User login
Official Newspaper of the American College of Surgeons
ASCO offers practice guidance on small renal masses
All patients with small renal masses detected on imaging should be considered for renal tumor biopsy when there is a likelihood that the results may affect management of the patient, says a new clinical oncology practice guideline from the American Society of Clinical Oncology.
The guideline defines small renal masses as incidentally image-detected, contrast-enhancing renal tumors 4 cm in diameter or less that are usually consistent with stage T1a renal cell carcinoma (RCC). Approximately one-fourth of all small renal masses turn out to be benign lesions such as oncocytoma or metanephric adenoma, and another 25% may be indolent tumors that can be managed more conservatively, the guidelines note.
Not too long ago, nearly all patients with small renal masses would have undergone radical nephrectomy for lesions of any size. Today, however, partial nephrectomy and percutaneous thermal ablation are safe and less debilitating surgical options for many patients, the authors point out. The purpose of the guideline, therefore, is to help clinicians manage patients with clinically localized small renal masses with evidence-based clinical recommendations.
Recommendations summarized
The guideline, developed with consensus from a multidisciplinary panel, includes six evidence-based recommendations, all based on intermediate quality sources, with recommendation strengths running from moderate to strong. In summary, the guideline recommends:
- All patients with a small renal mass should be considered for renal tumor biopsy “when the results may alter management.”
- For patients with significant comorbidities and a limited life expectancy, active surveillance should be one of the initial management options. Absolute indications for active surveillance include if the patient is at high risk for anesthesia and intervention or has a life expectancy of less than 5 years. Active surveillance is a relative indication for those patients with significant risk of end-stage renal disease if treated, small renal masses less than 1 cm, or a life expectancy of less than 10 years.
- For all patients for whom an intervention is indicated and who have a tumor amenable to limited resection, partial nephrectomy should be the standard treatment offered.
- Percutaneous thermal ablation can be considered as an option for patients whose tumors can be completely ablated. A biopsy should be performed either prior to or at the time of ablation.
- Radical nephrectomy for small renal masses should be reserved only for patients whose tumors are significantly complex to allow for successful partial nephrectomy or for whom or where partial nephrectomy “may result in unacceptable morbidity even when performed at centers with expertise. Referral to a surgeon and a center with experience in partial nephrectomy should be considered.”
- If the patient has chronic kidney disease (CKD), defined as an estimated glomerular filtration rate less than 45 mL/min per 1.73 m2, or develops progressive CKD after treatment, he or she should be considered for referral to a nephrologist, especially if the CKD is associated with proteinuria.
The guideline also offers advice for clinicians on communicating with patients and coordinating all aspects of care in a complex care environment.
“To begin, remember that today’s empowered patient will expect a greater role in his or her care. This means taking steps to ensure the patient is well educated and informed. Clinicians should take the time to orient the patient to his or her care but also make available recommended sources for information, including both print materials and online information,” the guideline authors advise.
They also recommend that clinicians share the details of pathology reports and test results with patients, families, and caregivers using terminology they can understand, including a thorough explanation of cancer staging, tumor types, and clinical options. Patients should also be informed, if appropriate, about the availability of clinical trials.
The guideline is sponsored by ASCO, Dr. Finelli and multiple coauthors disclosed relationships with various drug and/or device companies.
All patients with small renal masses detected on imaging should be considered for renal tumor biopsy when there is a likelihood that the results may affect management of the patient, says a new clinical oncology practice guideline from the American Society of Clinical Oncology.
The guideline defines small renal masses as incidentally image-detected, contrast-enhancing renal tumors 4 cm in diameter or less that are usually consistent with stage T1a renal cell carcinoma (RCC). Approximately one-fourth of all small renal masses turn out to be benign lesions such as oncocytoma or metanephric adenoma, and another 25% may be indolent tumors that can be managed more conservatively, the guidelines note.
Not too long ago, nearly all patients with small renal masses would have undergone radical nephrectomy for lesions of any size. Today, however, partial nephrectomy and percutaneous thermal ablation are safe and less debilitating surgical options for many patients, the authors point out. The purpose of the guideline, therefore, is to help clinicians manage patients with clinically localized small renal masses with evidence-based clinical recommendations.
Recommendations summarized
The guideline, developed with consensus from a multidisciplinary panel, includes six evidence-based recommendations, all based on intermediate quality sources, with recommendation strengths running from moderate to strong. In summary, the guideline recommends:
- All patients with a small renal mass should be considered for renal tumor biopsy “when the results may alter management.”
- For patients with significant comorbidities and a limited life expectancy, active surveillance should be one of the initial management options. Absolute indications for active surveillance include if the patient is at high risk for anesthesia and intervention or has a life expectancy of less than 5 years. Active surveillance is a relative indication for those patients with significant risk of end-stage renal disease if treated, small renal masses less than 1 cm, or a life expectancy of less than 10 years.
- For all patients for whom an intervention is indicated and who have a tumor amenable to limited resection, partial nephrectomy should be the standard treatment offered.
- Percutaneous thermal ablation can be considered as an option for patients whose tumors can be completely ablated. A biopsy should be performed either prior to or at the time of ablation.
- Radical nephrectomy for small renal masses should be reserved only for patients whose tumors are significantly complex to allow for successful partial nephrectomy or for whom or where partial nephrectomy “may result in unacceptable morbidity even when performed at centers with expertise. Referral to a surgeon and a center with experience in partial nephrectomy should be considered.”
- If the patient has chronic kidney disease (CKD), defined as an estimated glomerular filtration rate less than 45 mL/min per 1.73 m2, or develops progressive CKD after treatment, he or she should be considered for referral to a nephrologist, especially if the CKD is associated with proteinuria.
The guideline also offers advice for clinicians on communicating with patients and coordinating all aspects of care in a complex care environment.
“To begin, remember that today’s empowered patient will expect a greater role in his or her care. This means taking steps to ensure the patient is well educated and informed. Clinicians should take the time to orient the patient to his or her care but also make available recommended sources for information, including both print materials and online information,” the guideline authors advise.
They also recommend that clinicians share the details of pathology reports and test results with patients, families, and caregivers using terminology they can understand, including a thorough explanation of cancer staging, tumor types, and clinical options. Patients should also be informed, if appropriate, about the availability of clinical trials.
The guideline is sponsored by ASCO, Dr. Finelli and multiple coauthors disclosed relationships with various drug and/or device companies.
All patients with small renal masses detected on imaging should be considered for renal tumor biopsy when there is a likelihood that the results may affect management of the patient, says a new clinical oncology practice guideline from the American Society of Clinical Oncology.
The guideline defines small renal masses as incidentally image-detected, contrast-enhancing renal tumors 4 cm in diameter or less that are usually consistent with stage T1a renal cell carcinoma (RCC). Approximately one-fourth of all small renal masses turn out to be benign lesions such as oncocytoma or metanephric adenoma, and another 25% may be indolent tumors that can be managed more conservatively, the guidelines note.
Not too long ago, nearly all patients with small renal masses would have undergone radical nephrectomy for lesions of any size. Today, however, partial nephrectomy and percutaneous thermal ablation are safe and less debilitating surgical options for many patients, the authors point out. The purpose of the guideline, therefore, is to help clinicians manage patients with clinically localized small renal masses with evidence-based clinical recommendations.
Recommendations summarized
The guideline, developed with consensus from a multidisciplinary panel, includes six evidence-based recommendations, all based on intermediate quality sources, with recommendation strengths running from moderate to strong. In summary, the guideline recommends:
- All patients with a small renal mass should be considered for renal tumor biopsy “when the results may alter management.”
- For patients with significant comorbidities and a limited life expectancy, active surveillance should be one of the initial management options. Absolute indications for active surveillance include if the patient is at high risk for anesthesia and intervention or has a life expectancy of less than 5 years. Active surveillance is a relative indication for those patients with significant risk of end-stage renal disease if treated, small renal masses less than 1 cm, or a life expectancy of less than 10 years.
- For all patients for whom an intervention is indicated and who have a tumor amenable to limited resection, partial nephrectomy should be the standard treatment offered.
- Percutaneous thermal ablation can be considered as an option for patients whose tumors can be completely ablated. A biopsy should be performed either prior to or at the time of ablation.
- Radical nephrectomy for small renal masses should be reserved only for patients whose tumors are significantly complex to allow for successful partial nephrectomy or for whom or where partial nephrectomy “may result in unacceptable morbidity even when performed at centers with expertise. Referral to a surgeon and a center with experience in partial nephrectomy should be considered.”
- If the patient has chronic kidney disease (CKD), defined as an estimated glomerular filtration rate less than 45 mL/min per 1.73 m2, or develops progressive CKD after treatment, he or she should be considered for referral to a nephrologist, especially if the CKD is associated with proteinuria.
The guideline also offers advice for clinicians on communicating with patients and coordinating all aspects of care in a complex care environment.
“To begin, remember that today’s empowered patient will expect a greater role in his or her care. This means taking steps to ensure the patient is well educated and informed. Clinicians should take the time to orient the patient to his or her care but also make available recommended sources for information, including both print materials and online information,” the guideline authors advise.
They also recommend that clinicians share the details of pathology reports and test results with patients, families, and caregivers using terminology they can understand, including a thorough explanation of cancer staging, tumor types, and clinical options. Patients should also be informed, if appropriate, about the availability of clinical trials.
The guideline is sponsored by ASCO, Dr. Finelli and multiple coauthors disclosed relationships with various drug and/or device companies.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The guideline recommends renal tumor biopsy for most patients with incidentally detected renal masses 4 cm or smaller.
Major finding: Approximately 25% of patients with incidental small renal masses will have benign lesions.
Data source: Evidence-based clinical guideline developed by a multidisciplinary panel.
Disclosures: The guideline is sponsored by ASCO, Dr. Finelli and multiple coauthors disclosed relationships with various drug and/or device companies.
Expert panel reaches consensus on hernia management recommendations
Those are key conclusions from a consensus statement based on a systematic review of existing evidence in the medical literature about ventral hernia management that were published in the January 2017 issue of the Annals of Surgery.
“Despite ventral hernias (VH) being one of the most common pathologies seen by clinicians, significant variability in management exists,” wrote the researchers, led by Mike K. Liang, MD, of the University of Texas Health Science Center at Houston. “Surveys of clinicians and review of nationwide databases of patients undergoing elective ventral hernia repair (VHR) demonstrate substantial heterogeneity in patient selection and clinical practice.”
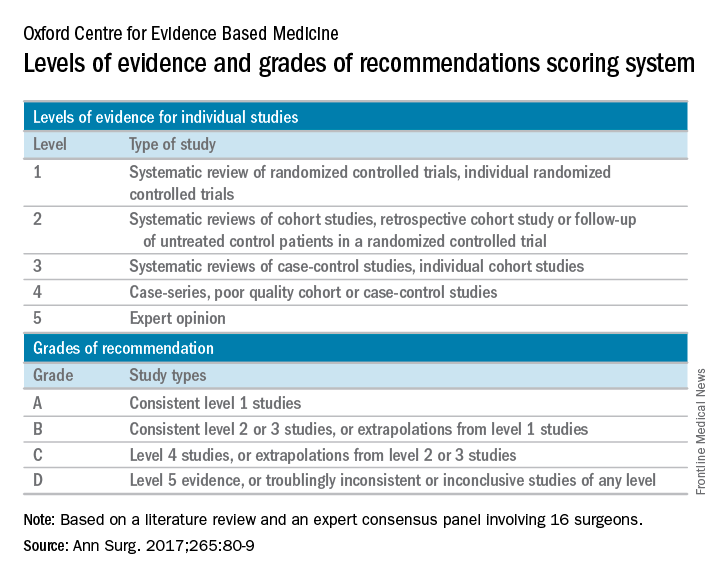
The panelists agreed that complications with VHR increase in obese patients (grade A evidence), current smokers (grade A), and in patients with glycosylated hemoglobin A1c (HbA1c) of 6.5% or greater (grade B). They did not recommend elective VHR in patients with a body mass index of 50 kg/m2 or greater (grade C), in current smokers (grade A), or patients with an HbA1c of 8.0% or greater (grade B). They also agreed that patients with a BMI of 30-50 kg/m2 or an HbA1c level of 6.5%-8.0% require individualized interventions to reduce surgical risk (grade C, grade B, respectively). The panelists considered nonoperative management to have a low risk of short-term morbidity (grade C) and they recommended mesh reinforcement for repair of hernias 2 cm or greater in size (grade A).
The panelists failed to reach agreement on several areas where high-quality data were limited, including mesh type. “Categories include ultra-light weight, light-weight, mid-weight, heavy-weight, and super-heavy weight, though precise definitions for each category are variable,” authors of the consensus statement wrote. “Randomized controlled trials are needed to compare synthetic, biological, and bioabsorbable meshes in all VH types and clinical settings.”
The authors of the consensus statement also called for further high-quality studies to better assess the management of VH in complex patients, which they defined as those presenting acutely, patients with cirrhosis, patients with inflammatory bowel disease, and patients who are pregnant.
The authors acknowledged certain limitations of the statement, including the fact that not all VH experts were included on the consensus panel. “However, the panel consisted of a large group of national experts with a primary practice focus of VHR,” they wrote. “The panelists have diverse views and unique areas of knowledge in the realm of hernia repair. The differing backgrounds among panelists was intended to make the guidelines that were developed more generalizable, as there is a wide variety of experience and skill level in the surgical community. In addition, there are no objective criteria to define an ‘expert’ in VH management.”
This work was supported by the Center for Clinical and Translational Sciences. The authors reported having no financial disclosures.
[email protected]
Those are key conclusions from a consensus statement based on a systematic review of existing evidence in the medical literature about ventral hernia management that were published in the January 2017 issue of the Annals of Surgery.
“Despite ventral hernias (VH) being one of the most common pathologies seen by clinicians, significant variability in management exists,” wrote the researchers, led by Mike K. Liang, MD, of the University of Texas Health Science Center at Houston. “Surveys of clinicians and review of nationwide databases of patients undergoing elective ventral hernia repair (VHR) demonstrate substantial heterogeneity in patient selection and clinical practice.”
The panelists agreed that complications with VHR increase in obese patients (grade A evidence), current smokers (grade A), and in patients with glycosylated hemoglobin A1c (HbA1c) of 6.5% or greater (grade B). They did not recommend elective VHR in patients with a body mass index of 50 kg/m2 or greater (grade C), in current smokers (grade A), or patients with an HbA1c of 8.0% or greater (grade B). They also agreed that patients with a BMI of 30-50 kg/m2 or an HbA1c level of 6.5%-8.0% require individualized interventions to reduce surgical risk (grade C, grade B, respectively). The panelists considered nonoperative management to have a low risk of short-term morbidity (grade C) and they recommended mesh reinforcement for repair of hernias 2 cm or greater in size (grade A).
The panelists failed to reach agreement on several areas where high-quality data were limited, including mesh type. “Categories include ultra-light weight, light-weight, mid-weight, heavy-weight, and super-heavy weight, though precise definitions for each category are variable,” authors of the consensus statement wrote. “Randomized controlled trials are needed to compare synthetic, biological, and bioabsorbable meshes in all VH types and clinical settings.”

The authors of the consensus statement also called for further high-quality studies to better assess the management of VH in complex patients, which they defined as those presenting acutely, patients with cirrhosis, patients with inflammatory bowel disease, and patients who are pregnant.
The authors acknowledged certain limitations of the statement, including the fact that not all VH experts were included on the consensus panel. “However, the panel consisted of a large group of national experts with a primary practice focus of VHR,” they wrote. “The panelists have diverse views and unique areas of knowledge in the realm of hernia repair. The differing backgrounds among panelists was intended to make the guidelines that were developed more generalizable, as there is a wide variety of experience and skill level in the surgical community. In addition, there are no objective criteria to define an ‘expert’ in VH management.”
