User login
Doug Brunk is a San Diego-based award-winning reporter who began covering health care in 1991. Before joining the company, he wrote for the health sciences division of Columbia University and was an associate editor at Contemporary Long Term Care magazine when it won a Jesse H. Neal Award. His work has been syndicated by the Los Angeles Times and he is the author of two books related to the University of Kentucky Wildcats men's basketball program. Doug has a master’s degree in magazine journalism from the S.I. Newhouse School of Public Communications at Syracuse University. Follow him on Twitter @dougbrunk.
HT not linked with increased mortality
AT ENDO 2015
SAN DIEGO – Hormone therapy in postmenopausal women does not seem to affect risk of mortality, results from a meta-analysis demonstrated.
“Most menopausal women suffer from a lot of bothersome symptoms, like hot flashes, sleep problems, and night sweats,” Dr. Khalid Benkhadra said during a press briefing at the meeting of the Endocrine Society. “Hormone therapy is usually very effective in alleviating these symptoms. However, women are usually concerned about the long-term safety [of HT].”
To assess all-cause and specific mortality associated with hormone therapy in postmenopausal women, Dr. Benkhadra and his associates searched databases for randomized trials published before August 2013 that compared hormone therapy with placebo or no treatment, with a follow-up of at least 6 months. The databases included MEDLINE and the Cochrane Central Register of Controlled Trials.
The outcome of interest was death from any cause, death from cardiac events, and death from breast cancer, lung cancer, ovarian cancer, colorectal cancer, and stroke.
The final meta-analysis included 43 clinical trials involving more than 52,000 women, mean age 62 years. Dr. Benkhadra, a research fellow at Mayo Clinic, Rochester, Minn., reported that no statistically significant association was observed between the use of hormone therapy and all-cause mortality (risk ratio, 0.99), with no significant interaction based on hormone type or preexisting heart disease. In addition, no association was observed between use of hormone therapy and death from myocardial infarction (RR, 1.04), stroke (RR, 1.49), and breast cancer (RR, 0.93), nor lung cancer, ovarian cancer, and colorectal cancer. “In each of these categories, there was no significant effect of HR on the risk of dying,” Dr. Benkhadra said. “Results were similar for estrogen only versus combined estrogen and progesterone therapy.”
He acknowledged certain limitations of the study, including the fact that the evidence in the 43 trials was rated as “low to moderate” and the length of follow-up was limited to 5 years.
Based on the findings, he concluded that postmenopausal women with bothersome symptoms “can engage with their physician in shared decision making to discuss treatment [with HT].”
Dr. Benkhadra reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
SAN DIEGO – Hormone therapy in postmenopausal women does not seem to affect risk of mortality, results from a meta-analysis demonstrated.
“Most menopausal women suffer from a lot of bothersome symptoms, like hot flashes, sleep problems, and night sweats,” Dr. Khalid Benkhadra said during a press briefing at the meeting of the Endocrine Society. “Hormone therapy is usually very effective in alleviating these symptoms. However, women are usually concerned about the long-term safety [of HT].”
To assess all-cause and specific mortality associated with hormone therapy in postmenopausal women, Dr. Benkhadra and his associates searched databases for randomized trials published before August 2013 that compared hormone therapy with placebo or no treatment, with a follow-up of at least 6 months. The databases included MEDLINE and the Cochrane Central Register of Controlled Trials.
The outcome of interest was death from any cause, death from cardiac events, and death from breast cancer, lung cancer, ovarian cancer, colorectal cancer, and stroke.
The final meta-analysis included 43 clinical trials involving more than 52,000 women, mean age 62 years. Dr. Benkhadra, a research fellow at Mayo Clinic, Rochester, Minn., reported that no statistically significant association was observed between the use of hormone therapy and all-cause mortality (risk ratio, 0.99), with no significant interaction based on hormone type or preexisting heart disease. In addition, no association was observed between use of hormone therapy and death from myocardial infarction (RR, 1.04), stroke (RR, 1.49), and breast cancer (RR, 0.93), nor lung cancer, ovarian cancer, and colorectal cancer. “In each of these categories, there was no significant effect of HR on the risk of dying,” Dr. Benkhadra said. “Results were similar for estrogen only versus combined estrogen and progesterone therapy.”
He acknowledged certain limitations of the study, including the fact that the evidence in the 43 trials was rated as “low to moderate” and the length of follow-up was limited to 5 years.
Based on the findings, he concluded that postmenopausal women with bothersome symptoms “can engage with their physician in shared decision making to discuss treatment [with HT].”
Dr. Benkhadra reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
SAN DIEGO – Hormone therapy in postmenopausal women does not seem to affect risk of mortality, results from a meta-analysis demonstrated.
“Most menopausal women suffer from a lot of bothersome symptoms, like hot flashes, sleep problems, and night sweats,” Dr. Khalid Benkhadra said during a press briefing at the meeting of the Endocrine Society. “Hormone therapy is usually very effective in alleviating these symptoms. However, women are usually concerned about the long-term safety [of HT].”
To assess all-cause and specific mortality associated with hormone therapy in postmenopausal women, Dr. Benkhadra and his associates searched databases for randomized trials published before August 2013 that compared hormone therapy with placebo or no treatment, with a follow-up of at least 6 months. The databases included MEDLINE and the Cochrane Central Register of Controlled Trials.
The outcome of interest was death from any cause, death from cardiac events, and death from breast cancer, lung cancer, ovarian cancer, colorectal cancer, and stroke.
The final meta-analysis included 43 clinical trials involving more than 52,000 women, mean age 62 years. Dr. Benkhadra, a research fellow at Mayo Clinic, Rochester, Minn., reported that no statistically significant association was observed between the use of hormone therapy and all-cause mortality (risk ratio, 0.99), with no significant interaction based on hormone type or preexisting heart disease. In addition, no association was observed between use of hormone therapy and death from myocardial infarction (RR, 1.04), stroke (RR, 1.49), and breast cancer (RR, 0.93), nor lung cancer, ovarian cancer, and colorectal cancer. “In each of these categories, there was no significant effect of HR on the risk of dying,” Dr. Benkhadra said. “Results were similar for estrogen only versus combined estrogen and progesterone therapy.”
He acknowledged certain limitations of the study, including the fact that the evidence in the 43 trials was rated as “low to moderate” and the length of follow-up was limited to 5 years.
Based on the findings, he concluded that postmenopausal women with bothersome symptoms “can engage with their physician in shared decision making to discuss treatment [with HT].”
Dr. Benkhadra reported having no relevant financial conflicts.
On Twitter @dougbrunk
Key clinical point: Use of hormone therapy by postmenopausal women was not associated with an increased risk of mortality.
Major finding: No statistically significant association was observed between the use of hormone therapy and all-cause mortality (risk ratio, 0.99).
Data source: A meta-analysis of 43 clinical trials involving more than 52,000 women, mean age 62 years.
Disclosures: Dr. Benkhadra reported having no relevant financial conflicts.
Alternatives to colonoscopy for CRC screening are works in progress
SAN FRANCISCO – Colonoscopy may be the most commonly used technique to screen for colorectal cancer in the United States, but it’s far from perfect, according to Dr. David A. Lieberman.
For one thing, serious complications occur in 3-5 per 1,000 procedures at 30 days of follow-up, “and if you look at individuals over 65, the rates almost double in terms of serious complications,” Dr. Lieberman, chief of the division of gastroenterology and hepatology at Oregon Health and Science University, said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology. “The rate of perforation over age 65 is about 1 per 1,000,” he said.
Colonoscopy is invasive, “and it would never be conceived of as the ideal test for population-based screening,” he added. “It’s expensive, and [clinicians] in many countries simply do not have the resources and expertise to do it.”
Compared with current screening rates in the United States for cervical and breast cancer, a “screening gap” exists in the percentage of adults who are up to date in tests for colorectal cancer, he said. “If you look at what’s been accomplished with cervical cancer screening and mammography, we see that there’s a ways to go for colonoscopy,” he said.
The ideal screening test for colorectal cancer would accurately identify individuals at risk for colon cancer at an early, preventable stage, Dr. Lieberman said. That prevention element “really averts the downstream colon cancer care costs, which would be very attractive in a single-payer health care system,” he said. “If we had a system that took care of us for life, that system would be very invested in trying to prevent colon cancer, because [it would] be worried about those downstream costs.”
One potential alternative to colonoscopy is fecal immunochemical testing (FIT). A recent meta-analysis suggested that FIT detected cancer with a 79% sensitivity (Ann. Intern. Med. 2014;160:171-81), and a separate, recent prospective study found a 74% sensitivity rate in detecting cancer, but a 24% sensitivity rate in detecting advanced adenoma (N. Engl. J. Med. 2014;370:1287-97). “So in terms of early detection, FIT is pretty good,” Dr. Lieberman said. “In terms of cancer prevention? Maybe not so good. There are also issues around the testing program, which requires adherence to both positive and negative tests. Some of these behavioral factors play a major role in the actual effectiveness. So you can have a great test, but if it’s not getting done properly then there’s going to be an issue. There is some potential for CRC prevention, but it’s going to be less than for some others.”
What about stool DNA? A landmark study found that stool DNA detected cancer with a sensitivity of 92%, compared with FIT at 74% (N. Engl. J Med. 2014;370:1287-97). However, the sensitivity for detecting adenomas was relatively low for both methods (42% for stool DNA, compared with 24% for FIT). Stool DNA testing “appears to be an effective screening test [but] the appropriate interval for testing is still uncertain,” Dr. Lieberman said. “The recommendation is a 3-year interval.”
He characterized serum testing as the “holy grail” of screening for colorectal cancer, “because it would be ideal to have a noninvasive test. You’d come in, get a blood draw, and get risk stratified. There are a number of different possible pathways, one looking at specific genetic markers, others looking at genetic fingerprinting: in other words, seeing a pattern of genes that are associated with colon cancer versus patients who do not have colon cancer. Proteomics is another fingerprinting technique, as is metabolomics: looking at a variety of metabolites that may be altered by the presence of having neoplasia. This is an intriguing area but so far there hasn’t been a lot of data.”
To date, the most progress involved in this area involves molecular tests of DNA or proteins. “These studies are all relatively small and somewhat promising in terms of cancer sensitivity, but not so great for adenoma sensitivity,” he said. “It’s fair to say that this is still a work in progress.”
Dr. Lieberman reported having served on the scientific advisory board of Exact Sciences.
In a separate presentation, Dr. Uri Ladabaum updated attendees on the status of PillCam, also known as colon capsule endoscopy (CCE), for detecting colon cancer. The PillCam is cleared by the Food and Drug Administration for detection of colon polyps in patients after an incomplete optical colonoscopy with adequate preparation, or if a complete evaluation of the colon was not technically possible. In a large meta-analysis, the first-generation PillCam achieved a sensitivity of 71% and a specificity of 75% in detecting polyps of any size and a sensitivity of 68% and a specificity of 82% in detecting polyps 6 mm in size or larger (Clin. Gastroenterol. Hepatol. 2010;8:515-22). “The results were somewhat disappointing for a test like this, not hitting the bar that we would want,” said Dr. Ladabaum, professor of medicine in the division of gastroenterology and hepatology at Stanford (Calif.) University.
The second generation PillCam features an improved angle of the imagers, from 156 degrees to 172 degrees, “so you get a bigger view all the way around,” he said. Another new feature is an adaptive imaging rate. The first-generation device captured 4 images per second regardless of where the capsule was moving. The new system “goes at 4 images per second when stationary, goes as fast as 35 imagers per second while moving, and 14 images per second before it reaches the small bowel,” Dr. Ladabaum said.
In an early study of the second-generation system, the device achieved a sensitivity of 89% and a specificity of 76% in detecting polyps of 6 mm in size or larger and a sensitivity of 88% and a specificity of 89% in detecting polyps 10 mm in size or larger (Endoscopy 2009;41:1026-31).
More recently, researchers examined consecutive patients who had incomplete colonoscopy and compared a follow-up with either second-generation CCE or CT colonography (Gut 2015;64:272-81). The diagnostic yield of CCE and CT colonography was 25% and 12%, respectively, for polyps 6 mm or larger and 5% vs. 3% for polyps 10 mm or larger.
