User login
Short-term approach is best for seizure prevention after intracerebral hemorrhage
(sICH), new research shows.
Investigators created a model that simulated common clinical scenarios to compare four antiseizure drug strategies – conservative, moderate, aggressive, and risk-guided. They used the 2HELPS2B score as a risk stratification tool to guide clinical decisions.
The investigators found that the short-term, early-seizure prophylaxis strategies “dominated” long-term therapy under most clinical scenarios, underscoring the importance of early discontinuation of antiseizure drug therapy.
“The main message here was that strategies that involved long-term antiseizure drug prescription (moderate and aggressive) fail to provide better outcomes in most clinical scenarios, when compared with strategies using short-term prophylaxis (conservative and risk-guided),” senior investigator Lidia M.V.R. Moura, MD, MPH, assistant professor of neurology, Harvard Medical School, Boston, said in an interview.
The study was published online July 26 in JAMA Neurology.
Common complication
“Acute asymptomatic seizures [early seizures ≤7 days after stroke] are a common complication of sICH,” the authors noted.
Potential safety concerns have prompted recommendations against the use of antiseizure medications for primary prophylaxis. However, approximately 40% of U.S. patients with sICH do receive prophylactic levetiracetam before seizure development. For these patients, the duration of prophylaxis varies widely.
“Because seizure risk is a key determinant of which patient groups might benefit most from different prophylaxis strategies, validated tools for predicting early ... and late ... seizure risks could aid physicians in treatment decisions. However, no clinical trials or prospective studies have evaluated the net benefit of various strategies after sICH,” the investigators noted.
“Our patients who were survivors of an intracerebral hemorrhage motivated us to conduct the study,” said Dr. Moura, who is also director of the MGH NeuroValue Laboratory. “Some would come to the clinic with a long list of medications; some of them were taking antiseizure drugs for many years, but they never had a documented seizure.” These patients did not know why they had been taking an antiseizure drug for so long.
“In these conversations, we noted so much variability in indications and variability in patient access to specialty care to make treatment decisions. We noted that the evidence behind our current guidelines on seizure management was limited,” she added.
Dr. Moura and colleagues were “committed to improve outcome for people with neurological conditions by leveraging research methods that can help guide providers and systems, especially when data from clinical trials is lacking,” so they “decided to compare different strategies head to head using available data and generate evidence that could be used in situations with many trade-offs in risks and benefits.”
To investigate, the researchers used a simulation model and decision analysis to compare four treatment strategies on the basis of type of therapy (primary vs. secondary prophylaxis), timing of event (early vs. late seizures), and duration of therapy (1-week [short-term] versus indefinite [long-term] therapy).
These four strategies were as follows:
- Conservative: short-term (7-day) secondary early-seizure prophylaxis with long-term therapy after late seizure
- Moderate: long-term secondary early-seizure prophylaxis or late-seizure therapy
- Aggressive: long-term primary prophylaxis
- Risk-guided: short-term secondary early-seizure prophylaxis among low-risk patients (2HELPS2b score, 0), short-term primary prophylaxis among patients at higher risk (2HELPS2B score ≥1), and long-term secondary therapy for late seizure
The decision tree’s outcome measure was the number of expected quality-adjusted life-years.
Primary prophylaxis was defined as “treatment initiated immediately on hospital admission.” Secondary prophylaxis was defined as “treatment started after a seizure” and was subdivided into secondary early-seizure prophylaxis, defined as treatment started after a seizure occurring in the first 7 days after the stroke, or secondary late-seizure therapy, defined as treatment started or restarted after a seizure occurring after the first poststroke week.
Incorporate early-risk stratification tool
The researchers created four common clinical scenarios and then applied the decision-making model to each. They found that the preferred strategies differed, depending on the particular scenario.
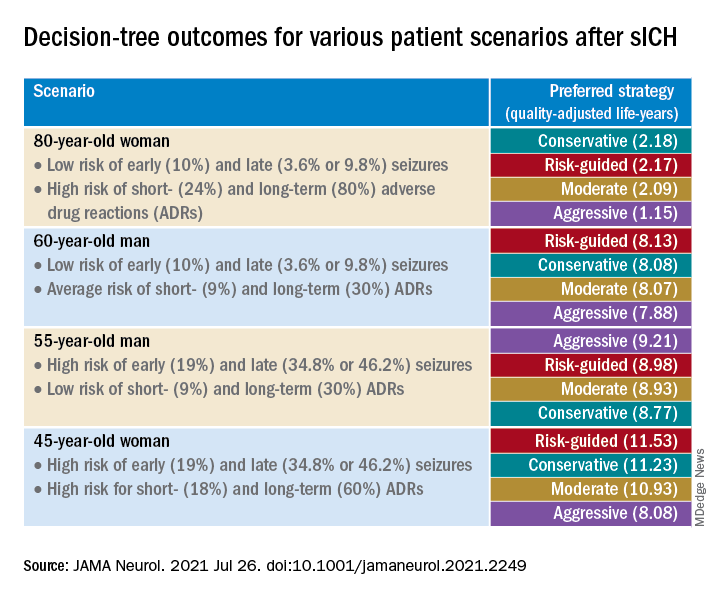
Sensitivity analyses revealed that short-term strategies, including the conservative and risk-guided approaches, were preferable in most cases, with the risk-guided strategy performing comparably or even better than alternative strategies in most cases.
“Our findings suggest that a strategy that incorporates an early-seizure risk stratification tool [2HELPS2B] is favored over alternative strategies in most settings,” Dr. Moura commented.
“Current services with rapidly available EEG may consider using a 1-hour screening with EEG upon admission for all patients presenting with sICH to risk-stratify those patients, using the 2HELPS2B tool,” she continued. “If EEG is unavailable for early-seizure risk stratification, the conservative strategy seems most reasonable.”
‘Potential fallacies’
Commenting on the study, José Biller, MD, professor and chairman, department of neurology, Loyola University Chicago, Maywood, Ill., called it a “well-written and intriguing contribution [to the field], with potential fallacies.”
The bottom line, he said, is that only a randomized, long-term, prospective, multicenter, high-quality study with larger cohorts can prove or disprove the investigators’ assumption.
The authors acknowledged that a limitation of the study was the use of published literature to obtain data to estimate model parameters and that they did not account for other possible factors that might modify some parameter estimates.
Nevertheless, Dr. Moura said the findings have important practical implications because they “highlight the importance of discontinuing antiseizure medications that were started during a hospitalization for sICH in patients that only had an early seizure.”
It is “of great importance for all providers to reassess the indication of antiseizure medications. Those drugs are not free of risks and can impact the patient’s health and quality of life,” she added.
The study was supported by grants from the National Institutes of Health. Dr. Moura reported receiving funding from the Centers for Disease Control and Prevention, the NIH, and the Epilepsy Foundation of America (Epilepsy Learning Healthcare System) as the director of the data coordinating center. Dr. Biller is the editor-in-chief of the Journal of Stroke and Cerebrovascular Diseases and a section editor of UpToDate.
A version of this article first appeared on Medscape.com.
(sICH), new research shows.
Investigators created a model that simulated common clinical scenarios to compare four antiseizure drug strategies – conservative, moderate, aggressive, and risk-guided. They used the 2HELPS2B score as a risk stratification tool to guide clinical decisions.
The investigators found that the short-term, early-seizure prophylaxis strategies “dominated” long-term therapy under most clinical scenarios, underscoring the importance of early discontinuation of antiseizure drug therapy.
“The main message here was that strategies that involved long-term antiseizure drug prescription (moderate and aggressive) fail to provide better outcomes in most clinical scenarios, when compared with strategies using short-term prophylaxis (conservative and risk-guided),” senior investigator Lidia M.V.R. Moura, MD, MPH, assistant professor of neurology, Harvard Medical School, Boston, said in an interview.
The study was published online July 26 in JAMA Neurology.
Common complication
“Acute asymptomatic seizures [early seizures ≤7 days after stroke] are a common complication of sICH,” the authors noted.
Potential safety concerns have prompted recommendations against the use of antiseizure medications for primary prophylaxis. However, approximately 40% of U.S. patients with sICH do receive prophylactic levetiracetam before seizure development. For these patients, the duration of prophylaxis varies widely.
“Because seizure risk is a key determinant of which patient groups might benefit most from different prophylaxis strategies, validated tools for predicting early ... and late ... seizure risks could aid physicians in treatment decisions. However, no clinical trials or prospective studies have evaluated the net benefit of various strategies after sICH,” the investigators noted.
“Our patients who were survivors of an intracerebral hemorrhage motivated us to conduct the study,” said Dr. Moura, who is also director of the MGH NeuroValue Laboratory. “Some would come to the clinic with a long list of medications; some of them were taking antiseizure drugs for many years, but they never had a documented seizure.” These patients did not know why they had been taking an antiseizure drug for so long.
“In these conversations, we noted so much variability in indications and variability in patient access to specialty care to make treatment decisions. We noted that the evidence behind our current guidelines on seizure management was limited,” she added.
Dr. Moura and colleagues were “committed to improve outcome for people with neurological conditions by leveraging research methods that can help guide providers and systems, especially when data from clinical trials is lacking,” so they “decided to compare different strategies head to head using available data and generate evidence that could be used in situations with many trade-offs in risks and benefits.”
To investigate, the researchers used a simulation model and decision analysis to compare four treatment strategies on the basis of type of therapy (primary vs. secondary prophylaxis), timing of event (early vs. late seizures), and duration of therapy (1-week [short-term] versus indefinite [long-term] therapy).
These four strategies were as follows:
- Conservative: short-term (7-day) secondary early-seizure prophylaxis with long-term therapy after late seizure
- Moderate: long-term secondary early-seizure prophylaxis or late-seizure therapy
- Aggressive: long-term primary prophylaxis
- Risk-guided: short-term secondary early-seizure prophylaxis among low-risk patients (2HELPS2b score, 0), short-term primary prophylaxis among patients at higher risk (2HELPS2B score ≥1), and long-term secondary therapy for late seizure
The decision tree’s outcome measure was the number of expected quality-adjusted life-years.
Primary prophylaxis was defined as “treatment initiated immediately on hospital admission.” Secondary prophylaxis was defined as “treatment started after a seizure” and was subdivided into secondary early-seizure prophylaxis, defined as treatment started after a seizure occurring in the first 7 days after the stroke, or secondary late-seizure therapy, defined as treatment started or restarted after a seizure occurring after the first poststroke week.
Incorporate early-risk stratification tool
The researchers created four common clinical scenarios and then applied the decision-making model to each. They found that the preferred strategies differed, depending on the particular scenario.

Sensitivity analyses revealed that short-term strategies, including the conservative and risk-guided approaches, were preferable in most cases, with the risk-guided strategy performing comparably or even better than alternative strategies in most cases.
“Our findings suggest that a strategy that incorporates an early-seizure risk stratification tool [2HELPS2B] is favored over alternative strategies in most settings,” Dr. Moura commented.
“Current services with rapidly available EEG may consider using a 1-hour screening with EEG upon admission for all patients presenting with sICH to risk-stratify those patients, using the 2HELPS2B tool,” she continued. “If EEG is unavailable for early-seizure risk stratification, the conservative strategy seems most reasonable.”
‘Potential fallacies’
Commenting on the study, José Biller, MD, professor and chairman, department of neurology, Loyola University Chicago, Maywood, Ill., called it a “well-written and intriguing contribution [to the field], with potential fallacies.”
The bottom line, he said, is that only a randomized, long-term, prospective, multicenter, high-quality study with larger cohorts can prove or disprove the investigators’ assumption.
The authors acknowledged that a limitation of the study was the use of published literature to obtain data to estimate model parameters and that they did not account for other possible factors that might modify some parameter estimates.
Nevertheless, Dr. Moura said the findings have important practical implications because they “highlight the importance of discontinuing antiseizure medications that were started during a hospitalization for sICH in patients that only had an early seizure.”
It is “of great importance for all providers to reassess the indication of antiseizure medications. Those drugs are not free of risks and can impact the patient’s health and quality of life,” she added.
The study was supported by grants from the National Institutes of Health. Dr. Moura reported receiving funding from the Centers for Disease Control and Prevention, the NIH, and the Epilepsy Foundation of America (Epilepsy Learning Healthcare System) as the director of the data coordinating center. Dr. Biller is the editor-in-chief of the Journal of Stroke and Cerebrovascular Diseases and a section editor of UpToDate.
A version of this article first appeared on Medscape.com.
(sICH), new research shows.
Investigators created a model that simulated common clinical scenarios to compare four antiseizure drug strategies – conservative, moderate, aggressive, and risk-guided. They used the 2HELPS2B score as a risk stratification tool to guide clinical decisions.
The investigators found that the short-term, early-seizure prophylaxis strategies “dominated” long-term therapy under most clinical scenarios, underscoring the importance of early discontinuation of antiseizure drug therapy.
“The main message here was that strategies that involved long-term antiseizure drug prescription (moderate and aggressive) fail to provide better outcomes in most clinical scenarios, when compared with strategies using short-term prophylaxis (conservative and risk-guided),” senior investigator Lidia M.V.R. Moura, MD, MPH, assistant professor of neurology, Harvard Medical School, Boston, said in an interview.
The study was published online July 26 in JAMA Neurology.
Common complication
“Acute asymptomatic seizures [early seizures ≤7 days after stroke] are a common complication of sICH,” the authors noted.
Potential safety concerns have prompted recommendations against the use of antiseizure medications for primary prophylaxis. However, approximately 40% of U.S. patients with sICH do receive prophylactic levetiracetam before seizure development. For these patients, the duration of prophylaxis varies widely.
“Because seizure risk is a key determinant of which patient groups might benefit most from different prophylaxis strategies, validated tools for predicting early ... and late ... seizure risks could aid physicians in treatment decisions. However, no clinical trials or prospective studies have evaluated the net benefit of various strategies after sICH,” the investigators noted.
“Our patients who were survivors of an intracerebral hemorrhage motivated us to conduct the study,” said Dr. Moura, who is also director of the MGH NeuroValue Laboratory. “Some would come to the clinic with a long list of medications; some of them were taking antiseizure drugs for many years, but they never had a documented seizure.” These patients did not know why they had been taking an antiseizure drug for so long.
“In these conversations, we noted so much variability in indications and variability in patient access to specialty care to make treatment decisions. We noted that the evidence behind our current guidelines on seizure management was limited,” she added.
Dr. Moura and colleagues were “committed to improve outcome for people with neurological conditions by leveraging research methods that can help guide providers and systems, especially when data from clinical trials is lacking,” so they “decided to compare different strategies head to head using available data and generate evidence that could be used in situations with many trade-offs in risks and benefits.”
To investigate, the researchers used a simulation model and decision analysis to compare four treatment strategies on the basis of type of therapy (primary vs. secondary prophylaxis), timing of event (early vs. late seizures), and duration of therapy (1-week [short-term] versus indefinite [long-term] therapy).
These four strategies were as follows:
- Conservative: short-term (7-day) secondary early-seizure prophylaxis with long-term therapy after late seizure
- Moderate: long-term secondary early-seizure prophylaxis or late-seizure therapy
- Aggressive: long-term primary prophylaxis
- Risk-guided: short-term secondary early-seizure prophylaxis among low-risk patients (2HELPS2b score, 0), short-term primary prophylaxis among patients at higher risk (2HELPS2B score ≥1), and long-term secondary therapy for late seizure
The decision tree’s outcome measure was the number of expected quality-adjusted life-years.
Primary prophylaxis was defined as “treatment initiated immediately on hospital admission.” Secondary prophylaxis was defined as “treatment started after a seizure” and was subdivided into secondary early-seizure prophylaxis, defined as treatment started after a seizure occurring in the first 7 days after the stroke, or secondary late-seizure therapy, defined as treatment started or restarted after a seizure occurring after the first poststroke week.
Incorporate early-risk stratification tool
The researchers created four common clinical scenarios and then applied the decision-making model to each. They found that the preferred strategies differed, depending on the particular scenario.

Sensitivity analyses revealed that short-term strategies, including the conservative and risk-guided approaches, were preferable in most cases, with the risk-guided strategy performing comparably or even better than alternative strategies in most cases.
“Our findings suggest that a strategy that incorporates an early-seizure risk stratification tool [2HELPS2B] is favored over alternative strategies in most settings,” Dr. Moura commented.
“Current services with rapidly available EEG may consider using a 1-hour screening with EEG upon admission for all patients presenting with sICH to risk-stratify those patients, using the 2HELPS2B tool,” she continued. “If EEG is unavailable for early-seizure risk stratification, the conservative strategy seems most reasonable.”
‘Potential fallacies’
Commenting on the study, José Biller, MD, professor and chairman, department of neurology, Loyola University Chicago, Maywood, Ill., called it a “well-written and intriguing contribution [to the field], with potential fallacies.”
The bottom line, he said, is that only a randomized, long-term, prospective, multicenter, high-quality study with larger cohorts can prove or disprove the investigators’ assumption.
The authors acknowledged that a limitation of the study was the use of published literature to obtain data to estimate model parameters and that they did not account for other possible factors that might modify some parameter estimates.
Nevertheless, Dr. Moura said the findings have important practical implications because they “highlight the importance of discontinuing antiseizure medications that were started during a hospitalization for sICH in patients that only had an early seizure.”
It is “of great importance for all providers to reassess the indication of antiseizure medications. Those drugs are not free of risks and can impact the patient’s health and quality of life,” she added.
The study was supported by grants from the National Institutes of Health. Dr. Moura reported receiving funding from the Centers for Disease Control and Prevention, the NIH, and the Epilepsy Foundation of America (Epilepsy Learning Healthcare System) as the director of the data coordinating center. Dr. Biller is the editor-in-chief of the Journal of Stroke and Cerebrovascular Diseases and a section editor of UpToDate.
A version of this article first appeared on Medscape.com.
Virtual roller-coaster may explain the ups and downs of migraine
and may explain the mechanisms underlying common symptoms and increased activity in certain brain regions in migraine patients.
In a new study, the prevalence of dizziness was 65% among patients with migraine who underwent a virtual roller-coaster ride versus 30% among those without migraine. In addition, imaging showed greater neuronal activity after the simulation in those with migraine.
“Migraine patients reported more dizziness and motion sickness, as well as longer symptom duration and intensity, in a virtual roller-coaster ride,” even though the videos and timing were identical for both groups, said study investigator Arne May, MD, PhD, professor of neurology at the University of Hamburg (Germany).
“We found differences not just in behavioral results but also in specific activations of areas within the cerebellum and the frontal gyrus. Migraine patients process such visual input differently from controls and activate a specific brain network to do so,” he added.
The findings were published online July 21, 2021, in Neurology.
The brain’s response
Nausea, which is among the diagnostic criteria for migraine, is the main symptom of motion sickness. Vestibular symptoms such as dizziness are also components of migraine.
Previous research has examined how the brain processes visual and motion stimuli in migraine, but the reasons patients with migraine are susceptible to motion sickness and dizziness remain unclear.
The researchers used a simulated roller-coaster ride to study the clinical and brain responses to motion among participants with and participants without migraine. They enrolled 20 consecutive patients with migraine who presented to a tertiary headache clinic between January and March 2020 and enrolled 20 healthy participants from a university hospital and the community. The average age of the study population was 30 years, and more than 80% were women.
In response to a questionnaire, participants provided information about demographics and headache features, including onset, frequency, and intensity. They also provided information about their status within the migraine phase and about vestibular symptoms experienced in daily life.
While undergoing functional MRI (fMRI), all participants watched two short videos that provided a first-person perspective of a roller-coaster ride. During the videos, they wore ear buds that conveyed the sound of a car riding over the rails.
The first video included more horizontal perspectives, and the second had more vertical perspectives. Each video was shown three times in random order.
During fMRI, participants reported intensity of nausea and vestibular symptoms using an 11-point Likert scale. After the experiment, they responded to a questionnaire that evaluated intensity and duration of nausea, dizziness, and vertigo experienced during the videos.
Participants also were given the Simulator Sickness Questionnaire (SSQ), which assessed motion sickness. A 100-point visual analog scale (VAS) was used to rate how realistic the roller-coaster experience had been.
There were no differences in sex or age between the migraine group and the healthy control group. Half of the patients with migraine reported aura. The mean number of migraine attacks within the previous month was 3.7. The mean Migraine Disability Assessment score was 21.5, which indicates severe disability.
Nausea, dizziness often neglected
Baseline prevalence of vestibular symptoms was 75% in the migraine group and 5% in the control group (P < .0001). These symptoms included dizziness (60% and 5%, respectively; P < .0001) and postural symptoms (40% and 0%, respectively; P = .003).
At baseline, vestibular symptoms were more frequent (P = .001), more intense (P < .0001), and were associated with greater disability (P = .001) in patients with migraine, compared with participants without migraine. The patients with migraine were also more susceptible to motion sickness (P = .02) and had higher depression scores (P = .001).
During the roller-coaster simulation, dizziness was more prevalent among patients with migraine than among those without migraine (65% vs. 30%; P = .03). Patients with migraine also reported more motion sickness (SSQ score, 47.3 vs. 24.3; P = .004), longer symptom duration (1:19 minutes vs. 00:27 minutes; P = .03), and symptoms of greater intensity (VAS, 22.0 vs. 9.9; P = .03).
Brain activity also differed between groups. Among patients with migraine, neuronal activity was greater in clusters within the right superior and left inferior occipital gyrus, the left pontine nuclei, and the left cerebellar lobules V and VI.
There was a moderately negative correlation of activation of the inferior occipital gyrus with migraine disability (r = –0.46; P = .04). Activation within the pontine nuclei correlated positively with motion sickness scores (r = 0.32; P = .04).
In addition, among patients with migraine, activity in the cerebellar lobule VIIb and in the left middle frontal gyrus was decreased in comparison with persons without migraine. Also among patients with migraine, there was enhanced connectivity between the pontine nuclei, cerebellar areas V and VI, and the interior and superior occipital gyrus and numerous cortical areas.
Clinicians often neglect to treat dizziness and nausea in patients with migraine, said Dr. May. However, these symptoms are part of migraine, even when attacks are not occurring.