This work was supported by the Center for Clinical and Translational Sciences. The authors reported having no financial disclosures.
[email protected]
Those are key conclusions from a consensus statement based on a systematic review of existing evidence in the medical literature about ventral hernia management that were published in the January 2017 issue of the Annals of Surgery.
“Despite ventral hernias (VH) being one of the most common pathologies seen by clinicians, significant variability in management exists,” wrote the researchers, led by Mike K. Liang, MD, of the University of Texas Health Science Center at Houston. “Surveys of clinicians and review of nationwide databases of patients undergoing elective ventral hernia repair (VHR) demonstrate substantial heterogeneity in patient selection and clinical practice.”
The panelists agreed that complications with VHR increase in obese patients (grade A evidence), current smokers (grade A), and in patients with glycosylated hemoglobin A1c (HbA1c) of 6.5% or greater (grade B). They did not recommend elective VHR in patients with a body mass index of 50 kg/m2 or greater (grade C), in current smokers (grade A), or patients with an HbA1c of 8.0% or greater (grade B). They also agreed that patients with a BMI of 30-50 kg/m2 or an HbA1c level of 6.5%-8.0% require individualized interventions to reduce surgical risk (grade C, grade B, respectively). The panelists considered nonoperative management to have a low risk of short-term morbidity (grade C) and they recommended mesh reinforcement for repair of hernias 2 cm or greater in size (grade A).
The panelists failed to reach agreement on several areas where high-quality data were limited, including mesh type. “Categories include ultra-light weight, light-weight, mid-weight, heavy-weight, and super-heavy weight, though precise definitions for each category are variable,” authors of the consensus statement wrote. “Randomized controlled trials are needed to compare synthetic, biological, and bioabsorbable meshes in all VH types and clinical settings.”

The authors of the consensus statement also called for further high-quality studies to better assess the management of VH in complex patients, which they defined as those presenting acutely, patients with cirrhosis, patients with inflammatory bowel disease, and patients who are pregnant.
The authors acknowledged certain limitations of the statement, including the fact that not all VH experts were included on the consensus panel. “However, the panel consisted of a large group of national experts with a primary practice focus of VHR,” they wrote. “The panelists have diverse views and unique areas of knowledge in the realm of hernia repair. The differing backgrounds among panelists was intended to make the guidelines that were developed more generalizable, as there is a wide variety of experience and skill level in the surgical community. In addition, there are no objective criteria to define an ‘expert’ in VH management.”
This work was supported by the Center for Clinical and Translational Sciences. The authors reported having no financial disclosures.
[email protected]
FROM ANNALS OF SURGERY
Scleroligation procedure proposed for gastroesophageal varices
In the treatment of gastroesophageal varices, scleroligation – a hybrid procedure that combines sclerotherapy and band ligation – performed as well as did band ligation, but required fewer sessions and had a shorter overall treatment duration. Sclerotherapy involves the injection of sclerosant to prompt occlusion of the varices, while ligation involves banding the varices to cut off blood flow.
The new approach combines them. The researchers ligated the varix 3-5 cm from the gastroesophageal junction and injected the sclerosant into the varix, below the ligated section. They reasoned that ligation should increase the contact time between the sclerosant and endothelial cells, and thus improve efficacy.
He noted that in the United States, band ligation is the standard of therapy, and the new study won’t change that. “These preliminary results from a single center are quite promising, and if they can be confirmed in a larger, multicenter trial, then this is something that can be added to our therapeutic armamentarium,” said Dr. Kwo.
Band ligation replaced sclerotherapy as the preferred treatment for gastroesophageal varices because it has equal efficacy but fewer severe side effects. However, it also suffers from a higher rate of recurrence because the bands cannot destroy deeper varices and perforating veins.
The combination technique, scleroligation, has been demonstrated in the treatment of esophageal varices, which prompted the authors’ investigation into gastroesophageal varices.
At a single center, the researchers recruited 120 patients with cirrhosis and acute gastroesophageal variceal bleeding above the gastroesophageal junction. They were randomized 1:1 to undergo endoscopic band ligation (EBL) or scleroligation (SL).
On average, 15.6 weeks were required to obliterate the varices in the EBL group versus 8.64 weeks in the SL group (P less than .001). The EBL group required an average of 3.43 sessions to reach that endpoint, compared with 2.22 sessions in the SL group (P less than .001). The EBL group required an average of 13.72 bands per patient, compared with 8.88 bands in the SL group (P less than .001). The EBL group also had a higher average number of units of blood transfused (2.30 vs 1.53; P less than .001).
No patients in either group experienced perforation, chest empyema, pericardial effusion, or strictures.
The good safety outcomes may be related to the small volume of sclerosant used, just 2 mL. “It’s probably because of their meticulous approach that they were able to reduce the complications that we have historically seen with sclerotherapy alone,” said Dr. Kwo.
There was no difference in the number of rebleeds or recurrences at follow-up, total cost, mortality due to rebleeding, or 12-month survival.
No funding source was disclosed. The authors reported no financial disclosures.
In the treatment of gastroesophageal varices, scleroligation – a hybrid procedure that combines sclerotherapy and band ligation – performed as well as did band ligation, but required fewer sessions and had a shorter overall treatment duration. Sclerotherapy involves the injection of sclerosant to prompt occlusion of the varices, while ligation involves banding the varices to cut off blood flow.
The new approach combines them. The researchers ligated the varix 3-5 cm from the gastroesophageal junction and injected the sclerosant into the varix, below the ligated section. They reasoned that ligation should increase the contact time between the sclerosant and endothelial cells, and thus improve efficacy.
He noted that in the United States, band ligation is the standard of therapy, and the new study won’t change that. “These preliminary results from a single center are quite promising, and if they can be confirmed in a larger, multicenter trial, then this is something that can be added to our therapeutic armamentarium,” said Dr. Kwo.
Band ligation replaced sclerotherapy as the preferred treatment for gastroesophageal varices because it has equal efficacy but fewer severe side effects. However, it also suffers from a higher rate of recurrence because the bands cannot destroy deeper varices and perforating veins.
The combination technique, scleroligation, has been demonstrated in the treatment of esophageal varices, which prompted the authors’ investigation into gastroesophageal varices.
At a single center, the researchers recruited 120 patients with cirrhosis and acute gastroesophageal variceal bleeding above the gastroesophageal junction. They were randomized 1:1 to undergo endoscopic band ligation (EBL) or scleroligation (SL).
On average, 15.6 weeks were required to obliterate the varices in the EBL group versus 8.64 weeks in the SL group (P less than .001). The EBL group required an average of 3.43 sessions to reach that endpoint, compared with 2.22 sessions in the SL group (P less than .001). The EBL group required an average of 13.72 bands per patient, compared with 8.88 bands in the SL group (P less than .001). The EBL group also had a higher average number of units of blood transfused (2.30 vs 1.53; P less than .001).
No patients in either group experienced perforation, chest empyema, pericardial effusion, or strictures.
The good safety outcomes may be related to the small volume of sclerosant used, just 2 mL. “It’s probably because of their meticulous approach that they were able to reduce the complications that we have historically seen with sclerotherapy alone,” said Dr. Kwo.
There was no difference in the number of rebleeds or recurrences at follow-up, total cost, mortality due to rebleeding, or 12-month survival.
No funding source was disclosed. The authors reported no financial disclosures.
In the treatment of gastroesophageal varices, scleroligation – a hybrid procedure that combines sclerotherapy and band ligation – performed as well as did band ligation, but required fewer sessions and had a shorter overall treatment duration. Sclerotherapy involves the injection of sclerosant to prompt occlusion of the varices, while ligation involves banding the varices to cut off blood flow.
The new approach combines them. The researchers ligated the varix 3-5 cm from the gastroesophageal junction and injected the sclerosant into the varix, below the ligated section. They reasoned that ligation should increase the contact time between the sclerosant and endothelial cells, and thus improve efficacy.
He noted that in the United States, band ligation is the standard of therapy, and the new study won’t change that. “These preliminary results from a single center are quite promising, and if they can be confirmed in a larger, multicenter trial, then this is something that can be added to our therapeutic armamentarium,” said Dr. Kwo.
Band ligation replaced sclerotherapy as the preferred treatment for gastroesophageal varices because it has equal efficacy but fewer severe side effects. However, it also suffers from a higher rate of recurrence because the bands cannot destroy deeper varices and perforating veins.
The combination technique, scleroligation, has been demonstrated in the treatment of esophageal varices, which prompted the authors’ investigation into gastroesophageal varices.
At a single center, the researchers recruited 120 patients with cirrhosis and acute gastroesophageal variceal bleeding above the gastroesophageal junction. They were randomized 1:1 to undergo endoscopic band ligation (EBL) or scleroligation (SL).
On average, 15.6 weeks were required to obliterate the varices in the EBL group versus 8.64 weeks in the SL group (P less than .001). The EBL group required an average of 3.43 sessions to reach that endpoint, compared with 2.22 sessions in the SL group (P less than .001). The EBL group required an average of 13.72 bands per patient, compared with 8.88 bands in the SL group (P less than .001). The EBL group also had a higher average number of units of blood transfused (2.30 vs 1.53; P less than .001).
No patients in either group experienced perforation, chest empyema, pericardial effusion, or strictures.
The good safety outcomes may be related to the small volume of sclerosant used, just 2 mL. “It’s probably because of their meticulous approach that they were able to reduce the complications that we have historically seen with sclerotherapy alone,” said Dr. Kwo.
There was no difference in the number of rebleeds or recurrences at follow-up, total cost, mortality due to rebleeding, or 12-month survival.
No funding source was disclosed. The authors reported no financial disclosures.
FROM GASTROINTESTINAL ENDOSCOPY
Key clinical point:
Major finding: Scleroligation required 2.22 sessions on average, compared with 3.43 for endoscopic band ligation.
Data source: Single-center randomized trial of 120 patients.
Disclosures: No funding source was disclosed. The authors reported no financial disclosures.
21 Medicare health plans warned to fix provider directory errors
Federal officials this month warned 21 Medicare Advantage insurers with high rates of errors in their online network directories that they could face heavy fines or have to stop enrolling people if the problems are not fixed by Feb. 6.
Among the plans that were cited are Blue Cross Blue Shield of Michigan, Highmark of Pennsylvania, SCAN Health Plan of California as well as some regional plans owned by national carriers such as UnitedHealthcare and Humana.
The action follows the government’s first in-depth review of the accuracy of Medicare Advantage provider directories, which consumers and advocates have complained about for years. More than 17 million Americans, or nearly a third of Medicare beneficiaries, get coverage through private Medicare Advantage plans.
The Centers for Medicare & Medicaid Services in October reported some of the results of the audit, but they had not released names or statistics from the individual plans.
“Because Medicare Advantage members rely on provider directories to locate an in-network provider, these inaccuracies pose a significant access-to-care barrier,” Medicare officials wrote in a report outlining the problems.
Unlike traditional Medicare, the private Medicare plans typically restrict beneficiaries to a network of doctors and hospitals.
Piedmont Community Health Plan, a small Medicare plan with about 5,200 members in southwest Virginia, had the highest rate of inaccuracies among the 54 insurers examined. Officials found errors in the listings of 87 of 108 doctors checked in Piedmont’s directory, according to the report. Most of the errors involved providing the wrong locations for doctors and doctors who should not have been listed. Piedmont officials did not return calls for comment.
Piedmont and two other plans with the highest error rates – a WellCare plan in Illinois and Emblem Health’s ConnectiCare subsidiary – were required by Medicare to submit specific business plans detailing how they intend to address the issue.
The individual plans receiving warning letters cover more than 1.4 million beneficiaries. Most operate in numerous states, although CMS generally limited its review to a specific state or geographic area.
The federal review focused on primary care doctors, cardiologists, ophthalmologists, and oncologists. It involved individual calls to check on the listings for 108 doctors in each health plan. “We encountered several instances where a call to a provider’s office resulted in determining that the provider had been retired or deceased for a long period of time, sometimes years,” the report said.
Almost half of the 5,832 doctors listed had incorrect information, including wrong addresses and wrong phone numbers. Most health plans had inaccurate information for 30%-60% of their providers’ offices, the report said. The report blamed the insurers for failing to do enough to keep their directories accurate.
“We saw a general lack of internal audit and testing of directory accuracy among many” Medicare Advantage organizations, the report said.
CMS’ survey found the most error-prone listings involved doctors with multiple offices that did not serve health plan members at each location.
The health plans were sent the warning letters Jan. 6 and given 30 days to fix the mistakes or face possible fines or sanctions, which could include suspending marketing and enrollment. CMS officials said the report was not issued before the annual open enrollment period – which ended Dec. 7 – because of the need to allow the health plans to review the findings before the report was made public.
Medicare Advantage members have until Feb. 14 to disenroll and join traditional Medicare but after that they are locked into their plan for the rest of the year. Seniors may be able to request permission to change plans on a case-by-case basis by calling 800-MEDICARE.
Another 32 companies with less serious mistakes also received letters saying their directories did not comply with a rule that took effect last year requiring plans to contact doctors and other providers every 3 months and to update their online directories in “real time.”
ConnectiCare spokeswoman Kimberly Kann acknowledged the difficulties. “Keeping these directories up to date is a two-way street and we are working with doctors and other medical professionals to continue providing quality service,” she said.
WellCare spokeswoman Crystal Warwell Walker said the Tampa, Fla.–based company took the survey results seriously. “We modified our data gathering techniques and online reporting options to ensure that when more than one address is listed for a provider, that provider is practicing at that location on a routine basis and access to care is not compromised,” she said.
CMS is continuing its investigation of provider directories this year and expects to examine all 300 companies by end of 2018.
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Federal officials this month warned 21 Medicare Advantage insurers with high rates of errors in their online network directories that they could face heavy fines or have to stop enrolling people if the problems are not fixed by Feb. 6.
Among the plans that were cited are Blue Cross Blue Shield of Michigan, Highmark of Pennsylvania, SCAN Health Plan of California as well as some regional plans owned by national carriers such as UnitedHealthcare and Humana.
The action follows the government’s first in-depth review of the accuracy of Medicare Advantage provider directories, which consumers and advocates have complained about for years. More than 17 million Americans, or nearly a third of Medicare beneficiaries, get coverage through private Medicare Advantage plans.
The Centers for Medicare & Medicaid Services in October reported some of the results of the audit, but they had not released names or statistics from the individual plans.
“Because Medicare Advantage members rely on provider directories to locate an in-network provider, these inaccuracies pose a significant access-to-care barrier,” Medicare officials wrote in a report outlining the problems.
Unlike traditional Medicare, the private Medicare plans typically restrict beneficiaries to a network of doctors and hospitals.
Piedmont Community Health Plan, a small Medicare plan with about 5,200 members in southwest Virginia, had the highest rate of inaccuracies among the 54 insurers examined. Officials found errors in the listings of 87 of 108 doctors checked in Piedmont’s directory, according to the report. Most of the errors involved providing the wrong locations for doctors and doctors who should not have been listed. Piedmont officials did not return calls for comment.
Piedmont and two other plans with the highest error rates – a WellCare plan in Illinois and Emblem Health’s ConnectiCare subsidiary – were required by Medicare to submit specific business plans detailing how they intend to address the issue.
The individual plans receiving warning letters cover more than 1.4 million beneficiaries. Most operate in numerous states, although CMS generally limited its review to a specific state or geographic area.
The federal review focused on primary care doctors, cardiologists, ophthalmologists, and oncologists. It involved individual calls to check on the listings for 108 doctors in each health plan. “We encountered several instances where a call to a provider’s office resulted in determining that the provider had been retired or deceased for a long period of time, sometimes years,” the report said.
