User login
Deaths, despair tied to drug dependence are accelerating amid COVID-19
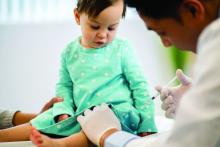
Patients with OUDs need assistance now more than ever.
The Centers for Disease Control and Prevention reported recently that opioid overdose deaths will increase to a new U.S. record, and more are expected as pandemic-related overdose deaths are yet to be counted.1
Specifically, according to the CDC, 70,980 people died from fatal overdoses in 2019,2 which is record high. Experts such as Bruce A. Goldberger, PhD, fear that the 2020 numbers could rise even higher, exacerbated by the coronavirus pandemic.
Deaths from drug overdoses remain higher than the peak yearly death totals ever recorded for car accidents, guns, or AIDS. Overdose deaths have accelerated further – pushing down overall life expectancy in the United States.3 Headlines purporting to identify good news in drug death figures don’t always get below top-level data. Deaths and despair tied to drug dependence are indeed accelerating. I am concerned about these alarmingly dangerous trends.
Synthetic opioids such as fentanyl accounted for about 3,000 deaths in 2013. By 2019, they accounted for more than 37,137.4 In addition, 16,539 deaths involved stimulants such as methamphetamine, and 16,196 deaths involved cocaine, the most recent CDC reporting shows. Opioids continue to play a role in U.S. “deaths of despair,” or rising fatalities from drugs, suicides, and alcohol among Americans without employment, hope of job opportunities, or college degrees.5 As the American Medical Association has warned,6 more people are dying from overdoses amid the COVID-19 pandemic. Clinicians need to be aware of trends so that we can help our patients navigate these challenges.
Fentanyl presents dangers
Experts had predicted that the pandemic, by limiting access to treatment, rescue, or overdose services, and increasing time at home and in the neighborhood, would result in more tragedy. In addition, the shift from prescription opioids to heroin and now to fentanyl has made deaths more common.
Fentanyls – synthetic opioids – are involved in more than half of overdose deaths, and in many of the cocaine and methamphetamine-related deaths, which also are on the rise. Fentanyl is about 100 times more potent than morphine and 50 times more potent than heroin. Breathing can stop after use of just 2 mg of fentanyl, which is about as much as trace amounts of table salt. Fentanyl has replaced heroin in many cities as the pandemic changed the relative ease of importing raw drugs such as heroin.
Another important trend is that fentanyl production and distribution throughout the United States have expanded. The ease of manufacture in unregulated sectors of the Chinese and Mexican economies is difficult for U.S. authorities to curb or eliminate. The Internet promotes novel strategies for synthesizing the substance, spreading its production across many labs; suppliers use the U.S. Postal Service for distribution, and e-commerce helps to get the drug from manufacturers to U.S. consumers for fentanyl transactions.
A recent RAND report observes that, for only $10 through the postal service, suppliers can ship a 1-kg parcel from China to the United States, and private shipments cost about $100.7 And with large volumes of legal trade between the two countries making rigorous scrutiny of products difficult, especially given the light weight of fentanyl, suppliers find it relatively easy to hide illicit substances in licit shipments. Opioid users have made the switch to fentanyl, and have seen fentanyl added to cocaine and methamphetamine they buy on the streets.
OUD and buprenorphine
Fentanyl is one part of the overdose crisis. Opioid use disorder (OUD) is the other. Both need to be addressed if we are to make any progress in this epidemic of death and dependency.
The OUD crisis continues amid the pandemic – and isn’t going away.8 Slips, relapses, and overdoses are all too common. Medication-assisted treatment (MAT) and OUD treatment programs are essential parts of our response to overdose initiatives. After naloxone rescue, the best anti-overdose response is to get the OUD patient into treatment with MATs. Patients with OUD have continuously high risks of overdose. The best outcomes appear to be related to treatment duration of greater than 2 years. But it is common to see patients with OUDs who have been in treatment multiple times, taking MATs, dropping out, overdosing, and dying. Some have been described as treatment resistant.9 It is clear that treatment can work, but also that even evidence-based treatments often fail.10
A recent study compared OUD patients who continued treatment for 6-9 months to those patients who had continued MAT treatment for 15-18 months. The longer the treatment, the fewer emergencies, prescriptions, or hospitalizations.11
But this study reminds us that all OUD patients, whether they are currently buprenorphine treated or not, experience overdoses and emergency department interventions. Short and longer treatment groups have a similar nonfatal overdose rate, about 6%, and went to the emergency department at a high rate, above 40%. Discontinuation of buprenorphine treatment is a major risk factor in opioid relapse, emergency department visits, and overdose. Cures are not common. Whether an OUD patient is being treated or has been treated in the past, carrying naloxone (brand name Narcan), makes sense and can save lives.
Methadone still considered most effective
Methadone is a synthetic opioid first studied as a treatment for OUD at Rockefeller University in New York City in the 1960s. Methadone may be the most effective treatment for OUD in promoting treatment retention for years, decreasing intravenous drug use, and decreasing deaths.12 It has been studied and safely used in treatment programs for decades. Methadone is typically administered in a clinic, daily, and with observation. In addition, methadone patients periodically take urine drug tests, which can distinguish methadone from substances of abuse. They also receive counseling. But methadone can be prescribed and administered only in methadone clinics in the United States. It is available for prescription in primary care clinics in Great Britain, Canada, and Australia.13 Numerous experts have suggested passing new legislation aimed at changing how methadone can be prescribed. Allowing primary care to administer methadone, just like buprenorphine, can improve access and benefit OUD patients.12
Availability of Narcan is critical
A comprehensive treatment model for OUDs includes prescribing naloxone, encouraging those patients with an OUD and their loved ones to have naloxone with them, and providing MATs and appropriate therapies, such as counseling.
As described by Allison L. Pitt and colleagues at Stanford (Calif.) University,14 the United States might be on track to have up to 500,000 deaths tied to opioid overdoses that might occur over the next 5 years. They modeled the effect on overdose of a long list of interventions, but only a few had an impact. At the top of the list was naloxone availability. We need to focus on saving lives by increasing naloxone availability, improving initiation, and expanding access to MAT, and increasing psychosocial treatment to improve outcomes, increase life-years and quality-adjusted life-years, and reduce opioid-related deaths. When Ms. Pitt and colleagues looked at what would make the most impact in reducing OUD deaths, it was naloxone. Pain patients on higher doses of opioids, nonprescription opioid users, OUD patients should be given naloxone prescriptions. While many can give a Heimlich to a choking person or CPR, few have naloxone to rescue a person who has overdosed on opioids. If an overdose is suspected, it should be administered by anyone who has it, as soon as possible. Then, the person who is intervening should call 911.
What we can do today
At this moment, clinicians can follow the Surgeon General’s advice,15 and prescribe naloxone.
We should give naloxone to OUD patients and their families, to pain patients at dosages of greater than or equal to 50 MME. Our top priorities should be patients with comorbid pain syndromes, those being treated with benzodiazepines and sleeping medications, and patients with alcohol use disorders. This is also an important intervention for those who binge drink, and have sleep apnea, and heart and respiratory diseases.
Naloxone is available without a prescription in at least 43 states. Naloxone is available in harm reduction programs and in hospitals, and is carried by emergency medical staff, law enforcement, and EMTs. It also is available on the streets, though it does not appear to have a dollar value like opioids or even buprenorphine. Also, the availability of naloxone in pharmacies has made it easier for family members and caregivers of pain patients or those with OUD to have it to administer in an emergency.
An excellent place for MDs to start is to do more to encourage all patients with OUD to carry naloxone, for their loved ones to carry naloxone, and for their homes to have naloxone nearby in the bedroom or bathroom. It is not logical to expect a person with an OUD to rescue themselves. Current and past OUD patients, as well as their loved ones, are at high risk – and should have naloxone nearby at all times.
Naloxone reverses an opioid overdose, but it should be thought about like cardioversion or CPR rather than a treatment for an underlying disease. Increasing access to buprenorphine, buprenorphine + naloxone, and naltrexone treatment for OUDs is an important organizing principle. Initiation of MAT treatment in the emergency setting or most anywhere and any place a patient with an OUD can begin treatment is necessary. Treatment with buprenorphine or methadone reduces opioid overdose and opioid-related acute care use.16
Reducing racial disparities in OUD treatment is necessary, because buprenorphine treatment is concentrated among White patients who either use private insurance or are self-pay.17 Reducing barriers to methadone program licenses, expanding sites for distribution,18 prescribing methadone in an office setting might help. Clinicians can do a better job of explaining the risks associated with opioid prescriptions, including diversion and overdose, and the benefits of OUD treatment. So, To reduce opioid overdoses, we must increase physician competencies in addiction medicine.
Dr. Gold is professor of psychiatry (adjunct) at Washington University, St. Louis. He is the 17th Distinguished Alumni Professor at the University of Florida, Gainesville. For more than 40 years, Dr. Gold has worked on developing models for understanding the effects of opioid, tobacco, cocaine, and other drugs, as well as food, on the brain and behavior. He disclosed financial ties with ADAPT Pharma and Magstim Ltd.
References
1. Kamp J. Overdose deaths rise, may reach record level, federal data show. Wall Street Journal. 2020 Jul 15.
2. 12 month–ending provisional number of drug overdose drugs. Centers for Disease Control and Prevention. 2020 Jul 5.
3. Katz J et al. In shadow of pandemic, U.S. drug overdose deaths resurge to record. New York Times. 2020 Jul 15.
4. Gold MS. The fentanyl crisis is only getting worse. Addiction Policy Forum. Updated 2020 Mar 12.
5. Gold MS. Mo Med. 2020-Mar-Apr;117(2):99-101.
6. Reports of increases in opioid-related overdoses and other concerns during the COVID-19 pandemic. American Medical Association. Issue brief. Updated 2020 Jul 20.
7. Pardo B et al. The future of fentanyl and other synthetic opioids. RAND report.
8. Gold MS. New challenges in the opioid epidemic. Addiction Policy Forum. 2020 Jun 4.
9. Patterson Silver Wolf DA and Gold MS. J Neurol Sci. 2020;411:116718.
10. Oesterle TS et al. Mayo Clin Proc. 2019;94(10):2072-86.
11. Connery HS and Weiss RD. Am J Psychiatry. 2020;177(2):104-6.
12. Kleber HD. JAMA. 2008;300(19):2303-5.
13. Samet JH et al. N Engl J Med. 2018;379(1):7-8.
14. Pitt AL et al. Am J Public Health. 2018;108(10):1394-1400.
15. U.S. Surgeon General’s Advisory on Naloxone and Opioid Overdose. hhs.gov.
16. Wakeman SE et al. JAMA Netw Open. 2020;3(2):e1920622.
17. Lagisetty PA et al. JAMA Psychiatry. 2019;76(9):979-81.
18. Kleinman RA. JAMA Psychiatry. 2020 Jul 15. doi: 10.1001/jamapsychiatry.2020.1624.
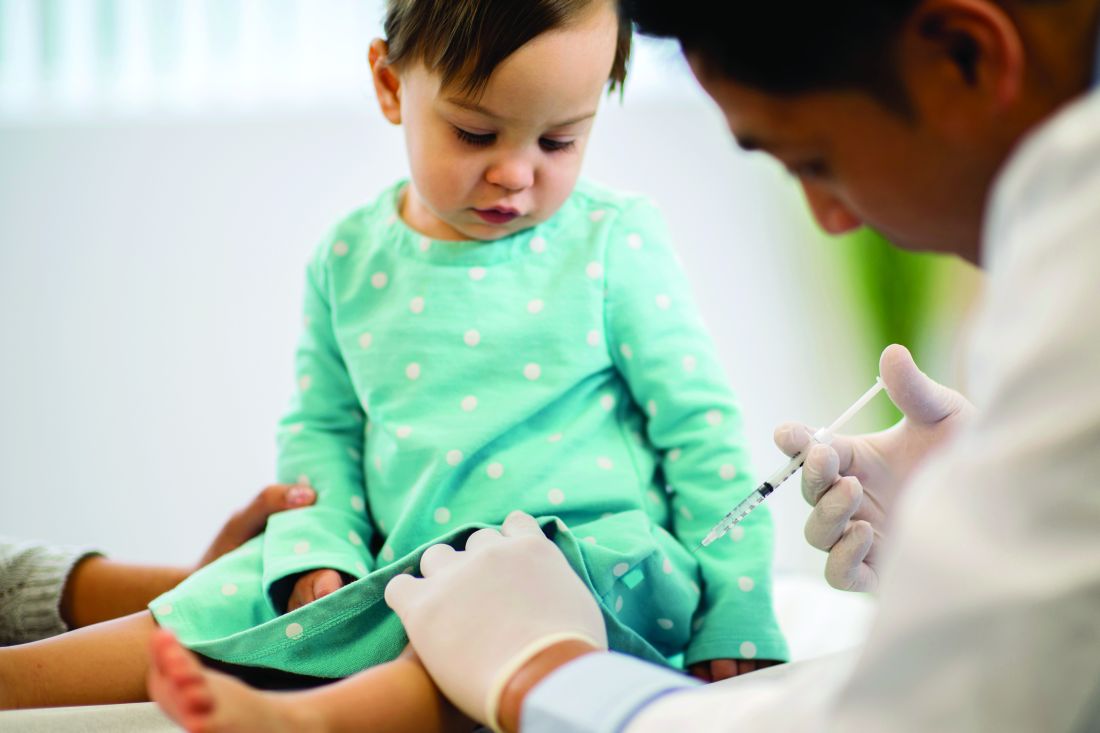
Patients with OUDs need assistance now more than ever.
Patients with OUDs need assistance now more than ever.
The Centers for Disease Control and Prevention reported recently that opioid overdose deaths will increase to a new U.S. record, and more are expected as pandemic-related overdose deaths are yet to be counted.1
Specifically, according to the CDC, 70,980 people died from fatal overdoses in 2019,2 which is record high. Experts such as Bruce A. Goldberger, PhD, fear that the 2020 numbers could rise even higher, exacerbated by the coronavirus pandemic.
Deaths from drug overdoses remain higher than the peak yearly death totals ever recorded for car accidents, guns, or AIDS. Overdose deaths have accelerated further – pushing down overall life expectancy in the United States.3 Headlines purporting to identify good news in drug death figures don’t always get below top-level data. Deaths and despair tied to drug dependence are indeed accelerating. I am concerned about these alarmingly dangerous trends.
Synthetic opioids such as fentanyl accounted for about 3,000 deaths in 2013. By 2019, they accounted for more than 37,137.4 In addition, 16,539 deaths involved stimulants such as methamphetamine, and 16,196 deaths involved cocaine, the most recent CDC reporting shows. Opioids continue to play a role in U.S. “deaths of despair,” or rising fatalities from drugs, suicides, and alcohol among Americans without employment, hope of job opportunities, or college degrees.5 As the American Medical Association has warned,6 more people are dying from overdoses amid the COVID-19 pandemic. Clinicians need to be aware of trends so that we can help our patients navigate these challenges.
Fentanyl presents dangers
Experts had predicted that the pandemic, by limiting access to treatment, rescue, or overdose services, and increasing time at home and in the neighborhood, would result in more tragedy. In addition, the shift from prescription opioids to heroin and now to fentanyl has made deaths more common.
Fentanyls – synthetic opioids – are involved in more than half of overdose deaths, and in many of the cocaine and methamphetamine-related deaths, which also are on the rise. Fentanyl is about 100 times more potent than morphine and 50 times more potent than heroin. Breathing can stop after use of just 2 mg of fentanyl, which is about as much as trace amounts of table salt. Fentanyl has replaced heroin in many cities as the pandemic changed the relative ease of importing raw drugs such as heroin.
Another important trend is that fentanyl production and distribution throughout the United States have expanded. The ease of manufacture in unregulated sectors of the Chinese and Mexican economies is difficult for U.S. authorities to curb or eliminate. The Internet promotes novel strategies for synthesizing the substance, spreading its production across many labs; suppliers use the U.S. Postal Service for distribution, and e-commerce helps to get the drug from manufacturers to U.S. consumers for fentanyl transactions.
A recent RAND report observes that, for only $10 through the postal service, suppliers can ship a 1-kg parcel from China to the United States, and private shipments cost about $100.7 And with large volumes of legal trade between the two countries making rigorous scrutiny of products difficult, especially given the light weight of fentanyl, suppliers find it relatively easy to hide illicit substances in licit shipments. Opioid users have made the switch to fentanyl, and have seen fentanyl added to cocaine and methamphetamine they buy on the streets.
OUD and buprenorphine
Fentanyl is one part of the overdose crisis. Opioid use disorder (OUD) is the other. Both need to be addressed if we are to make any progress in this epidemic of death and dependency.
The OUD crisis continues amid the pandemic – and isn’t going away.8 Slips, relapses, and overdoses are all too common. Medication-assisted treatment (MAT) and OUD treatment programs are essential parts of our response to overdose initiatives. After naloxone rescue, the best anti-overdose response is to get the OUD patient into treatment with MATs. Patients with OUD have continuously high risks of overdose. The best outcomes appear to be related to treatment duration of greater than 2 years. But it is common to see patients with OUDs who have been in treatment multiple times, taking MATs, dropping out, overdosing, and dying. Some have been described as treatment resistant.9 It is clear that treatment can work, but also that even evidence-based treatments often fail.10
A recent study compared OUD patients who continued treatment for 6-9 months to those patients who had continued MAT treatment for 15-18 months. The longer the treatment, the fewer emergencies, prescriptions, or hospitalizations.11
But this study reminds us that all OUD patients, whether they are currently buprenorphine treated or not, experience overdoses and emergency department interventions. Short and longer treatment groups have a similar nonfatal overdose rate, about 6%, and went to the emergency department at a high rate, above 40%. Discontinuation of buprenorphine treatment is a major risk factor in opioid relapse, emergency department visits, and overdose. Cures are not common. Whether an OUD patient is being treated or has been treated in the past, carrying naloxone (brand name Narcan), makes sense and can save lives.
Methadone still considered most effective
Methadone is a synthetic opioid first studied as a treatment for OUD at Rockefeller University in New York City in the 1960s. Methadone may be the most effective treatment for OUD in promoting treatment retention for years, decreasing intravenous drug use, and decreasing deaths.12 It has been studied and safely used in treatment programs for decades. Methadone is typically administered in a clinic, daily, and with observation. In addition, methadone patients periodically take urine drug tests, which can distinguish methadone from substances of abuse. They also receive counseling. But methadone can be prescribed and administered only in methadone clinics in the United States. It is available for prescription in primary care clinics in Great Britain, Canada, and Australia.13 Numerous experts have suggested passing new legislation aimed at changing how methadone can be prescribed. Allowing primary care to administer methadone, just like buprenorphine, can improve access and benefit OUD patients.12
Availability of Narcan is critical
A comprehensive treatment model for OUDs includes prescribing naloxone, encouraging those patients with an OUD and their loved ones to have naloxone with them, and providing MATs and appropriate therapies, such as counseling.
As described by Allison L. Pitt and colleagues at Stanford (Calif.) University,14 the United States might be on track to have up to 500,000 deaths tied to opioid overdoses that might occur over the next 5 years. They modeled the effect on overdose of a long list of interventions, but only a few had an impact. At the top of the list was naloxone availability. We need to focus on saving lives by increasing naloxone availability, improving initiation, and expanding access to MAT, and increasing psychosocial treatment to improve outcomes, increase life-years and quality-adjusted life-years, and reduce opioid-related deaths. When Ms. Pitt and colleagues looked at what would make the most impact in reducing OUD deaths, it was naloxone. Pain patients on higher doses of opioids, nonprescription opioid users, OUD patients should be given naloxone prescriptions. While many can give a Heimlich to a choking person or CPR, few have naloxone to rescue a person who has overdosed on opioids. If an overdose is suspected, it should be administered by anyone who has it, as soon as possible. Then, the person who is intervening should call 911.
What we can do today
At this moment, clinicians can follow the Surgeon General’s advice,15 and prescribe naloxone.
We should give naloxone to OUD patients and their families, to pain patients at dosages of greater than or equal to 50 MME. Our top priorities should be patients with comorbid pain syndromes, those being treated with benzodiazepines and sleeping medications, and patients with alcohol use disorders. This is also an important intervention for those who binge drink, and have sleep apnea, and heart and respiratory diseases.
Naloxone is available without a prescription in at least 43 states. Naloxone is available in harm reduction programs and in hospitals, and is carried by emergency medical staff, law enforcement, and EMTs. It also is available on the streets, though it does not appear to have a dollar value like opioids or even buprenorphine. Also, the availability of naloxone in pharmacies has made it easier for family members and caregivers of pain patients or those with OUD to have it to administer in an emergency.
An excellent place for MDs to start is to do more to encourage all patients with OUD to carry naloxone, for their loved ones to carry naloxone, and for their homes to have naloxone nearby in the bedroom or bathroom. It is not logical to expect a person with an OUD to rescue themselves. Current and past OUD patients, as well as their loved ones, are at high risk – and should have naloxone nearby at all times.
Naloxone reverses an opioid overdose, but it should be thought about like cardioversion or CPR rather than a treatment for an underlying disease. Increasing access to buprenorphine, buprenorphine + naloxone, and naltrexone treatment for OUDs is an important organizing principle. Initiation of MAT treatment in the emergency setting or most anywhere and any place a patient with an OUD can begin treatment is necessary. Treatment with buprenorphine or methadone reduces opioid overdose and opioid-related acute care use.16
Reducing racial disparities in OUD treatment is necessary, because buprenorphine treatment is concentrated among White patients who either use private insurance or are self-pay.17 Reducing barriers to methadone program licenses, expanding sites for distribution,18 prescribing methadone in an office setting might help. Clinicians can do a better job of explaining the risks associated with opioid prescriptions, including diversion and overdose, and the benefits of OUD treatment. So, To reduce opioid overdoses, we must increase physician competencies in addiction medicine.
Dr. Gold is professor of psychiatry (adjunct) at Washington University, St. Louis. He is the 17th Distinguished Alumni Professor at the University of Florida, Gainesville. For more than 40 years, Dr. Gold has worked on developing models for understanding the effects of opioid, tobacco, cocaine, and other drugs, as well as food, on the brain and behavior. He disclosed financial ties with ADAPT Pharma and Magstim Ltd.
References
1. Kamp J. Overdose deaths rise, may reach record level, federal data show. Wall Street Journal. 2020 Jul 15.
2. 12 month–ending provisional number of drug overdose drugs. Centers for Disease Control and Prevention. 2020 Jul 5.
3. Katz J et al. In shadow of pandemic, U.S. drug overdose deaths resurge to record. New York Times. 2020 Jul 15.
4. Gold MS. The fentanyl crisis is only getting worse. Addiction Policy Forum. Updated 2020 Mar 12.
5. Gold MS. Mo Med. 2020-Mar-Apr;117(2):99-101.
6. Reports of increases in opioid-related overdoses and other concerns during the COVID-19 pandemic. American Medical Association. Issue brief. Updated 2020 Jul 20.
7. Pardo B et al. The future of fentanyl and other synthetic opioids. RAND report.
8. Gold MS. New challenges in the opioid epidemic. Addiction Policy Forum. 2020 Jun 4.
9. Patterson Silver Wolf DA and Gold MS. J Neurol Sci. 2020;411:116718.
10. Oesterle TS et al. Mayo Clin Proc. 2019;94(10):2072-86.
11. Connery HS and Weiss RD. Am J Psychiatry. 2020;177(2):104-6.
12. Kleber HD. JAMA. 2008;300(19):2303-5.
13. Samet JH et al. N Engl J Med. 2018;379(1):7-8.
14. Pitt AL et al. Am J Public Health. 2018;108(10):1394-1400.
15. U.S. Surgeon General’s Advisory on Naloxone and Opioid Overdose. hhs.gov.
16. Wakeman SE et al. JAMA Netw Open. 2020;3(2):e1920622.
17. Lagisetty PA et al. JAMA Psychiatry. 2019;76(9):979-81.
18. Kleinman RA. JAMA Psychiatry. 2020 Jul 15. doi: 10.1001/jamapsychiatry.2020.1624.
The Centers for Disease Control and Prevention reported recently that opioid overdose deaths will increase to a new U.S. record, and more are expected as pandemic-related overdose deaths are yet to be counted.1
Specifically, according to the CDC, 70,980 people died from fatal overdoses in 2019,2 which is record high. Experts such as Bruce A. Goldberger, PhD, fear that the 2020 numbers could rise even higher, exacerbated by the coronavirus pandemic.
Deaths from drug overdoses remain higher than the peak yearly death totals ever recorded for car accidents, guns, or AIDS. Overdose deaths have accelerated further – pushing down overall life expectancy in the United States.3 Headlines purporting to identify good news in drug death figures don’t always get below top-level data. Deaths and despair tied to drug dependence are indeed accelerating. I am concerned about these alarmingly dangerous trends.
Synthetic opioids such as fentanyl accounted for about 3,000 deaths in 2013. By 2019, they accounted for more than 37,137.4 In addition, 16,539 deaths involved stimulants such as methamphetamine, and 16,196 deaths involved cocaine, the most recent CDC reporting shows. Opioids continue to play a role in U.S. “deaths of despair,” or rising fatalities from drugs, suicides, and alcohol among Americans without employment, hope of job opportunities, or college degrees.5 As the American Medical Association has warned,6 more people are dying from overdoses amid the COVID-19 pandemic. Clinicians need to be aware of trends so that we can help our patients navigate these challenges.
Fentanyl presents dangers
Experts had predicted that the pandemic, by limiting access to treatment, rescue, or overdose services, and increasing time at home and in the neighborhood, would result in more tragedy. In addition, the shift from prescription opioids to heroin and now to fentanyl has made deaths more common.
Fentanyls – synthetic opioids – are involved in more than half of overdose deaths, and in many of the cocaine and methamphetamine-related deaths, which also are on the rise. Fentanyl is about 100 times more potent than morphine and 50 times more potent than heroin. Breathing can stop after use of just 2 mg of fentanyl, which is about as much as trace amounts of table salt. Fentanyl has replaced heroin in many cities as the pandemic changed the relative ease of importing raw drugs such as heroin.
Another important trend is that fentanyl production and distribution throughout the United States have expanded. The ease of manufacture in unregulated sectors of the Chinese and Mexican economies is difficult for U.S. authorities to curb or eliminate. The Internet promotes novel strategies for synthesizing the substance, spreading its production across many labs; suppliers use the U.S. Postal Service for distribution, and e-commerce helps to get the drug from manufacturers to U.S. consumers for fentanyl transactions.
A recent RAND report observes that, for only $10 through the postal service, suppliers can ship a 1-kg parcel from China to the United States, and private shipments cost about $100.7 And with large volumes of legal trade between the two countries making rigorous scrutiny of products difficult, especially given the light weight of fentanyl, suppliers find it relatively easy to hide illicit substances in licit shipments. Opioid users have made the switch to fentanyl, and have seen fentanyl added to cocaine and methamphetamine they buy on the streets.
OUD and buprenorphine
Fentanyl is one part of the overdose crisis. Opioid use disorder (OUD) is the other. Both need to be addressed if we are to make any progress in this epidemic of death and dependency.
The OUD crisis continues amid the pandemic – and isn’t going away.8 Slips, relapses, and overdoses are all too common. Medication-assisted treatment (MAT) and OUD treatment programs are essential parts of our response to overdose initiatives. After naloxone rescue, the best anti-overdose response is to get the OUD patient into treatment with MATs. Patients with OUD have continuously high risks of overdose. The best outcomes appear to be related to treatment duration of greater than 2 years. But it is common to see patients with OUDs who have been in treatment multiple times, taking MATs, dropping out, overdosing, and dying. Some have been described as treatment resistant.9 It is clear that treatment can work, but also that even evidence-based treatments often fail.10
A recent study compared OUD patients who continued treatment for 6-9 months to those patients who had continued MAT treatment for 15-18 months. The longer the treatment, the fewer emergencies, prescriptions, or hospitalizations.11
But this study reminds us that all OUD patients, whether they are currently buprenorphine treated or not, experience overdoses and emergency department interventions. Short and longer treatment groups have a similar nonfatal overdose rate, about 6%, and went to the emergency department at a high rate, above 40%. Discontinuation of buprenorphine treatment is a major risk factor in opioid relapse, emergency department visits, and overdose. Cures are not common. Whether an OUD patient is being treated or has been treated in the past, carrying naloxone (brand name Narcan), makes sense and can save lives.
Methadone still considered most effective
Methadone is a synthetic opioid first studied as a treatment for OUD at Rockefeller University in New York City in the 1960s. Methadone may be the most effective treatment for OUD in promoting treatment retention for years, decreasing intravenous drug use, and decreasing deaths.12 It has been studied and safely used in treatment programs for decades. Methadone is typically administered in a clinic, daily, and with observation. In addition, methadone patients periodically take urine drug tests, which can distinguish methadone from substances of abuse. They also receive counseling. But methadone can be prescribed and administered only in methadone clinics in the United States. It is available for prescription in primary care clinics in Great Britain, Canada, and Australia.13 Numerous experts have suggested passing new legislation aimed at changing how methadone can be prescribed. Allowing primary care to administer methadone, just like buprenorphine, can improve access and benefit OUD patients.12
Availability of Narcan is critical
A comprehensive treatment model for OUDs includes prescribing naloxone, encouraging those patients with an OUD and their loved ones to have naloxone with them, and providing MATs and appropriate therapies, such as counseling.
As described by Allison L. Pitt and colleagues at Stanford (Calif.) University,14 the United States might be on track to have up to 500,000 deaths tied to opioid overdoses that might occur over the next 5 years. They modeled the effect on overdose of a long list of interventions, but only a few had an impact. At the top of the list was naloxone availability. We need to focus on saving lives by increasing naloxone availability, improving initiation, and expanding access to MAT, and increasing psychosocial treatment to improve outcomes, increase life-years and quality-adjusted life-years, and reduce opioid-related deaths. When Ms. Pitt and colleagues looked at what would make the most impact in reducing OUD deaths, it was naloxone. Pain patients on higher doses of opioids, nonprescription opioid users, OUD patients should be given naloxone prescriptions. While many can give a Heimlich to a choking person or CPR, few have naloxone to rescue a person who has overdosed on opioids. If an overdose is suspected, it should be administered by anyone who has it, as soon as possible. Then, the person who is intervening should call 911.
