User login
Menstrual cup use increases risk of IUD expulsion
Menstrual cup use is becoming increasingly popular as an option for menstrual hygiene among women in the United States, but little is known about the potential for IUD expulsion with menstrual cup use, Jill Long, MD, said at the 2020 virtual meeting of the American College of Obstetricians and Gynecologists.
Dr. Long cited a 2019 Internet survey of IUD users in which 11% reported menstrual cup use; reports of IUD expulsion were approximately three times higher among menstrual cup users. The results of the survey were limited by the self-reported responses and lack of data on when the expulsions occurred related to menstrual cup use.
Similar concerns about expulsion surfaced in an ongoing phase 3, randomized trial designed to support the marketing application of the Mona Lisa NT Cu380 Mini IUD, which is not currently approved in the United States.
“Nine months into the study, more expulsions were observed than expected, particularly among menstrual cup users,” said Dr. Long, medical officer and project officer for the Contraceptive Clinical Trials Network at the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The researchers then began to advise study participants to avoid menstrual cup use, and to collect data on use of menstrual hygiene products.
The preliminary study results reported by Dr. Long included 1,092 women assigned in a 4:1 ratio to receive the Mona Lisa NT Cu380 Mini and ParaGard, an IUD approved in the United States. Participants had follow-up visits at 6 weeks and at 3, 6, 12, 24, and 36 months after placement to verify the IUD position.
At 34 months, the overall rate of IUD expulsion was 9%. Of 277 women who reported menstrual cup use, the expulsion rate was 18%, compared with a 6% expulsion rates among the 704 women who reported tampon use.
Patient behavior persists despite advisory
Overall IUD expulsion was not significantly different among patients enrolled before and after the advisory against menstrual cup use (9.8% vs. 8.8%, respectively). In addition, menstrual cup use did not decrease after the implementation of the advisory (24% preadvisory vs. 28% post advisory).
The expulsion rates between menstrual cup users and nonusers were significantly different at both 12 months (14% vs. 5%) and 24 months (21% vs. 6%).
In addition, the researchers created a category of accidental self-removal, defined as the percent of expulsions occurring at the time the menstrual hygiene product was removed. Accidental self-removal was significantly higher among menstrual cup users, compared with tampon users (43% vs. 10%).
Study limitations included study blinding with regard to IUD type, parity, and age, so the impact of these factors on expulsion remains unclear, Dr. Long said. In addition, data on menstrual hygiene product use was collected retrospectively for the first 9 months, and no data were available on the impact on expulsion of combined use of menstrual cups and tampons.
Despite the apparent lack of impact of counseling, “women should be informed of the increased risk of expulsion if they choose to use menstrual cups concurrently with copper IUDs,” Dr. Long concluded.
More data to come from further analysis
During a question-and-answer session following the presentation, Dr. Long was asked whether suction might contribute to expulsion. “We advised subjects to break the seal on the menstrual cup prior to removal, but we did not see a decrease in expulsion rates with menstrual cup use after this advisory,” she said.
“The type of menstrual cup may be a factor,” she added. “We still need to analyze the data by the type of menstrual cup used as different cups have different degrees of suction.”
When asked about the potential role of coital activity on expulsion, Dr. Long said that the researchers had not yet reviewed coital activity logs to compare expulsion data with the timing of sexual activity.
Any increased pregnancy rates among menstrual cup users are “unlikely, but we don’t know for sure,” she added.
The current study also did not evaluate quantitative differences in bleeding, and participants are using bleeding diaries, Dr. Long noted. She added that participants are able to report problems with bleeding to the study sites, and that these are captured as adverse events.
The study was supported by a partnership between NICHD and FHI 360, a nonprofit human development organization based in North Carolina, with FHI 360 funding from the Bill and Melinda Gates Foundation. Dr. Long had no financial conflicts to disclose.
Menstrual cup use is becoming increasingly popular as an option for menstrual hygiene among women in the United States, but little is known about the potential for IUD expulsion with menstrual cup use, Jill Long, MD, said at the 2020 virtual meeting of the American College of Obstetricians and Gynecologists.
Dr. Long cited a 2019 Internet survey of IUD users in which 11% reported menstrual cup use; reports of IUD expulsion were approximately three times higher among menstrual cup users. The results of the survey were limited by the self-reported responses and lack of data on when the expulsions occurred related to menstrual cup use.
Similar concerns about expulsion surfaced in an ongoing phase 3, randomized trial designed to support the marketing application of the Mona Lisa NT Cu380 Mini IUD, which is not currently approved in the United States.
“Nine months into the study, more expulsions were observed than expected, particularly among menstrual cup users,” said Dr. Long, medical officer and project officer for the Contraceptive Clinical Trials Network at the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The researchers then began to advise study participants to avoid menstrual cup use, and to collect data on use of menstrual hygiene products.
The preliminary study results reported by Dr. Long included 1,092 women assigned in a 4:1 ratio to receive the Mona Lisa NT Cu380 Mini and ParaGard, an IUD approved in the United States. Participants had follow-up visits at 6 weeks and at 3, 6, 12, 24, and 36 months after placement to verify the IUD position.
At 34 months, the overall rate of IUD expulsion was 9%. Of 277 women who reported menstrual cup use, the expulsion rate was 18%, compared with a 6% expulsion rates among the 704 women who reported tampon use.
Patient behavior persists despite advisory
Overall IUD expulsion was not significantly different among patients enrolled before and after the advisory against menstrual cup use (9.8% vs. 8.8%, respectively). In addition, menstrual cup use did not decrease after the implementation of the advisory (24% preadvisory vs. 28% post advisory).
The expulsion rates between menstrual cup users and nonusers were significantly different at both 12 months (14% vs. 5%) and 24 months (21% vs. 6%).
In addition, the researchers created a category of accidental self-removal, defined as the percent of expulsions occurring at the time the menstrual hygiene product was removed. Accidental self-removal was significantly higher among menstrual cup users, compared with tampon users (43% vs. 10%).
Study limitations included study blinding with regard to IUD type, parity, and age, so the impact of these factors on expulsion remains unclear, Dr. Long said. In addition, data on menstrual hygiene product use was collected retrospectively for the first 9 months, and no data were available on the impact on expulsion of combined use of menstrual cups and tampons.
Despite the apparent lack of impact of counseling, “women should be informed of the increased risk of expulsion if they choose to use menstrual cups concurrently with copper IUDs,” Dr. Long concluded.
More data to come from further analysis
During a question-and-answer session following the presentation, Dr. Long was asked whether suction might contribute to expulsion. “We advised subjects to break the seal on the menstrual cup prior to removal, but we did not see a decrease in expulsion rates with menstrual cup use after this advisory,” she said.
“The type of menstrual cup may be a factor,” she added. “We still need to analyze the data by the type of menstrual cup used as different cups have different degrees of suction.”
When asked about the potential role of coital activity on expulsion, Dr. Long said that the researchers had not yet reviewed coital activity logs to compare expulsion data with the timing of sexual activity.
Any increased pregnancy rates among menstrual cup users are “unlikely, but we don’t know for sure,” she added.
The current study also did not evaluate quantitative differences in bleeding, and participants are using bleeding diaries, Dr. Long noted. She added that participants are able to report problems with bleeding to the study sites, and that these are captured as adverse events.
The study was supported by a partnership between NICHD and FHI 360, a nonprofit human development organization based in North Carolina, with FHI 360 funding from the Bill and Melinda Gates Foundation. Dr. Long had no financial conflicts to disclose.
Menstrual cup use is becoming increasingly popular as an option for menstrual hygiene among women in the United States, but little is known about the potential for IUD expulsion with menstrual cup use, Jill Long, MD, said at the 2020 virtual meeting of the American College of Obstetricians and Gynecologists.
Dr. Long cited a 2019 Internet survey of IUD users in which 11% reported menstrual cup use; reports of IUD expulsion were approximately three times higher among menstrual cup users. The results of the survey were limited by the self-reported responses and lack of data on when the expulsions occurred related to menstrual cup use.
Similar concerns about expulsion surfaced in an ongoing phase 3, randomized trial designed to support the marketing application of the Mona Lisa NT Cu380 Mini IUD, which is not currently approved in the United States.
“Nine months into the study, more expulsions were observed than expected, particularly among menstrual cup users,” said Dr. Long, medical officer and project officer for the Contraceptive Clinical Trials Network at the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The researchers then began to advise study participants to avoid menstrual cup use, and to collect data on use of menstrual hygiene products.
The preliminary study results reported by Dr. Long included 1,092 women assigned in a 4:1 ratio to receive the Mona Lisa NT Cu380 Mini and ParaGard, an IUD approved in the United States. Participants had follow-up visits at 6 weeks and at 3, 6, 12, 24, and 36 months after placement to verify the IUD position.
At 34 months, the overall rate of IUD expulsion was 9%. Of 277 women who reported menstrual cup use, the expulsion rate was 18%, compared with a 6% expulsion rates among the 704 women who reported tampon use.
Patient behavior persists despite advisory
Overall IUD expulsion was not significantly different among patients enrolled before and after the advisory against menstrual cup use (9.8% vs. 8.8%, respectively). In addition, menstrual cup use did not decrease after the implementation of the advisory (24% preadvisory vs. 28% post advisory).
The expulsion rates between menstrual cup users and nonusers were significantly different at both 12 months (14% vs. 5%) and 24 months (21% vs. 6%).
In addition, the researchers created a category of accidental self-removal, defined as the percent of expulsions occurring at the time the menstrual hygiene product was removed. Accidental self-removal was significantly higher among menstrual cup users, compared with tampon users (43% vs. 10%).
Study limitations included study blinding with regard to IUD type, parity, and age, so the impact of these factors on expulsion remains unclear, Dr. Long said. In addition, data on menstrual hygiene product use was collected retrospectively for the first 9 months, and no data were available on the impact on expulsion of combined use of menstrual cups and tampons.
Despite the apparent lack of impact of counseling, “women should be informed of the increased risk of expulsion if they choose to use menstrual cups concurrently with copper IUDs,” Dr. Long concluded.
More data to come from further analysis
During a question-and-answer session following the presentation, Dr. Long was asked whether suction might contribute to expulsion. “We advised subjects to break the seal on the menstrual cup prior to removal, but we did not see a decrease in expulsion rates with menstrual cup use after this advisory,” she said.
“The type of menstrual cup may be a factor,” she added. “We still need to analyze the data by the type of menstrual cup used as different cups have different degrees of suction.”
When asked about the potential role of coital activity on expulsion, Dr. Long said that the researchers had not yet reviewed coital activity logs to compare expulsion data with the timing of sexual activity.
Any increased pregnancy rates among menstrual cup users are “unlikely, but we don’t know for sure,” she added.
The current study also did not evaluate quantitative differences in bleeding, and participants are using bleeding diaries, Dr. Long noted. She added that participants are able to report problems with bleeding to the study sites, and that these are captured as adverse events.
The study was supported by a partnership between NICHD and FHI 360, a nonprofit human development organization based in North Carolina, with FHI 360 funding from the Bill and Melinda Gates Foundation. Dr. Long had no financial conflicts to disclose.
FROM ACOG 2020
New AHA scientific statement on menopause and CVD risk
Changes in hormones, body composition, lipids, and vascular health during the menopause transition can increase a woman’s chance of developing cardiovascular disease (CVD) after menopause, the American Heart Association said in a scientific statement.
“This statement aims to raise awareness of both healthcare providers and women about the menopause transition as a time of increasing heart disease risk,” Samar R. El Khoudary, PhD, MPH, who chaired the writing group, said in an interview.
“As such, it emphasizes the importance of monitoring women’s health during midlife and targeting this stage as a critical window for applying early intervention strategies that aim to maintain a healthy heart and reduce the risk of heart disease,” said Dr. El Khoudary, of the University of Pittsburgh.
The statement was published online Nov. 30 in Circulation.
Evolution in knowledge
During the past 20 years, knowledge of how menopause might contribute to CVD has evolved “dramatically,” Dr. El Khoudary noted. The accumulated data consistently point to the menopause transition as a time of change in heart health.
“Importantly,” she said, the latest AHA guidelines for CVD prevention in women, published in 2011, do not include data now available on the menopause transition as a time of increased CVD risk.
“As such, there is a compelling need to discuss the implications of the accumulating body of literature on this topic,” said Dr. El Khoudary.
The statement provides a contemporary synthesis of the existing data on menopause and how it relates to CVD, the leading cause of death of U.S. women.
Earlier age at natural menopause has generally been found to be a marker of greater CVD risk. Iatrogenically induced menopause (bilateral oophorectomy) during the premenopausal period is also associated with higher CVD risk, the data suggest.
Vasomotor symptoms are associated with worse levels of CVD risk factors and measures of subclinical atherosclerosis. Sleep disturbance has also been linked to greater risk for subclinical CVD and worse CV health indexes in women during midlife.
Increases in central/visceral fat and decreases in lean muscle mass are more pronounced during the menopause transition. This increased central adiposity is associated with increased risk for mortality, even among those with normal body mass index, the writing group found.
Increases in lipid levels (LDL cholesterol and apolipoprotein B), metabolic syndrome risk, and vascular remodeling at midlife are driven by the menopause transition more than aging, whereas increases in blood pressure, insulin level, and glucose level are likely more influenced by chronological aging, they reported.
Lifestyle interventions
The writing group noted that, because of the increase in overall life expectancy in the United States, a significant proportion of women will spend up to 40% of their lives after menopause.
Yet data suggest that only 7.2% of women transitioning to menopause are meeting physical activity guidelines and that fewer than 20% of those women are consistently maintaining a healthy diet.
Limited data from randomized, controlled trials suggest that a multidimensional lifestyle intervention during the menopause transition can prevent weight gain and reduce blood pressure and levels of triglycerides, blood glucose, and insulin and reduce the incidence of subclinical carotid atherosclerosis, they pointed out.
“Novel data” indicate a reversal in the associations of HDL cholesterol with CVD risk over the menopause transition, suggesting that higher HDL cholesterol levels may not consistently reflect good cardiovascular health in middle-aged women, the group noted.
There are also data suggesting that starting menopause hormone therapy when younger than 60 years or within 10 years of menopause is associated with reduced CVD risk.
The group said further research is needed into the cardiometabolic effects of menopause hormone therapy, including effects associated with form, route, and duration of administration, in women traversing menopause.
They also noted that data for the primary and secondary prevention of atherosclerotic CVD and improved survival with lipid-lowering interventions “remain elusive” for women and that further study is needed to develop evidence-based recommendations tailored specifically to women.
The research had no commercial funding. Dr. El Khoudary has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Changes in hormones, body composition, lipids, and vascular health during the menopause transition can increase a woman’s chance of developing cardiovascular disease (CVD) after menopause, the American Heart Association said in a scientific statement.
“This statement aims to raise awareness of both healthcare providers and women about the menopause transition as a time of increasing heart disease risk,” Samar R. El Khoudary, PhD, MPH, who chaired the writing group, said in an interview.
“As such, it emphasizes the importance of monitoring women’s health during midlife and targeting this stage as a critical window for applying early intervention strategies that aim to maintain a healthy heart and reduce the risk of heart disease,” said Dr. El Khoudary, of the University of Pittsburgh.
The statement was published online Nov. 30 in Circulation.
Evolution in knowledge
During the past 20 years, knowledge of how menopause might contribute to CVD has evolved “dramatically,” Dr. El Khoudary noted. The accumulated data consistently point to the menopause transition as a time of change in heart health.
“Importantly,” she said, the latest AHA guidelines for CVD prevention in women, published in 2011, do not include data now available on the menopause transition as a time of increased CVD risk.
“As such, there is a compelling need to discuss the implications of the accumulating body of literature on this topic,” said Dr. El Khoudary.
The statement provides a contemporary synthesis of the existing data on menopause and how it relates to CVD, the leading cause of death of U.S. women.
Earlier age at natural menopause has generally been found to be a marker of greater CVD risk. Iatrogenically induced menopause (bilateral oophorectomy) during the premenopausal period is also associated with higher CVD risk, the data suggest.
Vasomotor symptoms are associated with worse levels of CVD risk factors and measures of subclinical atherosclerosis. Sleep disturbance has also been linked to greater risk for subclinical CVD and worse CV health indexes in women during midlife.
Increases in central/visceral fat and decreases in lean muscle mass are more pronounced during the menopause transition. This increased central adiposity is associated with increased risk for mortality, even among those with normal body mass index, the writing group found.
Increases in lipid levels (LDL cholesterol and apolipoprotein B), metabolic syndrome risk, and vascular remodeling at midlife are driven by the menopause transition more than aging, whereas increases in blood pressure, insulin level, and glucose level are likely more influenced by chronological aging, they reported.
Lifestyle interventions
The writing group noted that, because of the increase in overall life expectancy in the United States, a significant proportion of women will spend up to 40% of their lives after menopause.
Yet data suggest that only 7.2% of women transitioning to menopause are meeting physical activity guidelines and that fewer than 20% of those women are consistently maintaining a healthy diet.
Limited data from randomized, controlled trials suggest that a multidimensional lifestyle intervention during the menopause transition can prevent weight gain and reduce blood pressure and levels of triglycerides, blood glucose, and insulin and reduce the incidence of subclinical carotid atherosclerosis, they pointed out.
“Novel data” indicate a reversal in the associations of HDL cholesterol with CVD risk over the menopause transition, suggesting that higher HDL cholesterol levels may not consistently reflect good cardiovascular health in middle-aged women, the group noted.
There are also data suggesting that starting menopause hormone therapy when younger than 60 years or within 10 years of menopause is associated with reduced CVD risk.
The group said further research is needed into the cardiometabolic effects of menopause hormone therapy, including effects associated with form, route, and duration of administration, in women traversing menopause.
They also noted that data for the primary and secondary prevention of atherosclerotic CVD and improved survival with lipid-lowering interventions “remain elusive” for women and that further study is needed to develop evidence-based recommendations tailored specifically to women.
The research had no commercial funding. Dr. El Khoudary has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Changes in hormones, body composition, lipids, and vascular health during the menopause transition can increase a woman’s chance of developing cardiovascular disease (CVD) after menopause, the American Heart Association said in a scientific statement.
“This statement aims to raise awareness of both healthcare providers and women about the menopause transition as a time of increasing heart disease risk,” Samar R. El Khoudary, PhD, MPH, who chaired the writing group, said in an interview.
“As such, it emphasizes the importance of monitoring women’s health during midlife and targeting this stage as a critical window for applying early intervention strategies that aim to maintain a healthy heart and reduce the risk of heart disease,” said Dr. El Khoudary, of the University of Pittsburgh.
The statement was published online Nov. 30 in Circulation.
Evolution in knowledge
During the past 20 years, knowledge of how menopause might contribute to CVD has evolved “dramatically,” Dr. El Khoudary noted. The accumulated data consistently point to the menopause transition as a time of change in heart health.
“Importantly,” she said, the latest AHA guidelines for CVD prevention in women, published in 2011, do not include data now available on the menopause transition as a time of increased CVD risk.
“As such, there is a compelling need to discuss the implications of the accumulating body of literature on this topic,” said Dr. El Khoudary.
The statement provides a contemporary synthesis of the existing data on menopause and how it relates to CVD, the leading cause of death of U.S. women.