Almost half of the 5,832 doctors listed had incorrect information, including wrong addresses and wrong phone numbers. Most health plans had inaccurate information for 30%-60% of their providers’ offices, the report said. The report blamed the insurers for failing to do enough to keep their directories accurate.
“We saw a general lack of internal audit and testing of directory accuracy among many” Medicare Advantage organizations, the report said.
CMS’ survey found the most error-prone listings involved doctors with multiple offices that did not serve health plan members at each location.
The health plans were sent the warning letters Jan. 6 and given 30 days to fix the mistakes or face possible fines or sanctions, which could include suspending marketing and enrollment. CMS officials said the report was not issued before the annual open enrollment period – which ended Dec. 7 – because of the need to allow the health plans to review the findings before the report was made public.
Medicare Advantage members have until Feb. 14 to disenroll and join traditional Medicare but after that they are locked into their plan for the rest of the year. Seniors may be able to request permission to change plans on a case-by-case basis by calling 800-MEDICARE.
Another 32 companies with less serious mistakes also received letters saying their directories did not comply with a rule that took effect last year requiring plans to contact doctors and other providers every 3 months and to update their online directories in “real time.”
ConnectiCare spokeswoman Kimberly Kann acknowledged the difficulties. “Keeping these directories up to date is a two-way street and we are working with doctors and other medical professionals to continue providing quality service,” she said.
WellCare spokeswoman Crystal Warwell Walker said the Tampa, Fla.–based company took the survey results seriously. “We modified our data gathering techniques and online reporting options to ensure that when more than one address is listed for a provider, that provider is practicing at that location on a routine basis and access to care is not compromised,” she said.
CMS is continuing its investigation of provider directories this year and expects to examine all 300 companies by end of 2018.
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Federal officials this month warned 21 Medicare Advantage insurers with high rates of errors in their online network directories that they could face heavy fines or have to stop enrolling people if the problems are not fixed by Feb. 6.
Among the plans that were cited are Blue Cross Blue Shield of Michigan, Highmark of Pennsylvania, SCAN Health Plan of California as well as some regional plans owned by national carriers such as UnitedHealthcare and Humana.
The action follows the government’s first in-depth review of the accuracy of Medicare Advantage provider directories, which consumers and advocates have complained about for years. More than 17 million Americans, or nearly a third of Medicare beneficiaries, get coverage through private Medicare Advantage plans.
The Centers for Medicare & Medicaid Services in October reported some of the results of the audit, but they had not released names or statistics from the individual plans.
“Because Medicare Advantage members rely on provider directories to locate an in-network provider, these inaccuracies pose a significant access-to-care barrier,” Medicare officials wrote in a report outlining the problems.
Unlike traditional Medicare, the private Medicare plans typically restrict beneficiaries to a network of doctors and hospitals.
Piedmont Community Health Plan, a small Medicare plan with about 5,200 members in southwest Virginia, had the highest rate of inaccuracies among the 54 insurers examined. Officials found errors in the listings of 87 of 108 doctors checked in Piedmont’s directory, according to the report. Most of the errors involved providing the wrong locations for doctors and doctors who should not have been listed. Piedmont officials did not return calls for comment.
Piedmont and two other plans with the highest error rates – a WellCare plan in Illinois and Emblem Health’s ConnectiCare subsidiary – were required by Medicare to submit specific business plans detailing how they intend to address the issue.
The individual plans receiving warning letters cover more than 1.4 million beneficiaries. Most operate in numerous states, although CMS generally limited its review to a specific state or geographic area.
The federal review focused on primary care doctors, cardiologists, ophthalmologists, and oncologists. It involved individual calls to check on the listings for 108 doctors in each health plan. “We encountered several instances where a call to a provider’s office resulted in determining that the provider had been retired or deceased for a long period of time, sometimes years,” the report said.
Almost half of the 5,832 doctors listed had incorrect information, including wrong addresses and wrong phone numbers. Most health plans had inaccurate information for 30%-60% of their providers’ offices, the report said. The report blamed the insurers for failing to do enough to keep their directories accurate.
“We saw a general lack of internal audit and testing of directory accuracy among many” Medicare Advantage organizations, the report said.
CMS’ survey found the most error-prone listings involved doctors with multiple offices that did not serve health plan members at each location.
The health plans were sent the warning letters Jan. 6 and given 30 days to fix the mistakes or face possible fines or sanctions, which could include suspending marketing and enrollment. CMS officials said the report was not issued before the annual open enrollment period – which ended Dec. 7 – because of the need to allow the health plans to review the findings before the report was made public.
Medicare Advantage members have until Feb. 14 to disenroll and join traditional Medicare but after that they are locked into their plan for the rest of the year. Seniors may be able to request permission to change plans on a case-by-case basis by calling 800-MEDICARE.
Another 32 companies with less serious mistakes also received letters saying their directories did not comply with a rule that took effect last year requiring plans to contact doctors and other providers every 3 months and to update their online directories in “real time.”
ConnectiCare spokeswoman Kimberly Kann acknowledged the difficulties. “Keeping these directories up to date is a two-way street and we are working with doctors and other medical professionals to continue providing quality service,” she said.
WellCare spokeswoman Crystal Warwell Walker said the Tampa, Fla.–based company took the survey results seriously. “We modified our data gathering techniques and online reporting options to ensure that when more than one address is listed for a provider, that provider is practicing at that location on a routine basis and access to care is not compromised,” she said.
CMS is continuing its investigation of provider directories this year and expects to examine all 300 companies by end of 2018.
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Repeal and replace: House bills offer potential road maps
As Republicans dig in to make good on their promise to repeal the Affordable Care Act, hints at what an eventual replacement may look like can be gleaned from legislation previously introduced in the House of Representatives.
One model could be the Empowering Patients First Act (H.R. 2300), sponsored by Rep. Tom Price (R-Ga.), a retired orthopedic surgeon and President-elect Trump’s nominee to head the Health and Human Services department. That legislation offers up a number of the usual GOP proposals related to health care, including refundable tax credits for low-income individuals buying insurance on the individual market; federal grants for states to provide health coverage through a high-risk pool, a reinsurance pool, or other mechanism to help subsidize the purchase of insurance; allowing individuals to purchase insurance through individual member associations; allowing small business owners to purchase insurance for their families and employees across state lines through their trade associations; and allowing insurance companies to sell coverage across state lines.
Another model can be found in House Republican’s health reform plan, called “A Better Way.”
The plan includes a number of provisions similar to those in Dr. Price’s plan, such as expanding consumer-directed health care options, allowing sale of insurance across state lines, expanding opportunities for pooling, and bringing about medical liability reform.
It also increases health insurance portability, helps to preserve the employer-sponsored insurance market, preserves wellness programs, promotes greater use of health savings accounts, and provides for greater opportunities to contribute and use them.
The Better Way plan maintains a few of the popular aspects of the Affordable Care Act, including a ban on coverage denial for preexisting conditions and the ability to keep adult children on parents’ health insurance up to age 26 years, in certain situations.
In addition, the plan would roll back a premium adjustment for older patients. The ACA mandates that premiums for older individuals could be no more than three times that of a younger enrollee. Prior to the ACA, the GOP plan notes that it was generally a limit of five times that of a younger enrollee, and the GOP vision is to bring that limit back.
“The ill-advised three-to-one policy is leading to artificially higher premiums for millions of Americans, especially younger and healthier patients,” according to the GOP plan.
The plan aims to repeal several ACA provisions including the Independent Payment Advisory Board, the Center for Medicare & Medicaid Innovations, and the ban on physician-owned hospitals. It would also extend value-based insurance design to Medicare Advantage, combine Medicare Parts A & B, and reform to uncompensated care.
President-elect Trump also gave hints as to what might be contained in a health reform plan he is formulating. In a Jan. 14 interview with the Washington Post, Mr. Trump said his plan aims to provide “insurance for all” while requiring drug manufacturers to negotiate with Medicare and Medicaid on pricing.
As Republicans dig in to make good on their promise to repeal the Affordable Care Act, hints at what an eventual replacement may look like can be gleaned from legislation previously introduced in the House of Representatives.
One model could be the Empowering Patients First Act (H.R. 2300), sponsored by Rep. Tom Price (R-Ga.), a retired orthopedic surgeon and President-elect Trump’s nominee to head the Health and Human Services department. That legislation offers up a number of the usual GOP proposals related to health care, including refundable tax credits for low-income individuals buying insurance on the individual market; federal grants for states to provide health coverage through a high-risk pool, a reinsurance pool, or other mechanism to help subsidize the purchase of insurance; allowing individuals to purchase insurance through individual member associations; allowing small business owners to purchase insurance for their families and employees across state lines through their trade associations; and allowing insurance companies to sell coverage across state lines.
Another model can be found in House Republican’s health reform plan, called “A Better Way.”
The plan includes a number of provisions similar to those in Dr. Price’s plan, such as expanding consumer-directed health care options, allowing sale of insurance across state lines, expanding opportunities for pooling, and bringing about medical liability reform.
It also increases health insurance portability, helps to preserve the employer-sponsored insurance market, preserves wellness programs, promotes greater use of health savings accounts, and provides for greater opportunities to contribute and use them.
The Better Way plan maintains a few of the popular aspects of the Affordable Care Act, including a ban on coverage denial for preexisting conditions and the ability to keep adult children on parents’ health insurance up to age 26 years, in certain situations.
In addition, the plan would roll back a premium adjustment for older patients. The ACA mandates that premiums for older individuals could be no more than three times that of a younger enrollee. Prior to the ACA, the GOP plan notes that it was generally a limit of five times that of a younger enrollee, and the GOP vision is to bring that limit back.
“The ill-advised three-to-one policy is leading to artificially higher premiums for millions of Americans, especially younger and healthier patients,” according to the GOP plan.
The plan aims to repeal several ACA provisions including the Independent Payment Advisory Board, the Center for Medicare & Medicaid Innovations, and the ban on physician-owned hospitals. It would also extend value-based insurance design to Medicare Advantage, combine Medicare Parts A & B, and reform to uncompensated care.
President-elect Trump also gave hints as to what might be contained in a health reform plan he is formulating. In a Jan. 14 interview with the Washington Post, Mr. Trump said his plan aims to provide “insurance for all” while requiring drug manufacturers to negotiate with Medicare and Medicaid on pricing.
As Republicans dig in to make good on their promise to repeal the Affordable Care Act, hints at what an eventual replacement may look like can be gleaned from legislation previously introduced in the House of Representatives.
One model could be the Empowering Patients First Act (H.R. 2300), sponsored by Rep. Tom Price (R-Ga.), a retired orthopedic surgeon and President-elect Trump’s nominee to head the Health and Human Services department. That legislation offers up a number of the usual GOP proposals related to health care, including refundable tax credits for low-income individuals buying insurance on the individual market; federal grants for states to provide health coverage through a high-risk pool, a reinsurance pool, or other mechanism to help subsidize the purchase of insurance; allowing individuals to purchase insurance through individual member associations; allowing small business owners to purchase insurance for their families and employees across state lines through their trade associations; and allowing insurance companies to sell coverage across state lines.
Another model can be found in House Republican’s health reform plan, called “A Better Way.”
The plan includes a number of provisions similar to those in Dr. Price’s plan, such as expanding consumer-directed health care options, allowing sale of insurance across state lines, expanding opportunities for pooling, and bringing about medical liability reform.
It also increases health insurance portability, helps to preserve the employer-sponsored insurance market, preserves wellness programs, promotes greater use of health savings accounts, and provides for greater opportunities to contribute and use them.
The Better Way plan maintains a few of the popular aspects of the Affordable Care Act, including a ban on coverage denial for preexisting conditions and the ability to keep adult children on parents’ health insurance up to age 26 years, in certain situations.
In addition, the plan would roll back a premium adjustment for older patients. The ACA mandates that premiums for older individuals could be no more than three times that of a younger enrollee. Prior to the ACA, the GOP plan notes that it was generally a limit of five times that of a younger enrollee, and the GOP vision is to bring that limit back.
“The ill-advised three-to-one policy is leading to artificially higher premiums for millions of Americans, especially younger and healthier patients,” according to the GOP plan.
The plan aims to repeal several ACA provisions including the Independent Payment Advisory Board, the Center for Medicare & Medicaid Innovations, and the ban on physician-owned hospitals. It would also extend value-based insurance design to Medicare Advantage, combine Medicare Parts A & B, and reform to uncompensated care.
President-elect Trump also gave hints as to what might be contained in a health reform plan he is formulating. In a Jan. 14 interview with the Washington Post, Mr. Trump said his plan aims to provide “insurance for all” while requiring drug manufacturers to negotiate with Medicare and Medicaid on pricing.
Dr. Price light on ACA replacement details at Senate hearing
WASHINGTON – Rep. Tom Price (R-Ga.) was light on specifics as to what he would favor in ACA replacement efforts, instead focused on broad goals for reform at a courtesy hearing Jan. 18 before the Senate Committee on Health, Education, Labor & Pensions.
Democratic senators on committee sought firm commitments on many issues – maintaining insurance coverage, women’s access to reproductive health care, coverage of mental health/substance use treatment, drug pricing, and reducing racial disparities – from Dr. Price, President-elect Trump’s nominee to lead the Health & Human Service department and a retired orthopedic surgeon. They also challenged Dr. Price on financial conflicts of interest related to legislation he supported.
Dr. Price consistently avoided committing to specific policies, but insisted that “individuals [should] have the opportunity to gain access to the kind of coverage they desire.”
Senators specifically queried Dr. Price as to whether he would commit to maintaining copay-free insurance coverage of all 18 forms of birth control for women approved by the Food and Drug Administration, as mandated by the ACA.
“Every single American ought to have access to the coverage and care that they desire,” Dr. Price responded.
Similarly, regarding coverage of mental health and substance use disorders, Dr. Price called it an “absolutely an imperative” that “every single American” have access to the care for these health issues.
When pressed by Sen. Maggie Hassan (D-N.H.) to commit to ensuring that there would be no cuts to Medicaid funding for mental health care/substance use disorders, Dr. Price noted that “we will address that need.”
Senators also queried Dr. Price’s commitment to maintaining the HHS Office of Minority Health. Sen. Murray offered a number of statistics demonstrating how minority women in particular have benefited with coverage and access to health care under the ACA.
Dr. Price stopped well short of committing to keeping the office, but instead returned to his desire to pursue policies that ensure “every American has access to the care that they desire.”
Dr. Price did not commit to upholding Mr. Trump’s campaign promise that no dollars would be cut from Medicare; instead, he argued that money spent is the wrong metric to measure health care quality.
Regarding the Center for Medicare & Medicaid Innovation, Dr. Price said that the center has “great promise,” but he “opposed the mandatory nature” of some of its programs, highlighting the comprehensive joint replacement bundle, which he said limits how orthopedic surgeons practice.
Senators also paid special attention to Dr. Price’s potential conflicts of interest. Several pointed to medical industry stock purchases that occurred around the time he introduced legislation that could benefit these companies, including a device manufacturer that would potentially benefit from Dr. Price’s challenging of the comprehensive joint replacement bundle and of pharmaceutical companies that might see benefit from the drug provisions in the 21st Century Cures Act.
He vehemently denied any wrongdoing, noting that he regularly and consistently disclosed all security holdings as required by congressional ethics rules and said he did nothing different from what many people in Congress currently do.
Despite his assurances that his ethics have not been compromised, Sen. Murray called for an ethics probe to address any potential conflicts of interest before his confirmation vote.
In closing the hearing, Chairman Lamar Alexander (R-Tenn.) reiterated his plan for a phased timeline for ACA repeal and replacement, to be completed so that no one would lose coverage. He suggested that while legislative action could be swift, implementation could span years to minimize impact on insurance coverage and access to health care.
Dr. Price’s official confirmation hearing before the Senate Finance Committee is scheduled for Jan. 24.
