User login
Long-term efficacy, safety data for ixekizumab in pediatric psoriasis reported
with the interleukin (IL)-17 inhibitor, investigators reported.
In addition, findings of a substudy, which evaluated randomized withdrawal of treatment after 60 weeks, suggest patients were able to regain benefit after not being treated for a period.
Ixekizumab (Taltz) was approved by the U.S. Food and Drug Administration for treating pediatric psoriasis in March 2020 for patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The trial (IXORA-PEDS) involved 171 patients aged 6-17 years (mean age, 13.5 years; 99 females and 72 males), who were randomly assigned to receive ixekizumab via subcutaneous administration every 4 weeks (115) or placebo for 12 weeks (56). Thereafter, 166 patients continued in an open-label maintenance period in which they were treated every 4 weeks for 12-60 weeks. This was followed by an extension period of up to 108 weeks, which was completed by 139 patients (83.7%). At baseline, the patients’ Psoriasis Area and Severity Index (PASI) score was 12 or higher, the static Physician’s Global Assessment (sPGA) score was 3 or higher, and 10% or more of body surface area was affected.
In the study, at 12 weeks, treatment with ixekizumab was superior to placebo, with sustained responses through 48 weeks. In the follow-up phase, primary and secondary endpoints were sustained through week 108, with patients achieving or maintaining PASI 75 (91.7%), PASI 90 (79%), PASI 100 (55.1%), sPGA 0 or 1 (78.3%), and sPGA 0 (52.4%). Significant improvements in itch were seen at 12 weeks and were sustained with “meaningful improvements in itch for 78.5% of these patients at week 108,” the investigators report.
Among the patients who received ixekizumab, clearance rates in areas that are difficult to treat increased from week 12 to week 108 among those affected. During this time, clearance of nail psoriasis increased from 22.8% to 68.1%, clearance of palmoplantar psoriasis increased from 46.2% to 90%, clearance of scalp psoriasis increased from 70.7% to 76.2%, and clearance of genital psoriasis increased from 83.3% to 87.5%.
No new safety findings during weeks 48-108 of the trial were reported, including no new cases of inflammatory bowel disease (IBD) or Candida infections. The results were reported in JAMA Dermatology.
“Safety is really what we think of most when we are talking about pediatric patients, especially since they may be on these for decades and ... since they most commonly start these therapies in adolescence,” said Amy Paller, MD, the study’s lead author, in an interview. “To be able to take this out 108 weeks, 2 years, is starting to get to a point where we are getting more comfortable with safety. Clearly, no new signals arose.” Dr. Paller is chair of the department of dermatology and professor of dermatology and pediatrics, Northwestern University, Chicago.
One of the biggest concerns with using IL-17 inhibitors such as ixekizumab to manage psoriasis is the development of IBD, said Dr. Paller. She noted that four cases of IBD were reported before the extension phase of the trial but that no new IBD cases were reported after week 48.
“We would not start this as a treatment of choice in someone with Crohn’s disease, or perhaps we would think twice about using it in someone with a strong family history [of Crohn’s disease],” said Dr. Paller, who is also the director of the Skin Biology and Diseases Resource-Based Center at Northwestern. “Otherwise, it does not make me concerned about its use.”
Commenting on the study, Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, said that the trial’s results provide additional evidence regarding the optimal management of pediatric psoriasis.
“The landscape has shifted toward involving more pediatric patients in clinical trials, thereby providing dermatologists with data to select safe and effective therapies to manage children with psoriasis,” Dr. Cordoro said in an interview. “We have data showing that children with psoriasis have been undertreated, likely because of concerns about safety. The more evidence available from trials such as this, the more likely children are to receive necessary treatment.”
The efficacy data from the study on difficult-to-treat areas of psoriasis, in addition to improvements in BSA and PASI measures, are significant for clinicians deciding on a therapy for patients with psoriasis concentrated in specific body sites. “It was very valuable that the efficacy data was provided by site, such as scalp, palmoplantar, nails, and genital psoriasis, as these are low-BSA but high-impact areas for patients,” said Dr. Cordoro.
The trial data on Crohn’s disease buttress her decision to continue to refrain from initiating ixekizumab in a child with IBD or who is at high risk for IBD. “I was happy to see that there was not a signal for Candida infection,” she added.
Interestingly, in the substudy in the European population, in which there was a double-blind, randomized withdrawal period, fewer patients who were reassigned to receive ixekizumab experienced relapse, compared with those who were reassigned to receive placebo. A total of 90.9% of patients who received placebo experienced relapse, compared with 17.6% of patients treated with ixekizumab. The median time to relapse in the placebo group was 149 days.
“There are data in the adult population that suggest intermittent treatment does allow for recapture of clinical response,” said Dr. Cordoro. “While it is not a large enough dataset to know definitively, this substudy of patients suggests the possibility of intermittent treatment and the ability to regain control [of psoriasis] after a period off drug.”
The study was funded by Eli Lilly. Dr. Paller is an investigator and consultant for Eli Lilly. Several other authors have received grants, personal fees, and/or were a consultant to Eli Lilly, and two authors are Eli Lilly employees. Dr. Cordoro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
with the interleukin (IL)-17 inhibitor, investigators reported.
In addition, findings of a substudy, which evaluated randomized withdrawal of treatment after 60 weeks, suggest patients were able to regain benefit after not being treated for a period.
Ixekizumab (Taltz) was approved by the U.S. Food and Drug Administration for treating pediatric psoriasis in March 2020 for patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The trial (IXORA-PEDS) involved 171 patients aged 6-17 years (mean age, 13.5 years; 99 females and 72 males), who were randomly assigned to receive ixekizumab via subcutaneous administration every 4 weeks (115) or placebo for 12 weeks (56). Thereafter, 166 patients continued in an open-label maintenance period in which they were treated every 4 weeks for 12-60 weeks. This was followed by an extension period of up to 108 weeks, which was completed by 139 patients (83.7%). At baseline, the patients’ Psoriasis Area and Severity Index (PASI) score was 12 or higher, the static Physician’s Global Assessment (sPGA) score was 3 or higher, and 10% or more of body surface area was affected.
In the study, at 12 weeks, treatment with ixekizumab was superior to placebo, with sustained responses through 48 weeks. In the follow-up phase, primary and secondary endpoints were sustained through week 108, with patients achieving or maintaining PASI 75 (91.7%), PASI 90 (79%), PASI 100 (55.1%), sPGA 0 or 1 (78.3%), and sPGA 0 (52.4%). Significant improvements in itch were seen at 12 weeks and were sustained with “meaningful improvements in itch for 78.5% of these patients at week 108,” the investigators report.
Among the patients who received ixekizumab, clearance rates in areas that are difficult to treat increased from week 12 to week 108 among those affected. During this time, clearance of nail psoriasis increased from 22.8% to 68.1%, clearance of palmoplantar psoriasis increased from 46.2% to 90%, clearance of scalp psoriasis increased from 70.7% to 76.2%, and clearance of genital psoriasis increased from 83.3% to 87.5%.
No new safety findings during weeks 48-108 of the trial were reported, including no new cases of inflammatory bowel disease (IBD) or Candida infections. The results were reported in JAMA Dermatology.
“Safety is really what we think of most when we are talking about pediatric patients, especially since they may be on these for decades and ... since they most commonly start these therapies in adolescence,” said Amy Paller, MD, the study’s lead author, in an interview. “To be able to take this out 108 weeks, 2 years, is starting to get to a point where we are getting more comfortable with safety. Clearly, no new signals arose.” Dr. Paller is chair of the department of dermatology and professor of dermatology and pediatrics, Northwestern University, Chicago.
One of the biggest concerns with using IL-17 inhibitors such as ixekizumab to manage psoriasis is the development of IBD, said Dr. Paller. She noted that four cases of IBD were reported before the extension phase of the trial but that no new IBD cases were reported after week 48.
“We would not start this as a treatment of choice in someone with Crohn’s disease, or perhaps we would think twice about using it in someone with a strong family history [of Crohn’s disease],” said Dr. Paller, who is also the director of the Skin Biology and Diseases Resource-Based Center at Northwestern. “Otherwise, it does not make me concerned about its use.”
Commenting on the study, Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, said that the trial’s results provide additional evidence regarding the optimal management of pediatric psoriasis.
“The landscape has shifted toward involving more pediatric patients in clinical trials, thereby providing dermatologists with data to select safe and effective therapies to manage children with psoriasis,” Dr. Cordoro said in an interview. “We have data showing that children with psoriasis have been undertreated, likely because of concerns about safety. The more evidence available from trials such as this, the more likely children are to receive necessary treatment.”
The efficacy data from the study on difficult-to-treat areas of psoriasis, in addition to improvements in BSA and PASI measures, are significant for clinicians deciding on a therapy for patients with psoriasis concentrated in specific body sites. “It was very valuable that the efficacy data was provided by site, such as scalp, palmoplantar, nails, and genital psoriasis, as these are low-BSA but high-impact areas for patients,” said Dr. Cordoro.
The trial data on Crohn’s disease buttress her decision to continue to refrain from initiating ixekizumab in a child with IBD or who is at high risk for IBD. “I was happy to see that there was not a signal for Candida infection,” she added.
Interestingly, in the substudy in the European population, in which there was a double-blind, randomized withdrawal period, fewer patients who were reassigned to receive ixekizumab experienced relapse, compared with those who were reassigned to receive placebo. A total of 90.9% of patients who received placebo experienced relapse, compared with 17.6% of patients treated with ixekizumab. The median time to relapse in the placebo group was 149 days.
“There are data in the adult population that suggest intermittent treatment does allow for recapture of clinical response,” said Dr. Cordoro. “While it is not a large enough dataset to know definitively, this substudy of patients suggests the possibility of intermittent treatment and the ability to regain control [of psoriasis] after a period off drug.”
The study was funded by Eli Lilly. Dr. Paller is an investigator and consultant for Eli Lilly. Several other authors have received grants, personal fees, and/or were a consultant to Eli Lilly, and two authors are Eli Lilly employees. Dr. Cordoro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
with the interleukin (IL)-17 inhibitor, investigators reported.
In addition, findings of a substudy, which evaluated randomized withdrawal of treatment after 60 weeks, suggest patients were able to regain benefit after not being treated for a period.
Ixekizumab (Taltz) was approved by the U.S. Food and Drug Administration for treating pediatric psoriasis in March 2020 for patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The trial (IXORA-PEDS) involved 171 patients aged 6-17 years (mean age, 13.5 years; 99 females and 72 males), who were randomly assigned to receive ixekizumab via subcutaneous administration every 4 weeks (115) or placebo for 12 weeks (56). Thereafter, 166 patients continued in an open-label maintenance period in which they were treated every 4 weeks for 12-60 weeks. This was followed by an extension period of up to 108 weeks, which was completed by 139 patients (83.7%). At baseline, the patients’ Psoriasis Area and Severity Index (PASI) score was 12 or higher, the static Physician’s Global Assessment (sPGA) score was 3 or higher, and 10% or more of body surface area was affected.
In the study, at 12 weeks, treatment with ixekizumab was superior to placebo, with sustained responses through 48 weeks. In the follow-up phase, primary and secondary endpoints were sustained through week 108, with patients achieving or maintaining PASI 75 (91.7%), PASI 90 (79%), PASI 100 (55.1%), sPGA 0 or 1 (78.3%), and sPGA 0 (52.4%). Significant improvements in itch were seen at 12 weeks and were sustained with “meaningful improvements in itch for 78.5% of these patients at week 108,” the investigators report.
Among the patients who received ixekizumab, clearance rates in areas that are difficult to treat increased from week 12 to week 108 among those affected. During this time, clearance of nail psoriasis increased from 22.8% to 68.1%, clearance of palmoplantar psoriasis increased from 46.2% to 90%, clearance of scalp psoriasis increased from 70.7% to 76.2%, and clearance of genital psoriasis increased from 83.3% to 87.5%.
No new safety findings during weeks 48-108 of the trial were reported, including no new cases of inflammatory bowel disease (IBD) or Candida infections. The results were reported in JAMA Dermatology.
“Safety is really what we think of most when we are talking about pediatric patients, especially since they may be on these for decades and ... since they most commonly start these therapies in adolescence,” said Amy Paller, MD, the study’s lead author, in an interview. “To be able to take this out 108 weeks, 2 years, is starting to get to a point where we are getting more comfortable with safety. Clearly, no new signals arose.” Dr. Paller is chair of the department of dermatology and professor of dermatology and pediatrics, Northwestern University, Chicago.
One of the biggest concerns with using IL-17 inhibitors such as ixekizumab to manage psoriasis is the development of IBD, said Dr. Paller. She noted that four cases of IBD were reported before the extension phase of the trial but that no new IBD cases were reported after week 48.
“We would not start this as a treatment of choice in someone with Crohn’s disease, or perhaps we would think twice about using it in someone with a strong family history [of Crohn’s disease],” said Dr. Paller, who is also the director of the Skin Biology and Diseases Resource-Based Center at Northwestern. “Otherwise, it does not make me concerned about its use.”
Commenting on the study, Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, said that the trial’s results provide additional evidence regarding the optimal management of pediatric psoriasis.
“The landscape has shifted toward involving more pediatric patients in clinical trials, thereby providing dermatologists with data to select safe and effective therapies to manage children with psoriasis,” Dr. Cordoro said in an interview. “We have data showing that children with psoriasis have been undertreated, likely because of concerns about safety. The more evidence available from trials such as this, the more likely children are to receive necessary treatment.”
The efficacy data from the study on difficult-to-treat areas of psoriasis, in addition to improvements in BSA and PASI measures, are significant for clinicians deciding on a therapy for patients with psoriasis concentrated in specific body sites. “It was very valuable that the efficacy data was provided by site, such as scalp, palmoplantar, nails, and genital psoriasis, as these are low-BSA but high-impact areas for patients,” said Dr. Cordoro.
The trial data on Crohn’s disease buttress her decision to continue to refrain from initiating ixekizumab in a child with IBD or who is at high risk for IBD. “I was happy to see that there was not a signal for Candida infection,” she added.
Interestingly, in the substudy in the European population, in which there was a double-blind, randomized withdrawal period, fewer patients who were reassigned to receive ixekizumab experienced relapse, compared with those who were reassigned to receive placebo. A total of 90.9% of patients who received placebo experienced relapse, compared with 17.6% of patients treated with ixekizumab. The median time to relapse in the placebo group was 149 days.
“There are data in the adult population that suggest intermittent treatment does allow for recapture of clinical response,” said Dr. Cordoro. “While it is not a large enough dataset to know definitively, this substudy of patients suggests the possibility of intermittent treatment and the ability to regain control [of psoriasis] after a period off drug.”
The study was funded by Eli Lilly. Dr. Paller is an investigator and consultant for Eli Lilly. Several other authors have received grants, personal fees, and/or were a consultant to Eli Lilly, and two authors are Eli Lilly employees. Dr. Cordoro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Secukinumab’s antipsoriatic effects confirmed in U.S. patient population
and those who up-titrated to 300 mg from the lower approved dose of 150 mg also saw benefits obtained at that level.
Researchers conducted a postmarketing trial of secukinumab in patients at U.S. centers, called CHOICE, after it was approved for psoriasis and PsA in 2015 and 2016 based on trials mainly conducted outside of the United States. The American patients in those studies “had a baseline clinical profile indicating harder-to-treat disease than the total study population, including higher body mass index (BMI), higher tender and swollen joint counts, increased prevalence of enthesitis and dactylitis, and more tumor necrosis factor inhibitor (TNFi) experience,” Tien Q. Nguyen, MD, a dermatologist in private practice in Irvine, Calif., and colleagues wrote in the Journal of Rheumatology.
In order to get a better sense of how secukinumab performs in U.S. patients who have not been treated with biologics, the researchers conducted the multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 4 CHOICE trial. It recruited patients for about 26 months at 67 U.S. centers during 2016-2018. The 258 patients randomized in the study to 300 mg (n = 103), 150 mg secukinumab (n = 103), or placebo (n = 52) had a mean time since PsA diagnosis of 3.0-3.9 years and all had a mean BMI of greater than 30 kg/m2, with dactylitis present in 48% and enthesitis in 73%. About one-third were taking methotrexate at baseline.
At week 16, patients taking secukinumab 300 mg were about 3.5 times more likely to have 20% improvement in American College of Rheumatology response criteria than with placebo (51.5% vs. 23.1%), whereas the response rate with 150 mg was not significantly different from placebo (36.9%). Rates of achieving ACR50 were significantly greater for both 300- and 150-mg doses versus placebo (28.2% and 24.3% vs. 5.8%), but only 300 mg led to a statistically significant difference in the rate of ACR70 responses, compared with placebo (17.5% vs. 1.9%).
In general, efficacy based on ACR20/50/70 responses and either remission or low disease activity on the Disease Activity in Psoriatic Arthritis index was lower among patients with less than 10 tender joints and less than 10 swollen joints at baseline. Methotrexate use at baseline did not affect ACR20 rates at week 16 in patients taking secukinumab, but the effect of methotrexate on ACR20 rates was noticeable among placebo-treated patients (38.9% vs. 14.7%). Enthesitis appeared to resolve significantly more often among patients on secukinumab, and more patients on secukinumab also had their dactylitis resolve, but the difference was not statistically significant.
Patients with psoriasis affecting more than 3% of their body surface area experienced higher response rates on the Psoriasis Area Severity Index (PASI) for 75%, 90%, and 100% skin lesion clearance than did patients taking placebo.
Patients who switched from 150 mg to 300 mg secukinumab after week 16 in the second treatment period of the trial more often achieved ACR20/50/70 responses by week 52, going from 2.4% to 65.9% of the up-titration subset for ACR20 and from 0% to 34.1% for ACR50 and to 12.2% for ACR70. Patients on placebo who switched also experienced increases in these response rates out to week 52. However, BMI above 30 kg/m2 led to numerically lower ACR50, ACR70, and PASI response rates at week 52.
The researchers noted that the response rates observed in CHOICE were lower than for the pivotal trials used for Food and Drug Administration approval for PsA, which “may have been due to patients in CHOICE having higher disease activity scores at baseline, compared with TNFi-naive patients” in the pivotal trials.
The safety profile of secukinumab appeared to be no different from what has been reported previously. The researchers said that, throughout the 52-week study, the most common adverse events in patients receiving secukinumab were upper respiratory tract infection in about 13% and diarrhea in about 7%. Most adverse events were mild or moderate, with serious adverse events occurring in 9.6% of patients taking secukinumab 300 mg and in 7.8% of patients taking secukinumab 150 mg over the 52 weeks.
“Overall, the findings from CHOICE were consistent with previous studies and demonstrated that secukinumab provides significant and sustained improvements in signs and symptoms of psoriatic arthritis. Our findings suggest that secukinumab 300 mg is safe and efficacious as a first-line biologic treatment for patients with PsA. Further studies will also help determine the optimal dose of secukinumab for treating overweight patients or those with high disease activity at treatment initiation,” the authors wrote.
The study was funded by Novartis, which manufactures secukinumab. Dr. Nguyen and some coauthors reported serving as a consultant, investigator, and/or speaker for numerous pharmaceutical companies, including Novartis.
and those who up-titrated to 300 mg from the lower approved dose of 150 mg also saw benefits obtained at that level.
Researchers conducted a postmarketing trial of secukinumab in patients at U.S. centers, called CHOICE, after it was approved for psoriasis and PsA in 2015 and 2016 based on trials mainly conducted outside of the United States. The American patients in those studies “had a baseline clinical profile indicating harder-to-treat disease than the total study population, including higher body mass index (BMI), higher tender and swollen joint counts, increased prevalence of enthesitis and dactylitis, and more tumor necrosis factor inhibitor (TNFi) experience,” Tien Q. Nguyen, MD, a dermatologist in private practice in Irvine, Calif., and colleagues wrote in the Journal of Rheumatology.
In order to get a better sense of how secukinumab performs in U.S. patients who have not been treated with biologics, the researchers conducted the multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 4 CHOICE trial. It recruited patients for about 26 months at 67 U.S. centers during 2016-2018. The 258 patients randomized in the study to 300 mg (n = 103), 150 mg secukinumab (n = 103), or placebo (n = 52) had a mean time since PsA diagnosis of 3.0-3.9 years and all had a mean BMI of greater than 30 kg/m2, with dactylitis present in 48% and enthesitis in 73%. About one-third were taking methotrexate at baseline.
At week 16, patients taking secukinumab 300 mg were about 3.5 times more likely to have 20% improvement in American College of Rheumatology response criteria than with placebo (51.5% vs. 23.1%), whereas the response rate with 150 mg was not significantly different from placebo (36.9%). Rates of achieving ACR50 were significantly greater for both 300- and 150-mg doses versus placebo (28.2% and 24.3% vs. 5.8%), but only 300 mg led to a statistically significant difference in the rate of ACR70 responses, compared with placebo (17.5% vs. 1.9%).
