User login
Coronavirus on the inpatient unit: A new challenge for psychiatry
For weeks now, the coronavirus epidemic has monopolized the media. As cases of COVID-19 have crossed borders and then oceans, the pandemic has caused fear and anxiety as Americans struggle with the uncertain nature of this highly contagious disease. Those exposed may be infectious before they are symptomatic, and the expression of the virus varies greatly: Some people have a mild illness and others quickly progress to severe pulmonary disease with a bilateral interstitial pneumonia that requires intubation and respiratory support. So far, the number of people infected and the absolute mortality is a fraction of what we have seen with this year’s seasonal flu, but in countries where the virus has spread quickly, medical systems have not been able to keep up with demand for high-intensity care and mortality rates have been many times higher than that of the flu. Italy, in particular, has not been able to halt the spread, even with the entire country on lockdown, and the medical system has been overwhelmed, resulting in rationing of care and many deaths.
COVID-19 represents a new challenge for the inpatient psychiatry unit. Some patients on an acute psychiatric unit may be agitated, uncooperative, or even violent, and it’s not hard to imagine the distress of anyone who has a patient spit on them as we’re all trying to remember not to shake hands. Inevitably, there will be patients who present for psychiatric admission with no respiratory symptoms, who are admitted and then become ill and are diagnosed with COVID-19. In the meantime, the potential is there for contagion to other patients on the unit, the hospital staff, and visitors to the unit.
While many hospital units treat infectious patients, the issues with psychiatry are different; psychiatry units are not set up to have aggressive infection control, staff and patients don’t typically wear protective gear, and people with psychiatric illnesses are ambulatory and interactive. The treatment of psychiatric illnesses involves more – not less – social interaction and patients attend groups and occupational therapy sessions; they dine in communal areas and watch television together in day rooms. Cell phones are typically not permitted for issues of privacy, and patients may use communal telephones. Patients who are very ill with psychiatric disorders may resist hygiene measures, and they may intrude on the personal space of others.
Patients with known COVID-19 can be isolated or transferred to another unit if more intensive medical care is necessary, but by that time, others have been exposed and potentially infected. How to contain this potential risk has been a topic of concern for psychiatric units everywhere. Following a potential or confirmed exposure, it’s not completely clear who should be sent home for self-quarantine: Do the staff who have had contact with the patient leave for 2 weeks, and if so, is there enough staff to replace them? Do they continue to work with protective equipment and leave only if they become symptomatic and test positive? Do staff remain at the hospital, or do they go home at night, potentially infecting those on public transportation and their family members? Presumably new patients would not be admitted to the unit, but our psychiatric system is taxed already with a lack of available beds.
In South Korea, patients and staff at the Daenam hospital reportedly faced this exact scenario. The hospital was locked down and 101 patients in the psychiatric facility developed COVID-19; 7 of those patients died, an outcome we hope to never see again. As of this writing, there are two patients on a 22-bed geropsychiatric unit at the UW Medical Center – Northwest in Seattle who developed COVID-19 after they were admitted to the unit. They have been isolated, and the rest of the patients on the unit have been quarantined. The staff are now wearing masks, gowns, and gloves.
“We started precautions for all 22 patients. ... We instituted our protocols for every room around, donning and doffing personal protective equipment (PPE). We had conversations with their family members,” said Santiago Neme, MD, MPH, an infectious disease physician at UW Medical Center – Northwest, in a press conference released by the university. “The patients were transferred and both remain stable. All patients on the unit were tested even though there were no concerning symptoms.”
These measures are necessary for infection control, but they are not helpful for the treatment of psychiatric disorders. Treatment consists, in part, of getting patients out of their rooms and involved in therapeutic activities in a milieu that removes them from the usual stresses of their daily lives. How insurance companies will respond to any need for extra days is one more concern to throw into the mix.
Paul Summergrad, MD, chairman of psychiatry at Tufts University in Boston, has been very interested in what facilities around the world have been doing. “In Washington state, after the nursing home infections, they sent home over a hundred staff members who had been exposed. In Hong Kong, the psychiatric hospitals have limited how patients circulate on the units even if no one is infected; this is not something that would go over well in the U.S.,” he said in an interview. Dr. Summergrad believes that higher death rates are seen in countries with higher smoking rates, and patients with psychiatric disorders are more likely to smoke than the general public, possibly placing them at higher risk for more severe morbidity and mortality.
Patrick Triplett, MD, clinical director of the department of psychiatry at Johns Hopkins University in Baltimore, communicated with me about their plans to manage a scenario in which a patient becomes ill on a psychiatry unit.
“If we think a patient might be infected, we will isolate them in a room with a closed door (We would need to account for their psychiatric needs as well during this period, say constant observation, for example.) and call the centralized command center, where the Hospital Epidemiology and Infection Control (HEIC) team gets involved. They will determine if the patient should be tested for COVID-19. If it’s determined that the patient is infected, they will likely be transferred to a floor with negative pressure rooms. We would coordinate psychiatric nursing needs with the receiving unit, based on the patient’s clinical needs.”
Dr. Triplett elaborated on the exposure of staff and visitors. “We take our lead on postexposure management from [HEIC] and Occupational Health. There are procedures in place for environmental cleaning, waste, linens, etc. The [Centers for Disease Control and Prevention] has guidelines on work restrictions for staff who have cared for patients with confirmed COVID-19, and HEIC helps determine the provider’s risk category. We would also involve them in determining risk levels and management for other patients on the floor and visitors. But prior to any known exposure, we are already limiting visitors for patients per the governor’s mandate of only one adult visitor per patient.”
The next couple of weeks will be telling, and as we readjust to a life of social distancing, it is certain to be a challenge to keep ourselves and our patients safe, healthy, and emotionally strong.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
For weeks now, the coronavirus epidemic has monopolized the media. As cases of COVID-19 have crossed borders and then oceans, the pandemic has caused fear and anxiety as Americans struggle with the uncertain nature of this highly contagious disease. Those exposed may be infectious before they are symptomatic, and the expression of the virus varies greatly: Some people have a mild illness and others quickly progress to severe pulmonary disease with a bilateral interstitial pneumonia that requires intubation and respiratory support. So far, the number of people infected and the absolute mortality is a fraction of what we have seen with this year’s seasonal flu, but in countries where the virus has spread quickly, medical systems have not been able to keep up with demand for high-intensity care and mortality rates have been many times higher than that of the flu. Italy, in particular, has not been able to halt the spread, even with the entire country on lockdown, and the medical system has been overwhelmed, resulting in rationing of care and many deaths.
COVID-19 represents a new challenge for the inpatient psychiatry unit. Some patients on an acute psychiatric unit may be agitated, uncooperative, or even violent, and it’s not hard to imagine the distress of anyone who has a patient spit on them as we’re all trying to remember not to shake hands. Inevitably, there will be patients who present for psychiatric admission with no respiratory symptoms, who are admitted and then become ill and are diagnosed with COVID-19. In the meantime, the potential is there for contagion to other patients on the unit, the hospital staff, and visitors to the unit.
While many hospital units treat infectious patients, the issues with psychiatry are different; psychiatry units are not set up to have aggressive infection control, staff and patients don’t typically wear protective gear, and people with psychiatric illnesses are ambulatory and interactive. The treatment of psychiatric illnesses involves more – not less – social interaction and patients attend groups and occupational therapy sessions; they dine in communal areas and watch television together in day rooms. Cell phones are typically not permitted for issues of privacy, and patients may use communal telephones. Patients who are very ill with psychiatric disorders may resist hygiene measures, and they may intrude on the personal space of others.
Patients with known COVID-19 can be isolated or transferred to another unit if more intensive medical care is necessary, but by that time, others have been exposed and potentially infected. How to contain this potential risk has been a topic of concern for psychiatric units everywhere. Following a potential or confirmed exposure, it’s not completely clear who should be sent home for self-quarantine: Do the staff who have had contact with the patient leave for 2 weeks, and if so, is there enough staff to replace them? Do they continue to work with protective equipment and leave only if they become symptomatic and test positive? Do staff remain at the hospital, or do they go home at night, potentially infecting those on public transportation and their family members? Presumably new patients would not be admitted to the unit, but our psychiatric system is taxed already with a lack of available beds.
In South Korea, patients and staff at the Daenam hospital reportedly faced this exact scenario. The hospital was locked down and 101 patients in the psychiatric facility developed COVID-19; 7 of those patients died, an outcome we hope to never see again. As of this writing, there are two patients on a 22-bed geropsychiatric unit at the UW Medical Center – Northwest in Seattle who developed COVID-19 after they were admitted to the unit. They have been isolated, and the rest of the patients on the unit have been quarantined. The staff are now wearing masks, gowns, and gloves.
“We started precautions for all 22 patients. ... We instituted our protocols for every room around, donning and doffing personal protective equipment (PPE). We had conversations with their family members,” said Santiago Neme, MD, MPH, an infectious disease physician at UW Medical Center – Northwest, in a press conference released by the university. “The patients were transferred and both remain stable. All patients on the unit were tested even though there were no concerning symptoms.”
These measures are necessary for infection control, but they are not helpful for the treatment of psychiatric disorders. Treatment consists, in part, of getting patients out of their rooms and involved in therapeutic activities in a milieu that removes them from the usual stresses of their daily lives. How insurance companies will respond to any need for extra days is one more concern to throw into the mix.
Paul Summergrad, MD, chairman of psychiatry at Tufts University in Boston, has been very interested in what facilities around the world have been doing. “In Washington state, after the nursing home infections, they sent home over a hundred staff members who had been exposed. In Hong Kong, the psychiatric hospitals have limited how patients circulate on the units even if no one is infected; this is not something that would go over well in the U.S.,” he said in an interview. Dr. Summergrad believes that higher death rates are seen in countries with higher smoking rates, and patients with psychiatric disorders are more likely to smoke than the general public, possibly placing them at higher risk for more severe morbidity and mortality.
Patrick Triplett, MD, clinical director of the department of psychiatry at Johns Hopkins University in Baltimore, communicated with me about their plans to manage a scenario in which a patient becomes ill on a psychiatry unit.
“If we think a patient might be infected, we will isolate them in a room with a closed door (We would need to account for their psychiatric needs as well during this period, say constant observation, for example.) and call the centralized command center, where the Hospital Epidemiology and Infection Control (HEIC) team gets involved. They will determine if the patient should be tested for COVID-19. If it’s determined that the patient is infected, they will likely be transferred to a floor with negative pressure rooms. We would coordinate psychiatric nursing needs with the receiving unit, based on the patient’s clinical needs.”
Dr. Triplett elaborated on the exposure of staff and visitors. “We take our lead on postexposure management from [HEIC] and Occupational Health. There are procedures in place for environmental cleaning, waste, linens, etc. The [Centers for Disease Control and Prevention] has guidelines on work restrictions for staff who have cared for patients with confirmed COVID-19, and HEIC helps determine the provider’s risk category. We would also involve them in determining risk levels and management for other patients on the floor and visitors. But prior to any known exposure, we are already limiting visitors for patients per the governor’s mandate of only one adult visitor per patient.”
The next couple of weeks will be telling, and as we readjust to a life of social distancing, it is certain to be a challenge to keep ourselves and our patients safe, healthy, and emotionally strong.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
For weeks now, the coronavirus epidemic has monopolized the media. As cases of COVID-19 have crossed borders and then oceans, the pandemic has caused fear and anxiety as Americans struggle with the uncertain nature of this highly contagious disease. Those exposed may be infectious before they are symptomatic, and the expression of the virus varies greatly: Some people have a mild illness and others quickly progress to severe pulmonary disease with a bilateral interstitial pneumonia that requires intubation and respiratory support. So far, the number of people infected and the absolute mortality is a fraction of what we have seen with this year’s seasonal flu, but in countries where the virus has spread quickly, medical systems have not been able to keep up with demand for high-intensity care and mortality rates have been many times higher than that of the flu. Italy, in particular, has not been able to halt the spread, even with the entire country on lockdown, and the medical system has been overwhelmed, resulting in rationing of care and many deaths.
COVID-19 represents a new challenge for the inpatient psychiatry unit. Some patients on an acute psychiatric unit may be agitated, uncooperative, or even violent, and it’s not hard to imagine the distress of anyone who has a patient spit on them as we’re all trying to remember not to shake hands. Inevitably, there will be patients who present for psychiatric admission with no respiratory symptoms, who are admitted and then become ill and are diagnosed with COVID-19. In the meantime, the potential is there for contagion to other patients on the unit, the hospital staff, and visitors to the unit.
While many hospital units treat infectious patients, the issues with psychiatry are different; psychiatry units are not set up to have aggressive infection control, staff and patients don’t typically wear protective gear, and people with psychiatric illnesses are ambulatory and interactive. The treatment of psychiatric illnesses involves more – not less – social interaction and patients attend groups and occupational therapy sessions; they dine in communal areas and watch television together in day rooms. Cell phones are typically not permitted for issues of privacy, and patients may use communal telephones. Patients who are very ill with psychiatric disorders may resist hygiene measures, and they may intrude on the personal space of others.
Patients with known COVID-19 can be isolated or transferred to another unit if more intensive medical care is necessary, but by that time, others have been exposed and potentially infected. How to contain this potential risk has been a topic of concern for psychiatric units everywhere. Following a potential or confirmed exposure, it’s not completely clear who should be sent home for self-quarantine: Do the staff who have had contact with the patient leave for 2 weeks, and if so, is there enough staff to replace them? Do they continue to work with protective equipment and leave only if they become symptomatic and test positive? Do staff remain at the hospital, or do they go home at night, potentially infecting those on public transportation and their family members? Presumably new patients would not be admitted to the unit, but our psychiatric system is taxed already with a lack of available beds.
In South Korea, patients and staff at the Daenam hospital reportedly faced this exact scenario. The hospital was locked down and 101 patients in the psychiatric facility developed COVID-19; 7 of those patients died, an outcome we hope to never see again. As of this writing, there are two patients on a 22-bed geropsychiatric unit at the UW Medical Center – Northwest in Seattle who developed COVID-19 after they were admitted to the unit. They have been isolated, and the rest of the patients on the unit have been quarantined. The staff are now wearing masks, gowns, and gloves.
“We started precautions for all 22 patients. ... We instituted our protocols for every room around, donning and doffing personal protective equipment (PPE). We had conversations with their family members,” said Santiago Neme, MD, MPH, an infectious disease physician at UW Medical Center – Northwest, in a press conference released by the university. “The patients were transferred and both remain stable. All patients on the unit were tested even though there were no concerning symptoms.”
These measures are necessary for infection control, but they are not helpful for the treatment of psychiatric disorders. Treatment consists, in part, of getting patients out of their rooms and involved in therapeutic activities in a milieu that removes them from the usual stresses of their daily lives. How insurance companies will respond to any need for extra days is one more concern to throw into the mix.
Paul Summergrad, MD, chairman of psychiatry at Tufts University in Boston, has been very interested in what facilities around the world have been doing. “In Washington state, after the nursing home infections, they sent home over a hundred staff members who had been exposed. In Hong Kong, the psychiatric hospitals have limited how patients circulate on the units even if no one is infected; this is not something that would go over well in the U.S.,” he said in an interview. Dr. Summergrad believes that higher death rates are seen in countries with higher smoking rates, and patients with psychiatric disorders are more likely to smoke than the general public, possibly placing them at higher risk for more severe morbidity and mortality.
Patrick Triplett, MD, clinical director of the department of psychiatry at Johns Hopkins University in Baltimore, communicated with me about their plans to manage a scenario in which a patient becomes ill on a psychiatry unit.
“If we think a patient might be infected, we will isolate them in a room with a closed door (We would need to account for their psychiatric needs as well during this period, say constant observation, for example.) and call the centralized command center, where the Hospital Epidemiology and Infection Control (HEIC) team gets involved. They will determine if the patient should be tested for COVID-19. If it’s determined that the patient is infected, they will likely be transferred to a floor with negative pressure rooms. We would coordinate psychiatric nursing needs with the receiving unit, based on the patient’s clinical needs.”
Dr. Triplett elaborated on the exposure of staff and visitors. “We take our lead on postexposure management from [HEIC] and Occupational Health. There are procedures in place for environmental cleaning, waste, linens, etc. The [Centers for Disease Control and Prevention] has guidelines on work restrictions for staff who have cared for patients with confirmed COVID-19, and HEIC helps determine the provider’s risk category. We would also involve them in determining risk levels and management for other patients on the floor and visitors. But prior to any known exposure, we are already limiting visitors for patients per the governor’s mandate of only one adult visitor per patient.”
The next couple of weeks will be telling, and as we readjust to a life of social distancing, it is certain to be a challenge to keep ourselves and our patients safe, healthy, and emotionally strong.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
Second-generation long-acting injectable antipsychotics: A practical guide
There are currently 7 FDA-approved second-generation long-acting injectable antipsychotics (LAIAs).1-7 These LAIAs provide a unique dosage form that allows patients to receive an antipsychotic without taking oral medications every day, or multiple times per day. This may be an appealing option for patients and clinicians, but because there are several types of LAIAs available, it may be difficult to determine which LAIA characteristics are best for a given patient.
Since the FDA approved the first second-generation LAIA, risperidone long-acting injectable (LAI),1 in 2003, 6 additional second-generation LAIAs have been approved:
- aripiprazole LAI
- aripiprazole lauroxil LAI
- olanzapine pamoate LAI
- paliperidone palmitate monthly injection
- paliperidone palmitate 3-month LAI
- risperidone LAI for subcutaneous (SQ) injection.
When discussing medication options with patients, clinicians need to consider factors that are unique to each LAIA. In this article, I describe the similarities and differences among the second-generation LAIAs, and address common questions about these medications.
A major potential benefit: Increased adherence
One potential benefit of all LAIAs is increased medication adherence compared with oral antipsychotics. One meta-analysis of 21 randomized controlled trials (RCTs) that compared LAIAs with oral antipsychotics and included 5,176 patients found that LAIAs had a similar efficacy to oral antipsychotics in preventing relapse.8 However, a meta-analysis of 25 mirror-image studies comparing LAIAs with oral antipsychotics that included 5,940 patients found that LAIAs were superior in preventing hospitalization.9 In these mirror-image studies, participants received oral antipsychotics first and then switched to LAIAs, and the 2 study periods were compared. Because mirror-image studies are observational, participants do not engage with research teams to the extent that they do in RCTs.9 Although mirror-image studies have limitations, participants in these studies may be a better representation of patients encountered in clinical practice due to the extensive monitoring and follow-up RCT participants typically receive.9
Differences in FDA-approved indications
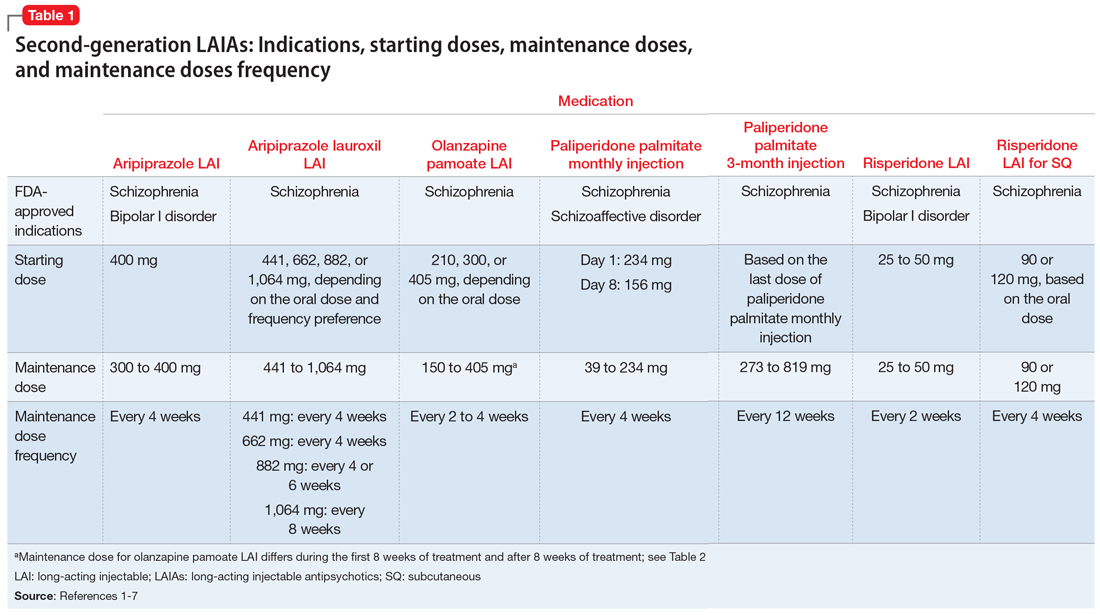
The 7 currently available LAIAs vary in terms of FDA-approved indications, dose options, frequency, need for oral antipsychotic overlap, route of administration, and other factors. Table 11-7 summarizes some of these differences. Although all second-generation LAIAs are approved for schizophrenia,1-7 risperidone LAI and aripiprazole LAI are also approved for bipolar I disorder.1,4 Paliperidone palmitate monthly injection is the only LAIA approved for treating patients with schizoaffective disorder.2
Starting doses
For most LAIAs, the starting dose is the same as the maintenance dose (Table 11-7). One exception is paliperidone palmitate monthly injection, which requires a 234-mg dose on Day 1 followed by a 156-mg dose on Day 8 for all patients, regardless of the maintenance dose required.2 The 156-mg dose may be given 4 days before or after Day 8.2 The first maintenance dose of paliperidone palmitate monthly injection should be administered 5 weeks after the 234-mg dose on Day 1.2 Before starting paliperidone palmitate 3-month injection, patients should be stable on paliperidone palmitate monthly injection for 4 months, and the 2 most recent doses of paliperidone palmitate monthly injection should be the same.3
Maintenance doses
Dosing frequency may be an important factor for some patients when deciding to receive a LAIA. The frequency of the maintenance doses for all second-generation LAIAs varies from every 2 weeks to 12 weeks (Table 11-7). Paliperidone palmitate 3-month LAI is the only LAIA that is administered every 12 weeks.3 Some dosages of aripiprazole lauroxil LAI are administered every 6 or 8 weeks.6 All other second-generation LAIAs are given every 2 to 4 weeks.
Continue to: Start with an oral antipsychotic
Start with an oral antipsychotic
Before starting any LAIA, patients should receive the oral formulation of that antipsychotic to establish tolerability.1-7 Four of the 7 available LAIAs have an oral-to-LAI dose equivalency recommendation in their prescribing information (Table 22,5-7). This can help clinicians estimate the LAIA maintenance dose required to control a patient’s symptoms. If a dose adjustment is needed once a patient starts an LAIA, the dose adjustment can be made when the next injection is due.2
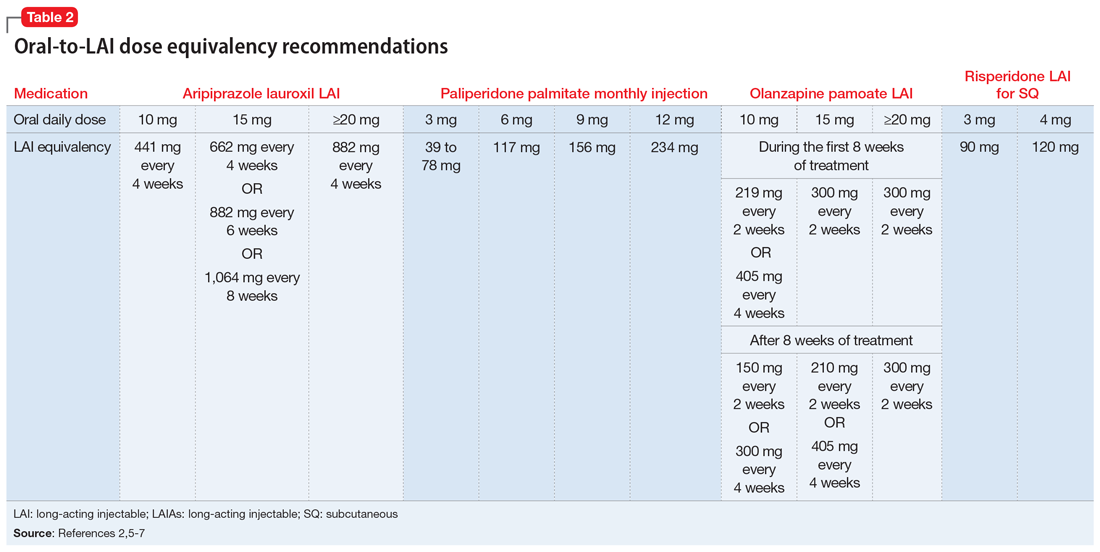
There are 2 important considerations when prescribing olanzapine pamoate LAI. First, the recommended dose for olanzapine pamoate LAI based on oral olanzapine doses differs during the first 8 weeks of treatment compared with after 8 weeks of treatment (Table 22,5-7). Additionally, because there are both short-acting and long-acting injections of olanzapine, it is essential to choose the correct formulation when prescribing this medication.5
Overlap with an oral antipsychotic might be necessary
Administration of several of the LAIAs may require overlap with an oral antipsychotic (Table 31,2,4-7). Patients who refuse to take oral medications may benefit from one of the LAIAs that does not require oral overlap—paliperidone palmitate monthly injection, olanzapine pamoate LAI, and risperidone LAI for SQ.2,5,7 Risperidone LAI requires overlap with oral risperidone for 3 weeks.1