“I have learned that if we can explain such symptoms, they are better accepted,” said Dr. May. “We need more and better basic research because we need to understand before we treat.”
Toward faster, more effective treatment
Commenting on the study, Erik Viirre, MD, PhD, professor in the department of neurosciences, University of California, San Diego, said, “we can be excited and celebrate that these researchers are using these news tools to investigate the operation of the migraine brain.
“That will combine with the new therapies and the genomics to give us a powerful approach to this particular condition,” said Dr. Viirre, who was not involved with the research.
The findings provide significant detail about the interconnections between the various brain regions affected by migraine, he noted. These regions include not just the sensory centers but also areas involved in higher executive function and emotional responses.
By identifying these regions, the findings show “some of the underlying mechanisms of these clinically relevant features,” said Dr. Viirre, who is also director of UCSD’s Arthur C. Clarke Center for Human Imagination.
The investigators set up the motion simulation well and used sound fMRI methodology, he added. However, imaging studies of the brain’s response to motion pose several challenges.
“The biggest challenge in any of these circumstances is that you can’t put an actual fMRI scanner on a roller-coaster,” said Dr. Viirre. “The actual acceleration and gravitational sensations delivered by a roller-coaster and gravity, of course, do not occur when you’re lying still in an MRI scanner.” Nevertheless, the pseudoacceleration produced by a visual stimulus is a reasonable proxy.
The findings also suggest that researchers in the future could examine whether any new therapeutic interventions for migraine modulate the brain functions differently for individuals with migraine than for those without migraine, he noted.
“That’s going to lead us to a faster, more effective, more reliable suite of migraine therapies,” said Dr. Viirre.
The study also reminds clinicians to take a broader approach to patients with migraine, and it underscores the value of strategies such as self-calming techniques, which can reduce the number and intensity of headaches, he said.
“Literally demonstrating these functional differences in the migraine brain is a hugely important message of advocacy for people with migraine,” Dr. Viirre concluded.
The study was funded by the German Research Foundation. Drs. May and Viirre have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
and may explain the mechanisms underlying common symptoms and increased activity in certain brain regions in migraine patients.
In a new study, the prevalence of dizziness was 65% among patients with migraine who underwent a virtual roller-coaster ride versus 30% among those without migraine. In addition, imaging showed greater neuronal activity after the simulation in those with migraine.
“Migraine patients reported more dizziness and motion sickness, as well as longer symptom duration and intensity, in a virtual roller-coaster ride,” even though the videos and timing were identical for both groups, said study investigator Arne May, MD, PhD, professor of neurology at the University of Hamburg (Germany).
“We found differences not just in behavioral results but also in specific activations of areas within the cerebellum and the frontal gyrus. Migraine patients process such visual input differently from controls and activate a specific brain network to do so,” he added.
The findings were published online July 21, 2021, in Neurology.
The brain’s response
Nausea, which is among the diagnostic criteria for migraine, is the main symptom of motion sickness. Vestibular symptoms such as dizziness are also components of migraine.
Previous research has examined how the brain processes visual and motion stimuli in migraine, but the reasons patients with migraine are susceptible to motion sickness and dizziness remain unclear.
The researchers used a simulated roller-coaster ride to study the clinical and brain responses to motion among participants with and participants without migraine. They enrolled 20 consecutive patients with migraine who presented to a tertiary headache clinic between January and March 2020 and enrolled 20 healthy participants from a university hospital and the community. The average age of the study population was 30 years, and more than 80% were women.
In response to a questionnaire, participants provided information about demographics and headache features, including onset, frequency, and intensity. They also provided information about their status within the migraine phase and about vestibular symptoms experienced in daily life.
While undergoing functional MRI (fMRI), all participants watched two short videos that provided a first-person perspective of a roller-coaster ride. During the videos, they wore ear buds that conveyed the sound of a car riding over the rails.
The first video included more horizontal perspectives, and the second had more vertical perspectives. Each video was shown three times in random order.
During fMRI, participants reported intensity of nausea and vestibular symptoms using an 11-point Likert scale. After the experiment, they responded to a questionnaire that evaluated intensity and duration of nausea, dizziness, and vertigo experienced during the videos.
Participants also were given the Simulator Sickness Questionnaire (SSQ), which assessed motion sickness. A 100-point visual analog scale (VAS) was used to rate how realistic the roller-coaster experience had been.
There were no differences in sex or age between the migraine group and the healthy control group. Half of the patients with migraine reported aura. The mean number of migraine attacks within the previous month was 3.7. The mean Migraine Disability Assessment score was 21.5, which indicates severe disability.
Nausea, dizziness often neglected
Baseline prevalence of vestibular symptoms was 75% in the migraine group and 5% in the control group (P < .0001). These symptoms included dizziness (60% and 5%, respectively; P < .0001) and postural symptoms (40% and 0%, respectively; P = .003).
At baseline, vestibular symptoms were more frequent (P = .001), more intense (P < .0001), and were associated with greater disability (P = .001) in patients with migraine, compared with participants without migraine. The patients with migraine were also more susceptible to motion sickness (P = .02) and had higher depression scores (P = .001).
During the roller-coaster simulation, dizziness was more prevalent among patients with migraine than among those without migraine (65% vs. 30%; P = .03). Patients with migraine also reported more motion sickness (SSQ score, 47.3 vs. 24.3; P = .004), longer symptom duration (1:19 minutes vs. 00:27 minutes; P = .03), and symptoms of greater intensity (VAS, 22.0 vs. 9.9; P = .03).
Brain activity also differed between groups. Among patients with migraine, neuronal activity was greater in clusters within the right superior and left inferior occipital gyrus, the left pontine nuclei, and the left cerebellar lobules V and VI.
There was a moderately negative correlation of activation of the inferior occipital gyrus with migraine disability (r = –0.46; P = .04). Activation within the pontine nuclei correlated positively with motion sickness scores (r = 0.32; P = .04).
In addition, among patients with migraine, activity in the cerebellar lobule VIIb and in the left middle frontal gyrus was decreased in comparison with persons without migraine. Also among patients with migraine, there was enhanced connectivity between the pontine nuclei, cerebellar areas V and VI, and the interior and superior occipital gyrus and numerous cortical areas.
Clinicians often neglect to treat dizziness and nausea in patients with migraine, said Dr. May. However, these symptoms are part of migraine, even when attacks are not occurring.
“I have learned that if we can explain such symptoms, they are better accepted,” said Dr. May. “We need more and better basic research because we need to understand before we treat.”
Toward faster, more effective treatment
Commenting on the study, Erik Viirre, MD, PhD, professor in the department of neurosciences, University of California, San Diego, said, “we can be excited and celebrate that these researchers are using these news tools to investigate the operation of the migraine brain.
“That will combine with the new therapies and the genomics to give us a powerful approach to this particular condition,” said Dr. Viirre, who was not involved with the research.
The findings provide significant detail about the interconnections between the various brain regions affected by migraine, he noted. These regions include not just the sensory centers but also areas involved in higher executive function and emotional responses.
By identifying these regions, the findings show “some of the underlying mechanisms of these clinically relevant features,” said Dr. Viirre, who is also director of UCSD’s Arthur C. Clarke Center for Human Imagination.
The investigators set up the motion simulation well and used sound fMRI methodology, he added. However, imaging studies of the brain’s response to motion pose several challenges.
“The biggest challenge in any of these circumstances is that you can’t put an actual fMRI scanner on a roller-coaster,” said Dr. Viirre. “The actual acceleration and gravitational sensations delivered by a roller-coaster and gravity, of course, do not occur when you’re lying still in an MRI scanner.” Nevertheless, the pseudoacceleration produced by a visual stimulus is a reasonable proxy.
The findings also suggest that researchers in the future could examine whether any new therapeutic interventions for migraine modulate the brain functions differently for individuals with migraine than for those without migraine, he noted.
“That’s going to lead us to a faster, more effective, more reliable suite of migraine therapies,” said Dr. Viirre.
The study also reminds clinicians to take a broader approach to patients with migraine, and it underscores the value of strategies such as self-calming techniques, which can reduce the number and intensity of headaches, he said.
“Literally demonstrating these functional differences in the migraine brain is a hugely important message of advocacy for people with migraine,” Dr. Viirre concluded.
The study was funded by the German Research Foundation. Drs. May and Viirre have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
and may explain the mechanisms underlying common symptoms and increased activity in certain brain regions in migraine patients.
In a new study, the prevalence of dizziness was 65% among patients with migraine who underwent a virtual roller-coaster ride versus 30% among those without migraine. In addition, imaging showed greater neuronal activity after the simulation in those with migraine.
“Migraine patients reported more dizziness and motion sickness, as well as longer symptom duration and intensity, in a virtual roller-coaster ride,” even though the videos and timing were identical for both groups, said study investigator Arne May, MD, PhD, professor of neurology at the University of Hamburg (Germany).
“We found differences not just in behavioral results but also in specific activations of areas within the cerebellum and the frontal gyrus. Migraine patients process such visual input differently from controls and activate a specific brain network to do so,” he added.
The findings were published online July 21, 2021, in Neurology.
The brain’s response
Nausea, which is among the diagnostic criteria for migraine, is the main symptom of motion sickness. Vestibular symptoms such as dizziness are also components of migraine.
Previous research has examined how the brain processes visual and motion stimuli in migraine, but the reasons patients with migraine are susceptible to motion sickness and dizziness remain unclear.
The researchers used a simulated roller-coaster ride to study the clinical and brain responses to motion among participants with and participants without migraine. They enrolled 20 consecutive patients with migraine who presented to a tertiary headache clinic between January and March 2020 and enrolled 20 healthy participants from a university hospital and the community. The average age of the study population was 30 years, and more than 80% were women.
In response to a questionnaire, participants provided information about demographics and headache features, including onset, frequency, and intensity. They also provided information about their status within the migraine phase and about vestibular symptoms experienced in daily life.
While undergoing functional MRI (fMRI), all participants watched two short videos that provided a first-person perspective of a roller-coaster ride. During the videos, they wore ear buds that conveyed the sound of a car riding over the rails.
The first video included more horizontal perspectives, and the second had more vertical perspectives. Each video was shown three times in random order.
During fMRI, participants reported intensity of nausea and vestibular symptoms using an 11-point Likert scale. After the experiment, they responded to a questionnaire that evaluated intensity and duration of nausea, dizziness, and vertigo experienced during the videos.
Participants also were given the Simulator Sickness Questionnaire (SSQ), which assessed motion sickness. A 100-point visual analog scale (VAS) was used to rate how realistic the roller-coaster experience had been.
There were no differences in sex or age between the migraine group and the healthy control group. Half of the patients with migraine reported aura. The mean number of migraine attacks within the previous month was 3.7. The mean Migraine Disability Assessment score was 21.5, which indicates severe disability.
Nausea, dizziness often neglected
Baseline prevalence of vestibular symptoms was 75% in the migraine group and 5% in the control group (P < .0001). These symptoms included dizziness (60% and 5%, respectively; P < .0001) and postural symptoms (40% and 0%, respectively; P = .003).
At baseline, vestibular symptoms were more frequent (P = .001), more intense (P < .0001), and were associated with greater disability (P = .001) in patients with migraine, compared with participants without migraine. The patients with migraine were also more susceptible to motion sickness (P = .02) and had higher depression scores (P = .001).
During the roller-coaster simulation, dizziness was more prevalent among patients with migraine than among those without migraine (65% vs. 30%; P = .03). Patients with migraine also reported more motion sickness (SSQ score, 47.3 vs. 24.3; P = .004), longer symptom duration (1:19 minutes vs. 00:27 minutes; P = .03), and symptoms of greater intensity (VAS, 22.0 vs. 9.9; P = .03).
Brain activity also differed between groups. Among patients with migraine, neuronal activity was greater in clusters within the right superior and left inferior occipital gyrus, the left pontine nuclei, and the left cerebellar lobules V and VI.
There was a moderately negative correlation of activation of the inferior occipital gyrus with migraine disability (r = –0.46; P = .04). Activation within the pontine nuclei correlated positively with motion sickness scores (r = 0.32; P = .04).
In addition, among patients with migraine, activity in the cerebellar lobule VIIb and in the left middle frontal gyrus was decreased in comparison with persons without migraine. Also among patients with migraine, there was enhanced connectivity between the pontine nuclei, cerebellar areas V and VI, and the interior and superior occipital gyrus and numerous cortical areas.
Clinicians often neglect to treat dizziness and nausea in patients with migraine, said Dr. May. However, these symptoms are part of migraine, even when attacks are not occurring.
“I have learned that if we can explain such symptoms, they are better accepted,” said Dr. May. “We need more and better basic research because we need to understand before we treat.”
Toward faster, more effective treatment
Commenting on the study, Erik Viirre, MD, PhD, professor in the department of neurosciences, University of California, San Diego, said, “we can be excited and celebrate that these researchers are using these news tools to investigate the operation of the migraine brain.
“That will combine with the new therapies and the genomics to give us a powerful approach to this particular condition,” said Dr. Viirre, who was not involved with the research.
The findings provide significant detail about the interconnections between the various brain regions affected by migraine, he noted. These regions include not just the sensory centers but also areas involved in higher executive function and emotional responses.
By identifying these regions, the findings show “some of the underlying mechanisms of these clinically relevant features,” said Dr. Viirre, who is also director of UCSD’s Arthur C. Clarke Center for Human Imagination.
The investigators set up the motion simulation well and used sound fMRI methodology, he added. However, imaging studies of the brain’s response to motion pose several challenges.
“The biggest challenge in any of these circumstances is that you can’t put an actual fMRI scanner on a roller-coaster,” said Dr. Viirre. “The actual acceleration and gravitational sensations delivered by a roller-coaster and gravity, of course, do not occur when you’re lying still in an MRI scanner.” Nevertheless, the pseudoacceleration produced by a visual stimulus is a reasonable proxy.
The findings also suggest that researchers in the future could examine whether any new therapeutic interventions for migraine modulate the brain functions differently for individuals with migraine than for those without migraine, he noted.
“That’s going to lead us to a faster, more effective, more reliable suite of migraine therapies,” said Dr. Viirre.
The study also reminds clinicians to take a broader approach to patients with migraine, and it underscores the value of strategies such as self-calming techniques, which can reduce the number and intensity of headaches, he said.
“Literally demonstrating these functional differences in the migraine brain is a hugely important message of advocacy for people with migraine,” Dr. Viirre concluded.
The study was funded by the German Research Foundation. Drs. May and Viirre have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Infusion shown effective for acquired von Willebrand disease
Acquired von Willebrand disease (aVWD) is a rare and serious condition associated with lymphoproliferative disorders, malignancy, autoimmune disorders, and cardiovascular disease. It is most commonly caused by monoclonal gammopathy of undetermined significance (MGUS), which acts to clear von Willebrand factor from the patient’s bloodstream. However, a continuous-infusion of plasma-derived von Willebrand factor (VWF) concentrate provided adequate hemostasis in aVWD resulting from MGUS, according to Kathryn E. Dane, PharmD, of Johns Hopkins University, Baltimore, and colleagues.
The infusion rapidly achieved target ristocetin cofactor activity with or without intravenous immunoglobulin in three patients, as detailed in the report published online in Blood Advances.
The three consecutive patients with aVWD were treated with plasma-derived VWF concentrate administered for periprocedural optimization (patient 1, an 85-year old woman) or to treat bleeding episodes (patient 2, an 88-year-old man; and patient 3, a 53-year-old woman). Factor VIII activity was measured via a 1-stage clotting test and von Willebrand factor activity was measured with a ristocetin cofactor assay.
Promising results
All three patients demonstrated increased VWF ristocetin cofactor and factor VIII activities within hours of initiation of the continuous infusion concentrate, according to the report.
“We hypothesize that the efficacy of CI VWF concentrate in aVWD may be related to continuous provision of VWF, allowing binding and neutralization of anti-VWF IgG antibodies, and providing adequate circulating unbound VWF for appropriate hemostatic efficacy,” the researchers concluded.
The authors reported that they had no competing financial interests.
Acquired von Willebrand disease (aVWD) is a rare and serious condition associated with lymphoproliferative disorders, malignancy, autoimmune disorders, and cardiovascular disease. It is most commonly caused by monoclonal gammopathy of undetermined significance (MGUS), which acts to clear von Willebrand factor from the patient’s bloodstream. However, a continuous-infusion of plasma-derived von Willebrand factor (VWF) concentrate provided adequate hemostasis in aVWD resulting from MGUS, according to Kathryn E. Dane, PharmD, of Johns Hopkins University, Baltimore, and colleagues.
The infusion rapidly achieved target ristocetin cofactor activity with or without intravenous immunoglobulin in three patients, as detailed in the report published online in Blood Advances.
The three consecutive patients with aVWD were treated with plasma-derived VWF concentrate administered for periprocedural optimization (patient 1, an 85-year old woman) or to treat bleeding episodes (patient 2, an 88-year-old man; and patient 3, a 53-year-old woman). Factor VIII activity was measured via a 1-stage clotting test and von Willebrand factor activity was measured with a ristocetin cofactor assay.
Promising results
All three patients demonstrated increased VWF ristocetin cofactor and factor VIII activities within hours of initiation of the continuous infusion concentrate, according to the report.
“We hypothesize that the efficacy of CI VWF concentrate in aVWD may be related to continuous provision of VWF, allowing binding and neutralization of anti-VWF IgG antibodies, and providing adequate circulating unbound VWF for appropriate hemostatic efficacy,” the researchers concluded.
The authors reported that they had no competing financial interests.
Acquired von Willebrand disease (aVWD) is a rare and serious condition associated with lymphoproliferative disorders, malignancy, autoimmune disorders, and cardiovascular disease. It is most commonly caused by monoclonal gammopathy of undetermined significance (MGUS), which acts to clear von Willebrand factor from the patient’s bloodstream. However, a continuous-infusion of plasma-derived von Willebrand factor (VWF) concentrate provided adequate hemostasis in aVWD resulting from MGUS, according to Kathryn E. Dane, PharmD, of Johns Hopkins University, Baltimore, and colleagues.
The infusion rapidly achieved target ristocetin cofactor activity with or without intravenous immunoglobulin in three patients, as detailed in the report published online in Blood Advances.
The three consecutive patients with aVWD were treated with plasma-derived VWF concentrate administered for periprocedural optimization (patient 1, an 85-year old woman) or to treat bleeding episodes (patient 2, an 88-year-old man; and patient 3, a 53-year-old woman). Factor VIII activity was measured via a 1-stage clotting test and von Willebrand factor activity was measured with a ristocetin cofactor assay.
Promising results
All three patients demonstrated increased VWF ristocetin cofactor and factor VIII activities within hours of initiation of the continuous infusion concentrate, according to the report.
“We hypothesize that the efficacy of CI VWF concentrate in aVWD may be related to continuous provision of VWF, allowing binding and neutralization of anti-VWF IgG antibodies, and providing adequate circulating unbound VWF for appropriate hemostatic efficacy,” the researchers concluded.
The authors reported that they had no competing financial interests.
FROM BLOOD ADVANCES
Internal mammary lymph node radiation safe over the long term
After a median follow-up 15.7 years among almost 4,000 women, for half of patients who received postoperative internal mammary and medial supraclavicular (IM-MS) lymph node irradiation, the “absolute rates and differences” of heart and lung complications “were very low, with no increased non–breast cancer–related mortality, even before introducing heart-sparing techniques,” said the investigators.
The findings come from the European Organization for Research and Treatment of Cancer (EORTC) trial. The investigators were led by Philip Poortmans, MD, PhD, a radiation oncologist at the University of Antwerp (Belgium).
The team had previously reported lower breast cancer mortality and breast cancer recurrence rates in the radiation group.
Women in the trial were treated from 1996 to 2004. “We expect that with contemporary volume-based radiation therapy outcomes will be even better, by improved coverage of target volumes, more homogeneous dose delivery and decreased doses to nontarget tissues,” the team wrote.
In the end, “our findings ... have important – reassuring – consequences for decision-making concerning elective lymph node treatment in breast cancer,” the researchers commented.
The study was published online on July 28, 2021, in the Journal of the National Cancer Institute.
Resolving the debate
There’s been debate for decades on whether the long-term risk associated with nodal irradiation, particularly collateral heart and lung damage from internal mammary irradiation, outweigh the benefits of better disease control, Julia White, MD, a radiation oncologist at the Ohio State University Breast Center, Columbus, noted in an accompanying editorial.
Concerns stem originally from trials conducted from the 1950s to the 1970s. In those trials, higher doses of radiation were delivered to the internal mammary node with far less precision than today. Subsequent studies have not laid the worry to rest, and protocols vary across institutions, Dr. White explained. Some treat IM nodes in high-risk patients, but others only treat the axilla and the medial supraclavicular lymph nodes.
Dr. White said the new EORTC trial “moves us one step closer to resolving the debate about the value of internal mammary nodal [IMN] radiation.”
She noted that, since 2014, advances in the field have led to an almost 50% reduction in cardiac radiation exposure during breast cancer treatment. Current guidelines recommend that internal mammary nodes “should generally be treated” as part of postmastectomy radiotherapy but that cardiopulmonary complications are still possible even with improved techniques, she wrote.
Mostly grade 1 morbidity
Women in the study had stage I-III breast cancer with axillary node involvement and/or medially located primary tumors. The median age at study entry was 54 years. The patients were treated at 46 centers in 13 countries.
The group that received IM-MS irradiation after surgery received 50 Gy in 25 fractions over 5 weeks.
The cumulative 15-year incidence of lung fibrosis was 5.7% among treated women versus 2.9% among control patients. The incidence of cardiac fibrosis was 1.9% with treatment, versus 1.1%.
The incidence of any cardiac disease was 11.1% in the radiation arm versus 9.4% in the control group.
Complications were mostly of grade 1. The only statistically significant difference in rates of events of grade 2 or higher was in the incidence of pulmonary morbidity, which was 0.8% with radiation versus 0.1% without. There were no differences in the incidence of second malignancies, contralateral breast cancer cases, or cardiovascular deaths with IMN irradiation.