In a larger, multisite study, researchers examined the efficacy of the second-generation PillCam as a primary screening tool (Gastroenterology 2015 [doi:10.1053/j.gastro.2015.01.025). Of 884 subjects enrolled, 85 were excluded from the analysis for a variety of reasons. Of these, 12 were excluded because the capsule did not reach the colon within 12 hours and 8 were excluded because the capsule did not pass the cecum within 12 hours. “So if used in practice, we’ll see this – [incomplete capsule studies],” Dr. Ladabaum said.
In addition, 77 were excluded for inadequate cleansing and capsule transit time through the colon in less than 40 minutes. “Maybe that’s due to the booster regimen,” he said. “There are still some things to work out in the bowel prep. Overall, there were a good number of people [in this study] who didn’t really have the exam [completed as intended]. Is that going to pan out as a big problem in clinical practice? That remains to be seen.”
The study found that the second-generation PillCam achieved a sensitivity of 87% and a specificity of 94% in detecting polyps 6 mm or larger and a sensitivity of 85% and a specificity of 97% in detecting polyps 10 mm in size or larger. “Is this good enough for this kind of technology?” Dr. Ladabaum asked. “There’s lots of room for debate here. Interestingly, the numbers for sessile serrated polyps didn’t look that good” (a sensitivity of 29% for polyps 6 mm in size or greater and 33% for polyps 10 mm in size or greater).
Dr. Ladabaum disclosed that he has served as a consultant for Given Imaging and Exact Sciences. He has also served on the advisory board for Mauna Kea Technologies.
In conclusion, according to Dr. Lieberman, there are several feasible alternatives to colonoscopy for primary colon cancer screening in average-risk individuals. Each has advantages and limitations and offers a less invasive approach to screening. The bar for screening has been raised from early cancer detection alone to both early detection and prevention. These less invasive tests will be expected to achieve accurate detection of individuals with early stage colon cancer and those at risk for colon cancer.
SAN FRANCISCO – Colonoscopy may be the most commonly used technique to screen for colorectal cancer in the United States, but it’s far from perfect, according to Dr. David A. Lieberman.
For one thing, serious complications occur in 3-5 per 1,000 procedures at 30 days of follow-up, “and if you look at individuals over 65, the rates almost double in terms of serious complications,” Dr. Lieberman, chief of the division of gastroenterology and hepatology at Oregon Health and Science University, said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology. “The rate of perforation over age 65 is about 1 per 1,000,” he said.
Colonoscopy is invasive, “and it would never be conceived of as the ideal test for population-based screening,” he added. “It’s expensive, and [clinicians] in many countries simply do not have the resources and expertise to do it.”
Compared with current screening rates in the United States for cervical and breast cancer, a “screening gap” exists in the percentage of adults who are up to date in tests for colorectal cancer, he said. “If you look at what’s been accomplished with cervical cancer screening and mammography, we see that there’s a ways to go for colonoscopy,” he said.
The ideal screening test for colorectal cancer would accurately identify individuals at risk for colon cancer at an early, preventable stage, Dr. Lieberman said. That prevention element “really averts the downstream colon cancer care costs, which would be very attractive in a single-payer health care system,” he said. “If we had a system that took care of us for life, that system would be very invested in trying to prevent colon cancer, because [it would] be worried about those downstream costs.”
One potential alternative to colonoscopy is fecal immunochemical testing (FIT). A recent meta-analysis suggested that FIT detected cancer with a 79% sensitivity (Ann. Intern. Med. 2014;160:171-81), and a separate, recent prospective study found a 74% sensitivity rate in detecting cancer, but a 24% sensitivity rate in detecting advanced adenoma (N. Engl. J. Med. 2014;370:1287-97). “So in terms of early detection, FIT is pretty good,” Dr. Lieberman said. “In terms of cancer prevention? Maybe not so good. There are also issues around the testing program, which requires adherence to both positive and negative tests. Some of these behavioral factors play a major role in the actual effectiveness. So you can have a great test, but if it’s not getting done properly then there’s going to be an issue. There is some potential for CRC prevention, but it’s going to be less than for some others.”
What about stool DNA? A landmark study found that stool DNA detected cancer with a sensitivity of 92%, compared with FIT at 74% (N. Engl. J Med. 2014;370:1287-97). However, the sensitivity for detecting adenomas was relatively low for both methods (42% for stool DNA, compared with 24% for FIT). Stool DNA testing “appears to be an effective screening test [but] the appropriate interval for testing is still uncertain,” Dr. Lieberman said. “The recommendation is a 3-year interval.”
He characterized serum testing as the “holy grail” of screening for colorectal cancer, “because it would be ideal to have a noninvasive test. You’d come in, get a blood draw, and get risk stratified. There are a number of different possible pathways, one looking at specific genetic markers, others looking at genetic fingerprinting: in other words, seeing a pattern of genes that are associated with colon cancer versus patients who do not have colon cancer. Proteomics is another fingerprinting technique, as is metabolomics: looking at a variety of metabolites that may be altered by the presence of having neoplasia. This is an intriguing area but so far there hasn’t been a lot of data.”
To date, the most progress involved in this area involves molecular tests of DNA or proteins. “These studies are all relatively small and somewhat promising in terms of cancer sensitivity, but not so great for adenoma sensitivity,” he said. “It’s fair to say that this is still a work in progress.”
Dr. Lieberman reported having served on the scientific advisory board of Exact Sciences.
In a separate presentation, Dr. Uri Ladabaum updated attendees on the status of PillCam, also known as colon capsule endoscopy (CCE), for detecting colon cancer. The PillCam is cleared by the Food and Drug Administration for detection of colon polyps in patients after an incomplete optical colonoscopy with adequate preparation, or if a complete evaluation of the colon was not technically possible. In a large meta-analysis, the first-generation PillCam achieved a sensitivity of 71% and a specificity of 75% in detecting polyps of any size and a sensitivity of 68% and a specificity of 82% in detecting polyps 6 mm in size or larger (Clin. Gastroenterol. Hepatol. 2010;8:515-22). “The results were somewhat disappointing for a test like this, not hitting the bar that we would want,” said Dr. Ladabaum, professor of medicine in the division of gastroenterology and hepatology at Stanford (Calif.) University.
The second generation PillCam features an improved angle of the imagers, from 156 degrees to 172 degrees, “so you get a bigger view all the way around,” he said. Another new feature is an adaptive imaging rate. The first-generation device captured 4 images per second regardless of where the capsule was moving. The new system “goes at 4 images per second when stationary, goes as fast as 35 imagers per second while moving, and 14 images per second before it reaches the small bowel,” Dr. Ladabaum said.
In an early study of the second-generation system, the device achieved a sensitivity of 89% and a specificity of 76% in detecting polyps of 6 mm in size or larger and a sensitivity of 88% and a specificity of 89% in detecting polyps 10 mm in size or larger (Endoscopy 2009;41:1026-31).
More recently, researchers examined consecutive patients who had incomplete colonoscopy and compared a follow-up with either second-generation CCE or CT colonography (Gut 2015;64:272-81). The diagnostic yield of CCE and CT colonography was 25% and 12%, respectively, for polyps 6 mm or larger and 5% vs. 3% for polyps 10 mm or larger.
In a larger, multisite study, researchers examined the efficacy of the second-generation PillCam as a primary screening tool (Gastroenterology 2015 [doi:10.1053/j.gastro.2015.01.025). Of 884 subjects enrolled, 85 were excluded from the analysis for a variety of reasons. Of these, 12 were excluded because the capsule did not reach the colon within 12 hours and 8 were excluded because the capsule did not pass the cecum within 12 hours. “So if used in practice, we’ll see this – [incomplete capsule studies],” Dr. Ladabaum said.
In addition, 77 were excluded for inadequate cleansing and capsule transit time through the colon in less than 40 minutes. “Maybe that’s due to the booster regimen,” he said. “There are still some things to work out in the bowel prep. Overall, there were a good number of people [in this study] who didn’t really have the exam [completed as intended]. Is that going to pan out as a big problem in clinical practice? That remains to be seen.”
The study found that the second-generation PillCam achieved a sensitivity of 87% and a specificity of 94% in detecting polyps 6 mm or larger and a sensitivity of 85% and a specificity of 97% in detecting polyps 10 mm in size or larger. “Is this good enough for this kind of technology?” Dr. Ladabaum asked. “There’s lots of room for debate here. Interestingly, the numbers for sessile serrated polyps didn’t look that good” (a sensitivity of 29% for polyps 6 mm in size or greater and 33% for polyps 10 mm in size or greater).
Dr. Ladabaum disclosed that he has served as a consultant for Given Imaging and Exact Sciences. He has also served on the advisory board for Mauna Kea Technologies.
In conclusion, according to Dr. Lieberman, there are several feasible alternatives to colonoscopy for primary colon cancer screening in average-risk individuals. Each has advantages and limitations and offers a less invasive approach to screening. The bar for screening has been raised from early cancer detection alone to both early detection and prevention. These less invasive tests will be expected to achieve accurate detection of individuals with early stage colon cancer and those at risk for colon cancer.
SAN FRANCISCO – Colonoscopy may be the most commonly used technique to screen for colorectal cancer in the United States, but it’s far from perfect, according to Dr. David A. Lieberman.
For one thing, serious complications occur in 3-5 per 1,000 procedures at 30 days of follow-up, “and if you look at individuals over 65, the rates almost double in terms of serious complications,” Dr. Lieberman, chief of the division of gastroenterology and hepatology at Oregon Health and Science University, said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology. “The rate of perforation over age 65 is about 1 per 1,000,” he said.
Colonoscopy is invasive, “and it would never be conceived of as the ideal test for population-based screening,” he added. “It’s expensive, and [clinicians] in many countries simply do not have the resources and expertise to do it.”
Compared with current screening rates in the United States for cervical and breast cancer, a “screening gap” exists in the percentage of adults who are up to date in tests for colorectal cancer, he said. “If you look at what’s been accomplished with cervical cancer screening and mammography, we see that there’s a ways to go for colonoscopy,” he said.
The ideal screening test for colorectal cancer would accurately identify individuals at risk for colon cancer at an early, preventable stage, Dr. Lieberman said. That prevention element “really averts the downstream colon cancer care costs, which would be very attractive in a single-payer health care system,” he said. “If we had a system that took care of us for life, that system would be very invested in trying to prevent colon cancer, because [it would] be worried about those downstream costs.”
One potential alternative to colonoscopy is fecal immunochemical testing (FIT). A recent meta-analysis suggested that FIT detected cancer with a 79% sensitivity (Ann. Intern. Med. 2014;160:171-81), and a separate, recent prospective study found a 74% sensitivity rate in detecting cancer, but a 24% sensitivity rate in detecting advanced adenoma (N. Engl. J. Med. 2014;370:1287-97). “So in terms of early detection, FIT is pretty good,” Dr. Lieberman said. “In terms of cancer prevention? Maybe not so good. There are also issues around the testing program, which requires adherence to both positive and negative tests. Some of these behavioral factors play a major role in the actual effectiveness. So you can have a great test, but if it’s not getting done properly then there’s going to be an issue. There is some potential for CRC prevention, but it’s going to be less than for some others.”
What about stool DNA? A landmark study found that stool DNA detected cancer with a sensitivity of 92%, compared with FIT at 74% (N. Engl. J Med. 2014;370:1287-97). However, the sensitivity for detecting adenomas was relatively low for both methods (42% for stool DNA, compared with 24% for FIT). Stool DNA testing “appears to be an effective screening test [but] the appropriate interval for testing is still uncertain,” Dr. Lieberman said. “The recommendation is a 3-year interval.”
He characterized serum testing as the “holy grail” of screening for colorectal cancer, “because it would be ideal to have a noninvasive test. You’d come in, get a blood draw, and get risk stratified. There are a number of different possible pathways, one looking at specific genetic markers, others looking at genetic fingerprinting: in other words, seeing a pattern of genes that are associated with colon cancer versus patients who do not have colon cancer. Proteomics is another fingerprinting technique, as is metabolomics: looking at a variety of metabolites that may be altered by the presence of having neoplasia. This is an intriguing area but so far there hasn’t been a lot of data.”
To date, the most progress involved in this area involves molecular tests of DNA or proteins. “These studies are all relatively small and somewhat promising in terms of cancer sensitivity, but not so great for adenoma sensitivity,” he said. “It’s fair to say that this is still a work in progress.”
Dr. Lieberman reported having served on the scientific advisory board of Exact Sciences.