Aripiprazole is available in 2 LAI formulations: aripiprazole LAI and aripiprazole lauroxil LAI. Aripiprazole lauroxil is a prodrug of aripiprazole, and these 2 LAI medications differ in available dose options and dosing frequency.4,6 Aripiprazole LAI requires an oral overlap for 2 weeks after the first injection, whereas aripiprazole lauroxil LAI requires 3 weeks of oral overlap unless aripiprazole lauroxil 675-mg LAI is administered (Figure6).4,6,10
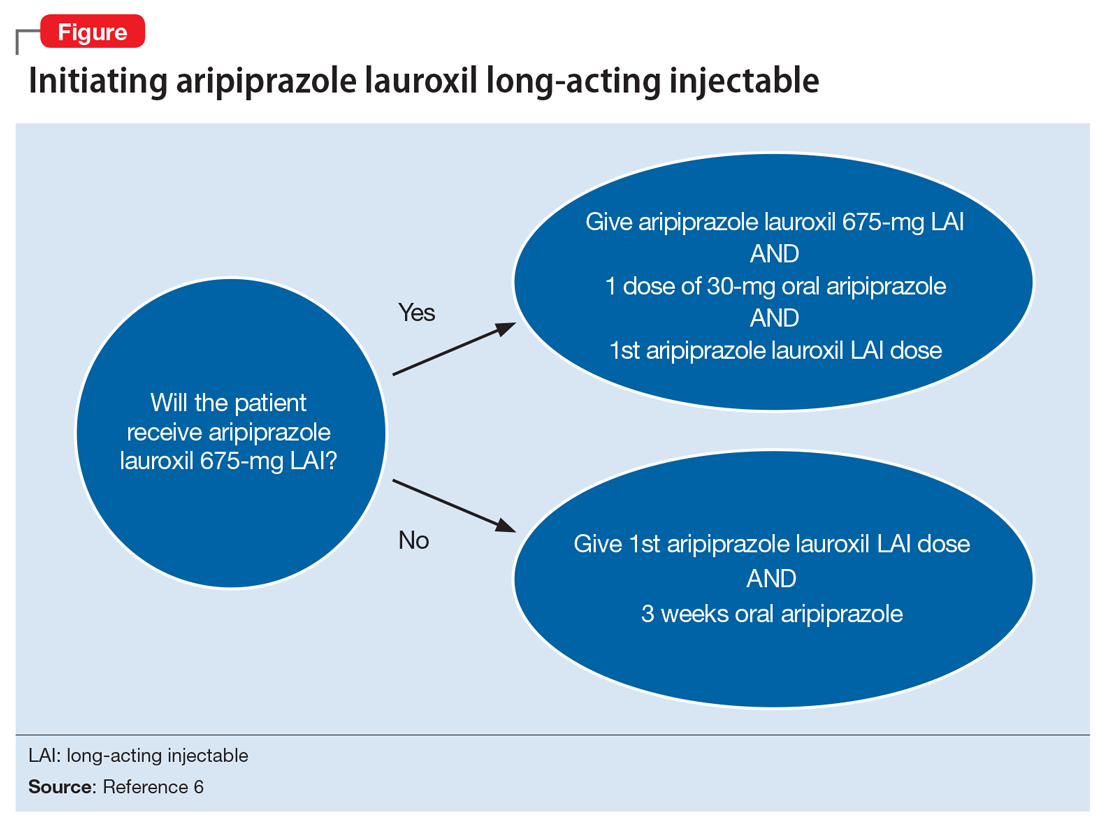
Aripiprazole lauroxil 675-mg LAI is formulated with drug particles that are smaller than those in aripiprazole lauroxil LAI.11 The smaller particle size results in faster dissolution and a more rapid increase in plasma aripiprazole levels. Aripiprazole lauroxil 675-mg LAI is a single injection that should be given with one 30-mg dose of oral aripiprazole.10 This combination results in aripiprazole concentrations that are comparable to aripiprazole lauroxil LAI and oral aripiprazole overlap for 3 weeks after the first injection.10
Continue to: The starting dose of aripiprazole lauroxil LAI...
The starting dose of aripiprazole lauroxil LAI may be administered on the same day as aripiprazole lauroxil 675-mg LAI and the 30-mg oral aripiprazole dose, or it may be administered up to 10 days after.10 Aripiprazole lauroxil LAI and aripiprazole lauroxil 675-mg LAI are not interchangeable due to differing pharmacokinetic profiles.6,10 Aripiprazole lauroxil 675-mg LAI may be used to re-initiate treatment in a patient who missed doses of aripiprazole lauroxil LAI.10 Aripiprazole lauroxil LAI and aripiprazole lauroxil 675 mg should not be injected together into the same deltoid or gluteal muscle.
Be mindful of differences in dosing windows
Each LAIA has a specific frequency recommendation, but due to scheduling or other factors, it may not be possible for patients to receive their injection on the specified day. The prescribing information for some LAIAs provides a dosing window (Table 41-7). The prescribing information for risperidone LAI, olanzapine pamoate LAI, and risperidone LAI for SQ does not specify how many days the injection can be administered before or after the due date; however, the prescribing information for risperidone LAI for SQ indicates that if the injection is not given on the due date, it should be administered as soon as possible after that.1,5,7
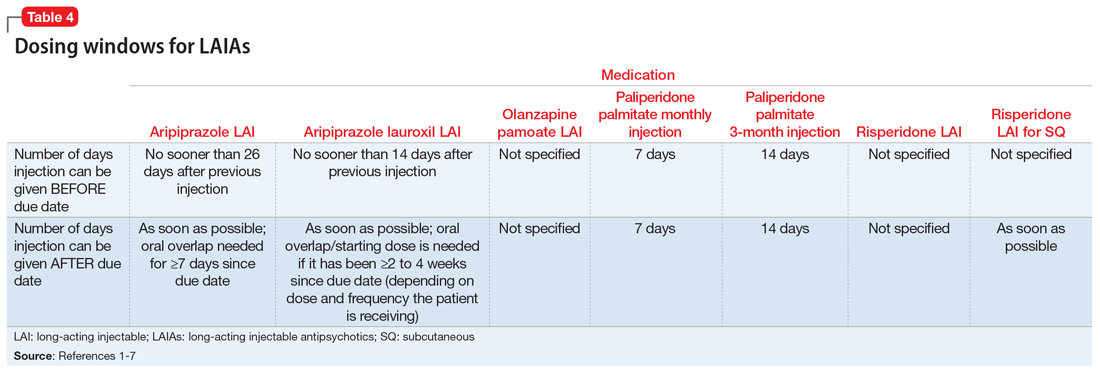
Paliperidone palmitate monthly injection and paliperidone palmitate 3-month LAI have the clearest recommendations for a dosing window. Paliperidone palmitate monthly injection may be administered 7 days before or after the 4-week due date, and paliperidone palmitate 3-month LAI can be administered 14 days before or after the 12-week due date.2,3
Aripiprazole LAI should not be administered sooner than 26 days after the previous injection, which means that it can be administered up to 2 days before the 4-week due date.4 If administered after the due date, it should be given as soon as possible, although oral overlap is not needed until ≥7 days past the due date.4
Aripiprazole lauroxil LAI has similar recommendations to aripiprazole LAI in that it should not be administered sooner than 14 days after the previous injection.6 If it is given after the due date, it should be administered as soon as possible; oral overlap/starting dose is needed if it has been ≥2 to 4 weeks since the due date, depending on which dose and frequency the patient is receiving.6
Continue to: Recommendations for missed doses
Recommendations for missed doses
Each LAIA has specific recommendations for missed dosing. Carpenter and Wong12 reviewed the recommendations for managing missed LAIA doses in
Consider patient preference
Patient preference for the type and location of the injection may factor into a clinician’s choice of LAIA (Table 51-7,10). Risperidone LAI for SQ is the only LAIA that is administered as an SQ abdominal injection.7 All other LAIAs are IM injections in the deltoid or gluteal muscle.1-6 All doses of risperidone LAI, paliperidone palmitate 3-month LAI, aripiprazole LAI, and aripiprazole lauroxil 675-mg LAI can be administered in the deltoid or gluteal muscle.1,3,4,10 Deltoid administration is required for the 2 starting doses of paliperidone palmitate monthly injection, but maintenance doses can be administered in the deltoid or gluteal muscle. Because administration into the deltoid results in a higher concentration of the drug compared with gluteal administration, administering the 2 starting doses of paliperidone palmitate monthly injection into the deltoid helps to rapidly attain therapeutic concentrations.2 Olanzapine pamoate LAI should be administered only in the gluteal muscle.5 The 441-mg dose of aripiprazole lauroxil LAI may be administered in the deltoid or gluteal muscle, but all other doses of aripiprazole lauroxil LAI should be administered only in the gluteal muscle.6
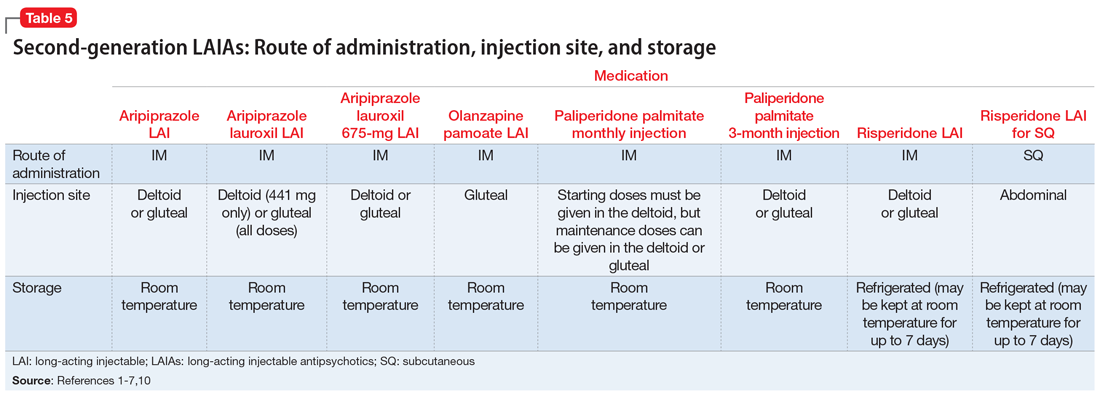
Storage
Most LAIAs can be stored at room temperature2-6; however, risperidone LAI and risperidone LAI for SQ need to be stored in the refrigerator. Both risperidone LAI and risperidone LAI for SQ may be kept at room temperature for up to 7 days. If they are not used within 7 days at room temperature, they should be discarded.1,7
Clinical pearls for specific LAIAs
Aripiprazole LAI. The recommended starting and maintenance dose for aripiprazole LAI is 400 mg monthly, unless the patient has drug interactions or other factors that require dose adjustment. If patients experience adverse reactions to the 400-mg dose, a reduction to 300 mg monthly could be considered.4
Olanzapine pamoate LAI has a Risk Evaluation and Mitigation Strategy (REMS) due to the potential for post-injection delirium/sedation syndrome (PDSS). Prescribing clinicians, dispensing pharmacies, and administering health care facilities must all be certified to prescribe, dispense, or administer olanzapine pamoate LAI. The patient must also be enrolled in the REMS program.13 Patients must be observed by health care staff for 3 hours after receiving a dose of olanzapine pamoate LAI to monitor for signs and symptoms of PDSS.5
Continue to: Risperidone LAI
Risperidone LAI. When increasing the dose of risperidone LAI, do not expect to see the clinical effects of the new dose earlier than 3 weeks after initiating the higher dose, because the main release of the medication starts at 3 weeks after the injection.1
Risperidone LAI for SQ has specific recommendations for the LAI dose based on whether the patient was stable when receiving 3 or 4 mg/d of oral risperidone. If patients are stable on <3 or >4 mg/d, they may not be candidates for risperidone LAI for SQ.7
Table 61-7,10 lists additional factors to consider when prescribing a specific LAIA.
Bottom Line
Second-generation long-acting injectable antipsychotics (LAIAs) have the potential to increase medication adherence. There are important differences among the 7 currently available LAIAs. For effective prescribing, clinicians need to understand each medication’s unique aspects, including dosing options, frequency, need for oral antipsychotic overlap, and route of administration.
Related Resources
- Correll CU, Citrome L, Haddad PM, et al. The use of long-acting injectable antipsychotics in schizophrenia: evaluating the evidence. J Clin Psychiatry. 2016;77(suppl 3):1-24.
- Peters L, Krogmann A, von Hardenberg L, et al. Long-acting injections in schizophrenia: a 3-year update on randomized controlled trials published January 2016-March 2019. Curr Psychiatry Rep. 2019;21(12):124.
Drug Brand Names
Aripiprazole • Abilify
Aripiprazole long-acting injectable • Abilify Maintena
Aripiprazole lauroxil extended-release injectable suspension • Aristada
Aripiprazole lauroxil 675 mg • Aristada Initio
Olanzapine pamoate long-acting injection • Zyprexa Relprevv
Paliperidone palmitate monthly long-acting injection • Invega Sustenna
Paliperidone palmitate 3-month injection • Invega Trinza
Risperidone • Risperdal
Risperidone long-acting injection • Risperdal Consta
Risperidone long-acting injection for SQ • Perseris
1. Risperdal Consta [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
2. Invega Sustenna [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
3. Invega Trinza [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
4. Abilify Maintena [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; 2019.
5. Zyprexa Relprevv [package insert]. Indianapolis; IN: Eli Lilly and Co.; 2019.
6. Aristada [package insert]. Waltham, MA: Alkermes, Inc.; 2019.
7. Perseris [package insert]. North Chesterfield, VA: Indivior, Inc.; 2018.
8. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213.
9. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965.
10. Aristada Initio [package insert]. Waltham, MA: Alkermes, Inc.; 2019.
11. Jain R, Meyer J, Wehr A, et al. Size matters: the importance of particle size in a newly developed injectable formulation for the treatment of schizophrenia. CNS Spectr. 2019:1-8.
12. Carpenter J, Wong KK. Long-acting injectable antipsychotics: what to do about missed doses. Current Psychiatry. 2018;17(7):10-12,14-19,56.
13. US Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS) zyprexa relprevv (olanzapine). https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=74. Updated April 11, 2019. Accessed January 27, 2020.
There are currently 7 FDA-approved second-generation long-acting injectable antipsychotics (LAIAs).1-7 These LAIAs provide a unique dosage form that allows patients to receive an antipsychotic without taking oral medications every day, or multiple times per day. This may be an appealing option for patients and clinicians, but because there are several types of LAIAs available, it may be difficult to determine which LAIA characteristics are best for a given patient.
Since the FDA approved the first second-generation LAIA, risperidone long-acting injectable (LAI),1 in 2003, 6 additional second-generation LAIAs have been approved:
- aripiprazole LAI
- aripiprazole lauroxil LAI
- olanzapine pamoate LAI
- paliperidone palmitate monthly injection
- paliperidone palmitate 3-month LAI
- risperidone LAI for subcutaneous (SQ) injection.
When discussing medication options with patients, clinicians need to consider factors that are unique to each LAIA. In this article, I describe the similarities and differences among the second-generation LAIAs, and address common questions about these medications.
A major potential benefit: Increased adherence
One potential benefit of all LAIAs is increased medication adherence compared with oral antipsychotics. One meta-analysis of 21 randomized controlled trials (RCTs) that compared LAIAs with oral antipsychotics and included 5,176 patients found that LAIAs had a similar efficacy to oral antipsychotics in preventing relapse.8 However, a meta-analysis of 25 mirror-image studies comparing LAIAs with oral antipsychotics that included 5,940 patients found that LAIAs were superior in preventing hospitalization.9 In these mirror-image studies, participants received oral antipsychotics first and then switched to LAIAs, and the 2 study periods were compared. Because mirror-image studies are observational, participants do not engage with research teams to the extent that they do in RCTs.9 Although mirror-image studies have limitations, participants in these studies may be a better representation of patients encountered in clinical practice due to the extensive monitoring and follow-up RCT participants typically receive.9
Differences in FDA-approved indications
The 7 currently available LAIAs vary in terms of FDA-approved indications, dose options, frequency, need for oral antipsychotic overlap, route of administration, and other factors. Table 11-7 summarizes some of these differences. Although all second-generation LAIAs are approved for schizophrenia,1-7 risperidone LAI and aripiprazole LAI are also approved for bipolar I disorder.1,4 Paliperidone palmitate monthly injection is the only LAIA approved for treating patients with schizoaffective disorder.2
Starting doses
For most LAIAs, the starting dose is the same as the maintenance dose (Table 11-7). One exception is paliperidone palmitate monthly injection, which requires a 234-mg dose on Day 1 followed by a 156-mg dose on Day 8 for all patients, regardless of the maintenance dose required.2 The 156-mg dose may be given 4 days before or after Day 8.2 The first maintenance dose of paliperidone palmitate monthly injection should be administered 5 weeks after the 234-mg dose on Day 1.2 Before starting paliperidone palmitate 3-month injection, patients should be stable on paliperidone palmitate monthly injection for 4 months, and the 2 most recent doses of paliperidone palmitate monthly injection should be the same.3

Maintenance doses
Dosing frequency may be an important factor for some patients when deciding to receive a LAIA. The frequency of the maintenance doses for all second-generation LAIAs varies from every 2 weeks to 12 weeks (Table 11-7). Paliperidone palmitate 3-month LAI is the only LAIA that is administered every 12 weeks.3 Some dosages of aripiprazole lauroxil LAI are administered every 6 or 8 weeks.6 All other second-generation LAIAs are given every 2 to 4 weeks.
Continue to: Start with an oral antipsychotic
Start with an oral antipsychotic
Before starting any LAIA, patients should receive the oral formulation of that antipsychotic to establish tolerability.1-7 Four of the 7 available LAIAs have an oral-to-LAI dose equivalency recommendation in their prescribing information (Table 22,5-7). This can help clinicians estimate the LAIA maintenance dose required to control a patient’s symptoms. If a dose adjustment is needed once a patient starts an LAIA, the dose adjustment can be made when the next injection is due.2

There are 2 important considerations when prescribing olanzapine pamoate LAI. First, the recommended dose for olanzapine pamoate LAI based on oral olanzapine doses differs during the first 8 weeks of treatment compared with after 8 weeks of treatment (Table 22,5-7). Additionally, because there are both short-acting and long-acting injections of olanzapine, it is essential to choose the correct formulation when prescribing this medication.5
Overlap with an oral antipsychotic might be necessary
Administration of several of the LAIAs may require overlap with an oral antipsychotic (Table 31,2,4-7). Patients who refuse to take oral medications may benefit from one of the LAIAs that does not require oral overlap—paliperidone palmitate monthly injection, olanzapine pamoate LAI, and risperidone LAI for SQ.2,5,7 Risperidone LAI requires overlap with oral risperidone for 3 weeks.1

Aripiprazole is available in 2 LAI formulations: aripiprazole LAI and aripiprazole lauroxil LAI. Aripiprazole lauroxil is a prodrug of aripiprazole, and these 2 LAI medications differ in available dose options and dosing frequency.4,6 Aripiprazole LAI requires an oral overlap for 2 weeks after the first injection, whereas aripiprazole lauroxil LAI requires 3 weeks of oral overlap unless aripiprazole lauroxil 675-mg LAI is administered (Figure6).4,6,10

Aripiprazole lauroxil 675-mg LAI is formulated with drug particles that are smaller than those in aripiprazole lauroxil LAI.11 The smaller particle size results in faster dissolution and a more rapid increase in plasma aripiprazole levels. Aripiprazole lauroxil 675-mg LAI is a single injection that should be given with one 30-mg dose of oral aripiprazole.10 This combination results in aripiprazole concentrations that are comparable to aripiprazole lauroxil LAI and oral aripiprazole overlap for 3 weeks after the first injection.10
Continue to: The starting dose of aripiprazole lauroxil LAI...
The starting dose of aripiprazole lauroxil LAI may be administered on the same day as aripiprazole lauroxil 675-mg LAI and the 30-mg oral aripiprazole dose, or it may be administered up to 10 days after.10 Aripiprazole lauroxil LAI and aripiprazole lauroxil 675-mg LAI are not interchangeable due to differing pharmacokinetic profiles.6,10 Aripiprazole lauroxil 675-mg LAI may be used to re-initiate treatment in a patient who missed doses of aripiprazole lauroxil LAI.10 Aripiprazole lauroxil LAI and aripiprazole lauroxil 675 mg should not be injected together into the same deltoid or gluteal muscle.
Be mindful of differences in dosing windows
Each LAIA has a specific frequency recommendation, but due to scheduling or other factors, it may not be possible for patients to receive their injection on the specified day. The prescribing information for some LAIAs provides a dosing window (Table 41-7). The prescribing information for risperidone LAI, olanzapine pamoate LAI, and risperidone LAI for SQ does not specify how many days the injection can be administered before or after the due date; however, the prescribing information for risperidone LAI for SQ indicates that if the injection is not given on the due date, it should be administered as soon as possible after that.1,5,7

Paliperidone palmitate monthly injection and paliperidone palmitate 3-month LAI have the clearest recommendations for a dosing window. Paliperidone palmitate monthly injection may be administered 7 days before or after the 4-week due date, and paliperidone palmitate 3-month LAI can be administered 14 days before or after the 12-week due date.2,3
Aripiprazole LAI should not be administered sooner than 26 days after the previous injection, which means that it can be administered up to 2 days before the 4-week due date.4 If administered after the due date, it should be given as soon as possible, although oral overlap is not needed until ≥7 days past the due date.4
Aripiprazole lauroxil LAI has similar recommendations to aripiprazole LAI in that it should not be administered sooner than 14 days after the previous injection.6 If it is given after the due date, it should be administered as soon as possible; oral overlap/starting dose is needed if it has been ≥2 to 4 weeks since the due date, depending on which dose and frequency the patient is receiving.6
Continue to: Recommendations for missed doses
Recommendations for missed doses
Each LAIA has specific recommendations for missed dosing. Carpenter and Wong12 reviewed the recommendations for managing missed LAIA doses in
Consider patient preference
Patient preference for the type and location of the injection may factor into a clinician’s choice of LAIA (Table 51-7,10). Risperidone LAI for SQ is the only LAIA that is administered as an SQ abdominal injection.7 All other LAIAs are IM injections in the deltoid or gluteal muscle.1-6 All doses of risperidone LAI, paliperidone palmitate 3-month LAI, aripiprazole LAI, and aripiprazole lauroxil 675-mg LAI can be administered in the deltoid or gluteal muscle.1,3,4,10 Deltoid administration is required for the 2 starting doses of paliperidone palmitate monthly injection, but maintenance doses can be administered in the deltoid or gluteal muscle. Because administration into the deltoid results in a higher concentration of the drug compared with gluteal administration, administering the 2 starting doses of paliperidone palmitate monthly injection into the deltoid helps to rapidly attain therapeutic concentrations.2 Olanzapine pamoate LAI should be administered only in the gluteal muscle.5 The 441-mg dose of aripiprazole lauroxil LAI may be administered in the deltoid or gluteal muscle, but all other doses of aripiprazole lauroxil LAI should be administered only in the gluteal muscle.6

Storage
Most LAIAs can be stored at room temperature2-6; however, risperidone LAI and risperidone LAI for SQ need to be stored in the refrigerator. Both risperidone LAI and risperidone LAI for SQ may be kept at room temperature for up to 7 days. If they are not used within 7 days at room temperature, they should be discarded.1,7
Clinical pearls for specific LAIAs
Aripiprazole LAI. The recommended starting and maintenance dose for aripiprazole LAI is 400 mg monthly, unless the patient has drug interactions or other factors that require dose adjustment. If patients experience adverse reactions to the 400-mg dose, a reduction to 300 mg monthly could be considered.4
Olanzapine pamoate LAI has a Risk Evaluation and Mitigation Strategy (REMS) due to the potential for post-injection delirium/sedation syndrome (PDSS). Prescribing clinicians, dispensing pharmacies, and administering health care facilities must all be certified to prescribe, dispense, or administer olanzapine pamoate LAI. The patient must also be enrolled in the REMS program.13 Patients must be observed by health care staff for 3 hours after receiving a dose of olanzapine pamoate LAI to monitor for signs and symptoms of PDSS.5
Continue to: Risperidone LAI
Risperidone LAI. When increasing the dose of risperidone LAI, do not expect to see the clinical effects of the new dose earlier than 3 weeks after initiating the higher dose, because the main release of the medication starts at 3 weeks after the injection.1
Risperidone LAI for SQ has specific recommendations for the LAI dose based on whether the patient was stable when receiving 3 or 4 mg/d of oral risperidone. If patients are stable on <3 or >4 mg/d, they may not be candidates for risperidone LAI for SQ.7
Table 61-7,10 lists additional factors to consider when prescribing a specific LAIA.
Bottom Line
Second-generation long-acting injectable antipsychotics (LAIAs) have the potential to increase medication adherence. There are important differences among the 7 currently available LAIAs. For effective prescribing, clinicians need to understand each medication’s unique aspects, including dosing options, frequency, need for oral antipsychotic overlap, and route of administration.
Related Resources
- Correll CU, Citrome L, Haddad PM, et al. The use of long-acting injectable antipsychotics in schizophrenia: evaluating the evidence. J Clin Psychiatry. 2016;77(suppl 3):1-24.
- Peters L, Krogmann A, von Hardenberg L, et al. Long-acting injections in schizophrenia: a 3-year update on randomized controlled trials published January 2016-March 2019. Curr Psychiatry Rep. 2019;21(12):124.
Drug Brand Names
Aripiprazole • Abilify
Aripiprazole long-acting injectable • Abilify Maintena
Aripiprazole lauroxil extended-release injectable suspension • Aristada
Aripiprazole lauroxil 675 mg • Aristada Initio
Olanzapine pamoate long-acting injection • Zyprexa Relprevv
Paliperidone palmitate monthly long-acting injection • Invega Sustenna
Paliperidone palmitate 3-month injection • Invega Trinza
Risperidone • Risperdal
Risperidone long-acting injection • Risperdal Consta
Risperidone long-acting injection for SQ • Perseris
There are currently 7 FDA-approved second-generation long-acting injectable antipsychotics (LAIAs).1-7 These LAIAs provide a unique dosage form that allows patients to receive an antipsychotic without taking oral medications every day, or multiple times per day. This may be an appealing option for patients and clinicians, but because there are several types of LAIAs available, it may be difficult to determine which LAIA characteristics are best for a given patient.
Since the FDA approved the first second-generation LAIA, risperidone long-acting injectable (LAI),1 in 2003, 6 additional second-generation LAIAs have been approved:
- aripiprazole LAI
- aripiprazole lauroxil LAI
- olanzapine pamoate LAI
- paliperidone palmitate monthly injection
- paliperidone palmitate 3-month LAI
- risperidone LAI for subcutaneous (SQ) injection.
When discussing medication options with patients, clinicians need to consider factors that are unique to each LAIA. In this article, I describe the similarities and differences among the second-generation LAIAs, and address common questions about these medications.
A major potential benefit: Increased adherence
One potential benefit of all LAIAs is increased medication adherence compared with oral antipsychotics. One meta-analysis of 21 randomized controlled trials (RCTs) that compared LAIAs with oral antipsychotics and included 5,176 patients found that LAIAs had a similar efficacy to oral antipsychotics in preventing relapse.8 However, a meta-analysis of 25 mirror-image studies comparing LAIAs with oral antipsychotics that included 5,940 patients found that LAIAs were superior in preventing hospitalization.9 In these mirror-image studies, participants received oral antipsychotics first and then switched to LAIAs, and the 2 study periods were compared. Because mirror-image studies are observational, participants do not engage with research teams to the extent that they do in RCTs.9 Although mirror-image studies have limitations, participants in these studies may be a better representation of patients encountered in clinical practice due to the extensive monitoring and follow-up RCT participants typically receive.9
Differences in FDA-approved indications
The 7 currently available LAIAs vary in terms of FDA-approved indications, dose options, frequency, need for oral antipsychotic overlap, route of administration, and other factors. Table 11-7 summarizes some of these differences. Although all second-generation LAIAs are approved for schizophrenia,1-7 risperidone LAI and aripiprazole LAI are also approved for bipolar I disorder.1,4 Paliperidone palmitate monthly injection is the only LAIA approved for treating patients with schizoaffective disorder.2
Starting doses
For most LAIAs, the starting dose is the same as the maintenance dose (Table 11-7). One exception is paliperidone palmitate monthly injection, which requires a 234-mg dose on Day 1 followed by a 156-mg dose on Day 8 for all patients, regardless of the maintenance dose required.2 The 156-mg dose may be given 4 days before or after Day 8.2 The first maintenance dose of paliperidone palmitate monthly injection should be administered 5 weeks after the 234-mg dose on Day 1.2 Before starting paliperidone palmitate 3-month injection, patients should be stable on paliperidone palmitate monthly injection for 4 months, and the 2 most recent doses of paliperidone palmitate monthly injection should be the same.3

Maintenance doses
Dosing frequency may be an important factor for some patients when deciding to receive a LAIA. The frequency of the maintenance doses for all second-generation LAIAs varies from every 2 weeks to 12 weeks (Table 11-7). Paliperidone palmitate 3-month LAI is the only LAIA that is administered every 12 weeks.3 Some dosages of aripiprazole lauroxil LAI are administered every 6 or 8 weeks.6 All other second-generation LAIAs are given every 2 to 4 weeks.
Continue to: Start with an oral antipsychotic
Start with an oral antipsychotic
Before starting any LAIA, patients should receive the oral formulation of that antipsychotic to establish tolerability.1-7 Four of the 7 available LAIAs have an oral-to-LAI dose equivalency recommendation in their prescribing information (Table 22,5-7). This can help clinicians estimate the LAIA maintenance dose required to control a patient’s symptoms. If a dose adjustment is needed once a patient starts an LAIA, the dose adjustment can be made when the next injection is due.2

There are 2 important considerations when prescribing olanzapine pamoate LAI. First, the recommended dose for olanzapine pamoate LAI based on oral olanzapine doses differs during the first 8 weeks of treatment compared with after 8 weeks of treatment (Table 22,5-7). Additionally, because there are both short-acting and long-acting injections of olanzapine, it is essential to choose the correct formulation when prescribing this medication.5
Overlap with an oral antipsychotic might be necessary
Administration of several of the LAIAs may require overlap with an oral antipsychotic (Table 31,2,4-7). Patients who refuse to take oral medications may benefit from one of the LAIAs that does not require oral overlap—paliperidone palmitate monthly injection, olanzapine pamoate LAI, and risperidone LAI for SQ.2,5,7 Risperidone LAI requires overlap with oral risperidone for 3 weeks.1

Aripiprazole is available in 2 LAI formulations: aripiprazole LAI and aripiprazole lauroxil LAI. Aripiprazole lauroxil is a prodrug of aripiprazole, and these 2 LAI medications differ in available dose options and dosing frequency.4,6 Aripiprazole LAI requires an oral overlap for 2 weeks after the first injection, whereas aripiprazole lauroxil LAI requires 3 weeks of oral overlap unless aripiprazole lauroxil 675-mg LAI is administered (Figure6).4,6,10

Aripiprazole lauroxil 675-mg LAI is formulated with drug particles that are smaller than those in aripiprazole lauroxil LAI.11 The smaller particle size results in faster dissolution and a more rapid increase in plasma aripiprazole levels. Aripiprazole lauroxil 675-mg LAI is a single injection that should be given with one 30-mg dose of oral aripiprazole.10 This combination results in aripiprazole concentrations that are comparable to aripiprazole lauroxil LAI and oral aripiprazole overlap for 3 weeks after the first injection.10
Continue to: The starting dose of aripiprazole lauroxil LAI...
The starting dose of aripiprazole lauroxil LAI may be administered on the same day as aripiprazole lauroxil 675-mg LAI and the 30-mg oral aripiprazole dose, or it may be administered up to 10 days after.10 Aripiprazole lauroxil LAI and aripiprazole lauroxil 675-mg LAI are not interchangeable due to differing pharmacokinetic profiles.6,10 Aripiprazole lauroxil 675-mg LAI may be used to re-initiate treatment in a patient who missed doses of aripiprazole lauroxil LAI.10 Aripiprazole lauroxil LAI and aripiprazole lauroxil 675 mg should not be injected together into the same deltoid or gluteal muscle.
Be mindful of differences in dosing windows
Each LAIA has a specific frequency recommendation, but due to scheduling or other factors, it may not be possible for patients to receive their injection on the specified day. The prescribing information for some LAIAs provides a dosing window (Table 41-7). The prescribing information for risperidone LAI, olanzapine pamoate LAI, and risperidone LAI for SQ does not specify how many days the injection can be administered before or after the due date; however, the prescribing information for risperidone LAI for SQ indicates that if the injection is not given on the due date, it should be administered as soon as possible after that.1,5,7

