User login
Anxiety associated with structural brain differences in children with epilepsy
MONTREAL – The brains of children who have been recently diagnosed with both epilepsy and anxiety are markedly different from those children who have seizures but are anxiety free, a study has shown.
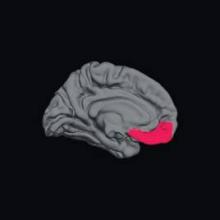
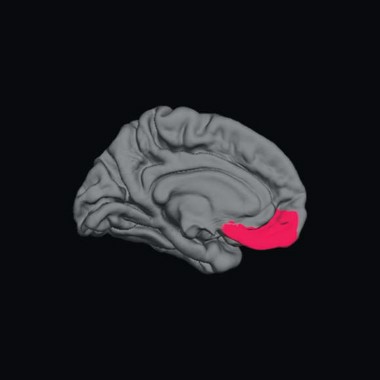
These differences include significantly larger left amygdala, and prefrontal cortices that are thinner in the left medial orbital, right lateral orbital, and right front pole regions – a pattern known to be associated with anxiety and mood disorders in the general population.
Since these children have been diagnosed with epilepsy recently, — and thus have not had years of seizures – these findings may shed new light on the "chicken or egg" association of epilepsy and anxiety, Jana E. Jones, Ph.D., said at the 30th International Epilepsy Conference.
"Seizures are unpredictable, frightening, and are often viewed as a significant factor precipitating anxiety," said Dr. Jones of the University of Wisconsin, Madison. "In contrast, these results suggest the presence of abnormal underlying neurobiology affecting subcortical and cortical regions implicated in anxiety in the general population."
She and her colleagues used magnetic resonance imaging (MRI) to examine brain structure in a cohort of 139 children aged 8-18 years: 25 with epilepsy and anxiety, 64 with epilepsy and no anxiety, and 50 healthy age-matched controls. The children’s average age was 13 years, with an average age of seizure onset of 12 years in those without anxiety and 11 years in those with anxiety. Most were taking one antiepileptic medication, but six in each group were not taking any. Two in the epilepsy/anxiety group were taking multiple antiepileptic medications.
In the epilepsy-only group, most (56%) had an idiopathic generalized epilepsy syndrome; 42% had a localization-related syndrome, and one was unclassified.
In the epilepsy/anxiety group, 68% had a localization-related syndrome and 32%, an idiopathic generalized syndrome.
All of the patients were assessed within 12 months of their epilepsy diagnosis. They all had a normal neurologic exam and a normal clinical MRI. They completed the Kiddie-Schedule for Affective Disorders and Schizophrenia questionnaire. The T1-weighted MRI scans were analyzed with the Free Surfer image analysis program.
In the epilepsy/anxiety group, 11 children had a specific phobia, 8 had separation anxiety disorder, 6 had a social phobia, 5 had generalized anxiety disorder, and 3 had an anxiety disorder not otherwise specified. Eight children had a diagnosis of more than one anxiety disorder.
The control group had a significantly higher average IQ (108) than did the epileptic children with and without anxiety (99.8 and 101.4, respectively). Only 10% of the control subjects had academic problems – children with epilepsy had significantly more academic problems in both groups (44% in the epilepsy/anxiety group and 50% of the epilepsy-only group).
Structural brain measurements showed differences between the two patient groups, relative to the healthy control group. The children with epilepsy and anxiety had significantly larger left amygdala volume than did the epilepsy-only group.
Dr. Jones also found significant differences in cortical thickness in several key areas of the prefrontal cortex. In the epilepsy/anxiety group, the left medial orbital, right lateral orbital, and right frontal pole regions of cortex were significantly thinner than in the epilepsy-only group.
These observations support previous findings in children with disorders such as social anxiety, generalized anxiety, and phobias in the general population, and may be indicative of an abnormal system of emotion regulation involving top-down control of the amygdala via prefrontal cortical connections, Dr. Jones said in an interview.
Her group is currently examining this brain circuitry using diffusion tensor imaging, which allows researchers to image connective fibers in the brain. "Perhaps most importantly, these results suggest that it is important to recognize and treat anxiety early in the course of epilepsy, particularly because there is a relationship between anxiety in childhood leading to ongoing mental health issues in adulthood," she said.
Dr. Jana Jones said she had no relevant financial disclosures.
On Twitter @Alz_Gal
MONTREAL – The brains of children who have been recently diagnosed with both epilepsy and anxiety are markedly different from those children who have seizures but are anxiety free, a study has shown.
These differences include significantly larger left amygdala, and prefrontal cortices that are thinner in the left medial orbital, right lateral orbital, and right front pole regions – a pattern known to be associated with anxiety and mood disorders in the general population.
Since these children have been diagnosed with epilepsy recently, — and thus have not had years of seizures – these findings may shed new light on the "chicken or egg" association of epilepsy and anxiety, Jana E. Jones, Ph.D., said at the 30th International Epilepsy Conference.
"Seizures are unpredictable, frightening, and are often viewed as a significant factor precipitating anxiety," said Dr. Jones of the University of Wisconsin, Madison. "In contrast, these results suggest the presence of abnormal underlying neurobiology affecting subcortical and cortical regions implicated in anxiety in the general population."
She and her colleagues used magnetic resonance imaging (MRI) to examine brain structure in a cohort of 139 children aged 8-18 years: 25 with epilepsy and anxiety, 64 with epilepsy and no anxiety, and 50 healthy age-matched controls. The children’s average age was 13 years, with an average age of seizure onset of 12 years in those without anxiety and 11 years in those with anxiety. Most were taking one antiepileptic medication, but six in each group were not taking any. Two in the epilepsy/anxiety group were taking multiple antiepileptic medications.
In the epilepsy-only group, most (56%) had an idiopathic generalized epilepsy syndrome; 42% had a localization-related syndrome, and one was unclassified.
In the epilepsy/anxiety group, 68% had a localization-related syndrome and 32%, an idiopathic generalized syndrome.
All of the patients were assessed within 12 months of their epilepsy diagnosis. They all had a normal neurologic exam and a normal clinical MRI. They completed the Kiddie-Schedule for Affective Disorders and Schizophrenia questionnaire. The T1-weighted MRI scans were analyzed with the Free Surfer image analysis program.
In the epilepsy/anxiety group, 11 children had a specific phobia, 8 had separation anxiety disorder, 6 had a social phobia, 5 had generalized anxiety disorder, and 3 had an anxiety disorder not otherwise specified. Eight children had a diagnosis of more than one anxiety disorder.
The control group had a significantly higher average IQ (108) than did the epileptic children with and without anxiety (99.8 and 101.4, respectively). Only 10% of the control subjects had academic problems – children with epilepsy had significantly more academic problems in both groups (44% in the epilepsy/anxiety group and 50% of the epilepsy-only group).
Structural brain measurements showed differences between the two patient groups, relative to the healthy control group. The children with epilepsy and anxiety had significantly larger left amygdala volume than did the epilepsy-only group.
Dr. Jones also found significant differences in cortical thickness in several key areas of the prefrontal cortex. In the epilepsy/anxiety group, the left medial orbital, right lateral orbital, and right frontal pole regions of cortex were significantly thinner than in the epilepsy-only group.
These observations support previous findings in children with disorders such as social anxiety, generalized anxiety, and phobias in the general population, and may be indicative of an abnormal system of emotion regulation involving top-down control of the amygdala via prefrontal cortical connections, Dr. Jones said in an interview.
Her group is currently examining this brain circuitry using diffusion tensor imaging, which allows researchers to image connective fibers in the brain. "Perhaps most importantly, these results suggest that it is important to recognize and treat anxiety early in the course of epilepsy, particularly because there is a relationship between anxiety in childhood leading to ongoing mental health issues in adulthood," she said.
Dr. Jana Jones said she had no relevant financial disclosures.
On Twitter @Alz_Gal
MONTREAL – The brains of children who have been recently diagnosed with both epilepsy and anxiety are markedly different from those children who have seizures but are anxiety free, a study has shown.
These differences include significantly larger left amygdala, and prefrontal cortices that are thinner in the left medial orbital, right lateral orbital, and right front pole regions – a pattern known to be associated with anxiety and mood disorders in the general population.
Since these children have been diagnosed with epilepsy recently, — and thus have not had years of seizures – these findings may shed new light on the "chicken or egg" association of epilepsy and anxiety, Jana E. Jones, Ph.D., said at the 30th International Epilepsy Conference.
"Seizures are unpredictable, frightening, and are often viewed as a significant factor precipitating anxiety," said Dr. Jones of the University of Wisconsin, Madison. "In contrast, these results suggest the presence of abnormal underlying neurobiology affecting subcortical and cortical regions implicated in anxiety in the general population."
She and her colleagues used magnetic resonance imaging (MRI) to examine brain structure in a cohort of 139 children aged 8-18 years: 25 with epilepsy and anxiety, 64 with epilepsy and no anxiety, and 50 healthy age-matched controls. The children’s average age was 13 years, with an average age of seizure onset of 12 years in those without anxiety and 11 years in those with anxiety. Most were taking one antiepileptic medication, but six in each group were not taking any. Two in the epilepsy/anxiety group were taking multiple antiepileptic medications.
In the epilepsy-only group, most (56%) had an idiopathic generalized epilepsy syndrome; 42% had a localization-related syndrome, and one was unclassified.
In the epilepsy/anxiety group, 68% had a localization-related syndrome and 32%, an idiopathic generalized syndrome.
All of the patients were assessed within 12 months of their epilepsy diagnosis. They all had a normal neurologic exam and a normal clinical MRI. They completed the Kiddie-Schedule for Affective Disorders and Schizophrenia questionnaire. The T1-weighted MRI scans were analyzed with the Free Surfer image analysis program.
In the epilepsy/anxiety group, 11 children had a specific phobia, 8 had separation anxiety disorder, 6 had a social phobia, 5 had generalized anxiety disorder, and 3 had an anxiety disorder not otherwise specified. Eight children had a diagnosis of more than one anxiety disorder.
The control group had a significantly higher average IQ (108) than did the epileptic children with and without anxiety (99.8 and 101.4, respectively). Only 10% of the control subjects had academic problems – children with epilepsy had significantly more academic problems in both groups (44% in the epilepsy/anxiety group and 50% of the epilepsy-only group).
Structural brain measurements showed differences between the two patient groups, relative to the healthy control group. The children with epilepsy and anxiety had significantly larger left amygdala volume than did the epilepsy-only group.
Dr. Jones also found significant differences in cortical thickness in several key areas of the prefrontal cortex. In the epilepsy/anxiety group, the left medial orbital, right lateral orbital, and right frontal pole regions of cortex were significantly thinner than in the epilepsy-only group.
These observations support previous findings in children with disorders such as social anxiety, generalized anxiety, and phobias in the general population, and may be indicative of an abnormal system of emotion regulation involving top-down control of the amygdala via prefrontal cortical connections, Dr. Jones said in an interview.
Her group is currently examining this brain circuitry using diffusion tensor imaging, which allows researchers to image connective fibers in the brain. "Perhaps most importantly, these results suggest that it is important to recognize and treat anxiety early in the course of epilepsy, particularly because there is a relationship between anxiety in childhood leading to ongoing mental health issues in adulthood," she said.
Dr. Jana Jones said she had no relevant financial disclosures.
On Twitter @Alz_Gal
AT IEC 2013
Major finding: Compared with the children with epilepsy alone, the brains of children with newly diagnosed epilepsy and comorbid anxiety showed significant differences in key areas associated with mood regulation.
Data source: A study comparing brain imaging among 25 children with epilepsy and anxiety, 64 with epilepsy only, and 50 healthy, age-matched controls.
Disclosures: Dr. Jana Jones said she had no relevant financial disclosures.
Pimavanserin reduced Parkinson’s psychosis without motor worsening
BOSTON – A novel 5-HT2A receptor inverse agonist significantly improved psychotic symptoms in Parkinson’s disease patients without the worsening of motor symptoms that usually occurs with antipsychotic treatment, according to the results of a randomized, placebo-controlled phase III trial.*
In addition to benefiting patients, the drug eased caregiver burden, Dr. Clive Ballard said at the Alzheimer’s Association International Conference 2013.
"By 4 weeks into the study, the benefit for caregivers began to appear, and it continued to increase" until the study ended at 6 weeks, said Dr. Ballard, professor of age-related diseases at King’s College London.
Parkinson’s psychosis is a common manifestation of the disease, and about 50% of patients will experience it, he said. "It’s not just frequent. It’s impactful. It’s distressing for patients and for those caring for them. It’s also associated with more impairment, with mortality, and it’s the leading cause of nursing home placement."
Sadly, he noted that there are not many medical options for the problem. Most antipsychotics produce extrapyramidal symptoms, which worsen parkinsonism. Atypical antipsychotics may be well tolerated, but are not very effective against the delusions and hallucinations that can occur in Parkinson’s patients.
"The situation in Parkinson’s is even worse than it is in Alzheimer’s. At least we do have some options for Alzheimer’s psychosis, even if they’re not great," Dr. Ballard said.
The atypical antipsychotics quetiapine and clozapine are the only well-tolerated options, he said. "Quetiapine is ineffective, though, leaving only clozapine. It’s well tolerated from a motor point of view, but it has other safety and tolerability issues that limit its use in clinical practice."
As a selective 5-HT2A inverse agonist, pimavanserin is in a unique drug class. According to Acadia Pharmaceuticals, the company developing the drug, it has the benefits of a 5HT2A agonist but doesn’t affect the dopaminergic, histaminergic, adrenergic, or muscarinic systems.
"Previous studies suggested that it was well tolerated and had some signal of potential benefit in psychosis," Dr. Ballard said. But those trials posted an unusually large placebo effect as well, making the results "difficult to interpret."
Investigators in the pimavanserin phase III study made a number of study design changes to compensate for the placebo response. In order to qualify for the trial, for example, patients had to have high baseline minimum scores on the Neuropsychiatric Inventory and the Scale for the Assessment of Positive Symptoms (SAPS).
They also took part in a brief psychosocial intervention before randomization. "In this intervention, the caregiver spends about 10 minutes each day with the patient, enabling the patient to do an activity they enjoy." This reduces the placebo response rate when the study medication commences, Dr. Ballard said.
The study also centralized any robust key findings, which were interpreted by independent, blinded raters.
The primary endpoint was antipsychotic efficacy as measured using the SAPS-PD, a nine-item scale adapted from the hallucinations and delusions domains of SAPS.
Secondary endpoints included the entire 20-item SAPS, the SAPS hallucinations and delusions subscores, the Clinical Global Impression (CGI) score, Unified Parkinson’s Disease Rating Scale (UPDRS) domains I and II, and a measure of caregiver burden.
