User login
Therapeutic drug monitoring of antipsychotics
Mr. Q, age 36, has a history of schizophrenia. He is brought to the hospital due to persistent auditory hallucinations and paranoid delusions. His history documents a trial and failure of risperidone, 4 mg twice daily, and aripiprazole, 20 mg/d. Based on this, the treatment team initiates haloperidol, 5 mg twice daily. Because he experiences persistent auditory hallucinations and paranoid delusions, Mr. Q is titrated to increasing doses of haloperidol over 2 weeks during the course of the hospitalization. Once Mr. Q is receiving a total haloperidol dose of 30 mg/d, the team decides to obtain a serum haloperidol level due to his persistent psychotic symptoms and the development of drug-induced parkinsonism. His serum haloperidol level is 24 ng/mL, which is within the expected range for his dose, but above the therapeutic window for efficacy. The team decides that the severity of Mr. Q’s illness and documented treatment resistance (failing at least 2 adequate trials of antipsychotics) warrant a trial of clozapine.
Despite a long history of therapeutic drug monitoring (TDM) within psychiatry, routine monitoring of antipsychotic serum levels has not been unanimously adopted as standard practice. Clinical practice typically results in mostly a subjective assessment of the safety and efficacy of antipsychotics. This practice is in contrast to agents such as valproic acid and lithium, which are routinely monitored for safety and efficacy using both subjective and laboratory measures. Clinicians may adhere to these monitoring practices for lithium and valproic acid because of these agents’ narrow “therapeutic window” between toxicity and efficacy. However, antipsychotics can be viewed in a similar fashion.
To help conceptualize the therapeutic window for antipsychotics, it is important to understand that in most cases, the pharmacologic target for antipsychotics is dopamine (D2)receptor antagonism between 60% to 80%.1 Total drug exposure would thus determine a patient’s likelihood of minimizing positive symptoms, or exposure to adverse effects related to total dopamine antagonism. Serum drug concentrations are a better metric than total daily dose for determining drug exposure and achieving the pharmacologic target.2 Evaluating serum antipsychotic levels also is a better method of determining true treatment failure than relying on the clinical judgment of the treating psychiatrist.3
Pros and cons of TDM
Benefits of using TDM for patients being treated with antipsychotics include4:
- ensuring adherence
- quantitatively adjusting dosages for medication interactions or genetic variations
- ensuring an adequate trial of a medication before considering it a treatment failure.
Potential drawbacks to TDM include:
- Delayed results. Access to expeditious testing may not be possible in certain laboratories, and this may require send-out testing, which could result in a delay in obtaining results. Continued advocacy and research on the value of TDM in antipsychotics may improve access to these resources in the future. Nonetheless, obtaining antipsychotic serum levels will still give clinicians insight into the antipsychotic exposure at a given dose. Further, obtaining antipsychotic serum levels may strengthen decisions about treatment resistance and the assessment of interactions, adherence, or the likelihood of adverse effects.
- Lack of guidance. Unfortunately, there is no established guidance outlining what to do once antipsychotic serum levels are obtained. The correlation of serum levels of commonly used second-generation antipsychotics with clinical efficacy needs to be more closely investigated; however, certain agents do have more data associated with appropriate ranges for efficacy/toxicity. While researchers should continue to study the precise relationship between antipsychotic serum levels and effect, clinicians still have resources available to help determine what the expected serum value for a given patient may be. Knowing an expected serum level may help clinicians determine whether there is an unknown interaction or genetic variation that is causing lower- or higher-than-expected levels. This may also help determine whether a patient is adhering to their medication regimen.
Growing evidence for TDM
In recent years, evidence supporting the use of TDM in patients receiving antipsychotics has been increasing, and recommendations from consensus groups have been strengthened. One of the most comprehensive assessments of these practices was published by the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie (AGNP), a German-based psychopharmacology group consisting of researchers and clinicians. In 2018, the AGNP published consensus guidelines for TDM in various neuropsychiatric medication classes and recommendations for collecting, interpreting, and decision-making elements of the process.5 The Table5 lists the AGNP-recommended therapeutic serum range for several commonly used second-generation antipsychotics.
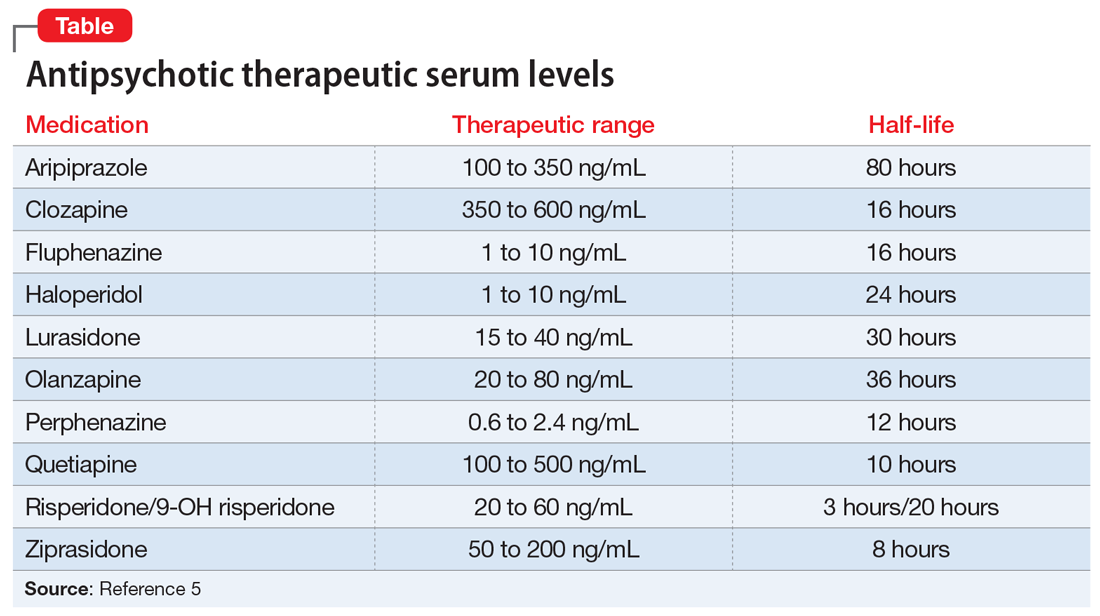
Researchers should be encouraged to contribute to the body of knowledge on the correlation of clinical response to serum level. However, there is compelling evidence for the use of TDM across many first- and second-generation antipsychotics. Of the most common, haloperidol and clozapine have evidence of a therapeutic range that is better correlated with serum level than daily dose. Specifically, haloperidol appears to lose benefit with dose increases beyond serum levels of approximately 10 ng/mL.6
Continue to: Clozapine levels may be...
Clozapine levels may be reported by measuring the metabolite norclozapine, which is not correlated with efficacy, or as a total level (combination of clozapine and norclozapine). While norclozapine is not associated with efficacy, the ratio of clozapine to norclozapine may indicate adherence to the medication, or any enzymatic modulation (genetic or drug–drug interaction) that may increase or decrease total exposure. A ratio of 1.5 to 2.0 (clozapine to norclozapine) is optimal; a ratio <0.5 may indicate nonadherence; and a ratio >2.0 may indicate inhibited drug clearance. A 12-hour serum clozapine level of ≥350 ng/mL is more likely to predict treatment response.7
CASE CONTINUED
Mr. Q is carefully tapered from haloperidol while initiating clozapine at 25 mg/d. As he is titrated on clozapine, Mr. Q’s serum levels are periodically checked and compared with expected levels and levels associated with efficacy. Eventually, Mr. Q is titrated to a clozapine dose of 400 mg/d at bedtime.
While receiving clozapine for 4 weeks, Mr. Q’s psychotic symptoms resolve, and he is scheduled for follow-up in the outpatient clozapine clinic.
Related Resources
- De Leon J. A critical commentary on the 2017 AGNP consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology. Pharmacopsychiatry. 2018; 51(1-02):63-68.
- Meyer JM. Is monitoring of plasma antipsychotic levels useful? Current Psychiatry. 2015;14(11):16,19-20.
Drug Brand Names
Aripiprazole • Abilify
Clozapine • Clozaril
Fluphenazine • Prolixin
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Olanzapine • Zyprexa
Perphenazine • Trilafon
Quetiapine • Seroquel
Risperidone • Risperdal
Valproic acid • Depakene, Depakote
Ziprasidone • Geodon
1. Stahl SM. Stahl’s essential psychopharmacology, neuroscientific basis and practical applications, 4th ed. Cambridge, United Kingdom: Cambridge University Press; 2013:129-236.
2. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181.
3. McCutcheon R, Beck K, D’Ambrosio E, et al. Antipsychotic plasma levels in the assessment of poor treatment response in schizophrenia. Acta Psychiatr Scand. 2018;137(1):39-46.
4. Horvitz-Lennon M, Mattke S, Predmore Z, et al. The role of antipsychotic plasma levels in the treatment of schizophrenia. Am J Psychiatry. 2017;174(5):421-426.
5. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62.
6. Van Putten T, Marder SR, Mintz J, et al. Haloperidol plasma levels and clinical response: a therapeutic window relationship. Am J Psychiatry. 1992;149(4):500-505.
7. Couchman L, Morgan PE, Spencer EP, et al. Plasma clozapine, norclozapine, and the clozapine:norclozapine ratio in relation to prescribed dose and other factors: data from a therapeutic drug monitoring service, 1993-2007. Ther Drug Monit. 2010;32(4):438-447.
Mr. Q, age 36, has a history of schizophrenia. He is brought to the hospital due to persistent auditory hallucinations and paranoid delusions. His history documents a trial and failure of risperidone, 4 mg twice daily, and aripiprazole, 20 mg/d. Based on this, the treatment team initiates haloperidol, 5 mg twice daily. Because he experiences persistent auditory hallucinations and paranoid delusions, Mr. Q is titrated to increasing doses of haloperidol over 2 weeks during the course of the hospitalization. Once Mr. Q is receiving a total haloperidol dose of 30 mg/d, the team decides to obtain a serum haloperidol level due to his persistent psychotic symptoms and the development of drug-induced parkinsonism. His serum haloperidol level is 24 ng/mL, which is within the expected range for his dose, but above the therapeutic window for efficacy. The team decides that the severity of Mr. Q’s illness and documented treatment resistance (failing at least 2 adequate trials of antipsychotics) warrant a trial of clozapine.
Despite a long history of therapeutic drug monitoring (TDM) within psychiatry, routine monitoring of antipsychotic serum levels has not been unanimously adopted as standard practice. Clinical practice typically results in mostly a subjective assessment of the safety and efficacy of antipsychotics. This practice is in contrast to agents such as valproic acid and lithium, which are routinely monitored for safety and efficacy using both subjective and laboratory measures. Clinicians may adhere to these monitoring practices for lithium and valproic acid because of these agents’ narrow “therapeutic window” between toxicity and efficacy. However, antipsychotics can be viewed in a similar fashion.
To help conceptualize the therapeutic window for antipsychotics, it is important to understand that in most cases, the pharmacologic target for antipsychotics is dopamine (D2)receptor antagonism between 60% to 80%.1 Total drug exposure would thus determine a patient’s likelihood of minimizing positive symptoms, or exposure to adverse effects related to total dopamine antagonism. Serum drug concentrations are a better metric than total daily dose for determining drug exposure and achieving the pharmacologic target.2 Evaluating serum antipsychotic levels also is a better method of determining true treatment failure than relying on the clinical judgment of the treating psychiatrist.3
Pros and cons of TDM
Benefits of using TDM for patients being treated with antipsychotics include4:
- ensuring adherence
- quantitatively adjusting dosages for medication interactions or genetic variations
- ensuring an adequate trial of a medication before considering it a treatment failure.
Potential drawbacks to TDM include:
- Delayed results. Access to expeditious testing may not be possible in certain laboratories, and this may require send-out testing, which could result in a delay in obtaining results. Continued advocacy and research on the value of TDM in antipsychotics may improve access to these resources in the future. Nonetheless, obtaining antipsychotic serum levels will still give clinicians insight into the antipsychotic exposure at a given dose. Further, obtaining antipsychotic serum levels may strengthen decisions about treatment resistance and the assessment of interactions, adherence, or the likelihood of adverse effects.
- Lack of guidance. Unfortunately, there is no established guidance outlining what to do once antipsychotic serum levels are obtained. The correlation of serum levels of commonly used second-generation antipsychotics with clinical efficacy needs to be more closely investigated; however, certain agents do have more data associated with appropriate ranges for efficacy/toxicity. While researchers should continue to study the precise relationship between antipsychotic serum levels and effect, clinicians still have resources available to help determine what the expected serum value for a given patient may be. Knowing an expected serum level may help clinicians determine whether there is an unknown interaction or genetic variation that is causing lower- or higher-than-expected levels. This may also help determine whether a patient is adhering to their medication regimen.
Growing evidence for TDM
In recent years, evidence supporting the use of TDM in patients receiving antipsychotics has been increasing, and recommendations from consensus groups have been strengthened. One of the most comprehensive assessments of these practices was published by the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie (AGNP), a German-based psychopharmacology group consisting of researchers and clinicians. In 2018, the AGNP published consensus guidelines for TDM in various neuropsychiatric medication classes and recommendations for collecting, interpreting, and decision-making elements of the process.5 The Table5 lists the AGNP-recommended therapeutic serum range for several commonly used second-generation antipsychotics.

Researchers should be encouraged to contribute to the body of knowledge on the correlation of clinical response to serum level. However, there is compelling evidence for the use of TDM across many first- and second-generation antipsychotics. Of the most common, haloperidol and clozapine have evidence of a therapeutic range that is better correlated with serum level than daily dose. Specifically, haloperidol appears to lose benefit with dose increases beyond serum levels of approximately 10 ng/mL.6
Continue to: Clozapine levels may be...
Clozapine levels may be reported by measuring the metabolite norclozapine, which is not correlated with efficacy, or as a total level (combination of clozapine and norclozapine). While norclozapine is not associated with efficacy, the ratio of clozapine to norclozapine may indicate adherence to the medication, or any enzymatic modulation (genetic or drug–drug interaction) that may increase or decrease total exposure. A ratio of 1.5 to 2.0 (clozapine to norclozapine) is optimal; a ratio <0.5 may indicate nonadherence; and a ratio >2.0 may indicate inhibited drug clearance. A 12-hour serum clozapine level of ≥350 ng/mL is more likely to predict treatment response.7
CASE CONTINUED
Mr. Q is carefully tapered from haloperidol while initiating clozapine at 25 mg/d. As he is titrated on clozapine, Mr. Q’s serum levels are periodically checked and compared with expected levels and levels associated with efficacy. Eventually, Mr. Q is titrated to a clozapine dose of 400 mg/d at bedtime.
While receiving clozapine for 4 weeks, Mr. Q’s psychotic symptoms resolve, and he is scheduled for follow-up in the outpatient clozapine clinic.
Related Resources
- De Leon J. A critical commentary on the 2017 AGNP consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology. Pharmacopsychiatry. 2018; 51(1-02):63-68.
- Meyer JM. Is monitoring of plasma antipsychotic levels useful? Current Psychiatry. 2015;14(11):16,19-20.
Drug Brand Names
Aripiprazole • Abilify
Clozapine • Clozaril
Fluphenazine • Prolixin
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Olanzapine • Zyprexa
Perphenazine • Trilafon
Quetiapine • Seroquel
Risperidone • Risperdal
Valproic acid • Depakene, Depakote
Ziprasidone • Geodon
Mr. Q, age 36, has a history of schizophrenia. He is brought to the hospital due to persistent auditory hallucinations and paranoid delusions. His history documents a trial and failure of risperidone, 4 mg twice daily, and aripiprazole, 20 mg/d. Based on this, the treatment team initiates haloperidol, 5 mg twice daily. Because he experiences persistent auditory hallucinations and paranoid delusions, Mr. Q is titrated to increasing doses of haloperidol over 2 weeks during the course of the hospitalization. Once Mr. Q is receiving a total haloperidol dose of 30 mg/d, the team decides to obtain a serum haloperidol level due to his persistent psychotic symptoms and the development of drug-induced parkinsonism. His serum haloperidol level is 24 ng/mL, which is within the expected range for his dose, but above the therapeutic window for efficacy. The team decides that the severity of Mr. Q’s illness and documented treatment resistance (failing at least 2 adequate trials of antipsychotics) warrant a trial of clozapine.
Despite a long history of therapeutic drug monitoring (TDM) within psychiatry, routine monitoring of antipsychotic serum levels has not been unanimously adopted as standard practice. Clinical practice typically results in mostly a subjective assessment of the safety and efficacy of antipsychotics. This practice is in contrast to agents such as valproic acid and lithium, which are routinely monitored for safety and efficacy using both subjective and laboratory measures. Clinicians may adhere to these monitoring practices for lithium and valproic acid because of these agents’ narrow “therapeutic window” between toxicity and efficacy. However, antipsychotics can be viewed in a similar fashion.
To help conceptualize the therapeutic window for antipsychotics, it is important to understand that in most cases, the pharmacologic target for antipsychotics is dopamine (D2)receptor antagonism between 60% to 80%.1 Total drug exposure would thus determine a patient’s likelihood of minimizing positive symptoms, or exposure to adverse effects related to total dopamine antagonism. Serum drug concentrations are a better metric than total daily dose for determining drug exposure and achieving the pharmacologic target.2 Evaluating serum antipsychotic levels also is a better method of determining true treatment failure than relying on the clinical judgment of the treating psychiatrist.3
Pros and cons of TDM
Benefits of using TDM for patients being treated with antipsychotics include4:
- ensuring adherence
- quantitatively adjusting dosages for medication interactions or genetic variations
- ensuring an adequate trial of a medication before considering it a treatment failure.
Potential drawbacks to TDM include:
- Delayed results. Access to expeditious testing may not be possible in certain laboratories, and this may require send-out testing, which could result in a delay in obtaining results. Continued advocacy and research on the value of TDM in antipsychotics may improve access to these resources in the future. Nonetheless, obtaining antipsychotic serum levels will still give clinicians insight into the antipsychotic exposure at a given dose. Further, obtaining antipsychotic serum levels may strengthen decisions about treatment resistance and the assessment of interactions, adherence, or the likelihood of adverse effects.
- Lack of guidance. Unfortunately, there is no established guidance outlining what to do once antipsychotic serum levels are obtained. The correlation of serum levels of commonly used second-generation antipsychotics with clinical efficacy needs to be more closely investigated; however, certain agents do have more data associated with appropriate ranges for efficacy/toxicity. While researchers should continue to study the precise relationship between antipsychotic serum levels and effect, clinicians still have resources available to help determine what the expected serum value for a given patient may be. Knowing an expected serum level may help clinicians determine whether there is an unknown interaction or genetic variation that is causing lower- or higher-than-expected levels. This may also help determine whether a patient is adhering to their medication regimen.
Growing evidence for TDM
In recent years, evidence supporting the use of TDM in patients receiving antipsychotics has been increasing, and recommendations from consensus groups have been strengthened. One of the most comprehensive assessments of these practices was published by the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie (AGNP), a German-based psychopharmacology group consisting of researchers and clinicians. In 2018, the AGNP published consensus guidelines for TDM in various neuropsychiatric medication classes and recommendations for collecting, interpreting, and decision-making elements of the process.5 The Table5 lists the AGNP-recommended therapeutic serum range for several commonly used second-generation antipsychotics.

Researchers should be encouraged to contribute to the body of knowledge on the correlation of clinical response to serum level. However, there is compelling evidence for the use of TDM across many first- and second-generation antipsychotics. Of the most common, haloperidol and clozapine have evidence of a therapeutic range that is better correlated with serum level than daily dose. Specifically, haloperidol appears to lose benefit with dose increases beyond serum levels of approximately 10 ng/mL.6
Continue to: Clozapine levels may be...
Clozapine levels may be reported by measuring the metabolite norclozapine, which is not correlated with efficacy, or as a total level (combination of clozapine and norclozapine). While norclozapine is not associated with efficacy, the ratio of clozapine to norclozapine may indicate adherence to the medication, or any enzymatic modulation (genetic or drug–drug interaction) that may increase or decrease total exposure. A ratio of 1.5 to 2.0 (clozapine to norclozapine) is optimal; a ratio <0.5 may indicate nonadherence; and a ratio >2.0 may indicate inhibited drug clearance. A 12-hour serum clozapine level of ≥350 ng/mL is more likely to predict treatment response.7
CASE CONTINUED
Mr. Q is carefully tapered from haloperidol while initiating clozapine at 25 mg/d. As he is titrated on clozapine, Mr. Q’s serum levels are periodically checked and compared with expected levels and levels associated with efficacy. Eventually, Mr. Q is titrated to a clozapine dose of 400 mg/d at bedtime.
While receiving clozapine for 4 weeks, Mr. Q’s psychotic symptoms resolve, and he is scheduled for follow-up in the outpatient clozapine clinic.
Related Resources
- De Leon J. A critical commentary on the 2017 AGNP consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology. Pharmacopsychiatry. 2018; 51(1-02):63-68.
- Meyer JM. Is monitoring of plasma antipsychotic levels useful? Current Psychiatry. 2015;14(11):16,19-20.
Drug Brand Names
Aripiprazole • Abilify
Clozapine • Clozaril
Fluphenazine • Prolixin
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Olanzapine • Zyprexa
Perphenazine • Trilafon
Quetiapine • Seroquel
Risperidone • Risperdal
Valproic acid • Depakene, Depakote
Ziprasidone • Geodon
1. Stahl SM. Stahl’s essential psychopharmacology, neuroscientific basis and practical applications, 4th ed. Cambridge, United Kingdom: Cambridge University Press; 2013:129-236.
2. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181.
