User login
IQ and concussion recovery
Pediatric concussion is one of those rare phenomena in which we may be witnessing its emergence and clarification in a generation. When I was serving as the game doctor for our local high school football team in the 1970s, I and many other physicians had a very simplistic view of concussion. If the patient never lost conscious and had a reasonably intact short-term memory, we didn’t seriously entertain concussion as a diagnosis. “What’s the score and who is the president?” Were my favorite screening questions.
Obviously, we were underdiagnosing and mismanaging concussion. In part thanks to some high-profile athletes who suffered multiple concussions and eventually chronic traumatic encephalopathy (CTE) physicians began to realize that they should be looking more closely at children who sustained a head injury. The diagnostic criteria were expanded to include any injury that even temporarily effected brain function.
With the new appreciation for the risk of multiple concussions, the focus broadened to include the question of when is it safe for the athlete to return to competition. What signs or symptoms can the patient offer us so we can be sure his or her brain is sufficiently recovered? Here we stepped off into a deep abyss of ignorance. Fortunately, it became obvious fairly quickly that imaging studies weren’t going to help us, as they were invariably normal or at least didn’t tell us anything that wasn’t obvious on a physical exam.
If the patient had a headache, complained of dizziness, or manifested amnesia, monitoring the patient was fairly straightforward. But, in the absence of symptoms and no obvious way to determine the pace of recovery of an organ we couldn’t visualize, clinicians were pulling criteria and time tables out of thin air. Guessing that the concussed brain was in some ways like a torn muscle or overstretched tendon, “brain rest” was often suggested. So no TV, no reading, and certainly none of the cerebral challenging activity of school. Fortunately, we don’t hear much about the notion of brain rest anymore and there is at least one study that suggests that patients kept home from school recover more slowly.
But . Sometimes they describe headache or dizziness but often they complain of a vague mental unwellness. “Brain fog,” a term that has emerged in the wake of the COVID pandemic, might be an apt descriptor. Management of these slow recoverers has been a challenge.
However, two recent articles in the journal Pediatrics may provide some clarity and offer guidance in their management. In a study coming from the psychology department at Georgia State University, researchers reported that they have been able to find “no evidence of clinical meaningful differences in IQ after pediatric concussion.” In their words there is “strong evidence against reduced intelligence in the first few weeks to month after pediatric concussion.”
While their findings may simply toss the IQ onto the pile of worthless measures of healing, a companion commentary by Talin Babikian, PhD, a psychologist at the Semel Institute for Neuroscience and Human Behavior at UCLA, provides a more nuanced interpretation. He writes that if we are looking for an explanation when a patient’s recovery is taking longer than we might expect we need to look beyond some structural damage. Maybe the patient has a previously undiagnosed premorbid condition effecting his or her intellectual, cognitive, or learning abilities. Could the stall in improvement be the result of other symptoms? Here fatigue and sleep deprivation may be the culprits. Could some underlying emotional factor such as anxiety or depression be the problem? For example, I have seen patients whose fear of re-injury has prevented their return to full function. And, finally, the patient may be avoiding a “nonpreferred or challenging situation” unrelated to the injury.
In other words, the concussion may simply be the most obvious rip in a fabric that was already frayed and under stress. This kind of broad holistic (a word I usually like to avoid) thinking may be what is lacking as we struggle to understand other mysterious and chronic conditions such as Lyme disease and chronic fatigue syndrome.
While these two papers help provide some clarity in the management of pediatric concussion, what they fail to address is the bigger question of the relationship between head injury and CTE. The answers to that conundrum are enshrouded in a mix of politics and publicity that I doubt will clear in the near future.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Pediatric concussion is one of those rare phenomena in which we may be witnessing its emergence and clarification in a generation. When I was serving as the game doctor for our local high school football team in the 1970s, I and many other physicians had a very simplistic view of concussion. If the patient never lost conscious and had a reasonably intact short-term memory, we didn’t seriously entertain concussion as a diagnosis. “What’s the score and who is the president?” Were my favorite screening questions.
Obviously, we were underdiagnosing and mismanaging concussion. In part thanks to some high-profile athletes who suffered multiple concussions and eventually chronic traumatic encephalopathy (CTE) physicians began to realize that they should be looking more closely at children who sustained a head injury. The diagnostic criteria were expanded to include any injury that even temporarily effected brain function.
With the new appreciation for the risk of multiple concussions, the focus broadened to include the question of when is it safe for the athlete to return to competition. What signs or symptoms can the patient offer us so we can be sure his or her brain is sufficiently recovered? Here we stepped off into a deep abyss of ignorance. Fortunately, it became obvious fairly quickly that imaging studies weren’t going to help us, as they were invariably normal or at least didn’t tell us anything that wasn’t obvious on a physical exam.
If the patient had a headache, complained of dizziness, or manifested amnesia, monitoring the patient was fairly straightforward. But, in the absence of symptoms and no obvious way to determine the pace of recovery of an organ we couldn’t visualize, clinicians were pulling criteria and time tables out of thin air. Guessing that the concussed brain was in some ways like a torn muscle or overstretched tendon, “brain rest” was often suggested. So no TV, no reading, and certainly none of the cerebral challenging activity of school. Fortunately, we don’t hear much about the notion of brain rest anymore and there is at least one study that suggests that patients kept home from school recover more slowly.
But . Sometimes they describe headache or dizziness but often they complain of a vague mental unwellness. “Brain fog,” a term that has emerged in the wake of the COVID pandemic, might be an apt descriptor. Management of these slow recoverers has been a challenge.
However, two recent articles in the journal Pediatrics may provide some clarity and offer guidance in their management. In a study coming from the psychology department at Georgia State University, researchers reported that they have been able to find “no evidence of clinical meaningful differences in IQ after pediatric concussion.” In their words there is “strong evidence against reduced intelligence in the first few weeks to month after pediatric concussion.”
While their findings may simply toss the IQ onto the pile of worthless measures of healing, a companion commentary by Talin Babikian, PhD, a psychologist at the Semel Institute for Neuroscience and Human Behavior at UCLA, provides a more nuanced interpretation. He writes that if we are looking for an explanation when a patient’s recovery is taking longer than we might expect we need to look beyond some structural damage. Maybe the patient has a previously undiagnosed premorbid condition effecting his or her intellectual, cognitive, or learning abilities. Could the stall in improvement be the result of other symptoms? Here fatigue and sleep deprivation may be the culprits. Could some underlying emotional factor such as anxiety or depression be the problem? For example, I have seen patients whose fear of re-injury has prevented their return to full function. And, finally, the patient may be avoiding a “nonpreferred or challenging situation” unrelated to the injury.
In other words, the concussion may simply be the most obvious rip in a fabric that was already frayed and under stress. This kind of broad holistic (a word I usually like to avoid) thinking may be what is lacking as we struggle to understand other mysterious and chronic conditions such as Lyme disease and chronic fatigue syndrome.
While these two papers help provide some clarity in the management of pediatric concussion, what they fail to address is the bigger question of the relationship between head injury and CTE. The answers to that conundrum are enshrouded in a mix of politics and publicity that I doubt will clear in the near future.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Pediatric concussion is one of those rare phenomena in which we may be witnessing its emergence and clarification in a generation. When I was serving as the game doctor for our local high school football team in the 1970s, I and many other physicians had a very simplistic view of concussion. If the patient never lost conscious and had a reasonably intact short-term memory, we didn’t seriously entertain concussion as a diagnosis. “What’s the score and who is the president?” Were my favorite screening questions.
Obviously, we were underdiagnosing and mismanaging concussion. In part thanks to some high-profile athletes who suffered multiple concussions and eventually chronic traumatic encephalopathy (CTE) physicians began to realize that they should be looking more closely at children who sustained a head injury. The diagnostic criteria were expanded to include any injury that even temporarily effected brain function.
With the new appreciation for the risk of multiple concussions, the focus broadened to include the question of when is it safe for the athlete to return to competition. What signs or symptoms can the patient offer us so we can be sure his or her brain is sufficiently recovered? Here we stepped off into a deep abyss of ignorance. Fortunately, it became obvious fairly quickly that imaging studies weren’t going to help us, as they were invariably normal or at least didn’t tell us anything that wasn’t obvious on a physical exam.
If the patient had a headache, complained of dizziness, or manifested amnesia, monitoring the patient was fairly straightforward. But, in the absence of symptoms and no obvious way to determine the pace of recovery of an organ we couldn’t visualize, clinicians were pulling criteria and time tables out of thin air. Guessing that the concussed brain was in some ways like a torn muscle or overstretched tendon, “brain rest” was often suggested. So no TV, no reading, and certainly none of the cerebral challenging activity of school. Fortunately, we don’t hear much about the notion of brain rest anymore and there is at least one study that suggests that patients kept home from school recover more slowly.
But . Sometimes they describe headache or dizziness but often they complain of a vague mental unwellness. “Brain fog,” a term that has emerged in the wake of the COVID pandemic, might be an apt descriptor. Management of these slow recoverers has been a challenge.
However, two recent articles in the journal Pediatrics may provide some clarity and offer guidance in their management. In a study coming from the psychology department at Georgia State University, researchers reported that they have been able to find “no evidence of clinical meaningful differences in IQ after pediatric concussion.” In their words there is “strong evidence against reduced intelligence in the first few weeks to month after pediatric concussion.”
While their findings may simply toss the IQ onto the pile of worthless measures of healing, a companion commentary by Talin Babikian, PhD, a psychologist at the Semel Institute for Neuroscience and Human Behavior at UCLA, provides a more nuanced interpretation. He writes that if we are looking for an explanation when a patient’s recovery is taking longer than we might expect we need to look beyond some structural damage. Maybe the patient has a previously undiagnosed premorbid condition effecting his or her intellectual, cognitive, or learning abilities. Could the stall in improvement be the result of other symptoms? Here fatigue and sleep deprivation may be the culprits. Could some underlying emotional factor such as anxiety or depression be the problem? For example, I have seen patients whose fear of re-injury has prevented their return to full function. And, finally, the patient may be avoiding a “nonpreferred or challenging situation” unrelated to the injury.
In other words, the concussion may simply be the most obvious rip in a fabric that was already frayed and under stress. This kind of broad holistic (a word I usually like to avoid) thinking may be what is lacking as we struggle to understand other mysterious and chronic conditions such as Lyme disease and chronic fatigue syndrome.
While these two papers help provide some clarity in the management of pediatric concussion, what they fail to address is the bigger question of the relationship between head injury and CTE. The answers to that conundrum are enshrouded in a mix of politics and publicity that I doubt will clear in the near future.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Can a decrease in dopamine lead to binge eating?
In medical school, we were repeatedly advised that there is both a science and an art to the practice of medicine. In these days of doc-in-a-box online consultations for obesity, it’s tempting to think that there’s a one-size-fits-all purely scientific approach for these new weight loss medications. Yet, for every nine patients who lose weight seemingly effortlessly on this class of medication, there is always one whose body stubbornly refuses to submit.
Adam is a 58-year-old man who came to me recently because he was having difficulty losing weight. Over the past 20 years, he’d been steadily gaining weight and now, technically has morbid obesity (a term which should arguably be obsolete). His weight gain is complicated by high blood pressure, high cholesterol, and obstructive sleep apnea. His sleep apnea has caused such profound exhaustion that he no longer has the energy to work out. He also has significant ADHD, which has been left untreated because of his ability to white-knuckle it through his many daily meetings and calls. A married father of three, he is a successful portfolio manager at a high-yield bond fund.
Adam tends to eat minimally during the day, thereby baffling his colleagues with the stark contrast between his minimal caloric intake and his large belly. However, when he returns from work late at night (kids safely tucked into bed), the floodgates open. He reports polishing off pints of ice cream, scarfing down bags of cookies, inhaling trays of brownies. No carbohydrate is off limits to him once he steps off the Metro North train and crosses the threshold from work to home.
Does Adam simply lack the desire or common-sense willpower to make the necessary changes in his lifestyle or is there something more complicated at play?
I would argue that Adam’s ADHD triggered a binge-eating disorder (BED) that festered unchecked over the past 20 years. Patients with BED typically eat massive quantities of food over short periods of time – often when they’re not even hungry. Adam admitted that he would generally continue to eat well after feeling stuffed to the brim.
The answer probably lies with dopamine, a neurotransmitter produced in the reward centers of the brain that regulates how people experience pleasure and control impulses. We believe that people with ADHD have low levels of dopamine (it’s actually a bit more complicated, but this is the general idea). These low levels of dopamine lead people to self-medicate with sugars, salt, and fats to increase dopamine levels.
Lisdexamfetamine (Vyvanse) is a Food and Drug Administration–approved treatment option for both ADHD and binge eating. It raises the levels of dopamine (as well as norepinephrine) in the brain’s reward center. Often, the strong urge to binge subsides rapidly once ADHD is properly treated.
Rather than starting Adam on a semaglutide or similar agent, I opted to start him on lisdexamfetamine. When I spoke to him 1 week later, he confided that the world suddenly shifted into focus, and he was able to plan his meals throughout the day and resist the urge to binge late at night.
I may eventually add a semaglutide-like medication if his weight loss plateaus, but for now, I will focus on raising his dopamine levels to tackle the underlying cause of his weight gain.
Dr. Messer is a clinical assistant professor at the Icahn School of Medicine at Mount Sinai, New York. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In medical school, we were repeatedly advised that there is both a science and an art to the practice of medicine. In these days of doc-in-a-box online consultations for obesity, it’s tempting to think that there’s a one-size-fits-all purely scientific approach for these new weight loss medications. Yet, for every nine patients who lose weight seemingly effortlessly on this class of medication, there is always one whose body stubbornly refuses to submit.
Adam is a 58-year-old man who came to me recently because he was having difficulty losing weight. Over the past 20 years, he’d been steadily gaining weight and now, technically has morbid obesity (a term which should arguably be obsolete). His weight gain is complicated by high blood pressure, high cholesterol, and obstructive sleep apnea. His sleep apnea has caused such profound exhaustion that he no longer has the energy to work out. He also has significant ADHD, which has been left untreated because of his ability to white-knuckle it through his many daily meetings and calls. A married father of three, he is a successful portfolio manager at a high-yield bond fund.
Adam tends to eat minimally during the day, thereby baffling his colleagues with the stark contrast between his minimal caloric intake and his large belly. However, when he returns from work late at night (kids safely tucked into bed), the floodgates open. He reports polishing off pints of ice cream, scarfing down bags of cookies, inhaling trays of brownies. No carbohydrate is off limits to him once he steps off the Metro North train and crosses the threshold from work to home.
Does Adam simply lack the desire or common-sense willpower to make the necessary changes in his lifestyle or is there something more complicated at play?
I would argue that Adam’s ADHD triggered a binge-eating disorder (BED) that festered unchecked over the past 20 years. Patients with BED typically eat massive quantities of food over short periods of time – often when they’re not even hungry. Adam admitted that he would generally continue to eat well after feeling stuffed to the brim.
The answer probably lies with dopamine, a neurotransmitter produced in the reward centers of the brain that regulates how people experience pleasure and control impulses. We believe that people with ADHD have low levels of dopamine (it’s actually a bit more complicated, but this is the general idea). These low levels of dopamine lead people to self-medicate with sugars, salt, and fats to increase dopamine levels.
Lisdexamfetamine (Vyvanse) is a Food and Drug Administration–approved treatment option for both ADHD and binge eating. It raises the levels of dopamine (as well as norepinephrine) in the brain’s reward center. Often, the strong urge to binge subsides rapidly once ADHD is properly treated.
Rather than starting Adam on a semaglutide or similar agent, I opted to start him on lisdexamfetamine. When I spoke to him 1 week later, he confided that the world suddenly shifted into focus, and he was able to plan his meals throughout the day and resist the urge to binge late at night.
I may eventually add a semaglutide-like medication if his weight loss plateaus, but for now, I will focus on raising his dopamine levels to tackle the underlying cause of his weight gain.
Dr. Messer is a clinical assistant professor at the Icahn School of Medicine at Mount Sinai, New York. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In medical school, we were repeatedly advised that there is both a science and an art to the practice of medicine. In these days of doc-in-a-box online consultations for obesity, it’s tempting to think that there’s a one-size-fits-all purely scientific approach for these new weight loss medications. Yet, for every nine patients who lose weight seemingly effortlessly on this class of medication, there is always one whose body stubbornly refuses to submit.
Adam is a 58-year-old man who came to me recently because he was having difficulty losing weight. Over the past 20 years, he’d been steadily gaining weight and now, technically has morbid obesity (a term which should arguably be obsolete). His weight gain is complicated by high blood pressure, high cholesterol, and obstructive sleep apnea. His sleep apnea has caused such profound exhaustion that he no longer has the energy to work out. He also has significant ADHD, which has been left untreated because of his ability to white-knuckle it through his many daily meetings and calls. A married father of three, he is a successful portfolio manager at a high-yield bond fund.
Adam tends to eat minimally during the day, thereby baffling his colleagues with the stark contrast between his minimal caloric intake and his large belly. However, when he returns from work late at night (kids safely tucked into bed), the floodgates open. He reports polishing off pints of ice cream, scarfing down bags of cookies, inhaling trays of brownies. No carbohydrate is off limits to him once he steps off the Metro North train and crosses the threshold from work to home.
Does Adam simply lack the desire or common-sense willpower to make the necessary changes in his lifestyle or is there something more complicated at play?
I would argue that Adam’s ADHD triggered a binge-eating disorder (BED) that festered unchecked over the past 20 years. Patients with BED typically eat massive quantities of food over short periods of time – often when they’re not even hungry. Adam admitted that he would generally continue to eat well after feeling stuffed to the brim.
The answer probably lies with dopamine, a neurotransmitter produced in the reward centers of the brain that regulates how people experience pleasure and control impulses. We believe that people with ADHD have low levels of dopamine (it’s actually a bit more complicated, but this is the general idea). These low levels of dopamine lead people to self-medicate with sugars, salt, and fats to increase dopamine levels.