Paliperidone palmitate monthly injection and paliperidone palmitate 3-month LAI have the clearest recommendations for a dosing window. Paliperidone palmitate monthly injection may be administered 7 days before or after the 4-week due date, and paliperidone palmitate 3-month LAI can be administered 14 days before or after the 12-week due date.2,3
Aripiprazole LAI should not be administered sooner than 26 days after the previous injection, which means that it can be administered up to 2 days before the 4-week due date.4 If administered after the due date, it should be given as soon as possible, although oral overlap is not needed until ≥7 days past the due date.4
Aripiprazole lauroxil LAI has similar recommendations to aripiprazole LAI in that it should not be administered sooner than 14 days after the previous injection.6 If it is given after the due date, it should be administered as soon as possible; oral overlap/starting dose is needed if it has been ≥2 to 4 weeks since the due date, depending on which dose and frequency the patient is receiving.6
Continue to: Recommendations for missed doses
Recommendations for missed doses
Each LAIA has specific recommendations for missed dosing. Carpenter and Wong12 reviewed the recommendations for managing missed LAIA doses in
Consider patient preference
Patient preference for the type and location of the injection may factor into a clinician’s choice of LAIA (Table 51-7,10). Risperidone LAI for SQ is the only LAIA that is administered as an SQ abdominal injection.7 All other LAIAs are IM injections in the deltoid or gluteal muscle.1-6 All doses of risperidone LAI, paliperidone palmitate 3-month LAI, aripiprazole LAI, and aripiprazole lauroxil 675-mg LAI can be administered in the deltoid or gluteal muscle.1,3,4,10 Deltoid administration is required for the 2 starting doses of paliperidone palmitate monthly injection, but maintenance doses can be administered in the deltoid or gluteal muscle. Because administration into the deltoid results in a higher concentration of the drug compared with gluteal administration, administering the 2 starting doses of paliperidone palmitate monthly injection into the deltoid helps to rapidly attain therapeutic concentrations.2 Olanzapine pamoate LAI should be administered only in the gluteal muscle.5 The 441-mg dose of aripiprazole lauroxil LAI may be administered in the deltoid or gluteal muscle, but all other doses of aripiprazole lauroxil LAI should be administered only in the gluteal muscle.6

