User login
Time to retire haloperidol?
For more than half a century, haloperidol has been used as a first-line medication for psychiatric agitation constituting a “behavioral emergency” when a patient cannot or will not take oral medication. Today, haloperidol is most commonly administered as an IM injection along with an anticholinergic medication to minimize extrapyramidal symptoms (EPS) and a benzodiazepine for additional sedation. The multiple-medication “cocktail” is often referred to by double-entendre nicknames, such as “B-52” or “5250” (ie, haloperidol, 5 mg; lorazepam, 2 mg; and
Earlier evidence of haloperidol’s efficacy
The initial “discovery” of antipsychotic medications was made in 1951 based on the inadvertent observation that chlorpromazine had the potential to calm surgical patients with autonomic activation. This calming effect, described as “désintéressment” (meaning a kind of “indifference to the world”),1 resulted in a new class of medications replacing barbiturates and bromides as go-to options to achieve “rapid tranquilization” of psychiatric agitation.2 Although the ability of antipsychotic medications to gradually reduce positive symptoms, such as delusions and hallucinations, has been attributed to dopamine (D2) antagonism, their more immediate sedating and anti-agitation effects are the result of broader effects as histamine (H1) and alpha-1 adrenergic antagonists.
In the 1970s, haloperidol emerged as a first-line option to manage agitation due to its IM and IV availability, as well as its relative lack of sedation and orthostasis compared with low-potency D2 antagonists such as chlorpromazine. However, haloperidol was observed to have a significant risk of acute EPS, including dystonic reactions.2 From the 1970s to the 1990s, numerous prospective clinical trials of haloperidol for the treatment of acute psychotic agitation, including several randomized controlled trials (RCTs) comparing haloperidol to lorazepam, were conducted.3 The design and outcomes of the haloperidol vs lorazepam RCTs were fairly consistent4-7:
- adult participants with acute agitation and a variety of psychiatric diagnoses, for whom informed consent often was waived due to agitation severity
- randomization to either IM haloperidol, 5 mg, or IM lorazepam, 2 mg, administered every 30 minutes until agitation resolved
- behavioral outcomes measured over several hours using various rating scales, without consistent assessment of EPS
- equivalent efficacy of haloperidol and lorazepam, with symptom resolution usually achieved after 1 to 2 doses (in 30 to 60 minutes), but sometimes longer
- anticholinergic “rescue” allowed for EPS, but not administered prophylactically
- EPS, including dystonia and akathisia, were significantly more frequent with haloperidol compared with lorazepam.8
In recognition of the greater risk of EPS with haloperidol compared with lorazepam, and the fact that most study participants were already taking standing doses of antipsychotic medications, some researchers have recommended using benzodiazepines alone as the optimal treatment for agitation.4,9 A 2012 Cochrane review concluded that the involuntary use of haloperidol alone “could be considered unethical.”10,11 However, other studies that examined the combination of haloperidol and lorazepam compared with either medication alone found that the combination of the 2 medications was associated with a more rapid resolution of symptoms, which suggests a superior synergistic effect.6,7,12 By the late 1990s, combined haloperidol and lorazepam, often mixed within a single injection, became the most common strategy to achieve rapid tranquilization in the psychiatric emergency setting.13 However, while the combination has been justified as a way to reduce the antipsychotic medication dose and EPS risk,2 few studies have compared combinations containing <5 mg of haloperidol. As a result, the apparent superiority of combined haloperidol and lorazepam compared with either medication alone may be a simple cumulative dose effect rather than true synergism. It is also important to note that adding lorazepam to haloperidol does not mitigate the risk of EPS such as dystonia in the absence of anticholinergic medication.8 To date, however, there have been no clinical trials investigating the efficacy of IM haloperidol, lorazepam, and
Newer RCTs tell a different story
With the availability of second-generation antipsychotics (SGAs) in IM formulations, clinical trials over the past 2 decades have focused on comparing SGAs with haloperidol alone as the “gold standard” control for acute agitation. Compared with previous trials of
- Study participants who signed informed consent (and were likely less agitated)
- IM haloperidol doses typically >5 mg (eg, 6.5 to 10 mg).
As with studies comparing lorazepam with haloperidol, the results of these RCTs revealed that IM
An updated 2017 Cochrane review of haloperidol for psychosis-induced aggression or agitation concluded that9:
- haloperidol is an effective intervention, although the evidence is “weak”
- significant treatment effects may take as long as 1 to 2 hours following multiple IM injections
- in contrast to SGAs, treatment with haloperidol carries a significant risk of EPS
- adding a benzodiazepine “does not have strong evidence of benefit and carries risk of additional harm.”
Continue to: Haloperidol's well-known toxicity
Haloperidol’s well-known toxicity
Haloperidol has been associated with numerous adverse effects:
Akathisia and other acute EPS. Treatment with even a single dose of IM haloperidol can result in acute EPS, including dystonia and akathisia. At best, such adverse effects are subjectively troubling and unpleasant; at worst, akathisia can exacerbate and be mistaken for agitation, leading to administration of more medication23 and the possible development of suicidal or violent behavior.24-25 In the studies reviewed above, the overall rate of EPS was as high as 21% after treatment with haloperidol,16 with parkinsonism occurring in up to 17% of patients,19 dystonia in up to 11%,7 and akathisia in up to 10%.15 However, because specific EPS were assessed inconsistently, and sometimes not at all, the rate of akathisia—arguably the most relevant and counter-therapeutic adverse effect related to agitation—remains unclear.
In another study that specifically assessed for akathisia in patients treated with haloperidol, up to 40% experienced akathisia 6 hours after a single oral dose of 5 mg.26 Even a single dose of IV
Although anticholinergic medications or benzodiazepinesare often administered as part of a haloperidol “cocktail,” these medications often do not adequately resolve emergent akathisia.26,28 No clinical trials of IM haloperidol combined with benztropine or diphenhydramine have been published, but several studies suggest that combining haloperidol with
Cardiotoxicity. Although low-potency antipsychotic medications such as
Continue to: Although there is no direct evidence...
Although there is no direct evidence that the cardiac risks associated with IV haloperidol apply to IM administration, epidemiologic studies indicate that oral haloperidol carries an elevated risk of ventricular arrhythmia and sudden cardiac death,35,36 with 1 study reporting greater risk compared with other SGAs.37 Haloperidol, whether administered orally or IM, may therefore be an especially poor choice for patients with agitation who are at risk for arrhythmia, including those with relevant medical comorbidities or delirium.34
Neuronal cell death. Several lines of research evidence have demonstrated that haloperidol can cause cellular injury or death in neuronal tissue in a dose-dependent fashion through a variety of mechanisms.38 By contrast, SGAs have been shown to have neuroprotective effects.39 While these findings have mostly come from studies conducted in animals or in vitro human tumor cell lines, some researchers have nonetheless called for haloperidol to be banned, noting that if its neurotoxic effects were more widely known, “we would realize what a travesty it is to use [such] a brain-unfriendly drug.”40
Several reasonable alternatives
Echoing the earlier Cochrane review of haloperidol for psychosis-induced aggression or agitation,10 a 2017 update concluded, “If no other alternative exists, sole use of intramuscular haloperidol could be life-saving. Where additional drugs are available, sole use of haloperidol for extreme emergency could be considered unethical.”9
What then are reasonable alternatives to replace IM haloperidol for agitation? Clinicians should consider the following nonpharmacologic and pharmacologic interventions:
Nonpharmacologic interventions. Several behavioral interventions have been demonstrated to be effective for managing acute agitation, including verbal de-escalation, enhanced “programming” on the inpatient units, and the judicious use of seclusion.41-43 While such interventions may demand additional staff or resources, they have the potential to lower long-term costs, reduce injuries to patients and staff, and improve the quality of care.43 The use of IM haloperidol as a form of “chemical restraint” does not represent standard-of-care treatment,3 and from an ethical perspective, should never be implemented punitively or to compensate for substandard care in the form of inadequate staffing or staff training.
Continue to: Benzodiazepines
Benzodiazepines. Lorazepam offers an attractive alternative to haloperidol without the risk of EPS.2,4,8 However, lorazepam alone may be perceived as less efficacious than a haloperidol “cocktail” because it represents less overall medication. Some evidence has suggested that lorazepam, 4 mg, might be the most appropriate dose, although it has only rarely been studied in clinical trials of acute agitation.3
Respiratory depression is frequently cited as an argument against using lorazepam for agitation, as if the therapeutic window is extremely narrow with ineffectiveness at 2 mg, but potential lethality beyond that dose. In fact, serious respiratory depression with lorazepam is unlikely in the absence of chronic obstructive pulmonary disease (COPD), obstructive sleep apnea, or concomitant alcohol or other sedative use.46 Case reports have documented therapeutic lorazepam dosing of 2 to 4 mg every 2 hours up to 20 to 30 mg/d in patients with manic agitation.47 Even in patients with COPD, significant respiratory depression tends not to occur at doses <8 mg.48 A more evidence-based concern about lorazepam dosing is that 2 mg might be ineffective in patients with established tolerance. For example, 1 report described a patient in acute alcohol withdrawal who required dosing lorazepam to 1,600 mg within 24 hours.49 Collectively, these reports suggest that lorazepam has a much wider therapeutic window than is typically perceived, and that dosing with 3 to 4 mg IM is a reasonable option for agitation when 2 mg is likely to be inadequate.
Paradoxical disinhibition is another concern that might prevent benzodiazepines from being used alone as a first-line intervention for emergency treatment of agitation. However, similar to respiratory depression, this adverse event is relatively rare and tends to occur in children and geriatric patients, individuals intoxicated with alcohol or other sedatives, and patients with brain injury, developmental delay, or dementia.23,46 Although exacerbation of aggression has not been demonstrated in the RCTs examining benzodiazepines for agitation reviewed above, based on other research, some clinicians have expressed concerns about the potential for benzodiazepines to exacerbate aggression in patients with impulse control disorders and a history of violent behavior.50
The 2005 Expert Consensus Panel for Behavioral Emergencies51 recommended the use of lorazepam alone over haloperidol for agitation for patients for whom the diagnosis is unknown or includes the following:
- stimulant intoxication
- personality disorder
- comorbid obesity
- comorbid cardiac arrhythmia
- a history of akathisia and other EPS
- a history of amenorrhea/galactorrhea
- a history of seizures.
In surveys, patients have ranked lorazepam as the preferred medication for emergency agitation, whereas haloperidol was ranked as one of the least-preferred options.51,52
Continue to: Second-generation antipsychotics
Second-generation antipsychotics. The SGAs available in IM formulations, such as aripiprazole, olanzapine, and ziprasidone, have been shown to be at least as effective as haloperidol for the treatment of acute agitation (in 2015, the short-acting injectable formulation of aripiprazole was discontinued in the United States independent of safety or efficacy issues53). A review of RCTs examining IM SGAs for the treatment of agitation concluded that the number needed to treat for response compared with placebo was 5 for aripiprazole, 3 for olanzapine, and 3 for ziprasidone.54 In terms of safety, a meta-analysis of studies examining IM medications for agitation confirmed that the risk of acute EPS, including dystonia, akathisia, and parkinsonism, is significantly lower with SGAs compared with haloperidol.55 An RCT comparing IM ziprasidone with haloperidol found equivalently modest effects on QTc prolongation.56 Therefore, SGAs are an obvious and evidence-based option for replacing haloperidol as a treatment for acute agitation.
Unfortunately, for clinicians hoping to replace haloperidol within a multiple-medication IM “cocktail,” there have been no published controlled trials of SGAs combined with benzodiazepines. Although a short report indicated that aripiprazole and lorazepam are chemically compatible to be combined within a single injection,57 the package insert for aripiprazole warns that “If parenteral benzodiazepine therapy is deemed necessary in addition to ABILIFY injection treatment, patients should be monitored for excessive sedation and for orthostatic hypotension.”58 The package insert for olanzapine likewise lists the combination of lorazepam and olanzapine as a drug interaction that can potentiate sedation, and the manufacturer issued specific warnings about parenteral combination.59,60 A single published case of significant hypotension with combined IM olanzapine and lorazepam,60 together with the fact that IM olanzapine can cause hypotension by itself,61 has discouraged the coadministration of these medications. Nonetheless, the combination is used in some emergency settings, with several retrospective studies failing to provide evidence of hypotension or respiratory depression as adverse effects.62-64
Droperidol.
Over the past decade, however, droperidol has returned to the US market68 and its IV and IM usage has been revitalized for managing patients with agitation within or en route to the ED. Studies have demonstrated droperidol efficacy comparable to midazolam, ziprasidone, or olanzapine, as well as effectiveness as an IV adjunct to midazolam.69-71 In contrast to the FDA black-box warning, retrospective studies and RCTs of both IV and IM droperidol suggest that QTc prolongation and torsades de pointes are rare events that do not occur any more frequently than they do with haloperidol, even at doses >10 mg.72,73 However, in studies involving patients with drug intoxication and treatment with multiple medications, oversedation to the point of needing rescue intervention was reported. In an emergency setting where these issues are relatively easily managed, such risks may be better tolerated than in psychiatric settings.
With earlier studies examining the use of droperidol in an acute psychiatric setting that reported a more rapid onset of action than haloperidol,65-67 a 2016 Cochrane review concluded that there was high-quality evidence to support droperidol’s use for psychosis-induced agitation.74 However, a 2015 RCT comparing IM droperidol, 10 mg, to haloperidol, 10 mg, found equivalent efficacy and response times (with maximal response occurring within 2 hours) and concluded that droperidol had no advantage over haloperidol.75 Because none of the clinical trials that evaluated droperidol have included assessments for EPS, its risk of akathisia remains uncertain.
Continue to: Ketamine
Ketamine. In recent years, ketamine has been used to treat acute agitation within or en route to the ED. Preliminary observational studies support ketamine’s efficacy when administered via IV or IM routes,76 with more rapid symptomatic improvement compared with haloperidol, lorazepam, or midazolam alone.77 Reported adverse effects of ketamine include dissociation, psychotic exacerbation, and respiratory depression,76 although 1 small naturalistic study found no evidence of exacerbation of psychotic or other psychiatric symptoms.78 An ongoing RCT is comparing IM ketamine, 5 mg/kg, to combined IM haloperidol, 5 mg, and midazolam, 5 mg.79 Although various ketamine formulations are increasingly being used in psychiatry, active psychosis is generally regarded as a contraindication. It is premature to recommend parenteral ketamine administration for agitation within most psychiatric settings until more research on safety has been completed.
Haloperidol, or something else? Practical considerations
Consider the following factors when deciding whether to use haloperidol or one of its alternatives:
Limitations of the evidence. Modern clinical trials requiring informed consent often do not include the kind of severe agitation that clinicians encounter in acute psychiatric, emergency, or forensic settings. In addition, standard interventions, such as 3-medication haloperidol “cocktails,” have not been evaluated in clinical trials. Clinicians are therefore often in the dark about optimal evidence-based practices.
Treatment goals. Psychiatric agitation has many causes, with a range of severity that warrants a commensurate range of responses. Protocols for managing acute agitation should include graded interventions that begin with nonpharmacologic interventions and voluntary oral medications, and move to involuntary IM medications when necessary.
While treatment guidelines clearly recommend against IM medications as “chemical restraint” with a goal of sedating a patient until he/she is unconscious,3,51 such outcomes are nonetheless often sought by staff who are concerned about the risk of injuries during a behavioral emergency. In such instances, the risks of violence towards patients and staff may outweigh concerns about adverse effects in a risk-benefit analysis. Consequently, clinicians may be prone to “skip over” graded interventions because they assume they “won’t work” in favor of administering involuntary multiple-medication haloperidol “cocktails” despite risks of excess sedation, EPS, and cardiotoxicity. Treatment settings should critically evaluate such biased preferences, with a goal of developing tailored, evidence-based strategies that maximize benefits while minimizing excess sedation and other untoward adverse effects, with an eye towards promoting better overall patient care and reducing length of stay.42,43,80
Continue to: Limitations of available medications
Limitations of available medications. There is no perfect medication for the management of acute agitation. Evidence indicates that pharmacologic options take 15 minutes to several hours to resolve acute agitation, even potentially more rapid-acting medications such as midazolam and droperidol. This is well beyond most clinicians’ desired window for response time in a behavioral emergency. Multiple-medication “cocktails” may be used with the hope of hastening response time, but may not achieve this goal at the expense of increasing the risk of adverse effects and the likelihood that a patient will remain sedated for a prolonged time. In the real world, this often means that by the time a psychiatrist comes to evaluate a patient who has been given emergency medications, the patient cannot be aroused for an interview. Ideally, medications would calm an agitated patient rapidly, without excess or prolonged sedation.80 Less-sedating SGAs, such as ziprasidone, might have this potential, but can sometimes be perceived as ineffective.
Avoiding akathisia. Akathisia’s potential to worsen and be mistaken for agitation makes it an especially concerning, if underappreciated, adverse effect of haloperidol that is often not adequately assessed in clinical trials or practice. In light of evidence that akathisia can occur in nearly half of patients receiving a single 5 mg-dose of haloperidol, it is difficult to justify the use of this medication for agitation when equally effective options exist with a lower risk of EPS.
While haloperidol-induced akathisia could in theory be mitigated by adding anticholinergic medications or benzodiazepines, previous studies have found that such strategies have limited effectiveness compared to “gold standard” treatment with propranolol.28,81,82 Furthermore, the half-lives of anticholinergic medications, such as benztropine or diphenhydramine, are significantly shorter than that of a single dose of haloperidol, which can be as long as 37 hours.83 Therefore, akathisia and other EPS could emerge or worsen several hours or even days after receiving an IM haloperidol “cocktail” as the shorter-acting medications wear off. Akathisia is best minimized by avoiding FGAs, such as haloperidol, when treating acute agitation.
Promoting adherence. Although haloperidol is often recommended for acute agitation in patients with schizophrenia or bipolar disorder on the basis that it would treat the underlying condition, many patients who receive IM medications for acute agitation are already prescribed standing doses of oral medication, which increases the risk of cumulative toxicity. In addition, receiving a medication likely to cause acute EPS that is ranked near the bottom of patient preferences may erode the potential for a therapeutic alliance and hamper longer-term antipsychotic medication adherence.

Time for a change
For nearly half a century, haloperidol has been a “gold standard” intervention for IM control in patients with agitation. However, given its potential to produce adverse effects, including a significant risk of akathisia that can worsen agitation, along with the availability of newer pharmacologic options that are at least as effective (Table 1, and Table 2), haloperidol should be retired as a first-line medication for the treatment of agitation. Clinicians would benefit from RCTs investigating the safety and efficacy of novel interventions including frequently-used, but untested medication combinations, as well as nonpharmacologic interventions.

Continue to: Bottom Line
Bottom Line
Although there is no perfect IM medication to treat acute agitation, haloperidol’s higher risk of adverse effects relative to newer alternatives suggest that it should no longer be considered a first-line intervention.
Related Resources
- Zun LS. Evidence-based review of pharmacotherapy for acute agitation. Part 1: onset of efficacy. J Emerg Med. 2018;54(3):364-374.
- Zun LS. Evidence-based review of pharmacotherapy for acute agitation. Part 2: safety. J Emerg Med. 2018;54(4): 522-532.
Drug Brand Names
Aripiprazole • Abilify
Benztropine • Cogentin
Chlorpromazine • Thorazine
Diphenhydramine • Benadryl
Droperidol • Inapsine
Haloperidol • Haldol
Ketamine • Ketalar
Lorazepam • Ativan
Midazolam • Versed
Olanzapine • Zyprexa
Prochlorperazine • Compazine
Promethazine • Phenergan
Propranolol • Inderal, Pronol
Ziprasidone • Geodon
1. Shorter E. A history of psychiatry. New York, NY: John Wiley & Sons, Inc.; 1997:249.
2. Salzman C, Green AI, Rodriguez-Villa F, et al. Benzodiazepines combined with neuroleptics for management of severe disruptive behavior. Psychosomatics. 1986;27(suppl 1):17-22.
3. Allen MH. Managing the agitated psychotic patient: a reappraisal of the evidence. J Clin Psychiatr. 2000;61(suppl 14):11-20.
4. Salzman C, Solomon D, Miyawaki E, et al. Parenteral lorazepam versus parenteral haloperidol for the control of psychotic disruptive behavior. J Clin Psychiatr. 1991:52(4):177-180.
5. Allen MH, Currier GW, Hughes DH, et al. The expert consensus guideline series: treatment of behavioral emergencies. Postgrad Med. 2001;(Spec No):1-88; quiz 89-90.
6. Foster S, Kessel J, Berman ME, et al. Efficacy of lorazepam and haloperidol for rapid tranquilization in a psychiatric emergency room setting. Int Clin Psychopharmacol. 1997;12(3):175-179.
7. Garza-Trevino WS, Hollister LE, Overall JE, et al. Efficacy of combinations of intramuscular antipsychotics and sedative-hypnotics for control of psychotic agitation. Am J Psychiatr. 1989:146(12):1598-1601.
8. Battaglia J, Moss S, Rush J, et al. Haloperidol, lorazepam, or both for psychotic agitation? A multicenter, prospective double-blind, emergency study. Am J Emerg Med 1997;15(4):335-340.
9. Ostinelli EG, Brooke-Powney MJ, Li X, et al. Haloperidol for psychosis-induced aggression or agitation (rapid tranquillisation). Cochrane Database Syst Rev. 2017; 7:CD009377. doi: 10.1002/14651858.CD009377.pub3.
10. Powney MJ, Adams CE, Jones H. Haloperidol for psychosis-induced aggression or agitation (rapid tranquillisation). Cochrane Database Syst Rev. 2012;11:CD009377. doi: 10.1002/14651858.CD009377.pub2.
11. Citrome L. Review: limited evidence on effects of haloperidol alone for rapid tranquillisation in psychosis-induced aggression. Evid Based Ment Health. 2013;16(2):47.
12. Bienek SA, Ownby R, Penalver A, et al. A double-blind study of lorazepam versus the combination of haloperidol and lorazepam in managing agitation. Pharmacother. 1998;18(1):57-62.
13. Binder RL, McNiel DE. Contemporary practices in managing acutely violent patients in 20 psychiatric emergency rooms. Psychiatric Serv. 1999;50(2):1553-1554.
14. Andrezina R, Josiassen RC, Marcus RN, et al. Intramuscular aripiprazole for the treatment of acute agitation in patients with schizophrenia or schizoaffective disorder: a double-blind, placebo-controlled comparison with intramuscular haloperidol. Psychopharmacology (Berl). 2006;188(3):281-292.
15. Tran-Johnson TK, Sack DA, Marcus RN, et al. Efficacy and safety of intramuscular aripiprazole in patients with acute agitation: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatr. 2007;68(1):111-119.
16. Brook S, Lucey JV, Gunn KP. Intramuscular ziprasidone compared with intramuscular haloperidol in the treatment of acute psychosis. J Clin Psychiatr. 2000;61(12):933-941.
17. Brook S, Walden J, Benattia I, et al. Ziprasidone and haloperidol in the treatment of acute exacerbation of schizophrenia and schizoaffective disorder: comparison of intramuscular and oral formulations in a 6-week, randomized, blinded-assessment study. Psychopharmacology (Berl). 2005;178(4):514-523.
18. Wright P, Birkett M, David SR, et al. Double-blind, placebo-controlled comparison of intramuscular olanzapine and intramuscular haloperidol in the treatment of acute agitation in schizophrenia. Am J Psychiatr. 2001;158(7):1149-1151.
19. Breier A, Meehan K, Birkett M, et al. A double-blind, placebo-controlled dose-response comparison of intramuscular olanzapine and haloperidol in the treatment of acute agitation in schizophrenia. Arch Gen Psych. 2002;59(5):441-448.
20. Hsu W, Huang S, Lee B, et al. Comparison of intramuscular olanzapine, orally disintegrating olanzapine tablets, oral risperidone solution, and intramuscular haloperidol in the management of acute agitation in an acute care psychiatric ward in Taiwan. J Clin Psychopharmacol. 2010;30(3):230-234.
21. Chan H, Ree S, Su L, et al. A double-blind, randomized comparison study of efficacy and safety of intramuscular olanzapine and intramuscular haloperidol in patients with schizophrenia and acute agitated behavior. J Clin Psychopharmacol. 2014;34(3):355-358.
22. Baldaçara L, Sanches M, Cordeiro DC, et al. Rapid tranquilization for agitated patients in emergency psychiatric rooms: a randomized trial of olanzapine, ziprasidone, haloperidol plus promethazine, haloperidol plus midazolam and haloperidol alone. Braz J Psychiatry. 2011;33(1):30-39.
23. Hillard JR. Defusing patient violence. Current Psychiatry. 2002;1(4):22-29.
24. Seemüller F, Schennach R, Mayr A, et al. Akathisia and suicidal ideation in first-episode schizophrenia. J Clin Psychopharmacol. 2012;32(5):694-698.
25. Eikelenboom-Schieveld SJM, Lucire Y, Fogleman JC. The relevance of cytochrome P450 polymorphism in forensic medicine and akathisia-related violence and suicide. J Forens Leg Med. 2016;41:65-71.
26. Van Putten T, May PRA, Marder SR. Akathisia with haloperidol and thiothixene. Arch Gen Psych. 1984;41:1036-1039.
27. Drotts DL, Vinson DR. Prochlorperazine induced akathisia in emergency patients. Ann Emerg Med. 1999;34(4):469-475.
28. Salem H, Negpal C, Pigott T. Revisiting antipsychotic-induced akathisia: current issues and prospective challenges. Curr Neuropharmacol. 2017;15(5):789-798.
29. Huf G, Coutinho ESF, Adams CE. Rapid tranquilization in psychiatric emergency settings in Brazil: pragmatic randomized controlled trial of intramuscular haloperidol versus intramuscular haloperidol plus promethazine. BMJ. 2007;335(7625):869.
30. Mantovani C, Labate CM, Sponholz A, et al. Are low doses of antipsychotics effective in the management of psychomotor agitation? A randomized, rated-blind trial of 4 intramuscular interventions. J Clin Psychopharmacol. 2013;33(3):306-312.
31. Darwish H, Grant R, Haslam R, et al. Promethazine-induced acute dystonic reactions. Am J Dis Child. 1980;134(10):990-991.
32. Jyothi CH, Rudraiah HGM, Vidya HK, et al. Promethazine induced acute dystonia: a case report. Manipal J Med Sci. 2016;1(2):63-64.
33. Ames D, Carr-Lopez SM, Gutierrez MA, et al. Detecting and managing adverse effects of antipsychotic medications: current state of play. Psychiatr Clin North Am. 2016;39(2):275-311.
34. Meyer-Massetti C, Cheng CM, Sharpe MA, et al. The FDA extended warning for intravenous haloperidol and torsades de pointes: how should institutions respond? J Hosp Med. 2010;5(4):E8-E16. doi: 10.1002/jhm.691.
35. Wu C, Tsai Y, Tsai H. Antipsychotic drugs and the risk of ventricular arrhythmia and/or sudden cardiac death: a nation-wide case-crossover study. J Am Heart Dis. 2015;4(2):e001568. doi: 10.1161/JAHA.114.001568.
36. Beach SR, Celano CM, Sugrue AM, et al. QT prolongation, torsades de pointe, and psychotropic medications: a 5-year update. Psychosomatics. 2018;59(1):105-122.
37. Leonard CE, Freeman CP, Newcomb CW, et al. Antipsychotics and the risks of sudden cardiac death and all-cause death: cohort studies in Medicaid and dually-eligible Medicaid-Medicare beneficiaries of five states. J Clin Exp Cardiol. 2013;suppl 10(6):1-9.
38. Nasrallah H, Chen AT. Multiple neurotoxic effects of haloperidol resulting in neuronal death. Ann Clin Psychiatr. 2017;29(3):195-202.
39. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
40. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2013;12(7):7-8.
41. Corrigan PW, Yudofsky SC, Silver JM. Pharmacological and behavioral treatments for aggressive psychiatric inpatients. Hosp Comm Psychiatr. 1993;44(2):125-133.
42. Zeller SL, Citrome L. Managing agitation associated with schizophrenia and bipolar disorder in the emergency setting. West J Emerg Med. 2016;17(2):165-172.
43. Vieta E, Garriga M, Cardete L, et al. Protocol for the management of psychiatric patients with psychomotor agitation. BMC Psychiatr. 2017;17:328.
44. Nobay F, Simon BC, Levitt A, et al. A prospective, double-blind, randomized trial of midazolam versus haloperidol versus lorazepam in the chemical restraint of violent and severely agitated patients. Acad Emerg Med. 2004;11(7):744-749.
45. Klein LR, Driver BE, Miner JR, et al. Intramuscular midazolam, olanzapine, ziprasidone, or haloperidol for treating acute agitation in the emergency department. Ann Emerg Med. 2018;72(4):374-385.
46. Hillard JR. Emergency treatment of acute psychosis. J Clin Psychiatr. 1998;59(suppl 1):57-60.
47. Modell JG, Lenox RH, Weiner S. Inpatient clinical trial of lorazepam for the management of manic agitation. J Clin Psychopharmacol. 1985;5(2):109-110.
48. Denaut M, Yernault JC, De Coster A. Double-blind comparison of the respiratory effects of parenteral lorazepam and diazepam in patients with chronic obstructive lung disease. Curr Med Res Opin. 1975;2(10):611-615.
49. Kahn DR, Barnhorst AV, Bourgeois JA. A case of alcohol withdrawal requiring 1,600 mg of lorazepam in 24 hours. CNS Spectr. 2009;14(7):385-389.
50. Jones KA. Benzodiazepines: their role in aggression and why GPs should prescribe with caution. Austral Fam Physician. 2011;40(11):862-865.
51. Allen MH, Currier GW, Carpenter D, et al. The expert consensus guideline series. Treatment of behavioral emergencies 2005. J Psychiatr Pract. 2005;11(suppl 1):5-108.
52. Allen MH, Carpenter D, Sheets JL, et al. What do consumers say they want and need during a psychiatric emergency? J Psychiatr Pract. 2003;9(1):39-58.
53. Han DH. Some Abilify formulations to discontinue in 2015. MPR. https://www.empr.com/home/news/some-abilify-formulations-to-discontinue-in-2015/. Published January 13, 2015. Accessed April 17, 2020.
54. Citrome L. Comparison of intramuscular ziprasidone, olanzapine, or aripiprazole for agitation: a quantitative review of efficacy and safety. J Clin Psychiatry. 2007;68(12):1876-1885.
55. Satterthwaite TD, Wolf DH, Rosenheck RA, et al. A meta-analysis of the risk of acute extrapyramidal symptoms with intramuscular antipsychotics for the treatment for agitation. J Clin Psychiatr. 2008;69(12):1869-1879.
56. Miceli JJ, Tensfeldt TG, Shiovitz T, et al. Effects of high-dose ziprasidone and haloperidol on the QTc interval after intramuscular administration: a randomized, single-blind, parallel-group study in patients with schizophrenia or schizoaffective disorder. Clin Ther. 2010;32(3):472-491.
57. Kovalick LJ, Pikalov AA, Ni N, et al. Short-term physical compatibility of intramuscular aripiprazole with intramuscular lorazepam. Am J Health-Syst Pharm. 2008;65(21):2007-2008.
58. Abilify [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2014.
59. Zyprexa [package insert]. Indianapolis, IN: Lilly Research Laboratories; 2005.
60. Zacher JL, Roche-Desilets J. Hypotension secondary to the combination of intramuscular olanzapine and intramuscular lorazepam. J Clin Psychiatr. 2005;66(12):1614-1615.
61. Marder SR, Sorsaburu S, Dunayevich E, et al. Case reports of postmarketing adverse event experiences with olanzapine intramuscular treatment in patients with agitation. J Clin Psychiatr 2010;71(4):433-441.
62. Wilson MP, MacDonald K, Vilke GM, et al. A comparison of the safety of olanzapine and haloperidol in combination with benzodiazepines in emergency department patients with acute agitation. J Emerg Med. 2012;43(5):790-797.
63. Wilson MP, MacDonald K, Vilke GM, et al. Potential complications of combining intramuscular olanzapine with benzodiazepines in emergency department patients. J Emerg Med. 2012;43(5):889-896.
64. Williams AM. Coadministration of intramuscular olanzapine and benzodiazepines in agitated patients with mental illness. Ment Health Clin [Internet]. 2018;8(5):208-213.
65. Resnick M, Burton BT. Droperidol vs. haloperidol in the initial management of acutely agitated patients. J Clin Psychiatry. 1984;45(7):298-299.
66. Thomas H, Schwartz E, Petrilli R. Droperidol versus haloperidol for chemical restraint of agitated and combative patients. Ann Emerg Med. 1992;21(4):407-413.
67. Richards JR, Derlet RW, Duncan DR. Chemical restraint for the agitated patient in the emergency department: lorazepam versus droperidol. J Emerg Med. 1998;16(4):567-573.
68. Boyer EW. Droperidol is back (and here’s what you need to know). ACEP Now. https://www.acepnow.com/article/droperidol-is-back-and-heres-what-you-need-to-know/. Published September 16, 2019. Accessed April 17, 2020.
69. Martel M, Sterzinger A, Miner J, et al. Management of acute undifferentiated agitation in the emergency department: a randomized double-blind trial of droperidol, ziprasidone, and midazolam. Acad Emerg Med. 2005;12(12):1167-1172.
70. Chan EW, Taylor DM, Knott JC, et al. Intravenous droperidol or olanzapine as an adjunct to midazolam for the acutely agitated patient: a multicenter, randomized, double-blind, placebo-controlled clinical trial. Ann Emerg Med. 2013;61(1):72-81.
71. Isbister GK, Calver LA, Page CB, et al. Randomized controlled trial of intramuscular droperidol versus midazolam for violence and acute behavioral disturbance: the DORM study. Ann Emerg Med. 2010;56(4):392-401.
72. Macht M, Mull AC, McVaney KE, et al. Comparison of droperidol and haloperidol for use by paramedics assessment of safety and effectiveness. Prehosp Emerg Care. 2014;18(3):375-380.
73. Calver L, Page CB, Downes MA, et al. The safety and effectiveness of droperidol for sedation of acute behavioral disturbance in the emergency department. Ann Emerg Med. 2015;66(3):230-238.
74. Kohokar MA, Rathbone J. Droperidol for psychosis-induced aggression or agitation. Cochrane Database Syst Rev. 2016;12:CD002830.
75. Calver L, Drinkwater V, Gupta R, et al. Droperidol v. haloperidol for sedation of aggressive behavior in acute mental health: randomized controlled trial. Brit J Psychiatr. 2015;206(3):223-228.
76. Hopper AB, Vilke GM, Castillo EM, et al. Ketamine use for acute agitation in the emergency department. J Emerg Med. 2015;48(6):712-719.
77. Riddell J, Tran A, Bengiamin R, et al. Ketamine as a first-line treatment for severely agitated emergency department patients. Am J Emerg Med. 2017;35:1000-1004.
78. Lebin JA, Akhavan AR, Hippe DS, et al. Psychiatric outcomes of patients with severe agitation following administration of prehospital ketamine. Acad Emerg Med. 2019;26(8):889-896.
79. Barbic D, Andolfatto G, Grunau B, et al. Rapid agitation control with ketamine in the emergency department (RACKED): a randomized controlled trial protocol. Trials. 2018;19(1):651.
80. Garriga M, Pacchiarotti I, Kasper S, et al. Assessment and management of agitation in psychiatry: expert consensus. World J Biol Psychiatr. 2016;17(2):86-128.
81. Adler L, Angrist B, Peselow E, et al. Efficacy of propranolol in neuroleptic-induced akathesia. J Clin Psychopharmacol. 1985;5(3):164-166.
82. Adler LA, Reiter S, Corwin J, et al. Neuroleptic-induced akathisia: propranolol versus benztropine. Biol Psychiatry. 1988;23(2):211-213.
83. de Leon J, Diaz FJ, Wedlund P, et al. Haloperidol half-life after chronic dosing. J Clin Psychopharmacol. 2004;24(6):656-660.
For more than half a century, haloperidol has been used as a first-line medication for psychiatric agitation constituting a “behavioral emergency” when a patient cannot or will not take oral medication. Today, haloperidol is most commonly administered as an IM injection along with an anticholinergic medication to minimize extrapyramidal symptoms (EPS) and a benzodiazepine for additional sedation. The multiple-medication “cocktail” is often referred to by double-entendre nicknames, such as “B-52” or “5250” (ie, haloperidol, 5 mg; lorazepam, 2 mg; and
Earlier evidence of haloperidol’s efficacy
The initial “discovery” of antipsychotic medications was made in 1951 based on the inadvertent observation that chlorpromazine had the potential to calm surgical patients with autonomic activation. This calming effect, described as “désintéressment” (meaning a kind of “indifference to the world”),1 resulted in a new class of medications replacing barbiturates and bromides as go-to options to achieve “rapid tranquilization” of psychiatric agitation.2 Although the ability of antipsychotic medications to gradually reduce positive symptoms, such as delusions and hallucinations, has been attributed to dopamine (D2) antagonism, their more immediate sedating and anti-agitation effects are the result of broader effects as histamine (H1) and alpha-1 adrenergic antagonists.
In the 1970s, haloperidol emerged as a first-line option to manage agitation due to its IM and IV availability, as well as its relative lack of sedation and orthostasis compared with low-potency D2 antagonists such as chlorpromazine. However, haloperidol was observed to have a significant risk of acute EPS, including dystonic reactions.2 From the 1970s to the 1990s, numerous prospective clinical trials of haloperidol for the treatment of acute psychotic agitation, including several randomized controlled trials (RCTs) comparing haloperidol to lorazepam, were conducted.3 The design and outcomes of the haloperidol vs lorazepam RCTs were fairly consistent4-7:
- adult participants with acute agitation and a variety of psychiatric diagnoses, for whom informed consent often was waived due to agitation severity
- randomization to either IM haloperidol, 5 mg, or IM lorazepam, 2 mg, administered every 30 minutes until agitation resolved
- behavioral outcomes measured over several hours using various rating scales, without consistent assessment of EPS
- equivalent efficacy of haloperidol and lorazepam, with symptom resolution usually achieved after 1 to 2 doses (in 30 to 60 minutes), but sometimes longer
- anticholinergic “rescue” allowed for EPS, but not administered prophylactically
- EPS, including dystonia and akathisia, were significantly more frequent with haloperidol compared with lorazepam.8
In recognition of the greater risk of EPS with haloperidol compared with lorazepam, and the fact that most study participants were already taking standing doses of antipsychotic medications, some researchers have recommended using benzodiazepines alone as the optimal treatment for agitation.4,9 A 2012 Cochrane review concluded that the involuntary use of haloperidol alone “could be considered unethical.”10,11 However, other studies that examined the combination of haloperidol and lorazepam compared with either medication alone found that the combination of the 2 medications was associated with a more rapid resolution of symptoms, which suggests a superior synergistic effect.6,7,12 By the late 1990s, combined haloperidol and lorazepam, often mixed within a single injection, became the most common strategy to achieve rapid tranquilization in the psychiatric emergency setting.13 However, while the combination has been justified as a way to reduce the antipsychotic medication dose and EPS risk,2 few studies have compared combinations containing <5 mg of haloperidol. As a result, the apparent superiority of combined haloperidol and lorazepam compared with either medication alone may be a simple cumulative dose effect rather than true synergism. It is also important to note that adding lorazepam to haloperidol does not mitigate the risk of EPS such as dystonia in the absence of anticholinergic medication.8 To date, however, there have been no clinical trials investigating the efficacy of IM haloperidol, lorazepam, and
Newer RCTs tell a different story
With the availability of second-generation antipsychotics (SGAs) in IM formulations, clinical trials over the past 2 decades have focused on comparing SGAs with haloperidol alone as the “gold standard” control for acute agitation. Compared with previous trials of
- Study participants who signed informed consent (and were likely less agitated)
- IM haloperidol doses typically >5 mg (eg, 6.5 to 10 mg).
As with studies comparing lorazepam with haloperidol, the results of these RCTs revealed that IM
An updated 2017 Cochrane review of haloperidol for psychosis-induced aggression or agitation concluded that9:
- haloperidol is an effective intervention, although the evidence is “weak”
- significant treatment effects may take as long as 1 to 2 hours following multiple IM injections
- in contrast to SGAs, treatment with haloperidol carries a significant risk of EPS
- adding a benzodiazepine “does not have strong evidence of benefit and carries risk of additional harm.”
Continue to: Haloperidol's well-known toxicity
Haloperidol’s well-known toxicity
Haloperidol has been associated with numerous adverse effects:
Akathisia and other acute EPS. Treatment with even a single dose of IM haloperidol can result in acute EPS, including dystonia and akathisia. At best, such adverse effects are subjectively troubling and unpleasant; at worst, akathisia can exacerbate and be mistaken for agitation, leading to administration of more medication23 and the possible development of suicidal or violent behavior.24-25 In the studies reviewed above, the overall rate of EPS was as high as 21% after treatment with haloperidol,16 with parkinsonism occurring in up to 17% of patients,19 dystonia in up to 11%,7 and akathisia in up to 10%.15 However, because specific EPS were assessed inconsistently, and sometimes not at all, the rate of akathisia—arguably the most relevant and counter-therapeutic adverse effect related to agitation—remains unclear.
In another study that specifically assessed for akathisia in patients treated with haloperidol, up to 40% experienced akathisia 6 hours after a single oral dose of 5 mg.26 Even a single dose of IV
Although anticholinergic medications or benzodiazepinesare often administered as part of a haloperidol “cocktail,” these medications often do not adequately resolve emergent akathisia.26,28 No clinical trials of IM haloperidol combined with benztropine or diphenhydramine have been published, but several studies suggest that combining haloperidol with
Cardiotoxicity. Although low-potency antipsychotic medications such as
Continue to: Although there is no direct evidence...
Although there is no direct evidence that the cardiac risks associated with IV haloperidol apply to IM administration, epidemiologic studies indicate that oral haloperidol carries an elevated risk of ventricular arrhythmia and sudden cardiac death,35,36 with 1 study reporting greater risk compared with other SGAs.37 Haloperidol, whether administered orally or IM, may therefore be an especially poor choice for patients with agitation who are at risk for arrhythmia, including those with relevant medical comorbidities or delirium.34
Neuronal cell death. Several lines of research evidence have demonstrated that haloperidol can cause cellular injury or death in neuronal tissue in a dose-dependent fashion through a variety of mechanisms.38 By contrast, SGAs have been shown to have neuroprotective effects.39 While these findings have mostly come from studies conducted in animals or in vitro human tumor cell lines, some researchers have nonetheless called for haloperidol to be banned, noting that if its neurotoxic effects were more widely known, “we would realize what a travesty it is to use [such] a brain-unfriendly drug.”40
Several reasonable alternatives
Echoing the earlier Cochrane review of haloperidol for psychosis-induced aggression or agitation,10 a 2017 update concluded, “If no other alternative exists, sole use of intramuscular haloperidol could be life-saving. Where additional drugs are available, sole use of haloperidol for extreme emergency could be considered unethical.”9
What then are reasonable alternatives to replace IM haloperidol for agitation? Clinicians should consider the following nonpharmacologic and pharmacologic interventions:
Nonpharmacologic interventions. Several behavioral interventions have been demonstrated to be effective for managing acute agitation, including verbal de-escalation, enhanced “programming” on the inpatient units, and the judicious use of seclusion.41-43 While such interventions may demand additional staff or resources, they have the potential to lower long-term costs, reduce injuries to patients and staff, and improve the quality of care.43 The use of IM haloperidol as a form of “chemical restraint” does not represent standard-of-care treatment,3 and from an ethical perspective, should never be implemented punitively or to compensate for substandard care in the form of inadequate staffing or staff training.
Continue to: Benzodiazepines
Benzodiazepines. Lorazepam offers an attractive alternative to haloperidol without the risk of EPS.2,4,8 However, lorazepam alone may be perceived as less efficacious than a haloperidol “cocktail” because it represents less overall medication. Some evidence has suggested that lorazepam, 4 mg, might be the most appropriate dose, although it has only rarely been studied in clinical trials of acute agitation.3
Respiratory depression is frequently cited as an argument against using lorazepam for agitation, as if the therapeutic window is extremely narrow with ineffectiveness at 2 mg, but potential lethality beyond that dose. In fact, serious respiratory depression with lorazepam is unlikely in the absence of chronic obstructive pulmonary disease (COPD), obstructive sleep apnea, or concomitant alcohol or other sedative use.46 Case reports have documented therapeutic lorazepam dosing of 2 to 4 mg every 2 hours up to 20 to 30 mg/d in patients with manic agitation.47 Even in patients with COPD, significant respiratory depression tends not to occur at doses <8 mg.48 A more evidence-based concern about lorazepam dosing is that 2 mg might be ineffective in patients with established tolerance. For example, 1 report described a patient in acute alcohol withdrawal who required dosing lorazepam to 1,600 mg within 24 hours.49 Collectively, these reports suggest that lorazepam has a much wider therapeutic window than is typically perceived, and that dosing with 3 to 4 mg IM is a reasonable option for agitation when 2 mg is likely to be inadequate.
Paradoxical disinhibition is another concern that might prevent benzodiazepines from being used alone as a first-line intervention for emergency treatment of agitation. However, similar to respiratory depression, this adverse event is relatively rare and tends to occur in children and geriatric patients, individuals intoxicated with alcohol or other sedatives, and patients with brain injury, developmental delay, or dementia.23,46 Although exacerbation of aggression has not been demonstrated in the RCTs examining benzodiazepines for agitation reviewed above, based on other research, some clinicians have expressed concerns about the potential for benzodiazepines to exacerbate aggression in patients with impulse control disorders and a history of violent behavior.50
The 2005 Expert Consensus Panel for Behavioral Emergencies51 recommended the use of lorazepam alone over haloperidol for agitation for patients for whom the diagnosis is unknown or includes the following:
- stimulant intoxication
- personality disorder
- comorbid obesity
- comorbid cardiac arrhythmia
- a history of akathisia and other EPS
- a history of amenorrhea/galactorrhea
- a history of seizures.
In surveys, patients have ranked lorazepam as the preferred medication for emergency agitation, whereas haloperidol was ranked as one of the least-preferred options.51,52
Continue to: Second-generation antipsychotics
Second-generation antipsychotics. The SGAs available in IM formulations, such as aripiprazole, olanzapine, and ziprasidone, have been shown to be at least as effective as haloperidol for the treatment of acute agitation (in 2015, the short-acting injectable formulation of aripiprazole was discontinued in the United States independent of safety or efficacy issues53). A review of RCTs examining IM SGAs for the treatment of agitation concluded that the number needed to treat for response compared with placebo was 5 for aripiprazole, 3 for olanzapine, and 3 for ziprasidone.54 In terms of safety, a meta-analysis of studies examining IM medications for agitation confirmed that the risk of acute EPS, including dystonia, akathisia, and parkinsonism, is significantly lower with SGAs compared with haloperidol.55 An RCT comparing IM ziprasidone with haloperidol found equivalently modest effects on QTc prolongation.56 Therefore, SGAs are an obvious and evidence-based option for replacing haloperidol as a treatment for acute agitation.
Unfortunately, for clinicians hoping to replace haloperidol within a multiple-medication IM “cocktail,” there have been no published controlled trials of SGAs combined with benzodiazepines. Although a short report indicated that aripiprazole and lorazepam are chemically compatible to be combined within a single injection,57 the package insert for aripiprazole warns that “If parenteral benzodiazepine therapy is deemed necessary in addition to ABILIFY injection treatment, patients should be monitored for excessive sedation and for orthostatic hypotension.”58 The package insert for olanzapine likewise lists the combination of lorazepam and olanzapine as a drug interaction that can potentiate sedation, and the manufacturer issued specific warnings about parenteral combination.59,60 A single published case of significant hypotension with combined IM olanzapine and lorazepam,60 together with the fact that IM olanzapine can cause hypotension by itself,61 has discouraged the coadministration of these medications. Nonetheless, the combination is used in some emergency settings, with several retrospective studies failing to provide evidence of hypotension or respiratory depression as adverse effects.62-64
Droperidol.
Over the past decade, however, droperidol has returned to the US market68 and its IV and IM usage has been revitalized for managing patients with agitation within or en route to the ED. Studies have demonstrated droperidol efficacy comparable to midazolam, ziprasidone, or olanzapine, as well as effectiveness as an IV adjunct to midazolam.69-71 In contrast to the FDA black-box warning, retrospective studies and RCTs of both IV and IM droperidol suggest that QTc prolongation and torsades de pointes are rare events that do not occur any more frequently than they do with haloperidol, even at doses >10 mg.72,73 However, in studies involving patients with drug intoxication and treatment with multiple medications, oversedation to the point of needing rescue intervention was reported. In an emergency setting where these issues are relatively easily managed, such risks may be better tolerated than in psychiatric settings.
With earlier studies examining the use of droperidol in an acute psychiatric setting that reported a more rapid onset of action than haloperidol,65-67 a 2016 Cochrane review concluded that there was high-quality evidence to support droperidol’s use for psychosis-induced agitation.74 However, a 2015 RCT comparing IM droperidol, 10 mg, to haloperidol, 10 mg, found equivalent efficacy and response times (with maximal response occurring within 2 hours) and concluded that droperidol had no advantage over haloperidol.75 Because none of the clinical trials that evaluated droperidol have included assessments for EPS, its risk of akathisia remains uncertain.
Continue to: Ketamine
Ketamine. In recent years, ketamine has been used to treat acute agitation within or en route to the ED. Preliminary observational studies support ketamine’s efficacy when administered via IV or IM routes,76 with more rapid symptomatic improvement compared with haloperidol, lorazepam, or midazolam alone.77 Reported adverse effects of ketamine include dissociation, psychotic exacerbation, and respiratory depression,76 although 1 small naturalistic study found no evidence of exacerbation of psychotic or other psychiatric symptoms.78 An ongoing RCT is comparing IM ketamine, 5 mg/kg, to combined IM haloperidol, 5 mg, and midazolam, 5 mg.79 Although various ketamine formulations are increasingly being used in psychiatry, active psychosis is generally regarded as a contraindication. It is premature to recommend parenteral ketamine administration for agitation within most psychiatric settings until more research on safety has been completed.
Haloperidol, or something else? Practical considerations
Consider the following factors when deciding whether to use haloperidol or one of its alternatives:
Limitations of the evidence. Modern clinical trials requiring informed consent often do not include the kind of severe agitation that clinicians encounter in acute psychiatric, emergency, or forensic settings. In addition, standard interventions, such as 3-medication haloperidol “cocktails,” have not been evaluated in clinical trials. Clinicians are therefore often in the dark about optimal evidence-based practices.
Treatment goals. Psychiatric agitation has many causes, with a range of severity that warrants a commensurate range of responses. Protocols for managing acute agitation should include graded interventions that begin with nonpharmacologic interventions and voluntary oral medications, and move to involuntary IM medications when necessary.
While treatment guidelines clearly recommend against IM medications as “chemical restraint” with a goal of sedating a patient until he/she is unconscious,3,51 such outcomes are nonetheless often sought by staff who are concerned about the risk of injuries during a behavioral emergency. In such instances, the risks of violence towards patients and staff may outweigh concerns about adverse effects in a risk-benefit analysis. Consequently, clinicians may be prone to “skip over” graded interventions because they assume they “won’t work” in favor of administering involuntary multiple-medication haloperidol “cocktails” despite risks of excess sedation, EPS, and cardiotoxicity. Treatment settings should critically evaluate such biased preferences, with a goal of developing tailored, evidence-based strategies that maximize benefits while minimizing excess sedation and other untoward adverse effects, with an eye towards promoting better overall patient care and reducing length of stay.42,43,80
Continue to: Limitations of available medications
Limitations of available medications. There is no perfect medication for the management of acute agitation. Evidence indicates that pharmacologic options take 15 minutes to several hours to resolve acute agitation, even potentially more rapid-acting medications such as midazolam and droperidol. This is well beyond most clinicians’ desired window for response time in a behavioral emergency. Multiple-medication “cocktails” may be used with the hope of hastening response time, but may not achieve this goal at the expense of increasing the risk of adverse effects and the likelihood that a patient will remain sedated for a prolonged time. In the real world, this often means that by the time a psychiatrist comes to evaluate a patient who has been given emergency medications, the patient cannot be aroused for an interview. Ideally, medications would calm an agitated patient rapidly, without excess or prolonged sedation.80 Less-sedating SGAs, such as ziprasidone, might have this potential, but can sometimes be perceived as ineffective.
Avoiding akathisia. Akathisia’s potential to worsen and be mistaken for agitation makes it an especially concerning, if underappreciated, adverse effect of haloperidol that is often not adequately assessed in clinical trials or practice. In light of evidence that akathisia can occur in nearly half of patients receiving a single 5 mg-dose of haloperidol, it is difficult to justify the use of this medication for agitation when equally effective options exist with a lower risk of EPS.
While haloperidol-induced akathisia could in theory be mitigated by adding anticholinergic medications or benzodiazepines, previous studies have found that such strategies have limited effectiveness compared to “gold standard” treatment with propranolol.28,81,82 Furthermore, the half-lives of anticholinergic medications, such as benztropine or diphenhydramine, are significantly shorter than that of a single dose of haloperidol, which can be as long as 37 hours.83 Therefore, akathisia and other EPS could emerge or worsen several hours or even days after receiving an IM haloperidol “cocktail” as the shorter-acting medications wear off. Akathisia is best minimized by avoiding FGAs, such as haloperidol, when treating acute agitation.
Promoting adherence. Although haloperidol is often recommended for acute agitation in patients with schizophrenia or bipolar disorder on the basis that it would treat the underlying condition, many patients who receive IM medications for acute agitation are already prescribed standing doses of oral medication, which increases the risk of cumulative toxicity. In addition, receiving a medication likely to cause acute EPS that is ranked near the bottom of patient preferences may erode the potential for a therapeutic alliance and hamper longer-term antipsychotic medication adherence.

