User login
Clinical Endocrinology News is an independent news source that provides endocrinologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the endocrinologist's practice. Specialty topics include Diabetes, Lipid & Metabolic Disorders Menopause, Obesity, Osteoporosis, Pediatric Endocrinology, Pituitary, Thyroid & Adrenal Disorders, and Reproductive Endocrinology. Featured content includes Commentaries, Implementin Health Reform, Law & Medicine, and In the Loop, the blog of Clinical Endocrinology News. Clinical Endocrinology News is owned by Frontline Medical Communications.
addict
addicted
addicting
addiction
adult sites
alcohol
antibody
ass
attorney
audit
auditor
babies
babpa
baby
ban
banned
banning
best
bisexual
bitch
bleach
blog
blow job
bondage
boobs
booty
buy
cannabis
certificate
certification
certified
cheap
cheapest
class action
cocaine
cock
counterfeit drug
crack
crap
crime
criminal
cunt
curable
cure
dangerous
dangers
dead
deadly
death
defend
defended
depedent
dependence
dependent
detergent
dick
die
dildo
drug abuse
drug recall
dying
fag
fake
fatal
fatalities
fatality
free
fuck
gangs
gingivitis
guns
hardcore
herbal
herbs
heroin
herpes
home remedies
homo
horny
hypersensitivity
hypoglycemia treatment
illegal drug use
illegal use of prescription
incest
infant
infants
job
ketoacidosis
kill
killer
killing
kinky
law suit
lawsuit
lawyer
lesbian
marijuana
medicine for hypoglycemia
murder
naked
natural
newborn
nigger
noise
nude
nudity
orgy
over the counter
overdosage
overdose
overdosed
overdosing
penis
pimp
pistol
porn
porno
pornographic
pornography
prison
profanity
purchase
purchasing
pussy
queer
rape
rapist
recall
recreational drug
rob
robberies
sale
sales
sex
sexual
shit
shoot
slut
slutty
stole
stolen
store
sue
suicidal
suicide
supplements
supply company
theft
thief
thieves
tit
toddler
toddlers
toxic
toxin
tragedy
treating dka
treating hypoglycemia
treatment for hypoglycemia
vagina
violence
whore
withdrawal
without prescription
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-imn')]
div[contains(@class, 'pane-pub-home-imn')]
div[contains(@class, 'pane-pub-topic-imn')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Supercharge your medical practice with ChatGPT: Here’s why you should upgrade
Artificial intelligence (AI) has already demonstrated its potential in various areas of healthcare, from early disease detection and drug discovery to genomics and personalized care. OpenAI’s ChatGPT, a large language model, is one AI tool that has been transforming practices across the globe, including mine.
Let me walk you through it.
ChatGPT is essentially an AI-fueled assistant, capable of interpreting and generating human-like text in response to user inputs. Imagine a well-informed and competent trainee working with you, ready to tackle tasks from handling patient inquiries to summarizing intricate medical literature.
Currently, ChatGPT works on the “freemium” pricing model; there is a free version built upon GPT-3.5 as well as a subscription “ChatGPT Plus” version based on GPT-4 which offers additional features such as the use of third-party plug-ins.
Now, you may ask, “Isn’t the free version enough?” The free version is indeed impressive, but upgrading to the paid version for $20 per month unlocks the full potential of this tool, particularly if we add plug-ins.
Here are some of the best ways to incorporate ChatGPT Plus into your practice.
Time saver and efficiency multiplier. The paid version of ChatGPT is an extraordinary time-saving tool. It can help you sort through vast amounts of medical literature in a fraction of the time it would normally take. Imagine having to sift through hundreds of articles to find the latest research relevant to a patient’s case. With the paid version of ChatGPT, you can simply ask it to provide summaries of the most recent and relevant studies, all in seconds.
Did you forget about that PowerPoint you need to make but know the potential papers you would use? No problem. ChatGPT can create slides in a few minutes. It becomes your on-demand research assistant.
Of course, you need to provide the source you find most relevant to you. Using plug-ins such as ScholarAI and Link Reader are great.
Improved patient communication. Explaining complex medical terminology and procedures to patients can sometimes be a challenge. ChatGPT can generate simplified and personalized explanations for your patients, fostering their understanding and involvement in their care process.
Epic is currently collaborating with Nuance Communications, Microsoft’s speech recognition subsidiary, to use generative AI tools for medical note-taking in the electronic health record. However, you do not need to wait for it; it just takes a prompt in ChatGPT and then copying/pasting the results into the chart.
Smoother administrative management. The premium version of ChatGPT can automate administrative tasks such as creating letters of medical necessity, clearance to other physicians for services, or even communications to staff on specific topics. This frees you to focus more on your core work: providing patient care.
Precision medicine aid. ChatGPT can be a powerful ally in the field of precision medicine. Its capabilities for analyzing large datasets and unearthing valuable insights can help deliver more personalized and potentially effective treatment plans. For example, one can prompt ChatGPT to query the reported frequency of certain genomic variants and their implications; with the upgraded version and plug-ins, the results will have fewer hallucinations — inaccurate results — and key data references.
Unlimited accessibility. Uninterrupted access is a compelling reason to upgrade. While the free version may have usage limitations, the premium version provides unrestricted, round-the-clock access. Be it a late-night research quest or an early-morning patient query, your AI assistant will always be available.
Strengthened privacy and security. The premium version of ChatGPT includes heightened privacy and security measures. Just make sure to follow HIPAA and not include identifiers when making queries.
Embracing AI tools like ChatGPT in your practice can help you stay at the cutting edge of medical care, saving you time, enhancing patient communication, and supporting you in providing personalized care.
While the free version can serve as a good starting point (there are apps for both iOS and Android), upgrading to the paid version opens up a world of possibilities that can truly supercharge your practice.
I would love to hear your comments on this column or on future topics. Contact me at [email protected].
Arturo Loaiza-Bonilla, MD, MSEd, is the cofounder and chief medical officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr. Loaiza-Bonilla is Assistant Professor of Medicine, Drexel University School of Medicine, Philadelphia, Pennsylvania, and serves as medical director of oncology research at Capital Health in New Jersey, where he maintains a connection to patient care by attending to patients 2 days a week. He has financial relationships with Verify, PSI CRO, Bayer, AstraZeneca, Cardinal Health, BrightInsight, The Lynx Group, Fresenius, Pfizer, Ipsen, Guardant, Amgen, Eisai, Natera, Merck, and Bristol Myers Squibb.
A version of this article appeared on Medscape.com.
Artificial intelligence (AI) has already demonstrated its potential in various areas of healthcare, from early disease detection and drug discovery to genomics and personalized care. OpenAI’s ChatGPT, a large language model, is one AI tool that has been transforming practices across the globe, including mine.
Let me walk you through it.
ChatGPT is essentially an AI-fueled assistant, capable of interpreting and generating human-like text in response to user inputs. Imagine a well-informed and competent trainee working with you, ready to tackle tasks from handling patient inquiries to summarizing intricate medical literature.
Currently, ChatGPT works on the “freemium” pricing model; there is a free version built upon GPT-3.5 as well as a subscription “ChatGPT Plus” version based on GPT-4 which offers additional features such as the use of third-party plug-ins.
Now, you may ask, “Isn’t the free version enough?” The free version is indeed impressive, but upgrading to the paid version for $20 per month unlocks the full potential of this tool, particularly if we add plug-ins.
Here are some of the best ways to incorporate ChatGPT Plus into your practice.
Time saver and efficiency multiplier. The paid version of ChatGPT is an extraordinary time-saving tool. It can help you sort through vast amounts of medical literature in a fraction of the time it would normally take. Imagine having to sift through hundreds of articles to find the latest research relevant to a patient’s case. With the paid version of ChatGPT, you can simply ask it to provide summaries of the most recent and relevant studies, all in seconds.
Did you forget about that PowerPoint you need to make but know the potential papers you would use? No problem. ChatGPT can create slides in a few minutes. It becomes your on-demand research assistant.
Of course, you need to provide the source you find most relevant to you. Using plug-ins such as ScholarAI and Link Reader are great.
Improved patient communication. Explaining complex medical terminology and procedures to patients can sometimes be a challenge. ChatGPT can generate simplified and personalized explanations for your patients, fostering their understanding and involvement in their care process.
Epic is currently collaborating with Nuance Communications, Microsoft’s speech recognition subsidiary, to use generative AI tools for medical note-taking in the electronic health record. However, you do not need to wait for it; it just takes a prompt in ChatGPT and then copying/pasting the results into the chart.
Smoother administrative management. The premium version of ChatGPT can automate administrative tasks such as creating letters of medical necessity, clearance to other physicians for services, or even communications to staff on specific topics. This frees you to focus more on your core work: providing patient care.
Precision medicine aid. ChatGPT can be a powerful ally in the field of precision medicine. Its capabilities for analyzing large datasets and unearthing valuable insights can help deliver more personalized and potentially effective treatment plans. For example, one can prompt ChatGPT to query the reported frequency of certain genomic variants and their implications; with the upgraded version and plug-ins, the results will have fewer hallucinations — inaccurate results — and key data references.
Unlimited accessibility. Uninterrupted access is a compelling reason to upgrade. While the free version may have usage limitations, the premium version provides unrestricted, round-the-clock access. Be it a late-night research quest or an early-morning patient query, your AI assistant will always be available.
Strengthened privacy and security. The premium version of ChatGPT includes heightened privacy and security measures. Just make sure to follow HIPAA and not include identifiers when making queries.
Embracing AI tools like ChatGPT in your practice can help you stay at the cutting edge of medical care, saving you time, enhancing patient communication, and supporting you in providing personalized care.
While the free version can serve as a good starting point (there are apps for both iOS and Android), upgrading to the paid version opens up a world of possibilities that can truly supercharge your practice.
I would love to hear your comments on this column or on future topics. Contact me at [email protected].
Arturo Loaiza-Bonilla, MD, MSEd, is the cofounder and chief medical officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr. Loaiza-Bonilla is Assistant Professor of Medicine, Drexel University School of Medicine, Philadelphia, Pennsylvania, and serves as medical director of oncology research at Capital Health in New Jersey, where he maintains a connection to patient care by attending to patients 2 days a week. He has financial relationships with Verify, PSI CRO, Bayer, AstraZeneca, Cardinal Health, BrightInsight, The Lynx Group, Fresenius, Pfizer, Ipsen, Guardant, Amgen, Eisai, Natera, Merck, and Bristol Myers Squibb.
A version of this article appeared on Medscape.com.
Artificial intelligence (AI) has already demonstrated its potential in various areas of healthcare, from early disease detection and drug discovery to genomics and personalized care. OpenAI’s ChatGPT, a large language model, is one AI tool that has been transforming practices across the globe, including mine.
Let me walk you through it.
ChatGPT is essentially an AI-fueled assistant, capable of interpreting and generating human-like text in response to user inputs. Imagine a well-informed and competent trainee working with you, ready to tackle tasks from handling patient inquiries to summarizing intricate medical literature.
Currently, ChatGPT works on the “freemium” pricing model; there is a free version built upon GPT-3.5 as well as a subscription “ChatGPT Plus” version based on GPT-4 which offers additional features such as the use of third-party plug-ins.
Now, you may ask, “Isn’t the free version enough?” The free version is indeed impressive, but upgrading to the paid version for $20 per month unlocks the full potential of this tool, particularly if we add plug-ins.
Here are some of the best ways to incorporate ChatGPT Plus into your practice.
Time saver and efficiency multiplier. The paid version of ChatGPT is an extraordinary time-saving tool. It can help you sort through vast amounts of medical literature in a fraction of the time it would normally take. Imagine having to sift through hundreds of articles to find the latest research relevant to a patient’s case. With the paid version of ChatGPT, you can simply ask it to provide summaries of the most recent and relevant studies, all in seconds.
Did you forget about that PowerPoint you need to make but know the potential papers you would use? No problem. ChatGPT can create slides in a few minutes. It becomes your on-demand research assistant.
Of course, you need to provide the source you find most relevant to you. Using plug-ins such as ScholarAI and Link Reader are great.
Improved patient communication. Explaining complex medical terminology and procedures to patients can sometimes be a challenge. ChatGPT can generate simplified and personalized explanations for your patients, fostering their understanding and involvement in their care process.
Epic is currently collaborating with Nuance Communications, Microsoft’s speech recognition subsidiary, to use generative AI tools for medical note-taking in the electronic health record. However, you do not need to wait for it; it just takes a prompt in ChatGPT and then copying/pasting the results into the chart.
Smoother administrative management. The premium version of ChatGPT can automate administrative tasks such as creating letters of medical necessity, clearance to other physicians for services, or even communications to staff on specific topics. This frees you to focus more on your core work: providing patient care.
Precision medicine aid. ChatGPT can be a powerful ally in the field of precision medicine. Its capabilities for analyzing large datasets and unearthing valuable insights can help deliver more personalized and potentially effective treatment plans. For example, one can prompt ChatGPT to query the reported frequency of certain genomic variants and their implications; with the upgraded version and plug-ins, the results will have fewer hallucinations — inaccurate results — and key data references.
Unlimited accessibility. Uninterrupted access is a compelling reason to upgrade. While the free version may have usage limitations, the premium version provides unrestricted, round-the-clock access. Be it a late-night research quest or an early-morning patient query, your AI assistant will always be available.
Strengthened privacy and security. The premium version of ChatGPT includes heightened privacy and security measures. Just make sure to follow HIPAA and not include identifiers when making queries.
Embracing AI tools like ChatGPT in your practice can help you stay at the cutting edge of medical care, saving you time, enhancing patient communication, and supporting you in providing personalized care.
While the free version can serve as a good starting point (there are apps for both iOS and Android), upgrading to the paid version opens up a world of possibilities that can truly supercharge your practice.
I would love to hear your comments on this column or on future topics. Contact me at [email protected].
Arturo Loaiza-Bonilla, MD, MSEd, is the cofounder and chief medical officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr. Loaiza-Bonilla is Assistant Professor of Medicine, Drexel University School of Medicine, Philadelphia, Pennsylvania, and serves as medical director of oncology research at Capital Health in New Jersey, where he maintains a connection to patient care by attending to patients 2 days a week. He has financial relationships with Verify, PSI CRO, Bayer, AstraZeneca, Cardinal Health, BrightInsight, The Lynx Group, Fresenius, Pfizer, Ipsen, Guardant, Amgen, Eisai, Natera, Merck, and Bristol Myers Squibb.
A version of this article appeared on Medscape.com.
What if a single GLP-1 shot could last for months?
As revolutionary as glucagon-like peptide 1 (GLP-1) drugs are, they still last for only so long in the body. Patients with diabetes typically must be injected once or twice a day (liraglutide) or once a week (semaglutide). This could hinder proper diabetes management, as adherence tends to go down the more frequent the dose.
But what if a single GLP-1 injection could last for 4 months?
“melts away like a sugar cube dissolving in water, molecule by molecule,” said Eric Appel, PhD, the project’s principal investigator and an associate professor of materials science and engineering at Stanford (Calif.) University.
So far, the team has tested the new drug delivery system in rats, and they say human clinical trials could start within 2 years.
Mathematical modeling indicated that one shot of liraglutide could maintain exposure in humans for 120 days, or about 4 months, according to their study in Cell Reports Medicine.
“Patient adherence is of critical importance to diabetes care,” said Alex Abramson, PhD, assistant professor in the chemical and biomolecular engineering department at Georgia Tech, who was not involved in the study. “It’s very exciting to have a potential new system that can last 4 months on a single injection.”
Long-Acting Injectables Have Come a Long Way
The first long-acting injectable — Lupron Depot, a monthly treatment for advanced prostate cancer — was approved in 1989. Since then, long-acting injectable depots have revolutionized the treatment and management of conditions ranging from osteoarthritis knee pain to schizophrenia to opioid use disorder. In 2021, the US Food and Drug Administration approved Apretude — an injectable treatment for HIV pre-exposure prevention that needs to be given every 2 months, compared with daily for the pill equivalent. Other new and innovative developments are underway: Researchers at the University of Connecticut are working on a transdermal microneedle patch — with many tiny vaccine-loaded needles — that could provide multiple doses of a vaccine over time, no boosters needed.
