User login
For MD-IQ use only
Hospital acquisition had no significant change in the rate of readmission or mortality
Background: Prior studies have examined the impact of hospital system mergers on health care costs, but few studies have previously examined impact on quality and patient experience.
Study design: Retrospective, difference-in-difference analysis.
Setting: 2,232 U.S. hospitals during 2007-2016.
Synopsis: The authors identified 2,232 hospitals, including 246 hospitals that were acquired between 2009 and 2013 and 1,986 control hospitals that were not acquired during this period. They used a difference-in-difference analysis to compare hospital performance on quality and patient experience measures from before and after an acquisition to concurrent changes in control hospitals. Hospital acquisition was associated with a significant decline in measured patient experience. There was no significant differential change in 30-day readmission or mortality. Although there was an association between acquisition and significant improvement in clinical process metrics, the authors found that this improvement occurred almost entirely prior to acquisition.
Bottom line: Hospital acquisition was associated with worse experience for patients and had no significant impact on readmission or mortality rates.
Citation: Beaulieu ND et al. Changes in quality of care after hospital mergers and acquisitions. N Engl J Med. 2020 Jan 2;382:51-9.
Dr. Midha is a hospitalist at Beth Israel Deaconess Medical Center, instructor of medicine, Boston University, and part-time instructor in medicine, Harvard Medical School, all in Boston.
Background: Prior studies have examined the impact of hospital system mergers on health care costs, but few studies have previously examined impact on quality and patient experience.
Study design: Retrospective, difference-in-difference analysis.
Setting: 2,232 U.S. hospitals during 2007-2016.
Synopsis: The authors identified 2,232 hospitals, including 246 hospitals that were acquired between 2009 and 2013 and 1,986 control hospitals that were not acquired during this period. They used a difference-in-difference analysis to compare hospital performance on quality and patient experience measures from before and after an acquisition to concurrent changes in control hospitals. Hospital acquisition was associated with a significant decline in measured patient experience. There was no significant differential change in 30-day readmission or mortality. Although there was an association between acquisition and significant improvement in clinical process metrics, the authors found that this improvement occurred almost entirely prior to acquisition.
Bottom line: Hospital acquisition was associated with worse experience for patients and had no significant impact on readmission or mortality rates.
Citation: Beaulieu ND et al. Changes in quality of care after hospital mergers and acquisitions. N Engl J Med. 2020 Jan 2;382:51-9.
Dr. Midha is a hospitalist at Beth Israel Deaconess Medical Center, instructor of medicine, Boston University, and part-time instructor in medicine, Harvard Medical School, all in Boston.
Background: Prior studies have examined the impact of hospital system mergers on health care costs, but few studies have previously examined impact on quality and patient experience.
Study design: Retrospective, difference-in-difference analysis.
Setting: 2,232 U.S. hospitals during 2007-2016.
Synopsis: The authors identified 2,232 hospitals, including 246 hospitals that were acquired between 2009 and 2013 and 1,986 control hospitals that were not acquired during this period. They used a difference-in-difference analysis to compare hospital performance on quality and patient experience measures from before and after an acquisition to concurrent changes in control hospitals. Hospital acquisition was associated with a significant decline in measured patient experience. There was no significant differential change in 30-day readmission or mortality. Although there was an association between acquisition and significant improvement in clinical process metrics, the authors found that this improvement occurred almost entirely prior to acquisition.
Bottom line: Hospital acquisition was associated with worse experience for patients and had no significant impact on readmission or mortality rates.
Citation: Beaulieu ND et al. Changes in quality of care after hospital mergers and acquisitions. N Engl J Med. 2020 Jan 2;382:51-9.
Dr. Midha is a hospitalist at Beth Israel Deaconess Medical Center, instructor of medicine, Boston University, and part-time instructor in medicine, Harvard Medical School, all in Boston.
New obesity target? Dopamine circuit in brainstem affects satiety
Researchers have discovered a new dopaminergic neural circuit leading to the hindbrain that is involved in satiety (feeling full and eating cessation) in mice, which may eventually lead to new ways to treat obesity.
Moreover, when mice were given methylphenidate (Ritalin, Concerta) – a stimulant approved to treat attention deficit hyperactivity disorder (ADHD) with a well-known side effect of decreasing appetite – signals in this dopaminergic pathway were enhanced and the mice ate less.
The study by Yong Han, PhD, a postdoctoral associate at Baylor College of Medicine, Houston, and colleagues was published online May 27 in Science Advances.
“We identified a new dopamine neural circuit from the midbrain to the hindbrain (brainstem) that regulates feeding behavior through an enhanced satiation response,” senior author Qi Wu, PhD, assistant professor in pediatrics-nutrition at Baylor College of Medicine, summarized in an interview.
The findings suggest that “people with obesity have a compromised dopaminergic neural pathway, presumably in ways that delay the satiation response, which makes them eat more, have a larger meal,” he explained.
Newly identified brain circuit plays a key role in satiety response
The study is about a circuit in the brain that helps precisely regulate the size of food portion consumed, Dr. Wu emphasized in a statement from the university, adding that the satiation response is as important as appetite.
Importantly, the results also provide clues about how methylphenidate can lead to weight loss.
Regulators have deemed that methylphenidate, a controlled substance with other side effects such as anxiety and a fast heart rate, is safe and effective for ADHD, Dr. Wu noted.
He speculated that, “If researchers want to do clinical trials of methylphenidate for obesity, it ultimately could evolve to be an anti-obesity drug, alone or combined with other drugs, or possibly derivatives of methylphenidate could be tested.”
The brain circuit “we discovered is the first to be fully described to regulate portion size via dopamine signaling,” Dr. Han stressed in the statement.
“Our new study shows that a circuit connecting neurons that produce dopamine, a chemical messenger previously known for the regulation of motivation and pleasure, has a new [critical] role in the control of feeding through dynamically regulating the satiety response,” he explained.
Brain signals that control portion size
Earlier studies that investigated how the dopaminergic system may regulate food intake, appetite, and body weight, have produced conflicting results, Dr. Wu said.
The researchers performed several experiments in mice that included the use of cell-specific circuitry mapping, optogenetics, and real-time recordings of brain activity.
They identified a new dopaminergic neural circuit comprised of dopaminergic neurons in the caudal ventral tegmental area (DA-VTA neurons) in the midbrain that directly innervate dopamine receptor D1-expressing neurons within the lateral parabrachial nucleus (DRD1-LPBN neurons) in the hindbrain.
There were four main findings:
- DA-VTA neurons were activated immediately before the cessation of each feeding bout.
- Actively inhibiting DA-VTA neurons before the end of each feeding bout prolonged the feeding.
- Activating DRD1-LPBN neurons inhibited feeding.
- Mice that lacked the DRD1 gene ate much more and gained weight.
“Our study illuminates a hindbrain dopaminergic circuit that controls feeding through dynamic regulation in satiety response and meal structure,” the researchers reiterate.
The study was supported by grants from the National Institutes of Health, NIH Digestive Diseases Center, Pew Charitable Trust, American Diabetes Association, Baylor Collaborative Faculty Research Investment Program, USDA/CRIS, USDA/ARS, American Heart Association, and NIH Centers of Biomedical Research Excellence, and by Pew and Kavli scholarships. The researchers have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Researchers have discovered a new dopaminergic neural circuit leading to the hindbrain that is involved in satiety (feeling full and eating cessation) in mice, which may eventually lead to new ways to treat obesity.
Moreover, when mice were given methylphenidate (Ritalin, Concerta) – a stimulant approved to treat attention deficit hyperactivity disorder (ADHD) with a well-known side effect of decreasing appetite – signals in this dopaminergic pathway were enhanced and the mice ate less.
The study by Yong Han, PhD, a postdoctoral associate at Baylor College of Medicine, Houston, and colleagues was published online May 27 in Science Advances.
“We identified a new dopamine neural circuit from the midbrain to the hindbrain (brainstem) that regulates feeding behavior through an enhanced satiation response,” senior author Qi Wu, PhD, assistant professor in pediatrics-nutrition at Baylor College of Medicine, summarized in an interview.
The findings suggest that “people with obesity have a compromised dopaminergic neural pathway, presumably in ways that delay the satiation response, which makes them eat more, have a larger meal,” he explained.
Newly identified brain circuit plays a key role in satiety response
The study is about a circuit in the brain that helps precisely regulate the size of food portion consumed, Dr. Wu emphasized in a statement from the university, adding that the satiation response is as important as appetite.
Importantly, the results also provide clues about how methylphenidate can lead to weight loss.