Time for a change
For nearly half a century, haloperidol has been a “gold standard” intervention for IM control in patients with agitation. However, given its potential to produce adverse effects, including a significant risk of akathisia that can worsen agitation, along with the availability of newer pharmacologic options that are at least as effective (Table 1, and Table 2), haloperidol should be retired as a first-line medication for the treatment of agitation. Clinicians would benefit from RCTs investigating the safety and efficacy of novel interventions including frequently-used, but untested medication combinations, as well as nonpharmacologic interventions.

Continue to: Bottom Line
Bottom Line
Although there is no perfect IM medication to treat acute agitation, haloperidol’s higher risk of adverse effects relative to newer alternatives suggest that it should no longer be considered a first-line intervention.
Related Resources
- Zun LS. Evidence-based review of pharmacotherapy for acute agitation. Part 1: onset of efficacy. J Emerg Med. 2018;54(3):364-374.
- Zun LS. Evidence-based review of pharmacotherapy for acute agitation. Part 2: safety. J Emerg Med. 2018;54(4): 522-532.
Drug Brand Names
Aripiprazole • Abilify
Benztropine • Cogentin
Chlorpromazine • Thorazine
Diphenhydramine • Benadryl
Droperidol • Inapsine
Haloperidol • Haldol
Ketamine • Ketalar
Lorazepam • Ativan
Midazolam • Versed
Olanzapine • Zyprexa
Prochlorperazine • Compazine
Promethazine • Phenergan
Propranolol • Inderal, Pronol
Ziprasidone • Geodon
For more than half a century, haloperidol has been used as a first-line medication for psychiatric agitation constituting a “behavioral emergency” when a patient cannot or will not take oral medication. Today, haloperidol is most commonly administered as an IM injection along with an anticholinergic medication to minimize extrapyramidal symptoms (EPS) and a benzodiazepine for additional sedation. The multiple-medication “cocktail” is often referred to by double-entendre nicknames, such as “B-52” or “5250” (ie, haloperidol, 5 mg; lorazepam, 2 mg; and
Earlier evidence of haloperidol’s efficacy
The initial “discovery” of antipsychotic medications was made in 1951 based on the inadvertent observation that chlorpromazine had the potential to calm surgical patients with autonomic activation. This calming effect, described as “désintéressment” (meaning a kind of “indifference to the world”),1 resulted in a new class of medications replacing barbiturates and bromides as go-to options to achieve “rapid tranquilization” of psychiatric agitation.2 Although the ability of antipsychotic medications to gradually reduce positive symptoms, such as delusions and hallucinations, has been attributed to dopamine (D2) antagonism, their more immediate sedating and anti-agitation effects are the result of broader effects as histamine (H1) and alpha-1 adrenergic antagonists.
In the 1970s, haloperidol emerged as a first-line option to manage agitation due to its IM and IV availability, as well as its relative lack of sedation and orthostasis compared with low-potency D2 antagonists such as chlorpromazine. However, haloperidol was observed to have a significant risk of acute EPS, including dystonic reactions.2 From the 1970s to the 1990s, numerous prospective clinical trials of haloperidol for the treatment of acute psychotic agitation, including several randomized controlled trials (RCTs) comparing haloperidol to lorazepam, were conducted.3 The design and outcomes of the haloperidol vs lorazepam RCTs were fairly consistent4-7:
- adult participants with acute agitation and a variety of psychiatric diagnoses, for whom informed consent often was waived due to agitation severity
- randomization to either IM haloperidol, 5 mg, or IM lorazepam, 2 mg, administered every 30 minutes until agitation resolved
- behavioral outcomes measured over several hours using various rating scales, without consistent assessment of EPS
- equivalent efficacy of haloperidol and lorazepam, with symptom resolution usually achieved after 1 to 2 doses (in 30 to 60 minutes), but sometimes longer
- anticholinergic “rescue” allowed for EPS, but not administered prophylactically
- EPS, including dystonia and akathisia, were significantly more frequent with haloperidol compared with lorazepam.8
In recognition of the greater risk of EPS with haloperidol compared with lorazepam, and the fact that most study participants were already taking standing doses of antipsychotic medications, some researchers have recommended using benzodiazepines alone as the optimal treatment for agitation.4,9 A 2012 Cochrane review concluded that the involuntary use of haloperidol alone “could be considered unethical.”10,11 However, other studies that examined the combination of haloperidol and lorazepam compared with either medication alone found that the combination of the 2 medications was associated with a more rapid resolution of symptoms, which suggests a superior synergistic effect.6,7,12 By the late 1990s, combined haloperidol and lorazepam, often mixed within a single injection, became the most common strategy to achieve rapid tranquilization in the psychiatric emergency setting.13 However, while the combination has been justified as a way to reduce the antipsychotic medication dose and EPS risk,2 few studies have compared combinations containing <5 mg of haloperidol. As a result, the apparent superiority of combined haloperidol and lorazepam compared with either medication alone may be a simple cumulative dose effect rather than true synergism. It is also important to note that adding lorazepam to haloperidol does not mitigate the risk of EPS such as dystonia in the absence of anticholinergic medication.8 To date, however, there have been no clinical trials investigating the efficacy of IM haloperidol, lorazepam, and
Newer RCTs tell a different story
With the availability of second-generation antipsychotics (SGAs) in IM formulations, clinical trials over the past 2 decades have focused on comparing SGAs with haloperidol alone as the “gold standard” control for acute agitation. Compared with previous trials of
- Study participants who signed informed consent (and were likely less agitated)
- IM haloperidol doses typically >5 mg (eg, 6.5 to 10 mg).
As with studies comparing lorazepam with haloperidol, the results of these RCTs revealed that IM
An updated 2017 Cochrane review of haloperidol for psychosis-induced aggression or agitation concluded that9:
- haloperidol is an effective intervention, although the evidence is “weak”
- significant treatment effects may take as long as 1 to 2 hours following multiple IM injections
- in contrast to SGAs, treatment with haloperidol carries a significant risk of EPS
- adding a benzodiazepine “does not have strong evidence of benefit and carries risk of additional harm.”
Continue to: Haloperidol's well-known toxicity
Haloperidol’s well-known toxicity
Haloperidol has been associated with numerous adverse effects:
Akathisia and other acute EPS. Treatment with even a single dose of IM haloperidol can result in acute EPS, including dystonia and akathisia. At best, such adverse effects are subjectively troubling and unpleasant; at worst, akathisia can exacerbate and be mistaken for agitation, leading to administration of more medication23 and the possible development of suicidal or violent behavior.24-25 In the studies reviewed above, the overall rate of EPS was as high as 21% after treatment with haloperidol,16 with parkinsonism occurring in up to 17% of patients,19 dystonia in up to 11%,7 and akathisia in up to 10%.15 However, because specific EPS were assessed inconsistently, and sometimes not at all, the rate of akathisia—arguably the most relevant and counter-therapeutic adverse effect related to agitation—remains unclear.
In another study that specifically assessed for akathisia in patients treated with haloperidol, up to 40% experienced akathisia 6 hours after a single oral dose of 5 mg.26 Even a single dose of IV
Although anticholinergic medications or benzodiazepinesare often administered as part of a haloperidol “cocktail,” these medications often do not adequately resolve emergent akathisia.26,28 No clinical trials of IM haloperidol combined with benztropine or diphenhydramine have been published, but several studies suggest that combining haloperidol with
Cardiotoxicity. Although low-potency antipsychotic medications such as
Continue to: Although there is no direct evidence...
Although there is no direct evidence that the cardiac risks associated with IV haloperidol apply to IM administration, epidemiologic studies indicate that oral haloperidol carries an elevated risk of ventricular arrhythmia and sudden cardiac death,35,36 with 1 study reporting greater risk compared with other SGAs.37 Haloperidol, whether administered orally or IM, may therefore be an especially poor choice for patients with agitation who are at risk for arrhythmia, including those with relevant medical comorbidities or delirium.34
Neuronal cell death. Several lines of research evidence have demonstrated that haloperidol can cause cellular injury or death in neuronal tissue in a dose-dependent fashion through a variety of mechanisms.38 By contrast, SGAs have been shown to have neuroprotective effects.39 While these findings have mostly come from studies conducted in animals or in vitro human tumor cell lines, some researchers have nonetheless called for haloperidol to be banned, noting that if its neurotoxic effects were more widely known, “we would realize what a travesty it is to use [such] a brain-unfriendly drug.”40
Several reasonable alternatives
Echoing the earlier Cochrane review of haloperidol for psychosis-induced aggression or agitation,10 a 2017 update concluded, “If no other alternative exists, sole use of intramuscular haloperidol could be life-saving. Where additional drugs are available, sole use of haloperidol for extreme emergency could be considered unethical.”9
What then are reasonable alternatives to replace IM haloperidol for agitation? Clinicians should consider the following nonpharmacologic and pharmacologic interventions:
Nonpharmacologic interventions. Several behavioral interventions have been demonstrated to be effective for managing acute agitation, including verbal de-escalation, enhanced “programming” on the inpatient units, and the judicious use of seclusion.41-43 While such interventions may demand additional staff or resources, they have the potential to lower long-term costs, reduce injuries to patients and staff, and improve the quality of care.43 The use of IM haloperidol as a form of “chemical restraint” does not represent standard-of-care treatment,3 and from an ethical perspective, should never be implemented punitively or to compensate for substandard care in the form of inadequate staffing or staff training.
Continue to: Benzodiazepines
Benzodiazepines. Lorazepam offers an attractive alternative to haloperidol without the risk of EPS.2,4,8 However, lorazepam alone may be perceived as less efficacious than a haloperidol “cocktail” because it represents less overall medication. Some evidence has suggested that lorazepam, 4 mg, might be the most appropriate dose, although it has only rarely been studied in clinical trials of acute agitation.3
Respiratory depression is frequently cited as an argument against using lorazepam for agitation, as if the therapeutic window is extremely narrow with ineffectiveness at 2 mg, but potential lethality beyond that dose. In fact, serious respiratory depression with lorazepam is unlikely in the absence of chronic obstructive pulmonary disease (COPD), obstructive sleep apnea, or concomitant alcohol or other sedative use.46 Case reports have documented therapeutic lorazepam dosing of 2 to 4 mg every 2 hours up to 20 to 30 mg/d in patients with manic agitation.47 Even in patients with COPD, significant respiratory depression tends not to occur at doses <8 mg.48 A more evidence-based concern about lorazepam dosing is that 2 mg might be ineffective in patients with established tolerance. For example, 1 report described a patient in acute alcohol withdrawal who required dosing lorazepam to 1,600 mg within 24 hours.49 Collectively, these reports suggest that lorazepam has a much wider therapeutic window than is typically perceived, and that dosing with 3 to 4 mg IM is a reasonable option for agitation when 2 mg is likely to be inadequate.
Paradoxical disinhibition is another concern that might prevent benzodiazepines from being used alone as a first-line intervention for emergency treatment of agitation. However, similar to respiratory depression, this adverse event is relatively rare and tends to occur in children and geriatric patients, individuals intoxicated with alcohol or other sedatives, and patients with brain injury, developmental delay, or dementia.23,46 Although exacerbation of aggression has not been demonstrated in the RCTs examining benzodiazepines for agitation reviewed above, based on other research, some clinicians have expressed concerns about the potential for benzodiazepines to exacerbate aggression in patients with impulse control disorders and a history of violent behavior.50
The 2005 Expert Consensus Panel for Behavioral Emergencies51 recommended the use of lorazepam alone over haloperidol for agitation for patients for whom the diagnosis is unknown or includes the following:
- stimulant intoxication
- personality disorder
- comorbid obesity
- comorbid cardiac arrhythmia
- a history of akathisia and other EPS
- a history of amenorrhea/galactorrhea
- a history of seizures.
In surveys, patients have ranked lorazepam as the preferred medication for emergency agitation, whereas haloperidol was ranked as one of the least-preferred options.51,52
Continue to: Second-generation antipsychotics
Second-generation antipsychotics. The SGAs available in IM formulations, such as aripiprazole, olanzapine, and ziprasidone, have been shown to be at least as effective as haloperidol for the treatment of acute agitation (in 2015, the short-acting injectable formulation of aripiprazole was discontinued in the United States independent of safety or efficacy issues53). A review of RCTs examining IM SGAs for the treatment of agitation concluded that the number needed to treat for response compared with placebo was 5 for aripiprazole, 3 for olanzapine, and 3 for ziprasidone.54 In terms of safety, a meta-analysis of studies examining IM medications for agitation confirmed that the risk of acute EPS, including dystonia, akathisia, and parkinsonism, is significantly lower with SGAs compared with haloperidol.55 An RCT comparing IM ziprasidone with haloperidol found equivalently modest effects on QTc prolongation.56 Therefore, SGAs are an obvious and evidence-based option for replacing haloperidol as a treatment for acute agitation.
Unfortunately, for clinicians hoping to replace haloperidol within a multiple-medication IM “cocktail,” there have been no published controlled trials of SGAs combined with benzodiazepines. Although a short report indicated that aripiprazole and lorazepam are chemically compatible to be combined within a single injection,57 the package insert for aripiprazole warns that “If parenteral benzodiazepine therapy is deemed necessary in addition to ABILIFY injection treatment, patients should be monitored for excessive sedation and for orthostatic hypotension.”58 The package insert for olanzapine likewise lists the combination of lorazepam and olanzapine as a drug interaction that can potentiate sedation, and the manufacturer issued specific warnings about parenteral combination.59,60 A single published case of significant hypotension with combined IM olanzapine and lorazepam,60 together with the fact that IM olanzapine can cause hypotension by itself,61 has discouraged the coadministration of these medications. Nonetheless, the combination is used in some emergency settings, with several retrospective studies failing to provide evidence of hypotension or respiratory depression as adverse effects.62-64
Droperidol.
Over the past decade, however, droperidol has returned to the US market68 and its IV and IM usage has been revitalized for managing patients with agitation within or en route to the ED. Studies have demonstrated droperidol efficacy comparable to midazolam, ziprasidone, or olanzapine, as well as effectiveness as an IV adjunct to midazolam.69-71 In contrast to the FDA black-box warning, retrospective studies and RCTs of both IV and IM droperidol suggest that QTc prolongation and torsades de pointes are rare events that do not occur any more frequently than they do with haloperidol, even at doses >10 mg.72,73 However, in studies involving patients with drug intoxication and treatment with multiple medications, oversedation to the point of needing rescue intervention was reported. In an emergency setting where these issues are relatively easily managed, such risks may be better tolerated than in psychiatric settings.
With earlier studies examining the use of droperidol in an acute psychiatric setting that reported a more rapid onset of action than haloperidol,65-67 a 2016 Cochrane review concluded that there was high-quality evidence to support droperidol’s use for psychosis-induced agitation.74 However, a 2015 RCT comparing IM droperidol, 10 mg, to haloperidol, 10 mg, found equivalent efficacy and response times (with maximal response occurring within 2 hours) and concluded that droperidol had no advantage over haloperidol.75 Because none of the clinical trials that evaluated droperidol have included assessments for EPS, its risk of akathisia remains uncertain.
Continue to: Ketamine
Ketamine. In recent years, ketamine has been used to treat acute agitation within or en route to the ED. Preliminary observational studies support ketamine’s efficacy when administered via IV or IM routes,76 with more rapid symptomatic improvement compared with haloperidol, lorazepam, or midazolam alone.77 Reported adverse effects of ketamine include dissociation, psychotic exacerbation, and respiratory depression,76 although 1 small naturalistic study found no evidence of exacerbation of psychotic or other psychiatric symptoms.78 An ongoing RCT is comparing IM ketamine, 5 mg/kg, to combined IM haloperidol, 5 mg, and midazolam, 5 mg.79 Although various ketamine formulations are increasingly being used in psychiatry, active psychosis is generally regarded as a contraindication. It is premature to recommend parenteral ketamine administration for agitation within most psychiatric settings until more research on safety has been completed.
Haloperidol, or something else? Practical considerations
Consider the following factors when deciding whether to use haloperidol or one of its alternatives:
Limitations of the evidence. Modern clinical trials requiring informed consent often do not include the kind of severe agitation that clinicians encounter in acute psychiatric, emergency, or forensic settings. In addition, standard interventions, such as 3-medication haloperidol “cocktails,” have not been evaluated in clinical trials. Clinicians are therefore often in the dark about optimal evidence-based practices.
Treatment goals. Psychiatric agitation has many causes, with a range of severity that warrants a commensurate range of responses. Protocols for managing acute agitation should include graded interventions that begin with nonpharmacologic interventions and voluntary oral medications, and move to involuntary IM medications when necessary.
While treatment guidelines clearly recommend against IM medications as “chemical restraint” with a goal of sedating a patient until he/she is unconscious,3,51 such outcomes are nonetheless often sought by staff who are concerned about the risk of injuries during a behavioral emergency. In such instances, the risks of violence towards patients and staff may outweigh concerns about adverse effects in a risk-benefit analysis. Consequently, clinicians may be prone to “skip over” graded interventions because they assume they “won’t work” in favor of administering involuntary multiple-medication haloperidol “cocktails” despite risks of excess sedation, EPS, and cardiotoxicity. Treatment settings should critically evaluate such biased preferences, with a goal of developing tailored, evidence-based strategies that maximize benefits while minimizing excess sedation and other untoward adverse effects, with an eye towards promoting better overall patient care and reducing length of stay.42,43,80
Continue to: Limitations of available medications
Limitations of available medications. There is no perfect medication for the management of acute agitation. Evidence indicates that pharmacologic options take 15 minutes to several hours to resolve acute agitation, even potentially more rapid-acting medications such as midazolam and droperidol. This is well beyond most clinicians’ desired window for response time in a behavioral emergency. Multiple-medication “cocktails” may be used with the hope of hastening response time, but may not achieve this goal at the expense of increasing the risk of adverse effects and the likelihood that a patient will remain sedated for a prolonged time. In the real world, this often means that by the time a psychiatrist comes to evaluate a patient who has been given emergency medications, the patient cannot be aroused for an interview. Ideally, medications would calm an agitated patient rapidly, without excess or prolonged sedation.80 Less-sedating SGAs, such as ziprasidone, might have this potential, but can sometimes be perceived as ineffective.
Avoiding akathisia. Akathisia’s potential to worsen and be mistaken for agitation makes it an especially concerning, if underappreciated, adverse effect of haloperidol that is often not adequately assessed in clinical trials or practice. In light of evidence that akathisia can occur in nearly half of patients receiving a single 5 mg-dose of haloperidol, it is difficult to justify the use of this medication for agitation when equally effective options exist with a lower risk of EPS.
While haloperidol-induced akathisia could in theory be mitigated by adding anticholinergic medications or benzodiazepines, previous studies have found that such strategies have limited effectiveness compared to “gold standard” treatment with propranolol.28,81,82 Furthermore, the half-lives of anticholinergic medications, such as benztropine or diphenhydramine, are significantly shorter than that of a single dose of haloperidol, which can be as long as 37 hours.83 Therefore, akathisia and other EPS could emerge or worsen several hours or even days after receiving an IM haloperidol “cocktail” as the shorter-acting medications wear off. Akathisia is best minimized by avoiding FGAs, such as haloperidol, when treating acute agitation.
Promoting adherence. Although haloperidol is often recommended for acute agitation in patients with schizophrenia or bipolar disorder on the basis that it would treat the underlying condition, many patients who receive IM medications for acute agitation are already prescribed standing doses of oral medication, which increases the risk of cumulative toxicity. In addition, receiving a medication likely to cause acute EPS that is ranked near the bottom of patient preferences may erode the potential for a therapeutic alliance and hamper longer-term antipsychotic medication adherence.

Time for a change
For nearly half a century, haloperidol has been a “gold standard” intervention for IM control in patients with agitation. However, given its potential to produce adverse effects, including a significant risk of akathisia that can worsen agitation, along with the availability of newer pharmacologic options that are at least as effective (Table 1, and Table 2), haloperidol should be retired as a first-line medication for the treatment of agitation. Clinicians would benefit from RCTs investigating the safety and efficacy of novel interventions including frequently-used, but untested medication combinations, as well as nonpharmacologic interventions.

