User login
Primary Care Offices Unprepared for Allergen Therapy Emergencies
ORLANDO – A survey of primary care offices revealed that the majority were not fully equipped to handle adverse reactions to allergen immunotherapy.
"Primary care offices require further education on administration of allergen immunotherapy to ensure adherence to the updated practice parameter, as well as to ensure safety," Dr. Vinitha Reddy said at the annual meeting of the American Academy of Allergy, Asthma and Immunology.
The telephone survey involved a questionnaire administered to nurses at 43 primary care offices (35 family medicine, 4 pediatric, and 4 internal medicine) that administer allergen immunotherapy (AIT). The practices employed a total 194 physicians, and administered AIT to approximately 500 patients.
The survey assessed adherence to the recommendations in "Allergen immunotherapy: a practice parameter third update," a joint guideline issued by the American Academy of Allergy, Asthma, and Immunology and the American College of Allergy, Asthma, and Immunology (J. Allergy Clin. Immunol. 2011;127:S1-55).
The guideline states that allergists’ offices are the preferred setting, but that primary care offices can perform AIT if the appropriate personnel and equipment are available to identify and treat anaphylaxis or other emergencies. The risk is low – approximately 0.2% of patients experience systemic reactions per injection, and deaths occur in just 1 per 2.5 million injections. However, the risk is not nonexistent, said Dr. Reddy, a second-year postgraduate fellow at Pennsylvania State University, Hershey.
Only 53% of the offices reported performing the recommended preinjection health assessment, which should include a determination of the presence of asthma and the degree of asthma control. Only 23% reported measuring peak flow, which is suggested as a method for assessing lung function in asthma patients.
Just 33% of the practices reported routinely having patients wait the recommended 30 minutes following AIT before leaving the office. Another 42% had patients wait just 20 minutes, and the other 25% had patients wait only 10 minutes.
Adverse reactions in the past 6 months, including hives, wheezing, or shortness of breath, were reported by 30% of the offices. None of the nurses reported having had a case of anaphylaxis, Dr. Reddy said.
All of the offices’ nurses said that they felt prepared to handle adverse reactions, and all said that they stock epinephrine, oxygen, and needles. The ability to administer antihistamines, corticosteroids, airway maintenance, and intravenous fluids was reported by 91%, 93%, 93%, and 70% of offices, respectively.
When offered in-service education on the AIT practice parameter, only 42% expressed interest. The other 58% stated that they did not have the time, Dr. Reddy reported.
During the question and answer period, an audience member commented that "it would be interesting" to conduct the same assessment in allergy practices, noting that "it may be about equal."
Dr. Reddy stated that she had no financial disclosures.
ORLANDO – A survey of primary care offices revealed that the majority were not fully equipped to handle adverse reactions to allergen immunotherapy.
"Primary care offices require further education on administration of allergen immunotherapy to ensure adherence to the updated practice parameter, as well as to ensure safety," Dr. Vinitha Reddy said at the annual meeting of the American Academy of Allergy, Asthma and Immunology.
The telephone survey involved a questionnaire administered to nurses at 43 primary care offices (35 family medicine, 4 pediatric, and 4 internal medicine) that administer allergen immunotherapy (AIT). The practices employed a total 194 physicians, and administered AIT to approximately 500 patients.
The survey assessed adherence to the recommendations in "Allergen immunotherapy: a practice parameter third update," a joint guideline issued by the American Academy of Allergy, Asthma, and Immunology and the American College of Allergy, Asthma, and Immunology (J. Allergy Clin. Immunol. 2011;127:S1-55).
The guideline states that allergists’ offices are the preferred setting, but that primary care offices can perform AIT if the appropriate personnel and equipment are available to identify and treat anaphylaxis or other emergencies. The risk is low – approximately 0.2% of patients experience systemic reactions per injection, and deaths occur in just 1 per 2.5 million injections. However, the risk is not nonexistent, said Dr. Reddy, a second-year postgraduate fellow at Pennsylvania State University, Hershey.
Only 53% of the offices reported performing the recommended preinjection health assessment, which should include a determination of the presence of asthma and the degree of asthma control. Only 23% reported measuring peak flow, which is suggested as a method for assessing lung function in asthma patients.
Just 33% of the practices reported routinely having patients wait the recommended 30 minutes following AIT before leaving the office. Another 42% had patients wait just 20 minutes, and the other 25% had patients wait only 10 minutes.
Adverse reactions in the past 6 months, including hives, wheezing, or shortness of breath, were reported by 30% of the offices. None of the nurses reported having had a case of anaphylaxis, Dr. Reddy said.
All of the offices’ nurses said that they felt prepared to handle adverse reactions, and all said that they stock epinephrine, oxygen, and needles. The ability to administer antihistamines, corticosteroids, airway maintenance, and intravenous fluids was reported by 91%, 93%, 93%, and 70% of offices, respectively.
When offered in-service education on the AIT practice parameter, only 42% expressed interest. The other 58% stated that they did not have the time, Dr. Reddy reported.
During the question and answer period, an audience member commented that "it would be interesting" to conduct the same assessment in allergy practices, noting that "it may be about equal."
Dr. Reddy stated that she had no financial disclosures.
ORLANDO – A survey of primary care offices revealed that the majority were not fully equipped to handle adverse reactions to allergen immunotherapy.
"Primary care offices require further education on administration of allergen immunotherapy to ensure adherence to the updated practice parameter, as well as to ensure safety," Dr. Vinitha Reddy said at the annual meeting of the American Academy of Allergy, Asthma and Immunology.
The telephone survey involved a questionnaire administered to nurses at 43 primary care offices (35 family medicine, 4 pediatric, and 4 internal medicine) that administer allergen immunotherapy (AIT). The practices employed a total 194 physicians, and administered AIT to approximately 500 patients.
The survey assessed adherence to the recommendations in "Allergen immunotherapy: a practice parameter third update," a joint guideline issued by the American Academy of Allergy, Asthma, and Immunology and the American College of Allergy, Asthma, and Immunology (J. Allergy Clin. Immunol. 2011;127:S1-55).
The guideline states that allergists’ offices are the preferred setting, but that primary care offices can perform AIT if the appropriate personnel and equipment are available to identify and treat anaphylaxis or other emergencies. The risk is low – approximately 0.2% of patients experience systemic reactions per injection, and deaths occur in just 1 per 2.5 million injections. However, the risk is not nonexistent, said Dr. Reddy, a second-year postgraduate fellow at Pennsylvania State University, Hershey.
Only 53% of the offices reported performing the recommended preinjection health assessment, which should include a determination of the presence of asthma and the degree of asthma control. Only 23% reported measuring peak flow, which is suggested as a method for assessing lung function in asthma patients.
Just 33% of the practices reported routinely having patients wait the recommended 30 minutes following AIT before leaving the office. Another 42% had patients wait just 20 minutes, and the other 25% had patients wait only 10 minutes.
Adverse reactions in the past 6 months, including hives, wheezing, or shortness of breath, were reported by 30% of the offices. None of the nurses reported having had a case of anaphylaxis, Dr. Reddy said.
All of the offices’ nurses said that they felt prepared to handle adverse reactions, and all said that they stock epinephrine, oxygen, and needles. The ability to administer antihistamines, corticosteroids, airway maintenance, and intravenous fluids was reported by 91%, 93%, 93%, and 70% of offices, respectively.
When offered in-service education on the AIT practice parameter, only 42% expressed interest. The other 58% stated that they did not have the time, Dr. Reddy reported.
During the question and answer period, an audience member commented that "it would be interesting" to conduct the same assessment in allergy practices, noting that "it may be about equal."
Dr. Reddy stated that she had no financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF ALLERGY, ASTHMA AND IMMUNOLOGY
Asthma Meds: Men Prefer to Inhale, Women to Take Pills
ORLANDO – Men may be more adherent than women are in their use of asthma controller medications, but adherence overall is poor, according to an analysis of claims from a large database.
Older adults were also more adherent than were younger adults in the retrospective cohort study, which included medical and pharmacy claims for 145,950 patients aged 18-65 years who were seen from January 2006 to December 2008.
To be included, patients included had to have 12 months or more of continuous medical and pharmacy eligibility following the index date (6 months after first medical claim for asthma), said Leslie J. Hinyard, Ph.D. at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Mean age at enrollment was 41 years for the 53,532 men, and 43 years for the 92,418 women. Adherence was defined by the Medication Possession Ratio (MPR), calculated as the number of days for which the medication is supplied divided by the number of days from first fill through the end of study eligibility.
Overall adherence, stratified by gender, ranged from a low MPR of 0.246 for inhaled corticosteroid use among women to a high of 0.597 for omalizumab use in women.
"I was actually surprised by how poor adherence was across the board. It’s actually consistent with what you find throughout the literature; but the adherence to what should be medicines taken daily over a long period of time is rather poor," said Dr. Hinyard, who is with the St. Louis University Center for Outcomes Research and also the university’s department of internal medicine, division of allergy.
Men had significantly greater adherence than did women for inhaled corticosteroid (MPR, 0.259 vs. 0.246), long-acting beta-agonists (0.384 vs. 0.345), combinations of the two (0.388 vs. 0.352), and for mast cell–stabilizing agents (0.238 vs. 0.205). On the other hand, women were significantly more adherent than men were for leukotriene modifiers (0.499 vs. 0.472).
"I was intrigued by the fact that, across the board, men seemed to have slightly higher adherence than women, except for leukotriene modifiers, which are pills. The rest are all inhaled," Dr. Hinyard said in an interview.
She noted that the explanation could be that women are more accustomed than are men to taking pills, rather than that men are more apt to use inhalers. However, she emphasized, "I’m completely speculating."
There was no statistically significant difference between men and women for oral steroid use, monoclonal antibodies, or theophylline.
Examined by age tertiles of 18-34 years, 35-49 years, and 50-65 years, there was a positive linear association between age and adherence for all drug categories except for chronic oral steroids.
For example, with inhaled corticosteroids, 45% of those aged 50-65 years fell into the top tertile for adherence, compared with 16% of the 18- to 34-year-olds. "Older adults were more adherent, especially compared to the younger adults," she noted.
"We need to understand if there’s a minimal threshold for adherence that will keep people from having exacerbations," Dr. Hinyard said. "We’re never going to get perfect adherence, but perhaps we can set a more realistic goal for people to strive for."
Dr. Hinyard had no relevant disclosures.
ORLANDO – Men may be more adherent than women are in their use of asthma controller medications, but adherence overall is poor, according to an analysis of claims from a large database.
Older adults were also more adherent than were younger adults in the retrospective cohort study, which included medical and pharmacy claims for 145,950 patients aged 18-65 years who were seen from January 2006 to December 2008.
To be included, patients included had to have 12 months or more of continuous medical and pharmacy eligibility following the index date (6 months after first medical claim for asthma), said Leslie J. Hinyard, Ph.D. at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Mean age at enrollment was 41 years for the 53,532 men, and 43 years for the 92,418 women. Adherence was defined by the Medication Possession Ratio (MPR), calculated as the number of days for which the medication is supplied divided by the number of days from first fill through the end of study eligibility.
Overall adherence, stratified by gender, ranged from a low MPR of 0.246 for inhaled corticosteroid use among women to a high of 0.597 for omalizumab use in women.
"I was actually surprised by how poor adherence was across the board. It’s actually consistent with what you find throughout the literature; but the adherence to what should be medicines taken daily over a long period of time is rather poor," said Dr. Hinyard, who is with the St. Louis University Center for Outcomes Research and also the university’s department of internal medicine, division of allergy.
Men had significantly greater adherence than did women for inhaled corticosteroid (MPR, 0.259 vs. 0.246), long-acting beta-agonists (0.384 vs. 0.345), combinations of the two (0.388 vs. 0.352), and for mast cell–stabilizing agents (0.238 vs. 0.205). On the other hand, women were significantly more adherent than men were for leukotriene modifiers (0.499 vs. 0.472).
"I was intrigued by the fact that, across the board, men seemed to have slightly higher adherence than women, except for leukotriene modifiers, which are pills. The rest are all inhaled," Dr. Hinyard said in an interview.
She noted that the explanation could be that women are more accustomed than are men to taking pills, rather than that men are more apt to use inhalers. However, she emphasized, "I’m completely speculating."
There was no statistically significant difference between men and women for oral steroid use, monoclonal antibodies, or theophylline.
Examined by age tertiles of 18-34 years, 35-49 years, and 50-65 years, there was a positive linear association between age and adherence for all drug categories except for chronic oral steroids.
For example, with inhaled corticosteroids, 45% of those aged 50-65 years fell into the top tertile for adherence, compared with 16% of the 18- to 34-year-olds. "Older adults were more adherent, especially compared to the younger adults," she noted.
"We need to understand if there’s a minimal threshold for adherence that will keep people from having exacerbations," Dr. Hinyard said. "We’re never going to get perfect adherence, but perhaps we can set a more realistic goal for people to strive for."
Dr. Hinyard had no relevant disclosures.
ORLANDO – Men may be more adherent than women are in their use of asthma controller medications, but adherence overall is poor, according to an analysis of claims from a large database.
Older adults were also more adherent than were younger adults in the retrospective cohort study, which included medical and pharmacy claims for 145,950 patients aged 18-65 years who were seen from January 2006 to December 2008.