WASHINGTON – Rep. Tom Price (R-Ga.) was light on specifics as to what he would favor in ACA replacement efforts, instead focused on broad goals for reform at a courtesy hearing Jan. 18 before the Senate Committee on Health, Education, Labor & Pensions.
Democratic senators on committee sought firm commitments on many issues – maintaining insurance coverage, women’s access to reproductive health care, coverage of mental health/substance use treatment, drug pricing, and reducing racial disparities – from Dr. Price, President-elect Trump’s nominee to lead the Health & Human Service department and a retired orthopedic surgeon. They also challenged Dr. Price on financial conflicts of interest related to legislation he supported.
Dr. Price consistently avoided committing to specific policies, but insisted that “individuals [should] have the opportunity to gain access to the kind of coverage they desire.”
Senators specifically queried Dr. Price as to whether he would commit to maintaining copay-free insurance coverage of all 18 forms of birth control for women approved by the Food and Drug Administration, as mandated by the ACA.
“Every single American ought to have access to the coverage and care that they desire,” Dr. Price responded.
Similarly, regarding coverage of mental health and substance use disorders, Dr. Price called it an “absolutely an imperative” that “every single American” have access to the care for these health issues.
When pressed by Sen. Maggie Hassan (D-N.H.) to commit to ensuring that there would be no cuts to Medicaid funding for mental health care/substance use disorders, Dr. Price noted that “we will address that need.”
Senators also queried Dr. Price’s commitment to maintaining the HHS Office of Minority Health. Sen. Murray offered a number of statistics demonstrating how minority women in particular have benefited with coverage and access to health care under the ACA.
Dr. Price stopped well short of committing to keeping the office, but instead returned to his desire to pursue policies that ensure “every American has access to the care that they desire.”
Dr. Price did not commit to upholding Mr. Trump’s campaign promise that no dollars would be cut from Medicare; instead, he argued that money spent is the wrong metric to measure health care quality.
Regarding the Center for Medicare & Medicaid Innovation, Dr. Price said that the center has “great promise,” but he “opposed the mandatory nature” of some of its programs, highlighting the comprehensive joint replacement bundle, which he said limits how orthopedic surgeons practice.
Senators also paid special attention to Dr. Price’s potential conflicts of interest. Several pointed to medical industry stock purchases that occurred around the time he introduced legislation that could benefit these companies, including a device manufacturer that would potentially benefit from Dr. Price’s challenging of the comprehensive joint replacement bundle and of pharmaceutical companies that might see benefit from the drug provisions in the 21st Century Cures Act.
He vehemently denied any wrongdoing, noting that he regularly and consistently disclosed all security holdings as required by congressional ethics rules and said he did nothing different from what many people in Congress currently do.
Despite his assurances that his ethics have not been compromised, Sen. Murray called for an ethics probe to address any potential conflicts of interest before his confirmation vote.
In closing the hearing, Chairman Lamar Alexander (R-Tenn.) reiterated his plan for a phased timeline for ACA repeal and replacement, to be completed so that no one would lose coverage. He suggested that while legislative action could be swift, implementation could span years to minimize impact on insurance coverage and access to health care.
Dr. Price’s official confirmation hearing before the Senate Finance Committee is scheduled for Jan. 24.
WASHINGTON – Rep. Tom Price (R-Ga.) was light on specifics as to what he would favor in ACA replacement efforts, instead focused on broad goals for reform at a courtesy hearing Jan. 18 before the Senate Committee on Health, Education, Labor & Pensions.
Democratic senators on committee sought firm commitments on many issues – maintaining insurance coverage, women’s access to reproductive health care, coverage of mental health/substance use treatment, drug pricing, and reducing racial disparities – from Dr. Price, President-elect Trump’s nominee to lead the Health & Human Service department and a retired orthopedic surgeon. They also challenged Dr. Price on financial conflicts of interest related to legislation he supported.
Dr. Price consistently avoided committing to specific policies, but insisted that “individuals [should] have the opportunity to gain access to the kind of coverage they desire.”
Senators specifically queried Dr. Price as to whether he would commit to maintaining copay-free insurance coverage of all 18 forms of birth control for women approved by the Food and Drug Administration, as mandated by the ACA.
“Every single American ought to have access to the coverage and care that they desire,” Dr. Price responded.
Similarly, regarding coverage of mental health and substance use disorders, Dr. Price called it an “absolutely an imperative” that “every single American” have access to the care for these health issues.
When pressed by Sen. Maggie Hassan (D-N.H.) to commit to ensuring that there would be no cuts to Medicaid funding for mental health care/substance use disorders, Dr. Price noted that “we will address that need.”
Senators also queried Dr. Price’s commitment to maintaining the HHS Office of Minority Health. Sen. Murray offered a number of statistics demonstrating how minority women in particular have benefited with coverage and access to health care under the ACA.
Dr. Price stopped well short of committing to keeping the office, but instead returned to his desire to pursue policies that ensure “every American has access to the care that they desire.”
Dr. Price did not commit to upholding Mr. Trump’s campaign promise that no dollars would be cut from Medicare; instead, he argued that money spent is the wrong metric to measure health care quality.
Regarding the Center for Medicare & Medicaid Innovation, Dr. Price said that the center has “great promise,” but he “opposed the mandatory nature” of some of its programs, highlighting the comprehensive joint replacement bundle, which he said limits how orthopedic surgeons practice.
Senators also paid special attention to Dr. Price’s potential conflicts of interest. Several pointed to medical industry stock purchases that occurred around the time he introduced legislation that could benefit these companies, including a device manufacturer that would potentially benefit from Dr. Price’s challenging of the comprehensive joint replacement bundle and of pharmaceutical companies that might see benefit from the drug provisions in the 21st Century Cures Act.
He vehemently denied any wrongdoing, noting that he regularly and consistently disclosed all security holdings as required by congressional ethics rules and said he did nothing different from what many people in Congress currently do.
Despite his assurances that his ethics have not been compromised, Sen. Murray called for an ethics probe to address any potential conflicts of interest before his confirmation vote.
In closing the hearing, Chairman Lamar Alexander (R-Tenn.) reiterated his plan for a phased timeline for ACA repeal and replacement, to be completed so that no one would lose coverage. He suggested that while legislative action could be swift, implementation could span years to minimize impact on insurance coverage and access to health care.
Dr. Price’s official confirmation hearing before the Senate Finance Committee is scheduled for Jan. 24.
Radiosurgery found not superior to open surgery for temporal lobe epilepsy
HOUSTON – Despite enrollment difficulties that limited the study, a recently completed randomized trial comparing radiosurgery with open lobectomy to treat temporal lobe epilepsy offers some guidance for patients and their physicians.
Radiosurgery’s noninferiority to open lobectomy couldn’t be shown from the ROSE (Radiosurgery or Open Surgery for Epilepsy) trial, but language deficits were similar – and quite small – by 3 years after either procedure. Expected visual field deficits were similar in each procedure as well. However, since the trial didn’t reach its target enrollment, several primary outcome measures could not be fully assessed.
On the face of it, radiosurgery has significant appeal. Although open resective surgery is effective, there’s still some risk of infection and blood loss, and neuropsychological changes as well as other focal neurologic deficits are seen. Still, the study saw many challenges, but the largest, according to the investigators, was in recruitment. “Patients like to choose,” said Nicholas M. Barbaro, MD, chair of the department of neurosurgery at Indiana University, Indianapolis. Dr. Barbaro, one of several ROSE coinvestigators who presented the study findings at the annual meeting of the American Epilepsy Society, noted that if patients felt that lobectomy was the best choice, then there would be no incentive to enter a trial where they might be randomized to radiosurgery. Also, he said, some patients might be reluctant to be irradiated, fearing short-term or long-term toxicity.
Trial hypotheses and protocols
The ROSE trial aimed to show that stereotactic radiosurgery (SRS) would not be inferior to anterior temporal lobectomy (ATL) in achieving a seizure-free state by months 25-36 post procedure. The lag to response after radiosurgery is about 1 year; seizure freedom, defined as 12 consecutive months with no seizures, was assessed from months 25 to 36 of the study for the primary outcome of seizure freedom.
Investigators also hypothesized that fewer SRS patients would have significant reductions in measures of language function; further, they predicted that patients in both treatment arms would experience improvements in quality of life (QOL), and that QOL would improve as seizure freedom increased. Finally, the trial sought to show that SRS was cost effective, compared with ATL, with the marginal cost-utility ratio dropping below $50,000 per quality-adjusted life-year (QALY).
Patients in the ATL arm received a standard “Spencer” ATL, with adequacy of resection assessed by MRI performed 3 months after surgery. An inadequate resection would have been classified as an adverse event, but all ATL patients had an adequate resection by study criteria, and all those whose histopathology was available (n = 20) had some hippocampal sclerosis.
Patients in the SRS arm had the amygdala and anterior 2 cm of the hippocampus, as well as the adjacent parahippocampal gyrus, irradiated. This resulted in a total treatment volume ranging from 5.5 to 7.5 cc. Patients received 4 Gy to the 50% isodose line, and treatment could involve an unlimited number of isocenters. The brain stem could receive no more than 10 Gy and the optic nerve and chiasm no more than 8 Gy. All treatment plans were cleared by the ROSE steering committee. The SRS patients had some variation in dose and volumes treated, but all were within the approved limits of the study.
Trial outcomes
As expected, the surgery arm achieved rapid seizure remission, while the SRS arm saw a steady increase in seizure-free numbers beginning at about 12 months after surgery. During study months 25-36, 78% of the ATL arm and 52% of the SRS arm were seizure free. “The null hypothesis of inferiority of SRS was not rejected,” said Mark Quigg, MD, professor of neurology at the University of Virginia, Charlottesville.
Most patients in both groups had no or minimal changes in verbal memory, with no significant differences between the groups at 36 months after treatment.
QOL measures improved rapidly for those who received open surgery, and more slowly for those in the radiosurgery arm, a pattern “consistent with the known association between improved seizure control and quality of life,” said John Langfitt, PhD, a neuropsychologist and professor of neurology and psychiatry at the University of Rochester (N.Y.). However, the study was underpowered to show noninferiority of SRS for QOL measures at 36 months.
“There was a preliminary trend toward reduced health care use over time in the open surgery arm,” said Dr. Langfitt, again noting that the earlier seizure control achieved in surgery reduced health care utilization for that group sooner than for the SRS group. “The power may be limited by sample size and the tendency of utilization to be highly skewed,” he said.
Also as expected, the ATL arm saw early surgery-related adverse events such as scalp wound infections, subdural hematomas, and deep vein thromboses. These were infrequent overall. In contrast, the SRS group saw more cerebral edema–related adverse events during months 9-18, with headaches, new neurologic deficits, and transient seizure exacerbation.
All but three patients received postoperative visual field testing. Of the patients receiving SRS, 34% (10 of 29) had an upper superior quadrant visual field defect, as did 42% (11 of 26) of patients in the ATL arm.
Since the primary treating surgeon and neurologist could not be blinded as to study arm, another neurologist who was blinded was responsible for assessing the outcome measures, and also could identify adverse events. The trial’s steering committee was also blinded to ongoing outcomes.
Pilot study results
A pilot study had previously found that SRS was comparable to the efficacy that had been seen in larger, prospective trials of open surgery, with about two-thirds of patients seizure free at 36 months. Although most patients experienced brief exacerbation of auras or complex partial seizures after radiosurgery, visual field defects were similar to those experienced by patients undergoing standard ATL. Overall, neuropsychological outcomes for those undergoing SRS in the pilot were good, with a low incidence of declines in language and verbal memory function of the dominant hemisphere, and no short-term affective changes were seen. SRS patients who were seizure free after the procedure experienced a significant improvement in QOL.
The promising pilot results contrasted with the limited findings of the ROSE study. In regard to seizure freedom in ROSE, said Dr. Quigg, “The data appear to show that radiosurgery is inferior to ATL, but the low power of the study means that we cannot conclude this with sufficient confidence. Nor can we conclude that the two treatments are noninferior.”
The study was partially funded by Elekta, the manufacturer of the Gamma Knife radiosurgery device used in the study. Dr. Barbaro reported no other disclosures. Dr. Langfitt reported being a consultant for Monteris. Dr. Quigg reported being an investigator for several antiepileptic drug trials sponsored by pharmaceutical companies.
[email protected]
On Twitter @karioakes
HOUSTON – Despite enrollment difficulties that limited the study, a recently completed randomized trial comparing radiosurgery with open lobectomy to treat temporal lobe epilepsy offers some guidance for patients and their physicians.
Radiosurgery’s noninferiority to open lobectomy couldn’t be shown from the ROSE (Radiosurgery or Open Surgery for Epilepsy) trial, but language deficits were similar – and quite small – by 3 years after either procedure. Expected visual field deficits were similar in each procedure as well. However, since the trial didn’t reach its target enrollment, several primary outcome measures could not be fully assessed.
On the face of it, radiosurgery has significant appeal. Although open resective surgery is effective, there’s still some risk of infection and blood loss, and neuropsychological changes as well as other focal neurologic deficits are seen. Still, the study saw many challenges, but the largest, according to the investigators, was in recruitment. “Patients like to choose,” said Nicholas M. Barbaro, MD, chair of the department of neurosurgery at Indiana University, Indianapolis. Dr. Barbaro, one of several ROSE coinvestigators who presented the study findings at the annual meeting of the American Epilepsy Society, noted that if patients felt that lobectomy was the best choice, then there would be no incentive to enter a trial where they might be randomized to radiosurgery. Also, he said, some patients might be reluctant to be irradiated, fearing short-term or long-term toxicity.
Trial hypotheses and protocols
The ROSE trial aimed to show that stereotactic radiosurgery (SRS) would not be inferior to anterior temporal lobectomy (ATL) in achieving a seizure-free state by months 25-36 post procedure. The lag to response after radiosurgery is about 1 year; seizure freedom, defined as 12 consecutive months with no seizures, was assessed from months 25 to 36 of the study for the primary outcome of seizure freedom.
Investigators also hypothesized that fewer SRS patients would have significant reductions in measures of language function; further, they predicted that patients in both treatment arms would experience improvements in quality of life (QOL), and that QOL would improve as seizure freedom increased. Finally, the trial sought to show that SRS was cost effective, compared with ATL, with the marginal cost-utility ratio dropping below $50,000 per quality-adjusted life-year (QALY).
Patients in the ATL arm received a standard “Spencer” ATL, with adequacy of resection assessed by MRI performed 3 months after surgery. An inadequate resection would have been classified as an adverse event, but all ATL patients had an adequate resection by study criteria, and all those whose histopathology was available (n = 20) had some hippocampal sclerosis.
Patients in the SRS arm had the amygdala and anterior 2 cm of the hippocampus, as well as the adjacent parahippocampal gyrus, irradiated. This resulted in a total treatment volume ranging from 5.5 to 7.5 cc. Patients received 4 Gy to the 50% isodose line, and treatment could involve an unlimited number of isocenters. The brain stem could receive no more than 10 Gy and the optic nerve and chiasm no more than 8 Gy. All treatment plans were cleared by the ROSE steering committee. The SRS patients had some variation in dose and volumes treated, but all were within the approved limits of the study.
Trial outcomes
As expected, the surgery arm achieved rapid seizure remission, while the SRS arm saw a steady increase in seizure-free numbers beginning at about 12 months after surgery. During study months 25-36, 78% of the ATL arm and 52% of the SRS arm were seizure free. “The null hypothesis of inferiority of SRS was not rejected,” said Mark Quigg, MD, professor of neurology at the University of Virginia, Charlottesville.
Most patients in both groups had no or minimal changes in verbal memory, with no significant differences between the groups at 36 months after treatment.
QOL measures improved rapidly for those who received open surgery, and more slowly for those in the radiosurgery arm, a pattern “consistent with the known association between improved seizure control and quality of life,” said John Langfitt, PhD, a neuropsychologist and professor of neurology and psychiatry at the University of Rochester (N.Y.). However, the study was underpowered to show noninferiority of SRS for QOL measures at 36 months.