The authors noted that their results conflict with a 2013 study that found a relative increase in major coronary events of 7.4% per Gy mean heart dose. The women in that trial were treated in Sweden and Denmark between 1958 and 2001.
Dr. Poortmans and colleagues noted, however, that this 2013 study and others found a proportional and not an absolute increase in risk. With a baseline risk of 10%, for instance, a 7% increase per 1 Gy translates to a total risk of 10.07%.
Also, no increased risk has been reported in more recently published trials, and a meta-analysis found no increase in non–breast cancer–related mortality with trials that began after 1988.
Still, “it seems logical to take the preexisting cardiac comorbidity of patients into consideration,” the investigators concluded. For patients with higher baseline cardiopulmonary risk factors, lower mean heart doses should be used, and such patients should undergo longer-term follow-up.
The study was funded by La Ligue Nationale Contre Le Cancer and the KWF Kanker Bestrijding from the Netherlands. The investigators and Dr. White disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
After a median follow-up 15.7 years among almost 4,000 women, for half of patients who received postoperative internal mammary and medial supraclavicular (IM-MS) lymph node irradiation, the “absolute rates and differences” of heart and lung complications “were very low, with no increased non–breast cancer–related mortality, even before introducing heart-sparing techniques,” said the investigators.
The findings come from the European Organization for Research and Treatment of Cancer (EORTC) trial. The investigators were led by Philip Poortmans, MD, PhD, a radiation oncologist at the University of Antwerp (Belgium).
The team had previously reported lower breast cancer mortality and breast cancer recurrence rates in the radiation group.
Women in the trial were treated from 1996 to 2004. “We expect that with contemporary volume-based radiation therapy outcomes will be even better, by improved coverage of target volumes, more homogeneous dose delivery and decreased doses to nontarget tissues,” the team wrote.
In the end, “our findings ... have important – reassuring – consequences for decision-making concerning elective lymph node treatment in breast cancer,” the researchers commented.
The study was published online on July 28, 2021, in the Journal of the National Cancer Institute.
Resolving the debate
There’s been debate for decades on whether the long-term risk associated with nodal irradiation, particularly collateral heart and lung damage from internal mammary irradiation, outweigh the benefits of better disease control, Julia White, MD, a radiation oncologist at the Ohio State University Breast Center, Columbus, noted in an accompanying editorial.
Concerns stem originally from trials conducted from the 1950s to the 1970s. In those trials, higher doses of radiation were delivered to the internal mammary node with far less precision than today. Subsequent studies have not laid the worry to rest, and protocols vary across institutions, Dr. White explained. Some treat IM nodes in high-risk patients, but others only treat the axilla and the medial supraclavicular lymph nodes.
Dr. White said the new EORTC trial “moves us one step closer to resolving the debate about the value of internal mammary nodal [IMN] radiation.”
She noted that, since 2014, advances in the field have led to an almost 50% reduction in cardiac radiation exposure during breast cancer treatment. Current guidelines recommend that internal mammary nodes “should generally be treated” as part of postmastectomy radiotherapy but that cardiopulmonary complications are still possible even with improved techniques, she wrote.
Mostly grade 1 morbidity
Women in the study had stage I-III breast cancer with axillary node involvement and/or medially located primary tumors. The median age at study entry was 54 years. The patients were treated at 46 centers in 13 countries.
The group that received IM-MS irradiation after surgery received 50 Gy in 25 fractions over 5 weeks.
The cumulative 15-year incidence of lung fibrosis was 5.7% among treated women versus 2.9% among control patients. The incidence of cardiac fibrosis was 1.9% with treatment, versus 1.1%.
The incidence of any cardiac disease was 11.1% in the radiation arm versus 9.4% in the control group.
Complications were mostly of grade 1. The only statistically significant difference in rates of events of grade 2 or higher was in the incidence of pulmonary morbidity, which was 0.8% with radiation versus 0.1% without. There were no differences in the incidence of second malignancies, contralateral breast cancer cases, or cardiovascular deaths with IMN irradiation.
The authors noted that their results conflict with a 2013 study that found a relative increase in major coronary events of 7.4% per Gy mean heart dose. The women in that trial were treated in Sweden and Denmark between 1958 and 2001.
Dr. Poortmans and colleagues noted, however, that this 2013 study and others found a proportional and not an absolute increase in risk. With a baseline risk of 10%, for instance, a 7% increase per 1 Gy translates to a total risk of 10.07%.
Also, no increased risk has been reported in more recently published trials, and a meta-analysis found no increase in non–breast cancer–related mortality with trials that began after 1988.
Still, “it seems logical to take the preexisting cardiac comorbidity of patients into consideration,” the investigators concluded. For patients with higher baseline cardiopulmonary risk factors, lower mean heart doses should be used, and such patients should undergo longer-term follow-up.
The study was funded by La Ligue Nationale Contre Le Cancer and the KWF Kanker Bestrijding from the Netherlands. The investigators and Dr. White disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
After a median follow-up 15.7 years among almost 4,000 women, for half of patients who received postoperative internal mammary and medial supraclavicular (IM-MS) lymph node irradiation, the “absolute rates and differences” of heart and lung complications “were very low, with no increased non–breast cancer–related mortality, even before introducing heart-sparing techniques,” said the investigators.
The findings come from the European Organization for Research and Treatment of Cancer (EORTC) trial. The investigators were led by Philip Poortmans, MD, PhD, a radiation oncologist at the University of Antwerp (Belgium).
The team had previously reported lower breast cancer mortality and breast cancer recurrence rates in the radiation group.
Women in the trial were treated from 1996 to 2004. “We expect that with contemporary volume-based radiation therapy outcomes will be even better, by improved coverage of target volumes, more homogeneous dose delivery and decreased doses to nontarget tissues,” the team wrote.
In the end, “our findings ... have important – reassuring – consequences for decision-making concerning elective lymph node treatment in breast cancer,” the researchers commented.
The study was published online on July 28, 2021, in the Journal of the National Cancer Institute.
Resolving the debate
There’s been debate for decades on whether the long-term risk associated with nodal irradiation, particularly collateral heart and lung damage from internal mammary irradiation, outweigh the benefits of better disease control, Julia White, MD, a radiation oncologist at the Ohio State University Breast Center, Columbus, noted in an accompanying editorial.
Concerns stem originally from trials conducted from the 1950s to the 1970s. In those trials, higher doses of radiation were delivered to the internal mammary node with far less precision than today. Subsequent studies have not laid the worry to rest, and protocols vary across institutions, Dr. White explained. Some treat IM nodes in high-risk patients, but others only treat the axilla and the medial supraclavicular lymph nodes.
Dr. White said the new EORTC trial “moves us one step closer to resolving the debate about the value of internal mammary nodal [IMN] radiation.”
She noted that, since 2014, advances in the field have led to an almost 50% reduction in cardiac radiation exposure during breast cancer treatment. Current guidelines recommend that internal mammary nodes “should generally be treated” as part of postmastectomy radiotherapy but that cardiopulmonary complications are still possible even with improved techniques, she wrote.
Mostly grade 1 morbidity
Women in the study had stage I-III breast cancer with axillary node involvement and/or medially located primary tumors. The median age at study entry was 54 years. The patients were treated at 46 centers in 13 countries.
The group that received IM-MS irradiation after surgery received 50 Gy in 25 fractions over 5 weeks.
The cumulative 15-year incidence of lung fibrosis was 5.7% among treated women versus 2.9% among control patients. The incidence of cardiac fibrosis was 1.9% with treatment, versus 1.1%.
The incidence of any cardiac disease was 11.1% in the radiation arm versus 9.4% in the control group.
Complications were mostly of grade 1. The only statistically significant difference in rates of events of grade 2 or higher was in the incidence of pulmonary morbidity, which was 0.8% with radiation versus 0.1% without. There were no differences in the incidence of second malignancies, contralateral breast cancer cases, or cardiovascular deaths with IMN irradiation.
The authors noted that their results conflict with a 2013 study that found a relative increase in major coronary events of 7.4% per Gy mean heart dose. The women in that trial were treated in Sweden and Denmark between 1958 and 2001.
Dr. Poortmans and colleagues noted, however, that this 2013 study and others found a proportional and not an absolute increase in risk. With a baseline risk of 10%, for instance, a 7% increase per 1 Gy translates to a total risk of 10.07%.
Also, no increased risk has been reported in more recently published trials, and a meta-analysis found no increase in non–breast cancer–related mortality with trials that began after 1988.
Still, “it seems logical to take the preexisting cardiac comorbidity of patients into consideration,” the investigators concluded. For patients with higher baseline cardiopulmonary risk factors, lower mean heart doses should be used, and such patients should undergo longer-term follow-up.
The study was funded by La Ligue Nationale Contre Le Cancer and the KWF Kanker Bestrijding from the Netherlands. The investigators and Dr. White disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Myasthenic Crisis After Recurrent COVID-19 Infection
A patient with myasthenia gravis who survived 2 COVID-19 infections required plasmapheresis to recover from an acute crisis.
COVID-19 is still in the early stages of understanding, although it is known to be complicated by individual patient comorbidities. The management and treatment of COVID-19 continues to quickly evolve as more is discovered regarding the virus. Multiple treatments have been preliminarily tested and used under a Food and Drug Administration emergency use authorization (EUA) determination. The long-term success of these therapies, however, is yet to be determined. Additionally, if a patient has a second clinical presentation for COVID-19, it is not known whether this represents latency with subsequent reactivation from the previous infection or a second de novo infection. The uncertainty calls into question the duration of immunity, if any, following a primary infection.
COVID-19 management becomes more complicated when patients have complex medical conditions, such as myasthenia gravis (MG). This autoimmune neuromuscular disorder can present with varying weakness, and many patients are on immunomodulator medications. The weakness can worsen into a myasthenic crisis (MC), resulting in profound weakness of the respiratory muscles. Therefore, patients with MG are at increased risk for COVID-19 and may have a more complicated course when infected.
Our patient with MG presented for severe COVID-19 symptoms twice and later developed MC. He received 2 treatment modalities available under an EUA (remdesivir and convalescent plasma) for COVID-19, resulting in symptom resolution and a negative polymerize chain reaction (PCR) test result for the virus. However, after receiving his typical maintenance therapy of IV immunoglobulin (IVIG) for his MG, he again developed symptoms consistent with COVID-19 and tested positive. After recovering from the second episode of COVID-19, the patient went into MC requiring plasmapheresis.
Case Presentation
A 56-year-old male, US Army veteran presented to Carl R. Darnall Army Medical Center emergency department (ED) 6 days after testing positive for COVID-19, with worsening sputum, cough, congestion, dyspnea, and fever. Due to his MG, the patient had a home oxygen monitor and reported that his oxygenation saturation dropped below 90% with minimal exertion. His medical history was significant for MG, status postthymectomy and radiation treatment, left hemidiaphragm paralysis secondary to phrenic nerve injury, and corticosteroid-induced insulin-dependent diabetes mellitus. His current home medications included pyridostigmine 60 mg 3 times a day, mycophenolate (MMF) 1500 mg twice daily, IV immunoglobulin (IVIG) every 3 weeks, insulin aspart up to 16 U per meal, insulin glargine 30 U twice a day, dulaglutide 0.75 mg every week, and metformin 1000 mg twice daily.
On initial examination, the patient’s heart rate (HR) was 111 beats/min, respiratory rate (RR), 22 breaths/min, blood pressure (BP), 138/88 mm Hg, temperature, 100.9 oF, and his initial pulse oximetry, 91% on room air. On physical examination, the patient was tachypneic, though without other signs of respiratory distress. Lung auscultation revealed no adventitial lung sounds. His cardiac examination was notable only for tachycardia. His neurologic examination demonstrated intact cranial nerves, with 5 out of 5 (scale 1 to 5) strength throughout the upper and lower extremities, sensation was intact to light touch, and he had normal cerebellar function. The rest of the examination was normal.
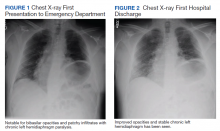
Initial laboratory investigation was notable for a white blood cell count of 14.15x103 cells/mcL with 84% neutrophils, and 6% lymphocytes. Additional tests revealed a C-reactive protein (CRP) level, 17.97 mg/dL (reference range, 0-0.5 mg/dL), ferritin level, 647 ng/mL (reference range, 22-274 ng/mL), d-dimer, 0.64 mcg/mL (reference range, 0-0.47mcg/mL), and a repeated positive COVID-19 PCR test. A portable chest X-ray showed bibasilar opacities (Figure 1).
The patient was diagnosed with COVID-19 and admitted to the intensive care unit (ICU). In the ICU, the patient received 1 U of convalescent plasma (CP) and started on a course of IV remdesivir 100 mg/d consistent with the EUA. He also received a 5-day course of ceftriaxone and azithromycin for possible community acquired pneumonia (CAP). As part of the patient’s MG maintenance medications, he received IVIG 4 g while in the ICU. Throughout his ICU stay, he required supplemental nasal cannula oxygenation to maintain his oxygen saturation > 93%. After 8 days in the ICU, his oxygen requirements decreased, and the patient was transferred out of the ICU and remdesivir was discontinued. On hospital day 10, a repeat COVID-19 PCR test was negative, inflammatory markers returned to within normal limits, and a repeat chest X-ray showed improvement from admission (Figure 2). Having recovered significantly, he was discharged home.
Three weeks later, the patient again presented to the MTF with 3 days of dyspnea, cough, fever, nausea, and vomiting. One day before symptom onset, he had received his maintenance IVIG infusion. The patient reported that his home oxygen saturation was 82% with minimal exertion. On ED presentation his HR was 107 beats/min, RR, 28 breaths/min, temperature, 98.1 oF, BP 118/71 mm Hg, and oxygen saturation, 92% on 2L nasal cannula. His examination was most notable for tachypnea with accessory muscle use. At this time, his neurologic examination was unchanged from prior admission with grossly intact cranial nerves and symmetric 5 of 5 motor strength in all extremities.
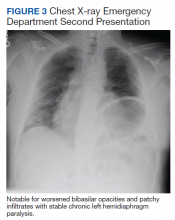
At this second ED visit, laboratory results demonstrated a CRP of 3.44 mg/dL, ferritin 2019 ng/mL, d-dimer, 3.39 mcg/mL, and a positive COVID-19 PCR result. His chest X-ray demonstrated new peripheral opacities compared with the X-ray at discharge (Figure 3). He required ICU admission again for his COVID-19 symptoms.
During his ICU course he continued to require supplemental oxygen by nasal cannula, though never required intubation. This second admission, he was again treated empirically for CAP with levofloxacin 750 mg daily for 5 days. He was discharged after 14 days with symptom resolution and down trending of inflammatory markers, though he was not retested for COVID-19.
Four days after his second discharge, he presented to the ED for a third time with diffuse weakness, dysphagia, and dysarthria of 1 day. His HR was 87/beats/min; RR, 17 breaths/min; temperature, 98.7 oF; BP, 144/81 mm Hg; and oxygen saturation, 98% on room air. His examination was significant for slurred speech, bilateral ptosis, 3 of 5 strength in bilateral finger flexion/abduction, wrist extension, knee and ankle flexion/extension; 4 of 5 strength in bilateral proximal muscle testing of deltoid, and hip; normal sensation, cerebellar function and reflexes. His negative inspiratory force (NIF) maximal effort was −30 cmH2O. He was determined to be in MC without evidence of COIVD-19 symptoms, and laboratory results were within normal limits, including a negative COVID-19 PCR. As he received IVIG as maintenance therapy, plasmapheresis was recommended to treat his MC, which required transfer to an outside civilian facility.
At the outside hospital, the patient underwent 5 rounds of plasmapheresis over 10 days. By the third treatment his strength had returned with resolution of the bulbar symptoms and no supplemental oxygen requirements. The patient was discharged and continued his original dosages of MMF and pyridostigmine. At 3 months, he remained asymptomatic from a COVID-19 standpoint and stable from a MG standpoint.
Discussion
Reinfection with the COVID-19 has been continuously debated with alternative explanations suggested for a positive test after a previous negative PCR test in the setting of symptom resolution.1,2 Proposed causes include dynamic PCR results due to prolonged viral shedding and inaccurate or poorly sensitive tests. The repeat positive cases in these scenarios, however, occurred in asymptomatic patients.1,2 COVID-19 shedding averages 20 to 22 days after symptom onset but has been seen up to 36 days after symptom resolution.2,3 This would suggest that fluctuating results during the immediate postsymptom period may be due to variations in viral shedding load and or sampling error—especially in asymptomatic patients. On the other hand, patients who experience return of symptoms days to weeks after previous convalescence leave clinicians wondering whether this represents clinical latency with reactivation or COVID-19 reinfection. A separate case of initial COVID-19 in a patient that had subsequent clinical recovery with a negative PCR developed recurrent respiratory symptoms and had a positive PCR test only 10 days later, further highlighting the reinfection vs reactivation issue of COVID-19.2 Further understanding of this issue may have implications on the extent of natural immunity following primary infection; potential vaccine dosage schedules; and global public health policies.
Although reactivation may be plausible given his immunomodulatory therapy, our patient’s second COVID-19 symptoms started 40 days after the initial symptoms, and 26 days after the initial course resolution; previous cases of return of severe symptoms occurred between 3 and 6 days.1 Given our patient’s time course between resolution and return of symptoms, if latency is the mechanism at play, this case demonstrates an exceptionally longer latency period than the ones that have been reported. Additionally, if latency is an issue in COVID-19, using remdesivir as a treatment further complicates the understanding of this disease.
Remdesivir, a nucleoside analogue antiviral, was shown to benefit recovery in patients with severe symptoms in the Adaptive COVID-19 Treatment Trial-1 study.4 Our patient had originally been placed on a 10-day course; however, on treatment day 8, his symptoms resolved and the remdesivir was discontinued. This is a similar finding to half the patients in the 10-day arm of the study by McCreary and colleagues.5 Although our patient was asymptomatic 4 weeks after the start of remdesivir, consistent with the majority of patients in the McCreary 10-day study arm, further comparison of the presented patient is limited due to study length and follow-up considerations.5 No previous data exist on reactivation, reinfection, or long-term mortality after being treated with remdesivir for COVID-19 infection.
IVIG is being studied in the treatment of COVID-19 and bears consideration as it relates to our patient. There is no evidence that IVIG used in the treatment of autoimmune diseases increases the risk of infection compared with that of other medications used in the treatment of such diseases. Furthermore, the current guidance from the MG expert panel does not suggest that IVIG increases the risk of contracting COVID-19 aside from the risks of exposure to hospital infrastructure.6 Yet the guidance does not discuss the use of IVIG for MG in patients who are already symptomatic from COVID-19 or for patients recovering from the clinical disease or does it discuss a possible compounding risk of thromboembolic events associated with IVIG and COVID-19.6,7 Our patient received his maintenance IVIG during his first admission without any worsening of symptoms or increased oxygen requirements. The day following our patient’s next scheduled IVIG infusion—while asymptomatic—he again developed respiratory symptoms; this could suggest that IVIG did not contribute to his second clinical course nor protect against.
CP is a treatment modality that has been used and studied in previous infectious outbreaks such as the first severe acute respiratory syndrome, and the H1N1 influenza virus.8 Current data on CP for COVID-19 are limited, but early descriptive studies have shown a benefit in improvement of symptoms 5 days sooner in those requiring supplemental oxygen, but no benefit for those requiring mechanical ventilation.9 Like patients that benefitted in these studies, our patient received CP early, 6 days after first testing positive and onset of symptoms. This patient’s reinfection or return of symptoms draws into question the hindrance or even prevention of long-term immunity from administration of CP.
COVID-19 presents many challenges when managing this patient’s coexisting MG, especially as the patient was already being treated with immunosuppressing therapies. The guidance does recommend continuation of standard MG therapies during hospitalizations, including immunosuppression medications such as MMF.6 Immunosuppression is associated with worsened severity of COVID-19 symptoms, although no relation exists to degree of immunosuppression and severity.7,10 To the best of our knowledge there has been no case report of reinfection or reactivation of COVID-19 associated with immunosuppressive agents used in the treatment of MG.
Our patient also was taking pyridostigmine for the treatment of his MG. There is no evidence this medication increases the risk of infection; but the cholinergic activity can increase bronchial secretions, which could theoretically worsen the COVID-19 respiratory symptoms.6,11 During both ICU admissions, our patient continued pyridostigmine use, observing complete return to baseline after discharge. Given the possible association with worsened respiratory outcomes after the second ICU admission, the balance between managing MG symptoms and COVID-19 symptoms needs further examination.
The patient was in MC during his third presentation to the ED. Although respiratory symptoms may be difficult to differentiate from COVID-19, the additional neurologic symptoms seen in this patient allowed for quick determination of the need for MC treatment. There are many potential etiologies contributing to the development of the MC presented here, and it was likely due to multifactorial precipitants. A common cause of MC is viral upper respiratory infections, further challenging the care of these patients during this pandemic.12 Many medications have been cited as causing a MC, 2 of which our patient received during admission for COVID-19: azithromycin and levoquin.12 Although the patient did not receive hydroxychloroquine, which was still being considered as an appropriate COVID-19 treatment at the time, it also is a drug known for precipitating MC and its use scrutinized in patients with MG.12
A key aspect to diagnosing and guiding therapies in myasthenic crisis in addition to the clinical symptoms of acute weakness is respiratory assessment through the nonaerosolizing NIF test.12 Our patient’s NIF measured < 30 cmH2O when in MC, while the reference range is < 75 cmH2O, and for mechanical ventilation is recommended at 20 cmH2O. Although the patient was maintaining O2 saturation > 95%, his NIF value was concerning, and preparations were made in case of precipitous decline. Compounding the NIF assessment in this patient is his history of left phrenic nerve palsy. Without a documented baseline NIF, results were limited in determining his diaphragm strength.13 Treatment for MC includes IVIG or plasmapheresis, since this patient had failed his maintenance therapy IVIG, plasmapheresis was coordinated for definitive therapy.
