User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
After Rapid Weight Loss, Monitor Antiobesity Drug Dosing
A patient who developed atrial fibrillation resulting from the failure to adjust the levothyroxine dose after rapid, significant weight loss while on the antiobesity drug tirzepatide (Zepbound) serves as a key reminder in managing patients experiencing rapid weight loss, either from antiobesity medications or any other means: Patients taking medications with weight-based dosing need to have their doses closely monitored.
“Failing to monitor and adjust dosing of these [and other] medications during a period of rapid weight loss may lead to supratherapeutic — even toxic — levels, as was seen in this [case],” underscore the authors of an editorial regarding the Teachable Moment case, published in JAMA Internal Medicine.
Toxicities from excessive doses can have a range of detrimental effects. In terms of thyroid medicine, the failure to adjust levothyroxine treatment for hypothyroidism in cases of rapid weight loss can lead to thyrotoxicosis, and in older patients in particular, a resulting thyrotropin level < 0.1 mIU/L is associated with as much as a threefold increased risk for atrial fibrillation, as observed in the report.
Case Demonstrates Risks
The case involved a 62-year-old man with obesity, hypothyroidism, and type 1 diabetes who presented to the emergency department with palpitations, excessive sweating, confusion, fever, and hand tremors. Upon being diagnosed with atrial fibrillation, the patient was immediately treated.
His medical history revealed the underlying culprit: Six months earlier, the patient had started treatment with the gastric inhibitory polypeptide (GIP)/glucagon-like peptide (GLP) 1 dual agonist tirzepatide. As is typical with the drug, the patient’s weight quickly plummeted, dropping from a starting body mass index of 44.4 down to 31.2 after 6 months and a decrease in body weight from 132 kg to 93 kg (a loss of 39 kg [approximately 86 lb]).
When he was prescribed tirzepatide, 2.5 mg weekly, for obesity, the patient had been recommended to increase the dose every 4 weeks as tolerated and, importantly, to have a follow-up visit in a month. But because he lived in different states seasonally, the follow-up never occurred.
Upon his emergency department visit, the patient’s thyrotropin level had dropped from 1.9 mIU/L at the first visit 6 months earlier to 0.001 mIU/L (well within the atrial fibrillation risk range), and his free thyroxine level (fT4) was 7.26 ng/ dL — substantially outside of the normal range of about 0.9-1.7 ng/dL for adults.
“The patient had 4-times higher fT4 levels of the upper limit,” first author Kagan E. Karakus, MD, of the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus, Aurora, told this news organization. “That is why he had experienced the adverse event of atrial fibrillation.”
Thyrotoxicosis Symptoms Can Be ‘Insidious,’ Levothyroxine Should Be Monitored
Although tirzepatide has not been approved by the US Food and Drug Administration for the treatment of type 1 diabetes, obesity is on the rise among patients with this disorder and recent research has shown a more than 10% reduction in body weight in 6 months and significant reductions in A1c with various doses.
Of note, in the current case, although the patient’s levothyroxine dose was not adjusted, his insulin dose was gradually self-decreased during his tirzepatide treatment to prevent hypoglycemia.
“If insulin treatment is excessive in diabetes, it causes hypoglycemia, [and] people with type 1 diabetes will recognize the signs of hypoglycemia related to excessive insulin earlier,” Dr. Karakus said.
If symptoms appear, patients can reduce their insulin doses on their own; however, the symptoms of thyrotoxicosis caused by excessive levothyroxine can be more insidious compared with hypoglycemia, he explained.
“Although patients can change their insulin doses, they cannot change the levothyroxine doses since it requires a blood test [thyroid-stimulating hormone; TSH] and a new prescription of the new dose.”
The key lesson is that “following levothyroxine treatment initiation or dose adjustment, 4-6 weeks is the optimal duration to recheck [the] thyrotropin level and adjust the dose as needed,” Dr. Karakus said.
Key Medications to Monitor
Other common outpatient medications that should be closely monitored in patients experiencing rapid weight loss, by any method, range from anticoagulants, anticonvulsants, and antituberculosis drugs to antibiotics and antifungals, the authors note.
Of note, medications with a narrow therapeutic index include phenytoin, warfarin, lithium carbonate, digoxin theophylline, tacrolimus, valproic acid, carbamazepine, and cyclosporine.
The failure to make necessary dose adjustments “is seen more often since the newer antiobesity drugs reduce a great amount of weight within months, almost as rapidly as bariatric surgery,” Dr. Karakus said.
“It is very important for physicians to be aware of the weight-based medications and narrow therapeutic index medications since their doses should be adjusted carefully, especially during weight loss,” he added.
Furthermore, “the patient should also know that weight reduction medication may cause adverse effects like nausea, vomiting and also may affect metabolism of other medications such that some medication doses should be adjusted regularly.”
In the editorial published with the study, Tyrone A. Johnson, MD, of the Department of Medicine, University of California, San Francisco, and colleagues note that the need for close monitoring is particularly important with older patients, who, in addition to having a higher likelihood of comorbidities, commonly have polypharmacy that could increase the potential for adverse effects.
Another key area concern is the emergence of direct-to-consumer avenues for GLP-1/GIP agonists for the many who either cannot afford or do not have access to the drugs, providing further opportunities for treatment without appropriate clinical oversight, they add.
Overall, the case “highlights the potential dangers underlying under-supervised prescribing of GLP-1/GIP receptor agonists and affirms the need for strong partnerships between patients and their clinicians during their use,” they wrote.
“These medications are best used in collaboration with continuity care teams, in context of a patient’s entire health, and in comprehensive risk-benefit assessment throughout the entire duration of treatment.”
A Caveat: Subclinical Levothyroxine Dosing
Commenting on the study, Matthew Ettleson, MD, a clinical instructor of medicine in the Section of Endocrinology, Diabetes, & Metabolism, University of Chicago, noted the important caveat that patients with hypothyroidism are commonly on subclinical doses, with varying dose adjustment needs.
“The patient in the case was clearly on a replacement level dose. However, many patients are on low doses of levothyroxine (75 µg or lower) for subclinical hypothyroidism, and, in general, I think the risks are lower with patients with subclinical hypothyroidism on lower doses of levothyroxine,” he told this news organization.
Because of that, “frequent TSH monitoring may be excessive in this population,” he said. “I would hesitate to empirically lower the dose with weight loss, unless it was clear that the patient was unlikely to follow up.
“Checking TSH at a more frequent interval and adjusting the dose accordingly should be adequate to prevent situations like this case.”
Dr. Karakus, Dr. Ettleson, and the editorial authors had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
A patient who developed atrial fibrillation resulting from the failure to adjust the levothyroxine dose after rapid, significant weight loss while on the antiobesity drug tirzepatide (Zepbound) serves as a key reminder in managing patients experiencing rapid weight loss, either from antiobesity medications or any other means: Patients taking medications with weight-based dosing need to have their doses closely monitored.
“Failing to monitor and adjust dosing of these [and other] medications during a period of rapid weight loss may lead to supratherapeutic — even toxic — levels, as was seen in this [case],” underscore the authors of an editorial regarding the Teachable Moment case, published in JAMA Internal Medicine.
Toxicities from excessive doses can have a range of detrimental effects. In terms of thyroid medicine, the failure to adjust levothyroxine treatment for hypothyroidism in cases of rapid weight loss can lead to thyrotoxicosis, and in older patients in particular, a resulting thyrotropin level < 0.1 mIU/L is associated with as much as a threefold increased risk for atrial fibrillation, as observed in the report.
Case Demonstrates Risks
The case involved a 62-year-old man with obesity, hypothyroidism, and type 1 diabetes who presented to the emergency department with palpitations, excessive sweating, confusion, fever, and hand tremors. Upon being diagnosed with atrial fibrillation, the patient was immediately treated.
His medical history revealed the underlying culprit: Six months earlier, the patient had started treatment with the gastric inhibitory polypeptide (GIP)/glucagon-like peptide (GLP) 1 dual agonist tirzepatide. As is typical with the drug, the patient’s weight quickly plummeted, dropping from a starting body mass index of 44.4 down to 31.2 after 6 months and a decrease in body weight from 132 kg to 93 kg (a loss of 39 kg [approximately 86 lb]).
When he was prescribed tirzepatide, 2.5 mg weekly, for obesity, the patient had been recommended to increase the dose every 4 weeks as tolerated and, importantly, to have a follow-up visit in a month. But because he lived in different states seasonally, the follow-up never occurred.
Upon his emergency department visit, the patient’s thyrotropin level had dropped from 1.9 mIU/L at the first visit 6 months earlier to 0.001 mIU/L (well within the atrial fibrillation risk range), and his free thyroxine level (fT4) was 7.26 ng/ dL — substantially outside of the normal range of about 0.9-1.7 ng/dL for adults.
“The patient had 4-times higher fT4 levels of the upper limit,” first author Kagan E. Karakus, MD, of the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus, Aurora, told this news organization. “That is why he had experienced the adverse event of atrial fibrillation.”
Thyrotoxicosis Symptoms Can Be ‘Insidious,’ Levothyroxine Should Be Monitored
Although tirzepatide has not been approved by the US Food and Drug Administration for the treatment of type 1 diabetes, obesity is on the rise among patients with this disorder and recent research has shown a more than 10% reduction in body weight in 6 months and significant reductions in A1c with various doses.
Of note, in the current case, although the patient’s levothyroxine dose was not adjusted, his insulin dose was gradually self-decreased during his tirzepatide treatment to prevent hypoglycemia.
“If insulin treatment is excessive in diabetes, it causes hypoglycemia, [and] people with type 1 diabetes will recognize the signs of hypoglycemia related to excessive insulin earlier,” Dr. Karakus said.
If symptoms appear, patients can reduce their insulin doses on their own; however, the symptoms of thyrotoxicosis caused by excessive levothyroxine can be more insidious compared with hypoglycemia, he explained.
“Although patients can change their insulin doses, they cannot change the levothyroxine doses since it requires a blood test [thyroid-stimulating hormone; TSH] and a new prescription of the new dose.”
The key lesson is that “following levothyroxine treatment initiation or dose adjustment, 4-6 weeks is the optimal duration to recheck [the] thyrotropin level and adjust the dose as needed,” Dr. Karakus said.
Key Medications to Monitor
Other common outpatient medications that should be closely monitored in patients experiencing rapid weight loss, by any method, range from anticoagulants, anticonvulsants, and antituberculosis drugs to antibiotics and antifungals, the authors note.
Of note, medications with a narrow therapeutic index include phenytoin, warfarin, lithium carbonate, digoxin theophylline, tacrolimus, valproic acid, carbamazepine, and cyclosporine.
The failure to make necessary dose adjustments “is seen more often since the newer antiobesity drugs reduce a great amount of weight within months, almost as rapidly as bariatric surgery,” Dr. Karakus said.
“It is very important for physicians to be aware of the weight-based medications and narrow therapeutic index medications since their doses should be adjusted carefully, especially during weight loss,” he added.
Furthermore, “the patient should also know that weight reduction medication may cause adverse effects like nausea, vomiting and also may affect metabolism of other medications such that some medication doses should be adjusted regularly.”
In the editorial published with the study, Tyrone A. Johnson, MD, of the Department of Medicine, University of California, San Francisco, and colleagues note that the need for close monitoring is particularly important with older patients, who, in addition to having a higher likelihood of comorbidities, commonly have polypharmacy that could increase the potential for adverse effects.
Another key area concern is the emergence of direct-to-consumer avenues for GLP-1/GIP agonists for the many who either cannot afford or do not have access to the drugs, providing further opportunities for treatment without appropriate clinical oversight, they add.
Overall, the case “highlights the potential dangers underlying under-supervised prescribing of GLP-1/GIP receptor agonists and affirms the need for strong partnerships between patients and their clinicians during their use,” they wrote.
“These medications are best used in collaboration with continuity care teams, in context of a patient’s entire health, and in comprehensive risk-benefit assessment throughout the entire duration of treatment.”
A Caveat: Subclinical Levothyroxine Dosing
Commenting on the study, Matthew Ettleson, MD, a clinical instructor of medicine in the Section of Endocrinology, Diabetes, & Metabolism, University of Chicago, noted the important caveat that patients with hypothyroidism are commonly on subclinical doses, with varying dose adjustment needs.
“The patient in the case was clearly on a replacement level dose. However, many patients are on low doses of levothyroxine (75 µg or lower) for subclinical hypothyroidism, and, in general, I think the risks are lower with patients with subclinical hypothyroidism on lower doses of levothyroxine,” he told this news organization.
Because of that, “frequent TSH monitoring may be excessive in this population,” he said. “I would hesitate to empirically lower the dose with weight loss, unless it was clear that the patient was unlikely to follow up.
“Checking TSH at a more frequent interval and adjusting the dose accordingly should be adequate to prevent situations like this case.”
Dr. Karakus, Dr. Ettleson, and the editorial authors had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
A patient who developed atrial fibrillation resulting from the failure to adjust the levothyroxine dose after rapid, significant weight loss while on the antiobesity drug tirzepatide (Zepbound) serves as a key reminder in managing patients experiencing rapid weight loss, either from antiobesity medications or any other means: Patients taking medications with weight-based dosing need to have their doses closely monitored.
“Failing to monitor and adjust dosing of these [and other] medications during a period of rapid weight loss may lead to supratherapeutic — even toxic — levels, as was seen in this [case],” underscore the authors of an editorial regarding the Teachable Moment case, published in JAMA Internal Medicine.
Toxicities from excessive doses can have a range of detrimental effects. In terms of thyroid medicine, the failure to adjust levothyroxine treatment for hypothyroidism in cases of rapid weight loss can lead to thyrotoxicosis, and in older patients in particular, a resulting thyrotropin level < 0.1 mIU/L is associated with as much as a threefold increased risk for atrial fibrillation, as observed in the report.
Case Demonstrates Risks
The case involved a 62-year-old man with obesity, hypothyroidism, and type 1 diabetes who presented to the emergency department with palpitations, excessive sweating, confusion, fever, and hand tremors. Upon being diagnosed with atrial fibrillation, the patient was immediately treated.
His medical history revealed the underlying culprit: Six months earlier, the patient had started treatment with the gastric inhibitory polypeptide (GIP)/glucagon-like peptide (GLP) 1 dual agonist tirzepatide. As is typical with the drug, the patient’s weight quickly plummeted, dropping from a starting body mass index of 44.4 down to 31.2 after 6 months and a decrease in body weight from 132 kg to 93 kg (a loss of 39 kg [approximately 86 lb]).
When he was prescribed tirzepatide, 2.5 mg weekly, for obesity, the patient had been recommended to increase the dose every 4 weeks as tolerated and, importantly, to have a follow-up visit in a month. But because he lived in different states seasonally, the follow-up never occurred.
Upon his emergency department visit, the patient’s thyrotropin level had dropped from 1.9 mIU/L at the first visit 6 months earlier to 0.001 mIU/L (well within the atrial fibrillation risk range), and his free thyroxine level (fT4) was 7.26 ng/ dL — substantially outside of the normal range of about 0.9-1.7 ng/dL for adults.