“There was a preliminary trend toward reduced health care use over time in the open surgery arm,” said Dr. Langfitt, again noting that the earlier seizure control achieved in surgery reduced health care utilization for that group sooner than for the SRS group. “The power may be limited by sample size and the tendency of utilization to be highly skewed,” he said.
Also as expected, the ATL arm saw early surgery-related adverse events such as scalp wound infections, subdural hematomas, and deep vein thromboses. These were infrequent overall. In contrast, the SRS group saw more cerebral edema–related adverse events during months 9-18, with headaches, new neurologic deficits, and transient seizure exacerbation.
All but three patients received postoperative visual field testing. Of the patients receiving SRS, 34% (10 of 29) had an upper superior quadrant visual field defect, as did 42% (11 of 26) of patients in the ATL arm.
Since the primary treating surgeon and neurologist could not be blinded as to study arm, another neurologist who was blinded was responsible for assessing the outcome measures, and also could identify adverse events. The trial’s steering committee was also blinded to ongoing outcomes.
Pilot study results
A pilot study had previously found that SRS was comparable to the efficacy that had been seen in larger, prospective trials of open surgery, with about two-thirds of patients seizure free at 36 months. Although most patients experienced brief exacerbation of auras or complex partial seizures after radiosurgery, visual field defects were similar to those experienced by patients undergoing standard ATL. Overall, neuropsychological outcomes for those undergoing SRS in the pilot were good, with a low incidence of declines in language and verbal memory function of the dominant hemisphere, and no short-term affective changes were seen. SRS patients who were seizure free after the procedure experienced a significant improvement in QOL.
The promising pilot results contrasted with the limited findings of the ROSE study. In regard to seizure freedom in ROSE, said Dr. Quigg, “The data appear to show that radiosurgery is inferior to ATL, but the low power of the study means that we cannot conclude this with sufficient confidence. Nor can we conclude that the two treatments are noninferior.”
The study was partially funded by Elekta, the manufacturer of the Gamma Knife radiosurgery device used in the study. Dr. Barbaro reported no other disclosures. Dr. Langfitt reported being a consultant for Monteris. Dr. Quigg reported being an investigator for several antiepileptic drug trials sponsored by pharmaceutical companies.
[email protected]
On Twitter @karioakes
HOUSTON – Despite enrollment difficulties that limited the study, a recently completed randomized trial comparing radiosurgery with open lobectomy to treat temporal lobe epilepsy offers some guidance for patients and their physicians.
Radiosurgery’s noninferiority to open lobectomy couldn’t be shown from the ROSE (Radiosurgery or Open Surgery for Epilepsy) trial, but language deficits were similar – and quite small – by 3 years after either procedure. Expected visual field deficits were similar in each procedure as well. However, since the trial didn’t reach its target enrollment, several primary outcome measures could not be fully assessed.
On the face of it, radiosurgery has significant appeal. Although open resective surgery is effective, there’s still some risk of infection and blood loss, and neuropsychological changes as well as other focal neurologic deficits are seen. Still, the study saw many challenges, but the largest, according to the investigators, was in recruitment. “Patients like to choose,” said Nicholas M. Barbaro, MD, chair of the department of neurosurgery at Indiana University, Indianapolis. Dr. Barbaro, one of several ROSE coinvestigators who presented the study findings at the annual meeting of the American Epilepsy Society, noted that if patients felt that lobectomy was the best choice, then there would be no incentive to enter a trial where they might be randomized to radiosurgery. Also, he said, some patients might be reluctant to be irradiated, fearing short-term or long-term toxicity.
Trial hypotheses and protocols
The ROSE trial aimed to show that stereotactic radiosurgery (SRS) would not be inferior to anterior temporal lobectomy (ATL) in achieving a seizure-free state by months 25-36 post procedure. The lag to response after radiosurgery is about 1 year; seizure freedom, defined as 12 consecutive months with no seizures, was assessed from months 25 to 36 of the study for the primary outcome of seizure freedom.
Investigators also hypothesized that fewer SRS patients would have significant reductions in measures of language function; further, they predicted that patients in both treatment arms would experience improvements in quality of life (QOL), and that QOL would improve as seizure freedom increased. Finally, the trial sought to show that SRS was cost effective, compared with ATL, with the marginal cost-utility ratio dropping below $50,000 per quality-adjusted life-year (QALY).
Patients in the ATL arm received a standard “Spencer” ATL, with adequacy of resection assessed by MRI performed 3 months after surgery. An inadequate resection would have been classified as an adverse event, but all ATL patients had an adequate resection by study criteria, and all those whose histopathology was available (n = 20) had some hippocampal sclerosis.
Patients in the SRS arm had the amygdala and anterior 2 cm of the hippocampus, as well as the adjacent parahippocampal gyrus, irradiated. This resulted in a total treatment volume ranging from 5.5 to 7.5 cc. Patients received 4 Gy to the 50% isodose line, and treatment could involve an unlimited number of isocenters. The brain stem could receive no more than 10 Gy and the optic nerve and chiasm no more than 8 Gy. All treatment plans were cleared by the ROSE steering committee. The SRS patients had some variation in dose and volumes treated, but all were within the approved limits of the study.
Trial outcomes
As expected, the surgery arm achieved rapid seizure remission, while the SRS arm saw a steady increase in seizure-free numbers beginning at about 12 months after surgery. During study months 25-36, 78% of the ATL arm and 52% of the SRS arm were seizure free. “The null hypothesis of inferiority of SRS was not rejected,” said Mark Quigg, MD, professor of neurology at the University of Virginia, Charlottesville.
Most patients in both groups had no or minimal changes in verbal memory, with no significant differences between the groups at 36 months after treatment.
QOL measures improved rapidly for those who received open surgery, and more slowly for those in the radiosurgery arm, a pattern “consistent with the known association between improved seizure control and quality of life,” said John Langfitt, PhD, a neuropsychologist and professor of neurology and psychiatry at the University of Rochester (N.Y.). However, the study was underpowered to show noninferiority of SRS for QOL measures at 36 months.
“There was a preliminary trend toward reduced health care use over time in the open surgery arm,” said Dr. Langfitt, again noting that the earlier seizure control achieved in surgery reduced health care utilization for that group sooner than for the SRS group. “The power may be limited by sample size and the tendency of utilization to be highly skewed,” he said.
Also as expected, the ATL arm saw early surgery-related adverse events such as scalp wound infections, subdural hematomas, and deep vein thromboses. These were infrequent overall. In contrast, the SRS group saw more cerebral edema–related adverse events during months 9-18, with headaches, new neurologic deficits, and transient seizure exacerbation.
All but three patients received postoperative visual field testing. Of the patients receiving SRS, 34% (10 of 29) had an upper superior quadrant visual field defect, as did 42% (11 of 26) of patients in the ATL arm.
Since the primary treating surgeon and neurologist could not be blinded as to study arm, another neurologist who was blinded was responsible for assessing the outcome measures, and also could identify adverse events. The trial’s steering committee was also blinded to ongoing outcomes.
Pilot study results
A pilot study had previously found that SRS was comparable to the efficacy that had been seen in larger, prospective trials of open surgery, with about two-thirds of patients seizure free at 36 months. Although most patients experienced brief exacerbation of auras or complex partial seizures after radiosurgery, visual field defects were similar to those experienced by patients undergoing standard ATL. Overall, neuropsychological outcomes for those undergoing SRS in the pilot were good, with a low incidence of declines in language and verbal memory function of the dominant hemisphere, and no short-term affective changes were seen. SRS patients who were seizure free after the procedure experienced a significant improvement in QOL.
The promising pilot results contrasted with the limited findings of the ROSE study. In regard to seizure freedom in ROSE, said Dr. Quigg, “The data appear to show that radiosurgery is inferior to ATL, but the low power of the study means that we cannot conclude this with sufficient confidence. Nor can we conclude that the two treatments are noninferior.”
The study was partially funded by Elekta, the manufacturer of the Gamma Knife radiosurgery device used in the study. Dr. Barbaro reported no other disclosures. Dr. Langfitt reported being a consultant for Monteris. Dr. Quigg reported being an investigator for several antiepileptic drug trials sponsored by pharmaceutical companies.
[email protected]
On Twitter @karioakes
AT AES 2016
Key clinical point:
Major finding: During study months 25-36, 78% of the ATL arm and 52% of the SRS arm were seizure free.
Data source: Trial of 58 patients with temporal lobe epilepsy randomized to receive ATL or SRS.
Disclosures: The study was partially funded by Elekta, the manufacturer of the Gamma Knife radiosurgery device used in the study. Several of the presenting ROSE steering committee members reported financial relationships with pharmaceutical companies.
Burnout: Time to stop blaming the victims
Most surgeons today are familiar with professional burnout – in their colleagues, in surgical trainees, and perhaps, in themselves. But the understanding of burnout is evolving. The discussion is moving away from blaming physicians for their poor coping skills toward identifying the structural and organizational roots of burnout.
Burnout is a syndrome cause by work-related stress that features emotional exhaustion, depersonalization, and a decreased sense of personal accomplishment. A recent study of nearly 7,000 physicians using the Maslach Burnout Inventory found that 54.4% of those surveyed reported at least one symptom of burnout (Mayo Clin Proc. 2015 Dec;90[12]:1600-19). Other studies have found similar rates of burnout in the surgical specialties such as orthopedic, oncologic, cardiothoracic, and plastic surgery (JAMA Surg. 2014 Sep;149:948-53; Ann Surg Oncol 2011 May;18:1229-35; Internat J Cardiol. 2015 Jan 20;179:7-72; Aesthet Surg J. 2016 Sep 27. E-pub ahead of print).
A new paradigm of burnout
The paradigm of burnout as a personal issue that can be managed by individual coping strategies is giving way to an understanding that the structural roots of burnout require the shared responsibility of individuals and their work organizations to solve the problem. A revised approach has emerged: Physician burnout as a symptom not of personal failure to cope, but of institutional failure to adapt to new circumstances in the health care milieu. The growing number of physicians employed in large group practices and medical centers has come with a whole array of management problems that are only beginning to be recognized, and burnout may be one of the most challenging.
Tait D. Shanafelt, MD, of the Mayo Foundation for Medical Education and Research, and John H. Noseworthy, MD, president and CEO of the Mayo Clinic, both in Rochester, Minn., have partnered to distill years of study and practice on the issue of burnout to a set of organizational strategies to tackle the problem and describe the Mayo Clinic experience. The study, “Executive Leadership and Physician Well-Being: Nine Organizational Strategies to Promote Engagement and Reduce Burnout” (Mayo Clin Proc. 2016 Nov 18. doi. org/10.1016/j.mayocp.2016.10.004) reverses the conventional “blame the victim” approach and identifies instead institutional responsibility to address burnout.
“Increasing evidence over the last 10 years demonstrating links to quality of care, productivity, and turnover have raised appreciation … by organizations that they have a substantial stake in this issue and that they control many of the factors that contribute to this problem,” said Dr. Shanafelt in an interview.
Unintended consequences of the individual solution
The focus on individual responsibility can have unintended consequences. A physician suffering from burnout can take action by leaving his or her job or cutting back. Staff turnover, a phenomenon closely tied to burnout, is costly and damaging to productivity and patient care (Physician Leadersh J. 2015 May-Jun;2[3]:22-5); Health Care Manage Rev. 2004;29[1]2-7). These personal strategies may help individuals cope but can end up harming the institution and the work life of other staff members. Physicians experiencing burnout in their own lives can trigger the same condition in their colleagues.
The Mayo paper by Dr. Shanafelt and Dr. Noseworthy states, “Mistakenly, most hospitals, medical centers, and practice groups operate under the framework that burnout and professional satisfaction are solely the responsibility of the individual physician. This frequently results in organizations pursuing a narrow list of ‘solutions’ that are unlikely to result in meaningful progress (e.g., stress management workshops and individual training in mindfulness/resilience). Such strategies neglect the organizational factors that are the primary drivers of physician burnout and are correctly viewed with skepticism by physicians as an insincere effort by the organization to address the problem.”
Organizational strategies to reduce burnout
Dr. Shanafelt and Dr. Noseworthy developed a list of nine organizational strategies that have been shown to reduce burnout among doctors. A critical piece of this approach is the accumulated evidence of the financial burden of burnout among physicians in health care institutions. The approach is based on an informed leadership that recognizes the costs of inaction, without which a systemic solution is unlikely to be achieved.
1. Naming the issue and assessing the problem
Acknowledgment of burnout as an institutional problem and meaningful measurement of physician well-being are the initial steps in tackling the problem. This requires a sincere commitment at the highest level of management to listen and to recognize what staff physicians are saying. “At Mayo Clinic, we have incorporated town halls, radio broadcasts, letters, and video interviews along with face-to-face meetings involving clinical divisions, work units, and small groups as formats or [by using] the CEO to reach the staff.” Assessing physician well-being and quality of work life using one or more of the many available tools has to be an ongoing “a barometer of organization health,” and not just a one-off, crisis management activity.
2. Harnessing the power of leadership
Studies have found that management behaviors and strategies of supervisors are key components of physician well-being. The bottom line is that physician supervisors must accept a share of responsibility for burnout in those they manage. Leaders can be chosen on the basis of their ability to listen, engage, develop, and lead, and but they can also be trained to improve. In addition, leaders should be regularly assessed by those whom they lead. Dr. Shanafelt and Dr. Noseworthy argue that a crucial element of successful leadership involves recognizing unique interests and talents of individual physicians whom they manage and facilitating professional development so that each staff member spends about 20% of work time engaged in activities that he or she finds most meaningful.
3. Developing targeted interventions
Just as all politics is local, the study suggests that many sources of burnout are local as well. For example, although a high clerical burden on physicians may be a universal driver of burnout, it manifests differently in each institutional setting. The key here is to dig into the specific structural driver at the unit or ward level, engage physicians in analysis and problem-solving, and implement a plan to address the problem.
Dr. Shanafelt noted, “We organize the drivers of engagement and burnout around seven dimensions: workload, efficiency, flexibility/control, community at work, organizational culture and values, work-life integration, and meaning in work. Each of these dimensions has organizational and individual components. Work units should begin by identifying which one or two dimensions are the biggest challenges for the group and then begin a stepwise process to address them.”
4. Cultivating community at work
Peer support, a long-standing source of strength among surgeons and other physicians, unintentionally has been eroded in many modern medical institutions. There is ample evidence that this loss of collegiality is tied to burnout. “These interactions have been an unintended casualty of increasing productivity expectations, documentation requirements, and clerical burden. [Many organizations have eliminated] formal spaces for physicians to interact (e.g., physicians’ lounge or dining room) without recognizing the important role that this dedicated space played in fostering interpersonal connections among physicians.” The Mayo Clinic and other institutions are reversing this trend by creating dedicated physician rooms for breaks, snacks, and a venue for peer interaction and camaraderie.
5. Rethinking rewards and incentives
Compensation is now commonly linked to productivity in many health care organizations, but this approach has some profound drawbacks: It can lead to physician burnout. Incentive structures based on patient satisfaction and quality metrics can have similar unintended consequences. All these incentive structures can combine to drive physicians to overwork. “Physicians may be particularly vulnerable to overwork due to high levels of education debt, their desire to ‘do everything for their patients,’ unhealthy role modeling by colleagues, and normalization of extreme work hours during the training process.” The investigators do not claim to have the ultimate answer to the problem of incentives that create unhealthy work patterns, but they argue that it is critical for leaders to recognize the potential unintended consequences of the productivity reward/incentive model and consider strategies to prevent overwork leading to burnout.