Conclusions
Federal facilities have seen an increase in the amount of respiratory complaints over the past months. Although COVID-19 is a concerning diagnosis, it is crucial to consider comorbidities in the diagnostic workup of each, even with a previous recent diagnosis of COVID-19. As treatment recommendations for COVID-19 continue to fluctuate coupled with the limitations and difficulties associated with MG patients, so too treatment and evaluation must be carefully considered at each presentation.
1. Gousseff M, Penot P, Gallay L, et al. Clinical recurrences of COVID-19 symptoms after recovery: viral relapse, reinfection or inflammatory rebound? J Infect. 2020;81(5):816-846. doi:10.1016/j.jinf.2020.06.073
2. Duggan NM, Ludy SM, Shannon BC, Reisner AT, Wilcox SR. Is novel coronavirus 2019 reinfection possible? Interpreting dynamic SARS-CoV-2 test results. Am J Emerg Med. 2021;39:256.e1-256.e3. doi:10.1016/j.ajem.2020.06.079
3. Li J, Zhang L, Liu B, Song D. Case report: viral shedding for 60 days in a woman with COVID-19. Am J Trop Med Hyg. 2020;102(6):1210-1213. doi:10.4269/ajtmh.20-0275
4. Beigel JH, Tomashek KM, Dodd LE. Remdesivir for the treatment of Covid-19 - preliminary report. Reply. N Engl J Med. 2020;383(10):994. doi:10.1056/NEJMc2022236
5. McCreary EK, Angus DC. Efficacy of remdesivir in COVID-19. JAMA. 2020;324(11):1041-1042. doi:10.1001/jama.2020.16337
6. International MG/COVID-19 Working Group; Jacob S, Muppidi S, Gordon A, et al. Guidance for the management of myasthenia gravis (MG) and Lambert-Eaton myasthenic syndrome (LEMS) during the COVID-19 pandemic. J Neurol Sci. 2020;412:116803. doi:10.1016/j.jns.2020.116803
7. Anand P, Slama MCC, Kaku M, et al. COVID-19 in patients with myasthenia gravis. Muscle Nerve. 2020;62(2):254-258. doi:10.1002/mus.26918
8. Wooding DJ, Bach H. Treatment of COVID-19 with convalescent plasma: lessons from past coronavirus outbreaks. Clin Microbiol Infect. 2020;26(10):1436-1446. doi:10.1016/j.cmi.2020.08.005
9. Salazar E, Perez KK, Ashraf M, et al. Treatment of coronavirus disease 2019 (covid-19) patients with convalescent plasma. Am J Pathol. 2020;190(8):1680-1690. doi:10.1016/j.ajpath.2020.05.014
10. Ryan C, Minc A, Caceres J, et al. Predicting severe outcomes in Covid-19 related illness using only patient demographics, comorbidities and symptoms [published online ahead of print, 2020 Sep 9]. Am J Emerg Med. 2020;S0735-6757(20)30809-3. doi:10.1016/j.ajem.2020.09.017
11. Singh S, Govindarajan R. COVID-19 and generalized myasthenia gravis exacerbation: a case report. Clin Neurol Neurosurg. 2020;196:106045. doi:10.1016/j.clineuro.2020.106045
12. Wendell LC, Levine JM. Myasthenic crisis. Neurohospitalist. 2011;1(1):16-22. doi:10.1177/1941875210382918
13. Dubé BP, Dres M. Diaphragm dysfunction: diagnostic approaches and management strategies. J Clin Med. 2016;5(12):113. Published 2016 Dec 5. doi:10.3390/jcm5120113
A patient with myasthenia gravis who survived 2 COVID-19 infections required plasmapheresis to recover from an acute crisis.
A patient with myasthenia gravis who survived 2 COVID-19 infections required plasmapheresis to recover from an acute crisis.
COVID-19 is still in the early stages of understanding, although it is known to be complicated by individual patient comorbidities. The management and treatment of COVID-19 continues to quickly evolve as more is discovered regarding the virus. Multiple treatments have been preliminarily tested and used under a Food and Drug Administration emergency use authorization (EUA) determination. The long-term success of these therapies, however, is yet to be determined. Additionally, if a patient has a second clinical presentation for COVID-19, it is not known whether this represents latency with subsequent reactivation from the previous infection or a second de novo infection. The uncertainty calls into question the duration of immunity, if any, following a primary infection.
COVID-19 management becomes more complicated when patients have complex medical conditions, such as myasthenia gravis (MG). This autoimmune neuromuscular disorder can present with varying weakness, and many patients are on immunomodulator medications. The weakness can worsen into a myasthenic crisis (MC), resulting in profound weakness of the respiratory muscles. Therefore, patients with MG are at increased risk for COVID-19 and may have a more complicated course when infected.
Our patient with MG presented for severe COVID-19 symptoms twice and later developed MC. He received 2 treatment modalities available under an EUA (remdesivir and convalescent plasma) for COVID-19, resulting in symptom resolution and a negative polymerize chain reaction (PCR) test result for the virus. However, after receiving his typical maintenance therapy of IV immunoglobulin (IVIG) for his MG, he again developed symptoms consistent with COVID-19 and tested positive. After recovering from the second episode of COVID-19, the patient went into MC requiring plasmapheresis.
Case Presentation
A 56-year-old male, US Army veteran presented to Carl R. Darnall Army Medical Center emergency department (ED) 6 days after testing positive for COVID-19, with worsening sputum, cough, congestion, dyspnea, and fever. Due to his MG, the patient had a home oxygen monitor and reported that his oxygenation saturation dropped below 90% with minimal exertion. His medical history was significant for MG, status postthymectomy and radiation treatment, left hemidiaphragm paralysis secondary to phrenic nerve injury, and corticosteroid-induced insulin-dependent diabetes mellitus. His current home medications included pyridostigmine 60 mg 3 times a day, mycophenolate (MMF) 1500 mg twice daily, IV immunoglobulin (IVIG) every 3 weeks, insulin aspart up to 16 U per meal, insulin glargine 30 U twice a day, dulaglutide 0.75 mg every week, and metformin 1000 mg twice daily.
On initial examination, the patient’s heart rate (HR) was 111 beats/min, respiratory rate (RR), 22 breaths/min, blood pressure (BP), 138/88 mm Hg, temperature, 100.9 oF, and his initial pulse oximetry, 91% on room air. On physical examination, the patient was tachypneic, though without other signs of respiratory distress. Lung auscultation revealed no adventitial lung sounds. His cardiac examination was notable only for tachycardia. His neurologic examination demonstrated intact cranial nerves, with 5 out of 5 (scale 1 to 5) strength throughout the upper and lower extremities, sensation was intact to light touch, and he had normal cerebellar function. The rest of the examination was normal.
Initial laboratory investigation was notable for a white blood cell count of 14.15x103 cells/mcL with 84% neutrophils, and 6% lymphocytes. Additional tests revealed a C-reactive protein (CRP) level, 17.97 mg/dL (reference range, 0-0.5 mg/dL), ferritin level, 647 ng/mL (reference range, 22-274 ng/mL), d-dimer, 0.64 mcg/mL (reference range, 0-0.47mcg/mL), and a repeated positive COVID-19 PCR test. A portable chest X-ray showed bibasilar opacities (Figure 1).
The patient was diagnosed with COVID-19 and admitted to the intensive care unit (ICU). In the ICU, the patient received 1 U of convalescent plasma (CP) and started on a course of IV remdesivir 100 mg/d consistent with the EUA. He also received a 5-day course of ceftriaxone and azithromycin for possible community acquired pneumonia (CAP). As part of the patient’s MG maintenance medications, he received IVIG 4 g while in the ICU. Throughout his ICU stay, he required supplemental nasal cannula oxygenation to maintain his oxygen saturation > 93%. After 8 days in the ICU, his oxygen requirements decreased, and the patient was transferred out of the ICU and remdesivir was discontinued. On hospital day 10, a repeat COVID-19 PCR test was negative, inflammatory markers returned to within normal limits, and a repeat chest X-ray showed improvement from admission (Figure 2). Having recovered significantly, he was discharged home.
Three weeks later, the patient again presented to the MTF with 3 days of dyspnea, cough, fever, nausea, and vomiting. One day before symptom onset, he had received his maintenance IVIG infusion. The patient reported that his home oxygen saturation was 82% with minimal exertion. On ED presentation his HR was 107 beats/min, RR, 28 breaths/min, temperature, 98.1 oF, BP 118/71 mm Hg, and oxygen saturation, 92% on 2L nasal cannula. His examination was most notable for tachypnea with accessory muscle use. At this time, his neurologic examination was unchanged from prior admission with grossly intact cranial nerves and symmetric 5 of 5 motor strength in all extremities.
At this second ED visit, laboratory results demonstrated a CRP of 3.44 mg/dL, ferritin 2019 ng/mL, d-dimer, 3.39 mcg/mL, and a positive COVID-19 PCR result. His chest X-ray demonstrated new peripheral opacities compared with the X-ray at discharge (Figure 3). He required ICU admission again for his COVID-19 symptoms.
During his ICU course he continued to require supplemental oxygen by nasal cannula, though never required intubation. This second admission, he was again treated empirically for CAP with levofloxacin 750 mg daily for 5 days. He was discharged after 14 days with symptom resolution and down trending of inflammatory markers, though he was not retested for COVID-19.
Four days after his second discharge, he presented to the ED for a third time with diffuse weakness, dysphagia, and dysarthria of 1 day. His HR was 87/beats/min; RR, 17 breaths/min; temperature, 98.7 oF; BP, 144/81 mm Hg; and oxygen saturation, 98% on room air. His examination was significant for slurred speech, bilateral ptosis, 3 of 5 strength in bilateral finger flexion/abduction, wrist extension, knee and ankle flexion/extension; 4 of 5 strength in bilateral proximal muscle testing of deltoid, and hip; normal sensation, cerebellar function and reflexes. His negative inspiratory force (NIF) maximal effort was −30 cmH2O. He was determined to be in MC without evidence of COIVD-19 symptoms, and laboratory results were within normal limits, including a negative COVID-19 PCR. As he received IVIG as maintenance therapy, plasmapheresis was recommended to treat his MC, which required transfer to an outside civilian facility.
At the outside hospital, the patient underwent 5 rounds of plasmapheresis over 10 days. By the third treatment his strength had returned with resolution of the bulbar symptoms and no supplemental oxygen requirements. The patient was discharged and continued his original dosages of MMF and pyridostigmine. At 3 months, he remained asymptomatic from a COVID-19 standpoint and stable from a MG standpoint.
Discussion
Reinfection with the COVID-19 has been continuously debated with alternative explanations suggested for a positive test after a previous negative PCR test in the setting of symptom resolution.1,2 Proposed causes include dynamic PCR results due to prolonged viral shedding and inaccurate or poorly sensitive tests. The repeat positive cases in these scenarios, however, occurred in asymptomatic patients.1,2 COVID-19 shedding averages 20 to 22 days after symptom onset but has been seen up to 36 days after symptom resolution.2,3 This would suggest that fluctuating results during the immediate postsymptom period may be due to variations in viral shedding load and or sampling error—especially in asymptomatic patients. On the other hand, patients who experience return of symptoms days to weeks after previous convalescence leave clinicians wondering whether this represents clinical latency with reactivation or COVID-19 reinfection. A separate case of initial COVID-19 in a patient that had subsequent clinical recovery with a negative PCR developed recurrent respiratory symptoms and had a positive PCR test only 10 days later, further highlighting the reinfection vs reactivation issue of COVID-19.2 Further understanding of this issue may have implications on the extent of natural immunity following primary infection; potential vaccine dosage schedules; and global public health policies.
Although reactivation may be plausible given his immunomodulatory therapy, our patient’s second COVID-19 symptoms started 40 days after the initial symptoms, and 26 days after the initial course resolution; previous cases of return of severe symptoms occurred between 3 and 6 days.1 Given our patient’s time course between resolution and return of symptoms, if latency is the mechanism at play, this case demonstrates an exceptionally longer latency period than the ones that have been reported. Additionally, if latency is an issue in COVID-19, using remdesivir as a treatment further complicates the understanding of this disease.
Remdesivir, a nucleoside analogue antiviral, was shown to benefit recovery in patients with severe symptoms in the Adaptive COVID-19 Treatment Trial-1 study.4 Our patient had originally been placed on a 10-day course; however, on treatment day 8, his symptoms resolved and the remdesivir was discontinued. This is a similar finding to half the patients in the 10-day arm of the study by McCreary and colleagues.5 Although our patient was asymptomatic 4 weeks after the start of remdesivir, consistent with the majority of patients in the McCreary 10-day study arm, further comparison of the presented patient is limited due to study length and follow-up considerations.5 No previous data exist on reactivation, reinfection, or long-term mortality after being treated with remdesivir for COVID-19 infection.
IVIG is being studied in the treatment of COVID-19 and bears consideration as it relates to our patient. There is no evidence that IVIG used in the treatment of autoimmune diseases increases the risk of infection compared with that of other medications used in the treatment of such diseases. Furthermore, the current guidance from the MG expert panel does not suggest that IVIG increases the risk of contracting COVID-19 aside from the risks of exposure to hospital infrastructure.6 Yet the guidance does not discuss the use of IVIG for MG in patients who are already symptomatic from COVID-19 or for patients recovering from the clinical disease or does it discuss a possible compounding risk of thromboembolic events associated with IVIG and COVID-19.6,7 Our patient received his maintenance IVIG during his first admission without any worsening of symptoms or increased oxygen requirements. The day following our patient’s next scheduled IVIG infusion—while asymptomatic—he again developed respiratory symptoms; this could suggest that IVIG did not contribute to his second clinical course nor protect against.
CP is a treatment modality that has been used and studied in previous infectious outbreaks such as the first severe acute respiratory syndrome, and the H1N1 influenza virus.8 Current data on CP for COVID-19 are limited, but early descriptive studies have shown a benefit in improvement of symptoms 5 days sooner in those requiring supplemental oxygen, but no benefit for those requiring mechanical ventilation.9 Like patients that benefitted in these studies, our patient received CP early, 6 days after first testing positive and onset of symptoms. This patient’s reinfection or return of symptoms draws into question the hindrance or even prevention of long-term immunity from administration of CP.
COVID-19 presents many challenges when managing this patient’s coexisting MG, especially as the patient was already being treated with immunosuppressing therapies. The guidance does recommend continuation of standard MG therapies during hospitalizations, including immunosuppression medications such as MMF.6 Immunosuppression is associated with worsened severity of COVID-19 symptoms, although no relation exists to degree of immunosuppression and severity.7,10 To the best of our knowledge there has been no case report of reinfection or reactivation of COVID-19 associated with immunosuppressive agents used in the treatment of MG.
Our patient also was taking pyridostigmine for the treatment of his MG. There is no evidence this medication increases the risk of infection; but the cholinergic activity can increase bronchial secretions, which could theoretically worsen the COVID-19 respiratory symptoms.6,11 During both ICU admissions, our patient continued pyridostigmine use, observing complete return to baseline after discharge. Given the possible association with worsened respiratory outcomes after the second ICU admission, the balance between managing MG symptoms and COVID-19 symptoms needs further examination.
The patient was in MC during his third presentation to the ED. Although respiratory symptoms may be difficult to differentiate from COVID-19, the additional neurologic symptoms seen in this patient allowed for quick determination of the need for MC treatment. There are many potential etiologies contributing to the development of the MC presented here, and it was likely due to multifactorial precipitants. A common cause of MC is viral upper respiratory infections, further challenging the care of these patients during this pandemic.12 Many medications have been cited as causing a MC, 2 of which our patient received during admission for COVID-19: azithromycin and levoquin.12 Although the patient did not receive hydroxychloroquine, which was still being considered as an appropriate COVID-19 treatment at the time, it also is a drug known for precipitating MC and its use scrutinized in patients with MG.12
A key aspect to diagnosing and guiding therapies in myasthenic crisis in addition to the clinical symptoms of acute weakness is respiratory assessment through the nonaerosolizing NIF test.12 Our patient’s NIF measured < 30 cmH2O when in MC, while the reference range is < 75 cmH2O, and for mechanical ventilation is recommended at 20 cmH2O. Although the patient was maintaining O2 saturation > 95%, his NIF value was concerning, and preparations were made in case of precipitous decline. Compounding the NIF assessment in this patient is his history of left phrenic nerve palsy. Without a documented baseline NIF, results were limited in determining his diaphragm strength.13 Treatment for MC includes IVIG or plasmapheresis, since this patient had failed his maintenance therapy IVIG, plasmapheresis was coordinated for definitive therapy.
Conclusions
Federal facilities have seen an increase in the amount of respiratory complaints over the past months. Although COVID-19 is a concerning diagnosis, it is crucial to consider comorbidities in the diagnostic workup of each, even with a previous recent diagnosis of COVID-19. As treatment recommendations for COVID-19 continue to fluctuate coupled with the limitations and difficulties associated with MG patients, so too treatment and evaluation must be carefully considered at each presentation.
COVID-19 is still in the early stages of understanding, although it is known to be complicated by individual patient comorbidities. The management and treatment of COVID-19 continues to quickly evolve as more is discovered regarding the virus. Multiple treatments have been preliminarily tested and used under a Food and Drug Administration emergency use authorization (EUA) determination. The long-term success of these therapies, however, is yet to be determined. Additionally, if a patient has a second clinical presentation for COVID-19, it is not known whether this represents latency with subsequent reactivation from the previous infection or a second de novo infection. The uncertainty calls into question the duration of immunity, if any, following a primary infection.
COVID-19 management becomes more complicated when patients have complex medical conditions, such as myasthenia gravis (MG). This autoimmune neuromuscular disorder can present with varying weakness, and many patients are on immunomodulator medications. The weakness can worsen into a myasthenic crisis (MC), resulting in profound weakness of the respiratory muscles. Therefore, patients with MG are at increased risk for COVID-19 and may have a more complicated course when infected.
Our patient with MG presented for severe COVID-19 symptoms twice and later developed MC. He received 2 treatment modalities available under an EUA (remdesivir and convalescent plasma) for COVID-19, resulting in symptom resolution and a negative polymerize chain reaction (PCR) test result for the virus. However, after receiving his typical maintenance therapy of IV immunoglobulin (IVIG) for his MG, he again developed symptoms consistent with COVID-19 and tested positive. After recovering from the second episode of COVID-19, the patient went into MC requiring plasmapheresis.
Case Presentation
A 56-year-old male, US Army veteran presented to Carl R. Darnall Army Medical Center emergency department (ED) 6 days after testing positive for COVID-19, with worsening sputum, cough, congestion, dyspnea, and fever. Due to his MG, the patient had a home oxygen monitor and reported that his oxygenation saturation dropped below 90% with minimal exertion. His medical history was significant for MG, status postthymectomy and radiation treatment, left hemidiaphragm paralysis secondary to phrenic nerve injury, and corticosteroid-induced insulin-dependent diabetes mellitus. His current home medications included pyridostigmine 60 mg 3 times a day, mycophenolate (MMF) 1500 mg twice daily, IV immunoglobulin (IVIG) every 3 weeks, insulin aspart up to 16 U per meal, insulin glargine 30 U twice a day, dulaglutide 0.75 mg every week, and metformin 1000 mg twice daily.
On initial examination, the patient’s heart rate (HR) was 111 beats/min, respiratory rate (RR), 22 breaths/min, blood pressure (BP), 138/88 mm Hg, temperature, 100.9 oF, and his initial pulse oximetry, 91% on room air. On physical examination, the patient was tachypneic, though without other signs of respiratory distress. Lung auscultation revealed no adventitial lung sounds. His cardiac examination was notable only for tachycardia. His neurologic examination demonstrated intact cranial nerves, with 5 out of 5 (scale 1 to 5) strength throughout the upper and lower extremities, sensation was intact to light touch, and he had normal cerebellar function. The rest of the examination was normal.
Initial laboratory investigation was notable for a white blood cell count of 14.15x103 cells/mcL with 84% neutrophils, and 6% lymphocytes. Additional tests revealed a C-reactive protein (CRP) level, 17.97 mg/dL (reference range, 0-0.5 mg/dL), ferritin level, 647 ng/mL (reference range, 22-274 ng/mL), d-dimer, 0.64 mcg/mL (reference range, 0-0.47mcg/mL), and a repeated positive COVID-19 PCR test. A portable chest X-ray showed bibasilar opacities (Figure 1).
The patient was diagnosed with COVID-19 and admitted to the intensive care unit (ICU). In the ICU, the patient received 1 U of convalescent plasma (CP) and started on a course of IV remdesivir 100 mg/d consistent with the EUA. He also received a 5-day course of ceftriaxone and azithromycin for possible community acquired pneumonia (CAP). As part of the patient’s MG maintenance medications, he received IVIG 4 g while in the ICU. Throughout his ICU stay, he required supplemental nasal cannula oxygenation to maintain his oxygen saturation > 93%. After 8 days in the ICU, his oxygen requirements decreased, and the patient was transferred out of the ICU and remdesivir was discontinued. On hospital day 10, a repeat COVID-19 PCR test was negative, inflammatory markers returned to within normal limits, and a repeat chest X-ray showed improvement from admission (Figure 2). Having recovered significantly, he was discharged home.
Three weeks later, the patient again presented to the MTF with 3 days of dyspnea, cough, fever, nausea, and vomiting. One day before symptom onset, he had received his maintenance IVIG infusion. The patient reported that his home oxygen saturation was 82% with minimal exertion. On ED presentation his HR was 107 beats/min, RR, 28 breaths/min, temperature, 98.1 oF, BP 118/71 mm Hg, and oxygen saturation, 92% on 2L nasal cannula. His examination was most notable for tachypnea with accessory muscle use. At this time, his neurologic examination was unchanged from prior admission with grossly intact cranial nerves and symmetric 5 of 5 motor strength in all extremities.
At this second ED visit, laboratory results demonstrated a CRP of 3.44 mg/dL, ferritin 2019 ng/mL, d-dimer, 3.39 mcg/mL, and a positive COVID-19 PCR result. His chest X-ray demonstrated new peripheral opacities compared with the X-ray at discharge (Figure 3). He required ICU admission again for his COVID-19 symptoms.
