User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Freshwater aquarium provides source for melioidosis infection
A Maryland woman came down with a severe tropical infection called melioidosis from her freshwater home aquarium, says a report in Emerging Infectious Diseases describing a new route of transmission. Melioidosis is caused by the bacteria Burkholderia pseudomallei in soil or water.
Until last year, almost all U.S. cases of melioidosis were from people who lived or traveled to disease-endemic areas. It has been a rare infection in the United States.
But this is not the first case of melioidosis from an unusual source. Earlier in 2021, CDC and state epidemiologists traced an outbreak of melioidosis in Georgia, Kansas, Minnesota, and Texas to B pseudomallei in a bottle of “Better Homes & Gardens Lavender & Chamomile Essential Oil Infused Aromatherapy Room Spray with Gemstones.”
In the aquarium case, the patient was a 56-year-old woman with diabetes and rheumatologic disease. She had been on immunosuppressives (methotrexate, azathioprine, and prednisone) until 1 month before she became symptomatic. She was hospitalized for fever and pneumonia.
Multiple blood cultures obtained on days 1-4 grew B. pseudomallei, but she had no evidence of endocarditis or intravascular seeding. Despite weeks of meropenem (Merrem), she developed evidence of a lung abscess, and trimethoprim/sulfamethoxazole (Bactrim) was added. Ultimately, the patient required a 12-week course of antibiotics.
CDC epidemiologist Patrick Dawson, PhD, first author of the report, told this news organization that although outbreak investigators always ask about pet ownership, they have not explicitly asked about fish. In this case, the patient did not volunteer exposure to the fish.
When state epidemiologists visited the patient’s home, “one of the first things they saw was a few aquariums,” Dr. Dawson said. Seeing the water and knowing “that most freshwater tropical fish in the U.S. are imported from Southeast Asia” led them to culture specifically for B. pseudomallei, which can be difficult for the microbiology lab to identify.
From there, Dr. Dawson explained, “The Maryland Department of Health sent a team to the local pet store” but did not find any of the bacteria there. (The patient had bought her fish 6 months earlier.) The investigators then worked with the national brand “to identify where they had actually sourced the fish from.”
Two retailers supply almost all of U.S. guppies and plants. While investigators could not find an exact matching isolate after so many months had elapsed, they found a positive PCR for B. pseudomallei in a water sample from imported fish in Los Angeles.
Dr. Dawson said tropical fish are imported from southeast Asia and typically come from small family fish farms. The fish import industry has “certain products that they add to the water to hopefully kill any bacteria.” He was unaware whether this included antibiotics but suggested, “we would have seen many more cases [of antibiotic resistance] by now” if it did.
In general advice for the public, Dr. Dawson said, “I would recommend washing hands before and after contact with the aquarium. If you have cuts or wounds on your hands, it’s really important to wear gloves if you have to go clean or maintain the aquarium and you’re putting your hands in the water, just for that extra layer of protection. It’s probably a strong idea to just avoid that altogether if someone’s immunocompromised. And not letting young children under 5 years old clean aquariums.” These are the “simplest things to do to protect yourself.”
Stephen A. Smith, DVM, PhD, a professor in the Aquatic Medicine Program at Virginia-Maryland College of Veterinary Medicine, Blacksburg, also stressed the importance of careful hand hygiene when caring for aquariums. He said that the filter, filter floss, biofilm, charcoal, and gravel might have exceptionally high concentrations of bacteria. Dr. Smith also recommended gloves when cleaning aquariums and not doing this task if immunocompromised.
Dr. Smith, who was not involved in the CDC study, shared a broader perspective, noting that “the reason why it’s important to federal regulators is that [B. pseudomallei] is a tier 1 select agent. And so, when that was isolated, it sent up all the red flags.” The far more common Mycobacterium marinum, or fish handler’s disease, is not reportable.
Mycobacterium marinum is another pathogen of concern that can be acquired from aquariums. These infections typically occur as nodular lesions on the arms and require months of therapy.
Dr. Smith stressed the importance of physicians eliciting a careful exposure history as the key to diagnosing zoonoses. For most exotic aquarium animals, he noted, “They’re caught in the wild wherever they are. They’re transported to a major hub to transport to the U.S., and a lot of times, we don’t have quarantine for those animals.”
Dr. Smith said.
Many infections also occur in the course of water sports – or even hiking and getting a cut or abrasion wet from a stream or lake. Aeromonas hydrophila can cause life-threatening infections. Vibrio vulnificus infections from salt-water injuries can cause sepsis and characteristic hemorrhagic bullae – large, discolored blisters filled with body fluid – during the summer. And eating contaminated shellfish has a 50%-60% death rate.
Other exposures to water-loving bacteria happen during fishing or cleaning/preparing fish. For example, Streptococcus iniae has caused cellulitis, arthritis, endocarditis, and meningitis following superficial or puncture injuries, notably from cleaning tilapia.
Other infections from contact with fish include Erysipelothrix rhusiopathiae (primarily skin infections) and gastroenteritis from Plesiomonas shigelloides, Campylobacter spp, and Salmonella spp.
Each of these zoonoses illustrates the importance of a careful exposure history when there’s an atypical presentation or an infection that is not responding promptly to empiric treatment. The aquarium case broadens the differential to include melioidosis, a serious disease from southeast Asia.
Dr. Dawson and Dr. Smith have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A Maryland woman came down with a severe tropical infection called melioidosis from her freshwater home aquarium, says a report in Emerging Infectious Diseases describing a new route of transmission. Melioidosis is caused by the bacteria Burkholderia pseudomallei in soil or water.
Until last year, almost all U.S. cases of melioidosis were from people who lived or traveled to disease-endemic areas. It has been a rare infection in the United States.
But this is not the first case of melioidosis from an unusual source. Earlier in 2021, CDC and state epidemiologists traced an outbreak of melioidosis in Georgia, Kansas, Minnesota, and Texas to B pseudomallei in a bottle of “Better Homes & Gardens Lavender & Chamomile Essential Oil Infused Aromatherapy Room Spray with Gemstones.”
In the aquarium case, the patient was a 56-year-old woman with diabetes and rheumatologic disease. She had been on immunosuppressives (methotrexate, azathioprine, and prednisone) until 1 month before she became symptomatic. She was hospitalized for fever and pneumonia.
Multiple blood cultures obtained on days 1-4 grew B. pseudomallei, but she had no evidence of endocarditis or intravascular seeding. Despite weeks of meropenem (Merrem), she developed evidence of a lung abscess, and trimethoprim/sulfamethoxazole (Bactrim) was added. Ultimately, the patient required a 12-week course of antibiotics.
CDC epidemiologist Patrick Dawson, PhD, first author of the report, told this news organization that although outbreak investigators always ask about pet ownership, they have not explicitly asked about fish. In this case, the patient did not volunteer exposure to the fish.
When state epidemiologists visited the patient’s home, “one of the first things they saw was a few aquariums,” Dr. Dawson said. Seeing the water and knowing “that most freshwater tropical fish in the U.S. are imported from Southeast Asia” led them to culture specifically for B. pseudomallei, which can be difficult for the microbiology lab to identify.
From there, Dr. Dawson explained, “The Maryland Department of Health sent a team to the local pet store” but did not find any of the bacteria there. (The patient had bought her fish 6 months earlier.) The investigators then worked with the national brand “to identify where they had actually sourced the fish from.”
Two retailers supply almost all of U.S. guppies and plants. While investigators could not find an exact matching isolate after so many months had elapsed, they found a positive PCR for B. pseudomallei in a water sample from imported fish in Los Angeles.
Dr. Dawson said tropical fish are imported from southeast Asia and typically come from small family fish farms. The fish import industry has “certain products that they add to the water to hopefully kill any bacteria.” He was unaware whether this included antibiotics but suggested, “we would have seen many more cases [of antibiotic resistance] by now” if it did.
In general advice for the public, Dr. Dawson said, “I would recommend washing hands before and after contact with the aquarium. If you have cuts or wounds on your hands, it’s really important to wear gloves if you have to go clean or maintain the aquarium and you’re putting your hands in the water, just for that extra layer of protection. It’s probably a strong idea to just avoid that altogether if someone’s immunocompromised. And not letting young children under 5 years old clean aquariums.” These are the “simplest things to do to protect yourself.”
Stephen A. Smith, DVM, PhD, a professor in the Aquatic Medicine Program at Virginia-Maryland College of Veterinary Medicine, Blacksburg, also stressed the importance of careful hand hygiene when caring for aquariums. He said that the filter, filter floss, biofilm, charcoal, and gravel might have exceptionally high concentrations of bacteria. Dr. Smith also recommended gloves when cleaning aquariums and not doing this task if immunocompromised.
Dr. Smith, who was not involved in the CDC study, shared a broader perspective, noting that “the reason why it’s important to federal regulators is that [B. pseudomallei] is a tier 1 select agent. And so, when that was isolated, it sent up all the red flags.” The far more common Mycobacterium marinum, or fish handler’s disease, is not reportable.
Mycobacterium marinum is another pathogen of concern that can be acquired from aquariums. These infections typically occur as nodular lesions on the arms and require months of therapy.
Dr. Smith stressed the importance of physicians eliciting a careful exposure history as the key to diagnosing zoonoses. For most exotic aquarium animals, he noted, “They’re caught in the wild wherever they are. They’re transported to a major hub to transport to the U.S., and a lot of times, we don’t have quarantine for those animals.”
Dr. Smith said.
Many infections also occur in the course of water sports – or even hiking and getting a cut or abrasion wet from a stream or lake. Aeromonas hydrophila can cause life-threatening infections. Vibrio vulnificus infections from salt-water injuries can cause sepsis and characteristic hemorrhagic bullae – large, discolored blisters filled with body fluid – during the summer. And eating contaminated shellfish has a 50%-60% death rate.
Other exposures to water-loving bacteria happen during fishing or cleaning/preparing fish. For example, Streptococcus iniae has caused cellulitis, arthritis, endocarditis, and meningitis following superficial or puncture injuries, notably from cleaning tilapia.
Other infections from contact with fish include Erysipelothrix rhusiopathiae (primarily skin infections) and gastroenteritis from Plesiomonas shigelloides, Campylobacter spp, and Salmonella spp.
Each of these zoonoses illustrates the importance of a careful exposure history when there’s an atypical presentation or an infection that is not responding promptly to empiric treatment. The aquarium case broadens the differential to include melioidosis, a serious disease from southeast Asia.
Dr. Dawson and Dr. Smith have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A Maryland woman came down with a severe tropical infection called melioidosis from her freshwater home aquarium, says a report in Emerging Infectious Diseases describing a new route of transmission. Melioidosis is caused by the bacteria Burkholderia pseudomallei in soil or water.
Until last year, almost all U.S. cases of melioidosis were from people who lived or traveled to disease-endemic areas. It has been a rare infection in the United States.
But this is not the first case of melioidosis from an unusual source. Earlier in 2021, CDC and state epidemiologists traced an outbreak of melioidosis in Georgia, Kansas, Minnesota, and Texas to B pseudomallei in a bottle of “Better Homes & Gardens Lavender & Chamomile Essential Oil Infused Aromatherapy Room Spray with Gemstones.”
In the aquarium case, the patient was a 56-year-old woman with diabetes and rheumatologic disease. She had been on immunosuppressives (methotrexate, azathioprine, and prednisone) until 1 month before she became symptomatic. She was hospitalized for fever and pneumonia.
Multiple blood cultures obtained on days 1-4 grew B. pseudomallei, but she had no evidence of endocarditis or intravascular seeding. Despite weeks of meropenem (Merrem), she developed evidence of a lung abscess, and trimethoprim/sulfamethoxazole (Bactrim) was added. Ultimately, the patient required a 12-week course of antibiotics.
CDC epidemiologist Patrick Dawson, PhD, first author of the report, told this news organization that although outbreak investigators always ask about pet ownership, they have not explicitly asked about fish. In this case, the patient did not volunteer exposure to the fish.
When state epidemiologists visited the patient’s home, “one of the first things they saw was a few aquariums,” Dr. Dawson said. Seeing the water and knowing “that most freshwater tropical fish in the U.S. are imported from Southeast Asia” led them to culture specifically for B. pseudomallei, which can be difficult for the microbiology lab to identify.
From there, Dr. Dawson explained, “The Maryland Department of Health sent a team to the local pet store” but did not find any of the bacteria there. (The patient had bought her fish 6 months earlier.) The investigators then worked with the national brand “to identify where they had actually sourced the fish from.”
Two retailers supply almost all of U.S. guppies and plants. While investigators could not find an exact matching isolate after so many months had elapsed, they found a positive PCR for B. pseudomallei in a water sample from imported fish in Los Angeles.
Dr. Dawson said tropical fish are imported from southeast Asia and typically come from small family fish farms. The fish import industry has “certain products that they add to the water to hopefully kill any bacteria.” He was unaware whether this included antibiotics but suggested, “we would have seen many more cases [of antibiotic resistance] by now” if it did.
In general advice for the public, Dr. Dawson said, “I would recommend washing hands before and after contact with the aquarium. If you have cuts or wounds on your hands, it’s really important to wear gloves if you have to go clean or maintain the aquarium and you’re putting your hands in the water, just for that extra layer of protection. It’s probably a strong idea to just avoid that altogether if someone’s immunocompromised. And not letting young children under 5 years old clean aquariums.” These are the “simplest things to do to protect yourself.”
Stephen A. Smith, DVM, PhD, a professor in the Aquatic Medicine Program at Virginia-Maryland College of Veterinary Medicine, Blacksburg, also stressed the importance of careful hand hygiene when caring for aquariums. He said that the filter, filter floss, biofilm, charcoal, and gravel might have exceptionally high concentrations of bacteria. Dr. Smith also recommended gloves when cleaning aquariums and not doing this task if immunocompromised.
Dr. Smith, who was not involved in the CDC study, shared a broader perspective, noting that “the reason why it’s important to federal regulators is that [B. pseudomallei] is a tier 1 select agent. And so, when that was isolated, it sent up all the red flags.” The far more common Mycobacterium marinum, or fish handler’s disease, is not reportable.
Mycobacterium marinum is another pathogen of concern that can be acquired from aquariums. These infections typically occur as nodular lesions on the arms and require months of therapy.
Dr. Smith stressed the importance of physicians eliciting a careful exposure history as the key to diagnosing zoonoses. For most exotic aquarium animals, he noted, “They’re caught in the wild wherever they are. They’re transported to a major hub to transport to the U.S., and a lot of times, we don’t have quarantine for those animals.”