6. Aligning values and strengthening culture
The investigators also describe Mayo’s efforts to pursue self-appraisal of alignment of mission, values, and culture. They also describe the regular use of an all-staff survey, which has on occasion yielded candid feedback that, while not always flattering, has been the basis of a profound institutional rethink. The willingness of leadership to be receptive to hard truths from physicians is the foundation of institutional learning about burnout prevention and encourages engagement of the staff.
7. Promoting flexibility and work-life integration
Allowing employees greater flexibility in how and when they work is a management strategy that is gaining ground in many industries. Increasing part-time positions and expanding options for the work day have both been found to help prevent burnout and also help physicians recover from burnout. In addition, “institutions should also comprehensively examine the structure of their vacation benefits, coverage for life events (e.g., birth of a child, illness/death in family), approach to scheduling, and strategy for coverage of nights and weekends. Compensation practices that disincentivize using vacation time are shortsighted and should be eliminated.”
8. Providing resources to promote resilience and self-care
The solutions to burnout have been aimed at the individual and involve stress-reduction training and other personal management strategies. A metastudy of the interventions mentions psychoeducation, counseling, wellness management, interpersonal communication, and mindfulness meditation (J Nerv Ment Dis. 2014 May;202:353-9). But without concomitant structural reform, these individual solutions can backfire. “When individually focused offerings are not coupled with sincere efforts to address the system-based issues contributing to burnout, this approach is typically met with skepticism and resistance by physicians (‘They are implying I am the problem’). In this context, the response to well-intentioned ‘resilience training’ is frequently a cynical one (‘You only want to make me more resilient so you can further increase my workload’).”
9. Funding organizational research
Organizational science is a well-developed field of study. But cutting-edge management models such as the learning organization, participatory management, and collaborative action planning have been slow in coming to health care institutions. Dr. Shanafelt and Dr. Noseworthy argue that “vanguard institutions” such as the Mayo Clinic (which began its Program on Physician Well-Being in 2008), Stanford (Calif.) University, and other innovative institutions have the responsibility of developing evidence-based strategies to combat burnout that smaller institutions can implement.
Burnout among physicians is now on the radar of leadership in many health care institutions. Evidence on the cost and consequences is accumulating, and it becoming increasingly difficult to ignore what is happening to many physicians. Dr. Shanafelt projected an increasing need for operational solutions at the organizational level to address the problem. He said, “We need evidence to guide organizations to implement changes that truly make a difference, not well-intentioned but ineffective programs. Now that organizations recognize the strong business case, they are ready to invest resources to address this issue but they need to know it is money well spent and that there is an evidence-base to justify the investment.”
Most surgeons today are familiar with professional burnout – in their colleagues, in surgical trainees, and perhaps, in themselves. But the understanding of burnout is evolving. The discussion is moving away from blaming physicians for their poor coping skills toward identifying the structural and organizational roots of burnout.
Burnout is a syndrome cause by work-related stress that features emotional exhaustion, depersonalization, and a decreased sense of personal accomplishment. A recent study of nearly 7,000 physicians using the Maslach Burnout Inventory found that 54.4% of those surveyed reported at least one symptom of burnout (Mayo Clin Proc. 2015 Dec;90[12]:1600-19). Other studies have found similar rates of burnout in the surgical specialties such as orthopedic, oncologic, cardiothoracic, and plastic surgery (JAMA Surg. 2014 Sep;149:948-53; Ann Surg Oncol 2011 May;18:1229-35; Internat J Cardiol. 2015 Jan 20;179:7-72; Aesthet Surg J. 2016 Sep 27. E-pub ahead of print).
A new paradigm of burnout
The paradigm of burnout as a personal issue that can be managed by individual coping strategies is giving way to an understanding that the structural roots of burnout require the shared responsibility of individuals and their work organizations to solve the problem. A revised approach has emerged: Physician burnout as a symptom not of personal failure to cope, but of institutional failure to adapt to new circumstances in the health care milieu. The growing number of physicians employed in large group practices and medical centers has come with a whole array of management problems that are only beginning to be recognized, and burnout may be one of the most challenging.
Tait D. Shanafelt, MD, of the Mayo Foundation for Medical Education and Research, and John H. Noseworthy, MD, president and CEO of the Mayo Clinic, both in Rochester, Minn., have partnered to distill years of study and practice on the issue of burnout to a set of organizational strategies to tackle the problem and describe the Mayo Clinic experience. The study, “Executive Leadership and Physician Well-Being: Nine Organizational Strategies to Promote Engagement and Reduce Burnout” (Mayo Clin Proc. 2016 Nov 18. doi. org/10.1016/j.mayocp.2016.10.004) reverses the conventional “blame the victim” approach and identifies instead institutional responsibility to address burnout.
“Increasing evidence over the last 10 years demonstrating links to quality of care, productivity, and turnover have raised appreciation … by organizations that they have a substantial stake in this issue and that they control many of the factors that contribute to this problem,” said Dr. Shanafelt in an interview.
Unintended consequences of the individual solution
The focus on individual responsibility can have unintended consequences. A physician suffering from burnout can take action by leaving his or her job or cutting back. Staff turnover, a phenomenon closely tied to burnout, is costly and damaging to productivity and patient care (Physician Leadersh J. 2015 May-Jun;2[3]:22-5); Health Care Manage Rev. 2004;29[1]2-7). These personal strategies may help individuals cope but can end up harming the institution and the work life of other staff members. Physicians experiencing burnout in their own lives can trigger the same condition in their colleagues.
The Mayo paper by Dr. Shanafelt and Dr. Noseworthy states, “Mistakenly, most hospitals, medical centers, and practice groups operate under the framework that burnout and professional satisfaction are solely the responsibility of the individual physician. This frequently results in organizations pursuing a narrow list of ‘solutions’ that are unlikely to result in meaningful progress (e.g., stress management workshops and individual training in mindfulness/resilience). Such strategies neglect the organizational factors that are the primary drivers of physician burnout and are correctly viewed with skepticism by physicians as an insincere effort by the organization to address the problem.”
Organizational strategies to reduce burnout
Dr. Shanafelt and Dr. Noseworthy developed a list of nine organizational strategies that have been shown to reduce burnout among doctors. A critical piece of this approach is the accumulated evidence of the financial burden of burnout among physicians in health care institutions. The approach is based on an informed leadership that recognizes the costs of inaction, without which a systemic solution is unlikely to be achieved.
1. Naming the issue and assessing the problem
Acknowledgment of burnout as an institutional problem and meaningful measurement of physician well-being are the initial steps in tackling the problem. This requires a sincere commitment at the highest level of management to listen and to recognize what staff physicians are saying. “At Mayo Clinic, we have incorporated town halls, radio broadcasts, letters, and video interviews along with face-to-face meetings involving clinical divisions, work units, and small groups as formats or [by using] the CEO to reach the staff.” Assessing physician well-being and quality of work life using one or more of the many available tools has to be an ongoing “a barometer of organization health,” and not just a one-off, crisis management activity.
2. Harnessing the power of leadership
Studies have found that management behaviors and strategies of supervisors are key components of physician well-being. The bottom line is that physician supervisors must accept a share of responsibility for burnout in those they manage. Leaders can be chosen on the basis of their ability to listen, engage, develop, and lead, and but they can also be trained to improve. In addition, leaders should be regularly assessed by those whom they lead. Dr. Shanafelt and Dr. Noseworthy argue that a crucial element of successful leadership involves recognizing unique interests and talents of individual physicians whom they manage and facilitating professional development so that each staff member spends about 20% of work time engaged in activities that he or she finds most meaningful.
3. Developing targeted interventions
Just as all politics is local, the study suggests that many sources of burnout are local as well. For example, although a high clerical burden on physicians may be a universal driver of burnout, it manifests differently in each institutional setting. The key here is to dig into the specific structural driver at the unit or ward level, engage physicians in analysis and problem-solving, and implement a plan to address the problem.
Dr. Shanafelt noted, “We organize the drivers of engagement and burnout around seven dimensions: workload, efficiency, flexibility/control, community at work, organizational culture and values, work-life integration, and meaning in work. Each of these dimensions has organizational and individual components. Work units should begin by identifying which one or two dimensions are the biggest challenges for the group and then begin a stepwise process to address them.”
4. Cultivating community at work
Peer support, a long-standing source of strength among surgeons and other physicians, unintentionally has been eroded in many modern medical institutions. There is ample evidence that this loss of collegiality is tied to burnout. “These interactions have been an unintended casualty of increasing productivity expectations, documentation requirements, and clerical burden. [Many organizations have eliminated] formal spaces for physicians to interact (e.g., physicians’ lounge or dining room) without recognizing the important role that this dedicated space played in fostering interpersonal connections among physicians.” The Mayo Clinic and other institutions are reversing this trend by creating dedicated physician rooms for breaks, snacks, and a venue for peer interaction and camaraderie.
5. Rethinking rewards and incentives
Compensation is now commonly linked to productivity in many health care organizations, but this approach has some profound drawbacks: It can lead to physician burnout. Incentive structures based on patient satisfaction and quality metrics can have similar unintended consequences. All these incentive structures can combine to drive physicians to overwork. “Physicians may be particularly vulnerable to overwork due to high levels of education debt, their desire to ‘do everything for their patients,’ unhealthy role modeling by colleagues, and normalization of extreme work hours during the training process.” The investigators do not claim to have the ultimate answer to the problem of incentives that create unhealthy work patterns, but they argue that it is critical for leaders to recognize the potential unintended consequences of the productivity reward/incentive model and consider strategies to prevent overwork leading to burnout.
6. Aligning values and strengthening culture
The investigators also describe Mayo’s efforts to pursue self-appraisal of alignment of mission, values, and culture. They also describe the regular use of an all-staff survey, which has on occasion yielded candid feedback that, while not always flattering, has been the basis of a profound institutional rethink. The willingness of leadership to be receptive to hard truths from physicians is the foundation of institutional learning about burnout prevention and encourages engagement of the staff.
7. Promoting flexibility and work-life integration
Allowing employees greater flexibility in how and when they work is a management strategy that is gaining ground in many industries. Increasing part-time positions and expanding options for the work day have both been found to help prevent burnout and also help physicians recover from burnout. In addition, “institutions should also comprehensively examine the structure of their vacation benefits, coverage for life events (e.g., birth of a child, illness/death in family), approach to scheduling, and strategy for coverage of nights and weekends. Compensation practices that disincentivize using vacation time are shortsighted and should be eliminated.”
8. Providing resources to promote resilience and self-care
The solutions to burnout have been aimed at the individual and involve stress-reduction training and other personal management strategies. A metastudy of the interventions mentions psychoeducation, counseling, wellness management, interpersonal communication, and mindfulness meditation (J Nerv Ment Dis. 2014 May;202:353-9). But without concomitant structural reform, these individual solutions can backfire. “When individually focused offerings are not coupled with sincere efforts to address the system-based issues contributing to burnout, this approach is typically met with skepticism and resistance by physicians (‘They are implying I am the problem’). In this context, the response to well-intentioned ‘resilience training’ is frequently a cynical one (‘You only want to make me more resilient so you can further increase my workload’).”
9. Funding organizational research
Organizational science is a well-developed field of study. But cutting-edge management models such as the learning organization, participatory management, and collaborative action planning have been slow in coming to health care institutions. Dr. Shanafelt and Dr. Noseworthy argue that “vanguard institutions” such as the Mayo Clinic (which began its Program on Physician Well-Being in 2008), Stanford (Calif.) University, and other innovative institutions have the responsibility of developing evidence-based strategies to combat burnout that smaller institutions can implement.
Burnout among physicians is now on the radar of leadership in many health care institutions. Evidence on the cost and consequences is accumulating, and it becoming increasingly difficult to ignore what is happening to many physicians. Dr. Shanafelt projected an increasing need for operational solutions at the organizational level to address the problem. He said, “We need evidence to guide organizations to implement changes that truly make a difference, not well-intentioned but ineffective programs. Now that organizations recognize the strong business case, they are ready to invest resources to address this issue but they need to know it is money well spent and that there is an evidence-base to justify the investment.”
Most surgeons today are familiar with professional burnout – in their colleagues, in surgical trainees, and perhaps, in themselves. But the understanding of burnout is evolving. The discussion is moving away from blaming physicians for their poor coping skills toward identifying the structural and organizational roots of burnout.
Burnout is a syndrome cause by work-related stress that features emotional exhaustion, depersonalization, and a decreased sense of personal accomplishment. A recent study of nearly 7,000 physicians using the Maslach Burnout Inventory found that 54.4% of those surveyed reported at least one symptom of burnout (Mayo Clin Proc. 2015 Dec;90[12]:1600-19). Other studies have found similar rates of burnout in the surgical specialties such as orthopedic, oncologic, cardiothoracic, and plastic surgery (JAMA Surg. 2014 Sep;149:948-53; Ann Surg Oncol 2011 May;18:1229-35; Internat J Cardiol. 2015 Jan 20;179:7-72; Aesthet Surg J. 2016 Sep 27. E-pub ahead of print).
A new paradigm of burnout
The paradigm of burnout as a personal issue that can be managed by individual coping strategies is giving way to an understanding that the structural roots of burnout require the shared responsibility of individuals and their work organizations to solve the problem. A revised approach has emerged: Physician burnout as a symptom not of personal failure to cope, but of institutional failure to adapt to new circumstances in the health care milieu. The growing number of physicians employed in large group practices and medical centers has come with a whole array of management problems that are only beginning to be recognized, and burnout may be one of the most challenging.
Tait D. Shanafelt, MD, of the Mayo Foundation for Medical Education and Research, and John H. Noseworthy, MD, president and CEO of the Mayo Clinic, both in Rochester, Minn., have partnered to distill years of study and practice on the issue of burnout to a set of organizational strategies to tackle the problem and describe the Mayo Clinic experience. The study, “Executive Leadership and Physician Well-Being: Nine Organizational Strategies to Promote Engagement and Reduce Burnout” (Mayo Clin Proc. 2016 Nov 18. doi. org/10.1016/j.mayocp.2016.10.004) reverses the conventional “blame the victim” approach and identifies instead institutional responsibility to address burnout.
“Increasing evidence over the last 10 years demonstrating links to quality of care, productivity, and turnover have raised appreciation … by organizations that they have a substantial stake in this issue and that they control many of the factors that contribute to this problem,” said Dr. Shanafelt in an interview.
Unintended consequences of the individual solution
The focus on individual responsibility can have unintended consequences. A physician suffering from burnout can take action by leaving his or her job or cutting back. Staff turnover, a phenomenon closely tied to burnout, is costly and damaging to productivity and patient care (Physician Leadersh J. 2015 May-Jun;2[3]:22-5); Health Care Manage Rev. 2004;29[1]2-7). These personal strategies may help individuals cope but can end up harming the institution and the work life of other staff members. Physicians experiencing burnout in their own lives can trigger the same condition in their colleagues.
The Mayo paper by Dr. Shanafelt and Dr. Noseworthy states, “Mistakenly, most hospitals, medical centers, and practice groups operate under the framework that burnout and professional satisfaction are solely the responsibility of the individual physician. This frequently results in organizations pursuing a narrow list of ‘solutions’ that are unlikely to result in meaningful progress (e.g., stress management workshops and individual training in mindfulness/resilience). Such strategies neglect the organizational factors that are the primary drivers of physician burnout and are correctly viewed with skepticism by physicians as an insincere effort by the organization to address the problem.”
Organizational strategies to reduce burnout
Dr. Shanafelt and Dr. Noseworthy developed a list of nine organizational strategies that have been shown to reduce burnout among doctors. A critical piece of this approach is the accumulated evidence of the financial burden of burnout among physicians in health care institutions. The approach is based on an informed leadership that recognizes the costs of inaction, without which a systemic solution is unlikely to be achieved.
