User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


Medicare study evaluates impact of U.S. Hospital Readmissions Reduction Program
Research offers evidence against calls to curtail the program
Among Medicare beneficiaries admitted to the hospital between 2008 and 2016, there was an increase in postdischarge 30-day mortality for patients with heart failure, but not for those with acute myocardial infarction or pneumonia.
The finding comes from an effort to evaluate the use of services soon after discharge for conditions targeted in the U.S. Hospital Readmissions Reduction Program (HRRP), and patients’ outcomes.
“The announcement and implementation of the HRRP were associated with a reduction in readmissions within 30 days of discharge for heart failure, acute myocardial infarction, and pneumonia, as shown by a decrease in the overall national rate of readmissions,” first author Rohan Khera, MD, and colleagues wrote in a study published online Jan. 15, 2020, in the British Medical Journal (doi:10.1136/bmj.l6831).
“Concerns existed that pressures to reduce readmissions had led to the evolution of care patterns that may have adverse consequences through reducing access to care in appropriate settings. Therefore, determining whether patients who are seen in acute care settings, but not admitted to hospital, experience an increased risk of mortality is essential.”
Dr. Khera, a cardiologist at the University of Texas Southwestern Medical Center, Dallas, and colleagues limited the analysis to Medicare claims data from patients who were admitted to the hospital with heart failure, acute myocardial infarction (MI), or pneumonia between 2008 and 2016. Key outcomes of interest were: (1) postdischarge 30-day mortality; and (2) acute care utilization in inpatient units, observation units, and the ED during the postdischarge period.
During the study period there were 3,772,924 hospital admissions for heart failure, 1,570,113 for acute MI, and 3,131,162 for pneumonia. The greatest number of readmissions within 30 days of discharge was for heart failure patients (22.5%), followed by acute MI (17.5%), and pneumonia (17.2%).
The overall rates of observation stays were 1.7% for heart failure, 2.6% for acute MI, and 1.4% for pneumonia, while the overall rates of emergency department visits were 6.4% for heart failure, 6.8% for acute MI, and 6.3% for pneumonia. Cumulatively, about one-third of all admissions – 30.7% for heart failure, 26.9% for acute MI, and 24.8% for pneumonia – received postdischarge care in any acute care setting.
Dr. Khera and colleagues found that overall postdischarge 30-day mortality was 8.7% for heart failure, 7.3% for acute MI, and 8.4% for pneumonia. At the same time, postdischarge 30-day mortality was higher in patients with readmissions (13.2% for heart failure, 12.7% for acute MI, and 15.3% for pneumonia), compared with those who had observation stays (4.5% for heart failure, 2.7% for acute MI, and 4.6% for pneumonia), emergency department visits (9.7% for heart failure, 8.8% for acute MI, and 7.8% for pneumonia), or no postdischarge acute care (7.2% for heart failure, 6.0% for acute MI, and 6.9% for pneumonia). Risk adjusted mortality increased annually by 0.05% only for heart failure, while it decreased by 0.06% for acute MI, and did not significantly change for pneumonia.
“The study strongly suggests that the HRRP did not lead to harm through inappropriate triage of patients at high risk to observation units and the emergency department, and therefore provides evidence against calls to curtail the program owing to this theoretical concern (see JAMA 2018;320:2539-41),” the researchers concluded.
They acknowledged certain limitations of the study, including the fact that they were “unable to identify patterns of acute care during the index hospital admission that would be associated with a higher rate of postdischarge acute care in observation units and emergency departments and whether these visits represented avenues for planned postdischarge follow-up care. Moreover, the proportion of these care encounters that were preventable remains poorly understood.”
Dr. Khera disclosed that he is supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. His coauthors reported having numerous disclosures.
SOURCE: Khera et al. BMJ 2020;368:l6831.
Research offers evidence against calls to curtail the program
Research offers evidence against calls to curtail the program
Among Medicare beneficiaries admitted to the hospital between 2008 and 2016, there was an increase in postdischarge 30-day mortality for patients with heart failure, but not for those with acute myocardial infarction or pneumonia.
The finding comes from an effort to evaluate the use of services soon after discharge for conditions targeted in the U.S. Hospital Readmissions Reduction Program (HRRP), and patients’ outcomes.
“The announcement and implementation of the HRRP were associated with a reduction in readmissions within 30 days of discharge for heart failure, acute myocardial infarction, and pneumonia, as shown by a decrease in the overall national rate of readmissions,” first author Rohan Khera, MD, and colleagues wrote in a study published online Jan. 15, 2020, in the British Medical Journal (doi:10.1136/bmj.l6831).
“Concerns existed that pressures to reduce readmissions had led to the evolution of care patterns that may have adverse consequences through reducing access to care in appropriate settings. Therefore, determining whether patients who are seen in acute care settings, but not admitted to hospital, experience an increased risk of mortality is essential.”
Dr. Khera, a cardiologist at the University of Texas Southwestern Medical Center, Dallas, and colleagues limited the analysis to Medicare claims data from patients who were admitted to the hospital with heart failure, acute myocardial infarction (MI), or pneumonia between 2008 and 2016. Key outcomes of interest were: (1) postdischarge 30-day mortality; and (2) acute care utilization in inpatient units, observation units, and the ED during the postdischarge period.
During the study period there were 3,772,924 hospital admissions for heart failure, 1,570,113 for acute MI, and 3,131,162 for pneumonia. The greatest number of readmissions within 30 days of discharge was for heart failure patients (22.5%), followed by acute MI (17.5%), and pneumonia (17.2%).
The overall rates of observation stays were 1.7% for heart failure, 2.6% for acute MI, and 1.4% for pneumonia, while the overall rates of emergency department visits were 6.4% for heart failure, 6.8% for acute MI, and 6.3% for pneumonia. Cumulatively, about one-third of all admissions – 30.7% for heart failure, 26.9% for acute MI, and 24.8% for pneumonia – received postdischarge care in any acute care setting.
Dr. Khera and colleagues found that overall postdischarge 30-day mortality was 8.7% for heart failure, 7.3% for acute MI, and 8.4% for pneumonia. At the same time, postdischarge 30-day mortality was higher in patients with readmissions (13.2% for heart failure, 12.7% for acute MI, and 15.3% for pneumonia), compared with those who had observation stays (4.5% for heart failure, 2.7% for acute MI, and 4.6% for pneumonia), emergency department visits (9.7% for heart failure, 8.8% for acute MI, and 7.8% for pneumonia), or no postdischarge acute care (7.2% for heart failure, 6.0% for acute MI, and 6.9% for pneumonia). Risk adjusted mortality increased annually by 0.05% only for heart failure, while it decreased by 0.06% for acute MI, and did not significantly change for pneumonia.
“The study strongly suggests that the HRRP did not lead to harm through inappropriate triage of patients at high risk to observation units and the emergency department, and therefore provides evidence against calls to curtail the program owing to this theoretical concern (see JAMA 2018;320:2539-41),” the researchers concluded.
They acknowledged certain limitations of the study, including the fact that they were “unable to identify patterns of acute care during the index hospital admission that would be associated with a higher rate of postdischarge acute care in observation units and emergency departments and whether these visits represented avenues for planned postdischarge follow-up care. Moreover, the proportion of these care encounters that were preventable remains poorly understood.”
Dr. Khera disclosed that he is supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. His coauthors reported having numerous disclosures.
SOURCE: Khera et al. BMJ 2020;368:l6831.
Among Medicare beneficiaries admitted to the hospital between 2008 and 2016, there was an increase in postdischarge 30-day mortality for patients with heart failure, but not for those with acute myocardial infarction or pneumonia.
The finding comes from an effort to evaluate the use of services soon after discharge for conditions targeted in the U.S. Hospital Readmissions Reduction Program (HRRP), and patients’ outcomes.
“The announcement and implementation of the HRRP were associated with a reduction in readmissions within 30 days of discharge for heart failure, acute myocardial infarction, and pneumonia, as shown by a decrease in the overall national rate of readmissions,” first author Rohan Khera, MD, and colleagues wrote in a study published online Jan. 15, 2020, in the British Medical Journal (doi:10.1136/bmj.l6831).
“Concerns existed that pressures to reduce readmissions had led to the evolution of care patterns that may have adverse consequences through reducing access to care in appropriate settings. Therefore, determining whether patients who are seen in acute care settings, but not admitted to hospital, experience an increased risk of mortality is essential.”
Dr. Khera, a cardiologist at the University of Texas Southwestern Medical Center, Dallas, and colleagues limited the analysis to Medicare claims data from patients who were admitted to the hospital with heart failure, acute myocardial infarction (MI), or pneumonia between 2008 and 2016. Key outcomes of interest were: (1) postdischarge 30-day mortality; and (2) acute care utilization in inpatient units, observation units, and the ED during the postdischarge period.
During the study period there were 3,772,924 hospital admissions for heart failure, 1,570,113 for acute MI, and 3,131,162 for pneumonia. The greatest number of readmissions within 30 days of discharge was for heart failure patients (22.5%), followed by acute MI (17.5%), and pneumonia (17.2%).
The overall rates of observation stays were 1.7% for heart failure, 2.6% for acute MI, and 1.4% for pneumonia, while the overall rates of emergency department visits were 6.4% for heart failure, 6.8% for acute MI, and 6.3% for pneumonia. Cumulatively, about one-third of all admissions – 30.7% for heart failure, 26.9% for acute MI, and 24.8% for pneumonia – received postdischarge care in any acute care setting.
Dr. Khera and colleagues found that overall postdischarge 30-day mortality was 8.7% for heart failure, 7.3% for acute MI, and 8.4% for pneumonia. At the same time, postdischarge 30-day mortality was higher in patients with readmissions (13.2% for heart failure, 12.7% for acute MI, and 15.3% for pneumonia), compared with those who had observation stays (4.5% for heart failure, 2.7% for acute MI, and 4.6% for pneumonia), emergency department visits (9.7% for heart failure, 8.8% for acute MI, and 7.8% for pneumonia), or no postdischarge acute care (7.2% for heart failure, 6.0% for acute MI, and 6.9% for pneumonia). Risk adjusted mortality increased annually by 0.05% only for heart failure, while it decreased by 0.06% for acute MI, and did not significantly change for pneumonia.
“The study strongly suggests that the HRRP did not lead to harm through inappropriate triage of patients at high risk to observation units and the emergency department, and therefore provides evidence against calls to curtail the program owing to this theoretical concern (see JAMA 2018;320:2539-41),” the researchers concluded.
They acknowledged certain limitations of the study, including the fact that they were “unable to identify patterns of acute care during the index hospital admission that would be associated with a higher rate of postdischarge acute care in observation units and emergency departments and whether these visits represented avenues for planned postdischarge follow-up care. Moreover, the proportion of these care encounters that were preventable remains poorly understood.”
Dr. Khera disclosed that he is supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. His coauthors reported having numerous disclosures.
SOURCE: Khera et al. BMJ 2020;368:l6831.
FROM BMJ
Flu activity increases for third straight week
For the second time during the 2019-2020 flu season, activity measures have climbed into noteworthy territory.
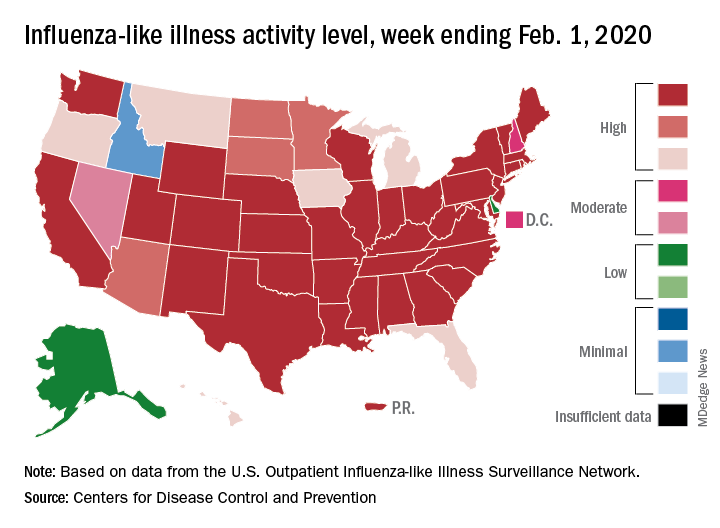
The proportion of outpatient visits for influenza-like illness (ILI) reached its highest December level, 7.1%, since 2003 and then dropped for 2 weeks. Three weeks of increases since then, however, have the outpatient-visit rate at 6.7% for the week ending Feb. 1, 2020, the Centers for Disease Control and Prevention reported. The baseline rate for the United States is 2.4%.