In a separate presentation, Dr. Uri Ladabaum updated attendees on the status of PillCam, also known as colon capsule endoscopy (CCE), for detecting colon cancer. The PillCam is cleared by the Food and Drug Administration for detection of colon polyps in patients after an incomplete optical colonoscopy with adequate preparation, or if a complete evaluation of the colon was not technically possible. In a large meta-analysis, the first-generation PillCam achieved a sensitivity of 71% and a specificity of 75% in detecting polyps of any size and a sensitivity of 68% and a specificity of 82% in detecting polyps 6 mm in size or larger (Clin. Gastroenterol. Hepatol. 2010;8:515-22). “The results were somewhat disappointing for a test like this, not hitting the bar that we would want,” said Dr. Ladabaum, professor of medicine in the division of gastroenterology and hepatology at Stanford (Calif.) University.
The second generation PillCam features an improved angle of the imagers, from 156 degrees to 172 degrees, “so you get a bigger view all the way around,” he said. Another new feature is an adaptive imaging rate. The first-generation device captured 4 images per second regardless of where the capsule was moving. The new system “goes at 4 images per second when stationary, goes as fast as 35 imagers per second while moving, and 14 images per second before it reaches the small bowel,” Dr. Ladabaum said.
In an early study of the second-generation system, the device achieved a sensitivity of 89% and a specificity of 76% in detecting polyps of 6 mm in size or larger and a sensitivity of 88% and a specificity of 89% in detecting polyps 10 mm in size or larger (Endoscopy 2009;41:1026-31).
More recently, researchers examined consecutive patients who had incomplete colonoscopy and compared a follow-up with either second-generation CCE or CT colonography (Gut 2015;64:272-81). The diagnostic yield of CCE and CT colonography was 25% and 12%, respectively, for polyps 6 mm or larger and 5% vs. 3% for polyps 10 mm or larger.
In a larger, multisite study, researchers examined the efficacy of the second-generation PillCam as a primary screening tool (Gastroenterology 2015 [doi:10.1053/j.gastro.2015.01.025). Of 884 subjects enrolled, 85 were excluded from the analysis for a variety of reasons. Of these, 12 were excluded because the capsule did not reach the colon within 12 hours and 8 were excluded because the capsule did not pass the cecum within 12 hours. “So if used in practice, we’ll see this – [incomplete capsule studies],” Dr. Ladabaum said.
In addition, 77 were excluded for inadequate cleansing and capsule transit time through the colon in less than 40 minutes. “Maybe that’s due to the booster regimen,” he said. “There are still some things to work out in the bowel prep. Overall, there were a good number of people [in this study] who didn’t really have the exam [completed as intended]. Is that going to pan out as a big problem in clinical practice? That remains to be seen.”
The study found that the second-generation PillCam achieved a sensitivity of 87% and a specificity of 94% in detecting polyps 6 mm or larger and a sensitivity of 85% and a specificity of 97% in detecting polyps 10 mm in size or larger. “Is this good enough for this kind of technology?” Dr. Ladabaum asked. “There’s lots of room for debate here. Interestingly, the numbers for sessile serrated polyps didn’t look that good” (a sensitivity of 29% for polyps 6 mm in size or greater and 33% for polyps 10 mm in size or greater).
Dr. Ladabaum disclosed that he has served as a consultant for Given Imaging and Exact Sciences. He has also served on the advisory board for Mauna Kea Technologies.
In conclusion, according to Dr. Lieberman, there are several feasible alternatives to colonoscopy for primary colon cancer screening in average-risk individuals. Each has advantages and limitations and offers a less invasive approach to screening. The bar for screening has been raised from early cancer detection alone to both early detection and prevention. These less invasive tests will be expected to achieve accurate detection of individuals with early stage colon cancer and those at risk for colon cancer.
AT THE AGA 2015 TECH SUMMIT
Update on Covidien and Boston Scientific Awards
SAN FRANCISCO – At the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology, two researchers provided attendees with an update on their innovative research projects made possible thanks to grants from the AGA Research Foundation.
First, Dr. Richard Kwon updated attendees on the status of his research supported by the inaugural AGA–Covidien Research & Development Pilot Award in Technology. Announced in the fall of 2014, this award is supported by a generous grant from Covidien, a leading global provider of health care products. With this grant, Dr. Kwon and his associates at the University of Michigan are investigating how an image processing and analysis method known as analytic morphomics can be used to improve the care of patients with pancreatic cysts. To date, the inability to accurately distinguish the cyst types has led to a significant amount of unnecessary surgeries and surveillance.
“Nonmucinous cysts generally are comprised of serous cystadenomas as well as inflammatory cells and a small handful of zebra cysts,” explained Dr. Kwon, a gastroenterologist at the university. “These are generally considered benign and do not require follow-up. In contrast, mucinous cysts are made of mucinous cystic neoplasms, which are characterized by a variant stroma and IPMN [intraductal papillary mucinous neoplasm]. This is important because these are the cysts that have malignant potential and therefore require some sort of follow-up and/or surgery.”
He described analytic morphomics as “a novel method to quantitatively analyze structural data such as shape and surface contour (which are otherwise descriptive) from CT scans in order to predict disease states. In our minds, the benefit is that it overcomes some of the shortcomings of imaging curvilinear organs. It has the potential to overcome qualitative descriptions and the limitations of characterizing these cysts based on one cut or one slice of imaging. Hopefully, it will allow us to establish quantifiable criteria, it can be performed on existing studies so you don’t need a new CT scan, and there’s no specialized laboratory or equipment that you need other than a high-powered computer.”
Dr. Kwon presented results from 81 cystic lesions processed with the method. Analytic morphomics yielded a sensitivity of 78% in predicting serous cystadenomas (SCA), a specificity of 90%, a positive predictive value of 73%, and negative predictive value of 88%. The accuracy was 84%. He and his associates are in the process of validating the findings and are in the planning stages of a prospective study comparing morphomics and cyst fluid analysis. “The accuracy of the SCA prediction model appears equal to cyst fluid carcinoembryonic antigen testing, suggesting morphomic data have the potential to replace endoscopic ultrasound/fine needle aspiration in certain scenarios,” he said.
During a separate presentation, Dr. Ashish Nimgaonkar updated attendees on the status of his research, supported by the inaugural AGA–Boston Scientific Career Development Technology & Innovation Award. Bestowed in the fall of 2014, this award is supported by a grant from Boston Scientific, a leading innovator of medical solutions. Dr. Nimgaonkar’s research focuses on developing technology to manage patients with refractory ascites – a condition in which fluid builds up in the abdomen. This fluid accumulation eventually becomes resistant to medical therapy and the only definitive treatment at this stage is liver transplantation, which is limited by organ availability. The only option for them is removal of this fluid every few weeks in a hospital or clinical setting. “When we started to look at this problem, the biggest driving factor for a [solution] was a way to keep these patients out of the hospital and to be able to do this at home,” said Dr. Nimgaonkar, associate medical director at the Center for Bioengineering Innovation and Design in the department of biomedical engineering at Johns Hopkins University, Baltimore.
He and his colleagues have developed a wireless implantable shunt technology to pull the fluid from the peritoneal space into the stomach. With this approach, patients can manage their fluid drainage needs at home – significantly improving their quality of life, as well as reducing the cost of care associated with frequent hospital visits. “You can think of it as a combination of a peritoneal drain and a percutaneous gastrostomy tube, which is a feeding tube hooked but completely implanted in the subcutaneous space,” he said of the technology, which is currently being refined and tested in animal models. “It can be done under conscious sedation. The current design we are working on is a simple, nonelectromechanical solution – silicone-based – which uses the diaphragmatic movements of the patient to drive the fluid. It turns out that the pressure differential between the peritoneal space and the intragastric cavity is minimal, so you need some kind of actuation mechanism.”
Dr. Nimgaonkar and his colleagues hope to study the technology in humans “in the next year or so.”
SAN FRANCISCO – At the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology, two researchers provided attendees with an update on their innovative research projects made possible thanks to grants from the AGA Research Foundation.
First, Dr. Richard Kwon updated attendees on the status of his research supported by the inaugural AGA–Covidien Research & Development Pilot Award in Technology. Announced in the fall of 2014, this award is supported by a generous grant from Covidien, a leading global provider of health care products. With this grant, Dr. Kwon and his associates at the University of Michigan are investigating how an image processing and analysis method known as analytic morphomics can be used to improve the care of patients with pancreatic cysts. To date, the inability to accurately distinguish the cyst types has led to a significant amount of unnecessary surgeries and surveillance.
“Nonmucinous cysts generally are comprised of serous cystadenomas as well as inflammatory cells and a small handful of zebra cysts,” explained Dr. Kwon, a gastroenterologist at the university. “These are generally considered benign and do not require follow-up. In contrast, mucinous cysts are made of mucinous cystic neoplasms, which are characterized by a variant stroma and IPMN [intraductal papillary mucinous neoplasm]. This is important because these are the cysts that have malignant potential and therefore require some sort of follow-up and/or surgery.”
He described analytic morphomics as “a novel method to quantitatively analyze structural data such as shape and surface contour (which are otherwise descriptive) from CT scans in order to predict disease states. In our minds, the benefit is that it overcomes some of the shortcomings of imaging curvilinear organs. It has the potential to overcome qualitative descriptions and the limitations of characterizing these cysts based on one cut or one slice of imaging. Hopefully, it will allow us to establish quantifiable criteria, it can be performed on existing studies so you don’t need a new CT scan, and there’s no specialized laboratory or equipment that you need other than a high-powered computer.”
Dr. Kwon presented results from 81 cystic lesions processed with the method. Analytic morphomics yielded a sensitivity of 78% in predicting serous cystadenomas (SCA), a specificity of 90%, a positive predictive value of 73%, and negative predictive value of 88%. The accuracy was 84%. He and his associates are in the process of validating the findings and are in the planning stages of a prospective study comparing morphomics and cyst fluid analysis. “The accuracy of the SCA prediction model appears equal to cyst fluid carcinoembryonic antigen testing, suggesting morphomic data have the potential to replace endoscopic ultrasound/fine needle aspiration in certain scenarios,” he said.
During a separate presentation, Dr. Ashish Nimgaonkar updated attendees on the status of his research, supported by the inaugural AGA–Boston Scientific Career Development Technology & Innovation Award. Bestowed in the fall of 2014, this award is supported by a grant from Boston Scientific, a leading innovator of medical solutions. Dr. Nimgaonkar’s research focuses on developing technology to manage patients with refractory ascites – a condition in which fluid builds up in the abdomen. This fluid accumulation eventually becomes resistant to medical therapy and the only definitive treatment at this stage is liver transplantation, which is limited by organ availability. The only option for them is removal of this fluid every few weeks in a hospital or clinical setting. “When we started to look at this problem, the biggest driving factor for a [solution] was a way to keep these patients out of the hospital and to be able to do this at home,” said Dr. Nimgaonkar, associate medical director at the Center for Bioengineering Innovation and Design in the department of biomedical engineering at Johns Hopkins University, Baltimore.
He and his colleagues have developed a wireless implantable shunt technology to pull the fluid from the peritoneal space into the stomach. With this approach, patients can manage their fluid drainage needs at home – significantly improving their quality of life, as well as reducing the cost of care associated with frequent hospital visits. “You can think of it as a combination of a peritoneal drain and a percutaneous gastrostomy tube, which is a feeding tube hooked but completely implanted in the subcutaneous space,” he said of the technology, which is currently being refined and tested in animal models. “It can be done under conscious sedation. The current design we are working on is a simple, nonelectromechanical solution – silicone-based – which uses the diaphragmatic movements of the patient to drive the fluid. It turns out that the pressure differential between the peritoneal space and the intragastric cavity is minimal, so you need some kind of actuation mechanism.”
Dr. Nimgaonkar and his colleagues hope to study the technology in humans “in the next year or so.”
SAN FRANCISCO – At the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology, two researchers provided attendees with an update on their innovative research projects made possible thanks to grants from the AGA Research Foundation.
First, Dr. Richard Kwon updated attendees on the status of his research supported by the inaugural AGA–Covidien Research & Development Pilot Award in Technology. Announced in the fall of 2014, this award is supported by a generous grant from Covidien, a leading global provider of health care products. With this grant, Dr. Kwon and his associates at the University of Michigan are investigating how an image processing and analysis method known as analytic morphomics can be used to improve the care of patients with pancreatic cysts. To date, the inability to accurately distinguish the cyst types has led to a significant amount of unnecessary surgeries and surveillance.