Earlier age at natural menopause has generally been found to be a marker of greater CVD risk. Iatrogenically induced menopause (bilateral oophorectomy) during the premenopausal period is also associated with higher CVD risk, the data suggest.
Vasomotor symptoms are associated with worse levels of CVD risk factors and measures of subclinical atherosclerosis. Sleep disturbance has also been linked to greater risk for subclinical CVD and worse CV health indexes in women during midlife.
Increases in central/visceral fat and decreases in lean muscle mass are more pronounced during the menopause transition. This increased central adiposity is associated with increased risk for mortality, even among those with normal body mass index, the writing group found.
Increases in lipid levels (LDL cholesterol and apolipoprotein B), metabolic syndrome risk, and vascular remodeling at midlife are driven by the menopause transition more than aging, whereas increases in blood pressure, insulin level, and glucose level are likely more influenced by chronological aging, they reported.
Lifestyle interventions
The writing group noted that, because of the increase in overall life expectancy in the United States, a significant proportion of women will spend up to 40% of their lives after menopause.
Yet data suggest that only 7.2% of women transitioning to menopause are meeting physical activity guidelines and that fewer than 20% of those women are consistently maintaining a healthy diet.
Limited data from randomized, controlled trials suggest that a multidimensional lifestyle intervention during the menopause transition can prevent weight gain and reduce blood pressure and levels of triglycerides, blood glucose, and insulin and reduce the incidence of subclinical carotid atherosclerosis, they pointed out.
“Novel data” indicate a reversal in the associations of HDL cholesterol with CVD risk over the menopause transition, suggesting that higher HDL cholesterol levels may not consistently reflect good cardiovascular health in middle-aged women, the group noted.
There are also data suggesting that starting menopause hormone therapy when younger than 60 years or within 10 years of menopause is associated with reduced CVD risk.
The group said further research is needed into the cardiometabolic effects of menopause hormone therapy, including effects associated with form, route, and duration of administration, in women traversing menopause.
They also noted that data for the primary and secondary prevention of atherosclerotic CVD and improved survival with lipid-lowering interventions “remain elusive” for women and that further study is needed to develop evidence-based recommendations tailored specifically to women.
The research had no commercial funding. Dr. El Khoudary has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Treating insomnia, anxiety in a pandemic
Since the start of the pandemic, we have been conducting an extra hour of Virtual Rounds at the Center for Women’s Mental Health. Virtual Rounds has been an opportunity to discuss cases around a spectrum of clinical management issues with respect to depression, bipolar disorder, and a spectrum of anxiety disorders like obsessive-compulsive disorder (OCD), posttraumatic stress disorder (PTSD), and generalized anxiety disorder. How to apply the calculus of risk-benefit decision-making around management of psychiatric disorder during pregnancy and the postpartum period has been the cornerstone of the work at our center for over 2 decades.
When we went virtual at our center in the early Spring, we decided to keep the format of our faculty rounds the way they have been for years and to sustain cohesiveness of our program during the pandemic. But we thought the needs of pregnant and postpartum women warranted being addressed in a context more specific to COVID-19, and also that reproductive psychiatrists and other clinicians could learn from each other about novel issues coming up for this group of patients during the pandemic. With that backdrop, Marlene Freeman, MD, and I founded “Virtual Rounds at the Center” to respond to queries from our colleagues across the country; we do this just after our own rounds on Wednesdays at 2:00 p.m.
As the pandemic has progressed, Virtual Rounds has blossomed into a virtual community on the Zoom platform, where social workers, psychologists, nurse prescribers, psychiatrists, and obstetricians discuss the needs of pregnant and postpartum women specific to COVID-19. Frequently, our discussions involve a review of the risks and benefits of treatment before, during, and after pregnancy.
Seemingly, week to week, more and more colleagues raise questions about the treatment of anxiety and insomnia during pregnancy and the postpartum period. I’ve spoken in previous columns about the enhanced use of telemedicine. Telemedicine not only facilitates efforts like Virtual Rounds and our ability to reach out to colleagues across the country and share cases, but also has allowed us to keep even closer tabs on the emotional well-being of our pregnant and postpartum women during COVID-19.
The question is not just about the effects of a medicine that a woman might take to treat anxiety or insomnia during pregnancy, but the experience of the pandemic per se, which we are measuring in multiple studies now using a variety of psychological instruments that patients complete. The pandemic is unequivocally taking a still unquantified toll on the mental health of Americans and potentially on the next generation to come.
Midcycle awakening during pregnancy
Complaints of insomnia and midcycle awakening during pregnancy are not new – it is the rule, rather than the exception for many pregnant women, particularly later in pregnancy. We have unequivocally seen a worsening of complaints of sleep disruption including insomnia and midcycle awakening during the pandemic that is greater than what we have seen previously. Both patients and colleagues have asked us the safest ways to manage it. One of the first things we consider when we hear about insomnia is whether it is part of an underlying mood disorder. While we see primary insomnia clinically, it really is important to remember that insomnia can be part and parcel of an underlying mood disorder.
With that in mind, what are the options? During the pandemic, we’ve seen an increased use of digital cognitive behavioral therapy for insomnia (CBT-I) for patients who cannot initiate sleep, which has a very strong evidence base for effectiveness as a first-line intervention for many.
If a patient has an incomplete response to CBT-I, what might be pursued next? In our center, we have a low threshold for using low doses of benzodiazepines, such as lorazepam or clonazepam, because the majority of data do not support an increased risk of major congenital malformations even when used in the first trimester. It is quite common to see medicines such as newer nonbenzodiazepine sedative hypnotics such as Ambien CR (zolpidem) or Lunesta (eszopiclone) used by our colleagues in ob.gyn. The reproductive safety data on those medicines are particularly sparse, and they may have greater risk of cognitive side effects the next day, so we tend to avoid them.
Another sometimes-forgotten option to consider is using low doses of tricyclic antidepressants (i.e., 10-25 mg of nortriptyline at bedtime), with tricyclics having a 40-year history and at least one pooled analysis showing the absence of increased risk for major congenital malformations when used. This may be a very easy way of managing insomnia, with low-dose tricyclics having an anxiolytic effect as well.
Anxiety during pregnancy
The most common rise in symptoms during COVID-19 for women who are pregnant or post partum has been an increase in anxiety. Women present with a spectrum of concerns leading to anxiety symptoms in the context of the pandemic. Earlier on in the pandemic, concerns focused mostly on how to stay healthy, and how to mitigate risk and not catch SARS-CoV-2 during pregnancy, as well as the very complex issues that were playing out in real time as hospital systems were figuring out how to manage pregnant women in labor and to keep both them and staff safe. Over time, anxiety has shifted to still staying safe during the pandemic and the potential impact of SARS-CoV-2 infection on pregnancy outcomes. The No. 1 concern is what the implications of COVID-19 disease are on mother and child. New mothers also are anxious about how they will practically navigate life with a newborn in the postpartum setting.
Early on in the pandemic, some hospital systems severely limited who was in the room with a woman during labor, potentially impeding the wishes of women during delivery who would have wanted their loved ones and/or a doula present, as an example. With enhanced testing available now, protocols have since relaxed in many hospitals to allow partners – but not a team – to remain in the hospital during the labor process. Still, the prospect of delivering during a pandemic is undoubtedly a source of anxiety for some women.
This sort of anxiety, particularly in patients with preexisting anxiety disorders, can be particularly challenging. Fortunately, there has been a rapid increase over the last several years of digital apps to mitigate anxiety. While many of them have not been systematically studied, the data on biobehavioral intervention for anxiety is enormous, and this should be used as first-line treatment for patients with mild to moderate symptoms; so many women would prefer to avoid pharmacological intervention during pregnancy, if possible, to avoid fetal drug exposure. For patients who meet criteria for frank anxiety disorder, other nonpharmacologic interventions such as CBT have been shown to be effective.
Frequently, we see women who are experiencing levels of anxiety where nonpharmacological interventions have an incomplete response, and colleagues have asked about the safest way to treat these patients. As has been discussed in multiple previous columns, selective serotonin reuptake inhibitors (SSRIs) should be thought of sooner rather than later, particularly with medicines with good reproductive safety data such as sertraline, citalopram, or fluoxetine.
We also reported over 15 years ago that at least 30%-40% of women presenting with histories of recurrent major depression at the beginning of pregnancy had comorbid anxiety disorders, and that the use of benzodiazepines in that population in addition to SSRIs was exceedingly common, with doses of approximately 0.5-1.5 mg of clonazepam or lorazepam being standard fare. Again, this is very appropriate treatment to mitigate anxiety symptoms because now have enough data as a field that support the existence of adverse outcomes associated with untreated anxiety during pregnancy in terms of both adverse obstetric and neonatal outcomes, higher rates of preterm birth, and other obstetric complications. Hence, managing anxiety during pregnancy should be considered like managing a toxic exposure – the same way that one would be concerned about anything else that a pregnant woman could be exposed to.
Lastly, although no atypical antipsychotic has been approved for the treatment of anxiety, its use off label is extremely common. More and more data support the absence of a signal of teratogenicity across the family of molecules including atypical antipsychotics. Beyond potential use of atypical antipsychotics, at Virtual Rounds last week, a colleague asked about the use of gabapentin in a patient who was diagnosed with substance use disorder and who had inadvertently conceived on gabapentin, which was being used to treat both anxiety and insomnia. We have typically avoided the use of gabapentin during pregnancy because prospective data have been limited to relatively small case series and one report, with a total of exposures in roughly the 300 range.
However, our colleagues at the Harvard School of Public Health have recently published an article that looked at the United States Medicaid Analytic eXtract (MAX) dataset, which has been used to publish other articles addressing atypical antipsychotics, SSRIs, lithium, and pharmacovigilance investigations among other important topics. In this study, the database was used to look specifically at 4,642 pregnancies with gabapentin exposure relative to 1,744,447 unexposed pregnancies, without a significant finding for increased risk for major congenital malformations.
The question of an increased risk of cardiac malformations and of increased risk for obstetric complications are difficult to untangle from anxiety and depression, as they also are associated with those same outcomes. With that said, the analysis is a welcome addition to our knowledge base for a medicine used more widely to treat symptoms such as anxiety and insomnia in the general population, with a question mark around where it may fit into the algorithm during pregnancy.
In our center, gabapentin still would not be used as a first-line treatment for the management of anxiety or insomnia during pregnancy. But these new data still are reassuring for patients who come in, frequently with unplanned pregnancies. It is an important reminder to those of us taking care of patients during the pandemic to review use of contraception, because although data are unavailable specific to the period of the pandemic, what is clear is that, even prior to COVID-19, 50% of pregnancies in America were unplanned. Addressing issues of reliable use of contraception, particularly during the pandemic, is that much more important.
In this particular case, our clinician colleague in Virtual Rounds decided to continue gabapentin across pregnancy in the context of these reassuring data, but others may choose to discontinue or pursue some of the other treatment options noted above.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital (MGH) in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Since the start of the pandemic, we have been conducting an extra hour of Virtual Rounds at the Center for Women’s Mental Health. Virtual Rounds has been an opportunity to discuss cases around a spectrum of clinical management issues with respect to depression, bipolar disorder, and a spectrum of anxiety disorders like obsessive-compulsive disorder (OCD), posttraumatic stress disorder (PTSD), and generalized anxiety disorder. How to apply the calculus of risk-benefit decision-making around management of psychiatric disorder during pregnancy and the postpartum period has been the cornerstone of the work at our center for over 2 decades.
When we went virtual at our center in the early Spring, we decided to keep the format of our faculty rounds the way they have been for years and to sustain cohesiveness of our program during the pandemic. But we thought the needs of pregnant and postpartum women warranted being addressed in a context more specific to COVID-19, and also that reproductive psychiatrists and other clinicians could learn from each other about novel issues coming up for this group of patients during the pandemic. With that backdrop, Marlene Freeman, MD, and I founded “Virtual Rounds at the Center” to respond to queries from our colleagues across the country; we do this just after our own rounds on Wednesdays at 2:00 p.m.
As the pandemic has progressed, Virtual Rounds has blossomed into a virtual community on the Zoom platform, where social workers, psychologists, nurse prescribers, psychiatrists, and obstetricians discuss the needs of pregnant and postpartum women specific to COVID-19. Frequently, our discussions involve a review of the risks and benefits of treatment before, during, and after pregnancy.
Seemingly, week to week, more and more colleagues raise questions about the treatment of anxiety and insomnia during pregnancy and the postpartum period. I’ve spoken in previous columns about the enhanced use of telemedicine. Telemedicine not only facilitates efforts like Virtual Rounds and our ability to reach out to colleagues across the country and share cases, but also has allowed us to keep even closer tabs on the emotional well-being of our pregnant and postpartum women during COVID-19.
The question is not just about the effects of a medicine that a woman might take to treat anxiety or insomnia during pregnancy, but the experience of the pandemic per se, which we are measuring in multiple studies now using a variety of psychological instruments that patients complete. The pandemic is unequivocally taking a still unquantified toll on the mental health of Americans and potentially on the next generation to come.
Midcycle awakening during pregnancy
Complaints of insomnia and midcycle awakening during pregnancy are not new – it is the rule, rather than the exception for many pregnant women, particularly later in pregnancy. We have unequivocally seen a worsening of complaints of sleep disruption including insomnia and midcycle awakening during the pandemic that is greater than what we have seen previously. Both patients and colleagues have asked us the safest ways to manage it. One of the first things we consider when we hear about insomnia is whether it is part of an underlying mood disorder. While we see primary insomnia clinically, it really is important to remember that insomnia can be part and parcel of an underlying mood disorder.
With that in mind, what are the options? During the pandemic, we’ve seen an increased use of digital cognitive behavioral therapy for insomnia (CBT-I) for patients who cannot initiate sleep, which has a very strong evidence base for effectiveness as a first-line intervention for many.
If a patient has an incomplete response to CBT-I, what might be pursued next? In our center, we have a low threshold for using low doses of benzodiazepines, such as lorazepam or clonazepam, because the majority of data do not support an increased risk of major congenital malformations even when used in the first trimester. It is quite common to see medicines such as newer nonbenzodiazepine sedative hypnotics such as Ambien CR (zolpidem) or Lunesta (eszopiclone) used by our colleagues in ob.gyn. The reproductive safety data on those medicines are particularly sparse, and they may have greater risk of cognitive side effects the next day, so we tend to avoid them.
Another sometimes-forgotten option to consider is using low doses of tricyclic antidepressants (i.e., 10-25 mg of nortriptyline at bedtime), with tricyclics having a 40-year history and at least one pooled analysis showing the absence of increased risk for major congenital malformations when used. This may be a very easy way of managing insomnia, with low-dose tricyclics having an anxiolytic effect as well.
Anxiety during pregnancy
The most common rise in symptoms during COVID-19 for women who are pregnant or post partum has been an increase in anxiety. Women present with a spectrum of concerns leading to anxiety symptoms in the context of the pandemic. Earlier on in the pandemic, concerns focused mostly on how to stay healthy, and how to mitigate risk and not catch SARS-CoV-2 during pregnancy, as well as the very complex issues that were playing out in real time as hospital systems were figuring out how to manage pregnant women in labor and to keep both them and staff safe. Over time, anxiety has shifted to still staying safe during the pandemic and the potential impact of SARS-CoV-2 infection on pregnancy outcomes. The No. 1 concern is what the implications of COVID-19 disease are on mother and child. New mothers also are anxious about how they will practically navigate life with a newborn in the postpartum setting.
Early on in the pandemic, some hospital systems severely limited who was in the room with a woman during labor, potentially impeding the wishes of women during delivery who would have wanted their loved ones and/or a doula present, as an example. With enhanced testing available now, protocols have since relaxed in many hospitals to allow partners – but not a team – to remain in the hospital during the labor process. Still, the prospect of delivering during a pandemic is undoubtedly a source of anxiety for some women.
This sort of anxiety, particularly in patients with preexisting anxiety disorders, can be particularly challenging. Fortunately, there has been a rapid increase over the last several years of digital apps to mitigate anxiety. While many of them have not been systematically studied, the data on biobehavioral intervention for anxiety is enormous, and this should be used as first-line treatment for patients with mild to moderate symptoms; so many women would prefer to avoid pharmacological intervention during pregnancy, if possible, to avoid fetal drug exposure. For patients who meet criteria for frank anxiety disorder, other nonpharmacologic interventions such as CBT have been shown to be effective.
Frequently, we see women who are experiencing levels of anxiety where nonpharmacological interventions have an incomplete response, and colleagues have asked about the safest way to treat these patients. As has been discussed in multiple previous columns, selective serotonin reuptake inhibitors (SSRIs) should be thought of sooner rather than later, particularly with medicines with good reproductive safety data such as sertraline, citalopram, or fluoxetine.
We also reported over 15 years ago that at least 30%-40% of women presenting with histories of recurrent major depression at the beginning of pregnancy had comorbid anxiety disorders, and that the use of benzodiazepines in that population in addition to SSRIs was exceedingly common, with doses of approximately 0.5-1.5 mg of clonazepam or lorazepam being standard fare. Again, this is very appropriate treatment to mitigate anxiety symptoms because now have enough data as a field that support the existence of adverse outcomes associated with untreated anxiety during pregnancy in terms of both adverse obstetric and neonatal outcomes, higher rates of preterm birth, and other obstetric complications. Hence, managing anxiety during pregnancy should be considered like managing a toxic exposure – the same way that one would be concerned about anything else that a pregnant woman could be exposed to.
Lastly, although no atypical antipsychotic has been approved for the treatment of anxiety, its use off label is extremely common. More and more data support the absence of a signal of teratogenicity across the family of molecules including atypical antipsychotics. Beyond potential use of atypical antipsychotics, at Virtual Rounds last week, a colleague asked about the use of gabapentin in a patient who was diagnosed with substance use disorder and who had inadvertently conceived on gabapentin, which was being used to treat both anxiety and insomnia. We have typically avoided the use of gabapentin during pregnancy because prospective data have been limited to relatively small case series and one report, with a total of exposures in roughly the 300 range.
However, our colleagues at the Harvard School of Public Health have recently published an article that looked at the United States Medicaid Analytic eXtract (MAX) dataset, which has been used to publish other articles addressing atypical antipsychotics, SSRIs, lithium, and pharmacovigilance investigations among other important topics. In this study, the database was used to look specifically at 4,642 pregnancies with gabapentin exposure relative to 1,744,447 unexposed pregnancies, without a significant finding for increased risk for major congenital malformations.
The question of an increased risk of cardiac malformations and of increased risk for obstetric complications are difficult to untangle from anxiety and depression, as they also are associated with those same outcomes. With that said, the analysis is a welcome addition to our knowledge base for a medicine used more widely to treat symptoms such as anxiety and insomnia in the general population, with a question mark around where it may fit into the algorithm during pregnancy.
In our center, gabapentin still would not be used as a first-line treatment for the management of anxiety or insomnia during pregnancy. But these new data still are reassuring for patients who come in, frequently with unplanned pregnancies. It is an important reminder to those of us taking care of patients during the pandemic to review use of contraception, because although data are unavailable specific to the period of the pandemic, what is clear is that, even prior to COVID-19, 50% of pregnancies in America were unplanned. Addressing issues of reliable use of contraception, particularly during the pandemic, is that much more important.
