User login
Recovery of Hair in the Psoriatic Plaques of a Patient With Coexistent Alopecia Universalis
To the Editor:
Both alopecia areata (AA) and psoriasis vulgaris are chronic relapsing autoimmune diseases, with AA causing nonscarring hair loss in approximately 0.1% to 0.2%1 of the population with a lifetime risk of 1.7%,2 and psoriasis more broadly impacting 1.5% to 2% of the population.3 The helper T cell (TH1) cytokine milieu is pathogenic in both conditions.4-6 IFN-γ knockout mice, unlike their wild-type counterparts, do not exhibit AA.7 Psoriasis is notably improved by IL-10 injections, which dampen the TH1 response.8 Distinct from AA, TH17 and TH22 cells have been implicated as key players in psoriasis pathogenesis, along with the associated IL-17 and IL-22 cytokines.9-12
Few cases of patients with concurrent AA and psoriasis have been described. Interestingly, these cases document normal hair regrowth in the areas of psoriasis.13-16 These cases may offer unique insight into the immune factors driving each disease. We describe a case of a man with both alopecia universalis (AU) and psoriasis who developed hair regrowth in some of the psoriatic plaques.
A 34-year-old man with concurrent AU and psoriasis who had not used any systemic or topical medication for either condition in the last year presented to our clinic seeking treatment. The patient had a history of alopecia totalis as a toddler that completely resolved by 4 years of age with the use of squaric acid dibutylester (SADBE). At 31 years of age, the alopecia recurred and was localized to the scalp. It was partially responsive to intralesional triamcinolone acetonide. The patient’s alopecia worsened over the 2 years following recurrence, ultimately progressing to AU. Two months after the alopecia recurrence, he developed the first psoriatic plaques. As the plaque psoriasis progressed, systemic therapy was initiated, first methotrexate and then etanercept. Shortly after developing AU, he lost his health insurance and discontinued all therapy. The patient’s psoriasis began to recur approximately 3 months after stopping etanercept. He was not using any other psoriasis medications. At that time, he noted terminal hair regrowth within some of the psoriatic plaques. No terminal hairs grew outside of the psoriatic plaques, and all regions with growth had previously been without hair for an extended period of time. The patient presented to our clinic approximately 1 year later. He had no other medical conditions and no relevant family history.
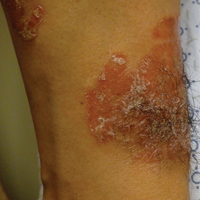
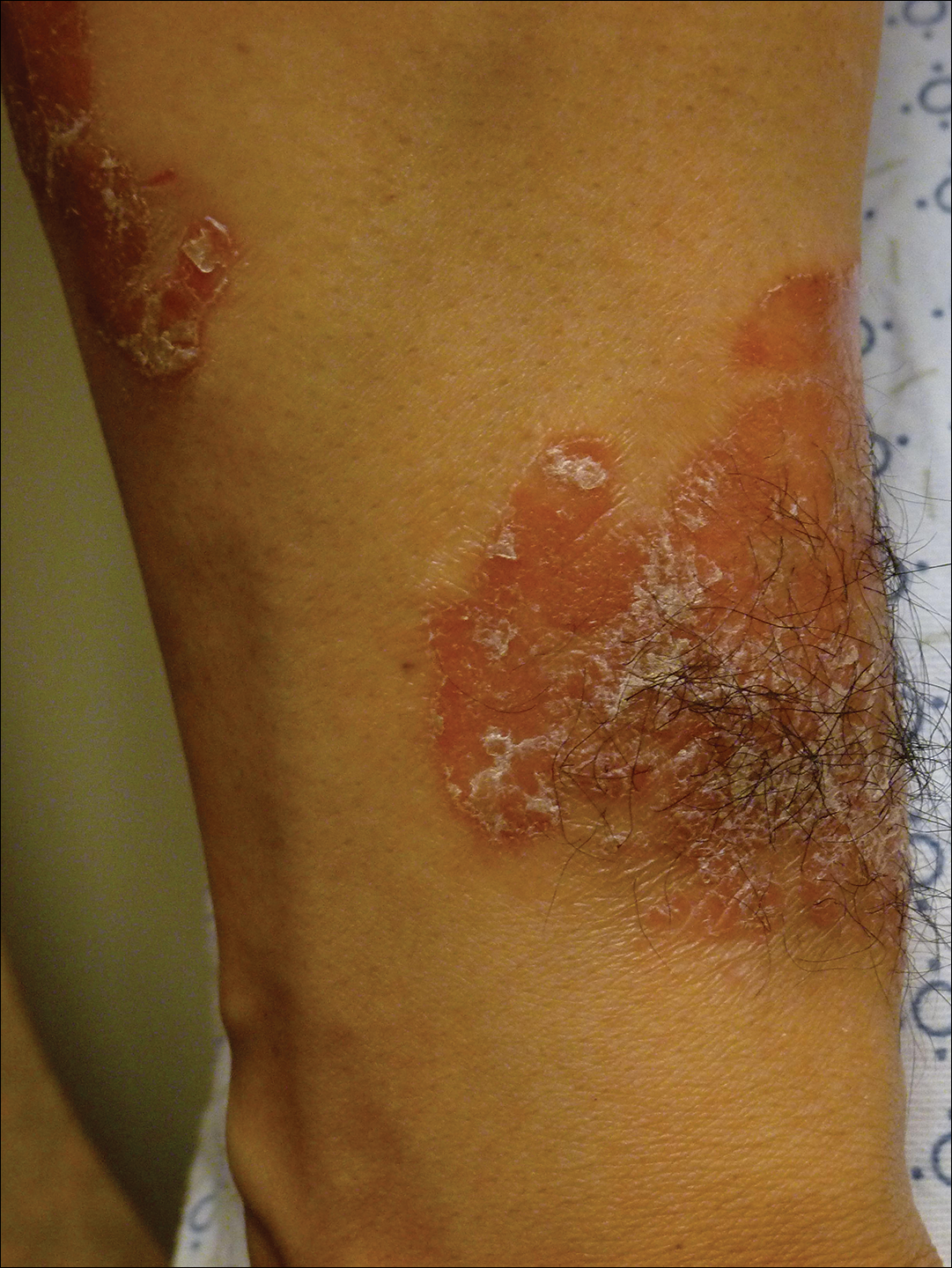
On initial physical examination, he had nonscarring hair loss involving nearly 100% of the body with psoriatic plaques on approximately 30% of the body surface area. Regions of terminal hair growth were confined to some but not all of the psoriatic plaques (Figure). Interestingly, the terminal hairs were primarily localized to the thickest central regions of the plaques. The patient’s psoriasis was treated with a combination of topical clobetasol and calcipotriene. In addition, he was started on tacrolimus ointment to the face and eyebrows for the AA. Maintenance of terminal hair within a region of topically treated psoriasis on the forearm persisted at the 2-month follow-up despite complete clearance of the corresponding psoriatic plaque. A small psoriatic plaque on the scalp cleared early with topical therapy without noticeable hair regrowth. The patient subsequently was started on contact immunotherapy with SADBE and intralesional triamcinolone acetonide for the scalp alopecia without satisfactory response. He decided to discontinue further attempts at treating the alopecia and requested to be restarted on etanercept therapy for recalcitrant psoriatic plaques. His psoriasis responded well to this therapy and he continues to be followed in our psoriasis clinic. One year after clearance of the treated psoriatic plaques, the corresponding terminal hairs persist.
Contact immunotherapy, most commonly with diphenylcyclopropenone or SADBE, is reported to have a 50% to 60% success rate in extensive AA, with a broad range of 9% to 87%17; however, randomized controlled trials testing the efficacy of contact immunotherapy are lacking. Although the mechanism of action of these topical sensitizers is not clearly delineated, it has been postulated that by inducing a new type of inflammatory response in the region, the immunologic milieu is changed, allowing the hair to grow. Some proposed mechanisms include promoting perifollicular lymphocyte apoptosis, preventing new recruitment of autoreactive lymphocytes, and allowing for the correction of aberrant major histocompatibility complex expression on the hair matrix epithelium to regain follicle immune privilege.18-20
Iatrogenic immunotherapy may work analogously to the natural immune system deviation demonstrated in our patient. Psoriasis and AA are believed to form competing immune cells and cytokine milieus, thus explaining how an individual with AA could regain normal hair growth in areas of psoriasis.15,16 The Renbök phenomenon, or reverse Köbner phenomenon, coined by Happle et al13 can be used to describe both the iatrogenic and natural cases of dermatologic disease improvement in response to secondary insults.14
A complex cascade of immune cells and cytokines coordinate AA pathogenesis. In the acute stage of AA, an inflammatory infiltrate of CD4+ T cells, CD8+ T cells, and antigen-presenting cells target anagen phase follicles, with a higher CD4+:CD8+ ratio in clinically active disease.21-23 Subcutaneous injections of either CD4+ or CD8+ lymphocyte subsets from mice with AA into normal-haired mice induces disease. However, CD8+ T cell injections rapidly produce apparent hair loss, whereas CD4+ T cells cause hair loss after several weeks, suggesting that CD8+ T cells directly modulate AA hair loss and CD4+ T cells act as an aide.24 The growth, differentiation, and survival of CD8+ T cells are stimulated by IL-2 and IFN-γ. Alopecia areata biopsies demonstrate a prevalence of TH1 cytokines, and patients with localized AA, alopecia totalis, and AU have notably higher serum IFN-γ levels compared to controls.25 In murine models, IL-1α and IL-1β increase during the catagen phase of the hair cycle and peak during the telogen phase.26 Excessive IL-1β expression is detected in the early stages of human disease, and certain IL-1β polymorphisms are associated with severe forms of AA.26 The role of tumor necrosis factor (TNF) α in AA is not well understood. In vitro studies show it inhibits hair growth, suggesting the cytokine may play a role in AA.27 However, anti–TNF-α therapy is not effective in AA, and case reports propose these therapies rarely induce AA.28-31
The TH1 response is likewise critical to psoriatic plaque development. IFN-γ and TNF-α are overexpressed in psoriatic plaques.32 IFN-γ has an antiproliferative and differentiation-inducing effect on normal keratinocytes, but psoriatic epithelial cells in vitro respond differently to the cytokine with a notably diminished growth inhibition.33,34 One explanation for the role of IFN-γ is that it stimulates dendritic cells to produce IL-1 and IL-23.35 IL-23 activates TH17 cells36; TH1 and TH17 conditions produce IL-22 whose serum level correlates with disease severity.37-39 IL-22 induces keratinocyte proliferation and migration and inhibits keratinocyte differentiation, helping account for hallmarks of the disease.40 Patients with psoriasis have increased levels of TH1, TH17, and TH22 cells, as well as their associated cytokines, in the skin and blood compared to controls.4,11,32,39,41
Alopecia areata and psoriasis are regulated by complex and still not entirely understood immune interactions. The fact that many of the same therapies are used to treat both diseases emphasizes both their overlapping characteristics and the lack of targeted therapy. It is unclear if and how the topical or systemic therapies used in our patient to treat one disease affected the natural history of the other condition. It is important to highlight, however, that the patient had not been treated for months when he developed the psoriatic plaques with hair regrowth. Other case reports also document hair regrowth in untreated plaques,13,16 making it unlikely to be a side effect of the medication regimen. For both psoriasis and AA, the immune cell composition and cytokine levels in the skin or serum vary throughout a patient’s disease course depending on severity of disease or response to treatment.6,39,42,43 Therefore, we hypothesize that the 2 conditions interact in a similarly distinct manner based on each disease’s stage and intensity in the patient. Both our patient’s course thus far and the various presentations described by other groups support this hypothesis. Our patient had a small region of psoriasis on the scalp that cleared without any terminal hair growth. He also had larger plaques on the forearms that developed hair growth most predominantly within the thicker regions of the plaques. His unique presentation highlights the fluidity of the immune factors driving psoriasis vulgaris and AA.
- Safavi K. Prevalence of alopecia areata in the First National Health and Nutrition Examination Survey. Arch Dermatol. 1992;128:702.
- Safavi KH, Muller SA, Suman VJ, et al. Incidence of alopecia areata in Olmsted County, Minnesota, 1975 through 1989. Mayo Clin Proc. 1995;70:628-633.
- Wolff K, Johnson RA. Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. 6th ed. New York, NY: McGraw-Hill; 2009.
- Austin LM, Ozawa M, Kikuchi T, et al. The majority of epidermal T cells in psoriasis vulgaris lesions can produce type 1 cytokines, interferon-gamma, interleukin-2, and tumor necrosis factor-alpha, defining TC1 (cytotoxic T lymphocyte) and TH1 effector populations: a type 1 differentiation bias is also measured in circulating blood T cells in psoriatic patients. J Invest Dermatol. 1999;113:752-759.
- Ghoreishi M, Martinka M, Dutz JP. Type 1 interferon signature in the scalp lesions of alopecia areata. Br J Dermatol. 2010;163:57-62.
- Rossi A, Cantisani C, Carlesimo M, et al. Serum concentrations of IL-2, IL-6, IL-12 and TNF-α in patients with alopecia areata. Int J Immunopathol Pharmacol. 2012;25:781-788.
- Freyschmidt-Paul P, McElwee KJ, Hoffmann R, et al. Interferon-gamma-deficient mice are resistant to the development of alopecia areata. Br J Dermatol. 2006;155:515-521.
- Reich K, Garbe C, Blaschke V, et al. Response of psoriasis to interleukin-10 is associated with suppression of cutaneous type 1 inflammation, downregulation of the epidermal interleukin-8/CXCR2 pathway and normalization of keratinocyte maturation. J Invest Dermatol. 2001;116:319-329.
- Teunissen MB, Koomen CW, de Waal Malefyt R, et al. Interleukin-17 and interferon-gamma synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J Invest Dermatol. 1998;111:645-649.
- Zheng Y, Danilenko DM, Valdez P, et al. Interleukin-22, a T(H)17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature. 2007;445:648-651.
- Boniface K, Guignouard E, Pedretti N, et al. A role for T cell-derived interleukin 22 in psoriatic skin inflammation. Clin Exp Immunol. 2007;150:407-415.
- Zaba LC, Suárez-Fariñas M, Fuentes-Duculan J, et al. Effective treatment of psoriasis with etanercept is linked to suppression of IL-17 signaling, not immediate response TNF genes. J Allergy Clin Immunol. 2009;124:1022-1030.e395.
- Happle R, van der Steen PHM, Perret CM. The Renbök phenomenon: an inverse Köebner reaction observed in alopecia areata. Eur J Dermatol. 1991;2:39-40.
- Ito T, Hashizume H, Takigawa M. Contact immunotherapy-induced Renbök phenomenon in a patient with alopecia areata and psoriasis vulgaris. Eur J Dermatol. 2010;20:126-127.
- Criado PR, Valente NY, Michalany NS, et al. An unusual association between scalp psoriasis and ophiasic alopecia areata: the Renbök phenomenon. Clin Exp Dermatol. 2007;32:320-321.
- Harris JE, Seykora JT, Lee RA. Renbök phenomenon and contact sensitization in a patient with alopecia universalis. Arch Dermatol. 2010;146:422-425.
- Alkhalifah A. Topical and intralesional therapies for alopecia areata. Dermatol Ther. 2011;24:355-363.
- Herbst V, Zöller M, Kissling S, et al. Diphenylcyclopropenone treatment of alopecia areata induces apoptosis of perifollicular lymphocytes. Eur J Dermatol. 2006;16:537-542.
- Zöller M, Freyschmidt-Paul P, Vitacolonna M, et al. Chronic delayed-type hypersensitivity reaction as a means to treat alopecia areata. Clin Exp Immunol. 2004;135:398-408.
- Bröcker EB, Echternacht-Happle K, Hamm H, et al. Abnormal expression of class I and class II major histocompatibility antigens in alopecia areata: modulation by topical immunotherapy. J Invest Dermatol. 1987;88:564-568.
- Todes-Taylor N, Turner R, Wood GS, et al. T cell subpopulations in alopecia areata. J Am Acad Dermatol. 1984;11:216-223.
- Perret C, Wiesner-Menzel L, Happle R. Immunohistochemical analysis of T-cell subsets in the peribulbar and intrabulbar infiltrates of alopecia areata. Acta Derm Venereol. 1984;64:26-30.
- Wiesner-Menzel L, Happle R. Intrabulbar and peribulbar accumulation of dendritic OKT 6-positive cells in alopecia areata. Arch Dermatol Res. 1984;276:333-334.
- McElwee KJ, Freyschmidt-Paul P, Hoffmann R, et al. Transfer of CD8+ cells induces localized hair loss whereas CD4+/CD25– cells promote systemic alopecia areata and CD4+/CD25+ cells blockade disease onset in the C3H/HeJ mouse model. J Invest Dermatol. 2005;124:947-957.
- Arca E, Muşabak U, Akar A, et al. Interferon-gamma in alopecia areata. Eur J Dermatol. 2004;14:33-36.