Storage
Most LAIAs can be stored at room temperature2-6; however, risperidone LAI and risperidone LAI for SQ need to be stored in the refrigerator. Both risperidone LAI and risperidone LAI for SQ may be kept at room temperature for up to 7 days. If they are not used within 7 days at room temperature, they should be discarded.1,7
Clinical pearls for specific LAIAs
Aripiprazole LAI. The recommended starting and maintenance dose for aripiprazole LAI is 400 mg monthly, unless the patient has drug interactions or other factors that require dose adjustment. If patients experience adverse reactions to the 400-mg dose, a reduction to 300 mg monthly could be considered.4
Olanzapine pamoate LAI has a Risk Evaluation and Mitigation Strategy (REMS) due to the potential for post-injection delirium/sedation syndrome (PDSS). Prescribing clinicians, dispensing pharmacies, and administering health care facilities must all be certified to prescribe, dispense, or administer olanzapine pamoate LAI. The patient must also be enrolled in the REMS program.13 Patients must be observed by health care staff for 3 hours after receiving a dose of olanzapine pamoate LAI to monitor for signs and symptoms of PDSS.5
Continue to: Risperidone LAI
Risperidone LAI. When increasing the dose of risperidone LAI, do not expect to see the clinical effects of the new dose earlier than 3 weeks after initiating the higher dose, because the main release of the medication starts at 3 weeks after the injection.1
Risperidone LAI for SQ has specific recommendations for the LAI dose based on whether the patient was stable when receiving 3 or 4 mg/d of oral risperidone. If patients are stable on <3 or >4 mg/d, they may not be candidates for risperidone LAI for SQ.7
Table 61-7,10 lists additional factors to consider when prescribing a specific LAIA.
Bottom Line
Second-generation long-acting injectable antipsychotics (LAIAs) have the potential to increase medication adherence. There are important differences among the 7 currently available LAIAs. For effective prescribing, clinicians need to understand each medication’s unique aspects, including dosing options, frequency, need for oral antipsychotic overlap, and route of administration.
Related Resources
- Correll CU, Citrome L, Haddad PM, et al. The use of long-acting injectable antipsychotics in schizophrenia: evaluating the evidence. J Clin Psychiatry. 2016;77(suppl 3):1-24.
- Peters L, Krogmann A, von Hardenberg L, et al. Long-acting injections in schizophrenia: a 3-year update on randomized controlled trials published January 2016-March 2019. Curr Psychiatry Rep. 2019;21(12):124.
Drug Brand Names
Aripiprazole • Abilify
Aripiprazole long-acting injectable • Abilify Maintena
Aripiprazole lauroxil extended-release injectable suspension • Aristada
Aripiprazole lauroxil 675 mg • Aristada Initio
Olanzapine pamoate long-acting injection • Zyprexa Relprevv
Paliperidone palmitate monthly long-acting injection • Invega Sustenna
Paliperidone palmitate 3-month injection • Invega Trinza
Risperidone • Risperdal
Risperidone long-acting injection • Risperdal Consta
Risperidone long-acting injection for SQ • Perseris
1. Risperdal Consta [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
2. Invega Sustenna [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
3. Invega Trinza [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
4. Abilify Maintena [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; 2019.
5. Zyprexa Relprevv [package insert]. Indianapolis; IN: Eli Lilly and Co.; 2019.
6. Aristada [package insert]. Waltham, MA: Alkermes, Inc.; 2019.
7. Perseris [package insert]. North Chesterfield, VA: Indivior, Inc.; 2018.
8. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213.
9. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965.
10. Aristada Initio [package insert]. Waltham, MA: Alkermes, Inc.; 2019.
11. Jain R, Meyer J, Wehr A, et al. Size matters: the importance of particle size in a newly developed injectable formulation for the treatment of schizophrenia. CNS Spectr. 2019:1-8.
12. Carpenter J, Wong KK. Long-acting injectable antipsychotics: what to do about missed doses. Current Psychiatry. 2018;17(7):10-12,14-19,56.
13. US Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS) zyprexa relprevv (olanzapine). https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=74. Updated April 11, 2019. Accessed January 27, 2020.
1. Risperdal Consta [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
2. Invega Sustenna [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
3. Invega Trinza [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019.
4. Abilify Maintena [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; 2019.
5. Zyprexa Relprevv [package insert]. Indianapolis; IN: Eli Lilly and Co.; 2019.
6. Aristada [package insert]. Waltham, MA: Alkermes, Inc.; 2019.
7. Perseris [package insert]. North Chesterfield, VA: Indivior, Inc.; 2018.
8. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213.
9. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965.
10. Aristada Initio [package insert]. Waltham, MA: Alkermes, Inc.; 2019.
11. Jain R, Meyer J, Wehr A, et al. Size matters: the importance of particle size in a newly developed injectable formulation for the treatment of schizophrenia. CNS Spectr. 2019:1-8.
12. Carpenter J, Wong KK. Long-acting injectable antipsychotics: what to do about missed doses. Current Psychiatry. 2018;17(7):10-12,14-19,56.
13. US Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS) zyprexa relprevv (olanzapine). https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=74. Updated April 11, 2019. Accessed January 27, 2020.
Transporting the high-risk psychiatric patient: Clinical and legal challenges
“I’ve given up, doctor. We gave it our best, but I am at the point where I want to end my life.” You receive this call at 2 a.m., and you’re flooded with a series of emotions and are bewildered – until your training kicks in.
Countless hours of working with patients in emergency department settings while on call as a resident inform your ability to triage the situation. Years of supervision guide your supportive statements as you work collaboratively with your patient to address the emotional and existential distress. As the call proceeds and you realize that your patient will require hospitalization, you are struck by a sobering question: “How am I going to arrange for my patient to go to the nearest hospital in the middle of the night?”
The options for transporting patients with serious mental illness (SMI) typically vary from bad to worse and usually filter down to three possibilities:
1. Get a friend or family member to transport them to the nearest ED.
2. Call emergency medical services (EMS) for transport to the nearest ED.
3. Call the police and request transport to the nearest ED.
Several factors would determine which of those options you would use alone or in combination. Current training paradigms for mental health professionals offer a limited body of literature on evidenced-based strategies for patients with SMI. At best – if not handled properly – these experiences can aggravate patients’ mental health conditions. At worst, they can lead to the loss of our patients. Together, we have more than 40 years’ experience working in complex mental health care systems that run the gamut, from providing direct clinical care to directing mental health care divisions.
Sobering statistics prevail
In 2017, suicide was the 10th leading cause of death in the United States, accounting for more than 47,000 deaths. Suicide was the second-leading cause of death among individuals aged 10-34 and the fourth leading cause of death for individuals aged 35- 54.1 In 2017, more than 70,200 Americans died from a drug overdose, including illicit drugs and prescription opioids.2 Drug overdose deaths nearly quadrupled between 1999 and 2017, from 16,849 deaths to 70,237, respectively.2
The life expectancy of an American with SMI is 14-32 years less than that of the average population.3 Those numbers are on par with many sub-Saharan countries, including Sudan and Ethiopia, and surpass the health disparities for most racial and ethnic groups.
The decrease in life expectancy for people with SMI is rarely the result of suicide but rather the effect of medical comorbidities, including heart disease, stroke, pulmonary disease, diabetes, and cancer.3 Individuals with SMI are much more likely to suffer from chronic illnesses that are associated with co-occurring addictions, side effects of psychotropic medications, and social determinants of mental health, such as poverty.
Major depressive disorder among people with SMI presents acute and chronic medical risks. For example, people with major depressive disorder are at a higher risk for stroke and cardiovascular disease.4 There is a threefold increase in cardiac-related deaths for individuals who experience depression after a heart attack.5 In addition, depression increases the risk of cardiac-related death after a heart attack more than any other risk factor, except for congestive heart failure.6
The transportation challenge
When the complexity of psychiatric conditions is conceptualized to include decreased life expectancy because of medical comorbidities, medical complications that can result from psychotropic medications, and the high incidence of co-occurring substance use disorders (SUDs), it is clear why we are concerned about the type of transport used for patients experiencing a psychiatric emergency.
A common practice for patients presenting for medical treatment for a mental health condition is for them to be transported by law enforcement in handcuffs, or by private vehicle, because of a lack of medical transport options.
One tragic example occurred on Sept. 18, 2018, when two Horry County, S.C., deputies were providing a hospital-to-hospital transport for two mental health patients. The patients, who had no legal charges and were not under arrest, were locked in a transport cage in the back of a law enforcement vehicle for transport during Hurricane Florence. While the driver tried to move through floodwaters, the car stalled and became submerged. The patients were unable to open the locked cage door, and the deputies responsible for the transport did not have a key to unlock the cage.
The two women died as a result of the incident. Both deputies are facing two counts of involuntary manslaughter – with a potential prison sentence of 5 years each.7
The system that promotes the use of law enforcement to transport medical patients who are not in legal custody perpetuates a high risk to patients. Also at risk are law enforcement professionals who often are not adequately trained to evaluate, intervene, and manage patients in need of medical treatment (for acute medical, SUDs, or mental illness).
Recommendations for transporting high-risk patients
The legal and regulatory standards required by the Centers for Medicare & Medicaid Services guidelines8 for behavioral health patients are consistent in saying that law enforcement restraints cannot be used in hospital settings – unless the patient is under law enforcement custody. If the patient is admitted to an inpatient psychiatric unit, law enforcement restraints are never allowed to be used. Despite those guidelines for handling these situations within hospital settings, a similar standard does not exist for interhospital transport of high-risk behavioral health patients.
Patients often are transported between facilities by law enforcement, and at times, with the use of law enforcement restraints. However, use of law enforcement for interhospital transport of patients not in the custody of law enforcement is both detrimental to and clinically dangerous for the patient. Not only does the scenario put the patient at risk, but it also places law enforcement professionals at risk with the potential for legal liability if the patient medically decompensates during the transport.
Also, using law enforcement to intervene and provide transportation for behavioral health patients in crisis might cause further harm to the patient’s psychological condition and deter the patient from presenting for treatment.
The appropriate level of interfacility transport for a behavioral health patient requiring an inpatient level of care is secure transport with trained medical professionals. If necessary, restraint guidelines should follow the same standard as they do in hospital settings. In situations with high risk for violence or agitation, law enforcement should be requested to accompany EMS during the transport. Handling the situation in this way allows for a collaborative approach to provide adequate medical support, and provide for the physical safety and security of the patient – and of EMS personnel.
Across police and sheriffs’ departments nationwide, law enforcement officers are required to transport mental health patients from one location or hospital to another. Hospitals and outpatient locations, often with no alternative, rely on local law enforcement agencies to provide safe transport between acute and mental health facilities. Departmental policies and the widespread belief that mental health patients automatically pose a substantial danger to themselves or others have led to these patients being handcuffed and put in the back of police cruisers. Essentially, they are treated like criminals who are under arrest.
The CMS has strict criteria for ordering, applying, evaluating, reevaluating, and reporting restraint-associated deaths. Likewise, The Joint Commission, during its accreditation survey, rightly scrutinizes a facility’s use of restraints. Both the CMS and The Joint Commission define restraint by the function of the device and not the type of the device.
According to the CMS, a restraint is any manual method, physical or mechanic device, material, or equipment that immobilizes or reduces the ability of a patient to move his or her arms, legs, body, or head freely. Within a police cruiser, handcuffs on a mental health patient who is not under arrest would fall squarely within this definition of restraint.
A patient’s current behavior or clinical needs – not history or diagnosis – should determine whether a restraint is warranted. A patient experiencing a psychiatric crisis who is not under arrest might very well escalate and become destructive and combative when being placed in handcuffs in the back of a police vehicle. What should police do at that point? Should they arrest the individual? We offer the following five recommendations.
Key recommendations
- Patients who require interhospital transport for an acute behavioral health crisis should be transported by medical professionals with the training and expertise to manage mental health and medical conditions.
- Interhospital transport restraint guidelines should be in line with CMS guidelines for hospital restraints. No patient that is not under law enforcement custody should be transported in law enforcement restraints.
- Patients at high risk for violence/agitation (an underlying condition causing violence/agitation may include dementia, traumatic brain injuries, delirium, SUDs, or psychiatric conditions) should be managed in accordance with medically indicated intervention such as medical restraints or medications.
- If required, a request should be made for law enforcement officers to accompany EMS to provide support and to ensure the safety of the patient and EMS professionals during the transport.
- EMS professionals should receive specific training to manage acute psychiatric and SUD conditions, and should be aware of the life-threatening medical complications that can result from SUDs and psychiatric conditions/medications.
We recognize that patients with SMI require clinicians to work collaboratively, perhaps with law enforcement, to safely transport them from one point to another. Only when the rules we have outlined are followed will our patients get transported in such ways that allow them to get the care they need.
Dr. Norris is editor-in-chief of MDedge Psychiatry, and assistant professor of psychiatry and behavioral sciences at George Washington University, Washington. He serves as assistant dean of student affairs at the university, and medical director of psychiatric and behavioral sciences at GWU Hospital. Ms. Palmieri is a managing partner of Healthcare Legal Education & Consulting Network (HLECN), and the chief clinical officer and cofounder of XFERALL. HLECN focuses on clinical education and training, and XFERALL is a company that provides a technology platform aimed at improving access to care for behavioral health and medical patients. Ms. Slater is a managing partner of legal education for HLECN, and a trial attorney at Colley Shroyer & Abraham in Columbus, Ohio. Mr. Whaley is director of the safety and security department at GWU Hospital.
References
1. National Institute of Mental Health: Suicide.
2. National Institute on Drug Abuse. Overdose death rates. Revised January 2019.
3. Post by former NIMH Director Thomas Insel: No health without mental health. Sept. 6, 2011.
4. Dhar AK and Barton DA. Depression and the link with cardiovascular disease. Front Psychiatry. 2016 Mar 21;7:33.
5. Lichtman JH et al. Depression and coronary heart disease. Circulation. 2008;118:1768-75.
6. Hare DL et al. Depression and cardiovascular disease: A clinical review. European Heart J. 2014 Jun;35(21):1365-72.
7. “ ‘How many people have to die?’ SC mental health patients endure nightmare transport.” The News & Observer. 2019 May 29.
8. 42 C.F.R. Part 482. Part IV. Department of Health and Human Services. Medicare & Medicaid programs; Hospital conditions of participation; Patients’ rights; Final rule. 2006 Dec 8.
“I’ve given up, doctor. We gave it our best, but I am at the point where I want to end my life.” You receive this call at 2 a.m., and you’re flooded with a series of emotions and are bewildered – until your training kicks in.
Countless hours of working with patients in emergency department settings while on call as a resident inform your ability to triage the situation. Years of supervision guide your supportive statements as you work collaboratively with your patient to address the emotional and existential distress. As the call proceeds and you realize that your patient will require hospitalization, you are struck by a sobering question: “How am I going to arrange for my patient to go to the nearest hospital in the middle of the night?”
The options for transporting patients with serious mental illness (SMI) typically vary from bad to worse and usually filter down to three possibilities:
1. Get a friend or family member to transport them to the nearest ED.
2. Call emergency medical services (EMS) for transport to the nearest ED.
3. Call the police and request transport to the nearest ED.
Several factors would determine which of those options you would use alone or in combination. Current training paradigms for mental health professionals offer a limited body of literature on evidenced-based strategies for patients with SMI. At best – if not handled properly – these experiences can aggravate patients’ mental health conditions. At worst, they can lead to the loss of our patients. Together, we have more than 40 years’ experience working in complex mental health care systems that run the gamut, from providing direct clinical care to directing mental health care divisions.
Sobering statistics prevail
In 2017, suicide was the 10th leading cause of death in the United States, accounting for more than 47,000 deaths. Suicide was the second-leading cause of death among individuals aged 10-34 and the fourth leading cause of death for individuals aged 35- 54.1 In 2017, more than 70,200 Americans died from a drug overdose, including illicit drugs and prescription opioids.2 Drug overdose deaths nearly quadrupled between 1999 and 2017, from 16,849 deaths to 70,237, respectively.2
The life expectancy of an American with SMI is 14-32 years less than that of the average population.3 Those numbers are on par with many sub-Saharan countries, including Sudan and Ethiopia, and surpass the health disparities for most racial and ethnic groups.
The decrease in life expectancy for people with SMI is rarely the result of suicide but rather the effect of medical comorbidities, including heart disease, stroke, pulmonary disease, diabetes, and cancer.3 Individuals with SMI are much more likely to suffer from chronic illnesses that are associated with co-occurring addictions, side effects of psychotropic medications, and social determinants of mental health, such as poverty.
Major depressive disorder among people with SMI presents acute and chronic medical risks. For example, people with major depressive disorder are at a higher risk for stroke and cardiovascular disease.4 There is a threefold increase in cardiac-related deaths for individuals who experience depression after a heart attack.5 In addition, depression increases the risk of cardiac-related death after a heart attack more than any other risk factor, except for congestive heart failure.6
The transportation challenge
When the complexity of psychiatric conditions is conceptualized to include decreased life expectancy because of medical comorbidities, medical complications that can result from psychotropic medications, and the high incidence of co-occurring substance use disorders (SUDs), it is clear why we are concerned about the type of transport used for patients experiencing a psychiatric emergency.
A common practice for patients presenting for medical treatment for a mental health condition is for them to be transported by law enforcement in handcuffs, or by private vehicle, because of a lack of medical transport options.
One tragic example occurred on Sept. 18, 2018, when two Horry County, S.C., deputies were providing a hospital-to-hospital transport for two mental health patients. The patients, who had no legal charges and were not under arrest, were locked in a transport cage in the back of a law enforcement vehicle for transport during Hurricane Florence. While the driver tried to move through floodwaters, the car stalled and became submerged. The patients were unable to open the locked cage door, and the deputies responsible for the transport did not have a key to unlock the cage.
The two women died as a result of the incident. Both deputies are facing two counts of involuntary manslaughter – with a potential prison sentence of 5 years each.7
The system that promotes the use of law enforcement to transport medical patients who are not in legal custody perpetuates a high risk to patients. Also at risk are law enforcement professionals who often are not adequately trained to evaluate, intervene, and manage patients in need of medical treatment (for acute medical, SUDs, or mental illness).
Recommendations for transporting high-risk patients
The legal and regulatory standards required by the Centers for Medicare & Medicaid Services guidelines8 for behavioral health patients are consistent in saying that law enforcement restraints cannot be used in hospital settings – unless the patient is under law enforcement custody. If the patient is admitted to an inpatient psychiatric unit, law enforcement restraints are never allowed to be used. Despite those guidelines for handling these situations within hospital settings, a similar standard does not exist for interhospital transport of high-risk behavioral health patients.
Patients often are transported between facilities by law enforcement, and at times, with the use of law enforcement restraints. However, use of law enforcement for interhospital transport of patients not in the custody of law enforcement is both detrimental to and clinically dangerous for the patient. Not only does the scenario put the patient at risk, but it also places law enforcement professionals at risk with the potential for legal liability if the patient medically decompensates during the transport.
Also, using law enforcement to intervene and provide transportation for behavioral health patients in crisis might cause further harm to the patient’s psychological condition and deter the patient from presenting for treatment.
The appropriate level of interfacility transport for a behavioral health patient requiring an inpatient level of care is secure transport with trained medical professionals. If necessary, restraint guidelines should follow the same standard as they do in hospital settings. In situations with high risk for violence or agitation, law enforcement should be requested to accompany EMS during the transport. Handling the situation in this way allows for a collaborative approach to provide adequate medical support, and provide for the physical safety and security of the patient – and of EMS personnel.
Across police and sheriffs’ departments nationwide, law enforcement officers are required to transport mental health patients from one location or hospital to another. Hospitals and outpatient locations, often with no alternative, rely on local law enforcement agencies to provide safe transport between acute and mental health facilities. Departmental policies and the widespread belief that mental health patients automatically pose a substantial danger to themselves or others have led to these patients being handcuffed and put in the back of police cruisers. Essentially, they are treated like criminals who are under arrest.
The CMS has strict criteria for ordering, applying, evaluating, reevaluating, and reporting restraint-associated deaths. Likewise, The Joint Commission, during its accreditation survey, rightly scrutinizes a facility’s use of restraints. Both the CMS and The Joint Commission define restraint by the function of the device and not the type of the device.
According to the CMS, a restraint is any manual method, physical or mechanic device, material, or equipment that immobilizes or reduces the ability of a patient to move his or her arms, legs, body, or head freely. Within a police cruiser, handcuffs on a mental health patient who is not under arrest would fall squarely within this definition of restraint.
A patient’s current behavior or clinical needs – not history or diagnosis – should determine whether a restraint is warranted. A patient experiencing a psychiatric crisis who is not under arrest might very well escalate and become destructive and combative when being placed in handcuffs in the back of a police vehicle. What should police do at that point? Should they arrest the individual? We offer the following five recommendations.
Key recommendations
- Patients who require interhospital transport for an acute behavioral health crisis should be transported by medical professionals with the training and expertise to manage mental health and medical conditions.
- Interhospital transport restraint guidelines should be in line with CMS guidelines for hospital restraints. No patient that is not under law enforcement custody should be transported in law enforcement restraints.
- Patients at high risk for violence/agitation (an underlying condition causing violence/agitation may include dementia, traumatic brain injuries, delirium, SUDs, or psychiatric conditions) should be managed in accordance with medically indicated intervention such as medical restraints or medications.
- If required, a request should be made for law enforcement officers to accompany EMS to provide support and to ensure the safety of the patient and EMS professionals during the transport.
- EMS professionals should receive specific training to manage acute psychiatric and SUD conditions, and should be aware of the life-threatening medical complications that can result from SUDs and psychiatric conditions/medications.
We recognize that patients with SMI require clinicians to work collaboratively, perhaps with law enforcement, to safely transport them from one point to another. Only when the rules we have outlined are followed will our patients get transported in such ways that allow them to get the care they need.
Dr. Norris is editor-in-chief of MDedge Psychiatry, and assistant professor of psychiatry and behavioral sciences at George Washington University, Washington. He serves as assistant dean of student affairs at the university, and medical director of psychiatric and behavioral sciences at GWU Hospital. Ms. Palmieri is a managing partner of Healthcare Legal Education & Consulting Network (HLECN), and the chief clinical officer and cofounder of XFERALL. HLECN focuses on clinical education and training, and XFERALL is a company that provides a technology platform aimed at improving access to care for behavioral health and medical patients. Ms. Slater is a managing partner of legal education for HLECN, and a trial attorney at Colley Shroyer & Abraham in Columbus, Ohio. Mr. Whaley is director of the safety and security department at GWU Hospital.
References
1. National Institute of Mental Health: Suicide.
2. National Institute on Drug Abuse. Overdose death rates. Revised January 2019.
3. Post by former NIMH Director Thomas Insel: No health without mental health. Sept. 6, 2011.
4. Dhar AK and Barton DA. Depression and the link with cardiovascular disease. Front Psychiatry. 2016 Mar 21;7:33.
5. Lichtman JH et al. Depression and coronary heart disease. Circulation. 2008;118:1768-75.
6. Hare DL et al. Depression and cardiovascular disease: A clinical review. European Heart J. 2014 Jun;35(21):1365-72.
7. “ ‘How many people have to die?’ SC mental health patients endure nightmare transport.” The News & Observer. 2019 May 29.
8. 42 C.F.R. Part 482. Part IV. Department of Health and Human Services. Medicare & Medicaid programs; Hospital conditions of participation; Patients’ rights; Final rule. 2006 Dec 8.
“I’ve given up, doctor. We gave it our best, but I am at the point where I want to end my life.” You receive this call at 2 a.m., and you’re flooded with a series of emotions and are bewildered – until your training kicks in.
Countless hours of working with patients in emergency department settings while on call as a resident inform your ability to triage the situation. Years of supervision guide your supportive statements as you work collaboratively with your patient to address the emotional and existential distress. As the call proceeds and you realize that your patient will require hospitalization, you are struck by a sobering question: “How am I going to arrange for my patient to go to the nearest hospital in the middle of the night?”
The options for transporting patients with serious mental illness (SMI) typically vary from bad to worse and usually filter down to three possibilities:
1. Get a friend or family member to transport them to the nearest ED.
2. Call emergency medical services (EMS) for transport to the nearest ED.
3. Call the police and request transport to the nearest ED.
Several factors would determine which of those options you would use alone or in combination. Current training paradigms for mental health professionals offer a limited body of literature on evidenced-based strategies for patients with SMI. At best – if not handled properly – these experiences can aggravate patients’ mental health conditions. At worst, they can lead to the loss of our patients. Together, we have more than 40 years’ experience working in complex mental health care systems that run the gamut, from providing direct clinical care to directing mental health care divisions.
Sobering statistics prevail
In 2017, suicide was the 10th leading cause of death in the United States, accounting for more than 47,000 deaths. Suicide was the second-leading cause of death among individuals aged 10-34 and the fourth leading cause of death for individuals aged 35- 54.1 In 2017, more than 70,200 Americans died from a drug overdose, including illicit drugs and prescription opioids.2 Drug overdose deaths nearly quadrupled between 1999 and 2017, from 16,849 deaths to 70,237, respectively.2
The life expectancy of an American with SMI is 14-32 years less than that of the average population.3 Those numbers are on par with many sub-Saharan countries, including Sudan and Ethiopia, and surpass the health disparities for most racial and ethnic groups.
The decrease in life expectancy for people with SMI is rarely the result of suicide but rather the effect of medical comorbidities, including heart disease, stroke, pulmonary disease, diabetes, and cancer.3 Individuals with SMI are much more likely to suffer from chronic illnesses that are associated with co-occurring addictions, side effects of psychotropic medications, and social determinants of mental health, such as poverty.
Major depressive disorder among people with SMI presents acute and chronic medical risks. For example, people with major depressive disorder are at a higher risk for stroke and cardiovascular disease.4 There is a threefold increase in cardiac-related deaths for individuals who experience depression after a heart attack.5 In addition, depression increases the risk of cardiac-related death after a heart attack more than any other risk factor, except for congestive heart failure.6
The transportation challenge
When the complexity of psychiatric conditions is conceptualized to include decreased life expectancy because of medical comorbidities, medical complications that can result from psychotropic medications, and the high incidence of co-occurring substance use disorders (SUDs), it is clear why we are concerned about the type of transport used for patients experiencing a psychiatric emergency.
A common practice for patients presenting for medical treatment for a mental health condition is for them to be transported by law enforcement in handcuffs, or by private vehicle, because of a lack of medical transport options.
One tragic example occurred on Sept. 18, 2018, when two Horry County, S.C., deputies were providing a hospital-to-hospital transport for two mental health patients. The patients, who had no legal charges and were not under arrest, were locked in a transport cage in the back of a law enforcement vehicle for transport during Hurricane Florence. While the driver tried to move through floodwaters, the car stalled and became submerged. The patients were unable to open the locked cage door, and the deputies responsible for the transport did not have a key to unlock the cage.
The two women died as a result of the incident. Both deputies are facing two counts of involuntary manslaughter – with a potential prison sentence of 5 years each.7
The system that promotes the use of law enforcement to transport medical patients who are not in legal custody perpetuates a high risk to patients. Also at risk are law enforcement professionals who often are not adequately trained to evaluate, intervene, and manage patients in need of medical treatment (for acute medical, SUDs, or mental illness).
Recommendations for transporting high-risk patients
The legal and regulatory standards required by the Centers for Medicare & Medicaid Services guidelines8 for behavioral health patients are consistent in saying that law enforcement restraints cannot be used in hospital settings – unless the patient is under law enforcement custody. If the patient is admitted to an inpatient psychiatric unit, law enforcement restraints are never allowed to be used. Despite those guidelines for handling these situations within hospital settings, a similar standard does not exist for interhospital transport of high-risk behavioral health patients.
Patients often are transported between facilities by law enforcement, and at times, with the use of law enforcement restraints. However, use of law enforcement for interhospital transport of patients not in the custody of law enforcement is both detrimental to and clinically dangerous for the patient. Not only does the scenario put the patient at risk, but it also places law enforcement professionals at risk with the potential for legal liability if the patient medically decompensates during the transport.
Also, using law enforcement to intervene and provide transportation for behavioral health patients in crisis might cause further harm to the patient’s psychological condition and deter the patient from presenting for treatment.
The appropriate level of interfacility transport for a behavioral health patient requiring an inpatient level of care is secure transport with trained medical professionals. If necessary, restraint guidelines should follow the same standard as they do in hospital settings. In situations with high risk for violence or agitation, law enforcement should be requested to accompany EMS during the transport. Handling the situation in this way allows for a collaborative approach to provide adequate medical support, and provide for the physical safety and security of the patient – and of EMS personnel.
Across police and sheriffs’ departments nationwide, law enforcement officers are required to transport mental health patients from one location or hospital to another. Hospitals and outpatient locations, often with no alternative, rely on local law enforcement agencies to provide safe transport between acute and mental health facilities. Departmental policies and the widespread belief that mental health patients automatically pose a substantial danger to themselves or others have led to these patients being handcuffed and put in the back of police cruisers. Essentially, they are treated like criminals who are under arrest.
The CMS has strict criteria for ordering, applying, evaluating, reevaluating, and reporting restraint-associated deaths. Likewise, The Joint Commission, during its accreditation survey, rightly scrutinizes a facility’s use of restraints. Both the CMS and The Joint Commission define restraint by the function of the device and not the type of the device.
According to the CMS, a restraint is any manual method, physical or mechanic device, material, or equipment that immobilizes or reduces the ability of a patient to move his or her arms, legs, body, or head freely. Within a police cruiser, handcuffs on a mental health patient who is not under arrest would fall squarely within this definition of restraint.
A patient’s current behavior or clinical needs – not history or diagnosis – should determine whether a restraint is warranted. A patient experiencing a psychiatric crisis who is not under arrest might very well escalate and become destructive and combative when being placed in handcuffs in the back of a police vehicle. What should police do at that point? Should they arrest the individual? We offer the following five recommendations.
Key recommendations
- Patients who require interhospital transport for an acute behavioral health crisis should be transported by medical professionals with the training and expertise to manage mental health and medical conditions.
- Interhospital transport restraint guidelines should be in line with CMS guidelines for hospital restraints. No patient that is not under law enforcement custody should be transported in law enforcement restraints.
- Patients at high risk for violence/agitation (an underlying condition causing violence/agitation may include dementia, traumatic brain injuries, delirium, SUDs, or psychiatric conditions) should be managed in accordance with medically indicated intervention such as medical restraints or medications.
- If required, a request should be made for law enforcement officers to accompany EMS to provide support and to ensure the safety of the patient and EMS professionals during the transport.
- EMS professionals should receive specific training to manage acute psychiatric and SUD conditions, and should be aware of the life-threatening medical complications that can result from SUDs and psychiatric conditions/medications.
We recognize that patients with SMI require clinicians to work collaboratively, perhaps with law enforcement, to safely transport them from one point to another. Only when the rules we have outlined are followed will our patients get transported in such ways that allow them to get the care they need.
Dr. Norris is editor-in-chief of MDedge Psychiatry, and assistant professor of psychiatry and behavioral sciences at George Washington University, Washington. He serves as assistant dean of student affairs at the university, and medical director of psychiatric and behavioral sciences at GWU Hospital. Ms. Palmieri is a managing partner of Healthcare Legal Education & Consulting Network (HLECN), and the chief clinical officer and cofounder of XFERALL. HLECN focuses on clinical education and training, and XFERALL is a company that provides a technology platform aimed at improving access to care for behavioral health and medical patients. Ms. Slater is a managing partner of legal education for HLECN, and a trial attorney at Colley Shroyer & Abraham in Columbus, Ohio. Mr. Whaley is director of the safety and security department at GWU Hospital.
References
1. National Institute of Mental Health: Suicide.
2. National Institute on Drug Abuse. Overdose death rates. Revised January 2019.
3. Post by former NIMH Director Thomas Insel: No health without mental health. Sept. 6, 2011.
4. Dhar AK and Barton DA. Depression and the link with cardiovascular disease. Front Psychiatry. 2016 Mar 21;7:33.
5. Lichtman JH et al. Depression and coronary heart disease. Circulation. 2008;118:1768-75.
6. Hare DL et al. Depression and cardiovascular disease: A clinical review. European Heart J. 2014 Jun;35(21):1365-72.
7. “ ‘How many people have to die?’ SC mental health patients endure nightmare transport.” The News & Observer. 2019 May 29.
8. 42 C.F.R. Part 482. Part IV. Department of Health and Human Services. Medicare & Medicaid programs; Hospital conditions of participation; Patients’ rights; Final rule. 2006 Dec 8.
Severe infection tied to substance-induced psychosis, conversion to schizophrenia
Severe infection is linked to an increased risk of substance-induced psychosis and subsequent conversion to schizophrenia, new research suggests.
Results of the large, population-based study showed any infection was associated with a 30% increased risk for substance-induced psychosis. However, with more than a threefold increased risk for substance-induced psychosis, hepatitis was the infection most strongly linked to psychosis and the only infection associated with conversion to schizophrenia.
“Severe infections are associated with an increased risk of developing a substance-induced psychosis. Furthermore, hepatitis following substance-induced psychosis is associated with an increase in the risk of conversion to schizophrenia. Both of these observations support the hypothesis of an immunological component to psychosis,” wrote the investigators, led by Carsten Hjorthøj, PhD, MSc, Copenhagen Research Center for Mental Health, Denmark.
The study was published online Feb. 12 in the American Journal of Psychiatry.
Mechanism still poorly understood
Previous research suggests that infection increases the risk for schizophrenia, but this new study is the first to investigate the association between infection and substance-induced psychosis.
Using Danish national registry data, the researchers analyzed data on all individuals born in Denmark since 1981.
Any infection increased the risk for substance-induced psychosis in the fully-adjusted model (hazard ratio = 1.30; 95% confidence interval, 1.22–1.39; P less than .001). The risk was doubled in the first 2 years following a severe infection and stayed elevated for more than 20 years.
Hepatitis was the infection most strongly associated with substance-induced psychosis (HR = 3.42; 95% CI, 2.47–4.74; P less than .001) and only hepatitis predicted conversion to schizophrenia after substance-induced psychosis (HR = 1.87; 95% CI, 1.07–3.26; P = .03).
These results, the investigators note, “mirror previous findings on the association between infections and schizophrenia, including previous observations that the link is particularly strong for hepatitis.”
They also point out that the biological mechanisms through which infections would increase the risk for psychosis, including substance-induced psychosis, remain poorly understood.
“If the exact mechanisms underlying the psychotogenic properties of infections or the immune response can be identified, this is likely to lead to improvements in treatment for psychotic disorders. A further hope is that it may even be possible to use this knowledge for primary prevention of psychosis,” the authors wrote.
Interpret with caution
Commenting on the study, Ole Köhler-Forsberg, MD, from the Psychosis Research Unit, Aarhus University Hospital, Denmark, said in an interview that the findings support a potential immunologic link to schizophrenia.
“However,” he added, “as in every register-based study, no causality can be assumed, only associations. As mentioned by the authors, there may be residual confounding and confounding by indication.”
Also commenting on the study, René Kahn, MD, PhD, professor and chair of psychiatry at the Icahn School of Medicine at Mount Sinai in New York, noted in an interview that there is a lot of “indirect evidence” from epidemiologic studies like this one to suggest an immune component to psychosis.
“However, there is not a single piece of direct evidence linking the immune system to schizophrenia so far,” Dr. Kahn cautioned.
“Yes, people who have hepatitis have a higher risk of developing substance-induced psychosis, but this may be a spurious finding. It may very well be that people who are prone to hepatitis for whatever reasons are at higher risk for psychosis but there is no direct evidence that the two are related. This study doesn’t convince me at all that immune pathology is related to schizophrenia,” Dr. Kahn said.
The study was supported by a grant from the Lundbeck Foundation Initiative for Integrative Psychiatric Research (iPSYCH). The study authors, Dr. Kahn, and Dr. Köhler-Forsberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Severe infection is linked to an increased risk of substance-induced psychosis and subsequent conversion to schizophrenia, new research suggests.
Results of the large, population-based study showed any infection was associated with a 30% increased risk for substance-induced psychosis. However, with more than a threefold increased risk for substance-induced psychosis, hepatitis was the infection most strongly linked to psychosis and the only infection associated with conversion to schizophrenia.
“Severe infections are associated with an increased risk of developing a substance-induced psychosis. Furthermore, hepatitis following substance-induced psychosis is associated with an increase in the risk of conversion to schizophrenia. Both of these observations support the hypothesis of an immunological component to psychosis,” wrote the investigators, led by Carsten Hjorthøj, PhD, MSc, Copenhagen Research Center for Mental Health, Denmark.
The study was published online Feb. 12 in the American Journal of Psychiatry.
Mechanism still poorly understood
Previous research suggests that infection increases the risk for schizophrenia, but this new study is the first to investigate the association between infection and substance-induced psychosis.
Using Danish national registry data, the researchers analyzed data on all individuals born in Denmark since 1981.
Any infection increased the risk for substance-induced psychosis in the fully-adjusted model (hazard ratio = 1.30; 95% confidence interval, 1.22–1.39; P less than .001). The risk was doubled in the first 2 years following a severe infection and stayed elevated for more than 20 years.
Hepatitis was the infection most strongly associated with substance-induced psychosis (HR = 3.42; 95% CI, 2.47–4.74; P less than .001) and only hepatitis predicted conversion to schizophrenia after substance-induced psychosis (HR = 1.87; 95% CI, 1.07–3.26; P = .03).
These results, the investigators note, “mirror previous findings on the association between infections and schizophrenia, including previous observations that the link is particularly strong for hepatitis.”
They also point out that the biological mechanisms through which infections would increase the risk for psychosis, including substance-induced psychosis, remain poorly understood.
“If the exact mechanisms underlying the psychotogenic properties of infections or the immune response can be identified, this is likely to lead to improvements in treatment for psychotic disorders. A further hope is that it may even be possible to use this knowledge for primary prevention of psychosis,” the authors wrote.
Interpret with caution
Commenting on the study, Ole Köhler-Forsberg, MD, from the Psychosis Research Unit, Aarhus University Hospital, Denmark, said in an interview that the findings support a potential immunologic link to schizophrenia.
“However,” he added, “as in every register-based study, no causality can be assumed, only associations. As mentioned by the authors, there may be residual confounding and confounding by indication.”
Also commenting on the study, René Kahn, MD, PhD, professor and chair of psychiatry at the Icahn School of Medicine at Mount Sinai in New York, noted in an interview that there is a lot of “indirect evidence” from epidemiologic studies like this one to suggest an immune component to psychosis.
“However, there is not a single piece of direct evidence linking the immune system to schizophrenia so far,” Dr. Kahn cautioned.
“Yes, people who have hepatitis have a higher risk of developing substance-induced psychosis, but this may be a spurious finding. It may very well be that people who are prone to hepatitis for whatever reasons are at higher risk for psychosis but there is no direct evidence that the two are related. This study doesn’t convince me at all that immune pathology is related to schizophrenia,” Dr. Kahn said.
The study was supported by a grant from the Lundbeck Foundation Initiative for Integrative Psychiatric Research (iPSYCH). The study authors, Dr. Kahn, and Dr. Köhler-Forsberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Severe infection is linked to an increased risk of substance-induced psychosis and subsequent conversion to schizophrenia, new research suggests.
Results of the large, population-based study showed any infection was associated with a 30% increased risk for substance-induced psychosis. However, with more than a threefold increased risk for substance-induced psychosis, hepatitis was the infection most strongly linked to psychosis and the only infection associated with conversion to schizophrenia.
“Severe infections are associated with an increased risk of developing a substance-induced psychosis. Furthermore, hepatitis following substance-induced psychosis is associated with an increase in the risk of conversion to schizophrenia. Both of these observations support the hypothesis of an immunological component to psychosis,” wrote the investigators, led by Carsten Hjorthøj, PhD, MSc, Copenhagen Research Center for Mental Health, Denmark.
The study was published online Feb. 12 in the American Journal of Psychiatry.
Mechanism still poorly understood
Previous research suggests that infection increases the risk for schizophrenia, but this new study is the first to investigate the association between infection and substance-induced psychosis.
Using Danish national registry data, the researchers analyzed data on all individuals born in Denmark since 1981.
Any infection increased the risk for substance-induced psychosis in the fully-adjusted model (hazard ratio = 1.30; 95% confidence interval, 1.22–1.39; P less than .001). The risk was doubled in the first 2 years following a severe infection and stayed elevated for more than 20 years.
Hepatitis was the infection most strongly associated with substance-induced psychosis (HR = 3.42; 95% CI, 2.47–4.74; P less than .001) and only hepatitis predicted conversion to schizophrenia after substance-induced psychosis (HR = 1.87; 95% CI, 1.07–3.26; P = .03).
These results, the investigators note, “mirror previous findings on the association between infections and schizophrenia, including previous observations that the link is particularly strong for hepatitis.”
They also point out that the biological mechanisms through which infections would increase the risk for psychosis, including substance-induced psychosis, remain poorly understood.
“If the exact mechanisms underlying the psychotogenic properties of infections or the immune response can be identified, this is likely to lead to improvements in treatment for psychotic disorders. A further hope is that it may even be possible to use this knowledge for primary prevention of psychosis,” the authors wrote.
Interpret with caution
Commenting on the study, Ole Köhler-Forsberg, MD, from the Psychosis Research Unit, Aarhus University Hospital, Denmark, said in an interview that the findings support a potential immunologic link to schizophrenia.
“However,” he added, “as in every register-based study, no causality can be assumed, only associations. As mentioned by the authors, there may be residual confounding and confounding by indication.”
Also commenting on the study, René Kahn, MD, PhD, professor and chair of psychiatry at the Icahn School of Medicine at Mount Sinai in New York, noted in an interview that there is a lot of “indirect evidence” from epidemiologic studies like this one to suggest an immune component to psychosis.
“However, there is not a single piece of direct evidence linking the immune system to schizophrenia so far,” Dr. Kahn cautioned.
“Yes, people who have hepatitis have a higher risk of developing substance-induced psychosis, but this may be a spurious finding. It may very well be that people who are prone to hepatitis for whatever reasons are at higher risk for psychosis but there is no direct evidence that the two are related. This study doesn’t convince me at all that immune pathology is related to schizophrenia,” Dr. Kahn said.
The study was supported by a grant from the Lundbeck Foundation Initiative for Integrative Psychiatric Research (iPSYCH). The study authors, Dr. Kahn, and Dr. Köhler-Forsberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TNF-alpha, oxidative stress disturbance may play role in schizophrenia pathophysiology
Disturbance of tumor necrosis factor (TNF)–alpha and oxidative stress status may be involved in the pathophysiology of schizophrenia, new study results suggest.
In a study published in Psychoneuroendocrinology, the investigators collected blood samples from 119 patients with schizophrenia and 135 controls. Along with TNF-alpha, assays for the oxidative stress markers superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT), and malondialdehyde (MDA) were measured. The average illness duration in patients with schizophrenia was 8.23 months, and their average total Positive and Negative Syndrome Scale score was 87.64, reported Shiguang Zhu of Nanjing (China) Medical University and associates.
Serum levels of TNF-alpha and MDA were significantly higher (P = .007 for both), and GSH-Px levels were significantly lower (P = .005), in patients with schizophrenia, compared with controls, after Bonferroni correction. The interaction between GSH-Px and TNF-alpha was negatively associated with the presence of schizophrenia (odds ratio, 0.99; 95% confidence interval, 0.98-0.99; P = .001), and the interaction between MDA and TNF-alpha was positively associated with schizophrenia risk (OR, 1.61, 95% CI, 1.16-2.24, P = .004).
“It is worth[while] to note that [the] immune-inflammatory and oxidative stress hypothesis are just one of the theories for schizophrenic development, and other neurobiological theories such as neurodevelopmental dysfunction and hypothalamus-pituitary-adrenal axis hormones disturbance should be considered,” the investigators wrote. However, their study “suggests that TNF-alpha and disturbance of oxidative stress status as well as their interaction may be involved in the pathophysiology of schizophrenia.”
The study was supported by the National Natural Science Foundation of China, Shanghai Jiao Tong University Medical Engineering Foundation, Shanghai Jiao Tong University School of Medicine, and CAS Key Laboratory of Mental Health. The investigators reported that they had no conflicts of interest.
SOURCE: Zhu S et al. Psychoneuroendocrinology. 2020 Jan 30. doi: 10.1016/j.psyneuen.2020.104595.
Disturbance of tumor necrosis factor (TNF)–alpha and oxidative stress status may be involved in the pathophysiology of schizophrenia, new study results suggest.
In a study published in Psychoneuroendocrinology, the investigators collected blood samples from 119 patients with schizophrenia and 135 controls. Along with TNF-alpha, assays for the oxidative stress markers superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT), and malondialdehyde (MDA) were measured. The average illness duration in patients with schizophrenia was 8.23 months, and their average total Positive and Negative Syndrome Scale score was 87.64, reported Shiguang Zhu of Nanjing (China) Medical University and associates.
Serum levels of TNF-alpha and MDA were significantly higher (P = .007 for both), and GSH-Px levels were significantly lower (P = .005), in patients with schizophrenia, compared with controls, after Bonferroni correction. The interaction between GSH-Px and TNF-alpha was negatively associated with the presence of schizophrenia (odds ratio, 0.99; 95% confidence interval, 0.98-0.99; P = .001), and the interaction between MDA and TNF-alpha was positively associated with schizophrenia risk (OR, 1.61, 95% CI, 1.16-2.24, P = .004).
“It is worth[while] to note that [the] immune-inflammatory and oxidative stress hypothesis are just one of the theories for schizophrenic development, and other neurobiological theories such as neurodevelopmental dysfunction and hypothalamus-pituitary-adrenal axis hormones disturbance should be considered,” the investigators wrote. However, their study “suggests that TNF-alpha and disturbance of oxidative stress status as well as their interaction may be involved in the pathophysiology of schizophrenia.”
The study was supported by the National Natural Science Foundation of China, Shanghai Jiao Tong University Medical Engineering Foundation, Shanghai Jiao Tong University School of Medicine, and CAS Key Laboratory of Mental Health. The investigators reported that they had no conflicts of interest.
SOURCE: Zhu S et al. Psychoneuroendocrinology. 2020 Jan 30. doi: 10.1016/j.psyneuen.2020.104595.
Disturbance of tumor necrosis factor (TNF)–alpha and oxidative stress status may be involved in the pathophysiology of schizophrenia, new study results suggest.
In a study published in Psychoneuroendocrinology, the investigators collected blood samples from 119 patients with schizophrenia and 135 controls. Along with TNF-alpha, assays for the oxidative stress markers superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT), and malondialdehyde (MDA) were measured. The average illness duration in patients with schizophrenia was 8.23 months, and their average total Positive and Negative Syndrome Scale score was 87.64, reported Shiguang Zhu of Nanjing (China) Medical University and associates.
Serum levels of TNF-alpha and MDA were significantly higher (P = .007 for both), and GSH-Px levels were significantly lower (P = .005), in patients with schizophrenia, compared with controls, after Bonferroni correction. The interaction between GSH-Px and TNF-alpha was negatively associated with the presence of schizophrenia (odds ratio, 0.99; 95% confidence interval, 0.98-0.99; P = .001), and the interaction between MDA and TNF-alpha was positively associated with schizophrenia risk (OR, 1.61, 95% CI, 1.16-2.24, P = .004).
“It is worth[while] to note that [the] immune-inflammatory and oxidative stress hypothesis are just one of the theories for schizophrenic development, and other neurobiological theories such as neurodevelopmental dysfunction and hypothalamus-pituitary-adrenal axis hormones disturbance should be considered,” the investigators wrote. However, their study “suggests that TNF-alpha and disturbance of oxidative stress status as well as their interaction may be involved in the pathophysiology of schizophrenia.”
The study was supported by the National Natural Science Foundation of China, Shanghai Jiao Tong University Medical Engineering Foundation, Shanghai Jiao Tong University School of Medicine, and CAS Key Laboratory of Mental Health. The investigators reported that they had no conflicts of interest.
SOURCE: Zhu S et al. Psychoneuroendocrinology. 2020 Jan 30. doi: 10.1016/j.psyneuen.2020.104595.
FROM PSYCHONEUROENDOCRINOLOGY
Cigarette smoking is associated with prefrontal function in patients with schizophrenia
Patients with schizophrenia have decreased chronnectomic density in the dorsolateral prefrontal cortex, compared with healthy controls, and cigarette smoking in patients with schizophrenia may be associated with a degree of preserved function in that brain region, researchers reported. The results indicate that smoking may be associated with a preservation effect, but it “cannot restore patients’ prefrontal dysfunction to normal levels,” the researchers said.
The chronnectome depicts how brain functional connectivity patterns (i.e., the connectome) vary over time. “Therefore, the chronnectome may be an effective index to evaluate the smoking-related prefrontal functional changes in schizophrenia,” said Yun-Shuang Fan, a researcher at the Clinical Hospital of Chengdu Brain Science Institute in China, and colleagues in the report, which was published in Progress in Neuro-Psychopharmacology & Biological Psychiatry.
The investigators studied 49 patients with schizophrenia, including 22 smokers and 27 nonsmokers, and 43 healthy controls, including 22 smokers and 21 nonsmokers. Participants underwent resting-state functional magnetic resonance imaging, and the researchers analyzed chronnectomic density using a sliding-window method. The investigators examined interactions between smoking status and diagnosis.
Smoking was associated with reduced chronnectomic density in healthy controls, but increased density in patients with schizophrenia. The study provides a “framework to elaborate upon the self-medication hypothesis in schizophrenia” and sheds “some fresh light on the elevated rates of smoking in schizophrenia,” they said.
The study was relatively small, and patients’ use of antipsychotic medications, which can affect the connectome, may limit the results. In addition, the study’s cross-sectional design precludes knowing whether “smoking behavior is the cause or result of the prefrontal chronnectome alterations in schizophrenia,” the authors added.
The study was supported by the National Natural Science Foundation of China and the Sichuan Science and Technology Program. The researchers had no conflicts of interest.
SOURCE: Fan YS et al. Prog Neuropsychopharmacol Biol Psychiatry. 2020 Apr 20. doi: 10.1016/j.pnpbp.2020.109860.
Patients with schizophrenia have decreased chronnectomic density in the dorsolateral prefrontal cortex, compared with healthy controls, and cigarette smoking in patients with schizophrenia may be associated with a degree of preserved function in that brain region, researchers reported. The results indicate that smoking may be associated with a preservation effect, but it “cannot restore patients’ prefrontal dysfunction to normal levels,” the researchers said.
The chronnectome depicts how brain functional connectivity patterns (i.e., the connectome) vary over time. “Therefore, the chronnectome may be an effective index to evaluate the smoking-related prefrontal functional changes in schizophrenia,” said Yun-Shuang Fan, a researcher at the Clinical Hospital of Chengdu Brain Science Institute in China, and colleagues in the report, which was published in Progress in Neuro-Psychopharmacology & Biological Psychiatry.
The investigators studied 49 patients with schizophrenia, including 22 smokers and 27 nonsmokers, and 43 healthy controls, including 22 smokers and 21 nonsmokers. Participants underwent resting-state functional magnetic resonance imaging, and the researchers analyzed chronnectomic density using a sliding-window method. The investigators examined interactions between smoking status and diagnosis.
Smoking was associated with reduced chronnectomic density in healthy controls, but increased density in patients with schizophrenia. The study provides a “framework to elaborate upon the self-medication hypothesis in schizophrenia” and sheds “some fresh light on the elevated rates of smoking in schizophrenia,” they said.
The study was relatively small, and patients’ use of antipsychotic medications, which can affect the connectome, may limit the results. In addition, the study’s cross-sectional design precludes knowing whether “smoking behavior is the cause or result of the prefrontal chronnectome alterations in schizophrenia,” the authors added.
The study was supported by the National Natural Science Foundation of China and the Sichuan Science and Technology Program. The researchers had no conflicts of interest.
SOURCE: Fan YS et al. Prog Neuropsychopharmacol Biol Psychiatry. 2020 Apr 20. doi: 10.1016/j.pnpbp.2020.109860.
Patients with schizophrenia have decreased chronnectomic density in the dorsolateral prefrontal cortex, compared with healthy controls, and cigarette smoking in patients with schizophrenia may be associated with a degree of preserved function in that brain region, researchers reported. The results indicate that smoking may be associated with a preservation effect, but it “cannot restore patients’ prefrontal dysfunction to normal levels,” the researchers said.
The chronnectome depicts how brain functional connectivity patterns (i.e., the connectome) vary over time. “Therefore, the chronnectome may be an effective index to evaluate the smoking-related prefrontal functional changes in schizophrenia,” said Yun-Shuang Fan, a researcher at the Clinical Hospital of Chengdu Brain Science Institute in China, and colleagues in the report, which was published in Progress in Neuro-Psychopharmacology & Biological Psychiatry.
The investigators studied 49 patients with schizophrenia, including 22 smokers and 27 nonsmokers, and 43 healthy controls, including 22 smokers and 21 nonsmokers. Participants underwent resting-state functional magnetic resonance imaging, and the researchers analyzed chronnectomic density using a sliding-window method. The investigators examined interactions between smoking status and diagnosis.
Smoking was associated with reduced chronnectomic density in healthy controls, but increased density in patients with schizophrenia. The study provides a “framework to elaborate upon the self-medication hypothesis in schizophrenia” and sheds “some fresh light on the elevated rates of smoking in schizophrenia,” they said.
The study was relatively small, and patients’ use of antipsychotic medications, which can affect the connectome, may limit the results. In addition, the study’s cross-sectional design precludes knowing whether “smoking behavior is the cause or result of the prefrontal chronnectome alterations in schizophrenia,” the authors added.
The study was supported by the National Natural Science Foundation of China and the Sichuan Science and Technology Program. The researchers had no conflicts of interest.
SOURCE: Fan YS et al. Prog Neuropsychopharmacol Biol Psychiatry. 2020 Apr 20. doi: 10.1016/j.pnpbp.2020.109860.
FROM PROGRESS IN NEURO-PSYCHOPHARMACOLOGY & BIOLOGICAL PSYCHIATRY
Early cognitive screening is key for schizophrenia spectrum disorder
, compared with a group of controls, results from a novel study show.
“Based on these findings, we recommend that neurocognitive assessment should be performed as early as possible after illness onset,” researchers led by Lars Helldin, MD, PhD, of the department of psychiatry at NU Health-Care Hospital, Region Västra Götaland, Sweden, wrote in a study published in Schizophrenia Research: Cognition (2020 Jun doi: 10.1016/j.scog.2020.100172). “Early identification of cognitive risk factors for poor real-life functional outcome is necessary in order to alert the clinical and rehabilitation services about patients in need of extra care.”
For the study, 291 men and women suffering from schizophrenia spectrum disorder (SSD) and 302 controls underwent assessment with a series of comprehensive neurocognitive tests, including the Global Assessment of Functioning (GAF), the Positive and Negative Syndrome Scale (PANSS), the Specific Level of Functioning Scale (SLOF), the Rey Auditory Verbal Learning Test (RAVLT), and the Wisconsin Card Sorting Test (WCST). The researchers found that the neurocognitive function of the SSD patients was significantly lower than that of the healthy controls on all assessments, with very large effect sizes. “There was considerable diversity within each group, as subgroups of patients scored higher than the control mean and subgroups of controls scored lower than the patient mean, particularly on tests of working memory, verbal learning and memory, and executive function,” wrote Dr. Helldin and associates.
As for the WSCT score, the cognitively intact group had a significantly lower PANSS negative symptom level (P less than .01), a lower PANSS general pathology level (P less than .05), and a lower PANSS total symptom level (P less than .01). As for the WAIS Vocabulary score, the patient group with a higher score than the controls had a significantly lower PANSS negative symptom level (P less than .05).
“Here, we have linked neurocognitive heterogeneity to functional outcome differences, and suggest that personalized treatment with emphasis on practical daily skills may be of great significance especially for those with large baseline cognitive deficits,” the researchers concluded. “Such efforts are imperative not only in order to reduce personal suffering and increase quality of life for the patients, but also to reduce the enormous society level economic costs of functional deficits.”
The study was funded by the Regional Health Authority, VG Region, Sweden. The authors reported having no financial disclosures.
, compared with a group of controls, results from a novel study show.
“Based on these findings, we recommend that neurocognitive assessment should be performed as early as possible after illness onset,” researchers led by Lars Helldin, MD, PhD, of the department of psychiatry at NU Health-Care Hospital, Region Västra Götaland, Sweden, wrote in a study published in Schizophrenia Research: Cognition (2020 Jun doi: 10.1016/j.scog.2020.100172). “Early identification of cognitive risk factors for poor real-life functional outcome is necessary in order to alert the clinical and rehabilitation services about patients in need of extra care.”
For the study, 291 men and women suffering from schizophrenia spectrum disorder (SSD) and 302 controls underwent assessment with a series of comprehensive neurocognitive tests, including the Global Assessment of Functioning (GAF), the Positive and Negative Syndrome Scale (PANSS), the Specific Level of Functioning Scale (SLOF), the Rey Auditory Verbal Learning Test (RAVLT), and the Wisconsin Card Sorting Test (WCST). The researchers found that the neurocognitive function of the SSD patients was significantly lower than that of the healthy controls on all assessments, with very large effect sizes. “There was considerable diversity within each group, as subgroups of patients scored higher than the control mean and subgroups of controls scored lower than the patient mean, particularly on tests of working memory, verbal learning and memory, and executive function,” wrote Dr. Helldin and associates.
As for the WSCT score, the cognitively intact group had a significantly lower PANSS negative symptom level (P less than .01), a lower PANSS general pathology level (P less than .05), and a lower PANSS total symptom level (P less than .01). As for the WAIS Vocabulary score, the patient group with a higher score than the controls had a significantly lower PANSS negative symptom level (P less than .05).
“Here, we have linked neurocognitive heterogeneity to functional outcome differences, and suggest that personalized treatment with emphasis on practical daily skills may be of great significance especially for those with large baseline cognitive deficits,” the researchers concluded. “Such efforts are imperative not only in order to reduce personal suffering and increase quality of life for the patients, but also to reduce the enormous society level economic costs of functional deficits.”
The study was funded by the Regional Health Authority, VG Region, Sweden. The authors reported having no financial disclosures.
, compared with a group of controls, results from a novel study show.
“Based on these findings, we recommend that neurocognitive assessment should be performed as early as possible after illness onset,” researchers led by Lars Helldin, MD, PhD, of the department of psychiatry at NU Health-Care Hospital, Region Västra Götaland, Sweden, wrote in a study published in Schizophrenia Research: Cognition (2020 Jun doi: 10.1016/j.scog.2020.100172). “Early identification of cognitive risk factors for poor real-life functional outcome is necessary in order to alert the clinical and rehabilitation services about patients in need of extra care.”
For the study, 291 men and women suffering from schizophrenia spectrum disorder (SSD) and 302 controls underwent assessment with a series of comprehensive neurocognitive tests, including the Global Assessment of Functioning (GAF), the Positive and Negative Syndrome Scale (PANSS), the Specific Level of Functioning Scale (SLOF), the Rey Auditory Verbal Learning Test (RAVLT), and the Wisconsin Card Sorting Test (WCST). The researchers found that the neurocognitive function of the SSD patients was significantly lower than that of the healthy controls on all assessments, with very large effect sizes. “There was considerable diversity within each group, as subgroups of patients scored higher than the control mean and subgroups of controls scored lower than the patient mean, particularly on tests of working memory, verbal learning and memory, and executive function,” wrote Dr. Helldin and associates.
As for the WSCT score, the cognitively intact group had a significantly lower PANSS negative symptom level (P less than .01), a lower PANSS general pathology level (P less than .05), and a lower PANSS total symptom level (P less than .01). As for the WAIS Vocabulary score, the patient group with a higher score than the controls had a significantly lower PANSS negative symptom level (P less than .05).
“Here, we have linked neurocognitive heterogeneity to functional outcome differences, and suggest that personalized treatment with emphasis on practical daily skills may be of great significance especially for those with large baseline cognitive deficits,” the researchers concluded. “Such efforts are imperative not only in order to reduce personal suffering and increase quality of life for the patients, but also to reduce the enormous society level economic costs of functional deficits.”
The study was funded by the Regional Health Authority, VG Region, Sweden. The authors reported having no financial disclosures.
FROM SCHIZOPHRENIA RESEARCH: COGNITION
Cardiovascular disease risk higher in patients with schizophrenia, metabolic syndrome
Metabolic syndrome is common among patients with schizophrenia, and those with metabolic syndrome are at significantly higher risk for cardiovascular disease, according to Shadi Naderyan Fe’li of the department of biostatistics and epidemiology at Shahid Sadoughi University of Medical Sciences in Yazd, Iran, and associates.
The cross-sectional study, performed on 100 patients with schizophrenia (83 men, 17 women), was published in the Medical Journal of the Islamic Republic of Iran. (men, 21.7%; women, 52.9%); the most common component of metabolic disorder was low HDL cholesterol in males and abdominal adiposity in females.
Based on Framingham Risk Scores, 76% of study participants had a low risk of cardiovascular disease, 16% had intermediate risk, and 8% had high risk. However, patients were almost twice as likely to have intermediate or high risk of cardiovascular disease if they also had metabolic syndrome (P = .042).
“Considering the findings of this study as well as other recent reports, psychiatrists and health care staff should be informed about the potential metabolic side effects of antipsychotics and unhealthy lifestyles among these patients. Furthermore, regular monitoring of metabolic risk factors is suggested. In addition, medical and behavioral interventions should be conducted for patients with [metabolic syndrome],” the investigators concluded.
The investigators reported that they had no conflicts of interest.
SOURCE: Fe’li SN et al. Med J Islam Repub Iran. 2019 Sep 16. doi: 10.34171/mjiri.33.97.
Metabolic syndrome is common among patients with schizophrenia, and those with metabolic syndrome are at significantly higher risk for cardiovascular disease, according to Shadi Naderyan Fe’li of the department of biostatistics and epidemiology at Shahid Sadoughi University of Medical Sciences in Yazd, Iran, and associates.
The cross-sectional study, performed on 100 patients with schizophrenia (83 men, 17 women), was published in the Medical Journal of the Islamic Republic of Iran. (men, 21.7%; women, 52.9%); the most common component of metabolic disorder was low HDL cholesterol in males and abdominal adiposity in females.
Based on Framingham Risk Scores, 76% of study participants had a low risk of cardiovascular disease, 16% had intermediate risk, and 8% had high risk. However, patients were almost twice as likely to have intermediate or high risk of cardiovascular disease if they also had metabolic syndrome (P = .042).
“Considering the findings of this study as well as other recent reports, psychiatrists and health care staff should be informed about the potential metabolic side effects of antipsychotics and unhealthy lifestyles among these patients. Furthermore, regular monitoring of metabolic risk factors is suggested. In addition, medical and behavioral interventions should be conducted for patients with [metabolic syndrome],” the investigators concluded.
The investigators reported that they had no conflicts of interest.
SOURCE: Fe’li SN et al. Med J Islam Repub Iran. 2019 Sep 16. doi: 10.34171/mjiri.33.97.
Metabolic syndrome is common among patients with schizophrenia, and those with metabolic syndrome are at significantly higher risk for cardiovascular disease, according to Shadi Naderyan Fe’li of the department of biostatistics and epidemiology at Shahid Sadoughi University of Medical Sciences in Yazd, Iran, and associates.
The cross-sectional study, performed on 100 patients with schizophrenia (83 men, 17 women), was published in the Medical Journal of the Islamic Republic of Iran. (men, 21.7%; women, 52.9%); the most common component of metabolic disorder was low HDL cholesterol in males and abdominal adiposity in females.
Based on Framingham Risk Scores, 76% of study participants had a low risk of cardiovascular disease, 16% had intermediate risk, and 8% had high risk. However, patients were almost twice as likely to have intermediate or high risk of cardiovascular disease if they also had metabolic syndrome (P = .042).
“Considering the findings of this study as well as other recent reports, psychiatrists and health care staff should be informed about the potential metabolic side effects of antipsychotics and unhealthy lifestyles among these patients. Furthermore, regular monitoring of metabolic risk factors is suggested. In addition, medical and behavioral interventions should be conducted for patients with [metabolic syndrome],” the investigators concluded.
The investigators reported that they had no conflicts of interest.
SOURCE: Fe’li SN et al. Med J Islam Repub Iran. 2019 Sep 16. doi: 10.34171/mjiri.33.97.
FROM THE MEDICAL JOURNAL OF THE ISLAMIC REPUBLIC OF IRAN
Top research findings of 2018-2019 for clinical practice
In Part 1 of this article, published in
1. Han LKM, Aghajani M, Clark SL, et al. Epigenetic aging in major depressive disorder. Am J Psychiatry. 2018;175(8):774-782.
In light of the association of major depressive disorder (MDD) with an increased risk of aging-related diseases, Han et al2 examined whether MDD was associated with higher epigenetic aging in blood as measured by DNA methylation patterns. They also studied whether clinical characteristics of MDD had a further impact on these patterns, and whether the findings replicated in brain tissue. Many differentially methylated regions of our DNA tend to change as we age. Han et al2 used these age-sensitive differentially methylated regions to estimate chronological age, using DNA extracted from various tissues, including blood and brain.
Study design
- As a part of the Netherlands Study of Depression and Anxiety (NESDA), this study included 811 patients with MDD and 319 control participants with no lifetime psychiatric disorders and low depressive symptoms (Inventory of Depressive Symptomatology score <14).
- Diagnosis of MDD and clinical characteristics were assessed by questionnaires and psychiatric interviews. Childhood trauma was assessed using the NEMESIS childhood trauma interview, which included a structured inventory of trauma exposure during childhood.
- DNA methylation age was estimated using all methylation sites in the blood of 811 patients with MDD and 319 control participants. The residuals of the DNA methylation age estimates regressed on chronological age were calculated to indicate epigenetic aging.
- Analyses were adjusted for sociodemographic characteristics, lifestyle, and health status.
- Postmortem brain samples of 74 patients with MDD and 64 control participants were used for replication.
Outcomes
- Significantly higher epigenetic aging was observed in patients with MDD compared with control participants (Cohen’s d = 0.18), which suggests that patients with MDD are biologically older than their corresponding chronological age. There was a significant dose effect with increasing symptom severity in the overall sample.
- In the MDD group, epigenetic aging was positively and significantly associated with childhood trauma.
- The case-control difference was replicated in an independent analysis of postmortem brain samples.
Conclusion
- These findings suggest that patients with MDD and people with a history of childhood trauma may biologically age relatively faster than those without MDD or childhood trauma. These findings may represent a biomarker of aging and might help identify patients who may benefit from early and intensive interventions to reduce the physical comorbidities of MDD.
- This study raises the possibility that MDD may be causally related to epigenetic age acceleration. However, it only points out the associations; there are other possible explanations for this correlation, including the possibility that a shared risk factor accounts for the observed association.
2. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526-535.
Delirium is common and often goes underdiagnosed. It is particularly prevalent among hospitalized geriatric patients. Several medications have been suggested to have a role in treating or preventing delirium. However, it remains uncertain which medications provide the best response rate, the lowest rate of delirium occurrence, and the best tolerability. In an attempt to find answers to these questions, Wu et al3 reviewed studies that evaluated the use of various medications used for delirium.
Study design
- Researchers conducted a systematic review and network meta-analysis of randomized controlled trials (RCTs) that investigated various pharmacologic agents used to treat or prevent delirium.
- Fifty-eight RCTs were included in the analyses. Of these, 20 RCTs with a total of 1,435 participants compared the outcomes of treatments of delirium, and 38 RCTs with a total of 8,168 participants examined prevention.
- A network meta-analysis was performed to determine if an agent or combinations of agents were superior to placebo or widely used medications.
Continue to: Outcomes
Outcomes
- Haloperidol plus lorazepam provided the best response rate for treating delirium compared with placebo/control.
- For delirium prevention, patients who received ramelteon, olanzapine, risperidone, or dexmedetomidine had significantly lower delirium occurrence rates than those receiving placebo/control.
- None of the pharmacologic treatments were significantly associated with a higher risk of all-cause mortality compared with placebo/control.
Conclusion
- Haloperidol plus lorazepam might be the best treatment and ramelteon the best preventive medicine for delirium. None of the pharmacologic interventions for treatment or prophylaxis increased all-cause mortality.
- However, network meta-analyses involve extrapolating treatment comparisons that are not made directly. As Blazer8 pointed out, both findings in this study (that haloperidol plus lorazepam is a unique intervention among the treatment trials and ramelteon is a unique intervention for prevention) seemed to be driven by 2 of the 58 studies that Wu et al3 examined.Wu et al3 also cautioned that both of these interventions needed to be further researched for efficacy.
3. Simpson TL, Saxon AJ, Stappenbeck C, et al. Double-blind randomized clinical trial of prazosin for alcohol use disorder. Am J Psychiatry. 2018;175(12):1216-1224.
While some evidence suggests that elevated brain noradrenergic activity is involved in the initiation and maintenance of alcohol use disorder,9 current medications used to treat alcohol use disorder do not target brain noradrenergic pathways. In an RCT, Simpson et al4 tested prazosin, an alpha-1 adrenergic receptor antagonist, for the treatment of alcohol use disorder.
Study design
- In this 12-week double-blind study, 92 participants with alcohol use disorder were randomly assigned to receive prazosin or placebo. Individuals with posttraumatic stress disorder were excluded.
- Prazosin was titrated to a target dosing schedule of 4 mg in the morning, 4 mg in the afternoon, and 8 mg at bedtime by the end of Week 2. The behavioral platform was medical management. Participants provided daily data on their alcohol consumption.
- Generalized linear mixed-effects models were used to examine the impact of prazosin compared with placebo on number of drinks per week, number of drinking days per week, and number of heavy drinking days per week.
Outcomes
- Among the 80 participants who completed the titration period and were included in the primary analyses, prazosin was associated with self-reported fewer heavy drinking days, and fewer drinks per week (Palatino LT Std−8 vs Palatino LT Std−1.5 with placebo). Drinking days per week and craving showed no group differences.
- The rate of drinking and the probability of heavy drinking showed a greater decrease over time for participants receiving prazosin compared with those receiving placebo.
Continue to: Conclusion
Conclusion
- These findings of moderate reductions in heavy drinking days and drinks per week with prazosin suggest that prazosin may be a promising harm-reduction treatment for alcohol use disorder.
4. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
Postpartum depression is among the most common complications of childbirth. It can result in considerable suffering for mothers, children, and families. Gamma-aminobutyric acid (GABA) signaling has previously been reported to be involved in the pathophysiology of postpartum depression. Meltzer-Brody et al5 conducted 2 double-blind, randomized, placebo-controlled, phase 3 trials comparing brexanolone with placebo in women with postpartum depression at 30 clinical research centers and specialized psychiatric units in the United States.
Study design
- Participants were women age 18 to 45, Palatino LT Std≤6 months postpartum at screening, with postpartum depression as indicated by a qualifying 17-item Hamilton Depression Rating Scale (HAM-D) score of ≥26 for Study 1 or 20 to 25 for Study 2.
- Of the 375 women who were screened simultaneously across both studies, 138 were randomly assigned (1:1:1) to receive a single IV injection of brexanolone, 90 μg/kg per hour (BRX90) (n = 45), brexanolone, 60 μg/kg per hour (BRX60) (n = 47), or placebo (n = 46) for 60 hours in Study 1, and 108 were randomly assigned (1:1) to receive BRX90 (n = 54) or placebo (n = 54) for 60 hours in Study 2.
- The primary efficacy endpoint was change in total score on the HAM-D from baseline to 60 hours. Patients were followed until Day 30.
Outcomes
- In Study 1, at 60 hours, the least-squares (LS) mean reduction in HAM-D total score from baseline was 19.5 points (standard error [SE] 1.2) in the BRX60 group and 17.7 points (SE 1.2) in the BRX90 group, compared with 14.0 points (SE 1.1) in the placebo group.
- In Study 2, at 60 hours, the LS mean reduction in HAM-D total score from baseline was 14.6 points (SE 0.8) in the BRX90 group compared with 12.1 points (SE 0.8) for the placebo group.
- In Study 1, one patient in the BRX60 group had 2 serious adverse events (suicidal ideation and intentional overdose attempt during follow-up). In Study 2, one patient in the BRX90 group had 2 serious adverse events (altered state of consciousness and syncope), which were considered treatment-related.
Conclusion
- Administration of brexanolone injection for postpartum depression resulted in significant, clinically meaningful reductions in HAM-D total score at 60 hours compared with placebo, with a rapid onset of action and durable treatment response during the study period. These results suggest that brexanolone injection has the potential to improve treatment options for women with this disorder.
Continue to: #5
5. Tiihonen J, Taipale H, Mehtälä J, et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry. 2019;76(5):499-507.
In clinical practice, the use of multiple antipsychotic agents for the maintenance treatment of schizophrenia is common but generally not recommended. The effectiveness of antipsychotic polypharmacy in preventing relapse of schizophrenia has not been established, and whether specific antipsychotic combinations are superior to monotherapies for maintenance treatment of schizophrenia is unknown. Tiihonen et al6 investigated the association of specific antipsychotic combinations with psychiatric rehospitalization, which was used as a marker for relapse.
Study design
- This study included 62,250 patients with schizophrenia, treated between January 1, 1996 and December 31, 2015, in a comprehensive, nationwide cohort in Finland. Overall, 31,257 individuals (50.2%) were men, and the median age was 45.6 (interquartile range, 34.6 to 57.9).
- Patients were receiving 29 different antipsychotic monotherapy or polypharmacy regimens.
- Researchers analyzed data from April 24 to June 15, 2018 using psychiatric rehospitalization as a marker for relapse. To minimize selection bias, rehospitalization risks were investigated using within-individual analyses.
- The main outcome was the hazard ratio (HR) for psychiatric rehospitalization during use of polypharmacy vs monotherapy by the same patient.
Outcomes
- Clozapine plus aripiprazole was associated with the lowest risk of psychiatric rehospitalization, with a difference of 14% (HR, .86; CI, .79 to .94) compared with clozapine monotherapy in the analysis that included all polypharmacy periods, and 18% (HR, .82; CI, .75 to .89) in the conservatively defined polypharmacy analysis that excluded periods <90 days.
- Among patients experiencing their first episode of schizophrenia, the differences between clozapine plus aripiprazole vs clozapine monotherapy were greater, with a difference of 22% in the analysis that included all polypharmacy periods, and 23% in the conservatively defined polypharmacy analysis.
- At the aggregate level, any antipsychotic polypharmacy was associated with a 7% to 13% lower risk of psychiatric rehospitalization compared with any monotherapy.
- Clozapine was the only monotherapy among the 10 best treatments.
- Results on all-cause and somatic hospitalization, mortality, and other sensitivity analyses were in line with the primary outcomes.
Conclusion
- This study suggests that certain types of antipsychotic polypharmacy may reduce the risk of rehospitalization in patients with schizophrenia. Current treatment guidelines state that clinicians should prefer antipsychotic monotherapy and avoid polypharmacy. Tiihonen et al6 raise the question whether current treatment guidelines should continue to discourage antipsychotic polypharmacy in the maintenance treatment of schizophrenia.
- Despite the large administrative databases and sophisticated statistical methods used in this study, this approach has important limitations. As Goff10 points out, despite efforts to minimize bias, these results should be considered preliminary until confirmed by RCTs.
6. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of adjunctive psychotropic medications in patients with schizophrenia. JAMA Psychiatry. 2019;76(5):508-515.
In routine clinical practice, patients with schizophrenia are often treated with combinations of antipsychotics and other psychotropic medications. However, there is little evidence about the comparative effectiveness of these adjunctive treatment strategies. Stroup et al7 investigated the comparative real-world effectiveness of adjunctive psychotropic treatments for patients with schizophrenia.
Continue to: Study design
Study design
- This comparative effectiveness study used US Medicaid data from January 1, 2001, to December 31, 2010. Data analysis was performed from January 1, 2017, to June 30, 2018.
- The study cohort included 81,921 adult outpatients diagnosed with schizophrenia with a mean age of 40.7 (range: 18 to 64), including 37,515 women (45.8%). All patients were stably treated with a single antipsychotic and then started on an adjunctive antidepressant (n = 31,117), benzodiazepine (n = 11,941), mood stabilizer (n = 12,849), or another antipsychotic (n = 26,014).
- Researchers used multinomial logistic regression models to estimate propensity scores to balance covariates across the 4 medication groups. Weighted Cox proportional hazards regression models were used to compare treatment outcomes during 365 days on an intention-to-treat basis.
- The main outcomes and measures included risk of hospitalization for a mental disorder (primary), emergency department (ED) visits for a mental disorder, and all-cause mortality.
Outcomes
- Compared with starting another antipsychotic, initiating use of an antidepressant was associated with a lower risk of psychiatric hospitalization, and initiating use of a benzodiazepine was associated with a higher risk. Initiating use of a mood stabilizer was not significantly different from initiating use of another antipsychotic.
- A similar pattern of associations was observed in psychiatric ED visits for initiating use of an antidepressant, benzodiazepine, or mood stabilizer.
- Initiating use of a mood stabilizer was associated with an increased risk of mortality.
Conclusion
- Compared with the addition of a second antipsychotic, adding an antidepressant was associated with substantially reduced rates of hospitalization, whereas adding a benzodiazepine was associated with a modest increase in the risk of hospitalization. While the addition of a mood stabilizer was not associated with a significant difference in the risk of hospitalization, it was associated with higher mortality.
- Despite the limitations associated with this study, the associations of benzodiazepines and mood stabilizers with poorer outcomes warrant clinical caution and further investigation.
Bottom Line
Significantly higher epigenetic aging has been observed in patients with major depressive disorder. Haloperidol plus lorazepam might be an effective treatment for delirium; and ramelteon may be effective for preventing delirium. Prazosin reduces heavy drinking in patients with alcohol use disorder. A 60-hour infusion of brexanolone can help alleviate postpartum depression. Clozapine plus aripiprazole reduces the risk of rehospitalization among patients with schizophrenia. Adding an antidepressant to an antipsychotic also can reduce the risk of rehospitalization among patients with schizophrenia.
Related Resources
- NEJM Journal Watch. www.jwatch.org.
- F1000 Prime. https://f1000.com/prime/home.
- BMJ Journals Evidence-Based Mental Health. https://ebmh.bmj.com.
Drug Brand Names
Aripiprazole • Abilify
Brexanolone • Zulresso
Clozapine • Clozaril
Dexmedetomidine • Precedex
Haloperidol • Haldol
Lorazepam • Ativan
Olanzapine • Zyprexa
Prazosin • Minipress
Ramelteon • Rozerem
Risperidone • Risperdal
1. Saeed SA, Stanley JB. Top research findings of 2018-2019. First of 2 parts. Current Psychiatry. 2020;19(1):13-18.
2. Han LKM, Aghajani M, Clark SL, et al. Epigenetic aging in major depressive disorder. Am J Psychiatry. 2018;175(8):774-782.
3. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526-535.
4. Simpson TL, Saxon AJ, Stappenbeck C, et al. Double-blind randomized clinical trial of prazosin for alcohol use disorder. Am J Psychiatry. 2018;175(12):1216-1224.
5. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
6. Tiihonen J, Taipale H, Mehtälä J, et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry. 2019;76(5):499-507.
7. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of adjunctive psychotropic medications in patients with schizophrenia. JAMA Psychiatry. 2019;76(5):508-515.
8. Blazer DG. Pharmacologic intervention for the treatment and prevention of delirium: looking beneath the modeling. JAMA Psychiatry. 2019;76(5):472-473.
9. Koob GF. Brain stress systems in the amygdala and addiction. Brain Res. 2009;1293:61-75.
10. Goff DC. Can adjunctive pharmacotherapy reduce hospitalization in schizophrenia? Insights from administrative databases. JAMA Psychiatry. 2019;76(5):468-469.
In Part 1 of this article, published in