At Stanford, Appel’s lab has spent years developing gels for drug delivery. His team uses a class of hydrogel called polymer-nanoparticle (PNP), which features weakly bound polymers and nanoparticles that can dissipate slowly over time.
The goal is to address a longstanding challenge with long-acting formulations: Achieving steady release. Because the hydrogel is “self-healing” — able to repair damages and restore its shape — it’s less likely to burst and release its drug cargo too early.
“Our PNP hydrogels possess a number of really unique characteristics,” Dr. Appel said. They have “excellent” biocompatibility, based on animal studies, and could work with a wide range of drugs. In proof-of-concept mouse studies, Dr. Appel and his team have shown that these hydrogels could also be used to make vaccines last longer, ferry cancer immunotherapies directly to tumors, and deliver antibodies for the prevention of infectious diseases like SARS-CoV-2.
Though the recent study on GLP-1s focused on treating type 2 diabetes, the same formulation could also be used to treat obesity, said Dr. Appel.
The researchers tested the tech using two GLP-1 receptor agonists — semaglutide and liraglutide. In rats, one shot maintained therapeutic serum concentrations of semaglutide or liraglutide over 42 days. With semaglutide, a significant portion was released quickly, followed by controlled release. Liraglutide, on the other hand, was released gradually as the hydrogel dissolved. This suggests the liraglutide hydrogel may be better tolerated, as a sudden peak in drug serum concentration is associated with adverse effects.
The researchers used pharmacokinetic modeling to predict how liraglutide would behave in humans with a larger injection volume, finding that a single dose could maintain therapeutic levels for about 4 months.
“Moving forward, it will be important to determine whether a burst release from the formulation causes any side effects,” Dr. Abramson noted. “Furthermore, it will be important to minimize the injection volumes in humans.”
But first, more studies in larger animals are needed. Next, Dr. Appel and his team plan to test the technology in pigs, whose skin and endocrine systems are most like humans’. If those trials go well, Dr. Appel said, human clinical trials could start within 2 years.
A version of this article appeared on Medscape.com.
As revolutionary as glucagon-like peptide 1 (GLP-1) drugs are, they still last for only so long in the body. Patients with diabetes typically must be injected once or twice a day (liraglutide) or once a week (semaglutide). This could hinder proper diabetes management, as adherence tends to go down the more frequent the dose.
But what if a single GLP-1 injection could last for 4 months?
“melts away like a sugar cube dissolving in water, molecule by molecule,” said Eric Appel, PhD, the project’s principal investigator and an associate professor of materials science and engineering at Stanford (Calif.) University.
So far, the team has tested the new drug delivery system in rats, and they say human clinical trials could start within 2 years.
Mathematical modeling indicated that one shot of liraglutide could maintain exposure in humans for 120 days, or about 4 months, according to their study in Cell Reports Medicine.
“Patient adherence is of critical importance to diabetes care,” said Alex Abramson, PhD, assistant professor in the chemical and biomolecular engineering department at Georgia Tech, who was not involved in the study. “It’s very exciting to have a potential new system that can last 4 months on a single injection.”
Long-Acting Injectables Have Come a Long Way
The first long-acting injectable — Lupron Depot, a monthly treatment for advanced prostate cancer — was approved in 1989. Since then, long-acting injectable depots have revolutionized the treatment and management of conditions ranging from osteoarthritis knee pain to schizophrenia to opioid use disorder. In 2021, the US Food and Drug Administration approved Apretude — an injectable treatment for HIV pre-exposure prevention that needs to be given every 2 months, compared with daily for the pill equivalent. Other new and innovative developments are underway: Researchers at the University of Connecticut are working on a transdermal microneedle patch — with many tiny vaccine-loaded needles — that could provide multiple doses of a vaccine over time, no boosters needed.
At Stanford, Appel’s lab has spent years developing gels for drug delivery. His team uses a class of hydrogel called polymer-nanoparticle (PNP), which features weakly bound polymers and nanoparticles that can dissipate slowly over time.
The goal is to address a longstanding challenge with long-acting formulations: Achieving steady release. Because the hydrogel is “self-healing” — able to repair damages and restore its shape — it’s less likely to burst and release its drug cargo too early.
“Our PNP hydrogels possess a number of really unique characteristics,” Dr. Appel said. They have “excellent” biocompatibility, based on animal studies, and could work with a wide range of drugs. In proof-of-concept mouse studies, Dr. Appel and his team have shown that these hydrogels could also be used to make vaccines last longer, ferry cancer immunotherapies directly to tumors, and deliver antibodies for the prevention of infectious diseases like SARS-CoV-2.
Though the recent study on GLP-1s focused on treating type 2 diabetes, the same formulation could also be used to treat obesity, said Dr. Appel.
The researchers tested the tech using two GLP-1 receptor agonists — semaglutide and liraglutide. In rats, one shot maintained therapeutic serum concentrations of semaglutide or liraglutide over 42 days. With semaglutide, a significant portion was released quickly, followed by controlled release. Liraglutide, on the other hand, was released gradually as the hydrogel dissolved. This suggests the liraglutide hydrogel may be better tolerated, as a sudden peak in drug serum concentration is associated with adverse effects.
The researchers used pharmacokinetic modeling to predict how liraglutide would behave in humans with a larger injection volume, finding that a single dose could maintain therapeutic levels for about 4 months.
“Moving forward, it will be important to determine whether a burst release from the formulation causes any side effects,” Dr. Abramson noted. “Furthermore, it will be important to minimize the injection volumes in humans.”
But first, more studies in larger animals are needed. Next, Dr. Appel and his team plan to test the technology in pigs, whose skin and endocrine systems are most like humans’. If those trials go well, Dr. Appel said, human clinical trials could start within 2 years.
A version of this article appeared on Medscape.com.
As revolutionary as glucagon-like peptide 1 (GLP-1) drugs are, they still last for only so long in the body. Patients with diabetes typically must be injected once or twice a day (liraglutide) or once a week (semaglutide). This could hinder proper diabetes management, as adherence tends to go down the more frequent the dose.
But what if a single GLP-1 injection could last for 4 months?
“melts away like a sugar cube dissolving in water, molecule by molecule,” said Eric Appel, PhD, the project’s principal investigator and an associate professor of materials science and engineering at Stanford (Calif.) University.
So far, the team has tested the new drug delivery system in rats, and they say human clinical trials could start within 2 years.
Mathematical modeling indicated that one shot of liraglutide could maintain exposure in humans for 120 days, or about 4 months, according to their study in Cell Reports Medicine.
“Patient adherence is of critical importance to diabetes care,” said Alex Abramson, PhD, assistant professor in the chemical and biomolecular engineering department at Georgia Tech, who was not involved in the study. “It’s very exciting to have a potential new system that can last 4 months on a single injection.”
Long-Acting Injectables Have Come a Long Way
The first long-acting injectable — Lupron Depot, a monthly treatment for advanced prostate cancer — was approved in 1989. Since then, long-acting injectable depots have revolutionized the treatment and management of conditions ranging from osteoarthritis knee pain to schizophrenia to opioid use disorder. In 2021, the US Food and Drug Administration approved Apretude — an injectable treatment for HIV pre-exposure prevention that needs to be given every 2 months, compared with daily for the pill equivalent. Other new and innovative developments are underway: Researchers at the University of Connecticut are working on a transdermal microneedle patch — with many tiny vaccine-loaded needles — that could provide multiple doses of a vaccine over time, no boosters needed.
At Stanford, Appel’s lab has spent years developing gels for drug delivery. His team uses a class of hydrogel called polymer-nanoparticle (PNP), which features weakly bound polymers and nanoparticles that can dissipate slowly over time.
The goal is to address a longstanding challenge with long-acting formulations: Achieving steady release. Because the hydrogel is “self-healing” — able to repair damages and restore its shape — it’s less likely to burst and release its drug cargo too early.
“Our PNP hydrogels possess a number of really unique characteristics,” Dr. Appel said. They have “excellent” biocompatibility, based on animal studies, and could work with a wide range of drugs. In proof-of-concept mouse studies, Dr. Appel and his team have shown that these hydrogels could also be used to make vaccines last longer, ferry cancer immunotherapies directly to tumors, and deliver antibodies for the prevention of infectious diseases like SARS-CoV-2.
Though the recent study on GLP-1s focused on treating type 2 diabetes, the same formulation could also be used to treat obesity, said Dr. Appel.
The researchers tested the tech using two GLP-1 receptor agonists — semaglutide and liraglutide. In rats, one shot maintained therapeutic serum concentrations of semaglutide or liraglutide over 42 days. With semaglutide, a significant portion was released quickly, followed by controlled release. Liraglutide, on the other hand, was released gradually as the hydrogel dissolved. This suggests the liraglutide hydrogel may be better tolerated, as a sudden peak in drug serum concentration is associated with adverse effects.
The researchers used pharmacokinetic modeling to predict how liraglutide would behave in humans with a larger injection volume, finding that a single dose could maintain therapeutic levels for about 4 months.
“Moving forward, it will be important to determine whether a burst release from the formulation causes any side effects,” Dr. Abramson noted. “Furthermore, it will be important to minimize the injection volumes in humans.”
But first, more studies in larger animals are needed. Next, Dr. Appel and his team plan to test the technology in pigs, whose skin and endocrine systems are most like humans’. If those trials go well, Dr. Appel said, human clinical trials could start within 2 years.
A version of this article appeared on Medscape.com.
FROM CELL REPORTS MEDICINE
Electronic Health Records — Recent Survey Results
I have been writing about electronic health records since the mid-1990s. While the basic concept has always been sound, I have always been (and continue to be) a critic of its implementation, which I have compared to the work of the Underpants Gnomes from the television show South Park.
You may recall that Phase One of the Gnomes’ grand scheme was to collect underpants, and Phase Three was to reap enormous profits. Unfortunately, they never quite figured out Phase Two.
EHR’s problems have run a similar course, ever since George W. Bush introduced the EHR Incentive Program (later renamed the Promoting Interoperability Program) in 2000. “By computerizing health records,” the president said, “we can avoid dangerous medical mistakes, reduce costs, and improve care.” That was the ultimate goal — Phase Three, if you will — but nearly a quarter-century later, we are still struggling with Phase Two.
According to the results of a recent survey by this news organization, progress has been made, but issues with usability, reliability, and patient privacy remain.
surveys, respectively. But 56% of them continue to worry about harmful effects from incorrect or misdirected information as a result of inputs from multiple sources, and the rapid turnover of staff that is doing the inputting. Many doctors worry about the potential for incorrect medications and “rule out” diagnoses getting embedded in some patients’ records and undermining future care.
The lack of information sharing among different EHR systems has been the technology’s greatest unmet promise, according to the survey. A lack of interoperability was cited as the most common reason for switching EHR systems. Other reasons included difficulties in clinical documentation and extracting data for quality reporting, as well as the inability to merge inpatient and outpatient records.
A clear majority (72%) felt EHR systems are getting easier to use. The recent decrease in government mandates has freed vendors to work on improving ease of documentation and information retrieval. The incorporation of virtual assistants and other artificial intelligence–based features (as I discussed in two recent columns) have also contributed to improved overall usability. Some newer applications even allow users to build workarounds to compensate for inherent deficiencies in the system.
Physicians tended to be most praiseworthy of functions related to electronic prescribing and retrieval of individual patient data. They felt that much more improvement was needed in helpful prompt features, internal messaging, and communications from patients.
The survey found that 38% of physicians “always” or “often” copy and paste information in patient charts, with another 37% doing so “occasionally.” Noting some of the problems inherent in copy and paste, such as note bloat, internal inconsistencies, error propagation, and documentation in the wrong patient chart, the survey authors suggest that EHR developers could help by shifting away from timelines that appear as one long note. They could also add functionality to allow new information to be displayed as updates on a digital chart.
Improvement is also needed in the way the EHR affects patient interactions, according to the survey results. Physicians are still often forced to click to a different screen to find lab results, another for current medications, and still another for past notes, all while trying to communicate with the patient. Such issues are likely to decrease in the next few years as doctors gain the ability to give voice commands to AI-based system add-ons to obtain this information.
Security concerns seem to be decreasing. In this year’s survey, nearly half of all physicians voiced no EHR privacy problems or concerns, even though a recent review of medical literature concluded that security risks remain meaningful. Those who did have privacy concerns were mostly worried about hackers and other unauthorized access to patient information.
The survey found that around 40% of EHR systems are not using patient portals to post lab results, diagnoses and procedure notes, or prescriptions. However, other physicians complained that their systems were too prompt in posting results, so that patients often received them before the doctor did. This is certainly another area where improvement at both extremes is necessary.
Other areas in which physicians saw a need for improvement were in system reliability, user training, and ongoing customer service. And among the dwindling ranks of physicians with no EHR experience, the most common reasons given for refusing to invest in an EHR system were affordability and interference with the doctor-patient relationship.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
I have been writing about electronic health records since the mid-1990s. While the basic concept has always been sound, I have always been (and continue to be) a critic of its implementation, which I have compared to the work of the Underpants Gnomes from the television show South Park.
You may recall that Phase One of the Gnomes’ grand scheme was to collect underpants, and Phase Three was to reap enormous profits. Unfortunately, they never quite figured out Phase Two.
EHR’s problems have run a similar course, ever since George W. Bush introduced the EHR Incentive Program (later renamed the Promoting Interoperability Program) in 2000. “By computerizing health records,” the president said, “we can avoid dangerous medical mistakes, reduce costs, and improve care.” That was the ultimate goal — Phase Three, if you will — but nearly a quarter-century later, we are still struggling with Phase Two.
According to the results of a recent survey by this news organization, progress has been made, but issues with usability, reliability, and patient privacy remain.
surveys, respectively. But 56% of them continue to worry about harmful effects from incorrect or misdirected information as a result of inputs from multiple sources, and the rapid turnover of staff that is doing the inputting. Many doctors worry about the potential for incorrect medications and “rule out” diagnoses getting embedded in some patients’ records and undermining future care.
The lack of information sharing among different EHR systems has been the technology’s greatest unmet promise, according to the survey. A lack of interoperability was cited as the most common reason for switching EHR systems. Other reasons included difficulties in clinical documentation and extracting data for quality reporting, as well as the inability to merge inpatient and outpatient records.
A clear majority (72%) felt EHR systems are getting easier to use. The recent decrease in government mandates has freed vendors to work on improving ease of documentation and information retrieval. The incorporation of virtual assistants and other artificial intelligence–based features (as I discussed in two recent columns) have also contributed to improved overall usability. Some newer applications even allow users to build workarounds to compensate for inherent deficiencies in the system.
Physicians tended to be most praiseworthy of functions related to electronic prescribing and retrieval of individual patient data. They felt that much more improvement was needed in helpful prompt features, internal messaging, and communications from patients.
The survey found that 38% of physicians “always” or “often” copy and paste information in patient charts, with another 37% doing so “occasionally.” Noting some of the problems inherent in copy and paste, such as note bloat, internal inconsistencies, error propagation, and documentation in the wrong patient chart, the survey authors suggest that EHR developers could help by shifting away from timelines that appear as one long note. They could also add functionality to allow new information to be displayed as updates on a digital chart.
Improvement is also needed in the way the EHR affects patient interactions, according to the survey results. Physicians are still often forced to click to a different screen to find lab results, another for current medications, and still another for past notes, all while trying to communicate with the patient. Such issues are likely to decrease in the next few years as doctors gain the ability to give voice commands to AI-based system add-ons to obtain this information.
Security concerns seem to be decreasing. In this year’s survey, nearly half of all physicians voiced no EHR privacy problems or concerns, even though a recent review of medical literature concluded that security risks remain meaningful. Those who did have privacy concerns were mostly worried about hackers and other unauthorized access to patient information.