Continue to: Bottom Line
Bottom Line
Although there is no perfect IM medication to treat acute agitation, haloperidol’s higher risk of adverse effects relative to newer alternatives suggest that it should no longer be considered a first-line intervention.
Related Resources
- Zun LS. Evidence-based review of pharmacotherapy for acute agitation. Part 1: onset of efficacy. J Emerg Med. 2018;54(3):364-374.
- Zun LS. Evidence-based review of pharmacotherapy for acute agitation. Part 2: safety. J Emerg Med. 2018;54(4): 522-532.
Drug Brand Names
Aripiprazole • Abilify
Benztropine • Cogentin
Chlorpromazine • Thorazine
Diphenhydramine • Benadryl
Droperidol • Inapsine
Haloperidol • Haldol
Ketamine • Ketalar
Lorazepam • Ativan
Midazolam • Versed
Olanzapine • Zyprexa
Prochlorperazine • Compazine
Promethazine • Phenergan
Propranolol • Inderal, Pronol
Ziprasidone • Geodon
1. Shorter E. A history of psychiatry. New York, NY: John Wiley & Sons, Inc.; 1997:249.
2. Salzman C, Green AI, Rodriguez-Villa F, et al. Benzodiazepines combined with neuroleptics for management of severe disruptive behavior. Psychosomatics. 1986;27(suppl 1):17-22.
3. Allen MH. Managing the agitated psychotic patient: a reappraisal of the evidence. J Clin Psychiatr. 2000;61(suppl 14):11-20.
4. Salzman C, Solomon D, Miyawaki E, et al. Parenteral lorazepam versus parenteral haloperidol for the control of psychotic disruptive behavior. J Clin Psychiatr. 1991:52(4):177-180.
5. Allen MH, Currier GW, Hughes DH, et al. The expert consensus guideline series: treatment of behavioral emergencies. Postgrad Med. 2001;(Spec No):1-88; quiz 89-90.
6. Foster S, Kessel J, Berman ME, et al. Efficacy of lorazepam and haloperidol for rapid tranquilization in a psychiatric emergency room setting. Int Clin Psychopharmacol. 1997;12(3):175-179.
7. Garza-Trevino WS, Hollister LE, Overall JE, et al. Efficacy of combinations of intramuscular antipsychotics and sedative-hypnotics for control of psychotic agitation. Am J Psychiatr. 1989:146(12):1598-1601.
8. Battaglia J, Moss S, Rush J, et al. Haloperidol, lorazepam, or both for psychotic agitation? A multicenter, prospective double-blind, emergency study. Am J Emerg Med 1997;15(4):335-340.
9. Ostinelli EG, Brooke-Powney MJ, Li X, et al. Haloperidol for psychosis-induced aggression or agitation (rapid tranquillisation). Cochrane Database Syst Rev. 2017; 7:CD009377. doi: 10.1002/14651858.CD009377.pub3.
10. Powney MJ, Adams CE, Jones H. Haloperidol for psychosis-induced aggression or agitation (rapid tranquillisation). Cochrane Database Syst Rev. 2012;11:CD009377. doi: 10.1002/14651858.CD009377.pub2.
11. Citrome L. Review: limited evidence on effects of haloperidol alone for rapid tranquillisation in psychosis-induced aggression. Evid Based Ment Health. 2013;16(2):47.
12. Bienek SA, Ownby R, Penalver A, et al. A double-blind study of lorazepam versus the combination of haloperidol and lorazepam in managing agitation. Pharmacother. 1998;18(1):57-62.
13. Binder RL, McNiel DE. Contemporary practices in managing acutely violent patients in 20 psychiatric emergency rooms. Psychiatric Serv. 1999;50(2):1553-1554.
14. Andrezina R, Josiassen RC, Marcus RN, et al. Intramuscular aripiprazole for the treatment of acute agitation in patients with schizophrenia or schizoaffective disorder: a double-blind, placebo-controlled comparison with intramuscular haloperidol. Psychopharmacology (Berl). 2006;188(3):281-292.
15. Tran-Johnson TK, Sack DA, Marcus RN, et al. Efficacy and safety of intramuscular aripiprazole in patients with acute agitation: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatr. 2007;68(1):111-119.
16. Brook S, Lucey JV, Gunn KP. Intramuscular ziprasidone compared with intramuscular haloperidol in the treatment of acute psychosis. J Clin Psychiatr. 2000;61(12):933-941.
17. Brook S, Walden J, Benattia I, et al. Ziprasidone and haloperidol in the treatment of acute exacerbation of schizophrenia and schizoaffective disorder: comparison of intramuscular and oral formulations in a 6-week, randomized, blinded-assessment study. Psychopharmacology (Berl). 2005;178(4):514-523.
18. Wright P, Birkett M, David SR, et al. Double-blind, placebo-controlled comparison of intramuscular olanzapine and intramuscular haloperidol in the treatment of acute agitation in schizophrenia. Am J Psychiatr. 2001;158(7):1149-1151.
19. Breier A, Meehan K, Birkett M, et al. A double-blind, placebo-controlled dose-response comparison of intramuscular olanzapine and haloperidol in the treatment of acute agitation in schizophrenia. Arch Gen Psych. 2002;59(5):441-448.
20. Hsu W, Huang S, Lee B, et al. Comparison of intramuscular olanzapine, orally disintegrating olanzapine tablets, oral risperidone solution, and intramuscular haloperidol in the management of acute agitation in an acute care psychiatric ward in Taiwan. J Clin Psychopharmacol. 2010;30(3):230-234.
21. Chan H, Ree S, Su L, et al. A double-blind, randomized comparison study of efficacy and safety of intramuscular olanzapine and intramuscular haloperidol in patients with schizophrenia and acute agitated behavior. J Clin Psychopharmacol. 2014;34(3):355-358.
22. Baldaçara L, Sanches M, Cordeiro DC, et al. Rapid tranquilization for agitated patients in emergency psychiatric rooms: a randomized trial of olanzapine, ziprasidone, haloperidol plus promethazine, haloperidol plus midazolam and haloperidol alone. Braz J Psychiatry. 2011;33(1):30-39.
23. Hillard JR. Defusing patient violence. Current Psychiatry. 2002;1(4):22-29.
24. Seemüller F, Schennach R, Mayr A, et al. Akathisia and suicidal ideation in first-episode schizophrenia. J Clin Psychopharmacol. 2012;32(5):694-698.
25. Eikelenboom-Schieveld SJM, Lucire Y, Fogleman JC. The relevance of cytochrome P450 polymorphism in forensic medicine and akathisia-related violence and suicide. J Forens Leg Med. 2016;41:65-71.
26. Van Putten T, May PRA, Marder SR. Akathisia with haloperidol and thiothixene. Arch Gen Psych. 1984;41:1036-1039.
27. Drotts DL, Vinson DR. Prochlorperazine induced akathisia in emergency patients. Ann Emerg Med. 1999;34(4):469-475.
28. Salem H, Negpal C, Pigott T. Revisiting antipsychotic-induced akathisia: current issues and prospective challenges. Curr Neuropharmacol. 2017;15(5):789-798.
29. Huf G, Coutinho ESF, Adams CE. Rapid tranquilization in psychiatric emergency settings in Brazil: pragmatic randomized controlled trial of intramuscular haloperidol versus intramuscular haloperidol plus promethazine. BMJ. 2007;335(7625):869.
30. Mantovani C, Labate CM, Sponholz A, et al. Are low doses of antipsychotics effective in the management of psychomotor agitation? A randomized, rated-blind trial of 4 intramuscular interventions. J Clin Psychopharmacol. 2013;33(3):306-312.
31. Darwish H, Grant R, Haslam R, et al. Promethazine-induced acute dystonic reactions. Am J Dis Child. 1980;134(10):990-991.
32. Jyothi CH, Rudraiah HGM, Vidya HK, et al. Promethazine induced acute dystonia: a case report. Manipal J Med Sci. 2016;1(2):63-64.
33. Ames D, Carr-Lopez SM, Gutierrez MA, et al. Detecting and managing adverse effects of antipsychotic medications: current state of play. Psychiatr Clin North Am. 2016;39(2):275-311.
34. Meyer-Massetti C, Cheng CM, Sharpe MA, et al. The FDA extended warning for intravenous haloperidol and torsades de pointes: how should institutions respond? J Hosp Med. 2010;5(4):E8-E16. doi: 10.1002/jhm.691.
35. Wu C, Tsai Y, Tsai H. Antipsychotic drugs and the risk of ventricular arrhythmia and/or sudden cardiac death: a nation-wide case-crossover study. J Am Heart Dis. 2015;4(2):e001568. doi: 10.1161/JAHA.114.001568.
36. Beach SR, Celano CM, Sugrue AM, et al. QT prolongation, torsades de pointe, and psychotropic medications: a 5-year update. Psychosomatics. 2018;59(1):105-122.
37. Leonard CE, Freeman CP, Newcomb CW, et al. Antipsychotics and the risks of sudden cardiac death and all-cause death: cohort studies in Medicaid and dually-eligible Medicaid-Medicare beneficiaries of five states. J Clin Exp Cardiol. 2013;suppl 10(6):1-9.
38. Nasrallah H, Chen AT. Multiple neurotoxic effects of haloperidol resulting in neuronal death. Ann Clin Psychiatr. 2017;29(3):195-202.
39. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
40. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2013;12(7):7-8.
41. Corrigan PW, Yudofsky SC, Silver JM. Pharmacological and behavioral treatments for aggressive psychiatric inpatients. Hosp Comm Psychiatr. 1993;44(2):125-133.
42. Zeller SL, Citrome L. Managing agitation associated with schizophrenia and bipolar disorder in the emergency setting. West J Emerg Med. 2016;17(2):165-172.
43. Vieta E, Garriga M, Cardete L, et al. Protocol for the management of psychiatric patients with psychomotor agitation. BMC Psychiatr. 2017;17:328.
44. Nobay F, Simon BC, Levitt A, et al. A prospective, double-blind, randomized trial of midazolam versus haloperidol versus lorazepam in the chemical restraint of violent and severely agitated patients. Acad Emerg Med. 2004;11(7):744-749.
45. Klein LR, Driver BE, Miner JR, et al. Intramuscular midazolam, olanzapine, ziprasidone, or haloperidol for treating acute agitation in the emergency department. Ann Emerg Med. 2018;72(4):374-385.
46. Hillard JR. Emergency treatment of acute psychosis. J Clin Psychiatr. 1998;59(suppl 1):57-60.
47. Modell JG, Lenox RH, Weiner S. Inpatient clinical trial of lorazepam for the management of manic agitation. J Clin Psychopharmacol. 1985;5(2):109-110.
48. Denaut M, Yernault JC, De Coster A. Double-blind comparison of the respiratory effects of parenteral lorazepam and diazepam in patients with chronic obstructive lung disease. Curr Med Res Opin. 1975;2(10):611-615.
49. Kahn DR, Barnhorst AV, Bourgeois JA. A case of alcohol withdrawal requiring 1,600 mg of lorazepam in 24 hours. CNS Spectr. 2009;14(7):385-389.
50. Jones KA. Benzodiazepines: their role in aggression and why GPs should prescribe with caution. Austral Fam Physician. 2011;40(11):862-865.
51. Allen MH, Currier GW, Carpenter D, et al. The expert consensus guideline series. Treatment of behavioral emergencies 2005. J Psychiatr Pract. 2005;11(suppl 1):5-108.
52. Allen MH, Carpenter D, Sheets JL, et al. What do consumers say they want and need during a psychiatric emergency? J Psychiatr Pract. 2003;9(1):39-58.
53. Han DH. Some Abilify formulations to discontinue in 2015. MPR. https://www.empr.com/home/news/some-abilify-formulations-to-discontinue-in-2015/. Published January 13, 2015. Accessed April 17, 2020.
54. Citrome L. Comparison of intramuscular ziprasidone, olanzapine, or aripiprazole for agitation: a quantitative review of efficacy and safety. J Clin Psychiatry. 2007;68(12):1876-1885.
55. Satterthwaite TD, Wolf DH, Rosenheck RA, et al. A meta-analysis of the risk of acute extrapyramidal symptoms with intramuscular antipsychotics for the treatment for agitation. J Clin Psychiatr. 2008;69(12):1869-1879.
56. Miceli JJ, Tensfeldt TG, Shiovitz T, et al. Effects of high-dose ziprasidone and haloperidol on the QTc interval after intramuscular administration: a randomized, single-blind, parallel-group study in patients with schizophrenia or schizoaffective disorder. Clin Ther. 2010;32(3):472-491.
57. Kovalick LJ, Pikalov AA, Ni N, et al. Short-term physical compatibility of intramuscular aripiprazole with intramuscular lorazepam. Am J Health-Syst Pharm. 2008;65(21):2007-2008.
58. Abilify [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2014.
59. Zyprexa [package insert]. Indianapolis, IN: Lilly Research Laboratories; 2005.
60. Zacher JL, Roche-Desilets J. Hypotension secondary to the combination of intramuscular olanzapine and intramuscular lorazepam. J Clin Psychiatr. 2005;66(12):1614-1615.
61. Marder SR, Sorsaburu S, Dunayevich E, et al. Case reports of postmarketing adverse event experiences with olanzapine intramuscular treatment in patients with agitation. J Clin Psychiatr 2010;71(4):433-441.
62. Wilson MP, MacDonald K, Vilke GM, et al. A comparison of the safety of olanzapine and haloperidol in combination with benzodiazepines in emergency department patients with acute agitation. J Emerg Med. 2012;43(5):790-797.
63. Wilson MP, MacDonald K, Vilke GM, et al. Potential complications of combining intramuscular olanzapine with benzodiazepines in emergency department patients. J Emerg Med. 2012;43(5):889-896.
64. Williams AM. Coadministration of intramuscular olanzapine and benzodiazepines in agitated patients with mental illness. Ment Health Clin [Internet]. 2018;8(5):208-213.
65. Resnick M, Burton BT. Droperidol vs. haloperidol in the initial management of acutely agitated patients. J Clin Psychiatry. 1984;45(7):298-299.
66. Thomas H, Schwartz E, Petrilli R. Droperidol versus haloperidol for chemical restraint of agitated and combative patients. Ann Emerg Med. 1992;21(4):407-413.
67. Richards JR, Derlet RW, Duncan DR. Chemical restraint for the agitated patient in the emergency department: lorazepam versus droperidol. J Emerg Med. 1998;16(4):567-573.
68. Boyer EW. Droperidol is back (and here’s what you need to know). ACEP Now. https://www.acepnow.com/article/droperidol-is-back-and-heres-what-you-need-to-know/. Published September 16, 2019. Accessed April 17, 2020.
69. Martel M, Sterzinger A, Miner J, et al. Management of acute undifferentiated agitation in the emergency department: a randomized double-blind trial of droperidol, ziprasidone, and midazolam. Acad Emerg Med. 2005;12(12):1167-1172.
70. Chan EW, Taylor DM, Knott JC, et al. Intravenous droperidol or olanzapine as an adjunct to midazolam for the acutely agitated patient: a multicenter, randomized, double-blind, placebo-controlled clinical trial. Ann Emerg Med. 2013;61(1):72-81.
71. Isbister GK, Calver LA, Page CB, et al. Randomized controlled trial of intramuscular droperidol versus midazolam for violence and acute behavioral disturbance: the DORM study. Ann Emerg Med. 2010;56(4):392-401.
72. Macht M, Mull AC, McVaney KE, et al. Comparison of droperidol and haloperidol for use by paramedics assessment of safety and effectiveness. Prehosp Emerg Care. 2014;18(3):375-380.
73. Calver L, Page CB, Downes MA, et al. The safety and effectiveness of droperidol for sedation of acute behavioral disturbance in the emergency department. Ann Emerg Med. 2015;66(3):230-238.
74. Kohokar MA, Rathbone J. Droperidol for psychosis-induced aggression or agitation. Cochrane Database Syst Rev. 2016;12:CD002830.
75. Calver L, Drinkwater V, Gupta R, et al. Droperidol v. haloperidol for sedation of aggressive behavior in acute mental health: randomized controlled trial. Brit J Psychiatr. 2015;206(3):223-228.
76. Hopper AB, Vilke GM, Castillo EM, et al. Ketamine use for acute agitation in the emergency department. J Emerg Med. 2015;48(6):712-719.
77. Riddell J, Tran A, Bengiamin R, et al. Ketamine as a first-line treatment for severely agitated emergency department patients. Am J Emerg Med. 2017;35:1000-1004.
78. Lebin JA, Akhavan AR, Hippe DS, et al. Psychiatric outcomes of patients with severe agitation following administration of prehospital ketamine. Acad Emerg Med. 2019;26(8):889-896.
79. Barbic D, Andolfatto G, Grunau B, et al. Rapid agitation control with ketamine in the emergency department (RACKED): a randomized controlled trial protocol. Trials. 2018;19(1):651.
80. Garriga M, Pacchiarotti I, Kasper S, et al. Assessment and management of agitation in psychiatry: expert consensus. World J Biol Psychiatr. 2016;17(2):86-128.
81. Adler L, Angrist B, Peselow E, et al. Efficacy of propranolol in neuroleptic-induced akathesia. J Clin Psychopharmacol. 1985;5(3):164-166.
82. Adler LA, Reiter S, Corwin J, et al. Neuroleptic-induced akathisia: propranolol versus benztropine. Biol Psychiatry. 1988;23(2):211-213.
83. de Leon J, Diaz FJ, Wedlund P, et al. Haloperidol half-life after chronic dosing. J Clin Psychopharmacol. 2004;24(6):656-660.
1. Shorter E. A history of psychiatry. New York, NY: John Wiley & Sons, Inc.; 1997:249.
2. Salzman C, Green AI, Rodriguez-Villa F, et al. Benzodiazepines combined with neuroleptics for management of severe disruptive behavior. Psychosomatics. 1986;27(suppl 1):17-22.
3. Allen MH. Managing the agitated psychotic patient: a reappraisal of the evidence. J Clin Psychiatr. 2000;61(suppl 14):11-20.
4. Salzman C, Solomon D, Miyawaki E, et al. Parenteral lorazepam versus parenteral haloperidol for the control of psychotic disruptive behavior. J Clin Psychiatr. 1991:52(4):177-180.
5. Allen MH, Currier GW, Hughes DH, et al. The expert consensus guideline series: treatment of behavioral emergencies. Postgrad Med. 2001;(Spec No):1-88; quiz 89-90.
6. Foster S, Kessel J, Berman ME, et al. Efficacy of lorazepam and haloperidol for rapid tranquilization in a psychiatric emergency room setting. Int Clin Psychopharmacol. 1997;12(3):175-179.
7. Garza-Trevino WS, Hollister LE, Overall JE, et al. Efficacy of combinations of intramuscular antipsychotics and sedative-hypnotics for control of psychotic agitation. Am J Psychiatr. 1989:146(12):1598-1601.
8. Battaglia J, Moss S, Rush J, et al. Haloperidol, lorazepam, or both for psychotic agitation? A multicenter, prospective double-blind, emergency study. Am J Emerg Med 1997;15(4):335-340.
9. Ostinelli EG, Brooke-Powney MJ, Li X, et al. Haloperidol for psychosis-induced aggression or agitation (rapid tranquillisation). Cochrane Database Syst Rev. 2017; 7:CD009377. doi: 10.1002/14651858.CD009377.pub3.
10. Powney MJ, Adams CE, Jones H. Haloperidol for psychosis-induced aggression or agitation (rapid tranquillisation). Cochrane Database Syst Rev. 2012;11:CD009377. doi: 10.1002/14651858.CD009377.pub2.
11. Citrome L. Review: limited evidence on effects of haloperidol alone for rapid tranquillisation in psychosis-induced aggression. Evid Based Ment Health. 2013;16(2):47.
12. Bienek SA, Ownby R, Penalver A, et al. A double-blind study of lorazepam versus the combination of haloperidol and lorazepam in managing agitation. Pharmacother. 1998;18(1):57-62.
13. Binder RL, McNiel DE. Contemporary practices in managing acutely violent patients in 20 psychiatric emergency rooms. Psychiatric Serv. 1999;50(2):1553-1554.
14. Andrezina R, Josiassen RC, Marcus RN, et al. Intramuscular aripiprazole for the treatment of acute agitation in patients with schizophrenia or schizoaffective disorder: a double-blind, placebo-controlled comparison with intramuscular haloperidol. Psychopharmacology (Berl). 2006;188(3):281-292.
15. Tran-Johnson TK, Sack DA, Marcus RN, et al. Efficacy and safety of intramuscular aripiprazole in patients with acute agitation: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatr. 2007;68(1):111-119.
16. Brook S, Lucey JV, Gunn KP. Intramuscular ziprasidone compared with intramuscular haloperidol in the treatment of acute psychosis. J Clin Psychiatr. 2000;61(12):933-941.
17. Brook S, Walden J, Benattia I, et al. Ziprasidone and haloperidol in the treatment of acute exacerbation of schizophrenia and schizoaffective disorder: comparison of intramuscular and oral formulations in a 6-week, randomized, blinded-assessment study. Psychopharmacology (Berl). 2005;178(4):514-523.
18. Wright P, Birkett M, David SR, et al. Double-blind, placebo-controlled comparison of intramuscular olanzapine and intramuscular haloperidol in the treatment of acute agitation in schizophrenia. Am J Psychiatr. 2001;158(7):1149-1151.
19. Breier A, Meehan K, Birkett M, et al. A double-blind, placebo-controlled dose-response comparison of intramuscular olanzapine and haloperidol in the treatment of acute agitation in schizophrenia. Arch Gen Psych. 2002;59(5):441-448.
20. Hsu W, Huang S, Lee B, et al. Comparison of intramuscular olanzapine, orally disintegrating olanzapine tablets, oral risperidone solution, and intramuscular haloperidol in the management of acute agitation in an acute care psychiatric ward in Taiwan. J Clin Psychopharmacol. 2010;30(3):230-234.
21. Chan H, Ree S, Su L, et al. A double-blind, randomized comparison study of efficacy and safety of intramuscular olanzapine and intramuscular haloperidol in patients with schizophrenia and acute agitated behavior. J Clin Psychopharmacol. 2014;34(3):355-358.
22. Baldaçara L, Sanches M, Cordeiro DC, et al. Rapid tranquilization for agitated patients in emergency psychiatric rooms: a randomized trial of olanzapine, ziprasidone, haloperidol plus promethazine, haloperidol plus midazolam and haloperidol alone. Braz J Psychiatry. 2011;33(1):30-39.
23. Hillard JR. Defusing patient violence. Current Psychiatry. 2002;1(4):22-29.
24. Seemüller F, Schennach R, Mayr A, et al. Akathisia and suicidal ideation in first-episode schizophrenia. J Clin Psychopharmacol. 2012;32(5):694-698.
25. Eikelenboom-Schieveld SJM, Lucire Y, Fogleman JC. The relevance of cytochrome P450 polymorphism in forensic medicine and akathisia-related violence and suicide. J Forens Leg Med. 2016;41:65-71.
26. Van Putten T, May PRA, Marder SR. Akathisia with haloperidol and thiothixene. Arch Gen Psych. 1984;41:1036-1039.
27. Drotts DL, Vinson DR. Prochlorperazine induced akathisia in emergency patients. Ann Emerg Med. 1999;34(4):469-475.
28. Salem H, Negpal C, Pigott T. Revisiting antipsychotic-induced akathisia: current issues and prospective challenges. Curr Neuropharmacol. 2017;15(5):789-798.
29. Huf G, Coutinho ESF, Adams CE. Rapid tranquilization in psychiatric emergency settings in Brazil: pragmatic randomized controlled trial of intramuscular haloperidol versus intramuscular haloperidol plus promethazine. BMJ. 2007;335(7625):869.
30. Mantovani C, Labate CM, Sponholz A, et al. Are low doses of antipsychotics effective in the management of psychomotor agitation? A randomized, rated-blind trial of 4 intramuscular interventions. J Clin Psychopharmacol. 2013;33(3):306-312.
31. Darwish H, Grant R, Haslam R, et al. Promethazine-induced acute dystonic reactions. Am J Dis Child. 1980;134(10):990-991.
32. Jyothi CH, Rudraiah HGM, Vidya HK, et al. Promethazine induced acute dystonia: a case report. Manipal J Med Sci. 2016;1(2):63-64.
33. Ames D, Carr-Lopez SM, Gutierrez MA, et al. Detecting and managing adverse effects of antipsychotic medications: current state of play. Psychiatr Clin North Am. 2016;39(2):275-311.
34. Meyer-Massetti C, Cheng CM, Sharpe MA, et al. The FDA extended warning for intravenous haloperidol and torsades de pointes: how should institutions respond? J Hosp Med. 2010;5(4):E8-E16. doi: 10.1002/jhm.691.
35. Wu C, Tsai Y, Tsai H. Antipsychotic drugs and the risk of ventricular arrhythmia and/or sudden cardiac death: a nation-wide case-crossover study. J Am Heart Dis. 2015;4(2):e001568. doi: 10.1161/JAHA.114.001568.
36. Beach SR, Celano CM, Sugrue AM, et al. QT prolongation, torsades de pointe, and psychotropic medications: a 5-year update. Psychosomatics. 2018;59(1):105-122.
37. Leonard CE, Freeman CP, Newcomb CW, et al. Antipsychotics and the risks of sudden cardiac death and all-cause death: cohort studies in Medicaid and dually-eligible Medicaid-Medicare beneficiaries of five states. J Clin Exp Cardiol. 2013;suppl 10(6):1-9.
38. Nasrallah H, Chen AT. Multiple neurotoxic effects of haloperidol resulting in neuronal death. Ann Clin Psychiatr. 2017;29(3):195-202.
39. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
40. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2013;12(7):7-8.
41. Corrigan PW, Yudofsky SC, Silver JM. Pharmacological and behavioral treatments for aggressive psychiatric inpatients. Hosp Comm Psychiatr. 1993;44(2):125-133.
42. Zeller SL, Citrome L. Managing agitation associated with schizophrenia and bipolar disorder in the emergency setting. West J Emerg Med. 2016;17(2):165-172.
43. Vieta E, Garriga M, Cardete L, et al. Protocol for the management of psychiatric patients with psychomotor agitation. BMC Psychiatr. 2017;17:328.
44. Nobay F, Simon BC, Levitt A, et al. A prospective, double-blind, randomized trial of midazolam versus haloperidol versus lorazepam in the chemical restraint of violent and severely agitated patients. Acad Emerg Med. 2004;11(7):744-749.
45. Klein LR, Driver BE, Miner JR, et al. Intramuscular midazolam, olanzapine, ziprasidone, or haloperidol for treating acute agitation in the emergency department. Ann Emerg Med. 2018;72(4):374-385.
46. Hillard JR. Emergency treatment of acute psychosis. J Clin Psychiatr. 1998;59(suppl 1):57-60.
47. Modell JG, Lenox RH, Weiner S. Inpatient clinical trial of lorazepam for the management of manic agitation. J Clin Psychopharmacol. 1985;5(2):109-110.
48. Denaut M, Yernault JC, De Coster A. Double-blind comparison of the respiratory effects of parenteral lorazepam and diazepam in patients with chronic obstructive lung disease. Curr Med Res Opin. 1975;2(10):611-615.
49. Kahn DR, Barnhorst AV, Bourgeois JA. A case of alcohol withdrawal requiring 1,600 mg of lorazepam in 24 hours. CNS Spectr. 2009;14(7):385-389.
50. Jones KA. Benzodiazepines: their role in aggression and why GPs should prescribe with caution. Austral Fam Physician. 2011;40(11):862-865.
51. Allen MH, Currier GW, Carpenter D, et al. The expert consensus guideline series. Treatment of behavioral emergencies 2005. J Psychiatr Pract. 2005;11(suppl 1):5-108.
52. Allen MH, Carpenter D, Sheets JL, et al. What do consumers say they want and need during a psychiatric emergency? J Psychiatr Pract. 2003;9(1):39-58.
53. Han DH. Some Abilify formulations to discontinue in 2015. MPR. https://www.empr.com/home/news/some-abilify-formulations-to-discontinue-in-2015/. Published January 13, 2015. Accessed April 17, 2020.
54. Citrome L. Comparison of intramuscular ziprasidone, olanzapine, or aripiprazole for agitation: a quantitative review of efficacy and safety. J Clin Psychiatry. 2007;68(12):1876-1885.
55. Satterthwaite TD, Wolf DH, Rosenheck RA, et al. A meta-analysis of the risk of acute extrapyramidal symptoms with intramuscular antipsychotics for the treatment for agitation. J Clin Psychiatr. 2008;69(12):1869-1879.
56. Miceli JJ, Tensfeldt TG, Shiovitz T, et al. Effects of high-dose ziprasidone and haloperidol on the QTc interval after intramuscular administration: a randomized, single-blind, parallel-group study in patients with schizophrenia or schizoaffective disorder. Clin Ther. 2010;32(3):472-491.
57. Kovalick LJ, Pikalov AA, Ni N, et al. Short-term physical compatibility of intramuscular aripiprazole with intramuscular lorazepam. Am J Health-Syst Pharm. 2008;65(21):2007-2008.
58. Abilify [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2014.
59. Zyprexa [package insert]. Indianapolis, IN: Lilly Research Laboratories; 2005.
60. Zacher JL, Roche-Desilets J. Hypotension secondary to the combination of intramuscular olanzapine and intramuscular lorazepam. J Clin Psychiatr. 2005;66(12):1614-1615.
61. Marder SR, Sorsaburu S, Dunayevich E, et al. Case reports of postmarketing adverse event experiences with olanzapine intramuscular treatment in patients with agitation. J Clin Psychiatr 2010;71(4):433-441.
62. Wilson MP, MacDonald K, Vilke GM, et al. A comparison of the safety of olanzapine and haloperidol in combination with benzodiazepines in emergency department patients with acute agitation. J Emerg Med. 2012;43(5):790-797.
63. Wilson MP, MacDonald K, Vilke GM, et al. Potential complications of combining intramuscular olanzapine with benzodiazepines in emergency department patients. J Emerg Med. 2012;43(5):889-896.
64. Williams AM. Coadministration of intramuscular olanzapine and benzodiazepines in agitated patients with mental illness. Ment Health Clin [Internet]. 2018;8(5):208-213.
65. Resnick M, Burton BT. Droperidol vs. haloperidol in the initial management of acutely agitated patients. J Clin Psychiatry. 1984;45(7):298-299.
66. Thomas H, Schwartz E, Petrilli R. Droperidol versus haloperidol for chemical restraint of agitated and combative patients. Ann Emerg Med. 1992;21(4):407-413.
67. Richards JR, Derlet RW, Duncan DR. Chemical restraint for the agitated patient in the emergency department: lorazepam versus droperidol. J Emerg Med. 1998;16(4):567-573.
68. Boyer EW. Droperidol is back (and here’s what you need to know). ACEP Now. https://www.acepnow.com/article/droperidol-is-back-and-heres-what-you-need-to-know/. Published September 16, 2019. Accessed April 17, 2020.
69. Martel M, Sterzinger A, Miner J, et al. Management of acute undifferentiated agitation in the emergency department: a randomized double-blind trial of droperidol, ziprasidone, and midazolam. Acad Emerg Med. 2005;12(12):1167-1172.
70. Chan EW, Taylor DM, Knott JC, et al. Intravenous droperidol or olanzapine as an adjunct to midazolam for the acutely agitated patient: a multicenter, randomized, double-blind, placebo-controlled clinical trial. Ann Emerg Med. 2013;61(1):72-81.
71. Isbister GK, Calver LA, Page CB, et al. Randomized controlled trial of intramuscular droperidol versus midazolam for violence and acute behavioral disturbance: the DORM study. Ann Emerg Med. 2010;56(4):392-401.
72. Macht M, Mull AC, McVaney KE, et al. Comparison of droperidol and haloperidol for use by paramedics assessment of safety and effectiveness. Prehosp Emerg Care. 2014;18(3):375-380.
73. Calver L, Page CB, Downes MA, et al. The safety and effectiveness of droperidol for sedation of acute behavioral disturbance in the emergency department. Ann Emerg Med. 2015;66(3):230-238.
74. Kohokar MA, Rathbone J. Droperidol for psychosis-induced aggression or agitation. Cochrane Database Syst Rev. 2016;12:CD002830.
75. Calver L, Drinkwater V, Gupta R, et al. Droperidol v. haloperidol for sedation of aggressive behavior in acute mental health: randomized controlled trial. Brit J Psychiatr. 2015;206(3):223-228.
76. Hopper AB, Vilke GM, Castillo EM, et al. Ketamine use for acute agitation in the emergency department. J Emerg Med. 2015;48(6):712-719.
77. Riddell J, Tran A, Bengiamin R, et al. Ketamine as a first-line treatment for severely agitated emergency department patients. Am J Emerg Med. 2017;35:1000-1004.
78. Lebin JA, Akhavan AR, Hippe DS, et al. Psychiatric outcomes of patients with severe agitation following administration of prehospital ketamine. Acad Emerg Med. 2019;26(8):889-896.
79. Barbic D, Andolfatto G, Grunau B, et al. Rapid agitation control with ketamine in the emergency department (RACKED): a randomized controlled trial protocol. Trials. 2018;19(1):651.
80. Garriga M, Pacchiarotti I, Kasper S, et al. Assessment and management of agitation in psychiatry: expert consensus. World J Biol Psychiatr. 2016;17(2):86-128.
81. Adler L, Angrist B, Peselow E, et al. Efficacy of propranolol in neuroleptic-induced akathesia. J Clin Psychopharmacol. 1985;5(3):164-166.
82. Adler LA, Reiter S, Corwin J, et al. Neuroleptic-induced akathisia: propranolol versus benztropine. Biol Psychiatry. 1988;23(2):211-213.
83. de Leon J, Diaz FJ, Wedlund P, et al. Haloperidol half-life after chronic dosing. J Clin Psychopharmacol. 2004;24(6):656-660.
Anti-NMDAR encephalitis or primary psychiatric disorder?
New insights and ‘red flags’ provide clues to diagnosis
It remains difficult to distinguish anti-NMDA receptor encephalitis from a primary psychiatric disorder, but recent studies have identified clinical features and proposed screening criteria that could make it easier to identify these patients who would benefit from immunotherapy, according to an expert in the neurologic disease.
Most patients with confirmed anti-NMDA receptor encephalitis will experience substantial improvement after treatment with immunotherapy and other modalities, said Josep Dalmau, MD, PhD, professor at the Catalan Institute for Research and Advanced Studies at the University of Barcelona and adjunct professor of neurology at the University of Pennsylvania, Philadelphia.
“In our experience, being aggressive with immune therapy ... the patients do quite well, which means that basically 85%-90% of the patients substantially improved over the next few months,” Dr. Dalmau said at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Identified for the first time a little more than a decade ago, anti-NMDA receptor encephalitis is a rare, immune-mediated disease that is usually found in children and young adults and is more common among women. It is frequently associated with ovarian tumors and teratomas, said Dr. Dalmau, and in about 90% of cases, patients will have prominent psychiatric and behavioral symptoms.
Patients develop IgG antibodies against the GluN1 subunit of the NMDA receptor. These autoantibodies represent not only a diagnostic marker of the disease, but are also pathogenic, altering NMDA receptor–related synaptic transmission, Dr. Dalmau said.
In several recent studies, investigators have attempted to cobble together a distinct phenotype on anti-NMDA receptor encephalitis to aid psychiatrists who might encounter patients with the disease, he said.
In one of the most recent studies, researchers combed the medical literature and found that, among 544 individuals with the disease, the most common psychiatric symptoms were agitation, seen in 59%, and psychotic symptoms (particularly visual-auditory hallucinations and disorganized behavior) in 54%; catatonia was seen in 42% of adults and 35% of children.
according to a report from researchers in Berlin, Dr. Dalmau added. By picking up on those clinical signs, which included seizures, catatonia, autonomic instability, or hyperkinesia, the time from symptom onset to diagnosis could be cut in half, the researchers found.
There’s also a handy acronym that could serve as a mnemonic to pick up on “diagnostic clues” of anti-NMDA receptor encephalitis among patients with new-onset psychiatric symptoms, Dr. Dalmau said.
That acronym, published in a review article by Dr. Dalmau and colleagues, is SEARCH For NMDAR-A, covering, in order: sleep dysfunction, excitement, agitation, rapid onset, child and young adult predominance, history of psychiatric disease (absent), fluctuating catatonia, negative and positive symptoms, memory deficit, decreased verbal output, antipsychotic intolerance, rule out neuroleptic malignant syndrome, and of course, antibodies (though the final “A” also stands for additional testing, including magnetic resonance imaging, cerebrospinal fluid studies, and electroencephalogram).
While the disease can be lethal, Dr. Dalmau said most patients respond to immunotherapy, and if applicable, treatment of the underlying tumor can help. The most common first-line treatments include steroids, intravenous immunoglobulin, and plasma exchange, he said, while second-line treatments include the monoclonal anti-CD20 antibody rituximab and cyclophosphamide.
Beyond immunotherapy, patients may benefit from supportive care and psychiatric treatment. Benzodiazepines are well tolerated, but Dr. Dalmau said antipsychotic intolerance is frequent, and electroconvulsive therapy has “mixed results” in these patients.
The recovery process can take months and may be complicated by hypersomnia, hyperphagia, and hypersexuality, he added.
“Some patients improve dramatically in 1 month, but this is uncommon, really,” he said, adding that an early recovery may be a “red flag” that the underlying condition is something other than anti-NMDA receptor encephalitis.
Dr. Dalmau provided disclosures related to Cellex Foundation, Safra Foundation, Caixa Health Project Foundation, and Sage Therapeutics.
SOURCE: Dalmau J. APA 2020, Abstract.
New insights and ‘red flags’ provide clues to diagnosis
New insights and ‘red flags’ provide clues to diagnosis
It remains difficult to distinguish anti-NMDA receptor encephalitis from a primary psychiatric disorder, but recent studies have identified clinical features and proposed screening criteria that could make it easier to identify these patients who would benefit from immunotherapy, according to an expert in the neurologic disease.
Most patients with confirmed anti-NMDA receptor encephalitis will experience substantial improvement after treatment with immunotherapy and other modalities, said Josep Dalmau, MD, PhD, professor at the Catalan Institute for Research and Advanced Studies at the University of Barcelona and adjunct professor of neurology at the University of Pennsylvania, Philadelphia.
“In our experience, being aggressive with immune therapy ... the patients do quite well, which means that basically 85%-90% of the patients substantially improved over the next few months,” Dr. Dalmau said at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Identified for the first time a little more than a decade ago, anti-NMDA receptor encephalitis is a rare, immune-mediated disease that is usually found in children and young adults and is more common among women. It is frequently associated with ovarian tumors and teratomas, said Dr. Dalmau, and in about 90% of cases, patients will have prominent psychiatric and behavioral symptoms.
Patients develop IgG antibodies against the GluN1 subunit of the NMDA receptor. These autoantibodies represent not only a diagnostic marker of the disease, but are also pathogenic, altering NMDA receptor–related synaptic transmission, Dr. Dalmau said.
In several recent studies, investigators have attempted to cobble together a distinct phenotype on anti-NMDA receptor encephalitis to aid psychiatrists who might encounter patients with the disease, he said.
In one of the most recent studies, researchers combed the medical literature and found that, among 544 individuals with the disease, the most common psychiatric symptoms were agitation, seen in 59%, and psychotic symptoms (particularly visual-auditory hallucinations and disorganized behavior) in 54%; catatonia was seen in 42% of adults and 35% of children.
according to a report from researchers in Berlin, Dr. Dalmau added. By picking up on those clinical signs, which included seizures, catatonia, autonomic instability, or hyperkinesia, the time from symptom onset to diagnosis could be cut in half, the researchers found.
There’s also a handy acronym that could serve as a mnemonic to pick up on “diagnostic clues” of anti-NMDA receptor encephalitis among patients with new-onset psychiatric symptoms, Dr. Dalmau said.
That acronym, published in a review article by Dr. Dalmau and colleagues, is SEARCH For NMDAR-A, covering, in order: sleep dysfunction, excitement, agitation, rapid onset, child and young adult predominance, history of psychiatric disease (absent), fluctuating catatonia, negative and positive symptoms, memory deficit, decreased verbal output, antipsychotic intolerance, rule out neuroleptic malignant syndrome, and of course, antibodies (though the final “A” also stands for additional testing, including magnetic resonance imaging, cerebrospinal fluid studies, and electroencephalogram).
While the disease can be lethal, Dr. Dalmau said most patients respond to immunotherapy, and if applicable, treatment of the underlying tumor can help. The most common first-line treatments include steroids, intravenous immunoglobulin, and plasma exchange, he said, while second-line treatments include the monoclonal anti-CD20 antibody rituximab and cyclophosphamide.
Beyond immunotherapy, patients may benefit from supportive care and psychiatric treatment. Benzodiazepines are well tolerated, but Dr. Dalmau said antipsychotic intolerance is frequent, and electroconvulsive therapy has “mixed results” in these patients.
The recovery process can take months and may be complicated by hypersomnia, hyperphagia, and hypersexuality, he added.
“Some patients improve dramatically in 1 month, but this is uncommon, really,” he said, adding that an early recovery may be a “red flag” that the underlying condition is something other than anti-NMDA receptor encephalitis.
Dr. Dalmau provided disclosures related to Cellex Foundation, Safra Foundation, Caixa Health Project Foundation, and Sage Therapeutics.
SOURCE: Dalmau J. APA 2020, Abstract.
It remains difficult to distinguish anti-NMDA receptor encephalitis from a primary psychiatric disorder, but recent studies have identified clinical features and proposed screening criteria that could make it easier to identify these patients who would benefit from immunotherapy, according to an expert in the neurologic disease.
Most patients with confirmed anti-NMDA receptor encephalitis will experience substantial improvement after treatment with immunotherapy and other modalities, said Josep Dalmau, MD, PhD, professor at the Catalan Institute for Research and Advanced Studies at the University of Barcelona and adjunct professor of neurology at the University of Pennsylvania, Philadelphia.
“In our experience, being aggressive with immune therapy ... the patients do quite well, which means that basically 85%-90% of the patients substantially improved over the next few months,” Dr. Dalmau said at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Identified for the first time a little more than a decade ago, anti-NMDA receptor encephalitis is a rare, immune-mediated disease that is usually found in children and young adults and is more common among women. It is frequently associated with ovarian tumors and teratomas, said Dr. Dalmau, and in about 90% of cases, patients will have prominent psychiatric and behavioral symptoms.
Patients develop IgG antibodies against the GluN1 subunit of the NMDA receptor. These autoantibodies represent not only a diagnostic marker of the disease, but are also pathogenic, altering NMDA receptor–related synaptic transmission, Dr. Dalmau said.
In several recent studies, investigators have attempted to cobble together a distinct phenotype on anti-NMDA receptor encephalitis to aid psychiatrists who might encounter patients with the disease, he said.
In one of the most recent studies, researchers combed the medical literature and found that, among 544 individuals with the disease, the most common psychiatric symptoms were agitation, seen in 59%, and psychotic symptoms (particularly visual-auditory hallucinations and disorganized behavior) in 54%; catatonia was seen in 42% of adults and 35% of children.
according to a report from researchers in Berlin, Dr. Dalmau added. By picking up on those clinical signs, which included seizures, catatonia, autonomic instability, or hyperkinesia, the time from symptom onset to diagnosis could be cut in half, the researchers found.
There’s also a handy acronym that could serve as a mnemonic to pick up on “diagnostic clues” of anti-NMDA receptor encephalitis among patients with new-onset psychiatric symptoms, Dr. Dalmau said.
That acronym, published in a review article by Dr. Dalmau and colleagues, is SEARCH For NMDAR-A, covering, in order: sleep dysfunction, excitement, agitation, rapid onset, child and young adult predominance, history of psychiatric disease (absent), fluctuating catatonia, negative and positive symptoms, memory deficit, decreased verbal output, antipsychotic intolerance, rule out neuroleptic malignant syndrome, and of course, antibodies (though the final “A” also stands for additional testing, including magnetic resonance imaging, cerebrospinal fluid studies, and electroencephalogram).
While the disease can be lethal, Dr. Dalmau said most patients respond to immunotherapy, and if applicable, treatment of the underlying tumor can help. The most common first-line treatments include steroids, intravenous immunoglobulin, and plasma exchange, he said, while second-line treatments include the monoclonal anti-CD20 antibody rituximab and cyclophosphamide.
Beyond immunotherapy, patients may benefit from supportive care and psychiatric treatment. Benzodiazepines are well tolerated, but Dr. Dalmau said antipsychotic intolerance is frequent, and electroconvulsive therapy has “mixed results” in these patients.
The recovery process can take months and may be complicated by hypersomnia, hyperphagia, and hypersexuality, he added.
“Some patients improve dramatically in 1 month, but this is uncommon, really,” he said, adding that an early recovery may be a “red flag” that the underlying condition is something other than anti-NMDA receptor encephalitis.
Dr. Dalmau provided disclosures related to Cellex Foundation, Safra Foundation, Caixa Health Project Foundation, and Sage Therapeutics.
SOURCE: Dalmau J. APA 2020, Abstract.
FROM APA 2020
Drive-up pharmacotherapy during the COVID-19 pandemic
My medical career began during a tragedy. I started medical school in August 2001 at New York University, a few dozen blocks north of the World Trade Center in Manhattan. Several weeks later, the September 11 terrorist attacks devastated the city, and the rest of our country. Though we knew virtually nothing yet about practicing medicine, my entire class put on our scrubs and ran to the Bellevue Hospital emergency department to see if there was anything we could do to help. In the end, there was not much we could do that day, but the experience seared into us the notion that a physician stands tall in a crisis and does whatever it takes to help.
For me, the recent emergency we are facing with the coronavirus disease 2019 (COVID-19) pandemic has brought back bone-chilling memories of that time, especially because New York City has been one of the hardest-hit cities in the world. It’s hard for anyone to change routines on a dime, but I’m fortunate to run a solo private practice with a small administrative staff. I was able to pivot my medication management and therapy patients to 100% telepsychiatry overnight, even though I quite dislike the emotional distancing that the physical separation creates. However, I do administer some treatments that require my patients’ physical presence: long-acting injectable (LAI) antipsychotics, and intranasal esketamine. I consider both to be life-saving interventions, so I had to figure out how to continue offering those services while doing my part to keep everyone healthy.
Drive-up LAI antipsychotics
Many of my patients who receive LAIs are on formulations that are injected into the deltoid, so I transitioned to having them drive up to the front door of my office and roll up their sleeve so I could administer the injection without them leaving their car. If it was possible to convert a monthly deltoid injection to an equivalent quarterly deltoid injection, I accelerated that process. It took a little more thought to figure out how to best manage patients who had been getting gluteal injections. Deltoid injections are more convenient, but for certain antipsychotics, the only available LAI formulations that allow intervals longer than 1 month require gluteal administration due to the injection volume and pharmacokinetic considerations. Because of privacy and safety considerations, I didn’t feel gluteal injections would be feasible or appropriate for drive-up administration.
Maintaining patients on their gluteal injections would provide a longer duration between doses, but because patients would have to come inside the office to get them, there would be a higher risk of COVID-19 transmission. Converting them to a once-monthly equivalent with the same molecule and comparable dosage given in the deltoid via drive-up would reduce the risk of viral transmission, but requiring more frequent injections would increase the likelihood they might not show up for all doses during this crisis. I spoke with several other psychiatrists about this dilemma, and several of them favored lengthening the injection cycle as the top priority during this time. However, given the exponential curve of viral transmission in a pandemic, time is of the essence to “flatten the curve.” I decided that prioritizing the reduction of infection risk was paramount, and so I began switching my patients receiving gluteal injections with a longer duration to deltoid injections with a shorter duration. I can only hope I made the right decision for my patients, staff, and family.
Drive-up esketamine
Then came the hardest question—how do I continue to provide intranasal esketamine to my patients? There is an (appropriately) rigid Risk Evaluation and Mitigation Strategy protocol in place that requires patients to be monitored in a medically supervised health care setting for 2 hours after receiving esketamine. Having a patient in the office for at least 2 hours would create a tremendous risk for viral transmission, even in the best-case scenario of using personal protective equipment and stringent efforts to sterilize the space. I didn’t consider putting the treatments on hold because esketamine is indicated solely for patients with treatment-resistant depression, and these patients couldn’t be effectively managed with conventional oral antidepressants. I decided I’d have to figure out a way to adapt the drive-up LAI administration process for esketamine treatments as well.
In my practice, esketamine monitoring usually occurs in a treatment room that has a back entrance to a small, private parking lot. I realized that if I had the patients pull around the building and park in the spot right outside the window, we could maintain direct observation from inside the office while they sat in their car! Patients are not permitted to drive after receiving an esketamine treatment, so we take possession of their car keys to prevent them from driving off before the end of the monitoring period. We give them one of our automatic blood pressure cuffs to take the required blood pressure readings, and they relay the results through a video telemedicine connection. We also enlist the patient’s designated driver to provide an additional set of eyes for monitoring. When the observation period ends, the cuff is retrieved and sanitized.
Meeting our patients’ needs
Our duty to our patients is vital during a crisis, and they deserve everything in our power that we can offer them. We can’t be complacent in our routines and let our fears of what might or might not happen paralyze us from moving forward. If we are flexible and creative, we can rise to overcome any challenge to meeting our patients’ needs. Throughout this ordeal, I’ve seen some of the patients I was most worried about turn out to be some of the most resilient. When our patients have risen to the occasion, what excuse do we have not to do the same?
My medical career began during a tragedy. I started medical school in August 2001 at New York University, a few dozen blocks north of the World Trade Center in Manhattan. Several weeks later, the September 11 terrorist attacks devastated the city, and the rest of our country. Though we knew virtually nothing yet about practicing medicine, my entire class put on our scrubs and ran to the Bellevue Hospital emergency department to see if there was anything we could do to help. In the end, there was not much we could do that day, but the experience seared into us the notion that a physician stands tall in a crisis and does whatever it takes to help.
For me, the recent emergency we are facing with the coronavirus disease 2019 (COVID-19) pandemic has brought back bone-chilling memories of that time, especially because New York City has been one of the hardest-hit cities in the world. It’s hard for anyone to change routines on a dime, but I’m fortunate to run a solo private practice with a small administrative staff. I was able to pivot my medication management and therapy patients to 100% telepsychiatry overnight, even though I quite dislike the emotional distancing that the physical separation creates. However, I do administer some treatments that require my patients’ physical presence: long-acting injectable (LAI) antipsychotics, and intranasal esketamine. I consider both to be life-saving interventions, so I had to figure out how to continue offering those services while doing my part to keep everyone healthy.
Drive-up LAI antipsychotics
Many of my patients who receive LAIs are on formulations that are injected into the deltoid, so I transitioned to having them drive up to the front door of my office and roll up their sleeve so I could administer the injection without them leaving their car. If it was possible to convert a monthly deltoid injection to an equivalent quarterly deltoid injection, I accelerated that process. It took a little more thought to figure out how to best manage patients who had been getting gluteal injections. Deltoid injections are more convenient, but for certain antipsychotics, the only available LAI formulations that allow intervals longer than 1 month require gluteal administration due to the injection volume and pharmacokinetic considerations. Because of privacy and safety considerations, I didn’t feel gluteal injections would be feasible or appropriate for drive-up administration.
Maintaining patients on their gluteal injections would provide a longer duration between doses, but because patients would have to come inside the office to get them, there would be a higher risk of COVID-19 transmission. Converting them to a once-monthly equivalent with the same molecule and comparable dosage given in the deltoid via drive-up would reduce the risk of viral transmission, but requiring more frequent injections would increase the likelihood they might not show up for all doses during this crisis. I spoke with several other psychiatrists about this dilemma, and several of them favored lengthening the injection cycle as the top priority during this time. However, given the exponential curve of viral transmission in a pandemic, time is of the essence to “flatten the curve.” I decided that prioritizing the reduction of infection risk was paramount, and so I began switching my patients receiving gluteal injections with a longer duration to deltoid injections with a shorter duration. I can only hope I made the right decision for my patients, staff, and family.
Drive-up esketamine
Then came the hardest question—how do I continue to provide intranasal esketamine to my patients? There is an (appropriately) rigid Risk Evaluation and Mitigation Strategy protocol in place that requires patients to be monitored in a medically supervised health care setting for 2 hours after receiving esketamine. Having a patient in the office for at least 2 hours would create a tremendous risk for viral transmission, even in the best-case scenario of using personal protective equipment and stringent efforts to sterilize the space. I didn’t consider putting the treatments on hold because esketamine is indicated solely for patients with treatment-resistant depression, and these patients couldn’t be effectively managed with conventional oral antidepressants. I decided I’d have to figure out a way to adapt the drive-up LAI administration process for esketamine treatments as well.
In my practice, esketamine monitoring usually occurs in a treatment room that has a back entrance to a small, private parking lot. I realized that if I had the patients pull around the building and park in the spot right outside the window, we could maintain direct observation from inside the office while they sat in their car! Patients are not permitted to drive after receiving an esketamine treatment, so we take possession of their car keys to prevent them from driving off before the end of the monitoring period. We give them one of our automatic blood pressure cuffs to take the required blood pressure readings, and they relay the results through a video telemedicine connection. We also enlist the patient’s designated driver to provide an additional set of eyes for monitoring. When the observation period ends, the cuff is retrieved and sanitized.