Lisdexamfetamine (Vyvanse) is a Food and Drug Administration–approved treatment option for both ADHD and binge eating. It raises the levels of dopamine (as well as norepinephrine) in the brain’s reward center. Often, the strong urge to binge subsides rapidly once ADHD is properly treated.
Rather than starting Adam on a semaglutide or similar agent, I opted to start him on lisdexamfetamine. When I spoke to him 1 week later, he confided that the world suddenly shifted into focus, and he was able to plan his meals throughout the day and resist the urge to binge late at night.
I may eventually add a semaglutide-like medication if his weight loss plateaus, but for now, I will focus on raising his dopamine levels to tackle the underlying cause of his weight gain.
Dr. Messer is a clinical assistant professor at the Icahn School of Medicine at Mount Sinai, New York. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
When does a bicarb drip make sense?
A 70-year-old woman is admitted to the intensive care unit with a pH of 7.1, an acute kidney injury (AKI), and ketonuria. She is volume depleted and her history is consistent with starvation ketosis. This LOL truly is in NAD (that’s little old lady in no acute distress, for those who haven’t read The House of God). She is clinically stable and seemingly unperturbed by the flurry of activity surrounding her admission.
Your resident is concerned by the severity of the acidosis and suggests starting an intravenous bicarbonate drip. The fellow is adamantly against it. He’s been taught that intravenous bicarbonate increases the serum pH but paradoxically causes intracellular acidosis. As the attending you elect to observe fellow autonomy – no bicarb is given. Because any debate on rounds is a “teachable moment,” you decide to review the evidence and physiology behind infusing bicarbonate.
What do the data reveal?
An excellent review published in CHEST in 2000 covers the physiologic effects of bicarbonate, specifically related to lactic acidosis, which our patient didn’t have. Aside from that difference, the review validates the fellow’s opinion. In short, It is unlikely to provoke hemodynamic or respiratory compromise outside the setting of shock or hypercapnia. Intravenous bicarbonate can lead to intracellular acidosis, hypercapnia, hypocalcemia, and a reduction in oxygen delivery via the Bohr effect. The authors concluded that because the benefits are unproven and the negative effects are real, intravenous bicarbonate should not be used to correct a metabolic acidosis.
The CHEST review hardly settles the issue, though. A survey published a few years later found a majority of intensivists and nephrologists used intravenous bicarbonate to treat metabolic acidosis while the Surviving Sepsis Campaign Guidelines for the Management of Sepsis and Septic Shock published in 2017 recommended against bicarbonate for acidosis. It wasn’t until 2018 that we reached the holy grail: a randomized controlled trial.
The BICAR-ICU study randomly assigned patients with a pH of 7.20 or less, PCO2 of 45 mm Hg or less, and sodium bicarbonate concentration of 20 mmol/L or less to receive no bicarbonate versus a sodium bicarbonate drip to maintain a pH greater than 7.30. There’s additional nuance to the trial design and even more detail in the results. To summarize, there was signal for an improvement in renal outcomes across all patients, and those with AKI saw a mortality benefit. Post–BICAR-ICU iterations of the Surviving Sepsis Campaign Guidelines have incorporated these findings by recommending intravenous bicarbonate for patients with sepsis who have AKI and a pH of 7.20 or less.
That’s not to say BICAR-ICU has settled the issue. Although it’s far and away the best we have, there were fewer than 400 total patients in their intention-to-treat analysis. It was open label, with lots of crossover. The primary outcome was negative for the entire population, with only a subgroup (albeit a prespecified one) showing benefit. Finally, the results weren’t stratified by etiology for the metabolic acidosis. There was also evidence of alkalosis and hypocalcemia in the treatment group.
Last but not least in terms of importance, in most cases when bicarbonate is being considered, wouldn’t some form of renal replacement therapy (RRT) be preferred? This point was raised by nephrologists and intensivists when we covered BICAR-ICU in a journal club at my former program. It’s also mentioned in an accompanying editorial. RRT timing is controversial, and a detailed discussion is outside the scope of this piece and beyond the limits of my current knowledge base. But I do know that the A in the A-E-I-O-U acute indications for dialysis pneumonic stands for acidosis.
Our patient had AKI, a pH of 7.20 or less, and a pCO2 well under 45 mm Hg. Does BICAR-ICU support the resident’s inclination to start a drip? Sort of. The majority of patients enrolled in BICAR-ICU were in shock or were recovering from cardiac arrest, so it’s not clear the results can be generalized to our LOL with starvation ketosis. Extrapolating from studies of diabetic ketoacidosis (DKA) seems more appropriate, and here the data are poor but equivocal. Reviews are generally negative but don’t rule out the use of intravenous bicarbonate in certain patients with DKA.
Key takeaways
Our patient survived a 24-hour ICU stay with neither cardiopulmonary decompensation nor a need for RRT. Not sure how she did out of the ICU; presumably she was discharged soon after transfer. As is always the case with anecdotal medicine, the absence of a control prevents assessment of the counterfactual. Is it possible she may have done “better” with intravenous bicarbonate? Seems unlikely to me, though I doubt there would have been demonstrable adverse effects. Perhaps next time the fellow can observe resident autonomy?
Aaron B. Holley, MD, is a professor of medicine at Uniformed Services University of the Health Sciences, Bethesda, Md., and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center. He reported conflicts of interest with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
A 70-year-old woman is admitted to the intensive care unit with a pH of 7.1, an acute kidney injury (AKI), and ketonuria. She is volume depleted and her history is consistent with starvation ketosis. This LOL truly is in NAD (that’s little old lady in no acute distress, for those who haven’t read The House of God). She is clinically stable and seemingly unperturbed by the flurry of activity surrounding her admission.
Your resident is concerned by the severity of the acidosis and suggests starting an intravenous bicarbonate drip. The fellow is adamantly against it. He’s been taught that intravenous bicarbonate increases the serum pH but paradoxically causes intracellular acidosis. As the attending you elect to observe fellow autonomy – no bicarb is given. Because any debate on rounds is a “teachable moment,” you decide to review the evidence and physiology behind infusing bicarbonate.
What do the data reveal?
An excellent review published in CHEST in 2000 covers the physiologic effects of bicarbonate, specifically related to lactic acidosis, which our patient didn’t have. Aside from that difference, the review validates the fellow’s opinion. In short, It is unlikely to provoke hemodynamic or respiratory compromise outside the setting of shock or hypercapnia. Intravenous bicarbonate can lead to intracellular acidosis, hypercapnia, hypocalcemia, and a reduction in oxygen delivery via the Bohr effect. The authors concluded that because the benefits are unproven and the negative effects are real, intravenous bicarbonate should not be used to correct a metabolic acidosis.
The CHEST review hardly settles the issue, though. A survey published a few years later found a majority of intensivists and nephrologists used intravenous bicarbonate to treat metabolic acidosis while the Surviving Sepsis Campaign Guidelines for the Management of Sepsis and Septic Shock published in 2017 recommended against bicarbonate for acidosis. It wasn’t until 2018 that we reached the holy grail: a randomized controlled trial.
The BICAR-ICU study randomly assigned patients with a pH of 7.20 or less, PCO2 of 45 mm Hg or less, and sodium bicarbonate concentration of 20 mmol/L or less to receive no bicarbonate versus a sodium bicarbonate drip to maintain a pH greater than 7.30. There’s additional nuance to the trial design and even more detail in the results. To summarize, there was signal for an improvement in renal outcomes across all patients, and those with AKI saw a mortality benefit. Post–BICAR-ICU iterations of the Surviving Sepsis Campaign Guidelines have incorporated these findings by recommending intravenous bicarbonate for patients with sepsis who have AKI and a pH of 7.20 or less.
That’s not to say BICAR-ICU has settled the issue. Although it’s far and away the best we have, there were fewer than 400 total patients in their intention-to-treat analysis. It was open label, with lots of crossover. The primary outcome was negative for the entire population, with only a subgroup (albeit a prespecified one) showing benefit. Finally, the results weren’t stratified by etiology for the metabolic acidosis. There was also evidence of alkalosis and hypocalcemia in the treatment group.
Last but not least in terms of importance, in most cases when bicarbonate is being considered, wouldn’t some form of renal replacement therapy (RRT) be preferred? This point was raised by nephrologists and intensivists when we covered BICAR-ICU in a journal club at my former program. It’s also mentioned in an accompanying editorial. RRT timing is controversial, and a detailed discussion is outside the scope of this piece and beyond the limits of my current knowledge base. But I do know that the A in the A-E-I-O-U acute indications for dialysis pneumonic stands for acidosis.
Our patient had AKI, a pH of 7.20 or less, and a pCO2 well under 45 mm Hg. Does BICAR-ICU support the resident’s inclination to start a drip? Sort of. The majority of patients enrolled in BICAR-ICU were in shock or were recovering from cardiac arrest, so it’s not clear the results can be generalized to our LOL with starvation ketosis. Extrapolating from studies of diabetic ketoacidosis (DKA) seems more appropriate, and here the data are poor but equivocal. Reviews are generally negative but don’t rule out the use of intravenous bicarbonate in certain patients with DKA.
Key takeaways
Our patient survived a 24-hour ICU stay with neither cardiopulmonary decompensation nor a need for RRT. Not sure how she did out of the ICU; presumably she was discharged soon after transfer. As is always the case with anecdotal medicine, the absence of a control prevents assessment of the counterfactual. Is it possible she may have done “better” with intravenous bicarbonate? Seems unlikely to me, though I doubt there would have been demonstrable adverse effects. Perhaps next time the fellow can observe resident autonomy?
Aaron B. Holley, MD, is a professor of medicine at Uniformed Services University of the Health Sciences, Bethesda, Md., and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center. He reported conflicts of interest with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
A 70-year-old woman is admitted to the intensive care unit with a pH of 7.1, an acute kidney injury (AKI), and ketonuria. She is volume depleted and her history is consistent with starvation ketosis. This LOL truly is in NAD (that’s little old lady in no acute distress, for those who haven’t read The House of God). She is clinically stable and seemingly unperturbed by the flurry of activity surrounding her admission.
Your resident is concerned by the severity of the acidosis and suggests starting an intravenous bicarbonate drip. The fellow is adamantly against it. He’s been taught that intravenous bicarbonate increases the serum pH but paradoxically causes intracellular acidosis. As the attending you elect to observe fellow autonomy – no bicarb is given. Because any debate on rounds is a “teachable moment,” you decide to review the evidence and physiology behind infusing bicarbonate.
What do the data reveal?
An excellent review published in CHEST in 2000 covers the physiologic effects of bicarbonate, specifically related to lactic acidosis, which our patient didn’t have. Aside from that difference, the review validates the fellow’s opinion. In short, It is unlikely to provoke hemodynamic or respiratory compromise outside the setting of shock or hypercapnia. Intravenous bicarbonate can lead to intracellular acidosis, hypercapnia, hypocalcemia, and a reduction in oxygen delivery via the Bohr effect. The authors concluded that because the benefits are unproven and the negative effects are real, intravenous bicarbonate should not be used to correct a metabolic acidosis.
The CHEST review hardly settles the issue, though. A survey published a few years later found a majority of intensivists and nephrologists used intravenous bicarbonate to treat metabolic acidosis while the Surviving Sepsis Campaign Guidelines for the Management of Sepsis and Septic Shock published in 2017 recommended against bicarbonate for acidosis. It wasn’t until 2018 that we reached the holy grail: a randomized controlled trial.
The BICAR-ICU study randomly assigned patients with a pH of 7.20 or less, PCO2 of 45 mm Hg or less, and sodium bicarbonate concentration of 20 mmol/L or less to receive no bicarbonate versus a sodium bicarbonate drip to maintain a pH greater than 7.30. There’s additional nuance to the trial design and even more detail in the results. To summarize, there was signal for an improvement in renal outcomes across all patients, and those with AKI saw a mortality benefit. Post–BICAR-ICU iterations of the Surviving Sepsis Campaign Guidelines have incorporated these findings by recommending intravenous bicarbonate for patients with sepsis who have AKI and a pH of 7.20 or less.
That’s not to say BICAR-ICU has settled the issue. Although it’s far and away the best we have, there were fewer than 400 total patients in their intention-to-treat analysis. It was open label, with lots of crossover. The primary outcome was negative for the entire population, with only a subgroup (albeit a prespecified one) showing benefit. Finally, the results weren’t stratified by etiology for the metabolic acidosis. There was also evidence of alkalosis and hypocalcemia in the treatment group.
Last but not least in terms of importance, in most cases when bicarbonate is being considered, wouldn’t some form of renal replacement therapy (RRT) be preferred? This point was raised by nephrologists and intensivists when we covered BICAR-ICU in a journal club at my former program. It’s also mentioned in an accompanying editorial. RRT timing is controversial, and a detailed discussion is outside the scope of this piece and beyond the limits of my current knowledge base. But I do know that the A in the A-E-I-O-U acute indications for dialysis pneumonic stands for acidosis.
Our patient had AKI, a pH of 7.20 or less, and a pCO2 well under 45 mm Hg. Does BICAR-ICU support the resident’s inclination to start a drip? Sort of. The majority of patients enrolled in BICAR-ICU were in shock or were recovering from cardiac arrest, so it’s not clear the results can be generalized to our LOL with starvation ketosis. Extrapolating from studies of diabetic ketoacidosis (DKA) seems more appropriate, and here the data are poor but equivocal. Reviews are generally negative but don’t rule out the use of intravenous bicarbonate in certain patients with DKA.
Key takeaways
Our patient survived a 24-hour ICU stay with neither cardiopulmonary decompensation nor a need for RRT. Not sure how she did out of the ICU; presumably she was discharged soon after transfer. As is always the case with anecdotal medicine, the absence of a control prevents assessment of the counterfactual. Is it possible she may have done “better” with intravenous bicarbonate? Seems unlikely to me, though I doubt there would have been demonstrable adverse effects. Perhaps next time the fellow can observe resident autonomy?
Aaron B. Holley, MD, is a professor of medicine at Uniformed Services University of the Health Sciences, Bethesda, Md., and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center. He reported conflicts of interest with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
The magic of music
I’m really going to miss Jimmy Buffett.
I’ve liked his music as far back as I can remember, and was lucky enough to see him in person in the mid-90s.
I’ve written about music before, but its affect on us never fails to amaze me. Songs can be background noise conducive to getting things done. They can also be in the foreground, serving as a mental vacation (or accompanying a real one). They can transport you to another place, briefly clearing your head from the daily goings-on around you. Even if it’s just during the drive home, it’s a welcome escape to a virtual beach and tropical drink.
Songs can bring back memories of certain events or people that we link them to. My dad loved anything by Neil Diamond, and nothing brings back thoughts of Dad more than when my iTunes randomly picks “I Am ... I Said.” Or John Williams’ Star Wars theme, taking me back to the summer of 1977 when I sat, spellbound, by this incredible movie whose magic is still going strong two generations later.
It’s amazing how our brain tries to make music out of nothing. Even in silence we have ear worms, the songs stuck in our head for hours to days (recently I’ve had “I Sing the Body Electric” from the 1980 movie Fame playing in there).
My office is over an MRI scanner, so I can always hear the chiller pumps softly running in the background. Sometimes my brain will turn their rhythmic chirping into a song, altering the pace of the song to fit them. The soft clicking of the ceiling fan, in my home office, does the same thing (for some reason my brain usually tries to fit “Yellow Submarine” to that one, no idea why).
Music is a part of that mysterious essence that makes us human. It touches all of us in some way, which varies between people, songs, and artists.
Jimmy Buffet’s music has a vacation vibe. Songs of the Caribbean & Keys, beaches, bars, boats, and tropical drinks. The 4:12 running time of his most well-known song, “Margaritaville,” gives a brief respite from my day when it comes on.
He passes into the beyond, to the sadness of his family, friends, and fans. But, unlike people, music can be immortal, and so he lives on through his creations. Like, Bach, Lennon, Bowie, Joplin, Sousa, and too many others to count, his work – and the enjoyment we get from it – are a gift left behind for the future.
Tight lines, Jimmy.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I’m really going to miss Jimmy Buffett.
I’ve liked his music as far back as I can remember, and was lucky enough to see him in person in the mid-90s.
I’ve written about music before, but its affect on us never fails to amaze me. Songs can be background noise conducive to getting things done. They can also be in the foreground, serving as a mental vacation (or accompanying a real one). They can transport you to another place, briefly clearing your head from the daily goings-on around you. Even if it’s just during the drive home, it’s a welcome escape to a virtual beach and tropical drink.
Songs can bring back memories of certain events or people that we link them to. My dad loved anything by Neil Diamond, and nothing brings back thoughts of Dad more than when my iTunes randomly picks “I Am ... I Said.” Or John Williams’ Star Wars theme, taking me back to the summer of 1977 when I sat, spellbound, by this incredible movie whose magic is still going strong two generations later.
It’s amazing how our brain tries to make music out of nothing. Even in silence we have ear worms, the songs stuck in our head for hours to days (recently I’ve had “I Sing the Body Electric” from the 1980 movie Fame playing in there).
My office is over an MRI scanner, so I can always hear the chiller pumps softly running in the background. Sometimes my brain will turn their rhythmic chirping into a song, altering the pace of the song to fit them. The soft clicking of the ceiling fan, in my home office, does the same thing (for some reason my brain usually tries to fit “Yellow Submarine” to that one, no idea why).
Music is a part of that mysterious essence that makes us human. It touches all of us in some way, which varies between people, songs, and artists.
Jimmy Buffet’s music has a vacation vibe. Songs of the Caribbean & Keys, beaches, bars, boats, and tropical drinks. The 4:12 running time of his most well-known song, “Margaritaville,” gives a brief respite from my day when it comes on.