To be included, patients included had to have 12 months or more of continuous medical and pharmacy eligibility following the index date (6 months after first medical claim for asthma), said Leslie J. Hinyard, Ph.D. at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Mean age at enrollment was 41 years for the 53,532 men, and 43 years for the 92,418 women. Adherence was defined by the Medication Possession Ratio (MPR), calculated as the number of days for which the medication is supplied divided by the number of days from first fill through the end of study eligibility.
Overall adherence, stratified by gender, ranged from a low MPR of 0.246 for inhaled corticosteroid use among women to a high of 0.597 for omalizumab use in women.
"I was actually surprised by how poor adherence was across the board. It’s actually consistent with what you find throughout the literature; but the adherence to what should be medicines taken daily over a long period of time is rather poor," said Dr. Hinyard, who is with the St. Louis University Center for Outcomes Research and also the university’s department of internal medicine, division of allergy.
Men had significantly greater adherence than did women for inhaled corticosteroid (MPR, 0.259 vs. 0.246), long-acting beta-agonists (0.384 vs. 0.345), combinations of the two (0.388 vs. 0.352), and for mast cell–stabilizing agents (0.238 vs. 0.205). On the other hand, women were significantly more adherent than men were for leukotriene modifiers (0.499 vs. 0.472).
"I was intrigued by the fact that, across the board, men seemed to have slightly higher adherence than women, except for leukotriene modifiers, which are pills. The rest are all inhaled," Dr. Hinyard said in an interview.
She noted that the explanation could be that women are more accustomed than are men to taking pills, rather than that men are more apt to use inhalers. However, she emphasized, "I’m completely speculating."
There was no statistically significant difference between men and women for oral steroid use, monoclonal antibodies, or theophylline.
Examined by age tertiles of 18-34 years, 35-49 years, and 50-65 years, there was a positive linear association between age and adherence for all drug categories except for chronic oral steroids.
For example, with inhaled corticosteroids, 45% of those aged 50-65 years fell into the top tertile for adherence, compared with 16% of the 18- to 34-year-olds. "Older adults were more adherent, especially compared to the younger adults," she noted.
"We need to understand if there’s a minimal threshold for adherence that will keep people from having exacerbations," Dr. Hinyard said. "We’re never going to get perfect adherence, but perhaps we can set a more realistic goal for people to strive for."
Dr. Hinyard had no relevant disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF ALLERGY, ASTHMA, AND IMMUNOLOGY
Asthma Guidelines Are Not Always Followed
ORLANDO – The 2007 National Asthma Education and Prevention Program guidelines are viewed as cumbersome and too time-consuming by some primary care physicians caring for children.
That finding, from a 23-question survey sent to 80 pediatricians, suggests that "there is room for improvement. I think the allergist can play a big role in helping co-manage severe asthmatic patients," Dr. Nabeel Farooqui of Ohio State University and Nationwide Children’s Hospital, Columbus, Ohio, said in an interview at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The Expert Panel Report 3 (EPR3): Guidelines for the Diagnosis and Management of Asthma, published by the National Heart, Lung, and Blood Institute, recommend referral to an asthma subspecialist for patients with difficult-to-manage moderate to severe persistent asthma, when a diagnosis is in question, or for patients who need additional testing. Such a subspecialist could be either an allergist or a pulmonologist, but only the allergist can do an allergy assessment, Dr. Farooqui noted.
"Many children with asthma also have allergic rhinitis. These children should be skin-tested. Allergists can help diagnose and treat some of the allergic comorbidities associated with asthma such as allergic rhinitis, and thereby get better control of the asthma," he said.
Of the 28 pediatricians who responded to the anonymous survey, 13 (46%) said that they "always" follow the NAEPP EPR 3 guidelines. Another 8 (29%) said they follow the guidelines "most of the time." Among those who didn’t always follow the guidelines, the most common reason, given by 11 respondents (39% of the total), was that they are "too cumbersome."
Nearly two-thirds of the respondents (17) reported always initiating inhaled steroids for patients with persistent asthma, whereas the other 11 expressed at least some hesitation. "Concern about nonadherence" to inhaled steroids was listed most often as the reason.
Just 15 of the 28 respondents (54%) reported reviewing a written asthma action plan with their patients at every visit as recommended, whereas another 12 (43%) did so only when they made a change in the patient’s care. Lack of time was listed by eight respondents as the reason not to review the plan at every visit, while another six said they did not think that such plans improve management or outcomes.
While all of the respondents endorsed influenza immunization for all asthma patients, nine mistakenly believed that egg allergy is a contraindication to receipt of the vaccine. (The contraindication was lifted in June 2011.)
While all but one respondent said that they refer difficult-to-manage asthma patients to subspecialists, only half referred for additional testing and less than half for a questionable diagnosis. The majority (69%) refer to pulmonologists, while the other 31% referred patients to both a pulmonologist and an allergist/immunologist. "The only time referral to allergist is made is when the patient is sent to pulmonary as well," Dr. Farooqui commented.
"Lack of timely appointment" was the most common reason given for not referring to a subspecialist.
For patients with well-controlled asthma who are on a daily controller medication, half of the respondents endorsed the recommended evaluation at 3-6 months for consideration of step-down therapy. However, the other half reported waiting a year or never stepping down therapy.
"Allergists should continue to promote their role, both in the community and academic centers, as specialists in the evaluation and management of asthma," Dr. Farooqui advised his colleagues.
Dr. Farooqui said he had no relevant financial disclosures.
ORLANDO – The 2007 National Asthma Education and Prevention Program guidelines are viewed as cumbersome and too time-consuming by some primary care physicians caring for children.
That finding, from a 23-question survey sent to 80 pediatricians, suggests that "there is room for improvement. I think the allergist can play a big role in helping co-manage severe asthmatic patients," Dr. Nabeel Farooqui of Ohio State University and Nationwide Children’s Hospital, Columbus, Ohio, said in an interview at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The Expert Panel Report 3 (EPR3): Guidelines for the Diagnosis and Management of Asthma, published by the National Heart, Lung, and Blood Institute, recommend referral to an asthma subspecialist for patients with difficult-to-manage moderate to severe persistent asthma, when a diagnosis is in question, or for patients who need additional testing. Such a subspecialist could be either an allergist or a pulmonologist, but only the allergist can do an allergy assessment, Dr. Farooqui noted.
"Many children with asthma also have allergic rhinitis. These children should be skin-tested. Allergists can help diagnose and treat some of the allergic comorbidities associated with asthma such as allergic rhinitis, and thereby get better control of the asthma," he said.
Of the 28 pediatricians who responded to the anonymous survey, 13 (46%) said that they "always" follow the NAEPP EPR 3 guidelines. Another 8 (29%) said they follow the guidelines "most of the time." Among those who didn’t always follow the guidelines, the most common reason, given by 11 respondents (39% of the total), was that they are "too cumbersome."
Nearly two-thirds of the respondents (17) reported always initiating inhaled steroids for patients with persistent asthma, whereas the other 11 expressed at least some hesitation. "Concern about nonadherence" to inhaled steroids was listed most often as the reason.
Just 15 of the 28 respondents (54%) reported reviewing a written asthma action plan with their patients at every visit as recommended, whereas another 12 (43%) did so only when they made a change in the patient’s care. Lack of time was listed by eight respondents as the reason not to review the plan at every visit, while another six said they did not think that such plans improve management or outcomes.
While all of the respondents endorsed influenza immunization for all asthma patients, nine mistakenly believed that egg allergy is a contraindication to receipt of the vaccine. (The contraindication was lifted in June 2011.)
While all but one respondent said that they refer difficult-to-manage asthma patients to subspecialists, only half referred for additional testing and less than half for a questionable diagnosis. The majority (69%) refer to pulmonologists, while the other 31% referred patients to both a pulmonologist and an allergist/immunologist. "The only time referral to allergist is made is when the patient is sent to pulmonary as well," Dr. Farooqui commented.
"Lack of timely appointment" was the most common reason given for not referring to a subspecialist.
For patients with well-controlled asthma who are on a daily controller medication, half of the respondents endorsed the recommended evaluation at 3-6 months for consideration of step-down therapy. However, the other half reported waiting a year or never stepping down therapy.
"Allergists should continue to promote their role, both in the community and academic centers, as specialists in the evaluation and management of asthma," Dr. Farooqui advised his colleagues.
Dr. Farooqui said he had no relevant financial disclosures.
ORLANDO – The 2007 National Asthma Education and Prevention Program guidelines are viewed as cumbersome and too time-consuming by some primary care physicians caring for children.
That finding, from a 23-question survey sent to 80 pediatricians, suggests that "there is room for improvement. I think the allergist can play a big role in helping co-manage severe asthmatic patients," Dr. Nabeel Farooqui of Ohio State University and Nationwide Children’s Hospital, Columbus, Ohio, said in an interview at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The Expert Panel Report 3 (EPR3): Guidelines for the Diagnosis and Management of Asthma, published by the National Heart, Lung, and Blood Institute, recommend referral to an asthma subspecialist for patients with difficult-to-manage moderate to severe persistent asthma, when a diagnosis is in question, or for patients who need additional testing. Such a subspecialist could be either an allergist or a pulmonologist, but only the allergist can do an allergy assessment, Dr. Farooqui noted.
"Many children with asthma also have allergic rhinitis. These children should be skin-tested. Allergists can help diagnose and treat some of the allergic comorbidities associated with asthma such as allergic rhinitis, and thereby get better control of the asthma," he said.
Of the 28 pediatricians who responded to the anonymous survey, 13 (46%) said that they "always" follow the NAEPP EPR 3 guidelines. Another 8 (29%) said they follow the guidelines "most of the time." Among those who didn’t always follow the guidelines, the most common reason, given by 11 respondents (39% of the total), was that they are "too cumbersome."
Nearly two-thirds of the respondents (17) reported always initiating inhaled steroids for patients with persistent asthma, whereas the other 11 expressed at least some hesitation. "Concern about nonadherence" to inhaled steroids was listed most often as the reason.
Just 15 of the 28 respondents (54%) reported reviewing a written asthma action plan with their patients at every visit as recommended, whereas another 12 (43%) did so only when they made a change in the patient’s care. Lack of time was listed by eight respondents as the reason not to review the plan at every visit, while another six said they did not think that such plans improve management or outcomes.
While all of the respondents endorsed influenza immunization for all asthma patients, nine mistakenly believed that egg allergy is a contraindication to receipt of the vaccine. (The contraindication was lifted in June 2011.)
While all but one respondent said that they refer difficult-to-manage asthma patients to subspecialists, only half referred for additional testing and less than half for a questionable diagnosis. The majority (69%) refer to pulmonologists, while the other 31% referred patients to both a pulmonologist and an allergist/immunologist. "The only time referral to allergist is made is when the patient is sent to pulmonary as well," Dr. Farooqui commented.
"Lack of timely appointment" was the most common reason given for not referring to a subspecialist.
For patients with well-controlled asthma who are on a daily controller medication, half of the respondents endorsed the recommended evaluation at 3-6 months for consideration of step-down therapy. However, the other half reported waiting a year or never stepping down therapy.
"Allergists should continue to promote their role, both in the community and academic centers, as specialists in the evaluation and management of asthma," Dr. Farooqui advised his colleagues.
Dr. Farooqui said he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF ALLERGY, ASTHMA, AND IMMUNOLOGY
Major Finding: Forty-six percent of pediatricians responded that they "always" follow the NAEPP EPR 3 guidelines. Another 29% said they follow the guidelines "most of the time." Among those who didn’t always follow the guidelines, the most common reason, given by 11 respondents (39% of the total), was that they are "too cumbersome."
Data Source: These were results of a 23-question survey sent to 80 pediatricians, of whom 28 responded.
Disclosures: Dr. Farooqui said he had no relevant financial disclosures.
Panel Explores Hepatitis B Protection Strategies for Providers
ATLANTA – A federal vaccine advisory panel is considering options for ensuring that health care personnel who were vaccinated against hepatitis B in the remote past are currently protected.
Increasing proportions of health care personnel were vaccinated against hepatitis B in infancy, or as a catch-up during adolescence. An estimated 90%-95% of healthy adults aged 40 years and younger achieve a protective antibody titer (defined as anti–hepatitis B surface antigen concentrations of 10 mIU/mL or greater) after receiving the three-dose series (MMWR 2011;60[RR07]:1-45).
However, there is currently no recommendation for postvaccination serologic testing to ensure protection, and records may no longer be available for some people who did have such testing earlier in their careers. Health care schools and other institutions are now seeking guidance to ensure hepatitis B protection for students and workers, said Dr. Sarah Schillie of the Centers for Disease Control and Prevention.
Current CDC recommendations for immunization of health care providers call for all unvaccinated individuals whose activities involve "reasonably anticipated risk for exposure to blood or other infectious body fluid" to be vaccinated with the complete three-dose hepatitis B vaccine series prior to contact with blood. Postvaccination serologic testing (at 1-2 months after the last dose) is recommended for all providers at high risk for occupational exposure to blood or body fluids. Revaccination is recommended for those with hepatitis B surface antigen (anti-HBs) concentrations less than 10 mIU/mL.
However, anti-HBs levels wane over time following vaccination, and an anti-HBs level of less than 10 mIU/mL years after vaccination doesn’t distinguish between people who responded initially and are still protected, those who have a delayed response and would likely respond to a second series of three additional doses, and nonresponders who would be susceptible to infection even after six doses (or who might have past or chronic HBV infection), Dr. Schillie explained.