Dr. Smith said.
Many infections also occur in the course of water sports – or even hiking and getting a cut or abrasion wet from a stream or lake. Aeromonas hydrophila can cause life-threatening infections. Vibrio vulnificus infections from salt-water injuries can cause sepsis and characteristic hemorrhagic bullae – large, discolored blisters filled with body fluid – during the summer. And eating contaminated shellfish has a 50%-60% death rate.
Other exposures to water-loving bacteria happen during fishing or cleaning/preparing fish. For example, Streptococcus iniae has caused cellulitis, arthritis, endocarditis, and meningitis following superficial or puncture injuries, notably from cleaning tilapia.
Other infections from contact with fish include Erysipelothrix rhusiopathiae (primarily skin infections) and gastroenteritis from Plesiomonas shigelloides, Campylobacter spp, and Salmonella spp.
Each of these zoonoses illustrates the importance of a careful exposure history when there’s an atypical presentation or an infection that is not responding promptly to empiric treatment. The aquarium case broadens the differential to include melioidosis, a serious disease from southeast Asia.
Dr. Dawson and Dr. Smith have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Frail COPD patients at high risk of disability and death
, a prospective cohort study of community-dwelling adults has shown.
“Frailty, a widely recognized geriatric syndrome characterized by multidimensional functional decline in bio-psycho-social factors, is associated with functional disability and mortality,” senior author Tze Pin Ng, MD, National University of Singapore, and colleagues explain.“Our results ... suggest that beyond traditional prognostic markers such as FEV1% (forced expiratory volume in 1 second) and dyspnea, the physical frailty phenotype provides additional useful prognostic information on future risks of disability and mortality,” the authors suggest.
The study was published online Dec. 12 in the journal CHEST®.
SLAS-1 and SLAS-2
Data from the Singapore Longitudinal Ageing Study (SLAS-1) and SLAS-2 were collected and analyzed. SLAS-1 recruited 2,804 participants 55 years of age and older from Sept. 2003 through Dec. 2004, while SLAS-2 recruited 3,270 participants of the same age between March 2009 and June 2013. “Follow-up visits and assessments were conducted approximately 3-5 years apart,” the investigators noted.
Mortality was determined at a mean of 9.5 years of follow-up for SLAS-1 participants and a mean of 6.5 years’ follow-up for SLAS-2 participants. A total of 4,627 participants were eventually included in the analysis, of whom 1,162 patients had COPD and 3,465 patients did not. COPD was classified as mild if FEV1% was greater than or equal to 80%; moderate if FEV1% was greater than or equal to 50% to less than 80%, and severe if FEV1% was less than 50%.
Frailty in turn was based on five clinical criteria, including weakness, slowness, low physical activity, exhaustion, and shrinking. Participants were classified as frail if they met three or more of these criteria and prefrail if they met one or two criteria.
Adverse health outcomes were judged on the basis of instrumental or basic activities of daily living (IADL/ADL), while disability was judged by self-reported difficulties in or requiring assistance with at least one IADL or ADL.
Frail or prefrail
Almost half of the participants were frail or prefrail, as the authors reported, while 25% had COPD. Among the participants with COPD, 30% had moderate to severe COPD, 6.4% had dyspnea, and almost half had prefrailty, while approximately 7% were classified as frail.
This percentage was 86% higher than it was for participants without COPD, among whom just 3.2% were assessed as frail, at an odds ratio of 1.86 (95% CI, 1.35-2.56). Further adjustments for possible confounders reduced the gap between frail COPD and frail non-COPD participants, but frailty remained significantly associated with COPD, at an OR of 1.61 (95% CI, 1.15-2.26), the investigators note.
Furthermore, compared to those without COPD, a diagnosis of COPD without and with dyspnea was associated with a 1.5- and 4.2-fold increase in prevalent frailty (95% CI, 1.04-2.08; 1.84-9.19), respectively, although not with prefrailty. Again, adjusting for multiple confounders, FEV1%, dyspnea, and both prefrailty and frailty were associated with an approximately twofold higher prevalence of IADL/ADL disability, while the prevalence of IADL/ADL disability for participants with COPD was approximately fourfold higher in those with co-occurring FEV1% less than 80% with either prefrailty, frailty, or dyspnea.
Furthermore, the presence of prefrailty or frailty in combination with a lower FEV1% or dyspnea was associated with a 3.7- to 3.8-fold increased risk of having an IADL or ADL disability.
Frailty and mortality
Some 1,116 participants with COPD were followed for a mean of 2,981 days for mortality outcomes. Both FEV1% less than 50% and the presence of prefrailty and frailty almost doubled the risk of mortality, at an adjusted hazard ratio of 1.8 (95% CI, 1.24-2.68) compared to patients with an FEV1% greater than or equal to 80%. In combination with either FEV1% less than 80% or prefrailty/frailty, dyspnea almost more than doubled the risk of mortality, at an HR of 2.4 for both combinations.
“However, the mortality risk of participants with COPD was highest among those with FEV1% less than 80% and prefrailty/frailty,” the authors note, more than tripling mortality risk at an adjusted HR of 3.25 (95% CI, 1.97-5.36). Interestingly, FEV1 less than 80% and prefrailty/frailty – both alone and in combination – were also associated with a twofold to fourfold increased risk of IADL or ADL disability in participants without COPD but were less strongly associated with mortality.
Researchers then went on to create a summary risk score containing all relevant variables with values ranging from 0 to 5. The highest risk category of 3 to 5 was associated with a 7- to 8.5-fold increased risk for IADL and ADL disability and mortality among participants with COPD, and that risk remained high after adjusting for multiple confounders.
Interestingly, frailty did not significantly predict mortality in women, while dyspnea did not significantly predict mortality in men. “Recognition and assessment of physical frailty in addition to FEV1% and dyspnea would allow for more accurate identification and targeted treatment of COPD at risk of future adverse outcomes,” the authors suggest.
Frailty scoring system
Asked to comment on the study, Sachin Gupta, MD, a pulmonologist and critical care specialist at Alameda Health System in Oakland, Calif., noted that the current study adds to the body of literature that outcomes in patients with COPD depend as much on objectively measured variables as on qualitative measures. “By applying a frailty scoring system, these researchers were able to categorize frailty and study its impact on patient characteristics and outcomes,” he told this news organization in an email.
The summary risk assessment tool developed and assessed is familiar: It carries parallels to the widely utilized BODE Index, replacing body mass index and 6-minute walk distance with the frailty scale, he added. “Findings from this study support the idea that what meets the eye in face-to-face visits – frailty – can be codified and be part of a tool that is predictive of outcomes,” Dr. Gupta underscored.
The authors had no conflicts of interest to declare. Dr. Gupta disclosed that he is also an employee and shareholder at Genentech.
A version of this article first appeared on Medscape.com.
, a prospective cohort study of community-dwelling adults has shown.
“Frailty, a widely recognized geriatric syndrome characterized by multidimensional functional decline in bio-psycho-social factors, is associated with functional disability and mortality,” senior author Tze Pin Ng, MD, National University of Singapore, and colleagues explain.“Our results ... suggest that beyond traditional prognostic markers such as FEV1% (forced expiratory volume in 1 second) and dyspnea, the physical frailty phenotype provides additional useful prognostic information on future risks of disability and mortality,” the authors suggest.
The study was published online Dec. 12 in the journal CHEST®.
SLAS-1 and SLAS-2
Data from the Singapore Longitudinal Ageing Study (SLAS-1) and SLAS-2 were collected and analyzed. SLAS-1 recruited 2,804 participants 55 years of age and older from Sept. 2003 through Dec. 2004, while SLAS-2 recruited 3,270 participants of the same age between March 2009 and June 2013. “Follow-up visits and assessments were conducted approximately 3-5 years apart,” the investigators noted.
Mortality was determined at a mean of 9.5 years of follow-up for SLAS-1 participants and a mean of 6.5 years’ follow-up for SLAS-2 participants. A total of 4,627 participants were eventually included in the analysis, of whom 1,162 patients had COPD and 3,465 patients did not. COPD was classified as mild if FEV1% was greater than or equal to 80%; moderate if FEV1% was greater than or equal to 50% to less than 80%, and severe if FEV1% was less than 50%.
Frailty in turn was based on five clinical criteria, including weakness, slowness, low physical activity, exhaustion, and shrinking. Participants were classified as frail if they met three or more of these criteria and prefrail if they met one or two criteria.
Adverse health outcomes were judged on the basis of instrumental or basic activities of daily living (IADL/ADL), while disability was judged by self-reported difficulties in or requiring assistance with at least one IADL or ADL.
Frail or prefrail
Almost half of the participants were frail or prefrail, as the authors reported, while 25% had COPD. Among the participants with COPD, 30% had moderate to severe COPD, 6.4% had dyspnea, and almost half had prefrailty, while approximately 7% were classified as frail.
This percentage was 86% higher than it was for participants without COPD, among whom just 3.2% were assessed as frail, at an odds ratio of 1.86 (95% CI, 1.35-2.56). Further adjustments for possible confounders reduced the gap between frail COPD and frail non-COPD participants, but frailty remained significantly associated with COPD, at an OR of 1.61 (95% CI, 1.15-2.26), the investigators note.
Furthermore, compared to those without COPD, a diagnosis of COPD without and with dyspnea was associated with a 1.5- and 4.2-fold increase in prevalent frailty (95% CI, 1.04-2.08; 1.84-9.19), respectively, although not with prefrailty. Again, adjusting for multiple confounders, FEV1%, dyspnea, and both prefrailty and frailty were associated with an approximately twofold higher prevalence of IADL/ADL disability, while the prevalence of IADL/ADL disability for participants with COPD was approximately fourfold higher in those with co-occurring FEV1% less than 80% with either prefrailty, frailty, or dyspnea.
Furthermore, the presence of prefrailty or frailty in combination with a lower FEV1% or dyspnea was associated with a 3.7- to 3.8-fold increased risk of having an IADL or ADL disability.
Frailty and mortality
Some 1,116 participants with COPD were followed for a mean of 2,981 days for mortality outcomes. Both FEV1% less than 50% and the presence of prefrailty and frailty almost doubled the risk of mortality, at an adjusted hazard ratio of 1.8 (95% CI, 1.24-2.68) compared to patients with an FEV1% greater than or equal to 80%. In combination with either FEV1% less than 80% or prefrailty/frailty, dyspnea almost more than doubled the risk of mortality, at an HR of 2.4 for both combinations.
“However, the mortality risk of participants with COPD was highest among those with FEV1% less than 80% and prefrailty/frailty,” the authors note, more than tripling mortality risk at an adjusted HR of 3.25 (95% CI, 1.97-5.36). Interestingly, FEV1 less than 80% and prefrailty/frailty – both alone and in combination – were also associated with a twofold to fourfold increased risk of IADL or ADL disability in participants without COPD but were less strongly associated with mortality.
Researchers then went on to create a summary risk score containing all relevant variables with values ranging from 0 to 5. The highest risk category of 3 to 5 was associated with a 7- to 8.5-fold increased risk for IADL and ADL disability and mortality among participants with COPD, and that risk remained high after adjusting for multiple confounders.
Interestingly, frailty did not significantly predict mortality in women, while dyspnea did not significantly predict mortality in men. “Recognition and assessment of physical frailty in addition to FEV1% and dyspnea would allow for more accurate identification and targeted treatment of COPD at risk of future adverse outcomes,” the authors suggest.
Frailty scoring system
Asked to comment on the study, Sachin Gupta, MD, a pulmonologist and critical care specialist at Alameda Health System in Oakland, Calif., noted that the current study adds to the body of literature that outcomes in patients with COPD depend as much on objectively measured variables as on qualitative measures. “By applying a frailty scoring system, these researchers were able to categorize frailty and study its impact on patient characteristics and outcomes,” he told this news organization in an email.
The summary risk assessment tool developed and assessed is familiar: It carries parallels to the widely utilized BODE Index, replacing body mass index and 6-minute walk distance with the frailty scale, he added. “Findings from this study support the idea that what meets the eye in face-to-face visits – frailty – can be codified and be part of a tool that is predictive of outcomes,” Dr. Gupta underscored.
The authors had no conflicts of interest to declare. Dr. Gupta disclosed that he is also an employee and shareholder at Genentech.
A version of this article first appeared on Medscape.com.
, a prospective cohort study of community-dwelling adults has shown.
“Frailty, a widely recognized geriatric syndrome characterized by multidimensional functional decline in bio-psycho-social factors, is associated with functional disability and mortality,” senior author Tze Pin Ng, MD, National University of Singapore, and colleagues explain.“Our results ... suggest that beyond traditional prognostic markers such as FEV1% (forced expiratory volume in 1 second) and dyspnea, the physical frailty phenotype provides additional useful prognostic information on future risks of disability and mortality,” the authors suggest.
The study was published online Dec. 12 in the journal CHEST®.
SLAS-1 and SLAS-2
Data from the Singapore Longitudinal Ageing Study (SLAS-1) and SLAS-2 were collected and analyzed. SLAS-1 recruited 2,804 participants 55 years of age and older from Sept. 2003 through Dec. 2004, while SLAS-2 recruited 3,270 participants of the same age between March 2009 and June 2013. “Follow-up visits and assessments were conducted approximately 3-5 years apart,” the investigators noted.
Mortality was determined at a mean of 9.5 years of follow-up for SLAS-1 participants and a mean of 6.5 years’ follow-up for SLAS-2 participants. A total of 4,627 participants were eventually included in the analysis, of whom 1,162 patients had COPD and 3,465 patients did not. COPD was classified as mild if FEV1% was greater than or equal to 80%; moderate if FEV1% was greater than or equal to 50% to less than 80%, and severe if FEV1% was less than 50%.
Frailty in turn was based on five clinical criteria, including weakness, slowness, low physical activity, exhaustion, and shrinking. Participants were classified as frail if they met three or more of these criteria and prefrail if they met one or two criteria.
Adverse health outcomes were judged on the basis of instrumental or basic activities of daily living (IADL/ADL), while disability was judged by self-reported difficulties in or requiring assistance with at least one IADL or ADL.
Frail or prefrail
Almost half of the participants were frail or prefrail, as the authors reported, while 25% had COPD. Among the participants with COPD, 30% had moderate to severe COPD, 6.4% had dyspnea, and almost half had prefrailty, while approximately 7% were classified as frail.