3. McCutcheon R, Beck K, D’Ambrosio E, et al. Antipsychotic plasma levels in the assessment of poor treatment response in schizophrenia. Acta Psychiatr Scand. 2018;137(1):39-46.
4. Horvitz-Lennon M, Mattke S, Predmore Z, et al. The role of antipsychotic plasma levels in the treatment of schizophrenia. Am J Psychiatry. 2017;174(5):421-426.
5. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62.
6. Van Putten T, Marder SR, Mintz J, et al. Haloperidol plasma levels and clinical response: a therapeutic window relationship. Am J Psychiatry. 1992;149(4):500-505.
7. Couchman L, Morgan PE, Spencer EP, et al. Plasma clozapine, norclozapine, and the clozapine:norclozapine ratio in relation to prescribed dose and other factors: data from a therapeutic drug monitoring service, 1993-2007. Ther Drug Monit. 2010;32(4):438-447.
1. Stahl SM. Stahl’s essential psychopharmacology, neuroscientific basis and practical applications, 4th ed. Cambridge, United Kingdom: Cambridge University Press; 2013:129-236.
2. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181.
3. McCutcheon R, Beck K, D’Ambrosio E, et al. Antipsychotic plasma levels in the assessment of poor treatment response in schizophrenia. Acta Psychiatr Scand. 2018;137(1):39-46.
4. Horvitz-Lennon M, Mattke S, Predmore Z, et al. The role of antipsychotic plasma levels in the treatment of schizophrenia. Am J Psychiatry. 2017;174(5):421-426.
5. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62.
6. Van Putten T, Marder SR, Mintz J, et al. Haloperidol plasma levels and clinical response: a therapeutic window relationship. Am J Psychiatry. 1992;149(4):500-505.
7. Couchman L, Morgan PE, Spencer EP, et al. Plasma clozapine, norclozapine, and the clozapine:norclozapine ratio in relation to prescribed dose and other factors: data from a therapeutic drug monitoring service, 1993-2007. Ther Drug Monit. 2010;32(4):438-447.
Understanding postpartum psychosis: From course to treatment
Although the last decade has brought appropriate increased interest in the diagnosis and treatment of postpartum depression, with screening initiatives across more than 40 states in place and even new medications being brought to market for treatment, far less attention has been given to diagnosis and treatment of a particularly serious psychiatric illness: postpartum psychosis.
Clinically, women can experience rapid mood changes, most often with the presentation that is consistent with a manic-like psychosis, with associated symptoms of delusional thinking, hallucinations, paranoia and either depression or elation, or an amalgam of these so-called “mixed symptoms.” Onset of symptoms typically is early, within 72 hours as is classically described, but may have a somewhat later time of onset in some women.
Many investigators have studied risk factors for postpartum psychosis, and it has been well established that a history of mood disorder, particularly bipolar disorder, is one of the strongest predictors of risk for postpartum psychosis. Women with histories of postpartum psychosis are at very high risk of recurrence, with as many as 70%-90% of women experiencing recurrence if not prophylaxed with an appropriate agent. From a clinical point of view, women with postpartum psychosis typically are hospitalized, given that this is both a psychiatric and potential obstetrical emergency. In fact, the data would suggest that although postpartum suicide and infanticide are not common, they can be a tragic concomitant of postpartum psychosis (Am J Psychiatry. 2016 Dec 1;173[12]:1179-88).
A great amount of interest has been placed on the etiology of postpartum psychosis, as it’s a dramatic presentation with very rapid onset in the acute postpartum period. A rich evidence base with respect to an algorithm of treatment that maximizes likelihood of full recovery or sustaining of euthymia after recovery is limited. Few studies have looked systematically at the optimum way to treat postpartum psychosis. Clinical wisdom has dictated that, given the dramatic symptoms with which these patients present, most patients are treated with lithium and an antipsychotic medication as if they have a manic-like psychosis. It may take brief or extended periods of time for patients to stabilize. Once they are stabilized, one of the most challenging questions for clinicians is how long to treat. Again, an evidence base clearly informing this question is lacking.
Over the years, many clinicians have treated patients with postpartum psychosis as if they have bipolar disorder, given the index presentation of the illness, so some of these patients are treated with antimanic drugs indefinitely. However, clinical experience from several centers that treat women with postpartum psychosis suggests that in remitted patients, a proportion of them may be able to taper and discontinue treatment, then sustain well-being for protracted periods.
One obstacle with respect to treatment of postpartum psychosis derives from the short length of stay after delivery for many women. Some women who present with symptoms of postpartum psychosis in the first 24-48 hours frequently are managed with direct admission to an inpatient psychiatric service. But others may not develop symptoms until they are home, which may place both mother and newborn at risk.
Given that the risk for recurrent postpartum psychosis is so great (70%-90%), women with histories of postpartum psychosis invariably are prophylaxed with mood stabilizer prior to delivery in a subsequent pregnancy. In our own center, we have published on the value of such prophylactic intervention, not just in women with postpartum psychosis, but in women with bipolar disorder, who are, as noted, at great risk for developing postpartum psychotic symptoms (Am J Psychiatry. 1995 Nov;152[11]:1641-5.)
Although postpartum psychosis may be rare, over the last 3 decades we have seen a substantial number of women with postpartum psychosis and have been fascinated with the spectrum of symptoms with which some women with postpartum psychotic illness present. We also have been impressed with the time required for some women to recompensate from their illness and the course of their disorder after they have seemingly remitted. Some women appear to be able to discontinue treatment as noted above; others, particularly if there is any history of bipolar disorder, need to be maintained on treatment with mood stabilizer indefinitely.
To better understand the phenomenology of postpartum psychosis, as well as the longitudinal course of the illness, in 2019, the Mass General Hospital Postpartum Psychosis Project (MGHP3) was established. The project is conducted as a hospital-based registry where women with histories of postpartum psychosis over the last decade are invited to participate in an in-depth interview to understand both symptoms and course of underlying illness. This is complemented by obtaining a sample of saliva, which is used for genetic testing to try to identify a genetic underpinning associated with postpartum psychosis, as the question of genetic etiology of postpartum psychosis is still an open one.
As part of the MGHP3 project, clinicians across the country are able to contact perinatal psychiatrists in our center with expertise in the treatment of postpartum psychosis. Our psychiatrists also can counsel clinicians on issues regarding long-term management of postpartum psychosis because for many, knowledge of precisely how to manage this disorder or the follow-up treatment may be incomplete.
From a clinical point of view, the relevant questions really include not only acute treatment, which has already been outlined, but also the issue of duration of treatment. While some patients may be able to taper and discontinue treatment after, for example, a year of being totally well, to date we are unable to know who those patients are. We tend to be more conservative in our own center and treat patients with puerperal psychosis for a more protracted period of time, usually over several years. We also ask women about their family history of bipolar disorder or postpartum psychosis. Depending on the clinical course (if the patient really has sustained euthymia), we consider slow taper and ultimate discontinuation. As always, treatment decisions are tailored to individual clinical history, course, and patient wishes.
Postpartum psychosis remains one of the most serious illnesses that we find in reproductive psychiatry, and incomplete attention has been given to this devastating illness, which we read about periodically in newspapers and magazines. Greater understanding of postpartum psychosis will lead to a more precision-like psychiatric approach, tailoring treatment to the invariable heterogeneity of this illness.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Although the last decade has brought appropriate increased interest in the diagnosis and treatment of postpartum depression, with screening initiatives across more than 40 states in place and even new medications being brought to market for treatment, far less attention has been given to diagnosis and treatment of a particularly serious psychiatric illness: postpartum psychosis.
Clinically, women can experience rapid mood changes, most often with the presentation that is consistent with a manic-like psychosis, with associated symptoms of delusional thinking, hallucinations, paranoia and either depression or elation, or an amalgam of these so-called “mixed symptoms.” Onset of symptoms typically is early, within 72 hours as is classically described, but may have a somewhat later time of onset in some women.
Many investigators have studied risk factors for postpartum psychosis, and it has been well established that a history of mood disorder, particularly bipolar disorder, is one of the strongest predictors of risk for postpartum psychosis. Women with histories of postpartum psychosis are at very high risk of recurrence, with as many as 70%-90% of women experiencing recurrence if not prophylaxed with an appropriate agent. From a clinical point of view, women with postpartum psychosis typically are hospitalized, given that this is both a psychiatric and potential obstetrical emergency. In fact, the data would suggest that although postpartum suicide and infanticide are not common, they can be a tragic concomitant of postpartum psychosis (Am J Psychiatry. 2016 Dec 1;173[12]:1179-88).
A great amount of interest has been placed on the etiology of postpartum psychosis, as it’s a dramatic presentation with very rapid onset in the acute postpartum period. A rich evidence base with respect to an algorithm of treatment that maximizes likelihood of full recovery or sustaining of euthymia after recovery is limited. Few studies have looked systematically at the optimum way to treat postpartum psychosis. Clinical wisdom has dictated that, given the dramatic symptoms with which these patients present, most patients are treated with lithium and an antipsychotic medication as if they have a manic-like psychosis. It may take brief or extended periods of time for patients to stabilize. Once they are stabilized, one of the most challenging questions for clinicians is how long to treat. Again, an evidence base clearly informing this question is lacking.
Over the years, many clinicians have treated patients with postpartum psychosis as if they have bipolar disorder, given the index presentation of the illness, so some of these patients are treated with antimanic drugs indefinitely. However, clinical experience from several centers that treat women with postpartum psychosis suggests that in remitted patients, a proportion of them may be able to taper and discontinue treatment, then sustain well-being for protracted periods.
One obstacle with respect to treatment of postpartum psychosis derives from the short length of stay after delivery for many women. Some women who present with symptoms of postpartum psychosis in the first 24-48 hours frequently are managed with direct admission to an inpatient psychiatric service. But others may not develop symptoms until they are home, which may place both mother and newborn at risk.
Given that the risk for recurrent postpartum psychosis is so great (70%-90%), women with histories of postpartum psychosis invariably are prophylaxed with mood stabilizer prior to delivery in a subsequent pregnancy. In our own center, we have published on the value of such prophylactic intervention, not just in women with postpartum psychosis, but in women with bipolar disorder, who are, as noted, at great risk for developing postpartum psychotic symptoms (Am J Psychiatry. 1995 Nov;152[11]:1641-5.)
Although postpartum psychosis may be rare, over the last 3 decades we have seen a substantial number of women with postpartum psychosis and have been fascinated with the spectrum of symptoms with which some women with postpartum psychotic illness present. We also have been impressed with the time required for some women to recompensate from their illness and the course of their disorder after they have seemingly remitted. Some women appear to be able to discontinue treatment as noted above; others, particularly if there is any history of bipolar disorder, need to be maintained on treatment with mood stabilizer indefinitely.
To better understand the phenomenology of postpartum psychosis, as well as the longitudinal course of the illness, in 2019, the Mass General Hospital Postpartum Psychosis Project (MGHP3) was established. The project is conducted as a hospital-based registry where women with histories of postpartum psychosis over the last decade are invited to participate in an in-depth interview to understand both symptoms and course of underlying illness. This is complemented by obtaining a sample of saliva, which is used for genetic testing to try to identify a genetic underpinning associated with postpartum psychosis, as the question of genetic etiology of postpartum psychosis is still an open one.
As part of the MGHP3 project, clinicians across the country are able to contact perinatal psychiatrists in our center with expertise in the treatment of postpartum psychosis. Our psychiatrists also can counsel clinicians on issues regarding long-term management of postpartum psychosis because for many, knowledge of precisely how to manage this disorder or the follow-up treatment may be incomplete.
From a clinical point of view, the relevant questions really include not only acute treatment, which has already been outlined, but also the issue of duration of treatment. While some patients may be able to taper and discontinue treatment after, for example, a year of being totally well, to date we are unable to know who those patients are. We tend to be more conservative in our own center and treat patients with puerperal psychosis for a more protracted period of time, usually over several years. We also ask women about their family history of bipolar disorder or postpartum psychosis. Depending on the clinical course (if the patient really has sustained euthymia), we consider slow taper and ultimate discontinuation. As always, treatment decisions are tailored to individual clinical history, course, and patient wishes.
Postpartum psychosis remains one of the most serious illnesses that we find in reproductive psychiatry, and incomplete attention has been given to this devastating illness, which we read about periodically in newspapers and magazines. Greater understanding of postpartum psychosis will lead to a more precision-like psychiatric approach, tailoring treatment to the invariable heterogeneity of this illness.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Although the last decade has brought appropriate increased interest in the diagnosis and treatment of postpartum depression, with screening initiatives across more than 40 states in place and even new medications being brought to market for treatment, far less attention has been given to diagnosis and treatment of a particularly serious psychiatric illness: postpartum psychosis.
Clinically, women can experience rapid mood changes, most often with the presentation that is consistent with a manic-like psychosis, with associated symptoms of delusional thinking, hallucinations, paranoia and either depression or elation, or an amalgam of these so-called “mixed symptoms.” Onset of symptoms typically is early, within 72 hours as is classically described, but may have a somewhat later time of onset in some women.
Many investigators have studied risk factors for postpartum psychosis, and it has been well established that a history of mood disorder, particularly bipolar disorder, is one of the strongest predictors of risk for postpartum psychosis. Women with histories of postpartum psychosis are at very high risk of recurrence, with as many as 70%-90% of women experiencing recurrence if not prophylaxed with an appropriate agent. From a clinical point of view, women with postpartum psychosis typically are hospitalized, given that this is both a psychiatric and potential obstetrical emergency. In fact, the data would suggest that although postpartum suicide and infanticide are not common, they can be a tragic concomitant of postpartum psychosis (Am J Psychiatry. 2016 Dec 1;173[12]:1179-88).
A great amount of interest has been placed on the etiology of postpartum psychosis, as it’s a dramatic presentation with very rapid onset in the acute postpartum period. A rich evidence base with respect to an algorithm of treatment that maximizes likelihood of full recovery or sustaining of euthymia after recovery is limited. Few studies have looked systematically at the optimum way to treat postpartum psychosis. Clinical wisdom has dictated that, given the dramatic symptoms with which these patients present, most patients are treated with lithium and an antipsychotic medication as if they have a manic-like psychosis. It may take brief or extended periods of time for patients to stabilize. Once they are stabilized, one of the most challenging questions for clinicians is how long to treat. Again, an evidence base clearly informing this question is lacking.
Over the years, many clinicians have treated patients with postpartum psychosis as if they have bipolar disorder, given the index presentation of the illness, so some of these patients are treated with antimanic drugs indefinitely. However, clinical experience from several centers that treat women with postpartum psychosis suggests that in remitted patients, a proportion of them may be able to taper and discontinue treatment, then sustain well-being for protracted periods.
One obstacle with respect to treatment of postpartum psychosis derives from the short length of stay after delivery for many women. Some women who present with symptoms of postpartum psychosis in the first 24-48 hours frequently are managed with direct admission to an inpatient psychiatric service. But others may not develop symptoms until they are home, which may place both mother and newborn at risk.
Given that the risk for recurrent postpartum psychosis is so great (70%-90%), women with histories of postpartum psychosis invariably are prophylaxed with mood stabilizer prior to delivery in a subsequent pregnancy. In our own center, we have published on the value of such prophylactic intervention, not just in women with postpartum psychosis, but in women with bipolar disorder, who are, as noted, at great risk for developing postpartum psychotic symptoms (Am J Psychiatry. 1995 Nov;152[11]:1641-5.)
Although postpartum psychosis may be rare, over the last 3 decades we have seen a substantial number of women with postpartum psychosis and have been fascinated with the spectrum of symptoms with which some women with postpartum psychotic illness present. We also have been impressed with the time required for some women to recompensate from their illness and the course of their disorder after they have seemingly remitted. Some women appear to be able to discontinue treatment as noted above; others, particularly if there is any history of bipolar disorder, need to be maintained on treatment with mood stabilizer indefinitely.
To better understand the phenomenology of postpartum psychosis, as well as the longitudinal course of the illness, in 2019, the Mass General Hospital Postpartum Psychosis Project (MGHP3) was established. The project is conducted as a hospital-based registry where women with histories of postpartum psychosis over the last decade are invited to participate in an in-depth interview to understand both symptoms and course of underlying illness. This is complemented by obtaining a sample of saliva, which is used for genetic testing to try to identify a genetic underpinning associated with postpartum psychosis, as the question of genetic etiology of postpartum psychosis is still an open one.
As part of the MGHP3 project, clinicians across the country are able to contact perinatal psychiatrists in our center with expertise in the treatment of postpartum psychosis. Our psychiatrists also can counsel clinicians on issues regarding long-term management of postpartum psychosis because for many, knowledge of precisely how to manage this disorder or the follow-up treatment may be incomplete.
From a clinical point of view, the relevant questions really include not only acute treatment, which has already been outlined, but also the issue of duration of treatment. While some patients may be able to taper and discontinue treatment after, for example, a year of being totally well, to date we are unable to know who those patients are. We tend to be more conservative in our own center and treat patients with puerperal psychosis for a more protracted period of time, usually over several years. We also ask women about their family history of bipolar disorder or postpartum psychosis. Depending on the clinical course (if the patient really has sustained euthymia), we consider slow taper and ultimate discontinuation. As always, treatment decisions are tailored to individual clinical history, course, and patient wishes.
Postpartum psychosis remains one of the most serious illnesses that we find in reproductive psychiatry, and incomplete attention has been given to this devastating illness, which we read about periodically in newspapers and magazines. Greater understanding of postpartum psychosis will lead to a more precision-like psychiatric approach, tailoring treatment to the invariable heterogeneity of this illness.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
FDA strengthens warning regarding clozapine, serious bowel complication risk
The Food and Drug Administration is strengthening a previous warning regarding the uncommon risk of serious bowel complications associated with the schizophrenia medication clozapine (Clozaril, FazaClo ODT, Versacloz).
According to the FDA press release, dated Jan. 28, clozapine affects bowel function in a majority of patients, and constipation is a common adverse event associated with clozapine use. This can uncommonly progress to serious bowel complications, including complete bowel blockage, and can result in hospitalization or even death if the constipation is not diagnosed and treated quickly.
Patients should contact their health care clinician if their bowel movements are less frequent, they have a bowel movement less than three times a week, they have hard or dry stool, or they have difficulty passing gas. Urgent care is needed if patients are experiencing nausea, vomiting, belly pain, or bloating, according to the FDA.
In addition, , avoid coprescribing with other anticholinergic medicines, advise and question patients about the risks of clozapine and their bowel movements, monitor patients for complications, and consider prophylactic laxative treatment in patients with a history of constipation or bowel obstruction, the FDA added.
The Food and Drug Administration is strengthening a previous warning regarding the uncommon risk of serious bowel complications associated with the schizophrenia medication clozapine (Clozaril, FazaClo ODT, Versacloz).
According to the FDA press release, dated Jan. 28, clozapine affects bowel function in a majority of patients, and constipation is a common adverse event associated with clozapine use. This can uncommonly progress to serious bowel complications, including complete bowel blockage, and can result in hospitalization or even death if the constipation is not diagnosed and treated quickly.
Patients should contact their health care clinician if their bowel movements are less frequent, they have a bowel movement less than three times a week, they have hard or dry stool, or they have difficulty passing gas. Urgent care is needed if patients are experiencing nausea, vomiting, belly pain, or bloating, according to the FDA.
In addition, , avoid coprescribing with other anticholinergic medicines, advise and question patients about the risks of clozapine and their bowel movements, monitor patients for complications, and consider prophylactic laxative treatment in patients with a history of constipation or bowel obstruction, the FDA added.
The Food and Drug Administration is strengthening a previous warning regarding the uncommon risk of serious bowel complications associated with the schizophrenia medication clozapine (Clozaril, FazaClo ODT, Versacloz).
According to the FDA press release, dated Jan. 28, clozapine affects bowel function in a majority of patients, and constipation is a common adverse event associated with clozapine use. This can uncommonly progress to serious bowel complications, including complete bowel blockage, and can result in hospitalization or even death if the constipation is not diagnosed and treated quickly.
Patients should contact their health care clinician if their bowel movements are less frequent, they have a bowel movement less than three times a week, they have hard or dry stool, or they have difficulty passing gas. Urgent care is needed if patients are experiencing nausea, vomiting, belly pain, or bloating, according to the FDA.
In addition, , avoid coprescribing with other anticholinergic medicines, advise and question patients about the risks of clozapine and their bowel movements, monitor patients for complications, and consider prophylactic laxative treatment in patients with a history of constipation or bowel obstruction, the FDA added.
Half of SLE patients have incident neuropsychiatric events
Neuropsychiatric events occurred in just over half of all patients recently diagnosed with systemic lupus erythematosus and followed for an average of nearly 8 years in an international study of more than 1,800 patients.
Up to 30% of these neuropsychiatric (NP) events in up to 20% of the followed cohort were directly attributable to systemic lupus erythematosus (SLE) in a representative patient population, wrote John G. Hanly, MD, and associates in Annals of the Rheumatic Diseases. Their findings were consistent with prior reports, they added.
Another notable finding from follow-up of these 1,827 SLE patients was that among those without a history of SLE-related NP events at baseline, 74% remained free of NP events during the subsequent 10 years, wrote Dr. Hanly, professor of medicine and director of the lupus clinic at Dalhousie University, Halifax, N.S., and coauthors. Among patients free from SLE-associated NP events after 2 years, 84% remained event free during their remaining follow-up. SLE patients with a history of an NP event that subsequently resolved had a 72% rate of freedom from another NP event during 10 years of follow-up.
These findings came from patients recently diagnosed with SLE (within the preceding 15 months) and enrolled at any of 31 participating academic medical centers in North America, Europe, and Asia. The investigators considered preenrollment NP events to include those starting from 6 months prior to diagnosis of SLE until the time patients entered the study. They used case definitions for 19 SLE-associated NP events published by the American College of Rheumatology (Arthritis Rheum. 1999 Apr;42[4]:599-608). All enrolled patients underwent annual assessment for NP events, with follow-up continuing as long as 18 years.