“The patient had 4-times higher fT4 levels of the upper limit,” first author Kagan E. Karakus, MD, of the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus, Aurora, told this news organization. “That is why he had experienced the adverse event of atrial fibrillation.”
Thyrotoxicosis Symptoms Can Be ‘Insidious,’ Levothyroxine Should Be Monitored
Although tirzepatide has not been approved by the US Food and Drug Administration for the treatment of type 1 diabetes, obesity is on the rise among patients with this disorder and recent research has shown a more than 10% reduction in body weight in 6 months and significant reductions in A1c with various doses.
Of note, in the current case, although the patient’s levothyroxine dose was not adjusted, his insulin dose was gradually self-decreased during his tirzepatide treatment to prevent hypoglycemia.
“If insulin treatment is excessive in diabetes, it causes hypoglycemia, [and] people with type 1 diabetes will recognize the signs of hypoglycemia related to excessive insulin earlier,” Dr. Karakus said.
If symptoms appear, patients can reduce their insulin doses on their own; however, the symptoms of thyrotoxicosis caused by excessive levothyroxine can be more insidious compared with hypoglycemia, he explained.
“Although patients can change their insulin doses, they cannot change the levothyroxine doses since it requires a blood test [thyroid-stimulating hormone; TSH] and a new prescription of the new dose.”
The key lesson is that “following levothyroxine treatment initiation or dose adjustment, 4-6 weeks is the optimal duration to recheck [the] thyrotropin level and adjust the dose as needed,” Dr. Karakus said.
Key Medications to Monitor
Other common outpatient medications that should be closely monitored in patients experiencing rapid weight loss, by any method, range from anticoagulants, anticonvulsants, and antituberculosis drugs to antibiotics and antifungals, the authors note.
Of note, medications with a narrow therapeutic index include phenytoin, warfarin, lithium carbonate, digoxin theophylline, tacrolimus, valproic acid, carbamazepine, and cyclosporine.
The failure to make necessary dose adjustments “is seen more often since the newer antiobesity drugs reduce a great amount of weight within months, almost as rapidly as bariatric surgery,” Dr. Karakus said.
“It is very important for physicians to be aware of the weight-based medications and narrow therapeutic index medications since their doses should be adjusted carefully, especially during weight loss,” he added.
Furthermore, “the patient should also know that weight reduction medication may cause adverse effects like nausea, vomiting and also may affect metabolism of other medications such that some medication doses should be adjusted regularly.”
In the editorial published with the study, Tyrone A. Johnson, MD, of the Department of Medicine, University of California, San Francisco, and colleagues note that the need for close monitoring is particularly important with older patients, who, in addition to having a higher likelihood of comorbidities, commonly have polypharmacy that could increase the potential for adverse effects.
Another key area concern is the emergence of direct-to-consumer avenues for GLP-1/GIP agonists for the many who either cannot afford or do not have access to the drugs, providing further opportunities for treatment without appropriate clinical oversight, they add.
Overall, the case “highlights the potential dangers underlying under-supervised prescribing of GLP-1/GIP receptor agonists and affirms the need for strong partnerships between patients and their clinicians during their use,” they wrote.
“These medications are best used in collaboration with continuity care teams, in context of a patient’s entire health, and in comprehensive risk-benefit assessment throughout the entire duration of treatment.”
A Caveat: Subclinical Levothyroxine Dosing
Commenting on the study, Matthew Ettleson, MD, a clinical instructor of medicine in the Section of Endocrinology, Diabetes, & Metabolism, University of Chicago, noted the important caveat that patients with hypothyroidism are commonly on subclinical doses, with varying dose adjustment needs.
“The patient in the case was clearly on a replacement level dose. However, many patients are on low doses of levothyroxine (75 µg or lower) for subclinical hypothyroidism, and, in general, I think the risks are lower with patients with subclinical hypothyroidism on lower doses of levothyroxine,” he told this news organization.
Because of that, “frequent TSH monitoring may be excessive in this population,” he said. “I would hesitate to empirically lower the dose with weight loss, unless it was clear that the patient was unlikely to follow up.
“Checking TSH at a more frequent interval and adjusting the dose accordingly should be adequate to prevent situations like this case.”
Dr. Karakus, Dr. Ettleson, and the editorial authors had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
1 in 4 Unresponsive Coma Patients May Retain Some Awareness
“We found that at least 1 in 4 patients who are unresponsive to commands might actually be quite present and highly cognitive,” said study investigator Nicholas D. Schiff, MD, Feil Family Brain & Mind Research Institute and Department of Neurology, Weill Cornell Medicine, Rockefeller University Hospital, New York.
“In other words, if you go to the bedside and carefully examine someone with a severe brain injury and find no evidence of responsiveness, no one has been able to give you an a priori number to say how likely you are to be wrong in thinking this person is actually unaware, not processing language, and not capable of high-level cognitive work. And the answer to that now is at least 1 in 4 times.”
The findings were published online in The New England Journal of Medicine.
Clinical Implications?
Cognitive motor dissociation (CMD) is a condition whereby patients with a severe brain injury who are unresponsive to commands at the bedside show brain activity on functional MRI (fMRI) or electroencephalography (EEG) when presented with selective motor imagery commands, such as “imagine playing tennis,” or “ imagine opening and closing your hand.”
Previous research shows that CMD is present in 10%-20% of people with a disorder of consciousness, a rate similar to that in patients with acute or chronic brain injury.
Understanding that a patient who appears unconscious has signs of cognitive processing could change the way clinicians and family interact with such individuals. Unresponsive patients who are aware may eventually be able to harness emerging communication technologies such as brain-computer interfaces.
In addition, knowing an individual’s CMD status could aid in prognosis. “We know from one study that there’s a four times increased likelihood that patients will be independent in a year in their function if they have cognitive motor dissociation,” said Dr. Schiff.
Unlike most previous studies of CMD, which were conducted at single sites and had relatively small cohorts, this new study included 353 adults with a disorder of consciousness (mean age, 37.9 years; 64% male) at six multinational sites.
Participants were recruited using a variety of methods, including consecutive enrollment of critically ill patients in the intensive care unit and enrollment of those with chronic illness or injury who were in the postacute phase of brain injury.
Response to Commands
Study participants were at different stages of recovery from an acute brain injury that had occurred an average of 8 months before the study started.
To determine the presence or absence of an observable response to commands among participants, trained staff used the Coma Recovery Scale–Revised (CRS-R); scores on this instrument range from 0 to 23, and higher scores indicate better neurobehavioral function.
About 40% of individuals were diagnosed with coma or vegetative state, 29% with minimally conscious state–minus, and 22% with minimally conscious state–plus. In all, 10% had emerged from a minimally conscious state.
Researchers assessed response to timed and repeated commands using fMRI or EEG in participants without an observable response to verbal commands, including those with a behavioral diagnosis of coma, vegetative state, or minimally conscious state–minus, and in participants with an observable response to verbal commands.
Of the 353 study participants, 61% underwent at least one fMRI assessment and 74% at least one EEG assessment. Both fMRI and EEG were performed in 35% of participants.
Dr. Schiff explained the two assessment types provide slightly different information, in that they measuring different types of brain signals. He also noted that although “every medical center in the world” has EEG, many do not have fMRI.
The brain imaging assessments captured brain activity within the motor area of the frontal cortex when tasked with motor imagery.
Of the 241 participants deemed to be in a coma or vegetative state or minimally conscious state–minus on the basis of CRS-R score, 60 (25%) had a response to commands on task-based fMRI, task-based EEG, or both.
The percentage of participants with CMD varied across study sites, from 2% to 45%, but Dr. Schiff said the reason for this is unclear.
The proportion of participants with CMD may have been even higher if all individuals had been assessed with both imaging techniques, he said.
Higher Rate of Awareness Than in Previous Research
The investigators noted that the percentage of participants with CMD in their study was up to 10 percentage points higher than in previous studies. This may be due to the multimodal approach that classified participants undergoing assessment with both fMRI and EEG on the basis of responses on either technique, they said.
The median age was lower among participants with CMD than those without CMD (30.5 years vs 45.3 years).
Compared with participants without CMD, a higher percentage of those with such dissociation had brain trauma as an etiologic factor (65% vs 38%) and a diagnosis of minimally conscious state–minus on the CRS-R (53% vs 38%).
Among people with CMD, 18% were assessed with fMRI only, 22% with EEG only, and 60% with both fMRI and EEG.
Dr. Schiff noted that the use of both fMRI and EEG appears to be more sensitive in detecting brain activity during tasks compared with use of one of these techniques alone.
Of the 112 participants with a diagnosis of minimally conscious state–plus or who had emerged from the minimally conscious state, 38% had a response to commands on task-based fMRI, task-based EEG, or both. Among these participants, 23% were assessed with fMRI only, 19% with EEG only, and 58% with both fMRI and EEG.
Research shows “it’s very clear that people with severe brain injury continue to get better over time,” noted Dr. Schiff. “Every month and week matters, and so it probably is the case that a lot of these patients are picking up the level of recovery, and the later we go out to measure them, the more likely we are to find people who are CMD than not.”
These new results should prompt further study to explore whether detection of CMD can lead to improved outcomes, the investigators noted. “In addition, the standardization, validation, and simplification of task-based fMRI and EEG methods that are used to detect cognitive motor dissociation are needed to prompt widespread clinical integration of these techniques and investigation of the bioethical implications of the findings.”
All study participants with chronic brain injury had survived their initial illness or injury and had access to a research facility with advanced fMRI and EEG capabilities. “This survival bias may reflect greater cognitive reserve and resilience over time among the participants. As such, the results of our study may not be generalizable to the overall population of patients with cognitive motor dissociation,” the investigators wrote.
Another study limitation was that participating sites used heterogeneous strategies to acquire, analyze, and interpret data, which led to differences in the number, type, and ordering of the cognitive tasks assessed on fMRI and EEG.
“These differences, along with variations in recruitment strategies and participant characteristics, may have contributed to the unequal percentage of participants with cognitive motor dissociation observed at each site. Our findings may therefore not be generalizable across all centers,” the researchers wrote.
Only a few academic medical centers have the specially trained personnel and techniques needed to assess patients for CMD — which, the researchers noted, limits the feasibility of performing these assessments in general practice.
Challenging Research
Commenting on the research, Aarti Sarwal, MD, professor of neurology and section chief, Neurocritical Care, Virginia Commonwealth University, Richmond, Virginia, noted that this was a “very challenging” study to perform, given that only a few academic centers are equipped to perform both fMRI and quantitative EEG analysis.
“In general, finding patients this far out, who have access to clinical, radiological, and electrophysiological testing and were provided good care enough to receive these, is a mammoth task in itself.”
Dr. Sarwal said the study builds on efforts of the Curing Coma campaign , a clinical, scientific, and public health effort of the Neurocritical Care Society to tackle the concept of coma as a treatable medical entity.
“It continues to highlight the challenges of prognostication in acute brain injured patients by showing a higher presence of cognitive function than previously perceived,” she said.
Dr. Sarwal believes that the study’s largest impact is underscoring the need for more research into understanding the degree and quality of cognitive processing in patients with a disorder of consciousness. But it also underlines the need for a “healthy debate” on the cost/benefit analysis of pursuing such research, given the limited number of patients with access to resources.
“This debate needs to include the caregivers and families outside the traditional realms of stakeholders overseeing the science.”
Although communication with comatose patients is still “a ways away,” this research is “a step in the right direction,” said Dr. Sarwal.
The study was funded by the James S. McDonnell Foundation and others. Dr. Schiff and Dr. Sarwal report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
“We found that at least 1 in 4 patients who are unresponsive to commands might actually be quite present and highly cognitive,” said study investigator Nicholas D. Schiff, MD, Feil Family Brain & Mind Research Institute and Department of Neurology, Weill Cornell Medicine, Rockefeller University Hospital, New York.
“In other words, if you go to the bedside and carefully examine someone with a severe brain injury and find no evidence of responsiveness, no one has been able to give you an a priori number to say how likely you are to be wrong in thinking this person is actually unaware, not processing language, and not capable of high-level cognitive work. And the answer to that now is at least 1 in 4 times.”
The findings were published online in The New England Journal of Medicine.
Clinical Implications?
Cognitive motor dissociation (CMD) is a condition whereby patients with a severe brain injury who are unresponsive to commands at the bedside show brain activity on functional MRI (fMRI) or electroencephalography (EEG) when presented with selective motor imagery commands, such as “imagine playing tennis,” or “ imagine opening and closing your hand.”
Previous research shows that CMD is present in 10%-20% of people with a disorder of consciousness, a rate similar to that in patients with acute or chronic brain injury.
Understanding that a patient who appears unconscious has signs of cognitive processing could change the way clinicians and family interact with such individuals. Unresponsive patients who are aware may eventually be able to harness emerging communication technologies such as brain-computer interfaces.
In addition, knowing an individual’s CMD status could aid in prognosis. “We know from one study that there’s a four times increased likelihood that patients will be independent in a year in their function if they have cognitive motor dissociation,” said Dr. Schiff.
Unlike most previous studies of CMD, which were conducted at single sites and had relatively small cohorts, this new study included 353 adults with a disorder of consciousness (mean age, 37.9 years; 64% male) at six multinational sites.
Participants were recruited using a variety of methods, including consecutive enrollment of critically ill patients in the intensive care unit and enrollment of those with chronic illness or injury who were in the postacute phase of brain injury.
Response to Commands
Study participants were at different stages of recovery from an acute brain injury that had occurred an average of 8 months before the study started.
To determine the presence or absence of an observable response to commands among participants, trained staff used the Coma Recovery Scale–Revised (CRS-R); scores on this instrument range from 0 to 23, and higher scores indicate better neurobehavioral function.
About 40% of individuals were diagnosed with coma or vegetative state, 29% with minimally conscious state–minus, and 22% with minimally conscious state–plus. In all, 10% had emerged from a minimally conscious state.
Researchers assessed response to timed and repeated commands using fMRI or EEG in participants without an observable response to verbal commands, including those with a behavioral diagnosis of coma, vegetative state, or minimally conscious state–minus, and in participants with an observable response to verbal commands.
Of the 353 study participants, 61% underwent at least one fMRI assessment and 74% at least one EEG assessment. Both fMRI and EEG were performed in 35% of participants.
Dr. Schiff explained the two assessment types provide slightly different information, in that they measuring different types of brain signals. He also noted that although “every medical center in the world” has EEG, many do not have fMRI.
The brain imaging assessments captured brain activity within the motor area of the frontal cortex when tasked with motor imagery.
Of the 241 participants deemed to be in a coma or vegetative state or minimally conscious state–minus on the basis of CRS-R score, 60 (25%) had a response to commands on task-based fMRI, task-based EEG, or both.
The percentage of participants with CMD varied across study sites, from 2% to 45%, but Dr. Schiff said the reason for this is unclear.