During his ICU course he continued to require supplemental oxygen by nasal cannula, though never required intubation. This second admission, he was again treated empirically for CAP with levofloxacin 750 mg daily for 5 days. He was discharged after 14 days with symptom resolution and down trending of inflammatory markers, though he was not retested for COVID-19.
Four days after his second discharge, he presented to the ED for a third time with diffuse weakness, dysphagia, and dysarthria of 1 day. His HR was 87/beats/min; RR, 17 breaths/min; temperature, 98.7 oF; BP, 144/81 mm Hg; and oxygen saturation, 98% on room air. His examination was significant for slurred speech, bilateral ptosis, 3 of 5 strength in bilateral finger flexion/abduction, wrist extension, knee and ankle flexion/extension; 4 of 5 strength in bilateral proximal muscle testing of deltoid, and hip; normal sensation, cerebellar function and reflexes. His negative inspiratory force (NIF) maximal effort was −30 cmH2O. He was determined to be in MC without evidence of COIVD-19 symptoms, and laboratory results were within normal limits, including a negative COVID-19 PCR. As he received IVIG as maintenance therapy, plasmapheresis was recommended to treat his MC, which required transfer to an outside civilian facility.
At the outside hospital, the patient underwent 5 rounds of plasmapheresis over 10 days. By the third treatment his strength had returned with resolution of the bulbar symptoms and no supplemental oxygen requirements. The patient was discharged and continued his original dosages of MMF and pyridostigmine. At 3 months, he remained asymptomatic from a COVID-19 standpoint and stable from a MG standpoint.
Discussion
Reinfection with the COVID-19 has been continuously debated with alternative explanations suggested for a positive test after a previous negative PCR test in the setting of symptom resolution.1,2 Proposed causes include dynamic PCR results due to prolonged viral shedding and inaccurate or poorly sensitive tests. The repeat positive cases in these scenarios, however, occurred in asymptomatic patients.1,2 COVID-19 shedding averages 20 to 22 days after symptom onset but has been seen up to 36 days after symptom resolution.2,3 This would suggest that fluctuating results during the immediate postsymptom period may be due to variations in viral shedding load and or sampling error—especially in asymptomatic patients. On the other hand, patients who experience return of symptoms days to weeks after previous convalescence leave clinicians wondering whether this represents clinical latency with reactivation or COVID-19 reinfection. A separate case of initial COVID-19 in a patient that had subsequent clinical recovery with a negative PCR developed recurrent respiratory symptoms and had a positive PCR test only 10 days later, further highlighting the reinfection vs reactivation issue of COVID-19.2 Further understanding of this issue may have implications on the extent of natural immunity following primary infection; potential vaccine dosage schedules; and global public health policies.
Although reactivation may be plausible given his immunomodulatory therapy, our patient’s second COVID-19 symptoms started 40 days after the initial symptoms, and 26 days after the initial course resolution; previous cases of return of severe symptoms occurred between 3 and 6 days.1 Given our patient’s time course between resolution and return of symptoms, if latency is the mechanism at play, this case demonstrates an exceptionally longer latency period than the ones that have been reported. Additionally, if latency is an issue in COVID-19, using remdesivir as a treatment further complicates the understanding of this disease.
Remdesivir, a nucleoside analogue antiviral, was shown to benefit recovery in patients with severe symptoms in the Adaptive COVID-19 Treatment Trial-1 study.4 Our patient had originally been placed on a 10-day course; however, on treatment day 8, his symptoms resolved and the remdesivir was discontinued. This is a similar finding to half the patients in the 10-day arm of the study by McCreary and colleagues.5 Although our patient was asymptomatic 4 weeks after the start of remdesivir, consistent with the majority of patients in the McCreary 10-day study arm, further comparison of the presented patient is limited due to study length and follow-up considerations.5 No previous data exist on reactivation, reinfection, or long-term mortality after being treated with remdesivir for COVID-19 infection.
IVIG is being studied in the treatment of COVID-19 and bears consideration as it relates to our patient. There is no evidence that IVIG used in the treatment of autoimmune diseases increases the risk of infection compared with that of other medications used in the treatment of such diseases. Furthermore, the current guidance from the MG expert panel does not suggest that IVIG increases the risk of contracting COVID-19 aside from the risks of exposure to hospital infrastructure.6 Yet the guidance does not discuss the use of IVIG for MG in patients who are already symptomatic from COVID-19 or for patients recovering from the clinical disease or does it discuss a possible compounding risk of thromboembolic events associated with IVIG and COVID-19.6,7 Our patient received his maintenance IVIG during his first admission without any worsening of symptoms or increased oxygen requirements. The day following our patient’s next scheduled IVIG infusion—while asymptomatic—he again developed respiratory symptoms; this could suggest that IVIG did not contribute to his second clinical course nor protect against.
CP is a treatment modality that has been used and studied in previous infectious outbreaks such as the first severe acute respiratory syndrome, and the H1N1 influenza virus.8 Current data on CP for COVID-19 are limited, but early descriptive studies have shown a benefit in improvement of symptoms 5 days sooner in those requiring supplemental oxygen, but no benefit for those requiring mechanical ventilation.9 Like patients that benefitted in these studies, our patient received CP early, 6 days after first testing positive and onset of symptoms. This patient’s reinfection or return of symptoms draws into question the hindrance or even prevention of long-term immunity from administration of CP.
COVID-19 presents many challenges when managing this patient’s coexisting MG, especially as the patient was already being treated with immunosuppressing therapies. The guidance does recommend continuation of standard MG therapies during hospitalizations, including immunosuppression medications such as MMF.6 Immunosuppression is associated with worsened severity of COVID-19 symptoms, although no relation exists to degree of immunosuppression and severity.7,10 To the best of our knowledge there has been no case report of reinfection or reactivation of COVID-19 associated with immunosuppressive agents used in the treatment of MG.
Our patient also was taking pyridostigmine for the treatment of his MG. There is no evidence this medication increases the risk of infection; but the cholinergic activity can increase bronchial secretions, which could theoretically worsen the COVID-19 respiratory symptoms.6,11 During both ICU admissions, our patient continued pyridostigmine use, observing complete return to baseline after discharge. Given the possible association with worsened respiratory outcomes after the second ICU admission, the balance between managing MG symptoms and COVID-19 symptoms needs further examination.
The patient was in MC during his third presentation to the ED. Although respiratory symptoms may be difficult to differentiate from COVID-19, the additional neurologic symptoms seen in this patient allowed for quick determination of the need for MC treatment. There are many potential etiologies contributing to the development of the MC presented here, and it was likely due to multifactorial precipitants. A common cause of MC is viral upper respiratory infections, further challenging the care of these patients during this pandemic.12 Many medications have been cited as causing a MC, 2 of which our patient received during admission for COVID-19: azithromycin and levoquin.12 Although the patient did not receive hydroxychloroquine, which was still being considered as an appropriate COVID-19 treatment at the time, it also is a drug known for precipitating MC and its use scrutinized in patients with MG.12
A key aspect to diagnosing and guiding therapies in myasthenic crisis in addition to the clinical symptoms of acute weakness is respiratory assessment through the nonaerosolizing NIF test.12 Our patient’s NIF measured < 30 cmH2O when in MC, while the reference range is < 75 cmH2O, and for mechanical ventilation is recommended at 20 cmH2O. Although the patient was maintaining O2 saturation > 95%, his NIF value was concerning, and preparations were made in case of precipitous decline. Compounding the NIF assessment in this patient is his history of left phrenic nerve palsy. Without a documented baseline NIF, results were limited in determining his diaphragm strength.13 Treatment for MC includes IVIG or plasmapheresis, since this patient had failed his maintenance therapy IVIG, plasmapheresis was coordinated for definitive therapy.
Conclusions
Federal facilities have seen an increase in the amount of respiratory complaints over the past months. Although COVID-19 is a concerning diagnosis, it is crucial to consider comorbidities in the diagnostic workup of each, even with a previous recent diagnosis of COVID-19. As treatment recommendations for COVID-19 continue to fluctuate coupled with the limitations and difficulties associated with MG patients, so too treatment and evaluation must be carefully considered at each presentation.
1. Gousseff M, Penot P, Gallay L, et al. Clinical recurrences of COVID-19 symptoms after recovery: viral relapse, reinfection or inflammatory rebound? J Infect. 2020;81(5):816-846. doi:10.1016/j.jinf.2020.06.073
2. Duggan NM, Ludy SM, Shannon BC, Reisner AT, Wilcox SR. Is novel coronavirus 2019 reinfection possible? Interpreting dynamic SARS-CoV-2 test results. Am J Emerg Med. 2021;39:256.e1-256.e3. doi:10.1016/j.ajem.2020.06.079
3. Li J, Zhang L, Liu B, Song D. Case report: viral shedding for 60 days in a woman with COVID-19. Am J Trop Med Hyg. 2020;102(6):1210-1213. doi:10.4269/ajtmh.20-0275
4. Beigel JH, Tomashek KM, Dodd LE. Remdesivir for the treatment of Covid-19 - preliminary report. Reply. N Engl J Med. 2020;383(10):994. doi:10.1056/NEJMc2022236
5. McCreary EK, Angus DC. Efficacy of remdesivir in COVID-19. JAMA. 2020;324(11):1041-1042. doi:10.1001/jama.2020.16337
6. International MG/COVID-19 Working Group; Jacob S, Muppidi S, Gordon A, et al. Guidance for the management of myasthenia gravis (MG) and Lambert-Eaton myasthenic syndrome (LEMS) during the COVID-19 pandemic. J Neurol Sci. 2020;412:116803. doi:10.1016/j.jns.2020.116803
7. Anand P, Slama MCC, Kaku M, et al. COVID-19 in patients with myasthenia gravis. Muscle Nerve. 2020;62(2):254-258. doi:10.1002/mus.26918
8. Wooding DJ, Bach H. Treatment of COVID-19 with convalescent plasma: lessons from past coronavirus outbreaks. Clin Microbiol Infect. 2020;26(10):1436-1446. doi:10.1016/j.cmi.2020.08.005
9. Salazar E, Perez KK, Ashraf M, et al. Treatment of coronavirus disease 2019 (covid-19) patients with convalescent plasma. Am J Pathol. 2020;190(8):1680-1690. doi:10.1016/j.ajpath.2020.05.014
10. Ryan C, Minc A, Caceres J, et al. Predicting severe outcomes in Covid-19 related illness using only patient demographics, comorbidities and symptoms [published online ahead of print, 2020 Sep 9]. Am J Emerg Med. 2020;S0735-6757(20)30809-3. doi:10.1016/j.ajem.2020.09.017
11. Singh S, Govindarajan R. COVID-19 and generalized myasthenia gravis exacerbation: a case report. Clin Neurol Neurosurg. 2020;196:106045. doi:10.1016/j.clineuro.2020.106045
12. Wendell LC, Levine JM. Myasthenic crisis. Neurohospitalist. 2011;1(1):16-22. doi:10.1177/1941875210382918
13. Dubé BP, Dres M. Diaphragm dysfunction: diagnostic approaches and management strategies. J Clin Med. 2016;5(12):113. Published 2016 Dec 5. doi:10.3390/jcm5120113
1. Gousseff M, Penot P, Gallay L, et al. Clinical recurrences of COVID-19 symptoms after recovery: viral relapse, reinfection or inflammatory rebound? J Infect. 2020;81(5):816-846. doi:10.1016/j.jinf.2020.06.073
2. Duggan NM, Ludy SM, Shannon BC, Reisner AT, Wilcox SR. Is novel coronavirus 2019 reinfection possible? Interpreting dynamic SARS-CoV-2 test results. Am J Emerg Med. 2021;39:256.e1-256.e3. doi:10.1016/j.ajem.2020.06.079
3. Li J, Zhang L, Liu B, Song D. Case report: viral shedding for 60 days in a woman with COVID-19. Am J Trop Med Hyg. 2020;102(6):1210-1213. doi:10.4269/ajtmh.20-0275
4. Beigel JH, Tomashek KM, Dodd LE. Remdesivir for the treatment of Covid-19 - preliminary report. Reply. N Engl J Med. 2020;383(10):994. doi:10.1056/NEJMc2022236
5. McCreary EK, Angus DC. Efficacy of remdesivir in COVID-19. JAMA. 2020;324(11):1041-1042. doi:10.1001/jama.2020.16337
6. International MG/COVID-19 Working Group; Jacob S, Muppidi S, Gordon A, et al. Guidance for the management of myasthenia gravis (MG) and Lambert-Eaton myasthenic syndrome (LEMS) during the COVID-19 pandemic. J Neurol Sci. 2020;412:116803. doi:10.1016/j.jns.2020.116803
7. Anand P, Slama MCC, Kaku M, et al. COVID-19 in patients with myasthenia gravis. Muscle Nerve. 2020;62(2):254-258. doi:10.1002/mus.26918
8. Wooding DJ, Bach H. Treatment of COVID-19 with convalescent plasma: lessons from past coronavirus outbreaks. Clin Microbiol Infect. 2020;26(10):1436-1446. doi:10.1016/j.cmi.2020.08.005
9. Salazar E, Perez KK, Ashraf M, et al. Treatment of coronavirus disease 2019 (covid-19) patients with convalescent plasma. Am J Pathol. 2020;190(8):1680-1690. doi:10.1016/j.ajpath.2020.05.014
10. Ryan C, Minc A, Caceres J, et al. Predicting severe outcomes in Covid-19 related illness using only patient demographics, comorbidities and symptoms [published online ahead of print, 2020 Sep 9]. Am J Emerg Med. 2020;S0735-6757(20)30809-3. doi:10.1016/j.ajem.2020.09.017
11. Singh S, Govindarajan R. COVID-19 and generalized myasthenia gravis exacerbation: a case report. Clin Neurol Neurosurg. 2020;196:106045. doi:10.1016/j.clineuro.2020.106045
12. Wendell LC, Levine JM. Myasthenic crisis. Neurohospitalist. 2011;1(1):16-22. doi:10.1177/1941875210382918
13. Dubé BP, Dres M. Diaphragm dysfunction: diagnostic approaches and management strategies. J Clin Med. 2016;5(12):113. Published 2016 Dec 5. doi:10.3390/jcm5120113
Mobile stroke teams treat patients faster and reduce disability
Having a mobile interventional stroke team (MIST) travel to treat stroke patients soon after stroke onset may improve patient outcomes, according to a new study. A retrospective analysis of a pilot program in New York found that
“The use of a Mobile Interventional Stroke Team (MIST) traveling to Thrombectomy Capable Stroke Centers to perform endovascular thrombectomy has been shown to be significantly faster with improved discharge outcomes,” wrote lead author Jacob Morey, a doctoral Candidate at Icahn School of Medicine at Mount Sinai in New York and coauthors in the paper. Prior to this study, “the effect of the MIST model stratified by time of presentation” had yet to be studied.
The findings were published online on Aug. 5 in Stroke.
MIST model versus drip-and-ship
The researchers analyzed 226 patients who underwent endovascular thrombectomy between January 2017 and February 2020 at four hospitals in the Mount Sinai health system using the NYC MIST Trial and a stroke database. At baseline, all patients were functionally independent as assessed by the modified Rankin Scale (mRS, score of 0-2). 106 patients were treated by a MIST team – staffed by a neurointerventionalist, a fellow or physician assistant, and radiologic technologist – that traveled to the patient’s location. A total of 120 patients were transferred to a comprehensive stroke center (CSC) or a hospital with endovascular thrombectomy expertise. The analysis was stratified based on whether the patient presented in the early time window (≤ 6 hours) or late time window (> 6 hours).
Patients treated in the early time window were significantly more likely to be mobile and able to perform daily tasks (mRS ≤ 2) 90 days after the procedure in the MIST group (54%), compared with the transferred group (28%, P < 0.01). Outcomes did not differ significantly between groups in the late time window (35% vs. 41%, P = 0.77).
Similarly, early-time-window patients in the MIST group were more likely to have higher functionality at discharge, compared with transferred patients, based on the on the National Institutes of Health Stroke Scale (median score of 5.0 vs. 12.0, P < 0.01). There was no significant difference between groups treated in the late time window (median score of 5.0 vs. 11.0, P = 0.11).
“Ischemic strokes often progress rapidly and can cause severe damage because brain tissue dies quickly without oxygen, resulting in serious long-term disabilities or death,“ said Johanna Fifi, MD, of Icahn School of Medicine, said in a statement to the American Heart Association. “Assessing and treating stroke patients in the early window means that a greater number of fast-progressing strokes are identified and treated.”
Time is brain
Endovascular thrombectomy is a time-sensitive surgical procedure to remove large blood clots in acute ischemic stroke that has “historically been limited to comprehensive stroke centers,” the authors wrote in their paper. It is considered the standard of care in ischemic strokes, which make up 90% of all strokes. “Less than 50% of Americans have direct access to endovascular thrombectomy, the others must be transferred to a thrombectomy-capable hospital for treatment, often losing over 2 hours of time to treatment,” said Dr. Fifi. “Every minute is precious in treating stroke, and getting to a center that offers thrombectomy is very important. The MIST model would address this by providing faster access to this potentially life-saving, disability-reducing procedure.”
Access to timely endovascular thrombectomy is gradually improving as “more institutions and cities have implemented the [MIST] model.” Dr. Fifi said.
“This study stresses the importance of ‘time is brain,’ especially for patients in the early time window. Although the study is limited by the observational, retrospective design and was performed at a single integrated center, the findings are provocative,” said Louise McCullough, MD, of the University of Texas Health Science Center at Houston said in a statement to the American Heart Association. “The use of a MIST model highlights the potential benefit of early and urgent treatment for patients with large-vessel stroke. Stroke systems of care need to take advantage of any opportunity to treat patients early, wherever they are.”
The study was partly funded by a Stryker Foundation grant.
Having a mobile interventional stroke team (MIST) travel to treat stroke patients soon after stroke onset may improve patient outcomes, according to a new study. A retrospective analysis of a pilot program in New York found that
“The use of a Mobile Interventional Stroke Team (MIST) traveling to Thrombectomy Capable Stroke Centers to perform endovascular thrombectomy has been shown to be significantly faster with improved discharge outcomes,” wrote lead author Jacob Morey, a doctoral Candidate at Icahn School of Medicine at Mount Sinai in New York and coauthors in the paper. Prior to this study, “the effect of the MIST model stratified by time of presentation” had yet to be studied.
The findings were published online on Aug. 5 in Stroke.
MIST model versus drip-and-ship
The researchers analyzed 226 patients who underwent endovascular thrombectomy between January 2017 and February 2020 at four hospitals in the Mount Sinai health system using the NYC MIST Trial and a stroke database. At baseline, all patients were functionally independent as assessed by the modified Rankin Scale (mRS, score of 0-2). 106 patients were treated by a MIST team – staffed by a neurointerventionalist, a fellow or physician assistant, and radiologic technologist – that traveled to the patient’s location. A total of 120 patients were transferred to a comprehensive stroke center (CSC) or a hospital with endovascular thrombectomy expertise. The analysis was stratified based on whether the patient presented in the early time window (≤ 6 hours) or late time window (> 6 hours).
Patients treated in the early time window were significantly more likely to be mobile and able to perform daily tasks (mRS ≤ 2) 90 days after the procedure in the MIST group (54%), compared with the transferred group (28%, P < 0.01). Outcomes did not differ significantly between groups in the late time window (35% vs. 41%, P = 0.77).
Similarly, early-time-window patients in the MIST group were more likely to have higher functionality at discharge, compared with transferred patients, based on the on the National Institutes of Health Stroke Scale (median score of 5.0 vs. 12.0, P < 0.01). There was no significant difference between groups treated in the late time window (median score of 5.0 vs. 11.0, P = 0.11).
“Ischemic strokes often progress rapidly and can cause severe damage because brain tissue dies quickly without oxygen, resulting in serious long-term disabilities or death,“ said Johanna Fifi, MD, of Icahn School of Medicine, said in a statement to the American Heart Association. “Assessing and treating stroke patients in the early window means that a greater number of fast-progressing strokes are identified and treated.”
Time is brain
Endovascular thrombectomy is a time-sensitive surgical procedure to remove large blood clots in acute ischemic stroke that has “historically been limited to comprehensive stroke centers,” the authors wrote in their paper. It is considered the standard of care in ischemic strokes, which make up 90% of all strokes. “Less than 50% of Americans have direct access to endovascular thrombectomy, the others must be transferred to a thrombectomy-capable hospital for treatment, often losing over 2 hours of time to treatment,” said Dr. Fifi. “Every minute is precious in treating stroke, and getting to a center that offers thrombectomy is very important. The MIST model would address this by providing faster access to this potentially life-saving, disability-reducing procedure.”
Access to timely endovascular thrombectomy is gradually improving as “more institutions and cities have implemented the [MIST] model.” Dr. Fifi said.
“This study stresses the importance of ‘time is brain,’ especially for patients in the early time window. Although the study is limited by the observational, retrospective design and was performed at a single integrated center, the findings are provocative,” said Louise McCullough, MD, of the University of Texas Health Science Center at Houston said in a statement to the American Heart Association. “The use of a MIST model highlights the potential benefit of early and urgent treatment for patients with large-vessel stroke. Stroke systems of care need to take advantage of any opportunity to treat patients early, wherever they are.”
The study was partly funded by a Stryker Foundation grant.
Having a mobile interventional stroke team (MIST) travel to treat stroke patients soon after stroke onset may improve patient outcomes, according to a new study. A retrospective analysis of a pilot program in New York found that
“The use of a Mobile Interventional Stroke Team (MIST) traveling to Thrombectomy Capable Stroke Centers to perform endovascular thrombectomy has been shown to be significantly faster with improved discharge outcomes,” wrote lead author Jacob Morey, a doctoral Candidate at Icahn School of Medicine at Mount Sinai in New York and coauthors in the paper. Prior to this study, “the effect of the MIST model stratified by time of presentation” had yet to be studied.
The findings were published online on Aug. 5 in Stroke.