Regulators have deemed that methylphenidate, a controlled substance with other side effects such as anxiety and a fast heart rate, is safe and effective for ADHD, Dr. Wu noted.
He speculated that, “If researchers want to do clinical trials of methylphenidate for obesity, it ultimately could evolve to be an anti-obesity drug, alone or combined with other drugs, or possibly derivatives of methylphenidate could be tested.”
The brain circuit “we discovered is the first to be fully described to regulate portion size via dopamine signaling,” Dr. Han stressed in the statement.
“Our new study shows that a circuit connecting neurons that produce dopamine, a chemical messenger previously known for the regulation of motivation and pleasure, has a new [critical] role in the control of feeding through dynamically regulating the satiety response,” he explained.
Brain signals that control portion size
Earlier studies that investigated how the dopaminergic system may regulate food intake, appetite, and body weight, have produced conflicting results, Dr. Wu said.
The researchers performed several experiments in mice that included the use of cell-specific circuitry mapping, optogenetics, and real-time recordings of brain activity.
They identified a new dopaminergic neural circuit comprised of dopaminergic neurons in the caudal ventral tegmental area (DA-VTA neurons) in the midbrain that directly innervate dopamine receptor D1-expressing neurons within the lateral parabrachial nucleus (DRD1-LPBN neurons) in the hindbrain.
There were four main findings:
- DA-VTA neurons were activated immediately before the cessation of each feeding bout.
- Actively inhibiting DA-VTA neurons before the end of each feeding bout prolonged the feeding.
- Activating DRD1-LPBN neurons inhibited feeding.
- Mice that lacked the DRD1 gene ate much more and gained weight.
“Our study illuminates a hindbrain dopaminergic circuit that controls feeding through dynamic regulation in satiety response and meal structure,” the researchers reiterate.
The study was supported by grants from the National Institutes of Health, NIH Digestive Diseases Center, Pew Charitable Trust, American Diabetes Association, Baylor Collaborative Faculty Research Investment Program, USDA/CRIS, USDA/ARS, American Heart Association, and NIH Centers of Biomedical Research Excellence, and by Pew and Kavli scholarships. The researchers have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Researchers have discovered a new dopaminergic neural circuit leading to the hindbrain that is involved in satiety (feeling full and eating cessation) in mice, which may eventually lead to new ways to treat obesity.
Moreover, when mice were given methylphenidate (Ritalin, Concerta) – a stimulant approved to treat attention deficit hyperactivity disorder (ADHD) with a well-known side effect of decreasing appetite – signals in this dopaminergic pathway were enhanced and the mice ate less.
The study by Yong Han, PhD, a postdoctoral associate at Baylor College of Medicine, Houston, and colleagues was published online May 27 in Science Advances.
“We identified a new dopamine neural circuit from the midbrain to the hindbrain (brainstem) that regulates feeding behavior through an enhanced satiation response,” senior author Qi Wu, PhD, assistant professor in pediatrics-nutrition at Baylor College of Medicine, summarized in an interview.
The findings suggest that “people with obesity have a compromised dopaminergic neural pathway, presumably in ways that delay the satiation response, which makes them eat more, have a larger meal,” he explained.
Newly identified brain circuit plays a key role in satiety response
The study is about a circuit in the brain that helps precisely regulate the size of food portion consumed, Dr. Wu emphasized in a statement from the university, adding that the satiation response is as important as appetite.
Importantly, the results also provide clues about how methylphenidate can lead to weight loss.
Regulators have deemed that methylphenidate, a controlled substance with other side effects such as anxiety and a fast heart rate, is safe and effective for ADHD, Dr. Wu noted.
He speculated that, “If researchers want to do clinical trials of methylphenidate for obesity, it ultimately could evolve to be an anti-obesity drug, alone or combined with other drugs, or possibly derivatives of methylphenidate could be tested.”
The brain circuit “we discovered is the first to be fully described to regulate portion size via dopamine signaling,” Dr. Han stressed in the statement.
“Our new study shows that a circuit connecting neurons that produce dopamine, a chemical messenger previously known for the regulation of motivation and pleasure, has a new [critical] role in the control of feeding through dynamically regulating the satiety response,” he explained.
Brain signals that control portion size
Earlier studies that investigated how the dopaminergic system may regulate food intake, appetite, and body weight, have produced conflicting results, Dr. Wu said.
The researchers performed several experiments in mice that included the use of cell-specific circuitry mapping, optogenetics, and real-time recordings of brain activity.
They identified a new dopaminergic neural circuit comprised of dopaminergic neurons in the caudal ventral tegmental area (DA-VTA neurons) in the midbrain that directly innervate dopamine receptor D1-expressing neurons within the lateral parabrachial nucleus (DRD1-LPBN neurons) in the hindbrain.
There were four main findings:
- DA-VTA neurons were activated immediately before the cessation of each feeding bout.
- Actively inhibiting DA-VTA neurons before the end of each feeding bout prolonged the feeding.
- Activating DRD1-LPBN neurons inhibited feeding.
- Mice that lacked the DRD1 gene ate much more and gained weight.
“Our study illuminates a hindbrain dopaminergic circuit that controls feeding through dynamic regulation in satiety response and meal structure,” the researchers reiterate.
The study was supported by grants from the National Institutes of Health, NIH Digestive Diseases Center, Pew Charitable Trust, American Diabetes Association, Baylor Collaborative Faculty Research Investment Program, USDA/CRIS, USDA/ARS, American Heart Association, and NIH Centers of Biomedical Research Excellence, and by Pew and Kavli scholarships. The researchers have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Electronic frailty index based on routine blood tests may help identify at-risk seniors
Background: Accurate identification of frail older patients at hospital admission may help target interventions; however, the extent to which risk prediction tools such as the frailty index can be utilized in the acute setting remains unclear.
Study design: Single-center prospective cohort study, during April 2015–January 2017.
Setting: A tertiary care, academic medical center in the United Kingdom.
Synopsis: This study enrolled 1,750 older adults, comprising 2,552 hospital admissions. For each admission, the authors generated a frailty index, called FI-Laboratory, based on the proportion of abnormal results from 27 of the most common admission laboratory tests. The authors found that an increase in the FI-Lab was significantly associated, independent of an existing chronic frailty score, with increased proportion of inpatient days, discharge to a higher level of care, readmission rates, and mortality. Notably, researchers were unable to calculate the FI-Lab score in 11.6% of cases because of insufficient laboratory information. The single-center design of this study may limit its generalizability.
Bottom line: The FI-Laboratory may provide information, complementary to existing frailty assessments, to help identify older adults at increased risk of inpatient adverse outcomes.
Citation: Logan Ellis H et al. Complementing chronic frailty assessment at hospital admission with an electronic frailty index (FI-Laboratory) comprising routine blood test results. CMAJ. 2020;192(1)e3-8.
Dr. Hu is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, both in Boston.
Background: Accurate identification of frail older patients at hospital admission may help target interventions; however, the extent to which risk prediction tools such as the frailty index can be utilized in the acute setting remains unclear.
Study design: Single-center prospective cohort study, during April 2015–January 2017.
Setting: A tertiary care, academic medical center in the United Kingdom.
Synopsis: This study enrolled 1,750 older adults, comprising 2,552 hospital admissions. For each admission, the authors generated a frailty index, called FI-Laboratory, based on the proportion of abnormal results from 27 of the most common admission laboratory tests. The authors found that an increase in the FI-Lab was significantly associated, independent of an existing chronic frailty score, with increased proportion of inpatient days, discharge to a higher level of care, readmission rates, and mortality. Notably, researchers were unable to calculate the FI-Lab score in 11.6% of cases because of insufficient laboratory information. The single-center design of this study may limit its generalizability.
Bottom line: The FI-Laboratory may provide information, complementary to existing frailty assessments, to help identify older adults at increased risk of inpatient adverse outcomes.
Citation: Logan Ellis H et al. Complementing chronic frailty assessment at hospital admission with an electronic frailty index (FI-Laboratory) comprising routine blood test results. CMAJ. 2020;192(1)e3-8.
Dr. Hu is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, both in Boston.
Background: Accurate identification of frail older patients at hospital admission may help target interventions; however, the extent to which risk prediction tools such as the frailty index can be utilized in the acute setting remains unclear.
Study design: Single-center prospective cohort study, during April 2015–January 2017.
Setting: A tertiary care, academic medical center in the United Kingdom.
