User login
Official Newspaper of the American College of Surgeons
Medicare's version of surgical quality
The quality of surgical care should be on everyone’s minds. As we all know, the government’s current plan to halt the nonsustainable rate of health care expenditure increases includes the use of Accountable Care Organizations (ACOs).
The idea is to make care "accountable" by partnering providers and hospitals to provide more efficient care. Efficient care in this context translates to reductions in unnecessary expenditures that do not add value, whether they are imaging, physician office visits, specialist consultations, lab tests, or procedures while retaining quality.
The high degree of variability of some expenditures by region and the lack of positive correlation between expenditures on the one hand, and healthcare outcome, patient satisfaction, or quality measures on the other, suggests that reducing expenditures without harming patients is possible. To date, it remains unclear how surgical practice will be affected by the introduction of ACOs, although it is hard to imagine how it could be immune to these pressures. One thing that seems certain is that quality of care will be a centerpiece of the new strategy. Therefore, it seems inevitable that quality standards will eventually be applied to surgical care. Your wound infection rate is statistically above average? Either fix it or stop operating.
Does it sound far-fetched that a regulatory body could determine the quality of a surgeon’s work based purely on measurement of outcome, and further, that this body could determine who can practice and who can’t? Consider the case of organ transplantation. Medicare began certifying heart, liver, and lung transplant programs in 1986, 1991, and 1995, respectively. In order to meet Medicare’s standard and to be Medicare approved, programs had to meet a raw survival threshold as well as a volume standard. The survival standard for heart transplantation in 1986 was 73% 1-year patient surviva
For liver transplantation, the initial standard was 77% 1-year survival; and for lung transplantation; the standard was 79%. Over the next decades survival rates gradually rose as better immunosuppression appeared and other advances in the field were made. As a consequence, the Medicare standards were eventually out of date since they no longer represented state of the art care. Surprisingly, Medicare never actually enforced the quality, nor the volume standards until a string of high-profile scandals involving several transplant programs prompted the agency to release a new set of "Conditions of Participation" in 2007. This document was published in the Federal Register meaning the rules essentially have the force of federal law. Among these rules was the establishment of minimum outcome standards for patient and graft survival. Want to do organ transplants? You must work in a program in which outcomes are within two standard deviations of the risk-adjusted national norm.
The approximately 250 hospitals in the United States at which organ transplants are performed have been functioning under the Conditions of Participation for more than 5 years now. Several things are becoming very clear. First, while Medicare approval, meaning compliance with Medicare standards, is necessary only in order to transplant Medicare beneficiaries, it is impossible for a program to be financially solvent if it is not Medicare approved. In addition to having to turn away Medicare patients, the other players in the field of organ transplantation are large transplant networks of secondary insurers, all of which require Medicare approval as a prerequisite to inclusion in their network. Medicare approval is therefore understood with in the transplant community to be mandatory. Second, the agency is serious about enforcing these regulations and holding transplant programs accountable for outcomes. Programs that do not meet their standards receive a written notice that their Medicare approval has been terminated. Appeal, through a process called mitigating factors, is allowed, as is a structured attempt to correct deficiencies called a Service Level Agreement. The sense in the transplantation community is that Medicare is pleased by the effect the new standards have had. The standards are viewed as a model for how regulation could be applied to future areas of medicine. Surgical care, since it tends to have easily tracked binary outcomes, will likely be affected in the future by Medicare experience with standard setting to date. This last bit is why surgeons outside of Transplantation need to pay attention.
Only time will tell if the outcome requirements for transplantation have had an overall positive impact. One benefit is that programs are clearly doing a better job now of tracking and paying attention to their outcomes. Quality-improvement programs have absolutely been made more robust. On the other hand, concerns have been raised that such standards will stifle innovation. Medicare has said they are willing to listen and discuss how this problem can be alleviated. For now, there is a profound disincentive to perform transplants on patients whose chance of survival is poorly measured by current algorithms or is unknown. At one point not that long ago, hepatocellular carcinoma was considered to be a contraindication to liver transplantation. Thanks to groundbreaking work at a few centers that refused to deny transplant patients purely because outcome has historically bad, hepatocellular cancer is one of the most common indications for liver transplantation today. Would these advances have been made if programs were watching outcomes like a hawk and basing decisions only on likely outcomes? Concerns have also been raised that these rules will limit access to transplantation through a variety of mechanisms. When programs are terminated by Medicare, the patients on their waiting list must be transferred to other programs. This process takes time and patients’ lives will hang in the balance. Finally, the issue of whether the standards really measure quality of care is now being thought about in much greater depth. The risk-adjustment methodologies were designed with quality improvement in mind, not to be a test for programmatic effectiveness. These issues will be included in a future editorial on the evaluation of surgical quality in the context of organ transplantation. Stay tuned.
Dr. Punch is an ACS Fellow and Jeremiah and Clare Turcotte Professor of Surgery, University of Michigan, Ann Arbor.
The quality of surgical care should be on everyone’s minds. As we all know, the government’s current plan to halt the nonsustainable rate of health care expenditure increases includes the use of Accountable Care Organizations (ACOs).
The idea is to make care "accountable" by partnering providers and hospitals to provide more efficient care. Efficient care in this context translates to reductions in unnecessary expenditures that do not add value, whether they are imaging, physician office visits, specialist consultations, lab tests, or procedures while retaining quality.
The high degree of variability of some expenditures by region and the lack of positive correlation between expenditures on the one hand, and healthcare outcome, patient satisfaction, or quality measures on the other, suggests that reducing expenditures without harming patients is possible. To date, it remains unclear how surgical practice will be affected by the introduction of ACOs, although it is hard to imagine how it could be immune to these pressures. One thing that seems certain is that quality of care will be a centerpiece of the new strategy. Therefore, it seems inevitable that quality standards will eventually be applied to surgical care. Your wound infection rate is statistically above average? Either fix it or stop operating.
Does it sound far-fetched that a regulatory body could determine the quality of a surgeon’s work based purely on measurement of outcome, and further, that this body could determine who can practice and who can’t? Consider the case of organ transplantation. Medicare began certifying heart, liver, and lung transplant programs in 1986, 1991, and 1995, respectively. In order to meet Medicare’s standard and to be Medicare approved, programs had to meet a raw survival threshold as well as a volume standard. The survival standard for heart transplantation in 1986 was 73% 1-year patient surviva
For liver transplantation, the initial standard was 77% 1-year survival; and for lung transplantation; the standard was 79%. Over the next decades survival rates gradually rose as better immunosuppression appeared and other advances in the field were made. As a consequence, the Medicare standards were eventually out of date since they no longer represented state of the art care. Surprisingly, Medicare never actually enforced the quality, nor the volume standards until a string of high-profile scandals involving several transplant programs prompted the agency to release a new set of "Conditions of Participation" in 2007. This document was published in the Federal Register meaning the rules essentially have the force of federal law. Among these rules was the establishment of minimum outcome standards for patient and graft survival. Want to do organ transplants? You must work in a program in which outcomes are within two standard deviations of the risk-adjusted national norm.
The approximately 250 hospitals in the United States at which organ transplants are performed have been functioning under the Conditions of Participation for more than 5 years now. Several things are becoming very clear. First, while Medicare approval, meaning compliance with Medicare standards, is necessary only in order to transplant Medicare beneficiaries, it is impossible for a program to be financially solvent if it is not Medicare approved. In addition to having to turn away Medicare patients, the other players in the field of organ transplantation are large transplant networks of secondary insurers, all of which require Medicare approval as a prerequisite to inclusion in their network. Medicare approval is therefore understood with in the transplant community to be mandatory. Second, the agency is serious about enforcing these regulations and holding transplant programs accountable for outcomes. Programs that do not meet their standards receive a written notice that their Medicare approval has been terminated. Appeal, through a process called mitigating factors, is allowed, as is a structured attempt to correct deficiencies called a Service Level Agreement. The sense in the transplantation community is that Medicare is pleased by the effect the new standards have had. The standards are viewed as a model for how regulation could be applied to future areas of medicine. Surgical care, since it tends to have easily tracked binary outcomes, will likely be affected in the future by Medicare experience with standard setting to date. This last bit is why surgeons outside of Transplantation need to pay attention.
Only time will tell if the outcome requirements for transplantation have had an overall positive impact. One benefit is that programs are clearly doing a better job now of tracking and paying attention to their outcomes. Quality-improvement programs have absolutely been made more robust. On the other hand, concerns have been raised that such standards will stifle innovation. Medicare has said they are willing to listen and discuss how this problem can be alleviated. For now, there is a profound disincentive to perform transplants on patients whose chance of survival is poorly measured by current algorithms or is unknown. At one point not that long ago, hepatocellular carcinoma was considered to be a contraindication to liver transplantation. Thanks to groundbreaking work at a few centers that refused to deny transplant patients purely because outcome has historically bad, hepatocellular cancer is one of the most common indications for liver transplantation today. Would these advances have been made if programs were watching outcomes like a hawk and basing decisions only on likely outcomes? Concerns have also been raised that these rules will limit access to transplantation through a variety of mechanisms. When programs are terminated by Medicare, the patients on their waiting list must be transferred to other programs. This process takes time and patients’ lives will hang in the balance. Finally, the issue of whether the standards really measure quality of care is now being thought about in much greater depth. The risk-adjustment methodologies were designed with quality improvement in mind, not to be a test for programmatic effectiveness. These issues will be included in a future editorial on the evaluation of surgical quality in the context of organ transplantation. Stay tuned.
Dr. Punch is an ACS Fellow and Jeremiah and Clare Turcotte Professor of Surgery, University of Michigan, Ann Arbor.
The quality of surgical care should be on everyone’s minds. As we all know, the government’s current plan to halt the nonsustainable rate of health care expenditure increases includes the use of Accountable Care Organizations (ACOs).
The idea is to make care "accountable" by partnering providers and hospitals to provide more efficient care. Efficient care in this context translates to reductions in unnecessary expenditures that do not add value, whether they are imaging, physician office visits, specialist consultations, lab tests, or procedures while retaining quality.
The high degree of variability of some expenditures by region and the lack of positive correlation between expenditures on the one hand, and healthcare outcome, patient satisfaction, or quality measures on the other, suggests that reducing expenditures without harming patients is possible. To date, it remains unclear how surgical practice will be affected by the introduction of ACOs, although it is hard to imagine how it could be immune to these pressures. One thing that seems certain is that quality of care will be a centerpiece of the new strategy. Therefore, it seems inevitable that quality standards will eventually be applied to surgical care. Your wound infection rate is statistically above average? Either fix it or stop operating.
Does it sound far-fetched that a regulatory body could determine the quality of a surgeon’s work based purely on measurement of outcome, and further, that this body could determine who can practice and who can’t? Consider the case of organ transplantation. Medicare began certifying heart, liver, and lung transplant programs in 1986, 1991, and 1995, respectively. In order to meet Medicare’s standard and to be Medicare approved, programs had to meet a raw survival threshold as well as a volume standard. The survival standard for heart transplantation in 1986 was 73% 1-year patient surviva
For liver transplantation, the initial standard was 77% 1-year survival; and for lung transplantation; the standard was 79%. Over the next decades survival rates gradually rose as better immunosuppression appeared and other advances in the field were made. As a consequence, the Medicare standards were eventually out of date since they no longer represented state of the art care. Surprisingly, Medicare never actually enforced the quality, nor the volume standards until a string of high-profile scandals involving several transplant programs prompted the agency to release a new set of "Conditions of Participation" in 2007. This document was published in the Federal Register meaning the rules essentially have the force of federal law. Among these rules was the establishment of minimum outcome standards for patient and graft survival. Want to do organ transplants? You must work in a program in which outcomes are within two standard deviations of the risk-adjusted national norm.
The approximately 250 hospitals in the United States at which organ transplants are performed have been functioning under the Conditions of Participation for more than 5 years now. Several things are becoming very clear. First, while Medicare approval, meaning compliance with Medicare standards, is necessary only in order to transplant Medicare beneficiaries, it is impossible for a program to be financially solvent if it is not Medicare approved. In addition to having to turn away Medicare patients, the other players in the field of organ transplantation are large transplant networks of secondary insurers, all of which require Medicare approval as a prerequisite to inclusion in their network. Medicare approval is therefore understood with in the transplant community to be mandatory. Second, the agency is serious about enforcing these regulations and holding transplant programs accountable for outcomes. Programs that do not meet their standards receive a written notice that their Medicare approval has been terminated. Appeal, through a process called mitigating factors, is allowed, as is a structured attempt to correct deficiencies called a Service Level Agreement. The sense in the transplantation community is that Medicare is pleased by the effect the new standards have had. The standards are viewed as a model for how regulation could be applied to future areas of medicine. Surgical care, since it tends to have easily tracked binary outcomes, will likely be affected in the future by Medicare experience with standard setting to date. This last bit is why surgeons outside of Transplantation need to pay attention.