The patients were randomized to 40 mg of pimavanserin daily (n = 105) or to placebo (n = 94) for 6 weeks. They were assessed at baseline and at 2, 4, and 6 weeks. After the randomization period, they could join a long-term, open-label extension study at the 40-mg/day dose. Most of the patients completed the trial protocol (87 in the placebo group and 89 in the pimavanserin group). Seven placebo patients dropped out, including two because of an adverse event. Of 15 patients in the pimavanserin group who dropped out, 10 did so because of adverse events. One patient randomized to pimavanserin did not take the medication.
At 6 weeks, patients in the active treatment group had significantly greater improvement on the SAPS-PD, the primary endpoint. By 15 days, both groups had experienced a significant improvement from baseline. Thereafter, the curves separated, with the placebo group becoming stable; Dr. Ballard said this indicated that the study design compensated for a large placebo response. The pimavanserin group, however, continued to improve. By the study’s end, there was a mean SAPS-PD decrease of 5.79 points in the active group, significantly better than the mean 2.73-point decrease in the placebo group. This translated to a 37% improvement for the study drug, compared with a 14% improvement for placebo.
The scores also translated into a meaningful clinical benefit, Dr. Ballard added. By the SAPS-PD measurement, response rates were 42% and 65%, respectively – a significant difference.
Changes on the CGI subscores were also significantly different. Patients in the placebo group experienced a mean decrease of less than half a point on the improvement subscale, compared with a decrease of about 1 point in the pimavanserin group. On the severity subscale, the placebo group stayed close to baseline, while the pimavanserin group decreased by about 1 point. About 27% of those taking placebo and 49% of those taking pimavanserin were considered responders.
Caregiver burden also significantly improved with the study drug.
In an exploratory analysis examining pimavanserin’s effect on sleep, the drug was associated with improvements in both nighttime sleep and daytime wakefulness on the Scale for Outcomes in Parkinson’s Disease sleep measure, Dr. Ballard said.
There were three deaths during the study – one in the placebo group (sudden cardiac death) and two in the active group (sepsis and septic shock). There were also four urinary tract infections – one in the placebo group and three in the active group. Other adverse events reported during the trial included peripheral edema (7% active vs. 3% placebo), falls (11% vs. 6%), confusion (6% vs. 3%), headache (1% vs. 5%), and hallucination (7% vs. 4%).
Based on the favorable results, Acadia announced that it had discontinued its work on a planned confirmatory phase III study. A New Drug Application for pimavanserin is in preparation, Dr. Ballard said.*
Acadia is also planning to study the drug’s effect in Alzheimer’s-associated psychosis in a phase II feasibility trial later this year, according to the company’s website.
"Similar to Parkinson’s disease psychosis, there is no therapy approved to treat Alzheimer’s psychosis in the U.S.," the company said in a written statement. "As symptoms progress and become more severe, physicians often resort to off-label use of antipsychotic medications in these patients. ... Antipsychotic drugs may exacerbate the cognitive disturbances associated with Alzheimer’s disease and also have a black box warning for use in elderly patients with dementia-related psychosis due to increased risks of mortality and morbidity."
The trial was funded by Acadia. Dr. Ballard disclosed that he has received honoraria and consulting fees from the company.
[email protected]
On Twitter @Alz_Gal
* Correction 7/30/13: An earlier version of this article misstated pimavanserin's mechanism of action and the status of its New Drug Application.
BOSTON – A novel 5-HT2A receptor inverse agonist significantly improved psychotic symptoms in Parkinson’s disease patients without the worsening of motor symptoms that usually occurs with antipsychotic treatment, according to the results of a randomized, placebo-controlled phase III trial.*
In addition to benefiting patients, the drug eased caregiver burden, Dr. Clive Ballard said at the Alzheimer’s Association International Conference 2013.
"By 4 weeks into the study, the benefit for caregivers began to appear, and it continued to increase" until the study ended at 6 weeks, said Dr. Ballard, professor of age-related diseases at King’s College London.
Parkinson’s psychosis is a common manifestation of the disease, and about 50% of patients will experience it, he said. "It’s not just frequent. It’s impactful. It’s distressing for patients and for those caring for them. It’s also associated with more impairment, with mortality, and it’s the leading cause of nursing home placement."
Sadly, he noted that there are not many medical options for the problem. Most antipsychotics produce extrapyramidal symptoms, which worsen parkinsonism. Atypical antipsychotics may be well tolerated, but are not very effective against the delusions and hallucinations that can occur in Parkinson’s patients.
"The situation in Parkinson’s is even worse than it is in Alzheimer’s. At least we do have some options for Alzheimer’s psychosis, even if they’re not great," Dr. Ballard said.
The atypical antipsychotics quetiapine and clozapine are the only well-tolerated options, he said. "Quetiapine is ineffective, though, leaving only clozapine. It’s well tolerated from a motor point of view, but it has other safety and tolerability issues that limit its use in clinical practice."
As a selective 5-HT2A inverse agonist, pimavanserin is in a unique drug class. According to Acadia Pharmaceuticals, the company developing the drug, it has the benefits of a 5HT2A agonist but doesn’t affect the dopaminergic, histaminergic, adrenergic, or muscarinic systems.
"Previous studies suggested that it was well tolerated and had some signal of potential benefit in psychosis," Dr. Ballard said. But those trials posted an unusually large placebo effect as well, making the results "difficult to interpret."
Investigators in the pimavanserin phase III study made a number of study design changes to compensate for the placebo response. In order to qualify for the trial, for example, patients had to have high baseline minimum scores on the Neuropsychiatric Inventory and the Scale for the Assessment of Positive Symptoms (SAPS).
They also took part in a brief psychosocial intervention before randomization. "In this intervention, the caregiver spends about 10 minutes each day with the patient, enabling the patient to do an activity they enjoy." This reduces the placebo response rate when the study medication commences, Dr. Ballard said.
The study also centralized any robust key findings, which were interpreted by independent, blinded raters.
The primary endpoint was antipsychotic efficacy as measured using the SAPS-PD, a nine-item scale adapted from the hallucinations and delusions domains of SAPS.
Secondary endpoints included the entire 20-item SAPS, the SAPS hallucinations and delusions subscores, the Clinical Global Impression (CGI) score, Unified Parkinson’s Disease Rating Scale (UPDRS) domains I and II, and a measure of caregiver burden.
The patients were randomized to 40 mg of pimavanserin daily (n = 105) or to placebo (n = 94) for 6 weeks. They were assessed at baseline and at 2, 4, and 6 weeks. After the randomization period, they could join a long-term, open-label extension study at the 40-mg/day dose. Most of the patients completed the trial protocol (87 in the placebo group and 89 in the pimavanserin group). Seven placebo patients dropped out, including two because of an adverse event. Of 15 patients in the pimavanserin group who dropped out, 10 did so because of adverse events. One patient randomized to pimavanserin did not take the medication.
At 6 weeks, patients in the active treatment group had significantly greater improvement on the SAPS-PD, the primary endpoint. By 15 days, both groups had experienced a significant improvement from baseline. Thereafter, the curves separated, with the placebo group becoming stable; Dr. Ballard said this indicated that the study design compensated for a large placebo response. The pimavanserin group, however, continued to improve. By the study’s end, there was a mean SAPS-PD decrease of 5.79 points in the active group, significantly better than the mean 2.73-point decrease in the placebo group. This translated to a 37% improvement for the study drug, compared with a 14% improvement for placebo.
The scores also translated into a meaningful clinical benefit, Dr. Ballard added. By the SAPS-PD measurement, response rates were 42% and 65%, respectively – a significant difference.
Changes on the CGI subscores were also significantly different. Patients in the placebo group experienced a mean decrease of less than half a point on the improvement subscale, compared with a decrease of about 1 point in the pimavanserin group. On the severity subscale, the placebo group stayed close to baseline, while the pimavanserin group decreased by about 1 point. About 27% of those taking placebo and 49% of those taking pimavanserin were considered responders.
Caregiver burden also significantly improved with the study drug.
In an exploratory analysis examining pimavanserin’s effect on sleep, the drug was associated with improvements in both nighttime sleep and daytime wakefulness on the Scale for Outcomes in Parkinson’s Disease sleep measure, Dr. Ballard said.
There were three deaths during the study – one in the placebo group (sudden cardiac death) and two in the active group (sepsis and septic shock). There were also four urinary tract infections – one in the placebo group and three in the active group. Other adverse events reported during the trial included peripheral edema (7% active vs. 3% placebo), falls (11% vs. 6%), confusion (6% vs. 3%), headache (1% vs. 5%), and hallucination (7% vs. 4%).
Based on the favorable results, Acadia announced that it had discontinued its work on a planned confirmatory phase III study. A New Drug Application for pimavanserin is in preparation, Dr. Ballard said.*
Acadia is also planning to study the drug’s effect in Alzheimer’s-associated psychosis in a phase II feasibility trial later this year, according to the company’s website.
"Similar to Parkinson’s disease psychosis, there is no therapy approved to treat Alzheimer’s psychosis in the U.S.," the company said in a written statement. "As symptoms progress and become more severe, physicians often resort to off-label use of antipsychotic medications in these patients. ... Antipsychotic drugs may exacerbate the cognitive disturbances associated with Alzheimer’s disease and also have a black box warning for use in elderly patients with dementia-related psychosis due to increased risks of mortality and morbidity."
The trial was funded by Acadia. Dr. Ballard disclosed that he has received honoraria and consulting fees from the company.
[email protected]
On Twitter @Alz_Gal
* Correction 7/30/13: An earlier version of this article misstated pimavanserin's mechanism of action and the status of its New Drug Application.
BOSTON – A novel 5-HT2A receptor inverse agonist significantly improved psychotic symptoms in Parkinson’s disease patients without the worsening of motor symptoms that usually occurs with antipsychotic treatment, according to the results of a randomized, placebo-controlled phase III trial.*
In addition to benefiting patients, the drug eased caregiver burden, Dr. Clive Ballard said at the Alzheimer’s Association International Conference 2013.
"By 4 weeks into the study, the benefit for caregivers began to appear, and it continued to increase" until the study ended at 6 weeks, said Dr. Ballard, professor of age-related diseases at King’s College London.
Parkinson’s psychosis is a common manifestation of the disease, and about 50% of patients will experience it, he said. "It’s not just frequent. It’s impactful. It’s distressing for patients and for those caring for them. It’s also associated with more impairment, with mortality, and it’s the leading cause of nursing home placement."
Sadly, he noted that there are not many medical options for the problem. Most antipsychotics produce extrapyramidal symptoms, which worsen parkinsonism. Atypical antipsychotics may be well tolerated, but are not very effective against the delusions and hallucinations that can occur in Parkinson’s patients.
"The situation in Parkinson’s is even worse than it is in Alzheimer’s. At least we do have some options for Alzheimer’s psychosis, even if they’re not great," Dr. Ballard said.
The atypical antipsychotics quetiapine and clozapine are the only well-tolerated options, he said. "Quetiapine is ineffective, though, leaving only clozapine. It’s well tolerated from a motor point of view, but it has other safety and tolerability issues that limit its use in clinical practice."
As a selective 5-HT2A inverse agonist, pimavanserin is in a unique drug class. According to Acadia Pharmaceuticals, the company developing the drug, it has the benefits of a 5HT2A agonist but doesn’t affect the dopaminergic, histaminergic, adrenergic, or muscarinic systems.
"Previous studies suggested that it was well tolerated and had some signal of potential benefit in psychosis," Dr. Ballard said. But those trials posted an unusually large placebo effect as well, making the results "difficult to interpret."
Investigators in the pimavanserin phase III study made a number of study design changes to compensate for the placebo response. In order to qualify for the trial, for example, patients had to have high baseline minimum scores on the Neuropsychiatric Inventory and the Scale for the Assessment of Positive Symptoms (SAPS).
They also took part in a brief psychosocial intervention before randomization. "In this intervention, the caregiver spends about 10 minutes each day with the patient, enabling the patient to do an activity they enjoy." This reduces the placebo response rate when the study medication commences, Dr. Ballard said.
The study also centralized any robust key findings, which were interpreted by independent, blinded raters.
The primary endpoint was antipsychotic efficacy as measured using the SAPS-PD, a nine-item scale adapted from the hallucinations and delusions domains of SAPS.
Secondary endpoints included the entire 20-item SAPS, the SAPS hallucinations and delusions subscores, the Clinical Global Impression (CGI) score, Unified Parkinson’s Disease Rating Scale (UPDRS) domains I and II, and a measure of caregiver burden.
The patients were randomized to 40 mg of pimavanserin daily (n = 105) or to placebo (n = 94) for 6 weeks. They were assessed at baseline and at 2, 4, and 6 weeks. After the randomization period, they could join a long-term, open-label extension study at the 40-mg/day dose. Most of the patients completed the trial protocol (87 in the placebo group and 89 in the pimavanserin group). Seven placebo patients dropped out, including two because of an adverse event. Of 15 patients in the pimavanserin group who dropped out, 10 did so because of adverse events. One patient randomized to pimavanserin did not take the medication.
At 6 weeks, patients in the active treatment group had significantly greater improvement on the SAPS-PD, the primary endpoint. By 15 days, both groups had experienced a significant improvement from baseline. Thereafter, the curves separated, with the placebo group becoming stable; Dr. Ballard said this indicated that the study design compensated for a large placebo response. The pimavanserin group, however, continued to improve. By the study’s end, there was a mean SAPS-PD decrease of 5.79 points in the active group, significantly better than the mean 2.73-point decrease in the placebo group. This translated to a 37% improvement for the study drug, compared with a 14% improvement for placebo.
The scores also translated into a meaningful clinical benefit, Dr. Ballard added. By the SAPS-PD measurement, response rates were 42% and 65%, respectively – a significant difference.
Changes on the CGI subscores were also significantly different. Patients in the placebo group experienced a mean decrease of less than half a point on the improvement subscale, compared with a decrease of about 1 point in the pimavanserin group. On the severity subscale, the placebo group stayed close to baseline, while the pimavanserin group decreased by about 1 point. About 27% of those taking placebo and 49% of those taking pimavanserin were considered responders.
Caregiver burden also significantly improved with the study drug.
In an exploratory analysis examining pimavanserin’s effect on sleep, the drug was associated with improvements in both nighttime sleep and daytime wakefulness on the Scale for Outcomes in Parkinson’s Disease sleep measure, Dr. Ballard said.