The survey found that around 40% of EHR systems are not using patient portals to post lab results, diagnoses and procedure notes, or prescriptions. However, other physicians complained that their systems were too prompt in posting results, so that patients often received them before the doctor did. This is certainly another area where improvement at both extremes is necessary.
Other areas in which physicians saw a need for improvement were in system reliability, user training, and ongoing customer service. And among the dwindling ranks of physicians with no EHR experience, the most common reasons given for refusing to invest in an EHR system were affordability and interference with the doctor-patient relationship.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
I have been writing about electronic health records since the mid-1990s. While the basic concept has always been sound, I have always been (and continue to be) a critic of its implementation, which I have compared to the work of the Underpants Gnomes from the television show South Park.
You may recall that Phase One of the Gnomes’ grand scheme was to collect underpants, and Phase Three was to reap enormous profits. Unfortunately, they never quite figured out Phase Two.
EHR’s problems have run a similar course, ever since George W. Bush introduced the EHR Incentive Program (later renamed the Promoting Interoperability Program) in 2000. “By computerizing health records,” the president said, “we can avoid dangerous medical mistakes, reduce costs, and improve care.” That was the ultimate goal — Phase Three, if you will — but nearly a quarter-century later, we are still struggling with Phase Two.
According to the results of a recent survey by this news organization, progress has been made, but issues with usability, reliability, and patient privacy remain.
surveys, respectively. But 56% of them continue to worry about harmful effects from incorrect or misdirected information as a result of inputs from multiple sources, and the rapid turnover of staff that is doing the inputting. Many doctors worry about the potential for incorrect medications and “rule out” diagnoses getting embedded in some patients’ records and undermining future care.
The lack of information sharing among different EHR systems has been the technology’s greatest unmet promise, according to the survey. A lack of interoperability was cited as the most common reason for switching EHR systems. Other reasons included difficulties in clinical documentation and extracting data for quality reporting, as well as the inability to merge inpatient and outpatient records.
A clear majority (72%) felt EHR systems are getting easier to use. The recent decrease in government mandates has freed vendors to work on improving ease of documentation and information retrieval. The incorporation of virtual assistants and other artificial intelligence–based features (as I discussed in two recent columns) have also contributed to improved overall usability. Some newer applications even allow users to build workarounds to compensate for inherent deficiencies in the system.
Physicians tended to be most praiseworthy of functions related to electronic prescribing and retrieval of individual patient data. They felt that much more improvement was needed in helpful prompt features, internal messaging, and communications from patients.
The survey found that 38% of physicians “always” or “often” copy and paste information in patient charts, with another 37% doing so “occasionally.” Noting some of the problems inherent in copy and paste, such as note bloat, internal inconsistencies, error propagation, and documentation in the wrong patient chart, the survey authors suggest that EHR developers could help by shifting away from timelines that appear as one long note. They could also add functionality to allow new information to be displayed as updates on a digital chart.
Improvement is also needed in the way the EHR affects patient interactions, according to the survey results. Physicians are still often forced to click to a different screen to find lab results, another for current medications, and still another for past notes, all while trying to communicate with the patient. Such issues are likely to decrease in the next few years as doctors gain the ability to give voice commands to AI-based system add-ons to obtain this information.
Security concerns seem to be decreasing. In this year’s survey, nearly half of all physicians voiced no EHR privacy problems or concerns, even though a recent review of medical literature concluded that security risks remain meaningful. Those who did have privacy concerns were mostly worried about hackers and other unauthorized access to patient information.
The survey found that around 40% of EHR systems are not using patient portals to post lab results, diagnoses and procedure notes, or prescriptions. However, other physicians complained that their systems were too prompt in posting results, so that patients often received them before the doctor did. This is certainly another area where improvement at both extremes is necessary.
Other areas in which physicians saw a need for improvement were in system reliability, user training, and ongoing customer service. And among the dwindling ranks of physicians with no EHR experience, the most common reasons given for refusing to invest in an EHR system were affordability and interference with the doctor-patient relationship.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
How to prescribe Zepbound
December marks the advent of the approval of tirzepatide (Zepbound) for on-label treatment of obesity. In November 2023, the US Food and Drug Administration (FDA) approved it for the treatment of obesity in adults.
In May 2022, the FDA approved Mounjaro, which is tirzepatide, for type 2 diabetes. Since then, many physicians, including myself, have prescribed it off-label for obesity. As an endocrinologist treating both obesity and diabetes, 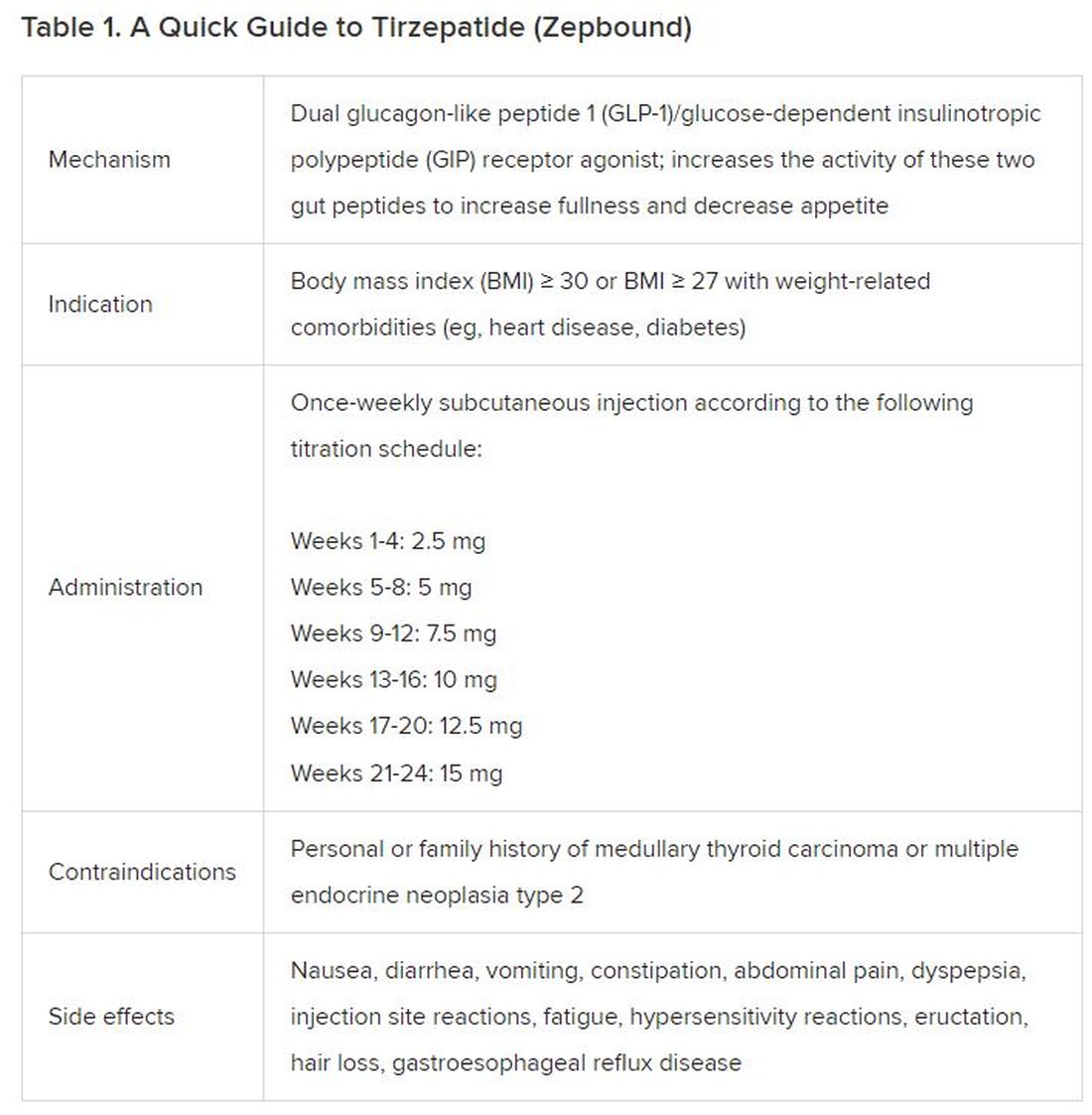
The Expertise
Because GLP-1 receptor agonists have been around since 2005, we’ve had over a decade of clinical experience with these medications. Table 2 provides more nuanced information on tirzepatide (as Zepbound, for obesity) based on our experiences with dulaglutide, liraglutide, semaglutide, and tirzepatide (as Mounjaro).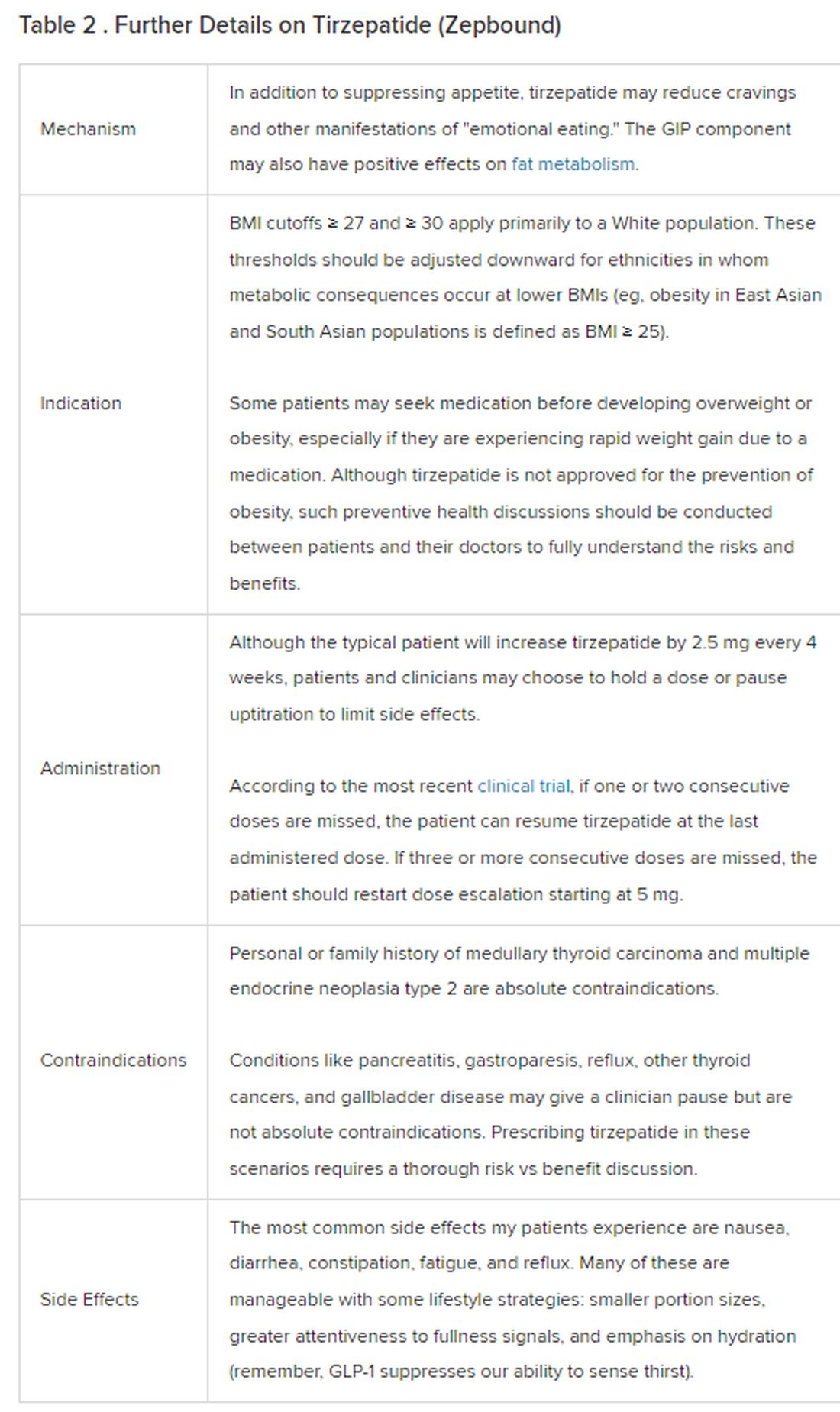
The Reality
In today’s increasingly complex healthcare system, the reality of providing high-quality obesity care is challenging. When discussing tirzepatide with patients, I use a 4 Cs schematic — comorbidities, cautions, costs, choices — to cover the most frequently asked questions.
Comorbidities
In trials, tirzepatide reduced A1c by about 2%. In one diabetes trial, tirzepatide reduced liver fat content significantly more than the comparator (insulin), and trials of tirzepatide in nonalcoholic steatohepatitis are ongoing. A prespecified meta-analysis of tirzepatide and cardiovascular disease estimated a 20% reduction in the risk for cardiovascular death, myocardial infarction, stroke, and hospitalized unstable angina. Tirzepatide as well as other GLP-1 agonists may be beneficial in alcohol use disorder. Prescribing tirzepatide to patients who have or are at risk of developing such comorbidities is an ideal way to target multiple metabolic diseases with one agent.
Cautions
The first principle of medicine is “do no harm.” Tirzepatide may be a poor option for individuals with a history of pancreatitis, gastroparesis, or severe gastroesophageal reflux disease. Because tirzepatide may interfere with the efficacy of estrogen-containing contraceptives during its uptitration phase, women should speak with their doctors about appropriate birth control options (eg, progestin-only, barrier methods). In clinical trials of tirzepatide, male participants were also advised to use reliable contraception. If patients are family-planning, tirzepatide should be discontinued 2 months (for women) and 4 months (for men) before conception, because its effects on fertility or pregnancy are currently unknown.
Costs
At a retail price of $1279 per month, Zepbound is only slightly more affordable than its main competitor, Wegovy (semaglutide 2.4 mg). Complex pharmacy negotiations may reduce this cost, but even with rebates, coupons, and commercial insurance, these costs still place tirzepatide out of reach for many patients. For patients who cannot access tirzepatide, clinicians should discuss more cost-feasible, evidence-based alternatives: for example, phentermine, phentermine-topiramate, naltrexone-bupropion, metformin, bupropion, or topiramate.
Choices
Patient preference drives much of today’s clinical decision-making. Some patients may be switching from semaglutide to tirzepatide, whether by choice or on the basis of physician recommendation. Although no head-to-head obesity trial exists, data from SURPASS-2 and SUSTAIN-FORTE can inform therapeutic equivalence:
- Semaglutide 1.0 mg to tirzepatide 2.5 mg will be a step-down; 5 mg will be a step-up
- Semaglutide 2.0 or 2.4 mg to tirzepatide 5 mg is probably equivalent
The decision to switch therapeutics may depend on weight loss goals, side effect tolerability, or insurance coverage. As with all medications, the use of tirzepatide should progress with shared decision-making, thorough discussions of risks vs benefits, and individualized regimens tailored to each patient’s needs.
The newly approved Zepbound is a valuable addition to our toolbox of obesity treatments. Patients and providers alike are excited for its potential as a highly effective antiobesity medication that can cause a degree of weight loss necessary to reverse comorbidities. The medical management of obesity with agents like tirzepatide holds great promise in addressing today’s obesity epidemic.
Dr. Tchang is Assistant Professor, Clinical Medicine, Division of Endocrinology, Diabetes, and Metabolism, Weill Cornell Medicine; Physician, Department of Medicine, Iris Cantor Women’s Health Center, Comprehensive Weight Control Center, New York, NY. She disclosed ties to Gelesis and Novo Nordisk.
A version of this article appeared on Medscape.com.
December marks the advent of the approval of tirzepatide (Zepbound) for on-label treatment of obesity. In November 2023, the US Food and Drug Administration (FDA) approved it for the treatment of obesity in adults.
In May 2022, the FDA approved Mounjaro, which is tirzepatide, for type 2 diabetes. Since then, many physicians, including myself, have prescribed it off-label for obesity. As an endocrinologist treating both obesity and diabetes, 
The Expertise
Because GLP-1 receptor agonists have been around since 2005, we’ve had over a decade of clinical experience with these medications. Table 2 provides more nuanced information on tirzepatide (as Zepbound, for obesity) based on our experiences with dulaglutide, liraglutide, semaglutide, and tirzepatide (as Mounjaro).