The researchers identified NP events in 955 of the 1,827 enrolled patients, a 52% incidence, including 1,910 unique NP events that included episodes from each of the 19 NP event types, with 92% involving the central nervous system and 8% involving the peripheral nervous system. The percentage of NP events attributable to SLE ranged from 17% to 31%, and they occurred in 14%-21% of the studied patients, with the range reflecting various attribution models used in the analyses. Some patients remained in the same NP state, while others progressed through more than one state.
The study did not receive commercial funding. Dr. Hanly had no disclosures.
SOURCE: Hanly JG et al. Ann Rheum Dis. 2020 Jan 8. doi: 10.1136/annrheumdis-2019-216150.
Neuropsychiatric events occurred in just over half of all patients recently diagnosed with systemic lupus erythematosus and followed for an average of nearly 8 years in an international study of more than 1,800 patients.
Up to 30% of these neuropsychiatric (NP) events in up to 20% of the followed cohort were directly attributable to systemic lupus erythematosus (SLE) in a representative patient population, wrote John G. Hanly, MD, and associates in Annals of the Rheumatic Diseases. Their findings were consistent with prior reports, they added.
Another notable finding from follow-up of these 1,827 SLE patients was that among those without a history of SLE-related NP events at baseline, 74% remained free of NP events during the subsequent 10 years, wrote Dr. Hanly, professor of medicine and director of the lupus clinic at Dalhousie University, Halifax, N.S., and coauthors. Among patients free from SLE-associated NP events after 2 years, 84% remained event free during their remaining follow-up. SLE patients with a history of an NP event that subsequently resolved had a 72% rate of freedom from another NP event during 10 years of follow-up.
These findings came from patients recently diagnosed with SLE (within the preceding 15 months) and enrolled at any of 31 participating academic medical centers in North America, Europe, and Asia. The investigators considered preenrollment NP events to include those starting from 6 months prior to diagnosis of SLE until the time patients entered the study. They used case definitions for 19 SLE-associated NP events published by the American College of Rheumatology (Arthritis Rheum. 1999 Apr;42[4]:599-608). All enrolled patients underwent annual assessment for NP events, with follow-up continuing as long as 18 years.
The researchers identified NP events in 955 of the 1,827 enrolled patients, a 52% incidence, including 1,910 unique NP events that included episodes from each of the 19 NP event types, with 92% involving the central nervous system and 8% involving the peripheral nervous system. The percentage of NP events attributable to SLE ranged from 17% to 31%, and they occurred in 14%-21% of the studied patients, with the range reflecting various attribution models used in the analyses. Some patients remained in the same NP state, while others progressed through more than one state.
The study did not receive commercial funding. Dr. Hanly had no disclosures.
SOURCE: Hanly JG et al. Ann Rheum Dis. 2020 Jan 8. doi: 10.1136/annrheumdis-2019-216150.
Neuropsychiatric events occurred in just over half of all patients recently diagnosed with systemic lupus erythematosus and followed for an average of nearly 8 years in an international study of more than 1,800 patients.
Up to 30% of these neuropsychiatric (NP) events in up to 20% of the followed cohort were directly attributable to systemic lupus erythematosus (SLE) in a representative patient population, wrote John G. Hanly, MD, and associates in Annals of the Rheumatic Diseases. Their findings were consistent with prior reports, they added.
Another notable finding from follow-up of these 1,827 SLE patients was that among those without a history of SLE-related NP events at baseline, 74% remained free of NP events during the subsequent 10 years, wrote Dr. Hanly, professor of medicine and director of the lupus clinic at Dalhousie University, Halifax, N.S., and coauthors. Among patients free from SLE-associated NP events after 2 years, 84% remained event free during their remaining follow-up. SLE patients with a history of an NP event that subsequently resolved had a 72% rate of freedom from another NP event during 10 years of follow-up.
These findings came from patients recently diagnosed with SLE (within the preceding 15 months) and enrolled at any of 31 participating academic medical centers in North America, Europe, and Asia. The investigators considered preenrollment NP events to include those starting from 6 months prior to diagnosis of SLE until the time patients entered the study. They used case definitions for 19 SLE-associated NP events published by the American College of Rheumatology (Arthritis Rheum. 1999 Apr;42[4]:599-608). All enrolled patients underwent annual assessment for NP events, with follow-up continuing as long as 18 years.
The researchers identified NP events in 955 of the 1,827 enrolled patients, a 52% incidence, including 1,910 unique NP events that included episodes from each of the 19 NP event types, with 92% involving the central nervous system and 8% involving the peripheral nervous system. The percentage of NP events attributable to SLE ranged from 17% to 31%, and they occurred in 14%-21% of the studied patients, with the range reflecting various attribution models used in the analyses. Some patients remained in the same NP state, while others progressed through more than one state.
The study did not receive commercial funding. Dr. Hanly had no disclosures.
SOURCE: Hanly JG et al. Ann Rheum Dis. 2020 Jan 8. doi: 10.1136/annrheumdis-2019-216150.
FROM ANNALS OF THE RHEUMATIC DISEASES
Antipsychotics, dopamine, and pain
Our understanding of pain mechanisms continues to evolve and, accordingly, so do our treatment strategies. The fundamental differences between acute and chronic pain were only recently recognized; this lack of recognition led to the application of acute pain treatments to chronic pain, contributing to the opioid epidemic in the United States.
With the diminishing emphasis on opioid medications, researchers are exploring other pharmacologic modalities for treating pain. Many nonopioid psychiatric medications are used off-label for the treatment of pain. Psychiatric medications play a larger role in the management of pain as pain becomes more chronic (Table 11). For simplicity, acute pain may be seen as nociception colored by emotions, and chronic pain as emotions colored by nociception. Protracted pain connects those extremes with a diminishing role of nociception and an increasing role of emotion,1 which may increase the potential role of psychiatric medications, including antipsychotics.
In this article, I discuss the potential role of dopamine in the perception of pain, and review the potential use of first- and second-generation antipsychotics for treating various pain syndromes.
Role of dopamine in pain
There is increasing interest in exploring antipsychotics to treat chronic pain2 because dopamine dysfunction is part of pathological pain perception. Excess dopamine is associated with headaches (dopamine hypersensitivity hypothesis3,4) and dopamine dysfunction is a part of posttraumatic stress disorder (PTSD),5 dissociation,6 paranoia,7 and catastrophizing.8 Somatic psychosis, like any psychosis, can be based on dopamine pathology. Dopaminergic neurons affect nociceptive function in the spinal dorsal horn,9 and dopamine receptors are altered in atypical facial pain,10 burning mouth syndrome,11 and fibromyalgia.12
In normal circumstances, dopamine is fundamentally a protective neurotransmitter. In acute pain, dopamine is powerfully released, making the pain bearable. A patient may describe acute pain as seeming “like it was not happening to me” or “it was like a dream”; both are examples of dopamine-caused dissociation and a possible prediction of subsequent chronification. In chronic pain, pathological mechanisms settle in and take root; therefore, keeping protective dopamine levels high becomes a priority. This is especially common in patients who have experienced abuse or PTSD. The only natural way to keep dopamine up for prolonged periods of time is to decrease pain and stress thresholds. Both phenomena are readily observed in patients with pain. In extreme cases, self-mutilation and involvement in conflicts become pathologically gratifying.
The dopaminergic system is essential for pain control with a tissue injury.13 It becomes pathologically stimulated and increasingly dysfunctional as algopathy (a pathological pain perception) develops. At the same time, a flood or drought of any neurotransmitter is equally bad and may produce similar clinical pictures. Both a lack of and excess of dopamine are associated with pain.14 This is why opposite treatments may be beneficial in different patients with chronic pain. As an example, the use of stimulants15 and bupropion16 has been reported in the treatment of abdominal pain. And, reversely, antipsychotics, especially first-generation agents, may be associated with chronic (tardive) pain, including orofacial and genital pain.17
First-generation antipsychotics
First-generation antipsychotics (FGAs) have been used to treat various nonpsychiatric conditions (Table 2). Although they are powerful D2 receptor inhibitors, FGAs lack the intrinsic ability to counteract the unwanted adverse effects of strong inhibition. As a result, movement disorders and prolactinemia are commonly induced by FGAs. The most dangerous consequence of treatment with these agents is neuroleptic malignant syndrome (NMS).
Continue to: Haloperidol
Haloperidol is prescribed widely by nonpsychiatrists, primarily to treat agitation. Intravenous haloperidol has been used for the abortive treatment of headaches.18 Paradoxically, IV haloperidol is less likely to induce extrapyramidal symptoms (EPS) than the oral formulation because of a more pronounced anticholinergic action in IV use. Haloperidol can help relieve gastroparesis and nausea, especially in IV administration,19 but prolonged oral administration is associated with unwanted movement problems and should be avoided.20
Chlorpromazine is more anticholinergic than haloperidol. It can be used in the abortive treatment of headaches (preferably via IV and IM administration), nausea, hiccups, porphyria, and serotonin syndrome, but it is very sedating and frequently produces hypotension, dangerous QT prolongation, and sensations of thought-blocking.21
Pimozide is reported to help with skin picking, trichotillomania, and somatic hallucinations.22
Droperidol, promethazine, and prochlorperazine are used off-label to treat nausea and headaches. Primary care clinicians may not be aware that these commonly used medications are antipsychotics. Similar to other FGAs, these 3 agents may produce NMS and tardive dyskinesia (TD). The same applies to the prokinetic drug metoclopramide.
Second-generation antipsychotics
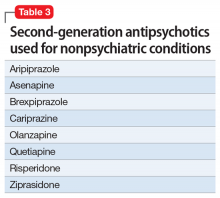
Second-generation antipsychotics (SGAs) work with various serotonin receptors, offsetting and enhancing the antipsychotic function of dopamine blockade. This diminishes but does not eliminate EPS and the risk of TD. Fortunately, the risk of NMS is lower with SGAs than with FGAs. Many SGAs are FDA-approved for treating schizophrenia and other psychiatric disorders, and some have relevance for pain management (Table 3). Many SGAs help with depressive symptoms and are powerful mood stabilizers. As such, they may diminish central over-firing of dopaminergic and serotonergic neurons involved in the pain cascade, which in turn decreases pain transmission and perception. The downside is that in general, SGAs increase the risk of diabetes and hyperlipidemia.
Continue to: Risperidone
Risperidone was the second FDA-approved SGA. Pain practitioners primarily prescribe it for treatmeant-resistant headaches, but patients with fibromyalgia and those with phantom and thalamic pain also may respond. Because risperidone’s properties are similar to that of many FGAs, it may potently cause EPS, TD, and prolactinemia. Neuroleptic malignant syndrome also has been reported.23
Ziprasidone is frequently overlooked by clinicians who treat pain. Although ziprasidone may be sedating, it is powerful as both a preventive and abortive (in an IM formulation) agent for treatment-resistant headaches. This might be attributed to its effects on the 5HT9 receptor. It is approved for treating bipolar depression and has been prescribed to effectively treat anxiety. For patients receiving ziprasidone, QT prolongation needs to be monitored closely.24
Olanzapine was modeled after clozapine and is effective as a mood stabilizer and an antianxiety, antipsychotic, and sleep-promoting medication. It has a useful “mellowing” effect and helps with central pain syndrome management. Patients with fibromyalgia respond well; in some cases, patients with phantom and thalamic pain also respond. Among SGAs prescribed to treat chronic pain, olanzapine has the most published studies. However, the downside is the risk of severe weight gain and diabetes. Usually, if a patient is already overweight, they gain less, but these patients typically are concerned about any additional weight gain.25
Aripiprazole is a partial dopamine agonist. It increases dopamine function in the prefrontal cortex, and by doing so it possibly improves cognition, mental acuity, goal-oriented activity, and attention. At the same time, it decreases dopamine activity in the basal ganglia and limbic system, improving catastrophizing, paranoia, abnormal pain perception, and multiple homeostasis functions. This combination of effects can be invaluable for some patients, but depending on individual susceptibility, aripiprazole might be too activating (causing agitation and akathisia) or too sedating.26
Brexpiprazole is a relative of aripiprazole, but for some patients it is better tolerated, and compliance with this medication usually is good. It partially antagonizes the D2 and 5HT1A receptors while antagonizing the 5HT2A receptors (which decreases the dopamine release in the striatum) and mimics the mechanism of action of an antidepressant. Through alpha-1-adrenergic receptor antagonism, it reduces EPS. All these effects are also part of the mechanisms of action of quetiapine, clozapine, and iloperidone, but brexpiprazole is considered to be the most alpha-1 antagonistic, which is a mechanism of action of other potential pain-controlling medications such as clonidine and tizanidine. In patients with pain who have an overactive noradrenergic system, this property may be beneficial. Its major problem stems from cytochrome P450 2D6 (CYP2D6) enzyme-dependent metabolism, which causes an approximately 5-fold increase in brexpiprazole blood level in poor CYP2D6 metabolizers. Therefore, combining brexpiprazole with CYP2D6 inhibitors such as fluoxetine, paroxetine, and duloxetine would be unwise. Aripiprazole and brexpiprazole are less associated with diabetes and sexual adverse effects than many other SGAs.27
Continue to: Asenapine
Asenapine is an underutilized antipsychotic. Its mechanism of action spans multiple receptors and is less specific in individual receptor activity than other dopamine blockers. It is administered under the tongue due to poor absorption when swallowed, and its molecule has an anesthetic property that causes mouth and tongue numbness/paresthesia. This function may help patients with orofacial pain. Significant somnolence and weight gain (although less than with olanzapine) limit its use. Some patients cannot tolerate the taste.28
Quetiapine is prescribed rather frequently due to its significant antianxiety effect. It is also reported to be beneficial in pain control.29 Weight gain may be severe. In doses smaller than typically administered to patients with bipolar disorder or schizophrenia, quetiapine is widely prescribed off-label for sleep. In lower doses, it acts primarily as an antihistamine (hence the sedation), but at an increased dose it activates the adrenergic system, which offsets sedation. Quetiapine antagonizes H1 histamine and 5HT2
Cariprazine is typically well tolerated because of its benign metabolic profile. It does not increase the QT interval and is not sedating. Cariprazine is a D2 and D3 partial receptor agonist. This allows the medication to inhibit overstimulated dopamine receptors (a desirable effect in pain management) and induces them when the endogenous dopamine level is low (helping with cognition, volition, and attention). Pro-cognitive effects are always beneficial for patients with pain. Cariprazine produces less EPS due to more ventral striatum vs dorsal striatum activity. Mood improvement caused by this medication is attributed to its 5HT2A, 5HT2B, and 5HT2C inverse agonism, which modulates the serotonergic system. Cariprazine will likely have a positive future in pain management because it has shown efficacy in the chronic stress model.33
A complex condition
No single medication or group of medications may be exclusively relied on for treating patients with chronic pain. Identifying alternatives to opioids for treating pain brings more attention to centrally-acting medications that may aid in the stabilization of the nervous system, which can decrease pathological pain perception and help patients cope with chronic painful conditions.
Bottom Line
Antipsychotics may be a valuable asset in the treatment of chronic pain, offering a potential alternative to prescribing opioids for pain. More research is needed to identify specific ways of using dopamine blockade or dopamine enhancement to help patients with chronic pain.
Continue to: Related Resource
Related Resource
- Tripathi A. Antipsychotics for migraines, cluster headaches, and nausea. Current Psychiatry. 2013;12(2):E1-E4.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Bupropion • Wellbutrin, Zyban
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Clonidine • Catapres
Clozapine • Clozaril
Droperidol • Inapsine
Duloxetine • Cymbalta
Fluoxetine • Prozac
Haloperidol • Haldol
Iloperidone • Fanapt
Metoclopramide • Reglan
Olanzapine • Zyprexa
Paroxetine • Paxil
Pimozide • Orap
Prochlorperazine • Compazine
Promethazine • Phenergan
Quetiapine • Seroquel
Risperidone • Risperdal
Tizanidine • Zanaflex
Ziprasidone • Geodon
1. Arbuck D, Pergolizzi J. Algopathy—acknowledging the pathological process of pain chronification. Pract Pain Manag. 2017;17(4):4,26-32.
2. Shin SW, Lee JS, Abdi S, et al. Antipsychotics for patients with pain. Korean J Pain. 2019;32(1):3-11.
3. D’Andrea G, Leone M, Bussone G, et al. Abnormal tyrosine metabolism in chronic cluster headache. Cephalalgia. 2017;37(2):148-153.
4. D’Andrea G, Granella F, Perini F, et al. Platelet levels of dopamine are increased in migraine and cluster headache. Headache. 2006;46(4):585-591.
5. Wolf EJ, Mitchell KS, Logue MW, et al. The dopamine D3 receptor gene, and posttraumatic stress disorder. J Trauma Stress. 2014;27(4):379-387.
6. den Ouden HEM, Daw ND, Fernandez G, et al. Dissociable effects of dopamine and serotonin on reversal learning. Neuron. 2013;80(4):1090-1100.
7. Nour MM, Dahoun T, Schwartenbeck P, et al. Dopaminergic basis for signaling belief updates, but not surprise, and the link to paranoia. Proc Natl Acad Sci U S A. 2018;115(43):E10167-E10176.
8. Zhu H, Clemens S, Sawchuk M, et al. Expression and distribution of all dopamine receptor subtypes (D(1)-D(5)) in the mouse lumbar spinal cord: a real-time polymerase chain reaction and non-autoradiographic in situ hybridization study. Neuroscience. 2007;149:885-897.
9. Wood PB, Schweinhardt P, Jaeger E, et al. Fibromyalgia patients show an abnormal dopamine response to pain. Eur J Neurosci. 2007;25:3576-3582.
10. Hagelberg N, Fossell H, Aalto S, et al. Altered dopamine D2 receptor binding in atypical facial pain. Pain. 2003;106(1-2):43-48.
11. Hagelberg N, Fossell H, Rinne JD, et al. Striatal dopamine D1 and D2 receptors in burning mouth syndrome. Pain. 2003;101(1-2):149-154.
12. Elman I, Borsook D. Common brain mechanisms of chronic pain and addiction. Neuron. 2016;89(1):11-36.
13. Siahposht-Khachaki A, Pourreza P, Ezzatpanah S, et al. Nucleus accumbens dopamine receptors mediate hypothalamus-induced antinociception in the rat formalin test. Eur J Pain. 2017;21(7):1285-1294.
14. Thompson T, Gallop K, Correll CU, et al. Pain perception in Parkinson’s disease: a systematic review and meta-analysis of experimental studies. Aging Res Rev. 2017;35:74-86.
15. Check JH. Chronic unremitting lower abdominal pain quickly abrogated following treatment with amphetamine. Clin Exp Obstet Gynecol. 2016;43(1):109-111.
16. Wilkes S. Bupropion. Drugs Today (Barc). 2006;42(10):671-681.
17. Frei K, Truong DD, Fahn S, et al. The nosology of tardive syndromes. J Neurol Sci. 2018;389:10-16.
18. Honkaniemi J, Liimatainen S, Rainesalo S, et al. Haloperidol in the acute treatment of migraine: a randomized, double-blind, placebo-controlled study. Headache. 2006;46(5):781-787.
19. Murray-Brown F, Dorman S. Haloperidol for the treatment of nausea and vomiting in palliative care patients. Cochrane Database Syst Rev. 2015;(11):CD006271.
20. Gaffigan ME, Bruner DI, Wason C, et al. A randomized controlled trial of intravenous haloperidol vs. intravenous metoclopramide for acute migraine therapy in the emergency department. J Emerg Med. 2015;49(3):326-334.
21. Weinman D, Nicastro O, Akala O, et al. Parenteral treatment of episodic tension-type headache: a systematic review. Headache. 2014;54(2):260-268.
22. Arnold LM, Auchenbach MB, McElroy SL. Psychogenic excoriation. Clinical features, proposed diagnostic criteria, epidemiology, and approaches to treatment. CNS Drugs. 2001;15(5):351-359.
23. Khouzam HR. Psychopharmacology of chronic pain: a focus on antidepressants and atypical antipsychotics. Postgrad Med. 2016;128(3):323-330.
24. Landsness EC, Wang LH, Bucelli RC. Ziprasidone as a potential abortive therapy for status migrainosus. Neurohospitalist. 2016;6(4):151-156.
25. Jimenez XF, Sundararajan T, Covington EC. A systematic review of atypical antipsychotics in chronic pain management: olanzapine demonstrates potential in central sensitization, fibromyalgia, and headache/migraine. Clin J Pain. 2018;34(6):585-591.
26. Fei L, Abrardi L, Mediati RD. Unexpected effect of aripiprazole on nociceptive pain. Ther Adv Psychopharmacol. 2012;2(5):211-212.
27. Markovic M, Gallipani A, Patel KH, et al. Brexpiprazole. Ann Pharmacother. 2017;51(4):315-322.
28. Gerrits M, de Greef R, Peeters P. Effect of absorption site on the pharmacokinetics of sublingual asenapine in healthy male subjects. Biopharm Drug Dispos. 2010;31(5-6):351-357.
29. Heo MH, Kim JY, Hwang I, et al. Analgesic effect of quetiapine in a mouse model of cancer-induced bone pain. Korean J Intern Med. 2017;32(6):1069-1074.
30. Tamburello AC, Lieberman JA, Baum RM, et al. Successful removal of quetiapine from a correctional formulary. J Am Acad Psychiatry Law. 2012;40(4):502-508.