There were three deaths during the study – one in the placebo group (sudden cardiac death) and two in the active group (sepsis and septic shock). There were also four urinary tract infections – one in the placebo group and three in the active group. Other adverse events reported during the trial included peripheral edema (7% active vs. 3% placebo), falls (11% vs. 6%), confusion (6% vs. 3%), headache (1% vs. 5%), and hallucination (7% vs. 4%).
Based on the favorable results, Acadia announced that it had discontinued its work on a planned confirmatory phase III study. A New Drug Application for pimavanserin is in preparation, Dr. Ballard said.*
Acadia is also planning to study the drug’s effect in Alzheimer’s-associated psychosis in a phase II feasibility trial later this year, according to the company’s website.
"Similar to Parkinson’s disease psychosis, there is no therapy approved to treat Alzheimer’s psychosis in the U.S.," the company said in a written statement. "As symptoms progress and become more severe, physicians often resort to off-label use of antipsychotic medications in these patients. ... Antipsychotic drugs may exacerbate the cognitive disturbances associated with Alzheimer’s disease and also have a black box warning for use in elderly patients with dementia-related psychosis due to increased risks of mortality and morbidity."
The trial was funded by Acadia. Dr. Ballard disclosed that he has received honoraria and consulting fees from the company.
[email protected]
On Twitter @Alz_Gal
* Correction 7/30/13: An earlier version of this article misstated pimavanserin's mechanism of action and the status of its New Drug Application.
AT AAIC2013
Major finding: Pimavanserin improved symptoms of Parkinson’s disease psychosis by 37%, compared with a 14% improvement for placebo, without inducing extrapyramidal symptoms.
Data source: The phase III study randomized 199 patients to either placebo or pimavanserin 40 mg/day for 6 weeks.
Disclosures: Acadia Pharmaceuticals funded the study. Dr. Ballard disclosed that he has received honoraria and consulting fees from the company.
Brain atrophy rate may predict later cognitive decline
BOSTON – For people with mild cognitive impairment, the rate of brain atrophy now may predict the level of cognitive decline years from now.
Every 1% increase in the rate of whole brain atrophy during a single year translated into 1.7-point decline on the Mini Mental State Examination (MMSE) score at 3 years, Kelvin Leung, Ph.D., said at the Alzheimer’s Association International Conference 2013.
"Many studies have shown associations between concurrent brain atrophy rates and cognitive decline, when compared and measured over the same time period," said Dr. Leung of the University College, London. "However, fewer have looked at the association between brain atrophy rates and future cognitive decline."
To examine that question, Dr. Leung used cognitive and imaging data from the Alzheimer’s Disease Neuroimaging Initiative. His study cohort included 471 patients (295 with mild cognitive impairment and 176 normal controls). The primary outcome was 36-month change on the MMSE. Secondary measures included changes in auditory verbal learning, immediate recall, category fluency, trail making, and backward digit span.
He used a magnetic resonance imaging measurement called boundary shift integral to calculate brain volume changes from baseline to 12 months. The boundary shift integral takes into account whole brain shrinkage, hippocampus shrinkage, and ventricle expansion to calculate a percentage difference from one time point to another.
When looking at the raw scores, every 1% increase in the 1-year brain atrophy rate was associated with a statistically significant 1.7-point decrease on the MMSE.
That atrophy rate also was significantly associated with a 2-point decline on the audio verbal learning test, a 2.8-point decline on the category fluency test, and an 11-point decline on the Trail Making Test A. The control subjects showed no significant cognitive changes.
Dr. Leung saw similar changes when he looked only at the change in hippocampal volume. For every 1% increase in the 1-year rate of hippocampal atrophy, patients with mild cognitive impairment experienced significant declines in the MMSE, audio verbal learning test, immediate memory, category fluency, and trail making. Again, control patients showed no changes.
There was a nearly identical correlation between ventricular volume expansion and the cognitive measures, he said.
The next step will be to investigate which specific cognitive changes are most associated with specific brain region changes.
Dr. Leung made no financial disclosures.
BOSTON – For people with mild cognitive impairment, the rate of brain atrophy now may predict the level of cognitive decline years from now.
Every 1% increase in the rate of whole brain atrophy during a single year translated into 1.7-point decline on the Mini Mental State Examination (MMSE) score at 3 years, Kelvin Leung, Ph.D., said at the Alzheimer’s Association International Conference 2013.
"Many studies have shown associations between concurrent brain atrophy rates and cognitive decline, when compared and measured over the same time period," said Dr. Leung of the University College, London. "However, fewer have looked at the association between brain atrophy rates and future cognitive decline."
To examine that question, Dr. Leung used cognitive and imaging data from the Alzheimer’s Disease Neuroimaging Initiative. His study cohort included 471 patients (295 with mild cognitive impairment and 176 normal controls). The primary outcome was 36-month change on the MMSE. Secondary measures included changes in auditory verbal learning, immediate recall, category fluency, trail making, and backward digit span.
He used a magnetic resonance imaging measurement called boundary shift integral to calculate brain volume changes from baseline to 12 months. The boundary shift integral takes into account whole brain shrinkage, hippocampus shrinkage, and ventricle expansion to calculate a percentage difference from one time point to another.
When looking at the raw scores, every 1% increase in the 1-year brain atrophy rate was associated with a statistically significant 1.7-point decrease on the MMSE.
That atrophy rate also was significantly associated with a 2-point decline on the audio verbal learning test, a 2.8-point decline on the category fluency test, and an 11-point decline on the Trail Making Test A. The control subjects showed no significant cognitive changes.
Dr. Leung saw similar changes when he looked only at the change in hippocampal volume. For every 1% increase in the 1-year rate of hippocampal atrophy, patients with mild cognitive impairment experienced significant declines in the MMSE, audio verbal learning test, immediate memory, category fluency, and trail making. Again, control patients showed no changes.
There was a nearly identical correlation between ventricular volume expansion and the cognitive measures, he said.
The next step will be to investigate which specific cognitive changes are most associated with specific brain region changes.
Dr. Leung made no financial disclosures.
BOSTON – For people with mild cognitive impairment, the rate of brain atrophy now may predict the level of cognitive decline years from now.
Every 1% increase in the rate of whole brain atrophy during a single year translated into 1.7-point decline on the Mini Mental State Examination (MMSE) score at 3 years, Kelvin Leung, Ph.D., said at the Alzheimer’s Association International Conference 2013.
"Many studies have shown associations between concurrent brain atrophy rates and cognitive decline, when compared and measured over the same time period," said Dr. Leung of the University College, London. "However, fewer have looked at the association between brain atrophy rates and future cognitive decline."
To examine that question, Dr. Leung used cognitive and imaging data from the Alzheimer’s Disease Neuroimaging Initiative. His study cohort included 471 patients (295 with mild cognitive impairment and 176 normal controls). The primary outcome was 36-month change on the MMSE. Secondary measures included changes in auditory verbal learning, immediate recall, category fluency, trail making, and backward digit span.
He used a magnetic resonance imaging measurement called boundary shift integral to calculate brain volume changes from baseline to 12 months. The boundary shift integral takes into account whole brain shrinkage, hippocampus shrinkage, and ventricle expansion to calculate a percentage difference from one time point to another.
When looking at the raw scores, every 1% increase in the 1-year brain atrophy rate was associated with a statistically significant 1.7-point decrease on the MMSE.
That atrophy rate also was significantly associated with a 2-point decline on the audio verbal learning test, a 2.8-point decline on the category fluency test, and an 11-point decline on the Trail Making Test A. The control subjects showed no significant cognitive changes.
Dr. Leung saw similar changes when he looked only at the change in hippocampal volume. For every 1% increase in the 1-year rate of hippocampal atrophy, patients with mild cognitive impairment experienced significant declines in the MMSE, audio verbal learning test, immediate memory, category fluency, and trail making. Again, control patients showed no changes.
There was a nearly identical correlation between ventricular volume expansion and the cognitive measures, he said.
The next step will be to investigate which specific cognitive changes are most associated with specific brain region changes.
Dr. Leung made no financial disclosures.
AT AAIC2013
Major finding: For patients with mild cognitive impairment, every 1% increase in the rate of whole brain atrophy during a single year was associated with a 1.7-point MMSE score decrease 3 years later.
Data source: Retrospective study of 471 patients from the Alzheimer’s Disease Neuroimaging Initiative.
Disclosures: Dr. Leung had no financial disclosures.
Epilepsy patients can face long-term social problems
MONTREAL – Adults who have long-standing epilepsy often have many social difficulties to contend with regardless of whether their childhood seizures have remitted or not, according to a 30-year longitudinal study.
The outcomes of adults who had three different types of childhood epilepsies were "amazingly similar," with high rates of social and romantic isolation, low educational achievement, and psychiatric diagnoses, Dr. Carol Camfield said at the 30th International Epilepsy Congress.
Even those whose seizures had remitted and who were off medication were still likely to have at least one marker of poor social outcome, said Dr. Camfield, a professor of pediatrics at the Dalhousie University in Halifax, N.S.
The finding highlights the need for more research into interventions that can address these long-term issues, she said. "They require from all of us prospective interventions in schooling, socialization, employment, and sexuality," she said. "And that’s a lot to ask from a pediatrician."
Her study looked at almost 3 decades of follow-up data on patients included in the Nova Scotia childhood population-based epilepsy cohort. This cohort comprises 692 adults – all the children who were diagnosed with epilepsy in the province in 1977-1985.
From this group, she selected 137 who had epilepsy only, normal intelligence, and a normal neurologic exam. These patients included 23 with juvenile myoclonic epilepsy (JME), 34 with generalized tonic-clonic seizures alone (GTCA), and 80 with focal seizures with secondary generalization (SEC gen).
The median age of onset ranged from 7 years in the GTCA and SEC gen groups to 10 years in the JME group. Age at last follow-up visit ranged from a median of 29 years for GTCA patients, to 35 years for SEC gen patients, to 36 years for JME patients.
Dr. Camfield examined eight social outcomes in each group:
• High school graduation. Subjects with JME fared the best in education. A high school diploma was not achieved by 13% of JME patients, compared with 40% of GTCA and 32% of SEC gen patients.
• Unable to name a single close friend. A quarter of GTCA patients were unable to name a close friend, compared with 9% of JME and 8% of SEC gen patients.
• Unemployment. The groups had similar rates of unemployment (31% JME, 33% GTCA, and 23% SEC gen).
• A psychiatric diagnosis other than attention-deficit/hyperactivity disorder. SEC gen patients were significantly less likely than were the others to have a psychiatric diagnosis other than ADHD (15% vs. 39% JME and 27% GTCA).
• A criminal conviction. All three groups had similar rates of convictions (4% JME, 7% GTCA, and 4% SEC gen).
• No romantic relationship of more than 3 months. Patients with GTCA were more likely to have never experienced a lasting romantic relationship (24% vs. 17% JME and 14% SEC gen)
• Living alone at the end of follow-up. At the final follow-up, SEC gen patients were significantly less likely to be living alone than were the other groups (8% vs. 30% JME and 23% GTCA).
• Pregnancy outside a stable relationship of more than 6 months. There was some indication of sexual impulsivity in the JME and GTCA groups, Dr. Camfield said. If these patients had become pregnant or caused a pregnancy, it was much more likely to have occurred outside of a stable relationship of at least 6 months’ duration, compared with those in the SEC gen group (79% and 65% vs. 37%).
However, when looking at social outcomes as a whole, there were no significant between-group differences. The majority of each group had at least one of the outcome measures (74% JMW, 76% GTCA, 62% SEC gen). It was not uncommon for patients to have two or more of the outcomes, Dr. Camfield said (22% JME, 21% GTCA, 10% SEC gen).
Remission, defined as being seizure free for at least 5 years and off all antiepileptic drugs, occurred in 40% of the JME group, 75% of the GTCA group, and 81% of the SEC gen group. This clearly indicates that social outcomes remained independent of remission status, Dr. Camfield said.
She had no financial declarations.
On Twitter @Alz_Gal
MONTREAL – Adults who have long-standing epilepsy often have many social difficulties to contend with regardless of whether their childhood seizures have remitted or not, according to a 30-year longitudinal study.
The outcomes of adults who had three different types of childhood epilepsies were "amazingly similar," with high rates of social and romantic isolation, low educational achievement, and psychiatric diagnoses, Dr. Carol Camfield said at the 30th International Epilepsy Congress.
Even those whose seizures had remitted and who were off medication were still likely to have at least one marker of poor social outcome, said Dr. Camfield, a professor of pediatrics at the Dalhousie University in Halifax, N.S.
The finding highlights the need for more research into interventions that can address these long-term issues, she said. "They require from all of us prospective interventions in schooling, socialization, employment, and sexuality," she said. "And that’s a lot to ask from a pediatrician."
Her study looked at almost 3 decades of follow-up data on patients included in the Nova Scotia childhood population-based epilepsy cohort. This cohort comprises 692 adults – all the children who were diagnosed with epilepsy in the province in 1977-1985.
From this group, she selected 137 who had epilepsy only, normal intelligence, and a normal neurologic exam. These patients included 23 with juvenile myoclonic epilepsy (JME), 34 with generalized tonic-clonic seizures alone (GTCA), and 80 with focal seizures with secondary generalization (SEC gen).
The median age of onset ranged from 7 years in the GTCA and SEC gen groups to 10 years in the JME group. Age at last follow-up visit ranged from a median of 29 years for GTCA patients, to 35 years for SEC gen patients, to 36 years for JME patients.
Dr. Camfield examined eight social outcomes in each group:
• High school graduation. Subjects with JME fared the best in education. A high school diploma was not achieved by 13% of JME patients, compared with 40% of GTCA and 32% of SEC gen patients.
• Unable to name a single close friend. A quarter of GTCA patients were unable to name a close friend, compared with 9% of JME and 8% of SEC gen patients.
• Unemployment. The groups had similar rates of unemployment (31% JME, 33% GTCA, and 23% SEC gen).
• A psychiatric diagnosis other than attention-deficit/hyperactivity disorder. SEC gen patients were significantly less likely than were the others to have a psychiatric diagnosis other than ADHD (15% vs. 39% JME and 27% GTCA).
• A criminal conviction. All three groups had similar rates of convictions (4% JME, 7% GTCA, and 4% SEC gen).
• No romantic relationship of more than 3 months. Patients with GTCA were more likely to have never experienced a lasting romantic relationship (24% vs. 17% JME and 14% SEC gen)
• Living alone at the end of follow-up. At the final follow-up, SEC gen patients were significantly less likely to be living alone than were the other groups (8% vs. 30% JME and 23% GTCA).