That rate of 6.7% is already above the highest rates recorded in eight of the last nine flu seasons, and another increase could mean a second, separate trip above 7.0% in the 2019-2020 season – something that has not occurred since national tracking began in 1997, CDC data show.
Those same data also show that,
Another important measure on the rise, the proportion of respiratory specimens testing positive for influenza, reached a new high for the season, 29.8%, during the week of Feb. 1, the CDC’s influenza division said.
Tests at clinical laboratories also show that predominance is continuing to switch from type B (45.6%) to type A (54.4%), the influenza division noted. Overall predominance for the season, however, continues to favor type B, 59.3% to 40.7%.
The percentage of deaths caused by pneumonia and influenza, which passed the threshold for epidemic of 7.2% back in early January, has been trending downward for the last 3 weeks and was 7.1% as of Feb. 1, according to the influenza division.
ILI-related deaths among children continue to remain high, with a total count of 78 for the season after another 10 deaths were reported during the week ending Feb. 1, the CDC reported. Comparable numbers for the last three seasons are 44 (2018-2019), 97 (2017-2018), and 35 (2016-2017).
The CDC estimates put the total number of ILIs at around 22 million for the season so far, leading to 210,000 hospitalizations. The agency said that it expects to release estimates of vaccine effectiveness later this month.
For the second time during the 2019-2020 flu season, activity measures have climbed into noteworthy territory.

The proportion of outpatient visits for influenza-like illness (ILI) reached its highest December level, 7.1%, since 2003 and then dropped for 2 weeks. Three weeks of increases since then, however, have the outpatient-visit rate at 6.7% for the week ending Feb. 1, 2020, the Centers for Disease Control and Prevention reported. The baseline rate for the United States is 2.4%.
That rate of 6.7% is already above the highest rates recorded in eight of the last nine flu seasons, and another increase could mean a second, separate trip above 7.0% in the 2019-2020 season – something that has not occurred since national tracking began in 1997, CDC data show.
Those same data also show that,
Another important measure on the rise, the proportion of respiratory specimens testing positive for influenza, reached a new high for the season, 29.8%, during the week of Feb. 1, the CDC’s influenza division said.
Tests at clinical laboratories also show that predominance is continuing to switch from type B (45.6%) to type A (54.4%), the influenza division noted. Overall predominance for the season, however, continues to favor type B, 59.3% to 40.7%.
The percentage of deaths caused by pneumonia and influenza, which passed the threshold for epidemic of 7.2% back in early January, has been trending downward for the last 3 weeks and was 7.1% as of Feb. 1, according to the influenza division.
ILI-related deaths among children continue to remain high, with a total count of 78 for the season after another 10 deaths were reported during the week ending Feb. 1, the CDC reported. Comparable numbers for the last three seasons are 44 (2018-2019), 97 (2017-2018), and 35 (2016-2017).
The CDC estimates put the total number of ILIs at around 22 million for the season so far, leading to 210,000 hospitalizations. The agency said that it expects to release estimates of vaccine effectiveness later this month.
For the second time during the 2019-2020 flu season, activity measures have climbed into noteworthy territory.

The proportion of outpatient visits for influenza-like illness (ILI) reached its highest December level, 7.1%, since 2003 and then dropped for 2 weeks. Three weeks of increases since then, however, have the outpatient-visit rate at 6.7% for the week ending Feb. 1, 2020, the Centers for Disease Control and Prevention reported. The baseline rate for the United States is 2.4%.
That rate of 6.7% is already above the highest rates recorded in eight of the last nine flu seasons, and another increase could mean a second, separate trip above 7.0% in the 2019-2020 season – something that has not occurred since national tracking began in 1997, CDC data show.
Those same data also show that,
Another important measure on the rise, the proportion of respiratory specimens testing positive for influenza, reached a new high for the season, 29.8%, during the week of Feb. 1, the CDC’s influenza division said.
Tests at clinical laboratories also show that predominance is continuing to switch from type B (45.6%) to type A (54.4%), the influenza division noted. Overall predominance for the season, however, continues to favor type B, 59.3% to 40.7%.
The percentage of deaths caused by pneumonia and influenza, which passed the threshold for epidemic of 7.2% back in early January, has been trending downward for the last 3 weeks and was 7.1% as of Feb. 1, according to the influenza division.
ILI-related deaths among children continue to remain high, with a total count of 78 for the season after another 10 deaths were reported during the week ending Feb. 1, the CDC reported. Comparable numbers for the last three seasons are 44 (2018-2019), 97 (2017-2018), and 35 (2016-2017).
The CDC estimates put the total number of ILIs at around 22 million for the season so far, leading to 210,000 hospitalizations. The agency said that it expects to release estimates of vaccine effectiveness later this month.
Myth busting: Sudden cardiac death in athletes
SNOWMASS, COLO. – Myths and misconceptions abound regarding the merits of universal incorporation of the resting 12-lead ECG into preparticipation cardiovascular screening of young athletes, Aaron L. Baggish, MD, declared at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
Dr. Baggish, director of the Cardiovascular Performance Program at Massachusetts General Hospital and a cardiologist at Harvard Medical School, Boston, set out to pop the balloons of a handful of these widely floating myths. These are commonly held fictions: In an electronic poll at the outset of his talk, only one in five members of his large audience recognized all of the following boldface statements as false.
“Preparticipation cardiovascular screening (PPCVS) has been shown to reduce the incidence of sudden cardiac death (SCD) among young competitive athletes.”
FALSE. Not for PPCVS by history and physical examination alone, or with the addition of a screening 12-lead ECG. In Italy, where a cluster of high-profile sudden cardiac deaths led to passage of a 1982 national law mandating 12-lead ECG screening as part of the PPCVS, investigators presented studies purporting to demonstrate a subsequent reduction in the risk of SCD. But those studies were subsequently shown to be fraught with problems. And a high-quality study capable of convincingly demonstrating such a benefit would need to be prohibitively large and expensive. “Don’t hold your breath waiting for that to happen anytime soon,” advised Dr. Baggish, who is medical director for the Boston Marathon, as well as team cardiologist for Harvard University Athletics, the New England Patriots, the Boston Bruins, USRowing, and U.S. Soccer.
“Hypertrophic cardiomyopathy is the leading cause of sudden death among young competitive athletes.”
FALSE. A study of the National Collegiate Athletic Association (NCAA) comprehensive database, with 4.2 million athlete-years of follow-up, showed that the most common cause of SCD was autopsy-negative sudden unexplained death (SUD), accounting for 25% of cases. Hypertrophic cardiomyopathy was deemed the cause of 8% of the SCDs (Circulation. 2015 Jul 7;132[1]:10-9).
“The same thing has been shown in studies done in the United Kingdom and in Australia: . Over the next 10 years, I suspect that one of the most important areas that we’ll be looking into will be this SUD area, perhaps using molecular autopsy to make some headway there,” according to the cardiologist.
SCD is rare. In the NCAA study, the incidence was 1 in 53,703 athlete-years. In sobering contrast, accidents, suicide, and homicide accounted for 50% of all deaths in the collegiate athletes.
“When you think about what’s important in terms of educating young people to be safe, the history and physical exam and 12-lead ECG are nowhere near as important as talking with them about minimizing accident risk and staying away from guns,” Dr. Baggish commented.
“Contemporary ECG interpretation criteria designed specifically for use in young athletes have eliminated the problem of false-positive testing.”
FALSE. The story of adding ECG screening to the PPCVS is one of dramatically improved sensitivity over history and physical exam alone, but always at the cost of reduced specificity. In the Harvard Athlete Initiative Study, Dr. Baggish and coworkers reported that adding the 12-lead ECG resulted in a 17% false-positive rate (Ann Intern Med. 2010 Mar 2;152[5]:269-75). Similar findings were reported in independent studies at two other large universities.
“An ECG false-positive rate of 16%-20%? That’s big trouble. Remember, the conditions we’re looking for are uncommon, with a prevalence of maybe 1 in 500 at most. So if you’re flagging one-fifth or one-sixth of your athletes, the ECG is really not an appropriate tool for screening,” he commented.
Recognition of this limitation has led to development of refined, improved ECG criteria: most notably, the 2012 Seattle criteria, with an associated false-positive rate of 4%-8%, followed by the 2017 International Consensus Criteria (J Am Coll Cardiol. 2017 Feb 28;69[8]:1057-75), with a false-positive rate of 1%-2%. That’s a great improvement. Still, when Dr. Baggish, a marathoner himself, thinks about the roughly 32,000 Boston Marathon runners at the starting line each year, that false-positive rate would translate into 320-640 of those individuals being needlessly subjected to the not-insignificant time and expense of further testing, along with considerable anxiety for the runners and their families, and perhaps even inappropriate disqualification.
“Current ACC/AHA guidelines recommend against the use of the 12-lead ECG during the PPCVS.”
FALSE. Dr. Baggish was a coauthor of the current guidelines, which he described as “an open-door invitation to local decisions, with some important caveats” (Circulation. 2015 Dec 1;132[22]:e267-72).
The guidelines state that the minimum requirement and legal standard for PPCVS of young competitive athletes is a focused history and physical examination, such as the American College of Cardiology/American Heart Association 14-point screen, which consists of 10 elements addressing personal and family history and 4 focused on the physical examination, or the American Academy of Pediatrics Preparticipation Physical Evaluation. Further, while mandatory universal inclusion of the 12-lead ECG is not recommended – it’s rated Class III, meaning don’t do it – the guidelines state that screening programs are at liberty to choose the 12-lead ECG as an additional tool, “provided that close physician involvement and sufficient quality control can be achieved. If undertaken, such initiatives should recognize the known and anticipated limitations of the 12-lead ECG as a population screening test, including the expected frequency of false-positive and false-negative test results, as well as the cost required to support these initiatives over time.”
Dr. Baggish considers the ACC/AHA guidelines to be one of the two most important developments in the field of SCD during sports in recent years. The other is the NCAA-sponsored multidisciplinary Interassociation Consensus Statement on Cardiovascular Care of College Student-Athletes, which he also coauthored (J Am Coll Cardiol. 2016 Jun 28;67[25]:2981-95).
The report lays out the case for a much broader than traditional view of the PPCVS, with “goals that extend beyond detection of occult high-risk pathology.”
“The NCAA has done something very interesting,” Dr. Baggish explained. “It has said that, if we’re going to be screening, we should be thinking about screening with a much broader rationale. It’s not just about finding the needle-in-a-haystack hypertrophic cardiomyopathy or anomalous coronary arteries, it’s about engaging student-athletes at an early point in their collegiate career and trying to improve their health overall – and not just while they’re in college, but over their lifespan.”
He reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – Myths and misconceptions abound regarding the merits of universal incorporation of the resting 12-lead ECG into preparticipation cardiovascular screening of young athletes, Aaron L. Baggish, MD, declared at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
Dr. Baggish, director of the Cardiovascular Performance Program at Massachusetts General Hospital and a cardiologist at Harvard Medical School, Boston, set out to pop the balloons of a handful of these widely floating myths. These are commonly held fictions: In an electronic poll at the outset of his talk, only one in five members of his large audience recognized all of the following boldface statements as false.
“Preparticipation cardiovascular screening (PPCVS) has been shown to reduce the incidence of sudden cardiac death (SCD) among young competitive athletes.”
FALSE. Not for PPCVS by history and physical examination alone, or with the addition of a screening 12-lead ECG. In Italy, where a cluster of high-profile sudden cardiac deaths led to passage of a 1982 national law mandating 12-lead ECG screening as part of the PPCVS, investigators presented studies purporting to demonstrate a subsequent reduction in the risk of SCD. But those studies were subsequently shown to be fraught with problems. And a high-quality study capable of convincingly demonstrating such a benefit would need to be prohibitively large and expensive. “Don’t hold your breath waiting for that to happen anytime soon,” advised Dr. Baggish, who is medical director for the Boston Marathon, as well as team cardiologist for Harvard University Athletics, the New England Patriots, the Boston Bruins, USRowing, and U.S. Soccer.
“Hypertrophic cardiomyopathy is the leading cause of sudden death among young competitive athletes.”
FALSE. A study of the National Collegiate Athletic Association (NCAA) comprehensive database, with 4.2 million athlete-years of follow-up, showed that the most common cause of SCD was autopsy-negative sudden unexplained death (SUD), accounting for 25% of cases. Hypertrophic cardiomyopathy was deemed the cause of 8% of the SCDs (Circulation. 2015 Jul 7;132[1]:10-9).
“The same thing has been shown in studies done in the United Kingdom and in Australia: . Over the next 10 years, I suspect that one of the most important areas that we’ll be looking into will be this SUD area, perhaps using molecular autopsy to make some headway there,” according to the cardiologist.