The Reality
In today’s increasingly complex healthcare system, the reality of providing high-quality obesity care is challenging. When discussing tirzepatide with patients, I use a 4 Cs schematic — comorbidities, cautions, costs, choices — to cover the most frequently asked questions.
Comorbidities
In trials, tirzepatide reduced A1c by about 2%. In one diabetes trial, tirzepatide reduced liver fat content significantly more than the comparator (insulin), and trials of tirzepatide in nonalcoholic steatohepatitis are ongoing. A prespecified meta-analysis of tirzepatide and cardiovascular disease estimated a 20% reduction in the risk for cardiovascular death, myocardial infarction, stroke, and hospitalized unstable angina. Tirzepatide as well as other GLP-1 agonists may be beneficial in alcohol use disorder. Prescribing tirzepatide to patients who have or are at risk of developing such comorbidities is an ideal way to target multiple metabolic diseases with one agent.
Cautions
The first principle of medicine is “do no harm.” Tirzepatide may be a poor option for individuals with a history of pancreatitis, gastroparesis, or severe gastroesophageal reflux disease. Because tirzepatide may interfere with the efficacy of estrogen-containing contraceptives during its uptitration phase, women should speak with their doctors about appropriate birth control options (eg, progestin-only, barrier methods). In clinical trials of tirzepatide, male participants were also advised to use reliable contraception. If patients are family-planning, tirzepatide should be discontinued 2 months (for women) and 4 months (for men) before conception, because its effects on fertility or pregnancy are currently unknown.
Costs
At a retail price of $1279 per month, Zepbound is only slightly more affordable than its main competitor, Wegovy (semaglutide 2.4 mg). Complex pharmacy negotiations may reduce this cost, but even with rebates, coupons, and commercial insurance, these costs still place tirzepatide out of reach for many patients. For patients who cannot access tirzepatide, clinicians should discuss more cost-feasible, evidence-based alternatives: for example, phentermine, phentermine-topiramate, naltrexone-bupropion, metformin, bupropion, or topiramate.
Choices
Patient preference drives much of today’s clinical decision-making. Some patients may be switching from semaglutide to tirzepatide, whether by choice or on the basis of physician recommendation. Although no head-to-head obesity trial exists, data from SURPASS-2 and SUSTAIN-FORTE can inform therapeutic equivalence:
- Semaglutide 1.0 mg to tirzepatide 2.5 mg will be a step-down; 5 mg will be a step-up
- Semaglutide 2.0 or 2.4 mg to tirzepatide 5 mg is probably equivalent
The decision to switch therapeutics may depend on weight loss goals, side effect tolerability, or insurance coverage. As with all medications, the use of tirzepatide should progress with shared decision-making, thorough discussions of risks vs benefits, and individualized regimens tailored to each patient’s needs.
The newly approved Zepbound is a valuable addition to our toolbox of obesity treatments. Patients and providers alike are excited for its potential as a highly effective antiobesity medication that can cause a degree of weight loss necessary to reverse comorbidities. The medical management of obesity with agents like tirzepatide holds great promise in addressing today’s obesity epidemic.
Dr. Tchang is Assistant Professor, Clinical Medicine, Division of Endocrinology, Diabetes, and Metabolism, Weill Cornell Medicine; Physician, Department of Medicine, Iris Cantor Women’s Health Center, Comprehensive Weight Control Center, New York, NY. She disclosed ties to Gelesis and Novo Nordisk.
A version of this article appeared on Medscape.com.
December marks the advent of the approval of tirzepatide (Zepbound) for on-label treatment of obesity. In November 2023, the US Food and Drug Administration (FDA) approved it for the treatment of obesity in adults.
In May 2022, the FDA approved Mounjaro, which is tirzepatide, for type 2 diabetes. Since then, many physicians, including myself, have prescribed it off-label for obesity. As an endocrinologist treating both obesity and diabetes, 
The Expertise
Because GLP-1 receptor agonists have been around since 2005, we’ve had over a decade of clinical experience with these medications. Table 2 provides more nuanced information on tirzepatide (as Zepbound, for obesity) based on our experiences with dulaglutide, liraglutide, semaglutide, and tirzepatide (as Mounjaro).
The Reality
In today’s increasingly complex healthcare system, the reality of providing high-quality obesity care is challenging. When discussing tirzepatide with patients, I use a 4 Cs schematic — comorbidities, cautions, costs, choices — to cover the most frequently asked questions.
Comorbidities
In trials, tirzepatide reduced A1c by about 2%. In one diabetes trial, tirzepatide reduced liver fat content significantly more than the comparator (insulin), and trials of tirzepatide in nonalcoholic steatohepatitis are ongoing. A prespecified meta-analysis of tirzepatide and cardiovascular disease estimated a 20% reduction in the risk for cardiovascular death, myocardial infarction, stroke, and hospitalized unstable angina. Tirzepatide as well as other GLP-1 agonists may be beneficial in alcohol use disorder. Prescribing tirzepatide to patients who have or are at risk of developing such comorbidities is an ideal way to target multiple metabolic diseases with one agent.
Cautions
The first principle of medicine is “do no harm.” Tirzepatide may be a poor option for individuals with a history of pancreatitis, gastroparesis, or severe gastroesophageal reflux disease. Because tirzepatide may interfere with the efficacy of estrogen-containing contraceptives during its uptitration phase, women should speak with their doctors about appropriate birth control options (eg, progestin-only, barrier methods). In clinical trials of tirzepatide, male participants were also advised to use reliable contraception. If patients are family-planning, tirzepatide should be discontinued 2 months (for women) and 4 months (for men) before conception, because its effects on fertility or pregnancy are currently unknown.
Costs
At a retail price of $1279 per month, Zepbound is only slightly more affordable than its main competitor, Wegovy (semaglutide 2.4 mg). Complex pharmacy negotiations may reduce this cost, but even with rebates, coupons, and commercial insurance, these costs still place tirzepatide out of reach for many patients. For patients who cannot access tirzepatide, clinicians should discuss more cost-feasible, evidence-based alternatives: for example, phentermine, phentermine-topiramate, naltrexone-bupropion, metformin, bupropion, or topiramate.
Choices
Patient preference drives much of today’s clinical decision-making. Some patients may be switching from semaglutide to tirzepatide, whether by choice or on the basis of physician recommendation. Although no head-to-head obesity trial exists, data from SURPASS-2 and SUSTAIN-FORTE can inform therapeutic equivalence:
- Semaglutide 1.0 mg to tirzepatide 2.5 mg will be a step-down; 5 mg will be a step-up
- Semaglutide 2.0 or 2.4 mg to tirzepatide 5 mg is probably equivalent
The decision to switch therapeutics may depend on weight loss goals, side effect tolerability, or insurance coverage. As with all medications, the use of tirzepatide should progress with shared decision-making, thorough discussions of risks vs benefits, and individualized regimens tailored to each patient’s needs.
The newly approved Zepbound is a valuable addition to our toolbox of obesity treatments. Patients and providers alike are excited for its potential as a highly effective antiobesity medication that can cause a degree of weight loss necessary to reverse comorbidities. The medical management of obesity with agents like tirzepatide holds great promise in addressing today’s obesity epidemic.
Dr. Tchang is Assistant Professor, Clinical Medicine, Division of Endocrinology, Diabetes, and Metabolism, Weill Cornell Medicine; Physician, Department of Medicine, Iris Cantor Women’s Health Center, Comprehensive Weight Control Center, New York, NY. She disclosed ties to Gelesis and Novo Nordisk.
A version of this article appeared on Medscape.com.
What is the link between cellphones and male fertility?
Infertility affects approximately one in six couples worldwide. More than half the time, it is the man’s low sperm quality that is to blame. Over the last three decades, sperm quality seems to have declined for no clearly identifiable reason. Theories are running rampant without anyone having the proof to back them up.
Potential Causes
The environment, lifestyle, excess weight or obesity, smoking, alcohol consumption, and psychological stress have all been alternately offered up as potential causes, following low-quality epidemiological studies. Cellphones are not exempt from this list, due to their emission of high-frequency (800-2200 MHz) electromagnetic waves that can be absorbed by the body.
Clinical trials conducted in rats or mice suggest that these waves can affect sperm quality and lead to histological changes to the testicles, bearing in mind that the conditions met in these trials are very far from our day-to-day exposure to electromagnetic waves, mostly via our cellphones.
The same observation can be made about experiments conducted on human sperm in vitro, but changes to the latter caused by electromagnetic waves leave doubts. Observational studies are rare, carried out in small cohorts, and marred by largely conflicting results. Publication bias plays a major role, just as much as the abundance of potential confounding factors does.
Swiss Observational Study
An observational study carried out in Switzerland had the benefit of involving a large cohort of 2886 young men who were representative of the general population. The participants completed an online questionnaire describing their relationship with their cellphone in detail and in qualitative and quantitative terms.
The study was launched in 2005, before cellphone use became so widespread, and this timeline was considered when looking for a link between cellphone exposure and sperm quality. In addition, multiple adjustments were made in the multivariate analyses to account for as many potential confounding factors as possible.
The participants, aged between 18 and 22 years, were recruited during a 3-day period to assess their suitability for military service. Each year, this cohort makes up 97% of the male population in Switzerland in this age range, with the remaining 3% being excluded from the selection process due to disability or chronic illness.
Regardless of the review board’s decision, subjects wishing to take part in the study were given a detailed description of what it involved, a consent form, and two questionnaires. The first focused on the individual directly, asking questions about his health and lifestyle. The second, intended for his parents, dealt with the period before conception.
This recruitment, which took place between September 2005 and November 2018, involved the researchers contacting 106,924 men. Ultimately, only 5.3% of subjects contacted returned the completed documentation. In the end, the study involved 2886 participants (3.1%) who provided all the necessary information, especially the laboratory testing (including a sperm analysis) needed to meet the study objectives. The number of hours spent on a smartphone and how it was used were routinely considered, as was sperm quality (volume, concentration, and total sperm count, as well as sperm mobility and morphology).
Significant Associations
A data analysis using an adjusted linear model revealed a significant association between frequent phone use (> 20 times per day) and lower sperm concentration (in mL) (adjusted β: -0.152, 95% CI -0.316 to 0.011). The same was found for their total concentration in ejaculate (adjusted β: -0.271, 95% CI -0.515 to -0.027).
An adjusted logistic regression analysis estimated that the risk for subnormal male fertility levels, as determined by the World Health Organization (WHO), was increased by at most 30%, when referring to the concentration of sperm per mL (21% in terms of total concentration). This inverse link was shown to be more pronounced during the first phase of the study (2005-2007), compared with the other two phases (2008-2011 and 2012-2018). Yet no links involving sperm mobility or morphology were found, and carrying a cellphone in a trouser pocket had no impact on the results.
This study certainly involves a large cohort of nearly 3000 young men. It is, nonetheless, retrospective, and its methodology, despite being better than that of previous studies, is still open to criticism. Its results can only fuel hypotheses, nothing more. Only prospective cohort studies will allow conclusions to be drawn and, in the meantime,
This article was translated from JIM, which is part of the Medscape professional network. A version of this article appeared on Medscape.com.
Infertility affects approximately one in six couples worldwide. More than half the time, it is the man’s low sperm quality that is to blame. Over the last three decades, sperm quality seems to have declined for no clearly identifiable reason. Theories are running rampant without anyone having the proof to back them up.
Potential Causes
The environment, lifestyle, excess weight or obesity, smoking, alcohol consumption, and psychological stress have all been alternately offered up as potential causes, following low-quality epidemiological studies. Cellphones are not exempt from this list, due to their emission of high-frequency (800-2200 MHz) electromagnetic waves that can be absorbed by the body.
Clinical trials conducted in rats or mice suggest that these waves can affect sperm quality and lead to histological changes to the testicles, bearing in mind that the conditions met in these trials are very far from our day-to-day exposure to electromagnetic waves, mostly via our cellphones.
The same observation can be made about experiments conducted on human sperm in vitro, but changes to the latter caused by electromagnetic waves leave doubts. Observational studies are rare, carried out in small cohorts, and marred by largely conflicting results. Publication bias plays a major role, just as much as the abundance of potential confounding factors does.
Swiss Observational Study
An observational study carried out in Switzerland had the benefit of involving a large cohort of 2886 young men who were representative of the general population. The participants completed an online questionnaire describing their relationship with their cellphone in detail and in qualitative and quantitative terms.
The study was launched in 2005, before cellphone use became so widespread, and this timeline was considered when looking for a link between cellphone exposure and sperm quality. In addition, multiple adjustments were made in the multivariate analyses to account for as many potential confounding factors as possible.
The participants, aged between 18 and 22 years, were recruited during a 3-day period to assess their suitability for military service. Each year, this cohort makes up 97% of the male population in Switzerland in this age range, with the remaining 3% being excluded from the selection process due to disability or chronic illness.
Regardless of the review board’s decision, subjects wishing to take part in the study were given a detailed description of what it involved, a consent form, and two questionnaires. The first focused on the individual directly, asking questions about his health and lifestyle. The second, intended for his parents, dealt with the period before conception.
This recruitment, which took place between September 2005 and November 2018, involved the researchers contacting 106,924 men. Ultimately, only 5.3% of subjects contacted returned the completed documentation. In the end, the study involved 2886 participants (3.1%) who provided all the necessary information, especially the laboratory testing (including a sperm analysis) needed to meet the study objectives. The number of hours spent on a smartphone and how it was used were routinely considered, as was sperm quality (volume, concentration, and total sperm count, as well as sperm mobility and morphology).
Significant Associations
A data analysis using an adjusted linear model revealed a significant association between frequent phone use (> 20 times per day) and lower sperm concentration (in mL) (adjusted β: -0.152, 95% CI -0.316 to 0.011). The same was found for their total concentration in ejaculate (adjusted β: -0.271, 95% CI -0.515 to -0.027).
An adjusted logistic regression analysis estimated that the risk for subnormal male fertility levels, as determined by the World Health Organization (WHO), was increased by at most 30%, when referring to the concentration of sperm per mL (21% in terms of total concentration). This inverse link was shown to be more pronounced during the first phase of the study (2005-2007), compared with the other two phases (2008-2011 and 2012-2018). Yet no links involving sperm mobility or morphology were found, and carrying a cellphone in a trouser pocket had no impact on the results.
This study certainly involves a large cohort of nearly 3000 young men. It is, nonetheless, retrospective, and its methodology, despite being better than that of previous studies, is still open to criticism. Its results can only fuel hypotheses, nothing more. Only prospective cohort studies will allow conclusions to be drawn and, in the meantime,
This article was translated from JIM, which is part of the Medscape professional network. A version of this article appeared on Medscape.com.
Infertility affects approximately one in six couples worldwide. More than half the time, it is the man’s low sperm quality that is to blame. Over the last three decades, sperm quality seems to have declined for no clearly identifiable reason. Theories are running rampant without anyone having the proof to back them up.
Potential Causes
The environment, lifestyle, excess weight or obesity, smoking, alcohol consumption, and psychological stress have all been alternately offered up as potential causes, following low-quality epidemiological studies. Cellphones are not exempt from this list, due to their emission of high-frequency (800-2200 MHz) electromagnetic waves that can be absorbed by the body.
Clinical trials conducted in rats or mice suggest that these waves can affect sperm quality and lead to histological changes to the testicles, bearing in mind that the conditions met in these trials are very far from our day-to-day exposure to electromagnetic waves, mostly via our cellphones.
The same observation can be made about experiments conducted on human sperm in vitro, but changes to the latter caused by electromagnetic waves leave doubts. Observational studies are rare, carried out in small cohorts, and marred by largely conflicting results. Publication bias plays a major role, just as much as the abundance of potential confounding factors does.
Swiss Observational Study
An observational study carried out in Switzerland had the benefit of involving a large cohort of 2886 young men who were representative of the general population. The participants completed an online questionnaire describing their relationship with their cellphone in detail and in qualitative and quantitative terms.
