User login
Diffuse idiopathic skeletal hyperostosis heart risk higher than expected
More people with diffuse idiopathic skeletal hyperostosis (DISH) develop cardiovascular disease (CVD) than is predicted by the Framingham Risk Score, results of an observational study have shown.
Notably, a higher rate of myocardial infarction (MI) was seen in those with DISH than in those without DISH over the 10-year follow-up period (24.4% vs. 4.3%; P = .0055).
“We propose more scrutiny is warranted in evaluating CV risk in these patients, more demanding treatment target goals should be established, and as a result, earlier and more aggressive preventive medical interventions instituted,” corresponding author Reuven Mader, MD, and associates wrote in Arthritis Research & Therapy.
“What Mader’s study is pointing out is that it’s worth the radiologist reporting [DISH],” Elizabeth A. Regan, MD, PhD, from the National Jewish Health Center in Denver, said in an interview.
DISH on a chest x-ray or CT scan should be another “red flag to be even more attentive to cardiovascular risk,” she added, particularly because studies have shown that people with DISH tend to be obese, have metabolic syndrome, or diabetes – all of which independently increase their risk for cardiovascular disease.
An old condition often found by accident
Physicians have known about DISH for many years, Dr. Mader of Ha’Emek Medical Center in Afula, Israel, observed in an interview. Historical evidence suggests it was present more than a thousand years ago, but it wasn’t until the 1950s that it gained scientific interest. Originally coined Forestier’s disease, it was renamed DISH in the late 1960s following the realization that it was not limited to the spine.
“It is a condition which is characterized by new bone formation,” Dr. Mader explained. This new bone formation has some predilection for the entheses – the tendons, ligaments, or joint capsules, that attach to the bone.
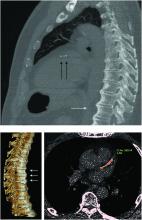
“Diagnosis of the disease is based mainly on radiographs, especially of the thoracic spine, and it requires the formation of bridges that connect at least four contiguous vertebra,” he continued.
“The bridges are usually right-sided and usually the intervertebral spaces are spared. Classically there is no involvement of the sacroiliac joints, although there are some changes that might involve the sacroiliac joints but in a different manner than in inflammatory sacroiliitis.”
DISH was originally thought to be a pain syndrome, which has “not played out,” Dr. Regan noted in her interview. While there may be people who experience pain as a result of DISH, most cases are asymptomatic and usually picked up incidentally on a chest x-ray or CT scan.
“It’s something that’s not obvious,” she said. One of the main problems it can cause is stiffness and lack of mobility in the spine and this can lead to quite severe fractures in some cases, such as during a car accident. Hence spinal surgeons and other orthopedic specialists, such as Dr. Regan, have also taken an interest in the condition.
“Apart from the thoracic spine, DISH may also involve the cervical spine; there have been many reports about difficulty in swallowing, breathing, and in the lumbar spine, spinal stenosis and so forth,” Dr. Mader said. The differential diagnosis includes ankylosing spondylitis, although there is some evidence that the two can coexist.
“The diagnosis depends on the alertness of the examining physician,” he added, noting that rheumatologists and other specialists would be “very aware of this condition” and “sensitive to changes that we see when we examine these patients.”
DISH and heightened cardiovascular risk
Previous work by Dr. Mader and associates has shown that people with DISH are more often affected by the metabolic syndrome than are those without DISH. The cross-sectional study had excluded those with preexisting CVD and found that people with DISH had a significantly higher Framingham Risk Score, compared with a control group of people with osteoarthritis and no DISH (P = .004), which in turn meant they had a significantly (P = .007) higher 10-year risk for developing CVD.
The aim of their most recent study was to compare the actual rate of CV events in 2016 versus those predicted by the Framingham Risk Score in 2006. To do this, they compared the available electronic medical records of 45 individuals with DISH and 47 without it.
The results showed that almost 39% of people with DISH had developed CVD, whereas the Framingham Risk Score had estimated that just under 27% would develop CVD.
For every 1% increase in the CVD risk calculated by the Framingham Risk Score, the odds of CVD increased by 4% in the DISH group versus the control group (P = .02).
While there was a significant (P < .003) difference in the Framingham Risk Score between the DISH and control groups in 2006 (28.6% vs. 17.8%), there was no overall statistical difference (P = .2) in the composite CVD outcome (38.8% vs. 25.5%) 10 years later, as calculated by the revised Framingham Risk Score, which included MI, cerebrovascular accident, transient ischemic attack, peripheral artery disease, and heart failure with preserved ejection fraction.
“We are dealing with patients who are in their 70s. So, it is expected that this group of patients will be more often affected by cardiovascular disease” than younger individuals, Dr. Mader observed. That said, the study’s findings “confirm the theory that patients with DISH have a high likelihood of developing cardiovascular disease,” he added, acknowledging that it was only the risk for MI that was statistically significantly higher in people with DISH than in the controls.
DISH and coronary artery calcification
“It might be even more interesting to have a different control population that had no osteoarthritis,” Dr. Regan observed.
As the associate director of the COPDGene study, Dr. Regan has access to data collected from a large cohort of people with chronic obstructive pulmonary disease (COPD; n = 2,728), around 13% of whom were identified as having DISH in one recent study.
In that study, the presence of DISH versus no DISH was associated with a 37% higher risk for having coronary artery calcification (CAC) – a marker for atherosclerosis and cardiovascular disease. Two-thirds of people with DISH had CAC, compared with 46.9% of those without DISH (P < .001). The prevalence of DISH was 8.8% in those without CAC, 12.8% in those with a CAC score of 1-100, 20% in those with a CAC score of 100-400, and 24.7% in those with a CAC score of more than 400, which is associated with a very high risk for coronary artery disease.
Dr. Regan observed that information on heart attacks and strokes were collected within the COPDGene study, so it would be possible to look at cardiovascular risk in their patients with DISH and confirm the findings of Mader and colleagues.
“I think the most important thing is recognizing that there are things going on in the spine that are important to people’s general health,” Dr. Regan said.
Dr. Mader noted: “It makes sense that patients with DISH should be more meticulously followed for at least the traditional risk factors and better treated because they are at a higher risk for these events.”
The study received no financial support. Neither Dr. Mader nor Dr. Regan had any conflicts of interest to disclose.
SOURCE: Glick K et al. Arthritis Res Ther. 2020. doi: 10.1186/s13075-020-02278-w.
More people with diffuse idiopathic skeletal hyperostosis (DISH) develop cardiovascular disease (CVD) than is predicted by the Framingham Risk Score, results of an observational study have shown.
Notably, a higher rate of myocardial infarction (MI) was seen in those with DISH than in those without DISH over the 10-year follow-up period (24.4% vs. 4.3%; P = .0055).
“We propose more scrutiny is warranted in evaluating CV risk in these patients, more demanding treatment target goals should be established, and as a result, earlier and more aggressive preventive medical interventions instituted,” corresponding author Reuven Mader, MD, and associates wrote in Arthritis Research & Therapy.
“What Mader’s study is pointing out is that it’s worth the radiologist reporting [DISH],” Elizabeth A. Regan, MD, PhD, from the National Jewish Health Center in Denver, said in an interview.
DISH on a chest x-ray or CT scan should be another “red flag to be even more attentive to cardiovascular risk,” she added, particularly because studies have shown that people with DISH tend to be obese, have metabolic syndrome, or diabetes – all of which independently increase their risk for cardiovascular disease.
An old condition often found by accident
Physicians have known about DISH for many years, Dr. Mader of Ha’Emek Medical Center in Afula, Israel, observed in an interview. Historical evidence suggests it was present more than a thousand years ago, but it wasn’t until the 1950s that it gained scientific interest. Originally coined Forestier’s disease, it was renamed DISH in the late 1960s following the realization that it was not limited to the spine.
“It is a condition which is characterized by new bone formation,” Dr. Mader explained. This new bone formation has some predilection for the entheses – the tendons, ligaments, or joint capsules, that attach to the bone.
“Diagnosis of the disease is based mainly on radiographs, especially of the thoracic spine, and it requires the formation of bridges that connect at least four contiguous vertebra,” he continued.
“The bridges are usually right-sided and usually the intervertebral spaces are spared. Classically there is no involvement of the sacroiliac joints, although there are some changes that might involve the sacroiliac joints but in a different manner than in inflammatory sacroiliitis.”
DISH was originally thought to be a pain syndrome, which has “not played out,” Dr. Regan noted in her interview. While there may be people who experience pain as a result of DISH, most cases are asymptomatic and usually picked up incidentally on a chest x-ray or CT scan.
“It’s something that’s not obvious,” she said. One of the main problems it can cause is stiffness and lack of mobility in the spine and this can lead to quite severe fractures in some cases, such as during a car accident. Hence spinal surgeons and other orthopedic specialists, such as Dr. Regan, have also taken an interest in the condition.
“Apart from the thoracic spine, DISH may also involve the cervical spine; there have been many reports about difficulty in swallowing, breathing, and in the lumbar spine, spinal stenosis and so forth,” Dr. Mader said. The differential diagnosis includes ankylosing spondylitis, although there is some evidence that the two can coexist.
“The diagnosis depends on the alertness of the examining physician,” he added, noting that rheumatologists and other specialists would be “very aware of this condition” and “sensitive to changes that we see when we examine these patients.”
DISH and heightened cardiovascular risk
Previous work by Dr. Mader and associates has shown that people with DISH are more often affected by the metabolic syndrome than are those without DISH. The cross-sectional study had excluded those with preexisting CVD and found that people with DISH had a significantly higher Framingham Risk Score, compared with a control group of people with osteoarthritis and no DISH (P = .004), which in turn meant they had a significantly (P = .007) higher 10-year risk for developing CVD.
The aim of their most recent study was to compare the actual rate of CV events in 2016 versus those predicted by the Framingham Risk Score in 2006. To do this, they compared the available electronic medical records of 45 individuals with DISH and 47 without it.
The results showed that almost 39% of people with DISH had developed CVD, whereas the Framingham Risk Score had estimated that just under 27% would develop CVD.
For every 1% increase in the CVD risk calculated by the Framingham Risk Score, the odds of CVD increased by 4% in the DISH group versus the control group (P = .02).
While there was a significant (P < .003) difference in the Framingham Risk Score between the DISH and control groups in 2006 (28.6% vs. 17.8%), there was no overall statistical difference (P = .2) in the composite CVD outcome (38.8% vs. 25.5%) 10 years later, as calculated by the revised Framingham Risk Score, which included MI, cerebrovascular accident, transient ischemic attack, peripheral artery disease, and heart failure with preserved ejection fraction.
“We are dealing with patients who are in their 70s. So, it is expected that this group of patients will be more often affected by cardiovascular disease” than younger individuals, Dr. Mader observed. That said, the study’s findings “confirm the theory that patients with DISH have a high likelihood of developing cardiovascular disease,” he added, acknowledging that it was only the risk for MI that was statistically significantly higher in people with DISH than in the controls.
DISH and coronary artery calcification
“It might be even more interesting to have a different control population that had no osteoarthritis,” Dr. Regan observed.
As the associate director of the COPDGene study, Dr. Regan has access to data collected from a large cohort of people with chronic obstructive pulmonary disease (COPD; n = 2,728), around 13% of whom were identified as having DISH in one recent study.
In that study, the presence of DISH versus no DISH was associated with a 37% higher risk for having coronary artery calcification (CAC) – a marker for atherosclerosis and cardiovascular disease. Two-thirds of people with DISH had CAC, compared with 46.9% of those without DISH (P < .001). The prevalence of DISH was 8.8% in those without CAC, 12.8% in those with a CAC score of 1-100, 20% in those with a CAC score of 100-400, and 24.7% in those with a CAC score of more than 400, which is associated with a very high risk for coronary artery disease.
Dr. Regan observed that information on heart attacks and strokes were collected within the COPDGene study, so it would be possible to look at cardiovascular risk in their patients with DISH and confirm the findings of Mader and colleagues.
“I think the most important thing is recognizing that there are things going on in the spine that are important to people’s general health,” Dr. Regan said.
Dr. Mader noted: “It makes sense that patients with DISH should be more meticulously followed for at least the traditional risk factors and better treated because they are at a higher risk for these events.”
The study received no financial support. Neither Dr. Mader nor Dr. Regan had any conflicts of interest to disclose.
SOURCE: Glick K et al. Arthritis Res Ther. 2020. doi: 10.1186/s13075-020-02278-w.
More people with diffuse idiopathic skeletal hyperostosis (DISH) develop cardiovascular disease (CVD) than is predicted by the Framingham Risk Score, results of an observational study have shown.
Notably, a higher rate of myocardial infarction (MI) was seen in those with DISH than in those without DISH over the 10-year follow-up period (24.4% vs. 4.3%; P = .0055).
“We propose more scrutiny is warranted in evaluating CV risk in these patients, more demanding treatment target goals should be established, and as a result, earlier and more aggressive preventive medical interventions instituted,” corresponding author Reuven Mader, MD, and associates wrote in Arthritis Research & Therapy.
“What Mader’s study is pointing out is that it’s worth the radiologist reporting [DISH],” Elizabeth A. Regan, MD, PhD, from the National Jewish Health Center in Denver, said in an interview.
DISH on a chest x-ray or CT scan should be another “red flag to be even more attentive to cardiovascular risk,” she added, particularly because studies have shown that people with DISH tend to be obese, have metabolic syndrome, or diabetes – all of which independently increase their risk for cardiovascular disease.
An old condition often found by accident
Physicians have known about DISH for many years, Dr. Mader of Ha’Emek Medical Center in Afula, Israel, observed in an interview. Historical evidence suggests it was present more than a thousand years ago, but it wasn’t until the 1950s that it gained scientific interest. Originally coined Forestier’s disease, it was renamed DISH in the late 1960s following the realization that it was not limited to the spine.
“It is a condition which is characterized by new bone formation,” Dr. Mader explained. This new bone formation has some predilection for the entheses – the tendons, ligaments, or joint capsules, that attach to the bone.
“Diagnosis of the disease is based mainly on radiographs, especially of the thoracic spine, and it requires the formation of bridges that connect at least four contiguous vertebra,” he continued.
“The bridges are usually right-sided and usually the intervertebral spaces are spared. Classically there is no involvement of the sacroiliac joints, although there are some changes that might involve the sacroiliac joints but in a different manner than in inflammatory sacroiliitis.”
DISH was originally thought to be a pain syndrome, which has “not played out,” Dr. Regan noted in her interview. While there may be people who experience pain as a result of DISH, most cases are asymptomatic and usually picked up incidentally on a chest x-ray or CT scan.
“It’s something that’s not obvious,” she said. One of the main problems it can cause is stiffness and lack of mobility in the spine and this can lead to quite severe fractures in some cases, such as during a car accident. Hence spinal surgeons and other orthopedic specialists, such as Dr. Regan, have also taken an interest in the condition.
“Apart from the thoracic spine, DISH may also involve the cervical spine; there have been many reports about difficulty in swallowing, breathing, and in the lumbar spine, spinal stenosis and so forth,” Dr. Mader said. The differential diagnosis includes ankylosing spondylitis, although there is some evidence that the two can coexist.
“The diagnosis depends on the alertness of the examining physician,” he added, noting that rheumatologists and other specialists would be “very aware of this condition” and “sensitive to changes that we see when we examine these patients.”
DISH and heightened cardiovascular risk
Previous work by Dr. Mader and associates has shown that people with DISH are more often affected by the metabolic syndrome than are those without DISH. The cross-sectional study had excluded those with preexisting CVD and found that people with DISH had a significantly higher Framingham Risk Score, compared with a control group of people with osteoarthritis and no DISH (P = .004), which in turn meant they had a significantly (P = .007) higher 10-year risk for developing CVD.
The aim of their most recent study was to compare the actual rate of CV events in 2016 versus those predicted by the Framingham Risk Score in 2006. To do this, they compared the available electronic medical records of 45 individuals with DISH and 47 without it.
The results showed that almost 39% of people with DISH had developed CVD, whereas the Framingham Risk Score had estimated that just under 27% would develop CVD.
For every 1% increase in the CVD risk calculated by the Framingham Risk Score, the odds of CVD increased by 4% in the DISH group versus the control group (P = .02).
While there was a significant (P < .003) difference in the Framingham Risk Score between the DISH and control groups in 2006 (28.6% vs. 17.8%), there was no overall statistical difference (P = .2) in the composite CVD outcome (38.8% vs. 25.5%) 10 years later, as calculated by the revised Framingham Risk Score, which included MI, cerebrovascular accident, transient ischemic attack, peripheral artery disease, and heart failure with preserved ejection fraction.
“We are dealing with patients who are in their 70s. So, it is expected that this group of patients will be more often affected by cardiovascular disease” than younger individuals, Dr. Mader observed. That said, the study’s findings “confirm the theory that patients with DISH have a high likelihood of developing cardiovascular disease,” he added, acknowledging that it was only the risk for MI that was statistically significantly higher in people with DISH than in the controls.
DISH and coronary artery calcification
“It might be even more interesting to have a different control population that had no osteoarthritis,” Dr. Regan observed.
As the associate director of the COPDGene study, Dr. Regan has access to data collected from a large cohort of people with chronic obstructive pulmonary disease (COPD; n = 2,728), around 13% of whom were identified as having DISH in one recent study.
In that study, the presence of DISH versus no DISH was associated with a 37% higher risk for having coronary artery calcification (CAC) – a marker for atherosclerosis and cardiovascular disease. Two-thirds of people with DISH had CAC, compared with 46.9% of those without DISH (P < .001). The prevalence of DISH was 8.8% in those without CAC, 12.8% in those with a CAC score of 1-100, 20% in those with a CAC score of 100-400, and 24.7% in those with a CAC score of more than 400, which is associated with a very high risk for coronary artery disease.
Dr. Regan observed that information on heart attacks and strokes were collected within the COPDGene study, so it would be possible to look at cardiovascular risk in their patients with DISH and confirm the findings of Mader and colleagues.
“I think the most important thing is recognizing that there are things going on in the spine that are important to people’s general health,” Dr. Regan said.
Dr. Mader noted: “It makes sense that patients with DISH should be more meticulously followed for at least the traditional risk factors and better treated because they are at a higher risk for these events.”
The study received no financial support. Neither Dr. Mader nor Dr. Regan had any conflicts of interest to disclose.
SOURCE: Glick K et al. Arthritis Res Ther. 2020. doi: 10.1186/s13075-020-02278-w.
FROM ARTHRITIS RESEARCH & THERAPY
COVID-19 outcomes no worse in patients on TNF inhibitors or methotrexate
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
More dairy lowers risk of falls, fractures in frail elderly
Consuming more milk, cheese, or yogurt might be a simple, low-cost way to boost bone health and prevent some falls and fractures in older people living in long-term care facilities, according to a new randomized study from Australia.
“Supplementation using dairy foods is likely to be an effective, safe, widely available, and low cost means of curtailing the public health burden of fractures,” said Sandra Iuliano, PhD, from the University of Melbourne, who presented the findings during the virtual American Society of Bone and Mineral Research 2020 annual meeting.
The researchers randomized 60 old-age institutions to provide residents with their usual menus or a diet with more milk, cheese, or yogurt for 2 years.
The residents with the altered menus increased their dairy consumption from 2 servings/day to 3.5 servings/day, which was reflected in a greater intake of calcium and protein, along with fewer falls, total fractures, and hip fractures than in the control group.
“This is the first randomized trial to show a benefit of dairy food intake on risk of fractures,” Walter Willett, MD, DrPH, professor of nutrition and epidemiology at the Harvard School of Public Health, Boston, said in an interview.
The results are “not surprising” because supplements of calcium plus vitamin D have reduced the risk of fractures in a similar population of older residents living in special living facilities, said Dr. Willett, coauthor of a recent review article, “Milk and Health,” published in the New England Journal of Medicine.
“It is important for everyone to have adequate intake of calcium and vitamin D,” he said. However, “it isn’t clear whether it is better to ensure this clinically by supplements, overall healthy diet, or extra dairy intake,” he added, noting that consuming the amount of dairy given in this Australian study is not environmentally sustainable.
Clifford Rosen, MD, professor of medicine, Tufts University, Boston, said in an interview that the Australian researchers studied the impact of increased dietary calcium and protein, not the impact of vitamin D via supplements.
“This is progress toward getting interventions to our most needy residents to prevent fractures – probably the most compelling data that we have had in a number of years,” he noted.
The current study shows “it’s not [the] vitamin D,” because the residents had initial low calcium levels but normal vitamin D levels. “For too long we’ve been stuck on the idea that it is [increasing] vitamin D in the elderly that causes a reduction in fractures,” said Dr. Rosen. “The data are not very supportive of it, but people continue to think that’s the most important element.”
On the other hand, the current study raises certain questions. “What we don’t know is, is it the calcium, or is it the protein, or the combination, that had an impact?”
Would upping dairy decrease falls?
Older adults living in institutions have a high risk of falls and fractures, including hip fractures, and “malnutrition is common,” said Dr. Iuliano during her presentation.
Prior studies have reported that such residents have a daily dietary calcium intake of 635 mg (half the recommended 1,300 mg), a protein intake of 0.8 g/kg body weight (less than the recommended 1 g/kg body weight), and a dairy intake of 1.5 servings (about a third of the recommended amount), she said.
The group hypothesized that upping dairy intake of elderly residents living in long-term care institutions would reduce the risk of fractures. They performed a 2-year cluster-randomized trial in 60 facilities in Melbourne and surrounding areas.
Half gave their 3,301 residents menus with a higher dairy content, and the other half gave their 3,894 residents (controls) the usual menus.
The residents in both groups had similar characteristics: they were a mean age of 87 years and 68% were women. A subgroup had blood tests and bone morphology studies at baseline and 1 year.
Researchers verified nutrient intake by analyzing the menus and doing plate waste analysis for a subgroup, and they determined the number of falls and fractures from incident and hospital x-ray reports, respectively.
One-third fewer fractures in the higher-dairy group
At the study start, residents in both groups had similar vitamin D levels (72 nmol/L) and bone morphology. They were consuming two servings of dairy food and drink a day, where a serving was 250 mL of milk (including lactose-free milk) or 200 g of yogurt or 40 g of cheese.
Their initial daily calcium intake was 650 mg, which stayed the same in the control group, but increased to >1100 mg in the intervention group.
Their initial daily protein intake was around 59 g, which remained the same in the control group, but increased to about 72 grams (1.1 g/kg body weight) in the intervention group.
At 2 years, the 1.5 servings/day increase in dairy intake in the control versus intervention group was associated with an 11% reduction in falls (62% vs. 57%), a 33% reduction in fractures (5.2% vs. 3.7%), a 46% reduction in hip fractures (2.4% vs. 1.3%), and no difference in mortality (28% in both groups).
The intervention was also associated with a slowing in bone loss and an increase in insulinlike growth factor–1.
Four dairy servings a day “is high”
Dr. Willett said that “it is reasonable for seniors to take one or two servings of dairy per day, but four servings per day, as in this study, is probably not necessary.”
Moreover, “dairy production has a major impact on greenhouse gas emissions, and even two servings per day would not be environmentally sustainable if everyone were to consume this amount,” he observed.
“Because the world is facing an existential threat from climate change, general advice to consume high amounts of dairy products would be irresponsible as we can get all essential nutrients from other sources,” he added. “That said, modest amounts of dairy foods, such as one to two servings per day could be reasonable. There is some suggestive evidence that dairy in the form of yogurt may have particular benefits.”
The study was funded by Melbourne University and various dietary councils. Dr. Iuliano reported receiving lecture fees from Abbott. Dr. Rosen and Dr. Willett reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Consuming more milk, cheese, or yogurt might be a simple, low-cost way to boost bone health and prevent some falls and fractures in older people living in long-term care facilities, according to a new randomized study from Australia.
“Supplementation using dairy foods is likely to be an effective, safe, widely available, and low cost means of curtailing the public health burden of fractures,” said Sandra Iuliano, PhD, from the University of Melbourne, who presented the findings during the virtual American Society of Bone and Mineral Research 2020 annual meeting.
The researchers randomized 60 old-age institutions to provide residents with their usual menus or a diet with more milk, cheese, or yogurt for 2 years.
The residents with the altered menus increased their dairy consumption from 2 servings/day to 3.5 servings/day, which was reflected in a greater intake of calcium and protein, along with fewer falls, total fractures, and hip fractures than in the control group.
“This is the first randomized trial to show a benefit of dairy food intake on risk of fractures,” Walter Willett, MD, DrPH, professor of nutrition and epidemiology at the Harvard School of Public Health, Boston, said in an interview.
The results are “not surprising” because supplements of calcium plus vitamin D have reduced the risk of fractures in a similar population of older residents living in special living facilities, said Dr. Willett, coauthor of a recent review article, “Milk and Health,” published in the New England Journal of Medicine.
“It is important for everyone to have adequate intake of calcium and vitamin D,” he said. However, “it isn’t clear whether it is better to ensure this clinically by supplements, overall healthy diet, or extra dairy intake,” he added, noting that consuming the amount of dairy given in this Australian study is not environmentally sustainable.