The proportion of participants with CMD may have been even higher if all individuals had been assessed with both imaging techniques, he said.
Higher Rate of Awareness Than in Previous Research
The investigators noted that the percentage of participants with CMD in their study was up to 10 percentage points higher than in previous studies. This may be due to the multimodal approach that classified participants undergoing assessment with both fMRI and EEG on the basis of responses on either technique, they said.
The median age was lower among participants with CMD than those without CMD (30.5 years vs 45.3 years).
Compared with participants without CMD, a higher percentage of those with such dissociation had brain trauma as an etiologic factor (65% vs 38%) and a diagnosis of minimally conscious state–minus on the CRS-R (53% vs 38%).
Among people with CMD, 18% were assessed with fMRI only, 22% with EEG only, and 60% with both fMRI and EEG.
Dr. Schiff noted that the use of both fMRI and EEG appears to be more sensitive in detecting brain activity during tasks compared with use of one of these techniques alone.
Of the 112 participants with a diagnosis of minimally conscious state–plus or who had emerged from the minimally conscious state, 38% had a response to commands on task-based fMRI, task-based EEG, or both. Among these participants, 23% were assessed with fMRI only, 19% with EEG only, and 58% with both fMRI and EEG.
Research shows “it’s very clear that people with severe brain injury continue to get better over time,” noted Dr. Schiff. “Every month and week matters, and so it probably is the case that a lot of these patients are picking up the level of recovery, and the later we go out to measure them, the more likely we are to find people who are CMD than not.”
These new results should prompt further study to explore whether detection of CMD can lead to improved outcomes, the investigators noted. “In addition, the standardization, validation, and simplification of task-based fMRI and EEG methods that are used to detect cognitive motor dissociation are needed to prompt widespread clinical integration of these techniques and investigation of the bioethical implications of the findings.”
All study participants with chronic brain injury had survived their initial illness or injury and had access to a research facility with advanced fMRI and EEG capabilities. “This survival bias may reflect greater cognitive reserve and resilience over time among the participants. As such, the results of our study may not be generalizable to the overall population of patients with cognitive motor dissociation,” the investigators wrote.
Another study limitation was that participating sites used heterogeneous strategies to acquire, analyze, and interpret data, which led to differences in the number, type, and ordering of the cognitive tasks assessed on fMRI and EEG.
“These differences, along with variations in recruitment strategies and participant characteristics, may have contributed to the unequal percentage of participants with cognitive motor dissociation observed at each site. Our findings may therefore not be generalizable across all centers,” the researchers wrote.
Only a few academic medical centers have the specially trained personnel and techniques needed to assess patients for CMD — which, the researchers noted, limits the feasibility of performing these assessments in general practice.
Challenging Research
Commenting on the research, Aarti Sarwal, MD, professor of neurology and section chief, Neurocritical Care, Virginia Commonwealth University, Richmond, Virginia, noted that this was a “very challenging” study to perform, given that only a few academic centers are equipped to perform both fMRI and quantitative EEG analysis.
“In general, finding patients this far out, who have access to clinical, radiological, and electrophysiological testing and were provided good care enough to receive these, is a mammoth task in itself.”
Dr. Sarwal said the study builds on efforts of the Curing Coma campaign , a clinical, scientific, and public health effort of the Neurocritical Care Society to tackle the concept of coma as a treatable medical entity.
“It continues to highlight the challenges of prognostication in acute brain injured patients by showing a higher presence of cognitive function than previously perceived,” she said.
Dr. Sarwal believes that the study’s largest impact is underscoring the need for more research into understanding the degree and quality of cognitive processing in patients with a disorder of consciousness. But it also underlines the need for a “healthy debate” on the cost/benefit analysis of pursuing such research, given the limited number of patients with access to resources.
“This debate needs to include the caregivers and families outside the traditional realms of stakeholders overseeing the science.”
Although communication with comatose patients is still “a ways away,” this research is “a step in the right direction,” said Dr. Sarwal.
The study was funded by the James S. McDonnell Foundation and others. Dr. Schiff and Dr. Sarwal report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
“We found that at least 1 in 4 patients who are unresponsive to commands might actually be quite present and highly cognitive,” said study investigator Nicholas D. Schiff, MD, Feil Family Brain & Mind Research Institute and Department of Neurology, Weill Cornell Medicine, Rockefeller University Hospital, New York.
“In other words, if you go to the bedside and carefully examine someone with a severe brain injury and find no evidence of responsiveness, no one has been able to give you an a priori number to say how likely you are to be wrong in thinking this person is actually unaware, not processing language, and not capable of high-level cognitive work. And the answer to that now is at least 1 in 4 times.”
The findings were published online in The New England Journal of Medicine.
Clinical Implications?
Cognitive motor dissociation (CMD) is a condition whereby patients with a severe brain injury who are unresponsive to commands at the bedside show brain activity on functional MRI (fMRI) or electroencephalography (EEG) when presented with selective motor imagery commands, such as “imagine playing tennis,” or “ imagine opening and closing your hand.”
Previous research shows that CMD is present in 10%-20% of people with a disorder of consciousness, a rate similar to that in patients with acute or chronic brain injury.
Understanding that a patient who appears unconscious has signs of cognitive processing could change the way clinicians and family interact with such individuals. Unresponsive patients who are aware may eventually be able to harness emerging communication technologies such as brain-computer interfaces.
In addition, knowing an individual’s CMD status could aid in prognosis. “We know from one study that there’s a four times increased likelihood that patients will be independent in a year in their function if they have cognitive motor dissociation,” said Dr. Schiff.
Unlike most previous studies of CMD, which were conducted at single sites and had relatively small cohorts, this new study included 353 adults with a disorder of consciousness (mean age, 37.9 years; 64% male) at six multinational sites.
Participants were recruited using a variety of methods, including consecutive enrollment of critically ill patients in the intensive care unit and enrollment of those with chronic illness or injury who were in the postacute phase of brain injury.
Response to Commands
Study participants were at different stages of recovery from an acute brain injury that had occurred an average of 8 months before the study started.
To determine the presence or absence of an observable response to commands among participants, trained staff used the Coma Recovery Scale–Revised (CRS-R); scores on this instrument range from 0 to 23, and higher scores indicate better neurobehavioral function.
About 40% of individuals were diagnosed with coma or vegetative state, 29% with minimally conscious state–minus, and 22% with minimally conscious state–plus. In all, 10% had emerged from a minimally conscious state.
Researchers assessed response to timed and repeated commands using fMRI or EEG in participants without an observable response to verbal commands, including those with a behavioral diagnosis of coma, vegetative state, or minimally conscious state–minus, and in participants with an observable response to verbal commands.
Of the 353 study participants, 61% underwent at least one fMRI assessment and 74% at least one EEG assessment. Both fMRI and EEG were performed in 35% of participants.
Dr. Schiff explained the two assessment types provide slightly different information, in that they measuring different types of brain signals. He also noted that although “every medical center in the world” has EEG, many do not have fMRI.
The brain imaging assessments captured brain activity within the motor area of the frontal cortex when tasked with motor imagery.
Of the 241 participants deemed to be in a coma or vegetative state or minimally conscious state–minus on the basis of CRS-R score, 60 (25%) had a response to commands on task-based fMRI, task-based EEG, or both.
The percentage of participants with CMD varied across study sites, from 2% to 45%, but Dr. Schiff said the reason for this is unclear.
The proportion of participants with CMD may have been even higher if all individuals had been assessed with both imaging techniques, he said.
Higher Rate of Awareness Than in Previous Research
The investigators noted that the percentage of participants with CMD in their study was up to 10 percentage points higher than in previous studies. This may be due to the multimodal approach that classified participants undergoing assessment with both fMRI and EEG on the basis of responses on either technique, they said.
The median age was lower among participants with CMD than those without CMD (30.5 years vs 45.3 years).
Compared with participants without CMD, a higher percentage of those with such dissociation had brain trauma as an etiologic factor (65% vs 38%) and a diagnosis of minimally conscious state–minus on the CRS-R (53% vs 38%).
Among people with CMD, 18% were assessed with fMRI only, 22% with EEG only, and 60% with both fMRI and EEG.
Dr. Schiff noted that the use of both fMRI and EEG appears to be more sensitive in detecting brain activity during tasks compared with use of one of these techniques alone.
Of the 112 participants with a diagnosis of minimally conscious state–plus or who had emerged from the minimally conscious state, 38% had a response to commands on task-based fMRI, task-based EEG, or both. Among these participants, 23% were assessed with fMRI only, 19% with EEG only, and 58% with both fMRI and EEG.
Research shows “it’s very clear that people with severe brain injury continue to get better over time,” noted Dr. Schiff. “Every month and week matters, and so it probably is the case that a lot of these patients are picking up the level of recovery, and the later we go out to measure them, the more likely we are to find people who are CMD than not.”
These new results should prompt further study to explore whether detection of CMD can lead to improved outcomes, the investigators noted. “In addition, the standardization, validation, and simplification of task-based fMRI and EEG methods that are used to detect cognitive motor dissociation are needed to prompt widespread clinical integration of these techniques and investigation of the bioethical implications of the findings.”
All study participants with chronic brain injury had survived their initial illness or injury and had access to a research facility with advanced fMRI and EEG capabilities. “This survival bias may reflect greater cognitive reserve and resilience over time among the participants. As such, the results of our study may not be generalizable to the overall population of patients with cognitive motor dissociation,” the investigators wrote.
Another study limitation was that participating sites used heterogeneous strategies to acquire, analyze, and interpret data, which led to differences in the number, type, and ordering of the cognitive tasks assessed on fMRI and EEG.
“These differences, along with variations in recruitment strategies and participant characteristics, may have contributed to the unequal percentage of participants with cognitive motor dissociation observed at each site. Our findings may therefore not be generalizable across all centers,” the researchers wrote.
Only a few academic medical centers have the specially trained personnel and techniques needed to assess patients for CMD — which, the researchers noted, limits the feasibility of performing these assessments in general practice.
Challenging Research
Commenting on the research, Aarti Sarwal, MD, professor of neurology and section chief, Neurocritical Care, Virginia Commonwealth University, Richmond, Virginia, noted that this was a “very challenging” study to perform, given that only a few academic centers are equipped to perform both fMRI and quantitative EEG analysis.
“In general, finding patients this far out, who have access to clinical, radiological, and electrophysiological testing and were provided good care enough to receive these, is a mammoth task in itself.”
Dr. Sarwal said the study builds on efforts of the Curing Coma campaign , a clinical, scientific, and public health effort of the Neurocritical Care Society to tackle the concept of coma as a treatable medical entity.
“It continues to highlight the challenges of prognostication in acute brain injured patients by showing a higher presence of cognitive function than previously perceived,” she said.
Dr. Sarwal believes that the study’s largest impact is underscoring the need for more research into understanding the degree and quality of cognitive processing in patients with a disorder of consciousness. But it also underlines the need for a “healthy debate” on the cost/benefit analysis of pursuing such research, given the limited number of patients with access to resources.
“This debate needs to include the caregivers and families outside the traditional realms of stakeholders overseeing the science.”
Although communication with comatose patients is still “a ways away,” this research is “a step in the right direction,” said Dr. Sarwal.
The study was funded by the James S. McDonnell Foundation and others. Dr. Schiff and Dr. Sarwal report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
FTC Interim Report on Pharmacy Middlemen Is First Step of Many Needed in Addressing Drug Costs, Access
Rising consolidation among pharmacy benefit managers (PBMs) allows the companies to profit at the expense of patients and independent pharmacists. That’s the conclusion of a recent Federal Trade Commission (FTC) report on interim findings from the agency’s ongoing investigation of PBMs.
Lawmakers are increasingly scrutinizing the industry amid growing concern among physicians and consumers about how PBMs exploit their market dominance. The top six PBMs managed 94% of US drug claims in 2023, with the majority handled by the industry’s three giants: CVS Caremark, Cigna’s Express Scripts, and United Healthcare’s OptumRx.
PBMs manage prescription drug benefits for health insurers, Medicare Part D drug plans, and large employers. They act as middlemen between health insurers and pharmacies, developing formularies of covered drugs and promising savings from the discounts and rebates they negotiate with drugmakers.
The FTC’s interim report found that the giant PBMs often exercise significant control over what drugs are available and at what price and which pharmacies patients can use to access their prescribed medications. Consumers suffer as a result, the report concluded.
Madelaine A. Feldman, MD, vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations, shared her perspective on the FTC report in an email Q&A with this news organization. She is affiliated with The Rheumatology Group, based in Metairie, Louisiana.
Dr. Feldman has long tracked the PBM industry and appeared as a witness before influential government panels, including the House Energy and Commerce Committee. She has highlighted for lawmakers the challenges physicians face in helping patients get needed medicines.
For example, she shared cases of PBMs steering patients toward the more expensive of three widely used rheumatoid arthritis medicines that have a similar mechanism of action, the Janus kinase (JAK) inhibitors, Dr. Feldman said.
One of the drugs cost roughly half of the other two — about $30,000 per year vs $65,000-$70,000. Yet only the two expensive drugs were included in the PBM formulary. As a result, the cheapest drug holds only a sliver of market share; the remainder is dominated by the two expensive products, she told the House Oversight and Accountability Committee in 2021.
This Q&A has been edited for length and clarity.
What would you want federal and state policymakers to do in response to the FTC’s report?
I think Congress needs to clearly delineate the differences between anticompetitive pharmacy issues, drug pricing issues, and their effect on formulary construction issues.
Lawmakers should demand more transparency and consider legislation that would remove perverse incentives that prompt PBMs to choose higher priced drugs for their formularies.
That may require other regulatory or legislative actions to ensure lower prices (not higher kickbacks) are incentivized. Ultimately, in order to gain true competition within the health insurance business, these oligopolies of multiple businesses need to be broken up. Anything less seems to be nibbling around the edges and allows the Big Three to continue their “whack-a mole” in circumventing piecemeal regulatory and legislative policies.
You’ve followed PBM practices closely for many years. Was there anything in this interim FTC report that surprised you?
Though not surprised, I am glad that it was released because it had been a year in investigation and there were many requests for some type of substantive report.
Two things that are missing that I feel are paramount are investigating how the three big PBMs are causing physical harm to patients as a result of the profit component in formulary construction and the profound financial impact of hidden PBM profit centers in self-insured employer health plans.
What we have seen over the years is the result of the perverse incentives for the PBMs to prefer the most profitable medications on their formularies.
They use utilization management tools such as step therapy, nonmedical switching, and exclusions to maintain their formularies’ profitability. These tools have been shown to delay and deny the proper care of patients, resulting in not just monetary but physical harm as well.
I would think the physical harm done to patients in manipulating the formularies should be addressed in this report as well and, in fact, may be the most important aspect of consumer protection of this issue.
In terms of the FTC’s mission to not “unduly burden” legitimate business, I would like to see the sector of self-insured employers addressed.