The study was launched in 2005, before cellphone use became so widespread, and this timeline was considered when looking for a link between cellphone exposure and sperm quality. In addition, multiple adjustments were made in the multivariate analyses to account for as many potential confounding factors as possible.
The participants, aged between 18 and 22 years, were recruited during a 3-day period to assess their suitability for military service. Each year, this cohort makes up 97% of the male population in Switzerland in this age range, with the remaining 3% being excluded from the selection process due to disability or chronic illness.
Regardless of the review board’s decision, subjects wishing to take part in the study were given a detailed description of what it involved, a consent form, and two questionnaires. The first focused on the individual directly, asking questions about his health and lifestyle. The second, intended for his parents, dealt with the period before conception.
This recruitment, which took place between September 2005 and November 2018, involved the researchers contacting 106,924 men. Ultimately, only 5.3% of subjects contacted returned the completed documentation. In the end, the study involved 2886 participants (3.1%) who provided all the necessary information, especially the laboratory testing (including a sperm analysis) needed to meet the study objectives. The number of hours spent on a smartphone and how it was used were routinely considered, as was sperm quality (volume, concentration, and total sperm count, as well as sperm mobility and morphology).
Significant Associations
A data analysis using an adjusted linear model revealed a significant association between frequent phone use (> 20 times per day) and lower sperm concentration (in mL) (adjusted β: -0.152, 95% CI -0.316 to 0.011). The same was found for their total concentration in ejaculate (adjusted β: -0.271, 95% CI -0.515 to -0.027).
An adjusted logistic regression analysis estimated that the risk for subnormal male fertility levels, as determined by the World Health Organization (WHO), was increased by at most 30%, when referring to the concentration of sperm per mL (21% in terms of total concentration). This inverse link was shown to be more pronounced during the first phase of the study (2005-2007), compared with the other two phases (2008-2011 and 2012-2018). Yet no links involving sperm mobility or morphology were found, and carrying a cellphone in a trouser pocket had no impact on the results.
This study certainly involves a large cohort of nearly 3000 young men. It is, nonetheless, retrospective, and its methodology, despite being better than that of previous studies, is still open to criticism. Its results can only fuel hypotheses, nothing more. Only prospective cohort studies will allow conclusions to be drawn and, in the meantime,
This article was translated from JIM, which is part of the Medscape professional network. A version of this article appeared on Medscape.com.
Children who are overweight at risk for chronic kidney disease
TOPLINE
, with the association, though weaker, still significant among those who do not develop type 2 diabetes or hypertension, in a large cohort study.
METHODOLOGY
- The study included data on 593,660 adolescents aged 16-20, born after January 1, 1975, who had medical assessments as part of mandatory military service in Israel.
- The mean age at study entry was 17.2 and 54.5% were male.
- Early CKD was defined as stage 1 to 2 CKD with moderately or severely increased albuminuria, with an estimated glomerular filtration rate of 60 mL/min/1.73 m2 or higher.
- The study excluded those with kidney pathology, albuminuria, hypertension, dysglycemia, or missing blood pressure or BMI data.
- Participants were followed up until early CKD onset, death, the last day insured, or August 23, 2020.
TAKEAWAY
- With a mean follow-up of 13.4 years, 1963 adolescents (0.3%) overall developed early chronic kidney disease. Among males, an increased risk of developing CKD was observed with a high-normal BMI in adolescence (hazard ratio [HR], 1.8); with overweight BMI (HR, 4.0); with mild obesity (HR, 6.7); and severe obesity (HR, 9.4).
- Among females, the increased risk was also observed with high-normal BMI (HR 1.4); overweight (HR, 2.3); mild obesity (HR, 2.7); and severe obesity (HR, 4.3).
- In excluding those who developed diabetes or hypertension, the overall rate of early CKD in the cohort was 0.2%.
- For males without diabetes or hypertension, the adjusted HR for early CKD with high-normal weight was 1.2; for overweight, HR 1.6; for mild obesity, HR 2.2; and for severe obesity, HR 2.7.
- For females without diabetes or hypertension, the corresponding increased risk for early CKD was HR 1.2 for high-normal BMI; HR 1.8 for overweight; 1.5 for mild obesity and 2.3 for severe obesity.
IN PRACTICE
“These findings suggest that adolescent obesity is a major risk factor for early CKD in young adulthood; this underscores the importance of mitigating adolescent obesity rates and managing risk factors for kidney disease in adolescents with high BMI,” the authors report.
“The association was evident even in persons with high-normal BMI in adolescence, was more pronounced in men, and appeared before the age of 30 years,” they say.
“Given the increasing obesity rates among adolescents, our findings are a harbinger of the potentially preventable increasing burden of CKD and subsequent cardiovascular disease.”
SOURCE
The study was conducted by first author Avishai M. Tsur, MD, of the Israel Defense Forces, Medical Corps, Tel Hashomer, Ramat Gan, Israel and Department of Military Medicine, Hebrew University of Jerusalem Faculty of Medicine, Jerusalem, Israel, and colleagues. The study was published online in JAMA Pediatrics.
LIMITATIONS
The study lacked longitudinal data on clinical and lifestyle factors, including stress, diet and physical activity. While adolescents were screened using urine dipstick, a lack of serum creatinine measurements could have missed some adolescents with reduced eGFR at the study entry. The generalizability of the results is limited by the lack of people from West Africa and East Asia in the study population.
DISCLOSURES
Coauthor Josef Coresh, MD, reported receiving grants from the National Institutes of Health outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE
, with the association, though weaker, still significant among those who do not develop type 2 diabetes or hypertension, in a large cohort study.
METHODOLOGY
- The study included data on 593,660 adolescents aged 16-20, born after January 1, 1975, who had medical assessments as part of mandatory military service in Israel.
- The mean age at study entry was 17.2 and 54.5% were male.
- Early CKD was defined as stage 1 to 2 CKD with moderately or severely increased albuminuria, with an estimated glomerular filtration rate of 60 mL/min/1.73 m2 or higher.
- The study excluded those with kidney pathology, albuminuria, hypertension, dysglycemia, or missing blood pressure or BMI data.
- Participants were followed up until early CKD onset, death, the last day insured, or August 23, 2020.
TAKEAWAY
- With a mean follow-up of 13.4 years, 1963 adolescents (0.3%) overall developed early chronic kidney disease. Among males, an increased risk of developing CKD was observed with a high-normal BMI in adolescence (hazard ratio [HR], 1.8); with overweight BMI (HR, 4.0); with mild obesity (HR, 6.7); and severe obesity (HR, 9.4).
- Among females, the increased risk was also observed with high-normal BMI (HR 1.4); overweight (HR, 2.3); mild obesity (HR, 2.7); and severe obesity (HR, 4.3).
- In excluding those who developed diabetes or hypertension, the overall rate of early CKD in the cohort was 0.2%.
- For males without diabetes or hypertension, the adjusted HR for early CKD with high-normal weight was 1.2; for overweight, HR 1.6; for mild obesity, HR 2.2; and for severe obesity, HR 2.7.
- For females without diabetes or hypertension, the corresponding increased risk for early CKD was HR 1.2 for high-normal BMI; HR 1.8 for overweight; 1.5 for mild obesity and 2.3 for severe obesity.
IN PRACTICE
“These findings suggest that adolescent obesity is a major risk factor for early CKD in young adulthood; this underscores the importance of mitigating adolescent obesity rates and managing risk factors for kidney disease in adolescents with high BMI,” the authors report.
“The association was evident even in persons with high-normal BMI in adolescence, was more pronounced in men, and appeared before the age of 30 years,” they say.
“Given the increasing obesity rates among adolescents, our findings are a harbinger of the potentially preventable increasing burden of CKD and subsequent cardiovascular disease.”
SOURCE
The study was conducted by first author Avishai M. Tsur, MD, of the Israel Defense Forces, Medical Corps, Tel Hashomer, Ramat Gan, Israel and Department of Military Medicine, Hebrew University of Jerusalem Faculty of Medicine, Jerusalem, Israel, and colleagues. The study was published online in JAMA Pediatrics.
LIMITATIONS
The study lacked longitudinal data on clinical and lifestyle factors, including stress, diet and physical activity. While adolescents were screened using urine dipstick, a lack of serum creatinine measurements could have missed some adolescents with reduced eGFR at the study entry. The generalizability of the results is limited by the lack of people from West Africa and East Asia in the study population.
DISCLOSURES
Coauthor Josef Coresh, MD, reported receiving grants from the National Institutes of Health outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE
, with the association, though weaker, still significant among those who do not develop type 2 diabetes or hypertension, in a large cohort study.
METHODOLOGY
- The study included data on 593,660 adolescents aged 16-20, born after January 1, 1975, who had medical assessments as part of mandatory military service in Israel.
- The mean age at study entry was 17.2 and 54.5% were male.
- Early CKD was defined as stage 1 to 2 CKD with moderately or severely increased albuminuria, with an estimated glomerular filtration rate of 60 mL/min/1.73 m2 or higher.
- The study excluded those with kidney pathology, albuminuria, hypertension, dysglycemia, or missing blood pressure or BMI data.
- Participants were followed up until early CKD onset, death, the last day insured, or August 23, 2020.
TAKEAWAY
- With a mean follow-up of 13.4 years, 1963 adolescents (0.3%) overall developed early chronic kidney disease. Among males, an increased risk of developing CKD was observed with a high-normal BMI in adolescence (hazard ratio [HR], 1.8); with overweight BMI (HR, 4.0); with mild obesity (HR, 6.7); and severe obesity (HR, 9.4).
- Among females, the increased risk was also observed with high-normal BMI (HR 1.4); overweight (HR, 2.3); mild obesity (HR, 2.7); and severe obesity (HR, 4.3).
- In excluding those who developed diabetes or hypertension, the overall rate of early CKD in the cohort was 0.2%.
- For males without diabetes or hypertension, the adjusted HR for early CKD with high-normal weight was 1.2; for overweight, HR 1.6; for mild obesity, HR 2.2; and for severe obesity, HR 2.7.
- For females without diabetes or hypertension, the corresponding increased risk for early CKD was HR 1.2 for high-normal BMI; HR 1.8 for overweight; 1.5 for mild obesity and 2.3 for severe obesity.
IN PRACTICE
“These findings suggest that adolescent obesity is a major risk factor for early CKD in young adulthood; this underscores the importance of mitigating adolescent obesity rates and managing risk factors for kidney disease in adolescents with high BMI,” the authors report.
“The association was evident even in persons with high-normal BMI in adolescence, was more pronounced in men, and appeared before the age of 30 years,” they say.
“Given the increasing obesity rates among adolescents, our findings are a harbinger of the potentially preventable increasing burden of CKD and subsequent cardiovascular disease.”
SOURCE
The study was conducted by first author Avishai M. Tsur, MD, of the Israel Defense Forces, Medical Corps, Tel Hashomer, Ramat Gan, Israel and Department of Military Medicine, Hebrew University of Jerusalem Faculty of Medicine, Jerusalem, Israel, and colleagues. The study was published online in JAMA Pediatrics.
LIMITATIONS
The study lacked longitudinal data on clinical and lifestyle factors, including stress, diet and physical activity. While adolescents were screened using urine dipstick, a lack of serum creatinine measurements could have missed some adolescents with reduced eGFR at the study entry. The generalizability of the results is limited by the lack of people from West Africa and East Asia in the study population.
DISCLOSURES
Coauthor Josef Coresh, MD, reported receiving grants from the National Institutes of Health outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
New KDIGO guideline encourages use of HCV-positive kidneys for HCV-negative recipients
The Kidney Disease: Improving Global Outcomes (KDIGO) Work Group has updated its guideline concerning the prevention, diagnosis, evaluation, and treatment of hepatitis C virus (HCV) infection in patients with chronic kidney disease (CKD).
Of note, KDIGO now supports transplant of HCV-positive kidneys to HCV-negative recipients.
The guidance document, authored by Ahmed Arslan Yousuf Awan, MD, of Baylor College of Medicine, Houston, and colleagues, was written in light of new evidence that has emerged since the 2018 guideline was published.
“The focused update was triggered by new data on antiviral treatment in patients with advanced stages of CKD (G4, G5, or G5D), transplant of HCV-infected kidneys into uninfected recipients, and evolution of the viewpoint on the role of kidney biopsy in managing kidney disease caused by HCV,” the guideline panelists wrote in Annals of Internal Medicine. “This update is intended to assist clinicians in the care of patients with HCV infection and CKD, including patients receiving dialysis (CKD G5D) and patients with a kidney transplant (CKD G1T-G5T).”
Anjay Rastogi, MD, PhD, professor and clinical chief of nephrology at the David Geffen School of Medicine at UCLA, said the update is both “timely and relevant,” and “will really have an impact on the organ shortage that we have for kidney transplant”
The updates are outlined below.
Expanded Access to HCV-Positive Kidneys
While the 2018 guideline recommended that HCV-positive kidneys be directed to HCV-positive recipients, the new guideline suggests that these kidneys are appropriate for all patients regardless of HCV status.
In support, the panelists cited a follow-up of THINKER-1 trial, which showed that eGFR and quality of life were not negatively affected when HCV-negative patients received an HCV-positive kidney, compared with an HCV-negative kidney. Data from 525 unmatched recipients in 16 other studies support this conclusion, the panelists noted.
Jose Debes, MD, PhD, associate professor at the University of Minnesota, Minneapolis, suggested that this is the most important update to the KDIGO guidelines.
“That [change] would be the main impact of these recommendations,” Dr. Debes said in an interview. “Several centers were already doing this, since some data [were] out there, but I think the fact that they’re making this into a guideline is quite important.”
Dr. Rastogi agreed that this recommendation is the most impactful update.
“That’s a big move,” Dr. Rastogi said in an interview. He predicted that the change will “definitely increase the donor pool, which is very, very important.”
For this new recommendation to have the greatest positive effect, however, Dr. Rastogi suggested that health care providers and treatment centers need to prepare an effective implementation strategy. He emphasized the importance of early communication with patients concerning the safety of HCV-positive kidneys, which depends on early initiation of direct-acting antiviral (DAA) therapy.
In the guideline, Dr. Awan and colleagues reported three documented cases of fibrosing cholestatic hepatitis occurred in patients who did not begin DAA therapy until 30 days after transplant.
“[Patients] should start [DAA treatment] right away,” Dr. Rastogi said, “and sometimes even before the transplant.”
This will require institutional support, he noted, as centers need to ensure that patients are covered for DAA therapy and medication is readily available.
Sofosbuvir Given the Green Light
Compared with the 2018 guideline, which recommended against sofosbuvir in patients with CKD G4 and G5, including those on dialysis, because of concerns about metabolization via the kidneys, the new guideline suggests that sofosbuvir-based DAA regimens are appropriate in patients with glomerular filtration rate (GFR) less than 30 mL/min per 1.73 m2, including those receiving dialysis.
This recommendation was based on a systematic review of 106 studies including both sofosbuvir-based and non-sofosbuvir-based DAA regimens that showed high safety and efficacy for all DAA regimen types across a broad variety of patient types.
“DAAs are highly effective and well tolerated treatments for hepatitis C in patients across all stages of CKD, including those undergoing dialysis and kidney transplant recipients, with no need for dose adjustment,” Dr. Awan and colleagues wrote.
Loosened Biopsy Requirements
Unlike the 2018 guideline, which advised kidney biopsy in HCV-positive patients with clinical evidence of glomerular disease prior to initiating DAA treatment, the new guideline suggests that HCV-infected patients with a typical presentation of immune-complex proliferative glomerulonephritis do not require confirmatory kidney biopsy.
“Because almost all patients with chronic hepatitis C (with or without glomerulonephritis) should be treated with DAAs, a kidney biopsy is unlikely to change management in most patients with hepatitis C and clinical glomerulonephritis,” the panelists wrote.
If kidney disease does not stabilize or improve with achievement of sustained virologic response, or if there is evidence of rapidly progressive glomerulonephritis, then a kidney biopsy should be considered before beginning immunosuppressive therapy, according to the guideline, which includes a flow chart to guide clinicians through this decision-making process.
