User login
Marital status plays modest role in gastric cancer overall survival
, according to research published in the Journal of Investigative Medicine.
Tumor size remained the largest contributor to overall survival, but marital status was among several other significant factors, such as age, race, gender, treatment style, and pathologic stage, that can provide insight into a patient’s likelihood of overall survival, as it does with several other cancers.
“Married patients had the best prognosis, followed by single patients, and the prognosis of separated patients was the worst,” write Lixiang Zhang and colleagues at the First Affiliated Hospital of Anhui Medical University, Hefei, China. “We speculate that this might be due to the fact that married patients had better financial conditions and emotional encouragement, while separated patients might be more likely to experience financial difficulties [and] emotional loss.”
The results were not necessarily surprising to Richard M. Peek, Jr., MD, director of the division of gastroenterology and a professor of medicine at Vanderbilt University Medical Center, who was not involved in the research.
“Marital status is a reflection of support systems, and a strong support system is a prognosticator for increased compliance with medical appointments and medical therapies,” Dr. Peek told this news organization. “It is something to consider when somebody is being treated for gastric cancer, because if they don’t have a strong support system – and marital status can be a proxy for that – then they may need more intensive follow-up and surveillance, for example, than somebody who does not have that support system.”
Exploring the marital status–cancer survival connection
Gastric cancer is the third leading cause of cancer deaths across the world, causing 780,000 deaths in 2018, the authors note. Yet it’s difficult to accurately predict the prognosis in patients who undergo treatment for early stage gastric cancer. Previous research has found marital status to be associated with survival in prostate, cervical, and rectal cancers.
Mark A. Lewis, MD, director of gastrointestinal oncology at Intermountain Healthcare Cancer Center, Utah, told this news organization that the connection between marital status and cancer outcomes has been described previously, including in an even larger analysis using the U.S. Surveillance, Epidemiology, and End Results (SEER) database from 2013. That study found that “unmarried patients are at significantly higher risk of presentation with metastatic cancer, undertreatment, and death resulting from their cancer.”
In this study, the researchers compared marital status and survival rates among 3,647 patients with early-stage gastric cancer, using data from the SEER database. The study only included patients with tumors in the lamina propria, mucosa, and submucosa and excluded those with distant metastasis or distant lymph node metastases, a second cancer, no data on chemotherapy received, or unknown survival time.
Because they were using a nomogram and building a new predictive nomogram that would include marital status, the researchers divided the patient population into a training set of 2,719 patients and a testing set of 928 patients. Using overall survival as the primary endpoint, the analysis included the variables of “age at diagnosis, race, gender, tumor location, histology, grade, stage_T and stage_N, surgery in the primary site, lymph node dissection, chemotherapy, radiation, tumor size, insurance, and marital status,” the authors report.
Among the study population, 53.7% were married, 17.3% were widowed, 14% were single and never married, 7.5% were divorced, 1.1% were separated, and the status of 6.4% was unknown. Age at diagnosis, race, gender, histology, tumor grade, stage T, stage N, surgery type, tumor size, and insurance status were all significantly different between the marital status subgroups.
Married patients had the best prognosis, with an average overall survival of 72 months, compared with an average 60 months in widowed persons, the group with the poorest overall survival. Overall survival was higher in married women (76 months) than in married men (69 months). The same pattern held for women (62 months) and men (52 months) who had been widowed.
“It is worthy to note that survival was significantly better in divorced female patients than in divorced male patients,” the authors report. “Survival was better in female patients than in male patients” across all marital groups.
What long-term relationships reveal
These findings do not mean that simply getting married changes one’s likelihood of survival, however. Rather, a long-term relationship is revealing about other aspects in a person’s life.
“I think it represents more stability in the supportive relationship that you need to really deal with a serious disease like cancer,” Dr. Peek said.
If a patient does not have a long-term partner, their care team can ask other questions to get a sense of what their support network is like, Dr. Peek added. “We want to know, does anybody else live in the house with them? Do they have adequate transportation? Can they make medical appointments? Do they have somebody who can help with the medical issues that are going to come up? Do they have family in the area?”
Cancer treatment requires a multidisciplinary approach, and having someone other than just the patient around to help bring together the different aspects of care from different care teams can make a difference in how the patient fares, Dr. Peek explained. Patients without a strong support system may need closer follow-up and other accommodations, he said.
Providers “may schedule their clinical appointments closer together if they don’t have a support system, or they may be able to reach out and offer transportation assistance and those kinds of things that somebody living alone may need,” Dr. Peek said. Outside resources may be a higher priority for those who lack a support system at home, he added.
Dr. Peek also noted other factors that may play a role in a patient’s survival that these researchers did not have the data to address, such as socioeconomic status, employment, alcohol use, smoking, and infection with Helicobacter pylori, the strongest known risk factor for gastric cancer.
A potentially relevant limitation of the study is that it probably has some selection bias, because the patients who were included probably had the means to have received an earlier diagnosis, said Dr. Lewis, who was not involved in the research.
“Furthermore, just in terms of the group sizes, the baseline characteristics section makes it clear that the preponderance of patients were married, lending that group more statistical weight,” Dr. Lewis said.
“Of the seven attributes in the nomogram, the impact of the marital status seems comparatively meager relative to conventional clinicopathology risk factors like T stage,” he added.
“All in all, I think this study reinforces our awareness that socioeconomic status and social determinants of health play a huge role in cancer outcomes, but it’s not entirely clear that’s modifiable just by getting married,” Dr. Lewis said. “There is a saying in oncology that ‘expensive liquor causes less cancer than cheap liquor,’ which is not negating the carcinogenicity of alcohol but rather identifying different outcomes by socioeconomic status.”
The research was funded by the Natural Science Foundation of Anhui Province. The authors report no relevant financial relationships. Dr. Peek reports no relevant financial relationships. Dr. Lewis reports receiving speaking fees for AstraZeneca/Daiichi Sankyo and having done educational videos for Astellas.
A version of this article first appeared on Medscape.com.
, according to research published in the Journal of Investigative Medicine.
Tumor size remained the largest contributor to overall survival, but marital status was among several other significant factors, such as age, race, gender, treatment style, and pathologic stage, that can provide insight into a patient’s likelihood of overall survival, as it does with several other cancers.
“Married patients had the best prognosis, followed by single patients, and the prognosis of separated patients was the worst,” write Lixiang Zhang and colleagues at the First Affiliated Hospital of Anhui Medical University, Hefei, China. “We speculate that this might be due to the fact that married patients had better financial conditions and emotional encouragement, while separated patients might be more likely to experience financial difficulties [and] emotional loss.”
The results were not necessarily surprising to Richard M. Peek, Jr., MD, director of the division of gastroenterology and a professor of medicine at Vanderbilt University Medical Center, who was not involved in the research.
“Marital status is a reflection of support systems, and a strong support system is a prognosticator for increased compliance with medical appointments and medical therapies,” Dr. Peek told this news organization. “It is something to consider when somebody is being treated for gastric cancer, because if they don’t have a strong support system – and marital status can be a proxy for that – then they may need more intensive follow-up and surveillance, for example, than somebody who does not have that support system.”
Exploring the marital status–cancer survival connection
Gastric cancer is the third leading cause of cancer deaths across the world, causing 780,000 deaths in 2018, the authors note. Yet it’s difficult to accurately predict the prognosis in patients who undergo treatment for early stage gastric cancer. Previous research has found marital status to be associated with survival in prostate, cervical, and rectal cancers.
Mark A. Lewis, MD, director of gastrointestinal oncology at Intermountain Healthcare Cancer Center, Utah, told this news organization that the connection between marital status and cancer outcomes has been described previously, including in an even larger analysis using the U.S. Surveillance, Epidemiology, and End Results (SEER) database from 2013. That study found that “unmarried patients are at significantly higher risk of presentation with metastatic cancer, undertreatment, and death resulting from their cancer.”
In this study, the researchers compared marital status and survival rates among 3,647 patients with early-stage gastric cancer, using data from the SEER database. The study only included patients with tumors in the lamina propria, mucosa, and submucosa and excluded those with distant metastasis or distant lymph node metastases, a second cancer, no data on chemotherapy received, or unknown survival time.
Because they were using a nomogram and building a new predictive nomogram that would include marital status, the researchers divided the patient population into a training set of 2,719 patients and a testing set of 928 patients. Using overall survival as the primary endpoint, the analysis included the variables of “age at diagnosis, race, gender, tumor location, histology, grade, stage_T and stage_N, surgery in the primary site, lymph node dissection, chemotherapy, radiation, tumor size, insurance, and marital status,” the authors report.
Among the study population, 53.7% were married, 17.3% were widowed, 14% were single and never married, 7.5% were divorced, 1.1% were separated, and the status of 6.4% was unknown. Age at diagnosis, race, gender, histology, tumor grade, stage T, stage N, surgery type, tumor size, and insurance status were all significantly different between the marital status subgroups.
Married patients had the best prognosis, with an average overall survival of 72 months, compared with an average 60 months in widowed persons, the group with the poorest overall survival. Overall survival was higher in married women (76 months) than in married men (69 months). The same pattern held for women (62 months) and men (52 months) who had been widowed.
“It is worthy to note that survival was significantly better in divorced female patients than in divorced male patients,” the authors report. “Survival was better in female patients than in male patients” across all marital groups.
What long-term relationships reveal
These findings do not mean that simply getting married changes one’s likelihood of survival, however. Rather, a long-term relationship is revealing about other aspects in a person’s life.
“I think it represents more stability in the supportive relationship that you need to really deal with a serious disease like cancer,” Dr. Peek said.
If a patient does not have a long-term partner, their care team can ask other questions to get a sense of what their support network is like, Dr. Peek added. “We want to know, does anybody else live in the house with them? Do they have adequate transportation? Can they make medical appointments? Do they have somebody who can help with the medical issues that are going to come up? Do they have family in the area?”
Cancer treatment requires a multidisciplinary approach, and having someone other than just the patient around to help bring together the different aspects of care from different care teams can make a difference in how the patient fares, Dr. Peek explained. Patients without a strong support system may need closer follow-up and other accommodations, he said.
Providers “may schedule their clinical appointments closer together if they don’t have a support system, or they may be able to reach out and offer transportation assistance and those kinds of things that somebody living alone may need,” Dr. Peek said. Outside resources may be a higher priority for those who lack a support system at home, he added.
Dr. Peek also noted other factors that may play a role in a patient’s survival that these researchers did not have the data to address, such as socioeconomic status, employment, alcohol use, smoking, and infection with Helicobacter pylori, the strongest known risk factor for gastric cancer.
A potentially relevant limitation of the study is that it probably has some selection bias, because the patients who were included probably had the means to have received an earlier diagnosis, said Dr. Lewis, who was not involved in the research.
“Furthermore, just in terms of the group sizes, the baseline characteristics section makes it clear that the preponderance of patients were married, lending that group more statistical weight,” Dr. Lewis said.
“Of the seven attributes in the nomogram, the impact of the marital status seems comparatively meager relative to conventional clinicopathology risk factors like T stage,” he added.
“All in all, I think this study reinforces our awareness that socioeconomic status and social determinants of health play a huge role in cancer outcomes, but it’s not entirely clear that’s modifiable just by getting married,” Dr. Lewis said. “There is a saying in oncology that ‘expensive liquor causes less cancer than cheap liquor,’ which is not negating the carcinogenicity of alcohol but rather identifying different outcomes by socioeconomic status.”
The research was funded by the Natural Science Foundation of Anhui Province. The authors report no relevant financial relationships. Dr. Peek reports no relevant financial relationships. Dr. Lewis reports receiving speaking fees for AstraZeneca/Daiichi Sankyo and having done educational videos for Astellas.
A version of this article first appeared on Medscape.com.
, according to research published in the Journal of Investigative Medicine.
Tumor size remained the largest contributor to overall survival, but marital status was among several other significant factors, such as age, race, gender, treatment style, and pathologic stage, that can provide insight into a patient’s likelihood of overall survival, as it does with several other cancers.
“Married patients had the best prognosis, followed by single patients, and the prognosis of separated patients was the worst,” write Lixiang Zhang and colleagues at the First Affiliated Hospital of Anhui Medical University, Hefei, China. “We speculate that this might be due to the fact that married patients had better financial conditions and emotional encouragement, while separated patients might be more likely to experience financial difficulties [and] emotional loss.”
The results were not necessarily surprising to Richard M. Peek, Jr., MD, director of the division of gastroenterology and a professor of medicine at Vanderbilt University Medical Center, who was not involved in the research.
“Marital status is a reflection of support systems, and a strong support system is a prognosticator for increased compliance with medical appointments and medical therapies,” Dr. Peek told this news organization. “It is something to consider when somebody is being treated for gastric cancer, because if they don’t have a strong support system – and marital status can be a proxy for that – then they may need more intensive follow-up and surveillance, for example, than somebody who does not have that support system.”
Exploring the marital status–cancer survival connection
Gastric cancer is the third leading cause of cancer deaths across the world, causing 780,000 deaths in 2018, the authors note. Yet it’s difficult to accurately predict the prognosis in patients who undergo treatment for early stage gastric cancer. Previous research has found marital status to be associated with survival in prostate, cervical, and rectal cancers.
Mark A. Lewis, MD, director of gastrointestinal oncology at Intermountain Healthcare Cancer Center, Utah, told this news organization that the connection between marital status and cancer outcomes has been described previously, including in an even larger analysis using the U.S. Surveillance, Epidemiology, and End Results (SEER) database from 2013. That study found that “unmarried patients are at significantly higher risk of presentation with metastatic cancer, undertreatment, and death resulting from their cancer.”
In this study, the researchers compared marital status and survival rates among 3,647 patients with early-stage gastric cancer, using data from the SEER database. The study only included patients with tumors in the lamina propria, mucosa, and submucosa and excluded those with distant metastasis or distant lymph node metastases, a second cancer, no data on chemotherapy received, or unknown survival time.
Because they were using a nomogram and building a new predictive nomogram that would include marital status, the researchers divided the patient population into a training set of 2,719 patients and a testing set of 928 patients. Using overall survival as the primary endpoint, the analysis included the variables of “age at diagnosis, race, gender, tumor location, histology, grade, stage_T and stage_N, surgery in the primary site, lymph node dissection, chemotherapy, radiation, tumor size, insurance, and marital status,” the authors report.
Among the study population, 53.7% were married, 17.3% were widowed, 14% were single and never married, 7.5% were divorced, 1.1% were separated, and the status of 6.4% was unknown. Age at diagnosis, race, gender, histology, tumor grade, stage T, stage N, surgery type, tumor size, and insurance status were all significantly different between the marital status subgroups.
Married patients had the best prognosis, with an average overall survival of 72 months, compared with an average 60 months in widowed persons, the group with the poorest overall survival. Overall survival was higher in married women (76 months) than in married men (69 months). The same pattern held for women (62 months) and men (52 months) who had been widowed.
“It is worthy to note that survival was significantly better in divorced female patients than in divorced male patients,” the authors report. “Survival was better in female patients than in male patients” across all marital groups.
What long-term relationships reveal
These findings do not mean that simply getting married changes one’s likelihood of survival, however. Rather, a long-term relationship is revealing about other aspects in a person’s life.
“I think it represents more stability in the supportive relationship that you need to really deal with a serious disease like cancer,” Dr. Peek said.
If a patient does not have a long-term partner, their care team can ask other questions to get a sense of what their support network is like, Dr. Peek added. “We want to know, does anybody else live in the house with them? Do they have adequate transportation? Can they make medical appointments? Do they have somebody who can help with the medical issues that are going to come up? Do they have family in the area?”
Cancer treatment requires a multidisciplinary approach, and having someone other than just the patient around to help bring together the different aspects of care from different care teams can make a difference in how the patient fares, Dr. Peek explained. Patients without a strong support system may need closer follow-up and other accommodations, he said.
Providers “may schedule their clinical appointments closer together if they don’t have a support system, or they may be able to reach out and offer transportation assistance and those kinds of things that somebody living alone may need,” Dr. Peek said. Outside resources may be a higher priority for those who lack a support system at home, he added.
Dr. Peek also noted other factors that may play a role in a patient’s survival that these researchers did not have the data to address, such as socioeconomic status, employment, alcohol use, smoking, and infection with Helicobacter pylori, the strongest known risk factor for gastric cancer.
A potentially relevant limitation of the study is that it probably has some selection bias, because the patients who were included probably had the means to have received an earlier diagnosis, said Dr. Lewis, who was not involved in the research.
“Furthermore, just in terms of the group sizes, the baseline characteristics section makes it clear that the preponderance of patients were married, lending that group more statistical weight,” Dr. Lewis said.
“Of the seven attributes in the nomogram, the impact of the marital status seems comparatively meager relative to conventional clinicopathology risk factors like T stage,” he added.
“All in all, I think this study reinforces our awareness that socioeconomic status and social determinants of health play a huge role in cancer outcomes, but it’s not entirely clear that’s modifiable just by getting married,” Dr. Lewis said. “There is a saying in oncology that ‘expensive liquor causes less cancer than cheap liquor,’ which is not negating the carcinogenicity of alcohol but rather identifying different outcomes by socioeconomic status.”
The research was funded by the Natural Science Foundation of Anhui Province. The authors report no relevant financial relationships. Dr. Peek reports no relevant financial relationships. Dr. Lewis reports receiving speaking fees for AstraZeneca/Daiichi Sankyo and having done educational videos for Astellas.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF INVESTIGATIVE MEDICINE
CRC screening disparities greatest among those under 55
Adults younger than 55 years were least likely to get screened for colorectal cancer over the past 2 decades, particularly if they were Hispanic or Asian or had a low income, lower education level, or no health insurance, according to a new study published online in Cancer Epidemiology, Biomarkers & Prevention.
The findings have raised concerns that disparities in screening rates will be even greater in adults aged 45-49 years, prompting the need for increased awareness and outreach to ensure that underserved groups have access to screenings.
“Differences in prevalence of screening by race and ethnicity, educational attainment, household income, and health insurance were most pronounced for those ages 50-54 years, whereas older adults experienced larger increases in prevalence across these groups,” wrote Po-Hong Liu, MD, MPH, a clinical investigator at Harvard University, Boston, and his colleagues. “The persistent and worsening disparities we observed in adults 50-54 years may extend to those ages 45-49 as they become eligible for screening.”
The U.S. Preventive Services Task Force shifted their recommendation for colorectal cancer screening in May 2021 to 5 years earlier, advising people to start screenings at 45 instead of 50, which aligns with the recommendations the American Cancer Society made 3 years earlier.
Both organizations made the change because of increasing rates of colorectal cancer in adults under age 50 and research indicating that beginning screenings at age 45 results in fewer cases, fewer deaths, and more life years gained.
“Across all age groups, colorectal cancer screening participation remains below national goals, and the benefits of screening are not equally realized across populations,” senior author Caitlin Murphy, PhD, MPH, associate professor, UTHealth School of Public Health, Houston, said in a prepared statement. “Extra care must be taken to ensure that expanding screening to younger ages does not negatively impact efforts to eliminate disparities in colorectal screening and outcomes nor jeopardize efforts to increase screening initiation among older adults who remain unscreened.”
Data analyzed from 8 years over 2 decades
The researchers analyzed data from the CDC’s cross-sectional National Health Interview Survey during 8 years over the past 2 decades: 2000, 2003, 2005, 2008, 2010, 2013, 2015, and 2018.
The number of participants each year ranged from a low of 21,781 in 2008 to a high of 34,557 in 2013. After excluding participants with a history of colorectal cancer or missing information on screenings, the total population sample included 80,220 participants 50-75 years old.
The researchers considered a person as having been screened if they received at least one recommended screening test within the year covered by the survey, regardless of why they underwent the test.
Recommended tests included sigmoidoscopy, colonoscopy, and stool-based tests for all survey years. In addition, the surveys for 2010, 2015, and 2018 included CT colonography, and the 2018 survey included FIT-DNA.
Screening across population groups
Colorectal cancer screening rates have doubled in the past 2 decades, from 36.7% in 2000 to 66.1% in 2018.
Rates are considerably lower, however, for several key groups, including the youngest group. Less than half (47.6%) of those aged 50-54 years received screenings in 2018, though this was still a nearly 20-point improvement over the 28.2% in this age group who were screened in 2000.
Separate from age, several other groups continue to have low screening rates in general, including Hispanics (56.5%, up from 25.9% in 2000), Asians (57.1%, up from 22.6% in 2000), those who have not received a high school degree (53.6%, up from 26.8% in 2000), and those from low income families (56.6%, up from 30.2% in 2000).
The group with the greatest need for more outreach and screenings are people without insurance, only 39.7% of whom were screened in 2018, a modest increase from 30.2% in 2000.
The biggest increase in screenings over time occurred in those aged 70-75 years, from 46.4% in 2000 to 78% in 2018 overall.
Racial/ethnic, economic, education, and insurance-based disparities were particularly evident the younger people were, including in terms of progress made over time.
For example, screenings of non-Hispanic White people aged 50-54 years improved 21 points (30.3% to 51%) between 2000 and 2018, compared with 19 points in Hispanics (16.7% to 35.5%) and 15 points in Asians (17.3% to 32.3%). Fortunately, Black Americans made even greater strides than White Americans with a 27-point increase during that time (23.4% to 50%).
Similarly, income correlated with expansion in screening rates for 50- to 54-year-olds: Those earning at least 400% over the federal poverty line improved 20 points (from 33.5% to 53.8%), compared with a 16-point improvement in those earning less than 200% above the poverty line (from 19.3% to 35%).
Those with private insurance likewise improved 21 points (from 30.7% to 51.7%), while those in this age group without insurance declined, with just 21.2% getting screened in 2018, compared with 28.2% in 2000. Those on public insurance saw a 15-point improvement, from 27.8% in 2000 to 43.1% in 2018.
“The individual and societal burden of colorectal cancer is especially great among younger adults,” the authors wrote.
The reasons for the much lower prevalence of screening in those under 55, the authors suggested, is likely due to less concern about colorectal cancer, less access to medical care (including being underinsured or uninsured), and the barriers created by competing priorities, such as work schedules, family responsibilities, and caregiving. The latter may be particularly true in underserved populations, the authors noted.
“Screening programs must consider the barriers unique to younger adults, ensuring the benefits of screening are equally realized by all population groups,” the authors concluded.
The research was funded by the National Institutes of Health and the Cancer Prevention and Research Institute of Texas. One author reported grants from Epigenomics and Freenome and personal fees from Guardant Health. Another author reported personal fees from Freenome, and a third author reported personal fees from Exact Sciences. No other authors had industry disclosures.
A version of this article first appeared on Medscape.com.
Adults younger than 55 years were least likely to get screened for colorectal cancer over the past 2 decades, particularly if they were Hispanic or Asian or had a low income, lower education level, or no health insurance, according to a new study published online in Cancer Epidemiology, Biomarkers & Prevention.
The findings have raised concerns that disparities in screening rates will be even greater in adults aged 45-49 years, prompting the need for increased awareness and outreach to ensure that underserved groups have access to screenings.
“Differences in prevalence of screening by race and ethnicity, educational attainment, household income, and health insurance were most pronounced for those ages 50-54 years, whereas older adults experienced larger increases in prevalence across these groups,” wrote Po-Hong Liu, MD, MPH, a clinical investigator at Harvard University, Boston, and his colleagues. “The persistent and worsening disparities we observed in adults 50-54 years may extend to those ages 45-49 as they become eligible for screening.”
The U.S. Preventive Services Task Force shifted their recommendation for colorectal cancer screening in May 2021 to 5 years earlier, advising people to start screenings at 45 instead of 50, which aligns with the recommendations the American Cancer Society made 3 years earlier.
Both organizations made the change because of increasing rates of colorectal cancer in adults under age 50 and research indicating that beginning screenings at age 45 results in fewer cases, fewer deaths, and more life years gained.
“Across all age groups, colorectal cancer screening participation remains below national goals, and the benefits of screening are not equally realized across populations,” senior author Caitlin Murphy, PhD, MPH, associate professor, UTHealth School of Public Health, Houston, said in a prepared statement. “Extra care must be taken to ensure that expanding screening to younger ages does not negatively impact efforts to eliminate disparities in colorectal screening and outcomes nor jeopardize efforts to increase screening initiation among older adults who remain unscreened.”
Data analyzed from 8 years over 2 decades
The researchers analyzed data from the CDC’s cross-sectional National Health Interview Survey during 8 years over the past 2 decades: 2000, 2003, 2005, 2008, 2010, 2013, 2015, and 2018.
The number of participants each year ranged from a low of 21,781 in 2008 to a high of 34,557 in 2013. After excluding participants with a history of colorectal cancer or missing information on screenings, the total population sample included 80,220 participants 50-75 years old.
The researchers considered a person as having been screened if they received at least one recommended screening test within the year covered by the survey, regardless of why they underwent the test.
Recommended tests included sigmoidoscopy, colonoscopy, and stool-based tests for all survey years. In addition, the surveys for 2010, 2015, and 2018 included CT colonography, and the 2018 survey included FIT-DNA.
Screening across population groups
Colorectal cancer screening rates have doubled in the past 2 decades, from 36.7% in 2000 to 66.1% in 2018.
Rates are considerably lower, however, for several key groups, including the youngest group. Less than half (47.6%) of those aged 50-54 years received screenings in 2018, though this was still a nearly 20-point improvement over the 28.2% in this age group who were screened in 2000.
Separate from age, several other groups continue to have low screening rates in general, including Hispanics (56.5%, up from 25.9% in 2000), Asians (57.1%, up from 22.6% in 2000), those who have not received a high school degree (53.6%, up from 26.8% in 2000), and those from low income families (56.6%, up from 30.2% in 2000).
The group with the greatest need for more outreach and screenings are people without insurance, only 39.7% of whom were screened in 2018, a modest increase from 30.2% in 2000.
The biggest increase in screenings over time occurred in those aged 70-75 years, from 46.4% in 2000 to 78% in 2018 overall.
Racial/ethnic, economic, education, and insurance-based disparities were particularly evident the younger people were, including in terms of progress made over time.