The report details how PBMs steer prescriptions to their affiliated pharmacies. The FTC says that can push smaller pharmacies out of the market, ultimately leading to higher costs and lower quality services for people. What’s your perspective?
Having more community pharmacies is better than having less. We are seeing more “pharmacy deserts” in rural areas as a result of many community pharmacies having to close.
The FTC voted 4-1 to allow staff to issue the interim report, with Commissioner Melissa Holyoak voting no. And some FTC commissioners seem divided on the usefulness of the report. Why?
Commissioner Holyoak states the “the Report leaves us without a better understanding of the competition concerns surrounding PBMs or how consumers are impacted by PBM practices.”
I do agree with her that the harm to patients’ medical status was not even addressed as far as I could tell in this report. There are multiple news articles and reports on the harms inflicted upon patients by the UM tools that drive the construction of ever changing formularies, all based on contracting with manufacturers that result in the highest profit for the PBM.
Holyoak also states, “Among other critical conclusions, the Report does not address the seemingly contradictory conclusions in the 2005 Report that PBMs, including vertically owned PBMs, generated cost savings for consumers.”
That may be true, but in 2005, the rise of PBMs was just beginning and the huge vertical and horizontal integration had yet to begin. Also, 2005 was still in the beginning of the biologic drug deluge, which did create competition to get on the formulary. Since then, PBMs have done nothing to control the rise in prices but instead, apparently have used the competition to get higher price concessions from manufacturers based on a percentage of the list price to line their pockets.
Commissioner Ferguson agreed with releasing the report but he had many issues with this report including the lack of PBM response.
I do agree with him that the FTC should have used some type of “force” to get the information they needed from the PBMs. The Big Three are known for obfuscation and delaying providing information to legislative and regulatory agencies.
A version of this article appeared on Medscape.com.
Rising consolidation among pharmacy benefit managers (PBMs) allows the companies to profit at the expense of patients and independent pharmacists. That’s the conclusion of a recent Federal Trade Commission (FTC) report on interim findings from the agency’s ongoing investigation of PBMs.
Lawmakers are increasingly scrutinizing the industry amid growing concern among physicians and consumers about how PBMs exploit their market dominance. The top six PBMs managed 94% of US drug claims in 2023, with the majority handled by the industry’s three giants: CVS Caremark, Cigna’s Express Scripts, and United Healthcare’s OptumRx.
PBMs manage prescription drug benefits for health insurers, Medicare Part D drug plans, and large employers. They act as middlemen between health insurers and pharmacies, developing formularies of covered drugs and promising savings from the discounts and rebates they negotiate with drugmakers.
The FTC’s interim report found that the giant PBMs often exercise significant control over what drugs are available and at what price and which pharmacies patients can use to access their prescribed medications. Consumers suffer as a result, the report concluded.
Madelaine A. Feldman, MD, vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations, shared her perspective on the FTC report in an email Q&A with this news organization. She is affiliated with The Rheumatology Group, based in Metairie, Louisiana.
Dr. Feldman has long tracked the PBM industry and appeared as a witness before influential government panels, including the House Energy and Commerce Committee. She has highlighted for lawmakers the challenges physicians face in helping patients get needed medicines.
For example, she shared cases of PBMs steering patients toward the more expensive of three widely used rheumatoid arthritis medicines that have a similar mechanism of action, the Janus kinase (JAK) inhibitors, Dr. Feldman said.
One of the drugs cost roughly half of the other two — about $30,000 per year vs $65,000-$70,000. Yet only the two expensive drugs were included in the PBM formulary. As a result, the cheapest drug holds only a sliver of market share; the remainder is dominated by the two expensive products, she told the House Oversight and Accountability Committee in 2021.
This Q&A has been edited for length and clarity.
What would you want federal and state policymakers to do in response to the FTC’s report?
I think Congress needs to clearly delineate the differences between anticompetitive pharmacy issues, drug pricing issues, and their effect on formulary construction issues.
Lawmakers should demand more transparency and consider legislation that would remove perverse incentives that prompt PBMs to choose higher priced drugs for their formularies.
That may require other regulatory or legislative actions to ensure lower prices (not higher kickbacks) are incentivized. Ultimately, in order to gain true competition within the health insurance business, these oligopolies of multiple businesses need to be broken up. Anything less seems to be nibbling around the edges and allows the Big Three to continue their “whack-a mole” in circumventing piecemeal regulatory and legislative policies.
You’ve followed PBM practices closely for many years. Was there anything in this interim FTC report that surprised you?
Though not surprised, I am glad that it was released because it had been a year in investigation and there were many requests for some type of substantive report.
Two things that are missing that I feel are paramount are investigating how the three big PBMs are causing physical harm to patients as a result of the profit component in formulary construction and the profound financial impact of hidden PBM profit centers in self-insured employer health plans.
What we have seen over the years is the result of the perverse incentives for the PBMs to prefer the most profitable medications on their formularies.
They use utilization management tools such as step therapy, nonmedical switching, and exclusions to maintain their formularies’ profitability. These tools have been shown to delay and deny the proper care of patients, resulting in not just monetary but physical harm as well.
I would think the physical harm done to patients in manipulating the formularies should be addressed in this report as well and, in fact, may be the most important aspect of consumer protection of this issue.
In terms of the FTC’s mission to not “unduly burden” legitimate business, I would like to see the sector of self-insured employers addressed.
The report details how PBMs steer prescriptions to their affiliated pharmacies. The FTC says that can push smaller pharmacies out of the market, ultimately leading to higher costs and lower quality services for people. What’s your perspective?
Having more community pharmacies is better than having less. We are seeing more “pharmacy deserts” in rural areas as a result of many community pharmacies having to close.
The FTC voted 4-1 to allow staff to issue the interim report, with Commissioner Melissa Holyoak voting no. And some FTC commissioners seem divided on the usefulness of the report. Why?
Commissioner Holyoak states the “the Report leaves us without a better understanding of the competition concerns surrounding PBMs or how consumers are impacted by PBM practices.”
I do agree with her that the harm to patients’ medical status was not even addressed as far as I could tell in this report. There are multiple news articles and reports on the harms inflicted upon patients by the UM tools that drive the construction of ever changing formularies, all based on contracting with manufacturers that result in the highest profit for the PBM.
Holyoak also states, “Among other critical conclusions, the Report does not address the seemingly contradictory conclusions in the 2005 Report that PBMs, including vertically owned PBMs, generated cost savings for consumers.”
That may be true, but in 2005, the rise of PBMs was just beginning and the huge vertical and horizontal integration had yet to begin. Also, 2005 was still in the beginning of the biologic drug deluge, which did create competition to get on the formulary. Since then, PBMs have done nothing to control the rise in prices but instead, apparently have used the competition to get higher price concessions from manufacturers based on a percentage of the list price to line their pockets.
Commissioner Ferguson agreed with releasing the report but he had many issues with this report including the lack of PBM response.
I do agree with him that the FTC should have used some type of “force” to get the information they needed from the PBMs. The Big Three are known for obfuscation and delaying providing information to legislative and regulatory agencies.
A version of this article appeared on Medscape.com.
Rising consolidation among pharmacy benefit managers (PBMs) allows the companies to profit at the expense of patients and independent pharmacists. That’s the conclusion of a recent Federal Trade Commission (FTC) report on interim findings from the agency’s ongoing investigation of PBMs.
Lawmakers are increasingly scrutinizing the industry amid growing concern among physicians and consumers about how PBMs exploit their market dominance. The top six PBMs managed 94% of US drug claims in 2023, with the majority handled by the industry’s three giants: CVS Caremark, Cigna’s Express Scripts, and United Healthcare’s OptumRx.
PBMs manage prescription drug benefits for health insurers, Medicare Part D drug plans, and large employers. They act as middlemen between health insurers and pharmacies, developing formularies of covered drugs and promising savings from the discounts and rebates they negotiate with drugmakers.
The FTC’s interim report found that the giant PBMs often exercise significant control over what drugs are available and at what price and which pharmacies patients can use to access their prescribed medications. Consumers suffer as a result, the report concluded.
Madelaine A. Feldman, MD, vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations, shared her perspective on the FTC report in an email Q&A with this news organization. She is affiliated with The Rheumatology Group, based in Metairie, Louisiana.
Dr. Feldman has long tracked the PBM industry and appeared as a witness before influential government panels, including the House Energy and Commerce Committee. She has highlighted for lawmakers the challenges physicians face in helping patients get needed medicines.
For example, she shared cases of PBMs steering patients toward the more expensive of three widely used rheumatoid arthritis medicines that have a similar mechanism of action, the Janus kinase (JAK) inhibitors, Dr. Feldman said.
One of the drugs cost roughly half of the other two — about $30,000 per year vs $65,000-$70,000. Yet only the two expensive drugs were included in the PBM formulary. As a result, the cheapest drug holds only a sliver of market share; the remainder is dominated by the two expensive products, she told the House Oversight and Accountability Committee in 2021.
This Q&A has been edited for length and clarity.
What would you want federal and state policymakers to do in response to the FTC’s report?
I think Congress needs to clearly delineate the differences between anticompetitive pharmacy issues, drug pricing issues, and their effect on formulary construction issues.
Lawmakers should demand more transparency and consider legislation that would remove perverse incentives that prompt PBMs to choose higher priced drugs for their formularies.
That may require other regulatory or legislative actions to ensure lower prices (not higher kickbacks) are incentivized. Ultimately, in order to gain true competition within the health insurance business, these oligopolies of multiple businesses need to be broken up. Anything less seems to be nibbling around the edges and allows the Big Three to continue their “whack-a mole” in circumventing piecemeal regulatory and legislative policies.
You’ve followed PBM practices closely for many years. Was there anything in this interim FTC report that surprised you?
Though not surprised, I am glad that it was released because it had been a year in investigation and there were many requests for some type of substantive report.
Two things that are missing that I feel are paramount are investigating how the three big PBMs are causing physical harm to patients as a result of the profit component in formulary construction and the profound financial impact of hidden PBM profit centers in self-insured employer health plans.
What we have seen over the years is the result of the perverse incentives for the PBMs to prefer the most profitable medications on their formularies.
They use utilization management tools such as step therapy, nonmedical switching, and exclusions to maintain their formularies’ profitability. These tools have been shown to delay and deny the proper care of patients, resulting in not just monetary but physical harm as well.
I would think the physical harm done to patients in manipulating the formularies should be addressed in this report as well and, in fact, may be the most important aspect of consumer protection of this issue.
In terms of the FTC’s mission to not “unduly burden” legitimate business, I would like to see the sector of self-insured employers addressed.
The report details how PBMs steer prescriptions to their affiliated pharmacies. The FTC says that can push smaller pharmacies out of the market, ultimately leading to higher costs and lower quality services for people. What’s your perspective?
Having more community pharmacies is better than having less. We are seeing more “pharmacy deserts” in rural areas as a result of many community pharmacies having to close.
The FTC voted 4-1 to allow staff to issue the interim report, with Commissioner Melissa Holyoak voting no. And some FTC commissioners seem divided on the usefulness of the report. Why?
Commissioner Holyoak states the “the Report leaves us without a better understanding of the competition concerns surrounding PBMs or how consumers are impacted by PBM practices.”
I do agree with her that the harm to patients’ medical status was not even addressed as far as I could tell in this report. There are multiple news articles and reports on the harms inflicted upon patients by the UM tools that drive the construction of ever changing formularies, all based on contracting with manufacturers that result in the highest profit for the PBM.
Holyoak also states, “Among other critical conclusions, the Report does not address the seemingly contradictory conclusions in the 2005 Report that PBMs, including vertically owned PBMs, generated cost savings for consumers.”
That may be true, but in 2005, the rise of PBMs was just beginning and the huge vertical and horizontal integration had yet to begin. Also, 2005 was still in the beginning of the biologic drug deluge, which did create competition to get on the formulary. Since then, PBMs have done nothing to control the rise in prices but instead, apparently have used the competition to get higher price concessions from manufacturers based on a percentage of the list price to line their pockets.
Commissioner Ferguson agreed with releasing the report but he had many issues with this report including the lack of PBM response.
I do agree with him that the FTC should have used some type of “force” to get the information they needed from the PBMs. The Big Three are known for obfuscation and delaying providing information to legislative and regulatory agencies.
A version of this article appeared on Medscape.com.
Could Targeting ‘Zombie Cells’ Extend a Healthy Lifespan?
What if a drug could help you live a longer, healthier life?
Scientists at the University of Connecticut are working on it. In a new study in Cell Metabolism, researchers described how to target specific cells to extend the lifespan and improve the health of mice late in life.
The study builds on a growing body of research, mostly in animals, testing interventions to slow aging and prolong health span, the length of time that one is not just alive but also healthy.
“Aging is the most important risk factor for every disease that we deal with in adult human beings,” said cardiologist Douglas Vaughan, MD, director of the Potocsnak Longevity Institute at Northwestern University’s Feinberg School of Medicine, Chicago. (Dr. Vaughan was not involved in the new study.) “So the big hypothesis is: If we could slow down aging just a little bit, we can push back the onset of disease.”
Senescent cells — or “zombie cells” — secrete harmful substances that disrupt tissue functioning. They’ve been linked to chronic inflammation, tissue damage, and the development of age-related diseases.
Senescence can be characterized by the accumulation of cells with high levels of specific markers like p21, or p21high cells. Almost any cell can become a p21high cell, and they accumulate with age, said Ming Xu, PhD, a professor at the UConn Center on Aging, UConn Health, Farmington, Connecticut, who led the study.
By targeting and eliminating p21high senescent cells, Dr. Xu hopes to develop novel therapies that might help people live longer and enjoy more years in good health.
Such a treatment could be ready for human trials in 2-5 years, Dr. Xu said.
What the Researchers Did
Xu and colleagues used genetic engineering to eliminate p21high cells in mice, introducing into their genome something they describe as an inducible “suicide gene.” Giving the mice a certain drug (a low dose of tamoxifen) activated the suicide gene in all p21high cells, causing them to die. Administering this treatment once a month, from age 20 months (older age) until the end of life, significantly extended the rodents’ lifespan, reduced inflammation, and decreased gene activity linked to aging.
Treated mice lived, on average, for 33 months — 3 months longer than the untreated mice. The oldest treated mouse lived to 43 months — roughly 130 in human years.
But the treated mice didn’t just live longer; they were also healthier. In humans, walking speed and grip strength can be clues of overall health and vitality. The old, treated mice were able to walk faster and grip objects with greater strength than untreated mice of the same age.
Dr. Xu’s lab is now testing drugs that target p21high cells in hopes of finding one that would work in humans. Leveraging immunotherapy technology to target these cells could be another option, Dr. Xu said.
The team also plans to test whether eliminating p21high cells could prevent or alleviate diabetes or Alzheimer’s disease.
Challenges and Criticisms
The research provides “important evidence that targeting senescence and the molecular components of that pathway might provide some benefit in the long term,” Dr. Vaughan said.
But killing senescent cells could come with downsides.