- Hoffmann R. The potential role of cytokines and T cells in alopecia areata. J Investig Dermatol Symp Proc. 1999;4:235-238.
- Philpott MP, Sanders DA, Bowen J, et al. Effects of interleukins, colony-stimulating factor and tumour necrosis factor on human hair follicle growth in vitro: a possible role for interleukin-1 and tumour necrosis factor-alpha in alopecia areata. Br J Dermatol. 1996;135:942-948.
- Le Bidre E, Chaby G, Martin L, et al. Alopecia areata during anti-TNF alpha therapy: nine cases. Ann Dermatol Venereol. 2011;138:285-293.
- Ferran M, Calvet J, Almirall M, et al. Alopecia areata as another immune-mediated disease developed in patients treated with tumour necrosis factor-α blocker agents: report of five cases and review of the literature. J Eur Acad Dermatol Venereol. 2011;25:479-484.
- Pan Y, Rao NA. Alopecia areata during etanercept therapy. Ocul Immunol Inflamm. 2009;17:127-129.
- Pelivani N, Hassan AS, Braathen LR, et al. Alopecia areata universalis elicited during treatment with adalimumab. Dermatology. 2008;216:320-323.
- Uyemura K, Yamamura M, Fivenson DF, et al. The cytokine network in lesional and lesion-free psoriatic skin is characterized by a T-helper type 1 cell-mediated response. J Invest Dermatol. 1993;101:701-705.
- Baker BS, Powles AV, Valdimarsson H, et al. An altered response by psoriatic keratinocytes to gamma interferon. Scan J Immunol. 1988;28:735-740.
- Jackson M, Howie SE, Weller R, et al. Psoriatic keratinocytes show reduced IRF-1 and STAT-1alpha activation in response to gamma-IFN. FASEB J. 1999;13:495-502.
- Perera GK, Di Meglio P, Nestle FO. Psoriasis. Annu Rev Pathol. 2012;7:385-422.
- McGeachy MJ, Chen Y, Tato CM, et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol. 2009;10:314-324.
- Volpe E, Servant N, Zollinger R, et al. A critical function for transforming growth factor-beta, interleukin 23 and proinflammatory cytokines in driving and modulating human T(H)-17 responses. Nat Immunol. 2008;9:650-657.
- Boniface K, Blumenschein WM, Brovont-Porth K, et al. Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage. J Immunol. 2010;185:679-687.
- Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol. 2010;130:1373-1383.
- Boniface K, Bernard FX, Garcia M, et al. IL-22 inhibits epidermal differentiation and induces proinflammatory gene expression and migration of human keratinocytes. J Immunol. 2005;174:3695-3702.
- Harper EG, Guo C, Rizzo H, et al. Th17 cytokines stimulate CCL20 expression in keratinocytes in vitro and in vivo: implications for psoriasis pathogenesis. J Invest Dermatol. 2009;129:2175-2183.
- Bowcock AM, Krueger JG. Getting under the skin: the immunogenetics of psoriasis. Nat Rev Immunol. 2005;5:699-711.
- Hoffmann R, Wenzel E, Huth A, et al. Cytokine mRNA levels in alopecia areata before and after treatment with the contact allergen diphenylcyclopropenone. J Invest Dermatol. 1994;103:530-533.
To the Editor:
Both alopecia areata (AA) and psoriasis vulgaris are chronic relapsing autoimmune diseases, with AA causing nonscarring hair loss in approximately 0.1% to 0.2%1 of the population with a lifetime risk of 1.7%,2 and psoriasis more broadly impacting 1.5% to 2% of the population.3 The helper T cell (TH1) cytokine milieu is pathogenic in both conditions.4-6 IFN-γ knockout mice, unlike their wild-type counterparts, do not exhibit AA.7 Psoriasis is notably improved by IL-10 injections, which dampen the TH1 response.8 Distinct from AA, TH17 and TH22 cells have been implicated as key players in psoriasis pathogenesis, along with the associated IL-17 and IL-22 cytokines.9-12
Few cases of patients with concurrent AA and psoriasis have been described. Interestingly, these cases document normal hair regrowth in the areas of psoriasis.13-16 These cases may offer unique insight into the immune factors driving each disease. We describe a case of a man with both alopecia universalis (AU) and psoriasis who developed hair regrowth in some of the psoriatic plaques.
A 34-year-old man with concurrent AU and psoriasis who had not used any systemic or topical medication for either condition in the last year presented to our clinic seeking treatment. The patient had a history of alopecia totalis as a toddler that completely resolved by 4 years of age with the use of squaric acid dibutylester (SADBE). At 31 years of age, the alopecia recurred and was localized to the scalp. It was partially responsive to intralesional triamcinolone acetonide. The patient’s alopecia worsened over the 2 years following recurrence, ultimately progressing to AU. Two months after the alopecia recurrence, he developed the first psoriatic plaques. As the plaque psoriasis progressed, systemic therapy was initiated, first methotrexate and then etanercept. Shortly after developing AU, he lost his health insurance and discontinued all therapy. The patient’s psoriasis began to recur approximately 3 months after stopping etanercept. He was not using any other psoriasis medications. At that time, he noted terminal hair regrowth within some of the psoriatic plaques. No terminal hairs grew outside of the psoriatic plaques, and all regions with growth had previously been without hair for an extended period of time. The patient presented to our clinic approximately 1 year later. He had no other medical conditions and no relevant family history.
On initial physical examination, he had nonscarring hair loss involving nearly 100% of the body with psoriatic plaques on approximately 30% of the body surface area. Regions of terminal hair growth were confined to some but not all of the psoriatic plaques (Figure). Interestingly, the terminal hairs were primarily localized to the thickest central regions of the plaques. The patient’s psoriasis was treated with a combination of topical clobetasol and calcipotriene. In addition, he was started on tacrolimus ointment to the face and eyebrows for the AA. Maintenance of terminal hair within a region of topically treated psoriasis on the forearm persisted at the 2-month follow-up despite complete clearance of the corresponding psoriatic plaque. A small psoriatic plaque on the scalp cleared early with topical therapy without noticeable hair regrowth. The patient subsequently was started on contact immunotherapy with SADBE and intralesional triamcinolone acetonide for the scalp alopecia without satisfactory response. He decided to discontinue further attempts at treating the alopecia and requested to be restarted on etanercept therapy for recalcitrant psoriatic plaques. His psoriasis responded well to this therapy and he continues to be followed in our psoriasis clinic. One year after clearance of the treated psoriatic plaques, the corresponding terminal hairs persist.

Contact immunotherapy, most commonly with diphenylcyclopropenone or SADBE, is reported to have a 50% to 60% success rate in extensive AA, with a broad range of 9% to 87%17; however, randomized controlled trials testing the efficacy of contact immunotherapy are lacking. Although the mechanism of action of these topical sensitizers is not clearly delineated, it has been postulated that by inducing a new type of inflammatory response in the region, the immunologic milieu is changed, allowing the hair to grow. Some proposed mechanisms include promoting perifollicular lymphocyte apoptosis, preventing new recruitment of autoreactive lymphocytes, and allowing for the correction of aberrant major histocompatibility complex expression on the hair matrix epithelium to regain follicle immune privilege.18-20
Iatrogenic immunotherapy may work analogously to the natural immune system deviation demonstrated in our patient. Psoriasis and AA are believed to form competing immune cells and cytokine milieus, thus explaining how an individual with AA could regain normal hair growth in areas of psoriasis.15,16 The Renbök phenomenon, or reverse Köbner phenomenon, coined by Happle et al13 can be used to describe both the iatrogenic and natural cases of dermatologic disease improvement in response to secondary insults.14
A complex cascade of immune cells and cytokines coordinate AA pathogenesis. In the acute stage of AA, an inflammatory infiltrate of CD4+ T cells, CD8+ T cells, and antigen-presenting cells target anagen phase follicles, with a higher CD4+:CD8+ ratio in clinically active disease.21-23 Subcutaneous injections of either CD4+ or CD8+ lymphocyte subsets from mice with AA into normal-haired mice induces disease. However, CD8+ T cell injections rapidly produce apparent hair loss, whereas CD4+ T cells cause hair loss after several weeks, suggesting that CD8+ T cells directly modulate AA hair loss and CD4+ T cells act as an aide.24 The growth, differentiation, and survival of CD8+ T cells are stimulated by IL-2 and IFN-γ. Alopecia areata biopsies demonstrate a prevalence of TH1 cytokines, and patients with localized AA, alopecia totalis, and AU have notably higher serum IFN-γ levels compared to controls.25 In murine models, IL-1α and IL-1β increase during the catagen phase of the hair cycle and peak during the telogen phase.26 Excessive IL-1β expression is detected in the early stages of human disease, and certain IL-1β polymorphisms are associated with severe forms of AA.26 The role of tumor necrosis factor (TNF) α in AA is not well understood. In vitro studies show it inhibits hair growth, suggesting the cytokine may play a role in AA.27 However, anti–TNF-α therapy is not effective in AA, and case reports propose these therapies rarely induce AA.28-31
The TH1 response is likewise critical to psoriatic plaque development. IFN-γ and TNF-α are overexpressed in psoriatic plaques.32 IFN-γ has an antiproliferative and differentiation-inducing effect on normal keratinocytes, but psoriatic epithelial cells in vitro respond differently to the cytokine with a notably diminished growth inhibition.33,34 One explanation for the role of IFN-γ is that it stimulates dendritic cells to produce IL-1 and IL-23.35 IL-23 activates TH17 cells36; TH1 and TH17 conditions produce IL-22 whose serum level correlates with disease severity.37-39 IL-22 induces keratinocyte proliferation and migration and inhibits keratinocyte differentiation, helping account for hallmarks of the disease.40 Patients with psoriasis have increased levels of TH1, TH17, and TH22 cells, as well as their associated cytokines, in the skin and blood compared to controls.4,11,32,39,41
Alopecia areata and psoriasis are regulated by complex and still not entirely understood immune interactions. The fact that many of the same therapies are used to treat both diseases emphasizes both their overlapping characteristics and the lack of targeted therapy. It is unclear if and how the topical or systemic therapies used in our patient to treat one disease affected the natural history of the other condition. It is important to highlight, however, that the patient had not been treated for months when he developed the psoriatic plaques with hair regrowth. Other case reports also document hair regrowth in untreated plaques,13,16 making it unlikely to be a side effect of the medication regimen. For both psoriasis and AA, the immune cell composition and cytokine levels in the skin or serum vary throughout a patient’s disease course depending on severity of disease or response to treatment.6,39,42,43 Therefore, we hypothesize that the 2 conditions interact in a similarly distinct manner based on each disease’s stage and intensity in the patient. Both our patient’s course thus far and the various presentations described by other groups support this hypothesis. Our patient had a small region of psoriasis on the scalp that cleared without any terminal hair growth. He also had larger plaques on the forearms that developed hair growth most predominantly within the thicker regions of the plaques. His unique presentation highlights the fluidity of the immune factors driving psoriasis vulgaris and AA.
To the Editor:
Both alopecia areata (AA) and psoriasis vulgaris are chronic relapsing autoimmune diseases, with AA causing nonscarring hair loss in approximately 0.1% to 0.2%1 of the population with a lifetime risk of 1.7%,2 and psoriasis more broadly impacting 1.5% to 2% of the population.3 The helper T cell (TH1) cytokine milieu is pathogenic in both conditions.4-6 IFN-γ knockout mice, unlike their wild-type counterparts, do not exhibit AA.7 Psoriasis is notably improved by IL-10 injections, which dampen the TH1 response.8 Distinct from AA, TH17 and TH22 cells have been implicated as key players in psoriasis pathogenesis, along with the associated IL-17 and IL-22 cytokines.9-12
Few cases of patients with concurrent AA and psoriasis have been described. Interestingly, these cases document normal hair regrowth in the areas of psoriasis.13-16 These cases may offer unique insight into the immune factors driving each disease. We describe a case of a man with both alopecia universalis (AU) and psoriasis who developed hair regrowth in some of the psoriatic plaques.
A 34-year-old man with concurrent AU and psoriasis who had not used any systemic or topical medication for either condition in the last year presented to our clinic seeking treatment. The patient had a history of alopecia totalis as a toddler that completely resolved by 4 years of age with the use of squaric acid dibutylester (SADBE). At 31 years of age, the alopecia recurred and was localized to the scalp. It was partially responsive to intralesional triamcinolone acetonide. The patient’s alopecia worsened over the 2 years following recurrence, ultimately progressing to AU. Two months after the alopecia recurrence, he developed the first psoriatic plaques. As the plaque psoriasis progressed, systemic therapy was initiated, first methotrexate and then etanercept. Shortly after developing AU, he lost his health insurance and discontinued all therapy. The patient’s psoriasis began to recur approximately 3 months after stopping etanercept. He was not using any other psoriasis medications. At that time, he noted terminal hair regrowth within some of the psoriatic plaques. No terminal hairs grew outside of the psoriatic plaques, and all regions with growth had previously been without hair for an extended period of time. The patient presented to our clinic approximately 1 year later. He had no other medical conditions and no relevant family history.
On initial physical examination, he had nonscarring hair loss involving nearly 100% of the body with psoriatic plaques on approximately 30% of the body surface area. Regions of terminal hair growth were confined to some but not all of the psoriatic plaques (Figure). Interestingly, the terminal hairs were primarily localized to the thickest central regions of the plaques. The patient’s psoriasis was treated with a combination of topical clobetasol and calcipotriene. In addition, he was started on tacrolimus ointment to the face and eyebrows for the AA. Maintenance of terminal hair within a region of topically treated psoriasis on the forearm persisted at the 2-month follow-up despite complete clearance of the corresponding psoriatic plaque. A small psoriatic plaque on the scalp cleared early with topical therapy without noticeable hair regrowth. The patient subsequently was started on contact immunotherapy with SADBE and intralesional triamcinolone acetonide for the scalp alopecia without satisfactory response. He decided to discontinue further attempts at treating the alopecia and requested to be restarted on etanercept therapy for recalcitrant psoriatic plaques. His psoriasis responded well to this therapy and he continues to be followed in our psoriasis clinic. One year after clearance of the treated psoriatic plaques, the corresponding terminal hairs persist.

Contact immunotherapy, most commonly with diphenylcyclopropenone or SADBE, is reported to have a 50% to 60% success rate in extensive AA, with a broad range of 9% to 87%17; however, randomized controlled trials testing the efficacy of contact immunotherapy are lacking. Although the mechanism of action of these topical sensitizers is not clearly delineated, it has been postulated that by inducing a new type of inflammatory response in the region, the immunologic milieu is changed, allowing the hair to grow. Some proposed mechanisms include promoting perifollicular lymphocyte apoptosis, preventing new recruitment of autoreactive lymphocytes, and allowing for the correction of aberrant major histocompatibility complex expression on the hair matrix epithelium to regain follicle immune privilege.18-20
Iatrogenic immunotherapy may work analogously to the natural immune system deviation demonstrated in our patient. Psoriasis and AA are believed to form competing immune cells and cytokine milieus, thus explaining how an individual with AA could regain normal hair growth in areas of psoriasis.15,16 The Renbök phenomenon, or reverse Köbner phenomenon, coined by Happle et al13 can be used to describe both the iatrogenic and natural cases of dermatologic disease improvement in response to secondary insults.14
A complex cascade of immune cells and cytokines coordinate AA pathogenesis. In the acute stage of AA, an inflammatory infiltrate of CD4+ T cells, CD8+ T cells, and antigen-presenting cells target anagen phase follicles, with a higher CD4+:CD8+ ratio in clinically active disease.21-23 Subcutaneous injections of either CD4+ or CD8+ lymphocyte subsets from mice with AA into normal-haired mice induces disease. However, CD8+ T cell injections rapidly produce apparent hair loss, whereas CD4+ T cells cause hair loss after several weeks, suggesting that CD8+ T cells directly modulate AA hair loss and CD4+ T cells act as an aide.24 The growth, differentiation, and survival of CD8+ T cells are stimulated by IL-2 and IFN-γ. Alopecia areata biopsies demonstrate a prevalence of TH1 cytokines, and patients with localized AA, alopecia totalis, and AU have notably higher serum IFN-γ levels compared to controls.25 In murine models, IL-1α and IL-1β increase during the catagen phase of the hair cycle and peak during the telogen phase.26 Excessive IL-1β expression is detected in the early stages of human disease, and certain IL-1β polymorphisms are associated with severe forms of AA.26 The role of tumor necrosis factor (TNF) α in AA is not well understood. In vitro studies show it inhibits hair growth, suggesting the cytokine may play a role in AA.27 However, anti–TNF-α therapy is not effective in AA, and case reports propose these therapies rarely induce AA.28-31
The TH1 response is likewise critical to psoriatic plaque development. IFN-γ and TNF-α are overexpressed in psoriatic plaques.32 IFN-γ has an antiproliferative and differentiation-inducing effect on normal keratinocytes, but psoriatic epithelial cells in vitro respond differently to the cytokine with a notably diminished growth inhibition.33,34 One explanation for the role of IFN-γ is that it stimulates dendritic cells to produce IL-1 and IL-23.35 IL-23 activates TH17 cells36; TH1 and TH17 conditions produce IL-22 whose serum level correlates with disease severity.37-39 IL-22 induces keratinocyte proliferation and migration and inhibits keratinocyte differentiation, helping account for hallmarks of the disease.40 Patients with psoriasis have increased levels of TH1, TH17, and TH22 cells, as well as their associated cytokines, in the skin and blood compared to controls.4,11,32,39,41
Alopecia areata and psoriasis are regulated by complex and still not entirely understood immune interactions. The fact that many of the same therapies are used to treat both diseases emphasizes both their overlapping characteristics and the lack of targeted therapy. It is unclear if and how the topical or systemic therapies used in our patient to treat one disease affected the natural history of the other condition. It is important to highlight, however, that the patient had not been treated for months when he developed the psoriatic plaques with hair regrowth. Other case reports also document hair regrowth in untreated plaques,13,16 making it unlikely to be a side effect of the medication regimen. For both psoriasis and AA, the immune cell composition and cytokine levels in the skin or serum vary throughout a patient’s disease course depending on severity of disease or response to treatment.6,39,42,43 Therefore, we hypothesize that the 2 conditions interact in a similarly distinct manner based on each disease’s stage and intensity in the patient. Both our patient’s course thus far and the various presentations described by other groups support this hypothesis. Our patient had a small region of psoriasis on the scalp that cleared without any terminal hair growth. He also had larger plaques on the forearms that developed hair growth most predominantly within the thicker regions of the plaques. His unique presentation highlights the fluidity of the immune factors driving psoriasis vulgaris and AA.