1. Naming the issue and assessing the problem
Acknowledgment of burnout as an institutional problem and meaningful measurement of physician well-being are the initial steps in tackling the problem. This requires a sincere commitment at the highest level of management to listen and to recognize what staff physicians are saying. “At Mayo Clinic, we have incorporated town halls, radio broadcasts, letters, and video interviews along with face-to-face meetings involving clinical divisions, work units, and small groups as formats or [by using] the CEO to reach the staff.” Assessing physician well-being and quality of work life using one or more of the many available tools has to be an ongoing “a barometer of organization health,” and not just a one-off, crisis management activity.
2. Harnessing the power of leadership
Studies have found that management behaviors and strategies of supervisors are key components of physician well-being. The bottom line is that physician supervisors must accept a share of responsibility for burnout in those they manage. Leaders can be chosen on the basis of their ability to listen, engage, develop, and lead, and but they can also be trained to improve. In addition, leaders should be regularly assessed by those whom they lead. Dr. Shanafelt and Dr. Noseworthy argue that a crucial element of successful leadership involves recognizing unique interests and talents of individual physicians whom they manage and facilitating professional development so that each staff member spends about 20% of work time engaged in activities that he or she finds most meaningful.
3. Developing targeted interventions
Just as all politics is local, the study suggests that many sources of burnout are local as well. For example, although a high clerical burden on physicians may be a universal driver of burnout, it manifests differently in each institutional setting. The key here is to dig into the specific structural driver at the unit or ward level, engage physicians in analysis and problem-solving, and implement a plan to address the problem.
Dr. Shanafelt noted, “We organize the drivers of engagement and burnout around seven dimensions: workload, efficiency, flexibility/control, community at work, organizational culture and values, work-life integration, and meaning in work. Each of these dimensions has organizational and individual components. Work units should begin by identifying which one or two dimensions are the biggest challenges for the group and then begin a stepwise process to address them.”
4. Cultivating community at work
Peer support, a long-standing source of strength among surgeons and other physicians, unintentionally has been eroded in many modern medical institutions. There is ample evidence that this loss of collegiality is tied to burnout. “These interactions have been an unintended casualty of increasing productivity expectations, documentation requirements, and clerical burden. [Many organizations have eliminated] formal spaces for physicians to interact (e.g., physicians’ lounge or dining room) without recognizing the important role that this dedicated space played in fostering interpersonal connections among physicians.” The Mayo Clinic and other institutions are reversing this trend by creating dedicated physician rooms for breaks, snacks, and a venue for peer interaction and camaraderie.
5. Rethinking rewards and incentives
Compensation is now commonly linked to productivity in many health care organizations, but this approach has some profound drawbacks: It can lead to physician burnout. Incentive structures based on patient satisfaction and quality metrics can have similar unintended consequences. All these incentive structures can combine to drive physicians to overwork. “Physicians may be particularly vulnerable to overwork due to high levels of education debt, their desire to ‘do everything for their patients,’ unhealthy role modeling by colleagues, and normalization of extreme work hours during the training process.” The investigators do not claim to have the ultimate answer to the problem of incentives that create unhealthy work patterns, but they argue that it is critical for leaders to recognize the potential unintended consequences of the productivity reward/incentive model and consider strategies to prevent overwork leading to burnout.
6. Aligning values and strengthening culture
The investigators also describe Mayo’s efforts to pursue self-appraisal of alignment of mission, values, and culture. They also describe the regular use of an all-staff survey, which has on occasion yielded candid feedback that, while not always flattering, has been the basis of a profound institutional rethink. The willingness of leadership to be receptive to hard truths from physicians is the foundation of institutional learning about burnout prevention and encourages engagement of the staff.
7. Promoting flexibility and work-life integration
Allowing employees greater flexibility in how and when they work is a management strategy that is gaining ground in many industries. Increasing part-time positions and expanding options for the work day have both been found to help prevent burnout and also help physicians recover from burnout. In addition, “institutions should also comprehensively examine the structure of their vacation benefits, coverage for life events (e.g., birth of a child, illness/death in family), approach to scheduling, and strategy for coverage of nights and weekends. Compensation practices that disincentivize using vacation time are shortsighted and should be eliminated.”
8. Providing resources to promote resilience and self-care
The solutions to burnout have been aimed at the individual and involve stress-reduction training and other personal management strategies. A metastudy of the interventions mentions psychoeducation, counseling, wellness management, interpersonal communication, and mindfulness meditation (J Nerv Ment Dis. 2014 May;202:353-9). But without concomitant structural reform, these individual solutions can backfire. “When individually focused offerings are not coupled with sincere efforts to address the system-based issues contributing to burnout, this approach is typically met with skepticism and resistance by physicians (‘They are implying I am the problem’). In this context, the response to well-intentioned ‘resilience training’ is frequently a cynical one (‘You only want to make me more resilient so you can further increase my workload’).”
9. Funding organizational research
Organizational science is a well-developed field of study. But cutting-edge management models such as the learning organization, participatory management, and collaborative action planning have been slow in coming to health care institutions. Dr. Shanafelt and Dr. Noseworthy argue that “vanguard institutions” such as the Mayo Clinic (which began its Program on Physician Well-Being in 2008), Stanford (Calif.) University, and other innovative institutions have the responsibility of developing evidence-based strategies to combat burnout that smaller institutions can implement.
Burnout among physicians is now on the radar of leadership in many health care institutions. Evidence on the cost and consequences is accumulating, and it becoming increasingly difficult to ignore what is happening to many physicians. Dr. Shanafelt projected an increasing need for operational solutions at the organizational level to address the problem. He said, “We need evidence to guide organizations to implement changes that truly make a difference, not well-intentioned but ineffective programs. Now that organizations recognize the strong business case, they are ready to invest resources to address this issue but they need to know it is money well spent and that there is an evidence-base to justify the investment.”
How to limit radiation in endovascular procedures
CHICAGO – Applying the key principles for limiting radiation exposure for vascular surgeons and staff – not to mention patients – during endovascular procedures involves a thorough understanding of dose metrics as well as risk factors for high-dose interventions, according to recent findings reported at a symposium on vascular surgery sponsored by Northwestern University.
Melissa Kirkwood, MD, of the University of Texas Southwestern Medical Center in Dallas, reported on her institution’s experience with limiting radiation exposure in typically high-dose cases. “What we found was that even though we had a substantial amount of dose in these cases – they included a significant proportion of 5- to 10-Gy and greater than 10-Gy cases – there were still no skin injuries detected in these patients,” she said, referencing retrospective and prospective analyses (J Vasc Surg. 2014;60:742-8; J Vasc Surg. 2015;61:902-6).
Vigilance regarding these principles for vascular surgeons is paramount, Dr. Kirkwood said, noting that the National Council on Radiation Protection and Measurements threshold for substantial radiation dose is 5 Gy or greater. “When you’re doing complex endovascular work, your doses can get that high,” she said.
As a means of measuring dose, Dr. Kirkwood called fluoroscopy time a “universally poor indicator” because in current practice vascular surgeons use digital acquisition mode in addition to fluoroscopy. “The digital acquisitions generate 10-100 times more dose than fluoroscopy, so if you’re only looking at fluoroscopy time, your potentially missing the majority of the dose for that case,” she said.
More applicable dose measures, she said, are kerma area product that measures total radiation beam output from the x-ray tube, which she called “a better reflection of operator exposure,” and reference air kerma (RAK), a measure of the dose at a reference point 15 cm along the beam axis toward the focal spot from the isocenter, which she said is the best approximation for patient peak skin dose exposure. However, the latter does not account for angle of the x-ray tube or patient position, which can vary based on the type of procedure or the patient’s size.
Dr. Kirkwood’s work at UTSW also determined that operator exposure during an endovascular procedure depends on where they stand. “Doubling the distance from the source can decrease the radiation level by a factor of four,” she said. For femoral access in the right groin, the operator is at greatest risk for exposure followed by the assistant when the assistant is standing to the right of the operator. The left brachial access site carries an even higher exposure for the operator, she said.
The table-mounted lead skirt plays a key role in limiting operator exposure, Dr. Kirkwood said. “It can be cumbersome, but it is very important in lowering your lower-body dose,” she said, because it will block radiation scatter coming off the bottom of the table.
At UTSW, the endovascular operators had a tutorial with the staff medical physicist on best practices to limit radiation exposure. “What we found was that we were significantly able to decrease the dose across all cases by simply going over a few principles,” she said.
Among those principles: “Always be aware when you’re on the fluoroscopy pedal and always use the lowest fluoroscopy mode possible,” she said. However, she noted that in difficult-to-visualize cases, a short-duration boost in fluoroscopy level might reduce overall fluoroscopy time and hence limit exposure. To limit digital acquisition mode, the use of fluoroscopic looping can allow for review of images during the procedure with a fraction of the dose that would be needed for a digital acquisition run.
Limiting magnification and using collimation can be complementary, Dr. Kirkwood said. “If you really have to magnify to see the area of interest, make sure you have tight collimation to try to decrease the scatter to you and your colleagues in the OR,” she said.
Dr. Kirkwood noted that raising the angio table as high as is comfortable and decreasing the distance between the source and image detector can limit patient exposure. Operators should avoid steep angulations of the x-ray tube, she said, but when angulations are necessary, operators should stand on the opposite side of the x-ray tube. “The best operating practice if you know you’re going to have a high-dose case with a lot of gantry angulation would be to tightly collimate to the area of interest and minimize the magnification,” she said.
Though not necessarily a principle, keeping up with software advances for imaging devices can also prove valuable for limiting radiation exposure, Dr. Kirkwood said. “It’s important to know about them because if you are purchasing new equipment, they are not necessarily included if you’re institution is looking to hold down costs,” she said.
Dr. Kirkwood had no relevant financial disclosures.
CHICAGO – Applying the key principles for limiting radiation exposure for vascular surgeons and staff – not to mention patients – during endovascular procedures involves a thorough understanding of dose metrics as well as risk factors for high-dose interventions, according to recent findings reported at a symposium on vascular surgery sponsored by Northwestern University.
Melissa Kirkwood, MD, of the University of Texas Southwestern Medical Center in Dallas, reported on her institution’s experience with limiting radiation exposure in typically high-dose cases. “What we found was that even though we had a substantial amount of dose in these cases – they included a significant proportion of 5- to 10-Gy and greater than 10-Gy cases – there were still no skin injuries detected in these patients,” she said, referencing retrospective and prospective analyses (J Vasc Surg. 2014;60:742-8; J Vasc Surg. 2015;61:902-6).
Vigilance regarding these principles for vascular surgeons is paramount, Dr. Kirkwood said, noting that the National Council on Radiation Protection and Measurements threshold for substantial radiation dose is 5 Gy or greater. “When you’re doing complex endovascular work, your doses can get that high,” she said.
As a means of measuring dose, Dr. Kirkwood called fluoroscopy time a “universally poor indicator” because in current practice vascular surgeons use digital acquisition mode in addition to fluoroscopy. “The digital acquisitions generate 10-100 times more dose than fluoroscopy, so if you’re only looking at fluoroscopy time, your potentially missing the majority of the dose for that case,” she said.
More applicable dose measures, she said, are kerma area product that measures total radiation beam output from the x-ray tube, which she called “a better reflection of operator exposure,” and reference air kerma (RAK), a measure of the dose at a reference point 15 cm along the beam axis toward the focal spot from the isocenter, which she said is the best approximation for patient peak skin dose exposure. However, the latter does not account for angle of the x-ray tube or patient position, which can vary based on the type of procedure or the patient’s size.
Dr. Kirkwood’s work at UTSW also determined that operator exposure during an endovascular procedure depends on where they stand. “Doubling the distance from the source can decrease the radiation level by a factor of four,” she said. For femoral access in the right groin, the operator is at greatest risk for exposure followed by the assistant when the assistant is standing to the right of the operator. The left brachial access site carries an even higher exposure for the operator, she said.
The table-mounted lead skirt plays a key role in limiting operator exposure, Dr. Kirkwood said. “It can be cumbersome, but it is very important in lowering your lower-body dose,” she said, because it will block radiation scatter coming off the bottom of the table.
At UTSW, the endovascular operators had a tutorial with the staff medical physicist on best practices to limit radiation exposure. “What we found was that we were significantly able to decrease the dose across all cases by simply going over a few principles,” she said.
Among those principles: “Always be aware when you’re on the fluoroscopy pedal and always use the lowest fluoroscopy mode possible,” she said. However, she noted that in difficult-to-visualize cases, a short-duration boost in fluoroscopy level might reduce overall fluoroscopy time and hence limit exposure. To limit digital acquisition mode, the use of fluoroscopic looping can allow for review of images during the procedure with a fraction of the dose that would be needed for a digital acquisition run.
Limiting magnification and using collimation can be complementary, Dr. Kirkwood said. “If you really have to magnify to see the area of interest, make sure you have tight collimation to try to decrease the scatter to you and your colleagues in the OR,” she said.
Dr. Kirkwood noted that raising the angio table as high as is comfortable and decreasing the distance between the source and image detector can limit patient exposure. Operators should avoid steep angulations of the x-ray tube, she said, but when angulations are necessary, operators should stand on the opposite side of the x-ray tube. “The best operating practice if you know you’re going to have a high-dose case with a lot of gantry angulation would be to tightly collimate to the area of interest and minimize the magnification,” she said.
Though not necessarily a principle, keeping up with software advances for imaging devices can also prove valuable for limiting radiation exposure, Dr. Kirkwood said. “It’s important to know about them because if you are purchasing new equipment, they are not necessarily included if you’re institution is looking to hold down costs,” she said.
Dr. Kirkwood had no relevant financial disclosures.
CHICAGO – Applying the key principles for limiting radiation exposure for vascular surgeons and staff – not to mention patients – during endovascular procedures involves a thorough understanding of dose metrics as well as risk factors for high-dose interventions, according to recent findings reported at a symposium on vascular surgery sponsored by Northwestern University.
Melissa Kirkwood, MD, of the University of Texas Southwestern Medical Center in Dallas, reported on her institution’s experience with limiting radiation exposure in typically high-dose cases. “What we found was that even though we had a substantial amount of dose in these cases – they included a significant proportion of 5- to 10-Gy and greater than 10-Gy cases – there were still no skin injuries detected in these patients,” she said, referencing retrospective and prospective analyses (J Vasc Surg. 2014;60:742-8; J Vasc Surg. 2015;61:902-6).
Vigilance regarding these principles for vascular surgeons is paramount, Dr. Kirkwood said, noting that the National Council on Radiation Protection and Measurements threshold for substantial radiation dose is 5 Gy or greater. “When you’re doing complex endovascular work, your doses can get that high,” she said.
As a means of measuring dose, Dr. Kirkwood called fluoroscopy time a “universally poor indicator” because in current practice vascular surgeons use digital acquisition mode in addition to fluoroscopy. “The digital acquisitions generate 10-100 times more dose than fluoroscopy, so if you’re only looking at fluoroscopy time, your potentially missing the majority of the dose for that case,” she said.
More applicable dose measures, she said, are kerma area product that measures total radiation beam output from the x-ray tube, which she called “a better reflection of operator exposure,” and reference air kerma (RAK), a measure of the dose at a reference point 15 cm along the beam axis toward the focal spot from the isocenter, which she said is the best approximation for patient peak skin dose exposure. However, the latter does not account for angle of the x-ray tube or patient position, which can vary based on the type of procedure or the patient’s size.
Dr. Kirkwood’s work at UTSW also determined that operator exposure during an endovascular procedure depends on where they stand. “Doubling the distance from the source can decrease the radiation level by a factor of four,” she said. For femoral access in the right groin, the operator is at greatest risk for exposure followed by the assistant when the assistant is standing to the right of the operator. The left brachial access site carries an even higher exposure for the operator, she said.