SCD is rare. In the NCAA study, the incidence was 1 in 53,703 athlete-years. In sobering contrast, accidents, suicide, and homicide accounted for 50% of all deaths in the collegiate athletes.
“When you think about what’s important in terms of educating young people to be safe, the history and physical exam and 12-lead ECG are nowhere near as important as talking with them about minimizing accident risk and staying away from guns,” Dr. Baggish commented.
“Contemporary ECG interpretation criteria designed specifically for use in young athletes have eliminated the problem of false-positive testing.”
FALSE. The story of adding ECG screening to the PPCVS is one of dramatically improved sensitivity over history and physical exam alone, but always at the cost of reduced specificity. In the Harvard Athlete Initiative Study, Dr. Baggish and coworkers reported that adding the 12-lead ECG resulted in a 17% false-positive rate (Ann Intern Med. 2010 Mar 2;152[5]:269-75). Similar findings were reported in independent studies at two other large universities.
“An ECG false-positive rate of 16%-20%? That’s big trouble. Remember, the conditions we’re looking for are uncommon, with a prevalence of maybe 1 in 500 at most. So if you’re flagging one-fifth or one-sixth of your athletes, the ECG is really not an appropriate tool for screening,” he commented.
Recognition of this limitation has led to development of refined, improved ECG criteria: most notably, the 2012 Seattle criteria, with an associated false-positive rate of 4%-8%, followed by the 2017 International Consensus Criteria (J Am Coll Cardiol. 2017 Feb 28;69[8]:1057-75), with a false-positive rate of 1%-2%. That’s a great improvement. Still, when Dr. Baggish, a marathoner himself, thinks about the roughly 32,000 Boston Marathon runners at the starting line each year, that false-positive rate would translate into 320-640 of those individuals being needlessly subjected to the not-insignificant time and expense of further testing, along with considerable anxiety for the runners and their families, and perhaps even inappropriate disqualification.
“Current ACC/AHA guidelines recommend against the use of the 12-lead ECG during the PPCVS.”
FALSE. Dr. Baggish was a coauthor of the current guidelines, which he described as “an open-door invitation to local decisions, with some important caveats” (Circulation. 2015 Dec 1;132[22]:e267-72).
The guidelines state that the minimum requirement and legal standard for PPCVS of young competitive athletes is a focused history and physical examination, such as the American College of Cardiology/American Heart Association 14-point screen, which consists of 10 elements addressing personal and family history and 4 focused on the physical examination, or the American Academy of Pediatrics Preparticipation Physical Evaluation. Further, while mandatory universal inclusion of the 12-lead ECG is not recommended – it’s rated Class III, meaning don’t do it – the guidelines state that screening programs are at liberty to choose the 12-lead ECG as an additional tool, “provided that close physician involvement and sufficient quality control can be achieved. If undertaken, such initiatives should recognize the known and anticipated limitations of the 12-lead ECG as a population screening test, including the expected frequency of false-positive and false-negative test results, as well as the cost required to support these initiatives over time.”
Dr. Baggish considers the ACC/AHA guidelines to be one of the two most important developments in the field of SCD during sports in recent years. The other is the NCAA-sponsored multidisciplinary Interassociation Consensus Statement on Cardiovascular Care of College Student-Athletes, which he also coauthored (J Am Coll Cardiol. 2016 Jun 28;67[25]:2981-95).
The report lays out the case for a much broader than traditional view of the PPCVS, with “goals that extend beyond detection of occult high-risk pathology.”
“The NCAA has done something very interesting,” Dr. Baggish explained. “It has said that, if we’re going to be screening, we should be thinking about screening with a much broader rationale. It’s not just about finding the needle-in-a-haystack hypertrophic cardiomyopathy or anomalous coronary arteries, it’s about engaging student-athletes at an early point in their collegiate career and trying to improve their health overall – and not just while they’re in college, but over their lifespan.”
He reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – Myths and misconceptions abound regarding the merits of universal incorporation of the resting 12-lead ECG into preparticipation cardiovascular screening of young athletes, Aaron L. Baggish, MD, declared at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
Dr. Baggish, director of the Cardiovascular Performance Program at Massachusetts General Hospital and a cardiologist at Harvard Medical School, Boston, set out to pop the balloons of a handful of these widely floating myths. These are commonly held fictions: In an electronic poll at the outset of his talk, only one in five members of his large audience recognized all of the following boldface statements as false.
“Preparticipation cardiovascular screening (PPCVS) has been shown to reduce the incidence of sudden cardiac death (SCD) among young competitive athletes.”
FALSE. Not for PPCVS by history and physical examination alone, or with the addition of a screening 12-lead ECG. In Italy, where a cluster of high-profile sudden cardiac deaths led to passage of a 1982 national law mandating 12-lead ECG screening as part of the PPCVS, investigators presented studies purporting to demonstrate a subsequent reduction in the risk of SCD. But those studies were subsequently shown to be fraught with problems. And a high-quality study capable of convincingly demonstrating such a benefit would need to be prohibitively large and expensive. “Don’t hold your breath waiting for that to happen anytime soon,” advised Dr. Baggish, who is medical director for the Boston Marathon, as well as team cardiologist for Harvard University Athletics, the New England Patriots, the Boston Bruins, USRowing, and U.S. Soccer.
“Hypertrophic cardiomyopathy is the leading cause of sudden death among young competitive athletes.”
FALSE. A study of the National Collegiate Athletic Association (NCAA) comprehensive database, with 4.2 million athlete-years of follow-up, showed that the most common cause of SCD was autopsy-negative sudden unexplained death (SUD), accounting for 25% of cases. Hypertrophic cardiomyopathy was deemed the cause of 8% of the SCDs (Circulation. 2015 Jul 7;132[1]:10-9).
“The same thing has been shown in studies done in the United Kingdom and in Australia: . Over the next 10 years, I suspect that one of the most important areas that we’ll be looking into will be this SUD area, perhaps using molecular autopsy to make some headway there,” according to the cardiologist.
SCD is rare. In the NCAA study, the incidence was 1 in 53,703 athlete-years. In sobering contrast, accidents, suicide, and homicide accounted for 50% of all deaths in the collegiate athletes.
“When you think about what’s important in terms of educating young people to be safe, the history and physical exam and 12-lead ECG are nowhere near as important as talking with them about minimizing accident risk and staying away from guns,” Dr. Baggish commented.
“Contemporary ECG interpretation criteria designed specifically for use in young athletes have eliminated the problem of false-positive testing.”
FALSE. The story of adding ECG screening to the PPCVS is one of dramatically improved sensitivity over history and physical exam alone, but always at the cost of reduced specificity. In the Harvard Athlete Initiative Study, Dr. Baggish and coworkers reported that adding the 12-lead ECG resulted in a 17% false-positive rate (Ann Intern Med. 2010 Mar 2;152[5]:269-75). Similar findings were reported in independent studies at two other large universities.
“An ECG false-positive rate of 16%-20%? That’s big trouble. Remember, the conditions we’re looking for are uncommon, with a prevalence of maybe 1 in 500 at most. So if you’re flagging one-fifth or one-sixth of your athletes, the ECG is really not an appropriate tool for screening,” he commented.
Recognition of this limitation has led to development of refined, improved ECG criteria: most notably, the 2012 Seattle criteria, with an associated false-positive rate of 4%-8%, followed by the 2017 International Consensus Criteria (J Am Coll Cardiol. 2017 Feb 28;69[8]:1057-75), with a false-positive rate of 1%-2%. That’s a great improvement. Still, when Dr. Baggish, a marathoner himself, thinks about the roughly 32,000 Boston Marathon runners at the starting line each year, that false-positive rate would translate into 320-640 of those individuals being needlessly subjected to the not-insignificant time and expense of further testing, along with considerable anxiety for the runners and their families, and perhaps even inappropriate disqualification.
“Current ACC/AHA guidelines recommend against the use of the 12-lead ECG during the PPCVS.”
FALSE. Dr. Baggish was a coauthor of the current guidelines, which he described as “an open-door invitation to local decisions, with some important caveats” (Circulation. 2015 Dec 1;132[22]:e267-72).
The guidelines state that the minimum requirement and legal standard for PPCVS of young competitive athletes is a focused history and physical examination, such as the American College of Cardiology/American Heart Association 14-point screen, which consists of 10 elements addressing personal and family history and 4 focused on the physical examination, or the American Academy of Pediatrics Preparticipation Physical Evaluation. Further, while mandatory universal inclusion of the 12-lead ECG is not recommended – it’s rated Class III, meaning don’t do it – the guidelines state that screening programs are at liberty to choose the 12-lead ECG as an additional tool, “provided that close physician involvement and sufficient quality control can be achieved. If undertaken, such initiatives should recognize the known and anticipated limitations of the 12-lead ECG as a population screening test, including the expected frequency of false-positive and false-negative test results, as well as the cost required to support these initiatives over time.”
Dr. Baggish considers the ACC/AHA guidelines to be one of the two most important developments in the field of SCD during sports in recent years. The other is the NCAA-sponsored multidisciplinary Interassociation Consensus Statement on Cardiovascular Care of College Student-Athletes, which he also coauthored (J Am Coll Cardiol. 2016 Jun 28;67[25]:2981-95).
The report lays out the case for a much broader than traditional view of the PPCVS, with “goals that extend beyond detection of occult high-risk pathology.”
“The NCAA has done something very interesting,” Dr. Baggish explained. “It has said that, if we’re going to be screening, we should be thinking about screening with a much broader rationale. It’s not just about finding the needle-in-a-haystack hypertrophic cardiomyopathy or anomalous coronary arteries, it’s about engaging student-athletes at an early point in their collegiate career and trying to improve their health overall – and not just while they’re in college, but over their lifespan.”
He reported having no financial conflicts regarding his presentation.
REPORTING FROM ACC SNOWMASS 2020
CMS proposes second specialty tier for Medicare drugs
The Centers for Medicare & Medicaid Services’ latest maneuver to combat rising drug prices is the proposed addition of a second specialty drug tier for the Medicare Part D prescription drug benefit.
The proposal is part of a broader proposed update to Medicare Parts C and D for contract years 2021 and 2022.
In a fact sheet highlighting various elements of the overall proposal, CMS noted that Part D plan sponsors and pharmacy benefit managers have been requesting the option to add a second “preferred” specialty tier that would “encourage the use of more preferred, less expensive agents, reduce enrollee cost sharing, and reduce costs to CMS.”
Currently, all pharmaceuticals with a cost greater than $670 are placed in a single specialty tier.
During a Feb. 5 press briefing, CMS Administrator Seema Verma described this change as “giving plans more negotiating power so they can lower prices for beneficiaries even further.”
Ms. Verma used a hypothetical example of two rheumatoid arthritis drugs to illustrate how the change will work. Currently, if both are over the $670 threshold, they would both be on the specialty tier with the same cost sharing. “Creating a second preferred specialty tier would allow for a different copay and fosters a more competitive environment that places Part D plans in a better position to negotiate the price of similar drugs and pass those savings onto the patient through lower cost sharing,” she said.
CMS is proposing to allow plans to implement a preferred specialty tier for the 2021 plan year.
The agency is also seeking to drive more generic drug use as a means of lowering costs.
Ms. Verma noted that, typically, even after a generic drug is launched, health plan sponsors prefer to drive patients to the brand name product, if they can secure a greater rebate from the manufacturer.
In a separate Feb. 5 blog post, Ms. Verma noted that when a brand was included on a formulary, the generic was also on the formulary 91.8% of the time. For the times in which the generic was not, it was typically because the wholesale cost of the generic was only 5%-15% lower than the brand wholesale cost.
In an effort to encourage use of generics, CMS is seeking comment on the development of measures of generic and biosimilar use in Medicare Part D that could be incorporated in health plan star ratings.
Some of the measures proposed in the blog post include the generic substitution rate, the generic therapeutic alternative opportunity rate (which measures the number of brand fills divided by the sum of the brand and generic fills when both are available), and the biosimilar utilization rate.
[email protected]
The Centers for Medicare & Medicaid Services’ latest maneuver to combat rising drug prices is the proposed addition of a second specialty drug tier for the Medicare Part D prescription drug benefit.
The proposal is part of a broader proposed update to Medicare Parts C and D for contract years 2021 and 2022.
In a fact sheet highlighting various elements of the overall proposal, CMS noted that Part D plan sponsors and pharmacy benefit managers have been requesting the option to add a second “preferred” specialty tier that would “encourage the use of more preferred, less expensive agents, reduce enrollee cost sharing, and reduce costs to CMS.”
Currently, all pharmaceuticals with a cost greater than $670 are placed in a single specialty tier.
During a Feb. 5 press briefing, CMS Administrator Seema Verma described this change as “giving plans more negotiating power so they can lower prices for beneficiaries even further.”