Meeting our patients’ needs
Our duty to our patients is vital during a crisis, and they deserve everything in our power that we can offer them. We can’t be complacent in our routines and let our fears of what might or might not happen paralyze us from moving forward. If we are flexible and creative, we can rise to overcome any challenge to meeting our patients’ needs. Throughout this ordeal, I’ve seen some of the patients I was most worried about turn out to be some of the most resilient. When our patients have risen to the occasion, what excuse do we have not to do the same?
My medical career began during a tragedy. I started medical school in August 2001 at New York University, a few dozen blocks north of the World Trade Center in Manhattan. Several weeks later, the September 11 terrorist attacks devastated the city, and the rest of our country. Though we knew virtually nothing yet about practicing medicine, my entire class put on our scrubs and ran to the Bellevue Hospital emergency department to see if there was anything we could do to help. In the end, there was not much we could do that day, but the experience seared into us the notion that a physician stands tall in a crisis and does whatever it takes to help.
For me, the recent emergency we are facing with the coronavirus disease 2019 (COVID-19) pandemic has brought back bone-chilling memories of that time, especially because New York City has been one of the hardest-hit cities in the world. It’s hard for anyone to change routines on a dime, but I’m fortunate to run a solo private practice with a small administrative staff. I was able to pivot my medication management and therapy patients to 100% telepsychiatry overnight, even though I quite dislike the emotional distancing that the physical separation creates. However, I do administer some treatments that require my patients’ physical presence: long-acting injectable (LAI) antipsychotics, and intranasal esketamine. I consider both to be life-saving interventions, so I had to figure out how to continue offering those services while doing my part to keep everyone healthy.
Drive-up LAI antipsychotics
Many of my patients who receive LAIs are on formulations that are injected into the deltoid, so I transitioned to having them drive up to the front door of my office and roll up their sleeve so I could administer the injection without them leaving their car. If it was possible to convert a monthly deltoid injection to an equivalent quarterly deltoid injection, I accelerated that process. It took a little more thought to figure out how to best manage patients who had been getting gluteal injections. Deltoid injections are more convenient, but for certain antipsychotics, the only available LAI formulations that allow intervals longer than 1 month require gluteal administration due to the injection volume and pharmacokinetic considerations. Because of privacy and safety considerations, I didn’t feel gluteal injections would be feasible or appropriate for drive-up administration.
Maintaining patients on their gluteal injections would provide a longer duration between doses, but because patients would have to come inside the office to get them, there would be a higher risk of COVID-19 transmission. Converting them to a once-monthly equivalent with the same molecule and comparable dosage given in the deltoid via drive-up would reduce the risk of viral transmission, but requiring more frequent injections would increase the likelihood they might not show up for all doses during this crisis. I spoke with several other psychiatrists about this dilemma, and several of them favored lengthening the injection cycle as the top priority during this time. However, given the exponential curve of viral transmission in a pandemic, time is of the essence to “flatten the curve.” I decided that prioritizing the reduction of infection risk was paramount, and so I began switching my patients receiving gluteal injections with a longer duration to deltoid injections with a shorter duration. I can only hope I made the right decision for my patients, staff, and family.
Drive-up esketamine
Then came the hardest question—how do I continue to provide intranasal esketamine to my patients? There is an (appropriately) rigid Risk Evaluation and Mitigation Strategy protocol in place that requires patients to be monitored in a medically supervised health care setting for 2 hours after receiving esketamine. Having a patient in the office for at least 2 hours would create a tremendous risk for viral transmission, even in the best-case scenario of using personal protective equipment and stringent efforts to sterilize the space. I didn’t consider putting the treatments on hold because esketamine is indicated solely for patients with treatment-resistant depression, and these patients couldn’t be effectively managed with conventional oral antidepressants. I decided I’d have to figure out a way to adapt the drive-up LAI administration process for esketamine treatments as well.
In my practice, esketamine monitoring usually occurs in a treatment room that has a back entrance to a small, private parking lot. I realized that if I had the patients pull around the building and park in the spot right outside the window, we could maintain direct observation from inside the office while they sat in their car! Patients are not permitted to drive after receiving an esketamine treatment, so we take possession of their car keys to prevent them from driving off before the end of the monitoring period. We give them one of our automatic blood pressure cuffs to take the required blood pressure readings, and they relay the results through a video telemedicine connection. We also enlist the patient’s designated driver to provide an additional set of eyes for monitoring. When the observation period ends, the cuff is retrieved and sanitized.
Meeting our patients’ needs
Our duty to our patients is vital during a crisis, and they deserve everything in our power that we can offer them. We can’t be complacent in our routines and let our fears of what might or might not happen paralyze us from moving forward. If we are flexible and creative, we can rise to overcome any challenge to meeting our patients’ needs. Throughout this ordeal, I’ve seen some of the patients I was most worried about turn out to be some of the most resilient. When our patients have risen to the occasion, what excuse do we have not to do the same?
First-in-class schizophrenia drug safe, effective, durable
A novel first-in-class antipsychotic that has a completely different mechanism of action than other currently available agents shows long-term efficacy and a good safety profile in patients with schizophrenia.
SEP-363856 (Sunovion Pharmaceuticals) was described as a potential “game changer” for schizophrenia after initial results were presented at the American Psychiatric Association 2019 Annual Meeting. The drug has been granted breakthrough therapy designation by the Food and Drug Administration.
Additional data from this study, as well as results from a 6-month open-label extension trial were published April 15 in The New England Journal of Medicine.
“This is the first safety and effectiveness data for 6 months on therapy,” lead author Kenneth Koblan, PhD, chief scientific officer at Sunovion, said in an interview.
To be able to show “durable” benefit and that the drug is “benign with respect to weight gain, lipids, and extrapyramidal side effects really provides us with a view of what could be a whole new class of molecules with respect to safety and efficacy,” Dr. Koblan said.
Clinically meaningful improvement
Although the exact mechanism of action is unknown, It does not bind to dopamine 2 (D2) or serotonin 2A (5-HT2A) receptors, which are thought to mediate the effects of currently available antipsychotics.
The original SEP 361-201 study was a phase 2, 4-week, double-blind, placebo-controlled trial comprising 245 adults hospitalized with an acute exacerbation of schizophrenia. Patients were randomly allocated to receive SEP-363856 (50 mg or 75 mg once daily) or placebo.
Demographics of the treatment groups were similar at baseline. The SEP-363856 group included 120 patients (64% men; mean age, 30.0 years; Positive and Negative Syndrome Scale [PANSS] total score, 101.4). The placebo group included 125 patients (63% men; mean age, 30.6 years; PANSS total score, 99.7).
After 4 weeks of treatment, patients taking SEP-363856 showed statistically significant and clinically meaningful improvement in PANSS total scores (the primary outcome), compared with patients taking placebo (–17.2 vs –9.7; P = .001; effect size = 0.45).
SEP-363856 also led to significant improvement in several secondary outcomes, including overall illness severity as judged by the Clinical Global Impression of Severity score, PANSS positive subscale score, PANSS negative subscale score, and PANSS general psychopathology subscale score.
A total of 156 patients, representing 81% of those who completed the 4-week trial, were enrolled in an extension study. They received treatment with SEP-363856 for 26 weeks.
Among 77 patients who had initially been assigned to SEP-363856 in the double-blind trial and then continued to receive treatment in the open-label extension, the mean change in PANSS total score from extension-study baseline to week 26 was −17.1 points.
Among 79 patients who had initially been assigned to placebo and then switched to open-label SEP-363856, the mean change in PANSS total score was −27.9 points.
Safety and tolerability of SEP-363856 were generally similar to placebo.
The active treatment and placebo arms were similar in number of patients who reported extrapyramidal symptoms (3.3% vs. 3.2%, respectively) and who used medications to treat extrapyramidal symptoms. The findings were also similar on movement disorder scales.
In addition, the novel drug had minimal effects on the protein hormone prolactin. “These findings are consistent with the absence of D2-receptor binding for SEP-363856,” the investigators wrote.
Adverse events (AEs) occurring with an incidence of at least 2%, and with a higher incidence in SEP-363856 than placebo, were somnolence (6.7% vs. 4.8%, respectively), agitation (5% vs. 4.8%), nausea (5% vs. 3.2%), diarrhea (2.5% vs. 0.8%), and dyspepsia (2.5% vs. 0%).
Two serious AEs that occurred in the SEP-363856 arm were worsening of schizophrenia and acute cardiovascular insufficiency in a 37-year-old woman, which led to her death 1 week after taking the first 50-mg dose of treatment. However, the woman had a history of essential hypertension and, at autopsy, was found to have coronary artery disease and pulmonary embolism.
A large phase 3 program is now underway to confirm safety and efficacy of SEP-363856 for schizophrenia, Dr. Koblan said. It includes four studies that are open and enrolling.
Benefit beyond psychosis?
In an accompanying editorial , Donald C. Goff, MD, of the Nathan Kline Institute at New York University, wrote that if the findings were replicated in future trials with more patients and of longer duration, “this class of drugs may provide a valuable new therapeutic option for psychosis and the negative symptoms of schizophrenia, possibly without the adverse effects associated with direct D2-receptor antagonism.”
It is also possible that SEP-363856 will have effects “beyond D2-receptor antagonists and hence beyond the treatment of psychosis. Other potential therapeutic indications, including cognitive impairment, depression, and substance abuse, remain to be explored,” Dr. Goff wrote.
This study “not only introduces a promising therapeutic compound but also provides additional evidence, on the basis of an agnostic drug-discovery process, that TAAR1 is a promising target,” he added.
“This is very welcome news given the great need for new pharmacologic treatments for patients with schizophrenia,” Dr. Goff concluded.
The study was funded by Sunovion Pharmaceuticals. Dr. Koblan is an employee of the company. Disclosures for the other authors are listed in the original article. Dr. Goff reported having received grants and nonfinancial support from Avanir Pharmaceuticals and from Takeda.
A version of this article first appeared on Medscape.com.
A novel first-in-class antipsychotic that has a completely different mechanism of action than other currently available agents shows long-term efficacy and a good safety profile in patients with schizophrenia.
SEP-363856 (Sunovion Pharmaceuticals) was described as a potential “game changer” for schizophrenia after initial results were presented at the American Psychiatric Association 2019 Annual Meeting. The drug has been granted breakthrough therapy designation by the Food and Drug Administration.
Additional data from this study, as well as results from a 6-month open-label extension trial were published April 15 in The New England Journal of Medicine.
“This is the first safety and effectiveness data for 6 months on therapy,” lead author Kenneth Koblan, PhD, chief scientific officer at Sunovion, said in an interview.
To be able to show “durable” benefit and that the drug is “benign with respect to weight gain, lipids, and extrapyramidal side effects really provides us with a view of what could be a whole new class of molecules with respect to safety and efficacy,” Dr. Koblan said.
Clinically meaningful improvement
Although the exact mechanism of action is unknown, It does not bind to dopamine 2 (D2) or serotonin 2A (5-HT2A) receptors, which are thought to mediate the effects of currently available antipsychotics.
The original SEP 361-201 study was a phase 2, 4-week, double-blind, placebo-controlled trial comprising 245 adults hospitalized with an acute exacerbation of schizophrenia. Patients were randomly allocated to receive SEP-363856 (50 mg or 75 mg once daily) or placebo.
Demographics of the treatment groups were similar at baseline. The SEP-363856 group included 120 patients (64% men; mean age, 30.0 years; Positive and Negative Syndrome Scale [PANSS] total score, 101.4). The placebo group included 125 patients (63% men; mean age, 30.6 years; PANSS total score, 99.7).
After 4 weeks of treatment, patients taking SEP-363856 showed statistically significant and clinically meaningful improvement in PANSS total scores (the primary outcome), compared with patients taking placebo (–17.2 vs –9.7; P = .001; effect size = 0.45).
SEP-363856 also led to significant improvement in several secondary outcomes, including overall illness severity as judged by the Clinical Global Impression of Severity score, PANSS positive subscale score, PANSS negative subscale score, and PANSS general psychopathology subscale score.
A total of 156 patients, representing 81% of those who completed the 4-week trial, were enrolled in an extension study. They received treatment with SEP-363856 for 26 weeks.
Among 77 patients who had initially been assigned to SEP-363856 in the double-blind trial and then continued to receive treatment in the open-label extension, the mean change in PANSS total score from extension-study baseline to week 26 was −17.1 points.
Among 79 patients who had initially been assigned to placebo and then switched to open-label SEP-363856, the mean change in PANSS total score was −27.9 points.
Safety and tolerability of SEP-363856 were generally similar to placebo.
The active treatment and placebo arms were similar in number of patients who reported extrapyramidal symptoms (3.3% vs. 3.2%, respectively) and who used medications to treat extrapyramidal symptoms. The findings were also similar on movement disorder scales.
In addition, the novel drug had minimal effects on the protein hormone prolactin. “These findings are consistent with the absence of D2-receptor binding for SEP-363856,” the investigators wrote.
Adverse events (AEs) occurring with an incidence of at least 2%, and with a higher incidence in SEP-363856 than placebo, were somnolence (6.7% vs. 4.8%, respectively), agitation (5% vs. 4.8%), nausea (5% vs. 3.2%), diarrhea (2.5% vs. 0.8%), and dyspepsia (2.5% vs. 0%).
Two serious AEs that occurred in the SEP-363856 arm were worsening of schizophrenia and acute cardiovascular insufficiency in a 37-year-old woman, which led to her death 1 week after taking the first 50-mg dose of treatment. However, the woman had a history of essential hypertension and, at autopsy, was found to have coronary artery disease and pulmonary embolism.
A large phase 3 program is now underway to confirm safety and efficacy of SEP-363856 for schizophrenia, Dr. Koblan said. It includes four studies that are open and enrolling.
Benefit beyond psychosis?
In an accompanying editorial , Donald C. Goff, MD, of the Nathan Kline Institute at New York University, wrote that if the findings were replicated in future trials with more patients and of longer duration, “this class of drugs may provide a valuable new therapeutic option for psychosis and the negative symptoms of schizophrenia, possibly without the adverse effects associated with direct D2-receptor antagonism.”
It is also possible that SEP-363856 will have effects “beyond D2-receptor antagonists and hence beyond the treatment of psychosis. Other potential therapeutic indications, including cognitive impairment, depression, and substance abuse, remain to be explored,” Dr. Goff wrote.
This study “not only introduces a promising therapeutic compound but also provides additional evidence, on the basis of an agnostic drug-discovery process, that TAAR1 is a promising target,” he added.
“This is very welcome news given the great need for new pharmacologic treatments for patients with schizophrenia,” Dr. Goff concluded.
The study was funded by Sunovion Pharmaceuticals. Dr. Koblan is an employee of the company. Disclosures for the other authors are listed in the original article. Dr. Goff reported having received grants and nonfinancial support from Avanir Pharmaceuticals and from Takeda.
A version of this article first appeared on Medscape.com.
A novel first-in-class antipsychotic that has a completely different mechanism of action than other currently available agents shows long-term efficacy and a good safety profile in patients with schizophrenia.
SEP-363856 (Sunovion Pharmaceuticals) was described as a potential “game changer” for schizophrenia after initial results were presented at the American Psychiatric Association 2019 Annual Meeting. The drug has been granted breakthrough therapy designation by the Food and Drug Administration.
Additional data from this study, as well as results from a 6-month open-label extension trial were published April 15 in The New England Journal of Medicine.
“This is the first safety and effectiveness data for 6 months on therapy,” lead author Kenneth Koblan, PhD, chief scientific officer at Sunovion, said in an interview.
To be able to show “durable” benefit and that the drug is “benign with respect to weight gain, lipids, and extrapyramidal side effects really provides us with a view of what could be a whole new class of molecules with respect to safety and efficacy,” Dr. Koblan said.
Clinically meaningful improvement
Although the exact mechanism of action is unknown, It does not bind to dopamine 2 (D2) or serotonin 2A (5-HT2A) receptors, which are thought to mediate the effects of currently available antipsychotics.
The original SEP 361-201 study was a phase 2, 4-week, double-blind, placebo-controlled trial comprising 245 adults hospitalized with an acute exacerbation of schizophrenia. Patients were randomly allocated to receive SEP-363856 (50 mg or 75 mg once daily) or placebo.
Demographics of the treatment groups were similar at baseline. The SEP-363856 group included 120 patients (64% men; mean age, 30.0 years; Positive and Negative Syndrome Scale [PANSS] total score, 101.4). The placebo group included 125 patients (63% men; mean age, 30.6 years; PANSS total score, 99.7).
After 4 weeks of treatment, patients taking SEP-363856 showed statistically significant and clinically meaningful improvement in PANSS total scores (the primary outcome), compared with patients taking placebo (–17.2 vs –9.7; P = .001; effect size = 0.45).
SEP-363856 also led to significant improvement in several secondary outcomes, including overall illness severity as judged by the Clinical Global Impression of Severity score, PANSS positive subscale score, PANSS negative subscale score, and PANSS general psychopathology subscale score.
A total of 156 patients, representing 81% of those who completed the 4-week trial, were enrolled in an extension study. They received treatment with SEP-363856 for 26 weeks.
Among 77 patients who had initially been assigned to SEP-363856 in the double-blind trial and then continued to receive treatment in the open-label extension, the mean change in PANSS total score from extension-study baseline to week 26 was −17.1 points.
Among 79 patients who had initially been assigned to placebo and then switched to open-label SEP-363856, the mean change in PANSS total score was −27.9 points.
Safety and tolerability of SEP-363856 were generally similar to placebo.
The active treatment and placebo arms were similar in number of patients who reported extrapyramidal symptoms (3.3% vs. 3.2%, respectively) and who used medications to treat extrapyramidal symptoms. The findings were also similar on movement disorder scales.
In addition, the novel drug had minimal effects on the protein hormone prolactin. “These findings are consistent with the absence of D2-receptor binding for SEP-363856,” the investigators wrote.
Adverse events (AEs) occurring with an incidence of at least 2%, and with a higher incidence in SEP-363856 than placebo, were somnolence (6.7% vs. 4.8%, respectively), agitation (5% vs. 4.8%), nausea (5% vs. 3.2%), diarrhea (2.5% vs. 0.8%), and dyspepsia (2.5% vs. 0%).
Two serious AEs that occurred in the SEP-363856 arm were worsening of schizophrenia and acute cardiovascular insufficiency in a 37-year-old woman, which led to her death 1 week after taking the first 50-mg dose of treatment. However, the woman had a history of essential hypertension and, at autopsy, was found to have coronary artery disease and pulmonary embolism.
A large phase 3 program is now underway to confirm safety and efficacy of SEP-363856 for schizophrenia, Dr. Koblan said. It includes four studies that are open and enrolling.
Benefit beyond psychosis?
In an accompanying editorial , Donald C. Goff, MD, of the Nathan Kline Institute at New York University, wrote that if the findings were replicated in future trials with more patients and of longer duration, “this class of drugs may provide a valuable new therapeutic option for psychosis and the negative symptoms of schizophrenia, possibly without the adverse effects associated with direct D2-receptor antagonism.”
It is also possible that SEP-363856 will have effects “beyond D2-receptor antagonists and hence beyond the treatment of psychosis. Other potential therapeutic indications, including cognitive impairment, depression, and substance abuse, remain to be explored,” Dr. Goff wrote.
This study “not only introduces a promising therapeutic compound but also provides additional evidence, on the basis of an agnostic drug-discovery process, that TAAR1 is a promising target,” he added.
“This is very welcome news given the great need for new pharmacologic treatments for patients with schizophrenia,” Dr. Goff concluded.
The study was funded by Sunovion Pharmaceuticals. Dr. Koblan is an employee of the company. Disclosures for the other authors are listed in the original article. Dr. Goff reported having received grants and nonfinancial support from Avanir Pharmaceuticals and from Takeda.
A version of this article first appeared on Medscape.com.
The ABCDs of treating tardive dyskinesia
Tardive dyskinesia (TD)—involuntary movement persisting for >1 month—is often caused by exposure to dopamine receptor–blocking agents such as antipsychotics.1 The pathophysiology of TD is attributed to dopamine receptor hypersensitivity and upregulation of dopamine receptors in response to chronic receptor blockade, although striatal dysfunction, oxidative stress, and gamma-aminobutyric acid (GABA) dysfunction may play a role.1 Because discontinuing the antipsychotic may not improve the patient’s TD symptoms and may worsen mood or psychosis, clinicians often prescribe adjunctive agents to reduce TD symptoms while continuing the antipsychotic. Clinicians can use the mnemonic ABCD to help recall 4 evidence-based treatments for TD.
Amantadine is an N-methyl-
Ginkgo Biloba contains antioxidant properties that may help reduce TD symptoms by alleviating oxidative stress. In a meta-analysis of 3 randomized controlled trials from China (N = 299), ginkgo biloba extract, 240 mg/d, significantly improved symptoms of TD compared with placebo.3 Ginkgo biloba has an antiplatelet effect and therefore should not be used in patients with an increased bleeding risk.
Clonazepam. Several small studies have examined the use of this GABA agonist for TD. In a study of 19 patients with TD, researchers found a symptom reduction of up to 35% with doses up to 4.5 mg/d.4 However, many studies have had small sample sizes or poor methodology. A 2018 Cochrane review recommended using other agents before considering clonazepam for TD because this medication has uncertain efficacy in treating TD, and it can cause sedation and dependence.5
Deutetrabenazine and valbenazine, the only FDA-approved treatments for TD, are vesicular monoamine transporter 2 (VMAT2) inhibitors, which inhibit dopamine release and decrease dopamine receptor hypersensitivity.6 In a 12-week, randomized, double-blind, placebo-controlled study of 117 patients with moderate-to-severe TD, those who received deutetrabenazine (up to 48 mg/d) had a significant mean reduction in AIMS score (3 points) compared with placebo.7 In the 1-year KINECT 3 study, 124 patients with TD who received valbenazine, 40 or 80 mg/d, had significant mean reductions in AIMS scores of 3.0 and 4.8 points, respectively.8 Adverse effects of these medications include somnolence, headache, akathisia, urinary tract infection, worsening mood, and suicidality. Tetrabenazine is another VMAT2 inhibitor that may be effective in doses up to 150 mg/d, but its off-label use is limited by the need for frequent dosing and a risk for suicidality.6
Other adjunctive treatments, such as vitamin B6, vitamin E, zonisamide, and levetiracetam, might offer some benefit in TD.6 However, further evidence is needed to support including these interventions in treatment guidelines.
1. Elkurd MT, Bahroo L. Keeping up with the clinical advances: tardive dyskinesia. CNS Spectr. 2019;24(suppl 1):70-81.
2. Pappa S, Tsouli S, Apostolu G, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271-275.
3. Zheng W, Xiang YQ, Ng CH, et al. Extract of ginkgo biloba for tardive dyskinesia: meta-analysis of randomized controlled trials. Pharmacopsychiatry. 2016;49(3):107-111.
4. Thaker GK, Nguyen JA, Strauss ME, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445-451.
5. Bergman H, Bhoopathi PS, Soares-Weiser K. Benzodiazepines for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:CD000205.
6. Sreeram V, Shagufta S, Kagadkar F. Role of vesicular monoamine transporter 2 inhibitors in tardive dyskinesia management. Cureus. 2019;11(8):e5471. doi: 10.7759/cureus.5471.
7. Fernandez HH, Factor SA, Hauser RA. Randomized controlled trial of deutetrabenazine for tardive dyskinesia. The ARM-TD study. Neurology. 2017;88(21):2003-2010.
8. Factor SA, Remington G, Comella CL, et al. The effects of valbenazine in participants with tardive dyskinesia: results of the 1-year KINECT 3 extension study. J Clin Psychiatry. 2017;78(9):1344-1350.
Tardive dyskinesia (TD)—involuntary movement persisting for >1 month—is often caused by exposure to dopamine receptor–blocking agents such as antipsychotics.1 The pathophysiology of TD is attributed to dopamine receptor hypersensitivity and upregulation of dopamine receptors in response to chronic receptor blockade, although striatal dysfunction, oxidative stress, and gamma-aminobutyric acid (GABA) dysfunction may play a role.1 Because discontinuing the antipsychotic may not improve the patient’s TD symptoms and may worsen mood or psychosis, clinicians often prescribe adjunctive agents to reduce TD symptoms while continuing the antipsychotic. Clinicians can use the mnemonic ABCD to help recall 4 evidence-based treatments for TD.
Amantadine is an N-methyl-
Ginkgo Biloba contains antioxidant properties that may help reduce TD symptoms by alleviating oxidative stress. In a meta-analysis of 3 randomized controlled trials from China (N = 299), ginkgo biloba extract, 240 mg/d, significantly improved symptoms of TD compared with placebo.3 Ginkgo biloba has an antiplatelet effect and therefore should not be used in patients with an increased bleeding risk.
Clonazepam. Several small studies have examined the use of this GABA agonist for TD. In a study of 19 patients with TD, researchers found a symptom reduction of up to 35% with doses up to 4.5 mg/d.4 However, many studies have had small sample sizes or poor methodology. A 2018 Cochrane review recommended using other agents before considering clonazepam for TD because this medication has uncertain efficacy in treating TD, and it can cause sedation and dependence.5
Deutetrabenazine and valbenazine, the only FDA-approved treatments for TD, are vesicular monoamine transporter 2 (VMAT2) inhibitors, which inhibit dopamine release and decrease dopamine receptor hypersensitivity.6 In a 12-week, randomized, double-blind, placebo-controlled study of 117 patients with moderate-to-severe TD, those who received deutetrabenazine (up to 48 mg/d) had a significant mean reduction in AIMS score (3 points) compared with placebo.7 In the 1-year KINECT 3 study, 124 patients with TD who received valbenazine, 40 or 80 mg/d, had significant mean reductions in AIMS scores of 3.0 and 4.8 points, respectively.8 Adverse effects of these medications include somnolence, headache, akathisia, urinary tract infection, worsening mood, and suicidality. Tetrabenazine is another VMAT2 inhibitor that may be effective in doses up to 150 mg/d, but its off-label use is limited by the need for frequent dosing and a risk for suicidality.6
Other adjunctive treatments, such as vitamin B6, vitamin E, zonisamide, and levetiracetam, might offer some benefit in TD.6 However, further evidence is needed to support including these interventions in treatment guidelines.
Tardive dyskinesia (TD)—involuntary movement persisting for >1 month—is often caused by exposure to dopamine receptor–blocking agents such as antipsychotics.1 The pathophysiology of TD is attributed to dopamine receptor hypersensitivity and upregulation of dopamine receptors in response to chronic receptor blockade, although striatal dysfunction, oxidative stress, and gamma-aminobutyric acid (GABA) dysfunction may play a role.1 Because discontinuing the antipsychotic may not improve the patient’s TD symptoms and may worsen mood or psychosis, clinicians often prescribe adjunctive agents to reduce TD symptoms while continuing the antipsychotic. Clinicians can use the mnemonic ABCD to help recall 4 evidence-based treatments for TD.
Amantadine is an N-methyl-
Ginkgo Biloba contains antioxidant properties that may help reduce TD symptoms by alleviating oxidative stress. In a meta-analysis of 3 randomized controlled trials from China (N = 299), ginkgo biloba extract, 240 mg/d, significantly improved symptoms of TD compared with placebo.3 Ginkgo biloba has an antiplatelet effect and therefore should not be used in patients with an increased bleeding risk.
Clonazepam. Several small studies have examined the use of this GABA agonist for TD. In a study of 19 patients with TD, researchers found a symptom reduction of up to 35% with doses up to 4.5 mg/d.4 However, many studies have had small sample sizes or poor methodology. A 2018 Cochrane review recommended using other agents before considering clonazepam for TD because this medication has uncertain efficacy in treating TD, and it can cause sedation and dependence.5
Deutetrabenazine and valbenazine, the only FDA-approved treatments for TD, are vesicular monoamine transporter 2 (VMAT2) inhibitors, which inhibit dopamine release and decrease dopamine receptor hypersensitivity.6 In a 12-week, randomized, double-blind, placebo-controlled study of 117 patients with moderate-to-severe TD, those who received deutetrabenazine (up to 48 mg/d) had a significant mean reduction in AIMS score (3 points) compared with placebo.7 In the 1-year KINECT 3 study, 124 patients with TD who received valbenazine, 40 or 80 mg/d, had significant mean reductions in AIMS scores of 3.0 and 4.8 points, respectively.8 Adverse effects of these medications include somnolence, headache, akathisia, urinary tract infection, worsening mood, and suicidality. Tetrabenazine is another VMAT2 inhibitor that may be effective in doses up to 150 mg/d, but its off-label use is limited by the need for frequent dosing and a risk for suicidality.6
Other adjunctive treatments, such as vitamin B6, vitamin E, zonisamide, and levetiracetam, might offer some benefit in TD.6 However, further evidence is needed to support including these interventions in treatment guidelines.
1. Elkurd MT, Bahroo L. Keeping up with the clinical advances: tardive dyskinesia. CNS Spectr. 2019;24(suppl 1):70-81.
2. Pappa S, Tsouli S, Apostolu G, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271-275.
3. Zheng W, Xiang YQ, Ng CH, et al. Extract of ginkgo biloba for tardive dyskinesia: meta-analysis of randomized controlled trials. Pharmacopsychiatry. 2016;49(3):107-111.
4. Thaker GK, Nguyen JA, Strauss ME, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445-451.
5. Bergman H, Bhoopathi PS, Soares-Weiser K. Benzodiazepines for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:CD000205.
6. Sreeram V, Shagufta S, Kagadkar F. Role of vesicular monoamine transporter 2 inhibitors in tardive dyskinesia management. Cureus. 2019;11(8):e5471. doi: 10.7759/cureus.5471.
7. Fernandez HH, Factor SA, Hauser RA. Randomized controlled trial of deutetrabenazine for tardive dyskinesia. The ARM-TD study. Neurology. 2017;88(21):2003-2010.
8. Factor SA, Remington G, Comella CL, et al. The effects of valbenazine in participants with tardive dyskinesia: results of the 1-year KINECT 3 extension study. J Clin Psychiatry. 2017;78(9):1344-1350.
1. Elkurd MT, Bahroo L. Keeping up with the clinical advances: tardive dyskinesia. CNS Spectr. 2019;24(suppl 1):70-81.
2. Pappa S, Tsouli S, Apostolu G, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271-275.
3. Zheng W, Xiang YQ, Ng CH, et al. Extract of ginkgo biloba for tardive dyskinesia: meta-analysis of randomized controlled trials. Pharmacopsychiatry. 2016;49(3):107-111.
4. Thaker GK, Nguyen JA, Strauss ME, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445-451.
5. Bergman H, Bhoopathi PS, Soares-Weiser K. Benzodiazepines for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:CD000205.
6. Sreeram V, Shagufta S, Kagadkar F. Role of vesicular monoamine transporter 2 inhibitors in tardive dyskinesia management. Cureus. 2019;11(8):e5471. doi: 10.7759/cureus.5471.
7. Fernandez HH, Factor SA, Hauser RA. Randomized controlled trial of deutetrabenazine for tardive dyskinesia. The ARM-TD study. Neurology. 2017;88(21):2003-2010.
8. Factor SA, Remington G, Comella CL, et al. The effects of valbenazine in participants with tardive dyskinesia: results of the 1-year KINECT 3 extension study. J Clin Psychiatry. 2017;78(9):1344-1350.
Is psychosis toxic to the brain?
Schizophrenia has been described as the “worst disease” to afflict mankind.1 It causes psychosis, which is an abnormal state of mind marked by hyperarousal, overactivation of brain circuits, and emotional distress. An untreated episode of psychosis can result in structural brain damage due to neurotoxicity. Patients who experience psychosis may be affected by inflammatory processes, oxidative and nitrosative reactions, mitochondrial dysfunction, decreased synaptic plasticity and neurogenesis, demyelination, and autoimmune attacks—all of which can contribute to cell necrosis and irreversible neuronal atrophy.2-4
The impacts of untreated psychosis
First-episode psychosis (FEP) can result in a loss of up to 1% of total brain volume and up to 3% of cortical gray matter.4,5 When FEP goes untreated, approximately 10 to 12 cc of brain tissue—basically a tablespoon of cells and myelin—could be permanently damaged.2,6,7 This explains why enlarged ventricles are a common radiologic finding in patients with schizophrenia.2 In such patients, imaging of the brain will show these as hollow, fluid-filled spaces that appear expanded.
Repeated episodes of untreated psychosis could result in progressively lower levels of baseline functioning, and patients may require longer hospitalizations to achieve stabilization and higher doses of medications to achieve remission.4,7 Greater brain volume losses are associated with poorer outcomes.3 Brain volume loss is also detectable in patients with untreated major depressive episodes,8 and recurrent episodes of bipolar I disorder can also result in the loss of gray matter and structural brain damage.9 The progressive decline in cognitive and functional outcomes and eventual development of treatment resistance are likely due to a kindling phenomenon or receptor sensitization.10
Act fast to prevent brain damage
Since it was first identified, schizophrenia has been recognized as a degenerative disease. However, the progression to structural brain damage is not inevitable, and can be arrested with expeditious, decisive treatment. Some agents, such as certain antipsychotic medications10 and omega-3 fatty acids, can be neuroprotective.11 The early use of a long-acting injectable antipsychotic also may help prevent relapse and additional psychotic episodes.5
Psychosis requires expedient and competent intervention to improve outcomes and reduce disease burden. Timely psychiatric treatment can improve not only immediate functioning, but also long-term prognosis. Because untreated psychosis can result in irreversible structural brain damage, clinicians must act swiftly to provide assertive treatment.
1. Where next with psychiatric illness? Nature. 1998;336(6195):95-96.
2. Salisbury DF, Kuroki N, Kasai K, et al. Progressive and interrelated functional and structural evidence of post-onset brain reduction in schizophrenia. Arch Gen Psychiatry. 2007;64(5):521-529.
3. van Haren NE, Hulshoff HE, Shnack HG, et al. Progressive brain volume loss in schizophrenia over the course of the illness: evidence of maturational abnormalities in early adulthood. Biol Psychiatry. 2008;63(1):106-113.
4. Cahn W, Hulshoff Pol HE, Lems EB, et al. Brain volume changes in first-episode schizophrenia: a 1-year follow-up study. Arch Gen Psychiatry. 2002;59(11):1002-1010.
5. Subotnik KL, Casaus LR, Ventura J, et al. Long-acting injectable risperidone for relapse prevention and control of breakthrough symptoms after a recent first episode of schizophrenia. a randomized clinical trial. JAMA Psychiatry. 2015;72(8):822-829.
6. Nasrallah HA. FAST and RAPID: acronyms to prevent brain damage in stroke and psychosis. Current Psychiatry. 2018;17(8):6-8.
7. Nasrallah HA. For first-episode psychosis, psychiatrists should behave like cardiologists. Current Psychiatry. 2017;16(8):4-7.
8. Moylan S, Maes M, Wray NR, et al. The neuroprogressive nature of major depressive disorder: pathways to disease evolution and resistance, and therapeutic implications. Mol Psychiatry. 2013;18(5):595-606.
9. Kozicky JM, McGirr A, Bond DJ, et al. Neuroprogression and episode recurrence in bipolar I disorder: a study of gray matter volume changes in first-episode mania and association with clinical outcome. Bipolar Disord. 2016;18(6):511-519.
10. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
11. Amminger GP, Schäfer MR, Schlögelhofer M, et al. Longer-term outcome in the prevention of psychotic disorders by the Vienna omega-3 study. Nat Commun. 2015;6:7934. doi: 10.1038/ncomms8934.
Schizophrenia has been described as the “worst disease” to afflict mankind.1 It causes psychosis, which is an abnormal state of mind marked by hyperarousal, overactivation of brain circuits, and emotional distress. An untreated episode of psychosis can result in structural brain damage due to neurotoxicity. Patients who experience psychosis may be affected by inflammatory processes, oxidative and nitrosative reactions, mitochondrial dysfunction, decreased synaptic plasticity and neurogenesis, demyelination, and autoimmune attacks—all of which can contribute to cell necrosis and irreversible neuronal atrophy.2-4
The impacts of untreated psychosis
First-episode psychosis (FEP) can result in a loss of up to 1% of total brain volume and up to 3% of cortical gray matter.4,5 When FEP goes untreated, approximately 10 to 12 cc of brain tissue—basically a tablespoon of cells and myelin—could be permanently damaged.2,6,7 This explains why enlarged ventricles are a common radiologic finding in patients with schizophrenia.2 In such patients, imaging of the brain will show these as hollow, fluid-filled spaces that appear expanded.
Repeated episodes of untreated psychosis could result in progressively lower levels of baseline functioning, and patients may require longer hospitalizations to achieve stabilization and higher doses of medications to achieve remission.4,7 Greater brain volume losses are associated with poorer outcomes.3 Brain volume loss is also detectable in patients with untreated major depressive episodes,8 and recurrent episodes of bipolar I disorder can also result in the loss of gray matter and structural brain damage.9 The progressive decline in cognitive and functional outcomes and eventual development of treatment resistance are likely due to a kindling phenomenon or receptor sensitization.10
Act fast to prevent brain damage
Since it was first identified, schizophrenia has been recognized as a degenerative disease. However, the progression to structural brain damage is not inevitable, and can be arrested with expeditious, decisive treatment. Some agents, such as certain antipsychotic medications10 and omega-3 fatty acids, can be neuroprotective.11 The early use of a long-acting injectable antipsychotic also may help prevent relapse and additional psychotic episodes.5
Psychosis requires expedient and competent intervention to improve outcomes and reduce disease burden. Timely psychiatric treatment can improve not only immediate functioning, but also long-term prognosis. Because untreated psychosis can result in irreversible structural brain damage, clinicians must act swiftly to provide assertive treatment.
Schizophrenia has been described as the “worst disease” to afflict mankind.1 It causes psychosis, which is an abnormal state of mind marked by hyperarousal, overactivation of brain circuits, and emotional distress. An untreated episode of psychosis can result in structural brain damage due to neurotoxicity. Patients who experience psychosis may be affected by inflammatory processes, oxidative and nitrosative reactions, mitochondrial dysfunction, decreased synaptic plasticity and neurogenesis, demyelination, and autoimmune attacks—all of which can contribute to cell necrosis and irreversible neuronal atrophy.2-4
The impacts of untreated psychosis
First-episode psychosis (FEP) can result in a loss of up to 1% of total brain volume and up to 3% of cortical gray matter.4,5 When FEP goes untreated, approximately 10 to 12 cc of brain tissue—basically a tablespoon of cells and myelin—could be permanently damaged.2,6,7 This explains why enlarged ventricles are a common radiologic finding in patients with schizophrenia.2 In such patients, imaging of the brain will show these as hollow, fluid-filled spaces that appear expanded.
Repeated episodes of untreated psychosis could result in progressively lower levels of baseline functioning, and patients may require longer hospitalizations to achieve stabilization and higher doses of medications to achieve remission.4,7 Greater brain volume losses are associated with poorer outcomes.3 Brain volume loss is also detectable in patients with untreated major depressive episodes,8 and recurrent episodes of bipolar I disorder can also result in the loss of gray matter and structural brain damage.9 The progressive decline in cognitive and functional outcomes and eventual development of treatment resistance are likely due to a kindling phenomenon or receptor sensitization.10
Act fast to prevent brain damage
Since it was first identified, schizophrenia has been recognized as a degenerative disease. However, the progression to structural brain damage is not inevitable, and can be arrested with expeditious, decisive treatment. Some agents, such as certain antipsychotic medications10 and omega-3 fatty acids, can be neuroprotective.11 The early use of a long-acting injectable antipsychotic also may help prevent relapse and additional psychotic episodes.5
Psychosis requires expedient and competent intervention to improve outcomes and reduce disease burden. Timely psychiatric treatment can improve not only immediate functioning, but also long-term prognosis. Because untreated psychosis can result in irreversible structural brain damage, clinicians must act swiftly to provide assertive treatment.
1. Where next with psychiatric illness? Nature. 1998;336(6195):95-96.
2. Salisbury DF, Kuroki N, Kasai K, et al. Progressive and interrelated functional and structural evidence of post-onset brain reduction in schizophrenia. Arch Gen Psychiatry. 2007;64(5):521-529.
3. van Haren NE, Hulshoff HE, Shnack HG, et al. Progressive brain volume loss in schizophrenia over the course of the illness: evidence of maturational abnormalities in early adulthood. Biol Psychiatry. 2008;63(1):106-113.
4. Cahn W, Hulshoff Pol HE, Lems EB, et al. Brain volume changes in first-episode schizophrenia: a 1-year follow-up study. Arch Gen Psychiatry. 2002;59(11):1002-1010.
5. Subotnik KL, Casaus LR, Ventura J, et al. Long-acting injectable risperidone for relapse prevention and control of breakthrough symptoms after a recent first episode of schizophrenia. a randomized clinical trial. JAMA Psychiatry. 2015;72(8):822-829.
6. Nasrallah HA. FAST and RAPID: acronyms to prevent brain damage in stroke and psychosis. Current Psychiatry. 2018;17(8):6-8.
7. Nasrallah HA. For first-episode psychosis, psychiatrists should behave like cardiologists. Current Psychiatry. 2017;16(8):4-7.
8. Moylan S, Maes M, Wray NR, et al. The neuroprogressive nature of major depressive disorder: pathways to disease evolution and resistance, and therapeutic implications. Mol Psychiatry. 2013;18(5):595-606.
9. Kozicky JM, McGirr A, Bond DJ, et al. Neuroprogression and episode recurrence in bipolar I disorder: a study of gray matter volume changes in first-episode mania and association with clinical outcome. Bipolar Disord. 2016;18(6):511-519.
10. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
11. Amminger GP, Schäfer MR, Schlögelhofer M, et al. Longer-term outcome in the prevention of psychotic disorders by the Vienna omega-3 study. Nat Commun. 2015;6:7934. doi: 10.1038/ncomms8934.
1. Where next with psychiatric illness? Nature. 1998;336(6195):95-96.
2. Salisbury DF, Kuroki N, Kasai K, et al. Progressive and interrelated functional and structural evidence of post-onset brain reduction in schizophrenia. Arch Gen Psychiatry. 2007;64(5):521-529.
3. van Haren NE, Hulshoff HE, Shnack HG, et al. Progressive brain volume loss in schizophrenia over the course of the illness: evidence of maturational abnormalities in early adulthood. Biol Psychiatry. 2008;63(1):106-113.
4. Cahn W, Hulshoff Pol HE, Lems EB, et al. Brain volume changes in first-episode schizophrenia: a 1-year follow-up study. Arch Gen Psychiatry. 2002;59(11):1002-1010.
5. Subotnik KL, Casaus LR, Ventura J, et al. Long-acting injectable risperidone for relapse prevention and control of breakthrough symptoms after a recent first episode of schizophrenia. a randomized clinical trial. JAMA Psychiatry. 2015;72(8):822-829.
6. Nasrallah HA. FAST and RAPID: acronyms to prevent brain damage in stroke and psychosis. Current Psychiatry. 2018;17(8):6-8.
7. Nasrallah HA. For first-episode psychosis, psychiatrists should behave like cardiologists. Current Psychiatry. 2017;16(8):4-7.
8. Moylan S, Maes M, Wray NR, et al. The neuroprogressive nature of major depressive disorder: pathways to disease evolution and resistance, and therapeutic implications. Mol Psychiatry. 2013;18(5):595-606.
9. Kozicky JM, McGirr A, Bond DJ, et al. Neuroprogression and episode recurrence in bipolar I disorder: a study of gray matter volume changes in first-episode mania and association with clinical outcome. Bipolar Disord. 2016;18(6):511-519.
10. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
11. Amminger GP, Schäfer MR, Schlögelhofer M, et al. Longer-term outcome in the prevention of psychotic disorders by the Vienna omega-3 study. Nat Commun. 2015;6:7934. doi: 10.1038/ncomms8934.
Command hallucinations, but is it really psychosis?
CASE Frequent hospitalizations
Ms. D, age 26, presents to the emergency department (ED) after drinking a bottle of hand sanitizer in a suicide attempt. She is admitted to an inpatient psychiatric unit, where she spends 50 days, followed by a transfer to a step-down unit, where she spends 26 days. Upon discharge, her diagnosis is schizoaffective disorder–bipolar type.
Shortly before this, Ms. D had intentionally ingested 20 vitamin pills to “make her heart stop” after a conflict at home. After ingesting the pills, Ms. D presented to the ED, where she stated that if she were discharged, she would kill herself by taking “better pills.” She was then admitted to an inpatient psychiatric unit, where she spent 60 days before being moved to an extended-care step-down facility, where she resided for 42 days.
HISTORY A challenging past
Ms. D has a history of >25 psychiatric hospitalizations with varying discharge diagnoses, including schizophrenia, schizoaffective disorder, borderline personality disorder (BPD), and borderline intellectual functioning.
Ms. D was raised in a 2-parent home with 3 older half-brothers and 3 sisters. She was sexually assaulted by a cousin when she was 12. Ms. D recalls one event of self-injury/cutting behavior at age 15 after she was bullied by peers. Her family history is significant for schizophrenia (mother), alcohol use disorder (both parents), and bipolar disorder (sister). Her mother, who is now deceased, was admitted to state psychiatric hospitals for extended periods.
Her medication regimen has changed with nearly every hospitalization but generally has included ≥1 antipsychotic, a mood stabilizer, an antidepressant, and a benzodiazepine (often prescribed on an as-needed basis). Ms. D is obese and has difficulty sleeping, hypothyroidism, gastroesophageal reflux disease (GERD), hypertension, and iron deficiency anemia. She receives medications to manage each of these conditions.
Ms. D’s previous psychotic symptoms included auditory command hallucinations. These occurred under stressful circumstances, such as during severe family conflicts that often led to her feeling abandoned. She reported that the “voice” she heard was usually her own instructing her to “take pills.” There was no prior evidence of bizarre delusions, negative symptoms, or disorganized thoughts or speech.
During episodes of decompensation, Ms. D did not report symptoms of mania, sustained depressed mood, or anxiety, nor were these symptoms observed. Although Ms. D endorsed suicidal ideation with a plan, intent, and means, during several of her previous ED presentations, she told clinicians that her intent was not to end her life but rather to evoke concern in her family members.
Continue to: After her mother died...
After her mother died when Ms. D was 19, she began to have nightmares of wanting to hurt herself and others and began experiencing multiple hospitalizations. In 2010, Ms. D was referred to an assertive community treatment (ACT) program for individuals age 16 to 27 because of her inability to participate in traditional community-based services and her historical need for advanced services, in order to provide psychiatric care in the least restrictive means possible.
Despite receiving intensive ACT services, and in addition to the numerous inpatient psychiatric hospitalizations, over 7 years, Ms. D accumulated 8 additional general-medical hospitalizations and >50 visits to hospital EDs and urgent care facilities. These hospitalizations typically followed arguments at home, strained family dynamics, and not feeling wanted. Ms. D would ingest large quantities of prescription or over-the-counter medications as a way of coping, which often occurred while she was residing in a step-down facility after hospital discharge.
[polldaddy:10528342]
The authors’ observations
The treatment team decided to transition Ms. D to an LTSR with full continuum of treatment. While some clinicians might be concerned with potential iatrogenic harm of LTSR placement and might instead recommend less restrictive residential support and an IOP. However, in Ms. D’s case, her numerous admissions to EDs, urgent care facilities, and medical and psychiatric hospitals, her failed step-down facility placements, and her family conflicts and poor dynamics limited the efficacy of her natural support system and drove the recommendation for an LTSR.
Previously, Ms. D’s experience with ACT services had centered on managing acute crises, with brief periods of stabilization that insufficiently engaged her in a consistent and meaningful treatment plan. Ms. D’s insurance company agreed to pay for the LTSR after lengthy discussions with the clinical leadership at the ACT program and the LTSR demonstrated that she was a high utilizer of health care services. They concluded that Ms. D’s stay at the LTSR would be less expensive than the frequent use of expensive hospital services and care.
EVALUATION A consensus on the diagnosis
During the first few weeks of Ms. D’s admission to the LTSR, the treatment team takes a thorough history and reviews her medical records, which they obtained from several past inpatient admissions and therapists who previously treated Ms. D. The team also collects collateral information from Ms. D’s family members. Based on this information, interviews, and composite behavioral observations from the first few weeks of Ms. D’s time at the LTSR, the psychiatrists and treatment team at the LTSR and ACT program determine that Ms. D meets the criteria for a primary diagnosis of BPD. Previous discharge diagnoses of schizoaffective disorder–bipolar type (Table 11), schizophrenia, or bipolar disorder could not be affirmed.