What we can do today
At this moment, clinicians can follow the Surgeon General’s advice,15 and prescribe naloxone.
We should give naloxone to OUD patients and their families, to pain patients at dosages of greater than or equal to 50 MME. Our top priorities should be patients with comorbid pain syndromes, those being treated with benzodiazepines and sleeping medications, and patients with alcohol use disorders. This is also an important intervention for those who binge drink, and have sleep apnea, and heart and respiratory diseases.
Naloxone is available without a prescription in at least 43 states. Naloxone is available in harm reduction programs and in hospitals, and is carried by emergency medical staff, law enforcement, and EMTs. It also is available on the streets, though it does not appear to have a dollar value like opioids or even buprenorphine. Also, the availability of naloxone in pharmacies has made it easier for family members and caregivers of pain patients or those with OUD to have it to administer in an emergency.
An excellent place for MDs to start is to do more to encourage all patients with OUD to carry naloxone, for their loved ones to carry naloxone, and for their homes to have naloxone nearby in the bedroom or bathroom. It is not logical to expect a person with an OUD to rescue themselves. Current and past OUD patients, as well as their loved ones, are at high risk – and should have naloxone nearby at all times.
Naloxone reverses an opioid overdose, but it should be thought about like cardioversion or CPR rather than a treatment for an underlying disease. Increasing access to buprenorphine, buprenorphine + naloxone, and naltrexone treatment for OUDs is an important organizing principle. Initiation of MAT treatment in the emergency setting or most anywhere and any place a patient with an OUD can begin treatment is necessary. Treatment with buprenorphine or methadone reduces opioid overdose and opioid-related acute care use.16
Reducing racial disparities in OUD treatment is necessary, because buprenorphine treatment is concentrated among White patients who either use private insurance or are self-pay.17 Reducing barriers to methadone program licenses, expanding sites for distribution,18 prescribing methadone in an office setting might help. Clinicians can do a better job of explaining the risks associated with opioid prescriptions, including diversion and overdose, and the benefits of OUD treatment. So, To reduce opioid overdoses, we must increase physician competencies in addiction medicine.
Dr. Gold is professor of psychiatry (adjunct) at Washington University, St. Louis. He is the 17th Distinguished Alumni Professor at the University of Florida, Gainesville. For more than 40 years, Dr. Gold has worked on developing models for understanding the effects of opioid, tobacco, cocaine, and other drugs, as well as food, on the brain and behavior. He disclosed financial ties with ADAPT Pharma and Magstim Ltd.
References
1. Kamp J. Overdose deaths rise, may reach record level, federal data show. Wall Street Journal. 2020 Jul 15.
2. 12 month–ending provisional number of drug overdose drugs. Centers for Disease Control and Prevention. 2020 Jul 5.
3. Katz J et al. In shadow of pandemic, U.S. drug overdose deaths resurge to record. New York Times. 2020 Jul 15.
4. Gold MS. The fentanyl crisis is only getting worse. Addiction Policy Forum. Updated 2020 Mar 12.
5. Gold MS. Mo Med. 2020-Mar-Apr;117(2):99-101.
6. Reports of increases in opioid-related overdoses and other concerns during the COVID-19 pandemic. American Medical Association. Issue brief. Updated 2020 Jul 20.
7. Pardo B et al. The future of fentanyl and other synthetic opioids. RAND report.
8. Gold MS. New challenges in the opioid epidemic. Addiction Policy Forum. 2020 Jun 4.
9. Patterson Silver Wolf DA and Gold MS. J Neurol Sci. 2020;411:116718.
10. Oesterle TS et al. Mayo Clin Proc. 2019;94(10):2072-86.
11. Connery HS and Weiss RD. Am J Psychiatry. 2020;177(2):104-6.
12. Kleber HD. JAMA. 2008;300(19):2303-5.
13. Samet JH et al. N Engl J Med. 2018;379(1):7-8.
14. Pitt AL et al. Am J Public Health. 2018;108(10):1394-1400.
15. U.S. Surgeon General’s Advisory on Naloxone and Opioid Overdose. hhs.gov.
16. Wakeman SE et al. JAMA Netw Open. 2020;3(2):e1920622.
17. Lagisetty PA et al. JAMA Psychiatry. 2019;76(9):979-81.
18. Kleinman RA. JAMA Psychiatry. 2020 Jul 15. doi: 10.1001/jamapsychiatry.2020.1624.
Action and awareness are needed to increase immunization rates
August was National Immunization Awareness Month. ... just in time to address the precipitous drop in immunization delivered during the early months of the pandemic.
In May, the Centers for Disease Control and Prevention reported substantial reductions in vaccine doses ordered through the Vaccines for Children program after the declaration of national emergency because of COVID-19 on March 13. Approximately 2.5 million fewer doses of routine, noninfluenza vaccines were administered between Jan. 6 and April 2020, compared with a similar period last year (MMWR Morb Mortal Wkly Rep. 2020 May 15;69[19]:591-3). Declines in immunization rates were echoed by states and municipalities across the United States. Last month, the health system in which I work reported 40,000 children behind on at least one vaccine.
We all know that, when immunization rates drop, outbreaks of vaccine-preventable diseases follow. In order and that is going to take more than a single month.
Identify patients who’ve missed vaccinations
Simply being open and ready to vaccinate is not enough. The Centers for Disease Control and Prevention urges providers to identify patients who have missed vaccines, and call them to schedule in-person visits. Proactively let parents know about strategies implemented in your office to ensure a safe environment.
Pediatricians are accustomed to an influx of patients in the summer, as parents make sure their children have all of the vaccines required for school attendance. As noted in a Washington Post article from Aug. 4, 2020, schools have traditionally served as a backstop for immunization rates. But as many school districts opt to take education online this fall, the implications for vaccine requirements are unclear. District of Columbia public schools continue to require immunization for virtual school attendance, but it is not clear how easily this can be enforced. To read about how other school districts have chosen to address – or not address – immunization requirements for school, visit the the Immunization Action Coalition’s Repository of Resources for Maintaining Immunization during the COVID-19 Pandemic. The repository links to international, national, and state-level policies and guidance and advocacy materials, including talking points, webinars, press releases, media articles from around the United States and social media posts, as well as telehealth resources.
Get some inspiration to talk about vaccination
Need a little inspiration for talking to parents about vaccines? Check out the CDC’s #HowIRecommend video series. These are short videos, most under a minute in length, that explain the importance of vaccination, how to effectively address questions from parents about vaccine safety, and how clinicians routinely recommend same day vaccination to their patients. These videos are part of the CDC’s National Immunization Awareness Month (NIAM) toolkit for communication with health care professionals. A companion toolkit for communicating with parents and patients contains sample social media messages with graphics, along with educational resources to share with parents.
The “Comprehensive Vaccine Education Program – From Training to Practice,” a free online program offered by the Pediatric Infectious Diseases Society, takes a deeper dive into strategies to combat vaccine misinformation and address vaccine hesitancy. Available modules cover vaccine fundamentals, vaccine safety, clinical manifestations of vaccine-preventable diseases, and communication skills that lead to more effective conversations with patients and parents. The curriculum also includes the newest edition of The Vaccine Handbook app, a comprehensive source of practical information for vaccine providers.
Educate young children about vaccines
Don’t leave young children out of the conversation. Vax-Force is a children’s book that explores how vaccination works inside the human body. Dr. Vaxson the pediatrician explains how trusted doctors and scientists made Vicky the Vaccine. Her mission is to tell Willy the White Blood Cell and his Antibuddies how to find and fight bad-guy germs like measles, tetanus, and polio. The book was written by Kelsey Rowe, MD, while she was a medical student at Saint Louis University School of Medicine. Dr. Rowe, now a pediatric resident, notes, “In a world where anti-vaccination rhetoric threatens the health of our global community, this book’s mission is to teach children and adults alike that getting vaccinations is a safe, effective, and even exciting thing to do.” The book is available for purchase at https://www.vax-force.com/, and a small part of every sale is donated to Unicef USA.
Consider vaccination advocacy in your communities
Vaccinate Your Family, a national, nonprofit organization dedicated to protecting people of all ages from vaccine-preventable diseases, suggests that health care providers need to take an active role in raising immunization rates, not just in their own practices, but in their communities. One way to do this is to submit an opinion piece or letter to the editor to a local newspaper describing why it’s important for parents to make sure their child’s immunizations are current. Those who have never written an opinion-editorial should look at the guidance developed by Voices for Vaccines.
How are we doing?
Early data suggest a rebound in immunization rates in May and June, but that is unlikely to close the gap created by disruptions in health care delivery earlier in the year. Collectively, we need to set ambitious goals. Are we just trying to reach prepandemic immunization levels? In Kentucky, where I practice, only 71% of kids aged 19-45 months had received all doses of seven routinely recommended vaccines (≥4 DTaP doses, ≥3 polio doses, ≥1 MMR dose, Hib full series, ≥3 HepB doses, ≥1 varicella dose, and ≥4 PCV doses) based on 2017 National Immunization Survey data. The Healthy People 2020 target goal is 80%. Only 55% of Kentucky girls aged 13-17 years received at least one dose of HPV vaccine, and rates in boys were even lower. Flu vaccine coverage in children 6 months to 17 years also was 55%. The status quo sets the bar too low. To see how your state is doing, check out the interactive map developed by the American Academy of Pediatrics.
Are we attempting to avoid disaster or can we seize the opportunity to protect more children than ever from vaccine-preventable diseases? The latter would really be something to celebrate.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She said she had no relevant financial disclosures. Email her at [email protected].
August was National Immunization Awareness Month. ... just in time to address the precipitous drop in immunization delivered during the early months of the pandemic.
In May, the Centers for Disease Control and Prevention reported substantial reductions in vaccine doses ordered through the Vaccines for Children program after the declaration of national emergency because of COVID-19 on March 13. Approximately 2.5 million fewer doses of routine, noninfluenza vaccines were administered between Jan. 6 and April 2020, compared with a similar period last year (MMWR Morb Mortal Wkly Rep. 2020 May 15;69[19]:591-3). Declines in immunization rates were echoed by states and municipalities across the United States. Last month, the health system in which I work reported 40,000 children behind on at least one vaccine.
We all know that, when immunization rates drop, outbreaks of vaccine-preventable diseases follow. In order and that is going to take more than a single month.
Identify patients who’ve missed vaccinations
Simply being open and ready to vaccinate is not enough. The Centers for Disease Control and Prevention urges providers to identify patients who have missed vaccines, and call them to schedule in-person visits. Proactively let parents know about strategies implemented in your office to ensure a safe environment.
Pediatricians are accustomed to an influx of patients in the summer, as parents make sure their children have all of the vaccines required for school attendance. As noted in a Washington Post article from Aug. 4, 2020, schools have traditionally served as a backstop for immunization rates. But as many school districts opt to take education online this fall, the implications for vaccine requirements are unclear. District of Columbia public schools continue to require immunization for virtual school attendance, but it is not clear how easily this can be enforced. To read about how other school districts have chosen to address – or not address – immunization requirements for school, visit the the Immunization Action Coalition’s Repository of Resources for Maintaining Immunization during the COVID-19 Pandemic. The repository links to international, national, and state-level policies and guidance and advocacy materials, including talking points, webinars, press releases, media articles from around the United States and social media posts, as well as telehealth resources.
Get some inspiration to talk about vaccination
Need a little inspiration for talking to parents about vaccines? Check out the CDC’s #HowIRecommend video series. These are short videos, most under a minute in length, that explain the importance of vaccination, how to effectively address questions from parents about vaccine safety, and how clinicians routinely recommend same day vaccination to their patients. These videos are part of the CDC’s National Immunization Awareness Month (NIAM) toolkit for communication with health care professionals. A companion toolkit for communicating with parents and patients contains sample social media messages with graphics, along with educational resources to share with parents.
The “Comprehensive Vaccine Education Program – From Training to Practice,” a free online program offered by the Pediatric Infectious Diseases Society, takes a deeper dive into strategies to combat vaccine misinformation and address vaccine hesitancy. Available modules cover vaccine fundamentals, vaccine safety, clinical manifestations of vaccine-preventable diseases, and communication skills that lead to more effective conversations with patients and parents. The curriculum also includes the newest edition of The Vaccine Handbook app, a comprehensive source of practical information for vaccine providers.
Educate young children about vaccines
Don’t leave young children out of the conversation. Vax-Force is a children’s book that explores how vaccination works inside the human body. Dr. Vaxson the pediatrician explains how trusted doctors and scientists made Vicky the Vaccine. Her mission is to tell Willy the White Blood Cell and his Antibuddies how to find and fight bad-guy germs like measles, tetanus, and polio. The book was written by Kelsey Rowe, MD, while she was a medical student at Saint Louis University School of Medicine. Dr. Rowe, now a pediatric resident, notes, “In a world where anti-vaccination rhetoric threatens the health of our global community, this book’s mission is to teach children and adults alike that getting vaccinations is a safe, effective, and even exciting thing to do.” The book is available for purchase at https://www.vax-force.com/, and a small part of every sale is donated to Unicef USA.
Consider vaccination advocacy in your communities
Vaccinate Your Family, a national, nonprofit organization dedicated to protecting people of all ages from vaccine-preventable diseases, suggests that health care providers need to take an active role in raising immunization rates, not just in their own practices, but in their communities. One way to do this is to submit an opinion piece or letter to the editor to a local newspaper describing why it’s important for parents to make sure their child’s immunizations are current. Those who have never written an opinion-editorial should look at the guidance developed by Voices for Vaccines.
How are we doing?
Early data suggest a rebound in immunization rates in May and June, but that is unlikely to close the gap created by disruptions in health care delivery earlier in the year. Collectively, we need to set ambitious goals. Are we just trying to reach prepandemic immunization levels? In Kentucky, where I practice, only 71% of kids aged 19-45 months had received all doses of seven routinely recommended vaccines (≥4 DTaP doses, ≥3 polio doses, ≥1 MMR dose, Hib full series, ≥3 HepB doses, ≥1 varicella dose, and ≥4 PCV doses) based on 2017 National Immunization Survey data. The Healthy People 2020 target goal is 80%. Only 55% of Kentucky girls aged 13-17 years received at least one dose of HPV vaccine, and rates in boys were even lower. Flu vaccine coverage in children 6 months to 17 years also was 55%. The status quo sets the bar too low. To see how your state is doing, check out the interactive map developed by the American Academy of Pediatrics.
Are we attempting to avoid disaster or can we seize the opportunity to protect more children than ever from vaccine-preventable diseases? The latter would really be something to celebrate.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She said she had no relevant financial disclosures. Email her at [email protected].
August was National Immunization Awareness Month. ... just in time to address the precipitous drop in immunization delivered during the early months of the pandemic.
In May, the Centers for Disease Control and Prevention reported substantial reductions in vaccine doses ordered through the Vaccines for Children program after the declaration of national emergency because of COVID-19 on March 13. Approximately 2.5 million fewer doses of routine, noninfluenza vaccines were administered between Jan. 6 and April 2020, compared with a similar period last year (MMWR Morb Mortal Wkly Rep. 2020 May 15;69[19]:591-3). Declines in immunization rates were echoed by states and municipalities across the United States. Last month, the health system in which I work reported 40,000 children behind on at least one vaccine.
We all know that, when immunization rates drop, outbreaks of vaccine-preventable diseases follow. In order and that is going to take more than a single month.
Identify patients who’ve missed vaccinations
Simply being open and ready to vaccinate is not enough. The Centers for Disease Control and Prevention urges providers to identify patients who have missed vaccines, and call them to schedule in-person visits. Proactively let parents know about strategies implemented in your office to ensure a safe environment.
Pediatricians are accustomed to an influx of patients in the summer, as parents make sure their children have all of the vaccines required for school attendance. As noted in a Washington Post article from Aug. 4, 2020, schools have traditionally served as a backstop for immunization rates. But as many school districts opt to take education online this fall, the implications for vaccine requirements are unclear. District of Columbia public schools continue to require immunization for virtual school attendance, but it is not clear how easily this can be enforced. To read about how other school districts have chosen to address – or not address – immunization requirements for school, visit the the Immunization Action Coalition’s Repository of Resources for Maintaining Immunization during the COVID-19 Pandemic. The repository links to international, national, and state-level policies and guidance and advocacy materials, including talking points, webinars, press releases, media articles from around the United States and social media posts, as well as telehealth resources.
Get some inspiration to talk about vaccination
Need a little inspiration for talking to parents about vaccines? Check out the CDC’s #HowIRecommend video series. These are short videos, most under a minute in length, that explain the importance of vaccination, how to effectively address questions from parents about vaccine safety, and how clinicians routinely recommend same day vaccination to their patients. These videos are part of the CDC’s National Immunization Awareness Month (NIAM) toolkit for communication with health care professionals. A companion toolkit for communicating with parents and patients contains sample social media messages with graphics, along with educational resources to share with parents.
The “Comprehensive Vaccine Education Program – From Training to Practice,” a free online program offered by the Pediatric Infectious Diseases Society, takes a deeper dive into strategies to combat vaccine misinformation and address vaccine hesitancy. Available modules cover vaccine fundamentals, vaccine safety, clinical manifestations of vaccine-preventable diseases, and communication skills that lead to more effective conversations with patients and parents. The curriculum also includes the newest edition of The Vaccine Handbook app, a comprehensive source of practical information for vaccine providers.
Educate young children about vaccines
Don’t leave young children out of the conversation. Vax-Force is a children’s book that explores how vaccination works inside the human body. Dr. Vaxson the pediatrician explains how trusted doctors and scientists made Vicky the Vaccine. Her mission is to tell Willy the White Blood Cell and his Antibuddies how to find and fight bad-guy germs like measles, tetanus, and polio. The book was written by Kelsey Rowe, MD, while she was a medical student at Saint Louis University School of Medicine. Dr. Rowe, now a pediatric resident, notes, “In a world where anti-vaccination rhetoric threatens the health of our global community, this book’s mission is to teach children and adults alike that getting vaccinations is a safe, effective, and even exciting thing to do.” The book is available for purchase at https://www.vax-force.com/, and a small part of every sale is donated to Unicef USA.
Consider vaccination advocacy in your communities
Vaccinate Your Family, a national, nonprofit organization dedicated to protecting people of all ages from vaccine-preventable diseases, suggests that health care providers need to take an active role in raising immunization rates, not just in their own practices, but in their communities. One way to do this is to submit an opinion piece or letter to the editor to a local newspaper describing why it’s important for parents to make sure their child’s immunizations are current. Those who have never written an opinion-editorial should look at the guidance developed by Voices for Vaccines.
How are we doing?
Early data suggest a rebound in immunization rates in May and June, but that is unlikely to close the gap created by disruptions in health care delivery earlier in the year. Collectively, we need to set ambitious goals. Are we just trying to reach prepandemic immunization levels? In Kentucky, where I practice, only 71% of kids aged 19-45 months had received all doses of seven routinely recommended vaccines (≥4 DTaP doses, ≥3 polio doses, ≥1 MMR dose, Hib full series, ≥3 HepB doses, ≥1 varicella dose, and ≥4 PCV doses) based on 2017 National Immunization Survey data. The Healthy People 2020 target goal is 80%. Only 55% of Kentucky girls aged 13-17 years received at least one dose of HPV vaccine, and rates in boys were even lower. Flu vaccine coverage in children 6 months to 17 years also was 55%. The status quo sets the bar too low. To see how your state is doing, check out the interactive map developed by the American Academy of Pediatrics.
Are we attempting to avoid disaster or can we seize the opportunity to protect more children than ever from vaccine-preventable diseases? The latter would really be something to celebrate.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She said she had no relevant financial disclosures. Email her at [email protected].
Back to school: How pediatricians can help LGBTQ youth
September every year means one thing to students across the country: Summer break is over, and it is time to go back to school. For LGBTQ youth, this can be both a blessing and a curse. Schools can be a refuge from being stuck at home with unsupportive family, but it also can mean returning to hallways full of harassment from other students and/or staff. Groups such as a gender-sexuality alliance (GSA) or a chapter of the Gay, Lesbian, and Straight Education Network (GLSEN) can provide a safe space for these students at school. Pediatricians can play an important role in ensuring that their patients know about access to these resources.
Gender-sexuality alliances, or gay-straight alliances as they have been more commonly known, have been around since the late 1980s. The first one was founded at Concord Academy in Massachusetts in 1988 by a straight student who was upset at how her gay classmates were being treated. Today’s GSAs continue this mission to create a welcoming environment for students of all gender identities and sexual orientations to gather, increase awareness on their campus of LGBTQ issues, and make the school environment safer for all students. According to the GSA network, there are over 4,000 active GSAs today in the United States located in 40 states.1
GLSEN was founded in 1990 initially as a network of gay and lesbian educators who wanted to create safer spaces in schools for LGBTQ students. Over the last 30 years, GLSEN continues to support this mission but has expanded into research and advocacy as well. There are currently 43 chapters of GLSEN in 30 states.2 GLSEN sponsors a number of national events throughout the year to raise awareness of LGBTQ issues in schools, including No Name Calling Week and the Day of Silence. Many chapters provide mentoring to local GSAs and volunteering as a mentor can be a great way for pediatricians to become involved in their local schools.
You may be asking yourself, why are GSAs important? According to GLSEN’s 2017 National School Climate Survey, nearly 35% of LGBTQ students missed at least 1 day of school in the previous month because of feeling unsafe, and nearly 57% of students reported hearing homophobic remarks from teachers and staff at their school.3 Around 10% of LGBTQ students reported being physically assaulted based on their sexual orientation and/or gender identity. Those LGBTQ students who experienced discrimination based on their sexual orientation and/or gender identity were more likely to have lower grade point averages and were more likely to be disciplined than those students who had not experienced discrimination.3 The cumulative effect of these negative experiences at school lead a sizable portion of affected students to drop out of school and possibly not pursue postsecondary education. This then leads to decreased job opportunities or career advancement, which could then lead to unemployment or low-wage jobs. Creating safe spaces for education to take place can have a lasting effect on the lives of LGBTQ students.
The 53% of students who reported having a GSA at their school in the National School Climate survey were less likely to report hearing negative comments about LGBTQ students, were less likely to miss school, experienced lower levels of victimization, and reported higher levels of supportive teachers and staff. All of these factors taken together ensure that LGBTQ students are more likely to complete their high school education. Russell B. Toomey, PhD, and colleagues were able to show that LGBTQ students with a perceived effective GSA were two times more likely than those without an effective GSA to attain a college education.4 Research also has shown that the presence of a GSA can have a beneficial impact on reducing bullying in general for all students, whether they identify as LGBTQ or not.5
What active steps can a pediatrician take to support their LGBTQ students? First, If the families run into trouble from the school, have your social workers help them connect with legal resources, as many court cases have established precedent that public schools cannot have a blanket ban on GSAs solely because they focus on LGBTQ issues. Second, if your patient has a GSA at their school and seems to be struggling with his/her sexual orientation and/or gender identity, encourage that student to consider attending their GSA so that they are able to spend time with other students like themselves. Third, as many schools will be starting virtually this year, you can provide your LGBTQ patients with a list of local online groups that students can participate in virtually if their school’s GSA is not meeting (see my LGBTQ Youth Consult column entitled, “Resources for LGBTQ youth during challenging times” at mdedge.com/pediatrics for a few ideas).* Lastly, be an active advocate in your own local school district for the inclusion of comprehensive nondiscrimination policies and the presence of GSAs for students. These small steps can go a long way to helping your LGBTQ patients thrive and succeed in school.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. Dr. Cooper has no relevant financial disclosures. Email him at [email protected].
References
1. gsanetwork.org/mission-vision-history/.
2. www.glsen.org/find_chapter?field_chapter_state_target_id=All.
3. live-glsen-website.pantheonsite.io/sites/default/files/2019-10/GLSEN-2017-National-School-Climate-Survey-NSCS-Full-Report.pdf.
4. Appl Dev Sci. 2011 Nov 7;15(4):175-85.
5.www.usnews.com/news/articles/2016-08-04/gay-straight-alliances-in-schools-pay-off-for-all-students-study-finds.
*This article was updated 8/17/2020.
September every year means one thing to students across the country: Summer break is over, and it is time to go back to school. For LGBTQ youth, this can be both a blessing and a curse. Schools can be a refuge from being stuck at home with unsupportive family, but it also can mean returning to hallways full of harassment from other students and/or staff. Groups such as a gender-sexuality alliance (GSA) or a chapter of the Gay, Lesbian, and Straight Education Network (GLSEN) can provide a safe space for these students at school. Pediatricians can play an important role in ensuring that their patients know about access to these resources.
Gender-sexuality alliances, or gay-straight alliances as they have been more commonly known, have been around since the late 1980s. The first one was founded at Concord Academy in Massachusetts in 1988 by a straight student who was upset at how her gay classmates were being treated. Today’s GSAs continue this mission to create a welcoming environment for students of all gender identities and sexual orientations to gather, increase awareness on their campus of LGBTQ issues, and make the school environment safer for all students. According to the GSA network, there are over 4,000 active GSAs today in the United States located in 40 states.1
GLSEN was founded in 1990 initially as a network of gay and lesbian educators who wanted to create safer spaces in schools for LGBTQ students. Over the last 30 years, GLSEN continues to support this mission but has expanded into research and advocacy as well. There are currently 43 chapters of GLSEN in 30 states.2 GLSEN sponsors a number of national events throughout the year to raise awareness of LGBTQ issues in schools, including No Name Calling Week and the Day of Silence. Many chapters provide mentoring to local GSAs and volunteering as a mentor can be a great way for pediatricians to become involved in their local schools.
You may be asking yourself, why are GSAs important? According to GLSEN’s 2017 National School Climate Survey, nearly 35% of LGBTQ students missed at least 1 day of school in the previous month because of feeling unsafe, and nearly 57% of students reported hearing homophobic remarks from teachers and staff at their school.3 Around 10% of LGBTQ students reported being physically assaulted based on their sexual orientation and/or gender identity. Those LGBTQ students who experienced discrimination based on their sexual orientation and/or gender identity were more likely to have lower grade point averages and were more likely to be disciplined than those students who had not experienced discrimination.3 The cumulative effect of these negative experiences at school lead a sizable portion of affected students to drop out of school and possibly not pursue postsecondary education. This then leads to decreased job opportunities or career advancement, which could then lead to unemployment or low-wage jobs. Creating safe spaces for education to take place can have a lasting effect on the lives of LGBTQ students.
The 53% of students who reported having a GSA at their school in the National School Climate survey were less likely to report hearing negative comments about LGBTQ students, were less likely to miss school, experienced lower levels of victimization, and reported higher levels of supportive teachers and staff. All of these factors taken together ensure that LGBTQ students are more likely to complete their high school education. Russell B. Toomey, PhD, and colleagues were able to show that LGBTQ students with a perceived effective GSA were two times more likely than those without an effective GSA to attain a college education.4 Research also has shown that the presence of a GSA can have a beneficial impact on reducing bullying in general for all students, whether they identify as LGBTQ or not.5
What active steps can a pediatrician take to support their LGBTQ students? First, If the families run into trouble from the school, have your social workers help them connect with legal resources, as many court cases have established precedent that public schools cannot have a blanket ban on GSAs solely because they focus on LGBTQ issues. Second, if your patient has a GSA at their school and seems to be struggling with his/her sexual orientation and/or gender identity, encourage that student to consider attending their GSA so that they are able to spend time with other students like themselves. Third, as many schools will be starting virtually this year, you can provide your LGBTQ patients with a list of local online groups that students can participate in virtually if their school’s GSA is not meeting (see my LGBTQ Youth Consult column entitled, “Resources for LGBTQ youth during challenging times” at mdedge.com/pediatrics for a few ideas).* Lastly, be an active advocate in your own local school district for the inclusion of comprehensive nondiscrimination policies and the presence of GSAs for students. These small steps can go a long way to helping your LGBTQ patients thrive and succeed in school.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. Dr. Cooper has no relevant financial disclosures. Email him at [email protected].
References
1. gsanetwork.org/mission-vision-history/.
2. www.glsen.org/find_chapter?field_chapter_state_target_id=All.
3. live-glsen-website.pantheonsite.io/sites/default/files/2019-10/GLSEN-2017-National-School-Climate-Survey-NSCS-Full-Report.pdf.
4. Appl Dev Sci. 2011 Nov 7;15(4):175-85.
5.www.usnews.com/news/articles/2016-08-04/gay-straight-alliances-in-schools-pay-off-for-all-students-study-finds.
*This article was updated 8/17/2020.
September every year means one thing to students across the country: Summer break is over, and it is time to go back to school. For LGBTQ youth, this can be both a blessing and a curse. Schools can be a refuge from being stuck at home with unsupportive family, but it also can mean returning to hallways full of harassment from other students and/or staff. Groups such as a gender-sexuality alliance (GSA) or a chapter of the Gay, Lesbian, and Straight Education Network (GLSEN) can provide a safe space for these students at school. Pediatricians can play an important role in ensuring that their patients know about access to these resources.
Gender-sexuality alliances, or gay-straight alliances as they have been more commonly known, have been around since the late 1980s. The first one was founded at Concord Academy in Massachusetts in 1988 by a straight student who was upset at how her gay classmates were being treated. Today’s GSAs continue this mission to create a welcoming environment for students of all gender identities and sexual orientations to gather, increase awareness on their campus of LGBTQ issues, and make the school environment safer for all students. According to the GSA network, there are over 4,000 active GSAs today in the United States located in 40 states.1
GLSEN was founded in 1990 initially as a network of gay and lesbian educators who wanted to create safer spaces in schools for LGBTQ students. Over the last 30 years, GLSEN continues to support this mission but has expanded into research and advocacy as well. There are currently 43 chapters of GLSEN in 30 states.2 GLSEN sponsors a number of national events throughout the year to raise awareness of LGBTQ issues in schools, including No Name Calling Week and the Day of Silence. Many chapters provide mentoring to local GSAs and volunteering as a mentor can be a great way for pediatricians to become involved in their local schools.