Ms. Verma used a hypothetical example of two rheumatoid arthritis drugs to illustrate how the change will work. Currently, if both are over the $670 threshold, they would both be on the specialty tier with the same cost sharing. “Creating a second preferred specialty tier would allow for a different copay and fosters a more competitive environment that places Part D plans in a better position to negotiate the price of similar drugs and pass those savings onto the patient through lower cost sharing,” she said.
CMS is proposing to allow plans to implement a preferred specialty tier for the 2021 plan year.
The agency is also seeking to drive more generic drug use as a means of lowering costs.
Ms. Verma noted that, typically, even after a generic drug is launched, health plan sponsors prefer to drive patients to the brand name product, if they can secure a greater rebate from the manufacturer.
In a separate Feb. 5 blog post, Ms. Verma noted that when a brand was included on a formulary, the generic was also on the formulary 91.8% of the time. For the times in which the generic was not, it was typically because the wholesale cost of the generic was only 5%-15% lower than the brand wholesale cost.
In an effort to encourage use of generics, CMS is seeking comment on the development of measures of generic and biosimilar use in Medicare Part D that could be incorporated in health plan star ratings.
Some of the measures proposed in the blog post include the generic substitution rate, the generic therapeutic alternative opportunity rate (which measures the number of brand fills divided by the sum of the brand and generic fills when both are available), and the biosimilar utilization rate.
[email protected]
The Centers for Medicare & Medicaid Services’ latest maneuver to combat rising drug prices is the proposed addition of a second specialty drug tier for the Medicare Part D prescription drug benefit.
The proposal is part of a broader proposed update to Medicare Parts C and D for contract years 2021 and 2022.
In a fact sheet highlighting various elements of the overall proposal, CMS noted that Part D plan sponsors and pharmacy benefit managers have been requesting the option to add a second “preferred” specialty tier that would “encourage the use of more preferred, less expensive agents, reduce enrollee cost sharing, and reduce costs to CMS.”
Currently, all pharmaceuticals with a cost greater than $670 are placed in a single specialty tier.
During a Feb. 5 press briefing, CMS Administrator Seema Verma described this change as “giving plans more negotiating power so they can lower prices for beneficiaries even further.”
Ms. Verma used a hypothetical example of two rheumatoid arthritis drugs to illustrate how the change will work. Currently, if both are over the $670 threshold, they would both be on the specialty tier with the same cost sharing. “Creating a second preferred specialty tier would allow for a different copay and fosters a more competitive environment that places Part D plans in a better position to negotiate the price of similar drugs and pass those savings onto the patient through lower cost sharing,” she said.
CMS is proposing to allow plans to implement a preferred specialty tier for the 2021 plan year.
The agency is also seeking to drive more generic drug use as a means of lowering costs.
Ms. Verma noted that, typically, even after a generic drug is launched, health plan sponsors prefer to drive patients to the brand name product, if they can secure a greater rebate from the manufacturer.
In a separate Feb. 5 blog post, Ms. Verma noted that when a brand was included on a formulary, the generic was also on the formulary 91.8% of the time. For the times in which the generic was not, it was typically because the wholesale cost of the generic was only 5%-15% lower than the brand wholesale cost.
In an effort to encourage use of generics, CMS is seeking comment on the development of measures of generic and biosimilar use in Medicare Part D that could be incorporated in health plan star ratings.
Some of the measures proposed in the blog post include the generic substitution rate, the generic therapeutic alternative opportunity rate (which measures the number of brand fills divided by the sum of the brand and generic fills when both are available), and the biosimilar utilization rate.
[email protected]
Remdesivir under study as treatment for novel coronavirus

“What they’re looking at is the effect of this drug -- either the drug plus standard of care versus standard of care alone,” Anthony S. Fauci, MD, reported Feb. 7 during a press briefing held by members of President Trump’s Coronavirus Task Force. “I think pretty soon we are going to get a definitive answer, whether one of these among several drugs works.”
Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases, added that several organizations and individual investigators are developing vaccines for 2019-nCoV. In one such effort, the National Institutes of Health is working with Moderna Inc. to develop a vaccine built on a messenger RNA platform. “One of the first steps is to successfully get that [novel coronavirus] gene and insert it into the messenger RNA platform successfully and allow it to express proteins,” Dr. Fauci explained. “We’ve succeeded in that. The next [step] is to put it in a mouse animal model to induce immunogenicity, and to get the company to make [gold nanoparticle] products. All of those have been successfully implemented. There have been no glitches so far. If that continues, we will be in Phase 1 trials in people within the next two-and-a-half months.”
In another development on the same day, Robert R. Redfield, MD, director of the Centers for Disease Control and Prevention, announced that Heath & Human Services issued an interim final rule to amend foreign quarantine regulations in the wake of the public health threat posed by the 2019-nCoV. “This will enable CDC to collect certain contact information data regarding airline passengers and crew when they arrive from other countries. . .and may be exposed to communicable disease,” Dr. Redfield said. “This action is part of our multi-layered approach to the U.S. response and demonstrates our commitment to take all necessary actions to protect the American people.”
According to Alex Azar, secretary of Health and Human Services, and chair of President Trump’s Coronavirus Task Force, there are 12 confirmed cases of the novel coronavirus in the United States, including two cases of transmission to people who had not recently been in China. “Although the virus represents a potentially very serious public health threat, and we expect to continue seeing more cases here, the immediate risk to the American public is low at this time,” Mr. Azar said. “We are working as quickly as possible on the many unanswered questions about this virus. That includes exactly how it spreads, how deadly it is, whether it’s commonly transmitted by patients who are not yet displaying symptoms, and other issues.”

“What they’re looking at is the effect of this drug -- either the drug plus standard of care versus standard of care alone,” Anthony S. Fauci, MD, reported Feb. 7 during a press briefing held by members of President Trump’s Coronavirus Task Force. “I think pretty soon we are going to get a definitive answer, whether one of these among several drugs works.”
Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases, added that several organizations and individual investigators are developing vaccines for 2019-nCoV. In one such effort, the National Institutes of Health is working with Moderna Inc. to develop a vaccine built on a messenger RNA platform. “One of the first steps is to successfully get that [novel coronavirus] gene and insert it into the messenger RNA platform successfully and allow it to express proteins,” Dr. Fauci explained. “We’ve succeeded in that. The next [step] is to put it in a mouse animal model to induce immunogenicity, and to get the company to make [gold nanoparticle] products. All of those have been successfully implemented. There have been no glitches so far. If that continues, we will be in Phase 1 trials in people within the next two-and-a-half months.”
In another development on the same day, Robert R. Redfield, MD, director of the Centers for Disease Control and Prevention, announced that Heath & Human Services issued an interim final rule to amend foreign quarantine regulations in the wake of the public health threat posed by the 2019-nCoV. “This will enable CDC to collect certain contact information data regarding airline passengers and crew when they arrive from other countries. . .and may be exposed to communicable disease,” Dr. Redfield said. “This action is part of our multi-layered approach to the U.S. response and demonstrates our commitment to take all necessary actions to protect the American people.”
According to Alex Azar, secretary of Health and Human Services, and chair of President Trump’s Coronavirus Task Force, there are 12 confirmed cases of the novel coronavirus in the United States, including two cases of transmission to people who had not recently been in China. “Although the virus represents a potentially very serious public health threat, and we expect to continue seeing more cases here, the immediate risk to the American public is low at this time,” Mr. Azar said. “We are working as quickly as possible on the many unanswered questions about this virus. That includes exactly how it spreads, how deadly it is, whether it’s commonly transmitted by patients who are not yet displaying symptoms, and other issues.”

“What they’re looking at is the effect of this drug -- either the drug plus standard of care versus standard of care alone,” Anthony S. Fauci, MD, reported Feb. 7 during a press briefing held by members of President Trump’s Coronavirus Task Force. “I think pretty soon we are going to get a definitive answer, whether one of these among several drugs works.”
Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases, added that several organizations and individual investigators are developing vaccines for 2019-nCoV. In one such effort, the National Institutes of Health is working with Moderna Inc. to develop a vaccine built on a messenger RNA platform. “One of the first steps is to successfully get that [novel coronavirus] gene and insert it into the messenger RNA platform successfully and allow it to express proteins,” Dr. Fauci explained. “We’ve succeeded in that. The next [step] is to put it in a mouse animal model to induce immunogenicity, and to get the company to make [gold nanoparticle] products. All of those have been successfully implemented. There have been no glitches so far. If that continues, we will be in Phase 1 trials in people within the next two-and-a-half months.”
In another development on the same day, Robert R. Redfield, MD, director of the Centers for Disease Control and Prevention, announced that Heath & Human Services issued an interim final rule to amend foreign quarantine regulations in the wake of the public health threat posed by the 2019-nCoV. “This will enable CDC to collect certain contact information data regarding airline passengers and crew when they arrive from other countries. . .and may be exposed to communicable disease,” Dr. Redfield said. “This action is part of our multi-layered approach to the U.S. response and demonstrates our commitment to take all necessary actions to protect the American people.”
According to Alex Azar, secretary of Health and Human Services, and chair of President Trump’s Coronavirus Task Force, there are 12 confirmed cases of the novel coronavirus in the United States, including two cases of transmission to people who had not recently been in China. “Although the virus represents a potentially very serious public health threat, and we expect to continue seeing more cases here, the immediate risk to the American public is low at this time,” Mr. Azar said. “We are working as quickly as possible on the many unanswered questions about this virus. That includes exactly how it spreads, how deadly it is, whether it’s commonly transmitted by patients who are not yet displaying symptoms, and other issues.”
Report chastises government for allowing flavored e-cigarettes
according to a report on federal and state policies.
In its annual “State of Tobacco Control” report, the American Lung Association called out the federal government for issuing “inadequate guidance on flavored e-cigarettes that leaves thousands of flavored e-cigarettes on the market.” The organization urged Congress and the Food and Drug Administration “to eliminate all flavored tobacco products from the marketplace, including menthol cigarettes, flavored cigars, and e-cigarettes,” in 2020.
“Flavored tobacco products cause kids to become hooked, and now more than one in four teens (27.5%) are vaping, a staggering 135% increase over the past 2 years,” the association wrote in a news release. Federal guidance on Jan. 2, 2020, permits the sale of flavored e-cigarettes that do not use cartridges. This guidance represented a reversal after officials said in a prior announcement that regulators would “clear the market” of flavored e-cigarettes.
Graphic warning labels
The report also asked the FDA to reject product marketing applications that fail to meet public health standards, calls on the U.S. Department of Health & Human Services to “clarify and ensure that all tobacco users have access to a comprehensive tobacco cessation benefit,” and urges Congress to increase federal funding for the Centers for Disease Control and Prevention’s Office on Smoking and Health to help stop youth e-cigarette use.
“Raising the federal minimum age of sale to 21, which took effect immediately on Dec. 30, was an important first step forward,” the report says. “The American Lung Association successfully advocated for the legislation to be comprehensive and to close state exemptions, such as for military personnel, while also not limiting states from pursuing stronger protections. Additional rules from FDA to provide guidance on the law’s implementation are forthcoming.”
The FDA is expected to release graphic warning labels for cigarette packs in March. After legal setbacks to the Tobacco Control Act of 2009, which required the FDA to ensure all cigarette packs had graphic warning labels by 2011, a judgment “compels FDA to release final graphic warnings by March 15, 2020, with the warning labels appearing on all cigarette packs by June of 2021,” the American Lung Association report said.
“While the American Lung Association recognizes the federal government with an A grade for passage of a strong federal Tobacco 21 law [raising the minimum age of purchase], it also earns an F for its failure to comprehensively oversee tobacco products,” said Harold P. Wimmer, national president and CEO of the American Lung Association, in the news release. “Without meaningful actions by the federal government, the health and the future of our nation’s children are being compromised.”
The federal government received an F for its tobacco tax policies, a D for cessation coverage, and an A for its mass media campaigns, “Tips from Former Smokers” and “The Real Cost.”
Grading states
In addition, the report graded each state and the District of Columbia in terms of funding for tobacco prevention programs, strength of smoke-free workplace laws, level of state tobacco taxes, and coverage of and access to services to quit tobacco. None scored all A’s, but California, the District of Columbia, Maine, New York, and Vermont ranked the highest. Alabama, Mississippi, and North Carolina, on the other hand, received all F’s.
In November, Massachusetts became the first state to prohibit the sale of flavored tobacco products, including menthol cigarettes, and more states should follow suit, according to the association.
according to a report on federal and state policies.