In general, efficacy based on ACR20/50/70 responses and either remission or low disease activity on the Disease Activity in Psoriatic Arthritis index was lower among patients with less than 10 tender joints and less than 10 swollen joints at baseline. Methotrexate use at baseline did not affect ACR20 rates at week 16 in patients taking secukinumab, but the effect of methotrexate on ACR20 rates was noticeable among placebo-treated patients (38.9% vs. 14.7%). Enthesitis appeared to resolve significantly more often among patients on secukinumab, and more patients on secukinumab also had their dactylitis resolve, but the difference was not statistically significant.
Patients with psoriasis affecting more than 3% of their body surface area experienced higher response rates on the Psoriasis Area Severity Index (PASI) for 75%, 90%, and 100% skin lesion clearance than did patients taking placebo.
Patients who switched from 150 mg to 300 mg secukinumab after week 16 in the second treatment period of the trial more often achieved ACR20/50/70 responses by week 52, going from 2.4% to 65.9% of the up-titration subset for ACR20 and from 0% to 34.1% for ACR50 and to 12.2% for ACR70. Patients on placebo who switched also experienced increases in these response rates out to week 52. However, BMI above 30 kg/m2 led to numerically lower ACR50, ACR70, and PASI response rates at week 52.
The researchers noted that the response rates observed in CHOICE were lower than for the pivotal trials used for Food and Drug Administration approval for PsA, which “may have been due to patients in CHOICE having higher disease activity scores at baseline, compared with TNFi-naive patients” in the pivotal trials.
The safety profile of secukinumab appeared to be no different from what has been reported previously. The researchers said that, throughout the 52-week study, the most common adverse events in patients receiving secukinumab were upper respiratory tract infection in about 13% and diarrhea in about 7%. Most adverse events were mild or moderate, with serious adverse events occurring in 9.6% of patients taking secukinumab 300 mg and in 7.8% of patients taking secukinumab 150 mg over the 52 weeks.
“Overall, the findings from CHOICE were consistent with previous studies and demonstrated that secukinumab provides significant and sustained improvements in signs and symptoms of psoriatic arthritis. Our findings suggest that secukinumab 300 mg is safe and efficacious as a first-line biologic treatment for patients with PsA. Further studies will also help determine the optimal dose of secukinumab for treating overweight patients or those with high disease activity at treatment initiation,” the authors wrote.
The study was funded by Novartis, which manufactures secukinumab. Dr. Nguyen and some coauthors reported serving as a consultant, investigator, and/or speaker for numerous pharmaceutical companies, including Novartis.
and those who up-titrated to 300 mg from the lower approved dose of 150 mg also saw benefits obtained at that level.
Researchers conducted a postmarketing trial of secukinumab in patients at U.S. centers, called CHOICE, after it was approved for psoriasis and PsA in 2015 and 2016 based on trials mainly conducted outside of the United States. The American patients in those studies “had a baseline clinical profile indicating harder-to-treat disease than the total study population, including higher body mass index (BMI), higher tender and swollen joint counts, increased prevalence of enthesitis and dactylitis, and more tumor necrosis factor inhibitor (TNFi) experience,” Tien Q. Nguyen, MD, a dermatologist in private practice in Irvine, Calif., and colleagues wrote in the Journal of Rheumatology.
In order to get a better sense of how secukinumab performs in U.S. patients who have not been treated with biologics, the researchers conducted the multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 4 CHOICE trial. It recruited patients for about 26 months at 67 U.S. centers during 2016-2018. The 258 patients randomized in the study to 300 mg (n = 103), 150 mg secukinumab (n = 103), or placebo (n = 52) had a mean time since PsA diagnosis of 3.0-3.9 years and all had a mean BMI of greater than 30 kg/m2, with dactylitis present in 48% and enthesitis in 73%. About one-third were taking methotrexate at baseline.
At week 16, patients taking secukinumab 300 mg were about 3.5 times more likely to have 20% improvement in American College of Rheumatology response criteria than with placebo (51.5% vs. 23.1%), whereas the response rate with 150 mg was not significantly different from placebo (36.9%). Rates of achieving ACR50 were significantly greater for both 300- and 150-mg doses versus placebo (28.2% and 24.3% vs. 5.8%), but only 300 mg led to a statistically significant difference in the rate of ACR70 responses, compared with placebo (17.5% vs. 1.9%).
In general, efficacy based on ACR20/50/70 responses and either remission or low disease activity on the Disease Activity in Psoriatic Arthritis index was lower among patients with less than 10 tender joints and less than 10 swollen joints at baseline. Methotrexate use at baseline did not affect ACR20 rates at week 16 in patients taking secukinumab, but the effect of methotrexate on ACR20 rates was noticeable among placebo-treated patients (38.9% vs. 14.7%). Enthesitis appeared to resolve significantly more often among patients on secukinumab, and more patients on secukinumab also had their dactylitis resolve, but the difference was not statistically significant.
Patients with psoriasis affecting more than 3% of their body surface area experienced higher response rates on the Psoriasis Area Severity Index (PASI) for 75%, 90%, and 100% skin lesion clearance than did patients taking placebo.
Patients who switched from 150 mg to 300 mg secukinumab after week 16 in the second treatment period of the trial more often achieved ACR20/50/70 responses by week 52, going from 2.4% to 65.9% of the up-titration subset for ACR20 and from 0% to 34.1% for ACR50 and to 12.2% for ACR70. Patients on placebo who switched also experienced increases in these response rates out to week 52. However, BMI above 30 kg/m2 led to numerically lower ACR50, ACR70, and PASI response rates at week 52.
The researchers noted that the response rates observed in CHOICE were lower than for the pivotal trials used for Food and Drug Administration approval for PsA, which “may have been due to patients in CHOICE having higher disease activity scores at baseline, compared with TNFi-naive patients” in the pivotal trials.
The safety profile of secukinumab appeared to be no different from what has been reported previously. The researchers said that, throughout the 52-week study, the most common adverse events in patients receiving secukinumab were upper respiratory tract infection in about 13% and diarrhea in about 7%. Most adverse events were mild or moderate, with serious adverse events occurring in 9.6% of patients taking secukinumab 300 mg and in 7.8% of patients taking secukinumab 150 mg over the 52 weeks.
“Overall, the findings from CHOICE were consistent with previous studies and demonstrated that secukinumab provides significant and sustained improvements in signs and symptoms of psoriatic arthritis. Our findings suggest that secukinumab 300 mg is safe and efficacious as a first-line biologic treatment for patients with PsA. Further studies will also help determine the optimal dose of secukinumab for treating overweight patients or those with high disease activity at treatment initiation,” the authors wrote.
The study was funded by Novartis, which manufactures secukinumab. Dr. Nguyen and some coauthors reported serving as a consultant, investigator, and/or speaker for numerous pharmaceutical companies, including Novartis.
FROM THE JOURNAL OF RHEUMATOLOGY
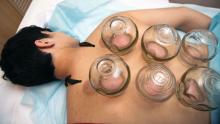
Cupping in dermatology
My inspiration to write about cupping this month stems from the perception that everyone seems to be talking about it, from a facialist who suggested it for me to a coworker who swears by cupping to treat her allergies. Cupping is by no means a novel procedure. Its use as a health therapy dates back thousands of years to ancient Egypt (1500 BCE), ancient Greece (described by Hippocrates), ancient Rome (described by the Greek physician Galen), China (during the Han dynasty, 206 BCE to 220 CE) and traditional Islamic culture.1 Over the past decade, the popularity of this ancient procedure has been increasing in the United States.1 Cupping has been applied as a remedy for various dermatologic and medical conditions, including herpes zoster, headaches, diminished appetite, maldigestion, abscess evacuation, narcolepsy, pain, fever, dysmenorrhea, and gout.1,2
Theories on the mechanism(s) of action
The practice of cupping is differentiated into dry and wet cupping.1,2 Traditionally, with dry cupping, a flame is applied to heat the air inside a thick glass cup (rather than the cup itself).1 The cup is placed on the skin surface, and negative pressure suctions the skin into the cup. Wet cupping differs mainly from dry cupping in that it involves blood-letting. Cups made of either silicone or glass of varying size and shapes are used. Modern adaptations to cupping include needle, herbal, and pulsatile cupping, as well as a “moving cupping” technique (vs. traditionally stationary cups).1
There are several theories, many of which are derived from the nondermatologic literature (that is, pain management), as to how cupping may deliver a clinical benefit. Some theories are based in scientific and medical principles, whereas other theories are more whimsical – specifically, that cupping draws out evil spirits.2 Studies of dry cupping have suggested that the procedure results in increased oxygenation of muscles via a local increase in oxygenated hemoglobin, which may help improve muscular activity and reduce pain.1 As theorized by Lowe in 2017, negative pressure exerted by dry cupping leads to stretching and dilation of capillaries, which increases blood flow.3 Wet cupping has been shown to increase heat shock protein 70 (HSP70) and beta-endorphin expression in rat models, which is thought to facilitate pain management.1 Removal of oxidants and reduction of reactive oxygen species in the blood is believed to be among the benefits of wet cupping.1
Cupping in general dermatology
While , as well as various inflammatory conditions.
Herpes zoster
In 2010, Cao et al. reported on their systematic review of wet cupping after completing searches of multiple databases (that is, PubMed, the Cochrane Library [Issue 3, 2008], China Network Knowledge Infrastructure, Chinese Scientific Journal Database, and Wan Fang Database). They identified eight randomized controlled trials involving 651 patients, with meta-analyses revealing that wet cupping performed better than medications in terms of the number of “cured” patients, number of patients with improved symptoms, and a lower incidence of postherpetic neuralgia. Wet cupping, in addition to medication, was also found to be superior to medication alone in multiple patients. The researchers concluded that wet cupping appears to effectively treat herpes zoster.4 However, the study failed to identify which medications were used to treat herpes zoster. In the United States, common medications for herpes zoster include acyclovir, valacyclovir, steroids, gabapentin, and other neuromodulators. Without knowing which medications were used, it is difficult to compare cupping to medication in terms of efficacy in treating herpes zoster.
Urticaria
Urticaria (hives) is an inflammatory skin condition that can be very uncomfortable for patients but often resolves without intervention within several months after onset. In 2001, Li and Ding reported on the treatment with cupping of 40 patients with urticaria. The cure rate among the treatment group was cited as 55%, compared with 30% in the control group, who were treated with a traditional Chinese remedy and an unidentified first-generation antihistamine.1,5 In 2020, Xiao et al. conducted a systematic review and meta-analysis of cupping therapy for patients with chronic urticaria. They identified 13 comparisons from 12 randomized controlled trials involving 842 subjects. The investigators found no significant differences between wet cupping and medication usage. They also found that cupping combined with antihistamine treatment was superior to antihistamines alone, and cupping therapy with acupuncture was more effective than acupuncture alone. The investigators did call for caution, citing the poor quality of the studies reviewed.6
It is important to note that it is difficult to attribute resolution of urticaria to the use of cupping given the self-resolution often associated with this condition. Antihistamines are the mainstay of therapy for urticaria, but in my personal experience, patients are not entirely satisfied with the level of symptom control with antihistamines alone and often search for alternative therapies to control the pesky hives and associated itch. In 2014, omalizumab (Xolair) was approved for treating chronic idiopathic urticaria, which has helped patients control symptoms of chronic idiopathic urticaria without needing to take antihistamines. There was no indication that the studies reviewed by Xiao et al. compared cupping against this new effective treatment. Therefore, these studies comparing cupping to medical management are outdated.
Acne, eczema, and psoriasis
Soliman’s 2018 review of cupping in dermatology included a few studies on these common cutaneous conditions. For instance, a 2013 single-blind prospective study by Xu et al. reported on the results of patients with moderate acne who received wet cupping (in the form of prickling bloodletting) twice weekly for 6 weeks.7 They reported that patients demonstrated improvement in the global acne grading system (GAGS) score by the end of the trial.1,7 Unfortunately, cupping was not compared with standard acne treatments (that is, benzoyl peroxide, topical and oral antibiotics, isotretinoin, topical retinoids, spironolactone).
In evaluating cupping for acute eczema, wet cupping was compared with oral loratadine and topical ointments in a 2007 study by Yao and Li. They divided 88 cases into treatment and control groups, with the former group (n = 46) receiving bloodletting puncturing and cupping and the control group (n = 42) receiving oral loratadine and topical Pairuisong (an herbal ointment used in Chinese medicine). The investigators observed no significant difference in total effective rates but a superior difference in the rates of responses that were considered “cured” and “markedly effective” in favor of the cupping treatment.1,8 However, a case report by Hon et al. has indicated that cupping therapy may be associated with more harm than benefit when used as an eczema treatment.1,9
In addition, it is important to note that the past 5 years have been gamechanging in the management of chronic eczema in terms of the array of novel and effective therapies (e.g., dupilumab and JAK inhibitors) and chronic moderate-to-severe eczema has become very treatable. Similarly, acute eczema is often successfully managed with topical steroids, calcineurin inhibitors, and emollients. As such, there is no compelling reason to consider an unproven treatment such as cupping.
In 2020, Xing et al. reviewed 16 randomized controlled trials assessing the use of “moving cupping” for plaque psoriasis, with 1,164 patients meeting inclusion criteria. Moving cupping was found to be significantly more effective than “no-moving” cupping therapy, and moving cupping, combined with medications, performed better than medications alone.10 None of the trials evaluated in this study included randomized controlled trials that compared patients using any of the more modern psoriasis medications, specifically biologics. And, again, the studies evaluated were not of the highest quality.
The data that support cupping, as summarized above, are based mostly on case reports, and strong double-blind prospective studies are lacking. Additionally, most of the studies cited gauged the efficacy of cupping using qualitative endpoints, rather than standardized quantitative endpoints and scales. Moreover, spontaneous remission of various dermatoses can occur, or they can improve over time, including acute eczema, psoriasis, and, especially, urticaria.
Adverse effects of cupping
Often alternative therapies are seen as “benign” and without adverse effects. However, complications can result from cupping. Trauma can be induced from the cupping itself by damaging superficial blood vessels and causing bruising.1,11 Blistering can also occur secondary to the suction effect, and the epidermal and dermal layers of the skin can be separated.1,11 Further, burns and discoloration have also been noted secondary to heat, trauma, and post inflammatory pigmentary changes.1,11 Another risk of cupping is the Koebner phenomenon, which occurs with psoriasis, with new lesions appearing in traumatized skin.12 Other adverse outcomes that have been reported with cupping include reactivation of herpes simplex virus secondary to skin trauma, iron deficiency anemia (secondary to blood loss), panniculitis, infections, and residual marks mistaken for signs of child abuse.1,11
Cupping in aesthetic dermatology
Facial cupping, a distinct practice from body cupping used to treat general dermatology conditions described previously, is also increasing in popularity. This practice is usually conducted in association with a facial or facial acupuncture by an aesthetician or other licensed professional. It can also be performed using at-home kits. The marketing claims for facial cupping cite improved tightening and contouring of facial skin, increased facial microcirculation and collagen synthesis, and enhanced lymphatic flow to aid with facial puffiness or swelling. One supposed mechanism for these benefits is that cupping increases blood flow. Interestingly, there was a 2020 animal study in which photoacoustic imaging of a mouse ear revealed increased temporary blood flow in the cupping microenvironment.13 Currently, however, there is no evidence in the English scientific literature that supports facial cupping. The benefits attributed to facial cupping for aesthetic purposes have emerged only in personal anecdotes. The temporary increase in blood flow may induce inflammation and swelling that adds volume to the face and temporarily diminishes wrinkles. However, this temporary plumpness may be associated with adverse effects, such as local trauma, irritation, bruising, postinflammatory pigmentary alteration, or even herpes reactivation. In my opinion, the possible adverse effects of cupping outweigh any potential benefit, especially given the insufficient evidence supporting the utility of cupping for cosmetic enhancement.
Summary
There is increasing interest among patients to incorporate complementary and alternative medicine – including the ancient tradition of cupping – in managing medical dermatologic conditions. However, current evidence supporting cupping as an effective therapeutic strategy is not strong, with most studies to date appearing to be of poor quality or not sufficiently convincing to displace standard therapies. Our medical strategies for managing chronic dermatologic conditions, particularly inflammatory disorders, continue to improve from both a safety and a proven efficacy standpoint. Therefore, I would not forgo medical management in favor of cupping. While cupping can be used as an adjunct therapy, I would caution patients about possible adverse side effects. In the aesthetic world, cupping is also gaining popularity, but this trend is also not supported by current evidence or studies, at least in the Western literature.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Write to her at [email protected] or message her on Instragram @DrChloeGoldman. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other disclosures.
References
1. Soliman Y et al. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Jun;27(2):103-7.
2. França K and Lotti T. Advances in Integrative Dermatology. John Wiley & Sons, 2019.
3. Lowe DT. Complement Ther Clin Pract. 2017 Nov;29:162-8.
4.Cao H et al. Altern Ther Health Med. 2010 Nov-Dec;16(6):48-54.
5. Li L and Ding J. J Tradit Chin Med. 2001 Mar;21(1):37-8.
6. Xiao XJ et al. J Integr Med. 2020 Jul;18(4):303-12.
7. Xu J et al. J Tradit Chin Med. 2013 Dec;33(6):752-6.
8. Yao J et al. Zhongguo Zhen Jiu. 2007; Jun;27(6):424-6.
9. Hon KL et al. Case Rep Pediatr. 2013;2013:605829.
10. Xing M et al. Medicine (Baltimore). 2020 Oct 9;99(41):e22539.
11. Kim TH et al. Eur J Integr Med. 2014 Aug 1;6(4):434-40.
12. Vender R and Vender R. J Cutan Med Surg. 2015 May-Jun;19(3):320-2.
13. Zhou Y et al. Biomed Opt Express. 2020 Apr 6;11(5):2394-401.
This article was updated 4/25/22.
My inspiration to write about cupping this month stems from the perception that everyone seems to be talking about it, from a facialist who suggested it for me to a coworker who swears by cupping to treat her allergies. Cupping is by no means a novel procedure. Its use as a health therapy dates back thousands of years to ancient Egypt (1500 BCE), ancient Greece (described by Hippocrates), ancient Rome (described by the Greek physician Galen), China (during the Han dynasty, 206 BCE to 220 CE) and traditional Islamic culture.1 Over the past decade, the popularity of this ancient procedure has been increasing in the United States.1 Cupping has been applied as a remedy for various dermatologic and medical conditions, including herpes zoster, headaches, diminished appetite, maldigestion, abscess evacuation, narcolepsy, pain, fever, dysmenorrhea, and gout.1,2
Theories on the mechanism(s) of action
The practice of cupping is differentiated into dry and wet cupping.1,2 Traditionally, with dry cupping, a flame is applied to heat the air inside a thick glass cup (rather than the cup itself).1 The cup is placed on the skin surface, and negative pressure suctions the skin into the cup. Wet cupping differs mainly from dry cupping in that it involves blood-letting. Cups made of either silicone or glass of varying size and shapes are used. Modern adaptations to cupping include needle, herbal, and pulsatile cupping, as well as a “moving cupping” technique (vs. traditionally stationary cups).1
There are several theories, many of which are derived from the nondermatologic literature (that is, pain management), as to how cupping may deliver a clinical benefit. Some theories are based in scientific and medical principles, whereas other theories are more whimsical – specifically, that cupping draws out evil spirits.2 Studies of dry cupping have suggested that the procedure results in increased oxygenation of muscles via a local increase in oxygenated hemoglobin, which may help improve muscular activity and reduce pain.1 As theorized by Lowe in 2017, negative pressure exerted by dry cupping leads to stretching and dilation of capillaries, which increases blood flow.3 Wet cupping has been shown to increase heat shock protein 70 (HSP70) and beta-endorphin expression in rat models, which is thought to facilitate pain management.1 Removal of oxidants and reduction of reactive oxygen species in the blood is believed to be among the benefits of wet cupping.1
Cupping in general dermatology
While , as well as various inflammatory conditions.
Herpes zoster
In 2010, Cao et al. reported on their systematic review of wet cupping after completing searches of multiple databases (that is, PubMed, the Cochrane Library [Issue 3, 2008], China Network Knowledge Infrastructure, Chinese Scientific Journal Database, and Wan Fang Database). They identified eight randomized controlled trials involving 651 patients, with meta-analyses revealing that wet cupping performed better than medications in terms of the number of “cured” patients, number of patients with improved symptoms, and a lower incidence of postherpetic neuralgia. Wet cupping, in addition to medication, was also found to be superior to medication alone in multiple patients. The researchers concluded that wet cupping appears to effectively treat herpes zoster.4 However, the study failed to identify which medications were used to treat herpes zoster. In the United States, common medications for herpes zoster include acyclovir, valacyclovir, steroids, gabapentin, and other neuromodulators. Without knowing which medications were used, it is difficult to compare cupping to medication in terms of efficacy in treating herpes zoster.
