User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
Suicide prevention, NRA team up for safer gun storage
WASHINGTON – A suicide prevention organization based at the University of Washington partnered with the National Rifle Association and other gun-user organizations to launch a state-wide program in Washington aimed at safe firearm storage to cut suicide rates.
with plans to expand these activities, Jennifer P. Stuber, Ph.D., said at the annual conference of the American Association of Suicidology.

The idea behind SAFER Homes came from recognition that a majority of suicides are caused by firearms, a majority of deaths from firearms are suicides, and that a potentially effective way to cut suicide numbers is by restricting unauthorized or unintended firearm access, especially to middle-aged men, said Dr. Stuber, a health policy researcher at the University of Washington, Seattle, and founder and policy director of Forefront Suicide Prevention, a social impact organization operated by the university’s school of social work. SAFER Homes also came into being as Dr. Stuber realized that she had a receptive audience on the issue of safe gun storage to prevent suicides among gun owners, gun retailers, and gun-rights proponents.
“There is remarkably little push back. These are folks who care about suicide. Seventy percent of gun-shop owners have experienced a personal suicide loss, and shop owners don’t want to be the one who sells someone a gun who then uses it to end their life,” Dr. Stuber said in an interview.
The Second Amendment Foundation gun-rights organization has been the main partner with Forefront Suicide Prevention in launching SAFER Homes Suicide Aware, but the campaign also received endorsement from the National Rifle Association. Dr. Stuber also worked with the Second Amendment Foundation and the National Rifle Association to get legislation passed in Washington to expand education on suicide prevention through improved gun safety. Dr. Stuber became an advocate for suicide prevention and safer gun storage following the firearm suicide of her husband in 2011, an experience she wrote about in a 2016 essay.

Preventing suicide through safer storage of firearms makes sense because the U.S. demographic subgroup with the highest number of suicides is men 35-64 years old. In this subgroup, the majority of those killed by suicide were never identified as depressed or having a mental health problem, and more than 80% never had a prior suicide attempt, said Jeffrey C. Sung, MD, a psychiatrist at the University of Washington who has worked with Dr. Stuber on the SAFER Homes campaign. He cited a study of 310 suicides among patients in the Veterans Health Administration system (93% men, 76% aged 50 years or older) that showed 72% reported having no suicide ideation on the most recent health questionnaire prior to their suicide (Psych Services. 2016 May;67[5]:517-22).
Because men like these do not come to clinical attention prior to their suicide, the best preventive strategy is to focus on improved safety measures to control potential means of lethality, said Dr. Sung, who is also immediate past president of the Washington State Psychiatric Society.
SAFER Homes “goes to community-based settings, like gun shows, where men in their middle years can be reached,” Dr. Stuber noted. By talking about taking steps to reduce the risk of firearm suicide with attendees at gun shows and with gun retailers, “you’re pushing on an open door,” Dr. Sung added.
Dr. Stuber and Dr. Sung had no disclosures.
SOURCE: Stuber J et al. Annual conference of the American Association of Suicidology.
WASHINGTON – A suicide prevention organization based at the University of Washington partnered with the National Rifle Association and other gun-user organizations to launch a state-wide program in Washington aimed at safe firearm storage to cut suicide rates.
with plans to expand these activities, Jennifer P. Stuber, Ph.D., said at the annual conference of the American Association of Suicidology.

The idea behind SAFER Homes came from recognition that a majority of suicides are caused by firearms, a majority of deaths from firearms are suicides, and that a potentially effective way to cut suicide numbers is by restricting unauthorized or unintended firearm access, especially to middle-aged men, said Dr. Stuber, a health policy researcher at the University of Washington, Seattle, and founder and policy director of Forefront Suicide Prevention, a social impact organization operated by the university’s school of social work. SAFER Homes also came into being as Dr. Stuber realized that she had a receptive audience on the issue of safe gun storage to prevent suicides among gun owners, gun retailers, and gun-rights proponents.
“There is remarkably little push back. These are folks who care about suicide. Seventy percent of gun-shop owners have experienced a personal suicide loss, and shop owners don’t want to be the one who sells someone a gun who then uses it to end their life,” Dr. Stuber said in an interview.
The Second Amendment Foundation gun-rights organization has been the main partner with Forefront Suicide Prevention in launching SAFER Homes Suicide Aware, but the campaign also received endorsement from the National Rifle Association. Dr. Stuber also worked with the Second Amendment Foundation and the National Rifle Association to get legislation passed in Washington to expand education on suicide prevention through improved gun safety. Dr. Stuber became an advocate for suicide prevention and safer gun storage following the firearm suicide of her husband in 2011, an experience she wrote about in a 2016 essay.

Preventing suicide through safer storage of firearms makes sense because the U.S. demographic subgroup with the highest number of suicides is men 35-64 years old. In this subgroup, the majority of those killed by suicide were never identified as depressed or having a mental health problem, and more than 80% never had a prior suicide attempt, said Jeffrey C. Sung, MD, a psychiatrist at the University of Washington who has worked with Dr. Stuber on the SAFER Homes campaign. He cited a study of 310 suicides among patients in the Veterans Health Administration system (93% men, 76% aged 50 years or older) that showed 72% reported having no suicide ideation on the most recent health questionnaire prior to their suicide (Psych Services. 2016 May;67[5]:517-22).
Because men like these do not come to clinical attention prior to their suicide, the best preventive strategy is to focus on improved safety measures to control potential means of lethality, said Dr. Sung, who is also immediate past president of the Washington State Psychiatric Society.
SAFER Homes “goes to community-based settings, like gun shows, where men in their middle years can be reached,” Dr. Stuber noted. By talking about taking steps to reduce the risk of firearm suicide with attendees at gun shows and with gun retailers, “you’re pushing on an open door,” Dr. Sung added.
Dr. Stuber and Dr. Sung had no disclosures.
SOURCE: Stuber J et al. Annual conference of the American Association of Suicidology.
WASHINGTON – A suicide prevention organization based at the University of Washington partnered with the National Rifle Association and other gun-user organizations to launch a state-wide program in Washington aimed at safe firearm storage to cut suicide rates.
with plans to expand these activities, Jennifer P. Stuber, Ph.D., said at the annual conference of the American Association of Suicidology.

The idea behind SAFER Homes came from recognition that a majority of suicides are caused by firearms, a majority of deaths from firearms are suicides, and that a potentially effective way to cut suicide numbers is by restricting unauthorized or unintended firearm access, especially to middle-aged men, said Dr. Stuber, a health policy researcher at the University of Washington, Seattle, and founder and policy director of Forefront Suicide Prevention, a social impact organization operated by the university’s school of social work. SAFER Homes also came into being as Dr. Stuber realized that she had a receptive audience on the issue of safe gun storage to prevent suicides among gun owners, gun retailers, and gun-rights proponents.
“There is remarkably little push back. These are folks who care about suicide. Seventy percent of gun-shop owners have experienced a personal suicide loss, and shop owners don’t want to be the one who sells someone a gun who then uses it to end their life,” Dr. Stuber said in an interview.
The Second Amendment Foundation gun-rights organization has been the main partner with Forefront Suicide Prevention in launching SAFER Homes Suicide Aware, but the campaign also received endorsement from the National Rifle Association. Dr. Stuber also worked with the Second Amendment Foundation and the National Rifle Association to get legislation passed in Washington to expand education on suicide prevention through improved gun safety. Dr. Stuber became an advocate for suicide prevention and safer gun storage following the firearm suicide of her husband in 2011, an experience she wrote about in a 2016 essay.

Preventing suicide through safer storage of firearms makes sense because the U.S. demographic subgroup with the highest number of suicides is men 35-64 years old. In this subgroup, the majority of those killed by suicide were never identified as depressed or having a mental health problem, and more than 80% never had a prior suicide attempt, said Jeffrey C. Sung, MD, a psychiatrist at the University of Washington who has worked with Dr. Stuber on the SAFER Homes campaign. He cited a study of 310 suicides among patients in the Veterans Health Administration system (93% men, 76% aged 50 years or older) that showed 72% reported having no suicide ideation on the most recent health questionnaire prior to their suicide (Psych Services. 2016 May;67[5]:517-22).
Because men like these do not come to clinical attention prior to their suicide, the best preventive strategy is to focus on improved safety measures to control potential means of lethality, said Dr. Sung, who is also immediate past president of the Washington State Psychiatric Society.
SAFER Homes “goes to community-based settings, like gun shows, where men in their middle years can be reached,” Dr. Stuber noted. By talking about taking steps to reduce the risk of firearm suicide with attendees at gun shows and with gun retailers, “you’re pushing on an open door,” Dr. Sung added.
Dr. Stuber and Dr. Sung had no disclosures.
SOURCE: Stuber J et al. Annual conference of the American Association of Suicidology.
EXPERT ANALYSIS FROM THE AAS ANNUAL CONFERENCE
VIDEO: Screening ECG patch boosts AF diagnoses ninefold
ORLANDO – An ECG patch worn twice for a total of about 24 days produced a nearly ninefold increase in the number of high-risk people diagnosed with atrial fibrillation, compared with those followed with usual care in a randomized trial with 2,655 people.
During 4 months of follow-up, 1,364 high-risk people assigned to ECG patch screening had a 5.1% rate of new atrial fibrillation (AF) diagnoses, compared with a 0.6% rate among 1,291 controls who wore the patch but were identified with new-onset AF using standard follow-up that did not take the patch data into account. This was a statistically significant difference for the study’s primary endpoint, Steven R. Steinhubl, MD, said at the annual meeting of the American College of Cardiology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In addition to proving that the ECG patch can better identify asymptomatic people who have AF than conventional means – usually waiting until a stroke occurs or for symptoms to appear – the noninvasive and relatively low-cost patch also gives researchers a new way to try to address the more fundamental medical question created by this line of investigation: How clinically important are relatively brief, asymptomatic episodes of atrial fibrillation, and are patient outcomes improved by treatments begun in this early phase?
The study results “show we can look beyond implantable devices with a less expensive, noninvasive way” to identify patients with asymptomatic AF and determine its natural history and need for intervention, Dr. Steinhubl said in a video interview.
The mSToP (mHealth Screening to Prevent Strokes) trial ran at Scripps and began by identifying more than 359,000 U.S. residents with Aetna health insurance who met the study’s definition of having high AF risk, either by being at least 75 years old, or at least 55 years old and male or at least 65 years old and female. To qualify as high risk those younger than 75 years also had to have at least one clinical risk factor, which could include a prior cerebrovascular event, heart failure, hypertension plus diabetes, obstructive sleep apnea, or any of six other comorbidities. The researchers also excluded potential participants because of several factors, including a history of atrial fibrillation or flutter, current treatment with an anticoagulant, end-stage renal disease, and patients with an implanted pacemaker or defibrillator.
They invited more than 100,000 of these qualifying Aetna beneficiaries to participate, and 2,655 agreed and received by mail a pair of ECG measurement patches (Zio) with instructions to wear one for 2 weeks at the start of the study and to wear the second during the final 2 weeks of the 4-month study period. The participants averaged 73 years of age, and their average CHA2DS2-VASc score was 3.
All patients in the study were told to wear their patches and mail them in, but the researchers used the collected ECG data for diagnosing AF in only the 1,364 patients randomized to the active arm. The ECG findings for the 1,291 controls wasn’t provided to their physicians, and so any new-onset AF had to be found either by symptom onset or incidentally. About one-third of the people assigned to each of the study arms never wore their patches. Those who wore their patches did so for an average of nearly 12 days each. Diagnosis of new-onset AF was based on finding either at least one AF episode recorded by the patches that lasted at least 30 seconds or an AF diagnosis appearing in the patient’s record. The average AF burden – the percentage of time a person with incident AF had an abnormal sinus rhythm – was 0.9%.
Even though many patients did not use their patches, the investigators assessed the primary endpoint of new AF diagnoses during the 4-month study period on an intention-to-treat basis. Their analysis showed an 8.8-fold higher rate of new AF diagnoses among people in the intervention arm whose patch data were used for immediate diagnosis, reported Dr. Steinhubl, an interventional cardiologist and director of digital medicine at the Scripps Translational Science Institute in La Jolla, Ca.
As a secondary endpoint, the researchers merged the entire group of 1,738 participants who had sent in patches with ECG data and compared their 1-year incidence of diagnosed AF against 3,476 matched controls from the Aetna database. After 1 year, the rate of new AF diagnoses was 6.3% in those with patch information and 2.3% among the controls, a threefold difference in diagnosis rates after adjustment for potential confounders.
“The clinical significance of the short AF episodes” manifested by many patch users identified with AF “requires greater clarity, especially in terms of stroke risk,” Dr. Steinhubl said. But he added, “I like to think that, as we learn more, we can look at more than just anticoagulation” as intervention options. For example, if a morbidly obese patient has asymptomatic AF found by patch screening, it might strengthen the case for bariatric surgery if it’s eventually shown that weight loss after bariatric surgery slows AF progression. The same holds true for more aggressive sleep apnea intervention in patients with sleep apnea and asymptomatic AF, as well as for patients with asymptomatic AF and another type of associated comorbidity.
SOURCE: Steinhubl S. ACC 18, Abstract 402-19.
Results from several studies have now shown that some kind of monitoring for AF in asymptomatic, at-risk people results in an increased diagnosis of subclinical AF. Study results also suggest that, in general, people diagnosed with subclinical AF are at a lower risk of stroke than patients with symptomatic AF. As of now, no prospective study has evaluated the efficacy of anticoagulant therapy in people diagnosed with subclinical AF, although such studies are now in progress. Until we have these results, the question of how to manage patients with subclinical AF remains unanswered.
N.A. Mark Estes, MD , is professor of medicine and director of the New England Cardiac Arrhythmia Center at Tufts Medical Center in Boston. He has been a consultant to Boston Scientific, Medtronic, and St. Jude. He made these comments as designated discussant for the mSToPS report.
Results from several studies have now shown that some kind of monitoring for AF in asymptomatic, at-risk people results in an increased diagnosis of subclinical AF. Study results also suggest that, in general, people diagnosed with subclinical AF are at a lower risk of stroke than patients with symptomatic AF. As of now, no prospective study has evaluated the efficacy of anticoagulant therapy in people diagnosed with subclinical AF, although such studies are now in progress. Until we have these results, the question of how to manage patients with subclinical AF remains unanswered.
N.A. Mark Estes, MD , is professor of medicine and director of the New England Cardiac Arrhythmia Center at Tufts Medical Center in Boston. He has been a consultant to Boston Scientific, Medtronic, and St. Jude. He made these comments as designated discussant for the mSToPS report.
Results from several studies have now shown that some kind of monitoring for AF in asymptomatic, at-risk people results in an increased diagnosis of subclinical AF. Study results also suggest that, in general, people diagnosed with subclinical AF are at a lower risk of stroke than patients with symptomatic AF. As of now, no prospective study has evaluated the efficacy of anticoagulant therapy in people diagnosed with subclinical AF, although such studies are now in progress. Until we have these results, the question of how to manage patients with subclinical AF remains unanswered.
N.A. Mark Estes, MD , is professor of medicine and director of the New England Cardiac Arrhythmia Center at Tufts Medical Center in Boston. He has been a consultant to Boston Scientific, Medtronic, and St. Jude. He made these comments as designated discussant for the mSToPS report.
ORLANDO – An ECG patch worn twice for a total of about 24 days produced a nearly ninefold increase in the number of high-risk people diagnosed with atrial fibrillation, compared with those followed with usual care in a randomized trial with 2,655 people.
During 4 months of follow-up, 1,364 high-risk people assigned to ECG patch screening had a 5.1% rate of new atrial fibrillation (AF) diagnoses, compared with a 0.6% rate among 1,291 controls who wore the patch but were identified with new-onset AF using standard follow-up that did not take the patch data into account. This was a statistically significant difference for the study’s primary endpoint, Steven R. Steinhubl, MD, said at the annual meeting of the American College of Cardiology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In addition to proving that the ECG patch can better identify asymptomatic people who have AF than conventional means – usually waiting until a stroke occurs or for symptoms to appear – the noninvasive and relatively low-cost patch also gives researchers a new way to try to address the more fundamental medical question created by this line of investigation: How clinically important are relatively brief, asymptomatic episodes of atrial fibrillation, and are patient outcomes improved by treatments begun in this early phase?