This percentage was 86% higher than it was for participants without COPD, among whom just 3.2% were assessed as frail, at an odds ratio of 1.86 (95% CI, 1.35-2.56). Further adjustments for possible confounders reduced the gap between frail COPD and frail non-COPD participants, but frailty remained significantly associated with COPD, at an OR of 1.61 (95% CI, 1.15-2.26), the investigators note.
Furthermore, compared to those without COPD, a diagnosis of COPD without and with dyspnea was associated with a 1.5- and 4.2-fold increase in prevalent frailty (95% CI, 1.04-2.08; 1.84-9.19), respectively, although not with prefrailty. Again, adjusting for multiple confounders, FEV1%, dyspnea, and both prefrailty and frailty were associated with an approximately twofold higher prevalence of IADL/ADL disability, while the prevalence of IADL/ADL disability for participants with COPD was approximately fourfold higher in those with co-occurring FEV1% less than 80% with either prefrailty, frailty, or dyspnea.
Furthermore, the presence of prefrailty or frailty in combination with a lower FEV1% or dyspnea was associated with a 3.7- to 3.8-fold increased risk of having an IADL or ADL disability.
Frailty and mortality
Some 1,116 participants with COPD were followed for a mean of 2,981 days for mortality outcomes. Both FEV1% less than 50% and the presence of prefrailty and frailty almost doubled the risk of mortality, at an adjusted hazard ratio of 1.8 (95% CI, 1.24-2.68) compared to patients with an FEV1% greater than or equal to 80%. In combination with either FEV1% less than 80% or prefrailty/frailty, dyspnea almost more than doubled the risk of mortality, at an HR of 2.4 for both combinations.
“However, the mortality risk of participants with COPD was highest among those with FEV1% less than 80% and prefrailty/frailty,” the authors note, more than tripling mortality risk at an adjusted HR of 3.25 (95% CI, 1.97-5.36). Interestingly, FEV1 less than 80% and prefrailty/frailty – both alone and in combination – were also associated with a twofold to fourfold increased risk of IADL or ADL disability in participants without COPD but were less strongly associated with mortality.
Researchers then went on to create a summary risk score containing all relevant variables with values ranging from 0 to 5. The highest risk category of 3 to 5 was associated with a 7- to 8.5-fold increased risk for IADL and ADL disability and mortality among participants with COPD, and that risk remained high after adjusting for multiple confounders.
Interestingly, frailty did not significantly predict mortality in women, while dyspnea did not significantly predict mortality in men. “Recognition and assessment of physical frailty in addition to FEV1% and dyspnea would allow for more accurate identification and targeted treatment of COPD at risk of future adverse outcomes,” the authors suggest.
Frailty scoring system
Asked to comment on the study, Sachin Gupta, MD, a pulmonologist and critical care specialist at Alameda Health System in Oakland, Calif., noted that the current study adds to the body of literature that outcomes in patients with COPD depend as much on objectively measured variables as on qualitative measures. “By applying a frailty scoring system, these researchers were able to categorize frailty and study its impact on patient characteristics and outcomes,” he told this news organization in an email.
The summary risk assessment tool developed and assessed is familiar: It carries parallels to the widely utilized BODE Index, replacing body mass index and 6-minute walk distance with the frailty scale, he added. “Findings from this study support the idea that what meets the eye in face-to-face visits – frailty – can be codified and be part of a tool that is predictive of outcomes,” Dr. Gupta underscored.
The authors had no conflicts of interest to declare. Dr. Gupta disclosed that he is also an employee and shareholder at Genentech.
A version of this article first appeared on Medscape.com.
FROM CHEST
COVID affects executive functioning in young to middle-age adults: Study
than people in the general population with no such infection, according to new data published on the preprint server medRxiv.
Researchers, led by Peter A. Hall, PhD, with the University of Waterloo (Ont.), found that COVID infection is associated with executive dysfunction among young and middle-aged adults, including for those not exposed to intubation or hospitalization.
The findings have not been peer reviewed.
The study included a representative cohort of 1,958 community-dwelling young and middle-aged adults. It used a balanced proportion of infected and uninfected people to estimate the link between SARS-CoV-2 infection and cognitive/executive dysfunction.
The authors noted that the survey was conducted from Sept. 28 to Oct. 21, 2021, when the primary variant in Canada was Delta.
The research was a cross-sectional observational study with data from the ongoing Canadian COVID-19 Experiences Survey. It included equal representation of vaccinated and vaccine-hesitant adults aged 18-54 years. COVID-19 symptoms ranged from negligible to life-threatening cases requiring hospitalization.
Half in the cohort (50.2%) received two vaccine shots; 43.3% had received no shots; and 5.5% received one shot, but were not intending to receive a second shot.
Dose-response relationship
According to the paper, those with prior COVID-19 infection, regardless of symptom severity, reported a significantly higher number of symptoms of executive dysfunction than their noninfected counterparts (mechanical adjustment, 1.63, standard error, 0.08; 95% confidence interval, 1.47-1.80; P = .001).
The researchers also found a dose-response relationship between COVID-19 symptom severity and cognitive dysfunction. Those with moderate and very/extremely severe COVID-19 symptoms were linked with significantly greater dysfunction.
“This reinforces what we’re hearing about – that COVID is not ‘one and done.’ It can have lasting and quite subtle and damaging effects on the human body,” William Schaffner, MD, infectious disease specialist with Vanderbilt University, Nashville, Tenn., said in an interview.
Measuring executive functioning – including the ability to make sound decisions – is something other studies haven’t typically addressed, he said.
Men were likely to report more cognitive dysfunction symptoms than women (beta, 0.15; P < .001). Younger adults (25-39 years) were more likely to experience cognitive dysfunction than those age 40-54 (beta, 0.30; P < .001).
Dr. Schaffner said it was troubling that young people are more likely to experience the dysfunction.
“When we think of ‘brain fog’ we think of older persons who are already predisposed to have more memory lapses as they get older,” he said.
The link between cognitive dysfunction and COVID-19 infection has been shown in other studies, but many have not used representative samples and have not compared results with noninfected controls in the general population, the authors wrote.
Executive dysfunction was measured using four questions from the Deficits in Executive Functioning Scale. Respondents were asked how often they have experienced these scenarios in the past 6 months:
- “I am unable to inhibit my reactions or responses to events or to other people.”
- “I make impulsive comments to others.”
- “I am likely to do things without considering the consequences for doing them.”
- “I act without thinking.”
“This makes it even more important that we talk about vaccination,” Dr. Schaffner said, “because clearly the more seriously ill you are, the more likely this sort of thing is likely to happen and vaccines have been shown time and again to avert hospitalizations and more serious illness. It also makes more important the monoclonal antibody treatments we have and the antivirals, which will prevent the evolution of mild disease into something more serious.”
This research was supported by a grant from the Canadian Institutes for Health Research, Institute for Population and Public Health. The study authors and Dr. Schaffner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
than people in the general population with no such infection, according to new data published on the preprint server medRxiv.
Researchers, led by Peter A. Hall, PhD, with the University of Waterloo (Ont.), found that COVID infection is associated with executive dysfunction among young and middle-aged adults, including for those not exposed to intubation or hospitalization.
The findings have not been peer reviewed.
The study included a representative cohort of 1,958 community-dwelling young and middle-aged adults. It used a balanced proportion of infected and uninfected people to estimate the link between SARS-CoV-2 infection and cognitive/executive dysfunction.
The authors noted that the survey was conducted from Sept. 28 to Oct. 21, 2021, when the primary variant in Canada was Delta.
The research was a cross-sectional observational study with data from the ongoing Canadian COVID-19 Experiences Survey. It included equal representation of vaccinated and vaccine-hesitant adults aged 18-54 years. COVID-19 symptoms ranged from negligible to life-threatening cases requiring hospitalization.
Half in the cohort (50.2%) received two vaccine shots; 43.3% had received no shots; and 5.5% received one shot, but were not intending to receive a second shot.
Dose-response relationship
According to the paper, those with prior COVID-19 infection, regardless of symptom severity, reported a significantly higher number of symptoms of executive dysfunction than their noninfected counterparts (mechanical adjustment, 1.63, standard error, 0.08; 95% confidence interval, 1.47-1.80; P = .001).
The researchers also found a dose-response relationship between COVID-19 symptom severity and cognitive dysfunction. Those with moderate and very/extremely severe COVID-19 symptoms were linked with significantly greater dysfunction.
“This reinforces what we’re hearing about – that COVID is not ‘one and done.’ It can have lasting and quite subtle and damaging effects on the human body,” William Schaffner, MD, infectious disease specialist with Vanderbilt University, Nashville, Tenn., said in an interview.
Measuring executive functioning – including the ability to make sound decisions – is something other studies haven’t typically addressed, he said.
Men were likely to report more cognitive dysfunction symptoms than women (beta, 0.15; P < .001). Younger adults (25-39 years) were more likely to experience cognitive dysfunction than those age 40-54 (beta, 0.30; P < .001).
Dr. Schaffner said it was troubling that young people are more likely to experience the dysfunction.
“When we think of ‘brain fog’ we think of older persons who are already predisposed to have more memory lapses as they get older,” he said.
The link between cognitive dysfunction and COVID-19 infection has been shown in other studies, but many have not used representative samples and have not compared results with noninfected controls in the general population, the authors wrote.
Executive dysfunction was measured using four questions from the Deficits in Executive Functioning Scale. Respondents were asked how often they have experienced these scenarios in the past 6 months:
- “I am unable to inhibit my reactions or responses to events or to other people.”
- “I make impulsive comments to others.”
- “I am likely to do things without considering the consequences for doing them.”
- “I act without thinking.”
“This makes it even more important that we talk about vaccination,” Dr. Schaffner said, “because clearly the more seriously ill you are, the more likely this sort of thing is likely to happen and vaccines have been shown time and again to avert hospitalizations and more serious illness. It also makes more important the monoclonal antibody treatments we have and the antivirals, which will prevent the evolution of mild disease into something more serious.”
This research was supported by a grant from the Canadian Institutes for Health Research, Institute for Population and Public Health. The study authors and Dr. Schaffner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
than people in the general population with no such infection, according to new data published on the preprint server medRxiv.
Researchers, led by Peter A. Hall, PhD, with the University of Waterloo (Ont.), found that COVID infection is associated with executive dysfunction among young and middle-aged adults, including for those not exposed to intubation or hospitalization.
The findings have not been peer reviewed.
The study included a representative cohort of 1,958 community-dwelling young and middle-aged adults. It used a balanced proportion of infected and uninfected people to estimate the link between SARS-CoV-2 infection and cognitive/executive dysfunction.
The authors noted that the survey was conducted from Sept. 28 to Oct. 21, 2021, when the primary variant in Canada was Delta.
The research was a cross-sectional observational study with data from the ongoing Canadian COVID-19 Experiences Survey. It included equal representation of vaccinated and vaccine-hesitant adults aged 18-54 years. COVID-19 symptoms ranged from negligible to life-threatening cases requiring hospitalization.
Half in the cohort (50.2%) received two vaccine shots; 43.3% had received no shots; and 5.5% received one shot, but were not intending to receive a second shot.
Dose-response relationship
According to the paper, those with prior COVID-19 infection, regardless of symptom severity, reported a significantly higher number of symptoms of executive dysfunction than their noninfected counterparts (mechanical adjustment, 1.63, standard error, 0.08; 95% confidence interval, 1.47-1.80; P = .001).
The researchers also found a dose-response relationship between COVID-19 symptom severity and cognitive dysfunction. Those with moderate and very/extremely severe COVID-19 symptoms were linked with significantly greater dysfunction.
“This reinforces what we’re hearing about – that COVID is not ‘one and done.’ It can have lasting and quite subtle and damaging effects on the human body,” William Schaffner, MD, infectious disease specialist with Vanderbilt University, Nashville, Tenn., said in an interview.
Measuring executive functioning – including the ability to make sound decisions – is something other studies haven’t typically addressed, he said.
Men were likely to report more cognitive dysfunction symptoms than women (beta, 0.15; P < .001). Younger adults (25-39 years) were more likely to experience cognitive dysfunction than those age 40-54 (beta, 0.30; P < .001).
Dr. Schaffner said it was troubling that young people are more likely to experience the dysfunction.
“When we think of ‘brain fog’ we think of older persons who are already predisposed to have more memory lapses as they get older,” he said.
The link between cognitive dysfunction and COVID-19 infection has been shown in other studies, but many have not used representative samples and have not compared results with noninfected controls in the general population, the authors wrote.
Executive dysfunction was measured using four questions from the Deficits in Executive Functioning Scale. Respondents were asked how often they have experienced these scenarios in the past 6 months:
- “I am unable to inhibit my reactions or responses to events or to other people.”
- “I make impulsive comments to others.”
- “I am likely to do things without considering the consequences for doing them.”
- “I act without thinking.”
“This makes it even more important that we talk about vaccination,” Dr. Schaffner said, “because clearly the more seriously ill you are, the more likely this sort of thing is likely to happen and vaccines have been shown time and again to avert hospitalizations and more serious illness. It also makes more important the monoclonal antibody treatments we have and the antivirals, which will prevent the evolution of mild disease into something more serious.”
This research was supported by a grant from the Canadian Institutes for Health Research, Institute for Population and Public Health. The study authors and Dr. Schaffner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM MEDRXIV
Surgeon General releases child mental health call to action
The nation’s Surgeon General, Vice Admiral Vivek H. Murthy, MD, MBA, recently released an advisory report on the current state of youth mental health and recommendations to improve well-being. This action follows a number of emergency declarations that have been made by professional organizations such as the American Academy of Child and Adolescent Psychiatry (AACAP), the American Academy of Pediatrics (AAP), and other health care groups to raise awareness about the alarming increase of depression, suicide, anxiety, and other mental health problems in youth.
These reports can be helpful in focusing attention and resources for important public health problems. Many still reference the 1999 report from former Surgeon General David Satcher, MD, PhD, which offered a number of eye-opening statistics regarding the prevalence of mental health conditions and the amount of disability associated with them.
Sadly, the present report indicates that many of these indices have grown worse in the past 20 years. For example, the advisory notes that, even before COVID-19, fully half of female high school students reported persistent feelings of sadness or hopelessness (up 40% from 2009). The report then goes on to cite a number of studies documenting even further rises in youth mental health problems associated with the pandemic.
Most of the advisory, however, is devoted to actions that can be taken by different groups, including young people themselves, parents, educators, the government, and even social media and video game companies, to support mental health and well-being. Multiple online resources are provided at the end of each of these sections.