MIST model versus drip-and-ship
The researchers analyzed 226 patients who underwent endovascular thrombectomy between January 2017 and February 2020 at four hospitals in the Mount Sinai health system using the NYC MIST Trial and a stroke database. At baseline, all patients were functionally independent as assessed by the modified Rankin Scale (mRS, score of 0-2). 106 patients were treated by a MIST team – staffed by a neurointerventionalist, a fellow or physician assistant, and radiologic technologist – that traveled to the patient’s location. A total of 120 patients were transferred to a comprehensive stroke center (CSC) or a hospital with endovascular thrombectomy expertise. The analysis was stratified based on whether the patient presented in the early time window (≤ 6 hours) or late time window (> 6 hours).
Patients treated in the early time window were significantly more likely to be mobile and able to perform daily tasks (mRS ≤ 2) 90 days after the procedure in the MIST group (54%), compared with the transferred group (28%, P < 0.01). Outcomes did not differ significantly between groups in the late time window (35% vs. 41%, P = 0.77).
Similarly, early-time-window patients in the MIST group were more likely to have higher functionality at discharge, compared with transferred patients, based on the on the National Institutes of Health Stroke Scale (median score of 5.0 vs. 12.0, P < 0.01). There was no significant difference between groups treated in the late time window (median score of 5.0 vs. 11.0, P = 0.11).
“Ischemic strokes often progress rapidly and can cause severe damage because brain tissue dies quickly without oxygen, resulting in serious long-term disabilities or death,“ said Johanna Fifi, MD, of Icahn School of Medicine, said in a statement to the American Heart Association. “Assessing and treating stroke patients in the early window means that a greater number of fast-progressing strokes are identified and treated.”
Time is brain
Endovascular thrombectomy is a time-sensitive surgical procedure to remove large blood clots in acute ischemic stroke that has “historically been limited to comprehensive stroke centers,” the authors wrote in their paper. It is considered the standard of care in ischemic strokes, which make up 90% of all strokes. “Less than 50% of Americans have direct access to endovascular thrombectomy, the others must be transferred to a thrombectomy-capable hospital for treatment, often losing over 2 hours of time to treatment,” said Dr. Fifi. “Every minute is precious in treating stroke, and getting to a center that offers thrombectomy is very important. The MIST model would address this by providing faster access to this potentially life-saving, disability-reducing procedure.”
Access to timely endovascular thrombectomy is gradually improving as “more institutions and cities have implemented the [MIST] model.” Dr. Fifi said.
“This study stresses the importance of ‘time is brain,’ especially for patients in the early time window. Although the study is limited by the observational, retrospective design and was performed at a single integrated center, the findings are provocative,” said Louise McCullough, MD, of the University of Texas Health Science Center at Houston said in a statement to the American Heart Association. “The use of a MIST model highlights the potential benefit of early and urgent treatment for patients with large-vessel stroke. Stroke systems of care need to take advantage of any opportunity to treat patients early, wherever they are.”
The study was partly funded by a Stryker Foundation grant.
FROM STROKE
Fulminant Hemorrhagic Bullae of the Upper Extremities Arising in the Setting of IV Placement During Severe COVID-19 Infection: Observations From a Major Consultative Practice
To the Editor:
A range of dermatologic manifestations of COVID-19 have been reported, including nonspecific maculopapular exanthems, urticaria, and varicellalike eruptions.1 Additionally, there have been sporadic accounts of cutaneous vasculopathic signs such as perniolike lesions, acro-ischemia, livedo reticularis, and retiform purpura.2 We describe exuberant hemorrhagic bullae occurring on the extremities of 2 critically ill patients with COVID-19. We hypothesized that the bullae were vasculopathic in nature and possibly exacerbated by peripheral intravenous (IV)–related injury.
A 62-year-old woman with a history of diabetes mellitus and chronic obstructive pulmonary disease was admitted to the intensive care unit for acute hypoxemic respiratory failure secondary to COVID-19 infection. Dermatology was consulted for evaluation of blisters on the right arm. A new peripheral IV line was inserted into the patient’s right forearm for treatment of secondary methicillin-resistant Staphylococcus aureus pneumonia. The peripheral IV was inserted into the right proximal forearm for 2 days prior to development of ecchymosis and blisters. Intravenous medications included vancomycin, cefepime, methylprednisolone, and famotidine, as well as maintenance fluids (normal saline). Physical examination revealed extensive confluent ecchymoses with overlying tense bullae (Figure 1). Notable laboratory findings included an elevated D-dimer (peak of 8.67 μg/mL fibrinogen-equivalent units [FEUs], reference range <0.5 μg/mL FEU) and fibrinogen (789 mg/dL, reference range 200–400 mg/dL) levels. Three days later she developed worsening edema of the right arm, accompanied by more extensive bullae formation (Figure 2). Computed tomography of the right arm showed extensive subcutaneous stranding and subcutaneous edema. An orthopedic consultation determined that there was no compartment syndrome, and surgical intervention was not recommended. The patient’s course was complicated by multiorgan failure, and she died 18 days after admission.
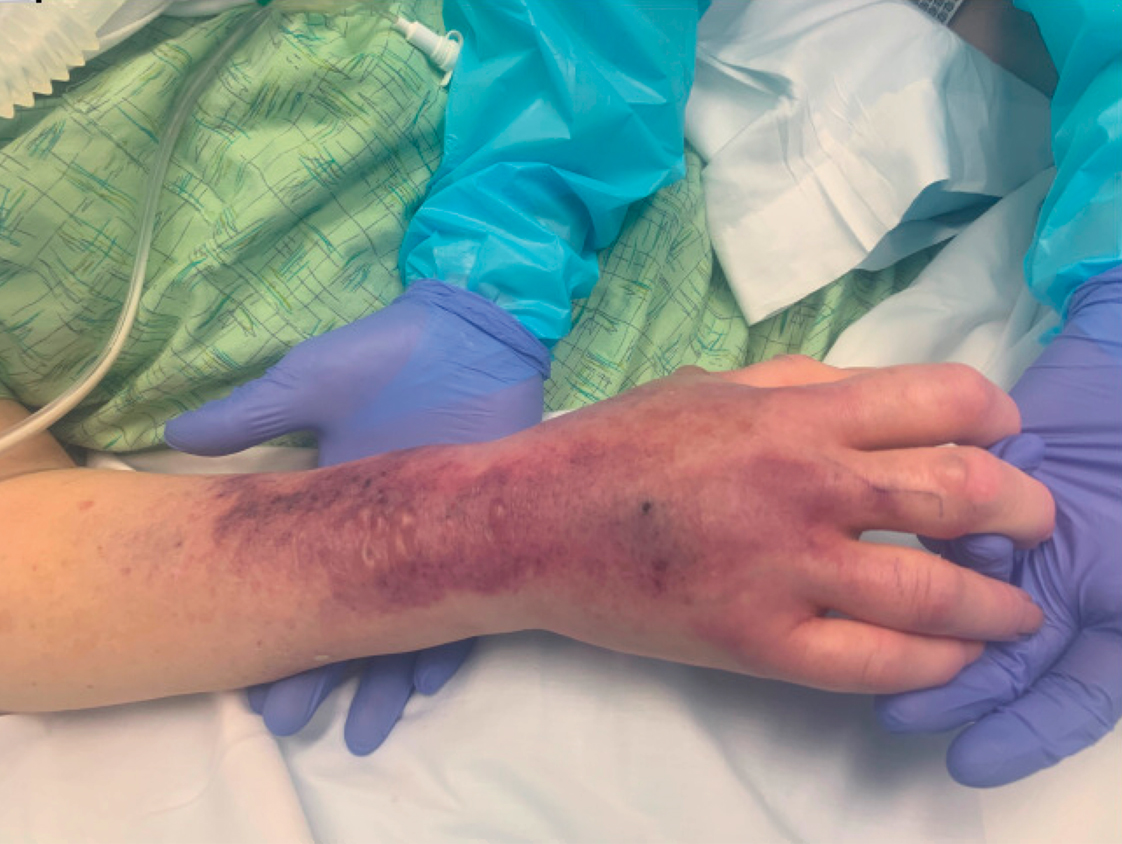
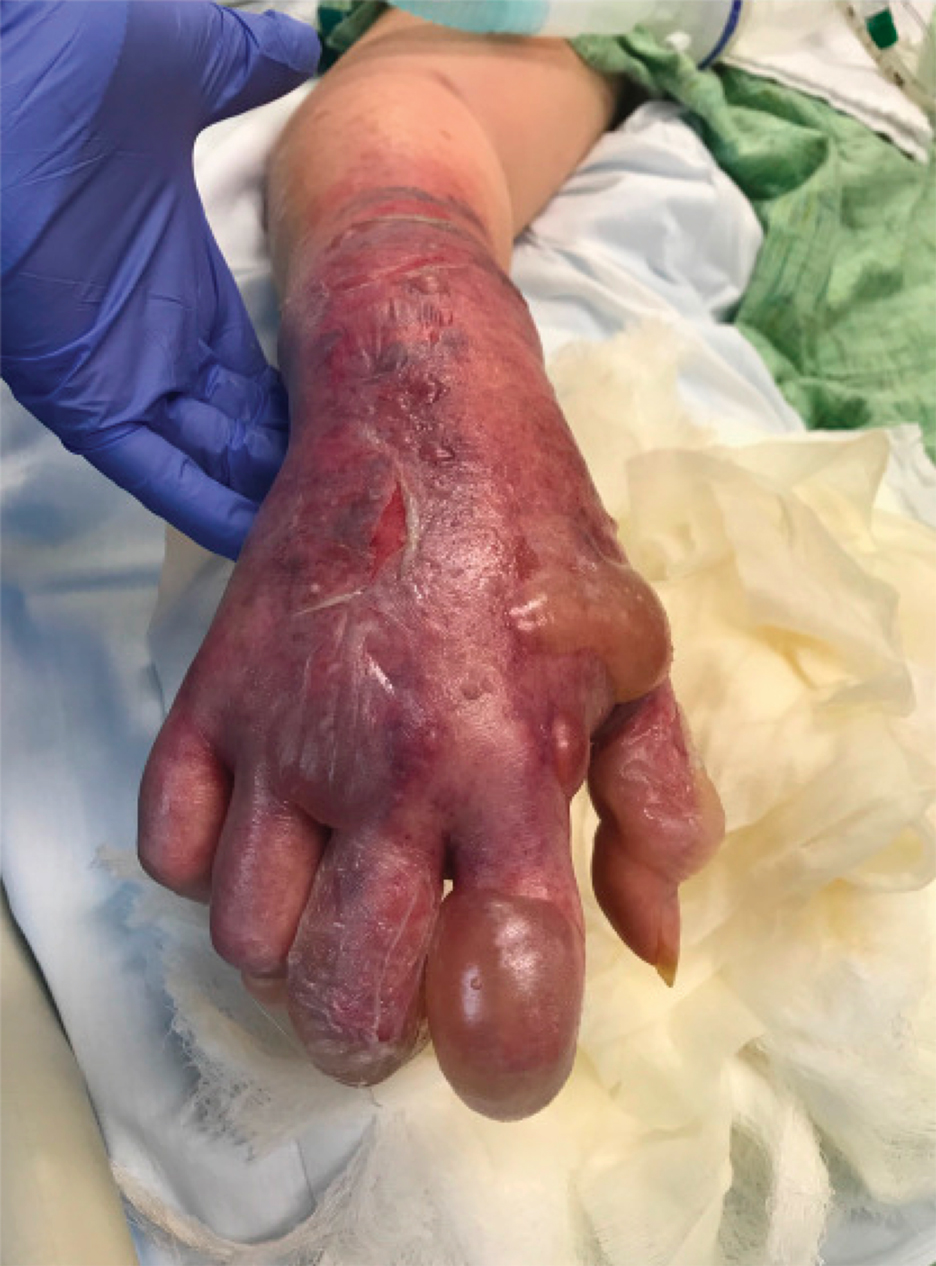
A 67-year-old man with coronary artery disease, diabetes mellitus, and hemiparesis secondary to stroke was admitted to the intensive care unit due to hypoxemia secondary to COVID-19 pneumonia. Dermatology was consulted for the evaluation of blisters on both arms. The right forearm peripheral IV line was used for 4 days prior to the development of cutaneous symptoms. Intravenous medications included cefepime, famotidine, and methylprednisolone. The left forearm peripheral IV line was in place for 1 day prior to the development of blisters and was used for the infusion of maintenance fluids (lactated Ringer’s solution). On the first day of the eruption, small bullae were noted at sites of prior peripheral IV lines (Figure 3). On day 3 of admission, the eruption progressed to larger and more confluent tense bullae with ecchymosis (Figure 4). Additionally, laboratory test results were notable for an elevated D-dimer (peak of >20.00 ug/mL FEU) and fibrinogen (748 mg/dL) levels. Computed tomography of the arms showed extensive subcutaneous stranding and fluid along the fascial planes of the arms, with no gas or abscess formation. Surgical intervention was not recommended following an orthopedic consultation. The patient’s course was complicated by acute kidney injury and rhabdomyolysis; he was later discharged to a skilled nursing facility in stable condition.

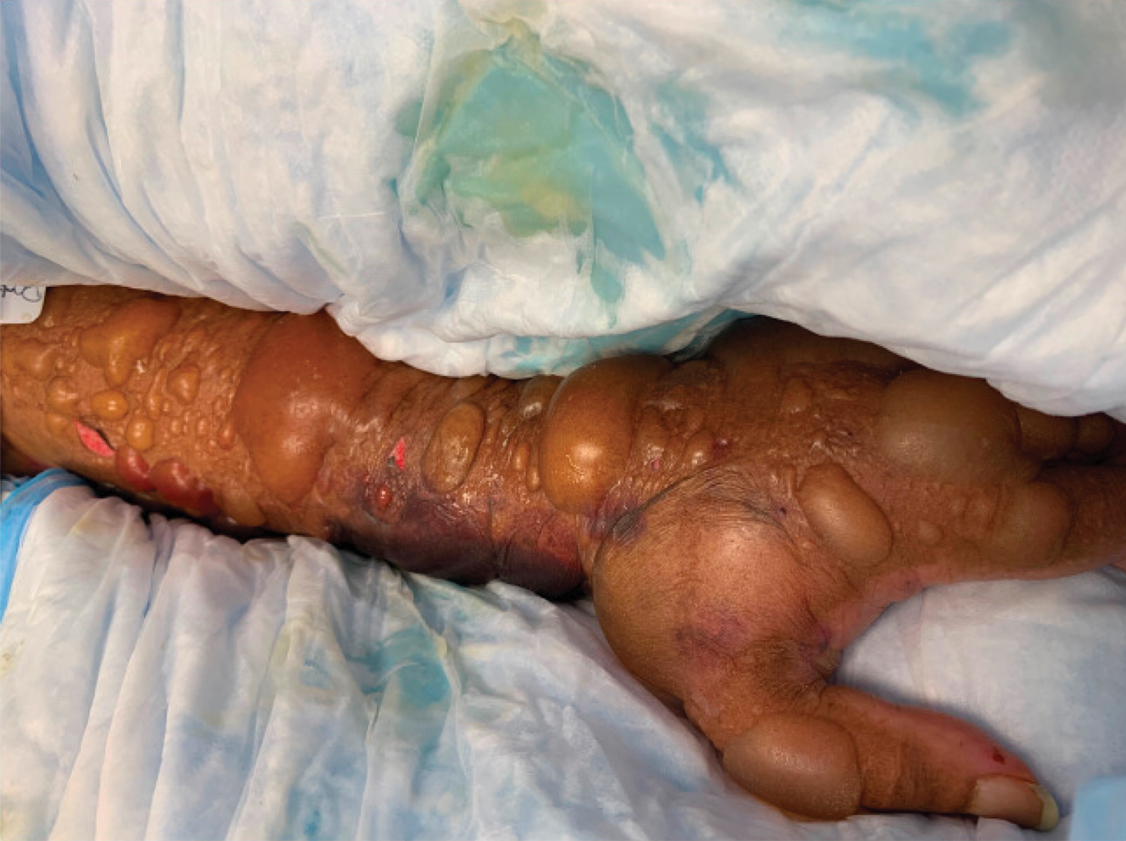
Reports from China indicate that approximately 50% of COVID-19 patients have elevated D-dimer levels and are at risk for thrombosis.3 We hypothesize that the exuberant hemorrhagic bullous eruptions in our 2 cases may be mediated in part by a hypercoagulable state secondary to COVID-19 infection combined with IV-related trauma or extravasation injury. However, a direct cytotoxic effect of the virus cannot be entirely excluded as a potential inciting factor. Other entities considered in the differential for localized bullae included trauma-induced bullous pemphigoid as well as bullous cellulitis. Both patients were treated with high-dose steroids as well as broad-spectrum antibiotics, which were expected to lead to improvement in symptoms of bullous pemphigoid and cellulitis, respectively; however, they did not lead to symptom improvement.
Extravasation injury results from unintentional administration of potentially vesicant substances into tissues surrounding the intended vascular channel.4 The mechanism of action of these injuries is postulated to arise from direct tissue injury from cytotoxic substances, elevated osmotic pressure, and reduced blood supply if vasoconstrictive substances are infused.5 In our patients, these injuries also may have promoted vascular occlusion leading to the brisk reaction observed. Although ecchymoses typically are associated with hypocoagulable states, both of our patients were noted to have normal platelet levels throughout hospitalization. Additionally, findings of elevated D-dimer and fibrinogen levels point to a hypercoagulable state. However, there is a possibility of platelet dysfunction leading to the observed cutaneous findings of ecchymoses. Thrombocytopenia is a common finding in patients with COVID-19 and is found to be associated with increased in-hospital mortality.6 Additional study of these reactions is needed given the propensity for multiorgan failure and death in patients with COVID-19 from suspected diffuse microvascular damage.3
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective [published online March 26, 2020]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.16387
- Zhang Y, Cao W, Xiao M, et al. Clinical and coagulation characteristics of 7 patients with critical COVID-19 pneumonia and acro-ischemia [in Chinese][published online March 28, 2020]. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E006.
- Mei H, Hu Y. Characteristics, causes, diagnosis and treatment of coagulation dysfunction in patients with COVID-19 [in Chinese][published online March 14, 2020]. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E002.
- Sauerland C, Engelking C, Wickham R, et al. Vesicant extravasation part I: mechanisms, pathogenesis, and nursing care to reduce risk. Oncol Nurs Forum. 2006;33:1134-1141.
- Reynolds PM, MacLaren R, Mueller SW, et al. Management of extravasation injuries: a focused evaluation of noncytotoxic medications. Pharmacotherapy. 2014;34:617-632.
- Yang X, Yang Q, Wang Y, et al. Thrombocytopenia and its association with mortality in patients with COVID-19. J Thromb Haemost. 2020;18:1469‐1472.
To the Editor:
A range of dermatologic manifestations of COVID-19 have been reported, including nonspecific maculopapular exanthems, urticaria, and varicellalike eruptions.1 Additionally, there have been sporadic accounts of cutaneous vasculopathic signs such as perniolike lesions, acro-ischemia, livedo reticularis, and retiform purpura.2 We describe exuberant hemorrhagic bullae occurring on the extremities of 2 critically ill patients with COVID-19. We hypothesized that the bullae were vasculopathic in nature and possibly exacerbated by peripheral intravenous (IV)–related injury.
A 62-year-old woman with a history of diabetes mellitus and chronic obstructive pulmonary disease was admitted to the intensive care unit for acute hypoxemic respiratory failure secondary to COVID-19 infection. Dermatology was consulted for evaluation of blisters on the right arm. A new peripheral IV line was inserted into the patient’s right forearm for treatment of secondary methicillin-resistant Staphylococcus aureus pneumonia. The peripheral IV was inserted into the right proximal forearm for 2 days prior to development of ecchymosis and blisters. Intravenous medications included vancomycin, cefepime, methylprednisolone, and famotidine, as well as maintenance fluids (normal saline). Physical examination revealed extensive confluent ecchymoses with overlying tense bullae (Figure 1). Notable laboratory findings included an elevated D-dimer (peak of 8.67 μg/mL fibrinogen-equivalent units [FEUs], reference range <0.5 μg/mL FEU) and fibrinogen (789 mg/dL, reference range 200–400 mg/dL) levels. Three days later she developed worsening edema of the right arm, accompanied by more extensive bullae formation (Figure 2). Computed tomography of the right arm showed extensive subcutaneous stranding and subcutaneous edema. An orthopedic consultation determined that there was no compartment syndrome, and surgical intervention was not recommended. The patient’s course was complicated by multiorgan failure, and she died 18 days after admission.


A 67-year-old man with coronary artery disease, diabetes mellitus, and hemiparesis secondary to stroke was admitted to the intensive care unit due to hypoxemia secondary to COVID-19 pneumonia. Dermatology was consulted for the evaluation of blisters on both arms. The right forearm peripheral IV line was used for 4 days prior to the development of cutaneous symptoms. Intravenous medications included cefepime, famotidine, and methylprednisolone. The left forearm peripheral IV line was in place for 1 day prior to the development of blisters and was used for the infusion of maintenance fluids (lactated Ringer’s solution). On the first day of the eruption, small bullae were noted at sites of prior peripheral IV lines (Figure 3). On day 3 of admission, the eruption progressed to larger and more confluent tense bullae with ecchymosis (Figure 4). Additionally, laboratory test results were notable for an elevated D-dimer (peak of >20.00 ug/mL FEU) and fibrinogen (748 mg/dL) levels. Computed tomography of the arms showed extensive subcutaneous stranding and fluid along the fascial planes of the arms, with no gas or abscess formation. Surgical intervention was not recommended following an orthopedic consultation. The patient’s course was complicated by acute kidney injury and rhabdomyolysis; he was later discharged to a skilled nursing facility in stable condition.


Reports from China indicate that approximately 50% of COVID-19 patients have elevated D-dimer levels and are at risk for thrombosis.3 We hypothesize that the exuberant hemorrhagic bullous eruptions in our 2 cases may be mediated in part by a hypercoagulable state secondary to COVID-19 infection combined with IV-related trauma or extravasation injury. However, a direct cytotoxic effect of the virus cannot be entirely excluded as a potential inciting factor. Other entities considered in the differential for localized bullae included trauma-induced bullous pemphigoid as well as bullous cellulitis. Both patients were treated with high-dose steroids as well as broad-spectrum antibiotics, which were expected to lead to improvement in symptoms of bullous pemphigoid and cellulitis, respectively; however, they did not lead to symptom improvement.