The study results “show we can look beyond implantable devices with a less expensive, noninvasive way” to identify patients with asymptomatic AF and determine its natural history and need for intervention, Dr. Steinhubl said in a video interview.
The mSToP (mHealth Screening to Prevent Strokes) trial ran at Scripps and began by identifying more than 359,000 U.S. residents with Aetna health insurance who met the study’s definition of having high AF risk, either by being at least 75 years old, or at least 55 years old and male or at least 65 years old and female. To qualify as high risk those younger than 75 years also had to have at least one clinical risk factor, which could include a prior cerebrovascular event, heart failure, hypertension plus diabetes, obstructive sleep apnea, or any of six other comorbidities. The researchers also excluded potential participants because of several factors, including a history of atrial fibrillation or flutter, current treatment with an anticoagulant, end-stage renal disease, and patients with an implanted pacemaker or defibrillator.
They invited more than 100,000 of these qualifying Aetna beneficiaries to participate, and 2,655 agreed and received by mail a pair of ECG measurement patches (Zio) with instructions to wear one for 2 weeks at the start of the study and to wear the second during the final 2 weeks of the 4-month study period. The participants averaged 73 years of age, and their average CHA2DS2-VASc score was 3.
All patients in the study were told to wear their patches and mail them in, but the researchers used the collected ECG data for diagnosing AF in only the 1,364 patients randomized to the active arm. The ECG findings for the 1,291 controls wasn’t provided to their physicians, and so any new-onset AF had to be found either by symptom onset or incidentally. About one-third of the people assigned to each of the study arms never wore their patches. Those who wore their patches did so for an average of nearly 12 days each. Diagnosis of new-onset AF was based on finding either at least one AF episode recorded by the patches that lasted at least 30 seconds or an AF diagnosis appearing in the patient’s record. The average AF burden – the percentage of time a person with incident AF had an abnormal sinus rhythm – was 0.9%.
Even though many patients did not use their patches, the investigators assessed the primary endpoint of new AF diagnoses during the 4-month study period on an intention-to-treat basis. Their analysis showed an 8.8-fold higher rate of new AF diagnoses among people in the intervention arm whose patch data were used for immediate diagnosis, reported Dr. Steinhubl, an interventional cardiologist and director of digital medicine at the Scripps Translational Science Institute in La Jolla, Ca.
As a secondary endpoint, the researchers merged the entire group of 1,738 participants who had sent in patches with ECG data and compared their 1-year incidence of diagnosed AF against 3,476 matched controls from the Aetna database. After 1 year, the rate of new AF diagnoses was 6.3% in those with patch information and 2.3% among the controls, a threefold difference in diagnosis rates after adjustment for potential confounders.
“The clinical significance of the short AF episodes” manifested by many patch users identified with AF “requires greater clarity, especially in terms of stroke risk,” Dr. Steinhubl said. But he added, “I like to think that, as we learn more, we can look at more than just anticoagulation” as intervention options. For example, if a morbidly obese patient has asymptomatic AF found by patch screening, it might strengthen the case for bariatric surgery if it’s eventually shown that weight loss after bariatric surgery slows AF progression. The same holds true for more aggressive sleep apnea intervention in patients with sleep apnea and asymptomatic AF, as well as for patients with asymptomatic AF and another type of associated comorbidity.
SOURCE: Steinhubl S. ACC 18, Abstract 402-19.
ORLANDO – An ECG patch worn twice for a total of about 24 days produced a nearly ninefold increase in the number of high-risk people diagnosed with atrial fibrillation, compared with those followed with usual care in a randomized trial with 2,655 people.
During 4 months of follow-up, 1,364 high-risk people assigned to ECG patch screening had a 5.1% rate of new atrial fibrillation (AF) diagnoses, compared with a 0.6% rate among 1,291 controls who wore the patch but were identified with new-onset AF using standard follow-up that did not take the patch data into account. This was a statistically significant difference for the study’s primary endpoint, Steven R. Steinhubl, MD, said at the annual meeting of the American College of Cardiology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In addition to proving that the ECG patch can better identify asymptomatic people who have AF than conventional means – usually waiting until a stroke occurs or for symptoms to appear – the noninvasive and relatively low-cost patch also gives researchers a new way to try to address the more fundamental medical question created by this line of investigation: How clinically important are relatively brief, asymptomatic episodes of atrial fibrillation, and are patient outcomes improved by treatments begun in this early phase?
The study results “show we can look beyond implantable devices with a less expensive, noninvasive way” to identify patients with asymptomatic AF and determine its natural history and need for intervention, Dr. Steinhubl said in a video interview.
The mSToP (mHealth Screening to Prevent Strokes) trial ran at Scripps and began by identifying more than 359,000 U.S. residents with Aetna health insurance who met the study’s definition of having high AF risk, either by being at least 75 years old, or at least 55 years old and male or at least 65 years old and female. To qualify as high risk those younger than 75 years also had to have at least one clinical risk factor, which could include a prior cerebrovascular event, heart failure, hypertension plus diabetes, obstructive sleep apnea, or any of six other comorbidities. The researchers also excluded potential participants because of several factors, including a history of atrial fibrillation or flutter, current treatment with an anticoagulant, end-stage renal disease, and patients with an implanted pacemaker or defibrillator.
They invited more than 100,000 of these qualifying Aetna beneficiaries to participate, and 2,655 agreed and received by mail a pair of ECG measurement patches (Zio) with instructions to wear one for 2 weeks at the start of the study and to wear the second during the final 2 weeks of the 4-month study period. The participants averaged 73 years of age, and their average CHA2DS2-VASc score was 3.
All patients in the study were told to wear their patches and mail them in, but the researchers used the collected ECG data for diagnosing AF in only the 1,364 patients randomized to the active arm. The ECG findings for the 1,291 controls wasn’t provided to their physicians, and so any new-onset AF had to be found either by symptom onset or incidentally. About one-third of the people assigned to each of the study arms never wore their patches. Those who wore their patches did so for an average of nearly 12 days each. Diagnosis of new-onset AF was based on finding either at least one AF episode recorded by the patches that lasted at least 30 seconds or an AF diagnosis appearing in the patient’s record. The average AF burden – the percentage of time a person with incident AF had an abnormal sinus rhythm – was 0.9%.
Even though many patients did not use their patches, the investigators assessed the primary endpoint of new AF diagnoses during the 4-month study period on an intention-to-treat basis. Their analysis showed an 8.8-fold higher rate of new AF diagnoses among people in the intervention arm whose patch data were used for immediate diagnosis, reported Dr. Steinhubl, an interventional cardiologist and director of digital medicine at the Scripps Translational Science Institute in La Jolla, Ca.
As a secondary endpoint, the researchers merged the entire group of 1,738 participants who had sent in patches with ECG data and compared their 1-year incidence of diagnosed AF against 3,476 matched controls from the Aetna database. After 1 year, the rate of new AF diagnoses was 6.3% in those with patch information and 2.3% among the controls, a threefold difference in diagnosis rates after adjustment for potential confounders.
“The clinical significance of the short AF episodes” manifested by many patch users identified with AF “requires greater clarity, especially in terms of stroke risk,” Dr. Steinhubl said. But he added, “I like to think that, as we learn more, we can look at more than just anticoagulation” as intervention options. For example, if a morbidly obese patient has asymptomatic AF found by patch screening, it might strengthen the case for bariatric surgery if it’s eventually shown that weight loss after bariatric surgery slows AF progression. The same holds true for more aggressive sleep apnea intervention in patients with sleep apnea and asymptomatic AF, as well as for patients with asymptomatic AF and another type of associated comorbidity.
SOURCE: Steinhubl S. ACC 18, Abstract 402-19.
REPORTING FROM ACC 18
Key clinical point: .
Major finding: After 4 months, new AF diagnoses occurred in 5.1% of patch users and 0.6% of usual-care controls.
Study details: mSToPS, a single-center, randomized study with 2,655 people at high risk for developing AF.
Disclosures: mSToPS received support from Aetna, Janssen, and iRhythm. Dr. Steinhubl has been an advisor to Airstrip, DynoSense, EasyG, FocusMotion, LifeWatch, MyoKardia, Novartis, and Spry Health, he serves on the board of Celes Health, and he has received research support from Janssen and Novartis.
Source: Steinhubl S. ACC 18, Abstract 402-19.
Disproportionately low U.S. research funding targets gynecologic cancers
NEW ORLEANS – The National Cancer Institute is woefully underfunding gynecologic cancer research, compared with several other cancer types, when the money the institute is spending annually is factored by the incidence and lethal impact of each cancer using U.S. data from 2007 to 2014.
That period featured “systematic and pervasive underfunding of gynecologic cancers in relation to other cancer sites,” Ryan J. Spencer, MD, said at the annual meeting of the Society of Gynecologic Oncology. The trends over the period he studied worsened with time and pose the risk that progress in gynecologic cancers – uterine, cervical, and ovarian – will “lag behind” other cancers’ progress in prevention, treatment, and improved survival, said Dr. Spencer, a gynecologic oncologist at the University of Wisconsin–Madison.
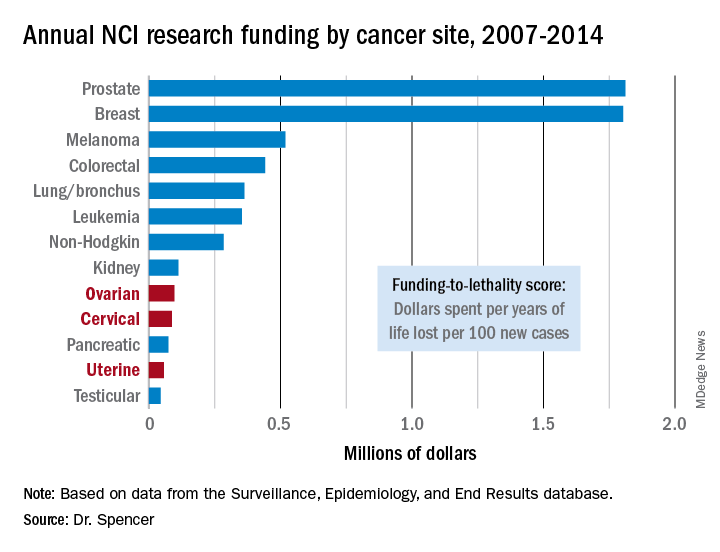
Additional time trend analyses showed that the annual funding-to-lethality score for each of the three gynecologic cancers declined during the period studied.
“We must do everything we can to reverse these trends,” Dr. Spencer concluded.
SOURCE: Spencer R et al. SGO 2018, Abstract 3.
The data reported by Dr. Spencer and his associates are very sobering. They present an elegant analysis that documents a lag and decline in funding for gynecologic cancers that factors in the lethality of various cancers. By several other measures as well, funding for research into gynecologic cancers has been slipping in recent years. During 2011-2016, we saw a 90% drop in enrollment into U.S. clinical trials for gynecologic cancers, and from a peak in 2012-2016 the total number of trials for gynecologic cancers fell by more than two-thirds.
Paola A. Gehrig, MD , is professor of ob.gyn. and director of gynecologic oncology at the University of North Carolina at Chapel Hill. She had no disclosures. She made these comments as designated discussant for the report.
The data reported by Dr. Spencer and his associates are very sobering. They present an elegant analysis that documents a lag and decline in funding for gynecologic cancers that factors in the lethality of various cancers. By several other measures as well, funding for research into gynecologic cancers has been slipping in recent years. During 2011-2016, we saw a 90% drop in enrollment into U.S. clinical trials for gynecologic cancers, and from a peak in 2012-2016 the total number of trials for gynecologic cancers fell by more than two-thirds.
Paola A. Gehrig, MD , is professor of ob.gyn. and director of gynecologic oncology at the University of North Carolina at Chapel Hill. She had no disclosures. She made these comments as designated discussant for the report.
The data reported by Dr. Spencer and his associates are very sobering. They present an elegant analysis that documents a lag and decline in funding for gynecologic cancers that factors in the lethality of various cancers. By several other measures as well, funding for research into gynecologic cancers has been slipping in recent years. During 2011-2016, we saw a 90% drop in enrollment into U.S. clinical trials for gynecologic cancers, and from a peak in 2012-2016 the total number of trials for gynecologic cancers fell by more than two-thirds.
Paola A. Gehrig, MD , is professor of ob.gyn. and director of gynecologic oncology at the University of North Carolina at Chapel Hill. She had no disclosures. She made these comments as designated discussant for the report.
NEW ORLEANS – The National Cancer Institute is woefully underfunding gynecologic cancer research, compared with several other cancer types, when the money the institute is spending annually is factored by the incidence and lethal impact of each cancer using U.S. data from 2007 to 2014.
That period featured “systematic and pervasive underfunding of gynecologic cancers in relation to other cancer sites,” Ryan J. Spencer, MD, said at the annual meeting of the Society of Gynecologic Oncology. The trends over the period he studied worsened with time and pose the risk that progress in gynecologic cancers – uterine, cervical, and ovarian – will “lag behind” other cancers’ progress in prevention, treatment, and improved survival, said Dr. Spencer, a gynecologic oncologist at the University of Wisconsin–Madison.

Additional time trend analyses showed that the annual funding-to-lethality score for each of the three gynecologic cancers declined during the period studied.
“We must do everything we can to reverse these trends,” Dr. Spencer concluded.
SOURCE: Spencer R et al. SGO 2018, Abstract 3.
NEW ORLEANS – The National Cancer Institute is woefully underfunding gynecologic cancer research, compared with several other cancer types, when the money the institute is spending annually is factored by the incidence and lethal impact of each cancer using U.S. data from 2007 to 2014.
That period featured “systematic and pervasive underfunding of gynecologic cancers in relation to other cancer sites,” Ryan J. Spencer, MD, said at the annual meeting of the Society of Gynecologic Oncology. The trends over the period he studied worsened with time and pose the risk that progress in gynecologic cancers – uterine, cervical, and ovarian – will “lag behind” other cancers’ progress in prevention, treatment, and improved survival, said Dr. Spencer, a gynecologic oncologist at the University of Wisconsin–Madison.

Additional time trend analyses showed that the annual funding-to-lethality score for each of the three gynecologic cancers declined during the period studied.
“We must do everything we can to reverse these trends,” Dr. Spencer concluded.
SOURCE: Spencer R et al. SGO 2018, Abstract 3.
REPORTING FROM SGO 2018
Key clinical point: The National Cancer Institute underfunds gynecologic cancer research.
Major finding: Ovarian cancer research funding averaged $97,000 per year of life lost per 100 new cases, compared with $1.8 million for both breast and prostate cancer.
Study details: A review of U.S. data collected by the National Cancer Institute during 2007-2014.
Disclosures: Dr. Spencer had no disclosures.
Source: Spencer R et al. SGO 2018, Abstract 3.
VIDEO: Indocyanine green finds more sentinel lymph nodes
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – Indocyanine green (ICG) worked better than isosulfan blue for mapping sentinel lymph nodes (SLNs) in a pivotal phase 3 trial with 176 patients who had stage I endometrial or cervical cancer.
Four injections of ICG resulted in detection of 96% of the identified SLNs in these patients, including bilateral SLNs in 78% of the patients. In contrast, four injections with isosulfan blue dye led to detection of 74% of all SLNs and identified bilateral SLNs in 31% of the patients, Michael M. Frumovitz, MD, said at the annual meeting of the Society for Gynecologic Oncology.
