User login
Prosody recognition associated with functioning in first-episode schizophrenia
Affective prosody recognition is associated with role and social functioning in patients with a recent first episode of schizophrenia, according to Kelsey A. Bonfils, PhD, and associates.
The investigators conducted an analysis of 49 patients aged between 18 and 45 years with a recent first episode of schizophrenia who were participating in a larger randomized, controlled trial. Symptoms of schizophrenia were assessed using a 24-item version of the Brief Psychiatric Rating Scale (BPRS) and functioning was assessed using the Global Functioning Scale (GFS) and Role Functioning Scale (RFS). Study participants took the Prosody Task, which assessed the ability to recognize happiness, sadness, anger, fear, and disgust, and the Facial Emotion Identification Test (FEIT), which assesses the ability to recognize happiness, sadness, anger, fear, surprise, and disgust, reported Dr. Bonfils of the Veterans Affairs Pittsburgh Healthcare System and the department of psychiatry at the University of Pittsburgh. The study was published in Schizophrenia Research: Cognition.
In the Prosody Task, patients were significantly more likely to recognize anger (45.6% correct) and sadness (43.8%), and significantly less likely to recognize disgust (21.9%). In the FEIT, patients were most likely to recognize happiness (97.5%), followed by surprise (90.0%), anger (85.0%), sadness (77.5%), disgust (73.8%), and fear (55.0%).
Performance in the Prosody Task was associated with GFS role functioning and RFS social functioning, while FEIT performance was not significantly associated with any functioning measure. In terms of symptoms, Prosody Task performance was negatively associated with disorganization in the BPRS, and FEIT performance was associated with disorganization, reality distortion, and positive symptoms.
“These findings are consistent with the view that emotion recognition deficits could be contributing to deficits in the ability of people with first-episode schizophrenia to adequately function in the real world, both in relationships with friends and in normative young adult roles,” the investigators wrote.
Dr. Bonfils reported no conflicts of interest. Three coauthors reported receiving support, research grants, and funding from several pharmaceutical companies.
SOURCE: Bonfils KA et al. Schizophr Res Cogn. 2019. doi: 10.1016/j.scog.2019.100153.
Affective prosody recognition is associated with role and social functioning in patients with a recent first episode of schizophrenia, according to Kelsey A. Bonfils, PhD, and associates.
The investigators conducted an analysis of 49 patients aged between 18 and 45 years with a recent first episode of schizophrenia who were participating in a larger randomized, controlled trial. Symptoms of schizophrenia were assessed using a 24-item version of the Brief Psychiatric Rating Scale (BPRS) and functioning was assessed using the Global Functioning Scale (GFS) and Role Functioning Scale (RFS). Study participants took the Prosody Task, which assessed the ability to recognize happiness, sadness, anger, fear, and disgust, and the Facial Emotion Identification Test (FEIT), which assesses the ability to recognize happiness, sadness, anger, fear, surprise, and disgust, reported Dr. Bonfils of the Veterans Affairs Pittsburgh Healthcare System and the department of psychiatry at the University of Pittsburgh. The study was published in Schizophrenia Research: Cognition.
In the Prosody Task, patients were significantly more likely to recognize anger (45.6% correct) and sadness (43.8%), and significantly less likely to recognize disgust (21.9%). In the FEIT, patients were most likely to recognize happiness (97.5%), followed by surprise (90.0%), anger (85.0%), sadness (77.5%), disgust (73.8%), and fear (55.0%).
Performance in the Prosody Task was associated with GFS role functioning and RFS social functioning, while FEIT performance was not significantly associated with any functioning measure. In terms of symptoms, Prosody Task performance was negatively associated with disorganization in the BPRS, and FEIT performance was associated with disorganization, reality distortion, and positive symptoms.
“These findings are consistent with the view that emotion recognition deficits could be contributing to deficits in the ability of people with first-episode schizophrenia to adequately function in the real world, both in relationships with friends and in normative young adult roles,” the investigators wrote.
Dr. Bonfils reported no conflicts of interest. Three coauthors reported receiving support, research grants, and funding from several pharmaceutical companies.
SOURCE: Bonfils KA et al. Schizophr Res Cogn. 2019. doi: 10.1016/j.scog.2019.100153.
Affective prosody recognition is associated with role and social functioning in patients with a recent first episode of schizophrenia, according to Kelsey A. Bonfils, PhD, and associates.
The investigators conducted an analysis of 49 patients aged between 18 and 45 years with a recent first episode of schizophrenia who were participating in a larger randomized, controlled trial. Symptoms of schizophrenia were assessed using a 24-item version of the Brief Psychiatric Rating Scale (BPRS) and functioning was assessed using the Global Functioning Scale (GFS) and Role Functioning Scale (RFS). Study participants took the Prosody Task, which assessed the ability to recognize happiness, sadness, anger, fear, and disgust, and the Facial Emotion Identification Test (FEIT), which assesses the ability to recognize happiness, sadness, anger, fear, surprise, and disgust, reported Dr. Bonfils of the Veterans Affairs Pittsburgh Healthcare System and the department of psychiatry at the University of Pittsburgh. The study was published in Schizophrenia Research: Cognition.
In the Prosody Task, patients were significantly more likely to recognize anger (45.6% correct) and sadness (43.8%), and significantly less likely to recognize disgust (21.9%). In the FEIT, patients were most likely to recognize happiness (97.5%), followed by surprise (90.0%), anger (85.0%), sadness (77.5%), disgust (73.8%), and fear (55.0%).
Performance in the Prosody Task was associated with GFS role functioning and RFS social functioning, while FEIT performance was not significantly associated with any functioning measure. In terms of symptoms, Prosody Task performance was negatively associated with disorganization in the BPRS, and FEIT performance was associated with disorganization, reality distortion, and positive symptoms.
“These findings are consistent with the view that emotion recognition deficits could be contributing to deficits in the ability of people with first-episode schizophrenia to adequately function in the real world, both in relationships with friends and in normative young adult roles,” the investigators wrote.
Dr. Bonfils reported no conflicts of interest. Three coauthors reported receiving support, research grants, and funding from several pharmaceutical companies.
SOURCE: Bonfils KA et al. Schizophr Res Cogn. 2019. doi: 10.1016/j.scog.2019.100153.
FROM SCHIZOPHRENIA RESEARCH: COGNITION
Black-box warnings: How they can improve your clinical practice
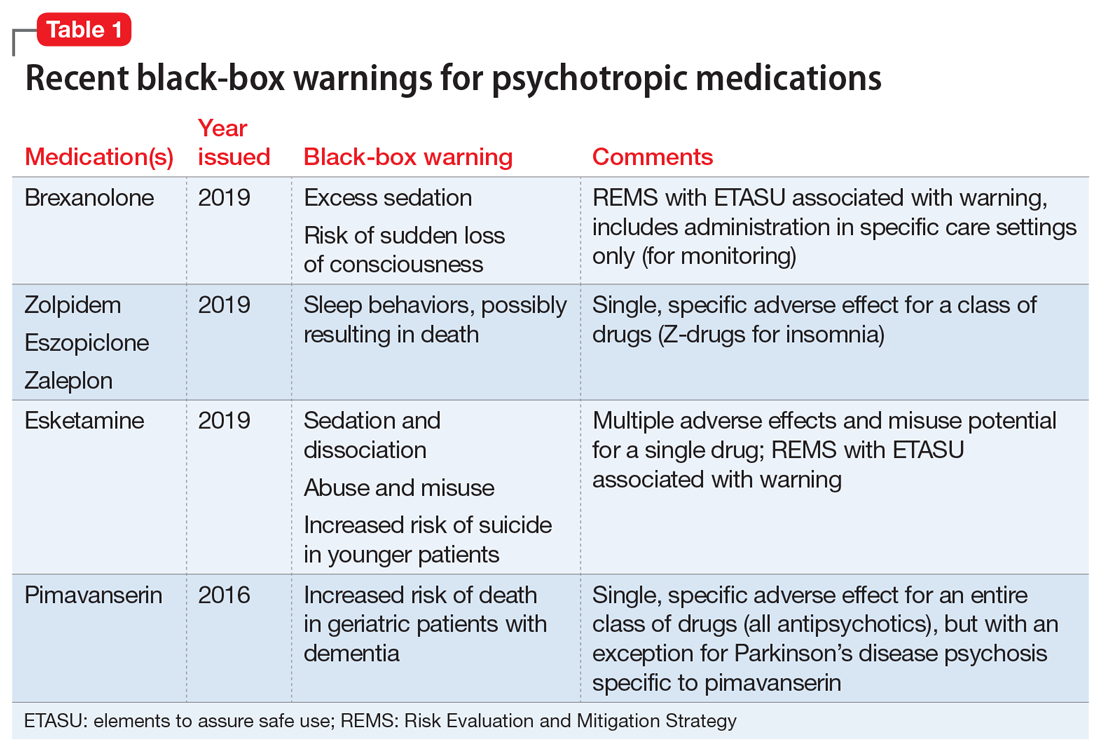
Recently, the FDA issued “black-box” warnings, its most prominent drug safety statements, for esketamine,1 which is indicated for treatment-resistant depression, and the Z-drugs, which are indicated for insomnia2 (Table 1). A black-box warning also comes with brexanolone, which was recently approved for postpartum depression.3 While these newly issued warnings serve as a timely reminder of the importance of black-box warnings, older black-box warnings also cover large areas of psychiatric prescribing, including all medications indicated for treating psychosis or schizophrenia (increased mortality in patients with dementia), and all psychotropic medications with a depression indication (suicidality in younger people).
In this article, we help busy prescribers navigate the landscape of black-box warnings by providing a concise review of how to use them in clinical practice, and where to find information to keep up-to-date.
What are black-box warnings?
A black-box warning is a summary of the potential serious or life-threatening risks of a specific prescription medication. The black-box warning is formatted within a black border found at the top of the manufacturer’s prescribing information document (also known as the package insert or product label). Below the black-box warning, potential risks appear in descending order in sections titled “Contraindications,” “Warnings and Precautions,” and “Adverse Reactions.”4 The FDA issues black-box warnings either during drug development, to take effect upon approval of a new agent, or (more commonly) based on post-marketing safety information,5 which the FDA continuously gathers from reports by patients, clinicians, and industry.6 Federal law mandates the existence of black-box warnings, stating in part that, “special problems, particularly those that may lead to death or serious injury, may be required by the [FDA] to be placed in a prominently displayed box” (21 CFR 201.57(e)).
When is a black-box warning necessary?
The FDA issues a black-box warning based upon its judgment of the seriousness of the adverse effect. However, by definition, these risks do not inherently outweigh the benefits a medication may offer to certain patients. According to the FDA,7 black-box warnings are placed when:
- an adverse reaction so significant exists that this potential negative effect must be considered in risks and benefits when prescribing the medication
- a serious adverse reaction exists that can be prevented, or the risk reduced, by appropriate use of the medication
- the FDA has approved the medication with restrictions to ensure safe use.
Table 2 shows examples of scenarios where black-box warnings have been issued.8 Black-box warnings may be placed on an individual agent or on an entire class of medications. For example, both antipsychotics and antidepressants have class-wide warnings. Finally, black-box warnings are not static, and their content may change; in a study of black-box warnings issued from 2007 to 2015, 29% were entirely new, 32% were considered major updates to existing black-box warnings, and 40% were minor updates.5

Critiques of black-box warnings focus on the absence of published, formal criteria for instituting such warnings, the lack of a consistent approach in their content, and the infrequent inclusion of any information on the relative size of the risk.9 Suggestions for improvement include offering guidance on how to implement the black-box warnings in a patient-centered, shared decision-making model by adding evidence profiles and implementation guides.10 Less frequently considered, black-box warnings may be discontinued if new evidence demonstrates that the risk is lower than previously appreciated; however, similarly to their placement, no explicit criteria for the removal of black-box warnings have been made public.11
When a medication poses an especially high safety risk, the FDA may require the manufacturer to implement a Risk Evaluation and Mitigation Strategy (REMS) program. These programs can describe specific steps to improve medication safety, known as elements to assure safe use (ETASU).4 A familiar example is the clozapine REMS. In order to reduce the risk of severe neutropenia, the clozapine REMS requires prescribers (and pharmacists) to complete specialized training (making up the ETASU). Surprisingly, not every medication with a REMS has a corresponding black-box warning12; more understandably, many medications with black-box warnings do not have an associated REMS, because their risks are evaluated to be manageable by an individual prescriber’s clinical judgment. Most recently, esketamine carries both a black-box warning and a REMS. The black-box warning focuses on adverse effects (Table 1), while the REMS focuses on specific steps used to lessen these risks, including requiring use of a patient enrollment and monitoring form, a fact sheet for patients, and health care setting and pharmacy enrollment forms.13
Continue to: Psychotropic medications and black-box warnings
Psychotropic medications and black-box warnings
Psychotropic medications have a large number of black-box warnings.14 Because it is difficult to find black-box warnings for multiple medications in one place, we have provided 2 convenient resources to address this gap: a concise summary guide (Table 3) and a more detailed database (Table 4, Table 5, Table 6, Table 7, and Table 8). In these Tables, the possible risk mitigations, off-label uses, and monitoring are not meant to be formal recommendations or endorsements but are for independent clinician consideration only.
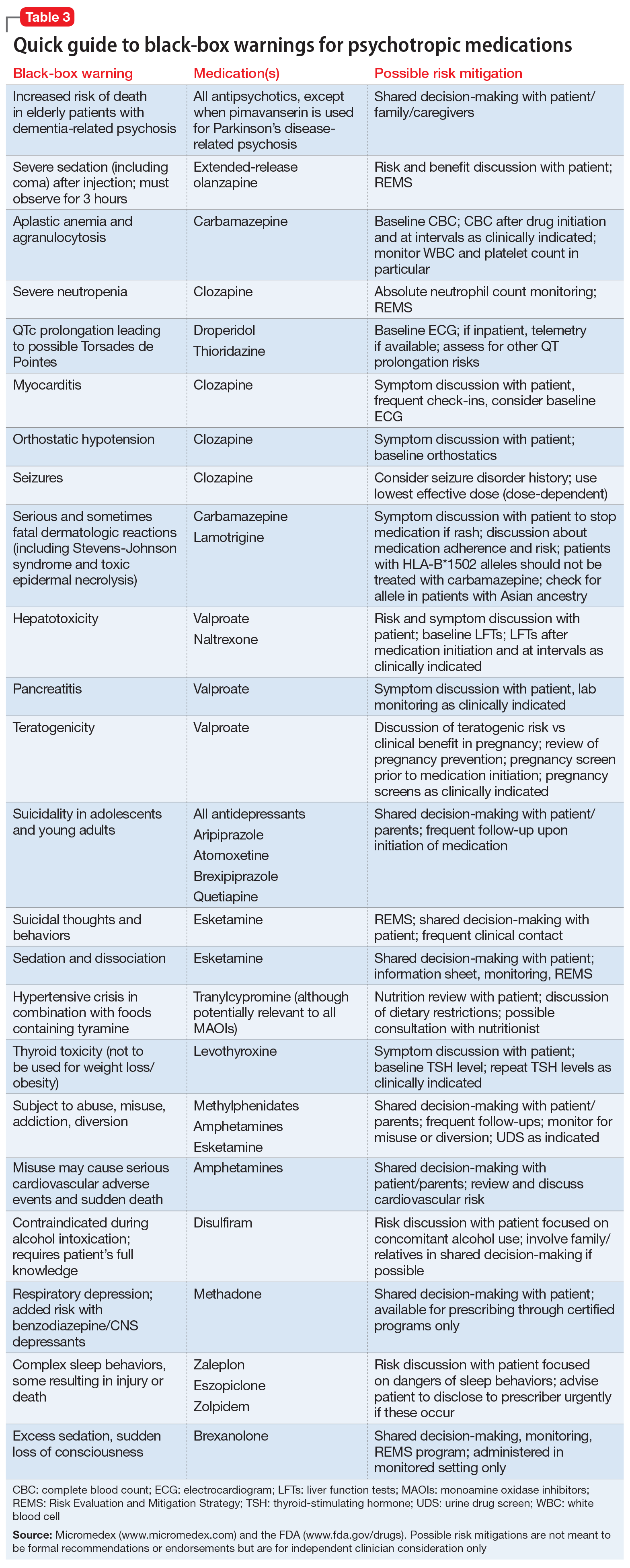
The information in these Tables was drawn from publicly available data, primarily the Micromedex and FDA web sites (see Related Resources). Because this information changes over time, at the end of this article we suggest ways for clinicians to stay updated with black-box warnings and build on the information provided in this article. These tools can be useful for day-to-day clinical practice in addition to studying for professional examinations. The following are selected high-profile black-box warnings.
Antidepressants and suicide risk. As a class, antidepressants carry a black-box warning on suicide risk in patients age ≤24. Initially issued in 2005, this warning was extended in 2007 to indicate that depression itself is associated with an increased risk of suicide. This black-box warning is used for an entire class of medications as well as for a specific patient population (age ≤24). Moreover, it indicates that suicide rates in patients age >65 were lower among patients using antidepressants.
Among psychotropic medication black-box warnings, this warning has perhaps been the most controversial. For example, it has been suggested that this black-box warning may have inadvertently increased suicide rates by discouraging clinicians from prescribing antidepressants,15 although this also has been called into question.16 This black-box warning illustrates that the consequences of issuing black-box warnings can be very difficult to assess, which makes their clinical effects highly complex and challenging to evaluate.14
Antipsychotics and dementia-related psychosis. This warning was initially issued in 2005 for second-generation antipsychotics and extended to first-generation antipsychotics in 2008. Antipsychotics as a class carry a black-box warning for increased risk of death in patients with dementia (major neurocognitive disorder). This warning extends to the recently approved antipsychotic pimavanserin, even though this agent’s proposed mechanism of action differs from that of other antipsychotics.17 However, it specifically allows for use in Parkinson’s disease psychosis, which is pimavanserin’s indication.18 In light of recent research suggesting pimavanserin is effective in dementia-related psychosis,19 it bears watching whether this agent becomes the first antipsychotic to have this warning removed.
Continue to: This class warning has...
This class warning has had widespread effects. For example, it has prompted less use of antipsychotics in nursing home facilities, as a result of stricter Centers for Medicare and Medicaid Services regulations20; overall, there is some evidence that there has been reduced prescribing of antipsychotics in general.21 Additionally, this black-box warning is unusual in that it warns about a specific off-label indication, which is itself poorly supported by evidence.21 Concomitantly, few other treatment options are available for this clinical situation. These medications are often seen as the only option for patients with dementia complicated by severe behavioral disturbance, and thus this black-box warning reflects real-world practices.14
Varenicline and neuropsychiatric complications. The withdrawal of the black-box warning on potential neuropsychiatric complications of using varenicline for smoking cessation shows that black-box warnings are not static and can, though infrequently, be removed as more safety data accumulates.11 As additional post-marketing information emerged on this risk, this black-box warning was reconsidered and withdrawn in 2016.22 Its withdrawal could potentially make clinicians more comfortable prescribing varenicline and in turn, help to reduce smoking rates.
How to use black-box warnings
To enhance their clinical practice, prescribers can use black-box warnings to inform safe prescribing practices, to guide shared decision-making, and to improve documentation of their treatment decisions.
Informing safe prescribing practices. A prescriber should be aware of the main safety concerns contained in a medication’s black-box warning; at the same time, these warnings are not meant to unduly limit use when crucial treatment is needed.14 In issuing a black-box warning, the FDA has clearly stated the priority and seriousness of its concern. These safety issues must be balanced against the medication’s utility for a given patient, at the prescriber’s clinical judgment.
Guiding shared decision-making. Clinicians are not required to disclose black-box warnings to patients, and there are no criteria that clearly define the role of these warnings in patient care. As is often noted, the FDA does not regulate the practice of medicine.6 However, given the seriousness of the potential adverse effects delineated by black-box warnings, it is reasonable for clinicians to have a solid grasp of black-box warnings for all medications they prescribe, and to be able to relate these warnings to patients, in appropriate language. This patient-centered discussion should include weighing the risks and benefits with the patient and educating the patient about the risks and strategies to mitigate those risks. This discussion can be augmented by patient handouts, which are often offered by pharmaceutical manufacturers, and by shared decision-making tools. A proactive discussion with patients and families about black-box warnings and other risks discussed in product labels can help reduce fears associated with taking medications and may improve adherence.
Continue to: Improving documentation of treatment decisions
Improving documentation of treatment decisions. Fluent knowledge of black-box warnings may help clinicians improve documentation of their treatment decisions, particularly the risks and benefits of their medication choices. Fluency with black-box warnings will help clinicians accurately document both their awareness of these risks, and how these risks informed their risk-benefit analysis in specific clinical situations.
Despite the clear importance the FDA places on black-box warnings, they are not often a topic of study in training or in postgraduate continuing education, and as a result, not all clinicians may be equally conversant with black-box warnings. While black-box warnings do change over time, many psychotropic medication black-box warnings are long-standing and well-established, and they evolve slowly enough to make mastering these warnings worthwhile in order to make the most informed clinical decisions for patient care.
Keeping up-to-date
There are practical and useful ways for busy clinicians to stay up-to-date with black-box warnings. Although these resources exist in multiple locations, together they provide convenient ways to keep current.
The FDA provides access to black-box warnings via its comprehensive database, DRUGS@FDA (https://www.accessdata.fda.gov/scripts/cder/daf/). Detailed information about REMS (and corresponding ETASU and other information related to REMS programs) is available at REMS@FDA (https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm). Clinicians can make safety reports that may contribute to FDA decision-making on black-box warnings by contacting MedWatch (https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program), the FDA’s adverse events reporting system. MedWatch releases safety information reports, which can be followed on Twitter @FDAMedWatch. Note that FDA information generally is organized by specific drug, and not into categories, such as psychotropic medications.
BlackBoxRx (www.blackboxrx.com) is a subscription-based web service that some clinicians may have access to via facility or academic resources as part of a larger FormWeb software package. Individuals also can subscribe (currently, $89/year).
Continue to: Micromedex
Micromedex (www.micromedex.com), which is widely available through medical libraries, is a subscription-based web service that provides black-box warning information from a separate tab that is easily accessed in each drug’s information front page. There is also an alphabetical list of black-box warnings under a separate tab on the Micromedex landing page.
ePocrates (www.epocrates.com) is a subscription-based service that provides extensive drug information, including black-box warnings, in a convenient mobile app.
Bottom Line
Black-box warnings are the most prominent drug safety warnings issued by the FDA. Many psychotropic medications carry black-box warnings that are crucial to everyday psychiatric prescribing. A better understanding of blackbox warnings can enhance your clinical practice by informing safe prescribing practices, guiding shared decision-making, and improving documentation of your treatment decisions.
Related Resources
- US Food and Drug Administration. DRUGS@FDA: FDAapproved drug products. www.accessdata.fda.gov/scripts/cder/daf/.
- US Food and Drug Administration. Drug safety and availability. www.fda.gov/drugs/drug-safety-and-availability. Updated October 10, 2019.
- BlackBoxRx. www.blackboxrx.com. (Subscription required.)
- Mircromedex. www.micromedex.com. (Subscription required.)
- ePocrates. www.epocrates.com. (Subscription required.)