The following three options are currently under consideration by the CDC’s Advisory Committee on Immunization Practices (ACIP):
• No action unless exposed. This strategy relies on the provider’s timely reporting of a percutaneous or mucosal exposure to blood or body fluids. The individual is then assessed for both hepatitis B vaccination history and serologic anti-HBs testing, and the source patient is tested for anti-HBs. The decision to give hepatitis B vaccination (HBV) and/or hepatitis B immune globulin is based on the results.
• Pre-exposure anti-HBs. If the level is above 10 mIU/mL, no further action is needed. If the anti-HBs level is less than 10 mIU/mL, one dose of HBV is given and the individual is retested 1-2 months later. If the anti-HBs level is still less than 10 mIU/mL, current recommendations for revaccination and retesting should be followed.
• Challenge dose of HBV. One dose is given at the time of school matriculation or hiring, and the anti-HBs level is measured 1-2 months later. If the level is more than 10 mIU/mL, no further action is needed. If it’s less than 10 mIU/mL, then current recommendations for revaccination and retesting should be followed.
Many health care systems in the United States are already using one of these options, Dr. Schillie noted.
Dr. Mark H. Sawyer, chair of the ACIP hepatitis B working group, said that a cost-effectiveness analysis of the three options will be presented to the ACIP at its June 2012 meeting, and a vote could be taken at its October 2012 meeting.
Dr. Sawyer and Dr. Schillie have no relevant disclosures.
ATLANTA – A federal vaccine advisory panel is considering options for ensuring that health care personnel who were vaccinated against hepatitis B in the remote past are currently protected.
Increasing proportions of health care personnel were vaccinated against hepatitis B in infancy, or as a catch-up during adolescence. An estimated 90%-95% of healthy adults aged 40 years and younger achieve a protective antibody titer (defined as anti–hepatitis B surface antigen concentrations of 10 mIU/mL or greater) after receiving the three-dose series (MMWR 2011;60[RR07]:1-45).
However, there is currently no recommendation for postvaccination serologic testing to ensure protection, and records may no longer be available for some people who did have such testing earlier in their careers. Health care schools and other institutions are now seeking guidance to ensure hepatitis B protection for students and workers, said Dr. Sarah Schillie of the Centers for Disease Control and Prevention.
Current CDC recommendations for immunization of health care providers call for all unvaccinated individuals whose activities involve "reasonably anticipated risk for exposure to blood or other infectious body fluid" to be vaccinated with the complete three-dose hepatitis B vaccine series prior to contact with blood. Postvaccination serologic testing (at 1-2 months after the last dose) is recommended for all providers at high risk for occupational exposure to blood or body fluids. Revaccination is recommended for those with hepatitis B surface antigen (anti-HBs) concentrations less than 10 mIU/mL.
However, anti-HBs levels wane over time following vaccination, and an anti-HBs level of less than 10 mIU/mL years after vaccination doesn’t distinguish between people who responded initially and are still protected, those who have a delayed response and would likely respond to a second series of three additional doses, and nonresponders who would be susceptible to infection even after six doses (or who might have past or chronic HBV infection), Dr. Schillie explained.
The following three options are currently under consideration by the CDC’s Advisory Committee on Immunization Practices (ACIP):
• No action unless exposed. This strategy relies on the provider’s timely reporting of a percutaneous or mucosal exposure to blood or body fluids. The individual is then assessed for both hepatitis B vaccination history and serologic anti-HBs testing, and the source patient is tested for anti-HBs. The decision to give hepatitis B vaccination (HBV) and/or hepatitis B immune globulin is based on the results.
• Pre-exposure anti-HBs. If the level is above 10 mIU/mL, no further action is needed. If the anti-HBs level is less than 10 mIU/mL, one dose of HBV is given and the individual is retested 1-2 months later. If the anti-HBs level is still less than 10 mIU/mL, current recommendations for revaccination and retesting should be followed.
• Challenge dose of HBV. One dose is given at the time of school matriculation or hiring, and the anti-HBs level is measured 1-2 months later. If the level is more than 10 mIU/mL, no further action is needed. If it’s less than 10 mIU/mL, then current recommendations for revaccination and retesting should be followed.
Many health care systems in the United States are already using one of these options, Dr. Schillie noted.
Dr. Mark H. Sawyer, chair of the ACIP hepatitis B working group, said that a cost-effectiveness analysis of the three options will be presented to the ACIP at its June 2012 meeting, and a vote could be taken at its October 2012 meeting.
Dr. Sawyer and Dr. Schillie have no relevant disclosures.
ATLANTA – A federal vaccine advisory panel is considering options for ensuring that health care personnel who were vaccinated against hepatitis B in the remote past are currently protected.
Increasing proportions of health care personnel were vaccinated against hepatitis B in infancy, or as a catch-up during adolescence. An estimated 90%-95% of healthy adults aged 40 years and younger achieve a protective antibody titer (defined as anti–hepatitis B surface antigen concentrations of 10 mIU/mL or greater) after receiving the three-dose series (MMWR 2011;60[RR07]:1-45).
However, there is currently no recommendation for postvaccination serologic testing to ensure protection, and records may no longer be available for some people who did have such testing earlier in their careers. Health care schools and other institutions are now seeking guidance to ensure hepatitis B protection for students and workers, said Dr. Sarah Schillie of the Centers for Disease Control and Prevention.
Current CDC recommendations for immunization of health care providers call for all unvaccinated individuals whose activities involve "reasonably anticipated risk for exposure to blood or other infectious body fluid" to be vaccinated with the complete three-dose hepatitis B vaccine series prior to contact with blood. Postvaccination serologic testing (at 1-2 months after the last dose) is recommended for all providers at high risk for occupational exposure to blood or body fluids. Revaccination is recommended for those with hepatitis B surface antigen (anti-HBs) concentrations less than 10 mIU/mL.
However, anti-HBs levels wane over time following vaccination, and an anti-HBs level of less than 10 mIU/mL years after vaccination doesn’t distinguish between people who responded initially and are still protected, those who have a delayed response and would likely respond to a second series of three additional doses, and nonresponders who would be susceptible to infection even after six doses (or who might have past or chronic HBV infection), Dr. Schillie explained.
The following three options are currently under consideration by the CDC’s Advisory Committee on Immunization Practices (ACIP):
• No action unless exposed. This strategy relies on the provider’s timely reporting of a percutaneous or mucosal exposure to blood or body fluids. The individual is then assessed for both hepatitis B vaccination history and serologic anti-HBs testing, and the source patient is tested for anti-HBs. The decision to give hepatitis B vaccination (HBV) and/or hepatitis B immune globulin is based on the results.
• Pre-exposure anti-HBs. If the level is above 10 mIU/mL, no further action is needed. If the anti-HBs level is less than 10 mIU/mL, one dose of HBV is given and the individual is retested 1-2 months later. If the anti-HBs level is still less than 10 mIU/mL, current recommendations for revaccination and retesting should be followed.
• Challenge dose of HBV. One dose is given at the time of school matriculation or hiring, and the anti-HBs level is measured 1-2 months later. If the level is more than 10 mIU/mL, no further action is needed. If it’s less than 10 mIU/mL, then current recommendations for revaccination and retesting should be followed.
Many health care systems in the United States are already using one of these options, Dr. Schillie noted.
Dr. Mark H. Sawyer, chair of the ACIP hepatitis B working group, said that a cost-effectiveness analysis of the three options will be presented to the ACIP at its June 2012 meeting, and a vote could be taken at its October 2012 meeting.
Dr. Sawyer and Dr. Schillie have no relevant disclosures.
FROM A MEETING OF THE CENTERS FOR DISEASE CONTROL AND PREVENTION'S ADVISORY COMMITTEE ON IMMUNIZATION PRACTICES
Vaccine Advisory Panel Holds Off on Adult PCV13 Recommendation
ATLANTA – The Centers for Disease Control and Prevention’s vaccine advisory panel is holding off on a recommendation for use of the 13-valent conjugate pneumococcal vaccine in adults, despite its December 2011 licensure for those aged 50 years and older.
At its February 2012 meeting, the Advisory Committee on Immunization Practices reviewed the evidence for the use of PCV13 in adults aged 65 years and older and in immunocompromised adults. The full committee agreed with a prior recommendation from its Pneumococcal Vaccines Work Group that there is insufficient evidence at this time to determine the value of immunizing adults with PCV13. Currently missing are data on efficacy, expected to be available in 2013 from an ongoing Dutch trial, and information about the indirect impact of the current routine use of PCV13 in children on herd immunity in adults, work group chair Dr. Nancy M. Bennett said.
In contrast to ACIP’s evidence-based approach, the Food and Drug Administration based its adult approval of PCV13 (Pfizer’s Prevnar 13) on noninferiority, compared with the 23-valent pneumococcal polysaccharide (PPSV23) vaccine, and on safety, explained Dr. Bennett, professor of medicine and community and preventive medicine at the University of Rochester (N.Y.).
PCV13 "is very promising in the prevention of pneumococcal disease in all age groups. ... The ACIP was asked to consider all of the evidence related to the use of the vaccine in adults. At this time, we are not prepared to make a recommendation. We are in the process of reevaluating all the evidence that’s available. The important thing to realize is that there’s a difference between licensure by the FDA and a recommendation from the ACIP," she said in an interview.
The current plan – subject to revision – is for ACIP to vote at its June 2012 meeting on use of PCV13 in immunocompromised adults (for whom the data in favor of PCV13 use are stronger) and to review data from the CDC’s Active Bacterial Core surveillance to see whether routine use of PCV13 in children indirectly reduces the rates of disease in adults, as occurred with the previous 7-valent pneumococcal conjugate vaccine (J. Infect. Dis. 2010;201:32-41)
In 2013, ACIP is expected to review data from the Community Acquired Pneumonia Immunization Trial in Adults (CAPITA), a randomized, placebo-controlled trial involving 85,000 community-dwelling, pneumococcal vaccine–naive adults aged 65 years and older being conducted in the Netherlands. The study’s primary objective is to determine the efficacy of PCV13 against vaccine-serotype community-acquired pneumonia (CAP). Secondary objectives are to evaluate efficacy against nonbacteremic vaccine-type CAP and vaccine-type invasive pneumococcal disease, all pneumococcal CAP, and death (Neth. J. Med. 2008;66:378-83).
Depending on when those data become available, a vote on the use of PCV13 in adults aged 65 and older (the age group currently targeted for PPSV23) could take place in February or June 2013, Dr. Bennett said.
Of course, PCV13 is licensed for adults aged 50 years and older, so physicians are able to use the vaccine in those individuals if they so choose. In an interview, Dr. William Schaffner, a member of the pneumococcal work group, said that he has received numerous questions from physicians about use of PCV13 in adults. For example, they want to know if patients who are already immunized with PPSV23 might benefit from receiving a dose of PCV13 since it provides better immunogenicity, better boosting, and unlike PPSV23, is likely to eliminate pneumococcal carriage.
Moreover, physicians have asked him whether new patients who are eligible for pneumococcal vaccine should be given PCV13, PPSV23, or perhaps both (to obtain both the presumed advantages of PCV13 along with protection against the extra 10 serotypes from PPSV23). And if they should be given both, can the two vaccines be administered simultaneously or must they be given in sequence? And if in sequence, which vaccine should be given first and what should be the interval in between?
Those questions have not been answered as yet, but, Dr. Schaffner said in an interview, "If I have a 67-year-old patient who is overweight, has diabetes, and has smoked – not an unusual combination – I want to do everything I can to make this specific patient who is sitting in front of me have the maximum protection. [Then] there may be a clinical reason to use this vaccine. Sometimes the public health decision may be different from an individualized clinical decision."
Dr. Schaffner, chair of the department of preventive medicine at Vanderbilt University in Nashville, Tenn., said data suggest that such a patient would likely receive maximal protection with a dose of PCV13 first, followed by a PPSV23 dose 6-8 weeks later. He acknowledged that this might be impractical for a variety of reasons. For one thing, although Medicare is expected to cover PCV13 under Part B, it is not clear whether it would cover both vaccines given in sequence. Thus, he said, "at the moment I would say to physicians, ‘Keep doing what you’re doing, and stay tuned.’ "
Dr. Bennett stated that she has no disclosures. Dr. Schaffner serves on the data safety monitoring board for Sanofi-Pasteur and Merck, and is an occasional consultant for Novartis, GlaxoSmithKline, and Pfizer.
ATLANTA – The Centers for Disease Control and Prevention’s vaccine advisory panel is holding off on a recommendation for use of the 13-valent conjugate pneumococcal vaccine in adults, despite its December 2011 licensure for those aged 50 years and older.
At its February 2012 meeting, the Advisory Committee on Immunization Practices reviewed the evidence for the use of PCV13 in adults aged 65 years and older and in immunocompromised adults. The full committee agreed with a prior recommendation from its Pneumococcal Vaccines Work Group that there is insufficient evidence at this time to determine the value of immunizing adults with PCV13. Currently missing are data on efficacy, expected to be available in 2013 from an ongoing Dutch trial, and information about the indirect impact of the current routine use of PCV13 in children on herd immunity in adults, work group chair Dr. Nancy M. Bennett said.