Synopsis: This study enrolled 1,750 older adults, comprising 2,552 hospital admissions. For each admission, the authors generated a frailty index, called FI-Laboratory, based on the proportion of abnormal results from 27 of the most common admission laboratory tests. The authors found that an increase in the FI-Lab was significantly associated, independent of an existing chronic frailty score, with increased proportion of inpatient days, discharge to a higher level of care, readmission rates, and mortality. Notably, researchers were unable to calculate the FI-Lab score in 11.6% of cases because of insufficient laboratory information. The single-center design of this study may limit its generalizability.
Bottom line: The FI-Laboratory may provide information, complementary to existing frailty assessments, to help identify older adults at increased risk of inpatient adverse outcomes.
Citation: Logan Ellis H et al. Complementing chronic frailty assessment at hospital admission with an electronic frailty index (FI-Laboratory) comprising routine blood test results. CMAJ. 2020;192(1)e3-8.
Dr. Hu is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, both in Boston.
FDA approves ‘game changer’ semaglutide for weight loss
The U.S. Food and Drug Administration has approved a 2.4 mg/week subcutaneous dose of the glucagonlike peptide–1 (GLP-1) receptor agonist semaglutide (Wegovy, Novo Nordisk) for weight loss.
Specifically, this drug format and dosage are approved as an adjunct to a reduced-calorie diet and increased physical activity to treat adults who have obesity (body mass index [BMI] ≥ 30 kg/m2) or are overweight (BMI ≥ 27 kg/m2) with at least one weight-related comorbidity.
Semaglutide “induces weight loss by reducing hunger, increasing feelings of fullness, and thereby helping people eat less and reduce their calorie intake,” according to a company statement.
Novo Nordisk plans to launch Wegovy later this month in the United States. The prescribing information can be found here.
This weight-loss drug is currently under review by the European Medicines Agency.
Several experts told Medscape that they believe the approval of this drug – as long as it is reimbursed – has the potential to change the paradigm of care when it comes to weight loss.
‘Game changer’ drug tested in STEP clinical trial program
The favorable FDA ruling is based on results from the Semaglutide Treatment Effect in People With Obesity (STEP) program of four phase 3 clinical trials that tested the drug’s safety and efficacy in more than 4,500 adults with overweight or obesity obesity who were randomized to receive a reduced a calorie meal plan and increased physical activity (placebo) or this lifestyle intervention plus semaglutide.
The four 68-week trials of subcutaneous semaglutide 2.4 mg/week versus placebo were published in February and March 2021.
As previously reported by this news organization, all trials were in adults with overweight or obesity:
- was in 1,961 adults (N Engl J Med. 2021 March 18;384:989-1002).
- was in 1,210 adults who also had diabetes (Lancet. 2021 Mar 13;397;971-84).
- was in 611 adults, where those in the treatment group also underwent an intensive lifestyle intervention (JAMA. 2021 Feb 24;325:1403-13.
- was in 803 adults who had reached a target dose of 2.4 mg semaglutide after a 20-week run-in (and the trial examined further weight loss in the subsequent 48 weeks) (JAMA 2021 Mar 23;325:1414-25).
In the STEP 1, 2, and 4 trials of individuals with overweight and obesity, those in the semaglutide groups attained a 15%-18% weight loss over 68 weeks.
The dosage was well-tolerated. The most common side effects were gastrointestinal, and they were transient and mild or moderate in severity.
The side effects, contraindications, and a black box warning about thyroid C-cell tumors are spelled out in the prescribing information.
A coauthor of the STEP 1 trial, Rachel Batterham, MBBS, PhD, of the Centre for Obesity Research at University College London, said at the time of publication: “The findings of this study represent a major breakthrough for improving the health of people with obesity.”
“No other drug has come close to producing this level of weight loss – this really is a gamechanger. For the first time, people can achieve through drugs what was only possible through weight-loss surgery,” she added.
Welcome Addition, But Will Insurance Coverage, Price Thwart Access?
Thomas A. Wadden, PhD, from the Perelman School of Medicine at the University of Pennsylvania, Philadelphia, and lead author of STEP 3, commented in an email to this news organization that “semaglutide 2.4 mg appears to be the breakthrough in weight management that healthcare providers and their patients with obesity have been waiting for.”
The mean 15% weight loss at 68 weeks is nearly twice what is seen with other FDA-approved anti-obesity medications, he noted, and moreover, 70% of patients taking semaglutide lost at least 10% of their initial weight, which is associated with clinically meaningful improvements in obesity-related type 2 diabetes, hypertension, obstructive sleep apnea, and impaired quality of life.
And “nearly one-third of users are likely to lose 20% or more of their starting weight, an outcome which eludes traditional diet and exercise interventions and which approaches weight losses produced by the most widely performed bariatric surgery, sleeve gastrectomy (with mean losses of 25% of initial weight at 1 year).” Dr. Wadden stressed.
Thus “the efficacy of semaglutide 2.4 mg, combined with its favorable safety profile, makes this medication a potential game changer,” he summarized, echoing Dr. Batterham.
However, insurance coverage and price could block uptake.
“I hope that the millions of people – in the U.S. and worldwide – who could benefit from this medication eventually will have access to it,” said Dr. Wadden. “In the U.S., the coverage of anti-obesity medications by insurers and employers will need to improve to ensure this happens, and the medication must be reasonably priced. These changes are critical to making this medication the game changer it could be.”
“This approval is an important development,” Scott Kahan, MD, director of the National Center for Weight and Wellness, Washington, who was not involved in the clinical trials of this drug, similarly wrote in an email.
“In a field with relatively few medication options, the availability of additional obesity pharmacotherapy agents is welcome,” he said. “In particular, semaglutide has shown impressive efficacy and safety data; as such it should be a valuable clinical option for many patients.”
However, it is concerning that “access to obesity treatments has traditionally been a challenge,” Dr. Kahan warned. “Novo Nordisk’s other obesity medication, Saxenda, has been a valuable tool, but one that exceedingly few patients are able to utilize due to minimal insurance reimbursement and very high cost.”
“It remains to be seen how accessible semaglutide will be for patients,” according to Dr. Kahan, “Still, if the challenge of limited coverage and high cost can be mitigated, this medication has a chance to significantly change the current paradigm of care, which until till now has included minimal use of pharmacotherapy outside specialty clinics,” he maintains.
Lower-dose injectable and pill already approved for diabetes
Subcutaneous semaglutide at doses up to 1 mg/week (Ozempic, Novo Nordisk), which comes as prefilled pens at doses of 0.5 mg or 1.0 mg, is already approved for the treatment of type 2 diabetes.
The company is also applying for approval for a higher dose of semaglutide, 2 mg/week, for use in type 2 diabetes, and has just resubmitted its label expansion application to the FDA, after the agency issued a refusal to file letter in March.
And in September 2019, the FDA approved oral semaglutide (Rybelsus, Novo Nordisk), in doses of 7 and 14 mg/day, to improve glycemic control in type 2 diabetes, making it the first GLP-1 receptor agonist available in tablet form.
CVOT and oral format trials for obesity on the horizon
The ongoing Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT) trial will shed light on cardiovascular outcomes after 2.5-5 years in patients with cardiovascular disease and overweight or obesity but without type 2 diabetes. Participants will receive semaglutide in doses up to a maximum of 2.4 mg/week, or placebo, as an adjunct to lifestyle recommendations focused on cardiovascular risk reduction. The study is expected to complete in 2023.
And Novo Nordisk plans to initiate a global 68-week phase 3 trial in the second half of 2021 on the efficacy and safety of oral semaglutide 50 mg compared with placebo in 1000 people with obesity or overweight and comorbidities.
A version of this article first appeared on Medscape.com.
This article was updated 6/7/21.
The U.S. Food and Drug Administration has approved a 2.4 mg/week subcutaneous dose of the glucagonlike peptide–1 (GLP-1) receptor agonist semaglutide (Wegovy, Novo Nordisk) for weight loss.
Specifically, this drug format and dosage are approved as an adjunct to a reduced-calorie diet and increased physical activity to treat adults who have obesity (body mass index [BMI] ≥ 30 kg/m2) or are overweight (BMI ≥ 27 kg/m2) with at least one weight-related comorbidity.
Semaglutide “induces weight loss by reducing hunger, increasing feelings of fullness, and thereby helping people eat less and reduce their calorie intake,” according to a company statement.
Novo Nordisk plans to launch Wegovy later this month in the United States. The prescribing information can be found here.
This weight-loss drug is currently under review by the European Medicines Agency.
Several experts told Medscape that they believe the approval of this drug – as long as it is reimbursed – has the potential to change the paradigm of care when it comes to weight loss.
‘Game changer’ drug tested in STEP clinical trial program
The favorable FDA ruling is based on results from the Semaglutide Treatment Effect in People With Obesity (STEP) program of four phase 3 clinical trials that tested the drug’s safety and efficacy in more than 4,500 adults with overweight or obesity obesity who were randomized to receive a reduced a calorie meal plan and increased physical activity (placebo) or this lifestyle intervention plus semaglutide.