Drug Brand Names
Amitriptyline • Elavil, Vanatrip
Amoxatine • Strattera
Amoxapine • Asendin
Aripiprazole • Abilify
Asenapine • Saphris
Brexanolone • Zulresso
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dexmethylphenidate • Focalin
Dextroamphetamine/amphetamine • Adderall
Disulfiram • Antabuse
Doxepin • Prudoxin, Silenor
Droperidol • Inapsine
Duloxetine • Cymbalta
Escitalopram • Lexapro
Esketamine • Spravato
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Haloperidol • Haldol
Iloperidone • Fanapt
Imipramine • Tofranil
Isocarboxazid • Marplan
Lamotrigine • Lamictal
Levomilnacipran • Fetzima
Levothyroxine • Synthroid
Linezolid • Zyvox
Lisdexamfetamine • Vyvanse
Lithium • Eskalith, Lithobid
Loxapine • Loxitane
Lurasidone • Latuda
Maprotiline • Ludiomil
Methadone • Dolophine, Methadose
Methylphenidate • Ritalin, Concerta
Midazolam • Versed
Milnacipran • Savella
Mirtazapine • Remeron
Naltrexone • Revia, Vivitrol
Nefazodone • Serzone
Nortriptyline • Aventyl, Pamelor
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Perphenazine • Trilafon
Phenelzine • Nardil
Pimavanserin • Nuplazid
Prochlorperazine • Compro
Protriptyline • Vivactil
Quetiapine • Seroquel
Risperidone • Risperdal
Selegiline • Emsam
Sertraline • Zoloft
Thioridazine • Mellaril
Thiothixene • Navane
Tranylcypromine • Parnate
Trazodone • Desyrel, Oleptro
Trifluoperazine • Stelazine
Trimipramine • Surmontil
Valproate • Depakote
Varenicline • Chantix, Wellbutrin
Vilazodone • Viibryd
Venlafaxine • Effexor
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
1. Spravato [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
2. U.S. Food and Drug Administration. FDA drug safety announcement: FDA adds boxed warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. https://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Published April 30, 2019. Accessed October 28, 2019.
3. Zulresso [package insert]. Cambridge, Mass.: Sage Therapeutics Inc.; 2019.
4. Gassman AL, Nguyen CP, Joffe HV. FDA regulation of prescription drugs. N Engl J Med. 2017;376(7):674-682.
5. Solotke MT, Dhruva SS, Downing NS, et al. New and incremental FDA black box warnings from 2008 to 2015. Expert Opin Drug Saf. 2018;17(2):117-123.
6. Murphy S, Roberts R. “Black box” 101: how the Food and Drug Administration evaluates, communicates, and manages drug benefit/risk. J Allergy Clin Immunol. 2006;117(1):34-39.
7. U.S. Food and Drug Administration. Guidance document: Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products – content and format. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/warnings-and-precautions-contraindications-and-boxed-warning-sections-labeling-human-prescription. Published October 2011. Accessed October 28, 2019.
8. Beach JE, Faich GA, Bormel FG, et al. Black box warnings in prescription drug labeling: results of a survey of 206 drugs. Food Drug Law J. 1998;53(3):403-411.
9. Matlock A, Allan N, Wills B, et al. A continuing black hole? The FDA boxed warning: an appeal to improve its clinical utility. Clinical Toxicol (Phila). 2011;49(6):443-447.
10. Elraiyah T, Gionfriddo MR, Montori VM, et al. Content, consistency, and quality of black box warnings: time for a change. Ann Intern Med. 2015;163(11):875-876.
11. Yeh JS, Sarpatwari A, Kesselheim AS. Ethical and practical considerations in removing black box warnings from drug labels. Drug Saf. 2016;39(8):709-714.
12. Boudes PF. Risk Evaluation and Mitigation Strategies (REMSs): are they improving drug safety? A critical review of REMSs requiring Elements to Assure Safe Use (ETASU). Drugs R D. 2017;17(2):245-254.
13. U.S. Food and Drug Administration. Approved risk evaluation mitigation strategies (REMS): Spravato (esketamine) REMS program. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386. Updated June 25, 2019. Accessed October 28, 2018.
14. Stevens JR, Jarrahzadeh T, Brendel RW, et al. Strategies for the prescription of psychotropic drugs with black box warnings. Psychosomatics. 2014;55(2):123-133.
15. Friedman RA. Antidepressants’ black-box warning--10 years later. N Engl J Med. 2014;371(18):1666-1668.
16. Stone MB. The FDA warning on antidepressants and suicidality--why the controversy? N Engl J Med. 2014;371(18):1668-1671.
17. Mathis MV, Muoio BM, Andreason P, et al. The US Food and Drug Administration’s perspective on the new antipsychotic pimavanserin. J Clin Psychiatry. 2017;78(6):e668-e673. doi: 10.4088/JCP.16r11119.
18. Nuplazid [package insert]. San Diego, CA: Acadia Pharmaceuticals Inc.; May 2019.
19. Ballard C, Banister C, Khan Z, et al. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in patients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
20. Maust DT, Kim HM, Chiang C, et al. Association of the Centers for Medicare & Medicaid Services’ National Partnership to Improve Dementia Care with the use of antipsychotics and other psychotropics in long-term care in the United States from 2009 to 2014. JAMA Intern Med. 2018;178(5):640-647.
21. Dorsey ER, Rabbani A, Gallagher SA, et al. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170(1):96-103.
22. U.S. Food and Drug Administration. FDA drug safety communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking. Published December 16, 2016. Accessed October 28, 2019.
Recently, the FDA issued “black-box” warnings, its most prominent drug safety statements, for esketamine,1 which is indicated for treatment-resistant depression, and the Z-drugs, which are indicated for insomnia2 (Table 1). A black-box warning also comes with brexanolone, which was recently approved for postpartum depression.3 While these newly issued warnings serve as a timely reminder of the importance of black-box warnings, older black-box warnings also cover large areas of psychiatric prescribing, including all medications indicated for treating psychosis or schizophrenia (increased mortality in patients with dementia), and all psychotropic medications with a depression indication (suicidality in younger people).

In this article, we help busy prescribers navigate the landscape of black-box warnings by providing a concise review of how to use them in clinical practice, and where to find information to keep up-to-date.
What are black-box warnings?
A black-box warning is a summary of the potential serious or life-threatening risks of a specific prescription medication. The black-box warning is formatted within a black border found at the top of the manufacturer’s prescribing information document (also known as the package insert or product label). Below the black-box warning, potential risks appear in descending order in sections titled “Contraindications,” “Warnings and Precautions,” and “Adverse Reactions.”4 The FDA issues black-box warnings either during drug development, to take effect upon approval of a new agent, or (more commonly) based on post-marketing safety information,5 which the FDA continuously gathers from reports by patients, clinicians, and industry.6 Federal law mandates the existence of black-box warnings, stating in part that, “special problems, particularly those that may lead to death or serious injury, may be required by the [FDA] to be placed in a prominently displayed box” (21 CFR 201.57(e)).
When is a black-box warning necessary?
The FDA issues a black-box warning based upon its judgment of the seriousness of the adverse effect. However, by definition, these risks do not inherently outweigh the benefits a medication may offer to certain patients. According to the FDA,7 black-box warnings are placed when:
- an adverse reaction so significant exists that this potential negative effect must be considered in risks and benefits when prescribing the medication
- a serious adverse reaction exists that can be prevented, or the risk reduced, by appropriate use of the medication
- the FDA has approved the medication with restrictions to ensure safe use.
Table 2 shows examples of scenarios where black-box warnings have been issued.8 Black-box warnings may be placed on an individual agent or on an entire class of medications. For example, both antipsychotics and antidepressants have class-wide warnings. Finally, black-box warnings are not static, and their content may change; in a study of black-box warnings issued from 2007 to 2015, 29% were entirely new, 32% were considered major updates to existing black-box warnings, and 40% were minor updates.5

Critiques of black-box warnings focus on the absence of published, formal criteria for instituting such warnings, the lack of a consistent approach in their content, and the infrequent inclusion of any information on the relative size of the risk.9 Suggestions for improvement include offering guidance on how to implement the black-box warnings in a patient-centered, shared decision-making model by adding evidence profiles and implementation guides.10 Less frequently considered, black-box warnings may be discontinued if new evidence demonstrates that the risk is lower than previously appreciated; however, similarly to their placement, no explicit criteria for the removal of black-box warnings have been made public.11
When a medication poses an especially high safety risk, the FDA may require the manufacturer to implement a Risk Evaluation and Mitigation Strategy (REMS) program. These programs can describe specific steps to improve medication safety, known as elements to assure safe use (ETASU).4 A familiar example is the clozapine REMS. In order to reduce the risk of severe neutropenia, the clozapine REMS requires prescribers (and pharmacists) to complete specialized training (making up the ETASU). Surprisingly, not every medication with a REMS has a corresponding black-box warning12; more understandably, many medications with black-box warnings do not have an associated REMS, because their risks are evaluated to be manageable by an individual prescriber’s clinical judgment. Most recently, esketamine carries both a black-box warning and a REMS. The black-box warning focuses on adverse effects (Table 1), while the REMS focuses on specific steps used to lessen these risks, including requiring use of a patient enrollment and monitoring form, a fact sheet for patients, and health care setting and pharmacy enrollment forms.13
Continue to: Psychotropic medications and black-box warnings
Psychotropic medications and black-box warnings
Psychotropic medications have a large number of black-box warnings.14 Because it is difficult to find black-box warnings for multiple medications in one place, we have provided 2 convenient resources to address this gap: a concise summary guide (Table 3) and a more detailed database (Table 4, Table 5, Table 6, Table 7, and Table 8). In these Tables, the possible risk mitigations, off-label uses, and monitoring are not meant to be formal recommendations or endorsements but are for independent clinician consideration only.

The information in these Tables was drawn from publicly available data, primarily the Micromedex and FDA web sites (see Related Resources). Because this information changes over time, at the end of this article we suggest ways for clinicians to stay updated with black-box warnings and build on the information provided in this article. These tools can be useful for day-to-day clinical practice in addition to studying for professional examinations. The following are selected high-profile black-box warnings.
Antidepressants and suicide risk. As a class, antidepressants carry a black-box warning on suicide risk in patients age ≤24. Initially issued in 2005, this warning was extended in 2007 to indicate that depression itself is associated with an increased risk of suicide. This black-box warning is used for an entire class of medications as well as for a specific patient population (age ≤24). Moreover, it indicates that suicide rates in patients age >65 were lower among patients using antidepressants.
Among psychotropic medication black-box warnings, this warning has perhaps been the most controversial. For example, it has been suggested that this black-box warning may have inadvertently increased suicide rates by discouraging clinicians from prescribing antidepressants,15 although this also has been called into question.16 This black-box warning illustrates that the consequences of issuing black-box warnings can be very difficult to assess, which makes their clinical effects highly complex and challenging to evaluate.14
Antipsychotics and dementia-related psychosis. This warning was initially issued in 2005 for second-generation antipsychotics and extended to first-generation antipsychotics in 2008. Antipsychotics as a class carry a black-box warning for increased risk of death in patients with dementia (major neurocognitive disorder). This warning extends to the recently approved antipsychotic pimavanserin, even though this agent’s proposed mechanism of action differs from that of other antipsychotics.17 However, it specifically allows for use in Parkinson’s disease psychosis, which is pimavanserin’s indication.18 In light of recent research suggesting pimavanserin is effective in dementia-related psychosis,19 it bears watching whether this agent becomes the first antipsychotic to have this warning removed.
Continue to: This class warning has...
This class warning has had widespread effects. For example, it has prompted less use of antipsychotics in nursing home facilities, as a result of stricter Centers for Medicare and Medicaid Services regulations20; overall, there is some evidence that there has been reduced prescribing of antipsychotics in general.21 Additionally, this black-box warning is unusual in that it warns about a specific off-label indication, which is itself poorly supported by evidence.21 Concomitantly, few other treatment options are available for this clinical situation. These medications are often seen as the only option for patients with dementia complicated by severe behavioral disturbance, and thus this black-box warning reflects real-world practices.14
Varenicline and neuropsychiatric complications. The withdrawal of the black-box warning on potential neuropsychiatric complications of using varenicline for smoking cessation shows that black-box warnings are not static and can, though infrequently, be removed as more safety data accumulates.11 As additional post-marketing information emerged on this risk, this black-box warning was reconsidered and withdrawn in 2016.22 Its withdrawal could potentially make clinicians more comfortable prescribing varenicline and in turn, help to reduce smoking rates.
How to use black-box warnings
To enhance their clinical practice, prescribers can use black-box warnings to inform safe prescribing practices, to guide shared decision-making, and to improve documentation of their treatment decisions.
Informing safe prescribing practices. A prescriber should be aware of the main safety concerns contained in a medication’s black-box warning; at the same time, these warnings are not meant to unduly limit use when crucial treatment is needed.14 In issuing a black-box warning, the FDA has clearly stated the priority and seriousness of its concern. These safety issues must be balanced against the medication’s utility for a given patient, at the prescriber’s clinical judgment.
Guiding shared decision-making. Clinicians are not required to disclose black-box warnings to patients, and there are no criteria that clearly define the role of these warnings in patient care. As is often noted, the FDA does not regulate the practice of medicine.6 However, given the seriousness of the potential adverse effects delineated by black-box warnings, it is reasonable for clinicians to have a solid grasp of black-box warnings for all medications they prescribe, and to be able to relate these warnings to patients, in appropriate language. This patient-centered discussion should include weighing the risks and benefits with the patient and educating the patient about the risks and strategies to mitigate those risks. This discussion can be augmented by patient handouts, which are often offered by pharmaceutical manufacturers, and by shared decision-making tools. A proactive discussion with patients and families about black-box warnings and other risks discussed in product labels can help reduce fears associated with taking medications and may improve adherence.
Continue to: Improving documentation of treatment decisions
Improving documentation of treatment decisions. Fluent knowledge of black-box warnings may help clinicians improve documentation of their treatment decisions, particularly the risks and benefits of their medication choices. Fluency with black-box warnings will help clinicians accurately document both their awareness of these risks, and how these risks informed their risk-benefit analysis in specific clinical situations.
Despite the clear importance the FDA places on black-box warnings, they are not often a topic of study in training or in postgraduate continuing education, and as a result, not all clinicians may be equally conversant with black-box warnings. While black-box warnings do change over time, many psychotropic medication black-box warnings are long-standing and well-established, and they evolve slowly enough to make mastering these warnings worthwhile in order to make the most informed clinical decisions for patient care.
Keeping up-to-date
There are practical and useful ways for busy clinicians to stay up-to-date with black-box warnings. Although these resources exist in multiple locations, together they provide convenient ways to keep current.
The FDA provides access to black-box warnings via its comprehensive database, DRUGS@FDA (https://www.accessdata.fda.gov/scripts/cder/daf/). Detailed information about REMS (and corresponding ETASU and other information related to REMS programs) is available at REMS@FDA (https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm). Clinicians can make safety reports that may contribute to FDA decision-making on black-box warnings by contacting MedWatch (https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program), the FDA’s adverse events reporting system. MedWatch releases safety information reports, which can be followed on Twitter @FDAMedWatch. Note that FDA information generally is organized by specific drug, and not into categories, such as psychotropic medications.
BlackBoxRx (www.blackboxrx.com) is a subscription-based web service that some clinicians may have access to via facility or academic resources as part of a larger FormWeb software package. Individuals also can subscribe (currently, $89/year).
Continue to: Micromedex
Micromedex (www.micromedex.com), which is widely available through medical libraries, is a subscription-based web service that provides black-box warning information from a separate tab that is easily accessed in each drug’s information front page. There is also an alphabetical list of black-box warnings under a separate tab on the Micromedex landing page.
ePocrates (www.epocrates.com) is a subscription-based service that provides extensive drug information, including black-box warnings, in a convenient mobile app.
Bottom Line
Black-box warnings are the most prominent drug safety warnings issued by the FDA. Many psychotropic medications carry black-box warnings that are crucial to everyday psychiatric prescribing. A better understanding of blackbox warnings can enhance your clinical practice by informing safe prescribing practices, guiding shared decision-making, and improving documentation of your treatment decisions.
Related Resources
- US Food and Drug Administration. DRUGS@FDA: FDAapproved drug products. www.accessdata.fda.gov/scripts/cder/daf/.
- US Food and Drug Administration. Drug safety and availability. www.fda.gov/drugs/drug-safety-and-availability. Updated October 10, 2019.
- BlackBoxRx. www.blackboxrx.com. (Subscription required.)
- Mircromedex. www.micromedex.com. (Subscription required.)
- ePocrates. www.epocrates.com. (Subscription required.)
Drug Brand Names
Amitriptyline • Elavil, Vanatrip
Amoxatine • Strattera
Amoxapine • Asendin
Aripiprazole • Abilify
Asenapine • Saphris
Brexanolone • Zulresso
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dexmethylphenidate • Focalin
Dextroamphetamine/amphetamine • Adderall
Disulfiram • Antabuse
Doxepin • Prudoxin, Silenor
Droperidol • Inapsine
Duloxetine • Cymbalta
Escitalopram • Lexapro
Esketamine • Spravato
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Haloperidol • Haldol
Iloperidone • Fanapt
Imipramine • Tofranil
Isocarboxazid • Marplan
Lamotrigine • Lamictal
Levomilnacipran • Fetzima
Levothyroxine • Synthroid
Linezolid • Zyvox
Lisdexamfetamine • Vyvanse
Lithium • Eskalith, Lithobid
Loxapine • Loxitane
Lurasidone • Latuda
Maprotiline • Ludiomil
Methadone • Dolophine, Methadose
Methylphenidate • Ritalin, Concerta
Midazolam • Versed
Milnacipran • Savella
Mirtazapine • Remeron
Naltrexone • Revia, Vivitrol
Nefazodone • Serzone
Nortriptyline • Aventyl, Pamelor
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Perphenazine • Trilafon
Phenelzine • Nardil
Pimavanserin • Nuplazid
Prochlorperazine • Compro
Protriptyline • Vivactil
Quetiapine • Seroquel
Risperidone • Risperdal
Selegiline • Emsam
Sertraline • Zoloft
Thioridazine • Mellaril
Thiothixene • Navane
Tranylcypromine • Parnate
Trazodone • Desyrel, Oleptro
Trifluoperazine • Stelazine
Trimipramine • Surmontil
Valproate • Depakote
Varenicline • Chantix, Wellbutrin
Vilazodone • Viibryd
Venlafaxine • Effexor
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
Recently, the FDA issued “black-box” warnings, its most prominent drug safety statements, for esketamine,1 which is indicated for treatment-resistant depression, and the Z-drugs, which are indicated for insomnia2 (Table 1). A black-box warning also comes with brexanolone, which was recently approved for postpartum depression.3 While these newly issued warnings serve as a timely reminder of the importance of black-box warnings, older black-box warnings also cover large areas of psychiatric prescribing, including all medications indicated for treating psychosis or schizophrenia (increased mortality in patients with dementia), and all psychotropic medications with a depression indication (suicidality in younger people).

In this article, we help busy prescribers navigate the landscape of black-box warnings by providing a concise review of how to use them in clinical practice, and where to find information to keep up-to-date.
What are black-box warnings?
A black-box warning is a summary of the potential serious or life-threatening risks of a specific prescription medication. The black-box warning is formatted within a black border found at the top of the manufacturer’s prescribing information document (also known as the package insert or product label). Below the black-box warning, potential risks appear in descending order in sections titled “Contraindications,” “Warnings and Precautions,” and “Adverse Reactions.”4 The FDA issues black-box warnings either during drug development, to take effect upon approval of a new agent, or (more commonly) based on post-marketing safety information,5 which the FDA continuously gathers from reports by patients, clinicians, and industry.6 Federal law mandates the existence of black-box warnings, stating in part that, “special problems, particularly those that may lead to death or serious injury, may be required by the [FDA] to be placed in a prominently displayed box” (21 CFR 201.57(e)).
When is a black-box warning necessary?
The FDA issues a black-box warning based upon its judgment of the seriousness of the adverse effect. However, by definition, these risks do not inherently outweigh the benefits a medication may offer to certain patients. According to the FDA,7 black-box warnings are placed when:
- an adverse reaction so significant exists that this potential negative effect must be considered in risks and benefits when prescribing the medication
- a serious adverse reaction exists that can be prevented, or the risk reduced, by appropriate use of the medication
- the FDA has approved the medication with restrictions to ensure safe use.
Table 2 shows examples of scenarios where black-box warnings have been issued.8 Black-box warnings may be placed on an individual agent or on an entire class of medications. For example, both antipsychotics and antidepressants have class-wide warnings. Finally, black-box warnings are not static, and their content may change; in a study of black-box warnings issued from 2007 to 2015, 29% were entirely new, 32% were considered major updates to existing black-box warnings, and 40% were minor updates.5

Critiques of black-box warnings focus on the absence of published, formal criteria for instituting such warnings, the lack of a consistent approach in their content, and the infrequent inclusion of any information on the relative size of the risk.9 Suggestions for improvement include offering guidance on how to implement the black-box warnings in a patient-centered, shared decision-making model by adding evidence profiles and implementation guides.10 Less frequently considered, black-box warnings may be discontinued if new evidence demonstrates that the risk is lower than previously appreciated; however, similarly to their placement, no explicit criteria for the removal of black-box warnings have been made public.11
When a medication poses an especially high safety risk, the FDA may require the manufacturer to implement a Risk Evaluation and Mitigation Strategy (REMS) program. These programs can describe specific steps to improve medication safety, known as elements to assure safe use (ETASU).4 A familiar example is the clozapine REMS. In order to reduce the risk of severe neutropenia, the clozapine REMS requires prescribers (and pharmacists) to complete specialized training (making up the ETASU). Surprisingly, not every medication with a REMS has a corresponding black-box warning12; more understandably, many medications with black-box warnings do not have an associated REMS, because their risks are evaluated to be manageable by an individual prescriber’s clinical judgment. Most recently, esketamine carries both a black-box warning and a REMS. The black-box warning focuses on adverse effects (Table 1), while the REMS focuses on specific steps used to lessen these risks, including requiring use of a patient enrollment and monitoring form, a fact sheet for patients, and health care setting and pharmacy enrollment forms.13
Continue to: Psychotropic medications and black-box warnings
Psychotropic medications and black-box warnings
Psychotropic medications have a large number of black-box warnings.14 Because it is difficult to find black-box warnings for multiple medications in one place, we have provided 2 convenient resources to address this gap: a concise summary guide (Table 3) and a more detailed database (Table 4, Table 5, Table 6, Table 7, and Table 8). In these Tables, the possible risk mitigations, off-label uses, and monitoring are not meant to be formal recommendations or endorsements but are for independent clinician consideration only.

The information in these Tables was drawn from publicly available data, primarily the Micromedex and FDA web sites (see Related Resources). Because this information changes over time, at the end of this article we suggest ways for clinicians to stay updated with black-box warnings and build on the information provided in this article. These tools can be useful for day-to-day clinical practice in addition to studying for professional examinations. The following are selected high-profile black-box warnings.
Antidepressants and suicide risk. As a class, antidepressants carry a black-box warning on suicide risk in patients age ≤24. Initially issued in 2005, this warning was extended in 2007 to indicate that depression itself is associated with an increased risk of suicide. This black-box warning is used for an entire class of medications as well as for a specific patient population (age ≤24). Moreover, it indicates that suicide rates in patients age >65 were lower among patients using antidepressants.
Among psychotropic medication black-box warnings, this warning has perhaps been the most controversial. For example, it has been suggested that this black-box warning may have inadvertently increased suicide rates by discouraging clinicians from prescribing antidepressants,15 although this also has been called into question.16 This black-box warning illustrates that the consequences of issuing black-box warnings can be very difficult to assess, which makes their clinical effects highly complex and challenging to evaluate.14
Antipsychotics and dementia-related psychosis. This warning was initially issued in 2005 for second-generation antipsychotics and extended to first-generation antipsychotics in 2008. Antipsychotics as a class carry a black-box warning for increased risk of death in patients with dementia (major neurocognitive disorder). This warning extends to the recently approved antipsychotic pimavanserin, even though this agent’s proposed mechanism of action differs from that of other antipsychotics.17 However, it specifically allows for use in Parkinson’s disease psychosis, which is pimavanserin’s indication.18 In light of recent research suggesting pimavanserin is effective in dementia-related psychosis,19 it bears watching whether this agent becomes the first antipsychotic to have this warning removed.