He passes into the beyond, to the sadness of his family, friends, and fans. But, unlike people, music can be immortal, and so he lives on through his creations. Like, Bach, Lennon, Bowie, Joplin, Sousa, and too many others to count, his work – and the enjoyment we get from it – are a gift left behind for the future.
Tight lines, Jimmy.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I’m really going to miss Jimmy Buffett.
I’ve liked his music as far back as I can remember, and was lucky enough to see him in person in the mid-90s.
I’ve written about music before, but its affect on us never fails to amaze me. Songs can be background noise conducive to getting things done. They can also be in the foreground, serving as a mental vacation (or accompanying a real one). They can transport you to another place, briefly clearing your head from the daily goings-on around you. Even if it’s just during the drive home, it’s a welcome escape to a virtual beach and tropical drink.
Songs can bring back memories of certain events or people that we link them to. My dad loved anything by Neil Diamond, and nothing brings back thoughts of Dad more than when my iTunes randomly picks “I Am ... I Said.” Or John Williams’ Star Wars theme, taking me back to the summer of 1977 when I sat, spellbound, by this incredible movie whose magic is still going strong two generations later.
It’s amazing how our brain tries to make music out of nothing. Even in silence we have ear worms, the songs stuck in our head for hours to days (recently I’ve had “I Sing the Body Electric” from the 1980 movie Fame playing in there).
My office is over an MRI scanner, so I can always hear the chiller pumps softly running in the background. Sometimes my brain will turn their rhythmic chirping into a song, altering the pace of the song to fit them. The soft clicking of the ceiling fan, in my home office, does the same thing (for some reason my brain usually tries to fit “Yellow Submarine” to that one, no idea why).
Music is a part of that mysterious essence that makes us human. It touches all of us in some way, which varies between people, songs, and artists.
Jimmy Buffet’s music has a vacation vibe. Songs of the Caribbean & Keys, beaches, bars, boats, and tropical drinks. The 4:12 running time of his most well-known song, “Margaritaville,” gives a brief respite from my day when it comes on.
He passes into the beyond, to the sadness of his family, friends, and fans. But, unlike people, music can be immortal, and so he lives on through his creations. Like, Bach, Lennon, Bowie, Joplin, Sousa, and too many others to count, his work – and the enjoyment we get from it – are a gift left behind for the future.
Tight lines, Jimmy.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
The most important study from ESC: FRAIL-AF
One of the hardest tasks of a clinician is applying evidence from trials to the person in your office. At the annual congress of the European Society of Cardiology, the surprising and unexpected results of the FRAIL-AF trial confirm the massive challenge of evidence translation.
FRAIL-AF investigators set out to study the question of whether frail, elderly patients with atrial fibrillation who were doing well with vitamin K antagonists (VKA) should be switched to direct-acting oral anticoagulants (DOAC).
Senior author Geert-Jan Geersing, MD, PhD, from the University Medical Center Utrecht (the Netherlands), told me that frustration led him to design this study. He was frustrated that colleagues assumed that evidence in nonfrail patients can always be translated to frail patients.
Dr. Geersing offered two reasons why common wisdom may be wrong. First was that the large DOAC versus warfarin trials included few elderly patients with frailty. Second, first author Linda Joosten, MD, made it clear in her presentation that frailty is a lot more than aging. It is a clinical syndrome, which entails a “high burden of comorbidities, dependency on others, and a reduced ability to resist stressors.”
The FRAIL-AF trial
The investigators recruited elderly, frail patients with fibrillation who were treated with VKAs and had stable international normalized ratios from outpatient clinics throughout the Netherlands. They screened about 2,600 patients and enrolled nearly 1,400. Most were excluded for not being frail.
Half the group was randomized to switching to a DOAC – drug choice was left to the treating clinician – and the other half remained on VKAs. Patients were 83 years of age on average with a mean CHA2DS2-VASc score of 4. All four classes of DOAC were used in the switching arm.
The primary endpoint was major or clinically relevant nonmajor bleeding, whichever came first, accounting for death as a competing risk. Follow-up was 1 year.
The results for switching to DOAC vs. VKA
Dr. Joosten started her presentation with this: “The results turned out to be different than we expected.” The authors designed the trial with the idea that switching to DOACs would be superior in safety to remaining on VKAs.
But the trial was halted after an interim analysis found a rate of major bleeding in the switching arm of 15.3% versus 9.4% in the arm staying on VKA (hazard ratio, 1.69; 95% confidence interval, 1.23-2.32; P = .0012).
The Kaplan-Meier event curves reveal that the excess risk of bleeding occurred after 100 days and increased with time. This argued against an early effect from transitioning the drugs.
An analysis looking at specific DOAC drugs revealed similar hazards for the two most common ones used – apixaban and rivaroxaban.
Thrombotic events were a secondary endpoint and were low in absolute numbers, 2.4% versus 2.0%, for remaining on VKA and switching to DOAC, respectively (HR, 1.26; 95% CI, 0.60-2.61).
The time in therapeutic range in FRAIL-AF was similar to that in the seminal DOAC trials.
Comments
Three reasons lead me to choose FRAIL-AF as the most important study from the 2023 ESC congress.
First is the specific lesson about switching drugs. Note that FRAIL-AF did not address the question of starting anticoagulation. The trial results show that if you have a frail older patient who is doing well on VKA, don’t change to a DOAC. That is important to know, but it is not what gives this study its heft.
The second reason centers on the investigators choice to do this trial. Dr. Geersing had a feeling that common wisdom was wrong. He did not try to persuade colleagues with anecdote or plausibility or meta-analyses of observational studies. He set out to answer a question in the correct way – with a randomized trial.
This is the path forward in medicine. I’ve often heard proponents of observational research declare that many topics in medicine cannot be studied with trials. I could hear people arguing that it’s not feasible to study mostly home-bound, elderly frail patients. And the fact that there exist so few trials in this space would support that argument.
But the FRAIL-AF authors showed that it is possible. This is the kind of science that medicine should celebrate. There were no soft endpoints, financial conflicts, or spin. If medical science had science as its incentive, rather than attention, FRAIL-AF easily wins top honors.
The third reason FRAIL-AF is so important is that it teaches us the humility required in translating evidence in our clinics. I like to say evidence is what separates doctors from palm readers. But using this evidence requires thinking hard about how average effects in trial environments apply to our patient.
Yes, of course, there is clear evidence from tens of thousands of patients in the DOAC versus warfarin trials, that, for those patients, on average, DOACs compare favorably with VKA. The average age of patients in these trials was 70-73 years; the average age in FRAIL-AF was 83 years. And that is just age. A substudy of the ENGAGE AF-TIMI 48 trial found that only 360 of more than 20,000 patients in the trial had severe frailty.
That lesson extends to nearly every common therapy in medicine today. It also casts great doubt on the soft-thinking idea of using evidence from trials to derive quality metrics. As if the nuance of evidence translation can be captured in an electronic health record.
The skillful use of evidence will be one of the main challenges of the next generation of clinicians. Thanks to advances in medical science, more patients will live long enough to become frail. And the so-called “guideline-directed” therapies may not apply to them.
Dr. Joosten, Dr. Geersing, and the FRAIL-AF team have taught us specific lessons about anticoagulation, but their greatest contribution has been to demonstrate the value of humility in science and the practice of evidence-based medicine.
If you treat patients, no trial at this meeting is more important.
Dr. Mandrola is a clinical electrophysiologist at Baptist Medical Associates, Louisville, Ky. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
One of the hardest tasks of a clinician is applying evidence from trials to the person in your office. At the annual congress of the European Society of Cardiology, the surprising and unexpected results of the FRAIL-AF trial confirm the massive challenge of evidence translation.
FRAIL-AF investigators set out to study the question of whether frail, elderly patients with atrial fibrillation who were doing well with vitamin K antagonists (VKA) should be switched to direct-acting oral anticoagulants (DOAC).
Senior author Geert-Jan Geersing, MD, PhD, from the University Medical Center Utrecht (the Netherlands), told me that frustration led him to design this study. He was frustrated that colleagues assumed that evidence in nonfrail patients can always be translated to frail patients.
Dr. Geersing offered two reasons why common wisdom may be wrong. First was that the large DOAC versus warfarin trials included few elderly patients with frailty. Second, first author Linda Joosten, MD, made it clear in her presentation that frailty is a lot more than aging. It is a clinical syndrome, which entails a “high burden of comorbidities, dependency on others, and a reduced ability to resist stressors.”
The FRAIL-AF trial
The investigators recruited elderly, frail patients with fibrillation who were treated with VKAs and had stable international normalized ratios from outpatient clinics throughout the Netherlands. They screened about 2,600 patients and enrolled nearly 1,400. Most were excluded for not being frail.
Half the group was randomized to switching to a DOAC – drug choice was left to the treating clinician – and the other half remained on VKAs. Patients were 83 years of age on average with a mean CHA2DS2-VASc score of 4. All four classes of DOAC were used in the switching arm.
The primary endpoint was major or clinically relevant nonmajor bleeding, whichever came first, accounting for death as a competing risk. Follow-up was 1 year.
The results for switching to DOAC vs. VKA
Dr. Joosten started her presentation with this: “The results turned out to be different than we expected.” The authors designed the trial with the idea that switching to DOACs would be superior in safety to remaining on VKAs.
But the trial was halted after an interim analysis found a rate of major bleeding in the switching arm of 15.3% versus 9.4% in the arm staying on VKA (hazard ratio, 1.69; 95% confidence interval, 1.23-2.32; P = .0012).
The Kaplan-Meier event curves reveal that the excess risk of bleeding occurred after 100 days and increased with time. This argued against an early effect from transitioning the drugs.
An analysis looking at specific DOAC drugs revealed similar hazards for the two most common ones used – apixaban and rivaroxaban.
Thrombotic events were a secondary endpoint and were low in absolute numbers, 2.4% versus 2.0%, for remaining on VKA and switching to DOAC, respectively (HR, 1.26; 95% CI, 0.60-2.61).
The time in therapeutic range in FRAIL-AF was similar to that in the seminal DOAC trials.
Comments
Three reasons lead me to choose FRAIL-AF as the most important study from the 2023 ESC congress.
First is the specific lesson about switching drugs. Note that FRAIL-AF did not address the question of starting anticoagulation. The trial results show that if you have a frail older patient who is doing well on VKA, don’t change to a DOAC. That is important to know, but it is not what gives this study its heft.
The second reason centers on the investigators choice to do this trial. Dr. Geersing had a feeling that common wisdom was wrong. He did not try to persuade colleagues with anecdote or plausibility or meta-analyses of observational studies. He set out to answer a question in the correct way – with a randomized trial.
This is the path forward in medicine. I’ve often heard proponents of observational research declare that many topics in medicine cannot be studied with trials. I could hear people arguing that it’s not feasible to study mostly home-bound, elderly frail patients. And the fact that there exist so few trials in this space would support that argument.
But the FRAIL-AF authors showed that it is possible. This is the kind of science that medicine should celebrate. There were no soft endpoints, financial conflicts, or spin. If medical science had science as its incentive, rather than attention, FRAIL-AF easily wins top honors.
The third reason FRAIL-AF is so important is that it teaches us the humility required in translating evidence in our clinics. I like to say evidence is what separates doctors from palm readers. But using this evidence requires thinking hard about how average effects in trial environments apply to our patient.
Yes, of course, there is clear evidence from tens of thousands of patients in the DOAC versus warfarin trials, that, for those patients, on average, DOACs compare favorably with VKA. The average age of patients in these trials was 70-73 years; the average age in FRAIL-AF was 83 years. And that is just age. A substudy of the ENGAGE AF-TIMI 48 trial found that only 360 of more than 20,000 patients in the trial had severe frailty.
That lesson extends to nearly every common therapy in medicine today. It also casts great doubt on the soft-thinking idea of using evidence from trials to derive quality metrics. As if the nuance of evidence translation can be captured in an electronic health record.
The skillful use of evidence will be one of the main challenges of the next generation of clinicians. Thanks to advances in medical science, more patients will live long enough to become frail. And the so-called “guideline-directed” therapies may not apply to them.
Dr. Joosten, Dr. Geersing, and the FRAIL-AF team have taught us specific lessons about anticoagulation, but their greatest contribution has been to demonstrate the value of humility in science and the practice of evidence-based medicine.
If you treat patients, no trial at this meeting is more important.
Dr. Mandrola is a clinical electrophysiologist at Baptist Medical Associates, Louisville, Ky. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
One of the hardest tasks of a clinician is applying evidence from trials to the person in your office. At the annual congress of the European Society of Cardiology, the surprising and unexpected results of the FRAIL-AF trial confirm the massive challenge of evidence translation.
FRAIL-AF investigators set out to study the question of whether frail, elderly patients with atrial fibrillation who were doing well with vitamin K antagonists (VKA) should be switched to direct-acting oral anticoagulants (DOAC).
Senior author Geert-Jan Geersing, MD, PhD, from the University Medical Center Utrecht (the Netherlands), told me that frustration led him to design this study. He was frustrated that colleagues assumed that evidence in nonfrail patients can always be translated to frail patients.
Dr. Geersing offered two reasons why common wisdom may be wrong. First was that the large DOAC versus warfarin trials included few elderly patients with frailty. Second, first author Linda Joosten, MD, made it clear in her presentation that frailty is a lot more than aging. It is a clinical syndrome, which entails a “high burden of comorbidities, dependency on others, and a reduced ability to resist stressors.”
The FRAIL-AF trial
The investigators recruited elderly, frail patients with fibrillation who were treated with VKAs and had stable international normalized ratios from outpatient clinics throughout the Netherlands. They screened about 2,600 patients and enrolled nearly 1,400. Most were excluded for not being frail.
Half the group was randomized to switching to a DOAC – drug choice was left to the treating clinician – and the other half remained on VKAs. Patients were 83 years of age on average with a mean CHA2DS2-VASc score of 4. All four classes of DOAC were used in the switching arm.
The primary endpoint was major or clinically relevant nonmajor bleeding, whichever came first, accounting for death as a competing risk. Follow-up was 1 year.
The results for switching to DOAC vs. VKA
Dr. Joosten started her presentation with this: “The results turned out to be different than we expected.” The authors designed the trial with the idea that switching to DOACs would be superior in safety to remaining on VKAs.
But the trial was halted after an interim analysis found a rate of major bleeding in the switching arm of 15.3% versus 9.4% in the arm staying on VKA (hazard ratio, 1.69; 95% confidence interval, 1.23-2.32; P = .0012).
The Kaplan-Meier event curves reveal that the excess risk of bleeding occurred after 100 days and increased with time. This argued against an early effect from transitioning the drugs.
An analysis looking at specific DOAC drugs revealed similar hazards for the two most common ones used – apixaban and rivaroxaban.
Thrombotic events were a secondary endpoint and were low in absolute numbers, 2.4% versus 2.0%, for remaining on VKA and switching to DOAC, respectively (HR, 1.26; 95% CI, 0.60-2.61).
The time in therapeutic range in FRAIL-AF was similar to that in the seminal DOAC trials.
Comments
Three reasons lead me to choose FRAIL-AF as the most important study from the 2023 ESC congress.
First is the specific lesson about switching drugs. Note that FRAIL-AF did not address the question of starting anticoagulation. The trial results show that if you have a frail older patient who is doing well on VKA, don’t change to a DOAC. That is important to know, but it is not what gives this study its heft.
The second reason centers on the investigators choice to do this trial. Dr. Geersing had a feeling that common wisdom was wrong. He did not try to persuade colleagues with anecdote or plausibility or meta-analyses of observational studies. He set out to answer a question in the correct way – with a randomized trial.
This is the path forward in medicine. I’ve often heard proponents of observational research declare that many topics in medicine cannot be studied with trials. I could hear people arguing that it’s not feasible to study mostly home-bound, elderly frail patients. And the fact that there exist so few trials in this space would support that argument.
But the FRAIL-AF authors showed that it is possible. This is the kind of science that medicine should celebrate. There were no soft endpoints, financial conflicts, or spin. If medical science had science as its incentive, rather than attention, FRAIL-AF easily wins top honors.
The third reason FRAIL-AF is so important is that it teaches us the humility required in translating evidence in our clinics. I like to say evidence is what separates doctors from palm readers. But using this evidence requires thinking hard about how average effects in trial environments apply to our patient.
Yes, of course, there is clear evidence from tens of thousands of patients in the DOAC versus warfarin trials, that, for those patients, on average, DOACs compare favorably with VKA. The average age of patients in these trials was 70-73 years; the average age in FRAIL-AF was 83 years. And that is just age. A substudy of the ENGAGE AF-TIMI 48 trial found that only 360 of more than 20,000 patients in the trial had severe frailty.
That lesson extends to nearly every common therapy in medicine today. It also casts great doubt on the soft-thinking idea of using evidence from trials to derive quality metrics. As if the nuance of evidence translation can be captured in an electronic health record.
The skillful use of evidence will be one of the main challenges of the next generation of clinicians. Thanks to advances in medical science, more patients will live long enough to become frail. And the so-called “guideline-directed” therapies may not apply to them.
Dr. Joosten, Dr. Geersing, and the FRAIL-AF team have taught us specific lessons about anticoagulation, but their greatest contribution has been to demonstrate the value of humility in science and the practice of evidence-based medicine.
If you treat patients, no trial at this meeting is more important.
Dr. Mandrola is a clinical electrophysiologist at Baptist Medical Associates, Louisville, Ky. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
‘Decapitated’ boy saved by surgery team
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: I am joined today by Dr. Ohad Einav. He’s a staff surgeon in orthopedics at Hadassah Medical Center in Jerusalem. He’s with me to talk about an absolutely incredible surgical case, something that is terrifying to most non–orthopedic surgeons and I imagine is fairly scary for spine surgeons like him as well. But what we don’t have is information about how this works from a medical perspective. So, first of all, Dr. Einav, thank you for taking time to speak with me today.