“Nonmucinous cysts generally are comprised of serous cystadenomas as well as inflammatory cells and a small handful of zebra cysts,” explained Dr. Kwon, a gastroenterologist at the university. “These are generally considered benign and do not require follow-up. In contrast, mucinous cysts are made of mucinous cystic neoplasms, which are characterized by a variant stroma and IPMN [intraductal papillary mucinous neoplasm]. This is important because these are the cysts that have malignant potential and therefore require some sort of follow-up and/or surgery.”
He described analytic morphomics as “a novel method to quantitatively analyze structural data such as shape and surface contour (which are otherwise descriptive) from CT scans in order to predict disease states. In our minds, the benefit is that it overcomes some of the shortcomings of imaging curvilinear organs. It has the potential to overcome qualitative descriptions and the limitations of characterizing these cysts based on one cut or one slice of imaging. Hopefully, it will allow us to establish quantifiable criteria, it can be performed on existing studies so you don’t need a new CT scan, and there’s no specialized laboratory or equipment that you need other than a high-powered computer.”
Dr. Kwon presented results from 81 cystic lesions processed with the method. Analytic morphomics yielded a sensitivity of 78% in predicting serous cystadenomas (SCA), a specificity of 90%, a positive predictive value of 73%, and negative predictive value of 88%. The accuracy was 84%. He and his associates are in the process of validating the findings and are in the planning stages of a prospective study comparing morphomics and cyst fluid analysis. “The accuracy of the SCA prediction model appears equal to cyst fluid carcinoembryonic antigen testing, suggesting morphomic data have the potential to replace endoscopic ultrasound/fine needle aspiration in certain scenarios,” he said.
During a separate presentation, Dr. Ashish Nimgaonkar updated attendees on the status of his research, supported by the inaugural AGA–Boston Scientific Career Development Technology & Innovation Award. Bestowed in the fall of 2014, this award is supported by a grant from Boston Scientific, a leading innovator of medical solutions. Dr. Nimgaonkar’s research focuses on developing technology to manage patients with refractory ascites – a condition in which fluid builds up in the abdomen. This fluid accumulation eventually becomes resistant to medical therapy and the only definitive treatment at this stage is liver transplantation, which is limited by organ availability. The only option for them is removal of this fluid every few weeks in a hospital or clinical setting. “When we started to look at this problem, the biggest driving factor for a [solution] was a way to keep these patients out of the hospital and to be able to do this at home,” said Dr. Nimgaonkar, associate medical director at the Center for Bioengineering Innovation and Design in the department of biomedical engineering at Johns Hopkins University, Baltimore.
He and his colleagues have developed a wireless implantable shunt technology to pull the fluid from the peritoneal space into the stomach. With this approach, patients can manage their fluid drainage needs at home – significantly improving their quality of life, as well as reducing the cost of care associated with frequent hospital visits. “You can think of it as a combination of a peritoneal drain and a percutaneous gastrostomy tube, which is a feeding tube hooked but completely implanted in the subcutaneous space,” he said of the technology, which is currently being refined and tested in animal models. “It can be done under conscious sedation. The current design we are working on is a simple, nonelectromechanical solution – silicone-based – which uses the diaphragmatic movements of the patient to drive the fluid. It turns out that the pressure differential between the peritoneal space and the intragastric cavity is minimal, so you need some kind of actuation mechanism.”
Dr. Nimgaonkar and his colleagues hope to study the technology in humans “in the next year or so.”
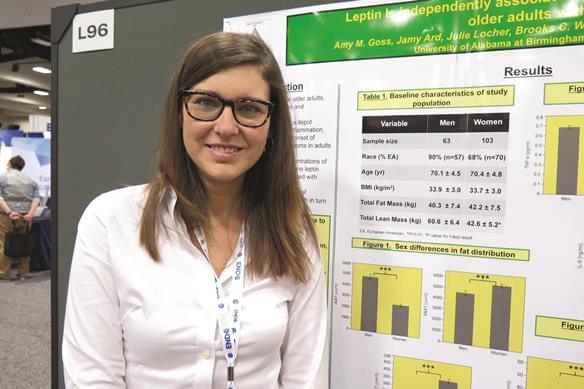
Elevated leptin linked to depression in obese older adults
SAN DIEGO – Elevated leptin may play a role in the relationship between excess adiposity and depressive symptoms in older adults, judging from the results of a cross-sectional survey.
While depression is common among older adults, particularly among those who are obese, “what’s not known is the underlying physiological mechanisms of what’s linking depression with excess fat mass,” Amy M. Goss, Ph.D., a postdoctoral research fellow in the department of nutrition sciences at the University of Alabama at Birmingham, said in an interview at the meeting of the Endocrine Society. “There’s a lot of work showing that visceral fat in particular is linked to depression. It’s thought that inflammation that emanates from visceral fat can lead to depressive symptoms. This is a particular problem in older adults, because they tend to have a lot of visceral adiposity.”
In an effort to investigate the independent relationships among depressive symptoms, total fat mass, intra-abdominal adipose tissue, markers of inflammation, and leptin in aging men and women, Dr. Goss and her associates evaluated 164 obese adults older than 65 years. The researchers used the Center for Epidemiologic Studies Depression (CES-D) 20-item scale to assess depressive symptoms, DXA to assess body composition, MRI to assess fat distribution, and a fasting blood sample to assess leptin, high-sensitivity C-reactive protein (hsCRP), interleukin-6, and tumor necrosis factor–alpha.
Compared with men, women had significantly greater levels of leptin (+100%; P less than .001), hsCRP (+63.9%; P less than .01), and CES-D scores (+52.7%; P less than .001). At the same time, men had significantly greater intra-abdominal adipose tissue (+82%; P less than .001) and lean mass (+42%; P less than .001), compared with women.
Multiple linear regression analysis revealed that only leptin was positively associated with CES-D scores, independent of gender and of all other markers studied (P less than .05).
In their abstract, the researchers concluded that elevated leptin “may, in part, mediate the observed relationship between excess adiposity and onset of depressive symptoms in aging, obese adults. Higher leptin and hsCRP concentration in women, compared to men may indicate greater proinflammatory state and explain sex differences in depressive symptoms in this population.”
Dr. Goss said that a key limitation of the study is its cross-sectional design. “We can’t determine the cause and effect based on this study, but it does lead the way to design randomized, clinical trials to test how we can improve depression in older adults with obesity,” she said.
She reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – Elevated leptin may play a role in the relationship between excess adiposity and depressive symptoms in older adults, judging from the results of a cross-sectional survey.
While depression is common among older adults, particularly among those who are obese, “what’s not known is the underlying physiological mechanisms of what’s linking depression with excess fat mass,” Amy M. Goss, Ph.D., a postdoctoral research fellow in the department of nutrition sciences at the University of Alabama at Birmingham, said in an interview at the meeting of the Endocrine Society. “There’s a lot of work showing that visceral fat in particular is linked to depression. It’s thought that inflammation that emanates from visceral fat can lead to depressive symptoms. This is a particular problem in older adults, because they tend to have a lot of visceral adiposity.”
In an effort to investigate the independent relationships among depressive symptoms, total fat mass, intra-abdominal adipose tissue, markers of inflammation, and leptin in aging men and women, Dr. Goss and her associates evaluated 164 obese adults older than 65 years. The researchers used the Center for Epidemiologic Studies Depression (CES-D) 20-item scale to assess depressive symptoms, DXA to assess body composition, MRI to assess fat distribution, and a fasting blood sample to assess leptin, high-sensitivity C-reactive protein (hsCRP), interleukin-6, and tumor necrosis factor–alpha.
Compared with men, women had significantly greater levels of leptin (+100%; P less than .001), hsCRP (+63.9%; P less than .01), and CES-D scores (+52.7%; P less than .001). At the same time, men had significantly greater intra-abdominal adipose tissue (+82%; P less than .001) and lean mass (+42%; P less than .001), compared with women.
Multiple linear regression analysis revealed that only leptin was positively associated with CES-D scores, independent of gender and of all other markers studied (P less than .05).
In their abstract, the researchers concluded that elevated leptin “may, in part, mediate the observed relationship between excess adiposity and onset of depressive symptoms in aging, obese adults. Higher leptin and hsCRP concentration in women, compared to men may indicate greater proinflammatory state and explain sex differences in depressive symptoms in this population.”
Dr. Goss said that a key limitation of the study is its cross-sectional design. “We can’t determine the cause and effect based on this study, but it does lead the way to design randomized, clinical trials to test how we can improve depression in older adults with obesity,” she said.
She reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – Elevated leptin may play a role in the relationship between excess adiposity and depressive symptoms in older adults, judging from the results of a cross-sectional survey.
While depression is common among older adults, particularly among those who are obese, “what’s not known is the underlying physiological mechanisms of what’s linking depression with excess fat mass,” Amy M. Goss, Ph.D., a postdoctoral research fellow in the department of nutrition sciences at the University of Alabama at Birmingham, said in an interview at the meeting of the Endocrine Society. “There’s a lot of work showing that visceral fat in particular is linked to depression. It’s thought that inflammation that emanates from visceral fat can lead to depressive symptoms. This is a particular problem in older adults, because they tend to have a lot of visceral adiposity.”
In an effort to investigate the independent relationships among depressive symptoms, total fat mass, intra-abdominal adipose tissue, markers of inflammation, and leptin in aging men and women, Dr. Goss and her associates evaluated 164 obese adults older than 65 years. The researchers used the Center for Epidemiologic Studies Depression (CES-D) 20-item scale to assess depressive symptoms, DXA to assess body composition, MRI to assess fat distribution, and a fasting blood sample to assess leptin, high-sensitivity C-reactive protein (hsCRP), interleukin-6, and tumor necrosis factor–alpha.
Compared with men, women had significantly greater levels of leptin (+100%; P less than .001), hsCRP (+63.9%; P less than .01), and CES-D scores (+52.7%; P less than .001). At the same time, men had significantly greater intra-abdominal adipose tissue (+82%; P less than .001) and lean mass (+42%; P less than .001), compared with women.
Multiple linear regression analysis revealed that only leptin was positively associated with CES-D scores, independent of gender and of all other markers studied (P less than .05).
In their abstract, the researchers concluded that elevated leptin “may, in part, mediate the observed relationship between excess adiposity and onset of depressive symptoms in aging, obese adults. Higher leptin and hsCRP concentration in women, compared to men may indicate greater proinflammatory state and explain sex differences in depressive symptoms in this population.”
Dr. Goss said that a key limitation of the study is its cross-sectional design. “We can’t determine the cause and effect based on this study, but it does lead the way to design randomized, clinical trials to test how we can improve depression in older adults with obesity,” she said.
She reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
Key clinical point: Leptin may play a role in the onset of depressive symptoms in aging, obese adults.
Major finding: Only leptin was positively associated with Center for Epidemiologic Studies Depression (CES-D) scores, independent of gender and of all other markers studied (P less than .05).
Data source: A cross-sectional study of 164 obese adults over the age of 65 years.
Disclosures: Dr. Goss reported having no relevant financial conflicts.
Lowering dietary fat, not carbs, led to greater body fat loss
SAN DIEGO – In adults with obesity, lowering intake of dietary fat may lead to greater body fat loss than lowering intake of dietary carbohydrates, results from a small federally funded study showed.
“A lot of focus has been placed on the idea of the competing importance of dietary carbohydrate versus dietary fat in the treatment and prevention of obesity,” lead study author Kevin D. Hall, Ph.D., said during a press briefing at the meeting of the Endocrine Society. “The fashion of which is the evil macronutrient of choice at one time or another is vacillating back and forth. More recently I would say that dietary carbohydrate has been the one demonized, with sugars and refined carbohydrates being the main culprit.”
Dr. Hall, a senior investigator at the National Institute of Diabetes and Digestive and Kidney Diseases, went on to note that at the level of the whole body, “you can think of fat balance as a balance between the intake of fat and how much fat is being burned. So if you’re eating a certain amount of fat and burning that same amount of fat, your body fat will be stable. The only way to perturb that fat balance at the whole-body level is to induce an imbalance between intake of fat and the amount of fat that is being burned.” The low carbohydrate advocates will suggest that an obese adult should decrease carbohydrate intake in the diet, thereby lowering insulin. This in turn has the effect of releasing fat from the fat cells, thereby leading to an imbalance that will eventually rebalance at a lower level of body fat. Alternatively, another way an obese adult could potentially achieve the same results is to cut dietary fat. The resulting state of fat imbalance would promote loss of body fat, according to Dr. Hall.