Continue to: The authors' observations
The authors’ observations
During Ms. D’s LTSR placement, it became clear that her self-harm behaviors and numerous visits to the ED and urgent care facilities involved severe and intense emotional dysregulation and maladaptive behaviors. These behaviors had developed over time in response to acute stressors and past trauma, and not as a result of a sustained mood or psychotic disorder. Before her LTSR placement, Ms. D was unable to use more adaptive coping skills, such as skills building, learning, and coaching. Ms. D typically “thrived” with medical attention in the ED or hospital, and once the stressor dissipated, she was discharged back to the same stressful living environment associated with her maladaptive coping.
Table 2 outlines the rationale for long-term residential treatment for Ms. D.

TREATMENT Developing more effective skills
Bolstered by a clearer diagnostic formulation of BPD, Ms. D’s initial treatment goals at the LTSR include developing effective skills (eg, mindfulness, interpersonal effectiveness, emotion regulation, and distress tolerance) to cope with family conflicts and other stressors while she is outside the facility on a therapeutic pass. Ms. D’s treatment focuses on skills learning and coaching, and behavior chain analyses, which are conducted by her therapist from the ACT program.
Ms. D remains clinically stable throughout her LTSR placement, and benefits from ongoing skills building and learning, coaching, and community integration efforts.
[polldaddy:10528348]
The authors’ observations
Several systematic reviews2-5 have found that there is a lack of high-quality evidence for the use of various psychotropic medications for patients with BPD, yet polypharmacy is common. Many patients with BPD receive ≥2 medications and >25% of patients receive ≥4 medications, typically for prolonged periods. Stoffers et al4 suggested that FGAs and antidepressants have marginal effects of for patients with BPD; however, their use cannot be ruled out because they may be helpful for comorbid symptoms that are often observed in patients with BPD. There is better evidence for SGAs, mood stabilizers, and omega-3 fatty acids; however, most effect estimates were based on single studies, and there is minimal data on long-term use of these agents.4
Continue to: A recent review highlighted...
A recent review highlighted 2 trends in medication prescribing for individuals with BPD3:
- a decrease in the use of benzodiazepines and antidepressants
- an increase in or preference for mood stabilizers and SGAs, especially valproate and quetiapine.
In terms of which medications can be used to target specific symptoms, the same researchers also noted from previous studies3:
- The prior use of SSRIs to target affective dysregulation, anxiety, and impulsive- behavior dyscontrol
- mood stabilizers (notably anticonvulsants) and SGAs to target “core symptoms” of BPD, including affective dysregulation, impulsive-behavioral dyscontrol, and cognitive-perceptual distortions
- omega-3 fatty acids for mood stabilization, impulsive-behavior dyscontrol, and possibly to reduce self-harm behaviors.
TREATMENT Medication adjustments
The treatment team reviews the lack of evidence for the long-term use of psychotropic medications in the treatment of BPD with Ms. D and her relatives,2-5 and develops a medication regimen that is clinically appropriate for managing the symptoms of BPD, while also being mindful of adverse effects.
When Ms. D was admitted to the LTSR from the hospital, her psychotropic medication regimen included haloperidol, 150 mg IM every month; olanzapine, 20 mg at bedtime; benztropine, 1 mg twice daily; and melatonin, 9 mg at bedtime.
Following discussions with Ms. D and her older sister, the team initiates a taper of olanzapine because of metabolic concerns. Ms. D has gained >40 lb while receiving this medication and had hypertension. Olanzapine was tapered and discontinued over the course of 3 months with no reemergence of sustained mood or psychotic symptoms (Table 3). During this period, Ms. D also participates in dietary counselling, follows a portion-controlled regimen, and loses >30 lb. Her wellness plan focuses on nutrition and exercise to improve her overall physical health.

Continue to: Six months into her stay...
Six months into her stay at the LTSR, Ms. D remains clinically stable and is able to leave the LTSR placement to go on home passes. At this time, the team begins to taper the haloperidol long-acting injection. One month prior to discharge from the LTSR, haloperidol is discontinued entirely. The treatment team simultaneously tapers and discontinues benztropine. No recurrence of extrapyramidal symptoms is observed by staff or noted by the patient.
A treatment plan is developed to address Ms. D’s medical conditions, including hypothyroidism, GERD, and obesity. Ms. D does not appear to have difficulty sleeping at the LTSR, so melatonin is tapered by 3-mg decrements and stopped after 2 months. However, shortly thereafter, she develops insomnia, so a 3-mg dose is re-initiated, and her complaints abate. Her primary care physician discontinues hydrochlorothiazide, an antihypertensive medication.
Ms. D’s medication regimen consists of melatonin, 3 mg at bedtime; pantoprazole, 40 mg before breakfast, for GERD; senna, 8.6 mg at bedtime, and polyethylene glycol, 17 gm/d, for constipation; levothyroxine, 125 mcg/d, for hypothyroidism; metoprolol extended-release, 50 mg/d, for hypertension; and ferrous sulfate, 325 mg/d, for iron deficiency anemia.
OUTCOME Improved functioning
After 11 months at the LTSR, Ms. D is discharged home. She continues to receive outpatient services in the community through the ACT program, meeting with her therapist for cognitive-behavioral therapy, skills building and learning, and integration.
Approximately 9 months later, Ms. D is re-started on an SSRI (sertraline, 50 mg/d, which is increased to 100 mg/d 9 months later) to target symptoms of anxiety, which primarily manifest as excessive worrying. Hydroxyzine, 50 mg 3 times daily as needed, is added to this regimen, for breakthrough anxiety symptoms. Hydroxyzine is prescribed instead of a benzodiazepine to avoid potential addiction and abuse.
Continue to: Oral ziprasidone...
Oral ziprasidone, 20 mg/d twice daily, is initiated during 2 brief inpatient psychiatric admissions; however, it is successfully tapered within 1 week of discharge, in partnership with the ACT program.
In the 23 months after her discharge, Ms. D has had 1 ED visit and 2 brief inpatient psychiatric hospitalizations, which is markedly fewer encounters than she had in the 2 years before her LTSR placement. She has also lost an additional 30 lb since her LTSR discharge through a healthy diet and exercise.
Ms. D is now considering transitioning to living independently in the community through a residential supported housing program.
Bottom Line
Psychotic symptoms in patients with borderline personality disorder (BPD) are typically fleeting and mostly occur in the context of intense interpersonal conflicts and real or imagined abandonment. Long-term structured residence placement for patients with BPD can allow for careful formulation of a treatment plan, and help patients gain effective skills to cope with difficult family dynamics and other stressors, with the ultimate goal of gradual community integration.
Related Resource
- National Education Alliance for Borderline Personality Disorder. https://www.borderlinepersonalitydisorder.org.
Drug Brand Names
Benztropine • Cogentin
Haloperidol • Haldol
Hydrochlorothiazide • Microzide, HydroDiuril
Hydroxyzine • Vistaril
Levothyroxine • Synthroid,
Metoprolol ER • Toprol XL
Olanzapine • Zyprexa
Pantoprazole • Protonix
Polyethylene glycol • MiraLax, Glycolax
Quetiapine • Seroquel
Senna • Senokot
Sertraline • Zoloft
Valproate • Depakene, Depakote
Ziprasidone • Geodon
1. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Hancock-Johnson E, Griffiths C, Picchioni M. A focused systematic review of pharmacological treatment for borderline personality disorder. CNS Drugs. 2017;31:345-356.
3. Starcevic V, Janca A. Pharmacotherapy of borderline personality disorder: replacing confusion with prudent pragmatism. Curr Opin Psychiatry. 2018;31(1):69-73.
4. Stoffers J, Völlm BA, Rücker G, et al. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2010;6:CD005653. doi: 10.1002/14651858.CD005653.pub2.
5. Stoffers-Winterling JM, Storebo OJ, Völlm BA, et al. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2018;3:CD012956. doi: 10.1002/14651858.CD012956.
CASE Frequent hospitalizations
Ms. D, age 26, presents to the emergency department (ED) after drinking a bottle of hand sanitizer in a suicide attempt. She is admitted to an inpatient psychiatric unit, where she spends 50 days, followed by a transfer to a step-down unit, where she spends 26 days. Upon discharge, her diagnosis is schizoaffective disorder–bipolar type.
Shortly before this, Ms. D had intentionally ingested 20 vitamin pills to “make her heart stop” after a conflict at home. After ingesting the pills, Ms. D presented to the ED, where she stated that if she were discharged, she would kill herself by taking “better pills.” She was then admitted to an inpatient psychiatric unit, where she spent 60 days before being moved to an extended-care step-down facility, where she resided for 42 days.
HISTORY A challenging past
Ms. D has a history of >25 psychiatric hospitalizations with varying discharge diagnoses, including schizophrenia, schizoaffective disorder, borderline personality disorder (BPD), and borderline intellectual functioning.
Ms. D was raised in a 2-parent home with 3 older half-brothers and 3 sisters. She was sexually assaulted by a cousin when she was 12. Ms. D recalls one event of self-injury/cutting behavior at age 15 after she was bullied by peers. Her family history is significant for schizophrenia (mother), alcohol use disorder (both parents), and bipolar disorder (sister). Her mother, who is now deceased, was admitted to state psychiatric hospitals for extended periods.
Her medication regimen has changed with nearly every hospitalization but generally has included ≥1 antipsychotic, a mood stabilizer, an antidepressant, and a benzodiazepine (often prescribed on an as-needed basis). Ms. D is obese and has difficulty sleeping, hypothyroidism, gastroesophageal reflux disease (GERD), hypertension, and iron deficiency anemia. She receives medications to manage each of these conditions.
Ms. D’s previous psychotic symptoms included auditory command hallucinations. These occurred under stressful circumstances, such as during severe family conflicts that often led to her feeling abandoned. She reported that the “voice” she heard was usually her own instructing her to “take pills.” There was no prior evidence of bizarre delusions, negative symptoms, or disorganized thoughts or speech.
During episodes of decompensation, Ms. D did not report symptoms of mania, sustained depressed mood, or anxiety, nor were these symptoms observed. Although Ms. D endorsed suicidal ideation with a plan, intent, and means, during several of her previous ED presentations, she told clinicians that her intent was not to end her life but rather to evoke concern in her family members.
Continue to: After her mother died...
After her mother died when Ms. D was 19, she began to have nightmares of wanting to hurt herself and others and began experiencing multiple hospitalizations. In 2010, Ms. D was referred to an assertive community treatment (ACT) program for individuals age 16 to 27 because of her inability to participate in traditional community-based services and her historical need for advanced services, in order to provide psychiatric care in the least restrictive means possible.
Despite receiving intensive ACT services, and in addition to the numerous inpatient psychiatric hospitalizations, over 7 years, Ms. D accumulated 8 additional general-medical hospitalizations and >50 visits to hospital EDs and urgent care facilities. These hospitalizations typically followed arguments at home, strained family dynamics, and not feeling wanted. Ms. D would ingest large quantities of prescription or over-the-counter medications as a way of coping, which often occurred while she was residing in a step-down facility after hospital discharge.
[polldaddy:10528342]
The authors’ observations
The treatment team decided to transition Ms. D to an LTSR with full continuum of treatment. While some clinicians might be concerned with potential iatrogenic harm of LTSR placement and might instead recommend less restrictive residential support and an IOP. However, in Ms. D’s case, her numerous admissions to EDs, urgent care facilities, and medical and psychiatric hospitals, her failed step-down facility placements, and her family conflicts and poor dynamics limited the efficacy of her natural support system and drove the recommendation for an LTSR.
Previously, Ms. D’s experience with ACT services had centered on managing acute crises, with brief periods of stabilization that insufficiently engaged her in a consistent and meaningful treatment plan. Ms. D’s insurance company agreed to pay for the LTSR after lengthy discussions with the clinical leadership at the ACT program and the LTSR demonstrated that she was a high utilizer of health care services. They concluded that Ms. D’s stay at the LTSR would be less expensive than the frequent use of expensive hospital services and care.
EVALUATION A consensus on the diagnosis
During the first few weeks of Ms. D’s admission to the LTSR, the treatment team takes a thorough history and reviews her medical records, which they obtained from several past inpatient admissions and therapists who previously treated Ms. D. The team also collects collateral information from Ms. D’s family members. Based on this information, interviews, and composite behavioral observations from the first few weeks of Ms. D’s time at the LTSR, the psychiatrists and treatment team at the LTSR and ACT program determine that Ms. D meets the criteria for a primary diagnosis of BPD. Previous discharge diagnoses of schizoaffective disorder–bipolar type (Table 11), schizophrenia, or bipolar disorder could not be affirmed.

Continue to: The authors' observations
The authors’ observations
During Ms. D’s LTSR placement, it became clear that her self-harm behaviors and numerous visits to the ED and urgent care facilities involved severe and intense emotional dysregulation and maladaptive behaviors. These behaviors had developed over time in response to acute stressors and past trauma, and not as a result of a sustained mood or psychotic disorder. Before her LTSR placement, Ms. D was unable to use more adaptive coping skills, such as skills building, learning, and coaching. Ms. D typically “thrived” with medical attention in the ED or hospital, and once the stressor dissipated, she was discharged back to the same stressful living environment associated with her maladaptive coping.
Table 2 outlines the rationale for long-term residential treatment for Ms. D.

TREATMENT Developing more effective skills
Bolstered by a clearer diagnostic formulation of BPD, Ms. D’s initial treatment goals at the LTSR include developing effective skills (eg, mindfulness, interpersonal effectiveness, emotion regulation, and distress tolerance) to cope with family conflicts and other stressors while she is outside the facility on a therapeutic pass. Ms. D’s treatment focuses on skills learning and coaching, and behavior chain analyses, which are conducted by her therapist from the ACT program.
Ms. D remains clinically stable throughout her LTSR placement, and benefits from ongoing skills building and learning, coaching, and community integration efforts.
[polldaddy:10528348]
The authors’ observations
Several systematic reviews2-5 have found that there is a lack of high-quality evidence for the use of various psychotropic medications for patients with BPD, yet polypharmacy is common. Many patients with BPD receive ≥2 medications and >25% of patients receive ≥4 medications, typically for prolonged periods. Stoffers et al4 suggested that FGAs and antidepressants have marginal effects of for patients with BPD; however, their use cannot be ruled out because they may be helpful for comorbid symptoms that are often observed in patients with BPD. There is better evidence for SGAs, mood stabilizers, and omega-3 fatty acids; however, most effect estimates were based on single studies, and there is minimal data on long-term use of these agents.4
Continue to: A recent review highlighted...
A recent review highlighted 2 trends in medication prescribing for individuals with BPD3:
- a decrease in the use of benzodiazepines and antidepressants
- an increase in or preference for mood stabilizers and SGAs, especially valproate and quetiapine.
In terms of which medications can be used to target specific symptoms, the same researchers also noted from previous studies3:
- The prior use of SSRIs to target affective dysregulation, anxiety, and impulsive- behavior dyscontrol
- mood stabilizers (notably anticonvulsants) and SGAs to target “core symptoms” of BPD, including affective dysregulation, impulsive-behavioral dyscontrol, and cognitive-perceptual distortions
- omega-3 fatty acids for mood stabilization, impulsive-behavior dyscontrol, and possibly to reduce self-harm behaviors.
TREATMENT Medication adjustments
The treatment team reviews the lack of evidence for the long-term use of psychotropic medications in the treatment of BPD with Ms. D and her relatives,2-5 and develops a medication regimen that is clinically appropriate for managing the symptoms of BPD, while also being mindful of adverse effects.
When Ms. D was admitted to the LTSR from the hospital, her psychotropic medication regimen included haloperidol, 150 mg IM every month; olanzapine, 20 mg at bedtime; benztropine, 1 mg twice daily; and melatonin, 9 mg at bedtime.
Following discussions with Ms. D and her older sister, the team initiates a taper of olanzapine because of metabolic concerns. Ms. D has gained >40 lb while receiving this medication and had hypertension. Olanzapine was tapered and discontinued over the course of 3 months with no reemergence of sustained mood or psychotic symptoms (Table 3). During this period, Ms. D also participates in dietary counselling, follows a portion-controlled regimen, and loses >30 lb. Her wellness plan focuses on nutrition and exercise to improve her overall physical health.

Continue to: Six months into her stay...
Six months into her stay at the LTSR, Ms. D remains clinically stable and is able to leave the LTSR placement to go on home passes. At this time, the team begins to taper the haloperidol long-acting injection. One month prior to discharge from the LTSR, haloperidol is discontinued entirely. The treatment team simultaneously tapers and discontinues benztropine. No recurrence of extrapyramidal symptoms is observed by staff or noted by the patient.
A treatment plan is developed to address Ms. D’s medical conditions, including hypothyroidism, GERD, and obesity. Ms. D does not appear to have difficulty sleeping at the LTSR, so melatonin is tapered by 3-mg decrements and stopped after 2 months. However, shortly thereafter, she develops insomnia, so a 3-mg dose is re-initiated, and her complaints abate. Her primary care physician discontinues hydrochlorothiazide, an antihypertensive medication.
Ms. D’s medication regimen consists of melatonin, 3 mg at bedtime; pantoprazole, 40 mg before breakfast, for GERD; senna, 8.6 mg at bedtime, and polyethylene glycol, 17 gm/d, for constipation; levothyroxine, 125 mcg/d, for hypothyroidism; metoprolol extended-release, 50 mg/d, for hypertension; and ferrous sulfate, 325 mg/d, for iron deficiency anemia.
OUTCOME Improved functioning
After 11 months at the LTSR, Ms. D is discharged home. She continues to receive outpatient services in the community through the ACT program, meeting with her therapist for cognitive-behavioral therapy, skills building and learning, and integration.
Approximately 9 months later, Ms. D is re-started on an SSRI (sertraline, 50 mg/d, which is increased to 100 mg/d 9 months later) to target symptoms of anxiety, which primarily manifest as excessive worrying. Hydroxyzine, 50 mg 3 times daily as needed, is added to this regimen, for breakthrough anxiety symptoms. Hydroxyzine is prescribed instead of a benzodiazepine to avoid potential addiction and abuse.
Continue to: Oral ziprasidone...
Oral ziprasidone, 20 mg/d twice daily, is initiated during 2 brief inpatient psychiatric admissions; however, it is successfully tapered within 1 week of discharge, in partnership with the ACT program.
In the 23 months after her discharge, Ms. D has had 1 ED visit and 2 brief inpatient psychiatric hospitalizations, which is markedly fewer encounters than she had in the 2 years before her LTSR placement. She has also lost an additional 30 lb since her LTSR discharge through a healthy diet and exercise.
Ms. D is now considering transitioning to living independently in the community through a residential supported housing program.
Bottom Line
Psychotic symptoms in patients with borderline personality disorder (BPD) are typically fleeting and mostly occur in the context of intense interpersonal conflicts and real or imagined abandonment. Long-term structured residence placement for patients with BPD can allow for careful formulation of a treatment plan, and help patients gain effective skills to cope with difficult family dynamics and other stressors, with the ultimate goal of gradual community integration.
Related Resource
- National Education Alliance for Borderline Personality Disorder. https://www.borderlinepersonalitydisorder.org.
Drug Brand Names
Benztropine • Cogentin
Haloperidol • Haldol
Hydrochlorothiazide • Microzide, HydroDiuril
Hydroxyzine • Vistaril
Levothyroxine • Synthroid,
Metoprolol ER • Toprol XL
Olanzapine • Zyprexa
Pantoprazole • Protonix
Polyethylene glycol • MiraLax, Glycolax
Quetiapine • Seroquel
Senna • Senokot
Sertraline • Zoloft
Valproate • Depakene, Depakote
Ziprasidone • Geodon
CASE Frequent hospitalizations
Ms. D, age 26, presents to the emergency department (ED) after drinking a bottle of hand sanitizer in a suicide attempt. She is admitted to an inpatient psychiatric unit, where she spends 50 days, followed by a transfer to a step-down unit, where she spends 26 days. Upon discharge, her diagnosis is schizoaffective disorder–bipolar type.
Shortly before this, Ms. D had intentionally ingested 20 vitamin pills to “make her heart stop” after a conflict at home. After ingesting the pills, Ms. D presented to the ED, where she stated that if she were discharged, she would kill herself by taking “better pills.” She was then admitted to an inpatient psychiatric unit, where she spent 60 days before being moved to an extended-care step-down facility, where she resided for 42 days.
HISTORY A challenging past
Ms. D has a history of >25 psychiatric hospitalizations with varying discharge diagnoses, including schizophrenia, schizoaffective disorder, borderline personality disorder (BPD), and borderline intellectual functioning.
Ms. D was raised in a 2-parent home with 3 older half-brothers and 3 sisters. She was sexually assaulted by a cousin when she was 12. Ms. D recalls one event of self-injury/cutting behavior at age 15 after she was bullied by peers. Her family history is significant for schizophrenia (mother), alcohol use disorder (both parents), and bipolar disorder (sister). Her mother, who is now deceased, was admitted to state psychiatric hospitals for extended periods.
Her medication regimen has changed with nearly every hospitalization but generally has included ≥1 antipsychotic, a mood stabilizer, an antidepressant, and a benzodiazepine (often prescribed on an as-needed basis). Ms. D is obese and has difficulty sleeping, hypothyroidism, gastroesophageal reflux disease (GERD), hypertension, and iron deficiency anemia. She receives medications to manage each of these conditions.
Ms. D’s previous psychotic symptoms included auditory command hallucinations. These occurred under stressful circumstances, such as during severe family conflicts that often led to her feeling abandoned. She reported that the “voice” she heard was usually her own instructing her to “take pills.” There was no prior evidence of bizarre delusions, negative symptoms, or disorganized thoughts or speech.
During episodes of decompensation, Ms. D did not report symptoms of mania, sustained depressed mood, or anxiety, nor were these symptoms observed. Although Ms. D endorsed suicidal ideation with a plan, intent, and means, during several of her previous ED presentations, she told clinicians that her intent was not to end her life but rather to evoke concern in her family members.
Continue to: After her mother died...
After her mother died when Ms. D was 19, she began to have nightmares of wanting to hurt herself and others and began experiencing multiple hospitalizations. In 2010, Ms. D was referred to an assertive community treatment (ACT) program for individuals age 16 to 27 because of her inability to participate in traditional community-based services and her historical need for advanced services, in order to provide psychiatric care in the least restrictive means possible.
Despite receiving intensive ACT services, and in addition to the numerous inpatient psychiatric hospitalizations, over 7 years, Ms. D accumulated 8 additional general-medical hospitalizations and >50 visits to hospital EDs and urgent care facilities. These hospitalizations typically followed arguments at home, strained family dynamics, and not feeling wanted. Ms. D would ingest large quantities of prescription or over-the-counter medications as a way of coping, which often occurred while she was residing in a step-down facility after hospital discharge.
[polldaddy:10528342]
The authors’ observations
The treatment team decided to transition Ms. D to an LTSR with full continuum of treatment. While some clinicians might be concerned with potential iatrogenic harm of LTSR placement and might instead recommend less restrictive residential support and an IOP. However, in Ms. D’s case, her numerous admissions to EDs, urgent care facilities, and medical and psychiatric hospitals, her failed step-down facility placements, and her family conflicts and poor dynamics limited the efficacy of her natural support system and drove the recommendation for an LTSR.
Previously, Ms. D’s experience with ACT services had centered on managing acute crises, with brief periods of stabilization that insufficiently engaged her in a consistent and meaningful treatment plan. Ms. D’s insurance company agreed to pay for the LTSR after lengthy discussions with the clinical leadership at the ACT program and the LTSR demonstrated that she was a high utilizer of health care services. They concluded that Ms. D’s stay at the LTSR would be less expensive than the frequent use of expensive hospital services and care.
EVALUATION A consensus on the diagnosis
During the first few weeks of Ms. D’s admission to the LTSR, the treatment team takes a thorough history and reviews her medical records, which they obtained from several past inpatient admissions and therapists who previously treated Ms. D. The team also collects collateral information from Ms. D’s family members. Based on this information, interviews, and composite behavioral observations from the first few weeks of Ms. D’s time at the LTSR, the psychiatrists and treatment team at the LTSR and ACT program determine that Ms. D meets the criteria for a primary diagnosis of BPD. Previous discharge diagnoses of schizoaffective disorder–bipolar type (Table 11), schizophrenia, or bipolar disorder could not be affirmed.

Continue to: The authors' observations
The authors’ observations
During Ms. D’s LTSR placement, it became clear that her self-harm behaviors and numerous visits to the ED and urgent care facilities involved severe and intense emotional dysregulation and maladaptive behaviors. These behaviors had developed over time in response to acute stressors and past trauma, and not as a result of a sustained mood or psychotic disorder. Before her LTSR placement, Ms. D was unable to use more adaptive coping skills, such as skills building, learning, and coaching. Ms. D typically “thrived” with medical attention in the ED or hospital, and once the stressor dissipated, she was discharged back to the same stressful living environment associated with her maladaptive coping.
Table 2 outlines the rationale for long-term residential treatment for Ms. D.