For example, screenings of non-Hispanic White people aged 50-54 years improved 21 points (30.3% to 51%) between 2000 and 2018, compared with 19 points in Hispanics (16.7% to 35.5%) and 15 points in Asians (17.3% to 32.3%). Fortunately, Black Americans made even greater strides than White Americans with a 27-point increase during that time (23.4% to 50%).
Similarly, income correlated with expansion in screening rates for 50- to 54-year-olds: Those earning at least 400% over the federal poverty line improved 20 points (from 33.5% to 53.8%), compared with a 16-point improvement in those earning less than 200% above the poverty line (from 19.3% to 35%).
Those with private insurance likewise improved 21 points (from 30.7% to 51.7%), while those in this age group without insurance declined, with just 21.2% getting screened in 2018, compared with 28.2% in 2000. Those on public insurance saw a 15-point improvement, from 27.8% in 2000 to 43.1% in 2018.
“The individual and societal burden of colorectal cancer is especially great among younger adults,” the authors wrote.
The reasons for the much lower prevalence of screening in those under 55, the authors suggested, is likely due to less concern about colorectal cancer, less access to medical care (including being underinsured or uninsured), and the barriers created by competing priorities, such as work schedules, family responsibilities, and caregiving. The latter may be particularly true in underserved populations, the authors noted.
“Screening programs must consider the barriers unique to younger adults, ensuring the benefits of screening are equally realized by all population groups,” the authors concluded.
The research was funded by the National Institutes of Health and the Cancer Prevention and Research Institute of Texas. One author reported grants from Epigenomics and Freenome and personal fees from Guardant Health. Another author reported personal fees from Freenome, and a third author reported personal fees from Exact Sciences. No other authors had industry disclosures.
A version of this article first appeared on Medscape.com.
Adults younger than 55 years were least likely to get screened for colorectal cancer over the past 2 decades, particularly if they were Hispanic or Asian or had a low income, lower education level, or no health insurance, according to a new study published online in Cancer Epidemiology, Biomarkers & Prevention.
The findings have raised concerns that disparities in screening rates will be even greater in adults aged 45-49 years, prompting the need for increased awareness and outreach to ensure that underserved groups have access to screenings.
“Differences in prevalence of screening by race and ethnicity, educational attainment, household income, and health insurance were most pronounced for those ages 50-54 years, whereas older adults experienced larger increases in prevalence across these groups,” wrote Po-Hong Liu, MD, MPH, a clinical investigator at Harvard University, Boston, and his colleagues. “The persistent and worsening disparities we observed in adults 50-54 years may extend to those ages 45-49 as they become eligible for screening.”
The U.S. Preventive Services Task Force shifted their recommendation for colorectal cancer screening in May 2021 to 5 years earlier, advising people to start screenings at 45 instead of 50, which aligns with the recommendations the American Cancer Society made 3 years earlier.
Both organizations made the change because of increasing rates of colorectal cancer in adults under age 50 and research indicating that beginning screenings at age 45 results in fewer cases, fewer deaths, and more life years gained.
“Across all age groups, colorectal cancer screening participation remains below national goals, and the benefits of screening are not equally realized across populations,” senior author Caitlin Murphy, PhD, MPH, associate professor, UTHealth School of Public Health, Houston, said in a prepared statement. “Extra care must be taken to ensure that expanding screening to younger ages does not negatively impact efforts to eliminate disparities in colorectal screening and outcomes nor jeopardize efforts to increase screening initiation among older adults who remain unscreened.”
Data analyzed from 8 years over 2 decades
The researchers analyzed data from the CDC’s cross-sectional National Health Interview Survey during 8 years over the past 2 decades: 2000, 2003, 2005, 2008, 2010, 2013, 2015, and 2018.
The number of participants each year ranged from a low of 21,781 in 2008 to a high of 34,557 in 2013. After excluding participants with a history of colorectal cancer or missing information on screenings, the total population sample included 80,220 participants 50-75 years old.
The researchers considered a person as having been screened if they received at least one recommended screening test within the year covered by the survey, regardless of why they underwent the test.
Recommended tests included sigmoidoscopy, colonoscopy, and stool-based tests for all survey years. In addition, the surveys for 2010, 2015, and 2018 included CT colonography, and the 2018 survey included FIT-DNA.
Screening across population groups
Colorectal cancer screening rates have doubled in the past 2 decades, from 36.7% in 2000 to 66.1% in 2018.
Rates are considerably lower, however, for several key groups, including the youngest group. Less than half (47.6%) of those aged 50-54 years received screenings in 2018, though this was still a nearly 20-point improvement over the 28.2% in this age group who were screened in 2000.
Separate from age, several other groups continue to have low screening rates in general, including Hispanics (56.5%, up from 25.9% in 2000), Asians (57.1%, up from 22.6% in 2000), those who have not received a high school degree (53.6%, up from 26.8% in 2000), and those from low income families (56.6%, up from 30.2% in 2000).
The group with the greatest need for more outreach and screenings are people without insurance, only 39.7% of whom were screened in 2018, a modest increase from 30.2% in 2000.
The biggest increase in screenings over time occurred in those aged 70-75 years, from 46.4% in 2000 to 78% in 2018 overall.
Racial/ethnic, economic, education, and insurance-based disparities were particularly evident the younger people were, including in terms of progress made over time.
For example, screenings of non-Hispanic White people aged 50-54 years improved 21 points (30.3% to 51%) between 2000 and 2018, compared with 19 points in Hispanics (16.7% to 35.5%) and 15 points in Asians (17.3% to 32.3%). Fortunately, Black Americans made even greater strides than White Americans with a 27-point increase during that time (23.4% to 50%).
Similarly, income correlated with expansion in screening rates for 50- to 54-year-olds: Those earning at least 400% over the federal poverty line improved 20 points (from 33.5% to 53.8%), compared with a 16-point improvement in those earning less than 200% above the poverty line (from 19.3% to 35%).
Those with private insurance likewise improved 21 points (from 30.7% to 51.7%), while those in this age group without insurance declined, with just 21.2% getting screened in 2018, compared with 28.2% in 2000. Those on public insurance saw a 15-point improvement, from 27.8% in 2000 to 43.1% in 2018.
“The individual and societal burden of colorectal cancer is especially great among younger adults,” the authors wrote.
The reasons for the much lower prevalence of screening in those under 55, the authors suggested, is likely due to less concern about colorectal cancer, less access to medical care (including being underinsured or uninsured), and the barriers created by competing priorities, such as work schedules, family responsibilities, and caregiving. The latter may be particularly true in underserved populations, the authors noted.
“Screening programs must consider the barriers unique to younger adults, ensuring the benefits of screening are equally realized by all population groups,” the authors concluded.
The research was funded by the National Institutes of Health and the Cancer Prevention and Research Institute of Texas. One author reported grants from Epigenomics and Freenome and personal fees from Guardant Health. Another author reported personal fees from Freenome, and a third author reported personal fees from Exact Sciences. No other authors had industry disclosures.
A version of this article first appeared on Medscape.com.
FROM CANCER EPIDEMIOLOGY, BIOMARKERS AN PREVENTION
Roe reversal may go well beyond abortion
Kami, a mother of one daughter in central Texas, lost three pregnancies in 2008. The third one nearly killed her.
The embryo became implanted in one of the fallopian tubes connecting her ovaries to her uterus. Because fallopian tubes can’t stretch to accommodate a fetus, patients must undergo surgery to remove the embryo before the tube ruptures. Failure to do so can result in internal bleeding and death.
But when Kami – who did not want to use her last name to avoid harassment – underwent an ultrasound to start the process of extracting the embryo, her doctor miscalculated how far along in the pregnancy she was and told her to come back in a few weeks.
She eventually did return, but only after passing out in the bathtub and waking up in a pool of her own blood. The tube had ruptured, and to remove it, emergency surgery was necessary.
Stories such as Kami’s could become more common in the aftermath of the U.S. Supreme Court’s decision to overturn Roe v. Wade, the 1973 case that created a right to an abortion.
Experts fear that antiabortion laws that take effect in the United States following the court’s decision will lead to a medical and legal limbo for thousands of people like Kami – people with uncommon reproductive conditions whose treatments involve the termination of pregnancies or the destruction of embryos.
Vague exceptions prompt concerns
According to the Guttmacher Institute, a nonprofit group for reproductive health, 13 states currently have trigger laws on the books that make abortion illegal in the absence of Roe. Nine other states have laws that would outlaw or severely restrict abortion without a federal right to the procedure.
Each of these laws carves out exceptions that allow the termination of a pregnancy to prevent the death of the pregnant individual. But the language of the provisions is not always precise in describing what those exceptions mean in practice, according to Elizabeth Nash, the principal policy associate for state issues at the Guttmacher Institute.
“These exceptions are designed to be extraordinarily narrow. These aren’t really designed to be usable exceptions,” Ms. Nash said. “There’s so much misinformation about abortion that there are probably legislators out there who think that it’s never needed to save a life.”
Tubal pregnancies
One of the best examples of a pregnancy termination that’s necessary to avoid death is in the case of an ectopic pregnancy such as Kami experienced. Without treatment to end the pregnancy, the embryo will eventually grow so large that the tube ruptures, causing massive bleeding that can kill the mother.
Most state laws regarding abortion exclude treatment of ectopic pregnancy, according to Ms. Nash. But, “if the state does not exclude ectopic pregnancy from all the regulations, then people might not be able to get the care that they need when they need it.”
The current abortion law in Texas, for example, prohibits ending a pregnancy after 6 weeks, or after cardiac activity becomes present. Cardiac activity can be present in cases of ectopic pregnancies, which account for between 1% and 2% of all pregnancies and are the leading cause of maternal deaths in the first trimester. And treatment definitely ends the life of the embryo or fetus in the fallopian tube, said Lisa Harris, MD, PhD, an ob.gyn. and medical ethicist at the University of Michigan, Ann Arbor.
Dr. Harris said she has never doubted that an ectopic pregnancy cannot possibly result in a live birth. But she recalled an encounter with another clinician on a surgical team for an ectopic pregnancy who said: “So you’re going to take it out of the tube and put it in the uterus, right?”
“It was a startling moment,” Dr. Harris recalled. She regarded the procedure as a “lifesaving, obvious surgery,” but her colleague, whose suggestion was a medical impossibility since the window of implantation is very brief after fertilization, viewed it “as an abortion, as killing an embryo or fetus.”
Dr. Harris said she isn’t concerned that physicians would stop treating ectopic pregnancies in a post-Roe world. Rather, she worries about two other possibilities: an overzealous prosecutor might not believe it was an ectopic pregnancy and press charges; or laws will cause physicians to second guess their clinical decisions for patients.
“What it means, in the middle of the night, when someone comes in with a 10-week ectopic pregnancy with a heartbeat, is the doctor may hesitate,” Dr. Harris said. Despite knowing the appropriate treatment, the doctor may want to speak with a lawyer or ethicist first to ensure they are covered legally. “And as that process unfolds, which could take hours or days, the person might have a complication.” s
Not treating an ectopic pregnancy would be malpractice, but “some doctors may not provide the standard of care that they would have ordinarily provided because they don’t want to risk breaking the law,” she said.
Even more ambiguous are cornual ectopic pregnancies, in which the implantation occurs at the junction of a fallopian tube and the uterus. These pregnancies, which make up 2%-4% of all tubal pregnancies, are immediately adjacent to the uterus. If an abortion is defined as the termination of an embryo or fetus in the womb, how such a legal definition would apply to these pregnancies is unclear.
An ob.gyn. wouldn’t regard ending an ectopic pregnancy as an abortion, but “this is not about logic or clinical meaning,” Dr. Harris said. “This is people outside of medicine making determinations that all pregnancies must continue, and when you think of a ban that way, you could see why a doctor would be frightened to end a pregnancy, whether it might be viable in the future or not.”
That’s true even if the pregnancy is located fully in the uterus. Dr. Harris described a pregnant patient she saw who had traveled from Texas to Michigan with a fetus that had a lethal defect.
The fetus had “an anomaly where the lungs couldn’t develop, where there were no kidneys. There was no chance this baby could be born and live. Her doctors were very clear that there will never be a baby that [she could] take home at the end of this pregnancy, yet they would not end her pregnancy because that would be an abortion,” Dr. Harris said.
Texas law “doesn’t make any allowances for whether a pregnancy will ever actually result in a baby or not,” Dr. Harris said. “The law, in effect, just says all pregnancies must continue.”
Selective reduction
How abortion laws in different states might affect selective reduction, which is used in some pregnancies to reduce the total number of fetuses a person is carrying, is even more ambiguous. The goal of selective reduction is to decrease health risks to the pregnant individual and increase the likelihood of survival for the remaining fetuses. Current Texas law prohibits these procedures.
Someone pregnant with quintuplets, for example, might seek selection reduction to reduce the pregnancy outcome to triplets or twins. A related procedure, selective termination, is used to terminate the life of a fetus with abnormalities while the pregnancy of the fetus’ in utero siblings continues.
The advent of assisted reproduction methods, such as in vitro fertilization (IVF), greatly increased the incidence of higher-order multiples, those with three or more fetuses. The first IVF baby was born in 1978. By 1998, the rate of higher-order multiple births was 1.9 per 1,000 births, five times the figure in 1980. The rate has since decreased by nearly half, to 1 per 1,000 births, but with 3.75 million live births a year, that’s still a lot of pregnancies with higher-order multiples.
The American College of Obstetricians and Gynecologists does not provide explicit guidance on when selective reduction is warranted, but its committee opinion on multifetal pregnancy reduction provides an ethical framework for providers to use when counseling people with pregnancies of three or more fetuses. How would various state laws that outlaw abortion affect these decisions? No one knows.
“Selective reduction ends the life of a fetus or embryo, but it doesn’t end the pregnancy,” Dr. Harris said. “So, if the pregnancy continues but it kills an embryo or fetus, is that an abortion?”
‘The question of the hour’
Dr. Harris and other doctors are haunted by potential medical cases in which continuing a pregnancy may result in the death of the person carrying the fetus but in which such death may not be so imminent that the law would allow immediate termination of the pregnancy.
Michael Northrup, MD, an intensive care pediatrician in Winston-Salem, N.C., recalled a particularly harrowing case that illustrates the peril in deciding when someone’s life is “enough” in danger to qualify as an exception to abortion bans.
The 14-year-old girl had severe lupus and kidney failure that required treatment with methotrexate and immediate dialysis to replace her electrolytes. A standard pretreatment pregnancy test revealed that she had been carrying a child for at least 10 weeks. Her pregnancy presented two problems. Methotrexate is so severely toxic that it’s sometimes used to end pregnancies. Even at low doses, fetuses that survive usually have severe deformities. In addition, dialysis requires administration of a blood thinner. If the teen miscarried while taking a blood thinner during dialysis, she risked bleeding to death.
Treatment could be delayed until week 24 of pregnancy, at which time delivery could be attempted, but the patient likely wouldn’t have any kidney function left by then. In addition, at 24 weeks, it was unlikely that the baby would survive anyway.
Dr. Northrup said that, had she chosen that route, “I’m not sure she would have made it. This was a religious family, people who very much were believers. They had their head of church come in, who fairly quickly determined that the best thing for her health was to terminate this pregnancy immediately and get the treatment she needed for her body.”
Would such a situation qualify for an emergency termination? The girl wasn’t going to die within 24 or 48 hours, but it may not have been possible to pinpoint the time of death within a day or 2.
“The family was sad, but they made that choice, and I wonder, would we have to justify that with these new laws?” Dr. Northrup said. “You definitely worry, being in the hot seat, ‘Does this count enough? Is she close enough to death?’ ”
The same question comes up when someone’s water breaks early in the second trimester. Since a live-birth delivery would be highly unlikely, given the age of the fetus, the standard of care is to offer to terminate the pregnancy to avoid a serious infection, Dr. Harris said. But if the infection hasn’t yet developed – even if it’s likely to develop soon – doctors in a state that outlaws abortion would not be able to offer termination. But as providers wait for an infection to develop, the person’s risk of dying from infection rapidly increases.
“How likely does someone need to be to die for it to count to get a life-preserving abortion?” Dr. Harris asked. “That, I think, is the question of the hour.”
Different institutions may decide to determine their own risk thresholds. One hospital, for example, may decide that any health threat that is associated with a 10% risk of death qualifies for a lifesaving abortion. But for many people, a 1 in 10 chance of dying is quite high.
“Who gets to decide what’s meaningful?” Dr. Harris asked, especially if the patient is already a parent of living children and doesn’t want to take any risk at all of orphaning them for a pregnancy with severe complications.
“The point is that this is way more complicated than anybody really knows, way more complicated than any legislator or justice could possibly know, and it creates all kinds of complicated ambiguities, some of which could result in harm to women,” she said. “I’ve been a doctor almost 30 years, and every week, sometimes every day, I’m humbled by how complicated pregnancy is and how complicated people’s bodies and life situations are.”
That’s what makes it so dangerous for policymakers to “insert themselves into medical practice,” Ms. Nash said. She worries about the legal ramifications of overturning Roe, such as prosecution of people who illegally undergo an abortion or of physicians who perform a procedure that a judge deems to be in violation of abortion law.
“There are already local prosecutors who have misused the law to go after people who have managed their own abortions,” Ms. Nash said. “Criminal abortion law, fetal homicide, child neglect, practicing medicine without a license – these are things people have actually been arrested and convicted under.”
Some laws may target the person seeking an abortion, whereas others may target clinicians providing abortions, or even people who simply help someone obtain an abortion, as the Texas law does. In Dr. Harris’s own state of Michigan, a group of Republican lawmakers recently introduced a bill that would imprison abortion providers for up to 10 years and anyone creating or distributing abortion medication for up to 20 years.
Michigan Gov. Gretchen Whitmer, a Democrat who called the proposed legislation “disturbing” and “infuriating,” would almost certainly veto the bill, but it’s just one of dozens already filed or that are expected to be filed across the United States.
The antiabortion organization National Right to Life has published a “post-Roe model abortion law” for states to adopt. The model includes an exemption for abortions that, “based on reasonable medical judgment, [were] necessary to prevent the death of the pregnant woman” – but, again, it does not clarify what that means in practice.
Lectures from strangers
Four years after nearly dying, Kami gave birth to a healthy girl following an uncomplicated pregnancy. But her journey to having more children presented more challenges.
Two years after the birth of her child, she had another ectopic pregnancy. Her doctor sent her prescriptions for medication that would end this pregnancy, but a pharmacist refused to fill the prescription.
“Do you know these are very serious medications?” the pharmacist asked her. She did – she had taken them once before for another ectopic pregnancy. She was with her daughter, feeling devastated about losing yet another desired pregnancy. She simply wanted to get the medication and go home.
“‘So you’re trying to have a cheap abortion,’ he said, and 30 heads turned and looked at me. The whole pharmacy heard,” Kami said.
She told the pharmacist that she’d miscarried. She said he responded with: “So you have a dead baby in your body.”
Even after her doctor called to insist on filling the order, the man refused to fill it.
Kami left without the prescription, and her doctor performed a surgical dilation and curettage to remove the embryo from her fallopian tube for no fee.
Kami later tried again to have more children. She experienced another ruptured tube that she said nearly killed her.
“There was such a sense of pain knowing that I couldn’t have any more kids, but also the relief of knowing that I don’t have to go through this again,” Kami said. Now, however, with the Supreme Court having overturned Roe v. Wade, she has a new worry: “That my daughter will not have the same rights and access to health care that I did.”
A version of this article first appeared on Medscape.com.
Kami, a mother of one daughter in central Texas, lost three pregnancies in 2008. The third one nearly killed her.
The embryo became implanted in one of the fallopian tubes connecting her ovaries to her uterus. Because fallopian tubes can’t stretch to accommodate a fetus, patients must undergo surgery to remove the embryo before the tube ruptures. Failure to do so can result in internal bleeding and death.
But when Kami – who did not want to use her last name to avoid harassment – underwent an ultrasound to start the process of extracting the embryo, her doctor miscalculated how far along in the pregnancy she was and told her to come back in a few weeks.
She eventually did return, but only after passing out in the bathtub and waking up in a pool of her own blood. The tube had ruptured, and to remove it, emergency surgery was necessary.
Stories such as Kami’s could become more common in the aftermath of the U.S. Supreme Court’s decision to overturn Roe v. Wade, the 1973 case that created a right to an abortion.
Experts fear that antiabortion laws that take effect in the United States following the court’s decision will lead to a medical and legal limbo for thousands of people like Kami – people with uncommon reproductive conditions whose treatments involve the termination of pregnancies or the destruction of embryos.
Vague exceptions prompt concerns
According to the Guttmacher Institute, a nonprofit group for reproductive health, 13 states currently have trigger laws on the books that make abortion illegal in the absence of Roe. Nine other states have laws that would outlaw or severely restrict abortion without a federal right to the procedure.
Each of these laws carves out exceptions that allow the termination of a pregnancy to prevent the death of the pregnant individual. But the language of the provisions is not always precise in describing what those exceptions mean in practice, according to Elizabeth Nash, the principal policy associate for state issues at the Guttmacher Institute.
“These exceptions are designed to be extraordinarily narrow. These aren’t really designed to be usable exceptions,” Ms. Nash said. “There’s so much misinformation about abortion that there are probably legislators out there who think that it’s never needed to save a life.”
Tubal pregnancies
One of the best examples of a pregnancy termination that’s necessary to avoid death is in the case of an ectopic pregnancy such as Kami experienced. Without treatment to end the pregnancy, the embryo will eventually grow so large that the tube ruptures, causing massive bleeding that can kill the mother.
Most state laws regarding abortion exclude treatment of ectopic pregnancy, according to Ms. Nash. But, “if the state does not exclude ectopic pregnancy from all the regulations, then people might not be able to get the care that they need when they need it.”
The current abortion law in Texas, for example, prohibits ending a pregnancy after 6 weeks, or after cardiac activity becomes present. Cardiac activity can be present in cases of ectopic pregnancies, which account for between 1% and 2% of all pregnancies and are the leading cause of maternal deaths in the first trimester. And treatment definitely ends the life of the embryo or fetus in the fallopian tube, said Lisa Harris, MD, PhD, an ob.gyn. and medical ethicist at the University of Michigan, Ann Arbor.
Dr. Harris said she has never doubted that an ectopic pregnancy cannot possibly result in a live birth. But she recalled an encounter with another clinician on a surgical team for an ectopic pregnancy who said: “So you’re going to take it out of the tube and put it in the uterus, right?”
“It was a startling moment,” Dr. Harris recalled. She regarded the procedure as a “lifesaving, obvious surgery,” but her colleague, whose suggestion was a medical impossibility since the window of implantation is very brief after fertilization, viewed it “as an abortion, as killing an embryo or fetus.”
Dr. Harris said she isn’t concerned that physicians would stop treating ectopic pregnancies in a post-Roe world. Rather, she worries about two other possibilities: an overzealous prosecutor might not believe it was an ectopic pregnancy and press charges; or laws will cause physicians to second guess their clinical decisions for patients.
“What it means, in the middle of the night, when someone comes in with a 10-week ectopic pregnancy with a heartbeat, is the doctor may hesitate,” Dr. Harris said. Despite knowing the appropriate treatment, the doctor may want to speak with a lawyer or ethicist first to ensure they are covered legally. “And as that process unfolds, which could take hours or days, the person might have a complication.” s
Not treating an ectopic pregnancy would be malpractice, but “some doctors may not provide the standard of care that they would have ordinarily provided because they don’t want to risk breaking the law,” she said.
Even more ambiguous are cornual ectopic pregnancies, in which the implantation occurs at the junction of a fallopian tube and the uterus. These pregnancies, which make up 2%-4% of all tubal pregnancies, are immediately adjacent to the uterus. If an abortion is defined as the termination of an embryo or fetus in the womb, how such a legal definition would apply to these pregnancies is unclear.
An ob.gyn. wouldn’t regard ending an ectopic pregnancy as an abortion, but “this is not about logic or clinical meaning,” Dr. Harris said. “This is people outside of medicine making determinations that all pregnancies must continue, and when you think of a ban that way, you could see why a doctor would be frightened to end a pregnancy, whether it might be viable in the future or not.”
That’s true even if the pregnancy is located fully in the uterus. Dr. Harris described a pregnant patient she saw who had traveled from Texas to Michigan with a fetus that had a lethal defect.
The fetus had “an anomaly where the lungs couldn’t develop, where there were no kidneys. There was no chance this baby could be born and live. Her doctors were very clear that there will never be a baby that [she could] take home at the end of this pregnancy, yet they would not end her pregnancy because that would be an abortion,” Dr. Harris said.
Texas law “doesn’t make any allowances for whether a pregnancy will ever actually result in a baby or not,” Dr. Harris said. “The law, in effect, just says all pregnancies must continue.”
Selective reduction
How abortion laws in different states might affect selective reduction, which is used in some pregnancies to reduce the total number of fetuses a person is carrying, is even more ambiguous. The goal of selective reduction is to decrease health risks to the pregnant individual and increase the likelihood of survival for the remaining fetuses. Current Texas law prohibits these procedures.
Someone pregnant with quintuplets, for example, might seek selection reduction to reduce the pregnancy outcome to triplets or twins. A related procedure, selective termination, is used to terminate the life of a fetus with abnormalities while the pregnancy of the fetus’ in utero siblings continues.
The advent of assisted reproduction methods, such as in vitro fertilization (IVF), greatly increased the incidence of higher-order multiples, those with three or more fetuses. The first IVF baby was born in 1978. By 1998, the rate of higher-order multiple births was 1.9 per 1,000 births, five times the figure in 1980. The rate has since decreased by nearly half, to 1 per 1,000 births, but with 3.75 million live births a year, that’s still a lot of pregnancies with higher-order multiples.
The American College of Obstetricians and Gynecologists does not provide explicit guidance on when selective reduction is warranted, but its committee opinion on multifetal pregnancy reduction provides an ethical framework for providers to use when counseling people with pregnancies of three or more fetuses. How would various state laws that outlaw abortion affect these decisions? No one knows.