Continue to: This class warning has...
This class warning has had widespread effects. For example, it has prompted less use of antipsychotics in nursing home facilities, as a result of stricter Centers for Medicare and Medicaid Services regulations20; overall, there is some evidence that there has been reduced prescribing of antipsychotics in general.21 Additionally, this black-box warning is unusual in that it warns about a specific off-label indication, which is itself poorly supported by evidence.21 Concomitantly, few other treatment options are available for this clinical situation. These medications are often seen as the only option for patients with dementia complicated by severe behavioral disturbance, and thus this black-box warning reflects real-world practices.14
Varenicline and neuropsychiatric complications. The withdrawal of the black-box warning on potential neuropsychiatric complications of using varenicline for smoking cessation shows that black-box warnings are not static and can, though infrequently, be removed as more safety data accumulates.11 As additional post-marketing information emerged on this risk, this black-box warning was reconsidered and withdrawn in 2016.22 Its withdrawal could potentially make clinicians more comfortable prescribing varenicline and in turn, help to reduce smoking rates.
How to use black-box warnings
To enhance their clinical practice, prescribers can use black-box warnings to inform safe prescribing practices, to guide shared decision-making, and to improve documentation of their treatment decisions.
Informing safe prescribing practices. A prescriber should be aware of the main safety concerns contained in a medication’s black-box warning; at the same time, these warnings are not meant to unduly limit use when crucial treatment is needed.14 In issuing a black-box warning, the FDA has clearly stated the priority and seriousness of its concern. These safety issues must be balanced against the medication’s utility for a given patient, at the prescriber’s clinical judgment.
Guiding shared decision-making. Clinicians are not required to disclose black-box warnings to patients, and there are no criteria that clearly define the role of these warnings in patient care. As is often noted, the FDA does not regulate the practice of medicine.6 However, given the seriousness of the potential adverse effects delineated by black-box warnings, it is reasonable for clinicians to have a solid grasp of black-box warnings for all medications they prescribe, and to be able to relate these warnings to patients, in appropriate language. This patient-centered discussion should include weighing the risks and benefits with the patient and educating the patient about the risks and strategies to mitigate those risks. This discussion can be augmented by patient handouts, which are often offered by pharmaceutical manufacturers, and by shared decision-making tools. A proactive discussion with patients and families about black-box warnings and other risks discussed in product labels can help reduce fears associated with taking medications and may improve adherence.
Continue to: Improving documentation of treatment decisions
Improving documentation of treatment decisions. Fluent knowledge of black-box warnings may help clinicians improve documentation of their treatment decisions, particularly the risks and benefits of their medication choices. Fluency with black-box warnings will help clinicians accurately document both their awareness of these risks, and how these risks informed their risk-benefit analysis in specific clinical situations.
Despite the clear importance the FDA places on black-box warnings, they are not often a topic of study in training or in postgraduate continuing education, and as a result, not all clinicians may be equally conversant with black-box warnings. While black-box warnings do change over time, many psychotropic medication black-box warnings are long-standing and well-established, and they evolve slowly enough to make mastering these warnings worthwhile in order to make the most informed clinical decisions for patient care.
Keeping up-to-date
There are practical and useful ways for busy clinicians to stay up-to-date with black-box warnings. Although these resources exist in multiple locations, together they provide convenient ways to keep current.
The FDA provides access to black-box warnings via its comprehensive database, DRUGS@FDA (https://www.accessdata.fda.gov/scripts/cder/daf/). Detailed information about REMS (and corresponding ETASU and other information related to REMS programs) is available at REMS@FDA (https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm). Clinicians can make safety reports that may contribute to FDA decision-making on black-box warnings by contacting MedWatch (https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program), the FDA’s adverse events reporting system. MedWatch releases safety information reports, which can be followed on Twitter @FDAMedWatch. Note that FDA information generally is organized by specific drug, and not into categories, such as psychotropic medications.
BlackBoxRx (www.blackboxrx.com) is a subscription-based web service that some clinicians may have access to via facility or academic resources as part of a larger FormWeb software package. Individuals also can subscribe (currently, $89/year).
Continue to: Micromedex
Micromedex (www.micromedex.com), which is widely available through medical libraries, is a subscription-based web service that provides black-box warning information from a separate tab that is easily accessed in each drug’s information front page. There is also an alphabetical list of black-box warnings under a separate tab on the Micromedex landing page.
ePocrates (www.epocrates.com) is a subscription-based service that provides extensive drug information, including black-box warnings, in a convenient mobile app.
Bottom Line
Black-box warnings are the most prominent drug safety warnings issued by the FDA. Many psychotropic medications carry black-box warnings that are crucial to everyday psychiatric prescribing. A better understanding of blackbox warnings can enhance your clinical practice by informing safe prescribing practices, guiding shared decision-making, and improving documentation of your treatment decisions.
Related Resources
- US Food and Drug Administration. DRUGS@FDA: FDAapproved drug products. www.accessdata.fda.gov/scripts/cder/daf/.
- US Food and Drug Administration. Drug safety and availability. www.fda.gov/drugs/drug-safety-and-availability. Updated October 10, 2019.
- BlackBoxRx. www.blackboxrx.com. (Subscription required.)
- Mircromedex. www.micromedex.com. (Subscription required.)
- ePocrates. www.epocrates.com. (Subscription required.)
Drug Brand Names
Amitriptyline • Elavil, Vanatrip
Amoxatine • Strattera
Amoxapine • Asendin
Aripiprazole • Abilify
Asenapine • Saphris
Brexanolone • Zulresso
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dexmethylphenidate • Focalin
Dextroamphetamine/amphetamine • Adderall
Disulfiram • Antabuse
Doxepin • Prudoxin, Silenor
Droperidol • Inapsine
Duloxetine • Cymbalta
Escitalopram • Lexapro
Esketamine • Spravato
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Haloperidol • Haldol
Iloperidone • Fanapt
Imipramine • Tofranil
Isocarboxazid • Marplan
Lamotrigine • Lamictal
Levomilnacipran • Fetzima
Levothyroxine • Synthroid
Linezolid • Zyvox
Lisdexamfetamine • Vyvanse
Lithium • Eskalith, Lithobid
Loxapine • Loxitane
Lurasidone • Latuda
Maprotiline • Ludiomil
Methadone • Dolophine, Methadose
Methylphenidate • Ritalin, Concerta
Midazolam • Versed
Milnacipran • Savella
Mirtazapine • Remeron
Naltrexone • Revia, Vivitrol
Nefazodone • Serzone
Nortriptyline • Aventyl, Pamelor
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Perphenazine • Trilafon
Phenelzine • Nardil
Pimavanserin • Nuplazid
Prochlorperazine • Compro
Protriptyline • Vivactil
Quetiapine • Seroquel
Risperidone • Risperdal
Selegiline • Emsam
Sertraline • Zoloft
Thioridazine • Mellaril
Thiothixene • Navane
Tranylcypromine • Parnate
Trazodone • Desyrel, Oleptro
Trifluoperazine • Stelazine
Trimipramine • Surmontil
Valproate • Depakote
Varenicline • Chantix, Wellbutrin
Vilazodone • Viibryd
Venlafaxine • Effexor
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
1. Spravato [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
2. U.S. Food and Drug Administration. FDA drug safety announcement: FDA adds boxed warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. https://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Published April 30, 2019. Accessed October 28, 2019.
3. Zulresso [package insert]. Cambridge, Mass.: Sage Therapeutics Inc.; 2019.
4. Gassman AL, Nguyen CP, Joffe HV. FDA regulation of prescription drugs. N Engl J Med. 2017;376(7):674-682.
5. Solotke MT, Dhruva SS, Downing NS, et al. New and incremental FDA black box warnings from 2008 to 2015. Expert Opin Drug Saf. 2018;17(2):117-123.
6. Murphy S, Roberts R. “Black box” 101: how the Food and Drug Administration evaluates, communicates, and manages drug benefit/risk. J Allergy Clin Immunol. 2006;117(1):34-39.
7. U.S. Food and Drug Administration. Guidance document: Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products – content and format. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/warnings-and-precautions-contraindications-and-boxed-warning-sections-labeling-human-prescription. Published October 2011. Accessed October 28, 2019.
8. Beach JE, Faich GA, Bormel FG, et al. Black box warnings in prescription drug labeling: results of a survey of 206 drugs. Food Drug Law J. 1998;53(3):403-411.
9. Matlock A, Allan N, Wills B, et al. A continuing black hole? The FDA boxed warning: an appeal to improve its clinical utility. Clinical Toxicol (Phila). 2011;49(6):443-447.
10. Elraiyah T, Gionfriddo MR, Montori VM, et al. Content, consistency, and quality of black box warnings: time for a change. Ann Intern Med. 2015;163(11):875-876.
11. Yeh JS, Sarpatwari A, Kesselheim AS. Ethical and practical considerations in removing black box warnings from drug labels. Drug Saf. 2016;39(8):709-714.
12. Boudes PF. Risk Evaluation and Mitigation Strategies (REMSs): are they improving drug safety? A critical review of REMSs requiring Elements to Assure Safe Use (ETASU). Drugs R D. 2017;17(2):245-254.
13. U.S. Food and Drug Administration. Approved risk evaluation mitigation strategies (REMS): Spravato (esketamine) REMS program. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386. Updated June 25, 2019. Accessed October 28, 2018.
14. Stevens JR, Jarrahzadeh T, Brendel RW, et al. Strategies for the prescription of psychotropic drugs with black box warnings. Psychosomatics. 2014;55(2):123-133.
15. Friedman RA. Antidepressants’ black-box warning--10 years later. N Engl J Med. 2014;371(18):1666-1668.
16. Stone MB. The FDA warning on antidepressants and suicidality--why the controversy? N Engl J Med. 2014;371(18):1668-1671.
17. Mathis MV, Muoio BM, Andreason P, et al. The US Food and Drug Administration’s perspective on the new antipsychotic pimavanserin. J Clin Psychiatry. 2017;78(6):e668-e673. doi: 10.4088/JCP.16r11119.
18. Nuplazid [package insert]. San Diego, CA: Acadia Pharmaceuticals Inc.; May 2019.
19. Ballard C, Banister C, Khan Z, et al. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in patients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
20. Maust DT, Kim HM, Chiang C, et al. Association of the Centers for Medicare & Medicaid Services’ National Partnership to Improve Dementia Care with the use of antipsychotics and other psychotropics in long-term care in the United States from 2009 to 2014. JAMA Intern Med. 2018;178(5):640-647.
21. Dorsey ER, Rabbani A, Gallagher SA, et al. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170(1):96-103.
22. U.S. Food and Drug Administration. FDA drug safety communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking. Published December 16, 2016. Accessed October 28, 2019.
1. Spravato [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
2. U.S. Food and Drug Administration. FDA drug safety announcement: FDA adds boxed warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. https://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Published April 30, 2019. Accessed October 28, 2019.
3. Zulresso [package insert]. Cambridge, Mass.: Sage Therapeutics Inc.; 2019.
4. Gassman AL, Nguyen CP, Joffe HV. FDA regulation of prescription drugs. N Engl J Med. 2017;376(7):674-682.
5. Solotke MT, Dhruva SS, Downing NS, et al. New and incremental FDA black box warnings from 2008 to 2015. Expert Opin Drug Saf. 2018;17(2):117-123.
6. Murphy S, Roberts R. “Black box” 101: how the Food and Drug Administration evaluates, communicates, and manages drug benefit/risk. J Allergy Clin Immunol. 2006;117(1):34-39.
7. U.S. Food and Drug Administration. Guidance document: Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products – content and format. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/warnings-and-precautions-contraindications-and-boxed-warning-sections-labeling-human-prescription. Published October 2011. Accessed October 28, 2019.
8. Beach JE, Faich GA, Bormel FG, et al. Black box warnings in prescription drug labeling: results of a survey of 206 drugs. Food Drug Law J. 1998;53(3):403-411.
9. Matlock A, Allan N, Wills B, et al. A continuing black hole? The FDA boxed warning: an appeal to improve its clinical utility. Clinical Toxicol (Phila). 2011;49(6):443-447.
10. Elraiyah T, Gionfriddo MR, Montori VM, et al. Content, consistency, and quality of black box warnings: time for a change. Ann Intern Med. 2015;163(11):875-876.
11. Yeh JS, Sarpatwari A, Kesselheim AS. Ethical and practical considerations in removing black box warnings from drug labels. Drug Saf. 2016;39(8):709-714.
12. Boudes PF. Risk Evaluation and Mitigation Strategies (REMSs): are they improving drug safety? A critical review of REMSs requiring Elements to Assure Safe Use (ETASU). Drugs R D. 2017;17(2):245-254.
13. U.S. Food and Drug Administration. Approved risk evaluation mitigation strategies (REMS): Spravato (esketamine) REMS program. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386. Updated June 25, 2019. Accessed October 28, 2018.
14. Stevens JR, Jarrahzadeh T, Brendel RW, et al. Strategies for the prescription of psychotropic drugs with black box warnings. Psychosomatics. 2014;55(2):123-133.
15. Friedman RA. Antidepressants’ black-box warning--10 years later. N Engl J Med. 2014;371(18):1666-1668.
16. Stone MB. The FDA warning on antidepressants and suicidality--why the controversy? N Engl J Med. 2014;371(18):1668-1671.
17. Mathis MV, Muoio BM, Andreason P, et al. The US Food and Drug Administration’s perspective on the new antipsychotic pimavanserin. J Clin Psychiatry. 2017;78(6):e668-e673. doi: 10.4088/JCP.16r11119.
18. Nuplazid [package insert]. San Diego, CA: Acadia Pharmaceuticals Inc.; May 2019.
19. Ballard C, Banister C, Khan Z, et al. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in patients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
20. Maust DT, Kim HM, Chiang C, et al. Association of the Centers for Medicare & Medicaid Services’ National Partnership to Improve Dementia Care with the use of antipsychotics and other psychotropics in long-term care in the United States from 2009 to 2014. JAMA Intern Med. 2018;178(5):640-647.
21. Dorsey ER, Rabbani A, Gallagher SA, et al. Impact of FDA black box advisory on antipsychotic medication use. Arch Intern Med. 2010;170(1):96-103.
22. U.S. Food and Drug Administration. FDA drug safety communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking. Published December 16, 2016. Accessed October 28, 2019.
Alkermes submits NDA for new schizophrenia, bipolar I treatment
Alkermes has announced that it has submitted a New Drug Application to the Food and Drug Administration for the approval of ALKS 3831 (olanzapine/samidorphan) for the treatment of schizophrenia and bipolar I disorder.
Included in the application for the investigational, novel, once-daily, oral atypical antipsychotic drug candidate is data from the ENLIGHTEN-1 study, which evaluated the antipsychotic efficacy of ALKS 3831, compared with a placebo, over a 4-week period, as well as data from ENLIGHTEN-2, which compared weight gain with ALKS 3831 and olanzapine alone over a 6-month period.
“Antipsychotic medications are an important part of the treatment paradigm for both schizophrenia and bipolar I disorder, yet there remains a persistent unmet need for new treatments,” Craig Hopkinson, MD, chief medical officer and senior vice president of medicines development and medical affairs at Alkermes, said in a press release.
As a combination of olanzapine and samidorphan, Samidorphan, an opioid receptor antagonist, is structurally related to naltrexone.
Alkermes is seeking an indication for the treatment of schizophrenia and an indication for the treatment of manic or mixed episodes associated with bipolar I disorder as monotherapy or as an adjunct to lithium or valproate, as well as for maintenance treatment of bipolar I. Dosage strength would be 10 mg of samidorphan with 5, 10, 15, or 20 mg of olanzapine.
Find the full press release on the Alkermes website.
Alkermes has announced that it has submitted a New Drug Application to the Food and Drug Administration for the approval of ALKS 3831 (olanzapine/samidorphan) for the treatment of schizophrenia and bipolar I disorder.
Included in the application for the investigational, novel, once-daily, oral atypical antipsychotic drug candidate is data from the ENLIGHTEN-1 study, which evaluated the antipsychotic efficacy of ALKS 3831, compared with a placebo, over a 4-week period, as well as data from ENLIGHTEN-2, which compared weight gain with ALKS 3831 and olanzapine alone over a 6-month period.
“Antipsychotic medications are an important part of the treatment paradigm for both schizophrenia and bipolar I disorder, yet there remains a persistent unmet need for new treatments,” Craig Hopkinson, MD, chief medical officer and senior vice president of medicines development and medical affairs at Alkermes, said in a press release.
As a combination of olanzapine and samidorphan, Samidorphan, an opioid receptor antagonist, is structurally related to naltrexone.
Alkermes is seeking an indication for the treatment of schizophrenia and an indication for the treatment of manic or mixed episodes associated with bipolar I disorder as monotherapy or as an adjunct to lithium or valproate, as well as for maintenance treatment of bipolar I. Dosage strength would be 10 mg of samidorphan with 5, 10, 15, or 20 mg of olanzapine.
Find the full press release on the Alkermes website.
Alkermes has announced that it has submitted a New Drug Application to the Food and Drug Administration for the approval of ALKS 3831 (olanzapine/samidorphan) for the treatment of schizophrenia and bipolar I disorder.
Included in the application for the investigational, novel, once-daily, oral atypical antipsychotic drug candidate is data from the ENLIGHTEN-1 study, which evaluated the antipsychotic efficacy of ALKS 3831, compared with a placebo, over a 4-week period, as well as data from ENLIGHTEN-2, which compared weight gain with ALKS 3831 and olanzapine alone over a 6-month period.
“Antipsychotic medications are an important part of the treatment paradigm for both schizophrenia and bipolar I disorder, yet there remains a persistent unmet need for new treatments,” Craig Hopkinson, MD, chief medical officer and senior vice president of medicines development and medical affairs at Alkermes, said in a press release.
As a combination of olanzapine and samidorphan, Samidorphan, an opioid receptor antagonist, is structurally related to naltrexone.
Alkermes is seeking an indication for the treatment of schizophrenia and an indication for the treatment of manic or mixed episodes associated with bipolar I disorder as monotherapy or as an adjunct to lithium or valproate, as well as for maintenance treatment of bipolar I. Dosage strength would be 10 mg of samidorphan with 5, 10, 15, or 20 mg of olanzapine.
Find the full press release on the Alkermes website.
Will paying patients to take oral antipsychotics boost adherence?
COPENHAGEN – Paying patients with schizophrenia a modest sum to take their long-acting injectable antipsychotic agents is a proven, evidence-based strategy for achieving improved adherence. Now it’s time to find out whether the same approach will work in patients on oral antipsychotics, Daniel Guinart, MD, said at the annual congress of the European College of Neuropsychopharmacology.
He presented a systematic literature review that turned up four studies, including two randomized clinical trials, of economic incentives aimed at improving adherence to long-acting injectable antipsychotics. The studies, all positive, showed that small financial incentives improved adherence by 12%-15%. However, once the intervention ended, adherence drifted back downward, so the financial reinforcement needs to be ongoing.
This approach has not previously been tested in outpatients with severe mental illness who are on oral antipsychotics because of the intrinsic limitations of reliance upon patient self-reported adherence. But Dr. Guinart and his coinvestigator, John M. Kane, MD, professor and chair of the department of psychiatry at Hofstra University in Hempstead, N.Y., have launched a 25-patient proof-of-concept study using technology to measure outpatient adherence. Provided that the pilot study shows this approach is feasible, the plan is to conduct a larger, longer-term, randomized controlled trial, said Dr. Guinart, also at Hofstra University.
If the modest financial expenditures involved in payment for pill taking do indeed prove to improve outpatient adherence to oral antipsychotics, it could reap major dividends in terms of fewer relapses, emergency department visits, hospitalizations, suicide attempts, and legal problems for patients, he noted.
The financial incentive to take oral antipsychotics needs to be big enough to promote behavioral change, but not so large that it poses ethical issues or encourages patients to game the system by lowering their adherence in order to gain entrance into the program. Fortunately, prior studies of successful behavioral incentives to take antihypertensive medications and other oral nonpsychotrophic drugs provide guidance on this score.
“One to two dollars per day is considered a reasonable incentive because it generates behavioral change, yet $30 per month doesn’t really serve as a financial aid,” the psychiatrist explained in an interview.
In the pilot study, adherence to oral antipsychotics is being assessed by having patients snap a cell phone photo of their daily medications being held in hand. Proprietary software analyzes whether those are the correct pills as prescribed. If so, the patient gets rewarded.
“It’s possible that after taking the photo some patients may throw the pills away. In adherence studies, we’re not the police, so at some point we have to trust that the patient is taking the medication,” he said.
Dr. Guinart reported having no financial conflicts of interest regarding his presentation.
COPENHAGEN – Paying patients with schizophrenia a modest sum to take their long-acting injectable antipsychotic agents is a proven, evidence-based strategy for achieving improved adherence. Now it’s time to find out whether the same approach will work in patients on oral antipsychotics, Daniel Guinart, MD, said at the annual congress of the European College of Neuropsychopharmacology.
He presented a systematic literature review that turned up four studies, including two randomized clinical trials, of economic incentives aimed at improving adherence to long-acting injectable antipsychotics. The studies, all positive, showed that small financial incentives improved adherence by 12%-15%. However, once the intervention ended, adherence drifted back downward, so the financial reinforcement needs to be ongoing.
This approach has not previously been tested in outpatients with severe mental illness who are on oral antipsychotics because of the intrinsic limitations of reliance upon patient self-reported adherence. But Dr. Guinart and his coinvestigator, John M. Kane, MD, professor and chair of the department of psychiatry at Hofstra University in Hempstead, N.Y., have launched a 25-patient proof-of-concept study using technology to measure outpatient adherence. Provided that the pilot study shows this approach is feasible, the plan is to conduct a larger, longer-term, randomized controlled trial, said Dr. Guinart, also at Hofstra University.
If the modest financial expenditures involved in payment for pill taking do indeed prove to improve outpatient adherence to oral antipsychotics, it could reap major dividends in terms of fewer relapses, emergency department visits, hospitalizations, suicide attempts, and legal problems for patients, he noted.
The financial incentive to take oral antipsychotics needs to be big enough to promote behavioral change, but not so large that it poses ethical issues or encourages patients to game the system by lowering their adherence in order to gain entrance into the program. Fortunately, prior studies of successful behavioral incentives to take antihypertensive medications and other oral nonpsychotrophic drugs provide guidance on this score.
“One to two dollars per day is considered a reasonable incentive because it generates behavioral change, yet $30 per month doesn’t really serve as a financial aid,” the psychiatrist explained in an interview.
In the pilot study, adherence to oral antipsychotics is being assessed by having patients snap a cell phone photo of their daily medications being held in hand. Proprietary software analyzes whether those are the correct pills as prescribed. If so, the patient gets rewarded.
“It’s possible that after taking the photo some patients may throw the pills away. In adherence studies, we’re not the police, so at some point we have to trust that the patient is taking the medication,” he said.
Dr. Guinart reported having no financial conflicts of interest regarding his presentation.
COPENHAGEN – Paying patients with schizophrenia a modest sum to take their long-acting injectable antipsychotic agents is a proven, evidence-based strategy for achieving improved adherence. Now it’s time to find out whether the same approach will work in patients on oral antipsychotics, Daniel Guinart, MD, said at the annual congress of the European College of Neuropsychopharmacology.