The four 68-week trials of subcutaneous semaglutide 2.4 mg/week versus placebo were published in February and March 2021.
As previously reported by this news organization, all trials were in adults with overweight or obesity:
- was in 1,961 adults (N Engl J Med. 2021 March 18;384:989-1002).
- was in 1,210 adults who also had diabetes (Lancet. 2021 Mar 13;397;971-84).
- was in 611 adults, where those in the treatment group also underwent an intensive lifestyle intervention (JAMA. 2021 Feb 24;325:1403-13.
- was in 803 adults who had reached a target dose of 2.4 mg semaglutide after a 20-week run-in (and the trial examined further weight loss in the subsequent 48 weeks) (JAMA 2021 Mar 23;325:1414-25).
In the STEP 1, 2, and 4 trials of individuals with overweight and obesity, those in the semaglutide groups attained a 15%-18% weight loss over 68 weeks.
The dosage was well-tolerated. The most common side effects were gastrointestinal, and they were transient and mild or moderate in severity.
The side effects, contraindications, and a black box warning about thyroid C-cell tumors are spelled out in the prescribing information.
A coauthor of the STEP 1 trial, Rachel Batterham, MBBS, PhD, of the Centre for Obesity Research at University College London, said at the time of publication: “The findings of this study represent a major breakthrough for improving the health of people with obesity.”
“No other drug has come close to producing this level of weight loss – this really is a gamechanger. For the first time, people can achieve through drugs what was only possible through weight-loss surgery,” she added.
Welcome Addition, But Will Insurance Coverage, Price Thwart Access?
Thomas A. Wadden, PhD, from the Perelman School of Medicine at the University of Pennsylvania, Philadelphia, and lead author of STEP 3, commented in an email to this news organization that “semaglutide 2.4 mg appears to be the breakthrough in weight management that healthcare providers and their patients with obesity have been waiting for.”
The mean 15% weight loss at 68 weeks is nearly twice what is seen with other FDA-approved anti-obesity medications, he noted, and moreover, 70% of patients taking semaglutide lost at least 10% of their initial weight, which is associated with clinically meaningful improvements in obesity-related type 2 diabetes, hypertension, obstructive sleep apnea, and impaired quality of life.
And “nearly one-third of users are likely to lose 20% or more of their starting weight, an outcome which eludes traditional diet and exercise interventions and which approaches weight losses produced by the most widely performed bariatric surgery, sleeve gastrectomy (with mean losses of 25% of initial weight at 1 year).” Dr. Wadden stressed.
Thus “the efficacy of semaglutide 2.4 mg, combined with its favorable safety profile, makes this medication a potential game changer,” he summarized, echoing Dr. Batterham.
However, insurance coverage and price could block uptake.
“I hope that the millions of people – in the U.S. and worldwide – who could benefit from this medication eventually will have access to it,” said Dr. Wadden. “In the U.S., the coverage of anti-obesity medications by insurers and employers will need to improve to ensure this happens, and the medication must be reasonably priced. These changes are critical to making this medication the game changer it could be.”
“This approval is an important development,” Scott Kahan, MD, director of the National Center for Weight and Wellness, Washington, who was not involved in the clinical trials of this drug, similarly wrote in an email.
“In a field with relatively few medication options, the availability of additional obesity pharmacotherapy agents is welcome,” he said. “In particular, semaglutide has shown impressive efficacy and safety data; as such it should be a valuable clinical option for many patients.”
However, it is concerning that “access to obesity treatments has traditionally been a challenge,” Dr. Kahan warned. “Novo Nordisk’s other obesity medication, Saxenda, has been a valuable tool, but one that exceedingly few patients are able to utilize due to minimal insurance reimbursement and very high cost.”
“It remains to be seen how accessible semaglutide will be for patients,” according to Dr. Kahan, “Still, if the challenge of limited coverage and high cost can be mitigated, this medication has a chance to significantly change the current paradigm of care, which until till now has included minimal use of pharmacotherapy outside specialty clinics,” he maintains.
Lower-dose injectable and pill already approved for diabetes
Subcutaneous semaglutide at doses up to 1 mg/week (Ozempic, Novo Nordisk), which comes as prefilled pens at doses of 0.5 mg or 1.0 mg, is already approved for the treatment of type 2 diabetes.
The company is also applying for approval for a higher dose of semaglutide, 2 mg/week, for use in type 2 diabetes, and has just resubmitted its label expansion application to the FDA, after the agency issued a refusal to file letter in March.
And in September 2019, the FDA approved oral semaglutide (Rybelsus, Novo Nordisk), in doses of 7 and 14 mg/day, to improve glycemic control in type 2 diabetes, making it the first GLP-1 receptor agonist available in tablet form.
CVOT and oral format trials for obesity on the horizon
The ongoing Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT) trial will shed light on cardiovascular outcomes after 2.5-5 years in patients with cardiovascular disease and overweight or obesity but without type 2 diabetes. Participants will receive semaglutide in doses up to a maximum of 2.4 mg/week, or placebo, as an adjunct to lifestyle recommendations focused on cardiovascular risk reduction. The study is expected to complete in 2023.
And Novo Nordisk plans to initiate a global 68-week phase 3 trial in the second half of 2021 on the efficacy and safety of oral semaglutide 50 mg compared with placebo in 1000 people with obesity or overweight and comorbidities.
A version of this article first appeared on Medscape.com.
This article was updated 6/7/21.
The U.S. Food and Drug Administration has approved a 2.4 mg/week subcutaneous dose of the glucagonlike peptide–1 (GLP-1) receptor agonist semaglutide (Wegovy, Novo Nordisk) for weight loss.
Specifically, this drug format and dosage are approved as an adjunct to a reduced-calorie diet and increased physical activity to treat adults who have obesity (body mass index [BMI] ≥ 30 kg/m2) or are overweight (BMI ≥ 27 kg/m2) with at least one weight-related comorbidity.
Semaglutide “induces weight loss by reducing hunger, increasing feelings of fullness, and thereby helping people eat less and reduce their calorie intake,” according to a company statement.
Novo Nordisk plans to launch Wegovy later this month in the United States. The prescribing information can be found here.
This weight-loss drug is currently under review by the European Medicines Agency.
Several experts told Medscape that they believe the approval of this drug – as long as it is reimbursed – has the potential to change the paradigm of care when it comes to weight loss.
‘Game changer’ drug tested in STEP clinical trial program
The favorable FDA ruling is based on results from the Semaglutide Treatment Effect in People With Obesity (STEP) program of four phase 3 clinical trials that tested the drug’s safety and efficacy in more than 4,500 adults with overweight or obesity obesity who were randomized to receive a reduced a calorie meal plan and increased physical activity (placebo) or this lifestyle intervention plus semaglutide.
The four 68-week trials of subcutaneous semaglutide 2.4 mg/week versus placebo were published in February and March 2021.
As previously reported by this news organization, all trials were in adults with overweight or obesity:
- was in 1,961 adults (N Engl J Med. 2021 March 18;384:989-1002).
- was in 1,210 adults who also had diabetes (Lancet. 2021 Mar 13;397;971-84).
- was in 611 adults, where those in the treatment group also underwent an intensive lifestyle intervention (JAMA. 2021 Feb 24;325:1403-13.
- was in 803 adults who had reached a target dose of 2.4 mg semaglutide after a 20-week run-in (and the trial examined further weight loss in the subsequent 48 weeks) (JAMA 2021 Mar 23;325:1414-25).
In the STEP 1, 2, and 4 trials of individuals with overweight and obesity, those in the semaglutide groups attained a 15%-18% weight loss over 68 weeks.
The dosage was well-tolerated. The most common side effects were gastrointestinal, and they were transient and mild or moderate in severity.
The side effects, contraindications, and a black box warning about thyroid C-cell tumors are spelled out in the prescribing information.
A coauthor of the STEP 1 trial, Rachel Batterham, MBBS, PhD, of the Centre for Obesity Research at University College London, said at the time of publication: “The findings of this study represent a major breakthrough for improving the health of people with obesity.”
“No other drug has come close to producing this level of weight loss – this really is a gamechanger. For the first time, people can achieve through drugs what was only possible through weight-loss surgery,” she added.
Welcome Addition, But Will Insurance Coverage, Price Thwart Access?