“Senescence protects us from hyperproliferative responses,” potentially blocking cells from becoming malignant, Dr. Vaughan said. “There’s this effect on aging that is desirable, but at the same time, you may enhance your risk of cancer or malignancy or excessive proliferation in some cells.”
And of course, we don’t necessarily need drugs to prolong healthy life, Dr. Vaughan pointed out.
For many people, a long healthy life is already within reach. Humans live longer on average than they used to, and simple lifestyle choices — nourishing your body well, staying active, and maintaining a healthy weight — can increase one’s chances of good health.
The most consistently demonstrated intervention for extending lifespan “in almost every animal species is caloric restriction,” Dr. Vaughan said. (Dr. Xu’s team is also investigating whether fasting and exercise can lead to a decrease in p21high cells.)
As for brain health, Dr. Vaughan and colleagues at Northwestern are studying “super agers,” people who are cognitively intact into their 90s.
“The one single thing that they found that contributes to that process, and contributes to that success, is really a social network and human bonds and interaction,” Dr. Vaughan said.
A version of this article appeared on Medscape.com.
What if a drug could help you live a longer, healthier life?
Scientists at the University of Connecticut are working on it. In a new study in Cell Metabolism, researchers described how to target specific cells to extend the lifespan and improve the health of mice late in life.
The study builds on a growing body of research, mostly in animals, testing interventions to slow aging and prolong health span, the length of time that one is not just alive but also healthy.
“Aging is the most important risk factor for every disease that we deal with in adult human beings,” said cardiologist Douglas Vaughan, MD, director of the Potocsnak Longevity Institute at Northwestern University’s Feinberg School of Medicine, Chicago. (Dr. Vaughan was not involved in the new study.) “So the big hypothesis is: If we could slow down aging just a little bit, we can push back the onset of disease.”
Senescent cells — or “zombie cells” — secrete harmful substances that disrupt tissue functioning. They’ve been linked to chronic inflammation, tissue damage, and the development of age-related diseases.
Senescence can be characterized by the accumulation of cells with high levels of specific markers like p21, or p21high cells. Almost any cell can become a p21high cell, and they accumulate with age, said Ming Xu, PhD, a professor at the UConn Center on Aging, UConn Health, Farmington, Connecticut, who led the study.
By targeting and eliminating p21high senescent cells, Dr. Xu hopes to develop novel therapies that might help people live longer and enjoy more years in good health.
Such a treatment could be ready for human trials in 2-5 years, Dr. Xu said.
What the Researchers Did
Xu and colleagues used genetic engineering to eliminate p21high cells in mice, introducing into their genome something they describe as an inducible “suicide gene.” Giving the mice a certain drug (a low dose of tamoxifen) activated the suicide gene in all p21high cells, causing them to die. Administering this treatment once a month, from age 20 months (older age) until the end of life, significantly extended the rodents’ lifespan, reduced inflammation, and decreased gene activity linked to aging.
Treated mice lived, on average, for 33 months — 3 months longer than the untreated mice. The oldest treated mouse lived to 43 months — roughly 130 in human years.
But the treated mice didn’t just live longer; they were also healthier. In humans, walking speed and grip strength can be clues of overall health and vitality. The old, treated mice were able to walk faster and grip objects with greater strength than untreated mice of the same age.
Dr. Xu’s lab is now testing drugs that target p21high cells in hopes of finding one that would work in humans. Leveraging immunotherapy technology to target these cells could be another option, Dr. Xu said.
The team also plans to test whether eliminating p21high cells could prevent or alleviate diabetes or Alzheimer’s disease.
Challenges and Criticisms
The research provides “important evidence that targeting senescence and the molecular components of that pathway might provide some benefit in the long term,” Dr. Vaughan said.
But killing senescent cells could come with downsides.
“Senescence protects us from hyperproliferative responses,” potentially blocking cells from becoming malignant, Dr. Vaughan said. “There’s this effect on aging that is desirable, but at the same time, you may enhance your risk of cancer or malignancy or excessive proliferation in some cells.”
And of course, we don’t necessarily need drugs to prolong healthy life, Dr. Vaughan pointed out.
For many people, a long healthy life is already within reach. Humans live longer on average than they used to, and simple lifestyle choices — nourishing your body well, staying active, and maintaining a healthy weight — can increase one’s chances of good health.
The most consistently demonstrated intervention for extending lifespan “in almost every animal species is caloric restriction,” Dr. Vaughan said. (Dr. Xu’s team is also investigating whether fasting and exercise can lead to a decrease in p21high cells.)
As for brain health, Dr. Vaughan and colleagues at Northwestern are studying “super agers,” people who are cognitively intact into their 90s.
“The one single thing that they found that contributes to that process, and contributes to that success, is really a social network and human bonds and interaction,” Dr. Vaughan said.
A version of this article appeared on Medscape.com.
What if a drug could help you live a longer, healthier life?
Scientists at the University of Connecticut are working on it. In a new study in Cell Metabolism, researchers described how to target specific cells to extend the lifespan and improve the health of mice late in life.
The study builds on a growing body of research, mostly in animals, testing interventions to slow aging and prolong health span, the length of time that one is not just alive but also healthy.
“Aging is the most important risk factor for every disease that we deal with in adult human beings,” said cardiologist Douglas Vaughan, MD, director of the Potocsnak Longevity Institute at Northwestern University’s Feinberg School of Medicine, Chicago. (Dr. Vaughan was not involved in the new study.) “So the big hypothesis is: If we could slow down aging just a little bit, we can push back the onset of disease.”
Senescent cells — or “zombie cells” — secrete harmful substances that disrupt tissue functioning. They’ve been linked to chronic inflammation, tissue damage, and the development of age-related diseases.
Senescence can be characterized by the accumulation of cells with high levels of specific markers like p21, or p21high cells. Almost any cell can become a p21high cell, and they accumulate with age, said Ming Xu, PhD, a professor at the UConn Center on Aging, UConn Health, Farmington, Connecticut, who led the study.
By targeting and eliminating p21high senescent cells, Dr. Xu hopes to develop novel therapies that might help people live longer and enjoy more years in good health.
Such a treatment could be ready for human trials in 2-5 years, Dr. Xu said.
What the Researchers Did
Xu and colleagues used genetic engineering to eliminate p21high cells in mice, introducing into their genome something they describe as an inducible “suicide gene.” Giving the mice a certain drug (a low dose of tamoxifen) activated the suicide gene in all p21high cells, causing them to die. Administering this treatment once a month, from age 20 months (older age) until the end of life, significantly extended the rodents’ lifespan, reduced inflammation, and decreased gene activity linked to aging.
Treated mice lived, on average, for 33 months — 3 months longer than the untreated mice. The oldest treated mouse lived to 43 months — roughly 130 in human years.
But the treated mice didn’t just live longer; they were also healthier. In humans, walking speed and grip strength can be clues of overall health and vitality. The old, treated mice were able to walk faster and grip objects with greater strength than untreated mice of the same age.
Dr. Xu’s lab is now testing drugs that target p21high cells in hopes of finding one that would work in humans. Leveraging immunotherapy technology to target these cells could be another option, Dr. Xu said.
The team also plans to test whether eliminating p21high cells could prevent or alleviate diabetes or Alzheimer’s disease.
Challenges and Criticisms
The research provides “important evidence that targeting senescence and the molecular components of that pathway might provide some benefit in the long term,” Dr. Vaughan said.
But killing senescent cells could come with downsides.
“Senescence protects us from hyperproliferative responses,” potentially blocking cells from becoming malignant, Dr. Vaughan said. “There’s this effect on aging that is desirable, but at the same time, you may enhance your risk of cancer or malignancy or excessive proliferation in some cells.”
And of course, we don’t necessarily need drugs to prolong healthy life, Dr. Vaughan pointed out.
For many people, a long healthy life is already within reach. Humans live longer on average than they used to, and simple lifestyle choices — nourishing your body well, staying active, and maintaining a healthy weight — can increase one’s chances of good health.
The most consistently demonstrated intervention for extending lifespan “in almost every animal species is caloric restriction,” Dr. Vaughan said. (Dr. Xu’s team is also investigating whether fasting and exercise can lead to a decrease in p21high cells.)
As for brain health, Dr. Vaughan and colleagues at Northwestern are studying “super agers,” people who are cognitively intact into their 90s.
“The one single thing that they found that contributes to that process, and contributes to that success, is really a social network and human bonds and interaction,” Dr. Vaughan said.
A version of this article appeared on Medscape.com.
Ultraprocessed Foods Upped Risk for Diabetic Complications
TOPLINE:
In patients with type 2 diabetes (T2D), eating more ultraprocessed food (UPF) increased the overall risk for microvascular complications and for diabetic kidney disease in particular. The risk was partly mediated by biomarkers related to body weight, lipid metabolism, and inflammation.
METHODOLOGY:
- Researchers evaluated the association between the intake of UPF and the risk for diabetic microvascular complications in a prospective cohort of 5685 participants with T2D (mean age, 59.7 years; 63.8% men) from the UK Biobank.
- Dietary information of participants was collected with a web-based 24-hour dietary recall tool that recorded the frequency of consumption of 206 foods and 32 beverages.
- Researchers found five patterns that accounted for one third of UPF intake variation by estimated weight (not calories): Bread and spreads; cereal with liquids; high dairy and low cured meat; sugary beverages and snacks; and mixed beverages and savory snacks.
- The outcomes included the risk for overall microvascular complications; for diabetic retinopathy, diabetic neuropathy, and diabetic kidney disease; and for biomarkers related to microvascular complications.
TAKEAWAY:
- During a median follow-up duration of 12.7 years, 1243 composite microvascular complications were reported, including 599 diabetic retinopathy, 237 diabetic neuropathy, and 662 diabetic kidney disease events.
- Each 10% increase in the proportion of UPF consumption increased the risk for composite microvascular complications by 8% (hazard ratio [HR], 1.08; 95% CI, 1.03-1.13) and diabetic kidney disease by 13% (HR 1.13; 95% CI, 1.06-1.20). No significant UPF intake association was found with diabetic retinopathy or diabetic neuropathy.
- In the biomarker mediation analysis, body mass index, triglycerides, and C-reactive protein collectively explained 22% (P < .001) and 15.8% (P < .001) of the associations of UPF consumption with composite microvascular complications and diabetic kidney disease, respectively.
- The food pattern rich in sugary beverages and snacks increased the risk for diabetic kidney disease, whereas the pattern rich in mixed beverages and savory snacks increased the risk for composite microvascular complications and diabetic retinopathy.
IN PRACTICE:
“In view of microvascular complications, our findings further support adhering to the recommendations outlined in the American Diabetes Association’s 2022 guidelines, which advocate for the preference of whole foods over highly processed ones,” the authors wrote.
SOURCE:
The study was led by Yue Li, MBBS, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. It was published online in The American Journal of Clinical Nutrition.
LIMITATIONS:
The dietary recall used in the UK Biobank was not specifically designed to collect dietary data according to the Nova food categories used in the study, which may have led to misclassifications. Data on usual dietary intake may not have been captured accurately, as all participants did not provide multiple dietary recalls. Individuals with T2D who completed dietary assessments were more likely to have a higher socioeconomic status and healthier lifestyle than those not filling the assessment, which could have resulted in an underrepresentation of high UPF consumers.
DISCLOSURES:
Some authors received funding from the National Natural Science Foundation of China, the National Key Research and Development Program of China, and other government sources. None of the authors declared any competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In patients with type 2 diabetes (T2D), eating more ultraprocessed food (UPF) increased the overall risk for microvascular complications and for diabetic kidney disease in particular. The risk was partly mediated by biomarkers related to body weight, lipid metabolism, and inflammation.
METHODOLOGY:
- Researchers evaluated the association between the intake of UPF and the risk for diabetic microvascular complications in a prospective cohort of 5685 participants with T2D (mean age, 59.7 years; 63.8% men) from the UK Biobank.
- Dietary information of participants was collected with a web-based 24-hour dietary recall tool that recorded the frequency of consumption of 206 foods and 32 beverages.
- Researchers found five patterns that accounted for one third of UPF intake variation by estimated weight (not calories): Bread and spreads; cereal with liquids; high dairy and low cured meat; sugary beverages and snacks; and mixed beverages and savory snacks.
- The outcomes included the risk for overall microvascular complications; for diabetic retinopathy, diabetic neuropathy, and diabetic kidney disease; and for biomarkers related to microvascular complications.
TAKEAWAY:
- During a median follow-up duration of 12.7 years, 1243 composite microvascular complications were reported, including 599 diabetic retinopathy, 237 diabetic neuropathy, and 662 diabetic kidney disease events.
- Each 10% increase in the proportion of UPF consumption increased the risk for composite microvascular complications by 8% (hazard ratio [HR], 1.08; 95% CI, 1.03-1.13) and diabetic kidney disease by 13% (HR 1.13; 95% CI, 1.06-1.20). No significant UPF intake association was found with diabetic retinopathy or diabetic neuropathy.
- In the biomarker mediation analysis, body mass index, triglycerides, and C-reactive protein collectively explained 22% (P < .001) and 15.8% (P < .001) of the associations of UPF consumption with composite microvascular complications and diabetic kidney disease, respectively.
- The food pattern rich in sugary beverages and snacks increased the risk for diabetic kidney disease, whereas the pattern rich in mixed beverages and savory snacks increased the risk for composite microvascular complications and diabetic retinopathy.
IN PRACTICE:
“In view of microvascular complications, our findings further support adhering to the recommendations outlined in the American Diabetes Association’s 2022 guidelines, which advocate for the preference of whole foods over highly processed ones,” the authors wrote.
SOURCE:
The study was led by Yue Li, MBBS, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. It was published online in The American Journal of Clinical Nutrition.
LIMITATIONS:
The dietary recall used in the UK Biobank was not specifically designed to collect dietary data according to the Nova food categories used in the study, which may have led to misclassifications. Data on usual dietary intake may not have been captured accurately, as all participants did not provide multiple dietary recalls. Individuals with T2D who completed dietary assessments were more likely to have a higher socioeconomic status and healthier lifestyle than those not filling the assessment, which could have resulted in an underrepresentation of high UPF consumers.
DISCLOSURES:
Some authors received funding from the National Natural Science Foundation of China, the National Key Research and Development Program of China, and other government sources. None of the authors declared any competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In patients with type 2 diabetes (T2D), eating more ultraprocessed food (UPF) increased the overall risk for microvascular complications and for diabetic kidney disease in particular. The risk was partly mediated by biomarkers related to body weight, lipid metabolism, and inflammation.
METHODOLOGY:
- Researchers evaluated the association between the intake of UPF and the risk for diabetic microvascular complications in a prospective cohort of 5685 participants with T2D (mean age, 59.7 years; 63.8% men) from the UK Biobank.
- Dietary information of participants was collected with a web-based 24-hour dietary recall tool that recorded the frequency of consumption of 206 foods and 32 beverages.