Ohad Einav, MD: Thank you for having me.
Dr. Wilson: Can you tell us about Suleiman Hassan and what happened to him before he came into your care?
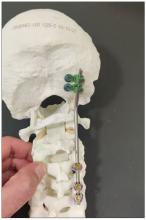
Dr. Einav: Hassan is a 12-year-old child who was riding his bicycle on the West Bank, about 40 minutes from here. Unfortunately, he was involved in a motor vehicle accident and he suffered injuries to his abdomen and cervical spine. He was transported to our service by helicopter from the scene of the accident.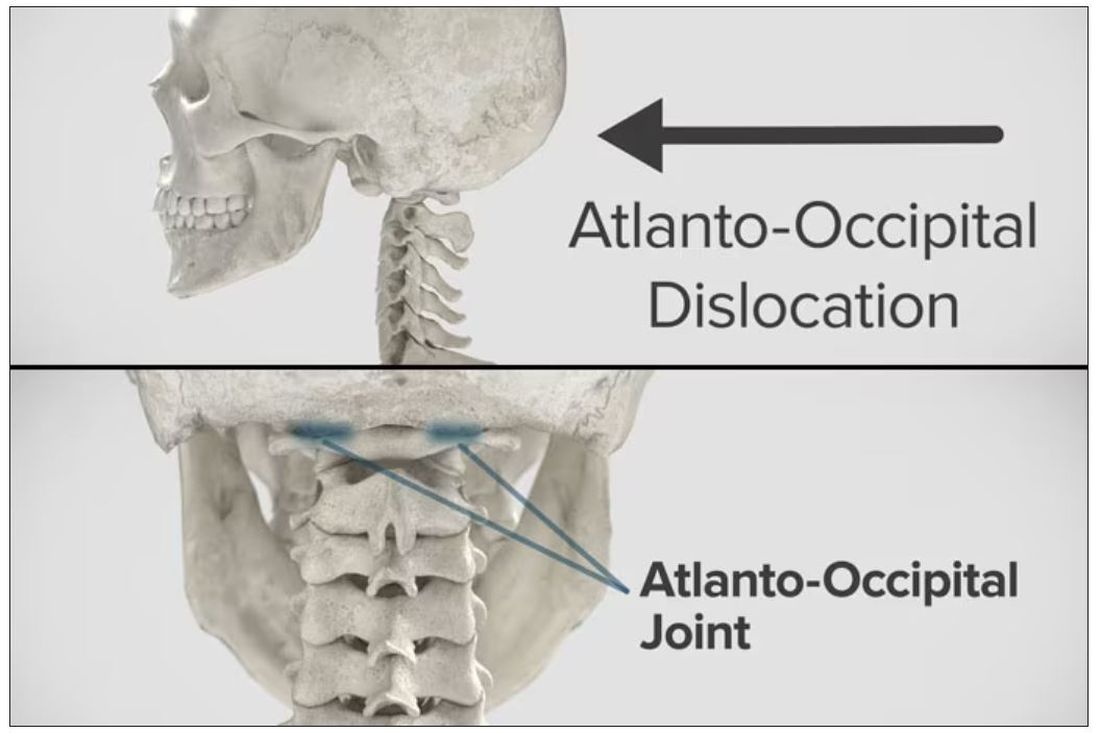
Dr. Wilson: “Injury to the cervical spine” might be something of an understatement. He had what’s called atlanto-occipital dislocation, colloquially often referred to as internal decapitation. Can you tell us what that means? It sounds terrifying.
Dr. Einav: It’s an injury to the ligaments between the occiput and the upper cervical spine, with or without bony fracture. The atlanto-occipital joint is formed by the superior articular facet of the atlas and the occipital condyle, stabilized by an articular capsule between the head and neck, and is supported by various ligaments around it that stabilize the joint and allow joint movements, including flexion, extension, and some rotation in the lower levels.
Dr. Wilson: This joint has several degrees of freedom, which means it needs a lot of support. With this type of injury, where essentially you have severing of the ligaments, is it usually survivable? How dangerous is this?
Dr. Einav: The mortality rate is 50%-60%, depending on the primary impact, the injury, transportation later on, and then the surgery and surgical management.
Dr. Wilson: Tell us a bit about this patient’s status when he came to your medical center. I assume he was in bad shape.
Dr. Einav: Hassan arrived at our medical center with a Glasgow Coma Scale score of 15. He was fully conscious. He was hemodynamically stable except for a bad laceration on his abdomen. He had a Philadelphia collar around his neck. He was transported by chopper because the paramedics suspected that he had a cervical spine injury and decided to bring him to a Level 1 trauma center.
He was monitored and we treated him according to the ATLS [advanced trauma life support] protocol. He didn’t have any gross sensory deficits, but he was a little confused about the whole situation and the accident. Therefore, we could do a general examination but we couldn’t rely on that regarding any sensory deficit that he may or may not have. We decided as a team that it would be better to slow down and control the situation. We decided not to operate on him immediately. We basically stabilized him and made sure that he didn’t have any traumatic internal organ damage. Later on we took him to the OR and performed surgery.
Dr. Wilson: It’s amazing that he had intact motor function, considering the extent of his injury. The spinal cord was spared somewhat during the injury. There must have been a moment when you realized that this kid, who was conscious and could move all four extremities, had a very severe neck injury. Was that due to a CT scan or physical exam? And what was your feeling when you saw that he had atlanto-occipital dislocation?
Dr. Einav: As a surgeon, you have a gut feeling in regard to the general examination of the patient. But I never rely on gut feelings. On the CT, I understood exactly what he had, what we needed to do, and the time frame.
Dr. Wilson: You’ve done these types of surgeries before, right? Obviously, no one has done a lot of them because this isn’t very common. But you knew what to do. Did you have a plan? Where does your experience come into play in a situation like this?
Dr. Einav: I graduated from the spine program of Toronto University, where I did a fellowship in trauma of the spine and complex spine surgery. I had very good teachers, and during my fellowship I treated a few cases in older patients that were similar but not the same. Therefore, I knew exactly what needed to be done.
Dr. Wilson: For those of us who aren’t surgeons, take us into the OR with you. This is obviously an incredibly delicate procedure. You are high up in the spinal cord at the base of the brain. The slightest mistake could have devastating consequences. What are the key elements of this procedure? What can go wrong here? What is the number-one thing you have to look out for when you’re trying to fix an internal decapitation?
Dr. Einav: The key element in surgeries of the cervical spine – trauma and complex spine surgery – is planning. I never go to the OR without knowing what I’m going to do. I have a few plans – plan A, plan B, plan C – in case something fails. So, I definitely know what the next step will be. I always think about the surgery a few hours before, if I have time to prepare.
The second thing that is very important is teamwork. The team needs to be coordinated. Everybody needs to know what their job is. With these types of injuries, it’s not the time for rookies. If you are new, please stand back and let the more experienced people do that job. I’m talking about surgeons, nurses, anesthesiologists – everyone.
Another important thing in planning is choosing the right hardware. For example, in this case we had a problem because most of the hardware is designed for adults, and we had to improvise because there isn’t a lot of hardware on the market for the pediatric population. The adult plates and screws are too big, so we had to improvise.
Dr. Wilson: Tell us more about that. How do you improvise spinal hardware for a 12-year-old?
Dr. Einav: In this case, I chose to use hardware from one of the companies that works with us.
You can see in this model the area of the injury, and the area that we worked on. To perform the surgery, I had to use some plates and rods from a different company. This company’s (NuVasive) hardware has a small attachment to the skull, which was helpful for affixing the skull to the cervical spine, instead of using a big plate that would sit at the base of the skull and would not be very good for him. Most of the hardware is made for adults and not for kids.
Dr. Wilson: Will that hardware preserve the motor function of his neck? Will he be able to turn his head and extend and flex it?
Dr. Einav: The injury leads to instability and destruction of both articulations between the head and neck. Therefore, those articulations won’t be able to function the same way in the future. There is a decrease of something like 50% of the flexion and extension of Hassan’s cervical spine. Therefore, I decided that in this case there would be no chance of saving Hassan’s motor function unless we performed a fusion between the head and the neck, and therefore I decided that this would be the best procedure with the best survival rate. So, in the future, he will have some diminished flexion, extension, and rotation of his head.
Dr. Wilson: How long did his surgery take?
Dr. Einav: To be honest, I don’t remember. But I can tell you that it took us time. It was very challenging to coordinate with everyone. The most problematic part of the surgery to perform is what we call “flip-over.”
The anesthesiologist intubated the patient when he was supine, and later on, we flipped him prone to operate on the spine. This maneuver can actually lead to injury by itself, and injury at this level is fatal. So, we took our time and got Hassan into the OR. The anesthesiologist did a great job with the GlideScope – inserting the endotracheal tube. Later on, we neuromonitored him. Basically, we connected Hassan’s peripheral nerves to a computer and monitored his motor function. Gently we flipped him over, and after that we saw a little change in his motor function, so we had to modify his position so we could preserve his motor function. We then started the procedure, which took a few hours. I don’t know exactly how many.
Dr. Wilson: That just speaks to how delicate this is for everything from the intubation, where typically you’re manipulating the head, to the repositioning. Clearly this requires a lot of teamwork.
What happened after the operation? How is he doing?
Dr. Einav: After the operation, Hassan had a great recovery. He’s doing well. He doesn’t have any motor or sensory deficits. He’s able to ambulate without any aid. He had no signs of infection, which can happen after a car accident, neither from his abdominal wound nor from the occipital cervical surgery. He feels well. We saw him in the clinic. We removed his collar. We monitored him at the clinic. He looked amazing.
Dr. Wilson: That’s incredible. Are there long-term risks for him that you need to be looking out for?
Dr. Einav: Yes, and that’s the reason that we are monitoring him post surgery. While he was in the hospital, we monitored his motor and sensory functions, as well as his wound healing. Later on, in the clinic, for a few weeks after surgery we monitored for any failure of the hardware and bone graft. We check for healing of the bone graft and bone substitutes we put in to heal those bones.
Dr. Wilson: He will grow, right? He’s only 12, so he still has some years of growth in him. Is he going to need more surgery or any kind of hardware upgrade?
Dr. Einav: I hope not. In my surgeries, I never rely on the hardware for long durations. If I decide to do, for example, fusion, I rely on the hardware for a certain amount of time. And then I plan that the biology will do the work. If I plan for fusion, I put bone grafts in the preferred area for a fusion. Then if the hardware fails, I wouldn’t need to take out the hardware, and there would be no change in the condition of the patient.
Dr. Wilson: What an incredible story. It’s clear that you and your team kept your cool despite a very high-acuity situation with a ton of risk. What a tremendous outcome that this boy is not only alive but fully functional. So, congratulations to you and your team. That was very strong work.
Dr. Einav: Thank you very much. I would like to thank our team. We have to remember that the surgeon is not standing alone in the war. Hassan’s story is a success story of a very big group of people from various backgrounds and religions. They work day and night to help people and save lives. To the paramedics, the physiologists, the traumatologists, the pediatricians, the nurses, the physiotherapists, and obviously the surgeons, a big thank you. His story is our success story.
Dr. Wilson: It’s inspiring to see so many people come together to do what we all are here for, which is to fight against suffering, disease, and death. Thank you for keeping up that fight. And thank you for joining me here.
Dr. Einav: Thank you very much.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: I am joined today by Dr. Ohad Einav. He’s a staff surgeon in orthopedics at Hadassah Medical Center in Jerusalem. He’s with me to talk about an absolutely incredible surgical case, something that is terrifying to most non–orthopedic surgeons and I imagine is fairly scary for spine surgeons like him as well. But what we don’t have is information about how this works from a medical perspective. So, first of all, Dr. Einav, thank you for taking time to speak with me today.
Ohad Einav, MD: Thank you for having me.
Dr. Wilson: Can you tell us about Suleiman Hassan and what happened to him before he came into your care?
Dr. Einav: Hassan is a 12-year-old child who was riding his bicycle on the West Bank, about 40 minutes from here. Unfortunately, he was involved in a motor vehicle accident and he suffered injuries to his abdomen and cervical spine. He was transported to our service by helicopter from the scene of the accident.
Dr. Wilson: “Injury to the cervical spine” might be something of an understatement. He had what’s called atlanto-occipital dislocation, colloquially often referred to as internal decapitation. Can you tell us what that means? It sounds terrifying.
Dr. Einav: It’s an injury to the ligaments between the occiput and the upper cervical spine, with or without bony fracture. The atlanto-occipital joint is formed by the superior articular facet of the atlas and the occipital condyle, stabilized by an articular capsule between the head and neck, and is supported by various ligaments around it that stabilize the joint and allow joint movements, including flexion, extension, and some rotation in the lower levels.
Dr. Wilson: This joint has several degrees of freedom, which means it needs a lot of support. With this type of injury, where essentially you have severing of the ligaments, is it usually survivable? How dangerous is this?
Dr. Einav: The mortality rate is 50%-60%, depending on the primary impact, the injury, transportation later on, and then the surgery and surgical management.
Dr. Wilson: Tell us a bit about this patient’s status when he came to your medical center. I assume he was in bad shape.
Dr. Einav: Hassan arrived at our medical center with a Glasgow Coma Scale score of 15. He was fully conscious. He was hemodynamically stable except for a bad laceration on his abdomen. He had a Philadelphia collar around his neck. He was transported by chopper because the paramedics suspected that he had a cervical spine injury and decided to bring him to a Level 1 trauma center.
He was monitored and we treated him according to the ATLS [advanced trauma life support] protocol. He didn’t have any gross sensory deficits, but he was a little confused about the whole situation and the accident. Therefore, we could do a general examination but we couldn’t rely on that regarding any sensory deficit that he may or may not have. We decided as a team that it would be better to slow down and control the situation. We decided not to operate on him immediately. We basically stabilized him and made sure that he didn’t have any traumatic internal organ damage. Later on we took him to the OR and performed surgery.
Dr. Wilson: It’s amazing that he had intact motor function, considering the extent of his injury. The spinal cord was spared somewhat during the injury. There must have been a moment when you realized that this kid, who was conscious and could move all four extremities, had a very severe neck injury. Was that due to a CT scan or physical exam? And what was your feeling when you saw that he had atlanto-occipital dislocation?
Dr. Einav: As a surgeon, you have a gut feeling in regard to the general examination of the patient. But I never rely on gut feelings. On the CT, I understood exactly what he had, what we needed to do, and the time frame.
Dr. Wilson: You’ve done these types of surgeries before, right? Obviously, no one has done a lot of them because this isn’t very common. But you knew what to do. Did you have a plan? Where does your experience come into play in a situation like this?
Dr. Einav: I graduated from the spine program of Toronto University, where I did a fellowship in trauma of the spine and complex spine surgery. I had very good teachers, and during my fellowship I treated a few cases in older patients that were similar but not the same. Therefore, I knew exactly what needed to be done.
Dr. Wilson: For those of us who aren’t surgeons, take us into the OR with you. This is obviously an incredibly delicate procedure. You are high up in the spinal cord at the base of the brain. The slightest mistake could have devastating consequences. What are the key elements of this procedure? What can go wrong here? What is the number-one thing you have to look out for when you’re trying to fix an internal decapitation?
Dr. Einav: The key element in surgeries of the cervical spine – trauma and complex spine surgery – is planning. I never go to the OR without knowing what I’m going to do. I have a few plans – plan A, plan B, plan C – in case something fails. So, I definitely know what the next step will be. I always think about the surgery a few hours before, if I have time to prepare.
The second thing that is very important is teamwork. The team needs to be coordinated. Everybody needs to know what their job is. With these types of injuries, it’s not the time for rookies. If you are new, please stand back and let the more experienced people do that job. I’m talking about surgeons, nurses, anesthesiologists – everyone.
Another important thing in planning is choosing the right hardware. For example, in this case we had a problem because most of the hardware is designed for adults, and we had to improvise because there isn’t a lot of hardware on the market for the pediatric population. The adult plates and screws are too big, so we had to improvise.
Dr. Wilson: Tell us more about that. How do you improvise spinal hardware for a 12-year-old?
Dr. Einav: In this case, I chose to use hardware from one of the companies that works with us.
You can see in this model the area of the injury, and the area that we worked on. To perform the surgery, I had to use some plates and rods from a different company. This company’s (NuVasive) hardware has a small attachment to the skull, which was helpful for affixing the skull to the cervical spine, instead of using a big plate that would sit at the base of the skull and would not be very good for him. Most of the hardware is made for adults and not for kids.
Dr. Wilson: Will that hardware preserve the motor function of his neck? Will he be able to turn his head and extend and flex it?
Dr. Einav: The injury leads to instability and destruction of both articulations between the head and neck. Therefore, those articulations won’t be able to function the same way in the future. There is a decrease of something like 50% of the flexion and extension of Hassan’s cervical spine. Therefore, I decided that in this case there would be no chance of saving Hassan’s motor function unless we performed a fusion between the head and the neck, and therefore I decided that this would be the best procedure with the best survival rate. So, in the future, he will have some diminished flexion, extension, and rotation of his head.
Dr. Wilson: How long did his surgery take?
Dr. Einav: To be honest, I don’t remember. But I can tell you that it took us time. It was very challenging to coordinate with everyone. The most problematic part of the surgery to perform is what we call “flip-over.”
The anesthesiologist intubated the patient when he was supine, and later on, we flipped him prone to operate on the spine. This maneuver can actually lead to injury by itself, and injury at this level is fatal. So, we took our time and got Hassan into the OR. The anesthesiologist did a great job with the GlideScope – inserting the endotracheal tube. Later on, we neuromonitored him. Basically, we connected Hassan’s peripheral nerves to a computer and monitored his motor function. Gently we flipped him over, and after that we saw a little change in his motor function, so we had to modify his position so we could preserve his motor function. We then started the procedure, which took a few hours. I don’t know exactly how many.