- Safavi K. Prevalence of alopecia areata in the First National Health and Nutrition Examination Survey. Arch Dermatol. 1992;128:702.
- Safavi KH, Muller SA, Suman VJ, et al. Incidence of alopecia areata in Olmsted County, Minnesota, 1975 through 1989. Mayo Clin Proc. 1995;70:628-633.
- Wolff K, Johnson RA. Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. 6th ed. New York, NY: McGraw-Hill; 2009.
- Austin LM, Ozawa M, Kikuchi T, et al. The majority of epidermal T cells in psoriasis vulgaris lesions can produce type 1 cytokines, interferon-gamma, interleukin-2, and tumor necrosis factor-alpha, defining TC1 (cytotoxic T lymphocyte) and TH1 effector populations: a type 1 differentiation bias is also measured in circulating blood T cells in psoriatic patients. J Invest Dermatol. 1999;113:752-759.
- Ghoreishi M, Martinka M, Dutz JP. Type 1 interferon signature in the scalp lesions of alopecia areata. Br J Dermatol. 2010;163:57-62.
- Rossi A, Cantisani C, Carlesimo M, et al. Serum concentrations of IL-2, IL-6, IL-12 and TNF-α in patients with alopecia areata. Int J Immunopathol Pharmacol. 2012;25:781-788.
- Freyschmidt-Paul P, McElwee KJ, Hoffmann R, et al. Interferon-gamma-deficient mice are resistant to the development of alopecia areata. Br J Dermatol. 2006;155:515-521.
- Reich K, Garbe C, Blaschke V, et al. Response of psoriasis to interleukin-10 is associated with suppression of cutaneous type 1 inflammation, downregulation of the epidermal interleukin-8/CXCR2 pathway and normalization of keratinocyte maturation. J Invest Dermatol. 2001;116:319-329.
- Teunissen MB, Koomen CW, de Waal Malefyt R, et al. Interleukin-17 and interferon-gamma synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J Invest Dermatol. 1998;111:645-649.
- Zheng Y, Danilenko DM, Valdez P, et al. Interleukin-22, a T(H)17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature. 2007;445:648-651.
- Boniface K, Guignouard E, Pedretti N, et al. A role for T cell-derived interleukin 22 in psoriatic skin inflammation. Clin Exp Immunol. 2007;150:407-415.
- Zaba LC, Suárez-Fariñas M, Fuentes-Duculan J, et al. Effective treatment of psoriasis with etanercept is linked to suppression of IL-17 signaling, not immediate response TNF genes. J Allergy Clin Immunol. 2009;124:1022-1030.e395.
- Happle R, van der Steen PHM, Perret CM. The Renbök phenomenon: an inverse Köebner reaction observed in alopecia areata. Eur J Dermatol. 1991;2:39-40.
- Ito T, Hashizume H, Takigawa M. Contact immunotherapy-induced Renbök phenomenon in a patient with alopecia areata and psoriasis vulgaris. Eur J Dermatol. 2010;20:126-127.
- Criado PR, Valente NY, Michalany NS, et al. An unusual association between scalp psoriasis and ophiasic alopecia areata: the Renbök phenomenon. Clin Exp Dermatol. 2007;32:320-321.
- Harris JE, Seykora JT, Lee RA. Renbök phenomenon and contact sensitization in a patient with alopecia universalis. Arch Dermatol. 2010;146:422-425.
- Alkhalifah A. Topical and intralesional therapies for alopecia areata. Dermatol Ther. 2011;24:355-363.
- Herbst V, Zöller M, Kissling S, et al. Diphenylcyclopropenone treatment of alopecia areata induces apoptosis of perifollicular lymphocytes. Eur J Dermatol. 2006;16:537-542.
- Zöller M, Freyschmidt-Paul P, Vitacolonna M, et al. Chronic delayed-type hypersensitivity reaction as a means to treat alopecia areata. Clin Exp Immunol. 2004;135:398-408.
- Bröcker EB, Echternacht-Happle K, Hamm H, et al. Abnormal expression of class I and class II major histocompatibility antigens in alopecia areata: modulation by topical immunotherapy. J Invest Dermatol. 1987;88:564-568.
- Todes-Taylor N, Turner R, Wood GS, et al. T cell subpopulations in alopecia areata. J Am Acad Dermatol. 1984;11:216-223.
- Perret C, Wiesner-Menzel L, Happle R. Immunohistochemical analysis of T-cell subsets in the peribulbar and intrabulbar infiltrates of alopecia areata. Acta Derm Venereol. 1984;64:26-30.
- Wiesner-Menzel L, Happle R. Intrabulbar and peribulbar accumulation of dendritic OKT 6-positive cells in alopecia areata. Arch Dermatol Res. 1984;276:333-334.
- McElwee KJ, Freyschmidt-Paul P, Hoffmann R, et al. Transfer of CD8+ cells induces localized hair loss whereas CD4+/CD25– cells promote systemic alopecia areata and CD4+/CD25+ cells blockade disease onset in the C3H/HeJ mouse model. J Invest Dermatol. 2005;124:947-957.
- Arca E, Muşabak U, Akar A, et al. Interferon-gamma in alopecia areata. Eur J Dermatol. 2004;14:33-36.
- Hoffmann R. The potential role of cytokines and T cells in alopecia areata. J Investig Dermatol Symp Proc. 1999;4:235-238.
- Philpott MP, Sanders DA, Bowen J, et al. Effects of interleukins, colony-stimulating factor and tumour necrosis factor on human hair follicle growth in vitro: a possible role for interleukin-1 and tumour necrosis factor-alpha in alopecia areata. Br J Dermatol. 1996;135:942-948.
- Le Bidre E, Chaby G, Martin L, et al. Alopecia areata during anti-TNF alpha therapy: nine cases. Ann Dermatol Venereol. 2011;138:285-293.
- Ferran M, Calvet J, Almirall M, et al. Alopecia areata as another immune-mediated disease developed in patients treated with tumour necrosis factor-α blocker agents: report of five cases and review of the literature. J Eur Acad Dermatol Venereol. 2011;25:479-484.
- Pan Y, Rao NA. Alopecia areata during etanercept therapy. Ocul Immunol Inflamm. 2009;17:127-129.
- Pelivani N, Hassan AS, Braathen LR, et al. Alopecia areata universalis elicited during treatment with adalimumab. Dermatology. 2008;216:320-323.
- Uyemura K, Yamamura M, Fivenson DF, et al. The cytokine network in lesional and lesion-free psoriatic skin is characterized by a T-helper type 1 cell-mediated response. J Invest Dermatol. 1993;101:701-705.
- Baker BS, Powles AV, Valdimarsson H, et al. An altered response by psoriatic keratinocytes to gamma interferon. Scan J Immunol. 1988;28:735-740.
- Jackson M, Howie SE, Weller R, et al. Psoriatic keratinocytes show reduced IRF-1 and STAT-1alpha activation in response to gamma-IFN. FASEB J. 1999;13:495-502.
- Perera GK, Di Meglio P, Nestle FO. Psoriasis. Annu Rev Pathol. 2012;7:385-422.
- McGeachy MJ, Chen Y, Tato CM, et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol. 2009;10:314-324.
- Volpe E, Servant N, Zollinger R, et al. A critical function for transforming growth factor-beta, interleukin 23 and proinflammatory cytokines in driving and modulating human T(H)-17 responses. Nat Immunol. 2008;9:650-657.
- Boniface K, Blumenschein WM, Brovont-Porth K, et al. Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage. J Immunol. 2010;185:679-687.
- Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol. 2010;130:1373-1383.
- Boniface K, Bernard FX, Garcia M, et al. IL-22 inhibits epidermal differentiation and induces proinflammatory gene expression and migration of human keratinocytes. J Immunol. 2005;174:3695-3702.
- Harper EG, Guo C, Rizzo H, et al. Th17 cytokines stimulate CCL20 expression in keratinocytes in vitro and in vivo: implications for psoriasis pathogenesis. J Invest Dermatol. 2009;129:2175-2183.
- Bowcock AM, Krueger JG. Getting under the skin: the immunogenetics of psoriasis. Nat Rev Immunol. 2005;5:699-711.
- Hoffmann R, Wenzel E, Huth A, et al. Cytokine mRNA levels in alopecia areata before and after treatment with the contact allergen diphenylcyclopropenone. J Invest Dermatol. 1994;103:530-533.
- Safavi K. Prevalence of alopecia areata in the First National Health and Nutrition Examination Survey. Arch Dermatol. 1992;128:702.
- Safavi KH, Muller SA, Suman VJ, et al. Incidence of alopecia areata in Olmsted County, Minnesota, 1975 through 1989. Mayo Clin Proc. 1995;70:628-633.
- Wolff K, Johnson RA. Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. 6th ed. New York, NY: McGraw-Hill; 2009.
- Austin LM, Ozawa M, Kikuchi T, et al. The majority of epidermal T cells in psoriasis vulgaris lesions can produce type 1 cytokines, interferon-gamma, interleukin-2, and tumor necrosis factor-alpha, defining TC1 (cytotoxic T lymphocyte) and TH1 effector populations: a type 1 differentiation bias is also measured in circulating blood T cells in psoriatic patients. J Invest Dermatol. 1999;113:752-759.
- Ghoreishi M, Martinka M, Dutz JP. Type 1 interferon signature in the scalp lesions of alopecia areata. Br J Dermatol. 2010;163:57-62.
- Rossi A, Cantisani C, Carlesimo M, et al. Serum concentrations of IL-2, IL-6, IL-12 and TNF-α in patients with alopecia areata. Int J Immunopathol Pharmacol. 2012;25:781-788.
- Freyschmidt-Paul P, McElwee KJ, Hoffmann R, et al. Interferon-gamma-deficient mice are resistant to the development of alopecia areata. Br J Dermatol. 2006;155:515-521.
- Reich K, Garbe C, Blaschke V, et al. Response of psoriasis to interleukin-10 is associated with suppression of cutaneous type 1 inflammation, downregulation of the epidermal interleukin-8/CXCR2 pathway and normalization of keratinocyte maturation. J Invest Dermatol. 2001;116:319-329.
- Teunissen MB, Koomen CW, de Waal Malefyt R, et al. Interleukin-17 and interferon-gamma synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J Invest Dermatol. 1998;111:645-649.
- Zheng Y, Danilenko DM, Valdez P, et al. Interleukin-22, a T(H)17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature. 2007;445:648-651.
- Boniface K, Guignouard E, Pedretti N, et al. A role for T cell-derived interleukin 22 in psoriatic skin inflammation. Clin Exp Immunol. 2007;150:407-415.
- Zaba LC, Suárez-Fariñas M, Fuentes-Duculan J, et al. Effective treatment of psoriasis with etanercept is linked to suppression of IL-17 signaling, not immediate response TNF genes. J Allergy Clin Immunol. 2009;124:1022-1030.e395.
- Happle R, van der Steen PHM, Perret CM. The Renbök phenomenon: an inverse Köebner reaction observed in alopecia areata. Eur J Dermatol. 1991;2:39-40.
- Ito T, Hashizume H, Takigawa M. Contact immunotherapy-induced Renbök phenomenon in a patient with alopecia areata and psoriasis vulgaris. Eur J Dermatol. 2010;20:126-127.
- Criado PR, Valente NY, Michalany NS, et al. An unusual association between scalp psoriasis and ophiasic alopecia areata: the Renbök phenomenon. Clin Exp Dermatol. 2007;32:320-321.
- Harris JE, Seykora JT, Lee RA. Renbök phenomenon and contact sensitization in a patient with alopecia universalis. Arch Dermatol. 2010;146:422-425.
- Alkhalifah A. Topical and intralesional therapies for alopecia areata. Dermatol Ther. 2011;24:355-363.
- Herbst V, Zöller M, Kissling S, et al. Diphenylcyclopropenone treatment of alopecia areata induces apoptosis of perifollicular lymphocytes. Eur J Dermatol. 2006;16:537-542.
- Zöller M, Freyschmidt-Paul P, Vitacolonna M, et al. Chronic delayed-type hypersensitivity reaction as a means to treat alopecia areata. Clin Exp Immunol. 2004;135:398-408.
- Bröcker EB, Echternacht-Happle K, Hamm H, et al. Abnormal expression of class I and class II major histocompatibility antigens in alopecia areata: modulation by topical immunotherapy. J Invest Dermatol. 1987;88:564-568.
- Todes-Taylor N, Turner R, Wood GS, et al. T cell subpopulations in alopecia areata. J Am Acad Dermatol. 1984;11:216-223.
- Perret C, Wiesner-Menzel L, Happle R. Immunohistochemical analysis of T-cell subsets in the peribulbar and intrabulbar infiltrates of alopecia areata. Acta Derm Venereol. 1984;64:26-30.
- Wiesner-Menzel L, Happle R. Intrabulbar and peribulbar accumulation of dendritic OKT 6-positive cells in alopecia areata. Arch Dermatol Res. 1984;276:333-334.
- McElwee KJ, Freyschmidt-Paul P, Hoffmann R, et al. Transfer of CD8+ cells induces localized hair loss whereas CD4+/CD25– cells promote systemic alopecia areata and CD4+/CD25+ cells blockade disease onset in the C3H/HeJ mouse model. J Invest Dermatol. 2005;124:947-957.
- Arca E, Muşabak U, Akar A, et al. Interferon-gamma in alopecia areata. Eur J Dermatol. 2004;14:33-36.
- Hoffmann R. The potential role of cytokines and T cells in alopecia areata. J Investig Dermatol Symp Proc. 1999;4:235-238.
- Philpott MP, Sanders DA, Bowen J, et al. Effects of interleukins, colony-stimulating factor and tumour necrosis factor on human hair follicle growth in vitro: a possible role for interleukin-1 and tumour necrosis factor-alpha in alopecia areata. Br J Dermatol. 1996;135:942-948.
- Le Bidre E, Chaby G, Martin L, et al. Alopecia areata during anti-TNF alpha therapy: nine cases. Ann Dermatol Venereol. 2011;138:285-293.
- Ferran M, Calvet J, Almirall M, et al. Alopecia areata as another immune-mediated disease developed in patients treated with tumour necrosis factor-α blocker agents: report of five cases and review of the literature. J Eur Acad Dermatol Venereol. 2011;25:479-484.
- Pan Y, Rao NA. Alopecia areata during etanercept therapy. Ocul Immunol Inflamm. 2009;17:127-129.
- Pelivani N, Hassan AS, Braathen LR, et al. Alopecia areata universalis elicited during treatment with adalimumab. Dermatology. 2008;216:320-323.
- Uyemura K, Yamamura M, Fivenson DF, et al. The cytokine network in lesional and lesion-free psoriatic skin is characterized by a T-helper type 1 cell-mediated response. J Invest Dermatol. 1993;101:701-705.
- Baker BS, Powles AV, Valdimarsson H, et al. An altered response by psoriatic keratinocytes to gamma interferon. Scan J Immunol. 1988;28:735-740.
- Jackson M, Howie SE, Weller R, et al. Psoriatic keratinocytes show reduced IRF-1 and STAT-1alpha activation in response to gamma-IFN. FASEB J. 1999;13:495-502.
- Perera GK, Di Meglio P, Nestle FO. Psoriasis. Annu Rev Pathol. 2012;7:385-422.
- McGeachy MJ, Chen Y, Tato CM, et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol. 2009;10:314-324.