The FILM trial randomized 176 patients with stage I endometrial or cervical cancer at eight centers in the United States or Canada between December 2015 and May 2017. Patients first received one of the tagging agents and then the second, and then underwent mapping using white light to detect blue-tagged SLNs and near-infrared light to find green-tagged SLNs. The patients were aged 63 years on average, and 96% had endometrial cancer.
The researchers identified 279 sentinel lymph nodes that stained only green, nine SLNs that stained only blue, and 248 SLNs tagged with both dyes. They confirmed tumor cells within all nine of SLNs tagged with blue dye only, in 95% of those tagged with ICG only, and in 92% of the SLNs stained with both dyes. The isosulfan blue dye identified SLNs in two patients who did not have any SLNs detected by the ICG, whereas the ICG identified SLNs in 22 patients who did not have any SLNs detected using the blue dye. Sixteen patients had metastatic disease that had moved to 21 SLNs. The ICG system identified all 21 involved lymph nodes; the blue dye identified 13 of the 21 affected SLNs (62%).
Dr. Frumovitz and his associates designed FILM as primarily a test of noninferiority. The per-protocol analysis with 163 patients showed that ICG was noninferior to isosulfan blue (P less than .001). Once the results demonstrated noninferiority, the study protocol allowed the researchers to test for superiority in the full, intention-to-treat cohort of 176 patients. The results showed that ICG was significantly superior to isosulfan blue (P less than .001). In addition, ICG treatment produced no allergic or other adverse reactions, Dr. Frumovitz said.
Once ICG and the associated near-infrared detection camera receive FDA marketing approval, “I think this will become the standard within 5 years,” he predicted in an interview.
The results also showed that using both ICG and isosulfan blue was not better than using ICG alone. “If you’re using both dyes, you can drop the blue dye. At MD Anderson we’ve used only ICG for about the past year,” Dr. Frumovitz said.
FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
SOURCE: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
The results from the FILM trial are potentially practice changing. The findings presented by Michael M. Frumovitz, MD, and his associates showed that indocyanine green is superior to isosulfan blue dye for mapping sentinel lymph nodes in patients with stage I endometrial or cervical cancer. The results also showed that using both dyes was no better than using indocyanine green alone.
The report by Floor J. Backes, MD, addressed an important and still unresolved question in treating patients with stage I or II endometrial cancer: What is the significance of finding isolated tumor cells in sentinel lymph nodes in these patients? The retrospective findings she presented showed that the presence of isolated tumor cells had no apparent effect on recurrence-free survival, recurrence pattern, or patient response to various treatments. This suggested th at treatment decisions in these patients should depend on other high-risk uterine factors but not on whether some lymph nodes contained isolated tumor cells.
Brent Smith, MD , is a gynecologic oncologist at the Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The results from the FILM trial are potentially practice changing. The findings presented by Michael M. Frumovitz, MD, and his associates showed that indocyanine green is superior to isosulfan blue dye for mapping sentinel lymph nodes in patients with stage I endometrial or cervical cancer. The results also showed that using both dyes was no better than using indocyanine green alone.
The report by Floor J. Backes, MD, addressed an important and still unresolved question in treating patients with stage I or II endometrial cancer: What is the significance of finding isolated tumor cells in sentinel lymph nodes in these patients? The retrospective findings she presented showed that the presence of isolated tumor cells had no apparent effect on recurrence-free survival, recurrence pattern, or patient response to various treatments. This suggested th at treatment decisions in these patients should depend on other high-risk uterine factors but not on whether some lymph nodes contained isolated tumor cells.
Brent Smith, MD , is a gynecologic oncologist at the Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The results from the FILM trial are potentially practice changing. The findings presented by Michael M. Frumovitz, MD, and his associates showed that indocyanine green is superior to isosulfan blue dye for mapping sentinel lymph nodes in patients with stage I endometrial or cervical cancer. The results also showed that using both dyes was no better than using indocyanine green alone.
The report by Floor J. Backes, MD, addressed an important and still unresolved question in treating patients with stage I or II endometrial cancer: What is the significance of finding isolated tumor cells in sentinel lymph nodes in these patients? The retrospective findings she presented showed that the presence of isolated tumor cells had no apparent effect on recurrence-free survival, recurrence pattern, or patient response to various treatments. This suggested th at treatment decisions in these patients should depend on other high-risk uterine factors but not on whether some lymph nodes contained isolated tumor cells.
Brent Smith, MD , is a gynecologic oncologist at the Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – Indocyanine green (ICG) worked better than isosulfan blue for mapping sentinel lymph nodes (SLNs) in a pivotal phase 3 trial with 176 patients who had stage I endometrial or cervical cancer.
Four injections of ICG resulted in detection of 96% of the identified SLNs in these patients, including bilateral SLNs in 78% of the patients. In contrast, four injections with isosulfan blue dye led to detection of 74% of all SLNs and identified bilateral SLNs in 31% of the patients, Michael M. Frumovitz, MD, said at the annual meeting of the Society for Gynecologic Oncology.
The FILM trial randomized 176 patients with stage I endometrial or cervical cancer at eight centers in the United States or Canada between December 2015 and May 2017. Patients first received one of the tagging agents and then the second, and then underwent mapping using white light to detect blue-tagged SLNs and near-infrared light to find green-tagged SLNs. The patients were aged 63 years on average, and 96% had endometrial cancer.
The researchers identified 279 sentinel lymph nodes that stained only green, nine SLNs that stained only blue, and 248 SLNs tagged with both dyes. They confirmed tumor cells within all nine of SLNs tagged with blue dye only, in 95% of those tagged with ICG only, and in 92% of the SLNs stained with both dyes. The isosulfan blue dye identified SLNs in two patients who did not have any SLNs detected by the ICG, whereas the ICG identified SLNs in 22 patients who did not have any SLNs detected using the blue dye. Sixteen patients had metastatic disease that had moved to 21 SLNs. The ICG system identified all 21 involved lymph nodes; the blue dye identified 13 of the 21 affected SLNs (62%).
Dr. Frumovitz and his associates designed FILM as primarily a test of noninferiority. The per-protocol analysis with 163 patients showed that ICG was noninferior to isosulfan blue (P less than .001). Once the results demonstrated noninferiority, the study protocol allowed the researchers to test for superiority in the full, intention-to-treat cohort of 176 patients. The results showed that ICG was significantly superior to isosulfan blue (P less than .001). In addition, ICG treatment produced no allergic or other adverse reactions, Dr. Frumovitz said.
Once ICG and the associated near-infrared detection camera receive FDA marketing approval, “I think this will become the standard within 5 years,” he predicted in an interview.
The results also showed that using both ICG and isosulfan blue was not better than using ICG alone. “If you’re using both dyes, you can drop the blue dye. At MD Anderson we’ve used only ICG for about the past year,” Dr. Frumovitz said.
FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
SOURCE: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – Indocyanine green (ICG) worked better than isosulfan blue for mapping sentinel lymph nodes (SLNs) in a pivotal phase 3 trial with 176 patients who had stage I endometrial or cervical cancer.
Four injections of ICG resulted in detection of 96% of the identified SLNs in these patients, including bilateral SLNs in 78% of the patients. In contrast, four injections with isosulfan blue dye led to detection of 74% of all SLNs and identified bilateral SLNs in 31% of the patients, Michael M. Frumovitz, MD, said at the annual meeting of the Society for Gynecologic Oncology.
The FILM trial randomized 176 patients with stage I endometrial or cervical cancer at eight centers in the United States or Canada between December 2015 and May 2017. Patients first received one of the tagging agents and then the second, and then underwent mapping using white light to detect blue-tagged SLNs and near-infrared light to find green-tagged SLNs. The patients were aged 63 years on average, and 96% had endometrial cancer.
The researchers identified 279 sentinel lymph nodes that stained only green, nine SLNs that stained only blue, and 248 SLNs tagged with both dyes. They confirmed tumor cells within all nine of SLNs tagged with blue dye only, in 95% of those tagged with ICG only, and in 92% of the SLNs stained with both dyes. The isosulfan blue dye identified SLNs in two patients who did not have any SLNs detected by the ICG, whereas the ICG identified SLNs in 22 patients who did not have any SLNs detected using the blue dye. Sixteen patients had metastatic disease that had moved to 21 SLNs. The ICG system identified all 21 involved lymph nodes; the blue dye identified 13 of the 21 affected SLNs (62%).
Dr. Frumovitz and his associates designed FILM as primarily a test of noninferiority. The per-protocol analysis with 163 patients showed that ICG was noninferior to isosulfan blue (P less than .001). Once the results demonstrated noninferiority, the study protocol allowed the researchers to test for superiority in the full, intention-to-treat cohort of 176 patients. The results showed that ICG was significantly superior to isosulfan blue (P less than .001). In addition, ICG treatment produced no allergic or other adverse reactions, Dr. Frumovitz said.
Once ICG and the associated near-infrared detection camera receive FDA marketing approval, “I think this will become the standard within 5 years,” he predicted in an interview.
The results also showed that using both ICG and isosulfan blue was not better than using ICG alone. “If you’re using both dyes, you can drop the blue dye. At MD Anderson we’ve used only ICG for about the past year,” Dr. Frumovitz said.
FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
SOURCE: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
REPORTING FROM SGO 2018
Key clinical point: Indocyanine green surpassed isosulfan blue for sentinel lymph node mapping in a pivotal trial.
Major finding: Researchers mapped sentinel lymph nodes in 96% of patients with indocyanine green and in 74% with isosulfan blue.
Study details: FILM, a multicenter, randomized phase 3 trial with 176 patients.
Disclosures: FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
Source: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
Trastuzumab plus chemo shows efficacy for high HER2 endometrial cancer
NEW ORLEANS – Adding the anti-HER2 antibody trastuzumab to a standard chemotherapy regimen produced markedly improved responses among patients with an advanced stage or recurrent uterine serous carcinoma that also overexpressed the HER2/neu cell receptor protein in a controlled, multicenter, phase 2 study with 58 evaluable patients.
The 30 patients who received trastuzumab along with carboplatin plus paclitaxel had a median progression-free survival of 12.6 months compared with 8.0 months among the 28 control patients who received only carboplatin plus paclitaxel, a hazard reduction of 56% with trastuzumab that was statistically significant (P = .0052), Alessandro D. Santin, MD, said at the annual meeting of the Society of Gynecologic Oncology.
In the subgroup of 41 patients with advanced (not recurrent) uterine serous carcinoma (USC) the incremental difference in median progression-free survival among patients treated with trastuzumab was 8.6 months, based on a 17.9-month duration in the 20 patients who received trastuzumab and 9.3 months in 20 control patients on chemotherapy only (P = .013).
“We’ll push to have the trastuzumab, carboplatin, and paclitaxel regimen in the guidelines” for treating patients with USC that overexpresses HER2/neu, agreed Amanda N. Fader, MD, director of gynecologic oncology at Johns Hopkins Medicine in Baltimore and lead author on the study reported by Dr. Santin.
Shortly after Dr. Santin’s report at the meeting, the results appeared in an article published online March 27 in Journal of Clinical Oncology.
The phase 2 trial ran at 11 U.S. centers during 2011-2016, and randomized 61 patients with stage III or IV or recurrent USC and high HER2 protein expression measured by immunohistochemistry and amplification of the HER2 gene shown by fluorescence in situ hybridization. The researchers collected data from 58 treated patients. Among the 17 patients with recurrent disease, those who received trastuzumab had a median progression-free survival of 9.2 months compared with 6.0 months in the controls, a hazard reduction of 86% with trastuzumab that was statistically significant (P = .0029). The addition of trastuzumab to carboplatin and paclitaxel was well tolerated.
The greatest benefit from the trastuzumab plus chemotherapy regimen might occur when used as first-line treatment, Dr. Santin suggested. He expressed interest in running a second study to validate the current findings, and a new study that would combine a second antibody directed against HER2, pertuzumab (Perjeta) with trastuzumab, carboplatin, and paclitaxel.
A key factor in making the trastuzumab plus chemotherapy regimen more widely available to USC patients would be routinely screening these patients for high levels of HER2/neu expression immediately after USC is diagnosed. “At Yale and Johns Hopkins this testing is done on all USC patients. That’s not standard care everywhere, but it should be based on these data,” Dr. Santin said in an interview.
SOURCE: Fader A et al. Abstract 22. J Clin Oncol. 2018 Mar 27. doi: 10.1200/JCO.2017.76.5966.
NEW ORLEANS – Adding the anti-HER2 antibody trastuzumab to a standard chemotherapy regimen produced markedly improved responses among patients with an advanced stage or recurrent uterine serous carcinoma that also overexpressed the HER2/neu cell receptor protein in a controlled, multicenter, phase 2 study with 58 evaluable patients.
The 30 patients who received trastuzumab along with carboplatin plus paclitaxel had a median progression-free survival of 12.6 months compared with 8.0 months among the 28 control patients who received only carboplatin plus paclitaxel, a hazard reduction of 56% with trastuzumab that was statistically significant (P = .0052), Alessandro D. Santin, MD, said at the annual meeting of the Society of Gynecologic Oncology.
In the subgroup of 41 patients with advanced (not recurrent) uterine serous carcinoma (USC) the incremental difference in median progression-free survival among patients treated with trastuzumab was 8.6 months, based on a 17.9-month duration in the 20 patients who received trastuzumab and 9.3 months in 20 control patients on chemotherapy only (P = .013).
“We’ll push to have the trastuzumab, carboplatin, and paclitaxel regimen in the guidelines” for treating patients with USC that overexpresses HER2/neu, agreed Amanda N. Fader, MD, director of gynecologic oncology at Johns Hopkins Medicine in Baltimore and lead author on the study reported by Dr. Santin.
Shortly after Dr. Santin’s report at the meeting, the results appeared in an article published online March 27 in Journal of Clinical Oncology.
The phase 2 trial ran at 11 U.S. centers during 2011-2016, and randomized 61 patients with stage III or IV or recurrent USC and high HER2 protein expression measured by immunohistochemistry and amplification of the HER2 gene shown by fluorescence in situ hybridization. The researchers collected data from 58 treated patients. Among the 17 patients with recurrent disease, those who received trastuzumab had a median progression-free survival of 9.2 months compared with 6.0 months in the controls, a hazard reduction of 86% with trastuzumab that was statistically significant (P = .0029). The addition of trastuzumab to carboplatin and paclitaxel was well tolerated.
The greatest benefit from the trastuzumab plus chemotherapy regimen might occur when used as first-line treatment, Dr. Santin suggested. He expressed interest in running a second study to validate the current findings, and a new study that would combine a second antibody directed against HER2, pertuzumab (Perjeta) with trastuzumab, carboplatin, and paclitaxel.
A key factor in making the trastuzumab plus chemotherapy regimen more widely available to USC patients would be routinely screening these patients for high levels of HER2/neu expression immediately after USC is diagnosed. “At Yale and Johns Hopkins this testing is done on all USC patients. That’s not standard care everywhere, but it should be based on these data,” Dr. Santin said in an interview.
SOURCE: Fader A et al. Abstract 22. J Clin Oncol. 2018 Mar 27. doi: 10.1200/JCO.2017.76.5966.
NEW ORLEANS – Adding the anti-HER2 antibody trastuzumab to a standard chemotherapy regimen produced markedly improved responses among patients with an advanced stage or recurrent uterine serous carcinoma that also overexpressed the HER2/neu cell receptor protein in a controlled, multicenter, phase 2 study with 58 evaluable patients.
The 30 patients who received trastuzumab along with carboplatin plus paclitaxel had a median progression-free survival of 12.6 months compared with 8.0 months among the 28 control patients who received only carboplatin plus paclitaxel, a hazard reduction of 56% with trastuzumab that was statistically significant (P = .0052), Alessandro D. Santin, MD, said at the annual meeting of the Society of Gynecologic Oncology.
In the subgroup of 41 patients with advanced (not recurrent) uterine serous carcinoma (USC) the incremental difference in median progression-free survival among patients treated with trastuzumab was 8.6 months, based on a 17.9-month duration in the 20 patients who received trastuzumab and 9.3 months in 20 control patients on chemotherapy only (P = .013).