- Researchers found five patterns that accounted for one third of UPF intake variation by estimated weight (not calories): Bread and spreads; cereal with liquids; high dairy and low cured meat; sugary beverages and snacks; and mixed beverages and savory snacks.
- The outcomes included the risk for overall microvascular complications; for diabetic retinopathy, diabetic neuropathy, and diabetic kidney disease; and for biomarkers related to microvascular complications.
TAKEAWAY:
- During a median follow-up duration of 12.7 years, 1243 composite microvascular complications were reported, including 599 diabetic retinopathy, 237 diabetic neuropathy, and 662 diabetic kidney disease events.
- Each 10% increase in the proportion of UPF consumption increased the risk for composite microvascular complications by 8% (hazard ratio [HR], 1.08; 95% CI, 1.03-1.13) and diabetic kidney disease by 13% (HR 1.13; 95% CI, 1.06-1.20). No significant UPF intake association was found with diabetic retinopathy or diabetic neuropathy.
- In the biomarker mediation analysis, body mass index, triglycerides, and C-reactive protein collectively explained 22% (P < .001) and 15.8% (P < .001) of the associations of UPF consumption with composite microvascular complications and diabetic kidney disease, respectively.
- The food pattern rich in sugary beverages and snacks increased the risk for diabetic kidney disease, whereas the pattern rich in mixed beverages and savory snacks increased the risk for composite microvascular complications and diabetic retinopathy.
IN PRACTICE:
“In view of microvascular complications, our findings further support adhering to the recommendations outlined in the American Diabetes Association’s 2022 guidelines, which advocate for the preference of whole foods over highly processed ones,” the authors wrote.
SOURCE:
The study was led by Yue Li, MBBS, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. It was published online in The American Journal of Clinical Nutrition.
LIMITATIONS:
The dietary recall used in the UK Biobank was not specifically designed to collect dietary data according to the Nova food categories used in the study, which may have led to misclassifications. Data on usual dietary intake may not have been captured accurately, as all participants did not provide multiple dietary recalls. Individuals with T2D who completed dietary assessments were more likely to have a higher socioeconomic status and healthier lifestyle than those not filling the assessment, which could have resulted in an underrepresentation of high UPF consumers.
DISCLOSURES:
Some authors received funding from the National Natural Science Foundation of China, the National Key Research and Development Program of China, and other government sources. None of the authors declared any competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
What Would ‘Project 2025’ Mean for Health and Healthcare?
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A Racing Heart Signals Trouble in Chronic Kidney Disease
TOPLINE:
A higher resting heart rate, even within the normal range, is linked to an increased risk for mortality and cardiovascular events in patients with non–dialysis-dependent chronic kidney disease (CKD).
METHODOLOGY:
- An elevated resting heart rate is an independent risk factor for all-cause mortality and cardiovascular events in the general population; however, the correlation between heart rate and mortality in patients with CKD is unclear.
- Researchers analyzed the longitudinal data of patients with non–dialysis-dependent CKD enrolled in the Fukushima CKD Cohort Study to investigate the association between resting heart rate and adverse clinical outcomes.
- The patient cohort was stratified into four groups on the basis of resting heart rates: < 70, 70-79, 80-89, and ≥ 90 beats/min.
- The primary and secondary outcomes were all-cause mortality and cardiovascular events, respectively, the latter category including myocardial infarction, angina pectoris, and heart failure.
TAKEAWAY:
- Researchers enrolled 1353 patients with non–dialysis-dependent CKD (median age, 65 years; 56.7% men; median estimated glomerular filtration rate, 52.2 mL/min/1.73 m2) who had a median heart rate of 76 beats/min.
- During the median observation period of 4.9 years, 123 patients died and 163 developed cardiovascular events.
- Compared with patients with a resting heart rate < 70 beats/min, those with a resting heart rate of 80-89 and ≥ 90 beats/min had an adjusted hazard ratio of 1.74 and 2.61 for all-cause mortality, respectively.
- Similarly, the risk for cardiovascular events was higher in patients with a heart rate of 80-89 beats/min than in those with a heart rate < 70 beats/min (adjusted hazard ratio, 1.70).
IN PRACTICE:
“The present study supported the idea that reducing heart rate might be effective for CKD patients with a heart rate ≥ 70/min, since the lowest risk of mortality was seen in patients with heart rate < 70/min,” the authors concluded.
SOURCE:
This study was led by Hirotaka Saito, Department of Nephrology and Hypertension, Fukushima Medical University, Fukushima City, Japan. It was published online in Scientific Reports.
LIMITATIONS:
Heart rate was measured using a standard sphygmomanometer or an automated device, rather than an electrocardiograph, which may have introduced measurement variability. The observational nature of the study precluded the establishment of cause-and-effect relationships between heart rate and clinical outcomes. Additionally, variables such as lifestyle factors, underlying health conditions, and socioeconomic factors were not measured, which could have affected the results.
DISCLOSURES:
Some authors received research funding from Chugai Pharmaceutical, Kowa Pharmaceutical, Ono Pharmaceutical, and other sources. They declared having no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A higher resting heart rate, even within the normal range, is linked to an increased risk for mortality and cardiovascular events in patients with non–dialysis-dependent chronic kidney disease (CKD).
METHODOLOGY:
- An elevated resting heart rate is an independent risk factor for all-cause mortality and cardiovascular events in the general population; however, the correlation between heart rate and mortality in patients with CKD is unclear.
- Researchers analyzed the longitudinal data of patients with non–dialysis-dependent CKD enrolled in the Fukushima CKD Cohort Study to investigate the association between resting heart rate and adverse clinical outcomes.
- The patient cohort was stratified into four groups on the basis of resting heart rates: < 70, 70-79, 80-89, and ≥ 90 beats/min.
- The primary and secondary outcomes were all-cause mortality and cardiovascular events, respectively, the latter category including myocardial infarction, angina pectoris, and heart failure.
TAKEAWAY:
- Researchers enrolled 1353 patients with non–dialysis-dependent CKD (median age, 65 years; 56.7% men; median estimated glomerular filtration rate, 52.2 mL/min/1.73 m2) who had a median heart rate of 76 beats/min.
- During the median observation period of 4.9 years, 123 patients died and 163 developed cardiovascular events.
- Compared with patients with a resting heart rate < 70 beats/min, those with a resting heart rate of 80-89 and ≥ 90 beats/min had an adjusted hazard ratio of 1.74 and 2.61 for all-cause mortality, respectively.
- Similarly, the risk for cardiovascular events was higher in patients with a heart rate of 80-89 beats/min than in those with a heart rate < 70 beats/min (adjusted hazard ratio, 1.70).
IN PRACTICE:
“The present study supported the idea that reducing heart rate might be effective for CKD patients with a heart rate ≥ 70/min, since the lowest risk of mortality was seen in patients with heart rate < 70/min,” the authors concluded.
SOURCE:
This study was led by Hirotaka Saito, Department of Nephrology and Hypertension, Fukushima Medical University, Fukushima City, Japan. It was published online in Scientific Reports.
LIMITATIONS:
Heart rate was measured using a standard sphygmomanometer or an automated device, rather than an electrocardiograph, which may have introduced measurement variability. The observational nature of the study precluded the establishment of cause-and-effect relationships between heart rate and clinical outcomes. Additionally, variables such as lifestyle factors, underlying health conditions, and socioeconomic factors were not measured, which could have affected the results.
DISCLOSURES:
Some authors received research funding from Chugai Pharmaceutical, Kowa Pharmaceutical, Ono Pharmaceutical, and other sources. They declared having no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A higher resting heart rate, even within the normal range, is linked to an increased risk for mortality and cardiovascular events in patients with non–dialysis-dependent chronic kidney disease (CKD).
METHODOLOGY:
- An elevated resting heart rate is an independent risk factor for all-cause mortality and cardiovascular events in the general population; however, the correlation between heart rate and mortality in patients with CKD is unclear.
- Researchers analyzed the longitudinal data of patients with non–dialysis-dependent CKD enrolled in the Fukushima CKD Cohort Study to investigate the association between resting heart rate and adverse clinical outcomes.
- The patient cohort was stratified into four groups on the basis of resting heart rates: < 70, 70-79, 80-89, and ≥ 90 beats/min.
- The primary and secondary outcomes were all-cause mortality and cardiovascular events, respectively, the latter category including myocardial infarction, angina pectoris, and heart failure.
TAKEAWAY:
- Researchers enrolled 1353 patients with non–dialysis-dependent CKD (median age, 65 years; 56.7% men; median estimated glomerular filtration rate, 52.2 mL/min/1.73 m2) who had a median heart rate of 76 beats/min.
- During the median observation period of 4.9 years, 123 patients died and 163 developed cardiovascular events.
- Compared with patients with a resting heart rate < 70 beats/min, those with a resting heart rate of 80-89 and ≥ 90 beats/min had an adjusted hazard ratio of 1.74 and 2.61 for all-cause mortality, respectively.
- Similarly, the risk for cardiovascular events was higher in patients with a heart rate of 80-89 beats/min than in those with a heart rate < 70 beats/min (adjusted hazard ratio, 1.70).
IN PRACTICE:
“The present study supported the idea that reducing heart rate might be effective for CKD patients with a heart rate ≥ 70/min, since the lowest risk of mortality was seen in patients with heart rate < 70/min,” the authors concluded.
SOURCE:
This study was led by Hirotaka Saito, Department of Nephrology and Hypertension, Fukushima Medical University, Fukushima City, Japan. It was published online in Scientific Reports.
LIMITATIONS:
Heart rate was measured using a standard sphygmomanometer or an automated device, rather than an electrocardiograph, which may have introduced measurement variability. The observational nature of the study precluded the establishment of cause-and-effect relationships between heart rate and clinical outcomes. Additionally, variables such as lifestyle factors, underlying health conditions, and socioeconomic factors were not measured, which could have affected the results.
DISCLOSURES:
Some authors received research funding from Chugai Pharmaceutical, Kowa Pharmaceutical, Ono Pharmaceutical, and other sources. They declared having no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Remission or Not, Biologics May Mitigate Cardiovascular Risks of RA
TOPLINE:
, suggesting that biologics may reduce cardiovascular risk in RA even if remission is not achieved.
METHODOLOGY:
- Studies reported reduced cardiovascular risk in patients with RA who respond to tumor necrosis factor inhibitors but not in nonresponders, highlighting the importance of controlling inflammation for cardiovascular protection.
- Researchers assessed whether bDMARDs modify the impact of disease activity and systemic inflammation on cardiovascular risk in 4370 patients (mean age, 55 years) with RA without cardiovascular disease from a 10-country observational cohort.
- The severity of RA disease activity was assessed using C-reactive protein (CRP) levels and 28-joint Disease Activity Score based on CRP (DAS28-CRP).
- Endpoints were time to first MACE — a composite of cardiovascular death, myocardial infarction, and stroke — and time to first ischemic cardiovascular event (iCVE) — a composite of MACE plus revascularization, angina, transient ischemic attack, and peripheral arterial disease.
TAKEAWAY:
- The interaction between use of bDMARD and DAS28-CRP (P = .017) or CRP (P = .011) was significant for MACE.
- Each unit increase in DAS28-CRP increased the risk for MACE in bDMARD nonusers (hazard ratio [HR], 1.21; P = .002) but not in users.
- The per log unit increase in CRP was associated with a risk for MACE in bDMARD nonusers (HR, 1.16; P = .009) but not in users.
- No interaction was observed between bDMARD use and DAS28-CRP or CRP for the iCVE risk.
IN PRACTICE:
“This may indicate additional bDMARD-specific benefits directly on arterial wall inflammation and atherosclerotic plaque anatomy, stability, and biology, independently of systemic inflammation,” the authors wrote.
SOURCE:
The study, led by George Athanasios Karpouzas, MD, The Lundquist Institute, Torrance, California, was published online in RMD Open.
LIMITATIONS:
Patients with a particular interest in RA-associated cardiovascular disease were included, which may have introduced referral bias and affected the generalizability of the findings. Standard definitions were used for selected outcomes; however, differences in the reporting of outcomes may be plausible. Some patients were evaluated prospectively, while others were evaluated retrospectively, leading to differences in surveillance.
DISCLOSURES:
The study was supported by Pfizer. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
, suggesting that biologics may reduce cardiovascular risk in RA even if remission is not achieved.
METHODOLOGY:
- Studies reported reduced cardiovascular risk in patients with RA who respond to tumor necrosis factor inhibitors but not in nonresponders, highlighting the importance of controlling inflammation for cardiovascular protection.
- Researchers assessed whether bDMARDs modify the impact of disease activity and systemic inflammation on cardiovascular risk in 4370 patients (mean age, 55 years) with RA without cardiovascular disease from a 10-country observational cohort.
- The severity of RA disease activity was assessed using C-reactive protein (CRP) levels and 28-joint Disease Activity Score based on CRP (DAS28-CRP).
- Endpoints were time to first MACE — a composite of cardiovascular death, myocardial infarction, and stroke — and time to first ischemic cardiovascular event (iCVE) — a composite of MACE plus revascularization, angina, transient ischemic attack, and peripheral arterial disease.
TAKEAWAY:
- The interaction between use of bDMARD and DAS28-CRP (P = .017) or CRP (P = .011) was significant for MACE.
- Each unit increase in DAS28-CRP increased the risk for MACE in bDMARD nonusers (hazard ratio [HR], 1.21; P = .002) but not in users.
- The per log unit increase in CRP was associated with a risk for MACE in bDMARD nonusers (HR, 1.16; P = .009) but not in users.
- No interaction was observed between bDMARD use and DAS28-CRP or CRP for the iCVE risk.
IN PRACTICE:
“This may indicate additional bDMARD-specific benefits directly on arterial wall inflammation and atherosclerotic plaque anatomy, stability, and biology, independently of systemic inflammation,” the authors wrote.
SOURCE:
The study, led by George Athanasios Karpouzas, MD, The Lundquist Institute, Torrance, California, was published online in RMD Open.
LIMITATIONS:
Patients with a particular interest in RA-associated cardiovascular disease were included, which may have introduced referral bias and affected the generalizability of the findings. Standard definitions were used for selected outcomes; however, differences in the reporting of outcomes may be plausible. Some patients were evaluated prospectively, while others were evaluated retrospectively, leading to differences in surveillance.
DISCLOSURES:
The study was supported by Pfizer. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
, suggesting that biologics may reduce cardiovascular risk in RA even if remission is not achieved.
METHODOLOGY:
- Studies reported reduced cardiovascular risk in patients with RA who respond to tumor necrosis factor inhibitors but not in nonresponders, highlighting the importance of controlling inflammation for cardiovascular protection.
- Researchers assessed whether bDMARDs modify the impact of disease activity and systemic inflammation on cardiovascular risk in 4370 patients (mean age, 55 years) with RA without cardiovascular disease from a 10-country observational cohort.
- The severity of RA disease activity was assessed using C-reactive protein (CRP) levels and 28-joint Disease Activity Score based on CRP (DAS28-CRP).