- Volpe E, Servant N, Zollinger R, et al. A critical function for transforming growth factor-beta, interleukin 23 and proinflammatory cytokines in driving and modulating human T(H)-17 responses. Nat Immunol. 2008;9:650-657.
- Boniface K, Blumenschein WM, Brovont-Porth K, et al. Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage. J Immunol. 2010;185:679-687.
- Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol. 2010;130:1373-1383.
- Boniface K, Bernard FX, Garcia M, et al. IL-22 inhibits epidermal differentiation and induces proinflammatory gene expression and migration of human keratinocytes. J Immunol. 2005;174:3695-3702.
- Harper EG, Guo C, Rizzo H, et al. Th17 cytokines stimulate CCL20 expression in keratinocytes in vitro and in vivo: implications for psoriasis pathogenesis. J Invest Dermatol. 2009;129:2175-2183.
- Bowcock AM, Krueger JG. Getting under the skin: the immunogenetics of psoriasis. Nat Rev Immunol. 2005;5:699-711.
- Hoffmann R, Wenzel E, Huth A, et al. Cytokine mRNA levels in alopecia areata before and after treatment with the contact allergen diphenylcyclopropenone. J Invest Dermatol. 1994;103:530-533.
Practice Points
- The Renbök phenomenon, or reverse Köbner phenomenon, describes cases where secondary insults improve dermatologic disease.
- Current evidence suggests that alopecia areata (AA) is driven by a helper T cell (TH1) response whereas psoriasis vulgaris is driven by TH1, TH17, and TH22.
- Patients with concurrent AA and psoriasis can develop normal hair regrowth confined to the psoriatic plaques. Developing methods to artificially alter the cytokine milieu in affected skin may lead to new therapeutic options for each condition.
Study finds psoriasis link to melanoma and hematologic cancers
Patients with psoriasis may be at a greater risk of melanoma and hematologic cancers, compared with the general population, but treatments do not appear to increase risk, according to Shivani P. Reddy of the University of Illinois at Chicago, and associates.
In a retrospective cohort study, they identified 815,765 patients at Kaiser Permanente Southern California who had at least one medical encounter from January 2004 through December 2013. Of these patients, 8,161 (1%) met the diagnostic and inclusion criteria for psoriasis, and there were 7,167 (0.89%) cases of melanoma and 5,399 (0.66%) cases of lymphoma or leukemia.
Among the patients with psoriasis, there were 62 (0.87%) melanoma cases and 47 (0.87%) cases of lymphoma or leukemia.
Of the 109 patients with psoriasis who went on to develop melanoma or lymphoma, the time to diagnosis of melanoma or hematologic cancers was significantly less for patients with psoriasis than for patients without psoriasis (P = .01). The patients with psoriasis had a 1.53 times higher risk of developing a malignancy compared with patients without psoriasis (P less than .01) in the multivariable Cox proportional hazards model.
“There were no differences between patients with melanoma and hematologic cancer by treatment type,” which were phototherapy, tumor necrosis factor inhibitor therapy, and topical medications, they wrote.
“Our study demonstrates that the risk of hematologic cancer and melanoma is increased in patients with psoriasis over time, although this risk is not impacted by psoriasis therapies,” the researchers concluded. “Defining the risk of malignancy in these patients is important for proper workup and management.”
Read the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2016.09.047).
Patients with psoriasis may be at a greater risk of melanoma and hematologic cancers, compared with the general population, but treatments do not appear to increase risk, according to Shivani P. Reddy of the University of Illinois at Chicago, and associates.
In a retrospective cohort study, they identified 815,765 patients at Kaiser Permanente Southern California who had at least one medical encounter from January 2004 through December 2013. Of these patients, 8,161 (1%) met the diagnostic and inclusion criteria for psoriasis, and there were 7,167 (0.89%) cases of melanoma and 5,399 (0.66%) cases of lymphoma or leukemia.
Among the patients with psoriasis, there were 62 (0.87%) melanoma cases and 47 (0.87%) cases of lymphoma or leukemia.
Of the 109 patients with psoriasis who went on to develop melanoma or lymphoma, the time to diagnosis of melanoma or hematologic cancers was significantly less for patients with psoriasis than for patients without psoriasis (P = .01). The patients with psoriasis had a 1.53 times higher risk of developing a malignancy compared with patients without psoriasis (P less than .01) in the multivariable Cox proportional hazards model.
“There were no differences between patients with melanoma and hematologic cancer by treatment type,” which were phototherapy, tumor necrosis factor inhibitor therapy, and topical medications, they wrote.
“Our study demonstrates that the risk of hematologic cancer and melanoma is increased in patients with psoriasis over time, although this risk is not impacted by psoriasis therapies,” the researchers concluded. “Defining the risk of malignancy in these patients is important for proper workup and management.”
Read the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2016.09.047).
Patients with psoriasis may be at a greater risk of melanoma and hematologic cancers, compared with the general population, but treatments do not appear to increase risk, according to Shivani P. Reddy of the University of Illinois at Chicago, and associates.
In a retrospective cohort study, they identified 815,765 patients at Kaiser Permanente Southern California who had at least one medical encounter from January 2004 through December 2013. Of these patients, 8,161 (1%) met the diagnostic and inclusion criteria for psoriasis, and there were 7,167 (0.89%) cases of melanoma and 5,399 (0.66%) cases of lymphoma or leukemia.
Among the patients with psoriasis, there were 62 (0.87%) melanoma cases and 47 (0.87%) cases of lymphoma or leukemia.
Of the 109 patients with psoriasis who went on to develop melanoma or lymphoma, the time to diagnosis of melanoma or hematologic cancers was significantly less for patients with psoriasis than for patients without psoriasis (P = .01). The patients with psoriasis had a 1.53 times higher risk of developing a malignancy compared with patients without psoriasis (P less than .01) in the multivariable Cox proportional hazards model.
“There were no differences between patients with melanoma and hematologic cancer by treatment type,” which were phototherapy, tumor necrosis factor inhibitor therapy, and topical medications, they wrote.
“Our study demonstrates that the risk of hematologic cancer and melanoma is increased in patients with psoriasis over time, although this risk is not impacted by psoriasis therapies,” the researchers concluded. “Defining the risk of malignancy in these patients is important for proper workup and management.”
Read the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2016.09.047).
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Risankizumab tops ustekinumab in phase II psoriasis trial
At 12 weeks, 77% of psoriasis patients treated with risankizumab showed a 90% or greater reduction in Psoriasis Area and Severity Index (PASI) score, compared with 40% of ustekinumab patients, in a phase II randomized trial.
The results were published April 19 in the New England Journal of Medicine.
Ustekinumab (Stelara), approved by the Food and Drug Administration in 2009, blocks interleukin-12 and interleukin-23 and has demonstrated effectiveness in psoriasis patients. However, the humanized IgG1 monoclonal antibody risankizumab goes farther and “selectively inhibits interleukin-23 by specifically targeting p19,” wrote Kim A. Papp, MD, PhD, of K. Papp Clinical Research and Probity Medical Research, Waterloo, Ont., and associates (N. Engl. J. Med. 2017;376:1551-60. doi: 10.1056/NEJMoa1607017).
To compare clinical response and safety, the researchers enrolled 166 adults aged 18-75 years with moderate to severe plaque psoriasis, in the phase II study. Patients were randomized to subcutaneous injections of risankizumab at one of three doses, or ustekinumab at one of two doses. Risankizumab patients received a single 18-mg dose at week 0, or 90-mg or 180-mg doses at weeks 0, 4, and 16. Ustekinumab patients weighing 100 kg or less received 45 mg at weeks 0, 4, and 16; those weighing more than 100 kg received 90 mg at weeks 0, 4, and 16. Demographics were similar among the treatment groups.
The primary end point was a 90% or greater reduction in the PASI score at week 12, compared with baseline.
In pooled results of the risankizumab 90-mg and 180-mg groups, 77% of patients achieved a PASI 90 at 12 weeks (73% of the 90-mg group and 81% of the 180-mg group), vs. 40% of ustekinumab patients (P less than .001). Complete clearance of lesions (PASI 100) occurred among risankizumab patients in 14% of the 18-mg group, 41% of the 90-mg group, and 48% of the 180-mg group, compared with 18% of the ustekinumab group.
Among risankizumab patients, the rates of adverse events through 48 weeks were 81% in the 180-mg group, 80% in the 90-mg group, and 69% in the 180-mg group, compared with 72% in those on ustekinumab, with nasopharyngitis the most commonly reported adverse event in all the treatment groups. The rates of serious adverse events were 12% and 15% among those in the 18-mg and 90-mg risankizumab groups, respectively; 0% among those on the 180-mg dose, and 8% among those on ustekinumab.
The study was not large or long enough to provide conclusive safety data on risankizumab, and additional studies are needed to review psoriasis lesions over a longer time period and include both placebo and active comparators, researchers noted. However, the results suggest that “the selective blockade of interleukin-23 through the inhibition of the p19 subunit rather than p40 provides a more complete inhibition of interleukin-23 activity, potentially resulting in a greater efficacy in the treatment of plaque psoriasis at the doses used,” they said.
The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies. Several authors are Boehringer Ingelheim employees.
At 12 weeks, 77% of psoriasis patients treated with risankizumab showed a 90% or greater reduction in Psoriasis Area and Severity Index (PASI) score, compared with 40% of ustekinumab patients, in a phase II randomized trial.
The results were published April 19 in the New England Journal of Medicine.
Ustekinumab (Stelara), approved by the Food and Drug Administration in 2009, blocks interleukin-12 and interleukin-23 and has demonstrated effectiveness in psoriasis patients. However, the humanized IgG1 monoclonal antibody risankizumab goes farther and “selectively inhibits interleukin-23 by specifically targeting p19,” wrote Kim A. Papp, MD, PhD, of K. Papp Clinical Research and Probity Medical Research, Waterloo, Ont., and associates (N. Engl. J. Med. 2017;376:1551-60. doi: 10.1056/NEJMoa1607017).
To compare clinical response and safety, the researchers enrolled 166 adults aged 18-75 years with moderate to severe plaque psoriasis, in the phase II study. Patients were randomized to subcutaneous injections of risankizumab at one of three doses, or ustekinumab at one of two doses. Risankizumab patients received a single 18-mg dose at week 0, or 90-mg or 180-mg doses at weeks 0, 4, and 16. Ustekinumab patients weighing 100 kg or less received 45 mg at weeks 0, 4, and 16; those weighing more than 100 kg received 90 mg at weeks 0, 4, and 16. Demographics were similar among the treatment groups.
The primary end point was a 90% or greater reduction in the PASI score at week 12, compared with baseline.
In pooled results of the risankizumab 90-mg and 180-mg groups, 77% of patients achieved a PASI 90 at 12 weeks (73% of the 90-mg group and 81% of the 180-mg group), vs. 40% of ustekinumab patients (P less than .001). Complete clearance of lesions (PASI 100) occurred among risankizumab patients in 14% of the 18-mg group, 41% of the 90-mg group, and 48% of the 180-mg group, compared with 18% of the ustekinumab group.
Among risankizumab patients, the rates of adverse events through 48 weeks were 81% in the 180-mg group, 80% in the 90-mg group, and 69% in the 180-mg group, compared with 72% in those on ustekinumab, with nasopharyngitis the most commonly reported adverse event in all the treatment groups. The rates of serious adverse events were 12% and 15% among those in the 18-mg and 90-mg risankizumab groups, respectively; 0% among those on the 180-mg dose, and 8% among those on ustekinumab.
The study was not large or long enough to provide conclusive safety data on risankizumab, and additional studies are needed to review psoriasis lesions over a longer time period and include both placebo and active comparators, researchers noted. However, the results suggest that “the selective blockade of interleukin-23 through the inhibition of the p19 subunit rather than p40 provides a more complete inhibition of interleukin-23 activity, potentially resulting in a greater efficacy in the treatment of plaque psoriasis at the doses used,” they said.
The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies. Several authors are Boehringer Ingelheim employees.
At 12 weeks, 77% of psoriasis patients treated with risankizumab showed a 90% or greater reduction in Psoriasis Area and Severity Index (PASI) score, compared with 40% of ustekinumab patients, in a phase II randomized trial.
The results were published April 19 in the New England Journal of Medicine.
Ustekinumab (Stelara), approved by the Food and Drug Administration in 2009, blocks interleukin-12 and interleukin-23 and has demonstrated effectiveness in psoriasis patients. However, the humanized IgG1 monoclonal antibody risankizumab goes farther and “selectively inhibits interleukin-23 by specifically targeting p19,” wrote Kim A. Papp, MD, PhD, of K. Papp Clinical Research and Probity Medical Research, Waterloo, Ont., and associates (N. Engl. J. Med. 2017;376:1551-60. doi: 10.1056/NEJMoa1607017).
To compare clinical response and safety, the researchers enrolled 166 adults aged 18-75 years with moderate to severe plaque psoriasis, in the phase II study. Patients were randomized to subcutaneous injections of risankizumab at one of three doses, or ustekinumab at one of two doses. Risankizumab patients received a single 18-mg dose at week 0, or 90-mg or 180-mg doses at weeks 0, 4, and 16. Ustekinumab patients weighing 100 kg or less received 45 mg at weeks 0, 4, and 16; those weighing more than 100 kg received 90 mg at weeks 0, 4, and 16. Demographics were similar among the treatment groups.
The primary end point was a 90% or greater reduction in the PASI score at week 12, compared with baseline.
In pooled results of the risankizumab 90-mg and 180-mg groups, 77% of patients achieved a PASI 90 at 12 weeks (73% of the 90-mg group and 81% of the 180-mg group), vs. 40% of ustekinumab patients (P less than .001). Complete clearance of lesions (PASI 100) occurred among risankizumab patients in 14% of the 18-mg group, 41% of the 90-mg group, and 48% of the 180-mg group, compared with 18% of the ustekinumab group.
Among risankizumab patients, the rates of adverse events through 48 weeks were 81% in the 180-mg group, 80% in the 90-mg group, and 69% in the 180-mg group, compared with 72% in those on ustekinumab, with nasopharyngitis the most commonly reported adverse event in all the treatment groups. The rates of serious adverse events were 12% and 15% among those in the 18-mg and 90-mg risankizumab groups, respectively; 0% among those on the 180-mg dose, and 8% among those on ustekinumab.
The study was not large or long enough to provide conclusive safety data on risankizumab, and additional studies are needed to review psoriasis lesions over a longer time period and include both placebo and active comparators, researchers noted. However, the results suggest that “the selective blockade of interleukin-23 through the inhibition of the p19 subunit rather than p40 provides a more complete inhibition of interleukin-23 activity, potentially resulting in a greater efficacy in the treatment of plaque psoriasis at the doses used,” they said.
The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies. Several authors are Boehringer Ingelheim employees.
Key clinical point: Clinical responses in psoriasis patients treated with risankizumab were superior to responses in patients treated with ustekinumab.
Major finding: At 12 weeks, 77% of risankizumab patients showed a 90% or greater reduction in PASI score, compared with 40% of ustekinumab patients.
Data source: A phase II randomized trial of 166 adults with moderate to severe plaque psoriasis.
Disclosures: The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies; several were employees of Boehringer.
How to get the most out of methotrexate for psoriasis
WAILEA, HAWAII – In an era when affordable health insurance could become increasingly tough to come by, it’s worth emphasizing that methotrexate is the most cost-effective way to manage extensive psoriasis – and the liquid formulation designed for intramuscular injection can be taken orally to reduce the cost even further, according to Craig L. Leonardi, MD.
“The absolute cheapest way to manage the patient who has no insurance and has bad psoriasis is to put him on methotrexate and teach him how to draw the liquid solution up in a syringe, dump it in a cup of juice, and drink it,” Dr. Leonardi said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Research Foundation. “The bioavailability is equivalent to [that of] the tablets, and it’s only about one-tenth the cost.”
He urged his fellow dermatologists not to forget about methotrexate in the current flashy era of highly effective – and very expensive – biologic therapies for psoriasis.
“I was thinking methotrexate was going to go away, but it turns out I rely more on that drug than ever before. It’s very useful, and it’s safe if used correctly,” said Dr. Leonardi, a dermatologist at Saint Louis University and a prominent clinical trialist.
He highlighted numerous clinical scenarios in which methotrexate remains a valuable treatment in patients with moderate to severe psoriasis. He also touched on patient monitoring requirements, adverse events, and common reasons he receives referrals from physicians whose patients seem to be having problems on methotrexate – referrals that, in most cases, could be avoided, he said, if the referring physician had a fuller understanding of the drug.
“Patients are referred to me all the time for methotrexate intolerance,” Dr. Leonardi said. “When you take methotrexate, you get a brief, dramatic spike in transaminase levels that peaks within a day and then drops off. But, when you get lab tests in these patients, you want to look at trough levels. You want to see the best liver function test values for the week.
“That means you have to tell the patient what day to take the drug and what day to get labs drawn,” he continued. “I’m here to tell you that the vast majority of issues that I see where patients have elevated liver function tests involves them getting their testing done in the first 4 days after taking methotrexate. Take the time to ask about this, and I think you’ll be pleasantly surprised.