Individualizing Immunosuppressive Therapy
Consistent with the old guideline, the new guideline recommends DAA treatment with concurrent immunosuppressive therapy for patients with cryoglobulinemic flare or rapidly progressive kidney failure. But in contrast, the new guideline calls for an individualized approach to immunosuppression in patients with nephrotic syndrome.
Dr. Awan and colleagues suggested that “nephrotic-range proteinuria (greater than 3.5 g/d) alone does not warrant use of immunosuppressive treatment because such patients can achieve remission of proteinuria after treatment with DAAs.” Still, if other associated complications — such as anasarca, thromboembolic disease, or severe hypoalbuminemia — are present, then immunosuppressive therapy may be warranted, with rituximab remaining the preferred first-line agent.
More Work Is Needed
Dr. Awan and colleagues concluded the guideline by highlighting areas of unmet need, and how filling these knowledge gaps could lead to additional guideline updates.
“Future studies of kidney donations from HCV-positive donors to HCV-negative recipients are needed to refine and clarify the timing of initiation and duration of DAA therapy and to assess long-term outcomes associated with this practice,” they wrote. “Also, randomized controlled trials are needed to determine which patients with HCV-associated kidney disease can be treated with DAA therapy alone versus in combination with immunosuppression and plasma exchange. KDIGO will assess the currency of its recommendations and the need to update them in the next 3 years.”
The guideline was funded by KDIGO. The investigators disclosed relationships with GSK, Gilead, Intercept, Novo Nordisk, and others. Dr. Rastogi and Dr. Debes had no conflicts of interest.
The Kidney Disease: Improving Global Outcomes (KDIGO) Work Group has updated its guideline concerning the prevention, diagnosis, evaluation, and treatment of hepatitis C virus (HCV) infection in patients with chronic kidney disease (CKD).
Of note, KDIGO now supports transplant of HCV-positive kidneys to HCV-negative recipients.
The guidance document, authored by Ahmed Arslan Yousuf Awan, MD, of Baylor College of Medicine, Houston, and colleagues, was written in light of new evidence that has emerged since the 2018 guideline was published.
“The focused update was triggered by new data on antiviral treatment in patients with advanced stages of CKD (G4, G5, or G5D), transplant of HCV-infected kidneys into uninfected recipients, and evolution of the viewpoint on the role of kidney biopsy in managing kidney disease caused by HCV,” the guideline panelists wrote in Annals of Internal Medicine. “This update is intended to assist clinicians in the care of patients with HCV infection and CKD, including patients receiving dialysis (CKD G5D) and patients with a kidney transplant (CKD G1T-G5T).”
Anjay Rastogi, MD, PhD, professor and clinical chief of nephrology at the David Geffen School of Medicine at UCLA, said the update is both “timely and relevant,” and “will really have an impact on the organ shortage that we have for kidney transplant”
The updates are outlined below.
Expanded Access to HCV-Positive Kidneys
While the 2018 guideline recommended that HCV-positive kidneys be directed to HCV-positive recipients, the new guideline suggests that these kidneys are appropriate for all patients regardless of HCV status.
In support, the panelists cited a follow-up of THINKER-1 trial, which showed that eGFR and quality of life were not negatively affected when HCV-negative patients received an HCV-positive kidney, compared with an HCV-negative kidney. Data from 525 unmatched recipients in 16 other studies support this conclusion, the panelists noted.
Jose Debes, MD, PhD, associate professor at the University of Minnesota, Minneapolis, suggested that this is the most important update to the KDIGO guidelines.
“That [change] would be the main impact of these recommendations,” Dr. Debes said in an interview. “Several centers were already doing this, since some data [were] out there, but I think the fact that they’re making this into a guideline is quite important.”
Dr. Rastogi agreed that this recommendation is the most impactful update.
“That’s a big move,” Dr. Rastogi said in an interview. He predicted that the change will “definitely increase the donor pool, which is very, very important.”
For this new recommendation to have the greatest positive effect, however, Dr. Rastogi suggested that health care providers and treatment centers need to prepare an effective implementation strategy. He emphasized the importance of early communication with patients concerning the safety of HCV-positive kidneys, which depends on early initiation of direct-acting antiviral (DAA) therapy.
In the guideline, Dr. Awan and colleagues reported three documented cases of fibrosing cholestatic hepatitis occurred in patients who did not begin DAA therapy until 30 days after transplant.
“[Patients] should start [DAA treatment] right away,” Dr. Rastogi said, “and sometimes even before the transplant.”
This will require institutional support, he noted, as centers need to ensure that patients are covered for DAA therapy and medication is readily available.
Sofosbuvir Given the Green Light
Compared with the 2018 guideline, which recommended against sofosbuvir in patients with CKD G4 and G5, including those on dialysis, because of concerns about metabolization via the kidneys, the new guideline suggests that sofosbuvir-based DAA regimens are appropriate in patients with glomerular filtration rate (GFR) less than 30 mL/min per 1.73 m2, including those receiving dialysis.
This recommendation was based on a systematic review of 106 studies including both sofosbuvir-based and non-sofosbuvir-based DAA regimens that showed high safety and efficacy for all DAA regimen types across a broad variety of patient types.
“DAAs are highly effective and well tolerated treatments for hepatitis C in patients across all stages of CKD, including those undergoing dialysis and kidney transplant recipients, with no need for dose adjustment,” Dr. Awan and colleagues wrote.
Loosened Biopsy Requirements
Unlike the 2018 guideline, which advised kidney biopsy in HCV-positive patients with clinical evidence of glomerular disease prior to initiating DAA treatment, the new guideline suggests that HCV-infected patients with a typical presentation of immune-complex proliferative glomerulonephritis do not require confirmatory kidney biopsy.
“Because almost all patients with chronic hepatitis C (with or without glomerulonephritis) should be treated with DAAs, a kidney biopsy is unlikely to change management in most patients with hepatitis C and clinical glomerulonephritis,” the panelists wrote.
If kidney disease does not stabilize or improve with achievement of sustained virologic response, or if there is evidence of rapidly progressive glomerulonephritis, then a kidney biopsy should be considered before beginning immunosuppressive therapy, according to the guideline, which includes a flow chart to guide clinicians through this decision-making process.
Individualizing Immunosuppressive Therapy
Consistent with the old guideline, the new guideline recommends DAA treatment with concurrent immunosuppressive therapy for patients with cryoglobulinemic flare or rapidly progressive kidney failure. But in contrast, the new guideline calls for an individualized approach to immunosuppression in patients with nephrotic syndrome.
Dr. Awan and colleagues suggested that “nephrotic-range proteinuria (greater than 3.5 g/d) alone does not warrant use of immunosuppressive treatment because such patients can achieve remission of proteinuria after treatment with DAAs.” Still, if other associated complications — such as anasarca, thromboembolic disease, or severe hypoalbuminemia — are present, then immunosuppressive therapy may be warranted, with rituximab remaining the preferred first-line agent.
More Work Is Needed
Dr. Awan and colleagues concluded the guideline by highlighting areas of unmet need, and how filling these knowledge gaps could lead to additional guideline updates.
“Future studies of kidney donations from HCV-positive donors to HCV-negative recipients are needed to refine and clarify the timing of initiation and duration of DAA therapy and to assess long-term outcomes associated with this practice,” they wrote. “Also, randomized controlled trials are needed to determine which patients with HCV-associated kidney disease can be treated with DAA therapy alone versus in combination with immunosuppression and plasma exchange. KDIGO will assess the currency of its recommendations and the need to update them in the next 3 years.”
The guideline was funded by KDIGO. The investigators disclosed relationships with GSK, Gilead, Intercept, Novo Nordisk, and others. Dr. Rastogi and Dr. Debes had no conflicts of interest.
The Kidney Disease: Improving Global Outcomes (KDIGO) Work Group has updated its guideline concerning the prevention, diagnosis, evaluation, and treatment of hepatitis C virus (HCV) infection in patients with chronic kidney disease (CKD).
Of note, KDIGO now supports transplant of HCV-positive kidneys to HCV-negative recipients.
The guidance document, authored by Ahmed Arslan Yousuf Awan, MD, of Baylor College of Medicine, Houston, and colleagues, was written in light of new evidence that has emerged since the 2018 guideline was published.
“The focused update was triggered by new data on antiviral treatment in patients with advanced stages of CKD (G4, G5, or G5D), transplant of HCV-infected kidneys into uninfected recipients, and evolution of the viewpoint on the role of kidney biopsy in managing kidney disease caused by HCV,” the guideline panelists wrote in Annals of Internal Medicine. “This update is intended to assist clinicians in the care of patients with HCV infection and CKD, including patients receiving dialysis (CKD G5D) and patients with a kidney transplant (CKD G1T-G5T).”
Anjay Rastogi, MD, PhD, professor and clinical chief of nephrology at the David Geffen School of Medicine at UCLA, said the update is both “timely and relevant,” and “will really have an impact on the organ shortage that we have for kidney transplant”
The updates are outlined below.
Expanded Access to HCV-Positive Kidneys
While the 2018 guideline recommended that HCV-positive kidneys be directed to HCV-positive recipients, the new guideline suggests that these kidneys are appropriate for all patients regardless of HCV status.
In support, the panelists cited a follow-up of THINKER-1 trial, which showed that eGFR and quality of life were not negatively affected when HCV-negative patients received an HCV-positive kidney, compared with an HCV-negative kidney. Data from 525 unmatched recipients in 16 other studies support this conclusion, the panelists noted.
Jose Debes, MD, PhD, associate professor at the University of Minnesota, Minneapolis, suggested that this is the most important update to the KDIGO guidelines.
“That [change] would be the main impact of these recommendations,” Dr. Debes said in an interview. “Several centers were already doing this, since some data [were] out there, but I think the fact that they’re making this into a guideline is quite important.”
Dr. Rastogi agreed that this recommendation is the most impactful update.
“That’s a big move,” Dr. Rastogi said in an interview. He predicted that the change will “definitely increase the donor pool, which is very, very important.”
For this new recommendation to have the greatest positive effect, however, Dr. Rastogi suggested that health care providers and treatment centers need to prepare an effective implementation strategy. He emphasized the importance of early communication with patients concerning the safety of HCV-positive kidneys, which depends on early initiation of direct-acting antiviral (DAA) therapy.
In the guideline, Dr. Awan and colleagues reported three documented cases of fibrosing cholestatic hepatitis occurred in patients who did not begin DAA therapy until 30 days after transplant.
“[Patients] should start [DAA treatment] right away,” Dr. Rastogi said, “and sometimes even before the transplant.”
This will require institutional support, he noted, as centers need to ensure that patients are covered for DAA therapy and medication is readily available.
Sofosbuvir Given the Green Light
Compared with the 2018 guideline, which recommended against sofosbuvir in patients with CKD G4 and G5, including those on dialysis, because of concerns about metabolization via the kidneys, the new guideline suggests that sofosbuvir-based DAA regimens are appropriate in patients with glomerular filtration rate (GFR) less than 30 mL/min per 1.73 m2, including those receiving dialysis.
This recommendation was based on a systematic review of 106 studies including both sofosbuvir-based and non-sofosbuvir-based DAA regimens that showed high safety and efficacy for all DAA regimen types across a broad variety of patient types.
“DAAs are highly effective and well tolerated treatments for hepatitis C in patients across all stages of CKD, including those undergoing dialysis and kidney transplant recipients, with no need for dose adjustment,” Dr. Awan and colleagues wrote.
Loosened Biopsy Requirements
Unlike the 2018 guideline, which advised kidney biopsy in HCV-positive patients with clinical evidence of glomerular disease prior to initiating DAA treatment, the new guideline suggests that HCV-infected patients with a typical presentation of immune-complex proliferative glomerulonephritis do not require confirmatory kidney biopsy.
“Because almost all patients with chronic hepatitis C (with or without glomerulonephritis) should be treated with DAAs, a kidney biopsy is unlikely to change management in most patients with hepatitis C and clinical glomerulonephritis,” the panelists wrote.
If kidney disease does not stabilize or improve with achievement of sustained virologic response, or if there is evidence of rapidly progressive glomerulonephritis, then a kidney biopsy should be considered before beginning immunosuppressive therapy, according to the guideline, which includes a flow chart to guide clinicians through this decision-making process.
Individualizing Immunosuppressive Therapy
Consistent with the old guideline, the new guideline recommends DAA treatment with concurrent immunosuppressive therapy for patients with cryoglobulinemic flare or rapidly progressive kidney failure. But in contrast, the new guideline calls for an individualized approach to immunosuppression in patients with nephrotic syndrome.
Dr. Awan and colleagues suggested that “nephrotic-range proteinuria (greater than 3.5 g/d) alone does not warrant use of immunosuppressive treatment because such patients can achieve remission of proteinuria after treatment with DAAs.” Still, if other associated complications — such as anasarca, thromboembolic disease, or severe hypoalbuminemia — are present, then immunosuppressive therapy may be warranted, with rituximab remaining the preferred first-line agent.
More Work Is Needed
Dr. Awan and colleagues concluded the guideline by highlighting areas of unmet need, and how filling these knowledge gaps could lead to additional guideline updates.
“Future studies of kidney donations from HCV-positive donors to HCV-negative recipients are needed to refine and clarify the timing of initiation and duration of DAA therapy and to assess long-term outcomes associated with this practice,” they wrote. “Also, randomized controlled trials are needed to determine which patients with HCV-associated kidney disease can be treated with DAA therapy alone versus in combination with immunosuppression and plasma exchange. KDIGO will assess the currency of its recommendations and the need to update them in the next 3 years.”
The guideline was funded by KDIGO. The investigators disclosed relationships with GSK, Gilead, Intercept, Novo Nordisk, and others. Dr. Rastogi and Dr. Debes had no conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
Anti-Rheumatic Drugs Linked to Reduced Thyroid Disease Incidence
TOPLINE:
Patients with rheumatoid arthritis (RA) in a large Swedish population cohort show a reduced incidence of autoimmune thyroid diseases, such as hypothyroidism or hyperthyroidism, after being diagnosed with RA, with the effect being more pronounced among those treated with disease-modifying anti-rheumatic drugs (DMARDs), particularly TNF-inhibitors.
Although DMARDs are commonly used in the treatment of RA, the drugs are rarely used to treat autoimmune thyroid diseases. The new results support theories raised in previous smaller studies that DMARDs could have a protective effect against thyroid disease.
METHODOLOGY:
- The study involved 13,731 patients with new-onset RA who were listed in the Swedish Rheumatology Quality Register between 2006 and 2018.
- The patients were matched for sex, age, and residential area with up to five reference individuals in the general population of 63,201 comparators.
- Overall, patients with RA were 64.7% female, with a mean age of 59. They were followed up with their matched comparators until the development of autoimmune thyroid disease, death, emigration, or the end of the study period, December 2019.
- The relative risks of autoimmune thyroid disease following a diagnosis of RA and with treatment with DMARDs were compared with those risks in the general population.
- Participants with a non-autoimmune cause for thyroxine prescription were excluded, as were those with an autoimmune thyroid disease at the time of RA diagnosis.
TAKEAWAY:
- Following their RA diagnosis, 321 (2.3%) of patients developed an autoimmune thyroid disease, compared with 1838 (2.9%) in the general population comparators, representing an incidence of 3.7 vs 4.6 per 1000 person-years (hazard ratio [HR], 0.81).
- The lower incidence of autoimmune thyroid disease was more pronounced with longer RA duration. For instance, at 10-14 years after an RA diagnosis, the incidence was 2.9 vs. 4.5 autoimmune thyroid disease events per 1000 person-years, respectively (HR, 0.64).
- The decreased risk of incident autoimmune thyroid disease among RA patients compared with the general population was strongest among patients treated with biologic DMARDs (bDMARD), with an HR of 0.54.
- The reduced incidence of autoimmune thyroid disease with bDMARD use was most pronounced among users of TNF-inhibitors (HR, 0.67).
