User login
Ketamine may help refractory status epilepticus
MONTREAL – Intravenous ketamine resolved refractory convulsive status epilepticus in 14 of 17 cases in a small, uncontrolled prospective study.
In addition to its efficacy, ketamine has a unique safety advantage over more commonly used anesthetics. "It does not cause respiratory depression, so it allows us to avoid intubation and ventilatory support in these children," said Dr. Anna Rosati of the University of Florence (Italy). "Because intubation can worsen the prognosis, we believe ketamine should be considered before conventional anesthetics in children with status epilepticus."
The study examined 17 incidents of convulsive status epilepticus that occurred in 13 children. In 13 cases, first-line treatment with midazolam, propofol, or thiopental had failed. Ketamine was the first-line agent in the other four cases, she reported at the 30th International Epilepsy Congress.
The children ranged in age from 2 months to 10 years. The mean duration of status epilepticus before ketamine administration was 16 days, with a range of 5 hours to 86 days. Seizure etiology included focal cortical dysplasia (2), other cortical malformations (3), hydrocephalus (1), febrile infection–related epilepsy syndrome (2), and Rett syndrome (1). One patient had MELAS syndrome (mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes). The etiology was unknown in three.
The ketamine protocol began with two intravenous boluses of 2-3 mg/kg given 5 minutes apart. This was immediately followed by a continuous ketamine infusion, starting at 10 mcg/kg per minute and increasing by 10-mcg increments up to 60 mcg/kg per minute. The mean dosage was 32.5 mcg/kg per minute. Patients also received midazolam as an add-on to prevent emergence reactions. Ketamine was continued for a median of 5 days, with a range of 6 hours to 17 days.
Status epilepticus resolved in 10 of the 13 cases in which ketamine was administered after the first-line drugs had failed and in all 4 cases in which ketamine was the first-line agent. In those four cases, "we were able to avoid intubation," Dr. Rosati said.
The only adverse events noted were hypersalivation, which occurred in all cases, and mild, transient increases in liver enzymes in four cases.
Electroencephalographic changes mirrored clinical improvement, Dr. Rosati said. In the 14 resolved cases, 12 showed a burst suppression pattern and two showed diffuse theta-delta activity. No EEG changes occurred in the three cases without resolution.
Two of the three children who didn’t respond to ketamine had focal cortical dysplasia. Their status resolved after a surgical excision. The third patient had an unknown etiology; status resolved only with a very high thiopental dosage. However, Dr. Rosati noted, a second status episode in that child did resolve with ketamine as a first-line drug.
In light of the small study’s positive results, Dr. Rosati said she is planning to conduct a randomized controlled study of ketamine in status epilepticus.
She said she had no relevant financial disclosures.
MONTREAL – Intravenous ketamine resolved refractory convulsive status epilepticus in 14 of 17 cases in a small, uncontrolled prospective study.
In addition to its efficacy, ketamine has a unique safety advantage over more commonly used anesthetics. "It does not cause respiratory depression, so it allows us to avoid intubation and ventilatory support in these children," said Dr. Anna Rosati of the University of Florence (Italy). "Because intubation can worsen the prognosis, we believe ketamine should be considered before conventional anesthetics in children with status epilepticus."
The study examined 17 incidents of convulsive status epilepticus that occurred in 13 children. In 13 cases, first-line treatment with midazolam, propofol, or thiopental had failed. Ketamine was the first-line agent in the other four cases, she reported at the 30th International Epilepsy Congress.
The children ranged in age from 2 months to 10 years. The mean duration of status epilepticus before ketamine administration was 16 days, with a range of 5 hours to 86 days. Seizure etiology included focal cortical dysplasia (2), other cortical malformations (3), hydrocephalus (1), febrile infection–related epilepsy syndrome (2), and Rett syndrome (1). One patient had MELAS syndrome (mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes). The etiology was unknown in three.
The ketamine protocol began with two intravenous boluses of 2-3 mg/kg given 5 minutes apart. This was immediately followed by a continuous ketamine infusion, starting at 10 mcg/kg per minute and increasing by 10-mcg increments up to 60 mcg/kg per minute. The mean dosage was 32.5 mcg/kg per minute. Patients also received midazolam as an add-on to prevent emergence reactions. Ketamine was continued for a median of 5 days, with a range of 6 hours to 17 days.
Status epilepticus resolved in 10 of the 13 cases in which ketamine was administered after the first-line drugs had failed and in all 4 cases in which ketamine was the first-line agent. In those four cases, "we were able to avoid intubation," Dr. Rosati said.
The only adverse events noted were hypersalivation, which occurred in all cases, and mild, transient increases in liver enzymes in four cases.
Electroencephalographic changes mirrored clinical improvement, Dr. Rosati said. In the 14 resolved cases, 12 showed a burst suppression pattern and two showed diffuse theta-delta activity. No EEG changes occurred in the three cases without resolution.
Two of the three children who didn’t respond to ketamine had focal cortical dysplasia. Their status resolved after a surgical excision. The third patient had an unknown etiology; status resolved only with a very high thiopental dosage. However, Dr. Rosati noted, a second status episode in that child did resolve with ketamine as a first-line drug.
In light of the small study’s positive results, Dr. Rosati said she is planning to conduct a randomized controlled study of ketamine in status epilepticus.
She said she had no relevant financial disclosures.
MONTREAL – Intravenous ketamine resolved refractory convulsive status epilepticus in 14 of 17 cases in a small, uncontrolled prospective study.
In addition to its efficacy, ketamine has a unique safety advantage over more commonly used anesthetics. "It does not cause respiratory depression, so it allows us to avoid intubation and ventilatory support in these children," said Dr. Anna Rosati of the University of Florence (Italy). "Because intubation can worsen the prognosis, we believe ketamine should be considered before conventional anesthetics in children with status epilepticus."
The study examined 17 incidents of convulsive status epilepticus that occurred in 13 children. In 13 cases, first-line treatment with midazolam, propofol, or thiopental had failed. Ketamine was the first-line agent in the other four cases, she reported at the 30th International Epilepsy Congress.
The children ranged in age from 2 months to 10 years. The mean duration of status epilepticus before ketamine administration was 16 days, with a range of 5 hours to 86 days. Seizure etiology included focal cortical dysplasia (2), other cortical malformations (3), hydrocephalus (1), febrile infection–related epilepsy syndrome (2), and Rett syndrome (1). One patient had MELAS syndrome (mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes). The etiology was unknown in three.
The ketamine protocol began with two intravenous boluses of 2-3 mg/kg given 5 minutes apart. This was immediately followed by a continuous ketamine infusion, starting at 10 mcg/kg per minute and increasing by 10-mcg increments up to 60 mcg/kg per minute. The mean dosage was 32.5 mcg/kg per minute. Patients also received midazolam as an add-on to prevent emergence reactions. Ketamine was continued for a median of 5 days, with a range of 6 hours to 17 days.
Status epilepticus resolved in 10 of the 13 cases in which ketamine was administered after the first-line drugs had failed and in all 4 cases in which ketamine was the first-line agent. In those four cases, "we were able to avoid intubation," Dr. Rosati said.
The only adverse events noted were hypersalivation, which occurred in all cases, and mild, transient increases in liver enzymes in four cases.
Electroencephalographic changes mirrored clinical improvement, Dr. Rosati said. In the 14 resolved cases, 12 showed a burst suppression pattern and two showed diffuse theta-delta activity. No EEG changes occurred in the three cases without resolution.
Two of the three children who didn’t respond to ketamine had focal cortical dysplasia. Their status resolved after a surgical excision. The third patient had an unknown etiology; status resolved only with a very high thiopental dosage. However, Dr. Rosati noted, a second status episode in that child did resolve with ketamine as a first-line drug.
In light of the small study’s positive results, Dr. Rosati said she is planning to conduct a randomized controlled study of ketamine in status epilepticus.
She said she had no relevant financial disclosures.
AT IEC 2013
Major finding: Intravenous ketamine resolved 14 of 17 refractory status epilepticus cases in children.
Data source: A prospective study involving 17 episodes that occurred among 13 children.
Disclosures: Dr. Anna Rosati said she had no relevant financial disclosures.
Bone growth affected by ketogenic diet in children with epilepsy
MONTREAL – Epilepsy treatment with the ketogenic diet for more than 6 months slowed the long-term skeletal development of 29 children who participated in a prospective, longitudinal study.
Children who went on the diet fell behind by a mean of 0.1756 lumbar Z-units every year, compared with children who had normal bone growth, Dr. Mark Mackay said at the 30th International Epilepsy Congress.
"Put simply, they accrue bone mass at a slower rate than their age-matched peers," Dr. Mackay of the Royal Children’s Hospital Melbourne, Parkville, Australia, said in an interview.
Children in Dr. Mackay’s study were treated with the ketogenic diet for more than 6 months during 2002-2009. They were a mean of 6 years old when they started the diet, and they followed it for a mean of 6 years.
In addition to the high-fat dietary treatment, they received supplements of calcium and vitamin D to reach the minimum daily requirements. They had dual-energy x-ray absorptiometry (DXA) scans at baseline and every 6 months while on the diet. Normative bone data were used for comparison.
The children also had regular assessment of bone health biomarkers, including serum calcium, potassium, 25-hydroxyvitamin D, parathyroid hormone, bone alkaline phosphatase, osteocalcin, and urinary calcium/creatinine ratio.
Children were grouped at baseline according to mobility status on a 5-point scale, ranging from walking without limitations (1) to being transported in a manual wheelchair (5). At baseline, 11 children had a score of 1 or 2, while 18 scored greater than 2.
Before treatment, their mean bone mineral density lumbar Z-score was 0.99. Children who had no mobility problems had higher baseline scores than did those with mobility problems (mean, –0.125 vs. –1.18). This was not a surprise, said Dr. Mackay, a pediatric neurologist at Royal Children’s Hospital Melbourne. "Many of the children we treat have comorbid physical disability, which also places them at risk of poor bone health, due to factors including decreased weight bearing, physical activity, and sunlight exposure, which is important for vitamin D production."
Subjects on the ketogenic diet demonstrated a mean bone mineral density lumbar Z-score decrease of 0.1756 units/year. Bone loss was greater in children who had higher baseline Z-scores (–0.28 vs. –0.04 units/year).
Their mean serum alkaline phosphatase levels remained in the normal range throughout the study period. However, the investigators measured elevations in mean osteocalcin (26.5 nmol/L) and mean urinary calcium/creatinine ratio (0.77).
The findings highlight the risks of a ketogenic diet, which relies on fat metabolism to induce ketoacidosis. A neutral pH is necessary to mobilize calcium from bone, Dr. Mackay said.
Children who are on the diet are also at risk for deficiencies in micronutrients important for health, including vitamin D and calcium. The timing of diet initiation also plays an important role, he said.
"Childhood and adolescence are critical periods for the normal accrual of bone mass. The bone that is laid down needs to last that person for the rest of their life. Therefore any intervention that affects accrual of bone can have long-lasting health consequences for the child."
While the ketogenic diet is a very effective treatment for some seizure disorders, it’s not risk free, he added. "Some parents see the ketogenic diet as a ‘natural’ alternative to medications, which it is not. Therefore it is important to be improving our knowledge about potential serious long-term side effects so parents can make an informed decision about treatment."
The findings should prompt more study of the diet’s potential long-term impact on skeletal health, Dr. Mackay said. "These will inform development of guidelines for bone surveillance in this high-risk group of predominantly children to minimize potential negative health consequences of the ketogenic diet. ... However, I don’t think we can make any comments at this stage about the role of antiresorptive treatments."
The study was partially funded by Pfizer Australia. Dr. Mackay did not have any financial disclosures.
MONTREAL – Epilepsy treatment with the ketogenic diet for more than 6 months slowed the long-term skeletal development of 29 children who participated in a prospective, longitudinal study.
Children who went on the diet fell behind by a mean of 0.1756 lumbar Z-units every year, compared with children who had normal bone growth, Dr. Mark Mackay said at the 30th International Epilepsy Congress.
"Put simply, they accrue bone mass at a slower rate than their age-matched peers," Dr. Mackay of the Royal Children’s Hospital Melbourne, Parkville, Australia, said in an interview.
Children in Dr. Mackay’s study were treated with the ketogenic diet for more than 6 months during 2002-2009. They were a mean of 6 years old when they started the diet, and they followed it for a mean of 6 years.
In addition to the high-fat dietary treatment, they received supplements of calcium and vitamin D to reach the minimum daily requirements. They had dual-energy x-ray absorptiometry (DXA) scans at baseline and every 6 months while on the diet. Normative bone data were used for comparison.
The children also had regular assessment of bone health biomarkers, including serum calcium, potassium, 25-hydroxyvitamin D, parathyroid hormone, bone alkaline phosphatase, osteocalcin, and urinary calcium/creatinine ratio.
Children were grouped at baseline according to mobility status on a 5-point scale, ranging from walking without limitations (1) to being transported in a manual wheelchair (5). At baseline, 11 children had a score of 1 or 2, while 18 scored greater than 2.
Before treatment, their mean bone mineral density lumbar Z-score was 0.99. Children who had no mobility problems had higher baseline scores than did those with mobility problems (mean, –0.125 vs. –1.18). This was not a surprise, said Dr. Mackay, a pediatric neurologist at Royal Children’s Hospital Melbourne. "Many of the children we treat have comorbid physical disability, which also places them at risk of poor bone health, due to factors including decreased weight bearing, physical activity, and sunlight exposure, which is important for vitamin D production."