Thomas A. Wadden, PhD, from the Perelman School of Medicine at the University of Pennsylvania, Philadelphia, and lead author of STEP 3, commented in an email to this news organization that “semaglutide 2.4 mg appears to be the breakthrough in weight management that healthcare providers and their patients with obesity have been waiting for.”
The mean 15% weight loss at 68 weeks is nearly twice what is seen with other FDA-approved anti-obesity medications, he noted, and moreover, 70% of patients taking semaglutide lost at least 10% of their initial weight, which is associated with clinically meaningful improvements in obesity-related type 2 diabetes, hypertension, obstructive sleep apnea, and impaired quality of life.
And “nearly one-third of users are likely to lose 20% or more of their starting weight, an outcome which eludes traditional diet and exercise interventions and which approaches weight losses produced by the most widely performed bariatric surgery, sleeve gastrectomy (with mean losses of 25% of initial weight at 1 year).” Dr. Wadden stressed.
Thus “the efficacy of semaglutide 2.4 mg, combined with its favorable safety profile, makes this medication a potential game changer,” he summarized, echoing Dr. Batterham.
However, insurance coverage and price could block uptake.
“I hope that the millions of people – in the U.S. and worldwide – who could benefit from this medication eventually will have access to it,” said Dr. Wadden. “In the U.S., the coverage of anti-obesity medications by insurers and employers will need to improve to ensure this happens, and the medication must be reasonably priced. These changes are critical to making this medication the game changer it could be.”
“This approval is an important development,” Scott Kahan, MD, director of the National Center for Weight and Wellness, Washington, who was not involved in the clinical trials of this drug, similarly wrote in an email.
“In a field with relatively few medication options, the availability of additional obesity pharmacotherapy agents is welcome,” he said. “In particular, semaglutide has shown impressive efficacy and safety data; as such it should be a valuable clinical option for many patients.”
However, it is concerning that “access to obesity treatments has traditionally been a challenge,” Dr. Kahan warned. “Novo Nordisk’s other obesity medication, Saxenda, has been a valuable tool, but one that exceedingly few patients are able to utilize due to minimal insurance reimbursement and very high cost.”
“It remains to be seen how accessible semaglutide will be for patients,” according to Dr. Kahan, “Still, if the challenge of limited coverage and high cost can be mitigated, this medication has a chance to significantly change the current paradigm of care, which until till now has included minimal use of pharmacotherapy outside specialty clinics,” he maintains.
Lower-dose injectable and pill already approved for diabetes
Subcutaneous semaglutide at doses up to 1 mg/week (Ozempic, Novo Nordisk), which comes as prefilled pens at doses of 0.5 mg or 1.0 mg, is already approved for the treatment of type 2 diabetes.
The company is also applying for approval for a higher dose of semaglutide, 2 mg/week, for use in type 2 diabetes, and has just resubmitted its label expansion application to the FDA, after the agency issued a refusal to file letter in March.
And in September 2019, the FDA approved oral semaglutide (Rybelsus, Novo Nordisk), in doses of 7 and 14 mg/day, to improve glycemic control in type 2 diabetes, making it the first GLP-1 receptor agonist available in tablet form.
CVOT and oral format trials for obesity on the horizon
The ongoing Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity (SELECT) trial will shed light on cardiovascular outcomes after 2.5-5 years in patients with cardiovascular disease and overweight or obesity but without type 2 diabetes. Participants will receive semaglutide in doses up to a maximum of 2.4 mg/week, or placebo, as an adjunct to lifestyle recommendations focused on cardiovascular risk reduction. The study is expected to complete in 2023.
And Novo Nordisk plans to initiate a global 68-week phase 3 trial in the second half of 2021 on the efficacy and safety of oral semaglutide 50 mg compared with placebo in 1000 people with obesity or overweight and comorbidities.
A version of this article first appeared on Medscape.com.
This article was updated 6/7/21.
Better ways to handle in-hospital conflicts
Imagine a hospitalist, part of a group with 35 hospitalists, is in her second year of practice and is caring for a 55-year-old woman with a history of congestive heart failure and cirrhosis from hepatitis C due to heroin use. The patient was hospitalized with acute back pain and found to have vertebral osteomyelitis confirmed on MRI.
The hospitalist calls a surgeon to get a biopsy so that antibiotic therapy can be chosen. The surgeon says it’s the second time the patient has been hospitalized for this condition, and asks, “Why do you need me to see this patient?” He says the hospitalist should just give IV antibiotics and consult infectious disease.
The hospitalist says, “The patient needs this biopsy. I’ll just call your chair.”
In the course of a busy day, conflicts arise all the time in the hospital – between clinicians, between patients and clinicians, and as internal battles when clinicians face uncertain situations. There are ways to make these conflicts less tense and more in tune with patient care, panelists said recently during a session at SHM Converge, the annual conference of the Society of Hospital Medicine.
In the case of vertebral osteomyelitis, for instance, the hospitalist was using a “position-based” strategy to deal with the conflict with the surgeon – she came in knowing she wanted a biopsy – rather than an “interest-based” strategy, or what is in the patient’s interest, said Patrick Rendon, MD, FHM, assistant professor in the hospital medicine division at the University of New Mexico, Albuquerque.
“What we really need to do is realign the thinking from both the hospitalist as well as the consult perspective,” Dr. Rendon said. “It is not us versus the consultant or the consult versus us. It should be both, together, versus the problem.”
Instead of saying something like, “I need this biopsy,” it might be better to ask for an evaluation, he said.
Handling conflicts better can improve patient care but can also benefit the clinicians themselves. While hospitalists say they routinely experience “pushback” when making a request of a consultant, they also say that they prefer to receive instruction when consulting about a case. Dr. Rendon said that hospitalists also say they want this teaching done “in the right way,” and consultants routinely say that their instruction, when they give it, is often met with resistance.
“The idea here is to open up perspectives,” Dr. Rendon said.
Emily Gottenborg, MD, hospitalist and assistant professor of medicine at the University of Colorado, discussed the case of an intern caring for a patient who says something offensive.
Conflicts, she said, come in all sorts – intimidation, harassment, bias. And they can be based on race, gender, disability, and hierarchy, she said. When on the receiving end of offensive remarks from patients, it’s important for a clinician to set boundaries and quickly move on, with responses such as, “I care about you as a person, but I will not tolerate offensive behavior. Let’s focus on how I can help you today.”
“Practice that behavior so that you have a script in your mind and then use it when needed so that you can nip this behavior in the bud,” Dr. Gottenborg said.
In her hypothetical case, the intern asks for help from her program, and monthly morbidity and mortality workshops on bias and harassment are scheduled. She also receives counseling, and faculty and staff receive discrimination and bias training. Getting help from the institution can help systematically reduce these problems, Dr. Gottenborg said.
Ernie Esquivel, MD, SFHM, hospitalist and assistant professor of clinical medicine at Weill Cornell Medicine, New York, said internal conflicts test physicians routinely – and this has been especially true during the COVID-19 pandemic, in which urgent clinical situations arose with no clear answers.
“In the past year, physicians have experienced an incredible amount of anxiety and stress,” he said. “Tolerating uncertainty is probably one of the most mature skills that we need to learn as a physician.”
The culture of medicine, to a large degree, promotes the opposite tendency: value is placed on nailing down the diagnosis or achieving certainty. Confidence levels of physicians tend not to waver, even in the face of difficult cases full of uncertainty, Dr. Esquivel said.
He urged physicians to practice “deliberate clinical inertia” – to resist a quick response and to think more deeply and systematically about a situation. To show the importance of this, he asks residents to rank diagnoses, using sticky notes, as information about a case is provided. By the fourth round, when much more information is available, the diagnoses have changed dramatically.
Dr. Esquivel suggested physicians switch from thinking in terms of “diagnoses” to thinking in terms of “hypotheses.” That approach can help clinicians tolerate uncertainty, because it reinforces the idea that they are dealing with an “iterative process.”
“There may not be one diagnosis to consider,” he said, “but several in play at once.”
A version of this article first appeared on Medscape.com.
Imagine a hospitalist, part of a group with 35 hospitalists, is in her second year of practice and is caring for a 55-year-old woman with a history of congestive heart failure and cirrhosis from hepatitis C due to heroin use. The patient was hospitalized with acute back pain and found to have vertebral osteomyelitis confirmed on MRI.
The hospitalist calls a surgeon to get a biopsy so that antibiotic therapy can be chosen. The surgeon says it’s the second time the patient has been hospitalized for this condition, and asks, “Why do you need me to see this patient?” He says the hospitalist should just give IV antibiotics and consult infectious disease.
The hospitalist says, “The patient needs this biopsy. I’ll just call your chair.”