In an effort to investigate the whole-body energy expenditure and metabolic fat balance from selective restriction of dietary carbohydrate versus dietary fat, the researchers enrolled 19 nondiabetic volunteers with a mean age of 34 years and a mean body mass index of 36 mg/m2. Of the 19 volunteers, 10 were men. The volunteers were housed in a metabolic ward at the National Institutes of Health for a pair of 2-week inpatient visits. For 5 days, everyone was fed a eucaloric baseline diet of 50% carbohydrate, 35% fat, and 15% protein that was designed to provide them with the number of calories they needed to maintain their body weight. Next, the volunteers were randomized to one of two groups where they received a 30% reduced energy diet by restriction of their intake of either fat or carbohydrate. “We provided all of their food; they had no choice in what they ate,” Dr. Hall said.
After a 2-4-week washout period the volunteers returned to the ward and received the alternative diet after the same 5-day baseline run-in phase. “The idea here was [that] we knew exactly how much they ate, and during the time they spent in metabolic chambers we could measure how much fat they burned,” he explained. “So we could calculate how fat oxidation changed as well as the fat imbalance in the body. The low-carb diet selectively reduced carbohydrates, keeping fat and protein at the constant levels, whereas the low-fat diet selectively reduced fat, keeping carbohydrate and protein at baseline levels.”
During the 6-day diet phase the researchers cut 30% of participants’ calories, selectively from either carbohydrates or fat. This translated into about 800 calories per day being cut from both diets.
Dr. Hall reported that reduction of dietary carbohydrates led to a significant increase in whole-body 24-hour fat oxidation (P< .0001), while isocaloric reduction of dietary fat in the same subjects had no significant effect on 24-hour fat oxidation (P = .15). However, the cumulative metabolic fat balance (adding up the daily imbalance between intake and expenditure of fat) indicated that the reduced fat diet resulted in about 80% greater body fat loss, compared with the reduced carbohydrate diet (P = .0003).
Dr. Hall acknowledged certain limitations of the study, including the fact that it did not address the long-term efficacy of low-carbohydrate or low-fat diets. One remaining key unanswered question is whether “the low fat diet continue to outpace the low carbohydrate diet with respect to body fat?” he asked. “This is a short-term study, a small number of people with obesity under metabolic ward conditions. We can’t extrapolate about what might happen to their health with respect to these diets, if they were able to adhere to them for long periods of time.”
The study was funded by the Intramural Research Program of the NIDDK. Dr. Hall reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – In adults with obesity, lowering intake of dietary fat may lead to greater body fat loss than lowering intake of dietary carbohydrates, results from a small federally funded study showed.
“A lot of focus has been placed on the idea of the competing importance of dietary carbohydrate versus dietary fat in the treatment and prevention of obesity,” lead study author Kevin D. Hall, Ph.D., said during a press briefing at the meeting of the Endocrine Society. “The fashion of which is the evil macronutrient of choice at one time or another is vacillating back and forth. More recently I would say that dietary carbohydrate has been the one demonized, with sugars and refined carbohydrates being the main culprit.”
Dr. Hall, a senior investigator at the National Institute of Diabetes and Digestive and Kidney Diseases, went on to note that at the level of the whole body, “you can think of fat balance as a balance between the intake of fat and how much fat is being burned. So if you’re eating a certain amount of fat and burning that same amount of fat, your body fat will be stable. The only way to perturb that fat balance at the whole-body level is to induce an imbalance between intake of fat and the amount of fat that is being burned.” The low carbohydrate advocates will suggest that an obese adult should decrease carbohydrate intake in the diet, thereby lowering insulin. This in turn has the effect of releasing fat from the fat cells, thereby leading to an imbalance that will eventually rebalance at a lower level of body fat. Alternatively, another way an obese adult could potentially achieve the same results is to cut dietary fat. The resulting state of fat imbalance would promote loss of body fat, according to Dr. Hall.
In an effort to investigate the whole-body energy expenditure and metabolic fat balance from selective restriction of dietary carbohydrate versus dietary fat, the researchers enrolled 19 nondiabetic volunteers with a mean age of 34 years and a mean body mass index of 36 mg/m2. Of the 19 volunteers, 10 were men. The volunteers were housed in a metabolic ward at the National Institutes of Health for a pair of 2-week inpatient visits. For 5 days, everyone was fed a eucaloric baseline diet of 50% carbohydrate, 35% fat, and 15% protein that was designed to provide them with the number of calories they needed to maintain their body weight. Next, the volunteers were randomized to one of two groups where they received a 30% reduced energy diet by restriction of their intake of either fat or carbohydrate. “We provided all of their food; they had no choice in what they ate,” Dr. Hall said.
After a 2-4-week washout period the volunteers returned to the ward and received the alternative diet after the same 5-day baseline run-in phase. “The idea here was [that] we knew exactly how much they ate, and during the time they spent in metabolic chambers we could measure how much fat they burned,” he explained. “So we could calculate how fat oxidation changed as well as the fat imbalance in the body. The low-carb diet selectively reduced carbohydrates, keeping fat and protein at the constant levels, whereas the low-fat diet selectively reduced fat, keeping carbohydrate and protein at baseline levels.”
During the 6-day diet phase the researchers cut 30% of participants’ calories, selectively from either carbohydrates or fat. This translated into about 800 calories per day being cut from both diets.
Dr. Hall reported that reduction of dietary carbohydrates led to a significant increase in whole-body 24-hour fat oxidation (P< .0001), while isocaloric reduction of dietary fat in the same subjects had no significant effect on 24-hour fat oxidation (P = .15). However, the cumulative metabolic fat balance (adding up the daily imbalance between intake and expenditure of fat) indicated that the reduced fat diet resulted in about 80% greater body fat loss, compared with the reduced carbohydrate diet (P = .0003).
Dr. Hall acknowledged certain limitations of the study, including the fact that it did not address the long-term efficacy of low-carbohydrate or low-fat diets. One remaining key unanswered question is whether “the low fat diet continue to outpace the low carbohydrate diet with respect to body fat?” he asked. “This is a short-term study, a small number of people with obesity under metabolic ward conditions. We can’t extrapolate about what might happen to their health with respect to these diets, if they were able to adhere to them for long periods of time.”
The study was funded by the Intramural Research Program of the NIDDK. Dr. Hall reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – In adults with obesity, lowering intake of dietary fat may lead to greater body fat loss than lowering intake of dietary carbohydrates, results from a small federally funded study showed.
“A lot of focus has been placed on the idea of the competing importance of dietary carbohydrate versus dietary fat in the treatment and prevention of obesity,” lead study author Kevin D. Hall, Ph.D., said during a press briefing at the meeting of the Endocrine Society. “The fashion of which is the evil macronutrient of choice at one time or another is vacillating back and forth. More recently I would say that dietary carbohydrate has been the one demonized, with sugars and refined carbohydrates being the main culprit.”
Dr. Hall, a senior investigator at the National Institute of Diabetes and Digestive and Kidney Diseases, went on to note that at the level of the whole body, “you can think of fat balance as a balance between the intake of fat and how much fat is being burned. So if you’re eating a certain amount of fat and burning that same amount of fat, your body fat will be stable. The only way to perturb that fat balance at the whole-body level is to induce an imbalance between intake of fat and the amount of fat that is being burned.” The low carbohydrate advocates will suggest that an obese adult should decrease carbohydrate intake in the diet, thereby lowering insulin. This in turn has the effect of releasing fat from the fat cells, thereby leading to an imbalance that will eventually rebalance at a lower level of body fat. Alternatively, another way an obese adult could potentially achieve the same results is to cut dietary fat. The resulting state of fat imbalance would promote loss of body fat, according to Dr. Hall.
In an effort to investigate the whole-body energy expenditure and metabolic fat balance from selective restriction of dietary carbohydrate versus dietary fat, the researchers enrolled 19 nondiabetic volunteers with a mean age of 34 years and a mean body mass index of 36 mg/m2. Of the 19 volunteers, 10 were men. The volunteers were housed in a metabolic ward at the National Institutes of Health for a pair of 2-week inpatient visits. For 5 days, everyone was fed a eucaloric baseline diet of 50% carbohydrate, 35% fat, and 15% protein that was designed to provide them with the number of calories they needed to maintain their body weight. Next, the volunteers were randomized to one of two groups where they received a 30% reduced energy diet by restriction of their intake of either fat or carbohydrate. “We provided all of their food; they had no choice in what they ate,” Dr. Hall said.
After a 2-4-week washout period the volunteers returned to the ward and received the alternative diet after the same 5-day baseline run-in phase. “The idea here was [that] we knew exactly how much they ate, and during the time they spent in metabolic chambers we could measure how much fat they burned,” he explained. “So we could calculate how fat oxidation changed as well as the fat imbalance in the body. The low-carb diet selectively reduced carbohydrates, keeping fat and protein at the constant levels, whereas the low-fat diet selectively reduced fat, keeping carbohydrate and protein at baseline levels.”
During the 6-day diet phase the researchers cut 30% of participants’ calories, selectively from either carbohydrates or fat. This translated into about 800 calories per day being cut from both diets.
Dr. Hall reported that reduction of dietary carbohydrates led to a significant increase in whole-body 24-hour fat oxidation (P< .0001), while isocaloric reduction of dietary fat in the same subjects had no significant effect on 24-hour fat oxidation (P = .15). However, the cumulative metabolic fat balance (adding up the daily imbalance between intake and expenditure of fat) indicated that the reduced fat diet resulted in about 80% greater body fat loss, compared with the reduced carbohydrate diet (P = .0003).
Dr. Hall acknowledged certain limitations of the study, including the fact that it did not address the long-term efficacy of low-carbohydrate or low-fat diets. One remaining key unanswered question is whether “the low fat diet continue to outpace the low carbohydrate diet with respect to body fat?” he asked. “This is a short-term study, a small number of people with obesity under metabolic ward conditions. We can’t extrapolate about what might happen to their health with respect to these diets, if they were able to adhere to them for long periods of time.”
The study was funded by the Intramural Research Program of the NIDDK. Dr. Hall reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
Key clinical point: To reduce body fat, consuming less fat may be more effective than consuming fewer carbohydrates.
Major finding: Reduction of dietary carbohydrates led to a significant increase in whole-body 24-hour fat oxidation (P < .0001), while isocaloric reduction of dietary fat in the same subjects had no significant effect on 24-hour fat oxidation (P = .15).
Data source: An inpatient study of 19 obese adults, which set out to evaluate the selective effect of restricting carbohydrate versus fat on energy and macronutrient balance.
Disclosures: The study was funded by the Intramural Research Program of the NIDDK. Dr. Hall reported having no relevant financial conflicts.
Strong leadership is the backbone of effective commercialization efforts
SAN FRANCISCO–The way Dr. Amanda Christini sees it, universities and academic medical centers should care about commercialization of innovation, because it creates new opportunities to impact patient care.
“As academicians, we need to engage in cultural change from the ground up in order to be good at effective innovation and in order to contribute in a meaningful way,” Dr. Christini, director of strategic initiatives for the Penn Medicine Center for Health Care Innovation, Philadelphia, said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Traditionally, the “value” of commercialization from a university or academic medical standpoint has been defined by royalties from product sales. “But it turns out there are different kinds of value,” Dr. Christini said. “Value is created when we have an impact on patient care. Value is created when we disseminate something we do that has impact, and it’s adopted broadly within your own institution or by other centers.”
The current health care “ecosystem” consists of many stakeholders, including patients, academic medical centers, the government, payers, large industry partners, investors, and start-ups, she continued. Current options for commercializing a product include licensing a product or device to an industry partner, creating a broad partnership with an industry partner, “where they fund your research and invest in core infrastructure such as manufacturing,” or creating a new company altogether. Embarking on such endeavors at today’s universities and academic medical centers is challenging because “the historic focus on therapeutics is slow to change,” Dr. Christini explained. “It’s very product oriented. What winds up happening is, instead of asking ‘what is possible,’ [decision-makers at] universities and academic medical centers will often say ‘No, we don’t do that.’ There is a fundamental culture shift that has to happen in order to allow this to be successful.”