In contrast to ACIP’s evidence-based approach, the Food and Drug Administration based its adult approval of PCV13 (Pfizer’s Prevnar 13) on noninferiority, compared with the 23-valent pneumococcal polysaccharide (PPSV23) vaccine, and on safety, explained Dr. Bennett, professor of medicine and community and preventive medicine at the University of Rochester (N.Y.).
PCV13 "is very promising in the prevention of pneumococcal disease in all age groups. ... The ACIP was asked to consider all of the evidence related to the use of the vaccine in adults. At this time, we are not prepared to make a recommendation. We are in the process of reevaluating all the evidence that’s available. The important thing to realize is that there’s a difference between licensure by the FDA and a recommendation from the ACIP," she said in an interview.
The current plan – subject to revision – is for ACIP to vote at its June 2012 meeting on use of PCV13 in immunocompromised adults (for whom the data in favor of PCV13 use are stronger) and to review data from the CDC’s Active Bacterial Core surveillance to see whether routine use of PCV13 in children indirectly reduces the rates of disease in adults, as occurred with the previous 7-valent pneumococcal conjugate vaccine (J. Infect. Dis. 2010;201:32-41)
In 2013, ACIP is expected to review data from the Community Acquired Pneumonia Immunization Trial in Adults (CAPITA), a randomized, placebo-controlled trial involving 85,000 community-dwelling, pneumococcal vaccine–naive adults aged 65 years and older being conducted in the Netherlands. The study’s primary objective is to determine the efficacy of PCV13 against vaccine-serotype community-acquired pneumonia (CAP). Secondary objectives are to evaluate efficacy against nonbacteremic vaccine-type CAP and vaccine-type invasive pneumococcal disease, all pneumococcal CAP, and death (Neth. J. Med. 2008;66:378-83).
Depending on when those data become available, a vote on the use of PCV13 in adults aged 65 and older (the age group currently targeted for PPSV23) could take place in February or June 2013, Dr. Bennett said.
Of course, PCV13 is licensed for adults aged 50 years and older, so physicians are able to use the vaccine in those individuals if they so choose. In an interview, Dr. William Schaffner, a member of the pneumococcal work group, said that he has received numerous questions from physicians about use of PCV13 in adults. For example, they want to know if patients who are already immunized with PPSV23 might benefit from receiving a dose of PCV13 since it provides better immunogenicity, better boosting, and unlike PPSV23, is likely to eliminate pneumococcal carriage.
Moreover, physicians have asked him whether new patients who are eligible for pneumococcal vaccine should be given PCV13, PPSV23, or perhaps both (to obtain both the presumed advantages of PCV13 along with protection against the extra 10 serotypes from PPSV23). And if they should be given both, can the two vaccines be administered simultaneously or must they be given in sequence? And if in sequence, which vaccine should be given first and what should be the interval in between?
Those questions have not been answered as yet, but, Dr. Schaffner said in an interview, "If I have a 67-year-old patient who is overweight, has diabetes, and has smoked – not an unusual combination – I want to do everything I can to make this specific patient who is sitting in front of me have the maximum protection. [Then] there may be a clinical reason to use this vaccine. Sometimes the public health decision may be different from an individualized clinical decision."
Dr. Schaffner, chair of the department of preventive medicine at Vanderbilt University in Nashville, Tenn., said data suggest that such a patient would likely receive maximal protection with a dose of PCV13 first, followed by a PPSV23 dose 6-8 weeks later. He acknowledged that this might be impractical for a variety of reasons. For one thing, although Medicare is expected to cover PCV13 under Part B, it is not clear whether it would cover both vaccines given in sequence. Thus, he said, "at the moment I would say to physicians, ‘Keep doing what you’re doing, and stay tuned.’ "
Dr. Bennett stated that she has no disclosures. Dr. Schaffner serves on the data safety monitoring board for Sanofi-Pasteur and Merck, and is an occasional consultant for Novartis, GlaxoSmithKline, and Pfizer.
ATLANTA – The Centers for Disease Control and Prevention’s vaccine advisory panel is holding off on a recommendation for use of the 13-valent conjugate pneumococcal vaccine in adults, despite its December 2011 licensure for those aged 50 years and older.
At its February 2012 meeting, the Advisory Committee on Immunization Practices reviewed the evidence for the use of PCV13 in adults aged 65 years and older and in immunocompromised adults. The full committee agreed with a prior recommendation from its Pneumococcal Vaccines Work Group that there is insufficient evidence at this time to determine the value of immunizing adults with PCV13. Currently missing are data on efficacy, expected to be available in 2013 from an ongoing Dutch trial, and information about the indirect impact of the current routine use of PCV13 in children on herd immunity in adults, work group chair Dr. Nancy M. Bennett said.
In contrast to ACIP’s evidence-based approach, the Food and Drug Administration based its adult approval of PCV13 (Pfizer’s Prevnar 13) on noninferiority, compared with the 23-valent pneumococcal polysaccharide (PPSV23) vaccine, and on safety, explained Dr. Bennett, professor of medicine and community and preventive medicine at the University of Rochester (N.Y.).
PCV13 "is very promising in the prevention of pneumococcal disease in all age groups. ... The ACIP was asked to consider all of the evidence related to the use of the vaccine in adults. At this time, we are not prepared to make a recommendation. We are in the process of reevaluating all the evidence that’s available. The important thing to realize is that there’s a difference between licensure by the FDA and a recommendation from the ACIP," she said in an interview.
The current plan – subject to revision – is for ACIP to vote at its June 2012 meeting on use of PCV13 in immunocompromised adults (for whom the data in favor of PCV13 use are stronger) and to review data from the CDC’s Active Bacterial Core surveillance to see whether routine use of PCV13 in children indirectly reduces the rates of disease in adults, as occurred with the previous 7-valent pneumococcal conjugate vaccine (J. Infect. Dis. 2010;201:32-41)
In 2013, ACIP is expected to review data from the Community Acquired Pneumonia Immunization Trial in Adults (CAPITA), a randomized, placebo-controlled trial involving 85,000 community-dwelling, pneumococcal vaccine–naive adults aged 65 years and older being conducted in the Netherlands. The study’s primary objective is to determine the efficacy of PCV13 against vaccine-serotype community-acquired pneumonia (CAP). Secondary objectives are to evaluate efficacy against nonbacteremic vaccine-type CAP and vaccine-type invasive pneumococcal disease, all pneumococcal CAP, and death (Neth. J. Med. 2008;66:378-83).
Depending on when those data become available, a vote on the use of PCV13 in adults aged 65 and older (the age group currently targeted for PPSV23) could take place in February or June 2013, Dr. Bennett said.
Of course, PCV13 is licensed for adults aged 50 years and older, so physicians are able to use the vaccine in those individuals if they so choose. In an interview, Dr. William Schaffner, a member of the pneumococcal work group, said that he has received numerous questions from physicians about use of PCV13 in adults. For example, they want to know if patients who are already immunized with PPSV23 might benefit from receiving a dose of PCV13 since it provides better immunogenicity, better boosting, and unlike PPSV23, is likely to eliminate pneumococcal carriage.
Moreover, physicians have asked him whether new patients who are eligible for pneumococcal vaccine should be given PCV13, PPSV23, or perhaps both (to obtain both the presumed advantages of PCV13 along with protection against the extra 10 serotypes from PPSV23). And if they should be given both, can the two vaccines be administered simultaneously or must they be given in sequence? And if in sequence, which vaccine should be given first and what should be the interval in between?
Those questions have not been answered as yet, but, Dr. Schaffner said in an interview, "If I have a 67-year-old patient who is overweight, has diabetes, and has smoked – not an unusual combination – I want to do everything I can to make this specific patient who is sitting in front of me have the maximum protection. [Then] there may be a clinical reason to use this vaccine. Sometimes the public health decision may be different from an individualized clinical decision."
Dr. Schaffner, chair of the department of preventive medicine at Vanderbilt University in Nashville, Tenn., said data suggest that such a patient would likely receive maximal protection with a dose of PCV13 first, followed by a PPSV23 dose 6-8 weeks later. He acknowledged that this might be impractical for a variety of reasons. For one thing, although Medicare is expected to cover PCV13 under Part B, it is not clear whether it would cover both vaccines given in sequence. Thus, he said, "at the moment I would say to physicians, ‘Keep doing what you’re doing, and stay tuned.’ "
Dr. Bennett stated that she has no disclosures. Dr. Schaffner serves on the data safety monitoring board for Sanofi-Pasteur and Merck, and is an occasional consultant for Novartis, GlaxoSmithKline, and Pfizer.
FROM A MEETING OF THE CENTERS FOR DISEASE CONTROL AND PREVENTION'S ADVISORY COMMITTEE ON IMMUNIZATION PRACTICES
Case of the Month: Subungual Myxoma
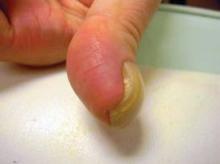
LAS VEGAS – A 39-year-old male patient presented with a 5-year history of an isolated, nonpainful, nonfluctuant left thumbnail deformity. He had no other skin findings, and no other known medical problems. His thumbnail was distinctly curved, with what appeared to be a slight pinkish growth underneath the nail. The nail was avulsed, revealing a readily visible and protruding tumor from the nail bed, extending from underneath the nail matrix to the level of the distal phalanx, with only a small portion of the nail bed not affected.
Digital myxomas are rare neoplasms, and the subungual location is rarer still, according to Dr. Miriam S. Bettencourt.
There have been just four mentions of subungual myxoma in the medical literature since 1982, all of which were case reports. Surgical excision is usually curative, without the need for repeat surgery. One previous case report described a midlateral approach that spared the nail apparatus and demonstrated remodeling of the nail despite a void left under the nail matrix after excision of the tumor (J. Hand Surg. Am. 1998;23:178-80).
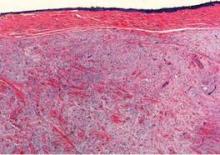
In this case, the patient’s histology revealed a large, circumscribed nonencapsulated nodular deposition of myxoid material, associated with a slight increase in spindle-shaped fibroblasts and thin intervening fibrocollagenous tissue underneath the nail bed epithelium. Scattered mast cells were visible within the lesion.
The tumor was sharply excised and the nail bed was roughly debrided using a small curette. A piece of tinfoil from a suture pack was fashioned into a pseudo nail shape and sutured beneath the patient’s cuticle in order to maintain the opening of the nail matrix during the excision, said Dr. Bettencourt, who presented the case at the Las Vegas Dermatology Seminar, sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis included subungual glomus tumor, which causes a bluish-red discoloration of the nail plate and, unlike myxoma, is painful and temperature-sensitive. Onychomatricoma can cause a nail curvature similar to that of myxoma. However, this rare benign tumor of the nail matrix causes a yellowish discoloration and splinter hemorrhages, which are not seen with myxoma.
Fibromas in the digits, including periungual fibroma (a marker of tuberous sclerosis), acquired digital fibrokeratoma, dermatofibroma, and recurrent digital fibrous tumors of childhood may also be considered in the differential diagnosis. So can myxoid cysts, for which there are three presentations in the nail apparatus: one related to the interphalangeal joint, a second having no connection with joints, and a third submatrical type. Patients with myxoid cysts tend to be female and middle-age or older. Squamous cell carcinomas, the most common of subungual malignancies, should also be considered, said Dr. Bettencourt, of the University of Nevada, Las Vegas.
When a subungual myxoma is identified, it is important to investigate the patient for signs of myxoma syndrome, the name of a group of diseases characterized by atrial and cutaneous myxomas, as well as lentigines.
The patient has not presented with any nail deformity or recurrence in the year since surgical recovery, she noted.
"Dermatologists need to and care for patients with nail problems, in place of having the patient be seen solely by a podiatrist, since nail diseases are part of our specialty," Dr. Bettencourt concluded.
She reported having no disclosures. SDEF and this news organization are owned by Elsevier.
LAS VEGAS – A 39-year-old male patient presented with a 5-year history of an isolated, nonpainful, nonfluctuant left thumbnail deformity. He had no other skin findings, and no other known medical problems. His thumbnail was distinctly curved, with what appeared to be a slight pinkish growth underneath the nail. The nail was avulsed, revealing a readily visible and protruding tumor from the nail bed, extending from underneath the nail matrix to the level of the distal phalanx, with only a small portion of the nail bed not affected.
Digital myxomas are rare neoplasms, and the subungual location is rarer still, according to Dr. Miriam S. Bettencourt.
There have been just four mentions of subungual myxoma in the medical literature since 1982, all of which were case reports. Surgical excision is usually curative, without the need for repeat surgery. One previous case report described a midlateral approach that spared the nail apparatus and demonstrated remodeling of the nail despite a void left under the nail matrix after excision of the tumor (J. Hand Surg. Am. 1998;23:178-80).
In this case, the patient’s histology revealed a large, circumscribed nonencapsulated nodular deposition of myxoid material, associated with a slight increase in spindle-shaped fibroblasts and thin intervening fibrocollagenous tissue underneath the nail bed epithelium. Scattered mast cells were visible within the lesion.
The tumor was sharply excised and the nail bed was roughly debrided using a small curette. A piece of tinfoil from a suture pack was fashioned into a pseudo nail shape and sutured beneath the patient’s cuticle in order to maintain the opening of the nail matrix during the excision, said Dr. Bettencourt, who presented the case at the Las Vegas Dermatology Seminar, sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis included subungual glomus tumor, which causes a bluish-red discoloration of the nail plate and, unlike myxoma, is painful and temperature-sensitive. Onychomatricoma can cause a nail curvature similar to that of myxoma. However, this rare benign tumor of the nail matrix causes a yellowish discoloration and splinter hemorrhages, which are not seen with myxoma.