He presented a systematic literature review that turned up four studies, including two randomized clinical trials, of economic incentives aimed at improving adherence to long-acting injectable antipsychotics. The studies, all positive, showed that small financial incentives improved adherence by 12%-15%. However, once the intervention ended, adherence drifted back downward, so the financial reinforcement needs to be ongoing.
This approach has not previously been tested in outpatients with severe mental illness who are on oral antipsychotics because of the intrinsic limitations of reliance upon patient self-reported adherence. But Dr. Guinart and his coinvestigator, John M. Kane, MD, professor and chair of the department of psychiatry at Hofstra University in Hempstead, N.Y., have launched a 25-patient proof-of-concept study using technology to measure outpatient adherence. Provided that the pilot study shows this approach is feasible, the plan is to conduct a larger, longer-term, randomized controlled trial, said Dr. Guinart, also at Hofstra University.
If the modest financial expenditures involved in payment for pill taking do indeed prove to improve outpatient adherence to oral antipsychotics, it could reap major dividends in terms of fewer relapses, emergency department visits, hospitalizations, suicide attempts, and legal problems for patients, he noted.
The financial incentive to take oral antipsychotics needs to be big enough to promote behavioral change, but not so large that it poses ethical issues or encourages patients to game the system by lowering their adherence in order to gain entrance into the program. Fortunately, prior studies of successful behavioral incentives to take antihypertensive medications and other oral nonpsychotrophic drugs provide guidance on this score.
“One to two dollars per day is considered a reasonable incentive because it generates behavioral change, yet $30 per month doesn’t really serve as a financial aid,” the psychiatrist explained in an interview.
In the pilot study, adherence to oral antipsychotics is being assessed by having patients snap a cell phone photo of their daily medications being held in hand. Proprietary software analyzes whether those are the correct pills as prescribed. If so, the patient gets rewarded.
“It’s possible that after taking the photo some patients may throw the pills away. In adherence studies, we’re not the police, so at some point we have to trust that the patient is taking the medication,” he said.
Dr. Guinart reported having no financial conflicts of interest regarding his presentation.
REPORTING FROM ECNP 2019
Resting-state cerebral blood flow tied to apathy in schizophrenia
Regional resting-state cerebral blood flow (rCBF) in the ventral and dorsal striatum is associated with apathy in patients with schizophrenia – but not with diminished expression, results of a small study suggest.
“This distinction is of high relevance when investigating striatal rCBF,” wrote Karoline Schneider, MD, and colleagues. “The paucity of reported associations beween striatal rCBF and negative symptoms could result from the fact that until now apathy and diminished expression have not been addressed separately.” The study was published in the Journal of Psychiatry & Neuroscience.
Dr. Schneider and colleagues used arterial spin labeling MRI to measure rCBF in 29 patients with schizophrenia from outpatient and inpatient units affiliated with the University of Zürich and in 20 controls without schizophrenia. Negative symptoms were assessed via the Brief Negative Symptom Scale and were divided into those of apathy or those of diminished expression.
, and between severity and higher rCBF in the left and right dorsal striatum (r = 0.48; P = .008). However, the correlations were nonsignificant for symptoms of diminished expression, reported Dr. Schneider, of the university’s department of psychiatry, psychotherapy, and psychosomatics.
“The association between increased striatal rCBF and the negative symptom dimension of apathy, but not diminished expression, provides further evidence for the assumption of different underlying neural bases,” Dr. Schneider and colleagues concluded. “These dimensions should be considered separately in future research on negative symptoms.”
Limitations of the study include its sample size. Another limitation is that all patients with schizophrenia in the study were taking second-generation antipsychotics, which in some research appears to influence striatal rCBF.
Grants from the Swiss National Science Foundation funded the study. Dr. Schneider reported no conflicts of interest. Two authors disclosed receiving grant support or honoraria from pharmaceutical companies but said those activities were unrelated to this study.
SOURCE: Schneider K et al. J Psychiatry Neurosci. 2019;44(2):102-10.
Regional resting-state cerebral blood flow (rCBF) in the ventral and dorsal striatum is associated with apathy in patients with schizophrenia – but not with diminished expression, results of a small study suggest.
“This distinction is of high relevance when investigating striatal rCBF,” wrote Karoline Schneider, MD, and colleagues. “The paucity of reported associations beween striatal rCBF and negative symptoms could result from the fact that until now apathy and diminished expression have not been addressed separately.” The study was published in the Journal of Psychiatry & Neuroscience.
Dr. Schneider and colleagues used arterial spin labeling MRI to measure rCBF in 29 patients with schizophrenia from outpatient and inpatient units affiliated with the University of Zürich and in 20 controls without schizophrenia. Negative symptoms were assessed via the Brief Negative Symptom Scale and were divided into those of apathy or those of diminished expression.
, and between severity and higher rCBF in the left and right dorsal striatum (r = 0.48; P = .008). However, the correlations were nonsignificant for symptoms of diminished expression, reported Dr. Schneider, of the university’s department of psychiatry, psychotherapy, and psychosomatics.
“The association between increased striatal rCBF and the negative symptom dimension of apathy, but not diminished expression, provides further evidence for the assumption of different underlying neural bases,” Dr. Schneider and colleagues concluded. “These dimensions should be considered separately in future research on negative symptoms.”
Limitations of the study include its sample size. Another limitation is that all patients with schizophrenia in the study were taking second-generation antipsychotics, which in some research appears to influence striatal rCBF.
Grants from the Swiss National Science Foundation funded the study. Dr. Schneider reported no conflicts of interest. Two authors disclosed receiving grant support or honoraria from pharmaceutical companies but said those activities were unrelated to this study.
SOURCE: Schneider K et al. J Psychiatry Neurosci. 2019;44(2):102-10.
Regional resting-state cerebral blood flow (rCBF) in the ventral and dorsal striatum is associated with apathy in patients with schizophrenia – but not with diminished expression, results of a small study suggest.
“This distinction is of high relevance when investigating striatal rCBF,” wrote Karoline Schneider, MD, and colleagues. “The paucity of reported associations beween striatal rCBF and negative symptoms could result from the fact that until now apathy and diminished expression have not been addressed separately.” The study was published in the Journal of Psychiatry & Neuroscience.
Dr. Schneider and colleagues used arterial spin labeling MRI to measure rCBF in 29 patients with schizophrenia from outpatient and inpatient units affiliated with the University of Zürich and in 20 controls without schizophrenia. Negative symptoms were assessed via the Brief Negative Symptom Scale and were divided into those of apathy or those of diminished expression.
, and between severity and higher rCBF in the left and right dorsal striatum (r = 0.48; P = .008). However, the correlations were nonsignificant for symptoms of diminished expression, reported Dr. Schneider, of the university’s department of psychiatry, psychotherapy, and psychosomatics.
“The association between increased striatal rCBF and the negative symptom dimension of apathy, but not diminished expression, provides further evidence for the assumption of different underlying neural bases,” Dr. Schneider and colleagues concluded. “These dimensions should be considered separately in future research on negative symptoms.”
Limitations of the study include its sample size. Another limitation is that all patients with schizophrenia in the study were taking second-generation antipsychotics, which in some research appears to influence striatal rCBF.
Grants from the Swiss National Science Foundation funded the study. Dr. Schneider reported no conflicts of interest. Two authors disclosed receiving grant support or honoraria from pharmaceutical companies but said those activities were unrelated to this study.
SOURCE: Schneider K et al. J Psychiatry Neurosci. 2019;44(2):102-10.
FROM THE JOURNAL OF PSYCHIATRY & NEUROSCIENCE
COGS update shows viability of endophenotypes in schizophrenia research
Lead author Tiffany A. Greenwood, PhD, noted that clinically diverse schizophrenia patients often are grouped together to get large sample sizes for genome-wide association studies, which can miss specific features of the heterogeneous disorder. Instead, in phase 2 of the Consortium on the Genetics of Schizophrenia (COGS) study, Dr. Greenwood and associates sought to connect features clustered into endophenotypes with certain genetic regions of interest.
“As stable biomarkers of the underlying brain dysfunctions, endophenotypes hold promise for parsing clinical heterogeneity of schizophrenia and refining the genetic signal,” they wrote. The study was published in JAMA Psychiatry.
Using this approach among 1,533 participants, they found seven regions exceeding the conventional genome-wide association significance of P less than 5 x 10–8, including regions associated with the endophenotypes of face memory (chromosome 3p21; effect size, –0.72; P = 4.2 x 10–8), antisaccade task (chromosome 9q31; effect size, –0.24; P = 3.5 x 10–8), and abstraction and mental flexibility (chromosome 10q23; effect size, –0.56; P = 1.5 x 10–8).
Those endophenotypes and genes intersect theoretical molecular and biological processes that have been identified in other research and could explain underlying mechanisms of schizophrenia. For example, research has suggested that NRG3, which is near the region associated with abstraction and mental flexibility, and affects certain cellular signaling pathways, could be a locus of susceptibility; in particular, some variants of NRG3 have been associated with cognitive and psychotic symptom severity in previous research.
“Although shared genetic substrates appear likely, this is not a study of schizophrenia but rather a study of neurophysiological and neurocognitive deficits that occur in the general population but are more pronounced in the context of schizophrenia and have implications for treatment,” wrote Dr. Greenwood, of the University of California, San Diego, and associates.
One limitation of the study is that, as investigators have demonstrated elsewhere, P values can prove highly variable over the course of replication studies, so the significance thresholds shown in this study still could run the risk of hiding false positives or negatives.
“As many of the 11 endophenotypes have been endorsed as targets for the development of novel treatments for schizophrenia, a better understanding of the corresponding cellular and molecular processes may pave the way for precision-based medicine in schizophrenia and perhaps other psychiatric illnesses with a shared genetic liability,” Dr. Greenwood and associates concluded.
SOURCE: Greenwood TA et al. JAMA Psychiatry. 2019 Oct 9. doi: 10.1001/jamapsychiatry.2019.2850.
Lead author Tiffany A. Greenwood, PhD, noted that clinically diverse schizophrenia patients often are grouped together to get large sample sizes for genome-wide association studies, which can miss specific features of the heterogeneous disorder. Instead, in phase 2 of the Consortium on the Genetics of Schizophrenia (COGS) study, Dr. Greenwood and associates sought to connect features clustered into endophenotypes with certain genetic regions of interest.
“As stable biomarkers of the underlying brain dysfunctions, endophenotypes hold promise for parsing clinical heterogeneity of schizophrenia and refining the genetic signal,” they wrote. The study was published in JAMA Psychiatry.
Using this approach among 1,533 participants, they found seven regions exceeding the conventional genome-wide association significance of P less than 5 x 10–8, including regions associated with the endophenotypes of face memory (chromosome 3p21; effect size, –0.72; P = 4.2 x 10–8), antisaccade task (chromosome 9q31; effect size, –0.24; P = 3.5 x 10–8), and abstraction and mental flexibility (chromosome 10q23; effect size, –0.56; P = 1.5 x 10–8).
Those endophenotypes and genes intersect theoretical molecular and biological processes that have been identified in other research and could explain underlying mechanisms of schizophrenia. For example, research has suggested that NRG3, which is near the region associated with abstraction and mental flexibility, and affects certain cellular signaling pathways, could be a locus of susceptibility; in particular, some variants of NRG3 have been associated with cognitive and psychotic symptom severity in previous research.
“Although shared genetic substrates appear likely, this is not a study of schizophrenia but rather a study of neurophysiological and neurocognitive deficits that occur in the general population but are more pronounced in the context of schizophrenia and have implications for treatment,” wrote Dr. Greenwood, of the University of California, San Diego, and associates.
One limitation of the study is that, as investigators have demonstrated elsewhere, P values can prove highly variable over the course of replication studies, so the significance thresholds shown in this study still could run the risk of hiding false positives or negatives.
“As many of the 11 endophenotypes have been endorsed as targets for the development of novel treatments for schizophrenia, a better understanding of the corresponding cellular and molecular processes may pave the way for precision-based medicine in schizophrenia and perhaps other psychiatric illnesses with a shared genetic liability,” Dr. Greenwood and associates concluded.
SOURCE: Greenwood TA et al. JAMA Psychiatry. 2019 Oct 9. doi: 10.1001/jamapsychiatry.2019.2850.
Lead author Tiffany A. Greenwood, PhD, noted that clinically diverse schizophrenia patients often are grouped together to get large sample sizes for genome-wide association studies, which can miss specific features of the heterogeneous disorder. Instead, in phase 2 of the Consortium on the Genetics of Schizophrenia (COGS) study, Dr. Greenwood and associates sought to connect features clustered into endophenotypes with certain genetic regions of interest.
“As stable biomarkers of the underlying brain dysfunctions, endophenotypes hold promise for parsing clinical heterogeneity of schizophrenia and refining the genetic signal,” they wrote. The study was published in JAMA Psychiatry.
Using this approach among 1,533 participants, they found seven regions exceeding the conventional genome-wide association significance of P less than 5 x 10–8, including regions associated with the endophenotypes of face memory (chromosome 3p21; effect size, –0.72; P = 4.2 x 10–8), antisaccade task (chromosome 9q31; effect size, –0.24; P = 3.5 x 10–8), and abstraction and mental flexibility (chromosome 10q23; effect size, –0.56; P = 1.5 x 10–8).
Those endophenotypes and genes intersect theoretical molecular and biological processes that have been identified in other research and could explain underlying mechanisms of schizophrenia. For example, research has suggested that NRG3, which is near the region associated with abstraction and mental flexibility, and affects certain cellular signaling pathways, could be a locus of susceptibility; in particular, some variants of NRG3 have been associated with cognitive and psychotic symptom severity in previous research.
“Although shared genetic substrates appear likely, this is not a study of schizophrenia but rather a study of neurophysiological and neurocognitive deficits that occur in the general population but are more pronounced in the context of schizophrenia and have implications for treatment,” wrote Dr. Greenwood, of the University of California, San Diego, and associates.
One limitation of the study is that, as investigators have demonstrated elsewhere, P values can prove highly variable over the course of replication studies, so the significance thresholds shown in this study still could run the risk of hiding false positives or negatives.
“As many of the 11 endophenotypes have been endorsed as targets for the development of novel treatments for schizophrenia, a better understanding of the corresponding cellular and molecular processes may pave the way for precision-based medicine in schizophrenia and perhaps other psychiatric illnesses with a shared genetic liability,” Dr. Greenwood and associates concluded.
SOURCE: Greenwood TA et al. JAMA Psychiatry. 2019 Oct 9. doi: 10.1001/jamapsychiatry.2019.2850.
FROM JAMA PSYCHIATRY
Should psychiatrists prescribe nonpsychotropic medications?
In our experience, most psychiatrists are uncomfortable with prescribing a medication when they feel that doing so would be outside their scope of practice. But there are many situations when prescribing a nonpsychotropic medication would be the correct choice. In this article, we discuss the scope of psychiatric practice, and present 4 case studies that illustrate situations in which psychiatrists should feel comfortable prescribing nonpsychotropic medications.
Defining the scope of practice
What is the scope of a psychiatrist’s practice? Scope of practice usually describes activities that a health care practitioner is allowed to undertake as defined by the terms of his/her license. A license to practice medicine does not include any stipulation restricting practice to a specific medical specialty. However, a local entity may delineate scope of practice within its organization. For instance, local practice standards held by the Detroit Wayne Mental Health Authority (DWMHA) state “Psychiatrists…shall not exceed their scope of practice as per DWMHA credentialing and privileging. For example, a Psychiatrist…who [has] not been appropriately privileged to deliver services to children shall not treat children, excepting crisis situations.”1
Like physicians in other specialties, psychiatrists are not limited to prescribing only a subset of medications commonly associated with their specialty. But for many psychiatrists, prescribing nonpsychotropic medications is complicated by individual and local factors. On one hand, some psychiatrists do not feel it is their role to prescribe nonpsychotropic medications,2 or even some psychotropic medications that may be more complex to prescribe, such as lithium, clozapine, or monoamine oxidase inhibitors.3-5 However, many feel comfortable prescribing complex combinations of psychotropic medications, or prescribing in a way that does not necessarily make sense (eg, prescribing benztropine as prophylaxis for dystonia when starting an antipsychotic).
Reviewing an average day at one urban psychiatric clinic, these questions seem to come up in half of the patient population, especially in patients with chronic mental illness, multiple medical comorbidities, and limited access to health care. When a young patient walks in without an appointment with an acute dystonic reaction secondary to the initiation of antipsychotics a couple of days ago, there is no hesitation to swiftly and appropriately prescribe an IM anticholinergic medication. But why are psychiatrists often hesitant to prescribe nonpsychotropic medications to treat other adverse effects of medications? Lack of knowledge? Lack of training?
Psychiatrists who practice in hospital systems often have immediate access to consultants, and this availability may encourage them to defer to the consultant for treatment of certain adverse effects. We have seen psychiatrists consult Neurology regarding the prescription of donepezil for mild neurocognitive disorder due to Alzheimer’s disease, or Endocrinology regarding prescription of levothyroxine for lithium-induced hypothyroidism.
However, there are numerous scenarios in which psychiatrists should feel comfortable prescribing nonpsychotropic medications or managing medication adverse effects, regardless of whether they consider it to be within or outside their scope of practice. The following case examples illustrate several such situations.
CASE 1
Ms. W, age 30, has been diagnosed with schizophrenia. She requests a refill of quetiapine, 800 mg/d. This medication has been clearly beneficial in alleviating her psychotic symptoms. However, since her last visit 3 months ago, her face appears more round, and she has gained 9 kg. Further evaluation indicates that she has developed metabolic syndrome and pre-diabetes.
Continue to: Metabolic adverse effects
Metabolic adverse effects, such as metabolic syndrome, diabetic ketoacidosis, and cardiovascular disease, are well-known risks of prescribing second-generation antipsychotics.6 In such situations, psychiatrists often advise patients to modify their diet, increase physical activity, and follow up with their primary care physician to determine if other medications are needed. However, getting a patient with a serious mental illness to exercise and modify her/his diet is difficult, and many of these patients do not have a primary care physician.
For patients such as Ms. W, a psychiatrist should consider prescribing metformin. Wu et al7 found that in addition to lifestyle modifications, metformin had the greatest effect on antipsychotic-induced weight gain. In this study, metformin alone had more impact on reversing weight gain and increasing insulin sensitivity than lifestyle modifications alone.7 This is crucial because these patients are especially vulnerable to cardiac disease.8 Metformin is well tolerated and has a low risk of causing hypoglycemia. Concerns regarding lactic acidosis have abated to the extent that the estimated glomerular filtration rate (eGFR) limits for using metformin have been lowered significantly. After reviewing the contraindications, the only knowledge needed to prescribe metformin is the patient’s kidney function and a brief understanding of the titration needed to minimize gastrointestinal adverse effects.9 Thus, prescribing metformin would be a fairly logical and easy first step for managing metabolic syndrome, especially in a patient whose motivation for increasing physical activity and modifying his/her diet is doubtful.
CASE 2
Mr. B, age 45, has major depressive disorder that has been well-controlled on paroxetine, 40 mg/d, for the past 2 years. He has no history of physical illness. On his most recent visit, he appears uncomfortable and nervous. After a long discussion, he discloses that his sex life isn’t what it used to be since starting paroxetine. He is bothered by erectile problems and asks whether he can “get some Viagra.”
Sexual adverse effects, such as erectile dysfunction, are frequently associated with the use of selective serotonin reuptake inhibitors.10 Although managing these adverse effects requires careful evaluation, in most cases, psychiatrists should be able to treat them.10 The logical choice in this case would be to prescribe one of the 4 FDA-approved phosphodiesterase-5 inhibitors (sildenafil [Viagra], tadalafil [Cialis], vardenafil [Levitra], and avanafil [Stendra]. However, Balon et al11 found that few psychiatrists prescribe phosphodiesterase-5 inhibitors, although they believed that they should be prescribing to treat their patients’ sexual dysfunction. Managing these adverse effects is important not only for the patient’s quality of life and relationship with his/her partner, but also for the therapeutic alliance. In a systematic review of 23 trials, Taylor et al12 examined >1,800 patients who were prescribed a medication to address sexual dysfunction secondary to antidepressants. They found that for men, adding a phosphodiesterase-5 inhibitor was appropriate and effective, and for women, adding bupropion at high doses should be considered.12 Like many other adverse effects, sexual adverse effects surely play a role in medication compliance. Dording et al13 found that the addition of sildenafil, 50 to 100 mg as needed, resulted in increased treatment satisfaction and overall contentment in 102 patients who complained of sexual dysfunction in the follow-up phase of the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) antidepressant trials. In most cases, with proper psychoeducation, prescription of
CASE 3
Ms. G, age 22, was recently discharged from an inpatient psychiatric unit after an episode of mania. She was prescribed carbamazepine, 600 mg/d, and ziprasidone, 40 mg twice a day, and appears to be doing well on this regimen. When asked about what led to her admission, she recalls having an elevated mood, increased energy, hypersexuality, impulsivity, and poor judgment. She reveals that she had several sexual partners during her manic episode, and worries that if such behavior occurs again, she may get pregnant. Yet Ms. G was not prescribed birth control upon discharge.
Continue to: Contraception
Contraception. We believe that psychiatrists have an obligation to protect patients from consequences of mental illness. Much the same way that psychiatrists hope to prevent suicide in a patient who has depression, patients should be protected from risks encountered in the manic phase of bipolar disorder. Another reason to prescribe contraceptives in such patients is the teratogenic effects of mood stabilizers. Pagano et al14 reviewed 6 studies that examined common forms of hormonal birth control to determine their safety in patients with depression or bipolar disorder. They found that overall, use of hormonal contraception was not associated with a worse clinical course of disease.
Many available forms of birth control are available. When prescribing in an outpatient setting, a daily oral medication or a monthly depot injection are convenient options.
CASE 4
Mr. P, age 65, has bipolar I disorder and is stable on risperidone long-acting injection, 37.7 mg bimonthly, and lithium, 1,200 mg/d. He reports that he is doing well but has noticed a recent decrease in energy and weight gain without any change in mood. Laboratory testing conducted prior to this visit revealed a thyroid-stimulating hormone (TSH) level of 4 mU/L (normal range: 0.4 to 4.0 mU/L). Six months ago, Mr. P’s TSH level was 2.8 mU/L. The resident supervisor suggests discussing the case with an endocrinologist.
Thyroid function. The impact of lithium on the thyroid gland is well established; however, psychiatrists’ response to such changes are not.15 Gitlin16 reviewed the many adverse effects of lithium and presented various management strategies to address findings such as Mr. P’s. Two important points are that lithium should not be discontinued in light of hypothyroidism, and synthetic thyroxine (levothyroxine) can be initiated and titrated to return TSH levels to a normal range.16 Levothyroxine can be started at low doses (eg, 25 to 50 mcg/d) and increased every 6 weeks until a normal TSH level is achieved.17 Managing lithium-induced clinical or subclinical hypothyroidism can prevent further pathology and possible relapse to depression.
Incorporating integrated care
In all these cases, the prescription of a medication with which some psychiatrists are not comfortable prescribing would have been the logical, easiest, and preferable choice. Of course, when initiating any medication, boxed warnings, contraindications, and drug–drug interactions should be reviewed. Initial dosages and titration schedules can be found in every medication’s FDA-approved prescribing information document (package insert), as well as in numerous reference books and articles.
Continue to: We acknowledge...
We acknowledge that prescribing a nonpsychotropic medication is not always a psychiatrist’s best choice, and that in patients with multiple medical comorbidities and drug–drug interactions that are not clearly defined, referring to or consulting a specialist is appropriate. We in no way support reckless prescribing, but instead present an opportunity to expand the perception of what should be considered within a psychiatrist’s scope of practice, and call for further education of psychiatrists so that they are more comfortable managing these adverse effects and/or prescribing at least some nonpsychotropic medications.