One of the segments is aimed at health care organizations and professionals. While first making a fairly sweeping statement that “our health care system today is not set up optimally to support the mental health and well-being of children and youth,” this part then outlines five broad recommendations that might help improve the fit. These include the following.
- Increase prevention efforts, such as coordination to enrichment programs and referrals for economic and legal supports for families in need.
- Screen routinely for mental health conditions and link those who screen in with appropriate care.
- Identify mental health needs in parents and caregivers such as depression and substance use that can have negative effects on children.
- Increase partnerships between health care groups and community organizations.
- Build multidisciplinary teams that are culturally appropriate and maximally engage children and caretakers in the decision-making process.
The current report is downloadable for free (see reference below) and it is certainly worthwhile for pediatricians to take a look. Dr. Murthy writes, regarding the current state of mental health, that “it would be a tragedy if we beat back one public health crisis only to allow another to grow in its place.”
The report also outlines specific areas where additional research is needed, such as data on racial and sexual minorities and research on innovative and scalable therapies. In addition to the online resources that are provided, the report is backed by over 250 references.
Since its release, the report has generally been well received, and, indeed, there is much to support. The well-known Child Mind Institute in New York tweeted that “this document is a wake-up call for the country and a long-overdue statement of leadership from the federal government.”
Many of the recommendations are admittedly somewhat commons sense, but there are some that are much less so. For example, one recommendation to youth themselves is to serve others – something that may first come across as counterintuitive but can indeed help children and adolescents develop a sense of purpose and self-worth. The call for pediatric health care professionals to screen parents in addition to the patients themselves will likely result in some debate as well. The recommendation to reduce access to lethal means, including the specific naming of firearms, is also a welcome addition. This report also rightly puts a spotlight on the role of societal factors such as racism and poverty in the development of mental health problems and in getting access to quality treatment.
Also worth noting is how much of the advisory examined the role of media in both the problem and the solution. While recognizing that technology, smartphones, and social media are here to stay, a number of suggestions were given to parents, media organizations, journalists, and entertainment companies to reduce the negative impacts these mediums can have. Explicitly recognized in the report is that “there can be tension between what’s best for the technology company and what’s best for the individual user or society.” Also acknowledged was that the link between media of various types and mental health is complex and inconsistent with there being a strong need for additional work in this area when it comes to academic research as well as product development within these companies themselves.
Yet while there is much to like about the advisory, there remain some areas that seem lacking. For example, the text about what causes mental health conditions gets a little dualistic in mentioning biological and environmental factors without much appreciation that these are hardly independent domains. Perhaps more substantially, there was surprisingly little airtime devoted to an enormous issue that underlies so many other challenges related to mental health care – namely an inadequate workforce that gets smaller by the minute. The topic was treated much too superficially with lots of vague calls to “expand” the workforce that lacked substance or detail.
Overall, however, the new Surgeon General’s Advisory is a welcome document that offers updated knowledge of our current challenges and provides practical responses that truly could make a difference. Now all we have to do is put these recommendations into action.
Dr. Rettew is a child and adolescent psychiatrist and medical director of Lane County Behavioral Health in Eugene, Ore. His latest book is “Parenting Made Complicated: What Science Really Knows About the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
Reference
“Protecting Youth Mental Health – The U.S. Surgeon General’s Advisory,” U.S. Department of Health & Human Services (2021).
The nation’s Surgeon General, Vice Admiral Vivek H. Murthy, MD, MBA, recently released an advisory report on the current state of youth mental health and recommendations to improve well-being. This action follows a number of emergency declarations that have been made by professional organizations such as the American Academy of Child and Adolescent Psychiatry (AACAP), the American Academy of Pediatrics (AAP), and other health care groups to raise awareness about the alarming increase of depression, suicide, anxiety, and other mental health problems in youth.
These reports can be helpful in focusing attention and resources for important public health problems. Many still reference the 1999 report from former Surgeon General David Satcher, MD, PhD, which offered a number of eye-opening statistics regarding the prevalence of mental health conditions and the amount of disability associated with them.
Sadly, the present report indicates that many of these indices have grown worse in the past 20 years. For example, the advisory notes that, even before COVID-19, fully half of female high school students reported persistent feelings of sadness or hopelessness (up 40% from 2009). The report then goes on to cite a number of studies documenting even further rises in youth mental health problems associated with the pandemic.
Most of the advisory, however, is devoted to actions that can be taken by different groups, including young people themselves, parents, educators, the government, and even social media and video game companies, to support mental health and well-being. Multiple online resources are provided at the end of each of these sections.
One of the segments is aimed at health care organizations and professionals. While first making a fairly sweeping statement that “our health care system today is not set up optimally to support the mental health and well-being of children and youth,” this part then outlines five broad recommendations that might help improve the fit. These include the following.
- Increase prevention efforts, such as coordination to enrichment programs and referrals for economic and legal supports for families in need.
- Screen routinely for mental health conditions and link those who screen in with appropriate care.
- Identify mental health needs in parents and caregivers such as depression and substance use that can have negative effects on children.
- Increase partnerships between health care groups and community organizations.
- Build multidisciplinary teams that are culturally appropriate and maximally engage children and caretakers in the decision-making process.
The current report is downloadable for free (see reference below) and it is certainly worthwhile for pediatricians to take a look. Dr. Murthy writes, regarding the current state of mental health, that “it would be a tragedy if we beat back one public health crisis only to allow another to grow in its place.”
The report also outlines specific areas where additional research is needed, such as data on racial and sexual minorities and research on innovative and scalable therapies. In addition to the online resources that are provided, the report is backed by over 250 references.
Since its release, the report has generally been well received, and, indeed, there is much to support. The well-known Child Mind Institute in New York tweeted that “this document is a wake-up call for the country and a long-overdue statement of leadership from the federal government.”
Many of the recommendations are admittedly somewhat commons sense, but there are some that are much less so. For example, one recommendation to youth themselves is to serve others – something that may first come across as counterintuitive but can indeed help children and adolescents develop a sense of purpose and self-worth. The call for pediatric health care professionals to screen parents in addition to the patients themselves will likely result in some debate as well. The recommendation to reduce access to lethal means, including the specific naming of firearms, is also a welcome addition. This report also rightly puts a spotlight on the role of societal factors such as racism and poverty in the development of mental health problems and in getting access to quality treatment.
Also worth noting is how much of the advisory examined the role of media in both the problem and the solution. While recognizing that technology, smartphones, and social media are here to stay, a number of suggestions were given to parents, media organizations, journalists, and entertainment companies to reduce the negative impacts these mediums can have. Explicitly recognized in the report is that “there can be tension between what’s best for the technology company and what’s best for the individual user or society.” Also acknowledged was that the link between media of various types and mental health is complex and inconsistent with there being a strong need for additional work in this area when it comes to academic research as well as product development within these companies themselves.
Yet while there is much to like about the advisory, there remain some areas that seem lacking. For example, the text about what causes mental health conditions gets a little dualistic in mentioning biological and environmental factors without much appreciation that these are hardly independent domains. Perhaps more substantially, there was surprisingly little airtime devoted to an enormous issue that underlies so many other challenges related to mental health care – namely an inadequate workforce that gets smaller by the minute. The topic was treated much too superficially with lots of vague calls to “expand” the workforce that lacked substance or detail.
Overall, however, the new Surgeon General’s Advisory is a welcome document that offers updated knowledge of our current challenges and provides practical responses that truly could make a difference. Now all we have to do is put these recommendations into action.
Dr. Rettew is a child and adolescent psychiatrist and medical director of Lane County Behavioral Health in Eugene, Ore. His latest book is “Parenting Made Complicated: What Science Really Knows About the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
Reference
“Protecting Youth Mental Health – The U.S. Surgeon General’s Advisory,” U.S. Department of Health & Human Services (2021).
The nation’s Surgeon General, Vice Admiral Vivek H. Murthy, MD, MBA, recently released an advisory report on the current state of youth mental health and recommendations to improve well-being. This action follows a number of emergency declarations that have been made by professional organizations such as the American Academy of Child and Adolescent Psychiatry (AACAP), the American Academy of Pediatrics (AAP), and other health care groups to raise awareness about the alarming increase of depression, suicide, anxiety, and other mental health problems in youth.
These reports can be helpful in focusing attention and resources for important public health problems. Many still reference the 1999 report from former Surgeon General David Satcher, MD, PhD, which offered a number of eye-opening statistics regarding the prevalence of mental health conditions and the amount of disability associated with them.
Sadly, the present report indicates that many of these indices have grown worse in the past 20 years. For example, the advisory notes that, even before COVID-19, fully half of female high school students reported persistent feelings of sadness or hopelessness (up 40% from 2009). The report then goes on to cite a number of studies documenting even further rises in youth mental health problems associated with the pandemic.
Most of the advisory, however, is devoted to actions that can be taken by different groups, including young people themselves, parents, educators, the government, and even social media and video game companies, to support mental health and well-being. Multiple online resources are provided at the end of each of these sections.
One of the segments is aimed at health care organizations and professionals. While first making a fairly sweeping statement that “our health care system today is not set up optimally to support the mental health and well-being of children and youth,” this part then outlines five broad recommendations that might help improve the fit. These include the following.
- Increase prevention efforts, such as coordination to enrichment programs and referrals for economic and legal supports for families in need.
- Screen routinely for mental health conditions and link those who screen in with appropriate care.
- Identify mental health needs in parents and caregivers such as depression and substance use that can have negative effects on children.
- Increase partnerships between health care groups and community organizations.
- Build multidisciplinary teams that are culturally appropriate and maximally engage children and caretakers in the decision-making process.
The current report is downloadable for free (see reference below) and it is certainly worthwhile for pediatricians to take a look. Dr. Murthy writes, regarding the current state of mental health, that “it would be a tragedy if we beat back one public health crisis only to allow another to grow in its place.”
The report also outlines specific areas where additional research is needed, such as data on racial and sexual minorities and research on innovative and scalable therapies. In addition to the online resources that are provided, the report is backed by over 250 references.
Since its release, the report has generally been well received, and, indeed, there is much to support. The well-known Child Mind Institute in New York tweeted that “this document is a wake-up call for the country and a long-overdue statement of leadership from the federal government.”
Many of the recommendations are admittedly somewhat commons sense, but there are some that are much less so. For example, one recommendation to youth themselves is to serve others – something that may first come across as counterintuitive but can indeed help children and adolescents develop a sense of purpose and self-worth. The call for pediatric health care professionals to screen parents in addition to the patients themselves will likely result in some debate as well. The recommendation to reduce access to lethal means, including the specific naming of firearms, is also a welcome addition. This report also rightly puts a spotlight on the role of societal factors such as racism and poverty in the development of mental health problems and in getting access to quality treatment.
Also worth noting is how much of the advisory examined the role of media in both the problem and the solution. While recognizing that technology, smartphones, and social media are here to stay, a number of suggestions were given to parents, media organizations, journalists, and entertainment companies to reduce the negative impacts these mediums can have. Explicitly recognized in the report is that “there can be tension between what’s best for the technology company and what’s best for the individual user or society.” Also acknowledged was that the link between media of various types and mental health is complex and inconsistent with there being a strong need for additional work in this area when it comes to academic research as well as product development within these companies themselves.
Yet while there is much to like about the advisory, there remain some areas that seem lacking. For example, the text about what causes mental health conditions gets a little dualistic in mentioning biological and environmental factors without much appreciation that these are hardly independent domains. Perhaps more substantially, there was surprisingly little airtime devoted to an enormous issue that underlies so many other challenges related to mental health care – namely an inadequate workforce that gets smaller by the minute. The topic was treated much too superficially with lots of vague calls to “expand” the workforce that lacked substance or detail.
Overall, however, the new Surgeon General’s Advisory is a welcome document that offers updated knowledge of our current challenges and provides practical responses that truly could make a difference. Now all we have to do is put these recommendations into action.
Dr. Rettew is a child and adolescent psychiatrist and medical director of Lane County Behavioral Health in Eugene, Ore. His latest book is “Parenting Made Complicated: What Science Really Knows About the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
Reference
“Protecting Youth Mental Health – The U.S. Surgeon General’s Advisory,” U.S. Department of Health & Human Services (2021).
Children and COVID: New cases, admissions are higher than ever
Weekly COVID-19 cases in children passed 300,000 for the first time since the pandemic started, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The rate of new COVID-related hospital admissions also reached a new high of 0.74 per 100,000 children as of Dec. 31. The highest rate seen before the current Omicron-fueled surge was 0.47 per 100,000 in early September, data from the Centers for Disease Control and Prevention show.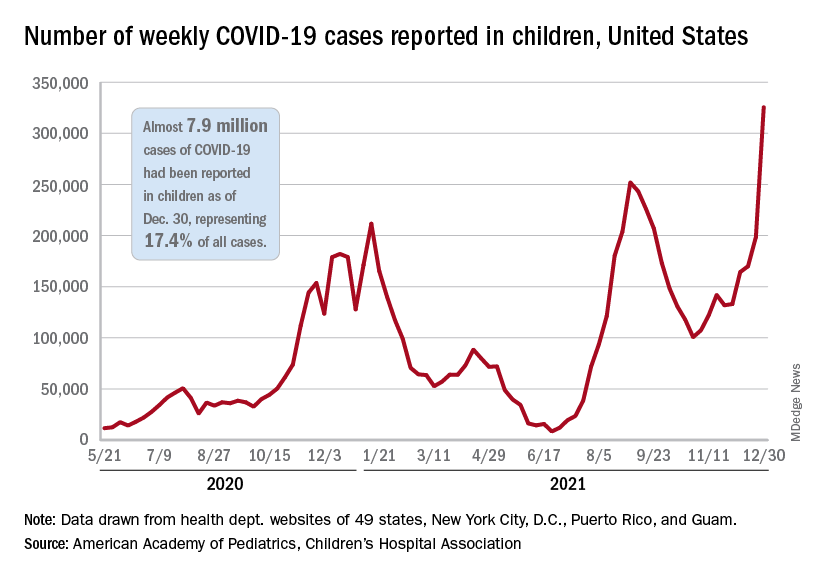
and exceeding the previous week’s count by almost 64%, the AAP and CHA said in their weekly COVID report.