In its annual “State of Tobacco Control” report, the American Lung Association called out the federal government for issuing “inadequate guidance on flavored e-cigarettes that leaves thousands of flavored e-cigarettes on the market.” The organization urged Congress and the Food and Drug Administration “to eliminate all flavored tobacco products from the marketplace, including menthol cigarettes, flavored cigars, and e-cigarettes,” in 2020.
“Flavored tobacco products cause kids to become hooked, and now more than one in four teens (27.5%) are vaping, a staggering 135% increase over the past 2 years,” the association wrote in a news release. Federal guidance on Jan. 2, 2020, permits the sale of flavored e-cigarettes that do not use cartridges. This guidance represented a reversal after officials said in a prior announcement that regulators would “clear the market” of flavored e-cigarettes.
Graphic warning labels
The report also asked the FDA to reject product marketing applications that fail to meet public health standards, calls on the U.S. Department of Health & Human Services to “clarify and ensure that all tobacco users have access to a comprehensive tobacco cessation benefit,” and urges Congress to increase federal funding for the Centers for Disease Control and Prevention’s Office on Smoking and Health to help stop youth e-cigarette use.
“Raising the federal minimum age of sale to 21, which took effect immediately on Dec. 30, was an important first step forward,” the report says. “The American Lung Association successfully advocated for the legislation to be comprehensive and to close state exemptions, such as for military personnel, while also not limiting states from pursuing stronger protections. Additional rules from FDA to provide guidance on the law’s implementation are forthcoming.”
The FDA is expected to release graphic warning labels for cigarette packs in March. After legal setbacks to the Tobacco Control Act of 2009, which required the FDA to ensure all cigarette packs had graphic warning labels by 2011, a judgment “compels FDA to release final graphic warnings by March 15, 2020, with the warning labels appearing on all cigarette packs by June of 2021,” the American Lung Association report said.
“While the American Lung Association recognizes the federal government with an A grade for passage of a strong federal Tobacco 21 law [raising the minimum age of purchase], it also earns an F for its failure to comprehensively oversee tobacco products,” said Harold P. Wimmer, national president and CEO of the American Lung Association, in the news release. “Without meaningful actions by the federal government, the health and the future of our nation’s children are being compromised.”
The federal government received an F for its tobacco tax policies, a D for cessation coverage, and an A for its mass media campaigns, “Tips from Former Smokers” and “The Real Cost.”
Grading states
In addition, the report graded each state and the District of Columbia in terms of funding for tobacco prevention programs, strength of smoke-free workplace laws, level of state tobacco taxes, and coverage of and access to services to quit tobacco. None scored all A’s, but California, the District of Columbia, Maine, New York, and Vermont ranked the highest. Alabama, Mississippi, and North Carolina, on the other hand, received all F’s.
In November, Massachusetts became the first state to prohibit the sale of flavored tobacco products, including menthol cigarettes, and more states should follow suit, according to the association.
according to a report on federal and state policies.
In its annual “State of Tobacco Control” report, the American Lung Association called out the federal government for issuing “inadequate guidance on flavored e-cigarettes that leaves thousands of flavored e-cigarettes on the market.” The organization urged Congress and the Food and Drug Administration “to eliminate all flavored tobacco products from the marketplace, including menthol cigarettes, flavored cigars, and e-cigarettes,” in 2020.
“Flavored tobacco products cause kids to become hooked, and now more than one in four teens (27.5%) are vaping, a staggering 135% increase over the past 2 years,” the association wrote in a news release. Federal guidance on Jan. 2, 2020, permits the sale of flavored e-cigarettes that do not use cartridges. This guidance represented a reversal after officials said in a prior announcement that regulators would “clear the market” of flavored e-cigarettes.
Graphic warning labels
The report also asked the FDA to reject product marketing applications that fail to meet public health standards, calls on the U.S. Department of Health & Human Services to “clarify and ensure that all tobacco users have access to a comprehensive tobacco cessation benefit,” and urges Congress to increase federal funding for the Centers for Disease Control and Prevention’s Office on Smoking and Health to help stop youth e-cigarette use.
“Raising the federal minimum age of sale to 21, which took effect immediately on Dec. 30, was an important first step forward,” the report says. “The American Lung Association successfully advocated for the legislation to be comprehensive and to close state exemptions, such as for military personnel, while also not limiting states from pursuing stronger protections. Additional rules from FDA to provide guidance on the law’s implementation are forthcoming.”
The FDA is expected to release graphic warning labels for cigarette packs in March. After legal setbacks to the Tobacco Control Act of 2009, which required the FDA to ensure all cigarette packs had graphic warning labels by 2011, a judgment “compels FDA to release final graphic warnings by March 15, 2020, with the warning labels appearing on all cigarette packs by June of 2021,” the American Lung Association report said.
“While the American Lung Association recognizes the federal government with an A grade for passage of a strong federal Tobacco 21 law [raising the minimum age of purchase], it also earns an F for its failure to comprehensively oversee tobacco products,” said Harold P. Wimmer, national president and CEO of the American Lung Association, in the news release. “Without meaningful actions by the federal government, the health and the future of our nation’s children are being compromised.”
The federal government received an F for its tobacco tax policies, a D for cessation coverage, and an A for its mass media campaigns, “Tips from Former Smokers” and “The Real Cost.”
Grading states
In addition, the report graded each state and the District of Columbia in terms of funding for tobacco prevention programs, strength of smoke-free workplace laws, level of state tobacco taxes, and coverage of and access to services to quit tobacco. None scored all A’s, but California, the District of Columbia, Maine, New York, and Vermont ranked the highest. Alabama, Mississippi, and North Carolina, on the other hand, received all F’s.
In November, Massachusetts became the first state to prohibit the sale of flavored tobacco products, including menthol cigarettes, and more states should follow suit, according to the association.
Funding failures: Tobacco prevention and cessation
When it comes to state funding for tobacco prevention and cessation, the American Lung Association grades on a curve. It did not help.

Each state’s annual funding for tobacco prevention and cessation was calculated and then compared with the Centers for Disease Control and Prevention’s recommended spending level. That percentage became the grade, with any level of funding at 80% or more of the CDC’s recommendation getting an A and anything below 50% getting an F, the ALA explained.
The three A’s went to Alaska – which spent $10.14 million, or 99.4% of the CDC-recommended $10.2 million – California (96.0%), and Maine (83.5%). The lowest levels of spending came from Georgia, which spend just 2.8% of the CDC’s recommendation of $106 million, and Missouri, which spent 3.0%, the ALA reported.
States’ grades were generally better in the four other areas of tobacco-control policy: There were 24 A’s and 9 F’s for smoke-free air laws, 1 A and 35 F’s for tobacco excise taxes, 3 A’s and 17 F’s for access to cessation treatment, and 10 A’s and 30 F’s for laws to raise the tobacco sales age to 21 years, the ALA said in the report.
Despite an overall grade of F, the federal government managed to earn some praise in that last area: “In what could only be described as unimaginable even 2 years ago, in December 2019, Congress passed bipartisan legislation to raise the minimum age of sale for tobacco products to 21,” the ALA said.
The federal government was strongly criticized on the subject of e-cigarettes. “The Trump Administration failed to prioritize public health over the tobacco industry with its Jan. 2, 2020, announcement that will leave thousands of flavored e-cigarettes on the market,” the ALA said, while concluding that the rising use of e-cigarettes in recent years “is a real-world demonstration of the failure of the U.S. Food and Drug Administration to properly oversee all tobacco products. … This failure places the lung health and lives of Americans at risk.”
When it comes to state funding for tobacco prevention and cessation, the American Lung Association grades on a curve. It did not help.

Each state’s annual funding for tobacco prevention and cessation was calculated and then compared with the Centers for Disease Control and Prevention’s recommended spending level. That percentage became the grade, with any level of funding at 80% or more of the CDC’s recommendation getting an A and anything below 50% getting an F, the ALA explained.
The three A’s went to Alaska – which spent $10.14 million, or 99.4% of the CDC-recommended $10.2 million – California (96.0%), and Maine (83.5%). The lowest levels of spending came from Georgia, which spend just 2.8% of the CDC’s recommendation of $106 million, and Missouri, which spent 3.0%, the ALA reported.
States’ grades were generally better in the four other areas of tobacco-control policy: There were 24 A’s and 9 F’s for smoke-free air laws, 1 A and 35 F’s for tobacco excise taxes, 3 A’s and 17 F’s for access to cessation treatment, and 10 A’s and 30 F’s for laws to raise the tobacco sales age to 21 years, the ALA said in the report.
Despite an overall grade of F, the federal government managed to earn some praise in that last area: “In what could only be described as unimaginable even 2 years ago, in December 2019, Congress passed bipartisan legislation to raise the minimum age of sale for tobacco products to 21,” the ALA said.
The federal government was strongly criticized on the subject of e-cigarettes. “The Trump Administration failed to prioritize public health over the tobacco industry with its Jan. 2, 2020, announcement that will leave thousands of flavored e-cigarettes on the market,” the ALA said, while concluding that the rising use of e-cigarettes in recent years “is a real-world demonstration of the failure of the U.S. Food and Drug Administration to properly oversee all tobacco products. … This failure places the lung health and lives of Americans at risk.”
When it comes to state funding for tobacco prevention and cessation, the American Lung Association grades on a curve. It did not help.

Each state’s annual funding for tobacco prevention and cessation was calculated and then compared with the Centers for Disease Control and Prevention’s recommended spending level. That percentage became the grade, with any level of funding at 80% or more of the CDC’s recommendation getting an A and anything below 50% getting an F, the ALA explained.
The three A’s went to Alaska – which spent $10.14 million, or 99.4% of the CDC-recommended $10.2 million – California (96.0%), and Maine (83.5%). The lowest levels of spending came from Georgia, which spend just 2.8% of the CDC’s recommendation of $106 million, and Missouri, which spent 3.0%, the ALA reported.
States’ grades were generally better in the four other areas of tobacco-control policy: There were 24 A’s and 9 F’s for smoke-free air laws, 1 A and 35 F’s for tobacco excise taxes, 3 A’s and 17 F’s for access to cessation treatment, and 10 A’s and 30 F’s for laws to raise the tobacco sales age to 21 years, the ALA said in the report.
Despite an overall grade of F, the federal government managed to earn some praise in that last area: “In what could only be described as unimaginable even 2 years ago, in December 2019, Congress passed bipartisan legislation to raise the minimum age of sale for tobacco products to 21,” the ALA said.
The federal government was strongly criticized on the subject of e-cigarettes. “The Trump Administration failed to prioritize public health over the tobacco industry with its Jan. 2, 2020, announcement that will leave thousands of flavored e-cigarettes on the market,” the ALA said, while concluding that the rising use of e-cigarettes in recent years “is a real-world demonstration of the failure of the U.S. Food and Drug Administration to properly oversee all tobacco products. … This failure places the lung health and lives of Americans at risk.”
Uptick in lung cancer in younger women, not related to smoking
A study of lung cancer in younger adults (less than 50 years) has found a recent trend of higher lung cancer rates in women, compared with men. The increase is driven by cases of adenocarcinoma of the lung.
The “emerging pattern of higher lung cancer incidence in young females” is not confined to geographic areas and income levels and “is not fully explained by sex-differences in smoking prevalence,” the authors comment.
Miranda M. Fidler-Benaoudia, PhD, Cancer Control Alberta, Alberta Health Services, Calgary, and colleagues examined lung cancer cases in 40 countries from 1993 to 2012.
They found that the female-to-male incidence rate ratio (IRR) had significantly crossed over from men to women in six countries, including the United States and Canada, and had nonsignificantly crossed over in a further 23 countries.
The research was published online Feb. 5 in the International Journal of Cancer.
These findings “forewarn of a higher lung cancer burden in women than men at older ages in the decades to follow, especially in higher-income settings,” write the authors. They highlight “the need for etiologic studies.”
Historically, lung cancer higher in men
Historically, lung cancer rates have been higher among men than women, owing to the fact that men start smoking in large numbers earlier and smoke at higher rates, the researchers comment.
However, there has been a convergence in lung cancer incidence between men and women. A recent study suggests that, in the United States, the incidence in young women is higher than that in their male counterparts.
To determine the degree to which this phenomenon is occurring globally, the team used national or subnational registry data from Cancer Incidence in Five Continents, volumes VIII–XI.
These included lung and bronchial cancer cases in 40 countries from 1993 to 2012, divided into 5-year periods. Individuals were categorized into 5-year age bands.
In addition, the team used the Global Health Data Exchange to extract data from the Global Burden of Disease Study 2015 and derive country- and sex-specific daily smoking prevalence rates.
The researchers found that among young men and women, there were three patterns in the occurrence of lung cancer between the periods 1993-1997 and 2008-2012:
- A significant crossover from male to female dominance, seen in six countries.