Stanford (Calif.) University and the Massachusetts Institute of Technology, Cambridge, have been especially successful in fostering collaboration among stakeholders in the health care ecosystem, Dr. Christini said. In fact, Stanford alumni have launched 40,000 companies resulting in $2.7 trillion in annual revenue, while MIT grads have launched 25,800 companies resulting in $2.2 trillion in annual revenue. Strong leadership skills form the backbone of such success. “And it’s not just leadership that gets up and says the word ‘innovation’ over and over again,” she said. “It’s leadership that actually changes what happens inside the institution. One way to do that is to create metrics that measure what you actually care about – not just royalty stream. [You want to measure things] like how long does it take to get a transaction done? Is the faculty happy? Is the faculty satisfied? Do they feel like they are being served by these [shareholder] groups? How many start-ups with funding above a certain number have been created as a result of the work that’s done? These kinds of things can have a dramatic impact on how successful you are.”
SAN FRANCISCO–The way Dr. Amanda Christini sees it, universities and academic medical centers should care about commercialization of innovation, because it creates new opportunities to impact patient care.
“As academicians, we need to engage in cultural change from the ground up in order to be good at effective innovation and in order to contribute in a meaningful way,” Dr. Christini, director of strategic initiatives for the Penn Medicine Center for Health Care Innovation, Philadelphia, said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Traditionally, the “value” of commercialization from a university or academic medical standpoint has been defined by royalties from product sales. “But it turns out there are different kinds of value,” Dr. Christini said. “Value is created when we have an impact on patient care. Value is created when we disseminate something we do that has impact, and it’s adopted broadly within your own institution or by other centers.”
The current health care “ecosystem” consists of many stakeholders, including patients, academic medical centers, the government, payers, large industry partners, investors, and start-ups, she continued. Current options for commercializing a product include licensing a product or device to an industry partner, creating a broad partnership with an industry partner, “where they fund your research and invest in core infrastructure such as manufacturing,” or creating a new company altogether. Embarking on such endeavors at today’s universities and academic medical centers is challenging because “the historic focus on therapeutics is slow to change,” Dr. Christini explained. “It’s very product oriented. What winds up happening is, instead of asking ‘what is possible,’ [decision-makers at] universities and academic medical centers will often say ‘No, we don’t do that.’ There is a fundamental culture shift that has to happen in order to allow this to be successful.”
Stanford (Calif.) University and the Massachusetts Institute of Technology, Cambridge, have been especially successful in fostering collaboration among stakeholders in the health care ecosystem, Dr. Christini said. In fact, Stanford alumni have launched 40,000 companies resulting in $2.7 trillion in annual revenue, while MIT grads have launched 25,800 companies resulting in $2.2 trillion in annual revenue. Strong leadership skills form the backbone of such success. “And it’s not just leadership that gets up and says the word ‘innovation’ over and over again,” she said. “It’s leadership that actually changes what happens inside the institution. One way to do that is to create metrics that measure what you actually care about – not just royalty stream. [You want to measure things] like how long does it take to get a transaction done? Is the faculty happy? Is the faculty satisfied? Do they feel like they are being served by these [shareholder] groups? How many start-ups with funding above a certain number have been created as a result of the work that’s done? These kinds of things can have a dramatic impact on how successful you are.”
SAN FRANCISCO–The way Dr. Amanda Christini sees it, universities and academic medical centers should care about commercialization of innovation, because it creates new opportunities to impact patient care.
“As academicians, we need to engage in cultural change from the ground up in order to be good at effective innovation and in order to contribute in a meaningful way,” Dr. Christini, director of strategic initiatives for the Penn Medicine Center for Health Care Innovation, Philadelphia, said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Traditionally, the “value” of commercialization from a university or academic medical standpoint has been defined by royalties from product sales. “But it turns out there are different kinds of value,” Dr. Christini said. “Value is created when we have an impact on patient care. Value is created when we disseminate something we do that has impact, and it’s adopted broadly within your own institution or by other centers.”
The current health care “ecosystem” consists of many stakeholders, including patients, academic medical centers, the government, payers, large industry partners, investors, and start-ups, she continued. Current options for commercializing a product include licensing a product or device to an industry partner, creating a broad partnership with an industry partner, “where they fund your research and invest in core infrastructure such as manufacturing,” or creating a new company altogether. Embarking on such endeavors at today’s universities and academic medical centers is challenging because “the historic focus on therapeutics is slow to change,” Dr. Christini explained. “It’s very product oriented. What winds up happening is, instead of asking ‘what is possible,’ [decision-makers at] universities and academic medical centers will often say ‘No, we don’t do that.’ There is a fundamental culture shift that has to happen in order to allow this to be successful.”
Stanford (Calif.) University and the Massachusetts Institute of Technology, Cambridge, have been especially successful in fostering collaboration among stakeholders in the health care ecosystem, Dr. Christini said. In fact, Stanford alumni have launched 40,000 companies resulting in $2.7 trillion in annual revenue, while MIT grads have launched 25,800 companies resulting in $2.2 trillion in annual revenue. Strong leadership skills form the backbone of such success. “And it’s not just leadership that gets up and says the word ‘innovation’ over and over again,” she said. “It’s leadership that actually changes what happens inside the institution. One way to do that is to create metrics that measure what you actually care about – not just royalty stream. [You want to measure things] like how long does it take to get a transaction done? Is the faculty happy? Is the faculty satisfied? Do they feel like they are being served by these [shareholder] groups? How many start-ups with funding above a certain number have been created as a result of the work that’s done? These kinds of things can have a dramatic impact on how successful you are.”
AT THE 2015 AGA TECH SUMMIT
Team approach best for making purchasing decisions
SAN FRANCISCO – The best decisions about purchasing supplies and equipment for a hospital or medical practice come from administrators and physicians working together as a team, speaker after speaker reiterated during the session on supply chain purchasing decisions at the 2015 AGA Tech Summit, which was sponsored by the AGA Center for GI Innovation and Technology.
“In this era of cost constraints, how can physicians and facilities align purchasing decisions so that everybody’s working together to provide the right service for the right patient in the right setting, in the right amount, and at the right time?” asked session moderator Dr. Joel V. Brill, a board-certified internist and gastroenterologist who is medical director of FAIR Health Inc. “Does that mean reducing inventory and suppliers of items? Reducing choice of equipment? Not being able to provide a new technology or procedure to patients? Even of more critical importance is that, if physicians and facilities don’t work collaboratively to address these issues, if you’re not providing a high-quality, value-driven product, you’re in danger of being priced out of the marketplace or deselected from the provider network.”
Kenneth Strople, a health care consultant with more than 25 years of experience as a hospital COO and CEO, advises administrators and physicians to consider a key question: “What is the desired device or equipment going to do for patient care? Is it going to improve the quality of care provided in the facility? Patient care is No. 1; that’s why we do what we do. The latest and greatest is not necessarily the best for your organization and patients.”
He shared three examples of purchasing decisions that can go awry, the first being a surgical robot, which creates increased operating room time and a significant increase in consumables: “$3,500 per case for which there is no additional reimbursement,” said Mr. Strople, who is managing director of California-based Palisades Healthcare Solutions LLC. Buying a surgical robot may enhance marketing efforts and reputation of your hospital or health care group, he acknowledged, but if it’s going to cost your organization additional funds every time the device is used, “my question is, why?,” he said. “Margins in hospitals are quite small. In California, for example, if you’re making 4%-5% on revenue you’re doing great. So when the cost per case increases substantially [hospital administrators] have a problem, because you can’t lose money on every case and make it up in volume.”
The second example he discussed was cardiac stents for balloon angioplasty. When first introduced to the marketplace there was minimal ability for hospitals to receive reimbursement for stents (which cost about $3,000 each), due to the fact that no DRG existed for them. “Commercial contracts didn’t account for the new technology, so hospitals ‘ate’ the cost of stents for a protracted period of time until the contracts could be adjusted,” Mr. Strople said.
Another key purchasing decision involves variation in cost among medical devices on the marketplace. For example, is the $15,000 total knee implant from company A better than the $9,000 implant from company B? “Maybe in some cases, but the sales rep will tell you the one from company A is better, and the rep is in the OR to coordinate things,” Mr. Strople said. “In fact, the rep’s commission may actually be more than the physician fee for the procedure.” He noted that reference pricing and purchaser transparency initiatives are beginning to impact patient behavior, “especially when the additional $6,000 cost is 100% patient responsibility.” The best way to approach such a purchasing decision is to call a meeting with all of the clinicians who use the device or equipment being considered. “Show them what the devices cost, and then you get them to make the responsible decision,” Mr. Strople said. “If you provide them the appropriate data and they believe the data, they’ll make the right decision. They’re not interested in causing the hospital to go out of business.”
Mr. Strople emphasized the importance of putting politics aside when making important purchasing decisions. For example, Dr. Jones may wield more political clout in a hospital than Dr. Smith does, but “a smart hospital administrator isn’t going to go out and just buy that device for Dr. Jones. He’s going to do the analysis, treat everybody the same, and say, ‘bring me the patient-related data and bring me the financial data. I will validate the financial data. I will evaluate cost, revenue, and the feedback I get on the patient care side.”
Another speaker, David A. Pierce, senior vice president and president of the endoscopy division for Boston Scientific in Marlboro, Mass., said that since the passage of the Affordable Care Act, “the future is going to be population health, so [device manufacturing] models and structures are going to have to be built to survive in capitation and bundling situations. For a device company, we’re going to have to demonstrate better outcomes and the ability to manage patients across the entire continuum.” He went on to note that “our whole existence has been in a price mix world, where innovation and technology have always been taken in [and] reimbursed aggressively. But in a population management world, we have to [adopt] a volume-based mindset; we have to make that switch. Our cost structure is probably misaligned with that reality that’s on the horizon. Return on investment for R&D is decreasing all the time.”
Dr. Brill, who is based in Paradise Valley, Ariz., characterized the supply chain purchasing environment as evolutionary. “Traditionally, the hospital was the workshop where the physician did his or her work,” Dr. Brill said. “You came in and performed surgery there, but the hospital still made the purchasing decisions. While the Ambulatory Surgery Center is often more physician-centric, frequently focusing on one specialty such as orthopedics or gastroenterology or ophthalmology, it still has the same issues of staffing and purchasing supplies and capital equipment. As we start thinking about the transition from volume to value, supply chain purchasing is of critical importance, for the margins and overhead will impact what you are willing to contract for with a purchaser or payer when providing an episode or bundled payment for a service, whether an endoscopic procedure or a condition such as reflux or obesity. As we move from silos to collaboration, health care is becoming a team sport: purchasing decisions are a critical component of health care professionals and facilities working together in order to achieve the desired outcomes, demonstrate value, and to drive patient volume.”
On Twitter @dougbrunk
SAN FRANCISCO – The best decisions about purchasing supplies and equipment for a hospital or medical practice come from administrators and physicians working together as a team, speaker after speaker reiterated during the session on supply chain purchasing decisions at the 2015 AGA Tech Summit, which was sponsored by the AGA Center for GI Innovation and Technology.
“In this era of cost constraints, how can physicians and facilities align purchasing decisions so that everybody’s working together to provide the right service for the right patient in the right setting, in the right amount, and at the right time?” asked session moderator Dr. Joel V. Brill, a board-certified internist and gastroenterologist who is medical director of FAIR Health Inc. “Does that mean reducing inventory and suppliers of items? Reducing choice of equipment? Not being able to provide a new technology or procedure to patients? Even of more critical importance is that, if physicians and facilities don’t work collaboratively to address these issues, if you’re not providing a high-quality, value-driven product, you’re in danger of being priced out of the marketplace or deselected from the provider network.”
Kenneth Strople, a health care consultant with more than 25 years of experience as a hospital COO and CEO, advises administrators and physicians to consider a key question: “What is the desired device or equipment going to do for patient care? Is it going to improve the quality of care provided in the facility? Patient care is No. 1; that’s why we do what we do. The latest and greatest is not necessarily the best for your organization and patients.”
He shared three examples of purchasing decisions that can go awry, the first being a surgical robot, which creates increased operating room time and a significant increase in consumables: “$3,500 per case for which there is no additional reimbursement,” said Mr. Strople, who is managing director of California-based Palisades Healthcare Solutions LLC. Buying a surgical robot may enhance marketing efforts and reputation of your hospital or health care group, he acknowledged, but if it’s going to cost your organization additional funds every time the device is used, “my question is, why?,” he said. “Margins in hospitals are quite small. In California, for example, if you’re making 4%-5% on revenue you’re doing great. So when the cost per case increases substantially [hospital administrators] have a problem, because you can’t lose money on every case and make it up in volume.”