You may be asking yourself, why are GSAs important? According to GLSEN’s 2017 National School Climate Survey, nearly 35% of LGBTQ students missed at least 1 day of school in the previous month because of feeling unsafe, and nearly 57% of students reported hearing homophobic remarks from teachers and staff at their school.3 Around 10% of LGBTQ students reported being physically assaulted based on their sexual orientation and/or gender identity. Those LGBTQ students who experienced discrimination based on their sexual orientation and/or gender identity were more likely to have lower grade point averages and were more likely to be disciplined than those students who had not experienced discrimination.3 The cumulative effect of these negative experiences at school lead a sizable portion of affected students to drop out of school and possibly not pursue postsecondary education. This then leads to decreased job opportunities or career advancement, which could then lead to unemployment or low-wage jobs. Creating safe spaces for education to take place can have a lasting effect on the lives of LGBTQ students.
The 53% of students who reported having a GSA at their school in the National School Climate survey were less likely to report hearing negative comments about LGBTQ students, were less likely to miss school, experienced lower levels of victimization, and reported higher levels of supportive teachers and staff. All of these factors taken together ensure that LGBTQ students are more likely to complete their high school education. Russell B. Toomey, PhD, and colleagues were able to show that LGBTQ students with a perceived effective GSA were two times more likely than those without an effective GSA to attain a college education.4 Research also has shown that the presence of a GSA can have a beneficial impact on reducing bullying in general for all students, whether they identify as LGBTQ or not.5
What active steps can a pediatrician take to support their LGBTQ students? First, If the families run into trouble from the school, have your social workers help them connect with legal resources, as many court cases have established precedent that public schools cannot have a blanket ban on GSAs solely because they focus on LGBTQ issues. Second, if your patient has a GSA at their school and seems to be struggling with his/her sexual orientation and/or gender identity, encourage that student to consider attending their GSA so that they are able to spend time with other students like themselves. Third, as many schools will be starting virtually this year, you can provide your LGBTQ patients with a list of local online groups that students can participate in virtually if their school’s GSA is not meeting (see my LGBTQ Youth Consult column entitled, “Resources for LGBTQ youth during challenging times” at mdedge.com/pediatrics for a few ideas).* Lastly, be an active advocate in your own local school district for the inclusion of comprehensive nondiscrimination policies and the presence of GSAs for students. These small steps can go a long way to helping your LGBTQ patients thrive and succeed in school.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. Dr. Cooper has no relevant financial disclosures. Email him at [email protected].
References
1. gsanetwork.org/mission-vision-history/.
2. www.glsen.org/find_chapter?field_chapter_state_target_id=All.
3. live-glsen-website.pantheonsite.io/sites/default/files/2019-10/GLSEN-2017-National-School-Climate-Survey-NSCS-Full-Report.pdf.
4. Appl Dev Sci. 2011 Nov 7;15(4):175-85.
5.www.usnews.com/news/articles/2016-08-04/gay-straight-alliances-in-schools-pay-off-for-all-students-study-finds.
*This article was updated 8/17/2020.
Long-lasting COVID-19 symptoms: Patients want answers
Q&A with Dr. Sachin Gupta
For some patients, a bout of COVID-19 may not be over after hospital discharge, acute symptoms subside, or a couple of tests for SARS-CoV-2 come back negative. Those who have reached these milestones of conquering the disease may find that their recovery journey has only begun. Debilitating symptoms such as fatigue, headache, and dyspnea may linger for weeks or longer. Patients with persistent symptoms, often referred to as “long haulers” in reference to the duration of their recovery, are looking for answers about their condition and when their COVID-19 illness will finally resolve.
Long-haul patients organize
What started as an accumulation of anecdotal evidence in social media, blogs, and the mainstream press about slow recovery and long-lasting symptoms of COVID-19 is now the focus of clinical trials in the population of recovering patients. Projects such as the COVID Symptom Study, initiated by the Massachusetts General Hospital, Boston; the Harvard School of Public Health, Boston; King’s College London; and Stanford (Calif.) University, are collecting data on symptoms from millions of patients and will eventually contribute to a better understanding of prolonged recovery.
Patients looking for answers have created groups on social media such as Facebook to exchange information about their experiences (e.g., Survivor Corps, COVID-19 Support Group, COVID-19 Recovered Survivors). Recovering patients have created patient-led research organizations (Body Politic COVID-19 Support Group) to explore persistent symptoms and begin to create data for research.
Some data on lingering symptoms
A small study of 143 previously hospitalized, recovering patients in Italy found that 87.4% of the cohort had at least one persistent symptom 2 months or longer after initial onset and at more than a month after discharge. In this sample, only 5% had been intubated. (JAMA 2020 Jul 9. doi: 10.1001/jama.2020.12603).
One study found that even patients who have had relatively mild symptoms and were not hospitalized can have persistent symptoms. The Centers for Disease Control and Prevention conducted a survey of adults who tested positive for the positive reverse transcription–polymerase chain reaction test for SARS-CoV-2 and found that, among the 292 respondents, 35% were still feeling the impact of the disease 2-3 weeks after testing. Fatigue (71%), cough (61%), and headache (61%) were the most commonly reported symptoms. The survey found that delayed recovery was evident in nearly a quarter of 18- to 34-year-olds and in a third of 35- to 49-year-olds who were not sick enough to require hospitalization (MMWR. 2020 Jul 24. doi: 10.15585/mmwr.mm6930e1).
Sachin Gupta, MD, FCCP, ATSF, a pulmonologist and member of the CHEST Physician editorial advisory board, has treated patients with COVID-19 and shared some of his thoughts on the problem of prolonged symptoms of COVID-19.
Q: Should clinicians expect to see COVID-19 patients who have symptoms persisting weeks after they are diagnosed?
Dr. Gupta: I think clinicians, especially in primary care, are already seeing many patients with lingering symptoms, both respiratory and nonrespiratory related, and debility. A few patients here in the San Francisco Bay Area that I have spoken with 4-6 weeks out from their acute illness have complained of persisting, though improving, fatigue and cough. Early studies are confirming this as a topical issue. There may be other long-lasting sequelae of COVID-19 beyond the common mild lingering symptoms. It will also be important to consider (and get more data on) to what degree asymptomatic patients develop some degree of mild inflammatory and subsequent fibrotic changes in organs like the lungs and heart
Q: How does the recovery phase of COVID-19 compare with recovery from severe influenza or ARDS?
Dr. Gupta: Most prior influenza and acute respiratory distress syndrome (ARDS) studies have provided initial follow-up at 3 months and beyond, so technically speaking, it is a little difficult to compare the symptomatology patterns in the JAMA study of 2 months on follow-up. Nevertheless, the key takeaway is that, even though few patients in the study had ARDS requiring intubation (severe disease), many patients with milder disease had significant lingering symptoms (55% with three or more symptoms) at 2 months.
This fits logically with the premise, which we have some limited data on with ARDS (N Engl J Med. 2003;348:683-93. doi: 10.1056/NEJMoa022450) and severe influenza infection survivors (Nature Sci Rep. 2017;7:17275. doi: 10.1038/s41598-017-17497-6) that varying degrees of the inflammation cascade triggered by certain viruses can lead to changes in important patient-reported outcomes, and objective measures such as pulmonary function over the long term.
Q: What can you do for patients with lingering symptoms of COVID-19 or what can you tell them about their symptoms?
Dr. Gupta: For many patients, there is fear, given the novel nature of the virus/pandemic, that their symptoms may persist long term. Acknowledgment of their symptoms is validating and important for us to recognize as we learn more about the virus. As we are finding, many patients are going online to find answers, after sometimes feeling rushed or dismissed initially in the clinical setting.
In my experience, the bar is fairly high for most patients to reach out to their physicians with complaints of lingering symptoms after acute infection. For the ones who do reach out, they tend to have either a greater constellation of symptoms or higher severity of one or two key symptoms. After assessing and, when appropriate, ruling out secondary infections or newly developed conditions, I shift toward symptom management. I encourage such patients to build up slowly. I suggest they work first on their activities of daily living (bathing, grooming), then their instrumental activities of daily living (cooking, cleaning, checking the mail), and then to engage, based on their tolerance of symptoms, to light purposeful exercise. There are many online resources for at-home exercise activities that I recommend to patients who are more debilitated; some larger centers are beginning to offer some forms of telepulmonary rehab.
Based on what we know about other causes of viral pneumonitis and ARDS, I ask such symptomatic patients to expect a slow, gradual, and in most cases a complete recovery, and depending on the individual case, I recommend pulmonary function tests and imaging that may be helpful to track that progress.
I remind myself, and patients, that our understanding may change as we learn more over time. Checking in at set intervals, even if not in person but through a phone call, can go a long way in a setting where we do not have a specific therapy, other than gradual exercise training, to help these patients recover faster. Reassurance and encouragement are vital for patients who are struggling with the lingering burden of disease and who may find it difficult to return to work or function as usual at home. The final point is to be mindful of our patient’s mental health and, where our reassurance is not enough, to consider appropriate mental health referrals.
Q: Can you handle this kind of problem with telemedicine or which patients with lingering symptoms need to come into the office – or failing that, the ED?
Dr. Gupta: Telemedicine in the outpatient setting provides a helpful tool to assess and manage patients, in my experience, with limited and straightforward complaints. Its scope is limited diagnostically (assessing symptoms and signs) as is its reach (ability to connect with elderly, disabled, or patients without/limited telemedicine access). In many instances, telemedicine limits our ability to connect with patients emotionally and build trust. Many patients who have gone through the acute illness that we see in pulmonary clinic on follow-up are older in age, and for many, video visits are not a practical solution. Telemedicine visits can sometimes present challenges for me as well in terms of thoroughly conveying lifestyle and symptom management strategies. Health literacy is typically easier to gauge and address in person.
For patients with any degree of enduring dyspnea, more so in the acute phase, I recommend home pulse oximetry for monitoring their oxygen saturation if it is financially and technically feasible for them to obtain one. Sending a patient to the ED is an option of last resort, but one that is necessary in some cases. I expect patients with lingering symptoms to tell me that symptoms may be persisting, hopefully gradually improving, and not getting worse. If post–COVID-19 symptoms such as fever, dyspnea, fatigue, or lightheadedness are new or worsening, particularly rapidly, the safest and best option I advise patients is to go to the ED for further assessment and testing. Postviral bacterial pneumonia is something we should consider, and there is some potential for aspergillosis as well.
Q: Do you have any concerns about patients with asthma, chronic obstructive pulmonary disease, or other pulmonary issues having lingering symptoms that may mask exacerbations or may cause exacerbation of their disease?
Dr. Gupta: So far, patients with chronic lung conditions do not appear to have not been disproportionately affected by the pandemic in terms of absolute numbers or percentage wise compared to the general public. I think that sheltering in place has been readily followed by many of these patients, and in addition, I assume better adherence to their maintenance therapies has likely helped. The very few cases of patients with underlying chronic obstructive pulmonary disease and interstitial lung disease that I have seen have fared very poorly when they were diagnosed with COVID-19 in the hospital. There are emerging data about short-term outcomes from severe COVID-19 infection in patients with interstitial lung disease in Europe (medRxiv. 2020 Jul 17. doi: 10.1101/2020.07.15.20152967), and from physicians treating pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension (Ann Am Thorac Soc. 2020 Jul 29. doi: 10.1513/AnnalsATS.202005-521OC). But so far, little has been published on the outcomes of mild disease in these patients with chronic lung disease.
Q: It’s still early days to know the significance of lingering symptoms. But at what point do you begin to consider the possibility of some kind of relapse? And what is your next move if the symptoms get worse?
Dr. Gupta: COVID-19 recurrence, whether because of reinfection or relapse, is a potential concern but not one that is very commonly seen so far in my purview. Generally, symptoms of post–COVID-19 infection that are lingering trend toward getting better, even if slowly. If post–COVID-19 infection symptoms are progressing (particularly if rapidly), that would be a strong indication to evaluate that patient in the ED (less likely in clinic), reswab them for SARS-CoV-2, and obtain further testing such as blood work and imaging. A significant challenge from a research perspective will be determining if coinfection with another virus is playing a role as we move closer to the fall season.
Q&A with Dr. Sachin Gupta
Q&A with Dr. Sachin Gupta
For some patients, a bout of COVID-19 may not be over after hospital discharge, acute symptoms subside, or a couple of tests for SARS-CoV-2 come back negative. Those who have reached these milestones of conquering the disease may find that their recovery journey has only begun. Debilitating symptoms such as fatigue, headache, and dyspnea may linger for weeks or longer. Patients with persistent symptoms, often referred to as “long haulers” in reference to the duration of their recovery, are looking for answers about their condition and when their COVID-19 illness will finally resolve.
Long-haul patients organize
What started as an accumulation of anecdotal evidence in social media, blogs, and the mainstream press about slow recovery and long-lasting symptoms of COVID-19 is now the focus of clinical trials in the population of recovering patients. Projects such as the COVID Symptom Study, initiated by the Massachusetts General Hospital, Boston; the Harvard School of Public Health, Boston; King’s College London; and Stanford (Calif.) University, are collecting data on symptoms from millions of patients and will eventually contribute to a better understanding of prolonged recovery.
Patients looking for answers have created groups on social media such as Facebook to exchange information about their experiences (e.g., Survivor Corps, COVID-19 Support Group, COVID-19 Recovered Survivors). Recovering patients have created patient-led research organizations (Body Politic COVID-19 Support Group) to explore persistent symptoms and begin to create data for research.
Some data on lingering symptoms
A small study of 143 previously hospitalized, recovering patients in Italy found that 87.4% of the cohort had at least one persistent symptom 2 months or longer after initial onset and at more than a month after discharge. In this sample, only 5% had been intubated. (JAMA 2020 Jul 9. doi: 10.1001/jama.2020.12603).
One study found that even patients who have had relatively mild symptoms and were not hospitalized can have persistent symptoms. The Centers for Disease Control and Prevention conducted a survey of adults who tested positive for the positive reverse transcription–polymerase chain reaction test for SARS-CoV-2 and found that, among the 292 respondents, 35% were still feeling the impact of the disease 2-3 weeks after testing. Fatigue (71%), cough (61%), and headache (61%) were the most commonly reported symptoms. The survey found that delayed recovery was evident in nearly a quarter of 18- to 34-year-olds and in a third of 35- to 49-year-olds who were not sick enough to require hospitalization (MMWR. 2020 Jul 24. doi: 10.15585/mmwr.mm6930e1).
Sachin Gupta, MD, FCCP, ATSF, a pulmonologist and member of the CHEST Physician editorial advisory board, has treated patients with COVID-19 and shared some of his thoughts on the problem of prolonged symptoms of COVID-19.
Q: Should clinicians expect to see COVID-19 patients who have symptoms persisting weeks after they are diagnosed?
Dr. Gupta: I think clinicians, especially in primary care, are already seeing many patients with lingering symptoms, both respiratory and nonrespiratory related, and debility. A few patients here in the San Francisco Bay Area that I have spoken with 4-6 weeks out from their acute illness have complained of persisting, though improving, fatigue and cough. Early studies are confirming this as a topical issue. There may be other long-lasting sequelae of COVID-19 beyond the common mild lingering symptoms. It will also be important to consider (and get more data on) to what degree asymptomatic patients develop some degree of mild inflammatory and subsequent fibrotic changes in organs like the lungs and heart
Q: How does the recovery phase of COVID-19 compare with recovery from severe influenza or ARDS?
Dr. Gupta: Most prior influenza and acute respiratory distress syndrome (ARDS) studies have provided initial follow-up at 3 months and beyond, so technically speaking, it is a little difficult to compare the symptomatology patterns in the JAMA study of 2 months on follow-up. Nevertheless, the key takeaway is that, even though few patients in the study had ARDS requiring intubation (severe disease), many patients with milder disease had significant lingering symptoms (55% with three or more symptoms) at 2 months.
This fits logically with the premise, which we have some limited data on with ARDS (N Engl J Med. 2003;348:683-93. doi: 10.1056/NEJMoa022450) and severe influenza infection survivors (Nature Sci Rep. 2017;7:17275. doi: 10.1038/s41598-017-17497-6) that varying degrees of the inflammation cascade triggered by certain viruses can lead to changes in important patient-reported outcomes, and objective measures such as pulmonary function over the long term.
Q: What can you do for patients with lingering symptoms of COVID-19 or what can you tell them about their symptoms?
Dr. Gupta: For many patients, there is fear, given the novel nature of the virus/pandemic, that their symptoms may persist long term. Acknowledgment of their symptoms is validating and important for us to recognize as we learn more about the virus. As we are finding, many patients are going online to find answers, after sometimes feeling rushed or dismissed initially in the clinical setting.
In my experience, the bar is fairly high for most patients to reach out to their physicians with complaints of lingering symptoms after acute infection. For the ones who do reach out, they tend to have either a greater constellation of symptoms or higher severity of one or two key symptoms. After assessing and, when appropriate, ruling out secondary infections or newly developed conditions, I shift toward symptom management. I encourage such patients to build up slowly. I suggest they work first on their activities of daily living (bathing, grooming), then their instrumental activities of daily living (cooking, cleaning, checking the mail), and then to engage, based on their tolerance of symptoms, to light purposeful exercise. There are many online resources for at-home exercise activities that I recommend to patients who are more debilitated; some larger centers are beginning to offer some forms of telepulmonary rehab.
Based on what we know about other causes of viral pneumonitis and ARDS, I ask such symptomatic patients to expect a slow, gradual, and in most cases a complete recovery, and depending on the individual case, I recommend pulmonary function tests and imaging that may be helpful to track that progress.
I remind myself, and patients, that our understanding may change as we learn more over time. Checking in at set intervals, even if not in person but through a phone call, can go a long way in a setting where we do not have a specific therapy, other than gradual exercise training, to help these patients recover faster. Reassurance and encouragement are vital for patients who are struggling with the lingering burden of disease and who may find it difficult to return to work or function as usual at home. The final point is to be mindful of our patient’s mental health and, where our reassurance is not enough, to consider appropriate mental health referrals.
Q: Can you handle this kind of problem with telemedicine or which patients with lingering symptoms need to come into the office – or failing that, the ED?
Dr. Gupta: Telemedicine in the outpatient setting provides a helpful tool to assess and manage patients, in my experience, with limited and straightforward complaints. Its scope is limited diagnostically (assessing symptoms and signs) as is its reach (ability to connect with elderly, disabled, or patients without/limited telemedicine access). In many instances, telemedicine limits our ability to connect with patients emotionally and build trust. Many patients who have gone through the acute illness that we see in pulmonary clinic on follow-up are older in age, and for many, video visits are not a practical solution. Telemedicine visits can sometimes present challenges for me as well in terms of thoroughly conveying lifestyle and symptom management strategies. Health literacy is typically easier to gauge and address in person.
For patients with any degree of enduring dyspnea, more so in the acute phase, I recommend home pulse oximetry for monitoring their oxygen saturation if it is financially and technically feasible for them to obtain one. Sending a patient to the ED is an option of last resort, but one that is necessary in some cases. I expect patients with lingering symptoms to tell me that symptoms may be persisting, hopefully gradually improving, and not getting worse. If post–COVID-19 symptoms such as fever, dyspnea, fatigue, or lightheadedness are new or worsening, particularly rapidly, the safest and best option I advise patients is to go to the ED for further assessment and testing. Postviral bacterial pneumonia is something we should consider, and there is some potential for aspergillosis as well.
Q: Do you have any concerns about patients with asthma, chronic obstructive pulmonary disease, or other pulmonary issues having lingering symptoms that may mask exacerbations or may cause exacerbation of their disease?
Dr. Gupta: So far, patients with chronic lung conditions do not appear to have not been disproportionately affected by the pandemic in terms of absolute numbers or percentage wise compared to the general public. I think that sheltering in place has been readily followed by many of these patients, and in addition, I assume better adherence to their maintenance therapies has likely helped. The very few cases of patients with underlying chronic obstructive pulmonary disease and interstitial lung disease that I have seen have fared very poorly when they were diagnosed with COVID-19 in the hospital. There are emerging data about short-term outcomes from severe COVID-19 infection in patients with interstitial lung disease in Europe (medRxiv. 2020 Jul 17. doi: 10.1101/2020.07.15.20152967), and from physicians treating pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension (Ann Am Thorac Soc. 2020 Jul 29. doi: 10.1513/AnnalsATS.202005-521OC). But so far, little has been published on the outcomes of mild disease in these patients with chronic lung disease.
Q: It’s still early days to know the significance of lingering symptoms. But at what point do you begin to consider the possibility of some kind of relapse? And what is your next move if the symptoms get worse?
Dr. Gupta: COVID-19 recurrence, whether because of reinfection or relapse, is a potential concern but not one that is very commonly seen so far in my purview. Generally, symptoms of post–COVID-19 infection that are lingering trend toward getting better, even if slowly. If post–COVID-19 infection symptoms are progressing (particularly if rapidly), that would be a strong indication to evaluate that patient in the ED (less likely in clinic), reswab them for SARS-CoV-2, and obtain further testing such as blood work and imaging. A significant challenge from a research perspective will be determining if coinfection with another virus is playing a role as we move closer to the fall season.
For some patients, a bout of COVID-19 may not be over after hospital discharge, acute symptoms subside, or a couple of tests for SARS-CoV-2 come back negative. Those who have reached these milestones of conquering the disease may find that their recovery journey has only begun. Debilitating symptoms such as fatigue, headache, and dyspnea may linger for weeks or longer. Patients with persistent symptoms, often referred to as “long haulers” in reference to the duration of their recovery, are looking for answers about their condition and when their COVID-19 illness will finally resolve.
Long-haul patients organize
What started as an accumulation of anecdotal evidence in social media, blogs, and the mainstream press about slow recovery and long-lasting symptoms of COVID-19 is now the focus of clinical trials in the population of recovering patients. Projects such as the COVID Symptom Study, initiated by the Massachusetts General Hospital, Boston; the Harvard School of Public Health, Boston; King’s College London; and Stanford (Calif.) University, are collecting data on symptoms from millions of patients and will eventually contribute to a better understanding of prolonged recovery.
Patients looking for answers have created groups on social media such as Facebook to exchange information about their experiences (e.g., Survivor Corps, COVID-19 Support Group, COVID-19 Recovered Survivors). Recovering patients have created patient-led research organizations (Body Politic COVID-19 Support Group) to explore persistent symptoms and begin to create data for research.
Some data on lingering symptoms
A small study of 143 previously hospitalized, recovering patients in Italy found that 87.4% of the cohort had at least one persistent symptom 2 months or longer after initial onset and at more than a month after discharge. In this sample, only 5% had been intubated. (JAMA 2020 Jul 9. doi: 10.1001/jama.2020.12603).
One study found that even patients who have had relatively mild symptoms and were not hospitalized can have persistent symptoms. The Centers for Disease Control and Prevention conducted a survey of adults who tested positive for the positive reverse transcription–polymerase chain reaction test for SARS-CoV-2 and found that, among the 292 respondents, 35% were still feeling the impact of the disease 2-3 weeks after testing. Fatigue (71%), cough (61%), and headache (61%) were the most commonly reported symptoms. The survey found that delayed recovery was evident in nearly a quarter of 18- to 34-year-olds and in a third of 35- to 49-year-olds who were not sick enough to require hospitalization (MMWR. 2020 Jul 24. doi: 10.15585/mmwr.mm6930e1).
Sachin Gupta, MD, FCCP, ATSF, a pulmonologist and member of the CHEST Physician editorial advisory board, has treated patients with COVID-19 and shared some of his thoughts on the problem of prolonged symptoms of COVID-19.
Q: Should clinicians expect to see COVID-19 patients who have symptoms persisting weeks after they are diagnosed?
Dr. Gupta: I think clinicians, especially in primary care, are already seeing many patients with lingering symptoms, both respiratory and nonrespiratory related, and debility. A few patients here in the San Francisco Bay Area that I have spoken with 4-6 weeks out from their acute illness have complained of persisting, though improving, fatigue and cough. Early studies are confirming this as a topical issue. There may be other long-lasting sequelae of COVID-19 beyond the common mild lingering symptoms. It will also be important to consider (and get more data on) to what degree asymptomatic patients develop some degree of mild inflammatory and subsequent fibrotic changes in organs like the lungs and heart
Q: How does the recovery phase of COVID-19 compare with recovery from severe influenza or ARDS?
Dr. Gupta: Most prior influenza and acute respiratory distress syndrome (ARDS) studies have provided initial follow-up at 3 months and beyond, so technically speaking, it is a little difficult to compare the symptomatology patterns in the JAMA study of 2 months on follow-up. Nevertheless, the key takeaway is that, even though few patients in the study had ARDS requiring intubation (severe disease), many patients with milder disease had significant lingering symptoms (55% with three or more symptoms) at 2 months.
This fits logically with the premise, which we have some limited data on with ARDS (N Engl J Med. 2003;348:683-93. doi: 10.1056/NEJMoa022450) and severe influenza infection survivors (Nature Sci Rep. 2017;7:17275. doi: 10.1038/s41598-017-17497-6) that varying degrees of the inflammation cascade triggered by certain viruses can lead to changes in important patient-reported outcomes, and objective measures such as pulmonary function over the long term.
Q: What can you do for patients with lingering symptoms of COVID-19 or what can you tell them about their symptoms?
Dr. Gupta: For many patients, there is fear, given the novel nature of the virus/pandemic, that their symptoms may persist long term. Acknowledgment of their symptoms is validating and important for us to recognize as we learn more about the virus. As we are finding, many patients are going online to find answers, after sometimes feeling rushed or dismissed initially in the clinical setting.
In my experience, the bar is fairly high for most patients to reach out to their physicians with complaints of lingering symptoms after acute infection. For the ones who do reach out, they tend to have either a greater constellation of symptoms or higher severity of one or two key symptoms. After assessing and, when appropriate, ruling out secondary infections or newly developed conditions, I shift toward symptom management. I encourage such patients to build up slowly. I suggest they work first on their activities of daily living (bathing, grooming), then their instrumental activities of daily living (cooking, cleaning, checking the mail), and then to engage, based on their tolerance of symptoms, to light purposeful exercise. There are many online resources for at-home exercise activities that I recommend to patients who are more debilitated; some larger centers are beginning to offer some forms of telepulmonary rehab.
Based on what we know about other causes of viral pneumonitis and ARDS, I ask such symptomatic patients to expect a slow, gradual, and in most cases a complete recovery, and depending on the individual case, I recommend pulmonary function tests and imaging that may be helpful to track that progress.
I remind myself, and patients, that our understanding may change as we learn more over time. Checking in at set intervals, even if not in person but through a phone call, can go a long way in a setting where we do not have a specific therapy, other than gradual exercise training, to help these patients recover faster. Reassurance and encouragement are vital for patients who are struggling with the lingering burden of disease and who may find it difficult to return to work or function as usual at home. The final point is to be mindful of our patient’s mental health and, where our reassurance is not enough, to consider appropriate mental health referrals.
Q: Can you handle this kind of problem with telemedicine or which patients with lingering symptoms need to come into the office – or failing that, the ED?
Dr. Gupta: Telemedicine in the outpatient setting provides a helpful tool to assess and manage patients, in my experience, with limited and straightforward complaints. Its scope is limited diagnostically (assessing symptoms and signs) as is its reach (ability to connect with elderly, disabled, or patients without/limited telemedicine access). In many instances, telemedicine limits our ability to connect with patients emotionally and build trust. Many patients who have gone through the acute illness that we see in pulmonary clinic on follow-up are older in age, and for many, video visits are not a practical solution. Telemedicine visits can sometimes present challenges for me as well in terms of thoroughly conveying lifestyle and symptom management strategies. Health literacy is typically easier to gauge and address in person.
For patients with any degree of enduring dyspnea, more so in the acute phase, I recommend home pulse oximetry for monitoring their oxygen saturation if it is financially and technically feasible for them to obtain one. Sending a patient to the ED is an option of last resort, but one that is necessary in some cases. I expect patients with lingering symptoms to tell me that symptoms may be persisting, hopefully gradually improving, and not getting worse. If post–COVID-19 symptoms such as fever, dyspnea, fatigue, or lightheadedness are new or worsening, particularly rapidly, the safest and best option I advise patients is to go to the ED for further assessment and testing. Postviral bacterial pneumonia is something we should consider, and there is some potential for aspergillosis as well.
Q: Do you have any concerns about patients with asthma, chronic obstructive pulmonary disease, or other pulmonary issues having lingering symptoms that may mask exacerbations or may cause exacerbation of their disease?
Dr. Gupta: So far, patients with chronic lung conditions do not appear to have not been disproportionately affected by the pandemic in terms of absolute numbers or percentage wise compared to the general public. I think that sheltering in place has been readily followed by many of these patients, and in addition, I assume better adherence to their maintenance therapies has likely helped. The very few cases of patients with underlying chronic obstructive pulmonary disease and interstitial lung disease that I have seen have fared very poorly when they were diagnosed with COVID-19 in the hospital. There are emerging data about short-term outcomes from severe COVID-19 infection in patients with interstitial lung disease in Europe (medRxiv. 2020 Jul 17. doi: 10.1101/2020.07.15.20152967), and from physicians treating pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension (Ann Am Thorac Soc. 2020 Jul 29. doi: 10.1513/AnnalsATS.202005-521OC). But so far, little has been published on the outcomes of mild disease in these patients with chronic lung disease.
Q: It’s still early days to know the significance of lingering symptoms. But at what point do you begin to consider the possibility of some kind of relapse? And what is your next move if the symptoms get worse?
Dr. Gupta: COVID-19 recurrence, whether because of reinfection or relapse, is a potential concern but not one that is very commonly seen so far in my purview. Generally, symptoms of post–COVID-19 infection that are lingering trend toward getting better, even if slowly. If post–COVID-19 infection symptoms are progressing (particularly if rapidly), that would be a strong indication to evaluate that patient in the ED (less likely in clinic), reswab them for SARS-CoV-2, and obtain further testing such as blood work and imaging. A significant challenge from a research perspective will be determining if coinfection with another virus is playing a role as we move closer to the fall season.
iResident: Virtual care on hospital medicine teaching services during a pandemic
At the start of each shift on his clinical service with rotating internal medicine residents, Benji Mathews, MD, SFHM, now adds a few components to his usual preparation. First, visiting the Minnesota Department of Health and various organizational websites to review the latest COVID-19 updates and guidelines. Next comes checking to see where he needs to pick up the surgical mask and eye protection that he will need to wear through the day. Last, he evaluates which of his patients are in telemedicine-equipped rooms; this last change has fast become a crucial part of working with his resident learners during a pandemic.