- An insignificant crossover from male to female dominance, found in 23 countries.
- A continued male dominance, observed in 11 countries.
Higher incidence in women in six countries
The six countries with significant crossover from male to female dominance were Canada, Denmark, Germany, New Zealand, the Netherlands, and the United States.
Further analysis showed that, in general, age-specific lung cancer incidence rates decreased in successive male birth cohorts in these six countries. There was more variation across female birth cohorts.
Calculating female-to-male incidence rate ratios, the team found, for example, the IRR increased in New Zealand from 1.0 in the 1953 birth cohort to 1.6 in the 1968 birth cohort for people aged 40-44 years.
In addition, among adults aged 45-49 years in the Netherlands, the IRR rose from 0.7 in those born in the circa 1948 cohort to 1.4 in those from the circa 1958 cohort.
Overall, female-to-male IRRs increased notably among the following groups:
- Individuals aged 30-34 years in Canada, Denmark, and Germany.
- Those aged 40-44 years in Germany, the Netherlands, and the United States.
- Those aged 44-50 years in the Netherlands and the United States.
- Those aged 50-54 years in Canada, Denmark, and New Zealand.
Countries with an insignificant crossover from male to female dominance of lung cancer were located across Africa, the Americas, Asia, Europe, and Oceania.
Again, incidence rates were typically characterized by falling rates of lung cancer among men in more recent birth cohorts, and lung cancer incidence trends were more variable in women.
The team writes: “Of note, the six countries demonstrating a significant crossover are among those considered to be more advanced in the tobacco epidemic.
“Many of the countries where the crossover was insignificant or when there was no crossover are considered to be late adopters of the tobacco epidemic, with the effects of the epidemic on the burden of lung cancer and other smoking-related diseases beginning to manifest more recently, or perhaps yet to come.”
They suggest that low- and middle-resource countries may not follow the tobacco epidemic pattern of high-income countries, and so “we may not see higher lung cancer incidence rates in women than men for the foreseeable future in these countries.”
No funding for the study has been disclosed. The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A study of lung cancer in younger adults (less than 50 years) has found a recent trend of higher lung cancer rates in women, compared with men. The increase is driven by cases of adenocarcinoma of the lung.
The “emerging pattern of higher lung cancer incidence in young females” is not confined to geographic areas and income levels and “is not fully explained by sex-differences in smoking prevalence,” the authors comment.
Miranda M. Fidler-Benaoudia, PhD, Cancer Control Alberta, Alberta Health Services, Calgary, and colleagues examined lung cancer cases in 40 countries from 1993 to 2012.
They found that the female-to-male incidence rate ratio (IRR) had significantly crossed over from men to women in six countries, including the United States and Canada, and had nonsignificantly crossed over in a further 23 countries.
The research was published online Feb. 5 in the International Journal of Cancer.
These findings “forewarn of a higher lung cancer burden in women than men at older ages in the decades to follow, especially in higher-income settings,” write the authors. They highlight “the need for etiologic studies.”
Historically, lung cancer higher in men
Historically, lung cancer rates have been higher among men than women, owing to the fact that men start smoking in large numbers earlier and smoke at higher rates, the researchers comment.
However, there has been a convergence in lung cancer incidence between men and women. A recent study suggests that, in the United States, the incidence in young women is higher than that in their male counterparts.
To determine the degree to which this phenomenon is occurring globally, the team used national or subnational registry data from Cancer Incidence in Five Continents, volumes VIII–XI.
These included lung and bronchial cancer cases in 40 countries from 1993 to 2012, divided into 5-year periods. Individuals were categorized into 5-year age bands.
In addition, the team used the Global Health Data Exchange to extract data from the Global Burden of Disease Study 2015 and derive country- and sex-specific daily smoking prevalence rates.
The researchers found that among young men and women, there were three patterns in the occurrence of lung cancer between the periods 1993-1997 and 2008-2012:
- A significant crossover from male to female dominance, seen in six countries.
- An insignificant crossover from male to female dominance, found in 23 countries.
- A continued male dominance, observed in 11 countries.
Higher incidence in women in six countries
The six countries with significant crossover from male to female dominance were Canada, Denmark, Germany, New Zealand, the Netherlands, and the United States.
Further analysis showed that, in general, age-specific lung cancer incidence rates decreased in successive male birth cohorts in these six countries. There was more variation across female birth cohorts.
Calculating female-to-male incidence rate ratios, the team found, for example, the IRR increased in New Zealand from 1.0 in the 1953 birth cohort to 1.6 in the 1968 birth cohort for people aged 40-44 years.
In addition, among adults aged 45-49 years in the Netherlands, the IRR rose from 0.7 in those born in the circa 1948 cohort to 1.4 in those from the circa 1958 cohort.
Overall, female-to-male IRRs increased notably among the following groups:
- Individuals aged 30-34 years in Canada, Denmark, and Germany.
- Those aged 40-44 years in Germany, the Netherlands, and the United States.
- Those aged 44-50 years in the Netherlands and the United States.
- Those aged 50-54 years in Canada, Denmark, and New Zealand.
Countries with an insignificant crossover from male to female dominance of lung cancer were located across Africa, the Americas, Asia, Europe, and Oceania.
Again, incidence rates were typically characterized by falling rates of lung cancer among men in more recent birth cohorts, and lung cancer incidence trends were more variable in women.
The team writes: “Of note, the six countries demonstrating a significant crossover are among those considered to be more advanced in the tobacco epidemic.
“Many of the countries where the crossover was insignificant or when there was no crossover are considered to be late adopters of the tobacco epidemic, with the effects of the epidemic on the burden of lung cancer and other smoking-related diseases beginning to manifest more recently, or perhaps yet to come.”
They suggest that low- and middle-resource countries may not follow the tobacco epidemic pattern of high-income countries, and so “we may not see higher lung cancer incidence rates in women than men for the foreseeable future in these countries.”
No funding for the study has been disclosed. The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A study of lung cancer in younger adults (less than 50 years) has found a recent trend of higher lung cancer rates in women, compared with men. The increase is driven by cases of adenocarcinoma of the lung.
The “emerging pattern of higher lung cancer incidence in young females” is not confined to geographic areas and income levels and “is not fully explained by sex-differences in smoking prevalence,” the authors comment.
Miranda M. Fidler-Benaoudia, PhD, Cancer Control Alberta, Alberta Health Services, Calgary, and colleagues examined lung cancer cases in 40 countries from 1993 to 2012.
They found that the female-to-male incidence rate ratio (IRR) had significantly crossed over from men to women in six countries, including the United States and Canada, and had nonsignificantly crossed over in a further 23 countries.
The research was published online Feb. 5 in the International Journal of Cancer.
These findings “forewarn of a higher lung cancer burden in women than men at older ages in the decades to follow, especially in higher-income settings,” write the authors. They highlight “the need for etiologic studies.”
Historically, lung cancer higher in men
Historically, lung cancer rates have been higher among men than women, owing to the fact that men start smoking in large numbers earlier and smoke at higher rates, the researchers comment.
However, there has been a convergence in lung cancer incidence between men and women. A recent study suggests that, in the United States, the incidence in young women is higher than that in their male counterparts.
To determine the degree to which this phenomenon is occurring globally, the team used national or subnational registry data from Cancer Incidence in Five Continents, volumes VIII–XI.
These included lung and bronchial cancer cases in 40 countries from 1993 to 2012, divided into 5-year periods. Individuals were categorized into 5-year age bands.
In addition, the team used the Global Health Data Exchange to extract data from the Global Burden of Disease Study 2015 and derive country- and sex-specific daily smoking prevalence rates.
The researchers found that among young men and women, there were three patterns in the occurrence of lung cancer between the periods 1993-1997 and 2008-2012:
- A significant crossover from male to female dominance, seen in six countries.
- An insignificant crossover from male to female dominance, found in 23 countries.
- A continued male dominance, observed in 11 countries.
Higher incidence in women in six countries
The six countries with significant crossover from male to female dominance were Canada, Denmark, Germany, New Zealand, the Netherlands, and the United States.
Further analysis showed that, in general, age-specific lung cancer incidence rates decreased in successive male birth cohorts in these six countries. There was more variation across female birth cohorts.
Calculating female-to-male incidence rate ratios, the team found, for example, the IRR increased in New Zealand from 1.0 in the 1953 birth cohort to 1.6 in the 1968 birth cohort for people aged 40-44 years.
In addition, among adults aged 45-49 years in the Netherlands, the IRR rose from 0.7 in those born in the circa 1948 cohort to 1.4 in those from the circa 1958 cohort.
Overall, female-to-male IRRs increased notably among the following groups:
- Individuals aged 30-34 years in Canada, Denmark, and Germany.
- Those aged 40-44 years in Germany, the Netherlands, and the United States.
- Those aged 44-50 years in the Netherlands and the United States.
- Those aged 50-54 years in Canada, Denmark, and New Zealand.
Countries with an insignificant crossover from male to female dominance of lung cancer were located across Africa, the Americas, Asia, Europe, and Oceania.
Again, incidence rates were typically characterized by falling rates of lung cancer among men in more recent birth cohorts, and lung cancer incidence trends were more variable in women.
The team writes: “Of note, the six countries demonstrating a significant crossover are among those considered to be more advanced in the tobacco epidemic.
“Many of the countries where the crossover was insignificant or when there was no crossover are considered to be late adopters of the tobacco epidemic, with the effects of the epidemic on the burden of lung cancer and other smoking-related diseases beginning to manifest more recently, or perhaps yet to come.”
They suggest that low- and middle-resource countries may not follow the tobacco epidemic pattern of high-income countries, and so “we may not see higher lung cancer incidence rates in women than men for the foreseeable future in these countries.”
No funding for the study has been disclosed. The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
New tools could help predict complication risks in lung and breast cancer
In this edition of “How I Will Treat My Next Patient,” I highlight the potential role of new models for predicting risks of common, clinically important situations in general oncology practice: severe neutropenia in lung cancer patients and locoregional recurrence of breast cancer.
Predicting neutropenia
Accurate, lung cancer–specific prediction models would be useful to estimate risk of chemotherapy-induced neutropenia (CIN), especially febrile neutropenia (FN), since that particular toxicity is linked to infection, dose delays and dose reductions that can compromise treatment efficacy, and poor health-related quality of life. Lung cancer patients are often older adults, with advanced disease and comorbid conditions, so they are a particularly vulnerable population for CIN.
Xiaowen Cao of Duke University, Durham, N.C., and coinvestigators published a model for predicting risk of severe CIN in advanced lung cancer patients, based on 10 pretreatment variables (Lung Cancer. 2020 Jan 5. doi: 10.1016/j.lungcan.2020.01.004). They developed their model to overcome limitations of the previously published work of Gary H. Lyman, MD, and colleagues that is not specific to lung cancer and incorporated relative dose intensity as a predictor (Cancer. 2011;117:1917-27). Relative dose intensity is not determined until after a treatment course is completed.
The new prediction model was based on a lung cancer data set encompassing 11,352 patients from 67 phase 2-3 cooperative group studies conducted between 1991 and 2010. In this data set, the Lyman model had an area under the curve of 0.8772 in patients with small cell lung cancer, but an area under the curve of just 0.6787 in non–small cell lung cancer.
The derivation model was derived from about two-thirds of the patients, randomly selected. The validation set was conducted using the remaining third. The variables included were readily clinically available: age, gender, weight, body mass index, insurance status, disease stage, number of metastatic sites, chemotherapy agents used, number of chemotherapy agents, planned growth factor use, duration of planned therapy, pleural effusion, presence of symptoms, and performance status. Their model had an area under the curve of 0.8348 in the training set and 0.8234 in the testing set.
How these results influence practice
The risk of an initial episode of FN is highest during a patient’s initial cycle of chemotherapy, when most patients are receiving full-dose treatment, often without prophylactic measures. Guidelines from the National Comprehensive Cancer Network suggest the use of prophylactic growth factors in patients with more than a 20% risk of FN, and considering using prophylaxis in patients with 10%-20% risk of FN. Underestimating those risks and failure to take adequate precautions may be particularly important for patients with lung cancer who are generally older adults, with comorbid conditions.
The comprehensive risk model for neutropenic complications that was developed by Dr. Lyman and colleagues was based on a large, prospective cohort including nearly 3,800 patients. The model had a 90% sensitivity and 96% predictive value, but was not lung cancer specific and, in this latest study, did not perform as well in the 85% of lung cancer patients with non–small cell lung cancer. The Lyman data, however, was obtained in cancer patients treated with investigator-choice chemotherapy in community practices. It remains the National Comprehensive Cancer Network standard for evaluating FN risk in patients embarking on chemotherapy for advanced malignancies. That should remain the case, pending the additional validation testing of the new lung cancer–specific model at independent institutions, treating heterogeneous patients in real-world settings.