Subjects on the ketogenic diet demonstrated a mean bone mineral density lumbar Z-score decrease of 0.1756 units/year. Bone loss was greater in children who had higher baseline Z-scores (–0.28 vs. –0.04 units/year).
Their mean serum alkaline phosphatase levels remained in the normal range throughout the study period. However, the investigators measured elevations in mean osteocalcin (26.5 nmol/L) and mean urinary calcium/creatinine ratio (0.77).
The findings highlight the risks of a ketogenic diet, which relies on fat metabolism to induce ketoacidosis. A neutral pH is necessary to mobilize calcium from bone, Dr. Mackay said.
Children who are on the diet are also at risk for deficiencies in micronutrients important for health, including vitamin D and calcium. The timing of diet initiation also plays an important role, he said.
"Childhood and adolescence are critical periods for the normal accrual of bone mass. The bone that is laid down needs to last that person for the rest of their life. Therefore any intervention that affects accrual of bone can have long-lasting health consequences for the child."
While the ketogenic diet is a very effective treatment for some seizure disorders, it’s not risk free, he added. "Some parents see the ketogenic diet as a ‘natural’ alternative to medications, which it is not. Therefore it is important to be improving our knowledge about potential serious long-term side effects so parents can make an informed decision about treatment."
The findings should prompt more study of the diet’s potential long-term impact on skeletal health, Dr. Mackay said. "These will inform development of guidelines for bone surveillance in this high-risk group of predominantly children to minimize potential negative health consequences of the ketogenic diet. ... However, I don’t think we can make any comments at this stage about the role of antiresorptive treatments."
The study was partially funded by Pfizer Australia. Dr. Mackay did not have any financial disclosures.
MONTREAL – Epilepsy treatment with the ketogenic diet for more than 6 months slowed the long-term skeletal development of 29 children who participated in a prospective, longitudinal study.
Children who went on the diet fell behind by a mean of 0.1756 lumbar Z-units every year, compared with children who had normal bone growth, Dr. Mark Mackay said at the 30th International Epilepsy Congress.
"Put simply, they accrue bone mass at a slower rate than their age-matched peers," Dr. Mackay of the Royal Children’s Hospital Melbourne, Parkville, Australia, said in an interview.
Children in Dr. Mackay’s study were treated with the ketogenic diet for more than 6 months during 2002-2009. They were a mean of 6 years old when they started the diet, and they followed it for a mean of 6 years.
In addition to the high-fat dietary treatment, they received supplements of calcium and vitamin D to reach the minimum daily requirements. They had dual-energy x-ray absorptiometry (DXA) scans at baseline and every 6 months while on the diet. Normative bone data were used for comparison.
The children also had regular assessment of bone health biomarkers, including serum calcium, potassium, 25-hydroxyvitamin D, parathyroid hormone, bone alkaline phosphatase, osteocalcin, and urinary calcium/creatinine ratio.
Children were grouped at baseline according to mobility status on a 5-point scale, ranging from walking without limitations (1) to being transported in a manual wheelchair (5). At baseline, 11 children had a score of 1 or 2, while 18 scored greater than 2.
Before treatment, their mean bone mineral density lumbar Z-score was 0.99. Children who had no mobility problems had higher baseline scores than did those with mobility problems (mean, –0.125 vs. –1.18). This was not a surprise, said Dr. Mackay, a pediatric neurologist at Royal Children’s Hospital Melbourne. "Many of the children we treat have comorbid physical disability, which also places them at risk of poor bone health, due to factors including decreased weight bearing, physical activity, and sunlight exposure, which is important for vitamin D production."
Subjects on the ketogenic diet demonstrated a mean bone mineral density lumbar Z-score decrease of 0.1756 units/year. Bone loss was greater in children who had higher baseline Z-scores (–0.28 vs. –0.04 units/year).
Their mean serum alkaline phosphatase levels remained in the normal range throughout the study period. However, the investigators measured elevations in mean osteocalcin (26.5 nmol/L) and mean urinary calcium/creatinine ratio (0.77).
The findings highlight the risks of a ketogenic diet, which relies on fat metabolism to induce ketoacidosis. A neutral pH is necessary to mobilize calcium from bone, Dr. Mackay said.
Children who are on the diet are also at risk for deficiencies in micronutrients important for health, including vitamin D and calcium. The timing of diet initiation also plays an important role, he said.
"Childhood and adolescence are critical periods for the normal accrual of bone mass. The bone that is laid down needs to last that person for the rest of their life. Therefore any intervention that affects accrual of bone can have long-lasting health consequences for the child."
While the ketogenic diet is a very effective treatment for some seizure disorders, it’s not risk free, he added. "Some parents see the ketogenic diet as a ‘natural’ alternative to medications, which it is not. Therefore it is important to be improving our knowledge about potential serious long-term side effects so parents can make an informed decision about treatment."
The findings should prompt more study of the diet’s potential long-term impact on skeletal health, Dr. Mackay said. "These will inform development of guidelines for bone surveillance in this high-risk group of predominantly children to minimize potential negative health consequences of the ketogenic diet. ... However, I don’t think we can make any comments at this stage about the role of antiresorptive treatments."
The study was partially funded by Pfizer Australia. Dr. Mackay did not have any financial disclosures.
AT IEC 2013
Major finding: Participants on the ketogenic diet demonstrated a mean bone mineral density lumbar Z-score decrease of 0.1756 units/year. Bone loss was greater in children who had higher baseline Z-scores (–0.28 vs. –0.04 units/year).
Data source: A prospective, longitudinal study of 29 children who were treated with the ketogenic diet for more than 6 months during 2002-2009.
Disclosures: The study was partially funded by Pfizer Australia. Dr. Mackay did not have any financial disclosures.
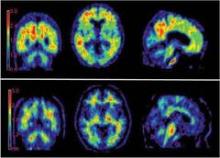
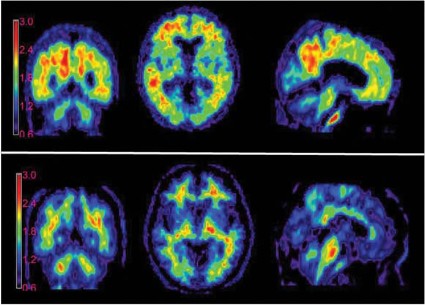
Medicare: Cover amyloid imaging in trials only
Medicare coverage for amyloid brain imaging should be severely limited, according to a proposed Medicare coverage decision released July 8.
The federal health program proposes to cover only one scan per patient and only as part of a clinical study or to rule out Alzheimer’s disease in narrowly defined and clinically difficult differential diagnoses. The procedure isn’t "reasonable and necessary" otherwise, because there are not enough data to conclude that beta-amyloid imaging with positron emission tomography (PET) improves outcomes for patients with Alzheimer’s disease.
Instead, the Centers for Medicare and Medicaid Services (CMS) proposes to cover the procedure under its Coverage with Evidence Development (CED) program, which provides coverage while collecting additional outcomes data. Coverage under CED contributes to a new technology’s developmental pathway by encouraging more studies that employ it, according to CMS.
Under the draft proposal, PET amyloid imaging would be covered only in CMS-approved, prospective, randomized trials that include subjects from appropriate populations, and, when appropriate, employ the gold standard of postmortem Alzheimer’s diagnosis.
Study populations must comprise Medicare beneficiaries with cognitive impairment suspicious for Alzheimer’s disease, or who may be at risk for developing the disease. Furthermore, studies must address at least one of the following questions:
• Does PET amyloid imaging lead to improved health outcomes, including avoidance of futile treatment or tests; improving or slowing the decline of quality of life; and survival?
• Does treatment guided by PET amyloid imaging identify specific subpopulations, patient characteristics, or differential diagnoses that predict improved health outcomes?
• Can PET amyloid imaging enrich the patient populations of Alzheimer’s trials by selecting patients on the basis of biological as well as clinical and epidemiologic factors? And if so, can this lead to improved health outcomes?
The payment structure will be reconsidered after another evidence review at some unspecified future time, CMS noted.
The draft decision is a disappointment for both patients and the drug companies that are developing amyloid imaging agents. Past CED programs have taken as long as 7 years to return enough evidence for a federal payment reevaluation, according to a statement issued by the Alzheimer’s Association.
"Neither families directly impacted by the disease, nor our federal government, can afford to wait as much as 7 years for a final decision about national coverage, as was the case with the National Oncology Patient Registry and the evaluation by CMS of FDG [fluorodeoxyglucose] PET coverage," the statement said. "The timeframe at which CMS has conducted CED processes is wholly unsuited and unacceptable to both the pace of scientific and technological innovation in the Alzheimer’s field, and more importantly, the rapidly increasing needs posed by the escalating Alzheimer’s epidemic. If the federal government follows this example and timeline, it will hinder coverage of a badly needed, already FDA-approved diagnostic tool in limited populations in which sufficient evidence indicates this technology has meaningful impact."
Avid Radiopharmaceuticals, which manufactures the imaging agent florbetapir F-18 injection (Amyvid), agreed.
"CMS appears to be challenging the value of an adjunctive tool that can assist physicians in making a more informed diagnosis for patients with cognitive impairment," said Dr. Daniel Skovronsky, the company’s president and chief executive officer. "Restricting coverage could hinder a timely and accurate diagnosis, which is in conflict with the advice of Alzheimer’s disease experts and with the administration’s National Alzheimer’s Project Act."
Last January, the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging (SNMMI) issued revised appropriate use criteria for these agents. Imaging should be reserved for those with persistent or progressive unexplained mild cognitive impairment, MCI that has an atypical presentation, and MCI which develops at an atypically young age, the document maintained.
Imaging was not considered appropriate for asymptomatic patients; for those with typical-onset dementia; as a substitute for risk genotyping in patients with a family history; or to determine dementia severity.
The SNMMI criteria address a small but important patient population not addressed under the CMS proposed payment scheme, said Dr. Richard J. Caselli, professor of neurology at the Mayo Clinic in Scottsdale, Ariz. He also is associate director and clinical core director of Mayo’s Alzheimer’s Disease Center.
"While I can understand the rationale for not reimbursing this in the typical elderly dementia patient, there is another population, albeit smaller, that would clearly benefit: the working patient," he said in an interview. "Imagine a 52-year-old man with no family history, who starts developing memory loss, has trouble on the job as a result, and seeks help. In this case, a diagnosis of Alzheimer’s is unexpected, and will result in disability."
While Medicare does not provide health insurance for working adults, such a diagnosis could lead to Medicare disability coverage.
"In this case, a [PET amyloid scan] with positive evidence for Alzheimer’s would in fact save everyone a lot of angst and expense, and the patient could be properly managed in terms of disability," Dr. Caselli said. "Further, young patients with altered mental status may have other serious illnesses, such as autoimmune encephalopathy, so having access to such a scan might save someone trials of steroids or other immunosuppressive agents."
CMS is accepting public comment on the draft recommendation through Aug. 3.
Dr. Caselli has no financial disclosures.
Medicare coverage for amyloid brain imaging should be severely limited, according to a proposed Medicare coverage decision released July 8.
The federal health program proposes to cover only one scan per patient and only as part of a clinical study or to rule out Alzheimer’s disease in narrowly defined and clinically difficult differential diagnoses. The procedure isn’t "reasonable and necessary" otherwise, because there are not enough data to conclude that beta-amyloid imaging with positron emission tomography (PET) improves outcomes for patients with Alzheimer’s disease.
Instead, the Centers for Medicare and Medicaid Services (CMS) proposes to cover the procedure under its Coverage with Evidence Development (CED) program, which provides coverage while collecting additional outcomes data. Coverage under CED contributes to a new technology’s developmental pathway by encouraging more studies that employ it, according to CMS.
Under the draft proposal, PET amyloid imaging would be covered only in CMS-approved, prospective, randomized trials that include subjects from appropriate populations, and, when appropriate, employ the gold standard of postmortem Alzheimer’s diagnosis.
Study populations must comprise Medicare beneficiaries with cognitive impairment suspicious for Alzheimer’s disease, or who may be at risk for developing the disease. Furthermore, studies must address at least one of the following questions:
• Does PET amyloid imaging lead to improved health outcomes, including avoidance of futile treatment or tests; improving or slowing the decline of quality of life; and survival?
• Does treatment guided by PET amyloid imaging identify specific subpopulations, patient characteristics, or differential diagnoses that predict improved health outcomes?
• Can PET amyloid imaging enrich the patient populations of Alzheimer’s trials by selecting patients on the basis of biological as well as clinical and epidemiologic factors? And if so, can this lead to improved health outcomes?
The payment structure will be reconsidered after another evidence review at some unspecified future time, CMS noted.
The draft decision is a disappointment for both patients and the drug companies that are developing amyloid imaging agents. Past CED programs have taken as long as 7 years to return enough evidence for a federal payment reevaluation, according to a statement issued by the Alzheimer’s Association.
"Neither families directly impacted by the disease, nor our federal government, can afford to wait as much as 7 years for a final decision about national coverage, as was the case with the National Oncology Patient Registry and the evaluation by CMS of FDG [fluorodeoxyglucose] PET coverage," the statement said. "The timeframe at which CMS has conducted CED processes is wholly unsuited and unacceptable to both the pace of scientific and technological innovation in the Alzheimer’s field, and more importantly, the rapidly increasing needs posed by the escalating Alzheimer’s epidemic. If the federal government follows this example and timeline, it will hinder coverage of a badly needed, already FDA-approved diagnostic tool in limited populations in which sufficient evidence indicates this technology has meaningful impact."
Avid Radiopharmaceuticals, which manufactures the imaging agent florbetapir F-18 injection (Amyvid), agreed.