- Endpoints were time to first MACE — a composite of cardiovascular death, myocardial infarction, and stroke — and time to first ischemic cardiovascular event (iCVE) — a composite of MACE plus revascularization, angina, transient ischemic attack, and peripheral arterial disease.
TAKEAWAY:
- The interaction between use of bDMARD and DAS28-CRP (P = .017) or CRP (P = .011) was significant for MACE.
- Each unit increase in DAS28-CRP increased the risk for MACE in bDMARD nonusers (hazard ratio [HR], 1.21; P = .002) but not in users.
- The per log unit increase in CRP was associated with a risk for MACE in bDMARD nonusers (HR, 1.16; P = .009) but not in users.
- No interaction was observed between bDMARD use and DAS28-CRP or CRP for the iCVE risk.
IN PRACTICE:
“This may indicate additional bDMARD-specific benefits directly on arterial wall inflammation and atherosclerotic plaque anatomy, stability, and biology, independently of systemic inflammation,” the authors wrote.
SOURCE:
The study, led by George Athanasios Karpouzas, MD, The Lundquist Institute, Torrance, California, was published online in RMD Open.
LIMITATIONS:
Patients with a particular interest in RA-associated cardiovascular disease were included, which may have introduced referral bias and affected the generalizability of the findings. Standard definitions were used for selected outcomes; however, differences in the reporting of outcomes may be plausible. Some patients were evaluated prospectively, while others were evaluated retrospectively, leading to differences in surveillance.
DISCLOSURES:
The study was supported by Pfizer. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
These Four Factors Account for 18 Years of Life Expectancy
This transcript has been edited for clarity.
Two individuals in the United States are celebrating their 30th birthdays. It’s a good day. They are entering the prime of their lives. One is a married White woman with a university degree. The other is a never-married White man with a high school diploma.
How many more years of life can these two individuals look forward to?
There’s a fairly dramatic difference. The man can expect 37.1 more years of life on average, living to be about 67. The woman can expect to live to age 85. That’s a life-expectancy discrepancy of 18 years based solely on gender, education, and marital status.
I’m using these cases to illustrate the extremes of life expectancy across four key social determinants of health: sex, race, marital status, and education. We all have some sense of how these factors play out in terms of health, but a new study suggests that it’s actually quite a bit more complicated than we thought.
Let me start by acknowledging my own bias here. As a clinical researcher, I sometimes find it hard to appreciate the value of actuarial-type studies that look at life expectancy (or any metric, really) between groups defined by marital status, for example. I’m never quite sure what to do with the conclusion. Married people live longer, the headline says. Okay, but as a doctor, what am I supposed to do about that? Encourage my patients to settle down and commit? Studies showing that women live longer than men or that White people live longer than Black people are also hard for me to incorporate into my practice. These are not easily changeable states.
But studies examining these groups are a reasonable starting point to ask more relevant questions. Why do women live longer than men? Is it behavioral (men take more risks and are less likely to see doctors)? Or is it hormonal (estrogen has a lot of protective effects that testosterone does not)? Or is it something else?
Integrating these social determinants of health into a cohesive story is a bit harder than it might seem, as this study, appearing in BMJ Open, illustrates.
In the context of this study, every person in America can be placed into one of 54 mutually exclusive groups. You can be male or female. You can be Black, White, or Hispanic. You can have a high school diploma or less, an associate degree, or a college degree; and you can be married, previously married, or never married.
Of course, this does not capture the beautiful tapestry that is American life, but let’s give them a pass. They are working with data from the American Community Survey, which contains 8634 people — the statistics would run into trouble with more granular divisions. This survey can be population weighted, so you can scale up the results to reasonably represent the population of the United States.
The survey collected data on the four broad categories of sex, race, education, and marital status and linked those survey results to the Multiple Cause of Death dataset from the CDC. From there, it’s a pretty simple task to rank the 54 categories in order from longest to shortest life expectancy, as you can see here.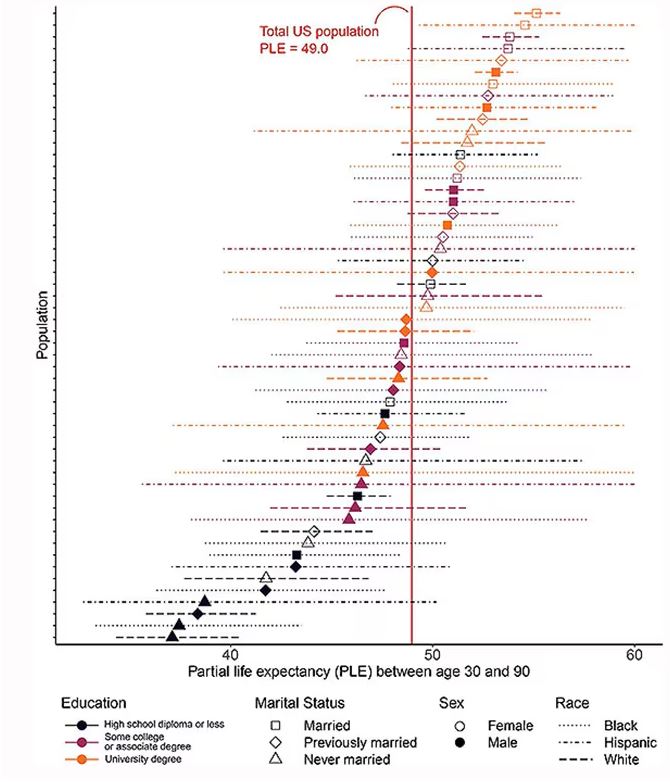
But that’s not really the interesting part of this study. Sure, there is a lot of variation; it’s interesting that these four factors explain about 18 years’ difference in life expectancy in this country. What strikes me here, actually, is the lack of an entirely consistent message across this spectrum.
Let me walk you through the second figure in this paper, because this nicely illustrates the surprising heterogeneity that exists here.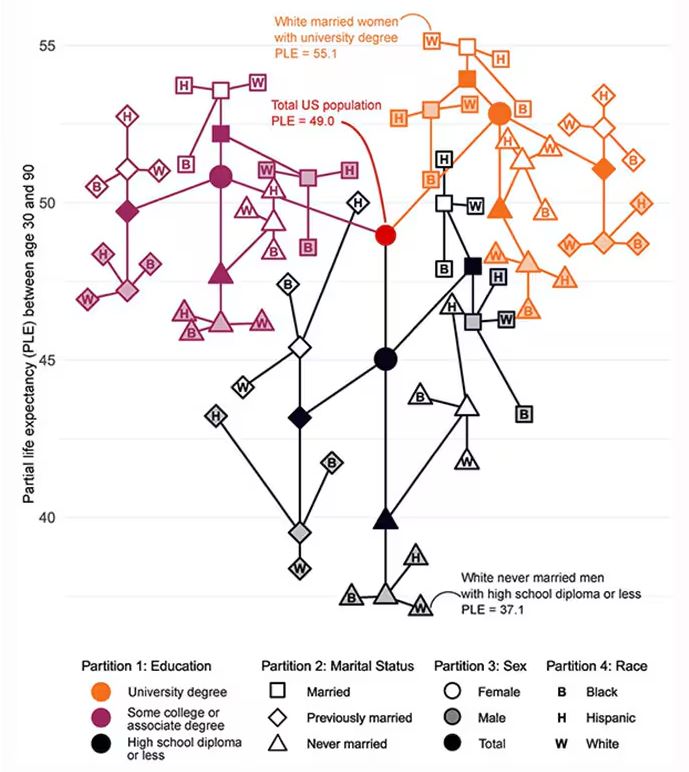
This may seem overwhelming, but basically, shapes that are higher up on the Y-axis represent the groups with longer life expectancy.
You can tell, for example, that shapes that are black in color (groups with high school educations or less) are generally lower. But not universally so. This box represents married, Hispanic females who do quite well in terms of life expectancy, even at that lower educational level.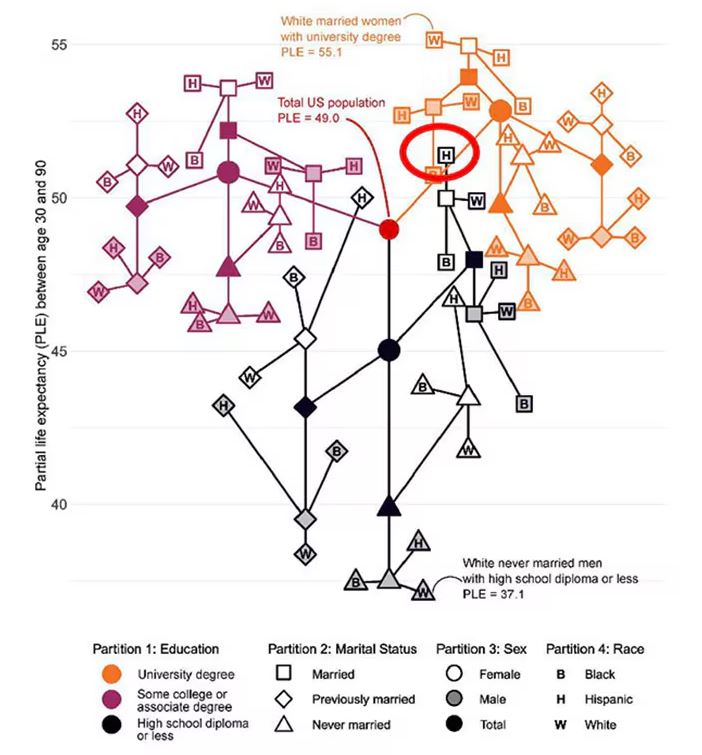
The authors quantify this phenomenon by creating a mortality risk score that integrates these findings. It looks something like this, with 0 being average morality for the United States.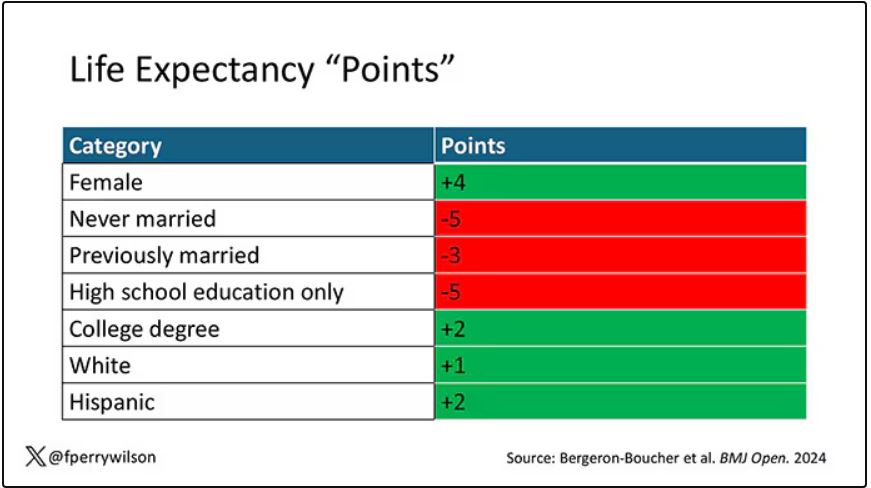
As you can see, you get a bunch of points for being female, but you lose a bunch for not being married. Education plays a large role, with a big hit for those who have a high school diploma or less, and a bonus for those with a college degree. Race plays a relatively more minor role.
This is all very interesting, but as I said at the beginning, this isn’t terribly useful to me as a physician. More important is figuring out why these differences exist. And there are some clues in the study data, particularly when we examine causes of death. This figure ranks those 54 groups again, from the married, White, college-educated females down to the never-married, White, high school–educated males. The boxes show how much more or less likely this group is to die of a given condition than the general population.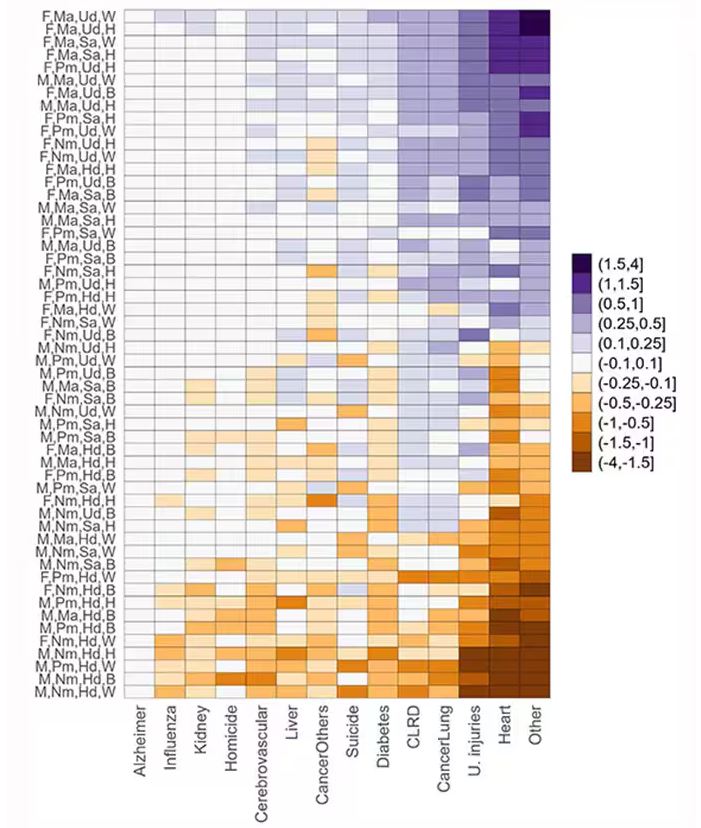
Looking at the bottom groups, you can see a dramatically increased risk for death from unintentional injuries, heart disease, and lung cancer. You see an increased risk for suicide as well. In the upper tiers, the only place where risk seems higher than expected is for the category of “other cancers,” reminding us that many types of cancer do not respect definitions of socioeconomic status.
You can even update the risk-scoring system to reflect the risk for different causes of death. You can see here how White people, for example, are at higher risk for death from unintentional injuries relative to other populations, despite having a lower mortality overall. 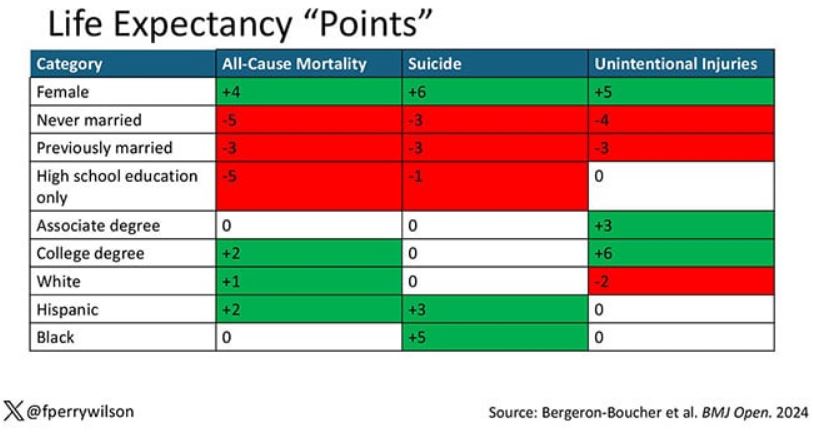
So maybe, through cause of death, we get a little closer to the answer of why. But this paper is really just a start. Its primary effect should be to surprise us — that in a country as wealthy as the United States, such dramatic variation exists based on factors that, with the exception of sex, I suppose, are not really biological. Which means that to find the why, we may need to turn from physiology to sociology.