The second example he discussed was cardiac stents for balloon angioplasty. When first introduced to the marketplace there was minimal ability for hospitals to receive reimbursement for stents (which cost about $3,000 each), due to the fact that no DRG existed for them. “Commercial contracts didn’t account for the new technology, so hospitals ‘ate’ the cost of stents for a protracted period of time until the contracts could be adjusted,” Mr. Strople said.
Another key purchasing decision involves variation in cost among medical devices on the marketplace. For example, is the $15,000 total knee implant from company A better than the $9,000 implant from company B? “Maybe in some cases, but the sales rep will tell you the one from company A is better, and the rep is in the OR to coordinate things,” Mr. Strople said. “In fact, the rep’s commission may actually be more than the physician fee for the procedure.” He noted that reference pricing and purchaser transparency initiatives are beginning to impact patient behavior, “especially when the additional $6,000 cost is 100% patient responsibility.” The best way to approach such a purchasing decision is to call a meeting with all of the clinicians who use the device or equipment being considered. “Show them what the devices cost, and then you get them to make the responsible decision,” Mr. Strople said. “If you provide them the appropriate data and they believe the data, they’ll make the right decision. They’re not interested in causing the hospital to go out of business.”
Mr. Strople emphasized the importance of putting politics aside when making important purchasing decisions. For example, Dr. Jones may wield more political clout in a hospital than Dr. Smith does, but “a smart hospital administrator isn’t going to go out and just buy that device for Dr. Jones. He’s going to do the analysis, treat everybody the same, and say, ‘bring me the patient-related data and bring me the financial data. I will validate the financial data. I will evaluate cost, revenue, and the feedback I get on the patient care side.”
Another speaker, David A. Pierce, senior vice president and president of the endoscopy division for Boston Scientific in Marlboro, Mass., said that since the passage of the Affordable Care Act, “the future is going to be population health, so [device manufacturing] models and structures are going to have to be built to survive in capitation and bundling situations. For a device company, we’re going to have to demonstrate better outcomes and the ability to manage patients across the entire continuum.” He went on to note that “our whole existence has been in a price mix world, where innovation and technology have always been taken in [and] reimbursed aggressively. But in a population management world, we have to [adopt] a volume-based mindset; we have to make that switch. Our cost structure is probably misaligned with that reality that’s on the horizon. Return on investment for R&D is decreasing all the time.”
Dr. Brill, who is based in Paradise Valley, Ariz., characterized the supply chain purchasing environment as evolutionary. “Traditionally, the hospital was the workshop where the physician did his or her work,” Dr. Brill said. “You came in and performed surgery there, but the hospital still made the purchasing decisions. While the Ambulatory Surgery Center is often more physician-centric, frequently focusing on one specialty such as orthopedics or gastroenterology or ophthalmology, it still has the same issues of staffing and purchasing supplies and capital equipment. As we start thinking about the transition from volume to value, supply chain purchasing is of critical importance, for the margins and overhead will impact what you are willing to contract for with a purchaser or payer when providing an episode or bundled payment for a service, whether an endoscopic procedure or a condition such as reflux or obesity. As we move from silos to collaboration, health care is becoming a team sport: purchasing decisions are a critical component of health care professionals and facilities working together in order to achieve the desired outcomes, demonstrate value, and to drive patient volume.”
On Twitter @dougbrunk
SAN FRANCISCO – The best decisions about purchasing supplies and equipment for a hospital or medical practice come from administrators and physicians working together as a team, speaker after speaker reiterated during the session on supply chain purchasing decisions at the 2015 AGA Tech Summit, which was sponsored by the AGA Center for GI Innovation and Technology.
“In this era of cost constraints, how can physicians and facilities align purchasing decisions so that everybody’s working together to provide the right service for the right patient in the right setting, in the right amount, and at the right time?” asked session moderator Dr. Joel V. Brill, a board-certified internist and gastroenterologist who is medical director of FAIR Health Inc. “Does that mean reducing inventory and suppliers of items? Reducing choice of equipment? Not being able to provide a new technology or procedure to patients? Even of more critical importance is that, if physicians and facilities don’t work collaboratively to address these issues, if you’re not providing a high-quality, value-driven product, you’re in danger of being priced out of the marketplace or deselected from the provider network.”
Kenneth Strople, a health care consultant with more than 25 years of experience as a hospital COO and CEO, advises administrators and physicians to consider a key question: “What is the desired device or equipment going to do for patient care? Is it going to improve the quality of care provided in the facility? Patient care is No. 1; that’s why we do what we do. The latest and greatest is not necessarily the best for your organization and patients.”
He shared three examples of purchasing decisions that can go awry, the first being a surgical robot, which creates increased operating room time and a significant increase in consumables: “$3,500 per case for which there is no additional reimbursement,” said Mr. Strople, who is managing director of California-based Palisades Healthcare Solutions LLC. Buying a surgical robot may enhance marketing efforts and reputation of your hospital or health care group, he acknowledged, but if it’s going to cost your organization additional funds every time the device is used, “my question is, why?,” he said. “Margins in hospitals are quite small. In California, for example, if you’re making 4%-5% on revenue you’re doing great. So when the cost per case increases substantially [hospital administrators] have a problem, because you can’t lose money on every case and make it up in volume.”
The second example he discussed was cardiac stents for balloon angioplasty. When first introduced to the marketplace there was minimal ability for hospitals to receive reimbursement for stents (which cost about $3,000 each), due to the fact that no DRG existed for them. “Commercial contracts didn’t account for the new technology, so hospitals ‘ate’ the cost of stents for a protracted period of time until the contracts could be adjusted,” Mr. Strople said.
Another key purchasing decision involves variation in cost among medical devices on the marketplace. For example, is the $15,000 total knee implant from company A better than the $9,000 implant from company B? “Maybe in some cases, but the sales rep will tell you the one from company A is better, and the rep is in the OR to coordinate things,” Mr. Strople said. “In fact, the rep’s commission may actually be more than the physician fee for the procedure.” He noted that reference pricing and purchaser transparency initiatives are beginning to impact patient behavior, “especially when the additional $6,000 cost is 100% patient responsibility.” The best way to approach such a purchasing decision is to call a meeting with all of the clinicians who use the device or equipment being considered. “Show them what the devices cost, and then you get them to make the responsible decision,” Mr. Strople said. “If you provide them the appropriate data and they believe the data, they’ll make the right decision. They’re not interested in causing the hospital to go out of business.”
Mr. Strople emphasized the importance of putting politics aside when making important purchasing decisions. For example, Dr. Jones may wield more political clout in a hospital than Dr. Smith does, but “a smart hospital administrator isn’t going to go out and just buy that device for Dr. Jones. He’s going to do the analysis, treat everybody the same, and say, ‘bring me the patient-related data and bring me the financial data. I will validate the financial data. I will evaluate cost, revenue, and the feedback I get on the patient care side.”
Another speaker, David A. Pierce, senior vice president and president of the endoscopy division for Boston Scientific in Marlboro, Mass., said that since the passage of the Affordable Care Act, “the future is going to be population health, so [device manufacturing] models and structures are going to have to be built to survive in capitation and bundling situations. For a device company, we’re going to have to demonstrate better outcomes and the ability to manage patients across the entire continuum.” He went on to note that “our whole existence has been in a price mix world, where innovation and technology have always been taken in [and] reimbursed aggressively. But in a population management world, we have to [adopt] a volume-based mindset; we have to make that switch. Our cost structure is probably misaligned with that reality that’s on the horizon. Return on investment for R&D is decreasing all the time.”
Dr. Brill, who is based in Paradise Valley, Ariz., characterized the supply chain purchasing environment as evolutionary. “Traditionally, the hospital was the workshop where the physician did his or her work,” Dr. Brill said. “You came in and performed surgery there, but the hospital still made the purchasing decisions. While the Ambulatory Surgery Center is often more physician-centric, frequently focusing on one specialty such as orthopedics or gastroenterology or ophthalmology, it still has the same issues of staffing and purchasing supplies and capital equipment. As we start thinking about the transition from volume to value, supply chain purchasing is of critical importance, for the margins and overhead will impact what you are willing to contract for with a purchaser or payer when providing an episode or bundled payment for a service, whether an endoscopic procedure or a condition such as reflux or obesity. As we move from silos to collaboration, health care is becoming a team sport: purchasing decisions are a critical component of health care professionals and facilities working together in order to achieve the desired outcomes, demonstrate value, and to drive patient volume.”
On Twitter @dougbrunk
AT THE 2015 AGA TECH SUMMIT
VIDEO: What makes people predictably irrational?
SAN FRANCISCO – Using behavioral economics to help patients identify and overcome psychological barriers to behavioral change has expanded our understanding of what makes people predictably irrational, according to Dr. David A. Asch.
In an interview at the 2015 AGA Tech Summit, Dr. Asch, executive director of the Penn Medicine Center for Health Care Innovation and professor of health care management at the Wharton School at the University of Pennsylvania, both in Philadelphia, discusses how physicians can incorporate the principles of behavioral economics into their practices to improve patient care.
Dr. Asch disclosed that he is a principal at VAL Health, a behavioral consulting firm.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @dougbrunk
SAN FRANCISCO – Using behavioral economics to help patients identify and overcome psychological barriers to behavioral change has expanded our understanding of what makes people predictably irrational, according to Dr. David A. Asch.
In an interview at the 2015 AGA Tech Summit, Dr. Asch, executive director of the Penn Medicine Center for Health Care Innovation and professor of health care management at the Wharton School at the University of Pennsylvania, both in Philadelphia, discusses how physicians can incorporate the principles of behavioral economics into their practices to improve patient care.
Dr. Asch disclosed that he is a principal at VAL Health, a behavioral consulting firm.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @dougbrunk
SAN FRANCISCO – Using behavioral economics to help patients identify and overcome psychological barriers to behavioral change has expanded our understanding of what makes people predictably irrational, according to Dr. David A. Asch.
In an interview at the 2015 AGA Tech Summit, Dr. Asch, executive director of the Penn Medicine Center for Health Care Innovation and professor of health care management at the Wharton School at the University of Pennsylvania, both in Philadelphia, discusses how physicians can incorporate the principles of behavioral economics into their practices to improve patient care.
Dr. Asch disclosed that he is a principal at VAL Health, a behavioral consulting firm.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @dougbrunk
AT THE 2015 AGA TECH SUMMIT
Gastric bypass patients on calcium had higher rates of kidney stone growth
SAN DIEGO – Patients taking calcium supplementation after gastric bypass surgery had higher rates of kidney stone growth, compared with those who received no calcium supplementation, results from a single-center retrospective study showed.
In addition, the majority of stones in patients taking calcium supplementation were comprised of calcium oxalate monohydrate, which is less amenable to extracorporeal shockwave therapy. “You need more invasive procedures to break up these kinds of stones,” lead author Christopher Loftus said in an interview at the meeting of the Endocrine Society, where the study was presented during a late-breaking abstract session.
Though it has been demonstrated that bariatric surgery is associated with an increased risk of kidney stone formation (Kidney Int. 2014 [doi:10.1038/ki.2014.352]), Mr. Loftus, a fourth-year medical student at Cleveland Clinic Lerner College of Medicine, and his associates set out to determine whether calcium supplementation increases the risk of nephrolithiasis in 60 stone-forming patients after gastric bypass surgery performed at the Cleveland Clinic. For each patient, two unenhanced CT scans at least 1 month apart and less than 2 years apart were selected at the start of the supplementation date and after the gastric bypass surgery date. The researchers calculated the rate of stone growth by the change in consecutive stone burden (the sum of maximum diameters of stones) divided by the elapsed time between scans.
Of the 60 patients, 31 received postoperative calcium supplementation (an average of 500 mg/day) and 29 did not. Compared with patients who did not take calcium supplementation, those who did were younger (a mean of 53 years vs. 58 years, respectively), more likely to be female (81% vs. 69%), and had a higher body mass index (34.5 kg/m2 vs. 32.7 kg/m2). In addition, a greater proportion of patients taking calcium supplements underwent Roux-en-Y bypass (83% vs. 64%; P = .19), had stones comprised of calcium oxalate (81% vs. 67%; P = .56), and a higher rate of stone growth (more than 10 mm/year vs. less than 5 mm/year; P = .0004). “We weren’t expecting such a pronounced effect,” Mr. Loftus said.