31. Fountoulakis KN, Iacovides A, Kaprinis SG, et al. Diffuse muscle pain with quetiapine. Br J Psychiatry. 2003;182:81.
32. Shintani F. Diminished pain perception in schizophrenia. Lancet. 2010;376(9735):87.
33. Duric V, Banasr M, Franklin T, et al. Cariprazine exhibits anxiolytic and dopamine D3 receptor-dependent antidepressant effects in the chronic stress model. Int J Neuropsychopharmacol. 2017;20(10):788-796
Our understanding of pain mechanisms continues to evolve and, accordingly, so do our treatment strategies. The fundamental differences between acute and chronic pain were only recently recognized; this lack of recognition led to the application of acute pain treatments to chronic pain, contributing to the opioid epidemic in the United States.
With the diminishing emphasis on opioid medications, researchers are exploring other pharmacologic modalities for treating pain. Many nonopioid psychiatric medications are used off-label for the treatment of pain. Psychiatric medications play a larger role in the management of pain as pain becomes more chronic (Table 11). For simplicity, acute pain may be seen as nociception colored by emotions, and chronic pain as emotions colored by nociception. Protracted pain connects those extremes with a diminishing role of nociception and an increasing role of emotion,1 which may increase the potential role of psychiatric medications, including antipsychotics.

In this article, I discuss the potential role of dopamine in the perception of pain, and review the potential use of first- and second-generation antipsychotics for treating various pain syndromes.
Role of dopamine in pain
There is increasing interest in exploring antipsychotics to treat chronic pain2 because dopamine dysfunction is part of pathological pain perception. Excess dopamine is associated with headaches (dopamine hypersensitivity hypothesis3,4) and dopamine dysfunction is a part of posttraumatic stress disorder (PTSD),5 dissociation,6 paranoia,7 and catastrophizing.8 Somatic psychosis, like any psychosis, can be based on dopamine pathology. Dopaminergic neurons affect nociceptive function in the spinal dorsal horn,9 and dopamine receptors are altered in atypical facial pain,10 burning mouth syndrome,11 and fibromyalgia.12
In normal circumstances, dopamine is fundamentally a protective neurotransmitter. In acute pain, dopamine is powerfully released, making the pain bearable. A patient may describe acute pain as seeming “like it was not happening to me” or “it was like a dream”; both are examples of dopamine-caused dissociation and a possible prediction of subsequent chronification. In chronic pain, pathological mechanisms settle in and take root; therefore, keeping protective dopamine levels high becomes a priority. This is especially common in patients who have experienced abuse or PTSD. The only natural way to keep dopamine up for prolonged periods of time is to decrease pain and stress thresholds. Both phenomena are readily observed in patients with pain. In extreme cases, self-mutilation and involvement in conflicts become pathologically gratifying.
The dopaminergic system is essential for pain control with a tissue injury.13 It becomes pathologically stimulated and increasingly dysfunctional as algopathy (a pathological pain perception) develops. At the same time, a flood or drought of any neurotransmitter is equally bad and may produce similar clinical pictures. Both a lack of and excess of dopamine are associated with pain.14 This is why opposite treatments may be beneficial in different patients with chronic pain. As an example, the use of stimulants15 and bupropion16 has been reported in the treatment of abdominal pain. And, reversely, antipsychotics, especially first-generation agents, may be associated with chronic (tardive) pain, including orofacial and genital pain.17
First-generation antipsychotics
First-generation antipsychotics (FGAs) have been used to treat various nonpsychiatric conditions (Table 2). Although they are powerful D2 receptor inhibitors, FGAs lack the intrinsic ability to counteract the unwanted adverse effects of strong inhibition. As a result, movement disorders and prolactinemia are commonly induced by FGAs. The most dangerous consequence of treatment with these agents is neuroleptic malignant syndrome (NMS).
Continue to: Haloperidol
Haloperidol is prescribed widely by nonpsychiatrists, primarily to treat agitation. Intravenous haloperidol has been used for the abortive treatment of headaches.18 Paradoxically, IV haloperidol is less likely to induce extrapyramidal symptoms (EPS) than the oral formulation because of a more pronounced anticholinergic action in IV use. Haloperidol can help relieve gastroparesis and nausea, especially in IV administration,19 but prolonged oral administration is associated with unwanted movement problems and should be avoided.20
Chlorpromazine is more anticholinergic than haloperidol. It can be used in the abortive treatment of headaches (preferably via IV and IM administration), nausea, hiccups, porphyria, and serotonin syndrome, but it is very sedating and frequently produces hypotension, dangerous QT prolongation, and sensations of thought-blocking.21
Pimozide is reported to help with skin picking, trichotillomania, and somatic hallucinations.22
Droperidol, promethazine, and prochlorperazine are used off-label to treat nausea and headaches. Primary care clinicians may not be aware that these commonly used medications are antipsychotics. Similar to other FGAs, these 3 agents may produce NMS and tardive dyskinesia (TD). The same applies to the prokinetic drug metoclopramide.
Second-generation antipsychotics
Second-generation antipsychotics (SGAs) work with various serotonin receptors, offsetting and enhancing the antipsychotic function of dopamine blockade. This diminishes but does not eliminate EPS and the risk of TD. Fortunately, the risk of NMS is lower with SGAs than with FGAs. Many SGAs are FDA-approved for treating schizophrenia and other psychiatric disorders, and some have relevance for pain management (Table 3). Many SGAs help with depressive symptoms and are powerful mood stabilizers. As such, they may diminish central over-firing of dopaminergic and serotonergic neurons involved in the pain cascade, which in turn decreases pain transmission and perception. The downside is that in general, SGAs increase the risk of diabetes and hyperlipidemia.
Continue to: Risperidone
Risperidone was the second FDA-approved SGA. Pain practitioners primarily prescribe it for treatmeant-resistant headaches, but patients with fibromyalgia and those with phantom and thalamic pain also may respond. Because risperidone’s properties are similar to that of many FGAs, it may potently cause EPS, TD, and prolactinemia. Neuroleptic malignant syndrome also has been reported.23
Ziprasidone is frequently overlooked by clinicians who treat pain. Although ziprasidone may be sedating, it is powerful as both a preventive and abortive (in an IM formulation) agent for treatment-resistant headaches. This might be attributed to its effects on the 5HT9 receptor. It is approved for treating bipolar depression and has been prescribed to effectively treat anxiety. For patients receiving ziprasidone, QT prolongation needs to be monitored closely.24
Olanzapine was modeled after clozapine and is effective as a mood stabilizer and an antianxiety, antipsychotic, and sleep-promoting medication. It has a useful “mellowing” effect and helps with central pain syndrome management. Patients with fibromyalgia respond well; in some cases, patients with phantom and thalamic pain also respond. Among SGAs prescribed to treat chronic pain, olanzapine has the most published studies. However, the downside is the risk of severe weight gain and diabetes. Usually, if a patient is already overweight, they gain less, but these patients typically are concerned about any additional weight gain.25
Aripiprazole is a partial dopamine agonist. It increases dopamine function in the prefrontal cortex, and by doing so it possibly improves cognition, mental acuity, goal-oriented activity, and attention. At the same time, it decreases dopamine activity in the basal ganglia and limbic system, improving catastrophizing, paranoia, abnormal pain perception, and multiple homeostasis functions. This combination of effects can be invaluable for some patients, but depending on individual susceptibility, aripiprazole might be too activating (causing agitation and akathisia) or too sedating.26
Brexpiprazole is a relative of aripiprazole, but for some patients it is better tolerated, and compliance with this medication usually is good. It partially antagonizes the D2 and 5HT1A receptors while antagonizing the 5HT2A receptors (which decreases the dopamine release in the striatum) and mimics the mechanism of action of an antidepressant. Through alpha-1-adrenergic receptor antagonism, it reduces EPS. All these effects are also part of the mechanisms of action of quetiapine, clozapine, and iloperidone, but brexpiprazole is considered to be the most alpha-1 antagonistic, which is a mechanism of action of other potential pain-controlling medications such as clonidine and tizanidine. In patients with pain who have an overactive noradrenergic system, this property may be beneficial. Its major problem stems from cytochrome P450 2D6 (CYP2D6) enzyme-dependent metabolism, which causes an approximately 5-fold increase in brexpiprazole blood level in poor CYP2D6 metabolizers. Therefore, combining brexpiprazole with CYP2D6 inhibitors such as fluoxetine, paroxetine, and duloxetine would be unwise. Aripiprazole and brexpiprazole are less associated with diabetes and sexual adverse effects than many other SGAs.27
Continue to: Asenapine
Asenapine is an underutilized antipsychotic. Its mechanism of action spans multiple receptors and is less specific in individual receptor activity than other dopamine blockers. It is administered under the tongue due to poor absorption when swallowed, and its molecule has an anesthetic property that causes mouth and tongue numbness/paresthesia. This function may help patients with orofacial pain. Significant somnolence and weight gain (although less than with olanzapine) limit its use. Some patients cannot tolerate the taste.28
Quetiapine is prescribed rather frequently due to its significant antianxiety effect. It is also reported to be beneficial in pain control.29 Weight gain may be severe. In doses smaller than typically administered to patients with bipolar disorder or schizophrenia, quetiapine is widely prescribed off-label for sleep. In lower doses, it acts primarily as an antihistamine (hence the sedation), but at an increased dose it activates the adrenergic system, which offsets sedation. Quetiapine antagonizes H1 histamine and 5HT2
Cariprazine is typically well tolerated because of its benign metabolic profile. It does not increase the QT interval and is not sedating. Cariprazine is a D2 and D3 partial receptor agonist. This allows the medication to inhibit overstimulated dopamine receptors (a desirable effect in pain management) and induces them when the endogenous dopamine level is low (helping with cognition, volition, and attention). Pro-cognitive effects are always beneficial for patients with pain. Cariprazine produces less EPS due to more ventral striatum vs dorsal striatum activity. Mood improvement caused by this medication is attributed to its 5HT2A, 5HT2B, and 5HT2C inverse agonism, which modulates the serotonergic system. Cariprazine will likely have a positive future in pain management because it has shown efficacy in the chronic stress model.33
A complex condition
No single medication or group of medications may be exclusively relied on for treating patients with chronic pain. Identifying alternatives to opioids for treating pain brings more attention to centrally-acting medications that may aid in the stabilization of the nervous system, which can decrease pathological pain perception and help patients cope with chronic painful conditions.
Bottom Line
Antipsychotics may be a valuable asset in the treatment of chronic pain, offering a potential alternative to prescribing opioids for pain. More research is needed to identify specific ways of using dopamine blockade or dopamine enhancement to help patients with chronic pain.
Continue to: Related Resource
Related Resource
- Tripathi A. Antipsychotics for migraines, cluster headaches, and nausea. Current Psychiatry. 2013;12(2):E1-E4.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Bupropion • Wellbutrin, Zyban
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Clonidine • Catapres
Clozapine • Clozaril
Droperidol • Inapsine
Duloxetine • Cymbalta
Fluoxetine • Prozac
Haloperidol • Haldol
Iloperidone • Fanapt
Metoclopramide • Reglan
Olanzapine • Zyprexa
Paroxetine • Paxil
Pimozide • Orap
Prochlorperazine • Compazine
Promethazine • Phenergan
Quetiapine • Seroquel
Risperidone • Risperdal
Tizanidine • Zanaflex
Ziprasidone • Geodon
Our understanding of pain mechanisms continues to evolve and, accordingly, so do our treatment strategies. The fundamental differences between acute and chronic pain were only recently recognized; this lack of recognition led to the application of acute pain treatments to chronic pain, contributing to the opioid epidemic in the United States.
With the diminishing emphasis on opioid medications, researchers are exploring other pharmacologic modalities for treating pain. Many nonopioid psychiatric medications are used off-label for the treatment of pain. Psychiatric medications play a larger role in the management of pain as pain becomes more chronic (Table 11). For simplicity, acute pain may be seen as nociception colored by emotions, and chronic pain as emotions colored by nociception. Protracted pain connects those extremes with a diminishing role of nociception and an increasing role of emotion,1 which may increase the potential role of psychiatric medications, including antipsychotics.

In this article, I discuss the potential role of dopamine in the perception of pain, and review the potential use of first- and second-generation antipsychotics for treating various pain syndromes.
Role of dopamine in pain
There is increasing interest in exploring antipsychotics to treat chronic pain2 because dopamine dysfunction is part of pathological pain perception. Excess dopamine is associated with headaches (dopamine hypersensitivity hypothesis3,4) and dopamine dysfunction is a part of posttraumatic stress disorder (PTSD),5 dissociation,6 paranoia,7 and catastrophizing.8 Somatic psychosis, like any psychosis, can be based on dopamine pathology. Dopaminergic neurons affect nociceptive function in the spinal dorsal horn,9 and dopamine receptors are altered in atypical facial pain,10 burning mouth syndrome,11 and fibromyalgia.12
In normal circumstances, dopamine is fundamentally a protective neurotransmitter. In acute pain, dopamine is powerfully released, making the pain bearable. A patient may describe acute pain as seeming “like it was not happening to me” or “it was like a dream”; both are examples of dopamine-caused dissociation and a possible prediction of subsequent chronification. In chronic pain, pathological mechanisms settle in and take root; therefore, keeping protective dopamine levels high becomes a priority. This is especially common in patients who have experienced abuse or PTSD. The only natural way to keep dopamine up for prolonged periods of time is to decrease pain and stress thresholds. Both phenomena are readily observed in patients with pain. In extreme cases, self-mutilation and involvement in conflicts become pathologically gratifying.
The dopaminergic system is essential for pain control with a tissue injury.13 It becomes pathologically stimulated and increasingly dysfunctional as algopathy (a pathological pain perception) develops. At the same time, a flood or drought of any neurotransmitter is equally bad and may produce similar clinical pictures. Both a lack of and excess of dopamine are associated with pain.14 This is why opposite treatments may be beneficial in different patients with chronic pain. As an example, the use of stimulants15 and bupropion16 has been reported in the treatment of abdominal pain. And, reversely, antipsychotics, especially first-generation agents, may be associated with chronic (tardive) pain, including orofacial and genital pain.17
First-generation antipsychotics
First-generation antipsychotics (FGAs) have been used to treat various nonpsychiatric conditions (Table 2). Although they are powerful D2 receptor inhibitors, FGAs lack the intrinsic ability to counteract the unwanted adverse effects of strong inhibition. As a result, movement disorders and prolactinemia are commonly induced by FGAs. The most dangerous consequence of treatment with these agents is neuroleptic malignant syndrome (NMS).
Continue to: Haloperidol
Haloperidol is prescribed widely by nonpsychiatrists, primarily to treat agitation. Intravenous haloperidol has been used for the abortive treatment of headaches.18 Paradoxically, IV haloperidol is less likely to induce extrapyramidal symptoms (EPS) than the oral formulation because of a more pronounced anticholinergic action in IV use. Haloperidol can help relieve gastroparesis and nausea, especially in IV administration,19 but prolonged oral administration is associated with unwanted movement problems and should be avoided.20
Chlorpromazine is more anticholinergic than haloperidol. It can be used in the abortive treatment of headaches (preferably via IV and IM administration), nausea, hiccups, porphyria, and serotonin syndrome, but it is very sedating and frequently produces hypotension, dangerous QT prolongation, and sensations of thought-blocking.21
Pimozide is reported to help with skin picking, trichotillomania, and somatic hallucinations.22
Droperidol, promethazine, and prochlorperazine are used off-label to treat nausea and headaches. Primary care clinicians may not be aware that these commonly used medications are antipsychotics. Similar to other FGAs, these 3 agents may produce NMS and tardive dyskinesia (TD). The same applies to the prokinetic drug metoclopramide.
Second-generation antipsychotics
Second-generation antipsychotics (SGAs) work with various serotonin receptors, offsetting and enhancing the antipsychotic function of dopamine blockade. This diminishes but does not eliminate EPS and the risk of TD. Fortunately, the risk of NMS is lower with SGAs than with FGAs. Many SGAs are FDA-approved for treating schizophrenia and other psychiatric disorders, and some have relevance for pain management (Table 3). Many SGAs help with depressive symptoms and are powerful mood stabilizers. As such, they may diminish central over-firing of dopaminergic and serotonergic neurons involved in the pain cascade, which in turn decreases pain transmission and perception. The downside is that in general, SGAs increase the risk of diabetes and hyperlipidemia.
Continue to: Risperidone
Risperidone was the second FDA-approved SGA. Pain practitioners primarily prescribe it for treatmeant-resistant headaches, but patients with fibromyalgia and those with phantom and thalamic pain also may respond. Because risperidone’s properties are similar to that of many FGAs, it may potently cause EPS, TD, and prolactinemia. Neuroleptic malignant syndrome also has been reported.23
Ziprasidone is frequently overlooked by clinicians who treat pain. Although ziprasidone may be sedating, it is powerful as both a preventive and abortive (in an IM formulation) agent for treatment-resistant headaches. This might be attributed to its effects on the 5HT9 receptor. It is approved for treating bipolar depression and has been prescribed to effectively treat anxiety. For patients receiving ziprasidone, QT prolongation needs to be monitored closely.24
Olanzapine was modeled after clozapine and is effective as a mood stabilizer and an antianxiety, antipsychotic, and sleep-promoting medication. It has a useful “mellowing” effect and helps with central pain syndrome management. Patients with fibromyalgia respond well; in some cases, patients with phantom and thalamic pain also respond. Among SGAs prescribed to treat chronic pain, olanzapine has the most published studies. However, the downside is the risk of severe weight gain and diabetes. Usually, if a patient is already overweight, they gain less, but these patients typically are concerned about any additional weight gain.25
Aripiprazole is a partial dopamine agonist. It increases dopamine function in the prefrontal cortex, and by doing so it possibly improves cognition, mental acuity, goal-oriented activity, and attention. At the same time, it decreases dopamine activity in the basal ganglia and limbic system, improving catastrophizing, paranoia, abnormal pain perception, and multiple homeostasis functions. This combination of effects can be invaluable for some patients, but depending on individual susceptibility, aripiprazole might be too activating (causing agitation and akathisia) or too sedating.26
Brexpiprazole is a relative of aripiprazole, but for some patients it is better tolerated, and compliance with this medication usually is good. It partially antagonizes the D2 and 5HT1A receptors while antagonizing the 5HT2A receptors (which decreases the dopamine release in the striatum) and mimics the mechanism of action of an antidepressant. Through alpha-1-adrenergic receptor antagonism, it reduces EPS. All these effects are also part of the mechanisms of action of quetiapine, clozapine, and iloperidone, but brexpiprazole is considered to be the most alpha-1 antagonistic, which is a mechanism of action of other potential pain-controlling medications such as clonidine and tizanidine. In patients with pain who have an overactive noradrenergic system, this property may be beneficial. Its major problem stems from cytochrome P450 2D6 (CYP2D6) enzyme-dependent metabolism, which causes an approximately 5-fold increase in brexpiprazole blood level in poor CYP2D6 metabolizers. Therefore, combining brexpiprazole with CYP2D6 inhibitors such as fluoxetine, paroxetine, and duloxetine would be unwise. Aripiprazole and brexpiprazole are less associated with diabetes and sexual adverse effects than many other SGAs.27
Continue to: Asenapine
Asenapine is an underutilized antipsychotic. Its mechanism of action spans multiple receptors and is less specific in individual receptor activity than other dopamine blockers. It is administered under the tongue due to poor absorption when swallowed, and its molecule has an anesthetic property that causes mouth and tongue numbness/paresthesia. This function may help patients with orofacial pain. Significant somnolence and weight gain (although less than with olanzapine) limit its use. Some patients cannot tolerate the taste.28
Quetiapine is prescribed rather frequently due to its significant antianxiety effect. It is also reported to be beneficial in pain control.29 Weight gain may be severe. In doses smaller than typically administered to patients with bipolar disorder or schizophrenia, quetiapine is widely prescribed off-label for sleep. In lower doses, it acts primarily as an antihistamine (hence the sedation), but at an increased dose it activates the adrenergic system, which offsets sedation. Quetiapine antagonizes H1 histamine and 5HT2
Cariprazine is typically well tolerated because of its benign metabolic profile. It does not increase the QT interval and is not sedating. Cariprazine is a D2 and D3 partial receptor agonist. This allows the medication to inhibit overstimulated dopamine receptors (a desirable effect in pain management) and induces them when the endogenous dopamine level is low (helping with cognition, volition, and attention). Pro-cognitive effects are always beneficial for patients with pain. Cariprazine produces less EPS due to more ventral striatum vs dorsal striatum activity. Mood improvement caused by this medication is attributed to its 5HT2A, 5HT2B, and 5HT2C inverse agonism, which modulates the serotonergic system. Cariprazine will likely have a positive future in pain management because it has shown efficacy in the chronic stress model.33
A complex condition
No single medication or group of medications may be exclusively relied on for treating patients with chronic pain. Identifying alternatives to opioids for treating pain brings more attention to centrally-acting medications that may aid in the stabilization of the nervous system, which can decrease pathological pain perception and help patients cope with chronic painful conditions.
Bottom Line
Antipsychotics may be a valuable asset in the treatment of chronic pain, offering a potential alternative to prescribing opioids for pain. More research is needed to identify specific ways of using dopamine blockade or dopamine enhancement to help patients with chronic pain.
Continue to: Related Resource
Related Resource
- Tripathi A. Antipsychotics for migraines, cluster headaches, and nausea. Current Psychiatry. 2013;12(2):E1-E4.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Bupropion • Wellbutrin, Zyban
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Clonidine • Catapres
Clozapine • Clozaril
Droperidol • Inapsine
Duloxetine • Cymbalta
Fluoxetine • Prozac
Haloperidol • Haldol
Iloperidone • Fanapt
Metoclopramide • Reglan
Olanzapine • Zyprexa
Paroxetine • Paxil
Pimozide • Orap
Prochlorperazine • Compazine
Promethazine • Phenergan
Quetiapine • Seroquel
Risperidone • Risperdal
Tizanidine • Zanaflex
Ziprasidone • Geodon
1. Arbuck D, Pergolizzi J. Algopathy—acknowledging the pathological process of pain chronification. Pract Pain Manag. 2017;17(4):4,26-32.