“If you just make an adjustment and get them on the right schedule, you’ll discover that the patient is tolerating the drug just fine,” Dr. Leonardi added. “We like getting labs on Monday and dosing on Tuesday.”
Methotrexate’s half-life is 3-15 hours. Psoriasis is often controlled at a methotrexate dose of about 15 mg/week.
Methotrexate’s advantages include ease of use. It’s a straightforward matter to start and stop the drug and to make small dose adjustments. Methotrexate comes in a variety of formulations: the 2.5-mg tablets, the 25 mg/mL solution for IM injection, and prefilled autoinjector devices for subcutaneous administration.
These preloaded pens for subcutaneous injection are just as effective as IM therapy, Dr. Leonardi said. Methotrexate is also better absorbed subcutaneously than it is orally. The bioavailability of oral methotrexate plateaus at a dose of about 22 mg/week, while subcutaneously delivered methotrexate does not. So, if a patient’s psoriasis isn’t adequately controlled on higher-dose oral therapy, it’s worth considering a switch to the preloaded pens.
“It’s a very simple injection, very cost-effective, and your patients may get more bang for the buck by doing that,” according to the dermatologist.
Also, nausea, vomiting, diarrhea, and abdominal pain have been shown to be significantly less frequent and intense with subcutaneous methotrexate than with the tablets. So, if a patient is experiencing limiting gastrointestinal issues on methotrexate tablets, a shift to subcutaneous therapy is often the solution.
What kind of efficacy can physicians expect with methotrexate monotherapy?
Dr. Leonardi cited the results of the European randomized, open-label RESTORE-1 trial (Br J Dermatol. 2011 Nov;165[5]:1109-17) as being consistent with his own extensive clinical experience: a week-16 PASI (Psoriasis Area and Severity Index ) 75 response rate of 42% with methotrexate, compared with 78% for infliximab (Remicade).
Of course, some patients can’t receive a biologic agent because of their age, lack of insurance coverage, or medical contraindications.
“I use methotrexate a lot in Medicare patients, where, with Part D, it’s hard to get access to biologics without really good coinsurance. I think all of us who prescribe biologics understand that,” he observed.
In pediatric patients with extensive psoriasis, he turns to methotrexate as first-line systemic therapy. After 3 months, if the young patient hasn’t responded satisfactorily, Dr. Leonardi asks the insurance company for access to a biologic agent and usually gets it.
Methotrexate really shines in combination with a biologic agent. It inhibits formation of antibiologic antibodies, an important cause of loss of effectiveness of monoclonal antibody therapy.
In one study, 28% of patients on adalimumab (Humira) developed antiadalimumab antibodies during the first 3 years of therapy. These patients were more likely to drop out of therapy for lack of effectiveness.
A key finding in this study was that two-thirds of patients who developed antiadalimumab antibodies did so during the first 28 weeks of therapy (JAMA. 2011 Apr 13;305[14]:1460-8). This time line has influenced Dr. Leonardi’s own clinical practice. He routinely keeps patients on methotrexate for their first 28-36 weeks on a biologic, then tapers the methotrexate in favor of biologic monotherapy.
Other benefits and guidelines
Methotrexate is also a boon in managing psoriasis flares in patients on a biologic.
“We’ve all had patients who are doing well on a biologic, they’re cruising along at 140 weeks, then they get strep throat or another infection, and, next thing you know, you have destabilized psoriasis,” Dr. Leonardi noted.
“One thing I’ll do is add methotrexate to the mix, try to get things under control, and, if we do, then we’ll try to taper the methotrexate,” he continued. “That whole episode might take 4-6 months to resolve before the patient might be able to tolerate biologic monotherapy, though. If they can’t at that point, you might consider rotating to another biologic.”
Current American Academy of Dermatology guidelines recommend that women on methotrexate limit their alcohol intake to one drink per day, two drinks for men. British Society for Rheumatology guidelines recommend a ceiling of 32 g to 64 g of ethanol per week.
“We don’t insist on abstinence. There’s no good evidence that it’s needed,” Dr. Leonardi noted. “If you dose this drug on Tuesday, you can be sure that it’s eliminated from the body on Friday, and that’s when I’ll generally green-light patients to socialize over the weekend. If you can make life a little more tolerable for your patients and they’re willing to follow your instructions, I think it’s a better deal all the way around.”
Prior to initiating methotrexate, he obtains a CBC with platelet count, liver function tests, serum urea nitrogen, creatinine, and screens for latent tuberculosis. In terms of on-treatment monitoring, he gets a CBC and liver function tests every 4-12 weeks and keeps an eye on renal function, especially in older patients, because methotrexate is eliminated renally.
“The guidelines have relaxed regarding the need for liver biopsies,” Dr. Leonardi said. “Most of us are not getting liver biopsies anymore, as is true of our friends in rheumatology.”
He reported having financial relationships with more than a dozen pharmaceutical companies. The SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – In an era when affordable health insurance could become increasingly tough to come by, it’s worth emphasizing that methotrexate is the most cost-effective way to manage extensive psoriasis – and the liquid formulation designed for intramuscular injection can be taken orally to reduce the cost even further, according to Craig L. Leonardi, MD.
“The absolute cheapest way to manage the patient who has no insurance and has bad psoriasis is to put him on methotrexate and teach him how to draw the liquid solution up in a syringe, dump it in a cup of juice, and drink it,” Dr. Leonardi said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Research Foundation. “The bioavailability is equivalent to [that of] the tablets, and it’s only about one-tenth the cost.”
He urged his fellow dermatologists not to forget about methotrexate in the current flashy era of highly effective – and very expensive – biologic therapies for psoriasis.
“I was thinking methotrexate was going to go away, but it turns out I rely more on that drug than ever before. It’s very useful, and it’s safe if used correctly,” said Dr. Leonardi, a dermatologist at Saint Louis University and a prominent clinical trialist.
He highlighted numerous clinical scenarios in which methotrexate remains a valuable treatment in patients with moderate to severe psoriasis. He also touched on patient monitoring requirements, adverse events, and common reasons he receives referrals from physicians whose patients seem to be having problems on methotrexate – referrals that, in most cases, could be avoided, he said, if the referring physician had a fuller understanding of the drug.
“Patients are referred to me all the time for methotrexate intolerance,” Dr. Leonardi said. “When you take methotrexate, you get a brief, dramatic spike in transaminase levels that peaks within a day and then drops off. But, when you get lab tests in these patients, you want to look at trough levels. You want to see the best liver function test values for the week.
“That means you have to tell the patient what day to take the drug and what day to get labs drawn,” he continued. “I’m here to tell you that the vast majority of issues that I see where patients have elevated liver function tests involves them getting their testing done in the first 4 days after taking methotrexate. Take the time to ask about this, and I think you’ll be pleasantly surprised.
“If you just make an adjustment and get them on the right schedule, you’ll discover that the patient is tolerating the drug just fine,” Dr. Leonardi added. “We like getting labs on Monday and dosing on Tuesday.”
Methotrexate’s half-life is 3-15 hours. Psoriasis is often controlled at a methotrexate dose of about 15 mg/week.
Methotrexate’s advantages include ease of use. It’s a straightforward matter to start and stop the drug and to make small dose adjustments. Methotrexate comes in a variety of formulations: the 2.5-mg tablets, the 25 mg/mL solution for IM injection, and prefilled autoinjector devices for subcutaneous administration.
These preloaded pens for subcutaneous injection are just as effective as IM therapy, Dr. Leonardi said. Methotrexate is also better absorbed subcutaneously than it is orally. The bioavailability of oral methotrexate plateaus at a dose of about 22 mg/week, while subcutaneously delivered methotrexate does not. So, if a patient’s psoriasis isn’t adequately controlled on higher-dose oral therapy, it’s worth considering a switch to the preloaded pens.
“It’s a very simple injection, very cost-effective, and your patients may get more bang for the buck by doing that,” according to the dermatologist.
Also, nausea, vomiting, diarrhea, and abdominal pain have been shown to be significantly less frequent and intense with subcutaneous methotrexate than with the tablets. So, if a patient is experiencing limiting gastrointestinal issues on methotrexate tablets, a shift to subcutaneous therapy is often the solution.
What kind of efficacy can physicians expect with methotrexate monotherapy?
Dr. Leonardi cited the results of the European randomized, open-label RESTORE-1 trial (Br J Dermatol. 2011 Nov;165[5]:1109-17) as being consistent with his own extensive clinical experience: a week-16 PASI (Psoriasis Area and Severity Index ) 75 response rate of 42% with methotrexate, compared with 78% for infliximab (Remicade).
Of course, some patients can’t receive a biologic agent because of their age, lack of insurance coverage, or medical contraindications.
“I use methotrexate a lot in Medicare patients, where, with Part D, it’s hard to get access to biologics without really good coinsurance. I think all of us who prescribe biologics understand that,” he observed.
In pediatric patients with extensive psoriasis, he turns to methotrexate as first-line systemic therapy. After 3 months, if the young patient hasn’t responded satisfactorily, Dr. Leonardi asks the insurance company for access to a biologic agent and usually gets it.
Methotrexate really shines in combination with a biologic agent. It inhibits formation of antibiologic antibodies, an important cause of loss of effectiveness of monoclonal antibody therapy.
In one study, 28% of patients on adalimumab (Humira) developed antiadalimumab antibodies during the first 3 years of therapy. These patients were more likely to drop out of therapy for lack of effectiveness.
A key finding in this study was that two-thirds of patients who developed antiadalimumab antibodies did so during the first 28 weeks of therapy (JAMA. 2011 Apr 13;305[14]:1460-8). This time line has influenced Dr. Leonardi’s own clinical practice. He routinely keeps patients on methotrexate for their first 28-36 weeks on a biologic, then tapers the methotrexate in favor of biologic monotherapy.
Other benefits and guidelines
Methotrexate is also a boon in managing psoriasis flares in patients on a biologic.
“We’ve all had patients who are doing well on a biologic, they’re cruising along at 140 weeks, then they get strep throat or another infection, and, next thing you know, you have destabilized psoriasis,” Dr. Leonardi noted.
“One thing I’ll do is add methotrexate to the mix, try to get things under control, and, if we do, then we’ll try to taper the methotrexate,” he continued. “That whole episode might take 4-6 months to resolve before the patient might be able to tolerate biologic monotherapy, though. If they can’t at that point, you might consider rotating to another biologic.”
Current American Academy of Dermatology guidelines recommend that women on methotrexate limit their alcohol intake to one drink per day, two drinks for men. British Society for Rheumatology guidelines recommend a ceiling of 32 g to 64 g of ethanol per week.
“We don’t insist on abstinence. There’s no good evidence that it’s needed,” Dr. Leonardi noted. “If you dose this drug on Tuesday, you can be sure that it’s eliminated from the body on Friday, and that’s when I’ll generally green-light patients to socialize over the weekend. If you can make life a little more tolerable for your patients and they’re willing to follow your instructions, I think it’s a better deal all the way around.”
Prior to initiating methotrexate, he obtains a CBC with platelet count, liver function tests, serum urea nitrogen, creatinine, and screens for latent tuberculosis. In terms of on-treatment monitoring, he gets a CBC and liver function tests every 4-12 weeks and keeps an eye on renal function, especially in older patients, because methotrexate is eliminated renally.
“The guidelines have relaxed regarding the need for liver biopsies,” Dr. Leonardi said. “Most of us are not getting liver biopsies anymore, as is true of our friends in rheumatology.”
He reported having financial relationships with more than a dozen pharmaceutical companies. The SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – In an era when affordable health insurance could become increasingly tough to come by, it’s worth emphasizing that methotrexate is the most cost-effective way to manage extensive psoriasis – and the liquid formulation designed for intramuscular injection can be taken orally to reduce the cost even further, according to Craig L. Leonardi, MD.
“The absolute cheapest way to manage the patient who has no insurance and has bad psoriasis is to put him on methotrexate and teach him how to draw the liquid solution up in a syringe, dump it in a cup of juice, and drink it,” Dr. Leonardi said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Research Foundation. “The bioavailability is equivalent to [that of] the tablets, and it’s only about one-tenth the cost.”
He urged his fellow dermatologists not to forget about methotrexate in the current flashy era of highly effective – and very expensive – biologic therapies for psoriasis.
“I was thinking methotrexate was going to go away, but it turns out I rely more on that drug than ever before. It’s very useful, and it’s safe if used correctly,” said Dr. Leonardi, a dermatologist at Saint Louis University and a prominent clinical trialist.
He highlighted numerous clinical scenarios in which methotrexate remains a valuable treatment in patients with moderate to severe psoriasis. He also touched on patient monitoring requirements, adverse events, and common reasons he receives referrals from physicians whose patients seem to be having problems on methotrexate – referrals that, in most cases, could be avoided, he said, if the referring physician had a fuller understanding of the drug.
“Patients are referred to me all the time for methotrexate intolerance,” Dr. Leonardi said. “When you take methotrexate, you get a brief, dramatic spike in transaminase levels that peaks within a day and then drops off. But, when you get lab tests in these patients, you want to look at trough levels. You want to see the best liver function test values for the week.
“That means you have to tell the patient what day to take the drug and what day to get labs drawn,” he continued. “I’m here to tell you that the vast majority of issues that I see where patients have elevated liver function tests involves them getting their testing done in the first 4 days after taking methotrexate. Take the time to ask about this, and I think you’ll be pleasantly surprised.
“If you just make an adjustment and get them on the right schedule, you’ll discover that the patient is tolerating the drug just fine,” Dr. Leonardi added. “We like getting labs on Monday and dosing on Tuesday.”
Methotrexate’s half-life is 3-15 hours. Psoriasis is often controlled at a methotrexate dose of about 15 mg/week.
Methotrexate’s advantages include ease of use. It’s a straightforward matter to start and stop the drug and to make small dose adjustments. Methotrexate comes in a variety of formulations: the 2.5-mg tablets, the 25 mg/mL solution for IM injection, and prefilled autoinjector devices for subcutaneous administration.
These preloaded pens for subcutaneous injection are just as effective as IM therapy, Dr. Leonardi said. Methotrexate is also better absorbed subcutaneously than it is orally. The bioavailability of oral methotrexate plateaus at a dose of about 22 mg/week, while subcutaneously delivered methotrexate does not. So, if a patient’s psoriasis isn’t adequately controlled on higher-dose oral therapy, it’s worth considering a switch to the preloaded pens.
“It’s a very simple injection, very cost-effective, and your patients may get more bang for the buck by doing that,” according to the dermatologist.
Also, nausea, vomiting, diarrhea, and abdominal pain have been shown to be significantly less frequent and intense with subcutaneous methotrexate than with the tablets. So, if a patient is experiencing limiting gastrointestinal issues on methotrexate tablets, a shift to subcutaneous therapy is often the solution.
What kind of efficacy can physicians expect with methotrexate monotherapy?
Dr. Leonardi cited the results of the European randomized, open-label RESTORE-1 trial (Br J Dermatol. 2011 Nov;165[5]:1109-17) as being consistent with his own extensive clinical experience: a week-16 PASI (Psoriasis Area and Severity Index ) 75 response rate of 42% with methotrexate, compared with 78% for infliximab (Remicade).
Of course, some patients can’t receive a biologic agent because of their age, lack of insurance coverage, or medical contraindications.
“I use methotrexate a lot in Medicare patients, where, with Part D, it’s hard to get access to biologics without really good coinsurance. I think all of us who prescribe biologics understand that,” he observed.
In pediatric patients with extensive psoriasis, he turns to methotrexate as first-line systemic therapy. After 3 months, if the young patient hasn’t responded satisfactorily, Dr. Leonardi asks the insurance company for access to a biologic agent and usually gets it.
Methotrexate really shines in combination with a biologic agent. It inhibits formation of antibiologic antibodies, an important cause of loss of effectiveness of monoclonal antibody therapy.
In one study, 28% of patients on adalimumab (Humira) developed antiadalimumab antibodies during the first 3 years of therapy. These patients were more likely to drop out of therapy for lack of effectiveness.
A key finding in this study was that two-thirds of patients who developed antiadalimumab antibodies did so during the first 28 weeks of therapy (JAMA. 2011 Apr 13;305[14]:1460-8). This time line has influenced Dr. Leonardi’s own clinical practice. He routinely keeps patients on methotrexate for their first 28-36 weeks on a biologic, then tapers the methotrexate in favor of biologic monotherapy.
Other benefits and guidelines
Methotrexate is also a boon in managing psoriasis flares in patients on a biologic.
“We’ve all had patients who are doing well on a biologic, they’re cruising along at 140 weeks, then they get strep throat or another infection, and, next thing you know, you have destabilized psoriasis,” Dr. Leonardi noted.
“One thing I’ll do is add methotrexate to the mix, try to get things under control, and, if we do, then we’ll try to taper the methotrexate,” he continued. “That whole episode might take 4-6 months to resolve before the patient might be able to tolerate biologic monotherapy, though. If they can’t at that point, you might consider rotating to another biologic.”