In the course of a busy day, conflicts arise all the time in the hospital – between clinicians, between patients and clinicians, and as internal battles when clinicians face uncertain situations. There are ways to make these conflicts less tense and more in tune with patient care, panelists said recently during a session at SHM Converge, the annual conference of the Society of Hospital Medicine.
In the case of vertebral osteomyelitis, for instance, the hospitalist was using a “position-based” strategy to deal with the conflict with the surgeon – she came in knowing she wanted a biopsy – rather than an “interest-based” strategy, or what is in the patient’s interest, said Patrick Rendon, MD, FHM, assistant professor in the hospital medicine division at the University of New Mexico, Albuquerque.
“What we really need to do is realign the thinking from both the hospitalist as well as the consult perspective,” Dr. Rendon said. “It is not us versus the consultant or the consult versus us. It should be both, together, versus the problem.”
Instead of saying something like, “I need this biopsy,” it might be better to ask for an evaluation, he said.
Handling conflicts better can improve patient care but can also benefit the clinicians themselves. While hospitalists say they routinely experience “pushback” when making a request of a consultant, they also say that they prefer to receive instruction when consulting about a case. Dr. Rendon said that hospitalists also say they want this teaching done “in the right way,” and consultants routinely say that their instruction, when they give it, is often met with resistance.
“The idea here is to open up perspectives,” Dr. Rendon said.
Emily Gottenborg, MD, hospitalist and assistant professor of medicine at the University of Colorado, discussed the case of an intern caring for a patient who says something offensive.
Conflicts, she said, come in all sorts – intimidation, harassment, bias. And they can be based on race, gender, disability, and hierarchy, she said. When on the receiving end of offensive remarks from patients, it’s important for a clinician to set boundaries and quickly move on, with responses such as, “I care about you as a person, but I will not tolerate offensive behavior. Let’s focus on how I can help you today.”
“Practice that behavior so that you have a script in your mind and then use it when needed so that you can nip this behavior in the bud,” Dr. Gottenborg said.
In her hypothetical case, the intern asks for help from her program, and monthly morbidity and mortality workshops on bias and harassment are scheduled. She also receives counseling, and faculty and staff receive discrimination and bias training. Getting help from the institution can help systematically reduce these problems, Dr. Gottenborg said.
Ernie Esquivel, MD, SFHM, hospitalist and assistant professor of clinical medicine at Weill Cornell Medicine, New York, said internal conflicts test physicians routinely – and this has been especially true during the COVID-19 pandemic, in which urgent clinical situations arose with no clear answers.
“In the past year, physicians have experienced an incredible amount of anxiety and stress,” he said. “Tolerating uncertainty is probably one of the most mature skills that we need to learn as a physician.”
The culture of medicine, to a large degree, promotes the opposite tendency: value is placed on nailing down the diagnosis or achieving certainty. Confidence levels of physicians tend not to waver, even in the face of difficult cases full of uncertainty, Dr. Esquivel said.
He urged physicians to practice “deliberate clinical inertia” – to resist a quick response and to think more deeply and systematically about a situation. To show the importance of this, he asks residents to rank diagnoses, using sticky notes, as information about a case is provided. By the fourth round, when much more information is available, the diagnoses have changed dramatically.
Dr. Esquivel suggested physicians switch from thinking in terms of “diagnoses” to thinking in terms of “hypotheses.” That approach can help clinicians tolerate uncertainty, because it reinforces the idea that they are dealing with an “iterative process.”
“There may not be one diagnosis to consider,” he said, “but several in play at once.”
A version of this article first appeared on Medscape.com.
Imagine a hospitalist, part of a group with 35 hospitalists, is in her second year of practice and is caring for a 55-year-old woman with a history of congestive heart failure and cirrhosis from hepatitis C due to heroin use. The patient was hospitalized with acute back pain and found to have vertebral osteomyelitis confirmed on MRI.
The hospitalist calls a surgeon to get a biopsy so that antibiotic therapy can be chosen. The surgeon says it’s the second time the patient has been hospitalized for this condition, and asks, “Why do you need me to see this patient?” He says the hospitalist should just give IV antibiotics and consult infectious disease.
The hospitalist says, “The patient needs this biopsy. I’ll just call your chair.”
In the course of a busy day, conflicts arise all the time in the hospital – between clinicians, between patients and clinicians, and as internal battles when clinicians face uncertain situations. There are ways to make these conflicts less tense and more in tune with patient care, panelists said recently during a session at SHM Converge, the annual conference of the Society of Hospital Medicine.
In the case of vertebral osteomyelitis, for instance, the hospitalist was using a “position-based” strategy to deal with the conflict with the surgeon – she came in knowing she wanted a biopsy – rather than an “interest-based” strategy, or what is in the patient’s interest, said Patrick Rendon, MD, FHM, assistant professor in the hospital medicine division at the University of New Mexico, Albuquerque.
“What we really need to do is realign the thinking from both the hospitalist as well as the consult perspective,” Dr. Rendon said. “It is not us versus the consultant or the consult versus us. It should be both, together, versus the problem.”
Instead of saying something like, “I need this biopsy,” it might be better to ask for an evaluation, he said.
Handling conflicts better can improve patient care but can also benefit the clinicians themselves. While hospitalists say they routinely experience “pushback” when making a request of a consultant, they also say that they prefer to receive instruction when consulting about a case. Dr. Rendon said that hospitalists also say they want this teaching done “in the right way,” and consultants routinely say that their instruction, when they give it, is often met with resistance.
“The idea here is to open up perspectives,” Dr. Rendon said.
Emily Gottenborg, MD, hospitalist and assistant professor of medicine at the University of Colorado, discussed the case of an intern caring for a patient who says something offensive.
Conflicts, she said, come in all sorts – intimidation, harassment, bias. And they can be based on race, gender, disability, and hierarchy, she said. When on the receiving end of offensive remarks from patients, it’s important for a clinician to set boundaries and quickly move on, with responses such as, “I care about you as a person, but I will not tolerate offensive behavior. Let’s focus on how I can help you today.”
“Practice that behavior so that you have a script in your mind and then use it when needed so that you can nip this behavior in the bud,” Dr. Gottenborg said.
In her hypothetical case, the intern asks for help from her program, and monthly morbidity and mortality workshops on bias and harassment are scheduled. She also receives counseling, and faculty and staff receive discrimination and bias training. Getting help from the institution can help systematically reduce these problems, Dr. Gottenborg said.
Ernie Esquivel, MD, SFHM, hospitalist and assistant professor of clinical medicine at Weill Cornell Medicine, New York, said internal conflicts test physicians routinely – and this has been especially true during the COVID-19 pandemic, in which urgent clinical situations arose with no clear answers.
“In the past year, physicians have experienced an incredible amount of anxiety and stress,” he said. “Tolerating uncertainty is probably one of the most mature skills that we need to learn as a physician.”
The culture of medicine, to a large degree, promotes the opposite tendency: value is placed on nailing down the diagnosis or achieving certainty. Confidence levels of physicians tend not to waver, even in the face of difficult cases full of uncertainty, Dr. Esquivel said.
He urged physicians to practice “deliberate clinical inertia” – to resist a quick response and to think more deeply and systematically about a situation. To show the importance of this, he asks residents to rank diagnoses, using sticky notes, as information about a case is provided. By the fourth round, when much more information is available, the diagnoses have changed dramatically.
Dr. Esquivel suggested physicians switch from thinking in terms of “diagnoses” to thinking in terms of “hypotheses.” That approach can help clinicians tolerate uncertainty, because it reinforces the idea that they are dealing with an “iterative process.”
“There may not be one diagnosis to consider,” he said, “but several in play at once.”
A version of this article first appeared on Medscape.com.
FROM SHM CONVERGE 2021
Adding daily steps linked to longer life
Taking more steps each day, in short spurts or longer bouts, was associated with a longer life in women older than 60 years, according to data from more than 16,000 participants in the ongoing Women’s Health Study.
The American Heart Association recommends at least 150 minutes per week of moderate physical activity, 75 minutes of vigorous physical activity, or a combination of both as fitness guidelines for adults. Walking is a safe and easy way for many adults to follow these guidelines, according to Christopher C. Moore, MS, a PhD candidate at the University of North Carolina at Chapel Hill.
The popularity of step counts reflect that they are simple and objective, and “focusing on steps can help promote an active lifestyle,” he said. Data on the impact of sporadic steps accumulated outside of longer bouts of activity on health outcomes are limited; however, technology advances in the form of fitness apps and wearable devices make it possible for researchers to track and measure the benefits of short periods of activity as well as longer periods.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting, sponsored by the AHA, Mr. Moore and colleagues assessed data from women older than 60 years who used wearable step-counting devices to measure their daily steps and walking patterns.