"CMS appears to be challenging the value of an adjunctive tool that can assist physicians in making a more informed diagnosis for patients with cognitive impairment," said Dr. Daniel Skovronsky, the company’s president and chief executive officer. "Restricting coverage could hinder a timely and accurate diagnosis, which is in conflict with the advice of Alzheimer’s disease experts and with the administration’s National Alzheimer’s Project Act."
Last January, the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging (SNMMI) issued revised appropriate use criteria for these agents. Imaging should be reserved for those with persistent or progressive unexplained mild cognitive impairment, MCI that has an atypical presentation, and MCI which develops at an atypically young age, the document maintained.
Imaging was not considered appropriate for asymptomatic patients; for those with typical-onset dementia; as a substitute for risk genotyping in patients with a family history; or to determine dementia severity.
The SNMMI criteria address a small but important patient population not addressed under the CMS proposed payment scheme, said Dr. Richard J. Caselli, professor of neurology at the Mayo Clinic in Scottsdale, Ariz. He also is associate director and clinical core director of Mayo’s Alzheimer’s Disease Center.
"While I can understand the rationale for not reimbursing this in the typical elderly dementia patient, there is another population, albeit smaller, that would clearly benefit: the working patient," he said in an interview. "Imagine a 52-year-old man with no family history, who starts developing memory loss, has trouble on the job as a result, and seeks help. In this case, a diagnosis of Alzheimer’s is unexpected, and will result in disability."
While Medicare does not provide health insurance for working adults, such a diagnosis could lead to Medicare disability coverage.
"In this case, a [PET amyloid scan] with positive evidence for Alzheimer’s would in fact save everyone a lot of angst and expense, and the patient could be properly managed in terms of disability," Dr. Caselli said. "Further, young patients with altered mental status may have other serious illnesses, such as autoimmune encephalopathy, so having access to such a scan might save someone trials of steroids or other immunosuppressive agents."
CMS is accepting public comment on the draft recommendation through Aug. 3.
Dr. Caselli has no financial disclosures.
Medicare coverage for amyloid brain imaging should be severely limited, according to a proposed Medicare coverage decision released July 8.
The federal health program proposes to cover only one scan per patient and only as part of a clinical study or to rule out Alzheimer’s disease in narrowly defined and clinically difficult differential diagnoses. The procedure isn’t "reasonable and necessary" otherwise, because there are not enough data to conclude that beta-amyloid imaging with positron emission tomography (PET) improves outcomes for patients with Alzheimer’s disease.
Instead, the Centers for Medicare and Medicaid Services (CMS) proposes to cover the procedure under its Coverage with Evidence Development (CED) program, which provides coverage while collecting additional outcomes data. Coverage under CED contributes to a new technology’s developmental pathway by encouraging more studies that employ it, according to CMS.
Under the draft proposal, PET amyloid imaging would be covered only in CMS-approved, prospective, randomized trials that include subjects from appropriate populations, and, when appropriate, employ the gold standard of postmortem Alzheimer’s diagnosis.
Study populations must comprise Medicare beneficiaries with cognitive impairment suspicious for Alzheimer’s disease, or who may be at risk for developing the disease. Furthermore, studies must address at least one of the following questions:
• Does PET amyloid imaging lead to improved health outcomes, including avoidance of futile treatment or tests; improving or slowing the decline of quality of life; and survival?
• Does treatment guided by PET amyloid imaging identify specific subpopulations, patient characteristics, or differential diagnoses that predict improved health outcomes?
• Can PET amyloid imaging enrich the patient populations of Alzheimer’s trials by selecting patients on the basis of biological as well as clinical and epidemiologic factors? And if so, can this lead to improved health outcomes?
The payment structure will be reconsidered after another evidence review at some unspecified future time, CMS noted.
The draft decision is a disappointment for both patients and the drug companies that are developing amyloid imaging agents. Past CED programs have taken as long as 7 years to return enough evidence for a federal payment reevaluation, according to a statement issued by the Alzheimer’s Association.
"Neither families directly impacted by the disease, nor our federal government, can afford to wait as much as 7 years for a final decision about national coverage, as was the case with the National Oncology Patient Registry and the evaluation by CMS of FDG [fluorodeoxyglucose] PET coverage," the statement said. "The timeframe at which CMS has conducted CED processes is wholly unsuited and unacceptable to both the pace of scientific and technological innovation in the Alzheimer’s field, and more importantly, the rapidly increasing needs posed by the escalating Alzheimer’s epidemic. If the federal government follows this example and timeline, it will hinder coverage of a badly needed, already FDA-approved diagnostic tool in limited populations in which sufficient evidence indicates this technology has meaningful impact."
Avid Radiopharmaceuticals, which manufactures the imaging agent florbetapir F-18 injection (Amyvid), agreed.
"CMS appears to be challenging the value of an adjunctive tool that can assist physicians in making a more informed diagnosis for patients with cognitive impairment," said Dr. Daniel Skovronsky, the company’s president and chief executive officer. "Restricting coverage could hinder a timely and accurate diagnosis, which is in conflict with the advice of Alzheimer’s disease experts and with the administration’s National Alzheimer’s Project Act."
Last January, the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging (SNMMI) issued revised appropriate use criteria for these agents. Imaging should be reserved for those with persistent or progressive unexplained mild cognitive impairment, MCI that has an atypical presentation, and MCI which develops at an atypically young age, the document maintained.
Imaging was not considered appropriate for asymptomatic patients; for those with typical-onset dementia; as a substitute for risk genotyping in patients with a family history; or to determine dementia severity.
The SNMMI criteria address a small but important patient population not addressed under the CMS proposed payment scheme, said Dr. Richard J. Caselli, professor of neurology at the Mayo Clinic in Scottsdale, Ariz. He also is associate director and clinical core director of Mayo’s Alzheimer’s Disease Center.
"While I can understand the rationale for not reimbursing this in the typical elderly dementia patient, there is another population, albeit smaller, that would clearly benefit: the working patient," he said in an interview. "Imagine a 52-year-old man with no family history, who starts developing memory loss, has trouble on the job as a result, and seeks help. In this case, a diagnosis of Alzheimer’s is unexpected, and will result in disability."
While Medicare does not provide health insurance for working adults, such a diagnosis could lead to Medicare disability coverage.
"In this case, a [PET amyloid scan] with positive evidence for Alzheimer’s would in fact save everyone a lot of angst and expense, and the patient could be properly managed in terms of disability," Dr. Caselli said. "Further, young patients with altered mental status may have other serious illnesses, such as autoimmune encephalopathy, so having access to such a scan might save someone trials of steroids or other immunosuppressive agents."
CMS is accepting public comment on the draft recommendation through Aug. 3.
Dr. Caselli has no financial disclosures.
ACPA-positivity in RA does not preclude drug-free remission
MADRID – Drug-free remission is possible for rheumatoid arthritis patients with anticitrullinated protein antibodies, although they may not be able to sustain it for as long as ACPA-negative patients.
Still, after treatment with methotrexate and tapered prednisone, more than one-third of ACPA-positive patients experienced drug-free remission for at least some period of time, Dr. Kirsten Wevers-de Boer said at the annual European Congress of Rheumatology.
"With this treatment strategy, the presence of ACPA appears not to preclude drug discontinuation," said Dr. Wevers-de Boer of the department of rheumatology at Leiden University Medical Center in the Netherlands.
She and her colleagues conducted a subanalysis of patients who achieved remission in the IMPROVED study, which examined remission rates in 610 patients with recent-onset rheumatoid or undifferentiated arthritis. They were treated with a two-step regimen of 7 weeks of methotrexate 25 mg/week along with a tapered course of prednisone. Prednisone was started at 60 mg/day and tapered to 7.5 mg/day by the end of the treatment period (Ann. Rheum. Dis. 2013 May 28 [doi:10.1136/annrheumdis-2013-203243]).
Patients not in remission 4 months after the treatment cycle were randomized to either one of two regimens – methotrexate, hydroxychloroquine, sulfasalazine, and low-dose prednisone, or methotrexate plus adalimumab.
Dr. Wevers-de Boer’s subanalysis focused on the 375 patients (61%) who achieved early drug-free remission. They were followed for up to 16 months.
At 1 year, 119 (32%) had remained in drug-free remission. Significantly more of the patients who were in drug-free remission at 1 year were ACPA negative than were those who were not in remission (62% vs. 50%).
At 16 months, 77 patients (65% of the entire group) were still in drug-free remission. Again, ACPA negativity was more often present among those who were still in drug-free remission than it was in those who were not in remission (80% vs. 58%).
Dr. Wevers-de Boer conducted regression analyses to look for any significant baseline factors that might predict the inability to sustain drug-free remission.
In the univariate analysis, significant factors were a high tender joint count at baseline, being ACPA-positive, and fulfilling the 2010 American College of Rheumatology-European League Against Rheumatism diagnostic criteria. In the multivariate analysis, the combination of high tender joint count and meeting the 2010 diagnostic criteria was significantly associated with an inability to sustain remission.
"Patients who are ACPA positive can achieve drug-free remission almost as often as patients who are ACPA negative, although they are somewhat less often able to sustain that," she said.
Dr. Wevers-de Boer disclosed that she has received grant funding from Abbott, which sponsored the IMPROVED study.
MADRID – Drug-free remission is possible for rheumatoid arthritis patients with anticitrullinated protein antibodies, although they may not be able to sustain it for as long as ACPA-negative patients.
Still, after treatment with methotrexate and tapered prednisone, more than one-third of ACPA-positive patients experienced drug-free remission for at least some period of time, Dr. Kirsten Wevers-de Boer said at the annual European Congress of Rheumatology.
"With this treatment strategy, the presence of ACPA appears not to preclude drug discontinuation," said Dr. Wevers-de Boer of the department of rheumatology at Leiden University Medical Center in the Netherlands.
She and her colleagues conducted a subanalysis of patients who achieved remission in the IMPROVED study, which examined remission rates in 610 patients with recent-onset rheumatoid or undifferentiated arthritis. They were treated with a two-step regimen of 7 weeks of methotrexate 25 mg/week along with a tapered course of prednisone. Prednisone was started at 60 mg/day and tapered to 7.5 mg/day by the end of the treatment period (Ann. Rheum. Dis. 2013 May 28 [doi:10.1136/annrheumdis-2013-203243]).
Patients not in remission 4 months after the treatment cycle were randomized to either one of two regimens – methotrexate, hydroxychloroquine, sulfasalazine, and low-dose prednisone, or methotrexate plus adalimumab.
Dr. Wevers-de Boer’s subanalysis focused on the 375 patients (61%) who achieved early drug-free remission. They were followed for up to 16 months.
At 1 year, 119 (32%) had remained in drug-free remission. Significantly more of the patients who were in drug-free remission at 1 year were ACPA negative than were those who were not in remission (62% vs. 50%).
At 16 months, 77 patients (65% of the entire group) were still in drug-free remission. Again, ACPA negativity was more often present among those who were still in drug-free remission than it was in those who were not in remission (80% vs. 58%).
Dr. Wevers-de Boer conducted regression analyses to look for any significant baseline factors that might predict the inability to sustain drug-free remission.
In the univariate analysis, significant factors were a high tender joint count at baseline, being ACPA-positive, and fulfilling the 2010 American College of Rheumatology-European League Against Rheumatism diagnostic criteria. In the multivariate analysis, the combination of high tender joint count and meeting the 2010 diagnostic criteria was significantly associated with an inability to sustain remission.
"Patients who are ACPA positive can achieve drug-free remission almost as often as patients who are ACPA negative, although they are somewhat less often able to sustain that," she said.
Dr. Wevers-de Boer disclosed that she has received grant funding from Abbott, which sponsored the IMPROVED study.
MADRID – Drug-free remission is possible for rheumatoid arthritis patients with anticitrullinated protein antibodies, although they may not be able to sustain it for as long as ACPA-negative patients.
Still, after treatment with methotrexate and tapered prednisone, more than one-third of ACPA-positive patients experienced drug-free remission for at least some period of time, Dr. Kirsten Wevers-de Boer said at the annual European Congress of Rheumatology.
"With this treatment strategy, the presence of ACPA appears not to preclude drug discontinuation," said Dr. Wevers-de Boer of the department of rheumatology at Leiden University Medical Center in the Netherlands.
She and her colleagues conducted a subanalysis of patients who achieved remission in the IMPROVED study, which examined remission rates in 610 patients with recent-onset rheumatoid or undifferentiated arthritis. They were treated with a two-step regimen of 7 weeks of methotrexate 25 mg/week along with a tapered course of prednisone. Prednisone was started at 60 mg/day and tapered to 7.5 mg/day by the end of the treatment period (Ann. Rheum. Dis. 2013 May 28 [doi:10.1136/annrheumdis-2013-203243]).
Patients not in remission 4 months after the treatment cycle were randomized to either one of two regimens – methotrexate, hydroxychloroquine, sulfasalazine, and low-dose prednisone, or methotrexate plus adalimumab.
Dr. Wevers-de Boer’s subanalysis focused on the 375 patients (61%) who achieved early drug-free remission. They were followed for up to 16 months.
At 1 year, 119 (32%) had remained in drug-free remission. Significantly more of the patients who were in drug-free remission at 1 year were ACPA negative than were those who were not in remission (62% vs. 50%).
At 16 months, 77 patients (65% of the entire group) were still in drug-free remission. Again, ACPA negativity was more often present among those who were still in drug-free remission than it was in those who were not in remission (80% vs. 58%).
Dr. Wevers-de Boer conducted regression analyses to look for any significant baseline factors that might predict the inability to sustain drug-free remission.