Dr. Wilson: That just speaks to how delicate this is for everything from the intubation, where typically you’re manipulating the head, to the repositioning. Clearly this requires a lot of teamwork.
What happened after the operation? How is he doing?
Dr. Einav: After the operation, Hassan had a great recovery. He’s doing well. He doesn’t have any motor or sensory deficits. He’s able to ambulate without any aid. He had no signs of infection, which can happen after a car accident, neither from his abdominal wound nor from the occipital cervical surgery. He feels well. We saw him in the clinic. We removed his collar. We monitored him at the clinic. He looked amazing.
Dr. Wilson: That’s incredible. Are there long-term risks for him that you need to be looking out for?
Dr. Einav: Yes, and that’s the reason that we are monitoring him post surgery. While he was in the hospital, we monitored his motor and sensory functions, as well as his wound healing. Later on, in the clinic, for a few weeks after surgery we monitored for any failure of the hardware and bone graft. We check for healing of the bone graft and bone substitutes we put in to heal those bones.
Dr. Wilson: He will grow, right? He’s only 12, so he still has some years of growth in him. Is he going to need more surgery or any kind of hardware upgrade?
Dr. Einav: I hope not. In my surgeries, I never rely on the hardware for long durations. If I decide to do, for example, fusion, I rely on the hardware for a certain amount of time. And then I plan that the biology will do the work. If I plan for fusion, I put bone grafts in the preferred area for a fusion. Then if the hardware fails, I wouldn’t need to take out the hardware, and there would be no change in the condition of the patient.
Dr. Wilson: What an incredible story. It’s clear that you and your team kept your cool despite a very high-acuity situation with a ton of risk. What a tremendous outcome that this boy is not only alive but fully functional. So, congratulations to you and your team. That was very strong work.
Dr. Einav: Thank you very much. I would like to thank our team. We have to remember that the surgeon is not standing alone in the war. Hassan’s story is a success story of a very big group of people from various backgrounds and religions. They work day and night to help people and save lives. To the paramedics, the physiologists, the traumatologists, the pediatricians, the nurses, the physiotherapists, and obviously the surgeons, a big thank you. His story is our success story.
Dr. Wilson: It’s inspiring to see so many people come together to do what we all are here for, which is to fight against suffering, disease, and death. Thank you for keeping up that fight. And thank you for joining me here.
Dr. Einav: Thank you very much.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: I am joined today by Dr. Ohad Einav. He’s a staff surgeon in orthopedics at Hadassah Medical Center in Jerusalem. He’s with me to talk about an absolutely incredible surgical case, something that is terrifying to most non–orthopedic surgeons and I imagine is fairly scary for spine surgeons like him as well. But what we don’t have is information about how this works from a medical perspective. So, first of all, Dr. Einav, thank you for taking time to speak with me today.
Ohad Einav, MD: Thank you for having me.
Dr. Wilson: Can you tell us about Suleiman Hassan and what happened to him before he came into your care?
Dr. Einav: Hassan is a 12-year-old child who was riding his bicycle on the West Bank, about 40 minutes from here. Unfortunately, he was involved in a motor vehicle accident and he suffered injuries to his abdomen and cervical spine. He was transported to our service by helicopter from the scene of the accident.
Dr. Wilson: “Injury to the cervical spine” might be something of an understatement. He had what’s called atlanto-occipital dislocation, colloquially often referred to as internal decapitation. Can you tell us what that means? It sounds terrifying.
Dr. Einav: It’s an injury to the ligaments between the occiput and the upper cervical spine, with or without bony fracture. The atlanto-occipital joint is formed by the superior articular facet of the atlas and the occipital condyle, stabilized by an articular capsule between the head and neck, and is supported by various ligaments around it that stabilize the joint and allow joint movements, including flexion, extension, and some rotation in the lower levels.
Dr. Wilson: This joint has several degrees of freedom, which means it needs a lot of support. With this type of injury, where essentially you have severing of the ligaments, is it usually survivable? How dangerous is this?
Dr. Einav: The mortality rate is 50%-60%, depending on the primary impact, the injury, transportation later on, and then the surgery and surgical management.
Dr. Wilson: Tell us a bit about this patient’s status when he came to your medical center. I assume he was in bad shape.
Dr. Einav: Hassan arrived at our medical center with a Glasgow Coma Scale score of 15. He was fully conscious. He was hemodynamically stable except for a bad laceration on his abdomen. He had a Philadelphia collar around his neck. He was transported by chopper because the paramedics suspected that he had a cervical spine injury and decided to bring him to a Level 1 trauma center.
He was monitored and we treated him according to the ATLS [advanced trauma life support] protocol. He didn’t have any gross sensory deficits, but he was a little confused about the whole situation and the accident. Therefore, we could do a general examination but we couldn’t rely on that regarding any sensory deficit that he may or may not have. We decided as a team that it would be better to slow down and control the situation. We decided not to operate on him immediately. We basically stabilized him and made sure that he didn’t have any traumatic internal organ damage. Later on we took him to the OR and performed surgery.
Dr. Wilson: It’s amazing that he had intact motor function, considering the extent of his injury. The spinal cord was spared somewhat during the injury. There must have been a moment when you realized that this kid, who was conscious and could move all four extremities, had a very severe neck injury. Was that due to a CT scan or physical exam? And what was your feeling when you saw that he had atlanto-occipital dislocation?
Dr. Einav: As a surgeon, you have a gut feeling in regard to the general examination of the patient. But I never rely on gut feelings. On the CT, I understood exactly what he had, what we needed to do, and the time frame.
Dr. Wilson: You’ve done these types of surgeries before, right? Obviously, no one has done a lot of them because this isn’t very common. But you knew what to do. Did you have a plan? Where does your experience come into play in a situation like this?
Dr. Einav: I graduated from the spine program of Toronto University, where I did a fellowship in trauma of the spine and complex spine surgery. I had very good teachers, and during my fellowship I treated a few cases in older patients that were similar but not the same. Therefore, I knew exactly what needed to be done.
Dr. Wilson: For those of us who aren’t surgeons, take us into the OR with you. This is obviously an incredibly delicate procedure. You are high up in the spinal cord at the base of the brain. The slightest mistake could have devastating consequences. What are the key elements of this procedure? What can go wrong here? What is the number-one thing you have to look out for when you’re trying to fix an internal decapitation?
Dr. Einav: The key element in surgeries of the cervical spine – trauma and complex spine surgery – is planning. I never go to the OR without knowing what I’m going to do. I have a few plans – plan A, plan B, plan C – in case something fails. So, I definitely know what the next step will be. I always think about the surgery a few hours before, if I have time to prepare.
The second thing that is very important is teamwork. The team needs to be coordinated. Everybody needs to know what their job is. With these types of injuries, it’s not the time for rookies. If you are new, please stand back and let the more experienced people do that job. I’m talking about surgeons, nurses, anesthesiologists – everyone.
Another important thing in planning is choosing the right hardware. For example, in this case we had a problem because most of the hardware is designed for adults, and we had to improvise because there isn’t a lot of hardware on the market for the pediatric population. The adult plates and screws are too big, so we had to improvise.
Dr. Wilson: Tell us more about that. How do you improvise spinal hardware for a 12-year-old?
Dr. Einav: In this case, I chose to use hardware from one of the companies that works with us.
You can see in this model the area of the injury, and the area that we worked on. To perform the surgery, I had to use some plates and rods from a different company. This company’s (NuVasive) hardware has a small attachment to the skull, which was helpful for affixing the skull to the cervical spine, instead of using a big plate that would sit at the base of the skull and would not be very good for him. Most of the hardware is made for adults and not for kids.
Dr. Wilson: Will that hardware preserve the motor function of his neck? Will he be able to turn his head and extend and flex it?
Dr. Einav: The injury leads to instability and destruction of both articulations between the head and neck. Therefore, those articulations won’t be able to function the same way in the future. There is a decrease of something like 50% of the flexion and extension of Hassan’s cervical spine. Therefore, I decided that in this case there would be no chance of saving Hassan’s motor function unless we performed a fusion between the head and the neck, and therefore I decided that this would be the best procedure with the best survival rate. So, in the future, he will have some diminished flexion, extension, and rotation of his head.
Dr. Wilson: How long did his surgery take?
Dr. Einav: To be honest, I don’t remember. But I can tell you that it took us time. It was very challenging to coordinate with everyone. The most problematic part of the surgery to perform is what we call “flip-over.”
The anesthesiologist intubated the patient when he was supine, and later on, we flipped him prone to operate on the spine. This maneuver can actually lead to injury by itself, and injury at this level is fatal. So, we took our time and got Hassan into the OR. The anesthesiologist did a great job with the GlideScope – inserting the endotracheal tube. Later on, we neuromonitored him. Basically, we connected Hassan’s peripheral nerves to a computer and monitored his motor function. Gently we flipped him over, and after that we saw a little change in his motor function, so we had to modify his position so we could preserve his motor function. We then started the procedure, which took a few hours. I don’t know exactly how many.
Dr. Wilson: That just speaks to how delicate this is for everything from the intubation, where typically you’re manipulating the head, to the repositioning. Clearly this requires a lot of teamwork.
What happened after the operation? How is he doing?
Dr. Einav: After the operation, Hassan had a great recovery. He’s doing well. He doesn’t have any motor or sensory deficits. He’s able to ambulate without any aid. He had no signs of infection, which can happen after a car accident, neither from his abdominal wound nor from the occipital cervical surgery. He feels well. We saw him in the clinic. We removed his collar. We monitored him at the clinic. He looked amazing.
Dr. Wilson: That’s incredible. Are there long-term risks for him that you need to be looking out for?
Dr. Einav: Yes, and that’s the reason that we are monitoring him post surgery. While he was in the hospital, we monitored his motor and sensory functions, as well as his wound healing. Later on, in the clinic, for a few weeks after surgery we monitored for any failure of the hardware and bone graft. We check for healing of the bone graft and bone substitutes we put in to heal those bones.
Dr. Wilson: He will grow, right? He’s only 12, so he still has some years of growth in him. Is he going to need more surgery or any kind of hardware upgrade?
Dr. Einav: I hope not. In my surgeries, I never rely on the hardware for long durations. If I decide to do, for example, fusion, I rely on the hardware for a certain amount of time. And then I plan that the biology will do the work. If I plan for fusion, I put bone grafts in the preferred area for a fusion. Then if the hardware fails, I wouldn’t need to take out the hardware, and there would be no change in the condition of the patient.
Dr. Wilson: What an incredible story. It’s clear that you and your team kept your cool despite a very high-acuity situation with a ton of risk. What a tremendous outcome that this boy is not only alive but fully functional. So, congratulations to you and your team. That was very strong work.
Dr. Einav: Thank you very much. I would like to thank our team. We have to remember that the surgeon is not standing alone in the war. Hassan’s story is a success story of a very big group of people from various backgrounds and religions. They work day and night to help people and save lives. To the paramedics, the physiologists, the traumatologists, the pediatricians, the nurses, the physiotherapists, and obviously the surgeons, a big thank you. His story is our success story.
Dr. Wilson: It’s inspiring to see so many people come together to do what we all are here for, which is to fight against suffering, disease, and death. Thank you for keeping up that fight. And thank you for joining me here.
Dr. Einav: Thank you very much.
A version of this article first appeared on Medscape.com.
Targeted warnings
I was probably about 9 or 10 and I am assuming it was early winter when my mother took me aside and said in her usual quiet tone, “Willy, don’t ever stick your tongue on a metal pipe when it is cold outside.”
Putting my tongue on a frozen pipe was something that had never occurred to me even in my wildest preadolescent dreams. My mother’s caution only served to pique my interest and provide me with one more tempting scenario to consider.
Recently, a prank has gone viral on TikTok that shows an adult, usually the parent, cracking (not smashing) an egg on the child’s head and then emptying the egg contents into a bowl. Unlike the tongue-pipe disaster, it is hard to imagine how this stunt can be dangerous as long as the child is old enough to be walking around. But, at least one pediatrician has warned that there is a risk to the child from contracting salmonella.
There may be a few young children who are frightened by having an egg cracked on their head, but I can’t imagine that it would leave any lasting emotional scars. Given the minuscule theoretical risk of infection and the fact that the videos have accumulated more than 670 million views, this is another example of when we “experts” should keep a low profile and let the virus fade into Internet oblivion.
There is, however, a difference between harmless foolishness and stupidity, and one wonders when and in what manner we pediatricians should become involved. For example, in a recent study published in the journal Pediatrics, the investigators searched through a national emergency department database and found that
There were two peaks of distribution, one at less than 1 year of age and another at age 4. The older children were more often injured playing on furniture, most often bunk beds. The younger children were more likely to have been injured by being lifted or tossed in the air. No deaths were reported.
Is this a phenomenon that demands a response by pediatricians? Do we have time to ask every family if they have a ceiling fan? Should we be handing out brochures to every family? To whom should we target our message? This is a situation that seems to sort easily into two categories. One that involves stupidity and a second that is ignorance that may respond to education.
Tossing young children in the air is fun for the tosser and the child. I am sure there are a few children every year who slip out of the grasp of an adult and are injured. I have never seen a child brought in with this history. But it must happen. The result is likely to trigger a very tricky child protective investigation. But tossing a child underneath a ceiling fan is just plain stupid. I’m not sure our intervention is going to prevent it from happening. Bunk beds and ceiling fans are a different story. Posters in our offices and warnings and labels at the point of purchase of both fans and bunk beds makes some sense.
And while we are sticking labels on furniture, we should take a hard look at couches. Researchers have recently found that the accumulation of sedentary time in childhood can lead to early evidence of heart damage, which may portend heart disease in adulthood. Instead of those tags under the cushions, we need a big blaze orange sticker in prominent view that warns of the danger of becoming a couch potato.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
I was probably about 9 or 10 and I am assuming it was early winter when my mother took me aside and said in her usual quiet tone, “Willy, don’t ever stick your tongue on a metal pipe when it is cold outside.”
Putting my tongue on a frozen pipe was something that had never occurred to me even in my wildest preadolescent dreams. My mother’s caution only served to pique my interest and provide me with one more tempting scenario to consider.
Recently, a prank has gone viral on TikTok that shows an adult, usually the parent, cracking (not smashing) an egg on the child’s head and then emptying the egg contents into a bowl. Unlike the tongue-pipe disaster, it is hard to imagine how this stunt can be dangerous as long as the child is old enough to be walking around. But, at least one pediatrician has warned that there is a risk to the child from contracting salmonella.
There may be a few young children who are frightened by having an egg cracked on their head, but I can’t imagine that it would leave any lasting emotional scars. Given the minuscule theoretical risk of infection and the fact that the videos have accumulated more than 670 million views, this is another example of when we “experts” should keep a low profile and let the virus fade into Internet oblivion.
There is, however, a difference between harmless foolishness and stupidity, and one wonders when and in what manner we pediatricians should become involved. For example, in a recent study published in the journal Pediatrics, the investigators searched through a national emergency department database and found that
There were two peaks of distribution, one at less than 1 year of age and another at age 4. The older children were more often injured playing on furniture, most often bunk beds. The younger children were more likely to have been injured by being lifted or tossed in the air. No deaths were reported.
Is this a phenomenon that demands a response by pediatricians? Do we have time to ask every family if they have a ceiling fan? Should we be handing out brochures to every family? To whom should we target our message? This is a situation that seems to sort easily into two categories. One that involves stupidity and a second that is ignorance that may respond to education.
Tossing young children in the air is fun for the tosser and the child. I am sure there are a few children every year who slip out of the grasp of an adult and are injured. I have never seen a child brought in with this history. But it must happen. The result is likely to trigger a very tricky child protective investigation. But tossing a child underneath a ceiling fan is just plain stupid. I’m not sure our intervention is going to prevent it from happening. Bunk beds and ceiling fans are a different story. Posters in our offices and warnings and labels at the point of purchase of both fans and bunk beds makes some sense.
And while we are sticking labels on furniture, we should take a hard look at couches. Researchers have recently found that the accumulation of sedentary time in childhood can lead to early evidence of heart damage, which may portend heart disease in adulthood. Instead of those tags under the cushions, we need a big blaze orange sticker in prominent view that warns of the danger of becoming a couch potato.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
I was probably about 9 or 10 and I am assuming it was early winter when my mother took me aside and said in her usual quiet tone, “Willy, don’t ever stick your tongue on a metal pipe when it is cold outside.”
Putting my tongue on a frozen pipe was something that had never occurred to me even in my wildest preadolescent dreams. My mother’s caution only served to pique my interest and provide me with one more tempting scenario to consider.
Recently, a prank has gone viral on TikTok that shows an adult, usually the parent, cracking (not smashing) an egg on the child’s head and then emptying the egg contents into a bowl. Unlike the tongue-pipe disaster, it is hard to imagine how this stunt can be dangerous as long as the child is old enough to be walking around. But, at least one pediatrician has warned that there is a risk to the child from contracting salmonella.
There may be a few young children who are frightened by having an egg cracked on their head, but I can’t imagine that it would leave any lasting emotional scars. Given the minuscule theoretical risk of infection and the fact that the videos have accumulated more than 670 million views, this is another example of when we “experts” should keep a low profile and let the virus fade into Internet oblivion.
There is, however, a difference between harmless foolishness and stupidity, and one wonders when and in what manner we pediatricians should become involved. For example, in a recent study published in the journal Pediatrics, the investigators searched through a national emergency department database and found that
There were two peaks of distribution, one at less than 1 year of age and another at age 4. The older children were more often injured playing on furniture, most often bunk beds. The younger children were more likely to have been injured by being lifted or tossed in the air. No deaths were reported.