• Pregnancy outside a stable relationship of more than 6 months. There was some indication of sexual impulsivity in the JME and GTCA groups, Dr. Camfield said. If these patients had become pregnant or caused a pregnancy, it was much more likely to have occurred outside of a stable relationship of at least 6 months’ duration, compared with those in the SEC gen group (79% and 65% vs. 37%).
However, when looking at social outcomes as a whole, there were no significant between-group differences. The majority of each group had at least one of the outcome measures (74% JMW, 76% GTCA, 62% SEC gen). It was not uncommon for patients to have two or more of the outcomes, Dr. Camfield said (22% JME, 21% GTCA, 10% SEC gen).
Remission, defined as being seizure free for at least 5 years and off all antiepileptic drugs, occurred in 40% of the JME group, 75% of the GTCA group, and 81% of the SEC gen group. This clearly indicates that social outcomes remained independent of remission status, Dr. Camfield said.
She had no financial declarations.
On Twitter @Alz_Gal
MONTREAL – Adults who have long-standing epilepsy often have many social difficulties to contend with regardless of whether their childhood seizures have remitted or not, according to a 30-year longitudinal study.
The outcomes of adults who had three different types of childhood epilepsies were "amazingly similar," with high rates of social and romantic isolation, low educational achievement, and psychiatric diagnoses, Dr. Carol Camfield said at the 30th International Epilepsy Congress.
Even those whose seizures had remitted and who were off medication were still likely to have at least one marker of poor social outcome, said Dr. Camfield, a professor of pediatrics at the Dalhousie University in Halifax, N.S.
The finding highlights the need for more research into interventions that can address these long-term issues, she said. "They require from all of us prospective interventions in schooling, socialization, employment, and sexuality," she said. "And that’s a lot to ask from a pediatrician."
Her study looked at almost 3 decades of follow-up data on patients included in the Nova Scotia childhood population-based epilepsy cohort. This cohort comprises 692 adults – all the children who were diagnosed with epilepsy in the province in 1977-1985.
From this group, she selected 137 who had epilepsy only, normal intelligence, and a normal neurologic exam. These patients included 23 with juvenile myoclonic epilepsy (JME), 34 with generalized tonic-clonic seizures alone (GTCA), and 80 with focal seizures with secondary generalization (SEC gen).
The median age of onset ranged from 7 years in the GTCA and SEC gen groups to 10 years in the JME group. Age at last follow-up visit ranged from a median of 29 years for GTCA patients, to 35 years for SEC gen patients, to 36 years for JME patients.
Dr. Camfield examined eight social outcomes in each group:
• High school graduation. Subjects with JME fared the best in education. A high school diploma was not achieved by 13% of JME patients, compared with 40% of GTCA and 32% of SEC gen patients.
• Unable to name a single close friend. A quarter of GTCA patients were unable to name a close friend, compared with 9% of JME and 8% of SEC gen patients.
• Unemployment. The groups had similar rates of unemployment (31% JME, 33% GTCA, and 23% SEC gen).
• A psychiatric diagnosis other than attention-deficit/hyperactivity disorder. SEC gen patients were significantly less likely than were the others to have a psychiatric diagnosis other than ADHD (15% vs. 39% JME and 27% GTCA).
• A criminal conviction. All three groups had similar rates of convictions (4% JME, 7% GTCA, and 4% SEC gen).
• No romantic relationship of more than 3 months. Patients with GTCA were more likely to have never experienced a lasting romantic relationship (24% vs. 17% JME and 14% SEC gen)
• Living alone at the end of follow-up. At the final follow-up, SEC gen patients were significantly less likely to be living alone than were the other groups (8% vs. 30% JME and 23% GTCA).
• Pregnancy outside a stable relationship of more than 6 months. There was some indication of sexual impulsivity in the JME and GTCA groups, Dr. Camfield said. If these patients had become pregnant or caused a pregnancy, it was much more likely to have occurred outside of a stable relationship of at least 6 months’ duration, compared with those in the SEC gen group (79% and 65% vs. 37%).
However, when looking at social outcomes as a whole, there were no significant between-group differences. The majority of each group had at least one of the outcome measures (74% JMW, 76% GTCA, 62% SEC gen). It was not uncommon for patients to have two or more of the outcomes, Dr. Camfield said (22% JME, 21% GTCA, 10% SEC gen).
Remission, defined as being seizure free for at least 5 years and off all antiepileptic drugs, occurred in 40% of the JME group, 75% of the GTCA group, and 81% of the SEC gen group. This clearly indicates that social outcomes remained independent of remission status, Dr. Camfield said.
She had no financial declarations.
On Twitter @Alz_Gal
AT IEC 2013
Major finding: More than half of adults with childhood generalized epilepsies had at least one problematic social issue.
Data source: The Nova Scotia childhood population-based epilepsy cohort provided almost 30 years’ worth of follow-up data on 137 patients.
Disclosures: Dr. Camfield had no financial disclosures.
In vivo tau imaging confirms patient’s Alzheimer’s diagnosis
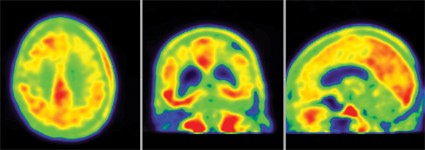
BOSTON – The first investigational tau radioligand for PET imaging has identified pathologic tangles in the brain of a man who was confirmed postmortem to have Alzheimer’s disease.
The patient who died was 1 of 12 participants in a study of the agent, [F-18]T808, and his imaging and neuropathologic results show that the agent binds exclusively to tau deposits, Hartmuth Kolb, Ph.D., said at a press conference at the Alzheimer’s Association International Conference 2013.
The 85-year-old man died of a pulmonary embolism 2 weeks after his [F-18]T808 scan. His death was unrelated to his diagnosis or to the imaging agent, Dr. Kolb said. Avid Radiopharmaceuticals, the company developing the tracer, obtained the brain for autopsy. The examination showed extensive tau tangles in exactly the same areas identified in a PET scan using [F-18]T808. Tau tangles show as red in the PET images; the warmer the shade, the more dense the tangles.
"The results were quite striking," said Dr. Kolb, senior vice president of research at Avid. "The Braak stage was a 5 or 6, which corresponded with a Mini-Mental State Examination [MMSE] score of 15. On the scan, tau was all over the brain except for a sparing of the sensorimotor cortex, and that was exactly what we had found," in the postmortem exam.
The first human imaging cohort comprised nine patients with diagnosed Alzheimer’s and three healthy control subjects. Investigators compared the in vivo PET images with immunohistochemistry and with staining from fluorescent tau ligand T557, which is used to identify the protein in postmortem exams.
In patients diagnosed with Alzheimer’s, the [F-18]T808 scan showed strong positive correlations with the clinical exam in the temporal lobe, frontal lobe, parietal lobe, and hippocampus. The posterior cingulate gyrus and putamen were relatively spared, and the cerebellum was negative. Patients with an MMSE score of 24 showed moderate tau deposition. Scans of three patients who had an MMSE score of 19, 14, and 3, respectively, showed a progressively denser tau concentration.
There was no significant tau accumulation in any of the three healthy control subjects.
There was one outlier – a patient with an MMSE score of 12 who showed only a very small amount of tau. Dr. Kolb said this patient was probably misdiagnosed as having Alzheimer’s – a problem that continues to plague both clinicians and researchers.
"We don’t have numbers of how many people are misdiagnosed every year, but we do know it’s a big problem," Heather Snyder, Ph.D., director of medical and scientific operations for the Alzheimer’s Association, said in an interview. Misdiagnosis is "not only a mentally difficult thing to go through, but it can potentially keep someone from getting the correct care."
In vivo imaging of Alzheimer’s brain pathology is now held to be an imperative for moving Alzheimer’s research forward. The first agent, Pittsburgh compound B (PiB), which binds to amyloid plaques, has virtually revolutionized research by allowing direct visualization of plaques in living patients. PiB has helped to build a new understanding of prodromal and presymptomatic Alzheimer’s, and it has contributed greatly to patient stratification in clinical trials and to the ability to track therapeutic response.
But PiB is not particularly useful in a clinical setting due to its short, 20-minute half-life. Florbetapir (Amyvid), the investigational PET radioligand florbetaben, and [F-18]T808 all use the same fluorine isotope, making their half-lives 120 minutes and much more friendly for clinical use. [F-18]T808 reaches a plateau of action at about 40 minutes post injection, Dr. Kolb said.
Avid performed its study under an Investigational New Drug designation granted last year, Dr. Kolb said. The company will continue to accrue patients toward an approval request, but it didn’t speculate as to how long that might take, he noted.
[F-18]T808 was being developed by Siemens Medical Solutions USA until last April, when Avid, a subsidiary of Eli Lilly, purchased it along with another investigational tau radiotracer.
The other agent is named [F-18]T807, Lilly spokesperson Eva Catherine Groves said in an interview. She noted that "both have reported similar results and, as such, we plan to complete additional development work on both tracers prior to selecting at least one for advancement into our Alzheimer’s research and development programs."
Dr. Kolb is an employee of Avid Radiopharmaceuticals.
[email protected]
On Twitter @Alz_Gal
BOSTON – The first investigational tau radioligand for PET imaging has identified pathologic tangles in the brain of a man who was confirmed postmortem to have Alzheimer’s disease.
The patient who died was 1 of 12 participants in a study of the agent, [F-18]T808, and his imaging and neuropathologic results show that the agent binds exclusively to tau deposits, Hartmuth Kolb, Ph.D., said at a press conference at the Alzheimer’s Association International Conference 2013.
The 85-year-old man died of a pulmonary embolism 2 weeks after his [F-18]T808 scan. His death was unrelated to his diagnosis or to the imaging agent, Dr. Kolb said. Avid Radiopharmaceuticals, the company developing the tracer, obtained the brain for autopsy. The examination showed extensive tau tangles in exactly the same areas identified in a PET scan using [F-18]T808. Tau tangles show as red in the PET images; the warmer the shade, the more dense the tangles.
"The results were quite striking," said Dr. Kolb, senior vice president of research at Avid. "The Braak stage was a 5 or 6, which corresponded with a Mini-Mental State Examination [MMSE] score of 15. On the scan, tau was all over the brain except for a sparing of the sensorimotor cortex, and that was exactly what we had found," in the postmortem exam.
The first human imaging cohort comprised nine patients with diagnosed Alzheimer’s and three healthy control subjects. Investigators compared the in vivo PET images with immunohistochemistry and with staining from fluorescent tau ligand T557, which is used to identify the protein in postmortem exams.
In patients diagnosed with Alzheimer’s, the [F-18]T808 scan showed strong positive correlations with the clinical exam in the temporal lobe, frontal lobe, parietal lobe, and hippocampus. The posterior cingulate gyrus and putamen were relatively spared, and the cerebellum was negative. Patients with an MMSE score of 24 showed moderate tau deposition. Scans of three patients who had an MMSE score of 19, 14, and 3, respectively, showed a progressively denser tau concentration.
There was no significant tau accumulation in any of the three healthy control subjects.
There was one outlier – a patient with an MMSE score of 12 who showed only a very small amount of tau. Dr. Kolb said this patient was probably misdiagnosed as having Alzheimer’s – a problem that continues to plague both clinicians and researchers.
"We don’t have numbers of how many people are misdiagnosed every year, but we do know it’s a big problem," Heather Snyder, Ph.D., director of medical and scientific operations for the Alzheimer’s Association, said in an interview. Misdiagnosis is "not only a mentally difficult thing to go through, but it can potentially keep someone from getting the correct care."
In vivo imaging of Alzheimer’s brain pathology is now held to be an imperative for moving Alzheimer’s research forward. The first agent, Pittsburgh compound B (PiB), which binds to amyloid plaques, has virtually revolutionized research by allowing direct visualization of plaques in living patients. PiB has helped to build a new understanding of prodromal and presymptomatic Alzheimer’s, and it has contributed greatly to patient stratification in clinical trials and to the ability to track therapeutic response.
But PiB is not particularly useful in a clinical setting due to its short, 20-minute half-life. Florbetapir (Amyvid), the investigational PET radioligand florbetaben, and [F-18]T808 all use the same fluorine isotope, making their half-lives 120 minutes and much more friendly for clinical use. [F-18]T808 reaches a plateau of action at about 40 minutes post injection, Dr. Kolb said.
Avid performed its study under an Investigational New Drug designation granted last year, Dr. Kolb said. The company will continue to accrue patients toward an approval request, but it didn’t speculate as to how long that might take, he noted.
[F-18]T808 was being developed by Siemens Medical Solutions USA until last April, when Avid, a subsidiary of Eli Lilly, purchased it along with another investigational tau radiotracer.
The other agent is named [F-18]T807, Lilly spokesperson Eva Catherine Groves said in an interview. She noted that "both have reported similar results and, as such, we plan to complete additional development work on both tracers prior to selecting at least one for advancement into our Alzheimer’s research and development programs."
Dr. Kolb is an employee of Avid Radiopharmaceuticals.
[email protected]
On Twitter @Alz_Gal
BOSTON – The first investigational tau radioligand for PET imaging has identified pathologic tangles in the brain of a man who was confirmed postmortem to have Alzheimer’s disease.
The patient who died was 1 of 12 participants in a study of the agent, [F-18]T808, and his imaging and neuropathologic results show that the agent binds exclusively to tau deposits, Hartmuth Kolb, Ph.D., said at a press conference at the Alzheimer’s Association International Conference 2013.
The 85-year-old man died of a pulmonary embolism 2 weeks after his [F-18]T808 scan. His death was unrelated to his diagnosis or to the imaging agent, Dr. Kolb said. Avid Radiopharmaceuticals, the company developing the tracer, obtained the brain for autopsy. The examination showed extensive tau tangles in exactly the same areas identified in a PET scan using [F-18]T808. Tau tangles show as red in the PET images; the warmer the shade, the more dense the tangles.
"The results were quite striking," said Dr. Kolb, senior vice president of research at Avid. "The Braak stage was a 5 or 6, which corresponded with a Mini-Mental State Examination [MMSE] score of 15. On the scan, tau was all over the brain except for a sparing of the sensorimotor cortex, and that was exactly what we had found," in the postmortem exam.
The first human imaging cohort comprised nine patients with diagnosed Alzheimer’s and three healthy control subjects. Investigators compared the in vivo PET images with immunohistochemistry and with staining from fluorescent tau ligand T557, which is used to identify the protein in postmortem exams.