New cases were up in all four regions of the United States, with the Northeast adding the most newly infected children while setting a new high for the fifth consecutive week. The South was just behind for the week but still well off the record it reached in September, the Midwest was third but recorded its busiest week ever, and the West was fourth and nowhere near its previous high, the AAP/CHA report indicated.
The total number of child cases since the pandemic began is almost 7.9 million, they said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. That figure represents 17.4% of all cases reported in the United States, and the cumulative rate of COVID infection is up to almost 10,500 per 100,000 children, meaning that 1 in 10 children have been infected.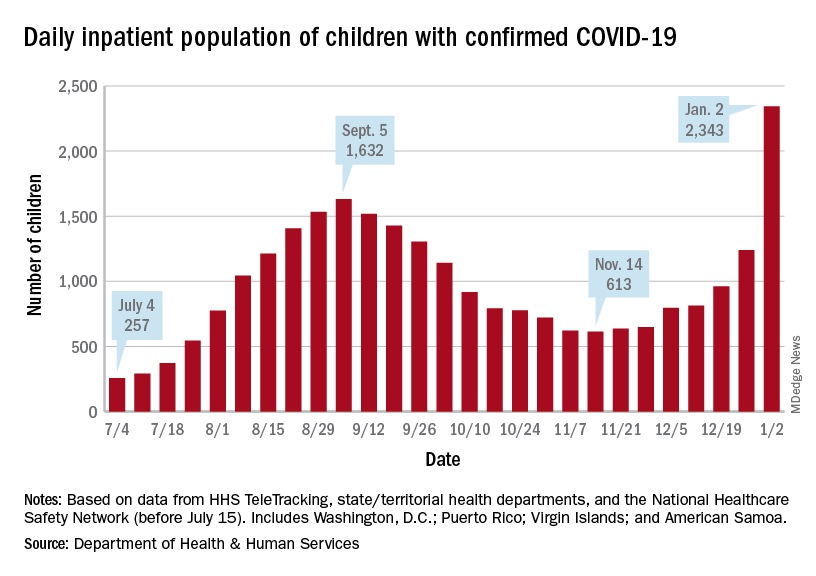
Children are still less likely to be hospitalized than adults, but the gap appears to be closing. On Jan. 2 there were 2,343 children and 87,690 adults in the hospital with confirmed COVID, a ratio of 37 adults for each child, but on Sept. 5, at the height of the previous surge, the ratio of hospitalized adults (93,647) to children (1,632) was 57:1, according to data from the Department of Health & Human Services.
New admissions show a similar pattern: The 0.74 admissions per 100,000 children recorded on Dec. 31 was lower than, for example, adults aged 30-39 years (2.7 per 100,000) or 50-59 years (4.25 per 100,000), but on Sept. 5 the corresponding figures were 0.46 (children), 2.74 (ages 30-39), and 5.03 (aged 50-59), based on the HHS data.
A look at vaccinations
The vaccination response to Omicron, however, has been more subdued and somewhat inconsistent. Vaccine initiation, not surprisingly, was down among eligible children for the week of Dec. 23-29. Before that, both the 5- to 11-year-olds and 12- to 15-year-olds were down for the second week of December and then up a bit (5.6% and 14.3%, respectively) during the third week, while the 16- to 17-year-olds, increased initiation by 63.2%, CDC’s COVID Data Tracker shows.
Less than a quarter (23.5%) of children aged 5-11 received at least one dose of the vaccine in the first 2 months of their eligibility, and only 14.7% are fully vaccinated. Among the older children, coverage looks like this: at least one dose for 61.2% of 12- to 15-year-olds and 67.4% of 16- to 17-year-olds and full vaccination for 51.3% and 57.6%, respectively, the CDC said.
At the state level, Massachusetts and Hawaii have the highest rates for children aged 12-17 years, with 86% having received a least one dose, and Vermont is highest for children aged 5-11 at 56%. The lowest rates can be found in Wyoming (38%) for 12- to 17-year-olds and in Mississippi (6%) for 5- to 11-year-olds, the AAP said in a separate report.
Weekly COVID-19 cases in children passed 300,000 for the first time since the pandemic started, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The rate of new COVID-related hospital admissions also reached a new high of 0.74 per 100,000 children as of Dec. 31. The highest rate seen before the current Omicron-fueled surge was 0.47 per 100,000 in early September, data from the Centers for Disease Control and Prevention show.
and exceeding the previous week’s count by almost 64%, the AAP and CHA said in their weekly COVID report.
New cases were up in all four regions of the United States, with the Northeast adding the most newly infected children while setting a new high for the fifth consecutive week. The South was just behind for the week but still well off the record it reached in September, the Midwest was third but recorded its busiest week ever, and the West was fourth and nowhere near its previous high, the AAP/CHA report indicated.
The total number of child cases since the pandemic began is almost 7.9 million, they said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. That figure represents 17.4% of all cases reported in the United States, and the cumulative rate of COVID infection is up to almost 10,500 per 100,000 children, meaning that 1 in 10 children have been infected.
Children are still less likely to be hospitalized than adults, but the gap appears to be closing. On Jan. 2 there were 2,343 children and 87,690 adults in the hospital with confirmed COVID, a ratio of 37 adults for each child, but on Sept. 5, at the height of the previous surge, the ratio of hospitalized adults (93,647) to children (1,632) was 57:1, according to data from the Department of Health & Human Services.
New admissions show a similar pattern: The 0.74 admissions per 100,000 children recorded on Dec. 31 was lower than, for example, adults aged 30-39 years (2.7 per 100,000) or 50-59 years (4.25 per 100,000), but on Sept. 5 the corresponding figures were 0.46 (children), 2.74 (ages 30-39), and 5.03 (aged 50-59), based on the HHS data.
A look at vaccinations
The vaccination response to Omicron, however, has been more subdued and somewhat inconsistent. Vaccine initiation, not surprisingly, was down among eligible children for the week of Dec. 23-29. Before that, both the 5- to 11-year-olds and 12- to 15-year-olds were down for the second week of December and then up a bit (5.6% and 14.3%, respectively) during the third week, while the 16- to 17-year-olds, increased initiation by 63.2%, CDC’s COVID Data Tracker shows.
Less than a quarter (23.5%) of children aged 5-11 received at least one dose of the vaccine in the first 2 months of their eligibility, and only 14.7% are fully vaccinated. Among the older children, coverage looks like this: at least one dose for 61.2% of 12- to 15-year-olds and 67.4% of 16- to 17-year-olds and full vaccination for 51.3% and 57.6%, respectively, the CDC said.
At the state level, Massachusetts and Hawaii have the highest rates for children aged 12-17 years, with 86% having received a least one dose, and Vermont is highest for children aged 5-11 at 56%. The lowest rates can be found in Wyoming (38%) for 12- to 17-year-olds and in Mississippi (6%) for 5- to 11-year-olds, the AAP said in a separate report.
Weekly COVID-19 cases in children passed 300,000 for the first time since the pandemic started, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The rate of new COVID-related hospital admissions also reached a new high of 0.74 per 100,000 children as of Dec. 31. The highest rate seen before the current Omicron-fueled surge was 0.47 per 100,000 in early September, data from the Centers for Disease Control and Prevention show.
and exceeding the previous week’s count by almost 64%, the AAP and CHA said in their weekly COVID report.
New cases were up in all four regions of the United States, with the Northeast adding the most newly infected children while setting a new high for the fifth consecutive week. The South was just behind for the week but still well off the record it reached in September, the Midwest was third but recorded its busiest week ever, and the West was fourth and nowhere near its previous high, the AAP/CHA report indicated.
The total number of child cases since the pandemic began is almost 7.9 million, they said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. That figure represents 17.4% of all cases reported in the United States, and the cumulative rate of COVID infection is up to almost 10,500 per 100,000 children, meaning that 1 in 10 children have been infected.
Children are still less likely to be hospitalized than adults, but the gap appears to be closing. On Jan. 2 there were 2,343 children and 87,690 adults in the hospital with confirmed COVID, a ratio of 37 adults for each child, but on Sept. 5, at the height of the previous surge, the ratio of hospitalized adults (93,647) to children (1,632) was 57:1, according to data from the Department of Health & Human Services.
New admissions show a similar pattern: The 0.74 admissions per 100,000 children recorded on Dec. 31 was lower than, for example, adults aged 30-39 years (2.7 per 100,000) or 50-59 years (4.25 per 100,000), but on Sept. 5 the corresponding figures were 0.46 (children), 2.74 (ages 30-39), and 5.03 (aged 50-59), based on the HHS data.
A look at vaccinations
The vaccination response to Omicron, however, has been more subdued and somewhat inconsistent. Vaccine initiation, not surprisingly, was down among eligible children for the week of Dec. 23-29. Before that, both the 5- to 11-year-olds and 12- to 15-year-olds were down for the second week of December and then up a bit (5.6% and 14.3%, respectively) during the third week, while the 16- to 17-year-olds, increased initiation by 63.2%, CDC’s COVID Data Tracker shows.
Less than a quarter (23.5%) of children aged 5-11 received at least one dose of the vaccine in the first 2 months of their eligibility, and only 14.7% are fully vaccinated. Among the older children, coverage looks like this: at least one dose for 61.2% of 12- to 15-year-olds and 67.4% of 16- to 17-year-olds and full vaccination for 51.3% and 57.6%, respectively, the CDC said.
At the state level, Massachusetts and Hawaii have the highest rates for children aged 12-17 years, with 86% having received a least one dose, and Vermont is highest for children aged 5-11 at 56%. The lowest rates can be found in Wyoming (38%) for 12- to 17-year-olds and in Mississippi (6%) for 5- to 11-year-olds, the AAP said in a separate report.
New CDC COVID-19 isolation guidelines still up for debate among experts
It’s a true Goldilocks debate: , with some calling them suitable, some saying they’re “reckless,” and at least one expert saying they’re “right in the middle.”
The controversy may lead to more updates. On Jan. 2, Anthony S. Fauci, MD, President Joe Biden’s chief medical adviser, said on CNN’s State of the Union that he anticipates further clarification of the guidelines soon.
Sparking the most debate: Infected people are not told to test before leaving isolation, the vaccinated and unvaccinated who are exposed are given some of the same advice, and the mask advice is not specific enough.
As issued on Dec. 27, the guidelines for the general public recommend:
- Anyone who tests positive should stay home and isolate for 5 days (instead of 10) and if the person has no symptoms or the symptoms resolve after 5 days, leaving the house is okay. A mask should be worn around others for 5 more days. In the event of a fever, the person must stay home until it resolves.
- If people are exposed to someone infected with COVID-19 and they have been boosted, finished the primary series of either the Pfizer or Moderna vaccine within the past 6 months, or finished the primary series of the Johnson & Johnson vaccine within the past 2 months, they should wear a mask around others for 10 days and, if possible, test on day 5. However, if symptoms develop, they should get a test and stay home.
- If people are exposed to someone infected with COVID-19 and they are unvaccinated or are more than 6 months out from their second dose of the Pfizer or Moderna vaccine (or more than 2 months after the J&J vaccine) and not boosted, they should quarantine for 5 days and then wear a mask for 5 more days. If quarantine is impossible, a mask should be worn for 10 days. A test on day 5 is suggested if possible. If symptoms occur, they should quarantine and test.
On social media and in interviews with this news organization, public health experts expressed an array of opinions.
A tweet from Eric Topol, MD, editor-in-chief of Medscape, posted the day after the new guidelines came out, had an empty box and this: “The data that support the new @CDCgov 5 day isolation period without a negative test.”
In a tweet on Jan. 2, Ashish K. Jha, MD, MPH, dean of the Brown University School of Public Health, said: “Hearing that CDC considering adding testing to isolation guidelines. That would be great. I’ve been arguing for a while that serial negative antigen tests provide a lot of confidence that someone is not contagious.”
Michael Mina, MD, PhD, chief science officer of eMed, a digital point-of-care platform enabling at-home diagnostic testing, tweeted: “CDC’s new guidance to drop isolation of positives to 5 days without a negative test is reckless. Some [people] stay infectious 3 days, some 12. I absolutely don’t want to sit next to someone who turned [positive] 5 days ago and hasn’t tested Neg. Test Neg to leave isolation early is just smart.”
Paul Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia and an infectious disease specialist, disagrees. Typically, he said, an infected person sheds virus for 7 days.
“If you are asymptomatic, the chances that you are shedding a significant amount of virus is very, very small,” he said in an interview.
Under debate
Testing: While many public health experts say a recommendation to test before leaving isolation is needed, CDC Director Rochelle Walensky, MD, explained testing was not recommended before leaving isolation because PCR testing can stay positive up to 12 weeks after a person is first infected with COVID-19.
Asked why there was not a recommendation for a rapid antigen test before leaving isolation, Dr. Walensky told CNN that it is not known how these tests perform at the end of infection and that the tests are not Food and Drug Administration–authorized for that purpose.
And while the guidelines suggest that those exposed – whether they are boosted, vaccinated, or not – should test on day 5 if possible, that recommendation should be stronger, some said. “At the very least recommend a test in those who can get it done,” said Dr. Topol.
However, making that recommendation is difficult when experts know how difficult it is for people to obtain tests now, William Schaffner, MD, professor of preventive medicine and an infectious disease specialist at Vanderbilt University, Nashville, Tenn., said in an interview.
“I am sure this was intensely debated,” Dr. Schaffner said of the recommendation on testing.
Vaccination status categories: Amesh Adalja, MD, senior scholar at the Johns Hopkins Center for Health Security, Baltimore, questioned the scientific basis behind treating the fully vaccinated (with two mRNA or one J&J vaccine) who are exposed ‘’as the equivalent of the unvaccinated when it comes to the quarantine requirement since the fully vaccinated are protected against what matters.”
Dr. Topol agreed: Guidelines “should be different for vaccinated versus unvaccinated.”
The recommendations for the exposed should definitely be simpler, Dr. Offit said. “I think it would be much simpler to just say, ‘If you are exposed, mask for 10 days,’ “ regardless of vaccination status.
Masks: The guidelines should also be more specific about the type of masks, Dr. Topol said. They should spell out that the masks need to be N95 or KN95, he said.
Science-driven or economy-driven? Was the guidance changed due more to concerns about the economy than to scientific information about infection and transmission? “It was,” Dr. Topol said.
Dr. Adalja sees it differently. “While it is true that this updated guidance will help the economy, it is based on a scientific foundation and should have been issued much earlier than it was.”
Tough decisions
The agency is walking a tightrope, Dr. Schaffner said, adding that he is in general agreement with what the CDC is trying to do. “The tightrope is between the public health ideal and trying to determine what will be acceptable,’’ he said.