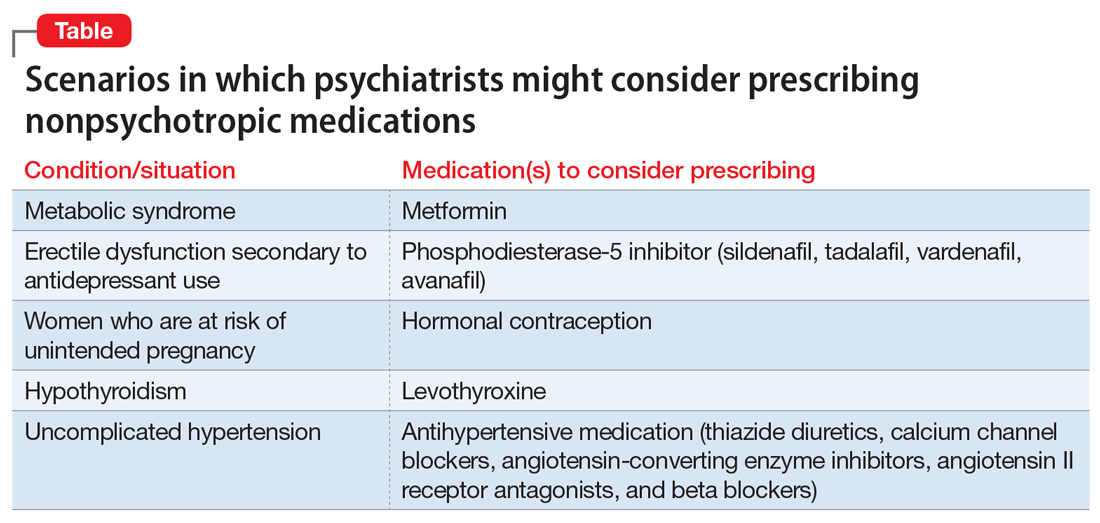
We exhort integrated medical care during this time of a physician shortage; however, we do not practice this way. Interestingly, physicians in primary care, such as those in family medicine or obstetrics and gynecology, frequently attempt to treat patients with psychiatric conditions in an attempt to provide integrated care. Numerous articles have discussed these efforts.18-20 However, this type of integrated care seems less frequent in psychiatry, even though the practice of modern psychiatry in the United States shows substantial overlap with the practice of physicians in primary care specialties.21 There are few articles or practical guidelines for psychiatrists who wish to treat patients’ physical illnesses, particularly patients with severe mental illness (see Related Resources, page 56). If we practice in an integrated manner to treat one of the simple conditions we described above, we can eliminate the need for a patient to visit a second physician, pay another co-pay, pay another bus fare, and take another day off work. This can be particularly helpful for patients who at times have to decide between paying for groceries or for medications. Having one clinician manage a patient’s medications also can decrease the risk of polypharmacy.
In addition to the case scenarios described in this article, there are more clinical situations and nonpsychotropic medications that psychiatrists could manage. Considering them outside the scope of psychiatric practice and being uncomfortable or ambivalent about them is not an excuse. We hope that psychiatrists can increase their expertise in this area, and can start to practice as the primary care physicians they claim they are, and should be.
Bottom Line
Many psychiatrists are uncomfortable prescribing nonpsychotropic medications, but there are numerous clinical scenarios in which the practice would make sense. This could include cases of metabolic syndrome, sexual dysfunction secondary to antidepressant use, or other adverse effects of commonly prescribed psychotropic medications.
Related Resources
- McCarron RM, Xiong GL, Keenan CR, et al. Preventive medical care in psychiatry. A practical guide for clinicians. Arlington, VA: American Psychiatric Association Publishing; 2015.
- McCarron RM, Xiong GL, Keenan CR, et al. Study guide to preventive medical care in psychiatry. Arlington, VA: American Psychiatric Association Publishing; 2017.
- Goldberg JF, Ernst CL. Managing the side effects of psychotropic medications. Washington, DC: American Psychiatric Association Publishing; 2019.
Drug Brand Names
Avanafil • Stendra
Benztropine • Cogentin
Bupropion • Wellbutrin, Zyban
Carbamazepine • Carbatrol, Tegretol
Clozapine • Clozaril
Donepezil • Aricept
Levothyroxine • Levoxyl, Synthroid
Lithium • Eskalith, Lithobid
Metformin • Fortamet, Glucophage
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone long-acting injection • Risperdal Consta
Sildenafil • Viagra
Tadalafil • Cialis
Vardenafil • Levitra
Ziprasidone • Geodon
1. Detroit Wayne Integrated Health Network. DWMHA psychiatric practice standards. http://dwihn.org/files/2015/6451/9628/Psychiatric_Practice_Standards.pdf. Revised June 2018. Accessed October 8, 2019.
2. Seaman JJ, Cornfield RM, Cummings DM, et al. Exploring psychiatric prescribing practices: the relationship between the role of the provider and the appropriateness of prescribing. Gen Hosp Psychiatry. 1987;9(3):220-224.
3. Zivanovic O. Lithium: a classic drug—frequently discussed, but, sadly, seldom prescribed! Aust N Z J Psychiatry. 2017;51(9):886-896.
4. Stroup TS, Gerhard T, Crystal S, et al. Geographic and clinical variation in clozapine use in the United States. Psychiatric Services. 2014;65(2):186-192.
5. Balon R, Mufti R, Arfken C. A survey of prescribing practices for monoamine oxidase inhibitors. Psychiatric Services. 1999;50(7):945-947.
6. Rummel-Kluge C, Komossa K, Schwarz S, et al. Head-to-head comparisons of metabolic side effects of second generation antipsychotics in the treatment of schizophrenia: a systematic review and meta-analysis. Schizophr Res. 2010;123(2-3):225-233.
7. Wu RR, Zhao JP, Jin H, et al. Lifestyle intervention and metformin for treatment of antipsychotic-induced weight gain: a randomized controlled trial. JAMA. 2008;299(2):185-193.
8. De Hert M, Correll CU, Bobes J, et al. Physical illness in patients with severe mental disorders. I. Prevalence, impact of medications and disparities in health care. World Psychiatry. 2011;10(1):52-77.
9. Kirpichnikov D, McFarlane SI, Sowers JR. Metformin: an update. Ann Internal Med. 2002;137(1):25-33.
10. Balon R. SSRI-associated sexual dysfunction. Am J Psychiatry. 2006;163(9):1504-1509.
11. Balon R, Morreale MK, Segraves RT. Prescribing of phosphodiesterase-5 inhibitors among psychiatrists. J Sex Marital Ther. 2014;40(3):165-169.
12. Taylor MJ, Rudkin L, Bullemor-Day P, et al. Strategies for managing sexual dysfunction induced by antidepressant medication. Cochrane Database Syst Rev. 2013;(5):CD003382.
13. Dording CM, LaRocca RA, Hails KA, et al. The effect of sildenafil on quality of life. Ann Clin Psychiatry. 2013;25(1):3-10.
14. Pagano HP, Zapata LB, Berry-Bibee EN, et al. Safety of hormonal contraception and intrauterine devices among women with depressive and bipolar disorders: a systematic review. Contraception. 2016;94(6):641-649.
15. Kibirige D, Luzinda K, Ssekitoleko R. Spectrum of lithium induced thyroid abnormalities: a current perspective. Thyroid Res. 2013;6(1):3.
16. Gitlin M. Lithium side effects and toxicity: prevalence and management strategies. Int J Bipolar Disord. 2016;4(1):27.
17. Devdhar M, Ousman YH, Burman KD. Hypothyroidism. Endocrinol Metab Clin North Am. 2007;36(3):595-615.
18. Hackley B, Sharma C, Kedzior A, et al. Managing mental health conditions in primary care settings. J Midwifery Women’s Health. 2010;55(1):9-19.
19. Fitelson E, McGibbon C. Evaluation and management of behavioral health disorders in women: an overview of major depression, bipolar disorder, anxiety disorders, and sleep in the primary care setting. Obstet Gynecol Clin North Am. 2016;43(2):231-246.
20. Colorafi K, Vanselow J, Nelson T. Treating anxiety and depression in primary care: reducing barriers to access. Fam Pract Manag. 2017;24(4):11-16.
21. McCall WV. Defining the unique scope of psychiatric practice in 2015. J ECT. 2015;31(4):203-204.
In our experience, most psychiatrists are uncomfortable with prescribing a medication when they feel that doing so would be outside their scope of practice. But there are many situations when prescribing a nonpsychotropic medication would be the correct choice. In this article, we discuss the scope of psychiatric practice, and present 4 case studies that illustrate situations in which psychiatrists should feel comfortable prescribing nonpsychotropic medications.
Defining the scope of practice
What is the scope of a psychiatrist’s practice? Scope of practice usually describes activities that a health care practitioner is allowed to undertake as defined by the terms of his/her license. A license to practice medicine does not include any stipulation restricting practice to a specific medical specialty. However, a local entity may delineate scope of practice within its organization. For instance, local practice standards held by the Detroit Wayne Mental Health Authority (DWMHA) state “Psychiatrists…shall not exceed their scope of practice as per DWMHA credentialing and privileging. For example, a Psychiatrist…who [has] not been appropriately privileged to deliver services to children shall not treat children, excepting crisis situations.”1
Like physicians in other specialties, psychiatrists are not limited to prescribing only a subset of medications commonly associated with their specialty. But for many psychiatrists, prescribing nonpsychotropic medications is complicated by individual and local factors. On one hand, some psychiatrists do not feel it is their role to prescribe nonpsychotropic medications,2 or even some psychotropic medications that may be more complex to prescribe, such as lithium, clozapine, or monoamine oxidase inhibitors.3-5 However, many feel comfortable prescribing complex combinations of psychotropic medications, or prescribing in a way that does not necessarily make sense (eg, prescribing benztropine as prophylaxis for dystonia when starting an antipsychotic).
Reviewing an average day at one urban psychiatric clinic, these questions seem to come up in half of the patient population, especially in patients with chronic mental illness, multiple medical comorbidities, and limited access to health care. When a young patient walks in without an appointment with an acute dystonic reaction secondary to the initiation of antipsychotics a couple of days ago, there is no hesitation to swiftly and appropriately prescribe an IM anticholinergic medication. But why are psychiatrists often hesitant to prescribe nonpsychotropic medications to treat other adverse effects of medications? Lack of knowledge? Lack of training?
Psychiatrists who practice in hospital systems often have immediate access to consultants, and this availability may encourage them to defer to the consultant for treatment of certain adverse effects. We have seen psychiatrists consult Neurology regarding the prescription of donepezil for mild neurocognitive disorder due to Alzheimer’s disease, or Endocrinology regarding prescription of levothyroxine for lithium-induced hypothyroidism.
However, there are numerous scenarios in which psychiatrists should feel comfortable prescribing nonpsychotropic medications or managing medication adverse effects, regardless of whether they consider it to be within or outside their scope of practice. The following case examples illustrate several such situations.
CASE 1
Ms. W, age 30, has been diagnosed with schizophrenia. She requests a refill of quetiapine, 800 mg/d. This medication has been clearly beneficial in alleviating her psychotic symptoms. However, since her last visit 3 months ago, her face appears more round, and she has gained 9 kg. Further evaluation indicates that she has developed metabolic syndrome and pre-diabetes.
Continue to: Metabolic adverse effects
Metabolic adverse effects, such as metabolic syndrome, diabetic ketoacidosis, and cardiovascular disease, are well-known risks of prescribing second-generation antipsychotics.6 In such situations, psychiatrists often advise patients to modify their diet, increase physical activity, and follow up with their primary care physician to determine if other medications are needed. However, getting a patient with a serious mental illness to exercise and modify her/his diet is difficult, and many of these patients do not have a primary care physician.
For patients such as Ms. W, a psychiatrist should consider prescribing metformin. Wu et al7 found that in addition to lifestyle modifications, metformin had the greatest effect on antipsychotic-induced weight gain. In this study, metformin alone had more impact on reversing weight gain and increasing insulin sensitivity than lifestyle modifications alone.7 This is crucial because these patients are especially vulnerable to cardiac disease.8 Metformin is well tolerated and has a low risk of causing hypoglycemia. Concerns regarding lactic acidosis have abated to the extent that the estimated glomerular filtration rate (eGFR) limits for using metformin have been lowered significantly. After reviewing the contraindications, the only knowledge needed to prescribe metformin is the patient’s kidney function and a brief understanding of the titration needed to minimize gastrointestinal adverse effects.9 Thus, prescribing metformin would be a fairly logical and easy first step for managing metabolic syndrome, especially in a patient whose motivation for increasing physical activity and modifying his/her diet is doubtful.
CASE 2
Mr. B, age 45, has major depressive disorder that has been well-controlled on paroxetine, 40 mg/d, for the past 2 years. He has no history of physical illness. On his most recent visit, he appears uncomfortable and nervous. After a long discussion, he discloses that his sex life isn’t what it used to be since starting paroxetine. He is bothered by erectile problems and asks whether he can “get some Viagra.”
Sexual adverse effects, such as erectile dysfunction, are frequently associated with the use of selective serotonin reuptake inhibitors.10 Although managing these adverse effects requires careful evaluation, in most cases, psychiatrists should be able to treat them.10 The logical choice in this case would be to prescribe one of the 4 FDA-approved phosphodiesterase-5 inhibitors (sildenafil [Viagra], tadalafil [Cialis], vardenafil [Levitra], and avanafil [Stendra]. However, Balon et al11 found that few psychiatrists prescribe phosphodiesterase-5 inhibitors, although they believed that they should be prescribing to treat their patients’ sexual dysfunction. Managing these adverse effects is important not only for the patient’s quality of life and relationship with his/her partner, but also for the therapeutic alliance. In a systematic review of 23 trials, Taylor et al12 examined >1,800 patients who were prescribed a medication to address sexual dysfunction secondary to antidepressants. They found that for men, adding a phosphodiesterase-5 inhibitor was appropriate and effective, and for women, adding bupropion at high doses should be considered.12 Like many other adverse effects, sexual adverse effects surely play a role in medication compliance. Dording et al13 found that the addition of sildenafil, 50 to 100 mg as needed, resulted in increased treatment satisfaction and overall contentment in 102 patients who complained of sexual dysfunction in the follow-up phase of the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) antidepressant trials. In most cases, with proper psychoeducation, prescription of
CASE 3
Ms. G, age 22, was recently discharged from an inpatient psychiatric unit after an episode of mania. She was prescribed carbamazepine, 600 mg/d, and ziprasidone, 40 mg twice a day, and appears to be doing well on this regimen. When asked about what led to her admission, she recalls having an elevated mood, increased energy, hypersexuality, impulsivity, and poor judgment. She reveals that she had several sexual partners during her manic episode, and worries that if such behavior occurs again, she may get pregnant. Yet Ms. G was not prescribed birth control upon discharge.
Continue to: Contraception
Contraception. We believe that psychiatrists have an obligation to protect patients from consequences of mental illness. Much the same way that psychiatrists hope to prevent suicide in a patient who has depression, patients should be protected from risks encountered in the manic phase of bipolar disorder. Another reason to prescribe contraceptives in such patients is the teratogenic effects of mood stabilizers. Pagano et al14 reviewed 6 studies that examined common forms of hormonal birth control to determine their safety in patients with depression or bipolar disorder. They found that overall, use of hormonal contraception was not associated with a worse clinical course of disease.
Many available forms of birth control are available. When prescribing in an outpatient setting, a daily oral medication or a monthly depot injection are convenient options.
CASE 4
Mr. P, age 65, has bipolar I disorder and is stable on risperidone long-acting injection, 37.7 mg bimonthly, and lithium, 1,200 mg/d. He reports that he is doing well but has noticed a recent decrease in energy and weight gain without any change in mood. Laboratory testing conducted prior to this visit revealed a thyroid-stimulating hormone (TSH) level of 4 mU/L (normal range: 0.4 to 4.0 mU/L). Six months ago, Mr. P’s TSH level was 2.8 mU/L. The resident supervisor suggests discussing the case with an endocrinologist.
Thyroid function. The impact of lithium on the thyroid gland is well established; however, psychiatrists’ response to such changes are not.15 Gitlin16 reviewed the many adverse effects of lithium and presented various management strategies to address findings such as Mr. P’s. Two important points are that lithium should not be discontinued in light of hypothyroidism, and synthetic thyroxine (levothyroxine) can be initiated and titrated to return TSH levels to a normal range.16 Levothyroxine can be started at low doses (eg, 25 to 50 mcg/d) and increased every 6 weeks until a normal TSH level is achieved.17 Managing lithium-induced clinical or subclinical hypothyroidism can prevent further pathology and possible relapse to depression.
Incorporating integrated care
In all these cases, the prescription of a medication with which some psychiatrists are not comfortable prescribing would have been the logical, easiest, and preferable choice. Of course, when initiating any medication, boxed warnings, contraindications, and drug–drug interactions should be reviewed. Initial dosages and titration schedules can be found in every medication’s FDA-approved prescribing information document (package insert), as well as in numerous reference books and articles.
Continue to: We acknowledge...
We acknowledge that prescribing a nonpsychotropic medication is not always a psychiatrist’s best choice, and that in patients with multiple medical comorbidities and drug–drug interactions that are not clearly defined, referring to or consulting a specialist is appropriate. We in no way support reckless prescribing, but instead present an opportunity to expand the perception of what should be considered within a psychiatrist’s scope of practice, and call for further education of psychiatrists so that they are more comfortable managing these adverse effects and/or prescribing at least some nonpsychotropic medications.

We exhort integrated medical care during this time of a physician shortage; however, we do not practice this way. Interestingly, physicians in primary care, such as those in family medicine or obstetrics and gynecology, frequently attempt to treat patients with psychiatric conditions in an attempt to provide integrated care. Numerous articles have discussed these efforts.18-20 However, this type of integrated care seems less frequent in psychiatry, even though the practice of modern psychiatry in the United States shows substantial overlap with the practice of physicians in primary care specialties.21 There are few articles or practical guidelines for psychiatrists who wish to treat patients’ physical illnesses, particularly patients with severe mental illness (see Related Resources, page 56). If we practice in an integrated manner to treat one of the simple conditions we described above, we can eliminate the need for a patient to visit a second physician, pay another co-pay, pay another bus fare, and take another day off work. This can be particularly helpful for patients who at times have to decide between paying for groceries or for medications. Having one clinician manage a patient’s medications also can decrease the risk of polypharmacy.
In addition to the case scenarios described in this article, there are more clinical situations and nonpsychotropic medications that psychiatrists could manage. Considering them outside the scope of psychiatric practice and being uncomfortable or ambivalent about them is not an excuse. We hope that psychiatrists can increase their expertise in this area, and can start to practice as the primary care physicians they claim they are, and should be.
Bottom Line
Many psychiatrists are uncomfortable prescribing nonpsychotropic medications, but there are numerous clinical scenarios in which the practice would make sense. This could include cases of metabolic syndrome, sexual dysfunction secondary to antidepressant use, or other adverse effects of commonly prescribed psychotropic medications.
Related Resources
- McCarron RM, Xiong GL, Keenan CR, et al. Preventive medical care in psychiatry. A practical guide for clinicians. Arlington, VA: American Psychiatric Association Publishing; 2015.
- McCarron RM, Xiong GL, Keenan CR, et al. Study guide to preventive medical care in psychiatry. Arlington, VA: American Psychiatric Association Publishing; 2017.
- Goldberg JF, Ernst CL. Managing the side effects of psychotropic medications. Washington, DC: American Psychiatric Association Publishing; 2019.
Drug Brand Names
Avanafil • Stendra
Benztropine • Cogentin
Bupropion • Wellbutrin, Zyban
Carbamazepine • Carbatrol, Tegretol
Clozapine • Clozaril
Donepezil • Aricept
Levothyroxine • Levoxyl, Synthroid
Lithium • Eskalith, Lithobid
Metformin • Fortamet, Glucophage
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone long-acting injection • Risperdal Consta
Sildenafil • Viagra
Tadalafil • Cialis
Vardenafil • Levitra
Ziprasidone • Geodon
In our experience, most psychiatrists are uncomfortable with prescribing a medication when they feel that doing so would be outside their scope of practice. But there are many situations when prescribing a nonpsychotropic medication would be the correct choice. In this article, we discuss the scope of psychiatric practice, and present 4 case studies that illustrate situations in which psychiatrists should feel comfortable prescribing nonpsychotropic medications.
Defining the scope of practice
What is the scope of a psychiatrist’s practice? Scope of practice usually describes activities that a health care practitioner is allowed to undertake as defined by the terms of his/her license. A license to practice medicine does not include any stipulation restricting practice to a specific medical specialty. However, a local entity may delineate scope of practice within its organization. For instance, local practice standards held by the Detroit Wayne Mental Health Authority (DWMHA) state “Psychiatrists…shall not exceed their scope of practice as per DWMHA credentialing and privileging. For example, a Psychiatrist…who [has] not been appropriately privileged to deliver services to children shall not treat children, excepting crisis situations.”1
Like physicians in other specialties, psychiatrists are not limited to prescribing only a subset of medications commonly associated with their specialty. But for many psychiatrists, prescribing nonpsychotropic medications is complicated by individual and local factors. On one hand, some psychiatrists do not feel it is their role to prescribe nonpsychotropic medications,2 or even some psychotropic medications that may be more complex to prescribe, such as lithium, clozapine, or monoamine oxidase inhibitors.3-5 However, many feel comfortable prescribing complex combinations of psychotropic medications, or prescribing in a way that does not necessarily make sense (eg, prescribing benztropine as prophylaxis for dystonia when starting an antipsychotic).
Reviewing an average day at one urban psychiatric clinic, these questions seem to come up in half of the patient population, especially in patients with chronic mental illness, multiple medical comorbidities, and limited access to health care. When a young patient walks in without an appointment with an acute dystonic reaction secondary to the initiation of antipsychotics a couple of days ago, there is no hesitation to swiftly and appropriately prescribe an IM anticholinergic medication. But why are psychiatrists often hesitant to prescribe nonpsychotropic medications to treat other adverse effects of medications? Lack of knowledge? Lack of training?
Psychiatrists who practice in hospital systems often have immediate access to consultants, and this availability may encourage them to defer to the consultant for treatment of certain adverse effects. We have seen psychiatrists consult Neurology regarding the prescription of donepezil for mild neurocognitive disorder due to Alzheimer’s disease, or Endocrinology regarding prescription of levothyroxine for lithium-induced hypothyroidism.
However, there are numerous scenarios in which psychiatrists should feel comfortable prescribing nonpsychotropic medications or managing medication adverse effects, regardless of whether they consider it to be within or outside their scope of practice. The following case examples illustrate several such situations.
CASE 1
Ms. W, age 30, has been diagnosed with schizophrenia. She requests a refill of quetiapine, 800 mg/d. This medication has been clearly beneficial in alleviating her psychotic symptoms. However, since her last visit 3 months ago, her face appears more round, and she has gained 9 kg. Further evaluation indicates that she has developed metabolic syndrome and pre-diabetes.
Continue to: Metabolic adverse effects
Metabolic adverse effects, such as metabolic syndrome, diabetic ketoacidosis, and cardiovascular disease, are well-known risks of prescribing second-generation antipsychotics.6 In such situations, psychiatrists often advise patients to modify their diet, increase physical activity, and follow up with their primary care physician to determine if other medications are needed. However, getting a patient with a serious mental illness to exercise and modify her/his diet is difficult, and many of these patients do not have a primary care physician.