In this particular case, our clinician colleague in Virtual Rounds decided to continue gabapentin across pregnancy in the context of these reassuring data, but others may choose to discontinue or pursue some of the other treatment options noted above.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital (MGH) in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Since the start of the pandemic, we have been conducting an extra hour of Virtual Rounds at the Center for Women’s Mental Health. Virtual Rounds has been an opportunity to discuss cases around a spectrum of clinical management issues with respect to depression, bipolar disorder, and a spectrum of anxiety disorders like obsessive-compulsive disorder (OCD), posttraumatic stress disorder (PTSD), and generalized anxiety disorder. How to apply the calculus of risk-benefit decision-making around management of psychiatric disorder during pregnancy and the postpartum period has been the cornerstone of the work at our center for over 2 decades.
When we went virtual at our center in the early Spring, we decided to keep the format of our faculty rounds the way they have been for years and to sustain cohesiveness of our program during the pandemic. But we thought the needs of pregnant and postpartum women warranted being addressed in a context more specific to COVID-19, and also that reproductive psychiatrists and other clinicians could learn from each other about novel issues coming up for this group of patients during the pandemic. With that backdrop, Marlene Freeman, MD, and I founded “Virtual Rounds at the Center” to respond to queries from our colleagues across the country; we do this just after our own rounds on Wednesdays at 2:00 p.m.
As the pandemic has progressed, Virtual Rounds has blossomed into a virtual community on the Zoom platform, where social workers, psychologists, nurse prescribers, psychiatrists, and obstetricians discuss the needs of pregnant and postpartum women specific to COVID-19. Frequently, our discussions involve a review of the risks and benefits of treatment before, during, and after pregnancy.
Seemingly, week to week, more and more colleagues raise questions about the treatment of anxiety and insomnia during pregnancy and the postpartum period. I’ve spoken in previous columns about the enhanced use of telemedicine. Telemedicine not only facilitates efforts like Virtual Rounds and our ability to reach out to colleagues across the country and share cases, but also has allowed us to keep even closer tabs on the emotional well-being of our pregnant and postpartum women during COVID-19.
The question is not just about the effects of a medicine that a woman might take to treat anxiety or insomnia during pregnancy, but the experience of the pandemic per se, which we are measuring in multiple studies now using a variety of psychological instruments that patients complete. The pandemic is unequivocally taking a still unquantified toll on the mental health of Americans and potentially on the next generation to come.
Midcycle awakening during pregnancy
Complaints of insomnia and midcycle awakening during pregnancy are not new – it is the rule, rather than the exception for many pregnant women, particularly later in pregnancy. We have unequivocally seen a worsening of complaints of sleep disruption including insomnia and midcycle awakening during the pandemic that is greater than what we have seen previously. Both patients and colleagues have asked us the safest ways to manage it. One of the first things we consider when we hear about insomnia is whether it is part of an underlying mood disorder. While we see primary insomnia clinically, it really is important to remember that insomnia can be part and parcel of an underlying mood disorder.
With that in mind, what are the options? During the pandemic, we’ve seen an increased use of digital cognitive behavioral therapy for insomnia (CBT-I) for patients who cannot initiate sleep, which has a very strong evidence base for effectiveness as a first-line intervention for many.
If a patient has an incomplete response to CBT-I, what might be pursued next? In our center, we have a low threshold for using low doses of benzodiazepines, such as lorazepam or clonazepam, because the majority of data do not support an increased risk of major congenital malformations even when used in the first trimester. It is quite common to see medicines such as newer nonbenzodiazepine sedative hypnotics such as Ambien CR (zolpidem) or Lunesta (eszopiclone) used by our colleagues in ob.gyn. The reproductive safety data on those medicines are particularly sparse, and they may have greater risk of cognitive side effects the next day, so we tend to avoid them.
Another sometimes-forgotten option to consider is using low doses of tricyclic antidepressants (i.e., 10-25 mg of nortriptyline at bedtime), with tricyclics having a 40-year history and at least one pooled analysis showing the absence of increased risk for major congenital malformations when used. This may be a very easy way of managing insomnia, with low-dose tricyclics having an anxiolytic effect as well.
Anxiety during pregnancy
The most common rise in symptoms during COVID-19 for women who are pregnant or post partum has been an increase in anxiety. Women present with a spectrum of concerns leading to anxiety symptoms in the context of the pandemic. Earlier on in the pandemic, concerns focused mostly on how to stay healthy, and how to mitigate risk and not catch SARS-CoV-2 during pregnancy, as well as the very complex issues that were playing out in real time as hospital systems were figuring out how to manage pregnant women in labor and to keep both them and staff safe. Over time, anxiety has shifted to still staying safe during the pandemic and the potential impact of SARS-CoV-2 infection on pregnancy outcomes. The No. 1 concern is what the implications of COVID-19 disease are on mother and child. New mothers also are anxious about how they will practically navigate life with a newborn in the postpartum setting.
Early on in the pandemic, some hospital systems severely limited who was in the room with a woman during labor, potentially impeding the wishes of women during delivery who would have wanted their loved ones and/or a doula present, as an example. With enhanced testing available now, protocols have since relaxed in many hospitals to allow partners – but not a team – to remain in the hospital during the labor process. Still, the prospect of delivering during a pandemic is undoubtedly a source of anxiety for some women.
This sort of anxiety, particularly in patients with preexisting anxiety disorders, can be particularly challenging. Fortunately, there has been a rapid increase over the last several years of digital apps to mitigate anxiety. While many of them have not been systematically studied, the data on biobehavioral intervention for anxiety is enormous, and this should be used as first-line treatment for patients with mild to moderate symptoms; so many women would prefer to avoid pharmacological intervention during pregnancy, if possible, to avoid fetal drug exposure. For patients who meet criteria for frank anxiety disorder, other nonpharmacologic interventions such as CBT have been shown to be effective.
Frequently, we see women who are experiencing levels of anxiety where nonpharmacological interventions have an incomplete response, and colleagues have asked about the safest way to treat these patients. As has been discussed in multiple previous columns, selective serotonin reuptake inhibitors (SSRIs) should be thought of sooner rather than later, particularly with medicines with good reproductive safety data such as sertraline, citalopram, or fluoxetine.
We also reported over 15 years ago that at least 30%-40% of women presenting with histories of recurrent major depression at the beginning of pregnancy had comorbid anxiety disorders, and that the use of benzodiazepines in that population in addition to SSRIs was exceedingly common, with doses of approximately 0.5-1.5 mg of clonazepam or lorazepam being standard fare. Again, this is very appropriate treatment to mitigate anxiety symptoms because now have enough data as a field that support the existence of adverse outcomes associated with untreated anxiety during pregnancy in terms of both adverse obstetric and neonatal outcomes, higher rates of preterm birth, and other obstetric complications. Hence, managing anxiety during pregnancy should be considered like managing a toxic exposure – the same way that one would be concerned about anything else that a pregnant woman could be exposed to.
Lastly, although no atypical antipsychotic has been approved for the treatment of anxiety, its use off label is extremely common. More and more data support the absence of a signal of teratogenicity across the family of molecules including atypical antipsychotics. Beyond potential use of atypical antipsychotics, at Virtual Rounds last week, a colleague asked about the use of gabapentin in a patient who was diagnosed with substance use disorder and who had inadvertently conceived on gabapentin, which was being used to treat both anxiety and insomnia. We have typically avoided the use of gabapentin during pregnancy because prospective data have been limited to relatively small case series and one report, with a total of exposures in roughly the 300 range.
However, our colleagues at the Harvard School of Public Health have recently published an article that looked at the United States Medicaid Analytic eXtract (MAX) dataset, which has been used to publish other articles addressing atypical antipsychotics, SSRIs, lithium, and pharmacovigilance investigations among other important topics. In this study, the database was used to look specifically at 4,642 pregnancies with gabapentin exposure relative to 1,744,447 unexposed pregnancies, without a significant finding for increased risk for major congenital malformations.
The question of an increased risk of cardiac malformations and of increased risk for obstetric complications are difficult to untangle from anxiety and depression, as they also are associated with those same outcomes. With that said, the analysis is a welcome addition to our knowledge base for a medicine used more widely to treat symptoms such as anxiety and insomnia in the general population, with a question mark around where it may fit into the algorithm during pregnancy.
In our center, gabapentin still would not be used as a first-line treatment for the management of anxiety or insomnia during pregnancy. But these new data still are reassuring for patients who come in, frequently with unplanned pregnancies. It is an important reminder to those of us taking care of patients during the pandemic to review use of contraception, because although data are unavailable specific to the period of the pandemic, what is clear is that, even prior to COVID-19, 50% of pregnancies in America were unplanned. Addressing issues of reliable use of contraception, particularly during the pandemic, is that much more important.
In this particular case, our clinician colleague in Virtual Rounds decided to continue gabapentin across pregnancy in the context of these reassuring data, but others may choose to discontinue or pursue some of the other treatment options noted above.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital (MGH) in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
The fourth trimester: Achieving improved postpartum care
The field of ob.gyn. has long focused significantly more attention on the prenatal period – on determining the optimal frequency of ultrasound examinations, for instance, and on screening for diabetes and other conditions – than on women’s health and well-being after delivery.
The traditional 6-week postpartum visit has too often been a quick and cursory visit, with new mothers typically navigating the preceding postpartum transitions on their own.
The need to redefine postpartum care was a central message of Haywood Brown, MD, who in 2017 served as the president of the America College of Obstetricians and Gynecologists. Dr. Brown established a task force whose work resulted in important guidance for taking a more comprehensive and patient-centered approach to postpartum care.1
Improved care in the “fourth trimester,” as it has come to be known, is comprehensive and includes ensuring that our patients have a solid transition to health care beyond the pregnancy. We also hope that it will help us to reduce maternal mortality, given that more than half of pregnancy-related deaths occur after delivery.
Timing and frequency of contact
Historically, we’ve had a single 6-week postpartum visit, with little or no maternal support or patient contact before this visit unless the patient reported a complication. In the new paradigm, as described in the ACOG committee opinion on optimizing postpartum care, maternal care should be an ongoing process.1
so that any questions or concerns may be addressed and support can be provided.
This should be followed by individualized, ongoing care until a comprehensive postpartum visit covering physical, social, and psychological well-being is conducted by 12 weeks after birth – anytime between 4 and 12 weeks.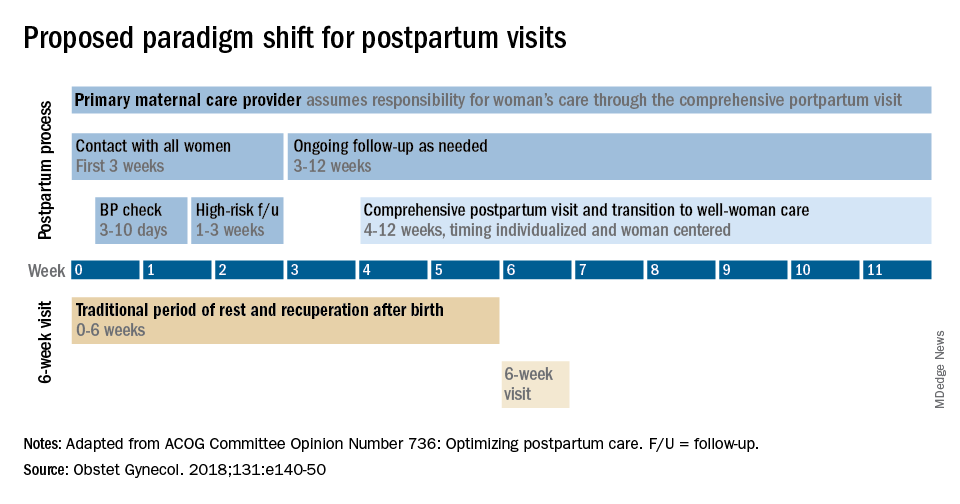
By stressing the importance of postpartum care during prenatal visits – and by talking about some of its key elements such as mental health, breastfeeding, and chronic disease management – we can let our patients know that postpartum care is not just an afterthought, but that it involves planning backed by evidence and expert opinion. Currently, as many as 40% of women do not attend a postpartum visit; early discussion, it is hoped, will increase attendance.
Certain high-risk groups should be seen or screened earlier than 3 weeks post partum. For instance, women who have hypertensive disorders of pregnancy should be evaluated no later than 7-10 days post partum, and women with severe hypertension should be seen within 72 hours, according to ACOG.
Early blood pressure checks – and follow-up as necessary – are critical for reducing the risk of postpartum stroke and other complications. I advocate uniformly checking blood pressure within several days after hospital discharge for all women who have hypertension at the end of their pregnancy.
Other high-risk conditions requiring early follow-up include diabetes and autoimmune conditions such as lupus, multiple sclerosis, and psoriasis that may flare in the postpartum period. Women with a history of postpartum depression similarly may benefit from early contact; they are at higher risk of having depression again, and there are clearly effective treatments, both medication and psychotherapy based.
In between the initial early contact (by 7-10 days post partum or by 3 weeks post partum) and the comprehensive visit between 4 and 12 weeks, the need for and timing of patient contact can be individualized. Some women will need only a brief contact and a visit at 8-10 weeks, while others will need much more. Our goal, as in all of medicine, is to provide individualized, patient-centered care.
Methods of contact
With the exception of the final comprehensive visit, postpartum care need not occur in person. Some conditions require an early office visit, but in general, as ACOG states, the usefulness of an in-person visit should be weighed against the burden of traveling to and attending that visit.
For many women, in-person visits are difficult, and we must be creative in utilizing telemedicine and phone support, text messaging, and app-based support. Having practiced during this pandemic, we are better positioned than ever before to make it relatively easy for new mothers to obtain ongoing postpartum care.
Notably, research is demonstrating that the use of technology may allow us to provide improved care and monitoring of hypertension in the postpartum period. For example, a randomized trial published in 2018 of over 200 women with a hypertensive disorder of pregnancy found that text-based surveillance with home blood pressure monitoring was more effective than usual in-person blood pressure checks in meeting clinical guidelines for postpartum monitoring.2
Women in the texting group were significantly more likely to have a single blood pressure obtained in the first 10 days post partum than women in the office group.
Postpartum care is also not a completely physician-driven endeavor. Much of what is needed to help women successfully navigate the fourth trimester can be provided by certified nurse midwives, advanced practice nurses, and other members of our maternal care teams.
Components of postpartum care
The postpartum care plan should be comprehensive, and having a checklist to guide one through initial and comprehensive visits may be helpful. The ACOG committee opinion categorizes the components of postpartum care into seven domains: mood and emotional well-being; infant care and feeding; sexuality, contraception, and birth spacing; sleep and fatigue; physical recovery from birth; chronic disease management; and health maintenance.1
The importance of screening for depression and anxiety cannot be emphasized enough. Perinatal depression is highly prevalent: It affects as many as one in seven women and can result in adverse short- and long-term effects on both the mother and child.
The U.S. Preventive Services Task Force has offered guidance for years, most recently in 2019 with its recommendations that clinicians refer pregnant and postpartum women who are at increased risk for depression to counseling interventions such as cognitive behavioral therapy and interpersonal therapy.3 There is evidence that some form of treatment for women who screen positive reduces the risk of perinatal depression.
Additionally, there is emerging evidence that postpartum PTSD may be as prevalent as postpartum depression.4 As ACOG points out, trauma is “in the eye of the beholder,” and an estimated 3%-16% of women have PTSD related to a traumatic birth experience. Complications like shoulder dystocia or postpartum hemorrhage, in which delivery processes rapidly change course, can be experienced as traumatic by women even though they and their infants are healthy. The risk of posttraumatic stress should be on our radar screen.
Interpregnancy intervals similarly are not discussed enough. We do not commonly talk to patients about how pregnancy and breastfeeding are nutritionally depleting and how it takes time to replenish these stores – yet birth spacing is so important.
Compared with interpregnancy intervals of at least 18 months, intervals shorter than 6 months were associated in a meta-analysis with increased risks of preterm birth, low birth weight, and small for gestational age.5 Optimal birth spacing is one of the few low-cost interventions available for reducing pregnancy complications in the future.
Finally, that chronic disease management is a domain of postpartum care warrants emphasis. We must work to ensure that patients have a solid plan of care in place for their diabetes, hypertension, lupus, or other chronic conditions. This includes who will provide that ongoing care, as well as when medical management should be restarted.
Some women are aware of the importance of timely care – of not waiting for 12 months, for instance, to see an internist or specialist – but others are not.
Again, certain health conditions such as multiple sclerosis and RA necessitate follow-up within a couple weeks after delivery so that medications can be restarted or dose adjustments made. The need for early postpartum follow-up can be discussed during prenatal visits, along with anticipatory guidance about breastfeeding, the signs and symptoms of perinatal depression and anxiety, and other components of the fourth trimester.
Dr. Macones has no relevant financial disclosures.
References
1. Obstet Gynecol. 2018 May;131(5):e140-50.
2. BMJ Qual Saf. 2018 Apr 27;27(11):871-7.
3. JAMA. 2019 Feb 12;321(6):580-7.
4. Clin Psychol Rev. 2014 Jul;34:389-401.JAMA. 2006 Apr 19;295(15):1809-23.
The field of ob.gyn. has long focused significantly more attention on the prenatal period – on determining the optimal frequency of ultrasound examinations, for instance, and on screening for diabetes and other conditions – than on women’s health and well-being after delivery.
The traditional 6-week postpartum visit has too often been a quick and cursory visit, with new mothers typically navigating the preceding postpartum transitions on their own.
The need to redefine postpartum care was a central message of Haywood Brown, MD, who in 2017 served as the president of the America College of Obstetricians and Gynecologists. Dr. Brown established a task force whose work resulted in important guidance for taking a more comprehensive and patient-centered approach to postpartum care.1
Improved care in the “fourth trimester,” as it has come to be known, is comprehensive and includes ensuring that our patients have a solid transition to health care beyond the pregnancy. We also hope that it will help us to reduce maternal mortality, given that more than half of pregnancy-related deaths occur after delivery.
Timing and frequency of contact
Historically, we’ve had a single 6-week postpartum visit, with little or no maternal support or patient contact before this visit unless the patient reported a complication. In the new paradigm, as described in the ACOG committee opinion on optimizing postpartum care, maternal care should be an ongoing process.1
so that any questions or concerns may be addressed and support can be provided.
This should be followed by individualized, ongoing care until a comprehensive postpartum visit covering physical, social, and psychological well-being is conducted by 12 weeks after birth – anytime between 4 and 12 weeks.
By stressing the importance of postpartum care during prenatal visits – and by talking about some of its key elements such as mental health, breastfeeding, and chronic disease management – we can let our patients know that postpartum care is not just an afterthought, but that it involves planning backed by evidence and expert opinion. Currently, as many as 40% of women do not attend a postpartum visit; early discussion, it is hoped, will increase attendance.
Certain high-risk groups should be seen or screened earlier than 3 weeks post partum. For instance, women who have hypertensive disorders of pregnancy should be evaluated no later than 7-10 days post partum, and women with severe hypertension should be seen within 72 hours, according to ACOG.