The table-mounted lead skirt plays a key role in limiting operator exposure, Dr. Kirkwood said. “It can be cumbersome, but it is very important in lowering your lower-body dose,” she said, because it will block radiation scatter coming off the bottom of the table.
At UTSW, the endovascular operators had a tutorial with the staff medical physicist on best practices to limit radiation exposure. “What we found was that we were significantly able to decrease the dose across all cases by simply going over a few principles,” she said.
Among those principles: “Always be aware when you’re on the fluoroscopy pedal and always use the lowest fluoroscopy mode possible,” she said. However, she noted that in difficult-to-visualize cases, a short-duration boost in fluoroscopy level might reduce overall fluoroscopy time and hence limit exposure. To limit digital acquisition mode, the use of fluoroscopic looping can allow for review of images during the procedure with a fraction of the dose that would be needed for a digital acquisition run.
Limiting magnification and using collimation can be complementary, Dr. Kirkwood said. “If you really have to magnify to see the area of interest, make sure you have tight collimation to try to decrease the scatter to you and your colleagues in the OR,” she said.
Dr. Kirkwood noted that raising the angio table as high as is comfortable and decreasing the distance between the source and image detector can limit patient exposure. Operators should avoid steep angulations of the x-ray tube, she said, but when angulations are necessary, operators should stand on the opposite side of the x-ray tube. “The best operating practice if you know you’re going to have a high-dose case with a lot of gantry angulation would be to tightly collimate to the area of interest and minimize the magnification,” she said.
Though not necessarily a principle, keeping up with software advances for imaging devices can also prove valuable for limiting radiation exposure, Dr. Kirkwood said. “It’s important to know about them because if you are purchasing new equipment, they are not necessarily included if you’re institution is looking to hold down costs,” she said.
Dr. Kirkwood had no relevant financial disclosures.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Key clinical point: Vascular surgeons can lower their radiation exposure during endovascular procedures by employing key principles like appropriate shielding.
Major finding: Familiarity with dose terminology and metrics, possible radiation-induced injuries, and techniques to lower radiation dosing are keys to limiting radiation exposure.
Data source: Review of literature, including National Council on Radiation and Protection guidelines and National Cancer Institute grades of skin toxicity for radiation dermatitis.
Disclosures: Dr. Kirkwood had no financial relationships to disclose.
SOFA score may be best to identify sepsis in the ICU
Among critically ill patients admitted to the ICU with a suspected infection, defining sepsis by an increase of 2 or more points in the Sequential Organ Failure Assessment score yielded greater prognostic accuracy for in-hospital mortality, compared with the quick SOFA and the systemic inflammatory response syndrome criteria, results from a large analysis showed.
“Accurate diagnostic criteria and consensus definitions have an important role in adult intensive care medicine, providing tools for research, benchmarking, performance monitoring, and accreditation,” researchers from The Australian and New Zealand Intensive Care Society Centre for Outcomes and Resource Evaluation reported in the Jan. 17, 2017 edition of JAMA. “Seymour and colleagues published data concerning the validity of a 2 or more–point change in the Sequential [Sepsis-related] Organ Failure Assessment (SOFA) score as a means of identifying sepsis among patients who are critically ill with suspected infection, assuming a SOFA of 0 for patients not known to have preexisting organ dysfunction (JAMA. 2016; 315[8]:762-74). In addition, the concept of the quick SOFA (qSOFA) score was introduced as a possible predictive tool among encounters with suspected infection outside the intensive care unit (ICU). These data were drawn from North American cohorts and a single German cohort and have not been validated externally.”
The mean age of the patients was 63 years, 45% were women, and the most common diagnosis was bacterial pneumonia (18%). Nearly 19% of patients died in the hospital and 56% died or experienced an ICU length of stay of at least 3 days or more. The researchers found that the SOFA score increased by 2 or more points in 90% of patients, while 87% manifested 2 or more SIRS criteria, and 54% had a qSOFA score of 2 or more points. In addition, discrimination of in-hospital mortality was significantly higher using SOFA (area under the receiving operating characteristic curve [AUROC], 0.753), compared with both SIRS criteria (AUROC, 0.589) and qSOFA (0.607); the between-group difference reached a P value of less than .001. Similar results were seen for the composite outcome of in-hospital mortality or an ICU length of stay of 3 days or more, which was higher using SOFA (AUROC, 0.736), compared with both SIRS criteria (AUROC, 0.609) and qSOFA (0.606); the between-group difference also reached a P value of less than .001.
The researchers acknowledged certain limitations of the study, including the fact that SOFA, SIRS criteria, and qSOFA could only be studied for the first 24 hours in the ICU. “Biochemical and physiological values could have come from any time within the first 24 hours of ICU admission and, as a result, could not be more accurately linked to the timing of the diagnosis of infection,” they wrote. “The SOFA score used should be considered a modification of the original because the cardiovascular component was estimated without knowledge of inotrope or vasopressor dose. The incidence of nosocomial infections and of infections in patients admitted with another diagnosis were unknown.”
Three of the seven study authors disclosed that they receive salary support from Monash University in Melbourne. The remainder reported having no financial disclosures.
It is neither surprising that qSOFA did not perform as well as the SOFA score in the ICU, given that this finding was already reported by Seymour et al. in their initial work, nor is it critically important because qSOFA is more likely to be useful outside of the ICU setting.
Thus, the findings ... support the results reported by Seymour et al. that qSOFA is potentially helpful in settings outside the ICU in rapidly identifying patients with suspected infection who have, or will likely develop, sepsis (JAMA. 2016;315[8]:762-74). However, qSOFA still warrants further testing, particularly in lower- and middle-income settings where context (for example, timing of presentation to the hospital among patients with a suspected infection) might vary considerably and such contextual factors might affect predictive validity. In addition, prospective studies may evaluate the utility of qSOFA when used longitudinally, with repeated measurements throughout the hospital stay. Arguably, the highest-quality evidence for validation of any tool to support clinical decision making would come from an analysis to establish whether decisions made with the support of the tool lead to better patient outcomes than those made by clinical judgment alone.
Ultimately, the utility of qSOFA will likely become surpassed if and when highly accurate, rapid diagnostic tests for sepsis emerge. For now, however, outside the ICU in the high-income settings where it has been tested, qSOFA appears a simple, rapid, inexpensive, and valid way to identify – among patients with suspected infection – those at a higher risk of having or developing sepsis.
Francois Lamontagne, MD, David A. Harrison, PhD, and Kathryn M. Rowan, PhD, are affiliated with the Intensive Care National Audit & Research Centre, London. Dr. Lamontagne reported serving as investigator for a study funded by GlaxoSmithKline and E-Motion. These comments are extracted from an editorial that appears in the Jan. 17, 2017 edition of JAMA (317[3]:267-8).
It is neither surprising that qSOFA did not perform as well as the SOFA score in the ICU, given that this finding was already reported by Seymour et al. in their initial work, nor is it critically important because qSOFA is more likely to be useful outside of the ICU setting.
Thus, the findings ... support the results reported by Seymour et al. that qSOFA is potentially helpful in settings outside the ICU in rapidly identifying patients with suspected infection who have, or will likely develop, sepsis (JAMA. 2016;315[8]:762-74). However, qSOFA still warrants further testing, particularly in lower- and middle-income settings where context (for example, timing of presentation to the hospital among patients with a suspected infection) might vary considerably and such contextual factors might affect predictive validity. In addition, prospective studies may evaluate the utility of qSOFA when used longitudinally, with repeated measurements throughout the hospital stay. Arguably, the highest-quality evidence for validation of any tool to support clinical decision making would come from an analysis to establish whether decisions made with the support of the tool lead to better patient outcomes than those made by clinical judgment alone.
Ultimately, the utility of qSOFA will likely become surpassed if and when highly accurate, rapid diagnostic tests for sepsis emerge. For now, however, outside the ICU in the high-income settings where it has been tested, qSOFA appears a simple, rapid, inexpensive, and valid way to identify – among patients with suspected infection – those at a higher risk of having or developing sepsis.
Francois Lamontagne, MD, David A. Harrison, PhD, and Kathryn M. Rowan, PhD, are affiliated with the Intensive Care National Audit & Research Centre, London. Dr. Lamontagne reported serving as investigator for a study funded by GlaxoSmithKline and E-Motion. These comments are extracted from an editorial that appears in the Jan. 17, 2017 edition of JAMA (317[3]:267-8).
It is neither surprising that qSOFA did not perform as well as the SOFA score in the ICU, given that this finding was already reported by Seymour et al. in their initial work, nor is it critically important because qSOFA is more likely to be useful outside of the ICU setting.
Thus, the findings ... support the results reported by Seymour et al. that qSOFA is potentially helpful in settings outside the ICU in rapidly identifying patients with suspected infection who have, or will likely develop, sepsis (JAMA. 2016;315[8]:762-74). However, qSOFA still warrants further testing, particularly in lower- and middle-income settings where context (for example, timing of presentation to the hospital among patients with a suspected infection) might vary considerably and such contextual factors might affect predictive validity. In addition, prospective studies may evaluate the utility of qSOFA when used longitudinally, with repeated measurements throughout the hospital stay. Arguably, the highest-quality evidence for validation of any tool to support clinical decision making would come from an analysis to establish whether decisions made with the support of the tool lead to better patient outcomes than those made by clinical judgment alone.
Ultimately, the utility of qSOFA will likely become surpassed if and when highly accurate, rapid diagnostic tests for sepsis emerge. For now, however, outside the ICU in the high-income settings where it has been tested, qSOFA appears a simple, rapid, inexpensive, and valid way to identify – among patients with suspected infection – those at a higher risk of having or developing sepsis.
Francois Lamontagne, MD, David A. Harrison, PhD, and Kathryn M. Rowan, PhD, are affiliated with the Intensive Care National Audit & Research Centre, London. Dr. Lamontagne reported serving as investigator for a study funded by GlaxoSmithKline and E-Motion. These comments are extracted from an editorial that appears in the Jan. 17, 2017 edition of JAMA (317[3]:267-8).
Among critically ill patients admitted to the ICU with a suspected infection, defining sepsis by an increase of 2 or more points in the Sequential Organ Failure Assessment score yielded greater prognostic accuracy for in-hospital mortality, compared with the quick SOFA and the systemic inflammatory response syndrome criteria, results from a large analysis showed.
“Accurate diagnostic criteria and consensus definitions have an important role in adult intensive care medicine, providing tools for research, benchmarking, performance monitoring, and accreditation,” researchers from The Australian and New Zealand Intensive Care Society Centre for Outcomes and Resource Evaluation reported in the Jan. 17, 2017 edition of JAMA. “Seymour and colleagues published data concerning the validity of a 2 or more–point change in the Sequential [Sepsis-related] Organ Failure Assessment (SOFA) score as a means of identifying sepsis among patients who are critically ill with suspected infection, assuming a SOFA of 0 for patients not known to have preexisting organ dysfunction (JAMA. 2016; 315[8]:762-74). In addition, the concept of the quick SOFA (qSOFA) score was introduced as a possible predictive tool among encounters with suspected infection outside the intensive care unit (ICU). These data were drawn from North American cohorts and a single German cohort and have not been validated externally.”
The mean age of the patients was 63 years, 45% were women, and the most common diagnosis was bacterial pneumonia (18%). Nearly 19% of patients died in the hospital and 56% died or experienced an ICU length of stay of at least 3 days or more. The researchers found that the SOFA score increased by 2 or more points in 90% of patients, while 87% manifested 2 or more SIRS criteria, and 54% had a qSOFA score of 2 or more points. In addition, discrimination of in-hospital mortality was significantly higher using SOFA (area under the receiving operating characteristic curve [AUROC], 0.753), compared with both SIRS criteria (AUROC, 0.589) and qSOFA (0.607); the between-group difference reached a P value of less than .001. Similar results were seen for the composite outcome of in-hospital mortality or an ICU length of stay of 3 days or more, which was higher using SOFA (AUROC, 0.736), compared with both SIRS criteria (AUROC, 0.609) and qSOFA (0.606); the between-group difference also reached a P value of less than .001.
The researchers acknowledged certain limitations of the study, including the fact that SOFA, SIRS criteria, and qSOFA could only be studied for the first 24 hours in the ICU. “Biochemical and physiological values could have come from any time within the first 24 hours of ICU admission and, as a result, could not be more accurately linked to the timing of the diagnosis of infection,” they wrote. “The SOFA score used should be considered a modification of the original because the cardiovascular component was estimated without knowledge of inotrope or vasopressor dose. The incidence of nosocomial infections and of infections in patients admitted with another diagnosis were unknown.”
Three of the seven study authors disclosed that they receive salary support from Monash University in Melbourne. The remainder reported having no financial disclosures.
Among critically ill patients admitted to the ICU with a suspected infection, defining sepsis by an increase of 2 or more points in the Sequential Organ Failure Assessment score yielded greater prognostic accuracy for in-hospital mortality, compared with the quick SOFA and the systemic inflammatory response syndrome criteria, results from a large analysis showed.
“Accurate diagnostic criteria and consensus definitions have an important role in adult intensive care medicine, providing tools for research, benchmarking, performance monitoring, and accreditation,” researchers from The Australian and New Zealand Intensive Care Society Centre for Outcomes and Resource Evaluation reported in the Jan. 17, 2017 edition of JAMA. “Seymour and colleagues published data concerning the validity of a 2 or more–point change in the Sequential [Sepsis-related] Organ Failure Assessment (SOFA) score as a means of identifying sepsis among patients who are critically ill with suspected infection, assuming a SOFA of 0 for patients not known to have preexisting organ dysfunction (JAMA. 2016; 315[8]:762-74). In addition, the concept of the quick SOFA (qSOFA) score was introduced as a possible predictive tool among encounters with suspected infection outside the intensive care unit (ICU). These data were drawn from North American cohorts and a single German cohort and have not been validated externally.”
The mean age of the patients was 63 years, 45% were women, and the most common diagnosis was bacterial pneumonia (18%). Nearly 19% of patients died in the hospital and 56% died or experienced an ICU length of stay of at least 3 days or more. The researchers found that the SOFA score increased by 2 or more points in 90% of patients, while 87% manifested 2 or more SIRS criteria, and 54% had a qSOFA score of 2 or more points. In addition, discrimination of in-hospital mortality was significantly higher using SOFA (area under the receiving operating characteristic curve [AUROC], 0.753), compared with both SIRS criteria (AUROC, 0.589) and qSOFA (0.607); the between-group difference reached a P value of less than .001. Similar results were seen for the composite outcome of in-hospital mortality or an ICU length of stay of 3 days or more, which was higher using SOFA (AUROC, 0.736), compared with both SIRS criteria (AUROC, 0.609) and qSOFA (0.606); the between-group difference also reached a P value of less than .001.
The researchers acknowledged certain limitations of the study, including the fact that SOFA, SIRS criteria, and qSOFA could only be studied for the first 24 hours in the ICU. “Biochemical and physiological values could have come from any time within the first 24 hours of ICU admission and, as a result, could not be more accurately linked to the timing of the diagnosis of infection,” they wrote. “The SOFA score used should be considered a modification of the original because the cardiovascular component was estimated without knowledge of inotrope or vasopressor dose. The incidence of nosocomial infections and of infections in patients admitted with another diagnosis were unknown.”
Three of the seven study authors disclosed that they receive salary support from Monash University in Melbourne. The remainder reported having no financial disclosures.
FROM JAMA
Key clinical point:
Major finding: The SOFA score increased by 2 or more points in 90% of patients, while 87% manifested 2 or more SIRS criteria, and 54% had a qSOFA score of 2 or more points.
Data source: A retrospective cohort study of 184,875 patients with an infection-related primary diagnosis who were admitted to 182 ICUs in Australia and New Zealand between 2000 and 2015.
Disclosures: Three of the seven study authors disclosed that they receive salary support from Monash University in Melbourne. The remainder reported having no financial disclosures.