“We’ll push to have the trastuzumab, carboplatin, and paclitaxel regimen in the guidelines” for treating patients with USC that overexpresses HER2/neu, agreed Amanda N. Fader, MD, director of gynecologic oncology at Johns Hopkins Medicine in Baltimore and lead author on the study reported by Dr. Santin.
Shortly after Dr. Santin’s report at the meeting, the results appeared in an article published online March 27 in Journal of Clinical Oncology.
The phase 2 trial ran at 11 U.S. centers during 2011-2016, and randomized 61 patients with stage III or IV or recurrent USC and high HER2 protein expression measured by immunohistochemistry and amplification of the HER2 gene shown by fluorescence in situ hybridization. The researchers collected data from 58 treated patients. Among the 17 patients with recurrent disease, those who received trastuzumab had a median progression-free survival of 9.2 months compared with 6.0 months in the controls, a hazard reduction of 86% with trastuzumab that was statistically significant (P = .0029). The addition of trastuzumab to carboplatin and paclitaxel was well tolerated.
The greatest benefit from the trastuzumab plus chemotherapy regimen might occur when used as first-line treatment, Dr. Santin suggested. He expressed interest in running a second study to validate the current findings, and a new study that would combine a second antibody directed against HER2, pertuzumab (Perjeta) with trastuzumab, carboplatin, and paclitaxel.
A key factor in making the trastuzumab plus chemotherapy regimen more widely available to USC patients would be routinely screening these patients for high levels of HER2/neu expression immediately after USC is diagnosed. “At Yale and Johns Hopkins this testing is done on all USC patients. That’s not standard care everywhere, but it should be based on these data,” Dr. Santin said in an interview.
SOURCE: Fader A et al. Abstract 22. J Clin Oncol. 2018 Mar 27. doi: 10.1200/JCO.2017.76.5966.
REPORTING FROM SGO 2018
Key clinical point: Trastuzumab plus chemotherapy improved progression-free survival in a phase 2 study.
Major finding: Median progression-free survival was 12.6 months with trastuzumab and 8.0 months in controls.
Study details: A multicenter, phase 2 study that enrolled 58 evaluable patients.
Disclosures: Trastuzumab was provided at no charge by Genentech. Dr. Santin and Dr. Fader had no relevant disclosures.
Source: Fader A et al. Abstract 22. J Clin Oncol. 2018 Mar 27. doi: 10.1200/JCO.2017.76.5966)
views
The biological relevance of HER2 amplification has been clearly established in the results from this and other studies. But we must keep in kind that the results reported by Dr. Santin have applicability to a subset of a subset of patients. Specifically, about 10% of endometrial cancers are uterine serous carcinoma (USC) (although they account for roughly 40% of deaths among endometrial cancer patients), about half of USC patients show a high level of HER2/neu expression by immunohistochemistry, and about 60% of these high-expressing USC also have HER2 gene amplification. The study presented by Dr. Santin ran at 11 centers for 5.5 years and enrolled just 61 patients, fewer than the planned enrollment target of 100 patients.
Michael A. Bookman, MD , is director of gynecologic oncology therapeutics at the Permanente Medical Group in San Francisco. He has been an adviser to AstraZeneca, Bayer, Clovis, Merck, Pfizer, and Tesaro, and he has participated in trials funded by Abbvie, Genentech, Immunogen, Mateon, and Roche. He made these comments as designated discussant for the study.
Residual single-site ovarian cancer surpasses multisite outcomes
NEW ORLEANS – When complete resection of advanced-stage, epithelial ovarian cancer is not possible, surgical resection that leaves a small volume of residual tumor at a single site produces significantly better outcomes than leaving minimal residual cancer at multiple sites, according to a review of 510 patients at two U.S. centers.
“In the past, we separated these patients based on whether they had a complete resection, R0 disease, or had 1 cm or less of residual disease” regardless of the number of sites with this small amount of residual tumor. The third category was patients with more than 1 cm of residual tumor at one or more sites, explained Dr. Manning-Geist in an interview. “What we did was break down the patients with 1 cm or less of residual tumor into those with one site or multiple sites. This is the first reported study to use number of sites” as a clinical characteristic for analysis in this context.
The message from the findings is that, while the goal of debulking surgery in patients with advanced epithelial ovarian cancer is complete tumor resection, if that can’t be achieved, the next goal is to leave residual tumor at just a single site, she concluded. A question that remains is whether primary debulking surgery is preferable to neoadjuvant treatment followed by interval debulking surgery. In the results Dr. Manning-Geist presented, patients who underwent primary debulking had better outcomes than those with neoadjuvant therapy followed by interval debulking, but these two subgroups also had different clinical characteristics.
The study used data from 510 patients with stage IIIC or IV epithelial ovarian cancer treated at either Brigham and Women’s or Massachusetts General Hospital during 2010-2015. The study cohort included 240 patients who underwent primary debulking surgery and 270 who first received neoadjuvant chemotherapy and then underwent interval debulking surgery. The patients who received neoadjuvant therapy were, on average, older (65 years vs. 63 years), had a higher prevalence of stage IV disease (44% vs. 16%), and had a higher prevalence of tumors with serous histology (93% vs. 77%), compared with patients who underwent primary debulking.
Complete tumor resections occurred in 39% of the primary debulking patients and in 64% of those who received neoadjuvant therapy; residual disease of 1 cm or less at one site occurred in 17% and 13%, respectively; minimal residual disease at multiple sites remained in 28% and 17% respectively; and the remaining patients had residual disease of more than 1 cm in at least one site, 16% and 6% respectively.
For this analysis, Dr. Manning-Geist and her associates considered residual disease at any of seven possible sites: diaphragm, upper abdomen, bowel mesentary, bowel serosa, abdominal peritoneum, pelvis, and nodal. Even if multiple individual metastases remained within one of these sites after surgery, it was categorized as a single site of residual disease.
Among patients who underwent primary debulking surgery, progression-free survival persisted for a median of 23 months among patients with full resection, 19 months in patients with a single site with minimal residual disease, 13 months among those with multiple sites of residual disease, and 10 months in patients with more than 1 cm of residual tumor. Median overall survival in these four subgroups was not yet reached, 64 months, 50 months, and 49 months, respectively.
Among patients who received neoadjuvant chemotherapy and then underwent interval debulking surgery, median durations of progression-free survival were 14 months, 12 months, 10 months, and 6 months, respectively. Median overall survival rates were 58 months, 37 months, 26 months, and 33 months, respectively. Within each of these four analyses, the differences in both survival and progression-free survival across the four subgroups was statistically significant, with a P less than .001 for each analysis.
In multivariate analyses, among patients who underwent primary debulking surgery, the significant linkages with worsening progression-free and overall survival were age, cancer stage, and amount and site number of residual disease. Among patients who received neoadjuvant chemotherapy followed by interval debulking residual disease diameter and site number of residual tumor was the only significant determinant for both progression-free and overall survival, Dr. Manning-Geist reported.
Dr. Manning-Geist had no disclosures.
SOURCE: Manning-Geist B et al. SGO 2018, Abstract 43.
NEW ORLEANS – When complete resection of advanced-stage, epithelial ovarian cancer is not possible, surgical resection that leaves a small volume of residual tumor at a single site produces significantly better outcomes than leaving minimal residual cancer at multiple sites, according to a review of 510 patients at two U.S. centers.
“In the past, we separated these patients based on whether they had a complete resection, R0 disease, or had 1 cm or less of residual disease” regardless of the number of sites with this small amount of residual tumor. The third category was patients with more than 1 cm of residual tumor at one or more sites, explained Dr. Manning-Geist in an interview. “What we did was break down the patients with 1 cm or less of residual tumor into those with one site or multiple sites. This is the first reported study to use number of sites” as a clinical characteristic for analysis in this context.
The message from the findings is that, while the goal of debulking surgery in patients with advanced epithelial ovarian cancer is complete tumor resection, if that can’t be achieved, the next goal is to leave residual tumor at just a single site, she concluded. A question that remains is whether primary debulking surgery is preferable to neoadjuvant treatment followed by interval debulking surgery. In the results Dr. Manning-Geist presented, patients who underwent primary debulking had better outcomes than those with neoadjuvant therapy followed by interval debulking, but these two subgroups also had different clinical characteristics.
The study used data from 510 patients with stage IIIC or IV epithelial ovarian cancer treated at either Brigham and Women’s or Massachusetts General Hospital during 2010-2015. The study cohort included 240 patients who underwent primary debulking surgery and 270 who first received neoadjuvant chemotherapy and then underwent interval debulking surgery. The patients who received neoadjuvant therapy were, on average, older (65 years vs. 63 years), had a higher prevalence of stage IV disease (44% vs. 16%), and had a higher prevalence of tumors with serous histology (93% vs. 77%), compared with patients who underwent primary debulking.
Complete tumor resections occurred in 39% of the primary debulking patients and in 64% of those who received neoadjuvant therapy; residual disease of 1 cm or less at one site occurred in 17% and 13%, respectively; minimal residual disease at multiple sites remained in 28% and 17% respectively; and the remaining patients had residual disease of more than 1 cm in at least one site, 16% and 6% respectively.
For this analysis, Dr. Manning-Geist and her associates considered residual disease at any of seven possible sites: diaphragm, upper abdomen, bowel mesentary, bowel serosa, abdominal peritoneum, pelvis, and nodal. Even if multiple individual metastases remained within one of these sites after surgery, it was categorized as a single site of residual disease.
Among patients who underwent primary debulking surgery, progression-free survival persisted for a median of 23 months among patients with full resection, 19 months in patients with a single site with minimal residual disease, 13 months among those with multiple sites of residual disease, and 10 months in patients with more than 1 cm of residual tumor. Median overall survival in these four subgroups was not yet reached, 64 months, 50 months, and 49 months, respectively.
Among patients who received neoadjuvant chemotherapy and then underwent interval debulking surgery, median durations of progression-free survival were 14 months, 12 months, 10 months, and 6 months, respectively. Median overall survival rates were 58 months, 37 months, 26 months, and 33 months, respectively. Within each of these four analyses, the differences in both survival and progression-free survival across the four subgroups was statistically significant, with a P less than .001 for each analysis.
In multivariate analyses, among patients who underwent primary debulking surgery, the significant linkages with worsening progression-free and overall survival were age, cancer stage, and amount and site number of residual disease. Among patients who received neoadjuvant chemotherapy followed by interval debulking residual disease diameter and site number of residual tumor was the only significant determinant for both progression-free and overall survival, Dr. Manning-Geist reported.
Dr. Manning-Geist had no disclosures.
SOURCE: Manning-Geist B et al. SGO 2018, Abstract 43.
NEW ORLEANS – When complete resection of advanced-stage, epithelial ovarian cancer is not possible, surgical resection that leaves a small volume of residual tumor at a single site produces significantly better outcomes than leaving minimal residual cancer at multiple sites, according to a review of 510 patients at two U.S. centers.
“In the past, we separated these patients based on whether they had a complete resection, R0 disease, or had 1 cm or less of residual disease” regardless of the number of sites with this small amount of residual tumor. The third category was patients with more than 1 cm of residual tumor at one or more sites, explained Dr. Manning-Geist in an interview. “What we did was break down the patients with 1 cm or less of residual tumor into those with one site or multiple sites. This is the first reported study to use number of sites” as a clinical characteristic for analysis in this context.
The message from the findings is that, while the goal of debulking surgery in patients with advanced epithelial ovarian cancer is complete tumor resection, if that can’t be achieved, the next goal is to leave residual tumor at just a single site, she concluded. A question that remains is whether primary debulking surgery is preferable to neoadjuvant treatment followed by interval debulking surgery. In the results Dr. Manning-Geist presented, patients who underwent primary debulking had better outcomes than those with neoadjuvant therapy followed by interval debulking, but these two subgroups also had different clinical characteristics.
The study used data from 510 patients with stage IIIC or IV epithelial ovarian cancer treated at either Brigham and Women’s or Massachusetts General Hospital during 2010-2015. The study cohort included 240 patients who underwent primary debulking surgery and 270 who first received neoadjuvant chemotherapy and then underwent interval debulking surgery. The patients who received neoadjuvant therapy were, on average, older (65 years vs. 63 years), had a higher prevalence of stage IV disease (44% vs. 16%), and had a higher prevalence of tumors with serous histology (93% vs. 77%), compared with patients who underwent primary debulking.
Complete tumor resections occurred in 39% of the primary debulking patients and in 64% of those who received neoadjuvant therapy; residual disease of 1 cm or less at one site occurred in 17% and 13%, respectively; minimal residual disease at multiple sites remained in 28% and 17% respectively; and the remaining patients had residual disease of more than 1 cm in at least one site, 16% and 6% respectively.
For this analysis, Dr. Manning-Geist and her associates considered residual disease at any of seven possible sites: diaphragm, upper abdomen, bowel mesentary, bowel serosa, abdominal peritoneum, pelvis, and nodal. Even if multiple individual metastases remained within one of these sites after surgery, it was categorized as a single site of residual disease.
Among patients who underwent primary debulking surgery, progression-free survival persisted for a median of 23 months among patients with full resection, 19 months in patients with a single site with minimal residual disease, 13 months among those with multiple sites of residual disease, and 10 months in patients with more than 1 cm of residual tumor. Median overall survival in these four subgroups was not yet reached, 64 months, 50 months, and 49 months, respectively.
Among patients who received neoadjuvant chemotherapy and then underwent interval debulking surgery, median durations of progression-free survival were 14 months, 12 months, 10 months, and 6 months, respectively. Median overall survival rates were 58 months, 37 months, 26 months, and 33 months, respectively. Within each of these four analyses, the differences in both survival and progression-free survival across the four subgroups was statistically significant, with a P less than .001 for each analysis.
In multivariate analyses, among patients who underwent primary debulking surgery, the significant linkages with worsening progression-free and overall survival were age, cancer stage, and amount and site number of residual disease. Among patients who received neoadjuvant chemotherapy followed by interval debulking residual disease diameter and site number of residual tumor was the only significant determinant for both progression-free and overall survival, Dr. Manning-Geist reported.
Dr. Manning-Geist had no disclosures.
SOURCE: Manning-Geist B et al. SGO 2018, Abstract 43.
REPORTING FROM SGO 2018
Key clinical point:
Major finding: Median overall survival after primary debulking was 64 months with single-site residual disease and 50 months with multisite disease.
Study details: Retrospective review of 510 patients from two U.S. centers.
Disclosures: Dr. Manning-Geist had no disclosures.
Source: Manning-Geist BL et al. SGO 2018, Abstract 43.
VIDEO: Interventions target opioid overprescribing after gynecologic surgery
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – U.S. clinicians prescribe opioid tablets to postsurgical patients too often and at too high a pill count, according to results from two independent studies that examined prescribing patterns and opioid use in patients following gynecologic surgery.
In addition, “setting preoperative expectations about pain management led to increased compliance at discharge,” said Dr. Mark, a gynecologic oncologist at Roswell Park Comprehensive Cancer Center in Buffalo, N.Y.
Findings from the second study, of 122 women who underwent gynecologic surgery at Women and Infants Hospital in Providence, R.I., showed that 32% did not use any opioids for pain following hospital discharge, and that opioid use during hospitalization was a significant predictor of postdischarge opioid needs. This finding provided a way to devise a new prescribing guide for postsurgical patients based on their opioid use while hospitalized, said Erica Weston, MD, a gynecologic oncologist at Johns Hopkins University, Baltimore.

“No question, we are overprescribing,” Dr. Dowdy said, and described a program he and his colleagues at Mayo recently put in place that capped routine opioid pill prescriptions following various surgeries based on historic patient needs. For example, most laparotomy patients receive a prescription for 10 opioid doses on discharge. Based on the first 6 months of this program, it’s on track to cut the annual number of opioid tablets prescribed to postsurgical patients at Mayo by 35,000 for all gynecologic surgeries and by 1.5 million tablets for all Mayo surgical subspecialties, he said.