Early blood pressure checks – and follow-up as necessary – are critical for reducing the risk of postpartum stroke and other complications. I advocate uniformly checking blood pressure within several days after hospital discharge for all women who have hypertension at the end of their pregnancy.
Other high-risk conditions requiring early follow-up include diabetes and autoimmune conditions such as lupus, multiple sclerosis, and psoriasis that may flare in the postpartum period. Women with a history of postpartum depression similarly may benefit from early contact; they are at higher risk of having depression again, and there are clearly effective treatments, both medication and psychotherapy based.
In between the initial early contact (by 7-10 days post partum or by 3 weeks post partum) and the comprehensive visit between 4 and 12 weeks, the need for and timing of patient contact can be individualized. Some women will need only a brief contact and a visit at 8-10 weeks, while others will need much more. Our goal, as in all of medicine, is to provide individualized, patient-centered care.
Methods of contact
With the exception of the final comprehensive visit, postpartum care need not occur in person. Some conditions require an early office visit, but in general, as ACOG states, the usefulness of an in-person visit should be weighed against the burden of traveling to and attending that visit.
For many women, in-person visits are difficult, and we must be creative in utilizing telemedicine and phone support, text messaging, and app-based support. Having practiced during this pandemic, we are better positioned than ever before to make it relatively easy for new mothers to obtain ongoing postpartum care.
Notably, research is demonstrating that the use of technology may allow us to provide improved care and monitoring of hypertension in the postpartum period. For example, a randomized trial published in 2018 of over 200 women with a hypertensive disorder of pregnancy found that text-based surveillance with home blood pressure monitoring was more effective than usual in-person blood pressure checks in meeting clinical guidelines for postpartum monitoring.2
Women in the texting group were significantly more likely to have a single blood pressure obtained in the first 10 days post partum than women in the office group.
Postpartum care is also not a completely physician-driven endeavor. Much of what is needed to help women successfully navigate the fourth trimester can be provided by certified nurse midwives, advanced practice nurses, and other members of our maternal care teams.
Components of postpartum care
The postpartum care plan should be comprehensive, and having a checklist to guide one through initial and comprehensive visits may be helpful. The ACOG committee opinion categorizes the components of postpartum care into seven domains: mood and emotional well-being; infant care and feeding; sexuality, contraception, and birth spacing; sleep and fatigue; physical recovery from birth; chronic disease management; and health maintenance.1
The importance of screening for depression and anxiety cannot be emphasized enough. Perinatal depression is highly prevalent: It affects as many as one in seven women and can result in adverse short- and long-term effects on both the mother and child.
The U.S. Preventive Services Task Force has offered guidance for years, most recently in 2019 with its recommendations that clinicians refer pregnant and postpartum women who are at increased risk for depression to counseling interventions such as cognitive behavioral therapy and interpersonal therapy.3 There is evidence that some form of treatment for women who screen positive reduces the risk of perinatal depression.
Additionally, there is emerging evidence that postpartum PTSD may be as prevalent as postpartum depression.4 As ACOG points out, trauma is “in the eye of the beholder,” and an estimated 3%-16% of women have PTSD related to a traumatic birth experience. Complications like shoulder dystocia or postpartum hemorrhage, in which delivery processes rapidly change course, can be experienced as traumatic by women even though they and their infants are healthy. The risk of posttraumatic stress should be on our radar screen.
Interpregnancy intervals similarly are not discussed enough. We do not commonly talk to patients about how pregnancy and breastfeeding are nutritionally depleting and how it takes time to replenish these stores – yet birth spacing is so important.
Compared with interpregnancy intervals of at least 18 months, intervals shorter than 6 months were associated in a meta-analysis with increased risks of preterm birth, low birth weight, and small for gestational age.5 Optimal birth spacing is one of the few low-cost interventions available for reducing pregnancy complications in the future.
Finally, that chronic disease management is a domain of postpartum care warrants emphasis. We must work to ensure that patients have a solid plan of care in place for their diabetes, hypertension, lupus, or other chronic conditions. This includes who will provide that ongoing care, as well as when medical management should be restarted.
Some women are aware of the importance of timely care – of not waiting for 12 months, for instance, to see an internist or specialist – but others are not.
Again, certain health conditions such as multiple sclerosis and RA necessitate follow-up within a couple weeks after delivery so that medications can be restarted or dose adjustments made. The need for early postpartum follow-up can be discussed during prenatal visits, along with anticipatory guidance about breastfeeding, the signs and symptoms of perinatal depression and anxiety, and other components of the fourth trimester.
Dr. Macones has no relevant financial disclosures.
References
1. Obstet Gynecol. 2018 May;131(5):e140-50.
2. BMJ Qual Saf. 2018 Apr 27;27(11):871-7.
3. JAMA. 2019 Feb 12;321(6):580-7.
4. Clin Psychol Rev. 2014 Jul;34:389-401.JAMA. 2006 Apr 19;295(15):1809-23.
The field of ob.gyn. has long focused significantly more attention on the prenatal period – on determining the optimal frequency of ultrasound examinations, for instance, and on screening for diabetes and other conditions – than on women’s health and well-being after delivery.
The traditional 6-week postpartum visit has too often been a quick and cursory visit, with new mothers typically navigating the preceding postpartum transitions on their own.
The need to redefine postpartum care was a central message of Haywood Brown, MD, who in 2017 served as the president of the America College of Obstetricians and Gynecologists. Dr. Brown established a task force whose work resulted in important guidance for taking a more comprehensive and patient-centered approach to postpartum care.1
Improved care in the “fourth trimester,” as it has come to be known, is comprehensive and includes ensuring that our patients have a solid transition to health care beyond the pregnancy. We also hope that it will help us to reduce maternal mortality, given that more than half of pregnancy-related deaths occur after delivery.
Timing and frequency of contact
Historically, we’ve had a single 6-week postpartum visit, with little or no maternal support or patient contact before this visit unless the patient reported a complication. In the new paradigm, as described in the ACOG committee opinion on optimizing postpartum care, maternal care should be an ongoing process.1
so that any questions or concerns may be addressed and support can be provided.
This should be followed by individualized, ongoing care until a comprehensive postpartum visit covering physical, social, and psychological well-being is conducted by 12 weeks after birth – anytime between 4 and 12 weeks.
By stressing the importance of postpartum care during prenatal visits – and by talking about some of its key elements such as mental health, breastfeeding, and chronic disease management – we can let our patients know that postpartum care is not just an afterthought, but that it involves planning backed by evidence and expert opinion. Currently, as many as 40% of women do not attend a postpartum visit; early discussion, it is hoped, will increase attendance.
Certain high-risk groups should be seen or screened earlier than 3 weeks post partum. For instance, women who have hypertensive disorders of pregnancy should be evaluated no later than 7-10 days post partum, and women with severe hypertension should be seen within 72 hours, according to ACOG.
Early blood pressure checks – and follow-up as necessary – are critical for reducing the risk of postpartum stroke and other complications. I advocate uniformly checking blood pressure within several days after hospital discharge for all women who have hypertension at the end of their pregnancy.
Other high-risk conditions requiring early follow-up include diabetes and autoimmune conditions such as lupus, multiple sclerosis, and psoriasis that may flare in the postpartum period. Women with a history of postpartum depression similarly may benefit from early contact; they are at higher risk of having depression again, and there are clearly effective treatments, both medication and psychotherapy based.
In between the initial early contact (by 7-10 days post partum or by 3 weeks post partum) and the comprehensive visit between 4 and 12 weeks, the need for and timing of patient contact can be individualized. Some women will need only a brief contact and a visit at 8-10 weeks, while others will need much more. Our goal, as in all of medicine, is to provide individualized, patient-centered care.
Methods of contact
With the exception of the final comprehensive visit, postpartum care need not occur in person. Some conditions require an early office visit, but in general, as ACOG states, the usefulness of an in-person visit should be weighed against the burden of traveling to and attending that visit.
For many women, in-person visits are difficult, and we must be creative in utilizing telemedicine and phone support, text messaging, and app-based support. Having practiced during this pandemic, we are better positioned than ever before to make it relatively easy for new mothers to obtain ongoing postpartum care.
Notably, research is demonstrating that the use of technology may allow us to provide improved care and monitoring of hypertension in the postpartum period. For example, a randomized trial published in 2018 of over 200 women with a hypertensive disorder of pregnancy found that text-based surveillance with home blood pressure monitoring was more effective than usual in-person blood pressure checks in meeting clinical guidelines for postpartum monitoring.2
Women in the texting group were significantly more likely to have a single blood pressure obtained in the first 10 days post partum than women in the office group.
Postpartum care is also not a completely physician-driven endeavor. Much of what is needed to help women successfully navigate the fourth trimester can be provided by certified nurse midwives, advanced practice nurses, and other members of our maternal care teams.
Components of postpartum care
The postpartum care plan should be comprehensive, and having a checklist to guide one through initial and comprehensive visits may be helpful. The ACOG committee opinion categorizes the components of postpartum care into seven domains: mood and emotional well-being; infant care and feeding; sexuality, contraception, and birth spacing; sleep and fatigue; physical recovery from birth; chronic disease management; and health maintenance.1
The importance of screening for depression and anxiety cannot be emphasized enough. Perinatal depression is highly prevalent: It affects as many as one in seven women and can result in adverse short- and long-term effects on both the mother and child.
The U.S. Preventive Services Task Force has offered guidance for years, most recently in 2019 with its recommendations that clinicians refer pregnant and postpartum women who are at increased risk for depression to counseling interventions such as cognitive behavioral therapy and interpersonal therapy.3 There is evidence that some form of treatment for women who screen positive reduces the risk of perinatal depression.
Additionally, there is emerging evidence that postpartum PTSD may be as prevalent as postpartum depression.4 As ACOG points out, trauma is “in the eye of the beholder,” and an estimated 3%-16% of women have PTSD related to a traumatic birth experience. Complications like shoulder dystocia or postpartum hemorrhage, in which delivery processes rapidly change course, can be experienced as traumatic by women even though they and their infants are healthy. The risk of posttraumatic stress should be on our radar screen.
Interpregnancy intervals similarly are not discussed enough. We do not commonly talk to patients about how pregnancy and breastfeeding are nutritionally depleting and how it takes time to replenish these stores – yet birth spacing is so important.
Compared with interpregnancy intervals of at least 18 months, intervals shorter than 6 months were associated in a meta-analysis with increased risks of preterm birth, low birth weight, and small for gestational age.5 Optimal birth spacing is one of the few low-cost interventions available for reducing pregnancy complications in the future.
Finally, that chronic disease management is a domain of postpartum care warrants emphasis. We must work to ensure that patients have a solid plan of care in place for their diabetes, hypertension, lupus, or other chronic conditions. This includes who will provide that ongoing care, as well as when medical management should be restarted.
Some women are aware of the importance of timely care – of not waiting for 12 months, for instance, to see an internist or specialist – but others are not.
Again, certain health conditions such as multiple sclerosis and RA necessitate follow-up within a couple weeks after delivery so that medications can be restarted or dose adjustments made. The need for early postpartum follow-up can be discussed during prenatal visits, along with anticipatory guidance about breastfeeding, the signs and symptoms of perinatal depression and anxiety, and other components of the fourth trimester.
Dr. Macones has no relevant financial disclosures.
References
1. Obstet Gynecol. 2018 May;131(5):e140-50.
2. BMJ Qual Saf. 2018 Apr 27;27(11):871-7.
3. JAMA. 2019 Feb 12;321(6):580-7.
4. Clin Psychol Rev. 2014 Jul;34:389-401.JAMA. 2006 Apr 19;295(15):1809-23.
The fourth trimester
As we approach the end of this year, one of the most surreal times in human history, we will look back on the many things we taught ourselves, the many things we took for granted, the many things we were grateful for, the many things we missed, and the many things we plan to do once we can do things again. Among the many things 2020 taught us to appreciate was the very real manifestation of the old adage, “prevention is the best medicine.” To prevent transmission of SARS-CoV-2, we wore masks, we sanitized everything, we avoided crowds, we traded in-person meetings for virtual meetings, we learned how to homeschool our children, and we delayed seeing relatives and friends.
Ob.gyns. in small and large practices around the world had the tremendous challenge of balancing necessary in-person prenatal care services with keeping their patients and babies safe. Labor and delivery units had even greater demands to keep women and neonates free of SARS-CoV-2 infection. Practices quickly put into place new treatment protocols and new management strategies to maintain the health of their staff while ensuring a high quality of care.
While we have focused much of our attention on greater precautions during pregnancy and childbirth, an important component of care is the immediate postpartum period – colloquially referred to as the “fourth trimester” – which remains critical to maintaining physical and mental health and well-being.
Despite concerns regarding COVID-19 safety, we should continue monitoring our patients during these crucial first weeks after childbirth. This year of social isolation, financial strain, and incredible uncertainty has created additional stress in many women’s lives. The usual support that some women would receive from family members, friends, and other mothers in the early days post partum may not be available. The pandemic also has further highlighted inequities in access to health care for vulnerable groups. In addition, restrictions have increased the incidence of intimate partner violence as many women and children have needed to shelter with their abusers. Perhaps now more than any time previously, ob.gyns. must be attuned to their patients’ needs and be ready to provide compassionate and sensitive care.
In this final month of the year, we have invited George A. Macones, MD, professor and chair of the department of women’s health at the University of Texas, Austin, to address the importance of care in the final “trimester” of pregnancy – the first 3 months post partum.
Dr. Reece, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He is the medical editor of this column. He said he had no relevant financial disclosures. Contact him at [email protected].
*This version has been updated to correct an erroneous byline, photo, and bio.
As we approach the end of this year, one of the most surreal times in human history, we will look back on the many things we taught ourselves, the many things we took for granted, the many things we were grateful for, the many things we missed, and the many things we plan to do once we can do things again. Among the many things 2020 taught us to appreciate was the very real manifestation of the old adage, “prevention is the best medicine.” To prevent transmission of SARS-CoV-2, we wore masks, we sanitized everything, we avoided crowds, we traded in-person meetings for virtual meetings, we learned how to homeschool our children, and we delayed seeing relatives and friends.
Ob.gyns. in small and large practices around the world had the tremendous challenge of balancing necessary in-person prenatal care services with keeping their patients and babies safe. Labor and delivery units had even greater demands to keep women and neonates free of SARS-CoV-2 infection. Practices quickly put into place new treatment protocols and new management strategies to maintain the health of their staff while ensuring a high quality of care.
While we have focused much of our attention on greater precautions during pregnancy and childbirth, an important component of care is the immediate postpartum period – colloquially referred to as the “fourth trimester” – which remains critical to maintaining physical and mental health and well-being.
Despite concerns regarding COVID-19 safety, we should continue monitoring our patients during these crucial first weeks after childbirth. This year of social isolation, financial strain, and incredible uncertainty has created additional stress in many women’s lives. The usual support that some women would receive from family members, friends, and other mothers in the early days post partum may not be available. The pandemic also has further highlighted inequities in access to health care for vulnerable groups. In addition, restrictions have increased the incidence of intimate partner violence as many women and children have needed to shelter with their abusers. Perhaps now more than any time previously, ob.gyns. must be attuned to their patients’ needs and be ready to provide compassionate and sensitive care.
In this final month of the year, we have invited George A. Macones, MD, professor and chair of the department of women’s health at the University of Texas, Austin, to address the importance of care in the final “trimester” of pregnancy – the first 3 months post partum.
Dr. Reece, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He is the medical editor of this column. He said he had no relevant financial disclosures. Contact him at [email protected].
*This version has been updated to correct an erroneous byline, photo, and bio.
As we approach the end of this year, one of the most surreal times in human history, we will look back on the many things we taught ourselves, the many things we took for granted, the many things we were grateful for, the many things we missed, and the many things we plan to do once we can do things again. Among the many things 2020 taught us to appreciate was the very real manifestation of the old adage, “prevention is the best medicine.” To prevent transmission of SARS-CoV-2, we wore masks, we sanitized everything, we avoided crowds, we traded in-person meetings for virtual meetings, we learned how to homeschool our children, and we delayed seeing relatives and friends.
Ob.gyns. in small and large practices around the world had the tremendous challenge of balancing necessary in-person prenatal care services with keeping their patients and babies safe. Labor and delivery units had even greater demands to keep women and neonates free of SARS-CoV-2 infection. Practices quickly put into place new treatment protocols and new management strategies to maintain the health of their staff while ensuring a high quality of care.
While we have focused much of our attention on greater precautions during pregnancy and childbirth, an important component of care is the immediate postpartum period – colloquially referred to as the “fourth trimester” – which remains critical to maintaining physical and mental health and well-being.
Despite concerns regarding COVID-19 safety, we should continue monitoring our patients during these crucial first weeks after childbirth. This year of social isolation, financial strain, and incredible uncertainty has created additional stress in many women’s lives. The usual support that some women would receive from family members, friends, and other mothers in the early days post partum may not be available. The pandemic also has further highlighted inequities in access to health care for vulnerable groups. In addition, restrictions have increased the incidence of intimate partner violence as many women and children have needed to shelter with their abusers. Perhaps now more than any time previously, ob.gyns. must be attuned to their patients’ needs and be ready to provide compassionate and sensitive care.
In this final month of the year, we have invited George A. Macones, MD, professor and chair of the department of women’s health at the University of Texas, Austin, to address the importance of care in the final “trimester” of pregnancy – the first 3 months post partum.
Dr. Reece, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He is the medical editor of this column. He said he had no relevant financial disclosures. Contact him at [email protected].
*This version has been updated to correct an erroneous byline, photo, and bio.
Prophylactic HIV treatment in female STI patients is rare
reported Kirk D. Henny, PhD, and colleagues of the Centers for Disease Control and Prevention.
In an effort to quantify HIV testing rates as well as the rate of pre-exposure prophylaxis (PrEP) among women with gonorrhea or syphilis, Dr. Henny and his colleagues performed a multivariate logistic regression analysis of 13,074 female patients aged 15-64 diagnosed with a STI in the absence of HIV. Data was pulled in 2017 from the IBM MarketScan commercial and Medicaid insurance databases, and the research was published in Obstetrics & Gynecology.
Medicaid patients were more likely to be tested for HIV
A total of 3,709 patients with commercial insurance were diagnosed with gonorrhea and 1,696 with syphilis. Among those with Medicaid, 6,172 were diagnosed with gonorrhea and 1,497 with syphilis. Medicaid patients diagnosed with either STI were more likely to be tested for HIV than the commercially insured patients. With an adjusted prevalence ratio, patients commercially insured with had either STI were more likely to be tested for HIV than patients who had no STI. Prophylactic treatment rates were similar in both insurance groups: 0.15% in the commercial insurance group and 0.26% in the Medicaid group. No patient from either group who was diagnosed with gonorrhea or syphilis and subsequently tested for HIV received pre-exposure prophylactic (PrEP) treatment.
STI diagnosis is a significant indicator of future HIV
Female patients diagnosed with either STI are more likely to contract HIV, the researchers noted. They cautioned that their findings of low HIV testing rates and the absence of prophylactic treatment means that “these missed opportunities for health care professionals to intervene with female patients diagnosed with gonorrhea or syphilis might have contributed to HIV infections that could have been averted.”