During the COVID-19 pandemic, residents and residency programs find themselves in a unique situation. Balancing the educational needs of a training program with the safety of trainees is a challenging task, specifically when taking care of patients who are COVID-19 positive or patients under investigation (PUI). One increasingly available tool that can help protect trainees while continuing to prioritize patient care and medical education is the use of telemedicine for virtual rounding. For our internal medicine residents through the University of Minnesota Internal Medicine Residency program rotating at Regions Hospital in Saint Paul, Minn., we have used video visits to continue our mandate as both health care and education professionals.
Virtual care decision tree
Virtual care can mitigate exposure risk, minimize use of personal protective equipment (PPE), and improve communications with patients and their families. To guide our teaching teams on the optimal situations for telemedicine, we needed to select those patients who would be most appropriate for a virtual visit.
For example, patients with advanced dementia, or intubated in the intensive care unit, would have less utility from a real-time video encounter. Further, we implemented a simple decision tree (Figure 1). First, the team needs to decide whether the patient needs an immediate in-person assessment; for instance, for critically ill patients or those who need end-of-life care discussions, telemedicine would not be an appropriate modality. Next, the decision is made on whether a patient requires an in-person exam at that time. The idea of forgoing the in-person physical exam may run counterintuitive to the core training medical providers undergo, but in certain circumstances telemedicine can still provide the appropriate level of care a patient requires.
Virtual rounding with residents: Pros and cons
Through the course of this pandemic, there have many questions raised regarding how to handle inpatient teaching services: Should resident teams be assigned COVID-19 positives or PUIs? How do you optimize assessing and learning from patients’ conditions that require human touch? Should all members of the teaching team be donning PPE and entering the patient room?
Internal medicine residents in our hospital have been assigned COVID-19 positive and PUI patients. With proper PPE, and donning and doffing practices, residents may continue to learn from this important training opportunity while also optimizing care for patients supplemented by telemedicine. This pandemic has flattened the hierarchy; often residents are teaching their attendings much of the latest literature and best practices around COVID-19. Residents also benefit by joining the organization’s daily virtual interprofessional COVID-19 huddle where they partner with infectious disease, critical care, pharmacy, and other experts to collaborate in the care of these patients.
There have been counterarguments made for residents joining the front lines with COVID-19 patients. Some have conditions that limit them from seeing this subgroup of patients, such as their immune status or other issues. For these residents, we do not assign COVID-19–positive patients. However, they may continue to support in virtually updating COVID-19 patients and their families. A second argument has been the use of PPE. We have implemented telemedicine to limit the total number of exposures and have a protocol for the fewest number of providers possible to see any at-risk or confirmed COVID-19 patient. For example, a resident who sees a COVID-19 patient in person may also be simultaneously virtually supervised by the attending.
Webside manner
The physical exam is only one of several operational considerations when delivering virtual care, whether with a teaching or nonteaching service. One important aspect is the “webside manner” of the provider, the virtual analogue to bedside manner.
Inherent parts of in-person encounters, such as eye contact and allowing for patients to finish their sentences, have added nuances with virtual care. For instance, providers must adjust to looking into the web camera to make eye contact, even though the patient’s face may be on the screen below. Additionally, for patients who are hard of hearing or unfamiliar with video calling, providers must be cognizant of projecting well over an Internet connection and timing responses to avoid overlapping conversation.
Similarly, there are nuances to the virtual physical exam, some specific to care in the COVID-19 era. In our previous virtual care practice, a bedside facilitator assisted in using tools such a digital stethoscope. In contrast, our current practice aims to refine the observational skills of our learners in conjunction with chart review, vital signs, and actively incorporating the patient in the physical exam. This does not mean asking them to auscultate themselves, but is more toward allowing patients to participate in focused evaluations, such as assessing abdominal tenderness or working through range of motion. Remote guidance for virtual exams also extends itself to teaching teams; for example, in our practice, we have been able to conduct bedside ultrasound teaching with in-person team members and a virtual facilitator.
Maskless connections: ‘Face-to-face’ visits with patients
As many hospitalists have witnessed, COVID-19 is so isolating for patients and their families. Patients have limited visitors, and their care team members are aiming to minimize exposures. Those who are entering the rooms wear masks and face shields that limit connecting with patients in a truly “face-to-face” manner. Telemedicine provides a face-to-face encounter that arguably improves upon portions of the traditional in-person encounter during this pandemic, with providers wearing PPE. For medical learners, gaining the interpersonal skills essential for health care professionals has been skewed with pandemic-related limitations; telemedicine can provide a tool to adapt to this unique era and augment this important educational piece.
Limitations, equity, and technological considerations
Realistically, the virtual exam during COVID-19 does have its limitations. An important part of virtual care and teaching services is instilling the appropriate times for use of telemedicine. If a patient has a clinical change (such as increase in FiO2 requirements) or other clinical need, there should be no hesitation for learners to conduct in-person assessments with appropriate PPE.
Nonexam indications are just as important – for example, if a patient requires extensive goals of care counseling, we recommend this not be done virtually. Other indications may vary between organizations; in our practice, we suggest at least one in-person assessment on the initial and discharge hospital days. Regardless of the specific indications, a successful virtual inpatient teaching service must be predicated on outlining the appropriate uses of telemedicine.
In the United States, there are already health care disparities for people of color and non–English speakers. If there is not a careful consideration for these marginalized groups, their health disparities could be further exacerbated – not just around COVID-19, but also for other inpatient conditions where telemedicine is being used. Groups whose equity must be thoughtfully managed include those who do not speak English and those who do not have access to smartphones or the Internet. Our HealthPartners organization has implemented the integration of interpreters for virtual three-way connections with patients and their clinicians to help mitigate this for non–English speakers. Additionally, utilizing easy-to-use tablets and telemedicine-capable carts has helped patients overcome technology barriers.
Last, the members of the teaching team must know the essential technical aspects of the technology they are using. Robust information technology (IT) support is also needed, but no matter how simple the equipment may be, staff and trainees must know how to both operate it and handle basic troubleshooting (such as audio or video disconnections). This also dovetails with the important element of on-boarding other members of the care team. In our practice, nursing staff, chaplains, interpreters, and dietitians also use virtual care as part of their workflow. However, even if it is used only by the teaching team, orienting other care team members will limit technical problems such as equipment being turned off or moved out of position.
Prior to the COVID-19 pandemic, telemedicine adoption was limited because of lack of awareness, barriers in training, understanding, and narrow beliefs regarding the innovation. The COVID-19 pandemic has resulted in a remarkable increase in the provision of telemedicine services in the inpatient hospital medicine services. Importantly, it is, and should be, a developing part of the education and training for health care learners. This pandemic has underscored the need for providing telemedicine services that will likely long outlast this crisis, and to support our health care learners in being effective “iResidents” on our care teams.
Takeaways
- The future of graduate medical education involves virtual care.
The COVID-19 pandemic response has demonstrated that virtual care plays an instrumental part in patient care, and its effects will not dissipate when the pandemic is done. The curriculum for health care trainees should incorporate telemedicine competencies so that they may more effectively leverage this technology for improving care delivery.
- Selection of telemedicine patients must be stratified.
In order to obtain the highest utility for medical learners on telemedicine, there needs to be a clear decision process for which patients can be seen virtually. This involves both clinical criteria, such as avoiding virtual care for end-of-life discussions, and patient criteria, such as those who are hard of hearing.
- Virtual communication requires new communication skills.
Seeing patients via telemedicine mandates a different skill set than in-person communication. Learners must improve their “webside manner” in order to build the patient-provider relationship. Instilling these tools can pay dividends in settings where telemedicine has high yield, such as maskless communication during a pandemic.
- Health disparities could be further exacerbated by telemedicine and should not be overlooked.
Equity in access to health care applies to telemedicine as it does to many other elements. There are multiple groups that can suffer from disparities, such as patients who need interpreters, or those who have lower technological literacy and access to digital devices. Creating awareness of these pitfalls in virtual care can help medical learners recognize and support in creative solutions for these factors.
Dr. Mathews is chief, hospital medicine, at Regions Hospital, HealthPartners, St. Paul, Minn. Dr. Doshi is telemedicine director, hospital medicine, HealthPartners.
At the start of each shift on his clinical service with rotating internal medicine residents, Benji Mathews, MD, SFHM, now adds a few components to his usual preparation. First, visiting the Minnesota Department of Health and various organizational websites to review the latest COVID-19 updates and guidelines. Next comes checking to see where he needs to pick up the surgical mask and eye protection that he will need to wear through the day. Last, he evaluates which of his patients are in telemedicine-equipped rooms; this last change has fast become a crucial part of working with his resident learners during a pandemic.
During the COVID-19 pandemic, residents and residency programs find themselves in a unique situation. Balancing the educational needs of a training program with the safety of trainees is a challenging task, specifically when taking care of patients who are COVID-19 positive or patients under investigation (PUI). One increasingly available tool that can help protect trainees while continuing to prioritize patient care and medical education is the use of telemedicine for virtual rounding. For our internal medicine residents through the University of Minnesota Internal Medicine Residency program rotating at Regions Hospital in Saint Paul, Minn., we have used video visits to continue our mandate as both health care and education professionals.
Virtual care decision tree
Virtual care can mitigate exposure risk, minimize use of personal protective equipment (PPE), and improve communications with patients and their families. To guide our teaching teams on the optimal situations for telemedicine, we needed to select those patients who would be most appropriate for a virtual visit.

For example, patients with advanced dementia, or intubated in the intensive care unit, would have less utility from a real-time video encounter. Further, we implemented a simple decision tree (Figure 1). First, the team needs to decide whether the patient needs an immediate in-person assessment; for instance, for critically ill patients or those who need end-of-life care discussions, telemedicine would not be an appropriate modality. Next, the decision is made on whether a patient requires an in-person exam at that time. The idea of forgoing the in-person physical exam may run counterintuitive to the core training medical providers undergo, but in certain circumstances telemedicine can still provide the appropriate level of care a patient requires.
Virtual rounding with residents: Pros and cons
Through the course of this pandemic, there have many questions raised regarding how to handle inpatient teaching services: Should resident teams be assigned COVID-19 positives or PUIs? How do you optimize assessing and learning from patients’ conditions that require human touch? Should all members of the teaching team be donning PPE and entering the patient room?
Internal medicine residents in our hospital have been assigned COVID-19 positive and PUI patients. With proper PPE, and donning and doffing practices, residents may continue to learn from this important training opportunity while also optimizing care for patients supplemented by telemedicine. This pandemic has flattened the hierarchy; often residents are teaching their attendings much of the latest literature and best practices around COVID-19. Residents also benefit by joining the organization’s daily virtual interprofessional COVID-19 huddle where they partner with infectious disease, critical care, pharmacy, and other experts to collaborate in the care of these patients.
There have been counterarguments made for residents joining the front lines with COVID-19 patients. Some have conditions that limit them from seeing this subgroup of patients, such as their immune status or other issues. For these residents, we do not assign COVID-19–positive patients. However, they may continue to support in virtually updating COVID-19 patients and their families. A second argument has been the use of PPE. We have implemented telemedicine to limit the total number of exposures and have a protocol for the fewest number of providers possible to see any at-risk or confirmed COVID-19 patient. For example, a resident who sees a COVID-19 patient in person may also be simultaneously virtually supervised by the attending.
Webside manner
The physical exam is only one of several operational considerations when delivering virtual care, whether with a teaching or nonteaching service. One important aspect is the “webside manner” of the provider, the virtual analogue to bedside manner.
Inherent parts of in-person encounters, such as eye contact and allowing for patients to finish their sentences, have added nuances with virtual care. For instance, providers must adjust to looking into the web camera to make eye contact, even though the patient’s face may be on the screen below. Additionally, for patients who are hard of hearing or unfamiliar with video calling, providers must be cognizant of projecting well over an Internet connection and timing responses to avoid overlapping conversation.
Similarly, there are nuances to the virtual physical exam, some specific to care in the COVID-19 era. In our previous virtual care practice, a bedside facilitator assisted in using tools such a digital stethoscope. In contrast, our current practice aims to refine the observational skills of our learners in conjunction with chart review, vital signs, and actively incorporating the patient in the physical exam. This does not mean asking them to auscultate themselves, but is more toward allowing patients to participate in focused evaluations, such as assessing abdominal tenderness or working through range of motion. Remote guidance for virtual exams also extends itself to teaching teams; for example, in our practice, we have been able to conduct bedside ultrasound teaching with in-person team members and a virtual facilitator.
Maskless connections: ‘Face-to-face’ visits with patients
As many hospitalists have witnessed, COVID-19 is so isolating for patients and their families. Patients have limited visitors, and their care team members are aiming to minimize exposures. Those who are entering the rooms wear masks and face shields that limit connecting with patients in a truly “face-to-face” manner. Telemedicine provides a face-to-face encounter that arguably improves upon portions of the traditional in-person encounter during this pandemic, with providers wearing PPE. For medical learners, gaining the interpersonal skills essential for health care professionals has been skewed with pandemic-related limitations; telemedicine can provide a tool to adapt to this unique era and augment this important educational piece.
Limitations, equity, and technological considerations
Realistically, the virtual exam during COVID-19 does have its limitations. An important part of virtual care and teaching services is instilling the appropriate times for use of telemedicine. If a patient has a clinical change (such as increase in FiO2 requirements) or other clinical need, there should be no hesitation for learners to conduct in-person assessments with appropriate PPE.
Nonexam indications are just as important – for example, if a patient requires extensive goals of care counseling, we recommend this not be done virtually. Other indications may vary between organizations; in our practice, we suggest at least one in-person assessment on the initial and discharge hospital days. Regardless of the specific indications, a successful virtual inpatient teaching service must be predicated on outlining the appropriate uses of telemedicine.
In the United States, there are already health care disparities for people of color and non–English speakers. If there is not a careful consideration for these marginalized groups, their health disparities could be further exacerbated – not just around COVID-19, but also for other inpatient conditions where telemedicine is being used. Groups whose equity must be thoughtfully managed include those who do not speak English and those who do not have access to smartphones or the Internet. Our HealthPartners organization has implemented the integration of interpreters for virtual three-way connections with patients and their clinicians to help mitigate this for non–English speakers. Additionally, utilizing easy-to-use tablets and telemedicine-capable carts has helped patients overcome technology barriers.
Last, the members of the teaching team must know the essential technical aspects of the technology they are using. Robust information technology (IT) support is also needed, but no matter how simple the equipment may be, staff and trainees must know how to both operate it and handle basic troubleshooting (such as audio or video disconnections). This also dovetails with the important element of on-boarding other members of the care team. In our practice, nursing staff, chaplains, interpreters, and dietitians also use virtual care as part of their workflow. However, even if it is used only by the teaching team, orienting other care team members will limit technical problems such as equipment being turned off or moved out of position.
Prior to the COVID-19 pandemic, telemedicine adoption was limited because of lack of awareness, barriers in training, understanding, and narrow beliefs regarding the innovation. The COVID-19 pandemic has resulted in a remarkable increase in the provision of telemedicine services in the inpatient hospital medicine services. Importantly, it is, and should be, a developing part of the education and training for health care learners. This pandemic has underscored the need for providing telemedicine services that will likely long outlast this crisis, and to support our health care learners in being effective “iResidents” on our care teams.
Takeaways
- The future of graduate medical education involves virtual care.
The COVID-19 pandemic response has demonstrated that virtual care plays an instrumental part in patient care, and its effects will not dissipate when the pandemic is done. The curriculum for health care trainees should incorporate telemedicine competencies so that they may more effectively leverage this technology for improving care delivery.
- Selection of telemedicine patients must be stratified.
In order to obtain the highest utility for medical learners on telemedicine, there needs to be a clear decision process for which patients can be seen virtually. This involves both clinical criteria, such as avoiding virtual care for end-of-life discussions, and patient criteria, such as those who are hard of hearing.
- Virtual communication requires new communication skills.
Seeing patients via telemedicine mandates a different skill set than in-person communication. Learners must improve their “webside manner” in order to build the patient-provider relationship. Instilling these tools can pay dividends in settings where telemedicine has high yield, such as maskless communication during a pandemic.
- Health disparities could be further exacerbated by telemedicine and should not be overlooked.
Equity in access to health care applies to telemedicine as it does to many other elements. There are multiple groups that can suffer from disparities, such as patients who need interpreters, or those who have lower technological literacy and access to digital devices. Creating awareness of these pitfalls in virtual care can help medical learners recognize and support in creative solutions for these factors.
Dr. Mathews is chief, hospital medicine, at Regions Hospital, HealthPartners, St. Paul, Minn. Dr. Doshi is telemedicine director, hospital medicine, HealthPartners.
At the start of each shift on his clinical service with rotating internal medicine residents, Benji Mathews, MD, SFHM, now adds a few components to his usual preparation. First, visiting the Minnesota Department of Health and various organizational websites to review the latest COVID-19 updates and guidelines. Next comes checking to see where he needs to pick up the surgical mask and eye protection that he will need to wear through the day. Last, he evaluates which of his patients are in telemedicine-equipped rooms; this last change has fast become a crucial part of working with his resident learners during a pandemic.
During the COVID-19 pandemic, residents and residency programs find themselves in a unique situation. Balancing the educational needs of a training program with the safety of trainees is a challenging task, specifically when taking care of patients who are COVID-19 positive or patients under investigation (PUI). One increasingly available tool that can help protect trainees while continuing to prioritize patient care and medical education is the use of telemedicine for virtual rounding. For our internal medicine residents through the University of Minnesota Internal Medicine Residency program rotating at Regions Hospital in Saint Paul, Minn., we have used video visits to continue our mandate as both health care and education professionals.
Virtual care decision tree
Virtual care can mitigate exposure risk, minimize use of personal protective equipment (PPE), and improve communications with patients and their families. To guide our teaching teams on the optimal situations for telemedicine, we needed to select those patients who would be most appropriate for a virtual visit.

For example, patients with advanced dementia, or intubated in the intensive care unit, would have less utility from a real-time video encounter. Further, we implemented a simple decision tree (Figure 1). First, the team needs to decide whether the patient needs an immediate in-person assessment; for instance, for critically ill patients or those who need end-of-life care discussions, telemedicine would not be an appropriate modality. Next, the decision is made on whether a patient requires an in-person exam at that time. The idea of forgoing the in-person physical exam may run counterintuitive to the core training medical providers undergo, but in certain circumstances telemedicine can still provide the appropriate level of care a patient requires.
Virtual rounding with residents: Pros and cons
Through the course of this pandemic, there have many questions raised regarding how to handle inpatient teaching services: Should resident teams be assigned COVID-19 positives or PUIs? How do you optimize assessing and learning from patients’ conditions that require human touch? Should all members of the teaching team be donning PPE and entering the patient room?
Internal medicine residents in our hospital have been assigned COVID-19 positive and PUI patients. With proper PPE, and donning and doffing practices, residents may continue to learn from this important training opportunity while also optimizing care for patients supplemented by telemedicine. This pandemic has flattened the hierarchy; often residents are teaching their attendings much of the latest literature and best practices around COVID-19. Residents also benefit by joining the organization’s daily virtual interprofessional COVID-19 huddle where they partner with infectious disease, critical care, pharmacy, and other experts to collaborate in the care of these patients.
There have been counterarguments made for residents joining the front lines with COVID-19 patients. Some have conditions that limit them from seeing this subgroup of patients, such as their immune status or other issues. For these residents, we do not assign COVID-19–positive patients. However, they may continue to support in virtually updating COVID-19 patients and their families. A second argument has been the use of PPE. We have implemented telemedicine to limit the total number of exposures and have a protocol for the fewest number of providers possible to see any at-risk or confirmed COVID-19 patient. For example, a resident who sees a COVID-19 patient in person may also be simultaneously virtually supervised by the attending.
Webside manner
The physical exam is only one of several operational considerations when delivering virtual care, whether with a teaching or nonteaching service. One important aspect is the “webside manner” of the provider, the virtual analogue to bedside manner.
Inherent parts of in-person encounters, such as eye contact and allowing for patients to finish their sentences, have added nuances with virtual care. For instance, providers must adjust to looking into the web camera to make eye contact, even though the patient’s face may be on the screen below. Additionally, for patients who are hard of hearing or unfamiliar with video calling, providers must be cognizant of projecting well over an Internet connection and timing responses to avoid overlapping conversation.
Similarly, there are nuances to the virtual physical exam, some specific to care in the COVID-19 era. In our previous virtual care practice, a bedside facilitator assisted in using tools such a digital stethoscope. In contrast, our current practice aims to refine the observational skills of our learners in conjunction with chart review, vital signs, and actively incorporating the patient in the physical exam. This does not mean asking them to auscultate themselves, but is more toward allowing patients to participate in focused evaluations, such as assessing abdominal tenderness or working through range of motion. Remote guidance for virtual exams also extends itself to teaching teams; for example, in our practice, we have been able to conduct bedside ultrasound teaching with in-person team members and a virtual facilitator.
Maskless connections: ‘Face-to-face’ visits with patients
As many hospitalists have witnessed, COVID-19 is so isolating for patients and their families. Patients have limited visitors, and their care team members are aiming to minimize exposures. Those who are entering the rooms wear masks and face shields that limit connecting with patients in a truly “face-to-face” manner. Telemedicine provides a face-to-face encounter that arguably improves upon portions of the traditional in-person encounter during this pandemic, with providers wearing PPE. For medical learners, gaining the interpersonal skills essential for health care professionals has been skewed with pandemic-related limitations; telemedicine can provide a tool to adapt to this unique era and augment this important educational piece.
Limitations, equity, and technological considerations
Realistically, the virtual exam during COVID-19 does have its limitations. An important part of virtual care and teaching services is instilling the appropriate times for use of telemedicine. If a patient has a clinical change (such as increase in FiO2 requirements) or other clinical need, there should be no hesitation for learners to conduct in-person assessments with appropriate PPE.
Nonexam indications are just as important – for example, if a patient requires extensive goals of care counseling, we recommend this not be done virtually. Other indications may vary between organizations; in our practice, we suggest at least one in-person assessment on the initial and discharge hospital days. Regardless of the specific indications, a successful virtual inpatient teaching service must be predicated on outlining the appropriate uses of telemedicine.
In the United States, there are already health care disparities for people of color and non–English speakers. If there is not a careful consideration for these marginalized groups, their health disparities could be further exacerbated – not just around COVID-19, but also for other inpatient conditions where telemedicine is being used. Groups whose equity must be thoughtfully managed include those who do not speak English and those who do not have access to smartphones or the Internet. Our HealthPartners organization has implemented the integration of interpreters for virtual three-way connections with patients and their clinicians to help mitigate this for non–English speakers. Additionally, utilizing easy-to-use tablets and telemedicine-capable carts has helped patients overcome technology barriers.
Last, the members of the teaching team must know the essential technical aspects of the technology they are using. Robust information technology (IT) support is also needed, but no matter how simple the equipment may be, staff and trainees must know how to both operate it and handle basic troubleshooting (such as audio or video disconnections). This also dovetails with the important element of on-boarding other members of the care team. In our practice, nursing staff, chaplains, interpreters, and dietitians also use virtual care as part of their workflow. However, even if it is used only by the teaching team, orienting other care team members will limit technical problems such as equipment being turned off or moved out of position.
Prior to the COVID-19 pandemic, telemedicine adoption was limited because of lack of awareness, barriers in training, understanding, and narrow beliefs regarding the innovation. The COVID-19 pandemic has resulted in a remarkable increase in the provision of telemedicine services in the inpatient hospital medicine services. Importantly, it is, and should be, a developing part of the education and training for health care learners. This pandemic has underscored the need for providing telemedicine services that will likely long outlast this crisis, and to support our health care learners in being effective “iResidents” on our care teams.
Takeaways
- The future of graduate medical education involves virtual care.
The COVID-19 pandemic response has demonstrated that virtual care plays an instrumental part in patient care, and its effects will not dissipate when the pandemic is done. The curriculum for health care trainees should incorporate telemedicine competencies so that they may more effectively leverage this technology for improving care delivery.
- Selection of telemedicine patients must be stratified.
In order to obtain the highest utility for medical learners on telemedicine, there needs to be a clear decision process for which patients can be seen virtually. This involves both clinical criteria, such as avoiding virtual care for end-of-life discussions, and patient criteria, such as those who are hard of hearing.
- Virtual communication requires new communication skills.
Seeing patients via telemedicine mandates a different skill set than in-person communication. Learners must improve their “webside manner” in order to build the patient-provider relationship. Instilling these tools can pay dividends in settings where telemedicine has high yield, such as maskless communication during a pandemic.
- Health disparities could be further exacerbated by telemedicine and should not be overlooked.
Equity in access to health care applies to telemedicine as it does to many other elements. There are multiple groups that can suffer from disparities, such as patients who need interpreters, or those who have lower technological literacy and access to digital devices. Creating awareness of these pitfalls in virtual care can help medical learners recognize and support in creative solutions for these factors.
Dr. Mathews is chief, hospital medicine, at Regions Hospital, HealthPartners, St. Paul, Minn. Dr. Doshi is telemedicine director, hospital medicine, HealthPartners.
Taming a terrible illness
Darth Vader is, to me, one of the most intimidating villains in movie history. I was 11 when Star Wars came out. I even cleaned my room so my mother would take me to see it.
When Darth Vader first walked on screen, it was striking. A tall, imposing figure in black, with harsh mechanical respirations. There was no question of who the bad guy was. As the movie progressed his darkness became more frightening until, in the first lightsaber battle any of us had seen, he cut down the benevolent Obi-Wan Kenobi.
Last year my family went to Disneyland. While browsing the park’s stores we saw numerous Darth Vader items ... with him now available as a teddy bear, and on T-shirts riding carousels and the Dumbo ride.
From terrifying villain to cutesy toy in 43 years.* Quite the fall from glory.
Diseases are often (and hopefully) like that. Syphilis, once the most common, feared, and incurable neurologic disease is now, for most, just a nuisance. The butt of jokes and sexual innuendos, rendered harmless by Alexander Fleming’s discoveries.
Bit by bit we see other diseases tamed. Multiple sclerosis, though still serious, becomes better controlled every year as new agents come out. The cure for Parkinson’s disease remains elusive, but agents to control the symptoms and improve quality of life are available. Even HIV, the most feared disease of the 80s and 90s, has been beaten back from a terrible death sentence to one where patients lead normal lives with antiviral therapy.
Today we face a new enemy, the COVID-19 pandemic. So far we have no definite treatments, nor shortage of ideas. Many companies are racing to develop a vaccine, and will likely, at some point, find one, but what and when are still in the future. too.
Alzheimer’s disease, for all practical purposes, remains untreatable and rightfully feared. Perhaps the only ones more terrifying are those we’ve reduced to just three letters: ALS (amyotrophic lateral sclerosis) and GBM (glioblastoma multiforme). Both have terrible courses and, in spite of years of research, nothing even close to an effective treatment.
I hope that changes, and soon, for all those affected by these (and many other) terrible disorders.
Like the Darth Vader teddy bear, I’ll be happy to see them become shells of their former selves, with the dread they bring now reduced to the lesser trepidation seen when facing a serious, but treatable, illness.
*Correction, 8/11/20: An earlier version of this column misstated the number of years since Star Wars debuted.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Darth Vader is, to me, one of the most intimidating villains in movie history. I was 11 when Star Wars came out. I even cleaned my room so my mother would take me to see it.
When Darth Vader first walked on screen, it was striking. A tall, imposing figure in black, with harsh mechanical respirations. There was no question of who the bad guy was. As the movie progressed his darkness became more frightening until, in the first lightsaber battle any of us had seen, he cut down the benevolent Obi-Wan Kenobi.
Last year my family went to Disneyland. While browsing the park’s stores we saw numerous Darth Vader items ... with him now available as a teddy bear, and on T-shirts riding carousels and the Dumbo ride.
From terrifying villain to cutesy toy in 43 years.* Quite the fall from glory.
Diseases are often (and hopefully) like that. Syphilis, once the most common, feared, and incurable neurologic disease is now, for most, just a nuisance. The butt of jokes and sexual innuendos, rendered harmless by Alexander Fleming’s discoveries.
Bit by bit we see other diseases tamed. Multiple sclerosis, though still serious, becomes better controlled every year as new agents come out. The cure for Parkinson’s disease remains elusive, but agents to control the symptoms and improve quality of life are available. Even HIV, the most feared disease of the 80s and 90s, has been beaten back from a terrible death sentence to one where patients lead normal lives with antiviral therapy.
Today we face a new enemy, the COVID-19 pandemic. So far we have no definite treatments, nor shortage of ideas. Many companies are racing to develop a vaccine, and will likely, at some point, find one, but what and when are still in the future. too.
Alzheimer’s disease, for all practical purposes, remains untreatable and rightfully feared. Perhaps the only ones more terrifying are those we’ve reduced to just three letters: ALS (amyotrophic lateral sclerosis) and GBM (glioblastoma multiforme). Both have terrible courses and, in spite of years of research, nothing even close to an effective treatment.
I hope that changes, and soon, for all those affected by these (and many other) terrible disorders.
Like the Darth Vader teddy bear, I’ll be happy to see them become shells of their former selves, with the dread they bring now reduced to the lesser trepidation seen when facing a serious, but treatable, illness.
*Correction, 8/11/20: An earlier version of this column misstated the number of years since Star Wars debuted.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Darth Vader is, to me, one of the most intimidating villains in movie history. I was 11 when Star Wars came out. I even cleaned my room so my mother would take me to see it.
When Darth Vader first walked on screen, it was striking. A tall, imposing figure in black, with harsh mechanical respirations. There was no question of who the bad guy was. As the movie progressed his darkness became more frightening until, in the first lightsaber battle any of us had seen, he cut down the benevolent Obi-Wan Kenobi.