The study population included 16,732 women enrolled in the Women’s Health Study, a longstanding study of heart disease, cancer, and disease prevention among women in the United States. The participants wore waist step counters 4-7 days a week during 2011-2015. The average of the women was 72 years; 96% were non-Hispanic White, and the average BMI was 26 kg/m2.
The researchers divided the total number of steps for each study participant into two groups: “bouted” steps, defined as 10 minutes or longer bouts of walking with few interruptions; and “sporadic” steps, defined as short spurts of walking during regular daily activities such as housework, taking the stairs, or walking to or from a car.
A total of 804 deaths occurred during an average of 6 years of follow-up. Each initial increase of 1,000 steps including sporadic or bouted steps was associated with a 28% decrease in death, compared with no daily steps (hazard ratio, 0.72).
Each increasing quartile of sporadic steps was linked with higher total steps per day, Mr. Moore said. “Initial increase in sporadic steps corresponded to the greatest reductions in mortality,” with a HR of 0.69 per additional sporadic steps below 3,200 per day, and the impact on reduced mortality plateaued at about 4,500 sporadic steps per day.
In further analysis, the researchers also found a roughly 32% decrease in death in participants who took more than 2,000 steps daily in uninterrupted bouts (HR, 0.69).
The study findings were limited by several factors, including the relatively short follow-up period and number of events, the assessment of steps at a single time point, and the mostly homogeneous population, Mr. Moore noted. Additional research is needed to assess whether the results are generalizable to men, younger women, and diverse racial and ethnic groups.
However, the results may have implications for public health messaging, he emphasized. The message is that, to impact longevity, the total volume of steps is more important than the type of activity through which they are accumulated.
“You can accumulate your steps through longer bouts of purposeful activity or through everyday behaviors such as walking to your car, taking the stairs, and doing housework,” Mr. Moore concluded.
Find a friend, both of you benefit
On the basis of this study and other available evidence, more steps daily are recommended for everyone, Nieca Goldberg, MD, a cardiologist at New York University Langone Health, said in an interview.
“You can increase minutes of walking and frequency of walking,” she said.
Dr. Goldberg emphasized that you don’t need a fancy app or wearable device to up your steps. She offered some tips to help overcome barriers to putting one foot in front of the other. “Take the steps instead of the elevator. Park your car farther from your destination so you can walk.” Also, you can help yourself and help a friend to better health. “Get a walking buddy so you can encourage each other to walk,” Dr. Goldberg added.
Mr. Moore and Dr. Goldberg had no financial conflicts to disclose. The Women’s Health Study is funded by Brigham and Women’s Hospital; the National Heart, Lung, and Blood Institute; and the National Cancer Institute. Mr. Moore was funded by a grant from the NHLBI but had no other financial conflicts to disclose.
Taking more steps each day, in short spurts or longer bouts, was associated with a longer life in women older than 60 years, according to data from more than 16,000 participants in the ongoing Women’s Health Study.
The American Heart Association recommends at least 150 minutes per week of moderate physical activity, 75 minutes of vigorous physical activity, or a combination of both as fitness guidelines for adults. Walking is a safe and easy way for many adults to follow these guidelines, according to Christopher C. Moore, MS, a PhD candidate at the University of North Carolina at Chapel Hill.
The popularity of step counts reflect that they are simple and objective, and “focusing on steps can help promote an active lifestyle,” he said. Data on the impact of sporadic steps accumulated outside of longer bouts of activity on health outcomes are limited; however, technology advances in the form of fitness apps and wearable devices make it possible for researchers to track and measure the benefits of short periods of activity as well as longer periods.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting, sponsored by the AHA, Mr. Moore and colleagues assessed data from women older than 60 years who used wearable step-counting devices to measure their daily steps and walking patterns.
The study population included 16,732 women enrolled in the Women’s Health Study, a longstanding study of heart disease, cancer, and disease prevention among women in the United States. The participants wore waist step counters 4-7 days a week during 2011-2015. The average of the women was 72 years; 96% were non-Hispanic White, and the average BMI was 26 kg/m2.
The researchers divided the total number of steps for each study participant into two groups: “bouted” steps, defined as 10 minutes or longer bouts of walking with few interruptions; and “sporadic” steps, defined as short spurts of walking during regular daily activities such as housework, taking the stairs, or walking to or from a car.
A total of 804 deaths occurred during an average of 6 years of follow-up. Each initial increase of 1,000 steps including sporadic or bouted steps was associated with a 28% decrease in death, compared with no daily steps (hazard ratio, 0.72).
Each increasing quartile of sporadic steps was linked with higher total steps per day, Mr. Moore said. “Initial increase in sporadic steps corresponded to the greatest reductions in mortality,” with a HR of 0.69 per additional sporadic steps below 3,200 per day, and the impact on reduced mortality plateaued at about 4,500 sporadic steps per day.
In further analysis, the researchers also found a roughly 32% decrease in death in participants who took more than 2,000 steps daily in uninterrupted bouts (HR, 0.69).
The study findings were limited by several factors, including the relatively short follow-up period and number of events, the assessment of steps at a single time point, and the mostly homogeneous population, Mr. Moore noted. Additional research is needed to assess whether the results are generalizable to men, younger women, and diverse racial and ethnic groups.
However, the results may have implications for public health messaging, he emphasized. The message is that, to impact longevity, the total volume of steps is more important than the type of activity through which they are accumulated.
“You can accumulate your steps through longer bouts of purposeful activity or through everyday behaviors such as walking to your car, taking the stairs, and doing housework,” Mr. Moore concluded.
Find a friend, both of you benefit
On the basis of this study and other available evidence, more steps daily are recommended for everyone, Nieca Goldberg, MD, a cardiologist at New York University Langone Health, said in an interview.
“You can increase minutes of walking and frequency of walking,” she said.
Dr. Goldberg emphasized that you don’t need a fancy app or wearable device to up your steps. She offered some tips to help overcome barriers to putting one foot in front of the other. “Take the steps instead of the elevator. Park your car farther from your destination so you can walk.” Also, you can help yourself and help a friend to better health. “Get a walking buddy so you can encourage each other to walk,” Dr. Goldberg added.
Mr. Moore and Dr. Goldberg had no financial conflicts to disclose. The Women’s Health Study is funded by Brigham and Women’s Hospital; the National Heart, Lung, and Blood Institute; and the National Cancer Institute. Mr. Moore was funded by a grant from the NHLBI but had no other financial conflicts to disclose.
Taking more steps each day, in short spurts or longer bouts, was associated with a longer life in women older than 60 years, according to data from more than 16,000 participants in the ongoing Women’s Health Study.
The American Heart Association recommends at least 150 minutes per week of moderate physical activity, 75 minutes of vigorous physical activity, or a combination of both as fitness guidelines for adults. Walking is a safe and easy way for many adults to follow these guidelines, according to Christopher C. Moore, MS, a PhD candidate at the University of North Carolina at Chapel Hill.
The popularity of step counts reflect that they are simple and objective, and “focusing on steps can help promote an active lifestyle,” he said. Data on the impact of sporadic steps accumulated outside of longer bouts of activity on health outcomes are limited; however, technology advances in the form of fitness apps and wearable devices make it possible for researchers to track and measure the benefits of short periods of activity as well as longer periods.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting, sponsored by the AHA, Mr. Moore and colleagues assessed data from women older than 60 years who used wearable step-counting devices to measure their daily steps and walking patterns.
The study population included 16,732 women enrolled in the Women’s Health Study, a longstanding study of heart disease, cancer, and disease prevention among women in the United States. The participants wore waist step counters 4-7 days a week during 2011-2015. The average of the women was 72 years; 96% were non-Hispanic White, and the average BMI was 26 kg/m2.
The researchers divided the total number of steps for each study participant into two groups: “bouted” steps, defined as 10 minutes or longer bouts of walking with few interruptions; and “sporadic” steps, defined as short spurts of walking during regular daily activities such as housework, taking the stairs, or walking to or from a car.
A total of 804 deaths occurred during an average of 6 years of follow-up. Each initial increase of 1,000 steps including sporadic or bouted steps was associated with a 28% decrease in death, compared with no daily steps (hazard ratio, 0.72).
Each increasing quartile of sporadic steps was linked with higher total steps per day, Mr. Moore said. “Initial increase in sporadic steps corresponded to the greatest reductions in mortality,” with a HR of 0.69 per additional sporadic steps below 3,200 per day, and the impact on reduced mortality plateaued at about 4,500 sporadic steps per day.