Only time will tell if the outcome requirements for transplantation have had an overall positive impact. One benefit is that programs are clearly doing a better job now of tracking and paying attention to their outcomes. Quality-improvement programs have absolutely been made more robust. On the other hand, concerns have been raised that such standards will stifle innovation. Medicare has said they are willing to listen and discuss how this problem can be alleviated. For now, there is a profound disincentive to perform transplants on patients whose chance of survival is poorly measured by current algorithms or is unknown. At one point not that long ago, hepatocellular carcinoma was considered to be a contraindication to liver transplantation. Thanks to groundbreaking work at a few centers that refused to deny transplant patients purely because outcome has historically bad, hepatocellular cancer is one of the most common indications for liver transplantation today. Would these advances have been made if programs were watching outcomes like a hawk and basing decisions only on likely outcomes? Concerns have also been raised that these rules will limit access to transplantation through a variety of mechanisms. When programs are terminated by Medicare, the patients on their waiting list must be transferred to other programs. This process takes time and patients’ lives will hang in the balance. Finally, the issue of whether the standards really measure quality of care is now being thought about in much greater depth. The risk-adjustment methodologies were designed with quality improvement in mind, not to be a test for programmatic effectiveness. These issues will be included in a future editorial on the evaluation of surgical quality in the context of organ transplantation. Stay tuned.
Dr. Punch is an ACS Fellow and Jeremiah and Clare Turcotte Professor of Surgery, University of Michigan, Ann Arbor.
'Don't tell her the diagnosis': Nondisclosure and the surgeon
It goes without saying that good surgical care is based on honesty in informed consent. The ethical basis of telling patients about their conditions and what needs to be done is central to what surgeons do. In this context, a request not to tell a patient a diagnosis is always jarring to me. One of the ethical principles that medicine has most fully embraced in the last few decades has been respect for patient autonomy. This principle is very much in opposition with the previous practice of paternalism in the prior era of medical care in which "the doctor knows best" and doctors made decisions for their patients. As a practicing surgeon today, I feel that there is very little that I know that I cannot disclose to my patient. However, occasionally cases challenge our underlying assumptions.
A few years ago, I saw an 11-year-old girl with a recent diagnosis of papillary thyroid cancer. Before I even saw her, the parents had called my office to be sure that I did not tell her the diagnosis of cancer. I found this request to be troubling. How could I discuss the operation with this child without telling her that she had cancer? Her parents assured me that she knew that she had a thyroid nodule and that on the basis of the biopsy, that she would need a thyroidectomy. The only thing that had not been explained to the child was the diagnosis of thyroid cancer.
Despite my initial concern with this request, in pediatrics, the parents are the decision makers for the child, so that there was no legal reason why the patient needed to be told that she had cancer. Nevertheless, the ethical imperative to include the diagnosis of cancer in the discussion about surgery weighed on me. Despite my initial opposition to being put in the position of not telling the patient of her diagnosis, I decided that I could do nothing more at that point. I hoped to convince the parents to let me share the diagnosis with their daughter at a later time.
When I met my patient, I found her to be a quiet and calm girl who seemed to me to be mature beyond her years. I proceeded to explain the risks of thyroidectomy to the patient and her parents. She seemed to take it all in and asked good questions about the operation and the recovery. She wanted to know how long before she could get back to school and sports. At the end of the consultation, the patient’s mother asked her to wait with her younger sister and her grandmother in the waiting room for a few minutes while the parents spoke to me alone.
Once she had left, the parents expressed their appreciation that I had not told her she had cancer. I told them how impressed I was with her poise and maturity and that although I did not agree with their decision not to tell her the diagnosis, I would certainly go along with it based on the assumption that they knew what would be in her best interests better than I. They seemed relieved that I was willing to go along with their decision. I realized at that point that the ethical arguments in favor of telling the patient of her diagnosis would likely be unconvincing for the parents, so I decided to focus instead on the practical problems with nondisclosure.
I asked the parents to consider that the operative schedule would include the diagnosis of thyroid cancer and that everyone seeing her in the hospital (doctors, nurses, etc.) would know her diagnosis. For all of these reasons, there would be a high likelihood that at some point during her hospital stay, someone would slip, and she would learn of the diagnosis in an uncontrolled manner from someone other than her parents or her doctor. In addition, I suggested that she would likely figure it out anyway even if no one told her. Finally, I asked them to consider the next few years. If they did not tell her the diagnosis of cancer now, at what point would they choose to do so? Certainly, at the point that she turned 18 years old, she would need to know the diagnosis, but would the parents want to hide it from her that long, even if they could?
The parents seemed to have not thought of all of these issues and answered that they fully wanted to tell her, but they were concerned about doing so when they, themselves, were still so upset by the diagnosis. They explained that they planned to tell her when they felt more in control of their own emotions.
Two weeks later, on the morning of surgery, the parents told me how they had explained the diagnosis to their daughter and that she had then explained it to her younger sister. It was clear to me that the assurance that the parents had given to the patient had allowed her to be calm and positive when talking with her younger sister. It is unknown how things might have worked out had the parents not told the patient of her diagnosis when they did, but it was clear to me that the fact that the parents had been able to control some aspects of how the patient learned of her diagnosis had helped them to feel better about a difficult situation. In addition, the patient seemed to be reassured by having explained things to her sister. Although I continue to assume that disclosure is always the best approach, there may be cases, such as this one, in which the timing of the disclosure might allow for a good outcome.
Dr. Angelos is an ACS Fellow, the Linda Kohler Anderson Professor of Surgery and Surgical Ethics; chief, endocrine surgery, and associate director of the MacLean Center for Clinical Medical Ethics at the University of Chicago.
It goes without saying that good surgical care is based on honesty in informed consent. The ethical basis of telling patients about their conditions and what needs to be done is central to what surgeons do. In this context, a request not to tell a patient a diagnosis is always jarring to me. One of the ethical principles that medicine has most fully embraced in the last few decades has been respect for patient autonomy. This principle is very much in opposition with the previous practice of paternalism in the prior era of medical care in which "the doctor knows best" and doctors made decisions for their patients. As a practicing surgeon today, I feel that there is very little that I know that I cannot disclose to my patient. However, occasionally cases challenge our underlying assumptions.
A few years ago, I saw an 11-year-old girl with a recent diagnosis of papillary thyroid cancer. Before I even saw her, the parents had called my office to be sure that I did not tell her the diagnosis of cancer. I found this request to be troubling. How could I discuss the operation with this child without telling her that she had cancer? Her parents assured me that she knew that she had a thyroid nodule and that on the basis of the biopsy, that she would need a thyroidectomy. The only thing that had not been explained to the child was the diagnosis of thyroid cancer.
Despite my initial concern with this request, in pediatrics, the parents are the decision makers for the child, so that there was no legal reason why the patient needed to be told that she had cancer. Nevertheless, the ethical imperative to include the diagnosis of cancer in the discussion about surgery weighed on me. Despite my initial opposition to being put in the position of not telling the patient of her diagnosis, I decided that I could do nothing more at that point. I hoped to convince the parents to let me share the diagnosis with their daughter at a later time.
When I met my patient, I found her to be a quiet and calm girl who seemed to me to be mature beyond her years. I proceeded to explain the risks of thyroidectomy to the patient and her parents. She seemed to take it all in and asked good questions about the operation and the recovery. She wanted to know how long before she could get back to school and sports. At the end of the consultation, the patient’s mother asked her to wait with her younger sister and her grandmother in the waiting room for a few minutes while the parents spoke to me alone.
Once she had left, the parents expressed their appreciation that I had not told her she had cancer. I told them how impressed I was with her poise and maturity and that although I did not agree with their decision not to tell her the diagnosis, I would certainly go along with it based on the assumption that they knew what would be in her best interests better than I. They seemed relieved that I was willing to go along with their decision. I realized at that point that the ethical arguments in favor of telling the patient of her diagnosis would likely be unconvincing for the parents, so I decided to focus instead on the practical problems with nondisclosure.
I asked the parents to consider that the operative schedule would include the diagnosis of thyroid cancer and that everyone seeing her in the hospital (doctors, nurses, etc.) would know her diagnosis. For all of these reasons, there would be a high likelihood that at some point during her hospital stay, someone would slip, and she would learn of the diagnosis in an uncontrolled manner from someone other than her parents or her doctor. In addition, I suggested that she would likely figure it out anyway even if no one told her. Finally, I asked them to consider the next few years. If they did not tell her the diagnosis of cancer now, at what point would they choose to do so? Certainly, at the point that she turned 18 years old, she would need to know the diagnosis, but would the parents want to hide it from her that long, even if they could?
The parents seemed to have not thought of all of these issues and answered that they fully wanted to tell her, but they were concerned about doing so when they, themselves, were still so upset by the diagnosis. They explained that they planned to tell her when they felt more in control of their own emotions.
Two weeks later, on the morning of surgery, the parents told me how they had explained the diagnosis to their daughter and that she had then explained it to her younger sister. It was clear to me that the assurance that the parents had given to the patient had allowed her to be calm and positive when talking with her younger sister. It is unknown how things might have worked out had the parents not told the patient of her diagnosis when they did, but it was clear to me that the fact that the parents had been able to control some aspects of how the patient learned of her diagnosis had helped them to feel better about a difficult situation. In addition, the patient seemed to be reassured by having explained things to her sister. Although I continue to assume that disclosure is always the best approach, there may be cases, such as this one, in which the timing of the disclosure might allow for a good outcome.
Dr. Angelos is an ACS Fellow, the Linda Kohler Anderson Professor of Surgery and Surgical Ethics; chief, endocrine surgery, and associate director of the MacLean Center for Clinical Medical Ethics at the University of Chicago.
It goes without saying that good surgical care is based on honesty in informed consent. The ethical basis of telling patients about their conditions and what needs to be done is central to what surgeons do. In this context, a request not to tell a patient a diagnosis is always jarring to me. One of the ethical principles that medicine has most fully embraced in the last few decades has been respect for patient autonomy. This principle is very much in opposition with the previous practice of paternalism in the prior era of medical care in which "the doctor knows best" and doctors made decisions for their patients. As a practicing surgeon today, I feel that there is very little that I know that I cannot disclose to my patient. However, occasionally cases challenge our underlying assumptions.
A few years ago, I saw an 11-year-old girl with a recent diagnosis of papillary thyroid cancer. Before I even saw her, the parents had called my office to be sure that I did not tell her the diagnosis of cancer. I found this request to be troubling. How could I discuss the operation with this child without telling her that she had cancer? Her parents assured me that she knew that she had a thyroid nodule and that on the basis of the biopsy, that she would need a thyroidectomy. The only thing that had not been explained to the child was the diagnosis of thyroid cancer.
Despite my initial concern with this request, in pediatrics, the parents are the decision makers for the child, so that there was no legal reason why the patient needed to be told that she had cancer. Nevertheless, the ethical imperative to include the diagnosis of cancer in the discussion about surgery weighed on me. Despite my initial opposition to being put in the position of not telling the patient of her diagnosis, I decided that I could do nothing more at that point. I hoped to convince the parents to let me share the diagnosis with their daughter at a later time.
When I met my patient, I found her to be a quiet and calm girl who seemed to me to be mature beyond her years. I proceeded to explain the risks of thyroidectomy to the patient and her parents. She seemed to take it all in and asked good questions about the operation and the recovery. She wanted to know how long before she could get back to school and sports. At the end of the consultation, the patient’s mother asked her to wait with her younger sister and her grandmother in the waiting room for a few minutes while the parents spoke to me alone.
Once she had left, the parents expressed their appreciation that I had not told her she had cancer. I told them how impressed I was with her poise and maturity and that although I did not agree with their decision not to tell her the diagnosis, I would certainly go along with it based on the assumption that they knew what would be in her best interests better than I. They seemed relieved that I was willing to go along with their decision. I realized at that point that the ethical arguments in favor of telling the patient of her diagnosis would likely be unconvincing for the parents, so I decided to focus instead on the practical problems with nondisclosure.
I asked the parents to consider that the operative schedule would include the diagnosis of thyroid cancer and that everyone seeing her in the hospital (doctors, nurses, etc.) would know her diagnosis. For all of these reasons, there would be a high likelihood that at some point during her hospital stay, someone would slip, and she would learn of the diagnosis in an uncontrolled manner from someone other than her parents or her doctor. In addition, I suggested that she would likely figure it out anyway even if no one told her. Finally, I asked them to consider the next few years. If they did not tell her the diagnosis of cancer now, at what point would they choose to do so? Certainly, at the point that she turned 18 years old, she would need to know the diagnosis, but would the parents want to hide it from her that long, even if they could?
The parents seemed to have not thought of all of these issues and answered that they fully wanted to tell her, but they were concerned about doing so when they, themselves, were still so upset by the diagnosis. They explained that they planned to tell her when they felt more in control of their own emotions.