Current American Academy of Dermatology guidelines recommend that women on methotrexate limit their alcohol intake to one drink per day, two drinks for men. British Society for Rheumatology guidelines recommend a ceiling of 32 g to 64 g of ethanol per week.
“We don’t insist on abstinence. There’s no good evidence that it’s needed,” Dr. Leonardi noted. “If you dose this drug on Tuesday, you can be sure that it’s eliminated from the body on Friday, and that’s when I’ll generally green-light patients to socialize over the weekend. If you can make life a little more tolerable for your patients and they’re willing to follow your instructions, I think it’s a better deal all the way around.”
Prior to initiating methotrexate, he obtains a CBC with platelet count, liver function tests, serum urea nitrogen, creatinine, and screens for latent tuberculosis. In terms of on-treatment monitoring, he gets a CBC and liver function tests every 4-12 weeks and keeps an eye on renal function, especially in older patients, because methotrexate is eliminated renally.
“The guidelines have relaxed regarding the need for liver biopsies,” Dr. Leonardi said. “Most of us are not getting liver biopsies anymore, as is true of our friends in rheumatology.”
He reported having financial relationships with more than a dozen pharmaceutical companies. The SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Secukinumab beat etanercept in 52-week psoriasis quality of life analysis
Compared with etanercept, secukinumab was associated with significantly faster and greater improvements in skin-specific quality of life among adults with moderate to severe psoriasis, according to a pooled analysis of data from the randomized, multicenter phase III ERASURE and FIXTURE trials.
The findings confirm the clinical superiority of secukinumab and extend prior head-to-head comparisons of this IL-17A inhibitor and etanercept, said Bruce E. Strober, MD, PhD, of the University of Connecticut in Farmington, Conn.
ERASURE and FIXTURE compared secukinumab (weekly for 4 weeks, then once-monthly) with etanercept (twice weekly for 12 weeks, then once-weekly) and placebo. Both secukinumab doses outperformed etanercept and placebo based on coprimary endpoints of PASI 75 and the proportion of patients who were clear or almost clear on a 5-point modified investigator’s global assessment (N Engl J Med. 2014 Jul 24;371[4]:326-38). A subsequent analysis showed that the clinical superiority of secukinumab held up at 52 weeks (J Dermatolog Treat. 2016;27[1]:1).
Because health-related quality of life is of increasing interest in psoriasis, Dr. Strober and his associates analyzed ERASURE and FIXTURE data on the Dermatology Life Quality Index (DLQI), a 10-item assessment of daily activities, leisure, personal relationships, symptoms and feelings, treatment, and work or school. Their analysis included 572 patients who received 300 mg secukinumab, 572 patients who received 150 mg secukinumab, and 326 patients who received 50 mg etanercept.
Baseline DLQI scores were similar among treatment groups. By week 4, significantly more secukinumab than etanercept recipients reported DLQI scores of 0 or 1 (DLQI 0/1), indicating that psoriasis was not impairing their quality of life (P < .05). This difference remained significant through week 52 at the 300-mg dose. The 150-mg dose significantly outperformed etanercept by week 8, and the difference remained significant through week 48.
In all, 81% of patients who received 300 mg secukinumab reported DLQI 0/1 at week 24, compared with 71% of those who received 150 mg secukinumab and 55% of those who received etanercept (P < .001 for each comparison with etanercept).
Among patients with complete data available, 86% of 300-mg secukinumab recipients achieved DLQI 0/1 at week 24, maintained this response through 52 weeks, and achieved a 90%-100% reduction in PASI total score at week 24. That was true of only 75% of etanercept recipients (P = .02, compared with 300 mg secukinumab) and 79% of 150-mg secukinumab recipients (P = .4, compared with etanercept).
In an adjusted model, patients who received 300 mg secukinumab were 1.9 times more likely to reach DLQI 0/1 than those who received etanercept (95% confidence interval, 1.6-2.3). Median time to response to both doses of secukinumab was 12 weeks, versus 24 weeks for etanercept (P < .0001).
This analysis offers “a robust, longer-term head-to-head comparison” of how secukinumab and etanercept affect quality of life in psoriasis, the researchers wrote. “Future research should consider replicating this evaluation using real-world long-term data and including other aspects of patient-centered outcomes, such as work productivity and psoriasis-related symptoms.”
Novartis funded ERASURE and FIXTURE. Dr. Strober reported financial ties to AbbVie, Amgen, Novartis, and several other pharmaceutical companies.
Compared with etanercept, secukinumab was associated with significantly faster and greater improvements in skin-specific quality of life among adults with moderate to severe psoriasis, according to a pooled analysis of data from the randomized, multicenter phase III ERASURE and FIXTURE trials.
The findings confirm the clinical superiority of secukinumab and extend prior head-to-head comparisons of this IL-17A inhibitor and etanercept, said Bruce E. Strober, MD, PhD, of the University of Connecticut in Farmington, Conn.
ERASURE and FIXTURE compared secukinumab (weekly for 4 weeks, then once-monthly) with etanercept (twice weekly for 12 weeks, then once-weekly) and placebo. Both secukinumab doses outperformed etanercept and placebo based on coprimary endpoints of PASI 75 and the proportion of patients who were clear or almost clear on a 5-point modified investigator’s global assessment (N Engl J Med. 2014 Jul 24;371[4]:326-38). A subsequent analysis showed that the clinical superiority of secukinumab held up at 52 weeks (J Dermatolog Treat. 2016;27[1]:1).
Because health-related quality of life is of increasing interest in psoriasis, Dr. Strober and his associates analyzed ERASURE and FIXTURE data on the Dermatology Life Quality Index (DLQI), a 10-item assessment of daily activities, leisure, personal relationships, symptoms and feelings, treatment, and work or school. Their analysis included 572 patients who received 300 mg secukinumab, 572 patients who received 150 mg secukinumab, and 326 patients who received 50 mg etanercept.
Baseline DLQI scores were similar among treatment groups. By week 4, significantly more secukinumab than etanercept recipients reported DLQI scores of 0 or 1 (DLQI 0/1), indicating that psoriasis was not impairing their quality of life (P < .05). This difference remained significant through week 52 at the 300-mg dose. The 150-mg dose significantly outperformed etanercept by week 8, and the difference remained significant through week 48.
In all, 81% of patients who received 300 mg secukinumab reported DLQI 0/1 at week 24, compared with 71% of those who received 150 mg secukinumab and 55% of those who received etanercept (P < .001 for each comparison with etanercept).
Among patients with complete data available, 86% of 300-mg secukinumab recipients achieved DLQI 0/1 at week 24, maintained this response through 52 weeks, and achieved a 90%-100% reduction in PASI total score at week 24. That was true of only 75% of etanercept recipients (P = .02, compared with 300 mg secukinumab) and 79% of 150-mg secukinumab recipients (P = .4, compared with etanercept).
In an adjusted model, patients who received 300 mg secukinumab were 1.9 times more likely to reach DLQI 0/1 than those who received etanercept (95% confidence interval, 1.6-2.3). Median time to response to both doses of secukinumab was 12 weeks, versus 24 weeks for etanercept (P < .0001).
This analysis offers “a robust, longer-term head-to-head comparison” of how secukinumab and etanercept affect quality of life in psoriasis, the researchers wrote. “Future research should consider replicating this evaluation using real-world long-term data and including other aspects of patient-centered outcomes, such as work productivity and psoriasis-related symptoms.”
Novartis funded ERASURE and FIXTURE. Dr. Strober reported financial ties to AbbVie, Amgen, Novartis, and several other pharmaceutical companies.
Compared with etanercept, secukinumab was associated with significantly faster and greater improvements in skin-specific quality of life among adults with moderate to severe psoriasis, according to a pooled analysis of data from the randomized, multicenter phase III ERASURE and FIXTURE trials.
The findings confirm the clinical superiority of secukinumab and extend prior head-to-head comparisons of this IL-17A inhibitor and etanercept, said Bruce E. Strober, MD, PhD, of the University of Connecticut in Farmington, Conn.
ERASURE and FIXTURE compared secukinumab (weekly for 4 weeks, then once-monthly) with etanercept (twice weekly for 12 weeks, then once-weekly) and placebo. Both secukinumab doses outperformed etanercept and placebo based on coprimary endpoints of PASI 75 and the proportion of patients who were clear or almost clear on a 5-point modified investigator’s global assessment (N Engl J Med. 2014 Jul 24;371[4]:326-38). A subsequent analysis showed that the clinical superiority of secukinumab held up at 52 weeks (J Dermatolog Treat. 2016;27[1]:1).
Because health-related quality of life is of increasing interest in psoriasis, Dr. Strober and his associates analyzed ERASURE and FIXTURE data on the Dermatology Life Quality Index (DLQI), a 10-item assessment of daily activities, leisure, personal relationships, symptoms and feelings, treatment, and work or school. Their analysis included 572 patients who received 300 mg secukinumab, 572 patients who received 150 mg secukinumab, and 326 patients who received 50 mg etanercept.
Baseline DLQI scores were similar among treatment groups. By week 4, significantly more secukinumab than etanercept recipients reported DLQI scores of 0 or 1 (DLQI 0/1), indicating that psoriasis was not impairing their quality of life (P < .05). This difference remained significant through week 52 at the 300-mg dose. The 150-mg dose significantly outperformed etanercept by week 8, and the difference remained significant through week 48.
In all, 81% of patients who received 300 mg secukinumab reported DLQI 0/1 at week 24, compared with 71% of those who received 150 mg secukinumab and 55% of those who received etanercept (P < .001 for each comparison with etanercept).
Among patients with complete data available, 86% of 300-mg secukinumab recipients achieved DLQI 0/1 at week 24, maintained this response through 52 weeks, and achieved a 90%-100% reduction in PASI total score at week 24. That was true of only 75% of etanercept recipients (P = .02, compared with 300 mg secukinumab) and 79% of 150-mg secukinumab recipients (P = .4, compared with etanercept).
In an adjusted model, patients who received 300 mg secukinumab were 1.9 times more likely to reach DLQI 0/1 than those who received etanercept (95% confidence interval, 1.6-2.3). Median time to response to both doses of secukinumab was 12 weeks, versus 24 weeks for etanercept (P < .0001).
This analysis offers “a robust, longer-term head-to-head comparison” of how secukinumab and etanercept affect quality of life in psoriasis, the researchers wrote. “Future research should consider replicating this evaluation using real-world long-term data and including other aspects of patient-centered outcomes, such as work productivity and psoriasis-related symptoms.”
Novartis funded ERASURE and FIXTURE. Dr. Strober reported financial ties to AbbVie, Amgen, Novartis, and several other pharmaceutical companies.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: Secukinumab was associated with faster and greater improvements in self-reported quality of life, compared with etanercept, in adults with moderate to severe plaque psoriasis.
Major finding: In all, 81% of patients who received 300 mg secukinumab reported DLQI 0/1 at week 24, compared with 71% of those who received 150 mg secukinumab and 55% of those who received etanercept (P < .001 for each comparison with etanercept).
Data source: A pooled analysis of the randomized, multicenter, phase III ERASURE and FIXTURE trials.
Disclosures: Novartis funded ERASURE and FIXTURE. Dr. Strober reported financial ties to AbbVie, Amgen, Novartis, and several other pharmaceutical companies.
Debunking Psoriasis Myths: Do Treatments for Psoriasis Cause Suicide?
Myth: Psoriasis Therapies Can Cause Suicidal Ideation in Psoriasis Patients
Psoriasis takes a toll on patients, both physically and emotionally. Depression is one of the comorbidities of psoriasis due to biological changes that cause psoriasis as well as the stigma of visible psoriasis. Severe depression and suicidal ideation have been perceived to be features of life-threatening medical disorders, but dermatologists need to be aware of the relationship between depressive symptoms, suicidal ideation, and psoriasis severity.
A 2010 United Kingdom study of 916,948 patients with mild psoriasis, severe psoriasis, or controls without psoriasis indicated that patients with psoriasis have an increased risk for depression, anxiety, and suicidality. The relative risk of these outcomes is elevated in younger patients with psoriasis, with the greatest relative risk being for depression in patients with severe psoriasis.
Kimball et al conducted a study in the United States of 7404 patients with psoriasis and 37,020 controls without psoriasis (age, <18 years). They reported that pediatric patients with psoriasis were significantly more at risk of developing psychiatric disorders versus controls (P=.0001), especially depression (P=.0036) and anxiety (P=.0048).
In February 2017, the US Food and Drug Administration (FDA) announced approval of brodalumab for use in adults with moderate to severe plaque psoriasis. It is intended for patients who are candidates for systemic therapy or phototherapy but have failed to respond or have stopped responding to other systemic therapies. Lebwohl et al published the results of the phase 3 clinical trials, which showed that brodalumab was highly effective in reducing plaque psoriasis, even compared to ustekinumab. In fact, psoriasis area and severity index scores of 100 were significantly higher in the brodalumab 210-mg group versus ustekinumab group by week 12 (P<.001).
However, the approval is accompanied with a strict warning from the FDA and tightly regulated access to the drug, as suicidal ideation and behavior, including 4 suicides, occurred in patients treated with brodalumab during clinical trials, particularly patients with a history of depression or suicidality. According to the FDA, "[a] causal association between treatment with [brodalumab] and increased risk of suicidal ideation and behavior has not been established." The label includes a black box warning and the drug will only be available through a restricted Risk Evaluation and Mitigation Strategy program, which has the following requirements from the FDA:
- Prescribers must be certified with the program and counsel patients about this risk. Patients with new or worsening symptoms of depression or suicidality should be referred to a mental health professional, as appropriate.
- Patients must sign a Patient-Prescriber Agreement Form and be made aware of the need to seek medical attention should they experience new or worsening suicidal thoughts or behavior, feelings of depression, anxiety, or other mood changes.
- Pharmacies must be certified with the program and must only dispense to patients who are authorized to receive the drug.
A medication guide is available for patients to inform them of the risk for suicidal ideation and behavior. The benefit of treatment must be weighed carefully against the seriousness of the risks associated with use.
Regardless of the therapy prescribed, dermatologists should be aware of the symptoms of depression. The National Psoriasis Foundation suggests you ask patients how they dress: Do they always wear long-sleeved shirts when they leave the house? Do they wear black? These questions can help determine if patients feel socially isolated or stigmatized by the disease. The National Psoriasis Foundation offers a Patient Navigation Center to help patients find a psychologist who specializes in issues related to psoriatic disease. Antidepressants and seeing a mental health professional can help, but ultimately taking control of the disease is the best way to improve depression.
Expert Commentary
According to the prescribing information for brodalumab, "Eight of the 10 subjects who attempted or completed suicide had a history of depression and/or suicidal ideation or behavior." Thus, 80% of these cases were at risk even before receiving 1 injection of brodalumab. Long-term registries will determine if there is truly an increased risk for suicidal ideation or behavior when taking brodalumab.
Brodalumab will be commercially available around the fall 2017. Before prescribing brodalumab, I will counsel patients about this potential increased risk of suicidal ideation or behavior as noted in the prescribing information, but I will tell them that a true risk has not yet been determined in long-term registries. I will mention to patients that if they really do feel depressed or experience suicidal ideation or behavior after starting brodalumab, they should stop taking brodalumab and contact me or a mental health professional.
—Jashin J. Wu, MD (Los Angeles, California)
FDA approves new psoriasis drug [news release]. Silver Spring, MD: US Food and Drug Administration; February 15, 2017. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm541981.htm. Accessed April 5, 2017.
Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
Kimball AB, Wu EQ, Guérin A, et al. Risks of developing psychiatric disorders in pediatric patients with psoriasis. J Am Acad Dermatol. 2012;67:651-7.e1-651-7.e2.
Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
Lebwohl M, Strober B, Menter A, et al. Phase 3 studies comparing brodalumab with ustekinumab in psoriasis. N Engl J Med. 2015;373:1318-1328.
Life with psoriasis: depression. National Psoriasis Foundation website. https://www.psoriasis.org/life-with-psoriasis/depression. Accessed April 5, 2017.
Özkaya Ö. Biologic psoriasis treatment, Siliq, approved by FDA with strong warning of possible suicide risk. https://psoriasisnewstoday.com/2017/02/16/psoriasis-drug-siliq-approved-by-fda-with-warning-of-possible-suicide-risk/. Published February 16, 2017. Accessed April 5, 2017.
Myth: Psoriasis Therapies Can Cause Suicidal Ideation in Psoriasis Patients
Psoriasis takes a toll on patients, both physically and emotionally. Depression is one of the comorbidities of psoriasis due to biological changes that cause psoriasis as well as the stigma of visible psoriasis. Severe depression and suicidal ideation have been perceived to be features of life-threatening medical disorders, but dermatologists need to be aware of the relationship between depressive symptoms, suicidal ideation, and psoriasis severity.