TREATMENT Developing more effective skills
Bolstered by a clearer diagnostic formulation of BPD, Ms. D’s initial treatment goals at the LTSR include developing effective skills (eg, mindfulness, interpersonal effectiveness, emotion regulation, and distress tolerance) to cope with family conflicts and other stressors while she is outside the facility on a therapeutic pass. Ms. D’s treatment focuses on skills learning and coaching, and behavior chain analyses, which are conducted by her therapist from the ACT program.
Ms. D remains clinically stable throughout her LTSR placement, and benefits from ongoing skills building and learning, coaching, and community integration efforts.
[polldaddy:10528348]
The authors’ observations
Several systematic reviews2-5 have found that there is a lack of high-quality evidence for the use of various psychotropic medications for patients with BPD, yet polypharmacy is common. Many patients with BPD receive ≥2 medications and >25% of patients receive ≥4 medications, typically for prolonged periods. Stoffers et al4 suggested that FGAs and antidepressants have marginal effects of for patients with BPD; however, their use cannot be ruled out because they may be helpful for comorbid symptoms that are often observed in patients with BPD. There is better evidence for SGAs, mood stabilizers, and omega-3 fatty acids; however, most effect estimates were based on single studies, and there is minimal data on long-term use of these agents.4
Continue to: A recent review highlighted...
A recent review highlighted 2 trends in medication prescribing for individuals with BPD3:
- a decrease in the use of benzodiazepines and antidepressants
- an increase in or preference for mood stabilizers and SGAs, especially valproate and quetiapine.
In terms of which medications can be used to target specific symptoms, the same researchers also noted from previous studies3:
- The prior use of SSRIs to target affective dysregulation, anxiety, and impulsive- behavior dyscontrol
- mood stabilizers (notably anticonvulsants) and SGAs to target “core symptoms” of BPD, including affective dysregulation, impulsive-behavioral dyscontrol, and cognitive-perceptual distortions
- omega-3 fatty acids for mood stabilization, impulsive-behavior dyscontrol, and possibly to reduce self-harm behaviors.
TREATMENT Medication adjustments
The treatment team reviews the lack of evidence for the long-term use of psychotropic medications in the treatment of BPD with Ms. D and her relatives,2-5 and develops a medication regimen that is clinically appropriate for managing the symptoms of BPD, while also being mindful of adverse effects.
When Ms. D was admitted to the LTSR from the hospital, her psychotropic medication regimen included haloperidol, 150 mg IM every month; olanzapine, 20 mg at bedtime; benztropine, 1 mg twice daily; and melatonin, 9 mg at bedtime.
Following discussions with Ms. D and her older sister, the team initiates a taper of olanzapine because of metabolic concerns. Ms. D has gained >40 lb while receiving this medication and had hypertension. Olanzapine was tapered and discontinued over the course of 3 months with no reemergence of sustained mood or psychotic symptoms (Table 3). During this period, Ms. D also participates in dietary counselling, follows a portion-controlled regimen, and loses >30 lb. Her wellness plan focuses on nutrition and exercise to improve her overall physical health.

Continue to: Six months into her stay...
Six months into her stay at the LTSR, Ms. D remains clinically stable and is able to leave the LTSR placement to go on home passes. At this time, the team begins to taper the haloperidol long-acting injection. One month prior to discharge from the LTSR, haloperidol is discontinued entirely. The treatment team simultaneously tapers and discontinues benztropine. No recurrence of extrapyramidal symptoms is observed by staff or noted by the patient.
A treatment plan is developed to address Ms. D’s medical conditions, including hypothyroidism, GERD, and obesity. Ms. D does not appear to have difficulty sleeping at the LTSR, so melatonin is tapered by 3-mg decrements and stopped after 2 months. However, shortly thereafter, she develops insomnia, so a 3-mg dose is re-initiated, and her complaints abate. Her primary care physician discontinues hydrochlorothiazide, an antihypertensive medication.
Ms. D’s medication regimen consists of melatonin, 3 mg at bedtime; pantoprazole, 40 mg before breakfast, for GERD; senna, 8.6 mg at bedtime, and polyethylene glycol, 17 gm/d, for constipation; levothyroxine, 125 mcg/d, for hypothyroidism; metoprolol extended-release, 50 mg/d, for hypertension; and ferrous sulfate, 325 mg/d, for iron deficiency anemia.
OUTCOME Improved functioning
After 11 months at the LTSR, Ms. D is discharged home. She continues to receive outpatient services in the community through the ACT program, meeting with her therapist for cognitive-behavioral therapy, skills building and learning, and integration.
Approximately 9 months later, Ms. D is re-started on an SSRI (sertraline, 50 mg/d, which is increased to 100 mg/d 9 months later) to target symptoms of anxiety, which primarily manifest as excessive worrying. Hydroxyzine, 50 mg 3 times daily as needed, is added to this regimen, for breakthrough anxiety symptoms. Hydroxyzine is prescribed instead of a benzodiazepine to avoid potential addiction and abuse.
Continue to: Oral ziprasidone...
Oral ziprasidone, 20 mg/d twice daily, is initiated during 2 brief inpatient psychiatric admissions; however, it is successfully tapered within 1 week of discharge, in partnership with the ACT program.
In the 23 months after her discharge, Ms. D has had 1 ED visit and 2 brief inpatient psychiatric hospitalizations, which is markedly fewer encounters than she had in the 2 years before her LTSR placement. She has also lost an additional 30 lb since her LTSR discharge through a healthy diet and exercise.
Ms. D is now considering transitioning to living independently in the community through a residential supported housing program.
Bottom Line
Psychotic symptoms in patients with borderline personality disorder (BPD) are typically fleeting and mostly occur in the context of intense interpersonal conflicts and real or imagined abandonment. Long-term structured residence placement for patients with BPD can allow for careful formulation of a treatment plan, and help patients gain effective skills to cope with difficult family dynamics and other stressors, with the ultimate goal of gradual community integration.
Related Resource
- National Education Alliance for Borderline Personality Disorder. https://www.borderlinepersonalitydisorder.org.
Drug Brand Names
Benztropine • Cogentin
Haloperidol • Haldol
Hydrochlorothiazide • Microzide, HydroDiuril
Hydroxyzine • Vistaril
Levothyroxine • Synthroid,
Metoprolol ER • Toprol XL
Olanzapine • Zyprexa
Pantoprazole • Protonix
Polyethylene glycol • MiraLax, Glycolax
Quetiapine • Seroquel
Senna • Senokot
Sertraline • Zoloft
Valproate • Depakene, Depakote
Ziprasidone • Geodon
1. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Hancock-Johnson E, Griffiths C, Picchioni M. A focused systematic review of pharmacological treatment for borderline personality disorder. CNS Drugs. 2017;31:345-356.
3. Starcevic V, Janca A. Pharmacotherapy of borderline personality disorder: replacing confusion with prudent pragmatism. Curr Opin Psychiatry. 2018;31(1):69-73.
4. Stoffers J, Völlm BA, Rücker G, et al. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2010;6:CD005653. doi: 10.1002/14651858.CD005653.pub2.
5. Stoffers-Winterling JM, Storebo OJ, Völlm BA, et al. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2018;3:CD012956. doi: 10.1002/14651858.CD012956.
1. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Hancock-Johnson E, Griffiths C, Picchioni M. A focused systematic review of pharmacological treatment for borderline personality disorder. CNS Drugs. 2017;31:345-356.
3. Starcevic V, Janca A. Pharmacotherapy of borderline personality disorder: replacing confusion with prudent pragmatism. Curr Opin Psychiatry. 2018;31(1):69-73.
4. Stoffers J, Völlm BA, Rücker G, et al. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2010;6:CD005653. doi: 10.1002/14651858.CD005653.pub2.
5. Stoffers-Winterling JM, Storebo OJ, Völlm BA, et al. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2018;3:CD012956. doi: 10.1002/14651858.CD012956.
The psychiatric clinic of the future
Despite the tremendous advances in psychiatry in recent years, the current clinical practice of psychiatry continues to rely on data from intermittent assessments along with subjective and unquantifiable accounts from patients and caregivers. Furthermore, there continues to be significant diagnostic variations among practitioners. Fortunately, technology to address these issues appears to be on the horizon.
How might the psychiatric clinic of the future look? What changes could we envision? These 4 critical factors may soon bring about dynamic changes in the way we practice psychiatry:
- precision psychiatry
- digital psychiatry
- technology-enhanced psychotherapy
- electronic health record (EHR) reforms.
In this article, we review how advances in each of these areas might lead to improved care for our patients.
Precision psychiatry
Precision psychiatry takes into account each patient’s variability in genes, environment, and lifestyle to determine individualized treatment and prevention strategies. It relies on pharmacogenomic testing as the primary tool. Pharmacogenomics is the study of variability in drug response due to heredity.
Emerging data on the clinical utility and cost-effectiveness of pharmacogenomic testing are encouraging, but its routine use is not well supported by current evidence.2 One limit to using pharmacogenomic testing is that many genes simultaneously exert an effect on the structure and function of neurons and associated pathophysiology. According to the International Society of Psychiatric Genetics, no single genetic variant is sufficient to cause psychiatric disorders such as depression, bipolar disorder, substance dependence, or schizophrenia. This limits the possibility of using genetic tests to establish a diagnosis.3
In the future, better algorithms could promote more accurate pharmacogenomics profiles for individual patients, which could influence treatment.
Precision psychiatry could lead to:
- identification of novel targets for new medications
- pharmacogenetic profiling of the patient to predict disease susceptibility and medication response
- personalized therapy: the right drug at the right dose for the right patient.
- improved efficacy and fewer adverse medication reactions.
Continue to: Digital psychiatry
Digital psychiatry
Integrating computer-based technology into psychiatric practice has given birth to a new frontier that could be called digital psychiatry. This might encompass the following:
- telepsychiatry
- social media with a mental health focus
- web-based applications/devices
- artificial intelligence (AI).
Telepsychiatry. Videoconferencing is the most widely used form of telepsychiatry. It provides patients with easier access to mental health treatment.4 Telepsychiatry has the potential to match patients and clinicians with similar cultural backgrounds, thus minimizing cultural gaps and misunderstandings. Most importantly, it is comparable to face-to-face interviews in terms of the reliability of assessment and treatment outcomes.5
Telepsychiatry might be particularly helpful for patients with restricted mobility, such as those who live in remote areas, nursing homes, or correctional facilities. In correctional settings, transferring prisoners is expensive and carries the risk of escape. In a small study (N = 86) conducted in Hong Kong, Chen et al6 found that using videoconferencing to conduct clinical interviews of inmates was cost-efficient and scored high in terms of patient acceptability.
Social media. Social media could be a powerful platform for early detection of mental illness. Staying connected with patients on social media could allow psychiatrists to be more aware of their patient’s mood fluctuations, which might lead to more timely assessments. Physicians could be automatically notified about changes in their patients’ social media activity that indicate changes in mental state, which could solicit immediate intervention and treatment. On the other hand, such use of social media could blur professional boundaries. Psychiatrists also could use social media to promote awareness of mental health and educate the public on ways to improve or maintain their mental well-being.7
Web-based applications/devices. Real-time monitoring through applications or internet-based smart devices creates a new avenue for patients to receive personalized assessments, treatment, and intervention.8 Smartwatches with internet connectivity may offer a glimpse of the wearer’s sleep architecture and duration, thus providing real-time data on patients who have insomnia. We can now passively collect objective data from devices, such as smartphones and laptops, to phenotype an individual’s mood and mental state, a process called digital phenotyping. The Table9 lists examples of the types of mental health–related metrics that can be captured by smartphones, smartwatches, and similar technology. Information from these devices can be accumulated to create a database that can be used to predict symptoms.10 For example, the way people use a smartphone’s keyboard, including latency time between space and character types, can be used to generate variables for data. This type of information is being studied for use in screening depression and passively assessing mood in real time.11
Continue to: Artificial intelligence
Artificial intelligence—the development of computer systems able to perform tasks that normally require human intelligence—is being increasingly used in psychiatry. Some studies have suggested AI can be used to identify patients’ risk of suicide12-15 or psychosis.16,17 Kalanderian and Nasrallah18 reviewed several of these studies in
Other researchers have found clinical uses for machine learning, a subset of AI that uses methods to automatically detect patterns and make predictions based on those patterns. In one study, a machine learning analysis of functional MRI scans was able to identify 4 distinct subtypes of depression.19 In another study, a machine learning model was able to predict with 60% accuracy which patients with depression would respond to antidepressants.20
In the future, AI might be used to change mental health classification systems. Because many mental health disorders share similar symptom clusters, machine learning can help to identify associations between symptoms, behavior, brain function, and real-world function across different diagnoses, potentially affecting how we will classify mental disorders.21
Technology-enhanced psychotherapy
In the future, it might be common for psychotherapy to be provided by a computer, or “virtual therapist.” Several studies have evaluated the use of technology-enhanced psychotherapy.
Lucas et al22 investigated patients’ interactions with a virtual therapist. Participants were interviewed by an avatar named Ellie, who they saw on a TV screen. Half of the participants were told Ellie was not human, and half were told Ellie was being controlled remotely by a human. Three psychologists who were blinded to group allocations analyzed transcripts of the interviews and video recordings of participants’ facial expressions to quantify the participants’ fear, sadness, and other emotional responses during the interviews, as well as their openness to the questions. Participants who believed Ellie was fully automated reported significantly lower fear of self-disclosure and impression management (attempts to control how others perceive them) than participants who were told that Ellie was operated by a human. Additionally, participants who believed they were interacting with a computer were more open during the interview.22
Continue to: Researchers at the University of Southern California...
Researchers at the University of Southern California developed software that assessed 74 acoustic features, including pitch, volume, quality, shimmer, jitter, and prosody, to predict outcomes among patients receiving couples therapy. This software was able to predict marital discord at least as well as human therapists.23
Many mental health apps purport to implement specific components of psychotherapy. Many of these apps focus on cognitive-behavioral therapy worksheets, mindfulness exercises, and/or mood tracking. The features provided by such apps emulate the tasks and intended outcomes of traditional psychotherapy, but in an entirely decentralized venue.24
Some have expressed concern that an increased use of virtual therapists powered by AI might lead to a dehumanization of psychiatry (Box25,26).
Box
Whether there are aspects of the psychiatric patient encounter that cannot be managed by a “virtual clinician” created by artificial intelligence (AI) remains to be determined. Some of the benefits of using AI in this manner may be difficult to anticipate, or may be specific to an individual’s relationship with his/her clinician.25
On the other hand, AI systems blur previously assumed boundaries between reality and fiction, and this could have complex effects on patients. Similar to therapeutic relationships with a human clinician, there is the risk of transference of emotions, thoughts, and feelings to a virtual therapist powered by AI. Unlike with a psychiatrist or therapist, however, there is no person on the other side of this transference. Whether virtual clinicians will be able to manage such transference remains to be seen.
In Deep Medicine,26 cardiologist Eric Topol, MD, emphasizes a crucial component of a patient encounter that AI will be unlikely able to provide: empathy. Virtual therapists powered by AI will inherit the tasks best done by machines, leaving humans more time to do what they do best—providing empathy and being “present” for patients.
Electronic health record reforms
Although many clinicians find EHRs to be onerous and time-consuming, EHR technology is constantly improving, and EHRs have revolutionized documentation and order implementation. Several potential advances could improve clinical practice. For example, EHRs could incorporate a clinical decision support system that uses AI-based algorithms to assist psychiatrists with diagnosis, monitoring, and treatment.27 In the future, EHRs might have the ability to monitor and learn from errors and adverse events, and automatically design an algorithm to avoid them.28 They should be designed to better manage analysis of pharmacogenetic test results, which is challenging due to the amount and complexity of the data.29 Future EHRs should eliminate the non-intuitive and multi-click interfaces and cumbersome data searches of today’s EHRs.30
Technology brings new ethical considerations
Mental health interventions based on AI typically work with algorithms, and algorithms bring ethical issues. Mental health devices or systems that use AI could contain biases that have the potential to harm in unintended ways, such as a data-driven sexist or racist bias.31 This may require investing additional time to explain to patients (and their families) what an algorithm is and how it works in relation to the therapy provided.
Continue to: Another concern is patient...
Another concern is patient autonomy.32 For example, it would be ethically problematic if a patient were to assume that there was a human physician “at the other end” of a virtual therapist or other technology who is communicating or reviewing his/her messages. Similarly, an older adult or a patient with intellectual disabilities may not be able to understand advanced technology or what it does when it is installed in their home to monitor the patient’s activities. This would increase the risk of privacy violations, manipulation, or even coercion if the requirements for informed consent are not satisfied.
A flowchart for the future
Although current research and innovations typically target specific areas of psychiatry, these advances can be integrated by devising algorithms and protocols that will change the current practice of psychiatry. The Figure provides a glimpse of how the psychiatry clinic of the future might work. A maxim of management is that “the best way to predict the future is to create it.” However, the mere conception of a vision is not enough—working towards it is essential.
Bottom Line
With advances in technology, psychiatric practice will soon be radically different from what it is today. The expanded use of telepsychiatry, social media, artificial intelligence, and web-based applications/devices holds great promise for psychiatric assessment, diagnosis, and treatment, although certain ethical and privacy concerns need to be adequately addressed.
Related Resources
- National Institute of Mental Health. Technology and the future of mental health treatment. www.nimh.nih.gov/health/topics/technology-and-the-future-of-mental-health-treatment/index.shtml. Revised September 2019.
- Hays R, Farrell HM, Touros J. Mobile apps and mental health: using technology to quantify real-time clinical risk. Current Psychiatry. 2019;18(6):37-41.
- Torous J, Luo J, Chan SR. Mental health apps: what to tell patients. Current Psychiatry. 2018;17(3):21-25.
1. Pirmohamed M. Pharmacogenetics and pharmacogenomics. Br J Clin Pharmacol. 2001;52(4):345-347.
2. Benitez J, Cool CL, Scotti DJ. Use of combinatorial pharmacogenomic guidance in treating psychiatric disorders. Per Med. 2018;15(6):481-494.
3. Cannon TD. Candidate gene studies in the GWAS era: the MET proto-oncogene, neurocognition, and schizophrenia. Am J Psychiatry. 2010;167(4):4,369-372.
4. Greenwood J, Chamberlain C, Parker G. Evaluation of a rural telepsychiatry service. Australas Psychiatry. 2004;12(3):268-272.
5. Hubley S, Lynch SB, Schneck C, et al. Review of key telepsychiatry outcomes. World J Psychiatry. 2016;6(2):269-282.
6. Cheng KM, Siu BW, Yeung CC, et al. Telepsychiatry for stable Chinese psychiatric out-patients in custody in Hong Kong: a case-control pilot study. Hong Kong Med J. 2018;24(4):378-383.
7. Frankish K, Ryan C, Harris A. Psychiatry and online social media: potential, pitfalls and ethical guidelines for psychiatrists and trainees. Australasian Psychiatry. 2012;20(3):181-187.
8. de la Torre Díez I, Alonso SG, Hamrioui S, et al. IoT-based services and applications for mental health in the literature. J Med Syst. 2019;43(1):4-9.
9. Topol E. Deep Medicine. New York, NY: Basic Books; 2019:168.
10. Adams RA, Huys QJM, Roiser JP. Computational Psychiatry: towards a mathematically informed understanding of mental illness. J Neurol Neurosurg Psychiatry. 2016;87(1):53-63.
11. Insel TR. Bending the curve for mental health: technology for a public health approach. Am J Public Health. 2019;109(suppl 3):S168-S170.
12. Just MA, Pan L, Cherkassky VL, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. 2017;1:911-919.
13. Pestian J, Nasrallah H, Matykiewicz P, et al. Suicide note classification using natural language processing: a content analysis. Biomed Inform Insights. 2010;2010(3):19-28.
14. Walsh CG, Ribeiro JD, Franklin JC. Predicting risk of suicide attempts over time through machine learning. Clinical Psychological Science. 2017;5(3):457-469.
15. Pestian JP, Sorter M, Connolly B, et al; STM Research Group. A machine learning approach to identifying the thought markers of suicidal subjects: a prospective multicenter trial. Suicide Life Threat Behav. 2017;47(1):112-121.
16. Corcoran CM, Carrillo F, Fernández-Slezak D, et al. Prediction of psychosis across protocols and risk cohorts using automated language analysis. World Psychiatry. 2018;17(1):67-75.
17. Bedi G, Carrillo F, Cecchi GA, et al. Automated analysis of free speech predicts psychosis onset in high-risk youths. NPJ Schizophr. 2015;1:15030. doi: 10.1038/npjschz.2015.30.
18. Kalanderian H, Nasrallah HA. Artificial intelligence in psychiatry. Current Psychiatry. 2019;18(8):33-38.
19. Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. 2017;23(1):28-38.
20. Chekroud AM, Zotti RJ, Shehzad Z, et al. Cross-trial prediction of treatment outcome in depression: a machine learning approach. Lancet Psychiatry. 2016;3(3):243-250.
21. Grisanzio KA, Goldstein-Piekarski AN, Wang MY, et al. Transdiagnostic symptom clusters and associations with brain, behavior, and daily function in mood, anxiety, and trauma disorders. JAMA Psychiatry. 2018;75(2):201-209.
22. Lucas G, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Computers in Human Behavior. 2014;37:94-100.
23. Nasir M, Baucom BR, Georgiou P, et al. Predicting couple therapy outcomes based on speech acoustic features. PLoS One. 2017;12(9):e0185123. doi: 10.1371/journal.pone.0185123.
24. Huguet A, Rao S, McGrath PJ, et al. A systematic review of cognitive behavioral therapy and behavioral activation apps for depression. PLoS One. 2016;11(5):e0154248. doi: 10.1371/journal.pone.0154248.
25. Scholten MR, Kelders SM, Van Gemert-Pijnen JE. Self-guided web-based interventions: scoping review on user needs and the potential of embodied conversational agents to address them. J Med Internet Res. 2017;19(11):e383.
26. Topol E. Deep Medicine. New York, NY: Basic Books; 2019:283-310.
27. Abramson EL, McGinnis S, Edwards A, et al. Electronic health record adoption and health information exchange among hospitals in New York State. J Eval Clin Pract. 2012;18(6):1156-1162.
28. Meeks DW, Smith MW, Taylor L, et al. An analysis of electronic health record-related patient safety concerns. J Am Med Inform Assoc. 2014;21(6):1053-1059.
29. Kho AN, Rasmussen LV, Connolly JJ, et al. Practical challenges in integrating genomic data into the electronic health record. Genet Med. 2013;15(10):772-778.
30. Ornstein SM, Oates RB, Fox GN. The computer-based medical record: current status. J Fam Pract. 1992;35(5):556-565.
31. Corea F. Machine ethics and artificial moral agents. In: Applied artificial intelligence: where AI can be used in business. Basel, Switzerland: Springer; 2019:33-41.
32. Beauchamp T, Childress J. Principles of biomedical ethics. 7th ed. New York, NY: Oxford University Press; 2012:44.
Despite the tremendous advances in psychiatry in recent years, the current clinical practice of psychiatry continues to rely on data from intermittent assessments along with subjective and unquantifiable accounts from patients and caregivers. Furthermore, there continues to be significant diagnostic variations among practitioners. Fortunately, technology to address these issues appears to be on the horizon.
How might the psychiatric clinic of the future look? What changes could we envision? These 4 critical factors may soon bring about dynamic changes in the way we practice psychiatry:
- precision psychiatry
- digital psychiatry
- technology-enhanced psychotherapy
- electronic health record (EHR) reforms.
In this article, we review how advances in each of these areas might lead to improved care for our patients.
Precision psychiatry
Precision psychiatry takes into account each patient’s variability in genes, environment, and lifestyle to determine individualized treatment and prevention strategies. It relies on pharmacogenomic testing as the primary tool. Pharmacogenomics is the study of variability in drug response due to heredity.
Emerging data on the clinical utility and cost-effectiveness of pharmacogenomic testing are encouraging, but its routine use is not well supported by current evidence.2 One limit to using pharmacogenomic testing is that many genes simultaneously exert an effect on the structure and function of neurons and associated pathophysiology. According to the International Society of Psychiatric Genetics, no single genetic variant is sufficient to cause psychiatric disorders such as depression, bipolar disorder, substance dependence, or schizophrenia. This limits the possibility of using genetic tests to establish a diagnosis.3
In the future, better algorithms could promote more accurate pharmacogenomics profiles for individual patients, which could influence treatment.
Precision psychiatry could lead to:
- identification of novel targets for new medications
- pharmacogenetic profiling of the patient to predict disease susceptibility and medication response
- personalized therapy: the right drug at the right dose for the right patient.
- improved efficacy and fewer adverse medication reactions.
Continue to: Digital psychiatry
Digital psychiatry
Integrating computer-based technology into psychiatric practice has given birth to a new frontier that could be called digital psychiatry. This might encompass the following:
- telepsychiatry
- social media with a mental health focus
- web-based applications/devices
- artificial intelligence (AI).
Telepsychiatry. Videoconferencing is the most widely used form of telepsychiatry. It provides patients with easier access to mental health treatment.4 Telepsychiatry has the potential to match patients and clinicians with similar cultural backgrounds, thus minimizing cultural gaps and misunderstandings. Most importantly, it is comparable to face-to-face interviews in terms of the reliability of assessment and treatment outcomes.5
Telepsychiatry might be particularly helpful for patients with restricted mobility, such as those who live in remote areas, nursing homes, or correctional facilities. In correctional settings, transferring prisoners is expensive and carries the risk of escape. In a small study (N = 86) conducted in Hong Kong, Chen et al6 found that using videoconferencing to conduct clinical interviews of inmates was cost-efficient and scored high in terms of patient acceptability.
Social media. Social media could be a powerful platform for early detection of mental illness. Staying connected with patients on social media could allow psychiatrists to be more aware of their patient’s mood fluctuations, which might lead to more timely assessments. Physicians could be automatically notified about changes in their patients’ social media activity that indicate changes in mental state, which could solicit immediate intervention and treatment. On the other hand, such use of social media could blur professional boundaries. Psychiatrists also could use social media to promote awareness of mental health and educate the public on ways to improve or maintain their mental well-being.7
Web-based applications/devices. Real-time monitoring through applications or internet-based smart devices creates a new avenue for patients to receive personalized assessments, treatment, and intervention.8 Smartwatches with internet connectivity may offer a glimpse of the wearer’s sleep architecture and duration, thus providing real-time data on patients who have insomnia. We can now passively collect objective data from devices, such as smartphones and laptops, to phenotype an individual’s mood and mental state, a process called digital phenotyping. The Table9 lists examples of the types of mental health–related metrics that can be captured by smartphones, smartwatches, and similar technology. Information from these devices can be accumulated to create a database that can be used to predict symptoms.10 For example, the way people use a smartphone’s keyboard, including latency time between space and character types, can be used to generate variables for data. This type of information is being studied for use in screening depression and passively assessing mood in real time.11
Continue to: Artificial intelligence
Artificial intelligence—the development of computer systems able to perform tasks that normally require human intelligence—is being increasingly used in psychiatry. Some studies have suggested AI can be used to identify patients’ risk of suicide12-15 or psychosis.16,17 Kalanderian and Nasrallah18 reviewed several of these studies in
Other researchers have found clinical uses for machine learning, a subset of AI that uses methods to automatically detect patterns and make predictions based on those patterns. In one study, a machine learning analysis of functional MRI scans was able to identify 4 distinct subtypes of depression.19 In another study, a machine learning model was able to predict with 60% accuracy which patients with depression would respond to antidepressants.20
In the future, AI might be used to change mental health classification systems. Because many mental health disorders share similar symptom clusters, machine learning can help to identify associations between symptoms, behavior, brain function, and real-world function across different diagnoses, potentially affecting how we will classify mental disorders.21
Technology-enhanced psychotherapy
In the future, it might be common for psychotherapy to be provided by a computer, or “virtual therapist.” Several studies have evaluated the use of technology-enhanced psychotherapy.
Lucas et al22 investigated patients’ interactions with a virtual therapist. Participants were interviewed by an avatar named Ellie, who they saw on a TV screen. Half of the participants were told Ellie was not human, and half were told Ellie was being controlled remotely by a human. Three psychologists who were blinded to group allocations analyzed transcripts of the interviews and video recordings of participants’ facial expressions to quantify the participants’ fear, sadness, and other emotional responses during the interviews, as well as their openness to the questions. Participants who believed Ellie was fully automated reported significantly lower fear of self-disclosure and impression management (attempts to control how others perceive them) than participants who were told that Ellie was operated by a human. Additionally, participants who believed they were interacting with a computer were more open during the interview.22
Continue to: Researchers at the University of Southern California...
Researchers at the University of Southern California developed software that assessed 74 acoustic features, including pitch, volume, quality, shimmer, jitter, and prosody, to predict outcomes among patients receiving couples therapy. This software was able to predict marital discord at least as well as human therapists.23
Many mental health apps purport to implement specific components of psychotherapy. Many of these apps focus on cognitive-behavioral therapy worksheets, mindfulness exercises, and/or mood tracking. The features provided by such apps emulate the tasks and intended outcomes of traditional psychotherapy, but in an entirely decentralized venue.24
Some have expressed concern that an increased use of virtual therapists powered by AI might lead to a dehumanization of psychiatry (Box25,26).
Box
Whether there are aspects of the psychiatric patient encounter that cannot be managed by a “virtual clinician” created by artificial intelligence (AI) remains to be determined. Some of the benefits of using AI in this manner may be difficult to anticipate, or may be specific to an individual’s relationship with his/her clinician.25
On the other hand, AI systems blur previously assumed boundaries between reality and fiction, and this could have complex effects on patients. Similar to therapeutic relationships with a human clinician, there is the risk of transference of emotions, thoughts, and feelings to a virtual therapist powered by AI. Unlike with a psychiatrist or therapist, however, there is no person on the other side of this transference. Whether virtual clinicians will be able to manage such transference remains to be seen.
In Deep Medicine,26 cardiologist Eric Topol, MD, emphasizes a crucial component of a patient encounter that AI will be unlikely able to provide: empathy. Virtual therapists powered by AI will inherit the tasks best done by machines, leaving humans more time to do what they do best—providing empathy and being “present” for patients.
Electronic health record reforms
Although many clinicians find EHRs to be onerous and time-consuming, EHR technology is constantly improving, and EHRs have revolutionized documentation and order implementation. Several potential advances could improve clinical practice. For example, EHRs could incorporate a clinical decision support system that uses AI-based algorithms to assist psychiatrists with diagnosis, monitoring, and treatment.27 In the future, EHRs might have the ability to monitor and learn from errors and adverse events, and automatically design an algorithm to avoid them.28 They should be designed to better manage analysis of pharmacogenetic test results, which is challenging due to the amount and complexity of the data.29 Future EHRs should eliminate the non-intuitive and multi-click interfaces and cumbersome data searches of today’s EHRs.30
Technology brings new ethical considerations
Mental health interventions based on AI typically work with algorithms, and algorithms bring ethical issues. Mental health devices or systems that use AI could contain biases that have the potential to harm in unintended ways, such as a data-driven sexist or racist bias.31 This may require investing additional time to explain to patients (and their families) what an algorithm is and how it works in relation to the therapy provided.
Continue to: Another concern is patient...
Another concern is patient autonomy.32 For example, it would be ethically problematic if a patient were to assume that there was a human physician “at the other end” of a virtual therapist or other technology who is communicating or reviewing his/her messages. Similarly, an older adult or a patient with intellectual disabilities may not be able to understand advanced technology or what it does when it is installed in their home to monitor the patient’s activities. This would increase the risk of privacy violations, manipulation, or even coercion if the requirements for informed consent are not satisfied.
A flowchart for the future
Although current research and innovations typically target specific areas of psychiatry, these advances can be integrated by devising algorithms and protocols that will change the current practice of psychiatry. The Figure provides a glimpse of how the psychiatry clinic of the future might work. A maxim of management is that “the best way to predict the future is to create it.” However, the mere conception of a vision is not enough—working towards it is essential.
Bottom Line
With advances in technology, psychiatric practice will soon be radically different from what it is today. The expanded use of telepsychiatry, social media, artificial intelligence, and web-based applications/devices holds great promise for psychiatric assessment, diagnosis, and treatment, although certain ethical and privacy concerns need to be adequately addressed.
Related Resources
- National Institute of Mental Health. Technology and the future of mental health treatment. www.nimh.nih.gov/health/topics/technology-and-the-future-of-mental-health-treatment/index.shtml. Revised September 2019.
- Hays R, Farrell HM, Touros J. Mobile apps and mental health: using technology to quantify real-time clinical risk. Current Psychiatry. 2019;18(6):37-41.
- Torous J, Luo J, Chan SR. Mental health apps: what to tell patients. Current Psychiatry. 2018;17(3):21-25.
Despite the tremendous advances in psychiatry in recent years, the current clinical practice of psychiatry continues to rely on data from intermittent assessments along with subjective and unquantifiable accounts from patients and caregivers. Furthermore, there continues to be significant diagnostic variations among practitioners. Fortunately, technology to address these issues appears to be on the horizon.
How might the psychiatric clinic of the future look? What changes could we envision? These 4 critical factors may soon bring about dynamic changes in the way we practice psychiatry:
- precision psychiatry
- digital psychiatry
- technology-enhanced psychotherapy
- electronic health record (EHR) reforms.
In this article, we review how advances in each of these areas might lead to improved care for our patients.
Precision psychiatry
Precision psychiatry takes into account each patient’s variability in genes, environment, and lifestyle to determine individualized treatment and prevention strategies. It relies on pharmacogenomic testing as the primary tool. Pharmacogenomics is the study of variability in drug response due to heredity.
Emerging data on the clinical utility and cost-effectiveness of pharmacogenomic testing are encouraging, but its routine use is not well supported by current evidence.2 One limit to using pharmacogenomic testing is that many genes simultaneously exert an effect on the structure and function of neurons and associated pathophysiology. According to the International Society of Psychiatric Genetics, no single genetic variant is sufficient to cause psychiatric disorders such as depression, bipolar disorder, substance dependence, or schizophrenia. This limits the possibility of using genetic tests to establish a diagnosis.3
In the future, better algorithms could promote more accurate pharmacogenomics profiles for individual patients, which could influence treatment.
Precision psychiatry could lead to:
- identification of novel targets for new medications
- pharmacogenetic profiling of the patient to predict disease susceptibility and medication response
- personalized therapy: the right drug at the right dose for the right patient.
- improved efficacy and fewer adverse medication reactions.
Continue to: Digital psychiatry
Digital psychiatry
Integrating computer-based technology into psychiatric practice has given birth to a new frontier that could be called digital psychiatry. This might encompass the following:
- telepsychiatry
- social media with a mental health focus
- web-based applications/devices
- artificial intelligence (AI).
Telepsychiatry. Videoconferencing is the most widely used form of telepsychiatry. It provides patients with easier access to mental health treatment.4 Telepsychiatry has the potential to match patients and clinicians with similar cultural backgrounds, thus minimizing cultural gaps and misunderstandings. Most importantly, it is comparable to face-to-face interviews in terms of the reliability of assessment and treatment outcomes.5
Telepsychiatry might be particularly helpful for patients with restricted mobility, such as those who live in remote areas, nursing homes, or correctional facilities. In correctional settings, transferring prisoners is expensive and carries the risk of escape. In a small study (N = 86) conducted in Hong Kong, Chen et al6 found that using videoconferencing to conduct clinical interviews of inmates was cost-efficient and scored high in terms of patient acceptability.
Social media. Social media could be a powerful platform for early detection of mental illness. Staying connected with patients on social media could allow psychiatrists to be more aware of their patient’s mood fluctuations, which might lead to more timely assessments. Physicians could be automatically notified about changes in their patients’ social media activity that indicate changes in mental state, which could solicit immediate intervention and treatment. On the other hand, such use of social media could blur professional boundaries. Psychiatrists also could use social media to promote awareness of mental health and educate the public on ways to improve or maintain their mental well-being.7
Web-based applications/devices. Real-time monitoring through applications or internet-based smart devices creates a new avenue for patients to receive personalized assessments, treatment, and intervention.8 Smartwatches with internet connectivity may offer a glimpse of the wearer’s sleep architecture and duration, thus providing real-time data on patients who have insomnia. We can now passively collect objective data from devices, such as smartphones and laptops, to phenotype an individual’s mood and mental state, a process called digital phenotyping. The Table9 lists examples of the types of mental health–related metrics that can be captured by smartphones, smartwatches, and similar technology. Information from these devices can be accumulated to create a database that can be used to predict symptoms.10 For example, the way people use a smartphone’s keyboard, including latency time between space and character types, can be used to generate variables for data. This type of information is being studied for use in screening depression and passively assessing mood in real time.11
Continue to: Artificial intelligence
Artificial intelligence—the development of computer systems able to perform tasks that normally require human intelligence—is being increasingly used in psychiatry. Some studies have suggested AI can be used to identify patients’ risk of suicide12-15 or psychosis.16,17 Kalanderian and Nasrallah18 reviewed several of these studies in
Other researchers have found clinical uses for machine learning, a subset of AI that uses methods to automatically detect patterns and make predictions based on those patterns. In one study, a machine learning analysis of functional MRI scans was able to identify 4 distinct subtypes of depression.19 In another study, a machine learning model was able to predict with 60% accuracy which patients with depression would respond to antidepressants.20
In the future, AI might be used to change mental health classification systems. Because many mental health disorders share similar symptom clusters, machine learning can help to identify associations between symptoms, behavior, brain function, and real-world function across different diagnoses, potentially affecting how we will classify mental disorders.21
Technology-enhanced psychotherapy
In the future, it might be common for psychotherapy to be provided by a computer, or “virtual therapist.” Several studies have evaluated the use of technology-enhanced psychotherapy.
Lucas et al22 investigated patients’ interactions with a virtual therapist. Participants were interviewed by an avatar named Ellie, who they saw on a TV screen. Half of the participants were told Ellie was not human, and half were told Ellie was being controlled remotely by a human. Three psychologists who were blinded to group allocations analyzed transcripts of the interviews and video recordings of participants’ facial expressions to quantify the participants’ fear, sadness, and other emotional responses during the interviews, as well as their openness to the questions. Participants who believed Ellie was fully automated reported significantly lower fear of self-disclosure and impression management (attempts to control how others perceive them) than participants who were told that Ellie was operated by a human. Additionally, participants who believed they were interacting with a computer were more open during the interview.22
Continue to: Researchers at the University of Southern California...
Researchers at the University of Southern California developed software that assessed 74 acoustic features, including pitch, volume, quality, shimmer, jitter, and prosody, to predict outcomes among patients receiving couples therapy. This software was able to predict marital discord at least as well as human therapists.23
Many mental health apps purport to implement specific components of psychotherapy. Many of these apps focus on cognitive-behavioral therapy worksheets, mindfulness exercises, and/or mood tracking. The features provided by such apps emulate the tasks and intended outcomes of traditional psychotherapy, but in an entirely decentralized venue.24
Some have expressed concern that an increased use of virtual therapists powered by AI might lead to a dehumanization of psychiatry (Box25,26).
Box
Whether there are aspects of the psychiatric patient encounter that cannot be managed by a “virtual clinician” created by artificial intelligence (AI) remains to be determined. Some of the benefits of using AI in this manner may be difficult to anticipate, or may be specific to an individual’s relationship with his/her clinician.25
On the other hand, AI systems blur previously assumed boundaries between reality and fiction, and this could have complex effects on patients. Similar to therapeutic relationships with a human clinician, there is the risk of transference of emotions, thoughts, and feelings to a virtual therapist powered by AI. Unlike with a psychiatrist or therapist, however, there is no person on the other side of this transference. Whether virtual clinicians will be able to manage such transference remains to be seen.
In Deep Medicine,26 cardiologist Eric Topol, MD, emphasizes a crucial component of a patient encounter that AI will be unlikely able to provide: empathy. Virtual therapists powered by AI will inherit the tasks best done by machines, leaving humans more time to do what they do best—providing empathy and being “present” for patients.
Electronic health record reforms
Although many clinicians find EHRs to be onerous and time-consuming, EHR technology is constantly improving, and EHRs have revolutionized documentation and order implementation. Several potential advances could improve clinical practice. For example, EHRs could incorporate a clinical decision support system that uses AI-based algorithms to assist psychiatrists with diagnosis, monitoring, and treatment.27 In the future, EHRs might have the ability to monitor and learn from errors and adverse events, and automatically design an algorithm to avoid them.28 They should be designed to better manage analysis of pharmacogenetic test results, which is challenging due to the amount and complexity of the data.29 Future EHRs should eliminate the non-intuitive and multi-click interfaces and cumbersome data searches of today’s EHRs.30
Technology brings new ethical considerations
Mental health interventions based on AI typically work with algorithms, and algorithms bring ethical issues. Mental health devices or systems that use AI could contain biases that have the potential to harm in unintended ways, such as a data-driven sexist or racist bias.31 This may require investing additional time to explain to patients (and their families) what an algorithm is and how it works in relation to the therapy provided.
Continue to: Another concern is patient...
Another concern is patient autonomy.32 For example, it would be ethically problematic if a patient were to assume that there was a human physician “at the other end” of a virtual therapist or other technology who is communicating or reviewing his/her messages. Similarly, an older adult or a patient with intellectual disabilities may not be able to understand advanced technology or what it does when it is installed in their home to monitor the patient’s activities. This would increase the risk of privacy violations, manipulation, or even coercion if the requirements for informed consent are not satisfied.
A flowchart for the future
Although current research and innovations typically target specific areas of psychiatry, these advances can be integrated by devising algorithms and protocols that will change the current practice of psychiatry. The Figure provides a glimpse of how the psychiatry clinic of the future might work. A maxim of management is that “the best way to predict the future is to create it.” However, the mere conception of a vision is not enough—working towards it is essential.
Bottom Line
With advances in technology, psychiatric practice will soon be radically different from what it is today. The expanded use of telepsychiatry, social media, artificial intelligence, and web-based applications/devices holds great promise for psychiatric assessment, diagnosis, and treatment, although certain ethical and privacy concerns need to be adequately addressed.
Related Resources
- National Institute of Mental Health. Technology and the future of mental health treatment. www.nimh.nih.gov/health/topics/technology-and-the-future-of-mental-health-treatment/index.shtml. Revised September 2019.
- Hays R, Farrell HM, Touros J. Mobile apps and mental health: using technology to quantify real-time clinical risk. Current Psychiatry. 2019;18(6):37-41.
- Torous J, Luo J, Chan SR. Mental health apps: what to tell patients. Current Psychiatry. 2018;17(3):21-25.
1. Pirmohamed M. Pharmacogenetics and pharmacogenomics. Br J Clin Pharmacol. 2001;52(4):345-347.
2. Benitez J, Cool CL, Scotti DJ. Use of combinatorial pharmacogenomic guidance in treating psychiatric disorders. Per Med. 2018;15(6):481-494.
3. Cannon TD. Candidate gene studies in the GWAS era: the MET proto-oncogene, neurocognition, and schizophrenia. Am J Psychiatry. 2010;167(4):4,369-372.
4. Greenwood J, Chamberlain C, Parker G. Evaluation of a rural telepsychiatry service. Australas Psychiatry. 2004;12(3):268-272.
5. Hubley S, Lynch SB, Schneck C, et al. Review of key telepsychiatry outcomes. World J Psychiatry. 2016;6(2):269-282.
6. Cheng KM, Siu BW, Yeung CC, et al. Telepsychiatry for stable Chinese psychiatric out-patients in custody in Hong Kong: a case-control pilot study. Hong Kong Med J. 2018;24(4):378-383.
7. Frankish K, Ryan C, Harris A. Psychiatry and online social media: potential, pitfalls and ethical guidelines for psychiatrists and trainees. Australasian Psychiatry. 2012;20(3):181-187.
8. de la Torre Díez I, Alonso SG, Hamrioui S, et al. IoT-based services and applications for mental health in the literature. J Med Syst. 2019;43(1):4-9.
9. Topol E. Deep Medicine. New York, NY: Basic Books; 2019:168.
10. Adams RA, Huys QJM, Roiser JP. Computational Psychiatry: towards a mathematically informed understanding of mental illness. J Neurol Neurosurg Psychiatry. 2016;87(1):53-63.
11. Insel TR. Bending the curve for mental health: technology for a public health approach. Am J Public Health. 2019;109(suppl 3):S168-S170.
12. Just MA, Pan L, Cherkassky VL, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. 2017;1:911-919.
13. Pestian J, Nasrallah H, Matykiewicz P, et al. Suicide note classification using natural language processing: a content analysis. Biomed Inform Insights. 2010;2010(3):19-28.
14. Walsh CG, Ribeiro JD, Franklin JC. Predicting risk of suicide attempts over time through machine learning. Clinical Psychological Science. 2017;5(3):457-469.
15. Pestian JP, Sorter M, Connolly B, et al; STM Research Group. A machine learning approach to identifying the thought markers of suicidal subjects: a prospective multicenter trial. Suicide Life Threat Behav. 2017;47(1):112-121.
16. Corcoran CM, Carrillo F, Fernández-Slezak D, et al. Prediction of psychosis across protocols and risk cohorts using automated language analysis. World Psychiatry. 2018;17(1):67-75.
17. Bedi G, Carrillo F, Cecchi GA, et al. Automated analysis of free speech predicts psychosis onset in high-risk youths. NPJ Schizophr. 2015;1:15030. doi: 10.1038/npjschz.2015.30.
18. Kalanderian H, Nasrallah HA. Artificial intelligence in psychiatry. Current Psychiatry. 2019;18(8):33-38.
19. Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. 2017;23(1):28-38.
20. Chekroud AM, Zotti RJ, Shehzad Z, et al. Cross-trial prediction of treatment outcome in depression: a machine learning approach. Lancet Psychiatry. 2016;3(3):243-250.
21. Grisanzio KA, Goldstein-Piekarski AN, Wang MY, et al. Transdiagnostic symptom clusters and associations with brain, behavior, and daily function in mood, anxiety, and trauma disorders. JAMA Psychiatry. 2018;75(2):201-209.
22. Lucas G, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Computers in Human Behavior. 2014;37:94-100.
23. Nasir M, Baucom BR, Georgiou P, et al. Predicting couple therapy outcomes based on speech acoustic features. PLoS One. 2017;12(9):e0185123. doi: 10.1371/journal.pone.0185123.
24. Huguet A, Rao S, McGrath PJ, et al. A systematic review of cognitive behavioral therapy and behavioral activation apps for depression. PLoS One. 2016;11(5):e0154248. doi: 10.1371/journal.pone.0154248.
25. Scholten MR, Kelders SM, Van Gemert-Pijnen JE. Self-guided web-based interventions: scoping review on user needs and the potential of embodied conversational agents to address them. J Med Internet Res. 2017;19(11):e383.
26. Topol E. Deep Medicine. New York, NY: Basic Books; 2019:283-310.
27. Abramson EL, McGinnis S, Edwards A, et al. Electronic health record adoption and health information exchange among hospitals in New York State. J Eval Clin Pract. 2012;18(6):1156-1162.
28. Meeks DW, Smith MW, Taylor L, et al. An analysis of electronic health record-related patient safety concerns. J Am Med Inform Assoc. 2014;21(6):1053-1059.
29. Kho AN, Rasmussen LV, Connolly JJ, et al. Practical challenges in integrating genomic data into the electronic health record. Genet Med. 2013;15(10):772-778.
30. Ornstein SM, Oates RB, Fox GN. The computer-based medical record: current status. J Fam Pract. 1992;35(5):556-565.
31. Corea F. Machine ethics and artificial moral agents. In: Applied artificial intelligence: where AI can be used in business. Basel, Switzerland: Springer; 2019:33-41.
32. Beauchamp T, Childress J. Principles of biomedical ethics. 7th ed. New York, NY: Oxford University Press; 2012:44.
1. Pirmohamed M. Pharmacogenetics and pharmacogenomics. Br J Clin Pharmacol. 2001;52(4):345-347.
2. Benitez J, Cool CL, Scotti DJ. Use of combinatorial pharmacogenomic guidance in treating psychiatric disorders. Per Med. 2018;15(6):481-494.
3. Cannon TD. Candidate gene studies in the GWAS era: the MET proto-oncogene, neurocognition, and schizophrenia. Am J Psychiatry. 2010;167(4):4,369-372.
4. Greenwood J, Chamberlain C, Parker G. Evaluation of a rural telepsychiatry service. Australas Psychiatry. 2004;12(3):268-272.
5. Hubley S, Lynch SB, Schneck C, et al. Review of key telepsychiatry outcomes. World J Psychiatry. 2016;6(2):269-282.
6. Cheng KM, Siu BW, Yeung CC, et al. Telepsychiatry for stable Chinese psychiatric out-patients in custody in Hong Kong: a case-control pilot study. Hong Kong Med J. 2018;24(4):378-383.
7. Frankish K, Ryan C, Harris A. Psychiatry and online social media: potential, pitfalls and ethical guidelines for psychiatrists and trainees. Australasian Psychiatry. 2012;20(3):181-187.
8. de la Torre Díez I, Alonso SG, Hamrioui S, et al. IoT-based services and applications for mental health in the literature. J Med Syst. 2019;43(1):4-9.
9. Topol E. Deep Medicine. New York, NY: Basic Books; 2019:168.
10. Adams RA, Huys QJM, Roiser JP. Computational Psychiatry: towards a mathematically informed understanding of mental illness. J Neurol Neurosurg Psychiatry. 2016;87(1):53-63.
11. Insel TR. Bending the curve for mental health: technology for a public health approach. Am J Public Health. 2019;109(suppl 3):S168-S170.
12. Just MA, Pan L, Cherkassky VL, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. 2017;1:911-919.
13. Pestian J, Nasrallah H, Matykiewicz P, et al. Suicide note classification using natural language processing: a content analysis. Biomed Inform Insights. 2010;2010(3):19-28.
14. Walsh CG, Ribeiro JD, Franklin JC. Predicting risk of suicide attempts over time through machine learning. Clinical Psychological Science. 2017;5(3):457-469.
15. Pestian JP, Sorter M, Connolly B, et al; STM Research Group. A machine learning approach to identifying the thought markers of suicidal subjects: a prospective multicenter trial. Suicide Life Threat Behav. 2017;47(1):112-121.
16. Corcoran CM, Carrillo F, Fernández-Slezak D, et al. Prediction of psychosis across protocols and risk cohorts using automated language analysis. World Psychiatry. 2018;17(1):67-75.
17. Bedi G, Carrillo F, Cecchi GA, et al. Automated analysis of free speech predicts psychosis onset in high-risk youths. NPJ Schizophr. 2015;1:15030. doi: 10.1038/npjschz.2015.30.
18. Kalanderian H, Nasrallah HA. Artificial intelligence in psychiatry. Current Psychiatry. 2019;18(8):33-38.
19. Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. 2017;23(1):28-38.
20. Chekroud AM, Zotti RJ, Shehzad Z, et al. Cross-trial prediction of treatment outcome in depression: a machine learning approach. Lancet Psychiatry. 2016;3(3):243-250.
21. Grisanzio KA, Goldstein-Piekarski AN, Wang MY, et al. Transdiagnostic symptom clusters and associations with brain, behavior, and daily function in mood, anxiety, and trauma disorders. JAMA Psychiatry. 2018;75(2):201-209.
22. Lucas G, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Computers in Human Behavior. 2014;37:94-100.
23. Nasir M, Baucom BR, Georgiou P, et al. Predicting couple therapy outcomes based on speech acoustic features. PLoS One. 2017;12(9):e0185123. doi: 10.1371/journal.pone.0185123.
24. Huguet A, Rao S, McGrath PJ, et al. A systematic review of cognitive behavioral therapy and behavioral activation apps for depression. PLoS One. 2016;11(5):e0154248. doi: 10.1371/journal.pone.0154248.
25. Scholten MR, Kelders SM, Van Gemert-Pijnen JE. Self-guided web-based interventions: scoping review on user needs and the potential of embodied conversational agents to address them. J Med Internet Res. 2017;19(11):e383.
26. Topol E. Deep Medicine. New York, NY: Basic Books; 2019:283-310.
27. Abramson EL, McGinnis S, Edwards A, et al. Electronic health record adoption and health information exchange among hospitals in New York State. J Eval Clin Pract. 2012;18(6):1156-1162.
28. Meeks DW, Smith MW, Taylor L, et al. An analysis of electronic health record-related patient safety concerns. J Am Med Inform Assoc. 2014;21(6):1053-1059.
29. Kho AN, Rasmussen LV, Connolly JJ, et al. Practical challenges in integrating genomic data into the electronic health record. Genet Med. 2013;15(10):772-778.
30. Ornstein SM, Oates RB, Fox GN. The computer-based medical record: current status. J Fam Pract. 1992;35(5):556-565.
31. Corea F. Machine ethics and artificial moral agents. In: Applied artificial intelligence: where AI can be used in business. Basel, Switzerland: Springer; 2019:33-41.
32. Beauchamp T, Childress J. Principles of biomedical ethics. 7th ed. New York, NY: Oxford University Press; 2012:44.
Does your patient have the right to refuse medications?
Ms. T, age 48, is brought to the psychiatric emergency department after the police find her walking along the highway at 3:00
Once involuntarily committed, does Ms. T have the right to refuse treatment?
Every psychiatrist has faced the predicament of a patient who refuses treatment. This creates an ethical dilemma between respecting the patient’s autonomy vs forcing treatment to ameliorate symptoms and reduce suffering. This article addresses case law related to the models for administering psychiatric medications over objection. We also discuss case law regarding court-appointed guardianship, and treating medical issues without consent. While this article provides valuable information on these scenarios, it is crucial to remember that the legal processes required to administer medications over patient objection are state-specific. In order to ensure the best practice and patient care, you must research the legal procedures specific to your jurisdiction, consult your clinic/hospital attorney, and/or contact your state’s mental health board for further clarification.
History of involuntary treatment
Prior to the 1960s, Ms. T would likely have been unable to refuse treatment. All patients were considered involuntary, and the course of treatment was decided solely by the psychiatric institution. Well into the 20th century, patients with psychiatric illness remained feared and stigmatized, which led to potent and potentially harsh methods of treatment. Some patients experienced extreme isolation, whipping, bloodletting, experimental use of chemicals, and starvation (Table 11-3).
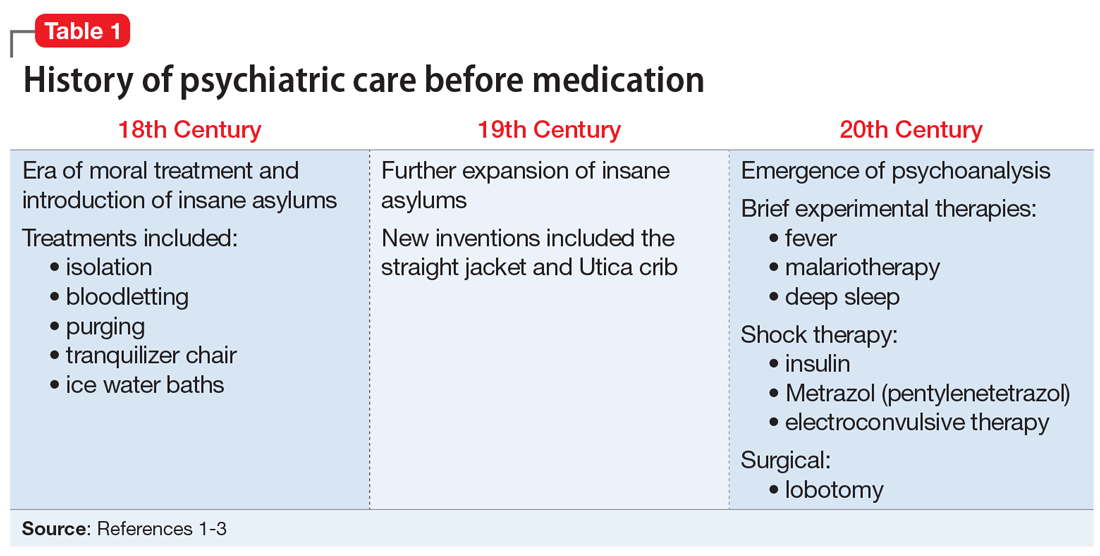
With the advent of psychotropic medications and a focus on civil liberties, the psychiatric mindset began to change from hospital-based treatment to a community-based approach. The value of psychotherapy was recognized, and by the 1960s, the establishment of community mental health centers was gaining momentum.
In the context of these changes, the civil rights movement pressed for stronger legislation regarding autonomy and the quality of treatment available to patients with psychiatric illness. In the 1960s and 1970s, Rouse v Cameron4 and Wyatt v Stickney5 dealt with a patient’s right to receive treatment while involuntarily committed. However, it was not until the 1980s that the courts addressed the issue of a patient’s right to refuse treatment.
The judicial system: A primer
When reviewing case law and its applicability to your patients, it is important to understand the various court systems. The judicial system is divided into state and federal courts, which are subdivided into trial, appellate, and supreme courts. When decisions at either the state or federal level require an ultimate decision maker, the US Supreme Court can choose to hear the case, or grant certiorari, and make a ruling, which is then binding law.6 Decisions made by any court are based on various degrees of stringency, called standards of proof (Table 27).
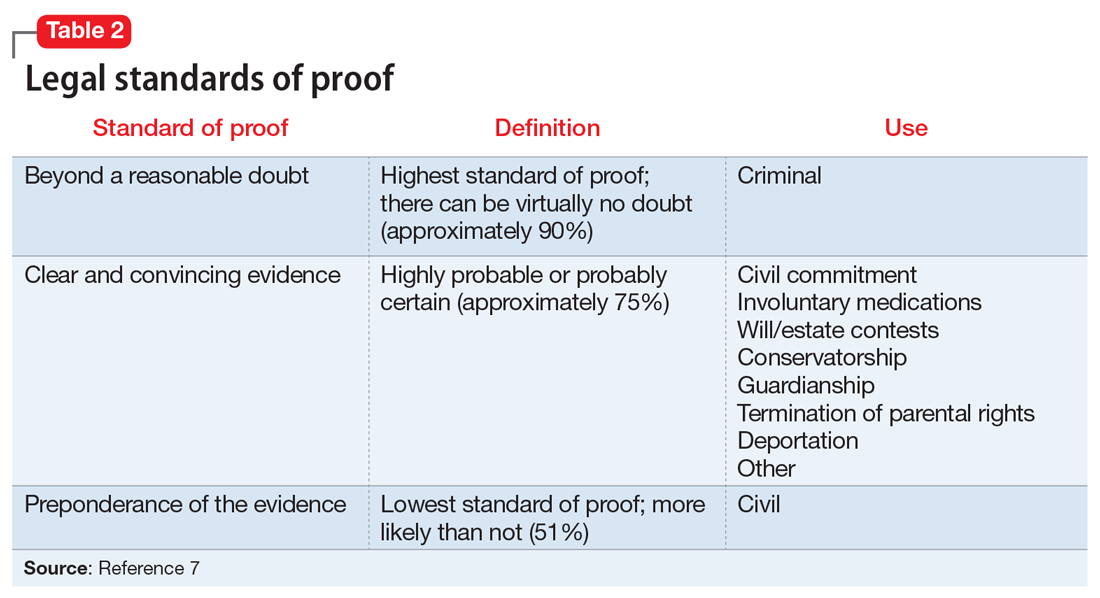
Continue to: For Ms. T's case...
For Ms. T’s case, civil commitment and involuntary medication hearings are held in probate court, which is a civil (not criminal) court. In addition to overseeing civil commitment and involuntary medications, probate courts adjudicate will and estate contests, conservatorship, and guardianship. Conservatorship hearings deal with financial issues, and guardianship cases encompass personal and health-related needs. Regardless of the court, an individual is guaranteed due process under the 5th Amendment (federal) and 14th Amendment (state).
Individuals are presumed competent to make their own decisions, but a court may call this into question. Competencies are specific to a variety of areas, such as criminal proceedings, medical decision making, writing a will (testimonial capacity), etc. Because each field applies its own standard of competence, an individual may be competent in one area but incompetent in another. Competence in medical decision making varies by state but generally consists of being able to communicate a choice, understand relevant information, appreciate one’s illness and its likely consequences, and rationally manipulate information.8
Box
Administering medications despite a patient’s objection differs from situations in which medications are provided during a psychiatric emergency. In an emergency, courts do not have time to weigh in. Instead, emergency medications (most often given as IM injections) are administered based on the physician’s clinical judgment. The criteria for psychiatric emergencies are delineated at the state level, but typically are defined as when a person with a mental illness creates an imminent risk of harm to self or others. Alternative approaches to resolving the emergency may include verbal de-escalation, quiet time in a room devoid of stimuli, locked seclusion, or physical restraints. These measures are often exhausted before emergency medications are administered.
Source: Reference 9
It is important to note that the legal process required before administering involuntary medications is distinct from situations in which medication needs to be provided during a psychiatric emergency. The Box9 outlines the difference between these 2 scenarios.