“Selective reduction ends the life of a fetus or embryo, but it doesn’t end the pregnancy,” Dr. Harris said. “So, if the pregnancy continues but it kills an embryo or fetus, is that an abortion?”
‘The question of the hour’
Dr. Harris and other doctors are haunted by potential medical cases in which continuing a pregnancy may result in the death of the person carrying the fetus but in which such death may not be so imminent that the law would allow immediate termination of the pregnancy.
Michael Northrup, MD, an intensive care pediatrician in Winston-Salem, N.C., recalled a particularly harrowing case that illustrates the peril in deciding when someone’s life is “enough” in danger to qualify as an exception to abortion bans.
The 14-year-old girl had severe lupus and kidney failure that required treatment with methotrexate and immediate dialysis to replace her electrolytes. A standard pretreatment pregnancy test revealed that she had been carrying a child for at least 10 weeks. Her pregnancy presented two problems. Methotrexate is so severely toxic that it’s sometimes used to end pregnancies. Even at low doses, fetuses that survive usually have severe deformities. In addition, dialysis requires administration of a blood thinner. If the teen miscarried while taking a blood thinner during dialysis, she risked bleeding to death.
Treatment could be delayed until week 24 of pregnancy, at which time delivery could be attempted, but the patient likely wouldn’t have any kidney function left by then. In addition, at 24 weeks, it was unlikely that the baby would survive anyway.
Dr. Northrup said that, had she chosen that route, “I’m not sure she would have made it. This was a religious family, people who very much were believers. They had their head of church come in, who fairly quickly determined that the best thing for her health was to terminate this pregnancy immediately and get the treatment she needed for her body.”
Would such a situation qualify for an emergency termination? The girl wasn’t going to die within 24 or 48 hours, but it may not have been possible to pinpoint the time of death within a day or 2.
“The family was sad, but they made that choice, and I wonder, would we have to justify that with these new laws?” Dr. Northrup said. “You definitely worry, being in the hot seat, ‘Does this count enough? Is she close enough to death?’ ”
The same question comes up when someone’s water breaks early in the second trimester. Since a live-birth delivery would be highly unlikely, given the age of the fetus, the standard of care is to offer to terminate the pregnancy to avoid a serious infection, Dr. Harris said. But if the infection hasn’t yet developed – even if it’s likely to develop soon – doctors in a state that outlaws abortion would not be able to offer termination. But as providers wait for an infection to develop, the person’s risk of dying from infection rapidly increases.
“How likely does someone need to be to die for it to count to get a life-preserving abortion?” Dr. Harris asked. “That, I think, is the question of the hour.”
Different institutions may decide to determine their own risk thresholds. One hospital, for example, may decide that any health threat that is associated with a 10% risk of death qualifies for a lifesaving abortion. But for many people, a 1 in 10 chance of dying is quite high.
“Who gets to decide what’s meaningful?” Dr. Harris asked, especially if the patient is already a parent of living children and doesn’t want to take any risk at all of orphaning them for a pregnancy with severe complications.
“The point is that this is way more complicated than anybody really knows, way more complicated than any legislator or justice could possibly know, and it creates all kinds of complicated ambiguities, some of which could result in harm to women,” she said. “I’ve been a doctor almost 30 years, and every week, sometimes every day, I’m humbled by how complicated pregnancy is and how complicated people’s bodies and life situations are.”
That’s what makes it so dangerous for policymakers to “insert themselves into medical practice,” Ms. Nash said. She worries about the legal ramifications of overturning Roe, such as prosecution of people who illegally undergo an abortion or of physicians who perform a procedure that a judge deems to be in violation of abortion law.
“There are already local prosecutors who have misused the law to go after people who have managed their own abortions,” Ms. Nash said. “Criminal abortion law, fetal homicide, child neglect, practicing medicine without a license – these are things people have actually been arrested and convicted under.”
Some laws may target the person seeking an abortion, whereas others may target clinicians providing abortions, or even people who simply help someone obtain an abortion, as the Texas law does. In Dr. Harris’s own state of Michigan, a group of Republican lawmakers recently introduced a bill that would imprison abortion providers for up to 10 years and anyone creating or distributing abortion medication for up to 20 years.
Michigan Gov. Gretchen Whitmer, a Democrat who called the proposed legislation “disturbing” and “infuriating,” would almost certainly veto the bill, but it’s just one of dozens already filed or that are expected to be filed across the United States.
The antiabortion organization National Right to Life has published a “post-Roe model abortion law” for states to adopt. The model includes an exemption for abortions that, “based on reasonable medical judgment, [were] necessary to prevent the death of the pregnant woman” – but, again, it does not clarify what that means in practice.
Lectures from strangers
Four years after nearly dying, Kami gave birth to a healthy girl following an uncomplicated pregnancy. But her journey to having more children presented more challenges.
Two years after the birth of her child, she had another ectopic pregnancy. Her doctor sent her prescriptions for medication that would end this pregnancy, but a pharmacist refused to fill the prescription.
“Do you know these are very serious medications?” the pharmacist asked her. She did – she had taken them once before for another ectopic pregnancy. She was with her daughter, feeling devastated about losing yet another desired pregnancy. She simply wanted to get the medication and go home.
“‘So you’re trying to have a cheap abortion,’ he said, and 30 heads turned and looked at me. The whole pharmacy heard,” Kami said.
She told the pharmacist that she’d miscarried. She said he responded with: “So you have a dead baby in your body.”
Even after her doctor called to insist on filling the order, the man refused to fill it.
Kami left without the prescription, and her doctor performed a surgical dilation and curettage to remove the embryo from her fallopian tube for no fee.
Kami later tried again to have more children. She experienced another ruptured tube that she said nearly killed her.
“There was such a sense of pain knowing that I couldn’t have any more kids, but also the relief of knowing that I don’t have to go through this again,” Kami said. Now, however, with the Supreme Court having overturned Roe v. Wade, she has a new worry: “That my daughter will not have the same rights and access to health care that I did.”
A version of this article first appeared on Medscape.com.
Kami, a mother of one daughter in central Texas, lost three pregnancies in 2008. The third one nearly killed her.
The embryo became implanted in one of the fallopian tubes connecting her ovaries to her uterus. Because fallopian tubes can’t stretch to accommodate a fetus, patients must undergo surgery to remove the embryo before the tube ruptures. Failure to do so can result in internal bleeding and death.
But when Kami – who did not want to use her last name to avoid harassment – underwent an ultrasound to start the process of extracting the embryo, her doctor miscalculated how far along in the pregnancy she was and told her to come back in a few weeks.
She eventually did return, but only after passing out in the bathtub and waking up in a pool of her own blood. The tube had ruptured, and to remove it, emergency surgery was necessary.
Stories such as Kami’s could become more common in the aftermath of the U.S. Supreme Court’s decision to overturn Roe v. Wade, the 1973 case that created a right to an abortion.
Experts fear that antiabortion laws that take effect in the United States following the court’s decision will lead to a medical and legal limbo for thousands of people like Kami – people with uncommon reproductive conditions whose treatments involve the termination of pregnancies or the destruction of embryos.
Vague exceptions prompt concerns
According to the Guttmacher Institute, a nonprofit group for reproductive health, 13 states currently have trigger laws on the books that make abortion illegal in the absence of Roe. Nine other states have laws that would outlaw or severely restrict abortion without a federal right to the procedure.
Each of these laws carves out exceptions that allow the termination of a pregnancy to prevent the death of the pregnant individual. But the language of the provisions is not always precise in describing what those exceptions mean in practice, according to Elizabeth Nash, the principal policy associate for state issues at the Guttmacher Institute.
“These exceptions are designed to be extraordinarily narrow. These aren’t really designed to be usable exceptions,” Ms. Nash said. “There’s so much misinformation about abortion that there are probably legislators out there who think that it’s never needed to save a life.”
Tubal pregnancies
One of the best examples of a pregnancy termination that’s necessary to avoid death is in the case of an ectopic pregnancy such as Kami experienced. Without treatment to end the pregnancy, the embryo will eventually grow so large that the tube ruptures, causing massive bleeding that can kill the mother.
Most state laws regarding abortion exclude treatment of ectopic pregnancy, according to Ms. Nash. But, “if the state does not exclude ectopic pregnancy from all the regulations, then people might not be able to get the care that they need when they need it.”
The current abortion law in Texas, for example, prohibits ending a pregnancy after 6 weeks, or after cardiac activity becomes present. Cardiac activity can be present in cases of ectopic pregnancies, which account for between 1% and 2% of all pregnancies and are the leading cause of maternal deaths in the first trimester. And treatment definitely ends the life of the embryo or fetus in the fallopian tube, said Lisa Harris, MD, PhD, an ob.gyn. and medical ethicist at the University of Michigan, Ann Arbor.
Dr. Harris said she has never doubted that an ectopic pregnancy cannot possibly result in a live birth. But she recalled an encounter with another clinician on a surgical team for an ectopic pregnancy who said: “So you’re going to take it out of the tube and put it in the uterus, right?”
“It was a startling moment,” Dr. Harris recalled. She regarded the procedure as a “lifesaving, obvious surgery,” but her colleague, whose suggestion was a medical impossibility since the window of implantation is very brief after fertilization, viewed it “as an abortion, as killing an embryo or fetus.”
Dr. Harris said she isn’t concerned that physicians would stop treating ectopic pregnancies in a post-Roe world. Rather, she worries about two other possibilities: an overzealous prosecutor might not believe it was an ectopic pregnancy and press charges; or laws will cause physicians to second guess their clinical decisions for patients.
“What it means, in the middle of the night, when someone comes in with a 10-week ectopic pregnancy with a heartbeat, is the doctor may hesitate,” Dr. Harris said. Despite knowing the appropriate treatment, the doctor may want to speak with a lawyer or ethicist first to ensure they are covered legally. “And as that process unfolds, which could take hours or days, the person might have a complication.” s
Not treating an ectopic pregnancy would be malpractice, but “some doctors may not provide the standard of care that they would have ordinarily provided because they don’t want to risk breaking the law,” she said.
Even more ambiguous are cornual ectopic pregnancies, in which the implantation occurs at the junction of a fallopian tube and the uterus. These pregnancies, which make up 2%-4% of all tubal pregnancies, are immediately adjacent to the uterus. If an abortion is defined as the termination of an embryo or fetus in the womb, how such a legal definition would apply to these pregnancies is unclear.
An ob.gyn. wouldn’t regard ending an ectopic pregnancy as an abortion, but “this is not about logic or clinical meaning,” Dr. Harris said. “This is people outside of medicine making determinations that all pregnancies must continue, and when you think of a ban that way, you could see why a doctor would be frightened to end a pregnancy, whether it might be viable in the future or not.”
That’s true even if the pregnancy is located fully in the uterus. Dr. Harris described a pregnant patient she saw who had traveled from Texas to Michigan with a fetus that had a lethal defect.
The fetus had “an anomaly where the lungs couldn’t develop, where there were no kidneys. There was no chance this baby could be born and live. Her doctors were very clear that there will never be a baby that [she could] take home at the end of this pregnancy, yet they would not end her pregnancy because that would be an abortion,” Dr. Harris said.
Texas law “doesn’t make any allowances for whether a pregnancy will ever actually result in a baby or not,” Dr. Harris said. “The law, in effect, just says all pregnancies must continue.”
Selective reduction
How abortion laws in different states might affect selective reduction, which is used in some pregnancies to reduce the total number of fetuses a person is carrying, is even more ambiguous. The goal of selective reduction is to decrease health risks to the pregnant individual and increase the likelihood of survival for the remaining fetuses. Current Texas law prohibits these procedures.
Someone pregnant with quintuplets, for example, might seek selection reduction to reduce the pregnancy outcome to triplets or twins. A related procedure, selective termination, is used to terminate the life of a fetus with abnormalities while the pregnancy of the fetus’ in utero siblings continues.
The advent of assisted reproduction methods, such as in vitro fertilization (IVF), greatly increased the incidence of higher-order multiples, those with three or more fetuses. The first IVF baby was born in 1978. By 1998, the rate of higher-order multiple births was 1.9 per 1,000 births, five times the figure in 1980. The rate has since decreased by nearly half, to 1 per 1,000 births, but with 3.75 million live births a year, that’s still a lot of pregnancies with higher-order multiples.
The American College of Obstetricians and Gynecologists does not provide explicit guidance on when selective reduction is warranted, but its committee opinion on multifetal pregnancy reduction provides an ethical framework for providers to use when counseling people with pregnancies of three or more fetuses. How would various state laws that outlaw abortion affect these decisions? No one knows.
“Selective reduction ends the life of a fetus or embryo, but it doesn’t end the pregnancy,” Dr. Harris said. “So, if the pregnancy continues but it kills an embryo or fetus, is that an abortion?”
‘The question of the hour’
Dr. Harris and other doctors are haunted by potential medical cases in which continuing a pregnancy may result in the death of the person carrying the fetus but in which such death may not be so imminent that the law would allow immediate termination of the pregnancy.
Michael Northrup, MD, an intensive care pediatrician in Winston-Salem, N.C., recalled a particularly harrowing case that illustrates the peril in deciding when someone’s life is “enough” in danger to qualify as an exception to abortion bans.
The 14-year-old girl had severe lupus and kidney failure that required treatment with methotrexate and immediate dialysis to replace her electrolytes. A standard pretreatment pregnancy test revealed that she had been carrying a child for at least 10 weeks. Her pregnancy presented two problems. Methotrexate is so severely toxic that it’s sometimes used to end pregnancies. Even at low doses, fetuses that survive usually have severe deformities. In addition, dialysis requires administration of a blood thinner. If the teen miscarried while taking a blood thinner during dialysis, she risked bleeding to death.
Treatment could be delayed until week 24 of pregnancy, at which time delivery could be attempted, but the patient likely wouldn’t have any kidney function left by then. In addition, at 24 weeks, it was unlikely that the baby would survive anyway.
Dr. Northrup said that, had she chosen that route, “I’m not sure she would have made it. This was a religious family, people who very much were believers. They had their head of church come in, who fairly quickly determined that the best thing for her health was to terminate this pregnancy immediately and get the treatment she needed for her body.”
Would such a situation qualify for an emergency termination? The girl wasn’t going to die within 24 or 48 hours, but it may not have been possible to pinpoint the time of death within a day or 2.
“The family was sad, but they made that choice, and I wonder, would we have to justify that with these new laws?” Dr. Northrup said. “You definitely worry, being in the hot seat, ‘Does this count enough? Is she close enough to death?’ ”
The same question comes up when someone’s water breaks early in the second trimester. Since a live-birth delivery would be highly unlikely, given the age of the fetus, the standard of care is to offer to terminate the pregnancy to avoid a serious infection, Dr. Harris said. But if the infection hasn’t yet developed – even if it’s likely to develop soon – doctors in a state that outlaws abortion would not be able to offer termination. But as providers wait for an infection to develop, the person’s risk of dying from infection rapidly increases.
“How likely does someone need to be to die for it to count to get a life-preserving abortion?” Dr. Harris asked. “That, I think, is the question of the hour.”
Different institutions may decide to determine their own risk thresholds. One hospital, for example, may decide that any health threat that is associated with a 10% risk of death qualifies for a lifesaving abortion. But for many people, a 1 in 10 chance of dying is quite high.
“Who gets to decide what’s meaningful?” Dr. Harris asked, especially if the patient is already a parent of living children and doesn’t want to take any risk at all of orphaning them for a pregnancy with severe complications.
“The point is that this is way more complicated than anybody really knows, way more complicated than any legislator or justice could possibly know, and it creates all kinds of complicated ambiguities, some of which could result in harm to women,” she said. “I’ve been a doctor almost 30 years, and every week, sometimes every day, I’m humbled by how complicated pregnancy is and how complicated people’s bodies and life situations are.”
That’s what makes it so dangerous for policymakers to “insert themselves into medical practice,” Ms. Nash said. She worries about the legal ramifications of overturning Roe, such as prosecution of people who illegally undergo an abortion or of physicians who perform a procedure that a judge deems to be in violation of abortion law.
“There are already local prosecutors who have misused the law to go after people who have managed their own abortions,” Ms. Nash said. “Criminal abortion law, fetal homicide, child neglect, practicing medicine without a license – these are things people have actually been arrested and convicted under.”
Some laws may target the person seeking an abortion, whereas others may target clinicians providing abortions, or even people who simply help someone obtain an abortion, as the Texas law does. In Dr. Harris’s own state of Michigan, a group of Republican lawmakers recently introduced a bill that would imprison abortion providers for up to 10 years and anyone creating or distributing abortion medication for up to 20 years.
Michigan Gov. Gretchen Whitmer, a Democrat who called the proposed legislation “disturbing” and “infuriating,” would almost certainly veto the bill, but it’s just one of dozens already filed or that are expected to be filed across the United States.
The antiabortion organization National Right to Life has published a “post-Roe model abortion law” for states to adopt. The model includes an exemption for abortions that, “based on reasonable medical judgment, [were] necessary to prevent the death of the pregnant woman” – but, again, it does not clarify what that means in practice.
Lectures from strangers
Four years after nearly dying, Kami gave birth to a healthy girl following an uncomplicated pregnancy. But her journey to having more children presented more challenges.
Two years after the birth of her child, she had another ectopic pregnancy. Her doctor sent her prescriptions for medication that would end this pregnancy, but a pharmacist refused to fill the prescription.
“Do you know these are very serious medications?” the pharmacist asked her. She did – she had taken them once before for another ectopic pregnancy. She was with her daughter, feeling devastated about losing yet another desired pregnancy. She simply wanted to get the medication and go home.
“‘So you’re trying to have a cheap abortion,’ he said, and 30 heads turned and looked at me. The whole pharmacy heard,” Kami said.
She told the pharmacist that she’d miscarried. She said he responded with: “So you have a dead baby in your body.”
Even after her doctor called to insist on filling the order, the man refused to fill it.
Kami left without the prescription, and her doctor performed a surgical dilation and curettage to remove the embryo from her fallopian tube for no fee.
Kami later tried again to have more children. She experienced another ruptured tube that she said nearly killed her.
“There was such a sense of pain knowing that I couldn’t have any more kids, but also the relief of knowing that I don’t have to go through this again,” Kami said. Now, however, with the Supreme Court having overturned Roe v. Wade, she has a new worry: “That my daughter will not have the same rights and access to health care that I did.”
A version of this article first appeared on Medscape.com.
Biomarkers may help to predict persistent oligoarticular JIA
Ongoing research in patients with oligoarticular juvenile idiopathic arthritis (JIA) so far suggests that a set of biomarkers in synovial fluid may help to predict which patients may be more likely to stay with persistent oligoarticular disease rather than progress to polyarticular disease, according to new research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year. Identifying biomarkers in synovial fluid or possibly serum could aid families and physicians in being more proactive in treatment protocols, said AnneMarie C. Brescia, MD, of Nemours Children’s Hospital in Wilmington, Del.
“JIA carries the risk of permanent joint damage and disability, which can result when joint involvement evolves from oligoarticular into a polyarticular course, termed extended oligoarticular disease,” Dr. Brescia told attendees. “Since disease progression increases the risk for disability, early prediction of this course is essential.”
This group – those whose oligoarticular disease will begin recruiting joints and ultimately become extended oligoarticular JIA – is “very important because they have been shown to have worse health-related quality of life and greater risk of needing a joint replacement than even polyarticular [JIA],” Dr. Brescia said. “So, our lab has really focused on trying to predict who will fall in this group.”
Melissa Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University in Indianapolis, was not involved in the study but agreed that having highly sensitive and specific biomarkers could be particularly helpful in clinical care.
“Biomarkers can help guide treatment decisions and help physicians and their patients share the decision-making about next choices and when to change,” Dr. Oliver told this news organization. “If a provider and parent know that their child has these markers in their serum or synovial fluid that may predict extension of their disease, then they may be more aggressive upfront with therapy.”
The study aimed to determine whether differential levels of synovial fluid proteins could be used to predict whether JIA would evolve into an extended course before it became clinically evident. Although early aggressive treatment is common with rheumatoid arthritis and can lead to remission, JIA treatment paradigms tend to be more reactive, Dr. Brescia said.
“It would be better to switch to proactive, that if we’re able to predict that this patient may have a more difficult course with extension to polyarticular, we could be prepared, we could inform the parents, and it would just help us have a more proactive approach,” she said.
The researchers used antibody arrays to detect the following inflammatory mediators in blinded samples: CD14, interleukin (IL)-1-alpha, IL-3, IL-5, IL-6, vascular endothelial growth factor (VEGF), and angiogenin. They analyzed 37 samples with persistent disease and 32 samples from disease that had not yet extended but would become extended in that patient. The samples came from patients who were taking no medicines or only NSAIDs. The researchers assessed the sensitivity and specificity of each biomarker. Sensitivity referred the biomarker’s ability to correctly indicate that the sample would extend, and specificity referred to the biomarker’s accuracy in determining that the disease in the sample would remain persistent.
Combining samples from cohorts at Nemours Children’s Health (14 persistent and 7 extended-to-be) and Cincinnati Children’s Hospital (23 persistent and 25 extended-to-be) yielded the following results: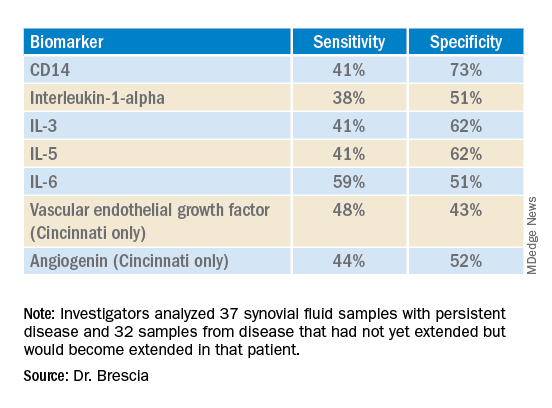
The findings revealed that the selected biomarkers were more accurate at predicting whose disease would remain persistent than predicting those that would extend, Dr. Brescia said. CD14 was the most specific biomarker, and IL-6 was the most sensitive biomarker in both groups.
When the researchers translated the findings from ELISA to the Luminex platform, positive results in synovial fluid for all these biomarkers were also positive in serum samples. Although the differences between persistent and extended-to-be samples did not reach statistical significance using Luminex, the pattern was the same for each biomarker.
“Luminex is more sensitive than ELISA. We believe that conducting an LDA [linear discriminant analysis] using these Luminex measurements will allow us to determine new cutoffs or new protein levels that are appropriate for Luminex to predict who will extend,” Dr. Brescia said. “It’s also our goal to develop a serum panel because ... being able to detect these markers in serum would expand the applicability of these markers to more patients.”
Dr. Brescia then described the group’s work in defining clinically relevant subpopulations of patients based on fibroblast-like synoviocytes (FLS) cells in the synovial intimal lining that produce inflammatory cytokines.
“Our compelling, single-cell, RNA sequencing preliminary data revealing multiple subpopulations within the total FLS population supports our hypothesis that distinct FLS subpopulations correlate with clinical outcome,” said Dr. Brescia. They looked at the percentage of chondrocyte-like, fibroblast-like, and smooth muscle-like subpopulations in samples from patients with oligoarticular JIA, extended-to-be JIA, and polyarticular JIA. Chondrocytes occurred in the largest proportion, and polyarticular JIA FLS had the largest percentage of chondrocytes, compared with the other two subpopulation groups.
“This is a work in progress,” Dr. Brescia said, “so hopefully you’ll hear about it next year.” In response to an attendee’s question, she said she believes identifying reliable biomarkers will eventually lead to refining treatment paradigms.
“I think it will at least change the guidance we can provide parents about making next choices and how quickly to accelerate to those next choices,” Dr. Brescia said. For example, if a child’s serum or synovial fluid has markers that show a very high likelihood of extension, the parent may decide to proceed to the next level medication sooner. “I do think it will push both parents and doctors to be a little more proactive instead of reactive when the poor patient comes back with 13 joints involved when they had just been an oligo for years.”
Dr. Oliver noted the promise of CD14 and IL-6 in potentially predicting which patients’ disease will stay persistent but cautioned that it’s still early in evaluating these biomarkers, especially with the limited patient samples in this study.
“I think these results are promising, and it’s great that there are groups out there working on this,” Dr. Oliver said. “Once we have a reliable, highly sensitive and specific biomarker, that will definitely help providers, parents, and patients be more informed.”
The research was supported by the Open Net Foundation, the Arthritis Foundation, Delaware Community Foundation, the Delaware Clinical and Translational Research (DE-CTR) ACCEL Program, the Nancy Taylor Foundation for Chronic Diseases, and CARRA. Dr. Brescia and Dr. Oliver have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ongoing research in patients with oligoarticular juvenile idiopathic arthritis (JIA) so far suggests that a set of biomarkers in synovial fluid may help to predict which patients may be more likely to stay with persistent oligoarticular disease rather than progress to polyarticular disease, according to new research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year. Identifying biomarkers in synovial fluid or possibly serum could aid families and physicians in being more proactive in treatment protocols, said AnneMarie C. Brescia, MD, of Nemours Children’s Hospital in Wilmington, Del.
“JIA carries the risk of permanent joint damage and disability, which can result when joint involvement evolves from oligoarticular into a polyarticular course, termed extended oligoarticular disease,” Dr. Brescia told attendees. “Since disease progression increases the risk for disability, early prediction of this course is essential.”
This group – those whose oligoarticular disease will begin recruiting joints and ultimately become extended oligoarticular JIA – is “very important because they have been shown to have worse health-related quality of life and greater risk of needing a joint replacement than even polyarticular [JIA],” Dr. Brescia said. “So, our lab has really focused on trying to predict who will fall in this group.”
Melissa Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University in Indianapolis, was not involved in the study but agreed that having highly sensitive and specific biomarkers could be particularly helpful in clinical care.
“Biomarkers can help guide treatment decisions and help physicians and their patients share the decision-making about next choices and when to change,” Dr. Oliver told this news organization. “If a provider and parent know that their child has these markers in their serum or synovial fluid that may predict extension of their disease, then they may be more aggressive upfront with therapy.”