Fibromas in the digits, including periungual fibroma (a marker of tuberous sclerosis), acquired digital fibrokeratoma, dermatofibroma, and recurrent digital fibrous tumors of childhood may also be considered in the differential diagnosis. So can myxoid cysts, for which there are three presentations in the nail apparatus: one related to the interphalangeal joint, a second having no connection with joints, and a third submatrical type. Patients with myxoid cysts tend to be female and middle-age or older. Squamous cell carcinomas, the most common of subungual malignancies, should also be considered, said Dr. Bettencourt, of the University of Nevada, Las Vegas.
When a subungual myxoma is identified, it is important to investigate the patient for signs of myxoma syndrome, the name of a group of diseases characterized by atrial and cutaneous myxomas, as well as lentigines.
The patient has not presented with any nail deformity or recurrence in the year since surgical recovery, she noted.
"Dermatologists need to and care for patients with nail problems, in place of having the patient be seen solely by a podiatrist, since nail diseases are part of our specialty," Dr. Bettencourt concluded.
She reported having no disclosures. SDEF and this news organization are owned by Elsevier.
LAS VEGAS – A 39-year-old male patient presented with a 5-year history of an isolated, nonpainful, nonfluctuant left thumbnail deformity. He had no other skin findings, and no other known medical problems. His thumbnail was distinctly curved, with what appeared to be a slight pinkish growth underneath the nail. The nail was avulsed, revealing a readily visible and protruding tumor from the nail bed, extending from underneath the nail matrix to the level of the distal phalanx, with only a small portion of the nail bed not affected.
Digital myxomas are rare neoplasms, and the subungual location is rarer still, according to Dr. Miriam S. Bettencourt.
There have been just four mentions of subungual myxoma in the medical literature since 1982, all of which were case reports. Surgical excision is usually curative, without the need for repeat surgery. One previous case report described a midlateral approach that spared the nail apparatus and demonstrated remodeling of the nail despite a void left under the nail matrix after excision of the tumor (J. Hand Surg. Am. 1998;23:178-80).
In this case, the patient’s histology revealed a large, circumscribed nonencapsulated nodular deposition of myxoid material, associated with a slight increase in spindle-shaped fibroblasts and thin intervening fibrocollagenous tissue underneath the nail bed epithelium. Scattered mast cells were visible within the lesion.
The tumor was sharply excised and the nail bed was roughly debrided using a small curette. A piece of tinfoil from a suture pack was fashioned into a pseudo nail shape and sutured beneath the patient’s cuticle in order to maintain the opening of the nail matrix during the excision, said Dr. Bettencourt, who presented the case at the Las Vegas Dermatology Seminar, sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis included subungual glomus tumor, which causes a bluish-red discoloration of the nail plate and, unlike myxoma, is painful and temperature-sensitive. Onychomatricoma can cause a nail curvature similar to that of myxoma. However, this rare benign tumor of the nail matrix causes a yellowish discoloration and splinter hemorrhages, which are not seen with myxoma.
Fibromas in the digits, including periungual fibroma (a marker of tuberous sclerosis), acquired digital fibrokeratoma, dermatofibroma, and recurrent digital fibrous tumors of childhood may also be considered in the differential diagnosis. So can myxoid cysts, for which there are three presentations in the nail apparatus: one related to the interphalangeal joint, a second having no connection with joints, and a third submatrical type. Patients with myxoid cysts tend to be female and middle-age or older. Squamous cell carcinomas, the most common of subungual malignancies, should also be considered, said Dr. Bettencourt, of the University of Nevada, Las Vegas.
When a subungual myxoma is identified, it is important to investigate the patient for signs of myxoma syndrome, the name of a group of diseases characterized by atrial and cutaneous myxomas, as well as lentigines.
The patient has not presented with any nail deformity or recurrence in the year since surgical recovery, she noted.
"Dermatologists need to and care for patients with nail problems, in place of having the patient be seen solely by a podiatrist, since nail diseases are part of our specialty," Dr. Bettencourt concluded.
She reported having no disclosures. SDEF and this news organization are owned by Elsevier.
CD4 Counts Predict Chemo Response in Laryngeal Cancer
PHOENIX – Pretreatment CD4 levels predicted response to induction chemotherapy among 97 patients with advanced laryngeal cancer, but not for 66 patients with advanced oropharyngeal cancer, according to a retrospective analysis of data from two clinical trials.
The two groups of head and neck cancer patients were enrolled in two identical prospective, phase II trials of induction chemotherapy and organ preservation, in which tumor response after one cycle of cisplatin and 5-fluorouracil was used to select those who would undergo surgery or definitive chemoradiation (J. Clin. Oncol. 2006;24:593-8 in laryngeal cancer; J. Clin. Oncol. 2008;26:3138-46 in oropharyngeal cancer).
Several lymphocyte subsets were measured before treatment via routine flow cytometry in peripheral blood in the laryngeal cancer patients, but only CD4 (helper cell) levels were significantly associated with chemotherapy response. Both absolute CD4 counts and CD4 percentages were higher among induction chemotherapy responders than nonresponders (P = .006 and P = .04, respectively).
Investigators also saw a trend for responders to have an increased percentage of CD3 cells (P = .13), decreased percentage of CD8 cells (P = .11), and higher CD4/CD8 ratios.
"Host immune parameters are important factors in treatment outcome, and may be useful in identifying subsets of patients with cancers that are responsive to organ-preserving therapy," said Dr. Gregory T. Wolf, who presented the data at a head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
"It is likely that immunobiology of head and neck cancers differ significantly by tumor site and predisposing factors," added Dr. Wolf, a professor in the department of otolaryngology at the University of Michigan, Ann Arbor.
The median length of follow-up in the two studies was 7.9 years for the patients with laryngeal cancer and 6.6 years for those with cancer of the oropharynx. Disease-free survival was 86% at 3 years and 82% at 5 years for laryngeal cancer, and 78% at 3 years and 76% at 4 years for oropharyngeal cancer. The proportions responding to induction chemotherapy were 75% in the laryngeal cancer trial and 82% in the oropharyngeal cancer study.
In an interview, Dr. Wolf said that having two identical treatment trials provided an opportunity to determine whether correlations of pretreatment CD4 levels with chemoresponse differed by tumor site. When the laryngeal and oropharyngeal cancer patient groups were combined, higher CD4 levels were still associated with response, but this was primarily because of the strong correlation among the larynx cancer patients.
Statistical regression testing determined that for patients with oropharyngeal cancer, CD8 cells were more closely associated with chemotherapy response, but the relationship was not as strong as the strong correlation of CD4 levels among laryngeal cancer patients.
There was a trend toward improved survival by both CD4 percentage (P = .36) and absolute CD4 count (P = .15) in the laryngeal cohort, but not in the oropharyngeal cohort. None of the other lymphocyte subsets predicted survival in either group, Dr. Wolf said.
Results for the oropharyngeal cancer patients were further stratified by human papillomavirus status, and were combined with the laryngeal cancer group. Both lower CD4/CD8 ratio and higher CD8 levels were consistent with better prognosis among patients who were HPV positive (P = .02 and P = .06, respectively).
"We combined the results to get the largest sample size and [to see] if the relationship was independent of tumor site, since the biology of these cancers is so different. It was remarkable that the prediction differed by tumor site, with CD4 cells being predictive for larynx and CD8 cells predictive for oropharynx," Dr. Wolf explained in an interview. This finding likely reflects the major biological differences between these cancers, and is why it was important to also include HPV status, he added.
Dr. Wolf is a consultant for IRX Therapeutics, Inc.
PHOENIX – Pretreatment CD4 levels predicted response to induction chemotherapy among 97 patients with advanced laryngeal cancer, but not for 66 patients with advanced oropharyngeal cancer, according to a retrospective analysis of data from two clinical trials.
The two groups of head and neck cancer patients were enrolled in two identical prospective, phase II trials of induction chemotherapy and organ preservation, in which tumor response after one cycle of cisplatin and 5-fluorouracil was used to select those who would undergo surgery or definitive chemoradiation (J. Clin. Oncol. 2006;24:593-8 in laryngeal cancer; J. Clin. Oncol. 2008;26:3138-46 in oropharyngeal cancer).
Several lymphocyte subsets were measured before treatment via routine flow cytometry in peripheral blood in the laryngeal cancer patients, but only CD4 (helper cell) levels were significantly associated with chemotherapy response. Both absolute CD4 counts and CD4 percentages were higher among induction chemotherapy responders than nonresponders (P = .006 and P = .04, respectively).
Investigators also saw a trend for responders to have an increased percentage of CD3 cells (P = .13), decreased percentage of CD8 cells (P = .11), and higher CD4/CD8 ratios.
"Host immune parameters are important factors in treatment outcome, and may be useful in identifying subsets of patients with cancers that are responsive to organ-preserving therapy," said Dr. Gregory T. Wolf, who presented the data at a head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
"It is likely that immunobiology of head and neck cancers differ significantly by tumor site and predisposing factors," added Dr. Wolf, a professor in the department of otolaryngology at the University of Michigan, Ann Arbor.
The median length of follow-up in the two studies was 7.9 years for the patients with laryngeal cancer and 6.6 years for those with cancer of the oropharynx. Disease-free survival was 86% at 3 years and 82% at 5 years for laryngeal cancer, and 78% at 3 years and 76% at 4 years for oropharyngeal cancer. The proportions responding to induction chemotherapy were 75% in the laryngeal cancer trial and 82% in the oropharyngeal cancer study.
In an interview, Dr. Wolf said that having two identical treatment trials provided an opportunity to determine whether correlations of pretreatment CD4 levels with chemoresponse differed by tumor site. When the laryngeal and oropharyngeal cancer patient groups were combined, higher CD4 levels were still associated with response, but this was primarily because of the strong correlation among the larynx cancer patients.
Statistical regression testing determined that for patients with oropharyngeal cancer, CD8 cells were more closely associated with chemotherapy response, but the relationship was not as strong as the strong correlation of CD4 levels among laryngeal cancer patients.
There was a trend toward improved survival by both CD4 percentage (P = .36) and absolute CD4 count (P = .15) in the laryngeal cohort, but not in the oropharyngeal cohort. None of the other lymphocyte subsets predicted survival in either group, Dr. Wolf said.
Results for the oropharyngeal cancer patients were further stratified by human papillomavirus status, and were combined with the laryngeal cancer group. Both lower CD4/CD8 ratio and higher CD8 levels were consistent with better prognosis among patients who were HPV positive (P = .02 and P = .06, respectively).
"We combined the results to get the largest sample size and [to see] if the relationship was independent of tumor site, since the biology of these cancers is so different. It was remarkable that the prediction differed by tumor site, with CD4 cells being predictive for larynx and CD8 cells predictive for oropharynx," Dr. Wolf explained in an interview. This finding likely reflects the major biological differences between these cancers, and is why it was important to also include HPV status, he added.
Dr. Wolf is a consultant for IRX Therapeutics, Inc.
PHOENIX – Pretreatment CD4 levels predicted response to induction chemotherapy among 97 patients with advanced laryngeal cancer, but not for 66 patients with advanced oropharyngeal cancer, according to a retrospective analysis of data from two clinical trials.
The two groups of head and neck cancer patients were enrolled in two identical prospective, phase II trials of induction chemotherapy and organ preservation, in which tumor response after one cycle of cisplatin and 5-fluorouracil was used to select those who would undergo surgery or definitive chemoradiation (J. Clin. Oncol. 2006;24:593-8 in laryngeal cancer; J. Clin. Oncol. 2008;26:3138-46 in oropharyngeal cancer).
Several lymphocyte subsets were measured before treatment via routine flow cytometry in peripheral blood in the laryngeal cancer patients, but only CD4 (helper cell) levels were significantly associated with chemotherapy response. Both absolute CD4 counts and CD4 percentages were higher among induction chemotherapy responders than nonresponders (P = .006 and P = .04, respectively).
Investigators also saw a trend for responders to have an increased percentage of CD3 cells (P = .13), decreased percentage of CD8 cells (P = .11), and higher CD4/CD8 ratios.
"Host immune parameters are important factors in treatment outcome, and may be useful in identifying subsets of patients with cancers that are responsive to organ-preserving therapy," said Dr. Gregory T. Wolf, who presented the data at a head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
"It is likely that immunobiology of head and neck cancers differ significantly by tumor site and predisposing factors," added Dr. Wolf, a professor in the department of otolaryngology at the University of Michigan, Ann Arbor.
The median length of follow-up in the two studies was 7.9 years for the patients with laryngeal cancer and 6.6 years for those with cancer of the oropharynx. Disease-free survival was 86% at 3 years and 82% at 5 years for laryngeal cancer, and 78% at 3 years and 76% at 4 years for oropharyngeal cancer. The proportions responding to induction chemotherapy were 75% in the laryngeal cancer trial and 82% in the oropharyngeal cancer study.
In an interview, Dr. Wolf said that having two identical treatment trials provided an opportunity to determine whether correlations of pretreatment CD4 levels with chemoresponse differed by tumor site. When the laryngeal and oropharyngeal cancer patient groups were combined, higher CD4 levels were still associated with response, but this was primarily because of the strong correlation among the larynx cancer patients.