Is this a phenomenon that demands a response by pediatricians? Do we have time to ask every family if they have a ceiling fan? Should we be handing out brochures to every family? To whom should we target our message? This is a situation that seems to sort easily into two categories. One that involves stupidity and a second that is ignorance that may respond to education.
Tossing young children in the air is fun for the tosser and the child. I am sure there are a few children every year who slip out of the grasp of an adult and are injured. I have never seen a child brought in with this history. But it must happen. The result is likely to trigger a very tricky child protective investigation. But tossing a child underneath a ceiling fan is just plain stupid. I’m not sure our intervention is going to prevent it from happening. Bunk beds and ceiling fans are a different story. Posters in our offices and warnings and labels at the point of purchase of both fans and bunk beds makes some sense.
And while we are sticking labels on furniture, we should take a hard look at couches. Researchers have recently found that the accumulation of sedentary time in childhood can lead to early evidence of heart damage, which may portend heart disease in adulthood. Instead of those tags under the cushions, we need a big blaze orange sticker in prominent view that warns of the danger of becoming a couch potato.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Mammography breast density reporting: What it means for clinicians
This transcript has been edited for clarity.
Today, I’m going to talk about the 2023 Food and Drug Administration regulation that requires breast density to be reported on all mammogram results nationwide, and for that report to go to both clinicians and patients. Previously this was the rule in some states, but not in others. This is important because 40%-50% of women have dense breasts. I’m going to discuss what that means for you, and for our patients.
First
Breast density describes the appearance of the breast on mammography. Appearance varies on the basis of breast tissue composition, with fibroglandular tissue being more dense than fatty tissue. Breast density is important because it relates to both the risk for cancer and the ability of mammography to detect cancer.
Breast density is defined and classified according to the American College of Radiology’s BI-RADS four-category scale. Categories 1 and 2 refer to breast tissue that is not dense, accounting for about 50% of the population. Categories 3 and 4 describe heterogeneously dense and extremely dense breast tissue, which occur in approximately 40% and 50% of women, respectively. When speaking about dense breast tissue readings on mammography, we are referring to categories 3 and 4.
Women with dense breast tissue have an increased risk of developing breast cancer and are less likely to have early breast cancer detected on mammography.
Let’s go over the details by category:
For women in categories 1 and 2 (considered not dense breast tissue), the sensitivity of mammography for detecting early breast cancer is 80%-90%. In categories 3 and 4, the sensitivity of mammography drops to 60%-70%.
Compared with women with average breast density, the risk of developing breast cancer is 20% higher in women with BI-RADS category 3 breasts, and more than twice as high (relative risk, 2.1) in those with BI-RADS category 4 breasts. Thus, the risk of developing breast cancer is higher, but the sensitivity of the test is lower.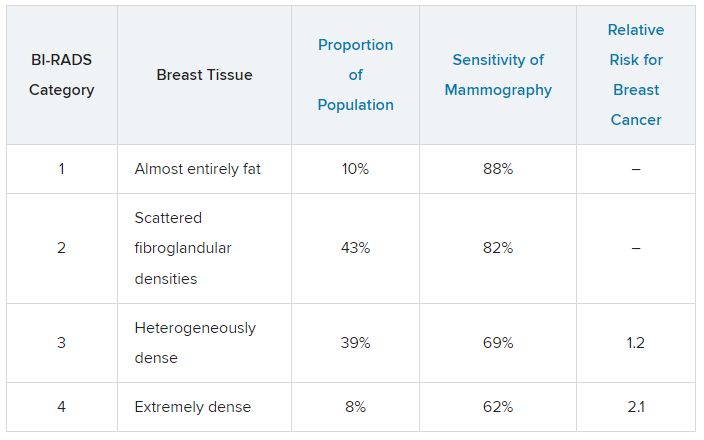
The clinical question is, what should we do about this? For women who have a normal mammogram with dense breasts, should follow-up testing be done, and if so, what test? The main follow-up testing options are either ultrasound or MRI, usually ultrasound. Additional testing will detect additional cancers that were not picked up on the initial mammogram and will also lead to additional biopsies for false-positive tests from the additional testing.
An American College of Gynecology and Obstetrics practice advisory nicely summarizes the evidence and clarifies that this decision is made in the context of a lack of published evidence demonstrating improved outcomes, specifically no reduction in breast cancer mortality, with supplemental testing. The official ACOG stance is that they “do not recommend routine use of alternative or adjunctive tests to screening mammography in women with dense breasts who are asymptomatic and have no additional risk factors.”
This is an area where it is important to understand the data. We are all going to be getting test results back that indicate level of breast density, and those test results will also be sent to our patients, so we are going to be asked about this by interested patients. Should this be something that we talk to patients about, utilizing shared decision-making to decide about whether follow-up testing is necessary in women with dense breasts? That is something each clinician will need to decide, and knowing the data is a critically important step in that decision.
Neil Skolnik, MD, is a professor, department of family medicine, at Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director, department of family medicine, Abington (Pennsylvania) Jefferson Health.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Today, I’m going to talk about the 2023 Food and Drug Administration regulation that requires breast density to be reported on all mammogram results nationwide, and for that report to go to both clinicians and patients. Previously this was the rule in some states, but not in others. This is important because 40%-50% of women have dense breasts. I’m going to discuss what that means for you, and for our patients.
First
Breast density describes the appearance of the breast on mammography. Appearance varies on the basis of breast tissue composition, with fibroglandular tissue being more dense than fatty tissue. Breast density is important because it relates to both the risk for cancer and the ability of mammography to detect cancer.
Breast density is defined and classified according to the American College of Radiology’s BI-RADS four-category scale. Categories 1 and 2 refer to breast tissue that is not dense, accounting for about 50% of the population. Categories 3 and 4 describe heterogeneously dense and extremely dense breast tissue, which occur in approximately 40% and 50% of women, respectively. When speaking about dense breast tissue readings on mammography, we are referring to categories 3 and 4.
Women with dense breast tissue have an increased risk of developing breast cancer and are less likely to have early breast cancer detected on mammography.
Let’s go over the details by category:
For women in categories 1 and 2 (considered not dense breast tissue), the sensitivity of mammography for detecting early breast cancer is 80%-90%. In categories 3 and 4, the sensitivity of mammography drops to 60%-70%.
Compared with women with average breast density, the risk of developing breast cancer is 20% higher in women with BI-RADS category 3 breasts, and more than twice as high (relative risk, 2.1) in those with BI-RADS category 4 breasts. Thus, the risk of developing breast cancer is higher, but the sensitivity of the test is lower.
The clinical question is, what should we do about this? For women who have a normal mammogram with dense breasts, should follow-up testing be done, and if so, what test? The main follow-up testing options are either ultrasound or MRI, usually ultrasound. Additional testing will detect additional cancers that were not picked up on the initial mammogram and will also lead to additional biopsies for false-positive tests from the additional testing.
An American College of Gynecology and Obstetrics practice advisory nicely summarizes the evidence and clarifies that this decision is made in the context of a lack of published evidence demonstrating improved outcomes, specifically no reduction in breast cancer mortality, with supplemental testing. The official ACOG stance is that they “do not recommend routine use of alternative or adjunctive tests to screening mammography in women with dense breasts who are asymptomatic and have no additional risk factors.”
This is an area where it is important to understand the data. We are all going to be getting test results back that indicate level of breast density, and those test results will also be sent to our patients, so we are going to be asked about this by interested patients. Should this be something that we talk to patients about, utilizing shared decision-making to decide about whether follow-up testing is necessary in women with dense breasts? That is something each clinician will need to decide, and knowing the data is a critically important step in that decision.
Neil Skolnik, MD, is a professor, department of family medicine, at Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director, department of family medicine, Abington (Pennsylvania) Jefferson Health.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Today, I’m going to talk about the 2023 Food and Drug Administration regulation that requires breast density to be reported on all mammogram results nationwide, and for that report to go to both clinicians and patients. Previously this was the rule in some states, but not in others. This is important because 40%-50% of women have dense breasts. I’m going to discuss what that means for you, and for our patients.
First
Breast density describes the appearance of the breast on mammography. Appearance varies on the basis of breast tissue composition, with fibroglandular tissue being more dense than fatty tissue. Breast density is important because it relates to both the risk for cancer and the ability of mammography to detect cancer.
Breast density is defined and classified according to the American College of Radiology’s BI-RADS four-category scale. Categories 1 and 2 refer to breast tissue that is not dense, accounting for about 50% of the population. Categories 3 and 4 describe heterogeneously dense and extremely dense breast tissue, which occur in approximately 40% and 50% of women, respectively. When speaking about dense breast tissue readings on mammography, we are referring to categories 3 and 4.
Women with dense breast tissue have an increased risk of developing breast cancer and are less likely to have early breast cancer detected on mammography.
Let’s go over the details by category:
For women in categories 1 and 2 (considered not dense breast tissue), the sensitivity of mammography for detecting early breast cancer is 80%-90%. In categories 3 and 4, the sensitivity of mammography drops to 60%-70%.
Compared with women with average breast density, the risk of developing breast cancer is 20% higher in women with BI-RADS category 3 breasts, and more than twice as high (relative risk, 2.1) in those with BI-RADS category 4 breasts. Thus, the risk of developing breast cancer is higher, but the sensitivity of the test is lower.
The clinical question is, what should we do about this? For women who have a normal mammogram with dense breasts, should follow-up testing be done, and if so, what test? The main follow-up testing options are either ultrasound or MRI, usually ultrasound. Additional testing will detect additional cancers that were not picked up on the initial mammogram and will also lead to additional biopsies for false-positive tests from the additional testing.
An American College of Gynecology and Obstetrics practice advisory nicely summarizes the evidence and clarifies that this decision is made in the context of a lack of published evidence demonstrating improved outcomes, specifically no reduction in breast cancer mortality, with supplemental testing. The official ACOG stance is that they “do not recommend routine use of alternative or adjunctive tests to screening mammography in women with dense breasts who are asymptomatic and have no additional risk factors.”
This is an area where it is important to understand the data. We are all going to be getting test results back that indicate level of breast density, and those test results will also be sent to our patients, so we are going to be asked about this by interested patients. Should this be something that we talk to patients about, utilizing shared decision-making to decide about whether follow-up testing is necessary in women with dense breasts? That is something each clinician will need to decide, and knowing the data is a critically important step in that decision.
Neil Skolnik, MD, is a professor, department of family medicine, at Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director, department of family medicine, Abington (Pennsylvania) Jefferson Health.
A version of this article first appeared on Medscape.com.
Introducing the ‘Ethics Corner’
In addition to reporting on scientific advances from our GI journals that can inform frontline clinical care of patients with gastrointestinal conditions, we have launched several new columns over the past year, including our monthly Member Spotlight and quarterly Health Policy and Advocacy columns, to diversify our content.
In our September 2023 issue, in addition to debuting a new cover design, we are pleased to introduce yet another new offering – the “Ethics Corner” column. It is intended to highlight important ethical considerations and challenges arising in GI practice and offer practical guidance on how to navigate them. In our inaugural Ethics Corner, AGA Ethics Committee members Dr. Sheldon Sloan and Dr. David Drew discuss the “good, the bad, and the ugly” of direct-to-consumer advertising (DTCA). They highlight the pros and cons of DTCA from an ethical perspective, illustrate how DTCA can impact everyday clinical interactions with patients, and provide insight into how to navigate these challenging conversations. We hope you enjoy this new addition to the newspaper and welcome your ideas for future Ethics Corner columns and other content.
Also in this month’s issue, we update you on AGA’s response to the American College of Physicians’ recent decision to recommend against initiating colon cancer screening at age 45, contrary to the recommendation of the GI community. We also present a story on Humira biosimilars and how they are likely to impact clinical practice. Finally, our September Member Spotlight features GI dietitian Renee Euler, MS, RD, LD, who discusses the intersection between diet and GI disorders, the importance of a team approach to GI care, and her work as a liaison between the AGA and Academy of Nutrition and Dietetics.
We hope you enjoy these, and all the stories featured in our September issue.
Megan A. Adams, MD, JD, MSc
In addition to reporting on scientific advances from our GI journals that can inform frontline clinical care of patients with gastrointestinal conditions, we have launched several new columns over the past year, including our monthly Member Spotlight and quarterly Health Policy and Advocacy columns, to diversify our content.
In our September 2023 issue, in addition to debuting a new cover design, we are pleased to introduce yet another new offering – the “Ethics Corner” column. It is intended to highlight important ethical considerations and challenges arising in GI practice and offer practical guidance on how to navigate them. In our inaugural Ethics Corner, AGA Ethics Committee members Dr. Sheldon Sloan and Dr. David Drew discuss the “good, the bad, and the ugly” of direct-to-consumer advertising (DTCA). They highlight the pros and cons of DTCA from an ethical perspective, illustrate how DTCA can impact everyday clinical interactions with patients, and provide insight into how to navigate these challenging conversations. We hope you enjoy this new addition to the newspaper and welcome your ideas for future Ethics Corner columns and other content.
Also in this month’s issue, we update you on AGA’s response to the American College of Physicians’ recent decision to recommend against initiating colon cancer screening at age 45, contrary to the recommendation of the GI community. We also present a story on Humira biosimilars and how they are likely to impact clinical practice. Finally, our September Member Spotlight features GI dietitian Renee Euler, MS, RD, LD, who discusses the intersection between diet and GI disorders, the importance of a team approach to GI care, and her work as a liaison between the AGA and Academy of Nutrition and Dietetics.
We hope you enjoy these, and all the stories featured in our September issue.
Megan A. Adams, MD, JD, MSc
In addition to reporting on scientific advances from our GI journals that can inform frontline clinical care of patients with gastrointestinal conditions, we have launched several new columns over the past year, including our monthly Member Spotlight and quarterly Health Policy and Advocacy columns, to diversify our content.
In our September 2023 issue, in addition to debuting a new cover design, we are pleased to introduce yet another new offering – the “Ethics Corner” column. It is intended to highlight important ethical considerations and challenges arising in GI practice and offer practical guidance on how to navigate them. In our inaugural Ethics Corner, AGA Ethics Committee members Dr. Sheldon Sloan and Dr. David Drew discuss the “good, the bad, and the ugly” of direct-to-consumer advertising (DTCA). They highlight the pros and cons of DTCA from an ethical perspective, illustrate how DTCA can impact everyday clinical interactions with patients, and provide insight into how to navigate these challenging conversations. We hope you enjoy this new addition to the newspaper and welcome your ideas for future Ethics Corner columns and other content.
Also in this month’s issue, we update you on AGA’s response to the American College of Physicians’ recent decision to recommend against initiating colon cancer screening at age 45, contrary to the recommendation of the GI community. We also present a story on Humira biosimilars and how they are likely to impact clinical practice. Finally, our September Member Spotlight features GI dietitian Renee Euler, MS, RD, LD, who discusses the intersection between diet and GI disorders, the importance of a team approach to GI care, and her work as a liaison between the AGA and Academy of Nutrition and Dietetics.
We hope you enjoy these, and all the stories featured in our September issue.
Megan A. Adams, MD, JD, MSc
Coping with burnout and repetitive injuries
Dear colleagues,
We are all part of one of the most exciting and varied fields of medicine and hope to have long and productive careers. In this month’s AGA Perspectives we explore two different impediments to longevity as a gastroenterologist: work-related disability and burnout.
Physician burnout has reached almost epidemic levels in medicine and is best approached in a multimodal manner, incorporating both institutional and individual changes. Dr. Sumeet Tewani discusses ways in which groups and institutions can foster physician wellness to reduce burnout. In particular, he will explore how flexibility in work schedules, among other initiatives, can improve workplace morale. In an accompanying perspective, Dr. Anna Lipowska and Dr. Amandeep Shergill explore how to incorporate ergonomics in endoscopy to prevent injury. Endoscopic practice, with its repetitive tasks and physical demands, can predispose to injury at all levels of training and experience. Ergonomics is thus a critical topic that is unfortunately covered too little, if at all, in our endoscopy training.
We hope these essays will help your medical practice and welcome your thoughts on these important issues at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Conn., and chief of endoscopy at West Haven (Conn.) VA Medical Center. He is an associate editor for GI & Hepatology News.
Fostering physician wellness to prevent burnout
BY SUMEET TEWANI, MD
Gastroenterology can be a challenging field, both professionally and personally, as it requires providers to have high clinical knowledge, expertise, and emotional intelligence. Burnout is a state of emotional, physical, and mental exhaustion, depersonalization, and a reduced sense of personal accomplishment, caused by prolonged or excessive stress. Burnout can be a serious and progressive chronic condition that negatively impacts the provider and patient experience, with serious consequences on the provider’s health, job performance, patient satisfaction, and personal relationships.
Adopting strategies to combat burnout are of vital importance to promoting provider wellness, a healthy work environment, and positive interactions with colleagues, staff, and patients. This also positively impacts providers’ out-of-work experiences and relationships. on an institutional level and on an individual level.
Multiple factors affecting burnout have been identified, including individual factors, work volume, professional risk and responsibility, resources, and relationships with colleagues and patients. Practicing gastroenterology frequently requires long and irregular work hours, heavy workloads, large panels of complex patients, invasive procedures, and high amounts of stress. Additional stressors may include an inefficient work environment, inadequate support, and loss of value and meaning in work. Nearly 50% of physicians meet criteria for burnout, citing such reasons as excessive bureaucratic tasks, lack of control, flexibility and autonomy, lack of peer respect, increasing computerization of practice, and lack of respect from patients.