The researchers also pointed out that, in a recent analysis of pharmacy data, prophylactic prescribing for female patients with clinical indications for PrEP was 6.6%, less than one-third the coverage provided to male patients.
Future research should target understanding “individual and contextual factors associated with low HIV testing” and PrEP treatment in female patients, especially those with STIs, Dr. Henny and his colleagues advised.
In a separate interview, Constance Bohon, MD FACOG, observed: “The authors present data to document the low incidence of pre-exposure prophylaxis in women who are at substantial risk of acquiring HIV and possible causes for the low utilization of this treatment.” It is important to identify barriers to diagnosis, counseling, and treatment, she advised.
“Multicenter studies to determine the best methodologies to improve the identification, management, and treatment of these at-risk women need to be done, and the conclusions disseminated to health care providers caring for women,” Dr. Bohon said.
PrEP is an important, simple strategy for reducing HIV transmission
“Pre-exposure prophylaxis has been demonstrated to decrease HIV acquisition in those at risk by up to 90% when taken appropriately,” and yet prescribing rates are extremely low (2%-6%) in at-risk women and especially women of color. These disparities have only grown over time, with prophylactic prescriptions for women at 5% between 2012 and 2017, compared with 68% for men, Catherine S. Eppes, MD, MPH, and Jennifer McKinney, MD, MPH, said in a related editorial commenting on the Research Letter by Dr. Henny and colleagues in Obstetrics & Gynecology (2020 Dec;136[6]:1080-2).
Given the abundant research demonstrating the importance and ease of prescribing PrEP, the question remains: “why does preexposure prophylaxis uptake remain so low, especially for women and women of color? There are three important issues about preexposure prophylaxis raised by this study: the research gap, the implementation gap, and the effect of systemic racism and bias,” noted Dr. Eppes and Dr. McKinney.
Women constitute a significant portion of the population that would benefit from HIV-prevention strategies, yet they continue to be excluded from research, they noted. “Much focus on research into barriers and implementation interventions for preexposure prophylaxis have focused on men who have sex with men and transgender women,” the authors of the editorial wrote.
Most women eligible for treatment would be willing to consider it if they were aware of the option, but numerous studies have cited a lack of awareness, especially among high-risk women of color in the United States, Dr. Eppes and Dr. McKinney noted.
Clinicians also need to add it to their growing checklist of mandatory appointment discussion topics, the editorialists said. “We propose standardized inclusion of preexposure prophylaxis counseling during reproductive healthcare visits. This could be aided through an electronic medical record-based best practice advisory alert. … Standardized order sets with the medication and laboratory studies necessary for safe monitoring could facilitate ease of incorporating into routine visits,” they suggested.
“Preexposure prophylaxis is extremely effective in preventing HIV, is safe, and is the only prevention method that leaves control entirely in the hands of the female partner. As a specialty, we have a responsibility to make sure our patients know about this option,” the editorialists concluded.
The authors had no financial disclosures to report. Dr. Bohon had no conflicts of interest to report.
SOURCE: Henny KD et al. Obstet Gynecol. 2020 Dec;136(6):1083-5.
reported Kirk D. Henny, PhD, and colleagues of the Centers for Disease Control and Prevention.
In an effort to quantify HIV testing rates as well as the rate of pre-exposure prophylaxis (PrEP) among women with gonorrhea or syphilis, Dr. Henny and his colleagues performed a multivariate logistic regression analysis of 13,074 female patients aged 15-64 diagnosed with a STI in the absence of HIV. Data was pulled in 2017 from the IBM MarketScan commercial and Medicaid insurance databases, and the research was published in Obstetrics & Gynecology.
Medicaid patients were more likely to be tested for HIV
A total of 3,709 patients with commercial insurance were diagnosed with gonorrhea and 1,696 with syphilis. Among those with Medicaid, 6,172 were diagnosed with gonorrhea and 1,497 with syphilis. Medicaid patients diagnosed with either STI were more likely to be tested for HIV than the commercially insured patients. With an adjusted prevalence ratio, patients commercially insured with had either STI were more likely to be tested for HIV than patients who had no STI. Prophylactic treatment rates were similar in both insurance groups: 0.15% in the commercial insurance group and 0.26% in the Medicaid group. No patient from either group who was diagnosed with gonorrhea or syphilis and subsequently tested for HIV received pre-exposure prophylactic (PrEP) treatment.
STI diagnosis is a significant indicator of future HIV
Female patients diagnosed with either STI are more likely to contract HIV, the researchers noted. They cautioned that their findings of low HIV testing rates and the absence of prophylactic treatment means that “these missed opportunities for health care professionals to intervene with female patients diagnosed with gonorrhea or syphilis might have contributed to HIV infections that could have been averted.”
The researchers also pointed out that, in a recent analysis of pharmacy data, prophylactic prescribing for female patients with clinical indications for PrEP was 6.6%, less than one-third the coverage provided to male patients.
Future research should target understanding “individual and contextual factors associated with low HIV testing” and PrEP treatment in female patients, especially those with STIs, Dr. Henny and his colleagues advised.
In a separate interview, Constance Bohon, MD FACOG, observed: “The authors present data to document the low incidence of pre-exposure prophylaxis in women who are at substantial risk of acquiring HIV and possible causes for the low utilization of this treatment.” It is important to identify barriers to diagnosis, counseling, and treatment, she advised.
“Multicenter studies to determine the best methodologies to improve the identification, management, and treatment of these at-risk women need to be done, and the conclusions disseminated to health care providers caring for women,” Dr. Bohon said.
PrEP is an important, simple strategy for reducing HIV transmission
“Pre-exposure prophylaxis has been demonstrated to decrease HIV acquisition in those at risk by up to 90% when taken appropriately,” and yet prescribing rates are extremely low (2%-6%) in at-risk women and especially women of color. These disparities have only grown over time, with prophylactic prescriptions for women at 5% between 2012 and 2017, compared with 68% for men, Catherine S. Eppes, MD, MPH, and Jennifer McKinney, MD, MPH, said in a related editorial commenting on the Research Letter by Dr. Henny and colleagues in Obstetrics & Gynecology (2020 Dec;136[6]:1080-2).
Given the abundant research demonstrating the importance and ease of prescribing PrEP, the question remains: “why does preexposure prophylaxis uptake remain so low, especially for women and women of color? There are three important issues about preexposure prophylaxis raised by this study: the research gap, the implementation gap, and the effect of systemic racism and bias,” noted Dr. Eppes and Dr. McKinney.
Women constitute a significant portion of the population that would benefit from HIV-prevention strategies, yet they continue to be excluded from research, they noted. “Much focus on research into barriers and implementation interventions for preexposure prophylaxis have focused on men who have sex with men and transgender women,” the authors of the editorial wrote.
Most women eligible for treatment would be willing to consider it if they were aware of the option, but numerous studies have cited a lack of awareness, especially among high-risk women of color in the United States, Dr. Eppes and Dr. McKinney noted.
Clinicians also need to add it to their growing checklist of mandatory appointment discussion topics, the editorialists said. “We propose standardized inclusion of preexposure prophylaxis counseling during reproductive healthcare visits. This could be aided through an electronic medical record-based best practice advisory alert. … Standardized order sets with the medication and laboratory studies necessary for safe monitoring could facilitate ease of incorporating into routine visits,” they suggested.
“Preexposure prophylaxis is extremely effective in preventing HIV, is safe, and is the only prevention method that leaves control entirely in the hands of the female partner. As a specialty, we have a responsibility to make sure our patients know about this option,” the editorialists concluded.
The authors had no financial disclosures to report. Dr. Bohon had no conflicts of interest to report.
SOURCE: Henny KD et al. Obstet Gynecol. 2020 Dec;136(6):1083-5.
reported Kirk D. Henny, PhD, and colleagues of the Centers for Disease Control and Prevention.
In an effort to quantify HIV testing rates as well as the rate of pre-exposure prophylaxis (PrEP) among women with gonorrhea or syphilis, Dr. Henny and his colleagues performed a multivariate logistic regression analysis of 13,074 female patients aged 15-64 diagnosed with a STI in the absence of HIV. Data was pulled in 2017 from the IBM MarketScan commercial and Medicaid insurance databases, and the research was published in Obstetrics & Gynecology.
Medicaid patients were more likely to be tested for HIV
A total of 3,709 patients with commercial insurance were diagnosed with gonorrhea and 1,696 with syphilis. Among those with Medicaid, 6,172 were diagnosed with gonorrhea and 1,497 with syphilis. Medicaid patients diagnosed with either STI were more likely to be tested for HIV than the commercially insured patients. With an adjusted prevalence ratio, patients commercially insured with had either STI were more likely to be tested for HIV than patients who had no STI. Prophylactic treatment rates were similar in both insurance groups: 0.15% in the commercial insurance group and 0.26% in the Medicaid group. No patient from either group who was diagnosed with gonorrhea or syphilis and subsequently tested for HIV received pre-exposure prophylactic (PrEP) treatment.
STI diagnosis is a significant indicator of future HIV
Female patients diagnosed with either STI are more likely to contract HIV, the researchers noted. They cautioned that their findings of low HIV testing rates and the absence of prophylactic treatment means that “these missed opportunities for health care professionals to intervene with female patients diagnosed with gonorrhea or syphilis might have contributed to HIV infections that could have been averted.”
The researchers also pointed out that, in a recent analysis of pharmacy data, prophylactic prescribing for female patients with clinical indications for PrEP was 6.6%, less than one-third the coverage provided to male patients.
Future research should target understanding “individual and contextual factors associated with low HIV testing” and PrEP treatment in female patients, especially those with STIs, Dr. Henny and his colleagues advised.
In a separate interview, Constance Bohon, MD FACOG, observed: “The authors present data to document the low incidence of pre-exposure prophylaxis in women who are at substantial risk of acquiring HIV and possible causes for the low utilization of this treatment.” It is important to identify barriers to diagnosis, counseling, and treatment, she advised.
“Multicenter studies to determine the best methodologies to improve the identification, management, and treatment of these at-risk women need to be done, and the conclusions disseminated to health care providers caring for women,” Dr. Bohon said.
PrEP is an important, simple strategy for reducing HIV transmission
“Pre-exposure prophylaxis has been demonstrated to decrease HIV acquisition in those at risk by up to 90% when taken appropriately,” and yet prescribing rates are extremely low (2%-6%) in at-risk women and especially women of color. These disparities have only grown over time, with prophylactic prescriptions for women at 5% between 2012 and 2017, compared with 68% for men, Catherine S. Eppes, MD, MPH, and Jennifer McKinney, MD, MPH, said in a related editorial commenting on the Research Letter by Dr. Henny and colleagues in Obstetrics & Gynecology (2020 Dec;136[6]:1080-2).
Given the abundant research demonstrating the importance and ease of prescribing PrEP, the question remains: “why does preexposure prophylaxis uptake remain so low, especially for women and women of color? There are three important issues about preexposure prophylaxis raised by this study: the research gap, the implementation gap, and the effect of systemic racism and bias,” noted Dr. Eppes and Dr. McKinney.
Women constitute a significant portion of the population that would benefit from HIV-prevention strategies, yet they continue to be excluded from research, they noted. “Much focus on research into barriers and implementation interventions for preexposure prophylaxis have focused on men who have sex with men and transgender women,” the authors of the editorial wrote.
Most women eligible for treatment would be willing to consider it if they were aware of the option, but numerous studies have cited a lack of awareness, especially among high-risk women of color in the United States, Dr. Eppes and Dr. McKinney noted.
Clinicians also need to add it to their growing checklist of mandatory appointment discussion topics, the editorialists said. “We propose standardized inclusion of preexposure prophylaxis counseling during reproductive healthcare visits. This could be aided through an electronic medical record-based best practice advisory alert. … Standardized order sets with the medication and laboratory studies necessary for safe monitoring could facilitate ease of incorporating into routine visits,” they suggested.
“Preexposure prophylaxis is extremely effective in preventing HIV, is safe, and is the only prevention method that leaves control entirely in the hands of the female partner. As a specialty, we have a responsibility to make sure our patients know about this option,” the editorialists concluded.
The authors had no financial disclosures to report. Dr. Bohon had no conflicts of interest to report.
SOURCE: Henny KD et al. Obstet Gynecol. 2020 Dec;136(6):1083-5.
FROM OBSTETRICS & GYNECOLOGY
Metformin improves most outcomes for T2D during pregnancy
including reduced weight gain, reduced insulin doses, and fewer large-for-gestational-age babies, suggest the results of a randomized controlled trial.
However, the drug was associated with an increased risk of small-for-gestational-age babies, which poses the question as to risk versus benefit of metformin on the health of offspring.
“Better understanding of the short- and long-term implications of these effects on infants will be important to properly advise patients with type 2 diabetes contemplating use of metformin during pregnancy,” said lead author Denice S. Feig, MD, Mount Sinai Hospital, Toronto.
The research was presented at the Diabetes UK Professional Conference: Online Series on Nov. 17 and recently published in The Lancet Diabetes & Endocrinology.
Summing up, Dr. Feig said that, on balance, she would be inclined to give metformin to most pregnant women with type 2 diabetes, perhaps with the exception of those who may have risk factors for small-for-gestational-age babies; for example, women who’ve had intrauterine growth restriction, who are smokers, and have significant renal disease, or have a lower body mass index.
Increased prevalence of type 2 diabetes in pregnancy
Dr. Feig said that across the developed world there have been huge increases in the prevalence of type 2 diabetes in pregnancy in recent years.
Insulin is the standard treatment for the management of type 2 diabetes in pregnancy, but these women have marked insulin resistance that worsens in pregnancy, which means their insulin requirements increase, leading to weight gain, painful injections, high cost, and noncompliance.
So despite treatment with insulin, these women continue to face increased rates of adverse maternal and fetal outcomes.
And although metformin is increasingly being used in women with type 2 diabetes during pregnancy, there is a scarcity of data on the benefits and harms of metformin use on pregnancy outcomes in these women.
The MiTy trial was therefore undertaken to determine whether metformin could improve outcomes.
The team recruited 502 women from 29 sites in Canada and Australia who had type 2 diabetes prior to pregnancy or were diagnosed during pregnancy, before 20 weeks’ gestation. The women were randomized to metformin 1 g twice daily or placebo, in addition to their usual insulin regimen, at between 6 and 28 weeks’ gestation.
Type 2 diabetes was diagnosed prior to pregnancy in 83% of women in the metformin group and in 90% of those assigned to placebo. The mean hemoglobin A1c level at randomization was 47 mmol/mol (6.5%) in both groups.
The average maternal age at baseline was approximately 35 years and mean gestational age at randomization was 16 weeks. Mean prepregnancy BMI was approximately 34 kg/m2.
Of note, only 30% were of European ethnicity.
Less weight gain, lower A1c, less insulin needed with metformin
Dr. Feig reported that there was no significant difference between the treatment groups in terms of the proportion of women with the composite primary outcome of pregnancy loss, preterm birth, birth injury, respiratory distress, neonatal hypoglycemia, or admission to neonatal intensive care lasting more than 24 hours (P = 0.86).
However, women in the metformin group had significantly less overall weight gain during pregnancy than did those in the placebo group, at –1.8 kg (P < .0001).
They also had a significantly lower last A1c level in pregnancy, at 41 mmol/mol (5.9%) versus 43.2 mmol/mol (6.1%) in those given placebo (P = .015), and required fewer insulin doses, at 1.1 versus 1.5 units/kg/day (P < .0001), which translated to a reduction of almost 44 units/day.
Women given metformin were also less likely to require Cesarean section delivery, at 53.4% versus 62.7% in the placebo group (P = .03), although there was no difference between groups in terms of gestational hypertension or preeclampsia.
The most common adverse events were gastrointestinal complications, which occurred in 27.3% of women in the metformin group and 22.3% of those given placebo.
There were no significant differences between the metformin and placebo groups in rates of pregnancy loss (P = .81), preterm birth (P = .16), birth injury (P = .37), respiratory distress (P = .49), and congenital anomalies (P = .16).
Average birth weight lower with metformin
However, Dr. Feig showed that the average birth weight was lower for offspring of women given metformin than those assigned to placebo, at 3.2 kg (7.05 lb) versus 3.4 kg (7.4 lb) (P = .002).
Women given metformin were also less likely to have a baby with a birth weight of 4 kg (8.8 lb) or more, at 12.1% versus 19.2%, or a relative risk of 0.65 (P = .046), and a baby that was extremely large for gestational age, at 8.6% versus 14.8%, or a relative risk of 0.58 (P = .046).
But of concern, metformin was associated with an increased risk of small-for-gestational-age babies, at 12.9% versus 6.6% with placebo, or a relative risk of 1.96 (P = .03).
Dr. Feig suggested that this may be due to a direct effect of metformin “because as we know metformin inhibits the mTOR pathway,” which is a “primary nutrient sensor in the placenta” and could “attenuate nutrient flux and fetal growth.”
She said it is not clear whether the small-for-gestational-age babies were “healthy or unhealthy.”
To investigate further, the team has launched the MiTy Kids study, which will follow the offspring in the MiTy trial to determine whether metformin during pregnancy is associated with a reduction in adiposity and improvement in insulin resistance in the babies at 2 years of age.
Who should be given metformin?
During the discussion, Helen R. Murphy, MD, PhD, Norwich Medical School, University of East Anglia, England, asked whether Dr. Feig would recommend continuing metformin in pregnancy if it was started preconception for fertility issues rather than diabetes.
She replied: “If they don’t have diabetes and it’s simply for PCOS [polycystic ovary syndrome], then I have either stopped it as soon as they got pregnant or sometimes continued it through the first trimester, and then stopped.
“If the person has diabetes, however, I think given this work, for most people I would continue it,” she said.
The study was funded by the Canadian Institutes of Health Research, Lunenfeld-Tanenbaum Research Institute, and the University of Toronto. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
including reduced weight gain, reduced insulin doses, and fewer large-for-gestational-age babies, suggest the results of a randomized controlled trial.
However, the drug was associated with an increased risk of small-for-gestational-age babies, which poses the question as to risk versus benefit of metformin on the health of offspring.
“Better understanding of the short- and long-term implications of these effects on infants will be important to properly advise patients with type 2 diabetes contemplating use of metformin during pregnancy,” said lead author Denice S. Feig, MD, Mount Sinai Hospital, Toronto.
The research was presented at the Diabetes UK Professional Conference: Online Series on Nov. 17 and recently published in The Lancet Diabetes & Endocrinology.
Summing up, Dr. Feig said that, on balance, she would be inclined to give metformin to most pregnant women with type 2 diabetes, perhaps with the exception of those who may have risk factors for small-for-gestational-age babies; for example, women who’ve had intrauterine growth restriction, who are smokers, and have significant renal disease, or have a lower body mass index.
Increased prevalence of type 2 diabetes in pregnancy
Dr. Feig said that across the developed world there have been huge increases in the prevalence of type 2 diabetes in pregnancy in recent years.
Insulin is the standard treatment for the management of type 2 diabetes in pregnancy, but these women have marked insulin resistance that worsens in pregnancy, which means their insulin requirements increase, leading to weight gain, painful injections, high cost, and noncompliance.