Locoregional recurrence
A retrospective cohort analysis of SWOG 8814, a phase 3 study of tamoxifen alone versus chemotherapy plus by tamoxifen in postmenopausal, node-positive, hormone receptor–positive breast cancer patients suggests that the 21-gene assay recurrence score (RS) can aid decisions about radiotherapy (RT).
Wendy A. Woodward, MD, PhD, and colleagues, analyzed patients who underwent mastectomy or breast-conserving surgery as their local therapy (JAMA Oncol. 2020 Jan 9. doi: 10.1001/jamaoncol.2019.5559). They found that patients with an intermediate or high RS – according to the 21-gene assay OncotypeDX – had more locoregional recurrences (LRR; breast, chest wall, axilla, internal mammary, supraclavicular or infraclavicular nodes).
There were 367 patients in SWOG 8814 who received tamoxifen alone or cyclophosphamide, doxorubicin, and fluorouracil followed by tamoxifen. LRR was observed in 5.8% of patients with a low RS (less than 18) and in 13.8% of patients with an intermediate or high RS (more than 18). The estimated 10-year cumulative LRR incidence rates were 9.7% and 16.5%, respectively (P = .02).
In the subset of patients with one to three positive nodes who had mastectomy without radiotherapy, the LRR was 1.5% for those with low RS and 11.1% for those with intermediate or high RS (P = .051). No difference by RS was found in the 10-year rates of LRR among patients with four or more involved nodes who received a mastectomy without RT (25.9% vs. 27.0%; P = .27).
In multivariate analysis, incorporating RS, type of surgery, and number of involved nodes, intermediate or high RS was a significant predictor of LRR, with a hazard ratio of 2.36 (P = .04). The investigators suggested that RS, when available, should be one of the factors considered in selecting patients for postmastectomy RT.
How these results influence practice
Selecting the node-positive, hormone receptor–positive, breast cancer patients who should receive postmastectomy RT is difficult and controversial. This is particularly true for those postmenopausal patients with fewer than four involved nodes, no lymphatic or vascular invasion, and no extracapsular spread of disease into the axillary fat. Limited information exists on the ability of genomic assays to identify LRR risk.
Eleftherios P. Mamounas, MD, and colleagues examined the results of NSABP B-28, a trial of chemotherapy plus tamoxifen (J Natl Cancer Inst. 2017;109[4]. doi:10.1093/jnci/djw259). Postmastectomy RT was not permitted. They found high RS correlated with greater LRR and low RS with decreased LRR among patients with one to three positive nodes. At first blush, the prospectively treated cohort of SWOG 8814 represents a uniformly treated cohort with long-term follow-up (median, 8.5 years) and extends in an independent analysis the findings of NSABP B-28.
However, as Dr. Woodward and colleagues point out, the current study has limitations. The use of RT was extracted retrospectively and may be underreported. More modern chemotherapy and RT may lower LRR from the risks observed in SWOG 8814. Finally, the modest numbers of LRR events precluded secondary analysis of RS as a continuous variable. This is important because the risk group cutoffs suggested by the authors are not aligned with those in the recently published TailorRx study or the ongoing RxPonder trial.
The TailorRT (Regional Radiotherapy in Biomarker Low Risk Node Positive Breast Cancer) study examines the safety of omitting RT among patients with low RS and one to three positive nodes. Until the TailorRT results are reported, the controversy regarding the role of postmastectomy RT in this group will continue for patients with low nodal tumor burden and less aggressive tumor features, including low RS.
An observed LRR risk of 11.1% in SWOG 8814 among patients with N1 disease and an RS above 18 suggest that genomic risk could be one of the factors that may justify postmastectomy RT in postmenopausal patients with node-positive, hormone receptor–positive breast cancer until additional data emerge from the contemporary trials.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight the potential role of new models for predicting risks of common, clinically important situations in general oncology practice: severe neutropenia in lung cancer patients and locoregional recurrence of breast cancer.
Predicting neutropenia
Accurate, lung cancer–specific prediction models would be useful to estimate risk of chemotherapy-induced neutropenia (CIN), especially febrile neutropenia (FN), since that particular toxicity is linked to infection, dose delays and dose reductions that can compromise treatment efficacy, and poor health-related quality of life. Lung cancer patients are often older adults, with advanced disease and comorbid conditions, so they are a particularly vulnerable population for CIN.
Xiaowen Cao of Duke University, Durham, N.C., and coinvestigators published a model for predicting risk of severe CIN in advanced lung cancer patients, based on 10 pretreatment variables (Lung Cancer. 2020 Jan 5. doi: 10.1016/j.lungcan.2020.01.004). They developed their model to overcome limitations of the previously published work of Gary H. Lyman, MD, and colleagues that is not specific to lung cancer and incorporated relative dose intensity as a predictor (Cancer. 2011;117:1917-27). Relative dose intensity is not determined until after a treatment course is completed.
The new prediction model was based on a lung cancer data set encompassing 11,352 patients from 67 phase 2-3 cooperative group studies conducted between 1991 and 2010. In this data set, the Lyman model had an area under the curve of 0.8772 in patients with small cell lung cancer, but an area under the curve of just 0.6787 in non–small cell lung cancer.
The derivation model was derived from about two-thirds of the patients, randomly selected. The validation set was conducted using the remaining third. The variables included were readily clinically available: age, gender, weight, body mass index, insurance status, disease stage, number of metastatic sites, chemotherapy agents used, number of chemotherapy agents, planned growth factor use, duration of planned therapy, pleural effusion, presence of symptoms, and performance status. Their model had an area under the curve of 0.8348 in the training set and 0.8234 in the testing set.
How these results influence practice
The risk of an initial episode of FN is highest during a patient’s initial cycle of chemotherapy, when most patients are receiving full-dose treatment, often without prophylactic measures. Guidelines from the National Comprehensive Cancer Network suggest the use of prophylactic growth factors in patients with more than a 20% risk of FN, and considering using prophylaxis in patients with 10%-20% risk of FN. Underestimating those risks and failure to take adequate precautions may be particularly important for patients with lung cancer who are generally older adults, with comorbid conditions.
The comprehensive risk model for neutropenic complications that was developed by Dr. Lyman and colleagues was based on a large, prospective cohort including nearly 3,800 patients. The model had a 90% sensitivity and 96% predictive value, but was not lung cancer specific and, in this latest study, did not perform as well in the 85% of lung cancer patients with non–small cell lung cancer. The Lyman data, however, was obtained in cancer patients treated with investigator-choice chemotherapy in community practices. It remains the National Comprehensive Cancer Network standard for evaluating FN risk in patients embarking on chemotherapy for advanced malignancies. That should remain the case, pending the additional validation testing of the new lung cancer–specific model at independent institutions, treating heterogeneous patients in real-world settings.
Locoregional recurrence
A retrospective cohort analysis of SWOG 8814, a phase 3 study of tamoxifen alone versus chemotherapy plus by tamoxifen in postmenopausal, node-positive, hormone receptor–positive breast cancer patients suggests that the 21-gene assay recurrence score (RS) can aid decisions about radiotherapy (RT).
Wendy A. Woodward, MD, PhD, and colleagues, analyzed patients who underwent mastectomy or breast-conserving surgery as their local therapy (JAMA Oncol. 2020 Jan 9. doi: 10.1001/jamaoncol.2019.5559). They found that patients with an intermediate or high RS – according to the 21-gene assay OncotypeDX – had more locoregional recurrences (LRR; breast, chest wall, axilla, internal mammary, supraclavicular or infraclavicular nodes).
There were 367 patients in SWOG 8814 who received tamoxifen alone or cyclophosphamide, doxorubicin, and fluorouracil followed by tamoxifen. LRR was observed in 5.8% of patients with a low RS (less than 18) and in 13.8% of patients with an intermediate or high RS (more than 18). The estimated 10-year cumulative LRR incidence rates were 9.7% and 16.5%, respectively (P = .02).
In the subset of patients with one to three positive nodes who had mastectomy without radiotherapy, the LRR was 1.5% for those with low RS and 11.1% for those with intermediate or high RS (P = .051). No difference by RS was found in the 10-year rates of LRR among patients with four or more involved nodes who received a mastectomy without RT (25.9% vs. 27.0%; P = .27).
In multivariate analysis, incorporating RS, type of surgery, and number of involved nodes, intermediate or high RS was a significant predictor of LRR, with a hazard ratio of 2.36 (P = .04). The investigators suggested that RS, when available, should be one of the factors considered in selecting patients for postmastectomy RT.
How these results influence practice
Selecting the node-positive, hormone receptor–positive, breast cancer patients who should receive postmastectomy RT is difficult and controversial. This is particularly true for those postmenopausal patients with fewer than four involved nodes, no lymphatic or vascular invasion, and no extracapsular spread of disease into the axillary fat. Limited information exists on the ability of genomic assays to identify LRR risk.
Eleftherios P. Mamounas, MD, and colleagues examined the results of NSABP B-28, a trial of chemotherapy plus tamoxifen (J Natl Cancer Inst. 2017;109[4]. doi:10.1093/jnci/djw259). Postmastectomy RT was not permitted. They found high RS correlated with greater LRR and low RS with decreased LRR among patients with one to three positive nodes. At first blush, the prospectively treated cohort of SWOG 8814 represents a uniformly treated cohort with long-term follow-up (median, 8.5 years) and extends in an independent analysis the findings of NSABP B-28.
However, as Dr. Woodward and colleagues point out, the current study has limitations. The use of RT was extracted retrospectively and may be underreported. More modern chemotherapy and RT may lower LRR from the risks observed in SWOG 8814. Finally, the modest numbers of LRR events precluded secondary analysis of RS as a continuous variable. This is important because the risk group cutoffs suggested by the authors are not aligned with those in the recently published TailorRx study or the ongoing RxPonder trial.
The TailorRT (Regional Radiotherapy in Biomarker Low Risk Node Positive Breast Cancer) study examines the safety of omitting RT among patients with low RS and one to three positive nodes. Until the TailorRT results are reported, the controversy regarding the role of postmastectomy RT in this group will continue for patients with low nodal tumor burden and less aggressive tumor features, including low RS.
An observed LRR risk of 11.1% in SWOG 8814 among patients with N1 disease and an RS above 18 suggest that genomic risk could be one of the factors that may justify postmastectomy RT in postmenopausal patients with node-positive, hormone receptor–positive breast cancer until additional data emerge from the contemporary trials.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight the potential role of new models for predicting risks of common, clinically important situations in general oncology practice: severe neutropenia in lung cancer patients and locoregional recurrence of breast cancer.
Predicting neutropenia
Accurate, lung cancer–specific prediction models would be useful to estimate risk of chemotherapy-induced neutropenia (CIN), especially febrile neutropenia (FN), since that particular toxicity is linked to infection, dose delays and dose reductions that can compromise treatment efficacy, and poor health-related quality of life. Lung cancer patients are often older adults, with advanced disease and comorbid conditions, so they are a particularly vulnerable population for CIN.
Xiaowen Cao of Duke University, Durham, N.C., and coinvestigators published a model for predicting risk of severe CIN in advanced lung cancer patients, based on 10 pretreatment variables (Lung Cancer. 2020 Jan 5. doi: 10.1016/j.lungcan.2020.01.004). They developed their model to overcome limitations of the previously published work of Gary H. Lyman, MD, and colleagues that is not specific to lung cancer and incorporated relative dose intensity as a predictor (Cancer. 2011;117:1917-27). Relative dose intensity is not determined until after a treatment course is completed.
The new prediction model was based on a lung cancer data set encompassing 11,352 patients from 67 phase 2-3 cooperative group studies conducted between 1991 and 2010. In this data set, the Lyman model had an area under the curve of 0.8772 in patients with small cell lung cancer, but an area under the curve of just 0.6787 in non–small cell lung cancer.
The derivation model was derived from about two-thirds of the patients, randomly selected. The validation set was conducted using the remaining third. The variables included were readily clinically available: age, gender, weight, body mass index, insurance status, disease stage, number of metastatic sites, chemotherapy agents used, number of chemotherapy agents, planned growth factor use, duration of planned therapy, pleural effusion, presence of symptoms, and performance status. Their model had an area under the curve of 0.8348 in the training set and 0.8234 in the testing set.
How these results influence practice
The risk of an initial episode of FN is highest during a patient’s initial cycle of chemotherapy, when most patients are receiving full-dose treatment, often without prophylactic measures. Guidelines from the National Comprehensive Cancer Network suggest the use of prophylactic growth factors in patients with more than a 20% risk of FN, and considering using prophylaxis in patients with 10%-20% risk of FN. Underestimating those risks and failure to take adequate precautions may be particularly important for patients with lung cancer who are generally older adults, with comorbid conditions.