2. Shin SW, Lee JS, Abdi S, et al. Antipsychotics for patients with pain. Korean J Pain. 2019;32(1):3-11.
3. D’Andrea G, Leone M, Bussone G, et al. Abnormal tyrosine metabolism in chronic cluster headache. Cephalalgia. 2017;37(2):148-153.
4. D’Andrea G, Granella F, Perini F, et al. Platelet levels of dopamine are increased in migraine and cluster headache. Headache. 2006;46(4):585-591.
5. Wolf EJ, Mitchell KS, Logue MW, et al. The dopamine D3 receptor gene, and posttraumatic stress disorder. J Trauma Stress. 2014;27(4):379-387.
6. den Ouden HEM, Daw ND, Fernandez G, et al. Dissociable effects of dopamine and serotonin on reversal learning. Neuron. 2013;80(4):1090-1100.
7. Nour MM, Dahoun T, Schwartenbeck P, et al. Dopaminergic basis for signaling belief updates, but not surprise, and the link to paranoia. Proc Natl Acad Sci U S A. 2018;115(43):E10167-E10176.
8. Zhu H, Clemens S, Sawchuk M, et al. Expression and distribution of all dopamine receptor subtypes (D(1)-D(5)) in the mouse lumbar spinal cord: a real-time polymerase chain reaction and non-autoradiographic in situ hybridization study. Neuroscience. 2007;149:885-897.
9. Wood PB, Schweinhardt P, Jaeger E, et al. Fibromyalgia patients show an abnormal dopamine response to pain. Eur J Neurosci. 2007;25:3576-3582.
10. Hagelberg N, Fossell H, Aalto S, et al. Altered dopamine D2 receptor binding in atypical facial pain. Pain. 2003;106(1-2):43-48.
11. Hagelberg N, Fossell H, Rinne JD, et al. Striatal dopamine D1 and D2 receptors in burning mouth syndrome. Pain. 2003;101(1-2):149-154.
12. Elman I, Borsook D. Common brain mechanisms of chronic pain and addiction. Neuron. 2016;89(1):11-36.
13. Siahposht-Khachaki A, Pourreza P, Ezzatpanah S, et al. Nucleus accumbens dopamine receptors mediate hypothalamus-induced antinociception in the rat formalin test. Eur J Pain. 2017;21(7):1285-1294.
14. Thompson T, Gallop K, Correll CU, et al. Pain perception in Parkinson’s disease: a systematic review and meta-analysis of experimental studies. Aging Res Rev. 2017;35:74-86.
15. Check JH. Chronic unremitting lower abdominal pain quickly abrogated following treatment with amphetamine. Clin Exp Obstet Gynecol. 2016;43(1):109-111.
16. Wilkes S. Bupropion. Drugs Today (Barc). 2006;42(10):671-681.
17. Frei K, Truong DD, Fahn S, et al. The nosology of tardive syndromes. J Neurol Sci. 2018;389:10-16.
18. Honkaniemi J, Liimatainen S, Rainesalo S, et al. Haloperidol in the acute treatment of migraine: a randomized, double-blind, placebo-controlled study. Headache. 2006;46(5):781-787.
19. Murray-Brown F, Dorman S. Haloperidol for the treatment of nausea and vomiting in palliative care patients. Cochrane Database Syst Rev. 2015;(11):CD006271.
20. Gaffigan ME, Bruner DI, Wason C, et al. A randomized controlled trial of intravenous haloperidol vs. intravenous metoclopramide for acute migraine therapy in the emergency department. J Emerg Med. 2015;49(3):326-334.
21. Weinman D, Nicastro O, Akala O, et al. Parenteral treatment of episodic tension-type headache: a systematic review. Headache. 2014;54(2):260-268.
22. Arnold LM, Auchenbach MB, McElroy SL. Psychogenic excoriation. Clinical features, proposed diagnostic criteria, epidemiology, and approaches to treatment. CNS Drugs. 2001;15(5):351-359.
23. Khouzam HR. Psychopharmacology of chronic pain: a focus on antidepressants and atypical antipsychotics. Postgrad Med. 2016;128(3):323-330.
24. Landsness EC, Wang LH, Bucelli RC. Ziprasidone as a potential abortive therapy for status migrainosus. Neurohospitalist. 2016;6(4):151-156.
25. Jimenez XF, Sundararajan T, Covington EC. A systematic review of atypical antipsychotics in chronic pain management: olanzapine demonstrates potential in central sensitization, fibromyalgia, and headache/migraine. Clin J Pain. 2018;34(6):585-591.
26. Fei L, Abrardi L, Mediati RD. Unexpected effect of aripiprazole on nociceptive pain. Ther Adv Psychopharmacol. 2012;2(5):211-212.
27. Markovic M, Gallipani A, Patel KH, et al. Brexpiprazole. Ann Pharmacother. 2017;51(4):315-322.
28. Gerrits M, de Greef R, Peeters P. Effect of absorption site on the pharmacokinetics of sublingual asenapine in healthy male subjects. Biopharm Drug Dispos. 2010;31(5-6):351-357.
29. Heo MH, Kim JY, Hwang I, et al. Analgesic effect of quetiapine in a mouse model of cancer-induced bone pain. Korean J Intern Med. 2017;32(6):1069-1074.
30. Tamburello AC, Lieberman JA, Baum RM, et al. Successful removal of quetiapine from a correctional formulary. J Am Acad Psychiatry Law. 2012;40(4):502-508.
31. Fountoulakis KN, Iacovides A, Kaprinis SG, et al. Diffuse muscle pain with quetiapine. Br J Psychiatry. 2003;182:81.
32. Shintani F. Diminished pain perception in schizophrenia. Lancet. 2010;376(9735):87.
33. Duric V, Banasr M, Franklin T, et al. Cariprazine exhibits anxiolytic and dopamine D3 receptor-dependent antidepressant effects in the chronic stress model. Int J Neuropsychopharmacol. 2017;20(10):788-796
1. Arbuck D, Pergolizzi J. Algopathy—acknowledging the pathological process of pain chronification. Pract Pain Manag. 2017;17(4):4,26-32.
2. Shin SW, Lee JS, Abdi S, et al. Antipsychotics for patients with pain. Korean J Pain. 2019;32(1):3-11.
3. D’Andrea G, Leone M, Bussone G, et al. Abnormal tyrosine metabolism in chronic cluster headache. Cephalalgia. 2017;37(2):148-153.
4. D’Andrea G, Granella F, Perini F, et al. Platelet levels of dopamine are increased in migraine and cluster headache. Headache. 2006;46(4):585-591.
5. Wolf EJ, Mitchell KS, Logue MW, et al. The dopamine D3 receptor gene, and posttraumatic stress disorder. J Trauma Stress. 2014;27(4):379-387.
6. den Ouden HEM, Daw ND, Fernandez G, et al. Dissociable effects of dopamine and serotonin on reversal learning. Neuron. 2013;80(4):1090-1100.
7. Nour MM, Dahoun T, Schwartenbeck P, et al. Dopaminergic basis for signaling belief updates, but not surprise, and the link to paranoia. Proc Natl Acad Sci U S A. 2018;115(43):E10167-E10176.
8. Zhu H, Clemens S, Sawchuk M, et al. Expression and distribution of all dopamine receptor subtypes (D(1)-D(5)) in the mouse lumbar spinal cord: a real-time polymerase chain reaction and non-autoradiographic in situ hybridization study. Neuroscience. 2007;149:885-897.
9. Wood PB, Schweinhardt P, Jaeger E, et al. Fibromyalgia patients show an abnormal dopamine response to pain. Eur J Neurosci. 2007;25:3576-3582.
10. Hagelberg N, Fossell H, Aalto S, et al. Altered dopamine D2 receptor binding in atypical facial pain. Pain. 2003;106(1-2):43-48.
11. Hagelberg N, Fossell H, Rinne JD, et al. Striatal dopamine D1 and D2 receptors in burning mouth syndrome. Pain. 2003;101(1-2):149-154.
12. Elman I, Borsook D. Common brain mechanisms of chronic pain and addiction. Neuron. 2016;89(1):11-36.
13. Siahposht-Khachaki A, Pourreza P, Ezzatpanah S, et al. Nucleus accumbens dopamine receptors mediate hypothalamus-induced antinociception in the rat formalin test. Eur J Pain. 2017;21(7):1285-1294.
14. Thompson T, Gallop K, Correll CU, et al. Pain perception in Parkinson’s disease: a systematic review and meta-analysis of experimental studies. Aging Res Rev. 2017;35:74-86.
15. Check JH. Chronic unremitting lower abdominal pain quickly abrogated following treatment with amphetamine. Clin Exp Obstet Gynecol. 2016;43(1):109-111.
16. Wilkes S. Bupropion. Drugs Today (Barc). 2006;42(10):671-681.
17. Frei K, Truong DD, Fahn S, et al. The nosology of tardive syndromes. J Neurol Sci. 2018;389:10-16.
18. Honkaniemi J, Liimatainen S, Rainesalo S, et al. Haloperidol in the acute treatment of migraine: a randomized, double-blind, placebo-controlled study. Headache. 2006;46(5):781-787.
19. Murray-Brown F, Dorman S. Haloperidol for the treatment of nausea and vomiting in palliative care patients. Cochrane Database Syst Rev. 2015;(11):CD006271.
20. Gaffigan ME, Bruner DI, Wason C, et al. A randomized controlled trial of intravenous haloperidol vs. intravenous metoclopramide for acute migraine therapy in the emergency department. J Emerg Med. 2015;49(3):326-334.
21. Weinman D, Nicastro O, Akala O, et al. Parenteral treatment of episodic tension-type headache: a systematic review. Headache. 2014;54(2):260-268.
22. Arnold LM, Auchenbach MB, McElroy SL. Psychogenic excoriation. Clinical features, proposed diagnostic criteria, epidemiology, and approaches to treatment. CNS Drugs. 2001;15(5):351-359.
23. Khouzam HR. Psychopharmacology of chronic pain: a focus on antidepressants and atypical antipsychotics. Postgrad Med. 2016;128(3):323-330.
24. Landsness EC, Wang LH, Bucelli RC. Ziprasidone as a potential abortive therapy for status migrainosus. Neurohospitalist. 2016;6(4):151-156.
25. Jimenez XF, Sundararajan T, Covington EC. A systematic review of atypical antipsychotics in chronic pain management: olanzapine demonstrates potential in central sensitization, fibromyalgia, and headache/migraine. Clin J Pain. 2018;34(6):585-591.
26. Fei L, Abrardi L, Mediati RD. Unexpected effect of aripiprazole on nociceptive pain. Ther Adv Psychopharmacol. 2012;2(5):211-212.
27. Markovic M, Gallipani A, Patel KH, et al. Brexpiprazole. Ann Pharmacother. 2017;51(4):315-322.
28. Gerrits M, de Greef R, Peeters P. Effect of absorption site on the pharmacokinetics of sublingual asenapine in healthy male subjects. Biopharm Drug Dispos. 2010;31(5-6):351-357.
29. Heo MH, Kim JY, Hwang I, et al. Analgesic effect of quetiapine in a mouse model of cancer-induced bone pain. Korean J Intern Med. 2017;32(6):1069-1074.
30. Tamburello AC, Lieberman JA, Baum RM, et al. Successful removal of quetiapine from a correctional formulary. J Am Acad Psychiatry Law. 2012;40(4):502-508.
31. Fountoulakis KN, Iacovides A, Kaprinis SG, et al. Diffuse muscle pain with quetiapine. Br J Psychiatry. 2003;182:81.
32. Shintani F. Diminished pain perception in schizophrenia. Lancet. 2010;376(9735):87.
33. Duric V, Banasr M, Franklin T, et al. Cariprazine exhibits anxiolytic and dopamine D3 receptor-dependent antidepressant effects in the chronic stress model. Int J Neuropsychopharmacol. 2017;20(10):788-796
FDA approves Caplyta to treat schizophrenia in adults
The Food and Drug Administration has approved lumateperone for the treatment of schizophrenia in adults.
Lumateperone, an atypical antipsychotic, will be marketed by Intra-Cellular Therapies as Caplyta, according to a Dec. 23 statement from the company.
In the first, 335 patients with schizophrenia were randomized to two doses of lumateperone, an active comparator, or placebo. Those randomized to the approved 42-mg dose of lumateperone showed a statistically significant reduction in total score on the Positive and Negative Syndrome Scale (PANSS), compared with patients in the other groups. The median age in this study was 42 years; 17% of patients were female, 19% were white, and 78% were black.
In the second study, 450 patients diagnosed with schizophrenia were randomized in a double-blind fashion to one of two doses of lumateperone or placebo. Patients taking the approved dose showed a statistically significant reduction from baseline to day 28 in PANSS total score. In this study, patients’ median age was 44 years; 23% were female, 26% were white, and 66% were black.
Treatment with lumateperone appears to feature a more favorable cardiometabolic profile than that of other approved antipsychotic agents.
Patients treated with lumateperone for at least a year showed significant reductions in LDL cholesterol, total cholesterol, serum prolactin, and body weight, compared with baseline values recorded when participants were on various standard-of-care antipsychotics prior to switching, Suresh Durgam, MD, reported at the annual congress of the European College of Neuropsychopharmacology. Other cardiometabolic parameters, including fasting blood glucose, insulin, triglycerides, and HDL cholesterol, showed only negligible change over the course of study, according to Dr. Durgam, a psychiatrist and senior vice president for late-stage clinical development and medical affairs at Intra-Cellular Therapies.
The most common adverse events with lumateperone were somnolence (24% vs. 10% on placebo) and dry mouth (6% vs. 2%).
Approval of lumateperone hit a snag last summer when the FDA canceled the Psychopharmacologic Drugs Advisory Committee meeting it previously had called for to review the new drug application for lumateperone. The agency said the meeting was canceled because of “new information regarding the application.”
At the time, Intra-Cellular Therapies noted that it had provided additional information to the FDA to meet agency requests. This information was related to nonclinical studies.
“The FDA canceled the advisory committee meeting to allow sufficient time to review this new and any forthcoming information as they continue” to review the new drug application for lumateperone, Intra-Cellular said a statement.
The company plans to launch Caplyta late in the first quarter of 2020.
Bruce Jancin and Kerry Dooley Young contributed to this report.
The Food and Drug Administration has approved lumateperone for the treatment of schizophrenia in adults.
Lumateperone, an atypical antipsychotic, will be marketed by Intra-Cellular Therapies as Caplyta, according to a Dec. 23 statement from the company.
In the first, 335 patients with schizophrenia were randomized to two doses of lumateperone, an active comparator, or placebo. Those randomized to the approved 42-mg dose of lumateperone showed a statistically significant reduction in total score on the Positive and Negative Syndrome Scale (PANSS), compared with patients in the other groups. The median age in this study was 42 years; 17% of patients were female, 19% were white, and 78% were black.
In the second study, 450 patients diagnosed with schizophrenia were randomized in a double-blind fashion to one of two doses of lumateperone or placebo. Patients taking the approved dose showed a statistically significant reduction from baseline to day 28 in PANSS total score. In this study, patients’ median age was 44 years; 23% were female, 26% were white, and 66% were black.
Treatment with lumateperone appears to feature a more favorable cardiometabolic profile than that of other approved antipsychotic agents.
Patients treated with lumateperone for at least a year showed significant reductions in LDL cholesterol, total cholesterol, serum prolactin, and body weight, compared with baseline values recorded when participants were on various standard-of-care antipsychotics prior to switching, Suresh Durgam, MD, reported at the annual congress of the European College of Neuropsychopharmacology. Other cardiometabolic parameters, including fasting blood glucose, insulin, triglycerides, and HDL cholesterol, showed only negligible change over the course of study, according to Dr. Durgam, a psychiatrist and senior vice president for late-stage clinical development and medical affairs at Intra-Cellular Therapies.
The most common adverse events with lumateperone were somnolence (24% vs. 10% on placebo) and dry mouth (6% vs. 2%).
Approval of lumateperone hit a snag last summer when the FDA canceled the Psychopharmacologic Drugs Advisory Committee meeting it previously had called for to review the new drug application for lumateperone. The agency said the meeting was canceled because of “new information regarding the application.”
At the time, Intra-Cellular Therapies noted that it had provided additional information to the FDA to meet agency requests. This information was related to nonclinical studies.
“The FDA canceled the advisory committee meeting to allow sufficient time to review this new and any forthcoming information as they continue” to review the new drug application for lumateperone, Intra-Cellular said a statement.
The company plans to launch Caplyta late in the first quarter of 2020.
Bruce Jancin and Kerry Dooley Young contributed to this report.
The Food and Drug Administration has approved lumateperone for the treatment of schizophrenia in adults.
Lumateperone, an atypical antipsychotic, will be marketed by Intra-Cellular Therapies as Caplyta, according to a Dec. 23 statement from the company.
In the first, 335 patients with schizophrenia were randomized to two doses of lumateperone, an active comparator, or placebo. Those randomized to the approved 42-mg dose of lumateperone showed a statistically significant reduction in total score on the Positive and Negative Syndrome Scale (PANSS), compared with patients in the other groups. The median age in this study was 42 years; 17% of patients were female, 19% were white, and 78% were black.
In the second study, 450 patients diagnosed with schizophrenia were randomized in a double-blind fashion to one of two doses of lumateperone or placebo. Patients taking the approved dose showed a statistically significant reduction from baseline to day 28 in PANSS total score. In this study, patients’ median age was 44 years; 23% were female, 26% were white, and 66% were black.
Treatment with lumateperone appears to feature a more favorable cardiometabolic profile than that of other approved antipsychotic agents.
Patients treated with lumateperone for at least a year showed significant reductions in LDL cholesterol, total cholesterol, serum prolactin, and body weight, compared with baseline values recorded when participants were on various standard-of-care antipsychotics prior to switching, Suresh Durgam, MD, reported at the annual congress of the European College of Neuropsychopharmacology. Other cardiometabolic parameters, including fasting blood glucose, insulin, triglycerides, and HDL cholesterol, showed only negligible change over the course of study, according to Dr. Durgam, a psychiatrist and senior vice president for late-stage clinical development and medical affairs at Intra-Cellular Therapies.
The most common adverse events with lumateperone were somnolence (24% vs. 10% on placebo) and dry mouth (6% vs. 2%).
Approval of lumateperone hit a snag last summer when the FDA canceled the Psychopharmacologic Drugs Advisory Committee meeting it previously had called for to review the new drug application for lumateperone. The agency said the meeting was canceled because of “new information regarding the application.”
At the time, Intra-Cellular Therapies noted that it had provided additional information to the FDA to meet agency requests. This information was related to nonclinical studies.
“The FDA canceled the advisory committee meeting to allow sufficient time to review this new and any forthcoming information as they continue” to review the new drug application for lumateperone, Intra-Cellular said a statement.
The company plans to launch Caplyta late in the first quarter of 2020.
Bruce Jancin and Kerry Dooley Young contributed to this report.
Schizophrenia, bipolar disorder associated with increased risk of secondary TD
Psychiatric inpatients, particularly those with schizophrenia or bipolar disorder, have both a greater risk of having a secondary diagnosis of tardive dyskinesia and having worse illness when tardive dyskinesia is also present, according to results of a case-control study of more than 77,000 inpatients.
For the study, the investigators conducted an analysis of 77,022 adults from the Nationwide Inpatient Sample who had been admitted between January 2010 and December 2014 for mood disorders and schizophrenia; 38,382 patients in this group also had a secondary diagnosis of tardive dyskinesia (TD), reported Rikinkumar S. Patel, MD, of the department of psychiatry at Griffin Memorial Hospital in Norman, Okla., and associates. The study was published in Heliyon.
They investigators found that patients with schizophrenia and bipolar disorder were four to five times more likely to also have TD, and patients with TD were six times more likely to have severe morbidity because of a major loss of function. Compared with non-TD controls, patients with TD had a longer hospital length of stay by 6.36 days and higher cost by $20,415.
More than 60% of TD patients came from below the 50th percentile in median household income, compared with less than 40% of the non-TD group. Dr. Patel and associates also found that almost half of the patients with TD were aged 40-60 years and that the prevalence of TD in the study population increased with age.
“Our findings support the previous evidence that advanced age is a risk factor for the development of TD,” they wrote, citing research by Criscely L. Go, MD, and associates (Parkinsonism Relat Disord. 2019. 15[9]:655-9).
Dr. Patel and associates concluded that more systematic research is needed to prevent TD and “optimize inpatient outcomes in psychiatric patients with TD.”
The study authors reported having no conflicts of interest.
SOURCE: Patel RS et al. Heliyon. 2019. doi: 10.1016/j.heliyon.2019.e01745.
Psychiatric inpatients, particularly those with schizophrenia or bipolar disorder, have both a greater risk of having a secondary diagnosis of tardive dyskinesia and having worse illness when tardive dyskinesia is also present, according to results of a case-control study of more than 77,000 inpatients.
For the study, the investigators conducted an analysis of 77,022 adults from the Nationwide Inpatient Sample who had been admitted between January 2010 and December 2014 for mood disorders and schizophrenia; 38,382 patients in this group also had a secondary diagnosis of tardive dyskinesia (TD), reported Rikinkumar S. Patel, MD, of the department of psychiatry at Griffin Memorial Hospital in Norman, Okla., and associates. The study was published in Heliyon.
They investigators found that patients with schizophrenia and bipolar disorder were four to five times more likely to also have TD, and patients with TD were six times more likely to have severe morbidity because of a major loss of function. Compared with non-TD controls, patients with TD had a longer hospital length of stay by 6.36 days and higher cost by $20,415.
More than 60% of TD patients came from below the 50th percentile in median household income, compared with less than 40% of the non-TD group. Dr. Patel and associates also found that almost half of the patients with TD were aged 40-60 years and that the prevalence of TD in the study population increased with age.