Two weeks later, on the morning of surgery, the parents told me how they had explained the diagnosis to their daughter and that she had then explained it to her younger sister. It was clear to me that the assurance that the parents had given to the patient had allowed her to be calm and positive when talking with her younger sister. It is unknown how things might have worked out had the parents not told the patient of her diagnosis when they did, but it was clear to me that the fact that the parents had been able to control some aspects of how the patient learned of her diagnosis had helped them to feel better about a difficult situation. In addition, the patient seemed to be reassured by having explained things to her sister. Although I continue to assume that disclosure is always the best approach, there may be cases, such as this one, in which the timing of the disclosure might allow for a good outcome.
Dr. Angelos is an ACS Fellow, the Linda Kohler Anderson Professor of Surgery and Surgical Ethics; chief, endocrine surgery, and associate director of the MacLean Center for Clinical Medical Ethics at the University of Chicago.
For empiric candidemia treatment, echinocandin tops fluconazole
LAS VEGAS – An echinocandin should be used for empiric therapy in critically ill candidemia patients awaiting culture results, according to investigators from Wayne State University in Detroit.
The reason is that Candida glabrata is on the rise in the critically ill, and it’s often resistant to fluconazole, the usual empiric choice, said Dr. Lisa Flynn, a vascular surgeon in the department of surgery at the university.
Dr. Flynn and her colleagues came to their conclusion after reviewing outcomes in 91 critically ill candidemia patients.
Just 40% (36) had the historic cause of candidemia, Candida albicans, which remains generally susceptible to fluconazole; 25% (23) had C. glabrata, and the rest had C. parapsilosis or other species.
Before those results were known, 53% (48) of patients were treated empirically with fluconazole and 36% (33), with the echinocandin micafungin. Most of the others received no treatment.
Seventy percent (16) of C. glabrata patients got fluconazole, the highest rate in the study of inappropriate initial antifungal therapy; probably not coincidently, 56% (13) of the C. glabrata patients died; the mortality rate in patients with other candida species was 32% (22). On univariate analysis, mortality increased from 18% to 37% if C. glabrata was cultured (P = .04).
"When we looked at glabrata versus all other candida species, we found significant increases in in-hospital mortality" that corresponded to a greater likelihood of inappropriate initial treatment, she said at the annual meeting of the Surgical Infection Society.
For that reason, "we are proposing that initial empiric antifungal therapy start with an echinocandin in the critically ill patient and then deescalate to fluconazole if [indicated by] culture data," she said.
It’s sound advice, so long as "your incidence of Candida glabrata is high," session moderator Dr. Addison May said after the presentation.
"It really depends on your hospital’s rate, and how frequently it’s [isolated]. It’s important to understand what you need to empirically treat with," but also important to use newer agents like micafungin judiciously, to prevent resistance, said Dr. May, professor of surgery and anesthesiology at Vanderbilt University in Nashville, Tenn.
C. glabrata patients were more likely than others to be over 60 years old; they had longer hospital and ICU stays, as well.
The mean APACHE II (Acute Physiology and Chronic Health Evaluation II) score in the study was 25, and the mean age was 57 years; 54% (49) of patients were men, and 68% (62) were black. In the previous month, almost half had surgery and a quarter had been on total parenteral nutrition.
Central lines were the source of infection in 84% (76).
On multivariate analysis, inappropriate initial antifungal treatment, vasopressor therapy, mechanical ventilation, and end-stage renal disease were all significant risk factors for death.
Dr. May and Dr. Flynn said they had no relevant financial disclosures.
LAS VEGAS – An echinocandin should be used for empiric therapy in critically ill candidemia patients awaiting culture results, according to investigators from Wayne State University in Detroit.
The reason is that Candida glabrata is on the rise in the critically ill, and it’s often resistant to fluconazole, the usual empiric choice, said Dr. Lisa Flynn, a vascular surgeon in the department of surgery at the university.
Dr. Flynn and her colleagues came to their conclusion after reviewing outcomes in 91 critically ill candidemia patients.
Just 40% (36) had the historic cause of candidemia, Candida albicans, which remains generally susceptible to fluconazole; 25% (23) had C. glabrata, and the rest had C. parapsilosis or other species.
Before those results were known, 53% (48) of patients were treated empirically with fluconazole and 36% (33), with the echinocandin micafungin. Most of the others received no treatment.
Seventy percent (16) of C. glabrata patients got fluconazole, the highest rate in the study of inappropriate initial antifungal therapy; probably not coincidently, 56% (13) of the C. glabrata patients died; the mortality rate in patients with other candida species was 32% (22). On univariate analysis, mortality increased from 18% to 37% if C. glabrata was cultured (P = .04).
"When we looked at glabrata versus all other candida species, we found significant increases in in-hospital mortality" that corresponded to a greater likelihood of inappropriate initial treatment, she said at the annual meeting of the Surgical Infection Society.
For that reason, "we are proposing that initial empiric antifungal therapy start with an echinocandin in the critically ill patient and then deescalate to fluconazole if [indicated by] culture data," she said.
It’s sound advice, so long as "your incidence of Candida glabrata is high," session moderator Dr. Addison May said after the presentation.
"It really depends on your hospital’s rate, and how frequently it’s [isolated]. It’s important to understand what you need to empirically treat with," but also important to use newer agents like micafungin judiciously, to prevent resistance, said Dr. May, professor of surgery and anesthesiology at Vanderbilt University in Nashville, Tenn.
C. glabrata patients were more likely than others to be over 60 years old; they had longer hospital and ICU stays, as well.
The mean APACHE II (Acute Physiology and Chronic Health Evaluation II) score in the study was 25, and the mean age was 57 years; 54% (49) of patients were men, and 68% (62) were black. In the previous month, almost half had surgery and a quarter had been on total parenteral nutrition.
Central lines were the source of infection in 84% (76).
On multivariate analysis, inappropriate initial antifungal treatment, vasopressor therapy, mechanical ventilation, and end-stage renal disease were all significant risk factors for death.
Dr. May and Dr. Flynn said they had no relevant financial disclosures.
LAS VEGAS – An echinocandin should be used for empiric therapy in critically ill candidemia patients awaiting culture results, according to investigators from Wayne State University in Detroit.
The reason is that Candida glabrata is on the rise in the critically ill, and it’s often resistant to fluconazole, the usual empiric choice, said Dr. Lisa Flynn, a vascular surgeon in the department of surgery at the university.
Dr. Flynn and her colleagues came to their conclusion after reviewing outcomes in 91 critically ill candidemia patients.
Just 40% (36) had the historic cause of candidemia, Candida albicans, which remains generally susceptible to fluconazole; 25% (23) had C. glabrata, and the rest had C. parapsilosis or other species.
Before those results were known, 53% (48) of patients were treated empirically with fluconazole and 36% (33), with the echinocandin micafungin. Most of the others received no treatment.
Seventy percent (16) of C. glabrata patients got fluconazole, the highest rate in the study of inappropriate initial antifungal therapy; probably not coincidently, 56% (13) of the C. glabrata patients died; the mortality rate in patients with other candida species was 32% (22). On univariate analysis, mortality increased from 18% to 37% if C. glabrata was cultured (P = .04).
"When we looked at glabrata versus all other candida species, we found significant increases in in-hospital mortality" that corresponded to a greater likelihood of inappropriate initial treatment, she said at the annual meeting of the Surgical Infection Society.
For that reason, "we are proposing that initial empiric antifungal therapy start with an echinocandin in the critically ill patient and then deescalate to fluconazole if [indicated by] culture data," she said.
It’s sound advice, so long as "your incidence of Candida glabrata is high," session moderator Dr. Addison May said after the presentation.
"It really depends on your hospital’s rate, and how frequently it’s [isolated]. It’s important to understand what you need to empirically treat with," but also important to use newer agents like micafungin judiciously, to prevent resistance, said Dr. May, professor of surgery and anesthesiology at Vanderbilt University in Nashville, Tenn.
C. glabrata patients were more likely than others to be over 60 years old; they had longer hospital and ICU stays, as well.
The mean APACHE II (Acute Physiology and Chronic Health Evaluation II) score in the study was 25, and the mean age was 57 years; 54% (49) of patients were men, and 68% (62) were black. In the previous month, almost half had surgery and a quarter had been on total parenteral nutrition.
Central lines were the source of infection in 84% (76).
On multivariate analysis, inappropriate initial antifungal treatment, vasopressor therapy, mechanical ventilation, and end-stage renal disease were all significant risk factors for death.
Dr. May and Dr. Flynn said they had no relevant financial disclosures.
AT THE SIS ANNUAL MEETING
Major finding: Of candidemia patients, 25% had C. glabrata, which is resistant to fluconazole and is associated with in-hospital mortality.
Data Source: A retrospective study of 91 candidemia patients
Disclosures: Dr. May and Dr. Flynn said they had no relevant financial disclosures.
Reablate, don't medicate, after failed AF ablation
DENVER – After a failed first ablation procedure for paroxysmal atrial fibrillation, redo ablation proved more effective than did antiarrhythmic drug therapy in a randomized trial, Dr. Jonathan Steinberg reported at the annual meeting of the Heart Rhythm Society.
The success rate of a first ablation procedure in patients with symptomatic paroxysmal AF is typically about 60%. What to do for the 40% who are nonresponders has been unclear, with no prior randomized clinical trial evidence available to guide decisions, noted Dr. Steinberg of Columbia University, New York.
The study comprised 154 patients with recurrent symptomatic paroxysmal AF 3 months after an initial ablation procedure involving only pulmonary vein isolation. All participants received an implantable loop recorder to track atrial arrhythmic events.
They were then randomized to redo-ablation limited to reisolation of the pulmonary vein, which was successfully accomplished in all instances, or to guideline-based antiarrhythmic drug therapy. The choice of drug was left to individual investigator discretion. The three options were propafenone at 450-900 mg/day, sotalol at 160-320 mg/day, or flecainide at 200-400 mg/day. Propafenone was selected in the majority of cases, at an average dose of 579 mg/day.
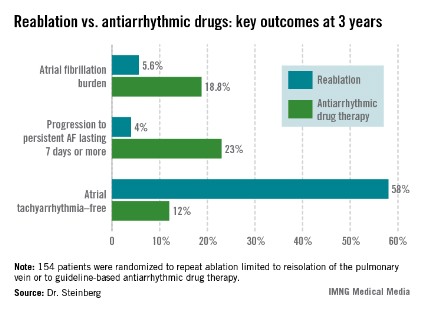
The average AF burden as measured by implantable loop recorder at randomization was 15%. The primary study endpoint was AF burden at 36 months of follow-up, which was 5.6% in the redo-ablation group compared with 18.8% in the antiarrhythmic drug group.
Secondary endpoints uniformly favored redo-ablation as well.
Data from the implantable loop recorders was evaluated every 3 months during 3 years of follow-up. As early as 3 months into the study, the group given antiarrhythmic drugs had an AF burden of 3.3%, significantly higher than the 1.9% rate seen in the redo-ablation group. Thereafter, the drug therapy group experienced a gradual increase in AF burden throughout the first 12-15 months, followed by a much more substantial increase during the remainder of the study.
"The redo-ablation group had a different pattern" of AF burden, Dr. Steinberg observed. "It was low throughout the first 12-15 months, with just a slight increase, and it then rose only gradually over time until the 36-month end of the study."
Freedom from any atrial tachyarrhythmia at 1 year was 30% in the antiarrhythmic drug therapy group and 75% in the redo-ablation group. By 3 years, 12% of those in the drug therapy group were free of atrial tachyarrhythmias as were 58% in the redo-ablation group.
Complications in the redo-ablation group consisted of two cases of cardiac tamponade. In contrast, 49 patients, or 64%, in the antiarrhythmic drug therapy group discontinued medication due to intolerance or ineffectiveness.
Session cochair Dr. Gordon Tomaselli of Johns Hopkins University, Baltimore, said that in light of the potential proarrhythmic effects of virtually all antiarrhythmic drugs, it would have been useful to include a no-antiarrhythmic drug control group in the study.
Dr. Steinberg reported having no conflicts of interest.
DENVER – After a failed first ablation procedure for paroxysmal atrial fibrillation, redo ablation proved more effective than did antiarrhythmic drug therapy in a randomized trial, Dr. Jonathan Steinberg reported at the annual meeting of the Heart Rhythm Society.
The success rate of a first ablation procedure in patients with symptomatic paroxysmal AF is typically about 60%. What to do for the 40% who are nonresponders has been unclear, with no prior randomized clinical trial evidence available to guide decisions, noted Dr. Steinberg of Columbia University, New York.
The study comprised 154 patients with recurrent symptomatic paroxysmal AF 3 months after an initial ablation procedure involving only pulmonary vein isolation. All participants received an implantable loop recorder to track atrial arrhythmic events.
They were then randomized to redo-ablation limited to reisolation of the pulmonary vein, which was successfully accomplished in all instances, or to guideline-based antiarrhythmic drug therapy. The choice of drug was left to individual investigator discretion. The three options were propafenone at 450-900 mg/day, sotalol at 160-320 mg/day, or flecainide at 200-400 mg/day. Propafenone was selected in the majority of cases, at an average dose of 579 mg/day.