For patients such as Ms. W, a psychiatrist should consider prescribing metformin. Wu et al7 found that in addition to lifestyle modifications, metformin had the greatest effect on antipsychotic-induced weight gain. In this study, metformin alone had more impact on reversing weight gain and increasing insulin sensitivity than lifestyle modifications alone.7 This is crucial because these patients are especially vulnerable to cardiac disease.8 Metformin is well tolerated and has a low risk of causing hypoglycemia. Concerns regarding lactic acidosis have abated to the extent that the estimated glomerular filtration rate (eGFR) limits for using metformin have been lowered significantly. After reviewing the contraindications, the only knowledge needed to prescribe metformin is the patient’s kidney function and a brief understanding of the titration needed to minimize gastrointestinal adverse effects.9 Thus, prescribing metformin would be a fairly logical and easy first step for managing metabolic syndrome, especially in a patient whose motivation for increasing physical activity and modifying his/her diet is doubtful.
CASE 2
Mr. B, age 45, has major depressive disorder that has been well-controlled on paroxetine, 40 mg/d, for the past 2 years. He has no history of physical illness. On his most recent visit, he appears uncomfortable and nervous. After a long discussion, he discloses that his sex life isn’t what it used to be since starting paroxetine. He is bothered by erectile problems and asks whether he can “get some Viagra.”
Sexual adverse effects, such as erectile dysfunction, are frequently associated with the use of selective serotonin reuptake inhibitors.10 Although managing these adverse effects requires careful evaluation, in most cases, psychiatrists should be able to treat them.10 The logical choice in this case would be to prescribe one of the 4 FDA-approved phosphodiesterase-5 inhibitors (sildenafil [Viagra], tadalafil [Cialis], vardenafil [Levitra], and avanafil [Stendra]. However, Balon et al11 found that few psychiatrists prescribe phosphodiesterase-5 inhibitors, although they believed that they should be prescribing to treat their patients’ sexual dysfunction. Managing these adverse effects is important not only for the patient’s quality of life and relationship with his/her partner, but also for the therapeutic alliance. In a systematic review of 23 trials, Taylor et al12 examined >1,800 patients who were prescribed a medication to address sexual dysfunction secondary to antidepressants. They found that for men, adding a phosphodiesterase-5 inhibitor was appropriate and effective, and for women, adding bupropion at high doses should be considered.12 Like many other adverse effects, sexual adverse effects surely play a role in medication compliance. Dording et al13 found that the addition of sildenafil, 50 to 100 mg as needed, resulted in increased treatment satisfaction and overall contentment in 102 patients who complained of sexual dysfunction in the follow-up phase of the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) antidepressant trials. In most cases, with proper psychoeducation, prescription of
CASE 3
Ms. G, age 22, was recently discharged from an inpatient psychiatric unit after an episode of mania. She was prescribed carbamazepine, 600 mg/d, and ziprasidone, 40 mg twice a day, and appears to be doing well on this regimen. When asked about what led to her admission, she recalls having an elevated mood, increased energy, hypersexuality, impulsivity, and poor judgment. She reveals that she had several sexual partners during her manic episode, and worries that if such behavior occurs again, she may get pregnant. Yet Ms. G was not prescribed birth control upon discharge.
Continue to: Contraception
Contraception. We believe that psychiatrists have an obligation to protect patients from consequences of mental illness. Much the same way that psychiatrists hope to prevent suicide in a patient who has depression, patients should be protected from risks encountered in the manic phase of bipolar disorder. Another reason to prescribe contraceptives in such patients is the teratogenic effects of mood stabilizers. Pagano et al14 reviewed 6 studies that examined common forms of hormonal birth control to determine their safety in patients with depression or bipolar disorder. They found that overall, use of hormonal contraception was not associated with a worse clinical course of disease.
Many available forms of birth control are available. When prescribing in an outpatient setting, a daily oral medication or a monthly depot injection are convenient options.
CASE 4
Mr. P, age 65, has bipolar I disorder and is stable on risperidone long-acting injection, 37.7 mg bimonthly, and lithium, 1,200 mg/d. He reports that he is doing well but has noticed a recent decrease in energy and weight gain without any change in mood. Laboratory testing conducted prior to this visit revealed a thyroid-stimulating hormone (TSH) level of 4 mU/L (normal range: 0.4 to 4.0 mU/L). Six months ago, Mr. P’s TSH level was 2.8 mU/L. The resident supervisor suggests discussing the case with an endocrinologist.
Thyroid function. The impact of lithium on the thyroid gland is well established; however, psychiatrists’ response to such changes are not.15 Gitlin16 reviewed the many adverse effects of lithium and presented various management strategies to address findings such as Mr. P’s. Two important points are that lithium should not be discontinued in light of hypothyroidism, and synthetic thyroxine (levothyroxine) can be initiated and titrated to return TSH levels to a normal range.16 Levothyroxine can be started at low doses (eg, 25 to 50 mcg/d) and increased every 6 weeks until a normal TSH level is achieved.17 Managing lithium-induced clinical or subclinical hypothyroidism can prevent further pathology and possible relapse to depression.
Incorporating integrated care
In all these cases, the prescription of a medication with which some psychiatrists are not comfortable prescribing would have been the logical, easiest, and preferable choice. Of course, when initiating any medication, boxed warnings, contraindications, and drug–drug interactions should be reviewed. Initial dosages and titration schedules can be found in every medication’s FDA-approved prescribing information document (package insert), as well as in numerous reference books and articles.
Continue to: We acknowledge...
We acknowledge that prescribing a nonpsychotropic medication is not always a psychiatrist’s best choice, and that in patients with multiple medical comorbidities and drug–drug interactions that are not clearly defined, referring to or consulting a specialist is appropriate. We in no way support reckless prescribing, but instead present an opportunity to expand the perception of what should be considered within a psychiatrist’s scope of practice, and call for further education of psychiatrists so that they are more comfortable managing these adverse effects and/or prescribing at least some nonpsychotropic medications.

We exhort integrated medical care during this time of a physician shortage; however, we do not practice this way. Interestingly, physicians in primary care, such as those in family medicine or obstetrics and gynecology, frequently attempt to treat patients with psychiatric conditions in an attempt to provide integrated care. Numerous articles have discussed these efforts.18-20 However, this type of integrated care seems less frequent in psychiatry, even though the practice of modern psychiatry in the United States shows substantial overlap with the practice of physicians in primary care specialties.21 There are few articles or practical guidelines for psychiatrists who wish to treat patients’ physical illnesses, particularly patients with severe mental illness (see Related Resources, page 56). If we practice in an integrated manner to treat one of the simple conditions we described above, we can eliminate the need for a patient to visit a second physician, pay another co-pay, pay another bus fare, and take another day off work. This can be particularly helpful for patients who at times have to decide between paying for groceries or for medications. Having one clinician manage a patient’s medications also can decrease the risk of polypharmacy.
In addition to the case scenarios described in this article, there are more clinical situations and nonpsychotropic medications that psychiatrists could manage. Considering them outside the scope of psychiatric practice and being uncomfortable or ambivalent about them is not an excuse. We hope that psychiatrists can increase their expertise in this area, and can start to practice as the primary care physicians they claim they are, and should be.
Bottom Line
Many psychiatrists are uncomfortable prescribing nonpsychotropic medications, but there are numerous clinical scenarios in which the practice would make sense. This could include cases of metabolic syndrome, sexual dysfunction secondary to antidepressant use, or other adverse effects of commonly prescribed psychotropic medications.
Related Resources
- McCarron RM, Xiong GL, Keenan CR, et al. Preventive medical care in psychiatry. A practical guide for clinicians. Arlington, VA: American Psychiatric Association Publishing; 2015.
- McCarron RM, Xiong GL, Keenan CR, et al. Study guide to preventive medical care in psychiatry. Arlington, VA: American Psychiatric Association Publishing; 2017.
- Goldberg JF, Ernst CL. Managing the side effects of psychotropic medications. Washington, DC: American Psychiatric Association Publishing; 2019.
Drug Brand Names
Avanafil • Stendra
Benztropine • Cogentin
Bupropion • Wellbutrin, Zyban
Carbamazepine • Carbatrol, Tegretol
Clozapine • Clozaril
Donepezil • Aricept
Levothyroxine • Levoxyl, Synthroid
Lithium • Eskalith, Lithobid
Metformin • Fortamet, Glucophage
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone long-acting injection • Risperdal Consta
Sildenafil • Viagra
Tadalafil • Cialis
Vardenafil • Levitra
Ziprasidone • Geodon
1. Detroit Wayne Integrated Health Network. DWMHA psychiatric practice standards. http://dwihn.org/files/2015/6451/9628/Psychiatric_Practice_Standards.pdf. Revised June 2018. Accessed October 8, 2019.
2. Seaman JJ, Cornfield RM, Cummings DM, et al. Exploring psychiatric prescribing practices: the relationship between the role of the provider and the appropriateness of prescribing. Gen Hosp Psychiatry. 1987;9(3):220-224.
3. Zivanovic O. Lithium: a classic drug—frequently discussed, but, sadly, seldom prescribed! Aust N Z J Psychiatry. 2017;51(9):886-896.
4. Stroup TS, Gerhard T, Crystal S, et al. Geographic and clinical variation in clozapine use in the United States. Psychiatric Services. 2014;65(2):186-192.
5. Balon R, Mufti R, Arfken C. A survey of prescribing practices for monoamine oxidase inhibitors. Psychiatric Services. 1999;50(7):945-947.
6. Rummel-Kluge C, Komossa K, Schwarz S, et al. Head-to-head comparisons of metabolic side effects of second generation antipsychotics in the treatment of schizophrenia: a systematic review and meta-analysis. Schizophr Res. 2010;123(2-3):225-233.
7. Wu RR, Zhao JP, Jin H, et al. Lifestyle intervention and metformin for treatment of antipsychotic-induced weight gain: a randomized controlled trial. JAMA. 2008;299(2):185-193.
8. De Hert M, Correll CU, Bobes J, et al. Physical illness in patients with severe mental disorders. I. Prevalence, impact of medications and disparities in health care. World Psychiatry. 2011;10(1):52-77.
9. Kirpichnikov D, McFarlane SI, Sowers JR. Metformin: an update. Ann Internal Med. 2002;137(1):25-33.
10. Balon R. SSRI-associated sexual dysfunction. Am J Psychiatry. 2006;163(9):1504-1509.
11. Balon R, Morreale MK, Segraves RT. Prescribing of phosphodiesterase-5 inhibitors among psychiatrists. J Sex Marital Ther. 2014;40(3):165-169.
12. Taylor MJ, Rudkin L, Bullemor-Day P, et al. Strategies for managing sexual dysfunction induced by antidepressant medication. Cochrane Database Syst Rev. 2013;(5):CD003382.
13. Dording CM, LaRocca RA, Hails KA, et al. The effect of sildenafil on quality of life. Ann Clin Psychiatry. 2013;25(1):3-10.
14. Pagano HP, Zapata LB, Berry-Bibee EN, et al. Safety of hormonal contraception and intrauterine devices among women with depressive and bipolar disorders: a systematic review. Contraception. 2016;94(6):641-649.
15. Kibirige D, Luzinda K, Ssekitoleko R. Spectrum of lithium induced thyroid abnormalities: a current perspective. Thyroid Res. 2013;6(1):3.
16. Gitlin M. Lithium side effects and toxicity: prevalence and management strategies. Int J Bipolar Disord. 2016;4(1):27.
17. Devdhar M, Ousman YH, Burman KD. Hypothyroidism. Endocrinol Metab Clin North Am. 2007;36(3):595-615.
18. Hackley B, Sharma C, Kedzior A, et al. Managing mental health conditions in primary care settings. J Midwifery Women’s Health. 2010;55(1):9-19.
19. Fitelson E, McGibbon C. Evaluation and management of behavioral health disorders in women: an overview of major depression, bipolar disorder, anxiety disorders, and sleep in the primary care setting. Obstet Gynecol Clin North Am. 2016;43(2):231-246.
20. Colorafi K, Vanselow J, Nelson T. Treating anxiety and depression in primary care: reducing barriers to access. Fam Pract Manag. 2017;24(4):11-16.
21. McCall WV. Defining the unique scope of psychiatric practice in 2015. J ECT. 2015;31(4):203-204.
1. Detroit Wayne Integrated Health Network. DWMHA psychiatric practice standards. http://dwihn.org/files/2015/6451/9628/Psychiatric_Practice_Standards.pdf. Revised June 2018. Accessed October 8, 2019.
2. Seaman JJ, Cornfield RM, Cummings DM, et al. Exploring psychiatric prescribing practices: the relationship between the role of the provider and the appropriateness of prescribing. Gen Hosp Psychiatry. 1987;9(3):220-224.
3. Zivanovic O. Lithium: a classic drug—frequently discussed, but, sadly, seldom prescribed! Aust N Z J Psychiatry. 2017;51(9):886-896.
4. Stroup TS, Gerhard T, Crystal S, et al. Geographic and clinical variation in clozapine use in the United States. Psychiatric Services. 2014;65(2):186-192.
5. Balon R, Mufti R, Arfken C. A survey of prescribing practices for monoamine oxidase inhibitors. Psychiatric Services. 1999;50(7):945-947.
6. Rummel-Kluge C, Komossa K, Schwarz S, et al. Head-to-head comparisons of metabolic side effects of second generation antipsychotics in the treatment of schizophrenia: a systematic review and meta-analysis. Schizophr Res. 2010;123(2-3):225-233.
7. Wu RR, Zhao JP, Jin H, et al. Lifestyle intervention and metformin for treatment of antipsychotic-induced weight gain: a randomized controlled trial. JAMA. 2008;299(2):185-193.
8. De Hert M, Correll CU, Bobes J, et al. Physical illness in patients with severe mental disorders. I. Prevalence, impact of medications and disparities in health care. World Psychiatry. 2011;10(1):52-77.
9. Kirpichnikov D, McFarlane SI, Sowers JR. Metformin: an update. Ann Internal Med. 2002;137(1):25-33.
10. Balon R. SSRI-associated sexual dysfunction. Am J Psychiatry. 2006;163(9):1504-1509.
11. Balon R, Morreale MK, Segraves RT. Prescribing of phosphodiesterase-5 inhibitors among psychiatrists. J Sex Marital Ther. 2014;40(3):165-169.
12. Taylor MJ, Rudkin L, Bullemor-Day P, et al. Strategies for managing sexual dysfunction induced by antidepressant medication. Cochrane Database Syst Rev. 2013;(5):CD003382.
13. Dording CM, LaRocca RA, Hails KA, et al. The effect of sildenafil on quality of life. Ann Clin Psychiatry. 2013;25(1):3-10.
14. Pagano HP, Zapata LB, Berry-Bibee EN, et al. Safety of hormonal contraception and intrauterine devices among women with depressive and bipolar disorders: a systematic review. Contraception. 2016;94(6):641-649.
15. Kibirige D, Luzinda K, Ssekitoleko R. Spectrum of lithium induced thyroid abnormalities: a current perspective. Thyroid Res. 2013;6(1):3.
16. Gitlin M. Lithium side effects and toxicity: prevalence and management strategies. Int J Bipolar Disord. 2016;4(1):27.
17. Devdhar M, Ousman YH, Burman KD. Hypothyroidism. Endocrinol Metab Clin North Am. 2007;36(3):595-615.
18. Hackley B, Sharma C, Kedzior A, et al. Managing mental health conditions in primary care settings. J Midwifery Women’s Health. 2010;55(1):9-19.
19. Fitelson E, McGibbon C. Evaluation and management of behavioral health disorders in women: an overview of major depression, bipolar disorder, anxiety disorders, and sleep in the primary care setting. Obstet Gynecol Clin North Am. 2016;43(2):231-246.
20. Colorafi K, Vanselow J, Nelson T. Treating anxiety and depression in primary care: reducing barriers to access. Fam Pract Manag. 2017;24(4):11-16.
21. McCall WV. Defining the unique scope of psychiatric practice in 2015. J ECT. 2015;31(4):203-204.
‘Joker’ filled with mental illness misconceptions
The Batman characters have been cultural icons for generations – spanning more than three-quarters of a century. How many of us had Batman (or the Joker) on our school lunch box or watched reruns of Adam West’s campy televised rendition of Batman? The October release of “Joker” has been breaking contemporary box office records.
(Spoiler alert!) The Todd Phillips film associates mental illness with violent acts, spurring a slew of articles explaining that this association is uncommon and may promote stigmatization and public fear of people with obvious symptoms of mental illness. The protagonist, Arthur Fleck (Joaquin Phoenix), suffers from a condition in which his affect and facial expressions are not appropriate to his emotions or to the situation. He laughs uncontrollably when a situation is sad or upsetting. Sometimes he laughs and cries at the same time. As a result, he often is misunderstood, ridiculed, and victimized – like many people with obvious mental illness.
Arthur Fleck is a loner who has difficulty with relationships and self-esteem, and is beaten severely while at work as a clown. Shortly after the incident, he is given a gun by one of his coworkers. He keeps it with him even when working as a clown in a children’s hospital – where it is accidentally revealed, and he is subsequently fired. Still in his clown garb, he later uses the gun when he is mocked and assaulted on the subway by three Gotham City bankers.
In an unusual tone, his mental health worker reminds him early in the film that he is prescribed seven different psychotropic medications, helping to cement for the viewer that mental illness is the cause of Arthur’s problems and the Joker’s origin story. Then the funding for Arthur’s mental health treatment (even if it was not good treatment) was cut – a problem not just in Gotham.
While some of Arthur Fleck’s symptoms are consistent with real mental illness, the combination of symptoms is unusual. Although he is being treated with a variety of medications, it is unclear whether any of them are helping him or what exactly they are helping him with. (Ironically, once he is off of his medications, he becomes a better dresser and a better dancer.) He writes in a disorganized way in his journal; the only intelligible sentence that is focused on is, “The worst part about having mental illness is people expect you to behave as if you DONT.” A smiley face in the ‘O’ suggests that his affect is inappropriate even in his writing. Arthur’s condition of uncontrollable laughing and/or crying, associated with head trauma, appears more consistent with the neurologic condition pseudobulbar affect rather than a mental illness. In addition to pseudobulbar affect, Arthur demonstrates a constellation of symptoms of different kinds of mental illness, including erotomanic delusions, ideas of reference, and disorganized thinking. He also does not appear to take social cues, such as knowing when he is being mocked. He appears to believe that his neighbor is his girlfriend (as the viewer was similarly led to believe), eventually breaking into her apartment where he thought he belonged, much to her horror when she finds him there. Some of his symptoms may run in his family (whether it be his biological or adoptive family).

Penny (Arthur’s mother) strongly believes (perhaps a delusion, perhaps not) that her previous employer Thomas Wayne (the future Batman’s father) is the father of her love-child, Arthur. When Arthur obtains Penny’s mental health records (through his own violent devices), he finds that she had been diagnosed with narcissistic personality disorder and a psychotic disorder. She had been found guilty of endangering the welfare of her (perhaps adopted, perhaps not) child Arthur, who had been malnourished, with severe head trauma, and tied to a radiator.
Arthur’s smothering of his mother with a pillow in her hospital bed, after he was devastated by both her stroke and this newfound data, occurred in a perfect storm. The killing is not portrayed as an act of euthanasia. We know that schizophrenia is overrepresented among matricide perpetrators and that long-term dysfunctional relationships between mother and (grown) child usually precede matricides. Mothers are often seen as controlling, fathers are often absent (as in Arthur’s case), and the child is often overly dependent. The mother and child (as seen here) often have a relationship marked by love and hate – mutual dependence and hostility. But Arthur is not the only character in the Batman universe to commit matricide. Recall that the Batman’s psychiatrist Amadeus Arkham himself killed his own mentally ill mother during his young adulthood.
Pop culture can give the public negative impressions of mental illness. While filmmakers need not portray actual mental illnesses or their symptoms in moving their stories forward, their portrayals have an impact on what the public sees as mental illness. This is similar to the current American president and others in political power asserting that mental illness causes mass shootings, and those in the public taking their word for it rather than the word of psychiatry.
In actuality, what felt the most true to life in the film was the early scene in which Arthur was seriously assaulted while waving the going-out-of-business sign on the sidewalk, just trying to make a living. As psychiatrists know, people with mental illness are more likely to be victimized by others in society than to be perpetrators of violence. To be sure, some of Arthur’s characteristics are dynamic risk factors, such as his unemployment and social isolation. However, society often conflates mental illness with dangerousness, but most people with mental illness are not violent.
In the final scenes, Arthur Fleck (who is now the Joker) is apparently back in the white-walled Arkham State Hospital, with an implication that he has gotten away with the murders, either found incompetent or insane. This, too, has negative implications for the public viewing the film – and further perpetuates the misunderstanding that people with mental illness “get away” with their crimes. In reality, depending on the study, approximately one-quarter of those who pleaded insanity were found insane, and those facing jury trials (and public perception) are less likely to be found insane than those with bench trials. Public misinterpretations and outrage over the idea that a mentally unwell person might be found insane rather than guilty have existed for centuries, perhaps most memorably when John Hinckley Jr. attempted to assassinate former President Ronald Reagan, after identifying with a character in the film “Taxi Driver.” Let’s presume that Gotham has an insanity defense similar to other places in America. Then, in order to be found insane, Arthur’s pseudobulbar affect or his (unclear) mental illness would have either caused him not to know the nature and consequences of his acts, and/or to appreciate the wrongfulness of his acts (if we are fairly certain that Gotham is actually New York City). Neither of these appear to be true from the film. He knew that he was killing. No delusions or hallucinations made him think his acts were not wrong. Rather, he had an arguably rational motive – certainly the multitudes wearing clown masks in the subsequent uprisings against the powerful also believed his motive to be rational. He deliberately killed the bankers who mocked and beat him. He was also able to defer his killings until what he calculated was the right time to have the most impact – for example, on live television, or when he was alone with his mother in the hospital.
In closing, unrealistic portrayals of the link between mental illness, violence, and forensic hospitalization are seen on the silver screen in “Joker.” We hope that others who feign mental illness symptoms to evade criminal responsibility will emulate Joaquin Phoenix’s Joker as it will make it much easier for forensic psychiatrists to ferret out malingerers!
Dr. Hatters Friedman serves as the Phillip Resnick Professor of Forensic Psychiatry at Case Western Reserve University, Cleveland. She is also editor of Family Murder: Pathologies of Love and Hate (Washington, D.C.: American Psychiatric Association Publishing [2019]), which was written by the Group for the Advancement of Psychiatry’s Committee on Psychiatry & Law. Dr. Rosenbaum is a clinical and forensic psychiatrist in private practice in New York. She is an assistant clinical professor at New York University Langone Medical Center and on the faculty at Weill-Cornell Medical Center.
The Batman characters have been cultural icons for generations – spanning more than three-quarters of a century. How many of us had Batman (or the Joker) on our school lunch box or watched reruns of Adam West’s campy televised rendition of Batman? The October release of “Joker” has been breaking contemporary box office records.
(Spoiler alert!) The Todd Phillips film associates mental illness with violent acts, spurring a slew of articles explaining that this association is uncommon and may promote stigmatization and public fear of people with obvious symptoms of mental illness. The protagonist, Arthur Fleck (Joaquin Phoenix), suffers from a condition in which his affect and facial expressions are not appropriate to his emotions or to the situation. He laughs uncontrollably when a situation is sad or upsetting. Sometimes he laughs and cries at the same time. As a result, he often is misunderstood, ridiculed, and victimized – like many people with obvious mental illness.
Arthur Fleck is a loner who has difficulty with relationships and self-esteem, and is beaten severely while at work as a clown. Shortly after the incident, he is given a gun by one of his coworkers. He keeps it with him even when working as a clown in a children’s hospital – where it is accidentally revealed, and he is subsequently fired. Still in his clown garb, he later uses the gun when he is mocked and assaulted on the subway by three Gotham City bankers.