The study aimed to determine whether differential levels of synovial fluid proteins could be used to predict whether JIA would evolve into an extended course before it became clinically evident. Although early aggressive treatment is common with rheumatoid arthritis and can lead to remission, JIA treatment paradigms tend to be more reactive, Dr. Brescia said.
“It would be better to switch to proactive, that if we’re able to predict that this patient may have a more difficult course with extension to polyarticular, we could be prepared, we could inform the parents, and it would just help us have a more proactive approach,” she said.
The researchers used antibody arrays to detect the following inflammatory mediators in blinded samples: CD14, interleukin (IL)-1-alpha, IL-3, IL-5, IL-6, vascular endothelial growth factor (VEGF), and angiogenin. They analyzed 37 samples with persistent disease and 32 samples from disease that had not yet extended but would become extended in that patient. The samples came from patients who were taking no medicines or only NSAIDs. The researchers assessed the sensitivity and specificity of each biomarker. Sensitivity referred the biomarker’s ability to correctly indicate that the sample would extend, and specificity referred to the biomarker’s accuracy in determining that the disease in the sample would remain persistent.
Combining samples from cohorts at Nemours Children’s Health (14 persistent and 7 extended-to-be) and Cincinnati Children’s Hospital (23 persistent and 25 extended-to-be) yielded the following results:
The findings revealed that the selected biomarkers were more accurate at predicting whose disease would remain persistent than predicting those that would extend, Dr. Brescia said. CD14 was the most specific biomarker, and IL-6 was the most sensitive biomarker in both groups.
When the researchers translated the findings from ELISA to the Luminex platform, positive results in synovial fluid for all these biomarkers were also positive in serum samples. Although the differences between persistent and extended-to-be samples did not reach statistical significance using Luminex, the pattern was the same for each biomarker.
“Luminex is more sensitive than ELISA. We believe that conducting an LDA [linear discriminant analysis] using these Luminex measurements will allow us to determine new cutoffs or new protein levels that are appropriate for Luminex to predict who will extend,” Dr. Brescia said. “It’s also our goal to develop a serum panel because ... being able to detect these markers in serum would expand the applicability of these markers to more patients.”
Dr. Brescia then described the group’s work in defining clinically relevant subpopulations of patients based on fibroblast-like synoviocytes (FLS) cells in the synovial intimal lining that produce inflammatory cytokines.
“Our compelling, single-cell, RNA sequencing preliminary data revealing multiple subpopulations within the total FLS population supports our hypothesis that distinct FLS subpopulations correlate with clinical outcome,” said Dr. Brescia. They looked at the percentage of chondrocyte-like, fibroblast-like, and smooth muscle-like subpopulations in samples from patients with oligoarticular JIA, extended-to-be JIA, and polyarticular JIA. Chondrocytes occurred in the largest proportion, and polyarticular JIA FLS had the largest percentage of chondrocytes, compared with the other two subpopulation groups.
“This is a work in progress,” Dr. Brescia said, “so hopefully you’ll hear about it next year.” In response to an attendee’s question, she said she believes identifying reliable biomarkers will eventually lead to refining treatment paradigms.
“I think it will at least change the guidance we can provide parents about making next choices and how quickly to accelerate to those next choices,” Dr. Brescia said. For example, if a child’s serum or synovial fluid has markers that show a very high likelihood of extension, the parent may decide to proceed to the next level medication sooner. “I do think it will push both parents and doctors to be a little more proactive instead of reactive when the poor patient comes back with 13 joints involved when they had just been an oligo for years.”
Dr. Oliver noted the promise of CD14 and IL-6 in potentially predicting which patients’ disease will stay persistent but cautioned that it’s still early in evaluating these biomarkers, especially with the limited patient samples in this study.
“I think these results are promising, and it’s great that there are groups out there working on this,” Dr. Oliver said. “Once we have a reliable, highly sensitive and specific biomarker, that will definitely help providers, parents, and patients be more informed.”
The research was supported by the Open Net Foundation, the Arthritis Foundation, Delaware Community Foundation, the Delaware Clinical and Translational Research (DE-CTR) ACCEL Program, the Nancy Taylor Foundation for Chronic Diseases, and CARRA. Dr. Brescia and Dr. Oliver have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ongoing research in patients with oligoarticular juvenile idiopathic arthritis (JIA) so far suggests that a set of biomarkers in synovial fluid may help to predict which patients may be more likely to stay with persistent oligoarticular disease rather than progress to polyarticular disease, according to new research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year. Identifying biomarkers in synovial fluid or possibly serum could aid families and physicians in being more proactive in treatment protocols, said AnneMarie C. Brescia, MD, of Nemours Children’s Hospital in Wilmington, Del.
“JIA carries the risk of permanent joint damage and disability, which can result when joint involvement evolves from oligoarticular into a polyarticular course, termed extended oligoarticular disease,” Dr. Brescia told attendees. “Since disease progression increases the risk for disability, early prediction of this course is essential.”
This group – those whose oligoarticular disease will begin recruiting joints and ultimately become extended oligoarticular JIA – is “very important because they have been shown to have worse health-related quality of life and greater risk of needing a joint replacement than even polyarticular [JIA],” Dr. Brescia said. “So, our lab has really focused on trying to predict who will fall in this group.”
Melissa Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University in Indianapolis, was not involved in the study but agreed that having highly sensitive and specific biomarkers could be particularly helpful in clinical care.
“Biomarkers can help guide treatment decisions and help physicians and their patients share the decision-making about next choices and when to change,” Dr. Oliver told this news organization. “If a provider and parent know that their child has these markers in their serum or synovial fluid that may predict extension of their disease, then they may be more aggressive upfront with therapy.”
The study aimed to determine whether differential levels of synovial fluid proteins could be used to predict whether JIA would evolve into an extended course before it became clinically evident. Although early aggressive treatment is common with rheumatoid arthritis and can lead to remission, JIA treatment paradigms tend to be more reactive, Dr. Brescia said.
“It would be better to switch to proactive, that if we’re able to predict that this patient may have a more difficult course with extension to polyarticular, we could be prepared, we could inform the parents, and it would just help us have a more proactive approach,” she said.
The researchers used antibody arrays to detect the following inflammatory mediators in blinded samples: CD14, interleukin (IL)-1-alpha, IL-3, IL-5, IL-6, vascular endothelial growth factor (VEGF), and angiogenin. They analyzed 37 samples with persistent disease and 32 samples from disease that had not yet extended but would become extended in that patient. The samples came from patients who were taking no medicines or only NSAIDs. The researchers assessed the sensitivity and specificity of each biomarker. Sensitivity referred the biomarker’s ability to correctly indicate that the sample would extend, and specificity referred to the biomarker’s accuracy in determining that the disease in the sample would remain persistent.
Combining samples from cohorts at Nemours Children’s Health (14 persistent and 7 extended-to-be) and Cincinnati Children’s Hospital (23 persistent and 25 extended-to-be) yielded the following results:
The findings revealed that the selected biomarkers were more accurate at predicting whose disease would remain persistent than predicting those that would extend, Dr. Brescia said. CD14 was the most specific biomarker, and IL-6 was the most sensitive biomarker in both groups.
When the researchers translated the findings from ELISA to the Luminex platform, positive results in synovial fluid for all these biomarkers were also positive in serum samples. Although the differences between persistent and extended-to-be samples did not reach statistical significance using Luminex, the pattern was the same for each biomarker.
“Luminex is more sensitive than ELISA. We believe that conducting an LDA [linear discriminant analysis] using these Luminex measurements will allow us to determine new cutoffs or new protein levels that are appropriate for Luminex to predict who will extend,” Dr. Brescia said. “It’s also our goal to develop a serum panel because ... being able to detect these markers in serum would expand the applicability of these markers to more patients.”
Dr. Brescia then described the group’s work in defining clinically relevant subpopulations of patients based on fibroblast-like synoviocytes (FLS) cells in the synovial intimal lining that produce inflammatory cytokines.
“Our compelling, single-cell, RNA sequencing preliminary data revealing multiple subpopulations within the total FLS population supports our hypothesis that distinct FLS subpopulations correlate with clinical outcome,” said Dr. Brescia. They looked at the percentage of chondrocyte-like, fibroblast-like, and smooth muscle-like subpopulations in samples from patients with oligoarticular JIA, extended-to-be JIA, and polyarticular JIA. Chondrocytes occurred in the largest proportion, and polyarticular JIA FLS had the largest percentage of chondrocytes, compared with the other two subpopulation groups.
“This is a work in progress,” Dr. Brescia said, “so hopefully you’ll hear about it next year.” In response to an attendee’s question, she said she believes identifying reliable biomarkers will eventually lead to refining treatment paradigms.
“I think it will at least change the guidance we can provide parents about making next choices and how quickly to accelerate to those next choices,” Dr. Brescia said. For example, if a child’s serum or synovial fluid has markers that show a very high likelihood of extension, the parent may decide to proceed to the next level medication sooner. “I do think it will push both parents and doctors to be a little more proactive instead of reactive when the poor patient comes back with 13 joints involved when they had just been an oligo for years.”
Dr. Oliver noted the promise of CD14 and IL-6 in potentially predicting which patients’ disease will stay persistent but cautioned that it’s still early in evaluating these biomarkers, especially with the limited patient samples in this study.
“I think these results are promising, and it’s great that there are groups out there working on this,” Dr. Oliver said. “Once we have a reliable, highly sensitive and specific biomarker, that will definitely help providers, parents, and patients be more informed.”
The research was supported by the Open Net Foundation, the Arthritis Foundation, Delaware Community Foundation, the Delaware Clinical and Translational Research (DE-CTR) ACCEL Program, the Nancy Taylor Foundation for Chronic Diseases, and CARRA. Dr. Brescia and Dr. Oliver have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CARRA 2022
‘Genetic’ height linked to peripheral neuropathy and certain skin and bone infections
, according to a study published in PLOS Genetics.
Prior studies have investigated height as a risk factor for chronic diseases, such as a higher risk for atrial fibrillation and a reduced risk of cardiovascular disease. It’s been consistently difficult, however, to eliminate the confounding influences of diet, socioeconomics, lifestyle behaviors, and other environmental factors that may interfere with a person’s reaching their expected height based on their genes.
This study, however, was able to better parse those differences by using Mendelian randomization within the comprehensive clinical and genetic dataset of a national health care system biobank. Mendelian randomization uses “genetic instruments for exposures of interest under the assumption that genotype is less susceptible to confounding than measured exposures,” the authors explained. The findings confirmed previously suspected associations between height and a range of cardiovascular and metabolic conditions as well as revealing new associations with several other conditions.
Prior associations confirmed, new associations uncovered
The results confirmed that being tall is linked to a higher risk of atrial fibrillation and varicose veins, and a lower risk of coronary heart disease, high blood pressure, and high cholesterol. The study also uncovered new associations between greater height and a higher risk of peripheral neuropathy, which is caused by damage to nerves on the extremities, as well as skin and bone infections, such as leg and foot ulcers.
The meta-analysis “identified five additional traits associated with genetically-predicted height,” wrote Sridharan Raghavan, MD, assistant professor of medicine at the University of Colorado Anschutz Medical Campus, and colleagues. “Two were genitourinary conditions – erectile dysfunction and urinary retention – that can be associated with neuropathy, and a third was a phecode for nonspecific skin disorders that may be related to skin infections – consistent with the race/ethnicity stratified results.”
Removing potential confounders
F. Perry Wilson, MD, associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study, said the findings were not particularly surprising overall, but it’s striking that the researchers had ”such a large cohort with such detailed electronic health records allowing for the comparison of genetic height with a variety of clinical outcomes.” He also noted the study’s strength in using Mendelian randomization so that the exposure is the predicted genetic height instead of a person’s measured height.
“This is key, since lots of things affect actual height – nutrition is an important one that could certainly be linked to disease as well,” Dr. Wilson said. ”By using genetic height, the authors remove these potential confounders. Since genetic height is “assigned” at birth (or conception), there is little opportunity for confounding. Of course, it is possible that some of the gene variants used to predict genetic height actually do something else, such as make you seek out less nutritious meals, but by and large this is how these types of studies need to be done.”
Height may impact over 100 clinical traits
The study relied on data from the U.S. Veteran Affairs Million Veteran Program with 222,300 non-Hispanic White and 58,151 non-Hispanic Black participants. The researchers first estimated the likelihood of participants’ genetic height based on 3,290 genetic variants determined to affect genetic height in a recent European-ancestry genome-wide meta-analysis. Then they compared these estimates with participants’ actual height in the VA medical record, adjusting for age, sex, and other genetic characteristics.
In doing so, the researchers found 345 clinical traits that were associated with the actual measured height in White participants plus another 17 clinical trials linked to actual measured height in Black participants. An overall 127 of these clinical traits were significantly associated with White participants’ genetically predicted height, and two of them were significantly associated with Black participants’ genetically predicted height.
In analyzing all these data together, the researchers were largely able to separate out those associations between genetically predicted height and certain health conditions from those associations between health conditions and a person’s actual measured height. They also determined that including body mass index as a covariate had little impact on the results. The researchers conducted the appropriate statistical correction to ensure the use of so many variables did not result in spurious statistical significance in some associations.
“Using genetic methods applied to the VA Million Veteran Program, we found evidence that adult height may impact over 100 clinical traits, including several conditions associated with poor outcomes and quality of life – peripheral neuropathy, lower extremity ulcers, and chronic venous insufficiency. We conclude that height may be an unrecognized nonmodifiable risk factor for several common conditions in adults.”
Height linked with health conditions
Genetically predicted height predicted a reduced risk of hyperlipidemia and hypertension independent of coronary heart disease, the analysis revealed. Genetically predicted height was also linked to an approximately 51% increased risk of atrial fibrillation in participants without coronary heart disease but, paradoxically, only a 39% increased risk in those with coronary heart disease, despite coronary heart disease being a risk factor for atrial fibrillation. Genetically predicted height was also associated with a greater risk of varicose veins in the legs and deep vein thrombosis.
Another novel association uncovered by the analysis was between women’s genetically predicted height and both asthma and nonspecific peripheral nerve disorders. “Whether these associations reflect differences by sex in disease pathophysiology related to height may warrant exploration in a sample with better balance between men and women,” the authors wrote. “In sum, our results suggest that an individual’s height may warrant consideration as a nonmodifiable predictor for several common conditions, particularly those affecting peripheral/distal extremities that are most physically impacted by tall stature.”
A substantial limitation of the study was its homogeneity of participants, who were 92% male with an average height of 176 cm and an average BMI of 30.1. The Black participants tended to be younger, with an average age of 58 compared with 64 years in the White participants, but the groups were otherwise similar in height and weight.* The database included data from Hispanic participants, but the researchers excluded these data because of the small sample size.
The smaller dataset for Black participants was a limitation as well as the fact that the genome-wide association study the researchers relied on came from a European population, which may not be as accurate in people with other ancestry, Dr. Wilson said. The bigger limitation, however, is what the findings’ clinical relevance is.
What does it all mean?
“Genetic height is in your genes – there is nothing to be done about it – so it is more of academic interest than clinical interest,” Dr. Wilson said. It’s not even clear whether incorporating a person’s height – actual or genetically predicted, if it could be easily determined for each person – into risk calculators. ”To know whether it would be beneficial to use height (or genetic height) as a risk factor, you’d need to examine each condition of interest, adjusting for all known risk factors, to see if height improved the prediction,” Dr. Wilson said. “I suspect for most conditions, the well-known risk factors would swamp height. For example, high genetic height might truly increase risk for neuropathy. But diabetes might increase the risk so much more that height is not particularly relevant.”
On the other hand, the fact that height in general has any potential influence at all on disease risk may inspire physicians to consider other risk factors in especially tall individuals.
”Physicians may find it interesting that we have some confirmation that height does increase the risk of certain conditions,” Dr. Wilson said. “While this is unlikely to dramatically change practice, they may be a bit more diligent in looking for other relevant risk factors for the diseases found in this study in their very tall patients.”
The research was funded by the U.S. Department of Veteran Affairs, the Boettcher Foundation’s Webb-Waring Biomedical Research Program, the National Institutes of Health, and a Linda Pechenik Montague Investigator award. One study coauthor is a full-time employee of Novartis Institutes of Biomedical Research. The other authors and Dr. Wilson had no disclosures.
*Correction, 6/29/22: An earlier version of this article misstated the average age of Black participants.
, according to a study published in PLOS Genetics.
Prior studies have investigated height as a risk factor for chronic diseases, such as a higher risk for atrial fibrillation and a reduced risk of cardiovascular disease. It’s been consistently difficult, however, to eliminate the confounding influences of diet, socioeconomics, lifestyle behaviors, and other environmental factors that may interfere with a person’s reaching their expected height based on their genes.
This study, however, was able to better parse those differences by using Mendelian randomization within the comprehensive clinical and genetic dataset of a national health care system biobank. Mendelian randomization uses “genetic instruments for exposures of interest under the assumption that genotype is less susceptible to confounding than measured exposures,” the authors explained. The findings confirmed previously suspected associations between height and a range of cardiovascular and metabolic conditions as well as revealing new associations with several other conditions.
Prior associations confirmed, new associations uncovered
The results confirmed that being tall is linked to a higher risk of atrial fibrillation and varicose veins, and a lower risk of coronary heart disease, high blood pressure, and high cholesterol. The study also uncovered new associations between greater height and a higher risk of peripheral neuropathy, which is caused by damage to nerves on the extremities, as well as skin and bone infections, such as leg and foot ulcers.
The meta-analysis “identified five additional traits associated with genetically-predicted height,” wrote Sridharan Raghavan, MD, assistant professor of medicine at the University of Colorado Anschutz Medical Campus, and colleagues. “Two were genitourinary conditions – erectile dysfunction and urinary retention – that can be associated with neuropathy, and a third was a phecode for nonspecific skin disorders that may be related to skin infections – consistent with the race/ethnicity stratified results.”
Removing potential confounders
F. Perry Wilson, MD, associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study, said the findings were not particularly surprising overall, but it’s striking that the researchers had ”such a large cohort with such detailed electronic health records allowing for the comparison of genetic height with a variety of clinical outcomes.” He also noted the study’s strength in using Mendelian randomization so that the exposure is the predicted genetic height instead of a person’s measured height.
“This is key, since lots of things affect actual height – nutrition is an important one that could certainly be linked to disease as well,” Dr. Wilson said. ”By using genetic height, the authors remove these potential confounders. Since genetic height is “assigned” at birth (or conception), there is little opportunity for confounding. Of course, it is possible that some of the gene variants used to predict genetic height actually do something else, such as make you seek out less nutritious meals, but by and large this is how these types of studies need to be done.”
Height may impact over 100 clinical traits
The study relied on data from the U.S. Veteran Affairs Million Veteran Program with 222,300 non-Hispanic White and 58,151 non-Hispanic Black participants. The researchers first estimated the likelihood of participants’ genetic height based on 3,290 genetic variants determined to affect genetic height in a recent European-ancestry genome-wide meta-analysis. Then they compared these estimates with participants’ actual height in the VA medical record, adjusting for age, sex, and other genetic characteristics.
In doing so, the researchers found 345 clinical traits that were associated with the actual measured height in White participants plus another 17 clinical trials linked to actual measured height in Black participants. An overall 127 of these clinical traits were significantly associated with White participants’ genetically predicted height, and two of them were significantly associated with Black participants’ genetically predicted height.
In analyzing all these data together, the researchers were largely able to separate out those associations between genetically predicted height and certain health conditions from those associations between health conditions and a person’s actual measured height. They also determined that including body mass index as a covariate had little impact on the results. The researchers conducted the appropriate statistical correction to ensure the use of so many variables did not result in spurious statistical significance in some associations.
“Using genetic methods applied to the VA Million Veteran Program, we found evidence that adult height may impact over 100 clinical traits, including several conditions associated with poor outcomes and quality of life – peripheral neuropathy, lower extremity ulcers, and chronic venous insufficiency. We conclude that height may be an unrecognized nonmodifiable risk factor for several common conditions in adults.”
Height linked with health conditions
Genetically predicted height predicted a reduced risk of hyperlipidemia and hypertension independent of coronary heart disease, the analysis revealed. Genetically predicted height was also linked to an approximately 51% increased risk of atrial fibrillation in participants without coronary heart disease but, paradoxically, only a 39% increased risk in those with coronary heart disease, despite coronary heart disease being a risk factor for atrial fibrillation. Genetically predicted height was also associated with a greater risk of varicose veins in the legs and deep vein thrombosis.
Another novel association uncovered by the analysis was between women’s genetically predicted height and both asthma and nonspecific peripheral nerve disorders. “Whether these associations reflect differences by sex in disease pathophysiology related to height may warrant exploration in a sample with better balance between men and women,” the authors wrote. “In sum, our results suggest that an individual’s height may warrant consideration as a nonmodifiable predictor for several common conditions, particularly those affecting peripheral/distal extremities that are most physically impacted by tall stature.”
A substantial limitation of the study was its homogeneity of participants, who were 92% male with an average height of 176 cm and an average BMI of 30.1. The Black participants tended to be younger, with an average age of 58 compared with 64 years in the White participants, but the groups were otherwise similar in height and weight.* The database included data from Hispanic participants, but the researchers excluded these data because of the small sample size.
The smaller dataset for Black participants was a limitation as well as the fact that the genome-wide association study the researchers relied on came from a European population, which may not be as accurate in people with other ancestry, Dr. Wilson said. The bigger limitation, however, is what the findings’ clinical relevance is.
What does it all mean?
“Genetic height is in your genes – there is nothing to be done about it – so it is more of academic interest than clinical interest,” Dr. Wilson said. It’s not even clear whether incorporating a person’s height – actual or genetically predicted, if it could be easily determined for each person – into risk calculators. ”To know whether it would be beneficial to use height (or genetic height) as a risk factor, you’d need to examine each condition of interest, adjusting for all known risk factors, to see if height improved the prediction,” Dr. Wilson said. “I suspect for most conditions, the well-known risk factors would swamp height. For example, high genetic height might truly increase risk for neuropathy. But diabetes might increase the risk so much more that height is not particularly relevant.”
On the other hand, the fact that height in general has any potential influence at all on disease risk may inspire physicians to consider other risk factors in especially tall individuals.
”Physicians may find it interesting that we have some confirmation that height does increase the risk of certain conditions,” Dr. Wilson said. “While this is unlikely to dramatically change practice, they may be a bit more diligent in looking for other relevant risk factors for the diseases found in this study in their very tall patients.”
The research was funded by the U.S. Department of Veteran Affairs, the Boettcher Foundation’s Webb-Waring Biomedical Research Program, the National Institutes of Health, and a Linda Pechenik Montague Investigator award. One study coauthor is a full-time employee of Novartis Institutes of Biomedical Research. The other authors and Dr. Wilson had no disclosures.
*Correction, 6/29/22: An earlier version of this article misstated the average age of Black participants.
, according to a study published in PLOS Genetics.
Prior studies have investigated height as a risk factor for chronic diseases, such as a higher risk for atrial fibrillation and a reduced risk of cardiovascular disease. It’s been consistently difficult, however, to eliminate the confounding influences of diet, socioeconomics, lifestyle behaviors, and other environmental factors that may interfere with a person’s reaching their expected height based on their genes.
This study, however, was able to better parse those differences by using Mendelian randomization within the comprehensive clinical and genetic dataset of a national health care system biobank. Mendelian randomization uses “genetic instruments for exposures of interest under the assumption that genotype is less susceptible to confounding than measured exposures,” the authors explained. The findings confirmed previously suspected associations between height and a range of cardiovascular and metabolic conditions as well as revealing new associations with several other conditions.
Prior associations confirmed, new associations uncovered
The results confirmed that being tall is linked to a higher risk of atrial fibrillation and varicose veins, and a lower risk of coronary heart disease, high blood pressure, and high cholesterol. The study also uncovered new associations between greater height and a higher risk of peripheral neuropathy, which is caused by damage to nerves on the extremities, as well as skin and bone infections, such as leg and foot ulcers.
The meta-analysis “identified five additional traits associated with genetically-predicted height,” wrote Sridharan Raghavan, MD, assistant professor of medicine at the University of Colorado Anschutz Medical Campus, and colleagues. “Two were genitourinary conditions – erectile dysfunction and urinary retention – that can be associated with neuropathy, and a third was a phecode for nonspecific skin disorders that may be related to skin infections – consistent with the race/ethnicity stratified results.”
Removing potential confounders
F. Perry Wilson, MD, associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study, said the findings were not particularly surprising overall, but it’s striking that the researchers had ”such a large cohort with such detailed electronic health records allowing for the comparison of genetic height with a variety of clinical outcomes.” He also noted the study’s strength in using Mendelian randomization so that the exposure is the predicted genetic height instead of a person’s measured height.
“This is key, since lots of things affect actual height – nutrition is an important one that could certainly be linked to disease as well,” Dr. Wilson said. ”By using genetic height, the authors remove these potential confounders. Since genetic height is “assigned” at birth (or conception), there is little opportunity for confounding. Of course, it is possible that some of the gene variants used to predict genetic height actually do something else, such as make you seek out less nutritious meals, but by and large this is how these types of studies need to be done.”
Height may impact over 100 clinical traits
The study relied on data from the U.S. Veteran Affairs Million Veteran Program with 222,300 non-Hispanic White and 58,151 non-Hispanic Black participants. The researchers first estimated the likelihood of participants’ genetic height based on 3,290 genetic variants determined to affect genetic height in a recent European-ancestry genome-wide meta-analysis. Then they compared these estimates with participants’ actual height in the VA medical record, adjusting for age, sex, and other genetic characteristics.
In doing so, the researchers found 345 clinical traits that were associated with the actual measured height in White participants plus another 17 clinical trials linked to actual measured height in Black participants. An overall 127 of these clinical traits were significantly associated with White participants’ genetically predicted height, and two of them were significantly associated with Black participants’ genetically predicted height.