The average AF burden as measured by implantable loop recorder at randomization was 15%. The primary study endpoint was AF burden at 36 months of follow-up, which was 5.6% in the redo-ablation group compared with 18.8% in the antiarrhythmic drug group.
Secondary endpoints uniformly favored redo-ablation as well.
Data from the implantable loop recorders was evaluated every 3 months during 3 years of follow-up. As early as 3 months into the study, the group given antiarrhythmic drugs had an AF burden of 3.3%, significantly higher than the 1.9% rate seen in the redo-ablation group. Thereafter, the drug therapy group experienced a gradual increase in AF burden throughout the first 12-15 months, followed by a much more substantial increase during the remainder of the study.
"The redo-ablation group had a different pattern" of AF burden, Dr. Steinberg observed. "It was low throughout the first 12-15 months, with just a slight increase, and it then rose only gradually over time until the 36-month end of the study."
Freedom from any atrial tachyarrhythmia at 1 year was 30% in the antiarrhythmic drug therapy group and 75% in the redo-ablation group. By 3 years, 12% of those in the drug therapy group were free of atrial tachyarrhythmias as were 58% in the redo-ablation group.
Complications in the redo-ablation group consisted of two cases of cardiac tamponade. In contrast, 49 patients, or 64%, in the antiarrhythmic drug therapy group discontinued medication due to intolerance or ineffectiveness.
Session cochair Dr. Gordon Tomaselli of Johns Hopkins University, Baltimore, said that in light of the potential proarrhythmic effects of virtually all antiarrhythmic drugs, it would have been useful to include a no-antiarrhythmic drug control group in the study.
Dr. Steinberg reported having no conflicts of interest.
DENVER – After a failed first ablation procedure for paroxysmal atrial fibrillation, redo ablation proved more effective than did antiarrhythmic drug therapy in a randomized trial, Dr. Jonathan Steinberg reported at the annual meeting of the Heart Rhythm Society.
The success rate of a first ablation procedure in patients with symptomatic paroxysmal AF is typically about 60%. What to do for the 40% who are nonresponders has been unclear, with no prior randomized clinical trial evidence available to guide decisions, noted Dr. Steinberg of Columbia University, New York.
The study comprised 154 patients with recurrent symptomatic paroxysmal AF 3 months after an initial ablation procedure involving only pulmonary vein isolation. All participants received an implantable loop recorder to track atrial arrhythmic events.
They were then randomized to redo-ablation limited to reisolation of the pulmonary vein, which was successfully accomplished in all instances, or to guideline-based antiarrhythmic drug therapy. The choice of drug was left to individual investigator discretion. The three options were propafenone at 450-900 mg/day, sotalol at 160-320 mg/day, or flecainide at 200-400 mg/day. Propafenone was selected in the majority of cases, at an average dose of 579 mg/day.
The average AF burden as measured by implantable loop recorder at randomization was 15%. The primary study endpoint was AF burden at 36 months of follow-up, which was 5.6% in the redo-ablation group compared with 18.8% in the antiarrhythmic drug group.
Secondary endpoints uniformly favored redo-ablation as well.
Data from the implantable loop recorders was evaluated every 3 months during 3 years of follow-up. As early as 3 months into the study, the group given antiarrhythmic drugs had an AF burden of 3.3%, significantly higher than the 1.9% rate seen in the redo-ablation group. Thereafter, the drug therapy group experienced a gradual increase in AF burden throughout the first 12-15 months, followed by a much more substantial increase during the remainder of the study.
"The redo-ablation group had a different pattern" of AF burden, Dr. Steinberg observed. "It was low throughout the first 12-15 months, with just a slight increase, and it then rose only gradually over time until the 36-month end of the study."
Freedom from any atrial tachyarrhythmia at 1 year was 30% in the antiarrhythmic drug therapy group and 75% in the redo-ablation group. By 3 years, 12% of those in the drug therapy group were free of atrial tachyarrhythmias as were 58% in the redo-ablation group.
Complications in the redo-ablation group consisted of two cases of cardiac tamponade. In contrast, 49 patients, or 64%, in the antiarrhythmic drug therapy group discontinued medication due to intolerance or ineffectiveness.
Session cochair Dr. Gordon Tomaselli of Johns Hopkins University, Baltimore, said that in light of the potential proarrhythmic effects of virtually all antiarrhythmic drugs, it would have been useful to include a no-antiarrhythmic drug control group in the study.
Dr. Steinberg reported having no conflicts of interest.
AT HEART RHYTHM 2013
Major Finding: At 3 years after a first ablation procedure had failed for symptomatic paroxysmal atrial fibrillation, 12% of those randomized to drug therapy and 58% of those in the redo-ablation group were free of atrial tachyarrhythmias.
Data Source: A randomized, prospective, multicenter clinical trial involving 154 patients whose atrial arrhythmia status was monitored via implantable loop recorder.
Disclosures: The presenter reported having no conflicts of interest.
ACP restates call for inpatient blood glucose of 140-200 mg/dL
Blood glucose levels should be targeted to 140-200 mg/dL in surgical or medical ICU patients on insulin therapy, according to advice the American College of Physicians published online May 24 in the American Journal of Medical Quality.
Also, "clinicians should avoid targets less than ... 140 mg/dL because harms are likely to increase with lower blood glucose targets," the group said (Am. J. Med. Qual. 2013 [doi: 10.1177/1062860613489339]).
The advice isn’t new, but instead a restatement of ACP’s 2011 inpatient glycemic control guidelines reissued as part of its "Best Practice Advice" campaign, said Paul G. Shekelle, Ph.D., senior author of both the advice paper and guidelines (Ann. Intern. Med. 2011;154:260-7).
"This is based on the prior guidelines, so there’s nothing new here in that sense. The Best Practice Advice series sometimes runs in parallel to the guidelines, sometimes it is something completely different than any ACP guidelines, and sometimes, like this case, it runs asynchronous to the guidelines. Ideally, these will be more synchronous in the future," Dr. Shekelle, director of the RAND Corporation’s Southern California Evidence-Based Practice Center, said in an interview.
ACP’s advice is largely in keeping with glucose control recommendations from other groups, which have tended toward liberalization in recent years amid evidence that aggressive, euglycemic control in hospitalized patients, even if they have diabetes, doesn’t improve outcomes and carries too high a risk of hypoglycemia and its attendant problems.
"Nobody is advocating tight glycemic control anymore in the hospital. It isn’t necessary and may be harmful," said Dr. Etie S. Moghissi, the lead author on a 2009 inpatient glycemic control consensus statement issued by the American Association of Clinical Endocrinologists and American Diabetes Association (Diabetes Care 2009;32:1119-31).
The consensus statement recommended an upper limit of 180 mg/dL based on the pivotal NICE-SUGAR study, instead of 200 mg/dL, which Dr. Shekelle said ACP chose because it was the upper target limit in several of the additional studies upon which the group based its 2011 guidelines (N. Engl. .J Med. 2009;360:1283-97).
But Dr. Moghissi, who is with the department of medicine at the University of California, Los Angeles, said she’s concerned that 200 mg/dL might be too high.
"We know that when we set targets, people do not achieve them. So when we set a higher target, most of the time people go above that. The concern" is that a target of 200 mg/dL "may be perceived [as meaning that] a little bit over 200 mg/dL is okay," but "above 200 mg/dL, usually there are issues with increased risk of infection, poor wound healing, volume depletion," and other problems, she said in an interview.
Dr. Shekelle and Dr. Moghissi said they have no relevant disclosures.
Although interesting that this best practice advice comes more than 2 years after the ACP guidelines, it is still relevant, especially to hospitalists working in open ICUs.
At this point, everyone agrees that intensive insulin therapy leads to increased risk of hypoglycemia, which may lead to worse outcomes. However, in the extensively quoted NICE-SUGAR study, the mean glucose level achieved in the conventional group was 144mg/dL, far below from the 200mg/dL upper limit set by the ACP. There are insufficient and inconclusive studies on the wards, and thus, target ranges and recommendations for the wards are cautionary extrapolations from ICU studies.

|
| Dr. Pejvak Salehi |
It is important to note that the ADA/ACE, SHM and ACC/AHA all agree with the lower target capillary blood glucose of 140mg/dL on the wards and ICU.
Interestingly, all other entities recommend an upper target of 180mg/dL. In addition, the Surgical Care Improvement Project CMS Core Measure for postoperative day 1 and 2:00-6:00 a.m. CBG for cardiac surgery patients is currently set at 200mg/dL. I would caution hospitals with setting upper limits at 200mg/dL in the CCU. Until this core measure has changed, hospitals may see their SCIP measures worsening.
Dr. Pejvak Salehi is with the department of medicine, Portland, Ore., Veterans Affairs Medical Center. He has worked in glycemic management for several years.
Although interesting that this best practice advice comes more than 2 years after the ACP guidelines, it is still relevant, especially to hospitalists working in open ICUs.
At this point, everyone agrees that intensive insulin therapy leads to increased risk of hypoglycemia, which may lead to worse outcomes. However, in the extensively quoted NICE-SUGAR study, the mean glucose level achieved in the conventional group was 144mg/dL, far below from the 200mg/dL upper limit set by the ACP. There are insufficient and inconclusive studies on the wards, and thus, target ranges and recommendations for the wards are cautionary extrapolations from ICU studies.

|
| Dr. Pejvak Salehi |
It is important to note that the ADA/ACE, SHM and ACC/AHA all agree with the lower target capillary blood glucose of 140mg/dL on the wards and ICU.
Interestingly, all other entities recommend an upper target of 180mg/dL. In addition, the Surgical Care Improvement Project CMS Core Measure for postoperative day 1 and 2:00-6:00 a.m. CBG for cardiac surgery patients is currently set at 200mg/dL. I would caution hospitals with setting upper limits at 200mg/dL in the CCU. Until this core measure has changed, hospitals may see their SCIP measures worsening.
Dr. Pejvak Salehi is with the department of medicine, Portland, Ore., Veterans Affairs Medical Center. He has worked in glycemic management for several years.
Although interesting that this best practice advice comes more than 2 years after the ACP guidelines, it is still relevant, especially to hospitalists working in open ICUs.
At this point, everyone agrees that intensive insulin therapy leads to increased risk of hypoglycemia, which may lead to worse outcomes. However, in the extensively quoted NICE-SUGAR study, the mean glucose level achieved in the conventional group was 144mg/dL, far below from the 200mg/dL upper limit set by the ACP. There are insufficient and inconclusive studies on the wards, and thus, target ranges and recommendations for the wards are cautionary extrapolations from ICU studies.

|
| Dr. Pejvak Salehi |
It is important to note that the ADA/ACE, SHM and ACC/AHA all agree with the lower target capillary blood glucose of 140mg/dL on the wards and ICU.
Interestingly, all other entities recommend an upper target of 180mg/dL. In addition, the Surgical Care Improvement Project CMS Core Measure for postoperative day 1 and 2:00-6:00 a.m. CBG for cardiac surgery patients is currently set at 200mg/dL. I would caution hospitals with setting upper limits at 200mg/dL in the CCU. Until this core measure has changed, hospitals may see their SCIP measures worsening.
Dr. Pejvak Salehi is with the department of medicine, Portland, Ore., Veterans Affairs Medical Center. He has worked in glycemic management for several years.
Blood glucose levels should be targeted to 140-200 mg/dL in surgical or medical ICU patients on insulin therapy, according to advice the American College of Physicians published online May 24 in the American Journal of Medical Quality.
Also, "clinicians should avoid targets less than ... 140 mg/dL because harms are likely to increase with lower blood glucose targets," the group said (Am. J. Med. Qual. 2013 [doi: 10.1177/1062860613489339]).
The advice isn’t new, but instead a restatement of ACP’s 2011 inpatient glycemic control guidelines reissued as part of its "Best Practice Advice" campaign, said Paul G. Shekelle, Ph.D., senior author of both the advice paper and guidelines (Ann. Intern. Med. 2011;154:260-7).
"This is based on the prior guidelines, so there’s nothing new here in that sense. The Best Practice Advice series sometimes runs in parallel to the guidelines, sometimes it is something completely different than any ACP guidelines, and sometimes, like this case, it runs asynchronous to the guidelines. Ideally, these will be more synchronous in the future," Dr. Shekelle, director of the RAND Corporation’s Southern California Evidence-Based Practice Center, said in an interview.
ACP’s advice is largely in keeping with glucose control recommendations from other groups, which have tended toward liberalization in recent years amid evidence that aggressive, euglycemic control in hospitalized patients, even if they have diabetes, doesn’t improve outcomes and carries too high a risk of hypoglycemia and its attendant problems.
"Nobody is advocating tight glycemic control anymore in the hospital. It isn’t necessary and may be harmful," said Dr. Etie S. Moghissi, the lead author on a 2009 inpatient glycemic control consensus statement issued by the American Association of Clinical Endocrinologists and American Diabetes Association (Diabetes Care 2009;32:1119-31).