So despite treatment with insulin, these women continue to face increased rates of adverse maternal and fetal outcomes.
And although metformin is increasingly being used in women with type 2 diabetes during pregnancy, there is a scarcity of data on the benefits and harms of metformin use on pregnancy outcomes in these women.
The MiTy trial was therefore undertaken to determine whether metformin could improve outcomes.
The team recruited 502 women from 29 sites in Canada and Australia who had type 2 diabetes prior to pregnancy or were diagnosed during pregnancy, before 20 weeks’ gestation. The women were randomized to metformin 1 g twice daily or placebo, in addition to their usual insulin regimen, at between 6 and 28 weeks’ gestation.
Type 2 diabetes was diagnosed prior to pregnancy in 83% of women in the metformin group and in 90% of those assigned to placebo. The mean hemoglobin A1c level at randomization was 47 mmol/mol (6.5%) in both groups.
The average maternal age at baseline was approximately 35 years and mean gestational age at randomization was 16 weeks. Mean prepregnancy BMI was approximately 34 kg/m2.
Of note, only 30% were of European ethnicity.
Less weight gain, lower A1c, less insulin needed with metformin
Dr. Feig reported that there was no significant difference between the treatment groups in terms of the proportion of women with the composite primary outcome of pregnancy loss, preterm birth, birth injury, respiratory distress, neonatal hypoglycemia, or admission to neonatal intensive care lasting more than 24 hours (P = 0.86).
However, women in the metformin group had significantly less overall weight gain during pregnancy than did those in the placebo group, at –1.8 kg (P < .0001).
They also had a significantly lower last A1c level in pregnancy, at 41 mmol/mol (5.9%) versus 43.2 mmol/mol (6.1%) in those given placebo (P = .015), and required fewer insulin doses, at 1.1 versus 1.5 units/kg/day (P < .0001), which translated to a reduction of almost 44 units/day.
Women given metformin were also less likely to require Cesarean section delivery, at 53.4% versus 62.7% in the placebo group (P = .03), although there was no difference between groups in terms of gestational hypertension or preeclampsia.
The most common adverse events were gastrointestinal complications, which occurred in 27.3% of women in the metformin group and 22.3% of those given placebo.
There were no significant differences between the metformin and placebo groups in rates of pregnancy loss (P = .81), preterm birth (P = .16), birth injury (P = .37), respiratory distress (P = .49), and congenital anomalies (P = .16).
Average birth weight lower with metformin
However, Dr. Feig showed that the average birth weight was lower for offspring of women given metformin than those assigned to placebo, at 3.2 kg (7.05 lb) versus 3.4 kg (7.4 lb) (P = .002).
Women given metformin were also less likely to have a baby with a birth weight of 4 kg (8.8 lb) or more, at 12.1% versus 19.2%, or a relative risk of 0.65 (P = .046), and a baby that was extremely large for gestational age, at 8.6% versus 14.8%, or a relative risk of 0.58 (P = .046).
But of concern, metformin was associated with an increased risk of small-for-gestational-age babies, at 12.9% versus 6.6% with placebo, or a relative risk of 1.96 (P = .03).
Dr. Feig suggested that this may be due to a direct effect of metformin “because as we know metformin inhibits the mTOR pathway,” which is a “primary nutrient sensor in the placenta” and could “attenuate nutrient flux and fetal growth.”
She said it is not clear whether the small-for-gestational-age babies were “healthy or unhealthy.”
To investigate further, the team has launched the MiTy Kids study, which will follow the offspring in the MiTy trial to determine whether metformin during pregnancy is associated with a reduction in adiposity and improvement in insulin resistance in the babies at 2 years of age.
Who should be given metformin?
During the discussion, Helen R. Murphy, MD, PhD, Norwich Medical School, University of East Anglia, England, asked whether Dr. Feig would recommend continuing metformin in pregnancy if it was started preconception for fertility issues rather than diabetes.
She replied: “If they don’t have diabetes and it’s simply for PCOS [polycystic ovary syndrome], then I have either stopped it as soon as they got pregnant or sometimes continued it through the first trimester, and then stopped.
“If the person has diabetes, however, I think given this work, for most people I would continue it,” she said.
The study was funded by the Canadian Institutes of Health Research, Lunenfeld-Tanenbaum Research Institute, and the University of Toronto. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
including reduced weight gain, reduced insulin doses, and fewer large-for-gestational-age babies, suggest the results of a randomized controlled trial.
However, the drug was associated with an increased risk of small-for-gestational-age babies, which poses the question as to risk versus benefit of metformin on the health of offspring.
“Better understanding of the short- and long-term implications of these effects on infants will be important to properly advise patients with type 2 diabetes contemplating use of metformin during pregnancy,” said lead author Denice S. Feig, MD, Mount Sinai Hospital, Toronto.
The research was presented at the Diabetes UK Professional Conference: Online Series on Nov. 17 and recently published in The Lancet Diabetes & Endocrinology.
Summing up, Dr. Feig said that, on balance, she would be inclined to give metformin to most pregnant women with type 2 diabetes, perhaps with the exception of those who may have risk factors for small-for-gestational-age babies; for example, women who’ve had intrauterine growth restriction, who are smokers, and have significant renal disease, or have a lower body mass index.
Increased prevalence of type 2 diabetes in pregnancy
Dr. Feig said that across the developed world there have been huge increases in the prevalence of type 2 diabetes in pregnancy in recent years.
Insulin is the standard treatment for the management of type 2 diabetes in pregnancy, but these women have marked insulin resistance that worsens in pregnancy, which means their insulin requirements increase, leading to weight gain, painful injections, high cost, and noncompliance.
So despite treatment with insulin, these women continue to face increased rates of adverse maternal and fetal outcomes.
And although metformin is increasingly being used in women with type 2 diabetes during pregnancy, there is a scarcity of data on the benefits and harms of metformin use on pregnancy outcomes in these women.
The MiTy trial was therefore undertaken to determine whether metformin could improve outcomes.
The team recruited 502 women from 29 sites in Canada and Australia who had type 2 diabetes prior to pregnancy or were diagnosed during pregnancy, before 20 weeks’ gestation. The women were randomized to metformin 1 g twice daily or placebo, in addition to their usual insulin regimen, at between 6 and 28 weeks’ gestation.
Type 2 diabetes was diagnosed prior to pregnancy in 83% of women in the metformin group and in 90% of those assigned to placebo. The mean hemoglobin A1c level at randomization was 47 mmol/mol (6.5%) in both groups.
The average maternal age at baseline was approximately 35 years and mean gestational age at randomization was 16 weeks. Mean prepregnancy BMI was approximately 34 kg/m2.
Of note, only 30% were of European ethnicity.
Less weight gain, lower A1c, less insulin needed with metformin
Dr. Feig reported that there was no significant difference between the treatment groups in terms of the proportion of women with the composite primary outcome of pregnancy loss, preterm birth, birth injury, respiratory distress, neonatal hypoglycemia, or admission to neonatal intensive care lasting more than 24 hours (P = 0.86).
However, women in the metformin group had significantly less overall weight gain during pregnancy than did those in the placebo group, at –1.8 kg (P < .0001).
They also had a significantly lower last A1c level in pregnancy, at 41 mmol/mol (5.9%) versus 43.2 mmol/mol (6.1%) in those given placebo (P = .015), and required fewer insulin doses, at 1.1 versus 1.5 units/kg/day (P < .0001), which translated to a reduction of almost 44 units/day.
Women given metformin were also less likely to require Cesarean section delivery, at 53.4% versus 62.7% in the placebo group (P = .03), although there was no difference between groups in terms of gestational hypertension or preeclampsia.
The most common adverse events were gastrointestinal complications, which occurred in 27.3% of women in the metformin group and 22.3% of those given placebo.
There were no significant differences between the metformin and placebo groups in rates of pregnancy loss (P = .81), preterm birth (P = .16), birth injury (P = .37), respiratory distress (P = .49), and congenital anomalies (P = .16).
Average birth weight lower with metformin
However, Dr. Feig showed that the average birth weight was lower for offspring of women given metformin than those assigned to placebo, at 3.2 kg (7.05 lb) versus 3.4 kg (7.4 lb) (P = .002).
Women given metformin were also less likely to have a baby with a birth weight of 4 kg (8.8 lb) or more, at 12.1% versus 19.2%, or a relative risk of 0.65 (P = .046), and a baby that was extremely large for gestational age, at 8.6% versus 14.8%, or a relative risk of 0.58 (P = .046).
But of concern, metformin was associated with an increased risk of small-for-gestational-age babies, at 12.9% versus 6.6% with placebo, or a relative risk of 1.96 (P = .03).
Dr. Feig suggested that this may be due to a direct effect of metformin “because as we know metformin inhibits the mTOR pathway,” which is a “primary nutrient sensor in the placenta” and could “attenuate nutrient flux and fetal growth.”
She said it is not clear whether the small-for-gestational-age babies were “healthy or unhealthy.”
To investigate further, the team has launched the MiTy Kids study, which will follow the offspring in the MiTy trial to determine whether metformin during pregnancy is associated with a reduction in adiposity and improvement in insulin resistance in the babies at 2 years of age.
Who should be given metformin?
During the discussion, Helen R. Murphy, MD, PhD, Norwich Medical School, University of East Anglia, England, asked whether Dr. Feig would recommend continuing metformin in pregnancy if it was started preconception for fertility issues rather than diabetes.
She replied: “If they don’t have diabetes and it’s simply for PCOS [polycystic ovary syndrome], then I have either stopped it as soon as they got pregnant or sometimes continued it through the first trimester, and then stopped.
“If the person has diabetes, however, I think given this work, for most people I would continue it,” she said.
The study was funded by the Canadian Institutes of Health Research, Lunenfeld-Tanenbaum Research Institute, and the University of Toronto. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Immunodeficiency strongly linked to mental illness, suicidal behavior
Patients with a primary humoral immunodeficiency (PID) are 91% more likely to have a psychiatric disorder and 84% more likely to exhibit suicidal behavior, compared against those without the condition, new research shows.
Results showed that this association, which was stronger in women, could not be fully explained by comorbid autoimmune diseases or by familial confounding.
These findings have important clinical implications, study investigator Josef Isung, MD, PhD, Centre for Psychiatry Research, Karolinska Institute, Stockholm, Sweden, told Medscape Medical News.
Clinicians managing patients with PID “should be aware of this increased association with psychiatric disorders and perhaps screen for them,” said Isung.
The study was published in the November issue of JAMA Psychiatry.
Registry study
Mounting evidence suggests immune disruption plays a role in psychiatric disorders through a range of mechanisms, including altered neurodevelopment. However, little is known about the neuropsychiatric consequences resulting from the underproduction of homeostatic antibodies.
They’re associated with an increased risk for recurrent infections and of developing autoimmune diseases.
The immunodeficiency can be severe, even life threatening, but can also be relatively mild. One of the less severe PID types is selective IgA deficiency, which is linked to increased infections within the mucosa-associated lymphoid tissue (MALT), an important immune barrier.
Experts have long suspected that infections within the MALT are associated with certain forms of psychopathology in children, particularly obsessive-compulsive disorder and chronic tic disorders.
While patients with this selective IgA subtype may be at some increased risk for infection and autoimmune disease, their overall health otherwise is good, said Isung.
The prevalence of PIDs ranges from about 1:250 to 1:20,000, depending on the type of humoral immunodeficiency, although most would fall into the relatively rare category, he added.
Using several linked national Swedish registries, researchers identified individuals with any PID diagnosis affecting immunoglobulin levels, their full siblings, and those with a lifetime diagnosis of selective IgA deficiency. In addition, they collected data on autoimmune diseases.
The study outcome was a lifetime record of a psychiatric disorder, a suicide attempt, or death by suicide.
Strong link to autism
Researchers identified 8378 patients (59% women) with PID affecting immunoglobulin levels (median age at first diagnosis, 47.8 years). They compared this group with almost 14.3 million subjects without PID.
In those with PID, 27.6% had an autoimmune disease vs 6.8% of those without PID, a statistically significant difference (P < .001).
About 20.5% of those with PID and 10.7% of unexposed subjects had at least one diagnosis of a psychiatric disorder.
In a model adjusted for year of birth, sex, and history of autoimmune disease, subjects with PID had a 91% higher likelihood of any psychiatric disorder (adjusted odds ratio [AOR] 1.91; 95% CI, 1.81 - 2.01; P < .001) vs their counterparts without PID.
The AORs for individual psychiatric disorders ranged from 1.34 (95% CI, 1.17 - 1.54; P < .001) for schizophrenia and other psychotic disorders to 2.99 (95% CI, 2.42 - 3.70; P < .001) for autism spectrum disorders (ASDs)
It’s unclear why the association with PID was strongest for autism, “but being a neurodevelopmental disorder, maybe autism is logically more associated with this type of disruption,” said Isung.
Research suggests that immunologic disruption may play a role in ASD, either through altered maternal immune function in utero or through immune disruption after birth, the researchers note.
Compared to those without PID, individuals with it had a significantly increased likelihood of any suicidal behavior (AOR, 1.84; 95% CI, 1.66 - 2.04, P < .001) as well as individual outcomes of death by suicide and suicide attempts.
The association with psychiatric disorders and suicidal behavior was markedly stronger for exposure to both PID and autoimmune disease than for exposure to either of these alone, which suggest an additive effect for these immune-related conditions.
Sex differences
“It was unclear to us why women seemed particularly vulnerable,” said Isung. He noted that PIDs are generally about as common in women as in men, but women tend to have higher rates of psychiatric disorders.
The analysis of the sibling cohort also revealed an elevated risk for psychiatric disorders, including ASD and suicidal behavior, but to a lesser degree.
“From this we could infer that at least part of the associations would be genetic, but part would be related to the disruption in itself,” said Isung.
An analysis examining selective IgA subtype also revealed a link with psychiatric disorders and suicidal behavior, suggesting this link is not exclusive to severe PID cases.
“Our conclusion here was that it seems like PID itself, or the immune disruption in itself, could explain the association rather than the burden of illness,” said Isung.
However, he acknowledged that the long-term stress and mental health fallout of having a chronic illness like PID may also explain some of the increased risk for psychiatric disorders.
This study, he said, provides more evidence that immune disruptions affect neurodevelopment and the brain. However, he added, the underlying mechanism still isn’t fully understood.
The results highlight the need to raise awareness of the association between immunodeficiency and mental illness, including suicidality among clinicians, patients, and advocates.
These findings may also have implications in patients with other immune deficiencies, said Isung, noting, “it would be interesting to further explore associations with other immunocompromised populations.”
No surprises
Commenting on the findings for Medscape Medical News, Igor Galynker, MD, professor of psychiatry at Icahn School of Medicine at Mount Sinai, New York City, said the study was “very well-done” and used “reliable and well-controlled” databases.
However, he added, the results “are neither particularly dramatic nor conclusive” as it makes sense that medical illnesses like PID would “increase risk of psychopathology,” said Galynker.
PID patients are much more likely to have contact with clinicians and to receive a psychiatric diagnosis, he said.
“People with a chronic illness are more stressed and generally have high incidences of depression, anxiety, and suicidal behavior. In addition to that, they may be more likely to be diagnosed with those conditions because they see a clinician more frequently.”
However, that reasoning doesn’t apply to autism, which manifests in early childhood and so is unlikely to be the result of stress, said Galynker, which is why he believes the finding that ASD is the psychiatric outcome most strongly associated with PID is “the most convincing.”
Galynker wasn’t surprised that the association between PID and psychiatric illnesses, and suicidal behaviors, was stronger among women.
“Women attempt suicide four times more often than men to begin with, so you would expect this to be more pronounced” in those with PID.
The study was supported by grants from the Centre for Psychiatry Research, Department of Clinical Neuroscience, Karolinska Institute; Stockholm Care Services; the Soderstrom Konig Foundation; and the Fredrik & Ingrid Thurings Foundation. Isung and Galynker have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with a primary humoral immunodeficiency (PID) are 91% more likely to have a psychiatric disorder and 84% more likely to exhibit suicidal behavior, compared against those without the condition, new research shows.
Results showed that this association, which was stronger in women, could not be fully explained by comorbid autoimmune diseases or by familial confounding.
These findings have important clinical implications, study investigator Josef Isung, MD, PhD, Centre for Psychiatry Research, Karolinska Institute, Stockholm, Sweden, told Medscape Medical News.
Clinicians managing patients with PID “should be aware of this increased association with psychiatric disorders and perhaps screen for them,” said Isung.
The study was published in the November issue of JAMA Psychiatry.
Registry study
Mounting evidence suggests immune disruption plays a role in psychiatric disorders through a range of mechanisms, including altered neurodevelopment. However, little is known about the neuropsychiatric consequences resulting from the underproduction of homeostatic antibodies.
They’re associated with an increased risk for recurrent infections and of developing autoimmune diseases.
The immunodeficiency can be severe, even life threatening, but can also be relatively mild. One of the less severe PID types is selective IgA deficiency, which is linked to increased infections within the mucosa-associated lymphoid tissue (MALT), an important immune barrier.
Experts have long suspected that infections within the MALT are associated with certain forms of psychopathology in children, particularly obsessive-compulsive disorder and chronic tic disorders.
While patients with this selective IgA subtype may be at some increased risk for infection and autoimmune disease, their overall health otherwise is good, said Isung.
The prevalence of PIDs ranges from about 1:250 to 1:20,000, depending on the type of humoral immunodeficiency, although most would fall into the relatively rare category, he added.
Using several linked national Swedish registries, researchers identified individuals with any PID diagnosis affecting immunoglobulin levels, their full siblings, and those with a lifetime diagnosis of selective IgA deficiency. In addition, they collected data on autoimmune diseases.
The study outcome was a lifetime record of a psychiatric disorder, a suicide attempt, or death by suicide.
Strong link to autism
Researchers identified 8378 patients (59% women) with PID affecting immunoglobulin levels (median age at first diagnosis, 47.8 years). They compared this group with almost 14.3 million subjects without PID.
In those with PID, 27.6% had an autoimmune disease vs 6.8% of those without PID, a statistically significant difference (P < .001).
About 20.5% of those with PID and 10.7% of unexposed subjects had at least one diagnosis of a psychiatric disorder.
In a model adjusted for year of birth, sex, and history of autoimmune disease, subjects with PID had a 91% higher likelihood of any psychiatric disorder (adjusted odds ratio [AOR] 1.91; 95% CI, 1.81 - 2.01; P < .001) vs their counterparts without PID.
The AORs for individual psychiatric disorders ranged from 1.34 (95% CI, 1.17 - 1.54; P < .001) for schizophrenia and other psychotic disorders to 2.99 (95% CI, 2.42 - 3.70; P < .001) for autism spectrum disorders (ASDs)
It’s unclear why the association with PID was strongest for autism, “but being a neurodevelopmental disorder, maybe autism is logically more associated with this type of disruption,” said Isung.
Research suggests that immunologic disruption may play a role in ASD, either through altered maternal immune function in utero or through immune disruption after birth, the researchers note.
Compared to those without PID, individuals with it had a significantly increased likelihood of any suicidal behavior (AOR, 1.84; 95% CI, 1.66 - 2.04, P < .001) as well as individual outcomes of death by suicide and suicide attempts.