The revised guidelines are more practical than before, others said. “The goal is harm reduction and many people just don’t do any isolation if they are faced with a 10-day period,” Dr. Adalja said.
Before issuing the new guidance, the CDC looked at the accumulating science and also took into account stresses on the health care system and other factors, Dr. Schaffner said. “Is it perfect?” Dr. Schaffner said of the new guideline. “No. Is it carefree? No. It’s right in the middle.”
Dr. Schaffner does think the messages about the new recommendations and how they were decided upon could have been communicated better, and in a more understandable manner. Some experts, for instance, led with the economy and the need for people to return to work and school when explaining the guidelines and then brought up the science behind the revisions.
That order should have been reversed, Dr. Schaffner said.
A version of this article first appeared on Medscape.com.
It’s a true Goldilocks debate: , with some calling them suitable, some saying they’re “reckless,” and at least one expert saying they’re “right in the middle.”
The controversy may lead to more updates. On Jan. 2, Anthony S. Fauci, MD, President Joe Biden’s chief medical adviser, said on CNN’s State of the Union that he anticipates further clarification of the guidelines soon.
Sparking the most debate: Infected people are not told to test before leaving isolation, the vaccinated and unvaccinated who are exposed are given some of the same advice, and the mask advice is not specific enough.
As issued on Dec. 27, the guidelines for the general public recommend:
- Anyone who tests positive should stay home and isolate for 5 days (instead of 10) and if the person has no symptoms or the symptoms resolve after 5 days, leaving the house is okay. A mask should be worn around others for 5 more days. In the event of a fever, the person must stay home until it resolves.
- If people are exposed to someone infected with COVID-19 and they have been boosted, finished the primary series of either the Pfizer or Moderna vaccine within the past 6 months, or finished the primary series of the Johnson & Johnson vaccine within the past 2 months, they should wear a mask around others for 10 days and, if possible, test on day 5. However, if symptoms develop, they should get a test and stay home.
- If people are exposed to someone infected with COVID-19 and they are unvaccinated or are more than 6 months out from their second dose of the Pfizer or Moderna vaccine (or more than 2 months after the J&J vaccine) and not boosted, they should quarantine for 5 days and then wear a mask for 5 more days. If quarantine is impossible, a mask should be worn for 10 days. A test on day 5 is suggested if possible. If symptoms occur, they should quarantine and test.
On social media and in interviews with this news organization, public health experts expressed an array of opinions.
A tweet from Eric Topol, MD, editor-in-chief of Medscape, posted the day after the new guidelines came out, had an empty box and this: “The data that support the new @CDCgov 5 day isolation period without a negative test.”
In a tweet on Jan. 2, Ashish K. Jha, MD, MPH, dean of the Brown University School of Public Health, said: “Hearing that CDC considering adding testing to isolation guidelines. That would be great. I’ve been arguing for a while that serial negative antigen tests provide a lot of confidence that someone is not contagious.”
Michael Mina, MD, PhD, chief science officer of eMed, a digital point-of-care platform enabling at-home diagnostic testing, tweeted: “CDC’s new guidance to drop isolation of positives to 5 days without a negative test is reckless. Some [people] stay infectious 3 days, some 12. I absolutely don’t want to sit next to someone who turned [positive] 5 days ago and hasn’t tested Neg. Test Neg to leave isolation early is just smart.”
Paul Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia and an infectious disease specialist, disagrees. Typically, he said, an infected person sheds virus for 7 days.
“If you are asymptomatic, the chances that you are shedding a significant amount of virus is very, very small,” he said in an interview.
Under debate
Testing: While many public health experts say a recommendation to test before leaving isolation is needed, CDC Director Rochelle Walensky, MD, explained testing was not recommended before leaving isolation because PCR testing can stay positive up to 12 weeks after a person is first infected with COVID-19.
Asked why there was not a recommendation for a rapid antigen test before leaving isolation, Dr. Walensky told CNN that it is not known how these tests perform at the end of infection and that the tests are not Food and Drug Administration–authorized for that purpose.
And while the guidelines suggest that those exposed – whether they are boosted, vaccinated, or not – should test on day 5 if possible, that recommendation should be stronger, some said. “At the very least recommend a test in those who can get it done,” said Dr. Topol.
However, making that recommendation is difficult when experts know how difficult it is for people to obtain tests now, William Schaffner, MD, professor of preventive medicine and an infectious disease specialist at Vanderbilt University, Nashville, Tenn., said in an interview.
“I am sure this was intensely debated,” Dr. Schaffner said of the recommendation on testing.
Vaccination status categories: Amesh Adalja, MD, senior scholar at the Johns Hopkins Center for Health Security, Baltimore, questioned the scientific basis behind treating the fully vaccinated (with two mRNA or one J&J vaccine) who are exposed ‘’as the equivalent of the unvaccinated when it comes to the quarantine requirement since the fully vaccinated are protected against what matters.”
Dr. Topol agreed: Guidelines “should be different for vaccinated versus unvaccinated.”
The recommendations for the exposed should definitely be simpler, Dr. Offit said. “I think it would be much simpler to just say, ‘If you are exposed, mask for 10 days,’ “ regardless of vaccination status.
Masks: The guidelines should also be more specific about the type of masks, Dr. Topol said. They should spell out that the masks need to be N95 or KN95, he said.
Science-driven or economy-driven? Was the guidance changed due more to concerns about the economy than to scientific information about infection and transmission? “It was,” Dr. Topol said.
Dr. Adalja sees it differently. “While it is true that this updated guidance will help the economy, it is based on a scientific foundation and should have been issued much earlier than it was.”
Tough decisions
The agency is walking a tightrope, Dr. Schaffner said, adding that he is in general agreement with what the CDC is trying to do. “The tightrope is between the public health ideal and trying to determine what will be acceptable,’’ he said.
The revised guidelines are more practical than before, others said. “The goal is harm reduction and many people just don’t do any isolation if they are faced with a 10-day period,” Dr. Adalja said.
Before issuing the new guidance, the CDC looked at the accumulating science and also took into account stresses on the health care system and other factors, Dr. Schaffner said. “Is it perfect?” Dr. Schaffner said of the new guideline. “No. Is it carefree? No. It’s right in the middle.”
Dr. Schaffner does think the messages about the new recommendations and how they were decided upon could have been communicated better, and in a more understandable manner. Some experts, for instance, led with the economy and the need for people to return to work and school when explaining the guidelines and then brought up the science behind the revisions.
That order should have been reversed, Dr. Schaffner said.
A version of this article first appeared on Medscape.com.
It’s a true Goldilocks debate: , with some calling them suitable, some saying they’re “reckless,” and at least one expert saying they’re “right in the middle.”
The controversy may lead to more updates. On Jan. 2, Anthony S. Fauci, MD, President Joe Biden’s chief medical adviser, said on CNN’s State of the Union that he anticipates further clarification of the guidelines soon.
Sparking the most debate: Infected people are not told to test before leaving isolation, the vaccinated and unvaccinated who are exposed are given some of the same advice, and the mask advice is not specific enough.
As issued on Dec. 27, the guidelines for the general public recommend:
- Anyone who tests positive should stay home and isolate for 5 days (instead of 10) and if the person has no symptoms or the symptoms resolve after 5 days, leaving the house is okay. A mask should be worn around others for 5 more days. In the event of a fever, the person must stay home until it resolves.
- If people are exposed to someone infected with COVID-19 and they have been boosted, finished the primary series of either the Pfizer or Moderna vaccine within the past 6 months, or finished the primary series of the Johnson & Johnson vaccine within the past 2 months, they should wear a mask around others for 10 days and, if possible, test on day 5. However, if symptoms develop, they should get a test and stay home.
- If people are exposed to someone infected with COVID-19 and they are unvaccinated or are more than 6 months out from their second dose of the Pfizer or Moderna vaccine (or more than 2 months after the J&J vaccine) and not boosted, they should quarantine for 5 days and then wear a mask for 5 more days. If quarantine is impossible, a mask should be worn for 10 days. A test on day 5 is suggested if possible. If symptoms occur, they should quarantine and test.
On social media and in interviews with this news organization, public health experts expressed an array of opinions.
A tweet from Eric Topol, MD, editor-in-chief of Medscape, posted the day after the new guidelines came out, had an empty box and this: “The data that support the new @CDCgov 5 day isolation period without a negative test.”
In a tweet on Jan. 2, Ashish K. Jha, MD, MPH, dean of the Brown University School of Public Health, said: “Hearing that CDC considering adding testing to isolation guidelines. That would be great. I’ve been arguing for a while that serial negative antigen tests provide a lot of confidence that someone is not contagious.”
Michael Mina, MD, PhD, chief science officer of eMed, a digital point-of-care platform enabling at-home diagnostic testing, tweeted: “CDC’s new guidance to drop isolation of positives to 5 days without a negative test is reckless. Some [people] stay infectious 3 days, some 12. I absolutely don’t want to sit next to someone who turned [positive] 5 days ago and hasn’t tested Neg. Test Neg to leave isolation early is just smart.”
Paul Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia and an infectious disease specialist, disagrees. Typically, he said, an infected person sheds virus for 7 days.
“If you are asymptomatic, the chances that you are shedding a significant amount of virus is very, very small,” he said in an interview.
Under debate
Testing: While many public health experts say a recommendation to test before leaving isolation is needed, CDC Director Rochelle Walensky, MD, explained testing was not recommended before leaving isolation because PCR testing can stay positive up to 12 weeks after a person is first infected with COVID-19.
Asked why there was not a recommendation for a rapid antigen test before leaving isolation, Dr. Walensky told CNN that it is not known how these tests perform at the end of infection and that the tests are not Food and Drug Administration–authorized for that purpose.
And while the guidelines suggest that those exposed – whether they are boosted, vaccinated, or not – should test on day 5 if possible, that recommendation should be stronger, some said. “At the very least recommend a test in those who can get it done,” said Dr. Topol.
However, making that recommendation is difficult when experts know how difficult it is for people to obtain tests now, William Schaffner, MD, professor of preventive medicine and an infectious disease specialist at Vanderbilt University, Nashville, Tenn., said in an interview.
“I am sure this was intensely debated,” Dr. Schaffner said of the recommendation on testing.
Vaccination status categories: Amesh Adalja, MD, senior scholar at the Johns Hopkins Center for Health Security, Baltimore, questioned the scientific basis behind treating the fully vaccinated (with two mRNA or one J&J vaccine) who are exposed ‘’as the equivalent of the unvaccinated when it comes to the quarantine requirement since the fully vaccinated are protected against what matters.”
Dr. Topol agreed: Guidelines “should be different for vaccinated versus unvaccinated.”
The recommendations for the exposed should definitely be simpler, Dr. Offit said. “I think it would be much simpler to just say, ‘If you are exposed, mask for 10 days,’ “ regardless of vaccination status.
Masks: The guidelines should also be more specific about the type of masks, Dr. Topol said. They should spell out that the masks need to be N95 or KN95, he said.
Science-driven or economy-driven? Was the guidance changed due more to concerns about the economy than to scientific information about infection and transmission? “It was,” Dr. Topol said.
Dr. Adalja sees it differently. “While it is true that this updated guidance will help the economy, it is based on a scientific foundation and should have been issued much earlier than it was.”
Tough decisions
The agency is walking a tightrope, Dr. Schaffner said, adding that he is in general agreement with what the CDC is trying to do. “The tightrope is between the public health ideal and trying to determine what will be acceptable,’’ he said.
The revised guidelines are more practical than before, others said. “The goal is harm reduction and many people just don’t do any isolation if they are faced with a 10-day period,” Dr. Adalja said.
Before issuing the new guidance, the CDC looked at the accumulating science and also took into account stresses on the health care system and other factors, Dr. Schaffner said. “Is it perfect?” Dr. Schaffner said of the new guideline. “No. Is it carefree? No. It’s right in the middle.”
Dr. Schaffner does think the messages about the new recommendations and how they were decided upon could have been communicated better, and in a more understandable manner. Some experts, for instance, led with the economy and the need for people to return to work and school when explaining the guidelines and then brought up the science behind the revisions.
That order should have been reversed, Dr. Schaffner said.
A version of this article first appeared on Medscape.com.
FDA approves first-in-class inclisiran to lower LDL-C
The Food and Drug Administration has approved inclisiran (Leqvio) as an adjunct to statins for further reduction of LDL cholesterol levels, the drug’s developer, Novartis, announced on Dec. 22, 2021.
The first-in-class small interfering RNA (siRNA) agent is also novel among peer drug therapies for its administration by injection initially, at 3 months, and thereafter twice per year.
Inclisiran is indicated for use atop maximally tolerated statins in adults with clinical cardiovascular disease or in patients with heterozygous familial hypercholesterolemia, the company reported.
Such patients who received inclisiran, compared with placebo, in the ORION-9, ORION-10, and ORION-11 randomized trials on which the FDA approval was based showed LDL-C reductions exceeding 50% over 1-2 years.
The drug works by “silencing” RNA involved in synthesis of PCSK9, which has a role in controlling the number of LDL cholesterol cell-surface receptors, a unique mechanism of action among available treatments for dyslipidemia.
Novartis, the company said, “has obtained global rights to develop, manufacture, and commercialize Leqvio under a license and collaboration agreement with Alnylam Pharmaceuticals.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved inclisiran (Leqvio) as an adjunct to statins for further reduction of LDL cholesterol levels, the drug’s developer, Novartis, announced on Dec. 22, 2021.
The first-in-class small interfering RNA (siRNA) agent is also novel among peer drug therapies for its administration by injection initially, at 3 months, and thereafter twice per year.
Inclisiran is indicated for use atop maximally tolerated statins in adults with clinical cardiovascular disease or in patients with heterozygous familial hypercholesterolemia, the company reported.
Such patients who received inclisiran, compared with placebo, in the ORION-9, ORION-10, and ORION-11 randomized trials on which the FDA approval was based showed LDL-C reductions exceeding 50% over 1-2 years.
The drug works by “silencing” RNA involved in synthesis of PCSK9, which has a role in controlling the number of LDL cholesterol cell-surface receptors, a unique mechanism of action among available treatments for dyslipidemia.
Novartis, the company said, “has obtained global rights to develop, manufacture, and commercialize Leqvio under a license and collaboration agreement with Alnylam Pharmaceuticals.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved inclisiran (Leqvio) as an adjunct to statins for further reduction of LDL cholesterol levels, the drug’s developer, Novartis, announced on Dec. 22, 2021.