The consensus statement recommended an upper limit of 180 mg/dL based on the pivotal NICE-SUGAR study, instead of 200 mg/dL, which Dr. Shekelle said ACP chose because it was the upper target limit in several of the additional studies upon which the group based its 2011 guidelines (N. Engl. .J Med. 2009;360:1283-97).
But Dr. Moghissi, who is with the department of medicine at the University of California, Los Angeles, said she’s concerned that 200 mg/dL might be too high.
"We know that when we set targets, people do not achieve them. So when we set a higher target, most of the time people go above that. The concern" is that a target of 200 mg/dL "may be perceived [as meaning that] a little bit over 200 mg/dL is okay," but "above 200 mg/dL, usually there are issues with increased risk of infection, poor wound healing, volume depletion," and other problems, she said in an interview.
Dr. Shekelle and Dr. Moghissi said they have no relevant disclosures.
Blood glucose levels should be targeted to 140-200 mg/dL in surgical or medical ICU patients on insulin therapy, according to advice the American College of Physicians published online May 24 in the American Journal of Medical Quality.
Also, "clinicians should avoid targets less than ... 140 mg/dL because harms are likely to increase with lower blood glucose targets," the group said (Am. J. Med. Qual. 2013 [doi: 10.1177/1062860613489339]).
The advice isn’t new, but instead a restatement of ACP’s 2011 inpatient glycemic control guidelines reissued as part of its "Best Practice Advice" campaign, said Paul G. Shekelle, Ph.D., senior author of both the advice paper and guidelines (Ann. Intern. Med. 2011;154:260-7).
"This is based on the prior guidelines, so there’s nothing new here in that sense. The Best Practice Advice series sometimes runs in parallel to the guidelines, sometimes it is something completely different than any ACP guidelines, and sometimes, like this case, it runs asynchronous to the guidelines. Ideally, these will be more synchronous in the future," Dr. Shekelle, director of the RAND Corporation’s Southern California Evidence-Based Practice Center, said in an interview.
ACP’s advice is largely in keeping with glucose control recommendations from other groups, which have tended toward liberalization in recent years amid evidence that aggressive, euglycemic control in hospitalized patients, even if they have diabetes, doesn’t improve outcomes and carries too high a risk of hypoglycemia and its attendant problems.
"Nobody is advocating tight glycemic control anymore in the hospital. It isn’t necessary and may be harmful," said Dr. Etie S. Moghissi, the lead author on a 2009 inpatient glycemic control consensus statement issued by the American Association of Clinical Endocrinologists and American Diabetes Association (Diabetes Care 2009;32:1119-31).
The consensus statement recommended an upper limit of 180 mg/dL based on the pivotal NICE-SUGAR study, instead of 200 mg/dL, which Dr. Shekelle said ACP chose because it was the upper target limit in several of the additional studies upon which the group based its 2011 guidelines (N. Engl. .J Med. 2009;360:1283-97).
But Dr. Moghissi, who is with the department of medicine at the University of California, Los Angeles, said she’s concerned that 200 mg/dL might be too high.
"We know that when we set targets, people do not achieve them. So when we set a higher target, most of the time people go above that. The concern" is that a target of 200 mg/dL "may be perceived [as meaning that] a little bit over 200 mg/dL is okay," but "above 200 mg/dL, usually there are issues with increased risk of infection, poor wound healing, volume depletion," and other problems, she said in an interview.
Dr. Shekelle and Dr. Moghissi said they have no relevant disclosures.
FROM THE AMERICAN JOURNAL OF MEDICAL QUALITY
Medicare uses more brand-name drugs than does VA
Among patients with diabetes, use of brand-name drugs was two to three times higher for Medicare than for the Department of Veterans Affairs in 2008, according to a new study in Annals of Internal Medicine.
That led to increased costs "at a time when the U.S. government is facing substantial budget pressures," wrote Dr. Walid F. Gellad of the Veterans Affairs Center for Heath Equity Research and Promotion, Pittsburgh, and his associates (Ann. Intern. Med. 2013 June 11 [doi:10.7326/0003-4819-159-2-201307160-00664]).
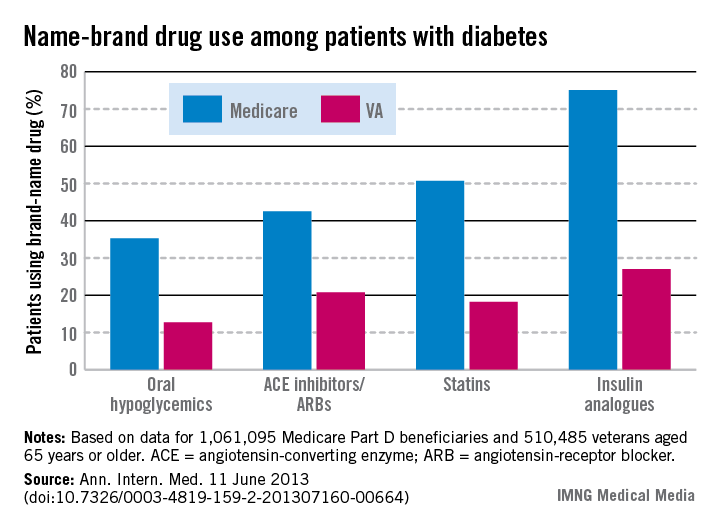
The investigators calculated the percentage of diabetes patients using brand name drugs for four common medication classes: oral hypoglycemics (35% for Medicare vs. 13% for VA), angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (43% for Medicare vs. 21% for VA), statins (51% vs. 18%), and insulin analogues (75% vs. 27%).
Had Medicare used generic drugs at a level comparable with the VA system, it would have saved over $1 billion on the diabetes population alone, the investigators noted.
The study was funded by the Department of Veterans Affairs, the National Institutes of Health, and the Robert Wood Johnson Foundation.
Among patients with diabetes, use of brand-name drugs was two to three times higher for Medicare than for the Department of Veterans Affairs in 2008, according to a new study in Annals of Internal Medicine.
That led to increased costs "at a time when the U.S. government is facing substantial budget pressures," wrote Dr. Walid F. Gellad of the Veterans Affairs Center for Heath Equity Research and Promotion, Pittsburgh, and his associates (Ann. Intern. Med. 2013 June 11 [doi:10.7326/0003-4819-159-2-201307160-00664]).
The investigators calculated the percentage of diabetes patients using brand name drugs for four common medication classes: oral hypoglycemics (35% for Medicare vs. 13% for VA), angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (43% for Medicare vs. 21% for VA), statins (51% vs. 18%), and insulin analogues (75% vs. 27%).
Had Medicare used generic drugs at a level comparable with the VA system, it would have saved over $1 billion on the diabetes population alone, the investigators noted.
The study was funded by the Department of Veterans Affairs, the National Institutes of Health, and the Robert Wood Johnson Foundation.
Among patients with diabetes, use of brand-name drugs was two to three times higher for Medicare than for the Department of Veterans Affairs in 2008, according to a new study in Annals of Internal Medicine.
That led to increased costs "at a time when the U.S. government is facing substantial budget pressures," wrote Dr. Walid F. Gellad of the Veterans Affairs Center for Heath Equity Research and Promotion, Pittsburgh, and his associates (Ann. Intern. Med. 2013 June 11 [doi:10.7326/0003-4819-159-2-201307160-00664]).
The investigators calculated the percentage of diabetes patients using brand name drugs for four common medication classes: oral hypoglycemics (35% for Medicare vs. 13% for VA), angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (43% for Medicare vs. 21% for VA), statins (51% vs. 18%), and insulin analogues (75% vs. 27%).
Had Medicare used generic drugs at a level comparable with the VA system, it would have saved over $1 billion on the diabetes population alone, the investigators noted.
The study was funded by the Department of Veterans Affairs, the National Institutes of Health, and the Robert Wood Johnson Foundation.
FROM ANNALS OF INTERNAL MEDICINE
Biomarkers predict response to cardiac resynchronization therapy
DENVER – Levels of troponin T and brain natriuretic peptide prior to implantation of a cardiac resynchronization device were strongly predictive of the risk of death or hospitalization for heart failure during the first year of device therapy in the BENEFIT study.
BENEFIT was a 1-year observational study undertaken to determine whether markers could predict which candidates for cardiac resynchronization therapy (CRT) were least likely to benefit from the costly device. At present, roughly 30% of patients who appropriately receive a CRT device do not respond to the treatment.
"We are all at this point frustrated with the persistent portion of CRT recipients who do not respond to therapy," BENEFIT principal investigator Dr. Alaa A. Shalaby said in presenting the study findings at the annual meeting of the Heart Rhythm Society. "We think our results suggest CRT should be offered earlier in the course of the disease"
BENEFIT included 267 CRT recipients at 33 centers. Patients were placed into one of three predefined risk categories based upon baseline levels of brain natriuretic peptide (BNP) and troponin T (TnT). On the basis os prior studies, high BNP was defined as 440 pg/mL or greater and a detectible TnT level was 0.01 ng/mL or greater. The low-risk group had an undetectable TnT and a BNP below 440 pg/mL. The intermediate-risk group had either an elevated BNP or TnT. The high-risk group had elevated BNP and TnT, reported Dr. Shalaby of the University of Pittsburgh.
The median baseline BNP in the study population was 198 pg/mL.
The three groups were similar in terms of baseline characteristics, including the proportion of patients with ischemic cardiomyopathy. Based on BNP and TnT results, 59% of patients were in the low-risk category, 33% had intermediate risk, and 8% were deemed high risk. The intermediate-risk group was evenly divided between patients who qualified on the basis of a high BNP and those with a detectable TnT.
During 12 months of follow-up there were 11 deaths and 19 heart failure hospitalizations. The incidence of either endpoint was 7% in the low-risk group, 15% in the intermediate-risk group, and 30% in the high-risk cohort.
After the researchers adjusted the results for age, left ventricular ejection fraction, QRS duration, and NYHA class, the risk of death or heart failure hospitalization was 2.5-fold greater in the group with intermediate-level baseline biomarkers as in the low-risk group. The high-biomarker group had a 7.3-fold increased risk, compared with the low-biomarker cohort. Both of these differences were statistically significant.
Changes in the biomarker levels after 6 and 12 months of CRT will be the subject of a future BENEFIT analysis, he noted.
The BENEFIT study was supported by St. Jude Medical. Dr. Shalaby reported having no conflicts of interest.
DENVER – Levels of troponin T and brain natriuretic peptide prior to implantation of a cardiac resynchronization device were strongly predictive of the risk of death or hospitalization for heart failure during the first year of device therapy in the BENEFIT study.
BENEFIT was a 1-year observational study undertaken to determine whether markers could predict which candidates for cardiac resynchronization therapy (CRT) were least likely to benefit from the costly device. At present, roughly 30% of patients who appropriately receive a CRT device do not respond to the treatment.
"We are all at this point frustrated with the persistent portion of CRT recipients who do not respond to therapy," BENEFIT principal investigator Dr. Alaa A. Shalaby said in presenting the study findings at the annual meeting of the Heart Rhythm Society. "We think our results suggest CRT should be offered earlier in the course of the disease"
BENEFIT included 267 CRT recipients at 33 centers. Patients were placed into one of three predefined risk categories based upon baseline levels of brain natriuretic peptide (BNP) and troponin T (TnT). On the basis os prior studies, high BNP was defined as 440 pg/mL or greater and a detectible TnT level was 0.01 ng/mL or greater. The low-risk group had an undetectable TnT and a BNP below 440 pg/mL. The intermediate-risk group had either an elevated BNP or TnT. The high-risk group had elevated BNP and TnT, reported Dr. Shalaby of the University of Pittsburgh.
The median baseline BNP in the study population was 198 pg/mL.
The three groups were similar in terms of baseline characteristics, including the proportion of patients with ischemic cardiomyopathy. Based on BNP and TnT results, 59% of patients were in the low-risk category, 33% had intermediate risk, and 8% were deemed high risk. The intermediate-risk group was evenly divided between patients who qualified on the basis of a high BNP and those with a detectable TnT.
During 12 months of follow-up there were 11 deaths and 19 heart failure hospitalizations. The incidence of either endpoint was 7% in the low-risk group, 15% in the intermediate-risk group, and 30% in the high-risk cohort.
After the researchers adjusted the results for age, left ventricular ejection fraction, QRS duration, and NYHA class, the risk of death or heart failure hospitalization was 2.5-fold greater in the group with intermediate-level baseline biomarkers as in the low-risk group. The high-biomarker group had a 7.3-fold increased risk, compared with the low-biomarker cohort. Both of these differences were statistically significant.
Changes in the biomarker levels after 6 and 12 months of CRT will be the subject of a future BENEFIT analysis, he noted.
The BENEFIT study was supported by St. Jude Medical. Dr. Shalaby reported having no conflicts of interest.