The association with psychiatric disorders and suicidal behavior was markedly stronger for exposure to both PID and autoimmune disease than for exposure to either of these alone, which suggest an additive effect for these immune-related conditions.
Sex differences
“It was unclear to us why women seemed particularly vulnerable,” said Isung. He noted that PIDs are generally about as common in women as in men, but women tend to have higher rates of psychiatric disorders.
The analysis of the sibling cohort also revealed an elevated risk for psychiatric disorders, including ASD and suicidal behavior, but to a lesser degree.
“From this we could infer that at least part of the associations would be genetic, but part would be related to the disruption in itself,” said Isung.
An analysis examining selective IgA subtype also revealed a link with psychiatric disorders and suicidal behavior, suggesting this link is not exclusive to severe PID cases.
“Our conclusion here was that it seems like PID itself, or the immune disruption in itself, could explain the association rather than the burden of illness,” said Isung.
However, he acknowledged that the long-term stress and mental health fallout of having a chronic illness like PID may also explain some of the increased risk for psychiatric disorders.
This study, he said, provides more evidence that immune disruptions affect neurodevelopment and the brain. However, he added, the underlying mechanism still isn’t fully understood.
The results highlight the need to raise awareness of the association between immunodeficiency and mental illness, including suicidality among clinicians, patients, and advocates.
These findings may also have implications in patients with other immune deficiencies, said Isung, noting, “it would be interesting to further explore associations with other immunocompromised populations.”
No surprises
Commenting on the findings for Medscape Medical News, Igor Galynker, MD, professor of psychiatry at Icahn School of Medicine at Mount Sinai, New York City, said the study was “very well-done” and used “reliable and well-controlled” databases.
However, he added, the results “are neither particularly dramatic nor conclusive” as it makes sense that medical illnesses like PID would “increase risk of psychopathology,” said Galynker.
PID patients are much more likely to have contact with clinicians and to receive a psychiatric diagnosis, he said.
“People with a chronic illness are more stressed and generally have high incidences of depression, anxiety, and suicidal behavior. In addition to that, they may be more likely to be diagnosed with those conditions because they see a clinician more frequently.”
However, that reasoning doesn’t apply to autism, which manifests in early childhood and so is unlikely to be the result of stress, said Galynker, which is why he believes the finding that ASD is the psychiatric outcome most strongly associated with PID is “the most convincing.”
Galynker wasn’t surprised that the association between PID and psychiatric illnesses, and suicidal behaviors, was stronger among women.
“Women attempt suicide four times more often than men to begin with, so you would expect this to be more pronounced” in those with PID.
The study was supported by grants from the Centre for Psychiatry Research, Department of Clinical Neuroscience, Karolinska Institute; Stockholm Care Services; the Soderstrom Konig Foundation; and the Fredrik & Ingrid Thurings Foundation. Isung and Galynker have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with a primary humoral immunodeficiency (PID) are 91% more likely to have a psychiatric disorder and 84% more likely to exhibit suicidal behavior, compared against those without the condition, new research shows.
Results showed that this association, which was stronger in women, could not be fully explained by comorbid autoimmune diseases or by familial confounding.
These findings have important clinical implications, study investigator Josef Isung, MD, PhD, Centre for Psychiatry Research, Karolinska Institute, Stockholm, Sweden, told Medscape Medical News.
Clinicians managing patients with PID “should be aware of this increased association with psychiatric disorders and perhaps screen for them,” said Isung.
The study was published in the November issue of JAMA Psychiatry.
Registry study
Mounting evidence suggests immune disruption plays a role in psychiatric disorders through a range of mechanisms, including altered neurodevelopment. However, little is known about the neuropsychiatric consequences resulting from the underproduction of homeostatic antibodies.
They’re associated with an increased risk for recurrent infections and of developing autoimmune diseases.
The immunodeficiency can be severe, even life threatening, but can also be relatively mild. One of the less severe PID types is selective IgA deficiency, which is linked to increased infections within the mucosa-associated lymphoid tissue (MALT), an important immune barrier.
Experts have long suspected that infections within the MALT are associated with certain forms of psychopathology in children, particularly obsessive-compulsive disorder and chronic tic disorders.
While patients with this selective IgA subtype may be at some increased risk for infection and autoimmune disease, their overall health otherwise is good, said Isung.
The prevalence of PIDs ranges from about 1:250 to 1:20,000, depending on the type of humoral immunodeficiency, although most would fall into the relatively rare category, he added.
Using several linked national Swedish registries, researchers identified individuals with any PID diagnosis affecting immunoglobulin levels, their full siblings, and those with a lifetime diagnosis of selective IgA deficiency. In addition, they collected data on autoimmune diseases.
The study outcome was a lifetime record of a psychiatric disorder, a suicide attempt, or death by suicide.
Strong link to autism
Researchers identified 8378 patients (59% women) with PID affecting immunoglobulin levels (median age at first diagnosis, 47.8 years). They compared this group with almost 14.3 million subjects without PID.
In those with PID, 27.6% had an autoimmune disease vs 6.8% of those without PID, a statistically significant difference (P < .001).
About 20.5% of those with PID and 10.7% of unexposed subjects had at least one diagnosis of a psychiatric disorder.
In a model adjusted for year of birth, sex, and history of autoimmune disease, subjects with PID had a 91% higher likelihood of any psychiatric disorder (adjusted odds ratio [AOR] 1.91; 95% CI, 1.81 - 2.01; P < .001) vs their counterparts without PID.
The AORs for individual psychiatric disorders ranged from 1.34 (95% CI, 1.17 - 1.54; P < .001) for schizophrenia and other psychotic disorders to 2.99 (95% CI, 2.42 - 3.70; P < .001) for autism spectrum disorders (ASDs)
It’s unclear why the association with PID was strongest for autism, “but being a neurodevelopmental disorder, maybe autism is logically more associated with this type of disruption,” said Isung.
Research suggests that immunologic disruption may play a role in ASD, either through altered maternal immune function in utero or through immune disruption after birth, the researchers note.
Compared to those without PID, individuals with it had a significantly increased likelihood of any suicidal behavior (AOR, 1.84; 95% CI, 1.66 - 2.04, P < .001) as well as individual outcomes of death by suicide and suicide attempts.
The association with psychiatric disorders and suicidal behavior was markedly stronger for exposure to both PID and autoimmune disease than for exposure to either of these alone, which suggest an additive effect for these immune-related conditions.
Sex differences
“It was unclear to us why women seemed particularly vulnerable,” said Isung. He noted that PIDs are generally about as common in women as in men, but women tend to have higher rates of psychiatric disorders.
The analysis of the sibling cohort also revealed an elevated risk for psychiatric disorders, including ASD and suicidal behavior, but to a lesser degree.
“From this we could infer that at least part of the associations would be genetic, but part would be related to the disruption in itself,” said Isung.
An analysis examining selective IgA subtype also revealed a link with psychiatric disorders and suicidal behavior, suggesting this link is not exclusive to severe PID cases.
“Our conclusion here was that it seems like PID itself, or the immune disruption in itself, could explain the association rather than the burden of illness,” said Isung.
However, he acknowledged that the long-term stress and mental health fallout of having a chronic illness like PID may also explain some of the increased risk for psychiatric disorders.
This study, he said, provides more evidence that immune disruptions affect neurodevelopment and the brain. However, he added, the underlying mechanism still isn’t fully understood.
The results highlight the need to raise awareness of the association between immunodeficiency and mental illness, including suicidality among clinicians, patients, and advocates.
These findings may also have implications in patients with other immune deficiencies, said Isung, noting, “it would be interesting to further explore associations with other immunocompromised populations.”
No surprises
Commenting on the findings for Medscape Medical News, Igor Galynker, MD, professor of psychiatry at Icahn School of Medicine at Mount Sinai, New York City, said the study was “very well-done” and used “reliable and well-controlled” databases.
However, he added, the results “are neither particularly dramatic nor conclusive” as it makes sense that medical illnesses like PID would “increase risk of psychopathology,” said Galynker.
PID patients are much more likely to have contact with clinicians and to receive a psychiatric diagnosis, he said.
“People with a chronic illness are more stressed and generally have high incidences of depression, anxiety, and suicidal behavior. In addition to that, they may be more likely to be diagnosed with those conditions because they see a clinician more frequently.”
However, that reasoning doesn’t apply to autism, which manifests in early childhood and so is unlikely to be the result of stress, said Galynker, which is why he believes the finding that ASD is the psychiatric outcome most strongly associated with PID is “the most convincing.”
Galynker wasn’t surprised that the association between PID and psychiatric illnesses, and suicidal behaviors, was stronger among women.
“Women attempt suicide four times more often than men to begin with, so you would expect this to be more pronounced” in those with PID.
The study was supported by grants from the Centre for Psychiatry Research, Department of Clinical Neuroscience, Karolinska Institute; Stockholm Care Services; the Soderstrom Konig Foundation; and the Fredrik & Ingrid Thurings Foundation. Isung and Galynker have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Sedentary postmenopausal women have higher heart failure risk
The more time older women spent sitting or lying down, the more likely their risk of hospitalization for heart failure, based on data from more than 80,000 postmenopausal women.
The 2018 Physical Activity Guidelines show evidence of the impact of physical activity on reducing heart failure risk, but the association between activity, sedentary behavior (SB) and heart failure (HF) in older women in particular has not been well studied, wrote Michael J. LaMonte, PhD, MPH, of the State University of New York at Buffalo, and colleagues in a study published in Circulation: Heart Failure. “Given the high prevalence of prolonged sedentary time among U.S. adults aged 65 and older, among whom HF burden is substantial, understanding the role SB has in HF development is relevant to future HF prevention strategies,” the researchers wrote.
The researchers identified 80,982 women aged 50-79 years who were enrolled in the Women’s Health Initiative Observational Study, had no known HF, and could walk at least one block unassisted. The average follow-up period was 9 years, and a total of 1,402 women were hospitalized for heart failure during the period of time they were observed.
The time spent sedentary (combined sitting or lying down) was divided into tertiles of 6.5 hours or less, 6.6-9.5 hours, and more than 9.5 hours. Time spent sitting was divided into tertiles of 4.5 hours or less; 4.6-8.5 hours; and more than 8.5 hours.
Heart failure risk goes up with more down time
After controlling for multiple variables including age, race, education, income, smoking status alcohol use, menopausal hormone therapy, and hysterectomy status, the researchers found that patients in the second tertile for sedentary behavior had a significantly increased heart failure risk than patients in the first tertile for sedentary behavior. This risk was even greater for patients falling in the third tertile for sedentary behavior. Odds ratios were 1.00 (referent), 1.15, and 1.42 for the lowest to highest tertiles for total sedentary behavior, respectively, and 1.00 (referent), 1.14, and 1.54 for sitting (P < .001 for both total sedentary behavior and sitting only).
The trends remained significant after controlling for comorbidities including MI and coronary revascularization, and the associations were similar among categories of women with additional HF risk factors, including body mass index, diabetes, hypertension, and coronary heart disease.
Notably, the association between hours spent sitting or lying down and HF risk persisted even in women who met recommended activity levels, the researchers wrote.
The study findings were limited by the use of self-reports and by the inability to evaluate SB patterns or SB and HF subtypes, the researchers noted. However, the results were strengthened by the large sample size, use of time-varying SB exposure, and extensive controlling, and the data support the risk of increased SB on adverse cardiovascular outcomes.
“Results of this study underscore the need for effective strategies to reduce daily SB time, in addition to increasing recreational physical activity, as part of population efforts for HF prevention,” they concluded.
Clinicians know the value of a physically active lifestyle for heart health, said lead author Dr. LaMonte in a statement accompanying the study’s release. “However, our study clearly shows that we also need to increase efforts to reduce daily sedentary time and encourage adults to frequently interrupt their sedentary time. This does not necessarily require an extended bout of physical activity; it might simply be standing up for 5 minutes or standing and moving one’s feet in place.
“We do not have sufficient evidence on the best approach to recommend for interrupting sedentary time. However, accumulating data suggest that habitual activities such as steps taken during household and other activities of daily living are an important aspect of cardiovascular disease prevention and healthy aging,” Dr. LaMonte added.
Promote more movement and less sitting
“This is the first study to assess sedentary time and the risk for incident heart failure hospitalization in postmenopausal women,” said Robert H. Hopkins Jr., MD, of the University of Arkansas for Medical Sciences, Little Rock, in an interview.
“Heart failure is the cause of approximately 35% of cardiovascular mortalities in women, and sedentary behaviors are common in older adults,” he noted.
Kashif J. Piracha, MD, of Houston Methodist Willowbrook Hospital, agreed that there is a lack of existing data looking at the relationship between sedentary behavior and the risk of the development of heart failure in postmenopausal women. In an interview, he cited this as a reason “it was important to conduct this study.”
Dr. Hopkins added that he was not surprised by the study results “There are a number of studies which have demonstrated reduction in risk for heart failure in men and in combined populations of men and women with increased physical activity.” There are fewer data (but similar outcomes) in studies of men with increased levels of sedentary behaviors, he said.
“This study adds one more reason that other clinicians in primary care and me need to encourage our older patients to get up and move,” said Dr. Hopkins, who also serves on the editorial advisory board of Internal Medicine News. “Many of us have focused our efforts in the past on achieving exercise goals and this study provides a foundation for a recommendation that ‘it is not just about exercise;’ we need to also encourage our patients to minimize their time in sedentary pursuits in addition to exercise if we are to optimize their health into older age.”
Dr. Hopkins noted that the large size of the study was a strength, but the observational design and use of patient surveys were limitations.
“We need further studies to better tease out whether there are risk differences in different sedentary behavior patterns, whether this applies across heart failure with reduced ejection fraction versus heart failure with preserved ejection fraction, and whether there are additional ways we can mitigate these risks as our society ages,” he said.
Findings differ from California Men’s Health Study’s
“The results corroborate the fact that there is less risk of heart failure in physically active patients,” Dr. Piracha noted.
The message for clinicians is to encourage postmenopausal female patients to engage in physical activity as much as possible, said Dr. Piracha. “Also, it appears that in this population, even with good physical activity, prolonged sedentary behavior of more than 8.5 hours a day was still associated with a higher risk of incident HF hospitalization. Therefore, a case can be made to focus on carrying out physical activity with an intensity that can be sustained for longer, rather than shorter periods of time.”
Notably, the finding of increased HF hospitalization in women who reported high amounts of physical activity but were still sedentary for more than 8.5 hours a day “is contrary to what was seen in the California Men’s Health Study.” In that study, “men with high physical activity levels who also had prolonged sitting time did not have increased risk of HF hospitalization,” Dr. Piracha noted. “Further research is needed to elucidate what hormonal or other factors contribute to this difference.”
The new study was supported by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Hopkins and Dr. Piracha had no financial conflicts to disclose.
SOURCE: LaMonte MJ et al. Circ Heart Fail. 2020 Nov 24. doi: 10.1161/CIRCHEARTFAILURE.120.007508.
The more time older women spent sitting or lying down, the more likely their risk of hospitalization for heart failure, based on data from more than 80,000 postmenopausal women.
The 2018 Physical Activity Guidelines show evidence of the impact of physical activity on reducing heart failure risk, but the association between activity, sedentary behavior (SB) and heart failure (HF) in older women in particular has not been well studied, wrote Michael J. LaMonte, PhD, MPH, of the State University of New York at Buffalo, and colleagues in a study published in Circulation: Heart Failure. “Given the high prevalence of prolonged sedentary time among U.S. adults aged 65 and older, among whom HF burden is substantial, understanding the role SB has in HF development is relevant to future HF prevention strategies,” the researchers wrote.
The researchers identified 80,982 women aged 50-79 years who were enrolled in the Women’s Health Initiative Observational Study, had no known HF, and could walk at least one block unassisted. The average follow-up period was 9 years, and a total of 1,402 women were hospitalized for heart failure during the period of time they were observed.
The time spent sedentary (combined sitting or lying down) was divided into tertiles of 6.5 hours or less, 6.6-9.5 hours, and more than 9.5 hours. Time spent sitting was divided into tertiles of 4.5 hours or less; 4.6-8.5 hours; and more than 8.5 hours.
Heart failure risk goes up with more down time
After controlling for multiple variables including age, race, education, income, smoking status alcohol use, menopausal hormone therapy, and hysterectomy status, the researchers found that patients in the second tertile for sedentary behavior had a significantly increased heart failure risk than patients in the first tertile for sedentary behavior. This risk was even greater for patients falling in the third tertile for sedentary behavior. Odds ratios were 1.00 (referent), 1.15, and 1.42 for the lowest to highest tertiles for total sedentary behavior, respectively, and 1.00 (referent), 1.14, and 1.54 for sitting (P < .001 for both total sedentary behavior and sitting only).
The trends remained significant after controlling for comorbidities including MI and coronary revascularization, and the associations were similar among categories of women with additional HF risk factors, including body mass index, diabetes, hypertension, and coronary heart disease.
Notably, the association between hours spent sitting or lying down and HF risk persisted even in women who met recommended activity levels, the researchers wrote.
The study findings were limited by the use of self-reports and by the inability to evaluate SB patterns or SB and HF subtypes, the researchers noted. However, the results were strengthened by the large sample size, use of time-varying SB exposure, and extensive controlling, and the data support the risk of increased SB on adverse cardiovascular outcomes.
“Results of this study underscore the need for effective strategies to reduce daily SB time, in addition to increasing recreational physical activity, as part of population efforts for HF prevention,” they concluded.
Clinicians know the value of a physically active lifestyle for heart health, said lead author Dr. LaMonte in a statement accompanying the study’s release. “However, our study clearly shows that we also need to increase efforts to reduce daily sedentary time and encourage adults to frequently interrupt their sedentary time. This does not necessarily require an extended bout of physical activity; it might simply be standing up for 5 minutes or standing and moving one’s feet in place.
“We do not have sufficient evidence on the best approach to recommend for interrupting sedentary time. However, accumulating data suggest that habitual activities such as steps taken during household and other activities of daily living are an important aspect of cardiovascular disease prevention and healthy aging,” Dr. LaMonte added.
Promote more movement and less sitting
“This is the first study to assess sedentary time and the risk for incident heart failure hospitalization in postmenopausal women,” said Robert H. Hopkins Jr., MD, of the University of Arkansas for Medical Sciences, Little Rock, in an interview.
“Heart failure is the cause of approximately 35% of cardiovascular mortalities in women, and sedentary behaviors are common in older adults,” he noted.
Kashif J. Piracha, MD, of Houston Methodist Willowbrook Hospital, agreed that there is a lack of existing data looking at the relationship between sedentary behavior and the risk of the development of heart failure in postmenopausal women. In an interview, he cited this as a reason “it was important to conduct this study.”
Dr. Hopkins added that he was not surprised by the study results “There are a number of studies which have demonstrated reduction in risk for heart failure in men and in combined populations of men and women with increased physical activity.” There are fewer data (but similar outcomes) in studies of men with increased levels of sedentary behaviors, he said.