Last year my family went to Disneyland. While browsing the park’s stores we saw numerous Darth Vader items ... with him now available as a teddy bear, and on T-shirts riding carousels and the Dumbo ride.
From terrifying villain to cutesy toy in 43 years.* Quite the fall from glory.
Diseases are often (and hopefully) like that. Syphilis, once the most common, feared, and incurable neurologic disease is now, for most, just a nuisance. The butt of jokes and sexual innuendos, rendered harmless by Alexander Fleming’s discoveries.
Bit by bit we see other diseases tamed. Multiple sclerosis, though still serious, becomes better controlled every year as new agents come out. The cure for Parkinson’s disease remains elusive, but agents to control the symptoms and improve quality of life are available. Even HIV, the most feared disease of the 80s and 90s, has been beaten back from a terrible death sentence to one where patients lead normal lives with antiviral therapy.
Today we face a new enemy, the COVID-19 pandemic. So far we have no definite treatments, nor shortage of ideas. Many companies are racing to develop a vaccine, and will likely, at some point, find one, but what and when are still in the future. too.
Alzheimer’s disease, for all practical purposes, remains untreatable and rightfully feared. Perhaps the only ones more terrifying are those we’ve reduced to just three letters: ALS (amyotrophic lateral sclerosis) and GBM (glioblastoma multiforme). Both have terrible courses and, in spite of years of research, nothing even close to an effective treatment.
I hope that changes, and soon, for all those affected by these (and many other) terrible disorders.
Like the Darth Vader teddy bear, I’ll be happy to see them become shells of their former selves, with the dread they bring now reduced to the lesser trepidation seen when facing a serious, but treatable, illness.
*Correction, 8/11/20: An earlier version of this column misstated the number of years since Star Wars debuted.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Coming soon: The 2020 SoHM Report!
On behalf of SHM’s Practice Analysis Committee, I am excited to announce the scheduled September 2020 release of the 2020 State of Hospital Medicine Report (SoHM)!
For reasons all too familiar, this year’s SoHM survey process was unlike any in SHM’s history. We were still collecting survey responses from a few stragglers in early March when the entire world shut down almost overnight to flatten the curve of a deadly pandemic. Hospital medicine group (HMG) leaders were suddenly either up to their eyeballs trying to figure out how to safely care for huge influxes of COVID-19 patients that overwhelmed established systems of care or were trying to figure out how to staff in a low-volume environment with few COVID patients, a relative trickle of ED admissions, and virtually no surgical care. And everywhere, hospitals and their HMGs were quickly stressed in ways that would have been unimaginable just a couple of months earlier – financially, operationally, epidemiologically, and culturally.
SHM offices closed, with all staff working from home. And the talented people who would normally have been working diligently on the survey data were suddenly redirected to focus on COVID-related issues, including tracking government announcements that were changing daily and providing needed resources to SHM members. By the time they could raise their heads and begin thinking about survey data, we were months behind schedule.
I need to give a huge shout-out to our survey manager extraordinaire Josh Lapps, SHM’s Director of Policy and Practice Management, and his survey support team including Luke Heisinger and Kim Schonberger. Once they were able to turn their focus back to the SoHM, they worked like demons to catch up. And in addition to the work of preparing the SoHM for publication, they helped issue and analyze a follow-up survey to investigate how HMGs adjusted their staffing and operations in response to COVID! As I write this, we appear to be back on schedule for a September SoHM release date, with the COVID supplemental survey report to follow soon after. Thanks also to PAC committee members who, despite their own stresses, rose to the challenge of participating in calls and planning the supplemental survey.
Despite the pandemic, HMGs found survey participation valuable. When all was said and done, we had a respectable number of respondent groups: 502 this year vs. 569 in 2018. Although the number of respondent groups is down, the average group size has increased, so that an all-time high of 10,122 employed/contracted full-time equivalent (FTE) hospitalists (plus 484 locum tenens FTEs) are represented in the data set. The respondents continue to be very diverse, representing all practice models and every state – and even a couple of other countries. One notable change is a significant increase in pediatric HM group participation, thanks to a recruitment charge led by PAC member Sandra Gage, associate division chief of hospital medicine at Phoenix Children’s Hospital, and supported by the inclusion of several new pediatric HM-specific questions to better capture unique attributes of these hospital medicine practices.
We had more multisite respondents than ever, and the multisite respondents overwhelmingly used the new “retake” feature in the online version of the survey. I’m happy to report that we received consistent positive feedback about our new electronic survey platform, and thanks to its capabilities data analysis has been significantly automated, enhancing both efficiency and data reliability.
The survey content is more wide ranging than ever. In addition to the usual topics such as scope of services, staffing and scheduling, compensation models, evaluation and management code distribution, and HM group finances, the 2020 report will include the afore-referenced information about HM groups serving children, expanded information on nurse practitioner (NPs)/physician assistant (PA) roles, and data on diversity in HM physician leadership. The follow-up COVID survey will be published separately as a supplement, available only to purchasers of the SoHM report.
Multiple options for SoHM report purchase. All survey participants will receive access to the online version of the survey. Others may purchase the hard copy report, online access, or both. The report has a colorful, easy-to-read layout, and many of the tables have been streamlined to make them easier to read. I encourage you to sign up to preorder your copy of the SoHM Report today at www.hospitalmedicine.org/sohm; you’ll almost certainly discover a treasure trove of worthwhile information.
Use the report to assess how your practice compares to other practices, but always keep in mind that surveys don’t tell you what should be; they only tell you what currently is the case – or at least, what was during the survey period. New best practices not yet reflected in survey data are emerging all the time, and that is probably more true today in the new world affected by this pandemic than ever before. And while the ways others do things won’t always be right for your group’s unique situation and needs, it always helps to know how you compare with others. Whether you are partners or employees, you and your colleagues “own” the success of your hospital medicine practice and, armed with the best available data, are the best judges of what is right for you.
Ms. Flores is a partner at Nelson Flores Hospital Medicine Consultants in La Quinta, Calif. She serves on SHM’s Practice Analysis and Annual Conference Committees and helps to coordinate SHM’s biannual State of Hospital Medicine survey.
On behalf of SHM’s Practice Analysis Committee, I am excited to announce the scheduled September 2020 release of the 2020 State of Hospital Medicine Report (SoHM)!
For reasons all too familiar, this year’s SoHM survey process was unlike any in SHM’s history. We were still collecting survey responses from a few stragglers in early March when the entire world shut down almost overnight to flatten the curve of a deadly pandemic. Hospital medicine group (HMG) leaders were suddenly either up to their eyeballs trying to figure out how to safely care for huge influxes of COVID-19 patients that overwhelmed established systems of care or were trying to figure out how to staff in a low-volume environment with few COVID patients, a relative trickle of ED admissions, and virtually no surgical care. And everywhere, hospitals and their HMGs were quickly stressed in ways that would have been unimaginable just a couple of months earlier – financially, operationally, epidemiologically, and culturally.
SHM offices closed, with all staff working from home. And the talented people who would normally have been working diligently on the survey data were suddenly redirected to focus on COVID-related issues, including tracking government announcements that were changing daily and providing needed resources to SHM members. By the time they could raise their heads and begin thinking about survey data, we were months behind schedule.
I need to give a huge shout-out to our survey manager extraordinaire Josh Lapps, SHM’s Director of Policy and Practice Management, and his survey support team including Luke Heisinger and Kim Schonberger. Once they were able to turn their focus back to the SoHM, they worked like demons to catch up. And in addition to the work of preparing the SoHM for publication, they helped issue and analyze a follow-up survey to investigate how HMGs adjusted their staffing and operations in response to COVID! As I write this, we appear to be back on schedule for a September SoHM release date, with the COVID supplemental survey report to follow soon after. Thanks also to PAC committee members who, despite their own stresses, rose to the challenge of participating in calls and planning the supplemental survey.
Despite the pandemic, HMGs found survey participation valuable. When all was said and done, we had a respectable number of respondent groups: 502 this year vs. 569 in 2018. Although the number of respondent groups is down, the average group size has increased, so that an all-time high of 10,122 employed/contracted full-time equivalent (FTE) hospitalists (plus 484 locum tenens FTEs) are represented in the data set. The respondents continue to be very diverse, representing all practice models and every state – and even a couple of other countries. One notable change is a significant increase in pediatric HM group participation, thanks to a recruitment charge led by PAC member Sandra Gage, associate division chief of hospital medicine at Phoenix Children’s Hospital, and supported by the inclusion of several new pediatric HM-specific questions to better capture unique attributes of these hospital medicine practices.
We had more multisite respondents than ever, and the multisite respondents overwhelmingly used the new “retake” feature in the online version of the survey. I’m happy to report that we received consistent positive feedback about our new electronic survey platform, and thanks to its capabilities data analysis has been significantly automated, enhancing both efficiency and data reliability.
The survey content is more wide ranging than ever. In addition to the usual topics such as scope of services, staffing and scheduling, compensation models, evaluation and management code distribution, and HM group finances, the 2020 report will include the afore-referenced information about HM groups serving children, expanded information on nurse practitioner (NPs)/physician assistant (PA) roles, and data on diversity in HM physician leadership. The follow-up COVID survey will be published separately as a supplement, available only to purchasers of the SoHM report.
Multiple options for SoHM report purchase. All survey participants will receive access to the online version of the survey. Others may purchase the hard copy report, online access, or both. The report has a colorful, easy-to-read layout, and many of the tables have been streamlined to make them easier to read. I encourage you to sign up to preorder your copy of the SoHM Report today at www.hospitalmedicine.org/sohm; you’ll almost certainly discover a treasure trove of worthwhile information.
Use the report to assess how your practice compares to other practices, but always keep in mind that surveys don’t tell you what should be; they only tell you what currently is the case – or at least, what was during the survey period. New best practices not yet reflected in survey data are emerging all the time, and that is probably more true today in the new world affected by this pandemic than ever before. And while the ways others do things won’t always be right for your group’s unique situation and needs, it always helps to know how you compare with others. Whether you are partners or employees, you and your colleagues “own” the success of your hospital medicine practice and, armed with the best available data, are the best judges of what is right for you.
Ms. Flores is a partner at Nelson Flores Hospital Medicine Consultants in La Quinta, Calif. She serves on SHM’s Practice Analysis and Annual Conference Committees and helps to coordinate SHM’s biannual State of Hospital Medicine survey.
On behalf of SHM’s Practice Analysis Committee, I am excited to announce the scheduled September 2020 release of the 2020 State of Hospital Medicine Report (SoHM)!
For reasons all too familiar, this year’s SoHM survey process was unlike any in SHM’s history. We were still collecting survey responses from a few stragglers in early March when the entire world shut down almost overnight to flatten the curve of a deadly pandemic. Hospital medicine group (HMG) leaders were suddenly either up to their eyeballs trying to figure out how to safely care for huge influxes of COVID-19 patients that overwhelmed established systems of care or were trying to figure out how to staff in a low-volume environment with few COVID patients, a relative trickle of ED admissions, and virtually no surgical care. And everywhere, hospitals and their HMGs were quickly stressed in ways that would have been unimaginable just a couple of months earlier – financially, operationally, epidemiologically, and culturally.
SHM offices closed, with all staff working from home. And the talented people who would normally have been working diligently on the survey data were suddenly redirected to focus on COVID-related issues, including tracking government announcements that were changing daily and providing needed resources to SHM members. By the time they could raise their heads and begin thinking about survey data, we were months behind schedule.
I need to give a huge shout-out to our survey manager extraordinaire Josh Lapps, SHM’s Director of Policy and Practice Management, and his survey support team including Luke Heisinger and Kim Schonberger. Once they were able to turn their focus back to the SoHM, they worked like demons to catch up. And in addition to the work of preparing the SoHM for publication, they helped issue and analyze a follow-up survey to investigate how HMGs adjusted their staffing and operations in response to COVID! As I write this, we appear to be back on schedule for a September SoHM release date, with the COVID supplemental survey report to follow soon after. Thanks also to PAC committee members who, despite their own stresses, rose to the challenge of participating in calls and planning the supplemental survey.
Despite the pandemic, HMGs found survey participation valuable. When all was said and done, we had a respectable number of respondent groups: 502 this year vs. 569 in 2018. Although the number of respondent groups is down, the average group size has increased, so that an all-time high of 10,122 employed/contracted full-time equivalent (FTE) hospitalists (plus 484 locum tenens FTEs) are represented in the data set. The respondents continue to be very diverse, representing all practice models and every state – and even a couple of other countries. One notable change is a significant increase in pediatric HM group participation, thanks to a recruitment charge led by PAC member Sandra Gage, associate division chief of hospital medicine at Phoenix Children’s Hospital, and supported by the inclusion of several new pediatric HM-specific questions to better capture unique attributes of these hospital medicine practices.
We had more multisite respondents than ever, and the multisite respondents overwhelmingly used the new “retake” feature in the online version of the survey. I’m happy to report that we received consistent positive feedback about our new electronic survey platform, and thanks to its capabilities data analysis has been significantly automated, enhancing both efficiency and data reliability.
The survey content is more wide ranging than ever. In addition to the usual topics such as scope of services, staffing and scheduling, compensation models, evaluation and management code distribution, and HM group finances, the 2020 report will include the afore-referenced information about HM groups serving children, expanded information on nurse practitioner (NPs)/physician assistant (PA) roles, and data on diversity in HM physician leadership. The follow-up COVID survey will be published separately as a supplement, available only to purchasers of the SoHM report.
Multiple options for SoHM report purchase. All survey participants will receive access to the online version of the survey. Others may purchase the hard copy report, online access, or both. The report has a colorful, easy-to-read layout, and many of the tables have been streamlined to make them easier to read. I encourage you to sign up to preorder your copy of the SoHM Report today at www.hospitalmedicine.org/sohm; you’ll almost certainly discover a treasure trove of worthwhile information.
Use the report to assess how your practice compares to other practices, but always keep in mind that surveys don’t tell you what should be; they only tell you what currently is the case – or at least, what was during the survey period. New best practices not yet reflected in survey data are emerging all the time, and that is probably more true today in the new world affected by this pandemic than ever before. And while the ways others do things won’t always be right for your group’s unique situation and needs, it always helps to know how you compare with others. Whether you are partners or employees, you and your colleagues “own” the success of your hospital medicine practice and, armed with the best available data, are the best judges of what is right for you.
Ms. Flores is a partner at Nelson Flores Hospital Medicine Consultants in La Quinta, Calif. She serves on SHM’s Practice Analysis and Annual Conference Committees and helps to coordinate SHM’s biannual State of Hospital Medicine survey.
Telemedicine in primary care
How to effectively utilize this tool
By now it is well known that the COVID-19 pandemic has significantly disrupted primary care. Office visits and revenues have precipitously dropped as physicians and patients alike fear in-person visits may increase their risks of contracting the virus. However, telemedicine has emerged as a lifeline of sorts for many practices, enabling them to conduct visits and maintain contact with patients.
Telemedicine is likely to continue to serve as a tool for primary care providers to improve access to convenient, cost-effective, high-quality care after the pandemic. Another benefit of telemedicine is it can help maintain a portion of a practice’s revenue stream for physicians during uncertain times.
Indeed, the nation has seen recent progress toward telemedicine parity, which refers to the concept of reimbursing providers’ telehealth visits at the same rates as similar in-person visits.
A challenge to adopting telemedicine is that it calls for adjusting established workflows for in-person encounters. A practice cannot simply replicate in-person processes to work for telehealth. While both in-person and virtual visits require adherence to HIPAA, for example, how you actually protect patient privacy will call for different measures. Harking back to the early days of EMR implementation, one does not need to like the telemedicine platform or process, but come to terms with the fact that it is a tool that is here to stay to deliver patient care.
Treat your practice like a laboratory
Adoption may vary between practices depending on many factors, including clinicians’ comfort with technology, clinical tolerance and triage rules for nontouch encounters, state regulations, and more. Every provider group should begin experimenting with telemedicine in specific ways that make sense for them.
One physician may practice telemedicine full-time while the rest abstain, or perhaps the practice prefers to offer telemedicine services during specific hours on specific days. Don’t be afraid to start slowly when you’re trying something new – but do get started with telehealth. It will increasingly be a mainstream medium and more patients will come to expect it.
Train the entire team
Many primary care practices do not enjoy the resources of an information technology team, so all team members essentially need to learn the new skill of telemedicine usage, in addition to assisting patients. That can’t happen without staff buy-in, so it is essential that everyone from the office manager to medical assistants have the training they need to make the technology work. Juggling schedules for telehealth and in-office, activating an account through email, starting and joining a telehealth meeting, and preparing a patient for a visit are just a handful of basic tasks your staff should be trained to do to contribute to the successful integration of telehealth.
Educate and encourage patients to use telehealth
While unfamiliarity with technology may represent a roadblock for some patients, others resist telemedicine simply because no one has explained to them why it’s so important and the benefits it can hold for them. Education and communication are critical, including the sometimes painstaking work of slowly walking patients through the process of performing important functions on the telemedicine app. By providing them with some friendly coaching, patients won’t feel lost or abandoned during what for some may be an unfamiliar and frustrating process.
Manage more behavioral health
Different states and health plans incentivize primary practices for integrating behavioral health into their offerings. Rather than dismiss this addition to your own practice as too cumbersome to take on, I would recommend using telehealth to expand behavioral health care services.
If your practice is working toward a team-based, interdisciplinary approach to care delivery, behavioral health is a critical component. While other elements of this “whole person” health care may be better suited for an office visit, the vast majority of behavioral health services can be delivered virtually.
To decide if your patient may benefit from behavioral health care, the primary care provider (PCP) can conduct a screening via telehealth. Once the screening is complete, the PCP can discuss results and refer the patient to a mental health professional – all via telehealth. While patients may be reluctant to receive behavioral health treatment, perhaps because of stigma or inexperience, they may appreciate the telemedicine option as they can remain in the comfort and familiarity of their homes.
Collaborative Care is both an in-person and virtual model that allows PCP practices to offer behavioral health services in a cost effective way by utilizing a psychiatrist as a “consultant” to the practice as opposed to hiring a full-time psychiatrist. All services within the Collaborative Care Model can be offered via telehealth, and all major insurance providers reimburse primary care providers for delivering Collaborative Care.
When PCPs provide behavioral health treatment as an “extension” of the primary care service offerings, the stigma is reduced and more patients are willing to accept the care they need.
Many areas of the country suffer from a lack of access to behavioral health specialists. In rural counties, for example, the nearest therapist may be located over an hour away. By integrating behavioral telehealth services into your practice’s offerings, you can remove geographic and transportation obstacles to care for your patient population.
Doing this can lead to providing more culturally competent care. It’s important that you’re able to offer mental health services to your patients from a professional with a similar ethnic or racial background. Language barriers and cultural differences may limit a provider’s ability to treat a patient, particularly if the patient faces health disparities related to race or ethnicity. If your practice needs to look outside of your community to tap into a more diverse pool of providers to better meet your patients’ needs, telehealth makes it easier to do that.
Adopting telemedicine for consultative patient visits offers primary care a path toward restoring patient volume and hope for a postpandemic future.
Mark Stephan, MD, is chief medical officer at Equality Health, a whole-health delivery system. He practiced family medicine for 19 years, including hospital medicine and obstetrics in rural and urban settings. Dr. Stephan has no conflicts related to the content of this piece.
How to effectively utilize this tool
How to effectively utilize this tool
By now it is well known that the COVID-19 pandemic has significantly disrupted primary care. Office visits and revenues have precipitously dropped as physicians and patients alike fear in-person visits may increase their risks of contracting the virus. However, telemedicine has emerged as a lifeline of sorts for many practices, enabling them to conduct visits and maintain contact with patients.
Telemedicine is likely to continue to serve as a tool for primary care providers to improve access to convenient, cost-effective, high-quality care after the pandemic. Another benefit of telemedicine is it can help maintain a portion of a practice’s revenue stream for physicians during uncertain times.
Indeed, the nation has seen recent progress toward telemedicine parity, which refers to the concept of reimbursing providers’ telehealth visits at the same rates as similar in-person visits.
A challenge to adopting telemedicine is that it calls for adjusting established workflows for in-person encounters. A practice cannot simply replicate in-person processes to work for telehealth. While both in-person and virtual visits require adherence to HIPAA, for example, how you actually protect patient privacy will call for different measures. Harking back to the early days of EMR implementation, one does not need to like the telemedicine platform or process, but come to terms with the fact that it is a tool that is here to stay to deliver patient care.
Treat your practice like a laboratory
Adoption may vary between practices depending on many factors, including clinicians’ comfort with technology, clinical tolerance and triage rules for nontouch encounters, state regulations, and more. Every provider group should begin experimenting with telemedicine in specific ways that make sense for them.
One physician may practice telemedicine full-time while the rest abstain, or perhaps the practice prefers to offer telemedicine services during specific hours on specific days. Don’t be afraid to start slowly when you’re trying something new – but do get started with telehealth. It will increasingly be a mainstream medium and more patients will come to expect it.
Train the entire team
Many primary care practices do not enjoy the resources of an information technology team, so all team members essentially need to learn the new skill of telemedicine usage, in addition to assisting patients. That can’t happen without staff buy-in, so it is essential that everyone from the office manager to medical assistants have the training they need to make the technology work. Juggling schedules for telehealth and in-office, activating an account through email, starting and joining a telehealth meeting, and preparing a patient for a visit are just a handful of basic tasks your staff should be trained to do to contribute to the successful integration of telehealth.
Educate and encourage patients to use telehealth
While unfamiliarity with technology may represent a roadblock for some patients, others resist telemedicine simply because no one has explained to them why it’s so important and the benefits it can hold for them. Education and communication are critical, including the sometimes painstaking work of slowly walking patients through the process of performing important functions on the telemedicine app. By providing them with some friendly coaching, patients won’t feel lost or abandoned during what for some may be an unfamiliar and frustrating process.
Manage more behavioral health
Different states and health plans incentivize primary practices for integrating behavioral health into their offerings. Rather than dismiss this addition to your own practice as too cumbersome to take on, I would recommend using telehealth to expand behavioral health care services.
If your practice is working toward a team-based, interdisciplinary approach to care delivery, behavioral health is a critical component. While other elements of this “whole person” health care may be better suited for an office visit, the vast majority of behavioral health services can be delivered virtually.
To decide if your patient may benefit from behavioral health care, the primary care provider (PCP) can conduct a screening via telehealth. Once the screening is complete, the PCP can discuss results and refer the patient to a mental health professional – all via telehealth. While patients may be reluctant to receive behavioral health treatment, perhaps because of stigma or inexperience, they may appreciate the telemedicine option as they can remain in the comfort and familiarity of their homes.
Collaborative Care is both an in-person and virtual model that allows PCP practices to offer behavioral health services in a cost effective way by utilizing a psychiatrist as a “consultant” to the practice as opposed to hiring a full-time psychiatrist. All services within the Collaborative Care Model can be offered via telehealth, and all major insurance providers reimburse primary care providers for delivering Collaborative Care.
When PCPs provide behavioral health treatment as an “extension” of the primary care service offerings, the stigma is reduced and more patients are willing to accept the care they need.
Many areas of the country suffer from a lack of access to behavioral health specialists. In rural counties, for example, the nearest therapist may be located over an hour away. By integrating behavioral telehealth services into your practice’s offerings, you can remove geographic and transportation obstacles to care for your patient population.
Doing this can lead to providing more culturally competent care. It’s important that you’re able to offer mental health services to your patients from a professional with a similar ethnic or racial background. Language barriers and cultural differences may limit a provider’s ability to treat a patient, particularly if the patient faces health disparities related to race or ethnicity. If your practice needs to look outside of your community to tap into a more diverse pool of providers to better meet your patients’ needs, telehealth makes it easier to do that.
Adopting telemedicine for consultative patient visits offers primary care a path toward restoring patient volume and hope for a postpandemic future.
Mark Stephan, MD, is chief medical officer at Equality Health, a whole-health delivery system. He practiced family medicine for 19 years, including hospital medicine and obstetrics in rural and urban settings. Dr. Stephan has no conflicts related to the content of this piece.
By now it is well known that the COVID-19 pandemic has significantly disrupted primary care. Office visits and revenues have precipitously dropped as physicians and patients alike fear in-person visits may increase their risks of contracting the virus. However, telemedicine has emerged as a lifeline of sorts for many practices, enabling them to conduct visits and maintain contact with patients.
Telemedicine is likely to continue to serve as a tool for primary care providers to improve access to convenient, cost-effective, high-quality care after the pandemic. Another benefit of telemedicine is it can help maintain a portion of a practice’s revenue stream for physicians during uncertain times.
Indeed, the nation has seen recent progress toward telemedicine parity, which refers to the concept of reimbursing providers’ telehealth visits at the same rates as similar in-person visits.
A challenge to adopting telemedicine is that it calls for adjusting established workflows for in-person encounters. A practice cannot simply replicate in-person processes to work for telehealth. While both in-person and virtual visits require adherence to HIPAA, for example, how you actually protect patient privacy will call for different measures. Harking back to the early days of EMR implementation, one does not need to like the telemedicine platform or process, but come to terms with the fact that it is a tool that is here to stay to deliver patient care.
Treat your practice like a laboratory
Adoption may vary between practices depending on many factors, including clinicians’ comfort with technology, clinical tolerance and triage rules for nontouch encounters, state regulations, and more. Every provider group should begin experimenting with telemedicine in specific ways that make sense for them.
One physician may practice telemedicine full-time while the rest abstain, or perhaps the practice prefers to offer telemedicine services during specific hours on specific days. Don’t be afraid to start slowly when you’re trying something new – but do get started with telehealth. It will increasingly be a mainstream medium and more patients will come to expect it.
Train the entire team
Many primary care practices do not enjoy the resources of an information technology team, so all team members essentially need to learn the new skill of telemedicine usage, in addition to assisting patients. That can’t happen without staff buy-in, so it is essential that everyone from the office manager to medical assistants have the training they need to make the technology work. Juggling schedules for telehealth and in-office, activating an account through email, starting and joining a telehealth meeting, and preparing a patient for a visit are just a handful of basic tasks your staff should be trained to do to contribute to the successful integration of telehealth.
Educate and encourage patients to use telehealth
While unfamiliarity with technology may represent a roadblock for some patients, others resist telemedicine simply because no one has explained to them why it’s so important and the benefits it can hold for them. Education and communication are critical, including the sometimes painstaking work of slowly walking patients through the process of performing important functions on the telemedicine app. By providing them with some friendly coaching, patients won’t feel lost or abandoned during what for some may be an unfamiliar and frustrating process.
Manage more behavioral health
Different states and health plans incentivize primary practices for integrating behavioral health into their offerings. Rather than dismiss this addition to your own practice as too cumbersome to take on, I would recommend using telehealth to expand behavioral health care services.
If your practice is working toward a team-based, interdisciplinary approach to care delivery, behavioral health is a critical component. While other elements of this “whole person” health care may be better suited for an office visit, the vast majority of behavioral health services can be delivered virtually.
To decide if your patient may benefit from behavioral health care, the primary care provider (PCP) can conduct a screening via telehealth. Once the screening is complete, the PCP can discuss results and refer the patient to a mental health professional – all via telehealth. While patients may be reluctant to receive behavioral health treatment, perhaps because of stigma or inexperience, they may appreciate the telemedicine option as they can remain in the comfort and familiarity of their homes.
Collaborative Care is both an in-person and virtual model that allows PCP practices to offer behavioral health services in a cost effective way by utilizing a psychiatrist as a “consultant” to the practice as opposed to hiring a full-time psychiatrist. All services within the Collaborative Care Model can be offered via telehealth, and all major insurance providers reimburse primary care providers for delivering Collaborative Care.
When PCPs provide behavioral health treatment as an “extension” of the primary care service offerings, the stigma is reduced and more patients are willing to accept the care they need.
Many areas of the country suffer from a lack of access to behavioral health specialists. In rural counties, for example, the nearest therapist may be located over an hour away. By integrating behavioral telehealth services into your practice’s offerings, you can remove geographic and transportation obstacles to care for your patient population.
Doing this can lead to providing more culturally competent care. It’s important that you’re able to offer mental health services to your patients from a professional with a similar ethnic or racial background. Language barriers and cultural differences may limit a provider’s ability to treat a patient, particularly if the patient faces health disparities related to race or ethnicity. If your practice needs to look outside of your community to tap into a more diverse pool of providers to better meet your patients’ needs, telehealth makes it easier to do that.
Adopting telemedicine for consultative patient visits offers primary care a path toward restoring patient volume and hope for a postpandemic future.
Mark Stephan, MD, is chief medical officer at Equality Health, a whole-health delivery system. He practiced family medicine for 19 years, including hospital medicine and obstetrics in rural and urban settings. Dr. Stephan has no conflicts related to the content of this piece.
Chronicles of cancer: A history of mammography, part 1
Technological imperatives
The history of mammography provides a powerful example of the connection between social factors and the rise of a medical technology. It is also an object lesson in the profound difficulties that the medical community faces when trying to evaluate and embrace new discoveries in such a complex area as cancer diagnosis and treatment, especially when tied to issues of sex-based bias and gender identity. Given its profound ties to women’s lives and women’s bodies, mammography holds a unique place in the history of cancer. Part 1 will examine the technological imperatives driving mammography forward, and part 2 will address the social factors that promoted and inhibited the developing technology.
All that glitters
Innovations in technology have contributed so greatly to the progress of medical science in saving and improving patients’ lives that the lure of new technology and the desire to see it succeed and to embrace it has become profound.
In a debate on the adoption of new technologies, Michael Rosen, MD, a surgeon at the Cleveland Clinic, Ohio, pointed out the inherent risks in the life cycle of medical technology: “The stages of surgical innovation have been well described as moving from the generation of a hypothesis with an early promising report to being accepted conclusively as a new standard without formal testing. As the life cycle continues and comparative effectiveness data begin to emerge slowly through appropriately designed trials, the procedure or device is often ultimately abandoned.”1
The history of mammography bears out this grim warning in example after example as an object lesson, revealing not only the difficulties involved in the development of new medical technologies, but also the profound problems involved in validating the effectiveness and appropriateness of a new technology from its inception to the present.