1. Han LKM, Aghajani M, Clark SL, et al. Epigenetic aging in major depressive disorder. Am J Psychiatry. 2018;175(8):774-782.
In light of the association of major depressive disorder (MDD) with an increased risk of aging-related diseases, Han et al2 examined whether MDD was associated with higher epigenetic aging in blood as measured by DNA methylation patterns. They also studied whether clinical characteristics of MDD had a further impact on these patterns, and whether the findings replicated in brain tissue. Many differentially methylated regions of our DNA tend to change as we age. Han et al2 used these age-sensitive differentially methylated regions to estimate chronological age, using DNA extracted from various tissues, including blood and brain.
Study design
- As a part of the Netherlands Study of Depression and Anxiety (NESDA), this study included 811 patients with MDD and 319 control participants with no lifetime psychiatric disorders and low depressive symptoms (Inventory of Depressive Symptomatology score <14).
- Diagnosis of MDD and clinical characteristics were assessed by questionnaires and psychiatric interviews. Childhood trauma was assessed using the NEMESIS childhood trauma interview, which included a structured inventory of trauma exposure during childhood.
- DNA methylation age was estimated using all methylation sites in the blood of 811 patients with MDD and 319 control participants. The residuals of the DNA methylation age estimates regressed on chronological age were calculated to indicate epigenetic aging.
- Analyses were adjusted for sociodemographic characteristics, lifestyle, and health status.
- Postmortem brain samples of 74 patients with MDD and 64 control participants were used for replication.
Outcomes
- Significantly higher epigenetic aging was observed in patients with MDD compared with control participants (Cohen’s d = 0.18), which suggests that patients with MDD are biologically older than their corresponding chronological age. There was a significant dose effect with increasing symptom severity in the overall sample.
- In the MDD group, epigenetic aging was positively and significantly associated with childhood trauma.
- The case-control difference was replicated in an independent analysis of postmortem brain samples.
Conclusion
- These findings suggest that patients with MDD and people with a history of childhood trauma may biologically age relatively faster than those without MDD or childhood trauma. These findings may represent a biomarker of aging and might help identify patients who may benefit from early and intensive interventions to reduce the physical comorbidities of MDD.
- This study raises the possibility that MDD may be causally related to epigenetic age acceleration. However, it only points out the associations; there are other possible explanations for this correlation, including the possibility that a shared risk factor accounts for the observed association.
2. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526-535.
Delirium is common and often goes underdiagnosed. It is particularly prevalent among hospitalized geriatric patients. Several medications have been suggested to have a role in treating or preventing delirium. However, it remains uncertain which medications provide the best response rate, the lowest rate of delirium occurrence, and the best tolerability. In an attempt to find answers to these questions, Wu et al3 reviewed studies that evaluated the use of various medications used for delirium.
Study design
- Researchers conducted a systematic review and network meta-analysis of randomized controlled trials (RCTs) that investigated various pharmacologic agents used to treat or prevent delirium.
- Fifty-eight RCTs were included in the analyses. Of these, 20 RCTs with a total of 1,435 participants compared the outcomes of treatments of delirium, and 38 RCTs with a total of 8,168 participants examined prevention.
- A network meta-analysis was performed to determine if an agent or combinations of agents were superior to placebo or widely used medications.
Continue to: Outcomes
Outcomes
- Haloperidol plus lorazepam provided the best response rate for treating delirium compared with placebo/control.
- For delirium prevention, patients who received ramelteon, olanzapine, risperidone, or dexmedetomidine had significantly lower delirium occurrence rates than those receiving placebo/control.
- None of the pharmacologic treatments were significantly associated with a higher risk of all-cause mortality compared with placebo/control.
Conclusion
- Haloperidol plus lorazepam might be the best treatment and ramelteon the best preventive medicine for delirium. None of the pharmacologic interventions for treatment or prophylaxis increased all-cause mortality.
- However, network meta-analyses involve extrapolating treatment comparisons that are not made directly. As Blazer8 pointed out, both findings in this study (that haloperidol plus lorazepam is a unique intervention among the treatment trials and ramelteon is a unique intervention for prevention) seemed to be driven by 2 of the 58 studies that Wu et al3 examined.Wu et al3 also cautioned that both of these interventions needed to be further researched for efficacy.
3. Simpson TL, Saxon AJ, Stappenbeck C, et al. Double-blind randomized clinical trial of prazosin for alcohol use disorder. Am J Psychiatry. 2018;175(12):1216-1224.
While some evidence suggests that elevated brain noradrenergic activity is involved in the initiation and maintenance of alcohol use disorder,9 current medications used to treat alcohol use disorder do not target brain noradrenergic pathways. In an RCT, Simpson et al4 tested prazosin, an alpha-1 adrenergic receptor antagonist, for the treatment of alcohol use disorder.
Study design
- In this 12-week double-blind study, 92 participants with alcohol use disorder were randomly assigned to receive prazosin or placebo. Individuals with posttraumatic stress disorder were excluded.
- Prazosin was titrated to a target dosing schedule of 4 mg in the morning, 4 mg in the afternoon, and 8 mg at bedtime by the end of Week 2. The behavioral platform was medical management. Participants provided daily data on their alcohol consumption.
- Generalized linear mixed-effects models were used to examine the impact of prazosin compared with placebo on number of drinks per week, number of drinking days per week, and number of heavy drinking days per week.
Outcomes
- Among the 80 participants who completed the titration period and were included in the primary analyses, prazosin was associated with self-reported fewer heavy drinking days, and fewer drinks per week (Palatino LT Std−8 vs Palatino LT Std−1.5 with placebo). Drinking days per week and craving showed no group differences.
- The rate of drinking and the probability of heavy drinking showed a greater decrease over time for participants receiving prazosin compared with those receiving placebo.
Continue to: Conclusion
Conclusion
- These findings of moderate reductions in heavy drinking days and drinks per week with prazosin suggest that prazosin may be a promising harm-reduction treatment for alcohol use disorder.
4. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
Postpartum depression is among the most common complications of childbirth. It can result in considerable suffering for mothers, children, and families. Gamma-aminobutyric acid (GABA) signaling has previously been reported to be involved in the pathophysiology of postpartum depression. Meltzer-Brody et al5 conducted 2 double-blind, randomized, placebo-controlled, phase 3 trials comparing brexanolone with placebo in women with postpartum depression at 30 clinical research centers and specialized psychiatric units in the United States.
Study design
- Participants were women age 18 to 45, Palatino LT Std≤6 months postpartum at screening, with postpartum depression as indicated by a qualifying 17-item Hamilton Depression Rating Scale (HAM-D) score of ≥26 for Study 1 or 20 to 25 for Study 2.
- Of the 375 women who were screened simultaneously across both studies, 138 were randomly assigned (1:1:1) to receive a single IV injection of brexanolone, 90 μg/kg per hour (BRX90) (n = 45), brexanolone, 60 μg/kg per hour (BRX60) (n = 47), or placebo (n = 46) for 60 hours in Study 1, and 108 were randomly assigned (1:1) to receive BRX90 (n = 54) or placebo (n = 54) for 60 hours in Study 2.
- The primary efficacy endpoint was change in total score on the HAM-D from baseline to 60 hours. Patients were followed until Day 30.
Outcomes
- In Study 1, at 60 hours, the least-squares (LS) mean reduction in HAM-D total score from baseline was 19.5 points (standard error [SE] 1.2) in the BRX60 group and 17.7 points (SE 1.2) in the BRX90 group, compared with 14.0 points (SE 1.1) in the placebo group.
- In Study 2, at 60 hours, the LS mean reduction in HAM-D total score from baseline was 14.6 points (SE 0.8) in the BRX90 group compared with 12.1 points (SE 0.8) for the placebo group.
- In Study 1, one patient in the BRX60 group had 2 serious adverse events (suicidal ideation and intentional overdose attempt during follow-up). In Study 2, one patient in the BRX90 group had 2 serious adverse events (altered state of consciousness and syncope), which were considered treatment-related.
Conclusion
- Administration of brexanolone injection for postpartum depression resulted in significant, clinically meaningful reductions in HAM-D total score at 60 hours compared with placebo, with a rapid onset of action and durable treatment response during the study period. These results suggest that brexanolone injection has the potential to improve treatment options for women with this disorder.
Continue to: #5
5. Tiihonen J, Taipale H, Mehtälä J, et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry. 2019;76(5):499-507.
In clinical practice, the use of multiple antipsychotic agents for the maintenance treatment of schizophrenia is common but generally not recommended. The effectiveness of antipsychotic polypharmacy in preventing relapse of schizophrenia has not been established, and whether specific antipsychotic combinations are superior to monotherapies for maintenance treatment of schizophrenia is unknown. Tiihonen et al6 investigated the association of specific antipsychotic combinations with psychiatric rehospitalization, which was used as a marker for relapse.
Study design
- This study included 62,250 patients with schizophrenia, treated between January 1, 1996 and December 31, 2015, in a comprehensive, nationwide cohort in Finland. Overall, 31,257 individuals (50.2%) were men, and the median age was 45.6 (interquartile range, 34.6 to 57.9).
- Patients were receiving 29 different antipsychotic monotherapy or polypharmacy regimens.
- Researchers analyzed data from April 24 to June 15, 2018 using psychiatric rehospitalization as a marker for relapse. To minimize selection bias, rehospitalization risks were investigated using within-individual analyses.
- The main outcome was the hazard ratio (HR) for psychiatric rehospitalization during use of polypharmacy vs monotherapy by the same patient.
Outcomes
- Clozapine plus aripiprazole was associated with the lowest risk of psychiatric rehospitalization, with a difference of 14% (HR, .86; CI, .79 to .94) compared with clozapine monotherapy in the analysis that included all polypharmacy periods, and 18% (HR, .82; CI, .75 to .89) in the conservatively defined polypharmacy analysis that excluded periods <90 days.
- Among patients experiencing their first episode of schizophrenia, the differences between clozapine plus aripiprazole vs clozapine monotherapy were greater, with a difference of 22% in the analysis that included all polypharmacy periods, and 23% in the conservatively defined polypharmacy analysis.
- At the aggregate level, any antipsychotic polypharmacy was associated with a 7% to 13% lower risk of psychiatric rehospitalization compared with any monotherapy.
- Clozapine was the only monotherapy among the 10 best treatments.
- Results on all-cause and somatic hospitalization, mortality, and other sensitivity analyses were in line with the primary outcomes.
Conclusion
- This study suggests that certain types of antipsychotic polypharmacy may reduce the risk of rehospitalization in patients with schizophrenia. Current treatment guidelines state that clinicians should prefer antipsychotic monotherapy and avoid polypharmacy. Tiihonen et al6 raise the question whether current treatment guidelines should continue to discourage antipsychotic polypharmacy in the maintenance treatment of schizophrenia.
- Despite the large administrative databases and sophisticated statistical methods used in this study, this approach has important limitations. As Goff10 points out, despite efforts to minimize bias, these results should be considered preliminary until confirmed by RCTs.
6. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of adjunctive psychotropic medications in patients with schizophrenia. JAMA Psychiatry. 2019;76(5):508-515.
In routine clinical practice, patients with schizophrenia are often treated with combinations of antipsychotics and other psychotropic medications. However, there is little evidence about the comparative effectiveness of these adjunctive treatment strategies. Stroup et al7 investigated the comparative real-world effectiveness of adjunctive psychotropic treatments for patients with schizophrenia.
Continue to: Study design
Study design
- This comparative effectiveness study used US Medicaid data from January 1, 2001, to December 31, 2010. Data analysis was performed from January 1, 2017, to June 30, 2018.
- The study cohort included 81,921 adult outpatients diagnosed with schizophrenia with a mean age of 40.7 (range: 18 to 64), including 37,515 women (45.8%). All patients were stably treated with a single antipsychotic and then started on an adjunctive antidepressant (n = 31,117), benzodiazepine (n = 11,941), mood stabilizer (n = 12,849), or another antipsychotic (n = 26,014).
- Researchers used multinomial logistic regression models to estimate propensity scores to balance covariates across the 4 medication groups. Weighted Cox proportional hazards regression models were used to compare treatment outcomes during 365 days on an intention-to-treat basis.
- The main outcomes and measures included risk of hospitalization for a mental disorder (primary), emergency department (ED) visits for a mental disorder, and all-cause mortality.
Outcomes
- Compared with starting another antipsychotic, initiating use of an antidepressant was associated with a lower risk of psychiatric hospitalization, and initiating use of a benzodiazepine was associated with a higher risk. Initiating use of a mood stabilizer was not significantly different from initiating use of another antipsychotic.
- A similar pattern of associations was observed in psychiatric ED visits for initiating use of an antidepressant, benzodiazepine, or mood stabilizer.
- Initiating use of a mood stabilizer was associated with an increased risk of mortality.
Conclusion
- Compared with the addition of a second antipsychotic, adding an antidepressant was associated with substantially reduced rates of hospitalization, whereas adding a benzodiazepine was associated with a modest increase in the risk of hospitalization. While the addition of a mood stabilizer was not associated with a significant difference in the risk of hospitalization, it was associated with higher mortality.
- Despite the limitations associated with this study, the associations of benzodiazepines and mood stabilizers with poorer outcomes warrant clinical caution and further investigation.
Bottom Line
Significantly higher epigenetic aging has been observed in patients with major depressive disorder. Haloperidol plus lorazepam might be an effective treatment for delirium; and ramelteon may be effective for preventing delirium. Prazosin reduces heavy drinking in patients with alcohol use disorder. A 60-hour infusion of brexanolone can help alleviate postpartum depression. Clozapine plus aripiprazole reduces the risk of rehospitalization among patients with schizophrenia. Adding an antidepressant to an antipsychotic also can reduce the risk of rehospitalization among patients with schizophrenia.
Related Resources
- NEJM Journal Watch. www.jwatch.org.
- F1000 Prime. https://f1000.com/prime/home.
- BMJ Journals Evidence-Based Mental Health. https://ebmh.bmj.com.
Drug Brand Names
Aripiprazole • Abilify
Brexanolone • Zulresso
Clozapine • Clozaril
Dexmedetomidine • Precedex
Haloperidol • Haldol
Lorazepam • Ativan
Olanzapine • Zyprexa
Prazosin • Minipress
Ramelteon • Rozerem
Risperidone • Risperdal
In Part 1 of this article, published in