“This study adds one more reason that other clinicians in primary care and me need to encourage our older patients to get up and move,” said Dr. Hopkins, who also serves on the editorial advisory board of Internal Medicine News. “Many of us have focused our efforts in the past on achieving exercise goals and this study provides a foundation for a recommendation that ‘it is not just about exercise;’ we need to also encourage our patients to minimize their time in sedentary pursuits in addition to exercise if we are to optimize their health into older age.”
Dr. Hopkins noted that the large size of the study was a strength, but the observational design and use of patient surveys were limitations.
“We need further studies to better tease out whether there are risk differences in different sedentary behavior patterns, whether this applies across heart failure with reduced ejection fraction versus heart failure with preserved ejection fraction, and whether there are additional ways we can mitigate these risks as our society ages,” he said.
Findings differ from California Men’s Health Study’s
“The results corroborate the fact that there is less risk of heart failure in physically active patients,” Dr. Piracha noted.
The message for clinicians is to encourage postmenopausal female patients to engage in physical activity as much as possible, said Dr. Piracha. “Also, it appears that in this population, even with good physical activity, prolonged sedentary behavior of more than 8.5 hours a day was still associated with a higher risk of incident HF hospitalization. Therefore, a case can be made to focus on carrying out physical activity with an intensity that can be sustained for longer, rather than shorter periods of time.”
Notably, the finding of increased HF hospitalization in women who reported high amounts of physical activity but were still sedentary for more than 8.5 hours a day “is contrary to what was seen in the California Men’s Health Study.” In that study, “men with high physical activity levels who also had prolonged sitting time did not have increased risk of HF hospitalization,” Dr. Piracha noted. “Further research is needed to elucidate what hormonal or other factors contribute to this difference.”
The new study was supported by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Hopkins and Dr. Piracha had no financial conflicts to disclose.
SOURCE: LaMonte MJ et al. Circ Heart Fail. 2020 Nov 24. doi: 10.1161/CIRCHEARTFAILURE.120.007508.
The more time older women spent sitting or lying down, the more likely their risk of hospitalization for heart failure, based on data from more than 80,000 postmenopausal women.
The 2018 Physical Activity Guidelines show evidence of the impact of physical activity on reducing heart failure risk, but the association between activity, sedentary behavior (SB) and heart failure (HF) in older women in particular has not been well studied, wrote Michael J. LaMonte, PhD, MPH, of the State University of New York at Buffalo, and colleagues in a study published in Circulation: Heart Failure. “Given the high prevalence of prolonged sedentary time among U.S. adults aged 65 and older, among whom HF burden is substantial, understanding the role SB has in HF development is relevant to future HF prevention strategies,” the researchers wrote.
The researchers identified 80,982 women aged 50-79 years who were enrolled in the Women’s Health Initiative Observational Study, had no known HF, and could walk at least one block unassisted. The average follow-up period was 9 years, and a total of 1,402 women were hospitalized for heart failure during the period of time they were observed.
The time spent sedentary (combined sitting or lying down) was divided into tertiles of 6.5 hours or less, 6.6-9.5 hours, and more than 9.5 hours. Time spent sitting was divided into tertiles of 4.5 hours or less; 4.6-8.5 hours; and more than 8.5 hours.
Heart failure risk goes up with more down time
After controlling for multiple variables including age, race, education, income, smoking status alcohol use, menopausal hormone therapy, and hysterectomy status, the researchers found that patients in the second tertile for sedentary behavior had a significantly increased heart failure risk than patients in the first tertile for sedentary behavior. This risk was even greater for patients falling in the third tertile for sedentary behavior. Odds ratios were 1.00 (referent), 1.15, and 1.42 for the lowest to highest tertiles for total sedentary behavior, respectively, and 1.00 (referent), 1.14, and 1.54 for sitting (P < .001 for both total sedentary behavior and sitting only).
The trends remained significant after controlling for comorbidities including MI and coronary revascularization, and the associations were similar among categories of women with additional HF risk factors, including body mass index, diabetes, hypertension, and coronary heart disease.
Notably, the association between hours spent sitting or lying down and HF risk persisted even in women who met recommended activity levels, the researchers wrote.
The study findings were limited by the use of self-reports and by the inability to evaluate SB patterns or SB and HF subtypes, the researchers noted. However, the results were strengthened by the large sample size, use of time-varying SB exposure, and extensive controlling, and the data support the risk of increased SB on adverse cardiovascular outcomes.
“Results of this study underscore the need for effective strategies to reduce daily SB time, in addition to increasing recreational physical activity, as part of population efforts for HF prevention,” they concluded.
Clinicians know the value of a physically active lifestyle for heart health, said lead author Dr. LaMonte in a statement accompanying the study’s release. “However, our study clearly shows that we also need to increase efforts to reduce daily sedentary time and encourage adults to frequently interrupt their sedentary time. This does not necessarily require an extended bout of physical activity; it might simply be standing up for 5 minutes or standing and moving one’s feet in place.
“We do not have sufficient evidence on the best approach to recommend for interrupting sedentary time. However, accumulating data suggest that habitual activities such as steps taken during household and other activities of daily living are an important aspect of cardiovascular disease prevention and healthy aging,” Dr. LaMonte added.
Promote more movement and less sitting
“This is the first study to assess sedentary time and the risk for incident heart failure hospitalization in postmenopausal women,” said Robert H. Hopkins Jr., MD, of the University of Arkansas for Medical Sciences, Little Rock, in an interview.
“Heart failure is the cause of approximately 35% of cardiovascular mortalities in women, and sedentary behaviors are common in older adults,” he noted.
Kashif J. Piracha, MD, of Houston Methodist Willowbrook Hospital, agreed that there is a lack of existing data looking at the relationship between sedentary behavior and the risk of the development of heart failure in postmenopausal women. In an interview, he cited this as a reason “it was important to conduct this study.”
Dr. Hopkins added that he was not surprised by the study results “There are a number of studies which have demonstrated reduction in risk for heart failure in men and in combined populations of men and women with increased physical activity.” There are fewer data (but similar outcomes) in studies of men with increased levels of sedentary behaviors, he said.
“This study adds one more reason that other clinicians in primary care and me need to encourage our older patients to get up and move,” said Dr. Hopkins, who also serves on the editorial advisory board of Internal Medicine News. “Many of us have focused our efforts in the past on achieving exercise goals and this study provides a foundation for a recommendation that ‘it is not just about exercise;’ we need to also encourage our patients to minimize their time in sedentary pursuits in addition to exercise if we are to optimize their health into older age.”
Dr. Hopkins noted that the large size of the study was a strength, but the observational design and use of patient surveys were limitations.
“We need further studies to better tease out whether there are risk differences in different sedentary behavior patterns, whether this applies across heart failure with reduced ejection fraction versus heart failure with preserved ejection fraction, and whether there are additional ways we can mitigate these risks as our society ages,” he said.
Findings differ from California Men’s Health Study’s
“The results corroborate the fact that there is less risk of heart failure in physically active patients,” Dr. Piracha noted.
The message for clinicians is to encourage postmenopausal female patients to engage in physical activity as much as possible, said Dr. Piracha. “Also, it appears that in this population, even with good physical activity, prolonged sedentary behavior of more than 8.5 hours a day was still associated with a higher risk of incident HF hospitalization. Therefore, a case can be made to focus on carrying out physical activity with an intensity that can be sustained for longer, rather than shorter periods of time.”
Notably, the finding of increased HF hospitalization in women who reported high amounts of physical activity but were still sedentary for more than 8.5 hours a day “is contrary to what was seen in the California Men’s Health Study.” In that study, “men with high physical activity levels who also had prolonged sitting time did not have increased risk of HF hospitalization,” Dr. Piracha noted. “Further research is needed to elucidate what hormonal or other factors contribute to this difference.”
The new study was supported by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Hopkins and Dr. Piracha had no financial conflicts to disclose.
SOURCE: LaMonte MJ et al. Circ Heart Fail. 2020 Nov 24. doi: 10.1161/CIRCHEARTFAILURE.120.007508.
FROM CIRCULATION: HEART FAILURE
Half of women treated for gynecologic cancers miss or skip doses of oral drugs
Oral agents are taking on an ever-greater role in the management of gynecologic cancers. However, women being treated for these cancers have less than ideal adherence to such medications, a new study has found – with just over half reporting taking them exactly as prescribed.
The findings reported in Obstetrics & Gynecology are consistent with reports from other populations taking oral anticancer agents, in which adherence is generally lower than with intravenous therapies.
For their research, Catherine Watson, MD, and colleagues at Duke University, Durham, N.C., recruited 100 women at their institution taking a variety of oral anticancer agents for uterine or ovarian cancer for 30 days or more (median time, 6 months). The women answered a questionnaire that measured adherence as well as health literacy, quality of life, and distress. The researchers also collected information on the subjects’ race, age, insurance type, medication burden, and medication costs.
Fourteen of the women in the study additionally underwent qualitative interviews about their experiences with oral anticancer drugs. The researchers also queried physicians and nurse practitioners about their thoughts on adherence.
Dr. Watson and colleagues reported that 54% of women self-reported perfect adherence to their medication in the previous week, while 21% had missed or skipped one dose, and 25% reported skipping or missing more than one dose.
The researchers saw no significant differences between the adherent and nonadherent groups corresponding with race, age, or other demographic or clinical characteristics, but they noted that their study was not powered to detect such associations. The small sample size and self-reported data were among this study’s limitations, Dr. Watson and colleagues acknowledged.
Interviews with patients revealed some surprising reasons for the less-than-optimal adherence, with 43% of women reporting feeling anxiety about the burden of administering medication at home. While patients acknowledged the convenience of oral regimens, some also expressed a wish for more physician contact and support. Some women who were nonadherent said they perceived the efficacy of oral agents to be less than intravenous therapies.
Physicians and nurse practitioners interviewed by the researchers “tended to assume that their patients were adherent to oral anticancer therapy because of the therapy’s importance, and many did not routinely ask their patients about adherence,” Dr. Watson and colleagues wrote.
Emma Rossi, MD, a gynecologic oncologist at the University of North Carolina at Chapel Hill, commented in an interview that the study highlighted “the importance of not generalizing our perception of patients. As providers we have to spend the time asking patients where they’re at and how they’re thinking about taking an oral medication, with special attention to their fears, their expectations, and their concerns. We should be touching base with them not just before starting drugs, but during the course of treatment.”
Dr. Rossi stressed that compliance in real-life settings is likely to be different from that seen in clinical trials of oral anticancer drugs. “It’s important to recognize that real-world efficacy of treatments may be different from trial efficacy. We see these differences a lot in medicine where studies show a large magnitude of effect that doesn’t play out in real life practice – because of factors like this.”
Some 57% of the women in the study were taking their medications as maintenance, while the rest were taking the medications as active treatment. This too might have an effect on adherence, she said, with the active-treatment group potentially more motivated to maintain perfect adherence.
“You see in the interviews that doctors assume patients would be compliant because of the seriousness of the disease,” Dr. Rossi said. “But some patients said they perceived oral drugs as less strong or effective. If a patient is cancer free on a scan and doesn’t have measurable disease, prescribing an oral medication may be sending the subliminal message that it’s not as important.”
Physicians “may need to take the extra steps to individualize our counseling of patients – especially with therapies that they’re responsible for administering,” Dr. Rossi continued. “As this study shows, every patient sees treatment through her own individual lens. We really need to meet them where they’re at to make them comfortable with their treatment and optimize compliance.”
Dr. Watson and colleagues’ study was supported by their institution. One coauthor reported financial ties with drug manufacturers in the form of grant and clinical trial support and honoraria. Another coauthor is the member of various boards and steering committees, which are uncompensated. Dr. Rossi reported no financial conflicts of interest.
SOURCE: Watson C et al. Obstet Gynecol. 2020. doi: 10.1097/AOG.0000000000004170.
Oral agents are taking on an ever-greater role in the management of gynecologic cancers. However, women being treated for these cancers have less than ideal adherence to such medications, a new study has found – with just over half reporting taking them exactly as prescribed.
The findings reported in Obstetrics & Gynecology are consistent with reports from other populations taking oral anticancer agents, in which adherence is generally lower than with intravenous therapies.
For their research, Catherine Watson, MD, and colleagues at Duke University, Durham, N.C., recruited 100 women at their institution taking a variety of oral anticancer agents for uterine or ovarian cancer for 30 days or more (median time, 6 months). The women answered a questionnaire that measured adherence as well as health literacy, quality of life, and distress. The researchers also collected information on the subjects’ race, age, insurance type, medication burden, and medication costs.
Fourteen of the women in the study additionally underwent qualitative interviews about their experiences with oral anticancer drugs. The researchers also queried physicians and nurse practitioners about their thoughts on adherence.
Dr. Watson and colleagues reported that 54% of women self-reported perfect adherence to their medication in the previous week, while 21% had missed or skipped one dose, and 25% reported skipping or missing more than one dose.
The researchers saw no significant differences between the adherent and nonadherent groups corresponding with race, age, or other demographic or clinical characteristics, but they noted that their study was not powered to detect such associations. The small sample size and self-reported data were among this study’s limitations, Dr. Watson and colleagues acknowledged.
Interviews with patients revealed some surprising reasons for the less-than-optimal adherence, with 43% of women reporting feeling anxiety about the burden of administering medication at home. While patients acknowledged the convenience of oral regimens, some also expressed a wish for more physician contact and support. Some women who were nonadherent said they perceived the efficacy of oral agents to be less than intravenous therapies.
Physicians and nurse practitioners interviewed by the researchers “tended to assume that their patients were adherent to oral anticancer therapy because of the therapy’s importance, and many did not routinely ask their patients about adherence,” Dr. Watson and colleagues wrote.
Emma Rossi, MD, a gynecologic oncologist at the University of North Carolina at Chapel Hill, commented in an interview that the study highlighted “the importance of not generalizing our perception of patients. As providers we have to spend the time asking patients where they’re at and how they’re thinking about taking an oral medication, with special attention to their fears, their expectations, and their concerns. We should be touching base with them not just before starting drugs, but during the course of treatment.”
Dr. Rossi stressed that compliance in real-life settings is likely to be different from that seen in clinical trials of oral anticancer drugs. “It’s important to recognize that real-world efficacy of treatments may be different from trial efficacy. We see these differences a lot in medicine where studies show a large magnitude of effect that doesn’t play out in real life practice – because of factors like this.”
Some 57% of the women in the study were taking their medications as maintenance, while the rest were taking the medications as active treatment. This too might have an effect on adherence, she said, with the active-treatment group potentially more motivated to maintain perfect adherence.
“You see in the interviews that doctors assume patients would be compliant because of the seriousness of the disease,” Dr. Rossi said. “But some patients said they perceived oral drugs as less strong or effective. If a patient is cancer free on a scan and doesn’t have measurable disease, prescribing an oral medication may be sending the subliminal message that it’s not as important.”
Physicians “may need to take the extra steps to individualize our counseling of patients – especially with therapies that they’re responsible for administering,” Dr. Rossi continued. “As this study shows, every patient sees treatment through her own individual lens. We really need to meet them where they’re at to make them comfortable with their treatment and optimize compliance.”
Dr. Watson and colleagues’ study was supported by their institution. One coauthor reported financial ties with drug manufacturers in the form of grant and clinical trial support and honoraria. Another coauthor is the member of various boards and steering committees, which are uncompensated. Dr. Rossi reported no financial conflicts of interest.
SOURCE: Watson C et al. Obstet Gynecol. 2020. doi: 10.1097/AOG.0000000000004170.
Oral agents are taking on an ever-greater role in the management of gynecologic cancers. However, women being treated for these cancers have less than ideal adherence to such medications, a new study has found – with just over half reporting taking them exactly as prescribed.
The findings reported in Obstetrics & Gynecology are consistent with reports from other populations taking oral anticancer agents, in which adherence is generally lower than with intravenous therapies.
For their research, Catherine Watson, MD, and colleagues at Duke University, Durham, N.C., recruited 100 women at their institution taking a variety of oral anticancer agents for uterine or ovarian cancer for 30 days or more (median time, 6 months). The women answered a questionnaire that measured adherence as well as health literacy, quality of life, and distress. The researchers also collected information on the subjects’ race, age, insurance type, medication burden, and medication costs.
Fourteen of the women in the study additionally underwent qualitative interviews about their experiences with oral anticancer drugs. The researchers also queried physicians and nurse practitioners about their thoughts on adherence.
Dr. Watson and colleagues reported that 54% of women self-reported perfect adherence to their medication in the previous week, while 21% had missed or skipped one dose, and 25% reported skipping or missing more than one dose.
The researchers saw no significant differences between the adherent and nonadherent groups corresponding with race, age, or other demographic or clinical characteristics, but they noted that their study was not powered to detect such associations. The small sample size and self-reported data were among this study’s limitations, Dr. Watson and colleagues acknowledged.
Interviews with patients revealed some surprising reasons for the less-than-optimal adherence, with 43% of women reporting feeling anxiety about the burden of administering medication at home. While patients acknowledged the convenience of oral regimens, some also expressed a wish for more physician contact and support. Some women who were nonadherent said they perceived the efficacy of oral agents to be less than intravenous therapies.
Physicians and nurse practitioners interviewed by the researchers “tended to assume that their patients were adherent to oral anticancer therapy because of the therapy’s importance, and many did not routinely ask their patients about adherence,” Dr. Watson and colleagues wrote.
Emma Rossi, MD, a gynecologic oncologist at the University of North Carolina at Chapel Hill, commented in an interview that the study highlighted “the importance of not generalizing our perception of patients. As providers we have to spend the time asking patients where they’re at and how they’re thinking about taking an oral medication, with special attention to their fears, their expectations, and their concerns. We should be touching base with them not just before starting drugs, but during the course of treatment.”
Dr. Rossi stressed that compliance in real-life settings is likely to be different from that seen in clinical trials of oral anticancer drugs. “It’s important to recognize that real-world efficacy of treatments may be different from trial efficacy. We see these differences a lot in medicine where studies show a large magnitude of effect that doesn’t play out in real life practice – because of factors like this.”
Some 57% of the women in the study were taking their medications as maintenance, while the rest were taking the medications as active treatment. This too might have an effect on adherence, she said, with the active-treatment group potentially more motivated to maintain perfect adherence.
“You see in the interviews that doctors assume patients would be compliant because of the seriousness of the disease,” Dr. Rossi said. “But some patients said they perceived oral drugs as less strong or effective. If a patient is cancer free on a scan and doesn’t have measurable disease, prescribing an oral medication may be sending the subliminal message that it’s not as important.”
Physicians “may need to take the extra steps to individualize our counseling of patients – especially with therapies that they’re responsible for administering,” Dr. Rossi continued. “As this study shows, every patient sees treatment through her own individual lens. We really need to meet them where they’re at to make them comfortable with their treatment and optimize compliance.”
Dr. Watson and colleagues’ study was supported by their institution. One coauthor reported financial ties with drug manufacturers in the form of grant and clinical trial support and honoraria. Another coauthor is the member of various boards and steering committees, which are uncompensated. Dr. Rossi reported no financial conflicts of interest.
SOURCE: Watson C et al. Obstet Gynecol. 2020. doi: 10.1097/AOG.0000000000004170.
FROM OBSTETRICS & GYNECOLOGY