The study reported by Dr. Mark ran after the Roswell Park gynecologic oncology department implemented new guidelines for dispensing pain control medications following surgery. The guidelines called for comprehensive teaching for patients about pain expectations and pain management both before and after surgery and also established four dispensing categories:
- Patients undergoing minimally invasive or outpatient surgery and with no history of chronic pain and low opioid need while hospitalized received the default dispense of 600 mg ibuprofen every 6 hours as needed for 7 days and 500 mg acetaminophen every 6 hours as needed for 7 days.
- Patients who underwent this surgery but required 5 or more opioid tablets while hospitalized or those with a history of chronic pain and opioid use received the ibuprofen and acetaminophen regimen plus 12 opioid tablets, a 3-day supply with 1 tablet taken every 6 hours as needed.
- Patients who underwent laparotomy and had no chronic pain and opioid history and low opioid use while hospitalized received the ibuprofen and acetaminophen regimen plus 12 opioid tablets, a 3-day supply.
- Patients who underwent laparotomy and showed a higher opioid need based on their use during the 24 hours before discharge received the ibuprofen and acetaminophen regimen plus 24 opioid tablets for 3 days so they could take 2 tablets every 6 hours as needed.
Dr. Mark and his associates collected data from 337 patients managed with these guidelines during June 2017–January 2018 and compared them with 626 patients who underwent gynecologic surgery at Roswell Park during July 2016–June 2017. The data showed the average number of opioid tablets dispensed per patient for all discharges fell from 31.7 before the new guideline to 3.5, an 89% reduction. For the subgroup of patients who had undergone a laparotomy, the average pill number fell from 43.6 to 11.6, a 72% drop. Among patients treated with minimally invasive or outpatient surgery, average tablets dispensed fell from 28.1 before to 0.9 after, a 97% reduction. The reduction among opioid-naive patients was 90%, and it was 83% among patients who used opioids prior to their surgery.
Under the new program, patients requested an opioid refill 14% of the time after laparotomy and 8% of the time after minimally invasive surgery, rates that did not significantly differ from the prior era. Average postoperative pain scores were identical among patients treated under the new dispensing guidelines and those treated during the prior years, and 96% of patients said they were satisfied with the care they received during the new, restricted dispensing period, Dr. Mark reported.
The single-center experience reviewed by Dr. Weston tracked opioid use by 122 women who underwent a minimally invasive hysterectomy at Women and Infants both as inpatients and out to both 1-2 weeks and 4-6 weeks following discharge. The patients were an average age of 61 years, and included 16% who reported chronic pain and 5% with a history of chronic opioid use.
During the inpatient phase, median opioid use was three doses, with 25% of the patients using no opioids. During the first 1-2 weeks following discharge, median opioid use was nine tablets, with 37% of the patients using no opioids. By the 4- to 6-week follow-up (which collected data from 114 of the patients), median opioid use was a cumulative 11 tablets with 67% of the patients reporting no opioid use during the time between their first and second follow-up visit. During the total postdischarge period, 90% of the patients used 30 or fewer opioid tablets.
A multivariate analysis of the findings showed that opioid use while in hospital was the only significant predictor of opioid use after discharge. Age of 65 years or older showed a nonsignificant trend toward less postdischarge opioid use.
Based on these data Dr. Weston and her associates proposed a formula for estimating a patient’s opioid needs at discharge: Gynecologic surgery patients who needed no opioid medication as inpatients could receive 1-5 opioid tablets at discharge, patients who used opioids at or below the median level should receive 10-15 tablets at discharge, and those who used more than the median number of opioid tablets as inpatients should receive 25-30 tablets at discharge. For patients who undergo surgery as outpatients and have no record of pain medication needs, Dr. Weston recommended discharging them with a prescription for 25-30 tablets, possibly reducing this to 10-15 tablets for patients aged 65 years or older.
Dr. Mark, Dr. Weston, and Dr. Dowdy had no disclosures.
SOURCE: Mark J et al. SGO 2018, Abstract 7. Weston E et al. SGO 2018, Abstract 8.
The important and thought provoking reports by Dr. Mark and by Dr. Weston and their associates present innovative ways for clinicians to address the opioid crisis by prescribing fewer narcotics to postsurgical patients when they leave the hospital. Their work suggests that doing this can have little or no negative impact on patient satisfaction with their care. Their findings give us important documentation for prescribing fewer opioid tablets, while still giving patients adequate pain relief.

Their findings also provide clear documentation that, in current practice, without guidance like this opioids are often overprescribed, not out of negligence but because clinicians are simply not sure what a patient will need once they go home following gynecologic surgery. By reviewing the pain medication a patient needed in hospital we can better estimate what patients will need when they go home, and we can better avoid giving patients more opioid tablets than they will really need.
Brent Smith, MD , is a gynecologic oncologist at Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The important and thought provoking reports by Dr. Mark and by Dr. Weston and their associates present innovative ways for clinicians to address the opioid crisis by prescribing fewer narcotics to postsurgical patients when they leave the hospital. Their work suggests that doing this can have little or no negative impact on patient satisfaction with their care. Their findings give us important documentation for prescribing fewer opioid tablets, while still giving patients adequate pain relief.

Their findings also provide clear documentation that, in current practice, without guidance like this opioids are often overprescribed, not out of negligence but because clinicians are simply not sure what a patient will need once they go home following gynecologic surgery. By reviewing the pain medication a patient needed in hospital we can better estimate what patients will need when they go home, and we can better avoid giving patients more opioid tablets than they will really need.
Brent Smith, MD , is a gynecologic oncologist at Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The important and thought provoking reports by Dr. Mark and by Dr. Weston and their associates present innovative ways for clinicians to address the opioid crisis by prescribing fewer narcotics to postsurgical patients when they leave the hospital. Their work suggests that doing this can have little or no negative impact on patient satisfaction with their care. Their findings give us important documentation for prescribing fewer opioid tablets, while still giving patients adequate pain relief.

Their findings also provide clear documentation that, in current practice, without guidance like this opioids are often overprescribed, not out of negligence but because clinicians are simply not sure what a patient will need once they go home following gynecologic surgery. By reviewing the pain medication a patient needed in hospital we can better estimate what patients will need when they go home, and we can better avoid giving patients more opioid tablets than they will really need.
Brent Smith, MD , is a gynecologic oncologist at Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – U.S. clinicians prescribe opioid tablets to postsurgical patients too often and at too high a pill count, according to results from two independent studies that examined prescribing patterns and opioid use in patients following gynecologic surgery.
In addition, “setting preoperative expectations about pain management led to increased compliance at discharge,” said Dr. Mark, a gynecologic oncologist at Roswell Park Comprehensive Cancer Center in Buffalo, N.Y.
Findings from the second study, of 122 women who underwent gynecologic surgery at Women and Infants Hospital in Providence, R.I., showed that 32% did not use any opioids for pain following hospital discharge, and that opioid use during hospitalization was a significant predictor of postdischarge opioid needs. This finding provided a way to devise a new prescribing guide for postsurgical patients based on their opioid use while hospitalized, said Erica Weston, MD, a gynecologic oncologist at Johns Hopkins University, Baltimore.

“No question, we are overprescribing,” Dr. Dowdy said, and described a program he and his colleagues at Mayo recently put in place that capped routine opioid pill prescriptions following various surgeries based on historic patient needs. For example, most laparotomy patients receive a prescription for 10 opioid doses on discharge. Based on the first 6 months of this program, it’s on track to cut the annual number of opioid tablets prescribed to postsurgical patients at Mayo by 35,000 for all gynecologic surgeries and by 1.5 million tablets for all Mayo surgical subspecialties, he said.
The study reported by Dr. Mark ran after the Roswell Park gynecologic oncology department implemented new guidelines for dispensing pain control medications following surgery. The guidelines called for comprehensive teaching for patients about pain expectations and pain management both before and after surgery and also established four dispensing categories:
- Patients undergoing minimally invasive or outpatient surgery and with no history of chronic pain and low opioid need while hospitalized received the default dispense of 600 mg ibuprofen every 6 hours as needed for 7 days and 500 mg acetaminophen every 6 hours as needed for 7 days.
- Patients who underwent this surgery but required 5 or more opioid tablets while hospitalized or those with a history of chronic pain and opioid use received the ibuprofen and acetaminophen regimen plus 12 opioid tablets, a 3-day supply with 1 tablet taken every 6 hours as needed.
- Patients who underwent laparotomy and had no chronic pain and opioid history and low opioid use while hospitalized received the ibuprofen and acetaminophen regimen plus 12 opioid tablets, a 3-day supply.
- Patients who underwent laparotomy and showed a higher opioid need based on their use during the 24 hours before discharge received the ibuprofen and acetaminophen regimen plus 24 opioid tablets for 3 days so they could take 2 tablets every 6 hours as needed.
Dr. Mark and his associates collected data from 337 patients managed with these guidelines during June 2017–January 2018 and compared them with 626 patients who underwent gynecologic surgery at Roswell Park during July 2016–June 2017. The data showed the average number of opioid tablets dispensed per patient for all discharges fell from 31.7 before the new guideline to 3.5, an 89% reduction. For the subgroup of patients who had undergone a laparotomy, the average pill number fell from 43.6 to 11.6, a 72% drop. Among patients treated with minimally invasive or outpatient surgery, average tablets dispensed fell from 28.1 before to 0.9 after, a 97% reduction. The reduction among opioid-naive patients was 90%, and it was 83% among patients who used opioids prior to their surgery.
Under the new program, patients requested an opioid refill 14% of the time after laparotomy and 8% of the time after minimally invasive surgery, rates that did not significantly differ from the prior era. Average postoperative pain scores were identical among patients treated under the new dispensing guidelines and those treated during the prior years, and 96% of patients said they were satisfied with the care they received during the new, restricted dispensing period, Dr. Mark reported.
The single-center experience reviewed by Dr. Weston tracked opioid use by 122 women who underwent a minimally invasive hysterectomy at Women and Infants both as inpatients and out to both 1-2 weeks and 4-6 weeks following discharge. The patients were an average age of 61 years, and included 16% who reported chronic pain and 5% with a history of chronic opioid use.
During the inpatient phase, median opioid use was three doses, with 25% of the patients using no opioids. During the first 1-2 weeks following discharge, median opioid use was nine tablets, with 37% of the patients using no opioids. By the 4- to 6-week follow-up (which collected data from 114 of the patients), median opioid use was a cumulative 11 tablets with 67% of the patients reporting no opioid use during the time between their first and second follow-up visit. During the total postdischarge period, 90% of the patients used 30 or fewer opioid tablets.
A multivariate analysis of the findings showed that opioid use while in hospital was the only significant predictor of opioid use after discharge. Age of 65 years or older showed a nonsignificant trend toward less postdischarge opioid use.
Based on these data Dr. Weston and her associates proposed a formula for estimating a patient’s opioid needs at discharge: Gynecologic surgery patients who needed no opioid medication as inpatients could receive 1-5 opioid tablets at discharge, patients who used opioids at or below the median level should receive 10-15 tablets at discharge, and those who used more than the median number of opioid tablets as inpatients should receive 25-30 tablets at discharge. For patients who undergo surgery as outpatients and have no record of pain medication needs, Dr. Weston recommended discharging them with a prescription for 25-30 tablets, possibly reducing this to 10-15 tablets for patients aged 65 years or older.
Dr. Mark, Dr. Weston, and Dr. Dowdy had no disclosures.
SOURCE: Mark J et al. SGO 2018, Abstract 7. Weston E et al. SGO 2018, Abstract 8.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – U.S. clinicians prescribe opioid tablets to postsurgical patients too often and at too high a pill count, according to results from two independent studies that examined prescribing patterns and opioid use in patients following gynecologic surgery.
In addition, “setting preoperative expectations about pain management led to increased compliance at discharge,” said Dr. Mark, a gynecologic oncologist at Roswell Park Comprehensive Cancer Center in Buffalo, N.Y.
Findings from the second study, of 122 women who underwent gynecologic surgery at Women and Infants Hospital in Providence, R.I., showed that 32% did not use any opioids for pain following hospital discharge, and that opioid use during hospitalization was a significant predictor of postdischarge opioid needs. This finding provided a way to devise a new prescribing guide for postsurgical patients based on their opioid use while hospitalized, said Erica Weston, MD, a gynecologic oncologist at Johns Hopkins University, Baltimore.

“No question, we are overprescribing,” Dr. Dowdy said, and described a program he and his colleagues at Mayo recently put in place that capped routine opioid pill prescriptions following various surgeries based on historic patient needs. For example, most laparotomy patients receive a prescription for 10 opioid doses on discharge. Based on the first 6 months of this program, it’s on track to cut the annual number of opioid tablets prescribed to postsurgical patients at Mayo by 35,000 for all gynecologic surgeries and by 1.5 million tablets for all Mayo surgical subspecialties, he said.
The study reported by Dr. Mark ran after the Roswell Park gynecologic oncology department implemented new guidelines for dispensing pain control medications following surgery. The guidelines called for comprehensive teaching for patients about pain expectations and pain management both before and after surgery and also established four dispensing categories:
- Patients undergoing minimally invasive or outpatient surgery and with no history of chronic pain and low opioid need while hospitalized received the default dispense of 600 mg ibuprofen every 6 hours as needed for 7 days and 500 mg acetaminophen every 6 hours as needed for 7 days.
- Patients who underwent this surgery but required 5 or more opioid tablets while hospitalized or those with a history of chronic pain and opioid use received the ibuprofen and acetaminophen regimen plus 12 opioid tablets, a 3-day supply with 1 tablet taken every 6 hours as needed.
- Patients who underwent laparotomy and had no chronic pain and opioid history and low opioid use while hospitalized received the ibuprofen and acetaminophen regimen plus 12 opioid tablets, a 3-day supply.
- Patients who underwent laparotomy and showed a higher opioid need based on their use during the 24 hours before discharge received the ibuprofen and acetaminophen regimen plus 24 opioid tablets for 3 days so they could take 2 tablets every 6 hours as needed.
Dr. Mark and his associates collected data from 337 patients managed with these guidelines during June 2017–January 2018 and compared them with 626 patients who underwent gynecologic surgery at Roswell Park during July 2016–June 2017. The data showed the average number of opioid tablets dispensed per patient for all discharges fell from 31.7 before the new guideline to 3.5, an 89% reduction. For the subgroup of patients who had undergone a laparotomy, the average pill number fell from 43.6 to 11.6, a 72% drop. Among patients treated with minimally invasive or outpatient surgery, average tablets dispensed fell from 28.1 before to 0.9 after, a 97% reduction. The reduction among opioid-naive patients was 90%, and it was 83% among patients who used opioids prior to their surgery.
Under the new program, patients requested an opioid refill 14% of the time after laparotomy and 8% of the time after minimally invasive surgery, rates that did not significantly differ from the prior era. Average postoperative pain scores were identical among patients treated under the new dispensing guidelines and those treated during the prior years, and 96% of patients said they were satisfied with the care they received during the new, restricted dispensing period, Dr. Mark reported.
The single-center experience reviewed by Dr. Weston tracked opioid use by 122 women who underwent a minimally invasive hysterectomy at Women and Infants both as inpatients and out to both 1-2 weeks and 4-6 weeks following discharge. The patients were an average age of 61 years, and included 16% who reported chronic pain and 5% with a history of chronic opioid use.
During the inpatient phase, median opioid use was three doses, with 25% of the patients using no opioids. During the first 1-2 weeks following discharge, median opioid use was nine tablets, with 37% of the patients using no opioids. By the 4- to 6-week follow-up (which collected data from 114 of the patients), median opioid use was a cumulative 11 tablets with 67% of the patients reporting no opioid use during the time between their first and second follow-up visit. During the total postdischarge period, 90% of the patients used 30 or fewer opioid tablets.
A multivariate analysis of the findings showed that opioid use while in hospital was the only significant predictor of opioid use after discharge. Age of 65 years or older showed a nonsignificant trend toward less postdischarge opioid use.