In analyzing all these data together, the researchers were largely able to separate out those associations between genetically predicted height and certain health conditions from those associations between health conditions and a person’s actual measured height. They also determined that including body mass index as a covariate had little impact on the results. The researchers conducted the appropriate statistical correction to ensure the use of so many variables did not result in spurious statistical significance in some associations.
“Using genetic methods applied to the VA Million Veteran Program, we found evidence that adult height may impact over 100 clinical traits, including several conditions associated with poor outcomes and quality of life – peripheral neuropathy, lower extremity ulcers, and chronic venous insufficiency. We conclude that height may be an unrecognized nonmodifiable risk factor for several common conditions in adults.”
Height linked with health conditions
Genetically predicted height predicted a reduced risk of hyperlipidemia and hypertension independent of coronary heart disease, the analysis revealed. Genetically predicted height was also linked to an approximately 51% increased risk of atrial fibrillation in participants without coronary heart disease but, paradoxically, only a 39% increased risk in those with coronary heart disease, despite coronary heart disease being a risk factor for atrial fibrillation. Genetically predicted height was also associated with a greater risk of varicose veins in the legs and deep vein thrombosis.
Another novel association uncovered by the analysis was between women’s genetically predicted height and both asthma and nonspecific peripheral nerve disorders. “Whether these associations reflect differences by sex in disease pathophysiology related to height may warrant exploration in a sample with better balance between men and women,” the authors wrote. “In sum, our results suggest that an individual’s height may warrant consideration as a nonmodifiable predictor for several common conditions, particularly those affecting peripheral/distal extremities that are most physically impacted by tall stature.”
A substantial limitation of the study was its homogeneity of participants, who were 92% male with an average height of 176 cm and an average BMI of 30.1. The Black participants tended to be younger, with an average age of 58 compared with 64 years in the White participants, but the groups were otherwise similar in height and weight.* The database included data from Hispanic participants, but the researchers excluded these data because of the small sample size.
The smaller dataset for Black participants was a limitation as well as the fact that the genome-wide association study the researchers relied on came from a European population, which may not be as accurate in people with other ancestry, Dr. Wilson said. The bigger limitation, however, is what the findings’ clinical relevance is.
What does it all mean?
“Genetic height is in your genes – there is nothing to be done about it – so it is more of academic interest than clinical interest,” Dr. Wilson said. It’s not even clear whether incorporating a person’s height – actual or genetically predicted, if it could be easily determined for each person – into risk calculators. ”To know whether it would be beneficial to use height (or genetic height) as a risk factor, you’d need to examine each condition of interest, adjusting for all known risk factors, to see if height improved the prediction,” Dr. Wilson said. “I suspect for most conditions, the well-known risk factors would swamp height. For example, high genetic height might truly increase risk for neuropathy. But diabetes might increase the risk so much more that height is not particularly relevant.”
On the other hand, the fact that height in general has any potential influence at all on disease risk may inspire physicians to consider other risk factors in especially tall individuals.
”Physicians may find it interesting that we have some confirmation that height does increase the risk of certain conditions,” Dr. Wilson said. “While this is unlikely to dramatically change practice, they may be a bit more diligent in looking for other relevant risk factors for the diseases found in this study in their very tall patients.”
The research was funded by the U.S. Department of Veteran Affairs, the Boettcher Foundation’s Webb-Waring Biomedical Research Program, the National Institutes of Health, and a Linda Pechenik Montague Investigator award. One study coauthor is a full-time employee of Novartis Institutes of Biomedical Research. The other authors and Dr. Wilson had no disclosures.
*Correction, 6/29/22: An earlier version of this article misstated the average age of Black participants.
FROM PLOS GENETICS
Lupus mutation may unlock targeted drugs for patient subset
Scientists have confirmed that a receptor long suspected to be linked to lupus is, in fact, a major driver of the autoimmune disease for at least some subset of patients, according to a study recently published in Nature. Researchers discovered the crucial role of toll-like receptor 7 (TLR7) because of a rare mutation in a pediatric patient with systemic lupus erythematosus (SLE) who had a particularly severe presentation.
“Sometimes it’s valuable to find these very severe cases where there is one mutation that has a strong effect because if we understand how those mutations work, the lessons we learn can generally tell us about disease mechanisms,” explained senior author Carola G. Vinuesa, MD, PhD, of the Centre for Personalised Immunology at Australian National University in Canberra and The Francis Crick Institute in London.

“It’s quite difficult to find one mutation that can alone cause the entire disease,” Dr. Vinuesa added, but what it reveals about how the disease develops may lead to more effective targeted therapies than the immune suppressants most often used to treat lupus currently.
The mutation they found was in the TLR7 gene that encodes the TLR7 protein. TLR7 is a receptor used by immune cells to identify viral RNA so they can fight off viral infections, including COVID-19. But if the body’s own genetic material binds to TLR7 in susceptible individuals, it can lead to an overproduction of type 1 interferons, which are cytokines that trigger or exacerbate the immune reactions that lead to lupus symptoms. The TLR7 gene occurs on the X chromosome, which may explain men’s greater susceptibility to COVID-19 and the greater incidence of lupus in women, who have two X chromosomes instead of the one that men have, Dr. Vinuesa said.
Previous research had shown an association between TLR7 and lupus, but this new study is the first to provide definitive proof that a TLR7 mutation by itself can directly cause human lupus. After discovering the variant in the patient, Dr. Vinuesa’s team used CRISPR to edit the genome of a mouse model and introduce the same mutation the patient had. “And they developed full-blown disease, just with this one single base-pair substitution – 1 letter in the 3 billion letters of the genome,” Dr. Vinuesa said. “It tells us that these receptors are not just there to recognize viral RNA, that in some circumstances, they could be triggered by our own nucleic acids.”
One pathway among many?
The finding does not mean that every lupus patient has this mutation, which remains rare, but suggests that overactivity in this receptor already reported in many lupus patients may be causally related to disease, Dr. Vinuesa said.
Noa Schwartz, MD, an assistant professor of medicine at Albert Einstein College of Medicine, New York, and director of the Montefiore-Einstein Institute for Lupus Care and Research, said in an interview that lupus is thought of as a syndrome, a collection of different but similar diseases that don’t necessarily have a single cause. But finding a single gene mutation that could potentially lead to lupus is an important piece of the puzzle, said Dr. Schwartz, who was not involved in the study. Based on past research in mice models, “we’ve hypothesized that TLR7 is important in humans as well, but this is the last nail in the coffin.”
One of the key questions this finding has prompted is how many patients’ disease results from TLR7 activity. “Because of the evidence from Ignacio Sanz’s group demonstrating TLR7 overactivity in a significant fraction of SLE patients, we believe that it is probably going to be pretty important,” Dr. Vinuesa said. “My feeling is that it is going to be quite a central pathway in lupus pathogenesis, if not the central pathway.”
Dr. Schwartz was more cautious, noting that it is probably important for a subset of patients but may “have a limited effect on the general lupus population.” While it’s not yet clear how large that subset is, it is possible it will include people with cutaneous lupus, those with primarily dermatologic symptoms.
“Hydroxychloroquine works particularly well for cutaneous manifestations of lupus, and one of the ways that works is by inhibiting TLR7 and TLR9, so this [finding] potentially matters for skin disease and lupus, but it’s very early,” Dr. Schwartz said. If it does turn out that TLR7 activity is particularly associated with cutaneous lupus, it may mean therapies with fewer side effects, she said. “Specifically for cutaneous lupus, the concept of suppressing the entire immune system for skin illness sometimes feels, especially to patients, very extreme, so they are [patients] who directed therapy could be so especially relevant for.”
Laura Lewandowski, MD, an assistant clinical investigator and head of the lupus genomics and global health disparities unit at the National Institute of Arthritis and Musculoskeletal and Skin Disease, described this study as particularly remarkable in the way it revealed the mechanism leading to lupus symptoms.
“As whole genome sequencing becomes faster and less expensive, more and more people are employing them in their studies,” most of which report changes in certain genes, Dr. Lewandowski said. “One of the most striking findings about this paper was that they took it to the next step and did a really elegant study on the exact way this gain-of-function TLR7 mutation leads to the autoimmunity that we see in lupus. The detail of mechanism in this paper is really unique.”
A step toward personalized medicine
Dr. Lewandowski is part of a team that recently presented a poster related to genomic sequencing in lupus patients at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance. Her study reported on the whole genome sequencing of patients with childhood-onset SLE who were already enrolled in the CARRA Lupus Registry. Children with lupus may be more likely than adults to have rare genetic variants, so a registry of childhood-onset SLE patients with fully sequenced genomes provides an opportunity to look for single-gene mutations specifically linked to lupus, said Dr. Lewandowski, who has recently begun a research collaboration with Dr. Vinuesa.
“As we move forward and more and more patients are included in these studies, we will understand a little bit more about the genetic architecture of patients who have rare variations leading to disease, or even common variations,” Dr. Lewandowski said about the intersection between her research and Dr. Vinuesa’s study. The more data they gather, the more they can explore the possible interactions of rare and common variants that play a role in SLE as well as what environmental triggers, such as viral infection or pollution exposure, might tip someone into having an autoimmune disease. “We’re just starting to peek under the hood,” Dr. Lewandowski said.
If further research can reveal the relative contribution of genetics to the disease and what those genetic drivers are, it may allow for greater precision in therapies and “ultimately improve the quality of life for our patients, the ultimate goal of all of these studies,” Dr. Lewandowski said.
Drugs that target TLR7 already exist for other indications, and clinical trials have already begun to see if these TLR7 inhibitors benefit lupus patients.
“If the clinical trials work, this will be quite a nice, targeted therapy with potentially much less side effects than other therapies on the market at the moment,” Dr. Vinuesa said. She is cautiously hopeful, saying it’s likely to make an impact on lupus treatment, but it’s too early to say precisely how much.
“It allows us to understand the disease mechanisms a little bit better and to try and assess what percentage of patients’ disease can be explained by overactivity in this receptor,” Dr. Vinuesa said. She thinks it’s possible that TLR7 over activation may be relevant to other systemic autoimmune diseases as well, such as Sjögren’s syndrome, rheumatoid arthritis, or juvenile dermatomyositis, but it will take more studies to find out.
“Right now, we have medicines that broadly inhibit the immune system and aren’t as targeted, but we have a lot more clinical and scientific work to do before we move this field forward for lupus patients,” Dr. Lewandowski said. “This is one case where they were able to find the exact molecular defect, and it’s not the end of the path of precision medicine — it’s the beginning.”
Dr. Vinuesa, Dr. Schwartz, and Dr. Lewandowski reported no disclosures.
A version of this article first appeared on Medscape.com.
Scientists have confirmed that a receptor long suspected to be linked to lupus is, in fact, a major driver of the autoimmune disease for at least some subset of patients, according to a study recently published in Nature. Researchers discovered the crucial role of toll-like receptor 7 (TLR7) because of a rare mutation in a pediatric patient with systemic lupus erythematosus (SLE) who had a particularly severe presentation.
“Sometimes it’s valuable to find these very severe cases where there is one mutation that has a strong effect because if we understand how those mutations work, the lessons we learn can generally tell us about disease mechanisms,” explained senior author Carola G. Vinuesa, MD, PhD, of the Centre for Personalised Immunology at Australian National University in Canberra and The Francis Crick Institute in London.

“It’s quite difficult to find one mutation that can alone cause the entire disease,” Dr. Vinuesa added, but what it reveals about how the disease develops may lead to more effective targeted therapies than the immune suppressants most often used to treat lupus currently.
The mutation they found was in the TLR7 gene that encodes the TLR7 protein. TLR7 is a receptor used by immune cells to identify viral RNA so they can fight off viral infections, including COVID-19. But if the body’s own genetic material binds to TLR7 in susceptible individuals, it can lead to an overproduction of type 1 interferons, which are cytokines that trigger or exacerbate the immune reactions that lead to lupus symptoms. The TLR7 gene occurs on the X chromosome, which may explain men’s greater susceptibility to COVID-19 and the greater incidence of lupus in women, who have two X chromosomes instead of the one that men have, Dr. Vinuesa said.
Previous research had shown an association between TLR7 and lupus, but this new study is the first to provide definitive proof that a TLR7 mutation by itself can directly cause human lupus. After discovering the variant in the patient, Dr. Vinuesa’s team used CRISPR to edit the genome of a mouse model and introduce the same mutation the patient had. “And they developed full-blown disease, just with this one single base-pair substitution – 1 letter in the 3 billion letters of the genome,” Dr. Vinuesa said. “It tells us that these receptors are not just there to recognize viral RNA, that in some circumstances, they could be triggered by our own nucleic acids.”
One pathway among many?
The finding does not mean that every lupus patient has this mutation, which remains rare, but suggests that overactivity in this receptor already reported in many lupus patients may be causally related to disease, Dr. Vinuesa said.
Noa Schwartz, MD, an assistant professor of medicine at Albert Einstein College of Medicine, New York, and director of the Montefiore-Einstein Institute for Lupus Care and Research, said in an interview that lupus is thought of as a syndrome, a collection of different but similar diseases that don’t necessarily have a single cause. But finding a single gene mutation that could potentially lead to lupus is an important piece of the puzzle, said Dr. Schwartz, who was not involved in the study. Based on past research in mice models, “we’ve hypothesized that TLR7 is important in humans as well, but this is the last nail in the coffin.”
One of the key questions this finding has prompted is how many patients’ disease results from TLR7 activity. “Because of the evidence from Ignacio Sanz’s group demonstrating TLR7 overactivity in a significant fraction of SLE patients, we believe that it is probably going to be pretty important,” Dr. Vinuesa said. “My feeling is that it is going to be quite a central pathway in lupus pathogenesis, if not the central pathway.”
Dr. Schwartz was more cautious, noting that it is probably important for a subset of patients but may “have a limited effect on the general lupus population.” While it’s not yet clear how large that subset is, it is possible it will include people with cutaneous lupus, those with primarily dermatologic symptoms.
“Hydroxychloroquine works particularly well for cutaneous manifestations of lupus, and one of the ways that works is by inhibiting TLR7 and TLR9, so this [finding] potentially matters for skin disease and lupus, but it’s very early,” Dr. Schwartz said. If it does turn out that TLR7 activity is particularly associated with cutaneous lupus, it may mean therapies with fewer side effects, she said. “Specifically for cutaneous lupus, the concept of suppressing the entire immune system for skin illness sometimes feels, especially to patients, very extreme, so they are [patients] who directed therapy could be so especially relevant for.”
Laura Lewandowski, MD, an assistant clinical investigator and head of the lupus genomics and global health disparities unit at the National Institute of Arthritis and Musculoskeletal and Skin Disease, described this study as particularly remarkable in the way it revealed the mechanism leading to lupus symptoms.
“As whole genome sequencing becomes faster and less expensive, more and more people are employing them in their studies,” most of which report changes in certain genes, Dr. Lewandowski said. “One of the most striking findings about this paper was that they took it to the next step and did a really elegant study on the exact way this gain-of-function TLR7 mutation leads to the autoimmunity that we see in lupus. The detail of mechanism in this paper is really unique.”
A step toward personalized medicine
Dr. Lewandowski is part of a team that recently presented a poster related to genomic sequencing in lupus patients at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance. Her study reported on the whole genome sequencing of patients with childhood-onset SLE who were already enrolled in the CARRA Lupus Registry. Children with lupus may be more likely than adults to have rare genetic variants, so a registry of childhood-onset SLE patients with fully sequenced genomes provides an opportunity to look for single-gene mutations specifically linked to lupus, said Dr. Lewandowski, who has recently begun a research collaboration with Dr. Vinuesa.
“As we move forward and more and more patients are included in these studies, we will understand a little bit more about the genetic architecture of patients who have rare variations leading to disease, or even common variations,” Dr. Lewandowski said about the intersection between her research and Dr. Vinuesa’s study. The more data they gather, the more they can explore the possible interactions of rare and common variants that play a role in SLE as well as what environmental triggers, such as viral infection or pollution exposure, might tip someone into having an autoimmune disease. “We’re just starting to peek under the hood,” Dr. Lewandowski said.
If further research can reveal the relative contribution of genetics to the disease and what those genetic drivers are, it may allow for greater precision in therapies and “ultimately improve the quality of life for our patients, the ultimate goal of all of these studies,” Dr. Lewandowski said.
Drugs that target TLR7 already exist for other indications, and clinical trials have already begun to see if these TLR7 inhibitors benefit lupus patients.
“If the clinical trials work, this will be quite a nice, targeted therapy with potentially much less side effects than other therapies on the market at the moment,” Dr. Vinuesa said. She is cautiously hopeful, saying it’s likely to make an impact on lupus treatment, but it’s too early to say precisely how much.
“It allows us to understand the disease mechanisms a little bit better and to try and assess what percentage of patients’ disease can be explained by overactivity in this receptor,” Dr. Vinuesa said. She thinks it’s possible that TLR7 over activation may be relevant to other systemic autoimmune diseases as well, such as Sjögren’s syndrome, rheumatoid arthritis, or juvenile dermatomyositis, but it will take more studies to find out.
“Right now, we have medicines that broadly inhibit the immune system and aren’t as targeted, but we have a lot more clinical and scientific work to do before we move this field forward for lupus patients,” Dr. Lewandowski said. “This is one case where they were able to find the exact molecular defect, and it’s not the end of the path of precision medicine — it’s the beginning.”
Dr. Vinuesa, Dr. Schwartz, and Dr. Lewandowski reported no disclosures.
A version of this article first appeared on Medscape.com.
Scientists have confirmed that a receptor long suspected to be linked to lupus is, in fact, a major driver of the autoimmune disease for at least some subset of patients, according to a study recently published in Nature. Researchers discovered the crucial role of toll-like receptor 7 (TLR7) because of a rare mutation in a pediatric patient with systemic lupus erythematosus (SLE) who had a particularly severe presentation.
“Sometimes it’s valuable to find these very severe cases where there is one mutation that has a strong effect because if we understand how those mutations work, the lessons we learn can generally tell us about disease mechanisms,” explained senior author Carola G. Vinuesa, MD, PhD, of the Centre for Personalised Immunology at Australian National University in Canberra and The Francis Crick Institute in London.

“It’s quite difficult to find one mutation that can alone cause the entire disease,” Dr. Vinuesa added, but what it reveals about how the disease develops may lead to more effective targeted therapies than the immune suppressants most often used to treat lupus currently.
The mutation they found was in the TLR7 gene that encodes the TLR7 protein. TLR7 is a receptor used by immune cells to identify viral RNA so they can fight off viral infections, including COVID-19. But if the body’s own genetic material binds to TLR7 in susceptible individuals, it can lead to an overproduction of type 1 interferons, which are cytokines that trigger or exacerbate the immune reactions that lead to lupus symptoms. The TLR7 gene occurs on the X chromosome, which may explain men’s greater susceptibility to COVID-19 and the greater incidence of lupus in women, who have two X chromosomes instead of the one that men have, Dr. Vinuesa said.
Previous research had shown an association between TLR7 and lupus, but this new study is the first to provide definitive proof that a TLR7 mutation by itself can directly cause human lupus. After discovering the variant in the patient, Dr. Vinuesa’s team used CRISPR to edit the genome of a mouse model and introduce the same mutation the patient had. “And they developed full-blown disease, just with this one single base-pair substitution – 1 letter in the 3 billion letters of the genome,” Dr. Vinuesa said. “It tells us that these receptors are not just there to recognize viral RNA, that in some circumstances, they could be triggered by our own nucleic acids.”
One pathway among many?
The finding does not mean that every lupus patient has this mutation, which remains rare, but suggests that overactivity in this receptor already reported in many lupus patients may be causally related to disease, Dr. Vinuesa said.
Noa Schwartz, MD, an assistant professor of medicine at Albert Einstein College of Medicine, New York, and director of the Montefiore-Einstein Institute for Lupus Care and Research, said in an interview that lupus is thought of as a syndrome, a collection of different but similar diseases that don’t necessarily have a single cause. But finding a single gene mutation that could potentially lead to lupus is an important piece of the puzzle, said Dr. Schwartz, who was not involved in the study. Based on past research in mice models, “we’ve hypothesized that TLR7 is important in humans as well, but this is the last nail in the coffin.”
One of the key questions this finding has prompted is how many patients’ disease results from TLR7 activity. “Because of the evidence from Ignacio Sanz’s group demonstrating TLR7 overactivity in a significant fraction of SLE patients, we believe that it is probably going to be pretty important,” Dr. Vinuesa said. “My feeling is that it is going to be quite a central pathway in lupus pathogenesis, if not the central pathway.”
Dr. Schwartz was more cautious, noting that it is probably important for a subset of patients but may “have a limited effect on the general lupus population.” While it’s not yet clear how large that subset is, it is possible it will include people with cutaneous lupus, those with primarily dermatologic symptoms.
“Hydroxychloroquine works particularly well for cutaneous manifestations of lupus, and one of the ways that works is by inhibiting TLR7 and TLR9, so this [finding] potentially matters for skin disease and lupus, but it’s very early,” Dr. Schwartz said. If it does turn out that TLR7 activity is particularly associated with cutaneous lupus, it may mean therapies with fewer side effects, she said. “Specifically for cutaneous lupus, the concept of suppressing the entire immune system for skin illness sometimes feels, especially to patients, very extreme, so they are [patients] who directed therapy could be so especially relevant for.”
Laura Lewandowski, MD, an assistant clinical investigator and head of the lupus genomics and global health disparities unit at the National Institute of Arthritis and Musculoskeletal and Skin Disease, described this study as particularly remarkable in the way it revealed the mechanism leading to lupus symptoms.
“As whole genome sequencing becomes faster and less expensive, more and more people are employing them in their studies,” most of which report changes in certain genes, Dr. Lewandowski said. “One of the most striking findings about this paper was that they took it to the next step and did a really elegant study on the exact way this gain-of-function TLR7 mutation leads to the autoimmunity that we see in lupus. The detail of mechanism in this paper is really unique.”
A step toward personalized medicine
Dr. Lewandowski is part of a team that recently presented a poster related to genomic sequencing in lupus patients at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance. Her study reported on the whole genome sequencing of patients with childhood-onset SLE who were already enrolled in the CARRA Lupus Registry. Children with lupus may be more likely than adults to have rare genetic variants, so a registry of childhood-onset SLE patients with fully sequenced genomes provides an opportunity to look for single-gene mutations specifically linked to lupus, said Dr. Lewandowski, who has recently begun a research collaboration with Dr. Vinuesa.
“As we move forward and more and more patients are included in these studies, we will understand a little bit more about the genetic architecture of patients who have rare variations leading to disease, or even common variations,” Dr. Lewandowski said about the intersection between her research and Dr. Vinuesa’s study. The more data they gather, the more they can explore the possible interactions of rare and common variants that play a role in SLE as well as what environmental triggers, such as viral infection or pollution exposure, might tip someone into having an autoimmune disease. “We’re just starting to peek under the hood,” Dr. Lewandowski said.
If further research can reveal the relative contribution of genetics to the disease and what those genetic drivers are, it may allow for greater precision in therapies and “ultimately improve the quality of life for our patients, the ultimate goal of all of these studies,” Dr. Lewandowski said.
Drugs that target TLR7 already exist for other indications, and clinical trials have already begun to see if these TLR7 inhibitors benefit lupus patients.
“If the clinical trials work, this will be quite a nice, targeted therapy with potentially much less side effects than other therapies on the market at the moment,” Dr. Vinuesa said. She is cautiously hopeful, saying it’s likely to make an impact on lupus treatment, but it’s too early to say precisely how much.
“It allows us to understand the disease mechanisms a little bit better and to try and assess what percentage of patients’ disease can be explained by overactivity in this receptor,” Dr. Vinuesa said. She thinks it’s possible that TLR7 over activation may be relevant to other systemic autoimmune diseases as well, such as Sjögren’s syndrome, rheumatoid arthritis, or juvenile dermatomyositis, but it will take more studies to find out.
“Right now, we have medicines that broadly inhibit the immune system and aren’t as targeted, but we have a lot more clinical and scientific work to do before we move this field forward for lupus patients,” Dr. Lewandowski said. “This is one case where they were able to find the exact molecular defect, and it’s not the end of the path of precision medicine — it’s the beginning.”
Dr. Vinuesa, Dr. Schwartz, and Dr. Lewandowski reported no disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE
Pfizer COVID vaccine performs well in youth with rheumatic diseases
The Pfizer-BioNTech mRNA vaccine (Comirnaty) showed a good safety profile with minimal short-term side effects and no negative impact on disease activity in a cohort of adolescents and young adults with rheumatic diseases, according to research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year.
Only 3% of patients experience a severe transient adverse event, according to Merav Heshin-Bekenstein, MD, of Dana-Dwek Children’s Hospital at the Tel Aviv Sourasky Medical Center in Israel. The findings were published in Rheumatology.
“We found that the mRNA Pfizer vaccine was immunogenic and induced an adequate humoral immune response in adolescent patients,” Dr. Heshin-Bekenstein told CARRA attendees. “It was definitely comparable to healthy controls and practically all patients were seropositive following the second vaccine, except for one patient with long-standing systemic sclerosis.”
The findings were not necessarily surprising but were encouraging to Melissa S. Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University, Indianapolis. Dr. Oliver wasn’t part of the study team.
“We know that the COVID vaccines in healthy adolescents have shown good efficacy with minimal side effects, and it’s good to see that this study showed that in those with rheumatic diseases on immunosuppressive therapy,” Dr. Oliver told this news organization.
Until now, the data on COVID-19 vaccines in teens with rheumatic illnesses has been limited, she said, so “many pediatric rheumatologists only have the data from adult studies to go on or personal experience with their own cohort of patients.”
But the high immunogenicity seen in the study was a pleasant surprise to Beth H. Rutstein, MD, assistant professor of clinical pediatrics in the division of rheumatology at Children’s Hospital of Philadelphia and the Perelman School of Medicine at the University of Pennsylvania.