4 Legal models
There are several legal models used to determine when a patient can be administered psychiatric medications over objection. Table 310,11 summarizes these models.
Rights-driven (Rogers) model. If Ms. T was involuntarily hospitalized in Massachusetts or another state that adopted the rights-driven model, she would retain the right to refuse treatment. These states require an external judicial review, and court approval is necessary before imposing any therapy. This model was established in Rogers v Commissioner,12 where 7 patients at the Boston State Hospital filed a lawsuit regarding their right to refuse medications. The Massachusetts Supreme Judicial Court ruled that, despite being involuntarily committed, a patient is considered competent to refuse treatment until found specifically incompetent to do so by the court. If a patient is found incompetent, the judge, using a full adversarial hearing, decides what the incompetent patient would have wanted if he/she were competent. The judge reaches a conclusion based on the substituted judgment model (Table 410). In Rogers v Commissioner,12 the court ruled that the right to decision making is not lost after becoming a patient at a mental health facility. The right is lost only if the patient is found incompetent by the judge. Thus, every individual has the right to “manage his own person” and “take care of himself.”
Continue to: An update to the rights-driven (Rogers) model
An update to the rights-driven (Rogers) model. Other states, such as Ohio, have adopted the Rogers model and addressed issues that arose subsequent to the aforementioned case. In Steele v Hamilton County,13 Jeffrey Steele was admitted and later civilly committed to the hospital. After 2 months, an involuntary medication hearing was completed in which 3 psychiatrists concluded that, although Mr. Steele was not a danger to himself or others while in the hospital, he would ultimately benefit from medications.
The probate court acknowledged that Mr. Steele lacked capacity and required hospitalization. However, because he was not imminently dangerous, medication should not be used involuntarily. After a series of appeals, the Ohio Supreme Court ruled that a court may authorize the administration of an antipsychotic medication against a patient’s wishes without a finding of dangerousness when clear and convincing evidence exists that:
- the patient lacks the capacity to give or withhold informed consent regarding treatment
- the proposed medication is in the patient’s best interest
- no less intrusive treatment will be as effective in treating the mental illness.
This ruling set a precedent that dangerousness is not a requirement for involuntary medications.
Treatment-driven (Rennie) model. As in the rights-driven model, in the treatment-driven model, Ms. T would retain the constitutional right to refuse treatment. However, the models differ in the amount of procedural due process required. The treatment-driven model derives from Rennie v Klein,14 in which John Rennie, a patient at Ancora State Psychiatric Hospital in New Jersey, filed a suit regarding the right of involuntarily committed patients to refuse antipsychotic medications. The Third Circuit Court of Appeals ruled that, if professional judgment deems a patient to be a danger to himself or others, then antipsychotics may be administered over individual objection. This professional judgment is typically based on the opinion of the treating physician, along with a second physician or panel.
Utah model. This model is based on A.E. and R.R. v Mitchell,15 in which the Utah District Court ruled that a civilly committed patient has no right to refuse treatment. This Utah model was created after state legislature determined that, in order to civilly commit a patient, hospitalization must be the least restrictive alternative and the patient is incompetent to consent to treatment. Unlike the 2 previous models, competency to refuse medications is not separated from a previous finding of civil commitment, but rather, they occur simultaneously.
Continue to: Rights in unique situations
Rights in unique situations
Correctional settings. If Ms. T was an inmate, would her right to refuse psychiatric medication change? This was addressed in the case of Washington v Harper.16 Walter Harper, serving time for a robbery conviction, filed a claim that his civil rights were being violated when he received involuntary medications based on the decision of a 3-person panel consisting of a psychiatrist, psychologist, and prison official. The US Supreme Court ruled that this process provided sufficient due process to mandate providing psychotropic medications against a patient’s will. This reduction in required procedures is related to the unique nature of the correctional environment and an increased need to maintain safety. This need was felt to outweigh an individual’s right to refuse medication.
Incompetent to stand trial. In Sell v U.S.,17 Charles Sell, a dentist, was charged with fraud and attempted murder. He underwent a competency evaluation and was found incompetent to stand trial because of delusional thinking. Mr. Sell was hospitalized for restorability but refused medications. The hospital held an administrative hearing to proceed with involuntary antipsychotic medications; however, Mr. Sell filed an order with the court to prevent this. Eventually, the US Supreme Court ruled that non-dangerous, incompetent defendants may be involuntarily medicated even if they do not pose a risk to self or others on the basis that it furthers the state’s interest in bringing to trial those charged with serious crimes. However, the following conditions must be met before involuntary medication can be administered:
- an important government issue must be at stake (determined case-by-case)
- a substantial probability must exist that the medication will enable the defendant to become competent without significant adverse effects
- the medication must be medically appropriate and necessary to restore competency, with no less restrictive alternative available.
This case suggests that, before one attempts to forcibly medicate a defendant for the purpose of competency restoration, one should exhaust the same judicial remedies one uses for civil patients first.
Court-appointed guardianship
In the case of Ms. T, what if her father requested to become her guardian? This question was explored in the matter of Guardianship of Richard Roe III.18 Mr. Roe was admitted to the Northampton State Hospital in Massachusetts, where he refused antipsychotic medications. Prior to his release, his father asked to be his guardian. The probate court obliged the request. However, Mr. Roe’s lawyer and guardian ad litem (a neutral temporary guardian often appointed when legal issues are pending) challenged the ruling, arguing the probate court cannot empower the guardian to consent to involuntary medication administration. On appeal, the court ruled:
- the guardianship was justified
- the standard of proof for establishment of a guardianship is preponderance of the evidence (Table 27)
- the guardian must seek from a court a “substituted judgment” to authorize forcible administration of antipsychotic medication.
The decision to establish the court as the final decision maker is based on the view that a patient’s relatives may be biased. Courts should take an objective approach that considers
- patient preference stated during periods of competency
- medication adverse effects
- consequences if treatment is refused
- prognosis with treatment
- religious beliefs
- impact on the patient’s family.
Continue to: This case set the stage for...
This case set the stage for later decisions that placed antipsychotic medications in the same category as electroconvulsive therapy and psychosurgery. This could mean a guardian would need specialized authorization to request antipsychotic treatment but could consent to an appendectomy without legal issue.
Fortunately, now most jurisdictions have remedied this cumbersome solution by requiring a higher standard of proof, clear and convincing evidence (Table 27), to establish guardianship but allowing the guardian more latitude to make decisions for their wards (such as those involving hospital admission or medications) without further court involvement.
Involuntary medical treatment
In order for a patient to consent for medical treatment, he/she must have the capacity to do so (Table 59). How do the courts handle the patient’s right to refuse medical treatment? This was addressed in the case of Georgetown College v Jones.19 Mrs. Jones, a 25-year-old Jehovah’s Witness and mother of a 7-month-old baby, suffered a ruptured ulcer and lost a life-threatening amount of blood. Due to her religious beliefs, Mrs. Jones refused a blood transfusion. The hospital quickly appealed to the court, who ruled the woman was help-seeking by going to the hospital, did not want to die, was in distress, and lacked capacity to make medical decisions. Acting in a parens patriae manner (when the government steps in to make decisions for its citizens who cannot), the court ordered the hospital to administer blood transfusions.
Proxy decision maker. When the situation is less emergent, a proxy decision maker can be appointed by the court. This was addressed in the case of Superintendent of Belchertown v Saikewicz.20 Mr. Saikewicz, a 67-year-old man with intellectual disability, was diagnosed with cancer and given weeks to months to live without treatment. However, treatment was only 50% effective and could potentially cause severe adverse effects. A guardian ad litem was appointed and recommended nontreatment, which the court upheld. The court ruled that the right to accept or reject medical treatment applies to both incompetent and competent persons. With incompetent persons, a “substituted judgment” analysis is used over the “best interest of the patient” doctrine.20 This falls in line with the Guardianship of Richard Roe III ruling,18 in which the court’s substituted judgment standard is enacted in an effort to respect patient autonomy.
Right to die. When does a patient have the right to die and what is the standard of proof? The US Supreme Court case Cruzan v Director21 addressed this. Nancy Cruzan was involved in a car crash, which left her in a persistent vegetative state with no significant cognitive function. She remained this way for 6 years before her parents sought to terminate life support. The hospital refused. The Missouri Supreme Court ruled that a standard of clear and convincing evidence (Table 27) is required to withdraw treatment, and in a 5-to-4 decision, the US Supreme Court upheld Missouri’s decision. This set the national standard for withdrawal of life-sustaining treatment. The moderate standard of proof is based on the court’s ruling that the decision to terminate life is a particularly important one.
Continue to: CASE
CASE CONTINUED
After having been civilly committed to your inpatient psychiatric facility, Ms. T’s paranoia and disorganized behavior persist. She continues to refuse medications.
There are 3 options: respect her decision, negotiate with her, or attempt to force medications through due process.11 In negotiating a compromise, it is best to understand the barriers to treatment. A patient may refuse medications due to poor insight into his/her illness, medication adverse effects, a preference for an alternative treatment, delusional concerns over contamination and/or poisoning, interpersonal conflicts with the treatment staff, a preference for symptoms (eg, mania) over wellness, medication ineffectiveness, length of treatment course, or stigma.22,23 However, a patient’s unwillingness to compromise creates the dilemma of autonomy vs treatment.
For Ms. T, the treatment team felt initiating involuntary medication was the best option for her quality of life and safety. Because she resides in Ohio, a Rogers-like model was applied. The probate court was petitioned and found her incompetent to make medical decisions. The court accepted the physician’s recommendation of treatment with antipsychotic medications. If this scenario took place in New Jersey, a Rennie model would apply, requiring due process through the second opinion of another physician. Lastly, if Ms. T lived in Utah, she would have been unable to refuse medications once civilly committed.
Pros and cons of each model
Over the years, various concerns about each of these models have been raised. Given the slow-moving wheels of justice, one concern was that perhaps patients would be left “rotting with their rights on,” or lingering in a psychotic state out of respect for their civil liberties.19 While court hearings do not always happen quickly, more often than not, a judge will agree with the psychiatrist seeking treatment because the judge likely has little experience with mental illness and will defer to the physician’s expertise. This means the Rogers model may be more likely to produce the desired outcome, just more slowly. With respect to the Rennie model, although it is often more expeditious, the second opinion of an independent psychiatrist may contradict that of the original physician because the consultant will rely on his/her own expertise. Finally, some were concerned that psychiatrists would view the Utah model as carte blanche to start whatever medications they wanted with no respect for patient preference. Based on our clinical experience, none of these concerns have come to fruition over time, and patients safely receive medications over objection in hospitals every day.
Consider why the patient refuses medication
Regardless of which involuntary medication model is employed, it is important to consider the underlying cause for medication refusal, because it may affect future compliance. If the refusal is the result of a religious belief, history of adverse effects, or other rational motive, then it may be reasonable to respect the patient’s autonomy.24 However, if the refusal is secondary to symptoms of mental illness, it is appropriate to move forward with an involuntary medication hearing and treat the underlying condition.
Continue to: In the case of Ms. T...
In the case of Ms. T, she appeared to be refusing medications because of her psychotic symptoms, which could be effectively treated with antipsychotic medications. Therefore, Ms. T’s current lack of capacity is hopefully a transient phenomenon that can be ameliorated by initiating medication. Typically, antipsychotic medications begin to reduce psychotic symptoms within the first week, with further improvement over time.25 The value of the inpatient psychiatric setting is that it allows for daily monitoring of a patient’s response to treatment. As capacity is regained, patient autonomy over medical decisions is reinstated.
Bottom Line
The legal processes required to administer medications over a patient’s objection are state-specific, and multiple models are used. In general, a patient’s right to refuse treatment can be overruled by obtaining adjudication through the courts (Rogers model) or the opinion of a second physician (Rennie model). In order to ensure the best practice and patient care, research the legal procedure specific to your jurisdiction, consult your clinic/hospital attorney, and/or contact your state’s mental health board for further clarification.
Related Resources
- Miller D. Is forced treatment in our outpatients’ best interests? Clinical Psychiatry News. https://www.mdedge.com/psychiatry/article/80277/forced-treatment-our-outpatients-best-interests.
- Miller D, Hanson A. Committed: The battle over involuntary psychiatric care. Baltimore, MD: Johns Hopkins University Press; 2016.
1. Laffey P. Psychiatric therapy in Georgian Britain. Psychol Med. 2003;33(7):1285-1297.
2. Porter R. Madness: a brief history. New York, NY: Oxford Press; 2002.
3. Stetka B, Watson J. Odd and outlandish psychiatric treatments throughout history. Medscape Psychiatry. https://www.medscape.com/features/slideshow/odd-psychiatric-treatments. Published April 13, 2016. Accessed February 26, 2020.
4. Rouse v Cameron, 373, F2d 451 (DC Cir 1966).
5. Wyatt v Stickney, 325 F Supp 781 (MD Ala 1971).
6. Administrative Office of the US Courts. Comparing federal and state Courts. United States Courts. https://www.uscourts.gov/about-federal-courts/court-role-and-structure/comparing-federal-state-courts. Accessed February 26, 2020.
7. Drogin E, Williams C. Introduction to the Legal System. In: Gold L, Frierson R, eds. Textbook of forensic psychiatry, 3rd ed. Arlington, VA: American Psychiatric Association Publishing; 2018:80-83.
8. Appelbaum P, Grisso T. Assessing patients’ capacities to consent to treatment. N Engl J Med. 1988;319(25):1635-1638.
9. Kambam P. Informed consent and competence. In: Rosnar R, Scott C, eds. Principles and practice of forensic psychiatry, 3rd ed. Boca Raton, FL: CRC press; 2017:115-121.
10. Wall B, Anfang S. Legal regulation of psychiatric treatment. In: Gold L, Frierson R, eds. Textbook of forensic psychiatry, 3rd ed. Arlington, VA: American Psychiatric Association Publishing; 2018:306-333.
11. Pinals D, Nesbit A, Hoge S. Treatment refusal in psychiatric practice. In: Rosnar R, Scott C, eds. Principles and practice of forensic psychiatry, 3rd ed. Boca Raton, FL: CRC press; 2017:155-163.
12. Rogers v Commissioner, 390 489 (Mass 1983).
13. Steele v Hamilton County, 90 Ohio St3d 176 (Ohio 2000).
14. Rennie v Klein, 462 F Supp 1131 (D NJ 1978).
15. AE and RR v Mitchell, 724 F.2d 864 (10th Cir 1983).
16. Washington v Harper, 494 US 210 (1990).
17. Sell v US, 539 US 166 (2003).
18. Guardianship of Richard Roe III, 383 415, 435 (Mass 1981).
19. Georgetown College v Jones, 331 F2d 1010 (DC Cir 1964).
20. Superintendent of Belchertown v Saikewicz, 370 NE 2d 417 (1977).
21. Cruzan v Director, 497 US 261 (1990).
22. Owiti J, Bowers L. A literature review: refusal of psychotropic medication in acute inpatient psychiatric care. J Psychiatr Ment Health Nurs. 2011;18(7):637-647.
23. Appelbaum P, Gutheil T. “Rotting with their rights on”: constitutional theory and clinical reality in drug refusal by psychiatric patients. Bull Am Acad Psychiatry Law. 1979;7(3):306-315.
24. Adelugba OO, Mela M, Haq IU. Psychotropic medication refusal: reasons and patients’ perception at a secure forensic psychiatric treatment centre. J Forensic Sci Med. 2016;2(1):12-17.
25. Agid O, Kapur S, Arenovich T, et al. Delayed-onset hypothesis of antipsychotic action: a hypothesis tested and rejected. Arch Gen Psychiatry. 2003;60(12):1228.
Ms. T, age 48, is brought to the psychiatric emergency department after the police find her walking along the highway at 3:00
Once involuntarily committed, does Ms. T have the right to refuse treatment?
Every psychiatrist has faced the predicament of a patient who refuses treatment. This creates an ethical dilemma between respecting the patient’s autonomy vs forcing treatment to ameliorate symptoms and reduce suffering. This article addresses case law related to the models for administering psychiatric medications over objection. We also discuss case law regarding court-appointed guardianship, and treating medical issues without consent. While this article provides valuable information on these scenarios, it is crucial to remember that the legal processes required to administer medications over patient objection are state-specific. In order to ensure the best practice and patient care, you must research the legal procedures specific to your jurisdiction, consult your clinic/hospital attorney, and/or contact your state’s mental health board for further clarification.
History of involuntary treatment
Prior to the 1960s, Ms. T would likely have been unable to refuse treatment. All patients were considered involuntary, and the course of treatment was decided solely by the psychiatric institution. Well into the 20th century, patients with psychiatric illness remained feared and stigmatized, which led to potent and potentially harsh methods of treatment. Some patients experienced extreme isolation, whipping, bloodletting, experimental use of chemicals, and starvation (Table 11-3).

With the advent of psychotropic medications and a focus on civil liberties, the psychiatric mindset began to change from hospital-based treatment to a community-based approach. The value of psychotherapy was recognized, and by the 1960s, the establishment of community mental health centers was gaining momentum.
In the context of these changes, the civil rights movement pressed for stronger legislation regarding autonomy and the quality of treatment available to patients with psychiatric illness. In the 1960s and 1970s, Rouse v Cameron4 and Wyatt v Stickney5 dealt with a patient’s right to receive treatment while involuntarily committed. However, it was not until the 1980s that the courts addressed the issue of a patient’s right to refuse treatment.
The judicial system: A primer
When reviewing case law and its applicability to your patients, it is important to understand the various court systems. The judicial system is divided into state and federal courts, which are subdivided into trial, appellate, and supreme courts. When decisions at either the state or federal level require an ultimate decision maker, the US Supreme Court can choose to hear the case, or grant certiorari, and make a ruling, which is then binding law.6 Decisions made by any court are based on various degrees of stringency, called standards of proof (Table 27).

Continue to: For Ms. T's case...
For Ms. T’s case, civil commitment and involuntary medication hearings are held in probate court, which is a civil (not criminal) court. In addition to overseeing civil commitment and involuntary medications, probate courts adjudicate will and estate contests, conservatorship, and guardianship. Conservatorship hearings deal with financial issues, and guardianship cases encompass personal and health-related needs. Regardless of the court, an individual is guaranteed due process under the 5th Amendment (federal) and 14th Amendment (state).
Individuals are presumed competent to make their own decisions, but a court may call this into question. Competencies are specific to a variety of areas, such as criminal proceedings, medical decision making, writing a will (testimonial capacity), etc. Because each field applies its own standard of competence, an individual may be competent in one area but incompetent in another. Competence in medical decision making varies by state but generally consists of being able to communicate a choice, understand relevant information, appreciate one’s illness and its likely consequences, and rationally manipulate information.8
Box
Administering medications despite a patient’s objection differs from situations in which medications are provided during a psychiatric emergency. In an emergency, courts do not have time to weigh in. Instead, emergency medications (most often given as IM injections) are administered based on the physician’s clinical judgment. The criteria for psychiatric emergencies are delineated at the state level, but typically are defined as when a person with a mental illness creates an imminent risk of harm to self or others. Alternative approaches to resolving the emergency may include verbal de-escalation, quiet time in a room devoid of stimuli, locked seclusion, or physical restraints. These measures are often exhausted before emergency medications are administered.
Source: Reference 9
It is important to note that the legal process required before administering involuntary medications is distinct from situations in which medication needs to be provided during a psychiatric emergency. The Box9 outlines the difference between these 2 scenarios.