In patients diagnosed with Alzheimer’s, the [F-18]T808 scan showed strong positive correlations with the clinical exam in the temporal lobe, frontal lobe, parietal lobe, and hippocampus. The posterior cingulate gyrus and putamen were relatively spared, and the cerebellum was negative. Patients with an MMSE score of 24 showed moderate tau deposition. Scans of three patients who had an MMSE score of 19, 14, and 3, respectively, showed a progressively denser tau concentration.
There was no significant tau accumulation in any of the three healthy control subjects.
There was one outlier – a patient with an MMSE score of 12 who showed only a very small amount of tau. Dr. Kolb said this patient was probably misdiagnosed as having Alzheimer’s – a problem that continues to plague both clinicians and researchers.
"We don’t have numbers of how many people are misdiagnosed every year, but we do know it’s a big problem," Heather Snyder, Ph.D., director of medical and scientific operations for the Alzheimer’s Association, said in an interview. Misdiagnosis is "not only a mentally difficult thing to go through, but it can potentially keep someone from getting the correct care."
In vivo imaging of Alzheimer’s brain pathology is now held to be an imperative for moving Alzheimer’s research forward. The first agent, Pittsburgh compound B (PiB), which binds to amyloid plaques, has virtually revolutionized research by allowing direct visualization of plaques in living patients. PiB has helped to build a new understanding of prodromal and presymptomatic Alzheimer’s, and it has contributed greatly to patient stratification in clinical trials and to the ability to track therapeutic response.
But PiB is not particularly useful in a clinical setting due to its short, 20-minute half-life. Florbetapir (Amyvid), the investigational PET radioligand florbetaben, and [F-18]T808 all use the same fluorine isotope, making their half-lives 120 minutes and much more friendly for clinical use. [F-18]T808 reaches a plateau of action at about 40 minutes post injection, Dr. Kolb said.
Avid performed its study under an Investigational New Drug designation granted last year, Dr. Kolb said. The company will continue to accrue patients toward an approval request, but it didn’t speculate as to how long that might take, he noted.
[F-18]T808 was being developed by Siemens Medical Solutions USA until last April, when Avid, a subsidiary of Eli Lilly, purchased it along with another investigational tau radiotracer.
The other agent is named [F-18]T807, Lilly spokesperson Eva Catherine Groves said in an interview. She noted that "both have reported similar results and, as such, we plan to complete additional development work on both tracers prior to selecting at least one for advancement into our Alzheimer’s research and development programs."
Dr. Kolb is an employee of Avid Radiopharmaceuticals.
[email protected]
On Twitter @Alz_Gal
AT AAIC 2013
Major finding: The results of in vivo tau tangle PET imaging with [F-18]T808 in a patient diagnosed with Alzheimer’s were later confirmed in a postmortem neuropathologic analysis.
Data source: A study of PET imaging with [F-18]T808 in nine patients diagnosed with Alzheimer’s disease and three healthy controls .
Disclosures: Dr. Kolb is an employee of Avid Radiopharmaceuticals.
IVIG falls down at GAP study’s finish line
BOSTON – Intravenous immunoglobulin is down, but may not be completely out as a potential treatment for Alzheimer’s disease.
In a disappointing follow-up to last year’s interim data, patients with mild to moderate disease who received intravenous immunoglobulin (IVIG) experienced no significant cognitive or functional benefits relative to the placebo group, Dr. Norman Relkin reported at a press briefing at the Alzheimer’s Association International Conference 2013.
But investigators in the phase III Gammaglobulin Alzheimer’s Partnership (GAP) study saw some positive results in cognition and function in the subgroup of patients with moderate disease and in those who carried the high-risk apolipoprotein E epsilon-4 (APOE epsilon-4) allele. Those hints of benefit are enough to justify keeping IVIG on the table, said Dr. Relkin, the study’s lead investigator.
"It’s too soon to be planning another study," he said. "I think it will be some time before we know how to move forward. But I’m optimistic that there are some signals in these data that are worthy of further study."
GAP randomized 390 patients with mild to moderate Alzheimer’s to placebo or biweekly infusions of 200 or 400 mg/kg IVIG for 18 months. A total of 309 completed the per protocol treatment.
Six-month results, reported at last year’s AAIC meeting, were positive. Those who received IVIG showed significantly slower cognitive decline than did those who got placebo. Patients who received the higher dose experienced virtually no decline from their baseline measurements.
But last May, Baxter, the study sponsor, reported negative results from GAP. After the full 18 months of treatment, there were no significant differences in the rate of cognitive decline between patients in the placebo or active treatment arms.
The results also did not indicate a statistically significant change in functional ability in comparison with placebo.
However, patients with moderate disease and those who were positive for APOE epsilon-4 appeared to reap some benefits from IVIG, said Dr. Relkin, a neurologist at New York–Presbyterian Hospital/Weill Cornell Medical Center, New York.
IVIG treatment in patients with moderate disease showed trends toward benefits relative to placebo on the cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-Cog) and the Modified Mini-Mental State Examination (MMSE), with P values of .083 and .067, respectively. Dr. Relkin noted that these subgroup analyses were not powered to detect such differences. APOE epsilon-4 carriers also had numerically better scores on the MMSE than did placebo patients, with a P value of .012.
A breakdown of APOE epsilon-4 patients by drug dosage indicated that those taking 400 mg/kg did better on tests of executive function (Trails B, clock draw, and digit symbol) than did those taking 200 mg/kg. Again, this analysis wasn’t powered to show statistical significance.
GAP also found some biomarker differences in patients treated with IVIG, Dr. Relkin noted. These included:
• A statistically significant, dose-dependent reduction in plasma beta-amyloid 42 levels (but not beta-amyloid 40).
• Statistically significant, dose-dependent increases in anti-oligomer and anti-fibril antibodies in cerebrospinal fluid and plasma.
• A reduction in brain fibrillar amyloid as measured by florbetapir PET scan in those who received the larger IVIG dose.
There was no effect on tau and phosphorylated tau levels in cerebrospinal fluid.
It’s tough to pinpoint just why IVIG didn’t live up to its 6-month promise, Dr. Relkin said. One reason might lie in trial design and patient selection – problems that continue to plague Alzheimer’s treatment studies.
"In these large clinical trials, a certain number of patients deemed to have Alzheimer’s actually don’t have it. A therapy designed to be effective in all disease stages is most likely to show benefit in those with moderate disease and in [APOE epsilon-4] carriers, because those are the ones most likely to be accurately diagnosed."
The positive subgroup findings might also be simply a "statistical fluke," he said.
"This was a pivotal trial and could have been used for drug approval, if the results had been positive. But there is a difference between a negative study and a failed study. A failed study is one that gives you no answer to the question that was asked. A negative study is one that answers that question – and in this case, the answer was ‘no.’ That’s one way in which this trial did succeed. It gave us a clear, unequivocal answer that we can interpret and translate into a clinically meaningful result."
The GAP study was sponsored by Baxter. Dr. Relkin receives study support from Baxter.
On Twitter @Alz_Gal
BOSTON – Intravenous immunoglobulin is down, but may not be completely out as a potential treatment for Alzheimer’s disease.
In a disappointing follow-up to last year’s interim data, patients with mild to moderate disease who received intravenous immunoglobulin (IVIG) experienced no significant cognitive or functional benefits relative to the placebo group, Dr. Norman Relkin reported at a press briefing at the Alzheimer’s Association International Conference 2013.
But investigators in the phase III Gammaglobulin Alzheimer’s Partnership (GAP) study saw some positive results in cognition and function in the subgroup of patients with moderate disease and in those who carried the high-risk apolipoprotein E epsilon-4 (APOE epsilon-4) allele. Those hints of benefit are enough to justify keeping IVIG on the table, said Dr. Relkin, the study’s lead investigator.
"It’s too soon to be planning another study," he said. "I think it will be some time before we know how to move forward. But I’m optimistic that there are some signals in these data that are worthy of further study."
GAP randomized 390 patients with mild to moderate Alzheimer’s to placebo or biweekly infusions of 200 or 400 mg/kg IVIG for 18 months. A total of 309 completed the per protocol treatment.
Six-month results, reported at last year’s AAIC meeting, were positive. Those who received IVIG showed significantly slower cognitive decline than did those who got placebo. Patients who received the higher dose experienced virtually no decline from their baseline measurements.
But last May, Baxter, the study sponsor, reported negative results from GAP. After the full 18 months of treatment, there were no significant differences in the rate of cognitive decline between patients in the placebo or active treatment arms.
The results also did not indicate a statistically significant change in functional ability in comparison with placebo.
However, patients with moderate disease and those who were positive for APOE epsilon-4 appeared to reap some benefits from IVIG, said Dr. Relkin, a neurologist at New York–Presbyterian Hospital/Weill Cornell Medical Center, New York.
IVIG treatment in patients with moderate disease showed trends toward benefits relative to placebo on the cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-Cog) and the Modified Mini-Mental State Examination (MMSE), with P values of .083 and .067, respectively. Dr. Relkin noted that these subgroup analyses were not powered to detect such differences. APOE epsilon-4 carriers also had numerically better scores on the MMSE than did placebo patients, with a P value of .012.
A breakdown of APOE epsilon-4 patients by drug dosage indicated that those taking 400 mg/kg did better on tests of executive function (Trails B, clock draw, and digit symbol) than did those taking 200 mg/kg. Again, this analysis wasn’t powered to show statistical significance.
GAP also found some biomarker differences in patients treated with IVIG, Dr. Relkin noted. These included:
• A statistically significant, dose-dependent reduction in plasma beta-amyloid 42 levels (but not beta-amyloid 40).
• Statistically significant, dose-dependent increases in anti-oligomer and anti-fibril antibodies in cerebrospinal fluid and plasma.
• A reduction in brain fibrillar amyloid as measured by florbetapir PET scan in those who received the larger IVIG dose.
There was no effect on tau and phosphorylated tau levels in cerebrospinal fluid.
It’s tough to pinpoint just why IVIG didn’t live up to its 6-month promise, Dr. Relkin said. One reason might lie in trial design and patient selection – problems that continue to plague Alzheimer’s treatment studies.
"In these large clinical trials, a certain number of patients deemed to have Alzheimer’s actually don’t have it. A therapy designed to be effective in all disease stages is most likely to show benefit in those with moderate disease and in [APOE epsilon-4] carriers, because those are the ones most likely to be accurately diagnosed."
The positive subgroup findings might also be simply a "statistical fluke," he said.
"This was a pivotal trial and could have been used for drug approval, if the results had been positive. But there is a difference between a negative study and a failed study. A failed study is one that gives you no answer to the question that was asked. A negative study is one that answers that question – and in this case, the answer was ‘no.’ That’s one way in which this trial did succeed. It gave us a clear, unequivocal answer that we can interpret and translate into a clinically meaningful result."
The GAP study was sponsored by Baxter. Dr. Relkin receives study support from Baxter.
On Twitter @Alz_Gal
BOSTON – Intravenous immunoglobulin is down, but may not be completely out as a potential treatment for Alzheimer’s disease.
In a disappointing follow-up to last year’s interim data, patients with mild to moderate disease who received intravenous immunoglobulin (IVIG) experienced no significant cognitive or functional benefits relative to the placebo group, Dr. Norman Relkin reported at a press briefing at the Alzheimer’s Association International Conference 2013.
But investigators in the phase III Gammaglobulin Alzheimer’s Partnership (GAP) study saw some positive results in cognition and function in the subgroup of patients with moderate disease and in those who carried the high-risk apolipoprotein E epsilon-4 (APOE epsilon-4) allele. Those hints of benefit are enough to justify keeping IVIG on the table, said Dr. Relkin, the study’s lead investigator.
"It’s too soon to be planning another study," he said. "I think it will be some time before we know how to move forward. But I’m optimistic that there are some signals in these data that are worthy of further study."
GAP randomized 390 patients with mild to moderate Alzheimer’s to placebo or biweekly infusions of 200 or 400 mg/kg IVIG for 18 months. A total of 309 completed the per protocol treatment.
Six-month results, reported at last year’s AAIC meeting, were positive. Those who received IVIG showed significantly slower cognitive decline than did those who got placebo. Patients who received the higher dose experienced virtually no decline from their baseline measurements.
But last May, Baxter, the study sponsor, reported negative results from GAP. After the full 18 months of treatment, there were no significant differences in the rate of cognitive decline between patients in the placebo or active treatment arms.
The results also did not indicate a statistically significant change in functional ability in comparison with placebo.
However, patients with moderate disease and those who were positive for APOE epsilon-4 appeared to reap some benefits from IVIG, said Dr. Relkin, a neurologist at New York–Presbyterian Hospital/Weill Cornell Medical Center, New York.
IVIG treatment in patients with moderate disease showed trends toward benefits relative to placebo on the cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-Cog) and the Modified Mini-Mental State Examination (MMSE), with P values of .083 and .067, respectively. Dr. Relkin noted that these subgroup analyses were not powered to detect such differences. APOE epsilon-4 carriers also had numerically better scores on the MMSE than did placebo patients, with a P value of .012.
A breakdown of APOE epsilon-4 patients by drug dosage indicated that those taking 400 mg/kg did better on tests of executive function (Trails B, clock draw, and digit symbol) than did those taking 200 mg/kg. Again, this analysis wasn’t powered to show statistical significance.
GAP also found some biomarker differences in patients treated with IVIG, Dr. Relkin noted. These included:
• A statistically significant, dose-dependent reduction in plasma beta-amyloid 42 levels (but not beta-amyloid 40).
• Statistically significant, dose-dependent increases in anti-oligomer and anti-fibril antibodies in cerebrospinal fluid and plasma.
• A reduction in brain fibrillar amyloid as measured by florbetapir PET scan in those who received the larger IVIG dose.
There was no effect on tau and phosphorylated tau levels in cerebrospinal fluid.
It’s tough to pinpoint just why IVIG didn’t live up to its 6-month promise, Dr. Relkin said. One reason might lie in trial design and patient selection – problems that continue to plague Alzheimer’s treatment studies.
"In these large clinical trials, a certain number of patients deemed to have Alzheimer’s actually don’t have it. A therapy designed to be effective in all disease stages is most likely to show benefit in those with moderate disease and in [APOE epsilon-4] carriers, because those are the ones most likely to be accurately diagnosed."
The positive subgroup findings might also be simply a "statistical fluke," he said.
"This was a pivotal trial and could have been used for drug approval, if the results had been positive. But there is a difference between a negative study and a failed study. A failed study is one that gives you no answer to the question that was asked. A negative study is one that answers that question – and in this case, the answer was ‘no.’ That’s one way in which this trial did succeed. It gave us a clear, unequivocal answer that we can interpret and translate into a clinically meaningful result."
The GAP study was sponsored by Baxter. Dr. Relkin receives study support from Baxter.