“Our findings support the previous evidence that advanced age is a risk factor for the development of TD,” they wrote, citing research by Criscely L. Go, MD, and associates (Parkinsonism Relat Disord. 2019. 15[9]:655-9).
Dr. Patel and associates concluded that more systematic research is needed to prevent TD and “optimize inpatient outcomes in psychiatric patients with TD.”
The study authors reported having no conflicts of interest.
SOURCE: Patel RS et al. Heliyon. 2019. doi: 10.1016/j.heliyon.2019.e01745.
Psychiatric inpatients, particularly those with schizophrenia or bipolar disorder, have both a greater risk of having a secondary diagnosis of tardive dyskinesia and having worse illness when tardive dyskinesia is also present, according to results of a case-control study of more than 77,000 inpatients.
For the study, the investigators conducted an analysis of 77,022 adults from the Nationwide Inpatient Sample who had been admitted between January 2010 and December 2014 for mood disorders and schizophrenia; 38,382 patients in this group also had a secondary diagnosis of tardive dyskinesia (TD), reported Rikinkumar S. Patel, MD, of the department of psychiatry at Griffin Memorial Hospital in Norman, Okla., and associates. The study was published in Heliyon.
They investigators found that patients with schizophrenia and bipolar disorder were four to five times more likely to also have TD, and patients with TD were six times more likely to have severe morbidity because of a major loss of function. Compared with non-TD controls, patients with TD had a longer hospital length of stay by 6.36 days and higher cost by $20,415.
More than 60% of TD patients came from below the 50th percentile in median household income, compared with less than 40% of the non-TD group. Dr. Patel and associates also found that almost half of the patients with TD were aged 40-60 years and that the prevalence of TD in the study population increased with age.
“Our findings support the previous evidence that advanced age is a risk factor for the development of TD,” they wrote, citing research by Criscely L. Go, MD, and associates (Parkinsonism Relat Disord. 2019. 15[9]:655-9).
Dr. Patel and associates concluded that more systematic research is needed to prevent TD and “optimize inpatient outcomes in psychiatric patients with TD.”
The study authors reported having no conflicts of interest.
SOURCE: Patel RS et al. Heliyon. 2019. doi: 10.1016/j.heliyon.2019.e01745.
FROM HELIYON
Sequential intercept model is really a ‘no-intercept model’
Ultimately, psychiatrists must take responsibility for complex patients.
In legal settings, the “sequential intercept model” for targeting people involved in the criminal justice system with mental illness has been proposed as an improvement for the status quo.
The model intends to divert individuals with mental illnesses at any one of five described stages in their journey through the legal system. In the first stage, a patient may be provided enough care in the community to never enter the criminal system. If that works, the patient may be diverted by first responders out of the legal system and back into treatment. Sequentially, throughout the remaining stages, the patient can be diverted by an attorney, the court, a presentencing correctional facility, the sentencing judge, a postsentencing correctional facility, or probation. The model rightfully encourages anyone in the continuum of care to take ownership of a situation and intervene.
I applaud the model for encouraging all participants to intervene in changing the course of our most challenging patients. However, I am reminded of the complexity of large systems trying to change. In practice, what I have seen is a series of half-hearted recommendations: Emergency responders who consider their role finished after giving a patient the number of the suicide hotline, attorneys who are satisfied by giving their clients an outdated list of community mental health clinics, judges who interpret their recommendations for treatment as a fait accompli, and correctional facilities that release patients with an absurdly short supply of medications and the address of an emergency room. I worry that by creating a model encouraging all to participate, we have just absolved ones who make any effort, even if inadequate.
In some ways, the sequential intercept model has similarities with modern mental health treatment teams. In many settings, a treatment team includes a series of providers who are sequentially involved in the life of a patient. A team can include a psychiatrist for psychopharmacology; a neuropsychologist for psychological testing; a social worker for psychotherapeutic strategies; another social worker to assist in obtaining social assistance; an addiction counselor for substance use disorder; another psychiatrist who monitors the administration of a single medication, like ketamine; and a pharmacist who approves the medication regimen. That’s several providers for the treatment of one patient.
As a forensic psychiatrist, I am often asked to review treatment plans of other providers. I am asked to comment on the appropriate nature of a given treatment. Often, insurance companies want to review the continued need for treatment or whether any treatment is warranted at all. Sometimes, employers want to review a treatment plan to ensure the safety of their employees. At times, courts will ask for a review and expectations from treatment of a defendant to assist in sentencing determinations. However, I have not yet been asked by anyone if the amount of care a patient is obtaining is too fragmented and without any clear leadership.
In our endless pursuit of medicalization and standardization of mental health, we have, especially in large systems, created specialization silos for the care of our patients. Many, if not most psychiatrists, do not participate in any psychotherapy; social workers and psychologists do not prescribe (for the most part); many substance abuse counselors only address sobriety and not other primary mental illness factors; and pharmacists cannot diagnose nor are they trained in psychosocial approaches. In many ways, we have defined participants not by what they do, but what they don’t do.
One also can be saddened by the enormous logistical complexity imposed on patients required to make numerous appointments, which can deprive them of time for recovery. However, my bigger concern is that the multiplicity of providers also permits the dissolution of accountability. In my experience, those large teams have an ability to deflect responsibility in ways that are unmatched by any single provider who cannot rely on putting the fault on someone else.
Sadly and ironically, those two parallel paradigms of mental illness and criminal care impose those problems on each other by averting any attempt at interception, a “no-intercept model.” Mental health programs will deny clients involved in the criminal justice system for requiring too much treatment, too little treatment, for lack of availability of one of the necessary providers, for requiring substance use treatment, or simply for being part of the criminal justice system. Accordingly, the legal system will fail to accept recommendations by mental health providers that mental health treatment is not paramount at this time and that the defendant would be better served by addressing his criminogenic risk factors. In response, the multitude of participants in the legal system will point to the mental health system for all answers.
Contrary to many if not most problems, I do not think that the solution lies somewhere in the middle, as this would require the five stages of the legal system to compromise with the nine hypothetical participants of the mental health system. For our part, as psychiatrists, we must accept that we are ultimately responsible for all levels of care. As a field, we are also responsible for educating the public and the legal system of our role and limitations in providing care as well as being available for providing such care. Correspondingly, the legal system is responsible for putting an adequate effort into diverting patients and having or obtaining adequate understanding of available and appropriate care for their defendants.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Among his writings is chapter 7 in the new book “Critical Psychiatry: Controversies and Clinical Implications” (Springer, 2019).
Ultimately, psychiatrists must take responsibility for complex patients.
Ultimately, psychiatrists must take responsibility for complex patients.
In legal settings, the “sequential intercept model” for targeting people involved in the criminal justice system with mental illness has been proposed as an improvement for the status quo.
The model intends to divert individuals with mental illnesses at any one of five described stages in their journey through the legal system. In the first stage, a patient may be provided enough care in the community to never enter the criminal system. If that works, the patient may be diverted by first responders out of the legal system and back into treatment. Sequentially, throughout the remaining stages, the patient can be diverted by an attorney, the court, a presentencing correctional facility, the sentencing judge, a postsentencing correctional facility, or probation. The model rightfully encourages anyone in the continuum of care to take ownership of a situation and intervene.
I applaud the model for encouraging all participants to intervene in changing the course of our most challenging patients. However, I am reminded of the complexity of large systems trying to change. In practice, what I have seen is a series of half-hearted recommendations: Emergency responders who consider their role finished after giving a patient the number of the suicide hotline, attorneys who are satisfied by giving their clients an outdated list of community mental health clinics, judges who interpret their recommendations for treatment as a fait accompli, and correctional facilities that release patients with an absurdly short supply of medications and the address of an emergency room. I worry that by creating a model encouraging all to participate, we have just absolved ones who make any effort, even if inadequate.
In some ways, the sequential intercept model has similarities with modern mental health treatment teams. In many settings, a treatment team includes a series of providers who are sequentially involved in the life of a patient. A team can include a psychiatrist for psychopharmacology; a neuropsychologist for psychological testing; a social worker for psychotherapeutic strategies; another social worker to assist in obtaining social assistance; an addiction counselor for substance use disorder; another psychiatrist who monitors the administration of a single medication, like ketamine; and a pharmacist who approves the medication regimen. That’s several providers for the treatment of one patient.
As a forensic psychiatrist, I am often asked to review treatment plans of other providers. I am asked to comment on the appropriate nature of a given treatment. Often, insurance companies want to review the continued need for treatment or whether any treatment is warranted at all. Sometimes, employers want to review a treatment plan to ensure the safety of their employees. At times, courts will ask for a review and expectations from treatment of a defendant to assist in sentencing determinations. However, I have not yet been asked by anyone if the amount of care a patient is obtaining is too fragmented and without any clear leadership.
In our endless pursuit of medicalization and standardization of mental health, we have, especially in large systems, created specialization silos for the care of our patients. Many, if not most psychiatrists, do not participate in any psychotherapy; social workers and psychologists do not prescribe (for the most part); many substance abuse counselors only address sobriety and not other primary mental illness factors; and pharmacists cannot diagnose nor are they trained in psychosocial approaches. In many ways, we have defined participants not by what they do, but what they don’t do.
One also can be saddened by the enormous logistical complexity imposed on patients required to make numerous appointments, which can deprive them of time for recovery. However, my bigger concern is that the multiplicity of providers also permits the dissolution of accountability. In my experience, those large teams have an ability to deflect responsibility in ways that are unmatched by any single provider who cannot rely on putting the fault on someone else.
Sadly and ironically, those two parallel paradigms of mental illness and criminal care impose those problems on each other by averting any attempt at interception, a “no-intercept model.” Mental health programs will deny clients involved in the criminal justice system for requiring too much treatment, too little treatment, for lack of availability of one of the necessary providers, for requiring substance use treatment, or simply for being part of the criminal justice system. Accordingly, the legal system will fail to accept recommendations by mental health providers that mental health treatment is not paramount at this time and that the defendant would be better served by addressing his criminogenic risk factors. In response, the multitude of participants in the legal system will point to the mental health system for all answers.
Contrary to many if not most problems, I do not think that the solution lies somewhere in the middle, as this would require the five stages of the legal system to compromise with the nine hypothetical participants of the mental health system. For our part, as psychiatrists, we must accept that we are ultimately responsible for all levels of care. As a field, we are also responsible for educating the public and the legal system of our role and limitations in providing care as well as being available for providing such care. Correspondingly, the legal system is responsible for putting an adequate effort into diverting patients and having or obtaining adequate understanding of available and appropriate care for their defendants.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Among his writings is chapter 7 in the new book “Critical Psychiatry: Controversies and Clinical Implications” (Springer, 2019).
In legal settings, the “sequential intercept model” for targeting people involved in the criminal justice system with mental illness has been proposed as an improvement for the status quo.
The model intends to divert individuals with mental illnesses at any one of five described stages in their journey through the legal system. In the first stage, a patient may be provided enough care in the community to never enter the criminal system. If that works, the patient may be diverted by first responders out of the legal system and back into treatment. Sequentially, throughout the remaining stages, the patient can be diverted by an attorney, the court, a presentencing correctional facility, the sentencing judge, a postsentencing correctional facility, or probation. The model rightfully encourages anyone in the continuum of care to take ownership of a situation and intervene.
I applaud the model for encouraging all participants to intervene in changing the course of our most challenging patients. However, I am reminded of the complexity of large systems trying to change. In practice, what I have seen is a series of half-hearted recommendations: Emergency responders who consider their role finished after giving a patient the number of the suicide hotline, attorneys who are satisfied by giving their clients an outdated list of community mental health clinics, judges who interpret their recommendations for treatment as a fait accompli, and correctional facilities that release patients with an absurdly short supply of medications and the address of an emergency room. I worry that by creating a model encouraging all to participate, we have just absolved ones who make any effort, even if inadequate.
In some ways, the sequential intercept model has similarities with modern mental health treatment teams. In many settings, a treatment team includes a series of providers who are sequentially involved in the life of a patient. A team can include a psychiatrist for psychopharmacology; a neuropsychologist for psychological testing; a social worker for psychotherapeutic strategies; another social worker to assist in obtaining social assistance; an addiction counselor for substance use disorder; another psychiatrist who monitors the administration of a single medication, like ketamine; and a pharmacist who approves the medication regimen. That’s several providers for the treatment of one patient.
As a forensic psychiatrist, I am often asked to review treatment plans of other providers. I am asked to comment on the appropriate nature of a given treatment. Often, insurance companies want to review the continued need for treatment or whether any treatment is warranted at all. Sometimes, employers want to review a treatment plan to ensure the safety of their employees. At times, courts will ask for a review and expectations from treatment of a defendant to assist in sentencing determinations. However, I have not yet been asked by anyone if the amount of care a patient is obtaining is too fragmented and without any clear leadership.
In our endless pursuit of medicalization and standardization of mental health, we have, especially in large systems, created specialization silos for the care of our patients. Many, if not most psychiatrists, do not participate in any psychotherapy; social workers and psychologists do not prescribe (for the most part); many substance abuse counselors only address sobriety and not other primary mental illness factors; and pharmacists cannot diagnose nor are they trained in psychosocial approaches. In many ways, we have defined participants not by what they do, but what they don’t do.
One also can be saddened by the enormous logistical complexity imposed on patients required to make numerous appointments, which can deprive them of time for recovery. However, my bigger concern is that the multiplicity of providers also permits the dissolution of accountability. In my experience, those large teams have an ability to deflect responsibility in ways that are unmatched by any single provider who cannot rely on putting the fault on someone else.
Sadly and ironically, those two parallel paradigms of mental illness and criminal care impose those problems on each other by averting any attempt at interception, a “no-intercept model.” Mental health programs will deny clients involved in the criminal justice system for requiring too much treatment, too little treatment, for lack of availability of one of the necessary providers, for requiring substance use treatment, or simply for being part of the criminal justice system. Accordingly, the legal system will fail to accept recommendations by mental health providers that mental health treatment is not paramount at this time and that the defendant would be better served by addressing his criminogenic risk factors. In response, the multitude of participants in the legal system will point to the mental health system for all answers.
Contrary to many if not most problems, I do not think that the solution lies somewhere in the middle, as this would require the five stages of the legal system to compromise with the nine hypothetical participants of the mental health system. For our part, as psychiatrists, we must accept that we are ultimately responsible for all levels of care. As a field, we are also responsible for educating the public and the legal system of our role and limitations in providing care as well as being available for providing such care. Correspondingly, the legal system is responsible for putting an adequate effort into diverting patients and having or obtaining adequate understanding of available and appropriate care for their defendants.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Among his writings is chapter 7 in the new book “Critical Psychiatry: Controversies and Clinical Implications” (Springer, 2019).
Pimavanserin reduced dementia-related psychotic symptoms without affecting cognition
SAN DIEGO – Pimavanserin, a second-generation antipsychotic approved for hallucinations and delusions in patients with Parkinson’s disease, may also be helpful for psychotic symptoms in other dementia patients, Erin P. Foff, MD, said at the Clinical Trials on Alzheimer’s Disease conference.
In fact, the phase 3 HARMONY trial was stopped early, after an interim efficacy analysis determined that treatment with pimavanserin (Nuplazid) had achieved its primary endpoint – a statistically significant threefold reduction in the risk of relapse (P less than .0033).
Importantly, pimavanserin didn’t significantly affect cognition nor, at least in this controlled setting, did it appear to increase falls or other adverse events often seen with antipsychotic use in elderly patients, said Dr. Foff, clinical lead for the dementia-related psychosis program at Acadia Pharmaceuticals, which makes the drug and sponsored the study.
Based on the positive results, Acadia intends to submit a supplemental new drug application for this indication, according to an investor presentation posted on the company website.
“There is a critical need for an intervention [for psychosis symptoms] in this population,” Dr. Foff said. “We saw a robust response that was well tolerated and well maintained with no negative impact on cognitive scores.”
The second-generation antipsychotic was approved in 2016 for treating hallucinations and delusions in patients with Parkinson’s disease.
The drug is a selective antagonist of 5-HT2 receptors, with low affinity for dopamine receptors. This slightly differentiates it from other second-generation antipsychotics that affect dopamine receptors as well as 5-HT2 receptors.
HARMONY was not a typical placebo-controlled, randomized efficacy trial. Rather, it employed a two-phase design: an open-label treatment response period followed by a placebo-controlled randomization limited to open-label responders. Overall, HARMONY involved 392 patients with mild to severe dementia of numerous etiologies, including Alzheimer’s disease (66.8%), Parkinson’s disease dementia (14.3%), frontotemporal dementia (1.8%), vascular dementia (9.7%), and dementia with Lewy bodies (7.4%). All patients entered a 12-week, open-label period during which they received pimavanserin 34 mg daily. The primary endpoint was a combination of least a 30% reduction on the total Scale for the Assessment of Positive Symptom–Hallucinations and Delusions (SAPS-HD) scale plus a score of 1-2 on the Clinical Global Impressions–Improvement (CGI-I) scale, meaning better or very much better.
At 12 weeks, all responders were then randomized to placebo or continued therapy for 26 weeks. The primary endpoint was relapse, defined as at least a 30% worsening of the SAPS-HD relative to open-label baseline, plus a CGI-I score of 6-7 (worse or very much worse).
Patients were aged a mean of 74 years. Most (about 90%) were living at home. Visual hallucinations occurred in 80% and delusions in 83%. At baseline, the mean SAPS-HD score was 24.4, and the mean CGI-Severity score was 4.7. The mean Mini-Mental State Exam (MMSE) score was 16.7.
In the open-label period, pimavanserin reduced the SAPS-HD score at 12 weeks by a mean of 75%. Symptoms began to decline in the first week of treatment, with continuing improvement throughout the treatment period. By week 4, 30% had hit the response target. This number increased steadily, with 51% responding by week 4, 75% by week 8, and 88% by week 12.
By probable diagnosis, response rates were 59.8% in Alzheimer’s patients, 45.5% for those with Lewy body dementia, 71.2% among patients with Parkinson’s disease, 71% in patients with vascular dementia, and 50% in patients with frontotemporal dementia. In the final analysis, 80% of patients overall were considered responders.
The randomized potion began immediately thereafter with no washout period. About 62% (194) of the entire cohort – all responders – entered into the placebo-controlled phase. The remaining patients were either not responders (20%), dropped out because of an adverse event (7.7%), or left the study for unspecified reasons (10%). There was one death, which was not related to the study medication. A total of 41 patients were still being treated when the study was discontinued, and they were excluded from the final analysis.
When the randomized study ended, relapses had occurred in 28.3% of those taking placebo and in 12.6% of those taking pimavanserin – a statistically significant difference (hazard ratio, 0.353). This translated to a 180% reduction in relapse.
The rate of adverse events was similar in both active and placebo groups (41% vs. 36.6%). Serious adverse events occurred in 4.8% and 3.6%, respectively. The most commonly reported adverse events were headache (9.5% vs. 4.5%) and urinary tract infection (6.7% vs. 3.6%). Asthenia occurred in 2.9% of treated patients and 0.9% of placebo patients, but no falls were reported. Anxiety and dizziness were also reported in three patients taking the study medication.
Three patients (2.9%) experienced a prolonged QT phase on ECG, with a mean delay of 5.4 milliseconds from baseline. “Pimavanserin is known to have this effect of QT prolongation,” Dr. Foff said. “This 5.4-ms change is exactly in line with what we already know about pimavanserin and is not clinically significant. We saw no effect on motor function, consistent with the mechanism of action, and very low levels of agitation or aggression.”
Pimavanserin didn’t significantly change cognition from baseline in the open-label period, and in the randomized period, MMSE never differed significantly between groups.
The company also conducted an exploratory subgroup analysis that looked at placebo versus pimavanserin relapse by probable clinical diagnosis. Among the types of dementia, relapse rates for placebo versus pimavanserin were 23% versus 13% among Alzheimer’s patients, 67% versus 0% in Lewy body dementia patients, 50% versus 7% in patients with Parkinson’s, and 17% each among vascular dementia patients. Only one patient in the randomized period had frontotemporal dementia, and that patient relapsed on treatment.
Whether pimavanserin is effective specifically for psychosis in Alzheimer’s disease patients, however, remains in question. In 2018, Acadia published a negative phase 2 trial in a targeted group of 181 Alzheimer’s patients. The primary outcome in each study was mean change on the Neuropsychiatric Inventory–Nursing Home Version psychosis score (NPI-NH-PS). Clive Ballard, MD, of the University of Exeter (England), was the primary investigator.
After 6 weeks, those taking pimavanserin had a 3.76-point change in the NPI-NH-PS, compared with a 1.93-point change in the placebo group. The mean 1.84-point difference was not statistically significant.
This Alzheimer’s-only cohort group also experienced more adverse events than the HARMONY mixed-diagnosis cohort did, although the differences between pimavanserin and placebo groups were not significant. Adverse events included falls (23% of each group) and agitation (21% with pimavanserin vs. 14% with placebo). Cognition was unaffected.
Later that year, Acadia published a subgroup analysis of the same cohort parsing response by symptom severity, again with Dr. Ballard as the lead investigator.
The analysis focused on 57 patients with a baseline NPI-NH-PS of at least 12, indicating severe symptoms of psychosis.
Treatment effects were more pronounced in this group, significantly favoring pimavanserin. On the NPI-NH-PS, 88.9% of the pimavanserin group and 43.3% of the placebo group had at least a 30% improvement; 77.8% and 43.3% experienced at least a 50% improvement. The rate of serious adverse events was similar (18% with pimavanserin and 17% with placebo) and cognition was unaffected. Falls occurred in 14% of the treated group and 20% of the placebo group.
“These findings coupled with the results from other studies of pimavanserin suggest a potential role for pimavanserin in treating psychosis in patients across a range of neuropsychiatric conditions,” Dr. Ballard wrote.