- The lower incidence of autoimmune thyroid disease following a diagnosis of RA contrasts with previous studies showing an increased risk for thyroid disease associated with RA.
- However, the decreased risk of thyroid disease following bDMARD treatment supports the theory that immunomodulatory treatment could also have an effect of blunting the inflammatory processes that can lead to overt clinical autoimmune thyroid disease.
IN PRACTICE:
“To our knowledge, no previous study has investigated whether the risk of new-onset autoimmune thyroid disease is affected by RA treatment in early RA,” the authors report.
“Our results demonstrate that compared to the general population, patients with RA treated with bDMARDs, TNF-inhibitors in particular, are at decreased risk of developing autoimmune thyroid disease, a finding that calls for replication and may open for drug-repurposing studies,” they note.
SOURCE:
The study was conducted by first author Kristin Waldenlind, PhD, of the Department of Medicine, Solna, Division of Clinical Epidemiology, Karolinska Institutet, Stockholm, Sweden, and colleagues. It was published online November 27 in the Journal of Internal Medicine.
LIMITATIONS:
The study lacked details on participants’ thyroid autoantibody and hormone levels.
The presence of autoimmune thyroid disease was determined based on prescriptions for thyroxine, hence the authors cannot exclude the possibility of a lower threshold for thyroxine prescription among patients treated with DMARDs.
Information was not available on potential risk factors for RA and autoimmune thyroid disease that might have introduced confounding, such as smoking or obesity.
DISCLOSURES:
The study received funding from the Swedish Research Council, the Swedish Heart Lung Foundation, Vinnova, and Region Stockholm/Karolinska Institutet (ALF). The authors’ disclosures are detailed in the published study.
A version of this article appeared on Medscape.com.
TOPLINE:
Patients with rheumatoid arthritis (RA) in a large Swedish population cohort show a reduced incidence of autoimmune thyroid diseases, such as hypothyroidism or hyperthyroidism, after being diagnosed with RA, with the effect being more pronounced among those treated with disease-modifying anti-rheumatic drugs (DMARDs), particularly TNF-inhibitors.
Although DMARDs are commonly used in the treatment of RA, the drugs are rarely used to treat autoimmune thyroid diseases. The new results support theories raised in previous smaller studies that DMARDs could have a protective effect against thyroid disease.
METHODOLOGY:
- The study involved 13,731 patients with new-onset RA who were listed in the Swedish Rheumatology Quality Register between 2006 and 2018.
- The patients were matched for sex, age, and residential area with up to five reference individuals in the general population of 63,201 comparators.
- Overall, patients with RA were 64.7% female, with a mean age of 59. They were followed up with their matched comparators until the development of autoimmune thyroid disease, death, emigration, or the end of the study period, December 2019.
- The relative risks of autoimmune thyroid disease following a diagnosis of RA and with treatment with DMARDs were compared with those risks in the general population.
- Participants with a non-autoimmune cause for thyroxine prescription were excluded, as were those with an autoimmune thyroid disease at the time of RA diagnosis.
TAKEAWAY:
- Following their RA diagnosis, 321 (2.3%) of patients developed an autoimmune thyroid disease, compared with 1838 (2.9%) in the general population comparators, representing an incidence of 3.7 vs 4.6 per 1000 person-years (hazard ratio [HR], 0.81).
- The lower incidence of autoimmune thyroid disease was more pronounced with longer RA duration. For instance, at 10-14 years after an RA diagnosis, the incidence was 2.9 vs. 4.5 autoimmune thyroid disease events per 1000 person-years, respectively (HR, 0.64).
- The decreased risk of incident autoimmune thyroid disease among RA patients compared with the general population was strongest among patients treated with biologic DMARDs (bDMARD), with an HR of 0.54.
- The reduced incidence of autoimmune thyroid disease with bDMARD use was most pronounced among users of TNF-inhibitors (HR, 0.67).
- The lower incidence of autoimmune thyroid disease following a diagnosis of RA contrasts with previous studies showing an increased risk for thyroid disease associated with RA.
- However, the decreased risk of thyroid disease following bDMARD treatment supports the theory that immunomodulatory treatment could also have an effect of blunting the inflammatory processes that can lead to overt clinical autoimmune thyroid disease.
IN PRACTICE:
“To our knowledge, no previous study has investigated whether the risk of new-onset autoimmune thyroid disease is affected by RA treatment in early RA,” the authors report.
“Our results demonstrate that compared to the general population, patients with RA treated with bDMARDs, TNF-inhibitors in particular, are at decreased risk of developing autoimmune thyroid disease, a finding that calls for replication and may open for drug-repurposing studies,” they note.
SOURCE:
The study was conducted by first author Kristin Waldenlind, PhD, of the Department of Medicine, Solna, Division of Clinical Epidemiology, Karolinska Institutet, Stockholm, Sweden, and colleagues. It was published online November 27 in the Journal of Internal Medicine.
LIMITATIONS:
The study lacked details on participants’ thyroid autoantibody and hormone levels.
The presence of autoimmune thyroid disease was determined based on prescriptions for thyroxine, hence the authors cannot exclude the possibility of a lower threshold for thyroxine prescription among patients treated with DMARDs.
Information was not available on potential risk factors for RA and autoimmune thyroid disease that might have introduced confounding, such as smoking or obesity.
DISCLOSURES:
The study received funding from the Swedish Research Council, the Swedish Heart Lung Foundation, Vinnova, and Region Stockholm/Karolinska Institutet (ALF). The authors’ disclosures are detailed in the published study.
A version of this article appeared on Medscape.com.
TOPLINE:
Patients with rheumatoid arthritis (RA) in a large Swedish population cohort show a reduced incidence of autoimmune thyroid diseases, such as hypothyroidism or hyperthyroidism, after being diagnosed with RA, with the effect being more pronounced among those treated with disease-modifying anti-rheumatic drugs (DMARDs), particularly TNF-inhibitors.
Although DMARDs are commonly used in the treatment of RA, the drugs are rarely used to treat autoimmune thyroid diseases. The new results support theories raised in previous smaller studies that DMARDs could have a protective effect against thyroid disease.
METHODOLOGY:
- The study involved 13,731 patients with new-onset RA who were listed in the Swedish Rheumatology Quality Register between 2006 and 2018.
- The patients were matched for sex, age, and residential area with up to five reference individuals in the general population of 63,201 comparators.
- Overall, patients with RA were 64.7% female, with a mean age of 59. They were followed up with their matched comparators until the development of autoimmune thyroid disease, death, emigration, or the end of the study period, December 2019.
- The relative risks of autoimmune thyroid disease following a diagnosis of RA and with treatment with DMARDs were compared with those risks in the general population.
- Participants with a non-autoimmune cause for thyroxine prescription were excluded, as were those with an autoimmune thyroid disease at the time of RA diagnosis.
TAKEAWAY:
- Following their RA diagnosis, 321 (2.3%) of patients developed an autoimmune thyroid disease, compared with 1838 (2.9%) in the general population comparators, representing an incidence of 3.7 vs 4.6 per 1000 person-years (hazard ratio [HR], 0.81).
- The lower incidence of autoimmune thyroid disease was more pronounced with longer RA duration. For instance, at 10-14 years after an RA diagnosis, the incidence was 2.9 vs. 4.5 autoimmune thyroid disease events per 1000 person-years, respectively (HR, 0.64).
- The decreased risk of incident autoimmune thyroid disease among RA patients compared with the general population was strongest among patients treated with biologic DMARDs (bDMARD), with an HR of 0.54.
- The reduced incidence of autoimmune thyroid disease with bDMARD use was most pronounced among users of TNF-inhibitors (HR, 0.67).
- The lower incidence of autoimmune thyroid disease following a diagnosis of RA contrasts with previous studies showing an increased risk for thyroid disease associated with RA.
- However, the decreased risk of thyroid disease following bDMARD treatment supports the theory that immunomodulatory treatment could also have an effect of blunting the inflammatory processes that can lead to overt clinical autoimmune thyroid disease.
IN PRACTICE:
“To our knowledge, no previous study has investigated whether the risk of new-onset autoimmune thyroid disease is affected by RA treatment in early RA,” the authors report.
“Our results demonstrate that compared to the general population, patients with RA treated with bDMARDs, TNF-inhibitors in particular, are at decreased risk of developing autoimmune thyroid disease, a finding that calls for replication and may open for drug-repurposing studies,” they note.
SOURCE:
The study was conducted by first author Kristin Waldenlind, PhD, of the Department of Medicine, Solna, Division of Clinical Epidemiology, Karolinska Institutet, Stockholm, Sweden, and colleagues. It was published online November 27 in the Journal of Internal Medicine.
LIMITATIONS:
The study lacked details on participants’ thyroid autoantibody and hormone levels.
The presence of autoimmune thyroid disease was determined based on prescriptions for thyroxine, hence the authors cannot exclude the possibility of a lower threshold for thyroxine prescription among patients treated with DMARDs.
Information was not available on potential risk factors for RA and autoimmune thyroid disease that might have introduced confounding, such as smoking or obesity.
DISCLOSURES:
The study received funding from the Swedish Research Council, the Swedish Heart Lung Foundation, Vinnova, and Region Stockholm/Karolinska Institutet (ALF). The authors’ disclosures are detailed in the published study.
A version of this article appeared on Medscape.com.
ADA issues new screening, obesity management recommendations
for 2024.
“The Standards of Care are essentially the global guidelines for the care of individuals with diabetes and those at risk,” ADA chief scientific and medical officer Robert Gabbay, MD, PhD, said during a briefing announcing the new Standards.
The document was developed via a scientific literature review by the ADA’s Professional Practice Committee. The panel comprises 21 professionals, including physicians from many specialties, nurse practitioners, certified diabetes care and education specialists, dietitians, and pharmacists. The chair is Nuha A. El Sayed, MD, ADA’s senior vice president of healthcare improvement.
Specific sections of the 2024 document have been endorsed by the American College of Cardiology, the American Society of Bone and Mineral Research, and the Obesity Society. It was published on December 11, 2023, as a supplement in Diabetes Care.
An introductory section summarizing the changes for 2024 spans six pages. Those addressed during the briefing included the following:
Heart Failure Screening: Two new recommendations have been added to include screening of adults with diabetes for asymptomatic heart failure by measuring natriuretic peptide levels to facilitate the prevention or progression to symptomatic stages of heart failure.
“This is a really important and exciting area. We know that people with type 2 diabetes in particular are at high risk for heart failure,” Dr. Gabbay said, adding that these recommendations “are to really more aggressively screen those at high risk for heart failure with a simple blood test and, based on those values, then be able to move on to further evaluation and echocardiography, for example. The recommendations are really to screen a broad number of individuals with type 2 diabetes because many are at risk, [particularly] those without symptoms.”
PAD Screening: A new strong recommendation is to screen for PAD with ankle-brachial index testing in asymptomatic people with diabetes who are aged ≥ 50 years and have microvascular disease in any location, foot complications, or any end-organ damage from diabetes. The document also advises consideration of PAD screening for all individuals who have had diabetes for ≥ 10 years.
Dr. Gabbay commented, “We know that amputation rates are rising, unlike many other complications. We know that there are incredible health disparities. Blacks are two to four times more likely than Whites to have an amputation.”
Dr. El Sayed added, “Many patients don’t show the common symptoms of peripheral arterial disease. Screening is the most important way to find out if they have it or not because it can be a very devastating disease.”
Type 1 Diabetes Screening: This involves several new recommendations, including a framework for investigating suspected type 1 diabetes in newly diagnosed adults using islet autoantibody tests and diagnostic criteria for preclinical stages based on the recent approval of teplizumab for delaying the onset of type 1 diabetes.
“Screening and capturing disease earlier so that we can intervene is really an important consideration here. That includes screening for type 1 diabetes and thinking about therapeutic options to delay the development of frank type 1 diabetes,” Dr. Gabbay said.
Screening first-degree relatives of people with type 1 diabetes is a high priority because they’re at an elevated risk, he added.
Obesity Management: New recommendations here include the use of anthropomorphic measurements beyond body mass index to include waist circumference and waist:hip ratio and individual assessment of body fat mass and distribution.
Individualization of obesity management including behavioral, pharmacologic, and surgical approaches is encouraged. The use of a glucagon-like peptide-1 (GLP-1) receptor agonist or a dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist with greater weight loss efficacy is preferred for obesity management in people with diabetes.
“Obesity management is one of the biggest changes over this last year,” Dr. Gabbay commented.
Other New Recommendations: Among the many other revisions in the 2024 document are new recommendations about regular evaluation and treatment for bone health, assessment of disability and guidance for referral, and alignment of guidance for liver disease screening and management with those of other professional societies. Regarding the last item, Dr. Gabbay noted, “I don’t think it’s gotten the attention it deserves. Diabetes and obesity are becoming the leading causes of liver disease.”
Clinicians can also download the Standards of Care app on their smartphones. “That can be really helpful when questions come up since you can’t remember everything in there. Here you can look it up in a matter of seconds,” Dr. Gabbay said.
Dr. El Sayed added that asking patients about their priorities is also important. “If they aren’t brought up during the visit, it’s unlikely to be as fruitful as it should be.”
Dr. El Sayed has no disclosures. Dr. Gabbay serves as a consultant and/or advisor for HealthReveal, Lark Technologies, Onduo, StartUp Health, Sweetech, and Vida Health.
A version of this article appeared on Medscape.com.
for 2024.
“The Standards of Care are essentially the global guidelines for the care of individuals with diabetes and those at risk,” ADA chief scientific and medical officer Robert Gabbay, MD, PhD, said during a briefing announcing the new Standards.
The document was developed via a scientific literature review by the ADA’s Professional Practice Committee. The panel comprises 21 professionals, including physicians from many specialties, nurse practitioners, certified diabetes care and education specialists, dietitians, and pharmacists. The chair is Nuha A. El Sayed, MD, ADA’s senior vice president of healthcare improvement.
Specific sections of the 2024 document have been endorsed by the American College of Cardiology, the American Society of Bone and Mineral Research, and the Obesity Society. It was published on December 11, 2023, as a supplement in Diabetes Care.
An introductory section summarizing the changes for 2024 spans six pages. Those addressed during the briefing included the following:
Heart Failure Screening: Two new recommendations have been added to include screening of adults with diabetes for asymptomatic heart failure by measuring natriuretic peptide levels to facilitate the prevention or progression to symptomatic stages of heart failure.
“This is a really important and exciting area. We know that people with type 2 diabetes in particular are at high risk for heart failure,” Dr. Gabbay said, adding that these recommendations “are to really more aggressively screen those at high risk for heart failure with a simple blood test and, based on those values, then be able to move on to further evaluation and echocardiography, for example. The recommendations are really to screen a broad number of individuals with type 2 diabetes because many are at risk, [particularly] those without symptoms.”
PAD Screening: A new strong recommendation is to screen for PAD with ankle-brachial index testing in asymptomatic people with diabetes who are aged ≥ 50 years and have microvascular disease in any location, foot complications, or any end-organ damage from diabetes. The document also advises consideration of PAD screening for all individuals who have had diabetes for ≥ 10 years.
Dr. Gabbay commented, “We know that amputation rates are rising, unlike many other complications. We know that there are incredible health disparities. Blacks are two to four times more likely than Whites to have an amputation.”
Dr. El Sayed added, “Many patients don’t show the common symptoms of peripheral arterial disease. Screening is the most important way to find out if they have it or not because it can be a very devastating disease.”
Type 1 Diabetes Screening: This involves several new recommendations, including a framework for investigating suspected type 1 diabetes in newly diagnosed adults using islet autoantibody tests and diagnostic criteria for preclinical stages based on the recent approval of teplizumab for delaying the onset of type 1 diabetes.
“Screening and capturing disease earlier so that we can intervene is really an important consideration here. That includes screening for type 1 diabetes and thinking about therapeutic options to delay the development of frank type 1 diabetes,” Dr. Gabbay said.
Screening first-degree relatives of people with type 1 diabetes is a high priority because they’re at an elevated risk, he added.