On Twitter @Alz_Gal
AT AAIC 2013
Major finding: The use of IVIG led to no significant cognitive or functional benefits in patients with mild to moderate Alzheimer’s disease.
Data source: GAP randomized 390 patients to placebo or to IVIG at doses of 200 or 400 mg/kg every 2 weeks for 18 months.
Disclosures: The GAP study was sponsored by Baxter. Dr. Relkin receives study support from Baxter.
Subanalysis spurs continued study of solanezumab for Alzheimer's
BOSTON – Another Eli Lilly–sponsored trial of solanezumab is in the works, bolstered by a subanalysis of two failed studies that picked up an efficacy signal among patients with mild Alzheimer’s.
"Although our primary objectives in those trials were not met, this prespecified secondary analysis did show a significant benefit" for patients who took the drug, compared with those who took placebo, Christopher Carlson, Ph.D., said at the Alzheimer’s Association International Conference 2013.
Dr. Carlson, an employee of Eli Lilly, presented the pooled analysis of EXPEDITION I and II. It found a 34% decline in cognitive slowing and a 17% decline in functional slowing, compared with placebo, among the 2,042 patients with mild disease.
Angina was the only adverse event seen in more than 1% of patients, which was also significantly different between the groups; it occurred in 1% of the solanezumab group and in 0.2% of the placebo group.
Amyloid-related imaging abnormalities (ARIAs) were numerically more common among those taking solanezumab, but the between-group differences were not statistically significant. Any increase of ARIA-H (microhemorrhage) in size or number over baseline occurred in 9% of the solanezumab patients and 7.5% of the placebo patients. ARIA-E, a measure of vasogenic edema and/or sulcal effusion, developed in 1.1% of those taking solanezumab and 0.5% of those taking placebo.
Dr. Carlson said the combination of some cognitive benefit and the low rate of adverse events was enough to prompt further study of the monoclonal antibody.
"The cognitive and functional effects and safety profile provide a favorable risk/benefit level. So we are now in the process of planning a large, multinational phase III trial for patients with mild Alzheimer’s and evidence of amyloid pathology."
Lilly announced its plans for the study during a July 12 webcast.
The new study – EXPEDITION III – will enroll 2,100 patients with mild Alzheimer’s. Treatment will consist of one 400-mg infusion of solanezumab or placebo every 4 weeks. Like EXPEDITION I and II, EXPEDITION III will run for 80 weeks and will be followed by an open-label extension study.
Co–primary endpoints will be the Alzheimer’s Disease Assessment Scale – cognitive subscale (ADAS-cog)-14 and the Alzheimer’s Disease Cooperative Study Activities of Daily Living (ADAS-ADL), and Instrumental Activities of Daily Living scales (IADL). Secondary endpoints include a variety of cognitive, functional, and quality of life measures, as well as changes in amyloid brain deposition as measured by florbetapir PET scan, changes in CSF biomarkers, and volumetric brain changes as measured on magnetic resonance imaging.
The trial design of EXPEDITION III includes a few changes that Lilly researchers hope will decrease noise in the data, making it easier to pinpoint any benefits the drug may confer. The failed EXPEDITION trials contained an unknown number of patients with mild disease who may not have had amyloid pathology – a problem that could have diluted the drug’s positive effects, Dr. Eric Siemers said on the Lilly conference call.
In those studies, "We tested a small subgroup of the mild patients with florbetapir PET. Among them, almost 26% were negative for amyloid pathology," said Dr. Siemers, senior medical director of the company’s Alzheimer’s disease team. It’s doubtful that an antibody designed to eliminate amyloid plaques could benefit a patient without such deposits, he said.
Another admission qualification of the new study – a Mini Mental State Examination score of 20-26 – will further help ensure that only patients with mild Alzheimer’s enter the trial, he added.
The tightened entry requirements mean that more patients will have to be screened for entry, Dr. Siemers noted. "The screen failure rate for EXPEDITION I and II was just under 25%. In EXPEDITION III it could be up to 50%." That means it could take 22 months to find the full complement of 2,100 patients.
"This would place the last patient visit around the end of 2016," he said.
Eli Lilly is the sponsor of all the solanezumab EXPEDITION trials.
BOSTON – Another Eli Lilly–sponsored trial of solanezumab is in the works, bolstered by a subanalysis of two failed studies that picked up an efficacy signal among patients with mild Alzheimer’s.
"Although our primary objectives in those trials were not met, this prespecified secondary analysis did show a significant benefit" for patients who took the drug, compared with those who took placebo, Christopher Carlson, Ph.D., said at the Alzheimer’s Association International Conference 2013.
Dr. Carlson, an employee of Eli Lilly, presented the pooled analysis of EXPEDITION I and II. It found a 34% decline in cognitive slowing and a 17% decline in functional slowing, compared with placebo, among the 2,042 patients with mild disease.
Angina was the only adverse event seen in more than 1% of patients, which was also significantly different between the groups; it occurred in 1% of the solanezumab group and in 0.2% of the placebo group.
Amyloid-related imaging abnormalities (ARIAs) were numerically more common among those taking solanezumab, but the between-group differences were not statistically significant. Any increase of ARIA-H (microhemorrhage) in size or number over baseline occurred in 9% of the solanezumab patients and 7.5% of the placebo patients. ARIA-E, a measure of vasogenic edema and/or sulcal effusion, developed in 1.1% of those taking solanezumab and 0.5% of those taking placebo.
Dr. Carlson said the combination of some cognitive benefit and the low rate of adverse events was enough to prompt further study of the monoclonal antibody.
"The cognitive and functional effects and safety profile provide a favorable risk/benefit level. So we are now in the process of planning a large, multinational phase III trial for patients with mild Alzheimer’s and evidence of amyloid pathology."
Lilly announced its plans for the study during a July 12 webcast.
The new study – EXPEDITION III – will enroll 2,100 patients with mild Alzheimer’s. Treatment will consist of one 400-mg infusion of solanezumab or placebo every 4 weeks. Like EXPEDITION I and II, EXPEDITION III will run for 80 weeks and will be followed by an open-label extension study.
Co–primary endpoints will be the Alzheimer’s Disease Assessment Scale – cognitive subscale (ADAS-cog)-14 and the Alzheimer’s Disease Cooperative Study Activities of Daily Living (ADAS-ADL), and Instrumental Activities of Daily Living scales (IADL). Secondary endpoints include a variety of cognitive, functional, and quality of life measures, as well as changes in amyloid brain deposition as measured by florbetapir PET scan, changes in CSF biomarkers, and volumetric brain changes as measured on magnetic resonance imaging.
The trial design of EXPEDITION III includes a few changes that Lilly researchers hope will decrease noise in the data, making it easier to pinpoint any benefits the drug may confer. The failed EXPEDITION trials contained an unknown number of patients with mild disease who may not have had amyloid pathology – a problem that could have diluted the drug’s positive effects, Dr. Eric Siemers said on the Lilly conference call.
In those studies, "We tested a small subgroup of the mild patients with florbetapir PET. Among them, almost 26% were negative for amyloid pathology," said Dr. Siemers, senior medical director of the company’s Alzheimer’s disease team. It’s doubtful that an antibody designed to eliminate amyloid plaques could benefit a patient without such deposits, he said.
Another admission qualification of the new study – a Mini Mental State Examination score of 20-26 – will further help ensure that only patients with mild Alzheimer’s enter the trial, he added.
The tightened entry requirements mean that more patients will have to be screened for entry, Dr. Siemers noted. "The screen failure rate for EXPEDITION I and II was just under 25%. In EXPEDITION III it could be up to 50%." That means it could take 22 months to find the full complement of 2,100 patients.
"This would place the last patient visit around the end of 2016," he said.
Eli Lilly is the sponsor of all the solanezumab EXPEDITION trials.
BOSTON – Another Eli Lilly–sponsored trial of solanezumab is in the works, bolstered by a subanalysis of two failed studies that picked up an efficacy signal among patients with mild Alzheimer’s.
"Although our primary objectives in those trials were not met, this prespecified secondary analysis did show a significant benefit" for patients who took the drug, compared with those who took placebo, Christopher Carlson, Ph.D., said at the Alzheimer’s Association International Conference 2013.
Dr. Carlson, an employee of Eli Lilly, presented the pooled analysis of EXPEDITION I and II. It found a 34% decline in cognitive slowing and a 17% decline in functional slowing, compared with placebo, among the 2,042 patients with mild disease.
Angina was the only adverse event seen in more than 1% of patients, which was also significantly different between the groups; it occurred in 1% of the solanezumab group and in 0.2% of the placebo group.
Amyloid-related imaging abnormalities (ARIAs) were numerically more common among those taking solanezumab, but the between-group differences were not statistically significant. Any increase of ARIA-H (microhemorrhage) in size or number over baseline occurred in 9% of the solanezumab patients and 7.5% of the placebo patients. ARIA-E, a measure of vasogenic edema and/or sulcal effusion, developed in 1.1% of those taking solanezumab and 0.5% of those taking placebo.
Dr. Carlson said the combination of some cognitive benefit and the low rate of adverse events was enough to prompt further study of the monoclonal antibody.
"The cognitive and functional effects and safety profile provide a favorable risk/benefit level. So we are now in the process of planning a large, multinational phase III trial for patients with mild Alzheimer’s and evidence of amyloid pathology."
Lilly announced its plans for the study during a July 12 webcast.
The new study – EXPEDITION III – will enroll 2,100 patients with mild Alzheimer’s. Treatment will consist of one 400-mg infusion of solanezumab or placebo every 4 weeks. Like EXPEDITION I and II, EXPEDITION III will run for 80 weeks and will be followed by an open-label extension study.
Co–primary endpoints will be the Alzheimer’s Disease Assessment Scale – cognitive subscale (ADAS-cog)-14 and the Alzheimer’s Disease Cooperative Study Activities of Daily Living (ADAS-ADL), and Instrumental Activities of Daily Living scales (IADL). Secondary endpoints include a variety of cognitive, functional, and quality of life measures, as well as changes in amyloid brain deposition as measured by florbetapir PET scan, changes in CSF biomarkers, and volumetric brain changes as measured on magnetic resonance imaging.
The trial design of EXPEDITION III includes a few changes that Lilly researchers hope will decrease noise in the data, making it easier to pinpoint any benefits the drug may confer. The failed EXPEDITION trials contained an unknown number of patients with mild disease who may not have had amyloid pathology – a problem that could have diluted the drug’s positive effects, Dr. Eric Siemers said on the Lilly conference call.
In those studies, "We tested a small subgroup of the mild patients with florbetapir PET. Among them, almost 26% were negative for amyloid pathology," said Dr. Siemers, senior medical director of the company’s Alzheimer’s disease team. It’s doubtful that an antibody designed to eliminate amyloid plaques could benefit a patient without such deposits, he said.
Another admission qualification of the new study – a Mini Mental State Examination score of 20-26 – will further help ensure that only patients with mild Alzheimer’s enter the trial, he added.
The tightened entry requirements mean that more patients will have to be screened for entry, Dr. Siemers noted. "The screen failure rate for EXPEDITION I and II was just under 25%. In EXPEDITION III it could be up to 50%." That means it could take 22 months to find the full complement of 2,100 patients.
"This would place the last patient visit around the end of 2016," he said.
Eli Lilly is the sponsor of all the solanezumab EXPEDITION trials.
AT AAIC 2013
Major finding: Solanezumab decreased cognitive decline by 34% and functional decline by 17% in a group of patients with mild Alzheimer’s disease.
Data source: A planned subgroup analysis of two randomized placebo-controlled trials, comprising 2,042 patients treated for 80 weeks.
Disclosures: Eli Lilly is the sponsor of all the solanezumab EXPEDITION trials. Dr. Christopher Carlson is a Lilly employee.
Insulin boosted memory in patients with high-risk Alzheimer's allele
BOSTON – Long-acting intranasal insulin imparted a significant memory boost to patients with mild cognitive impairment or Alzheimer’s disease who carried a high-risk allele for the disease in a randomized, placebo-controlled trial.
"It’s a very interesting result," principal study investigator Suzanne Craft, Ph.D., of Wake Forest University, Winston-Salem, N.C., said at the Alzheimer’s Association International Conference 2013. "It’s not what we have previously observed with regular insulin, and so far it’s very difficult to find a treatment that benefits these patients."
Intranasal insulin travels quickly up the trigeminal and olfactory nerves and penetrates the blood-brain barrier within 15-30 minutes. Dr. Craft has previously examined the effect of regular insulin delivered intranasally. In those studies, patients had improved cognition and cerebral glucose metabolism. The treatment also modulated levels of biomarkers in cerebrospinal fluid. But because regular insulin’s effect peaks shortly after administration and falls off over 8 hours, she and her colleagues reasoned that long-acting insulin might confer improved cognitive results. "The longer-acting insulins mimic basal insulin, which remains mildly elevated even postprandially," she said.
The trial, dubbed SNIFF-LONG (Study of Nasal Insulin to Fight Forgetfulness), gave 20 or 40 IU of long-acting insulin or placebo to 60 patients (mean age, 72 years) with mild cognitive impairment (MCI) or Alzheimer’s disease. Most (39) had a diagnosis of MCI, and the rest had Alzheimer’s. Patients had a mean Mini-Mental State Examination score of 26. Thirty carried the apolipoprotein E (apo E) epsilon 4 allele, putting them at a very high risk for developing Alzheimer’s.
The patients had an average insulin resistance index of 2.8. At baseline, 14 patients were taking an acetylcholinesterase inhibitor or memantine. The primary endpoints were scores on a test of composite memory; secondary endpoints included scores on pattern separation and visual memory counting tests. There was also a planned subgroup analysis that examined results by apo E epsilon 4 status.
Intranasal insulin and placebo were administered once a day for 3 weeks via a nasal delivery device that has been specially developed for these trials. The group underwent memory testing at baseline and after the treatment period.
Overall, there were no significant improvements on the primary endpoint measuring a composite of scores on verbal memory tests (the Story Recall Test and the Buschke Selective Reminding Test) in patients taking either the 20- or 40-IU dose.
However, in a planned subgroup analysis, patients carrying the apo E epsilon 4 allele performed significantly better than did those without it. Apo E epsilon 4 noncarriers declined about 1 point relative to their baseline score, while carriers improved about 1 point, said Dr. Craft, who conducted the trial when she was professor of psychiatry and behavioral sciences at the University of Washington, Seattle, and director of the Memory Disorders Clinic at the American Lakes division of the VA Puget Sound Health Care System. At Wake Forest, she is a professor of gerontology and geriatric medicine.