Statistical regression testing determined that for patients with oropharyngeal cancer, CD8 cells were more closely associated with chemotherapy response, but the relationship was not as strong as the strong correlation of CD4 levels among laryngeal cancer patients.
There was a trend toward improved survival by both CD4 percentage (P = .36) and absolute CD4 count (P = .15) in the laryngeal cohort, but not in the oropharyngeal cohort. None of the other lymphocyte subsets predicted survival in either group, Dr. Wolf said.
Results for the oropharyngeal cancer patients were further stratified by human papillomavirus status, and were combined with the laryngeal cancer group. Both lower CD4/CD8 ratio and higher CD8 levels were consistent with better prognosis among patients who were HPV positive (P = .02 and P = .06, respectively).
"We combined the results to get the largest sample size and [to see] if the relationship was independent of tumor site, since the biology of these cancers is so different. It was remarkable that the prediction differed by tumor site, with CD4 cells being predictive for larynx and CD8 cells predictive for oropharynx," Dr. Wolf explained in an interview. This finding likely reflects the major biological differences between these cancers, and is why it was important to also include HPV status, he added.
Dr. Wolf is a consultant for IRX Therapeutics, Inc.
FROM A HEAD AND NECK CANCER SYMPOSIUM SPONSORED BY THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Major Finding: Among laryngeal cancer patients, both absolute and percent CD4 counts were significantly higher among responders to induction chemotherapy than in nonresponders (P = .006 and P = .04, respectively).
Data Source: The data come from a retrospective analysis of head and neck cancer patients who were enrolled in two identical prospective, phase II trials of induction chemotherapy and organ preservation.
Disclosures: Dr. Wolf is a consultant for IRX Therapeutics.
Infrared Imaging Eyes Mucositis After Head & Neck Radiation
PHOENIX – Thermal imaging was able to detect small and early changes in the temperature of mucosal surfaces – a possible predictor of the development of mucositis – among 34 patients who were treated with chemoradiotherapy for locally advanced squamous cell carcinoma of the head and neck.
"Detection of these early changes using sensitive thermal imaging technology would allow identification of patients who will require more intensive supportive care," said Dr. Ezra Cohen, who presented the pilot study at the head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
Radiotherapy-associated mucositis manifests initially as erythematous areas in the treatment field, which are accompanied by an intense inflammatory response histologically. Thus, Dr. Cohen and his associates had hypothesized that patients destined to display severe mucocutaneous toxicity would demonstrate greater alterations in thermal intensity early in therapy, compared with identically treated counterparts who do not subsequently develop the toxicity.
The researchers further hypothesized that they could measure those changes with infrared thermal imaging, a noninvasive technique that allows visualization and quantification of changes in skin or mucosal surface temperature.
The 34 patients (28 male, mean age 58 years) in the pilot study were treated with identical chemoradiotherapy regimens of 5-fluorouracil and hydroxyurea with a median radiation dose of 74 Gy for cancers of the oral cavity or oropharynx.
Using a portable device developed at Argonne National Laboratory in Illinois, the investigators conducted noninvasive baseline and weekly thermal imaging. The device detects infrared light naturally emitted from the skin or mucosal surface and generates an electrical signal, which is amplified and converted into digital data flow that is visualized in color on a monitor.
Grade 3 mucositis based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events v3.0 was observed in 53% of the patients, and dermatitis in 21%. All patients displayed an increase in temperature within the radiation field.
Investigators charted a statistically significant positive association between an early rise in temperature in oral mucous membranes when compared with a reference area and mucositis grade (P = .03). For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis grade, reported Dr. Cohen, codirector of the head and neck cancer program at the University of Chicago.
Mucositis and its clinical sequelae are consistently reported as the most clinically significant acute toxicity in the treatment of locally advanced squamous cell carcinoma of the head and neck with chemoradiotherapy, according to Dr. Cohen, who also chaired the meeting. Patient to patient variability in mucositis is related to radiotherapy dosing, fractionation, and volumes, but there also appear to be individual differences in "normal tissue tolerance," even among patients on the same treatment regimen, he said.
"Larger studies with greater dynamic ranges in mucositis scoring are warranted to evaluate whether this tool can help predict which patients would be in need of early intervention to prevent acute complications," he said.
The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
PHOENIX – Thermal imaging was able to detect small and early changes in the temperature of mucosal surfaces – a possible predictor of the development of mucositis – among 34 patients who were treated with chemoradiotherapy for locally advanced squamous cell carcinoma of the head and neck.
"Detection of these early changes using sensitive thermal imaging technology would allow identification of patients who will require more intensive supportive care," said Dr. Ezra Cohen, who presented the pilot study at the head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
Radiotherapy-associated mucositis manifests initially as erythematous areas in the treatment field, which are accompanied by an intense inflammatory response histologically. Thus, Dr. Cohen and his associates had hypothesized that patients destined to display severe mucocutaneous toxicity would demonstrate greater alterations in thermal intensity early in therapy, compared with identically treated counterparts who do not subsequently develop the toxicity.
The researchers further hypothesized that they could measure those changes with infrared thermal imaging, a noninvasive technique that allows visualization and quantification of changes in skin or mucosal surface temperature.
The 34 patients (28 male, mean age 58 years) in the pilot study were treated with identical chemoradiotherapy regimens of 5-fluorouracil and hydroxyurea with a median radiation dose of 74 Gy for cancers of the oral cavity or oropharynx.
Using a portable device developed at Argonne National Laboratory in Illinois, the investigators conducted noninvasive baseline and weekly thermal imaging. The device detects infrared light naturally emitted from the skin or mucosal surface and generates an electrical signal, which is amplified and converted into digital data flow that is visualized in color on a monitor.
Grade 3 mucositis based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events v3.0 was observed in 53% of the patients, and dermatitis in 21%. All patients displayed an increase in temperature within the radiation field.
Investigators charted a statistically significant positive association between an early rise in temperature in oral mucous membranes when compared with a reference area and mucositis grade (P = .03). For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis grade, reported Dr. Cohen, codirector of the head and neck cancer program at the University of Chicago.
Mucositis and its clinical sequelae are consistently reported as the most clinically significant acute toxicity in the treatment of locally advanced squamous cell carcinoma of the head and neck with chemoradiotherapy, according to Dr. Cohen, who also chaired the meeting. Patient to patient variability in mucositis is related to radiotherapy dosing, fractionation, and volumes, but there also appear to be individual differences in "normal tissue tolerance," even among patients on the same treatment regimen, he said.
"Larger studies with greater dynamic ranges in mucositis scoring are warranted to evaluate whether this tool can help predict which patients would be in need of early intervention to prevent acute complications," he said.
The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
PHOENIX – Thermal imaging was able to detect small and early changes in the temperature of mucosal surfaces – a possible predictor of the development of mucositis – among 34 patients who were treated with chemoradiotherapy for locally advanced squamous cell carcinoma of the head and neck.
"Detection of these early changes using sensitive thermal imaging technology would allow identification of patients who will require more intensive supportive care," said Dr. Ezra Cohen, who presented the pilot study at the head and neck cancer symposium sponsored by the American Society for Radiation Oncology.
Radiotherapy-associated mucositis manifests initially as erythematous areas in the treatment field, which are accompanied by an intense inflammatory response histologically. Thus, Dr. Cohen and his associates had hypothesized that patients destined to display severe mucocutaneous toxicity would demonstrate greater alterations in thermal intensity early in therapy, compared with identically treated counterparts who do not subsequently develop the toxicity.
The researchers further hypothesized that they could measure those changes with infrared thermal imaging, a noninvasive technique that allows visualization and quantification of changes in skin or mucosal surface temperature.
The 34 patients (28 male, mean age 58 years) in the pilot study were treated with identical chemoradiotherapy regimens of 5-fluorouracil and hydroxyurea with a median radiation dose of 74 Gy for cancers of the oral cavity or oropharynx.
Using a portable device developed at Argonne National Laboratory in Illinois, the investigators conducted noninvasive baseline and weekly thermal imaging. The device detects infrared light naturally emitted from the skin or mucosal surface and generates an electrical signal, which is amplified and converted into digital data flow that is visualized in color on a monitor.
Grade 3 mucositis based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events v3.0 was observed in 53% of the patients, and dermatitis in 21%. All patients displayed an increase in temperature within the radiation field.
Investigators charted a statistically significant positive association between an early rise in temperature in oral mucous membranes when compared with a reference area and mucositis grade (P = .03). For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis grade, reported Dr. Cohen, codirector of the head and neck cancer program at the University of Chicago.
Mucositis and its clinical sequelae are consistently reported as the most clinically significant acute toxicity in the treatment of locally advanced squamous cell carcinoma of the head and neck with chemoradiotherapy, according to Dr. Cohen, who also chaired the meeting. Patient to patient variability in mucositis is related to radiotherapy dosing, fractionation, and volumes, but there also appear to be individual differences in "normal tissue tolerance," even among patients on the same treatment regimen, he said.
"Larger studies with greater dynamic ranges in mucositis scoring are warranted to evaluate whether this tool can help predict which patients would be in need of early intervention to prevent acute complications," he said.
The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
FROM A HEAD AND NECK CANCER SYMPOSIUM SPONSORED BY THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Major Finding: For every 1 C increase in temperature, compared with the reference (temperature near the medial angle of one eye), there was a 0.157 increase in average subsequent mucositis.
Data Source: Data come from a pilot study of functional infrared imaging in 34 patients with locally advanced head and neck cancer who were treated with chemoradiotherapy.
Disclosures: The study was funded by the National Institutes of Health and the University of Chicago Comprehensive Cancer Center. Dr. Cohen and his associates said they had no relevant financial disclosures.
Pertussis Immunization Recommended for Adults 65 and Older
ATLANTA – The tetanus-diphtheria-acellular pertussis vaccine should be given to adults aged 65 years and older, according to recommendations from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The recommendation, backed by a 14-1 vote among ACIP members, is expected to go into effect when the CDC publishes the guidance, along with other guidance related to use of the adolescent/adult formulation of the tetanus-diphtheria-acellular pertussis vaccine (Tdap).
Of the two currently licensed Tdap vaccines, only GlaxoSmithKline’s Boostrix is approved for adults aged 65 and older. Efficacy data for the other one, Sanofi-Pasteur’s Adacel, had been submitted to the Food and Drug Administration but did not meet immunogenicity end points, the FDA’s representative, Dr. Wellington Sun, told the committee.
The ACIP recommendation will express a preference for use of Boostrix in the 65+ age group but is expected to state that Adacel is acceptable and preferable to delay of vaccination if Boostrix is not available. There were differences of opinion among committee members about whether to express a preference or not, and the exact language for the recommendation was still to be worked out at the time of the vote.
Current recommendations call for a single dose of Tdap for adults aged 19-64 years. Previously, the vaccine had only been recommended for adults 65 and older who were in close contact with an infant less than 12 months of age. With the current vote, ACIP also recommended elimination of the "decennial" interval that had previously been applied to the tetanus-diphtheria vaccine, so that a single dose of Tdap can be given to any adult who has not previously received it, regardless of interval since the last Td, said Dr. Jennifer L. Liang who explained the ACIP Pertussis Vaccines Working Groups’ deliberations.
Liaisons from the American College of Physicians (ACP) and the American Academy of Family Physicians (AAFP) said they supported the vote. "I think the important thing that we did today was in essence make a universal use recommendation for Tdap in adults. Before, it was full of provisos that clinicians couldn’t remember, sort of like the flu [vaccine] story. We’ve made it easy for clinicians by saying every adult deserves a dose of Tdap," ACP liaison Gregory A. Poland said in an interview.
However, "There are unknowns yet. We don’t know the duration of efficacy or the exact level of efficacy, particularly in older adults. Those are data needs that we have," noted Dr. Poland, the Mary Lowell Leary Professor of Medicine and Infectious Diseases at the Mayo Clinic, Rochester, Minn.
According to AAFP liaison Dr. Jamie Loehr, owner of Cayuga Family Medicine, Ithaca, N.Y., "The [AAFP] will review this, and most likely support it as it has in the past. On a practical level, it will be much easier for family physicians to have a blanket immunization recommendation rather than having to pick and chose."
Surveillance data suggest that the incidence of pertussis overall among adults older than 65 in the U.S. ranges from 2.5/100,000 in those aged 65-69 years to 0.8/100,000 in those aged 85 years and older. However, reported rates for adults are believed to be greatly underestimated since adults often don’t exhibit the typical paroxysms, inspiratory whoop, and post-tussive vomiting seen in infants with pertussis. Indeed, data from Australian surveillance and from published studies on varying age groups, some including adults beyond age 65 years, suggest that the incidence actually ranges from 66 to 507 cases/100,000 population, the CDC’s Dr. Anna Acosta told the ACIP.
Girishanthy Krishnarajah of GlaxoSmithKline presented a cost-effectiveness analysis of a one-time Boostrix vaccination among a cohort of U.S. residents aged 65 and older, in which the cost per case averted ranged from $416 to $10,413, depending on the pertussis incidence level. The cost per quality-adjusted life year (QALY) ranged from $13,260 to $332,218, also depending on incidence. When the pertussis incidence in people aged 65 years and older exceeded 110/100,000, the incremental cost-effectiveness ratio falls below $50,000/QALY, the generally accepted cost-effectiveness threshold, she said.