Extravasation injury results from unintentional administration of potentially vesicant substances into tissues surrounding the intended vascular channel.4 The mechanism of action of these injuries is postulated to arise from direct tissue injury from cytotoxic substances, elevated osmotic pressure, and reduced blood supply if vasoconstrictive substances are infused.5 In our patients, these injuries also may have promoted vascular occlusion leading to the brisk reaction observed. Although ecchymoses typically are associated with hypocoagulable states, both of our patients were noted to have normal platelet levels throughout hospitalization. Additionally, findings of elevated D-dimer and fibrinogen levels point to a hypercoagulable state. However, there is a possibility of platelet dysfunction leading to the observed cutaneous findings of ecchymoses. Thrombocytopenia is a common finding in patients with COVID-19 and is found to be associated with increased in-hospital mortality.6 Additional study of these reactions is needed given the propensity for multiorgan failure and death in patients with COVID-19 from suspected diffuse microvascular damage.3
To the Editor:
A range of dermatologic manifestations of COVID-19 have been reported, including nonspecific maculopapular exanthems, urticaria, and varicellalike eruptions.1 Additionally, there have been sporadic accounts of cutaneous vasculopathic signs such as perniolike lesions, acro-ischemia, livedo reticularis, and retiform purpura.2 We describe exuberant hemorrhagic bullae occurring on the extremities of 2 critically ill patients with COVID-19. We hypothesized that the bullae were vasculopathic in nature and possibly exacerbated by peripheral intravenous (IV)–related injury.
A 62-year-old woman with a history of diabetes mellitus and chronic obstructive pulmonary disease was admitted to the intensive care unit for acute hypoxemic respiratory failure secondary to COVID-19 infection. Dermatology was consulted for evaluation of blisters on the right arm. A new peripheral IV line was inserted into the patient’s right forearm for treatment of secondary methicillin-resistant Staphylococcus aureus pneumonia. The peripheral IV was inserted into the right proximal forearm for 2 days prior to development of ecchymosis and blisters. Intravenous medications included vancomycin, cefepime, methylprednisolone, and famotidine, as well as maintenance fluids (normal saline). Physical examination revealed extensive confluent ecchymoses with overlying tense bullae (Figure 1). Notable laboratory findings included an elevated D-dimer (peak of 8.67 μg/mL fibrinogen-equivalent units [FEUs], reference range <0.5 μg/mL FEU) and fibrinogen (789 mg/dL, reference range 200–400 mg/dL) levels. Three days later she developed worsening edema of the right arm, accompanied by more extensive bullae formation (Figure 2). Computed tomography of the right arm showed extensive subcutaneous stranding and subcutaneous edema. An orthopedic consultation determined that there was no compartment syndrome, and surgical intervention was not recommended. The patient’s course was complicated by multiorgan failure, and she died 18 days after admission.


A 67-year-old man with coronary artery disease, diabetes mellitus, and hemiparesis secondary to stroke was admitted to the intensive care unit due to hypoxemia secondary to COVID-19 pneumonia. Dermatology was consulted for the evaluation of blisters on both arms. The right forearm peripheral IV line was used for 4 days prior to the development of cutaneous symptoms. Intravenous medications included cefepime, famotidine, and methylprednisolone. The left forearm peripheral IV line was in place for 1 day prior to the development of blisters and was used for the infusion of maintenance fluids (lactated Ringer’s solution). On the first day of the eruption, small bullae were noted at sites of prior peripheral IV lines (Figure 3). On day 3 of admission, the eruption progressed to larger and more confluent tense bullae with ecchymosis (Figure 4). Additionally, laboratory test results were notable for an elevated D-dimer (peak of >20.00 ug/mL FEU) and fibrinogen (748 mg/dL) levels. Computed tomography of the arms showed extensive subcutaneous stranding and fluid along the fascial planes of the arms, with no gas or abscess formation. Surgical intervention was not recommended following an orthopedic consultation. The patient’s course was complicated by acute kidney injury and rhabdomyolysis; he was later discharged to a skilled nursing facility in stable condition.


Reports from China indicate that approximately 50% of COVID-19 patients have elevated D-dimer levels and are at risk for thrombosis.3 We hypothesize that the exuberant hemorrhagic bullous eruptions in our 2 cases may be mediated in part by a hypercoagulable state secondary to COVID-19 infection combined with IV-related trauma or extravasation injury. However, a direct cytotoxic effect of the virus cannot be entirely excluded as a potential inciting factor. Other entities considered in the differential for localized bullae included trauma-induced bullous pemphigoid as well as bullous cellulitis. Both patients were treated with high-dose steroids as well as broad-spectrum antibiotics, which were expected to lead to improvement in symptoms of bullous pemphigoid and cellulitis, respectively; however, they did not lead to symptom improvement.
Extravasation injury results from unintentional administration of potentially vesicant substances into tissues surrounding the intended vascular channel.4 The mechanism of action of these injuries is postulated to arise from direct tissue injury from cytotoxic substances, elevated osmotic pressure, and reduced blood supply if vasoconstrictive substances are infused.5 In our patients, these injuries also may have promoted vascular occlusion leading to the brisk reaction observed. Although ecchymoses typically are associated with hypocoagulable states, both of our patients were noted to have normal platelet levels throughout hospitalization. Additionally, findings of elevated D-dimer and fibrinogen levels point to a hypercoagulable state. However, there is a possibility of platelet dysfunction leading to the observed cutaneous findings of ecchymoses. Thrombocytopenia is a common finding in patients with COVID-19 and is found to be associated with increased in-hospital mortality.6 Additional study of these reactions is needed given the propensity for multiorgan failure and death in patients with COVID-19 from suspected diffuse microvascular damage.3
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective [published online March 26, 2020]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.16387
- Zhang Y, Cao W, Xiao M, et al. Clinical and coagulation characteristics of 7 patients with critical COVID-19 pneumonia and acro-ischemia [in Chinese][published online March 28, 2020]. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E006.
- Mei H, Hu Y. Characteristics, causes, diagnosis and treatment of coagulation dysfunction in patients with COVID-19 [in Chinese][published online March 14, 2020]. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E002.
- Sauerland C, Engelking C, Wickham R, et al. Vesicant extravasation part I: mechanisms, pathogenesis, and nursing care to reduce risk. Oncol Nurs Forum. 2006;33:1134-1141.
- Reynolds PM, MacLaren R, Mueller SW, et al. Management of extravasation injuries: a focused evaluation of noncytotoxic medications. Pharmacotherapy. 2014;34:617-632.
- Yang X, Yang Q, Wang Y, et al. Thrombocytopenia and its association with mortality in patients with COVID-19. J Thromb Haemost. 2020;18:1469‐1472.
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective [published online March 26, 2020]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.16387
- Zhang Y, Cao W, Xiao M, et al. Clinical and coagulation characteristics of 7 patients with critical COVID-19 pneumonia and acro-ischemia [in Chinese][published online March 28, 2020]. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E006.
- Mei H, Hu Y. Characteristics, causes, diagnosis and treatment of coagulation dysfunction in patients with COVID-19 [in Chinese][published online March 14, 2020]. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E002.
- Sauerland C, Engelking C, Wickham R, et al. Vesicant extravasation part I: mechanisms, pathogenesis, and nursing care to reduce risk. Oncol Nurs Forum. 2006;33:1134-1141.
- Reynolds PM, MacLaren R, Mueller SW, et al. Management of extravasation injuries: a focused evaluation of noncytotoxic medications. Pharmacotherapy. 2014;34:617-632.
- Yang X, Yang Q, Wang Y, et al. Thrombocytopenia and its association with mortality in patients with COVID-19. J Thromb Haemost. 2020;18:1469‐1472.
Practice Points
- Hemorrhagic bullae are an uncommon cutaneous manifestation of COVID-19 infection in hospitalized individuals.
- Although there is no reported treatment for COVID-19–associated hemorrhagic bullae, we recommend supportive care and management of underlying etiology.
How many years of aromatase inhibitor therapy in breast cancer?
That’s a question that has been vexing oncologists for more than a decade, but final results from a large study now suggest that, after an initial 5 years of adjuvant endocrine therapy with tamoxifen and/or an AI, an additional 5 years of AI therapy offer no clinical benefits, compared with an additional 2 years of therapy.
What’s more, 5 years of additional treatment were associated with a significantly increased risk for fractures, compared with 2 years of therapy, Michael Gnant, MD, from the Medical University of Vienna, and colleagues reported in the phase 3 Austrian Breast and Colorectal Cancer Study Group Trial 16 (ABCSG-16).
“Thus, in postmenopausal women with hormone receptor–positive breast cancer who had received 5 years of adjuvant endocrine therapy, the extension of aromatase inhibitor therapy for 2 years rather than 5 years was sufficient to maximize the benefits of such therapy in most patients without extending the exposure to toxic effects,” they concluded.
The study was published online on July 28, 2021, in the New England Journal of Medicine.
The new results “in patients with hormone-receptor–positive breast cancer who are at low or average risk underscore the importance of avoiding iatrogenic complications when making these decisions,” Pamela J. Goodwin, MD, from the Lunenfeld–Tanenbaum Research Institute at the University of Toronto, wrote in an accompanying editorial.
She noted that these latest results echo those of two earlier studies: the DATA trial and the optimistically named IDEAL trial, both of which showed no benefit of short-term versus longer-term adjuvant endocrine therapy with regard to either disease-free survival (DFS) or overall survival (OS).
“These results provide strong evidence against the routine use of more than 2 years of extended aromatase inhibitor therapy in women who are at low or average risk, similar to those who were included in this trial,” Dr. Goodwin commented.
ABCSG-16 details
Dr. Gnant and colleagues enrolled 3,484 women aged 80 years or younger with HR-positive stage I, II, or III breast cancer for whom there was no evidence of recurrence at baseline. The patients had all received adjuvant tamoxifen or an AI for 5 years; for some patients, the two drugs were administered in sequence until 12 months before randomization.
The patients were randomly assigned to receive oral anastrozole 1 mg daily for either 2 or 5 additional years.
Among all patients, 3,208 had experienced no recurrence 2 years after randomization. These patients were included in the primary DFS analysis. Of this group, 1,635 (51%) had received tamoxifen alone for 5 years, 235 (7.3%) had received an AI alone, and 41.7% had received sequential AIs and tamoxifen.
At a median follow-up of 118 months after randomization, disease progression or death occurred in 670 women – 335 in each trial arm.
The rate of DFS 10 years after randomization was 73.6% in the 2-year additional-therapy group and 73.9% in the 5-year group. The hazard ratio for disease recurrence or death was 0.99 and was not significant. After adjustment for potential confounding factors, the HR remained essentially unchanged (HR, 1.0).
Overall survival at 8 years, a secondary endpoint, was virtually identical between the groups, at 87.5% in the 2-year group and 87.3% in the 5-year group. The HR for death from any cause was 1.02 and was not significant.
The HR for contralateral breast cancer was 1.15, and the HR for a second primary cancer was 1.06; neither was statistically significant.
Although the use of bone-targeted medication was similar between the groups, among patients in the 2-year group, the incidence of clinical bone fractures 5 years after randomization was lower, at 4.7% versus 6.3%, which translates to an HR for fracture with 5 additional years of AI therapy of 1.35 (95% confidence interval, 1.00-1.84).
Adverse events with anastrozole were consistent with its known toxicity profile. At least one serious adverse event occurred in 26.5% of patients in the 2-year group and in 40.2% in the 5-year group. Investigator-assessed serious adverse events that were judged to be related to anastrozole occurred in 2.3% and 4.0% of patients, respectively.
Osteoarthritis, the most frequently reported adverse event, was documented in 1.7% of patients in the 2-year group and in 4.3% in the 5-year group.
AI duration still unclear
“We did not investigate the value of extending adjuvant endocrine therapy per se, since the benefit of extending aromatase inhibitors after 5 years of adjuvant tamoxifen has been well established. In contrast, the most effective duration of adjuvant aromatase inhibitor therapy remains unclear in randomized trials,” Dr. Gnant and colleagues commented.
In her editorial, Dr. Goodwin summarized potential approaches for improving late outcomes for patients with hormone receptor–positive breast cancers, including the use of biomarkers to detect minimal residual disease and therapies to treat it.
She noted that the detection of circulating tumor cells in patients with HR-positive breast cancer 5 years after diagnosis is associated with a 13-fold increase in risk for recurrence and a median time to clinical recurrence of 2.8 years.
“This interval may be sufficiently long that therapeutic intervention can prevent the development of incurable clinical metastases. Continued improvement in these assays and the development of new therapies that target the unique biologic features of dormant cells will no doubt be required for a major reduction in late recurrence risk,” she wrote.
The study was supported by AstraZeneca and the Austrian Breast and Colorectal Cancer Study Group. Dr. Gnant has received lecture fees from AstraZeneca and others. Dr. Goodwin has received institutional research funding from the Breast Cancer Research Foundation and EPIC Sciences.
A version of this article first appeared on Medscape.com.
That’s a question that has been vexing oncologists for more than a decade, but final results from a large study now suggest that, after an initial 5 years of adjuvant endocrine therapy with tamoxifen and/or an AI, an additional 5 years of AI therapy offer no clinical benefits, compared with an additional 2 years of therapy.
What’s more, 5 years of additional treatment were associated with a significantly increased risk for fractures, compared with 2 years of therapy, Michael Gnant, MD, from the Medical University of Vienna, and colleagues reported in the phase 3 Austrian Breast and Colorectal Cancer Study Group Trial 16 (ABCSG-16).
“Thus, in postmenopausal women with hormone receptor–positive breast cancer who had received 5 years of adjuvant endocrine therapy, the extension of aromatase inhibitor therapy for 2 years rather than 5 years was sufficient to maximize the benefits of such therapy in most patients without extending the exposure to toxic effects,” they concluded.
The study was published online on July 28, 2021, in the New England Journal of Medicine.
The new results “in patients with hormone-receptor–positive breast cancer who are at low or average risk underscore the importance of avoiding iatrogenic complications when making these decisions,” Pamela J. Goodwin, MD, from the Lunenfeld–Tanenbaum Research Institute at the University of Toronto, wrote in an accompanying editorial.
She noted that these latest results echo those of two earlier studies: the DATA trial and the optimistically named IDEAL trial, both of which showed no benefit of short-term versus longer-term adjuvant endocrine therapy with regard to either disease-free survival (DFS) or overall survival (OS).
“These results provide strong evidence against the routine use of more than 2 years of extended aromatase inhibitor therapy in women who are at low or average risk, similar to those who were included in this trial,” Dr. Goodwin commented.
ABCSG-16 details
Dr. Gnant and colleagues enrolled 3,484 women aged 80 years or younger with HR-positive stage I, II, or III breast cancer for whom there was no evidence of recurrence at baseline. The patients had all received adjuvant tamoxifen or an AI for 5 years; for some patients, the two drugs were administered in sequence until 12 months before randomization.
The patients were randomly assigned to receive oral anastrozole 1 mg daily for either 2 or 5 additional years.
Among all patients, 3,208 had experienced no recurrence 2 years after randomization. These patients were included in the primary DFS analysis. Of this group, 1,635 (51%) had received tamoxifen alone for 5 years, 235 (7.3%) had received an AI alone, and 41.7% had received sequential AIs and tamoxifen.
At a median follow-up of 118 months after randomization, disease progression or death occurred in 670 women – 335 in each trial arm.
The rate of DFS 10 years after randomization was 73.6% in the 2-year additional-therapy group and 73.9% in the 5-year group. The hazard ratio for disease recurrence or death was 0.99 and was not significant. After adjustment for potential confounding factors, the HR remained essentially unchanged (HR, 1.0).
Overall survival at 8 years, a secondary endpoint, was virtually identical between the groups, at 87.5% in the 2-year group and 87.3% in the 5-year group. The HR for death from any cause was 1.02 and was not significant.
The HR for contralateral breast cancer was 1.15, and the HR for a second primary cancer was 1.06; neither was statistically significant.
Although the use of bone-targeted medication was similar between the groups, among patients in the 2-year group, the incidence of clinical bone fractures 5 years after randomization was lower, at 4.7% versus 6.3%, which translates to an HR for fracture with 5 additional years of AI therapy of 1.35 (95% confidence interval, 1.00-1.84).
Adverse events with anastrozole were consistent with its known toxicity profile. At least one serious adverse event occurred in 26.5% of patients in the 2-year group and in 40.2% in the 5-year group. Investigator-assessed serious adverse events that were judged to be related to anastrozole occurred in 2.3% and 4.0% of patients, respectively.
Osteoarthritis, the most frequently reported adverse event, was documented in 1.7% of patients in the 2-year group and in 4.3% in the 5-year group.
AI duration still unclear
“We did not investigate the value of extending adjuvant endocrine therapy per se, since the benefit of extending aromatase inhibitors after 5 years of adjuvant tamoxifen has been well established. In contrast, the most effective duration of adjuvant aromatase inhibitor therapy remains unclear in randomized trials,” Dr. Gnant and colleagues commented.
In her editorial, Dr. Goodwin summarized potential approaches for improving late outcomes for patients with hormone receptor–positive breast cancers, including the use of biomarkers to detect minimal residual disease and therapies to treat it.
She noted that the detection of circulating tumor cells in patients with HR-positive breast cancer 5 years after diagnosis is associated with a 13-fold increase in risk for recurrence and a median time to clinical recurrence of 2.8 years.
“This interval may be sufficiently long that therapeutic intervention can prevent the development of incurable clinical metastases. Continued improvement in these assays and the development of new therapies that target the unique biologic features of dormant cells will no doubt be required for a major reduction in late recurrence risk,” she wrote.
The study was supported by AstraZeneca and the Austrian Breast and Colorectal Cancer Study Group. Dr. Gnant has received lecture fees from AstraZeneca and others. Dr. Goodwin has received institutional research funding from the Breast Cancer Research Foundation and EPIC Sciences.
A version of this article first appeared on Medscape.com.
That’s a question that has been vexing oncologists for more than a decade, but final results from a large study now suggest that, after an initial 5 years of adjuvant endocrine therapy with tamoxifen and/or an AI, an additional 5 years of AI therapy offer no clinical benefits, compared with an additional 2 years of therapy.
What’s more, 5 years of additional treatment were associated with a significantly increased risk for fractures, compared with 2 years of therapy, Michael Gnant, MD, from the Medical University of Vienna, and colleagues reported in the phase 3 Austrian Breast and Colorectal Cancer Study Group Trial 16 (ABCSG-16).
“Thus, in postmenopausal women with hormone receptor–positive breast cancer who had received 5 years of adjuvant endocrine therapy, the extension of aromatase inhibitor therapy for 2 years rather than 5 years was sufficient to maximize the benefits of such therapy in most patients without extending the exposure to toxic effects,” they concluded.
The study was published online on July 28, 2021, in the New England Journal of Medicine.
The new results “in patients with hormone-receptor–positive breast cancer who are at low or average risk underscore the importance of avoiding iatrogenic complications when making these decisions,” Pamela J. Goodwin, MD, from the Lunenfeld–Tanenbaum Research Institute at the University of Toronto, wrote in an accompanying editorial.
She noted that these latest results echo those of two earlier studies: the DATA trial and the optimistically named IDEAL trial, both of which showed no benefit of short-term versus longer-term adjuvant endocrine therapy with regard to either disease-free survival (DFS) or overall survival (OS).
“These results provide strong evidence against the routine use of more than 2 years of extended aromatase inhibitor therapy in women who are at low or average risk, similar to those who were included in this trial,” Dr. Goodwin commented.
ABCSG-16 details
Dr. Gnant and colleagues enrolled 3,484 women aged 80 years or younger with HR-positive stage I, II, or III breast cancer for whom there was no evidence of recurrence at baseline. The patients had all received adjuvant tamoxifen or an AI for 5 years; for some patients, the two drugs were administered in sequence until 12 months before randomization.
The patients were randomly assigned to receive oral anastrozole 1 mg daily for either 2 or 5 additional years.
Among all patients, 3,208 had experienced no recurrence 2 years after randomization. These patients were included in the primary DFS analysis. Of this group, 1,635 (51%) had received tamoxifen alone for 5 years, 235 (7.3%) had received an AI alone, and 41.7% had received sequential AIs and tamoxifen.
At a median follow-up of 118 months after randomization, disease progression or death occurred in 670 women – 335 in each trial arm.
The rate of DFS 10 years after randomization was 73.6% in the 2-year additional-therapy group and 73.9% in the 5-year group. The hazard ratio for disease recurrence or death was 0.99 and was not significant. After adjustment for potential confounding factors, the HR remained essentially unchanged (HR, 1.0).
Overall survival at 8 years, a secondary endpoint, was virtually identical between the groups, at 87.5% in the 2-year group and 87.3% in the 5-year group. The HR for death from any cause was 1.02 and was not significant.
The HR for contralateral breast cancer was 1.15, and the HR for a second primary cancer was 1.06; neither was statistically significant.
Although the use of bone-targeted medication was similar between the groups, among patients in the 2-year group, the incidence of clinical bone fractures 5 years after randomization was lower, at 4.7% versus 6.3%, which translates to an HR for fracture with 5 additional years of AI therapy of 1.35 (95% confidence interval, 1.00-1.84).
Adverse events with anastrozole were consistent with its known toxicity profile. At least one serious adverse event occurred in 26.5% of patients in the 2-year group and in 40.2% in the 5-year group. Investigator-assessed serious adverse events that were judged to be related to anastrozole occurred in 2.3% and 4.0% of patients, respectively.
Osteoarthritis, the most frequently reported adverse event, was documented in 1.7% of patients in the 2-year group and in 4.3% in the 5-year group.
AI duration still unclear
“We did not investigate the value of extending adjuvant endocrine therapy per se, since the benefit of extending aromatase inhibitors after 5 years of adjuvant tamoxifen has been well established. In contrast, the most effective duration of adjuvant aromatase inhibitor therapy remains unclear in randomized trials,” Dr. Gnant and colleagues commented.