A 2010 United Kingdom study of 916,948 patients with mild psoriasis, severe psoriasis, or controls without psoriasis indicated that patients with psoriasis have an increased risk for depression, anxiety, and suicidality. The relative risk of these outcomes is elevated in younger patients with psoriasis, with the greatest relative risk being for depression in patients with severe psoriasis.
Kimball et al conducted a study in the United States of 7404 patients with psoriasis and 37,020 controls without psoriasis (age, <18 years). They reported that pediatric patients with psoriasis were significantly more at risk of developing psychiatric disorders versus controls (P=.0001), especially depression (P=.0036) and anxiety (P=.0048).
In February 2017, the US Food and Drug Administration (FDA) announced approval of brodalumab for use in adults with moderate to severe plaque psoriasis. It is intended for patients who are candidates for systemic therapy or phototherapy but have failed to respond or have stopped responding to other systemic therapies. Lebwohl et al published the results of the phase 3 clinical trials, which showed that brodalumab was highly effective in reducing plaque psoriasis, even compared to ustekinumab. In fact, psoriasis area and severity index scores of 100 were significantly higher in the brodalumab 210-mg group versus ustekinumab group by week 12 (P<.001).
However, the approval is accompanied with a strict warning from the FDA and tightly regulated access to the drug, as suicidal ideation and behavior, including 4 suicides, occurred in patients treated with brodalumab during clinical trials, particularly patients with a history of depression or suicidality. According to the FDA, "[a] causal association between treatment with [brodalumab] and increased risk of suicidal ideation and behavior has not been established." The label includes a black box warning and the drug will only be available through a restricted Risk Evaluation and Mitigation Strategy program, which has the following requirements from the FDA:
- Prescribers must be certified with the program and counsel patients about this risk. Patients with new or worsening symptoms of depression or suicidality should be referred to a mental health professional, as appropriate.
- Patients must sign a Patient-Prescriber Agreement Form and be made aware of the need to seek medical attention should they experience new or worsening suicidal thoughts or behavior, feelings of depression, anxiety, or other mood changes.
- Pharmacies must be certified with the program and must only dispense to patients who are authorized to receive the drug.
A medication guide is available for patients to inform them of the risk for suicidal ideation and behavior. The benefit of treatment must be weighed carefully against the seriousness of the risks associated with use.
Regardless of the therapy prescribed, dermatologists should be aware of the symptoms of depression. The National Psoriasis Foundation suggests you ask patients how they dress: Do they always wear long-sleeved shirts when they leave the house? Do they wear black? These questions can help determine if patients feel socially isolated or stigmatized by the disease. The National Psoriasis Foundation offers a Patient Navigation Center to help patients find a psychologist who specializes in issues related to psoriatic disease. Antidepressants and seeing a mental health professional can help, but ultimately taking control of the disease is the best way to improve depression.
Expert Commentary
According to the prescribing information for brodalumab, "Eight of the 10 subjects who attempted or completed suicide had a history of depression and/or suicidal ideation or behavior." Thus, 80% of these cases were at risk even before receiving 1 injection of brodalumab. Long-term registries will determine if there is truly an increased risk for suicidal ideation or behavior when taking brodalumab.
Brodalumab will be commercially available around the fall 2017. Before prescribing brodalumab, I will counsel patients about this potential increased risk of suicidal ideation or behavior as noted in the prescribing information, but I will tell them that a true risk has not yet been determined in long-term registries. I will mention to patients that if they really do feel depressed or experience suicidal ideation or behavior after starting brodalumab, they should stop taking brodalumab and contact me or a mental health professional.
—Jashin J. Wu, MD (Los Angeles, California)
Myth: Psoriasis Therapies Can Cause Suicidal Ideation in Psoriasis Patients
Psoriasis takes a toll on patients, both physically and emotionally. Depression is one of the comorbidities of psoriasis due to biological changes that cause psoriasis as well as the stigma of visible psoriasis. Severe depression and suicidal ideation have been perceived to be features of life-threatening medical disorders, but dermatologists need to be aware of the relationship between depressive symptoms, suicidal ideation, and psoriasis severity.
A 2010 United Kingdom study of 916,948 patients with mild psoriasis, severe psoriasis, or controls without psoriasis indicated that patients with psoriasis have an increased risk for depression, anxiety, and suicidality. The relative risk of these outcomes is elevated in younger patients with psoriasis, with the greatest relative risk being for depression in patients with severe psoriasis.
Kimball et al conducted a study in the United States of 7404 patients with psoriasis and 37,020 controls without psoriasis (age, <18 years). They reported that pediatric patients with psoriasis were significantly more at risk of developing psychiatric disorders versus controls (P=.0001), especially depression (P=.0036) and anxiety (P=.0048).
In February 2017, the US Food and Drug Administration (FDA) announced approval of brodalumab for use in adults with moderate to severe plaque psoriasis. It is intended for patients who are candidates for systemic therapy or phototherapy but have failed to respond or have stopped responding to other systemic therapies. Lebwohl et al published the results of the phase 3 clinical trials, which showed that brodalumab was highly effective in reducing plaque psoriasis, even compared to ustekinumab. In fact, psoriasis area and severity index scores of 100 were significantly higher in the brodalumab 210-mg group versus ustekinumab group by week 12 (P<.001).
However, the approval is accompanied with a strict warning from the FDA and tightly regulated access to the drug, as suicidal ideation and behavior, including 4 suicides, occurred in patients treated with brodalumab during clinical trials, particularly patients with a history of depression or suicidality. According to the FDA, "[a] causal association between treatment with [brodalumab] and increased risk of suicidal ideation and behavior has not been established." The label includes a black box warning and the drug will only be available through a restricted Risk Evaluation and Mitigation Strategy program, which has the following requirements from the FDA:
- Prescribers must be certified with the program and counsel patients about this risk. Patients with new or worsening symptoms of depression or suicidality should be referred to a mental health professional, as appropriate.
- Patients must sign a Patient-Prescriber Agreement Form and be made aware of the need to seek medical attention should they experience new or worsening suicidal thoughts or behavior, feelings of depression, anxiety, or other mood changes.
- Pharmacies must be certified with the program and must only dispense to patients who are authorized to receive the drug.
A medication guide is available for patients to inform them of the risk for suicidal ideation and behavior. The benefit of treatment must be weighed carefully against the seriousness of the risks associated with use.
Regardless of the therapy prescribed, dermatologists should be aware of the symptoms of depression. The National Psoriasis Foundation suggests you ask patients how they dress: Do they always wear long-sleeved shirts when they leave the house? Do they wear black? These questions can help determine if patients feel socially isolated or stigmatized by the disease. The National Psoriasis Foundation offers a Patient Navigation Center to help patients find a psychologist who specializes in issues related to psoriatic disease. Antidepressants and seeing a mental health professional can help, but ultimately taking control of the disease is the best way to improve depression.
Expert Commentary
According to the prescribing information for brodalumab, "Eight of the 10 subjects who attempted or completed suicide had a history of depression and/or suicidal ideation or behavior." Thus, 80% of these cases were at risk even before receiving 1 injection of brodalumab. Long-term registries will determine if there is truly an increased risk for suicidal ideation or behavior when taking brodalumab.
Brodalumab will be commercially available around the fall 2017. Before prescribing brodalumab, I will counsel patients about this potential increased risk of suicidal ideation or behavior as noted in the prescribing information, but I will tell them that a true risk has not yet been determined in long-term registries. I will mention to patients that if they really do feel depressed or experience suicidal ideation or behavior after starting brodalumab, they should stop taking brodalumab and contact me or a mental health professional.
—Jashin J. Wu, MD (Los Angeles, California)
FDA approves new psoriasis drug [news release]. Silver Spring, MD: US Food and Drug Administration; February 15, 2017. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm541981.htm. Accessed April 5, 2017.
Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
Kimball AB, Wu EQ, Guérin A, et al. Risks of developing psychiatric disorders in pediatric patients with psoriasis. J Am Acad Dermatol. 2012;67:651-7.e1-651-7.e2.
Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
Lebwohl M, Strober B, Menter A, et al. Phase 3 studies comparing brodalumab with ustekinumab in psoriasis. N Engl J Med. 2015;373:1318-1328.
Life with psoriasis: depression. National Psoriasis Foundation website. https://www.psoriasis.org/life-with-psoriasis/depression. Accessed April 5, 2017.
Özkaya Ö. Biologic psoriasis treatment, Siliq, approved by FDA with strong warning of possible suicide risk. https://psoriasisnewstoday.com/2017/02/16/psoriasis-drug-siliq-approved-by-fda-with-warning-of-possible-suicide-risk/. Published February 16, 2017. Accessed April 5, 2017.
FDA approves new psoriasis drug [news release]. Silver Spring, MD: US Food and Drug Administration; February 15, 2017. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm541981.htm. Accessed April 5, 2017.
Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
Kimball AB, Wu EQ, Guérin A, et al. Risks of developing psychiatric disorders in pediatric patients with psoriasis. J Am Acad Dermatol. 2012;67:651-7.e1-651-7.e2.
Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
Lebwohl M, Strober B, Menter A, et al. Phase 3 studies comparing brodalumab with ustekinumab in psoriasis. N Engl J Med. 2015;373:1318-1328.
Life with psoriasis: depression. National Psoriasis Foundation website. https://www.psoriasis.org/life-with-psoriasis/depression. Accessed April 5, 2017.
Özkaya Ö. Biologic psoriasis treatment, Siliq, approved by FDA with strong warning of possible suicide risk. https://psoriasisnewstoday.com/2017/02/16/psoriasis-drug-siliq-approved-by-fda-with-warning-of-possible-suicide-risk/. Published February 16, 2017. Accessed April 5, 2017.
What’s old is new in topical psoriasis therapy
WAILEA, HAWAII – A fixed combination of halobetasol and tazarotene formulated as a once-daily lotion proved safe and effective for patients with moderate or severe plaque psoriasis in a phase II randomized clinical trial, Linda Stein Gold, MD, reported at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
The rationale for developing this product is that it should provide the efficacy of a class 1 superpotent topical corticosteroid, albeit with the topical retinoid lessening concern about the possibility of steroid-induced skin atrophy and other long-term safety issues, explained Dr. Stein Gold, director of dermatology research at Henry Ford Health System in Detroit.
The double-blind, multicenter, 8-week, phase II study included 212 patients randomized 2:2:1 to once-daily application of fixed-combination halobetasol propionate 0.01% and tazarotene 0.045% lotion, either topical agent as monotherapy, or vehicle. Ninety percent of participants had moderate plaque psoriasis as defined by a baseline Investigator’s Global Assessment (IGA) score of 3 and a median 5% body surface area involvement. The remaining patients were classified as having severe psoriasis, with a baseline IGA of 4, but no one with psoriasis over more than 12% of their body surface area was eligible for the trial.
The fixed-combination lotion, known for now as IDP-118, established superior efficacy over placebo by 2 weeks. At week 8, treatment success, as defined by at least a two-grade improvement from baseline in IGA score plus a rating of clear or almost clear, was documented in 53% of the IDP-118 lotion group, compared with 33% on halobetasol propionate monotherapy, 19% with tazarotene alone, and 10% with vehicle, Dr. Stein Gold reported.
Each patient had a target lesion 25-32 cm2 in size, which was designated for careful assessment of changes in the domains of lesional erythema, plaque elevation, and scaling. At week 8, the IDP-118 had a target lesion treatment success rate of 54% for erythema, 68% for plaque elevation, and 64% for scaling, she said.
The treatment discontinuation rate was 3.4% in the IDP-118 group, 12.1% with tazarotene, zero for halobetasol propionate, and 3.2% for vehicle. The most frequently reported treatment-emergent adverse events were mild or moderate application-site reactions. Skin atrophy was rare. No treatment-related serious adverse events occurred.
A 555-patient, phase III, open-label, 12-month safety study of IDP-118 is due to be completed later in 2017.
The phase II study was funded by Valeant Pharmaceuticals. Dr. Stein Gold reported serving as an investigator, advisor, and speaker for the company. SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – A fixed combination of halobetasol and tazarotene formulated as a once-daily lotion proved safe and effective for patients with moderate or severe plaque psoriasis in a phase II randomized clinical trial, Linda Stein Gold, MD, reported at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
The rationale for developing this product is that it should provide the efficacy of a class 1 superpotent topical corticosteroid, albeit with the topical retinoid lessening concern about the possibility of steroid-induced skin atrophy and other long-term safety issues, explained Dr. Stein Gold, director of dermatology research at Henry Ford Health System in Detroit.
The double-blind, multicenter, 8-week, phase II study included 212 patients randomized 2:2:1 to once-daily application of fixed-combination halobetasol propionate 0.01% and tazarotene 0.045% lotion, either topical agent as monotherapy, or vehicle. Ninety percent of participants had moderate plaque psoriasis as defined by a baseline Investigator’s Global Assessment (IGA) score of 3 and a median 5% body surface area involvement. The remaining patients were classified as having severe psoriasis, with a baseline IGA of 4, but no one with psoriasis over more than 12% of their body surface area was eligible for the trial.
The fixed-combination lotion, known for now as IDP-118, established superior efficacy over placebo by 2 weeks. At week 8, treatment success, as defined by at least a two-grade improvement from baseline in IGA score plus a rating of clear or almost clear, was documented in 53% of the IDP-118 lotion group, compared with 33% on halobetasol propionate monotherapy, 19% with tazarotene alone, and 10% with vehicle, Dr. Stein Gold reported.
Each patient had a target lesion 25-32 cm2 in size, which was designated for careful assessment of changes in the domains of lesional erythema, plaque elevation, and scaling. At week 8, the IDP-118 had a target lesion treatment success rate of 54% for erythema, 68% for plaque elevation, and 64% for scaling, she said.
The treatment discontinuation rate was 3.4% in the IDP-118 group, 12.1% with tazarotene, zero for halobetasol propionate, and 3.2% for vehicle. The most frequently reported treatment-emergent adverse events were mild or moderate application-site reactions. Skin atrophy was rare. No treatment-related serious adverse events occurred.
A 555-patient, phase III, open-label, 12-month safety study of IDP-118 is due to be completed later in 2017.
The phase II study was funded by Valeant Pharmaceuticals. Dr. Stein Gold reported serving as an investigator, advisor, and speaker for the company. SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – A fixed combination of halobetasol and tazarotene formulated as a once-daily lotion proved safe and effective for patients with moderate or severe plaque psoriasis in a phase II randomized clinical trial, Linda Stein Gold, MD, reported at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
The rationale for developing this product is that it should provide the efficacy of a class 1 superpotent topical corticosteroid, albeit with the topical retinoid lessening concern about the possibility of steroid-induced skin atrophy and other long-term safety issues, explained Dr. Stein Gold, director of dermatology research at Henry Ford Health System in Detroit.
The double-blind, multicenter, 8-week, phase II study included 212 patients randomized 2:2:1 to once-daily application of fixed-combination halobetasol propionate 0.01% and tazarotene 0.045% lotion, either topical agent as monotherapy, or vehicle. Ninety percent of participants had moderate plaque psoriasis as defined by a baseline Investigator’s Global Assessment (IGA) score of 3 and a median 5% body surface area involvement. The remaining patients were classified as having severe psoriasis, with a baseline IGA of 4, but no one with psoriasis over more than 12% of their body surface area was eligible for the trial.
The fixed-combination lotion, known for now as IDP-118, established superior efficacy over placebo by 2 weeks. At week 8, treatment success, as defined by at least a two-grade improvement from baseline in IGA score plus a rating of clear or almost clear, was documented in 53% of the IDP-118 lotion group, compared with 33% on halobetasol propionate monotherapy, 19% with tazarotene alone, and 10% with vehicle, Dr. Stein Gold reported.
Each patient had a target lesion 25-32 cm2 in size, which was designated for careful assessment of changes in the domains of lesional erythema, plaque elevation, and scaling. At week 8, the IDP-118 had a target lesion treatment success rate of 54% for erythema, 68% for plaque elevation, and 64% for scaling, she said.
The treatment discontinuation rate was 3.4% in the IDP-118 group, 12.1% with tazarotene, zero for halobetasol propionate, and 3.2% for vehicle. The most frequently reported treatment-emergent adverse events were mild or moderate application-site reactions. Skin atrophy was rare. No treatment-related serious adverse events occurred.
A 555-patient, phase III, open-label, 12-month safety study of IDP-118 is due to be completed later in 2017.
The phase II study was funded by Valeant Pharmaceuticals. Dr. Stein Gold reported serving as an investigator, advisor, and speaker for the company. SDEF and this news organization are owned by the same parent company.
Key clinical point:
Major finding: An investigational fixed-combination lotion consisting of halobetasol propionate 0.01% and tazarotene 0.045% achieved scores of clear or almost clear in 53% of patients, significantly better than either component as monotherapy.
Data source: This phase II, double-blind, multicenter, vehicle-controlled, 8-week randomized clinical trial included 212 patients with moderate or severe plaque psoriasis.
Disclosures: The study was funded by Valeant Pharmaceuticals. The presenter reported serving as an investigator, advisor, and speaker for the company.