Obesity Management: New recommendations here include the use of anthropomorphic measurements beyond body mass index to include waist circumference and waist:hip ratio and individual assessment of body fat mass and distribution.
Individualization of obesity management including behavioral, pharmacologic, and surgical approaches is encouraged. The use of a glucagon-like peptide-1 (GLP-1) receptor agonist or a dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist with greater weight loss efficacy is preferred for obesity management in people with diabetes.
“Obesity management is one of the biggest changes over this last year,” Dr. Gabbay commented.
Other New Recommendations: Among the many other revisions in the 2024 document are new recommendations about regular evaluation and treatment for bone health, assessment of disability and guidance for referral, and alignment of guidance for liver disease screening and management with those of other professional societies. Regarding the last item, Dr. Gabbay noted, “I don’t think it’s gotten the attention it deserves. Diabetes and obesity are becoming the leading causes of liver disease.”
Clinicians can also download the Standards of Care app on their smartphones. “That can be really helpful when questions come up since you can’t remember everything in there. Here you can look it up in a matter of seconds,” Dr. Gabbay said.
Dr. El Sayed added that asking patients about their priorities is also important. “If they aren’t brought up during the visit, it’s unlikely to be as fruitful as it should be.”
Dr. El Sayed has no disclosures. Dr. Gabbay serves as a consultant and/or advisor for HealthReveal, Lark Technologies, Onduo, StartUp Health, Sweetech, and Vida Health.
A version of this article appeared on Medscape.com.
for 2024.
“The Standards of Care are essentially the global guidelines for the care of individuals with diabetes and those at risk,” ADA chief scientific and medical officer Robert Gabbay, MD, PhD, said during a briefing announcing the new Standards.
The document was developed via a scientific literature review by the ADA’s Professional Practice Committee. The panel comprises 21 professionals, including physicians from many specialties, nurse practitioners, certified diabetes care and education specialists, dietitians, and pharmacists. The chair is Nuha A. El Sayed, MD, ADA’s senior vice president of healthcare improvement.
Specific sections of the 2024 document have been endorsed by the American College of Cardiology, the American Society of Bone and Mineral Research, and the Obesity Society. It was published on December 11, 2023, as a supplement in Diabetes Care.
An introductory section summarizing the changes for 2024 spans six pages. Those addressed during the briefing included the following:
Heart Failure Screening: Two new recommendations have been added to include screening of adults with diabetes for asymptomatic heart failure by measuring natriuretic peptide levels to facilitate the prevention or progression to symptomatic stages of heart failure.
“This is a really important and exciting area. We know that people with type 2 diabetes in particular are at high risk for heart failure,” Dr. Gabbay said, adding that these recommendations “are to really more aggressively screen those at high risk for heart failure with a simple blood test and, based on those values, then be able to move on to further evaluation and echocardiography, for example. The recommendations are really to screen a broad number of individuals with type 2 diabetes because many are at risk, [particularly] those without symptoms.”
PAD Screening: A new strong recommendation is to screen for PAD with ankle-brachial index testing in asymptomatic people with diabetes who are aged ≥ 50 years and have microvascular disease in any location, foot complications, or any end-organ damage from diabetes. The document also advises consideration of PAD screening for all individuals who have had diabetes for ≥ 10 years.
Dr. Gabbay commented, “We know that amputation rates are rising, unlike many other complications. We know that there are incredible health disparities. Blacks are two to four times more likely than Whites to have an amputation.”
Dr. El Sayed added, “Many patients don’t show the common symptoms of peripheral arterial disease. Screening is the most important way to find out if they have it or not because it can be a very devastating disease.”
Type 1 Diabetes Screening: This involves several new recommendations, including a framework for investigating suspected type 1 diabetes in newly diagnosed adults using islet autoantibody tests and diagnostic criteria for preclinical stages based on the recent approval of teplizumab for delaying the onset of type 1 diabetes.
“Screening and capturing disease earlier so that we can intervene is really an important consideration here. That includes screening for type 1 diabetes and thinking about therapeutic options to delay the development of frank type 1 diabetes,” Dr. Gabbay said.
Screening first-degree relatives of people with type 1 diabetes is a high priority because they’re at an elevated risk, he added.
Obesity Management: New recommendations here include the use of anthropomorphic measurements beyond body mass index to include waist circumference and waist:hip ratio and individual assessment of body fat mass and distribution.
Individualization of obesity management including behavioral, pharmacologic, and surgical approaches is encouraged. The use of a glucagon-like peptide-1 (GLP-1) receptor agonist or a dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist with greater weight loss efficacy is preferred for obesity management in people with diabetes.
“Obesity management is one of the biggest changes over this last year,” Dr. Gabbay commented.
Other New Recommendations: Among the many other revisions in the 2024 document are new recommendations about regular evaluation and treatment for bone health, assessment of disability and guidance for referral, and alignment of guidance for liver disease screening and management with those of other professional societies. Regarding the last item, Dr. Gabbay noted, “I don’t think it’s gotten the attention it deserves. Diabetes and obesity are becoming the leading causes of liver disease.”
Clinicians can also download the Standards of Care app on their smartphones. “That can be really helpful when questions come up since you can’t remember everything in there. Here you can look it up in a matter of seconds,” Dr. Gabbay said.
Dr. El Sayed added that asking patients about their priorities is also important. “If they aren’t brought up during the visit, it’s unlikely to be as fruitful as it should be.”
Dr. El Sayed has no disclosures. Dr. Gabbay serves as a consultant and/or advisor for HealthReveal, Lark Technologies, Onduo, StartUp Health, Sweetech, and Vida Health.
A version of this article appeared on Medscape.com.
Telemedicine in diabetes care associated with worse outcomes
TOPLINE:
Adult patients with type 2 diabetes and complex care needs receiving endocrinology treatment through telemedicine alone show worse glycemic outcomes compared with those receiving treatment either in-person or in mixed-care models.
The findings contrast with some previous studies showing similar glycemic outcomes with telemedicine care vs in-person care for type 2 diabetes management.
The study is believed to be the first to examine telemedicine care outcomes specifically in the endocrinology setting and based on clinical factors that affect treatment complexity.
METHODOLOGY:
- The retrospective cohort study included 3778 adults with type 2 diabetes in a single, large integrated US health system who had received either telemedicine-only, in-person, or a mix of telemedicine and in-person care between May and October 2020.
- Patients were followed up through May 2022 and evaluated for estimated A1c change after 12 months within each treatment cohort, as well as factors associated with any changes.
- Of the patients, 1182 received telemedicine-only, 1049 received in-person, and 1547 received mixed care. Mean ages in the groups ranged from 57 to 63 years, and women made up between 55% and 63%.
TAKEAWAY:
- Over the 12-month evaluation period, patients receiving telemedicine-only care had no significant changes or improvements in adjusted A1c (−0.06; P = .55), those receiving in-person care had an improvement of 0.37% (P < .001), and those receiving mixed care had an improvement of 0.22% (P = .004).
- The glycemic outcome patterns were similar among patients with a baseline A1c of 8% or higher.
- Of those prescribed multiple daily injections vs no insulin, estimated changes in A1c were 0.25% higher for those receiving telemedicine than for those receiving in-person care (P = .03).
- No associations were observed between changes in A1c and comorbidities.
- Regarding reasons for the differences, the authors noted that “the strategies to support glycemic improvement that are available during in-person appointments have not consistently been translated to telemedicine care.”
- Essential components of telemedicine such as self-management education support may not currently be routinely available through telemedicine or at the point-of-care during telemedicine visits, they added.
- “In our prior work in this care setting, practitioners described how inferior availability of glucose data limited their ability to intensify treatment through telemedicine.”
- “Implementation of approaches to overcome these differences, such as team-based virtual care and technological tools to automate blood glucose data sharing, are needed to ensure all patients receive high-quality diabetes care regardless of care modality.”
IN PRACTICE:
“These findings suggest that patients with type 2 diabetes who rely on telemedicine alone to access endocrinology care may require additional support to achieve glycemic goals,” the authors reported.
“Since some patients with barriers to in-person endocrinology care will continue to rely on telemedicine to access care, structured approaches to ensure routine delivery of high-quality team-based diabetes care are needed,” they asserted.
“Translation of successful strategies from clinical trials into routine telemedicine care, especially targeted toward adults with more complex diabetes, is critical to improve clinical outcomes for patients who rely on this care modality.”
SOURCE:
The study was conducted by first author Margaret F. Zupa, MD, of the division of endocrinology and metabolism, University of Pittsburgh School of Medicine, Pennsylvania, and colleagues.
It was published in JAMA Network Open.
LIMITATIONS:
While demographic differences between the groups were included as covariates, the treatment modality cohorts were not balanced based on baseline characteristics that could be confounders.
Various factors, such as treatment complexity, glycemic control, and transportation barriers, could have affected whether patients received care with telemedicine; therefore, causal associations could not be established.
DISCLOSURES:
The study received funding from the National Center for Advancing Translational Sciences, National Institutes of Health, Pittsburgh Foundation, and Fraternal Order of the Eagles Charity Foundation Diabetes Fund. The authors’ disclosures are detailed in the study.
A version of this article appeared on Medscape.com.
TOPLINE:
Adult patients with type 2 diabetes and complex care needs receiving endocrinology treatment through telemedicine alone show worse glycemic outcomes compared with those receiving treatment either in-person or in mixed-care models.
The findings contrast with some previous studies showing similar glycemic outcomes with telemedicine care vs in-person care for type 2 diabetes management.
The study is believed to be the first to examine telemedicine care outcomes specifically in the endocrinology setting and based on clinical factors that affect treatment complexity.
METHODOLOGY:
- The retrospective cohort study included 3778 adults with type 2 diabetes in a single, large integrated US health system who had received either telemedicine-only, in-person, or a mix of telemedicine and in-person care between May and October 2020.
- Patients were followed up through May 2022 and evaluated for estimated A1c change after 12 months within each treatment cohort, as well as factors associated with any changes.
- Of the patients, 1182 received telemedicine-only, 1049 received in-person, and 1547 received mixed care. Mean ages in the groups ranged from 57 to 63 years, and women made up between 55% and 63%.
TAKEAWAY:
- Over the 12-month evaluation period, patients receiving telemedicine-only care had no significant changes or improvements in adjusted A1c (−0.06; P = .55), those receiving in-person care had an improvement of 0.37% (P < .001), and those receiving mixed care had an improvement of 0.22% (P = .004).
- The glycemic outcome patterns were similar among patients with a baseline A1c of 8% or higher.
- Of those prescribed multiple daily injections vs no insulin, estimated changes in A1c were 0.25% higher for those receiving telemedicine than for those receiving in-person care (P = .03).
- No associations were observed between changes in A1c and comorbidities.
- Regarding reasons for the differences, the authors noted that “the strategies to support glycemic improvement that are available during in-person appointments have not consistently been translated to telemedicine care.”
- Essential components of telemedicine such as self-management education support may not currently be routinely available through telemedicine or at the point-of-care during telemedicine visits, they added.
- “In our prior work in this care setting, practitioners described how inferior availability of glucose data limited their ability to intensify treatment through telemedicine.”
- “Implementation of approaches to overcome these differences, such as team-based virtual care and technological tools to automate blood glucose data sharing, are needed to ensure all patients receive high-quality diabetes care regardless of care modality.”
IN PRACTICE:
“These findings suggest that patients with type 2 diabetes who rely on telemedicine alone to access endocrinology care may require additional support to achieve glycemic goals,” the authors reported.
“Since some patients with barriers to in-person endocrinology care will continue to rely on telemedicine to access care, structured approaches to ensure routine delivery of high-quality team-based diabetes care are needed,” they asserted.
“Translation of successful strategies from clinical trials into routine telemedicine care, especially targeted toward adults with more complex diabetes, is critical to improve clinical outcomes for patients who rely on this care modality.”
SOURCE:
The study was conducted by first author Margaret F. Zupa, MD, of the division of endocrinology and metabolism, University of Pittsburgh School of Medicine, Pennsylvania, and colleagues.
It was published in JAMA Network Open.
LIMITATIONS:
While demographic differences between the groups were included as covariates, the treatment modality cohorts were not balanced based on baseline characteristics that could be confounders.
Various factors, such as treatment complexity, glycemic control, and transportation barriers, could have affected whether patients received care with telemedicine; therefore, causal associations could not be established.
DISCLOSURES:
The study received funding from the National Center for Advancing Translational Sciences, National Institutes of Health, Pittsburgh Foundation, and Fraternal Order of the Eagles Charity Foundation Diabetes Fund. The authors’ disclosures are detailed in the study.
A version of this article appeared on Medscape.com.
TOPLINE:
Adult patients with type 2 diabetes and complex care needs receiving endocrinology treatment through telemedicine alone show worse glycemic outcomes compared with those receiving treatment either in-person or in mixed-care models.
The findings contrast with some previous studies showing similar glycemic outcomes with telemedicine care vs in-person care for type 2 diabetes management.
The study is believed to be the first to examine telemedicine care outcomes specifically in the endocrinology setting and based on clinical factors that affect treatment complexity.
METHODOLOGY:
- The retrospective cohort study included 3778 adults with type 2 diabetes in a single, large integrated US health system who had received either telemedicine-only, in-person, or a mix of telemedicine and in-person care between May and October 2020.
- Patients were followed up through May 2022 and evaluated for estimated A1c change after 12 months within each treatment cohort, as well as factors associated with any changes.
- Of the patients, 1182 received telemedicine-only, 1049 received in-person, and 1547 received mixed care. Mean ages in the groups ranged from 57 to 63 years, and women made up between 55% and 63%.
TAKEAWAY:
- Over the 12-month evaluation period, patients receiving telemedicine-only care had no significant changes or improvements in adjusted A1c (−0.06; P = .55), those receiving in-person care had an improvement of 0.37% (P < .001), and those receiving mixed care had an improvement of 0.22% (P = .004).
- The glycemic outcome patterns were similar among patients with a baseline A1c of 8% or higher.
- Of those prescribed multiple daily injections vs no insulin, estimated changes in A1c were 0.25% higher for those receiving telemedicine than for those receiving in-person care (P = .03).
- No associations were observed between changes in A1c and comorbidities.
- Regarding reasons for the differences, the authors noted that “the strategies to support glycemic improvement that are available during in-person appointments have not consistently been translated to telemedicine care.”
- Essential components of telemedicine such as self-management education support may not currently be routinely available through telemedicine or at the point-of-care during telemedicine visits, they added.
- “In our prior work in this care setting, practitioners described how inferior availability of glucose data limited their ability to intensify treatment through telemedicine.”
- “Implementation of approaches to overcome these differences, such as team-based virtual care and technological tools to automate blood glucose data sharing, are needed to ensure all patients receive high-quality diabetes care regardless of care modality.”
IN PRACTICE:
“These findings suggest that patients with type 2 diabetes who rely on telemedicine alone to access endocrinology care may require additional support to achieve glycemic goals,” the authors reported.
“Since some patients with barriers to in-person endocrinology care will continue to rely on telemedicine to access care, structured approaches to ensure routine delivery of high-quality team-based diabetes care are needed,” they asserted.
“Translation of successful strategies from clinical trials into routine telemedicine care, especially targeted toward adults with more complex diabetes, is critical to improve clinical outcomes for patients who rely on this care modality.”
SOURCE:
The study was conducted by first author Margaret F. Zupa, MD, of the division of endocrinology and metabolism, University of Pittsburgh School of Medicine, Pennsylvania, and colleagues.
It was published in JAMA Network Open.
LIMITATIONS:
While demographic differences between the groups were included as covariates, the treatment modality cohorts were not balanced based on baseline characteristics that could be confounders.
Various factors, such as treatment complexity, glycemic control, and transportation barriers, could have affected whether patients received care with telemedicine; therefore, causal associations could not be established.
DISCLOSURES:
The study received funding from the National Center for Advancing Translational Sciences, National Institutes of Health, Pittsburgh Foundation, and Fraternal Order of the Eagles Charity Foundation Diabetes Fund. The authors’ disclosures are detailed in the study.
A version of this article appeared on Medscape.com.