DENVER – Levels of troponin T and brain natriuretic peptide prior to implantation of a cardiac resynchronization device were strongly predictive of the risk of death or hospitalization for heart failure during the first year of device therapy in the BENEFIT study.
BENEFIT was a 1-year observational study undertaken to determine whether markers could predict which candidates for cardiac resynchronization therapy (CRT) were least likely to benefit from the costly device. At present, roughly 30% of patients who appropriately receive a CRT device do not respond to the treatment.
"We are all at this point frustrated with the persistent portion of CRT recipients who do not respond to therapy," BENEFIT principal investigator Dr. Alaa A. Shalaby said in presenting the study findings at the annual meeting of the Heart Rhythm Society. "We think our results suggest CRT should be offered earlier in the course of the disease"
BENEFIT included 267 CRT recipients at 33 centers. Patients were placed into one of three predefined risk categories based upon baseline levels of brain natriuretic peptide (BNP) and troponin T (TnT). On the basis os prior studies, high BNP was defined as 440 pg/mL or greater and a detectible TnT level was 0.01 ng/mL or greater. The low-risk group had an undetectable TnT and a BNP below 440 pg/mL. The intermediate-risk group had either an elevated BNP or TnT. The high-risk group had elevated BNP and TnT, reported Dr. Shalaby of the University of Pittsburgh.
The median baseline BNP in the study population was 198 pg/mL.
The three groups were similar in terms of baseline characteristics, including the proportion of patients with ischemic cardiomyopathy. Based on BNP and TnT results, 59% of patients were in the low-risk category, 33% had intermediate risk, and 8% were deemed high risk. The intermediate-risk group was evenly divided between patients who qualified on the basis of a high BNP and those with a detectable TnT.
During 12 months of follow-up there were 11 deaths and 19 heart failure hospitalizations. The incidence of either endpoint was 7% in the low-risk group, 15% in the intermediate-risk group, and 30% in the high-risk cohort.
After the researchers adjusted the results for age, left ventricular ejection fraction, QRS duration, and NYHA class, the risk of death or heart failure hospitalization was 2.5-fold greater in the group with intermediate-level baseline biomarkers as in the low-risk group. The high-biomarker group had a 7.3-fold increased risk, compared with the low-biomarker cohort. Both of these differences were statistically significant.
Changes in the biomarker levels after 6 and 12 months of CRT will be the subject of a future BENEFIT analysis, he noted.
The BENEFIT study was supported by St. Jude Medical. Dr. Shalaby reported having no conflicts of interest.
AT HEART RHYTHM 2013
Major finding: Cardiac resynchronization therapy device recipients with a preimplantation brain natriuretic peptide level of at least 440 pg/mL and a detectable troponin T level were 7.3-fold more likely to die or be hospitalized for heart failure during the first 12 months after device implantation than were those with an undetectable troponin T level and a lower brain natriuretic peptide level.
Data source: The BENEFIT study was a prospective, 33-center study in which 267 patients who received a CRT device were followed for 1 year.
Disclosures: BENEFIT was supported by St. Jude Medical. The presenter reported having no conflicts of interest.
Consider extended postop thromboprophylaxis when VTE risk reaches 0.88%
ORLANDO – Thromboprophylaxis for 3 weeks after discharge following abdominal surgery is more cost effective than is inpatient thromboprophylaxis alone, given certain base case assumptions, according to a decision tree analysis.
The findings, which incorporate both cost- and patient-related factors, provide clarity for clinicians about when extended duration thromboprophylaxis is appropriate, Dr. James C. Iannuzzi said at the annual Digestive Disease Week.
The analysis indicates that extended-duration thromboprophylaxis with low-molecular-weight heparin for 3 weeks after discharge is preferable to 7 days of inpatient-only thromboprophylaxis in cases where venous thromboembolism (VTE) risk is estimated at 0.88% to 2.39%; patient preferences regarding costs and medication administration, including the need for self-administered injection of low-molecular-weight heparin (LMWH), should be considered in these cases, said Dr. Iannuzzi of surgical health outcomes and research enterprise at the University of Rochester (N.Y.) Medical Center.
Extended-duration thromboprophylaxis dominates in cases in which the estimated VTE risk exceeds 2.39%, he said.
"Current guidelines are inconsistent about what metrics should be used, which makes it difficult for providers to decide whether extended-duration thromboprophylaxis would be beneficial. This likely plays a significant role in the current lack of adherence to guidelines," he said in an interview, adding. "This study was really aimed at providing clinicians a better cut-off for when it should be used, incorporating both the cost and patient perspective."
To assess cost effectiveness of thromboprophylaxis, Dr. Iannuzzi and his colleagues compared extended-duration treatment for 21 days after discharge (following 7 days of inpatient prophylaxis) with inpatient prophylaxis alone in a hypothetical case involving abdominal oncologic resection without complications in an otherwise healthy 45-year-old male patient.
Willingness to pay was set at $50,000 per quality-adjusted life-year (QALY), and the probabilities of various factors and scenarios were determined based on the available literature – much of it coming from the orthopedic literature. Costs were in U.S. 2013 dollars adjusted using the consumer price index; effectiveness was based on QALY (utility considered over 1 year), and cost effectiveness was evaluated using an incremental cost-effectiveness ratio, Dr. Iannuzzi explained.
Sensitivity analyses were performed to assess uncertainty within the model, with particular interest in the threshold for cost effectiveness based on VTE incidence, he said.
The endpoints were pulmonary embolism or deep vein thrombosis with attendant costs and assigned effectiveness evaluated by QALY, he said.
Based on the predetermined probabilities, and assuming an annualized cost of $23,248 for pulmonary embolism, $21,540 for deep vein thrombosis, $14,363 for post-thrombotic syndrome, $706 for generic LMWH, and $872 for brand-name LMWH, the threshold for the relative cost effectiveness of extended-duration thromboprophylaxis was VTE probability of 1.65% for brand-name LMWH, and 0.88% for generic LMWH.
"The 0.88% threshold is near the range of most major abdominal surgeries, which highlights the importance of using extended-duration thromboprophylaxis. However, the model was sensitive to changes in medication costs and patient values until postdischarge VTE risk exceeded 2.4%," Dr. Iannuzzi explained, adding that the model sensitivity in the 0.88% to 2.39% range is the reason why it is important to consider the patient perspective and the availability of generic LWMH in such cases.
He and his colleagues recently developed a risk score to help with postdischarge VTE prediction and to guide decision-making. The risk score, published online in May in the Journal of Vascular Surgery (J. Vasc. Surg. 2013 [doi:10.106/j.jvs.2012.12.073]), considers patient, operative, and early outcome factors to identify patients at increased risk.
The current findings, which pave the way for patient-centered decision making, use cost effectiveness of extended-duration thromboprophylaxis as a measure for risk, and should inform future guidelines’ definition of high risk. They also suggest that while a blanket approach to prophylaxis is not warranted, payers should cover the cost of extended-duration treatment.
"When cost was analyzed alone – without taking patient discomfort and the burden of self-injections into account, the threshold for cost effectiveness was much lower, suggesting that from the payer perspective, significant cost savings would be derived by increasing extended-duration thromboprophylaxis use," he explained.
Dr. Iannuzzi reported having no disclosures.
ORLANDO – Thromboprophylaxis for 3 weeks after discharge following abdominal surgery is more cost effective than is inpatient thromboprophylaxis alone, given certain base case assumptions, according to a decision tree analysis.
The findings, which incorporate both cost- and patient-related factors, provide clarity for clinicians about when extended duration thromboprophylaxis is appropriate, Dr. James C. Iannuzzi said at the annual Digestive Disease Week.
The analysis indicates that extended-duration thromboprophylaxis with low-molecular-weight heparin for 3 weeks after discharge is preferable to 7 days of inpatient-only thromboprophylaxis in cases where venous thromboembolism (VTE) risk is estimated at 0.88% to 2.39%; patient preferences regarding costs and medication administration, including the need for self-administered injection of low-molecular-weight heparin (LMWH), should be considered in these cases, said Dr. Iannuzzi of surgical health outcomes and research enterprise at the University of Rochester (N.Y.) Medical Center.
Extended-duration thromboprophylaxis dominates in cases in which the estimated VTE risk exceeds 2.39%, he said.
"Current guidelines are inconsistent about what metrics should be used, which makes it difficult for providers to decide whether extended-duration thromboprophylaxis would be beneficial. This likely plays a significant role in the current lack of adherence to guidelines," he said in an interview, adding. "This study was really aimed at providing clinicians a better cut-off for when it should be used, incorporating both the cost and patient perspective."
To assess cost effectiveness of thromboprophylaxis, Dr. Iannuzzi and his colleagues compared extended-duration treatment for 21 days after discharge (following 7 days of inpatient prophylaxis) with inpatient prophylaxis alone in a hypothetical case involving abdominal oncologic resection without complications in an otherwise healthy 45-year-old male patient.
Willingness to pay was set at $50,000 per quality-adjusted life-year (QALY), and the probabilities of various factors and scenarios were determined based on the available literature – much of it coming from the orthopedic literature. Costs were in U.S. 2013 dollars adjusted using the consumer price index; effectiveness was based on QALY (utility considered over 1 year), and cost effectiveness was evaluated using an incremental cost-effectiveness ratio, Dr. Iannuzzi explained.
Sensitivity analyses were performed to assess uncertainty within the model, with particular interest in the threshold for cost effectiveness based on VTE incidence, he said.
The endpoints were pulmonary embolism or deep vein thrombosis with attendant costs and assigned effectiveness evaluated by QALY, he said.
Based on the predetermined probabilities, and assuming an annualized cost of $23,248 for pulmonary embolism, $21,540 for deep vein thrombosis, $14,363 for post-thrombotic syndrome, $706 for generic LMWH, and $872 for brand-name LMWH, the threshold for the relative cost effectiveness of extended-duration thromboprophylaxis was VTE probability of 1.65% for brand-name LMWH, and 0.88% for generic LMWH.
"The 0.88% threshold is near the range of most major abdominal surgeries, which highlights the importance of using extended-duration thromboprophylaxis. However, the model was sensitive to changes in medication costs and patient values until postdischarge VTE risk exceeded 2.4%," Dr. Iannuzzi explained, adding that the model sensitivity in the 0.88% to 2.39% range is the reason why it is important to consider the patient perspective and the availability of generic LWMH in such cases.
He and his colleagues recently developed a risk score to help with postdischarge VTE prediction and to guide decision-making. The risk score, published online in May in the Journal of Vascular Surgery (J. Vasc. Surg. 2013 [doi:10.106/j.jvs.2012.12.073]), considers patient, operative, and early outcome factors to identify patients at increased risk.
The current findings, which pave the way for patient-centered decision making, use cost effectiveness of extended-duration thromboprophylaxis as a measure for risk, and should inform future guidelines’ definition of high risk. They also suggest that while a blanket approach to prophylaxis is not warranted, payers should cover the cost of extended-duration treatment.
"When cost was analyzed alone – without taking patient discomfort and the burden of self-injections into account, the threshold for cost effectiveness was much lower, suggesting that from the payer perspective, significant cost savings would be derived by increasing extended-duration thromboprophylaxis use," he explained.
Dr. Iannuzzi reported having no disclosures.
ORLANDO – Thromboprophylaxis for 3 weeks after discharge following abdominal surgery is more cost effective than is inpatient thromboprophylaxis alone, given certain base case assumptions, according to a decision tree analysis.
The findings, which incorporate both cost- and patient-related factors, provide clarity for clinicians about when extended duration thromboprophylaxis is appropriate, Dr. James C. Iannuzzi said at the annual Digestive Disease Week.
The analysis indicates that extended-duration thromboprophylaxis with low-molecular-weight heparin for 3 weeks after discharge is preferable to 7 days of inpatient-only thromboprophylaxis in cases where venous thromboembolism (VTE) risk is estimated at 0.88% to 2.39%; patient preferences regarding costs and medication administration, including the need for self-administered injection of low-molecular-weight heparin (LMWH), should be considered in these cases, said Dr. Iannuzzi of surgical health outcomes and research enterprise at the University of Rochester (N.Y.) Medical Center.
Extended-duration thromboprophylaxis dominates in cases in which the estimated VTE risk exceeds 2.39%, he said.
"Current guidelines are inconsistent about what metrics should be used, which makes it difficult for providers to decide whether extended-duration thromboprophylaxis would be beneficial. This likely plays a significant role in the current lack of adherence to guidelines," he said in an interview, adding. "This study was really aimed at providing clinicians a better cut-off for when it should be used, incorporating both the cost and patient perspective."
To assess cost effectiveness of thromboprophylaxis, Dr. Iannuzzi and his colleagues compared extended-duration treatment for 21 days after discharge (following 7 days of inpatient prophylaxis) with inpatient prophylaxis alone in a hypothetical case involving abdominal oncologic resection without complications in an otherwise healthy 45-year-old male patient.