Based on these data Dr. Weston and her associates proposed a formula for estimating a patient’s opioid needs at discharge: Gynecologic surgery patients who needed no opioid medication as inpatients could receive 1-5 opioid tablets at discharge, patients who used opioids at or below the median level should receive 10-15 tablets at discharge, and those who used more than the median number of opioid tablets as inpatients should receive 25-30 tablets at discharge. For patients who undergo surgery as outpatients and have no record of pain medication needs, Dr. Weston recommended discharging them with a prescription for 25-30 tablets, possibly reducing this to 10-15 tablets for patients aged 65 years or older.
Dr. Mark, Dr. Weston, and Dr. Dowdy had no disclosures.
SOURCE: Mark J et al. SGO 2018, Abstract 7. Weston E et al. SGO 2018, Abstract 8.
REPORTING FROM SGO 2018
Key clinical point: Patients who underwent gynecologic surgery received adequate pain relief when receiving fewer opioid tablets.
Major finding: A protocol that restricted opioid dispensing successfully cut the discharge allotment of opioid tablets by 89%.
Study details: A single-center review of 337 patients, and a second single-center experience with 122 patients.
Disclosures: Dr. Mark, Dr. Weston, and Dr. Dowdy had no disclosures.
Source: Mark J et al. SGO 2018, Abstract 7. Weston E et al. SGO 2018, Abstract 8.
VIDEO: Triple-antihypertensive pill nails early therapy
ORLANDO – Hypertensive adults started on a triple-drug, single daily pill regimen as either initial or early treatment had a sharply better rate of reaching their goal blood pressure after 6 months, compared with usual-care controls, in a multicenter, randomized trial with 700 patients.
“Early use of a low-dose, three-in-one blood pressure lowering pill is safe and provides faster and better control of blood pressure compared with usual care,” Ruth Webster, PhD, said at the annual meeting of the American College of Cardiology.
The tested polypill contained half the standard doses of the angiotensin receptor blocker telmisartan (20 mg), the calcium channel blocker amlodipine (2.5 mg), and the diuretic chlorthalidone (12.5 mg). After 6 months on this regimen, 70% of patients were at their goal blood pressure, compared with 55% of the control patients, and patients on the polypill had on average a 10/5 mm Hg greater reduction in their blood pressure than did patients on usual care, reported Dr. Webster, head of research programs at the George Institute for Global Health in Sydney. Rates of total and serious adverse events and withdrawals because of adverse events were similar in the two study arms, and both arms also had nearly identical levels of treatment adherence, about 95%.
“No prior trial has evaluated a triple, low-dose pill for initial or early treatment,” she noted.
“This is a home run,” said Karol E. Watson, MD, professor of medicine and director of the Women’s Cardiovascular Health Center at the University of California, Los Angeles. “In the past, clinicians were told to pick one drug and push it as hard as you could and then maybe think about adding a second drug. Experience has shown that this does not increase efficacy, but it does increase adverse events, so current guidelines say start with two drugs. Now they are showing for the first time that you should start with three drugs. That goes with what we know.”
“Triple-drug therapy for the masses makes complete sense,” especially now that the blood pressure goal for most patients is less than 130/80 mm Hg, said William B. White, MD, professor of medicine and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington. Plus, “compliance is vastly improved when you use a combination-drug pill,” he noted.
The blood pressure targets that Dr. Webster and her associates used were less than 140/90 mm Hg except in patients with diabetes or chronic kidney disease, who had a target of less than 130/80 mm Hg. At the time researchers designed the trial the generally accepted blood pressure target for antihypertensive treatment was less than 140/90 mm Hg, Dr. Webster noted.
She also stressed that she did not believe the three specific drugs selected for the polypill made a difference. “The specific drugs we used was not that important. We would probably get the same result with different drugs. It’s about the strategy of using triple, low-dose therapy,” Dr. Webster suggested. Dr. Watson agreed.
The TRIUMPH (Triple Pill vs. Usual Care Management for Patients with Mild to Moderate Hypertension) study enrolled patients at 11 hospital outpatient clinics in Sri Lanka. The average age of the patients was 56 years. The average blood pressure was 154/90 mm Hg. About 59% of patients were not on any antihypertensive drug at baseline, with the rest on a single drug. The study protocol excluded patients on two or more drugs at entry. Roughly 30% of enrolled patients had diabetes, and 1%-2% had chronic kidney disease. Their target blood pressure on treatment during the study was less than 130/80 mm Hg.
The study’s primary endpoint was the percentage of patients at their goal blood pressure after 6 months. Patients in the triple-drug polypill group achieved their goal blood pressure 23% more often relative to the control, usual care patients, a statistically significant difference. The between group difference in achievement of goal blood pressure was apparent by the end of the first 6 weeks in the study. Patients in the control arm generally received either one or two drugs during the study, but often at full dose rather than the half doses used in the triple-drug patients. The study’s design specified that patients in the triple-drug arm who were not at their target blood pressure after 6 weeks could, at the discretion of their treating physician, switch to a second formulation that doubled the dosage of each of the three drugs. Patients in the usual care arm could have their treatment adjusted after 6 or 12 weeks as long as they continued to receive either one or two drugs. After 6 weeks, 68% of patients in the triple-drug arm and 44% receiving usual care were at their blood pressure goal. After 12 weeks, the percentages at goal were 73% of patients on the triple-drug pill and 47% on usual care.
Dr. Webster hypothesized that the triple-drug, low-dose strategy for initial or early treatment would surpass usual care not only in low- and middle-income countries, like Sri Lanka, but also in high-income, industrialized countries such as the United States.
TRIUMPH received no commercial funding. Dr. Webster had no disclosures. Dr. Watson has been a consultant to Amgen, AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and GlaxoSmithKline. Dr. White has been a consultant to Novartis.
SOURCE: Webster R. ACC 2018. Webster R et al. ACC 18 late breaker.
The TRIUMPH results showed the feasibility and efficacy of achieving good blood pressure control with a single pill containing low doses of three different antihypertensive drugs that are well tolerated and have different mechanisms of action. This strategy avoids the adverse effects from drugs used at their maximum dose.
An attraction of this strategy is how seamless it is for patients. They take a single pill with three drugs, which can enhance compliance and in routine practice can reduce their copay. It’s much easier for patients to take a single pill.
Eileen M. Handberg, PhD , is a research professor of medicine and director of the Clinical Trials Program at the University of Florida in Gainesville. She had no relevant disclosures. She made these comments in an interview.
The TRIUMPH results showed the feasibility and efficacy of achieving good blood pressure control with a single pill containing low doses of three different antihypertensive drugs that are well tolerated and have different mechanisms of action. This strategy avoids the adverse effects from drugs used at their maximum dose.
An attraction of this strategy is how seamless it is for patients. They take a single pill with three drugs, which can enhance compliance and in routine practice can reduce their copay. It’s much easier for patients to take a single pill.
Eileen M. Handberg, PhD , is a research professor of medicine and director of the Clinical Trials Program at the University of Florida in Gainesville. She had no relevant disclosures. She made these comments in an interview.
The TRIUMPH results showed the feasibility and efficacy of achieving good blood pressure control with a single pill containing low doses of three different antihypertensive drugs that are well tolerated and have different mechanisms of action. This strategy avoids the adverse effects from drugs used at their maximum dose.
An attraction of this strategy is how seamless it is for patients. They take a single pill with three drugs, which can enhance compliance and in routine practice can reduce their copay. It’s much easier for patients to take a single pill.
Eileen M. Handberg, PhD , is a research professor of medicine and director of the Clinical Trials Program at the University of Florida in Gainesville. She had no relevant disclosures. She made these comments in an interview.
ORLANDO – Hypertensive adults started on a triple-drug, single daily pill regimen as either initial or early treatment had a sharply better rate of reaching their goal blood pressure after 6 months, compared with usual-care controls, in a multicenter, randomized trial with 700 patients.
“Early use of a low-dose, three-in-one blood pressure lowering pill is safe and provides faster and better control of blood pressure compared with usual care,” Ruth Webster, PhD, said at the annual meeting of the American College of Cardiology.
The tested polypill contained half the standard doses of the angiotensin receptor blocker telmisartan (20 mg), the calcium channel blocker amlodipine (2.5 mg), and the diuretic chlorthalidone (12.5 mg). After 6 months on this regimen, 70% of patients were at their goal blood pressure, compared with 55% of the control patients, and patients on the polypill had on average a 10/5 mm Hg greater reduction in their blood pressure than did patients on usual care, reported Dr. Webster, head of research programs at the George Institute for Global Health in Sydney. Rates of total and serious adverse events and withdrawals because of adverse events were similar in the two study arms, and both arms also had nearly identical levels of treatment adherence, about 95%.
“No prior trial has evaluated a triple, low-dose pill for initial or early treatment,” she noted.
“This is a home run,” said Karol E. Watson, MD, professor of medicine and director of the Women’s Cardiovascular Health Center at the University of California, Los Angeles. “In the past, clinicians were told to pick one drug and push it as hard as you could and then maybe think about adding a second drug. Experience has shown that this does not increase efficacy, but it does increase adverse events, so current guidelines say start with two drugs. Now they are showing for the first time that you should start with three drugs. That goes with what we know.”
“Triple-drug therapy for the masses makes complete sense,” especially now that the blood pressure goal for most patients is less than 130/80 mm Hg, said William B. White, MD, professor of medicine and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington. Plus, “compliance is vastly improved when you use a combination-drug pill,” he noted.
The blood pressure targets that Dr. Webster and her associates used were less than 140/90 mm Hg except in patients with diabetes or chronic kidney disease, who had a target of less than 130/80 mm Hg. At the time researchers designed the trial the generally accepted blood pressure target for antihypertensive treatment was less than 140/90 mm Hg, Dr. Webster noted.
She also stressed that she did not believe the three specific drugs selected for the polypill made a difference. “The specific drugs we used was not that important. We would probably get the same result with different drugs. It’s about the strategy of using triple, low-dose therapy,” Dr. Webster suggested. Dr. Watson agreed.
The TRIUMPH (Triple Pill vs. Usual Care Management for Patients with Mild to Moderate Hypertension) study enrolled patients at 11 hospital outpatient clinics in Sri Lanka. The average age of the patients was 56 years. The average blood pressure was 154/90 mm Hg. About 59% of patients were not on any antihypertensive drug at baseline, with the rest on a single drug. The study protocol excluded patients on two or more drugs at entry. Roughly 30% of enrolled patients had diabetes, and 1%-2% had chronic kidney disease. Their target blood pressure on treatment during the study was less than 130/80 mm Hg.
The study’s primary endpoint was the percentage of patients at their goal blood pressure after 6 months. Patients in the triple-drug polypill group achieved their goal blood pressure 23% more often relative to the control, usual care patients, a statistically significant difference. The between group difference in achievement of goal blood pressure was apparent by the end of the first 6 weeks in the study. Patients in the control arm generally received either one or two drugs during the study, but often at full dose rather than the half doses used in the triple-drug patients. The study’s design specified that patients in the triple-drug arm who were not at their target blood pressure after 6 weeks could, at the discretion of their treating physician, switch to a second formulation that doubled the dosage of each of the three drugs. Patients in the usual care arm could have their treatment adjusted after 6 or 12 weeks as long as they continued to receive either one or two drugs. After 6 weeks, 68% of patients in the triple-drug arm and 44% receiving usual care were at their blood pressure goal. After 12 weeks, the percentages at goal were 73% of patients on the triple-drug pill and 47% on usual care.
Dr. Webster hypothesized that the triple-drug, low-dose strategy for initial or early treatment would surpass usual care not only in low- and middle-income countries, like Sri Lanka, but also in high-income, industrialized countries such as the United States.
TRIUMPH received no commercial funding. Dr. Webster had no disclosures. Dr. Watson has been a consultant to Amgen, AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and GlaxoSmithKline. Dr. White has been a consultant to Novartis.
SOURCE: Webster R. ACC 2018. Webster R et al. ACC 18 late breaker.
ORLANDO – Hypertensive adults started on a triple-drug, single daily pill regimen as either initial or early treatment had a sharply better rate of reaching their goal blood pressure after 6 months, compared with usual-care controls, in a multicenter, randomized trial with 700 patients.
“Early use of a low-dose, three-in-one blood pressure lowering pill is safe and provides faster and better control of blood pressure compared with usual care,” Ruth Webster, PhD, said at the annual meeting of the American College of Cardiology.
The tested polypill contained half the standard doses of the angiotensin receptor blocker telmisartan (20 mg), the calcium channel blocker amlodipine (2.5 mg), and the diuretic chlorthalidone (12.5 mg). After 6 months on this regimen, 70% of patients were at their goal blood pressure, compared with 55% of the control patients, and patients on the polypill had on average a 10/5 mm Hg greater reduction in their blood pressure than did patients on usual care, reported Dr. Webster, head of research programs at the George Institute for Global Health in Sydney. Rates of total and serious adverse events and withdrawals because of adverse events were similar in the two study arms, and both arms also had nearly identical levels of treatment adherence, about 95%.
“No prior trial has evaluated a triple, low-dose pill for initial or early treatment,” she noted.
“This is a home run,” said Karol E. Watson, MD, professor of medicine and director of the Women’s Cardiovascular Health Center at the University of California, Los Angeles. “In the past, clinicians were told to pick one drug and push it as hard as you could and then maybe think about adding a second drug. Experience has shown that this does not increase efficacy, but it does increase adverse events, so current guidelines say start with two drugs. Now they are showing for the first time that you should start with three drugs. That goes with what we know.”
“Triple-drug therapy for the masses makes complete sense,” especially now that the blood pressure goal for most patients is less than 130/80 mm Hg, said William B. White, MD, professor of medicine and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington. Plus, “compliance is vastly improved when you use a combination-drug pill,” he noted.
The blood pressure targets that Dr. Webster and her associates used were less than 140/90 mm Hg except in patients with diabetes or chronic kidney disease, who had a target of less than 130/80 mm Hg. At the time researchers designed the trial the generally accepted blood pressure target for antihypertensive treatment was less than 140/90 mm Hg, Dr. Webster noted.
She also stressed that she did not believe the three specific drugs selected for the polypill made a difference. “The specific drugs we used was not that important. We would probably get the same result with different drugs. It’s about the strategy of using triple, low-dose therapy,” Dr. Webster suggested. Dr. Watson agreed.
The TRIUMPH (Triple Pill vs. Usual Care Management for Patients with Mild to Moderate Hypertension) study enrolled patients at 11 hospital outpatient clinics in Sri Lanka. The average age of the patients was 56 years. The average blood pressure was 154/90 mm Hg. About 59% of patients were not on any antihypertensive drug at baseline, with the rest on a single drug. The study protocol excluded patients on two or more drugs at entry. Roughly 30% of enrolled patients had diabetes, and 1%-2% had chronic kidney disease. Their target blood pressure on treatment during the study was less than 130/80 mm Hg.
The study’s primary endpoint was the percentage of patients at their goal blood pressure after 6 months. Patients in the triple-drug polypill group achieved their goal blood pressure 23% more often relative to the control, usual care patients, a statistically significant difference. The between group difference in achievement of goal blood pressure was apparent by the end of the first 6 weeks in the study. Patients in the control arm generally received either one or two drugs during the study, but often at full dose rather than the half doses used in the triple-drug patients. The study’s design specified that patients in the triple-drug arm who were not at their target blood pressure after 6 weeks could, at the discretion of their treating physician, switch to a second formulation that doubled the dosage of each of the three drugs. Patients in the usual care arm could have their treatment adjusted after 6 or 12 weeks as long as they continued to receive either one or two drugs. After 6 weeks, 68% of patients in the triple-drug arm and 44% receiving usual care were at their blood pressure goal. After 12 weeks, the percentages at goal were 73% of patients on the triple-drug pill and 47% on usual care.