In the univariate analysis, significant factors were a high tender joint count at baseline, being ACPA-positive, and fulfilling the 2010 American College of Rheumatology-European League Against Rheumatism diagnostic criteria. In the multivariate analysis, the combination of high tender joint count and meeting the 2010 diagnostic criteria was significantly associated with an inability to sustain remission.
"Patients who are ACPA positive can achieve drug-free remission almost as often as patients who are ACPA negative, although they are somewhat less often able to sustain that," she said.
Dr. Wevers-de Boer disclosed that she has received grant funding from Abbott, which sponsored the IMPROVED study.
AT THE EULAR CONGRESS 2013
Major finding: Significantly more of these patients who were in drug-free remission at 1 year were ACPA-negative than were those who were not in remission (62% vs. 50%).
Data source: A subanalysis of 375 patients who achieved early drug-free remission in the IMPROVED study.
Disclosures: Dr. Wevers-de Boer reported receiving grant funding from Abbott, the IMPROVED study sponsor.
New biomarkers find 8% more patients with early RA
MADRID – Four newly identified serologic biomarkers have the potential to increase the chances of an early diagnosis for patients with rheumatoid arthritis.
When added to the standard biomarker panel, the new markers – UH-RA.1, UH-RA.9, UH-RA.14, and UH-RA.21 – found about 8% more patients with new-onset RA, Dr. Liesbeth De Winter reported at the annual European Congress of Rheumatology.
The current diagnostic methods, based on 2010 RA Classification Criteria and a two-antibody serological panel, miss about one-third of patients with new-onset disease, said Dr. De Winter of the Biomedical Research Institute of Hasselt University, Diepenbeek, Belgium. The resulting delay in diagnosis decreases the chance of a good outcome for the group.
"Early diagnosis is important, because there is a therapeutic window in the first few years during which early intervention and treatment yield a better outcome, and patients are much more likely to go into remission when treated at this time," she said.
Researchers at the same institute first published on these new biomarkers in 2011 (J. Autoimmun. 2011;36:33-46). Since then, Dr. De Winter said, they have been validated and found to be significantly associated with early-onset disease.
The team used the current diagnostic criteria in a group of 292 patients with RA, 97 healthy controls, and 90 rheumatic controls (including patients with arthritis, ankylosing spondylitis, osteoarthritis, and Sjögren’s syndrome). About one-third (34%) of the RA patients were seronegative for the diagnostic biomarkers of rheumatoid factor and anti-citrullinated protein antibodies (ACPAs). This 34% was termed the "serological gap."
But 26% of these patients were also positive for at least one of the four new biomarkers, "narrowing the serological gap by 8%," Dr. De Winter said. Of the 39 patients with very early RA, 13% were also positive for the new biomarkers, she added.
The test should be easy and inexpensive to implement, as it could simply be added to the current biomarker panel in a routine patient workup, she added.
The next step is to examine whether the markers can predict either disease course or therapeutic response, she said. "It’s already been shown that patients who are positive for ACPA respond differently to treatment. We also want to try and identify their role in the disease process, to help us gain some additional knowledge about it."
Identifying more early-stage patients could have a large impact on disease burden, Dr. Maya Buch said during a press briefing on the study.
"There’s currently a risk that some of our early-stage patients are being neglected because the diagnostic criteria as they are now are weighted toward ACPA-positive patients," said Dr. Buch of the University of Leeds, England. "I think it’s likely that even more biomarkers will be identified over time, further increasing our ability to catch these patients early. Incremental work like this fills the need for patients that we are not currently able to diagnose as early as necessary."
Dr. De Winter had no financial disclosures.
MADRID – Four newly identified serologic biomarkers have the potential to increase the chances of an early diagnosis for patients with rheumatoid arthritis.
When added to the standard biomarker panel, the new markers – UH-RA.1, UH-RA.9, UH-RA.14, and UH-RA.21 – found about 8% more patients with new-onset RA, Dr. Liesbeth De Winter reported at the annual European Congress of Rheumatology.
The current diagnostic methods, based on 2010 RA Classification Criteria and a two-antibody serological panel, miss about one-third of patients with new-onset disease, said Dr. De Winter of the Biomedical Research Institute of Hasselt University, Diepenbeek, Belgium. The resulting delay in diagnosis decreases the chance of a good outcome for the group.
"Early diagnosis is important, because there is a therapeutic window in the first few years during which early intervention and treatment yield a better outcome, and patients are much more likely to go into remission when treated at this time," she said.
Researchers at the same institute first published on these new biomarkers in 2011 (J. Autoimmun. 2011;36:33-46). Since then, Dr. De Winter said, they have been validated and found to be significantly associated with early-onset disease.
The team used the current diagnostic criteria in a group of 292 patients with RA, 97 healthy controls, and 90 rheumatic controls (including patients with arthritis, ankylosing spondylitis, osteoarthritis, and Sjögren’s syndrome). About one-third (34%) of the RA patients were seronegative for the diagnostic biomarkers of rheumatoid factor and anti-citrullinated protein antibodies (ACPAs). This 34% was termed the "serological gap."
But 26% of these patients were also positive for at least one of the four new biomarkers, "narrowing the serological gap by 8%," Dr. De Winter said. Of the 39 patients with very early RA, 13% were also positive for the new biomarkers, she added.
The test should be easy and inexpensive to implement, as it could simply be added to the current biomarker panel in a routine patient workup, she added.
The next step is to examine whether the markers can predict either disease course or therapeutic response, she said. "It’s already been shown that patients who are positive for ACPA respond differently to treatment. We also want to try and identify their role in the disease process, to help us gain some additional knowledge about it."
Identifying more early-stage patients could have a large impact on disease burden, Dr. Maya Buch said during a press briefing on the study.
"There’s currently a risk that some of our early-stage patients are being neglected because the diagnostic criteria as they are now are weighted toward ACPA-positive patients," said Dr. Buch of the University of Leeds, England. "I think it’s likely that even more biomarkers will be identified over time, further increasing our ability to catch these patients early. Incremental work like this fills the need for patients that we are not currently able to diagnose as early as necessary."
Dr. De Winter had no financial disclosures.
MADRID – Four newly identified serologic biomarkers have the potential to increase the chances of an early diagnosis for patients with rheumatoid arthritis.
When added to the standard biomarker panel, the new markers – UH-RA.1, UH-RA.9, UH-RA.14, and UH-RA.21 – found about 8% more patients with new-onset RA, Dr. Liesbeth De Winter reported at the annual European Congress of Rheumatology.
The current diagnostic methods, based on 2010 RA Classification Criteria and a two-antibody serological panel, miss about one-third of patients with new-onset disease, said Dr. De Winter of the Biomedical Research Institute of Hasselt University, Diepenbeek, Belgium. The resulting delay in diagnosis decreases the chance of a good outcome for the group.
"Early diagnosis is important, because there is a therapeutic window in the first few years during which early intervention and treatment yield a better outcome, and patients are much more likely to go into remission when treated at this time," she said.
Researchers at the same institute first published on these new biomarkers in 2011 (J. Autoimmun. 2011;36:33-46). Since then, Dr. De Winter said, they have been validated and found to be significantly associated with early-onset disease.
The team used the current diagnostic criteria in a group of 292 patients with RA, 97 healthy controls, and 90 rheumatic controls (including patients with arthritis, ankylosing spondylitis, osteoarthritis, and Sjögren’s syndrome). About one-third (34%) of the RA patients were seronegative for the diagnostic biomarkers of rheumatoid factor and anti-citrullinated protein antibodies (ACPAs). This 34% was termed the "serological gap."
But 26% of these patients were also positive for at least one of the four new biomarkers, "narrowing the serological gap by 8%," Dr. De Winter said. Of the 39 patients with very early RA, 13% were also positive for the new biomarkers, she added.
The test should be easy and inexpensive to implement, as it could simply be added to the current biomarker panel in a routine patient workup, she added.
The next step is to examine whether the markers can predict either disease course or therapeutic response, she said. "It’s already been shown that patients who are positive for ACPA respond differently to treatment. We also want to try and identify their role in the disease process, to help us gain some additional knowledge about it."
Identifying more early-stage patients could have a large impact on disease burden, Dr. Maya Buch said during a press briefing on the study.
"There’s currently a risk that some of our early-stage patients are being neglected because the diagnostic criteria as they are now are weighted toward ACPA-positive patients," said Dr. Buch of the University of Leeds, England. "I think it’s likely that even more biomarkers will be identified over time, further increasing our ability to catch these patients early. Incremental work like this fills the need for patients that we are not currently able to diagnose as early as necessary."
Dr. De Winter had no financial disclosures.
AT THE EULAR CONGRESS 2013
Major finding: A panel of four new serologic biomarkers found about 8% more patients with early rheumatoid arthritis, improving the rate of missed diagnosis from 34% to 26%.
Data source: A diagnostic study of 292 patients with RA, 97 healthy controls, and 90 rheumatic controls (including patients with arthritis, ankylosing spondylitis, osteoarthritis, and Sjögren’s syndrome).
Disclosures: Dr. De Winter had no financial disclosures.
RA remission similar for tocilizumab alone or with methotrexate
MADRID – Patients with early, active rheumatoid arthritis who took tocilizumab – either alone or in combination with methotrexate – continued to benefit from it by the end of a 2-year study.
About half of those on either treatment strategy in the study achieved remission by the end of the first year and this did not change appreciably by the end of the second year. There was also a very low rate of radiographic progression, Dr. Tom Huizinga said at the annual European Congress of Rheumatology.
The results confirm and extend the earlier findings of ACT-RAY, a 2-year, randomized, placebo-controlled study of tocilizumab employed as a switch or add-on therapy for patients with early, active rheumatoid arthritis (RA). The 24-week data, published earlier this year, showed that tocilizumab was just as effective without methotrexate as with it, suggesting that it could be employed as monotherapy (Ann. Rheum. Dis. 2013;72:43-50).
All 553 patients in ACT-RAY received open-label tocilizumab 8 mg/kg intravenously every 4 weeks. They were randomized to the switch strategy (tocilizumab 8 mg/kg IV every 4 weeks with oral placebo) or the add-on strategy (tocilizumab 8 mg/kg IV every 4 weeks plus 2.5 mg methotrexate), said Dr. Huizinga, head of the department of rheumatology at Leiden (The Netherlands) University Medical Center.
Most of the patients (81%) were women; mean age was 53 years. Patients had a mean disease duration of 8 years and a mean Disease Activity Score–28 (DAS28) of 6.4.
Most of the study group (433) completed the second year of treatment. Reasons for withdrawal included lack of efficacy (2% in the add-on strategy group and 5% in the switch strategy group) and adverse events (10% of add-on patients and 11% of switch). There were three deaths in the add-on group and six in the switch group.
Sustained remission was defined as a DAS28 of less than 2.6 at two consecutive visits separated by 12 weeks. By week 52, about 50% of the overall cohort (53% add-on strategy and 47% switch strategy) had achieved remission and were able to discontinue tocilizumab.
By week 104, 86% of the overall cohort had experienced a flare in disease activity, with a median time of 90 days from tocilizumab discontinuation. Most of those patients restarted tocilizumab. The medication continued to be effective. The mean DAS28 at flare was 4.46, dropping to a mean of 2.99 within 4 weeks of restarting treatment.
The mean DAS28 score at week 104 was unchanged from the score at week 52, decreasing by 3.6 from baseline in both groups. The large majority of each group experienced no radiographic progression during year 2 (94% of the add-on and 91% of the switch groups).
By the end of the study at week 104, 23% of the add-on group and 18% of the switch group were in remission as measured by the Clinical Disease Activity Index – virtually identical to CDAI remission rates at week 52.
The safety results were consistent with previous findings, Dr. Huizinga said. Serious adverse events and infections occurred in 15% of the add-on group and 4% of the switch group.
Liver enzyme elevations were more common among those in the add-on group. Elevations of up to three times the upper limit of normal of alanine aminotransferase occurred in 58% of the add-on group and 40% of the switch group. Elevations of up to five times the upper limit occurred in 13% of the add-on group and 5% of the switch group. Aspartate transaminase elevations of up to three times the upper limit of normal occurred in 51% of the add-on group and 30% of the switch group. Elevations of up to five times the upper limit of normal occurred in 5% of the add-on and 2% of the switch groups.
During a discussion period after his presentation, Dr. Huizinga addressed the issue of clinical impact, saying all of the information isn’t in yet. Patients will be followed for an additional year for structural or joint damage.
"Clinical meaningfulness is an intense discussion that we will have to conduct," he said. "I’m not quite sure of it myself yet."
The study was funded by F. Hoffmann-La Roche Ltd. Dr. Huizinga disclosed that he is a consultant for Abbott, Axis Shield Diagnostics, Biotest AG, Bristol-Myers Squibb, Crescendo Bioscience, Roche, Novartis, Schering-Plough, UCB, Wyeth, and Pfizer.
MADRID – Patients with early, active rheumatoid arthritis who took tocilizumab – either alone or in combination with methotrexate – continued to benefit from it by the end of a 2-year study.
About half of those on either treatment strategy in the study achieved remission by the end of the first year and this did not change appreciably by the end of the second year. There was also a very low rate of radiographic progression, Dr. Tom Huizinga said at the annual European Congress of Rheumatology.
The results confirm and extend the earlier findings of ACT-RAY, a 2-year, randomized, placebo-controlled study of tocilizumab employed as a switch or add-on therapy for patients with early, active rheumatoid arthritis (RA). The 24-week data, published earlier this year, showed that tocilizumab was just as effective without methotrexate as with it, suggesting that it could be employed as monotherapy (Ann. Rheum. Dis. 2013;72:43-50).