In an unusual tone, his mental health worker reminds him early in the film that he is prescribed seven different psychotropic medications, helping to cement for the viewer that mental illness is the cause of Arthur’s problems and the Joker’s origin story. Then the funding for Arthur’s mental health treatment (even if it was not good treatment) was cut – a problem not just in Gotham.
While some of Arthur Fleck’s symptoms are consistent with real mental illness, the combination of symptoms is unusual. Although he is being treated with a variety of medications, it is unclear whether any of them are helping him or what exactly they are helping him with. (Ironically, once he is off of his medications, he becomes a better dresser and a better dancer.) He writes in a disorganized way in his journal; the only intelligible sentence that is focused on is, “The worst part about having mental illness is people expect you to behave as if you DONT.” A smiley face in the ‘O’ suggests that his affect is inappropriate even in his writing. Arthur’s condition of uncontrollable laughing and/or crying, associated with head trauma, appears more consistent with the neurologic condition pseudobulbar affect rather than a mental illness. In addition to pseudobulbar affect, Arthur demonstrates a constellation of symptoms of different kinds of mental illness, including erotomanic delusions, ideas of reference, and disorganized thinking. He also does not appear to take social cues, such as knowing when he is being mocked. He appears to believe that his neighbor is his girlfriend (as the viewer was similarly led to believe), eventually breaking into her apartment where he thought he belonged, much to her horror when she finds him there. Some of his symptoms may run in his family (whether it be his biological or adoptive family).

Penny (Arthur’s mother) strongly believes (perhaps a delusion, perhaps not) that her previous employer Thomas Wayne (the future Batman’s father) is the father of her love-child, Arthur. When Arthur obtains Penny’s mental health records (through his own violent devices), he finds that she had been diagnosed with narcissistic personality disorder and a psychotic disorder. She had been found guilty of endangering the welfare of her (perhaps adopted, perhaps not) child Arthur, who had been malnourished, with severe head trauma, and tied to a radiator.
Arthur’s smothering of his mother with a pillow in her hospital bed, after he was devastated by both her stroke and this newfound data, occurred in a perfect storm. The killing is not portrayed as an act of euthanasia. We know that schizophrenia is overrepresented among matricide perpetrators and that long-term dysfunctional relationships between mother and (grown) child usually precede matricides. Mothers are often seen as controlling, fathers are often absent (as in Arthur’s case), and the child is often overly dependent. The mother and child (as seen here) often have a relationship marked by love and hate – mutual dependence and hostility. But Arthur is not the only character in the Batman universe to commit matricide. Recall that the Batman’s psychiatrist Amadeus Arkham himself killed his own mentally ill mother during his young adulthood.
Pop culture can give the public negative impressions of mental illness. While filmmakers need not portray actual mental illnesses or their symptoms in moving their stories forward, their portrayals have an impact on what the public sees as mental illness. This is similar to the current American president and others in political power asserting that mental illness causes mass shootings, and those in the public taking their word for it rather than the word of psychiatry.
In actuality, what felt the most true to life in the film was the early scene in which Arthur was seriously assaulted while waving the going-out-of-business sign on the sidewalk, just trying to make a living. As psychiatrists know, people with mental illness are more likely to be victimized by others in society than to be perpetrators of violence. To be sure, some of Arthur’s characteristics are dynamic risk factors, such as his unemployment and social isolation. However, society often conflates mental illness with dangerousness, but most people with mental illness are not violent.
In the final scenes, Arthur Fleck (who is now the Joker) is apparently back in the white-walled Arkham State Hospital, with an implication that he has gotten away with the murders, either found incompetent or insane. This, too, has negative implications for the public viewing the film – and further perpetuates the misunderstanding that people with mental illness “get away” with their crimes. In reality, depending on the study, approximately one-quarter of those who pleaded insanity were found insane, and those facing jury trials (and public perception) are less likely to be found insane than those with bench trials. Public misinterpretations and outrage over the idea that a mentally unwell person might be found insane rather than guilty have existed for centuries, perhaps most memorably when John Hinckley Jr. attempted to assassinate former President Ronald Reagan, after identifying with a character in the film “Taxi Driver.” Let’s presume that Gotham has an insanity defense similar to other places in America. Then, in order to be found insane, Arthur’s pseudobulbar affect or his (unclear) mental illness would have either caused him not to know the nature and consequences of his acts, and/or to appreciate the wrongfulness of his acts (if we are fairly certain that Gotham is actually New York City). Neither of these appear to be true from the film. He knew that he was killing. No delusions or hallucinations made him think his acts were not wrong. Rather, he had an arguably rational motive – certainly the multitudes wearing clown masks in the subsequent uprisings against the powerful also believed his motive to be rational. He deliberately killed the bankers who mocked and beat him. He was also able to defer his killings until what he calculated was the right time to have the most impact – for example, on live television, or when he was alone with his mother in the hospital.
In closing, unrealistic portrayals of the link between mental illness, violence, and forensic hospitalization are seen on the silver screen in “Joker.” We hope that others who feign mental illness symptoms to evade criminal responsibility will emulate Joaquin Phoenix’s Joker as it will make it much easier for forensic psychiatrists to ferret out malingerers!
Dr. Hatters Friedman serves as the Phillip Resnick Professor of Forensic Psychiatry at Case Western Reserve University, Cleveland. She is also editor of Family Murder: Pathologies of Love and Hate (Washington, D.C.: American Psychiatric Association Publishing [2019]), which was written by the Group for the Advancement of Psychiatry’s Committee on Psychiatry & Law. Dr. Rosenbaum is a clinical and forensic psychiatrist in private practice in New York. She is an assistant clinical professor at New York University Langone Medical Center and on the faculty at Weill-Cornell Medical Center.
The Batman characters have been cultural icons for generations – spanning more than three-quarters of a century. How many of us had Batman (or the Joker) on our school lunch box or watched reruns of Adam West’s campy televised rendition of Batman? The October release of “Joker” has been breaking contemporary box office records.
(Spoiler alert!) The Todd Phillips film associates mental illness with violent acts, spurring a slew of articles explaining that this association is uncommon and may promote stigmatization and public fear of people with obvious symptoms of mental illness. The protagonist, Arthur Fleck (Joaquin Phoenix), suffers from a condition in which his affect and facial expressions are not appropriate to his emotions or to the situation. He laughs uncontrollably when a situation is sad or upsetting. Sometimes he laughs and cries at the same time. As a result, he often is misunderstood, ridiculed, and victimized – like many people with obvious mental illness.
Arthur Fleck is a loner who has difficulty with relationships and self-esteem, and is beaten severely while at work as a clown. Shortly after the incident, he is given a gun by one of his coworkers. He keeps it with him even when working as a clown in a children’s hospital – where it is accidentally revealed, and he is subsequently fired. Still in his clown garb, he later uses the gun when he is mocked and assaulted on the subway by three Gotham City bankers.
In an unusual tone, his mental health worker reminds him early in the film that he is prescribed seven different psychotropic medications, helping to cement for the viewer that mental illness is the cause of Arthur’s problems and the Joker’s origin story. Then the funding for Arthur’s mental health treatment (even if it was not good treatment) was cut – a problem not just in Gotham.
While some of Arthur Fleck’s symptoms are consistent with real mental illness, the combination of symptoms is unusual. Although he is being treated with a variety of medications, it is unclear whether any of them are helping him or what exactly they are helping him with. (Ironically, once he is off of his medications, he becomes a better dresser and a better dancer.) He writes in a disorganized way in his journal; the only intelligible sentence that is focused on is, “The worst part about having mental illness is people expect you to behave as if you DONT.” A smiley face in the ‘O’ suggests that his affect is inappropriate even in his writing. Arthur’s condition of uncontrollable laughing and/or crying, associated with head trauma, appears more consistent with the neurologic condition pseudobulbar affect rather than a mental illness. In addition to pseudobulbar affect, Arthur demonstrates a constellation of symptoms of different kinds of mental illness, including erotomanic delusions, ideas of reference, and disorganized thinking. He also does not appear to take social cues, such as knowing when he is being mocked. He appears to believe that his neighbor is his girlfriend (as the viewer was similarly led to believe), eventually breaking into her apartment where he thought he belonged, much to her horror when she finds him there. Some of his symptoms may run in his family (whether it be his biological or adoptive family).

Penny (Arthur’s mother) strongly believes (perhaps a delusion, perhaps not) that her previous employer Thomas Wayne (the future Batman’s father) is the father of her love-child, Arthur. When Arthur obtains Penny’s mental health records (through his own violent devices), he finds that she had been diagnosed with narcissistic personality disorder and a psychotic disorder. She had been found guilty of endangering the welfare of her (perhaps adopted, perhaps not) child Arthur, who had been malnourished, with severe head trauma, and tied to a radiator.
Arthur’s smothering of his mother with a pillow in her hospital bed, after he was devastated by both her stroke and this newfound data, occurred in a perfect storm. The killing is not portrayed as an act of euthanasia. We know that schizophrenia is overrepresented among matricide perpetrators and that long-term dysfunctional relationships between mother and (grown) child usually precede matricides. Mothers are often seen as controlling, fathers are often absent (as in Arthur’s case), and the child is often overly dependent. The mother and child (as seen here) often have a relationship marked by love and hate – mutual dependence and hostility. But Arthur is not the only character in the Batman universe to commit matricide. Recall that the Batman’s psychiatrist Amadeus Arkham himself killed his own mentally ill mother during his young adulthood.
Pop culture can give the public negative impressions of mental illness. While filmmakers need not portray actual mental illnesses or their symptoms in moving their stories forward, their portrayals have an impact on what the public sees as mental illness. This is similar to the current American president and others in political power asserting that mental illness causes mass shootings, and those in the public taking their word for it rather than the word of psychiatry.
In actuality, what felt the most true to life in the film was the early scene in which Arthur was seriously assaulted while waving the going-out-of-business sign on the sidewalk, just trying to make a living. As psychiatrists know, people with mental illness are more likely to be victimized by others in society than to be perpetrators of violence. To be sure, some of Arthur’s characteristics are dynamic risk factors, such as his unemployment and social isolation. However, society often conflates mental illness with dangerousness, but most people with mental illness are not violent.
In the final scenes, Arthur Fleck (who is now the Joker) is apparently back in the white-walled Arkham State Hospital, with an implication that he has gotten away with the murders, either found incompetent or insane. This, too, has negative implications for the public viewing the film – and further perpetuates the misunderstanding that people with mental illness “get away” with their crimes. In reality, depending on the study, approximately one-quarter of those who pleaded insanity were found insane, and those facing jury trials (and public perception) are less likely to be found insane than those with bench trials. Public misinterpretations and outrage over the idea that a mentally unwell person might be found insane rather than guilty have existed for centuries, perhaps most memorably when John Hinckley Jr. attempted to assassinate former President Ronald Reagan, after identifying with a character in the film “Taxi Driver.” Let’s presume that Gotham has an insanity defense similar to other places in America. Then, in order to be found insane, Arthur’s pseudobulbar affect or his (unclear) mental illness would have either caused him not to know the nature and consequences of his acts, and/or to appreciate the wrongfulness of his acts (if we are fairly certain that Gotham is actually New York City). Neither of these appear to be true from the film. He knew that he was killing. No delusions or hallucinations made him think his acts were not wrong. Rather, he had an arguably rational motive – certainly the multitudes wearing clown masks in the subsequent uprisings against the powerful also believed his motive to be rational. He deliberately killed the bankers who mocked and beat him. He was also able to defer his killings until what he calculated was the right time to have the most impact – for example, on live television, or when he was alone with his mother in the hospital.
In closing, unrealistic portrayals of the link between mental illness, violence, and forensic hospitalization are seen on the silver screen in “Joker.” We hope that others who feign mental illness symptoms to evade criminal responsibility will emulate Joaquin Phoenix’s Joker as it will make it much easier for forensic psychiatrists to ferret out malingerers!
Dr. Hatters Friedman serves as the Phillip Resnick Professor of Forensic Psychiatry at Case Western Reserve University, Cleveland. She is also editor of Family Murder: Pathologies of Love and Hate (Washington, D.C.: American Psychiatric Association Publishing [2019]), which was written by the Group for the Advancement of Psychiatry’s Committee on Psychiatry & Law. Dr. Rosenbaum is a clinical and forensic psychiatrist in private practice in New York. She is an assistant clinical professor at New York University Langone Medical Center and on the faculty at Weill-Cornell Medical Center.
Spotlight is on promising investigational antipsychotics
COPENHAGEN –
Two of the highlighted agents – pimavanserin and SEP-363856 – were designed to eschew the traditional antipsychotic target, the dopamine D2 receptor, in favor of other mechanisms of action aimed at the negative symptoms of schizophrenia, for which there is a long-recognized major unmet need for better therapies.
A third agent, known for now as ALKS 3831, is composed of a combination of olanzapine and samidorphan, an opioid receptor antagonist. This once-daily oral combination of olanzapine/samidorphan (OLA/SAM) is designed to retain the clinical efficacy of olanzapine while mitigating the drug’s limiting side effect of substantial weight gain.
OLA/SAM New Drug Application expected soon
Christine Graham, PhD, presented highlights of the pivotal phase 3 ENLIGHTEN-2 study, a double-blind clinical trial in which 661 U.S. outpatients with schizophrenia were randomized to OLA/SAM or olanzapine alone at 10 or 20 mg/day for 24 weeks, at which point everyone was switched to open-label OLA/SAM at 10 or 20 mg/10 mg for an additional 52-week extension safety study.
At 24 weeks, the OLA/SAM group had a mean 4.21% weight gain from baseline, significantly less than the 6.59% gain with olanzapine alone. A clinically meaningful and unwelcome weight gain of 7% or greater occurred in 27.5% of OLA/SAM patients, compared with 42.7% of controls, for an adjusted 50% reduction in risk in the group on the investigational medication. Similarly, a 10% or greater weight gain occurred in 17.8% of OLA/SAM patients and 29.8% of controls; once again, that represented a 50% relative risk reduction. The two therapies were equally effective, achieving roughly 10-point reductions in the Positive and Negative Syndrome Scale (PANSS) for schizophrenia total score.
Both treatments showed similar weight gain trajectories for the first 4 weeks. However, by week 6 the trajectories diverged, with body weight plateauing in the OLA/SAM group and remaining stable throughout the remainder of the 76-week, two-part study. Meanwhile, body weight continued to climb in the olanzapine-only group throughout the 24 weeks, reported Dr. Graham, senior clinical research scientist at Alkermes, in Waltham, Mass.
“The waist circumference results were surprising: We saw that waist circumference separated between the two groups as early as week 1, considerably earlier than the week 6 separation in weight. This suggests to us that even when weight gain is similar between the two treatments, OLA/SAM is showing an early effect at limiting central fat accumulation – and this has important health implications, as central fat has been shown to be potentially pathogenic for developing diabetes, cardiovascular disease, and even some forms of cancer,” she said.
The safety profile of OLA/SAM was essentially the same as for olanzapine-only, with the exception of the weight gain.
Alkermes is planning to submit its New Drug Application for OLA/SAM to the Food and Drug Administration before the year’s end. FDA officials have urged the company to broaden the application to include not only the treatment of schizophrenia, but bipolar I disorder as well, since olanzapine is an approved, well-established treatment for that disorder. Dr. Graham and coinvestigators have demonstrated that OLA/SAM has no clinically significant effect on the pharmacokinetics of lithium or valproate (Clin Drug Investig. 2019 Oct 4. doi: 10.1007/s40261-019-00860-y).
Phase 3 trial on pimavanserin underway
Pimavanserin is an oral selective serotonin inverse agonist, or SSIA, with a high affinity for 5-HT2A receptors, very low affinity for 5-HT2C receptors, and “absolutely no affinity” for dopaminergic, histaminergic, adrenergic, or muscarinic receptors, explained Dragana Bugarski-Kirola, MD, a psychiatrist and vice president of clinical development at Acadia Pharmaceuticals in San Diego.
“Those sites are thought to contribute to sedation, cognitive impairment, and orthostatic hypotension,” she noted.
Pimavanserin is at present FDA approved for a narrow indication: Treatment of hallucinations and delusions associated with Parkinson’s disease psychosis. But the drug’s unique mechanism of action suggests broad efficacy across a range of psychiatric disorders.
Indeed, after a successful phase 2 clinical trial of pimavanserin for treatment of Alzheimer’s-related psychosis, a phase 3 randomized, double-blind, placebo-controlled clinical trial of the drug for relapse prevention in dementia-related psychosis is now enrolling a planned 360 outpatients at 95 centers in 13 countries. This 26-week study, known as HARMONY, is preceded by open-label psychotherapy to ensure that study participants truly need pharmacotherapy. Patients are eligible regardless of their type of dementia, because psychosis in patients with various forms of dementia is clinically pretty much the same, whether the underlying disorder is Alzheimer’s disease, vascular dementia, Parkinson’s disease, or Lewy body dementia, according to Dr. Bugarski-Kirola.
In addition, pimavanserin also is the subject of an ongoing phase 3 randomized trial in patients with major depressive disorder inadequately responsive to an selective serotonin reuptake inhibitor or a selective norepinephrine reuptake inhibitor. A 380-patient phase 2 study of the drug as adjunctive treatment for negative symptoms of schizophrenia also is underway based upon earlier promising results.
Across the board for these potential indications, the drug has been well tolerated, with a side effect profile similar to that of placebo. Importantly, pimavanserin has not been associated with cognitive impairment when used for dementia-related psychosis, unlike the antipsychotics now being used off label in clinical practice, the psychiatrist said.
SEP-363856 part of ‘novel class’
SEP-363856 is a nondopaminergic D2, trace amine-associated receptor agonist (TAAR1) under development for treatment of schizophrenia. Phase 3 trials in adults and adolescents with schizophrenia will begin before the end of the year on the strength of positive phase 2 results, according to Kenneth S. Koblan, PhD, head of global translational medicine and early development, as well as head of discovery sciences, at Sunovion Pharmaceuticals, Marlborough, Mass.
“We believe that SEP-363856 actually represents the first candidate in a novel class of antipsychotics. It’s a monoamine receptor activator, unlike the atypical antipsychotics, which work through blockade of the monoamine receptor via dopamine and serotonin. We believe that it’s the monoamine receptor activation that leads to the safety and efficacy of the class,” he explained.
In the four-country, double-blind, 4-week phase 2 trial conducted in 245 hospitalized acutely psychotic patients, oral SEP-363856 flexibly dosed at 50 or 75 mg/day had a side effect profile like that of placebo. Negative symptoms as assessed via the Brief Negative Symptom Scale improved by an average of 7.1 points at 4 weeks with SEP-363856, significantly more than the 2.7-point improvement with placebo. The PANSS total score improved by 17.2 points in the SEP-363856 group and 9.7 points in controls at 4 weeks, with a further 10-point drop in PANSS during a 6-month open-label extension phase of the study. Moreover, the SEP-363856 cohort showed significant functional improvement at 4 weeks in the UCSD Performance-Based Skills Assessment, with continued improvement during the open-label extension study.
Dr. Koblan said the pharmaceutical industry has overemphasized the development of dopaminergic D2-based drugs for schizophrenia. In the past 2 decades, roughly 30,000 patients have been enrolled in industry-sponsored, placebo-controlled, phase 2 or 3 randomized trials of drugs with that mechanism. Many of the those drugs have reached the marketplace. In contrast, there have been far fewer RCTs – and no product launches – of antipsychotics with non-D2 mechanisms of action.
“When you consider that the cost is about $50,000 per research subject and 50,000 subjects have been studied since 2000, the pharmaceutical industry has invested on the order of billions of dollars to try to come up with the next breakthrough medication,” he said.
COPENHAGEN –
Two of the highlighted agents – pimavanserin and SEP-363856 – were designed to eschew the traditional antipsychotic target, the dopamine D2 receptor, in favor of other mechanisms of action aimed at the negative symptoms of schizophrenia, for which there is a long-recognized major unmet need for better therapies.
A third agent, known for now as ALKS 3831, is composed of a combination of olanzapine and samidorphan, an opioid receptor antagonist. This once-daily oral combination of olanzapine/samidorphan (OLA/SAM) is designed to retain the clinical efficacy of olanzapine while mitigating the drug’s limiting side effect of substantial weight gain.
OLA/SAM New Drug Application expected soon
Christine Graham, PhD, presented highlights of the pivotal phase 3 ENLIGHTEN-2 study, a double-blind clinical trial in which 661 U.S. outpatients with schizophrenia were randomized to OLA/SAM or olanzapine alone at 10 or 20 mg/day for 24 weeks, at which point everyone was switched to open-label OLA/SAM at 10 or 20 mg/10 mg for an additional 52-week extension safety study.
At 24 weeks, the OLA/SAM group had a mean 4.21% weight gain from baseline, significantly less than the 6.59% gain with olanzapine alone. A clinically meaningful and unwelcome weight gain of 7% or greater occurred in 27.5% of OLA/SAM patients, compared with 42.7% of controls, for an adjusted 50% reduction in risk in the group on the investigational medication. Similarly, a 10% or greater weight gain occurred in 17.8% of OLA/SAM patients and 29.8% of controls; once again, that represented a 50% relative risk reduction. The two therapies were equally effective, achieving roughly 10-point reductions in the Positive and Negative Syndrome Scale (PANSS) for schizophrenia total score.
Both treatments showed similar weight gain trajectories for the first 4 weeks. However, by week 6 the trajectories diverged, with body weight plateauing in the OLA/SAM group and remaining stable throughout the remainder of the 76-week, two-part study. Meanwhile, body weight continued to climb in the olanzapine-only group throughout the 24 weeks, reported Dr. Graham, senior clinical research scientist at Alkermes, in Waltham, Mass.
“The waist circumference results were surprising: We saw that waist circumference separated between the two groups as early as week 1, considerably earlier than the week 6 separation in weight. This suggests to us that even when weight gain is similar between the two treatments, OLA/SAM is showing an early effect at limiting central fat accumulation – and this has important health implications, as central fat has been shown to be potentially pathogenic for developing diabetes, cardiovascular disease, and even some forms of cancer,” she said.
The safety profile of OLA/SAM was essentially the same as for olanzapine-only, with the exception of the weight gain.
Alkermes is planning to submit its New Drug Application for OLA/SAM to the Food and Drug Administration before the year’s end. FDA officials have urged the company to broaden the application to include not only the treatment of schizophrenia, but bipolar I disorder as well, since olanzapine is an approved, well-established treatment for that disorder. Dr. Graham and coinvestigators have demonstrated that OLA/SAM has no clinically significant effect on the pharmacokinetics of lithium or valproate (Clin Drug Investig. 2019 Oct 4. doi: 10.1007/s40261-019-00860-y).
Phase 3 trial on pimavanserin underway
Pimavanserin is an oral selective serotonin inverse agonist, or SSIA, with a high affinity for 5-HT2A receptors, very low affinity for 5-HT2C receptors, and “absolutely no affinity” for dopaminergic, histaminergic, adrenergic, or muscarinic receptors, explained Dragana Bugarski-Kirola, MD, a psychiatrist and vice president of clinical development at Acadia Pharmaceuticals in San Diego.
“Those sites are thought to contribute to sedation, cognitive impairment, and orthostatic hypotension,” she noted.
Pimavanserin is at present FDA approved for a narrow indication: Treatment of hallucinations and delusions associated with Parkinson’s disease psychosis. But the drug’s unique mechanism of action suggests broad efficacy across a range of psychiatric disorders.
Indeed, after a successful phase 2 clinical trial of pimavanserin for treatment of Alzheimer’s-related psychosis, a phase 3 randomized, double-blind, placebo-controlled clinical trial of the drug for relapse prevention in dementia-related psychosis is now enrolling a planned 360 outpatients at 95 centers in 13 countries. This 26-week study, known as HARMONY, is preceded by open-label psychotherapy to ensure that study participants truly need pharmacotherapy. Patients are eligible regardless of their type of dementia, because psychosis in patients with various forms of dementia is clinically pretty much the same, whether the underlying disorder is Alzheimer’s disease, vascular dementia, Parkinson’s disease, or Lewy body dementia, according to Dr. Bugarski-Kirola.