Willingness to pay was set at $50,000 per quality-adjusted life-year (QALY), and the probabilities of various factors and scenarios were determined based on the available literature – much of it coming from the orthopedic literature. Costs were in U.S. 2013 dollars adjusted using the consumer price index; effectiveness was based on QALY (utility considered over 1 year), and cost effectiveness was evaluated using an incremental cost-effectiveness ratio, Dr. Iannuzzi explained.
Sensitivity analyses were performed to assess uncertainty within the model, with particular interest in the threshold for cost effectiveness based on VTE incidence, he said.
The endpoints were pulmonary embolism or deep vein thrombosis with attendant costs and assigned effectiveness evaluated by QALY, he said.
Based on the predetermined probabilities, and assuming an annualized cost of $23,248 for pulmonary embolism, $21,540 for deep vein thrombosis, $14,363 for post-thrombotic syndrome, $706 for generic LMWH, and $872 for brand-name LMWH, the threshold for the relative cost effectiveness of extended-duration thromboprophylaxis was VTE probability of 1.65% for brand-name LMWH, and 0.88% for generic LMWH.
"The 0.88% threshold is near the range of most major abdominal surgeries, which highlights the importance of using extended-duration thromboprophylaxis. However, the model was sensitive to changes in medication costs and patient values until postdischarge VTE risk exceeded 2.4%," Dr. Iannuzzi explained, adding that the model sensitivity in the 0.88% to 2.39% range is the reason why it is important to consider the patient perspective and the availability of generic LWMH in such cases.
He and his colleagues recently developed a risk score to help with postdischarge VTE prediction and to guide decision-making. The risk score, published online in May in the Journal of Vascular Surgery (J. Vasc. Surg. 2013 [doi:10.106/j.jvs.2012.12.073]), considers patient, operative, and early outcome factors to identify patients at increased risk.
The current findings, which pave the way for patient-centered decision making, use cost effectiveness of extended-duration thromboprophylaxis as a measure for risk, and should inform future guidelines’ definition of high risk. They also suggest that while a blanket approach to prophylaxis is not warranted, payers should cover the cost of extended-duration treatment.
"When cost was analyzed alone – without taking patient discomfort and the burden of self-injections into account, the threshold for cost effectiveness was much lower, suggesting that from the payer perspective, significant cost savings would be derived by increasing extended-duration thromboprophylaxis use," he explained.
Dr. Iannuzzi reported having no disclosures.
AT DDW 2013
Major finding: Extended-duration thromboprophylaxis is preferable to inpatient-only thromboprophylaxis when VTE risk is 0.88% or greater.
Data source: A decision tree analysis.
Disclosures: Dr. Iannuzzi reported having no disclosures.
Four factors predict likelihood of future knee replacement
PHILADELPHIA – Knowledge of a patient’s age, gender, quality of life score, and radiographic knee osteoarthritis severity helps predict the odds of a patient undergoing total knee arthroplasty in the next 5 years.
Few studies have looked at the predictors of total knee arthroplasty (TKA) in community-based cohorts, despite the heavy burden of TKA in the United States, said Dr. Marc C. Hochberg, head of rheumatology and clinical immunology, University of Maryland, Baltimore. An estimated 4 million Americans already live with TKA, and more than half of adults diagnosed with knee osteoarthritis (OA) will undergo TKA (J. Bone Joint Surg. Am. 2013;95:385-92).
For the current analysis, Dr. Hochberg and his associates obtained 48 months of annual clinical follow-up data on 6,406 patients in the Osteoarthritis Initiative who had symptomatic, radiographic knee OA or who were at risk for the condition. Follow-up at 60 months involved only questionnaires. Consensus readings of knee radiographs were used for the analysis, along with knee-specific multiple variable regression models. The best models were selected based on Chi-square values and area under the receiver operating characteristic curve (AUC). Radiographic knee OA Kellgren-Lawrence (KL) severity grade 0, 1, 2, 3, and 4 was present in the right knee of 36%, 18%, 28%, 14%, and 3.5% of patients and in the left knee of 38%, 18%, 26%, 14.4%, and 3.3% of patients.
A little more than half of participants were women (58.4%), mean body mass index was 28.7 kg/m2, mean Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score was 2.47 (0-20 scale), mean WOMAC function score was 8.33 (0-68 scale), and mean Knee injury and Osteoarthritis Outcome Score (KOOS) (quality of life) was 66.10 (0-100 scale). Their average age was 62.7, he said at the World Congress on Osteoarthritis, sponsored by the Osteoarthritis Research Society International.
During the 60 months of follow-up, there were 91 right and 102 left TKAs. The AUC for age, sex, and BMI was 0.64 for the right knee and 0.67 for the left knee.
The best TKA prediction models included age, sex, BMI, and KOOS quality-of-life score, improving the AUC to 0.78 for the right knee and 0.80 for the left knee, Dr. Hochberg said. Addition of the WOMAC pain score was associated with a marginal, but significant improvement in the AUC (0.79 and 0.80, respectively). When KL grade was added to these models, the AUC reached 0.89 and 0.91 for the right and left knees, respectively, he said. Sensitivity analyses failed to demonstrate additional effects of other baseline variables when added.
Limitations of the study included the relatively few TKAs, that most patients had KL grade 2 at baseline, proportional hazards analyses were not used, and the study did not consider a correlation between knees or the change in independent variables over time, he said.
In a prospective Canadian study, willingness to undergo surgery was the strongest predictor of the time to first hip or knee total joint replacement (TJR) (Arthritis Rheum. 2006;54:3212-20), while an international OARSI Task Force reported that pain and disability was higher in patients recommended for hip or knee TJR (Osteoarthritis Cartilage 2011;19:147-54). The Task Force, which included Dr. Hochberg, could not, however, identify cut points for pain and disability that would discriminate between those who did or did not get the nod for surgery.
The Osteoarthritis Initiative is sponsored by the National Institutes of Health. Dr. Hochberg reported serving as a consultant for Allergan, Bioiberica, Iroko, Merck, and Pfizer, and as a scientific/medical advisory board member for Eli Lilly and Merck.
PHILADELPHIA – Knowledge of a patient’s age, gender, quality of life score, and radiographic knee osteoarthritis severity helps predict the odds of a patient undergoing total knee arthroplasty in the next 5 years.
Few studies have looked at the predictors of total knee arthroplasty (TKA) in community-based cohorts, despite the heavy burden of TKA in the United States, said Dr. Marc C. Hochberg, head of rheumatology and clinical immunology, University of Maryland, Baltimore. An estimated 4 million Americans already live with TKA, and more than half of adults diagnosed with knee osteoarthritis (OA) will undergo TKA (J. Bone Joint Surg. Am. 2013;95:385-92).
For the current analysis, Dr. Hochberg and his associates obtained 48 months of annual clinical follow-up data on 6,406 patients in the Osteoarthritis Initiative who had symptomatic, radiographic knee OA or who were at risk for the condition. Follow-up at 60 months involved only questionnaires. Consensus readings of knee radiographs were used for the analysis, along with knee-specific multiple variable regression models. The best models were selected based on Chi-square values and area under the receiver operating characteristic curve (AUC). Radiographic knee OA Kellgren-Lawrence (KL) severity grade 0, 1, 2, 3, and 4 was present in the right knee of 36%, 18%, 28%, 14%, and 3.5% of patients and in the left knee of 38%, 18%, 26%, 14.4%, and 3.3% of patients.
A little more than half of participants were women (58.4%), mean body mass index was 28.7 kg/m2, mean Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score was 2.47 (0-20 scale), mean WOMAC function score was 8.33 (0-68 scale), and mean Knee injury and Osteoarthritis Outcome Score (KOOS) (quality of life) was 66.10 (0-100 scale). Their average age was 62.7, he said at the World Congress on Osteoarthritis, sponsored by the Osteoarthritis Research Society International.
During the 60 months of follow-up, there were 91 right and 102 left TKAs. The AUC for age, sex, and BMI was 0.64 for the right knee and 0.67 for the left knee.
The best TKA prediction models included age, sex, BMI, and KOOS quality-of-life score, improving the AUC to 0.78 for the right knee and 0.80 for the left knee, Dr. Hochberg said. Addition of the WOMAC pain score was associated with a marginal, but significant improvement in the AUC (0.79 and 0.80, respectively). When KL grade was added to these models, the AUC reached 0.89 and 0.91 for the right and left knees, respectively, he said. Sensitivity analyses failed to demonstrate additional effects of other baseline variables when added.
Limitations of the study included the relatively few TKAs, that most patients had KL grade 2 at baseline, proportional hazards analyses were not used, and the study did not consider a correlation between knees or the change in independent variables over time, he said.
In a prospective Canadian study, willingness to undergo surgery was the strongest predictor of the time to first hip or knee total joint replacement (TJR) (Arthritis Rheum. 2006;54:3212-20), while an international OARSI Task Force reported that pain and disability was higher in patients recommended for hip or knee TJR (Osteoarthritis Cartilage 2011;19:147-54). The Task Force, which included Dr. Hochberg, could not, however, identify cut points for pain and disability that would discriminate between those who did or did not get the nod for surgery.
The Osteoarthritis Initiative is sponsored by the National Institutes of Health. Dr. Hochberg reported serving as a consultant for Allergan, Bioiberica, Iroko, Merck, and Pfizer, and as a scientific/medical advisory board member for Eli Lilly and Merck.
PHILADELPHIA – Knowledge of a patient’s age, gender, quality of life score, and radiographic knee osteoarthritis severity helps predict the odds of a patient undergoing total knee arthroplasty in the next 5 years.
Few studies have looked at the predictors of total knee arthroplasty (TKA) in community-based cohorts, despite the heavy burden of TKA in the United States, said Dr. Marc C. Hochberg, head of rheumatology and clinical immunology, University of Maryland, Baltimore. An estimated 4 million Americans already live with TKA, and more than half of adults diagnosed with knee osteoarthritis (OA) will undergo TKA (J. Bone Joint Surg. Am. 2013;95:385-92).
For the current analysis, Dr. Hochberg and his associates obtained 48 months of annual clinical follow-up data on 6,406 patients in the Osteoarthritis Initiative who had symptomatic, radiographic knee OA or who were at risk for the condition. Follow-up at 60 months involved only questionnaires. Consensus readings of knee radiographs were used for the analysis, along with knee-specific multiple variable regression models. The best models were selected based on Chi-square values and area under the receiver operating characteristic curve (AUC). Radiographic knee OA Kellgren-Lawrence (KL) severity grade 0, 1, 2, 3, and 4 was present in the right knee of 36%, 18%, 28%, 14%, and 3.5% of patients and in the left knee of 38%, 18%, 26%, 14.4%, and 3.3% of patients.
A little more than half of participants were women (58.4%), mean body mass index was 28.7 kg/m2, mean Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score was 2.47 (0-20 scale), mean WOMAC function score was 8.33 (0-68 scale), and mean Knee injury and Osteoarthritis Outcome Score (KOOS) (quality of life) was 66.10 (0-100 scale). Their average age was 62.7, he said at the World Congress on Osteoarthritis, sponsored by the Osteoarthritis Research Society International.
During the 60 months of follow-up, there were 91 right and 102 left TKAs. The AUC for age, sex, and BMI was 0.64 for the right knee and 0.67 for the left knee.
The best TKA prediction models included age, sex, BMI, and KOOS quality-of-life score, improving the AUC to 0.78 for the right knee and 0.80 for the left knee, Dr. Hochberg said. Addition of the WOMAC pain score was associated with a marginal, but significant improvement in the AUC (0.79 and 0.80, respectively). When KL grade was added to these models, the AUC reached 0.89 and 0.91 for the right and left knees, respectively, he said. Sensitivity analyses failed to demonstrate additional effects of other baseline variables when added.
Limitations of the study included the relatively few TKAs, that most patients had KL grade 2 at baseline, proportional hazards analyses were not used, and the study did not consider a correlation between knees or the change in independent variables over time, he said.
In a prospective Canadian study, willingness to undergo surgery was the strongest predictor of the time to first hip or knee total joint replacement (TJR) (Arthritis Rheum. 2006;54:3212-20), while an international OARSI Task Force reported that pain and disability was higher in patients recommended for hip or knee TJR (Osteoarthritis Cartilage 2011;19:147-54). The Task Force, which included Dr. Hochberg, could not, however, identify cut points for pain and disability that would discriminate between those who did or did not get the nod for surgery.
The Osteoarthritis Initiative is sponsored by the National Institutes of Health. Dr. Hochberg reported serving as a consultant for Allergan, Bioiberica, Iroko, Merck, and Pfizer, and as a scientific/medical advisory board member for Eli Lilly and Merck.
AT OARSI
Major finding: The best TKA prediction models included age, sex, BMI, and KOOS quality-of-life score, improving the AUC to 0.78 for the right knee and 0.80 for the left knee.
Data source: Retrospective analysis of 91 right and 102 left TKAs among 6,406 patients with knee osteoarthritis in the multicenter, observational Osteoarthritis Initiative study.