Urticaria
Urticaria (hives) is an inflammatory skin condition that can be very uncomfortable for patients but often resolves without intervention within several months after onset. In 2001, Li and Ding reported on the treatment with cupping of 40 patients with urticaria. The cure rate among the treatment group was cited as 55%, compared with 30% in the control group, who were treated with a traditional Chinese remedy and an unidentified first-generation antihistamine.1,5 In 2020, Xiao et al. conducted a systematic review and meta-analysis of cupping therapy for patients with chronic urticaria. They identified 13 comparisons from 12 randomized controlled trials involving 842 subjects. The investigators found no significant differences between wet cupping and medication usage. They also found that cupping combined with antihistamine treatment was superior to antihistamines alone, and cupping therapy with acupuncture was more effective than acupuncture alone. The investigators did call for caution, citing the poor quality of the studies reviewed.6
It is important to note that it is difficult to attribute resolution of urticaria to the use of cupping given the self-resolution often associated with this condition. Antihistamines are the mainstay of therapy for urticaria, but in my personal experience, patients are not entirely satisfied with the level of symptom control with antihistamines alone and often search for alternative therapies to control the pesky hives and associated itch. In 2014, omalizumab (Xolair) was approved for treating chronic idiopathic urticaria, which has helped patients control symptoms of chronic idiopathic urticaria without needing to take antihistamines. There was no indication that the studies reviewed by Xiao et al. compared cupping against this new effective treatment. Therefore, these studies comparing cupping to medical management are outdated.
Acne, eczema, and psoriasis
Soliman’s 2018 review of cupping in dermatology included a few studies on these common cutaneous conditions. For instance, a 2013 single-blind prospective study by Xu et al. reported on the results of patients with moderate acne who received wet cupping (in the form of prickling bloodletting) twice weekly for 6 weeks.7 They reported that patients demonstrated improvement in the global acne grading system (GAGS) score by the end of the trial.1,7 Unfortunately, cupping was not compared with standard acne treatments (that is, benzoyl peroxide, topical and oral antibiotics, isotretinoin, topical retinoids, spironolactone).
In evaluating cupping for acute eczema, wet cupping was compared with oral loratadine and topical ointments in a 2007 study by Yao and Li. They divided 88 cases into treatment and control groups, with the former group (n = 46) receiving bloodletting puncturing and cupping and the control group (n = 42) receiving oral loratadine and topical Pairuisong (an herbal ointment used in Chinese medicine). The investigators observed no significant difference in total effective rates but a superior difference in the rates of responses that were considered “cured” and “markedly effective” in favor of the cupping treatment.1,8 However, a case report by Hon et al. has indicated that cupping therapy may be associated with more harm than benefit when used as an eczema treatment.1,9
In addition, it is important to note that the past 5 years have been gamechanging in the management of chronic eczema in terms of the array of novel and effective therapies (e.g., dupilumab and JAK inhibitors) and chronic moderate-to-severe eczema has become very treatable. Similarly, acute eczema is often successfully managed with topical steroids, calcineurin inhibitors, and emollients. As such, there is no compelling reason to consider an unproven treatment such as cupping.
In 2020, Xing et al. reviewed 16 randomized controlled trials assessing the use of “moving cupping” for plaque psoriasis, with 1,164 patients meeting inclusion criteria. Moving cupping was found to be significantly more effective than “no-moving” cupping therapy, and moving cupping, combined with medications, performed better than medications alone.10 None of the trials evaluated in this study included randomized controlled trials that compared patients using any of the more modern psoriasis medications, specifically biologics. And, again, the studies evaluated were not of the highest quality.
The data that support cupping, as summarized above, are based mostly on case reports, and strong double-blind prospective studies are lacking. Additionally, most of the studies cited gauged the efficacy of cupping using qualitative endpoints, rather than standardized quantitative endpoints and scales. Moreover, spontaneous remission of various dermatoses can occur, or they can improve over time, including acute eczema, psoriasis, and, especially, urticaria.
Adverse effects of cupping
Often alternative therapies are seen as “benign” and without adverse effects. However, complications can result from cupping. Trauma can be induced from the cupping itself by damaging superficial blood vessels and causing bruising.1,11 Blistering can also occur secondary to the suction effect, and the epidermal and dermal layers of the skin can be separated.1,11 Further, burns and discoloration have also been noted secondary to heat, trauma, and post inflammatory pigmentary changes.1,11 Another risk of cupping is the Koebner phenomenon, which occurs with psoriasis, with new lesions appearing in traumatized skin.12 Other adverse outcomes that have been reported with cupping include reactivation of herpes simplex virus secondary to skin trauma, iron deficiency anemia (secondary to blood loss), panniculitis, infections, and residual marks mistaken for signs of child abuse.1,11
Cupping in aesthetic dermatology
Facial cupping, a distinct practice from body cupping used to treat general dermatology conditions described previously, is also increasing in popularity. This practice is usually conducted in association with a facial or facial acupuncture by an aesthetician or other licensed professional. It can also be performed using at-home kits. The marketing claims for facial cupping cite improved tightening and contouring of facial skin, increased facial microcirculation and collagen synthesis, and enhanced lymphatic flow to aid with facial puffiness or swelling. One supposed mechanism for these benefits is that cupping increases blood flow. Interestingly, there was a 2020 animal study in which photoacoustic imaging of a mouse ear revealed increased temporary blood flow in the cupping microenvironment.13 Currently, however, there is no evidence in the English scientific literature that supports facial cupping. The benefits attributed to facial cupping for aesthetic purposes have emerged only in personal anecdotes. The temporary increase in blood flow may induce inflammation and swelling that adds volume to the face and temporarily diminishes wrinkles. However, this temporary plumpness may be associated with adverse effects, such as local trauma, irritation, bruising, postinflammatory pigmentary alteration, or even herpes reactivation. In my opinion, the possible adverse effects of cupping outweigh any potential benefit, especially given the insufficient evidence supporting the utility of cupping for cosmetic enhancement.
Summary
There is increasing interest among patients to incorporate complementary and alternative medicine – including the ancient tradition of cupping – in managing medical dermatologic conditions. However, current evidence supporting cupping as an effective therapeutic strategy is not strong, with most studies to date appearing to be of poor quality or not sufficiently convincing to displace standard therapies. Our medical strategies for managing chronic dermatologic conditions, particularly inflammatory disorders, continue to improve from both a safety and a proven efficacy standpoint. Therefore, I would not forgo medical management in favor of cupping. While cupping can be used as an adjunct therapy, I would caution patients about possible adverse side effects. In the aesthetic world, cupping is also gaining popularity, but this trend is also not supported by current evidence or studies, at least in the Western literature.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Write to her at [email protected] or message her on Instragram @DrChloeGoldman. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other disclosures.
References
1. Soliman Y et al. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Jun;27(2):103-7.
2. França K and Lotti T. Advances in Integrative Dermatology. John Wiley & Sons, 2019.
3. Lowe DT. Complement Ther Clin Pract. 2017 Nov;29:162-8.
4.Cao H et al. Altern Ther Health Med. 2010 Nov-Dec;16(6):48-54.
5. Li L and Ding J. J Tradit Chin Med. 2001 Mar;21(1):37-8.
6. Xiao XJ et al. J Integr Med. 2020 Jul;18(4):303-12.
7. Xu J et al. J Tradit Chin Med. 2013 Dec;33(6):752-6.
8. Yao J et al. Zhongguo Zhen Jiu. 2007; Jun;27(6):424-6.
9. Hon KL et al. Case Rep Pediatr. 2013;2013:605829.
10. Xing M et al. Medicine (Baltimore). 2020 Oct 9;99(41):e22539.
11. Kim TH et al. Eur J Integr Med. 2014 Aug 1;6(4):434-40.
12. Vender R and Vender R. J Cutan Med Surg. 2015 May-Jun;19(3):320-2.
13. Zhou Y et al. Biomed Opt Express. 2020 Apr 6;11(5):2394-401.
This article was updated 4/25/22.
My inspiration to write about cupping this month stems from the perception that everyone seems to be talking about it, from a facialist who suggested it for me to a coworker who swears by cupping to treat her allergies. Cupping is by no means a novel procedure. Its use as a health therapy dates back thousands of years to ancient Egypt (1500 BCE), ancient Greece (described by Hippocrates), ancient Rome (described by the Greek physician Galen), China (during the Han dynasty, 206 BCE to 220 CE) and traditional Islamic culture.1 Over the past decade, the popularity of this ancient procedure has been increasing in the United States.1 Cupping has been applied as a remedy for various dermatologic and medical conditions, including herpes zoster, headaches, diminished appetite, maldigestion, abscess evacuation, narcolepsy, pain, fever, dysmenorrhea, and gout.1,2
Theories on the mechanism(s) of action
The practice of cupping is differentiated into dry and wet cupping.1,2 Traditionally, with dry cupping, a flame is applied to heat the air inside a thick glass cup (rather than the cup itself).1 The cup is placed on the skin surface, and negative pressure suctions the skin into the cup. Wet cupping differs mainly from dry cupping in that it involves blood-letting. Cups made of either silicone or glass of varying size and shapes are used. Modern adaptations to cupping include needle, herbal, and pulsatile cupping, as well as a “moving cupping” technique (vs. traditionally stationary cups).1
There are several theories, many of which are derived from the nondermatologic literature (that is, pain management), as to how cupping may deliver a clinical benefit. Some theories are based in scientific and medical principles, whereas other theories are more whimsical – specifically, that cupping draws out evil spirits.2 Studies of dry cupping have suggested that the procedure results in increased oxygenation of muscles via a local increase in oxygenated hemoglobin, which may help improve muscular activity and reduce pain.1 As theorized by Lowe in 2017, negative pressure exerted by dry cupping leads to stretching and dilation of capillaries, which increases blood flow.3 Wet cupping has been shown to increase heat shock protein 70 (HSP70) and beta-endorphin expression in rat models, which is thought to facilitate pain management.1 Removal of oxidants and reduction of reactive oxygen species in the blood is believed to be among the benefits of wet cupping.1
Cupping in general dermatology
While , as well as various inflammatory conditions.
Herpes zoster
In 2010, Cao et al. reported on their systematic review of wet cupping after completing searches of multiple databases (that is, PubMed, the Cochrane Library [Issue 3, 2008], China Network Knowledge Infrastructure, Chinese Scientific Journal Database, and Wan Fang Database). They identified eight randomized controlled trials involving 651 patients, with meta-analyses revealing that wet cupping performed better than medications in terms of the number of “cured” patients, number of patients with improved symptoms, and a lower incidence of postherpetic neuralgia. Wet cupping, in addition to medication, was also found to be superior to medication alone in multiple patients. The researchers concluded that wet cupping appears to effectively treat herpes zoster.4 However, the study failed to identify which medications were used to treat herpes zoster. In the United States, common medications for herpes zoster include acyclovir, valacyclovir, steroids, gabapentin, and other neuromodulators. Without knowing which medications were used, it is difficult to compare cupping to medication in terms of efficacy in treating herpes zoster.
Urticaria
Urticaria (hives) is an inflammatory skin condition that can be very uncomfortable for patients but often resolves without intervention within several months after onset. In 2001, Li and Ding reported on the treatment with cupping of 40 patients with urticaria. The cure rate among the treatment group was cited as 55%, compared with 30% in the control group, who were treated with a traditional Chinese remedy and an unidentified first-generation antihistamine.1,5 In 2020, Xiao et al. conducted a systematic review and meta-analysis of cupping therapy for patients with chronic urticaria. They identified 13 comparisons from 12 randomized controlled trials involving 842 subjects. The investigators found no significant differences between wet cupping and medication usage. They also found that cupping combined with antihistamine treatment was superior to antihistamines alone, and cupping therapy with acupuncture was more effective than acupuncture alone. The investigators did call for caution, citing the poor quality of the studies reviewed.6
It is important to note that it is difficult to attribute resolution of urticaria to the use of cupping given the self-resolution often associated with this condition. Antihistamines are the mainstay of therapy for urticaria, but in my personal experience, patients are not entirely satisfied with the level of symptom control with antihistamines alone and often search for alternative therapies to control the pesky hives and associated itch. In 2014, omalizumab (Xolair) was approved for treating chronic idiopathic urticaria, which has helped patients control symptoms of chronic idiopathic urticaria without needing to take antihistamines. There was no indication that the studies reviewed by Xiao et al. compared cupping against this new effective treatment. Therefore, these studies comparing cupping to medical management are outdated.
Acne, eczema, and psoriasis
Soliman’s 2018 review of cupping in dermatology included a few studies on these common cutaneous conditions. For instance, a 2013 single-blind prospective study by Xu et al. reported on the results of patients with moderate acne who received wet cupping (in the form of prickling bloodletting) twice weekly for 6 weeks.7 They reported that patients demonstrated improvement in the global acne grading system (GAGS) score by the end of the trial.1,7 Unfortunately, cupping was not compared with standard acne treatments (that is, benzoyl peroxide, topical and oral antibiotics, isotretinoin, topical retinoids, spironolactone).
In evaluating cupping for acute eczema, wet cupping was compared with oral loratadine and topical ointments in a 2007 study by Yao and Li. They divided 88 cases into treatment and control groups, with the former group (n = 46) receiving bloodletting puncturing and cupping and the control group (n = 42) receiving oral loratadine and topical Pairuisong (an herbal ointment used in Chinese medicine). The investigators observed no significant difference in total effective rates but a superior difference in the rates of responses that were considered “cured” and “markedly effective” in favor of the cupping treatment.1,8 However, a case report by Hon et al. has indicated that cupping therapy may be associated with more harm than benefit when used as an eczema treatment.1,9
In addition, it is important to note that the past 5 years have been gamechanging in the management of chronic eczema in terms of the array of novel and effective therapies (e.g., dupilumab and JAK inhibitors) and chronic moderate-to-severe eczema has become very treatable. Similarly, acute eczema is often successfully managed with topical steroids, calcineurin inhibitors, and emollients. As such, there is no compelling reason to consider an unproven treatment such as cupping.
In 2020, Xing et al. reviewed 16 randomized controlled trials assessing the use of “moving cupping” for plaque psoriasis, with 1,164 patients meeting inclusion criteria. Moving cupping was found to be significantly more effective than “no-moving” cupping therapy, and moving cupping, combined with medications, performed better than medications alone.10 None of the trials evaluated in this study included randomized controlled trials that compared patients using any of the more modern psoriasis medications, specifically biologics. And, again, the studies evaluated were not of the highest quality.
The data that support cupping, as summarized above, are based mostly on case reports, and strong double-blind prospective studies are lacking. Additionally, most of the studies cited gauged the efficacy of cupping using qualitative endpoints, rather than standardized quantitative endpoints and scales. Moreover, spontaneous remission of various dermatoses can occur, or they can improve over time, including acute eczema, psoriasis, and, especially, urticaria.
Adverse effects of cupping
Often alternative therapies are seen as “benign” and without adverse effects. However, complications can result from cupping. Trauma can be induced from the cupping itself by damaging superficial blood vessels and causing bruising.1,11 Blistering can also occur secondary to the suction effect, and the epidermal and dermal layers of the skin can be separated.1,11 Further, burns and discoloration have also been noted secondary to heat, trauma, and post inflammatory pigmentary changes.1,11 Another risk of cupping is the Koebner phenomenon, which occurs with psoriasis, with new lesions appearing in traumatized skin.12 Other adverse outcomes that have been reported with cupping include reactivation of herpes simplex virus secondary to skin trauma, iron deficiency anemia (secondary to blood loss), panniculitis, infections, and residual marks mistaken for signs of child abuse.1,11
Cupping in aesthetic dermatology
Facial cupping, a distinct practice from body cupping used to treat general dermatology conditions described previously, is also increasing in popularity. This practice is usually conducted in association with a facial or facial acupuncture by an aesthetician or other licensed professional. It can also be performed using at-home kits. The marketing claims for facial cupping cite improved tightening and contouring of facial skin, increased facial microcirculation and collagen synthesis, and enhanced lymphatic flow to aid with facial puffiness or swelling. One supposed mechanism for these benefits is that cupping increases blood flow. Interestingly, there was a 2020 animal study in which photoacoustic imaging of a mouse ear revealed increased temporary blood flow in the cupping microenvironment.13 Currently, however, there is no evidence in the English scientific literature that supports facial cupping. The benefits attributed to facial cupping for aesthetic purposes have emerged only in personal anecdotes. The temporary increase in blood flow may induce inflammation and swelling that adds volume to the face and temporarily diminishes wrinkles. However, this temporary plumpness may be associated with adverse effects, such as local trauma, irritation, bruising, postinflammatory pigmentary alteration, or even herpes reactivation. In my opinion, the possible adverse effects of cupping outweigh any potential benefit, especially given the insufficient evidence supporting the utility of cupping for cosmetic enhancement.
Summary
There is increasing interest among patients to incorporate complementary and alternative medicine – including the ancient tradition of cupping – in managing medical dermatologic conditions. However, current evidence supporting cupping as an effective therapeutic strategy is not strong, with most studies to date appearing to be of poor quality or not sufficiently convincing to displace standard therapies. Our medical strategies for managing chronic dermatologic conditions, particularly inflammatory disorders, continue to improve from both a safety and a proven efficacy standpoint. Therefore, I would not forgo medical management in favor of cupping. While cupping can be used as an adjunct therapy, I would caution patients about possible adverse side effects. In the aesthetic world, cupping is also gaining popularity, but this trend is also not supported by current evidence or studies, at least in the Western literature.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Write to her at [email protected] or message her on Instragram @DrChloeGoldman. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other disclosures.
References
1. Soliman Y et al. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Jun;27(2):103-7.
2. França K and Lotti T. Advances in Integrative Dermatology. John Wiley & Sons, 2019.
3. Lowe DT. Complement Ther Clin Pract. 2017 Nov;29:162-8.
4.Cao H et al. Altern Ther Health Med. 2010 Nov-Dec;16(6):48-54.
5. Li L and Ding J. J Tradit Chin Med. 2001 Mar;21(1):37-8.
6. Xiao XJ et al. J Integr Med. 2020 Jul;18(4):303-12.
7. Xu J et al. J Tradit Chin Med. 2013 Dec;33(6):752-6.
8. Yao J et al. Zhongguo Zhen Jiu. 2007; Jun;27(6):424-6.
9. Hon KL et al. Case Rep Pediatr. 2013;2013:605829.
10. Xing M et al. Medicine (Baltimore). 2020 Oct 9;99(41):e22539.
11. Kim TH et al. Eur J Integr Med. 2014 Aug 1;6(4):434-40.
12. Vender R and Vender R. J Cutan Med Surg. 2015 May-Jun;19(3):320-2.
13. Zhou Y et al. Biomed Opt Express. 2020 Apr 6;11(5):2394-401.
This article was updated 4/25/22.
Apremilast has neutral effect on vascular inflammation in psoriasis study
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Study links air pollution to psoriasis flares
Exposure to air pollution – even short term – may play a role in triggering psoriasis flares, according to new research from Italy, which found a significant association between exposure to higher levels of air pollution prior to patients presenting for psoriasis flares at medical visits, compared with visits unrelated to flares.
“We found that higher concentration of different air pollutants was associated with psoriasis flares in patients living in an industrialized city of the Po Valley” in Verona, Italy, report the authors of the study, published in JAMA Dermatology.
The findings underscore the need for clinicians to “consider environmental/external triggers in patients with chronic inflammatory diseases experiencing flares,” first author Francesco Bellinato, MD, of the Section of Dermatology and Venereology, University of Verona, Italy, told this news organization.
He and his coauthors conducted a case-crossover and cross-sectional longitudinal study that involved a retrospective analysis of data in 957 patients in Verona with chronic plaque psoriasis, who were evaluated every 3-4 months at an outpatient dermatology clinic for a median of 2.7 years.
Over the study period, disease flares, defined as an increase in the Psoriasis Area and Severity Index (PASI) of 5 or more points from the previous visit, occurred in 369 patients (38.6%), consistent with known flare rates in psoriasis. Participants in the study (mean age, 61) had median PASI scores of 12 during visits for psoriatic flares compared with PASI scores of 1 during control (no flare) visits (P < .001).
Evaluations of mean concentrations of several air pollutants within 10 miles of the patients over 4,398 visits showed that concentrations were significantly higher in the 60 days prior to the psoriasis flare, compared with control visits that were not related to flares (P < .05), after adjusting for factors including seasonality (by trimester, to adjust for weather conditions and UV/sunlight exposure) and the type of systemic psoriasis treatments patients were receiving (conventional or biological).
Increases in air pollutant levels prior to flares were observed among the 35.8% of patients who had a flare of at least a 50% increase in the PASI score, as well among the 47.2% of patients who had at least a 100% increase in PASI, compared with control visits not involving flares. In addition, mean and area-under-the-curve concentrations of air pollutants were higher in the 60 days before the visits among those with PASI 5 or greater, compared with those with PASI scores below 5, the authors add.
Dr. Bellinato noted that the associations were not limited to any particular subgroup. “The associations with air pollution and flares were observed in the entire population,” he said in an interview.
Vehicle, industry emissions
The pollutants that were measured were those mainly associated with fossil fuel combustion from vehicle and industry emissions, including carbon monoxide, nitrogen dioxide, other nitrogen oxides, benzene, coarse particulate matter (2.5-10.0 μm in diameter) and fine particulate matter (less than 2.5 μm in diameter).
They note that the risk of having a PASI score of 5 or greater was elevated even at thresholds of exposure that are largely considered safe. “Indeed, the risk for having a PASI score of 5 or greater was 40% to 50% higher at exposures as low as 20 μg/m3” of coarse particulate matter and 15 μg/m3 of fine particulate matter in the 60-day period prior to the visits, they write.
The authors referred to evidence linking air pollution with a worsening of a variety of inflammatory cutaneous diseases, including atopic dermatitis and acne, as well as photoaging. Psoriasis flares are known to be triggered by a variety of environmental factors, including infections or certain drugs; however, evidence of a role of air pollution has been lacking. Potential mechanisms linking the exposures to flares include the possibility that exhaust particles can activate skin resident T-cells, “resulting in abnormal production of proinflammatory cytokines including tumor necrosis factor α (TNF-α) and interleukins (ILs), including IL-1α, IL-1β, IL-6, and IL-8.8,” the authors write.
Their results, though inferring a causal relationship, fall short of showing a clear dose–response relationship between higher pollutant levels and an increased risk of psoriasis flares, possibly the result of a smaller sample size of subjects exposed to higher levels of pollution, they add.
Limitations of the study included the definition of flare, which used a clinical score that could be affected by other measurements, they point out, while strengths of the study included the large cohort of patients followed for over 7 years and the availability of daily measurements of air pollutants.
While the study suggests that environmental air pollutant fluctuations may affect psoriasis course,” the authors concluded, “further study is needed to examine whether these findings generalize to other populations and to better understand the mechanisms by which air pollution may affect psoriasis disease activity.”
Dr. Bellinato and four coauthors had no disclosures; the remaining authors had disclosures that included receiving personal fees from pharmaceutical companies that were outside of the submitted work.
A version of this article first appeared on Medscape.com.
Exposure to air pollution – even short term – may play a role in triggering psoriasis flares, according to new research from Italy, which found a significant association between exposure to higher levels of air pollution prior to patients presenting for psoriasis flares at medical visits, compared with visits unrelated to flares.
“We found that higher concentration of different air pollutants was associated with psoriasis flares in patients living in an industrialized city of the Po Valley” in Verona, Italy, report the authors of the study, published in JAMA Dermatology.
The findings underscore the need for clinicians to “consider environmental/external triggers in patients with chronic inflammatory diseases experiencing flares,” first author Francesco Bellinato, MD, of the Section of Dermatology and Venereology, University of Verona, Italy, told this news organization.
He and his coauthors conducted a case-crossover and cross-sectional longitudinal study that involved a retrospective analysis of data in 957 patients in Verona with chronic plaque psoriasis, who were evaluated every 3-4 months at an outpatient dermatology clinic for a median of 2.7 years.
Over the study period, disease flares, defined as an increase in the Psoriasis Area and Severity Index (PASI) of 5 or more points from the previous visit, occurred in 369 patients (38.6%), consistent with known flare rates in psoriasis. Participants in the study (mean age, 61) had median PASI scores of 12 during visits for psoriatic flares compared with PASI scores of 1 during control (no flare) visits (P < .001).
Evaluations of mean concentrations of several air pollutants within 10 miles of the patients over 4,398 visits showed that concentrations were significantly higher in the 60 days prior to the psoriasis flare, compared with control visits that were not related to flares (P < .05), after adjusting for factors including seasonality (by trimester, to adjust for weather conditions and UV/sunlight exposure) and the type of systemic psoriasis treatments patients were receiving (conventional or biological).
Increases in air pollutant levels prior to flares were observed among the 35.8% of patients who had a flare of at least a 50% increase in the PASI score, as well among the 47.2% of patients who had at least a 100% increase in PASI, compared with control visits not involving flares. In addition, mean and area-under-the-curve concentrations of air pollutants were higher in the 60 days before the visits among those with PASI 5 or greater, compared with those with PASI scores below 5, the authors add.
Dr. Bellinato noted that the associations were not limited to any particular subgroup. “The associations with air pollution and flares were observed in the entire population,” he said in an interview.
Vehicle, industry emissions
The pollutants that were measured were those mainly associated with fossil fuel combustion from vehicle and industry emissions, including carbon monoxide, nitrogen dioxide, other nitrogen oxides, benzene, coarse particulate matter (2.5-10.0 μm in diameter) and fine particulate matter (less than 2.5 μm in diameter).
They note that the risk of having a PASI score of 5 or greater was elevated even at thresholds of exposure that are largely considered safe. “Indeed, the risk for having a PASI score of 5 or greater was 40% to 50% higher at exposures as low as 20 μg/m3” of coarse particulate matter and 15 μg/m3 of fine particulate matter in the 60-day period prior to the visits, they write.
The authors referred to evidence linking air pollution with a worsening of a variety of inflammatory cutaneous diseases, including atopic dermatitis and acne, as well as photoaging. Psoriasis flares are known to be triggered by a variety of environmental factors, including infections or certain drugs; however, evidence of a role of air pollution has been lacking. Potential mechanisms linking the exposures to flares include the possibility that exhaust particles can activate skin resident T-cells, “resulting in abnormal production of proinflammatory cytokines including tumor necrosis factor α (TNF-α) and interleukins (ILs), including IL-1α, IL-1β, IL-6, and IL-8.8,” the authors write.
Their results, though inferring a causal relationship, fall short of showing a clear dose–response relationship between higher pollutant levels and an increased risk of psoriasis flares, possibly the result of a smaller sample size of subjects exposed to higher levels of pollution, they add.
Limitations of the study included the definition of flare, which used a clinical score that could be affected by other measurements, they point out, while strengths of the study included the large cohort of patients followed for over 7 years and the availability of daily measurements of air pollutants.
While the study suggests that environmental air pollutant fluctuations may affect psoriasis course,” the authors concluded, “further study is needed to examine whether these findings generalize to other populations and to better understand the mechanisms by which air pollution may affect psoriasis disease activity.”
Dr. Bellinato and four coauthors had no disclosures; the remaining authors had disclosures that included receiving personal fees from pharmaceutical companies that were outside of the submitted work.
A version of this article first appeared on Medscape.com.
Exposure to air pollution – even short term – may play a role in triggering psoriasis flares, according to new research from Italy, which found a significant association between exposure to higher levels of air pollution prior to patients presenting for psoriasis flares at medical visits, compared with visits unrelated to flares.
“We found that higher concentration of different air pollutants was associated with psoriasis flares in patients living in an industrialized city of the Po Valley” in Verona, Italy, report the authors of the study, published in JAMA Dermatology.
The findings underscore the need for clinicians to “consider environmental/external triggers in patients with chronic inflammatory diseases experiencing flares,” first author Francesco Bellinato, MD, of the Section of Dermatology and Venereology, University of Verona, Italy, told this news organization.
He and his coauthors conducted a case-crossover and cross-sectional longitudinal study that involved a retrospective analysis of data in 957 patients in Verona with chronic plaque psoriasis, who were evaluated every 3-4 months at an outpatient dermatology clinic for a median of 2.7 years.
Over the study period, disease flares, defined as an increase in the Psoriasis Area and Severity Index (PASI) of 5 or more points from the previous visit, occurred in 369 patients (38.6%), consistent with known flare rates in psoriasis. Participants in the study (mean age, 61) had median PASI scores of 12 during visits for psoriatic flares compared with PASI scores of 1 during control (no flare) visits (P < .001).
Evaluations of mean concentrations of several air pollutants within 10 miles of the patients over 4,398 visits showed that concentrations were significantly higher in the 60 days prior to the psoriasis flare, compared with control visits that were not related to flares (P < .05), after adjusting for factors including seasonality (by trimester, to adjust for weather conditions and UV/sunlight exposure) and the type of systemic psoriasis treatments patients were receiving (conventional or biological).
Increases in air pollutant levels prior to flares were observed among the 35.8% of patients who had a flare of at least a 50% increase in the PASI score, as well among the 47.2% of patients who had at least a 100% increase in PASI, compared with control visits not involving flares. In addition, mean and area-under-the-curve concentrations of air pollutants were higher in the 60 days before the visits among those with PASI 5 or greater, compared with those with PASI scores below 5, the authors add.
Dr. Bellinato noted that the associations were not limited to any particular subgroup. “The associations with air pollution and flares were observed in the entire population,” he said in an interview.
Vehicle, industry emissions
The pollutants that were measured were those mainly associated with fossil fuel combustion from vehicle and industry emissions, including carbon monoxide, nitrogen dioxide, other nitrogen oxides, benzene, coarse particulate matter (2.5-10.0 μm in diameter) and fine particulate matter (less than 2.5 μm in diameter).
They note that the risk of having a PASI score of 5 or greater was elevated even at thresholds of exposure that are largely considered safe. “Indeed, the risk for having a PASI score of 5 or greater was 40% to 50% higher at exposures as low as 20 μg/m3” of coarse particulate matter and 15 μg/m3 of fine particulate matter in the 60-day period prior to the visits, they write.
The authors referred to evidence linking air pollution with a worsening of a variety of inflammatory cutaneous diseases, including atopic dermatitis and acne, as well as photoaging. Psoriasis flares are known to be triggered by a variety of environmental factors, including infections or certain drugs; however, evidence of a role of air pollution has been lacking. Potential mechanisms linking the exposures to flares include the possibility that exhaust particles can activate skin resident T-cells, “resulting in abnormal production of proinflammatory cytokines including tumor necrosis factor α (TNF-α) and interleukins (ILs), including IL-1α, IL-1β, IL-6, and IL-8.8,” the authors write.
Their results, though inferring a causal relationship, fall short of showing a clear dose–response relationship between higher pollutant levels and an increased risk of psoriasis flares, possibly the result of a smaller sample size of subjects exposed to higher levels of pollution, they add.
Limitations of the study included the definition of flare, which used a clinical score that could be affected by other measurements, they point out, while strengths of the study included the large cohort of patients followed for over 7 years and the availability of daily measurements of air pollutants.
While the study suggests that environmental air pollutant fluctuations may affect psoriasis course,” the authors concluded, “further study is needed to examine whether these findings generalize to other populations and to better understand the mechanisms by which air pollution may affect psoriasis disease activity.”
Dr. Bellinato and four coauthors had no disclosures; the remaining authors had disclosures that included receiving personal fees from pharmaceutical companies that were outside of the submitted work.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
CV risk biomarkers tentatively identified in psoriatic disease
The risk of cardiovascular (CV) events in patients with psoriatic disease rises with higher levels of two cardiac biomarkers in a manner independent of risk calculated by the Framingham Risk Score (FRS), a longitudinal cohort study has shown. But researchers who conducted the study note that neither of the two biomarkers identified in the study – cardiac troponin I (cTnI) and N-terminal pro-brain-type natriuretic peptide (NT-proBNP) – led to an improvement in predictive performance when combined with the FRS, despite their association with carotid plaque burden.
Psoriasis and psoriatic arthritis are both associated with greater risk of CV morbidity and mortality, partly because of systemic inflammation that leads to atherogenesis. Measures of CV risk such as the FRS rely on traditional measures of CV risk and thus are likely to underestimate the CV event risk of people with psoriatic disease, according to the authors of the new study, published online in Arthritis & Rheumatology. The effort was led by Keith Colaço, MSc; Lihi Eder, MD, PhD; and other researchers affiliated with the University of Toronto.
“We are desperately in need of biomarker science advancement in psoriatic arthritis for a variety of places of guidance: How to choose a medication more accurately for the patient in front of us – that is, getting to be more like oncologists who use biomarkers to pick the best treatment or combination. That’s an important need. A second important need is how to guide clinicians regarding risk prediction for things like persistent, severe disease activity, progressive structural damage from disease, and, in this case, predicting a very common comorbidity that occurs in [psoriasis and] psoriatic arthritis patients,” Philip J. Mease, MD, told this news organization when asked to comment on the study.
Such biomarkers could assist with patient counseling, according to Dr. Mease, who is director of rheumatology research at Swedish Medical Center/Providence St. Joseph Health and is a clinical professor at the University of Washington, both in Seattle. Some patients may struggle with advice to lose weight or adopt lifestyle measures to limit CV risk, and more accurate predictions of risk may serve as further motivation. “It could well be that if you have a biomarker that accurately predicts a coming cataclysm, that it will lead you to redouble your efforts to do whatever it takes to reduce cardiovascular risk,” he said.
Both cTnI and NT-proBNP have been linked to increased CV risk in the general population, but little work has been done in the context of rheumatologic diseases.
The researchers analyzed data from 358 patients seen at the University of Toronto. The mean follow-up was 3.69 years. After adjustment for CV risk factors, lipid-lowering therapy, and creatinine levels, there was an association between cTnI levels and total carotid plaque area (adjusted beta coefficient, 0.21; 95% confidence interval, 0-0.41), but not for levels of NT-proBNP.
Atherosclerosis progressed in 89 participants overall, but multivariate adjustment revealed no significant relationship between progression and cTnI or NT-proBNP levels.
Separately, the researchers analyzed 1,000 individuals with psoriatic arthritis (n = 648) or with psoriasis and no arthritis (n = 352) whom they followed for a mean of 7.1 years after the patients underwent evaluation during 2002-2019. After adjustment for FRS, there was an association between the risk of a CV event and each 1–standard deviation increase in both cTnI (hazard ratio, 3.02; 95% CI, 1.12-8.16) and NT-proBNP (HR, 2.02; 95% CI, 1.28-3.18).
The combination of both biomarkers with the FRS predicted higher CV risk (HR, 1.91; 95% CI, 1.23-2.97). Neither biomarker made a statistically significant difference in changing CV risk prediction when added individually to FRS, although cTnI trended toward significance (HR, 2.60; 95% CI, 0.98-6.87).
Instead of the carotid plaque burden, Dr. Mease would have liked to have seen the authors evaluate calcium scores in coronary arteries as measured by CT. “I would have loved to have seen the researchers using that in addition to the carotid plaque assessment, to see what that would show us about these patients,” he said.
Only a small number of patients experienced CV events during the study period, which will likely make it necessary to conduct larger studies to identify a clear relationship. “You need a registry-type study with probably many hundreds if not thousands of patients in order to identify whether or not adding troponin could be useful to what we typically measure with patients when we’re trying to assess their risk,” Dr. Mease said.
The study was supported in part by the National Psoriasis Foundation and the Arthritis Society. Individual researchers have received support from a range of sources, including the Enid Walker Estate, the Women’s College Research Institute, the Arthritis Society, the National Psoriasis Foundation, the Edward Dunlop Foundation, the Ontario Ministry of Science and Innovation, and a Pfizer Chair Research Award. Some of the researchers have financial relationships with pharmaceutical companies that market drugs for psoriasis and psoriatic arthritis.
A version of this article first appeared on Medscape.com.
The risk of cardiovascular (CV) events in patients with psoriatic disease rises with higher levels of two cardiac biomarkers in a manner independent of risk calculated by the Framingham Risk Score (FRS), a longitudinal cohort study has shown. But researchers who conducted the study note that neither of the two biomarkers identified in the study – cardiac troponin I (cTnI) and N-terminal pro-brain-type natriuretic peptide (NT-proBNP) – led to an improvement in predictive performance when combined with the FRS, despite their association with carotid plaque burden.
Psoriasis and psoriatic arthritis are both associated with greater risk of CV morbidity and mortality, partly because of systemic inflammation that leads to atherogenesis. Measures of CV risk such as the FRS rely on traditional measures of CV risk and thus are likely to underestimate the CV event risk of people with psoriatic disease, according to the authors of the new study, published online in Arthritis & Rheumatology. The effort was led by Keith Colaço, MSc; Lihi Eder, MD, PhD; and other researchers affiliated with the University of Toronto.
“We are desperately in need of biomarker science advancement in psoriatic arthritis for a variety of places of guidance: How to choose a medication more accurately for the patient in front of us – that is, getting to be more like oncologists who use biomarkers to pick the best treatment or combination. That’s an important need. A second important need is how to guide clinicians regarding risk prediction for things like persistent, severe disease activity, progressive structural damage from disease, and, in this case, predicting a very common comorbidity that occurs in [psoriasis and] psoriatic arthritis patients,” Philip J. Mease, MD, told this news organization when asked to comment on the study.
Such biomarkers could assist with patient counseling, according to Dr. Mease, who is director of rheumatology research at Swedish Medical Center/Providence St. Joseph Health and is a clinical professor at the University of Washington, both in Seattle. Some patients may struggle with advice to lose weight or adopt lifestyle measures to limit CV risk, and more accurate predictions of risk may serve as further motivation. “It could well be that if you have a biomarker that accurately predicts a coming cataclysm, that it will lead you to redouble your efforts to do whatever it takes to reduce cardiovascular risk,” he said.
Both cTnI and NT-proBNP have been linked to increased CV risk in the general population, but little work has been done in the context of rheumatologic diseases.
The researchers analyzed data from 358 patients seen at the University of Toronto. The mean follow-up was 3.69 years. After adjustment for CV risk factors, lipid-lowering therapy, and creatinine levels, there was an association between cTnI levels and total carotid plaque area (adjusted beta coefficient, 0.21; 95% confidence interval, 0-0.41), but not for levels of NT-proBNP.
Atherosclerosis progressed in 89 participants overall, but multivariate adjustment revealed no significant relationship between progression and cTnI or NT-proBNP levels.
Separately, the researchers analyzed 1,000 individuals with psoriatic arthritis (n = 648) or with psoriasis and no arthritis (n = 352) whom they followed for a mean of 7.1 years after the patients underwent evaluation during 2002-2019. After adjustment for FRS, there was an association between the risk of a CV event and each 1–standard deviation increase in both cTnI (hazard ratio, 3.02; 95% CI, 1.12-8.16) and NT-proBNP (HR, 2.02; 95% CI, 1.28-3.18).
The combination of both biomarkers with the FRS predicted higher CV risk (HR, 1.91; 95% CI, 1.23-2.97). Neither biomarker made a statistically significant difference in changing CV risk prediction when added individually to FRS, although cTnI trended toward significance (HR, 2.60; 95% CI, 0.98-6.87).
Instead of the carotid plaque burden, Dr. Mease would have liked to have seen the authors evaluate calcium scores in coronary arteries as measured by CT. “I would have loved to have seen the researchers using that in addition to the carotid plaque assessment, to see what that would show us about these patients,” he said.
Only a small number of patients experienced CV events during the study period, which will likely make it necessary to conduct larger studies to identify a clear relationship. “You need a registry-type study with probably many hundreds if not thousands of patients in order to identify whether or not adding troponin could be useful to what we typically measure with patients when we’re trying to assess their risk,” Dr. Mease said.
The study was supported in part by the National Psoriasis Foundation and the Arthritis Society. Individual researchers have received support from a range of sources, including the Enid Walker Estate, the Women’s College Research Institute, the Arthritis Society, the National Psoriasis Foundation, the Edward Dunlop Foundation, the Ontario Ministry of Science and Innovation, and a Pfizer Chair Research Award. Some of the researchers have financial relationships with pharmaceutical companies that market drugs for psoriasis and psoriatic arthritis.
A version of this article first appeared on Medscape.com.
The risk of cardiovascular (CV) events in patients with psoriatic disease rises with higher levels of two cardiac biomarkers in a manner independent of risk calculated by the Framingham Risk Score (FRS), a longitudinal cohort study has shown. But researchers who conducted the study note that neither of the two biomarkers identified in the study – cardiac troponin I (cTnI) and N-terminal pro-brain-type natriuretic peptide (NT-proBNP) – led to an improvement in predictive performance when combined with the FRS, despite their association with carotid plaque burden.
Psoriasis and psoriatic arthritis are both associated with greater risk of CV morbidity and mortality, partly because of systemic inflammation that leads to atherogenesis. Measures of CV risk such as the FRS rely on traditional measures of CV risk and thus are likely to underestimate the CV event risk of people with psoriatic disease, according to the authors of the new study, published online in Arthritis & Rheumatology. The effort was led by Keith Colaço, MSc; Lihi Eder, MD, PhD; and other researchers affiliated with the University of Toronto.
“We are desperately in need of biomarker science advancement in psoriatic arthritis for a variety of places of guidance: How to choose a medication more accurately for the patient in front of us – that is, getting to be more like oncologists who use biomarkers to pick the best treatment or combination. That’s an important need. A second important need is how to guide clinicians regarding risk prediction for things like persistent, severe disease activity, progressive structural damage from disease, and, in this case, predicting a very common comorbidity that occurs in [psoriasis and] psoriatic arthritis patients,” Philip J. Mease, MD, told this news organization when asked to comment on the study.
Such biomarkers could assist with patient counseling, according to Dr. Mease, who is director of rheumatology research at Swedish Medical Center/Providence St. Joseph Health and is a clinical professor at the University of Washington, both in Seattle. Some patients may struggle with advice to lose weight or adopt lifestyle measures to limit CV risk, and more accurate predictions of risk may serve as further motivation. “It could well be that if you have a biomarker that accurately predicts a coming cataclysm, that it will lead you to redouble your efforts to do whatever it takes to reduce cardiovascular risk,” he said.
Both cTnI and NT-proBNP have been linked to increased CV risk in the general population, but little work has been done in the context of rheumatologic diseases.
The researchers analyzed data from 358 patients seen at the University of Toronto. The mean follow-up was 3.69 years. After adjustment for CV risk factors, lipid-lowering therapy, and creatinine levels, there was an association between cTnI levels and total carotid plaque area (adjusted beta coefficient, 0.21; 95% confidence interval, 0-0.41), but not for levels of NT-proBNP.
Atherosclerosis progressed in 89 participants overall, but multivariate adjustment revealed no significant relationship between progression and cTnI or NT-proBNP levels.
Separately, the researchers analyzed 1,000 individuals with psoriatic arthritis (n = 648) or with psoriasis and no arthritis (n = 352) whom they followed for a mean of 7.1 years after the patients underwent evaluation during 2002-2019. After adjustment for FRS, there was an association between the risk of a CV event and each 1–standard deviation increase in both cTnI (hazard ratio, 3.02; 95% CI, 1.12-8.16) and NT-proBNP (HR, 2.02; 95% CI, 1.28-3.18).
The combination of both biomarkers with the FRS predicted higher CV risk (HR, 1.91; 95% CI, 1.23-2.97). Neither biomarker made a statistically significant difference in changing CV risk prediction when added individually to FRS, although cTnI trended toward significance (HR, 2.60; 95% CI, 0.98-6.87).
Instead of the carotid plaque burden, Dr. Mease would have liked to have seen the authors evaluate calcium scores in coronary arteries as measured by CT. “I would have loved to have seen the researchers using that in addition to the carotid plaque assessment, to see what that would show us about these patients,” he said.
Only a small number of patients experienced CV events during the study period, which will likely make it necessary to conduct larger studies to identify a clear relationship. “You need a registry-type study with probably many hundreds if not thousands of patients in order to identify whether or not adding troponin could be useful to what we typically measure with patients when we’re trying to assess their risk,” Dr. Mease said.
The study was supported in part by the National Psoriasis Foundation and the Arthritis Society. Individual researchers have received support from a range of sources, including the Enid Walker Estate, the Women’s College Research Institute, the Arthritis Society, the National Psoriasis Foundation, the Edward Dunlop Foundation, the Ontario Ministry of Science and Innovation, and a Pfizer Chair Research Award. Some of the researchers have financial relationships with pharmaceutical companies that market drugs for psoriasis and psoriatic arthritis.
A version of this article first appeared on Medscape.com.
FROM ARTHRITIS & RHEUMATOLOGY
Drug survival study looks at what lasts longest in RA, axSpA, PsA, and psoriasis
Survival rates of biologics and other novel immunomodulatory drugs vary substantially across chronic inflammatory diseases, and rates are highest for rituximab in rheumatoid arthritis (RA) and golimumab in axial spondyloarthritis (axSpA), but with similar rates seen for most drugs used in the treatment of psoriasis and psoriatic arthritis (PsA), according to findings from a study of two Danish registries.
Drug survival refers to “the probability that patients will remain on a given drug, and is a proxy for efficacy as well as safety in daily clinical practice,” wrote Alexander Egeberg, MD, PhD, of the department of dermatology at Copenhagen University Hospital–Bispebjerg, and colleagues. Although the use of biologics has expanded for inflammatory diseases, real-world data on drug survival in newer agents such as interleukin (IL)-17, IL-23, and Janus kinase inhibitors are lacking, they said.
In a study published in Seminars in Arthritis and Rheumatism, the researchers reviewed data from the DANBIO and DERMBIO registries of patients in Denmark with inflammatory diseases including rheumatoid arthritis (RA), axial spondyloarthritis (AxSpA), psoriatic arthritis (PsA), and psoriasis.
The study population included 12,089 adults: 5,104 with RA, 2,157 with AxSpA, 2,251 with PsA, and 2,577 with psoriasis. Patients’ mean age at the time of first treatment for these conditions was 57.8 years, 42.3 years, 49 years, and 45 years, respectively. Participants were treated with biologics or novel small molecule therapies for RA, AxSpA, PsA, or psoriasis between January 2015 and May 2021 (from the DANBIO database) and November 2009 to November 2019 (DERMBIO database).
In adjusted models, drug survival in RA was highest for rituximab followed by baricitinib, etanercept, and tocilizumab. Drug survival in AxSpA was highest for golimumab, compared with all other drugs, followed by secukinumab and etanercept. Survival was lowest for infliximab. In PsA, drug survival was roughly equal for most drugs, including golimumab, secukinumab, and ixekizumab, with the lowest survival observed for tofacitinib and infliximab, compared with all other drugs. Drug survival in psoriasis was highest with guselkumab, followed by ustekinumab and IL-17 inhibitors.
However, the number of treatment series “was low for some drugs, and not all differences were statistically significant, which could influence the overall interpretability of these findings,” the researchers noted in their discussion.
Notably, the high treatment persistence for rituximab in RA patients needs further confirmation, the researchers said. “In Denmark, rituximab is often the biologic drug of choice in RA patients with a history of cancer while there is a reluctancy to use TNF [tumor necrosis factor] inhibitors in such patients; this may have prolonged the drug survival for rituximab treated patients due to limited treatment alternatives,” they said.
The findings were limited by several factors, including the observational study design and changes in guidelines over the course of the study, the researchers noted. Other limitations included the inability to adjust for certain variables, such as antibody status, body weight, and smoking, because of missing data, and a lack of data on the underlying reasons for drug discontinuation, they said.
However, the results were strengthened by the large number of patients and completeness of the registries, the researchers emphasized. The range in responses to different drug types across diseases supports the need for individualized treatments with attention to underlying disease, patient profile, and treatment history, they concluded.
The study received no outside funding. Eight coauthors reported financial ties to a number of pharmaceutical companies.
Survival rates of biologics and other novel immunomodulatory drugs vary substantially across chronic inflammatory diseases, and rates are highest for rituximab in rheumatoid arthritis (RA) and golimumab in axial spondyloarthritis (axSpA), but with similar rates seen for most drugs used in the treatment of psoriasis and psoriatic arthritis (PsA), according to findings from a study of two Danish registries.
Drug survival refers to “the probability that patients will remain on a given drug, and is a proxy for efficacy as well as safety in daily clinical practice,” wrote Alexander Egeberg, MD, PhD, of the department of dermatology at Copenhagen University Hospital–Bispebjerg, and colleagues. Although the use of biologics has expanded for inflammatory diseases, real-world data on drug survival in newer agents such as interleukin (IL)-17, IL-23, and Janus kinase inhibitors are lacking, they said.
In a study published in Seminars in Arthritis and Rheumatism, the researchers reviewed data from the DANBIO and DERMBIO registries of patients in Denmark with inflammatory diseases including rheumatoid arthritis (RA), axial spondyloarthritis (AxSpA), psoriatic arthritis (PsA), and psoriasis.
The study population included 12,089 adults: 5,104 with RA, 2,157 with AxSpA, 2,251 with PsA, and 2,577 with psoriasis. Patients’ mean age at the time of first treatment for these conditions was 57.8 years, 42.3 years, 49 years, and 45 years, respectively. Participants were treated with biologics or novel small molecule therapies for RA, AxSpA, PsA, or psoriasis between January 2015 and May 2021 (from the DANBIO database) and November 2009 to November 2019 (DERMBIO database).
In adjusted models, drug survival in RA was highest for rituximab followed by baricitinib, etanercept, and tocilizumab. Drug survival in AxSpA was highest for golimumab, compared with all other drugs, followed by secukinumab and etanercept. Survival was lowest for infliximab. In PsA, drug survival was roughly equal for most drugs, including golimumab, secukinumab, and ixekizumab, with the lowest survival observed for tofacitinib and infliximab, compared with all other drugs. Drug survival in psoriasis was highest with guselkumab, followed by ustekinumab and IL-17 inhibitors.
However, the number of treatment series “was low for some drugs, and not all differences were statistically significant, which could influence the overall interpretability of these findings,” the researchers noted in their discussion.
Notably, the high treatment persistence for rituximab in RA patients needs further confirmation, the researchers said. “In Denmark, rituximab is often the biologic drug of choice in RA patients with a history of cancer while there is a reluctancy to use TNF [tumor necrosis factor] inhibitors in such patients; this may have prolonged the drug survival for rituximab treated patients due to limited treatment alternatives,” they said.
The findings were limited by several factors, including the observational study design and changes in guidelines over the course of the study, the researchers noted. Other limitations included the inability to adjust for certain variables, such as antibody status, body weight, and smoking, because of missing data, and a lack of data on the underlying reasons for drug discontinuation, they said.
However, the results were strengthened by the large number of patients and completeness of the registries, the researchers emphasized. The range in responses to different drug types across diseases supports the need for individualized treatments with attention to underlying disease, patient profile, and treatment history, they concluded.
The study received no outside funding. Eight coauthors reported financial ties to a number of pharmaceutical companies.
Survival rates of biologics and other novel immunomodulatory drugs vary substantially across chronic inflammatory diseases, and rates are highest for rituximab in rheumatoid arthritis (RA) and golimumab in axial spondyloarthritis (axSpA), but with similar rates seen for most drugs used in the treatment of psoriasis and psoriatic arthritis (PsA), according to findings from a study of two Danish registries.
Drug survival refers to “the probability that patients will remain on a given drug, and is a proxy for efficacy as well as safety in daily clinical practice,” wrote Alexander Egeberg, MD, PhD, of the department of dermatology at Copenhagen University Hospital–Bispebjerg, and colleagues. Although the use of biologics has expanded for inflammatory diseases, real-world data on drug survival in newer agents such as interleukin (IL)-17, IL-23, and Janus kinase inhibitors are lacking, they said.
In a study published in Seminars in Arthritis and Rheumatism, the researchers reviewed data from the DANBIO and DERMBIO registries of patients in Denmark with inflammatory diseases including rheumatoid arthritis (RA), axial spondyloarthritis (AxSpA), psoriatic arthritis (PsA), and psoriasis.
The study population included 12,089 adults: 5,104 with RA, 2,157 with AxSpA, 2,251 with PsA, and 2,577 with psoriasis. Patients’ mean age at the time of first treatment for these conditions was 57.8 years, 42.3 years, 49 years, and 45 years, respectively. Participants were treated with biologics or novel small molecule therapies for RA, AxSpA, PsA, or psoriasis between January 2015 and May 2021 (from the DANBIO database) and November 2009 to November 2019 (DERMBIO database).
In adjusted models, drug survival in RA was highest for rituximab followed by baricitinib, etanercept, and tocilizumab. Drug survival in AxSpA was highest for golimumab, compared with all other drugs, followed by secukinumab and etanercept. Survival was lowest for infliximab. In PsA, drug survival was roughly equal for most drugs, including golimumab, secukinumab, and ixekizumab, with the lowest survival observed for tofacitinib and infliximab, compared with all other drugs. Drug survival in psoriasis was highest with guselkumab, followed by ustekinumab and IL-17 inhibitors.
However, the number of treatment series “was low for some drugs, and not all differences were statistically significant, which could influence the overall interpretability of these findings,” the researchers noted in their discussion.
Notably, the high treatment persistence for rituximab in RA patients needs further confirmation, the researchers said. “In Denmark, rituximab is often the biologic drug of choice in RA patients with a history of cancer while there is a reluctancy to use TNF [tumor necrosis factor] inhibitors in such patients; this may have prolonged the drug survival for rituximab treated patients due to limited treatment alternatives,” they said.
The findings were limited by several factors, including the observational study design and changes in guidelines over the course of the study, the researchers noted. Other limitations included the inability to adjust for certain variables, such as antibody status, body weight, and smoking, because of missing data, and a lack of data on the underlying reasons for drug discontinuation, they said.
However, the results were strengthened by the large number of patients and completeness of the registries, the researchers emphasized. The range in responses to different drug types across diseases supports the need for individualized treatments with attention to underlying disease, patient profile, and treatment history, they concluded.
The study received no outside funding. Eight coauthors reported financial ties to a number of pharmaceutical companies.
FROM SEMINARS IN ARTHRITIS AND RHEUMATISM
One-third of psoriatic arthritis patients could have metabolic syndrome, data analysis finds
of 724 individuals, as did approximately 23%-63% of patients across multiple studies, investigators from Spain report.
Previous studies of people with PsA in particular suggest they are at an increased risk of cardiovascular disease and have a higher prevalence of metabolic syndrome, prompting recommendations on cardiovascular risk management for patients with PsA, wrote the authors, Ana Urruticoechea-Arana, MD, of the department of rheumatology, Hospital Can Misses, Ibiza, Spain, and colleagues.
However, assessing the prevalence of metabolic syndrome remains a challenge because the definition varies across studies, they noted.
For a more thorough assessment of the prevalence of metabolic syndrome in this population, the researchers conducted a study using two sources: a systematic literature review of 18 studies published up to March 2019, and data on patients with PsA enrolled in the CARMA (Spanish Cardiovascular in Rheumatology) project, a longitudinal cohort observational study of adults with inflammatory diseases in Spain. The findings were published March 1 in the Journal of Clinical Rheumatology.
The literature review included a total of a total of 2,452 patients with PsA, with a mean age between 42 and 59 years, and a mean disease duration ranging from 3 to 14 years.
The definitions of metabolic syndrome varied; the most common was the definition from the National Cholesterol Education Program (NECP ATP III). Other definitions used in the studies included those issued by the International Diabetes Federation, the World Health Organization, and the American Heart Association.
Across these studies, the rate of metabolic syndrome ranged from 23.5% to 62.9%. Prevalence was similar between men and women. One study that included patients with a PsA disease duration of only 3 years showed a prevalence of 38%, similar to the average prevalence overall. Another study showed a significantly higher prevalence of metabolic syndrome in patients with PsA and cutaneous psoriasis, compared with those without psoriasis (40.8% vs. 13.16%; P = .006).
The CARMA study included 724 patients with PsA; 45.4% were women and 21.8% were smokers. The mean age of the population in this study was 51 years, and the mean disease duration was 9 years. Overall, 222 patients (30.7%) met at least three criteria for metabolic syndrome, based on the NCEP ATP III definition. The most common abnormal findings for traditional cardiovascular risk factors in the CARMA cohort were high blood pressure (66.8%), hyperglycemia (42.6%), and hypertriglyceridemia (30.6%).
Despite the variation in prevalence of metabolic syndrome, depending on the definition used, the authors wrote, “It can be stated that the rate of [metabolic syndrome] in patients with PsA is in general very high, especially if we take into account the mean age of patients included in the studies.”
“These findings support the hypotheses that this increase in the inflammatory pathway in PsA may contribute a higher risk of cardiovascular events and [metabolic syndrome] in patients with PsA than patients with psoriasis alone, the risk being even higher in severe PsA,” and that insulin resistance, metabolic syndrome, and atherosclerotic events “may have a common inflammatory basis,” the researchers wrote in their discussion of the results.
The study findings were limited by several factors, most importantly the variation in definitions of metabolic syndrome in the literature review, which limits the generalizability of the results, the researchers said. Limitations of the CARMA study include the focus only on patients who were being cared for in hospitals, which might yield an overestimation of metabolic syndrome, they added.
However, the results support findings from previous studies and highlight the need for proper assessment of body weight and cardiovascular risk factors in patients with PsA at the onset of disease, they said.
“Furthermore, it is necessary to conduct more research to standardize (and modify as appropriate) the definition of [metabolic syndrome] and establish the best strategy for managing it in these patients,” they concluded.
The study was funded by an independent grant from UCB Pharma. One author disclosed receiving grants from Pfizer, Abbvie, Novartis, Roche, UCB, Sanofi, BMS, Lilly, MSD, and Janssen. Lead author Dr. Urruticoechea-Arana and the other authors had no disclosures.
of 724 individuals, as did approximately 23%-63% of patients across multiple studies, investigators from Spain report.
Previous studies of people with PsA in particular suggest they are at an increased risk of cardiovascular disease and have a higher prevalence of metabolic syndrome, prompting recommendations on cardiovascular risk management for patients with PsA, wrote the authors, Ana Urruticoechea-Arana, MD, of the department of rheumatology, Hospital Can Misses, Ibiza, Spain, and colleagues.
However, assessing the prevalence of metabolic syndrome remains a challenge because the definition varies across studies, they noted.
For a more thorough assessment of the prevalence of metabolic syndrome in this population, the researchers conducted a study using two sources: a systematic literature review of 18 studies published up to March 2019, and data on patients with PsA enrolled in the CARMA (Spanish Cardiovascular in Rheumatology) project, a longitudinal cohort observational study of adults with inflammatory diseases in Spain. The findings were published March 1 in the Journal of Clinical Rheumatology.
The literature review included a total of a total of 2,452 patients with PsA, with a mean age between 42 and 59 years, and a mean disease duration ranging from 3 to 14 years.
The definitions of metabolic syndrome varied; the most common was the definition from the National Cholesterol Education Program (NECP ATP III). Other definitions used in the studies included those issued by the International Diabetes Federation, the World Health Organization, and the American Heart Association.
Across these studies, the rate of metabolic syndrome ranged from 23.5% to 62.9%. Prevalence was similar between men and women. One study that included patients with a PsA disease duration of only 3 years showed a prevalence of 38%, similar to the average prevalence overall. Another study showed a significantly higher prevalence of metabolic syndrome in patients with PsA and cutaneous psoriasis, compared with those without psoriasis (40.8% vs. 13.16%; P = .006).
The CARMA study included 724 patients with PsA; 45.4% were women and 21.8% were smokers. The mean age of the population in this study was 51 years, and the mean disease duration was 9 years. Overall, 222 patients (30.7%) met at least three criteria for metabolic syndrome, based on the NCEP ATP III definition. The most common abnormal findings for traditional cardiovascular risk factors in the CARMA cohort were high blood pressure (66.8%), hyperglycemia (42.6%), and hypertriglyceridemia (30.6%).
Despite the variation in prevalence of metabolic syndrome, depending on the definition used, the authors wrote, “It can be stated that the rate of [metabolic syndrome] in patients with PsA is in general very high, especially if we take into account the mean age of patients included in the studies.”
“These findings support the hypotheses that this increase in the inflammatory pathway in PsA may contribute a higher risk of cardiovascular events and [metabolic syndrome] in patients with PsA than patients with psoriasis alone, the risk being even higher in severe PsA,” and that insulin resistance, metabolic syndrome, and atherosclerotic events “may have a common inflammatory basis,” the researchers wrote in their discussion of the results.
The study findings were limited by several factors, most importantly the variation in definitions of metabolic syndrome in the literature review, which limits the generalizability of the results, the researchers said. Limitations of the CARMA study include the focus only on patients who were being cared for in hospitals, which might yield an overestimation of metabolic syndrome, they added.
However, the results support findings from previous studies and highlight the need for proper assessment of body weight and cardiovascular risk factors in patients with PsA at the onset of disease, they said.
“Furthermore, it is necessary to conduct more research to standardize (and modify as appropriate) the definition of [metabolic syndrome] and establish the best strategy for managing it in these patients,” they concluded.
The study was funded by an independent grant from UCB Pharma. One author disclosed receiving grants from Pfizer, Abbvie, Novartis, Roche, UCB, Sanofi, BMS, Lilly, MSD, and Janssen. Lead author Dr. Urruticoechea-Arana and the other authors had no disclosures.
of 724 individuals, as did approximately 23%-63% of patients across multiple studies, investigators from Spain report.
Previous studies of people with PsA in particular suggest they are at an increased risk of cardiovascular disease and have a higher prevalence of metabolic syndrome, prompting recommendations on cardiovascular risk management for patients with PsA, wrote the authors, Ana Urruticoechea-Arana, MD, of the department of rheumatology, Hospital Can Misses, Ibiza, Spain, and colleagues.
However, assessing the prevalence of metabolic syndrome remains a challenge because the definition varies across studies, they noted.
For a more thorough assessment of the prevalence of metabolic syndrome in this population, the researchers conducted a study using two sources: a systematic literature review of 18 studies published up to March 2019, and data on patients with PsA enrolled in the CARMA (Spanish Cardiovascular in Rheumatology) project, a longitudinal cohort observational study of adults with inflammatory diseases in Spain. The findings were published March 1 in the Journal of Clinical Rheumatology.
The literature review included a total of a total of 2,452 patients with PsA, with a mean age between 42 and 59 years, and a mean disease duration ranging from 3 to 14 years.
The definitions of metabolic syndrome varied; the most common was the definition from the National Cholesterol Education Program (NECP ATP III). Other definitions used in the studies included those issued by the International Diabetes Federation, the World Health Organization, and the American Heart Association.
Across these studies, the rate of metabolic syndrome ranged from 23.5% to 62.9%. Prevalence was similar between men and women. One study that included patients with a PsA disease duration of only 3 years showed a prevalence of 38%, similar to the average prevalence overall. Another study showed a significantly higher prevalence of metabolic syndrome in patients with PsA and cutaneous psoriasis, compared with those without psoriasis (40.8% vs. 13.16%; P = .006).
The CARMA study included 724 patients with PsA; 45.4% were women and 21.8% were smokers. The mean age of the population in this study was 51 years, and the mean disease duration was 9 years. Overall, 222 patients (30.7%) met at least three criteria for metabolic syndrome, based on the NCEP ATP III definition. The most common abnormal findings for traditional cardiovascular risk factors in the CARMA cohort were high blood pressure (66.8%), hyperglycemia (42.6%), and hypertriglyceridemia (30.6%).
Despite the variation in prevalence of metabolic syndrome, depending on the definition used, the authors wrote, “It can be stated that the rate of [metabolic syndrome] in patients with PsA is in general very high, especially if we take into account the mean age of patients included in the studies.”
“These findings support the hypotheses that this increase in the inflammatory pathway in PsA may contribute a higher risk of cardiovascular events and [metabolic syndrome] in patients with PsA than patients with psoriasis alone, the risk being even higher in severe PsA,” and that insulin resistance, metabolic syndrome, and atherosclerotic events “may have a common inflammatory basis,” the researchers wrote in their discussion of the results.
The study findings were limited by several factors, most importantly the variation in definitions of metabolic syndrome in the literature review, which limits the generalizability of the results, the researchers said. Limitations of the CARMA study include the focus only on patients who were being cared for in hospitals, which might yield an overestimation of metabolic syndrome, they added.
However, the results support findings from previous studies and highlight the need for proper assessment of body weight and cardiovascular risk factors in patients with PsA at the onset of disease, they said.
“Furthermore, it is necessary to conduct more research to standardize (and modify as appropriate) the definition of [metabolic syndrome] and establish the best strategy for managing it in these patients,” they concluded.
The study was funded by an independent grant from UCB Pharma. One author disclosed receiving grants from Pfizer, Abbvie, Novartis, Roche, UCB, Sanofi, BMS, Lilly, MSD, and Janssen. Lead author Dr. Urruticoechea-Arana and the other authors had no disclosures.
FROM JOURNAL OF CLINICAL RHEUMATOLOGY
An Update on JAK Inhibitors in Skin Disease
Atopic dermatitis (AD) is a chronic inflammatory skin disorder affecting 7% of adults and 13% of children in the United States.1,2 Atopic dermatitis is characterized by pruritus, dry skin, and pain, all of which can negatively impact quality of life and put patients at higher risk for psychiatric comorbidities such as anxiety and depression.3 The pathogenesis of AD is multifactorial, involving genetics, epidermal barrier dysfunction, and immune dysregulation. Overactivation of helper T cell (TH2) pathway cytokines, including IL-4, IL-13, and IL-31, is thought to propagate both inflammation and pruritus, which are central to AD. The JAK-STAT signaling pathway plays a pivotal role in the immune system dysregulation and exaggeration of TH2 cell response, making JAK-STAT inhibitors (or JAK inhibitors) strong theoretical candidates for the treatment of AD.4 In humans, the Janus kinases are composed of 4 different members—JAK1, JAK2, JAK3, and tyrosine kinase 2—all of which can be targeted by JAK inhibitors.5
JAK inhibitors such as tofacitinib have already been approved by the US Food and Drug Administration (FDA) to treat various inflammatory conditions, including rheumatoid arthritis, ulcerative colitis, and psoriatic arthritis; other JAK inhibitors such as baricitinib are only approved for patients with rheumatoid arthritis.6,7 The success of these small molecule inhibitors in these immune-mediated conditions make them attractive candidates for the treatment of AD. Several JAK inhibitors are in phase 2 and phase 3 clinical trials as oral therapies (moderate to severe AD) or as topical treatments (mild to moderate AD). Currently, ruxolitinib (RUX) is the only topical JAK inhibitor that is FDA approved for the treatment of AD in the United States.8 In this editorial, we focus on recent trials of JAK inhibitors tested in patients with AD, including topical RUX, as well as oral abrocitinib, upadacitinib, and baricitinib.
Topical RUX in AD
Ruxolitinib is a topical JAK1/2 small molecule inhibitor approved by the FDA for the treatment of AD in 2021. In a randomized trial by Kim et al9 in 2020, all tested regimens of RUX demonstrated significant improvement in eczema area and severity index (EASI) scores vs vehicle; notably, RUX cream 1.5% applied twice daily achieved the greatest mean percentage change in baseline EASI score vs vehicle at 4 weeks (76.1% vs 15.5%; P<.0001). Ruxolitinib cream was well tolerated through week 8 of the trial, and all adverse events (AEs) were mild to moderate in severity and comparable to those in the vehicle group.9
Topical JAK inhibitors appear to be effective for mild to moderate AD and have had an acceptable safety profile in clinical trials thus far. Although topical corticosteroids and calcineurin inhibitors can have great clinical benefit in AD, they are recommended for short-term use given side effects such as thinning of the skin, burning, or telangiectasia formation.10,11 The hope is that topical JAK inhibitors may be an alternative to standard topical treatments for AD, as they can be used for longer periods due to a safer side-effect profile.
Oral JAK Inhibitors in AD
Several oral JAK inhibitors are undergoing investigation for the systemic treatment of moderate to severe AD. Abrocitinib is an oral JAK1 inhibitor that has demonstrated efficacy in several phase 3 trials in patients with moderate to severe AD. In a 2021 trial, patients were randomized in a 2:2:2:1 ratio to receive abrocitinib 200 mg daily, abrocitinib 100 mg daily, subcutaneous dupilumab 300 mg every other week, or placebo, respectively.12 Patients in both abrocitinib groups showed significant improvement in AD vs placebo, and EASI-75 response was achieved in 70.3%, 58.7%, 58.1%, and 27.1% of patients, respectively (P<.001 for both abrocitinib doses vs placebo). Adverse events occurred more frequently in the abrocitinib 200-mg group vs placebo. Nausea, acne, nasopharyngitis, and headache were the most frequently reported AEs with abrocitinib.12 Another phase 3 trial by Silverberg et al13 (N=391) had similar treatment results, with 38.1% of participants receiving abrocitinib 200 mg and 28.4% of participants receiving abrocitinib 100 mg achieving investigator global assessment scores of 0 (clear) or 1 (almost clear) vs 9.1% of participants receiving placebo (P<.001). Abrocitinib was well tolerated in this trial with few serious AEs (ie, herpangina [0.6%], pneumonia [0.6%]).13 In both trials, there were rare instances of laboratory values indicating thrombocytopenia with the 200-mg dose (0.9%12 and 3.2%13) without any clinical manifestations. Although a decrease in platelets was observed, no thrombocytopenia occurred in the abrocitinib 100-mg group in the latter trial.13
Baricitinib is another oral inhibitor of JAK1 and JAK2 with potential for the treatment of AD. One randomized trial (N=329) demonstrated its efficacy in combination with a topical corticosteroid (TCS). At 16 weeks, a higher number of participants treated with baricitinib and TCS achieved investigator global assessment scores of 0 (clear) or 1 (almost clear) compared to those who received placebo and TCS (31% with baricitinib 4 mg + TCS, 24% with baricitinib 2 mg + TCS, and 15% with placebo + TCS).14 Similarly, in BREEZE-AD5,another phase 3 trial (N=440), baricitinib monotherapy demonstrated a higher rate of treatment success vs placebo.15 Specifically, 13% of patients treated with baricitinib 1 mg and 30% of those treated with baricitinib 2 mg achieved 75% or greater reduction in EASI scores compared to 8% in the placebo group. The most common AEs associated with baricitinib were nasopharyngitis and headache. Adverse events occurred with similar frequency across both experimental and control groups.15 Reich et al14 demonstrated a higher overall rate of AEs—most commonly nasopharyngitis, upper respiratory tract infections, and folliculitis—in baricitinib-treated patients; however, serious AEs occurred with similar frequency across all groups, including the control group.
The selective JAK1 inhibitor upadacitinib also is undergoing testing in treating moderate to severe AD. In one trial, 167 patients were randomized to once daily oral upadacitinib 7.5 mg, 15 mg, or 30 mg or placebo.16 All doses of upadacitinib demonstrated considerably higher percentage improvements from baseline in EASI scores compared to placebo at 16 weeks with a clear dose-response relationship (39%, 62%, and 74% vs 23%, respectively). In this trial, there were no dose-limiting safety events. Serious AEs were infrequent, occurring in 4.8%, 2.4%, and 0% of upadacitinib groups vs 2.5% for placebo. The serious AEs observed with upadacitinib were 1 case of appendicitis, lower jaw pericoronitis in a patient with a history of repeated tooth infections, and an exacerbation of AD.16
Tofacitinib, another JAK inhibitor, has been shown to increase the risk for blood clots and death in a large trial in the treatment of rheumatoid arthritis. Following this study, the FDA is requiring black box warnings for tofacitinib and also for the 2 JAK inhibitors baricitinib and upadacitinib regarding the risks for heart-related events, cancer, blood clots, and death. Given that these medications share a similar mechanism of action to tofacitinib, they may have similar risks, though they have not yet been fully evaluated in large safety trials.17
With more recent investigation into novel therapeutics for AD, oral JAK inhibitors may play an important role in the future to treat patients with moderate to severe AD with inadequate response or contraindications to other systemic therapies. In trials thus far, oral JAK inhibitors have exhibited acceptable safety profiles and have demonstrated treatment success in AD. More randomized, controlled, phase 3 studies with larger patient populations are required to confirm their potential as effective treatments and elucidate their long-term safety.
Deucravacitinib in Psoriasis
Deucravacitinib is a first-in-class, oral, selective TYK2 inhibitor currently undergoing testing for the treatment of psoriasis. A randomized phase 2 trial (N=267) found that deucravacitinib was more effective than placebo in treating chronic plaque psoriasis at doses of 3 to 12 mg daily.18 The percentage of participants with a 75% or greater reduction from baseline in the psoriasis area and severity index score was 7% with placebo, 9% with deucravacitinib 3 mg every other day (P=.49 vs placebo), 39% with 3 mg once daily (P<.001 vs placebo), 69% with 3 mg twice daily (P<.001 vs placebo), 67% with 6 mg twice daily (P<.001 vs placebo), and 75% with 12 mg once daily (P<.001 vs placebo). The most commonly reported AEs were nasopharyngitis, headache, diarrhea, nausea, and upper respiratory tract infection. Adverse events occurred in 51% of participants in the control group and in 55% to 80% of those in the experimental groups. Additionally, there was 1 reported case of melanoma (stage 0) 96 days after the start of treatment in a patient in the 3-mg once-daily group. Serious AEs occurred in only 0% to 2% of participants who received deucravacitinib.18
Two phase 3 trials—POETYK PSO-1 and POETYK PSO-2 (N=1686)—found deucravacitinib to be notably more effective than both placebo and apremilast in treating psoriasis.19 Among participants receiving deucravacitinib 6 mg daily, 58.7% and 53.6% in the 2 respective trials achieved psoriasis area and severity index 75 response vs 12.7% and 9.4% receiving placebo and 35.1% and 40.2% receiving apremilast. Overall, the treatment was well tolerated, with a low rate of discontinuation of deucravacitinib due to AEs (2.4% of patients on deucravacitinib compared to 3.8% on placebo and 5.2% on apremilast). The most frequently observed AEs with deucravacitinib were nasopharyngitis and upper respiratory tract infection. The full results of these trials are expected to be published soon.19,20
Final Thoughts
Overall, JAK inhibitors are a novel class of therapeutics that may have further success in the treatment of other dermatologic conditions that negatively affect patients’ quality of life and productivity. We should look forward to additional successful trials with these promising medications.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590.
- Silverberg JI , Simpson EL. Associations of childhood eczema severity: a US population-based study. Dermatitis. 2014;25:107-114.
- Schonmann Y, Mansfield KE, Hayes JF, et al. Atopic eczema in adulthood and risk of depression and anxiety: a population-based cohort study. J Allergy Clin Immunol Pract. 2020;8:248-257.e16.
- Bao L, Zhang H, Chan LS. The involvement of the JAK-STAT signaling pathway in chronic inflammatory skin disease atopic dermatitis. JAKSTAT. 2013;2:e24137.
- Villarino AV, Kanno Y, O’Shea JJ. Mechanisms and consequences of Jak-STAT signaling in the immune system. Nat Immunol. 2017;18:374-384.
- Xeljanz FDA approval history. Drugs.com website. Updated December 14, 2021. Accessed February 16, 2022. https://www.drugs.com/history/xeljanz.html
- Mullard A. FDA approves Eli Lilly’s baricitinib. Nat Rev Drug Discov. 2018;17:460.
- FDA approves Opzelura. Drugs.com website. Published September 2021. Accessed February 16, 2022. https://www.drugs.com/newdrugs/fda-approves-opzelura-ruxolitinib-cream-atopic-dermatitis-ad-5666.html
- Kim BS, Sun K, Papp K, et al. Effects of ruxolitinib cream on pruritus and quality of life in atopic dermatitis: results from a phase 2, randomized, dose-ranging, vehicle- and active-controlled study.J Am Acad Dermatol. 2020;82:1305-1313.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2, management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Wollenberg A, Barbarot S, Bieber T, et al. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: part I. J Eur Acad Dermatol Venereol. 2018;32:657-682.
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112.
- Silverberg JI, Simpson EL, Thyssen JP, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:863-873.
- Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:1333-1343.
- Simpson EL, Forman S, Silverberg JI, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis: results from a randomized monotherapy phase 3 trial in the United States and Canada (BREEZE-AD5). J Am Acad Dermatol. 2021;85:62-70.
- Guttman-Yassky E, Thaçi D, Pangan AL, et al. Upadacitinib in adults with moderate to severe atopic dermatitis: 16-week results from a randomized, placebo-controlled trial. J Allergy Clin Immunol. 2020;145:877-884.
- US Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. Published September 1, 2022. Accessed February 16, 2022. https://www.fda.gov/drugs/drug-safety-and-availability/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Papp K, Gordon K, Thaçi D, et al. Phase 2 trial of selective tyrosine kinase 2 inhibition in psoriasis. N Engl J Med. 2018;379:1313-1321.
- Bristol Myers Squibb presents positive data from two pivotal phase 3 psoriasis studies demonstrating superiority of deucravacitinib compared to placebo and Otezla® (apremilast). Press release. Bristol Meyers Squibb. April 23, 2021. Accessed February 16, 2022. https://news.bms.com/news/details/2021/Bristol-Myers-Squibb-Presents-Positive-Data-from-Two-Pivotal-Phase-3-Psoriasis-Studies-Demonstrating-Superiority-of-Deucravacitinib-Compared-to-Placebo-and-Otezla-apremilast/default.aspx
- Armstrong A, Gooderham M, Warren R, et al. Efficacy and safety of deucravacitinib, an oral, selective tyrosine kinase 2 (TYK2) inhibitor, compared with placebo and apremilast in moderate to severe plaque psoriasis: results from the POETYK PSO-1 study [abstract]. Abstract presented at: 2021 American Academy of Dermatology annual meeting; April 23-25, 2021; San Francisco, California.
Atopic dermatitis (AD) is a chronic inflammatory skin disorder affecting 7% of adults and 13% of children in the United States.1,2 Atopic dermatitis is characterized by pruritus, dry skin, and pain, all of which can negatively impact quality of life and put patients at higher risk for psychiatric comorbidities such as anxiety and depression.3 The pathogenesis of AD is multifactorial, involving genetics, epidermal barrier dysfunction, and immune dysregulation. Overactivation of helper T cell (TH2) pathway cytokines, including IL-4, IL-13, and IL-31, is thought to propagate both inflammation and pruritus, which are central to AD. The JAK-STAT signaling pathway plays a pivotal role in the immune system dysregulation and exaggeration of TH2 cell response, making JAK-STAT inhibitors (or JAK inhibitors) strong theoretical candidates for the treatment of AD.4 In humans, the Janus kinases are composed of 4 different members—JAK1, JAK2, JAK3, and tyrosine kinase 2—all of which can be targeted by JAK inhibitors.5
JAK inhibitors such as tofacitinib have already been approved by the US Food and Drug Administration (FDA) to treat various inflammatory conditions, including rheumatoid arthritis, ulcerative colitis, and psoriatic arthritis; other JAK inhibitors such as baricitinib are only approved for patients with rheumatoid arthritis.6,7 The success of these small molecule inhibitors in these immune-mediated conditions make them attractive candidates for the treatment of AD. Several JAK inhibitors are in phase 2 and phase 3 clinical trials as oral therapies (moderate to severe AD) or as topical treatments (mild to moderate AD). Currently, ruxolitinib (RUX) is the only topical JAK inhibitor that is FDA approved for the treatment of AD in the United States.8 In this editorial, we focus on recent trials of JAK inhibitors tested in patients with AD, including topical RUX, as well as oral abrocitinib, upadacitinib, and baricitinib.
Topical RUX in AD
Ruxolitinib is a topical JAK1/2 small molecule inhibitor approved by the FDA for the treatment of AD in 2021. In a randomized trial by Kim et al9 in 2020, all tested regimens of RUX demonstrated significant improvement in eczema area and severity index (EASI) scores vs vehicle; notably, RUX cream 1.5% applied twice daily achieved the greatest mean percentage change in baseline EASI score vs vehicle at 4 weeks (76.1% vs 15.5%; P<.0001). Ruxolitinib cream was well tolerated through week 8 of the trial, and all adverse events (AEs) were mild to moderate in severity and comparable to those in the vehicle group.9
Topical JAK inhibitors appear to be effective for mild to moderate AD and have had an acceptable safety profile in clinical trials thus far. Although topical corticosteroids and calcineurin inhibitors can have great clinical benefit in AD, they are recommended for short-term use given side effects such as thinning of the skin, burning, or telangiectasia formation.10,11 The hope is that topical JAK inhibitors may be an alternative to standard topical treatments for AD, as they can be used for longer periods due to a safer side-effect profile.
Oral JAK Inhibitors in AD
Several oral JAK inhibitors are undergoing investigation for the systemic treatment of moderate to severe AD. Abrocitinib is an oral JAK1 inhibitor that has demonstrated efficacy in several phase 3 trials in patients with moderate to severe AD. In a 2021 trial, patients were randomized in a 2:2:2:1 ratio to receive abrocitinib 200 mg daily, abrocitinib 100 mg daily, subcutaneous dupilumab 300 mg every other week, or placebo, respectively.12 Patients in both abrocitinib groups showed significant improvement in AD vs placebo, and EASI-75 response was achieved in 70.3%, 58.7%, 58.1%, and 27.1% of patients, respectively (P<.001 for both abrocitinib doses vs placebo). Adverse events occurred more frequently in the abrocitinib 200-mg group vs placebo. Nausea, acne, nasopharyngitis, and headache were the most frequently reported AEs with abrocitinib.12 Another phase 3 trial by Silverberg et al13 (N=391) had similar treatment results, with 38.1% of participants receiving abrocitinib 200 mg and 28.4% of participants receiving abrocitinib 100 mg achieving investigator global assessment scores of 0 (clear) or 1 (almost clear) vs 9.1% of participants receiving placebo (P<.001). Abrocitinib was well tolerated in this trial with few serious AEs (ie, herpangina [0.6%], pneumonia [0.6%]).13 In both trials, there were rare instances of laboratory values indicating thrombocytopenia with the 200-mg dose (0.9%12 and 3.2%13) without any clinical manifestations. Although a decrease in platelets was observed, no thrombocytopenia occurred in the abrocitinib 100-mg group in the latter trial.13
Baricitinib is another oral inhibitor of JAK1 and JAK2 with potential for the treatment of AD. One randomized trial (N=329) demonstrated its efficacy in combination with a topical corticosteroid (TCS). At 16 weeks, a higher number of participants treated with baricitinib and TCS achieved investigator global assessment scores of 0 (clear) or 1 (almost clear) compared to those who received placebo and TCS (31% with baricitinib 4 mg + TCS, 24% with baricitinib 2 mg + TCS, and 15% with placebo + TCS).14 Similarly, in BREEZE-AD5,another phase 3 trial (N=440), baricitinib monotherapy demonstrated a higher rate of treatment success vs placebo.15 Specifically, 13% of patients treated with baricitinib 1 mg and 30% of those treated with baricitinib 2 mg achieved 75% or greater reduction in EASI scores compared to 8% in the placebo group. The most common AEs associated with baricitinib were nasopharyngitis and headache. Adverse events occurred with similar frequency across both experimental and control groups.15 Reich et al14 demonstrated a higher overall rate of AEs—most commonly nasopharyngitis, upper respiratory tract infections, and folliculitis—in baricitinib-treated patients; however, serious AEs occurred with similar frequency across all groups, including the control group.
The selective JAK1 inhibitor upadacitinib also is undergoing testing in treating moderate to severe AD. In one trial, 167 patients were randomized to once daily oral upadacitinib 7.5 mg, 15 mg, or 30 mg or placebo.16 All doses of upadacitinib demonstrated considerably higher percentage improvements from baseline in EASI scores compared to placebo at 16 weeks with a clear dose-response relationship (39%, 62%, and 74% vs 23%, respectively). In this trial, there were no dose-limiting safety events. Serious AEs were infrequent, occurring in 4.8%, 2.4%, and 0% of upadacitinib groups vs 2.5% for placebo. The serious AEs observed with upadacitinib were 1 case of appendicitis, lower jaw pericoronitis in a patient with a history of repeated tooth infections, and an exacerbation of AD.16
Tofacitinib, another JAK inhibitor, has been shown to increase the risk for blood clots and death in a large trial in the treatment of rheumatoid arthritis. Following this study, the FDA is requiring black box warnings for tofacitinib and also for the 2 JAK inhibitors baricitinib and upadacitinib regarding the risks for heart-related events, cancer, blood clots, and death. Given that these medications share a similar mechanism of action to tofacitinib, they may have similar risks, though they have not yet been fully evaluated in large safety trials.17
With more recent investigation into novel therapeutics for AD, oral JAK inhibitors may play an important role in the future to treat patients with moderate to severe AD with inadequate response or contraindications to other systemic therapies. In trials thus far, oral JAK inhibitors have exhibited acceptable safety profiles and have demonstrated treatment success in AD. More randomized, controlled, phase 3 studies with larger patient populations are required to confirm their potential as effective treatments and elucidate their long-term safety.
Deucravacitinib in Psoriasis
Deucravacitinib is a first-in-class, oral, selective TYK2 inhibitor currently undergoing testing for the treatment of psoriasis. A randomized phase 2 trial (N=267) found that deucravacitinib was more effective than placebo in treating chronic plaque psoriasis at doses of 3 to 12 mg daily.18 The percentage of participants with a 75% or greater reduction from baseline in the psoriasis area and severity index score was 7% with placebo, 9% with deucravacitinib 3 mg every other day (P=.49 vs placebo), 39% with 3 mg once daily (P<.001 vs placebo), 69% with 3 mg twice daily (P<.001 vs placebo), 67% with 6 mg twice daily (P<.001 vs placebo), and 75% with 12 mg once daily (P<.001 vs placebo). The most commonly reported AEs were nasopharyngitis, headache, diarrhea, nausea, and upper respiratory tract infection. Adverse events occurred in 51% of participants in the control group and in 55% to 80% of those in the experimental groups. Additionally, there was 1 reported case of melanoma (stage 0) 96 days after the start of treatment in a patient in the 3-mg once-daily group. Serious AEs occurred in only 0% to 2% of participants who received deucravacitinib.18
Two phase 3 trials—POETYK PSO-1 and POETYK PSO-2 (N=1686)—found deucravacitinib to be notably more effective than both placebo and apremilast in treating psoriasis.19 Among participants receiving deucravacitinib 6 mg daily, 58.7% and 53.6% in the 2 respective trials achieved psoriasis area and severity index 75 response vs 12.7% and 9.4% receiving placebo and 35.1% and 40.2% receiving apremilast. Overall, the treatment was well tolerated, with a low rate of discontinuation of deucravacitinib due to AEs (2.4% of patients on deucravacitinib compared to 3.8% on placebo and 5.2% on apremilast). The most frequently observed AEs with deucravacitinib were nasopharyngitis and upper respiratory tract infection. The full results of these trials are expected to be published soon.19,20
Final Thoughts
Overall, JAK inhibitors are a novel class of therapeutics that may have further success in the treatment of other dermatologic conditions that negatively affect patients’ quality of life and productivity. We should look forward to additional successful trials with these promising medications.
Atopic dermatitis (AD) is a chronic inflammatory skin disorder affecting 7% of adults and 13% of children in the United States.1,2 Atopic dermatitis is characterized by pruritus, dry skin, and pain, all of which can negatively impact quality of life and put patients at higher risk for psychiatric comorbidities such as anxiety and depression.3 The pathogenesis of AD is multifactorial, involving genetics, epidermal barrier dysfunction, and immune dysregulation. Overactivation of helper T cell (TH2) pathway cytokines, including IL-4, IL-13, and IL-31, is thought to propagate both inflammation and pruritus, which are central to AD. The JAK-STAT signaling pathway plays a pivotal role in the immune system dysregulation and exaggeration of TH2 cell response, making JAK-STAT inhibitors (or JAK inhibitors) strong theoretical candidates for the treatment of AD.4 In humans, the Janus kinases are composed of 4 different members—JAK1, JAK2, JAK3, and tyrosine kinase 2—all of which can be targeted by JAK inhibitors.5
JAK inhibitors such as tofacitinib have already been approved by the US Food and Drug Administration (FDA) to treat various inflammatory conditions, including rheumatoid arthritis, ulcerative colitis, and psoriatic arthritis; other JAK inhibitors such as baricitinib are only approved for patients with rheumatoid arthritis.6,7 The success of these small molecule inhibitors in these immune-mediated conditions make them attractive candidates for the treatment of AD. Several JAK inhibitors are in phase 2 and phase 3 clinical trials as oral therapies (moderate to severe AD) or as topical treatments (mild to moderate AD). Currently, ruxolitinib (RUX) is the only topical JAK inhibitor that is FDA approved for the treatment of AD in the United States.8 In this editorial, we focus on recent trials of JAK inhibitors tested in patients with AD, including topical RUX, as well as oral abrocitinib, upadacitinib, and baricitinib.
Topical RUX in AD
Ruxolitinib is a topical JAK1/2 small molecule inhibitor approved by the FDA for the treatment of AD in 2021. In a randomized trial by Kim et al9 in 2020, all tested regimens of RUX demonstrated significant improvement in eczema area and severity index (EASI) scores vs vehicle; notably, RUX cream 1.5% applied twice daily achieved the greatest mean percentage change in baseline EASI score vs vehicle at 4 weeks (76.1% vs 15.5%; P<.0001). Ruxolitinib cream was well tolerated through week 8 of the trial, and all adverse events (AEs) were mild to moderate in severity and comparable to those in the vehicle group.9
Topical JAK inhibitors appear to be effective for mild to moderate AD and have had an acceptable safety profile in clinical trials thus far. Although topical corticosteroids and calcineurin inhibitors can have great clinical benefit in AD, they are recommended for short-term use given side effects such as thinning of the skin, burning, or telangiectasia formation.10,11 The hope is that topical JAK inhibitors may be an alternative to standard topical treatments for AD, as they can be used for longer periods due to a safer side-effect profile.
Oral JAK Inhibitors in AD
Several oral JAK inhibitors are undergoing investigation for the systemic treatment of moderate to severe AD. Abrocitinib is an oral JAK1 inhibitor that has demonstrated efficacy in several phase 3 trials in patients with moderate to severe AD. In a 2021 trial, patients were randomized in a 2:2:2:1 ratio to receive abrocitinib 200 mg daily, abrocitinib 100 mg daily, subcutaneous dupilumab 300 mg every other week, or placebo, respectively.12 Patients in both abrocitinib groups showed significant improvement in AD vs placebo, and EASI-75 response was achieved in 70.3%, 58.7%, 58.1%, and 27.1% of patients, respectively (P<.001 for both abrocitinib doses vs placebo). Adverse events occurred more frequently in the abrocitinib 200-mg group vs placebo. Nausea, acne, nasopharyngitis, and headache were the most frequently reported AEs with abrocitinib.12 Another phase 3 trial by Silverberg et al13 (N=391) had similar treatment results, with 38.1% of participants receiving abrocitinib 200 mg and 28.4% of participants receiving abrocitinib 100 mg achieving investigator global assessment scores of 0 (clear) or 1 (almost clear) vs 9.1% of participants receiving placebo (P<.001). Abrocitinib was well tolerated in this trial with few serious AEs (ie, herpangina [0.6%], pneumonia [0.6%]).13 In both trials, there were rare instances of laboratory values indicating thrombocytopenia with the 200-mg dose (0.9%12 and 3.2%13) without any clinical manifestations. Although a decrease in platelets was observed, no thrombocytopenia occurred in the abrocitinib 100-mg group in the latter trial.13
Baricitinib is another oral inhibitor of JAK1 and JAK2 with potential for the treatment of AD. One randomized trial (N=329) demonstrated its efficacy in combination with a topical corticosteroid (TCS). At 16 weeks, a higher number of participants treated with baricitinib and TCS achieved investigator global assessment scores of 0 (clear) or 1 (almost clear) compared to those who received placebo and TCS (31% with baricitinib 4 mg + TCS, 24% with baricitinib 2 mg + TCS, and 15% with placebo + TCS).14 Similarly, in BREEZE-AD5,another phase 3 trial (N=440), baricitinib monotherapy demonstrated a higher rate of treatment success vs placebo.15 Specifically, 13% of patients treated with baricitinib 1 mg and 30% of those treated with baricitinib 2 mg achieved 75% or greater reduction in EASI scores compared to 8% in the placebo group. The most common AEs associated with baricitinib were nasopharyngitis and headache. Adverse events occurred with similar frequency across both experimental and control groups.15 Reich et al14 demonstrated a higher overall rate of AEs—most commonly nasopharyngitis, upper respiratory tract infections, and folliculitis—in baricitinib-treated patients; however, serious AEs occurred with similar frequency across all groups, including the control group.
The selective JAK1 inhibitor upadacitinib also is undergoing testing in treating moderate to severe AD. In one trial, 167 patients were randomized to once daily oral upadacitinib 7.5 mg, 15 mg, or 30 mg or placebo.16 All doses of upadacitinib demonstrated considerably higher percentage improvements from baseline in EASI scores compared to placebo at 16 weeks with a clear dose-response relationship (39%, 62%, and 74% vs 23%, respectively). In this trial, there were no dose-limiting safety events. Serious AEs were infrequent, occurring in 4.8%, 2.4%, and 0% of upadacitinib groups vs 2.5% for placebo. The serious AEs observed with upadacitinib were 1 case of appendicitis, lower jaw pericoronitis in a patient with a history of repeated tooth infections, and an exacerbation of AD.16
Tofacitinib, another JAK inhibitor, has been shown to increase the risk for blood clots and death in a large trial in the treatment of rheumatoid arthritis. Following this study, the FDA is requiring black box warnings for tofacitinib and also for the 2 JAK inhibitors baricitinib and upadacitinib regarding the risks for heart-related events, cancer, blood clots, and death. Given that these medications share a similar mechanism of action to tofacitinib, they may have similar risks, though they have not yet been fully evaluated in large safety trials.17
With more recent investigation into novel therapeutics for AD, oral JAK inhibitors may play an important role in the future to treat patients with moderate to severe AD with inadequate response or contraindications to other systemic therapies. In trials thus far, oral JAK inhibitors have exhibited acceptable safety profiles and have demonstrated treatment success in AD. More randomized, controlled, phase 3 studies with larger patient populations are required to confirm their potential as effective treatments and elucidate their long-term safety.
Deucravacitinib in Psoriasis
Deucravacitinib is a first-in-class, oral, selective TYK2 inhibitor currently undergoing testing for the treatment of psoriasis. A randomized phase 2 trial (N=267) found that deucravacitinib was more effective than placebo in treating chronic plaque psoriasis at doses of 3 to 12 mg daily.18 The percentage of participants with a 75% or greater reduction from baseline in the psoriasis area and severity index score was 7% with placebo, 9% with deucravacitinib 3 mg every other day (P=.49 vs placebo), 39% with 3 mg once daily (P<.001 vs placebo), 69% with 3 mg twice daily (P<.001 vs placebo), 67% with 6 mg twice daily (P<.001 vs placebo), and 75% with 12 mg once daily (P<.001 vs placebo). The most commonly reported AEs were nasopharyngitis, headache, diarrhea, nausea, and upper respiratory tract infection. Adverse events occurred in 51% of participants in the control group and in 55% to 80% of those in the experimental groups. Additionally, there was 1 reported case of melanoma (stage 0) 96 days after the start of treatment in a patient in the 3-mg once-daily group. Serious AEs occurred in only 0% to 2% of participants who received deucravacitinib.18
Two phase 3 trials—POETYK PSO-1 and POETYK PSO-2 (N=1686)—found deucravacitinib to be notably more effective than both placebo and apremilast in treating psoriasis.19 Among participants receiving deucravacitinib 6 mg daily, 58.7% and 53.6% in the 2 respective trials achieved psoriasis area and severity index 75 response vs 12.7% and 9.4% receiving placebo and 35.1% and 40.2% receiving apremilast. Overall, the treatment was well tolerated, with a low rate of discontinuation of deucravacitinib due to AEs (2.4% of patients on deucravacitinib compared to 3.8% on placebo and 5.2% on apremilast). The most frequently observed AEs with deucravacitinib were nasopharyngitis and upper respiratory tract infection. The full results of these trials are expected to be published soon.19,20
Final Thoughts
Overall, JAK inhibitors are a novel class of therapeutics that may have further success in the treatment of other dermatologic conditions that negatively affect patients’ quality of life and productivity. We should look forward to additional successful trials with these promising medications.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590.
- Silverberg JI , Simpson EL. Associations of childhood eczema severity: a US population-based study. Dermatitis. 2014;25:107-114.
- Schonmann Y, Mansfield KE, Hayes JF, et al. Atopic eczema in adulthood and risk of depression and anxiety: a population-based cohort study. J Allergy Clin Immunol Pract. 2020;8:248-257.e16.
- Bao L, Zhang H, Chan LS. The involvement of the JAK-STAT signaling pathway in chronic inflammatory skin disease atopic dermatitis. JAKSTAT. 2013;2:e24137.
- Villarino AV, Kanno Y, O’Shea JJ. Mechanisms and consequences of Jak-STAT signaling in the immune system. Nat Immunol. 2017;18:374-384.
- Xeljanz FDA approval history. Drugs.com website. Updated December 14, 2021. Accessed February 16, 2022. https://www.drugs.com/history/xeljanz.html
- Mullard A. FDA approves Eli Lilly’s baricitinib. Nat Rev Drug Discov. 2018;17:460.
- FDA approves Opzelura. Drugs.com website. Published September 2021. Accessed February 16, 2022. https://www.drugs.com/newdrugs/fda-approves-opzelura-ruxolitinib-cream-atopic-dermatitis-ad-5666.html
- Kim BS, Sun K, Papp K, et al. Effects of ruxolitinib cream on pruritus and quality of life in atopic dermatitis: results from a phase 2, randomized, dose-ranging, vehicle- and active-controlled study.J Am Acad Dermatol. 2020;82:1305-1313.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2, management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Wollenberg A, Barbarot S, Bieber T, et al. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: part I. J Eur Acad Dermatol Venereol. 2018;32:657-682.
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112.
- Silverberg JI, Simpson EL, Thyssen JP, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:863-873.
- Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:1333-1343.
- Simpson EL, Forman S, Silverberg JI, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis: results from a randomized monotherapy phase 3 trial in the United States and Canada (BREEZE-AD5). J Am Acad Dermatol. 2021;85:62-70.
- Guttman-Yassky E, Thaçi D, Pangan AL, et al. Upadacitinib in adults with moderate to severe atopic dermatitis: 16-week results from a randomized, placebo-controlled trial. J Allergy Clin Immunol. 2020;145:877-884.
- US Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. Published September 1, 2022. Accessed February 16, 2022. https://www.fda.gov/drugs/drug-safety-and-availability/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Papp K, Gordon K, Thaçi D, et al. Phase 2 trial of selective tyrosine kinase 2 inhibition in psoriasis. N Engl J Med. 2018;379:1313-1321.
- Bristol Myers Squibb presents positive data from two pivotal phase 3 psoriasis studies demonstrating superiority of deucravacitinib compared to placebo and Otezla® (apremilast). Press release. Bristol Meyers Squibb. April 23, 2021. Accessed February 16, 2022. https://news.bms.com/news/details/2021/Bristol-Myers-Squibb-Presents-Positive-Data-from-Two-Pivotal-Phase-3-Psoriasis-Studies-Demonstrating-Superiority-of-Deucravacitinib-Compared-to-Placebo-and-Otezla-apremilast/default.aspx
- Armstrong A, Gooderham M, Warren R, et al. Efficacy and safety of deucravacitinib, an oral, selective tyrosine kinase 2 (TYK2) inhibitor, compared with placebo and apremilast in moderate to severe plaque psoriasis: results from the POETYK PSO-1 study [abstract]. Abstract presented at: 2021 American Academy of Dermatology annual meeting; April 23-25, 2021; San Francisco, California.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590.
- Silverberg JI , Simpson EL. Associations of childhood eczema severity: a US population-based study. Dermatitis. 2014;25:107-114.
- Schonmann Y, Mansfield KE, Hayes JF, et al. Atopic eczema in adulthood and risk of depression and anxiety: a population-based cohort study. J Allergy Clin Immunol Pract. 2020;8:248-257.e16.
- Bao L, Zhang H, Chan LS. The involvement of the JAK-STAT signaling pathway in chronic inflammatory skin disease atopic dermatitis. JAKSTAT. 2013;2:e24137.
- Villarino AV, Kanno Y, O’Shea JJ. Mechanisms and consequences of Jak-STAT signaling in the immune system. Nat Immunol. 2017;18:374-384.
- Xeljanz FDA approval history. Drugs.com website. Updated December 14, 2021. Accessed February 16, 2022. https://www.drugs.com/history/xeljanz.html
- Mullard A. FDA approves Eli Lilly’s baricitinib. Nat Rev Drug Discov. 2018;17:460.
- FDA approves Opzelura. Drugs.com website. Published September 2021. Accessed February 16, 2022. https://www.drugs.com/newdrugs/fda-approves-opzelura-ruxolitinib-cream-atopic-dermatitis-ad-5666.html
- Kim BS, Sun K, Papp K, et al. Effects of ruxolitinib cream on pruritus and quality of life in atopic dermatitis: results from a phase 2, randomized, dose-ranging, vehicle- and active-controlled study.J Am Acad Dermatol. 2020;82:1305-1313.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2, management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Wollenberg A, Barbarot S, Bieber T, et al. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: part I. J Eur Acad Dermatol Venereol. 2018;32:657-682.
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112.
- Silverberg JI, Simpson EL, Thyssen JP, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:863-873.
- Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:1333-1343.
- Simpson EL, Forman S, Silverberg JI, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis: results from a randomized monotherapy phase 3 trial in the United States and Canada (BREEZE-AD5). J Am Acad Dermatol. 2021;85:62-70.
- Guttman-Yassky E, Thaçi D, Pangan AL, et al. Upadacitinib in adults with moderate to severe atopic dermatitis: 16-week results from a randomized, placebo-controlled trial. J Allergy Clin Immunol. 2020;145:877-884.
- US Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. Published September 1, 2022. Accessed February 16, 2022. https://www.fda.gov/drugs/drug-safety-and-availability/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Papp K, Gordon K, Thaçi D, et al. Phase 2 trial of selective tyrosine kinase 2 inhibition in psoriasis. N Engl J Med. 2018;379:1313-1321.
- Bristol Myers Squibb presents positive data from two pivotal phase 3 psoriasis studies demonstrating superiority of deucravacitinib compared to placebo and Otezla® (apremilast). Press release. Bristol Meyers Squibb. April 23, 2021. Accessed February 16, 2022. https://news.bms.com/news/details/2021/Bristol-Myers-Squibb-Presents-Positive-Data-from-Two-Pivotal-Phase-3-Psoriasis-Studies-Demonstrating-Superiority-of-Deucravacitinib-Compared-to-Placebo-and-Otezla-apremilast/default.aspx
- Armstrong A, Gooderham M, Warren R, et al. Efficacy and safety of deucravacitinib, an oral, selective tyrosine kinase 2 (TYK2) inhibitor, compared with placebo and apremilast in moderate to severe plaque psoriasis: results from the POETYK PSO-1 study [abstract]. Abstract presented at: 2021 American Academy of Dermatology annual meeting; April 23-25, 2021; San Francisco, California.
How to make the most of your time with psoriasis patients
In the clinical experience of George Han, MD, PhD, .
“They come in with bags of topical products to show you what they’ve tried,” Dr. Han, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said during the ODAC Dermatology, Aesthetic & Surgical Conference. “And you’re supposed to see this patient, talk to them, and counsel them in about 10 minutes. How do you make time to conduct an efficient psoriasis visit?”
Patients have a long-term battle to get clear, and spending a little longer on the initial visit “pays a lot of dividends,” he said. “Some of these patients are the most thankful patients in our practices, and it truly is gratifying” to see how much they can improve.
Questions about diet
Dr. Han said that psoriasis patients often ask him if, what, or how much they’re eating affects their disease. “But how do you counsel patients about diet when we’re not dietitians? We can at least give some guidance based on available data.”
He referred to a nationwide study of psoriasis patient-reported outcomes and dietary behaviors, which found that the percentage of patients who reported skin improvement was greatest after reducing intake of alcohol (53.8%); gluten (53.4%); and nightshade vegetables, such as tomatoes, potatoes, and peppers (52.1%); and after adding fish oil/omega-3 (44.6%), vegetables (42.5%), and oral vitamin D (41%). He noted that there is a threefold increased incidence of celiac disease in patients with psoriasis.
As for nightshade vegetables, intake leads to increased alkaloids, “which have been known to worsen bowel inflammation such as in IBD [inflammatory bowel disease], but there is a lack of controlled trials examining this in the overall psoriasis population,” Dr. Han said. The Mediterranean diet, he added, “is sensible, and adding olive oil to your diet seems to have a positive effect on ... PASI, while fish oil seems to reduce C-reactive protein.” The data on the effect of vitamin D supplements are mixed, he said.
A separate randomized study evaluated the impact of weight loss in overweight or obese patients with psoriasis, who had not achieved clearance after 4 weeks of systemic treatment. Significantly more of those in the dietary intervention arm reached the weight loss goal of 5% at 20 weeks, and patients in this arm had a median reduction in the Psoriasis Area and Severity Index (PASI) score of almost 50%, compared with almost 26% among those without an active dietary intervention.
Joint pain, PsA
For psoriasis patients who complain of joint pain, he recommends administering quick measures like the five-question Psoriasis Epidemiology Screening Test (PEST) to screen for psoriatic arthritis (PsA), which is available on the National Psoriasis Foundation web site. “I ask patients about swollen, tender joints – specifically hands, wrists, ankles, feet, and toes,” Dr. Han said. Joint stiffness in the morning is a “concerning finding,” which is “more indicative of psoriatic arthritis than vague knee or back pain that worsens with use. If you have a younger patient with back pain who has a reduced ability to flex their spine, think axial disease.”
Tumor necrosis factor (TNF)–alpha inhibitors are considered first- and second-line treatment for PsA, but interleukin (IL)–17 inhibitors are generally considered just as effective overall. “The IL-23 inhibitors have mixed signals,” said Dr. Han, who is also on the NPF’s medical board. “We know that guselkumab is effective against psoriatic arthritis, but there is no inhibition of joint progression at the approved dosage on the label – though it was pretty close.”
Risankizumab (Skyrizi), an IL-23 inhibitor, was approved in January 2022 for adults with PsA and while the American College of Rheumatology response data “look reasonably good, the results for inhibition of radiographic progression are quite far off and it’s not in the label,” he said. Tildrakizumab (Ilumya), an IL-23 inhibitor, “looks impressive in phase 2b trials. It will be interesting to see if there is differentiation between the IL-23 agents in treating joint disease going forward.”
Dr. Han considers biologic therapy a good option for patients with questionable joint involvement or very limited joint disease. “If the patient has some evidence of PsA, as long as it’s a medication that has approval for that, I’m OK with starting it,” he said. “However, for patients whose joint pain dominates over the skin, or [who] have severe joint disease at presentation, I would prioritize the TNF-alpha inhibitors and IL-17s and refer them to rheumatology for shared management.”
Topical, oral treatments
As for topical approaches to treating psoriasis, adding halobetasol propionate 0.01% to tazarotene 0.045% may have a synergistic effect, while tapinarof 1% cream holds promise, he said. Tapinarof, which is expected to be approved this year, is an investigative aryl hydrocarbon agonist that inhibits an array of proinflammatory cytokines, including interferon-gamma and TNF-alpha. “It has been shown to have inhibitory effects both on Th17 cytokines and Th2 cytokines,” Dr. Han said. “What’s nice about this is that patients still appear to have treatment effect 1-2 months after stopping the drug.”
Another topical agent now under FDA review for psoriasis, is roflumilast, a phosphodiesterase type 4 (PDE4) inhibitor, which has been shown to have a treatment efficacy of 30% or more. “We’ll see how this works into our treatment regimen for psoriasis,” he said, as strategies targeting PDE4 have already been reported to help treat psoriasis.
With regards to oral therapies, he said that there are concerns about the efficacy of the oral PDE4 inhibitor apremilast, approved for psoriasis, compared with other biologics. Deucravacitinib, an oral selective tyrosine kinase 2 (TYK2) inhibitor also under FDA review for psoriasis, “may fill this gap, because its efficacy seems much stronger and really capitalizes on blocking IL-23, which we know is a central pathway in the pathogenesis of psoriasis.”
Phototherapy is another treatment option. Home narrowband-UVB devices cost $3,000-$5,000, “which is a fraction of 1 year of biologic treatment,” Dr. Han said. Older data on phototherapy suggest that “lesions can clear within 2-3 months, depending on how often you do the phototherapy, while newer data suggest that 75% of patients can achieve clear or minimal disease” with phototherapy.
Biologic therapy
If patients meet criteria for treatment with a biologic, he begins the conversation by saying, “I don’t want to give you an immunosuppressant, but your psoriasis represents an overactivation of inflammation in your body, so in some way we have to bring that down. Ideally, we would target your immune system in a way that targets psoriasis very narrowly, while leaving it to do what it needs to: protecting against infections and neoplasia.”
XXXIL-17 inhibitors generally have the fastest onset of action, Dr. Han noted. Authors of a review paper found that achievement of Psoriasis Area and Severity Index (PASI) 50 was 1.8 weeks with brodalumab, 1.9 weeks for ixekizumab, 3 weeks for high-dose secukinumab, 3.5 weeks for adalimumab, 3.7 weeks for infliximab, 5.1 weeks for low-dose ustekinumab, 6.5 weeks for high-dose etanercept, and 10.9 weeks with low-dose etanercept, while achievement of PASI 50 was closer to 1 month for IL-23 inhibitors.
“The conversation I have with patients on IL-23 inhibitors is, ‘we’re in this for the long haul,’ otherwise they come in 2 months later,” he said. “They may have gotten clearer but we’re talking about getting well over half of our patients to PASI 100, or to clear or minimal disease, and they may not have gotten there yet. It helps to frame expectations.”
Dr. Han disclosed that he is a consultant to, a speaker for, or has received research support from Beiersdorf, CeraVe, Celgene, Janssen, Lilly, MC2, Pfizer, UCB, Boehringer Ingelheim, Bond Avillion, Athenex, Amgen, AbbVie, Regeneron/Sanofi, LEO Pharma, Ortho Dermatologics, BMS, Sun Pharma, Dermavant, Dermtech, MedX, Novartis, and Castle Biosciences.
In the clinical experience of George Han, MD, PhD, .
“They come in with bags of topical products to show you what they’ve tried,” Dr. Han, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said during the ODAC Dermatology, Aesthetic & Surgical Conference. “And you’re supposed to see this patient, talk to them, and counsel them in about 10 minutes. How do you make time to conduct an efficient psoriasis visit?”
Patients have a long-term battle to get clear, and spending a little longer on the initial visit “pays a lot of dividends,” he said. “Some of these patients are the most thankful patients in our practices, and it truly is gratifying” to see how much they can improve.
Questions about diet
Dr. Han said that psoriasis patients often ask him if, what, or how much they’re eating affects their disease. “But how do you counsel patients about diet when we’re not dietitians? We can at least give some guidance based on available data.”
He referred to a nationwide study of psoriasis patient-reported outcomes and dietary behaviors, which found that the percentage of patients who reported skin improvement was greatest after reducing intake of alcohol (53.8%); gluten (53.4%); and nightshade vegetables, such as tomatoes, potatoes, and peppers (52.1%); and after adding fish oil/omega-3 (44.6%), vegetables (42.5%), and oral vitamin D (41%). He noted that there is a threefold increased incidence of celiac disease in patients with psoriasis.
As for nightshade vegetables, intake leads to increased alkaloids, “which have been known to worsen bowel inflammation such as in IBD [inflammatory bowel disease], but there is a lack of controlled trials examining this in the overall psoriasis population,” Dr. Han said. The Mediterranean diet, he added, “is sensible, and adding olive oil to your diet seems to have a positive effect on ... PASI, while fish oil seems to reduce C-reactive protein.” The data on the effect of vitamin D supplements are mixed, he said.
A separate randomized study evaluated the impact of weight loss in overweight or obese patients with psoriasis, who had not achieved clearance after 4 weeks of systemic treatment. Significantly more of those in the dietary intervention arm reached the weight loss goal of 5% at 20 weeks, and patients in this arm had a median reduction in the Psoriasis Area and Severity Index (PASI) score of almost 50%, compared with almost 26% among those without an active dietary intervention.
Joint pain, PsA
For psoriasis patients who complain of joint pain, he recommends administering quick measures like the five-question Psoriasis Epidemiology Screening Test (PEST) to screen for psoriatic arthritis (PsA), which is available on the National Psoriasis Foundation web site. “I ask patients about swollen, tender joints – specifically hands, wrists, ankles, feet, and toes,” Dr. Han said. Joint stiffness in the morning is a “concerning finding,” which is “more indicative of psoriatic arthritis than vague knee or back pain that worsens with use. If you have a younger patient with back pain who has a reduced ability to flex their spine, think axial disease.”
Tumor necrosis factor (TNF)–alpha inhibitors are considered first- and second-line treatment for PsA, but interleukin (IL)–17 inhibitors are generally considered just as effective overall. “The IL-23 inhibitors have mixed signals,” said Dr. Han, who is also on the NPF’s medical board. “We know that guselkumab is effective against psoriatic arthritis, but there is no inhibition of joint progression at the approved dosage on the label – though it was pretty close.”
Risankizumab (Skyrizi), an IL-23 inhibitor, was approved in January 2022 for adults with PsA and while the American College of Rheumatology response data “look reasonably good, the results for inhibition of radiographic progression are quite far off and it’s not in the label,” he said. Tildrakizumab (Ilumya), an IL-23 inhibitor, “looks impressive in phase 2b trials. It will be interesting to see if there is differentiation between the IL-23 agents in treating joint disease going forward.”
Dr. Han considers biologic therapy a good option for patients with questionable joint involvement or very limited joint disease. “If the patient has some evidence of PsA, as long as it’s a medication that has approval for that, I’m OK with starting it,” he said. “However, for patients whose joint pain dominates over the skin, or [who] have severe joint disease at presentation, I would prioritize the TNF-alpha inhibitors and IL-17s and refer them to rheumatology for shared management.”
Topical, oral treatments
As for topical approaches to treating psoriasis, adding halobetasol propionate 0.01% to tazarotene 0.045% may have a synergistic effect, while tapinarof 1% cream holds promise, he said. Tapinarof, which is expected to be approved this year, is an investigative aryl hydrocarbon agonist that inhibits an array of proinflammatory cytokines, including interferon-gamma and TNF-alpha. “It has been shown to have inhibitory effects both on Th17 cytokines and Th2 cytokines,” Dr. Han said. “What’s nice about this is that patients still appear to have treatment effect 1-2 months after stopping the drug.”
Another topical agent now under FDA review for psoriasis, is roflumilast, a phosphodiesterase type 4 (PDE4) inhibitor, which has been shown to have a treatment efficacy of 30% or more. “We’ll see how this works into our treatment regimen for psoriasis,” he said, as strategies targeting PDE4 have already been reported to help treat psoriasis.
With regards to oral therapies, he said that there are concerns about the efficacy of the oral PDE4 inhibitor apremilast, approved for psoriasis, compared with other biologics. Deucravacitinib, an oral selective tyrosine kinase 2 (TYK2) inhibitor also under FDA review for psoriasis, “may fill this gap, because its efficacy seems much stronger and really capitalizes on blocking IL-23, which we know is a central pathway in the pathogenesis of psoriasis.”
Phototherapy is another treatment option. Home narrowband-UVB devices cost $3,000-$5,000, “which is a fraction of 1 year of biologic treatment,” Dr. Han said. Older data on phototherapy suggest that “lesions can clear within 2-3 months, depending on how often you do the phototherapy, while newer data suggest that 75% of patients can achieve clear or minimal disease” with phototherapy.
Biologic therapy
If patients meet criteria for treatment with a biologic, he begins the conversation by saying, “I don’t want to give you an immunosuppressant, but your psoriasis represents an overactivation of inflammation in your body, so in some way we have to bring that down. Ideally, we would target your immune system in a way that targets psoriasis very narrowly, while leaving it to do what it needs to: protecting against infections and neoplasia.”
XXXIL-17 inhibitors generally have the fastest onset of action, Dr. Han noted. Authors of a review paper found that achievement of Psoriasis Area and Severity Index (PASI) 50 was 1.8 weeks with brodalumab, 1.9 weeks for ixekizumab, 3 weeks for high-dose secukinumab, 3.5 weeks for adalimumab, 3.7 weeks for infliximab, 5.1 weeks for low-dose ustekinumab, 6.5 weeks for high-dose etanercept, and 10.9 weeks with low-dose etanercept, while achievement of PASI 50 was closer to 1 month for IL-23 inhibitors.
“The conversation I have with patients on IL-23 inhibitors is, ‘we’re in this for the long haul,’ otherwise they come in 2 months later,” he said. “They may have gotten clearer but we’re talking about getting well over half of our patients to PASI 100, or to clear or minimal disease, and they may not have gotten there yet. It helps to frame expectations.”
Dr. Han disclosed that he is a consultant to, a speaker for, or has received research support from Beiersdorf, CeraVe, Celgene, Janssen, Lilly, MC2, Pfizer, UCB, Boehringer Ingelheim, Bond Avillion, Athenex, Amgen, AbbVie, Regeneron/Sanofi, LEO Pharma, Ortho Dermatologics, BMS, Sun Pharma, Dermavant, Dermtech, MedX, Novartis, and Castle Biosciences.
In the clinical experience of George Han, MD, PhD, .
“They come in with bags of topical products to show you what they’ve tried,” Dr. Han, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said during the ODAC Dermatology, Aesthetic & Surgical Conference. “And you’re supposed to see this patient, talk to them, and counsel them in about 10 minutes. How do you make time to conduct an efficient psoriasis visit?”
Patients have a long-term battle to get clear, and spending a little longer on the initial visit “pays a lot of dividends,” he said. “Some of these patients are the most thankful patients in our practices, and it truly is gratifying” to see how much they can improve.
Questions about diet
Dr. Han said that psoriasis patients often ask him if, what, or how much they’re eating affects their disease. “But how do you counsel patients about diet when we’re not dietitians? We can at least give some guidance based on available data.”
He referred to a nationwide study of psoriasis patient-reported outcomes and dietary behaviors, which found that the percentage of patients who reported skin improvement was greatest after reducing intake of alcohol (53.8%); gluten (53.4%); and nightshade vegetables, such as tomatoes, potatoes, and peppers (52.1%); and after adding fish oil/omega-3 (44.6%), vegetables (42.5%), and oral vitamin D (41%). He noted that there is a threefold increased incidence of celiac disease in patients with psoriasis.
As for nightshade vegetables, intake leads to increased alkaloids, “which have been known to worsen bowel inflammation such as in IBD [inflammatory bowel disease], but there is a lack of controlled trials examining this in the overall psoriasis population,” Dr. Han said. The Mediterranean diet, he added, “is sensible, and adding olive oil to your diet seems to have a positive effect on ... PASI, while fish oil seems to reduce C-reactive protein.” The data on the effect of vitamin D supplements are mixed, he said.
A separate randomized study evaluated the impact of weight loss in overweight or obese patients with psoriasis, who had not achieved clearance after 4 weeks of systemic treatment. Significantly more of those in the dietary intervention arm reached the weight loss goal of 5% at 20 weeks, and patients in this arm had a median reduction in the Psoriasis Area and Severity Index (PASI) score of almost 50%, compared with almost 26% among those without an active dietary intervention.
Joint pain, PsA
For psoriasis patients who complain of joint pain, he recommends administering quick measures like the five-question Psoriasis Epidemiology Screening Test (PEST) to screen for psoriatic arthritis (PsA), which is available on the National Psoriasis Foundation web site. “I ask patients about swollen, tender joints – specifically hands, wrists, ankles, feet, and toes,” Dr. Han said. Joint stiffness in the morning is a “concerning finding,” which is “more indicative of psoriatic arthritis than vague knee or back pain that worsens with use. If you have a younger patient with back pain who has a reduced ability to flex their spine, think axial disease.”
Tumor necrosis factor (TNF)–alpha inhibitors are considered first- and second-line treatment for PsA, but interleukin (IL)–17 inhibitors are generally considered just as effective overall. “The IL-23 inhibitors have mixed signals,” said Dr. Han, who is also on the NPF’s medical board. “We know that guselkumab is effective against psoriatic arthritis, but there is no inhibition of joint progression at the approved dosage on the label – though it was pretty close.”
Risankizumab (Skyrizi), an IL-23 inhibitor, was approved in January 2022 for adults with PsA and while the American College of Rheumatology response data “look reasonably good, the results for inhibition of radiographic progression are quite far off and it’s not in the label,” he said. Tildrakizumab (Ilumya), an IL-23 inhibitor, “looks impressive in phase 2b trials. It will be interesting to see if there is differentiation between the IL-23 agents in treating joint disease going forward.”
Dr. Han considers biologic therapy a good option for patients with questionable joint involvement or very limited joint disease. “If the patient has some evidence of PsA, as long as it’s a medication that has approval for that, I’m OK with starting it,” he said. “However, for patients whose joint pain dominates over the skin, or [who] have severe joint disease at presentation, I would prioritize the TNF-alpha inhibitors and IL-17s and refer them to rheumatology for shared management.”
Topical, oral treatments
As for topical approaches to treating psoriasis, adding halobetasol propionate 0.01% to tazarotene 0.045% may have a synergistic effect, while tapinarof 1% cream holds promise, he said. Tapinarof, which is expected to be approved this year, is an investigative aryl hydrocarbon agonist that inhibits an array of proinflammatory cytokines, including interferon-gamma and TNF-alpha. “It has been shown to have inhibitory effects both on Th17 cytokines and Th2 cytokines,” Dr. Han said. “What’s nice about this is that patients still appear to have treatment effect 1-2 months after stopping the drug.”
Another topical agent now under FDA review for psoriasis, is roflumilast, a phosphodiesterase type 4 (PDE4) inhibitor, which has been shown to have a treatment efficacy of 30% or more. “We’ll see how this works into our treatment regimen for psoriasis,” he said, as strategies targeting PDE4 have already been reported to help treat psoriasis.
With regards to oral therapies, he said that there are concerns about the efficacy of the oral PDE4 inhibitor apremilast, approved for psoriasis, compared with other biologics. Deucravacitinib, an oral selective tyrosine kinase 2 (TYK2) inhibitor also under FDA review for psoriasis, “may fill this gap, because its efficacy seems much stronger and really capitalizes on blocking IL-23, which we know is a central pathway in the pathogenesis of psoriasis.”
Phototherapy is another treatment option. Home narrowband-UVB devices cost $3,000-$5,000, “which is a fraction of 1 year of biologic treatment,” Dr. Han said. Older data on phototherapy suggest that “lesions can clear within 2-3 months, depending on how often you do the phototherapy, while newer data suggest that 75% of patients can achieve clear or minimal disease” with phototherapy.
Biologic therapy
If patients meet criteria for treatment with a biologic, he begins the conversation by saying, “I don’t want to give you an immunosuppressant, but your psoriasis represents an overactivation of inflammation in your body, so in some way we have to bring that down. Ideally, we would target your immune system in a way that targets psoriasis very narrowly, while leaving it to do what it needs to: protecting against infections and neoplasia.”
XXXIL-17 inhibitors generally have the fastest onset of action, Dr. Han noted. Authors of a review paper found that achievement of Psoriasis Area and Severity Index (PASI) 50 was 1.8 weeks with brodalumab, 1.9 weeks for ixekizumab, 3 weeks for high-dose secukinumab, 3.5 weeks for adalimumab, 3.7 weeks for infliximab, 5.1 weeks for low-dose ustekinumab, 6.5 weeks for high-dose etanercept, and 10.9 weeks with low-dose etanercept, while achievement of PASI 50 was closer to 1 month for IL-23 inhibitors.
“The conversation I have with patients on IL-23 inhibitors is, ‘we’re in this for the long haul,’ otherwise they come in 2 months later,” he said. “They may have gotten clearer but we’re talking about getting well over half of our patients to PASI 100, or to clear or minimal disease, and they may not have gotten there yet. It helps to frame expectations.”
Dr. Han disclosed that he is a consultant to, a speaker for, or has received research support from Beiersdorf, CeraVe, Celgene, Janssen, Lilly, MC2, Pfizer, UCB, Boehringer Ingelheim, Bond Avillion, Athenex, Amgen, AbbVie, Regeneron/Sanofi, LEO Pharma, Ortho Dermatologics, BMS, Sun Pharma, Dermavant, Dermtech, MedX, Novartis, and Castle Biosciences.
FROM ODAC 2022