A modern failure?
In fact, one of the more modern developments in mammography technology – digital imaging – has recently been called into question with regard to its effectiveness in saving lives, even as the technology continues to spread throughout the medical community.
A recent meta-analysis has shown that there is little or no improvement in outcomes of breast cancer screening when using digital analysis and screening mammograms vs. traditional film recording.
The meta-analysis assessed 24 studies with a combined total of 16,583,743 screening examinations (10,968,843 film and 5,614,900 digital). The study found that the difference in cancer detection rate using digital rather than film screening showed an increase of only 0.51 detections per 1,000 screens.
The researchers concluded “that while digital mammography is beneficial for medical facilities due to easier storage and handling of images, these results suggest the transition from film to digital mammography has not resulted in health benefits for screened women.”2
In fact, the researchers added that “This analysis reinforces the need to carefully evaluate effects of future changes in technology, such as tomosynthesis, to ensure new technology leads to improved health outcomes and beyond technical gains.”2
None of the nine main randomized clinical trials that were used to determine the effectiveness of mammography screening from the 1960s to the 1990s used digital or 3-D digital mammography (digital breast tomosynthesis or DBT). The earliest trial used direct-exposure film mammography and the others relied upon screen-film mammography.3 And yet the assumptions of the validity of the new digital technologies were predicated on the generalized acceptance of the validity of screening derived from these studies, and a corollary assumption that any technological improvement in the quality of the image must inherently be an improvement of the overall results of screening.
The failure of new technologies to meet expectations is a sobering corrective to the high hopes of researchers, practitioners, and patient groups alike, and is perhaps destined to contribute more to the parallel history of controversy and distrust concerning the risk/benefits of mammography that has been a media and scientific mainstay.
Too often the history of medical technology has found disappointment at the end of the road for new discoveries. But although the disappointing results of digital screening might be considered a failure in the progress of mammography, it is likely just another pause on the road of this technology, the history of which has been rocky from the start.
The need for a new way of looking
The rationale behind the original and continuing development of mammography is a simple one, common to all cancer screening methods – the belief that the earlier the detection of a cancer, the more likely it is to be treated effectively with the therapeutic regimens at hand. While there is some controversy regarding the cost-benefit ratio of screening, especially when therapies for breast cancer are not perfect and vary widely in expense and availability globally, the driving belief has been that mammography provides an outcomes benefit in allowing early surgical and chemoradiation therapy with a curative intent.
There were two main driving forces behind the early development of mammography. The first was the highly lethal nature of breast cancer, especially when it was caught too late and had spread too far to benefit from the only available option at the time – surgery. The second was the severity of the surgical treatment, the only therapeutic option at the time, and the distressing number of women who faced the radical mastectomy procedure pioneered by physicians William Stewart Halsted (1852-1922) at Johns Hopkins University, Baltimore, and Willy Meyer (1858-1932) in New York.
In 1894, in an era when the development of anesthetics and antisepsis made ever more difficult surgical procedures possible without inevitably killing the patient, both men separately published their results of a highly extensive operation that consisted of removal of the breast, chest muscles, and axillary lymph nodes.
As long as there was no presurgical method of determining the extent of a breast cancer’s spread, much less an ability to visually distinguish malignant from benign growths, this “better safe than sorry” approach became the default approach of an increasing number of surgeons, and the drastic solution of radical mastectomy was increasingly applied universally.
But in 1895, with the discovery of x-rays, medical science recognized a nearly miraculous technology for visualizing the inside of the body, and radioactive materials were also routinely used in medical therapies, by both legitimate practitioners and hucksters.
However, in the very early days, the users of x-rays were unaware that large radiation doses could have serious biological effects and had no way of determining radiation field strength and accumulating dosage.
In fact, early calibration of x-ray tubes was based on the amount of skin reddening (erythema) produced when the operator placed a hand directly in the x-ray beam.
It was in this environment that, within only a few decades, the new x-rays, especially with the development of improvements in mammography imaging, were able in many cases to identify smaller, more curable breast cancers. This eventually allowed surgeons to develop and use less extensive operations than the highly disfiguring radical mastectomy that was simultaneously dreaded for its invasiveness and embraced for its life-saving potential.4
Pioneering era
The technological history of mammography was thus driven by the quest for better imaging and reproducibility in order to further the hopes of curative surgical approaches.
In 1913, the German surgeon Albert Salomon (1883-1976) was the first to detect breast cancer using x-rays, but its clinical use was not established, as the images published in his “Beiträge zur pathologie und klinik der mammakarzinome (Contributions to the pathology and clinic of breast cancers)” were photographs of postsurgical breast specimens that illustrated the anatomy and spread of breast cancer tumors but were not adapted to presurgical screening.
After Salomon’s work was published in 1913, there was no new mammography literature published until 1927, when German surgeon Otto Kleinschmidt (1880-1948) published a report describing the world’s first authentic mammography, which he attributed to his mentor, the plastic surgeon Erwin Payr (1871-1946).5
This was followed soon after in 1930 by the work of radiologist Stafford L. Warren (1896-1981), of the University of Rochester (N.Y.), who published a paper on the use of standard roentgenograms for the in vivo preoperative assessment of breast malignancies. His technique involved the use of a stereoscopic system with a grid mechanism and intensifying screens to amplify the image. Breast compression was not involved in his mammogram technique. “Dr. Warren claimed to be correct 92% of the time when using this technique to predict malignancy.”5
His study of 119 women with a histopathologic diagnosis (61 benign and 58 malignant) demonstrated the feasibility of the technique for routine use and “created a surge of interest.”6
But the technology of the time proved difficult to use, and the results difficult to reproduce from laboratory to laboratory, and ultimately did not gain wide acceptance. Among Warren’s other claims to fame, he was a participant in the Manhattan Project and was a member of the teams sent to assess radiation damage in Hiroshima and Nagasaki after the dropping of the atomic bombs.
And in fact, future developments in mammography and all other x-ray screening techniques included attempts to minimize radiation exposure; such attempts were driven, in part, by the tragic impact of atomic bomb radiation and the medical studies carried out on the survivors.
An image more deadly than the disease
Further improvements in mammography technique occurred through the 1930s and 1940s, including better visualization of the mammary ducts based upon the pioneering studies of Emil Ries, MD, in Chicago, who, along with Nymphus Frederick Hicken, MD (1900-1998), reported on the use of contrast mammography (also known as ductography or galactography). On a side note, Dr. Hicken was responsible for introducing the terms mammogram and mammography in 1937.
Problems with ductography, which involved the injection of a radiographically opaque contrast agent into the nipple, occurred when the early contrast agents, such as oil-based lipiodol, proved to be toxic and capable of causing abscesses.7This advance led to the development of other agents, and among the most popular at the time was one that would prove deadly to many.
Thorotrast, first used in 1928, was widely embraced because of its lack of immediately noticeable side effects and the high-quality contrast it provided. Thorotrast was a suspension of radioactive thorium dioxide particles, which gained popularity for use as a radiological imaging agent from the 1930s to 1950s throughout the world, being used in an estimated 2-10 million radiographic exams, primarily for neurosurgery.
In the 1920s and 1930s, world governments had begun to recognize the dangers of radiation exposure, especially among workers, but thorotrast was a unique case because, unbeknownst to most practitioners at the time, thorium dioxide was retained in the body for the lifetime of the patient, with 70% deposited in the liver, 20% in the spleen, and the remaining in the bony medulla and in the peripheral lymph nodes.
Nineteen years after the first use of thorotrast, the first case of a human malignant tumor attributed to its exposure was reported. “Besides the liver neoplasm cases, aplastic anemia, leukemia and an impressive incidence of chromosome aberrations were registered in exposed individuals.”8
Despite its widespread adoption elsewhere, especially in Japan, the use of thorotrast never became popular in the United States, in part because in 1932 and 1937, warnings were issued by the American Medical Association to restrict its use.9
There was a shift to the use of iodinated hydrophilic molecules as contrast agents for conventional x-ray, computed tomography, and fluoroscopy procedures.9 However, it was discovered that these agents, too, have their own risks and dangerous side effects. They can cause severe adverse effects, including allergies, cardiovascular diseases, and nephrotoxicity in some patients.
Slow adoption and limited results
Between 1930 and 1950, Dr. Warren, Jacob Gershon-Cohen, MD (1899-1971) of Philadelphia, and radiologist Raul Leborgne of Uruguay “spread the gospel of mammography as an adjunct to physical examination for the diagnosis of breast cancer.”4 The latter also developed the breast compression technique to produce better quality images and lower the radiation exposure needed, and described the differences that could be visualized between benign and malign microcalcifications.
But despite the introduction of improvements such as double-emulsion film and breast compression to produce higher-quality images, “mammographic films often remained dark and hazy. Moreover, the new techniques, while improving the images, were not easily reproduced by other investigators and clinicians,” and therefore were still not widely adopted.4
Little noticeable effect of mammography
Although the technology existed and had its popularizers, mammography had little impact on an epidemiological level.
There was no major change in the mean maximum breast cancer tumor diameter and node positivity rate detected over the 20 years from 1929 to 1948.10 However, starting in the late 1940s, the American Cancer Society began public education campaigns and early detection education, and thereafter, there was a 3% decline in mean maximum diameter of tumor size seen every 10 years until 1968.
“We have interpreted this as the effect of public education and professional education about early detection through television, print media, and professional publications that began in 1947 because no other event was known to occur that would affect cancer detection beginning in the late 1940s.”10
However, the early detection methods at the time were self-examination and clinical examination for lumps, with mammography remaining a relatively limited tool until its general acceptance broadened a few decades later.
Robert Egan, “Father of Mammography,” et al.
The broad acceptance of mammography as a screening tool and its impacts on a broad population level resulted in large part from the work of Robert L. Egan, MD (1921-2001) in the late 1950s and 1960s.
Dr. Egan’s work was inspired in 1956 by a presentation by a visiting fellow, Jean Pierre Batiani, who brought a mammogram clearly showing a breast cancer from his institution, the Curie Foundation in Paris. The image had been made using very low kilowattage, high tube currents, and fine-grain film.
Dr. Egan, then a resident in radiology, was given the task by the head of his department of reproducing the results.
In 1959, Dr. Egan, then at the University of Texas MD Anderson Cancer Center, Houston, published a combined technique that used a high-milliamperage–low-voltage technique, a fine-grain intensifying screen, and single-emulsion films for mammography, thereby decreasing the radiation exposure significantly from previous x-ray techniques and improving the visualization and reproducibility of screening.
By 1960, Dr. Egan reported on 1,000 mammography cases at MD Anderson, demonstrating the ability of proper screening to detect unsuspected cancers and to limit mastectomies on benign masses. Of 245 breast cancers ultimately confirmed by biopsy, 238 were discovered by mammography, 19 of which were in women whose physical examinations had revealed no breast pathology. One of the cancers was only 8 mm in diameter when sectioned at biopsy.
Dr. Egan’s findings prompted an investigation by the Cancer Control Program (CCP) of the U.S. Public Health Service and led to a study jointly conducted by the National Cancer Institute and MD Anderson Hospital and the CCP, which involved 24 institutions and 1,500 patients.
“The results showed a 21% false-negative rate and a 79% true-positive rate for screening studies using Egan’s technique. This was a milestone for women’s imaging in the United States. Screening mammography was off to a tentative start.”5
“Egan was the man who developed a smooth-riding automobile compared to a Model T. He put mammography on the map and made it an intelligible, reproducible study. In short, he was the father of modern mammography,” according to his professor, mentor, and fellow mammography pioneer Gerald Dodd, MD (Emory School of Medicine website biography).
In 1964 Dr. Egan published his definitive book, “Mammography,” and in 1965 he hosted a 30-minute audiovisual presentation describing in detail his technique.11
The use of mammography was further powered by improved methods of preoperative needle localization, pioneered by Richard H. Gold, MD, in 1963 at Jefferson Medical College, Philadelphia, which eased obtaining a tissue diagnosis for any suspicious lesions detected in the mammogram. Dr. Gold performed needle localization of nonpalpable, mammographically visible lesions before biopsy, which allowed surgical resection of a smaller volume of breast tissue than was possible before.
Throughout the era, there were also incremental improvements in mammography machines and an increase in the number of commercial manufacturers.
Xeroradiography, an imaging technique adapted from xerographic photocopying, was seen as a major improvement over direct film imaging, and the technology became popular throughout the 1970s based on the research of John N. Wolfe, MD (1923-1993), who worked closely with the Xerox Corporation to improve the breast imaging process.6 However, this technology had all the same problems associated with running an office copying machine, including paper jams and toner issues, and the worst aspect was the high dose of radiation required. For this reason, it would quickly be superseded by the use of screen-film mammography, which eventually completely replaced the use of both xeromammography and direct-exposure film mammography.
The march of mammography
A series of nine randomized clinical trials (RCTs) between the 1960s and 1990s formed the foundation of the clinical use of mammography. These studies enrolled more than 600,000 women in the United States, Canada, the United Kingdom, and Sweden. The nine main RCTs of breast cancer screening were the Health Insurance Plan of Greater New York (HIP) trial, the Edinburgh trial, the Canadian National Breast Screening Study, the Canadian National Breast Screening Study 2, the United Kingdom Age trial, the Stockholm trial, the Malmö Mammographic Screening Trial, the Gothenburg trial, and the Swedish Two-County Study.3
These trials incorporated improvements in the technology as it developed, as seen in the fact that the earliest, the HIP trial, used direct-exposure film mammography and the other trials used screen-film mammography.3
Meta-analyses of the major nine screening trials indicated that reduced breast cancer mortality with screening was dependent on age. In particular, the results for women aged 40-49 years and 50-59 years showed only borderline statistical significance, and they varied depending on how cases were accrued in individual trials. “Assuming that differences actually exist, the absolute breast cancer mortality reduction per 10,000 women screened for 10 years ranged from 3 for age 39-49 years; 5-8 for age 50-59 years; and 12-21 for age 60-69 years.”3 In addition the estimates for women aged 70-74 years were limited by low numbers of events in trials that had smaller numbers of women in this age group.
However, at the time, the studies had a profound influence on increasing the popularity and spread of mammography.
As mammographies became more common, standardization became an important issue and a Mammography Accreditation Program began in 1987. Originally a voluntary program, it became mandatory with the Mammography Quality Standards Act of 1992, which required all U.S. mammography facilities to become accredited and certified.
In 1986, the American College of Radiology proposed its Breast Imaging Reporting and Data System (BI-RADS) initiative to enable standardized reporting of mammography; the first report was released in 1993.
BI-RADS is now on its fifth edition and has addressed the use of mammography, breast ultrasonography, and breast magnetic resonance imaging, developing standardized auditing approaches for all three techniques of breast cancer imaging.6
The digital era and beyond
With the dawn of the 21st century, the era of digital breast cancer screening began.
The screen-film mammography (SFM) technique employed throughout the 1980s and 1990s had significant advantages over earlier x-ray films for producing more vivid images of dense breast tissues. The next technology, digital mammography, was introduced in the late 20th century, and the first system was approved by the U.S. FDA in 2000.
One of the key benefits touted for digital mammograms is the fact that the radiologist can manipulate the contrast of the images, which allows for masses to be identified that might otherwise not be visible on standard film.
However, the recent meta-analysis discussed in the introduction calls such benefits into question, and a new controversy is likely to ensue on the question of the effectiveness of digital mammography on overall clinical outcomes.
But the technology continues to evolve.
“There has been a continuous and substantial technical development from SFM to full-field digital mammography and very recently also the introduction of digital breast tomosynthesis (DBT). This technical evolution calls for new evidence regarding the performance of screening using new mammography technologies, and the evidence needed to translate new technologies into screening practice,” according to an updated assessment by the U.S. Preventive Services Task Force.12
DBT was approved by the Food and Drug Administration in 2011. The technology involves the creation of a series of images, which are assembled into a 3-D–like image of breast slices. Traditional digital mammography creates a 2-D image of a flattened breast, and the radiologist must peer through the layers to find abnormalities. DBT uses a computer algorithm to reconstruct multiple low-dose digital images of the breast that can be displayed individually or in cinematic mode.13
Early trials showed a significant benefit of DBT in detecting new and smaller breast cancers, compared with standard digital mammography.
In women in their 40s, DBT found 1.7 more cancers than digital mammography for every 1,000 exams of women with normal breast tissue. In addition, 16.3% of women in this age group who were screened using digital mammography received callbacks, versus 11.7% of those screened using DBT. For younger women with dense breasts, the advantage of DBT was even greater, with 2.27 more cancers found for every 1,000 women screened. Whether such results will lead to clinically improved outcomes remains a question. “It can still miss cancers. Also, like traditional mammography, DBT might not reduce deaths from tumors that are very aggressive and fast-growing. And some women will still be called back unnecessarily for false-positive results.”14
But such technological advances further the hopes of researchers and patients alike.
Conclusion
Medical technology is driven both by advances in science and by the demands of patients and physicians for improved outcomes. The history of mammography, for example, is tied to the scientific advancements in x-ray technology, which allowed physicians for the first time to peer inside a living body without a scalpel at hand. But mammography was also an outgrowth of the profound need of the surgeon to identify cancerous masses in the breast at an early-enough stage to attempt a cure, while simultaneously minimizing the radical nature of the surgery required.
And while seeing is believing, the need to see and verify what was seen in order to make life-and-death decisions drove the demand for improvements in the technology of mammography throughout most of the 20th century and beyond.
The tortuous path from the early and continuing snafus with contrast agents to the apparent failure of the promise of digital technology serves as a continuing reminder of the hopes and perils that developing medical technologies present. It will be interesting to see if further refinements to mammography, such as DBT, will enhance the technology enough to have a major impact on countless women’s lives, or if new developments in magnetic resonance imaging and ultrasound make traditional mammography a relic of the past.
Part 2 of this history will present the social dynamics intimately involved with the rise and promulgation of mammography and how social need and public fears and controversies affected its development and spread as much, if not more, than technological innovation.
This article could only touch upon the myriad of details and technologies involved in the history of mammography, and I urge interested readers to check out the relevant references for far more in-depth and fascinating stories from its complex and controversial past.
References
1. Felix EL, Rosen M, Earle D. “Curbing Our Enthusiasm for Surgical Innovation: Is It a Good Thing or Bad Thing?” The Great Debates, General Surgery News, 2018 Oct 17
2. J Natl Cancer Inst. 2020 Jun 23. doi: 10.1093/jnci/djaa080.
3. Nelson H et al. Screening for Breast Cancer: A Systematic Review to Update the 2009 U.S. Preventive Services Task Force Recommendation. Evidence Synthesis No. 124. (Rockville, Md.: U.S. Agency for Healthcare Research and Quality, 2016 Jan, pp. 29-49)4. Lerner, BH. “To See Today With the Eyes of Tomorrow: A History of Screening Mammography,” background paper for Patlak M et al., Mammography and Beyond: Developing Technologies for the Early Detection of Breast Cancer (Washington: National Academies Press, 2001).
5. Grady I, Hansen P. Chapter 28: Mammography in “Kuerer’s Breast Surgical Oncology”(New York: McGaw-Hill Medical, 2010)
6. Radiology. 2014 Nov;273(2 Suppl):S23-44.
7. Bassett LW, Kim CH. (2003) Chapter 1: Ductography in Dershaw DD (eds) “Imaging-Guided Interventional Breast Techniques” (New York: Springer, 2003, pp. 1-30).
8. Cuperschmid EM, Ribeiro de Campos TP. 2009 International Nuclear Atlantic Conference, Rio de Janeiro, Sept 27–Oct 2, 2009
9. Bioscience Microflora. 2000;19(2):107-16.
10. Cady B. New era in breast cancer. Impact of screening on disease presentation. Surg Oncol Clin N Am. 1997 Apr;6(2):195-202.
11. Egan R. “Mammography Technique.” Audiovisual presentation. (Washington: U.S. Public Health Service, 1965).
12. Zackrisson S, Houssami N. Chapter 13: Evolution of Mammography Screening: From Film Screen to Digital Breast Tomosynthesis in “Breast Cancer Screening: An Examination of Scientific Evidence” (Cambridge, Mass.: Academic Press, 2016, pp. 323-46).13. Melnikow J et al. Screening for breast cancer with digital breast tomosynthesis. Evidence Synthesis No. 125 (Rockville, Md.: U.S. Agency for Healthcare Research and Quality, 2016 Jan).
14. Newer breast screening technology may spot more cancers. Harvard Women’s Health Watch online, June 2019.
Mark Lesney is the editor of Hematology News and the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has worked as a writer/editor for the American Chemical Society, and has served as an adjunct assistant professor in the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
Technological imperatives
Technological imperatives
The history of mammography provides a powerful example of the connection between social factors and the rise of a medical technology. It is also an object lesson in the profound difficulties that the medical community faces when trying to evaluate and embrace new discoveries in such a complex area as cancer diagnosis and treatment, especially when tied to issues of sex-based bias and gender identity. Given its profound ties to women’s lives and women’s bodies, mammography holds a unique place in the history of cancer. Part 1 will examine the technological imperatives driving mammography forward, and part 2 will address the social factors that promoted and inhibited the developing technology.
All that glitters
Innovations in technology have contributed so greatly to the progress of medical science in saving and improving patients’ lives that the lure of new technology and the desire to see it succeed and to embrace it has become profound.
In a debate on the adoption of new technologies, Michael Rosen, MD, a surgeon at the Cleveland Clinic, Ohio, pointed out the inherent risks in the life cycle of medical technology: “The stages of surgical innovation have been well described as moving from the generation of a hypothesis with an early promising report to being accepted conclusively as a new standard without formal testing. As the life cycle continues and comparative effectiveness data begin to emerge slowly through appropriately designed trials, the procedure or device is often ultimately abandoned.”1
The history of mammography bears out this grim warning in example after example as an object lesson, revealing not only the difficulties involved in the development of new medical technologies, but also the profound problems involved in validating the effectiveness and appropriateness of a new technology from its inception to the present.
A modern failure?
In fact, one of the more modern developments in mammography technology – digital imaging – has recently been called into question with regard to its effectiveness in saving lives, even as the technology continues to spread throughout the medical community.
A recent meta-analysis has shown that there is little or no improvement in outcomes of breast cancer screening when using digital analysis and screening mammograms vs. traditional film recording.
The meta-analysis assessed 24 studies with a combined total of 16,583,743 screening examinations (10,968,843 film and 5,614,900 digital). The study found that the difference in cancer detection rate using digital rather than film screening showed an increase of only 0.51 detections per 1,000 screens.
The researchers concluded “that while digital mammography is beneficial for medical facilities due to easier storage and handling of images, these results suggest the transition from film to digital mammography has not resulted in health benefits for screened women.”2
In fact, the researchers added that “This analysis reinforces the need to carefully evaluate effects of future changes in technology, such as tomosynthesis, to ensure new technology leads to improved health outcomes and beyond technical gains.”2
None of the nine main randomized clinical trials that were used to determine the effectiveness of mammography screening from the 1960s to the 1990s used digital or 3-D digital mammography (digital breast tomosynthesis or DBT). The earliest trial used direct-exposure film mammography and the others relied upon screen-film mammography.3 And yet the assumptions of the validity of the new digital technologies were predicated on the generalized acceptance of the validity of screening derived from these studies, and a corollary assumption that any technological improvement in the quality of the image must inherently be an improvement of the overall results of screening.
The failure of new technologies to meet expectations is a sobering corrective to the high hopes of researchers, practitioners, and patient groups alike, and is perhaps destined to contribute more to the parallel history of controversy and distrust concerning the risk/benefits of mammography that has been a media and scientific mainstay.
Too often the history of medical technology has found disappointment at the end of the road for new discoveries. But although the disappointing results of digital screening might be considered a failure in the progress of mammography, it is likely just another pause on the road of this technology, the history of which has been rocky from the start.
The need for a new way of looking
The rationale behind the original and continuing development of mammography is a simple one, common to all cancer screening methods – the belief that the earlier the detection of a cancer, the more likely it is to be treated effectively with the therapeutic regimens at hand. While there is some controversy regarding the cost-benefit ratio of screening, especially when therapies for breast cancer are not perfect and vary widely in expense and availability globally, the driving belief has been that mammography provides an outcomes benefit in allowing early surgical and chemoradiation therapy with a curative intent.
There were two main driving forces behind the early development of mammography. The first was the highly lethal nature of breast cancer, especially when it was caught too late and had spread too far to benefit from the only available option at the time – surgery. The second was the severity of the surgical treatment, the only therapeutic option at the time, and the distressing number of women who faced the radical mastectomy procedure pioneered by physicians William Stewart Halsted (1852-1922) at Johns Hopkins University, Baltimore, and Willy Meyer (1858-1932) in New York.
In 1894, in an era when the development of anesthetics and antisepsis made ever more difficult surgical procedures possible without inevitably killing the patient, both men separately published their results of a highly extensive operation that consisted of removal of the breast, chest muscles, and axillary lymph nodes.
As long as there was no presurgical method of determining the extent of a breast cancer’s spread, much less an ability to visually distinguish malignant from benign growths, this “better safe than sorry” approach became the default approach of an increasing number of surgeons, and the drastic solution of radical mastectomy was increasingly applied universally.
But in 1895, with the discovery of x-rays, medical science recognized a nearly miraculous technology for visualizing the inside of the body, and radioactive materials were also routinely used in medical therapies, by both legitimate practitioners and hucksters.
However, in the very early days, the users of x-rays were unaware that large radiation doses could have serious biological effects and had no way of determining radiation field strength and accumulating dosage.
In fact, early calibration of x-ray tubes was based on the amount of skin reddening (erythema) produced when the operator placed a hand directly in the x-ray beam.
It was in this environment that, within only a few decades, the new x-rays, especially with the development of improvements in mammography imaging, were able in many cases to identify smaller, more curable breast cancers. This eventually allowed surgeons to develop and use less extensive operations than the highly disfiguring radical mastectomy that was simultaneously dreaded for its invasiveness and embraced for its life-saving potential.4
Pioneering era
The technological history of mammography was thus driven by the quest for better imaging and reproducibility in order to further the hopes of curative surgical approaches.
In 1913, the German surgeon Albert Salomon (1883-1976) was the first to detect breast cancer using x-rays, but its clinical use was not established, as the images published in his “Beiträge zur pathologie und klinik der mammakarzinome (Contributions to the pathology and clinic of breast cancers)” were photographs of postsurgical breast specimens that illustrated the anatomy and spread of breast cancer tumors but were not adapted to presurgical screening.
After Salomon’s work was published in 1913, there was no new mammography literature published until 1927, when German surgeon Otto Kleinschmidt (1880-1948) published a report describing the world’s first authentic mammography, which he attributed to his mentor, the plastic surgeon Erwin Payr (1871-1946).5
This was followed soon after in 1930 by the work of radiologist Stafford L. Warren (1896-1981), of the University of Rochester (N.Y.), who published a paper on the use of standard roentgenograms for the in vivo preoperative assessment of breast malignancies. His technique involved the use of a stereoscopic system with a grid mechanism and intensifying screens to amplify the image. Breast compression was not involved in his mammogram technique. “Dr. Warren claimed to be correct 92% of the time when using this technique to predict malignancy.”5
His study of 119 women with a histopathologic diagnosis (61 benign and 58 malignant) demonstrated the feasibility of the technique for routine use and “created a surge of interest.”6
But the technology of the time proved difficult to use, and the results difficult to reproduce from laboratory to laboratory, and ultimately did not gain wide acceptance. Among Warren’s other claims to fame, he was a participant in the Manhattan Project and was a member of the teams sent to assess radiation damage in Hiroshima and Nagasaki after the dropping of the atomic bombs.
And in fact, future developments in mammography and all other x-ray screening techniques included attempts to minimize radiation exposure; such attempts were driven, in part, by the tragic impact of atomic bomb radiation and the medical studies carried out on the survivors.
An image more deadly than the disease
Further improvements in mammography technique occurred through the 1930s and 1940s, including better visualization of the mammary ducts based upon the pioneering studies of Emil Ries, MD, in Chicago, who, along with Nymphus Frederick Hicken, MD (1900-1998), reported on the use of contrast mammography (also known as ductography or galactography). On a side note, Dr. Hicken was responsible for introducing the terms mammogram and mammography in 1937.
Problems with ductography, which involved the injection of a radiographically opaque contrast agent into the nipple, occurred when the early contrast agents, such as oil-based lipiodol, proved to be toxic and capable of causing abscesses.7This advance led to the development of other agents, and among the most popular at the time was one that would prove deadly to many.
Thorotrast, first used in 1928, was widely embraced because of its lack of immediately noticeable side effects and the high-quality contrast it provided. Thorotrast was a suspension of radioactive thorium dioxide particles, which gained popularity for use as a radiological imaging agent from the 1930s to 1950s throughout the world, being used in an estimated 2-10 million radiographic exams, primarily for neurosurgery.
In the 1920s and 1930s, world governments had begun to recognize the dangers of radiation exposure, especially among workers, but thorotrast was a unique case because, unbeknownst to most practitioners at the time, thorium dioxide was retained in the body for the lifetime of the patient, with 70% deposited in the liver, 20% in the spleen, and the remaining in the bony medulla and in the peripheral lymph nodes.
Nineteen years after the first use of thorotrast, the first case of a human malignant tumor attributed to its exposure was reported. “Besides the liver neoplasm cases, aplastic anemia, leukemia and an impressive incidence of chromosome aberrations were registered in exposed individuals.”8
Despite its widespread adoption elsewhere, especially in Japan, the use of thorotrast never became popular in the United States, in part because in 1932 and 1937, warnings were issued by the American Medical Association to restrict its use.9
There was a shift to the use of iodinated hydrophilic molecules as contrast agents for conventional x-ray, computed tomography, and fluoroscopy procedures.9 However, it was discovered that these agents, too, have their own risks and dangerous side effects. They can cause severe adverse effects, including allergies, cardiovascular diseases, and nephrotoxicity in some patients.