All 553 patients in ACT-RAY received open-label tocilizumab 8 mg/kg intravenously every 4 weeks. They were randomized to the switch strategy (tocilizumab 8 mg/kg IV every 4 weeks with oral placebo) or the add-on strategy (tocilizumab 8 mg/kg IV every 4 weeks plus 2.5 mg methotrexate), said Dr. Huizinga, head of the department of rheumatology at Leiden (The Netherlands) University Medical Center.
Most of the patients (81%) were women; mean age was 53 years. Patients had a mean disease duration of 8 years and a mean Disease Activity Score–28 (DAS28) of 6.4.
Most of the study group (433) completed the second year of treatment. Reasons for withdrawal included lack of efficacy (2% in the add-on strategy group and 5% in the switch strategy group) and adverse events (10% of add-on patients and 11% of switch). There were three deaths in the add-on group and six in the switch group.
Sustained remission was defined as a DAS28 of less than 2.6 at two consecutive visits separated by 12 weeks. By week 52, about 50% of the overall cohort (53% add-on strategy and 47% switch strategy) had achieved remission and were able to discontinue tocilizumab.
By week 104, 86% of the overall cohort had experienced a flare in disease activity, with a median time of 90 days from tocilizumab discontinuation. Most of those patients restarted tocilizumab. The medication continued to be effective. The mean DAS28 at flare was 4.46, dropping to a mean of 2.99 within 4 weeks of restarting treatment.
The mean DAS28 score at week 104 was unchanged from the score at week 52, decreasing by 3.6 from baseline in both groups. The large majority of each group experienced no radiographic progression during year 2 (94% of the add-on and 91% of the switch groups).
By the end of the study at week 104, 23% of the add-on group and 18% of the switch group were in remission as measured by the Clinical Disease Activity Index – virtually identical to CDAI remission rates at week 52.
The safety results were consistent with previous findings, Dr. Huizinga said. Serious adverse events and infections occurred in 15% of the add-on group and 4% of the switch group.
Liver enzyme elevations were more common among those in the add-on group. Elevations of up to three times the upper limit of normal of alanine aminotransferase occurred in 58% of the add-on group and 40% of the switch group. Elevations of up to five times the upper limit occurred in 13% of the add-on group and 5% of the switch group. Aspartate transaminase elevations of up to three times the upper limit of normal occurred in 51% of the add-on group and 30% of the switch group. Elevations of up to five times the upper limit of normal occurred in 5% of the add-on and 2% of the switch groups.
During a discussion period after his presentation, Dr. Huizinga addressed the issue of clinical impact, saying all of the information isn’t in yet. Patients will be followed for an additional year for structural or joint damage.
"Clinical meaningfulness is an intense discussion that we will have to conduct," he said. "I’m not quite sure of it myself yet."
The study was funded by F. Hoffmann-La Roche Ltd. Dr. Huizinga disclosed that he is a consultant for Abbott, Axis Shield Diagnostics, Biotest AG, Bristol-Myers Squibb, Crescendo Bioscience, Roche, Novartis, Schering-Plough, UCB, Wyeth, and Pfizer.
MADRID – Patients with early, active rheumatoid arthritis who took tocilizumab – either alone or in combination with methotrexate – continued to benefit from it by the end of a 2-year study.
About half of those on either treatment strategy in the study achieved remission by the end of the first year and this did not change appreciably by the end of the second year. There was also a very low rate of radiographic progression, Dr. Tom Huizinga said at the annual European Congress of Rheumatology.
The results confirm and extend the earlier findings of ACT-RAY, a 2-year, randomized, placebo-controlled study of tocilizumab employed as a switch or add-on therapy for patients with early, active rheumatoid arthritis (RA). The 24-week data, published earlier this year, showed that tocilizumab was just as effective without methotrexate as with it, suggesting that it could be employed as monotherapy (Ann. Rheum. Dis. 2013;72:43-50).
All 553 patients in ACT-RAY received open-label tocilizumab 8 mg/kg intravenously every 4 weeks. They were randomized to the switch strategy (tocilizumab 8 mg/kg IV every 4 weeks with oral placebo) or the add-on strategy (tocilizumab 8 mg/kg IV every 4 weeks plus 2.5 mg methotrexate), said Dr. Huizinga, head of the department of rheumatology at Leiden (The Netherlands) University Medical Center.
Most of the patients (81%) were women; mean age was 53 years. Patients had a mean disease duration of 8 years and a mean Disease Activity Score–28 (DAS28) of 6.4.
Most of the study group (433) completed the second year of treatment. Reasons for withdrawal included lack of efficacy (2% in the add-on strategy group and 5% in the switch strategy group) and adverse events (10% of add-on patients and 11% of switch). There were three deaths in the add-on group and six in the switch group.
Sustained remission was defined as a DAS28 of less than 2.6 at two consecutive visits separated by 12 weeks. By week 52, about 50% of the overall cohort (53% add-on strategy and 47% switch strategy) had achieved remission and were able to discontinue tocilizumab.
By week 104, 86% of the overall cohort had experienced a flare in disease activity, with a median time of 90 days from tocilizumab discontinuation. Most of those patients restarted tocilizumab. The medication continued to be effective. The mean DAS28 at flare was 4.46, dropping to a mean of 2.99 within 4 weeks of restarting treatment.
The mean DAS28 score at week 104 was unchanged from the score at week 52, decreasing by 3.6 from baseline in both groups. The large majority of each group experienced no radiographic progression during year 2 (94% of the add-on and 91% of the switch groups).
By the end of the study at week 104, 23% of the add-on group and 18% of the switch group were in remission as measured by the Clinical Disease Activity Index – virtually identical to CDAI remission rates at week 52.
The safety results were consistent with previous findings, Dr. Huizinga said. Serious adverse events and infections occurred in 15% of the add-on group and 4% of the switch group.
Liver enzyme elevations were more common among those in the add-on group. Elevations of up to three times the upper limit of normal of alanine aminotransferase occurred in 58% of the add-on group and 40% of the switch group. Elevations of up to five times the upper limit occurred in 13% of the add-on group and 5% of the switch group. Aspartate transaminase elevations of up to three times the upper limit of normal occurred in 51% of the add-on group and 30% of the switch group. Elevations of up to five times the upper limit of normal occurred in 5% of the add-on and 2% of the switch groups.
During a discussion period after his presentation, Dr. Huizinga addressed the issue of clinical impact, saying all of the information isn’t in yet. Patients will be followed for an additional year for structural or joint damage.
"Clinical meaningfulness is an intense discussion that we will have to conduct," he said. "I’m not quite sure of it myself yet."
The study was funded by F. Hoffmann-La Roche Ltd. Dr. Huizinga disclosed that he is a consultant for Abbott, Axis Shield Diagnostics, Biotest AG, Bristol-Myers Squibb, Crescendo Bioscience, Roche, Novartis, Schering-Plough, UCB, Wyeth, and Pfizer.
AT THE EULAR CONGRESS 2013
Major finding: By week 52, about 50% of the overall cohort (53% add-on strategy and 47% switch strategy) had achieved remission and were able to discontinue tocilizumab.
Data source: The ACT-RAY study randomized 553 patients to tocilizumab plus placebo or tocilizumab plus methotrexate.
Disclosures: The study was funded by F. Hoffmann-La Roche Ltd. Dr. Huizinga disclosed that he is a consultant for Abbott, Axis Shield Diagnostics, Biotest AG, Bristol-Myers Squibb, Crescendo Bioscience, Roche, Novartis, Schering-Plough, UCB, Wyeth, and Pfizer.
Remission elusive in overweight, obese RA patients
MADRID – Being overweight or obese makes it much more difficult for patients with early rheumatoid arthritis to achieve remission and significantly increases the chance that they will need a biologic by the end of the first treatment year.
The finding speaks to the role that weight can play in RA management, Dr. Elisa Gremese said during the annual European Congress of Rheumatology.
"Obesity is considered a systemic low-grade inflammatory state, and adipose tissue is a proinflammatory cytokine-producing factory," Dr. Gremese said during a press briefing. "Studies continue to show that obesity may be contributing to the rise in RA and is associated with a worse course of disease and more disability."
She examined the relationship between weight and first-year therapeutic outcomes among 346 patients with early rheumatoid arthritis; 33% of these had very early disease of less than 6 months’ duration. Normal weight was considered to be a body mass index of less than 25 kg/m2; overweight was a BMI of 25-30 kg/m2, and obesity was a BMI of greater than 30 kg/m2. About half of the patients (47%) were normal weight, 39% were overweight, and the remainder were obese, said Dr. Gremese of the Università Cattolica del Sacro Cuore in Rome.
All patients received up to 25 mg methotrexate each week, with or without steroids. In cases in which a patient didn’t achieve at least a "good" response (a change of at least 1.2 on the Disease Activity Score [DAS] 28), a tumor necrosis factor (TNF) blocker was added to the treatment regimen.
Overweight and obese patients had a significantly lower rate of remission as measured by both the DAS and the Clinical Disease Activity Index (CDAI) Score.
At 12 months, 49% of the normal weight group, 29% of the overweight group, and 34% of the obese group reached remission as measured by the DAS. Remission according to the CDAI criteria occurred in 50% of the normal weight group, 37% of the overweight group, and 31% of the obese group.
By the end of the first year, significantly more overweight and obese patients were taking an anti-TNF medication than were normal weight patients (29% and 28%, compared with 16%, respectively).
A multivariate analysis found that having a body mass index of at least 25 kg/m2 at baseline more than doubled the risk of DAS nonremission (odds ratio, 2.4) at 12 months, and nearly doubled the risk of CDAI nonremission (OR, 1.8).
"In the general population, the prevalence of overweight and obesity now reaches 50%," Dr. Gremese said. "We should look at this as a modifiable risk factor" that can be changed to help patients reach a better outcome.
She added that her group has started a dedicated outpatient center with a multidisciplinary program to help patients with RA and other rheumatic diseases with weight loss. "They work with a dietitian and psychologist as well as rheumatologist," she said.
Dr. Gremese had no financial declarations.
MADRID – Being overweight or obese makes it much more difficult for patients with early rheumatoid arthritis to achieve remission and significantly increases the chance that they will need a biologic by the end of the first treatment year.
The finding speaks to the role that weight can play in RA management, Dr. Elisa Gremese said during the annual European Congress of Rheumatology.
"Obesity is considered a systemic low-grade inflammatory state, and adipose tissue is a proinflammatory cytokine-producing factory," Dr. Gremese said during a press briefing. "Studies continue to show that obesity may be contributing to the rise in RA and is associated with a worse course of disease and more disability."
She examined the relationship between weight and first-year therapeutic outcomes among 346 patients with early rheumatoid arthritis; 33% of these had very early disease of less than 6 months’ duration. Normal weight was considered to be a body mass index of less than 25 kg/m2; overweight was a BMI of 25-30 kg/m2, and obesity was a BMI of greater than 30 kg/m2. About half of the patients (47%) were normal weight, 39% were overweight, and the remainder were obese, said Dr. Gremese of the Università Cattolica del Sacro Cuore in Rome.
All patients received up to 25 mg methotrexate each week, with or without steroids. In cases in which a patient didn’t achieve at least a "good" response (a change of at least 1.2 on the Disease Activity Score [DAS] 28), a tumor necrosis factor (TNF) blocker was added to the treatment regimen.
Overweight and obese patients had a significantly lower rate of remission as measured by both the DAS and the Clinical Disease Activity Index (CDAI) Score.
At 12 months, 49% of the normal weight group, 29% of the overweight group, and 34% of the obese group reached remission as measured by the DAS. Remission according to the CDAI criteria occurred in 50% of the normal weight group, 37% of the overweight group, and 31% of the obese group.
By the end of the first year, significantly more overweight and obese patients were taking an anti-TNF medication than were normal weight patients (29% and 28%, compared with 16%, respectively).
A multivariate analysis found that having a body mass index of at least 25 kg/m2 at baseline more than doubled the risk of DAS nonremission (odds ratio, 2.4) at 12 months, and nearly doubled the risk of CDAI nonremission (OR, 1.8).
"In the general population, the prevalence of overweight and obesity now reaches 50%," Dr. Gremese said. "We should look at this as a modifiable risk factor" that can be changed to help patients reach a better outcome.
She added that her group has started a dedicated outpatient center with a multidisciplinary program to help patients with RA and other rheumatic diseases with weight loss. "They work with a dietitian and psychologist as well as rheumatologist," she said.
Dr. Gremese had no financial declarations.
MADRID – Being overweight or obese makes it much more difficult for patients with early rheumatoid arthritis to achieve remission and significantly increases the chance that they will need a biologic by the end of the first treatment year.
The finding speaks to the role that weight can play in RA management, Dr. Elisa Gremese said during the annual European Congress of Rheumatology.
"Obesity is considered a systemic low-grade inflammatory state, and adipose tissue is a proinflammatory cytokine-producing factory," Dr. Gremese said during a press briefing. "Studies continue to show that obesity may be contributing to the rise in RA and is associated with a worse course of disease and more disability."
She examined the relationship between weight and first-year therapeutic outcomes among 346 patients with early rheumatoid arthritis; 33% of these had very early disease of less than 6 months’ duration. Normal weight was considered to be a body mass index of less than 25 kg/m2; overweight was a BMI of 25-30 kg/m2, and obesity was a BMI of greater than 30 kg/m2. About half of the patients (47%) were normal weight, 39% were overweight, and the remainder were obese, said Dr. Gremese of the Università Cattolica del Sacro Cuore in Rome.