The first-in-class small interfering RNA (siRNA) agent is also novel among peer drug therapies for its administration by injection initially, at 3 months, and thereafter twice per year.
Inclisiran is indicated for use atop maximally tolerated statins in adults with clinical cardiovascular disease or in patients with heterozygous familial hypercholesterolemia, the company reported.
Such patients who received inclisiran, compared with placebo, in the ORION-9, ORION-10, and ORION-11 randomized trials on which the FDA approval was based showed LDL-C reductions exceeding 50% over 1-2 years.
The drug works by “silencing” RNA involved in synthesis of PCSK9, which has a role in controlling the number of LDL cholesterol cell-surface receptors, a unique mechanism of action among available treatments for dyslipidemia.
Novartis, the company said, “has obtained global rights to develop, manufacture, and commercialize Leqvio under a license and collaboration agreement with Alnylam Pharmaceuticals.”
A version of this article first appeared on Medscape.com.
FDA backs Pfizer booster for 12- to 15-year-olds
Besides updating the authorization for the Pfizer COVID-19 vaccine, the agency also shortened the recommended time between a second dose and the booster to 5 months or more, based on new evidence. In addition, a third primary series dose is now authorized for certain immunocompromised children 5 years to 11 years old. Full details are available in an FDA news release.
The amended emergency use authorization (EUA) only applies to the Pfizer vaccine, said acting FDA Commissioner Janet Woodcock, MD.
“Just to make sure every everyone is clear on this, right now: If you got [Johnson & Johnson’s one-dose vaccine], you get a booster after 2 months. If you got Moderna, you can get a booster at 6 months or beyond,” she said during a media briefing.
What is new, she said, is “if you got Pfizer as your primary series, you can get a booster at 5 months or beyond.”
A lower risk of myocarditis?
Asked about concerns about the risk of myocarditis with vaccination in the 12- to 15-year age group, Dr. Woodcock said they expect it would be “extremely rare with the third dose.”
“We have the real-world evidence from the Israeli experience to help us with that analysis,” she said.
The data so far consistently points to a higher risk of myocarditis after a second mRNA vaccine dose among males, from teenagers to 30-year-olds, with a peak at about 16 to 17 years of age, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said during the media call.
The risk of myocarditis is about 2 to 3 times higher after a second vaccine dose, compared to a booster shot, Dr. Marks said, based on available data. It may be related to the closer dose timing of the second dose versus a third, he added.
“The inference here is that on the risk of myocarditis with third doses in the 12- to 15-year age range is likely to be quite acceptable,” he said.
Dr. Marks also pointed out that most cases of myocarditis clear up quickly.
“We’re not seeing long-lasting effects. That’s not to say that we don’t care about this and that it’s not important,” he said.
“But what it is saying is that in the setting of a tremendous number of Omicron and Delta cases in this country, the potential benefits of getting vaccinated in this age group outweigh that risk,” Dr. Marks said. “We can look at that risk-benefit and still feel comfortable.”
He said that “the really overwhelming majority of these cases, 98%, have been mild” -- shown by a 1-day median hospital stay.
Even so, the FDA plans to continue monitoring for the risk of myocarditis “very closely,” he said.
Interestingly, swollen underarm lymph nodes were seen more frequently after the booster dose than after the second dose of a two-dose primary series, the FDA said.
Reducing the time between primary vaccination with the Pfizer vaccine -- two initial doses -- and the booster shot from 6 months to 5 months is based on decreasing efficacy data that the drugmaker submitted to the FDA.
The 5-month interval was evaluated in a study from Israel published Dec. 21 in the New England Journal of Medicine .
Mixing and matching vaccines
Less clear at the moment is guidance about boosters for people who opted to mix and match their primary vaccine series.
“There was a mix-and-match study that was done which showed that in some cases, the mixing and matching … of an adenoviral record vaccine and an mRNA vaccine seem to give a very good immune response,” Dr. Marks said.
Once more data comes in on mixing and matching, “we’ll analyze them and then potentially make recommendations,” he said.
‘It’s not too late’
No federal government media briefing on COVID-19 would be complete without a plea for the unvaccinated to get immunized.
“We’re talking a lot about boosters right now, but it’s not too late for those who have not gotten a vaccine to get a vaccine,” Dr. Marks said, referring to the tens of millions of Americans who remain unvaccinated at the beginning of 2022.
“We know from our previous studies that even a single dose of the vaccine -- and probably two doses -- can help prevent the worst outcomes from COVID-19, including hospitalization and death.”
A version of this article first appeared on WebMD.com.
Besides updating the authorization for the Pfizer COVID-19 vaccine, the agency also shortened the recommended time between a second dose and the booster to 5 months or more, based on new evidence. In addition, a third primary series dose is now authorized for certain immunocompromised children 5 years to 11 years old. Full details are available in an FDA news release.
The amended emergency use authorization (EUA) only applies to the Pfizer vaccine, said acting FDA Commissioner Janet Woodcock, MD.
“Just to make sure every everyone is clear on this, right now: If you got [Johnson & Johnson’s one-dose vaccine], you get a booster after 2 months. If you got Moderna, you can get a booster at 6 months or beyond,” she said during a media briefing.
What is new, she said, is “if you got Pfizer as your primary series, you can get a booster at 5 months or beyond.”
A lower risk of myocarditis?
Asked about concerns about the risk of myocarditis with vaccination in the 12- to 15-year age group, Dr. Woodcock said they expect it would be “extremely rare with the third dose.”
“We have the real-world evidence from the Israeli experience to help us with that analysis,” she said.
The data so far consistently points to a higher risk of myocarditis after a second mRNA vaccine dose among males, from teenagers to 30-year-olds, with a peak at about 16 to 17 years of age, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said during the media call.
The risk of myocarditis is about 2 to 3 times higher after a second vaccine dose, compared to a booster shot, Dr. Marks said, based on available data. It may be related to the closer dose timing of the second dose versus a third, he added.
“The inference here is that on the risk of myocarditis with third doses in the 12- to 15-year age range is likely to be quite acceptable,” he said.
Dr. Marks also pointed out that most cases of myocarditis clear up quickly.
“We’re not seeing long-lasting effects. That’s not to say that we don’t care about this and that it’s not important,” he said.
“But what it is saying is that in the setting of a tremendous number of Omicron and Delta cases in this country, the potential benefits of getting vaccinated in this age group outweigh that risk,” Dr. Marks said. “We can look at that risk-benefit and still feel comfortable.”
He said that “the really overwhelming majority of these cases, 98%, have been mild” -- shown by a 1-day median hospital stay.
Even so, the FDA plans to continue monitoring for the risk of myocarditis “very closely,” he said.
Interestingly, swollen underarm lymph nodes were seen more frequently after the booster dose than after the second dose of a two-dose primary series, the FDA said.
Reducing the time between primary vaccination with the Pfizer vaccine -- two initial doses -- and the booster shot from 6 months to 5 months is based on decreasing efficacy data that the drugmaker submitted to the FDA.
The 5-month interval was evaluated in a study from Israel published Dec. 21 in the New England Journal of Medicine .
Mixing and matching vaccines
Less clear at the moment is guidance about boosters for people who opted to mix and match their primary vaccine series.
“There was a mix-and-match study that was done which showed that in some cases, the mixing and matching … of an adenoviral record vaccine and an mRNA vaccine seem to give a very good immune response,” Dr. Marks said.
Once more data comes in on mixing and matching, “we’ll analyze them and then potentially make recommendations,” he said.
‘It’s not too late’
No federal government media briefing on COVID-19 would be complete without a plea for the unvaccinated to get immunized.
“We’re talking a lot about boosters right now, but it’s not too late for those who have not gotten a vaccine to get a vaccine,” Dr. Marks said, referring to the tens of millions of Americans who remain unvaccinated at the beginning of 2022.
“We know from our previous studies that even a single dose of the vaccine -- and probably two doses -- can help prevent the worst outcomes from COVID-19, including hospitalization and death.”
A version of this article first appeared on WebMD.com.
Besides updating the authorization for the Pfizer COVID-19 vaccine, the agency also shortened the recommended time between a second dose and the booster to 5 months or more, based on new evidence. In addition, a third primary series dose is now authorized for certain immunocompromised children 5 years to 11 years old. Full details are available in an FDA news release.
The amended emergency use authorization (EUA) only applies to the Pfizer vaccine, said acting FDA Commissioner Janet Woodcock, MD.
“Just to make sure every everyone is clear on this, right now: If you got [Johnson & Johnson’s one-dose vaccine], you get a booster after 2 months. If you got Moderna, you can get a booster at 6 months or beyond,” she said during a media briefing.
What is new, she said, is “if you got Pfizer as your primary series, you can get a booster at 5 months or beyond.”
A lower risk of myocarditis?
Asked about concerns about the risk of myocarditis with vaccination in the 12- to 15-year age group, Dr. Woodcock said they expect it would be “extremely rare with the third dose.”
“We have the real-world evidence from the Israeli experience to help us with that analysis,” she said.
The data so far consistently points to a higher risk of myocarditis after a second mRNA vaccine dose among males, from teenagers to 30-year-olds, with a peak at about 16 to 17 years of age, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said during the media call.
The risk of myocarditis is about 2 to 3 times higher after a second vaccine dose, compared to a booster shot, Dr. Marks said, based on available data. It may be related to the closer dose timing of the second dose versus a third, he added.
“The inference here is that on the risk of myocarditis with third doses in the 12- to 15-year age range is likely to be quite acceptable,” he said.
Dr. Marks also pointed out that most cases of myocarditis clear up quickly.
“We’re not seeing long-lasting effects. That’s not to say that we don’t care about this and that it’s not important,” he said.
“But what it is saying is that in the setting of a tremendous number of Omicron and Delta cases in this country, the potential benefits of getting vaccinated in this age group outweigh that risk,” Dr. Marks said. “We can look at that risk-benefit and still feel comfortable.”
He said that “the really overwhelming majority of these cases, 98%, have been mild” -- shown by a 1-day median hospital stay.
Even so, the FDA plans to continue monitoring for the risk of myocarditis “very closely,” he said.
Interestingly, swollen underarm lymph nodes were seen more frequently after the booster dose than after the second dose of a two-dose primary series, the FDA said.
Reducing the time between primary vaccination with the Pfizer vaccine -- two initial doses -- and the booster shot from 6 months to 5 months is based on decreasing efficacy data that the drugmaker submitted to the FDA.
The 5-month interval was evaluated in a study from Israel published Dec. 21 in the New England Journal of Medicine .
Mixing and matching vaccines
Less clear at the moment is guidance about boosters for people who opted to mix and match their primary vaccine series.
“There was a mix-and-match study that was done which showed that in some cases, the mixing and matching … of an adenoviral record vaccine and an mRNA vaccine seem to give a very good immune response,” Dr. Marks said.
Once more data comes in on mixing and matching, “we’ll analyze them and then potentially make recommendations,” he said.
‘It’s not too late’
No federal government media briefing on COVID-19 would be complete without a plea for the unvaccinated to get immunized.
“We’re talking a lot about boosters right now, but it’s not too late for those who have not gotten a vaccine to get a vaccine,” Dr. Marks said, referring to the tens of millions of Americans who remain unvaccinated at the beginning of 2022.
“We know from our previous studies that even a single dose of the vaccine -- and probably two doses -- can help prevent the worst outcomes from COVID-19, including hospitalization and death.”
A version of this article first appeared on WebMD.com.
Travel/school disruptions as COVID-19 cases grow in 2022
As the United States enters a third year of the COVID-19 pandemic,
The United States is reporting a 7-day average of more than 386,000 cases after several record-breaking days, according to the data tracker by the New York Times. The United States surpassed 585,000 cases on Dec. 30, setting a new record before the New Year’s holiday.
New York, Washington, D.C., and other states along the East Coast are leading the national surge. New York reported more than 85,000 new cases on the last day of 2021, marking the highest 1-day total in the state since the pandemic began.
“As we fight the winter surge, we need to keep the most vulnerable among us in mind – do what you can to keep others in your community safe from COVID-19,” New York Gov. Kathy Hochul said in a statement on Jan. 1, 2022.
“Wear a mask, wash your hands, and take advantage of the best tool we have at our disposal – the vaccine,” she said.
The 2021 winter surge in the United States peaked around Jan. 12, which may suggest that the country has a week or so before the current wave reaches its height and begins to drop, the newspaper reported.
In the meantime, people are dealing with disruptions as they return from holiday travel and begin the new year. Airlines canceled more than 2,700 flights on Jan. 1 and more than 1,900 flights on Jan. 2, bringing the total since Christmas Eve to more than 14,000 canceled flights.
About half of cancellations were connected to wintry weather at key airline hubs in Chicago and Denver, the newspaper reported, as well as ongoing flight crew shortages caused by the Omicron variant.
More disruptions could continue, the Federal Aviation Administration warned, as an increasing number of its air-traffic control employees test positive for COVID-19.
“To maintain safety, traffic volumes at some facilities could be reduced, which might result in delays during busy periods,” an FAA spokesman told The Wall Street Journal.
The current COVID-19 surge will also affect businesses and schools as the new year begins. A growing number of universities are opting to start the next semester with remote instruction.
American University, Duke University, and Michigan State University announced in recent days that they would delay in-person classes to slow the spread of the coronavirus on campus. They will begin classes online on Jan. 10 and return to campus the following week or later.
“I realize that students prefer to be in person, and so do I. But it is important that we do so in a safe manner,” Samuel Stanley Jr., MD, president of Michigan State University, said in a statement on New Year’s Eve.
K-12 school districts are deciding how to adapt as well. Some districts are bringing back mask requirements, and some are ramping up testing. Others are moving to remote learning – and signaling the need for flexibility as the Omicron variant brings new surprises.
“Change has been the only constant in this fight,” Roger Leon, the superintendent for Newark (N.J.) Public Schools, wrote in a note to parents. He announced on Dec. 30, 2021, that students would learn remotely for at least the first 2 weeks of the new year.
This continues “to be a brutal, relentless, and ruthless virus that rears its ugly head at inopportune times,” he said.
A version of this article first appeared on WebMD.com.
As the United States enters a third year of the COVID-19 pandemic,
The United States is reporting a 7-day average of more than 386,000 cases after several record-breaking days, according to the data tracker by the New York Times. The United States surpassed 585,000 cases on Dec. 30, setting a new record before the New Year’s holiday.