Disclosures: The Osteoarthritis Initiative is sponsored by the National Institutes of Health. Dr. Hochberg reported serving as a consultant for Allergan, Bioiberica, Iroko, Merck, and Pfizer, and as a scientific/medical advisory board member for Eli Lilly and Merck.
Vancomycin, properly dosed, is as safe on the kidneys as linezolid
LAS VEGAS – Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL, according to researchers from the University of Virginia in Charlottesville.
"When correct dosing is performed, the use of alternative agents to treat Gram-positive infections – specifically to avoid [vancomycin] nephrotoxicity – is unnecessary. Vancomycin use in critically ill patients is no different than linezolid use regarding nephrotoxicity or new-onset need for hemodialysis," said lead investigator Stephen Davies, a resident in the department of surgery, said at the annual meeting of the Surgical Infection Society.
The kidney safety of vancomycin has been in doubt, with mixed results from prior studies. To shed light on the issue, Dr. Davies and his team compared renal outcomes in 298 critically ill patients treated with vancomycin for 571 Gram-positive infections with outcomes in 247 patients treated with linezolid for 475 Gram-positive infections.
Infection sites included the lungs, peritoneum, blood stream, and urinary tract, among others, and isolates included Staphylococcus aureus (77 methicillin-resistant S. aureus), Enterococcus faecium (67 vancomycin-resistant Enterococcus), E. faecalis, and Streptococcus species. Vancomycin patients were dosed at 15-20 mg/kg, with serial trough monitoring, and treated for a mean of 16.2 days, vs. 14.3 days for linezolid. Patients were on no other nephrotoxic agents.
In the end, "there were no [statistically significant] differences between the two groups regarding maximum creatinine during treatment, final creatinine after treatment, change in creatinine maximum and initial values, and final and initial creatinine values." There were also no differences "in new-onset hemodialysis or death," Dr. Davies said.
APACHE II (Acute Physiology and Chronic Health Evaluation II) scores above 30 and initial creatinine levels above 1.2 mg/dL both predicted the need for new-onset hemodialysis, but antibiotic choice did not.
Those same two variables also predicted an increase in creatinine of more than 1.0 mg/dl during treatment; vancomycin use did, as well (relative risk, vancomycin 0.49; 95% confidence interval: 0.25-0.94). Concerned, the team looked into the issue. "What we found was that a rise in creatinine was typically not encountered until trough levels greater than 20 mg/dL were reached. As long as you stay within the recommended doses, you should be safe," Dr. Davies said.
There were no statistically significant baseline differences between the two groups in renal function, hemodialysis, creatinine levels, or APACHE II scores.
Despite the results, Dr. Philip Barie, who helped moderate Dr. Davies’ talk, said in an interview that he’s still concerned about vancomycin kidney safety.
"We are having to use higher doses to treat tougher bugs, and whereas nephrotoxicity pretty much went away with vancomycin after they purified the drug [decades ago], now we are having to use higher doses more, and nephrotoxicity is beginning to creep back into the picture. You have to dose really carefully, and if you have an organism that is among the more resistant to vancomycin, the safest thing to do with vancomycin is to use linezolid," said Dr. Barie, a professor of surgery and public heath at Cornell University, New York.
Dr. Davies and Dr. Barie reported no relevant disclosures.
Multiple factors must be considered in determining the optimal antibiotic treatment of gram-positive infections including but not limited to clinical and microbiological efficacy, development of resistant microorganisms, adverse effects, and costs. Several recent meta-analyses have reported conflicting results regarding whether linezolid is superior to vancomycin in clinical and/or microbiological efficacy, but all have reported increased nephrotoxicity with vancomycin (PLoS ONE 2013;8:e58240; Int. J. Antimicrob. Agents 2013;41:426-33; Eur. J. Clin. Microbiol. Infect. Dis. 2013).
However, none of these performed metaregression analyses to determine whether there is an association between vancomycin troughs and risk of nephrotoxicity. While the study by Dr. Davies et al. suggests such a relationship, there is inadequate data to determine if the higher vancomycin troughs, which were associated with creatinine elevations above 1.0 mg/dL, were necessary to eradicate the Gram-positive infections. Furthermore, despite the adjustment for severity of illness, there may have been a selection bias in the initial choice of antibiotics. Lastly, treatment selection for Gram-positive infections should also take into account: the costs and resources necessary to monitor vancomycin troughs, and the feasibility of performing timely and appropriate dose adjustments based on troughs to achieve a therapeutic yet nontoxic level.
Lillian S. Kao, MD, FACS, of the department of surgery, University of Texas Health Science Center at Houston.
Multiple factors must be considered in determining the optimal antibiotic treatment of gram-positive infections including but not limited to clinical and microbiological efficacy, development of resistant microorganisms, adverse effects, and costs. Several recent meta-analyses have reported conflicting results regarding whether linezolid is superior to vancomycin in clinical and/or microbiological efficacy, but all have reported increased nephrotoxicity with vancomycin (PLoS ONE 2013;8:e58240; Int. J. Antimicrob. Agents 2013;41:426-33; Eur. J. Clin. Microbiol. Infect. Dis. 2013).
However, none of these performed metaregression analyses to determine whether there is an association between vancomycin troughs and risk of nephrotoxicity. While the study by Dr. Davies et al. suggests such a relationship, there is inadequate data to determine if the higher vancomycin troughs, which were associated with creatinine elevations above 1.0 mg/dL, were necessary to eradicate the Gram-positive infections. Furthermore, despite the adjustment for severity of illness, there may have been a selection bias in the initial choice of antibiotics. Lastly, treatment selection for Gram-positive infections should also take into account: the costs and resources necessary to monitor vancomycin troughs, and the feasibility of performing timely and appropriate dose adjustments based on troughs to achieve a therapeutic yet nontoxic level.
Lillian S. Kao, MD, FACS, of the department of surgery, University of Texas Health Science Center at Houston.
Multiple factors must be considered in determining the optimal antibiotic treatment of gram-positive infections including but not limited to clinical and microbiological efficacy, development of resistant microorganisms, adverse effects, and costs. Several recent meta-analyses have reported conflicting results regarding whether linezolid is superior to vancomycin in clinical and/or microbiological efficacy, but all have reported increased nephrotoxicity with vancomycin (PLoS ONE 2013;8:e58240; Int. J. Antimicrob. Agents 2013;41:426-33; Eur. J. Clin. Microbiol. Infect. Dis. 2013).
However, none of these performed metaregression analyses to determine whether there is an association between vancomycin troughs and risk of nephrotoxicity. While the study by Dr. Davies et al. suggests such a relationship, there is inadequate data to determine if the higher vancomycin troughs, which were associated with creatinine elevations above 1.0 mg/dL, were necessary to eradicate the Gram-positive infections. Furthermore, despite the adjustment for severity of illness, there may have been a selection bias in the initial choice of antibiotics. Lastly, treatment selection for Gram-positive infections should also take into account: the costs and resources necessary to monitor vancomycin troughs, and the feasibility of performing timely and appropriate dose adjustments based on troughs to achieve a therapeutic yet nontoxic level.
Lillian S. Kao, MD, FACS, of the department of surgery, University of Texas Health Science Center at Houston.
LAS VEGAS – Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL, according to researchers from the University of Virginia in Charlottesville.
"When correct dosing is performed, the use of alternative agents to treat Gram-positive infections – specifically to avoid [vancomycin] nephrotoxicity – is unnecessary. Vancomycin use in critically ill patients is no different than linezolid use regarding nephrotoxicity or new-onset need for hemodialysis," said lead investigator Stephen Davies, a resident in the department of surgery, said at the annual meeting of the Surgical Infection Society.
The kidney safety of vancomycin has been in doubt, with mixed results from prior studies. To shed light on the issue, Dr. Davies and his team compared renal outcomes in 298 critically ill patients treated with vancomycin for 571 Gram-positive infections with outcomes in 247 patients treated with linezolid for 475 Gram-positive infections.
Infection sites included the lungs, peritoneum, blood stream, and urinary tract, among others, and isolates included Staphylococcus aureus (77 methicillin-resistant S. aureus), Enterococcus faecium (67 vancomycin-resistant Enterococcus), E. faecalis, and Streptococcus species. Vancomycin patients were dosed at 15-20 mg/kg, with serial trough monitoring, and treated for a mean of 16.2 days, vs. 14.3 days for linezolid. Patients were on no other nephrotoxic agents.
In the end, "there were no [statistically significant] differences between the two groups regarding maximum creatinine during treatment, final creatinine after treatment, change in creatinine maximum and initial values, and final and initial creatinine values." There were also no differences "in new-onset hemodialysis or death," Dr. Davies said.
APACHE II (Acute Physiology and Chronic Health Evaluation II) scores above 30 and initial creatinine levels above 1.2 mg/dL both predicted the need for new-onset hemodialysis, but antibiotic choice did not.
Those same two variables also predicted an increase in creatinine of more than 1.0 mg/dl during treatment; vancomycin use did, as well (relative risk, vancomycin 0.49; 95% confidence interval: 0.25-0.94). Concerned, the team looked into the issue. "What we found was that a rise in creatinine was typically not encountered until trough levels greater than 20 mg/dL were reached. As long as you stay within the recommended doses, you should be safe," Dr. Davies said.
There were no statistically significant baseline differences between the two groups in renal function, hemodialysis, creatinine levels, or APACHE II scores.
Despite the results, Dr. Philip Barie, who helped moderate Dr. Davies’ talk, said in an interview that he’s still concerned about vancomycin kidney safety.
"We are having to use higher doses to treat tougher bugs, and whereas nephrotoxicity pretty much went away with vancomycin after they purified the drug [decades ago], now we are having to use higher doses more, and nephrotoxicity is beginning to creep back into the picture. You have to dose really carefully, and if you have an organism that is among the more resistant to vancomycin, the safest thing to do with vancomycin is to use linezolid," said Dr. Barie, a professor of surgery and public heath at Cornell University, New York.
Dr. Davies and Dr. Barie reported no relevant disclosures.
LAS VEGAS – Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL, according to researchers from the University of Virginia in Charlottesville.
"When correct dosing is performed, the use of alternative agents to treat Gram-positive infections – specifically to avoid [vancomycin] nephrotoxicity – is unnecessary. Vancomycin use in critically ill patients is no different than linezolid use regarding nephrotoxicity or new-onset need for hemodialysis," said lead investigator Stephen Davies, a resident in the department of surgery, said at the annual meeting of the Surgical Infection Society.
The kidney safety of vancomycin has been in doubt, with mixed results from prior studies. To shed light on the issue, Dr. Davies and his team compared renal outcomes in 298 critically ill patients treated with vancomycin for 571 Gram-positive infections with outcomes in 247 patients treated with linezolid for 475 Gram-positive infections.
Infection sites included the lungs, peritoneum, blood stream, and urinary tract, among others, and isolates included Staphylococcus aureus (77 methicillin-resistant S. aureus), Enterococcus faecium (67 vancomycin-resistant Enterococcus), E. faecalis, and Streptococcus species. Vancomycin patients were dosed at 15-20 mg/kg, with serial trough monitoring, and treated for a mean of 16.2 days, vs. 14.3 days for linezolid. Patients were on no other nephrotoxic agents.
In the end, "there were no [statistically significant] differences between the two groups regarding maximum creatinine during treatment, final creatinine after treatment, change in creatinine maximum and initial values, and final and initial creatinine values." There were also no differences "in new-onset hemodialysis or death," Dr. Davies said.
APACHE II (Acute Physiology and Chronic Health Evaluation II) scores above 30 and initial creatinine levels above 1.2 mg/dL both predicted the need for new-onset hemodialysis, but antibiotic choice did not.
Those same two variables also predicted an increase in creatinine of more than 1.0 mg/dl during treatment; vancomycin use did, as well (relative risk, vancomycin 0.49; 95% confidence interval: 0.25-0.94). Concerned, the team looked into the issue. "What we found was that a rise in creatinine was typically not encountered until trough levels greater than 20 mg/dL were reached. As long as you stay within the recommended doses, you should be safe," Dr. Davies said.
There were no statistically significant baseline differences between the two groups in renal function, hemodialysis, creatinine levels, or APACHE II scores.
Despite the results, Dr. Philip Barie, who helped moderate Dr. Davies’ talk, said in an interview that he’s still concerned about vancomycin kidney safety.
"We are having to use higher doses to treat tougher bugs, and whereas nephrotoxicity pretty much went away with vancomycin after they purified the drug [decades ago], now we are having to use higher doses more, and nephrotoxicity is beginning to creep back into the picture. You have to dose really carefully, and if you have an organism that is among the more resistant to vancomycin, the safest thing to do with vancomycin is to use linezolid," said Dr. Barie, a professor of surgery and public heath at Cornell University, New York.
Dr. Davies and Dr. Barie reported no relevant disclosures.
AT THE SIS ANNUAL MEETING
Major finding: Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL.
Data source: Single-center, retrospective cohort study in 545 critically ill patients
Disclosures: The lead investigator said he has no financial conflicts.