All patients received up to 25 mg methotrexate each week, with or without steroids. In cases in which a patient didn’t achieve at least a "good" response (a change of at least 1.2 on the Disease Activity Score [DAS] 28), a tumor necrosis factor (TNF) blocker was added to the treatment regimen.
Overweight and obese patients had a significantly lower rate of remission as measured by both the DAS and the Clinical Disease Activity Index (CDAI) Score.
At 12 months, 49% of the normal weight group, 29% of the overweight group, and 34% of the obese group reached remission as measured by the DAS. Remission according to the CDAI criteria occurred in 50% of the normal weight group, 37% of the overweight group, and 31% of the obese group.
By the end of the first year, significantly more overweight and obese patients were taking an anti-TNF medication than were normal weight patients (29% and 28%, compared with 16%, respectively).
A multivariate analysis found that having a body mass index of at least 25 kg/m2 at baseline more than doubled the risk of DAS nonremission (odds ratio, 2.4) at 12 months, and nearly doubled the risk of CDAI nonremission (OR, 1.8).
"In the general population, the prevalence of overweight and obesity now reaches 50%," Dr. Gremese said. "We should look at this as a modifiable risk factor" that can be changed to help patients reach a better outcome.
She added that her group has started a dedicated outpatient center with a multidisciplinary program to help patients with RA and other rheumatic diseases with weight loss. "They work with a dietitian and psychologist as well as rheumatologist," she said.
Dr. Gremese had no financial declarations.
AT THE EULAR CONGRESS 2013
Major finding: After 1 year of treatment, remission occurred in 49% of early RA patients of normal weight, 29% of the overweight group, and 34% of the obese group.
Data source: The prospective study comprised 346 patients.
Disclosures: Dr. Gremese had no financial disclosures.
More than half of eligible girls don't get first HPV vaccine
WASHINGTON – Only about 40% of girls old enough to receive the HPV vaccine got the first dose during a year in which they were seen at least once in an urban community clinic, a study has shown.
The large cohort study also found that 53% of girls underwent a sexual health screening before initiating the HPV series, suggesting that many are already sexually active but unprotected, Dr. Annnika Hofstetter said at the annual meeting of the Pediatric Academic Societies.
The finding points out the importance of early education about HPV and sexual safety for both girls and parents, said Dr. Hofstetter of New York–Presbyterian Hospital.
"Even in settings with high HPV vaccine initiation rates, the series is often delayed," she said. "Promotion of early HPV vaccination consistent with [federal] guidelines is crucial."
Dr. Hofstetter examined the timing of the initial HPV vaccination in relation to age and sexual activity in 23,900 females aged 11-26 years. All had been seen at least once in any of 16 large urban health care clinics (family planning, ob.gyn., school-based health, pediatric, and family medicine). She divided the cohort into two subgroups: an age analysis of 10,489 who were 11-26 years old, and a sexual health screening analysis of 15,049 who were aged 11-19 years. The two subgroups add up to more that the total because there was overlap between them. The sexual health screening cohort included only girls who were getting the Pap smear and chlamydia test because they were considered to be sexually active.
Most of the cohort (78%) was Hispanic; 18% were black, and the rest were non-Hispanic whites. Most (72%) were also publicly insured; 23% had private insurance, and 5% had no health care coverage at all. Because all were at least 11 years old, all were eligible to receive the vaccine. If the vaccine wasn’t given during a visit, it was considered a "missed opportunity."
The first analysis included girls aged 11-26 years. It examined the age of first HPV vaccination in each year from 2007 to 2011. The percentage of girls who had at least the initial vaccination rose significantly over the study period, from about 35% in 2007 to 69% in 2011.
Vaccination rates were highest in the girls aged 11-12 and 13-17 years, but only about 40% of each group were vaccinated in each of the study years – leaving the other 60% unvaccinated.
When she compared the number of clinic visits with each girl’s immunization status, Dr. Hofstetter found a median of two to three missed vaccination opportunities for every year. About half of those who did get vaccinated had at least one missed opportunity before they got the first dose.
The second analysis examined sexual health screening – chlamydia testing and Pap smear – in relationship to vaccination timing. The tests were considered a proxy for sexual activity. Girls in this group were aged 11-19 years and had not yet been vaccinated.
More than half of this group (54%) had either a Pap smear (17%) or chlamydia test (37%). Of the chlamydia screens, 30% were negative and 7% were positive. Of the Pap smears, 11% were normal and 7% were abnormal.
During the discussion, a physician from Sweden said that some preschoolers in the country have been offered the series. "Giving it that early removes this entire question of prevaccination sexual activity," he said.
The study was partially funded by Merck. Dr. Hofstetter said she had no relevant financial disclosures.
WASHINGTON – Only about 40% of girls old enough to receive the HPV vaccine got the first dose during a year in which they were seen at least once in an urban community clinic, a study has shown.
The large cohort study also found that 53% of girls underwent a sexual health screening before initiating the HPV series, suggesting that many are already sexually active but unprotected, Dr. Annnika Hofstetter said at the annual meeting of the Pediatric Academic Societies.
The finding points out the importance of early education about HPV and sexual safety for both girls and parents, said Dr. Hofstetter of New York–Presbyterian Hospital.
"Even in settings with high HPV vaccine initiation rates, the series is often delayed," she said. "Promotion of early HPV vaccination consistent with [federal] guidelines is crucial."
Dr. Hofstetter examined the timing of the initial HPV vaccination in relation to age and sexual activity in 23,900 females aged 11-26 years. All had been seen at least once in any of 16 large urban health care clinics (family planning, ob.gyn., school-based health, pediatric, and family medicine). She divided the cohort into two subgroups: an age analysis of 10,489 who were 11-26 years old, and a sexual health screening analysis of 15,049 who were aged 11-19 years. The two subgroups add up to more that the total because there was overlap between them. The sexual health screening cohort included only girls who were getting the Pap smear and chlamydia test because they were considered to be sexually active.
Most of the cohort (78%) was Hispanic; 18% were black, and the rest were non-Hispanic whites. Most (72%) were also publicly insured; 23% had private insurance, and 5% had no health care coverage at all. Because all were at least 11 years old, all were eligible to receive the vaccine. If the vaccine wasn’t given during a visit, it was considered a "missed opportunity."
The first analysis included girls aged 11-26 years. It examined the age of first HPV vaccination in each year from 2007 to 2011. The percentage of girls who had at least the initial vaccination rose significantly over the study period, from about 35% in 2007 to 69% in 2011.
Vaccination rates were highest in the girls aged 11-12 and 13-17 years, but only about 40% of each group were vaccinated in each of the study years – leaving the other 60% unvaccinated.
When she compared the number of clinic visits with each girl’s immunization status, Dr. Hofstetter found a median of two to three missed vaccination opportunities for every year. About half of those who did get vaccinated had at least one missed opportunity before they got the first dose.
The second analysis examined sexual health screening – chlamydia testing and Pap smear – in relationship to vaccination timing. The tests were considered a proxy for sexual activity. Girls in this group were aged 11-19 years and had not yet been vaccinated.
More than half of this group (54%) had either a Pap smear (17%) or chlamydia test (37%). Of the chlamydia screens, 30% were negative and 7% were positive. Of the Pap smears, 11% were normal and 7% were abnormal.
During the discussion, a physician from Sweden said that some preschoolers in the country have been offered the series. "Giving it that early removes this entire question of prevaccination sexual activity," he said.
The study was partially funded by Merck. Dr. Hofstetter said she had no relevant financial disclosures.
WASHINGTON – Only about 40% of girls old enough to receive the HPV vaccine got the first dose during a year in which they were seen at least once in an urban community clinic, a study has shown.
The large cohort study also found that 53% of girls underwent a sexual health screening before initiating the HPV series, suggesting that many are already sexually active but unprotected, Dr. Annnika Hofstetter said at the annual meeting of the Pediatric Academic Societies.
The finding points out the importance of early education about HPV and sexual safety for both girls and parents, said Dr. Hofstetter of New York–Presbyterian Hospital.
"Even in settings with high HPV vaccine initiation rates, the series is often delayed," she said. "Promotion of early HPV vaccination consistent with [federal] guidelines is crucial."
Dr. Hofstetter examined the timing of the initial HPV vaccination in relation to age and sexual activity in 23,900 females aged 11-26 years. All had been seen at least once in any of 16 large urban health care clinics (family planning, ob.gyn., school-based health, pediatric, and family medicine). She divided the cohort into two subgroups: an age analysis of 10,489 who were 11-26 years old, and a sexual health screening analysis of 15,049 who were aged 11-19 years. The two subgroups add up to more that the total because there was overlap between them. The sexual health screening cohort included only girls who were getting the Pap smear and chlamydia test because they were considered to be sexually active.
Most of the cohort (78%) was Hispanic; 18% were black, and the rest were non-Hispanic whites. Most (72%) were also publicly insured; 23% had private insurance, and 5% had no health care coverage at all. Because all were at least 11 years old, all were eligible to receive the vaccine. If the vaccine wasn’t given during a visit, it was considered a "missed opportunity."
The first analysis included girls aged 11-26 years. It examined the age of first HPV vaccination in each year from 2007 to 2011. The percentage of girls who had at least the initial vaccination rose significantly over the study period, from about 35% in 2007 to 69% in 2011.
Vaccination rates were highest in the girls aged 11-12 and 13-17 years, but only about 40% of each group were vaccinated in each of the study years – leaving the other 60% unvaccinated.
When she compared the number of clinic visits with each girl’s immunization status, Dr. Hofstetter found a median of two to three missed vaccination opportunities for every year. About half of those who did get vaccinated had at least one missed opportunity before they got the first dose.
The second analysis examined sexual health screening – chlamydia testing and Pap smear – in relationship to vaccination timing. The tests were considered a proxy for sexual activity. Girls in this group were aged 11-19 years and had not yet been vaccinated.
More than half of this group (54%) had either a Pap smear (17%) or chlamydia test (37%). Of the chlamydia screens, 30% were negative and 7% were positive. Of the Pap smears, 11% were normal and 7% were abnormal.
During the discussion, a physician from Sweden said that some preschoolers in the country have been offered the series. "Giving it that early removes this entire question of prevaccination sexual activity," he said.
The study was partially funded by Merck. Dr. Hofstetter said she had no relevant financial disclosures.
AT THE PAS ANNUAL MEETING
Major finding: A comparison of the number of clinic visits with each girl’s HPV immunization status found a median of two to three missed vaccination opportunities for every year.
Data source: Two cohorts totaling 23,900 girls and women aged 11-26 years.
Disclosures: The study was partially funded by Merck. Dr. Hofstetter said she had no relevant financial disclosures.
Monotherapy as good as combo for kids with pneumonia
WASHINGTON – Beta-lactam monotherapy is likely to be just as effective as a combination of beta-lactam and macrolide therapy for the majority of children hospitalized with pneumonia, a study has shown.
A subanalysis of a large national study found no difference in the length of hospital stay for children with pneumonia, whether they had received ceftriaxone alone or in combination with azithromycin, Dr. Derek J. Williams said at the annual meeting of the Pediatric Academic Societies.
"This is an important finding, because we know that limiting antimicrobial use to the narrowest-spectrum agent for the shortest duration is imperative to slow the epidemic of antimicrobial resistance," said Dr. Williams, a pediatrician in the division of hospital medicine at Vanderbilt University, Nashville, Tenn.
His large retrospective study used data collected in the Etiology of Pneumonia in the Community (EPIC) study. Sponsored by the Centers for Disease Control and Prevention, EPIC now contains information on 1,496 children.
Dr. Williams’ study involved 941 children who were admitted to hospitals with presumed community-acquired pneumonia. Most of these (678) were treated with ceftriaxone alone; 263 received combination therapy with ceftriaxone and azithromycin.
There were some important baseline differences between the groups, Dr. Williams noted. Children in the combination group were significantly older than those in the monotherapy* group (median, 64 months vs. 19 months). They were also significantly more likely to have asthma (44% vs. 35%) or other high-risk comorbidities including cardiopulmonary, oncologic, or immunosuppressive conditions; hepatic or renal problems; metabolic or genetic issues; and neurodevelopmental disorders.
The median length of hospital stay was 64 hours in both groups. Neither an unadjusted nor adjusted analysis found any significant relationship between therapy type and length of stay.
A propensity score matching looked at the outcome in 190 children in each exposure group. It too found no significant relationships.
The results are in line with a national pediatric pneumonia treatment guideline, published in 2011 in Clinical Infectious Diseases (53:617-30),Dr. Williams said.
During discussion, however, he noted that the conclusions may be somewhat marred by the nature of the EPIC cohort. "Although more than 80% of EPIC children received antibiotics, a bacterial pathogen was identified in only 15%."
This prompted some comment from pediatric hospitalist Mark W. Shen of the Dell Children’s Medical Center of Central Texas, Austin.
"The EPIC study is probably one of our most robust studies looking at the etiology of community-acquired pneumonia in hospitalized kids," he said. "But it seems now that you could argue that potentially more than 85% of these could have a viral etiology for their disease. So that would make it exceedingly difficult to find any inherent differences in antibiotic treatment. Is there any gold standard for figuring out how we can assess the comparative effectiveness, if most of these cases could be viral?
"This is the big issue that we struggle with," said Dr. Williams. "How do we define bacterial pneumonia? Our methods for detecting viruses are far superior to those we use to detect bacteria. I think this is coming with biomarkers, but we are not there yet. I think we’re underestimating bacterial disease and coinfections, but I also think that retrospective data are just not as good as prospective. Until we are able to really define this, that’s going to be a difficult question to answer."