Dr. Wilson is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Two individuals in the United States are celebrating their 30th birthdays. It’s a good day. They are entering the prime of their lives. One is a married White woman with a university degree. The other is a never-married White man with a high school diploma.
How many more years of life can these two individuals look forward to?
There’s a fairly dramatic difference. The man can expect 37.1 more years of life on average, living to be about 67. The woman can expect to live to age 85. That’s a life-expectancy discrepancy of 18 years based solely on gender, education, and marital status.
I’m using these cases to illustrate the extremes of life expectancy across four key social determinants of health: sex, race, marital status, and education. We all have some sense of how these factors play out in terms of health, but a new study suggests that it’s actually quite a bit more complicated than we thought.
Let me start by acknowledging my own bias here. As a clinical researcher, I sometimes find it hard to appreciate the value of actuarial-type studies that look at life expectancy (or any metric, really) between groups defined by marital status, for example. I’m never quite sure what to do with the conclusion. Married people live longer, the headline says. Okay, but as a doctor, what am I supposed to do about that? Encourage my patients to settle down and commit? Studies showing that women live longer than men or that White people live longer than Black people are also hard for me to incorporate into my practice. These are not easily changeable states.
But studies examining these groups are a reasonable starting point to ask more relevant questions. Why do women live longer than men? Is it behavioral (men take more risks and are less likely to see doctors)? Or is it hormonal (estrogen has a lot of protective effects that testosterone does not)? Or is it something else?
Integrating these social determinants of health into a cohesive story is a bit harder than it might seem, as this study, appearing in BMJ Open, illustrates.
In the context of this study, every person in America can be placed into one of 54 mutually exclusive groups. You can be male or female. You can be Black, White, or Hispanic. You can have a high school diploma or less, an associate degree, or a college degree; and you can be married, previously married, or never married.
Of course, this does not capture the beautiful tapestry that is American life, but let’s give them a pass. They are working with data from the American Community Survey, which contains 8634 people — the statistics would run into trouble with more granular divisions. This survey can be population weighted, so you can scale up the results to reasonably represent the population of the United States.
The survey collected data on the four broad categories of sex, race, education, and marital status and linked those survey results to the Multiple Cause of Death dataset from the CDC. From there, it’s a pretty simple task to rank the 54 categories in order from longest to shortest life expectancy, as you can see here.
But that’s not really the interesting part of this study. Sure, there is a lot of variation; it’s interesting that these four factors explain about 18 years’ difference in life expectancy in this country. What strikes me here, actually, is the lack of an entirely consistent message across this spectrum.
Let me walk you through the second figure in this paper, because this nicely illustrates the surprising heterogeneity that exists here.
This may seem overwhelming, but basically, shapes that are higher up on the Y-axis represent the groups with longer life expectancy.
You can tell, for example, that shapes that are black in color (groups with high school educations or less) are generally lower. But not universally so. This box represents married, Hispanic females who do quite well in terms of life expectancy, even at that lower educational level.
The authors quantify this phenomenon by creating a mortality risk score that integrates these findings. It looks something like this, with 0 being average morality for the United States.
As you can see, you get a bunch of points for being female, but you lose a bunch for not being married. Education plays a large role, with a big hit for those who have a high school diploma or less, and a bonus for those with a college degree. Race plays a relatively more minor role.
This is all very interesting, but as I said at the beginning, this isn’t terribly useful to me as a physician. More important is figuring out why these differences exist. And there are some clues in the study data, particularly when we examine causes of death. This figure ranks those 54 groups again, from the married, White, college-educated females down to the never-married, White, high school–educated males. The boxes show how much more or less likely this group is to die of a given condition than the general population.
Looking at the bottom groups, you can see a dramatically increased risk for death from unintentional injuries, heart disease, and lung cancer. You see an increased risk for suicide as well. In the upper tiers, the only place where risk seems higher than expected is for the category of “other cancers,” reminding us that many types of cancer do not respect definitions of socioeconomic status.
You can even update the risk-scoring system to reflect the risk for different causes of death. You can see here how White people, for example, are at higher risk for death from unintentional injuries relative to other populations, despite having a lower mortality overall. 
So maybe, through cause of death, we get a little closer to the answer of why. But this paper is really just a start. Its primary effect should be to surprise us — that in a country as wealthy as the United States, such dramatic variation exists based on factors that, with the exception of sex, I suppose, are not really biological. Which means that to find the why, we may need to turn from physiology to sociology.
Dr. Wilson is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Two individuals in the United States are celebrating their 30th birthdays. It’s a good day. They are entering the prime of their lives. One is a married White woman with a university degree. The other is a never-married White man with a high school diploma.
How many more years of life can these two individuals look forward to?
There’s a fairly dramatic difference. The man can expect 37.1 more years of life on average, living to be about 67. The woman can expect to live to age 85. That’s a life-expectancy discrepancy of 18 years based solely on gender, education, and marital status.
I’m using these cases to illustrate the extremes of life expectancy across four key social determinants of health: sex, race, marital status, and education. We all have some sense of how these factors play out in terms of health, but a new study suggests that it’s actually quite a bit more complicated than we thought.
Let me start by acknowledging my own bias here. As a clinical researcher, I sometimes find it hard to appreciate the value of actuarial-type studies that look at life expectancy (or any metric, really) between groups defined by marital status, for example. I’m never quite sure what to do with the conclusion. Married people live longer, the headline says. Okay, but as a doctor, what am I supposed to do about that? Encourage my patients to settle down and commit? Studies showing that women live longer than men or that White people live longer than Black people are also hard for me to incorporate into my practice. These are not easily changeable states.
But studies examining these groups are a reasonable starting point to ask more relevant questions. Why do women live longer than men? Is it behavioral (men take more risks and are less likely to see doctors)? Or is it hormonal (estrogen has a lot of protective effects that testosterone does not)? Or is it something else?
Integrating these social determinants of health into a cohesive story is a bit harder than it might seem, as this study, appearing in BMJ Open, illustrates.
In the context of this study, every person in America can be placed into one of 54 mutually exclusive groups. You can be male or female. You can be Black, White, or Hispanic. You can have a high school diploma or less, an associate degree, or a college degree; and you can be married, previously married, or never married.
Of course, this does not capture the beautiful tapestry that is American life, but let’s give them a pass. They are working with data from the American Community Survey, which contains 8634 people — the statistics would run into trouble with more granular divisions. This survey can be population weighted, so you can scale up the results to reasonably represent the population of the United States.
The survey collected data on the four broad categories of sex, race, education, and marital status and linked those survey results to the Multiple Cause of Death dataset from the CDC. From there, it’s a pretty simple task to rank the 54 categories in order from longest to shortest life expectancy, as you can see here.
But that’s not really the interesting part of this study. Sure, there is a lot of variation; it’s interesting that these four factors explain about 18 years’ difference in life expectancy in this country. What strikes me here, actually, is the lack of an entirely consistent message across this spectrum.
Let me walk you through the second figure in this paper, because this nicely illustrates the surprising heterogeneity that exists here.
This may seem overwhelming, but basically, shapes that are higher up on the Y-axis represent the groups with longer life expectancy.
You can tell, for example, that shapes that are black in color (groups with high school educations or less) are generally lower. But not universally so. This box represents married, Hispanic females who do quite well in terms of life expectancy, even at that lower educational level.
The authors quantify this phenomenon by creating a mortality risk score that integrates these findings. It looks something like this, with 0 being average morality for the United States.
As you can see, you get a bunch of points for being female, but you lose a bunch for not being married. Education plays a large role, with a big hit for those who have a high school diploma or less, and a bonus for those with a college degree. Race plays a relatively more minor role.
This is all very interesting, but as I said at the beginning, this isn’t terribly useful to me as a physician. More important is figuring out why these differences exist. And there are some clues in the study data, particularly when we examine causes of death. This figure ranks those 54 groups again, from the married, White, college-educated females down to the never-married, White, high school–educated males. The boxes show how much more or less likely this group is to die of a given condition than the general population.
Looking at the bottom groups, you can see a dramatically increased risk for death from unintentional injuries, heart disease, and lung cancer. You see an increased risk for suicide as well. In the upper tiers, the only place where risk seems higher than expected is for the category of “other cancers,” reminding us that many types of cancer do not respect definitions of socioeconomic status.
You can even update the risk-scoring system to reflect the risk for different causes of death. You can see here how White people, for example, are at higher risk for death from unintentional injuries relative to other populations, despite having a lower mortality overall. 
So maybe, through cause of death, we get a little closer to the answer of why. But this paper is really just a start. Its primary effect should be to surprise us — that in a country as wealthy as the United States, such dramatic variation exists based on factors that, with the exception of sex, I suppose, are not really biological. Which means that to find the why, we may need to turn from physiology to sociology.
Dr. Wilson is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Xanthelasma Not Linked to Heart Diseases, Study Finds
TOPLINE:
Xanthelasma palpebrarum, characterized by yellowish plaques on the eyelids, is not associated with increased rates of dyslipidemia or cardiovascular disease.
METHODOLOGY:
- Researchers conducted a case-control study at a single tertiary care center in Israel and analyzed data from 35,452 individuals (mean age, 52.2 years; 69% men) who underwent medical screening from 2001 to 2020.
- They compared 203 patients with xanthelasma palpebrarum with 2030 individuals without the disease (control).
- Primary outcomes were prevalence of dyslipidemia and cardiovascular disease between the two groups.
TAKEAWAY:
- Lipid profiles were similar between the two groups, with no difference in total cholesterol, high- and low-density lipoprotein, and triglyceride levels (all P > .05).
- The prevalence of dyslipidemia was similar for patients with xanthelasma palpebrarum and controls (46% vs 42%, respectively; P = .29), as was the incidence of cardiovascular disease (8.9% vs 10%, respectively; P = .56).
- The incidence of diabetes (P = .13), cerebrovascular accidents (P > .99), ischemic heart disease (P = .73), and hypertension (P = .56) were not significantly different between the two groups.
IN PRACTICE:
“Our study conducted on a large population of individuals undergoing comprehensive ophthalmic and systemic screening tests did not find a significant association between xanthelasma palpebrarum and an increased prevalence of lipid abnormalities or cardiovascular disease,” the authors wrote.
SOURCE:
The study was led by Yael Lustig, MD, of the Goldschleger Eye Institute at Sheba Medical Center, in Ramat Gan, Israel. It was published online on August 5, 2024, in Ophthalmology.
LIMITATIONS:
The retrospective nature of the study and the single-center design may have limited the generalizability of the findings. The study population was self-selected, potentially introducing selection bias. Lack of histopathologic examination could have affected the accuracy of the diagnosis.
DISCLOSURES:
No funding sources were disclosed for this study. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Xanthelasma palpebrarum, characterized by yellowish plaques on the eyelids, is not associated with increased rates of dyslipidemia or cardiovascular disease.
METHODOLOGY:
- Researchers conducted a case-control study at a single tertiary care center in Israel and analyzed data from 35,452 individuals (mean age, 52.2 years; 69% men) who underwent medical screening from 2001 to 2020.
- They compared 203 patients with xanthelasma palpebrarum with 2030 individuals without the disease (control).
- Primary outcomes were prevalence of dyslipidemia and cardiovascular disease between the two groups.
TAKEAWAY:
- Lipid profiles were similar between the two groups, with no difference in total cholesterol, high- and low-density lipoprotein, and triglyceride levels (all P > .05).
- The prevalence of dyslipidemia was similar for patients with xanthelasma palpebrarum and controls (46% vs 42%, respectively; P = .29), as was the incidence of cardiovascular disease (8.9% vs 10%, respectively; P = .56).
- The incidence of diabetes (P = .13), cerebrovascular accidents (P > .99), ischemic heart disease (P = .73), and hypertension (P = .56) were not significantly different between the two groups.
IN PRACTICE:
“Our study conducted on a large population of individuals undergoing comprehensive ophthalmic and systemic screening tests did not find a significant association between xanthelasma palpebrarum and an increased prevalence of lipid abnormalities or cardiovascular disease,” the authors wrote.
SOURCE:
The study was led by Yael Lustig, MD, of the Goldschleger Eye Institute at Sheba Medical Center, in Ramat Gan, Israel. It was published online on August 5, 2024, in Ophthalmology.
LIMITATIONS:
The retrospective nature of the study and the single-center design may have limited the generalizability of the findings. The study population was self-selected, potentially introducing selection bias. Lack of histopathologic examination could have affected the accuracy of the diagnosis.
DISCLOSURES:
No funding sources were disclosed for this study. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Xanthelasma palpebrarum, characterized by yellowish plaques on the eyelids, is not associated with increased rates of dyslipidemia or cardiovascular disease.
METHODOLOGY:
- Researchers conducted a case-control study at a single tertiary care center in Israel and analyzed data from 35,452 individuals (mean age, 52.2 years; 69% men) who underwent medical screening from 2001 to 2020.
- They compared 203 patients with xanthelasma palpebrarum with 2030 individuals without the disease (control).
- Primary outcomes were prevalence of dyslipidemia and cardiovascular disease between the two groups.
TAKEAWAY:
- Lipid profiles were similar between the two groups, with no difference in total cholesterol, high- and low-density lipoprotein, and triglyceride levels (all P > .05).
- The prevalence of dyslipidemia was similar for patients with xanthelasma palpebrarum and controls (46% vs 42%, respectively; P = .29), as was the incidence of cardiovascular disease (8.9% vs 10%, respectively; P = .56).
- The incidence of diabetes (P = .13), cerebrovascular accidents (P > .99), ischemic heart disease (P = .73), and hypertension (P = .56) were not significantly different between the two groups.
IN PRACTICE:
“Our study conducted on a large population of individuals undergoing comprehensive ophthalmic and systemic screening tests did not find a significant association between xanthelasma palpebrarum and an increased prevalence of lipid abnormalities or cardiovascular disease,” the authors wrote.
SOURCE:
The study was led by Yael Lustig, MD, of the Goldschleger Eye Institute at Sheba Medical Center, in Ramat Gan, Israel. It was published online on August 5, 2024, in Ophthalmology.
LIMITATIONS:
The retrospective nature of the study and the single-center design may have limited the generalizability of the findings. The study population was self-selected, potentially introducing selection bias. Lack of histopathologic examination could have affected the accuracy of the diagnosis.
DISCLOSURES:
No funding sources were disclosed for this study. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.