"This was a very robust result that characterized the vast majority of patients in each group," she noted.
The results were also examined by the level of insulin resistance. "In those with the highest insulin resistance, the 40-IU dose showed a significant benefit [of about 1 point]; and those without insulin resistance did not, and, in fact, showed memory decline" of about 1 point, she said.
Both patient groups responded well to the 40-IU dose in the secondary endpoints of scores on tests of pattern separation (Benton Visual Retention Rest-Recognition) and visual memory counting (Dot Counting Test). "There were no interactions with [apo E epsilon 4] status or insulin resistance. Instead we saw a nice, robust improvement on both the pattern separation and dot counting tests" for patients taking that dose.
Dr. Craft will soon be recruiting patients for a 1-year study called SNIFF. This will include 240 subjects randomized to 20 IU of insulin (Humilin) inhaled twice daily (total dose, 40 IU) or placebo. At the end of the first 12 months, insulin will be given open-label to all participants for an additional 6 months. Tests of cognition are planned at baseline and at 3, 6, 9, 12, and 15 months. Memory and changes in daily functioning will be assessed at 6, 12, and 18 months.
The study was sponsored by the University of Washington and the National Institute on Aging. Dr. Craft had no financial disclosures.
On Twitter @Alz_Gal
BOSTON – Long-acting intranasal insulin imparted a significant memory boost to patients with mild cognitive impairment or Alzheimer’s disease who carried a high-risk allele for the disease in a randomized, placebo-controlled trial.
"It’s a very interesting result," principal study investigator Suzanne Craft, Ph.D., of Wake Forest University, Winston-Salem, N.C., said at the Alzheimer’s Association International Conference 2013. "It’s not what we have previously observed with regular insulin, and so far it’s very difficult to find a treatment that benefits these patients."
Intranasal insulin travels quickly up the trigeminal and olfactory nerves and penetrates the blood-brain barrier within 15-30 minutes. Dr. Craft has previously examined the effect of regular insulin delivered intranasally. In those studies, patients had improved cognition and cerebral glucose metabolism. The treatment also modulated levels of biomarkers in cerebrospinal fluid. But because regular insulin’s effect peaks shortly after administration and falls off over 8 hours, she and her colleagues reasoned that long-acting insulin might confer improved cognitive results. "The longer-acting insulins mimic basal insulin, which remains mildly elevated even postprandially," she said.
The trial, dubbed SNIFF-LONG (Study of Nasal Insulin to Fight Forgetfulness), gave 20 or 40 IU of long-acting insulin or placebo to 60 patients (mean age, 72 years) with mild cognitive impairment (MCI) or Alzheimer’s disease. Most (39) had a diagnosis of MCI, and the rest had Alzheimer’s. Patients had a mean Mini-Mental State Examination score of 26. Thirty carried the apolipoprotein E (apo E) epsilon 4 allele, putting them at a very high risk for developing Alzheimer’s.
The patients had an average insulin resistance index of 2.8. At baseline, 14 patients were taking an acetylcholinesterase inhibitor or memantine. The primary endpoints were scores on a test of composite memory; secondary endpoints included scores on pattern separation and visual memory counting tests. There was also a planned subgroup analysis that examined results by apo E epsilon 4 status.
Intranasal insulin and placebo were administered once a day for 3 weeks via a nasal delivery device that has been specially developed for these trials. The group underwent memory testing at baseline and after the treatment period.
Overall, there were no significant improvements on the primary endpoint measuring a composite of scores on verbal memory tests (the Story Recall Test and the Buschke Selective Reminding Test) in patients taking either the 20- or 40-IU dose.
However, in a planned subgroup analysis, patients carrying the apo E epsilon 4 allele performed significantly better than did those without it. Apo E epsilon 4 noncarriers declined about 1 point relative to their baseline score, while carriers improved about 1 point, said Dr. Craft, who conducted the trial when she was professor of psychiatry and behavioral sciences at the University of Washington, Seattle, and director of the Memory Disorders Clinic at the American Lakes division of the VA Puget Sound Health Care System. At Wake Forest, she is a professor of gerontology and geriatric medicine.
"This was a very robust result that characterized the vast majority of patients in each group," she noted.
The results were also examined by the level of insulin resistance. "In those with the highest insulin resistance, the 40-IU dose showed a significant benefit [of about 1 point]; and those without insulin resistance did not, and, in fact, showed memory decline" of about 1 point, she said.
Both patient groups responded well to the 40-IU dose in the secondary endpoints of scores on tests of pattern separation (Benton Visual Retention Rest-Recognition) and visual memory counting (Dot Counting Test). "There were no interactions with [apo E epsilon 4] status or insulin resistance. Instead we saw a nice, robust improvement on both the pattern separation and dot counting tests" for patients taking that dose.
Dr. Craft will soon be recruiting patients for a 1-year study called SNIFF. This will include 240 subjects randomized to 20 IU of insulin (Humilin) inhaled twice daily (total dose, 40 IU) or placebo. At the end of the first 12 months, insulin will be given open-label to all participants for an additional 6 months. Tests of cognition are planned at baseline and at 3, 6, 9, 12, and 15 months. Memory and changes in daily functioning will be assessed at 6, 12, and 18 months.
The study was sponsored by the University of Washington and the National Institute on Aging. Dr. Craft had no financial disclosures.
On Twitter @Alz_Gal
BOSTON – Long-acting intranasal insulin imparted a significant memory boost to patients with mild cognitive impairment or Alzheimer’s disease who carried a high-risk allele for the disease in a randomized, placebo-controlled trial.
"It’s a very interesting result," principal study investigator Suzanne Craft, Ph.D., of Wake Forest University, Winston-Salem, N.C., said at the Alzheimer’s Association International Conference 2013. "It’s not what we have previously observed with regular insulin, and so far it’s very difficult to find a treatment that benefits these patients."
Intranasal insulin travels quickly up the trigeminal and olfactory nerves and penetrates the blood-brain barrier within 15-30 minutes. Dr. Craft has previously examined the effect of regular insulin delivered intranasally. In those studies, patients had improved cognition and cerebral glucose metabolism. The treatment also modulated levels of biomarkers in cerebrospinal fluid. But because regular insulin’s effect peaks shortly after administration and falls off over 8 hours, she and her colleagues reasoned that long-acting insulin might confer improved cognitive results. "The longer-acting insulins mimic basal insulin, which remains mildly elevated even postprandially," she said.
The trial, dubbed SNIFF-LONG (Study of Nasal Insulin to Fight Forgetfulness), gave 20 or 40 IU of long-acting insulin or placebo to 60 patients (mean age, 72 years) with mild cognitive impairment (MCI) or Alzheimer’s disease. Most (39) had a diagnosis of MCI, and the rest had Alzheimer’s. Patients had a mean Mini-Mental State Examination score of 26. Thirty carried the apolipoprotein E (apo E) epsilon 4 allele, putting them at a very high risk for developing Alzheimer’s.
The patients had an average insulin resistance index of 2.8. At baseline, 14 patients were taking an acetylcholinesterase inhibitor or memantine. The primary endpoints were scores on a test of composite memory; secondary endpoints included scores on pattern separation and visual memory counting tests. There was also a planned subgroup analysis that examined results by apo E epsilon 4 status.
Intranasal insulin and placebo were administered once a day for 3 weeks via a nasal delivery device that has been specially developed for these trials. The group underwent memory testing at baseline and after the treatment period.
Overall, there were no significant improvements on the primary endpoint measuring a composite of scores on verbal memory tests (the Story Recall Test and the Buschke Selective Reminding Test) in patients taking either the 20- or 40-IU dose.
However, in a planned subgroup analysis, patients carrying the apo E epsilon 4 allele performed significantly better than did those without it. Apo E epsilon 4 noncarriers declined about 1 point relative to their baseline score, while carriers improved about 1 point, said Dr. Craft, who conducted the trial when she was professor of psychiatry and behavioral sciences at the University of Washington, Seattle, and director of the Memory Disorders Clinic at the American Lakes division of the VA Puget Sound Health Care System. At Wake Forest, she is a professor of gerontology and geriatric medicine.
"This was a very robust result that characterized the vast majority of patients in each group," she noted.
The results were also examined by the level of insulin resistance. "In those with the highest insulin resistance, the 40-IU dose showed a significant benefit [of about 1 point]; and those without insulin resistance did not, and, in fact, showed memory decline" of about 1 point, she said.
Both patient groups responded well to the 40-IU dose in the secondary endpoints of scores on tests of pattern separation (Benton Visual Retention Rest-Recognition) and visual memory counting (Dot Counting Test). "There were no interactions with [apo E epsilon 4] status or insulin resistance. Instead we saw a nice, robust improvement on both the pattern separation and dot counting tests" for patients taking that dose.
Dr. Craft will soon be recruiting patients for a 1-year study called SNIFF. This will include 240 subjects randomized to 20 IU of insulin (Humilin) inhaled twice daily (total dose, 40 IU) or placebo. At the end of the first 12 months, insulin will be given open-label to all participants for an additional 6 months. Tests of cognition are planned at baseline and at 3, 6, 9, 12, and 15 months. Memory and changes in daily functioning will be assessed at 6, 12, and 18 months.
The study was sponsored by the University of Washington and the National Institute on Aging. Dr. Craft had no financial disclosures.
On Twitter @Alz_Gal
AT AAIC 2013
Major finding: Apo E epsilon 4 noncarriers declined about 1 point relative to their baseline score on a composite endpoint of verbal memory tests, while carriers improved about 1 point.
Data source: SNIFF-LONG included 60 patients with mild cognitive impairment or Alzheimer’s disease.
Disclosures: Dr. Suzanne Craft had no financial disclosures. The study was sponsored by the University of Washington and the National Institute on Aging.
BACE1 inhibitor markedly decreased CSF amyloid in Alzheimer’s
BOSTON – A drug that blocks the formation of two brain-toxic amyloid beta peptides cleared most of the protein fragments from cerebrospinal fluid in patients with mild to moderate Alzheimer’s disease who took it in a 7-day phase I study.
The CSF load of amyloid beta 40 (Abeta40) dropped by a mean of 90% and that of Abeta42 by a mean of 86%, compared with a placebo group, Dr. Mark S. Forman reported at the Alzheimer’s Association International Conference 2013.
The drug, called MK-8931, is an inhibitor of the beta-secretase 1 enzyme (BACE1), which is also known as beta-site amyloid precursor protein (APP) cleaving enzyme 1. It changes the point at which the APP splits into the Abeta40 and Abeta42 peptide fragments. Researchers hope this alteration might prevent the proteins from forming, and so prevent the buildup of amyloid brain plaques that is a diagnostic hallmark of Alzheimer’s disease.
The placebo-controlled study randomized 30 subjects to three dosage groups (12, 40, and 60 mg) of eight patients each and a placebo group of six patients. The medications were administered once daily for 7 days.
Patients underwent lumbar puncture at baseline to assess CSF levels of Abeta40 and Abeta42. Samples were also drawn hourly for 36 hours after the last dose was administered.
"There was a profound, steady-state reduction in CSF Abeta peptides," Dr. Forman said. The reductions were similar to the results of a prior safety study performed in 40 healthy young adults.
Adverse events reported in two or more subjects in at least one dose group included headache, dizziness, nausea, vomiting, insomnia, and back pain. All adverse events were generally mild to moderate in intensity and transient in duration. No dose-dependent increase in the incidence of adverse events was observed.
The study didn’t examine any cognitive outcomes, Dr. Forman said in an interview. Those will be assessed in the next phase of development of MK-8931 – a phase II/III study called EPOCH, which began recruiting subjects in December 2012. The 78-week trial aims to enroll up to 1,700 patients with mild to moderate Alzheimer’s, randomizing them to the 12-, 40-, and 60-mg doses.
EPOCH’s primary endpoints are changes in the cognitive subscale score of the ADAS-Cog (Alzheimer’s Disease Assessment Scale) and the change from baseline in the score of the ADCS-ADL (Alzheimer’s Disease Cooperative Study – Activities of Daily Living) scale.
So far, manipulating APP to inhibit toxic amyloid production has been difficult. Earlier this year, Eli Lilly pulled the plug on its investigational BACE1 inhibitor, LY2886721, because of abnormal liver enzymes identified during routine monitoring of subjects taking it.
Safety concerns also caused Lilly to yank semagacestat from development in 2010. Semagacestat was also designed to change the APP cleavage point, but it targeted gamma-secretase instead of beta-secretase. Although the drug decreased CSF levels of Abeta42 by up to 65%, patients who took it actually had a significantly greater decline in cognition and function than did the placebo group. They also had a slight increase in skin cancers.
"One thing we are really concerned about is the long-term data from the semagacestat trial, where people actually got worse instead of better," Dr. Forman said. "We don’t know if that’s a reflection of some off-target activity or reducing amyloid in the brain. What will long-term BACE inhibition do? It’s an unknown. But we think the potential upsides outweigh the risks with this compound."
In an interview, Maria Carillo, Ph.D., agreed. "I’m excited that BACE1 is being pursued as an earlier intervention in the amyloid process. It will help prove that the amyloid hypothesis is worth pursing and give us an additional amyloid strategy."
Because Alzheimer’s seems to deliver a number of pathologic hits, effectively treating it will call for a varied armamentarium, said Dr. Carillo, vice president of medical and scientific relations for the Alzheimer’s Association. "At the end of the day, we have to know how to tackle all of them – tau, amyloids, inflammation, and energy pathways."
Merck funded the study of MK-8931. Dr. Forman is the company’s director of clinical research.
On Twitter @Alz_Gal
BOSTON – A drug that blocks the formation of two brain-toxic amyloid beta peptides cleared most of the protein fragments from cerebrospinal fluid in patients with mild to moderate Alzheimer’s disease who took it in a 7-day phase I study.
The CSF load of amyloid beta 40 (Abeta40) dropped by a mean of 90% and that of Abeta42 by a mean of 86%, compared with a placebo group, Dr. Mark S. Forman reported at the Alzheimer’s Association International Conference 2013.
The drug, called MK-8931, is an inhibitor of the beta-secretase 1 enzyme (BACE1), which is also known as beta-site amyloid precursor protein (APP) cleaving enzyme 1. It changes the point at which the APP splits into the Abeta40 and Abeta42 peptide fragments. Researchers hope this alteration might prevent the proteins from forming, and so prevent the buildup of amyloid brain plaques that is a diagnostic hallmark of Alzheimer’s disease.
The placebo-controlled study randomized 30 subjects to three dosage groups (12, 40, and 60 mg) of eight patients each and a placebo group of six patients. The medications were administered once daily for 7 days.