In her editorial, Dr. Goodwin summarized potential approaches for improving late outcomes for patients with hormone receptor–positive breast cancers, including the use of biomarkers to detect minimal residual disease and therapies to treat it.
She noted that the detection of circulating tumor cells in patients with HR-positive breast cancer 5 years after diagnosis is associated with a 13-fold increase in risk for recurrence and a median time to clinical recurrence of 2.8 years.
“This interval may be sufficiently long that therapeutic intervention can prevent the development of incurable clinical metastases. Continued improvement in these assays and the development of new therapies that target the unique biologic features of dormant cells will no doubt be required for a major reduction in late recurrence risk,” she wrote.
The study was supported by AstraZeneca and the Austrian Breast and Colorectal Cancer Study Group. Dr. Gnant has received lecture fees from AstraZeneca and others. Dr. Goodwin has received institutional research funding from the Breast Cancer Research Foundation and EPIC Sciences.
A version of this article first appeared on Medscape.com.
One in three cancer articles on social media has wrong info
Of the 200 most popular articles (50 each for prostate, lung, breast, and colorectal cancer), about a third (32.5%, n = 65) contained misinformation.
Among these articles containing misinformation, 76.9% (50/65) contained harmful information.
“The Internet is a leading source of health misinformation,” the study authors wrote. This is “particularly true for social media, where false information spreads faster and more broadly than fact-checked information,” they said, citing other research.
“We need to address these issues head on,” said lead author Skyler Johnson, MD, of the University of Utah’s Huntsman Cancer Institute in Salt Lake City.
“As a medical community, we can’t ignore the problem of cancer misinformation on social media or ask our patients to ignore it. We must empathize with our patients and help them when they encounter this type of information,” he said in a statement. “My goal is to help answer their questions, and provide cancer patients with accurate information that will give them the best chance for the best outcome.”
The study was published online July 22 in the Journal of the National Cancer Institute.
The study period ran from 2018 to 2019, and looked at articles posted on social media platforms Facebook, Reddit, Twitter, or Pinterest. Popularity was measured by engagement with readers, such as upvotes, comments, reactions, and shares.
Some of the articles came from long-established news entities such as CBS News, The New York Times, and medical journals, while others came from fleeting crowdfunding web pages and fledging nontraditional news sites.
One example of popular and harmful misinformation highlighted by Dr. Johnson in an interview was titled, “44-Year-Old Mother Claims CBD Oil Cured Her of Breast Cancer within 5 Months.” Posted on truththeory.com in February 2018, the article is tagged as “opinion” by the publisher and in turn links to another news story about the same woman in the UK’s Daily Mail newspaper.
The ideas and claims in such articles can be very influential, Jennifer L. Lycette, MD, suggested in a recent blog post.
“After 18 years as a cancer doctor, it sadly doesn’t come as a surprise anymore when a patient declines treatment recommendations and instead opts for ‘alternative’ treatment,” she wrote.
Sometimes, misinformation is not sensational but is still effective via clever wording and presentation, observed Brian G. Southwell, PhD, of Duke University, Durham, N.C., who has studied patients and misinformation.
“It isn’t the falsehood that is somehow magically attractive, per se, but the way that misinformation is often framed that can make it attractive,” he said in an interview.
Dr. Southwell recommends that clinicians be proactive about medical misinformation.
“Rather than expect patients to raise concerns without prompting, health care providers should invite conversations about potential misinformation with their patients,” he wrote in a recent essay in the American Journal of Public Health.
In short, ask patients what they know about the treatment of their cancer, he suggests.
“Patients don’t typically know that the misinformation they are encountering is misinformation,” said Dr. Southwell. “Approaching patients with compassion and empathy is a good first step.”
Study details
For the study, reported by Johnson et al., two National Comprehensive Cancer Network panel members were selected as content experts for each of the four cancers and were tasked with reviewing the primary medical claims in each article. The experts then completed a set of ratings to arrive at the proportion of misinformation and potential for harm in each article.
Of the 200 articles, 41.5% were from nontraditional news (digital only), 37.5% were from traditional news sources (online versions of print and/or broadcast media), 17% were from medical journals, 3% were from a crowdfunding site, and 1% were from personal blogs.
This expert review concluded that nearly one-third of the articles contained misinformation, as noted above. The misinformation was described as misleading (title not supported by text or statistics/data do not support conclusion, 28.8%), strength of the evidence mischaracterized (weak evidence portrayed as strong or vice versa, 27.7%) and unproven therapies (not studied or insufficient evidence, 26.7%).
Notably, the median number of engagements, such as likes on Twitter, for articles with misinformation was greater than that of factual articles (median, 2,300 vs. 1,600; P = .05).
In total, 30.5% of all 200 articles contained harmful information. This was described as harmful inaction (could lead to delay or not seeking medical attention for treatable/curable condition, 31.0%), economic harm (out-of-pocket financial costs associated with treatment/travel, 27.7%), harmful action (potentially toxic effects of the suggested test/treatment, 17.0%), and harmful interactions (known/unknown medical interactions with curative therapies, 16.2%).
The median number of engagements for articles with harmful information was statistically significantly greater than that of articles with correct information (median, 2,300 vs. 1,500; P = .007).
A limitation of the study is that it included only the most popular English language cancer articles.
This study was funded in part by the Huntsman Cancer Institute. Dr. Johnson, Dr. Lycette, and Dr. Southwell have disclosed no relevant financial relationships. Some study authors have ties to the pharmaceutical industry.
A version of this article first appeared on Medscape.com.
Of the 200 most popular articles (50 each for prostate, lung, breast, and colorectal cancer), about a third (32.5%, n = 65) contained misinformation.
Among these articles containing misinformation, 76.9% (50/65) contained harmful information.
“The Internet is a leading source of health misinformation,” the study authors wrote. This is “particularly true for social media, where false information spreads faster and more broadly than fact-checked information,” they said, citing other research.
“We need to address these issues head on,” said lead author Skyler Johnson, MD, of the University of Utah’s Huntsman Cancer Institute in Salt Lake City.
“As a medical community, we can’t ignore the problem of cancer misinformation on social media or ask our patients to ignore it. We must empathize with our patients and help them when they encounter this type of information,” he said in a statement. “My goal is to help answer their questions, and provide cancer patients with accurate information that will give them the best chance for the best outcome.”
The study was published online July 22 in the Journal of the National Cancer Institute.
The study period ran from 2018 to 2019, and looked at articles posted on social media platforms Facebook, Reddit, Twitter, or Pinterest. Popularity was measured by engagement with readers, such as upvotes, comments, reactions, and shares.
Some of the articles came from long-established news entities such as CBS News, The New York Times, and medical journals, while others came from fleeting crowdfunding web pages and fledging nontraditional news sites.
One example of popular and harmful misinformation highlighted by Dr. Johnson in an interview was titled, “44-Year-Old Mother Claims CBD Oil Cured Her of Breast Cancer within 5 Months.” Posted on truththeory.com in February 2018, the article is tagged as “opinion” by the publisher and in turn links to another news story about the same woman in the UK’s Daily Mail newspaper.
The ideas and claims in such articles can be very influential, Jennifer L. Lycette, MD, suggested in a recent blog post.
“After 18 years as a cancer doctor, it sadly doesn’t come as a surprise anymore when a patient declines treatment recommendations and instead opts for ‘alternative’ treatment,” she wrote.
Sometimes, misinformation is not sensational but is still effective via clever wording and presentation, observed Brian G. Southwell, PhD, of Duke University, Durham, N.C., who has studied patients and misinformation.
“It isn’t the falsehood that is somehow magically attractive, per se, but the way that misinformation is often framed that can make it attractive,” he said in an interview.
Dr. Southwell recommends that clinicians be proactive about medical misinformation.
“Rather than expect patients to raise concerns without prompting, health care providers should invite conversations about potential misinformation with their patients,” he wrote in a recent essay in the American Journal of Public Health.
In short, ask patients what they know about the treatment of their cancer, he suggests.
“Patients don’t typically know that the misinformation they are encountering is misinformation,” said Dr. Southwell. “Approaching patients with compassion and empathy is a good first step.”
Study details
For the study, reported by Johnson et al., two National Comprehensive Cancer Network panel members were selected as content experts for each of the four cancers and were tasked with reviewing the primary medical claims in each article. The experts then completed a set of ratings to arrive at the proportion of misinformation and potential for harm in each article.
Of the 200 articles, 41.5% were from nontraditional news (digital only), 37.5% were from traditional news sources (online versions of print and/or broadcast media), 17% were from medical journals, 3% were from a crowdfunding site, and 1% were from personal blogs.
This expert review concluded that nearly one-third of the articles contained misinformation, as noted above. The misinformation was described as misleading (title not supported by text or statistics/data do not support conclusion, 28.8%), strength of the evidence mischaracterized (weak evidence portrayed as strong or vice versa, 27.7%) and unproven therapies (not studied or insufficient evidence, 26.7%).
Notably, the median number of engagements, such as likes on Twitter, for articles with misinformation was greater than that of factual articles (median, 2,300 vs. 1,600; P = .05).
In total, 30.5% of all 200 articles contained harmful information. This was described as harmful inaction (could lead to delay or not seeking medical attention for treatable/curable condition, 31.0%), economic harm (out-of-pocket financial costs associated with treatment/travel, 27.7%), harmful action (potentially toxic effects of the suggested test/treatment, 17.0%), and harmful interactions (known/unknown medical interactions with curative therapies, 16.2%).
The median number of engagements for articles with harmful information was statistically significantly greater than that of articles with correct information (median, 2,300 vs. 1,500; P = .007).
A limitation of the study is that it included only the most popular English language cancer articles.
This study was funded in part by the Huntsman Cancer Institute. Dr. Johnson, Dr. Lycette, and Dr. Southwell have disclosed no relevant financial relationships. Some study authors have ties to the pharmaceutical industry.
A version of this article first appeared on Medscape.com.
Of the 200 most popular articles (50 each for prostate, lung, breast, and colorectal cancer), about a third (32.5%, n = 65) contained misinformation.
Among these articles containing misinformation, 76.9% (50/65) contained harmful information.
“The Internet is a leading source of health misinformation,” the study authors wrote. This is “particularly true for social media, where false information spreads faster and more broadly than fact-checked information,” they said, citing other research.
“We need to address these issues head on,” said lead author Skyler Johnson, MD, of the University of Utah’s Huntsman Cancer Institute in Salt Lake City.
“As a medical community, we can’t ignore the problem of cancer misinformation on social media or ask our patients to ignore it. We must empathize with our patients and help them when they encounter this type of information,” he said in a statement. “My goal is to help answer their questions, and provide cancer patients with accurate information that will give them the best chance for the best outcome.”
The study was published online July 22 in the Journal of the National Cancer Institute.
The study period ran from 2018 to 2019, and looked at articles posted on social media platforms Facebook, Reddit, Twitter, or Pinterest. Popularity was measured by engagement with readers, such as upvotes, comments, reactions, and shares.
Some of the articles came from long-established news entities such as CBS News, The New York Times, and medical journals, while others came from fleeting crowdfunding web pages and fledging nontraditional news sites.
One example of popular and harmful misinformation highlighted by Dr. Johnson in an interview was titled, “44-Year-Old Mother Claims CBD Oil Cured Her of Breast Cancer within 5 Months.” Posted on truththeory.com in February 2018, the article is tagged as “opinion” by the publisher and in turn links to another news story about the same woman in the UK’s Daily Mail newspaper.
The ideas and claims in such articles can be very influential, Jennifer L. Lycette, MD, suggested in a recent blog post.
“After 18 years as a cancer doctor, it sadly doesn’t come as a surprise anymore when a patient declines treatment recommendations and instead opts for ‘alternative’ treatment,” she wrote.
Sometimes, misinformation is not sensational but is still effective via clever wording and presentation, observed Brian G. Southwell, PhD, of Duke University, Durham, N.C., who has studied patients and misinformation.
“It isn’t the falsehood that is somehow magically attractive, per se, but the way that misinformation is often framed that can make it attractive,” he said in an interview.
Dr. Southwell recommends that clinicians be proactive about medical misinformation.
“Rather than expect patients to raise concerns without prompting, health care providers should invite conversations about potential misinformation with their patients,” he wrote in a recent essay in the American Journal of Public Health.
In short, ask patients what they know about the treatment of their cancer, he suggests.
“Patients don’t typically know that the misinformation they are encountering is misinformation,” said Dr. Southwell. “Approaching patients with compassion and empathy is a good first step.”
Study details
For the study, reported by Johnson et al., two National Comprehensive Cancer Network panel members were selected as content experts for each of the four cancers and were tasked with reviewing the primary medical claims in each article. The experts then completed a set of ratings to arrive at the proportion of misinformation and potential for harm in each article.
Of the 200 articles, 41.5% were from nontraditional news (digital only), 37.5% were from traditional news sources (online versions of print and/or broadcast media), 17% were from medical journals, 3% were from a crowdfunding site, and 1% were from personal blogs.
This expert review concluded that nearly one-third of the articles contained misinformation, as noted above. The misinformation was described as misleading (title not supported by text or statistics/data do not support conclusion, 28.8%), strength of the evidence mischaracterized (weak evidence portrayed as strong or vice versa, 27.7%) and unproven therapies (not studied or insufficient evidence, 26.7%).
Notably, the median number of engagements, such as likes on Twitter, for articles with misinformation was greater than that of factual articles (median, 2,300 vs. 1,600; P = .05).
In total, 30.5% of all 200 articles contained harmful information. This was described as harmful inaction (could lead to delay or not seeking medical attention for treatable/curable condition, 31.0%), economic harm (out-of-pocket financial costs associated with treatment/travel, 27.7%), harmful action (potentially toxic effects of the suggested test/treatment, 17.0%), and harmful interactions (known/unknown medical interactions with curative therapies, 16.2%).
The median number of engagements for articles with harmful information was statistically significantly greater than that of articles with correct information (median, 2,300 vs. 1,500; P = .007).
A limitation of the study is that it included only the most popular English language cancer articles.
This study was funded in part by the Huntsman Cancer Institute. Dr. Johnson, Dr. Lycette, and Dr. Southwell have disclosed no relevant financial relationships. Some study authors have ties to the pharmaceutical industry.
A version of this article first appeared on Medscape.com.
FDA approves neoadjuvant pembro for triple-negative breast cancer
This approval is based on findings from the randomized, phase 3 KEYNOTE-522 trial, which showed significantly prolonged event-free survival with the pembrolizumab regimen versus neoadjuvant chemotherapy alone for previously untreated stage II-III TNBC.
This is the 30th indication for pembrolizumab in the United States.
The immunotherapy received accelerated approval in November 2020 for adjuvant use in locally recurrent unresectable or metastatic TNBC for patients whose tumors express programmed death–ligand-1, as determined by an FDA-approved test. That accelerated approval was based on results from the phase 3 KEYNOTE-355 trial. The approval has now been converted to a full approval on the basis of confirmatory data from the KEYNOTE-522, notes a statement from the manufacturer, Merck.
“Triple-negative is a difficult-to-treat type of breast cancer that unfortunately is more common in the U.S. in younger women and in Black women,” commented Vicki Goodman, MD, vice president of clinical research, Merck Research Laboratories. “We are proud to offer a new treatment option for patients faced with this challenging cancer. This neoadjuvant and adjuvant combination with pembrolizumab is the first immunotherapy regimen to be approved in high-risk early-stage TNBC, marking a meaningful milestone for the breast cancer community.”
In KEYNOTE-522, participants were randomly assigned to receive either placebo or pembrolizumab plus chemotherapy with carboplatin and paclitaxel, followed by doxorubicin or epirubicin and cyclophosphamide before surgery, as well as placebo or pembrolizumab as single-agent therapy after surgery.
The results from this trial, first reported in 2019 at the annual meeting of the European Society of Medical Oncology, showed that, for patients in the pembrolizumab arm of the trial, the pathological complete response rate was nearly 65% versus 51% among the patients who received placebo. The benefit was seen both in those whose tumors were positive and those whose tumors were negative for PD-L1 expression.
Among patients in the pembrolizumab arm, there was a 37% reduction in the risk for disease progression that precluded definitive surgery, a local/distant recurrence, a second primary cancer, or death from any cause (hazard ratio, 0.63).
Pembrolizumab can be associated with immune-mediated adverse reactions that may be severe or fatal, Merck noted.
These events “can occur in any organ system or tissue and can affect more than one body system simultaneously. Immune-mediated adverse reactions can occur at any time during or after treatment,” Merck warned. The company states: “Early identification and management of immune-mediated adverse reactions are essential.”
Treatment may need to be withheld or permanently discontinued, and corticosteroids may be needed, depending on the severity of the adverse reaction, according to the statement.
Infusion-related reactions can also occur. Because of its mechanism of action, pembrolizumab can cause fetal harm when administered to women during pregnancy.
A version of this article first appeared on Medscape.com.
This approval is based on findings from the randomized, phase 3 KEYNOTE-522 trial, which showed significantly prolonged event-free survival with the pembrolizumab regimen versus neoadjuvant chemotherapy alone for previously untreated stage II-III TNBC.
This is the 30th indication for pembrolizumab in the United States.
The immunotherapy received accelerated approval in November 2020 for adjuvant use in locally recurrent unresectable or metastatic TNBC for patients whose tumors express programmed death–ligand-1, as determined by an FDA-approved test. That accelerated approval was based on results from the phase 3 KEYNOTE-355 trial. The approval has now been converted to a full approval on the basis of confirmatory data from the KEYNOTE-522, notes a statement from the manufacturer, Merck.
“Triple-negative is a difficult-to-treat type of breast cancer that unfortunately is more common in the U.S. in younger women and in Black women,” commented Vicki Goodman, MD, vice president of clinical research, Merck Research Laboratories. “We are proud to offer a new treatment option for patients faced with this challenging cancer. This neoadjuvant and adjuvant combination with pembrolizumab is the first immunotherapy regimen to be approved in high-risk early-stage TNBC, marking a meaningful milestone for the breast cancer community.”
In KEYNOTE-522, participants were randomly assigned to receive either placebo or pembrolizumab plus chemotherapy with carboplatin and paclitaxel, followed by doxorubicin or epirubicin and cyclophosphamide before surgery, as well as placebo or pembrolizumab as single-agent therapy after surgery.
The results from this trial, first reported in 2019 at the annual meeting of the European Society of Medical Oncology, showed that, for patients in the pembrolizumab arm of the trial, the pathological complete response rate was nearly 65% versus 51% among the patients who received placebo. The benefit was seen both in those whose tumors were positive and those whose tumors were negative for PD-L1 expression.
Among patients in the pembrolizumab arm, there was a 37% reduction in the risk for disease progression that precluded definitive surgery, a local/distant recurrence, a second primary cancer, or death from any cause (hazard ratio, 0.63).
Pembrolizumab can be associated with immune-mediated adverse reactions that may be severe or fatal, Merck noted.
These events “can occur in any organ system or tissue and can affect more than one body system simultaneously. Immune-mediated adverse reactions can occur at any time during or after treatment,” Merck warned. The company states: “Early identification and management of immune-mediated adverse reactions are essential.”
Treatment may need to be withheld or permanently discontinued, and corticosteroids may be needed, depending on the severity of the adverse reaction, according to the statement.
Infusion-related reactions can also occur. Because of its mechanism of action, pembrolizumab can cause fetal harm when administered to women during pregnancy.
A version of this article first appeared on Medscape.com.
This approval is based on findings from the randomized, phase 3 KEYNOTE-522 trial, which showed significantly prolonged event-free survival with the pembrolizumab regimen versus neoadjuvant chemotherapy alone for previously untreated stage II-III TNBC.
This is the 30th indication for pembrolizumab in the United States.
The immunotherapy received accelerated approval in November 2020 for adjuvant use in locally recurrent unresectable or metastatic TNBC for patients whose tumors express programmed death–ligand-1, as determined by an FDA-approved test. That accelerated approval was based on results from the phase 3 KEYNOTE-355 trial. The approval has now been converted to a full approval on the basis of confirmatory data from the KEYNOTE-522, notes a statement from the manufacturer, Merck.
“Triple-negative is a difficult-to-treat type of breast cancer that unfortunately is more common in the U.S. in younger women and in Black women,” commented Vicki Goodman, MD, vice president of clinical research, Merck Research Laboratories. “We are proud to offer a new treatment option for patients faced with this challenging cancer. This neoadjuvant and adjuvant combination with pembrolizumab is the first immunotherapy regimen to be approved in high-risk early-stage TNBC, marking a meaningful milestone for the breast cancer community.”
In KEYNOTE-522, participants were randomly assigned to receive either placebo or pembrolizumab plus chemotherapy with carboplatin and paclitaxel, followed by doxorubicin or epirubicin and cyclophosphamide before surgery, as well as placebo or pembrolizumab as single-agent therapy after surgery.
The results from this trial, first reported in 2019 at the annual meeting of the European Society of Medical Oncology, showed that, for patients in the pembrolizumab arm of the trial, the pathological complete response rate was nearly 65% versus 51% among the patients who received placebo. The benefit was seen both in those whose tumors were positive and those whose tumors were negative for PD-L1 expression.
Among patients in the pembrolizumab arm, there was a 37% reduction in the risk for disease progression that precluded definitive surgery, a local/distant recurrence, a second primary cancer, or death from any cause (hazard ratio, 0.63).
Pembrolizumab can be associated with immune-mediated adverse reactions that may be severe or fatal, Merck noted.
These events “can occur in any organ system or tissue and can affect more than one body system simultaneously. Immune-mediated adverse reactions can occur at any time during or after treatment,” Merck warned. The company states: “Early identification and management of immune-mediated adverse reactions are essential.”
Treatment may need to be withheld or permanently discontinued, and corticosteroids may be needed, depending on the severity of the adverse reaction, according to the statement.
Infusion-related reactions can also occur. Because of its mechanism of action, pembrolizumab can cause fetal harm when administered to women during pregnancy.
A version of this article first appeared on Medscape.com.