A CDC cost-effectiveness analysis produced similar results, according to Dr. Acosta.
Data suggest that safety and tolerability of Tdap in older adults is similar to that of the Td vaccine. While there are no efficacy data available specifically in the 65+ age group, data suggest that "older adults mount an immune response, likely to provide protection," the CDC’s Dr. Liang said.
Several ACIP members expressed concern that efficacy data were not available, but voted in favor of the recommendation regardless. Dr. Jeffrey Duchin was the sole committee member who cast a vote against the recommendation. "I would have liked to have seen more data on efficacy in the older population. It was assumed in the models. I would like to see more studies," Dr. Duchin, a professor of medicine at the University of Washington in Seattle, said in an interview.
As CDC employees, Dr. Acosta and Dr. Liang had no disclosures. Dr. Loehr and Dr. Duchin stated that they have no disclosures. Dr. Poland consults with companies on novel vaccines, but currently has no relationships with either GlaxoSmithKline or Sanofi-Pasteur.
ATLANTA – The tetanus-diphtheria-acellular pertussis vaccine should be given to adults aged 65 years and older, according to recommendations from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The recommendation, backed by a 14-1 vote among ACIP members, is expected to go into effect when the CDC publishes the guidance, along with other guidance related to use of the adolescent/adult formulation of the tetanus-diphtheria-acellular pertussis vaccine (Tdap).
Of the two currently licensed Tdap vaccines, only GlaxoSmithKline’s Boostrix is approved for adults aged 65 and older. Efficacy data for the other one, Sanofi-Pasteur’s Adacel, had been submitted to the Food and Drug Administration but did not meet immunogenicity end points, the FDA’s representative, Dr. Wellington Sun, told the committee.
The ACIP recommendation will express a preference for use of Boostrix in the 65+ age group but is expected to state that Adacel is acceptable and preferable to delay of vaccination if Boostrix is not available. There were differences of opinion among committee members about whether to express a preference or not, and the exact language for the recommendation was still to be worked out at the time of the vote.
Current recommendations call for a single dose of Tdap for adults aged 19-64 years. Previously, the vaccine had only been recommended for adults 65 and older who were in close contact with an infant less than 12 months of age. With the current vote, ACIP also recommended elimination of the "decennial" interval that had previously been applied to the tetanus-diphtheria vaccine, so that a single dose of Tdap can be given to any adult who has not previously received it, regardless of interval since the last Td, said Dr. Jennifer L. Liang who explained the ACIP Pertussis Vaccines Working Groups’ deliberations.
Liaisons from the American College of Physicians (ACP) and the American Academy of Family Physicians (AAFP) said they supported the vote. "I think the important thing that we did today was in essence make a universal use recommendation for Tdap in adults. Before, it was full of provisos that clinicians couldn’t remember, sort of like the flu [vaccine] story. We’ve made it easy for clinicians by saying every adult deserves a dose of Tdap," ACP liaison Gregory A. Poland said in an interview.
However, "There are unknowns yet. We don’t know the duration of efficacy or the exact level of efficacy, particularly in older adults. Those are data needs that we have," noted Dr. Poland, the Mary Lowell Leary Professor of Medicine and Infectious Diseases at the Mayo Clinic, Rochester, Minn.
According to AAFP liaison Dr. Jamie Loehr, owner of Cayuga Family Medicine, Ithaca, N.Y., "The [AAFP] will review this, and most likely support it as it has in the past. On a practical level, it will be much easier for family physicians to have a blanket immunization recommendation rather than having to pick and chose."
Surveillance data suggest that the incidence of pertussis overall among adults older than 65 in the U.S. ranges from 2.5/100,000 in those aged 65-69 years to 0.8/100,000 in those aged 85 years and older. However, reported rates for adults are believed to be greatly underestimated since adults often don’t exhibit the typical paroxysms, inspiratory whoop, and post-tussive vomiting seen in infants with pertussis. Indeed, data from Australian surveillance and from published studies on varying age groups, some including adults beyond age 65 years, suggest that the incidence actually ranges from 66 to 507 cases/100,000 population, the CDC’s Dr. Anna Acosta told the ACIP.
Girishanthy Krishnarajah of GlaxoSmithKline presented a cost-effectiveness analysis of a one-time Boostrix vaccination among a cohort of U.S. residents aged 65 and older, in which the cost per case averted ranged from $416 to $10,413, depending on the pertussis incidence level. The cost per quality-adjusted life year (QALY) ranged from $13,260 to $332,218, also depending on incidence. When the pertussis incidence in people aged 65 years and older exceeded 110/100,000, the incremental cost-effectiveness ratio falls below $50,000/QALY, the generally accepted cost-effectiveness threshold, she said.
A CDC cost-effectiveness analysis produced similar results, according to Dr. Acosta.
Data suggest that safety and tolerability of Tdap in older adults is similar to that of the Td vaccine. While there are no efficacy data available specifically in the 65+ age group, data suggest that "older adults mount an immune response, likely to provide protection," the CDC’s Dr. Liang said.
Several ACIP members expressed concern that efficacy data were not available, but voted in favor of the recommendation regardless. Dr. Jeffrey Duchin was the sole committee member who cast a vote against the recommendation. "I would have liked to have seen more data on efficacy in the older population. It was assumed in the models. I would like to see more studies," Dr. Duchin, a professor of medicine at the University of Washington in Seattle, said in an interview.
As CDC employees, Dr. Acosta and Dr. Liang had no disclosures. Dr. Loehr and Dr. Duchin stated that they have no disclosures. Dr. Poland consults with companies on novel vaccines, but currently has no relationships with either GlaxoSmithKline or Sanofi-Pasteur.
ATLANTA – The tetanus-diphtheria-acellular pertussis vaccine should be given to adults aged 65 years and older, according to recommendations from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The recommendation, backed by a 14-1 vote among ACIP members, is expected to go into effect when the CDC publishes the guidance, along with other guidance related to use of the adolescent/adult formulation of the tetanus-diphtheria-acellular pertussis vaccine (Tdap).
Of the two currently licensed Tdap vaccines, only GlaxoSmithKline’s Boostrix is approved for adults aged 65 and older. Efficacy data for the other one, Sanofi-Pasteur’s Adacel, had been submitted to the Food and Drug Administration but did not meet immunogenicity end points, the FDA’s representative, Dr. Wellington Sun, told the committee.
The ACIP recommendation will express a preference for use of Boostrix in the 65+ age group but is expected to state that Adacel is acceptable and preferable to delay of vaccination if Boostrix is not available. There were differences of opinion among committee members about whether to express a preference or not, and the exact language for the recommendation was still to be worked out at the time of the vote.
Current recommendations call for a single dose of Tdap for adults aged 19-64 years. Previously, the vaccine had only been recommended for adults 65 and older who were in close contact with an infant less than 12 months of age. With the current vote, ACIP also recommended elimination of the "decennial" interval that had previously been applied to the tetanus-diphtheria vaccine, so that a single dose of Tdap can be given to any adult who has not previously received it, regardless of interval since the last Td, said Dr. Jennifer L. Liang who explained the ACIP Pertussis Vaccines Working Groups’ deliberations.
Liaisons from the American College of Physicians (ACP) and the American Academy of Family Physicians (AAFP) said they supported the vote. "I think the important thing that we did today was in essence make a universal use recommendation for Tdap in adults. Before, it was full of provisos that clinicians couldn’t remember, sort of like the flu [vaccine] story. We’ve made it easy for clinicians by saying every adult deserves a dose of Tdap," ACP liaison Gregory A. Poland said in an interview.
However, "There are unknowns yet. We don’t know the duration of efficacy or the exact level of efficacy, particularly in older adults. Those are data needs that we have," noted Dr. Poland, the Mary Lowell Leary Professor of Medicine and Infectious Diseases at the Mayo Clinic, Rochester, Minn.
According to AAFP liaison Dr. Jamie Loehr, owner of Cayuga Family Medicine, Ithaca, N.Y., "The [AAFP] will review this, and most likely support it as it has in the past. On a practical level, it will be much easier for family physicians to have a blanket immunization recommendation rather than having to pick and chose."
Surveillance data suggest that the incidence of pertussis overall among adults older than 65 in the U.S. ranges from 2.5/100,000 in those aged 65-69 years to 0.8/100,000 in those aged 85 years and older. However, reported rates for adults are believed to be greatly underestimated since adults often don’t exhibit the typical paroxysms, inspiratory whoop, and post-tussive vomiting seen in infants with pertussis. Indeed, data from Australian surveillance and from published studies on varying age groups, some including adults beyond age 65 years, suggest that the incidence actually ranges from 66 to 507 cases/100,000 population, the CDC’s Dr. Anna Acosta told the ACIP.
Girishanthy Krishnarajah of GlaxoSmithKline presented a cost-effectiveness analysis of a one-time Boostrix vaccination among a cohort of U.S. residents aged 65 and older, in which the cost per case averted ranged from $416 to $10,413, depending on the pertussis incidence level. The cost per quality-adjusted life year (QALY) ranged from $13,260 to $332,218, also depending on incidence. When the pertussis incidence in people aged 65 years and older exceeded 110/100,000, the incremental cost-effectiveness ratio falls below $50,000/QALY, the generally accepted cost-effectiveness threshold, she said.
A CDC cost-effectiveness analysis produced similar results, according to Dr. Acosta.
Data suggest that safety and tolerability of Tdap in older adults is similar to that of the Td vaccine. While there are no efficacy data available specifically in the 65+ age group, data suggest that "older adults mount an immune response, likely to provide protection," the CDC’s Dr. Liang said.
Several ACIP members expressed concern that efficacy data were not available, but voted in favor of the recommendation regardless. Dr. Jeffrey Duchin was the sole committee member who cast a vote against the recommendation. "I would have liked to have seen more data on efficacy in the older population. It was assumed in the models. I would like to see more studies," Dr. Duchin, a professor of medicine at the University of Washington in Seattle, said in an interview.
As CDC employees, Dr. Acosta and Dr. Liang had no disclosures. Dr. Loehr and Dr. Duchin stated that they have no disclosures. Dr. Poland consults with companies on novel vaccines, but currently has no relationships with either GlaxoSmithKline or Sanofi-Pasteur.
Microwave Technology Outlasts Botulinum Toxin for Hyperhidrosis
Microwave technology is a new hyperhidrosis treatment that offers longer-term efficacy than botulinum toxin.
"With microwave thermolysis, we now have a reliable and safe method with long-term efficacy to treat our patients who suffer from hyperhidrosis," said Dr. Michael S. Kaminer, at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
A hyperhidrosis diagnosis is made in patients who have visible, excessive sweating for at least 6 months without apparent cause, and with at least two of the following characteristics (J. Am. Acad. Dermatol. 2004;51:274-86):
• Impairs daily activities.
• Frequency of at least one episode per week.
• Bilateral and symmetric.
• Age of onset before 25 years.
• Positive family history.
• Cessation during sleep.
In the United states, approximately 7.6 million people, or 4% of all U.S. adults, are estimated to have axillary hyperhidrosis with a level on the Hyperhidrosis Disease Severity Scale (HDSS) of either 3 ("My sweating is barely tolerable and frequently interferes with my daily activities.") or 4 ("My sweating is intolerable and always interferes with my daily activities."), according to Dr. Kaminer, managing partner, SkinCare Physicians, Chestnut Hill, Mass. Another 32.5 million are "sweat bothered," with an HDSS level of 2 ("My sweating is tolerable but sometimes interferes with my daily activities.") or 1 ("My sweating is never noticeable and never interferes with my daily activities.").
Axillary hyperhidrosis causes considerable distress among patients. In one survey of the Dermatology Life Quality Index, the condition scored just below atopic dermatitis, close to psoriasis, and above acne vulgaris in terms of quality of life impairment, with a mean baseline score of 10.45, compared with 11.20 for atopic dermatitis, 10.53 for psoriasis, and 7.45 for acne vulgaris (Br. J. Dermatol. 2008;159:997-1035).
Current treatment options include various topical over-the-counter products, iontophoresis, sympathectomy, botulinum toxin, ultrasound (in early trials), and now microwave thermolysis.
The proprietary microwave thermolysis system, MiraDry (Miramar Labs), focuses energy delivery to the dermal-fat interface. Microwave energy is absorbed by electric dipoles. The ion content of sweat is a perfect target for this, said Dr. Kaminer, who is also with the departments of dermatology at Yale University, New Haven, Conn.; Dartmouth College, Hanover, N.H.; and Brown University, Providence, R.I.
Energy becomes concentrated along this interface and creates a focal energy zone. Continuous hydroceramic cooling prevents thermal conduction of heat superficially, and creates a heat zone at the level of sweat glands resulting in thermolysis. The procedure involves injectable local anesthesia. Treatment time is about 30 minutes, with rapid recovery.
Long-term efficacy of the device was demonstrated in the DRIUP study (Dermatologic Reduction in Underarm Perspiration), in which 120 patients with HDSS scores of 3 or 4 were randomized in a 2:1 ratio to one to three active treatments (using a prior investigational version of the microwave device) or sham treatment at seven U.S. sites. Efficacy, defined as patients reporting a reduction to an HDSS score of 1 or 2, was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant.