Slow adoption and limited results
Between 1930 and 1950, Dr. Warren, Jacob Gershon-Cohen, MD (1899-1971) of Philadelphia, and radiologist Raul Leborgne of Uruguay “spread the gospel of mammography as an adjunct to physical examination for the diagnosis of breast cancer.”4 The latter also developed the breast compression technique to produce better quality images and lower the radiation exposure needed, and described the differences that could be visualized between benign and malign microcalcifications.
But despite the introduction of improvements such as double-emulsion film and breast compression to produce higher-quality images, “mammographic films often remained dark and hazy. Moreover, the new techniques, while improving the images, were not easily reproduced by other investigators and clinicians,” and therefore were still not widely adopted.4
Little noticeable effect of mammography
Although the technology existed and had its popularizers, mammography had little impact on an epidemiological level.
There was no major change in the mean maximum breast cancer tumor diameter and node positivity rate detected over the 20 years from 1929 to 1948.10 However, starting in the late 1940s, the American Cancer Society began public education campaigns and early detection education, and thereafter, there was a 3% decline in mean maximum diameter of tumor size seen every 10 years until 1968.
“We have interpreted this as the effect of public education and professional education about early detection through television, print media, and professional publications that began in 1947 because no other event was known to occur that would affect cancer detection beginning in the late 1940s.”10
However, the early detection methods at the time were self-examination and clinical examination for lumps, with mammography remaining a relatively limited tool until its general acceptance broadened a few decades later.
Robert Egan, “Father of Mammography,” et al.
The broad acceptance of mammography as a screening tool and its impacts on a broad population level resulted in large part from the work of Robert L. Egan, MD (1921-2001) in the late 1950s and 1960s.
Dr. Egan’s work was inspired in 1956 by a presentation by a visiting fellow, Jean Pierre Batiani, who brought a mammogram clearly showing a breast cancer from his institution, the Curie Foundation in Paris. The image had been made using very low kilowattage, high tube currents, and fine-grain film.
Dr. Egan, then a resident in radiology, was given the task by the head of his department of reproducing the results.
In 1959, Dr. Egan, then at the University of Texas MD Anderson Cancer Center, Houston, published a combined technique that used a high-milliamperage–low-voltage technique, a fine-grain intensifying screen, and single-emulsion films for mammography, thereby decreasing the radiation exposure significantly from previous x-ray techniques and improving the visualization and reproducibility of screening.
By 1960, Dr. Egan reported on 1,000 mammography cases at MD Anderson, demonstrating the ability of proper screening to detect unsuspected cancers and to limit mastectomies on benign masses. Of 245 breast cancers ultimately confirmed by biopsy, 238 were discovered by mammography, 19 of which were in women whose physical examinations had revealed no breast pathology. One of the cancers was only 8 mm in diameter when sectioned at biopsy.
Dr. Egan’s findings prompted an investigation by the Cancer Control Program (CCP) of the U.S. Public Health Service and led to a study jointly conducted by the National Cancer Institute and MD Anderson Hospital and the CCP, which involved 24 institutions and 1,500 patients.
“The results showed a 21% false-negative rate and a 79% true-positive rate for screening studies using Egan’s technique. This was a milestone for women’s imaging in the United States. Screening mammography was off to a tentative start.”5
“Egan was the man who developed a smooth-riding automobile compared to a Model T. He put mammography on the map and made it an intelligible, reproducible study. In short, he was the father of modern mammography,” according to his professor, mentor, and fellow mammography pioneer Gerald Dodd, MD (Emory School of Medicine website biography).
In 1964 Dr. Egan published his definitive book, “Mammography,” and in 1965 he hosted a 30-minute audiovisual presentation describing in detail his technique.11
The use of mammography was further powered by improved methods of preoperative needle localization, pioneered by Richard H. Gold, MD, in 1963 at Jefferson Medical College, Philadelphia, which eased obtaining a tissue diagnosis for any suspicious lesions detected in the mammogram. Dr. Gold performed needle localization of nonpalpable, mammographically visible lesions before biopsy, which allowed surgical resection of a smaller volume of breast tissue than was possible before.
Throughout the era, there were also incremental improvements in mammography machines and an increase in the number of commercial manufacturers.
Xeroradiography, an imaging technique adapted from xerographic photocopying, was seen as a major improvement over direct film imaging, and the technology became popular throughout the 1970s based on the research of John N. Wolfe, MD (1923-1993), who worked closely with the Xerox Corporation to improve the breast imaging process.6 However, this technology had all the same problems associated with running an office copying machine, including paper jams and toner issues, and the worst aspect was the high dose of radiation required. For this reason, it would quickly be superseded by the use of screen-film mammography, which eventually completely replaced the use of both xeromammography and direct-exposure film mammography.
The march of mammography
A series of nine randomized clinical trials (RCTs) between the 1960s and 1990s formed the foundation of the clinical use of mammography. These studies enrolled more than 600,000 women in the United States, Canada, the United Kingdom, and Sweden. The nine main RCTs of breast cancer screening were the Health Insurance Plan of Greater New York (HIP) trial, the Edinburgh trial, the Canadian National Breast Screening Study, the Canadian National Breast Screening Study 2, the United Kingdom Age trial, the Stockholm trial, the Malmö Mammographic Screening Trial, the Gothenburg trial, and the Swedish Two-County Study.3
These trials incorporated improvements in the technology as it developed, as seen in the fact that the earliest, the HIP trial, used direct-exposure film mammography and the other trials used screen-film mammography.3
Meta-analyses of the major nine screening trials indicated that reduced breast cancer mortality with screening was dependent on age. In particular, the results for women aged 40-49 years and 50-59 years showed only borderline statistical significance, and they varied depending on how cases were accrued in individual trials. “Assuming that differences actually exist, the absolute breast cancer mortality reduction per 10,000 women screened for 10 years ranged from 3 for age 39-49 years; 5-8 for age 50-59 years; and 12-21 for age 60-69 years.”3 In addition the estimates for women aged 70-74 years were limited by low numbers of events in trials that had smaller numbers of women in this age group.
However, at the time, the studies had a profound influence on increasing the popularity and spread of mammography.
As mammographies became more common, standardization became an important issue and a Mammography Accreditation Program began in 1987. Originally a voluntary program, it became mandatory with the Mammography Quality Standards Act of 1992, which required all U.S. mammography facilities to become accredited and certified.
In 1986, the American College of Radiology proposed its Breast Imaging Reporting and Data System (BI-RADS) initiative to enable standardized reporting of mammography; the first report was released in 1993.
BI-RADS is now on its fifth edition and has addressed the use of mammography, breast ultrasonography, and breast magnetic resonance imaging, developing standardized auditing approaches for all three techniques of breast cancer imaging.6
The digital era and beyond
With the dawn of the 21st century, the era of digital breast cancer screening began.
The screen-film mammography (SFM) technique employed throughout the 1980s and 1990s had significant advantages over earlier x-ray films for producing more vivid images of dense breast tissues. The next technology, digital mammography, was introduced in the late 20th century, and the first system was approved by the U.S. FDA in 2000.
One of the key benefits touted for digital mammograms is the fact that the radiologist can manipulate the contrast of the images, which allows for masses to be identified that might otherwise not be visible on standard film.
However, the recent meta-analysis discussed in the introduction calls such benefits into question, and a new controversy is likely to ensue on the question of the effectiveness of digital mammography on overall clinical outcomes.
But the technology continues to evolve.
“There has been a continuous and substantial technical development from SFM to full-field digital mammography and very recently also the introduction of digital breast tomosynthesis (DBT). This technical evolution calls for new evidence regarding the performance of screening using new mammography technologies, and the evidence needed to translate new technologies into screening practice,” according to an updated assessment by the U.S. Preventive Services Task Force.12
DBT was approved by the Food and Drug Administration in 2011. The technology involves the creation of a series of images, which are assembled into a 3-D–like image of breast slices. Traditional digital mammography creates a 2-D image of a flattened breast, and the radiologist must peer through the layers to find abnormalities. DBT uses a computer algorithm to reconstruct multiple low-dose digital images of the breast that can be displayed individually or in cinematic mode.13
Early trials showed a significant benefit of DBT in detecting new and smaller breast cancers, compared with standard digital mammography.
In women in their 40s, DBT found 1.7 more cancers than digital mammography for every 1,000 exams of women with normal breast tissue. In addition, 16.3% of women in this age group who were screened using digital mammography received callbacks, versus 11.7% of those screened using DBT. For younger women with dense breasts, the advantage of DBT was even greater, with 2.27 more cancers found for every 1,000 women screened. Whether such results will lead to clinically improved outcomes remains a question. “It can still miss cancers. Also, like traditional mammography, DBT might not reduce deaths from tumors that are very aggressive and fast-growing. And some women will still be called back unnecessarily for false-positive results.”14
But such technological advances further the hopes of researchers and patients alike.
Conclusion
Medical technology is driven both by advances in science and by the demands of patients and physicians for improved outcomes. The history of mammography, for example, is tied to the scientific advancements in x-ray technology, which allowed physicians for the first time to peer inside a living body without a scalpel at hand. But mammography was also an outgrowth of the profound need of the surgeon to identify cancerous masses in the breast at an early-enough stage to attempt a cure, while simultaneously minimizing the radical nature of the surgery required.
And while seeing is believing, the need to see and verify what was seen in order to make life-and-death decisions drove the demand for improvements in the technology of mammography throughout most of the 20th century and beyond.
The tortuous path from the early and continuing snafus with contrast agents to the apparent failure of the promise of digital technology serves as a continuing reminder of the hopes and perils that developing medical technologies present. It will be interesting to see if further refinements to mammography, such as DBT, will enhance the technology enough to have a major impact on countless women’s lives, or if new developments in magnetic resonance imaging and ultrasound make traditional mammography a relic of the past.
Part 2 of this history will present the social dynamics intimately involved with the rise and promulgation of mammography and how social need and public fears and controversies affected its development and spread as much, if not more, than technological innovation.
This article could only touch upon the myriad of details and technologies involved in the history of mammography, and I urge interested readers to check out the relevant references for far more in-depth and fascinating stories from its complex and controversial past.
References
1. Felix EL, Rosen M, Earle D. “Curbing Our Enthusiasm for Surgical Innovation: Is It a Good Thing or Bad Thing?” The Great Debates, General Surgery News, 2018 Oct 17
2. J Natl Cancer Inst. 2020 Jun 23. doi: 10.1093/jnci/djaa080.
3. Nelson H et al. Screening for Breast Cancer: A Systematic Review to Update the 2009 U.S. Preventive Services Task Force Recommendation. Evidence Synthesis No. 124. (Rockville, Md.: U.S. Agency for Healthcare Research and Quality, 2016 Jan, pp. 29-49)4. Lerner, BH. “To See Today With the Eyes of Tomorrow: A History of Screening Mammography,” background paper for Patlak M et al., Mammography and Beyond: Developing Technologies for the Early Detection of Breast Cancer (Washington: National Academies Press, 2001).
5. Grady I, Hansen P. Chapter 28: Mammography in “Kuerer’s Breast Surgical Oncology”(New York: McGaw-Hill Medical, 2010)
6. Radiology. 2014 Nov;273(2 Suppl):S23-44.
7. Bassett LW, Kim CH. (2003) Chapter 1: Ductography in Dershaw DD (eds) “Imaging-Guided Interventional Breast Techniques” (New York: Springer, 2003, pp. 1-30).
8. Cuperschmid EM, Ribeiro de Campos TP. 2009 International Nuclear Atlantic Conference, Rio de Janeiro, Sept 27–Oct 2, 2009
9. Bioscience Microflora. 2000;19(2):107-16.
10. Cady B. New era in breast cancer. Impact of screening on disease presentation. Surg Oncol Clin N Am. 1997 Apr;6(2):195-202.
11. Egan R. “Mammography Technique.” Audiovisual presentation. (Washington: U.S. Public Health Service, 1965).
12. Zackrisson S, Houssami N. Chapter 13: Evolution of Mammography Screening: From Film Screen to Digital Breast Tomosynthesis in “Breast Cancer Screening: An Examination of Scientific Evidence” (Cambridge, Mass.: Academic Press, 2016, pp. 323-46).13. Melnikow J et al. Screening for breast cancer with digital breast tomosynthesis. Evidence Synthesis No. 125 (Rockville, Md.: U.S. Agency for Healthcare Research and Quality, 2016 Jan).
14. Newer breast screening technology may spot more cancers. Harvard Women’s Health Watch online, June 2019.
Mark Lesney is the editor of Hematology News and the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has worked as a writer/editor for the American Chemical Society, and has served as an adjunct assistant professor in the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
The history of mammography provides a powerful example of the connection between social factors and the rise of a medical technology. It is also an object lesson in the profound difficulties that the medical community faces when trying to evaluate and embrace new discoveries in such a complex area as cancer diagnosis and treatment, especially when tied to issues of sex-based bias and gender identity. Given its profound ties to women’s lives and women’s bodies, mammography holds a unique place in the history of cancer. Part 1 will examine the technological imperatives driving mammography forward, and part 2 will address the social factors that promoted and inhibited the developing technology.
All that glitters
Innovations in technology have contributed so greatly to the progress of medical science in saving and improving patients’ lives that the lure of new technology and the desire to see it succeed and to embrace it has become profound.
In a debate on the adoption of new technologies, Michael Rosen, MD, a surgeon at the Cleveland Clinic, Ohio, pointed out the inherent risks in the life cycle of medical technology: “The stages of surgical innovation have been well described as moving from the generation of a hypothesis with an early promising report to being accepted conclusively as a new standard without formal testing. As the life cycle continues and comparative effectiveness data begin to emerge slowly through appropriately designed trials, the procedure or device is often ultimately abandoned.”1
The history of mammography bears out this grim warning in example after example as an object lesson, revealing not only the difficulties involved in the development of new medical technologies, but also the profound problems involved in validating the effectiveness and appropriateness of a new technology from its inception to the present.
A modern failure?
In fact, one of the more modern developments in mammography technology – digital imaging – has recently been called into question with regard to its effectiveness in saving lives, even as the technology continues to spread throughout the medical community.
A recent meta-analysis has shown that there is little or no improvement in outcomes of breast cancer screening when using digital analysis and screening mammograms vs. traditional film recording.
The meta-analysis assessed 24 studies with a combined total of 16,583,743 screening examinations (10,968,843 film and 5,614,900 digital). The study found that the difference in cancer detection rate using digital rather than film screening showed an increase of only 0.51 detections per 1,000 screens.
The researchers concluded “that while digital mammography is beneficial for medical facilities due to easier storage and handling of images, these results suggest the transition from film to digital mammography has not resulted in health benefits for screened women.”2
In fact, the researchers added that “This analysis reinforces the need to carefully evaluate effects of future changes in technology, such as tomosynthesis, to ensure new technology leads to improved health outcomes and beyond technical gains.”2
None of the nine main randomized clinical trials that were used to determine the effectiveness of mammography screening from the 1960s to the 1990s used digital or 3-D digital mammography (digital breast tomosynthesis or DBT). The earliest trial used direct-exposure film mammography and the others relied upon screen-film mammography.3 And yet the assumptions of the validity of the new digital technologies were predicated on the generalized acceptance of the validity of screening derived from these studies, and a corollary assumption that any technological improvement in the quality of the image must inherently be an improvement of the overall results of screening.
The failure of new technologies to meet expectations is a sobering corrective to the high hopes of researchers, practitioners, and patient groups alike, and is perhaps destined to contribute more to the parallel history of controversy and distrust concerning the risk/benefits of mammography that has been a media and scientific mainstay.
Too often the history of medical technology has found disappointment at the end of the road for new discoveries. But although the disappointing results of digital screening might be considered a failure in the progress of mammography, it is likely just another pause on the road of this technology, the history of which has been rocky from the start.
The need for a new way of looking
The rationale behind the original and continuing development of mammography is a simple one, common to all cancer screening methods – the belief that the earlier the detection of a cancer, the more likely it is to be treated effectively with the therapeutic regimens at hand. While there is some controversy regarding the cost-benefit ratio of screening, especially when therapies for breast cancer are not perfect and vary widely in expense and availability globally, the driving belief has been that mammography provides an outcomes benefit in allowing early surgical and chemoradiation therapy with a curative intent.
There were two main driving forces behind the early development of mammography. The first was the highly lethal nature of breast cancer, especially when it was caught too late and had spread too far to benefit from the only available option at the time – surgery. The second was the severity of the surgical treatment, the only therapeutic option at the time, and the distressing number of women who faced the radical mastectomy procedure pioneered by physicians William Stewart Halsted (1852-1922) at Johns Hopkins University, Baltimore, and Willy Meyer (1858-1932) in New York.
In 1894, in an era when the development of anesthetics and antisepsis made ever more difficult surgical procedures possible without inevitably killing the patient, both men separately published their results of a highly extensive operation that consisted of removal of the breast, chest muscles, and axillary lymph nodes.
As long as there was no presurgical method of determining the extent of a breast cancer’s spread, much less an ability to visually distinguish malignant from benign growths, this “better safe than sorry” approach became the default approach of an increasing number of surgeons, and the drastic solution of radical mastectomy was increasingly applied universally.
But in 1895, with the discovery of x-rays, medical science recognized a nearly miraculous technology for visualizing the inside of the body, and radioactive materials were also routinely used in medical therapies, by both legitimate practitioners and hucksters.
However, in the very early days, the users of x-rays were unaware that large radiation doses could have serious biological effects and had no way of determining radiation field strength and accumulating dosage.
In fact, early calibration of x-ray tubes was based on the amount of skin reddening (erythema) produced when the operator placed a hand directly in the x-ray beam.
It was in this environment that, within only a few decades, the new x-rays, especially with the development of improvements in mammography imaging, were able in many cases to identify smaller, more curable breast cancers. This eventually allowed surgeons to develop and use less extensive operations than the highly disfiguring radical mastectomy that was simultaneously dreaded for its invasiveness and embraced for its life-saving potential.4
Pioneering era
The technological history of mammography was thus driven by the quest for better imaging and reproducibility in order to further the hopes of curative surgical approaches.
In 1913, the German surgeon Albert Salomon (1883-1976) was the first to detect breast cancer using x-rays, but its clinical use was not established, as the images published in his “Beiträge zur pathologie und klinik der mammakarzinome (Contributions to the pathology and clinic of breast cancers)” were photographs of postsurgical breast specimens that illustrated the anatomy and spread of breast cancer tumors but were not adapted to presurgical screening.
After Salomon’s work was published in 1913, there was no new mammography literature published until 1927, when German surgeon Otto Kleinschmidt (1880-1948) published a report describing the world’s first authentic mammography, which he attributed to his mentor, the plastic surgeon Erwin Payr (1871-1946).5
This was followed soon after in 1930 by the work of radiologist Stafford L. Warren (1896-1981), of the University of Rochester (N.Y.), who published a paper on the use of standard roentgenograms for the in vivo preoperative assessment of breast malignancies. His technique involved the use of a stereoscopic system with a grid mechanism and intensifying screens to amplify the image. Breast compression was not involved in his mammogram technique. “Dr. Warren claimed to be correct 92% of the time when using this technique to predict malignancy.”5
His study of 119 women with a histopathologic diagnosis (61 benign and 58 malignant) demonstrated the feasibility of the technique for routine use and “created a surge of interest.”6
But the technology of the time proved difficult to use, and the results difficult to reproduce from laboratory to laboratory, and ultimately did not gain wide acceptance. Among Warren’s other claims to fame, he was a participant in the Manhattan Project and was a member of the teams sent to assess radiation damage in Hiroshima and Nagasaki after the dropping of the atomic bombs.
And in fact, future developments in mammography and all other x-ray screening techniques included attempts to minimize radiation exposure; such attempts were driven, in part, by the tragic impact of atomic bomb radiation and the medical studies carried out on the survivors.
An image more deadly than the disease
Further improvements in mammography technique occurred through the 1930s and 1940s, including better visualization of the mammary ducts based upon the pioneering studies of Emil Ries, MD, in Chicago, who, along with Nymphus Frederick Hicken, MD (1900-1998), reported on the use of contrast mammography (also known as ductography or galactography). On a side note, Dr. Hicken was responsible for introducing the terms mammogram and mammography in 1937.
Problems with ductography, which involved the injection of a radiographically opaque contrast agent into the nipple, occurred when the early contrast agents, such as oil-based lipiodol, proved to be toxic and capable of causing abscesses.7This advance led to the development of other agents, and among the most popular at the time was one that would prove deadly to many.
Thorotrast, first used in 1928, was widely embraced because of its lack of immediately noticeable side effects and the high-quality contrast it provided. Thorotrast was a suspension of radioactive thorium dioxide particles, which gained popularity for use as a radiological imaging agent from the 1930s to 1950s throughout the world, being used in an estimated 2-10 million radiographic exams, primarily for neurosurgery.
In the 1920s and 1930s, world governments had begun to recognize the dangers of radiation exposure, especially among workers, but thorotrast was a unique case because, unbeknownst to most practitioners at the time, thorium dioxide was retained in the body for the lifetime of the patient, with 70% deposited in the liver, 20% in the spleen, and the remaining in the bony medulla and in the peripheral lymph nodes.
Nineteen years after the first use of thorotrast, the first case of a human malignant tumor attributed to its exposure was reported. “Besides the liver neoplasm cases, aplastic anemia, leukemia and an impressive incidence of chromosome aberrations were registered in exposed individuals.”8
Despite its widespread adoption elsewhere, especially in Japan, the use of thorotrast never became popular in the United States, in part because in 1932 and 1937, warnings were issued by the American Medical Association to restrict its use.9
There was a shift to the use of iodinated hydrophilic molecules as contrast agents for conventional x-ray, computed tomography, and fluoroscopy procedures.9 However, it was discovered that these agents, too, have their own risks and dangerous side effects. They can cause severe adverse effects, including allergies, cardiovascular diseases, and nephrotoxicity in some patients.
Slow adoption and limited results
Between 1930 and 1950, Dr. Warren, Jacob Gershon-Cohen, MD (1899-1971) of Philadelphia, and radiologist Raul Leborgne of Uruguay “spread the gospel of mammography as an adjunct to physical examination for the diagnosis of breast cancer.”4 The latter also developed the breast compression technique to produce better quality images and lower the radiation exposure needed, and described the differences that could be visualized between benign and malign microcalcifications.
But despite the introduction of improvements such as double-emulsion film and breast compression to produce higher-quality images, “mammographic films often remained dark and hazy. Moreover, the new techniques, while improving the images, were not easily reproduced by other investigators and clinicians,” and therefore were still not widely adopted.4
Little noticeable effect of mammography
Although the technology existed and had its popularizers, mammography had little impact on an epidemiological level.
There was no major change in the mean maximum breast cancer tumor diameter and node positivity rate detected over the 20 years from 1929 to 1948.10 However, starting in the late 1940s, the American Cancer Society began public education campaigns and early detection education, and thereafter, there was a 3% decline in mean maximum diameter of tumor size seen every 10 years until 1968.
“We have interpreted this as the effect of public education and professional education about early detection through television, print media, and professional publications that began in 1947 because no other event was known to occur that would affect cancer detection beginning in the late 1940s.”10
However, the early detection methods at the time were self-examination and clinical examination for lumps, with mammography remaining a relatively limited tool until its general acceptance broadened a few decades later.
Robert Egan, “Father of Mammography,” et al.
The broad acceptance of mammography as a screening tool and its impacts on a broad population level resulted in large part from the work of Robert L. Egan, MD (1921-2001) in the late 1950s and 1960s.
Dr. Egan’s work was inspired in 1956 by a presentation by a visiting fellow, Jean Pierre Batiani, who brought a mammogram clearly showing a breast cancer from his institution, the Curie Foundation in Paris. The image had been made using very low kilowattage, high tube currents, and fine-grain film.
Dr. Egan, then a resident in radiology, was given the task by the head of his department of reproducing the results.
In 1959, Dr. Egan, then at the University of Texas MD Anderson Cancer Center, Houston, published a combined technique that used a high-milliamperage–low-voltage technique, a fine-grain intensifying screen, and single-emulsion films for mammography, thereby decreasing the radiation exposure significantly from previous x-ray techniques and improving the visualization and reproducibility of screening.
By 1960, Dr. Egan reported on 1,000 mammography cases at MD Anderson, demonstrating the ability of proper screening to detect unsuspected cancers and to limit mastectomies on benign masses. Of 245 breast cancers ultimately confirmed by biopsy, 238 were discovered by mammography, 19 of which were in women whose physical examinations had revealed no breast pathology. One of the cancers was only 8 mm in diameter when sectioned at biopsy.
Dr. Egan’s findings prompted an investigation by the Cancer Control Program (CCP) of the U.S. Public Health Service and led to a study jointly conducted by the National Cancer Institute and MD Anderson Hospital and the CCP, which involved 24 institutions and 1,500 patients.
“The results showed a 21% false-negative rate and a 79% true-positive rate for screening studies using Egan’s technique. This was a milestone for women’s imaging in the United States. Screening mammography was off to a tentative start.”5
“Egan was the man who developed a smooth-riding automobile compared to a Model T. He put mammography on the map and made it an intelligible, reproducible study. In short, he was the father of modern mammography,” according to his professor, mentor, and fellow mammography pioneer Gerald Dodd, MD (Emory School of Medicine website biography).
In 1964 Dr. Egan published his definitive book, “Mammography,” and in 1965 he hosted a 30-minute audiovisual presentation describing in detail his technique.11
The use of mammography was further powered by improved methods of preoperative needle localization, pioneered by Richard H. Gold, MD, in 1963 at Jefferson Medical College, Philadelphia, which eased obtaining a tissue diagnosis for any suspicious lesions detected in the mammogram. Dr. Gold performed needle localization of nonpalpable, mammographically visible lesions before biopsy, which allowed surgical resection of a smaller volume of breast tissue than was possible before.
Throughout the era, there were also incremental improvements in mammography machines and an increase in the number of commercial manufacturers.
Xeroradiography, an imaging technique adapted from xerographic photocopying, was seen as a major improvement over direct film imaging, and the technology became popular throughout the 1970s based on the research of John N. Wolfe, MD (1923-1993), who worked closely with the Xerox Corporation to improve the breast imaging process.6 However, this technology had all the same problems associated with running an office copying machine, including paper jams and toner issues, and the worst aspect was the high dose of radiation required. For this reason, it would quickly be superseded by the use of screen-film mammography, which eventually completely replaced the use of both xeromammography and direct-exposure film mammography.
The march of mammography
A series of nine randomized clinical trials (RCTs) between the 1960s and 1990s formed the foundation of the clinical use of mammography. These studies enrolled more than 600,000 women in the United States, Canada, the United Kingdom, and Sweden. The nine main RCTs of breast cancer screening were the Health Insurance Plan of Greater New York (HIP) trial, the Edinburgh trial, the Canadian National Breast Screening Study, the Canadian National Breast Screening Study 2, the United Kingdom Age trial, the Stockholm trial, the Malmö Mammographic Screening Trial, the Gothenburg trial, and the Swedish Two-County Study.3
These trials incorporated improvements in the technology as it developed, as seen in the fact that the earliest, the HIP trial, used direct-exposure film mammography and the other trials used screen-film mammography.3
Meta-analyses of the major nine screening trials indicated that reduced breast cancer mortality with screening was dependent on age. In particular, the results for women aged 40-49 years and 50-59 years showed only borderline statistical significance, and they varied depending on how cases were accrued in individual trials. “Assuming that differences actually exist, the absolute breast cancer mortality reduction per 10,000 women screened for 10 years ranged from 3 for age 39-49 years; 5-8 for age 50-59 years; and 12-21 for age 60-69 years.”3 In addition the estimates for women aged 70-74 years were limited by low numbers of events in trials that had smaller numbers of women in this age group.
However, at the time, the studies had a profound influence on increasing the popularity and spread of mammography.
As mammographies became more common, standardization became an important issue and a Mammography Accreditation Program began in 1987. Originally a voluntary program, it became mandatory with the Mammography Quality Standards Act of 1992, which required all U.S. mammography facilities to become accredited and certified.
In 1986, the American College of Radiology proposed its Breast Imaging Reporting and Data System (BI-RADS) initiative to enable standardized reporting of mammography; the first report was released in 1993.
BI-RADS is now on its fifth edition and has addressed the use of mammography, breast ultrasonography, and breast magnetic resonance imaging, developing standardized auditing approaches for all three techniques of breast cancer imaging.6
The digital era and beyond
With the dawn of the 21st century, the era of digital breast cancer screening began.
The screen-film mammography (SFM) technique employed throughout the 1980s and 1990s had significant advantages over earlier x-ray films for producing more vivid images of dense breast tissues. The next technology, digital mammography, was introduced in the late 20th century, and the first system was approved by the U.S. FDA in 2000.
One of the key benefits touted for digital mammograms is the fact that the radiologist can manipulate the contrast of the images, which allows for masses to be identified that might otherwise not be visible on standard film.
However, the recent meta-analysis discussed in the introduction calls such benefits into question, and a new controversy is likely to ensue on the question of the effectiveness of digital mammography on overall clinical outcomes.
But the technology continues to evolve.
“There has been a continuous and substantial technical development from SFM to full-field digital mammography and very recently also the introduction of digital breast tomosynthesis (DBT). This technical evolution calls for new evidence regarding the performance of screening using new mammography technologies, and the evidence needed to translate new technologies into screening practice,” according to an updated assessment by the U.S. Preventive Services Task Force.12
DBT was approved by the Food and Drug Administration in 2011. The technology involves the creation of a series of images, which are assembled into a 3-D–like image of breast slices. Traditional digital mammography creates a 2-D image of a flattened breast, and the radiologist must peer through the layers to find abnormalities. DBT uses a computer algorithm to reconstruct multiple low-dose digital images of the breast that can be displayed individually or in cinematic mode.13
Early trials showed a significant benefit of DBT in detecting new and smaller breast cancers, compared with standard digital mammography.
In women in their 40s, DBT found 1.7 more cancers than digital mammography for every 1,000 exams of women with normal breast tissue. In addition, 16.3% of women in this age group who were screened using digital mammography received callbacks, versus 11.7% of those screened using DBT. For younger women with dense breasts, the advantage of DBT was even greater, with 2.27 more cancers found for every 1,000 women screened. Whether such results will lead to clinically improved outcomes remains a question. “It can still miss cancers. Also, like traditional mammography, DBT might not reduce deaths from tumors that are very aggressive and fast-growing. And some women will still be called back unnecessarily for false-positive results.”14
But such technological advances further the hopes of researchers and patients alike.