“I was both surprised and thrilled with Dr. Heshin-Bekenstein’s findings suggesting near-universal seroconversion for patients with rheumatic disease regardless of underlying diagnosis or immunomodulatory therapy regimen, as much of the adult data has suggested a poorer seroconversion rate” and lower antibody titers in adults with similar illnesses, Dr. Rutstein said in an interview.
The study “provides essential reassurance that vaccination against COVID-19 does not increase the risk of disease flare or worsen disease severity scores,” said Dr. Rutstein, who was not associated with the research. “Rather than speaking purely anecdotally with our patients and their families, we can refer to the science – which is always more reassuring for both our patients and ourselves.”
Study included diverse conditions and therapies
Risk factors for poor outcomes with COVID-19 in children include obesity, cardiovascular disease, chronic lung disease, diabetes, and asthma, Dr. Heshin-Bekenstein told CARRA attendees. Multisystem inflammatory syndrome in children (MIS-C) and long COVID are also potential complications of COVID-19 with less understood risk factors.
Although COVID-19 is most often mild in children, certain severe, systemic rheumatic diseases increase hospitalization risk, including systemic lupus erythematosus (SLE) and vasculitis. Evidence has also shown that COVID-19 infection increases the risk of disease flare in teens with juvenile-onset rheumatic diseases, so it’s “crucial to prevent COVID-19 disease in this population,” Dr. Heshin-Bekenstein said.
Her study therefore aimed to assess the safety and immunogenicity of the Pfizer mRNA vaccine for teens with juvenile-onset rheumatic diseases and those taking immunomodulatory medications. The international prospective multicenter study ran from April to November 2021 at three pediatric rheumatology clinics in Israel and one in Slovenia. Endpoints included short-term side effects, vaccination impact on clinical disease activity, immunogenicity at 2-9 weeks after the second dose, and, secondarily, efficacy against COVID-19 infection.
The 91 participants included adolescents aged 12-18 and young adults aged 18-21. Nearly half of the participants (46%) had juvenile idiopathic arthritis (JIA), and 14% had SLE. Other participants’ conditions included systemic vasculitis, idiopathic uveitis, inflammatory bowel disease–related arthritis, systemic or localized scleroderma, juvenile dermatomyositis, or an autoinflammatory disease. Participants’ mean disease duration was 4.8 years.
The researchers compared the patients with a control group of 40 individuals with similar demographics but without rheumatic disease. The researchers used the LIAISON quantitative assay to assess serum IgG antibody levels against the SARS-CoV-2 spike protein in both groups.
Eight in 10 participants with rheumatic disease were taking an immunomodulatory medication, including a conventional synthetic disease-modifying antirheumatic drug (csDMARD) in 40%, a biologic DMARD in 37%, tumor necrosis factor (TNF) inhibitors in 32%, hydroxychloroquine (HCQ) in 19%, glucocorticoids in 14%, and mycophenolate in 11%. A smaller proportion were on other biologics: JAK inhibitors in 6.6%, anti-CD20 drugs in 4.4%, and an IL-6 inhibitor in 1%.
Side effects similar in both groups
None of the side effects reported by participants were statistically different between those with rheumatic disease and the control group. Localized pain was the most common side effect, reported by 73%-79% of participants after each dose. About twice as many participants with rheumatic disease experienced muscle aches and joint pains, compared with the control group, but the differences were not significant. Fever occurred more often in those with rheumatic disease (6%, five cases) than without (3%, one case). One-third of those with rheumatic disease felt tiredness, compared with 20% of the control group.
None of the healthy controls were hospitalized after vaccination, but three rheumatic patients were, including two after the first dose. Both were 17 years old, had systemic vasculitis with granulomatosis with polyangiitis (GPA), and were taking rituximab (Rituxan). One patient experienced acute onset of chronic renal failure, fever, dehydration, and high C-reactive protein within hours of vaccination. The other experienced new onset of pulmonary hemorrhage a week after vaccination.
In addition, a 14-year-old female with lupus, taking only HCQ, went to the emergency department with fever, headache, vomiting, and joint pain 1 day after the second vaccine dose. She had normal inflammatory markers and no change in disease activity score, and she was discharged with low-dose steroids tapered after 2 weeks.
Immune response high in patients with rheumatic disease
Immunogenicity was similar in both groups, with 97% seropositivity in the rheumatic disease group and 100% in the control group. Average IgG titers were 242 in the rheumatic group and 388 in the control group (P < .0001). Seropositivity was 88% in those taking mycophenolate with another drug (100% with mycophenolate monotherapy), 90% with HCQ, 94% with any csDMARDs and another drug (100% with csDMARD monotherapy), and 100% for all other drugs. During 3 months’ follow-up after vaccination, there were no COVID-19 cases among the participants.
Dr. Heshin-Bekenstein noted that their results showed better immunogenicity in teens, compared with adults, for two specific drugs. Seropositivity in teens taking methotrexate (Rheumatrex, Trexall) or rituximab was 100% in this study, compared with 84% in adults taking methotrexate and 39% in adults taking rituximab in a previous study. However, only three patients in this study were taking rituximab, and only seven were taking methotrexate.
The study’s heterogenous population was both a strength and a weakness of the study. “Due to the diversity of rheumatic diseases and medications included in this cohort, it was not possible to draw significant conclusions regarding the impact of the immunomodulatory medications and type of disease” on titers, Dr. Heshin-Bekenstein told attendees.
Still, “I think as pediatric rheumatologists, we can feel reassured in recommending the COVID-19 vaccine to our patients,” Dr. Oliver said. “I will add that every patient is different, and everyone should have a conversation with their physician about receiving the COVID-19 vaccine.” Dr. Oliver said she discusses vaccination, including COVID vaccination, with every patient, and it’s been challenging to address concerns in the midst of so much misinformation circulating about the vaccine.
These findings do raise questions about whether it’s still necessary to hold immunomodulatory medications to get the vaccine,” Dr. Rutstein said.
“Many families are nervous to pause their medications before and after the vaccine as is currently recommended for many therapies by the American College of Rheumatology, and I do share that concern for some of my patients with more clinically unstable disease, so I try to work with each family to decide on best timing and have delayed or deferred the series until some patients are on a steady dose of a new immunomodulatory medication if it has been recently started,” Dr. Rutstein said. “This is one of the reasons why Dr. Heshin-Bekenstein’s study is so important – we may be holding medications that can be safely continued and even further decrease the risk of disease flare.”
None of the physicians have disclosed any relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Pfizer-BioNTech mRNA vaccine (Comirnaty) showed a good safety profile with minimal short-term side effects and no negative impact on disease activity in a cohort of adolescents and young adults with rheumatic diseases, according to research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year.
Only 3% of patients experience a severe transient adverse event, according to Merav Heshin-Bekenstein, MD, of Dana-Dwek Children’s Hospital at the Tel Aviv Sourasky Medical Center in Israel. The findings were published in Rheumatology.
“We found that the mRNA Pfizer vaccine was immunogenic and induced an adequate humoral immune response in adolescent patients,” Dr. Heshin-Bekenstein told CARRA attendees. “It was definitely comparable to healthy controls and practically all patients were seropositive following the second vaccine, except for one patient with long-standing systemic sclerosis.”
The findings were not necessarily surprising but were encouraging to Melissa S. Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University, Indianapolis. Dr. Oliver wasn’t part of the study team.
“We know that the COVID vaccines in healthy adolescents have shown good efficacy with minimal side effects, and it’s good to see that this study showed that in those with rheumatic diseases on immunosuppressive therapy,” Dr. Oliver told this news organization.
Until now, the data on COVID-19 vaccines in teens with rheumatic illnesses has been limited, she said, so “many pediatric rheumatologists only have the data from adult studies to go on or personal experience with their own cohort of patients.”
But the high immunogenicity seen in the study was a pleasant surprise to Beth H. Rutstein, MD, assistant professor of clinical pediatrics in the division of rheumatology at Children’s Hospital of Philadelphia and the Perelman School of Medicine at the University of Pennsylvania.
“I was both surprised and thrilled with Dr. Heshin-Bekenstein’s findings suggesting near-universal seroconversion for patients with rheumatic disease regardless of underlying diagnosis or immunomodulatory therapy regimen, as much of the adult data has suggested a poorer seroconversion rate” and lower antibody titers in adults with similar illnesses, Dr. Rutstein said in an interview.
The study “provides essential reassurance that vaccination against COVID-19 does not increase the risk of disease flare or worsen disease severity scores,” said Dr. Rutstein, who was not associated with the research. “Rather than speaking purely anecdotally with our patients and their families, we can refer to the science – which is always more reassuring for both our patients and ourselves.”
Study included diverse conditions and therapies
Risk factors for poor outcomes with COVID-19 in children include obesity, cardiovascular disease, chronic lung disease, diabetes, and asthma, Dr. Heshin-Bekenstein told CARRA attendees. Multisystem inflammatory syndrome in children (MIS-C) and long COVID are also potential complications of COVID-19 with less understood risk factors.
Although COVID-19 is most often mild in children, certain severe, systemic rheumatic diseases increase hospitalization risk, including systemic lupus erythematosus (SLE) and vasculitis. Evidence has also shown that COVID-19 infection increases the risk of disease flare in teens with juvenile-onset rheumatic diseases, so it’s “crucial to prevent COVID-19 disease in this population,” Dr. Heshin-Bekenstein said.
Her study therefore aimed to assess the safety and immunogenicity of the Pfizer mRNA vaccine for teens with juvenile-onset rheumatic diseases and those taking immunomodulatory medications. The international prospective multicenter study ran from April to November 2021 at three pediatric rheumatology clinics in Israel and one in Slovenia. Endpoints included short-term side effects, vaccination impact on clinical disease activity, immunogenicity at 2-9 weeks after the second dose, and, secondarily, efficacy against COVID-19 infection.
The 91 participants included adolescents aged 12-18 and young adults aged 18-21. Nearly half of the participants (46%) had juvenile idiopathic arthritis (JIA), and 14% had SLE. Other participants’ conditions included systemic vasculitis, idiopathic uveitis, inflammatory bowel disease–related arthritis, systemic or localized scleroderma, juvenile dermatomyositis, or an autoinflammatory disease. Participants’ mean disease duration was 4.8 years.
The researchers compared the patients with a control group of 40 individuals with similar demographics but without rheumatic disease. The researchers used the LIAISON quantitative assay to assess serum IgG antibody levels against the SARS-CoV-2 spike protein in both groups.
Eight in 10 participants with rheumatic disease were taking an immunomodulatory medication, including a conventional synthetic disease-modifying antirheumatic drug (csDMARD) in 40%, a biologic DMARD in 37%, tumor necrosis factor (TNF) inhibitors in 32%, hydroxychloroquine (HCQ) in 19%, glucocorticoids in 14%, and mycophenolate in 11%. A smaller proportion were on other biologics: JAK inhibitors in 6.6%, anti-CD20 drugs in 4.4%, and an IL-6 inhibitor in 1%.
Side effects similar in both groups
None of the side effects reported by participants were statistically different between those with rheumatic disease and the control group. Localized pain was the most common side effect, reported by 73%-79% of participants after each dose. About twice as many participants with rheumatic disease experienced muscle aches and joint pains, compared with the control group, but the differences were not significant. Fever occurred more often in those with rheumatic disease (6%, five cases) than without (3%, one case). One-third of those with rheumatic disease felt tiredness, compared with 20% of the control group.
None of the healthy controls were hospitalized after vaccination, but three rheumatic patients were, including two after the first dose. Both were 17 years old, had systemic vasculitis with granulomatosis with polyangiitis (GPA), and were taking rituximab (Rituxan). One patient experienced acute onset of chronic renal failure, fever, dehydration, and high C-reactive protein within hours of vaccination. The other experienced new onset of pulmonary hemorrhage a week after vaccination.
In addition, a 14-year-old female with lupus, taking only HCQ, went to the emergency department with fever, headache, vomiting, and joint pain 1 day after the second vaccine dose. She had normal inflammatory markers and no change in disease activity score, and she was discharged with low-dose steroids tapered after 2 weeks.
Immune response high in patients with rheumatic disease
Immunogenicity was similar in both groups, with 97% seropositivity in the rheumatic disease group and 100% in the control group. Average IgG titers were 242 in the rheumatic group and 388 in the control group (P < .0001). Seropositivity was 88% in those taking mycophenolate with another drug (100% with mycophenolate monotherapy), 90% with HCQ, 94% with any csDMARDs and another drug (100% with csDMARD monotherapy), and 100% for all other drugs. During 3 months’ follow-up after vaccination, there were no COVID-19 cases among the participants.
Dr. Heshin-Bekenstein noted that their results showed better immunogenicity in teens, compared with adults, for two specific drugs. Seropositivity in teens taking methotrexate (Rheumatrex, Trexall) or rituximab was 100% in this study, compared with 84% in adults taking methotrexate and 39% in adults taking rituximab in a previous study. However, only three patients in this study were taking rituximab, and only seven were taking methotrexate.
The study’s heterogenous population was both a strength and a weakness of the study. “Due to the diversity of rheumatic diseases and medications included in this cohort, it was not possible to draw significant conclusions regarding the impact of the immunomodulatory medications and type of disease” on titers, Dr. Heshin-Bekenstein told attendees.
Still, “I think as pediatric rheumatologists, we can feel reassured in recommending the COVID-19 vaccine to our patients,” Dr. Oliver said. “I will add that every patient is different, and everyone should have a conversation with their physician about receiving the COVID-19 vaccine.” Dr. Oliver said she discusses vaccination, including COVID vaccination, with every patient, and it’s been challenging to address concerns in the midst of so much misinformation circulating about the vaccine.
These findings do raise questions about whether it’s still necessary to hold immunomodulatory medications to get the vaccine,” Dr. Rutstein said.
“Many families are nervous to pause their medications before and after the vaccine as is currently recommended for many therapies by the American College of Rheumatology, and I do share that concern for some of my patients with more clinically unstable disease, so I try to work with each family to decide on best timing and have delayed or deferred the series until some patients are on a steady dose of a new immunomodulatory medication if it has been recently started,” Dr. Rutstein said. “This is one of the reasons why Dr. Heshin-Bekenstein’s study is so important – we may be holding medications that can be safely continued and even further decrease the risk of disease flare.”
None of the physicians have disclosed any relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Pfizer-BioNTech mRNA vaccine (Comirnaty) showed a good safety profile with minimal short-term side effects and no negative impact on disease activity in a cohort of adolescents and young adults with rheumatic diseases, according to research presented at the annual scientific meeting of the Childhood Arthritis and Rheumatology Research Alliance, held virtually this year.
Only 3% of patients experience a severe transient adverse event, according to Merav Heshin-Bekenstein, MD, of Dana-Dwek Children’s Hospital at the Tel Aviv Sourasky Medical Center in Israel. The findings were published in Rheumatology.
“We found that the mRNA Pfizer vaccine was immunogenic and induced an adequate humoral immune response in adolescent patients,” Dr. Heshin-Bekenstein told CARRA attendees. “It was definitely comparable to healthy controls and practically all patients were seropositive following the second vaccine, except for one patient with long-standing systemic sclerosis.”
The findings were not necessarily surprising but were encouraging to Melissa S. Oliver, MD, assistant professor of clinical pediatrics in the division of pediatric rheumatology at Indiana University, Indianapolis. Dr. Oliver wasn’t part of the study team.
“We know that the COVID vaccines in healthy adolescents have shown good efficacy with minimal side effects, and it’s good to see that this study showed that in those with rheumatic diseases on immunosuppressive therapy,” Dr. Oliver told this news organization.
Until now, the data on COVID-19 vaccines in teens with rheumatic illnesses has been limited, she said, so “many pediatric rheumatologists only have the data from adult studies to go on or personal experience with their own cohort of patients.”
But the high immunogenicity seen in the study was a pleasant surprise to Beth H. Rutstein, MD, assistant professor of clinical pediatrics in the division of rheumatology at Children’s Hospital of Philadelphia and the Perelman School of Medicine at the University of Pennsylvania.
“I was both surprised and thrilled with Dr. Heshin-Bekenstein’s findings suggesting near-universal seroconversion for patients with rheumatic disease regardless of underlying diagnosis or immunomodulatory therapy regimen, as much of the adult data has suggested a poorer seroconversion rate” and lower antibody titers in adults with similar illnesses, Dr. Rutstein said in an interview.
The study “provides essential reassurance that vaccination against COVID-19 does not increase the risk of disease flare or worsen disease severity scores,” said Dr. Rutstein, who was not associated with the research. “Rather than speaking purely anecdotally with our patients and their families, we can refer to the science – which is always more reassuring for both our patients and ourselves.”
Study included diverse conditions and therapies
Risk factors for poor outcomes with COVID-19 in children include obesity, cardiovascular disease, chronic lung disease, diabetes, and asthma, Dr. Heshin-Bekenstein told CARRA attendees. Multisystem inflammatory syndrome in children (MIS-C) and long COVID are also potential complications of COVID-19 with less understood risk factors.
Although COVID-19 is most often mild in children, certain severe, systemic rheumatic diseases increase hospitalization risk, including systemic lupus erythematosus (SLE) and vasculitis. Evidence has also shown that COVID-19 infection increases the risk of disease flare in teens with juvenile-onset rheumatic diseases, so it’s “crucial to prevent COVID-19 disease in this population,” Dr. Heshin-Bekenstein said.
Her study therefore aimed to assess the safety and immunogenicity of the Pfizer mRNA vaccine for teens with juvenile-onset rheumatic diseases and those taking immunomodulatory medications. The international prospective multicenter study ran from April to November 2021 at three pediatric rheumatology clinics in Israel and one in Slovenia. Endpoints included short-term side effects, vaccination impact on clinical disease activity, immunogenicity at 2-9 weeks after the second dose, and, secondarily, efficacy against COVID-19 infection.
The 91 participants included adolescents aged 12-18 and young adults aged 18-21. Nearly half of the participants (46%) had juvenile idiopathic arthritis (JIA), and 14% had SLE. Other participants’ conditions included systemic vasculitis, idiopathic uveitis, inflammatory bowel disease–related arthritis, systemic or localized scleroderma, juvenile dermatomyositis, or an autoinflammatory disease. Participants’ mean disease duration was 4.8 years.
The researchers compared the patients with a control group of 40 individuals with similar demographics but without rheumatic disease. The researchers used the LIAISON quantitative assay to assess serum IgG antibody levels against the SARS-CoV-2 spike protein in both groups.
Eight in 10 participants with rheumatic disease were taking an immunomodulatory medication, including a conventional synthetic disease-modifying antirheumatic drug (csDMARD) in 40%, a biologic DMARD in 37%, tumor necrosis factor (TNF) inhibitors in 32%, hydroxychloroquine (HCQ) in 19%, glucocorticoids in 14%, and mycophenolate in 11%. A smaller proportion were on other biologics: JAK inhibitors in 6.6%, anti-CD20 drugs in 4.4%, and an IL-6 inhibitor in 1%.
Side effects similar in both groups
None of the side effects reported by participants were statistically different between those with rheumatic disease and the control group. Localized pain was the most common side effect, reported by 73%-79% of participants after each dose. About twice as many participants with rheumatic disease experienced muscle aches and joint pains, compared with the control group, but the differences were not significant. Fever occurred more often in those with rheumatic disease (6%, five cases) than without (3%, one case). One-third of those with rheumatic disease felt tiredness, compared with 20% of the control group.
None of the healthy controls were hospitalized after vaccination, but three rheumatic patients were, including two after the first dose. Both were 17 years old, had systemic vasculitis with granulomatosis with polyangiitis (GPA), and were taking rituximab (Rituxan). One patient experienced acute onset of chronic renal failure, fever, dehydration, and high C-reactive protein within hours of vaccination. The other experienced new onset of pulmonary hemorrhage a week after vaccination.
In addition, a 14-year-old female with lupus, taking only HCQ, went to the emergency department with fever, headache, vomiting, and joint pain 1 day after the second vaccine dose. She had normal inflammatory markers and no change in disease activity score, and she was discharged with low-dose steroids tapered after 2 weeks.
Immune response high in patients with rheumatic disease
Immunogenicity was similar in both groups, with 97% seropositivity in the rheumatic disease group and 100% in the control group. Average IgG titers were 242 in the rheumatic group and 388 in the control group (P < .0001). Seropositivity was 88% in those taking mycophenolate with another drug (100% with mycophenolate monotherapy), 90% with HCQ, 94% with any csDMARDs and another drug (100% with csDMARD monotherapy), and 100% for all other drugs. During 3 months’ follow-up after vaccination, there were no COVID-19 cases among the participants.
Dr. Heshin-Bekenstein noted that their results showed better immunogenicity in teens, compared with adults, for two specific drugs. Seropositivity in teens taking methotrexate (Rheumatrex, Trexall) or rituximab was 100% in this study, compared with 84% in adults taking methotrexate and 39% in adults taking rituximab in a previous study. However, only three patients in this study were taking rituximab, and only seven were taking methotrexate.
The study’s heterogenous population was both a strength and a weakness of the study. “Due to the diversity of rheumatic diseases and medications included in this cohort, it was not possible to draw significant conclusions regarding the impact of the immunomodulatory medications and type of disease” on titers, Dr. Heshin-Bekenstein told attendees.
Still, “I think as pediatric rheumatologists, we can feel reassured in recommending the COVID-19 vaccine to our patients,” Dr. Oliver said. “I will add that every patient is different, and everyone should have a conversation with their physician about receiving the COVID-19 vaccine.” Dr. Oliver said she discusses vaccination, including COVID vaccination, with every patient, and it’s been challenging to address concerns in the midst of so much misinformation circulating about the vaccine.
These findings do raise questions about whether it’s still necessary to hold immunomodulatory medications to get the vaccine,” Dr. Rutstein said.
“Many families are nervous to pause their medications before and after the vaccine as is currently recommended for many therapies by the American College of Rheumatology, and I do share that concern for some of my patients with more clinically unstable disease, so I try to work with each family to decide on best timing and have delayed or deferred the series until some patients are on a steady dose of a new immunomodulatory medication if it has been recently started,” Dr. Rutstein said. “This is one of the reasons why Dr. Heshin-Bekenstein’s study is so important – we may be holding medications that can be safely continued and even further decrease the risk of disease flare.”
None of the physicians have disclosed any relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CARRA 2022
Prior authorizations delay TNF inhibitors for children with JIA
Children with juvenile idiopathic arthritis (JIA) who need a tumor necrosis factor (TNF) inhibitor after failing conventional disease-modifying antirheumatic drug (DMARD) treatment often experience insurance delays before beginning the new drug because of prior authorization denials, according to research presented at the 2022 annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). The findings were also published as a research letter in JAMA Network Open.
“Prompt escalation to TNF inhibitors is recommended for children with JIA refractory to DMARDs,” author Jordan Roberts, MD, a clinical fellow of the Harvard Medical School Rheumatology Program, Boston, told CARRA attendees. TNF inhibitors are increasingly used as first-line treatment in JIA since growing evidence suggests better outcomes from early treatment with biologics. “Prior authorization requirements that delay TNF inhibitor initiation among children with JIA are common in clinical practice,” Dr. Roberts said, but little evidence exists to understand the extent of this problem and its causes.
The researchers therefore conducted a retrospective cohort study using a search of electronic health records from January 2018 to December 2019 to find all children at a single center with a new diagnosis of nonsystemic JIA. Then the authors pulled the timing of prior authorization requests, approvals, denials, and first TNF inhibitor dose from the medical notes. They also sought out any children who had been recommended a TNF inhibitor but never started one.
The total population included 54 children with an average age of 10 years, about two-thirds of whom had private insurance (63%). The group was predominantly White (63%), although 13% declined to provide race, and 7% were Hispanic. Most subtypes of disease were represented: oligoarticular persistent (28%), oligoarticular extended (2%), polyarticular rheumatoid factor-negative (15%), polyarticular rheumatoid factor-positive (15%), psoriatic arthritis (26%), enthesitis-related arthritis (12%), and undifferentiated arthritis (2%).
The 44 participants with private insurance had an average of two joints with active disease, while the 10 patients with public insurance had an average of four involved joints. Nearly all the patients (91%) had previously taken or were currently taking DMARDs when the prior authorization was submitted, and 61% had received NSAIDs.
All but one of the patients’ insurance plans required a prior authorization. The first prior authorization was denied for about one-third of the public insurance patients (30%) and a quarter of the private insurance patients. About 1 in 5 patients overall (22%) required a written appeal to override the denial, and 4% required peer-to-peer review. Meanwhile, 7% of patients began another medication because of the denial.
It took a median of 3 days for prior authorizations to be approved and a median of 24 days from the time the TNF inhibitor was recommended to the patient receiving the first dose. However, 22% of patients waited at least 2 weeks before the prior authorization was approved, and more than a quarter of the requests took over 30 days before the patient could begin the medication. In the public insurance group in particular, a quarter of children waited at least 19 days for approval and at least 44 days before starting the medication.
In fact, when the researchers looked at the difference in approval time between those who did and did not receive an initial denial, the difference was stark. Median approval time was 16 days when the prior authorization was denied, compared with a median of 5 days when the first prior authorization was approved. Similarly, time to initiation of the drug after recommendation was a median of 35 days for those whose prior authorization was first denied and 17 days for those with an initial approval.
The most common reason for an initial denial was the insurance company requiring a different TNF inhibitor than the one the rheumatologist wanted to prescribe. “These were all children whose rheumatologist has recommended either infliximab or etanercept that were required to use adalimumab instead,” Dr. Roberts said.
The other reasons for initial denial were similarly familiar ones:
- Required submission to another insurer
- Additional documentation required
- Lack of medical necessity
- Prescription was for an indication not approved by the Food and Drug Administration
- Age of patient
- Nonbiologic DMARD required
- NSAID required for step therapy
Only three children who were advised to begin a TNF inhibitor did not do so, including one who was lost to follow-up, one who had injection-related anxiety, and one who had safety concerns about the medication.