New York, Washington, D.C., and other states along the East Coast are leading the national surge. New York reported more than 85,000 new cases on the last day of 2021, marking the highest 1-day total in the state since the pandemic began.
“As we fight the winter surge, we need to keep the most vulnerable among us in mind – do what you can to keep others in your community safe from COVID-19,” New York Gov. Kathy Hochul said in a statement on Jan. 1, 2022.
“Wear a mask, wash your hands, and take advantage of the best tool we have at our disposal – the vaccine,” she said.
The 2021 winter surge in the United States peaked around Jan. 12, which may suggest that the country has a week or so before the current wave reaches its height and begins to drop, the newspaper reported.
In the meantime, people are dealing with disruptions as they return from holiday travel and begin the new year. Airlines canceled more than 2,700 flights on Jan. 1 and more than 1,900 flights on Jan. 2, bringing the total since Christmas Eve to more than 14,000 canceled flights.
About half of cancellations were connected to wintry weather at key airline hubs in Chicago and Denver, the newspaper reported, as well as ongoing flight crew shortages caused by the Omicron variant.
More disruptions could continue, the Federal Aviation Administration warned, as an increasing number of its air-traffic control employees test positive for COVID-19.
“To maintain safety, traffic volumes at some facilities could be reduced, which might result in delays during busy periods,” an FAA spokesman told The Wall Street Journal.
The current COVID-19 surge will also affect businesses and schools as the new year begins. A growing number of universities are opting to start the next semester with remote instruction.
American University, Duke University, and Michigan State University announced in recent days that they would delay in-person classes to slow the spread of the coronavirus on campus. They will begin classes online on Jan. 10 and return to campus the following week or later.
“I realize that students prefer to be in person, and so do I. But it is important that we do so in a safe manner,” Samuel Stanley Jr., MD, president of Michigan State University, said in a statement on New Year’s Eve.
K-12 school districts are deciding how to adapt as well. Some districts are bringing back mask requirements, and some are ramping up testing. Others are moving to remote learning – and signaling the need for flexibility as the Omicron variant brings new surprises.
“Change has been the only constant in this fight,” Roger Leon, the superintendent for Newark (N.J.) Public Schools, wrote in a note to parents. He announced on Dec. 30, 2021, that students would learn remotely for at least the first 2 weeks of the new year.
This continues “to be a brutal, relentless, and ruthless virus that rears its ugly head at inopportune times,” he said.
A version of this article first appeared on WebMD.com.
As the United States enters a third year of the COVID-19 pandemic,
The United States is reporting a 7-day average of more than 386,000 cases after several record-breaking days, according to the data tracker by the New York Times. The United States surpassed 585,000 cases on Dec. 30, setting a new record before the New Year’s holiday.
New York, Washington, D.C., and other states along the East Coast are leading the national surge. New York reported more than 85,000 new cases on the last day of 2021, marking the highest 1-day total in the state since the pandemic began.
“As we fight the winter surge, we need to keep the most vulnerable among us in mind – do what you can to keep others in your community safe from COVID-19,” New York Gov. Kathy Hochul said in a statement on Jan. 1, 2022.
“Wear a mask, wash your hands, and take advantage of the best tool we have at our disposal – the vaccine,” she said.
The 2021 winter surge in the United States peaked around Jan. 12, which may suggest that the country has a week or so before the current wave reaches its height and begins to drop, the newspaper reported.
In the meantime, people are dealing with disruptions as they return from holiday travel and begin the new year. Airlines canceled more than 2,700 flights on Jan. 1 and more than 1,900 flights on Jan. 2, bringing the total since Christmas Eve to more than 14,000 canceled flights.
About half of cancellations were connected to wintry weather at key airline hubs in Chicago and Denver, the newspaper reported, as well as ongoing flight crew shortages caused by the Omicron variant.
More disruptions could continue, the Federal Aviation Administration warned, as an increasing number of its air-traffic control employees test positive for COVID-19.
“To maintain safety, traffic volumes at some facilities could be reduced, which might result in delays during busy periods,” an FAA spokesman told The Wall Street Journal.
The current COVID-19 surge will also affect businesses and schools as the new year begins. A growing number of universities are opting to start the next semester with remote instruction.
American University, Duke University, and Michigan State University announced in recent days that they would delay in-person classes to slow the spread of the coronavirus on campus. They will begin classes online on Jan. 10 and return to campus the following week or later.
“I realize that students prefer to be in person, and so do I. But it is important that we do so in a safe manner,” Samuel Stanley Jr., MD, president of Michigan State University, said in a statement on New Year’s Eve.
K-12 school districts are deciding how to adapt as well. Some districts are bringing back mask requirements, and some are ramping up testing. Others are moving to remote learning – and signaling the need for flexibility as the Omicron variant brings new surprises.
“Change has been the only constant in this fight,” Roger Leon, the superintendent for Newark (N.J.) Public Schools, wrote in a note to parents. He announced on Dec. 30, 2021, that students would learn remotely for at least the first 2 weeks of the new year.
This continues “to be a brutal, relentless, and ruthless virus that rears its ugly head at inopportune times,” he said.
A version of this article first appeared on WebMD.com.
Medicaid implements waivers for some clinical trial coverage
Federal officials will allow some flexibility in meeting new requirements on covering the costs of clinical trials for people enrolled in Medicaid, seeking to accommodate states where legislatures will not meet in time to make needed changes in rules.
Congress in 2020 ordered U.S. states to have their Medicaid programs cover expenses related to participation in certain clinical trials, a move that was hailed by the American Society of Clinical Oncology (ASCO) and other groups as a boost to trials as well as to patients with serious illness who have lower incomes.
The mandate went into effect on Jan. 1, but the Centers for Medicare & Medicaid Services will allow accommodations in terms of implementation time for states that have not yet been able to make needed legislative changes, Daniel Tsai, deputy administrator and director of the Center for Medicaid and CHIP Services, wrote in a Dec. 7 letter. Mr. Tsai’s letter doesn’t mention specific states. The CMS did not immediately respond to a request seeking information on the states expected to apply for waivers.
Medicaid has in recent years been a rare large U.S. insurance program that does not cover the costs of clinical trials. The Affordable Care Act of 2010 mandated this coverage for people in private insurance plans. The federal government in 2000 decided that Medicare would do so.
‘A hidden opportunity’
A perspective article last May in the New England Journal of Medicine referred to the new Medicaid mandate on clinical trials as a “hidden opportunity,” referring to its genesis as an add-on in a massive federal spending package enacted in December 2020.
In the article, Samuel U. Takvorian, MD, MSHP, of the University of Pennsylvania, Philadelphia, and coauthors noted that rates of participation in clinical trials remain low for racial and ethnic minority groups, due in part to the lack of Medicaid coverage.
“For example, non-Hispanic White patients are nearly twice as likely as Black patients and three times as likely as Hispanic patients to enroll in cancer clinical trials – a gap that has widened over time,” Dr. Takvorian and coauthors wrote. “Inequities in enrollment have also manifested during the COVID-19 pandemic, which has disproportionately affected non-White patients, without their commensurate representation in trials of COVID-19 therapeutics.”
In October, researchers from the Arthur G. James Cancer Hospital and Ohio State University, Columbus, published results of a retrospective study of patients with stage I-IV pancreatic cancer that also found inequities in enrollment. Mariam F. Eskander, MD, MPH, and coauthors reported what they found by examining records for 1,127 patients (0.4%) enrolled in clinical trials and 301,340 (99.6%) who did not enroll. They found that enrollment in trials increased over the study period, but not for Black patients or patients on Medicaid.
In an interview, Dr. Eskander said the new Medicaid policy will remove a major obstacle to participation in clinical trials. An oncologist, Dr. Eskander said she is looking forward to being able to help more of her patients get access to experimental medicines and treatments.
But that may not be enough to draw more people with low incomes into these studies, said Dr. Eskander, who is now at Rutgers Cancer Institute of New Jersey in New Brunswick. She urges greater use of patient navigators to help people on Medicaid understand the resources available to them, as well as broad use of Medicaid’s nonemergency medical transportation (NEMT) benefit.
“Some patients will be offered clinical trial enrollment and some will accept, but I really worry about the challenges low-income people face with things like transportation and getting time off work,” she said.
A version of this article first appeared on Medscape.com.
Federal officials will allow some flexibility in meeting new requirements on covering the costs of clinical trials for people enrolled in Medicaid, seeking to accommodate states where legislatures will not meet in time to make needed changes in rules.
Congress in 2020 ordered U.S. states to have their Medicaid programs cover expenses related to participation in certain clinical trials, a move that was hailed by the American Society of Clinical Oncology (ASCO) and other groups as a boost to trials as well as to patients with serious illness who have lower incomes.
The mandate went into effect on Jan. 1, but the Centers for Medicare & Medicaid Services will allow accommodations in terms of implementation time for states that have not yet been able to make needed legislative changes, Daniel Tsai, deputy administrator and director of the Center for Medicaid and CHIP Services, wrote in a Dec. 7 letter. Mr. Tsai’s letter doesn’t mention specific states. The CMS did not immediately respond to a request seeking information on the states expected to apply for waivers.
Medicaid has in recent years been a rare large U.S. insurance program that does not cover the costs of clinical trials. The Affordable Care Act of 2010 mandated this coverage for people in private insurance plans. The federal government in 2000 decided that Medicare would do so.
‘A hidden opportunity’
A perspective article last May in the New England Journal of Medicine referred to the new Medicaid mandate on clinical trials as a “hidden opportunity,” referring to its genesis as an add-on in a massive federal spending package enacted in December 2020.
In the article, Samuel U. Takvorian, MD, MSHP, of the University of Pennsylvania, Philadelphia, and coauthors noted that rates of participation in clinical trials remain low for racial and ethnic minority groups, due in part to the lack of Medicaid coverage.
“For example, non-Hispanic White patients are nearly twice as likely as Black patients and three times as likely as Hispanic patients to enroll in cancer clinical trials – a gap that has widened over time,” Dr. Takvorian and coauthors wrote. “Inequities in enrollment have also manifested during the COVID-19 pandemic, which has disproportionately affected non-White patients, without their commensurate representation in trials of COVID-19 therapeutics.”
In October, researchers from the Arthur G. James Cancer Hospital and Ohio State University, Columbus, published results of a retrospective study of patients with stage I-IV pancreatic cancer that also found inequities in enrollment. Mariam F. Eskander, MD, MPH, and coauthors reported what they found by examining records for 1,127 patients (0.4%) enrolled in clinical trials and 301,340 (99.6%) who did not enroll. They found that enrollment in trials increased over the study period, but not for Black patients or patients on Medicaid.
In an interview, Dr. Eskander said the new Medicaid policy will remove a major obstacle to participation in clinical trials. An oncologist, Dr. Eskander said she is looking forward to being able to help more of her patients get access to experimental medicines and treatments.
But that may not be enough to draw more people with low incomes into these studies, said Dr. Eskander, who is now at Rutgers Cancer Institute of New Jersey in New Brunswick. She urges greater use of patient navigators to help people on Medicaid understand the resources available to them, as well as broad use of Medicaid’s nonemergency medical transportation (NEMT) benefit.
“Some patients will be offered clinical trial enrollment and some will accept, but I really worry about the challenges low-income people face with things like transportation and getting time off work,” she said.
A version of this article first appeared on Medscape.com.
Federal officials will allow some flexibility in meeting new requirements on covering the costs of clinical trials for people enrolled in Medicaid, seeking to accommodate states where legislatures will not meet in time to make needed changes in rules.
Congress in 2020 ordered U.S. states to have their Medicaid programs cover expenses related to participation in certain clinical trials, a move that was hailed by the American Society of Clinical Oncology (ASCO) and other groups as a boost to trials as well as to patients with serious illness who have lower incomes.
The mandate went into effect on Jan. 1, but the Centers for Medicare & Medicaid Services will allow accommodations in terms of implementation time for states that have not yet been able to make needed legislative changes, Daniel Tsai, deputy administrator and director of the Center for Medicaid and CHIP Services, wrote in a Dec. 7 letter. Mr. Tsai’s letter doesn’t mention specific states. The CMS did not immediately respond to a request seeking information on the states expected to apply for waivers.
Medicaid has in recent years been a rare large U.S. insurance program that does not cover the costs of clinical trials. The Affordable Care Act of 2010 mandated this coverage for people in private insurance plans. The federal government in 2000 decided that Medicare would do so.
‘A hidden opportunity’
A perspective article last May in the New England Journal of Medicine referred to the new Medicaid mandate on clinical trials as a “hidden opportunity,” referring to its genesis as an add-on in a massive federal spending package enacted in December 2020.
In the article, Samuel U. Takvorian, MD, MSHP, of the University of Pennsylvania, Philadelphia, and coauthors noted that rates of participation in clinical trials remain low for racial and ethnic minority groups, due in part to the lack of Medicaid coverage.
“For example, non-Hispanic White patients are nearly twice as likely as Black patients and three times as likely as Hispanic patients to enroll in cancer clinical trials – a gap that has widened over time,” Dr. Takvorian and coauthors wrote. “Inequities in enrollment have also manifested during the COVID-19 pandemic, which has disproportionately affected non-White patients, without their commensurate representation in trials of COVID-19 therapeutics.”
In October, researchers from the Arthur G. James Cancer Hospital and Ohio State University, Columbus, published results of a retrospective study of patients with stage I-IV pancreatic cancer that also found inequities in enrollment. Mariam F. Eskander, MD, MPH, and coauthors reported what they found by examining records for 1,127 patients (0.4%) enrolled in clinical trials and 301,340 (99.6%) who did not enroll. They found that enrollment in trials increased over the study period, but not for Black patients or patients on Medicaid.
In an interview, Dr. Eskander said the new Medicaid policy will remove a major obstacle to participation in clinical trials. An oncologist, Dr. Eskander said she is looking forward to being able to help more of her patients get access to experimental medicines and treatments.
But that may not be enough to draw more people with low incomes into these studies, said Dr. Eskander, who is now at Rutgers Cancer Institute of New Jersey in New Brunswick. She urges greater use of patient navigators to help people on Medicaid understand the resources available to them, as well as broad use of Medicaid’s nonemergency medical transportation (NEMT) benefit.
“Some patients will be offered clinical trial enrollment and some will accept, but I really worry about the challenges low-income people face with things like transportation and getting time off work,” she said.
A version of this article first appeared on Medscape.com.