In addition, pimavanserin also is the subject of an ongoing phase 3 randomized trial in patients with major depressive disorder inadequately responsive to an selective serotonin reuptake inhibitor or a selective norepinephrine reuptake inhibitor. A 380-patient phase 2 study of the drug as adjunctive treatment for negative symptoms of schizophrenia also is underway based upon earlier promising results.
Across the board for these potential indications, the drug has been well tolerated, with a side effect profile similar to that of placebo. Importantly, pimavanserin has not been associated with cognitive impairment when used for dementia-related psychosis, unlike the antipsychotics now being used off label in clinical practice, the psychiatrist said.
SEP-363856 part of ‘novel class’
SEP-363856 is a nondopaminergic D2, trace amine-associated receptor agonist (TAAR1) under development for treatment of schizophrenia. Phase 3 trials in adults and adolescents with schizophrenia will begin before the end of the year on the strength of positive phase 2 results, according to Kenneth S. Koblan, PhD, head of global translational medicine and early development, as well as head of discovery sciences, at Sunovion Pharmaceuticals, Marlborough, Mass.
“We believe that SEP-363856 actually represents the first candidate in a novel class of antipsychotics. It’s a monoamine receptor activator, unlike the atypical antipsychotics, which work through blockade of the monoamine receptor via dopamine and serotonin. We believe that it’s the monoamine receptor activation that leads to the safety and efficacy of the class,” he explained.
In the four-country, double-blind, 4-week phase 2 trial conducted in 245 hospitalized acutely psychotic patients, oral SEP-363856 flexibly dosed at 50 or 75 mg/day had a side effect profile like that of placebo. Negative symptoms as assessed via the Brief Negative Symptom Scale improved by an average of 7.1 points at 4 weeks with SEP-363856, significantly more than the 2.7-point improvement with placebo. The PANSS total score improved by 17.2 points in the SEP-363856 group and 9.7 points in controls at 4 weeks, with a further 10-point drop in PANSS during a 6-month open-label extension phase of the study. Moreover, the SEP-363856 cohort showed significant functional improvement at 4 weeks in the UCSD Performance-Based Skills Assessment, with continued improvement during the open-label extension study.
Dr. Koblan said the pharmaceutical industry has overemphasized the development of dopaminergic D2-based drugs for schizophrenia. In the past 2 decades, roughly 30,000 patients have been enrolled in industry-sponsored, placebo-controlled, phase 2 or 3 randomized trials of drugs with that mechanism. Many of the those drugs have reached the marketplace. In contrast, there have been far fewer RCTs – and no product launches – of antipsychotics with non-D2 mechanisms of action.
“When you consider that the cost is about $50,000 per research subject and 50,000 subjects have been studied since 2000, the pharmaceutical industry has invested on the order of billions of dollars to try to come up with the next breakthrough medication,” he said.
COPENHAGEN –
Two of the highlighted agents – pimavanserin and SEP-363856 – were designed to eschew the traditional antipsychotic target, the dopamine D2 receptor, in favor of other mechanisms of action aimed at the negative symptoms of schizophrenia, for which there is a long-recognized major unmet need for better therapies.
A third agent, known for now as ALKS 3831, is composed of a combination of olanzapine and samidorphan, an opioid receptor antagonist. This once-daily oral combination of olanzapine/samidorphan (OLA/SAM) is designed to retain the clinical efficacy of olanzapine while mitigating the drug’s limiting side effect of substantial weight gain.
OLA/SAM New Drug Application expected soon
Christine Graham, PhD, presented highlights of the pivotal phase 3 ENLIGHTEN-2 study, a double-blind clinical trial in which 661 U.S. outpatients with schizophrenia were randomized to OLA/SAM or olanzapine alone at 10 or 20 mg/day for 24 weeks, at which point everyone was switched to open-label OLA/SAM at 10 or 20 mg/10 mg for an additional 52-week extension safety study.
At 24 weeks, the OLA/SAM group had a mean 4.21% weight gain from baseline, significantly less than the 6.59% gain with olanzapine alone. A clinically meaningful and unwelcome weight gain of 7% or greater occurred in 27.5% of OLA/SAM patients, compared with 42.7% of controls, for an adjusted 50% reduction in risk in the group on the investigational medication. Similarly, a 10% or greater weight gain occurred in 17.8% of OLA/SAM patients and 29.8% of controls; once again, that represented a 50% relative risk reduction. The two therapies were equally effective, achieving roughly 10-point reductions in the Positive and Negative Syndrome Scale (PANSS) for schizophrenia total score.
Both treatments showed similar weight gain trajectories for the first 4 weeks. However, by week 6 the trajectories diverged, with body weight plateauing in the OLA/SAM group and remaining stable throughout the remainder of the 76-week, two-part study. Meanwhile, body weight continued to climb in the olanzapine-only group throughout the 24 weeks, reported Dr. Graham, senior clinical research scientist at Alkermes, in Waltham, Mass.
“The waist circumference results were surprising: We saw that waist circumference separated between the two groups as early as week 1, considerably earlier than the week 6 separation in weight. This suggests to us that even when weight gain is similar between the two treatments, OLA/SAM is showing an early effect at limiting central fat accumulation – and this has important health implications, as central fat has been shown to be potentially pathogenic for developing diabetes, cardiovascular disease, and even some forms of cancer,” she said.
The safety profile of OLA/SAM was essentially the same as for olanzapine-only, with the exception of the weight gain.
Alkermes is planning to submit its New Drug Application for OLA/SAM to the Food and Drug Administration before the year’s end. FDA officials have urged the company to broaden the application to include not only the treatment of schizophrenia, but bipolar I disorder as well, since olanzapine is an approved, well-established treatment for that disorder. Dr. Graham and coinvestigators have demonstrated that OLA/SAM has no clinically significant effect on the pharmacokinetics of lithium or valproate (Clin Drug Investig. 2019 Oct 4. doi: 10.1007/s40261-019-00860-y).
Phase 3 trial on pimavanserin underway
Pimavanserin is an oral selective serotonin inverse agonist, or SSIA, with a high affinity for 5-HT2A receptors, very low affinity for 5-HT2C receptors, and “absolutely no affinity” for dopaminergic, histaminergic, adrenergic, or muscarinic receptors, explained Dragana Bugarski-Kirola, MD, a psychiatrist and vice president of clinical development at Acadia Pharmaceuticals in San Diego.
“Those sites are thought to contribute to sedation, cognitive impairment, and orthostatic hypotension,” she noted.
Pimavanserin is at present FDA approved for a narrow indication: Treatment of hallucinations and delusions associated with Parkinson’s disease psychosis. But the drug’s unique mechanism of action suggests broad efficacy across a range of psychiatric disorders.
Indeed, after a successful phase 2 clinical trial of pimavanserin for treatment of Alzheimer’s-related psychosis, a phase 3 randomized, double-blind, placebo-controlled clinical trial of the drug for relapse prevention in dementia-related psychosis is now enrolling a planned 360 outpatients at 95 centers in 13 countries. This 26-week study, known as HARMONY, is preceded by open-label psychotherapy to ensure that study participants truly need pharmacotherapy. Patients are eligible regardless of their type of dementia, because psychosis in patients with various forms of dementia is clinically pretty much the same, whether the underlying disorder is Alzheimer’s disease, vascular dementia, Parkinson’s disease, or Lewy body dementia, according to Dr. Bugarski-Kirola.
In addition, pimavanserin also is the subject of an ongoing phase 3 randomized trial in patients with major depressive disorder inadequately responsive to an selective serotonin reuptake inhibitor or a selective norepinephrine reuptake inhibitor. A 380-patient phase 2 study of the drug as adjunctive treatment for negative symptoms of schizophrenia also is underway based upon earlier promising results.
Across the board for these potential indications, the drug has been well tolerated, with a side effect profile similar to that of placebo. Importantly, pimavanserin has not been associated with cognitive impairment when used for dementia-related psychosis, unlike the antipsychotics now being used off label in clinical practice, the psychiatrist said.
SEP-363856 part of ‘novel class’
SEP-363856 is a nondopaminergic D2, trace amine-associated receptor agonist (TAAR1) under development for treatment of schizophrenia. Phase 3 trials in adults and adolescents with schizophrenia will begin before the end of the year on the strength of positive phase 2 results, according to Kenneth S. Koblan, PhD, head of global translational medicine and early development, as well as head of discovery sciences, at Sunovion Pharmaceuticals, Marlborough, Mass.
“We believe that SEP-363856 actually represents the first candidate in a novel class of antipsychotics. It’s a monoamine receptor activator, unlike the atypical antipsychotics, which work through blockade of the monoamine receptor via dopamine and serotonin. We believe that it’s the monoamine receptor activation that leads to the safety and efficacy of the class,” he explained.
In the four-country, double-blind, 4-week phase 2 trial conducted in 245 hospitalized acutely psychotic patients, oral SEP-363856 flexibly dosed at 50 or 75 mg/day had a side effect profile like that of placebo. Negative symptoms as assessed via the Brief Negative Symptom Scale improved by an average of 7.1 points at 4 weeks with SEP-363856, significantly more than the 2.7-point improvement with placebo. The PANSS total score improved by 17.2 points in the SEP-363856 group and 9.7 points in controls at 4 weeks, with a further 10-point drop in PANSS during a 6-month open-label extension phase of the study. Moreover, the SEP-363856 cohort showed significant functional improvement at 4 weeks in the UCSD Performance-Based Skills Assessment, with continued improvement during the open-label extension study.
Dr. Koblan said the pharmaceutical industry has overemphasized the development of dopaminergic D2-based drugs for schizophrenia. In the past 2 decades, roughly 30,000 patients have been enrolled in industry-sponsored, placebo-controlled, phase 2 or 3 randomized trials of drugs with that mechanism. Many of the those drugs have reached the marketplace. In contrast, there have been far fewer RCTs – and no product launches – of antipsychotics with non-D2 mechanisms of action.
“When you consider that the cost is about $50,000 per research subject and 50,000 subjects have been studied since 2000, the pharmaceutical industry has invested on the order of billions of dollars to try to come up with the next breakthrough medication,” he said.
REPORTING FROM ECNP 2019
Will changing the names of psychiatric medications lead to better treatment?
Back in 1980, the American Psychiatric Association dropped the word “neurosis” from the DSM-III, so that if you had been neurotic, after 1980, you were neurotic no longer.
At the time, I discussed this on my daily radio show. For those folks who were nervous, worried, fearful, and full of anxieties about themselves, their families, welfare, health, and the environment around them, a new set of labels was introduced to more specifically describe one or more problems related to anxiety.
For codification, and at times, a clearer understanding of a specific problem, the change was made to be helpful. Certainly, for insurers and pharmacologic treatments, it worked. However, it’s interesting that the word and concept, neurosis, which still is used by some psychiatrists and psychologists – although not scientific – does offer a clear overall picture of a suffering, anxiety-ridden person who might have a combination of an anxiety disorder, panic attacks, somatic symptoms, and endless worry. This overlapping picture often is seen in clinical practice more than the multiple one-dimensional labels that are currently used. So be it.
This all leads me to what I’ve recently learned about the Neuroscience-based Nomenclature (NbN) Project. According to a recent article in the APA’s Psychiatric News, the group’s board of trustees has endorsed a proposal that would change or revise the names of psychiatric medications so that the names reflect their mechanism of action – a move seemingly focused on a pure biological model.
For example, according to the article, the medication perphenazine would be renamed a “D2 receptor antagonist” rather than an antipsychotic. For depression, we might have a serotonergic reuptake inhibitor, according to the report, and of course, the list of changes would go on – based on current knowledge of biological activity. It’s true that in general medicine, there are examples where mode of action is discussed. For example, in cardiology we have beta-blockers and alpha-blockers, which are descriptive of their actions. As doctors who have trained for years and know the mechanism of action of various medications, we will understand all this. But in patient care, both doctors and their patients often understand and feel comfortable using descriptive terms indicating the treatment modality, such as antibiotics, antivirals, antifungals, anti-inflammatory medications, as well as anti-itching, antiaging, and antispasmodic drugs.
So, I am concerned about these proposed changes. In an era focused on patient-centered care, where we seek to make it simpler for the patient/health care consumer, we might make it harder for the patient to grasp what’s going on.
It’s very important to keep in mind that we as physicians know the ins and outs of medications, and that even the most educated and bright patients who are not in medicine do not know what our education has taught us. For example, regardless of specialty, we all know the difference between left-sided and right-sided heart failure. Those outside of medicine, however, rarely know the difference. They understand heart disease as a rule. People in general might understand some general concepts, such as RBC, WBC, and platelets. A patient will speak of taking a blood thinner but rarely know or understand the differences between antiplatelets and anticoagulants. And why should they know this?
The point here is that I believe good patient care is keeping it simple and taking the time to explain what’s being treated, aiming to inform patients using down-to-earth, accessible language rather than the language of biochemistry.
It’s true that in psychiatry, wider use of certain medications than originally indicated has grown tremendously as well as off-label use. In light of that, the NbN idea is laudable. However, it would seem more practical to leave the traditional modes of action in place and expand our discussions with patients as to why we are using a specific medication. I have found a very simple and even rewarding way to explain to patients, for example, that yes, this is an antiseizure medication but it is now used in psychiatry as a mood stabilizer.
Another important point is the question of whether using nomenclature that describes the exact location of the problem is all that accurate. Currently, we know we still have a lot to learn about brain chemistry and neuronal transmission in mental disorders, just as in many medical disorders, there are gaps in our understanding of many illnesses and subsequent molecular changes.
Just as the DSM-III left behind the all-encompassing and descriptive word neurosis and the APA has changed labels in the DSM-IV and DSM-5, so the NbN project would change the nomenclature of current psychotropic medications. The intentions are good, but the idea that those changes will foster better patient understanding defies common sense. A better idea might be to continue use of both scientific names and names of commonly used actions of the medications, leaving both in place and letting clinicians decide what nomenclature best suits each patient.
It will be a sad day when psychiatrists become so medically and “scientifically” driven that we cannot explain to a patient, “I’m prescribing this antidepressant because it’s now used to treat anxiety,” or “Yes, this medicine is labeled ‘antipsychotic,’ but you’re not psychotic. It may help your mood swings and may even help you sleep better.” Now, is that hard? Is talking to a person and explaining the treatment no longer part of care? The take-home messages from the recent APA/Institute of Psychiatric Services meeting I attended seemed to suggest that human attention and care have great value. My father, a surgeon, always said that you learn a lot by simply talking to patients – and they learn from you.
Dr. London is a practicing psychiatrist and has been a newspaper columnist for 35 years, specializing in and writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019).
Back in 1980, the American Psychiatric Association dropped the word “neurosis” from the DSM-III, so that if you had been neurotic, after 1980, you were neurotic no longer.
At the time, I discussed this on my daily radio show. For those folks who were nervous, worried, fearful, and full of anxieties about themselves, their families, welfare, health, and the environment around them, a new set of labels was introduced to more specifically describe one or more problems related to anxiety.
For codification, and at times, a clearer understanding of a specific problem, the change was made to be helpful. Certainly, for insurers and pharmacologic treatments, it worked. However, it’s interesting that the word and concept, neurosis, which still is used by some psychiatrists and psychologists – although not scientific – does offer a clear overall picture of a suffering, anxiety-ridden person who might have a combination of an anxiety disorder, panic attacks, somatic symptoms, and endless worry. This overlapping picture often is seen in clinical practice more than the multiple one-dimensional labels that are currently used. So be it.
This all leads me to what I’ve recently learned about the Neuroscience-based Nomenclature (NbN) Project. According to a recent article in the APA’s Psychiatric News, the group’s board of trustees has endorsed a proposal that would change or revise the names of psychiatric medications so that the names reflect their mechanism of action – a move seemingly focused on a pure biological model.
For example, according to the article, the medication perphenazine would be renamed a “D2 receptor antagonist” rather than an antipsychotic. For depression, we might have a serotonergic reuptake inhibitor, according to the report, and of course, the list of changes would go on – based on current knowledge of biological activity. It’s true that in general medicine, there are examples where mode of action is discussed. For example, in cardiology we have beta-blockers and alpha-blockers, which are descriptive of their actions. As doctors who have trained for years and know the mechanism of action of various medications, we will understand all this. But in patient care, both doctors and their patients often understand and feel comfortable using descriptive terms indicating the treatment modality, such as antibiotics, antivirals, antifungals, anti-inflammatory medications, as well as anti-itching, antiaging, and antispasmodic drugs.
So, I am concerned about these proposed changes. In an era focused on patient-centered care, where we seek to make it simpler for the patient/health care consumer, we might make it harder for the patient to grasp what’s going on.
It’s very important to keep in mind that we as physicians know the ins and outs of medications, and that even the most educated and bright patients who are not in medicine do not know what our education has taught us. For example, regardless of specialty, we all know the difference between left-sided and right-sided heart failure. Those outside of medicine, however, rarely know the difference. They understand heart disease as a rule. People in general might understand some general concepts, such as RBC, WBC, and platelets. A patient will speak of taking a blood thinner but rarely know or understand the differences between antiplatelets and anticoagulants. And why should they know this?
The point here is that I believe good patient care is keeping it simple and taking the time to explain what’s being treated, aiming to inform patients using down-to-earth, accessible language rather than the language of biochemistry.
It’s true that in psychiatry, wider use of certain medications than originally indicated has grown tremendously as well as off-label use. In light of that, the NbN idea is laudable. However, it would seem more practical to leave the traditional modes of action in place and expand our discussions with patients as to why we are using a specific medication. I have found a very simple and even rewarding way to explain to patients, for example, that yes, this is an antiseizure medication but it is now used in psychiatry as a mood stabilizer.
Another important point is the question of whether using nomenclature that describes the exact location of the problem is all that accurate. Currently, we know we still have a lot to learn about brain chemistry and neuronal transmission in mental disorders, just as in many medical disorders, there are gaps in our understanding of many illnesses and subsequent molecular changes.
Just as the DSM-III left behind the all-encompassing and descriptive word neurosis and the APA has changed labels in the DSM-IV and DSM-5, so the NbN project would change the nomenclature of current psychotropic medications. The intentions are good, but the idea that those changes will foster better patient understanding defies common sense. A better idea might be to continue use of both scientific names and names of commonly used actions of the medications, leaving both in place and letting clinicians decide what nomenclature best suits each patient.
It will be a sad day when psychiatrists become so medically and “scientifically” driven that we cannot explain to a patient, “I’m prescribing this antidepressant because it’s now used to treat anxiety,” or “Yes, this medicine is labeled ‘antipsychotic,’ but you’re not psychotic. It may help your mood swings and may even help you sleep better.” Now, is that hard? Is talking to a person and explaining the treatment no longer part of care? The take-home messages from the recent APA/Institute of Psychiatric Services meeting I attended seemed to suggest that human attention and care have great value. My father, a surgeon, always said that you learn a lot by simply talking to patients – and they learn from you.
Dr. London is a practicing psychiatrist and has been a newspaper columnist for 35 years, specializing in and writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019).
Back in 1980, the American Psychiatric Association dropped the word “neurosis” from the DSM-III, so that if you had been neurotic, after 1980, you were neurotic no longer.
At the time, I discussed this on my daily radio show. For those folks who were nervous, worried, fearful, and full of anxieties about themselves, their families, welfare, health, and the environment around them, a new set of labels was introduced to more specifically describe one or more problems related to anxiety.
For codification, and at times, a clearer understanding of a specific problem, the change was made to be helpful. Certainly, for insurers and pharmacologic treatments, it worked. However, it’s interesting that the word and concept, neurosis, which still is used by some psychiatrists and psychologists – although not scientific – does offer a clear overall picture of a suffering, anxiety-ridden person who might have a combination of an anxiety disorder, panic attacks, somatic symptoms, and endless worry. This overlapping picture often is seen in clinical practice more than the multiple one-dimensional labels that are currently used. So be it.
This all leads me to what I’ve recently learned about the Neuroscience-based Nomenclature (NbN) Project. According to a recent article in the APA’s Psychiatric News, the group’s board of trustees has endorsed a proposal that would change or revise the names of psychiatric medications so that the names reflect their mechanism of action – a move seemingly focused on a pure biological model.
For example, according to the article, the medication perphenazine would be renamed a “D2 receptor antagonist” rather than an antipsychotic. For depression, we might have a serotonergic reuptake inhibitor, according to the report, and of course, the list of changes would go on – based on current knowledge of biological activity. It’s true that in general medicine, there are examples where mode of action is discussed. For example, in cardiology we have beta-blockers and alpha-blockers, which are descriptive of their actions. As doctors who have trained for years and know the mechanism of action of various medications, we will understand all this. But in patient care, both doctors and their patients often understand and feel comfortable using descriptive terms indicating the treatment modality, such as antibiotics, antivirals, antifungals, anti-inflammatory medications, as well as anti-itching, antiaging, and antispasmodic drugs.
So, I am concerned about these proposed changes. In an era focused on patient-centered care, where we seek to make it simpler for the patient/health care consumer, we might make it harder for the patient to grasp what’s going on.
It’s very important to keep in mind that we as physicians know the ins and outs of medications, and that even the most educated and bright patients who are not in medicine do not know what our education has taught us. For example, regardless of specialty, we all know the difference between left-sided and right-sided heart failure. Those outside of medicine, however, rarely know the difference. They understand heart disease as a rule. People in general might understand some general concepts, such as RBC, WBC, and platelets. A patient will speak of taking a blood thinner but rarely know or understand the differences between antiplatelets and anticoagulants. And why should they know this?
The point here is that I believe good patient care is keeping it simple and taking the time to explain what’s being treated, aiming to inform patients using down-to-earth, accessible language rather than the language of biochemistry.
It’s true that in psychiatry, wider use of certain medications than originally indicated has grown tremendously as well as off-label use. In light of that, the NbN idea is laudable. However, it would seem more practical to leave the traditional modes of action in place and expand our discussions with patients as to why we are using a specific medication. I have found a very simple and even rewarding way to explain to patients, for example, that yes, this is an antiseizure medication but it is now used in psychiatry as a mood stabilizer.
Another important point is the question of whether using nomenclature that describes the exact location of the problem is all that accurate. Currently, we know we still have a lot to learn about brain chemistry and neuronal transmission in mental disorders, just as in many medical disorders, there are gaps in our understanding of many illnesses and subsequent molecular changes.
Just as the DSM-III left behind the all-encompassing and descriptive word neurosis and the APA has changed labels in the DSM-IV and DSM-5, so the NbN project would change the nomenclature of current psychotropic medications. The intentions are good, but the idea that those changes will foster better patient understanding defies common sense. A better idea might be to continue use of both scientific names and names of commonly used actions of the medications, leaving both in place and letting clinicians decide what nomenclature best suits each patient.
It will be a sad day when psychiatrists become so medically and “scientifically” driven that we cannot explain to a patient, “I’m prescribing this antidepressant because it’s now used to treat anxiety,” or “Yes, this medicine is labeled ‘antipsychotic,’ but you’re not psychotic. It may help your mood swings and may even help you sleep better.” Now, is that hard? Is talking to a person and explaining the treatment no longer part of care? The take-home messages from the recent APA/Institute of Psychiatric Services meeting I attended seemed to suggest that human attention and care have great value. My father, a surgeon, always said that you learn a lot by simply talking to patients – and they learn from you.
Dr. London is a practicing psychiatrist and has been a newspaper columnist for 35 years, specializing in and writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019).