Preventing burnout
When coping with burnout, many providers choose positive mechanisms such as exercise, listening to music, meditation, and talking with family and friends. Others become more isolated, eat junk food or binge eat, or turn to drug or alcohol abuse. Our primary approach to preventing burnout at an individual level is to ensure providers have access to self-care techniques such as stress management and mindfulness, and to encourage wellness with regular exercise and healthy habits. Promoting a culture of work-life balance requires providing adequate time for personal activities and hobbies, rest, relaxation, and spending time with family and friends. Allowing providers to personally shape their career paths aligns their personal and professional goals, leading to greater satisfaction. To this end, providers may become involved in clinical research, medical education, and clinical and administrative committees through the practice, local medical school and hospitals, and local and national societies. We provide ample vacation time and CME opportunities for our providers. Vacation time is flexible and can be taken in half-day or full-day increments, or on an hourly basis for personal time as necessary. This allows for enhanced flexibility with scheduling time off from work.
On an institutional level, leadership plays a prime role in creating a healthy work environment. Having good leaders influences the well-being and satisfaction of everyone within the organization. Leaders can have a positive impact by aligning values and work culture, using incentives in a productive manner, and promoting strategies to reduce burnout. Involving physician partners in leadership on a rotating basis allows them to better understand the roles of the leaders in the organization and empowers them to have a voice in changing policies to reduce administrative burdens and foster wellness.
We promote the concept of working together as a team for the success of the practice. All partners have an equal say in the management of the group. We eliminated the stress of competition within the group, equalizing pay across all physician partners, while maintaining equal exposure to work and equal time off from work. This levels the playing field between physicians who have varied interests and expertise, so that everyone is working towards the success of the practice and not individual compensation. To that end, our providers do not have individual offices, but work out of a “bullpen” with an open concept where we have individual workspaces and interact with each other continuously throughout the day. This promotes cohesion and teamwork between the providers for all our patients and promotes professional relationships and peer support. Efforts to promote workplace morale include access to a fully stocked deli and a newly installed espresso machine.
The concept of teamwork
The concept of teamwork also needs to pervade through the entire organization. To manage the demands of a busy workday, we have directly trained advanced practice nurses in the clinic and inpatient settings, allowing our physicians to increase throughput and procedures while maintaining a high level of patient care, satisfaction, and efficiency. Providers report excessive administrative tasks and frustration with the electronic health record as major factors contributing to burnout. Delegating tasks to staff, commensurate with their training and scope of practice, alleviates some of this burden. Each of the providers in our practice has a triage nurse who functions in a key capacity to ensure the appropriate clinical and administrative tasks are complete. Medical scribes, medical assistants, nurses, and physician assistants can be utilized for data entry and other tasks. We have developed templates within the electronic health record that can be standardized across the practice. Promoting teamwork with staff also means respecting the staff and understanding their needs. A highly functioning health care team can provide comprehensive care proactively and efficiently, with improved professional satisfaction.
In summary, I identify several ways to promote physician wellness. Every GI practice should strive to implement local approaches to prevent physician burnout and help maintain a happy and productive workforce.
Dr. Tewani is a gastroenterologist with Rockford Gastroenterology Associates in Rockford, Ill. He has no relevant disclosures.
References
1. Koval ML. Medscape gastroenterologist lifestyle, happiness & burnout report 2023: Contentment amid stress. Medscape. 24 Feb 2023.
2. Ong J et al. The prevalence of burnout, risk factors, and job-related stressors in gastroenterologists: A systematic review. J Gastroenterol Hepatol. 2021 Sep;36(9):2338-48.
3. Anderson J et al. Strategies to combat physician burnout in gastroenterology. Am J Gastroenterol. 2017 Sep;112(9):1356-9.
4. Keswani R et al. Burnout in gastroenterologists and how to prevent it. Gastroenterology. 2014 Jul;147(1):11-4.
The hazards of endoscopy: Ergonomics guide the way
BY ANNA LIPOWSKA, MD, AND AMANDEEP SHERGILL, MD, MS
Preventing disability and promoting a long and successful endoscopic career involves proactive measures to support well-being, and ergonomics plays a key role. Ergonomics is the science of fitting a job to the worker, with a primary goal of working smarter and safer. When hazards are identified, mitigation measures, guided by a hierarchy of controls, must be implemented that improve the fit of the tool, task and job to the worker in order to reduce the risk of endoscopy-related injury (ERI). As more women enter the field and as the overall GI physician population ages, ensuring that endoscopy is designed to be safely performed within the capacity of a diverse group of workers will be critical to creating an inclusive and equitable work environment.
Ergonomic education is foundational: Awareness of ERI risk factors allows endoscopists to identify hazards and advocate for effective control solutions. Ergonomic education materials are available through all of the major GI societies. A road map for implementing an endoscopy ergonomics program has been previously published and provides guidance on risk assessment and mitigation measures.
Respect pain
Overuse injuries occur when the physical demands of a job are greater than tissue tolerances, leading to cumulative microtrauma. The first sign of microtraumatic injury is discomfort and pain. Studies have demonstrated that an estimated three-quarters of gastroenterologists experience ERI, with 20% requiring time off work and 12% requiring surgery. Gastroenterology trainees are also at risk, with 20% of surveyed fellows endorsing overuse injury, some even requiring work-related leaves of absence. In the early stages of ERI, aching and tiredness occur during the work shift only. In the intermediate stage, aching and tiredness occur early in the work shift and persist at night, and may be associated with a reduced capacity for repetitive work. In the late stages, aching, fatigue and weakness persist at rest and may be associated with inability to sleep and to perform light duties. Pain is an important signal indicating mitigation measures are required to control exposures.
Utilize the hierarchy of controls
The responsibility for adoption of ergonomically friendly practices does not lie solely with the physician; both institutional and industry-level support are key to its success. The hierarchy of controls defines which actions will best mitigate exposures to hazards in the workplace, highlighting that modifications to personal practice have the smallest impact. Current endoscope design does not accommodate the full range of hand strengths and sizes and contributes to ERI.
Advancements at the industry level by eliminating or substituting hazards, or designing engineering controls to reduce exposure, will be most effective at preventing distal upper extremity ERI. The next most effective controls are at the institutional level, with endoscopy units ensuring access to engineering controls and implementing effective administrative controls. For example, institutional support and investment in adjustable workstations is imperative to accommodate a range of anthropometric dimensions of the population. Support for ergonomic education, scheduling changes and a culture where safety is a priority can help reduce exposure to hazards and injury risk.
Adjust the endoscopy suite to achieve a comfortable position before every procedure
Neutral body posture is our position of greatest comfort and maximum strength, and any deviation from neutral posture decreases the amount of force the muscles can produce and causes the muscles to fatigue sooner. The most important factor affecting the endoscopists’ overall posture is the monitor position and height. Monitors must be adjustable. Place the monitor directly in front of you, with the center of the screen 15-25 degrees below eye height for a neutral neck position and resting eye position. Procedure bed height should be adjusted 0-10 cm below elbow height to allow for neutral elbow postures and relaxed shoulders. Antifatigue mats and shoes with supportive insoles can reduce fatigue. Two-piece lead aprons distribute a portion of the static load to the hips and decrease back strain. Incorporate a preprocedure ergonomic time-out, to assess proper room set up, body mechanics, equipment and team preparedness.
Give yourself a break
Breaks should be built into the endoscopy schedule, especially for a full day of endoscopy. At a minimum, incorporate microbreaks during procedures, which have been found to alleviate pain and improve performance. Exercises and stretching can be incorporated between cases, including routines designed specifically for endoscopists.
Getting older isn’t for the weak
Currently, 50% of our gastroenterologists are over 55 years old. The aging process leads to a distinct muscle mass and strength loss. Women are already at a disadvantage because, on average, they have less muscle mass than men in all age groups. Muscle starts to deteriorate when we reach our 30s, and after age 40, we lose on average 8% of our muscle mass every decade which accelerates at an even faster rate after age 60. Both resistance and aerobic exercise can be very useful to counteract sarcopenia and maintain strength. Given the physical demands of endoscopy, exercise can help safeguard career longevity and maintain overall wellness. Multiple resources are available to tailor a program that fits your time, budget and needs.
Optimize all of your workstations
Prolonged computer use and desk work is also a significant part of a gastroenterologist’s profession. If using a sitting desk, chair height should allow for 90-degree flexion at the hips and knees and for feet to rest flatly on the floor. The chair should also provide adequate back support for a relaxed upright position. Similar to endoscopy, place the monitor directly in front with the center of the screen slightly below eye level. For mouse and keyboard placement, aim to have the elbows at or slightly below 90 degrees and one’s wrists and fingers in neutral position.
Endoscopy can be hazardous to the endoscopists’ health. Incorporating ergonomic principles creates a safer and more efficient work environment. At the individual level, ergonomically optimized postures during endoscopy as well as during computer-related tasks, room set up, inclusion of microbreaks, and protective exercises can help decrease the risk of repetitive strain injury and prevent disability. Importantly, change at the industry and institutional level has the greatest potential for positive impact. Adoption of ergonomic practices promotes career longevity and ensures that gastroenterologists can continue successful and long careers and provide quality care to their patients without compromising their own health.
Dr. Lipowska is an assistant professor in the division of gastroenterology and hepatology, University of Illinois at Chicago. She disclosed no conflicts. Dr. Shergill is chief of gastroenterology for the San Francisco VA Health Care System. She disclosed consulting work for Boston Scientific and Neptune Medical, honoraria for visiting professorship with Intuitive Surgical, and a research gift from Pentax.
References
Shergill AK. Top tips for implementing an endoscopy ergonomics program. Gastrointest Endosc. 2023 Feb;97(2):361-4.
Pawa S et al. Are all endoscopy-related musculoskeletal injuries created equal? Results of a national gender-based survey. Am J Gastroenterol. 2021;116(3):530-8.
Austin K et al. Musculoskeletal injuries are commonly reported among gastroenterology trainees: Results of a national survey. Dig Dis Sci. 2019;64(6):1439-47.
Lipowska A et al. Ergonomics in the unit: Modeling the environment around the endoscopist. Tech Innov Gastrointest Endosc. 2021;23(3):256-62.
Park A et al. Intraoperative “Micro Breaks” with a targeted stretching enhance surgeon physical function and mental focus: A multicenter cohort study. Ann Surg. 2017;265(2):340-6.
Shergill A et al. Ergonomic endoscopy: An oxymoron or realistic goal? Gastrointest Endosc. 2019;90(6):966-70.
Dear colleagues,
We are all part of one of the most exciting and varied fields of medicine and hope to have long and productive careers. In this month’s AGA Perspectives we explore two different impediments to longevity as a gastroenterologist: work-related disability and burnout.
Physician burnout has reached almost epidemic levels in medicine and is best approached in a multimodal manner, incorporating both institutional and individual changes. Dr. Sumeet Tewani discusses ways in which groups and institutions can foster physician wellness to reduce burnout. In particular, he will explore how flexibility in work schedules, among other initiatives, can improve workplace morale. In an accompanying perspective, Dr. Anna Lipowska and Dr. Amandeep Shergill explore how to incorporate ergonomics in endoscopy to prevent injury. Endoscopic practice, with its repetitive tasks and physical demands, can predispose to injury at all levels of training and experience. Ergonomics is thus a critical topic that is unfortunately covered too little, if at all, in our endoscopy training.
We hope these essays will help your medical practice and welcome your thoughts on these important issues at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Conn., and chief of endoscopy at West Haven (Conn.) VA Medical Center. He is an associate editor for GI & Hepatology News.
Fostering physician wellness to prevent burnout
BY SUMEET TEWANI, MD
Gastroenterology can be a challenging field, both professionally and personally, as it requires providers to have high clinical knowledge, expertise, and emotional intelligence. Burnout is a state of emotional, physical, and mental exhaustion, depersonalization, and a reduced sense of personal accomplishment, caused by prolonged or excessive stress. Burnout can be a serious and progressive chronic condition that negatively impacts the provider and patient experience, with serious consequences on the provider’s health, job performance, patient satisfaction, and personal relationships.
Adopting strategies to combat burnout are of vital importance to promoting provider wellness, a healthy work environment, and positive interactions with colleagues, staff, and patients. This also positively impacts providers’ out-of-work experiences and relationships. on an institutional level and on an individual level.
Multiple factors affecting burnout have been identified, including individual factors, work volume, professional risk and responsibility, resources, and relationships with colleagues and patients. Practicing gastroenterology frequently requires long and irregular work hours, heavy workloads, large panels of complex patients, invasive procedures, and high amounts of stress. Additional stressors may include an inefficient work environment, inadequate support, and loss of value and meaning in work. Nearly 50% of physicians meet criteria for burnout, citing such reasons as excessive bureaucratic tasks, lack of control, flexibility and autonomy, lack of peer respect, increasing computerization of practice, and lack of respect from patients.
Preventing burnout
When coping with burnout, many providers choose positive mechanisms such as exercise, listening to music, meditation, and talking with family and friends. Others become more isolated, eat junk food or binge eat, or turn to drug or alcohol abuse. Our primary approach to preventing burnout at an individual level is to ensure providers have access to self-care techniques such as stress management and mindfulness, and to encourage wellness with regular exercise and healthy habits. Promoting a culture of work-life balance requires providing adequate time for personal activities and hobbies, rest, relaxation, and spending time with family and friends. Allowing providers to personally shape their career paths aligns their personal and professional goals, leading to greater satisfaction. To this end, providers may become involved in clinical research, medical education, and clinical and administrative committees through the practice, local medical school and hospitals, and local and national societies. We provide ample vacation time and CME opportunities for our providers. Vacation time is flexible and can be taken in half-day or full-day increments, or on an hourly basis for personal time as necessary. This allows for enhanced flexibility with scheduling time off from work.
On an institutional level, leadership plays a prime role in creating a healthy work environment. Having good leaders influences the well-being and satisfaction of everyone within the organization. Leaders can have a positive impact by aligning values and work culture, using incentives in a productive manner, and promoting strategies to reduce burnout. Involving physician partners in leadership on a rotating basis allows them to better understand the roles of the leaders in the organization and empowers them to have a voice in changing policies to reduce administrative burdens and foster wellness.
We promote the concept of working together as a team for the success of the practice. All partners have an equal say in the management of the group. We eliminated the stress of competition within the group, equalizing pay across all physician partners, while maintaining equal exposure to work and equal time off from work. This levels the playing field between physicians who have varied interests and expertise, so that everyone is working towards the success of the practice and not individual compensation. To that end, our providers do not have individual offices, but work out of a “bullpen” with an open concept where we have individual workspaces and interact with each other continuously throughout the day. This promotes cohesion and teamwork between the providers for all our patients and promotes professional relationships and peer support. Efforts to promote workplace morale include access to a fully stocked deli and a newly installed espresso machine.
The concept of teamwork
The concept of teamwork also needs to pervade through the entire organization. To manage the demands of a busy workday, we have directly trained advanced practice nurses in the clinic and inpatient settings, allowing our physicians to increase throughput and procedures while maintaining a high level of patient care, satisfaction, and efficiency. Providers report excessive administrative tasks and frustration with the electronic health record as major factors contributing to burnout. Delegating tasks to staff, commensurate with their training and scope of practice, alleviates some of this burden. Each of the providers in our practice has a triage nurse who functions in a key capacity to ensure the appropriate clinical and administrative tasks are complete. Medical scribes, medical assistants, nurses, and physician assistants can be utilized for data entry and other tasks. We have developed templates within the electronic health record that can be standardized across the practice. Promoting teamwork with staff also means respecting the staff and understanding their needs. A highly functioning health care team can provide comprehensive care proactively and efficiently, with improved professional satisfaction.
In summary, I identify several ways to promote physician wellness. Every GI practice should strive to implement local approaches to prevent physician burnout and help maintain a happy and productive workforce.
Dr. Tewani is a gastroenterologist with Rockford Gastroenterology Associates in Rockford, Ill. He has no relevant disclosures.
References
1. Koval ML. Medscape gastroenterologist lifestyle, happiness & burnout report 2023: Contentment amid stress. Medscape. 24 Feb 2023.
2. Ong J et al. The prevalence of burnout, risk factors, and job-related stressors in gastroenterologists: A systematic review. J Gastroenterol Hepatol. 2021 Sep;36(9):2338-48.
3. Anderson J et al. Strategies to combat physician burnout in gastroenterology. Am J Gastroenterol. 2017 Sep;112(9):1356-9.
4. Keswani R et al. Burnout in gastroenterologists and how to prevent it. Gastroenterology. 2014 Jul;147(1):11-4.
The hazards of endoscopy: Ergonomics guide the way
BY ANNA LIPOWSKA, MD, AND AMANDEEP SHERGILL, MD, MS
Preventing disability and promoting a long and successful endoscopic career involves proactive measures to support well-being, and ergonomics plays a key role. Ergonomics is the science of fitting a job to the worker, with a primary goal of working smarter and safer. When hazards are identified, mitigation measures, guided by a hierarchy of controls, must be implemented that improve the fit of the tool, task and job to the worker in order to reduce the risk of endoscopy-related injury (ERI). As more women enter the field and as the overall GI physician population ages, ensuring that endoscopy is designed to be safely performed within the capacity of a diverse group of workers will be critical to creating an inclusive and equitable work environment.
Ergonomic education is foundational: Awareness of ERI risk factors allows endoscopists to identify hazards and advocate for effective control solutions. Ergonomic education materials are available through all of the major GI societies. A road map for implementing an endoscopy ergonomics program has been previously published and provides guidance on risk assessment and mitigation measures.
Respect pain
Overuse injuries occur when the physical demands of a job are greater than tissue tolerances, leading to cumulative microtrauma. The first sign of microtraumatic injury is discomfort and pain. Studies have demonstrated that an estimated three-quarters of gastroenterologists experience ERI, with 20% requiring time off work and 12% requiring surgery. Gastroenterology trainees are also at risk, with 20% of surveyed fellows endorsing overuse injury, some even requiring work-related leaves of absence. In the early stages of ERI, aching and tiredness occur during the work shift only. In the intermediate stage, aching and tiredness occur early in the work shift and persist at night, and may be associated with a reduced capacity for repetitive work. In the late stages, aching, fatigue and weakness persist at rest and may be associated with inability to sleep and to perform light duties. Pain is an important signal indicating mitigation measures are required to control exposures.