Dr. Webster hypothesized that the triple-drug, low-dose strategy for initial or early treatment would surpass usual care not only in low- and middle-income countries, like Sri Lanka, but also in high-income, industrialized countries such as the United States.
TRIUMPH received no commercial funding. Dr. Webster had no disclosures. Dr. Watson has been a consultant to Amgen, AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and GlaxoSmithKline. Dr. White has been a consultant to Novartis.
SOURCE: Webster R. ACC 2018. Webster R et al. ACC 18 late breaker.
REPORTING FROM ACC 18
Key clinical point: Starting hypertensive patients on a single, triple-drug pill produced excellent control.
Major finding: After 6 months, 70% of patients on the triple-drug pill reached target blood pressure, compared with 55% of control patients.
Study details: TRIUMPH, a multicenter, randomized trial with 700 hypertensive adults.
Disclosures: TRIUMPH received no commercial funding. Dr. Webster had no disclosures. Dr. Watson has been a consultant to Amgen, AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and GlaxoSmithKline. Dr. White has been a consultant to Novartis.
Source: Webster R et al. ACC 18 late breaker.
VIDEO: Cervical cancer laparotomy outperforms minimally invasive surgery
NEW ORLEANS – Use of minimally invasive radical hysterectomy to treat early-stage cervical cancer has grown over the past decade, and in current U.S. practice, roughly half of these cases are done with a minimally-invasive approach, with the rest done by conventional laparotomy. But the first data ever reported from a large, prospective trial that compared the efficacy of both methods for cervical cancer had the unexpected finding that disease-free survival following minimally invasive procedures significantly lagged behind radical hysterectomies done by open laparotomy, Pedro T. Ramirez, MD, said at the annual meeting of the Society of Gynecologic Oncology.
Just after this report came results from a second study that used propensity score–adjusted observational data from the National Cancer Database and found significantly worse overall survival following minimally invasive radical hysterectomy for early-stage cervical cancer, compared with laparotomy, said J. Alejandro Rauh-Hain, MD, a gynecologic oncologist at the University of Texas MD Anderson Cancer Center in Houston.
Both findings were “very surprising,” said Dr. Rauh-Hain in a video interview. “I was pretty sure we’d see no difference” in outcomes between minimally invasive radical hysterectomies and the same surgery either done by laparoscope or robotically assisted.
Prior prospective comparisons of minimally invasive and open surgical methods for other cancer types, including endometrial, gastric, and ovarian, showed no differences in cancer recurrences and survival, which led to widening use of minimally invasive surgery (MIS) for cervical cancer despite no direct evidence supporting equivalence, Dr. Rauh-Hain noted. “We adopted it with no data. It made sense that cervical cancer would be the same as endometrial cancer,” he explained.
The Laparoscopic Approach to Cervical Cancer (LACC) trial ran at 33 centers in 12 countries, including six U.S. centers. The study randomized women during 2008-2017 who had stage 1A1, 1A2, or 1B1 cervical cancer to either MIS or open surgery for a radical hysterectomy. Each participating center had to submit to a trial review committee full case records for 10 patients and unedited surgical videos of two patients who had previously undergone a minimally invasive radical hysterectomy at the center to document local prowess with MIS.
Dr. Ramirez and his colleagues designed LACC to prove the noninferiority of MIS and calculated an expected enrollment of 740 patients based on statistical expectations, but the study stopped early after enrolling 631 patients because of the adverse outcomes identified in the MIS patients, with a median follow-up of 2.5 years instead of the planned follow-up of 4.5 years. The study reached the 4.5-year follow-up in about 39% of patients. Of the 312 patients randomized to undergo laparotomy, 88% actually underwent the surgery; of the 319 patients randomized to MIS, 91% received this surgery, with 16% of the MIS procedures done using robotic assistance.
The study’s primary endpoint was disease-free survival at 4.5 years, which occurred in 86% of the MIS patients and in 96.5% of the laparotomy patients, a difference that failed to meet the study’s prespecified definition of noninferiority for MIS, reported Dr. Ramirez, a professor of gynecologic oncology and director of Minimally Invasive Surgery Research and Education at the MD Anderson Cancer Center. In addition, several secondary analyses of the data all showed starkly superior outcomes in the laparotomy subgroup.
Disease-free survival among all patients regardless of follow-up duration occurred in 98% of laparotomy patients and 92% of MIS patients, which translated into a 3.74 hazard ratio (P = .002) for disease recurrence or death among the MIS patients when compared with laparotomy patients. The all-cause mortality rates were 1% in the laparotomy patients and 6% among the MIS patients, a hazard ratio of 6.00 (P = .004). The risk of local or regional recurrences was more than fourfold higher in the MIS patients. A blinded, central panel adjudicated all recurrences identified during the study.
The LACC results “should be discussed with patients scheduled to undergo radical hysterectomy” for cervical cancer, Dr. Ramirez concluded.
The observational data from the National Cancer Database used in the analysis led by Dr. Rauh-Hain came from 2,221 patients hospitalized and treated with radical hysterectomy and pelvic lymph node dissection at a U.S. center during 2010-2012 for either stage 1A2 or 1B1 cervical cancer. Among these patients, 47.5% underwent MIS, with 79% of those procedures done with robotic assistance, while the other 52.5% underwent open laparotomy, Dr. Rauh-Hain reported. Additional analysis of data from this database by the researchers showed that, although the first report of MIS for radical hysterectomy appeared in 1992, the approach remained largely unused in U.S. practice until 2007, when use of MIS began to sharply rise. By 2010, about a third of radical hysterectomies for cervical cancer involved MIS, and usage increased still further during 2011 and 2012 to produce a nearly 48% rate during the 3-year study period.
The primary endpoint of Dr. Rauh-Hain’s analysis was overall survival following propensity-score matching of the MIS and laparotomy patients using 13 demographic and clinical criteria. The analysis showed 4-year mortality rates of 5.8% among the laparotomy patients and 8.4% among the MIS patients, which calculated to a relatively increased mortality hazard from MIS of 48% (P = .02).
Dr. Rauh-Hain also reported results from an interrupted time series analysis using data from the Surveillance, Epidemiology, and End Results database of the National Cancer Institute. This analysis compared annual 4-year relative survival rates among women undergoing radical hysterectomy for cervical cancer and found that, after survival rates showed a gradual, steady rise during the years culminating in 2006, once MIS began being more widely used in 2007 survival rates began to drop, with a statistically significant annualized decline of 1% through 2010.
Based on the results from both studies, “at MD Anderson we discuss the results with patients,” with the consequence that the percentage of patients treated with laparotomy is now increasing, Dr. Rauh-Hain said. The results from both studies “are concerning,” he explained.
SOURCE: Ramirez PT and Rauh-Hain JA. SGO 2018, Late-Breaking Abstracts 1 and 2.
The findings from these studies appear valid and should be discussed with patients.
The findings raise a major question: Why has minimally invasive surgery (MIS) led to worse survival rates than laparotomy? Several possible explanations can be hypothesized: The uterine manipulator used in MIS led to local spread of cancer cells; MIS involves a learning curve and initial attempts at MIS did not remove enough of the tumor; and MIS led to increased exposure of the peritoneal cavity to the cancer. The findings also raise another question: Why has MIS for cervical cancer performed less well than MIS for cancers from other organs, such as endometrial and prostate?
The overall survival results from the LACC trial calculate out to 4.75 added deaths per year for every 1,000 patients treated with MIS, compared with laparoscopy. But the National Inpatient Sample data suggest that MIS cuts mortality by about two deaths per year per 1,000 patients, compared with laparotomy, and mortality data from a different analysis (Gynecol Oncol. 2012 Oct;127[1]:11-7) suggest that MIS might prevent six deaths annually for every 1,000 patients, compared with laparotomy. Overall, these three sets of findings suggest roughly comparable mortality outcomes from MIS and laparotomy, but with MIS having the bonus of fewer complications and less need for transfusions.
The cautions and concerns raised by the LACC trial and Dr. Rauh-Hain’s analysis of observational data cannot be easily dismissed. We need to figure out why the results from both studies show worse survival and recurrence rates with MIS, and we need to identify whether subgroups of patients exist who might clearly benefit from either the MIS or open-surgery approach.
Shitanshu Uppal, MD , is a gynecologic oncologist at the University of Michigan in Ann Arbor. He made these comments as designated discussant for the two studies. He had no disclosures.
The findings from these studies appear valid and should be discussed with patients.
The findings raise a major question: Why has minimally invasive surgery (MIS) led to worse survival rates than laparotomy? Several possible explanations can be hypothesized: The uterine manipulator used in MIS led to local spread of cancer cells; MIS involves a learning curve and initial attempts at MIS did not remove enough of the tumor; and MIS led to increased exposure of the peritoneal cavity to the cancer. The findings also raise another question: Why has MIS for cervical cancer performed less well than MIS for cancers from other organs, such as endometrial and prostate?
The overall survival results from the LACC trial calculate out to 4.75 added deaths per year for every 1,000 patients treated with MIS, compared with laparoscopy. But the National Inpatient Sample data suggest that MIS cuts mortality by about two deaths per year per 1,000 patients, compared with laparotomy, and mortality data from a different analysis (Gynecol Oncol. 2012 Oct;127[1]:11-7) suggest that MIS might prevent six deaths annually for every 1,000 patients, compared with laparotomy. Overall, these three sets of findings suggest roughly comparable mortality outcomes from MIS and laparotomy, but with MIS having the bonus of fewer complications and less need for transfusions.
The cautions and concerns raised by the LACC trial and Dr. Rauh-Hain’s analysis of observational data cannot be easily dismissed. We need to figure out why the results from both studies show worse survival and recurrence rates with MIS, and we need to identify whether subgroups of patients exist who might clearly benefit from either the MIS or open-surgery approach.
Shitanshu Uppal, MD , is a gynecologic oncologist at the University of Michigan in Ann Arbor. He made these comments as designated discussant for the two studies. He had no disclosures.
The findings from these studies appear valid and should be discussed with patients.
The findings raise a major question: Why has minimally invasive surgery (MIS) led to worse survival rates than laparotomy? Several possible explanations can be hypothesized: The uterine manipulator used in MIS led to local spread of cancer cells; MIS involves a learning curve and initial attempts at MIS did not remove enough of the tumor; and MIS led to increased exposure of the peritoneal cavity to the cancer. The findings also raise another question: Why has MIS for cervical cancer performed less well than MIS for cancers from other organs, such as endometrial and prostate?
The overall survival results from the LACC trial calculate out to 4.75 added deaths per year for every 1,000 patients treated with MIS, compared with laparoscopy. But the National Inpatient Sample data suggest that MIS cuts mortality by about two deaths per year per 1,000 patients, compared with laparotomy, and mortality data from a different analysis (Gynecol Oncol. 2012 Oct;127[1]:11-7) suggest that MIS might prevent six deaths annually for every 1,000 patients, compared with laparotomy. Overall, these three sets of findings suggest roughly comparable mortality outcomes from MIS and laparotomy, but with MIS having the bonus of fewer complications and less need for transfusions.
The cautions and concerns raised by the LACC trial and Dr. Rauh-Hain’s analysis of observational data cannot be easily dismissed. We need to figure out why the results from both studies show worse survival and recurrence rates with MIS, and we need to identify whether subgroups of patients exist who might clearly benefit from either the MIS or open-surgery approach.
Shitanshu Uppal, MD , is a gynecologic oncologist at the University of Michigan in Ann Arbor. He made these comments as designated discussant for the two studies. He had no disclosures.
NEW ORLEANS – Use of minimally invasive radical hysterectomy to treat early-stage cervical cancer has grown over the past decade, and in current U.S. practice, roughly half of these cases are done with a minimally-invasive approach, with the rest done by conventional laparotomy. But the first data ever reported from a large, prospective trial that compared the efficacy of both methods for cervical cancer had the unexpected finding that disease-free survival following minimally invasive procedures significantly lagged behind radical hysterectomies done by open laparotomy, Pedro T. Ramirez, MD, said at the annual meeting of the Society of Gynecologic Oncology.
Just after this report came results from a second study that used propensity score–adjusted observational data from the National Cancer Database and found significantly worse overall survival following minimally invasive radical hysterectomy for early-stage cervical cancer, compared with laparotomy, said J. Alejandro Rauh-Hain, MD, a gynecologic oncologist at the University of Texas MD Anderson Cancer Center in Houston.
Both findings were “very surprising,” said Dr. Rauh-Hain in a video interview. “I was pretty sure we’d see no difference” in outcomes between minimally invasive radical hysterectomies and the same surgery either done by laparoscope or robotically assisted.
Prior prospective comparisons of minimally invasive and open surgical methods for other cancer types, including endometrial, gastric, and ovarian, showed no differences in cancer recurrences and survival, which led to widening use of minimally invasive surgery (MIS) for cervical cancer despite no direct evidence supporting equivalence, Dr. Rauh-Hain noted. “We adopted it with no data. It made sense that cervical cancer would be the same as endometrial cancer,” he explained.
The Laparoscopic Approach to Cervical Cancer (LACC) trial ran at 33 centers in 12 countries, including six U.S. centers. The study randomized women during 2008-2017 who had stage 1A1, 1A2, or 1B1 cervical cancer to either MIS or open surgery for a radical hysterectomy. Each participating center had to submit to a trial review committee full case records for 10 patients and unedited surgical videos of two patients who had previously undergone a minimally invasive radical hysterectomy at the center to document local prowess with MIS.
Dr. Ramirez and his colleagues designed LACC to prove the noninferiority of MIS and calculated an expected enrollment of 740 patients based on statistical expectations, but the study stopped early after enrolling 631 patients because of the adverse outcomes identified in the MIS patients, with a median follow-up of 2.5 years instead of the planned follow-up of 4.5 years. The study reached the 4.5-year follow-up in about 39% of patients. Of the 312 patients randomized to undergo laparotomy, 88% actually underwent the surgery; of the 319 patients randomized to MIS, 91% received this surgery, with 16% of the MIS procedures done using robotic assistance.
The study’s primary endpoint was disease-free survival at 4.5 years, which occurred in 86% of the MIS patients and in 96.5% of the laparotomy patients, a difference that failed to meet the study’s prespecified definition of noninferiority for MIS, reported Dr. Ramirez, a professor of gynecologic oncology and director of Minimally Invasive Surgery Research and Education at the MD Anderson Cancer Center. In addition, several secondary analyses of the data all showed starkly superior outcomes in the laparotomy subgroup.
Disease-free survival among all patients regardless of follow-up duration occurred in 98% of laparotomy patients and 92% of MIS patients, which translated into a 3.74 hazard ratio (P = .002) for disease recurrence or death among the MIS patients when compared with laparotomy patients. The all-cause mortality rates were 1% in the laparotomy patients and 6% among the MIS patients, a hazard ratio of 6.00 (P = .004). The risk of local or regional recurrences was more than fourfold higher in the MIS patients. A blinded, central panel adjudicated all recurrences identified during the study.
The LACC results “should be discussed with patients scheduled to undergo radical hysterectomy” for cervical cancer, Dr. Ramirez concluded.
The observational data from the National Cancer Database used in the analysis led by Dr. Rauh-Hain came from 2,221 patients hospitalized and treated with radical hysterectomy and pelvic lymph node dissection at a U.S. center during 2010-2012 for either stage 1A2 or 1B1 cervical cancer. Among these patients, 47.5% underwent MIS, with 79% of those procedures done with robotic assistance, while the other 52.5% underwent open laparotomy, Dr. Rauh-Hain reported. Additional analysis of data from this database by the researchers showed that, although the first report of MIS for radical hysterectomy appeared in 1992, the approach remained largely unused in U.S. practice until 2007, when use of MIS began to sharply rise. By 2010, about a third of radical hysterectomies for cervical cancer involved MIS, and usage increased still further during 2011 and 2012 to produce a nearly 48% rate during the 3-year study period.