The comprehensive risk model for neutropenic complications that was developed by Dr. Lyman and colleagues was based on a large, prospective cohort including nearly 3,800 patients. The model had a 90% sensitivity and 96% predictive value, but was not lung cancer specific and, in this latest study, did not perform as well in the 85% of lung cancer patients with non–small cell lung cancer. The Lyman data, however, was obtained in cancer patients treated with investigator-choice chemotherapy in community practices. It remains the National Comprehensive Cancer Network standard for evaluating FN risk in patients embarking on chemotherapy for advanced malignancies. That should remain the case, pending the additional validation testing of the new lung cancer–specific model at independent institutions, treating heterogeneous patients in real-world settings.
Locoregional recurrence
A retrospective cohort analysis of SWOG 8814, a phase 3 study of tamoxifen alone versus chemotherapy plus by tamoxifen in postmenopausal, node-positive, hormone receptor–positive breast cancer patients suggests that the 21-gene assay recurrence score (RS) can aid decisions about radiotherapy (RT).
Wendy A. Woodward, MD, PhD, and colleagues, analyzed patients who underwent mastectomy or breast-conserving surgery as their local therapy (JAMA Oncol. 2020 Jan 9. doi: 10.1001/jamaoncol.2019.5559). They found that patients with an intermediate or high RS – according to the 21-gene assay OncotypeDX – had more locoregional recurrences (LRR; breast, chest wall, axilla, internal mammary, supraclavicular or infraclavicular nodes).
There were 367 patients in SWOG 8814 who received tamoxifen alone or cyclophosphamide, doxorubicin, and fluorouracil followed by tamoxifen. LRR was observed in 5.8% of patients with a low RS (less than 18) and in 13.8% of patients with an intermediate or high RS (more than 18). The estimated 10-year cumulative LRR incidence rates were 9.7% and 16.5%, respectively (P = .02).
In the subset of patients with one to three positive nodes who had mastectomy without radiotherapy, the LRR was 1.5% for those with low RS and 11.1% for those with intermediate or high RS (P = .051). No difference by RS was found in the 10-year rates of LRR among patients with four or more involved nodes who received a mastectomy without RT (25.9% vs. 27.0%; P = .27).
In multivariate analysis, incorporating RS, type of surgery, and number of involved nodes, intermediate or high RS was a significant predictor of LRR, with a hazard ratio of 2.36 (P = .04). The investigators suggested that RS, when available, should be one of the factors considered in selecting patients for postmastectomy RT.
How these results influence practice
Selecting the node-positive, hormone receptor–positive, breast cancer patients who should receive postmastectomy RT is difficult and controversial. This is particularly true for those postmenopausal patients with fewer than four involved nodes, no lymphatic or vascular invasion, and no extracapsular spread of disease into the axillary fat. Limited information exists on the ability of genomic assays to identify LRR risk.
Eleftherios P. Mamounas, MD, and colleagues examined the results of NSABP B-28, a trial of chemotherapy plus tamoxifen (J Natl Cancer Inst. 2017;109[4]. doi:10.1093/jnci/djw259). Postmastectomy RT was not permitted. They found high RS correlated with greater LRR and low RS with decreased LRR among patients with one to three positive nodes. At first blush, the prospectively treated cohort of SWOG 8814 represents a uniformly treated cohort with long-term follow-up (median, 8.5 years) and extends in an independent analysis the findings of NSABP B-28.
However, as Dr. Woodward and colleagues point out, the current study has limitations. The use of RT was extracted retrospectively and may be underreported. More modern chemotherapy and RT may lower LRR from the risks observed in SWOG 8814. Finally, the modest numbers of LRR events precluded secondary analysis of RS as a continuous variable. This is important because the risk group cutoffs suggested by the authors are not aligned with those in the recently published TailorRx study or the ongoing RxPonder trial.
The TailorRT (Regional Radiotherapy in Biomarker Low Risk Node Positive Breast Cancer) study examines the safety of omitting RT among patients with low RS and one to three positive nodes. Until the TailorRT results are reported, the controversy regarding the role of postmastectomy RT in this group will continue for patients with low nodal tumor burden and less aggressive tumor features, including low RS.
An observed LRR risk of 11.1% in SWOG 8814 among patients with N1 disease and an RS above 18 suggest that genomic risk could be one of the factors that may justify postmastectomy RT in postmenopausal patients with node-positive, hormone receptor–positive breast cancer until additional data emerge from the contemporary trials.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
The power of an odd couple
The time has come for good men and women to unite and rise up against a common foe. For too long nurses and doctors have labored under the tyranny of a dictator who claimed to help them provide high-quality care for their patients while at the same time cutting their paperwork to nil. But like most autocrats he failed to engage his subjects in a meaningful dialogue as each new version of his promised improvements rolled off the drawing board. When the caregivers were slow to adopt these new nonsystems he offered them financial incentives and issued threats to their survival. Although they were warned that there might be uncomfortable adjustment periods, the caregivers were promised that the steep learning curves would level out and their professional lives would again be valued and productive.
Of course, the dictator is not a single person but a motley and disorganized conglomerate of user- and patient-unfriendly electronic health record nonsystems. Ask almost any nurse or physician for her feelings about computer-based medical record systems, and you will hear tales of long hours, disengagement, and frustration. Caregivers are unhappy at all levels, and patients have grown tired of their nurses and physicians spending most of their time looking at computer screens.
You certainly have heard this all before. But you are hearing it in hospital hallways and grocery store checkout lines as a low rumble of discontent emerging from separate individuals, not as a well-articulated and widely distributed voice of physicians as a group. To some extent this relative silence is because there is no such group, at least not in same mold as a labor union. The term “labor union” may make you uncomfortable. But given the current climate in medicine, unionizing may be the best and only way to effect change.
But organizing to effect change in the workplace isn’t part of the physician genome. In the 1960s, a group of house officers in Boston engaged in a heal-in to successfully improve their salaries and working conditions. But over the ensuing half century physicians have remained tragically silent in the face of a changing workplace landscape in which they have gone from being independent owner operators in control of their destinies to becoming employees feeling powerless to improve their working conditions. This perceived impotence has escalated in the face of the challenge posed by the introduction of dysfunctional EHRs.
Ironically, a solution is at almost every physician’s elbow. In a recent New York Times opinion piece Theresa Brown and Stephen Bergman acknowledge that physicians don’t seem prepared to mount a meaningful response to the challenge to the failed promise of EHRs (“Doctors, Nurses and the Paperwork Crisis That Could Unite Them,” Dec. 31, 2019). They point out that, over the last half century, physicians have remained isolated on the sidelines, finding just enough voice to grumble. Nurses have in a variety of situations organized to effect change in their working conditions – in some cases by forming labor unions.
The authors of this op-ed piece, a physician and a nurse, make a strong argument that the time has come for nurses and doctors shake off the shackles of their stereotypic roles and join in creating a loud, forceful, and effective voice to demand a working environment in which the computer functions as an asset and no longer as the terrible burden it has become. Neither group has the power to do it alone, but together they may be able to turn the tide. For physicians it will probably mean venturing several steps outside of their comfort zone. But working shoulder to shoulder with nurses may provide the courage to speak out.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
The time has come for good men and women to unite and rise up against a common foe. For too long nurses and doctors have labored under the tyranny of a dictator who claimed to help them provide high-quality care for their patients while at the same time cutting their paperwork to nil. But like most autocrats he failed to engage his subjects in a meaningful dialogue as each new version of his promised improvements rolled off the drawing board. When the caregivers were slow to adopt these new nonsystems he offered them financial incentives and issued threats to their survival. Although they were warned that there might be uncomfortable adjustment periods, the caregivers were promised that the steep learning curves would level out and their professional lives would again be valued and productive.
Of course, the dictator is not a single person but a motley and disorganized conglomerate of user- and patient-unfriendly electronic health record nonsystems. Ask almost any nurse or physician for her feelings about computer-based medical record systems, and you will hear tales of long hours, disengagement, and frustration. Caregivers are unhappy at all levels, and patients have grown tired of their nurses and physicians spending most of their time looking at computer screens.
You certainly have heard this all before. But you are hearing it in hospital hallways and grocery store checkout lines as a low rumble of discontent emerging from separate individuals, not as a well-articulated and widely distributed voice of physicians as a group. To some extent this relative silence is because there is no such group, at least not in same mold as a labor union. The term “labor union” may make you uncomfortable. But given the current climate in medicine, unionizing may be the best and only way to effect change.
But organizing to effect change in the workplace isn’t part of the physician genome. In the 1960s, a group of house officers in Boston engaged in a heal-in to successfully improve their salaries and working conditions. But over the ensuing half century physicians have remained tragically silent in the face of a changing workplace landscape in which they have gone from being independent owner operators in control of their destinies to becoming employees feeling powerless to improve their working conditions. This perceived impotence has escalated in the face of the challenge posed by the introduction of dysfunctional EHRs.
Ironically, a solution is at almost every physician’s elbow. In a recent New York Times opinion piece Theresa Brown and Stephen Bergman acknowledge that physicians don’t seem prepared to mount a meaningful response to the challenge to the failed promise of EHRs (“Doctors, Nurses and the Paperwork Crisis That Could Unite Them,” Dec. 31, 2019). They point out that, over the last half century, physicians have remained isolated on the sidelines, finding just enough voice to grumble. Nurses have in a variety of situations organized to effect change in their working conditions – in some cases by forming labor unions.
The authors of this op-ed piece, a physician and a nurse, make a strong argument that the time has come for nurses and doctors shake off the shackles of their stereotypic roles and join in creating a loud, forceful, and effective voice to demand a working environment in which the computer functions as an asset and no longer as the terrible burden it has become. Neither group has the power to do it alone, but together they may be able to turn the tide. For physicians it will probably mean venturing several steps outside of their comfort zone. But working shoulder to shoulder with nurses may provide the courage to speak out.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
The time has come for good men and women to unite and rise up against a common foe. For too long nurses and doctors have labored under the tyranny of a dictator who claimed to help them provide high-quality care for their patients while at the same time cutting their paperwork to nil. But like most autocrats he failed to engage his subjects in a meaningful dialogue as each new version of his promised improvements rolled off the drawing board. When the caregivers were slow to adopt these new nonsystems he offered them financial incentives and issued threats to their survival. Although they were warned that there might be uncomfortable adjustment periods, the caregivers were promised that the steep learning curves would level out and their professional lives would again be valued and productive.
Of course, the dictator is not a single person but a motley and disorganized conglomerate of user- and patient-unfriendly electronic health record nonsystems. Ask almost any nurse or physician for her feelings about computer-based medical record systems, and you will hear tales of long hours, disengagement, and frustration. Caregivers are unhappy at all levels, and patients have grown tired of their nurses and physicians spending most of their time looking at computer screens.
You certainly have heard this all before. But you are hearing it in hospital hallways and grocery store checkout lines as a low rumble of discontent emerging from separate individuals, not as a well-articulated and widely distributed voice of physicians as a group. To some extent this relative silence is because there is no such group, at least not in same mold as a labor union. The term “labor union” may make you uncomfortable. But given the current climate in medicine, unionizing may be the best and only way to effect change.
But organizing to effect change in the workplace isn’t part of the physician genome. In the 1960s, a group of house officers in Boston engaged in a heal-in to successfully improve their salaries and working conditions. But over the ensuing half century physicians have remained tragically silent in the face of a changing workplace landscape in which they have gone from being independent owner operators in control of their destinies to becoming employees feeling powerless to improve their working conditions. This perceived impotence has escalated in the face of the challenge posed by the introduction of dysfunctional EHRs.
Ironically, a solution is at almost every physician’s elbow. In a recent New York Times opinion piece Theresa Brown and Stephen Bergman acknowledge that physicians don’t seem prepared to mount a meaningful response to the challenge to the failed promise of EHRs (“Doctors, Nurses and the Paperwork Crisis That Could Unite Them,” Dec. 31, 2019). They point out that, over the last half century, physicians have remained isolated on the sidelines, finding just enough voice to grumble. Nurses have in a variety of situations organized to effect change in their working conditions – in some cases by forming labor unions.
The authors of this op-ed piece, a physician and a nurse, make a strong argument that the time has come for nurses and doctors shake off the shackles of their stereotypic roles and join in creating a loud, forceful, and effective voice to demand a working environment in which the computer functions as an asset and no longer as the terrible burden it has become. Neither group has the power to do it alone, but together they may be able to turn the tide. For physicians it will probably mean venturing several steps outside of their comfort zone. But working shoulder to shoulder with nurses may provide the courage to speak out.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].