Conclusion
Medical technology is driven both by advances in science and by the demands of patients and physicians for improved outcomes. The history of mammography, for example, is tied to the scientific advancements in x-ray technology, which allowed physicians for the first time to peer inside a living body without a scalpel at hand. But mammography was also an outgrowth of the profound need of the surgeon to identify cancerous masses in the breast at an early-enough stage to attempt a cure, while simultaneously minimizing the radical nature of the surgery required.
And while seeing is believing, the need to see and verify what was seen in order to make life-and-death decisions drove the demand for improvements in the technology of mammography throughout most of the 20th century and beyond.
The tortuous path from the early and continuing snafus with contrast agents to the apparent failure of the promise of digital technology serves as a continuing reminder of the hopes and perils that developing medical technologies present. It will be interesting to see if further refinements to mammography, such as DBT, will enhance the technology enough to have a major impact on countless women’s lives, or if new developments in magnetic resonance imaging and ultrasound make traditional mammography a relic of the past.
Part 2 of this history will present the social dynamics intimately involved with the rise and promulgation of mammography and how social need and public fears and controversies affected its development and spread as much, if not more, than technological innovation.
This article could only touch upon the myriad of details and technologies involved in the history of mammography, and I urge interested readers to check out the relevant references for far more in-depth and fascinating stories from its complex and controversial past.
References
1. Felix EL, Rosen M, Earle D. “Curbing Our Enthusiasm for Surgical Innovation: Is It a Good Thing or Bad Thing?” The Great Debates, General Surgery News, 2018 Oct 17
2. J Natl Cancer Inst. 2020 Jun 23. doi: 10.1093/jnci/djaa080.
3. Nelson H et al. Screening for Breast Cancer: A Systematic Review to Update the 2009 U.S. Preventive Services Task Force Recommendation. Evidence Synthesis No. 124. (Rockville, Md.: U.S. Agency for Healthcare Research and Quality, 2016 Jan, pp. 29-49)4. Lerner, BH. “To See Today With the Eyes of Tomorrow: A History of Screening Mammography,” background paper for Patlak M et al., Mammography and Beyond: Developing Technologies for the Early Detection of Breast Cancer (Washington: National Academies Press, 2001).
5. Grady I, Hansen P. Chapter 28: Mammography in “Kuerer’s Breast Surgical Oncology”(New York: McGaw-Hill Medical, 2010)
6. Radiology. 2014 Nov;273(2 Suppl):S23-44.
7. Bassett LW, Kim CH. (2003) Chapter 1: Ductography in Dershaw DD (eds) “Imaging-Guided Interventional Breast Techniques” (New York: Springer, 2003, pp. 1-30).
8. Cuperschmid EM, Ribeiro de Campos TP. 2009 International Nuclear Atlantic Conference, Rio de Janeiro, Sept 27–Oct 2, 2009
9. Bioscience Microflora. 2000;19(2):107-16.
10. Cady B. New era in breast cancer. Impact of screening on disease presentation. Surg Oncol Clin N Am. 1997 Apr;6(2):195-202.
11. Egan R. “Mammography Technique.” Audiovisual presentation. (Washington: U.S. Public Health Service, 1965).
12. Zackrisson S, Houssami N. Chapter 13: Evolution of Mammography Screening: From Film Screen to Digital Breast Tomosynthesis in “Breast Cancer Screening: An Examination of Scientific Evidence” (Cambridge, Mass.: Academic Press, 2016, pp. 323-46).13. Melnikow J et al. Screening for breast cancer with digital breast tomosynthesis. Evidence Synthesis No. 125 (Rockville, Md.: U.S. Agency for Healthcare Research and Quality, 2016 Jan).
14. Newer breast screening technology may spot more cancers. Harvard Women’s Health Watch online, June 2019.
Mark Lesney is the editor of Hematology News and the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has worked as a writer/editor for the American Chemical Society, and has served as an adjunct assistant professor in the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
Welcome to HM20 Virtual
Welcome to the HM20 virtual conference! We’re glad to have you join us to virtually experience sessions from our most popular SHM annual conference tracks including Rapid Fire, Clinical Updates, and High-Value Care! We also have added some new timely topics given our current times that you won’t want to miss. We encourage you to engage with the larger community via social media at #HM20Virtual.
HM20 in San Diego, scheduled originally for April 2020, was trending to be the highest in-person attended SHM annual conference with a fantastic line-up of offerings. Unfortunately, then came our pandemic, or pandemics. In mid-March, the Society of Hospital Medicine board of directors concluded that it was impossible for SHM to move forward with Hospital Medicine 2020 in San Diego because of the continued spread of COVID-19. Canceling the in-person conference during this unprecedented time was the right thing to do. I have valued the SHM leadership team and the larger SHM community for their support in being even more engaged on the front lines and with each other across our world during this time.
The COVID-19 pandemic has created a systemic challenge for health care systems across the nation. As hospitalists continue to be on the front lines of care and also innovations, organizations have leveraged telemedicine to support their patients, protect their clinicians, and conserve scarce resources. It is hospital medicine that has been on the front lines of change and adaptations and have led in this pandemic in many organizations across the nation and the world.
Unfortunately, known health disparities have also been amplified and there came an acute worsening of the chronic issues in this nation. On March 13, 2020, 26-year-old Breonna Taylor was shot after police forcibly entered her home. Armaud Arbery was shot and killed by armed neighbors while running through a neighborhood in Brunswick, Ga. Then on May 25, 5 miles from where I call home here in the Twin Cities in Minnesota, George Floyd, a 46-year-old father arrested for suspected use of a counterfeit $20 bill, died after police kneeled on his neck for over 8 minutes. This pandemic has also shaken up the status quo and laid bare a lot of our country’s long and deep-seated issues – from massive economic inequities to ongoing racial disparities to immigration concerns. It’s woken a lot of our valued hospitalists to the fact that the old ways of doing things just don’t work.
I’m grateful our society has taken steps to speak into these timely topics, and to share via publications, Twitter chats, advocacy items, and more! I want to encourage all of us to use the immense network of our hospitalist communities to comfort each other, learn, grow, and engage. We have not achieved big changes by ourselves. We’ve created valued offerings and innovative changes, and we’ve led on the front lines, in policies and procedures, by doing it together. Meaningful change requires allies in a common cause. We stand with our black and brown brothers and sisters who are particularly attuned to injustice, inequality, and struggle. We in hospital medicine stand up with many others who are struggling, our African American, Latin American, Native American, immigrant, LGBTQ+ communities. This intersection of the crisis of the COVID-19 pandemic and the racism pandemic have led us to a pivotal point in the arc of change and justice. I invite you to comfort each other, learn from each other, and act together in this community. To this end we have included timely resources in our HM20 virtual offering on these topics.
This year has been a big transition year. Not only did 2020 usher in a new decade, along with COVID-19 and our double pandemic, SHM has also had important transitions within its senior leadership. We say farewell to Larry Wellikson, MD, who has been at the helm of SHM since the beginning. On behalf of this annual conference, we want to celebrate and thank you, Larry, for your years of dedication and service to SHM. You have taken the specialty of hospital medicine and created a movement in SHM, where the entire hospital medicine team may gather under a bigger tent for education, community, and for the betterment of care for our patients.
We extend a welcome to Eric Howell, MD, who succeeds Dr. Wellikson as SHM’s CEO. We also welcome Danielle Scheurer, MD, as the new SHM president, succeeding the great leadership offered this past year by Christopher Frost, MD. In addition, Jerome C. Siy, MD, was voted president-elect, Dr. Rachel Thompson, MD, was elected treasurer, Kris Rehm, MD, was voted secretary, and Darlene Tad-y, MD, was elected to the board of directors. We welcome these new officers.
HM20 Virtual will consist of prerecorded on-demand sessions that can be viewed at your convenience as well as live Q&A and attendee networking that will take place during specific dates/times. A few of the top-rated sessions from our historically popular tracks include: Update in Clinical Practice Guidelines, Antibiotics Made Ridiculously Simple, Getting to Know Oncology Emergencies, Inpatient Pain Management in the Era of the Opioid Epidemic, Updates in Heart Failure, and Hyponatremia: Don’t Drink the Water. Additionally, we have some of our perennial favorites including the Update in Hospital Medicine and Top Pediatric Articles of 2019. There will be COVID-19 specific content from expertise throughout the nation focusing on care pathways, clinical updates, telemedicine, point-of-care ultrasound, and more! To view the HM20 Virtual Opening Session and discover what you can expect in this educational experience, click here.
The Journal of Hospital Medicine has had a large presence in our meetings for many years. We are grateful for Samir Shah, MD, and his leadership during this double pandemic, for identifying areas where we can advance the field responsibly in the face of relatively limited evidence, and rapidly evolving news. As part of his commitment, all JHM articles related to COVID-19 and published during the pandemic are open access. A pre-COVID goal that has been realized during the pandemic was to bring more of the journal into our annual conference and the conference contents into the journal. We are proud to say this has been a great collaboration, particularly during this pandemic, and much thanks to Dr. Shah’s leadership for highlighting timely pieces. Kimberly Manning, MD, had an especially powerful piece on the topic of racism and our double pandemic, and she is a featured speaker during our HM20 Virtual offering, under the same title as her article: “When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics.” Additionally, Manpreet Malik, MD, and I will be copresenting on a timely topic about the “Immigrant Hospitalist during COVID-19.”
Aside from these sessions for HM20 Virtual, the real can’t miss(es) for the conference are the Research, Innovations, and Clinical Vignette (RIV) posters sessions. I am grateful for the leadership of Stephanie Mueller, MD, who served as chair for this year’s RIV. This unique year has led to the hosting of a virtual poster competition with judging and the opening of a virtual gallery. We are so pleased to be able to share and highlight the work of many of learners and staff hospitalists! I love that a hospitalist on one side of the country can help provide pearls on a case, an innovation, or a research idea that can help improve diagnosis for a patient at the other side of the country. Keep an eye on SHM’s social media and the presentation by Dr. Mueller for announcements of the winners.
A favorite reason many of us attend the annual conference is for the people and community. We wanted to keep this value as we shifted to a virtual offering. Networking will occur through a variety of offerings including Simulive sessions and Special Interest Forums. Simulive sessions will run for 3 weeks from August 11 to August 27. For those of you new to the term, Simulive may sound like a made-up word, but it is an actual amalgamation of a prerecorded webinar and a live interaction (simulated + live = Simulive). Simulive allows the faculty to sit in on their prerecorded session and interact with the audience via the chat feature during the live scheduled recording and spend time afterwards for a live Q&A from the audience.
There will also be over 20 Special Interest Forums hosted in the evenings after these Simulive sessions have concluded to give you a chance to connect with individuals, share experiences, and have meaningful discussions that can directly impact your practice. Samples of the forums include: Diversity and Inclusion, Rural Hospital Medicine, Pediatrics, NP/PA, Perioperative and Comanagement, Health Information Technology, and Point of Care Ultrasound! Take a look at the HM20 registration page for further information. You will receive direct information on how to attend. We encourage you to join!
HM20 also features a virtual 5K! Whether you run on a treadmill or jog in your neighborhood or local park, you can participate in HM20’s Virtual Fun Run or Walk. To participate, simply run your 5K during the weeks of HM20 Virtual and when you’re done, fill out our form to log your time. We encourage you to post a picture on social media as well with #HM20Virtual. You’ll also receive a certificate of completion at the close of HM20 Virtual.
All HM20 Virtual sessions will be available as on-demand after August 31. HM20 virtual offers more than 60 CME hours and over 35 MOC hours that you can claim at your convenience! That’s the most amount of CME and MOC ever offered at SHM for an event! This conference would not be possible without the tireless and relentless effort of SHM staff and leadership, our terrific speakers and faculty, and all the volunteer committee members of SHM. A huge thanks to the Annual Conference Committee who had the charge to develop the content for the Annual Conference, including topics, speakers, and learning objectives. I am grateful to have had the opportunity to serve on this committee for the past 7 years and to lead HM20 this year. Thanks to Brittany Evans, Hayleigh Lawrence, and Michelle Kann for their valued support this past year from an SHM staff perspective; to my assistant course director, Dan Steinberg, MD; and to the immediate past course director, Dustin Smith, MD, for their support.
Once again, we are excited to have you join, and we hope this conference elevates your education in hospital medicine, advances your career, stimulates innovative thinking, and provides you with enduring networking opportunities. We sincerely thank you for attending HM20 Virtual. Welcome!
Dr. Mathews is chief of hospital medicine at Regions Hospital, HealthPartners in St. Paul, Minn., an associate professor at the University of Minnesota, Minneapolis, and course director of HM20.
Welcome to the HM20 virtual conference! We’re glad to have you join us to virtually experience sessions from our most popular SHM annual conference tracks including Rapid Fire, Clinical Updates, and High-Value Care! We also have added some new timely topics given our current times that you won’t want to miss. We encourage you to engage with the larger community via social media at #HM20Virtual.
HM20 in San Diego, scheduled originally for April 2020, was trending to be the highest in-person attended SHM annual conference with a fantastic line-up of offerings. Unfortunately, then came our pandemic, or pandemics. In mid-March, the Society of Hospital Medicine board of directors concluded that it was impossible for SHM to move forward with Hospital Medicine 2020 in San Diego because of the continued spread of COVID-19. Canceling the in-person conference during this unprecedented time was the right thing to do. I have valued the SHM leadership team and the larger SHM community for their support in being even more engaged on the front lines and with each other across our world during this time.
The COVID-19 pandemic has created a systemic challenge for health care systems across the nation. As hospitalists continue to be on the front lines of care and also innovations, organizations have leveraged telemedicine to support their patients, protect their clinicians, and conserve scarce resources. It is hospital medicine that has been on the front lines of change and adaptations and have led in this pandemic in many organizations across the nation and the world.
Unfortunately, known health disparities have also been amplified and there came an acute worsening of the chronic issues in this nation. On March 13, 2020, 26-year-old Breonna Taylor was shot after police forcibly entered her home. Armaud Arbery was shot and killed by armed neighbors while running through a neighborhood in Brunswick, Ga. Then on May 25, 5 miles from where I call home here in the Twin Cities in Minnesota, George Floyd, a 46-year-old father arrested for suspected use of a counterfeit $20 bill, died after police kneeled on his neck for over 8 minutes. This pandemic has also shaken up the status quo and laid bare a lot of our country’s long and deep-seated issues – from massive economic inequities to ongoing racial disparities to immigration concerns. It’s woken a lot of our valued hospitalists to the fact that the old ways of doing things just don’t work.
I’m grateful our society has taken steps to speak into these timely topics, and to share via publications, Twitter chats, advocacy items, and more! I want to encourage all of us to use the immense network of our hospitalist communities to comfort each other, learn, grow, and engage. We have not achieved big changes by ourselves. We’ve created valued offerings and innovative changes, and we’ve led on the front lines, in policies and procedures, by doing it together. Meaningful change requires allies in a common cause. We stand with our black and brown brothers and sisters who are particularly attuned to injustice, inequality, and struggle. We in hospital medicine stand up with many others who are struggling, our African American, Latin American, Native American, immigrant, LGBTQ+ communities. This intersection of the crisis of the COVID-19 pandemic and the racism pandemic have led us to a pivotal point in the arc of change and justice. I invite you to comfort each other, learn from each other, and act together in this community. To this end we have included timely resources in our HM20 virtual offering on these topics.
This year has been a big transition year. Not only did 2020 usher in a new decade, along with COVID-19 and our double pandemic, SHM has also had important transitions within its senior leadership. We say farewell to Larry Wellikson, MD, who has been at the helm of SHM since the beginning. On behalf of this annual conference, we want to celebrate and thank you, Larry, for your years of dedication and service to SHM. You have taken the specialty of hospital medicine and created a movement in SHM, where the entire hospital medicine team may gather under a bigger tent for education, community, and for the betterment of care for our patients.
We extend a welcome to Eric Howell, MD, who succeeds Dr. Wellikson as SHM’s CEO. We also welcome Danielle Scheurer, MD, as the new SHM president, succeeding the great leadership offered this past year by Christopher Frost, MD. In addition, Jerome C. Siy, MD, was voted president-elect, Dr. Rachel Thompson, MD, was elected treasurer, Kris Rehm, MD, was voted secretary, and Darlene Tad-y, MD, was elected to the board of directors. We welcome these new officers.
HM20 Virtual will consist of prerecorded on-demand sessions that can be viewed at your convenience as well as live Q&A and attendee networking that will take place during specific dates/times. A few of the top-rated sessions from our historically popular tracks include: Update in Clinical Practice Guidelines, Antibiotics Made Ridiculously Simple, Getting to Know Oncology Emergencies, Inpatient Pain Management in the Era of the Opioid Epidemic, Updates in Heart Failure, and Hyponatremia: Don’t Drink the Water. Additionally, we have some of our perennial favorites including the Update in Hospital Medicine and Top Pediatric Articles of 2019. There will be COVID-19 specific content from expertise throughout the nation focusing on care pathways, clinical updates, telemedicine, point-of-care ultrasound, and more! To view the HM20 Virtual Opening Session and discover what you can expect in this educational experience, click here.
The Journal of Hospital Medicine has had a large presence in our meetings for many years. We are grateful for Samir Shah, MD, and his leadership during this double pandemic, for identifying areas where we can advance the field responsibly in the face of relatively limited evidence, and rapidly evolving news. As part of his commitment, all JHM articles related to COVID-19 and published during the pandemic are open access. A pre-COVID goal that has been realized during the pandemic was to bring more of the journal into our annual conference and the conference contents into the journal. We are proud to say this has been a great collaboration, particularly during this pandemic, and much thanks to Dr. Shah’s leadership for highlighting timely pieces. Kimberly Manning, MD, had an especially powerful piece on the topic of racism and our double pandemic, and she is a featured speaker during our HM20 Virtual offering, under the same title as her article: “When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics.” Additionally, Manpreet Malik, MD, and I will be copresenting on a timely topic about the “Immigrant Hospitalist during COVID-19.”
Aside from these sessions for HM20 Virtual, the real can’t miss(es) for the conference are the Research, Innovations, and Clinical Vignette (RIV) posters sessions. I am grateful for the leadership of Stephanie Mueller, MD, who served as chair for this year’s RIV. This unique year has led to the hosting of a virtual poster competition with judging and the opening of a virtual gallery. We are so pleased to be able to share and highlight the work of many of learners and staff hospitalists! I love that a hospitalist on one side of the country can help provide pearls on a case, an innovation, or a research idea that can help improve diagnosis for a patient at the other side of the country. Keep an eye on SHM’s social media and the presentation by Dr. Mueller for announcements of the winners.
A favorite reason many of us attend the annual conference is for the people and community. We wanted to keep this value as we shifted to a virtual offering. Networking will occur through a variety of offerings including Simulive sessions and Special Interest Forums. Simulive sessions will run for 3 weeks from August 11 to August 27. For those of you new to the term, Simulive may sound like a made-up word, but it is an actual amalgamation of a prerecorded webinar and a live interaction (simulated + live = Simulive). Simulive allows the faculty to sit in on their prerecorded session and interact with the audience via the chat feature during the live scheduled recording and spend time afterwards for a live Q&A from the audience.
There will also be over 20 Special Interest Forums hosted in the evenings after these Simulive sessions have concluded to give you a chance to connect with individuals, share experiences, and have meaningful discussions that can directly impact your practice. Samples of the forums include: Diversity and Inclusion, Rural Hospital Medicine, Pediatrics, NP/PA, Perioperative and Comanagement, Health Information Technology, and Point of Care Ultrasound! Take a look at the HM20 registration page for further information. You will receive direct information on how to attend. We encourage you to join!
HM20 also features a virtual 5K! Whether you run on a treadmill or jog in your neighborhood or local park, you can participate in HM20’s Virtual Fun Run or Walk. To participate, simply run your 5K during the weeks of HM20 Virtual and when you’re done, fill out our form to log your time. We encourage you to post a picture on social media as well with #HM20Virtual. You’ll also receive a certificate of completion at the close of HM20 Virtual.
All HM20 Virtual sessions will be available as on-demand after August 31. HM20 virtual offers more than 60 CME hours and over 35 MOC hours that you can claim at your convenience! That’s the most amount of CME and MOC ever offered at SHM for an event! This conference would not be possible without the tireless and relentless effort of SHM staff and leadership, our terrific speakers and faculty, and all the volunteer committee members of SHM. A huge thanks to the Annual Conference Committee who had the charge to develop the content for the Annual Conference, including topics, speakers, and learning objectives. I am grateful to have had the opportunity to serve on this committee for the past 7 years and to lead HM20 this year. Thanks to Brittany Evans, Hayleigh Lawrence, and Michelle Kann for their valued support this past year from an SHM staff perspective; to my assistant course director, Dan Steinberg, MD; and to the immediate past course director, Dustin Smith, MD, for their support.
Once again, we are excited to have you join, and we hope this conference elevates your education in hospital medicine, advances your career, stimulates innovative thinking, and provides you with enduring networking opportunities. We sincerely thank you for attending HM20 Virtual. Welcome!
Dr. Mathews is chief of hospital medicine at Regions Hospital, HealthPartners in St. Paul, Minn., an associate professor at the University of Minnesota, Minneapolis, and course director of HM20.
Welcome to the HM20 virtual conference! We’re glad to have you join us to virtually experience sessions from our most popular SHM annual conference tracks including Rapid Fire, Clinical Updates, and High-Value Care! We also have added some new timely topics given our current times that you won’t want to miss. We encourage you to engage with the larger community via social media at #HM20Virtual.
HM20 in San Diego, scheduled originally for April 2020, was trending to be the highest in-person attended SHM annual conference with a fantastic line-up of offerings. Unfortunately, then came our pandemic, or pandemics. In mid-March, the Society of Hospital Medicine board of directors concluded that it was impossible for SHM to move forward with Hospital Medicine 2020 in San Diego because of the continued spread of COVID-19. Canceling the in-person conference during this unprecedented time was the right thing to do. I have valued the SHM leadership team and the larger SHM community for their support in being even more engaged on the front lines and with each other across our world during this time.
The COVID-19 pandemic has created a systemic challenge for health care systems across the nation. As hospitalists continue to be on the front lines of care and also innovations, organizations have leveraged telemedicine to support their patients, protect their clinicians, and conserve scarce resources. It is hospital medicine that has been on the front lines of change and adaptations and have led in this pandemic in many organizations across the nation and the world.
Unfortunately, known health disparities have also been amplified and there came an acute worsening of the chronic issues in this nation. On March 13, 2020, 26-year-old Breonna Taylor was shot after police forcibly entered her home. Armaud Arbery was shot and killed by armed neighbors while running through a neighborhood in Brunswick, Ga. Then on May 25, 5 miles from where I call home here in the Twin Cities in Minnesota, George Floyd, a 46-year-old father arrested for suspected use of a counterfeit $20 bill, died after police kneeled on his neck for over 8 minutes. This pandemic has also shaken up the status quo and laid bare a lot of our country’s long and deep-seated issues – from massive economic inequities to ongoing racial disparities to immigration concerns. It’s woken a lot of our valued hospitalists to the fact that the old ways of doing things just don’t work.
I’m grateful our society has taken steps to speak into these timely topics, and to share via publications, Twitter chats, advocacy items, and more! I want to encourage all of us to use the immense network of our hospitalist communities to comfort each other, learn, grow, and engage. We have not achieved big changes by ourselves. We’ve created valued offerings and innovative changes, and we’ve led on the front lines, in policies and procedures, by doing it together. Meaningful change requires allies in a common cause. We stand with our black and brown brothers and sisters who are particularly attuned to injustice, inequality, and struggle. We in hospital medicine stand up with many others who are struggling, our African American, Latin American, Native American, immigrant, LGBTQ+ communities. This intersection of the crisis of the COVID-19 pandemic and the racism pandemic have led us to a pivotal point in the arc of change and justice. I invite you to comfort each other, learn from each other, and act together in this community. To this end we have included timely resources in our HM20 virtual offering on these topics.
This year has been a big transition year. Not only did 2020 usher in a new decade, along with COVID-19 and our double pandemic, SHM has also had important transitions within its senior leadership. We say farewell to Larry Wellikson, MD, who has been at the helm of SHM since the beginning. On behalf of this annual conference, we want to celebrate and thank you, Larry, for your years of dedication and service to SHM. You have taken the specialty of hospital medicine and created a movement in SHM, where the entire hospital medicine team may gather under a bigger tent for education, community, and for the betterment of care for our patients.
We extend a welcome to Eric Howell, MD, who succeeds Dr. Wellikson as SHM’s CEO. We also welcome Danielle Scheurer, MD, as the new SHM president, succeeding the great leadership offered this past year by Christopher Frost, MD. In addition, Jerome C. Siy, MD, was voted president-elect, Dr. Rachel Thompson, MD, was elected treasurer, Kris Rehm, MD, was voted secretary, and Darlene Tad-y, MD, was elected to the board of directors. We welcome these new officers.
HM20 Virtual will consist of prerecorded on-demand sessions that can be viewed at your convenience as well as live Q&A and attendee networking that will take place during specific dates/times. A few of the top-rated sessions from our historically popular tracks include: Update in Clinical Practice Guidelines, Antibiotics Made Ridiculously Simple, Getting to Know Oncology Emergencies, Inpatient Pain Management in the Era of the Opioid Epidemic, Updates in Heart Failure, and Hyponatremia: Don’t Drink the Water. Additionally, we have some of our perennial favorites including the Update in Hospital Medicine and Top Pediatric Articles of 2019. There will be COVID-19 specific content from expertise throughout the nation focusing on care pathways, clinical updates, telemedicine, point-of-care ultrasound, and more! To view the HM20 Virtual Opening Session and discover what you can expect in this educational experience, click here.
The Journal of Hospital Medicine has had a large presence in our meetings for many years. We are grateful for Samir Shah, MD, and his leadership during this double pandemic, for identifying areas where we can advance the field responsibly in the face of relatively limited evidence, and rapidly evolving news. As part of his commitment, all JHM articles related to COVID-19 and published during the pandemic are open access. A pre-COVID goal that has been realized during the pandemic was to bring more of the journal into our annual conference and the conference contents into the journal. We are proud to say this has been a great collaboration, particularly during this pandemic, and much thanks to Dr. Shah’s leadership for highlighting timely pieces. Kimberly Manning, MD, had an especially powerful piece on the topic of racism and our double pandemic, and she is a featured speaker during our HM20 Virtual offering, under the same title as her article: “When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics.” Additionally, Manpreet Malik, MD, and I will be copresenting on a timely topic about the “Immigrant Hospitalist during COVID-19.”
Aside from these sessions for HM20 Virtual, the real can’t miss(es) for the conference are the Research, Innovations, and Clinical Vignette (RIV) posters sessions. I am grateful for the leadership of Stephanie Mueller, MD, who served as chair for this year’s RIV. This unique year has led to the hosting of a virtual poster competition with judging and the opening of a virtual gallery. We are so pleased to be able to share and highlight the work of many of learners and staff hospitalists! I love that a hospitalist on one side of the country can help provide pearls on a case, an innovation, or a research idea that can help improve diagnosis for a patient at the other side of the country. Keep an eye on SHM’s social media and the presentation by Dr. Mueller for announcements of the winners.
A favorite reason many of us attend the annual conference is for the people and community. We wanted to keep this value as we shifted to a virtual offering. Networking will occur through a variety of offerings including Simulive sessions and Special Interest Forums. Simulive sessions will run for 3 weeks from August 11 to August 27. For those of you new to the term, Simulive may sound like a made-up word, but it is an actual amalgamation of a prerecorded webinar and a live interaction (simulated + live = Simulive). Simulive allows the faculty to sit in on their prerecorded session and interact with the audience via the chat feature during the live scheduled recording and spend time afterwards for a live Q&A from the audience.
There will also be over 20 Special Interest Forums hosted in the evenings after these Simulive sessions have concluded to give you a chance to connect with individuals, share experiences, and have meaningful discussions that can directly impact your practice. Samples of the forums include: Diversity and Inclusion, Rural Hospital Medicine, Pediatrics, NP/PA, Perioperative and Comanagement, Health Information Technology, and Point of Care Ultrasound! Take a look at the HM20 registration page for further information. You will receive direct information on how to attend. We encourage you to join!
HM20 also features a virtual 5K! Whether you run on a treadmill or jog in your neighborhood or local park, you can participate in HM20’s Virtual Fun Run or Walk. To participate, simply run your 5K during the weeks of HM20 Virtual and when you’re done, fill out our form to log your time. We encourage you to post a picture on social media as well with #HM20Virtual. You’ll also receive a certificate of completion at the close of HM20 Virtual.
All HM20 Virtual sessions will be available as on-demand after August 31. HM20 virtual offers more than 60 CME hours and over 35 MOC hours that you can claim at your convenience! That’s the most amount of CME and MOC ever offered at SHM for an event! This conference would not be possible without the tireless and relentless effort of SHM staff and leadership, our terrific speakers and faculty, and all the volunteer committee members of SHM. A huge thanks to the Annual Conference Committee who had the charge to develop the content for the Annual Conference, including topics, speakers, and learning objectives. I am grateful to have had the opportunity to serve on this committee for the past 7 years and to lead HM20 this year. Thanks to Brittany Evans, Hayleigh Lawrence, and Michelle Kann for their valued support this past year from an SHM staff perspective; to my assistant course director, Dan Steinberg, MD; and to the immediate past course director, Dustin Smith, MD, for their support.
Once again, we are excited to have you join, and we hope this conference elevates your education in hospital medicine, advances your career, stimulates innovative thinking, and provides you with enduring networking opportunities. We sincerely thank you for attending HM20 Virtual. Welcome!
Dr. Mathews is chief of hospital medicine at Regions Hospital, HealthPartners in St. Paul, Minn., an associate professor at the University of Minnesota, Minneapolis, and course director of HM20.