SOURCE: Foff EP et al. CTAD 2019, Late-breaker 1
SAN DIEGO – Pimavanserin, a second-generation antipsychotic approved for hallucinations and delusions in patients with Parkinson’s disease, may also be helpful for psychotic symptoms in other dementia patients, Erin P. Foff, MD, said at the Clinical Trials on Alzheimer’s Disease conference.
In fact, the phase 3 HARMONY trial was stopped early, after an interim efficacy analysis determined that treatment with pimavanserin (Nuplazid) had achieved its primary endpoint – a statistically significant threefold reduction in the risk of relapse (P less than .0033).
Importantly, pimavanserin didn’t significantly affect cognition nor, at least in this controlled setting, did it appear to increase falls or other adverse events often seen with antipsychotic use in elderly patients, said Dr. Foff, clinical lead for the dementia-related psychosis program at Acadia Pharmaceuticals, which makes the drug and sponsored the study.
Based on the positive results, Acadia intends to submit a supplemental new drug application for this indication, according to an investor presentation posted on the company website.
“There is a critical need for an intervention [for psychosis symptoms] in this population,” Dr. Foff said. “We saw a robust response that was well tolerated and well maintained with no negative impact on cognitive scores.”
The second-generation antipsychotic was approved in 2016 for treating hallucinations and delusions in patients with Parkinson’s disease.
The drug is a selective antagonist of 5-HT2 receptors, with low affinity for dopamine receptors. This slightly differentiates it from other second-generation antipsychotics that affect dopamine receptors as well as 5-HT2 receptors.
HARMONY was not a typical placebo-controlled, randomized efficacy trial. Rather, it employed a two-phase design: an open-label treatment response period followed by a placebo-controlled randomization limited to open-label responders. Overall, HARMONY involved 392 patients with mild to severe dementia of numerous etiologies, including Alzheimer’s disease (66.8%), Parkinson’s disease dementia (14.3%), frontotemporal dementia (1.8%), vascular dementia (9.7%), and dementia with Lewy bodies (7.4%). All patients entered a 12-week, open-label period during which they received pimavanserin 34 mg daily. The primary endpoint was a combination of least a 30% reduction on the total Scale for the Assessment of Positive Symptom–Hallucinations and Delusions (SAPS-HD) scale plus a score of 1-2 on the Clinical Global Impressions–Improvement (CGI-I) scale, meaning better or very much better.
At 12 weeks, all responders were then randomized to placebo or continued therapy for 26 weeks. The primary endpoint was relapse, defined as at least a 30% worsening of the SAPS-HD relative to open-label baseline, plus a CGI-I score of 6-7 (worse or very much worse).
Patients were aged a mean of 74 years. Most (about 90%) were living at home. Visual hallucinations occurred in 80% and delusions in 83%. At baseline, the mean SAPS-HD score was 24.4, and the mean CGI-Severity score was 4.7. The mean Mini-Mental State Exam (MMSE) score was 16.7.
In the open-label period, pimavanserin reduced the SAPS-HD score at 12 weeks by a mean of 75%. Symptoms began to decline in the first week of treatment, with continuing improvement throughout the treatment period. By week 4, 30% had hit the response target. This number increased steadily, with 51% responding by week 4, 75% by week 8, and 88% by week 12.
By probable diagnosis, response rates were 59.8% in Alzheimer’s patients, 45.5% for those with Lewy body dementia, 71.2% among patients with Parkinson’s disease, 71% in patients with vascular dementia, and 50% in patients with frontotemporal dementia. In the final analysis, 80% of patients overall were considered responders.
The randomized potion began immediately thereafter with no washout period. About 62% (194) of the entire cohort – all responders – entered into the placebo-controlled phase. The remaining patients were either not responders (20%), dropped out because of an adverse event (7.7%), or left the study for unspecified reasons (10%). There was one death, which was not related to the study medication. A total of 41 patients were still being treated when the study was discontinued, and they were excluded from the final analysis.
When the randomized study ended, relapses had occurred in 28.3% of those taking placebo and in 12.6% of those taking pimavanserin – a statistically significant difference (hazard ratio, 0.353). This translated to a 180% reduction in relapse.
The rate of adverse events was similar in both active and placebo groups (41% vs. 36.6%). Serious adverse events occurred in 4.8% and 3.6%, respectively. The most commonly reported adverse events were headache (9.5% vs. 4.5%) and urinary tract infection (6.7% vs. 3.6%). Asthenia occurred in 2.9% of treated patients and 0.9% of placebo patients, but no falls were reported. Anxiety and dizziness were also reported in three patients taking the study medication.
Three patients (2.9%) experienced a prolonged QT phase on ECG, with a mean delay of 5.4 milliseconds from baseline. “Pimavanserin is known to have this effect of QT prolongation,” Dr. Foff said. “This 5.4-ms change is exactly in line with what we already know about pimavanserin and is not clinically significant. We saw no effect on motor function, consistent with the mechanism of action, and very low levels of agitation or aggression.”
Pimavanserin didn’t significantly change cognition from baseline in the open-label period, and in the randomized period, MMSE never differed significantly between groups.
The company also conducted an exploratory subgroup analysis that looked at placebo versus pimavanserin relapse by probable clinical diagnosis. Among the types of dementia, relapse rates for placebo versus pimavanserin were 23% versus 13% among Alzheimer’s patients, 67% versus 0% in Lewy body dementia patients, 50% versus 7% in patients with Parkinson’s, and 17% each among vascular dementia patients. Only one patient in the randomized period had frontotemporal dementia, and that patient relapsed on treatment.
Whether pimavanserin is effective specifically for psychosis in Alzheimer’s disease patients, however, remains in question. In 2018, Acadia published a negative phase 2 trial in a targeted group of 181 Alzheimer’s patients. The primary outcome in each study was mean change on the Neuropsychiatric Inventory–Nursing Home Version psychosis score (NPI-NH-PS). Clive Ballard, MD, of the University of Exeter (England), was the primary investigator.
After 6 weeks, those taking pimavanserin had a 3.76-point change in the NPI-NH-PS, compared with a 1.93-point change in the placebo group. The mean 1.84-point difference was not statistically significant.
This Alzheimer’s-only cohort group also experienced more adverse events than the HARMONY mixed-diagnosis cohort did, although the differences between pimavanserin and placebo groups were not significant. Adverse events included falls (23% of each group) and agitation (21% with pimavanserin vs. 14% with placebo). Cognition was unaffected.
Later that year, Acadia published a subgroup analysis of the same cohort parsing response by symptom severity, again with Dr. Ballard as the lead investigator.
The analysis focused on 57 patients with a baseline NPI-NH-PS of at least 12, indicating severe symptoms of psychosis.
Treatment effects were more pronounced in this group, significantly favoring pimavanserin. On the NPI-NH-PS, 88.9% of the pimavanserin group and 43.3% of the placebo group had at least a 30% improvement; 77.8% and 43.3% experienced at least a 50% improvement. The rate of serious adverse events was similar (18% with pimavanserin and 17% with placebo) and cognition was unaffected. Falls occurred in 14% of the treated group and 20% of the placebo group.
“These findings coupled with the results from other studies of pimavanserin suggest a potential role for pimavanserin in treating psychosis in patients across a range of neuropsychiatric conditions,” Dr. Ballard wrote.
SOURCE: Foff EP et al. CTAD 2019, Late-breaker 1
SAN DIEGO – Pimavanserin, a second-generation antipsychotic approved for hallucinations and delusions in patients with Parkinson’s disease, may also be helpful for psychotic symptoms in other dementia patients, Erin P. Foff, MD, said at the Clinical Trials on Alzheimer’s Disease conference.
In fact, the phase 3 HARMONY trial was stopped early, after an interim efficacy analysis determined that treatment with pimavanserin (Nuplazid) had achieved its primary endpoint – a statistically significant threefold reduction in the risk of relapse (P less than .0033).
Importantly, pimavanserin didn’t significantly affect cognition nor, at least in this controlled setting, did it appear to increase falls or other adverse events often seen with antipsychotic use in elderly patients, said Dr. Foff, clinical lead for the dementia-related psychosis program at Acadia Pharmaceuticals, which makes the drug and sponsored the study.
Based on the positive results, Acadia intends to submit a supplemental new drug application for this indication, according to an investor presentation posted on the company website.
“There is a critical need for an intervention [for psychosis symptoms] in this population,” Dr. Foff said. “We saw a robust response that was well tolerated and well maintained with no negative impact on cognitive scores.”
The second-generation antipsychotic was approved in 2016 for treating hallucinations and delusions in patients with Parkinson’s disease.
The drug is a selective antagonist of 5-HT2 receptors, with low affinity for dopamine receptors. This slightly differentiates it from other second-generation antipsychotics that affect dopamine receptors as well as 5-HT2 receptors.
HARMONY was not a typical placebo-controlled, randomized efficacy trial. Rather, it employed a two-phase design: an open-label treatment response period followed by a placebo-controlled randomization limited to open-label responders. Overall, HARMONY involved 392 patients with mild to severe dementia of numerous etiologies, including Alzheimer’s disease (66.8%), Parkinson’s disease dementia (14.3%), frontotemporal dementia (1.8%), vascular dementia (9.7%), and dementia with Lewy bodies (7.4%). All patients entered a 12-week, open-label period during which they received pimavanserin 34 mg daily. The primary endpoint was a combination of least a 30% reduction on the total Scale for the Assessment of Positive Symptom–Hallucinations and Delusions (SAPS-HD) scale plus a score of 1-2 on the Clinical Global Impressions–Improvement (CGI-I) scale, meaning better or very much better.
At 12 weeks, all responders were then randomized to placebo or continued therapy for 26 weeks. The primary endpoint was relapse, defined as at least a 30% worsening of the SAPS-HD relative to open-label baseline, plus a CGI-I score of 6-7 (worse or very much worse).
Patients were aged a mean of 74 years. Most (about 90%) were living at home. Visual hallucinations occurred in 80% and delusions in 83%. At baseline, the mean SAPS-HD score was 24.4, and the mean CGI-Severity score was 4.7. The mean Mini-Mental State Exam (MMSE) score was 16.7.
In the open-label period, pimavanserin reduced the SAPS-HD score at 12 weeks by a mean of 75%. Symptoms began to decline in the first week of treatment, with continuing improvement throughout the treatment period. By week 4, 30% had hit the response target. This number increased steadily, with 51% responding by week 4, 75% by week 8, and 88% by week 12.
By probable diagnosis, response rates were 59.8% in Alzheimer’s patients, 45.5% for those with Lewy body dementia, 71.2% among patients with Parkinson’s disease, 71% in patients with vascular dementia, and 50% in patients with frontotemporal dementia. In the final analysis, 80% of patients overall were considered responders.
The randomized potion began immediately thereafter with no washout period. About 62% (194) of the entire cohort – all responders – entered into the placebo-controlled phase. The remaining patients were either not responders (20%), dropped out because of an adverse event (7.7%), or left the study for unspecified reasons (10%). There was one death, which was not related to the study medication. A total of 41 patients were still being treated when the study was discontinued, and they were excluded from the final analysis.
When the randomized study ended, relapses had occurred in 28.3% of those taking placebo and in 12.6% of those taking pimavanserin – a statistically significant difference (hazard ratio, 0.353). This translated to a 180% reduction in relapse.
The rate of adverse events was similar in both active and placebo groups (41% vs. 36.6%). Serious adverse events occurred in 4.8% and 3.6%, respectively. The most commonly reported adverse events were headache (9.5% vs. 4.5%) and urinary tract infection (6.7% vs. 3.6%). Asthenia occurred in 2.9% of treated patients and 0.9% of placebo patients, but no falls were reported. Anxiety and dizziness were also reported in three patients taking the study medication.
Three patients (2.9%) experienced a prolonged QT phase on ECG, with a mean delay of 5.4 milliseconds from baseline. “Pimavanserin is known to have this effect of QT prolongation,” Dr. Foff said. “This 5.4-ms change is exactly in line with what we already know about pimavanserin and is not clinically significant. We saw no effect on motor function, consistent with the mechanism of action, and very low levels of agitation or aggression.”
Pimavanserin didn’t significantly change cognition from baseline in the open-label period, and in the randomized period, MMSE never differed significantly between groups.
The company also conducted an exploratory subgroup analysis that looked at placebo versus pimavanserin relapse by probable clinical diagnosis. Among the types of dementia, relapse rates for placebo versus pimavanserin were 23% versus 13% among Alzheimer’s patients, 67% versus 0% in Lewy body dementia patients, 50% versus 7% in patients with Parkinson’s, and 17% each among vascular dementia patients. Only one patient in the randomized period had frontotemporal dementia, and that patient relapsed on treatment.
Whether pimavanserin is effective specifically for psychosis in Alzheimer’s disease patients, however, remains in question. In 2018, Acadia published a negative phase 2 trial in a targeted group of 181 Alzheimer’s patients. The primary outcome in each study was mean change on the Neuropsychiatric Inventory–Nursing Home Version psychosis score (NPI-NH-PS). Clive Ballard, MD, of the University of Exeter (England), was the primary investigator.
After 6 weeks, those taking pimavanserin had a 3.76-point change in the NPI-NH-PS, compared with a 1.93-point change in the placebo group. The mean 1.84-point difference was not statistically significant.
This Alzheimer’s-only cohort group also experienced more adverse events than the HARMONY mixed-diagnosis cohort did, although the differences between pimavanserin and placebo groups were not significant. Adverse events included falls (23% of each group) and agitation (21% with pimavanserin vs. 14% with placebo). Cognition was unaffected.
Later that year, Acadia published a subgroup analysis of the same cohort parsing response by symptom severity, again with Dr. Ballard as the lead investigator.
The analysis focused on 57 patients with a baseline NPI-NH-PS of at least 12, indicating severe symptoms of psychosis.
Treatment effects were more pronounced in this group, significantly favoring pimavanserin. On the NPI-NH-PS, 88.9% of the pimavanserin group and 43.3% of the placebo group had at least a 30% improvement; 77.8% and 43.3% experienced at least a 50% improvement. The rate of serious adverse events was similar (18% with pimavanserin and 17% with placebo) and cognition was unaffected. Falls occurred in 14% of the treated group and 20% of the placebo group.
“These findings coupled with the results from other studies of pimavanserin suggest a potential role for pimavanserin in treating psychosis in patients across a range of neuropsychiatric conditions,” Dr. Ballard wrote.
SOURCE: Foff EP et al. CTAD 2019, Late-breaker 1
REPORTING FROM CTAD 2019
The Great Pretender
Susannah Cahalan’s new book challenges an experiment that changed psychiatry
As an undergraduate psychology major, I was taught about the Rosenhan study in several of my courses. My professors lectured about the shocking findings psychologist David Rosenhan, PhD, documented in a 1973 Science article, “On Being Sane in Insane Places” and these findings lent themselves to lecture hall drama. Eight people presented to hospitals and said they heard voices saying: “empty, hollow, thud.” These “pseudopatients” exhibited no other psychiatric symptoms but were admitted, diagnosed with schizophrenia, and observations of their behavior were made. The charts included notes such as, “Patient exhibits writing behavior,” my professors said. The pseudopatients were kept for an average of 19 days, and one for as long as 51 days. The decades have passed, and there are many things I learned in college that I have since forgotten, but I remember “empty, hollow, thud,” and the famous Rosenhan experiment.
I was eager to read Susannah Cahalan’s book, “The Great Pretender” (Grand Central Publishing, 2019), which puts both Dr. Rosenhan and his pseudopatient study under a microscope. Ms. Cahalan is the author of the page-turner, Brain on Fire: My Month of Madness (Free Press, 2012), where she recounted her own struggle with a psychotic episode. Ms. Cahalan, a young reporter in New York City, became psychotic and then catatonic; her condition perplexed the neurologists who were treating her on an inpatient unit, and they were on the verge of transferring her to psychiatry when a diagnosis of anti-NMDA receptor encephalitis was suspected and then confirmed with a brain biopsy. Ms. Cahalan made a full recovery after treatment with steroids, intravenous immunoglobulin, and plasmapheresis. While Ms. Cahalan’s symptoms were classic for a severe psychotic disorder, there was reason to believe that this was not a primary psychiatric disorder: She was having grand mal seizures. Her book was a bestseller, and she has spoken widely to make others aware of this rare illness that masquerades as psychosis. I heard her speak at the opening session of the American Psychiatric Association’s annual meeting in May of 2017.“My family, like many families before them, fought against the tyranny of the mental illness label,” Ms. Cahalan writes at the very beginning of “The Great Pretender.” She goes on to talk about how psychiatry differs from other medical fields: It’s the only specialty where people can be treated against their will; psychiatry casts judgments on the person; mental illness is poorly defined – perhaps there is no clear divide between normal and mad; and psychiatric disorders are less “real” than other illnesses. Throughout the book she refers to psychiatrists as smug and arrogant.
Ms. Cahalan takes on the task of documenting the horrors of psychiatry’s often sordid history, starting with journalist Nellie Bly’s 1887 journey into to a psychiatric facility to expose the abuses there. Certainly, psychiatry’s history is sordid. Ms. Cahalan talks about inhumane conditions in overcrowded psychiatric hospitals, about our sad chapter of lobotomies, about the influence of psychoanalysis on diagnosis and treatment, and about how homosexuality was once an illness and now is not. She includes “One Flew Over the Cuckoo’s Nest,” “The Myth of Mental Illness,” big pharma, and the Goldwater fiasco. In her recounting of the history, it’s all bad. She mentions Benjamin Rush, MD, only once, as the creator of “ ‘... the tranquilizing chair’ (a case of the worst false advertising ever), a terrifying sensory-deprivation apparatus in which patients were strapped down to a chair with a wooden box placed over their heads to block stimulation, restrict movement, and reduce blood to the brain.” Dr. Rush’s role as the father of American psychiatry who challenged the belief that mental illness was the result of demonic possession, gets no mention. Nor does Ms. Cahalan note that he founded Pennsylvania Hospital, where moral and occupational therapy revolutionized the treatment of those with mental illness.
So there’s her story, her rendition of the history of American psychiatry, and through this she weaves in the story of the Rosenhan experiment.
“ ‘It all started out as a dare,’ Dr. Rosenhan told a local newspaper, ‘I was teaching psychology at Swarthmore, and my students were saying that the course was too conceptual and abstract. So I said, ‘Okay, if you really want to know what mental patients are like, become mental patients.’ ”
Really? I read this and wondered how a psychologist could talk about people who had been hospitalized with psychiatric disorders as though they were aliens. Certainly, some of these students, their family members, or their friends must have been hospitalized at some point. Yet all through, there is this sense that the patients are other, and the discovery of the undercover operation is that the patients are actually human beings! Dr. Rosenhan, who was one of the pseudopatients, goes on to conclude that the label is everything, that once labeled they are treated differently by the nurses in “cages” and the doctors who walk by and avert their gaze. A second man Ms. Cahalan named, also one of the pseudopatients, had a similar experience. A third subject she located was not included in the study: His experience was counter to the findings of the study, his time in the hospital was a positive, he found it comforting, and the experiences he had there had a lasting positive influence on his life.
Ms. Cahalan talks about the publication of “On Being Sane in Insane Places” as a study that was finally scientific, one that changed all of psychiatry, and was the driving force for the creation of the DSM-III and the closure of state hospitals. I wondered if it was as influential as Ms. Cahalan claims, and I asked some psychiatrists who were practicing in 1973 when the article was published. I wanted to know if this study rocked their world.
“At first, with the great amount of publicity the study generated, it was added fodder for the antipsychiatrists, including the Scientologists and Szaszians,” Steven Sharfstein, MD, a former president of the American Psychiatric Association, told me. “But as young psychiatrists in the trenches, business continued to boom, and we continued to do the best job we could with diagnosis, assessment of risk, involuntary commitment, and treatment. And from what I recall, morale was high in the 1970s. We had some new medications and psychotherapies, and there was community activism. Faking symptoms to gain admission seemed to be a no-brainer, but keeping people for long stays was more problematic.”
E. Fuller Torrey, MD, the founder of the Treatment Advocacy Center who worked for many years treating patients at St. Elizabeth’s Hospital, replied: “It is important to remember that this study was published at the height of the deinstitutionalization movement and quite likely accelerated it. As I recall, at the time it seemed odd that all eight patients claimed to have had similar experiences while hospitalized. I think the main effect of the study was to provide ammunition for the antipsychiatrists.”
Ms. Cahalan has bought into the antipsychiatry movement full force. It’s not until the very end that there is any acknowledgment that psychiatry ever helped anyone, and even then, it’s a bit begrudging. Worse, she neglects to mention that people with psychiatric disorders suffer because of their psychic pain; one could get through this book and believe that people with mental illness struggle only because they are labeled and then mistreated, and for someone who has suffered herself, she misses the essence of how awful it is to be ill, and that people are often helped by psychiatric treatments. When she finally adds a paragraph talking about the usefulness of psychotropics, it’s with a caveat. “But I’m not here to rail against the drugs. There are plenty of places you can get that perspective. I see that these drugs help many people lead full and meaningful lives. It would be folly to discount their worth. We also can’t deny that the situation is complicated.”
There are moments in the manuscript where I found it difficult to know what were Dr. Rosenhan’s interpretations and what were Ms. Callahan’s interpretations of Dr. Rosenhan’s experiences. A lot of assumptions are made – particularly about the motivations of the hospital staff – and I wasn’t always sure they were correct. For example, on his second day in the hospital, Dr. Rosenhan asked a nurse for the newspaper. When she tells him it hasn’t come yet, he concludes that the staff is keeping the newspapers from the patients. And when a staff member is initially chatty then later shuns Dr. Rosenhan, he concludes that the man initially mistook him for a psychiatrist because he looks professorial. Both Ms. Cahalan and Dr. Rosenhan approach psychiatry with biases, and they don’t always question their assumptions.