Patients underwent lumbar puncture at baseline to assess CSF levels of Abeta40 and Abeta42. Samples were also drawn hourly for 36 hours after the last dose was administered.
"There was a profound, steady-state reduction in CSF Abeta peptides," Dr. Forman said. The reductions were similar to the results of a prior safety study performed in 40 healthy young adults.
Adverse events reported in two or more subjects in at least one dose group included headache, dizziness, nausea, vomiting, insomnia, and back pain. All adverse events were generally mild to moderate in intensity and transient in duration. No dose-dependent increase in the incidence of adverse events was observed.
The study didn’t examine any cognitive outcomes, Dr. Forman said in an interview. Those will be assessed in the next phase of development of MK-8931 – a phase II/III study called EPOCH, which began recruiting subjects in December 2012. The 78-week trial aims to enroll up to 1,700 patients with mild to moderate Alzheimer’s, randomizing them to the 12-, 40-, and 60-mg doses.
EPOCH’s primary endpoints are changes in the cognitive subscale score of the ADAS-Cog (Alzheimer’s Disease Assessment Scale) and the change from baseline in the score of the ADCS-ADL (Alzheimer’s Disease Cooperative Study – Activities of Daily Living) scale.
So far, manipulating APP to inhibit toxic amyloid production has been difficult. Earlier this year, Eli Lilly pulled the plug on its investigational BACE1 inhibitor, LY2886721, because of abnormal liver enzymes identified during routine monitoring of subjects taking it.
Safety concerns also caused Lilly to yank semagacestat from development in 2010. Semagacestat was also designed to change the APP cleavage point, but it targeted gamma-secretase instead of beta-secretase. Although the drug decreased CSF levels of Abeta42 by up to 65%, patients who took it actually had a significantly greater decline in cognition and function than did the placebo group. They also had a slight increase in skin cancers.
"One thing we are really concerned about is the long-term data from the semagacestat trial, where people actually got worse instead of better," Dr. Forman said. "We don’t know if that’s a reflection of some off-target activity or reducing amyloid in the brain. What will long-term BACE inhibition do? It’s an unknown. But we think the potential upsides outweigh the risks with this compound."
In an interview, Maria Carillo, Ph.D., agreed. "I’m excited that BACE1 is being pursued as an earlier intervention in the amyloid process. It will help prove that the amyloid hypothesis is worth pursing and give us an additional amyloid strategy."
Because Alzheimer’s seems to deliver a number of pathologic hits, effectively treating it will call for a varied armamentarium, said Dr. Carillo, vice president of medical and scientific relations for the Alzheimer’s Association. "At the end of the day, we have to know how to tackle all of them – tau, amyloids, inflammation, and energy pathways."
Merck funded the study of MK-8931. Dr. Forman is the company’s director of clinical research.
On Twitter @Alz_Gal
BOSTON – A drug that blocks the formation of two brain-toxic amyloid beta peptides cleared most of the protein fragments from cerebrospinal fluid in patients with mild to moderate Alzheimer’s disease who took it in a 7-day phase I study.
The CSF load of amyloid beta 40 (Abeta40) dropped by a mean of 90% and that of Abeta42 by a mean of 86%, compared with a placebo group, Dr. Mark S. Forman reported at the Alzheimer’s Association International Conference 2013.
The drug, called MK-8931, is an inhibitor of the beta-secretase 1 enzyme (BACE1), which is also known as beta-site amyloid precursor protein (APP) cleaving enzyme 1. It changes the point at which the APP splits into the Abeta40 and Abeta42 peptide fragments. Researchers hope this alteration might prevent the proteins from forming, and so prevent the buildup of amyloid brain plaques that is a diagnostic hallmark of Alzheimer’s disease.
The placebo-controlled study randomized 30 subjects to three dosage groups (12, 40, and 60 mg) of eight patients each and a placebo group of six patients. The medications were administered once daily for 7 days.
Patients underwent lumbar puncture at baseline to assess CSF levels of Abeta40 and Abeta42. Samples were also drawn hourly for 36 hours after the last dose was administered.
"There was a profound, steady-state reduction in CSF Abeta peptides," Dr. Forman said. The reductions were similar to the results of a prior safety study performed in 40 healthy young adults.
Adverse events reported in two or more subjects in at least one dose group included headache, dizziness, nausea, vomiting, insomnia, and back pain. All adverse events were generally mild to moderate in intensity and transient in duration. No dose-dependent increase in the incidence of adverse events was observed.
The study didn’t examine any cognitive outcomes, Dr. Forman said in an interview. Those will be assessed in the next phase of development of MK-8931 – a phase II/III study called EPOCH, which began recruiting subjects in December 2012. The 78-week trial aims to enroll up to 1,700 patients with mild to moderate Alzheimer’s, randomizing them to the 12-, 40-, and 60-mg doses.
EPOCH’s primary endpoints are changes in the cognitive subscale score of the ADAS-Cog (Alzheimer’s Disease Assessment Scale) and the change from baseline in the score of the ADCS-ADL (Alzheimer’s Disease Cooperative Study – Activities of Daily Living) scale.
So far, manipulating APP to inhibit toxic amyloid production has been difficult. Earlier this year, Eli Lilly pulled the plug on its investigational BACE1 inhibitor, LY2886721, because of abnormal liver enzymes identified during routine monitoring of subjects taking it.
Safety concerns also caused Lilly to yank semagacestat from development in 2010. Semagacestat was also designed to change the APP cleavage point, but it targeted gamma-secretase instead of beta-secretase. Although the drug decreased CSF levels of Abeta42 by up to 65%, patients who took it actually had a significantly greater decline in cognition and function than did the placebo group. They also had a slight increase in skin cancers.
"One thing we are really concerned about is the long-term data from the semagacestat trial, where people actually got worse instead of better," Dr. Forman said. "We don’t know if that’s a reflection of some off-target activity or reducing amyloid in the brain. What will long-term BACE inhibition do? It’s an unknown. But we think the potential upsides outweigh the risks with this compound."
In an interview, Maria Carillo, Ph.D., agreed. "I’m excited that BACE1 is being pursued as an earlier intervention in the amyloid process. It will help prove that the amyloid hypothesis is worth pursing and give us an additional amyloid strategy."
Because Alzheimer’s seems to deliver a number of pathologic hits, effectively treating it will call for a varied armamentarium, said Dr. Carillo, vice president of medical and scientific relations for the Alzheimer’s Association. "At the end of the day, we have to know how to tackle all of them – tau, amyloids, inflammation, and energy pathways."
Merck funded the study of MK-8931. Dr. Forman is the company’s director of clinical research.
On Twitter @Alz_Gal
AT AAIC 2013
Major finding: MK-8931 decreased mean cerebrospinal fluid levels of amyloid beta 40 by 90% and amyloid beta 42 by 86%.
Data source: A randomized, placebo-controlled study of 30 patients with mild to moderate Alzheimer’s disease.
Disclosures: Merck funded the study of MK-8931. Dr. Forman is the company’s director of clinical research.
Structural brain abnormalities predict enduring seizures
MONTREAL – Early imaging may be the best clue about seizure remission in children with early intractable seizures, according to findings from a population-based, retrospective cohort study.
Children who had uncontrolled seizures at 2 years and structural brain abnormalities had a 9% rate of becoming seizure free during a median follow-up period of 12 years. However, 60% of those with structurally normal brains eventually did become seizure free, despite an early history of intractability, Dr. Elaine Wirrell said at the 30th International Epilepsy Congress.
"While a significant minority of children with early medical intractability ultimately achieved seizure control without surgery, those with an abnormal imaging study did poorly," said Dr. Wirrell of the Mayo Clinic, Rochester, Minn. "For this subgroup, early surgical intervention is strongly advised to limit the comorbidities of ongoing, intractable seizures. Conversely, a cautious approach is suggested for those with normal imaging, as most will remit with time."
She and her colleagues examined medical intractability in a group of 381 children who were diagnosed with epilepsy during 1980-2009 and who were followed for a median of 12 years. Of these, 75 (20%) showed early medical intractability, defined as failing at least two antiepileptic medications or continuing to have seizures more often than every 6 months.
The children were a mean of 7 years old when their epilepsy began. Most (75%) had a focal onset, while 23% had a generalized onset. Onset for the remainder was unknown. The etiology was unknown in 54%, genetic in 23%, and structural or metabolic in the remainder.
Overall, 74 children underwent either MRI or CT, and a structural brain abnormality was present in 35. There were 40 children who had structurally normal brains. (One patient was known to have a familial absence syndrome ruling out a structural abnormality and did not undergo brain imaging.)
After a median follow-up of 12 years, 48 patients were still having seizures or had undergone brain surgery; 32 of these initially had abnormal brain imaging. Only three children with a structural brain abnormality were seizure free off or on medications at follow-up. A total of 24 who had normal brain structure eventually became seizure free.
More than half of the patients with an abnormality were surgical candidates because of the presence of a single focal or hemispheric lesion (16), tuberous sclerosis (3), or mesio-temporal sclerosis with periventricular leukomalacia (1). Of 16 who underwent surgery, 7 became seizure free. Five were able to discontinue all antiepileptic drugs.
In a multivariate analysis, neuroimaging was a significant predictor of seizure outcome.
"Imaging that is done properly is important," Dr. Wirrell said in an interview. "There is value with going on with a medication trial, but if one fails, it’s reasonable to have the discussion of possible surgery as we are starting the next."
Dr. Wirrell had no financial disclosures.
MONTREAL – Early imaging may be the best clue about seizure remission in children with early intractable seizures, according to findings from a population-based, retrospective cohort study.
Children who had uncontrolled seizures at 2 years and structural brain abnormalities had a 9% rate of becoming seizure free during a median follow-up period of 12 years. However, 60% of those with structurally normal brains eventually did become seizure free, despite an early history of intractability, Dr. Elaine Wirrell said at the 30th International Epilepsy Congress.
"While a significant minority of children with early medical intractability ultimately achieved seizure control without surgery, those with an abnormal imaging study did poorly," said Dr. Wirrell of the Mayo Clinic, Rochester, Minn. "For this subgroup, early surgical intervention is strongly advised to limit the comorbidities of ongoing, intractable seizures. Conversely, a cautious approach is suggested for those with normal imaging, as most will remit with time."
She and her colleagues examined medical intractability in a group of 381 children who were diagnosed with epilepsy during 1980-2009 and who were followed for a median of 12 years. Of these, 75 (20%) showed early medical intractability, defined as failing at least two antiepileptic medications or continuing to have seizures more often than every 6 months.
The children were a mean of 7 years old when their epilepsy began. Most (75%) had a focal onset, while 23% had a generalized onset. Onset for the remainder was unknown. The etiology was unknown in 54%, genetic in 23%, and structural or metabolic in the remainder.
Overall, 74 children underwent either MRI or CT, and a structural brain abnormality was present in 35. There were 40 children who had structurally normal brains. (One patient was known to have a familial absence syndrome ruling out a structural abnormality and did not undergo brain imaging.)
After a median follow-up of 12 years, 48 patients were still having seizures or had undergone brain surgery; 32 of these initially had abnormal brain imaging. Only three children with a structural brain abnormality were seizure free off or on medications at follow-up. A total of 24 who had normal brain structure eventually became seizure free.
More than half of the patients with an abnormality were surgical candidates because of the presence of a single focal or hemispheric lesion (16), tuberous sclerosis (3), or mesio-temporal sclerosis with periventricular leukomalacia (1). Of 16 who underwent surgery, 7 became seizure free. Five were able to discontinue all antiepileptic drugs.
In a multivariate analysis, neuroimaging was a significant predictor of seizure outcome.
"Imaging that is done properly is important," Dr. Wirrell said in an interview. "There is value with going on with a medication trial, but if one fails, it’s reasonable to have the discussion of possible surgery as we are starting the next."
Dr. Wirrell had no financial disclosures.
MONTREAL – Early imaging may be the best clue about seizure remission in children with early intractable seizures, according to findings from a population-based, retrospective cohort study.
Children who had uncontrolled seizures at 2 years and structural brain abnormalities had a 9% rate of becoming seizure free during a median follow-up period of 12 years. However, 60% of those with structurally normal brains eventually did become seizure free, despite an early history of intractability, Dr. Elaine Wirrell said at the 30th International Epilepsy Congress.
"While a significant minority of children with early medical intractability ultimately achieved seizure control without surgery, those with an abnormal imaging study did poorly," said Dr. Wirrell of the Mayo Clinic, Rochester, Minn. "For this subgroup, early surgical intervention is strongly advised to limit the comorbidities of ongoing, intractable seizures. Conversely, a cautious approach is suggested for those with normal imaging, as most will remit with time."
She and her colleagues examined medical intractability in a group of 381 children who were diagnosed with epilepsy during 1980-2009 and who were followed for a median of 12 years. Of these, 75 (20%) showed early medical intractability, defined as failing at least two antiepileptic medications or continuing to have seizures more often than every 6 months.
The children were a mean of 7 years old when their epilepsy began. Most (75%) had a focal onset, while 23% had a generalized onset. Onset for the remainder was unknown. The etiology was unknown in 54%, genetic in 23%, and structural or metabolic in the remainder.
Overall, 74 children underwent either MRI or CT, and a structural brain abnormality was present in 35. There were 40 children who had structurally normal brains. (One patient was known to have a familial absence syndrome ruling out a structural abnormality and did not undergo brain imaging.)
After a median follow-up of 12 years, 48 patients were still having seizures or had undergone brain surgery; 32 of these initially had abnormal brain imaging. Only three children with a structural brain abnormality were seizure free off or on medications at follow-up. A total of 24 who had normal brain structure eventually became seizure free.
More than half of the patients with an abnormality were surgical candidates because of the presence of a single focal or hemispheric lesion (16), tuberous sclerosis (3), or mesio-temporal sclerosis with periventricular leukomalacia (1). Of 16 who underwent surgery, 7 became seizure free. Five were able to discontinue all antiepileptic drugs.
In a multivariate analysis, neuroimaging was a significant predictor of seizure outcome.
"Imaging that is done properly is important," Dr. Wirrell said in an interview. "There is value with going on with a medication trial, but if one fails, it’s reasonable to have the discussion of possible surgery as we are starting the next."
Dr. Wirrell had no financial disclosures.
AT IEC 2013
Major finding: Among children with an early history of medically intractable seizures, those with abnormal brain imaging were significantly less likely to become seizure free than were those with normal imaging (9% vs. 60%).
Data source: A population-based, retrospective cohort study of 75 children with early medically intractable epilepsy.
Disclosures: Dr. Elaine Wirrell had no financial disclosures.