Dr. Shen was also an author on a poster at the meeting. His study found that the hospitalist changed or stopped antibiotics in 93% of children with pneumonia who were started on one during the emergency department visit. "Sixty percent of the time, we found that there was no difference in their outcomes," he said.
The EPIC study is funded by the Centers for Disease Control and Prevention. Dr. Williams said he had no relevant financial disclosures. Dr. Shen was listed in the program as having no financial disclosures.
*Correction, 6/26/13: An earlier version of this story misstated the type of therapy group in the description of the study.
WASHINGTON – Beta-lactam monotherapy is likely to be just as effective as a combination of beta-lactam and macrolide therapy for the majority of children hospitalized with pneumonia, a study has shown.
A subanalysis of a large national study found no difference in the length of hospital stay for children with pneumonia, whether they had received ceftriaxone alone or in combination with azithromycin, Dr. Derek J. Williams said at the annual meeting of the Pediatric Academic Societies.
"This is an important finding, because we know that limiting antimicrobial use to the narrowest-spectrum agent for the shortest duration is imperative to slow the epidemic of antimicrobial resistance," said Dr. Williams, a pediatrician in the division of hospital medicine at Vanderbilt University, Nashville, Tenn.
His large retrospective study used data collected in the Etiology of Pneumonia in the Community (EPIC) study. Sponsored by the Centers for Disease Control and Prevention, EPIC now contains information on 1,496 children.
Dr. Williams’ study involved 941 children who were admitted to hospitals with presumed community-acquired pneumonia. Most of these (678) were treated with ceftriaxone alone; 263 received combination therapy with ceftriaxone and azithromycin.
There were some important baseline differences between the groups, Dr. Williams noted. Children in the combination group were significantly older than those in the monotherapy* group (median, 64 months vs. 19 months). They were also significantly more likely to have asthma (44% vs. 35%) or other high-risk comorbidities including cardiopulmonary, oncologic, or immunosuppressive conditions; hepatic or renal problems; metabolic or genetic issues; and neurodevelopmental disorders.
The median length of hospital stay was 64 hours in both groups. Neither an unadjusted nor adjusted analysis found any significant relationship between therapy type and length of stay.
A propensity score matching looked at the outcome in 190 children in each exposure group. It too found no significant relationships.
The results are in line with a national pediatric pneumonia treatment guideline, published in 2011 in Clinical Infectious Diseases (53:617-30),Dr. Williams said.
During discussion, however, he noted that the conclusions may be somewhat marred by the nature of the EPIC cohort. "Although more than 80% of EPIC children received antibiotics, a bacterial pathogen was identified in only 15%."
This prompted some comment from pediatric hospitalist Mark W. Shen of the Dell Children’s Medical Center of Central Texas, Austin.
"The EPIC study is probably one of our most robust studies looking at the etiology of community-acquired pneumonia in hospitalized kids," he said. "But it seems now that you could argue that potentially more than 85% of these could have a viral etiology for their disease. So that would make it exceedingly difficult to find any inherent differences in antibiotic treatment. Is there any gold standard for figuring out how we can assess the comparative effectiveness, if most of these cases could be viral?
"This is the big issue that we struggle with," said Dr. Williams. "How do we define bacterial pneumonia? Our methods for detecting viruses are far superior to those we use to detect bacteria. I think this is coming with biomarkers, but we are not there yet. I think we’re underestimating bacterial disease and coinfections, but I also think that retrospective data are just not as good as prospective. Until we are able to really define this, that’s going to be a difficult question to answer."
Dr. Shen was also an author on a poster at the meeting. His study found that the hospitalist changed or stopped antibiotics in 93% of children with pneumonia who were started on one during the emergency department visit. "Sixty percent of the time, we found that there was no difference in their outcomes," he said.
The EPIC study is funded by the Centers for Disease Control and Prevention. Dr. Williams said he had no relevant financial disclosures. Dr. Shen was listed in the program as having no financial disclosures.
*Correction, 6/26/13: An earlier version of this story misstated the type of therapy group in the description of the study.
WASHINGTON – Beta-lactam monotherapy is likely to be just as effective as a combination of beta-lactam and macrolide therapy for the majority of children hospitalized with pneumonia, a study has shown.
A subanalysis of a large national study found no difference in the length of hospital stay for children with pneumonia, whether they had received ceftriaxone alone or in combination with azithromycin, Dr. Derek J. Williams said at the annual meeting of the Pediatric Academic Societies.
"This is an important finding, because we know that limiting antimicrobial use to the narrowest-spectrum agent for the shortest duration is imperative to slow the epidemic of antimicrobial resistance," said Dr. Williams, a pediatrician in the division of hospital medicine at Vanderbilt University, Nashville, Tenn.
His large retrospective study used data collected in the Etiology of Pneumonia in the Community (EPIC) study. Sponsored by the Centers for Disease Control and Prevention, EPIC now contains information on 1,496 children.
Dr. Williams’ study involved 941 children who were admitted to hospitals with presumed community-acquired pneumonia. Most of these (678) were treated with ceftriaxone alone; 263 received combination therapy with ceftriaxone and azithromycin.
There were some important baseline differences between the groups, Dr. Williams noted. Children in the combination group were significantly older than those in the monotherapy* group (median, 64 months vs. 19 months). They were also significantly more likely to have asthma (44% vs. 35%) or other high-risk comorbidities including cardiopulmonary, oncologic, or immunosuppressive conditions; hepatic or renal problems; metabolic or genetic issues; and neurodevelopmental disorders.
The median length of hospital stay was 64 hours in both groups. Neither an unadjusted nor adjusted analysis found any significant relationship between therapy type and length of stay.
A propensity score matching looked at the outcome in 190 children in each exposure group. It too found no significant relationships.
The results are in line with a national pediatric pneumonia treatment guideline, published in 2011 in Clinical Infectious Diseases (53:617-30),Dr. Williams said.
During discussion, however, he noted that the conclusions may be somewhat marred by the nature of the EPIC cohort. "Although more than 80% of EPIC children received antibiotics, a bacterial pathogen was identified in only 15%."
This prompted some comment from pediatric hospitalist Mark W. Shen of the Dell Children’s Medical Center of Central Texas, Austin.
"The EPIC study is probably one of our most robust studies looking at the etiology of community-acquired pneumonia in hospitalized kids," he said. "But it seems now that you could argue that potentially more than 85% of these could have a viral etiology for their disease. So that would make it exceedingly difficult to find any inherent differences in antibiotic treatment. Is there any gold standard for figuring out how we can assess the comparative effectiveness, if most of these cases could be viral?
"This is the big issue that we struggle with," said Dr. Williams. "How do we define bacterial pneumonia? Our methods for detecting viruses are far superior to those we use to detect bacteria. I think this is coming with biomarkers, but we are not there yet. I think we’re underestimating bacterial disease and coinfections, but I also think that retrospective data are just not as good as prospective. Until we are able to really define this, that’s going to be a difficult question to answer."
Dr. Shen was also an author on a poster at the meeting. His study found that the hospitalist changed or stopped antibiotics in 93% of children with pneumonia who were started on one during the emergency department visit. "Sixty percent of the time, we found that there was no difference in their outcomes," he said.
The EPIC study is funded by the Centers for Disease Control and Prevention. Dr. Williams said he had no relevant financial disclosures. Dr. Shen was listed in the program as having no financial disclosures.
*Correction, 6/26/13: An earlier version of this story misstated the type of therapy group in the description of the study.
AT THE PAS ANNUAL MEETING
Major finding: Whether treated with ceftriaxone alone or with a combination of ceftriaxone and azithromycin, children with community-acquired pneumonia stayed in the hospital a median of 64 hours.
Data source: A subanalysis involving 941 children in the EPIC study.
Disclosures: The EPIC study is funded by the Centers for Disease Control and Prevention. Dr. Williams said he had no relevant financial disclosures. Dr. Shen was listed in the program as having no financial disclosures.
Heart failure guidelines: New hope in medical therapy
The newest heart failure management guidelines make a bold statement: Heart failure should no longer be considered a death sentence, but can instead be managed in a way that may add years of quality life for some patients.
“With optimal therapy applied to the right patient in the right manner at the right time, the risk of death can be markedly reduced, perhaps by as much as 50%. Treating fewer than 10 patients with all the correct therapies will easily save at least one life and one or more hospitalizations. Those are real benefits that dwarf the benefit of many of our other cardiovascular therapies,” said Dr. Clyde W. Yancy, chair of the joint guidelines writing committee. The document was published in the June 5 online edition of the Journal of the American College of Cardiology (2013 [doi:10.1016/j.jacc.2013.05.019]).
"For so long, we had assumed that, by definition, heart failure was a fatal diagnosis – that all we could do was tell patients to get their affairs in order and perhaps make them feel a little better, but that death was almost a fait accompli," said Dr. Yancy, the Magerstadt Professor of Medicine and chief of cardiology at Northwestern University, Chicago. "But, in the past few years, a lot of tough work has been done showing there are effective therapies and that when given correctly, major improvements in survival do occur."
A joint effort of the American College of Cardiology Foundation and the American Heart Association, the 2013 Heart Failure Guidelines represent the first update on the topic since 2009, Dr. Yancy said in an interview. Although the years between the documents are few, the strides in research have been many, he said.
"The emergence of new and important datasets generated the impetus for the 2013 guideline not as an update, but as a complete rewrite. All of the previously extant clinical practice guideline statements were subject to reanalysis, a change in level of evidence and most importantly, a change in the class of recommendation," he said.
The document is among the first in the United States to employ the concept of guideline-directed medical therapy (GDMT) – a new designation that allows clinicians to easily determine the best course of heart failure care for an individual patient. Schematic algorithms provide easy-to-follow treatment pathways that should be helpful for anyone who treats heart failure patients, from specialist to primary care provider, said Dr. Yancy.
A major focus of the guideline is treating heart failure with preserved ejection fraction (HFpEF), with the goal of preventing or delaying progression. HFpEF is "a real entity" that constitutes about half of heart failure diagnoses, Dr. Yancy said, but as yet, has no specific intervention.
Until research provides further answers, the best way to manage HFpEF is holistically. "About 90% of these patients have comorbid conditions like hypertension, coronary artery disease, diabetes, renal insufficiency and atrial fibrillation. In the absence of a specific intervention for HFpEF, focusing on these other conditions will provide us the opportunity to modify the natural history of this disease."
Dr. David E. Lanfear, a cardiologist specializing in advanced heart failure and transplantation at Henry Ford Hospital in Detroit, said the guidelines on HFpEF “are very reasonable. The recommendations appear similar to those in previous statements, on blood pressure control, volume control for symptoms, atrial fibrillation, and ischemia, without endorsing specific medications. The statement also eloquently points out the ways in which HFpEF represents a huge gap in the knowledge base.
The guidelines contain "critical" new indications for the use of aldosterone antagonists, Dr. Yancy said. The drugs saw a surge in use in the early 2000s, but the rush to embrace them brought challenges as well. "Some of the applications led to missteps resulting in elevated potassium levels and emergency admissions," Dr. Yancy said. Since then, additional trials have pinpointed the best ways to use aldosterone agonists in patients who have heart failure with reduced ejection fraction or cardiac injury after heart attack. Data now confirm their benefit in patients with mild and moderate disease, as well as those with more advanced disease.
"This is the first document in the United States to embrace the benefit of aldosterone antagonists for these patients," Dr. Yancy said. Provided that patient renal function is "reasonably intact," the drugs are a valuable addition to GDMT.
The guidelines also offer a refinement of the recommendations for cardiac resynchronization device therapy – another change supported by the results of recent, large-scale trials. "We now have three separate, well-done trials that suggest a significant benefit of cardiac resynchronization in patients with mild to moderate disease," Dr. Yancy said.
In addition to recommending the treatment for patients with mild to moderate disease, the guideline targets it more specifically. "We gave the greatest strength of recommendation for patients with a wide QRS interval and left bundle branch block, a modest recommendation for patients with a less wide interval, and an equivocal recommendation for those without left bundle branch block. We think this will allow better discrimination of those who are most likely to benefit from CRT from those unlikely to benefit."
There are also more plentiful data in favor of mechanical circulatory support for patients with advanced heart failure. "This is no longer a proof of concept strategy," Dr. Yancy said. "Left ventricular assist devices for advanced chronic heart failure represent an important component of a contemporary treatment algorithm for heart failure."
The guideline even reaches past the mechanics of heart failure into its possible genetic origins. "We’ve discovered that idiopathic dilated cardiomyopathy may not really be idiopathic, but instead related to genetic abnormality. We’ve coalesced observations and data from this emerging field to come up with recommendations about when we might consider genetic testing in patients and screening in family members. It’s something new, and we’re delighted that it’s presented in this document."
The guidelines also offer a new outlook on reducing heart failure readmissions – a problem that comes with a $25 billion/year price tag, Dr. Yancy said. Four simple, low-tech interventions stood out as practical and effective:
• Using in-hospital systems that identify heart failure patients appropriate for GDMT and prompt physicians to advance this care and assess response.
• Developing transitional care and discharge planning that emphasizes patient education to increase treatment compliance, manage comorbid conditions effectively, and tackle psychosocial barriers to care.
• Harnessing the cooperative power of a nurse-led multidisciplinary heart failure disease management program.
• Following up every patient with a phone call within 3 days of discharge and a physician appointment within 2 weeks.
"The beauty of this is that while everyone is looking for the silver bullet to decrease readmission – including high-tech interventions like device implantation and home electronic monitoring – we believe that these simple, straightforward, evidence-based approaches will work."