The primary endpoint of Dr. Rauh-Hain’s analysis was overall survival following propensity-score matching of the MIS and laparotomy patients using 13 demographic and clinical criteria. The analysis showed 4-year mortality rates of 5.8% among the laparotomy patients and 8.4% among the MIS patients, which calculated to a relatively increased mortality hazard from MIS of 48% (P = .02).
Dr. Rauh-Hain also reported results from an interrupted time series analysis using data from the Surveillance, Epidemiology, and End Results database of the National Cancer Institute. This analysis compared annual 4-year relative survival rates among women undergoing radical hysterectomy for cervical cancer and found that, after survival rates showed a gradual, steady rise during the years culminating in 2006, once MIS began being more widely used in 2007 survival rates began to drop, with a statistically significant annualized decline of 1% through 2010.
Based on the results from both studies, “at MD Anderson we discuss the results with patients,” with the consequence that the percentage of patients treated with laparotomy is now increasing, Dr. Rauh-Hain said. The results from both studies “are concerning,” he explained.
SOURCE: Ramirez PT and Rauh-Hain JA. SGO 2018, Late-Breaking Abstracts 1 and 2.
NEW ORLEANS – Use of minimally invasive radical hysterectomy to treat early-stage cervical cancer has grown over the past decade, and in current U.S. practice, roughly half of these cases are done with a minimally-invasive approach, with the rest done by conventional laparotomy. But the first data ever reported from a large, prospective trial that compared the efficacy of both methods for cervical cancer had the unexpected finding that disease-free survival following minimally invasive procedures significantly lagged behind radical hysterectomies done by open laparotomy, Pedro T. Ramirez, MD, said at the annual meeting of the Society of Gynecologic Oncology.
Just after this report came results from a second study that used propensity score–adjusted observational data from the National Cancer Database and found significantly worse overall survival following minimally invasive radical hysterectomy for early-stage cervical cancer, compared with laparotomy, said J. Alejandro Rauh-Hain, MD, a gynecologic oncologist at the University of Texas MD Anderson Cancer Center in Houston.
Both findings were “very surprising,” said Dr. Rauh-Hain in a video interview. “I was pretty sure we’d see no difference” in outcomes between minimally invasive radical hysterectomies and the same surgery either done by laparoscope or robotically assisted.
Prior prospective comparisons of minimally invasive and open surgical methods for other cancer types, including endometrial, gastric, and ovarian, showed no differences in cancer recurrences and survival, which led to widening use of minimally invasive surgery (MIS) for cervical cancer despite no direct evidence supporting equivalence, Dr. Rauh-Hain noted. “We adopted it with no data. It made sense that cervical cancer would be the same as endometrial cancer,” he explained.
The Laparoscopic Approach to Cervical Cancer (LACC) trial ran at 33 centers in 12 countries, including six U.S. centers. The study randomized women during 2008-2017 who had stage 1A1, 1A2, or 1B1 cervical cancer to either MIS or open surgery for a radical hysterectomy. Each participating center had to submit to a trial review committee full case records for 10 patients and unedited surgical videos of two patients who had previously undergone a minimally invasive radical hysterectomy at the center to document local prowess with MIS.
Dr. Ramirez and his colleagues designed LACC to prove the noninferiority of MIS and calculated an expected enrollment of 740 patients based on statistical expectations, but the study stopped early after enrolling 631 patients because of the adverse outcomes identified in the MIS patients, with a median follow-up of 2.5 years instead of the planned follow-up of 4.5 years. The study reached the 4.5-year follow-up in about 39% of patients. Of the 312 patients randomized to undergo laparotomy, 88% actually underwent the surgery; of the 319 patients randomized to MIS, 91% received this surgery, with 16% of the MIS procedures done using robotic assistance.
The study’s primary endpoint was disease-free survival at 4.5 years, which occurred in 86% of the MIS patients and in 96.5% of the laparotomy patients, a difference that failed to meet the study’s prespecified definition of noninferiority for MIS, reported Dr. Ramirez, a professor of gynecologic oncology and director of Minimally Invasive Surgery Research and Education at the MD Anderson Cancer Center. In addition, several secondary analyses of the data all showed starkly superior outcomes in the laparotomy subgroup.
Disease-free survival among all patients regardless of follow-up duration occurred in 98% of laparotomy patients and 92% of MIS patients, which translated into a 3.74 hazard ratio (P = .002) for disease recurrence or death among the MIS patients when compared with laparotomy patients. The all-cause mortality rates were 1% in the laparotomy patients and 6% among the MIS patients, a hazard ratio of 6.00 (P = .004). The risk of local or regional recurrences was more than fourfold higher in the MIS patients. A blinded, central panel adjudicated all recurrences identified during the study.
The LACC results “should be discussed with patients scheduled to undergo radical hysterectomy” for cervical cancer, Dr. Ramirez concluded.
The observational data from the National Cancer Database used in the analysis led by Dr. Rauh-Hain came from 2,221 patients hospitalized and treated with radical hysterectomy and pelvic lymph node dissection at a U.S. center during 2010-2012 for either stage 1A2 or 1B1 cervical cancer. Among these patients, 47.5% underwent MIS, with 79% of those procedures done with robotic assistance, while the other 52.5% underwent open laparotomy, Dr. Rauh-Hain reported. Additional analysis of data from this database by the researchers showed that, although the first report of MIS for radical hysterectomy appeared in 1992, the approach remained largely unused in U.S. practice until 2007, when use of MIS began to sharply rise. By 2010, about a third of radical hysterectomies for cervical cancer involved MIS, and usage increased still further during 2011 and 2012 to produce a nearly 48% rate during the 3-year study period.
The primary endpoint of Dr. Rauh-Hain’s analysis was overall survival following propensity-score matching of the MIS and laparotomy patients using 13 demographic and clinical criteria. The analysis showed 4-year mortality rates of 5.8% among the laparotomy patients and 8.4% among the MIS patients, which calculated to a relatively increased mortality hazard from MIS of 48% (P = .02).
Dr. Rauh-Hain also reported results from an interrupted time series analysis using data from the Surveillance, Epidemiology, and End Results database of the National Cancer Institute. This analysis compared annual 4-year relative survival rates among women undergoing radical hysterectomy for cervical cancer and found that, after survival rates showed a gradual, steady rise during the years culminating in 2006, once MIS began being more widely used in 2007 survival rates began to drop, with a statistically significant annualized decline of 1% through 2010.
Based on the results from both studies, “at MD Anderson we discuss the results with patients,” with the consequence that the percentage of patients treated with laparotomy is now increasing, Dr. Rauh-Hain said. The results from both studies “are concerning,” he explained.
SOURCE: Ramirez PT and Rauh-Hain JA. SGO 2018, Late-Breaking Abstracts 1 and 2.
REPORTING FROM SGO 2018
Key clinical point: Laparotomy produced better survival than did minimally invasive surgery for cervical cancer.
Major finding: Disease-free survival after 4.5 years was 96.5% with laparotomy and 86.0% with minimally invasive surgery.
Study details: LACC was a multicenter, randomized trial with 631 patients. The observational study included 2,221 patients from the National Cancer Database during 2010-2012.
Disclosures: Dr. Ramirez and Dr. Rauh-Hain had no disclosures.
Source: Ramirez PT and Rauh-Hain JA. SGO 2018, Late-Breaking Abstracts 1 and 2.
AAD guidelines’ conflict-of-interest policies discussed in pro-con debate
SAN DIEGO – The American Academy of Dermatology’s policies that regulate conflicts of interest among members of its guidelines panels are “pretty good, but could be improved,” Lionel G. Bercovitch, MD, said at the annual meeting of the American Academy Dermatology.
One positive step might be to tighten the current American Academy of Dermatology requirement that more than half of the members in clinical guideline work groups be free of any financial conflicts and the minimum be raised to a higher percentage, such as more than 70%, suggested Dr. Bercovitch, a professor of dermatology at Brown University, Providence, R.I.
But his concern over the adequacy of existing conflict barriers during the writing of clinical guidelines wasn’t shared by Clifford Perlis, MD, who countered that “there are reasons not to waste too much time wringing our hands over conflicts of interest.”
He offered four reasons to support his statement:
- Conflicts of interest are ubiquitous and thus impossible to eliminate.
- Excluding working group members with conflicts can deprive the guidelines of valuable expertise.
- Checks and balances that are already in place in guideline development prevent inappropriate influence from conflicts of interest.
- No evidence has shown that conflicts of interest have inappropriately influenced development of treatment guidelines.
Conflicts of interest may not be as well managed as AAD policies suggest, Dr. Bercovitch noted. He cited a report published in late 2017 that tallied the actual conflicts of 49 people who served as the authors of three AAD guidelines published during 2013-2016. To objectively double check each author’s conflicts the researchers used the Open Payments database run by the Centers for Medicare & Medicaid Services (JAMA Dermatol. 2017 Dec;153[12]:1229-35).
The analysis showed that 40 of the 49 authors (82%) had received some amount of industry payment, 63% had received more than $1,000, and 51% had received more than $10,000. The median amount received from industry was just over $33,000. The analysis also showed that 22 of the 40 authors who received an industry payment had disclosure statements for the guideline they participated in that did not agree with the information in the Open Payments database.
“The AAD relies on information obtained through its self-reported online member disclosure system. This internal system collects updates to disclosed relationships on a real-time, ongoing basis, allowing the AAD to regularly assess any changes,” wrote Dr. Lim and his coauthors. “This provides information in a more meaningful and time-sensitive way” than the Open Payments database. In addition, the Open Payments database “is known to be inaccurate,” while the AAD “relies on information obtained through its self-reported online member disclosure system.” This includes an assessment of the relevancy of the conflict to the guideline involved. “This critical evaluation of relevancy was not addressed in the authors’ analysis,” they added.
They reported an adjusted analysis of the percentage of authors with relevant conflicts for each of the guidelines examined in the initial study. The percentages shrank to zero, 40%, and 43% of the authors with relevant conflicts, percentages that fell within the AAD’s ceiling for an acceptable percentage of work group members with conflicts.
The discussion on this topic was presented during a forum on dermatoethics at the meeting, structured as a debate in which presenters are assigned an ethical argument or point-of-view to discuss and defend. The position taken by the speaker need not (and often does not) correspond to the speaker’s personal views.
SAN DIEGO – The American Academy of Dermatology’s policies that regulate conflicts of interest among members of its guidelines panels are “pretty good, but could be improved,” Lionel G. Bercovitch, MD, said at the annual meeting of the American Academy Dermatology.
One positive step might be to tighten the current American Academy of Dermatology requirement that more than half of the members in clinical guideline work groups be free of any financial conflicts and the minimum be raised to a higher percentage, such as more than 70%, suggested Dr. Bercovitch, a professor of dermatology at Brown University, Providence, R.I.
But his concern over the adequacy of existing conflict barriers during the writing of clinical guidelines wasn’t shared by Clifford Perlis, MD, who countered that “there are reasons not to waste too much time wringing our hands over conflicts of interest.”
He offered four reasons to support his statement:
- Conflicts of interest are ubiquitous and thus impossible to eliminate.
- Excluding working group members with conflicts can deprive the guidelines of valuable expertise.
- Checks and balances that are already in place in guideline development prevent inappropriate influence from conflicts of interest.
- No evidence has shown that conflicts of interest have inappropriately influenced development of treatment guidelines.
Conflicts of interest may not be as well managed as AAD policies suggest, Dr. Bercovitch noted. He cited a report published in late 2017 that tallied the actual conflicts of 49 people who served as the authors of three AAD guidelines published during 2013-2016. To objectively double check each author’s conflicts the researchers used the Open Payments database run by the Centers for Medicare & Medicaid Services (JAMA Dermatol. 2017 Dec;153[12]:1229-35).
The analysis showed that 40 of the 49 authors (82%) had received some amount of industry payment, 63% had received more than $1,000, and 51% had received more than $10,000. The median amount received from industry was just over $33,000. The analysis also showed that 22 of the 40 authors who received an industry payment had disclosure statements for the guideline they participated in that did not agree with the information in the Open Payments database.
“The AAD relies on information obtained through its self-reported online member disclosure system. This internal system collects updates to disclosed relationships on a real-time, ongoing basis, allowing the AAD to regularly assess any changes,” wrote Dr. Lim and his coauthors. “This provides information in a more meaningful and time-sensitive way” than the Open Payments database. In addition, the Open Payments database “is known to be inaccurate,” while the AAD “relies on information obtained through its self-reported online member disclosure system.” This includes an assessment of the relevancy of the conflict to the guideline involved. “This critical evaluation of relevancy was not addressed in the authors’ analysis,” they added.
They reported an adjusted analysis of the percentage of authors with relevant conflicts for each of the guidelines examined in the initial study. The percentages shrank to zero, 40%, and 43% of the authors with relevant conflicts, percentages that fell within the AAD’s ceiling for an acceptable percentage of work group members with conflicts.
The discussion on this topic was presented during a forum on dermatoethics at the meeting, structured as a debate in which presenters are assigned an ethical argument or point-of-view to discuss and defend. The position taken by the speaker need not (and often does not) correspond to the speaker’s personal views.
SAN DIEGO – The American Academy of Dermatology’s policies that regulate conflicts of interest among members of its guidelines panels are “pretty good, but could be improved,” Lionel G. Bercovitch, MD, said at the annual meeting of the American Academy Dermatology.
One positive step might be to tighten the current American Academy of Dermatology requirement that more than half of the members in clinical guideline work groups be free of any financial conflicts and the minimum be raised to a higher percentage, such as more than 70%, suggested Dr. Bercovitch, a professor of dermatology at Brown University, Providence, R.I.
But his concern over the adequacy of existing conflict barriers during the writing of clinical guidelines wasn’t shared by Clifford Perlis, MD, who countered that “there are reasons not to waste too much time wringing our hands over conflicts of interest.”
He offered four reasons to support his statement:
- Conflicts of interest are ubiquitous and thus impossible to eliminate.
- Excluding working group members with conflicts can deprive the guidelines of valuable expertise.
- Checks and balances that are already in place in guideline development prevent inappropriate influence from conflicts of interest.
- No evidence has shown that conflicts of interest have inappropriately influenced development of treatment guidelines.
Conflicts of interest may not be as well managed as AAD policies suggest, Dr. Bercovitch noted. He cited a report published in late 2017 that tallied the actual conflicts of 49 people who served as the authors of three AAD guidelines published during 2013-2016. To objectively double check each author’s conflicts the researchers used the Open Payments database run by the Centers for Medicare & Medicaid Services (JAMA Dermatol. 2017 Dec;153[12]:1229-35).
The analysis showed that 40 of the 49 authors (82%) had received some amount of industry payment, 63% had received more than $1,000, and 51% had received more than $10,000. The median amount received from industry was just over $33,000. The analysis also showed that 22 of the 40 authors who received an industry payment had disclosure statements for the guideline they participated in that did not agree with the information in the Open Payments database.
“The AAD relies on information obtained through its self-reported online member disclosure system. This internal system collects updates to disclosed relationships on a real-time, ongoing basis, allowing the AAD to regularly assess any changes,” wrote Dr. Lim and his coauthors. “This provides information in a more meaningful and time-sensitive way” than the Open Payments database. In addition, the Open Payments database “is known to be inaccurate,” while the AAD “relies on information obtained through its self-reported online member disclosure system.” This includes an assessment of the relevancy of the conflict to the guideline involved. “This critical evaluation of relevancy was not addressed in the authors’ analysis,” they added.
They reported an adjusted analysis of the percentage of authors with relevant conflicts for each of the guidelines examined in the initial study. The percentages shrank to zero, 40%, and 43% of the authors with relevant conflicts, percentages that fell within the AAD’s ceiling for an acceptable percentage of work group members with conflicts.
The discussion on this topic was presented during a forum on dermatoethics at the meeting, structured as a debate in which presenters are assigned an ethical argument or point-of-view to discuss and defend. The position taken by the speaker need not (and often does not) correspond to the speaker’s personal views.
EXPERT ANALYSIS FROM AAD 18



