In their abstract, the researchers said that further studies are required to elucidate the exact role of calcium supplementation on stone disease in this patient population.
The study was funded in part by a grant from the American Society of Nephrology. Mr. Loftus reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – Patients taking calcium supplementation after gastric bypass surgery had higher rates of kidney stone growth, compared with those who received no calcium supplementation, results from a single-center retrospective study showed.
In addition, the majority of stones in patients taking calcium supplementation were comprised of calcium oxalate monohydrate, which is less amenable to extracorporeal shockwave therapy. “You need more invasive procedures to break up these kinds of stones,” lead author Christopher Loftus said in an interview at the meeting of the Endocrine Society, where the study was presented during a late-breaking abstract session.
Though it has been demonstrated that bariatric surgery is associated with an increased risk of kidney stone formation (Kidney Int. 2014 [doi:10.1038/ki.2014.352]), Mr. Loftus, a fourth-year medical student at Cleveland Clinic Lerner College of Medicine, and his associates set out to determine whether calcium supplementation increases the risk of nephrolithiasis in 60 stone-forming patients after gastric bypass surgery performed at the Cleveland Clinic. For each patient, two unenhanced CT scans at least 1 month apart and less than 2 years apart were selected at the start of the supplementation date and after the gastric bypass surgery date. The researchers calculated the rate of stone growth by the change in consecutive stone burden (the sum of maximum diameters of stones) divided by the elapsed time between scans.
Of the 60 patients, 31 received postoperative calcium supplementation (an average of 500 mg/day) and 29 did not. Compared with patients who did not take calcium supplementation, those who did were younger (a mean of 53 years vs. 58 years, respectively), more likely to be female (81% vs. 69%), and had a higher body mass index (34.5 kg/m2 vs. 32.7 kg/m2). In addition, a greater proportion of patients taking calcium supplements underwent Roux-en-Y bypass (83% vs. 64%; P = .19), had stones comprised of calcium oxalate (81% vs. 67%; P = .56), and a higher rate of stone growth (more than 10 mm/year vs. less than 5 mm/year; P = .0004). “We weren’t expecting such a pronounced effect,” Mr. Loftus said.
In their abstract, the researchers said that further studies are required to elucidate the exact role of calcium supplementation on stone disease in this patient population.
The study was funded in part by a grant from the American Society of Nephrology. Mr. Loftus reported having no relevant financial conflicts.
On Twitter @dougbrunk
SAN DIEGO – Patients taking calcium supplementation after gastric bypass surgery had higher rates of kidney stone growth, compared with those who received no calcium supplementation, results from a single-center retrospective study showed.
In addition, the majority of stones in patients taking calcium supplementation were comprised of calcium oxalate monohydrate, which is less amenable to extracorporeal shockwave therapy. “You need more invasive procedures to break up these kinds of stones,” lead author Christopher Loftus said in an interview at the meeting of the Endocrine Society, where the study was presented during a late-breaking abstract session.
Though it has been demonstrated that bariatric surgery is associated with an increased risk of kidney stone formation (Kidney Int. 2014 [doi:10.1038/ki.2014.352]), Mr. Loftus, a fourth-year medical student at Cleveland Clinic Lerner College of Medicine, and his associates set out to determine whether calcium supplementation increases the risk of nephrolithiasis in 60 stone-forming patients after gastric bypass surgery performed at the Cleveland Clinic. For each patient, two unenhanced CT scans at least 1 month apart and less than 2 years apart were selected at the start of the supplementation date and after the gastric bypass surgery date. The researchers calculated the rate of stone growth by the change in consecutive stone burden (the sum of maximum diameters of stones) divided by the elapsed time between scans.
Of the 60 patients, 31 received postoperative calcium supplementation (an average of 500 mg/day) and 29 did not. Compared with patients who did not take calcium supplementation, those who did were younger (a mean of 53 years vs. 58 years, respectively), more likely to be female (81% vs. 69%), and had a higher body mass index (34.5 kg/m2 vs. 32.7 kg/m2). In addition, a greater proportion of patients taking calcium supplements underwent Roux-en-Y bypass (83% vs. 64%; P = .19), had stones comprised of calcium oxalate (81% vs. 67%; P = .56), and a higher rate of stone growth (more than 10 mm/year vs. less than 5 mm/year; P = .0004). “We weren’t expecting such a pronounced effect,” Mr. Loftus said.
In their abstract, the researchers said that further studies are required to elucidate the exact role of calcium supplementation on stone disease in this patient population.
The study was funded in part by a grant from the American Society of Nephrology. Mr. Loftus reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
Key clinical point: Calcium supplementation for post-gastric bypass surgery patients may increase the risk of kidney stone growth.
Major finding: Compared with patients who did not take calcium supplementation, those who did had a higher rate of kidney stone growth (more than 10 mm/year vs. less than 5 mm/year; P = .0004).
Data source: An analysis of 60 stone-forming patients who underwent gastric bypass surgery at the Cleveland Clinic.
Disclosures: The study was funded in part by a grant from the American Society of Nephrology. Mr. Loftus reported having no relevant financial conflicts.
Racial differences in birth outcomes noted in women using ART
AT ENDO 2015
SAN DIEGO – Compared to white women undergoing assisted reproductive technology, black women were more likely to have preterm births and deliver small-for-gestational-age infants, a large prospective cohort study demonstrated.
“What this means in the long-term outcomes and what this means as far as counseling [ART patients] is a tricky subject, but I think it does warrant more study,” Dr. Snigdha Alur, the study’s lead author, said in an interview at the meeting of the Endocrine Society.
Assisted reproductive technology “is an increasingly employed form of treatment, prompting interest in which factors could predict or influence its success. Despite black women having the highest prevalence of infertility and ample investigation on racial disparities influencing pregnancy outcomes, few have evaluated whether race also affects ART outcomes,” Dr. Alur of the department of obstetrics and gynecology at the University of Rochester (N.Y.) Medical Center, and her associates wrote in a late-breaking abstract.
In an effort to explore whether adverse pregnancy outcomes differ between white and black women using ART, the researchers evaluated data from 1,871 singleton births to women resulting from ART delivered in the Central and Finger Lakes regions of New York state between 2005 and 2013.
The women completed the state’s perinatal data system questionnaire pertaining to their and their partners’ background and their prenatal care, labor course, and birth outcome. These data were combined with obstetric records including birth size and gestational age. A preterm birth was defined as one delivered at less than 37 weeks’ gestational age, while a very preterm birth was defined as one delivered at less than 34 weeks’ gestational age. A small-for-gestational-age (SGA) infant and a very SGA infant were defined as gender-adjusted weight less than 10% and less than 5%, respectively.
Of the 1,871 singleton births studied, 47 were to black women and 1,824 were to white women. After the researchers adjusted for maternal age, smoking status, highest level of education, and other covariates, black women were significantly more likely to have preterm births (odds ratio, 2.63) but not very preterm births (OR, 2.17), compared with their white counterparts. They also had significantly increased odds of having a SGA (OR, 2.71) or a very SGA infant (OR, 3.01), compared with white women.
“It could be that we have to alter some of our counseling and education based on some of these findings,” Dr. Alur said. “Maybe it’s not just the ART, but there could be other factors playing a role [in poorer outcomes for black ART pregnancies] that need further research.”
Dr. Alur reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
SAN DIEGO – Compared to white women undergoing assisted reproductive technology, black women were more likely to have preterm births and deliver small-for-gestational-age infants, a large prospective cohort study demonstrated.
“What this means in the long-term outcomes and what this means as far as counseling [ART patients] is a tricky subject, but I think it does warrant more study,” Dr. Snigdha Alur, the study’s lead author, said in an interview at the meeting of the Endocrine Society.
Assisted reproductive technology “is an increasingly employed form of treatment, prompting interest in which factors could predict or influence its success. Despite black women having the highest prevalence of infertility and ample investigation on racial disparities influencing pregnancy outcomes, few have evaluated whether race also affects ART outcomes,” Dr. Alur of the department of obstetrics and gynecology at the University of Rochester (N.Y.) Medical Center, and her associates wrote in a late-breaking abstract.
In an effort to explore whether adverse pregnancy outcomes differ between white and black women using ART, the researchers evaluated data from 1,871 singleton births to women resulting from ART delivered in the Central and Finger Lakes regions of New York state between 2005 and 2013.
The women completed the state’s perinatal data system questionnaire pertaining to their and their partners’ background and their prenatal care, labor course, and birth outcome. These data were combined with obstetric records including birth size and gestational age. A preterm birth was defined as one delivered at less than 37 weeks’ gestational age, while a very preterm birth was defined as one delivered at less than 34 weeks’ gestational age. A small-for-gestational-age (SGA) infant and a very SGA infant were defined as gender-adjusted weight less than 10% and less than 5%, respectively.
Of the 1,871 singleton births studied, 47 were to black women and 1,824 were to white women. After the researchers adjusted for maternal age, smoking status, highest level of education, and other covariates, black women were significantly more likely to have preterm births (odds ratio, 2.63) but not very preterm births (OR, 2.17), compared with their white counterparts. They also had significantly increased odds of having a SGA (OR, 2.71) or a very SGA infant (OR, 3.01), compared with white women.
“It could be that we have to alter some of our counseling and education based on some of these findings,” Dr. Alur said. “Maybe it’s not just the ART, but there could be other factors playing a role [in poorer outcomes for black ART pregnancies] that need further research.”
Dr. Alur reported having no relevant financial conflicts.
On Twitter @dougbrunk
AT ENDO 2015
SAN DIEGO – Compared to white women undergoing assisted reproductive technology, black women were more likely to have preterm births and deliver small-for-gestational-age infants, a large prospective cohort study demonstrated.
“What this means in the long-term outcomes and what this means as far as counseling [ART patients] is a tricky subject, but I think it does warrant more study,” Dr. Snigdha Alur, the study’s lead author, said in an interview at the meeting of the Endocrine Society.
Assisted reproductive technology “is an increasingly employed form of treatment, prompting interest in which factors could predict or influence its success. Despite black women having the highest prevalence of infertility and ample investigation on racial disparities influencing pregnancy outcomes, few have evaluated whether race also affects ART outcomes,” Dr. Alur of the department of obstetrics and gynecology at the University of Rochester (N.Y.) Medical Center, and her associates wrote in a late-breaking abstract.
In an effort to explore whether adverse pregnancy outcomes differ between white and black women using ART, the researchers evaluated data from 1,871 singleton births to women resulting from ART delivered in the Central and Finger Lakes regions of New York state between 2005 and 2013.
The women completed the state’s perinatal data system questionnaire pertaining to their and their partners’ background and their prenatal care, labor course, and birth outcome. These data were combined with obstetric records including birth size and gestational age. A preterm birth was defined as one delivered at less than 37 weeks’ gestational age, while a very preterm birth was defined as one delivered at less than 34 weeks’ gestational age. A small-for-gestational-age (SGA) infant and a very SGA infant were defined as gender-adjusted weight less than 10% and less than 5%, respectively.
Of the 1,871 singleton births studied, 47 were to black women and 1,824 were to white women. After the researchers adjusted for maternal age, smoking status, highest level of education, and other covariates, black women were significantly more likely to have preterm births (odds ratio, 2.63) but not very preterm births (OR, 2.17), compared with their white counterparts. They also had significantly increased odds of having a SGA (OR, 2.71) or a very SGA infant (OR, 3.01), compared with white women.
“It could be that we have to alter some of our counseling and education based on some of these findings,” Dr. Alur said. “Maybe it’s not just the ART, but there could be other factors playing a role [in poorer outcomes for black ART pregnancies] that need further research.”
Dr. Alur reported having no relevant financial conflicts.
On Twitter @dougbrunk
Key clinical point: Black women with singleton births from assisted reproductive technology were more likely to have preterm births and deliver SGA infants when compared with white women who also used ART.
Major finding: Black women were significantly more likely to have preterm births (OR 2.63) but not very preterm births (OR 2.17), when compared with their white counterparts. They also had significantly increased odds of having a SGA (OR 2.71) or a very SGA infant (OR 3.01).
Data source: A prospective analysis of 1,871 births to women resulting from ART delivered in the Central and Finger Lakes regions of New York State between 2005 and 2013.
Disclosures: Dr. Alur reported having no relevant financial conflicts.