1. Han LKM, Aghajani M, Clark SL, et al. Epigenetic aging in major depressive disorder. Am J Psychiatry. 2018;175(8):774-782.
In light of the association of major depressive disorder (MDD) with an increased risk of aging-related diseases, Han et al2 examined whether MDD was associated with higher epigenetic aging in blood as measured by DNA methylation patterns. They also studied whether clinical characteristics of MDD had a further impact on these patterns, and whether the findings replicated in brain tissue. Many differentially methylated regions of our DNA tend to change as we age. Han et al2 used these age-sensitive differentially methylated regions to estimate chronological age, using DNA extracted from various tissues, including blood and brain.
Study design
- As a part of the Netherlands Study of Depression and Anxiety (NESDA), this study included 811 patients with MDD and 319 control participants with no lifetime psychiatric disorders and low depressive symptoms (Inventory of Depressive Symptomatology score <14).
- Diagnosis of MDD and clinical characteristics were assessed by questionnaires and psychiatric interviews. Childhood trauma was assessed using the NEMESIS childhood trauma interview, which included a structured inventory of trauma exposure during childhood.
- DNA methylation age was estimated using all methylation sites in the blood of 811 patients with MDD and 319 control participants. The residuals of the DNA methylation age estimates regressed on chronological age were calculated to indicate epigenetic aging.
- Analyses were adjusted for sociodemographic characteristics, lifestyle, and health status.
- Postmortem brain samples of 74 patients with MDD and 64 control participants were used for replication.
Outcomes
- Significantly higher epigenetic aging was observed in patients with MDD compared with control participants (Cohen’s d = 0.18), which suggests that patients with MDD are biologically older than their corresponding chronological age. There was a significant dose effect with increasing symptom severity in the overall sample.
- In the MDD group, epigenetic aging was positively and significantly associated with childhood trauma.
- The case-control difference was replicated in an independent analysis of postmortem brain samples.
Conclusion
- These findings suggest that patients with MDD and people with a history of childhood trauma may biologically age relatively faster than those without MDD or childhood trauma. These findings may represent a biomarker of aging and might help identify patients who may benefit from early and intensive interventions to reduce the physical comorbidities of MDD.
- This study raises the possibility that MDD may be causally related to epigenetic age acceleration. However, it only points out the associations; there are other possible explanations for this correlation, including the possibility that a shared risk factor accounts for the observed association.
2. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526-535.
Delirium is common and often goes underdiagnosed. It is particularly prevalent among hospitalized geriatric patients. Several medications have been suggested to have a role in treating or preventing delirium. However, it remains uncertain which medications provide the best response rate, the lowest rate of delirium occurrence, and the best tolerability. In an attempt to find answers to these questions, Wu et al3 reviewed studies that evaluated the use of various medications used for delirium.
Study design
- Researchers conducted a systematic review and network meta-analysis of randomized controlled trials (RCTs) that investigated various pharmacologic agents used to treat or prevent delirium.
- Fifty-eight RCTs were included in the analyses. Of these, 20 RCTs with a total of 1,435 participants compared the outcomes of treatments of delirium, and 38 RCTs with a total of 8,168 participants examined prevention.
- A network meta-analysis was performed to determine if an agent or combinations of agents were superior to placebo or widely used medications.
Continue to: Outcomes
Outcomes
- Haloperidol plus lorazepam provided the best response rate for treating delirium compared with placebo/control.
- For delirium prevention, patients who received ramelteon, olanzapine, risperidone, or dexmedetomidine had significantly lower delirium occurrence rates than those receiving placebo/control.
- None of the pharmacologic treatments were significantly associated with a higher risk of all-cause mortality compared with placebo/control.
Conclusion
- Haloperidol plus lorazepam might be the best treatment and ramelteon the best preventive medicine for delirium. None of the pharmacologic interventions for treatment or prophylaxis increased all-cause mortality.
- However, network meta-analyses involve extrapolating treatment comparisons that are not made directly. As Blazer8 pointed out, both findings in this study (that haloperidol plus lorazepam is a unique intervention among the treatment trials and ramelteon is a unique intervention for prevention) seemed to be driven by 2 of the 58 studies that Wu et al3 examined.Wu et al3 also cautioned that both of these interventions needed to be further researched for efficacy.
3. Simpson TL, Saxon AJ, Stappenbeck C, et al. Double-blind randomized clinical trial of prazosin for alcohol use disorder. Am J Psychiatry. 2018;175(12):1216-1224.
While some evidence suggests that elevated brain noradrenergic activity is involved in the initiation and maintenance of alcohol use disorder,9 current medications used to treat alcohol use disorder do not target brain noradrenergic pathways. In an RCT, Simpson et al4 tested prazosin, an alpha-1 adrenergic receptor antagonist, for the treatment of alcohol use disorder.
Study design
- In this 12-week double-blind study, 92 participants with alcohol use disorder were randomly assigned to receive prazosin or placebo. Individuals with posttraumatic stress disorder were excluded.
- Prazosin was titrated to a target dosing schedule of 4 mg in the morning, 4 mg in the afternoon, and 8 mg at bedtime by the end of Week 2. The behavioral platform was medical management. Participants provided daily data on their alcohol consumption.
- Generalized linear mixed-effects models were used to examine the impact of prazosin compared with placebo on number of drinks per week, number of drinking days per week, and number of heavy drinking days per week.
Outcomes
- Among the 80 participants who completed the titration period and were included in the primary analyses, prazosin was associated with self-reported fewer heavy drinking days, and fewer drinks per week (Palatino LT Std−8 vs Palatino LT Std−1.5 with placebo). Drinking days per week and craving showed no group differences.
- The rate of drinking and the probability of heavy drinking showed a greater decrease over time for participants receiving prazosin compared with those receiving placebo.
Continue to: Conclusion
Conclusion
- These findings of moderate reductions in heavy drinking days and drinks per week with prazosin suggest that prazosin may be a promising harm-reduction treatment for alcohol use disorder.
4. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
Postpartum depression is among the most common complications of childbirth. It can result in considerable suffering for mothers, children, and families. Gamma-aminobutyric acid (GABA) signaling has previously been reported to be involved in the pathophysiology of postpartum depression. Meltzer-Brody et al5 conducted 2 double-blind, randomized, placebo-controlled, phase 3 trials comparing brexanolone with placebo in women with postpartum depression at 30 clinical research centers and specialized psychiatric units in the United States.
Study design
- Participants were women age 18 to 45, Palatino LT Std≤6 months postpartum at screening, with postpartum depression as indicated by a qualifying 17-item Hamilton Depression Rating Scale (HAM-D) score of ≥26 for Study 1 or 20 to 25 for Study 2.
- Of the 375 women who were screened simultaneously across both studies, 138 were randomly assigned (1:1:1) to receive a single IV injection of brexanolone, 90 μg/kg per hour (BRX90) (n = 45), brexanolone, 60 μg/kg per hour (BRX60) (n = 47), or placebo (n = 46) for 60 hours in Study 1, and 108 were randomly assigned (1:1) to receive BRX90 (n = 54) or placebo (n = 54) for 60 hours in Study 2.
- The primary efficacy endpoint was change in total score on the HAM-D from baseline to 60 hours. Patients were followed until Day 30.
Outcomes
- In Study 1, at 60 hours, the least-squares (LS) mean reduction in HAM-D total score from baseline was 19.5 points (standard error [SE] 1.2) in the BRX60 group and 17.7 points (SE 1.2) in the BRX90 group, compared with 14.0 points (SE 1.1) in the placebo group.
- In Study 2, at 60 hours, the LS mean reduction in HAM-D total score from baseline was 14.6 points (SE 0.8) in the BRX90 group compared with 12.1 points (SE 0.8) for the placebo group.
- In Study 1, one patient in the BRX60 group had 2 serious adverse events (suicidal ideation and intentional overdose attempt during follow-up). In Study 2, one patient in the BRX90 group had 2 serious adverse events (altered state of consciousness and syncope), which were considered treatment-related.
Conclusion
- Administration of brexanolone injection for postpartum depression resulted in significant, clinically meaningful reductions in HAM-D total score at 60 hours compared with placebo, with a rapid onset of action and durable treatment response during the study period. These results suggest that brexanolone injection has the potential to improve treatment options for women with this disorder.
Continue to: #5
5. Tiihonen J, Taipale H, Mehtälä J, et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry. 2019;76(5):499-507.
In clinical practice, the use of multiple antipsychotic agents for the maintenance treatment of schizophrenia is common but generally not recommended. The effectiveness of antipsychotic polypharmacy in preventing relapse of schizophrenia has not been established, and whether specific antipsychotic combinations are superior to monotherapies for maintenance treatment of schizophrenia is unknown. Tiihonen et al6 investigated the association of specific antipsychotic combinations with psychiatric rehospitalization, which was used as a marker for relapse.
Study design
- This study included 62,250 patients with schizophrenia, treated between January 1, 1996 and December 31, 2015, in a comprehensive, nationwide cohort in Finland. Overall, 31,257 individuals (50.2%) were men, and the median age was 45.6 (interquartile range, 34.6 to 57.9).
- Patients were receiving 29 different antipsychotic monotherapy or polypharmacy regimens.
- Researchers analyzed data from April 24 to June 15, 2018 using psychiatric rehospitalization as a marker for relapse. To minimize selection bias, rehospitalization risks were investigated using within-individual analyses.
- The main outcome was the hazard ratio (HR) for psychiatric rehospitalization during use of polypharmacy vs monotherapy by the same patient.
Outcomes
- Clozapine plus aripiprazole was associated with the lowest risk of psychiatric rehospitalization, with a difference of 14% (HR, .86; CI, .79 to .94) compared with clozapine monotherapy in the analysis that included all polypharmacy periods, and 18% (HR, .82; CI, .75 to .89) in the conservatively defined polypharmacy analysis that excluded periods <90 days.
- Among patients experiencing their first episode of schizophrenia, the differences between clozapine plus aripiprazole vs clozapine monotherapy were greater, with a difference of 22% in the analysis that included all polypharmacy periods, and 23% in the conservatively defined polypharmacy analysis.
- At the aggregate level, any antipsychotic polypharmacy was associated with a 7% to 13% lower risk of psychiatric rehospitalization compared with any monotherapy.
- Clozapine was the only monotherapy among the 10 best treatments.
- Results on all-cause and somatic hospitalization, mortality, and other sensitivity analyses were in line with the primary outcomes.
Conclusion
- This study suggests that certain types of antipsychotic polypharmacy may reduce the risk of rehospitalization in patients with schizophrenia. Current treatment guidelines state that clinicians should prefer antipsychotic monotherapy and avoid polypharmacy. Tiihonen et al6 raise the question whether current treatment guidelines should continue to discourage antipsychotic polypharmacy in the maintenance treatment of schizophrenia.
- Despite the large administrative databases and sophisticated statistical methods used in this study, this approach has important limitations. As Goff10 points out, despite efforts to minimize bias, these results should be considered preliminary until confirmed by RCTs.
6. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of adjunctive psychotropic medications in patients with schizophrenia. JAMA Psychiatry. 2019;76(5):508-515.
In routine clinical practice, patients with schizophrenia are often treated with combinations of antipsychotics and other psychotropic medications. However, there is little evidence about the comparative effectiveness of these adjunctive treatment strategies. Stroup et al7 investigated the comparative real-world effectiveness of adjunctive psychotropic treatments for patients with schizophrenia.
Continue to: Study design
Study design
- This comparative effectiveness study used US Medicaid data from January 1, 2001, to December 31, 2010. Data analysis was performed from January 1, 2017, to June 30, 2018.
- The study cohort included 81,921 adult outpatients diagnosed with schizophrenia with a mean age of 40.7 (range: 18 to 64), including 37,515 women (45.8%). All patients were stably treated with a single antipsychotic and then started on an adjunctive antidepressant (n = 31,117), benzodiazepine (n = 11,941), mood stabilizer (n = 12,849), or another antipsychotic (n = 26,014).
- Researchers used multinomial logistic regression models to estimate propensity scores to balance covariates across the 4 medication groups. Weighted Cox proportional hazards regression models were used to compare treatment outcomes during 365 days on an intention-to-treat basis.
- The main outcomes and measures included risk of hospitalization for a mental disorder (primary), emergency department (ED) visits for a mental disorder, and all-cause mortality.
Outcomes
- Compared with starting another antipsychotic, initiating use of an antidepressant was associated with a lower risk of psychiatric hospitalization, and initiating use of a benzodiazepine was associated with a higher risk. Initiating use of a mood stabilizer was not significantly different from initiating use of another antipsychotic.
- A similar pattern of associations was observed in psychiatric ED visits for initiating use of an antidepressant, benzodiazepine, or mood stabilizer.
- Initiating use of a mood stabilizer was associated with an increased risk of mortality.
Conclusion
- Compared with the addition of a second antipsychotic, adding an antidepressant was associated with substantially reduced rates of hospitalization, whereas adding a benzodiazepine was associated with a modest increase in the risk of hospitalization. While the addition of a mood stabilizer was not associated with a significant difference in the risk of hospitalization, it was associated with higher mortality.
- Despite the limitations associated with this study, the associations of benzodiazepines and mood stabilizers with poorer outcomes warrant clinical caution and further investigation.
Bottom Line
Significantly higher epigenetic aging has been observed in patients with major depressive disorder. Haloperidol plus lorazepam might be an effective treatment for delirium; and ramelteon may be effective for preventing delirium. Prazosin reduces heavy drinking in patients with alcohol use disorder. A 60-hour infusion of brexanolone can help alleviate postpartum depression. Clozapine plus aripiprazole reduces the risk of rehospitalization among patients with schizophrenia. Adding an antidepressant to an antipsychotic also can reduce the risk of rehospitalization among patients with schizophrenia.
Related Resources
- NEJM Journal Watch. www.jwatch.org.
- F1000 Prime. https://f1000.com/prime/home.
- BMJ Journals Evidence-Based Mental Health. https://ebmh.bmj.com.
Drug Brand Names
Aripiprazole • Abilify
Brexanolone • Zulresso
Clozapine • Clozaril
Dexmedetomidine • Precedex
Haloperidol • Haldol
Lorazepam • Ativan
Olanzapine • Zyprexa
Prazosin • Minipress
Ramelteon • Rozerem
Risperidone • Risperdal
1. Saeed SA, Stanley JB. Top research findings of 2018-2019. First of 2 parts. Current Psychiatry. 2020;19(1):13-18.
2. Han LKM, Aghajani M, Clark SL, et al. Epigenetic aging in major depressive disorder. Am J Psychiatry. 2018;175(8):774-782.
3. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526-535.
4. Simpson TL, Saxon AJ, Stappenbeck C, et al. Double-blind randomized clinical trial of prazosin for alcohol use disorder. Am J Psychiatry. 2018;175(12):1216-1224.
5. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
6. Tiihonen J, Taipale H, Mehtälä J, et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry. 2019;76(5):499-507.
7. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of adjunctive psychotropic medications in patients with schizophrenia. JAMA Psychiatry. 2019;76(5):508-515.
8. Blazer DG. Pharmacologic intervention for the treatment and prevention of delirium: looking beneath the modeling. JAMA Psychiatry. 2019;76(5):472-473.
9. Koob GF. Brain stress systems in the amygdala and addiction. Brain Res. 2009;1293:61-75.
10. Goff DC. Can adjunctive pharmacotherapy reduce hospitalization in schizophrenia? Insights from administrative databases. JAMA Psychiatry. 2019;76(5):468-469.
1. Saeed SA, Stanley JB. Top research findings of 2018-2019. First of 2 parts. Current Psychiatry. 2020;19(1):13-18.
2. Han LKM, Aghajani M, Clark SL, et al. Epigenetic aging in major depressive disorder. Am J Psychiatry. 2018;175(8):774-782.
3. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76(5):526-535.
4. Simpson TL, Saxon AJ, Stappenbeck C, et al. Double-blind randomized clinical trial of prazosin for alcohol use disorder. Am J Psychiatry. 2018;175(12):1216-1224.
5. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
6. Tiihonen J, Taipale H, Mehtälä J, et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry. 2019;76(5):499-507.
7. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of adjunctive psychotropic medications in patients with schizophrenia. JAMA Psychiatry. 2019;76(5):508-515.
8. Blazer DG. Pharmacologic intervention for the treatment and prevention of delirium: looking beneath the modeling. JAMA Psychiatry. 2019;76(5):472-473.
9. Koob GF. Brain stress systems in the amygdala and addiction. Brain Res. 2009;1293:61-75.
10. Goff DC. Can adjunctive pharmacotherapy reduce hospitalization in schizophrenia? Insights from administrative databases. JAMA Psychiatry. 2019;76(5):468-469.
Lumateperone for schizophrenia
Antipsychotic nonadherence is a known contributor to relapse risk among patients with schizophrenia.1 Because relapse episodes may be associated with antipsychotic treatment resistance, this must be avoided as much as possible by appropriate medication selection.2 Adverse effect burden is an important factor leading to oral antipsychotic nonadherence, with patient-derived data indicating that extrapyramidal symptoms (EPS) (odds ratio [OR] 0.57, P = .0007), sedation/cognitive adverse effects (OR 0.70, P = .033), prolactin/endocrine effects (OR 0.69, P = .0342), and metabolic adverse effects (OR 0.64, P = .0079) are all significantly related to lower rates of adherence.3 With this in mind, successive generations of antipsychotics have been released, with fewer tolerability issues present than seen with earlier compounds.1,4 Although these newer second-generation antipsychotics (SGAs) have not proven more effective for schizophrenia than those first marketed in the 1990s, they generally possess lower rates of EPS, hyperprolactinemia, anticholinergic and antihistaminic properties, metabolic adverse effects, and orthostasis.5 This improved adverse effect profile will hopefully increase the chances of antipsychotic acceptance in patients with schizophrenia, and thereby promote improved adherence.
Lumateperone (Caplyta) is a novel oral antipsychotic approved for the treatment of adult patients with schizophrenia (Table 1). It possesses some properties seen with other SGAs, including high affinity for serotonin 5HT2A receptors (Ki 0.54 nM) and lower affinity for dopamine D2 receptors (Ki 32 nM), along with low affinity for alpha1-adrenergic receptors (Ki 73 nM), and muscarinic and histaminergic receptors (Ki > 100 nM).6,7 However, there are some distinguishing features: the ratio of 5HT2A receptor affinity to D2 affinity is 60, greater than that of other SGAs such as risperidone (12), olanzapine (12.4) or aripiprazole (0.18)8; at steady state, the D2 occupancy remains <40% (Figure) and the corresponding rates of EPS/akathisia were only 6.7% for lumateperone vs 6.3% for placebo in short-term clinical trials.7,9