Finally, Dr. Yancy said, document places great importance on patient-centric outcomes like quality-of-life issues, shared decision-making, care coordination, and palliative care. Over the past decade, the physician/patient relationship has changed from almost paternalistic to an active partnership. "We need to include the patient’s point of view in this whole process. We need to put a greater emphasis on quality of life, and we need not fear a discussion on quality of death."
Dr. Yancy expressed a firm belief that integrating the guidelines into daily practice could have an enormous impact on the way heart failure patients are treated.
"We have come so far in our understanding and ability to treat these patients. These are dramatically effective interventions. We can shift the culture to the belief that heart failure is something that we can treat – to the idea that you can help your patients feel better and live longer. If we use this correctly, we can make a difference."
Dr. Yancy had no financial declarations.
The newest heart failure management guidelines make a bold statement: Heart failure should no longer be considered a death sentence, but can instead be managed in a way that may add years of quality life for some patients.
“With optimal therapy applied to the right patient in the right manner at the right time, the risk of death can be markedly reduced, perhaps by as much as 50%. Treating fewer than 10 patients with all the correct therapies will easily save at least one life and one or more hospitalizations. Those are real benefits that dwarf the benefit of many of our other cardiovascular therapies,” said Dr. Clyde W. Yancy, chair of the joint guidelines writing committee. The document was published in the June 5 online edition of the Journal of the American College of Cardiology (2013 [doi:10.1016/j.jacc.2013.05.019]).
"For so long, we had assumed that, by definition, heart failure was a fatal diagnosis – that all we could do was tell patients to get their affairs in order and perhaps make them feel a little better, but that death was almost a fait accompli," said Dr. Yancy, the Magerstadt Professor of Medicine and chief of cardiology at Northwestern University, Chicago. "But, in the past few years, a lot of tough work has been done showing there are effective therapies and that when given correctly, major improvements in survival do occur."
A joint effort of the American College of Cardiology Foundation and the American Heart Association, the 2013 Heart Failure Guidelines represent the first update on the topic since 2009, Dr. Yancy said in an interview. Although the years between the documents are few, the strides in research have been many, he said.
"The emergence of new and important datasets generated the impetus for the 2013 guideline not as an update, but as a complete rewrite. All of the previously extant clinical practice guideline statements were subject to reanalysis, a change in level of evidence and most importantly, a change in the class of recommendation," he said.
The document is among the first in the United States to employ the concept of guideline-directed medical therapy (GDMT) – a new designation that allows clinicians to easily determine the best course of heart failure care for an individual patient. Schematic algorithms provide easy-to-follow treatment pathways that should be helpful for anyone who treats heart failure patients, from specialist to primary care provider, said Dr. Yancy.
A major focus of the guideline is treating heart failure with preserved ejection fraction (HFpEF), with the goal of preventing or delaying progression. HFpEF is "a real entity" that constitutes about half of heart failure diagnoses, Dr. Yancy said, but as yet, has no specific intervention.
Until research provides further answers, the best way to manage HFpEF is holistically. "About 90% of these patients have comorbid conditions like hypertension, coronary artery disease, diabetes, renal insufficiency and atrial fibrillation. In the absence of a specific intervention for HFpEF, focusing on these other conditions will provide us the opportunity to modify the natural history of this disease."
Dr. David E. Lanfear, a cardiologist specializing in advanced heart failure and transplantation at Henry Ford Hospital in Detroit, said the guidelines on HFpEF “are very reasonable. The recommendations appear similar to those in previous statements, on blood pressure control, volume control for symptoms, atrial fibrillation, and ischemia, without endorsing specific medications. The statement also eloquently points out the ways in which HFpEF represents a huge gap in the knowledge base.
The guidelines contain "critical" new indications for the use of aldosterone antagonists, Dr. Yancy said. The drugs saw a surge in use in the early 2000s, but the rush to embrace them brought challenges as well. "Some of the applications led to missteps resulting in elevated potassium levels and emergency admissions," Dr. Yancy said. Since then, additional trials have pinpointed the best ways to use aldosterone agonists in patients who have heart failure with reduced ejection fraction or cardiac injury after heart attack. Data now confirm their benefit in patients with mild and moderate disease, as well as those with more advanced disease.
"This is the first document in the United States to embrace the benefit of aldosterone antagonists for these patients," Dr. Yancy said. Provided that patient renal function is "reasonably intact," the drugs are a valuable addition to GDMT.
The guidelines also offer a refinement of the recommendations for cardiac resynchronization device therapy – another change supported by the results of recent, large-scale trials. "We now have three separate, well-done trials that suggest a significant benefit of cardiac resynchronization in patients with mild to moderate disease," Dr. Yancy said.
In addition to recommending the treatment for patients with mild to moderate disease, the guideline targets it more specifically. "We gave the greatest strength of recommendation for patients with a wide QRS interval and left bundle branch block, a modest recommendation for patients with a less wide interval, and an equivocal recommendation for those without left bundle branch block. We think this will allow better discrimination of those who are most likely to benefit from CRT from those unlikely to benefit."
There are also more plentiful data in favor of mechanical circulatory support for patients with advanced heart failure. "This is no longer a proof of concept strategy," Dr. Yancy said. "Left ventricular assist devices for advanced chronic heart failure represent an important component of a contemporary treatment algorithm for heart failure."
The guideline even reaches past the mechanics of heart failure into its possible genetic origins. "We’ve discovered that idiopathic dilated cardiomyopathy may not really be idiopathic, but instead related to genetic abnormality. We’ve coalesced observations and data from this emerging field to come up with recommendations about when we might consider genetic testing in patients and screening in family members. It’s something new, and we’re delighted that it’s presented in this document."
The guidelines also offer a new outlook on reducing heart failure readmissions – a problem that comes with a $25 billion/year price tag, Dr. Yancy said. Four simple, low-tech interventions stood out as practical and effective:
• Using in-hospital systems that identify heart failure patients appropriate for GDMT and prompt physicians to advance this care and assess response.
• Developing transitional care and discharge planning that emphasizes patient education to increase treatment compliance, manage comorbid conditions effectively, and tackle psychosocial barriers to care.
• Harnessing the cooperative power of a nurse-led multidisciplinary heart failure disease management program.
• Following up every patient with a phone call within 3 days of discharge and a physician appointment within 2 weeks.
"The beauty of this is that while everyone is looking for the silver bullet to decrease readmission – including high-tech interventions like device implantation and home electronic monitoring – we believe that these simple, straightforward, evidence-based approaches will work."
Finally, Dr. Yancy said, document places great importance on patient-centric outcomes like quality-of-life issues, shared decision-making, care coordination, and palliative care. Over the past decade, the physician/patient relationship has changed from almost paternalistic to an active partnership. "We need to include the patient’s point of view in this whole process. We need to put a greater emphasis on quality of life, and we need not fear a discussion on quality of death."
Dr. Yancy expressed a firm belief that integrating the guidelines into daily practice could have an enormous impact on the way heart failure patients are treated.
"We have come so far in our understanding and ability to treat these patients. These are dramatically effective interventions. We can shift the culture to the belief that heart failure is something that we can treat – to the idea that you can help your patients feel better and live longer. If we use this correctly, we can make a difference."
Dr. Yancy had no financial declarations.
The newest heart failure management guidelines make a bold statement: Heart failure should no longer be considered a death sentence, but can instead be managed in a way that may add years of quality life for some patients.
“With optimal therapy applied to the right patient in the right manner at the right time, the risk of death can be markedly reduced, perhaps by as much as 50%. Treating fewer than 10 patients with all the correct therapies will easily save at least one life and one or more hospitalizations. Those are real benefits that dwarf the benefit of many of our other cardiovascular therapies,” said Dr. Clyde W. Yancy, chair of the joint guidelines writing committee. The document was published in the June 5 online edition of the Journal of the American College of Cardiology (2013 [doi:10.1016/j.jacc.2013.05.019]).
"For so long, we had assumed that, by definition, heart failure was a fatal diagnosis – that all we could do was tell patients to get their affairs in order and perhaps make them feel a little better, but that death was almost a fait accompli," said Dr. Yancy, the Magerstadt Professor of Medicine and chief of cardiology at Northwestern University, Chicago. "But, in the past few years, a lot of tough work has been done showing there are effective therapies and that when given correctly, major improvements in survival do occur."
A joint effort of the American College of Cardiology Foundation and the American Heart Association, the 2013 Heart Failure Guidelines represent the first update on the topic since 2009, Dr. Yancy said in an interview. Although the years between the documents are few, the strides in research have been many, he said.
"The emergence of new and important datasets generated the impetus for the 2013 guideline not as an update, but as a complete rewrite. All of the previously extant clinical practice guideline statements were subject to reanalysis, a change in level of evidence and most importantly, a change in the class of recommendation," he said.
The document is among the first in the United States to employ the concept of guideline-directed medical therapy (GDMT) – a new designation that allows clinicians to easily determine the best course of heart failure care for an individual patient. Schematic algorithms provide easy-to-follow treatment pathways that should be helpful for anyone who treats heart failure patients, from specialist to primary care provider, said Dr. Yancy.
A major focus of the guideline is treating heart failure with preserved ejection fraction (HFpEF), with the goal of preventing or delaying progression. HFpEF is "a real entity" that constitutes about half of heart failure diagnoses, Dr. Yancy said, but as yet, has no specific intervention.
Until research provides further answers, the best way to manage HFpEF is holistically. "About 90% of these patients have comorbid conditions like hypertension, coronary artery disease, diabetes, renal insufficiency and atrial fibrillation. In the absence of a specific intervention for HFpEF, focusing on these other conditions will provide us the opportunity to modify the natural history of this disease."
Dr. David E. Lanfear, a cardiologist specializing in advanced heart failure and transplantation at Henry Ford Hospital in Detroit, said the guidelines on HFpEF “are very reasonable. The recommendations appear similar to those in previous statements, on blood pressure control, volume control for symptoms, atrial fibrillation, and ischemia, without endorsing specific medications. The statement also eloquently points out the ways in which HFpEF represents a huge gap in the knowledge base.
The guidelines contain "critical" new indications for the use of aldosterone antagonists, Dr. Yancy said. The drugs saw a surge in use in the early 2000s, but the rush to embrace them brought challenges as well. "Some of the applications led to missteps resulting in elevated potassium levels and emergency admissions," Dr. Yancy said. Since then, additional trials have pinpointed the best ways to use aldosterone agonists in patients who have heart failure with reduced ejection fraction or cardiac injury after heart attack. Data now confirm their benefit in patients with mild and moderate disease, as well as those with more advanced disease.
"This is the first document in the United States to embrace the benefit of aldosterone antagonists for these patients," Dr. Yancy said. Provided that patient renal function is "reasonably intact," the drugs are a valuable addition to GDMT.
The guidelines also offer a refinement of the recommendations for cardiac resynchronization device therapy – another change supported by the results of recent, large-scale trials. "We now have three separate, well-done trials that suggest a significant benefit of cardiac resynchronization in patients with mild to moderate disease," Dr. Yancy said.
In addition to recommending the treatment for patients with mild to moderate disease, the guideline targets it more specifically. "We gave the greatest strength of recommendation for patients with a wide QRS interval and left bundle branch block, a modest recommendation for patients with a less wide interval, and an equivocal recommendation for those without left bundle branch block. We think this will allow better discrimination of those who are most likely to benefit from CRT from those unlikely to benefit."
There are also more plentiful data in favor of mechanical circulatory support for patients with advanced heart failure. "This is no longer a proof of concept strategy," Dr. Yancy said. "Left ventricular assist devices for advanced chronic heart failure represent an important component of a contemporary treatment algorithm for heart failure."
The guideline even reaches past the mechanics of heart failure into its possible genetic origins. "We’ve discovered that idiopathic dilated cardiomyopathy may not really be idiopathic, but instead related to genetic abnormality. We’ve coalesced observations and data from this emerging field to come up with recommendations about when we might consider genetic testing in patients and screening in family members. It’s something new, and we’re delighted that it’s presented in this document."
The guidelines also offer a new outlook on reducing heart failure readmissions – a problem that comes with a $25 billion/year price tag, Dr. Yancy said. Four simple, low-tech interventions stood out as practical and effective:
• Using in-hospital systems that identify heart failure patients appropriate for GDMT and prompt physicians to advance this care and assess response.
• Developing transitional care and discharge planning that emphasizes patient education to increase treatment compliance, manage comorbid conditions effectively, and tackle psychosocial barriers to care.
• Harnessing the cooperative power of a nurse-led multidisciplinary heart failure disease management program.
• Following up every patient with a phone call within 3 days of discharge and a physician appointment within 2 weeks.
"The beauty of this is that while everyone is looking for the silver bullet to decrease readmission – including high-tech interventions like device implantation and home electronic monitoring – we believe that these simple, straightforward, evidence-based approaches will work."
Finally, Dr. Yancy said, document places great importance on patient-centric outcomes like quality-of-life issues, shared decision-making, care coordination, and palliative care. Over the past decade, the physician/patient relationship has changed from almost paternalistic to an active partnership. "We need to include the patient’s point of view in this whole process. We need to put a greater emphasis on quality of life, and we need not fear a discussion on quality of death."
Dr. Yancy expressed a firm belief that integrating the guidelines into daily practice could have an enormous impact on the way heart failure patients are treated.
"We have come so far in our understanding and ability to treat these patients. These are dramatically effective interventions. We can shift the culture to the belief that heart failure is something that we can treat – to the idea that you can help your patients feel better and live longer. If we use this correctly, we can make a difference."
Dr. Yancy had no financial declarations.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY