User login
COVID fallout: ‘Alarming’ dip in routine vax for pregnant women
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
FROM ACOG 2022
USPSTF recommendation roundup
In 2021, the US Preventive Services Task Force (USPSTF) considered 13 topics and made a total of 23 recommendations. They reviewed only 1 new topic. The other 12 were updates of topics previously addressed; no changes were made in 9 of them. In 3, the recommended age of screening or the criteria for screening were expanded. This Practice Alert will review the recommendations made and highlight new recommendations and any changes to previous ones. All complete recommendation statements, rationales, clinical considerations, and evidence reports can be found on the USPSTF website at https://uspreventiveservicestaskforce.org/uspstf/home.1
Dental caries in children
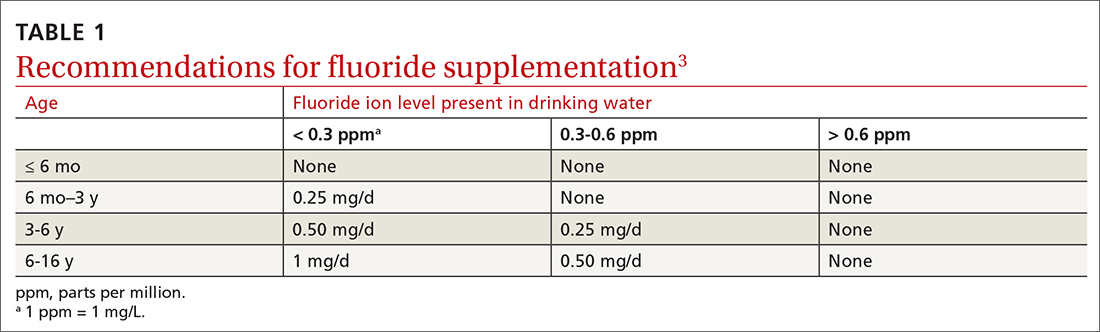
Dental caries affect about 23% of children between the ages of 2 and 5 years and are associated with multiple adverse social outcomes and medical conditions.2 The best way to prevent tooth decay, other than regular brushing with fluoride toothpaste, is to drink water with recommended amounts of fluoride (≥ 0.6 parts fluoride per million parts water).2 The USPSTF reaffirmed its recommendation from 2014 that stated when a local water supply lacks sufficient fluoride, primary care clinicians should prescribe oral supplementation for infants and children in the form of fluoride drops starting at age 6 months. The dosage of fluoride depends on patient age and fluoride concentration in the local water (TABLE 13). The USPSTF also recommends applying topical fluoride as 5% sodium fluoride varnish, every 6 months, starting when the primary teeth erupt.2
In addition to fluoride supplements and topical varnish, should clinicians perform screening examinations looking for dental caries? The USPSTF feels there is not enough evidence to assess this practice and gives it an “I” rating (insufficient evidence).
Preventive interventions in pregnancy
In 2021, the USPSTF assessed 3 topics related to pregnancy and prenatal care.
Screening for gestational diabetes. The USPSTF gave a “B” recommendation for screening at 24 weeks of pregnancy or after, but an “I” statement for screening prior to 24 weeks.4 Screening can involve a 1-step or 2-step protocol.
The 2-step protocol is most commonly used in the United States. It involves first measuring serum glucose after a nonfasting 50-g oral glucose challenge; if the resulting level is high, the second step is a 75- or 100-g oral glucose tolerance test lasting 3 hours. The 1-step protocol involves measuring a fasting glucose level, followed by a 75-g oral glucose challenge with glucose levels measured at 1 and 2 hours.
Healthy weight gain in pregnancy. This was the only new topic the USPSTF assessed last year. The resulting recommendation is to offer pregnant women behavioral counseling to promote healthy weight gain and to prevent excessive weight gain in pregnancy. The recommended weight gain depends on the mother’s prepregnancy weight status: 28 to 40 lbs if the mother is underweight; 25 to 35 lbs if she is not under- or overweight; 15 to 25 lbs if she is overweight; and 11 to 20 lbs if she is obese.5 Healthy weight gain contributes to preventing gestational diabetes, emergency cesarean sections, and infant macrosomia.
Continue to: Low-dose aspirin
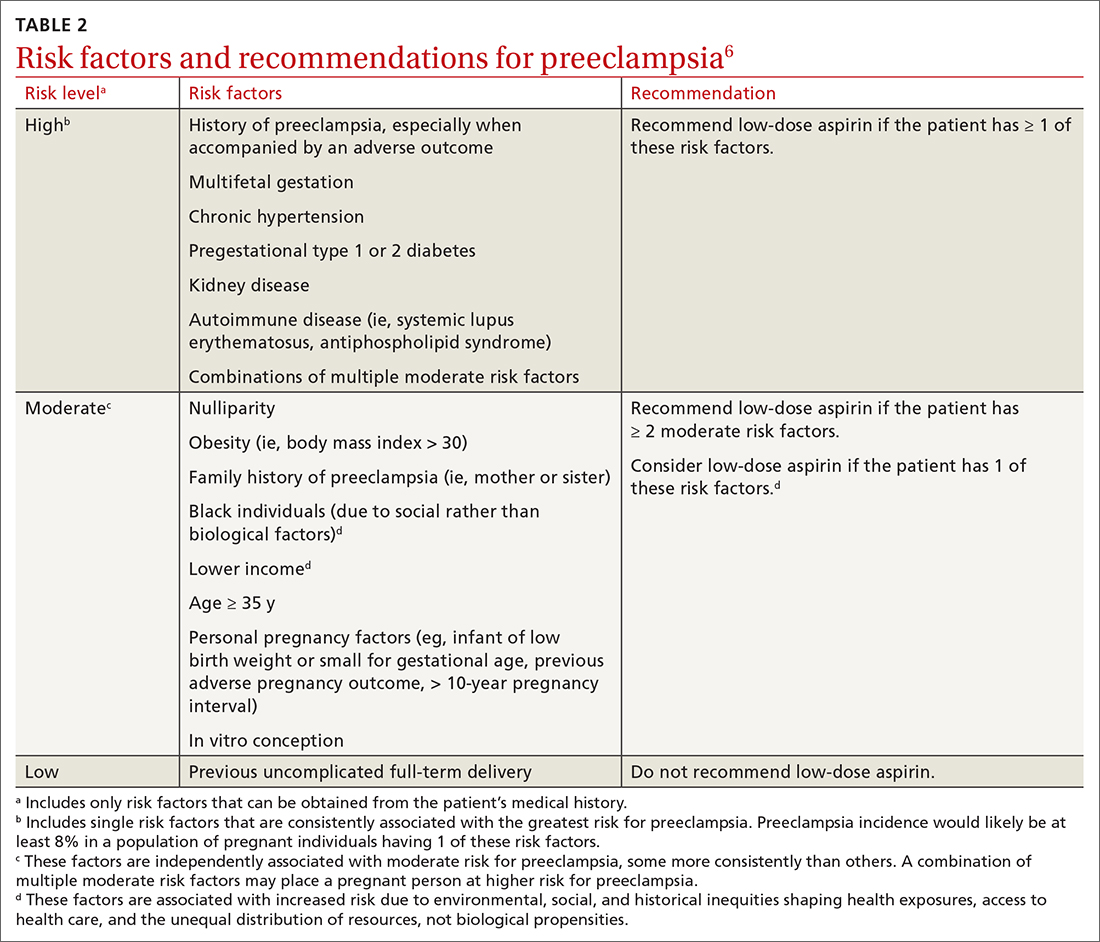
Low-dose aspirin. Reaffirming a recommendation from 2014, the USPSTF advises low-dose aspirin (81 mg/d) starting after 12 weeks’ gestation for all pregnant women who are at high risk for preeclampsia. TABLE 26 lists high- and moderate-risk conditions for preeclampsia and the recommendation for the use of low-dose aspirin.
Sexually transmitted infections
Screening for both chlamydia and gonorrhea in sexually active females through age 24 years was given a “B” recommendation, reaffirming the 2014 recommendation.7 Screening for these 2 sexually transmitted infections (STIs) is also recommended for women 25 years and older who are at increased risk of STIs. Risk is defined as having a new sex partner, more than 1 sex partner, a sex partner who has other sex partners, or a sex partner who has an STI; not using condoms consistently; having a previous STI; exchanging sex for money or drugs; or having a history of incarceration.
Screen for both infections simultaneously using a nucleic acid amplification test, testing all sites of sexual exposure. Urine testing can replace cervical, vaginal, and urethral testing. Those found to be positive for either STI should be treated according to the most recent treatment guidelines from the Centers for Disease Control and Prevention (CDC). And sexual partners should be advised to undergo testing.8,9
The USPSTF could not find evidence for the benefits and harms of screening for STIs in men. Remember that screening applies to those who are asymptomatic. Male sex partners of those found to be infected should be tested, as should those who show any signs or symptoms of an STI. A recent Practice Alert described the most current CDC guidance for diagnosing and treating STIs.9
Type 2 diabetes and prediabetes
Screening for type 2 diabetes (T2D) and prediabetes is now recommended for adults ages 35 to 70 years who are overweight or obese.10 The age to start screening has been lowered to 35 years from the previous recommendation in 2015, which recommended starting at age 40. In addition, the recommendation states that patients with prediabetes should be referred for preventive interventions. It is important that referral is included in the statement because the Affordable Care Act mandates that USPSTF “A” and “B” recommendations must be covered by commercial health insurance with no copay or deductible.
Continue to: Screening can be conducted...
Screening can be conducted using a fasting plasma glucose or A1C level, or with an oral glucose tolerance test. Interventions that can prevent or delay the onset of T2D in those with prediabetes include lifestyle interventions that focus on diet and physical activity, and the use of metformin (although metformin has not been approved for this by the US Food and Drug Administration).
Changes to cancer screening recommendations
In 2021, the USPSTF reviewed and modified its recommendations on screening for 2 types of cancer: colorectal and lung.
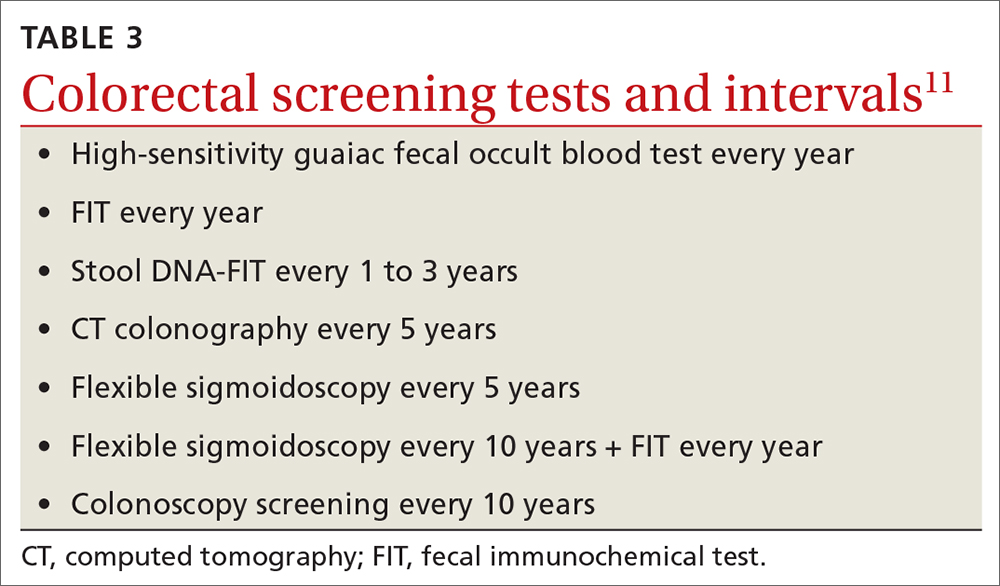
For colorectal cancer, the age at which to start screening was lowered from 50 years to 45 years.11 Screening at this earlier age is a “B” recommendation, because, while there is benefit from screening, it is less than for older age groups. Screening individuals ages 50 to 75 years remains an “A” recommendation, and for those ages 76 to 85 years it remains a “C” recommendation. A “C” recommendation means that the overall benefits are small but some individuals might benefit based on their overall health and prior screening results. In its clinical considerations, the USPSTF recommends against screening in those ages 85 and older but, curiously, does not list it as a “D” recommendation. The screening methods and recommended screening intervals for each appear in TABLE 3.11
For lung cancer, annual screening using low-dose computed tomography (CT) was first recommended by the USPSTF in 2013 for adults ages 55 to 80 years with a 30-pack-year smoking history. Screening could stop once 15 years had passed since smoking cessation. In 2021, the USPSTF lowered the age to initiate screening to 50 years, and the smoking history threshold to 20 pack-years.12 If these recommendations are followed, a current smoker who does not quit smoking could possibly receive 30 annual CT scans. The recommendation does state that screening should stop once a person develops a health condition that significantly affects life expectancy or ability to have lung surgery.
For primary prevention of lung cancer and other chronic diseases through smoking cessation, the USPSTF also reassessed its 2015 recommendations. It reaffirmed the “A” recommendation to ask adults about tobacco use and, for tobacco users, to recommend cessation and provide behavioral therapy and approved pharmacotherapy.13 The recommendation differed for pregnant adults in that the USPSTF is unsure about the potential harms of pharmacotherapy in pregnancy and gives that an “I” statement.13 An additional “I” statement was made about the use of electronic cigarettes for smoking cessation; the USPSTF recommends using behavioral and pharmacotherapy interventions with proven effectiveness and safety instead.
Continue to: 4 additional recommendation updates with no changes
4 additional recommendation updates with no changes
Screening for high blood pressure in adults ages 18 years and older continues to receive an “A” recommendation.14 Importantly, the recommendation states that confirmation of high blood pressure should be made in an out-of-office setting before initiating treatment. Screening for vitamin D deficiency in adults and hearing loss in older adults both continue with “I” statements,15,16 and screening for asymptomatic carotid artery stenosis continues to receive a “D” recommendation.17 The implications of the vitamin D “I” statement were discussed in a previous Practice Alert.18
Continuing value of the USPSTF
The USPSTF continues to set the gold standard for assessment of preventive interventions, and its decisions affect first-dollar coverage by commercial health insurance. The reaffirmation of past recommendations demonstrates the value of adhering to rigorous evidence-based methods (if they are done correctly, they rarely must be markedly changed). And the updating of screening criteria shows the need to constantly review the evolving evidence for current recommendations. Once again, however, funding and staffing limitations allowed the USPSTF to assess only 1 new topic. A listing of all the 2021 recommendations is in TABLE 4.1


1. USPSTF. Recommendation topics. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation-topics
2. USPSTF. Prevention of dental caries in children younger than 5 years: screening and interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1#bootstrap-panel—4
3. ADA. Dietary fluoride supplements: evidence-based clinical recommendations. Accessed April 14, 2022. www.ada.org/-/media/project/ada-organization/ada/ada-org/files/resources/research/ada_evidence-based_fluoride_supplement_chairside_guide.pdf?rev=60850dca0dcc41038efda83d42b1c2e0&hash=FEC2BBEA0C892FB12C098E33344E48B4
4. USPSTF. Gestational diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening
5. USPSTF. Healthy weight and weight gain in pregnancy: behavioral counseling interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/healthy-weight-and-weight-gain-during-pregnancy-behavioral-counseling-interventions
6. USPSTF. Aspirin use to prevent preeclampsia and related morbidity and mortality: preventive medication. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/low-dose-aspirin-use-for-the-prevention-of-morbidity-and-mortality-from-preeclampsia-preventive-medication
7. USPSTF. Chlamydia and gonorrhea: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/chlamydia-and-gonorrhea-screening
8. Workowski KA, Bauchman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70:1-187.
9. Campos-Outcalt D. CDC guidelines on sexually transmitted infections. J Fam Pract. 2021;70:506-509.
10. USPSTF. Prediabetes and type 2 diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/screening-for-prediabetes-and-type-2-diabetes
11. USPSTF. Colorectal cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
12. USPSTF. Lung cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
13. USPSTF. Tobacco smoking cessation in adults, including pregnant persons: interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
14. USPSTF. Hypertension in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hypertension-in-adults-screening
15. USPSTF. Vitamin D deficiency in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/vitamin-d-deficiency-screening
16. USPSTF. Hearing loss in older adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hearing-loss-in-older-adults-screening
17. USPSTF. Asymptomatic carotid artery stenosis: screening. Access April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
18. Campos-Outcalt D. How to proceed when it comes to vitamin D. J Fam Pract. 2021;70:289-292.
In 2021, the US Preventive Services Task Force (USPSTF) considered 13 topics and made a total of 23 recommendations. They reviewed only 1 new topic. The other 12 were updates of topics previously addressed; no changes were made in 9 of them. In 3, the recommended age of screening or the criteria for screening were expanded. This Practice Alert will review the recommendations made and highlight new recommendations and any changes to previous ones. All complete recommendation statements, rationales, clinical considerations, and evidence reports can be found on the USPSTF website at https://uspreventiveservicestaskforce.org/uspstf/home.1
Dental caries in children
Dental caries affect about 23% of children between the ages of 2 and 5 years and are associated with multiple adverse social outcomes and medical conditions.2 The best way to prevent tooth decay, other than regular brushing with fluoride toothpaste, is to drink water with recommended amounts of fluoride (≥ 0.6 parts fluoride per million parts water).2 The USPSTF reaffirmed its recommendation from 2014 that stated when a local water supply lacks sufficient fluoride, primary care clinicians should prescribe oral supplementation for infants and children in the form of fluoride drops starting at age 6 months. The dosage of fluoride depends on patient age and fluoride concentration in the local water (TABLE 13). The USPSTF also recommends applying topical fluoride as 5% sodium fluoride varnish, every 6 months, starting when the primary teeth erupt.2

In addition to fluoride supplements and topical varnish, should clinicians perform screening examinations looking for dental caries? The USPSTF feels there is not enough evidence to assess this practice and gives it an “I” rating (insufficient evidence).
Preventive interventions in pregnancy
In 2021, the USPSTF assessed 3 topics related to pregnancy and prenatal care.
Screening for gestational diabetes. The USPSTF gave a “B” recommendation for screening at 24 weeks of pregnancy or after, but an “I” statement for screening prior to 24 weeks.4 Screening can involve a 1-step or 2-step protocol.
The 2-step protocol is most commonly used in the United States. It involves first measuring serum glucose after a nonfasting 50-g oral glucose challenge; if the resulting level is high, the second step is a 75- or 100-g oral glucose tolerance test lasting 3 hours. The 1-step protocol involves measuring a fasting glucose level, followed by a 75-g oral glucose challenge with glucose levels measured at 1 and 2 hours.
Healthy weight gain in pregnancy. This was the only new topic the USPSTF assessed last year. The resulting recommendation is to offer pregnant women behavioral counseling to promote healthy weight gain and to prevent excessive weight gain in pregnancy. The recommended weight gain depends on the mother’s prepregnancy weight status: 28 to 40 lbs if the mother is underweight; 25 to 35 lbs if she is not under- or overweight; 15 to 25 lbs if she is overweight; and 11 to 20 lbs if she is obese.5 Healthy weight gain contributes to preventing gestational diabetes, emergency cesarean sections, and infant macrosomia.
Continue to: Low-dose aspirin
Low-dose aspirin. Reaffirming a recommendation from 2014, the USPSTF advises low-dose aspirin (81 mg/d) starting after 12 weeks’ gestation for all pregnant women who are at high risk for preeclampsia. TABLE 26 lists high- and moderate-risk conditions for preeclampsia and the recommendation for the use of low-dose aspirin.

Sexually transmitted infections
Screening for both chlamydia and gonorrhea in sexually active females through age 24 years was given a “B” recommendation, reaffirming the 2014 recommendation.7 Screening for these 2 sexually transmitted infections (STIs) is also recommended for women 25 years and older who are at increased risk of STIs. Risk is defined as having a new sex partner, more than 1 sex partner, a sex partner who has other sex partners, or a sex partner who has an STI; not using condoms consistently; having a previous STI; exchanging sex for money or drugs; or having a history of incarceration.
Screen for both infections simultaneously using a nucleic acid amplification test, testing all sites of sexual exposure. Urine testing can replace cervical, vaginal, and urethral testing. Those found to be positive for either STI should be treated according to the most recent treatment guidelines from the Centers for Disease Control and Prevention (CDC). And sexual partners should be advised to undergo testing.8,9
The USPSTF could not find evidence for the benefits and harms of screening for STIs in men. Remember that screening applies to those who are asymptomatic. Male sex partners of those found to be infected should be tested, as should those who show any signs or symptoms of an STI. A recent Practice Alert described the most current CDC guidance for diagnosing and treating STIs.9
Type 2 diabetes and prediabetes
Screening for type 2 diabetes (T2D) and prediabetes is now recommended for adults ages 35 to 70 years who are overweight or obese.10 The age to start screening has been lowered to 35 years from the previous recommendation in 2015, which recommended starting at age 40. In addition, the recommendation states that patients with prediabetes should be referred for preventive interventions. It is important that referral is included in the statement because the Affordable Care Act mandates that USPSTF “A” and “B” recommendations must be covered by commercial health insurance with no copay or deductible.
Continue to: Screening can be conducted...
Screening can be conducted using a fasting plasma glucose or A1C level, or with an oral glucose tolerance test. Interventions that can prevent or delay the onset of T2D in those with prediabetes include lifestyle interventions that focus on diet and physical activity, and the use of metformin (although metformin has not been approved for this by the US Food and Drug Administration).
Changes to cancer screening recommendations
In 2021, the USPSTF reviewed and modified its recommendations on screening for 2 types of cancer: colorectal and lung.
For colorectal cancer, the age at which to start screening was lowered from 50 years to 45 years.11 Screening at this earlier age is a “B” recommendation, because, while there is benefit from screening, it is less than for older age groups. Screening individuals ages 50 to 75 years remains an “A” recommendation, and for those ages 76 to 85 years it remains a “C” recommendation. A “C” recommendation means that the overall benefits are small but some individuals might benefit based on their overall health and prior screening results. In its clinical considerations, the USPSTF recommends against screening in those ages 85 and older but, curiously, does not list it as a “D” recommendation. The screening methods and recommended screening intervals for each appear in TABLE 3.11

For lung cancer, annual screening using low-dose computed tomography (CT) was first recommended by the USPSTF in 2013 for adults ages 55 to 80 years with a 30-pack-year smoking history. Screening could stop once 15 years had passed since smoking cessation. In 2021, the USPSTF lowered the age to initiate screening to 50 years, and the smoking history threshold to 20 pack-years.12 If these recommendations are followed, a current smoker who does not quit smoking could possibly receive 30 annual CT scans. The recommendation does state that screening should stop once a person develops a health condition that significantly affects life expectancy or ability to have lung surgery.
For primary prevention of lung cancer and other chronic diseases through smoking cessation, the USPSTF also reassessed its 2015 recommendations. It reaffirmed the “A” recommendation to ask adults about tobacco use and, for tobacco users, to recommend cessation and provide behavioral therapy and approved pharmacotherapy.13 The recommendation differed for pregnant adults in that the USPSTF is unsure about the potential harms of pharmacotherapy in pregnancy and gives that an “I” statement.13 An additional “I” statement was made about the use of electronic cigarettes for smoking cessation; the USPSTF recommends using behavioral and pharmacotherapy interventions with proven effectiveness and safety instead.
Continue to: 4 additional recommendation updates with no changes
4 additional recommendation updates with no changes
Screening for high blood pressure in adults ages 18 years and older continues to receive an “A” recommendation.14 Importantly, the recommendation states that confirmation of high blood pressure should be made in an out-of-office setting before initiating treatment. Screening for vitamin D deficiency in adults and hearing loss in older adults both continue with “I” statements,15,16 and screening for asymptomatic carotid artery stenosis continues to receive a “D” recommendation.17 The implications of the vitamin D “I” statement were discussed in a previous Practice Alert.18
Continuing value of the USPSTF
The USPSTF continues to set the gold standard for assessment of preventive interventions, and its decisions affect first-dollar coverage by commercial health insurance. The reaffirmation of past recommendations demonstrates the value of adhering to rigorous evidence-based methods (if they are done correctly, they rarely must be markedly changed). And the updating of screening criteria shows the need to constantly review the evolving evidence for current recommendations. Once again, however, funding and staffing limitations allowed the USPSTF to assess only 1 new topic. A listing of all the 2021 recommendations is in TABLE 4.1


In 2021, the US Preventive Services Task Force (USPSTF) considered 13 topics and made a total of 23 recommendations. They reviewed only 1 new topic. The other 12 were updates of topics previously addressed; no changes were made in 9 of them. In 3, the recommended age of screening or the criteria for screening were expanded. This Practice Alert will review the recommendations made and highlight new recommendations and any changes to previous ones. All complete recommendation statements, rationales, clinical considerations, and evidence reports can be found on the USPSTF website at https://uspreventiveservicestaskforce.org/uspstf/home.1
Dental caries in children
Dental caries affect about 23% of children between the ages of 2 and 5 years and are associated with multiple adverse social outcomes and medical conditions.2 The best way to prevent tooth decay, other than regular brushing with fluoride toothpaste, is to drink water with recommended amounts of fluoride (≥ 0.6 parts fluoride per million parts water).2 The USPSTF reaffirmed its recommendation from 2014 that stated when a local water supply lacks sufficient fluoride, primary care clinicians should prescribe oral supplementation for infants and children in the form of fluoride drops starting at age 6 months. The dosage of fluoride depends on patient age and fluoride concentration in the local water (TABLE 13). The USPSTF also recommends applying topical fluoride as 5% sodium fluoride varnish, every 6 months, starting when the primary teeth erupt.2

In addition to fluoride supplements and topical varnish, should clinicians perform screening examinations looking for dental caries? The USPSTF feels there is not enough evidence to assess this practice and gives it an “I” rating (insufficient evidence).
Preventive interventions in pregnancy
In 2021, the USPSTF assessed 3 topics related to pregnancy and prenatal care.
Screening for gestational diabetes. The USPSTF gave a “B” recommendation for screening at 24 weeks of pregnancy or after, but an “I” statement for screening prior to 24 weeks.4 Screening can involve a 1-step or 2-step protocol.
The 2-step protocol is most commonly used in the United States. It involves first measuring serum glucose after a nonfasting 50-g oral glucose challenge; if the resulting level is high, the second step is a 75- or 100-g oral glucose tolerance test lasting 3 hours. The 1-step protocol involves measuring a fasting glucose level, followed by a 75-g oral glucose challenge with glucose levels measured at 1 and 2 hours.
Healthy weight gain in pregnancy. This was the only new topic the USPSTF assessed last year. The resulting recommendation is to offer pregnant women behavioral counseling to promote healthy weight gain and to prevent excessive weight gain in pregnancy. The recommended weight gain depends on the mother’s prepregnancy weight status: 28 to 40 lbs if the mother is underweight; 25 to 35 lbs if she is not under- or overweight; 15 to 25 lbs if she is overweight; and 11 to 20 lbs if she is obese.5 Healthy weight gain contributes to preventing gestational diabetes, emergency cesarean sections, and infant macrosomia.
Continue to: Low-dose aspirin
Low-dose aspirin. Reaffirming a recommendation from 2014, the USPSTF advises low-dose aspirin (81 mg/d) starting after 12 weeks’ gestation for all pregnant women who are at high risk for preeclampsia. TABLE 26 lists high- and moderate-risk conditions for preeclampsia and the recommendation for the use of low-dose aspirin.

Sexually transmitted infections
Screening for both chlamydia and gonorrhea in sexually active females through age 24 years was given a “B” recommendation, reaffirming the 2014 recommendation.7 Screening for these 2 sexually transmitted infections (STIs) is also recommended for women 25 years and older who are at increased risk of STIs. Risk is defined as having a new sex partner, more than 1 sex partner, a sex partner who has other sex partners, or a sex partner who has an STI; not using condoms consistently; having a previous STI; exchanging sex for money or drugs; or having a history of incarceration.
Screen for both infections simultaneously using a nucleic acid amplification test, testing all sites of sexual exposure. Urine testing can replace cervical, vaginal, and urethral testing. Those found to be positive for either STI should be treated according to the most recent treatment guidelines from the Centers for Disease Control and Prevention (CDC). And sexual partners should be advised to undergo testing.8,9
The USPSTF could not find evidence for the benefits and harms of screening for STIs in men. Remember that screening applies to those who are asymptomatic. Male sex partners of those found to be infected should be tested, as should those who show any signs or symptoms of an STI. A recent Practice Alert described the most current CDC guidance for diagnosing and treating STIs.9
Type 2 diabetes and prediabetes
Screening for type 2 diabetes (T2D) and prediabetes is now recommended for adults ages 35 to 70 years who are overweight or obese.10 The age to start screening has been lowered to 35 years from the previous recommendation in 2015, which recommended starting at age 40. In addition, the recommendation states that patients with prediabetes should be referred for preventive interventions. It is important that referral is included in the statement because the Affordable Care Act mandates that USPSTF “A” and “B” recommendations must be covered by commercial health insurance with no copay or deductible.
Continue to: Screening can be conducted...
Screening can be conducted using a fasting plasma glucose or A1C level, or with an oral glucose tolerance test. Interventions that can prevent or delay the onset of T2D in those with prediabetes include lifestyle interventions that focus on diet and physical activity, and the use of metformin (although metformin has not been approved for this by the US Food and Drug Administration).
Changes to cancer screening recommendations
In 2021, the USPSTF reviewed and modified its recommendations on screening for 2 types of cancer: colorectal and lung.
For colorectal cancer, the age at which to start screening was lowered from 50 years to 45 years.11 Screening at this earlier age is a “B” recommendation, because, while there is benefit from screening, it is less than for older age groups. Screening individuals ages 50 to 75 years remains an “A” recommendation, and for those ages 76 to 85 years it remains a “C” recommendation. A “C” recommendation means that the overall benefits are small but some individuals might benefit based on their overall health and prior screening results. In its clinical considerations, the USPSTF recommends against screening in those ages 85 and older but, curiously, does not list it as a “D” recommendation. The screening methods and recommended screening intervals for each appear in TABLE 3.11

For lung cancer, annual screening using low-dose computed tomography (CT) was first recommended by the USPSTF in 2013 for adults ages 55 to 80 years with a 30-pack-year smoking history. Screening could stop once 15 years had passed since smoking cessation. In 2021, the USPSTF lowered the age to initiate screening to 50 years, and the smoking history threshold to 20 pack-years.12 If these recommendations are followed, a current smoker who does not quit smoking could possibly receive 30 annual CT scans. The recommendation does state that screening should stop once a person develops a health condition that significantly affects life expectancy or ability to have lung surgery.
For primary prevention of lung cancer and other chronic diseases through smoking cessation, the USPSTF also reassessed its 2015 recommendations. It reaffirmed the “A” recommendation to ask adults about tobacco use and, for tobacco users, to recommend cessation and provide behavioral therapy and approved pharmacotherapy.13 The recommendation differed for pregnant adults in that the USPSTF is unsure about the potential harms of pharmacotherapy in pregnancy and gives that an “I” statement.13 An additional “I” statement was made about the use of electronic cigarettes for smoking cessation; the USPSTF recommends using behavioral and pharmacotherapy interventions with proven effectiveness and safety instead.
Continue to: 4 additional recommendation updates with no changes
4 additional recommendation updates with no changes
Screening for high blood pressure in adults ages 18 years and older continues to receive an “A” recommendation.14 Importantly, the recommendation states that confirmation of high blood pressure should be made in an out-of-office setting before initiating treatment. Screening for vitamin D deficiency in adults and hearing loss in older adults both continue with “I” statements,15,16 and screening for asymptomatic carotid artery stenosis continues to receive a “D” recommendation.17 The implications of the vitamin D “I” statement were discussed in a previous Practice Alert.18
Continuing value of the USPSTF
The USPSTF continues to set the gold standard for assessment of preventive interventions, and its decisions affect first-dollar coverage by commercial health insurance. The reaffirmation of past recommendations demonstrates the value of adhering to rigorous evidence-based methods (if they are done correctly, they rarely must be markedly changed). And the updating of screening criteria shows the need to constantly review the evolving evidence for current recommendations. Once again, however, funding and staffing limitations allowed the USPSTF to assess only 1 new topic. A listing of all the 2021 recommendations is in TABLE 4.1


1. USPSTF. Recommendation topics. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation-topics
2. USPSTF. Prevention of dental caries in children younger than 5 years: screening and interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1#bootstrap-panel—4
3. ADA. Dietary fluoride supplements: evidence-based clinical recommendations. Accessed April 14, 2022. www.ada.org/-/media/project/ada-organization/ada/ada-org/files/resources/research/ada_evidence-based_fluoride_supplement_chairside_guide.pdf?rev=60850dca0dcc41038efda83d42b1c2e0&hash=FEC2BBEA0C892FB12C098E33344E48B4
4. USPSTF. Gestational diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening
5. USPSTF. Healthy weight and weight gain in pregnancy: behavioral counseling interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/healthy-weight-and-weight-gain-during-pregnancy-behavioral-counseling-interventions
6. USPSTF. Aspirin use to prevent preeclampsia and related morbidity and mortality: preventive medication. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/low-dose-aspirin-use-for-the-prevention-of-morbidity-and-mortality-from-preeclampsia-preventive-medication
7. USPSTF. Chlamydia and gonorrhea: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/chlamydia-and-gonorrhea-screening
8. Workowski KA, Bauchman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70:1-187.
9. Campos-Outcalt D. CDC guidelines on sexually transmitted infections. J Fam Pract. 2021;70:506-509.
10. USPSTF. Prediabetes and type 2 diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/screening-for-prediabetes-and-type-2-diabetes
11. USPSTF. Colorectal cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
12. USPSTF. Lung cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
13. USPSTF. Tobacco smoking cessation in adults, including pregnant persons: interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
14. USPSTF. Hypertension in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hypertension-in-adults-screening
15. USPSTF. Vitamin D deficiency in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/vitamin-d-deficiency-screening
16. USPSTF. Hearing loss in older adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hearing-loss-in-older-adults-screening
17. USPSTF. Asymptomatic carotid artery stenosis: screening. Access April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
18. Campos-Outcalt D. How to proceed when it comes to vitamin D. J Fam Pract. 2021;70:289-292.
1. USPSTF. Recommendation topics. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation-topics
2. USPSTF. Prevention of dental caries in children younger than 5 years: screening and interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1#bootstrap-panel—4
3. ADA. Dietary fluoride supplements: evidence-based clinical recommendations. Accessed April 14, 2022. www.ada.org/-/media/project/ada-organization/ada/ada-org/files/resources/research/ada_evidence-based_fluoride_supplement_chairside_guide.pdf?rev=60850dca0dcc41038efda83d42b1c2e0&hash=FEC2BBEA0C892FB12C098E33344E48B4
4. USPSTF. Gestational diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening
5. USPSTF. Healthy weight and weight gain in pregnancy: behavioral counseling interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/healthy-weight-and-weight-gain-during-pregnancy-behavioral-counseling-interventions
6. USPSTF. Aspirin use to prevent preeclampsia and related morbidity and mortality: preventive medication. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/low-dose-aspirin-use-for-the-prevention-of-morbidity-and-mortality-from-preeclampsia-preventive-medication
7. USPSTF. Chlamydia and gonorrhea: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/chlamydia-and-gonorrhea-screening
8. Workowski KA, Bauchman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70:1-187.
9. Campos-Outcalt D. CDC guidelines on sexually transmitted infections. J Fam Pract. 2021;70:506-509.
10. USPSTF. Prediabetes and type 2 diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/screening-for-prediabetes-and-type-2-diabetes
11. USPSTF. Colorectal cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
12. USPSTF. Lung cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
13. USPSTF. Tobacco smoking cessation in adults, including pregnant persons: interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
14. USPSTF. Hypertension in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hypertension-in-adults-screening
15. USPSTF. Vitamin D deficiency in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/vitamin-d-deficiency-screening
16. USPSTF. Hearing loss in older adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hearing-loss-in-older-adults-screening
17. USPSTF. Asymptomatic carotid artery stenosis: screening. Access April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
18. Campos-Outcalt D. How to proceed when it comes to vitamin D. J Fam Pract. 2021;70:289-292.
Screening for hypertensive disorders of pregnancy is often incomplete
Nearly three-quarters of clinicians reported screening patients for hypertensive disorders of pregnancy, but only one-quarter comprehensively identified cardiovascular risk, based on survey data from approximately 1,500 clinicians in the United States.
Rates of hypertensive disorders of pregnancy have been on the rise in the United States for the past decade, and women with a history of these disorders require cardiovascular risk monitoring during the postpartum period and beyond, wrote Nicole D. Ford, PhD, of the Centers for Disease Control and Prevention, Atlanta, and colleagues. Specifically, the American College of Obstetricians and Gynecologists recommends cardiovascular risk evaluation and lifestyle modification for these individuals, the researchers said.
The most effective management of women with a history of hypertensive disorders of pregnancy will likely involve a team effort by primary care, ob.gyns., and cardiologists, but data on clinician screening and referrals are limited, they added.
In a study published in Obstetrics & Gynecology, the researchers reviewed data from a cross-sectional, web-based survey of clinicians practicing in the United States (Fall DocStyles 2020). The study population of 1,502 respondents with complete surveys included 1,000 primary care physicians, 251 ob.gyns., and 251 nurse practitioners or physician assistants. Approximately 60% of the respondents were male, and approximately 65% had been in practice for at least 10 years.
Overall, 73.6% of clinicians reported screening patients for a history of hypertensive disorders of pregnancy. The screening rates were highest among ob.gyns. (94.8%).
However, although 93.9% of clinicians overall correctly identified at least one potential risk associated with hypertensive disorders of pregnancy, only 24.8% correctly identified all cardiovascular risks associated with hypertensive disorders of pregnancy listed in the survey, the researchers noted.
Screening rates ranged from 49% to 91% for pregnant women, 34%-75% for postpartum women, 26%-61% for nonpregnant reproductive-age women, 20%-45% for perimenopausal or menopausal women, and 1%-4% for others outside of these categories.
The most often–cited barriers to referral were lack of patient follow-through (51.5%) and patient refusal (33.6%). To improve and facilitate referrals, respondents’ most frequent resource request was for more referral options (42.9%), followed by patient education materials (36.2%), and professional guidelines (34.1%).
In a multivariate analysis, primary care physicians were more than five times as likely to report not screening patients for hypertensive disorders of pregnancy (adjusted prevalence ratio, 5.54); nurse practitioners and physician assistants were more than seven times as likely (adjusted prevalence ratio, 7.42).
The researchers also found that clinicians who saw fewer than 80 patients per week were almost twice as likely not to screen for hypertensive disorders of pregnancy than those who saw 110 or more patients per week (adjusted prevalence ratio, 1.81).
“Beyond the immediate postpartum period, there is a lack of clear guidance on CVD [cardiovascular disease] evaluation and ongoing monitoring in women with history of hypertensive disorders of pregnancy,” the researchers wrote in their discussion. “Recognizing hypertensive disorders of pregnancy as a risk factor for CVD may allow clinicians to identify women requiring early evaluation and intervention,” they said.
The study findings were limited by several factors including potentially biased estimates of screening practices, and the potential for selection bias because of the convenience sample used to recruit survey participants, the researchers noted.
However, the results were strengthened by the inclusion of data from several clinician types and the relatively large sample size, and are consistent with those of previous studies, they said. Based on the findings, addressing barriers at both the patient and clinician level and increasing both patient and clinician education about the long-term risks of hypertensive disorders of pregnancy might increase cardiovascular screening and subsequent referrals, they concluded.
More education, improved screening tools needed
“Unfortunately, most CVD risk stratification scores such as the Framingham score do not include pregnancy complications, despite excellent evidence that pregnancy complications increase risk of CVD,” said Catherine M. Albright, MD, MS, of the University of Washington, Seattle, in an interview. “This is likely because these scores were developed primarily to screen for CVD risk in men. Given the rising incidence of hypertensive disorders of pregnancy and the clear evidence that this is a risk factor for future CVD, more studies like this one are needed in order to help guide patient and provider education,” said Dr. Albright, who was not involved in the study.
“It is generally well reported within the ob.gyn. literature about the increased lifetime CVD risk related to hypertensive disorders of pregnancy and we, as ob.gyns., always ask about pregnancy history because of our specialty, which gives us the opportunity to counsel about future risks,” she said.
“Women’s health [including during pregnancy] has been undervalued and underresearched for a long time,” with limited focus on pregnancy-related issues until recently, Dr. Albright noted. “This is clear in the attitudes and education of the primary care providers in this study,” she said.
A major barrier to screening in clinical practice has been that the standard screening guidelines for CVD (for example, those published by the United States Preventive Services Taskforce) have not included pregnancy history, said Dr. Albright. “Subsequently, these questions are not asked during routine annual visits,” she said. Ideally, “we should be able to leverage the electronic medical record to prompt providers to view a previously recorded pregnancy history or to ask about pregnancy history as a routine part of CVD risk assessment, and, of course, additional education outside of ob.gyn. and cardiology is needed,” she said.
The clinical takeaway from the current study is that “every annual visit with a person who has been pregnant is an opportunity to ask about and document pregnancy history,” Dr. Albright said. “After the completion of childbearing, many patients no longer see an ob.gyn., so other providers need to feel comfortable asking about and counseling about risks related to pregnancy complications,” she added.
“It is clear that adverse pregnancy outcomes pose lifetime health risks,” said Dr. Albright. “We will continue to look into the mechanisms of this through research. However, right now the additional research that is needed is to determine the optimal screening and follow-up for patients with a history of hypertensive disorders of pregnancy, as well as to examine how existing CVD-screening algorithms can be modified to include adverse pregnancy outcomes,” she emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Albright had no financial conflicts to disclose.
Nearly three-quarters of clinicians reported screening patients for hypertensive disorders of pregnancy, but only one-quarter comprehensively identified cardiovascular risk, based on survey data from approximately 1,500 clinicians in the United States.
Rates of hypertensive disorders of pregnancy have been on the rise in the United States for the past decade, and women with a history of these disorders require cardiovascular risk monitoring during the postpartum period and beyond, wrote Nicole D. Ford, PhD, of the Centers for Disease Control and Prevention, Atlanta, and colleagues. Specifically, the American College of Obstetricians and Gynecologists recommends cardiovascular risk evaluation and lifestyle modification for these individuals, the researchers said.
The most effective management of women with a history of hypertensive disorders of pregnancy will likely involve a team effort by primary care, ob.gyns., and cardiologists, but data on clinician screening and referrals are limited, they added.
In a study published in Obstetrics & Gynecology, the researchers reviewed data from a cross-sectional, web-based survey of clinicians practicing in the United States (Fall DocStyles 2020). The study population of 1,502 respondents with complete surveys included 1,000 primary care physicians, 251 ob.gyns., and 251 nurse practitioners or physician assistants. Approximately 60% of the respondents were male, and approximately 65% had been in practice for at least 10 years.
Overall, 73.6% of clinicians reported screening patients for a history of hypertensive disorders of pregnancy. The screening rates were highest among ob.gyns. (94.8%).
However, although 93.9% of clinicians overall correctly identified at least one potential risk associated with hypertensive disorders of pregnancy, only 24.8% correctly identified all cardiovascular risks associated with hypertensive disorders of pregnancy listed in the survey, the researchers noted.
Screening rates ranged from 49% to 91% for pregnant women, 34%-75% for postpartum women, 26%-61% for nonpregnant reproductive-age women, 20%-45% for perimenopausal or menopausal women, and 1%-4% for others outside of these categories.
The most often–cited barriers to referral were lack of patient follow-through (51.5%) and patient refusal (33.6%). To improve and facilitate referrals, respondents’ most frequent resource request was for more referral options (42.9%), followed by patient education materials (36.2%), and professional guidelines (34.1%).
In a multivariate analysis, primary care physicians were more than five times as likely to report not screening patients for hypertensive disorders of pregnancy (adjusted prevalence ratio, 5.54); nurse practitioners and physician assistants were more than seven times as likely (adjusted prevalence ratio, 7.42).
The researchers also found that clinicians who saw fewer than 80 patients per week were almost twice as likely not to screen for hypertensive disorders of pregnancy than those who saw 110 or more patients per week (adjusted prevalence ratio, 1.81).
“Beyond the immediate postpartum period, there is a lack of clear guidance on CVD [cardiovascular disease] evaluation and ongoing monitoring in women with history of hypertensive disorders of pregnancy,” the researchers wrote in their discussion. “Recognizing hypertensive disorders of pregnancy as a risk factor for CVD may allow clinicians to identify women requiring early evaluation and intervention,” they said.
The study findings were limited by several factors including potentially biased estimates of screening practices, and the potential for selection bias because of the convenience sample used to recruit survey participants, the researchers noted.
However, the results were strengthened by the inclusion of data from several clinician types and the relatively large sample size, and are consistent with those of previous studies, they said. Based on the findings, addressing barriers at both the patient and clinician level and increasing both patient and clinician education about the long-term risks of hypertensive disorders of pregnancy might increase cardiovascular screening and subsequent referrals, they concluded.
More education, improved screening tools needed
“Unfortunately, most CVD risk stratification scores such as the Framingham score do not include pregnancy complications, despite excellent evidence that pregnancy complications increase risk of CVD,” said Catherine M. Albright, MD, MS, of the University of Washington, Seattle, in an interview. “This is likely because these scores were developed primarily to screen for CVD risk in men. Given the rising incidence of hypertensive disorders of pregnancy and the clear evidence that this is a risk factor for future CVD, more studies like this one are needed in order to help guide patient and provider education,” said Dr. Albright, who was not involved in the study.
“It is generally well reported within the ob.gyn. literature about the increased lifetime CVD risk related to hypertensive disorders of pregnancy and we, as ob.gyns., always ask about pregnancy history because of our specialty, which gives us the opportunity to counsel about future risks,” she said.
“Women’s health [including during pregnancy] has been undervalued and underresearched for a long time,” with limited focus on pregnancy-related issues until recently, Dr. Albright noted. “This is clear in the attitudes and education of the primary care providers in this study,” she said.
A major barrier to screening in clinical practice has been that the standard screening guidelines for CVD (for example, those published by the United States Preventive Services Taskforce) have not included pregnancy history, said Dr. Albright. “Subsequently, these questions are not asked during routine annual visits,” she said. Ideally, “we should be able to leverage the electronic medical record to prompt providers to view a previously recorded pregnancy history or to ask about pregnancy history as a routine part of CVD risk assessment, and, of course, additional education outside of ob.gyn. and cardiology is needed,” she said.
The clinical takeaway from the current study is that “every annual visit with a person who has been pregnant is an opportunity to ask about and document pregnancy history,” Dr. Albright said. “After the completion of childbearing, many patients no longer see an ob.gyn., so other providers need to feel comfortable asking about and counseling about risks related to pregnancy complications,” she added.
“It is clear that adverse pregnancy outcomes pose lifetime health risks,” said Dr. Albright. “We will continue to look into the mechanisms of this through research. However, right now the additional research that is needed is to determine the optimal screening and follow-up for patients with a history of hypertensive disorders of pregnancy, as well as to examine how existing CVD-screening algorithms can be modified to include adverse pregnancy outcomes,” she emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Albright had no financial conflicts to disclose.
Nearly three-quarters of clinicians reported screening patients for hypertensive disorders of pregnancy, but only one-quarter comprehensively identified cardiovascular risk, based on survey data from approximately 1,500 clinicians in the United States.
Rates of hypertensive disorders of pregnancy have been on the rise in the United States for the past decade, and women with a history of these disorders require cardiovascular risk monitoring during the postpartum period and beyond, wrote Nicole D. Ford, PhD, of the Centers for Disease Control and Prevention, Atlanta, and colleagues. Specifically, the American College of Obstetricians and Gynecologists recommends cardiovascular risk evaluation and lifestyle modification for these individuals, the researchers said.
The most effective management of women with a history of hypertensive disorders of pregnancy will likely involve a team effort by primary care, ob.gyns., and cardiologists, but data on clinician screening and referrals are limited, they added.
In a study published in Obstetrics & Gynecology, the researchers reviewed data from a cross-sectional, web-based survey of clinicians practicing in the United States (Fall DocStyles 2020). The study population of 1,502 respondents with complete surveys included 1,000 primary care physicians, 251 ob.gyns., and 251 nurse practitioners or physician assistants. Approximately 60% of the respondents were male, and approximately 65% had been in practice for at least 10 years.
Overall, 73.6% of clinicians reported screening patients for a history of hypertensive disorders of pregnancy. The screening rates were highest among ob.gyns. (94.8%).
However, although 93.9% of clinicians overall correctly identified at least one potential risk associated with hypertensive disorders of pregnancy, only 24.8% correctly identified all cardiovascular risks associated with hypertensive disorders of pregnancy listed in the survey, the researchers noted.
Screening rates ranged from 49% to 91% for pregnant women, 34%-75% for postpartum women, 26%-61% for nonpregnant reproductive-age women, 20%-45% for perimenopausal or menopausal women, and 1%-4% for others outside of these categories.
The most often–cited barriers to referral were lack of patient follow-through (51.5%) and patient refusal (33.6%). To improve and facilitate referrals, respondents’ most frequent resource request was for more referral options (42.9%), followed by patient education materials (36.2%), and professional guidelines (34.1%).
In a multivariate analysis, primary care physicians were more than five times as likely to report not screening patients for hypertensive disorders of pregnancy (adjusted prevalence ratio, 5.54); nurse practitioners and physician assistants were more than seven times as likely (adjusted prevalence ratio, 7.42).
The researchers also found that clinicians who saw fewer than 80 patients per week were almost twice as likely not to screen for hypertensive disorders of pregnancy than those who saw 110 or more patients per week (adjusted prevalence ratio, 1.81).
“Beyond the immediate postpartum period, there is a lack of clear guidance on CVD [cardiovascular disease] evaluation and ongoing monitoring in women with history of hypertensive disorders of pregnancy,” the researchers wrote in their discussion. “Recognizing hypertensive disorders of pregnancy as a risk factor for CVD may allow clinicians to identify women requiring early evaluation and intervention,” they said.
The study findings were limited by several factors including potentially biased estimates of screening practices, and the potential for selection bias because of the convenience sample used to recruit survey participants, the researchers noted.
However, the results were strengthened by the inclusion of data from several clinician types and the relatively large sample size, and are consistent with those of previous studies, they said. Based on the findings, addressing barriers at both the patient and clinician level and increasing both patient and clinician education about the long-term risks of hypertensive disorders of pregnancy might increase cardiovascular screening and subsequent referrals, they concluded.
More education, improved screening tools needed
“Unfortunately, most CVD risk stratification scores such as the Framingham score do not include pregnancy complications, despite excellent evidence that pregnancy complications increase risk of CVD,” said Catherine M. Albright, MD, MS, of the University of Washington, Seattle, in an interview. “This is likely because these scores were developed primarily to screen for CVD risk in men. Given the rising incidence of hypertensive disorders of pregnancy and the clear evidence that this is a risk factor for future CVD, more studies like this one are needed in order to help guide patient and provider education,” said Dr. Albright, who was not involved in the study.
“It is generally well reported within the ob.gyn. literature about the increased lifetime CVD risk related to hypertensive disorders of pregnancy and we, as ob.gyns., always ask about pregnancy history because of our specialty, which gives us the opportunity to counsel about future risks,” she said.
“Women’s health [including during pregnancy] has been undervalued and underresearched for a long time,” with limited focus on pregnancy-related issues until recently, Dr. Albright noted. “This is clear in the attitudes and education of the primary care providers in this study,” she said.
A major barrier to screening in clinical practice has been that the standard screening guidelines for CVD (for example, those published by the United States Preventive Services Taskforce) have not included pregnancy history, said Dr. Albright. “Subsequently, these questions are not asked during routine annual visits,” she said. Ideally, “we should be able to leverage the electronic medical record to prompt providers to view a previously recorded pregnancy history or to ask about pregnancy history as a routine part of CVD risk assessment, and, of course, additional education outside of ob.gyn. and cardiology is needed,” she said.
The clinical takeaway from the current study is that “every annual visit with a person who has been pregnant is an opportunity to ask about and document pregnancy history,” Dr. Albright said. “After the completion of childbearing, many patients no longer see an ob.gyn., so other providers need to feel comfortable asking about and counseling about risks related to pregnancy complications,” she added.
“It is clear that adverse pregnancy outcomes pose lifetime health risks,” said Dr. Albright. “We will continue to look into the mechanisms of this through research. However, right now the additional research that is needed is to determine the optimal screening and follow-up for patients with a history of hypertensive disorders of pregnancy, as well as to examine how existing CVD-screening algorithms can be modified to include adverse pregnancy outcomes,” she emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Albright had no financial conflicts to disclose.
FROM OBSTETRICS & GYNECOLOGY
Three-parent IVF now legal in two countries
: the United Kingdom and Australia.
Australia’s senate passed a bill on March 30 amending pre-existing laws to allow the procedure in certain circumstances.
The goal of this procedure is to prevent genetic disorders caused by defective mitochondria, the power plants inside our cells that provide energy for normal growth and development. When mitochondria don’t produce any energy at all, the resulting genetic disorders are quickly fatal. When mitochondria make only a little energy, children can have severe illnesses and disabilities.
“The outcomes from this problem are really severe, and it’s highly likely that the baby will be very sick or die,” says Arthur Caplan, PhD, head of the division of medical ethics at the New York University Grossman School of Medicine.
Mitochondria have a little bit of DNA, and children inherit them from their mother. To avoid children inheriting this damaged genetic material, mitochondrial donation, also known as three-parent in vitro fertilization (IVF), takes the nucleus, which contains most of the DNA that makes us who we are, from an egg of the mother and puts it into a donated egg from a woman with healthy mitochondria.
The egg is then fertilized with sperm through IVF, and the resulting embryo has genetic material from two women and one man.
One ethical conundrum about mitochondrial donation is that any child conceived this way would inherit modified DNA and pass that along to their own children.
“I think it’s likely that we are going to go down this road to repair disease,” Dr. Caplan says. “I don’t think all genetic engineering of embryos is wrong, but we have to draw the line between enhancement versus treating disease.”
For couples who want a child that shares at least some of their own DNA, there are other ways to have a child without damaged mitochondria. One option would be genetic screening of their embryos to find healthy embryos without this defect, which would work for some women who have relatively few mitochondrial mutations. Another alternative is using a donor egg from a woman with healthy mitochondria.
Mitochondrial donation may appeal to couples who want their children to have a genetic connection to both parents, Dr. Caplan says. But prospective parents also need to be aware that this procedure is relatively new and, unlike egg donation, doesn’t have a long track record of success.
“It looks promising, but we don’t have the full safety picture yet, and we’re not going to start to get it for another decade or so,” Dr. Caplan cautions. “I do think it’s worth offering as one option, but you also have to get people to think about how important it is to have a biological child together and make sure that they understand that even if we try this technique, we don’t know the long-term outcomes for children yet.”
A version of this article first appeared on WebMD.com.
: the United Kingdom and Australia.
Australia’s senate passed a bill on March 30 amending pre-existing laws to allow the procedure in certain circumstances.
The goal of this procedure is to prevent genetic disorders caused by defective mitochondria, the power plants inside our cells that provide energy for normal growth and development. When mitochondria don’t produce any energy at all, the resulting genetic disorders are quickly fatal. When mitochondria make only a little energy, children can have severe illnesses and disabilities.
“The outcomes from this problem are really severe, and it’s highly likely that the baby will be very sick or die,” says Arthur Caplan, PhD, head of the division of medical ethics at the New York University Grossman School of Medicine.
Mitochondria have a little bit of DNA, and children inherit them from their mother. To avoid children inheriting this damaged genetic material, mitochondrial donation, also known as three-parent in vitro fertilization (IVF), takes the nucleus, which contains most of the DNA that makes us who we are, from an egg of the mother and puts it into a donated egg from a woman with healthy mitochondria.
The egg is then fertilized with sperm through IVF, and the resulting embryo has genetic material from two women and one man.
One ethical conundrum about mitochondrial donation is that any child conceived this way would inherit modified DNA and pass that along to their own children.
“I think it’s likely that we are going to go down this road to repair disease,” Dr. Caplan says. “I don’t think all genetic engineering of embryos is wrong, but we have to draw the line between enhancement versus treating disease.”
For couples who want a child that shares at least some of their own DNA, there are other ways to have a child without damaged mitochondria. One option would be genetic screening of their embryos to find healthy embryos without this defect, which would work for some women who have relatively few mitochondrial mutations. Another alternative is using a donor egg from a woman with healthy mitochondria.
Mitochondrial donation may appeal to couples who want their children to have a genetic connection to both parents, Dr. Caplan says. But prospective parents also need to be aware that this procedure is relatively new and, unlike egg donation, doesn’t have a long track record of success.
“It looks promising, but we don’t have the full safety picture yet, and we’re not going to start to get it for another decade or so,” Dr. Caplan cautions. “I do think it’s worth offering as one option, but you also have to get people to think about how important it is to have a biological child together and make sure that they understand that even if we try this technique, we don’t know the long-term outcomes for children yet.”
A version of this article first appeared on WebMD.com.
: the United Kingdom and Australia.
Australia’s senate passed a bill on March 30 amending pre-existing laws to allow the procedure in certain circumstances.
The goal of this procedure is to prevent genetic disorders caused by defective mitochondria, the power plants inside our cells that provide energy for normal growth and development. When mitochondria don’t produce any energy at all, the resulting genetic disorders are quickly fatal. When mitochondria make only a little energy, children can have severe illnesses and disabilities.
“The outcomes from this problem are really severe, and it’s highly likely that the baby will be very sick or die,” says Arthur Caplan, PhD, head of the division of medical ethics at the New York University Grossman School of Medicine.
Mitochondria have a little bit of DNA, and children inherit them from their mother. To avoid children inheriting this damaged genetic material, mitochondrial donation, also known as three-parent in vitro fertilization (IVF), takes the nucleus, which contains most of the DNA that makes us who we are, from an egg of the mother and puts it into a donated egg from a woman with healthy mitochondria.
The egg is then fertilized with sperm through IVF, and the resulting embryo has genetic material from two women and one man.
One ethical conundrum about mitochondrial donation is that any child conceived this way would inherit modified DNA and pass that along to their own children.
“I think it’s likely that we are going to go down this road to repair disease,” Dr. Caplan says. “I don’t think all genetic engineering of embryos is wrong, but we have to draw the line between enhancement versus treating disease.”
For couples who want a child that shares at least some of their own DNA, there are other ways to have a child without damaged mitochondria. One option would be genetic screening of their embryos to find healthy embryos without this defect, which would work for some women who have relatively few mitochondrial mutations. Another alternative is using a donor egg from a woman with healthy mitochondria.
Mitochondrial donation may appeal to couples who want their children to have a genetic connection to both parents, Dr. Caplan says. But prospective parents also need to be aware that this procedure is relatively new and, unlike egg donation, doesn’t have a long track record of success.
“It looks promising, but we don’t have the full safety picture yet, and we’re not going to start to get it for another decade or so,” Dr. Caplan cautions. “I do think it’s worth offering as one option, but you also have to get people to think about how important it is to have a biological child together and make sure that they understand that even if we try this technique, we don’t know the long-term outcomes for children yet.”
A version of this article first appeared on WebMD.com.
Transvaginal mesh, native tissue repair have similar outcomes in 3-year trial
Transvaginal mesh was found to be safe and effective for patients with pelvic organ prolapse (POP) when compared with native tissue repair (NTR) in a 3-year trial.
Researchers, led by Bruce S. Kahn, MD, with the department of obstetrics & gynecology at Scripps Clinic in San Diego evaluated the two surgical treatment methods and published their findings in Obstetrics & Gynecology.
At completion of the 3-year follow-up in 2016, there were 401 participants in the transvaginal mesh group and 171 in the NTR group.
The prospective, nonrandomized, parallel-cohort, 27-site trial used a primary composite endpoint of anatomical success; subjective success (vaginal bulging); retreatment measures; and serious device-related or serious procedure-related adverse events.
The secondary endpoint was a composite outcome similar to the primary composite outcome but with anatomical success more stringently defined as POP quantification (POP-Q) point Ba < 0 and/or C < 0.
The secondary outcome was added to this trial because investigators had criticized the primary endpoint, set by the Food and Drug Administration, because it included anatomic outcome measures that were the same for inclusion criteria (POP-Q point Ba < 0 and/or C < 0.)
The secondary-outcome composite also included quality-of-life measures, mesh exposure, and mesh- and procedure-related complications.
Outcomes similar for both groups
The primary outcome demonstrated transvaginal mesh was not superior to native tissue repair (P =.056).
In the secondary outcome, superiority of transvaginal mesh over native tissue repair was shown (P =.009), with a propensity score–adjusted difference of 10.6% (90% confidence interval, 3.3%-17.9%) in favor of transvaginal mesh.
The authors noted that subjective success regarding vaginal bulging, which is important in patient satisfaction, was high and not statistically different between the two groups.
Additionally, transvaginal mesh repair was as safe as NTR regarding serious device-related and/or serious procedure-related side effects.
For the primary safety endpoint, 3.1% in the mesh group and 2.7% in the native tissue repair group experienced serious adverse events, demonstrating that mesh was noninferior to NTR.
Research results have been mixed
Unanswered questions surround surgical options for POP, which, the authors wrote, “affects 3%-6% of women based on symptoms and up to 50% of women based on vaginal examination.”
The FDA in 2011 issued 522 postmarket surveillance study orders for companies that market transvaginal mesh for POP.
Research results have varied and contentious debate has continued in the field. Some studies have shown that mesh has better subjective and objective outcomes than NTR in the anterior compartment. Others have found more complications with transvaginal mesh, such as mesh exposure and painful intercourse.
Complicating comparisons, early versions of the mesh used were larger and denser than today’s versions.
In this postmarket study, patients received either the Uphold LITE brand of transvaginal mesh or native tissue repair for surgical treatment of POP.
Expert: This study unlikely to change minds
In an accompanying editorial, John O.L. DeLancey, MD, professor of gynecology at the University of Michigan, Ann Arbor, pointed out that so far there’s been a lack of randomized trials that could answer whether mesh surgeries result in fewer symptoms or result in sufficient improvements in anatomy to justify their additional risk.
This study may not help with the decision. Dr. DeLancey wrote: “Will this study change the minds of either side of this debate? Probably not. The two sides are deeply entrenched in their positions.”
Two considerations are important in thinking about the issue, he said. Surgical outcomes for POP are “not as good as we would hope.” Also, many women have had serious complications with mesh operations.
He wrote: “Mesh litigation has resulted in more $8 billion in settlements, which is many times the $1 billion annual national cost of providing care for prolapse. Those of us who practice in referral centers have seen women with devastating problems, even though they probably represent a small fraction of cases.”
Dr. DeLancey highlighted some limitations of the study by Dr. Kahn and colleagues, especially regarding differences in the groups studied and the design of the study.
“For example,” he explained, “65% of individuals in the mesh-repair group had a prior hysterectomy as opposed to 30% in the native tissue repair group. In addition, some of the operations in the native tissue group are not typical choices; for example, hysteropexy was used for some patients and had a 47% failure rate.”
He said the all-or-nothing approach to surgical solutions may be clouding the debate – in other words mesh or no mesh for women as a group.
“Rather than asking whether mesh is better than no mesh, knowing which women (if any) stand to benefit from mesh is the critical question. We need to understand, for each woman, what structural failures exist so that we can target our interventions to correct them,” he wrote.
This study was sponsored by Boston Scientific. Dr. Kahn disclosed research support from Solaire, payments from AbbVie and Douchenay as a speaker, payments from Caldera and Cytuity (Boston Scientific) as a medical consultant, and payment from Johnson & Johnson as an expert witness. One coauthor disclosed that money was paid to her institution from Medtronic and Boston Scientific (both unrestricted educational grants for cadaveric lab). Another is chief medical officer at Axonics. One study coauthor receives research funding from Axonics and is a consultant for Group Dynamics, Medpace, and FirstThought. One coauthor received research support, is a consultant for Boston Scientific, and is an expert witness for Johnson & Johnson. Dr. DeLancey declared no relevant financial relationships.
Transvaginal mesh was found to be safe and effective for patients with pelvic organ prolapse (POP) when compared with native tissue repair (NTR) in a 3-year trial.
Researchers, led by Bruce S. Kahn, MD, with the department of obstetrics & gynecology at Scripps Clinic in San Diego evaluated the two surgical treatment methods and published their findings in Obstetrics & Gynecology.
At completion of the 3-year follow-up in 2016, there were 401 participants in the transvaginal mesh group and 171 in the NTR group.
The prospective, nonrandomized, parallel-cohort, 27-site trial used a primary composite endpoint of anatomical success; subjective success (vaginal bulging); retreatment measures; and serious device-related or serious procedure-related adverse events.
The secondary endpoint was a composite outcome similar to the primary composite outcome but with anatomical success more stringently defined as POP quantification (POP-Q) point Ba < 0 and/or C < 0.
The secondary outcome was added to this trial because investigators had criticized the primary endpoint, set by the Food and Drug Administration, because it included anatomic outcome measures that were the same for inclusion criteria (POP-Q point Ba < 0 and/or C < 0.)
The secondary-outcome composite also included quality-of-life measures, mesh exposure, and mesh- and procedure-related complications.
Outcomes similar for both groups
The primary outcome demonstrated transvaginal mesh was not superior to native tissue repair (P =.056).
In the secondary outcome, superiority of transvaginal mesh over native tissue repair was shown (P =.009), with a propensity score–adjusted difference of 10.6% (90% confidence interval, 3.3%-17.9%) in favor of transvaginal mesh.
The authors noted that subjective success regarding vaginal bulging, which is important in patient satisfaction, was high and not statistically different between the two groups.
Additionally, transvaginal mesh repair was as safe as NTR regarding serious device-related and/or serious procedure-related side effects.
For the primary safety endpoint, 3.1% in the mesh group and 2.7% in the native tissue repair group experienced serious adverse events, demonstrating that mesh was noninferior to NTR.
Research results have been mixed
Unanswered questions surround surgical options for POP, which, the authors wrote, “affects 3%-6% of women based on symptoms and up to 50% of women based on vaginal examination.”
The FDA in 2011 issued 522 postmarket surveillance study orders for companies that market transvaginal mesh for POP.
Research results have varied and contentious debate has continued in the field. Some studies have shown that mesh has better subjective and objective outcomes than NTR in the anterior compartment. Others have found more complications with transvaginal mesh, such as mesh exposure and painful intercourse.
Complicating comparisons, early versions of the mesh used were larger and denser than today’s versions.
In this postmarket study, patients received either the Uphold LITE brand of transvaginal mesh or native tissue repair for surgical treatment of POP.
Expert: This study unlikely to change minds
In an accompanying editorial, John O.L. DeLancey, MD, professor of gynecology at the University of Michigan, Ann Arbor, pointed out that so far there’s been a lack of randomized trials that could answer whether mesh surgeries result in fewer symptoms or result in sufficient improvements in anatomy to justify their additional risk.
This study may not help with the decision. Dr. DeLancey wrote: “Will this study change the minds of either side of this debate? Probably not. The two sides are deeply entrenched in their positions.”
Two considerations are important in thinking about the issue, he said. Surgical outcomes for POP are “not as good as we would hope.” Also, many women have had serious complications with mesh operations.
He wrote: “Mesh litigation has resulted in more $8 billion in settlements, which is many times the $1 billion annual national cost of providing care for prolapse. Those of us who practice in referral centers have seen women with devastating problems, even though they probably represent a small fraction of cases.”
Dr. DeLancey highlighted some limitations of the study by Dr. Kahn and colleagues, especially regarding differences in the groups studied and the design of the study.
“For example,” he explained, “65% of individuals in the mesh-repair group had a prior hysterectomy as opposed to 30% in the native tissue repair group. In addition, some of the operations in the native tissue group are not typical choices; for example, hysteropexy was used for some patients and had a 47% failure rate.”
He said the all-or-nothing approach to surgical solutions may be clouding the debate – in other words mesh or no mesh for women as a group.
“Rather than asking whether mesh is better than no mesh, knowing which women (if any) stand to benefit from mesh is the critical question. We need to understand, for each woman, what structural failures exist so that we can target our interventions to correct them,” he wrote.
This study was sponsored by Boston Scientific. Dr. Kahn disclosed research support from Solaire, payments from AbbVie and Douchenay as a speaker, payments from Caldera and Cytuity (Boston Scientific) as a medical consultant, and payment from Johnson & Johnson as an expert witness. One coauthor disclosed that money was paid to her institution from Medtronic and Boston Scientific (both unrestricted educational grants for cadaveric lab). Another is chief medical officer at Axonics. One study coauthor receives research funding from Axonics and is a consultant for Group Dynamics, Medpace, and FirstThought. One coauthor received research support, is a consultant for Boston Scientific, and is an expert witness for Johnson & Johnson. Dr. DeLancey declared no relevant financial relationships.
Transvaginal mesh was found to be safe and effective for patients with pelvic organ prolapse (POP) when compared with native tissue repair (NTR) in a 3-year trial.
Researchers, led by Bruce S. Kahn, MD, with the department of obstetrics & gynecology at Scripps Clinic in San Diego evaluated the two surgical treatment methods and published their findings in Obstetrics & Gynecology.
At completion of the 3-year follow-up in 2016, there were 401 participants in the transvaginal mesh group and 171 in the NTR group.
The prospective, nonrandomized, parallel-cohort, 27-site trial used a primary composite endpoint of anatomical success; subjective success (vaginal bulging); retreatment measures; and serious device-related or serious procedure-related adverse events.
The secondary endpoint was a composite outcome similar to the primary composite outcome but with anatomical success more stringently defined as POP quantification (POP-Q) point Ba < 0 and/or C < 0.
The secondary outcome was added to this trial because investigators had criticized the primary endpoint, set by the Food and Drug Administration, because it included anatomic outcome measures that were the same for inclusion criteria (POP-Q point Ba < 0 and/or C < 0.)
The secondary-outcome composite also included quality-of-life measures, mesh exposure, and mesh- and procedure-related complications.
Outcomes similar for both groups
The primary outcome demonstrated transvaginal mesh was not superior to native tissue repair (P =.056).
In the secondary outcome, superiority of transvaginal mesh over native tissue repair was shown (P =.009), with a propensity score–adjusted difference of 10.6% (90% confidence interval, 3.3%-17.9%) in favor of transvaginal mesh.
The authors noted that subjective success regarding vaginal bulging, which is important in patient satisfaction, was high and not statistically different between the two groups.
Additionally, transvaginal mesh repair was as safe as NTR regarding serious device-related and/or serious procedure-related side effects.
For the primary safety endpoint, 3.1% in the mesh group and 2.7% in the native tissue repair group experienced serious adverse events, demonstrating that mesh was noninferior to NTR.
Research results have been mixed
Unanswered questions surround surgical options for POP, which, the authors wrote, “affects 3%-6% of women based on symptoms and up to 50% of women based on vaginal examination.”
The FDA in 2011 issued 522 postmarket surveillance study orders for companies that market transvaginal mesh for POP.
Research results have varied and contentious debate has continued in the field. Some studies have shown that mesh has better subjective and objective outcomes than NTR in the anterior compartment. Others have found more complications with transvaginal mesh, such as mesh exposure and painful intercourse.
Complicating comparisons, early versions of the mesh used were larger and denser than today’s versions.
In this postmarket study, patients received either the Uphold LITE brand of transvaginal mesh or native tissue repair for surgical treatment of POP.
Expert: This study unlikely to change minds
In an accompanying editorial, John O.L. DeLancey, MD, professor of gynecology at the University of Michigan, Ann Arbor, pointed out that so far there’s been a lack of randomized trials that could answer whether mesh surgeries result in fewer symptoms or result in sufficient improvements in anatomy to justify their additional risk.
This study may not help with the decision. Dr. DeLancey wrote: “Will this study change the minds of either side of this debate? Probably not. The two sides are deeply entrenched in their positions.”
Two considerations are important in thinking about the issue, he said. Surgical outcomes for POP are “not as good as we would hope.” Also, many women have had serious complications with mesh operations.
He wrote: “Mesh litigation has resulted in more $8 billion in settlements, which is many times the $1 billion annual national cost of providing care for prolapse. Those of us who practice in referral centers have seen women with devastating problems, even though they probably represent a small fraction of cases.”
Dr. DeLancey highlighted some limitations of the study by Dr. Kahn and colleagues, especially regarding differences in the groups studied and the design of the study.
“For example,” he explained, “65% of individuals in the mesh-repair group had a prior hysterectomy as opposed to 30% in the native tissue repair group. In addition, some of the operations in the native tissue group are not typical choices; for example, hysteropexy was used for some patients and had a 47% failure rate.”
He said the all-or-nothing approach to surgical solutions may be clouding the debate – in other words mesh or no mesh for women as a group.
“Rather than asking whether mesh is better than no mesh, knowing which women (if any) stand to benefit from mesh is the critical question. We need to understand, for each woman, what structural failures exist so that we can target our interventions to correct them,” he wrote.
This study was sponsored by Boston Scientific. Dr. Kahn disclosed research support from Solaire, payments from AbbVie and Douchenay as a speaker, payments from Caldera and Cytuity (Boston Scientific) as a medical consultant, and payment from Johnson & Johnson as an expert witness. One coauthor disclosed that money was paid to her institution from Medtronic and Boston Scientific (both unrestricted educational grants for cadaveric lab). Another is chief medical officer at Axonics. One study coauthor receives research funding from Axonics and is a consultant for Group Dynamics, Medpace, and FirstThought. One coauthor received research support, is a consultant for Boston Scientific, and is an expert witness for Johnson & Johnson. Dr. DeLancey declared no relevant financial relationships.
FROM OBSTETRICS & GYNECOLOGY
Many state cancer plans drift from USPSTF breast cancer recommendations
When it comes to mammography recommendations state comprehensive cancer control (CCC) plans vary considerably and don’t always closely match the U.S. Preventive Services Task Force (USPSTF) recommendations for mammography frequency in women at average risk for breast cancer, according to a new cross-sectional study of CCC plans in all 50 states and the District of Columbia. The recommended age for initiation varied widely among CCC plans, and nearly one in five bore little resemblance at all to the USPTF recommendations.
According to the authors of the study, the variation among suggested ages of initiation may indicate a lack of consensus among state agencies. “For a recommendation tied to service coverage, this is a serious gap in public health policy,” they wrote in the study published online in JAMA Network Open.
CCC plans include goals, measurable objectives, and evidence-based strategies to combat cancers that are both common and preventable. They include input from multiple groups, frequently take 4-6 years to create, and should be updated regularly. Funding from the Centers for Disease Control and Prevention requires that the plans include data cancer screening prevalence rates and specific objectives and strategies.
Breast cancer is the most common cancer in women in the United States, and the second highest cause of cancer death. Regular, high-quality screening reduces breast cancer mortality by 25%-31% among women aged 50-69. As a result, the American Cancer Society, the USPSTF, the American College of Physicians, and the American Academy of Family Physicians have produced guidelines for mammography screening in women at average risk of breast cancer.
Although the benefits of screening are widely accepted, there is disagreement about the ages it should be initiated and ended. These inconsistencies stem from different evidence used to support recommendations, as well as different standards for benefits and harms from screening. Common concerns include overdiagnosis, false-positive results, and radiation damage from mammography.
Because these benefits and harms can vary based on age and values, there is an emphasis on shared decision-making between clinicians and women, especially those aged 40-49.
The most recent USPTF recommendation, issued in 2016, states that women aged 50-74 with average risk should be screened with mammography every 2 years. The choice of mammography in average-risk women under 50 should be approached on an individual basis. USPTF defines average risk as having no signs, symptoms, or previous diagnosis of breast cancer, and no family history or genetic causes for concern.
In the new study, researchers conducted a point-in-time evaluation of CCC plans from 50 states and the District of Columbia, between Nov. 1, 2019, and June 30, 2021.
Thirty-one percent of the plans included the complete USPTF recommendations of biennial mammography between ages 50 and 74; 51% included some, but not all of the USPTF recommendations; and 18% were not consistent at all with USPTF recommendations.
Overall, 59% of plans recommended initiation at age 50 and 37% at age 40, which is consistent with the older 2009 USPSTF recommendation. Eight percent of plans recommended starting at both 40 and 50, and 20% of plans had no recommended age of initiation.
Among the plans that were partially consistent with USPTF, 73% recommended initiation of mammography at age 40 and 31% at age 50. Eighty-five percent did not include an age to stop mammography; 15% did not include a recommended frequency; and 15% had an initiation age other than 40 or 50. Eighty-five percent of plans partially consistent with USPSTF included a recommendation that mammography should be conducted biennially.
The authors state that CCC plans could be improved by a unified emphasis on biennial screening of the general population of women aged 50-74, as well as clear differentiation between women at average risk and those at high risk, who could be screened at ages younger than 50 in consultation with their physician.
The study is limited by the fact that plans were reviewed a single time, while state CCC plans are updated with varying periodicity. The authors agree that implementation of population-based screening should be tailored to individual states and health care systems.
When it comes to mammography recommendations state comprehensive cancer control (CCC) plans vary considerably and don’t always closely match the U.S. Preventive Services Task Force (USPSTF) recommendations for mammography frequency in women at average risk for breast cancer, according to a new cross-sectional study of CCC plans in all 50 states and the District of Columbia. The recommended age for initiation varied widely among CCC plans, and nearly one in five bore little resemblance at all to the USPTF recommendations.
According to the authors of the study, the variation among suggested ages of initiation may indicate a lack of consensus among state agencies. “For a recommendation tied to service coverage, this is a serious gap in public health policy,” they wrote in the study published online in JAMA Network Open.
CCC plans include goals, measurable objectives, and evidence-based strategies to combat cancers that are both common and preventable. They include input from multiple groups, frequently take 4-6 years to create, and should be updated regularly. Funding from the Centers for Disease Control and Prevention requires that the plans include data cancer screening prevalence rates and specific objectives and strategies.
Breast cancer is the most common cancer in women in the United States, and the second highest cause of cancer death. Regular, high-quality screening reduces breast cancer mortality by 25%-31% among women aged 50-69. As a result, the American Cancer Society, the USPSTF, the American College of Physicians, and the American Academy of Family Physicians have produced guidelines for mammography screening in women at average risk of breast cancer.
Although the benefits of screening are widely accepted, there is disagreement about the ages it should be initiated and ended. These inconsistencies stem from different evidence used to support recommendations, as well as different standards for benefits and harms from screening. Common concerns include overdiagnosis, false-positive results, and radiation damage from mammography.
Because these benefits and harms can vary based on age and values, there is an emphasis on shared decision-making between clinicians and women, especially those aged 40-49.
The most recent USPTF recommendation, issued in 2016, states that women aged 50-74 with average risk should be screened with mammography every 2 years. The choice of mammography in average-risk women under 50 should be approached on an individual basis. USPTF defines average risk as having no signs, symptoms, or previous diagnosis of breast cancer, and no family history or genetic causes for concern.
In the new study, researchers conducted a point-in-time evaluation of CCC plans from 50 states and the District of Columbia, between Nov. 1, 2019, and June 30, 2021.
Thirty-one percent of the plans included the complete USPTF recommendations of biennial mammography between ages 50 and 74; 51% included some, but not all of the USPTF recommendations; and 18% were not consistent at all with USPTF recommendations.
Overall, 59% of plans recommended initiation at age 50 and 37% at age 40, which is consistent with the older 2009 USPSTF recommendation. Eight percent of plans recommended starting at both 40 and 50, and 20% of plans had no recommended age of initiation.
Among the plans that were partially consistent with USPTF, 73% recommended initiation of mammography at age 40 and 31% at age 50. Eighty-five percent did not include an age to stop mammography; 15% did not include a recommended frequency; and 15% had an initiation age other than 40 or 50. Eighty-five percent of plans partially consistent with USPSTF included a recommendation that mammography should be conducted biennially.
The authors state that CCC plans could be improved by a unified emphasis on biennial screening of the general population of women aged 50-74, as well as clear differentiation between women at average risk and those at high risk, who could be screened at ages younger than 50 in consultation with their physician.
The study is limited by the fact that plans were reviewed a single time, while state CCC plans are updated with varying periodicity. The authors agree that implementation of population-based screening should be tailored to individual states and health care systems.
When it comes to mammography recommendations state comprehensive cancer control (CCC) plans vary considerably and don’t always closely match the U.S. Preventive Services Task Force (USPSTF) recommendations for mammography frequency in women at average risk for breast cancer, according to a new cross-sectional study of CCC plans in all 50 states and the District of Columbia. The recommended age for initiation varied widely among CCC plans, and nearly one in five bore little resemblance at all to the USPTF recommendations.
According to the authors of the study, the variation among suggested ages of initiation may indicate a lack of consensus among state agencies. “For a recommendation tied to service coverage, this is a serious gap in public health policy,” they wrote in the study published online in JAMA Network Open.
CCC plans include goals, measurable objectives, and evidence-based strategies to combat cancers that are both common and preventable. They include input from multiple groups, frequently take 4-6 years to create, and should be updated regularly. Funding from the Centers for Disease Control and Prevention requires that the plans include data cancer screening prevalence rates and specific objectives and strategies.
Breast cancer is the most common cancer in women in the United States, and the second highest cause of cancer death. Regular, high-quality screening reduces breast cancer mortality by 25%-31% among women aged 50-69. As a result, the American Cancer Society, the USPSTF, the American College of Physicians, and the American Academy of Family Physicians have produced guidelines for mammography screening in women at average risk of breast cancer.
Although the benefits of screening are widely accepted, there is disagreement about the ages it should be initiated and ended. These inconsistencies stem from different evidence used to support recommendations, as well as different standards for benefits and harms from screening. Common concerns include overdiagnosis, false-positive results, and radiation damage from mammography.
Because these benefits and harms can vary based on age and values, there is an emphasis on shared decision-making between clinicians and women, especially those aged 40-49.
The most recent USPTF recommendation, issued in 2016, states that women aged 50-74 with average risk should be screened with mammography every 2 years. The choice of mammography in average-risk women under 50 should be approached on an individual basis. USPTF defines average risk as having no signs, symptoms, or previous diagnosis of breast cancer, and no family history or genetic causes for concern.
In the new study, researchers conducted a point-in-time evaluation of CCC plans from 50 states and the District of Columbia, between Nov. 1, 2019, and June 30, 2021.
Thirty-one percent of the plans included the complete USPTF recommendations of biennial mammography between ages 50 and 74; 51% included some, but not all of the USPTF recommendations; and 18% were not consistent at all with USPTF recommendations.
Overall, 59% of plans recommended initiation at age 50 and 37% at age 40, which is consistent with the older 2009 USPSTF recommendation. Eight percent of plans recommended starting at both 40 and 50, and 20% of plans had no recommended age of initiation.
Among the plans that were partially consistent with USPTF, 73% recommended initiation of mammography at age 40 and 31% at age 50. Eighty-five percent did not include an age to stop mammography; 15% did not include a recommended frequency; and 15% had an initiation age other than 40 or 50. Eighty-five percent of plans partially consistent with USPSTF included a recommendation that mammography should be conducted biennially.
The authors state that CCC plans could be improved by a unified emphasis on biennial screening of the general population of women aged 50-74, as well as clear differentiation between women at average risk and those at high risk, who could be screened at ages younger than 50 in consultation with their physician.
The study is limited by the fact that plans were reviewed a single time, while state CCC plans are updated with varying periodicity. The authors agree that implementation of population-based screening should be tailored to individual states and health care systems.
FROM JAMA NETWORK OPEN
Roe v. Wade reversal would rock ob.gyn. residencies
A decision by the U.S. Supreme Court to overturn Roe v. Wade could have far-reaching impacts on residents in ob.gyn. medical programs across the country, from learning simple procedures like ultrasounds to how to manage miscarriages.
On Monday, Politico published a leaked initial draft majority opinion written by Justice Samuel Alito, which declares Roe unconstitutional. The actual opinion is due at the end of June. If all the justices keep their positions for that vote, the 1973 court case that legalized abortion rights in the United States would no longer keep states from banning the procedure before viability, which is defined as 24-26 weeks into a pregnancy.
Loss of federal protections provided by Roe would leave a patchwork of states where providers would have to change reproductive health practices. Medical schools in those states would also have to curtail any abortion care training, experts told this news organization.
Constance Bohon, MD, co-chair of the Legislative and Liability Committee of the American College of Obstetricians and Gynecologists (ACOG), said: “As a profession, we are concerned about the possibility that there will be ob.gyn. residents who graduate without the experience and training needed to care for a patient who has complications from an abortion or a missed abortion. We are very concerned that without adequate training and access to care for these patients, the maternal mortality rate will rise.”
The loss of Roe “means that sadly, and realistically, many residents just won’t get the training, or we will need to think about mitigation strategies, like providing simulation training,” said Kavita Vinekar, MD, MPH, assistant clinical professor in the department of obstetrics and gynecology at the David Geffen School of Medicine at UCLA. “I can assure you that without access to abortion training, the quality of ob.gyn. training will absolutely suffer.”
Twenty-two states, including large swaths of the south and Midwest, already have laws that would go into effect immediately to ban abortion in the absence of Roe. Four states, including Florida, Montana, and Indiana, would likely ban abortion, according to an analysis from the Guttmacher Institute.
Almost 45% of ob.gyn. obstetrics and gynecology residency programs are in these states, totaling 2,638 residents as of 2022, according to a study published in April by researchers at the UCLA.
Most reproductive health care training lasts between 1-2 months during residency and includes instruction on ultrasounds, best practices in managing pregnancy complications, learning how to safely evacuate a uterus in the event of a stillbirth or miscarriage, and counseling for family planning options.
A glimpse at the future
The potential for a new reality is already playing out in Texas, where a law banning abortions after 6 weeks in pregnancy took effect last Sept. 1. The Ryan Program, a national initiative to teach abortion care to medical school residents, started a pilot to match resident physicians in Texas with hospital programs in other states without abortion restrictions.
The Ryan Program has matched over 40 residents since the law took effect, according to Jody Steinauer, MD, PhD, who oversees the Ryan Program at the Bixby Center for Global Reproductive Health at the University of California, San Francisco. Matching students is an arduous and timely process because students have hectic schedules, and state licensure and other regulatory issues must be worked out, she said.
“It will take a while to get systems in place to really support resident travel, and not every resident is going to be able to travel, so I’m just worried about the future impacts on patient care,” Dr. Steinauer told this news organization. “We have hundreds of residents who are not learning the skills they need, and they’re not going be able to provide evidence-based, patient-centered care.”
But Dr. Bohon expressed skepticism that the Ryan Program and other initiatives to provide training for travel residents would be sufficient. “Unfortunately, it is anticipated that this program will not be sufficient to provide training for all of the ob.gyn. residents who do not have access to abortion training because of the state where their residency is,” she said. “There are residency programs that have the capacity to have ob.gyns. get training at their programs, but there is a limited number of these positions available as well.”
She added that the Council on Resident Education in Obstetrics and Gynecology is actively pursuing options to provide abortion training for residents for whom such training is not available.
Spillover effect
Health care professionals in states without restrictive abortion laws will also see an increase in patients seeking abortion-related care. This spike already is happening in New Mexico, which borders not only Texas, but Oklahoma and Arizona – states that have severely restricted abortion rights.
“It’s difficult in the states that will be overloaded with additional abortion care to then incorporate learners on top of that,” said Eve Espey, MD, MPH, distinguished professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque. “Logistically, to fill that gap in care and train at the same time will be really challenging.”
Dr. Espey said the UNM Center for Reproductive Health has already seen patients from Texas who would likely fall under an exemption in that state’s law that allows abortions if a patient faces a medical emergency if the pregnancy continues.
“Providers are so afraid of these laws, and if they’re risk adverse, they’re always going to err on the side of not taking care of the patient if you think you can get in trouble,” Dr. Espey said, pointing to patients who presented with lethal fetal anomalies or ruptured fetal membranes early in pregnancy that might fall under the medical emergency exemption.
Dr. Steinauer with the Ryan Program said that many Texas residency program directors have told her they’ve heard from applicants who are giving low ranks to residency programs in the state.
“They’re saying, ‘if we want to be able to be trained as an ob.gyn., why would we want to go to a program with such significant restrictions?’” Dr. Steinauer said. “That is a real worry.”
Most ob.gyn. medical residency programs now provide built-in abortion training, which the Accreditation Council for Graduate Medical Education mandated in 1995. A small majority do not. According to a 2020 research letter from the ACOG, about 11% of ob.gyn. residents said their programs hypothetically offer training without a clear process to obtain it, and 8% reported that their programs offered no such training at all.
Residents can also opt out of abortion training, although students increasingly are choosing to only opt out of certain parts, such as training for abortions provided later in pregnancies, according to Dr. Steinauer.
A looming ‘pipeline problem’
There were about 3,550 abortion service clinicians in 2020, 72% of whom were ob.gyns., followed by family medicine doctors and advanced practice registered nurses, according to research published in March in JAMA Internal Medicine. This number is likely not an accurate tally, however, because the authors queried a national medical claims database that did not include self-pay patients.
Julia Strasser, DrPH, MPH, a senior research scientist at The George Washington University Milken Institute School of Public Health, Washington, D.C., who led the study, said states could create policies to broaden access to abortion for patients and training for medical residents.
Those include expanding laws to allow advanced practice clinicians to provide abortion care, allowing state Medicaid programs to opt-in for abortion payment, and establishing abortion care continuing medical education requirements for state boards of medicine for both ob.gyn. and primary care.
“Providers that practice in those restricted states will not only stop providing care there but also won’t be able to train the next generation of workforce to be able to provide that care,” Dr. Strasser said. “It’s a pipeline problem.”
States also could encourage their medical residency programs to not hold slots open for students who want to opt out of abortion care training. The University of Washington, Seattle, in 2000 eliminated slots it had been holding open for opt-out students because the state does not have restrictions on abortion.
As abortion becomes increasingly restricted across the country, Dr. Strasser said, for medical schools in “a state that continues to make abortion available, it’s essentially their duty to make that training available.”
A version of this article first appeared on Medscape.com.
A decision by the U.S. Supreme Court to overturn Roe v. Wade could have far-reaching impacts on residents in ob.gyn. medical programs across the country, from learning simple procedures like ultrasounds to how to manage miscarriages.
On Monday, Politico published a leaked initial draft majority opinion written by Justice Samuel Alito, which declares Roe unconstitutional. The actual opinion is due at the end of June. If all the justices keep their positions for that vote, the 1973 court case that legalized abortion rights in the United States would no longer keep states from banning the procedure before viability, which is defined as 24-26 weeks into a pregnancy.
Loss of federal protections provided by Roe would leave a patchwork of states where providers would have to change reproductive health practices. Medical schools in those states would also have to curtail any abortion care training, experts told this news organization.
Constance Bohon, MD, co-chair of the Legislative and Liability Committee of the American College of Obstetricians and Gynecologists (ACOG), said: “As a profession, we are concerned about the possibility that there will be ob.gyn. residents who graduate without the experience and training needed to care for a patient who has complications from an abortion or a missed abortion. We are very concerned that without adequate training and access to care for these patients, the maternal mortality rate will rise.”
The loss of Roe “means that sadly, and realistically, many residents just won’t get the training, or we will need to think about mitigation strategies, like providing simulation training,” said Kavita Vinekar, MD, MPH, assistant clinical professor in the department of obstetrics and gynecology at the David Geffen School of Medicine at UCLA. “I can assure you that without access to abortion training, the quality of ob.gyn. training will absolutely suffer.”
Twenty-two states, including large swaths of the south and Midwest, already have laws that would go into effect immediately to ban abortion in the absence of Roe. Four states, including Florida, Montana, and Indiana, would likely ban abortion, according to an analysis from the Guttmacher Institute.
Almost 45% of ob.gyn. obstetrics and gynecology residency programs are in these states, totaling 2,638 residents as of 2022, according to a study published in April by researchers at the UCLA.
Most reproductive health care training lasts between 1-2 months during residency and includes instruction on ultrasounds, best practices in managing pregnancy complications, learning how to safely evacuate a uterus in the event of a stillbirth or miscarriage, and counseling for family planning options.
A glimpse at the future
The potential for a new reality is already playing out in Texas, where a law banning abortions after 6 weeks in pregnancy took effect last Sept. 1. The Ryan Program, a national initiative to teach abortion care to medical school residents, started a pilot to match resident physicians in Texas with hospital programs in other states without abortion restrictions.
The Ryan Program has matched over 40 residents since the law took effect, according to Jody Steinauer, MD, PhD, who oversees the Ryan Program at the Bixby Center for Global Reproductive Health at the University of California, San Francisco. Matching students is an arduous and timely process because students have hectic schedules, and state licensure and other regulatory issues must be worked out, she said.
“It will take a while to get systems in place to really support resident travel, and not every resident is going to be able to travel, so I’m just worried about the future impacts on patient care,” Dr. Steinauer told this news organization. “We have hundreds of residents who are not learning the skills they need, and they’re not going be able to provide evidence-based, patient-centered care.”
But Dr. Bohon expressed skepticism that the Ryan Program and other initiatives to provide training for travel residents would be sufficient. “Unfortunately, it is anticipated that this program will not be sufficient to provide training for all of the ob.gyn. residents who do not have access to abortion training because of the state where their residency is,” she said. “There are residency programs that have the capacity to have ob.gyns. get training at their programs, but there is a limited number of these positions available as well.”
She added that the Council on Resident Education in Obstetrics and Gynecology is actively pursuing options to provide abortion training for residents for whom such training is not available.
Spillover effect
Health care professionals in states without restrictive abortion laws will also see an increase in patients seeking abortion-related care. This spike already is happening in New Mexico, which borders not only Texas, but Oklahoma and Arizona – states that have severely restricted abortion rights.
“It’s difficult in the states that will be overloaded with additional abortion care to then incorporate learners on top of that,” said Eve Espey, MD, MPH, distinguished professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque. “Logistically, to fill that gap in care and train at the same time will be really challenging.”
Dr. Espey said the UNM Center for Reproductive Health has already seen patients from Texas who would likely fall under an exemption in that state’s law that allows abortions if a patient faces a medical emergency if the pregnancy continues.
“Providers are so afraid of these laws, and if they’re risk adverse, they’re always going to err on the side of not taking care of the patient if you think you can get in trouble,” Dr. Espey said, pointing to patients who presented with lethal fetal anomalies or ruptured fetal membranes early in pregnancy that might fall under the medical emergency exemption.
Dr. Steinauer with the Ryan Program said that many Texas residency program directors have told her they’ve heard from applicants who are giving low ranks to residency programs in the state.
“They’re saying, ‘if we want to be able to be trained as an ob.gyn., why would we want to go to a program with such significant restrictions?’” Dr. Steinauer said. “That is a real worry.”
Most ob.gyn. medical residency programs now provide built-in abortion training, which the Accreditation Council for Graduate Medical Education mandated in 1995. A small majority do not. According to a 2020 research letter from the ACOG, about 11% of ob.gyn. residents said their programs hypothetically offer training without a clear process to obtain it, and 8% reported that their programs offered no such training at all.
Residents can also opt out of abortion training, although students increasingly are choosing to only opt out of certain parts, such as training for abortions provided later in pregnancies, according to Dr. Steinauer.
A looming ‘pipeline problem’
There were about 3,550 abortion service clinicians in 2020, 72% of whom were ob.gyns., followed by family medicine doctors and advanced practice registered nurses, according to research published in March in JAMA Internal Medicine. This number is likely not an accurate tally, however, because the authors queried a national medical claims database that did not include self-pay patients.
Julia Strasser, DrPH, MPH, a senior research scientist at The George Washington University Milken Institute School of Public Health, Washington, D.C., who led the study, said states could create policies to broaden access to abortion for patients and training for medical residents.
Those include expanding laws to allow advanced practice clinicians to provide abortion care, allowing state Medicaid programs to opt-in for abortion payment, and establishing abortion care continuing medical education requirements for state boards of medicine for both ob.gyn. and primary care.
“Providers that practice in those restricted states will not only stop providing care there but also won’t be able to train the next generation of workforce to be able to provide that care,” Dr. Strasser said. “It’s a pipeline problem.”
States also could encourage their medical residency programs to not hold slots open for students who want to opt out of abortion care training. The University of Washington, Seattle, in 2000 eliminated slots it had been holding open for opt-out students because the state does not have restrictions on abortion.
As abortion becomes increasingly restricted across the country, Dr. Strasser said, for medical schools in “a state that continues to make abortion available, it’s essentially their duty to make that training available.”
A version of this article first appeared on Medscape.com.
A decision by the U.S. Supreme Court to overturn Roe v. Wade could have far-reaching impacts on residents in ob.gyn. medical programs across the country, from learning simple procedures like ultrasounds to how to manage miscarriages.
On Monday, Politico published a leaked initial draft majority opinion written by Justice Samuel Alito, which declares Roe unconstitutional. The actual opinion is due at the end of June. If all the justices keep their positions for that vote, the 1973 court case that legalized abortion rights in the United States would no longer keep states from banning the procedure before viability, which is defined as 24-26 weeks into a pregnancy.
Loss of federal protections provided by Roe would leave a patchwork of states where providers would have to change reproductive health practices. Medical schools in those states would also have to curtail any abortion care training, experts told this news organization.
Constance Bohon, MD, co-chair of the Legislative and Liability Committee of the American College of Obstetricians and Gynecologists (ACOG), said: “As a profession, we are concerned about the possibility that there will be ob.gyn. residents who graduate without the experience and training needed to care for a patient who has complications from an abortion or a missed abortion. We are very concerned that without adequate training and access to care for these patients, the maternal mortality rate will rise.”
The loss of Roe “means that sadly, and realistically, many residents just won’t get the training, or we will need to think about mitigation strategies, like providing simulation training,” said Kavita Vinekar, MD, MPH, assistant clinical professor in the department of obstetrics and gynecology at the David Geffen School of Medicine at UCLA. “I can assure you that without access to abortion training, the quality of ob.gyn. training will absolutely suffer.”
Twenty-two states, including large swaths of the south and Midwest, already have laws that would go into effect immediately to ban abortion in the absence of Roe. Four states, including Florida, Montana, and Indiana, would likely ban abortion, according to an analysis from the Guttmacher Institute.
Almost 45% of ob.gyn. obstetrics and gynecology residency programs are in these states, totaling 2,638 residents as of 2022, according to a study published in April by researchers at the UCLA.
Most reproductive health care training lasts between 1-2 months during residency and includes instruction on ultrasounds, best practices in managing pregnancy complications, learning how to safely evacuate a uterus in the event of a stillbirth or miscarriage, and counseling for family planning options.
A glimpse at the future
The potential for a new reality is already playing out in Texas, where a law banning abortions after 6 weeks in pregnancy took effect last Sept. 1. The Ryan Program, a national initiative to teach abortion care to medical school residents, started a pilot to match resident physicians in Texas with hospital programs in other states without abortion restrictions.
The Ryan Program has matched over 40 residents since the law took effect, according to Jody Steinauer, MD, PhD, who oversees the Ryan Program at the Bixby Center for Global Reproductive Health at the University of California, San Francisco. Matching students is an arduous and timely process because students have hectic schedules, and state licensure and other regulatory issues must be worked out, she said.
“It will take a while to get systems in place to really support resident travel, and not every resident is going to be able to travel, so I’m just worried about the future impacts on patient care,” Dr. Steinauer told this news organization. “We have hundreds of residents who are not learning the skills they need, and they’re not going be able to provide evidence-based, patient-centered care.”
But Dr. Bohon expressed skepticism that the Ryan Program and other initiatives to provide training for travel residents would be sufficient. “Unfortunately, it is anticipated that this program will not be sufficient to provide training for all of the ob.gyn. residents who do not have access to abortion training because of the state where their residency is,” she said. “There are residency programs that have the capacity to have ob.gyns. get training at their programs, but there is a limited number of these positions available as well.”
She added that the Council on Resident Education in Obstetrics and Gynecology is actively pursuing options to provide abortion training for residents for whom such training is not available.
Spillover effect
Health care professionals in states without restrictive abortion laws will also see an increase in patients seeking abortion-related care. This spike already is happening in New Mexico, which borders not only Texas, but Oklahoma and Arizona – states that have severely restricted abortion rights.
“It’s difficult in the states that will be overloaded with additional abortion care to then incorporate learners on top of that,” said Eve Espey, MD, MPH, distinguished professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque. “Logistically, to fill that gap in care and train at the same time will be really challenging.”
Dr. Espey said the UNM Center for Reproductive Health has already seen patients from Texas who would likely fall under an exemption in that state’s law that allows abortions if a patient faces a medical emergency if the pregnancy continues.
“Providers are so afraid of these laws, and if they’re risk adverse, they’re always going to err on the side of not taking care of the patient if you think you can get in trouble,” Dr. Espey said, pointing to patients who presented with lethal fetal anomalies or ruptured fetal membranes early in pregnancy that might fall under the medical emergency exemption.
Dr. Steinauer with the Ryan Program said that many Texas residency program directors have told her they’ve heard from applicants who are giving low ranks to residency programs in the state.
“They’re saying, ‘if we want to be able to be trained as an ob.gyn., why would we want to go to a program with such significant restrictions?’” Dr. Steinauer said. “That is a real worry.”
Most ob.gyn. medical residency programs now provide built-in abortion training, which the Accreditation Council for Graduate Medical Education mandated in 1995. A small majority do not. According to a 2020 research letter from the ACOG, about 11% of ob.gyn. residents said their programs hypothetically offer training without a clear process to obtain it, and 8% reported that their programs offered no such training at all.
Residents can also opt out of abortion training, although students increasingly are choosing to only opt out of certain parts, such as training for abortions provided later in pregnancies, according to Dr. Steinauer.
A looming ‘pipeline problem’
There were about 3,550 abortion service clinicians in 2020, 72% of whom were ob.gyns., followed by family medicine doctors and advanced practice registered nurses, according to research published in March in JAMA Internal Medicine. This number is likely not an accurate tally, however, because the authors queried a national medical claims database that did not include self-pay patients.
Julia Strasser, DrPH, MPH, a senior research scientist at The George Washington University Milken Institute School of Public Health, Washington, D.C., who led the study, said states could create policies to broaden access to abortion for patients and training for medical residents.
Those include expanding laws to allow advanced practice clinicians to provide abortion care, allowing state Medicaid programs to opt-in for abortion payment, and establishing abortion care continuing medical education requirements for state boards of medicine for both ob.gyn. and primary care.
“Providers that practice in those restricted states will not only stop providing care there but also won’t be able to train the next generation of workforce to be able to provide that care,” Dr. Strasser said. “It’s a pipeline problem.”
States also could encourage their medical residency programs to not hold slots open for students who want to opt out of abortion care training. The University of Washington, Seattle, in 2000 eliminated slots it had been holding open for opt-out students because the state does not have restrictions on abortion.
As abortion becomes increasingly restricted across the country, Dr. Strasser said, for medical schools in “a state that continues to make abortion available, it’s essentially their duty to make that training available.”
A version of this article first appeared on Medscape.com.
CDC flags uptick in hypertensive disorders in pregnancy
Hypertensive disorders in pregnancy affect nearly 16% of women who give birth in U.S. hospitals and appear to be increasing, according to an April 29 report from the Centers for Disease Control and Prevention.
Older patients and Black women are substantially more likely to experience hypertension in pregnancy, the analysis found.
“Addressing hypertensive disorders in pregnancy is a key strategy in reducing inequities in pregnancy-related mortality,” study coauthor Wanda Barfield, MD, MPH, director of CDC’s Division of Reproductive Health, said in a statement.
Age, obesity, diabetes
The overall prevalence of hypertensive disorders in pregnancy increased from 13.3% in 2017 to 15.9% in 2019, the researchers reported in the CDC’s Morbidity and Mortality Weekly Report.
The uptick in hypertension coincides with trends toward older maternal age and higher rates of obesity and diabetes, which may explain the increase, they said.
For the study, Dr. Barfield and her colleagues analyzed nationally representative data from the National Inpatient Sample. They identified patients with a diagnosis of chronic hypertension, pregnancy-associated hypertension, or unspecified maternal hypertension during their hospitalization.
Among women aged 45-55 years, the prevalence of hypertension was 31%. Among those aged 35-44 years, it was 18%.
Hypertension diagnoses were more common in women who were Black (20.9%) or American Indian or Alaska Native (16.4%), than in other groups.
Of patients who died during delivery hospitalization, 31.6% had a hypertensive disorder.
The study shows a marked increase in hypertensive disorders over a relatively short time, according to Jane van Dis, MD, of the department of obstetrics and gynecology at the University of Rochester (N.Y.), who was not involved in the research. The phenomenon is consistent with her own experience, she said.
“When I am admitting patients, I’m oftentimes surprised when someone does not have a hypertensive disorder because I feel like the majority of patients these days do,” Dr. van Dis told this news organization.
Dr. Van Dis speculated that factors related to the environment, including air pollution and endocrine disrupters, could contribute to elevated rates of hypertensive disorders.
Natalie Bello, MD, MPH, director of hypertension research at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said rates of hypertension today could be even higher than in the study.
The CDC report relied on pre-COVID data, and the pandemic “increased disparities in health outcomes,” Dr. Bello said in an interview. “I’m worried that in actuality these numbers are an underestimation of the current state of hypertension in pregnancy.”
Dr. Bello, who has studied the need for better training in cardio-obstetrics, applauded Vice President Kamala Harris’ efforts to improve maternal health.
“The racial and geographic disparities that we continue to see in the field are disheartening but should be a call to action to redouble our work to improve maternal outcomes,” Dr. Bello told this news organization. “The good news is that a lot of morbidity related to hypertension can be avoided with timely diagnosis and treatment of blood pressure. However, we need to act to provide all pregnant persons with optimal care.”
Janet Wright, MD, director of CDC’s Division for Heart Disease and Stroke Prevention, said blood pressure home monitoring is a “great example” of a strategy clinicians can use to identify and manage patients with hypertension.
But one approach – self-monitoring blood pressure from home during pregnancy – did not significantly improve the health of pregnant women, according to new results from randomized trials in the United Kingdom.
Trial results published in JAMA show that blood pressure home-monitoring coupled to telemonitoring, as compared with usual care, did not significantly improve blood pressure control among patients with chronic or gestational hypertension.
A second trial published in JAMA that included patients at risk for preeclampsia found that self-monitoring with telemonitoring did not lead to significantly earlier diagnoses of hypertension.
“Individuals at risk for a hypertensive disorder of pregnancy, or with gestational or chronic hypertension, cannot be treated with a single approach,” Malavika Prabhu, MD, with Weill Cornell Medicine, New York, and coauthors write in an editorial accompanying the JAMA studies. Although the data suggest that self-monitoring of blood pressure is practical and tolerated, “More research is needed to determine optimal, high-value, equitable approaches to averting adverse perinatal outcomes associated with hypertensive disorders of pregnancy,” they write.
The CDC study authors and Dr. van Dis have disclosed no relevant financial relationships. Dr. Bello is funded by the National Institutes of Health to study blood pressure monitoring in pregnancy. The JAMA editorial authors disclosed university, government, and corporate grants and work with publishing companies.
A version of this article first appeared on Medscape.com.
Hypertensive disorders in pregnancy affect nearly 16% of women who give birth in U.S. hospitals and appear to be increasing, according to an April 29 report from the Centers for Disease Control and Prevention.
Older patients and Black women are substantially more likely to experience hypertension in pregnancy, the analysis found.
“Addressing hypertensive disorders in pregnancy is a key strategy in reducing inequities in pregnancy-related mortality,” study coauthor Wanda Barfield, MD, MPH, director of CDC’s Division of Reproductive Health, said in a statement.
Age, obesity, diabetes
The overall prevalence of hypertensive disorders in pregnancy increased from 13.3% in 2017 to 15.9% in 2019, the researchers reported in the CDC’s Morbidity and Mortality Weekly Report.
The uptick in hypertension coincides with trends toward older maternal age and higher rates of obesity and diabetes, which may explain the increase, they said.
For the study, Dr. Barfield and her colleagues analyzed nationally representative data from the National Inpatient Sample. They identified patients with a diagnosis of chronic hypertension, pregnancy-associated hypertension, or unspecified maternal hypertension during their hospitalization.
Among women aged 45-55 years, the prevalence of hypertension was 31%. Among those aged 35-44 years, it was 18%.
Hypertension diagnoses were more common in women who were Black (20.9%) or American Indian or Alaska Native (16.4%), than in other groups.
Of patients who died during delivery hospitalization, 31.6% had a hypertensive disorder.
The study shows a marked increase in hypertensive disorders over a relatively short time, according to Jane van Dis, MD, of the department of obstetrics and gynecology at the University of Rochester (N.Y.), who was not involved in the research. The phenomenon is consistent with her own experience, she said.
“When I am admitting patients, I’m oftentimes surprised when someone does not have a hypertensive disorder because I feel like the majority of patients these days do,” Dr. van Dis told this news organization.
Dr. Van Dis speculated that factors related to the environment, including air pollution and endocrine disrupters, could contribute to elevated rates of hypertensive disorders.
Natalie Bello, MD, MPH, director of hypertension research at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said rates of hypertension today could be even higher than in the study.
The CDC report relied on pre-COVID data, and the pandemic “increased disparities in health outcomes,” Dr. Bello said in an interview. “I’m worried that in actuality these numbers are an underestimation of the current state of hypertension in pregnancy.”
Dr. Bello, who has studied the need for better training in cardio-obstetrics, applauded Vice President Kamala Harris’ efforts to improve maternal health.
“The racial and geographic disparities that we continue to see in the field are disheartening but should be a call to action to redouble our work to improve maternal outcomes,” Dr. Bello told this news organization. “The good news is that a lot of morbidity related to hypertension can be avoided with timely diagnosis and treatment of blood pressure. However, we need to act to provide all pregnant persons with optimal care.”
Janet Wright, MD, director of CDC’s Division for Heart Disease and Stroke Prevention, said blood pressure home monitoring is a “great example” of a strategy clinicians can use to identify and manage patients with hypertension.
But one approach – self-monitoring blood pressure from home during pregnancy – did not significantly improve the health of pregnant women, according to new results from randomized trials in the United Kingdom.
Trial results published in JAMA show that blood pressure home-monitoring coupled to telemonitoring, as compared with usual care, did not significantly improve blood pressure control among patients with chronic or gestational hypertension.
A second trial published in JAMA that included patients at risk for preeclampsia found that self-monitoring with telemonitoring did not lead to significantly earlier diagnoses of hypertension.
“Individuals at risk for a hypertensive disorder of pregnancy, or with gestational or chronic hypertension, cannot be treated with a single approach,” Malavika Prabhu, MD, with Weill Cornell Medicine, New York, and coauthors write in an editorial accompanying the JAMA studies. Although the data suggest that self-monitoring of blood pressure is practical and tolerated, “More research is needed to determine optimal, high-value, equitable approaches to averting adverse perinatal outcomes associated with hypertensive disorders of pregnancy,” they write.
The CDC study authors and Dr. van Dis have disclosed no relevant financial relationships. Dr. Bello is funded by the National Institutes of Health to study blood pressure monitoring in pregnancy. The JAMA editorial authors disclosed university, government, and corporate grants and work with publishing companies.
A version of this article first appeared on Medscape.com.
Hypertensive disorders in pregnancy affect nearly 16% of women who give birth in U.S. hospitals and appear to be increasing, according to an April 29 report from the Centers for Disease Control and Prevention.
Older patients and Black women are substantially more likely to experience hypertension in pregnancy, the analysis found.
“Addressing hypertensive disorders in pregnancy is a key strategy in reducing inequities in pregnancy-related mortality,” study coauthor Wanda Barfield, MD, MPH, director of CDC’s Division of Reproductive Health, said in a statement.
Age, obesity, diabetes
The overall prevalence of hypertensive disorders in pregnancy increased from 13.3% in 2017 to 15.9% in 2019, the researchers reported in the CDC’s Morbidity and Mortality Weekly Report.
The uptick in hypertension coincides with trends toward older maternal age and higher rates of obesity and diabetes, which may explain the increase, they said.
For the study, Dr. Barfield and her colleagues analyzed nationally representative data from the National Inpatient Sample. They identified patients with a diagnosis of chronic hypertension, pregnancy-associated hypertension, or unspecified maternal hypertension during their hospitalization.
Among women aged 45-55 years, the prevalence of hypertension was 31%. Among those aged 35-44 years, it was 18%.
Hypertension diagnoses were more common in women who were Black (20.9%) or American Indian or Alaska Native (16.4%), than in other groups.
Of patients who died during delivery hospitalization, 31.6% had a hypertensive disorder.
The study shows a marked increase in hypertensive disorders over a relatively short time, according to Jane van Dis, MD, of the department of obstetrics and gynecology at the University of Rochester (N.Y.), who was not involved in the research. The phenomenon is consistent with her own experience, she said.
“When I am admitting patients, I’m oftentimes surprised when someone does not have a hypertensive disorder because I feel like the majority of patients these days do,” Dr. van Dis told this news organization.
Dr. Van Dis speculated that factors related to the environment, including air pollution and endocrine disrupters, could contribute to elevated rates of hypertensive disorders.
Natalie Bello, MD, MPH, director of hypertension research at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said rates of hypertension today could be even higher than in the study.
The CDC report relied on pre-COVID data, and the pandemic “increased disparities in health outcomes,” Dr. Bello said in an interview. “I’m worried that in actuality these numbers are an underestimation of the current state of hypertension in pregnancy.”
Dr. Bello, who has studied the need for better training in cardio-obstetrics, applauded Vice President Kamala Harris’ efforts to improve maternal health.
“The racial and geographic disparities that we continue to see in the field are disheartening but should be a call to action to redouble our work to improve maternal outcomes,” Dr. Bello told this news organization. “The good news is that a lot of morbidity related to hypertension can be avoided with timely diagnosis and treatment of blood pressure. However, we need to act to provide all pregnant persons with optimal care.”
Janet Wright, MD, director of CDC’s Division for Heart Disease and Stroke Prevention, said blood pressure home monitoring is a “great example” of a strategy clinicians can use to identify and manage patients with hypertension.
But one approach – self-monitoring blood pressure from home during pregnancy – did not significantly improve the health of pregnant women, according to new results from randomized trials in the United Kingdom.
Trial results published in JAMA show that blood pressure home-monitoring coupled to telemonitoring, as compared with usual care, did not significantly improve blood pressure control among patients with chronic or gestational hypertension.
A second trial published in JAMA that included patients at risk for preeclampsia found that self-monitoring with telemonitoring did not lead to significantly earlier diagnoses of hypertension.
“Individuals at risk for a hypertensive disorder of pregnancy, or with gestational or chronic hypertension, cannot be treated with a single approach,” Malavika Prabhu, MD, with Weill Cornell Medicine, New York, and coauthors write in an editorial accompanying the JAMA studies. Although the data suggest that self-monitoring of blood pressure is practical and tolerated, “More research is needed to determine optimal, high-value, equitable approaches to averting adverse perinatal outcomes associated with hypertensive disorders of pregnancy,” they write.
The CDC study authors and Dr. van Dis have disclosed no relevant financial relationships. Dr. Bello is funded by the National Institutes of Health to study blood pressure monitoring in pregnancy. The JAMA editorial authors disclosed university, government, and corporate grants and work with publishing companies.
A version of this article first appeared on Medscape.com.
Abortion politics lead to power struggles over family planning grants
BOZEMAN, Mont. – In a busy downtown coffee shop, a drawing of a ski lift with intrauterine devices for chairs draws the eyes of sleepy customers getting their morning underway with a caffeine jolt.
The flyer touts the services of Bridgercare, a nonprofit reproductive health clinic a few miles up the road. The clinic offers wellness exams, birth control, and LGBTQ+ services – and, starting in April, it oversees the state’s multimillion-dollar share of federal family planning program funding.
In March, Bridgercare beat out the state health department to become administrator of Montana’s $2.3 million Title X program, which helps pay for family planning and preventive health services. The organization applied for the grant because its leaders were concerned about a new state law that sought to restrict which local providers are funded.
What is happening in Montana is the latest example of an ongoing power struggle between nonprofits and conservative-leaning states over who receives federal family planning money. That has intensified in recent years as the Title X program has increasingly become entangled with the politics of abortion.
This year, the federal government set aside $257 million for family planning and preventive care. The providers that get that funding often serve families with low incomes, and Title X is one of the few federal programs in which people without legal permission to be in the United States can participate.
“The program permeates into communities that otherwise would be unreached by public health efforts,” said Rebecca Kreitzer, an associate professor of public policy at the University of North Carolina at Chapel Hill.
The Montana Department of Public Health and Human Services controlled the distribution of the state’s Title X funds for decades. Bridgercare sought the administrator role to circumvent a Republican-sponsored law passed last year that required the state to prioritize the money for local health departments and federally qualified health centers. That would have put the nonprofit – which doesn’t provide abortion procedures – and similar organizations at the bottom of the list. The law also banned clinics that perform abortions from receiving Title X funds from the state health department.
Bridgercare Executive Director Stephanie McDowell said the group applied for the grant to try to protect the program from decisions coming out of the state capitol. “Because of the politicization of Title X, we’re seeing how it’s run, swinging back and forth based on partisan leadership,” Ms. McDowell said.
A U.S. Department of Health & Human Services spokesperson, Tara Broido, didn’t answer a question about whether the agency intentionally awarded grants to nonprofits to avoid state politics. Instead, she said in a statement that applicants were evaluated in a competitive process by a panel of independent reviewers based on criteria to deliver high-quality, client-centered services.
Federal law prohibits the money from being used to perform abortions. But it can cover other services provided by groups that offer abortions – the largest and best-known by far is Planned Parenthood. In recent years, conservative politicians have tried to keep such providers from receiving Title X funding.
In some cases, contraception has entered the debate around which family planning services government should help fund. Some abortion opponents have raised concerns that long-lasting forms of birth control, such as IUDs, lead to abortions. Those claims are disputed by reproductive health experts.
In 2019, the Trump administration introduced several new rules for Title X, including disqualifying from receiving the funding family planning clinics that also offered abortion services or referrals. Many clinics across the nation left the program instead of conforming to the rules. Simultaneously, the spread of COVID-19 interrupted routine care. The number of patients served by Title X plummeted.
The Biden administration reversed most of those rules, including allowing providers with abortion services back into the Title X program. States also try to influence the funding’s reach, either through legislation or budget rules.
The current Title X funding cycle is 5 years, and the amount of money available each year could shift based on the state’s network of providers or federal budget changes. Jon Ebelt, a spokesperson for the Montana Department of Public Health and Human Services, didn’t answer when asked whether the state planned to reapply to administer the funding in 2027. He said the department was disappointed with the Biden administration’s “refusal” to renew the state’s funding.
“We recognize, however, that recent proabortion federal rule changes have distorted Title X and conflict with Montana law,” he said.
Conservative states have been tangling with nonprofits and the federal government over Title X funding for more than a decade. In 2011, during the Obama administration, Texas whittled down the state’s family planning spending and prioritized sending the federal money to general primary care providers over reproductive health clinics. As a result, 25% of family planning clinics in Texas closed. In 2013, a nonprofit now called Every Body Texas joined the competition to distribute the state’s Title X dollars and won.
“Filling and rebuilding those holes have taken this last decade, essentially,” said Berna Mason, director of service delivery improvement for Every Body Texas.
In 2019, the governor of Nebraska proposed a budget that would have prohibited the money from going to any organization that provided abortions or referred patients for abortions outside of an emergency. It also would have required that funding recipients be legally and financially separate from such clinics, a restriction that would have gone further than the Trump administration’s rules. Afterward, a family planning council won the right to administer Title X money.
In 2017, the nonprofit Arizona Family Health Partnership lost its status as that state’s only Title X administrator when the state health department was given 25% of the funding to deliver to providers. That came after Arizona lawmakers ordered the department to apply for the funds and distribute them first to state- or county-owned clinics, with the remaining money going to primary care facilities. The change was backed by groups that were opposed to abortion, and reproductive health care providers saw it as an attempt to weaken clinics that offer abortion services.
However, the state left nearly all the money it received untouched, and although it’s still required by law to apply for Title X funding, it hasn’t received a portion of the grant since.
Bré Thomas, CEO of Arizona Family Health Partnership, said that, even though the nonprofit is the sole administrator of the Title X funding again, the threat remains that some or all could be taken away because of politics. “We’re at the will of who’s in charge,” Ms. Thomas said.
Nonprofits say they have an advantage over state agencies in expanding services because they have more flexibility in fundraising and fewer administrative hurdles.
In April, Mississippi nonprofit Converge took over administration of Title X funds, a role the state had held for decades. The organization’s founders said they weren’t worried that conservative politicians would restrict access to services but simply believed they could do a better job. “Service quality was very low, and it was very hard to get appointments,” said cofounder Danielle Lampton.
A Mississippi State Department of Health spokesperson, Liz Sharlot, said the agency looks forward to working with Converge.
In Montana, Bridgercare plans to restore funding to Planned Parenthood clinics that have been cut off from the program since 2019, recruit more health centers to participate, and expand the program’s reach in rural, frontier, and tribal communities using telehealth services, Ms. McDowell said.
The organization’s goal is to increase the number of patients benefiting from the federal program by at least 10% in each year of the 5-year grant cycle. The clinic also plans to apply to keep its Title X role beyond this grant.
“In 5 years, our grant application should be a clear front-runner for funding,” she said. “It’s less about ‘How do we beat someone in 5 years?’ And more about ‘How do we grow this program to serve patients?’”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
BOZEMAN, Mont. – In a busy downtown coffee shop, a drawing of a ski lift with intrauterine devices for chairs draws the eyes of sleepy customers getting their morning underway with a caffeine jolt.
The flyer touts the services of Bridgercare, a nonprofit reproductive health clinic a few miles up the road. The clinic offers wellness exams, birth control, and LGBTQ+ services – and, starting in April, it oversees the state’s multimillion-dollar share of federal family planning program funding.
In March, Bridgercare beat out the state health department to become administrator of Montana’s $2.3 million Title X program, which helps pay for family planning and preventive health services. The organization applied for the grant because its leaders were concerned about a new state law that sought to restrict which local providers are funded.
What is happening in Montana is the latest example of an ongoing power struggle between nonprofits and conservative-leaning states over who receives federal family planning money. That has intensified in recent years as the Title X program has increasingly become entangled with the politics of abortion.
This year, the federal government set aside $257 million for family planning and preventive care. The providers that get that funding often serve families with low incomes, and Title X is one of the few federal programs in which people without legal permission to be in the United States can participate.
“The program permeates into communities that otherwise would be unreached by public health efforts,” said Rebecca Kreitzer, an associate professor of public policy at the University of North Carolina at Chapel Hill.
The Montana Department of Public Health and Human Services controlled the distribution of the state’s Title X funds for decades. Bridgercare sought the administrator role to circumvent a Republican-sponsored law passed last year that required the state to prioritize the money for local health departments and federally qualified health centers. That would have put the nonprofit – which doesn’t provide abortion procedures – and similar organizations at the bottom of the list. The law also banned clinics that perform abortions from receiving Title X funds from the state health department.
Bridgercare Executive Director Stephanie McDowell said the group applied for the grant to try to protect the program from decisions coming out of the state capitol. “Because of the politicization of Title X, we’re seeing how it’s run, swinging back and forth based on partisan leadership,” Ms. McDowell said.
A U.S. Department of Health & Human Services spokesperson, Tara Broido, didn’t answer a question about whether the agency intentionally awarded grants to nonprofits to avoid state politics. Instead, she said in a statement that applicants were evaluated in a competitive process by a panel of independent reviewers based on criteria to deliver high-quality, client-centered services.
Federal law prohibits the money from being used to perform abortions. But it can cover other services provided by groups that offer abortions – the largest and best-known by far is Planned Parenthood. In recent years, conservative politicians have tried to keep such providers from receiving Title X funding.
In some cases, contraception has entered the debate around which family planning services government should help fund. Some abortion opponents have raised concerns that long-lasting forms of birth control, such as IUDs, lead to abortions. Those claims are disputed by reproductive health experts.
In 2019, the Trump administration introduced several new rules for Title X, including disqualifying from receiving the funding family planning clinics that also offered abortion services or referrals. Many clinics across the nation left the program instead of conforming to the rules. Simultaneously, the spread of COVID-19 interrupted routine care. The number of patients served by Title X plummeted.
The Biden administration reversed most of those rules, including allowing providers with abortion services back into the Title X program. States also try to influence the funding’s reach, either through legislation or budget rules.
The current Title X funding cycle is 5 years, and the amount of money available each year could shift based on the state’s network of providers or federal budget changes. Jon Ebelt, a spokesperson for the Montana Department of Public Health and Human Services, didn’t answer when asked whether the state planned to reapply to administer the funding in 2027. He said the department was disappointed with the Biden administration’s “refusal” to renew the state’s funding.
“We recognize, however, that recent proabortion federal rule changes have distorted Title X and conflict with Montana law,” he said.
Conservative states have been tangling with nonprofits and the federal government over Title X funding for more than a decade. In 2011, during the Obama administration, Texas whittled down the state’s family planning spending and prioritized sending the federal money to general primary care providers over reproductive health clinics. As a result, 25% of family planning clinics in Texas closed. In 2013, a nonprofit now called Every Body Texas joined the competition to distribute the state’s Title X dollars and won.
“Filling and rebuilding those holes have taken this last decade, essentially,” said Berna Mason, director of service delivery improvement for Every Body Texas.
In 2019, the governor of Nebraska proposed a budget that would have prohibited the money from going to any organization that provided abortions or referred patients for abortions outside of an emergency. It also would have required that funding recipients be legally and financially separate from such clinics, a restriction that would have gone further than the Trump administration’s rules. Afterward, a family planning council won the right to administer Title X money.
In 2017, the nonprofit Arizona Family Health Partnership lost its status as that state’s only Title X administrator when the state health department was given 25% of the funding to deliver to providers. That came after Arizona lawmakers ordered the department to apply for the funds and distribute them first to state- or county-owned clinics, with the remaining money going to primary care facilities. The change was backed by groups that were opposed to abortion, and reproductive health care providers saw it as an attempt to weaken clinics that offer abortion services.
However, the state left nearly all the money it received untouched, and although it’s still required by law to apply for Title X funding, it hasn’t received a portion of the grant since.
Bré Thomas, CEO of Arizona Family Health Partnership, said that, even though the nonprofit is the sole administrator of the Title X funding again, the threat remains that some or all could be taken away because of politics. “We’re at the will of who’s in charge,” Ms. Thomas said.
Nonprofits say they have an advantage over state agencies in expanding services because they have more flexibility in fundraising and fewer administrative hurdles.
In April, Mississippi nonprofit Converge took over administration of Title X funds, a role the state had held for decades. The organization’s founders said they weren’t worried that conservative politicians would restrict access to services but simply believed they could do a better job. “Service quality was very low, and it was very hard to get appointments,” said cofounder Danielle Lampton.
A Mississippi State Department of Health spokesperson, Liz Sharlot, said the agency looks forward to working with Converge.
In Montana, Bridgercare plans to restore funding to Planned Parenthood clinics that have been cut off from the program since 2019, recruit more health centers to participate, and expand the program’s reach in rural, frontier, and tribal communities using telehealth services, Ms. McDowell said.
The organization’s goal is to increase the number of patients benefiting from the federal program by at least 10% in each year of the 5-year grant cycle. The clinic also plans to apply to keep its Title X role beyond this grant.
“In 5 years, our grant application should be a clear front-runner for funding,” she said. “It’s less about ‘How do we beat someone in 5 years?’ And more about ‘How do we grow this program to serve patients?’”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
BOZEMAN, Mont. – In a busy downtown coffee shop, a drawing of a ski lift with intrauterine devices for chairs draws the eyes of sleepy customers getting their morning underway with a caffeine jolt.
The flyer touts the services of Bridgercare, a nonprofit reproductive health clinic a few miles up the road. The clinic offers wellness exams, birth control, and LGBTQ+ services – and, starting in April, it oversees the state’s multimillion-dollar share of federal family planning program funding.
In March, Bridgercare beat out the state health department to become administrator of Montana’s $2.3 million Title X program, which helps pay for family planning and preventive health services. The organization applied for the grant because its leaders were concerned about a new state law that sought to restrict which local providers are funded.
What is happening in Montana is the latest example of an ongoing power struggle between nonprofits and conservative-leaning states over who receives federal family planning money. That has intensified in recent years as the Title X program has increasingly become entangled with the politics of abortion.
This year, the federal government set aside $257 million for family planning and preventive care. The providers that get that funding often serve families with low incomes, and Title X is one of the few federal programs in which people without legal permission to be in the United States can participate.
“The program permeates into communities that otherwise would be unreached by public health efforts,” said Rebecca Kreitzer, an associate professor of public policy at the University of North Carolina at Chapel Hill.
The Montana Department of Public Health and Human Services controlled the distribution of the state’s Title X funds for decades. Bridgercare sought the administrator role to circumvent a Republican-sponsored law passed last year that required the state to prioritize the money for local health departments and federally qualified health centers. That would have put the nonprofit – which doesn’t provide abortion procedures – and similar organizations at the bottom of the list. The law also banned clinics that perform abortions from receiving Title X funds from the state health department.
Bridgercare Executive Director Stephanie McDowell said the group applied for the grant to try to protect the program from decisions coming out of the state capitol. “Because of the politicization of Title X, we’re seeing how it’s run, swinging back and forth based on partisan leadership,” Ms. McDowell said.
A U.S. Department of Health & Human Services spokesperson, Tara Broido, didn’t answer a question about whether the agency intentionally awarded grants to nonprofits to avoid state politics. Instead, she said in a statement that applicants were evaluated in a competitive process by a panel of independent reviewers based on criteria to deliver high-quality, client-centered services.
Federal law prohibits the money from being used to perform abortions. But it can cover other services provided by groups that offer abortions – the largest and best-known by far is Planned Parenthood. In recent years, conservative politicians have tried to keep such providers from receiving Title X funding.
In some cases, contraception has entered the debate around which family planning services government should help fund. Some abortion opponents have raised concerns that long-lasting forms of birth control, such as IUDs, lead to abortions. Those claims are disputed by reproductive health experts.
In 2019, the Trump administration introduced several new rules for Title X, including disqualifying from receiving the funding family planning clinics that also offered abortion services or referrals. Many clinics across the nation left the program instead of conforming to the rules. Simultaneously, the spread of COVID-19 interrupted routine care. The number of patients served by Title X plummeted.
The Biden administration reversed most of those rules, including allowing providers with abortion services back into the Title X program. States also try to influence the funding’s reach, either through legislation or budget rules.
The current Title X funding cycle is 5 years, and the amount of money available each year could shift based on the state’s network of providers or federal budget changes. Jon Ebelt, a spokesperson for the Montana Department of Public Health and Human Services, didn’t answer when asked whether the state planned to reapply to administer the funding in 2027. He said the department was disappointed with the Biden administration’s “refusal” to renew the state’s funding.
“We recognize, however, that recent proabortion federal rule changes have distorted Title X and conflict with Montana law,” he said.
Conservative states have been tangling with nonprofits and the federal government over Title X funding for more than a decade. In 2011, during the Obama administration, Texas whittled down the state’s family planning spending and prioritized sending the federal money to general primary care providers over reproductive health clinics. As a result, 25% of family planning clinics in Texas closed. In 2013, a nonprofit now called Every Body Texas joined the competition to distribute the state’s Title X dollars and won.
“Filling and rebuilding those holes have taken this last decade, essentially,” said Berna Mason, director of service delivery improvement for Every Body Texas.
In 2019, the governor of Nebraska proposed a budget that would have prohibited the money from going to any organization that provided abortions or referred patients for abortions outside of an emergency. It also would have required that funding recipients be legally and financially separate from such clinics, a restriction that would have gone further than the Trump administration’s rules. Afterward, a family planning council won the right to administer Title X money.
In 2017, the nonprofit Arizona Family Health Partnership lost its status as that state’s only Title X administrator when the state health department was given 25% of the funding to deliver to providers. That came after Arizona lawmakers ordered the department to apply for the funds and distribute them first to state- or county-owned clinics, with the remaining money going to primary care facilities. The change was backed by groups that were opposed to abortion, and reproductive health care providers saw it as an attempt to weaken clinics that offer abortion services.
However, the state left nearly all the money it received untouched, and although it’s still required by law to apply for Title X funding, it hasn’t received a portion of the grant since.
Bré Thomas, CEO of Arizona Family Health Partnership, said that, even though the nonprofit is the sole administrator of the Title X funding again, the threat remains that some or all could be taken away because of politics. “We’re at the will of who’s in charge,” Ms. Thomas said.
Nonprofits say they have an advantage over state agencies in expanding services because they have more flexibility in fundraising and fewer administrative hurdles.
In April, Mississippi nonprofit Converge took over administration of Title X funds, a role the state had held for decades. The organization’s founders said they weren’t worried that conservative politicians would restrict access to services but simply believed they could do a better job. “Service quality was very low, and it was very hard to get appointments,” said cofounder Danielle Lampton.
A Mississippi State Department of Health spokesperson, Liz Sharlot, said the agency looks forward to working with Converge.
In Montana, Bridgercare plans to restore funding to Planned Parenthood clinics that have been cut off from the program since 2019, recruit more health centers to participate, and expand the program’s reach in rural, frontier, and tribal communities using telehealth services, Ms. McDowell said.
The organization’s goal is to increase the number of patients benefiting from the federal program by at least 10% in each year of the 5-year grant cycle. The clinic also plans to apply to keep its Title X role beyond this grant.
“In 5 years, our grant application should be a clear front-runner for funding,” she said. “It’s less about ‘How do we beat someone in 5 years?’ And more about ‘How do we grow this program to serve patients?’”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Maternal autoimmune diseases up risk of mental illness in children
Mental disorders were significantly more likely in children whose mothers had one of five common autoimmune diseases, a new study found.
Previous research has linked both maternal and paternal autoimmune diseases and specific mental disorders, such as attention-deficit/hyperactivity disorder (ADHD), but most of these studies focused on specific conditions in relatively small populations. The new study included data on more than 2 million births, making it one of the largest efforts to date to examine the association, according to the researchers, whose findings were published in JAMA Network Open.
Previous evidence of the possible association between certain maternal autoimmune diseases and mental disorders in offspring has been “scattered and limited,” which “hampered an overall understanding” of the link, Fei Li, MD, the corresponding author of the study, told this news organization.
Dr. Li, of Shanghai Jiao Tong University China, and colleagues reviewed data from a Danish registry cohort of singleton births with up to 38 years of follow-up. They explored associations between a range of maternal autoimmune diseases diagnosed before childbirth and the risks of mental disorders in children in early childhood through young adulthood.
The study population included 2,254,234 births and 38,916,359 person-years. Data on mental health were collected from the Psychiatric Central Research Register and the country’s National Patient Register. The median age of the children at the time of assessment was 16.7 years; approximately half were male.
A total of 50,863 children (2.26%) were born to mothers who had been diagnosed with autoimmune diseases before childbirth. During the follow-up period, 5,460 children of mothers with autoimmune diseases and 303,092 children of mothers without autoimmune diseases were diagnosed with a mental disorder (10.73% vs. 13.76%), according to the researchers.
The risk of being diagnosed with a mental disorder was significantly higher among children of mothers with any autoimmune disease (hazard ratio [HR,], 1.16), with an incidence of 9.38 vs. 7.91 per 1,000 person-years, the researchers reported.
The increased risk persisted when the results were classified by organ system, including connective tissue (HR, 1.11), endocrine (HR, 1.19), gastrointestinal (HR, 1.11), blood (HR, 1.10), nervous (HR, 1.17), and skin (HR, 1.19).
The five autoimmune diseases in mothers that were most commonly associated mental health disorders in children were type 1 diabetes, rheumatoid arthritis, systemic lupus erythematosus, multiple sclerosis, and psoriasis vulgaris.
The greatest risk for children of mothers with any autoimmune disease was observed for organic conditions such as delirium, (HR, 1.54), followed by obsessive-compulsive disorder (HR, 1.42), schizophrenia (HR, 1.54), and mood problems (HR, 1.12).
Children of mothers with any autoimmune disorder also had a significantly increased risk of autism (HR, 1.21), intellectual disability (HR, 1.19), and ADHD (HR, 1.19).
The results add to evidence that activation of the maternal immune system may drive changes in the brain and behavioral problems, which has been observed in animal studies, the researchers wrote.
Potential underlying mechanisms in need of more exploration include genetic risk factors, maternal transmission of autoantibodies to the fetus during pregnancy, and the increased risk of obstetric complications, such as preterm birth, for women with autoimmune disorders that could affect mental development in children, they added.
The study findings were limited by several factors, including the lack of data on potential exacerbation of autoimmune disease activity during pregnancy and its effect on the fetus, the researchers noted. Other limitations included potential detection bias, lack of data on mental disorders in adulthood, and potential changes in diagnostic criteria over the long study period.
The results were strengthened by the use of a population-based registry, the large sample size, and ability to consider a range of confounders, the researchers said.
“This study could help acquire a comprehensive compilation of the associations between maternal autoimmune disorders diagnosed before childbirth and offspring’s mental disorders from childhood through early adulthood,” Dr. Li said in an interview.
For clinicians, Dr. Li said, the findings suggest that the offspring of mothers with autoimmune diseases may benefit from long-term surveillance for mental health disorders.
“Further studies should provide more evidence on the detailed associations of specific maternal autoimmune diseases with a full spectrum of mental disorders in offspring, and more research on underlying mechanisms is needed as well,” she said.
Pay early attention
M. Susan Jay, MD, an adjunct professor of pediatrics at the Medical College of Wisconsin, Milwaukee, said previous efforts to examine the association between maternal autoimmunity were hampered by study design, small samples, and self-report of disease history – problems the new research avoids.
The large patient population allowed for detailed subgroup analysis of different conditions and outcomes. Another advantage was the availability of sociodemographic and clinical information, which allowed for the elimination of confounding factors, said Dr. Jay, who was not involved in the research.
“It would be prudent to follow children of mothers with autoimmune disorders before or during pregnancy for mental health issues, and if identified clinically, to offer psychological and developmental behavioral support options,” Dr. Jay added.
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mental disorders were significantly more likely in children whose mothers had one of five common autoimmune diseases, a new study found.
Previous research has linked both maternal and paternal autoimmune diseases and specific mental disorders, such as attention-deficit/hyperactivity disorder (ADHD), but most of these studies focused on specific conditions in relatively small populations. The new study included data on more than 2 million births, making it one of the largest efforts to date to examine the association, according to the researchers, whose findings were published in JAMA Network Open.
Previous evidence of the possible association between certain maternal autoimmune diseases and mental disorders in offspring has been “scattered and limited,” which “hampered an overall understanding” of the link, Fei Li, MD, the corresponding author of the study, told this news organization.
Dr. Li, of Shanghai Jiao Tong University China, and colleagues reviewed data from a Danish registry cohort of singleton births with up to 38 years of follow-up. They explored associations between a range of maternal autoimmune diseases diagnosed before childbirth and the risks of mental disorders in children in early childhood through young adulthood.
The study population included 2,254,234 births and 38,916,359 person-years. Data on mental health were collected from the Psychiatric Central Research Register and the country’s National Patient Register. The median age of the children at the time of assessment was 16.7 years; approximately half were male.
A total of 50,863 children (2.26%) were born to mothers who had been diagnosed with autoimmune diseases before childbirth. During the follow-up period, 5,460 children of mothers with autoimmune diseases and 303,092 children of mothers without autoimmune diseases were diagnosed with a mental disorder (10.73% vs. 13.76%), according to the researchers.
The risk of being diagnosed with a mental disorder was significantly higher among children of mothers with any autoimmune disease (hazard ratio [HR,], 1.16), with an incidence of 9.38 vs. 7.91 per 1,000 person-years, the researchers reported.
The increased risk persisted when the results were classified by organ system, including connective tissue (HR, 1.11), endocrine (HR, 1.19), gastrointestinal (HR, 1.11), blood (HR, 1.10), nervous (HR, 1.17), and skin (HR, 1.19).
The five autoimmune diseases in mothers that were most commonly associated mental health disorders in children were type 1 diabetes, rheumatoid arthritis, systemic lupus erythematosus, multiple sclerosis, and psoriasis vulgaris.
The greatest risk for children of mothers with any autoimmune disease was observed for organic conditions such as delirium, (HR, 1.54), followed by obsessive-compulsive disorder (HR, 1.42), schizophrenia (HR, 1.54), and mood problems (HR, 1.12).
Children of mothers with any autoimmune disorder also had a significantly increased risk of autism (HR, 1.21), intellectual disability (HR, 1.19), and ADHD (HR, 1.19).
The results add to evidence that activation of the maternal immune system may drive changes in the brain and behavioral problems, which has been observed in animal studies, the researchers wrote.
Potential underlying mechanisms in need of more exploration include genetic risk factors, maternal transmission of autoantibodies to the fetus during pregnancy, and the increased risk of obstetric complications, such as preterm birth, for women with autoimmune disorders that could affect mental development in children, they added.
The study findings were limited by several factors, including the lack of data on potential exacerbation of autoimmune disease activity during pregnancy and its effect on the fetus, the researchers noted. Other limitations included potential detection bias, lack of data on mental disorders in adulthood, and potential changes in diagnostic criteria over the long study period.
The results were strengthened by the use of a population-based registry, the large sample size, and ability to consider a range of confounders, the researchers said.
“This study could help acquire a comprehensive compilation of the associations between maternal autoimmune disorders diagnosed before childbirth and offspring’s mental disorders from childhood through early adulthood,” Dr. Li said in an interview.
For clinicians, Dr. Li said, the findings suggest that the offspring of mothers with autoimmune diseases may benefit from long-term surveillance for mental health disorders.
“Further studies should provide more evidence on the detailed associations of specific maternal autoimmune diseases with a full spectrum of mental disorders in offspring, and more research on underlying mechanisms is needed as well,” she said.
Pay early attention
M. Susan Jay, MD, an adjunct professor of pediatrics at the Medical College of Wisconsin, Milwaukee, said previous efforts to examine the association between maternal autoimmunity were hampered by study design, small samples, and self-report of disease history – problems the new research avoids.
The large patient population allowed for detailed subgroup analysis of different conditions and outcomes. Another advantage was the availability of sociodemographic and clinical information, which allowed for the elimination of confounding factors, said Dr. Jay, who was not involved in the research.
“It would be prudent to follow children of mothers with autoimmune disorders before or during pregnancy for mental health issues, and if identified clinically, to offer psychological and developmental behavioral support options,” Dr. Jay added.
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mental disorders were significantly more likely in children whose mothers had one of five common autoimmune diseases, a new study found.
Previous research has linked both maternal and paternal autoimmune diseases and specific mental disorders, such as attention-deficit/hyperactivity disorder (ADHD), but most of these studies focused on specific conditions in relatively small populations. The new study included data on more than 2 million births, making it one of the largest efforts to date to examine the association, according to the researchers, whose findings were published in JAMA Network Open.
Previous evidence of the possible association between certain maternal autoimmune diseases and mental disorders in offspring has been “scattered and limited,” which “hampered an overall understanding” of the link, Fei Li, MD, the corresponding author of the study, told this news organization.
Dr. Li, of Shanghai Jiao Tong University China, and colleagues reviewed data from a Danish registry cohort of singleton births with up to 38 years of follow-up. They explored associations between a range of maternal autoimmune diseases diagnosed before childbirth and the risks of mental disorders in children in early childhood through young adulthood.
The study population included 2,254,234 births and 38,916,359 person-years. Data on mental health were collected from the Psychiatric Central Research Register and the country’s National Patient Register. The median age of the children at the time of assessment was 16.7 years; approximately half were male.
A total of 50,863 children (2.26%) were born to mothers who had been diagnosed with autoimmune diseases before childbirth. During the follow-up period, 5,460 children of mothers with autoimmune diseases and 303,092 children of mothers without autoimmune diseases were diagnosed with a mental disorder (10.73% vs. 13.76%), according to the researchers.
The risk of being diagnosed with a mental disorder was significantly higher among children of mothers with any autoimmune disease (hazard ratio [HR,], 1.16), with an incidence of 9.38 vs. 7.91 per 1,000 person-years, the researchers reported.
The increased risk persisted when the results were classified by organ system, including connective tissue (HR, 1.11), endocrine (HR, 1.19), gastrointestinal (HR, 1.11), blood (HR, 1.10), nervous (HR, 1.17), and skin (HR, 1.19).
The five autoimmune diseases in mothers that were most commonly associated mental health disorders in children were type 1 diabetes, rheumatoid arthritis, systemic lupus erythematosus, multiple sclerosis, and psoriasis vulgaris.
The greatest risk for children of mothers with any autoimmune disease was observed for organic conditions such as delirium, (HR, 1.54), followed by obsessive-compulsive disorder (HR, 1.42), schizophrenia (HR, 1.54), and mood problems (HR, 1.12).
Children of mothers with any autoimmune disorder also had a significantly increased risk of autism (HR, 1.21), intellectual disability (HR, 1.19), and ADHD (HR, 1.19).
The results add to evidence that activation of the maternal immune system may drive changes in the brain and behavioral problems, which has been observed in animal studies, the researchers wrote.
Potential underlying mechanisms in need of more exploration include genetic risk factors, maternal transmission of autoantibodies to the fetus during pregnancy, and the increased risk of obstetric complications, such as preterm birth, for women with autoimmune disorders that could affect mental development in children, they added.
The study findings were limited by several factors, including the lack of data on potential exacerbation of autoimmune disease activity during pregnancy and its effect on the fetus, the researchers noted. Other limitations included potential detection bias, lack of data on mental disorders in adulthood, and potential changes in diagnostic criteria over the long study period.
The results were strengthened by the use of a population-based registry, the large sample size, and ability to consider a range of confounders, the researchers said.
“This study could help acquire a comprehensive compilation of the associations between maternal autoimmune disorders diagnosed before childbirth and offspring’s mental disorders from childhood through early adulthood,” Dr. Li said in an interview.
For clinicians, Dr. Li said, the findings suggest that the offspring of mothers with autoimmune diseases may benefit from long-term surveillance for mental health disorders.
“Further studies should provide more evidence on the detailed associations of specific maternal autoimmune diseases with a full spectrum of mental disorders in offspring, and more research on underlying mechanisms is needed as well,” she said.
Pay early attention
M. Susan Jay, MD, an adjunct professor of pediatrics at the Medical College of Wisconsin, Milwaukee, said previous efforts to examine the association between maternal autoimmunity were hampered by study design, small samples, and self-report of disease history – problems the new research avoids.
The large patient population allowed for detailed subgroup analysis of different conditions and outcomes. Another advantage was the availability of sociodemographic and clinical information, which allowed for the elimination of confounding factors, said Dr. Jay, who was not involved in the research.
“It would be prudent to follow children of mothers with autoimmune disorders before or during pregnancy for mental health issues, and if identified clinically, to offer psychological and developmental behavioral support options,” Dr. Jay added.
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.