“Several children were required to use alternative TNF inhibitors than the one that was recommended due to restricted formularies, which may reduce shared decisionmaking between physicians and families and may not be the optimal clinical choice for an individual child,” Dr. Roberts said in her conclusion. Most children, however, were able to get approval for the TNF inhibitor originally requested, “suggesting that utilization management strategies present barriers to timely care despite appropriate specialty medication requests,” she said. “Therefore, it’s important for us to advocate for access to medications for children with JIA.”
Findings are not surprising
“I have these same experiences at my institution – often insurance will dictate clinical practice, and step therapy is the only option, causing a delay to initiation of TNFi even if we think, as the pediatric rheumatologist, that a child needs this medicine to be initiated on presentation to our clinic,” Nayimisha Balmuri, MD, assistant professor of pediatrics in the division of allergy, immunology, and rheumatology at the Johns Hopkins School of Medicine, Baltimore, told this news organization.
Dr. Balmuri, who was not involved in the study, noted that in her clinic at Johns Hopkins, it is hit or miss if an appeal to insurance companies or to the state (if it is Medicaid coverage) will be successful. “Unfortunately, [we are] mostly unsuccessful, and we have to try another DMARD for 8 to 12 weeks first before trying to get TNFi,” she said.
Dr. Balmuri called for bringing these issues to the attention of state and federal legislators. “It’s so important for us to continue to advocate for our patients at the state and national level! We are the advocates for our patients, and we are uniquely trained to know the best medications to initiate to help patients maximize their chance to reach remission of arthritis. Insurance companies need to hear our voices!”
Dr. Roberts reported grants from CARRA, the Lupus Foundation of America, and the National Institute of Allergy and Infectious Diseases during the conduct of the study.
A version of this article first appeared on Medscape.com.
Children with juvenile idiopathic arthritis (JIA) who need a tumor necrosis factor (TNF) inhibitor after failing conventional disease-modifying antirheumatic drug (DMARD) treatment often experience insurance delays before beginning the new drug because of prior authorization denials, according to research presented at the 2022 annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). The findings were also published as a research letter in JAMA Network Open.
“Prompt escalation to TNF inhibitors is recommended for children with JIA refractory to DMARDs,” author Jordan Roberts, MD, a clinical fellow of the Harvard Medical School Rheumatology Program, Boston, told CARRA attendees. TNF inhibitors are increasingly used as first-line treatment in JIA since growing evidence suggests better outcomes from early treatment with biologics. “Prior authorization requirements that delay TNF inhibitor initiation among children with JIA are common in clinical practice,” Dr. Roberts said, but little evidence exists to understand the extent of this problem and its causes.
The researchers therefore conducted a retrospective cohort study using a search of electronic health records from January 2018 to December 2019 to find all children at a single center with a new diagnosis of nonsystemic JIA. Then the authors pulled the timing of prior authorization requests, approvals, denials, and first TNF inhibitor dose from the medical notes. They also sought out any children who had been recommended a TNF inhibitor but never started one.
The total population included 54 children with an average age of 10 years, about two-thirds of whom had private insurance (63%). The group was predominantly White (63%), although 13% declined to provide race, and 7% were Hispanic. Most subtypes of disease were represented: oligoarticular persistent (28%), oligoarticular extended (2%), polyarticular rheumatoid factor-negative (15%), polyarticular rheumatoid factor-positive (15%), psoriatic arthritis (26%), enthesitis-related arthritis (12%), and undifferentiated arthritis (2%).
The 44 participants with private insurance had an average of two joints with active disease, while the 10 patients with public insurance had an average of four involved joints. Nearly all the patients (91%) had previously taken or were currently taking DMARDs when the prior authorization was submitted, and 61% had received NSAIDs.
All but one of the patients’ insurance plans required a prior authorization. The first prior authorization was denied for about one-third of the public insurance patients (30%) and a quarter of the private insurance patients. About 1 in 5 patients overall (22%) required a written appeal to override the denial, and 4% required peer-to-peer review. Meanwhile, 7% of patients began another medication because of the denial.
It took a median of 3 days for prior authorizations to be approved and a median of 24 days from the time the TNF inhibitor was recommended to the patient receiving the first dose. However, 22% of patients waited at least 2 weeks before the prior authorization was approved, and more than a quarter of the requests took over 30 days before the patient could begin the medication. In the public insurance group in particular, a quarter of children waited at least 19 days for approval and at least 44 days before starting the medication.
In fact, when the researchers looked at the difference in approval time between those who did and did not receive an initial denial, the difference was stark. Median approval time was 16 days when the prior authorization was denied, compared with a median of 5 days when the first prior authorization was approved. Similarly, time to initiation of the drug after recommendation was a median of 35 days for those whose prior authorization was first denied and 17 days for those with an initial approval.
The most common reason for an initial denial was the insurance company requiring a different TNF inhibitor than the one the rheumatologist wanted to prescribe. “These were all children whose rheumatologist has recommended either infliximab or etanercept that were required to use adalimumab instead,” Dr. Roberts said.
The other reasons for initial denial were similarly familiar ones:
- Required submission to another insurer
- Additional documentation required
- Lack of medical necessity
- Prescription was for an indication not approved by the Food and Drug Administration
- Age of patient
- Nonbiologic DMARD required
- NSAID required for step therapy
Only three children who were advised to begin a TNF inhibitor did not do so, including one who was lost to follow-up, one who had injection-related anxiety, and one who had safety concerns about the medication.
“Several children were required to use alternative TNF inhibitors than the one that was recommended due to restricted formularies, which may reduce shared decisionmaking between physicians and families and may not be the optimal clinical choice for an individual child,” Dr. Roberts said in her conclusion. Most children, however, were able to get approval for the TNF inhibitor originally requested, “suggesting that utilization management strategies present barriers to timely care despite appropriate specialty medication requests,” she said. “Therefore, it’s important for us to advocate for access to medications for children with JIA.”
Findings are not surprising
“I have these same experiences at my institution – often insurance will dictate clinical practice, and step therapy is the only option, causing a delay to initiation of TNFi even if we think, as the pediatric rheumatologist, that a child needs this medicine to be initiated on presentation to our clinic,” Nayimisha Balmuri, MD, assistant professor of pediatrics in the division of allergy, immunology, and rheumatology at the Johns Hopkins School of Medicine, Baltimore, told this news organization.
Dr. Balmuri, who was not involved in the study, noted that in her clinic at Johns Hopkins, it is hit or miss if an appeal to insurance companies or to the state (if it is Medicaid coverage) will be successful. “Unfortunately, [we are] mostly unsuccessful, and we have to try another DMARD for 8 to 12 weeks first before trying to get TNFi,” she said.
Dr. Balmuri called for bringing these issues to the attention of state and federal legislators. “It’s so important for us to continue to advocate for our patients at the state and national level! We are the advocates for our patients, and we are uniquely trained to know the best medications to initiate to help patients maximize their chance to reach remission of arthritis. Insurance companies need to hear our voices!”
Dr. Roberts reported grants from CARRA, the Lupus Foundation of America, and the National Institute of Allergy and Infectious Diseases during the conduct of the study.
A version of this article first appeared on Medscape.com.
Children with juvenile idiopathic arthritis (JIA) who need a tumor necrosis factor (TNF) inhibitor after failing conventional disease-modifying antirheumatic drug (DMARD) treatment often experience insurance delays before beginning the new drug because of prior authorization denials, according to research presented at the 2022 annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). The findings were also published as a research letter in JAMA Network Open.
“Prompt escalation to TNF inhibitors is recommended for children with JIA refractory to DMARDs,” author Jordan Roberts, MD, a clinical fellow of the Harvard Medical School Rheumatology Program, Boston, told CARRA attendees. TNF inhibitors are increasingly used as first-line treatment in JIA since growing evidence suggests better outcomes from early treatment with biologics. “Prior authorization requirements that delay TNF inhibitor initiation among children with JIA are common in clinical practice,” Dr. Roberts said, but little evidence exists to understand the extent of this problem and its causes.
The researchers therefore conducted a retrospective cohort study using a search of electronic health records from January 2018 to December 2019 to find all children at a single center with a new diagnosis of nonsystemic JIA. Then the authors pulled the timing of prior authorization requests, approvals, denials, and first TNF inhibitor dose from the medical notes. They also sought out any children who had been recommended a TNF inhibitor but never started one.
The total population included 54 children with an average age of 10 years, about two-thirds of whom had private insurance (63%). The group was predominantly White (63%), although 13% declined to provide race, and 7% were Hispanic. Most subtypes of disease were represented: oligoarticular persistent (28%), oligoarticular extended (2%), polyarticular rheumatoid factor-negative (15%), polyarticular rheumatoid factor-positive (15%), psoriatic arthritis (26%), enthesitis-related arthritis (12%), and undifferentiated arthritis (2%).
The 44 participants with private insurance had an average of two joints with active disease, while the 10 patients with public insurance had an average of four involved joints. Nearly all the patients (91%) had previously taken or were currently taking DMARDs when the prior authorization was submitted, and 61% had received NSAIDs.
All but one of the patients’ insurance plans required a prior authorization. The first prior authorization was denied for about one-third of the public insurance patients (30%) and a quarter of the private insurance patients. About 1 in 5 patients overall (22%) required a written appeal to override the denial, and 4% required peer-to-peer review. Meanwhile, 7% of patients began another medication because of the denial.
It took a median of 3 days for prior authorizations to be approved and a median of 24 days from the time the TNF inhibitor was recommended to the patient receiving the first dose. However, 22% of patients waited at least 2 weeks before the prior authorization was approved, and more than a quarter of the requests took over 30 days before the patient could begin the medication. In the public insurance group in particular, a quarter of children waited at least 19 days for approval and at least 44 days before starting the medication.
In fact, when the researchers looked at the difference in approval time between those who did and did not receive an initial denial, the difference was stark. Median approval time was 16 days when the prior authorization was denied, compared with a median of 5 days when the first prior authorization was approved. Similarly, time to initiation of the drug after recommendation was a median of 35 days for those whose prior authorization was first denied and 17 days for those with an initial approval.
The most common reason for an initial denial was the insurance company requiring a different TNF inhibitor than the one the rheumatologist wanted to prescribe. “These were all children whose rheumatologist has recommended either infliximab or etanercept that were required to use adalimumab instead,” Dr. Roberts said.
The other reasons for initial denial were similarly familiar ones:
- Required submission to another insurer
- Additional documentation required
- Lack of medical necessity
- Prescription was for an indication not approved by the Food and Drug Administration
- Age of patient
- Nonbiologic DMARD required
- NSAID required for step therapy
Only three children who were advised to begin a TNF inhibitor did not do so, including one who was lost to follow-up, one who had injection-related anxiety, and one who had safety concerns about the medication.
“Several children were required to use alternative TNF inhibitors than the one that was recommended due to restricted formularies, which may reduce shared decisionmaking between physicians and families and may not be the optimal clinical choice for an individual child,” Dr. Roberts said in her conclusion. Most children, however, were able to get approval for the TNF inhibitor originally requested, “suggesting that utilization management strategies present barriers to timely care despite appropriate specialty medication requests,” she said. “Therefore, it’s important for us to advocate for access to medications for children with JIA.”
Findings are not surprising
“I have these same experiences at my institution – often insurance will dictate clinical practice, and step therapy is the only option, causing a delay to initiation of TNFi even if we think, as the pediatric rheumatologist, that a child needs this medicine to be initiated on presentation to our clinic,” Nayimisha Balmuri, MD, assistant professor of pediatrics in the division of allergy, immunology, and rheumatology at the Johns Hopkins School of Medicine, Baltimore, told this news organization.
Dr. Balmuri, who was not involved in the study, noted that in her clinic at Johns Hopkins, it is hit or miss if an appeal to insurance companies or to the state (if it is Medicaid coverage) will be successful. “Unfortunately, [we are] mostly unsuccessful, and we have to try another DMARD for 8 to 12 weeks first before trying to get TNFi,” she said.
Dr. Balmuri called for bringing these issues to the attention of state and federal legislators. “It’s so important for us to continue to advocate for our patients at the state and national level! We are the advocates for our patients, and we are uniquely trained to know the best medications to initiate to help patients maximize their chance to reach remission of arthritis. Insurance companies need to hear our voices!”
Dr. Roberts reported grants from CARRA, the Lupus Foundation of America, and the National Institute of Allergy and Infectious Diseases during the conduct of the study.
A version of this article first appeared on Medscape.com.
Gaps in follow-up care put kids with asthma at risk of severe recurrence
Jo Ward’s twin boys have been to the emergency department for respiratory problems about as many times as the dozen years they’ve been alive. Both have asthma and bronchopulmonary dysplasia, a form of chronic airway damage that can occur in children born premature, as the twins were. But each time Ms. Ward took them in for treatment during an acute bout of breathing distress, the staff told her to schedule a follow-up visit for the children with their physician only if they didn’t get better, not regardless of the outcome – as medical guidelines recommend.
“They asked questions, they did the exams, but they really didn’t give you a lot of information to help you at home,” Ms. Ward told this news organization. If they had, she doesn’t think she’d have needed to take them in for emergency care so often.
A new study, published in Academic Pediatrics, suggests she’s right.
Current clinical guidelines for asthma recommend that patients who visit the ED for an asthma-related problem should have a follow-up appointment within a month after the visit, independent of how well they have recovered once home, according to Naomi S. Bardach, MD, a professor of pediatrics and health policy at the University of California, San Francisco, who led the new study.
Her research found that children who have a follow-up appointment within 2 weeks of such a visit are less likely to come back again the next year. Yet the study also found that only about one in five youth had a follow-up visit within that 2-week window.
“The emergency department visit is probably a sign that they need some additional attention for their asthma,” Dr. Bardach said. “We know we can prevent emergency department visits if they get the right kind of medication or if they figure out how to avoid the things that are going to cause an asthma exacerbation or flare.”
For the study, Dr. Bardach and colleagues analyzed data from California, Vermont, and Massachusetts for all asthma-related emergency visits for patients aged 3-21 years between 2013 and 2016.
Out of the 90,267 such visits they identified, 22.6% of patients had a follow-up within 2 weeks, more often by patients who were younger, had commercial insurance, had evidence of prior asthma, or had complex chronic conditions.
Whereas 5.7% of patients who had follow-up visits returned to the ED within 60 days, 6.4% of those who didn’t came back – a 12% difference (P < .001). The gap was larger a year out, with 25% of those with follow-ups returning to the ED, compared with 28.3% of those without follow-ups returning (P < .001), according to the researchers.
Overall, Dr. Bardach’s group estimates that for every 30 children who have follow-up visits with a physician, one would avoid a return trip to the emergency department for asthma within a year.
But given the sheer number of asthma-related trips to the ED each year – 164,145 for kids age 1-17 years in the United States in 2016 alone – that translates into big numbers of kids not going back to the hospital: approximately 72,000 such trips avoided at a savings to the health care system of at least $8.6 million annually.
Missed opportunities
Had Ms. Ward’s boys been among the one in five to receive follow-up care earlier in their lives, she might have saved a significant amount of time, money, anxiety, and heartache. When the twins were 9 years old, she took them to a new pediatric pulmonologist. That changed everything. In that first visit, “they gave me way more information than I ever had in the first 9 years,” she said.
The doctor told Ms. Ward to keep steroids on hand, gave her a prescription for extra doses of the powerful medication, and explained that they needed to be used within 24 hours of the first sign of a breathing problem.
“She said if you give them the steroids right away, it keeps them out of the emergency room, and that’s actually worked,” Ms. Ward said. “She made sure we had care plans every visit and asked me each time if I still had it or we needed to rewrite it. They gave me signs to look for, for when to go to hospital visits. I think that when you go to the doctor, they should be telling you stuff like that.”
Dr. Bardach said visits with a primary care doctor or asthma specialist offer families a chance to receive information to keep the condition from becoming critical.
“Going to that follow-up visit, they can get access to education from the provider about how to avoid things that trigger asthma, and there’s medication that kids can take that keeps the lungs calm and less likely to have a big asthma reaction, so getting access to that medication can be really helpful,” she said.
That was the case for Amy Davenport, of Chapel Hill, N.C., whose 6-year-old son has been to the ED twice for his asthma.
The first time, when he was 3, he was having trouble breathing with a respiratory tract infection and received nebulizer treatment – although he received it in the ED since no beds were available in the ICU. The staff did tell Ms. Davenport to follow up with her primary care provider, but her son’s pediatrician was reluctant to diagnose him with asthma at such a young age and didn’t prescribe any maintenance medications.
A few months later, Ms. Davenport and her son found themselves back in the hospital, and an ICU bed was open this time. The critical care staff referred Davenport to a pediatric pulmonary specialist, and they haven’t been back to the hospital since. Ms. Davenport said she believes if they’d received a maintenance medication after the first visit, it likely would have prevented the second one.
“I’ve definitely seen now that, after the second admission, we got an asthma action plan and it said exactly what to do,” she said. “I felt like we had really good follow-up. We had that action plan on our refrigerator for a long time, and it helped us as parents with three small children to manage.”
Of course, follow-up care takes time – time away from work and school that not all families can spare, the researchers acknowledged. Telehealth may be an option, especially after its use expanded during the COVID-19 pandemic.
“We know that health systems have a hard time being flexible enough to actually have a kid be able to make an appointment within a short period of time, and we also know it’s hard for families sometimes to go back into a clinical setting within a certain period of time,” Dr. Bardach said. The urgency for the appointment may wane for those whose children seem to be doing better.
When the researchers adjusted their calculations for socioeconomic status, the results didn’t change much. But the study did find that patients with private insurance were about twice as likely to have follow-up visits as those on Medicaid (43.7% vs. 21.7%). And “the content and conduct” of the follow-up visit makes a difference as well.
Ms. Ward, whose boys are insured through Medicaid, recalled several visits to the ED where she had to push the staff to get the care her children needed. In one case, when one of her boys was a year old and struggling to breathe, the emergency doctor handed her a prescription and recommended she fill it at a neighborhood drugstore that would be cheaper than the hospital’s pharmacy. Then a nurse came in to begin the discharge process.
“I said no, ‘we’re not ready yet. Look at him,’” Ms. Ward said. The nurse took a pulse oximeter reading that showed the boy’s oxygen levels were at 84%, dangerously low. “If I wasn’t so knowledgeable and paid attention when they were born, since they were preemies, if it would have been somebody else, they probably would’ve went home and he’d have died.”
With the pediatric pulmonologist the boys have now, Ms. Ward said she feels more capable of managing their asthma and knowing how to reduce the likelihood that they’ll need to visit the ED.
“Part of what we’re seeing here is that having an existing and trusting relationship with a clinician can be helpful to kids with asthma,” Dr. Bardach said. “If we help establish and maintain those connections, and explain how important that connection can be, that can also help somebody with asthma overall.”
The research was funded by the Agency for Healthcare Research and Quality. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Jo Ward’s twin boys have been to the emergency department for respiratory problems about as many times as the dozen years they’ve been alive. Both have asthma and bronchopulmonary dysplasia, a form of chronic airway damage that can occur in children born premature, as the twins were. But each time Ms. Ward took them in for treatment during an acute bout of breathing distress, the staff told her to schedule a follow-up visit for the children with their physician only if they didn’t get better, not regardless of the outcome – as medical guidelines recommend.
“They asked questions, they did the exams, but they really didn’t give you a lot of information to help you at home,” Ms. Ward told this news organization. If they had, she doesn’t think she’d have needed to take them in for emergency care so often.
A new study, published in Academic Pediatrics, suggests she’s right.
Current clinical guidelines for asthma recommend that patients who visit the ED for an asthma-related problem should have a follow-up appointment within a month after the visit, independent of how well they have recovered once home, according to Naomi S. Bardach, MD, a professor of pediatrics and health policy at the University of California, San Francisco, who led the new study.
Her research found that children who have a follow-up appointment within 2 weeks of such a visit are less likely to come back again the next year. Yet the study also found that only about one in five youth had a follow-up visit within that 2-week window.
“The emergency department visit is probably a sign that they need some additional attention for their asthma,” Dr. Bardach said. “We know we can prevent emergency department visits if they get the right kind of medication or if they figure out how to avoid the things that are going to cause an asthma exacerbation or flare.”
For the study, Dr. Bardach and colleagues analyzed data from California, Vermont, and Massachusetts for all asthma-related emergency visits for patients aged 3-21 years between 2013 and 2016.
Out of the 90,267 such visits they identified, 22.6% of patients had a follow-up within 2 weeks, more often by patients who were younger, had commercial insurance, had evidence of prior asthma, or had complex chronic conditions.
Whereas 5.7% of patients who had follow-up visits returned to the ED within 60 days, 6.4% of those who didn’t came back – a 12% difference (P < .001). The gap was larger a year out, with 25% of those with follow-ups returning to the ED, compared with 28.3% of those without follow-ups returning (P < .001), according to the researchers.
Overall, Dr. Bardach’s group estimates that for every 30 children who have follow-up visits with a physician, one would avoid a return trip to the emergency department for asthma within a year.
But given the sheer number of asthma-related trips to the ED each year – 164,145 for kids age 1-17 years in the United States in 2016 alone – that translates into big numbers of kids not going back to the hospital: approximately 72,000 such trips avoided at a savings to the health care system of at least $8.6 million annually.
Missed opportunities
Had Ms. Ward’s boys been among the one in five to receive follow-up care earlier in their lives, she might have saved a significant amount of time, money, anxiety, and heartache. When the twins were 9 years old, she took them to a new pediatric pulmonologist. That changed everything. In that first visit, “they gave me way more information than I ever had in the first 9 years,” she said.
The doctor told Ms. Ward to keep steroids on hand, gave her a prescription for extra doses of the powerful medication, and explained that they needed to be used within 24 hours of the first sign of a breathing problem.
“She said if you give them the steroids right away, it keeps them out of the emergency room, and that’s actually worked,” Ms. Ward said. “She made sure we had care plans every visit and asked me each time if I still had it or we needed to rewrite it. They gave me signs to look for, for when to go to hospital visits. I think that when you go to the doctor, they should be telling you stuff like that.”
Dr. Bardach said visits with a primary care doctor or asthma specialist offer families a chance to receive information to keep the condition from becoming critical.
“Going to that follow-up visit, they can get access to education from the provider about how to avoid things that trigger asthma, and there’s medication that kids can take that keeps the lungs calm and less likely to have a big asthma reaction, so getting access to that medication can be really helpful,” she said.
That was the case for Amy Davenport, of Chapel Hill, N.C., whose 6-year-old son has been to the ED twice for his asthma.
The first time, when he was 3, he was having trouble breathing with a respiratory tract infection and received nebulizer treatment – although he received it in the ED since no beds were available in the ICU. The staff did tell Ms. Davenport to follow up with her primary care provider, but her son’s pediatrician was reluctant to diagnose him with asthma at such a young age and didn’t prescribe any maintenance medications.
A few months later, Ms. Davenport and her son found themselves back in the hospital, and an ICU bed was open this time. The critical care staff referred Davenport to a pediatric pulmonary specialist, and they haven’t been back to the hospital since. Ms. Davenport said she believes if they’d received a maintenance medication after the first visit, it likely would have prevented the second one.
“I’ve definitely seen now that, after the second admission, we got an asthma action plan and it said exactly what to do,” she said. “I felt like we had really good follow-up. We had that action plan on our refrigerator for a long time, and it helped us as parents with three small children to manage.”
Of course, follow-up care takes time – time away from work and school that not all families can spare, the researchers acknowledged. Telehealth may be an option, especially after its use expanded during the COVID-19 pandemic.
“We know that health systems have a hard time being flexible enough to actually have a kid be able to make an appointment within a short period of time, and we also know it’s hard for families sometimes to go back into a clinical setting within a certain period of time,” Dr. Bardach said. The urgency for the appointment may wane for those whose children seem to be doing better.
When the researchers adjusted their calculations for socioeconomic status, the results didn’t change much. But the study did find that patients with private insurance were about twice as likely to have follow-up visits as those on Medicaid (43.7% vs. 21.7%). And “the content and conduct” of the follow-up visit makes a difference as well.
Ms. Ward, whose boys are insured through Medicaid, recalled several visits to the ED where she had to push the staff to get the care her children needed. In one case, when one of her boys was a year old and struggling to breathe, the emergency doctor handed her a prescription and recommended she fill it at a neighborhood drugstore that would be cheaper than the hospital’s pharmacy. Then a nurse came in to begin the discharge process.
“I said no, ‘we’re not ready yet. Look at him,’” Ms. Ward said. The nurse took a pulse oximeter reading that showed the boy’s oxygen levels were at 84%, dangerously low. “If I wasn’t so knowledgeable and paid attention when they were born, since they were preemies, if it would have been somebody else, they probably would’ve went home and he’d have died.”
With the pediatric pulmonologist the boys have now, Ms. Ward said she feels more capable of managing their asthma and knowing how to reduce the likelihood that they’ll need to visit the ED.
“Part of what we’re seeing here is that having an existing and trusting relationship with a clinician can be helpful to kids with asthma,” Dr. Bardach said. “If we help establish and maintain those connections, and explain how important that connection can be, that can also help somebody with asthma overall.”
The research was funded by the Agency for Healthcare Research and Quality. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Jo Ward’s twin boys have been to the emergency department for respiratory problems about as many times as the dozen years they’ve been alive. Both have asthma and bronchopulmonary dysplasia, a form of chronic airway damage that can occur in children born premature, as the twins were. But each time Ms. Ward took them in for treatment during an acute bout of breathing distress, the staff told her to schedule a follow-up visit for the children with their physician only if they didn’t get better, not regardless of the outcome – as medical guidelines recommend.
“They asked questions, they did the exams, but they really didn’t give you a lot of information to help you at home,” Ms. Ward told this news organization. If they had, she doesn’t think she’d have needed to take them in for emergency care so often.
A new study, published in Academic Pediatrics, suggests she’s right.
Current clinical guidelines for asthma recommend that patients who visit the ED for an asthma-related problem should have a follow-up appointment within a month after the visit, independent of how well they have recovered once home, according to Naomi S. Bardach, MD, a professor of pediatrics and health policy at the University of California, San Francisco, who led the new study.
Her research found that children who have a follow-up appointment within 2 weeks of such a visit are less likely to come back again the next year. Yet the study also found that only about one in five youth had a follow-up visit within that 2-week window.