How it works
A unique aspect of lumateperone’s pharmacology may relate to differential actions at presynaptic and postsynaptic dopamine D2 receptors. Other antipsychotics possess comparable antagonist (or partial agonist) properties at postsynaptic D2 receptors (the D2 long isoform) and the presynaptic autoreceptor (the D2 short isoform). By blocking the presynaptic autoreceptor, feedback inhibition on dopamine release is removed; therefore, the required higher levels of postsynaptic D2 receptor occupancy needed for effective antipsychotic action (eg, 60% to 80% for antagonists, and 83% to 100% for partial agonists) may be a product of the need to oppose this increased presynaptic release of dopamine. In vitro assays show that lumateperone does not increase presynaptic dopamine release, indicating that it possesses agonist properties at the presynaptic D2 short receptor.10 That property may explain how lumateperone functions as an antipsychotic despite low levels of D2 receptor occupancy.10
Another hypothesis is based on our understanding of pimavanserin’s pharmacology. Pimavanserin is a selective 5HT2A antagonist FDA-approved for the treatment of Parkinson’s disease psychosis (PDP), with extremely high receptor affinity (Ki 0.087 nM) and no appreciable binding at dopamine receptors.5 Pimavanserin not only treats PDP, but is being evaluated in clinical trials for dementia-related psychosis, and has positive data for augmenting antipsychotics when there is a low level of D2 blockade.11,12 In a controlled trial, pimavanserin added to risperidone, 2 mg/d, was as effective as risperidone, 6 mg/d, illustrating the point that near-saturation of the 5HT2A receptor can increase antipsychotic efficacy when dopamine blockade is relatively low. For risperidone, 2 mg/d, the expected D2 occupancy is only 60%.13
Lumateperone also has moderate binding affinity for serotonin transporters (SERT) (Ki 33 nM). Serotonin transporter occupancy at the dose approved for schizophrenia (42 mg/d) is approximately 30%,14 below the ≥80% SERT occupancy seen with selective serotonin reuptake inhibitor (SSRI) antidepressants; nevertheless, there is evidence for antidepressant effects seen in preclinical assays, schizophrenia studies, and phase III trials for bipolar depression.8,15,16 It is hypothesized that near-saturation of the 5HT2A receptor might act synergistically with the modest extent of 5HT reuptake inhibition to promote downstream effects associated with effective antidepressant treatments.8 In vivo data also showed phosphorylation of N-methyl-
Clinical implications
Nonadherence with oral antipsychotics among patients with schizophrenia is often related to adverse effects.17 The SGAs marketed since 2000 generally have lower rates of sedation and metabolic and/or endocrine adverse events than earlier compounds, yet each has limitations:
- asenapine: sedation and weight gain
- the partial agonists (aripiprazole, brexpiprazole, cariprazine): akathisia
- lurasidone: dose-dependent EPS and akathisia
- iloperidone: orthostasis.18
Ziprasidone is an exception, because it had low rates of most adverse effects in schizophrenia trials, but the need to take it twice daily with a 500 kcal meal hampers its use. A meta-analysis of 32 oral antipsychotics, including first-generation agents, noted that the efficacy differences between medications are slight for patients without treatment-resistant schizophrenia, but “differences in side-effects are more marked.”18
Continue to: Until novel mechanisms are discovered...
Until novel mechanisms are discovered that increase schizophrenia response rates, the availability of newer antipsychotics with more favorable tolerability profiles presents clinicians and patients with added options when adverse effects interfere with prior treatment. In all phases of the adult schizophrenia trial program for lumateperone, 811 patients received short-term (4- to 6-week) exposure (dose range: 14 to 84 mg/d), while 329 had ≥6 months exposure and 108 had ≥1 year of exposure to the 42-mg/d dose. In these studies, there was no single adverse reaction leading to discontinuation that occurred at a rate >2%. The only adverse events that occurred at rates ≥5% and more than twice the rate of placebo were somnolence/sedation (lumateperone 24%, placebo 10%), and dry mouth (lumateperone 6%, placebo 2%). Nausea was present in 9% of the lumateperone group compared with 5% for placebo.7 In the short-term studies, the combined rate of EPS and akathisia was 6.7% for lumateperone and 6.3% for placebo.7 This difference translates to a number needed to harm of 250 for these neurologic adverse effects. The functional impact of lumateperone’s glutamatergic mechanisms is not well characterized within the schizophrenia population, but the antidepressant potential has been studied for patients with bipolar depression, with 1 positive phase III trial.19
Efficacy in adults with schizophrenia. The efficacy of lumateperone has been established in 2 pivotal, double-blind, placebo-controlled trials. The first was a 4-week, phase II trial (Study 005) in which 335 adults age 18 to 55 with an acute exacerbation of schizophrenia were randomized in a 1:1:1:1 manner to lumateperone, 42 mg/d (60 mg of lumateperone tosylate), lumateperone, 84 mg/d (120 mg of lumateperone tosylate), risperidone, 4 mg/d, or placebo, all taken once daily.20 For the 4 treatment arms, the least squares mean changes from baseline to the Day 28 endpoint on the primary outcome measure, Positive and Negative Syndrome Scale (PANSS) total score, were: lumateperone, 42 mg/d: −13.2 points; lumateperone, 84 mg/d: −8.3 points; risperidone, 4 mg/d: −13.4 points; and placebo: −7.4 points. Both lumateperone, 42 mg/d, and risperidone, 4 mg/d, were significantly different than placebo, and with identical moderate effect sizes of 0.4.20 Lumateperone, 84 mg/d, did not separate from placebo on the primary outcome. The responder analysis also indicated that a similar proportion of patients (40%) randomized to lumateperone, 42 mg/d, or risperidone, 4 mg/d, improved by ≥30% on PANSS total score.
The second pivotal trial (Study 301) was a phase III, double-blind, placebo-controlled trial of 450 adults, age 18 to 60, with an acute exacerbation of schizophrenia who were randomized in 1:1:1 manner to receive lumateperone, 42 mg/d (lumateperone tosylate 60 mg), lumateperone, 28 mg/d (lumateperone tosylate 40 mg), or placebo once daily for 4 weeks.21 For the 3 treatment arms, the least squares mean changes on PANSS total score from baseline to the Day 28 endpoint were: lumateperone, 42 mg/d: −14.5 points; lumateperone, 28 mg/d: −12.9 points; and placebo: −10.3 points. Lumateperone, 28 mg/d, did not separate from placebo on the primary outcome. The responder analysis also indicated that 36.5% of those receiving lumateperone, 42 mg/d, and 36.3% of those receiving lumateperone, 28 mg/d, improved by ≥30% on PANSS total score, compared with 25.5% of patients treated with placebo.
Unlike the 2 positive trials in which placebo change in total PANSS scores were −7.4 and −10.3 points, respectively, in a phase III trial (Study 302) with 696 participants, placebo showed a 15.1-point decrease from baseline PANSS total score.19 Among the 3 treatment arms of this study (lumateperone, 14 mg/d, lumateperone, 42 mg/d, and risperidone, 4 mg/d), only risperidone was superior to placebo.
Adverse events
In the phase II pivotal study, completion rates among the 4 arms were comparable: lumateperone, 42 mg/d: 71%; lumateperone, 84 mg/d: 76%; risperidone, 4 mg/d: 77%; and placebo: 72%.20 There were no serious adverse events (SAEs) associated with lumateperone; the 2 SAEs that occurred involved worsening of schizophrenia/psychotic disorder for risperidone (n = 1) and for placebo (n = 1). Five participants discontinued the study due to an adverse event: 2 who were receiving lumateperone (1 due to dry mouth, and 1 due to worsening of schizophrenia) and 3 who were receiving risperidone (2 due to akathisia, and 1 due to blood creatine phosphokinase increase).20 The most frequent adverse event was somnolence/sedation (placebo: 13%; lumateperone, 42 mg/d: 17%; risperidone, 4 mg/d: 21%; and lumateperone, 84 mg/d: 32.5%). Neither dose of lumateperone was associated with increased rates of EPS. Median weight gain to Day 28 was 1 kg for placebo and for each dose of lumateperone, and 2.5 kg for risperidone. Compared with risperidone, lumateperone showed statistically significantly lower prolactin levels (lumateperone, 42 mg/d and 84 mg/d: P < .001), and metabolic parameters, including fasting glucose (lumateperone 42 mg/d: P = .007; lumateperone, 84 mg/d: P = .023), total cholesterol (lumateperone, 42 mg/d: P = .012; lumateperone, 84 mg/d: P = .004), and triglycerides (lumateperone, 42 mg/d: P = .074; lumateperone, 84 mg/d: P = .002).20 There was no significant impact on the corrected QT interval.
Continue to: In the phase III trial...
In the phase III trial, completion rates among the 3 arms were lumateperone, 42 mg/d: 85%; lumateperone, 28 mg/d: 80%; and placebo: 74%. There was 1 SAE in a patient receiving lumateperone, 28 mg/d. This individual had preexisting risk factors and a history of seizures, and experienced a seizure during the study. Adverse events that occurred in either lumateperone group at a rate ≥5% and more than twice the rate of placebo were somnolence (lumateperone, 42 mg/d: 17.3%; lumateperone, 28 mg/d: 11.3%; and placebo: 4.0%); sedation (lumateperone, 42 mg/d: 12.7%; lumateperone, 28 mg/d: 9.3%; and placebo: 5.4%); fatigue (lumateperone, 42 mg/d: 5.3%; lumateperone, 28 mg/d: 4.7%; and placebo: 1.3%); and constipation (lumateperone, 42 mg/d: 6.7%; lumateperone, 28 mg/d: 4.0%; and placebo: 2.7%).21 No EPS-related adverse events occurred in ≥5% patients in any treatment arm. Median change in weight from baseline to Day 28 was 0.9 kg for lumateperone, 42 mg/d, 0.6 kg for lumateperone, 28 mg/d, and 0.7 kg for placebo. There were no significant mean changes in metabolic parameters for any treatment arm, and none of the patients had a corrected QT interval (QTc) >500 ms or a change in QTc >60 ms from baseline.21
Pharmacologic profile
Lumateperone’s in vitro binding profile includes high affinity for serotonin 5HT2A receptors (Ki 0.54 nM), lower affinity for dopamine D2 receptors (Ki 32 nM), moderate binding affinity for SERT (Ki 33 nM), and lower affinity for alpha 1-adrenergic receptors (Ki 73 nM) and muscarinic and histaminergic receptors (Ki >100 nM).6,7 As noted above, this 60-fold ratio of 5HT2A to D2 affinity is extremely high; moreover, imaging data reveal low D2 receptor occupancy, consistent with the lack of clinically significant EPS seen in the trials. In vitro assays also reveal impact on glutamate pathways, and pathways associated with antidepressant response.8 The clinical benefits of the glutamatergic properties remain theoretical, but the antidepressant benefit has been seen in a positive phase III trial for bipolar depression.19
Clinical considerations
Effect sizes in the 2 positive pivotal trials were 0.3 and 0.4, comparable with those for other antipsychotics approved within the last decade: brexpiprazole, 0.26; cariprazine, 0.34; and lurasidone, 0.36.21 The absence of clinically significant EPS, lack of impact on metabolic or endocrine parameters, and lack of titration are all appealing properties. That only 42 mg/d proved effective may reflect the fact that the other doses studied to date in randomized, fixed-dose studies were 14 mg/d (Study 302) and 84 mg/d (Study 005), evaluated in one study each. While those 2 doses might indeed be outside the therapeutic window, given the heterogeneity of schizophrenia, future studies might help further refine the therapeutic range for schizophrenia, especially for doses closer to 42 mg/d (eg, 28 mg/d, 63 mg/d). Should 42 mg/d not prove effective, there is no data for now to suggest whether a dose increase may be helpful. As there is only 1 marketed dose of lumateperone (42-mg capsules), and no easy way to modify this dose, lumateperone’s package insert includes cautionary language regarding situations where there will be less-than-expected drug exposure (use of cytochrome P450 [CYP] 3A4 inducers), greater-than-expected drug exposure (moderate or strong CYP 3A4 inhibitors), or use in patients with moderate or severe hepatic impairment as defined by Child-Pugh Criteria (Child-Pugh B or C). These are not contraindications.
Unique properties of lumateperone include the lack of presynaptic dopamine D2 antagonism, low D2 receptor occupancy, and the absence of significant EPS and metabolic or endocrine adverse effects. In vitro data indicate glutamatergic effects, and human data indicate antidepressant effects in bipolar patients. Despite the absence of significant histamine H1 or muscarinic affinity, the rate of somnolence/sedation was twice that of placebo (lumateperone 24%, placebo 10%).7
Why Rx? Reasons to prescribe lumateperone for adult patients with schizophrenia include:
- Favorable tolerability profile, including no significant signal for EPS or endocrine or metabolic adverse effects, and no QT prolongation
- No need for titration.
Dosing. There is only 1 dose available for lumateperone, 42-mg capsules (Table 2). As the dose cannot be modified, the package insert contains cautionary language regarding situations with less-than-expected drug exposure (use of CYP 3A4 inducers), greater-than-expected drug exposure (moderate or strong CYP 3A4 inhibitors), or use in patients with moderate or severe hepatic impairment as defined by Child-Pugh criteria (Child-Pugh B or C). These are not contraindications.
Contraindications. The only contraindication is known hypersensitivity to lumateperone.
Continue to: Bottom Line
Bottom Line
Lumateperone is a novel oral antipsychotic indicated for treating adults with schizophrenia. Its unique properties include the lack of presynaptic dopamine D2 antagonism, low D2 receptor occupancy, and the absence of significant extrapyramidal symptoms and metabolic or endocrine adverse effects. In clinical trials, the most frequent adverse event was somnolence/sedation.
Related Resource
- Fulton D. FDA approves Caplyta to treat schizophrenenia in adults. https://www.mdedge.com/psychiatry/article/214733/schizophrenia-other-psychotic-disorders/fda-approves-caplyta-treat.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Cariprazine • Vraylar
Iloperidone • Fanapt
Lumateperone • Caplyta
Lurasidone • Latuda
Olanzapine • Zyprexa
Pimavanserin • Nuplazid
Risperidone • Risperdal
Ziprasidone • Geodon
1. Dufort A, Zipursky RB. Understanding and managing treatment adherence in schizophrenia [published online January 3, 2019]. Clin Schizophr Relat Psychoses. 2019. doi: 10.3371/CSRP.ADRZ.121218.
2. Takeuchi H, Siu C, Remington G, et al. Does relapse contribute to treatment resistance? Antipsychotic response in first- vs. second-episode schizophrenia. Neuropsychopharmacology. 2019;44(6):1036-1042.
3. Dibonaventura M, Gabriel S, Dupclay L, et al. A patient perspective of the impact of medication side effects on adherence: results of a cross-sectional nationwide survey of patients with schizophrenia. BMC Psychiatry. 2012;12:20.
4. Kurokawa S, Kishimoto T, Su K-P, et al. Psychiatrists’ perceptions of medication adherence among patients with schizophrenia: an international survey. Schizophr Res. 2019;211:105-107.
5. Meyer JM. Pharmacotherapy of psychosis and mania. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. 13th ed. Chicago, Illinois: McGraw-Hill; 2018:279-302.
6. Davis RE, Correll CU. ITI-007 in the treatment of schizophrenia: from novel pharmacology to clinical outcomes. Expert Rev Neurother. 2016;16(6):601-614.
7. Caplyta [package Insert]. New York, NY: Intra-Cellular Therapies, Inc.; 2019.
8. Snyder GL, Vanover KE, Zhu H, et al. Functional profile of a novel modulator of serotonin, dopamine, and glutamate neurotransmission. Psychopharmacology (Berl). 2015;232(3):605-621.
9. Vanover KE, Davis RE, Zhou Y, et al. Dopamine D2 receptor occupancy of lumateperone (ITI-007): a positron emission tomography study in patients with schizophrenia. Neuropsychopharmacology. 2019;44(3):598-605.
10. Zhang L, Hendrick JP. The presynaptic D2 partial agonist lumateperone acts as a postsynaptic D2 antagonist. Matters. 2018. doi: 10.19185/matters.201712000006.
11. Meltzer HY, Elkis H, Vanover K, et al. Pimavanserin, a selective serotonin (5-HT)2A-inverse agonist, enhances the efficacy and safety of risperidone, 2mg/day, but does not enhance efficacy of haloperidol, 2mg/day: comparison with reference dose risperidone, 6mg/day. Schizophr Res. 2012;141(2-3):144-152.
12. Nasrallah HA, Fedora R, Morton R. Successful treatment of clozapine-nonresponsive refractory hallucinations and delusions with pimavanserin, a serotonin 5HT-2A receptor inverse agonist. Schizophr Res. 2019;208:217-220.
13. Remington G, Mamo D, Labelle A, et al. A PET study evaluating dopamine D2 receptor occupancy for long-acting injectable risperidone. Am J Psychiatry. 2006;163(3):396-401.
14. Davis RE, Vanover KE, Zhou Y, et al. ITI-007 demonstrates brain occupancy at serotonin 5-HT2A and dopamine D2 receptors and serotonin transporters using positron emission tomography in healthy volunteers. Psychopharmacology (Berl). 2015;232(15):2863-2872.
15. Kumar B, Kuhad A, Kuhad A. Lumateperone: a new treatment approach for neuropsychiatric disorders. Drugs Today (Barc). 2018;54(12):713-719.
16. Vanover K, Glass S, Kozauer S, et al. 30 lumateperone (ITI-007) for the treatment of schizophrenia: overview of placebo-controlled clinical trials and an open-label safety switching study. CNS Spectr. 2019;24(1):190-191.
17. Young SL, Taylor M, Lawrie SM. “First do no harm.” A systematic review of the prevalence and management of antipsychotic adverse effects. J Psychopharmacol. 2015;29(4):353-362.
18. Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394(10202):939-951.
19. Vyas P, Hwang BJ, Brašic ´ JR. An evaluation of lumateperone tosylate for the treatment of schizophrenia. Expert Opin Pharmacother. 2019;1-7.
20. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961.
21. Correll CU, Davis RE, Weingart M, et al. Efficacy and safety of lumateperone for treatment of schizophrenia [published online January 8, 2020]. JAMA Psychiatry. 2020;E1-E10.
Antipsychotic nonadherence is a known contributor to relapse risk among patients with schizophrenia.1 Because relapse episodes may be associated with antipsychotic treatment resistance, this must be avoided as much as possible by appropriate medication selection.2 Adverse effect burden is an important factor leading to oral antipsychotic nonadherence, with patient-derived data indicating that extrapyramidal symptoms (EPS) (odds ratio [OR] 0.57, P = .0007), sedation/cognitive adverse effects (OR 0.70, P = .033), prolactin/endocrine effects (OR 0.69, P = .0342), and metabolic adverse effects (OR 0.64, P = .0079) are all significantly related to lower rates of adherence.3 With this in mind, successive generations of antipsychotics have been released, with fewer tolerability issues present than seen with earlier compounds.1,4 Although these newer second-generation antipsychotics (SGAs) have not proven more effective for schizophrenia than those first marketed in the 1990s, they generally possess lower rates of EPS, hyperprolactinemia, anticholinergic and antihistaminic properties, metabolic adverse effects, and orthostasis.5 This improved adverse effect profile will hopefully increase the chances of antipsychotic acceptance in patients with schizophrenia, and thereby promote improved adherence.
Lumateperone (Caplyta) is a novel oral antipsychotic approved for the treatment of adult patients with schizophrenia (Table 1). It possesses some properties seen with other SGAs, including high affinity for serotonin 5HT2A receptors (Ki 0.54 nM) and lower affinity for dopamine D2 receptors (Ki 32 nM), along with low affinity for alpha1-adrenergic receptors (Ki 73 nM), and muscarinic and histaminergic receptors (Ki > 100 nM).6,7 However, there are some distinguishing features: the ratio of 5HT2A receptor affinity to D2 affinity is 60, greater than that of other SGAs such as risperidone (12), olanzapine (12.4) or aripiprazole (0.18)8; at steady state, the D2 occupancy remains <40% (Figure) and the corresponding rates of EPS/akathisia were only 6.7% for lumateperone vs 6.3% for placebo in short-term clinical trials.7,9

How it works
A unique aspect of lumateperone’s pharmacology may relate to differential actions at presynaptic and postsynaptic dopamine D2 receptors. Other antipsychotics possess comparable antagonist (or partial agonist) properties at postsynaptic D2 receptors (the D2 long isoform) and the presynaptic autoreceptor (the D2 short isoform). By blocking the presynaptic autoreceptor, feedback inhibition on dopamine release is removed; therefore, the required higher levels of postsynaptic D2 receptor occupancy needed for effective antipsychotic action (eg, 60% to 80% for antagonists, and 83% to 100% for partial agonists) may be a product of the need to oppose this increased presynaptic release of dopamine. In vitro assays show that lumateperone does not increase presynaptic dopamine release, indicating that it possesses agonist properties at the presynaptic D2 short receptor.10 That property may explain how lumateperone functions as an antipsychotic despite low levels of D2 receptor occupancy.10
Another hypothesis is based on our understanding of pimavanserin’s pharmacology. Pimavanserin is a selective 5HT2A antagonist FDA-approved for the treatment of Parkinson’s disease psychosis (PDP), with extremely high receptor affinity (Ki 0.087 nM) and no appreciable binding at dopamine receptors.5 Pimavanserin not only treats PDP, but is being evaluated in clinical trials for dementia-related psychosis, and has positive data for augmenting antipsychotics when there is a low level of D2 blockade.11,12 In a controlled trial, pimavanserin added to risperidone, 2 mg/d, was as effective as risperidone, 6 mg/d, illustrating the point that near-saturation of the 5HT2A receptor can increase antipsychotic efficacy when dopamine blockade is relatively low. For risperidone, 2 mg/d, the expected D2 occupancy is only 60%.13
Lumateperone also has moderate binding affinity for serotonin transporters (SERT) (Ki 33 nM). Serotonin transporter occupancy at the dose approved for schizophrenia (42 mg/d) is approximately 30%,14 below the ≥80% SERT occupancy seen with selective serotonin reuptake inhibitor (SSRI) antidepressants; nevertheless, there is evidence for antidepressant effects seen in preclinical assays, schizophrenia studies, and phase III trials for bipolar depression.8,15,16 It is hypothesized that near-saturation of the 5HT2A receptor might act synergistically with the modest extent of 5HT reuptake inhibition to promote downstream effects associated with effective antidepressant treatments.8 In vivo data also showed phosphorylation of N-methyl-
Clinical implications
Nonadherence with oral antipsychotics among patients with schizophrenia is often related to adverse effects.17 The SGAs marketed since 2000 generally have lower rates of sedation and metabolic and/or endocrine adverse events than earlier compounds, yet each has limitations:
- asenapine: sedation and weight gain
- the partial agonists (aripiprazole, brexpiprazole, cariprazine): akathisia
- lurasidone: dose-dependent EPS and akathisia
- iloperidone: orthostasis.18
Ziprasidone is an exception, because it had low rates of most adverse effects in schizophrenia trials, but the need to take it twice daily with a 500 kcal meal hampers its use. A meta-analysis of 32 oral antipsychotics, including first-generation agents, noted that the efficacy differences between medications are slight for patients without treatment-resistant schizophrenia, but “differences in side-effects are more marked.”18
Continue to: Until novel mechanisms are discovered...
Until novel mechanisms are discovered that increase schizophrenia response rates, the availability of newer antipsychotics with more favorable tolerability profiles presents clinicians and patients with added options when adverse effects interfere with prior treatment. In all phases of the adult schizophrenia trial program for lumateperone, 811 patients received short-term (4- to 6-week) exposure (dose range: 14 to 84 mg/d), while 329 had ≥6 months exposure and 108 had ≥1 year of exposure to the 42-mg/d dose. In these studies, there was no single adverse reaction leading to discontinuation that occurred at a rate >2%. The only adverse events that occurred at rates ≥5% and more than twice the rate of placebo were somnolence/sedation (lumateperone 24%, placebo 10%), and dry mouth (lumateperone 6%, placebo 2%). Nausea was present in 9% of the lumateperone group compared with 5% for placebo.7 In the short-term studies, the combined rate of EPS and akathisia was 6.7% for lumateperone and 6.3% for placebo.7 This difference translates to a number needed to harm of 250 for these neurologic adverse effects. The functional impact of lumateperone’s glutamatergic mechanisms is not well characterized within the schizophrenia population, but the antidepressant potential has been studied for patients with bipolar depression, with 1 positive phase III trial.19
Efficacy in adults with schizophrenia. The efficacy of lumateperone has been established in 2 pivotal, double-blind, placebo-controlled trials. The first was a 4-week, phase II trial (Study 005) in which 335 adults age 18 to 55 with an acute exacerbation of schizophrenia were randomized in a 1:1:1:1 manner to lumateperone, 42 mg/d (60 mg of lumateperone tosylate), lumateperone, 84 mg/d (120 mg of lumateperone tosylate), risperidone, 4 mg/d, or placebo, all taken once daily.20 For the 4 treatment arms, the least squares mean changes from baseline to the Day 28 endpoint on the primary outcome measure, Positive and Negative Syndrome Scale (PANSS) total score, were: lumateperone, 42 mg/d: −13.2 points; lumateperone, 84 mg/d: −8.3 points; risperidone, 4 mg/d: −13.4 points; and placebo: −7.4 points. Both lumateperone, 42 mg/d, and risperidone, 4 mg/d, were significantly different than placebo, and with identical moderate effect sizes of 0.4.20 Lumateperone, 84 mg/d, did not separate from placebo on the primary outcome. The responder analysis also indicated that a similar proportion of patients (40%) randomized to lumateperone, 42 mg/d, or risperidone, 4 mg/d, improved by ≥30% on PANSS total score.
The second pivotal trial (Study 301) was a phase III, double-blind, placebo-controlled trial of 450 adults, age 18 to 60, with an acute exacerbation of schizophrenia who were randomized in 1:1:1 manner to receive lumateperone, 42 mg/d (lumateperone tosylate 60 mg), lumateperone, 28 mg/d (lumateperone tosylate 40 mg), or placebo once daily for 4 weeks.21 For the 3 treatment arms, the least squares mean changes on PANSS total score from baseline to the Day 28 endpoint were: lumateperone, 42 mg/d: −14.5 points; lumateperone, 28 mg/d: −12.9 points; and placebo: −10.3 points. Lumateperone, 28 mg/d, did not separate from placebo on the primary outcome. The responder analysis also indicated that 36.5% of those receiving lumateperone, 42 mg/d, and 36.3% of those receiving lumateperone, 28 mg/d, improved by ≥30% on PANSS total score, compared with 25.5% of patients treated with placebo.
Unlike the 2 positive trials in which placebo change in total PANSS scores were −7.4 and −10.3 points, respectively, in a phase III trial (Study 302) with 696 participants, placebo showed a 15.1-point decrease from baseline PANSS total score.19 Among the 3 treatment arms of this study (lumateperone, 14 mg/d, lumateperone, 42 mg/d, and risperidone, 4 mg/d), only risperidone was superior to placebo.
Adverse events
In the phase II pivotal study, completion rates among the 4 arms were comparable: lumateperone, 42 mg/d: 71%; lumateperone, 84 mg/d: 76%; risperidone, 4 mg/d: 77%; and placebo: 72%.20 There were no serious adverse events (SAEs) associated with lumateperone; the 2 SAEs that occurred involved worsening of schizophrenia/psychotic disorder for risperidone (n = 1) and for placebo (n = 1). Five participants discontinued the study due to an adverse event: 2 who were receiving lumateperone (1 due to dry mouth, and 1 due to worsening of schizophrenia) and 3 who were receiving risperidone (2 due to akathisia, and 1 due to blood creatine phosphokinase increase).20 The most frequent adverse event was somnolence/sedation (placebo: 13%; lumateperone, 42 mg/d: 17%; risperidone, 4 mg/d: 21%; and lumateperone, 84 mg/d: 32.5%). Neither dose of lumateperone was associated with increased rates of EPS. Median weight gain to Day 28 was 1 kg for placebo and for each dose of lumateperone, and 2.5 kg for risperidone. Compared with risperidone, lumateperone showed statistically significantly lower prolactin levels (lumateperone, 42 mg/d and 84 mg/d: P < .001), and metabolic parameters, including fasting glucose (lumateperone 42 mg/d: P = .007; lumateperone, 84 mg/d: P = .023), total cholesterol (lumateperone, 42 mg/d: P = .012; lumateperone, 84 mg/d: P = .004), and triglycerides (lumateperone, 42 mg/d: P = .074; lumateperone, 84 mg/d: P = .002).20 There was no significant impact on the corrected QT interval.
Continue to: In the phase III trial...
In the phase III trial, completion rates among the 3 arms were lumateperone, 42 mg/d: 85%; lumateperone, 28 mg/d: 80%; and placebo: 74%. There was 1 SAE in a patient receiving lumateperone, 28 mg/d. This individual had preexisting risk factors and a history of seizures, and experienced a seizure during the study. Adverse events that occurred in either lumateperone group at a rate ≥5% and more than twice the rate of placebo were somnolence (lumateperone, 42 mg/d: 17.3%; lumateperone, 28 mg/d: 11.3%; and placebo: 4.0%); sedation (lumateperone, 42 mg/d: 12.7%; lumateperone, 28 mg/d: 9.3%; and placebo: 5.4%); fatigue (lumateperone, 42 mg/d: 5.3%; lumateperone, 28 mg/d: 4.7%; and placebo: 1.3%); and constipation (lumateperone, 42 mg/d: 6.7%; lumateperone, 28 mg/d: 4.0%; and placebo: 2.7%).21 No EPS-related adverse events occurred in ≥5% patients in any treatment arm. Median change in weight from baseline to Day 28 was 0.9 kg for lumateperone, 42 mg/d, 0.6 kg for lumateperone, 28 mg/d, and 0.7 kg for placebo. There were no significant mean changes in metabolic parameters for any treatment arm, and none of the patients had a corrected QT interval (QTc) >500 ms or a change in QTc >60 ms from baseline.21
Pharmacologic profile
Lumateperone’s in vitro binding profile includes high affinity for serotonin 5HT2A receptors (Ki 0.54 nM), lower affinity for dopamine D2 receptors (Ki 32 nM), moderate binding affinity for SERT (Ki 33 nM), and lower affinity for alpha 1-adrenergic receptors (Ki 73 nM) and muscarinic and histaminergic receptors (Ki >100 nM).6,7 As noted above, this 60-fold ratio of 5HT2A to D2 affinity is extremely high; moreover, imaging data reveal low D2 receptor occupancy, consistent with the lack of clinically significant EPS seen in the trials. In vitro assays also reveal impact on glutamate pathways, and pathways associated with antidepressant response.8 The clinical benefits of the glutamatergic properties remain theoretical, but the antidepressant benefit has been seen in a positive phase III trial for bipolar depression.19
Clinical considerations
Effect sizes in the 2 positive pivotal trials were 0.3 and 0.4, comparable with those for other antipsychotics approved within the last decade: brexpiprazole, 0.26; cariprazine, 0.34; and lurasidone, 0.36.21 The absence of clinically significant EPS, lack of impact on metabolic or endocrine parameters, and lack of titration are all appealing properties. That only 42 mg/d proved effective may reflect the fact that the other doses studied to date in randomized, fixed-dose studies were 14 mg/d (Study 302) and 84 mg/d (Study 005), evaluated in one study each. While those 2 doses might indeed be outside the therapeutic window, given the heterogeneity of schizophrenia, future studies might help further refine the therapeutic range for schizophrenia, especially for doses closer to 42 mg/d (eg, 28 mg/d, 63 mg/d). Should 42 mg/d not prove effective, there is no data for now to suggest whether a dose increase may be helpful. As there is only 1 marketed dose of lumateperone (42-mg capsules), and no easy way to modify this dose, lumateperone’s package insert includes cautionary language regarding situations where there will be less-than-expected drug exposure (use of cytochrome P450 [CYP] 3A4 inducers), greater-than-expected drug exposure (moderate or strong CYP 3A4 inhibitors), or use in patients with moderate or severe hepatic impairment as defined by Child-Pugh Criteria (Child-Pugh B or C). These are not contraindications.
Unique properties of lumateperone include the lack of presynaptic dopamine D2 antagonism, low D2 receptor occupancy, and the absence of significant EPS and metabolic or endocrine adverse effects. In vitro data indicate glutamatergic effects, and human data indicate antidepressant effects in bipolar patients. Despite the absence of significant histamine H1 or muscarinic affinity, the rate of somnolence/sedation was twice that of placebo (lumateperone 24%, placebo 10%).7
Why Rx? Reasons to prescribe lumateperone for adult patients with schizophrenia include:
- Favorable tolerability profile, including no significant signal for EPS or endocrine or metabolic adverse effects, and no QT prolongation
- No need for titration.
Dosing. There is only 1 dose available for lumateperone, 42-mg capsules (Table 2). As the dose cannot be modified, the package insert contains cautionary language regarding situations with less-than-expected drug exposure (use of CYP 3A4 inducers), greater-than-expected drug exposure (moderate or strong CYP 3A4 inhibitors), or use in patients with moderate or severe hepatic impairment as defined by Child-Pugh criteria (Child-Pugh B or C). These are not contraindications.
Contraindications. The only contraindication is known hypersensitivity to lumateperone.
Continue to: Bottom Line
Bottom Line
Lumateperone is a novel oral antipsychotic indicated for treating adults with schizophrenia. Its unique properties include the lack of presynaptic dopamine D2 antagonism, low D2 receptor occupancy, and the absence of significant extrapyramidal symptoms and metabolic or endocrine adverse effects. In clinical trials, the most frequent adverse event was somnolence/sedation.
Related Resource
- Fulton D. FDA approves Caplyta to treat schizophrenenia in adults. https://www.mdedge.com/psychiatry/article/214733/schizophrenia-other-psychotic-disorders/fda-approves-caplyta-treat.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Cariprazine • Vraylar
Iloperidone • Fanapt
Lumateperone • Caplyta
Lurasidone • Latuda
Olanzapine • Zyprexa
Pimavanserin • Nuplazid
Risperidone • Risperdal
Ziprasidone • Geodon
Antipsychotic nonadherence is a known contributor to relapse risk among patients with schizophrenia.1 Because relapse episodes may be associated with antipsychotic treatment resistance, this must be avoided as much as possible by appropriate medication selection.2 Adverse effect burden is an important factor leading to oral antipsychotic nonadherence, with patient-derived data indicating that extrapyramidal symptoms (EPS) (odds ratio [OR] 0.57, P = .0007), sedation/cognitive adverse effects (OR 0.70, P = .033), prolactin/endocrine effects (OR 0.69, P = .0342), and metabolic adverse effects (OR 0.64, P = .0079) are all significantly related to lower rates of adherence.3 With this in mind, successive generations of antipsychotics have been released, with fewer tolerability issues present than seen with earlier compounds.1,4 Although these newer second-generation antipsychotics (SGAs) have not proven more effective for schizophrenia than those first marketed in the 1990s, they generally possess lower rates of EPS, hyperprolactinemia, anticholinergic and antihistaminic properties, metabolic adverse effects, and orthostasis.5 This improved adverse effect profile will hopefully increase the chances of antipsychotic acceptance in patients with schizophrenia, and thereby promote improved adherence.
Lumateperone (Caplyta) is a novel oral antipsychotic approved for the treatment of adult patients with schizophrenia (Table 1). It possesses some properties seen with other SGAs, including high affinity for serotonin 5HT2A receptors (Ki 0.54 nM) and lower affinity for dopamine D2 receptors (Ki 32 nM), along with low affinity for alpha1-adrenergic receptors (Ki 73 nM), and muscarinic and histaminergic receptors (Ki > 100 nM).6,7 However, there are some distinguishing features: the ratio of 5HT2A receptor affinity to D2 affinity is 60, greater than that of other SGAs such as risperidone (12), olanzapine (12.4) or aripiprazole (0.18)8; at steady state, the D2 occupancy remains <40% (Figure) and the corresponding rates of EPS/akathisia were only 6.7% for lumateperone vs 6.3% for placebo in short-term clinical trials.7,9