Clifford Rosen, MD, professor of medicine, Tufts University, Boston, said in an interview that the Australian researchers studied the impact of increased dietary calcium and protein, not the impact of vitamin D via supplements.
“This is progress toward getting interventions to our most needy residents to prevent fractures – probably the most compelling data that we have had in a number of years,” he noted.
The current study shows “it’s not [the] vitamin D,” because the residents had initial low calcium levels but normal vitamin D levels. “For too long we’ve been stuck on the idea that it is [increasing] vitamin D in the elderly that causes a reduction in fractures,” said Dr. Rosen. “The data are not very supportive of it, but people continue to think that’s the most important element.”
On the other hand, the current study raises certain questions. “What we don’t know is, is it the calcium, or is it the protein, or the combination, that had an impact?”
Would upping dairy decrease falls?
Older adults living in institutions have a high risk of falls and fractures, including hip fractures, and “malnutrition is common,” said Dr. Iuliano during her presentation.
Prior studies have reported that such residents have a daily dietary calcium intake of 635 mg (half the recommended 1,300 mg), a protein intake of 0.8 g/kg body weight (less than the recommended 1 g/kg body weight), and a dairy intake of 1.5 servings (about a third of the recommended amount), she said.
The group hypothesized that upping dairy intake of elderly residents living in long-term care institutions would reduce the risk of fractures. They performed a 2-year cluster-randomized trial in 60 facilities in Melbourne and surrounding areas.
Half gave their 3,301 residents menus with a higher dairy content, and the other half gave their 3,894 residents (controls) the usual menus.
The residents in both groups had similar characteristics: they were a mean age of 87 years and 68% were women. A subgroup had blood tests and bone morphology studies at baseline and 1 year.
Researchers verified nutrient intake by analyzing the menus and doing plate waste analysis for a subgroup, and they determined the number of falls and fractures from incident and hospital x-ray reports, respectively.
One-third fewer fractures in the higher-dairy group
At the study start, residents in both groups had similar vitamin D levels (72 nmol/L) and bone morphology. They were consuming two servings of dairy food and drink a day, where a serving was 250 mL of milk (including lactose-free milk) or 200 g of yogurt or 40 g of cheese.
Their initial daily calcium intake was 650 mg, which stayed the same in the control group, but increased to >1100 mg in the intervention group.
Their initial daily protein intake was around 59 g, which remained the same in the control group, but increased to about 72 grams (1.1 g/kg body weight) in the intervention group.
At 2 years, the 1.5 servings/day increase in dairy intake in the control versus intervention group was associated with an 11% reduction in falls (62% vs. 57%), a 33% reduction in fractures (5.2% vs. 3.7%), a 46% reduction in hip fractures (2.4% vs. 1.3%), and no difference in mortality (28% in both groups).
The intervention was also associated with a slowing in bone loss and an increase in insulinlike growth factor–1.
Four dairy servings a day “is high”
Dr. Willett said that “it is reasonable for seniors to take one or two servings of dairy per day, but four servings per day, as in this study, is probably not necessary.”
Moreover, “dairy production has a major impact on greenhouse gas emissions, and even two servings per day would not be environmentally sustainable if everyone were to consume this amount,” he observed.
“Because the world is facing an existential threat from climate change, general advice to consume high amounts of dairy products would be irresponsible as we can get all essential nutrients from other sources,” he added. “That said, modest amounts of dairy foods, such as one to two servings per day could be reasonable. There is some suggestive evidence that dairy in the form of yogurt may have particular benefits.”
The study was funded by Melbourne University and various dietary councils. Dr. Iuliano reported receiving lecture fees from Abbott. Dr. Rosen and Dr. Willett reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Consuming more milk, cheese, or yogurt might be a simple, low-cost way to boost bone health and prevent some falls and fractures in older people living in long-term care facilities, according to a new randomized study from Australia.
“Supplementation using dairy foods is likely to be an effective, safe, widely available, and low cost means of curtailing the public health burden of fractures,” said Sandra Iuliano, PhD, from the University of Melbourne, who presented the findings during the virtual American Society of Bone and Mineral Research 2020 annual meeting.
The researchers randomized 60 old-age institutions to provide residents with their usual menus or a diet with more milk, cheese, or yogurt for 2 years.
The residents with the altered menus increased their dairy consumption from 2 servings/day to 3.5 servings/day, which was reflected in a greater intake of calcium and protein, along with fewer falls, total fractures, and hip fractures than in the control group.
“This is the first randomized trial to show a benefit of dairy food intake on risk of fractures,” Walter Willett, MD, DrPH, professor of nutrition and epidemiology at the Harvard School of Public Health, Boston, said in an interview.
The results are “not surprising” because supplements of calcium plus vitamin D have reduced the risk of fractures in a similar population of older residents living in special living facilities, said Dr. Willett, coauthor of a recent review article, “Milk and Health,” published in the New England Journal of Medicine.
“It is important for everyone to have adequate intake of calcium and vitamin D,” he said. However, “it isn’t clear whether it is better to ensure this clinically by supplements, overall healthy diet, or extra dairy intake,” he added, noting that consuming the amount of dairy given in this Australian study is not environmentally sustainable.
Clifford Rosen, MD, professor of medicine, Tufts University, Boston, said in an interview that the Australian researchers studied the impact of increased dietary calcium and protein, not the impact of vitamin D via supplements.
“This is progress toward getting interventions to our most needy residents to prevent fractures – probably the most compelling data that we have had in a number of years,” he noted.
The current study shows “it’s not [the] vitamin D,” because the residents had initial low calcium levels but normal vitamin D levels. “For too long we’ve been stuck on the idea that it is [increasing] vitamin D in the elderly that causes a reduction in fractures,” said Dr. Rosen. “The data are not very supportive of it, but people continue to think that’s the most important element.”
On the other hand, the current study raises certain questions. “What we don’t know is, is it the calcium, or is it the protein, or the combination, that had an impact?”
Would upping dairy decrease falls?
Older adults living in institutions have a high risk of falls and fractures, including hip fractures, and “malnutrition is common,” said Dr. Iuliano during her presentation.
Prior studies have reported that such residents have a daily dietary calcium intake of 635 mg (half the recommended 1,300 mg), a protein intake of 0.8 g/kg body weight (less than the recommended 1 g/kg body weight), and a dairy intake of 1.5 servings (about a third of the recommended amount), she said.
The group hypothesized that upping dairy intake of elderly residents living in long-term care institutions would reduce the risk of fractures. They performed a 2-year cluster-randomized trial in 60 facilities in Melbourne and surrounding areas.
Half gave their 3,301 residents menus with a higher dairy content, and the other half gave their 3,894 residents (controls) the usual menus.
The residents in both groups had similar characteristics: they were a mean age of 87 years and 68% were women. A subgroup had blood tests and bone morphology studies at baseline and 1 year.
Researchers verified nutrient intake by analyzing the menus and doing plate waste analysis for a subgroup, and they determined the number of falls and fractures from incident and hospital x-ray reports, respectively.
One-third fewer fractures in the higher-dairy group
At the study start, residents in both groups had similar vitamin D levels (72 nmol/L) and bone morphology. They were consuming two servings of dairy food and drink a day, where a serving was 250 mL of milk (including lactose-free milk) or 200 g of yogurt or 40 g of cheese.
Their initial daily calcium intake was 650 mg, which stayed the same in the control group, but increased to >1100 mg in the intervention group.
Their initial daily protein intake was around 59 g, which remained the same in the control group, but increased to about 72 grams (1.1 g/kg body weight) in the intervention group.
At 2 years, the 1.5 servings/day increase in dairy intake in the control versus intervention group was associated with an 11% reduction in falls (62% vs. 57%), a 33% reduction in fractures (5.2% vs. 3.7%), a 46% reduction in hip fractures (2.4% vs. 1.3%), and no difference in mortality (28% in both groups).
The intervention was also associated with a slowing in bone loss and an increase in insulinlike growth factor–1.
Four dairy servings a day “is high”
Dr. Willett said that “it is reasonable for seniors to take one or two servings of dairy per day, but four servings per day, as in this study, is probably not necessary.”
Moreover, “dairy production has a major impact on greenhouse gas emissions, and even two servings per day would not be environmentally sustainable if everyone were to consume this amount,” he observed.
“Because the world is facing an existential threat from climate change, general advice to consume high amounts of dairy products would be irresponsible as we can get all essential nutrients from other sources,” he added. “That said, modest amounts of dairy foods, such as one to two servings per day could be reasonable. There is some suggestive evidence that dairy in the form of yogurt may have particular benefits.”
The study was funded by Melbourne University and various dietary councils. Dr. Iuliano reported receiving lecture fees from Abbott. Dr. Rosen and Dr. Willett reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ASBMR 2020
Mega vitamin D harms bone in women, not men, without osteoporosis
“More is not necessarily better” when it comes to vitamin D supplements for women with adequate serum levels, new research suggests.
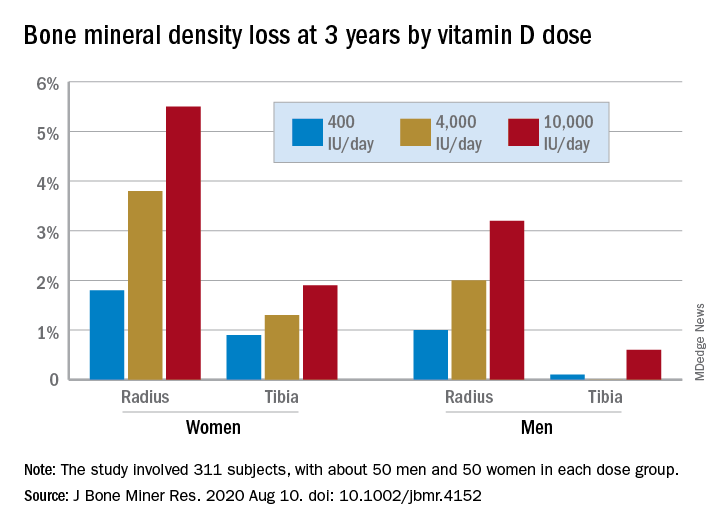
In a study of healthy 55- to 70-year-old women who took very-high-dose vitamin D supplements – either 4,000 IU/day or the previously identified “upper safe limit” of 10,000 IU/day – for 3 years had a significantly greater loss of total bone mineral density (BMD) at the radius and tibia than did women who took 400 IU/day. However, this effect was not seen in men. And the higher-dose vitamin D supplements did not improve bone strength in men or women.
But this was an exploratory post hoc analysis, and these were healthy community-dwelling adults with sufficient serum vitamin D levels (and no osteoporosis) at study entry, stressed lead researcher Lauren A. Burt, PhD, from the University of Calgary, in Alberta, Canada.
Dr. Burt presented these findings Sept. 11 at the virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and the study was also recently published online in the Journal of Bone and Mineral Research.
The results suggest that, “if you have normal bone density and adequate levels of vitamin D, there is no bone benefit in taking doses of vitamin D above the standard recommendations designed to prevent vitamin D deficiency, and doses at or above 4,000 IU/day might even be detrimental to bone, especially in females,” Dr. Burt said in an interview.
“These results are clinically relevant,” Dr. Burt and her coauthors wrote, “as vitamin D supplementation is widely administered to postmenopausal females for osteoporosis prevention.”
“Our findings do not support a benefit of high-dose vitamin D supplementation for bone health and raise the possibility of harm for females.”
Invited to comment, Meryl S. LeBoff, MD, of Harvard Medical School, Boston, said in an interview that this finding “warrants further research” because it is “important” to discover sex differences in bone responses to vitamin D.
“This doesn’t apply to osteoporosis”
Dr. LeBoff was lead author of a subanalysis of the Vitamin D and Omega-3 Trial (VITAL).
As she reported at last year’s ASBMR meeting, that analysis showed that, in healthy adults who did not have vitamin D insufficiency, taking vitamin D3 supplements for 2 years did not improve BMD, compared with placebo (recently published), nor was this linked with fewer fractures.
Dr. LeBoff pointed out that the current study investigated “very high doses of vitamin D” – at least double the 2,000 IU/day doses examined in VITAL.
Also, the serum vitamin D levels in this study were “above what we considered the upper normal limit for our assay in our hospital,” she noted, and there was no placebo control.
“We did not see any adverse effects of 2,000 IU/day vitamin D,” Dr. LeBoff stressed.
“At the same time, we didn’t see any significant benefits in terms of bone density because they already had achieved a normal level of vitamin D sufficient for bone.”
But “this doesn’t apply to patients with vitamin D deficiency, patients with osteoporosis, or low bone mass, in which case we would recommend vitamin D.”
Some patients take more vitamin D than they need because they think more is better, said LeBoff, but this study suggests “more is not necessarily better.”
“There’s been a concern for several years that too much vitamin D may be associated with increased fractures,” she emphasized.
Post hoc analysis
The current study analyzed new data from the Calgary Vitamin D study.
That study found no benefit in BMD or bone strength (JAMA. 2019;322[8]:736-45), contrary to the researchers’ hypothesis that high-dose vitamin D supplements would be associated with greater calcium absorption and parathyroid hormone suppression and, thus, reduced age-related bone loss (improved bone density and strength).
Instead, they found a negative dose-response relationship, which “should be regarded as hypothesis generating, requiring confirmation with further research,” they wrote.
The current study sought to determine if there were sex differences in the effect of vitamin D supplements on bone health in this population.
From October 2013 to December 2017, the Canada Vitamin D study enrolled 311 participants (53% male). To be eligible for the study, participants had to have serum 25-hydroxyvitamin D levels greater than 30 nmol/L and less than 125 nmol/L. They also needed to have adequate calcium intake (1,200 mg/day, as defined by the U.S. Institute of Medicine), or if not, they were instructed to take an appropriate calcium supplement dose.
Patients were randomized to receive 400, 4,000, or 10,000 IU/day of vitamin D3 cholecalciferol, given as 5 drops/day of liquid (Ddrops), with roughly 50 men and 50 women in each dose group.
Researchers selected the 400 IU/day dose as the comparator because the Institute of Medicine recommends a vitamin D intake of 600 IU/day for adults under age 70 years to provide the vitamin D needed for bone health. The typical Canadian diet includes 200-300 IU/day of vitamin D, so individuals would need a supplement of 400 IU/day to reach the recommended intake. The 4,000 IU/day dose is the recommended tolerable upper intake level, according to the Institute of Medicine. And the 10,000 IU/day dose is the tolerable upper intake level of vitamin D as identified in a review by Hathcock and colleagues (Am J Clin Nutr. 2007;85:6-18).
Participants underwent scans with high-resolution peripheral quantitative computed tomography (HR-pQCT) to measure total volumetric BMD at the radius and tibia at baseline, 6, 12, 24, and 36 months. Finite element analysis was used to estimate bone strength.
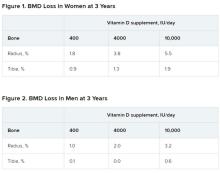
After 3 years, women had lost significantly more BMD at the radius after taking high-dose versus 400 IU/day of vitamin D. Losses in BMD at the tibia followed a similar trend but were smaller (Figure 1). There were no significant changes in this measure among men (Figure 2).
There were also no significant changes in bone strength among men or women.
Biological mechanism remains to be determined
Dr. LeBoff said a “possible biological explanation” for the findings is that “women, particularly when they are younger, lose more bone than men.”
“Postmenopausal females do lose bone at an accelerated rate compared with males,” Dr. Burt agreed, “but at the time the study was designed, there was no reason to believe that high-dose vitamin D supplementation would accelerate the problem.”
“The biological mechanism of the vitamin D–related bone loss needs further investigation,” Dr. Burt added, “but there are laboratory data suggesting that supraphysiologic doses of active metabolites of vitamin D may stimulate bone resorption.”
The study was funded by the Pure North S’Energy Foundation. Dr. Burt has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. LeBoff has reported receiving grants from the National Institutes of Health for the VITAL analysis.
A version of this article originally appeared on Medscape.com.
“More is not necessarily better” when it comes to vitamin D supplements for women with adequate serum levels, new research suggests.

In a study of healthy 55- to 70-year-old women who took very-high-dose vitamin D supplements – either 4,000 IU/day or the previously identified “upper safe limit” of 10,000 IU/day – for 3 years had a significantly greater loss of total bone mineral density (BMD) at the radius and tibia than did women who took 400 IU/day. However, this effect was not seen in men. And the higher-dose vitamin D supplements did not improve bone strength in men or women.
But this was an exploratory post hoc analysis, and these were healthy community-dwelling adults with sufficient serum vitamin D levels (and no osteoporosis) at study entry, stressed lead researcher Lauren A. Burt, PhD, from the University of Calgary, in Alberta, Canada.
Dr. Burt presented these findings Sept. 11 at the virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and the study was also recently published online in the Journal of Bone and Mineral Research.
The results suggest that, “if you have normal bone density and adequate levels of vitamin D, there is no bone benefit in taking doses of vitamin D above the standard recommendations designed to prevent vitamin D deficiency, and doses at or above 4,000 IU/day might even be detrimental to bone, especially in females,” Dr. Burt said in an interview.
“These results are clinically relevant,” Dr. Burt and her coauthors wrote, “as vitamin D supplementation is widely administered to postmenopausal females for osteoporosis prevention.”
“Our findings do not support a benefit of high-dose vitamin D supplementation for bone health and raise the possibility of harm for females.”
Invited to comment, Meryl S. LeBoff, MD, of Harvard Medical School, Boston, said in an interview that this finding “warrants further research” because it is “important” to discover sex differences in bone responses to vitamin D.
“This doesn’t apply to osteoporosis”
Dr. LeBoff was lead author of a subanalysis of the Vitamin D and Omega-3 Trial (VITAL).
As she reported at last year’s ASBMR meeting, that analysis showed that, in healthy adults who did not have vitamin D insufficiency, taking vitamin D3 supplements for 2 years did not improve BMD, compared with placebo (recently published), nor was this linked with fewer fractures.
Dr. LeBoff pointed out that the current study investigated “very high doses of vitamin D” – at least double the 2,000 IU/day doses examined in VITAL.
Also, the serum vitamin D levels in this study were “above what we considered the upper normal limit for our assay in our hospital,” she noted, and there was no placebo control.
“We did not see any adverse effects of 2,000 IU/day vitamin D,” Dr. LeBoff stressed.
“At the same time, we didn’t see any significant benefits in terms of bone density because they already had achieved a normal level of vitamin D sufficient for bone.”
But “this doesn’t apply to patients with vitamin D deficiency, patients with osteoporosis, or low bone mass, in which case we would recommend vitamin D.”
Some patients take more vitamin D than they need because they think more is better, said LeBoff, but this study suggests “more is not necessarily better.”
“There’s been a concern for several years that too much vitamin D may be associated with increased fractures,” she emphasized.
Post hoc analysis
The current study analyzed new data from the Calgary Vitamin D study.
That study found no benefit in BMD or bone strength (JAMA. 2019;322[8]:736-45), contrary to the researchers’ hypothesis that high-dose vitamin D supplements would be associated with greater calcium absorption and parathyroid hormone suppression and, thus, reduced age-related bone loss (improved bone density and strength).
Instead, they found a negative dose-response relationship, which “should be regarded as hypothesis generating, requiring confirmation with further research,” they wrote.
The current study sought to determine if there were sex differences in the effect of vitamin D supplements on bone health in this population.
From October 2013 to December 2017, the Canada Vitamin D study enrolled 311 participants (53% male). To be eligible for the study, participants had to have serum 25-hydroxyvitamin D levels greater than 30 nmol/L and less than 125 nmol/L. They also needed to have adequate calcium intake (1,200 mg/day, as defined by the U.S. Institute of Medicine), or if not, they were instructed to take an appropriate calcium supplement dose.
Patients were randomized to receive 400, 4,000, or 10,000 IU/day of vitamin D3 cholecalciferol, given as 5 drops/day of liquid (Ddrops), with roughly 50 men and 50 women in each dose group.
Researchers selected the 400 IU/day dose as the comparator because the Institute of Medicine recommends a vitamin D intake of 600 IU/day for adults under age 70 years to provide the vitamin D needed for bone health. The typical Canadian diet includes 200-300 IU/day of vitamin D, so individuals would need a supplement of 400 IU/day to reach the recommended intake. The 4,000 IU/day dose is the recommended tolerable upper intake level, according to the Institute of Medicine. And the 10,000 IU/day dose is the tolerable upper intake level of vitamin D as identified in a review by Hathcock and colleagues (Am J Clin Nutr. 2007;85:6-18).
Participants underwent scans with high-resolution peripheral quantitative computed tomography (HR-pQCT) to measure total volumetric BMD at the radius and tibia at baseline, 6, 12, 24, and 36 months. Finite element analysis was used to estimate bone strength.
After 3 years, women had lost significantly more BMD at the radius after taking high-dose versus 400 IU/day of vitamin D. Losses in BMD at the tibia followed a similar trend but were smaller (Figure 1). There were no significant changes in this measure among men (Figure 2).
There were also no significant changes in bone strength among men or women.
Biological mechanism remains to be determined
Dr. LeBoff said a “possible biological explanation” for the findings is that “women, particularly when they are younger, lose more bone than men.”
“Postmenopausal females do lose bone at an accelerated rate compared with males,” Dr. Burt agreed, “but at the time the study was designed, there was no reason to believe that high-dose vitamin D supplementation would accelerate the problem.”
“The biological mechanism of the vitamin D–related bone loss needs further investigation,” Dr. Burt added, “but there are laboratory data suggesting that supraphysiologic doses of active metabolites of vitamin D may stimulate bone resorption.”
The study was funded by the Pure North S’Energy Foundation. Dr. Burt has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. LeBoff has reported receiving grants from the National Institutes of Health for the VITAL analysis.
A version of this article originally appeared on Medscape.com.
“More is not necessarily better” when it comes to vitamin D supplements for women with adequate serum levels, new research suggests.

In a study of healthy 55- to 70-year-old women who took very-high-dose vitamin D supplements – either 4,000 IU/day or the previously identified “upper safe limit” of 10,000 IU/day – for 3 years had a significantly greater loss of total bone mineral density (BMD) at the radius and tibia than did women who took 400 IU/day. However, this effect was not seen in men. And the higher-dose vitamin D supplements did not improve bone strength in men or women.
But this was an exploratory post hoc analysis, and these were healthy community-dwelling adults with sufficient serum vitamin D levels (and no osteoporosis) at study entry, stressed lead researcher Lauren A. Burt, PhD, from the University of Calgary, in Alberta, Canada.
Dr. Burt presented these findings Sept. 11 at the virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and the study was also recently published online in the Journal of Bone and Mineral Research.
The results suggest that, “if you have normal bone density and adequate levels of vitamin D, there is no bone benefit in taking doses of vitamin D above the standard recommendations designed to prevent vitamin D deficiency, and doses at or above 4,000 IU/day might even be detrimental to bone, especially in females,” Dr. Burt said in an interview.
“These results are clinically relevant,” Dr. Burt and her coauthors wrote, “as vitamin D supplementation is widely administered to postmenopausal females for osteoporosis prevention.”
“Our findings do not support a benefit of high-dose vitamin D supplementation for bone health and raise the possibility of harm for females.”
Invited to comment, Meryl S. LeBoff, MD, of Harvard Medical School, Boston, said in an interview that this finding “warrants further research” because it is “important” to discover sex differences in bone responses to vitamin D.
“This doesn’t apply to osteoporosis”
Dr. LeBoff was lead author of a subanalysis of the Vitamin D and Omega-3 Trial (VITAL).
As she reported at last year’s ASBMR meeting, that analysis showed that, in healthy adults who did not have vitamin D insufficiency, taking vitamin D3 supplements for 2 years did not improve BMD, compared with placebo (recently published), nor was this linked with fewer fractures.
Dr. LeBoff pointed out that the current study investigated “very high doses of vitamin D” – at least double the 2,000 IU/day doses examined in VITAL.
Also, the serum vitamin D levels in this study were “above what we considered the upper normal limit for our assay in our hospital,” she noted, and there was no placebo control.
“We did not see any adverse effects of 2,000 IU/day vitamin D,” Dr. LeBoff stressed.
“At the same time, we didn’t see any significant benefits in terms of bone density because they already had achieved a normal level of vitamin D sufficient for bone.”
But “this doesn’t apply to patients with vitamin D deficiency, patients with osteoporosis, or low bone mass, in which case we would recommend vitamin D.”
Some patients take more vitamin D than they need because they think more is better, said LeBoff, but this study suggests “more is not necessarily better.”
“There’s been a concern for several years that too much vitamin D may be associated with increased fractures,” she emphasized.
Post hoc analysis
The current study analyzed new data from the Calgary Vitamin D study.
That study found no benefit in BMD or bone strength (JAMA. 2019;322[8]:736-45), contrary to the researchers’ hypothesis that high-dose vitamin D supplements would be associated with greater calcium absorption and parathyroid hormone suppression and, thus, reduced age-related bone loss (improved bone density and strength).
Instead, they found a negative dose-response relationship, which “should be regarded as hypothesis generating, requiring confirmation with further research,” they wrote.
The current study sought to determine if there were sex differences in the effect of vitamin D supplements on bone health in this population.
From October 2013 to December 2017, the Canada Vitamin D study enrolled 311 participants (53% male). To be eligible for the study, participants had to have serum 25-hydroxyvitamin D levels greater than 30 nmol/L and less than 125 nmol/L. They also needed to have adequate calcium intake (1,200 mg/day, as defined by the U.S. Institute of Medicine), or if not, they were instructed to take an appropriate calcium supplement dose.
Patients were randomized to receive 400, 4,000, or 10,000 IU/day of vitamin D3 cholecalciferol, given as 5 drops/day of liquid (Ddrops), with roughly 50 men and 50 women in each dose group.
Researchers selected the 400 IU/day dose as the comparator because the Institute of Medicine recommends a vitamin D intake of 600 IU/day for adults under age 70 years to provide the vitamin D needed for bone health. The typical Canadian diet includes 200-300 IU/day of vitamin D, so individuals would need a supplement of 400 IU/day to reach the recommended intake. The 4,000 IU/day dose is the recommended tolerable upper intake level, according to the Institute of Medicine. And the 10,000 IU/day dose is the tolerable upper intake level of vitamin D as identified in a review by Hathcock and colleagues (Am J Clin Nutr. 2007;85:6-18).
Participants underwent scans with high-resolution peripheral quantitative computed tomography (HR-pQCT) to measure total volumetric BMD at the radius and tibia at baseline, 6, 12, 24, and 36 months. Finite element analysis was used to estimate bone strength.
After 3 years, women had lost significantly more BMD at the radius after taking high-dose versus 400 IU/day of vitamin D. Losses in BMD at the tibia followed a similar trend but were smaller (Figure 1). There were no significant changes in this measure among men (Figure 2).
There were also no significant changes in bone strength among men or women.
Biological mechanism remains to be determined
Dr. LeBoff said a “possible biological explanation” for the findings is that “women, particularly when they are younger, lose more bone than men.”
“Postmenopausal females do lose bone at an accelerated rate compared with males,” Dr. Burt agreed, “but at the time the study was designed, there was no reason to believe that high-dose vitamin D supplementation would accelerate the problem.”
“The biological mechanism of the vitamin D–related bone loss needs further investigation,” Dr. Burt added, “but there are laboratory data suggesting that supraphysiologic doses of active metabolites of vitamin D may stimulate bone resorption.”
The study was funded by the Pure North S’Energy Foundation. Dr. Burt has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. LeBoff has reported receiving grants from the National Institutes of Health for the VITAL analysis.
A version of this article originally appeared on Medscape.com.
High disability after a year of RA treatment signals increased mortality risk
over the course of up to 10 years of follow-up, according to an analysis of patients enrolled in the Canadian Early Arthritis Cohort (CATCH).
Higher Disease Activity Score in 28 joints (DAS28) at follow-up was also associated with higher all-cause mortality among the patients, who all took at least one conventional synthetic or biologic disease-modifying antirheumatic drug during the first year. Higher DAS28 scores in previous studies has been associated with increased disability as measured by the HAQ, Safoora Fatima, MD, of the University of Western Ontario, London, and colleagues wrote in Arthritis & Rheumatology.
“Combining our study findings with this association suggests that poorer disease control (high DAS28) within the first treatment year for RA may lead to increased disability (high HAQ scores) which in turn may contribute to higher mortality. This may indicate that RA patients who do not have a deep response in the first year to treatment have higher subsequent mortality,” the researchers wrote.
In addition to higher HAQ scores, all-cause mortality was independently associated with age, male sex, lower education, smoking, more comorbidities, higher baseline disease activity, and glucocorticoid use. “This is helpful in a clinical setting as it can guide physician-patient discussions in terms of risk factors associated with prognosis, prescribing glucocorticoids, counseling on smoking cessation, monitoring treatment responses, and focusing on patient education,” the authors wrote.
While the impact of increased disease activity and damage likely plays a role in the association between high HAQ score and increased mortality, the authors noted that “comorbidities could be causing deaths and those with comorbidities in [early RA] have less chance of remission and more functional impairment at 1 year versus those without any comorbidities, as has been shown [before] in the CATCH [early RA] cohort.”
Dr. Fatima and associates studied 1,724 patients with RA who had a symptom duration of less than 1 year at the time of enrollment in CATCH during 2007-2017. These patients had a mean age of 55 years, and 72% were women. Over the 10-year follow up period, 62 patients (2.4%) died. HAQ scores proved to be significantly higher at both baseline and 1 year for those who died, going from 1.2 to 0.9, compared with scores moving from 1.0 to 0.5 among patients who did not die. (The HAQ has eight categories that are each scored 0-3, with 0 meaning no self-reported functional impairment and 3 meaning severe functional impairment.) Similarly, DAS28 scores were significantly higher at both time points for patients who died versus those who lived, declining from 5.4 to 3.6 for deceased and from 4.9 to 2.8 for nondeceased patients in a year.
Whereas HAQ at baseline was not significantly associated with all-cause mortality in a multivariate, discrete-time survival model that adjusted for age, gender, comorbidities, disease activity, smoking, education, seropositivity, symptom duration, and glucocorticoid use, the association between HAQ at 1 year and death remained statistically significant with a hazard ratio of 1.87.
The authors noted that potential confounders may not have been adjusted for in the comparisons, such as “variable access to advanced therapies, other comorbidities not in the standardized comorbidity questionnaire, [and] severity of comorbidities.”
CATCH has been funded over many years by multiple companies including Amgen and Pfizer Canada, AbbVie, Medexus, Eli Lilly Canada, Merck Canada, Sandoz, Hoffman–La Roche, Janssen, UCB Canada, Bristol-Myers Squibb Canada, and Sanofi Genzyme. The authors had no disclosures.
SOURCE: Fatima S et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41513.
over the course of up to 10 years of follow-up, according to an analysis of patients enrolled in the Canadian Early Arthritis Cohort (CATCH).
Higher Disease Activity Score in 28 joints (DAS28) at follow-up was also associated with higher all-cause mortality among the patients, who all took at least one conventional synthetic or biologic disease-modifying antirheumatic drug during the first year. Higher DAS28 scores in previous studies has been associated with increased disability as measured by the HAQ, Safoora Fatima, MD, of the University of Western Ontario, London, and colleagues wrote in Arthritis & Rheumatology.
“Combining our study findings with this association suggests that poorer disease control (high DAS28) within the first treatment year for RA may lead to increased disability (high HAQ scores) which in turn may contribute to higher mortality. This may indicate that RA patients who do not have a deep response in the first year to treatment have higher subsequent mortality,” the researchers wrote.
In addition to higher HAQ scores, all-cause mortality was independently associated with age, male sex, lower education, smoking, more comorbidities, higher baseline disease activity, and glucocorticoid use. “This is helpful in a clinical setting as it can guide physician-patient discussions in terms of risk factors associated with prognosis, prescribing glucocorticoids, counseling on smoking cessation, monitoring treatment responses, and focusing on patient education,” the authors wrote.
While the impact of increased disease activity and damage likely plays a role in the association between high HAQ score and increased mortality, the authors noted that “comorbidities could be causing deaths and those with comorbidities in [early RA] have less chance of remission and more functional impairment at 1 year versus those without any comorbidities, as has been shown [before] in the CATCH [early RA] cohort.”
Dr. Fatima and associates studied 1,724 patients with RA who had a symptom duration of less than 1 year at the time of enrollment in CATCH during 2007-2017. These patients had a mean age of 55 years, and 72% were women. Over the 10-year follow up period, 62 patients (2.4%) died. HAQ scores proved to be significantly higher at both baseline and 1 year for those who died, going from 1.2 to 0.9, compared with scores moving from 1.0 to 0.5 among patients who did not die. (The HAQ has eight categories that are each scored 0-3, with 0 meaning no self-reported functional impairment and 3 meaning severe functional impairment.) Similarly, DAS28 scores were significantly higher at both time points for patients who died versus those who lived, declining from 5.4 to 3.6 for deceased and from 4.9 to 2.8 for nondeceased patients in a year.
Whereas HAQ at baseline was not significantly associated with all-cause mortality in a multivariate, discrete-time survival model that adjusted for age, gender, comorbidities, disease activity, smoking, education, seropositivity, symptom duration, and glucocorticoid use, the association between HAQ at 1 year and death remained statistically significant with a hazard ratio of 1.87.
The authors noted that potential confounders may not have been adjusted for in the comparisons, such as “variable access to advanced therapies, other comorbidities not in the standardized comorbidity questionnaire, [and] severity of comorbidities.”
CATCH has been funded over many years by multiple companies including Amgen and Pfizer Canada, AbbVie, Medexus, Eli Lilly Canada, Merck Canada, Sandoz, Hoffman–La Roche, Janssen, UCB Canada, Bristol-Myers Squibb Canada, and Sanofi Genzyme. The authors had no disclosures.
SOURCE: Fatima S et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41513.
over the course of up to 10 years of follow-up, according to an analysis of patients enrolled in the Canadian Early Arthritis Cohort (CATCH).
Higher Disease Activity Score in 28 joints (DAS28) at follow-up was also associated with higher all-cause mortality among the patients, who all took at least one conventional synthetic or biologic disease-modifying antirheumatic drug during the first year. Higher DAS28 scores in previous studies has been associated with increased disability as measured by the HAQ, Safoora Fatima, MD, of the University of Western Ontario, London, and colleagues wrote in Arthritis & Rheumatology.
“Combining our study findings with this association suggests that poorer disease control (high DAS28) within the first treatment year for RA may lead to increased disability (high HAQ scores) which in turn may contribute to higher mortality. This may indicate that RA patients who do not have a deep response in the first year to treatment have higher subsequent mortality,” the researchers wrote.
In addition to higher HAQ scores, all-cause mortality was independently associated with age, male sex, lower education, smoking, more comorbidities, higher baseline disease activity, and glucocorticoid use. “This is helpful in a clinical setting as it can guide physician-patient discussions in terms of risk factors associated with prognosis, prescribing glucocorticoids, counseling on smoking cessation, monitoring treatment responses, and focusing on patient education,” the authors wrote.
While the impact of increased disease activity and damage likely plays a role in the association between high HAQ score and increased mortality, the authors noted that “comorbidities could be causing deaths and those with comorbidities in [early RA] have less chance of remission and more functional impairment at 1 year versus those without any comorbidities, as has been shown [before] in the CATCH [early RA] cohort.”
Dr. Fatima and associates studied 1,724 patients with RA who had a symptom duration of less than 1 year at the time of enrollment in CATCH during 2007-2017. These patients had a mean age of 55 years, and 72% were women. Over the 10-year follow up period, 62 patients (2.4%) died. HAQ scores proved to be significantly higher at both baseline and 1 year for those who died, going from 1.2 to 0.9, compared with scores moving from 1.0 to 0.5 among patients who did not die. (The HAQ has eight categories that are each scored 0-3, with 0 meaning no self-reported functional impairment and 3 meaning severe functional impairment.) Similarly, DAS28 scores were significantly higher at both time points for patients who died versus those who lived, declining from 5.4 to 3.6 for deceased and from 4.9 to 2.8 for nondeceased patients in a year.
Whereas HAQ at baseline was not significantly associated with all-cause mortality in a multivariate, discrete-time survival model that adjusted for age, gender, comorbidities, disease activity, smoking, education, seropositivity, symptom duration, and glucocorticoid use, the association between HAQ at 1 year and death remained statistically significant with a hazard ratio of 1.87.
The authors noted that potential confounders may not have been adjusted for in the comparisons, such as “variable access to advanced therapies, other comorbidities not in the standardized comorbidity questionnaire, [and] severity of comorbidities.”
CATCH has been funded over many years by multiple companies including Amgen and Pfizer Canada, AbbVie, Medexus, Eli Lilly Canada, Merck Canada, Sandoz, Hoffman–La Roche, Janssen, UCB Canada, Bristol-Myers Squibb Canada, and Sanofi Genzyme. The authors had no disclosures.
SOURCE: Fatima S et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41513.
FROM ARTHRITIS & RHEUMATOLOGY
A practical approach to knee OA
CASE A 73-year-old woman presents to your clinic with 1 year of gradual-onset left knee pain. The pain is worse at the medial knee and at the beginning and end of the day, with some mild improvement after activity in the morning. The patient has already tried oral acetaminophen, an over-the-counter menthol cream, and a soft elastic knee brace, but these interventions have helped only minimally.
On physical exam, there is no obvious deformity of the knee. There is a bit of small joint effusion without redness or warmth. There is mild tenderness to palpation of the medial joint line. Radiographic findings include osteophytes of the medial and lateral tibial plateaus and medial and lateral femoral condyles with mild joint-space narrowing of the medial compartment, consistent with mild osteoarthritis.
How would you manage this patient’s care?
The knee is the most common joint to be affected by osteoarthritis (OA) and accounts for the majority of the disease’s total burden.1 More than 19% of American adults ages ≥ 45 years have knee OA,1,2 and more than half of the people with symptomatic knee OA in the United States are younger than 65 years of age.3 Longer lifespan and increasing rates of obesity are thought to be driving the increasing prevalence of knee OA, although this remains debated.1 Risk factors for knee OA are outlined in TABLE.1,4-8

Diagnosis: Radiographs are helpful, not essential
The diagnosis of knee OA is relatively straightforward. Gradual onset of knee joint pain is present most days, with pain worse after activity and better with rest. Patients are usually middle-aged or older and/or have a distant history of knee joint injury. Other signs, symptoms, and physical exam findings associated with knee OA include: morning stiffness < 30 minutes, crepitus, instability, range-of-motion deficit, varus or valgus deformity, bony exostosis, joint-line tenderness, joint swelling/effusion, and the absence of erythema/warmth.1,9,10
Although radiographs are not necessary to diagnose knee OA, they can be helpful in confirming the diagnosis by assessing the degree and location of OA and ruling out other pathology. Standing, weight-bearing radiographs are particularly helpful for assessing the degree of joint-space narrowing. In addition to joint-space narrowing, radiographic findings indicative of knee OA include marginal osteophytes, subchondral sclerosis, and subchondral cysts. (See FIGURE 1.)
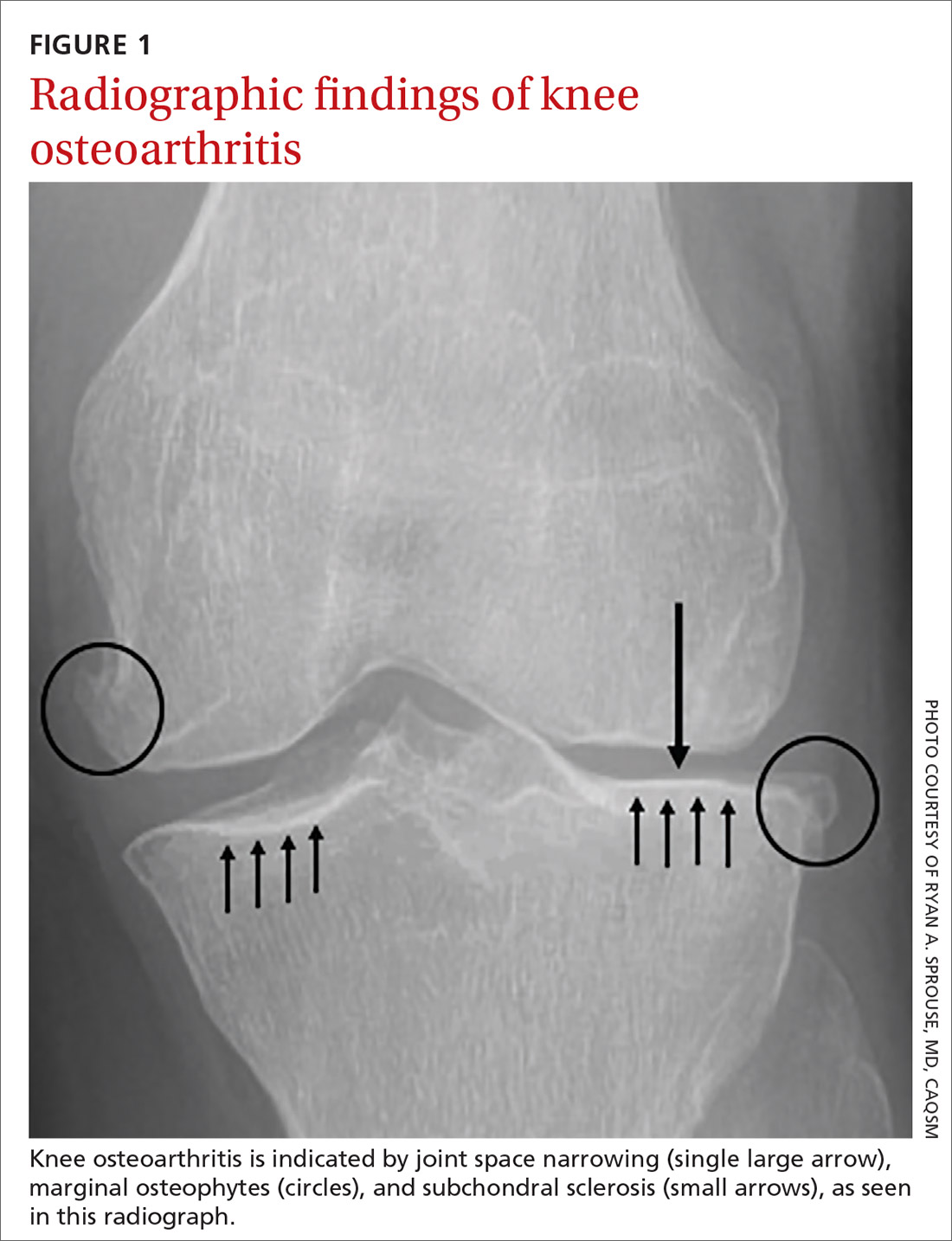
Keep in mind that radiographs are less sensitive for early OA, that the degree of OA seen on radiographs does not correlate well with symptoms, and that radiographic evidence of OA is a common incidental finding—especially in elderly individuals.11 Although not routinely utilized for knee OA diagnosis, magnetic resonance imaging (MRI) can be used to assess for earlier stages of the disease and to rule out pathology associated with the soft tissue and cartilage that is not directly associated with OA.
Continue to: Management
Management: Decrease pain, improve function, slow progression
Because there is no cure for OA, the primary goals of treatment are to decrease pain, improve function of the joint, and slow progression of the disease. As a result, a multifaceted treatment approach is usually undertaken that includes weight reduction and exercise therapy and may include pharmacotherapy, depending on the degree of symptoms. FIGURE 2 contains a summary of the stepwise management of knee OA.
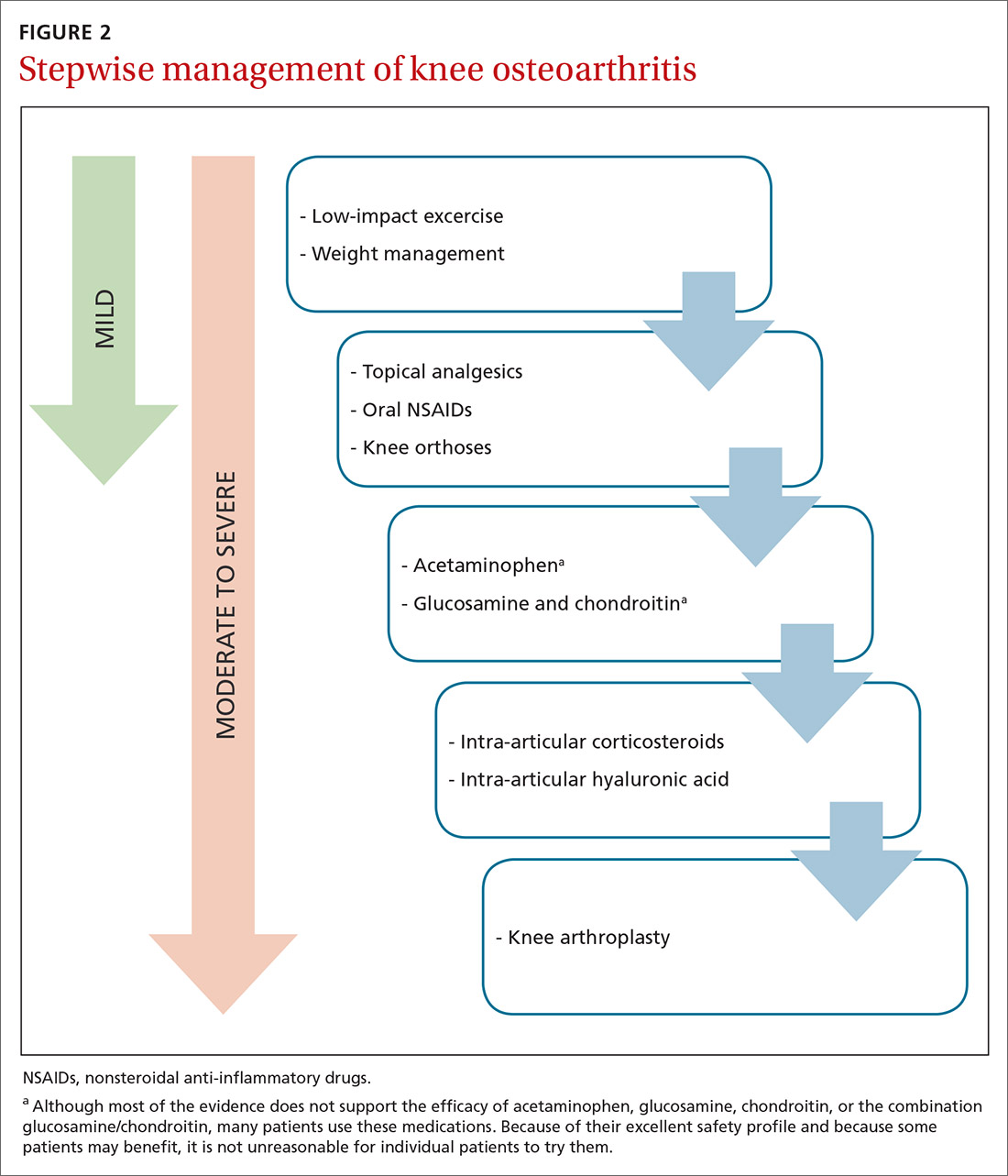
Weight management can slow progression of the disease
Obesity is a causative factor in knee OA.12,13 Patients with knee OA who achieve and maintain an appropriate body weight can potentially slow progression of the disease.13,14 One pound of weight loss can lead to a 4-fold reduction in the load exerted on the knee per step.15
Specific methods of weight reduction are beyond the scope of this article; however, one randomized controlled trial (RCT) involving 399 overweight and obese adults with knee OA found that individuals who participated in a dietary intervention or a combined diet and exercise intervention achieved more weight loss than those who undertook exercise alone.16 Additionally, the diet group had greater reductions in knee compression forces compared to the exercise group, and the combined diet and exercise group had less pain and better function than both the diet group and the exercise group.16 This would suggest that both diet and exercise interventions should be employed in the treatment of knee OA, not only for weight management, but also for knee joint health.
What kind of exercise? Evidence exists to support the utilization of various forms of exercise. In general, land-based therapeutic exercise improves knee pain, physical function, and quality of life, but these benefits often last less than 1 year because people often fail to maintain exercise programs for the long term.17
Specific therapies such as yoga, Tai Chi, balance training, and aquatic exercise have shown some minor improvement in symptoms related to knee OA.18-22 Weight-bearing strength training, non–weight-bearing strength training, and aerobic exercise have all been shown to be effective for short-term pain relief in knee OA, with non–weight-bearing strength training being the most effective.23
Continue to: Strengthening of the upper leg muscles...
Strengthening of the upper leg muscles is thought to be one of the factors involved in reducing pain associated with knee OA.24 Strength training, Tai Chi, and aerobic exercise have also been shown to decrease fall risk in the elderly with knee OA.25 In general, lower impact activities (eg, walking, swimming, biking, yoga) are preferred over higher impact activities (eg, running, jumping) in order to lessen pain with exercise.26-28
Knee orthoses: Many forms and mixed findings
Knee braces come in many forms, including soft braces (eg, elastic sleeves, simple hinged braces) and unloading braces. Many of these braces have been purported to help with knee OA although the evidence remains mixed, with a lack of high-quality trials. A systematic review of RCTs comparing various knee braces, foot orthotics, and conservative treatment for the management of medial compartment OA concluded that the optimal choice for orthosis remains unclear, and long-term evidence is lacking.29
The medial unloading (valgus) knee brace is often used to treat medial compartment OA and varus malalignment of the knee by applying a valgus force, thereby reducing the load on the medial compartment. One recent systematic review concluded that medial unloading braces improve pain from medial compartment OA, but whether they improve function and stiffness is unclear.30 Another study showed that compared to conservative treatment alone, valgus knee bracing has some benefit in decreasing pain and improving knee function.31 Additionally, an 8-year prospective study found that the valgus unloading brace can delay the time before patients need to undergo knee arthroplasty.32 However, another prospective study examining the efficacy of valgus bracing at 2.7 years and 11.2 years showed short-term but not long-term benefit.33
Soft knee braces include a variety of elastic sleeves and simple hinged knee braces. These braces are available commercially at most pharmacies and athletic retail stores. Soft braces are thought to improve pain by a thermal and compressive effect, and to provide stability to the knee joint. One systematic review concluded that soft knee braces have a moderate effect on pain and a small-to-moderate effect on self-reported physical function.34 A small trial showed that soft knee braces reduced pain and dynamic instability in individuals with knee OA.35
In summary, many types of soft knee braces exist, but the evidence for recommending them individually or collectively is limited, as high-quality trials are lacking. However, the available evidence does suggest some mild benefit with regard to pain and function with no concern for adverse effects.
Continue to: Pharmacotherapy
Pharmacotherapy: Oral agents
Acetaminophen. Although people commonly use this over-the-counter analgesic for knee OA pain, recent meta-analyses have shown that acetaminophen provides little to no benefit.36,37 Furthermore, although many believe acetaminophen causes fewer adverse effects than oral nonsteroidal anti-inflammatory drugs (NSAIDs), liver, gastrointestinal, and renal complications are not uncommon with long-term acetaminophen use. Nevertheless, a trial of acetaminophen may be beneficial in patients with cardiovascular disease or who are taking oral anticoagulants.
Oral NSAIDs. Many studies have concluded that NSAIDs are more effective at controlling pain from knee OA than acetaminophen.37,38 They are among the most commonly prescribed treatments for knee OA, but patients and their physicians should be cautious about long-term use because of potential cardiac, renal, gastrointestinal, and other adverse effects. Although evidence regarding optimal frequency of use is scarce, oral NSAIDs should be used intermittently and at the minimal effective dose in order to decrease the risk of adverse events.
One recent meta-analysis of RCTs concluded that diclofenac at a dose of 150 mg/d is the most effective NSAID for improving pain and function associated with knee OA.37 Another recent systematic review and meta-analysis analyzing multiple pharmacologic treatments found an association between celecoxib and decreased pain from knee OA.39 However, this study also concluded that uncertainty surrounded all of the estimates of effect size for change in pain compared to placebo for all of the pharmacologic treatments included in the study.39
A meta-analysis of RCTs comparing celecoxib to no treatment, placebo, naproxen, and diclofenac concluded that celecoxib is slightly better than placebo and the aforementioned NSAIDs in reducing pain and improving function in general OA. However, the authors had reservations regarding pharmaceutical industry involvement in the studies and overall limited data.40
With all of that said, the American Academy of Orthopaedic Surgeons (AAOS) recommends strongly for the use of oral NSAIDs in the management of knee OA.41
Continue to: Glucosamine and chondroitin
Glucosamine and chondroitin. Glucosamine and chondroitin are supplements that have gained popularity in the treatment of knee OA. These constituents are found naturally in articular cartilage, which explains the rationale for their use. Glucosamine and chondroitin (or a combination of the 2) are associated with few adverse effects, but the evidence to support their use in knee OA management is mixed.
One large double-blind RCT (the Glucosamine/Chondroitin Arthritis Intervention Trial [GAIT]) concluded that glucosamine, chondroitin, or the combination of the 2 did not have a significant effect on reducing pain from knee OA compared to placebo and did not slow structural joint disease.42 However, this same study found that in a subset of patients with moderate-to-severe knee OA, the combination of glucosamine and chondroitin was mildly effective in reducing pain.42
Multiple studies have shown either no benefit, inconsistent results, or limited benefit of glucosamine and chondroitin in the treatment of knee OA, with the patented crystalline form of glucosamine showing the most efficacy.43-47 The AAOS and the American College of Rheumatology (ACR) do not recommend glucosamine and chondroitin for knee OA management.10,41
In summary, the evidence for glucosamine, chondroitin, or a combination of the 2 for knee OA is mixed with likely limited benefit, but because they are associated with few adverse effects, patients may be offered a 3- to 6-month trial of these supplements if other effective options are exhausted.
Injections
Limited-quality evidence suggests that oral NSAIDs and intra-articular (IA) hyaluronic acid (HA) injections are equally efficacious for knee OA pain.38,48 There is insufficient evidence directly comparing oral NSAIDs with IA corticosteroid (CS) injections.
Continue to: HA is found naturally...
HA is found naturally in articular cartilage, which explains the rationale behind its use. A network meta-analysis performed by the American Medical Society for Sports Medicine concluded that knee OA is more likely to respond to IAHA than to IACS or IA placebo, leading the society to recommend the use of IAHA in knee OA management, especially for patients > 60 years with mild-to-moderate knee OA.9 Conversely, the AAOS does not recommend the use of IAHA, and the ACR does not recommend for or against the use of IAHA.10,41
IACSs are commonly used to provide pain relief in those with moderate-to-severe knee OA. There is evidence that a single IACS injection provides mild pain relief for up to 6 weeks.49 However, there is some concern that repetitive IACS injections may speed cartilage loss. A 2-year randomized double-blind placebo-controlled trial comparing the effectiveness of repetitive IA triamcinolone vs saline in knee OA found no difference in pain severity and concluded that there was greater cartilage volume loss in the triamcinolone group.50
AAOS does not recommend for or against the use of IACSs, whereas the ACR does recommend for the use of IACSs.10,41 Given the available evidence, conservative use of IACS injections remains an option for patients with refractory moderate-to-severe knee OA.
Topicals
Topical analgesics are often utilized for knee OA because of their efficacy, tolerability, low risk of adverse effects, and ease of use. They are generally recommended over oral NSAIDs in the elderly and in individuals at risk for cardiac, renal, and gastrointestinal complications from oral NSAIDs.
One review found that topical diclofenac and topical ketoprofen were comparable to the oral forms of these medications.51 One RCT concluded that topical and oral diclofenac were equally efficacious in treating knee OA symptoms, although topical diclofenac was associated with significantly fewer gastrointestinal adverse effects.52 In multiple randomized trials, topical diclofenac has shown efficacy compared to placebo.53-55 A recent systematic review and meta-analysis of RCTs concluded that topical NSAIDs were safe and effective for treating general OA compared to placebo, with diclofenac patches most effective for pain relief and piroxicam most effective for functional improvement.56
Continue to: Topical capsaicin has shown...
Topical capsaicin has shown some efficacy in treating pain associated with knee OA.57 One meta-analysis of RCTs concluded that topical NSAIDs and capsaicin may be equally efficacious for OA-associated pain relief, although none of the RCTs directly compared the two.58 The major limitation of capsaicin is a patient-reported mild-to-moderate burning sensation with application that may decrease compliance.
Emerging treatments: IA PRP & extended-release IA triamcinolone acetonide
IA platelet-rich plasma (PRP) has been investigated for efficacy in treating knee OA. PRP is thought to decrease inflammation in the joint, although its exact mechanism remains unknown.59 Multiple studies have shown some benefit of PRP in reducing pain and improving function in individuals with knee OA, but nearly all of these studies have failed to show a clear benefit of PRP over HA injections.59-63 Additionally, the authors of most of these studies mention a high risk of bias. PRP therapy is expensive and generally is not covered by insurance companies, which precludes its use for many people.
Extended-release (ER) IA triamcinolone acetonide (Zilretta) has shown some superiority to standard IA triamcinolone acetonide in both degree and duration of pain relief for knee OA.64-66 The ER version tolerability did not differ from placebo and also showed prolonged synovial presence, lower systemic absorption, and lower blood glucose elevations compared with standard triamcinolone.64-66
Surgical intervention: A last resort
Select patients with severe pain and disability from knee OA that is refractory to conservative management options should be referred for consideration of knee arthroplasty. Age, weight, OA location, and degree of OA are all considered with respect to knee arthroplasty timing and technique.
There is good evidence that arthroscopy with debridement, on the other hand, is no more effective than conservative management.67
Continue to: Unicompartmental or "partial"...
Unicompartmental or “partial” knee replacements are reserved for select cases when 1 knee compartment has a significantly higher degree of degenerative change.
CASE After reviewing the therapeutic options with your patient, you agree that she will undergo a course of physical therapy and try using topical diclofenac along with a hinged knee brace. Because of the patient’s age and co-morbidities of cardiovascular disease and mild chronic kidney disease, oral NSAIDs are avoided at this time.
The patient returns to the office in 2 months reporting mild improvement in her pain. To provide additional pain relief, an ultrasound-guided IA steroid injection is attempted. The patient also continues home physical therapy, activity modification, topical diclofenac, and use of a hinged knee brace.
She returns to the office 2 months later, reporting continued improvement in her pain. No further intervention is undertaken at this time.
CORRESPONDENCE
Ryan A. Sprouse, MD, CAQSM, West Virginia University School of Medicine–Eastern Campus, WVU Medicine Orthopaedics and Sports Medicine, 912 Somerset Boulevard, Charles Town, WV 25414; [email protected].
1. Wallace IJ, Worthington S,Felson DT, et al. Knee osteoarthritis has doubled in prevalence since the mid-20th century. Proc Natl Acad Sci. 2017;114:9332-9336.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35.
3. Vina ER, Kwoh CK. Epidemiology of osteoarthritis: literature update. Curr Opin Rheumatol. 2018;30:160-167.
4. Warner SC, Valdes AM. Genetic association studies in osteoarthritis: is it fairytale? Curr Opin Rheumatol. 2017;29:103-109.
5. Srikanth VK, Fryer JL, Zhai G, et al. A meta-analysis of sex differences prevalence, incidence and severity of osteoarthritis. Osteoarthritis Cartilage. 2005;13:769-781.
6. Palazzo C, Nguyen C, Lefevre-Colau MM, et al. Risk factors and burden of osteoarthritis. Ann Phys Rehabil Med. 2016;59:134-138.
7. Tanamas S, Hanna FS, Cicuttini FM, et al. Does knee malalignment increase the risk of development and progression of knee osteoarthritis? A systematic review. Arthritis Rheum. 2009;61:459-467.
8. Yucesoy B, Charles LE, Baker B, et al. Occupational and genetic risk factors for osteoarthritis: a review. Work. 2015;50:261-273.
9. Trojian TH, Concoff AL, Joy SM, et al. AMSSM scientific statement concerning viscosupplementation injections for knee osteoarthritis: importance for individual patient outcomes. Br J Sports Med. 2016;50:84-92.
10. Hochberg MC, Altman RD, April KT, et al. American College of Rheumatology 2012 Recommendations for the Use of Nonpharmacologic and Pharmacologic Therapies in Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res. 2012;64:465-474.
11. Bedson J, Croft PR. The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord. 2008;9:116.
12. Felson DT, Anderson JJ, Naimark A, et al. Obesity and knee osteoarthritis. The Framingham Study. Ann Intern Med. 1988;109:18-24.
13. Yusuf E, Bijsterbosch J, Slagboom PE, et al. Body mass index and alignment and their interaction as risk factors for progression of knees with radiographic signs of osteoarthritis. Osteoarthritis Cartilage. 2011;19:1117-1122.
14. Niu J, Zhang YQ, Torner J, et al. Is obesity a risk factor for progressive radiographic knee osteoarthritis? Arthritis Rheum. 2009;61:329-335.
15. Messier SP, Gutekunst DJ, Davis C, et al. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005;52:2026-2032.
16. Messier SP, Mihalko SL, Legault C, et al. Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis: the IDEA randomized clinical trial. JAMA. 2013;310:1263-1273.
17. Fransen M, McConnell S, Harmer AR, et al. Exercise for osteoarthritis of the knee: a Cochrane systematic review. Br J Sports Med.
18. Kan L, Zhang J, Yang Y, et al. The effects of yoga on pain, mobility, and quality of life in patients with knee osteoarthritis: a systematic review. Evid Based Complement Alternat Med. 2016;2016:6016532.
19. Chang WD, Chen S, Lee CL, et al. The effects of tai chi chuan on improving mind-body health for knee osteoarthritis patients: a systematic review and meta-analysis. Evid Based Complement Alternat Med. 2016;2016:1813979.
20. Takacs J, Krowchuk NM, Garland SJ, et al. Dynamic balance training improves physical function in individuals with knee osteoarthritis: a pilot randomized controlled trial. Arch Phys Med Rehabil. 2017;98:1586-1593.
21. Bartels EM, Juhl CB, Christensen R, et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev. 2016;(3):CD005523.
22. Hinman RS, Heywood SE, Day AR. Aquatic physical therapy for hip and knee osteoarthritis: results of a single-blind randomized controlled trial. Phys Ther. 2007;87:32-43.
23. Tanaka R, Ozawa J, Kito N, et al. Efficacy of strengthening or aerobic exercise on pain relief in people with knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Clin Rehabil. 2013;27:1059-1071.
24. Knoop J, Steultjens MP, Roorda LD, et al. Improvement in upper leg muscle strength underlies beneficial effects of exercise therapy in knee osteoarthritis: secondary analysis from a randomised controlled trial. Physiotherapy. 2015;101:171-177.
25. Mat S, Tan MP, Kamaruzzaman SB, et al. Physical therapies for improving balance and reducing falls risk in osteoarthritis of the knee: a systematic review. Age Ageing. 2015;44:16-24.
26. Peeler J, Christian M, Cooper J, et al. Managing knee osteoarthritis: the effects of body weight supported physical activity on joint pain, function, and thigh muscle strength. Clin J Sport Med. 2015;25:518-523.
27. Peeler J, Ripat J. The effect of low-load exercise on joint pain, function, and activities of daily living in patients with knee osteoarthritis. Knee. 2018;25:135-145.
28. Takacs J, Anderson JE, Leiter JR, et al. Lower body positive pressure: an emerging technology in the battle against knee osteoarthritis? Clin Interv Aging. 2013;8:983-991.
29. Duivenvoorden T, Brouwer RW, van Raaij TM, et al. Braces and orthoses for treating osteoarthritis of the knee. Cochrane Database Syst Rev. 2015;(3):CD004020.
30. Gohal C, Shanmugaraj A, Tate P, et al. Effectiveness of valgus offloading knee braces in the treatment of medial compartment knee osteoarthritis: a systematic review. Sports Health. 2018;10:500-514.
31. Brouwer RW, van Raaij TM, Verhaar JA, et al. Brace treatment for osteoarthritis of the knee: a prospective randomized multi-centre trial. Osteoarthritis Cartilage. 2006;14:777-783.
32. Lee PY, Winfield TG, Harris SR, et al. Unloading knee brace is a cost-effective method to bridge and delay surgery in unicompartmental knee arthritis. BMJ Open Sport Exerc Med. 2017;2:e000195.
33. Wilson B, Rankin H, Barnes CL. Long-term results of an unloader brace in patients with unicompartmental knee osteoarthritis. Orthopedics. 2011;34:334-347.
34. Cudejko T, van der Esch M, van der Leeden M, et al. Effect of soft braces on pain and physical function in patients with knee osteoarthritis: systematic review with meta-analyses. Arch Phys Med Rehabil. 2018;99:153-163.
35. Cudejko T, van der Esch M, van den Noort JC. Decreased pain and improved dynamic knee instability mediate the beneficial effect of wearing a soft knee brace on activity limitations in persons with knee osteoarthritis. Arthritis Care Res (Hoboken). 2019;71:1036-1043.
36. Machado GC, Maher CG, Ferreira PH, et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ. 2015;350:h1225.
37. da Costa BR, Reichenbach S, Keller N, et al. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: a network meta-analysis. Lancet. 2017;390:e21-e33.
38. Bannuru RR, Schmid CH, Kent DM, et al. Comparative effectiveness of pharmacologic interventions for knee osteoarthritis: a systematic review and network meta-analysis. Ann Intern Med. 2015;162:46-54.
39. Gregori D, Giacovelli G, Minto C, et al. Association of pharmacological treatments with long-term pain control in patients with knee osteoarthritis: a systematic review and meta-analysis. JAMA. 2018;320:2564-2579.
40. Puljak L, Marin A, Vrdoljak D, et al. Celecoxib for osteoarthritis. Cochrane Database Syst Rev. 2017;(5):CD009865.
41. Jevsevar DS. Treatment of osteoarthritis of the knee: evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013;9:571-576.
42. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
43. Singh JA, Noorbaloochi S, MacDonald R, et al. Chondroitin for osteoarthritis. Cochrane Database Syst Rev. 2015;(1):CD005614.
44. Yang S, Eaton CB, McAlindon TE, et al. Effects of glucosamine and chondroitin on treating knee osteoarthritis: an analysis with marginal structural models. Arthritis Rheumatol. 2015;67:714-723.
45. Ogata T, Yuki Ideno Y, Masami Akai M,et al. Effects of glucosamine in patients with osteoarthritis of the knee: a systematic review and meta-analysis. Clin Rheumatol. 2018;37:2479-2487.
46. Towheed TE, Maxwell L, Anastassiades TP, et al. Glucosamine therapy for treating osteoarthritis. Cochrane Database Syst Rev. 2009;(2):CD002946.
47. Bruyèreetal O, Cooper C, Pelletier JP, et al. A consensus statement on the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) algorithm for the management of knee osteoarthritis—from evidence-based medicine to the real-life setting. Semin Arthritis Rheum. 2016;45(4 suppl):S3-S11.
48. Ishijima M, Nakamura T, Shimizu K, et al. Intra-articular hyaluronic acid injection versus oral non-steroidal anti-inflammatory drug for the treatment of knee osteoarthritis: a multi-center, randomized, open-label, non-inferiority trial. Arthritis Res Ther. 2014;16:R18.
49. Juni P, Hari R, Rutjes AW, et al. Intra-articular corticosteroid for knee osteoarthritis. Cochrane Database Syst Rev. 2015;(10):CD005328.
50. McAlindon TE, LaValley MP, Harvey FW, et al. Effect of intra-articular triamcinolone vs saline on knee cartilage volume and pain in patients with knee osteoarthritis: a randomized clinical trial. JAMA. 2017;317:1967-1975.
51. Derry S, Conaghan P, Da Silva JA, et al. Topical NSAIDs for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2016;(4):CD007400.
52. Tugwell PS, Wells GA, Shainhouse JZ. Equivalence study of a topical diclofenac solution (pennsaid) compared with oral diclofenac in symptomatic treatment of osteoarthritis of the knee: a randomized controlled trial. J Rheumatol. 2004;31:2002-2012.
53. Wadsworth LT, Kent JD, Holt RJ. Efficacy and safety of diclofenac sodium 2% topical solution for osteoarthritis of the knee: a randomized, double-blind, vehicle-controlled, 4 week study. Curr Med Res Opin. 2016;32:241-250.
54. Roth SH, Shainhouse JZ. Efficacy and safety of a topical diclofenac solution (pennsaid) in the treatment of primary osteoarthritis of the knee: a randomized, double-blind, vehicle-controlled clinical trial. Arch Intern Med. 2004;164:2017-2023.
55. Baer PA, Thomas LM, Shainhouse Z. Treatment of osteoarthritis of the knee with a topical diclofenac solution: a randomised controlled, 6-week trial. BMC Musculoskelet Disord. 2005;6:44.
56. Zeng C, Wei J, Persson MSM, et al. Relative efficacy and safety of topical non-steroidal anti-inflammatory drugs for osteoarthritis: a systematic review and network meta-analysis of randomised controlled trials and observational studies. Br J Sports Med. 2018;52:642-650.
57. Guedes V, Castro JP, Brito I. Topical capsaicin for pain in osteoarthritis: a literature review. Reumatol Clin. 2018;14:40-45.
58. Persson MSM, Stocks J, Walsh DA, et al. The relative efficacy of topical non-steroidal anti-inflammatory drugs and capsaicin in osteoarthritis: a network meta-analysis of randomised controlled trials. Osteoarthritis Cartilage. 2018;26:1575-1582.
59. Cole BJ, Karas V, Hussey K, et al. Hyaluronic acid versus platelet-rich plasma: a prospective, double-blind randomized controlled trial comparing clinical outcomes and effects on intra-articular biology for the treatment of knee osteoarthritis. Am J Sports Med. 2017;45:339-346.
60. Laudy AB, Bakker EW, Rekers M, et al. Efficacy of platelet-rich plasma injections in osteoarthritis of the knee: a systematic review and meta-analysis. Br J Sports Med. 2015;49:657-672.
61. Han Y, Huang H, Pan J, et al. Meta-analysis comparing platelet-rich plasma vs hyaluronic acid injection in patients with knee osteoarthritis. Pain Med. 2019;20:1418-1429.
62. Filardo G, Di Matteo B, Di Martino A, et al. Platelet-rich plasma intra-articular knee injections show no superiority versus viscosupplementation: a randomized controlled trial. Am J Sports Med. 2015;43:1575-1582.
63. Di Martino A, Di Matteo B, Papio T, et al. Platelet-rich plasma versus hyaluronic acid injections for the treatment of knee osteoarthritis: results at 5 years of a double-blind, randomized controlled trial. Am J Sports Med. 2019;47:347-354.
64. Bodick N, Lufkin J, Willwerth C, et al. An intra-articular, extended-release formulation of triamcinolone acetonide prolongs and amplifies analgesic effect in patients with osteoarthritis of the knee: a randomized clinical trial. J Bone Joint Surg Am. 2015;97:877-888.
65. Conaghan PG, Cohen SB, Berenbaum F, et al. Brief report: a phase IIb trial of a novel extended-release microsphere formulation of triamcinolone acetonide for intraarticular injection in knee osteoarthritis. Arthritis Rheumatol. 2018;70:204-211.
66. Conaghan PG, Hunter DJ, Cohen SB, et al. Effects of a single intra-articular injection of a microsphere formulation of triamcinolone acetonide on knee osteoarthritis pain: a double-blinded, randomized, placebo-controlled, multinational study. J Bone Jt Surg Am. 2018;100:666–677.
67. Thorlund JB, Juhl CB, Roos EM, et al. Arthroscopic surgery for degenerative knee: systematic review and meta-analysis of benefits and harms. BMJ. 2015;350:h2747.
CASE A 73-year-old woman presents to your clinic with 1 year of gradual-onset left knee pain. The pain is worse at the medial knee and at the beginning and end of the day, with some mild improvement after activity in the morning. The patient has already tried oral acetaminophen, an over-the-counter menthol cream, and a soft elastic knee brace, but these interventions have helped only minimally.
On physical exam, there is no obvious deformity of the knee. There is a bit of small joint effusion without redness or warmth. There is mild tenderness to palpation of the medial joint line. Radiographic findings include osteophytes of the medial and lateral tibial plateaus and medial and lateral femoral condyles with mild joint-space narrowing of the medial compartment, consistent with mild osteoarthritis.
How would you manage this patient’s care?
The knee is the most common joint to be affected by osteoarthritis (OA) and accounts for the majority of the disease’s total burden.1 More than 19% of American adults ages ≥ 45 years have knee OA,1,2 and more than half of the people with symptomatic knee OA in the United States are younger than 65 years of age.3 Longer lifespan and increasing rates of obesity are thought to be driving the increasing prevalence of knee OA, although this remains debated.1 Risk factors for knee OA are outlined in TABLE.1,4-8

Diagnosis: Radiographs are helpful, not essential
The diagnosis of knee OA is relatively straightforward. Gradual onset of knee joint pain is present most days, with pain worse after activity and better with rest. Patients are usually middle-aged or older and/or have a distant history of knee joint injury. Other signs, symptoms, and physical exam findings associated with knee OA include: morning stiffness < 30 minutes, crepitus, instability, range-of-motion deficit, varus or valgus deformity, bony exostosis, joint-line tenderness, joint swelling/effusion, and the absence of erythema/warmth.1,9,10
Although radiographs are not necessary to diagnose knee OA, they can be helpful in confirming the diagnosis by assessing the degree and location of OA and ruling out other pathology. Standing, weight-bearing radiographs are particularly helpful for assessing the degree of joint-space narrowing. In addition to joint-space narrowing, radiographic findings indicative of knee OA include marginal osteophytes, subchondral sclerosis, and subchondral cysts. (See FIGURE 1.)

Keep in mind that radiographs are less sensitive for early OA, that the degree of OA seen on radiographs does not correlate well with symptoms, and that radiographic evidence of OA is a common incidental finding—especially in elderly individuals.11 Although not routinely utilized for knee OA diagnosis, magnetic resonance imaging (MRI) can be used to assess for earlier stages of the disease and to rule out pathology associated with the soft tissue and cartilage that is not directly associated with OA.
Continue to: Management
Management: Decrease pain, improve function, slow progression
Because there is no cure for OA, the primary goals of treatment are to decrease pain, improve function of the joint, and slow progression of the disease. As a result, a multifaceted treatment approach is usually undertaken that includes weight reduction and exercise therapy and may include pharmacotherapy, depending on the degree of symptoms. FIGURE 2 contains a summary of the stepwise management of knee OA.

Weight management can slow progression of the disease
Obesity is a causative factor in knee OA.12,13 Patients with knee OA who achieve and maintain an appropriate body weight can potentially slow progression of the disease.13,14 One pound of weight loss can lead to a 4-fold reduction in the load exerted on the knee per step.15
Specific methods of weight reduction are beyond the scope of this article; however, one randomized controlled trial (RCT) involving 399 overweight and obese adults with knee OA found that individuals who participated in a dietary intervention or a combined diet and exercise intervention achieved more weight loss than those who undertook exercise alone.16 Additionally, the diet group had greater reductions in knee compression forces compared to the exercise group, and the combined diet and exercise group had less pain and better function than both the diet group and the exercise group.16 This would suggest that both diet and exercise interventions should be employed in the treatment of knee OA, not only for weight management, but also for knee joint health.
What kind of exercise? Evidence exists to support the utilization of various forms of exercise. In general, land-based therapeutic exercise improves knee pain, physical function, and quality of life, but these benefits often last less than 1 year because people often fail to maintain exercise programs for the long term.17
Specific therapies such as yoga, Tai Chi, balance training, and aquatic exercise have shown some minor improvement in symptoms related to knee OA.18-22 Weight-bearing strength training, non–weight-bearing strength training, and aerobic exercise have all been shown to be effective for short-term pain relief in knee OA, with non–weight-bearing strength training being the most effective.23
Continue to: Strengthening of the upper leg muscles...
Strengthening of the upper leg muscles is thought to be one of the factors involved in reducing pain associated with knee OA.24 Strength training, Tai Chi, and aerobic exercise have also been shown to decrease fall risk in the elderly with knee OA.25 In general, lower impact activities (eg, walking, swimming, biking, yoga) are preferred over higher impact activities (eg, running, jumping) in order to lessen pain with exercise.26-28
Knee orthoses: Many forms and mixed findings
Knee braces come in many forms, including soft braces (eg, elastic sleeves, simple hinged braces) and unloading braces. Many of these braces have been purported to help with knee OA although the evidence remains mixed, with a lack of high-quality trials. A systematic review of RCTs comparing various knee braces, foot orthotics, and conservative treatment for the management of medial compartment OA concluded that the optimal choice for orthosis remains unclear, and long-term evidence is lacking.29
The medial unloading (valgus) knee brace is often used to treat medial compartment OA and varus malalignment of the knee by applying a valgus force, thereby reducing the load on the medial compartment. One recent systematic review concluded that medial unloading braces improve pain from medial compartment OA, but whether they improve function and stiffness is unclear.30 Another study showed that compared to conservative treatment alone, valgus knee bracing has some benefit in decreasing pain and improving knee function.31 Additionally, an 8-year prospective study found that the valgus unloading brace can delay the time before patients need to undergo knee arthroplasty.32 However, another prospective study examining the efficacy of valgus bracing at 2.7 years and 11.2 years showed short-term but not long-term benefit.33
Soft knee braces include a variety of elastic sleeves and simple hinged knee braces. These braces are available commercially at most pharmacies and athletic retail stores. Soft braces are thought to improve pain by a thermal and compressive effect, and to provide stability to the knee joint. One systematic review concluded that soft knee braces have a moderate effect on pain and a small-to-moderate effect on self-reported physical function.34 A small trial showed that soft knee braces reduced pain and dynamic instability in individuals with knee OA.35
In summary, many types of soft knee braces exist, but the evidence for recommending them individually or collectively is limited, as high-quality trials are lacking. However, the available evidence does suggest some mild benefit with regard to pain and function with no concern for adverse effects.
Continue to: Pharmacotherapy
Pharmacotherapy: Oral agents
Acetaminophen. Although people commonly use this over-the-counter analgesic for knee OA pain, recent meta-analyses have shown that acetaminophen provides little to no benefit.36,37 Furthermore, although many believe acetaminophen causes fewer adverse effects than oral nonsteroidal anti-inflammatory drugs (NSAIDs), liver, gastrointestinal, and renal complications are not uncommon with long-term acetaminophen use. Nevertheless, a trial of acetaminophen may be beneficial in patients with cardiovascular disease or who are taking oral anticoagulants.
Oral NSAIDs. Many studies have concluded that NSAIDs are more effective at controlling pain from knee OA than acetaminophen.37,38 They are among the most commonly prescribed treatments for knee OA, but patients and their physicians should be cautious about long-term use because of potential cardiac, renal, gastrointestinal, and other adverse effects. Although evidence regarding optimal frequency of use is scarce, oral NSAIDs should be used intermittently and at the minimal effective dose in order to decrease the risk of adverse events.
One recent meta-analysis of RCTs concluded that diclofenac at a dose of 150 mg/d is the most effective NSAID for improving pain and function associated with knee OA.37 Another recent systematic review and meta-analysis analyzing multiple pharmacologic treatments found an association between celecoxib and decreased pain from knee OA.39 However, this study also concluded that uncertainty surrounded all of the estimates of effect size for change in pain compared to placebo for all of the pharmacologic treatments included in the study.39
A meta-analysis of RCTs comparing celecoxib to no treatment, placebo, naproxen, and diclofenac concluded that celecoxib is slightly better than placebo and the aforementioned NSAIDs in reducing pain and improving function in general OA. However, the authors had reservations regarding pharmaceutical industry involvement in the studies and overall limited data.40
With all of that said, the American Academy of Orthopaedic Surgeons (AAOS) recommends strongly for the use of oral NSAIDs in the management of knee OA.41
Continue to: Glucosamine and chondroitin
Glucosamine and chondroitin. Glucosamine and chondroitin are supplements that have gained popularity in the treatment of knee OA. These constituents are found naturally in articular cartilage, which explains the rationale for their use. Glucosamine and chondroitin (or a combination of the 2) are associated with few adverse effects, but the evidence to support their use in knee OA management is mixed.
One large double-blind RCT (the Glucosamine/Chondroitin Arthritis Intervention Trial [GAIT]) concluded that glucosamine, chondroitin, or the combination of the 2 did not have a significant effect on reducing pain from knee OA compared to placebo and did not slow structural joint disease.42 However, this same study found that in a subset of patients with moderate-to-severe knee OA, the combination of glucosamine and chondroitin was mildly effective in reducing pain.42
Multiple studies have shown either no benefit, inconsistent results, or limited benefit of glucosamine and chondroitin in the treatment of knee OA, with the patented crystalline form of glucosamine showing the most efficacy.43-47 The AAOS and the American College of Rheumatology (ACR) do not recommend glucosamine and chondroitin for knee OA management.10,41
In summary, the evidence for glucosamine, chondroitin, or a combination of the 2 for knee OA is mixed with likely limited benefit, but because they are associated with few adverse effects, patients may be offered a 3- to 6-month trial of these supplements if other effective options are exhausted.
Injections
Limited-quality evidence suggests that oral NSAIDs and intra-articular (IA) hyaluronic acid (HA) injections are equally efficacious for knee OA pain.38,48 There is insufficient evidence directly comparing oral NSAIDs with IA corticosteroid (CS) injections.
Continue to: HA is found naturally...
HA is found naturally in articular cartilage, which explains the rationale behind its use. A network meta-analysis performed by the American Medical Society for Sports Medicine concluded that knee OA is more likely to respond to IAHA than to IACS or IA placebo, leading the society to recommend the use of IAHA in knee OA management, especially for patients > 60 years with mild-to-moderate knee OA.9 Conversely, the AAOS does not recommend the use of IAHA, and the ACR does not recommend for or against the use of IAHA.10,41
IACSs are commonly used to provide pain relief in those with moderate-to-severe knee OA. There is evidence that a single IACS injection provides mild pain relief for up to 6 weeks.49 However, there is some concern that repetitive IACS injections may speed cartilage loss. A 2-year randomized double-blind placebo-controlled trial comparing the effectiveness of repetitive IA triamcinolone vs saline in knee OA found no difference in pain severity and concluded that there was greater cartilage volume loss in the triamcinolone group.50
AAOS does not recommend for or against the use of IACSs, whereas the ACR does recommend for the use of IACSs.10,41 Given the available evidence, conservative use of IACS injections remains an option for patients with refractory moderate-to-severe knee OA.
Topicals
Topical analgesics are often utilized for knee OA because of their efficacy, tolerability, low risk of adverse effects, and ease of use. They are generally recommended over oral NSAIDs in the elderly and in individuals at risk for cardiac, renal, and gastrointestinal complications from oral NSAIDs.
One review found that topical diclofenac and topical ketoprofen were comparable to the oral forms of these medications.51 One RCT concluded that topical and oral diclofenac were equally efficacious in treating knee OA symptoms, although topical diclofenac was associated with significantly fewer gastrointestinal adverse effects.52 In multiple randomized trials, topical diclofenac has shown efficacy compared to placebo.53-55 A recent systematic review and meta-analysis of RCTs concluded that topical NSAIDs were safe and effective for treating general OA compared to placebo, with diclofenac patches most effective for pain relief and piroxicam most effective for functional improvement.56
Continue to: Topical capsaicin has shown...
Topical capsaicin has shown some efficacy in treating pain associated with knee OA.57 One meta-analysis of RCTs concluded that topical NSAIDs and capsaicin may be equally efficacious for OA-associated pain relief, although none of the RCTs directly compared the two.58 The major limitation of capsaicin is a patient-reported mild-to-moderate burning sensation with application that may decrease compliance.
Emerging treatments: IA PRP & extended-release IA triamcinolone acetonide
IA platelet-rich plasma (PRP) has been investigated for efficacy in treating knee OA. PRP is thought to decrease inflammation in the joint, although its exact mechanism remains unknown.59 Multiple studies have shown some benefit of PRP in reducing pain and improving function in individuals with knee OA, but nearly all of these studies have failed to show a clear benefit of PRP over HA injections.59-63 Additionally, the authors of most of these studies mention a high risk of bias. PRP therapy is expensive and generally is not covered by insurance companies, which precludes its use for many people.
Extended-release (ER) IA triamcinolone acetonide (Zilretta) has shown some superiority to standard IA triamcinolone acetonide in both degree and duration of pain relief for knee OA.64-66 The ER version tolerability did not differ from placebo and also showed prolonged synovial presence, lower systemic absorption, and lower blood glucose elevations compared with standard triamcinolone.64-66
Surgical intervention: A last resort
Select patients with severe pain and disability from knee OA that is refractory to conservative management options should be referred for consideration of knee arthroplasty. Age, weight, OA location, and degree of OA are all considered with respect to knee arthroplasty timing and technique.
There is good evidence that arthroscopy with debridement, on the other hand, is no more effective than conservative management.67
Continue to: Unicompartmental or "partial"...
Unicompartmental or “partial” knee replacements are reserved for select cases when 1 knee compartment has a significantly higher degree of degenerative change.
CASE After reviewing the therapeutic options with your patient, you agree that she will undergo a course of physical therapy and try using topical diclofenac along with a hinged knee brace. Because of the patient’s age and co-morbidities of cardiovascular disease and mild chronic kidney disease, oral NSAIDs are avoided at this time.
The patient returns to the office in 2 months reporting mild improvement in her pain. To provide additional pain relief, an ultrasound-guided IA steroid injection is attempted. The patient also continues home physical therapy, activity modification, topical diclofenac, and use of a hinged knee brace.
She returns to the office 2 months later, reporting continued improvement in her pain. No further intervention is undertaken at this time.
CORRESPONDENCE
Ryan A. Sprouse, MD, CAQSM, West Virginia University School of Medicine–Eastern Campus, WVU Medicine Orthopaedics and Sports Medicine, 912 Somerset Boulevard, Charles Town, WV 25414; [email protected].
CASE A 73-year-old woman presents to your clinic with 1 year of gradual-onset left knee pain. The pain is worse at the medial knee and at the beginning and end of the day, with some mild improvement after activity in the morning. The patient has already tried oral acetaminophen, an over-the-counter menthol cream, and a soft elastic knee brace, but these interventions have helped only minimally.
On physical exam, there is no obvious deformity of the knee. There is a bit of small joint effusion without redness or warmth. There is mild tenderness to palpation of the medial joint line. Radiographic findings include osteophytes of the medial and lateral tibial plateaus and medial and lateral femoral condyles with mild joint-space narrowing of the medial compartment, consistent with mild osteoarthritis.
How would you manage this patient’s care?
The knee is the most common joint to be affected by osteoarthritis (OA) and accounts for the majority of the disease’s total burden.1 More than 19% of American adults ages ≥ 45 years have knee OA,1,2 and more than half of the people with symptomatic knee OA in the United States are younger than 65 years of age.3 Longer lifespan and increasing rates of obesity are thought to be driving the increasing prevalence of knee OA, although this remains debated.1 Risk factors for knee OA are outlined in TABLE.1,4-8

Diagnosis: Radiographs are helpful, not essential
The diagnosis of knee OA is relatively straightforward. Gradual onset of knee joint pain is present most days, with pain worse after activity and better with rest. Patients are usually middle-aged or older and/or have a distant history of knee joint injury. Other signs, symptoms, and physical exam findings associated with knee OA include: morning stiffness < 30 minutes, crepitus, instability, range-of-motion deficit, varus or valgus deformity, bony exostosis, joint-line tenderness, joint swelling/effusion, and the absence of erythema/warmth.1,9,10
Although radiographs are not necessary to diagnose knee OA, they can be helpful in confirming the diagnosis by assessing the degree and location of OA and ruling out other pathology. Standing, weight-bearing radiographs are particularly helpful for assessing the degree of joint-space narrowing. In addition to joint-space narrowing, radiographic findings indicative of knee OA include marginal osteophytes, subchondral sclerosis, and subchondral cysts. (See FIGURE 1.)

Keep in mind that radiographs are less sensitive for early OA, that the degree of OA seen on radiographs does not correlate well with symptoms, and that radiographic evidence of OA is a common incidental finding—especially in elderly individuals.11 Although not routinely utilized for knee OA diagnosis, magnetic resonance imaging (MRI) can be used to assess for earlier stages of the disease and to rule out pathology associated with the soft tissue and cartilage that is not directly associated with OA.
Continue to: Management
Management: Decrease pain, improve function, slow progression
Because there is no cure for OA, the primary goals of treatment are to decrease pain, improve function of the joint, and slow progression of the disease. As a result, a multifaceted treatment approach is usually undertaken that includes weight reduction and exercise therapy and may include pharmacotherapy, depending on the degree of symptoms. FIGURE 2 contains a summary of the stepwise management of knee OA.

Weight management can slow progression of the disease
Obesity is a causative factor in knee OA.12,13 Patients with knee OA who achieve and maintain an appropriate body weight can potentially slow progression of the disease.13,14 One pound of weight loss can lead to a 4-fold reduction in the load exerted on the knee per step.15
Specific methods of weight reduction are beyond the scope of this article; however, one randomized controlled trial (RCT) involving 399 overweight and obese adults with knee OA found that individuals who participated in a dietary intervention or a combined diet and exercise intervention achieved more weight loss than those who undertook exercise alone.16 Additionally, the diet group had greater reductions in knee compression forces compared to the exercise group, and the combined diet and exercise group had less pain and better function than both the diet group and the exercise group.16 This would suggest that both diet and exercise interventions should be employed in the treatment of knee OA, not only for weight management, but also for knee joint health.
What kind of exercise? Evidence exists to support the utilization of various forms of exercise. In general, land-based therapeutic exercise improves knee pain, physical function, and quality of life, but these benefits often last less than 1 year because people often fail to maintain exercise programs for the long term.17
Specific therapies such as yoga, Tai Chi, balance training, and aquatic exercise have shown some minor improvement in symptoms related to knee OA.18-22 Weight-bearing strength training, non–weight-bearing strength training, and aerobic exercise have all been shown to be effective for short-term pain relief in knee OA, with non–weight-bearing strength training being the most effective.23
Continue to: Strengthening of the upper leg muscles...
Strengthening of the upper leg muscles is thought to be one of the factors involved in reducing pain associated with knee OA.24 Strength training, Tai Chi, and aerobic exercise have also been shown to decrease fall risk in the elderly with knee OA.25 In general, lower impact activities (eg, walking, swimming, biking, yoga) are preferred over higher impact activities (eg, running, jumping) in order to lessen pain with exercise.26-28
Knee orthoses: Many forms and mixed findings
Knee braces come in many forms, including soft braces (eg, elastic sleeves, simple hinged braces) and unloading braces. Many of these braces have been purported to help with knee OA although the evidence remains mixed, with a lack of high-quality trials. A systematic review of RCTs comparing various knee braces, foot orthotics, and conservative treatment for the management of medial compartment OA concluded that the optimal choice for orthosis remains unclear, and long-term evidence is lacking.29
The medial unloading (valgus) knee brace is often used to treat medial compartment OA and varus malalignment of the knee by applying a valgus force, thereby reducing the load on the medial compartment. One recent systematic review concluded that medial unloading braces improve pain from medial compartment OA, but whether they improve function and stiffness is unclear.30 Another study showed that compared to conservative treatment alone, valgus knee bracing has some benefit in decreasing pain and improving knee function.31 Additionally, an 8-year prospective study found that the valgus unloading brace can delay the time before patients need to undergo knee arthroplasty.32 However, another prospective study examining the efficacy of valgus bracing at 2.7 years and 11.2 years showed short-term but not long-term benefit.33
Soft knee braces include a variety of elastic sleeves and simple hinged knee braces. These braces are available commercially at most pharmacies and athletic retail stores. Soft braces are thought to improve pain by a thermal and compressive effect, and to provide stability to the knee joint. One systematic review concluded that soft knee braces have a moderate effect on pain and a small-to-moderate effect on self-reported physical function.34 A small trial showed that soft knee braces reduced pain and dynamic instability in individuals with knee OA.35
In summary, many types of soft knee braces exist, but the evidence for recommending them individually or collectively is limited, as high-quality trials are lacking. However, the available evidence does suggest some mild benefit with regard to pain and function with no concern for adverse effects.
Continue to: Pharmacotherapy
Pharmacotherapy: Oral agents
Acetaminophen. Although people commonly use this over-the-counter analgesic for knee OA pain, recent meta-analyses have shown that acetaminophen provides little to no benefit.36,37 Furthermore, although many believe acetaminophen causes fewer adverse effects than oral nonsteroidal anti-inflammatory drugs (NSAIDs), liver, gastrointestinal, and renal complications are not uncommon with long-term acetaminophen use. Nevertheless, a trial of acetaminophen may be beneficial in patients with cardiovascular disease or who are taking oral anticoagulants.
Oral NSAIDs. Many studies have concluded that NSAIDs are more effective at controlling pain from knee OA than acetaminophen.37,38 They are among the most commonly prescribed treatments for knee OA, but patients and their physicians should be cautious about long-term use because of potential cardiac, renal, gastrointestinal, and other adverse effects. Although evidence regarding optimal frequency of use is scarce, oral NSAIDs should be used intermittently and at the minimal effective dose in order to decrease the risk of adverse events.
One recent meta-analysis of RCTs concluded that diclofenac at a dose of 150 mg/d is the most effective NSAID for improving pain and function associated with knee OA.37 Another recent systematic review and meta-analysis analyzing multiple pharmacologic treatments found an association between celecoxib and decreased pain from knee OA.39 However, this study also concluded that uncertainty surrounded all of the estimates of effect size for change in pain compared to placebo for all of the pharmacologic treatments included in the study.39
A meta-analysis of RCTs comparing celecoxib to no treatment, placebo, naproxen, and diclofenac concluded that celecoxib is slightly better than placebo and the aforementioned NSAIDs in reducing pain and improving function in general OA. However, the authors had reservations regarding pharmaceutical industry involvement in the studies and overall limited data.40
With all of that said, the American Academy of Orthopaedic Surgeons (AAOS) recommends strongly for the use of oral NSAIDs in the management of knee OA.41
Continue to: Glucosamine and chondroitin
Glucosamine and chondroitin. Glucosamine and chondroitin are supplements that have gained popularity in the treatment of knee OA. These constituents are found naturally in articular cartilage, which explains the rationale for their use. Glucosamine and chondroitin (or a combination of the 2) are associated with few adverse effects, but the evidence to support their use in knee OA management is mixed.
One large double-blind RCT (the Glucosamine/Chondroitin Arthritis Intervention Trial [GAIT]) concluded that glucosamine, chondroitin, or the combination of the 2 did not have a significant effect on reducing pain from knee OA compared to placebo and did not slow structural joint disease.42 However, this same study found that in a subset of patients with moderate-to-severe knee OA, the combination of glucosamine and chondroitin was mildly effective in reducing pain.42
Multiple studies have shown either no benefit, inconsistent results, or limited benefit of glucosamine and chondroitin in the treatment of knee OA, with the patented crystalline form of glucosamine showing the most efficacy.43-47 The AAOS and the American College of Rheumatology (ACR) do not recommend glucosamine and chondroitin for knee OA management.10,41
In summary, the evidence for glucosamine, chondroitin, or a combination of the 2 for knee OA is mixed with likely limited benefit, but because they are associated with few adverse effects, patients may be offered a 3- to 6-month trial of these supplements if other effective options are exhausted.
Injections
Limited-quality evidence suggests that oral NSAIDs and intra-articular (IA) hyaluronic acid (HA) injections are equally efficacious for knee OA pain.38,48 There is insufficient evidence directly comparing oral NSAIDs with IA corticosteroid (CS) injections.
Continue to: HA is found naturally...
HA is found naturally in articular cartilage, which explains the rationale behind its use. A network meta-analysis performed by the American Medical Society for Sports Medicine concluded that knee OA is more likely to respond to IAHA than to IACS or IA placebo, leading the society to recommend the use of IAHA in knee OA management, especially for patients > 60 years with mild-to-moderate knee OA.9 Conversely, the AAOS does not recommend the use of IAHA, and the ACR does not recommend for or against the use of IAHA.10,41
IACSs are commonly used to provide pain relief in those with moderate-to-severe knee OA. There is evidence that a single IACS injection provides mild pain relief for up to 6 weeks.49 However, there is some concern that repetitive IACS injections may speed cartilage loss. A 2-year randomized double-blind placebo-controlled trial comparing the effectiveness of repetitive IA triamcinolone vs saline in knee OA found no difference in pain severity and concluded that there was greater cartilage volume loss in the triamcinolone group.50
AAOS does not recommend for or against the use of IACSs, whereas the ACR does recommend for the use of IACSs.10,41 Given the available evidence, conservative use of IACS injections remains an option for patients with refractory moderate-to-severe knee OA.
Topicals
Topical analgesics are often utilized for knee OA because of their efficacy, tolerability, low risk of adverse effects, and ease of use. They are generally recommended over oral NSAIDs in the elderly and in individuals at risk for cardiac, renal, and gastrointestinal complications from oral NSAIDs.
One review found that topical diclofenac and topical ketoprofen were comparable to the oral forms of these medications.51 One RCT concluded that topical and oral diclofenac were equally efficacious in treating knee OA symptoms, although topical diclofenac was associated with significantly fewer gastrointestinal adverse effects.52 In multiple randomized trials, topical diclofenac has shown efficacy compared to placebo.53-55 A recent systematic review and meta-analysis of RCTs concluded that topical NSAIDs were safe and effective for treating general OA compared to placebo, with diclofenac patches most effective for pain relief and piroxicam most effective for functional improvement.56
Continue to: Topical capsaicin has shown...
Topical capsaicin has shown some efficacy in treating pain associated with knee OA.57 One meta-analysis of RCTs concluded that topical NSAIDs and capsaicin may be equally efficacious for OA-associated pain relief, although none of the RCTs directly compared the two.58 The major limitation of capsaicin is a patient-reported mild-to-moderate burning sensation with application that may decrease compliance.
Emerging treatments: IA PRP & extended-release IA triamcinolone acetonide
IA platelet-rich plasma (PRP) has been investigated for efficacy in treating knee OA. PRP is thought to decrease inflammation in the joint, although its exact mechanism remains unknown.59 Multiple studies have shown some benefit of PRP in reducing pain and improving function in individuals with knee OA, but nearly all of these studies have failed to show a clear benefit of PRP over HA injections.59-63 Additionally, the authors of most of these studies mention a high risk of bias. PRP therapy is expensive and generally is not covered by insurance companies, which precludes its use for many people.
Extended-release (ER) IA triamcinolone acetonide (Zilretta) has shown some superiority to standard IA triamcinolone acetonide in both degree and duration of pain relief for knee OA.64-66 The ER version tolerability did not differ from placebo and also showed prolonged synovial presence, lower systemic absorption, and lower blood glucose elevations compared with standard triamcinolone.64-66
Surgical intervention: A last resort
Select patients with severe pain and disability from knee OA that is refractory to conservative management options should be referred for consideration of knee arthroplasty. Age, weight, OA location, and degree of OA are all considered with respect to knee arthroplasty timing and technique.
There is good evidence that arthroscopy with debridement, on the other hand, is no more effective than conservative management.67
Continue to: Unicompartmental or "partial"...
Unicompartmental or “partial” knee replacements are reserved for select cases when 1 knee compartment has a significantly higher degree of degenerative change.
CASE After reviewing the therapeutic options with your patient, you agree that she will undergo a course of physical therapy and try using topical diclofenac along with a hinged knee brace. Because of the patient’s age and co-morbidities of cardiovascular disease and mild chronic kidney disease, oral NSAIDs are avoided at this time.
The patient returns to the office in 2 months reporting mild improvement in her pain. To provide additional pain relief, an ultrasound-guided IA steroid injection is attempted. The patient also continues home physical therapy, activity modification, topical diclofenac, and use of a hinged knee brace.
She returns to the office 2 months later, reporting continued improvement in her pain. No further intervention is undertaken at this time.
CORRESPONDENCE
Ryan A. Sprouse, MD, CAQSM, West Virginia University School of Medicine–Eastern Campus, WVU Medicine Orthopaedics and Sports Medicine, 912 Somerset Boulevard, Charles Town, WV 25414; [email protected].
1. Wallace IJ, Worthington S,Felson DT, et al. Knee osteoarthritis has doubled in prevalence since the mid-20th century. Proc Natl Acad Sci. 2017;114:9332-9336.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35.
3. Vina ER, Kwoh CK. Epidemiology of osteoarthritis: literature update. Curr Opin Rheumatol. 2018;30:160-167.
4. Warner SC, Valdes AM. Genetic association studies in osteoarthritis: is it fairytale? Curr Opin Rheumatol. 2017;29:103-109.
5. Srikanth VK, Fryer JL, Zhai G, et al. A meta-analysis of sex differences prevalence, incidence and severity of osteoarthritis. Osteoarthritis Cartilage. 2005;13:769-781.
6. Palazzo C, Nguyen C, Lefevre-Colau MM, et al. Risk factors and burden of osteoarthritis. Ann Phys Rehabil Med. 2016;59:134-138.
7. Tanamas S, Hanna FS, Cicuttini FM, et al. Does knee malalignment increase the risk of development and progression of knee osteoarthritis? A systematic review. Arthritis Rheum. 2009;61:459-467.
8. Yucesoy B, Charles LE, Baker B, et al. Occupational and genetic risk factors for osteoarthritis: a review. Work. 2015;50:261-273.
9. Trojian TH, Concoff AL, Joy SM, et al. AMSSM scientific statement concerning viscosupplementation injections for knee osteoarthritis: importance for individual patient outcomes. Br J Sports Med. 2016;50:84-92.
10. Hochberg MC, Altman RD, April KT, et al. American College of Rheumatology 2012 Recommendations for the Use of Nonpharmacologic and Pharmacologic Therapies in Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res. 2012;64:465-474.
11. Bedson J, Croft PR. The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord. 2008;9:116.
12. Felson DT, Anderson JJ, Naimark A, et al. Obesity and knee osteoarthritis. The Framingham Study. Ann Intern Med. 1988;109:18-24.
13. Yusuf E, Bijsterbosch J, Slagboom PE, et al. Body mass index and alignment and their interaction as risk factors for progression of knees with radiographic signs of osteoarthritis. Osteoarthritis Cartilage. 2011;19:1117-1122.
14. Niu J, Zhang YQ, Torner J, et al. Is obesity a risk factor for progressive radiographic knee osteoarthritis? Arthritis Rheum. 2009;61:329-335.
15. Messier SP, Gutekunst DJ, Davis C, et al. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005;52:2026-2032.
16. Messier SP, Mihalko SL, Legault C, et al. Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis: the IDEA randomized clinical trial. JAMA. 2013;310:1263-1273.
17. Fransen M, McConnell S, Harmer AR, et al. Exercise for osteoarthritis of the knee: a Cochrane systematic review. Br J Sports Med.
18. Kan L, Zhang J, Yang Y, et al. The effects of yoga on pain, mobility, and quality of life in patients with knee osteoarthritis: a systematic review. Evid Based Complement Alternat Med. 2016;2016:6016532.
19. Chang WD, Chen S, Lee CL, et al. The effects of tai chi chuan on improving mind-body health for knee osteoarthritis patients: a systematic review and meta-analysis. Evid Based Complement Alternat Med. 2016;2016:1813979.
20. Takacs J, Krowchuk NM, Garland SJ, et al. Dynamic balance training improves physical function in individuals with knee osteoarthritis: a pilot randomized controlled trial. Arch Phys Med Rehabil. 2017;98:1586-1593.
21. Bartels EM, Juhl CB, Christensen R, et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev. 2016;(3):CD005523.
22. Hinman RS, Heywood SE, Day AR. Aquatic physical therapy for hip and knee osteoarthritis: results of a single-blind randomized controlled trial. Phys Ther. 2007;87:32-43.
23. Tanaka R, Ozawa J, Kito N, et al. Efficacy of strengthening or aerobic exercise on pain relief in people with knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Clin Rehabil. 2013;27:1059-1071.
24. Knoop J, Steultjens MP, Roorda LD, et al. Improvement in upper leg muscle strength underlies beneficial effects of exercise therapy in knee osteoarthritis: secondary analysis from a randomised controlled trial. Physiotherapy. 2015;101:171-177.
25. Mat S, Tan MP, Kamaruzzaman SB, et al. Physical therapies for improving balance and reducing falls risk in osteoarthritis of the knee: a systematic review. Age Ageing. 2015;44:16-24.
26. Peeler J, Christian M, Cooper J, et al. Managing knee osteoarthritis: the effects of body weight supported physical activity on joint pain, function, and thigh muscle strength. Clin J Sport Med. 2015;25:518-523.
27. Peeler J, Ripat J. The effect of low-load exercise on joint pain, function, and activities of daily living in patients with knee osteoarthritis. Knee. 2018;25:135-145.
28. Takacs J, Anderson JE, Leiter JR, et al. Lower body positive pressure: an emerging technology in the battle against knee osteoarthritis? Clin Interv Aging. 2013;8:983-991.
29. Duivenvoorden T, Brouwer RW, van Raaij TM, et al. Braces and orthoses for treating osteoarthritis of the knee. Cochrane Database Syst Rev. 2015;(3):CD004020.
30. Gohal C, Shanmugaraj A, Tate P, et al. Effectiveness of valgus offloading knee braces in the treatment of medial compartment knee osteoarthritis: a systematic review. Sports Health. 2018;10:500-514.
31. Brouwer RW, van Raaij TM, Verhaar JA, et al. Brace treatment for osteoarthritis of the knee: a prospective randomized multi-centre trial. Osteoarthritis Cartilage. 2006;14:777-783.
32. Lee PY, Winfield TG, Harris SR, et al. Unloading knee brace is a cost-effective method to bridge and delay surgery in unicompartmental knee arthritis. BMJ Open Sport Exerc Med. 2017;2:e000195.
33. Wilson B, Rankin H, Barnes CL. Long-term results of an unloader brace in patients with unicompartmental knee osteoarthritis. Orthopedics. 2011;34:334-347.
34. Cudejko T, van der Esch M, van der Leeden M, et al. Effect of soft braces on pain and physical function in patients with knee osteoarthritis: systematic review with meta-analyses. Arch Phys Med Rehabil. 2018;99:153-163.
35. Cudejko T, van der Esch M, van den Noort JC. Decreased pain and improved dynamic knee instability mediate the beneficial effect of wearing a soft knee brace on activity limitations in persons with knee osteoarthritis. Arthritis Care Res (Hoboken). 2019;71:1036-1043.
36. Machado GC, Maher CG, Ferreira PH, et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ. 2015;350:h1225.
37. da Costa BR, Reichenbach S, Keller N, et al. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: a network meta-analysis. Lancet. 2017;390:e21-e33.
38. Bannuru RR, Schmid CH, Kent DM, et al. Comparative effectiveness of pharmacologic interventions for knee osteoarthritis: a systematic review and network meta-analysis. Ann Intern Med. 2015;162:46-54.
39. Gregori D, Giacovelli G, Minto C, et al. Association of pharmacological treatments with long-term pain control in patients with knee osteoarthritis: a systematic review and meta-analysis. JAMA. 2018;320:2564-2579.
40. Puljak L, Marin A, Vrdoljak D, et al. Celecoxib for osteoarthritis. Cochrane Database Syst Rev. 2017;(5):CD009865.
41. Jevsevar DS. Treatment of osteoarthritis of the knee: evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013;9:571-576.
42. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
43. Singh JA, Noorbaloochi S, MacDonald R, et al. Chondroitin for osteoarthritis. Cochrane Database Syst Rev. 2015;(1):CD005614.
44. Yang S, Eaton CB, McAlindon TE, et al. Effects of glucosamine and chondroitin on treating knee osteoarthritis: an analysis with marginal structural models. Arthritis Rheumatol. 2015;67:714-723.
45. Ogata T, Yuki Ideno Y, Masami Akai M,et al. Effects of glucosamine in patients with osteoarthritis of the knee: a systematic review and meta-analysis. Clin Rheumatol. 2018;37:2479-2487.
46. Towheed TE, Maxwell L, Anastassiades TP, et al. Glucosamine therapy for treating osteoarthritis. Cochrane Database Syst Rev. 2009;(2):CD002946.
47. Bruyèreetal O, Cooper C, Pelletier JP, et al. A consensus statement on the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) algorithm for the management of knee osteoarthritis—from evidence-based medicine to the real-life setting. Semin Arthritis Rheum. 2016;45(4 suppl):S3-S11.
48. Ishijima M, Nakamura T, Shimizu K, et al. Intra-articular hyaluronic acid injection versus oral non-steroidal anti-inflammatory drug for the treatment of knee osteoarthritis: a multi-center, randomized, open-label, non-inferiority trial. Arthritis Res Ther. 2014;16:R18.
49. Juni P, Hari R, Rutjes AW, et al. Intra-articular corticosteroid for knee osteoarthritis. Cochrane Database Syst Rev. 2015;(10):CD005328.
50. McAlindon TE, LaValley MP, Harvey FW, et al. Effect of intra-articular triamcinolone vs saline on knee cartilage volume and pain in patients with knee osteoarthritis: a randomized clinical trial. JAMA. 2017;317:1967-1975.
51. Derry S, Conaghan P, Da Silva JA, et al. Topical NSAIDs for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2016;(4):CD007400.
52. Tugwell PS, Wells GA, Shainhouse JZ. Equivalence study of a topical diclofenac solution (pennsaid) compared with oral diclofenac in symptomatic treatment of osteoarthritis of the knee: a randomized controlled trial. J Rheumatol. 2004;31:2002-2012.
53. Wadsworth LT, Kent JD, Holt RJ. Efficacy and safety of diclofenac sodium 2% topical solution for osteoarthritis of the knee: a randomized, double-blind, vehicle-controlled, 4 week study. Curr Med Res Opin. 2016;32:241-250.
54. Roth SH, Shainhouse JZ. Efficacy and safety of a topical diclofenac solution (pennsaid) in the treatment of primary osteoarthritis of the knee: a randomized, double-blind, vehicle-controlled clinical trial. Arch Intern Med. 2004;164:2017-2023.
55. Baer PA, Thomas LM, Shainhouse Z. Treatment of osteoarthritis of the knee with a topical diclofenac solution: a randomised controlled, 6-week trial. BMC Musculoskelet Disord. 2005;6:44.
56. Zeng C, Wei J, Persson MSM, et al. Relative efficacy and safety of topical non-steroidal anti-inflammatory drugs for osteoarthritis: a systematic review and network meta-analysis of randomised controlled trials and observational studies. Br J Sports Med. 2018;52:642-650.
57. Guedes V, Castro JP, Brito I. Topical capsaicin for pain in osteoarthritis: a literature review. Reumatol Clin. 2018;14:40-45.
58. Persson MSM, Stocks J, Walsh DA, et al. The relative efficacy of topical non-steroidal anti-inflammatory drugs and capsaicin in osteoarthritis: a network meta-analysis of randomised controlled trials. Osteoarthritis Cartilage. 2018;26:1575-1582.
59. Cole BJ, Karas V, Hussey K, et al. Hyaluronic acid versus platelet-rich plasma: a prospective, double-blind randomized controlled trial comparing clinical outcomes and effects on intra-articular biology for the treatment of knee osteoarthritis. Am J Sports Med. 2017;45:339-346.
60. Laudy AB, Bakker EW, Rekers M, et al. Efficacy of platelet-rich plasma injections in osteoarthritis of the knee: a systematic review and meta-analysis. Br J Sports Med. 2015;49:657-672.
61. Han Y, Huang H, Pan J, et al. Meta-analysis comparing platelet-rich plasma vs hyaluronic acid injection in patients with knee osteoarthritis. Pain Med. 2019;20:1418-1429.
62. Filardo G, Di Matteo B, Di Martino A, et al. Platelet-rich plasma intra-articular knee injections show no superiority versus viscosupplementation: a randomized controlled trial. Am J Sports Med. 2015;43:1575-1582.
63. Di Martino A, Di Matteo B, Papio T, et al. Platelet-rich plasma versus hyaluronic acid injections for the treatment of knee osteoarthritis: results at 5 years of a double-blind, randomized controlled trial. Am J Sports Med. 2019;47:347-354.
64. Bodick N, Lufkin J, Willwerth C, et al. An intra-articular, extended-release formulation of triamcinolone acetonide prolongs and amplifies analgesic effect in patients with osteoarthritis of the knee: a randomized clinical trial. J Bone Joint Surg Am. 2015;97:877-888.
65. Conaghan PG, Cohen SB, Berenbaum F, et al. Brief report: a phase IIb trial of a novel extended-release microsphere formulation of triamcinolone acetonide for intraarticular injection in knee osteoarthritis. Arthritis Rheumatol. 2018;70:204-211.
66. Conaghan PG, Hunter DJ, Cohen SB, et al. Effects of a single intra-articular injection of a microsphere formulation of triamcinolone acetonide on knee osteoarthritis pain: a double-blinded, randomized, placebo-controlled, multinational study. J Bone Jt Surg Am. 2018;100:666–677.
67. Thorlund JB, Juhl CB, Roos EM, et al. Arthroscopic surgery for degenerative knee: systematic review and meta-analysis of benefits and harms. BMJ. 2015;350:h2747.
1. Wallace IJ, Worthington S,Felson DT, et al. Knee osteoarthritis has doubled in prevalence since the mid-20th century. Proc Natl Acad Sci. 2017;114:9332-9336.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35.
3. Vina ER, Kwoh CK. Epidemiology of osteoarthritis: literature update. Curr Opin Rheumatol. 2018;30:160-167.
4. Warner SC, Valdes AM. Genetic association studies in osteoarthritis: is it fairytale? Curr Opin Rheumatol. 2017;29:103-109.
5. Srikanth VK, Fryer JL, Zhai G, et al. A meta-analysis of sex differences prevalence, incidence and severity of osteoarthritis. Osteoarthritis Cartilage. 2005;13:769-781.
6. Palazzo C, Nguyen C, Lefevre-Colau MM, et al. Risk factors and burden of osteoarthritis. Ann Phys Rehabil Med. 2016;59:134-138.
7. Tanamas S, Hanna FS, Cicuttini FM, et al. Does knee malalignment increase the risk of development and progression of knee osteoarthritis? A systematic review. Arthritis Rheum. 2009;61:459-467.
8. Yucesoy B, Charles LE, Baker B, et al. Occupational and genetic risk factors for osteoarthritis: a review. Work. 2015;50:261-273.
9. Trojian TH, Concoff AL, Joy SM, et al. AMSSM scientific statement concerning viscosupplementation injections for knee osteoarthritis: importance for individual patient outcomes. Br J Sports Med. 2016;50:84-92.
10. Hochberg MC, Altman RD, April KT, et al. American College of Rheumatology 2012 Recommendations for the Use of Nonpharmacologic and Pharmacologic Therapies in Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res. 2012;64:465-474.
11. Bedson J, Croft PR. The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord. 2008;9:116.
12. Felson DT, Anderson JJ, Naimark A, et al. Obesity and knee osteoarthritis. The Framingham Study. Ann Intern Med. 1988;109:18-24.
13. Yusuf E, Bijsterbosch J, Slagboom PE, et al. Body mass index and alignment and their interaction as risk factors for progression of knees with radiographic signs of osteoarthritis. Osteoarthritis Cartilage. 2011;19:1117-1122.
14. Niu J, Zhang YQ, Torner J, et al. Is obesity a risk factor for progressive radiographic knee osteoarthritis? Arthritis Rheum. 2009;61:329-335.
15. Messier SP, Gutekunst DJ, Davis C, et al. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005;52:2026-2032.
16. Messier SP, Mihalko SL, Legault C, et al. Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis: the IDEA randomized clinical trial. JAMA. 2013;310:1263-1273.
17. Fransen M, McConnell S, Harmer AR, et al. Exercise for osteoarthritis of the knee: a Cochrane systematic review. Br J Sports Med.
18. Kan L, Zhang J, Yang Y, et al. The effects of yoga on pain, mobility, and quality of life in patients with knee osteoarthritis: a systematic review. Evid Based Complement Alternat Med. 2016;2016:6016532.
19. Chang WD, Chen S, Lee CL, et al. The effects of tai chi chuan on improving mind-body health for knee osteoarthritis patients: a systematic review and meta-analysis. Evid Based Complement Alternat Med. 2016;2016:1813979.
20. Takacs J, Krowchuk NM, Garland SJ, et al. Dynamic balance training improves physical function in individuals with knee osteoarthritis: a pilot randomized controlled trial. Arch Phys Med Rehabil. 2017;98:1586-1593.
21. Bartels EM, Juhl CB, Christensen R, et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev. 2016;(3):CD005523.
22. Hinman RS, Heywood SE, Day AR. Aquatic physical therapy for hip and knee osteoarthritis: results of a single-blind randomized controlled trial. Phys Ther. 2007;87:32-43.
23. Tanaka R, Ozawa J, Kito N, et al. Efficacy of strengthening or aerobic exercise on pain relief in people with knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Clin Rehabil. 2013;27:1059-1071.
24. Knoop J, Steultjens MP, Roorda LD, et al. Improvement in upper leg muscle strength underlies beneficial effects of exercise therapy in knee osteoarthritis: secondary analysis from a randomised controlled trial. Physiotherapy. 2015;101:171-177.
25. Mat S, Tan MP, Kamaruzzaman SB, et al. Physical therapies for improving balance and reducing falls risk in osteoarthritis of the knee: a systematic review. Age Ageing. 2015;44:16-24.
26. Peeler J, Christian M, Cooper J, et al. Managing knee osteoarthritis: the effects of body weight supported physical activity on joint pain, function, and thigh muscle strength. Clin J Sport Med. 2015;25:518-523.
27. Peeler J, Ripat J. The effect of low-load exercise on joint pain, function, and activities of daily living in patients with knee osteoarthritis. Knee. 2018;25:135-145.
28. Takacs J, Anderson JE, Leiter JR, et al. Lower body positive pressure: an emerging technology in the battle against knee osteoarthritis? Clin Interv Aging. 2013;8:983-991.
29. Duivenvoorden T, Brouwer RW, van Raaij TM, et al. Braces and orthoses for treating osteoarthritis of the knee. Cochrane Database Syst Rev. 2015;(3):CD004020.
30. Gohal C, Shanmugaraj A, Tate P, et al. Effectiveness of valgus offloading knee braces in the treatment of medial compartment knee osteoarthritis: a systematic review. Sports Health. 2018;10:500-514.
31. Brouwer RW, van Raaij TM, Verhaar JA, et al. Brace treatment for osteoarthritis of the knee: a prospective randomized multi-centre trial. Osteoarthritis Cartilage. 2006;14:777-783.
32. Lee PY, Winfield TG, Harris SR, et al. Unloading knee brace is a cost-effective method to bridge and delay surgery in unicompartmental knee arthritis. BMJ Open Sport Exerc Med. 2017;2:e000195.
33. Wilson B, Rankin H, Barnes CL. Long-term results of an unloader brace in patients with unicompartmental knee osteoarthritis. Orthopedics. 2011;34:334-347.
34. Cudejko T, van der Esch M, van der Leeden M, et al. Effect of soft braces on pain and physical function in patients with knee osteoarthritis: systematic review with meta-analyses. Arch Phys Med Rehabil. 2018;99:153-163.
35. Cudejko T, van der Esch M, van den Noort JC. Decreased pain and improved dynamic knee instability mediate the beneficial effect of wearing a soft knee brace on activity limitations in persons with knee osteoarthritis. Arthritis Care Res (Hoboken). 2019;71:1036-1043.
36. Machado GC, Maher CG, Ferreira PH, et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ. 2015;350:h1225.
37. da Costa BR, Reichenbach S, Keller N, et al. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: a network meta-analysis. Lancet. 2017;390:e21-e33.
38. Bannuru RR, Schmid CH, Kent DM, et al. Comparative effectiveness of pharmacologic interventions for knee osteoarthritis: a systematic review and network meta-analysis. Ann Intern Med. 2015;162:46-54.
39. Gregori D, Giacovelli G, Minto C, et al. Association of pharmacological treatments with long-term pain control in patients with knee osteoarthritis: a systematic review and meta-analysis. JAMA. 2018;320:2564-2579.
40. Puljak L, Marin A, Vrdoljak D, et al. Celecoxib for osteoarthritis. Cochrane Database Syst Rev. 2017;(5):CD009865.
41. Jevsevar DS. Treatment of osteoarthritis of the knee: evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013;9:571-576.
42. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
43. Singh JA, Noorbaloochi S, MacDonald R, et al. Chondroitin for osteoarthritis. Cochrane Database Syst Rev. 2015;(1):CD005614.
44. Yang S, Eaton CB, McAlindon TE, et al. Effects of glucosamine and chondroitin on treating knee osteoarthritis: an analysis with marginal structural models. Arthritis Rheumatol. 2015;67:714-723.
45. Ogata T, Yuki Ideno Y, Masami Akai M,et al. Effects of glucosamine in patients with osteoarthritis of the knee: a systematic review and meta-analysis. Clin Rheumatol. 2018;37:2479-2487.
46. Towheed TE, Maxwell L, Anastassiades TP, et al. Glucosamine therapy for treating osteoarthritis. Cochrane Database Syst Rev. 2009;(2):CD002946.
47. Bruyèreetal O, Cooper C, Pelletier JP, et al. A consensus statement on the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) algorithm for the management of knee osteoarthritis—from evidence-based medicine to the real-life setting. Semin Arthritis Rheum. 2016;45(4 suppl):S3-S11.
48. Ishijima M, Nakamura T, Shimizu K, et al. Intra-articular hyaluronic acid injection versus oral non-steroidal anti-inflammatory drug for the treatment of knee osteoarthritis: a multi-center, randomized, open-label, non-inferiority trial. Arthritis Res Ther. 2014;16:R18.
49. Juni P, Hari R, Rutjes AW, et al. Intra-articular corticosteroid for knee osteoarthritis. Cochrane Database Syst Rev. 2015;(10):CD005328.
50. McAlindon TE, LaValley MP, Harvey FW, et al. Effect of intra-articular triamcinolone vs saline on knee cartilage volume and pain in patients with knee osteoarthritis: a randomized clinical trial. JAMA. 2017;317:1967-1975.
51. Derry S, Conaghan P, Da Silva JA, et al. Topical NSAIDs for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2016;(4):CD007400.
52. Tugwell PS, Wells GA, Shainhouse JZ. Equivalence study of a topical diclofenac solution (pennsaid) compared with oral diclofenac in symptomatic treatment of osteoarthritis of the knee: a randomized controlled trial. J Rheumatol. 2004;31:2002-2012.
53. Wadsworth LT, Kent JD, Holt RJ. Efficacy and safety of diclofenac sodium 2% topical solution for osteoarthritis of the knee: a randomized, double-blind, vehicle-controlled, 4 week study. Curr Med Res Opin. 2016;32:241-250.
54. Roth SH, Shainhouse JZ. Efficacy and safety of a topical diclofenac solution (pennsaid) in the treatment of primary osteoarthritis of the knee: a randomized, double-blind, vehicle-controlled clinical trial. Arch Intern Med. 2004;164:2017-2023.
55. Baer PA, Thomas LM, Shainhouse Z. Treatment of osteoarthritis of the knee with a topical diclofenac solution: a randomised controlled, 6-week trial. BMC Musculoskelet Disord. 2005;6:44.
56. Zeng C, Wei J, Persson MSM, et al. Relative efficacy and safety of topical non-steroidal anti-inflammatory drugs for osteoarthritis: a systematic review and network meta-analysis of randomised controlled trials and observational studies. Br J Sports Med. 2018;52:642-650.
57. Guedes V, Castro JP, Brito I. Topical capsaicin for pain in osteoarthritis: a literature review. Reumatol Clin. 2018;14:40-45.
58. Persson MSM, Stocks J, Walsh DA, et al. The relative efficacy of topical non-steroidal anti-inflammatory drugs and capsaicin in osteoarthritis: a network meta-analysis of randomised controlled trials. Osteoarthritis Cartilage. 2018;26:1575-1582.
59. Cole BJ, Karas V, Hussey K, et al. Hyaluronic acid versus platelet-rich plasma: a prospective, double-blind randomized controlled trial comparing clinical outcomes and effects on intra-articular biology for the treatment of knee osteoarthritis. Am J Sports Med. 2017;45:339-346.
60. Laudy AB, Bakker EW, Rekers M, et al. Efficacy of platelet-rich plasma injections in osteoarthritis of the knee: a systematic review and meta-analysis. Br J Sports Med. 2015;49:657-672.
61. Han Y, Huang H, Pan J, et al. Meta-analysis comparing platelet-rich plasma vs hyaluronic acid injection in patients with knee osteoarthritis. Pain Med. 2019;20:1418-1429.
62. Filardo G, Di Matteo B, Di Martino A, et al. Platelet-rich plasma intra-articular knee injections show no superiority versus viscosupplementation: a randomized controlled trial. Am J Sports Med. 2015;43:1575-1582.
63. Di Martino A, Di Matteo B, Papio T, et al. Platelet-rich plasma versus hyaluronic acid injections for the treatment of knee osteoarthritis: results at 5 years of a double-blind, randomized controlled trial. Am J Sports Med. 2019;47:347-354.
64. Bodick N, Lufkin J, Willwerth C, et al. An intra-articular, extended-release formulation of triamcinolone acetonide prolongs and amplifies analgesic effect in patients with osteoarthritis of the knee: a randomized clinical trial. J Bone Joint Surg Am. 2015;97:877-888.
65. Conaghan PG, Cohen SB, Berenbaum F, et al. Brief report: a phase IIb trial of a novel extended-release microsphere formulation of triamcinolone acetonide for intraarticular injection in knee osteoarthritis. Arthritis Rheumatol. 2018;70:204-211.
66. Conaghan PG, Hunter DJ, Cohen SB, et al. Effects of a single intra-articular injection of a microsphere formulation of triamcinolone acetonide on knee osteoarthritis pain: a double-blinded, randomized, placebo-controlled, multinational study. J Bone Jt Surg Am. 2018;100:666–677.
67. Thorlund JB, Juhl CB, Roos EM, et al. Arthroscopic surgery for degenerative knee: systematic review and meta-analysis of benefits and harms. BMJ. 2015;350:h2747.
PRACTICE RECOMMENDATIONS
› Treat pain from knee osteoarthritis (OA) with weight management and low-impact exercise to decrease the risk of disease progression. A
› Prescribe oral or topical nonsteroidal anti-inflammatory drugs to relieve pain from knee OA, as both forms are equally effective. B
› Recommend a medial unloading (valgus) knee brace for short-term relief of medial knee OA. B
› Consider a trial of intra-articular corticosteroids or intra-articular hyaluronic acid derivatives for short-term relief of knee OA pain. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Phone outreach intervention feasible to reduce SLE readmissions
A nurse-led intervention aimed at reducing hospital readmission rates for systemic lupus erythematosus (SLE) is feasible but the jury is out as to whether it can achieve its primary goal, a study has found.
A paper published in Arthritis Care & Research presents the outcomes of a retrospective study using electronic health records that looked at the effect of a quality improvement initiative at the University of Colorado Hospital on readmission rates in two cohorts of 48 and 56 individuals with SLE.
Emily Bowers, MD, of the department of rheumatology at the University of Colorado at Denver, Aurora, and coauthors wrote that hospital readmission rates for SLE are as high as 36% for 30-day readmission. They are significantly higher than for other common chronic diseases such as heart failure, COPD, and diabetes. Readmission for SLE is associated with young age, ethnic or racial diversity, public health insurance, multiorgan involvement, and other comorbidities.
The intervention involved first alerting clinic nurses via the patient’s electronic medical record when the patient was discharged from hospital. The nurses would then call the patient within 48 hours to answer any questions and review their discharge information, and then consult with a rheumatologist on on-call if needed. This call was documented in the patient’s medical record.
In the preintervention cohort, there were 59 hospitalizations among 48 patients, 29% of which were followed by readmission within 30 days; 53% of these readmissions were lupus related. In the cohort that followed introduction of the intervention, there were 73 hospitalizations among 56 individuals, and 19% were followed by readmission within 30 days, 29% of which were lupus related.
After accounting for gender, age, race, and insurance type, the researchers calculated that there was an 89% higher odds of readmission in the nonintervention group than in the intervention group, but the difference was not statistically significant.
The authors noted that although the results were not statistically significant, the low cost of the intervention – requiring around 30 minutes of nursing time – meant even small reductions in the number of emergency department or hospital admissions would make it a cost-effective approach.
“Telephone outreach is an excellent method of providing additional support to patients, assessing clinical needs, reinforcing education about SLE, medications, and common complications such as drug side effects and infections, and allows for patients to ask pertinent questions to RN providers with expertise in the management of lupus,” the authors wrote.
The nurses also recorded qualitative information about the calls, which picked up some patient issues that could be addressed. For example, a patient was discharged with the wrong amount of prednisone, which the nurse was able to fix by adjusting the order and sending it to the pharmacy. Two other patients were confused by their medication instructions and were taking the medication incorrectly; the nurse arranged for the patients to come in for educational session. In another case, the nurse was able to arrange an infusion for the patient, and for one patient with concerns about infection, the nurse was able to advise that person on symptoms and how to seek care.
“To increase implementation of the intervention, we have discussed creating a discharge order set, which would include an automatic EMR message to the nurses,” the authors wrote. “Future studies should explore alternative ways of communicating with our patients after discharge, such as the use of text messaging, messaging through the patient portal in the EMR, or telehealth.”
The authors had no financial disclosures, and there was no outside financial support for the study.
SOURCE: Bowers E et al. Arthritis Care Res. 2020 Aug 29. doi: 10.1002/acr.24435.
A nurse-led intervention aimed at reducing hospital readmission rates for systemic lupus erythematosus (SLE) is feasible but the jury is out as to whether it can achieve its primary goal, a study has found.
A paper published in Arthritis Care & Research presents the outcomes of a retrospective study using electronic health records that looked at the effect of a quality improvement initiative at the University of Colorado Hospital on readmission rates in two cohorts of 48 and 56 individuals with SLE.
Emily Bowers, MD, of the department of rheumatology at the University of Colorado at Denver, Aurora, and coauthors wrote that hospital readmission rates for SLE are as high as 36% for 30-day readmission. They are significantly higher than for other common chronic diseases such as heart failure, COPD, and diabetes. Readmission for SLE is associated with young age, ethnic or racial diversity, public health insurance, multiorgan involvement, and other comorbidities.
The intervention involved first alerting clinic nurses via the patient’s electronic medical record when the patient was discharged from hospital. The nurses would then call the patient within 48 hours to answer any questions and review their discharge information, and then consult with a rheumatologist on on-call if needed. This call was documented in the patient’s medical record.
In the preintervention cohort, there were 59 hospitalizations among 48 patients, 29% of which were followed by readmission within 30 days; 53% of these readmissions were lupus related. In the cohort that followed introduction of the intervention, there were 73 hospitalizations among 56 individuals, and 19% were followed by readmission within 30 days, 29% of which were lupus related.
After accounting for gender, age, race, and insurance type, the researchers calculated that there was an 89% higher odds of readmission in the nonintervention group than in the intervention group, but the difference was not statistically significant.
The authors noted that although the results were not statistically significant, the low cost of the intervention – requiring around 30 minutes of nursing time – meant even small reductions in the number of emergency department or hospital admissions would make it a cost-effective approach.
“Telephone outreach is an excellent method of providing additional support to patients, assessing clinical needs, reinforcing education about SLE, medications, and common complications such as drug side effects and infections, and allows for patients to ask pertinent questions to RN providers with expertise in the management of lupus,” the authors wrote.
The nurses also recorded qualitative information about the calls, which picked up some patient issues that could be addressed. For example, a patient was discharged with the wrong amount of prednisone, which the nurse was able to fix by adjusting the order and sending it to the pharmacy. Two other patients were confused by their medication instructions and were taking the medication incorrectly; the nurse arranged for the patients to come in for educational session. In another case, the nurse was able to arrange an infusion for the patient, and for one patient with concerns about infection, the nurse was able to advise that person on symptoms and how to seek care.
“To increase implementation of the intervention, we have discussed creating a discharge order set, which would include an automatic EMR message to the nurses,” the authors wrote. “Future studies should explore alternative ways of communicating with our patients after discharge, such as the use of text messaging, messaging through the patient portal in the EMR, or telehealth.”
The authors had no financial disclosures, and there was no outside financial support for the study.
SOURCE: Bowers E et al. Arthritis Care Res. 2020 Aug 29. doi: 10.1002/acr.24435.
A nurse-led intervention aimed at reducing hospital readmission rates for systemic lupus erythematosus (SLE) is feasible but the jury is out as to whether it can achieve its primary goal, a study has found.
A paper published in Arthritis Care & Research presents the outcomes of a retrospective study using electronic health records that looked at the effect of a quality improvement initiative at the University of Colorado Hospital on readmission rates in two cohorts of 48 and 56 individuals with SLE.
Emily Bowers, MD, of the department of rheumatology at the University of Colorado at Denver, Aurora, and coauthors wrote that hospital readmission rates for SLE are as high as 36% for 30-day readmission. They are significantly higher than for other common chronic diseases such as heart failure, COPD, and diabetes. Readmission for SLE is associated with young age, ethnic or racial diversity, public health insurance, multiorgan involvement, and other comorbidities.
The intervention involved first alerting clinic nurses via the patient’s electronic medical record when the patient was discharged from hospital. The nurses would then call the patient within 48 hours to answer any questions and review their discharge information, and then consult with a rheumatologist on on-call if needed. This call was documented in the patient’s medical record.
In the preintervention cohort, there were 59 hospitalizations among 48 patients, 29% of which were followed by readmission within 30 days; 53% of these readmissions were lupus related. In the cohort that followed introduction of the intervention, there were 73 hospitalizations among 56 individuals, and 19% were followed by readmission within 30 days, 29% of which were lupus related.
After accounting for gender, age, race, and insurance type, the researchers calculated that there was an 89% higher odds of readmission in the nonintervention group than in the intervention group, but the difference was not statistically significant.
The authors noted that although the results were not statistically significant, the low cost of the intervention – requiring around 30 minutes of nursing time – meant even small reductions in the number of emergency department or hospital admissions would make it a cost-effective approach.
“Telephone outreach is an excellent method of providing additional support to patients, assessing clinical needs, reinforcing education about SLE, medications, and common complications such as drug side effects and infections, and allows for patients to ask pertinent questions to RN providers with expertise in the management of lupus,” the authors wrote.
The nurses also recorded qualitative information about the calls, which picked up some patient issues that could be addressed. For example, a patient was discharged with the wrong amount of prednisone, which the nurse was able to fix by adjusting the order and sending it to the pharmacy. Two other patients were confused by their medication instructions and were taking the medication incorrectly; the nurse arranged for the patients to come in for educational session. In another case, the nurse was able to arrange an infusion for the patient, and for one patient with concerns about infection, the nurse was able to advise that person on symptoms and how to seek care.
“To increase implementation of the intervention, we have discussed creating a discharge order set, which would include an automatic EMR message to the nurses,” the authors wrote. “Future studies should explore alternative ways of communicating with our patients after discharge, such as the use of text messaging, messaging through the patient portal in the EMR, or telehealth.”
The authors had no financial disclosures, and there was no outside financial support for the study.
SOURCE: Bowers E et al. Arthritis Care Res. 2020 Aug 29. doi: 10.1002/acr.24435.
FROM ARTHRITIS CARE & RESEARCH
Small-fiber polyneuropathy may underlie dysautonomia in ME/CFS
A significant proportion of patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia may have potentially treatable underlying autoimmune-associated small-fiber polyneuropathy (aaSFPN), pilot data suggest.
The findings, from a single-site study of 61 patients with ME/CFS, were presented August 21 at the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis by Ryan Whelan, BS, a research assistant at Simmaron Research Institute, Incline Village, Nevada.
Recent evidence suggests an autoimmune etiology for some patients with ME/CFS, which is defined as experiencing for a period of at least 6 months profound, unexplained fatigue, postexertional malaise, and unrefreshing sleep, as well as cognitive dysfunction and/or orthostatic intolerance (OI).
OI is part of a spectrum of autonomic dysfunction commonly seen in ME/CFS patients, which may also include postural orthostatic tachycardia (POTS), peripheral temperature dysregulation and light sensitivity, neuropathic pain, and gastrointestinal complaints. Many of these symptoms overlap those reported by patients with aaSFPN, a common but underdiagnosed neurodegenerative disorder characterized by the loss of peripheral autonomic nerve fibers, Whelan explained.
Findings from the current study show that in more than half of ME/CFS patients, levels of at least one autoantibody were elevated. A majority had comorbid POTS or OI, and over a third had biopsy-confirmed aaSFPN.
“Given the overlap of symptoms and common etiological basis, it may be important to identify ME/CFS patients who present with comorbid aaSFPN, as it has been shown that immune modulatory agents, including intravenous gamma globulin [IVIG], reduce the autonomic symptom burden in aaSFPN patients,” Whelan said.
He noted that Anne Louise Oaklander, MD, a neurologist at Massachusetts General Hospital, Harvard Medical School, Boston, and colleagues previously linked aaSFPN with fibromyalgia. In addition, they’ve found a connection between small-fiber dysfunction and postexertional malaise, which is a hallmark ME/CFS symptom.
Asked to comment on Whelan’s presentation, IACFSME co-president Lily Chu, MD, told Medscape Medical News that the new findings are “valuable, because ME/CFS has always been looked upon as just subjective symptoms. When people have laboratory abnormalities, it can be due to a bunch of other causes, but...here’s pathology, here’s a biopsy of actual damage. It’s not just a transient finding. You can actually see it. ... It’s a solid concrete piece of evidence vs something that can fluctuate.”
Autoantibodies, Autonomic Dysfunction, and Small-Fiber Polyneuropathy
Whelan and colleagues conducted an extensive analysis of medical records of 364 patients with ME/CFS (72% female) to identify potential aaSFPN comorbidity. Such identifications were made on the basis of progress notes documenting autonomic dysfunction, laboratory results for serum autoantibodies, and questionnaire symptom self-reports.
They identified 61 patients as possibly having comorbid aaSFPN. Of those, 52% tested positive for at least 1 of 4 autoantibodies, including antimuscarinic cholinergic receptor 4 (47%), anti-beta-2 adrenergic (27%), antimuscarinic cholinergic 3 (25%), and anti-beta-1 adrenergic (13%). These autoantibodies were linked to ME/CFS in a recent Swedish cohort study.
“Evidence supports that these autoantibodies may bind to receptor sites, blocking ligands from reaching these receptors. Disturbances of adrenergic and cholinergic receptors by these autoantibodies may contribute to symptoms of autonomic dysfunction in ME/CFS,” Whelan said.
Although 22% of patients in the study group had POTS and 59% had OI, the authors found no correlation between autoantibody levels and either OI or POTS. However, 38% were confirmed to have small-fiber polyneuropathy on skin biopsy, and the vast majority of those patients (93%) had either POTS or OI.
IVIG May Be a Potential Treatment
Whelan notes that some data suggest that IVIG might help patients with small-fiber neuropathy, including those with autoimmunity.
In addition, he described anecdotal data from a single patient with ME/CFS who had neuropathic symptoms. The patient was treated at Simmaron. The 56-year-old received two IVIG infusions given 6 months apart. The patient experienced a dramatic reduction in levels of all four of the relevant autoantibodies and favorable symptom reduction, as shown in clinician follow-up records. “With the success of this case study, we intend to further evaluate IVIG as a potential treatment in ME/CFS patients. With this research, we hope to identify a subset of ME/CFS patients who will respond favorably to IVIG,” Whelan concluded.
Regarding use of IVIG, Chu commented, “We don’t know exactly how it works, but it seems to help certain conditions.” She pointed to another recent small study that reported clinical improvement in patients with ME/CFS through a different approach, immunoadsorption, for reducing the autoantibody levels.
Overall, Chu said, this line of research “is important because it shows there’s some type of abnormal biomarker for ME/CFS. And, it may lay a path toward understanding the pathophysiology of the disease and why people have certain symptoms, and could be used to target therapies. ... It’s intriguing.”
Whelan and Chu have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A significant proportion of patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia may have potentially treatable underlying autoimmune-associated small-fiber polyneuropathy (aaSFPN), pilot data suggest.
The findings, from a single-site study of 61 patients with ME/CFS, were presented August 21 at the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis by Ryan Whelan, BS, a research assistant at Simmaron Research Institute, Incline Village, Nevada.
Recent evidence suggests an autoimmune etiology for some patients with ME/CFS, which is defined as experiencing for a period of at least 6 months profound, unexplained fatigue, postexertional malaise, and unrefreshing sleep, as well as cognitive dysfunction and/or orthostatic intolerance (OI).
OI is part of a spectrum of autonomic dysfunction commonly seen in ME/CFS patients, which may also include postural orthostatic tachycardia (POTS), peripheral temperature dysregulation and light sensitivity, neuropathic pain, and gastrointestinal complaints. Many of these symptoms overlap those reported by patients with aaSFPN, a common but underdiagnosed neurodegenerative disorder characterized by the loss of peripheral autonomic nerve fibers, Whelan explained.
Findings from the current study show that in more than half of ME/CFS patients, levels of at least one autoantibody were elevated. A majority had comorbid POTS or OI, and over a third had biopsy-confirmed aaSFPN.
“Given the overlap of symptoms and common etiological basis, it may be important to identify ME/CFS patients who present with comorbid aaSFPN, as it has been shown that immune modulatory agents, including intravenous gamma globulin [IVIG], reduce the autonomic symptom burden in aaSFPN patients,” Whelan said.
He noted that Anne Louise Oaklander, MD, a neurologist at Massachusetts General Hospital, Harvard Medical School, Boston, and colleagues previously linked aaSFPN with fibromyalgia. In addition, they’ve found a connection between small-fiber dysfunction and postexertional malaise, which is a hallmark ME/CFS symptom.
Asked to comment on Whelan’s presentation, IACFSME co-president Lily Chu, MD, told Medscape Medical News that the new findings are “valuable, because ME/CFS has always been looked upon as just subjective symptoms. When people have laboratory abnormalities, it can be due to a bunch of other causes, but...here’s pathology, here’s a biopsy of actual damage. It’s not just a transient finding. You can actually see it. ... It’s a solid concrete piece of evidence vs something that can fluctuate.”
Autoantibodies, Autonomic Dysfunction, and Small-Fiber Polyneuropathy
Whelan and colleagues conducted an extensive analysis of medical records of 364 patients with ME/CFS (72% female) to identify potential aaSFPN comorbidity. Such identifications were made on the basis of progress notes documenting autonomic dysfunction, laboratory results for serum autoantibodies, and questionnaire symptom self-reports.
They identified 61 patients as possibly having comorbid aaSFPN. Of those, 52% tested positive for at least 1 of 4 autoantibodies, including antimuscarinic cholinergic receptor 4 (47%), anti-beta-2 adrenergic (27%), antimuscarinic cholinergic 3 (25%), and anti-beta-1 adrenergic (13%). These autoantibodies were linked to ME/CFS in a recent Swedish cohort study.
“Evidence supports that these autoantibodies may bind to receptor sites, blocking ligands from reaching these receptors. Disturbances of adrenergic and cholinergic receptors by these autoantibodies may contribute to symptoms of autonomic dysfunction in ME/CFS,” Whelan said.
Although 22% of patients in the study group had POTS and 59% had OI, the authors found no correlation between autoantibody levels and either OI or POTS. However, 38% were confirmed to have small-fiber polyneuropathy on skin biopsy, and the vast majority of those patients (93%) had either POTS or OI.
IVIG May Be a Potential Treatment
Whelan notes that some data suggest that IVIG might help patients with small-fiber neuropathy, including those with autoimmunity.
In addition, he described anecdotal data from a single patient with ME/CFS who had neuropathic symptoms. The patient was treated at Simmaron. The 56-year-old received two IVIG infusions given 6 months apart. The patient experienced a dramatic reduction in levels of all four of the relevant autoantibodies and favorable symptom reduction, as shown in clinician follow-up records. “With the success of this case study, we intend to further evaluate IVIG as a potential treatment in ME/CFS patients. With this research, we hope to identify a subset of ME/CFS patients who will respond favorably to IVIG,” Whelan concluded.
Regarding use of IVIG, Chu commented, “We don’t know exactly how it works, but it seems to help certain conditions.” She pointed to another recent small study that reported clinical improvement in patients with ME/CFS through a different approach, immunoadsorption, for reducing the autoantibody levels.
Overall, Chu said, this line of research “is important because it shows there’s some type of abnormal biomarker for ME/CFS. And, it may lay a path toward understanding the pathophysiology of the disease and why people have certain symptoms, and could be used to target therapies. ... It’s intriguing.”
Whelan and Chu have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A significant proportion of patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia may have potentially treatable underlying autoimmune-associated small-fiber polyneuropathy (aaSFPN), pilot data suggest.
The findings, from a single-site study of 61 patients with ME/CFS, were presented August 21 at the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis by Ryan Whelan, BS, a research assistant at Simmaron Research Institute, Incline Village, Nevada.
Recent evidence suggests an autoimmune etiology for some patients with ME/CFS, which is defined as experiencing for a period of at least 6 months profound, unexplained fatigue, postexertional malaise, and unrefreshing sleep, as well as cognitive dysfunction and/or orthostatic intolerance (OI).
OI is part of a spectrum of autonomic dysfunction commonly seen in ME/CFS patients, which may also include postural orthostatic tachycardia (POTS), peripheral temperature dysregulation and light sensitivity, neuropathic pain, and gastrointestinal complaints. Many of these symptoms overlap those reported by patients with aaSFPN, a common but underdiagnosed neurodegenerative disorder characterized by the loss of peripheral autonomic nerve fibers, Whelan explained.
Findings from the current study show that in more than half of ME/CFS patients, levels of at least one autoantibody were elevated. A majority had comorbid POTS or OI, and over a third had biopsy-confirmed aaSFPN.
“Given the overlap of symptoms and common etiological basis, it may be important to identify ME/CFS patients who present with comorbid aaSFPN, as it has been shown that immune modulatory agents, including intravenous gamma globulin [IVIG], reduce the autonomic symptom burden in aaSFPN patients,” Whelan said.
He noted that Anne Louise Oaklander, MD, a neurologist at Massachusetts General Hospital, Harvard Medical School, Boston, and colleagues previously linked aaSFPN with fibromyalgia. In addition, they’ve found a connection between small-fiber dysfunction and postexertional malaise, which is a hallmark ME/CFS symptom.
Asked to comment on Whelan’s presentation, IACFSME co-president Lily Chu, MD, told Medscape Medical News that the new findings are “valuable, because ME/CFS has always been looked upon as just subjective symptoms. When people have laboratory abnormalities, it can be due to a bunch of other causes, but...here’s pathology, here’s a biopsy of actual damage. It’s not just a transient finding. You can actually see it. ... It’s a solid concrete piece of evidence vs something that can fluctuate.”
Autoantibodies, Autonomic Dysfunction, and Small-Fiber Polyneuropathy
Whelan and colleagues conducted an extensive analysis of medical records of 364 patients with ME/CFS (72% female) to identify potential aaSFPN comorbidity. Such identifications were made on the basis of progress notes documenting autonomic dysfunction, laboratory results for serum autoantibodies, and questionnaire symptom self-reports.
They identified 61 patients as possibly having comorbid aaSFPN. Of those, 52% tested positive for at least 1 of 4 autoantibodies, including antimuscarinic cholinergic receptor 4 (47%), anti-beta-2 adrenergic (27%), antimuscarinic cholinergic 3 (25%), and anti-beta-1 adrenergic (13%). These autoantibodies were linked to ME/CFS in a recent Swedish cohort study.
“Evidence supports that these autoantibodies may bind to receptor sites, blocking ligands from reaching these receptors. Disturbances of adrenergic and cholinergic receptors by these autoantibodies may contribute to symptoms of autonomic dysfunction in ME/CFS,” Whelan said.
Although 22% of patients in the study group had POTS and 59% had OI, the authors found no correlation between autoantibody levels and either OI or POTS. However, 38% were confirmed to have small-fiber polyneuropathy on skin biopsy, and the vast majority of those patients (93%) had either POTS or OI.
IVIG May Be a Potential Treatment
Whelan notes that some data suggest that IVIG might help patients with small-fiber neuropathy, including those with autoimmunity.
In addition, he described anecdotal data from a single patient with ME/CFS who had neuropathic symptoms. The patient was treated at Simmaron. The 56-year-old received two IVIG infusions given 6 months apart. The patient experienced a dramatic reduction in levels of all four of the relevant autoantibodies and favorable symptom reduction, as shown in clinician follow-up records. “With the success of this case study, we intend to further evaluate IVIG as a potential treatment in ME/CFS patients. With this research, we hope to identify a subset of ME/CFS patients who will respond favorably to IVIG,” Whelan concluded.
Regarding use of IVIG, Chu commented, “We don’t know exactly how it works, but it seems to help certain conditions.” She pointed to another recent small study that reported clinical improvement in patients with ME/CFS through a different approach, immunoadsorption, for reducing the autoantibody levels.
Overall, Chu said, this line of research “is important because it shows there’s some type of abnormal biomarker for ME/CFS. And, it may lay a path toward understanding the pathophysiology of the disease and why people have certain symptoms, and could be used to target therapies. ... It’s intriguing.”
Whelan and Chu have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Psoriasis, PsA, and pregnancy: Tailoring treatment with increasing data
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
The long road to a PsA prevention trial
About one-third of all patients with psoriasis will develop psoriatic arthritis (PsA), a condition that comes with a host of vague symptoms and no definitive blood test for diagnosis. Prevention trials could help to identify higher-risk groups for PsA, with a goal to catch disease early and improve outcomes. The challenge is finding enough participants in a disease that lacks a clear clinical profile, then tracking them for long periods of time to generate any significant data.
Researchers have been taking several approaches to improve outcomes in PsA, Christopher Ritchlin, MD, MPH, chief of the allergy/immunology and rheumatology division at the University of Rochester (N.Y.), said in an interview. “We are in the process of identifying biomarkers and imaging findings that characterize psoriasis patients at high risk to develop PsA.”
The next step would be to design an interventional trial to treat high-risk patients before they develop musculoskeletal inflammation, with a goal to delay onset, attenuate severity, or completely prevent arthritis. The issue now is “we don’t know which agents would be most effective in this prevention effort,” Dr. Ritchlin said. Biologics that target specific pathways significant in PsA pathogenesis are an appealing prospect. However, “it may be that alternative therapies such as methotrexate or ultraviolet A radiation therapy, for example, may help arrest the development of joint inflammation.”
Underdiagnosis impedes research
Several factors may undermine this important research.
For one, psoriasis patients are often unaware that they have PsA. “Many times they are diagnosed incorrectly by nonspecialists. As a consequence, they do not get access to appropriate medications,” said Lihi Eder, MD, PhD, staff rheumatologist and director of the psoriatic arthritis research program at the University of Toronto’s Women’s College Research Institute.
The condition also lacks a good diagnostic tool, Dr. Eder said. There’s no blood test that identifies this condition in the same manner as RA and lupus, for example. For these conditions, a general practitioner such as a family physician may conduct a blood test, and if positive, refer them to a rheumatologist. Such a system doesn’t exist for PsA. “Instead, nonspecialists are ordering tests and when they’re negative, they assume wrongly that these patients don’t have a rheumatic condition,” she explained.
Many clinicians aren’t that well versed in PsA, although dermatology has taken steps to become better educated. As a result, more dermatologists are referring patients to rheumatologists for PsA. Despite this small step forward, the heterogeneous clinical presentation of this condition makes diagnosis especially difficult. Unlike RA, which presents with inflammation in the joints, PsA can present as back or joint pain, “which makes our life as rheumatologists much more complex,” Dr. Eder said.
Defining a risk group
Most experts agree that the presence of psoriasis isn’t sufficient to conduct a prevention trial. Ideally, the goal of a prevention study would be to identify a critical subgroup of psoriasis patients at high risk of developing PsA.
However, this presents a challenging task, Dr. Eder said. Psoriasis is a risk factor for PsA, but most patients with psoriasis won’t actually develop it. Given that there’s an incidence rate of 2.7% a year, “you would need to recruit many hundreds of psoriasis patients and follow them for a long period of time until you have enough events.”
Moving forward with prevention studies calls for a better definition of the PsA risk group, according to Georg Schett, MD, chair of internal medicine in the department of internal medicine, rheumatology, and immunology at Friedrich‐Alexander University, Erlangen, Germany. “That’s very important, because you need to define such a group to make a prevention trial feasible. The whole benefit of such an approach is to catch the disease early, to say that psoriasis is a biomarker that’s linked to psoriatic arthritis.”
Indicators of risk other than psoriasis, such as pain, inflammation seen in ultrasound or MRI, and other specificities of psoriasis, could be used to define a population where interception can take place, Dr. Schett added. Although it’s not always clinically recognized, the combination of pain and structural lesions can be an indicator for developing PsA.
One prospective study he and his colleagues conducted in 114 psoriasis patients cited structural entheseal lesions and low cortical volumetric bone mineral density as risk factors in developing PsA. Keeping these factors in mind, Dr. Schett expects to see more studies in biointervention in these populations, “with the idea to prevent the onset of PsA and also decrease pain and subcutaneous inflammation.”
Researchers are currently working to identify those high-risk patients to include in an interventional trial, Dr. Ritchlin said.
That said, there’s been a great deal of “clinical trial angst” among investigators, Dr. Ritchlin noted. Outcomes in clinical trials for a wide range of biologic agents have not demonstrated significant advances in outcomes, compared with initial studies with anti–tumor necrosis factor–alpha (TNF-alpha) agents 20 years ago.
Combination biologics
One approach that’s generated some interest is the use of combination biologics medications. Sequential inhibition of cytokines such as interleukin-17A and TNF-alpha is of interest given their central contribution in joint inflammation and damage. “The challenge here of course is toxicity,” Dr. Ritchlin said. Trials that combined blockade of IL-1 and TNF-alpha in a RA trial years ago resulted in significant adverse events without improving outcomes.
Comparatively, a recent study in The Lancet Rheumatology reported success in using the IL-17A inhibitor secukinumab (Cosentyx) to reduce PsA symptoms. Tested on patients in the FUTURE 2 trial, investigators demonstrated that secukinumab in 300- and 150-mg doses safely reduced PsA signs and symptoms over a period of 5 years. Secukinumab also outperformed the TNF-alpha inhibitor adalimumab in 853 PsA patients in the 52-week, randomized, head-to-head, phase 3b EXCEED study, which was recently reported in The Lancet. Articular outcomes were similar between the two therapies, yet the secukinumab group did markedly better in Psoriasis Area and Severity Index scores, compared with the adalimumab group.
Based on these findings, “I suspect that studies examining the efficacy of combination biologics for treatment of PsA will surface in the near future,” Dr. Ritchlin said.
Yet another approach encompasses the spirit of personalized medicine. Clinicians often treat PsA patients empirically because they lack biomarkers that indicate which drug may be most effective for an individual patient, Dr. Ritchlin said. However, the technologies for investigating specific cell subsets in both the blood and tissues have advanced greatly over the last decade. “I am confident that a more precision medicine–based approach to the diagnosis and treatment of PsA is on the near horizon.”
Diet as an intervention
Other research has looked at the strong link between metabolic abnormalities and psoriasis and PsA. Some diets, such as the Mediterranean diet, show promise in improving the metabolic profile of these patients, making it a candidate as a potential intervention to reduce PsA risk. Another strategy would be to focus on limiting calories and promoting weight reduction.
One study in the British Journal of Dermatology looked at the associations between PsA and smoking, alcohol, and body mass index, identifying obesity as an important risk factor. Analyzing more than 90,000 psoriasis cases from the U.K. Clinical Practice Research Datalink between 1998 and 2014, researchers identified 1,409 PsA diagnoses. Among this cohort, researchers found an association between PsA and increased body mass index and moderate drinking. This finding underscores the need to support weight-reduction programs to reduce risk, Dr. Eder and Alexis Ogdie, MD, of the University of Pennsylvania, Philadelphia, wrote in a related editorial.
While observational studies such as this one provide further guidance for interventional trials, confounders can affect results. “Patients who lost weight could have made a positive lifestyle change (e.g., a dietary change) that was associated with the decreased risk for PsA rather than weight loss specifically, or they could have lost weight for unhealthy reasons,” Dr. Eder and Dr. Ogdie explained. Future research could address whether weight loss or other interventional factors may reduce PsA progression.
To make this work, “we would need to select patients that would benefit from diet. Secondly, we’d need to identify what kind of diet would be good for preventing PsA. And we don’t know that yet,” Dr. Eder further elaborated.
As with any prevention trial, the challenge is to follow patients over a long period of time, making sure they comply with the restrictions of the prescribed diet, Dr. Eder noted. “I do think it’s a really exciting type of intervention because it’s something that people are very interested in. There’s little risk of side effects, and it’s not very expensive.”
In other research on weight-loss methods, an observational study from Denmark found that bariatric surgery, especially gastric bypass, reduced the risk of developing PsA. This suggests that weight reduction by itself is important, “although we don’t know that yet,” Dr. Eder said.
A risk model for PsA
Dr. Eder and colleagues have been working on an algorithm that will incorporate clinical information (for example, the presence of nail lesions and the severity of psoriasis) to provide an estimated risk of developing PsA over the next 5 years. Subsequently, this information could be used to identify high-risk psoriasis patients as candidates for a prevention trial.
Other groups are looking at laboratory or imaging biomarkers to help develop PsA prediction models, she said. “Once we have these tools, we can move to next steps of prevention trials. What kinds of interventions should we apply? Are we talking biologic medications or other lifestyle interventions like diet? We are still at the early stages. However, with an improved understanding of the underlying mechanisms and risk factors we are expected to see prevention trials for PsA in the future.”
About one-third of all patients with psoriasis will develop psoriatic arthritis (PsA), a condition that comes with a host of vague symptoms and no definitive blood test for diagnosis. Prevention trials could help to identify higher-risk groups for PsA, with a goal to catch disease early and improve outcomes. The challenge is finding enough participants in a disease that lacks a clear clinical profile, then tracking them for long periods of time to generate any significant data.
Researchers have been taking several approaches to improve outcomes in PsA, Christopher Ritchlin, MD, MPH, chief of the allergy/immunology and rheumatology division at the University of Rochester (N.Y.), said in an interview. “We are in the process of identifying biomarkers and imaging findings that characterize psoriasis patients at high risk to develop PsA.”
The next step would be to design an interventional trial to treat high-risk patients before they develop musculoskeletal inflammation, with a goal to delay onset, attenuate severity, or completely prevent arthritis. The issue now is “we don’t know which agents would be most effective in this prevention effort,” Dr. Ritchlin said. Biologics that target specific pathways significant in PsA pathogenesis are an appealing prospect. However, “it may be that alternative therapies such as methotrexate or ultraviolet A radiation therapy, for example, may help arrest the development of joint inflammation.”
Underdiagnosis impedes research
Several factors may undermine this important research.
For one, psoriasis patients are often unaware that they have PsA. “Many times they are diagnosed incorrectly by nonspecialists. As a consequence, they do not get access to appropriate medications,” said Lihi Eder, MD, PhD, staff rheumatologist and director of the psoriatic arthritis research program at the University of Toronto’s Women’s College Research Institute.
The condition also lacks a good diagnostic tool, Dr. Eder said. There’s no blood test that identifies this condition in the same manner as RA and lupus, for example. For these conditions, a general practitioner such as a family physician may conduct a blood test, and if positive, refer them to a rheumatologist. Such a system doesn’t exist for PsA. “Instead, nonspecialists are ordering tests and when they’re negative, they assume wrongly that these patients don’t have a rheumatic condition,” she explained.
Many clinicians aren’t that well versed in PsA, although dermatology has taken steps to become better educated. As a result, more dermatologists are referring patients to rheumatologists for PsA. Despite this small step forward, the heterogeneous clinical presentation of this condition makes diagnosis especially difficult. Unlike RA, which presents with inflammation in the joints, PsA can present as back or joint pain, “which makes our life as rheumatologists much more complex,” Dr. Eder said.
Defining a risk group
Most experts agree that the presence of psoriasis isn’t sufficient to conduct a prevention trial. Ideally, the goal of a prevention study would be to identify a critical subgroup of psoriasis patients at high risk of developing PsA.
However, this presents a challenging task, Dr. Eder said. Psoriasis is a risk factor for PsA, but most patients with psoriasis won’t actually develop it. Given that there’s an incidence rate of 2.7% a year, “you would need to recruit many hundreds of psoriasis patients and follow them for a long period of time until you have enough events.”
Moving forward with prevention studies calls for a better definition of the PsA risk group, according to Georg Schett, MD, chair of internal medicine in the department of internal medicine, rheumatology, and immunology at Friedrich‐Alexander University, Erlangen, Germany. “That’s very important, because you need to define such a group to make a prevention trial feasible. The whole benefit of such an approach is to catch the disease early, to say that psoriasis is a biomarker that’s linked to psoriatic arthritis.”
Indicators of risk other than psoriasis, such as pain, inflammation seen in ultrasound or MRI, and other specificities of psoriasis, could be used to define a population where interception can take place, Dr. Schett added. Although it’s not always clinically recognized, the combination of pain and structural lesions can be an indicator for developing PsA.
One prospective study he and his colleagues conducted in 114 psoriasis patients cited structural entheseal lesions and low cortical volumetric bone mineral density as risk factors in developing PsA. Keeping these factors in mind, Dr. Schett expects to see more studies in biointervention in these populations, “with the idea to prevent the onset of PsA and also decrease pain and subcutaneous inflammation.”
Researchers are currently working to identify those high-risk patients to include in an interventional trial, Dr. Ritchlin said.
That said, there’s been a great deal of “clinical trial angst” among investigators, Dr. Ritchlin noted. Outcomes in clinical trials for a wide range of biologic agents have not demonstrated significant advances in outcomes, compared with initial studies with anti–tumor necrosis factor–alpha (TNF-alpha) agents 20 years ago.
Combination biologics
One approach that’s generated some interest is the use of combination biologics medications. Sequential inhibition of cytokines such as interleukin-17A and TNF-alpha is of interest given their central contribution in joint inflammation and damage. “The challenge here of course is toxicity,” Dr. Ritchlin said. Trials that combined blockade of IL-1 and TNF-alpha in a RA trial years ago resulted in significant adverse events without improving outcomes.
Comparatively, a recent study in The Lancet Rheumatology reported success in using the IL-17A inhibitor secukinumab (Cosentyx) to reduce PsA symptoms. Tested on patients in the FUTURE 2 trial, investigators demonstrated that secukinumab in 300- and 150-mg doses safely reduced PsA signs and symptoms over a period of 5 years. Secukinumab also outperformed the TNF-alpha inhibitor adalimumab in 853 PsA patients in the 52-week, randomized, head-to-head, phase 3b EXCEED study, which was recently reported in The Lancet. Articular outcomes were similar between the two therapies, yet the secukinumab group did markedly better in Psoriasis Area and Severity Index scores, compared with the adalimumab group.
Based on these findings, “I suspect that studies examining the efficacy of combination biologics for treatment of PsA will surface in the near future,” Dr. Ritchlin said.
Yet another approach encompasses the spirit of personalized medicine. Clinicians often treat PsA patients empirically because they lack biomarkers that indicate which drug may be most effective for an individual patient, Dr. Ritchlin said. However, the technologies for investigating specific cell subsets in both the blood and tissues have advanced greatly over the last decade. “I am confident that a more precision medicine–based approach to the diagnosis and treatment of PsA is on the near horizon.”
Diet as an intervention
Other research has looked at the strong link between metabolic abnormalities and psoriasis and PsA. Some diets, such as the Mediterranean diet, show promise in improving the metabolic profile of these patients, making it a candidate as a potential intervention to reduce PsA risk. Another strategy would be to focus on limiting calories and promoting weight reduction.
One study in the British Journal of Dermatology looked at the associations between PsA and smoking, alcohol, and body mass index, identifying obesity as an important risk factor. Analyzing more than 90,000 psoriasis cases from the U.K. Clinical Practice Research Datalink between 1998 and 2014, researchers identified 1,409 PsA diagnoses. Among this cohort, researchers found an association between PsA and increased body mass index and moderate drinking. This finding underscores the need to support weight-reduction programs to reduce risk, Dr. Eder and Alexis Ogdie, MD, of the University of Pennsylvania, Philadelphia, wrote in a related editorial.
While observational studies such as this one provide further guidance for interventional trials, confounders can affect results. “Patients who lost weight could have made a positive lifestyle change (e.g., a dietary change) that was associated with the decreased risk for PsA rather than weight loss specifically, or they could have lost weight for unhealthy reasons,” Dr. Eder and Dr. Ogdie explained. Future research could address whether weight loss or other interventional factors may reduce PsA progression.
To make this work, “we would need to select patients that would benefit from diet. Secondly, we’d need to identify what kind of diet would be good for preventing PsA. And we don’t know that yet,” Dr. Eder further elaborated.
As with any prevention trial, the challenge is to follow patients over a long period of time, making sure they comply with the restrictions of the prescribed diet, Dr. Eder noted. “I do think it’s a really exciting type of intervention because it’s something that people are very interested in. There’s little risk of side effects, and it’s not very expensive.”
In other research on weight-loss methods, an observational study from Denmark found that bariatric surgery, especially gastric bypass, reduced the risk of developing PsA. This suggests that weight reduction by itself is important, “although we don’t know that yet,” Dr. Eder said.
A risk model for PsA
Dr. Eder and colleagues have been working on an algorithm that will incorporate clinical information (for example, the presence of nail lesions and the severity of psoriasis) to provide an estimated risk of developing PsA over the next 5 years. Subsequently, this information could be used to identify high-risk psoriasis patients as candidates for a prevention trial.
Other groups are looking at laboratory or imaging biomarkers to help develop PsA prediction models, she said. “Once we have these tools, we can move to next steps of prevention trials. What kinds of interventions should we apply? Are we talking biologic medications or other lifestyle interventions like diet? We are still at the early stages. However, with an improved understanding of the underlying mechanisms and risk factors we are expected to see prevention trials for PsA in the future.”
About one-third of all patients with psoriasis will develop psoriatic arthritis (PsA), a condition that comes with a host of vague symptoms and no definitive blood test for diagnosis. Prevention trials could help to identify higher-risk groups for PsA, with a goal to catch disease early and improve outcomes. The challenge is finding enough participants in a disease that lacks a clear clinical profile, then tracking them for long periods of time to generate any significant data.
Researchers have been taking several approaches to improve outcomes in PsA, Christopher Ritchlin, MD, MPH, chief of the allergy/immunology and rheumatology division at the University of Rochester (N.Y.), said in an interview. “We are in the process of identifying biomarkers and imaging findings that characterize psoriasis patients at high risk to develop PsA.”
The next step would be to design an interventional trial to treat high-risk patients before they develop musculoskeletal inflammation, with a goal to delay onset, attenuate severity, or completely prevent arthritis. The issue now is “we don’t know which agents would be most effective in this prevention effort,” Dr. Ritchlin said. Biologics that target specific pathways significant in PsA pathogenesis are an appealing prospect. However, “it may be that alternative therapies such as methotrexate or ultraviolet A radiation therapy, for example, may help arrest the development of joint inflammation.”
Underdiagnosis impedes research
Several factors may undermine this important research.
For one, psoriasis patients are often unaware that they have PsA. “Many times they are diagnosed incorrectly by nonspecialists. As a consequence, they do not get access to appropriate medications,” said Lihi Eder, MD, PhD, staff rheumatologist and director of the psoriatic arthritis research program at the University of Toronto’s Women’s College Research Institute.
The condition also lacks a good diagnostic tool, Dr. Eder said. There’s no blood test that identifies this condition in the same manner as RA and lupus, for example. For these conditions, a general practitioner such as a family physician may conduct a blood test, and if positive, refer them to a rheumatologist. Such a system doesn’t exist for PsA. “Instead, nonspecialists are ordering tests and when they’re negative, they assume wrongly that these patients don’t have a rheumatic condition,” she explained.
Many clinicians aren’t that well versed in PsA, although dermatology has taken steps to become better educated. As a result, more dermatologists are referring patients to rheumatologists for PsA. Despite this small step forward, the heterogeneous clinical presentation of this condition makes diagnosis especially difficult. Unlike RA, which presents with inflammation in the joints, PsA can present as back or joint pain, “which makes our life as rheumatologists much more complex,” Dr. Eder said.
Defining a risk group
Most experts agree that the presence of psoriasis isn’t sufficient to conduct a prevention trial. Ideally, the goal of a prevention study would be to identify a critical subgroup of psoriasis patients at high risk of developing PsA.
However, this presents a challenging task, Dr. Eder said. Psoriasis is a risk factor for PsA, but most patients with psoriasis won’t actually develop it. Given that there’s an incidence rate of 2.7% a year, “you would need to recruit many hundreds of psoriasis patients and follow them for a long period of time until you have enough events.”
Moving forward with prevention studies calls for a better definition of the PsA risk group, according to Georg Schett, MD, chair of internal medicine in the department of internal medicine, rheumatology, and immunology at Friedrich‐Alexander University, Erlangen, Germany. “That’s very important, because you need to define such a group to make a prevention trial feasible. The whole benefit of such an approach is to catch the disease early, to say that psoriasis is a biomarker that’s linked to psoriatic arthritis.”
Indicators of risk other than psoriasis, such as pain, inflammation seen in ultrasound or MRI, and other specificities of psoriasis, could be used to define a population where interception can take place, Dr. Schett added. Although it’s not always clinically recognized, the combination of pain and structural lesions can be an indicator for developing PsA.
One prospective study he and his colleagues conducted in 114 psoriasis patients cited structural entheseal lesions and low cortical volumetric bone mineral density as risk factors in developing PsA. Keeping these factors in mind, Dr. Schett expects to see more studies in biointervention in these populations, “with the idea to prevent the onset of PsA and also decrease pain and subcutaneous inflammation.”
Researchers are currently working to identify those high-risk patients to include in an interventional trial, Dr. Ritchlin said.
That said, there’s been a great deal of “clinical trial angst” among investigators, Dr. Ritchlin noted. Outcomes in clinical trials for a wide range of biologic agents have not demonstrated significant advances in outcomes, compared with initial studies with anti–tumor necrosis factor–alpha (TNF-alpha) agents 20 years ago.
Combination biologics
One approach that’s generated some interest is the use of combination biologics medications. Sequential inhibition of cytokines such as interleukin-17A and TNF-alpha is of interest given their central contribution in joint inflammation and damage. “The challenge here of course is toxicity,” Dr. Ritchlin said. Trials that combined blockade of IL-1 and TNF-alpha in a RA trial years ago resulted in significant adverse events without improving outcomes.
Comparatively, a recent study in The Lancet Rheumatology reported success in using the IL-17A inhibitor secukinumab (Cosentyx) to reduce PsA symptoms. Tested on patients in the FUTURE 2 trial, investigators demonstrated that secukinumab in 300- and 150-mg doses safely reduced PsA signs and symptoms over a period of 5 years. Secukinumab also outperformed the TNF-alpha inhibitor adalimumab in 853 PsA patients in the 52-week, randomized, head-to-head, phase 3b EXCEED study, which was recently reported in The Lancet. Articular outcomes were similar between the two therapies, yet the secukinumab group did markedly better in Psoriasis Area and Severity Index scores, compared with the adalimumab group.
Based on these findings, “I suspect that studies examining the efficacy of combination biologics for treatment of PsA will surface in the near future,” Dr. Ritchlin said.
Yet another approach encompasses the spirit of personalized medicine. Clinicians often treat PsA patients empirically because they lack biomarkers that indicate which drug may be most effective for an individual patient, Dr. Ritchlin said. However, the technologies for investigating specific cell subsets in both the blood and tissues have advanced greatly over the last decade. “I am confident that a more precision medicine–based approach to the diagnosis and treatment of PsA is on the near horizon.”
Diet as an intervention
Other research has looked at the strong link between metabolic abnormalities and psoriasis and PsA. Some diets, such as the Mediterranean diet, show promise in improving the metabolic profile of these patients, making it a candidate as a potential intervention to reduce PsA risk. Another strategy would be to focus on limiting calories and promoting weight reduction.
One study in the British Journal of Dermatology looked at the associations between PsA and smoking, alcohol, and body mass index, identifying obesity as an important risk factor. Analyzing more than 90,000 psoriasis cases from the U.K. Clinical Practice Research Datalink between 1998 and 2014, researchers identified 1,409 PsA diagnoses. Among this cohort, researchers found an association between PsA and increased body mass index and moderate drinking. This finding underscores the need to support weight-reduction programs to reduce risk, Dr. Eder and Alexis Ogdie, MD, of the University of Pennsylvania, Philadelphia, wrote in a related editorial.
While observational studies such as this one provide further guidance for interventional trials, confounders can affect results. “Patients who lost weight could have made a positive lifestyle change (e.g., a dietary change) that was associated with the decreased risk for PsA rather than weight loss specifically, or they could have lost weight for unhealthy reasons,” Dr. Eder and Dr. Ogdie explained. Future research could address whether weight loss or other interventional factors may reduce PsA progression.
To make this work, “we would need to select patients that would benefit from diet. Secondly, we’d need to identify what kind of diet would be good for preventing PsA. And we don’t know that yet,” Dr. Eder further elaborated.
As with any prevention trial, the challenge is to follow patients over a long period of time, making sure they comply with the restrictions of the prescribed diet, Dr. Eder noted. “I do think it’s a really exciting type of intervention because it’s something that people are very interested in. There’s little risk of side effects, and it’s not very expensive.”
In other research on weight-loss methods, an observational study from Denmark found that bariatric surgery, especially gastric bypass, reduced the risk of developing PsA. This suggests that weight reduction by itself is important, “although we don’t know that yet,” Dr. Eder said.
A risk model for PsA
Dr. Eder and colleagues have been working on an algorithm that will incorporate clinical information (for example, the presence of nail lesions and the severity of psoriasis) to provide an estimated risk of developing PsA over the next 5 years. Subsequently, this information could be used to identify high-risk psoriasis patients as candidates for a prevention trial.
Other groups are looking at laboratory or imaging biomarkers to help develop PsA prediction models, she said. “Once we have these tools, we can move to next steps of prevention trials. What kinds of interventions should we apply? Are we talking biologic medications or other lifestyle interventions like diet? We are still at the early stages. However, with an improved understanding of the underlying mechanisms and risk factors we are expected to see prevention trials for PsA in the future.”