, a professor who didn’t treat patients. This intermixing of the two fields felt contrived to me, and gave too much credence to the idea that no one really knows sane from disordered, and everyone was embracing the antipsychiatry dogma. Surely, someone during the those years must have liked their psychiatrist.
That said, Ms. Cahalan does a phenomenal job of infiltrating the world of the late Dr. Rosenhan. She starts out enamored by him and by his finding that psychiatrists can’t tell real illness from faked disorder. She meets with his friends, his son, his colleagues, his students, and she flies all over the country to meet with those who can help her understand him. She gains access to his personal files and to the book he started to write about the experiment, then abandoned, which eventually resulted in a lawsuit by Doubleday to have the book’s advance returned. At one point, she even hires a private detective.
What is Ms. Cahalan looking for so desperately? She’s looking for these anonymous pseudopatients, the people who were admitted to these unnamed state hospitals, who made observations and took notes, who were diagnosed with schizophrenia and then finally released. She’s looking for the truth, and while she identifies Dr. Rosenhan and two other pseudopatients as people who faked their way into the hospital, she finds a mass of contradictions. The one pseudopatient was excluded from the study – he is the one who felt comforted by his time in the hospital. The other six pseudopatients could not be found, despite Ms. Cahalan’s heroic attempts. Furthermore, she found many inconsistencies in what Dr. Rosenhan reported, and his hospital notes revealed more than a presentation for voices saying “empty, hollow, thud.” He reported it had been going on for months, that he had put copper over his ears to block the sound, and that he felt suicidal.
Ultimately, Ms. Cahalan was left to conclude that the Rosenhan experiment was a lie, that the pseudopatients likely never existed and the article was a fabrication. She brings up other studies that have been proven to be fraudulent, and by this point, our faith in all of science is pretty shaken.
Ms. Cahalan took a long journey to get us to this place, one that spent a lot of effort in bashing psychiatry, finally concluding that, as a result of this fraudulent experiment, too many hospitals have been shuttered – leaving our sickest patients to the streets and to the jails – and that there are not enough mental health professionals. As a psychiatrist – one who is often willing to question our practices – I was distracted by the flagrant antipsychiatry sentiments. Reading past that, Ms. Cahalan’s remarkable detective work and creative intermingling of the Rosenhan experiment layered on the history of psychiatry, further layered on her own experience with psychosis, makes for an amazing story. The Rosenhan study may have rocked the world of psychiatry; the fact that it was fabricated should rock us even more.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
Susannah Cahalan’s new book challenges an experiment that changed psychiatry
Susannah Cahalan’s new book challenges an experiment that changed psychiatry
As an undergraduate psychology major, I was taught about the Rosenhan study in several of my courses. My professors lectured about the shocking findings psychologist David Rosenhan, PhD, documented in a 1973 Science article, “On Being Sane in Insane Places” and these findings lent themselves to lecture hall drama. Eight people presented to hospitals and said they heard voices saying: “empty, hollow, thud.” These “pseudopatients” exhibited no other psychiatric symptoms but were admitted, diagnosed with schizophrenia, and observations of their behavior were made. The charts included notes such as, “Patient exhibits writing behavior,” my professors said. The pseudopatients were kept for an average of 19 days, and one for as long as 51 days. The decades have passed, and there are many things I learned in college that I have since forgotten, but I remember “empty, hollow, thud,” and the famous Rosenhan experiment.
I was eager to read Susannah Cahalan’s book, “The Great Pretender” (Grand Central Publishing, 2019), which puts both Dr. Rosenhan and his pseudopatient study under a microscope. Ms. Cahalan is the author of the page-turner, Brain on Fire: My Month of Madness (Free Press, 2012), where she recounted her own struggle with a psychotic episode. Ms. Cahalan, a young reporter in New York City, became psychotic and then catatonic; her condition perplexed the neurologists who were treating her on an inpatient unit, and they were on the verge of transferring her to psychiatry when a diagnosis of anti-NMDA receptor encephalitis was suspected and then confirmed with a brain biopsy. Ms. Cahalan made a full recovery after treatment with steroids, intravenous immunoglobulin, and plasmapheresis. While Ms. Cahalan’s symptoms were classic for a severe psychotic disorder, there was reason to believe that this was not a primary psychiatric disorder: She was having grand mal seizures. Her book was a bestseller, and she has spoken widely to make others aware of this rare illness that masquerades as psychosis. I heard her speak at the opening session of the American Psychiatric Association’s annual meeting in May of 2017.“My family, like many families before them, fought against the tyranny of the mental illness label,” Ms. Cahalan writes at the very beginning of “The Great Pretender.” She goes on to talk about how psychiatry differs from other medical fields: It’s the only specialty where people can be treated against their will; psychiatry casts judgments on the person; mental illness is poorly defined – perhaps there is no clear divide between normal and mad; and psychiatric disorders are less “real” than other illnesses. Throughout the book she refers to psychiatrists as smug and arrogant.
Ms. Cahalan takes on the task of documenting the horrors of psychiatry’s often sordid history, starting with journalist Nellie Bly’s 1887 journey into to a psychiatric facility to expose the abuses there. Certainly, psychiatry’s history is sordid. Ms. Cahalan talks about inhumane conditions in overcrowded psychiatric hospitals, about our sad chapter of lobotomies, about the influence of psychoanalysis on diagnosis and treatment, and about how homosexuality was once an illness and now is not. She includes “One Flew Over the Cuckoo’s Nest,” “The Myth of Mental Illness,” big pharma, and the Goldwater fiasco. In her recounting of the history, it’s all bad. She mentions Benjamin Rush, MD, only once, as the creator of “ ‘... the tranquilizing chair’ (a case of the worst false advertising ever), a terrifying sensory-deprivation apparatus in which patients were strapped down to a chair with a wooden box placed over their heads to block stimulation, restrict movement, and reduce blood to the brain.” Dr. Rush’s role as the father of American psychiatry who challenged the belief that mental illness was the result of demonic possession, gets no mention. Nor does Ms. Cahalan note that he founded Pennsylvania Hospital, where moral and occupational therapy revolutionized the treatment of those with mental illness.
So there’s her story, her rendition of the history of American psychiatry, and through this she weaves in the story of the Rosenhan experiment.
“ ‘It all started out as a dare,’ Dr. Rosenhan told a local newspaper, ‘I was teaching psychology at Swarthmore, and my students were saying that the course was too conceptual and abstract. So I said, ‘Okay, if you really want to know what mental patients are like, become mental patients.’ ”
Really? I read this and wondered how a psychologist could talk about people who had been hospitalized with psychiatric disorders as though they were aliens. Certainly, some of these students, their family members, or their friends must have been hospitalized at some point. Yet all through, there is this sense that the patients are other, and the discovery of the undercover operation is that the patients are actually human beings! Dr. Rosenhan, who was one of the pseudopatients, goes on to conclude that the label is everything, that once labeled they are treated differently by the nurses in “cages” and the doctors who walk by and avert their gaze. A second man Ms. Cahalan named, also one of the pseudopatients, had a similar experience. A third subject she located was not included in the study: His experience was counter to the findings of the study, his time in the hospital was a positive, he found it comforting, and the experiences he had there had a lasting positive influence on his life.
Ms. Cahalan talks about the publication of “On Being Sane in Insane Places” as a study that was finally scientific, one that changed all of psychiatry, and was the driving force for the creation of the DSM-III and the closure of state hospitals. I wondered if it was as influential as Ms. Cahalan claims, and I asked some psychiatrists who were practicing in 1973 when the article was published. I wanted to know if this study rocked their world.
“At first, with the great amount of publicity the study generated, it was added fodder for the antipsychiatrists, including the Scientologists and Szaszians,” Steven Sharfstein, MD, a former president of the American Psychiatric Association, told me. “But as young psychiatrists in the trenches, business continued to boom, and we continued to do the best job we could with diagnosis, assessment of risk, involuntary commitment, and treatment. And from what I recall, morale was high in the 1970s. We had some new medications and psychotherapies, and there was community activism. Faking symptoms to gain admission seemed to be a no-brainer, but keeping people for long stays was more problematic.”
E. Fuller Torrey, MD, the founder of the Treatment Advocacy Center who worked for many years treating patients at St. Elizabeth’s Hospital, replied: “It is important to remember that this study was published at the height of the deinstitutionalization movement and quite likely accelerated it. As I recall, at the time it seemed odd that all eight patients claimed to have had similar experiences while hospitalized. I think the main effect of the study was to provide ammunition for the antipsychiatrists.”
Ms. Cahalan has bought into the antipsychiatry movement full force. It’s not until the very end that there is any acknowledgment that psychiatry ever helped anyone, and even then, it’s a bit begrudging. Worse, she neglects to mention that people with psychiatric disorders suffer because of their psychic pain; one could get through this book and believe that people with mental illness struggle only because they are labeled and then mistreated, and for someone who has suffered herself, she misses the essence of how awful it is to be ill, and that people are often helped by psychiatric treatments. When she finally adds a paragraph talking about the usefulness of psychotropics, it’s with a caveat. “But I’m not here to rail against the drugs. There are plenty of places you can get that perspective. I see that these drugs help many people lead full and meaningful lives. It would be folly to discount their worth. We also can’t deny that the situation is complicated.”
There are moments in the manuscript where I found it difficult to know what were Dr. Rosenhan’s interpretations and what were Ms. Callahan’s interpretations of Dr. Rosenhan’s experiences. A lot of assumptions are made – particularly about the motivations of the hospital staff – and I wasn’t always sure they were correct. For example, on his second day in the hospital, Dr. Rosenhan asked a nurse for the newspaper. When she tells him it hasn’t come yet, he concludes that the staff is keeping the newspapers from the patients. And when a staff member is initially chatty then later shuns Dr. Rosenhan, he concludes that the man initially mistook him for a psychiatrist because he looks professorial. Both Ms. Cahalan and Dr. Rosenhan approach psychiatry with biases, and they don’t always question their assumptions.
, a professor who didn’t treat patients. This intermixing of the two fields felt contrived to me, and gave too much credence to the idea that no one really knows sane from disordered, and everyone was embracing the antipsychiatry dogma. Surely, someone during the those years must have liked their psychiatrist.
That said, Ms. Cahalan does a phenomenal job of infiltrating the world of the late Dr. Rosenhan. She starts out enamored by him and by his finding that psychiatrists can’t tell real illness from faked disorder. She meets with his friends, his son, his colleagues, his students, and she flies all over the country to meet with those who can help her understand him. She gains access to his personal files and to the book he started to write about the experiment, then abandoned, which eventually resulted in a lawsuit by Doubleday to have the book’s advance returned. At one point, she even hires a private detective.
What is Ms. Cahalan looking for so desperately? She’s looking for these anonymous pseudopatients, the people who were admitted to these unnamed state hospitals, who made observations and took notes, who were diagnosed with schizophrenia and then finally released. She’s looking for the truth, and while she identifies Dr. Rosenhan and two other pseudopatients as people who faked their way into the hospital, she finds a mass of contradictions. The one pseudopatient was excluded from the study – he is the one who felt comforted by his time in the hospital. The other six pseudopatients could not be found, despite Ms. Cahalan’s heroic attempts. Furthermore, she found many inconsistencies in what Dr. Rosenhan reported, and his hospital notes revealed more than a presentation for voices saying “empty, hollow, thud.” He reported it had been going on for months, that he had put copper over his ears to block the sound, and that he felt suicidal.
Ultimately, Ms. Cahalan was left to conclude that the Rosenhan experiment was a lie, that the pseudopatients likely never existed and the article was a fabrication. She brings up other studies that have been proven to be fraudulent, and by this point, our faith in all of science is pretty shaken.
Ms. Cahalan took a long journey to get us to this place, one that spent a lot of effort in bashing psychiatry, finally concluding that, as a result of this fraudulent experiment, too many hospitals have been shuttered – leaving our sickest patients to the streets and to the jails – and that there are not enough mental health professionals. As a psychiatrist – one who is often willing to question our practices – I was distracted by the flagrant antipsychiatry sentiments. Reading past that, Ms. Cahalan’s remarkable detective work and creative intermingling of the Rosenhan experiment layered on the history of psychiatry, further layered on her own experience with psychosis, makes for an amazing story. The Rosenhan study may have rocked the world of psychiatry; the fact that it was fabricated should rock us even more.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
As an undergraduate psychology major, I was taught about the Rosenhan study in several of my courses. My professors lectured about the shocking findings psychologist David Rosenhan, PhD, documented in a 1973 Science article, “On Being Sane in Insane Places” and these findings lent themselves to lecture hall drama. Eight people presented to hospitals and said they heard voices saying: “empty, hollow, thud.” These “pseudopatients” exhibited no other psychiatric symptoms but were admitted, diagnosed with schizophrenia, and observations of their behavior were made. The charts included notes such as, “Patient exhibits writing behavior,” my professors said. The pseudopatients were kept for an average of 19 days, and one for as long as 51 days. The decades have passed, and there are many things I learned in college that I have since forgotten, but I remember “empty, hollow, thud,” and the famous Rosenhan experiment.
I was eager to read Susannah Cahalan’s book, “The Great Pretender” (Grand Central Publishing, 2019), which puts both Dr. Rosenhan and his pseudopatient study under a microscope. Ms. Cahalan is the author of the page-turner, Brain on Fire: My Month of Madness (Free Press, 2012), where she recounted her own struggle with a psychotic episode. Ms. Cahalan, a young reporter in New York City, became psychotic and then catatonic; her condition perplexed the neurologists who were treating her on an inpatient unit, and they were on the verge of transferring her to psychiatry when a diagnosis of anti-NMDA receptor encephalitis was suspected and then confirmed with a brain biopsy. Ms. Cahalan made a full recovery after treatment with steroids, intravenous immunoglobulin, and plasmapheresis. While Ms. Cahalan’s symptoms were classic for a severe psychotic disorder, there was reason to believe that this was not a primary psychiatric disorder: She was having grand mal seizures. Her book was a bestseller, and she has spoken widely to make others aware of this rare illness that masquerades as psychosis. I heard her speak at the opening session of the American Psychiatric Association’s annual meeting in May of 2017.“My family, like many families before them, fought against the tyranny of the mental illness label,” Ms. Cahalan writes at the very beginning of “The Great Pretender.” She goes on to talk about how psychiatry differs from other medical fields: It’s the only specialty where people can be treated against their will; psychiatry casts judgments on the person; mental illness is poorly defined – perhaps there is no clear divide between normal and mad; and psychiatric disorders are less “real” than other illnesses. Throughout the book she refers to psychiatrists as smug and arrogant.
Ms. Cahalan takes on the task of documenting the horrors of psychiatry’s often sordid history, starting with journalist Nellie Bly’s 1887 journey into to a psychiatric facility to expose the abuses there. Certainly, psychiatry’s history is sordid. Ms. Cahalan talks about inhumane conditions in overcrowded psychiatric hospitals, about our sad chapter of lobotomies, about the influence of psychoanalysis on diagnosis and treatment, and about how homosexuality was once an illness and now is not. She includes “One Flew Over the Cuckoo’s Nest,” “The Myth of Mental Illness,” big pharma, and the Goldwater fiasco. In her recounting of the history, it’s all bad. She mentions Benjamin Rush, MD, only once, as the creator of “ ‘... the tranquilizing chair’ (a case of the worst false advertising ever), a terrifying sensory-deprivation apparatus in which patients were strapped down to a chair with a wooden box placed over their heads to block stimulation, restrict movement, and reduce blood to the brain.” Dr. Rush’s role as the father of American psychiatry who challenged the belief that mental illness was the result of demonic possession, gets no mention. Nor does Ms. Cahalan note that he founded Pennsylvania Hospital, where moral and occupational therapy revolutionized the treatment of those with mental illness.
So there’s her story, her rendition of the history of American psychiatry, and through this she weaves in the story of the Rosenhan experiment.
“ ‘It all started out as a dare,’ Dr. Rosenhan told a local newspaper, ‘I was teaching psychology at Swarthmore, and my students were saying that the course was too conceptual and abstract. So I said, ‘Okay, if you really want to know what mental patients are like, become mental patients.’ ”
Really? I read this and wondered how a psychologist could talk about people who had been hospitalized with psychiatric disorders as though they were aliens. Certainly, some of these students, their family members, or their friends must have been hospitalized at some point. Yet all through, there is this sense that the patients are other, and the discovery of the undercover operation is that the patients are actually human beings! Dr. Rosenhan, who was one of the pseudopatients, goes on to conclude that the label is everything, that once labeled they are treated differently by the nurses in “cages” and the doctors who walk by and avert their gaze. A second man Ms. Cahalan named, also one of the pseudopatients, had a similar experience. A third subject she located was not included in the study: His experience was counter to the findings of the study, his time in the hospital was a positive, he found it comforting, and the experiences he had there had a lasting positive influence on his life.
Ms. Cahalan talks about the publication of “On Being Sane in Insane Places” as a study that was finally scientific, one that changed all of psychiatry, and was the driving force for the creation of the DSM-III and the closure of state hospitals. I wondered if it was as influential as Ms. Cahalan claims, and I asked some psychiatrists who were practicing in 1973 when the article was published. I wanted to know if this study rocked their world.
“At first, with the great amount of publicity the study generated, it was added fodder for the antipsychiatrists, including the Scientologists and Szaszians,” Steven Sharfstein, MD, a former president of the American Psychiatric Association, told me. “But as young psychiatrists in the trenches, business continued to boom, and we continued to do the best job we could with diagnosis, assessment of risk, involuntary commitment, and treatment. And from what I recall, morale was high in the 1970s. We had some new medications and psychotherapies, and there was community activism. Faking symptoms to gain admission seemed to be a no-brainer, but keeping people for long stays was more problematic.”
E. Fuller Torrey, MD, the founder of the Treatment Advocacy Center who worked for many years treating patients at St. Elizabeth’s Hospital, replied: “It is important to remember that this study was published at the height of the deinstitutionalization movement and quite likely accelerated it. As I recall, at the time it seemed odd that all eight patients claimed to have had similar experiences while hospitalized. I think the main effect of the study was to provide ammunition for the antipsychiatrists.”
Ms. Cahalan has bought into the antipsychiatry movement full force. It’s not until the very end that there is any acknowledgment that psychiatry ever helped anyone, and even then, it’s a bit begrudging. Worse, she neglects to mention that people with psychiatric disorders suffer because of their psychic pain; one could get through this book and believe that people with mental illness struggle only because they are labeled and then mistreated, and for someone who has suffered herself, she misses the essence of how awful it is to be ill, and that people are often helped by psychiatric treatments. When she finally adds a paragraph talking about the usefulness of psychotropics, it’s with a caveat. “But I’m not here to rail against the drugs. There are plenty of places you can get that perspective. I see that these drugs help many people lead full and meaningful lives. It would be folly to discount their worth. We also can’t deny that the situation is complicated.”
There are moments in the manuscript where I found it difficult to know what were Dr. Rosenhan’s interpretations and what were Ms. Callahan’s interpretations of Dr. Rosenhan’s experiences. A lot of assumptions are made – particularly about the motivations of the hospital staff – and I wasn’t always sure they were correct. For example, on his second day in the hospital, Dr. Rosenhan asked a nurse for the newspaper. When she tells him it hasn’t come yet, he concludes that the staff is keeping the newspapers from the patients. And when a staff member is initially chatty then later shuns Dr. Rosenhan, he concludes that the man initially mistook him for a psychiatrist because he looks professorial. Both Ms. Cahalan and Dr. Rosenhan approach psychiatry with biases, and they don’t always question their assumptions.
, a professor who didn’t treat patients. This intermixing of the two fields felt contrived to me, and gave too much credence to the idea that no one really knows sane from disordered, and everyone was embracing the antipsychiatry dogma. Surely, someone during the those years must have liked their psychiatrist.
That said, Ms. Cahalan does a phenomenal job of infiltrating the world of the late Dr. Rosenhan. She starts out enamored by him and by his finding that psychiatrists can’t tell real illness from faked disorder. She meets with his friends, his son, his colleagues, his students, and she flies all over the country to meet with those who can help her understand him. She gains access to his personal files and to the book he started to write about the experiment, then abandoned, which eventually resulted in a lawsuit by Doubleday to have the book’s advance returned. At one point, she even hires a private detective.
What is Ms. Cahalan looking for so desperately? She’s looking for these anonymous pseudopatients, the people who were admitted to these unnamed state hospitals, who made observations and took notes, who were diagnosed with schizophrenia and then finally released. She’s looking for the truth, and while she identifies Dr. Rosenhan and two other pseudopatients as people who faked their way into the hospital, she finds a mass of contradictions. The one pseudopatient was excluded from the study – he is the one who felt comforted by his time in the hospital. The other six pseudopatients could not be found, despite Ms. Cahalan’s heroic attempts. Furthermore, she found many inconsistencies in what Dr. Rosenhan reported, and his hospital notes revealed more than a presentation for voices saying “empty, hollow, thud.” He reported it had been going on for months, that he had put copper over his ears to block the sound, and that he felt suicidal.
Ultimately, Ms. Cahalan was left to conclude that the Rosenhan experiment was a lie, that the pseudopatients likely never existed and the article was a fabrication. She brings up other studies that have been proven to be fraudulent, and by this point, our faith in all of science is pretty shaken.
Ms. Cahalan took a long journey to get us to this place, one that spent a lot of effort in bashing psychiatry, finally concluding that, as a result of this fraudulent experiment, too many hospitals have been shuttered – leaving our sickest patients to the streets and to the jails – and that there are not enough mental health professionals. As a psychiatrist – one who is often willing to question our practices – I was distracted by the flagrant antipsychiatry sentiments. Reading past that, Ms. Cahalan’s remarkable detective work and creative intermingling of the Rosenhan experiment layered on the history of psychiatry, further layered on her own experience with psychosis, makes for an amazing story. The Rosenhan study may have rocked the world of psychiatry; the fact that it was fabricated should rock us even more.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.