There were 45 treatment-related adverse events, with 28% of the treatment group and 13% of the sham group patients experiencing at least one adverse event. The most common of these was an area of transient altered sensation in the arm, occurring in 10% of patients in the active treatment group. All but one adverse event resolved over time; the patient withdrew from the study, reporting altered sweating on the face. In all, 71% of the events were rated as mild in severity and didn’t affect patients’ daily activities (Dermatol Surg. 2012;38:185-91). Swelling was common, self-limited, and proportional to the energy delivered, Dr. Kaminer noted.
When compared with botulinum toxin, both approaches are effective, noninvasive (injections only), and involve minimal to no downtime. However, the duration of effect is 6.7 months for botulinum toxin, compared with stable efficacy beginning at 3 months and lasting through 12 months follow-up for microwave thermolysis, he said.
Overall, the advantages of microwave thermolysis over other treatments are its long-term efficacy (greater than 12 months, and likely longer), and its efficacy in more than 90% of patients with the latest-generation device. Disadvantages include the need for two procedures and the less than 100% responder rate. Botulinum toxin, in contrast, is easier to perform, and nearly 100% effective. However, it has the downside of the need for repeat treatments approximately every 6 months, and its cost, along with problematic insurance coverage.
Dr. Kaminer said that his practice participated in a registry-type study in the summer and fall of 2011 and was one of the first clinics in the country to purchase a MiraDry System, in December 2011. "Our staff has found the procedure easy to incorporate into the flow of our practice, and patients have found it to be rather easy to have done. We have seen some modest swelling for up to 1 week after treatment, but that is expected. So far, our experience has been pretty much what we would have expected, and if anything, a little better."
He added that, "Interest in offering this new treatment option to patients with excessive underarm sweating has been extremely high. Between clinics in Japan and the U.S., there have been over 1,500 patients treated."
In all, safety results have been consistent with prior experiences. "As with any procedure that we do, it is important to clearly communicate with the patients what to expect from the procedure and the recovery. Our feedback allowed the company to fine-tune their training program so that clinics that purchase the system have tools they can use to make the experience a very positive one for their patients."
Miramar is in the early stages of commercialization for the MiraDry system. It is being placed in a controlled strategy and is expected to reach "a significant number of practices across the U.S. by the middle of this year," he said.
Dr. Kaminer is chairman of the Miramar Labs scientific advisory board. His institution served as a research study site for the phase II study. SDEF and this news organization are owned by Elsevier.
Microwave technology is a new hyperhidrosis treatment that offers longer-term efficacy than botulinum toxin.
"With microwave thermolysis, we now have a reliable and safe method with long-term efficacy to treat our patients who suffer from hyperhidrosis," said Dr. Michael S. Kaminer, at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
A hyperhidrosis diagnosis is made in patients who have visible, excessive sweating for at least 6 months without apparent cause, and with at least two of the following characteristics (J. Am. Acad. Dermatol. 2004;51:274-86):
• Impairs daily activities.
• Frequency of at least one episode per week.
• Bilateral and symmetric.
• Age of onset before 25 years.
• Positive family history.
• Cessation during sleep.
In the United states, approximately 7.6 million people, or 4% of all U.S. adults, are estimated to have axillary hyperhidrosis with a level on the Hyperhidrosis Disease Severity Scale (HDSS) of either 3 ("My sweating is barely tolerable and frequently interferes with my daily activities.") or 4 ("My sweating is intolerable and always interferes with my daily activities."), according to Dr. Kaminer, managing partner, SkinCare Physicians, Chestnut Hill, Mass. Another 32.5 million are "sweat bothered," with an HDSS level of 2 ("My sweating is tolerable but sometimes interferes with my daily activities.") or 1 ("My sweating is never noticeable and never interferes with my daily activities.").
Axillary hyperhidrosis causes considerable distress among patients. In one survey of the Dermatology Life Quality Index, the condition scored just below atopic dermatitis, close to psoriasis, and above acne vulgaris in terms of quality of life impairment, with a mean baseline score of 10.45, compared with 11.20 for atopic dermatitis, 10.53 for psoriasis, and 7.45 for acne vulgaris (Br. J. Dermatol. 2008;159:997-1035).
Current treatment options include various topical over-the-counter products, iontophoresis, sympathectomy, botulinum toxin, ultrasound (in early trials), and now microwave thermolysis.
The proprietary microwave thermolysis system, MiraDry (Miramar Labs), focuses energy delivery to the dermal-fat interface. Microwave energy is absorbed by electric dipoles. The ion content of sweat is a perfect target for this, said Dr. Kaminer, who is also with the departments of dermatology at Yale University, New Haven, Conn.; Dartmouth College, Hanover, N.H.; and Brown University, Providence, R.I.
Energy becomes concentrated along this interface and creates a focal energy zone. Continuous hydroceramic cooling prevents thermal conduction of heat superficially, and creates a heat zone at the level of sweat glands resulting in thermolysis. The procedure involves injectable local anesthesia. Treatment time is about 30 minutes, with rapid recovery.
Long-term efficacy of the device was demonstrated in the DRIUP study (Dermatologic Reduction in Underarm Perspiration), in which 120 patients with HDSS scores of 3 or 4 were randomized in a 2:1 ratio to one to three active treatments (using a prior investigational version of the microwave device) or sham treatment at seven U.S. sites. Efficacy, defined as patients reporting a reduction to an HDSS score of 1 or 2, was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant.
There were 45 treatment-related adverse events, with 28% of the treatment group and 13% of the sham group patients experiencing at least one adverse event. The most common of these was an area of transient altered sensation in the arm, occurring in 10% of patients in the active treatment group. All but one adverse event resolved over time; the patient withdrew from the study, reporting altered sweating on the face. In all, 71% of the events were rated as mild in severity and didn’t affect patients’ daily activities (Dermatol Surg. 2012;38:185-91). Swelling was common, self-limited, and proportional to the energy delivered, Dr. Kaminer noted.
When compared with botulinum toxin, both approaches are effective, noninvasive (injections only), and involve minimal to no downtime. However, the duration of effect is 6.7 months for botulinum toxin, compared with stable efficacy beginning at 3 months and lasting through 12 months follow-up for microwave thermolysis, he said.
Overall, the advantages of microwave thermolysis over other treatments are its long-term efficacy (greater than 12 months, and likely longer), and its efficacy in more than 90% of patients with the latest-generation device. Disadvantages include the need for two procedures and the less than 100% responder rate. Botulinum toxin, in contrast, is easier to perform, and nearly 100% effective. However, it has the downside of the need for repeat treatments approximately every 6 months, and its cost, along with problematic insurance coverage.
Dr. Kaminer said that his practice participated in a registry-type study in the summer and fall of 2011 and was one of the first clinics in the country to purchase a MiraDry System, in December 2011. "Our staff has found the procedure easy to incorporate into the flow of our practice, and patients have found it to be rather easy to have done. We have seen some modest swelling for up to 1 week after treatment, but that is expected. So far, our experience has been pretty much what we would have expected, and if anything, a little better."
He added that, "Interest in offering this new treatment option to patients with excessive underarm sweating has been extremely high. Between clinics in Japan and the U.S., there have been over 1,500 patients treated."
In all, safety results have been consistent with prior experiences. "As with any procedure that we do, it is important to clearly communicate with the patients what to expect from the procedure and the recovery. Our feedback allowed the company to fine-tune their training program so that clinics that purchase the system have tools they can use to make the experience a very positive one for their patients."
Miramar is in the early stages of commercialization for the MiraDry system. It is being placed in a controlled strategy and is expected to reach "a significant number of practices across the U.S. by the middle of this year," he said.
Dr. Kaminer is chairman of the Miramar Labs scientific advisory board. His institution served as a research study site for the phase II study. SDEF and this news organization are owned by Elsevier.
Microwave technology is a new hyperhidrosis treatment that offers longer-term efficacy than botulinum toxin.
"With microwave thermolysis, we now have a reliable and safe method with long-term efficacy to treat our patients who suffer from hyperhidrosis," said Dr. Michael S. Kaminer, at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
A hyperhidrosis diagnosis is made in patients who have visible, excessive sweating for at least 6 months without apparent cause, and with at least two of the following characteristics (J. Am. Acad. Dermatol. 2004;51:274-86):
• Impairs daily activities.
• Frequency of at least one episode per week.
• Bilateral and symmetric.
• Age of onset before 25 years.
• Positive family history.
• Cessation during sleep.
In the United states, approximately 7.6 million people, or 4% of all U.S. adults, are estimated to have axillary hyperhidrosis with a level on the Hyperhidrosis Disease Severity Scale (HDSS) of either 3 ("My sweating is barely tolerable and frequently interferes with my daily activities.") or 4 ("My sweating is intolerable and always interferes with my daily activities."), according to Dr. Kaminer, managing partner, SkinCare Physicians, Chestnut Hill, Mass. Another 32.5 million are "sweat bothered," with an HDSS level of 2 ("My sweating is tolerable but sometimes interferes with my daily activities.") or 1 ("My sweating is never noticeable and never interferes with my daily activities.").
Axillary hyperhidrosis causes considerable distress among patients. In one survey of the Dermatology Life Quality Index, the condition scored just below atopic dermatitis, close to psoriasis, and above acne vulgaris in terms of quality of life impairment, with a mean baseline score of 10.45, compared with 11.20 for atopic dermatitis, 10.53 for psoriasis, and 7.45 for acne vulgaris (Br. J. Dermatol. 2008;159:997-1035).
Current treatment options include various topical over-the-counter products, iontophoresis, sympathectomy, botulinum toxin, ultrasound (in early trials), and now microwave thermolysis.
The proprietary microwave thermolysis system, MiraDry (Miramar Labs), focuses energy delivery to the dermal-fat interface. Microwave energy is absorbed by electric dipoles. The ion content of sweat is a perfect target for this, said Dr. Kaminer, who is also with the departments of dermatology at Yale University, New Haven, Conn.; Dartmouth College, Hanover, N.H.; and Brown University, Providence, R.I.
Energy becomes concentrated along this interface and creates a focal energy zone. Continuous hydroceramic cooling prevents thermal conduction of heat superficially, and creates a heat zone at the level of sweat glands resulting in thermolysis. The procedure involves injectable local anesthesia. Treatment time is about 30 minutes, with rapid recovery.
Long-term efficacy of the device was demonstrated in the DRIUP study (Dermatologic Reduction in Underarm Perspiration), in which 120 patients with HDSS scores of 3 or 4 were randomized in a 2:1 ratio to one to three active treatments (using a prior investigational version of the microwave device) or sham treatment at seven U.S. sites. Efficacy, defined as patients reporting a reduction to an HDSS score of 1 or 2, was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant.
There were 45 treatment-related adverse events, with 28% of the treatment group and 13% of the sham group patients experiencing at least one adverse event. The most common of these was an area of transient altered sensation in the arm, occurring in 10% of patients in the active treatment group. All but one adverse event resolved over time; the patient withdrew from the study, reporting altered sweating on the face. In all, 71% of the events were rated as mild in severity and didn’t affect patients’ daily activities (Dermatol Surg. 2012;38:185-91). Swelling was common, self-limited, and proportional to the energy delivered, Dr. Kaminer noted.
When compared with botulinum toxin, both approaches are effective, noninvasive (injections only), and involve minimal to no downtime. However, the duration of effect is 6.7 months for botulinum toxin, compared with stable efficacy beginning at 3 months and lasting through 12 months follow-up for microwave thermolysis, he said.
Overall, the advantages of microwave thermolysis over other treatments are its long-term efficacy (greater than 12 months, and likely longer), and its efficacy in more than 90% of patients with the latest-generation device. Disadvantages include the need for two procedures and the less than 100% responder rate. Botulinum toxin, in contrast, is easier to perform, and nearly 100% effective. However, it has the downside of the need for repeat treatments approximately every 6 months, and its cost, along with problematic insurance coverage.
Dr. Kaminer said that his practice participated in a registry-type study in the summer and fall of 2011 and was one of the first clinics in the country to purchase a MiraDry System, in December 2011. "Our staff has found the procedure easy to incorporate into the flow of our practice, and patients have found it to be rather easy to have done. We have seen some modest swelling for up to 1 week after treatment, but that is expected. So far, our experience has been pretty much what we would have expected, and if anything, a little better."
He added that, "Interest in offering this new treatment option to patients with excessive underarm sweating has been extremely high. Between clinics in Japan and the U.S., there have been over 1,500 patients treated."
In all, safety results have been consistent with prior experiences. "As with any procedure that we do, it is important to clearly communicate with the patients what to expect from the procedure and the recovery. Our feedback allowed the company to fine-tune their training program so that clinics that purchase the system have tools they can use to make the experience a very positive one for their patients."
Miramar is in the early stages of commercialization for the MiraDry system. It is being placed in a controlled strategy and is expected to reach "a significant number of practices across the U.S. by the middle of this year," he said.
Dr. Kaminer is chairman of the Miramar Labs scientific advisory board. His institution served as a research study site for the phase II study. SDEF and this news organization are owned by Elsevier.
FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Major Finding: Efficacy was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits.
Data Source: Patients (120) with HDSS scores of 3 or 4 were randomized in a 2:1 ratio to one to three active treatments (using a prior investigational version of the microwave device) or sham treatment at seven U.S. sites.
Disclosures: Dr. Kaminer is chairman of the Miramar Labs scientific advisory board. His institution served as a research study site for the phase II study. SDEF and this news organization are owned by Elsevier.