Utilize the hierarchy of controls
The responsibility for adoption of ergonomically friendly practices does not lie solely with the physician; both institutional and industry-level support are key to its success. The hierarchy of controls defines which actions will best mitigate exposures to hazards in the workplace, highlighting that modifications to personal practice have the smallest impact. Current endoscope design does not accommodate the full range of hand strengths and sizes and contributes to ERI.
Advancements at the industry level by eliminating or substituting hazards, or designing engineering controls to reduce exposure, will be most effective at preventing distal upper extremity ERI. The next most effective controls are at the institutional level, with endoscopy units ensuring access to engineering controls and implementing effective administrative controls. For example, institutional support and investment in adjustable workstations is imperative to accommodate a range of anthropometric dimensions of the population. Support for ergonomic education, scheduling changes and a culture where safety is a priority can help reduce exposure to hazards and injury risk.
Adjust the endoscopy suite to achieve a comfortable position before every procedure
Neutral body posture is our position of greatest comfort and maximum strength, and any deviation from neutral posture decreases the amount of force the muscles can produce and causes the muscles to fatigue sooner. The most important factor affecting the endoscopists’ overall posture is the monitor position and height. Monitors must be adjustable. Place the monitor directly in front of you, with the center of the screen 15-25 degrees below eye height for a neutral neck position and resting eye position. Procedure bed height should be adjusted 0-10 cm below elbow height to allow for neutral elbow postures and relaxed shoulders. Antifatigue mats and shoes with supportive insoles can reduce fatigue. Two-piece lead aprons distribute a portion of the static load to the hips and decrease back strain. Incorporate a preprocedure ergonomic time-out, to assess proper room set up, body mechanics, equipment and team preparedness.
Give yourself a break
Breaks should be built into the endoscopy schedule, especially for a full day of endoscopy. At a minimum, incorporate microbreaks during procedures, which have been found to alleviate pain and improve performance. Exercises and stretching can be incorporated between cases, including routines designed specifically for endoscopists.
Getting older isn’t for the weak
Currently, 50% of our gastroenterologists are over 55 years old. The aging process leads to a distinct muscle mass and strength loss. Women are already at a disadvantage because, on average, they have less muscle mass than men in all age groups. Muscle starts to deteriorate when we reach our 30s, and after age 40, we lose on average 8% of our muscle mass every decade which accelerates at an even faster rate after age 60. Both resistance and aerobic exercise can be very useful to counteract sarcopenia and maintain strength. Given the physical demands of endoscopy, exercise can help safeguard career longevity and maintain overall wellness. Multiple resources are available to tailor a program that fits your time, budget and needs.
Optimize all of your workstations
Prolonged computer use and desk work is also a significant part of a gastroenterologist’s profession. If using a sitting desk, chair height should allow for 90-degree flexion at the hips and knees and for feet to rest flatly on the floor. The chair should also provide adequate back support for a relaxed upright position. Similar to endoscopy, place the monitor directly in front with the center of the screen slightly below eye level. For mouse and keyboard placement, aim to have the elbows at or slightly below 90 degrees and one’s wrists and fingers in neutral position.
Endoscopy can be hazardous to the endoscopists’ health. Incorporating ergonomic principles creates a safer and more efficient work environment. At the individual level, ergonomically optimized postures during endoscopy as well as during computer-related tasks, room set up, inclusion of microbreaks, and protective exercises can help decrease the risk of repetitive strain injury and prevent disability. Importantly, change at the industry and institutional level has the greatest potential for positive impact. Adoption of ergonomic practices promotes career longevity and ensures that gastroenterologists can continue successful and long careers and provide quality care to their patients without compromising their own health.
Dr. Lipowska is an assistant professor in the division of gastroenterology and hepatology, University of Illinois at Chicago. She disclosed no conflicts. Dr. Shergill is chief of gastroenterology for the San Francisco VA Health Care System. She disclosed consulting work for Boston Scientific and Neptune Medical, honoraria for visiting professorship with Intuitive Surgical, and a research gift from Pentax.
References
Shergill AK. Top tips for implementing an endoscopy ergonomics program. Gastrointest Endosc. 2023 Feb;97(2):361-4.
Pawa S et al. Are all endoscopy-related musculoskeletal injuries created equal? Results of a national gender-based survey. Am J Gastroenterol. 2021;116(3):530-8.
Austin K et al. Musculoskeletal injuries are commonly reported among gastroenterology trainees: Results of a national survey. Dig Dis Sci. 2019;64(6):1439-47.
Lipowska A et al. Ergonomics in the unit: Modeling the environment around the endoscopist. Tech Innov Gastrointest Endosc. 2021;23(3):256-62.
Park A et al. Intraoperative “Micro Breaks” with a targeted stretching enhance surgeon physical function and mental focus: A multicenter cohort study. Ann Surg. 2017;265(2):340-6.
Shergill A et al. Ergonomic endoscopy: An oxymoron or realistic goal? Gastrointest Endosc. 2019;90(6):966-70.
Dear colleagues,
We are all part of one of the most exciting and varied fields of medicine and hope to have long and productive careers. In this month’s AGA Perspectives we explore two different impediments to longevity as a gastroenterologist: work-related disability and burnout.
Physician burnout has reached almost epidemic levels in medicine and is best approached in a multimodal manner, incorporating both institutional and individual changes. Dr. Sumeet Tewani discusses ways in which groups and institutions can foster physician wellness to reduce burnout. In particular, he will explore how flexibility in work schedules, among other initiatives, can improve workplace morale. In an accompanying perspective, Dr. Anna Lipowska and Dr. Amandeep Shergill explore how to incorporate ergonomics in endoscopy to prevent injury. Endoscopic practice, with its repetitive tasks and physical demands, can predispose to injury at all levels of training and experience. Ergonomics is thus a critical topic that is unfortunately covered too little, if at all, in our endoscopy training.
We hope these essays will help your medical practice and welcome your thoughts on these important issues at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Conn., and chief of endoscopy at West Haven (Conn.) VA Medical Center. He is an associate editor for GI & Hepatology News.
Fostering physician wellness to prevent burnout
BY SUMEET TEWANI, MD
Gastroenterology can be a challenging field, both professionally and personally, as it requires providers to have high clinical knowledge, expertise, and emotional intelligence. Burnout is a state of emotional, physical, and mental exhaustion, depersonalization, and a reduced sense of personal accomplishment, caused by prolonged or excessive stress. Burnout can be a serious and progressive chronic condition that negatively impacts the provider and patient experience, with serious consequences on the provider’s health, job performance, patient satisfaction, and personal relationships.
Adopting strategies to combat burnout are of vital importance to promoting provider wellness, a healthy work environment, and positive interactions with colleagues, staff, and patients. This also positively impacts providers’ out-of-work experiences and relationships. on an institutional level and on an individual level.
Multiple factors affecting burnout have been identified, including individual factors, work volume, professional risk and responsibility, resources, and relationships with colleagues and patients. Practicing gastroenterology frequently requires long and irregular work hours, heavy workloads, large panels of complex patients, invasive procedures, and high amounts of stress. Additional stressors may include an inefficient work environment, inadequate support, and loss of value and meaning in work. Nearly 50% of physicians meet criteria for burnout, citing such reasons as excessive bureaucratic tasks, lack of control, flexibility and autonomy, lack of peer respect, increasing computerization of practice, and lack of respect from patients.
Preventing burnout
When coping with burnout, many providers choose positive mechanisms such as exercise, listening to music, meditation, and talking with family and friends. Others become more isolated, eat junk food or binge eat, or turn to drug or alcohol abuse. Our primary approach to preventing burnout at an individual level is to ensure providers have access to self-care techniques such as stress management and mindfulness, and to encourage wellness with regular exercise and healthy habits. Promoting a culture of work-life balance requires providing adequate time for personal activities and hobbies, rest, relaxation, and spending time with family and friends. Allowing providers to personally shape their career paths aligns their personal and professional goals, leading to greater satisfaction. To this end, providers may become involved in clinical research, medical education, and clinical and administrative committees through the practice, local medical school and hospitals, and local and national societies. We provide ample vacation time and CME opportunities for our providers. Vacation time is flexible and can be taken in half-day or full-day increments, or on an hourly basis for personal time as necessary. This allows for enhanced flexibility with scheduling time off from work.
On an institutional level, leadership plays a prime role in creating a healthy work environment. Having good leaders influences the well-being and satisfaction of everyone within the organization. Leaders can have a positive impact by aligning values and work culture, using incentives in a productive manner, and promoting strategies to reduce burnout. Involving physician partners in leadership on a rotating basis allows them to better understand the roles of the leaders in the organization and empowers them to have a voice in changing policies to reduce administrative burdens and foster wellness.
We promote the concept of working together as a team for the success of the practice. All partners have an equal say in the management of the group. We eliminated the stress of competition within the group, equalizing pay across all physician partners, while maintaining equal exposure to work and equal time off from work. This levels the playing field between physicians who have varied interests and expertise, so that everyone is working towards the success of the practice and not individual compensation. To that end, our providers do not have individual offices, but work out of a “bullpen” with an open concept where we have individual workspaces and interact with each other continuously throughout the day. This promotes cohesion and teamwork between the providers for all our patients and promotes professional relationships and peer support. Efforts to promote workplace morale include access to a fully stocked deli and a newly installed espresso machine.
The concept of teamwork
The concept of teamwork also needs to pervade through the entire organization. To manage the demands of a busy workday, we have directly trained advanced practice nurses in the clinic and inpatient settings, allowing our physicians to increase throughput and procedures while maintaining a high level of patient care, satisfaction, and efficiency. Providers report excessive administrative tasks and frustration with the electronic health record as major factors contributing to burnout. Delegating tasks to staff, commensurate with their training and scope of practice, alleviates some of this burden. Each of the providers in our practice has a triage nurse who functions in a key capacity to ensure the appropriate clinical and administrative tasks are complete. Medical scribes, medical assistants, nurses, and physician assistants can be utilized for data entry and other tasks. We have developed templates within the electronic health record that can be standardized across the practice. Promoting teamwork with staff also means respecting the staff and understanding their needs. A highly functioning health care team can provide comprehensive care proactively and efficiently, with improved professional satisfaction.
In summary, I identify several ways to promote physician wellness. Every GI practice should strive to implement local approaches to prevent physician burnout and help maintain a happy and productive workforce.
Dr. Tewani is a gastroenterologist with Rockford Gastroenterology Associates in Rockford, Ill. He has no relevant disclosures.
References
1. Koval ML. Medscape gastroenterologist lifestyle, happiness & burnout report 2023: Contentment amid stress. Medscape. 24 Feb 2023.
2. Ong J et al. The prevalence of burnout, risk factors, and job-related stressors in gastroenterologists: A systematic review. J Gastroenterol Hepatol. 2021 Sep;36(9):2338-48.
3. Anderson J et al. Strategies to combat physician burnout in gastroenterology. Am J Gastroenterol. 2017 Sep;112(9):1356-9.
4. Keswani R et al. Burnout in gastroenterologists and how to prevent it. Gastroenterology. 2014 Jul;147(1):11-4.
The hazards of endoscopy: Ergonomics guide the way
BY ANNA LIPOWSKA, MD, AND AMANDEEP SHERGILL, MD, MS
Preventing disability and promoting a long and successful endoscopic career involves proactive measures to support well-being, and ergonomics plays a key role. Ergonomics is the science of fitting a job to the worker, with a primary goal of working smarter and safer. When hazards are identified, mitigation measures, guided by a hierarchy of controls, must be implemented that improve the fit of the tool, task and job to the worker in order to reduce the risk of endoscopy-related injury (ERI). As more women enter the field and as the overall GI physician population ages, ensuring that endoscopy is designed to be safely performed within the capacity of a diverse group of workers will be critical to creating an inclusive and equitable work environment.
Ergonomic education is foundational: Awareness of ERI risk factors allows endoscopists to identify hazards and advocate for effective control solutions. Ergonomic education materials are available through all of the major GI societies. A road map for implementing an endoscopy ergonomics program has been previously published and provides guidance on risk assessment and mitigation measures.
Respect pain
Overuse injuries occur when the physical demands of a job are greater than tissue tolerances, leading to cumulative microtrauma. The first sign of microtraumatic injury is discomfort and pain. Studies have demonstrated that an estimated three-quarters of gastroenterologists experience ERI, with 20% requiring time off work and 12% requiring surgery. Gastroenterology trainees are also at risk, with 20% of surveyed fellows endorsing overuse injury, some even requiring work-related leaves of absence. In the early stages of ERI, aching and tiredness occur during the work shift only. In the intermediate stage, aching and tiredness occur early in the work shift and persist at night, and may be associated with a reduced capacity for repetitive work. In the late stages, aching, fatigue and weakness persist at rest and may be associated with inability to sleep and to perform light duties. Pain is an important signal indicating mitigation measures are required to control exposures.
Utilize the hierarchy of controls
The responsibility for adoption of ergonomically friendly practices does not lie solely with the physician; both institutional and industry-level support are key to its success. The hierarchy of controls defines which actions will best mitigate exposures to hazards in the workplace, highlighting that modifications to personal practice have the smallest impact. Current endoscope design does not accommodate the full range of hand strengths and sizes and contributes to ERI.
Advancements at the industry level by eliminating or substituting hazards, or designing engineering controls to reduce exposure, will be most effective at preventing distal upper extremity ERI. The next most effective controls are at the institutional level, with endoscopy units ensuring access to engineering controls and implementing effective administrative controls. For example, institutional support and investment in adjustable workstations is imperative to accommodate a range of anthropometric dimensions of the population. Support for ergonomic education, scheduling changes and a culture where safety is a priority can help reduce exposure to hazards and injury risk.
Adjust the endoscopy suite to achieve a comfortable position before every procedure
Neutral body posture is our position of greatest comfort and maximum strength, and any deviation from neutral posture decreases the amount of force the muscles can produce and causes the muscles to fatigue sooner. The most important factor affecting the endoscopists’ overall posture is the monitor position and height. Monitors must be adjustable. Place the monitor directly in front of you, with the center of the screen 15-25 degrees below eye height for a neutral neck position and resting eye position. Procedure bed height should be adjusted 0-10 cm below elbow height to allow for neutral elbow postures and relaxed shoulders. Antifatigue mats and shoes with supportive insoles can reduce fatigue. Two-piece lead aprons distribute a portion of the static load to the hips and decrease back strain. Incorporate a preprocedure ergonomic time-out, to assess proper room set up, body mechanics, equipment and team preparedness.
Give yourself a break
Breaks should be built into the endoscopy schedule, especially for a full day of endoscopy. At a minimum, incorporate microbreaks during procedures, which have been found to alleviate pain and improve performance. Exercises and stretching can be incorporated between cases, including routines designed specifically for endoscopists.
Getting older isn’t for the weak
Currently, 50% of our gastroenterologists are over 55 years old. The aging process leads to a distinct muscle mass and strength loss. Women are already at a disadvantage because, on average, they have less muscle mass than men in all age groups. Muscle starts to deteriorate when we reach our 30s, and after age 40, we lose on average 8% of our muscle mass every decade which accelerates at an even faster rate after age 60. Both resistance and aerobic exercise can be very useful to counteract sarcopenia and maintain strength. Given the physical demands of endoscopy, exercise can help safeguard career longevity and maintain overall wellness. Multiple resources are available to tailor a program that fits your time, budget and needs.
Optimize all of your workstations
Prolonged computer use and desk work is also a significant part of a gastroenterologist’s profession. If using a sitting desk, chair height should allow for 90-degree flexion at the hips and knees and for feet to rest flatly on the floor. The chair should also provide adequate back support for a relaxed upright position. Similar to endoscopy, place the monitor directly in front with the center of the screen slightly below eye level. For mouse and keyboard placement, aim to have the elbows at or slightly below 90 degrees and one’s wrists and fingers in neutral position.
Endoscopy can be hazardous to the endoscopists’ health. Incorporating ergonomic principles creates a safer and more efficient work environment. At the individual level, ergonomically optimized postures during endoscopy as well as during computer-related tasks, room set up, inclusion of microbreaks, and protective exercises can help decrease the risk of repetitive strain injury and prevent disability. Importantly, change at the industry and institutional level has the greatest potential for positive impact. Adoption of ergonomic practices promotes career longevity and ensures that gastroenterologists can continue successful and long careers and provide quality care to their patients without compromising their own health.
Dr. Lipowska is an assistant professor in the division of gastroenterology and hepatology, University of Illinois at Chicago. She disclosed no conflicts. Dr. Shergill is chief of gastroenterology for the San Francisco VA Health Care System. She disclosed consulting work for Boston Scientific and Neptune Medical, honoraria for visiting professorship with Intuitive Surgical, and a research gift from Pentax.
References
Shergill AK. Top tips for implementing an endoscopy ergonomics program. Gastrointest Endosc. 2023 Feb;97(2):361-4.
Pawa S et al. Are all endoscopy-related musculoskeletal injuries created equal? Results of a national gender-based survey. Am J Gastroenterol. 2021;116(3):530-8.
Austin K et al. Musculoskeletal injuries are commonly reported among gastroenterology trainees: Results of a national survey. Dig Dis Sci. 2019;64(6):1439-47.
Lipowska A et al. Ergonomics in the unit: Modeling the environment around the endoscopist. Tech Innov Gastrointest Endosc. 2021;23(3):256-62.
Park A et al. Intraoperative “Micro Breaks” with a targeted stretching enhance surgeon physical function and mental focus: A multicenter cohort study. Ann Surg. 2017;265(2):340-6.
Shergill A et al. Ergonomic endoscopy: An oxymoron or realistic goal? Gastrointest Endosc. 2019;90(6):966-70.