“The emergency department visit is probably a sign that they need some additional attention for their asthma,” Dr. Bardach said. “We know we can prevent emergency department visits if they get the right kind of medication or if they figure out how to avoid the things that are going to cause an asthma exacerbation or flare.”
For the study, Dr. Bardach and colleagues analyzed data from California, Vermont, and Massachusetts for all asthma-related emergency visits for patients aged 3-21 years between 2013 and 2016.
Out of the 90,267 such visits they identified, 22.6% of patients had a follow-up within 2 weeks, more often by patients who were younger, had commercial insurance, had evidence of prior asthma, or had complex chronic conditions.
Whereas 5.7% of patients who had follow-up visits returned to the ED within 60 days, 6.4% of those who didn’t came back – a 12% difference (P < .001). The gap was larger a year out, with 25% of those with follow-ups returning to the ED, compared with 28.3% of those without follow-ups returning (P < .001), according to the researchers.
Overall, Dr. Bardach’s group estimates that for every 30 children who have follow-up visits with a physician, one would avoid a return trip to the emergency department for asthma within a year.
But given the sheer number of asthma-related trips to the ED each year – 164,145 for kids age 1-17 years in the United States in 2016 alone – that translates into big numbers of kids not going back to the hospital: approximately 72,000 such trips avoided at a savings to the health care system of at least $8.6 million annually.
Missed opportunities
Had Ms. Ward’s boys been among the one in five to receive follow-up care earlier in their lives, she might have saved a significant amount of time, money, anxiety, and heartache. When the twins were 9 years old, she took them to a new pediatric pulmonologist. That changed everything. In that first visit, “they gave me way more information than I ever had in the first 9 years,” she said.
The doctor told Ms. Ward to keep steroids on hand, gave her a prescription for extra doses of the powerful medication, and explained that they needed to be used within 24 hours of the first sign of a breathing problem.
“She said if you give them the steroids right away, it keeps them out of the emergency room, and that’s actually worked,” Ms. Ward said. “She made sure we had care plans every visit and asked me each time if I still had it or we needed to rewrite it. They gave me signs to look for, for when to go to hospital visits. I think that when you go to the doctor, they should be telling you stuff like that.”
Dr. Bardach said visits with a primary care doctor or asthma specialist offer families a chance to receive information to keep the condition from becoming critical.
“Going to that follow-up visit, they can get access to education from the provider about how to avoid things that trigger asthma, and there’s medication that kids can take that keeps the lungs calm and less likely to have a big asthma reaction, so getting access to that medication can be really helpful,” she said.
That was the case for Amy Davenport, of Chapel Hill, N.C., whose 6-year-old son has been to the ED twice for his asthma.
The first time, when he was 3, he was having trouble breathing with a respiratory tract infection and received nebulizer treatment – although he received it in the ED since no beds were available in the ICU. The staff did tell Ms. Davenport to follow up with her primary care provider, but her son’s pediatrician was reluctant to diagnose him with asthma at such a young age and didn’t prescribe any maintenance medications.
A few months later, Ms. Davenport and her son found themselves back in the hospital, and an ICU bed was open this time. The critical care staff referred Davenport to a pediatric pulmonary specialist, and they haven’t been back to the hospital since. Ms. Davenport said she believes if they’d received a maintenance medication after the first visit, it likely would have prevented the second one.
“I’ve definitely seen now that, after the second admission, we got an asthma action plan and it said exactly what to do,” she said. “I felt like we had really good follow-up. We had that action plan on our refrigerator for a long time, and it helped us as parents with three small children to manage.”
Of course, follow-up care takes time – time away from work and school that not all families can spare, the researchers acknowledged. Telehealth may be an option, especially after its use expanded during the COVID-19 pandemic.
“We know that health systems have a hard time being flexible enough to actually have a kid be able to make an appointment within a short period of time, and we also know it’s hard for families sometimes to go back into a clinical setting within a certain period of time,” Dr. Bardach said. The urgency for the appointment may wane for those whose children seem to be doing better.
When the researchers adjusted their calculations for socioeconomic status, the results didn’t change much. But the study did find that patients with private insurance were about twice as likely to have follow-up visits as those on Medicaid (43.7% vs. 21.7%). And “the content and conduct” of the follow-up visit makes a difference as well.
Ms. Ward, whose boys are insured through Medicaid, recalled several visits to the ED where she had to push the staff to get the care her children needed. In one case, when one of her boys was a year old and struggling to breathe, the emergency doctor handed her a prescription and recommended she fill it at a neighborhood drugstore that would be cheaper than the hospital’s pharmacy. Then a nurse came in to begin the discharge process.
“I said no, ‘we’re not ready yet. Look at him,’” Ms. Ward said. The nurse took a pulse oximeter reading that showed the boy’s oxygen levels were at 84%, dangerously low. “If I wasn’t so knowledgeable and paid attention when they were born, since they were preemies, if it would have been somebody else, they probably would’ve went home and he’d have died.”
With the pediatric pulmonologist the boys have now, Ms. Ward said she feels more capable of managing their asthma and knowing how to reduce the likelihood that they’ll need to visit the ED.
“Part of what we’re seeing here is that having an existing and trusting relationship with a clinician can be helpful to kids with asthma,” Dr. Bardach said. “If we help establish and maintain those connections, and explain how important that connection can be, that can also help somebody with asthma overall.”
The research was funded by the Agency for Healthcare Research and Quality. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACADEMIC PEDIATRICS
JIA disease activity, disability linked to social factors
For children with polyarticular juvenile idiopathic arthritis (pJIA), functional disability lasts longer and disease activity is increased among those who belong to a racial/ethnic minority or come from homes with low household income or low family education, according to a study published online in Pediatric Rheumatology. The findings also initially revealed a higher likelihood of functional disability among those living in a poorer community, but that association lost statistical significance after adjustment for confounders.
“We chose community poverty level as the primary predictor for outcomes in pJIA because the socioeconomic context of communities and neighborhoods affects the characteristics of the social, service, and physical environments to which all residents are exposed regardless of their own socioeconomic position and may have a greater negative impact on those with fewer individual resources,” the authors write. “While community poverty level was not associated with an increase in odds of moderate-to-severe disease activity, those with high community poverty level did have higher disease activity scores (0.33 points greater on average than those with low community poverty level, in adjusted analysis).”
Nayimisha Balmuri, MD, an assistant professor of pediatrics at Johns Hopkins Medicine and study coauthor, told this news organization that anecdotal experience from everyday practice has shown that “patients with myriad social determinants of health stacked against them present sicker, take longer to present, and require far more aggressive therapies and follow-up,” which wreaks havoc in terms of disease activity. “It’s really difficult, then, to play catch-up to other cohorts of patients,” Dr. Balmuri added.
Disparities in outcomes persist
A key clinical take-home message from these findings is that the differences in clinical outcomes are relevant throughout the entire year of therapy, Dr. Balmuri said. “Patients get better; however, they don’t get better the same,” she said, and this is because of a variety of reasons. “Getting in the door is one of [those reasons] but then continuing to follow-up care is another.” For general practitioners, it’s especially important to refer patients who complain of joint pains to a specialist and to then follow up to be sure they’re improving and they’re getting the care they need.
For pediatric rheumatologists and subspecialists, “it’s important for us to realize that the disparity doesn’t end when patients come into your door to begin with,” Dr. Balmuri said. “It continues over the short term and far past that into adulthood.”
Candace Feldman, MD, MPH, ScD, an assistant professor of medicine in the Division of Rheumatology, Inflammation, and Immunity at Brigham and Women’s Hospital, Boston, told this news organization that the research “provides an important foundation to the study of the impact of social determinants of health on disease activity and disability among children with JIA. Individuals with rheumatic conditions should be screened for social determinants of health–related needs, and infrastructure should exist within the rheumatology clinic to help address the needs uncovered.” Dr. Feldman was not involved in the study.
In addition to the results’ clinical significance, Dr. Feldman also noted the policy implications of these findings. “Physicians should advocate for efforts to dismantle structural racism, to address income inequality, and to mitigate the effects of climate change, which also disproportionately affect historically marginalized populations,” Dr. Feldman said. Although this study focused predominantly on poverty, she noted that financial insecurity, food insecurity, homelessness, or housing instability were other social determinants of health to consider in future research.
Dr. Balmuri and William Daniel Soulsby, MD, a clinical fellow in pediatric rheumatology at the University of California, San Francisco, who is the study’s lead author, said they focused on poverty in this study not only because it’s so understudied in patients with pJIA but also because research in adults with lupus has found that leaving poverty was associated with a reversal of accrued disease damage.
Interactions of social determinants
The authors analyzed retrospective data from 1,684 pediatric patients in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry covering the period of April 2015 to February 2020. All study participants had been diagnosed with pJIA. Symptom onset occurred before age 16, and at least five joints were involved. The authors excluded patients who had been diagnosed with other systemic inflammatory or autoimmune diseases.
The authors defined exposure to a high level of community poverty as living in a ZIP code where at least 20% of residents lived at or below the federal poverty level. The authors also collected data on household income, although these data were missing for more than a quarter of participants (27%) and were therefore included only in sensitivity analyses. They used the clinical Juvenile Arthritis Disease Activity Score–10 (cJADAS-10) and the Child Health Assessment Questionnaire (CHAQ) to assess disease activity and disability at baseline and 6 and 12 months later. A cutoff of 2.5 on the cJADAS-10 distinguished mild disease activity from moderate to high disease activity, and a CHAQ score of 0.25 was the cutoff for having functional disability.
Among those who reported household income, just over half the cohort had an income of at least $50,000. The study population was 74% White, and more non-White patients lived in high-poverty communities (36.4%) than did White patients (21.3%). Patients whose families had no more than a high school education (23.1% vs. 13.7%) and those with public insurance (43.0% vs. 21.5%) were also over-represented in poorer communities.
The median cJADAS-10 scores declined overall during patients’ first year of therapy. However, those with public insurance, a lower family education level, or residency in poorer communities made up the greatest proportion of patients who continued to have moderate to severe disease activity a year after diagnosis.
The unadjusted calculations showed that children living in high community poverty had 1.8 times greater odds of functional disability (odds ratio, 1.82; P < .001). However, after adjustment for age, sex, race/ethnicity, insurance status, family education, rheumatoid factor, and cyclic citrullinated peptide antibody, the association lost statistical significance (P = .3). Community poverty level was not associated with disease activity before or after adjustment.
“Race was adjusted for as a confounder; however, the association between race/ethnicity and social determinants of health is likely more complex,” Dr. Feldman said. “Interactions, for example, between individual race and area-level poverty could be investigated.”
Odds of persistent function disability were 1.5 times greater for children with public insurance (adjusted OR, 1.56; P = .023) and 1.9 times greater for those whose families had a lower education level (aOR, 1.89; P = .013). Children whose race/ethnicity was indicated as being other than White had more than double the odds of higher disease activity (aOR, 2.48; P = .002) and were nearly twice as likely to have persistent functional disability (aOR, 1.91; P = .031).
Future directions
Dr. Soulsby was struck by the difference in statistical significance between individual-level poverty, as measured by household income, and community-level poverty. “It’s interesting because it may suggest that both of these forms of poverty are different and have different impacts on disease,” he said. Dr. Balmuri elaborated on the nuances and interactions that exist with social determinants of health and how objective outcomes, such as disease activity as measured by clinical tools, can differ from subjective outcomes, such as patients’ reports of pain, daily disability, and social experiences.
“The human condition is far more complicated, unfortunately, than any dataset could have on their own collected,” Dr. Balmuri said. She said she plans to expand her pJIA research into other social determinants of health. “It’s first about getting people’s eyes and minds open to something we see every day that, for some reason, sometimes people are blinded to, [using] the data that we do have, and then our hope is to build upon that.”
Dr. Feldman noted that ZIP codes, which were used as a proxy for community poverty, may not provide the best perspective regarding a patient’s neighborhood, because significant variation may exist within a single ZIP code, which is something the authors noted as well. The investigators were limited in the data available from the registry, and Dr. Balmuri and Dr. Soulsby suggested that 9-digit ZIP codes or census tracts might better capture neighborhood deprivation.
The research was funded by the Arthritis Foundation and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Feldman has received research support from Pfizer and the Bristol-Myers Squibb Foundation. Dr. Soulsby and Dr. Balmuri have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For children with polyarticular juvenile idiopathic arthritis (pJIA), functional disability lasts longer and disease activity is increased among those who belong to a racial/ethnic minority or come from homes with low household income or low family education, according to a study published online in Pediatric Rheumatology. The findings also initially revealed a higher likelihood of functional disability among those living in a poorer community, but that association lost statistical significance after adjustment for confounders.
“We chose community poverty level as the primary predictor for outcomes in pJIA because the socioeconomic context of communities and neighborhoods affects the characteristics of the social, service, and physical environments to which all residents are exposed regardless of their own socioeconomic position and may have a greater negative impact on those with fewer individual resources,” the authors write. “While community poverty level was not associated with an increase in odds of moderate-to-severe disease activity, those with high community poverty level did have higher disease activity scores (0.33 points greater on average than those with low community poverty level, in adjusted analysis).”
Nayimisha Balmuri, MD, an assistant professor of pediatrics at Johns Hopkins Medicine and study coauthor, told this news organization that anecdotal experience from everyday practice has shown that “patients with myriad social determinants of health stacked against them present sicker, take longer to present, and require far more aggressive therapies and follow-up,” which wreaks havoc in terms of disease activity. “It’s really difficult, then, to play catch-up to other cohorts of patients,” Dr. Balmuri added.
Disparities in outcomes persist
A key clinical take-home message from these findings is that the differences in clinical outcomes are relevant throughout the entire year of therapy, Dr. Balmuri said. “Patients get better; however, they don’t get better the same,” she said, and this is because of a variety of reasons. “Getting in the door is one of [those reasons] but then continuing to follow-up care is another.” For general practitioners, it’s especially important to refer patients who complain of joint pains to a specialist and to then follow up to be sure they’re improving and they’re getting the care they need.
For pediatric rheumatologists and subspecialists, “it’s important for us to realize that the disparity doesn’t end when patients come into your door to begin with,” Dr. Balmuri said. “It continues over the short term and far past that into adulthood.”
Candace Feldman, MD, MPH, ScD, an assistant professor of medicine in the Division of Rheumatology, Inflammation, and Immunity at Brigham and Women’s Hospital, Boston, told this news organization that the research “provides an important foundation to the study of the impact of social determinants of health on disease activity and disability among children with JIA. Individuals with rheumatic conditions should be screened for social determinants of health–related needs, and infrastructure should exist within the rheumatology clinic to help address the needs uncovered.” Dr. Feldman was not involved in the study.
In addition to the results’ clinical significance, Dr. Feldman also noted the policy implications of these findings. “Physicians should advocate for efforts to dismantle structural racism, to address income inequality, and to mitigate the effects of climate change, which also disproportionately affect historically marginalized populations,” Dr. Feldman said. Although this study focused predominantly on poverty, she noted that financial insecurity, food insecurity, homelessness, or housing instability were other social determinants of health to consider in future research.
Dr. Balmuri and William Daniel Soulsby, MD, a clinical fellow in pediatric rheumatology at the University of California, San Francisco, who is the study’s lead author, said they focused on poverty in this study not only because it’s so understudied in patients with pJIA but also because research in adults with lupus has found that leaving poverty was associated with a reversal of accrued disease damage.
Interactions of social determinants
The authors analyzed retrospective data from 1,684 pediatric patients in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry covering the period of April 2015 to February 2020. All study participants had been diagnosed with pJIA. Symptom onset occurred before age 16, and at least five joints were involved. The authors excluded patients who had been diagnosed with other systemic inflammatory or autoimmune diseases.
The authors defined exposure to a high level of community poverty as living in a ZIP code where at least 20% of residents lived at or below the federal poverty level. The authors also collected data on household income, although these data were missing for more than a quarter of participants (27%) and were therefore included only in sensitivity analyses. They used the clinical Juvenile Arthritis Disease Activity Score–10 (cJADAS-10) and the Child Health Assessment Questionnaire (CHAQ) to assess disease activity and disability at baseline and 6 and 12 months later. A cutoff of 2.5 on the cJADAS-10 distinguished mild disease activity from moderate to high disease activity, and a CHAQ score of 0.25 was the cutoff for having functional disability.
Among those who reported household income, just over half the cohort had an income of at least $50,000. The study population was 74% White, and more non-White patients lived in high-poverty communities (36.4%) than did White patients (21.3%). Patients whose families had no more than a high school education (23.1% vs. 13.7%) and those with public insurance (43.0% vs. 21.5%) were also over-represented in poorer communities.
The median cJADAS-10 scores declined overall during patients’ first year of therapy. However, those with public insurance, a lower family education level, or residency in poorer communities made up the greatest proportion of patients who continued to have moderate to severe disease activity a year after diagnosis.
The unadjusted calculations showed that children living in high community poverty had 1.8 times greater odds of functional disability (odds ratio, 1.82; P < .001). However, after adjustment for age, sex, race/ethnicity, insurance status, family education, rheumatoid factor, and cyclic citrullinated peptide antibody, the association lost statistical significance (P = .3). Community poverty level was not associated with disease activity before or after adjustment.
“Race was adjusted for as a confounder; however, the association between race/ethnicity and social determinants of health is likely more complex,” Dr. Feldman said. “Interactions, for example, between individual race and area-level poverty could be investigated.”
Odds of persistent function disability were 1.5 times greater for children with public insurance (adjusted OR, 1.56; P = .023) and 1.9 times greater for those whose families had a lower education level (aOR, 1.89; P = .013). Children whose race/ethnicity was indicated as being other than White had more than double the odds of higher disease activity (aOR, 2.48; P = .002) and were nearly twice as likely to have persistent functional disability (aOR, 1.91; P = .031).
Future directions
Dr. Soulsby was struck by the difference in statistical significance between individual-level poverty, as measured by household income, and community-level poverty. “It’s interesting because it may suggest that both of these forms of poverty are different and have different impacts on disease,” he said. Dr. Balmuri elaborated on the nuances and interactions that exist with social determinants of health and how objective outcomes, such as disease activity as measured by clinical tools, can differ from subjective outcomes, such as patients’ reports of pain, daily disability, and social experiences.
“The human condition is far more complicated, unfortunately, than any dataset could have on their own collected,” Dr. Balmuri said. She said she plans to expand her pJIA research into other social determinants of health. “It’s first about getting people’s eyes and minds open to something we see every day that, for some reason, sometimes people are blinded to, [using] the data that we do have, and then our hope is to build upon that.”
Dr. Feldman noted that ZIP codes, which were used as a proxy for community poverty, may not provide the best perspective regarding a patient’s neighborhood, because significant variation may exist within a single ZIP code, which is something the authors noted as well. The investigators were limited in the data available from the registry, and Dr. Balmuri and Dr. Soulsby suggested that 9-digit ZIP codes or census tracts might better capture neighborhood deprivation.
The research was funded by the Arthritis Foundation and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Feldman has received research support from Pfizer and the Bristol-Myers Squibb Foundation. Dr. Soulsby and Dr. Balmuri have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For children with polyarticular juvenile idiopathic arthritis (pJIA), functional disability lasts longer and disease activity is increased among those who belong to a racial/ethnic minority or come from homes with low household income or low family education, according to a study published online in Pediatric Rheumatology. The findings also initially revealed a higher likelihood of functional disability among those living in a poorer community, but that association lost statistical significance after adjustment for confounders.
“We chose community poverty level as the primary predictor for outcomes in pJIA because the socioeconomic context of communities and neighborhoods affects the characteristics of the social, service, and physical environments to which all residents are exposed regardless of their own socioeconomic position and may have a greater negative impact on those with fewer individual resources,” the authors write. “While community poverty level was not associated with an increase in odds of moderate-to-severe disease activity, those with high community poverty level did have higher disease activity scores (0.33 points greater on average than those with low community poverty level, in adjusted analysis).”
Nayimisha Balmuri, MD, an assistant professor of pediatrics at Johns Hopkins Medicine and study coauthor, told this news organization that anecdotal experience from everyday practice has shown that “patients with myriad social determinants of health stacked against them present sicker, take longer to present, and require far more aggressive therapies and follow-up,” which wreaks havoc in terms of disease activity. “It’s really difficult, then, to play catch-up to other cohorts of patients,” Dr. Balmuri added.
Disparities in outcomes persist
A key clinical take-home message from these findings is that the differences in clinical outcomes are relevant throughout the entire year of therapy, Dr. Balmuri said. “Patients get better; however, they don’t get better the same,” she said, and this is because of a variety of reasons. “Getting in the door is one of [those reasons] but then continuing to follow-up care is another.” For general practitioners, it’s especially important to refer patients who complain of joint pains to a specialist and to then follow up to be sure they’re improving and they’re getting the care they need.
For pediatric rheumatologists and subspecialists, “it’s important for us to realize that the disparity doesn’t end when patients come into your door to begin with,” Dr. Balmuri said. “It continues over the short term and far past that into adulthood.”
Candace Feldman, MD, MPH, ScD, an assistant professor of medicine in the Division of Rheumatology, Inflammation, and Immunity at Brigham and Women’s Hospital, Boston, told this news organization that the research “provides an important foundation to the study of the impact of social determinants of health on disease activity and disability among children with JIA. Individuals with rheumatic conditions should be screened for social determinants of health–related needs, and infrastructure should exist within the rheumatology clinic to help address the needs uncovered.” Dr. Feldman was not involved in the study.
In addition to the results’ clinical significance, Dr. Feldman also noted the policy implications of these findings. “Physicians should advocate for efforts to dismantle structural racism, to address income inequality, and to mitigate the effects of climate change, which also disproportionately affect historically marginalized populations,” Dr. Feldman said. Although this study focused predominantly on poverty, she noted that financial insecurity, food insecurity, homelessness, or housing instability were other social determinants of health to consider in future research.
Dr. Balmuri and William Daniel Soulsby, MD, a clinical fellow in pediatric rheumatology at the University of California, San Francisco, who is the study’s lead author, said they focused on poverty in this study not only because it’s so understudied in patients with pJIA but also because research in adults with lupus has found that leaving poverty was associated with a reversal of accrued disease damage.
Interactions of social determinants
The authors analyzed retrospective data from 1,684 pediatric patients in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry covering the period of April 2015 to February 2020. All study participants had been diagnosed with pJIA. Symptom onset occurred before age 16, and at least five joints were involved. The authors excluded patients who had been diagnosed with other systemic inflammatory or autoimmune diseases.
The authors defined exposure to a high level of community poverty as living in a ZIP code where at least 20% of residents lived at or below the federal poverty level. The authors also collected data on household income, although these data were missing for more than a quarter of participants (27%) and were therefore included only in sensitivity analyses. They used the clinical Juvenile Arthritis Disease Activity Score–10 (cJADAS-10) and the Child Health Assessment Questionnaire (CHAQ) to assess disease activity and disability at baseline and 6 and 12 months later. A cutoff of 2.5 on the cJADAS-10 distinguished mild disease activity from moderate to high disease activity, and a CHAQ score of 0.25 was the cutoff for having functional disability.
Among those who reported household income, just over half the cohort had an income of at least $50,000. The study population was 74% White, and more non-White patients lived in high-poverty communities (36.4%) than did White patients (21.3%). Patients whose families had no more than a high school education (23.1% vs. 13.7%) and those with public insurance (43.0% vs. 21.5%) were also over-represented in poorer communities.
The median cJADAS-10 scores declined overall during patients’ first year of therapy. However, those with public insurance, a lower family education level, or residency in poorer communities made up the greatest proportion of patients who continued to have moderate to severe disease activity a year after diagnosis.
The unadjusted calculations showed that children living in high community poverty had 1.8 times greater odds of functional disability (odds ratio, 1.82; P < .001). However, after adjustment for age, sex, race/ethnicity, insurance status, family education, rheumatoid factor, and cyclic citrullinated peptide antibody, the association lost statistical significance (P = .3). Community poverty level was not associated with disease activity before or after adjustment.
“Race was adjusted for as a confounder; however, the association between race/ethnicity and social determinants of health is likely more complex,” Dr. Feldman said. “Interactions, for example, between individual race and area-level poverty could be investigated.”
Odds of persistent function disability were 1.5 times greater for children with public insurance (adjusted OR, 1.56; P = .023) and 1.9 times greater for those whose families had a lower education level (aOR, 1.89; P = .013). Children whose race/ethnicity was indicated as being other than White had more than double the odds of higher disease activity (aOR, 2.48; P = .002) and were nearly twice as likely to have persistent functional disability (aOR, 1.91; P = .031).
Future directions
Dr. Soulsby was struck by the difference in statistical significance between individual-level poverty, as measured by household income, and community-level poverty. “It’s interesting because it may suggest that both of these forms of poverty are different and have different impacts on disease,” he said. Dr. Balmuri elaborated on the nuances and interactions that exist with social determinants of health and how objective outcomes, such as disease activity as measured by clinical tools, can differ from subjective outcomes, such as patients’ reports of pain, daily disability, and social experiences.
“The human condition is far more complicated, unfortunately, than any dataset could have on their own collected,” Dr. Balmuri said. She said she plans to expand her pJIA research into other social determinants of health. “It’s first about getting people’s eyes and minds open to something we see every day that, for some reason, sometimes people are blinded to, [using] the data that we do have, and then our hope is to build upon that.”
Dr. Feldman noted that ZIP codes, which were used as a proxy for community poverty, may not provide the best perspective regarding a patient’s neighborhood, because significant variation may exist within a single ZIP code, which is something the authors noted as well. The investigators were limited in the data available from the registry, and Dr. Balmuri and Dr. Soulsby suggested that 9-digit ZIP codes or census tracts might better capture neighborhood deprivation.
The research was funded by the Arthritis Foundation and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Feldman has received research support from Pfizer and the Bristol-Myers Squibb Foundation. Dr. Soulsby and Dr. Balmuri have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PEDIATRIC RHEUMATOLOGY