4 Legal models
There are several legal models used to determine when a patient can be administered psychiatric medications over objection. Table 310,11 summarizes these models.
Rights-driven (Rogers) model. If Ms. T was involuntarily hospitalized in Massachusetts or another state that adopted the rights-driven model, she would retain the right to refuse treatment. These states require an external judicial review, and court approval is necessary before imposing any therapy. This model was established in Rogers v Commissioner,12 where 7 patients at the Boston State Hospital filed a lawsuit regarding their right to refuse medications. The Massachusetts Supreme Judicial Court ruled that, despite being involuntarily committed, a patient is considered competent to refuse treatment until found specifically incompetent to do so by the court. If a patient is found incompetent, the judge, using a full adversarial hearing, decides what the incompetent patient would have wanted if he/she were competent. The judge reaches a conclusion based on the substituted judgment model (Table 410). In Rogers v Commissioner,12 the court ruled that the right to decision making is not lost after becoming a patient at a mental health facility. The right is lost only if the patient is found incompetent by the judge. Thus, every individual has the right to “manage his own person” and “take care of himself.”
Continue to: An update to the rights-driven (Rogers) model
An update to the rights-driven (Rogers) model. Other states, such as Ohio, have adopted the Rogers model and addressed issues that arose subsequent to the aforementioned case. In Steele v Hamilton County,13 Jeffrey Steele was admitted and later civilly committed to the hospital. After 2 months, an involuntary medication hearing was completed in which 3 psychiatrists concluded that, although Mr. Steele was not a danger to himself or others while in the hospital, he would ultimately benefit from medications.
The probate court acknowledged that Mr. Steele lacked capacity and required hospitalization. However, because he was not imminently dangerous, medication should not be used involuntarily. After a series of appeals, the Ohio Supreme Court ruled that a court may authorize the administration of an antipsychotic medication against a patient’s wishes without a finding of dangerousness when clear and convincing evidence exists that:
- the patient lacks the capacity to give or withhold informed consent regarding treatment
- the proposed medication is in the patient’s best interest
- no less intrusive treatment will be as effective in treating the mental illness.
This ruling set a precedent that dangerousness is not a requirement for involuntary medications.
Treatment-driven (Rennie) model. As in the rights-driven model, in the treatment-driven model, Ms. T would retain the constitutional right to refuse treatment. However, the models differ in the amount of procedural due process required. The treatment-driven model derives from Rennie v Klein,14 in which John Rennie, a patient at Ancora State Psychiatric Hospital in New Jersey, filed a suit regarding the right of involuntarily committed patients to refuse antipsychotic medications. The Third Circuit Court of Appeals ruled that, if professional judgment deems a patient to be a danger to himself or others, then antipsychotics may be administered over individual objection. This professional judgment is typically based on the opinion of the treating physician, along with a second physician or panel.
Utah model. This model is based on A.E. and R.R. v Mitchell,15 in which the Utah District Court ruled that a civilly committed patient has no right to refuse treatment. This Utah model was created after state legislature determined that, in order to civilly commit a patient, hospitalization must be the least restrictive alternative and the patient is incompetent to consent to treatment. Unlike the 2 previous models, competency to refuse medications is not separated from a previous finding of civil commitment, but rather, they occur simultaneously.
Continue to: Rights in unique situations
Rights in unique situations
Correctional settings. If Ms. T was an inmate, would her right to refuse psychiatric medication change? This was addressed in the case of Washington v Harper.16 Walter Harper, serving time for a robbery conviction, filed a claim that his civil rights were being violated when he received involuntary medications based on the decision of a 3-person panel consisting of a psychiatrist, psychologist, and prison official. The US Supreme Court ruled that this process provided sufficient due process to mandate providing psychotropic medications against a patient’s will. This reduction in required procedures is related to the unique nature of the correctional environment and an increased need to maintain safety. This need was felt to outweigh an individual’s right to refuse medication.
Incompetent to stand trial. In Sell v U.S.,17 Charles Sell, a dentist, was charged with fraud and attempted murder. He underwent a competency evaluation and was found incompetent to stand trial because of delusional thinking. Mr. Sell was hospitalized for restorability but refused medications. The hospital held an administrative hearing to proceed with involuntary antipsychotic medications; however, Mr. Sell filed an order with the court to prevent this. Eventually, the US Supreme Court ruled that non-dangerous, incompetent defendants may be involuntarily medicated even if they do not pose a risk to self or others on the basis that it furthers the state’s interest in bringing to trial those charged with serious crimes. However, the following conditions must be met before involuntary medication can be administered:
- an important government issue must be at stake (determined case-by-case)
- a substantial probability must exist that the medication will enable the defendant to become competent without significant adverse effects
- the medication must be medically appropriate and necessary to restore competency, with no less restrictive alternative available.
This case suggests that, before one attempts to forcibly medicate a defendant for the purpose of competency restoration, one should exhaust the same judicial remedies one uses for civil patients first.
Court-appointed guardianship
In the case of Ms. T, what if her father requested to become her guardian? This question was explored in the matter of Guardianship of Richard Roe III.18 Mr. Roe was admitted to the Northampton State Hospital in Massachusetts, where he refused antipsychotic medications. Prior to his release, his father asked to be his guardian. The probate court obliged the request. However, Mr. Roe’s lawyer and guardian ad litem (a neutral temporary guardian often appointed when legal issues are pending) challenged the ruling, arguing the probate court cannot empower the guardian to consent to involuntary medication administration. On appeal, the court ruled:
- the guardianship was justified
- the standard of proof for establishment of a guardianship is preponderance of the evidence (Table 27)
- the guardian must seek from a court a “substituted judgment” to authorize forcible administration of antipsychotic medication.
The decision to establish the court as the final decision maker is based on the view that a patient’s relatives may be biased. Courts should take an objective approach that considers
- patient preference stated during periods of competency
- medication adverse effects
- consequences if treatment is refused
- prognosis with treatment
- religious beliefs
- impact on the patient’s family.
Continue to: This case set the stage for...
This case set the stage for later decisions that placed antipsychotic medications in the same category as electroconvulsive therapy and psychosurgery. This could mean a guardian would need specialized authorization to request antipsychotic treatment but could consent to an appendectomy without legal issue.
Fortunately, now most jurisdictions have remedied this cumbersome solution by requiring a higher standard of proof, clear and convincing evidence (Table 27), to establish guardianship but allowing the guardian more latitude to make decisions for their wards (such as those involving hospital admission or medications) without further court involvement.
Involuntary medical treatment
In order for a patient to consent for medical treatment, he/she must have the capacity to do so (Table 59). How do the courts handle the patient’s right to refuse medical treatment? This was addressed in the case of Georgetown College v Jones.19 Mrs. Jones, a 25-year-old Jehovah’s Witness and mother of a 7-month-old baby, suffered a ruptured ulcer and lost a life-threatening amount of blood. Due to her religious beliefs, Mrs. Jones refused a blood transfusion. The hospital quickly appealed to the court, who ruled the woman was help-seeking by going to the hospital, did not want to die, was in distress, and lacked capacity to make medical decisions. Acting in a parens patriae manner (when the government steps in to make decisions for its citizens who cannot), the court ordered the hospital to administer blood transfusions.
Proxy decision maker. When the situation is less emergent, a proxy decision maker can be appointed by the court. This was addressed in the case of Superintendent of Belchertown v Saikewicz.20 Mr. Saikewicz, a 67-year-old man with intellectual disability, was diagnosed with cancer and given weeks to months to live without treatment. However, treatment was only 50% effective and could potentially cause severe adverse effects. A guardian ad litem was appointed and recommended nontreatment, which the court upheld. The court ruled that the right to accept or reject medical treatment applies to both incompetent and competent persons. With incompetent persons, a “substituted judgment” analysis is used over the “best interest of the patient” doctrine.20 This falls in line with the Guardianship of Richard Roe III ruling,18 in which the court’s substituted judgment standard is enacted in an effort to respect patient autonomy.
Right to die. When does a patient have the right to die and what is the standard of proof? The US Supreme Court case Cruzan v Director21 addressed this. Nancy Cruzan was involved in a car crash, which left her in a persistent vegetative state with no significant cognitive function. She remained this way for 6 years before her parents sought to terminate life support. The hospital refused. The Missouri Supreme Court ruled that a standard of clear and convincing evidence (Table 27) is required to withdraw treatment, and in a 5-to-4 decision, the US Supreme Court upheld Missouri’s decision. This set the national standard for withdrawal of life-sustaining treatment. The moderate standard of proof is based on the court’s ruling that the decision to terminate life is a particularly important one.
Continue to: CASE
CASE CONTINUED
After having been civilly committed to your inpatient psychiatric facility, Ms. T’s paranoia and disorganized behavior persist. She continues to refuse medications.
There are 3 options: respect her decision, negotiate with her, or attempt to force medications through due process.11 In negotiating a compromise, it is best to understand the barriers to treatment. A patient may refuse medications due to poor insight into his/her illness, medication adverse effects, a preference for an alternative treatment, delusional concerns over contamination and/or poisoning, interpersonal conflicts with the treatment staff, a preference for symptoms (eg, mania) over wellness, medication ineffectiveness, length of treatment course, or stigma.22,23 However, a patient’s unwillingness to compromise creates the dilemma of autonomy vs treatment.
For Ms. T, the treatment team felt initiating involuntary medication was the best option for her quality of life and safety. Because she resides in Ohio, a Rogers-like model was applied. The probate court was petitioned and found her incompetent to make medical decisions. The court accepted the physician’s recommendation of treatment with antipsychotic medications. If this scenario took place in New Jersey, a Rennie model would apply, requiring due process through the second opinion of another physician. Lastly, if Ms. T lived in Utah, she would have been unable to refuse medications once civilly committed.
Pros and cons of each model
Over the years, various concerns about each of these models have been raised. Given the slow-moving wheels of justice, one concern was that perhaps patients would be left “rotting with their rights on,” or lingering in a psychotic state out of respect for their civil liberties.19 While court hearings do not always happen quickly, more often than not, a judge will agree with the psychiatrist seeking treatment because the judge likely has little experience with mental illness and will defer to the physician’s expertise. This means the Rogers model may be more likely to produce the desired outcome, just more slowly. With respect to the Rennie model, although it is often more expeditious, the second opinion of an independent psychiatrist may contradict that of the original physician because the consultant will rely on his/her own expertise. Finally, some were concerned that psychiatrists would view the Utah model as carte blanche to start whatever medications they wanted with no respect for patient preference. Based on our clinical experience, none of these concerns have come to fruition over time, and patients safely receive medications over objection in hospitals every day.
Consider why the patient refuses medication
Regardless of which involuntary medication model is employed, it is important to consider the underlying cause for medication refusal, because it may affect future compliance. If the refusal is the result of a religious belief, history of adverse effects, or other rational motive, then it may be reasonable to respect the patient’s autonomy.24 However, if the refusal is secondary to symptoms of mental illness, it is appropriate to move forward with an involuntary medication hearing and treat the underlying condition.
Continue to: In the case of Ms. T...
In the case of Ms. T, she appeared to be refusing medications because of her psychotic symptoms, which could be effectively treated with antipsychotic medications. Therefore, Ms. T’s current lack of capacity is hopefully a transient phenomenon that can be ameliorated by initiating medication. Typically, antipsychotic medications begin to reduce psychotic symptoms within the first week, with further improvement over time.25 The value of the inpatient psychiatric setting is that it allows for daily monitoring of a patient’s response to treatment. As capacity is regained, patient autonomy over medical decisions is reinstated.
Bottom Line
The legal processes required to administer medications over a patient’s objection are state-specific, and multiple models are used. In general, a patient’s right to refuse treatment can be overruled by obtaining adjudication through the courts (Rogers model) or the opinion of a second physician (Rennie model). In order to ensure the best practice and patient care, research the legal procedure specific to your jurisdiction, consult your clinic/hospital attorney, and/or contact your state’s mental health board for further clarification.
Related Resources
- Miller D. Is forced treatment in our outpatients’ best interests? Clinical Psychiatry News. https://www.mdedge.com/psychiatry/article/80277/forced-treatment-our-outpatients-best-interests.
- Miller D, Hanson A. Committed: The battle over involuntary psychiatric care. Baltimore, MD: Johns Hopkins University Press; 2016.
Ms. T, age 48, is brought to the psychiatric emergency department after the police find her walking along the highway at 3:00
Once involuntarily committed, does Ms. T have the right to refuse treatment?
Every psychiatrist has faced the predicament of a patient who refuses treatment. This creates an ethical dilemma between respecting the patient’s autonomy vs forcing treatment to ameliorate symptoms and reduce suffering. This article addresses case law related to the models for administering psychiatric medications over objection. We also discuss case law regarding court-appointed guardianship, and treating medical issues without consent. While this article provides valuable information on these scenarios, it is crucial to remember that the legal processes required to administer medications over patient objection are state-specific. In order to ensure the best practice and patient care, you must research the legal procedures specific to your jurisdiction, consult your clinic/hospital attorney, and/or contact your state’s mental health board for further clarification.
History of involuntary treatment
Prior to the 1960s, Ms. T would likely have been unable to refuse treatment. All patients were considered involuntary, and the course of treatment was decided solely by the psychiatric institution. Well into the 20th century, patients with psychiatric illness remained feared and stigmatized, which led to potent and potentially harsh methods of treatment. Some patients experienced extreme isolation, whipping, bloodletting, experimental use of chemicals, and starvation (Table 11-3).

With the advent of psychotropic medications and a focus on civil liberties, the psychiatric mindset began to change from hospital-based treatment to a community-based approach. The value of psychotherapy was recognized, and by the 1960s, the establishment of community mental health centers was gaining momentum.
In the context of these changes, the civil rights movement pressed for stronger legislation regarding autonomy and the quality of treatment available to patients with psychiatric illness. In the 1960s and 1970s, Rouse v Cameron4 and Wyatt v Stickney5 dealt with a patient’s right to receive treatment while involuntarily committed. However, it was not until the 1980s that the courts addressed the issue of a patient’s right to refuse treatment.
The judicial system: A primer
When reviewing case law and its applicability to your patients, it is important to understand the various court systems. The judicial system is divided into state and federal courts, which are subdivided into trial, appellate, and supreme courts. When decisions at either the state or federal level require an ultimate decision maker, the US Supreme Court can choose to hear the case, or grant certiorari, and make a ruling, which is then binding law.6 Decisions made by any court are based on various degrees of stringency, called standards of proof (Table 27).

Continue to: For Ms. T's case...
For Ms. T’s case, civil commitment and involuntary medication hearings are held in probate court, which is a civil (not criminal) court. In addition to overseeing civil commitment and involuntary medications, probate courts adjudicate will and estate contests, conservatorship, and guardianship. Conservatorship hearings deal with financial issues, and guardianship cases encompass personal and health-related needs. Regardless of the court, an individual is guaranteed due process under the 5th Amendment (federal) and 14th Amendment (state).
Individuals are presumed competent to make their own decisions, but a court may call this into question. Competencies are specific to a variety of areas, such as criminal proceedings, medical decision making, writing a will (testimonial capacity), etc. Because each field applies its own standard of competence, an individual may be competent in one area but incompetent in another. Competence in medical decision making varies by state but generally consists of being able to communicate a choice, understand relevant information, appreciate one’s illness and its likely consequences, and rationally manipulate information.8
Box
Administering medications despite a patient’s objection differs from situations in which medications are provided during a psychiatric emergency. In an emergency, courts do not have time to weigh in. Instead, emergency medications (most often given as IM injections) are administered based on the physician’s clinical judgment. The criteria for psychiatric emergencies are delineated at the state level, but typically are defined as when a person with a mental illness creates an imminent risk of harm to self or others. Alternative approaches to resolving the emergency may include verbal de-escalation, quiet time in a room devoid of stimuli, locked seclusion, or physical restraints. These measures are often exhausted before emergency medications are administered.
Source: Reference 9
It is important to note that the legal process required before administering involuntary medications is distinct from situations in which medication needs to be provided during a psychiatric emergency. The Box9 outlines the difference between these 2 scenarios.

4 Legal models
There are several legal models used to determine when a patient can be administered psychiatric medications over objection. Table 310,11 summarizes these models.
Rights-driven (Rogers) model. If Ms. T was involuntarily hospitalized in Massachusetts or another state that adopted the rights-driven model, she would retain the right to refuse treatment. These states require an external judicial review, and court approval is necessary before imposing any therapy. This model was established in Rogers v Commissioner,12 where 7 patients at the Boston State Hospital filed a lawsuit regarding their right to refuse medications. The Massachusetts Supreme Judicial Court ruled that, despite being involuntarily committed, a patient is considered competent to refuse treatment until found specifically incompetent to do so by the court. If a patient is found incompetent, the judge, using a full adversarial hearing, decides what the incompetent patient would have wanted if he/she were competent. The judge reaches a conclusion based on the substituted judgment model (Table 410). In Rogers v Commissioner,12 the court ruled that the right to decision making is not lost after becoming a patient at a mental health facility. The right is lost only if the patient is found incompetent by the judge. Thus, every individual has the right to “manage his own person” and “take care of himself.”
Continue to: An update to the rights-driven (Rogers) model
An update to the rights-driven (Rogers) model. Other states, such as Ohio, have adopted the Rogers model and addressed issues that arose subsequent to the aforementioned case. In Steele v Hamilton County,13 Jeffrey Steele was admitted and later civilly committed to the hospital. After 2 months, an involuntary medication hearing was completed in which 3 psychiatrists concluded that, although Mr. Steele was not a danger to himself or others while in the hospital, he would ultimately benefit from medications.
The probate court acknowledged that Mr. Steele lacked capacity and required hospitalization. However, because he was not imminently dangerous, medication should not be used involuntarily. After a series of appeals, the Ohio Supreme Court ruled that a court may authorize the administration of an antipsychotic medication against a patient’s wishes without a finding of dangerousness when clear and convincing evidence exists that:
- the patient lacks the capacity to give or withhold informed consent regarding treatment
- the proposed medication is in the patient’s best interest
- no less intrusive treatment will be as effective in treating the mental illness.
This ruling set a precedent that dangerousness is not a requirement for involuntary medications.
Treatment-driven (Rennie) model. As in the rights-driven model, in the treatment-driven model, Ms. T would retain the constitutional right to refuse treatment. However, the models differ in the amount of procedural due process required. The treatment-driven model derives from Rennie v Klein,14 in which John Rennie, a patient at Ancora State Psychiatric Hospital in New Jersey, filed a suit regarding the right of involuntarily committed patients to refuse antipsychotic medications. The Third Circuit Court of Appeals ruled that, if professional judgment deems a patient to be a danger to himself or others, then antipsychotics may be administered over individual objection. This professional judgment is typically based on the opinion of the treating physician, along with a second physician or panel.
Utah model. This model is based on A.E. and R.R. v Mitchell,15 in which the Utah District Court ruled that a civilly committed patient has no right to refuse treatment. This Utah model was created after state legislature determined that, in order to civilly commit a patient, hospitalization must be the least restrictive alternative and the patient is incompetent to consent to treatment. Unlike the 2 previous models, competency to refuse medications is not separated from a previous finding of civil commitment, but rather, they occur simultaneously.
Continue to: Rights in unique situations
Rights in unique situations
Correctional settings. If Ms. T was an inmate, would her right to refuse psychiatric medication change? This was addressed in the case of Washington v Harper.16 Walter Harper, serving time for a robbery conviction, filed a claim that his civil rights were being violated when he received involuntary medications based on the decision of a 3-person panel consisting of a psychiatrist, psychologist, and prison official. The US Supreme Court ruled that this process provided sufficient due process to mandate providing psychotropic medications against a patient’s will. This reduction in required procedures is related to the unique nature of the correctional environment and an increased need to maintain safety. This need was felt to outweigh an individual’s right to refuse medication.
Incompetent to stand trial. In Sell v U.S.,17 Charles Sell, a dentist, was charged with fraud and attempted murder. He underwent a competency evaluation and was found incompetent to stand trial because of delusional thinking. Mr. Sell was hospitalized for restorability but refused medications. The hospital held an administrative hearing to proceed with involuntary antipsychotic medications; however, Mr. Sell filed an order with the court to prevent this. Eventually, the US Supreme Court ruled that non-dangerous, incompetent defendants may be involuntarily medicated even if they do not pose a risk to self or others on the basis that it furthers the state’s interest in bringing to trial those charged with serious crimes. However, the following conditions must be met before involuntary medication can be administered:
- an important government issue must be at stake (determined case-by-case)
- a substantial probability must exist that the medication will enable the defendant to become competent without significant adverse effects
- the medication must be medically appropriate and necessary to restore competency, with no less restrictive alternative available.
This case suggests that, before one attempts to forcibly medicate a defendant for the purpose of competency restoration, one should exhaust the same judicial remedies one uses for civil patients first.
Court-appointed guardianship
In the case of Ms. T, what if her father requested to become her guardian? This question was explored in the matter of Guardianship of Richard Roe III.18 Mr. Roe was admitted to the Northampton State Hospital in Massachusetts, where he refused antipsychotic medications. Prior to his release, his father asked to be his guardian. The probate court obliged the request. However, Mr. Roe’s lawyer and guardian ad litem (a neutral temporary guardian often appointed when legal issues are pending) challenged the ruling, arguing the probate court cannot empower the guardian to consent to involuntary medication administration. On appeal, the court ruled:
- the guardianship was justified
- the standard of proof for establishment of a guardianship is preponderance of the evidence (Table 27)
- the guardian must seek from a court a “substituted judgment” to authorize forcible administration of antipsychotic medication.
The decision to establish the court as the final decision maker is based on the view that a patient’s relatives may be biased. Courts should take an objective approach that considers
- patient preference stated during periods of competency
- medication adverse effects
- consequences if treatment is refused
- prognosis with treatment
- religious beliefs
- impact on the patient’s family.
Continue to: This case set the stage for...
This case set the stage for later decisions that placed antipsychotic medications in the same category as electroconvulsive therapy and psychosurgery. This could mean a guardian would need specialized authorization to request antipsychotic treatment but could consent to an appendectomy without legal issue.
Fortunately, now most jurisdictions have remedied this cumbersome solution by requiring a higher standard of proof, clear and convincing evidence (Table 27), to establish guardianship but allowing the guardian more latitude to make decisions for their wards (such as those involving hospital admission or medications) without further court involvement.
Involuntary medical treatment
In order for a patient to consent for medical treatment, he/she must have the capacity to do so (Table 59). How do the courts handle the patient’s right to refuse medical treatment? This was addressed in the case of Georgetown College v Jones.19 Mrs. Jones, a 25-year-old Jehovah’s Witness and mother of a 7-month-old baby, suffered a ruptured ulcer and lost a life-threatening amount of blood. Due to her religious beliefs, Mrs. Jones refused a blood transfusion. The hospital quickly appealed to the court, who ruled the woman was help-seeking by going to the hospital, did not want to die, was in distress, and lacked capacity to make medical decisions. Acting in a parens patriae manner (when the government steps in to make decisions for its citizens who cannot), the court ordered the hospital to administer blood transfusions.
Proxy decision maker. When the situation is less emergent, a proxy decision maker can be appointed by the court. This was addressed in the case of Superintendent of Belchertown v Saikewicz.20 Mr. Saikewicz, a 67-year-old man with intellectual disability, was diagnosed with cancer and given weeks to months to live without treatment. However, treatment was only 50% effective and could potentially cause severe adverse effects. A guardian ad litem was appointed and recommended nontreatment, which the court upheld. The court ruled that the right to accept or reject medical treatment applies to both incompetent and competent persons. With incompetent persons, a “substituted judgment” analysis is used over the “best interest of the patient” doctrine.20 This falls in line with the Guardianship of Richard Roe III ruling,18 in which the court’s substituted judgment standard is enacted in an effort to respect patient autonomy.
Right to die. When does a patient have the right to die and what is the standard of proof? The US Supreme Court case Cruzan v Director21 addressed this. Nancy Cruzan was involved in a car crash, which left her in a persistent vegetative state with no significant cognitive function. She remained this way for 6 years before her parents sought to terminate life support. The hospital refused. The Missouri Supreme Court ruled that a standard of clear and convincing evidence (Table 27) is required to withdraw treatment, and in a 5-to-4 decision, the US Supreme Court upheld Missouri’s decision. This set the national standard for withdrawal of life-sustaining treatment. The moderate standard of proof is based on the court’s ruling that the decision to terminate life is a particularly important one.
Continue to: CASE
CASE CONTINUED
After having been civilly committed to your inpatient psychiatric facility, Ms. T’s paranoia and disorganized behavior persist. She continues to refuse medications.
There are 3 options: respect her decision, negotiate with her, or attempt to force medications through due process.11 In negotiating a compromise, it is best to understand the barriers to treatment. A patient may refuse medications due to poor insight into his/her illness, medication adverse effects, a preference for an alternative treatment, delusional concerns over contamination and/or poisoning, interpersonal conflicts with the treatment staff, a preference for symptoms (eg, mania) over wellness, medication ineffectiveness, length of treatment course, or stigma.22,23 However, a patient’s unwillingness to compromise creates the dilemma of autonomy vs treatment.
For Ms. T, the treatment team felt initiating involuntary medication was the best option for her quality of life and safety. Because she resides in Ohio, a Rogers-like model was applied. The probate court was petitioned and found her incompetent to make medical decisions. The court accepted the physician’s recommendation of treatment with antipsychotic medications. If this scenario took place in New Jersey, a Rennie model would apply, requiring due process through the second opinion of another physician. Lastly, if Ms. T lived in Utah, she would have been unable to refuse medications once civilly committed.
Pros and cons of each model
Over the years, various concerns about each of these models have been raised. Given the slow-moving wheels of justice, one concern was that perhaps patients would be left “rotting with their rights on,” or lingering in a psychotic state out of respect for their civil liberties.19 While court hearings do not always happen quickly, more often than not, a judge will agree with the psychiatrist seeking treatment because the judge likely has little experience with mental illness and will defer to the physician’s expertise. This means the Rogers model may be more likely to produce the desired outcome, just more slowly. With respect to the Rennie model, although it is often more expeditious, the second opinion of an independent psychiatrist may contradict that of the original physician because the consultant will rely on his/her own expertise. Finally, some were concerned that psychiatrists would view the Utah model as carte blanche to start whatever medications they wanted with no respect for patient preference. Based on our clinical experience, none of these concerns have come to fruition over time, and patients safely receive medications over objection in hospitals every day.
Consider why the patient refuses medication
Regardless of which involuntary medication model is employed, it is important to consider the underlying cause for medication refusal, because it may affect future compliance. If the refusal is the result of a religious belief, history of adverse effects, or other rational motive, then it may be reasonable to respect the patient’s autonomy.24 However, if the refusal is secondary to symptoms of mental illness, it is appropriate to move forward with an involuntary medication hearing and treat the underlying condition.
Continue to: In the case of Ms. T...
In the case of Ms. T, she appeared to be refusing medications because of her psychotic symptoms, which could be effectively treated with antipsychotic medications. Therefore, Ms. T’s current lack of capacity is hopefully a transient phenomenon that can be ameliorated by initiating medication. Typically, antipsychotic medications begin to reduce psychotic symptoms within the first week, with further improvement over time.25 The value of the inpatient psychiatric setting is that it allows for daily monitoring of a patient’s response to treatment. As capacity is regained, patient autonomy over medical decisions is reinstated.
Bottom Line
The legal processes required to administer medications over a patient’s objection are state-specific, and multiple models are used. In general, a patient’s right to refuse treatment can be overruled by obtaining adjudication through the courts (Rogers model) or the opinion of a second physician (Rennie model). In order to ensure the best practice and patient care, research the legal procedure specific to your jurisdiction, consult your clinic/hospital attorney, and/or contact your state’s mental health board for further clarification.
Related Resources
- Miller D. Is forced treatment in our outpatients’ best interests? Clinical Psychiatry News. https://www.mdedge.com/psychiatry/article/80277/forced-treatment-our-outpatients-best-interests.
- Miller D, Hanson A. Committed: The battle over involuntary psychiatric care. Baltimore, MD: Johns Hopkins University Press; 2016.
1. Laffey P. Psychiatric therapy in Georgian Britain. Psychol Med. 2003;33(7):1285-1297.
2. Porter R. Madness: a brief history. New York, NY: Oxford Press; 2002.
3. Stetka B, Watson J. Odd and outlandish psychiatric treatments throughout history. Medscape Psychiatry. https://www.medscape.com/features/slideshow/odd-psychiatric-treatments. Published April 13, 2016. Accessed February 26, 2020.
4. Rouse v Cameron, 373, F2d 451 (DC Cir 1966).
5. Wyatt v Stickney, 325 F Supp 781 (MD Ala 1971).
6. Administrative Office of the US Courts. Comparing federal and state Courts. United States Courts. https://www.uscourts.gov/about-federal-courts/court-role-and-structure/comparing-federal-state-courts. Accessed February 26, 2020.
7. Drogin E, Williams C. Introduction to the Legal System. In: Gold L, Frierson R, eds. Textbook of forensic psychiatry, 3rd ed. Arlington, VA: American Psychiatric Association Publishing; 2018:80-83.
8. Appelbaum P, Grisso T. Assessing patients’ capacities to consent to treatment. N Engl J Med. 1988;319(25):1635-1638.
9. Kambam P. Informed consent and competence. In: Rosnar R, Scott C, eds. Principles and practice of forensic psychiatry, 3rd ed. Boca Raton, FL: CRC press; 2017:115-121.
10. Wall B, Anfang S. Legal regulation of psychiatric treatment. In: Gold L, Frierson R, eds. Textbook of forensic psychiatry, 3rd ed. Arlington, VA: American Psychiatric Association Publishing; 2018:306-333.
11. Pinals D, Nesbit A, Hoge S. Treatment refusal in psychiatric practice. In: Rosnar R, Scott C, eds. Principles and practice of forensic psychiatry, 3rd ed. Boca Raton, FL: CRC press; 2017:155-163.
12. Rogers v Commissioner, 390 489 (Mass 1983).
13. Steele v Hamilton County, 90 Ohio St3d 176 (Ohio 2000).
14. Rennie v Klein, 462 F Supp 1131 (D NJ 1978).
15. AE and RR v Mitchell, 724 F.2d 864 (10th Cir 1983).
16. Washington v Harper, 494 US 210 (1990).
17. Sell v US, 539 US 166 (2003).
18. Guardianship of Richard Roe III, 383 415, 435 (Mass 1981).
19. Georgetown College v Jones, 331 F2d 1010 (DC Cir 1964).
20. Superintendent of Belchertown v Saikewicz, 370 NE 2d 417 (1977).
21. Cruzan v Director, 497 US 261 (1990).
22. Owiti J, Bowers L. A literature review: refusal of psychotropic medication in acute inpatient psychiatric care. J Psychiatr Ment Health Nurs. 2011;18(7):637-647.
23. Appelbaum P, Gutheil T. “Rotting with their rights on”: constitutional theory and clinical reality in drug refusal by psychiatric patients. Bull Am Acad Psychiatry Law. 1979;7(3):306-315.
24. Adelugba OO, Mela M, Haq IU. Psychotropic medication refusal: reasons and patients’ perception at a secure forensic psychiatric treatment centre. J Forensic Sci Med. 2016;2(1):12-17.
25. Agid O, Kapur S, Arenovich T, et al. Delayed-onset hypothesis of antipsychotic action: a hypothesis tested and rejected. Arch Gen Psychiatry. 2003;60(12):1228.
1. Laffey P. Psychiatric therapy in Georgian Britain. Psychol Med. 2003;33(7):1285-1297.
2. Porter R. Madness: a brief history. New York, NY: Oxford Press; 2002.
3. Stetka B, Watson J. Odd and outlandish psychiatric treatments throughout history. Medscape Psychiatry. https://www.medscape.com/features/slideshow/odd-psychiatric-treatments. Published April 13, 2016. Accessed February 26, 2020.
4. Rouse v Cameron, 373, F2d 451 (DC Cir 1966).
5. Wyatt v Stickney, 325 F Supp 781 (MD Ala 1971).
6. Administrative Office of the US Courts. Comparing federal and state Courts. United States Courts. https://www.uscourts.gov/about-federal-courts/court-role-and-structure/comparing-federal-state-courts. Accessed February 26, 2020.
7. Drogin E, Williams C. Introduction to the Legal System. In: Gold L, Frierson R, eds. Textbook of forensic psychiatry, 3rd ed. Arlington, VA: American Psychiatric Association Publishing; 2018:80-83.
8. Appelbaum P, Grisso T. Assessing patients’ capacities to consent to treatment. N Engl J Med. 1988;319(25):1635-1638.
9. Kambam P. Informed consent and competence. In: Rosnar R, Scott C, eds. Principles and practice of forensic psychiatry, 3rd ed. Boca Raton, FL: CRC press; 2017:115-121.
10. Wall B, Anfang S. Legal regulation of psychiatric treatment. In: Gold L, Frierson R, eds. Textbook of forensic psychiatry, 3rd ed. Arlington, VA: American Psychiatric Association Publishing; 2018:306-333.
11. Pinals D, Nesbit A, Hoge S. Treatment refusal in psychiatric practice. In: Rosnar R, Scott C, eds. Principles and practice of forensic psychiatry, 3rd ed. Boca Raton, FL: CRC press; 2017:155-163.
12. Rogers v Commissioner, 390 489 (Mass 1983).
13. Steele v Hamilton County, 90 Ohio St3d 176 (Ohio 2000).
14. Rennie v Klein, 462 F Supp 1131 (D NJ 1978).
15. AE and RR v Mitchell, 724 F.2d 864 (10th Cir 1983).
16. Washington v Harper, 494 US 210 (1990).
17. Sell v US, 539 US 166 (2003).
18. Guardianship of Richard Roe III, 383 415, 435 (Mass 1981).
19. Georgetown College v Jones, 331 F2d 1010 (DC Cir 1964).
20. Superintendent of Belchertown v Saikewicz, 370 NE 2d 417 (1977).
21. Cruzan v Director, 497 US 261 (1990).
22. Owiti J, Bowers L. A literature review: refusal of psychotropic medication in acute inpatient psychiatric care. J Psychiatr Ment Health Nurs. 2011;18(7):637-647.
23. Appelbaum P, Gutheil T. “Rotting with their rights on”: constitutional theory and clinical reality in drug refusal by psychiatric patients. Bull Am Acad Psychiatry Law. 1979;7(3):306-315.
24. Adelugba OO, Mela M, Haq IU. Psychotropic medication refusal: reasons and patients’ perception at a secure forensic psychiatric treatment centre. J Forensic Sci Med. 2016;2(1):12-17.
25. Agid O, Kapur S, Arenovich T, et al. Delayed-onset hypothesis of antipsychotic action: a hypothesis tested and rejected. Arch Gen Psychiatry. 2003;60(12):1228.
Psychiatric patients and pandemics
What can psychiatric clinicians do to keep their patients healthy in this coronavirus time?
In the 3 days between starting this column and finishing it, the world has gone into a tailspin. Perhaps what I write is no longer relevant. But hopefully it is.
I have no right or wrong answers here but thoughts about factors to consider.
- On inpatient psychiatry wards, the emphasis is on communal living. On our ward, bedrooms and bathrooms are shared. Patients eat together. There are numerous group therapies.
- We have decided to restrict visitors out of the concern that one may infect a ward of patients and staff. We are hoping to do video visitation, but that may take a while to implement.
- An open question is how we are going to provide our involuntary patients with access to the public defense attorneys. Public defenders still have the ability to come onto the inpatient ward, but we will start screening them first.
- In terms of sanitation, wall sanitizers are forbidden, since sanitizers may be drank or made into a firebomb. So we are incessantly wiping down the shared phones and game board pieces.
- Looking at the outpatient arena, we have moved our chairs around, so that there are 3 feet between chairs. We have opened up another waiting room to provide more distance.
- We are trying to decide whether to cancel groups. We did cancel our senior group, and I think I will cancel the rest of them shortly.
- We are seriously looking at telepsychiatry.
- Schools are closed. Many of my clinicians have young children, so they may be out. We are expecting many patients to cancel and will see how that plays out. Others of us have elderly parents. My mother’s assisted-living facility is on lockdown. So, having been locked out after a visit, she is with me tonight.
- Psychiatrists are expected to keep up their relative value unit count. Can they meet their targets? Probably not. Will it matter?
- And what about all our homeless patients, who cannot disinfect their tents or shelters?
- Conferences no longer seem so important. I am less worried about coverage for the American Psychiatric Association meeting, since the 2020 conference has been canceled.
On the rosy side, maybe this will be a wake-up call about climate change. So we live in interesting times.
Take care of your patients and each other.
Dr. Ritchie is chair of psychiatry at Medstar Washington Hospital Center and professor of psychiatry at Georgetown University, Washington. She has no disclosures.
What can psychiatric clinicians do to keep their patients healthy in this coronavirus time?
In the 3 days between starting this column and finishing it, the world has gone into a tailspin. Perhaps what I write is no longer relevant. But hopefully it is.
I have no right or wrong answers here but thoughts about factors to consider.
- On inpatient psychiatry wards, the emphasis is on communal living. On our ward, bedrooms and bathrooms are shared. Patients eat together. There are numerous group therapies.
- We have decided to restrict visitors out of the concern that one may infect a ward of patients and staff. We are hoping to do video visitation, but that may take a while to implement.
- An open question is how we are going to provide our involuntary patients with access to the public defense attorneys. Public defenders still have the ability to come onto the inpatient ward, but we will start screening them first.
- In terms of sanitation, wall sanitizers are forbidden, since sanitizers may be drank or made into a firebomb. So we are incessantly wiping down the shared phones and game board pieces.
- Looking at the outpatient arena, we have moved our chairs around, so that there are 3 feet between chairs. We have opened up another waiting room to provide more distance.
- We are trying to decide whether to cancel groups. We did cancel our senior group, and I think I will cancel the rest of them shortly.
- We are seriously looking at telepsychiatry.
- Schools are closed. Many of my clinicians have young children, so they may be out. We are expecting many patients to cancel and will see how that plays out. Others of us have elderly parents. My mother’s assisted-living facility is on lockdown. So, having been locked out after a visit, she is with me tonight.
- Psychiatrists are expected to keep up their relative value unit count. Can they meet their targets? Probably not. Will it matter?
- And what about all our homeless patients, who cannot disinfect their tents or shelters?
- Conferences no longer seem so important. I am less worried about coverage for the American Psychiatric Association meeting, since the 2020 conference has been canceled.
On the rosy side, maybe this will be a wake-up call about climate change. So we live in interesting times.
Take care of your patients and each other.
Dr. Ritchie is chair of psychiatry at Medstar Washington Hospital Center and professor of psychiatry at Georgetown University, Washington. She has no disclosures.
What can psychiatric clinicians do to keep their patients healthy in this coronavirus time?
In the 3 days between starting this column and finishing it, the world has gone into a tailspin. Perhaps what I write is no longer relevant. But hopefully it is.
I have no right or wrong answers here but thoughts about factors to consider.
- On inpatient psychiatry wards, the emphasis is on communal living. On our ward, bedrooms and bathrooms are shared. Patients eat together. There are numerous group therapies.
- We have decided to restrict visitors out of the concern that one may infect a ward of patients and staff. We are hoping to do video visitation, but that may take a while to implement.
- An open question is how we are going to provide our involuntary patients with access to the public defense attorneys. Public defenders still have the ability to come onto the inpatient ward, but we will start screening them first.
- In terms of sanitation, wall sanitizers are forbidden, since sanitizers may be drank or made into a firebomb. So we are incessantly wiping down the shared phones and game board pieces.
- Looking at the outpatient arena, we have moved our chairs around, so that there are 3 feet between chairs. We have opened up another waiting room to provide more distance.
- We are trying to decide whether to cancel groups. We did cancel our senior group, and I think I will cancel the rest of them shortly.
- We are seriously looking at telepsychiatry.
- Schools are closed. Many of my clinicians have young children, so they may be out. We are expecting many patients to cancel and will see how that plays out. Others of us have elderly parents. My mother’s assisted-living facility is on lockdown. So, having been locked out after a visit, she is with me tonight.
- Psychiatrists are expected to keep up their relative value unit count. Can they meet their targets? Probably not. Will it matter?
- And what about all our homeless patients, who cannot disinfect their tents or shelters?
- Conferences no longer seem so important. I am less worried about coverage for the American Psychiatric Association meeting, since the 2020 conference has been canceled.
On the rosy side, maybe this will be a wake-up call about climate change. So we live in interesting times.
Take care of your patients and each other.
Dr. Ritchie is chair of psychiatry at Medstar Washington Hospital Center and professor of psychiatry at Georgetown University, Washington. She has no disclosures.