Psoriasis Symptoms With the Greatest Impact on Patients
Flaking/scaling and itching, followed by dry cracked skin that may bleed, pain or soreness, and burning/stinging were noted by psoriasis patients as the symptoms with the most significant impact on daily life in a public meeting hosted by the US Food and Drug Administration (FDA) to hear patient perspectives on psoriasis. Approximately 70 psoriasis patients or patient representatives attended the meeting in person and others attended through a live webcast.
More than two-thirds of respondents identified flaking/scaling as one of their most significant symptoms of psoriasis, either localized to psoriasis-prone areas such as the elbows and knees or more widespread. Patients reported that this symptom is constant, leaving them to absentmindedly rub certain areas of the skin.
A similar number of respondents indicated that itching was their most significant symptom. One patient called it “an intense subcutaneous itch… deep down in the skin,” a description that resonated with other patients in the room.
Nearly 40% identified dry cracked skin that may bleed as a significant symptom, noting that areas where skin is thinner are affected more, such as the folds of the body. Patients described this symptom as interrelated with other symptoms such as itching. “The thicker the scales get on my skin, the more they itch, and the more they itch, the more I am likely to scratch them, and the more I scratch them, the more they start to crack, and then more come back and it keeps going and going,” one patient said.
More than one-quarter of respondents indicated that pain, soreness, or burning/stinging were the most significant symptoms. Patients indicated that the stinging/burning was more episodic, while the pain was more constant, with the pain being under the skin.
Triggers of these symptoms included stress (primary trigger), changes in weather, hormonal changes, diet, lotions, prolonged exposure to sunlight, sweat, aging, and other medical conditions.
Dermatologists may use these patient insights to prescribe therapies that target these symptoms.
The psoriasis public meeting in March 2016 was the FDA’s 18th patient-focused drug development meeting. The FDA sought this information to have a greater understanding of the burden of psoriasis on patients and the treatments currently used to treat psoriasis and its symptoms. This information will help guide the FDA as they consider future drug approvals.
Flaking/scaling and itching, followed by dry cracked skin that may bleed, pain or soreness, and burning/stinging were noted by psoriasis patients as the symptoms with the most significant impact on daily life in a public meeting hosted by the US Food and Drug Administration (FDA) to hear patient perspectives on psoriasis. Approximately 70 psoriasis patients or patient representatives attended the meeting in person and others attended through a live webcast.
More than two-thirds of respondents identified flaking/scaling as one of their most significant symptoms of psoriasis, either localized to psoriasis-prone areas such as the elbows and knees or more widespread. Patients reported that this symptom is constant, leaving them to absentmindedly rub certain areas of the skin.
A similar number of respondents indicated that itching was their most significant symptom. One patient called it “an intense subcutaneous itch… deep down in the skin,” a description that resonated with other patients in the room.
Nearly 40% identified dry cracked skin that may bleed as a significant symptom, noting that areas where skin is thinner are affected more, such as the folds of the body. Patients described this symptom as interrelated with other symptoms such as itching. “The thicker the scales get on my skin, the more they itch, and the more they itch, the more I am likely to scratch them, and the more I scratch them, the more they start to crack, and then more come back and it keeps going and going,” one patient said.
More than one-quarter of respondents indicated that pain, soreness, or burning/stinging were the most significant symptoms. Patients indicated that the stinging/burning was more episodic, while the pain was more constant, with the pain being under the skin.
Triggers of these symptoms included stress (primary trigger), changes in weather, hormonal changes, diet, lotions, prolonged exposure to sunlight, sweat, aging, and other medical conditions.
Dermatologists may use these patient insights to prescribe therapies that target these symptoms.
The psoriasis public meeting in March 2016 was the FDA’s 18th patient-focused drug development meeting. The FDA sought this information to have a greater understanding of the burden of psoriasis on patients and the treatments currently used to treat psoriasis and its symptoms. This information will help guide the FDA as they consider future drug approvals.
Flaking/scaling and itching, followed by dry cracked skin that may bleed, pain or soreness, and burning/stinging were noted by psoriasis patients as the symptoms with the most significant impact on daily life in a public meeting hosted by the US Food and Drug Administration (FDA) to hear patient perspectives on psoriasis. Approximately 70 psoriasis patients or patient representatives attended the meeting in person and others attended through a live webcast.
More than two-thirds of respondents identified flaking/scaling as one of their most significant symptoms of psoriasis, either localized to psoriasis-prone areas such as the elbows and knees or more widespread. Patients reported that this symptom is constant, leaving them to absentmindedly rub certain areas of the skin.
A similar number of respondents indicated that itching was their most significant symptom. One patient called it “an intense subcutaneous itch… deep down in the skin,” a description that resonated with other patients in the room.
Nearly 40% identified dry cracked skin that may bleed as a significant symptom, noting that areas where skin is thinner are affected more, such as the folds of the body. Patients described this symptom as interrelated with other symptoms such as itching. “The thicker the scales get on my skin, the more they itch, and the more they itch, the more I am likely to scratch them, and the more I scratch them, the more they start to crack, and then more come back and it keeps going and going,” one patient said.
More than one-quarter of respondents indicated that pain, soreness, or burning/stinging were the most significant symptoms. Patients indicated that the stinging/burning was more episodic, while the pain was more constant, with the pain being under the skin.
Triggers of these symptoms included stress (primary trigger), changes in weather, hormonal changes, diet, lotions, prolonged exposure to sunlight, sweat, aging, and other medical conditions.
Dermatologists may use these patient insights to prescribe therapies that target these symptoms.
The psoriasis public meeting in March 2016 was the FDA’s 18th patient-focused drug development meeting. The FDA sought this information to have a greater understanding of the burden of psoriasis on patients and the treatments currently used to treat psoriasis and its symptoms. This information will help guide the FDA as they consider future drug approvals.
Psoriasis on the Hands and Feet: How Patients Should Care for These Areas
What does your patient need to know at the first visit?
Patients with this condition need to avoid friction and excessive moisture. They should be counseled to use gloves for excessive wet work. I recommend they use cotton gloves on the hands, and then cover those with rubber gloves. Patients should use a hand emollient regularly, including after each time they wash their hands or have exposure to water. If the patient lifts weights, I recommend he/she use weight-lifting gloves to reduce friction.
What are your go to treatments? What are the side effects?
The first line of therapy for hand and foot psoriasis is a topical agent. I most often use a combination of topical steroids and a topical vitamin D analogue. If insurance is amenable, I may use a fixed combination of topical steroid and vitamin D analogue.
If topical therapies are not successful, I often consider using excimer laser therapy, which requires the patient to come to the office twice weekly, so it is important to determine if this therapy is compatible with the patient's schedule. Other options include oral and biological therapies. Apremilast is a reasonable first-line systemic therapy given that it is an oral therapy, requires no laboratory monitoring, and has a favorable safety profile. Alternatively, biologic agents can be utilized. There are several analyses available looking at the efficacy of different biologics in hand and foot psoriasis, but at this point there is no consensus first choice for a biologic in this condition. Many available biologics may have a notable impact though.
The side effects of therapies for psoriasis are well established. Topical therapies and excimer laser are relatively safe choices. Apremilast has been associated with early gastrointestinal tract side effects that tend to resolve over time. Each biologic has a unique safety profile, with a rare incidence of side effects that should be reviewed carefully with any prospective patients before starting therapy.
How do you keep patients compliant with treatment?
It is important to reinforce gentle hand care and foot care. Patients need to understand that lack of compliance with treatment will lead to recurrence of disease.
What do you do if patients refuse treatment?
I try to educate them as best as possible, and ask them to return and reconsider therapy if they find that this condition affects their quality of life.
What does your patient need to know at the first visit?
Patients with this condition need to avoid friction and excessive moisture. They should be counseled to use gloves for excessive wet work. I recommend they use cotton gloves on the hands, and then cover those with rubber gloves. Patients should use a hand emollient regularly, including after each time they wash their hands or have exposure to water. If the patient lifts weights, I recommend he/she use weight-lifting gloves to reduce friction.
What are your go to treatments? What are the side effects?
The first line of therapy for hand and foot psoriasis is a topical agent. I most often use a combination of topical steroids and a topical vitamin D analogue. If insurance is amenable, I may use a fixed combination of topical steroid and vitamin D analogue.
If topical therapies are not successful, I often consider using excimer laser therapy, which requires the patient to come to the office twice weekly, so it is important to determine if this therapy is compatible with the patient's schedule. Other options include oral and biological therapies. Apremilast is a reasonable first-line systemic therapy given that it is an oral therapy, requires no laboratory monitoring, and has a favorable safety profile. Alternatively, biologic agents can be utilized. There are several analyses available looking at the efficacy of different biologics in hand and foot psoriasis, but at this point there is no consensus first choice for a biologic in this condition. Many available biologics may have a notable impact though.
The side effects of therapies for psoriasis are well established. Topical therapies and excimer laser are relatively safe choices. Apremilast has been associated with early gastrointestinal tract side effects that tend to resolve over time. Each biologic has a unique safety profile, with a rare incidence of side effects that should be reviewed carefully with any prospective patients before starting therapy.
How do you keep patients compliant with treatment?
It is important to reinforce gentle hand care and foot care. Patients need to understand that lack of compliance with treatment will lead to recurrence of disease.
What do you do if patients refuse treatment?
I try to educate them as best as possible, and ask them to return and reconsider therapy if they find that this condition affects their quality of life.
What does your patient need to know at the first visit?
Patients with this condition need to avoid friction and excessive moisture. They should be counseled to use gloves for excessive wet work. I recommend they use cotton gloves on the hands, and then cover those with rubber gloves. Patients should use a hand emollient regularly, including after each time they wash their hands or have exposure to water. If the patient lifts weights, I recommend he/she use weight-lifting gloves to reduce friction.
What are your go to treatments? What are the side effects?
The first line of therapy for hand and foot psoriasis is a topical agent. I most often use a combination of topical steroids and a topical vitamin D analogue. If insurance is amenable, I may use a fixed combination of topical steroid and vitamin D analogue.
If topical therapies are not successful, I often consider using excimer laser therapy, which requires the patient to come to the office twice weekly, so it is important to determine if this therapy is compatible with the patient's schedule. Other options include oral and biological therapies. Apremilast is a reasonable first-line systemic therapy given that it is an oral therapy, requires no laboratory monitoring, and has a favorable safety profile. Alternatively, biologic agents can be utilized. There are several analyses available looking at the efficacy of different biologics in hand and foot psoriasis, but at this point there is no consensus first choice for a biologic in this condition. Many available biologics may have a notable impact though.
The side effects of therapies for psoriasis are well established. Topical therapies and excimer laser are relatively safe choices. Apremilast has been associated with early gastrointestinal tract side effects that tend to resolve over time. Each biologic has a unique safety profile, with a rare incidence of side effects that should be reviewed carefully with any prospective patients before starting therapy.
How do you keep patients compliant with treatment?
It is important to reinforce gentle hand care and foot care. Patients need to understand that lack of compliance with treatment will lead to recurrence of disease.
What do you do if patients refuse treatment?
I try to educate them as best as possible, and ask them to return and reconsider therapy if they find that this condition affects their quality of life.
Ixekizumab found superior to ustekinumab in psoriasis at 24 weeks
ORLANDO – The interleukin-17A inhibitor ixekizumab was associated with superior efficacy and safety when compared with ustekinumab at 24 weeks, a head-to-head trial of the two monoclonal antibodies in plaque psoriasis has shown.
The 24-week data from the IXORA-S trial were presented during a late-breaking clinical trial session at the annual meeting of the American Academy of Dermatology by Kristian Reich, MD, PhD, a founder of SCIderm in Hamburg, Germany.
At 24 weeks, 49% in the ixekizumab arm achieved a Psoriasis Area and Severity Index (PASI) 100 level of skin clearance, compared with 24% in the ustekinumab arm (P = .001). Ixekizumab treatment also reached significantly higher skin clearances than ustekinumab at 24 weeks at the level of PASI 90 (83% vs. 59%; P less than .001) and PASI 75 (91% vs. 82%; P = .015).
Treatment with ixekizumab produced a Static Physician’s Global Assessment (sPGA) score of 0 in 54% at 24 weeks, compared with 24% with ustekinumab (P less than .001). A sPGA score of 0 or 1 occurred in 87% of patients who took ixekizumab and in 59% of the ustekinumab group.
An improvement of 4 or more points in the pruritus Numeric Rating Scale was reported by 86% of ixekizumab patients at 24 weeks, compared with 72% of those who took ustekinumab (P = .018).
Dr. Reich reported that there were no deaths or any significant differences in overall treatment-related adverse events across both arms, although he cautioned against putting too much stock in 24-week safety data. “I hesitate to show safety data for only 300 patients at 24 weeks, but it’s good to see here that there doesn’t seem to be a difference. I still would want to see larger patient numbers and more long-term data,” he said.
Dr. Reich had numerous disclosures, including honoraria for serving as a speaker for Eli Lilly, the sponsor of this trial.
[email protected]
On Twitter @whitneymcknight
ORLANDO – The interleukin-17A inhibitor ixekizumab was associated with superior efficacy and safety when compared with ustekinumab at 24 weeks, a head-to-head trial of the two monoclonal antibodies in plaque psoriasis has shown.
The 24-week data from the IXORA-S trial were presented during a late-breaking clinical trial session at the annual meeting of the American Academy of Dermatology by Kristian Reich, MD, PhD, a founder of SCIderm in Hamburg, Germany.
At 24 weeks, 49% in the ixekizumab arm achieved a Psoriasis Area and Severity Index (PASI) 100 level of skin clearance, compared with 24% in the ustekinumab arm (P = .001). Ixekizumab treatment also reached significantly higher skin clearances than ustekinumab at 24 weeks at the level of PASI 90 (83% vs. 59%; P less than .001) and PASI 75 (91% vs. 82%; P = .015).
Treatment with ixekizumab produced a Static Physician’s Global Assessment (sPGA) score of 0 in 54% at 24 weeks, compared with 24% with ustekinumab (P less than .001). A sPGA score of 0 or 1 occurred in 87% of patients who took ixekizumab and in 59% of the ustekinumab group.
An improvement of 4 or more points in the pruritus Numeric Rating Scale was reported by 86% of ixekizumab patients at 24 weeks, compared with 72% of those who took ustekinumab (P = .018).
Dr. Reich reported that there were no deaths or any significant differences in overall treatment-related adverse events across both arms, although he cautioned against putting too much stock in 24-week safety data. “I hesitate to show safety data for only 300 patients at 24 weeks, but it’s good to see here that there doesn’t seem to be a difference. I still would want to see larger patient numbers and more long-term data,” he said.
Dr. Reich had numerous disclosures, including honoraria for serving as a speaker for Eli Lilly, the sponsor of this trial.
[email protected]
On Twitter @whitneymcknight
ORLANDO – The interleukin-17A inhibitor ixekizumab was associated with superior efficacy and safety when compared with ustekinumab at 24 weeks, a head-to-head trial of the two monoclonal antibodies in plaque psoriasis has shown.
The 24-week data from the IXORA-S trial were presented during a late-breaking clinical trial session at the annual meeting of the American Academy of Dermatology by Kristian Reich, MD, PhD, a founder of SCIderm in Hamburg, Germany.
At 24 weeks, 49% in the ixekizumab arm achieved a Psoriasis Area and Severity Index (PASI) 100 level of skin clearance, compared with 24% in the ustekinumab arm (P = .001). Ixekizumab treatment also reached significantly higher skin clearances than ustekinumab at 24 weeks at the level of PASI 90 (83% vs. 59%; P less than .001) and PASI 75 (91% vs. 82%; P = .015).
Treatment with ixekizumab produced a Static Physician’s Global Assessment (sPGA) score of 0 in 54% at 24 weeks, compared with 24% with ustekinumab (P less than .001). A sPGA score of 0 or 1 occurred in 87% of patients who took ixekizumab and in 59% of the ustekinumab group.
An improvement of 4 or more points in the pruritus Numeric Rating Scale was reported by 86% of ixekizumab patients at 24 weeks, compared with 72% of those who took ustekinumab (P = .018).
Dr. Reich reported that there were no deaths or any significant differences in overall treatment-related adverse events across both arms, although he cautioned against putting too much stock in 24-week safety data. “I hesitate to show safety data for only 300 patients at 24 weeks, but it’s good to see here that there doesn’t seem to be a difference. I still would want to see larger patient numbers and more long-term data,” he said.
Dr. Reich had numerous disclosures, including honoraria for serving as a speaker for Eli Lilly, the sponsor of this trial.
[email protected]
On Twitter @whitneymcknight
AT AAD 2017
Key clinical point:
Major finding: At 24 weeks, ixekizumab was associated with significantly better PASI response rates and reduction in itch than ustekinumab in patients with moderate to severe plaque psoriasis.
Data source: Head-to-head trial of 302 adults with moderate to severe plaque psoriasis treated with either ixekizumab or ustekinumab.
Disclosures: Dr. Reich had numerous disclosures, including honoraria for serving as a speaker for Eli Lilly, the sponsor of this trial.