How it works
A unique aspect of lumateperone’s pharmacology may relate to differential actions at presynaptic and postsynaptic dopamine D2 receptors. Other antipsychotics possess comparable antagonist (or partial agonist) properties at postsynaptic D2 receptors (the D2 long isoform) and the presynaptic autoreceptor (the D2 short isoform). By blocking the presynaptic autoreceptor, feedback inhibition on dopamine release is removed; therefore, the required higher levels of postsynaptic D2 receptor occupancy needed for effective antipsychotic action (eg, 60% to 80% for antagonists, and 83% to 100% for partial agonists) may be a product of the need to oppose this increased presynaptic release of dopamine. In vitro assays show that lumateperone does not increase presynaptic dopamine release, indicating that it possesses agonist properties at the presynaptic D2 short receptor.10 That property may explain how lumateperone functions as an antipsychotic despite low levels of D2 receptor occupancy.10
Another hypothesis is based on our understanding of pimavanserin’s pharmacology. Pimavanserin is a selective 5HT2A antagonist FDA-approved for the treatment of Parkinson’s disease psychosis (PDP), with extremely high receptor affinity (Ki 0.087 nM) and no appreciable binding at dopamine receptors.5 Pimavanserin not only treats PDP, but is being evaluated in clinical trials for dementia-related psychosis, and has positive data for augmenting antipsychotics when there is a low level of D2 blockade.11,12 In a controlled trial, pimavanserin added to risperidone, 2 mg/d, was as effective as risperidone, 6 mg/d, illustrating the point that near-saturation of the 5HT2A receptor can increase antipsychotic efficacy when dopamine blockade is relatively low. For risperidone, 2 mg/d, the expected D2 occupancy is only 60%.13
Lumateperone also has moderate binding affinity for serotonin transporters (SERT) (Ki 33 nM). Serotonin transporter occupancy at the dose approved for schizophrenia (42 mg/d) is approximately 30%,14 below the ≥80% SERT occupancy seen with selective serotonin reuptake inhibitor (SSRI) antidepressants; nevertheless, there is evidence for antidepressant effects seen in preclinical assays, schizophrenia studies, and phase III trials for bipolar depression.8,15,16 It is hypothesized that near-saturation of the 5HT2A receptor might act synergistically with the modest extent of 5HT reuptake inhibition to promote downstream effects associated with effective antidepressant treatments.8 In vivo data also showed phosphorylation of N-methyl-
Clinical implications
Nonadherence with oral antipsychotics among patients with schizophrenia is often related to adverse effects.17 The SGAs marketed since 2000 generally have lower rates of sedation and metabolic and/or endocrine adverse events than earlier compounds, yet each has limitations:
- asenapine: sedation and weight gain
- the partial agonists (aripiprazole, brexpiprazole, cariprazine): akathisia
- lurasidone: dose-dependent EPS and akathisia
- iloperidone: orthostasis.18
Ziprasidone is an exception, because it had low rates of most adverse effects in schizophrenia trials, but the need to take it twice daily with a 500 kcal meal hampers its use. A meta-analysis of 32 oral antipsychotics, including first-generation agents, noted that the efficacy differences between medications are slight for patients without treatment-resistant schizophrenia, but “differences in side-effects are more marked.”18
Continue to: Until novel mechanisms are discovered...
Until novel mechanisms are discovered that increase schizophrenia response rates, the availability of newer antipsychotics with more favorable tolerability profiles presents clinicians and patients with added options when adverse effects interfere with prior treatment. In all phases of the adult schizophrenia trial program for lumateperone, 811 patients received short-term (4- to 6-week) exposure (dose range: 14 to 84 mg/d), while 329 had ≥6 months exposure and 108 had ≥1 year of exposure to the 42-mg/d dose. In these studies, there was no single adverse reaction leading to discontinuation that occurred at a rate >2%. The only adverse events that occurred at rates ≥5% and more than twice the rate of placebo were somnolence/sedation (lumateperone 24%, placebo 10%), and dry mouth (lumateperone 6%, placebo 2%). Nausea was present in 9% of the lumateperone group compared with 5% for placebo.7 In the short-term studies, the combined rate of EPS and akathisia was 6.7% for lumateperone and 6.3% for placebo.7 This difference translates to a number needed to harm of 250 for these neurologic adverse effects. The functional impact of lumateperone’s glutamatergic mechanisms is not well characterized within the schizophrenia population, but the antidepressant potential has been studied for patients with bipolar depression, with 1 positive phase III trial.19
Efficacy in adults with schizophrenia. The efficacy of lumateperone has been established in 2 pivotal, double-blind, placebo-controlled trials. The first was a 4-week, phase II trial (Study 005) in which 335 adults age 18 to 55 with an acute exacerbation of schizophrenia were randomized in a 1:1:1:1 manner to lumateperone, 42 mg/d (60 mg of lumateperone tosylate), lumateperone, 84 mg/d (120 mg of lumateperone tosylate), risperidone, 4 mg/d, or placebo, all taken once daily.20 For the 4 treatment arms, the least squares mean changes from baseline to the Day 28 endpoint on the primary outcome measure, Positive and Negative Syndrome Scale (PANSS) total score, were: lumateperone, 42 mg/d: −13.2 points; lumateperone, 84 mg/d: −8.3 points; risperidone, 4 mg/d: −13.4 points; and placebo: −7.4 points. Both lumateperone, 42 mg/d, and risperidone, 4 mg/d, were significantly different than placebo, and with identical moderate effect sizes of 0.4.20 Lumateperone, 84 mg/d, did not separate from placebo on the primary outcome. The responder analysis also indicated that a similar proportion of patients (40%) randomized to lumateperone, 42 mg/d, or risperidone, 4 mg/d, improved by ≥30% on PANSS total score.
The second pivotal trial (Study 301) was a phase III, double-blind, placebo-controlled trial of 450 adults, age 18 to 60, with an acute exacerbation of schizophrenia who were randomized in 1:1:1 manner to receive lumateperone, 42 mg/d (lumateperone tosylate 60 mg), lumateperone, 28 mg/d (lumateperone tosylate 40 mg), or placebo once daily for 4 weeks.21 For the 3 treatment arms, the least squares mean changes on PANSS total score from baseline to the Day 28 endpoint were: lumateperone, 42 mg/d: −14.5 points; lumateperone, 28 mg/d: −12.9 points; and placebo: −10.3 points. Lumateperone, 28 mg/d, did not separate from placebo on the primary outcome. The responder analysis also indicated that 36.5% of those receiving lumateperone, 42 mg/d, and 36.3% of those receiving lumateperone, 28 mg/d, improved by ≥30% on PANSS total score, compared with 25.5% of patients treated with placebo.
Unlike the 2 positive trials in which placebo change in total PANSS scores were −7.4 and −10.3 points, respectively, in a phase III trial (Study 302) with 696 participants, placebo showed a 15.1-point decrease from baseline PANSS total score.19 Among the 3 treatment arms of this study (lumateperone, 14 mg/d, lumateperone, 42 mg/d, and risperidone, 4 mg/d), only risperidone was superior to placebo.
Adverse events
In the phase II pivotal study, completion rates among the 4 arms were comparable: lumateperone, 42 mg/d: 71%; lumateperone, 84 mg/d: 76%; risperidone, 4 mg/d: 77%; and placebo: 72%.20 There were no serious adverse events (SAEs) associated with lumateperone; the 2 SAEs that occurred involved worsening of schizophrenia/psychotic disorder for risperidone (n = 1) and for placebo (n = 1). Five participants discontinued the study due to an adverse event: 2 who were receiving lumateperone (1 due to dry mouth, and 1 due to worsening of schizophrenia) and 3 who were receiving risperidone (2 due to akathisia, and 1 due to blood creatine phosphokinase increase).20 The most frequent adverse event was somnolence/sedation (placebo: 13%; lumateperone, 42 mg/d: 17%; risperidone, 4 mg/d: 21%; and lumateperone, 84 mg/d: 32.5%). Neither dose of lumateperone was associated with increased rates of EPS. Median weight gain to Day 28 was 1 kg for placebo and for each dose of lumateperone, and 2.5 kg for risperidone. Compared with risperidone, lumateperone showed statistically significantly lower prolactin levels (lumateperone, 42 mg/d and 84 mg/d: P < .001), and metabolic parameters, including fasting glucose (lumateperone 42 mg/d: P = .007; lumateperone, 84 mg/d: P = .023), total cholesterol (lumateperone, 42 mg/d: P = .012; lumateperone, 84 mg/d: P = .004), and triglycerides (lumateperone, 42 mg/d: P = .074; lumateperone, 84 mg/d: P = .002).20 There was no significant impact on the corrected QT interval.
Continue to: In the phase III trial...
In the phase III trial, completion rates among the 3 arms were lumateperone, 42 mg/d: 85%; lumateperone, 28 mg/d: 80%; and placebo: 74%. There was 1 SAE in a patient receiving lumateperone, 28 mg/d. This individual had preexisting risk factors and a history of seizures, and experienced a seizure during the study. Adverse events that occurred in either lumateperone group at a rate ≥5% and more than twice the rate of placebo were somnolence (lumateperone, 42 mg/d: 17.3%; lumateperone, 28 mg/d: 11.3%; and placebo: 4.0%); sedation (lumateperone, 42 mg/d: 12.7%; lumateperone, 28 mg/d: 9.3%; and placebo: 5.4%); fatigue (lumateperone, 42 mg/d: 5.3%; lumateperone, 28 mg/d: 4.7%; and placebo: 1.3%); and constipation (lumateperone, 42 mg/d: 6.7%; lumateperone, 28 mg/d: 4.0%; and placebo: 2.7%).21 No EPS-related adverse events occurred in ≥5% patients in any treatment arm. Median change in weight from baseline to Day 28 was 0.9 kg for lumateperone, 42 mg/d, 0.6 kg for lumateperone, 28 mg/d, and 0.7 kg for placebo. There were no significant mean changes in metabolic parameters for any treatment arm, and none of the patients had a corrected QT interval (QTc) >500 ms or a change in QTc >60 ms from baseline.21
Pharmacologic profile
Lumateperone’s in vitro binding profile includes high affinity for serotonin 5HT2A receptors (Ki 0.54 nM), lower affinity for dopamine D2 receptors (Ki 32 nM), moderate binding affinity for SERT (Ki 33 nM), and lower affinity for alpha 1-adrenergic receptors (Ki 73 nM) and muscarinic and histaminergic receptors (Ki >100 nM).6,7 As noted above, this 60-fold ratio of 5HT2A to D2 affinity is extremely high; moreover, imaging data reveal low D2 receptor occupancy, consistent with the lack of clinically significant EPS seen in the trials. In vitro assays also reveal impact on glutamate pathways, and pathways associated with antidepressant response.8 The clinical benefits of the glutamatergic properties remain theoretical, but the antidepressant benefit has been seen in a positive phase III trial for bipolar depression.19
Clinical considerations
Effect sizes in the 2 positive pivotal trials were 0.3 and 0.4, comparable with those for other antipsychotics approved within the last decade: brexpiprazole, 0.26; cariprazine, 0.34; and lurasidone, 0.36.21 The absence of clinically significant EPS, lack of impact on metabolic or endocrine parameters, and lack of titration are all appealing properties. That only 42 mg/d proved effective may reflect the fact that the other doses studied to date in randomized, fixed-dose studies were 14 mg/d (Study 302) and 84 mg/d (Study 005), evaluated in one study each. While those 2 doses might indeed be outside the therapeutic window, given the heterogeneity of schizophrenia, future studies might help further refine the therapeutic range for schizophrenia, especially for doses closer to 42 mg/d (eg, 28 mg/d, 63 mg/d). Should 42 mg/d not prove effective, there is no data for now to suggest whether a dose increase may be helpful. As there is only 1 marketed dose of lumateperone (42-mg capsules), and no easy way to modify this dose, lumateperone’s package insert includes cautionary language regarding situations where there will be less-than-expected drug exposure (use of cytochrome P450 [CYP] 3A4 inducers), greater-than-expected drug exposure (moderate or strong CYP 3A4 inhibitors), or use in patients with moderate or severe hepatic impairment as defined by Child-Pugh Criteria (Child-Pugh B or C). These are not contraindications.
Unique properties of lumateperone include the lack of presynaptic dopamine D2 antagonism, low D2 receptor occupancy, and the absence of significant EPS and metabolic or endocrine adverse effects. In vitro data indicate glutamatergic effects, and human data indicate antidepressant effects in bipolar patients. Despite the absence of significant histamine H1 or muscarinic affinity, the rate of somnolence/sedation was twice that of placebo (lumateperone 24%, placebo 10%).7
Why Rx? Reasons to prescribe lumateperone for adult patients with schizophrenia include:
- Favorable tolerability profile, including no significant signal for EPS or endocrine or metabolic adverse effects, and no QT prolongation
- No need for titration.
Dosing. There is only 1 dose available for lumateperone, 42-mg capsules (Table 2). As the dose cannot be modified, the package insert contains cautionary language regarding situations with less-than-expected drug exposure (use of CYP 3A4 inducers), greater-than-expected drug exposure (moderate or strong CYP 3A4 inhibitors), or use in patients with moderate or severe hepatic impairment as defined by Child-Pugh criteria (Child-Pugh B or C). These are not contraindications.
Contraindications. The only contraindication is known hypersensitivity to lumateperone.
Continue to: Bottom Line
Bottom Line
Lumateperone is a novel oral antipsychotic indicated for treating adults with schizophrenia. Its unique properties include the lack of presynaptic dopamine D2 antagonism, low D2 receptor occupancy, and the absence of significant extrapyramidal symptoms and metabolic or endocrine adverse effects. In clinical trials, the most frequent adverse event was somnolence/sedation.
Related Resource
- Fulton D. FDA approves Caplyta to treat schizophrenenia in adults. https://www.mdedge.com/psychiatry/article/214733/schizophrenia-other-psychotic-disorders/fda-approves-caplyta-treat.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Cariprazine • Vraylar
Iloperidone • Fanapt
Lumateperone • Caplyta
Lurasidone • Latuda
Olanzapine • Zyprexa
Pimavanserin • Nuplazid
Risperidone • Risperdal
Ziprasidone • Geodon
1. Dufort A, Zipursky RB. Understanding and managing treatment adherence in schizophrenia [published online January 3, 2019]. Clin Schizophr Relat Psychoses. 2019. doi: 10.3371/CSRP.ADRZ.121218.
2. Takeuchi H, Siu C, Remington G, et al. Does relapse contribute to treatment resistance? Antipsychotic response in first- vs. second-episode schizophrenia. Neuropsychopharmacology. 2019;44(6):1036-1042.
3. Dibonaventura M, Gabriel S, Dupclay L, et al. A patient perspective of the impact of medication side effects on adherence: results of a cross-sectional nationwide survey of patients with schizophrenia. BMC Psychiatry. 2012;12:20.
4. Kurokawa S, Kishimoto T, Su K-P, et al. Psychiatrists’ perceptions of medication adherence among patients with schizophrenia: an international survey. Schizophr Res. 2019;211:105-107.
5. Meyer JM. Pharmacotherapy of psychosis and mania. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. 13th ed. Chicago, Illinois: McGraw-Hill; 2018:279-302.
6. Davis RE, Correll CU. ITI-007 in the treatment of schizophrenia: from novel pharmacology to clinical outcomes. Expert Rev Neurother. 2016;16(6):601-614.
7. Caplyta [package Insert]. New York, NY: Intra-Cellular Therapies, Inc.; 2019.
8. Snyder GL, Vanover KE, Zhu H, et al. Functional profile of a novel modulator of serotonin, dopamine, and glutamate neurotransmission. Psychopharmacology (Berl). 2015;232(3):605-621.
9. Vanover KE, Davis RE, Zhou Y, et al. Dopamine D2 receptor occupancy of lumateperone (ITI-007): a positron emission tomography study in patients with schizophrenia. Neuropsychopharmacology. 2019;44(3):598-605.
10. Zhang L, Hendrick JP. The presynaptic D2 partial agonist lumateperone acts as a postsynaptic D2 antagonist. Matters. 2018. doi: 10.19185/matters.201712000006.
11. Meltzer HY, Elkis H, Vanover K, et al. Pimavanserin, a selective serotonin (5-HT)2A-inverse agonist, enhances the efficacy and safety of risperidone, 2mg/day, but does not enhance efficacy of haloperidol, 2mg/day: comparison with reference dose risperidone, 6mg/day. Schizophr Res. 2012;141(2-3):144-152.
12. Nasrallah HA, Fedora R, Morton R. Successful treatment of clozapine-nonresponsive refractory hallucinations and delusions with pimavanserin, a serotonin 5HT-2A receptor inverse agonist. Schizophr Res. 2019;208:217-220.
13. Remington G, Mamo D, Labelle A, et al. A PET study evaluating dopamine D2 receptor occupancy for long-acting injectable risperidone. Am J Psychiatry. 2006;163(3):396-401.
14. Davis RE, Vanover KE, Zhou Y, et al. ITI-007 demonstrates brain occupancy at serotonin 5-HT2A and dopamine D2 receptors and serotonin transporters using positron emission tomography in healthy volunteers. Psychopharmacology (Berl). 2015;232(15):2863-2872.
15. Kumar B, Kuhad A, Kuhad A. Lumateperone: a new treatment approach for neuropsychiatric disorders. Drugs Today (Barc). 2018;54(12):713-719.
16. Vanover K, Glass S, Kozauer S, et al. 30 lumateperone (ITI-007) for the treatment of schizophrenia: overview of placebo-controlled clinical trials and an open-label safety switching study. CNS Spectr. 2019;24(1):190-191.
17. Young SL, Taylor M, Lawrie SM. “First do no harm.” A systematic review of the prevalence and management of antipsychotic adverse effects. J Psychopharmacol. 2015;29(4):353-362.
18. Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394(10202):939-951.
19. Vyas P, Hwang BJ, Brašic ´ JR. An evaluation of lumateperone tosylate for the treatment of schizophrenia. Expert Opin Pharmacother. 2019;1-7.
20. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961.
21. Correll CU, Davis RE, Weingart M, et al. Efficacy and safety of lumateperone for treatment of schizophrenia [published online January 8, 2020]. JAMA Psychiatry. 2020;E1-E10.
1. Dufort A, Zipursky RB. Understanding and managing treatment adherence in schizophrenia [published online January 3, 2019]. Clin Schizophr Relat Psychoses. 2019. doi: 10.3371/CSRP.ADRZ.121218.
2. Takeuchi H, Siu C, Remington G, et al. Does relapse contribute to treatment resistance? Antipsychotic response in first- vs. second-episode schizophrenia. Neuropsychopharmacology. 2019;44(6):1036-1042.
3. Dibonaventura M, Gabriel S, Dupclay L, et al. A patient perspective of the impact of medication side effects on adherence: results of a cross-sectional nationwide survey of patients with schizophrenia. BMC Psychiatry. 2012;12:20.
4. Kurokawa S, Kishimoto T, Su K-P, et al. Psychiatrists’ perceptions of medication adherence among patients with schizophrenia: an international survey. Schizophr Res. 2019;211:105-107.
5. Meyer JM. Pharmacotherapy of psychosis and mania. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. 13th ed. Chicago, Illinois: McGraw-Hill; 2018:279-302.
6. Davis RE, Correll CU. ITI-007 in the treatment of schizophrenia: from novel pharmacology to clinical outcomes. Expert Rev Neurother. 2016;16(6):601-614.
7. Caplyta [package Insert]. New York, NY: Intra-Cellular Therapies, Inc.; 2019.
8. Snyder GL, Vanover KE, Zhu H, et al. Functional profile of a novel modulator of serotonin, dopamine, and glutamate neurotransmission. Psychopharmacology (Berl). 2015;232(3):605-621.
9. Vanover KE, Davis RE, Zhou Y, et al. Dopamine D2 receptor occupancy of lumateperone (ITI-007): a positron emission tomography study in patients with schizophrenia. Neuropsychopharmacology. 2019;44(3):598-605.
10. Zhang L, Hendrick JP. The presynaptic D2 partial agonist lumateperone acts as a postsynaptic D2 antagonist. Matters. 2018. doi: 10.19185/matters.201712000006.
11. Meltzer HY, Elkis H, Vanover K, et al. Pimavanserin, a selective serotonin (5-HT)2A-inverse agonist, enhances the efficacy and safety of risperidone, 2mg/day, but does not enhance efficacy of haloperidol, 2mg/day: comparison with reference dose risperidone, 6mg/day. Schizophr Res. 2012;141(2-3):144-152.
12. Nasrallah HA, Fedora R, Morton R. Successful treatment of clozapine-nonresponsive refractory hallucinations and delusions with pimavanserin, a serotonin 5HT-2A receptor inverse agonist. Schizophr Res. 2019;208:217-220.
13. Remington G, Mamo D, Labelle A, et al. A PET study evaluating dopamine D2 receptor occupancy for long-acting injectable risperidone. Am J Psychiatry. 2006;163(3):396-401.
14. Davis RE, Vanover KE, Zhou Y, et al. ITI-007 demonstrates brain occupancy at serotonin 5-HT2A and dopamine D2 receptors and serotonin transporters using positron emission tomography in healthy volunteers. Psychopharmacology (Berl). 2015;232(15):2863-2872.
15. Kumar B, Kuhad A, Kuhad A. Lumateperone: a new treatment approach for neuropsychiatric disorders. Drugs Today (Barc). 2018;54(12):713-719.
16. Vanover K, Glass S, Kozauer S, et al. 30 lumateperone (ITI-007) for the treatment of schizophrenia: overview of placebo-controlled clinical trials and an open-label safety switching study. CNS Spectr. 2019;24(1):190-191.
17. Young SL, Taylor M, Lawrie SM. “First do no harm.” A systematic review of the prevalence and management of antipsychotic adverse effects. J Psychopharmacol. 2015;29(4):353-362.
18. Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394(10202):939-951.
19. Vyas P, Hwang BJ, Brašic ´ JR. An evaluation of lumateperone tosylate for the treatment of schizophrenia. Expert Opin Pharmacother. 2019;1-7.
20. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961.
21. Correll CU, Davis RE, Weingart M, et al. Efficacy and safety of lumateperone for treatment of schizophrenia [published online January 8, 2020]. JAMA Psychiatry. 2020;E1-E10.