In further analysis, the researchers also found a roughly 32% decrease in death in participants who took more than 2,000 steps daily in uninterrupted bouts (HR, 0.69).
The study findings were limited by several factors, including the relatively short follow-up period and number of events, the assessment of steps at a single time point, and the mostly homogeneous population, Mr. Moore noted. Additional research is needed to assess whether the results are generalizable to men, younger women, and diverse racial and ethnic groups.
However, the results may have implications for public health messaging, he emphasized. The message is that, to impact longevity, the total volume of steps is more important than the type of activity through which they are accumulated.
“You can accumulate your steps through longer bouts of purposeful activity or through everyday behaviors such as walking to your car, taking the stairs, and doing housework,” Mr. Moore concluded.
Find a friend, both of you benefit
On the basis of this study and other available evidence, more steps daily are recommended for everyone, Nieca Goldberg, MD, a cardiologist at New York University Langone Health, said in an interview.
“You can increase minutes of walking and frequency of walking,” she said.
Dr. Goldberg emphasized that you don’t need a fancy app or wearable device to up your steps. She offered some tips to help overcome barriers to putting one foot in front of the other. “Take the steps instead of the elevator. Park your car farther from your destination so you can walk.” Also, you can help yourself and help a friend to better health. “Get a walking buddy so you can encourage each other to walk,” Dr. Goldberg added.
Mr. Moore and Dr. Goldberg had no financial conflicts to disclose. The Women’s Health Study is funded by Brigham and Women’s Hospital; the National Heart, Lung, and Blood Institute; and the National Cancer Institute. Mr. Moore was funded by a grant from the NHLBI but had no other financial conflicts to disclose.
FROM EPI LIFESTYLE 2021
Hospitalists play key role in advance care planning
Advance care planning (ACP) is a process that supports adults at any age or stage of health in understanding and sharing their personal values, life goals, and preferences for future medical care, according to Meredith A. MacMartin, MD, director of inpatient palliative care at the Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
ACP “is really about planning for care in advance,” and in many ways, the inpatient setting is uniquely suited to this process, Dr. MacMartin said in a presentation at SHM Converge 2021, the annual conference of the Society of Hospital Medicine. “The key part is the advance part. You want conversations to happen before the care is actually needed,” she said.
Dr. MacMartin emphasized the importance of distinguishing between ACP and advance directives (ADs). ACP is a process, whereas ADs are documentation, “ideally of the content of advance care planning discussions,” she explained. ACP involves discussion about what is important to the patients, their goals, what information is helpful for them, and whether their current care is aligned with their goals, Dr. MacMartin said. ADs might involve a designated power of attorney for health care, a living will, and, in some states, specific clinician-signed orders regarding resuscitation or transport to hospital.
ACP is “more than whether a patient wants CPR [cardiopulmonary resuscitation] or not,” said Dr. MacMartin. ACP matters because it helps ensure that the care a patient receives aligns with the patient’s wishes and values, she said. ACP increases the likelihood that patients will die in their preferred locations, it allows them to discuss their wishes and prepare for decline, and it relieves family members of the burden of decision making, she said. From a hospital perspective, data show that use of an ACP can decrease intensive care unit (ICU) utilization and overall health care costs. “Often, when people are given the opportunity to express their wishes, they get less unnecessary care,” Dr. MacMartin noted.
Although ACP often takes place in an outpatient setting, hospitalists are in a unique position to conduct some ACP conversations with their patients, Dr. MacMartin said. “Hospitalists are available” and are physically present at least once a day, so there is a pragmatic advantage. Also, some data suggest that patients may feel more comfortable having ACP conversations with a hospitalist than with a primary care provider with whom they have a long-standing relationship, Dr. MacMartin added.
Another important advantage of ACP in the hospital setting is that, “as hospitalists, you are the expert on inpatient illness; you know what sick looks like, and you have a unique perspective on prognostication that may be harder to recreate in the outpatient setting,” Dr. MacMartin said.
Barriers to ACP include patient identification, logistics, attitudes
Settings in which ACP is appropriate include those in which a patient is undergoing “sentinel hospitalization,” meaning that the patient is at a transition point in the disease course. Examples are a patient newly diagnosed with metastatic solid cancer, a patient with progressive chronic kidney disease who is considering hemodialysis, or a patient who receives treatment in the ICU for longer than 7 days, Dr. MacMartin said.
Guidelines for identifying patients who might benefit from ACP include the use of the “surprise question” (“would you be surprised if this patient dies in the next year?”) as well as functional status assessments using tools such as the Australia-modified Karnofsky Performance Status or the Eastern Cooperative Oncology Group score, said Dr. MacMartin. Some studies suggest that any hospitalized patient older than 65 years should have an ACP discussion, she added.
Time pressure remains a significant barrier to ACP conversations. Some strategies to overcome this problem include enlisting help from other specialists, particularly social workers, Dr. MacMartin said. Social workers report a higher comfort level for talking to patients about death than any other medical specialty; “this is something they want to be doing,” she said. Also, the possibility of reimbursement may act as a buffer to create more time to have ACP conversations with patients, she noted.
Addressing clinicians’ discomfort with ACP conversations can be “a tougher nut to crack,” Dr. MacMartin acknowledged. Clinicians report that they don’t want to cause their patients distress, and some report that having conversations about end-of-life care is distressing for them as well. Some of these barriers can be overcome with skills training, including use of a prepared guideline or framework to help increase the comfort level for both clinicians and patients, said Dr. MacMartin.
A look ahead: Training strategies and COVID-19 impact
“For hospitalists interested in developing their ACP skills, I highly recommend two resources,” Dr. MacMartin said in an interview. “The Serious Illness Conversation Guide, from Ariadne Labs, is an excellent tool for any clinician to guide discussion about a patient’s goals and values,” she said.
“For clinicians wanting to build or improve their communication, including advance care planning discussions but also topics like responding to patient’s emotions, VitalTalk training offers a deeper dive into core communications skills,” she added.
“If your hospital has a palliative care team, they may also have more local resources available to you. To learn more about billing for ACP discussions, I recommend starting with your institutional billing and coding group, as these practices vary some between practices, and they will be able to provide the best guidance for clinicians. These are new codes that aren’t yet being very widely used so it’s a chance to innovate,” Dr. MacMartin noted.
“The hospital setting is an opportunity for patients to reflect on their health, both present and in the future, with a physician who has expertise in acute illness and prognostication and who is available for discussion on a daily basis during the hospitalization,” Dr. MacMartin emphasized.
As for whether the COVID-19 pandemic has affected ACP in the inpatient setting, the data are limited, but more information is forthcoming, Dr. MacMartin said. “In my personal experience and in talking to colleagues elsewhere, the pandemic has highlighted the need for ACP in some ways, as we have tried to ensure that people who wouldn’t want things like intensive care are identified early,” she said. “I hope that some of the workflows developed to identify patients who should get ACP in the hospital stay in practice and are strengthened over time,” she added.
Dr. MacMartin has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Advance care planning (ACP) is a process that supports adults at any age or stage of health in understanding and sharing their personal values, life goals, and preferences for future medical care, according to Meredith A. MacMartin, MD, director of inpatient palliative care at the Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
ACP “is really about planning for care in advance,” and in many ways, the inpatient setting is uniquely suited to this process, Dr. MacMartin said in a presentation at SHM Converge 2021, the annual conference of the Society of Hospital Medicine. “The key part is the advance part. You want conversations to happen before the care is actually needed,” she said.
Dr. MacMartin emphasized the importance of distinguishing between ACP and advance directives (ADs). ACP is a process, whereas ADs are documentation, “ideally of the content of advance care planning discussions,” she explained. ACP involves discussion about what is important to the patients, their goals, what information is helpful for them, and whether their current care is aligned with their goals, Dr. MacMartin said. ADs might involve a designated power of attorney for health care, a living will, and, in some states, specific clinician-signed orders regarding resuscitation or transport to hospital.
ACP is “more than whether a patient wants CPR [cardiopulmonary resuscitation] or not,” said Dr. MacMartin. ACP matters because it helps ensure that the care a patient receives aligns with the patient’s wishes and values, she said. ACP increases the likelihood that patients will die in their preferred locations, it allows them to discuss their wishes and prepare for decline, and it relieves family members of the burden of decision making, she said. From a hospital perspective, data show that use of an ACP can decrease intensive care unit (ICU) utilization and overall health care costs. “Often, when people are given the opportunity to express their wishes, they get less unnecessary care,” Dr. MacMartin noted.
Although ACP often takes place in an outpatient setting, hospitalists are in a unique position to conduct some ACP conversations with their patients, Dr. MacMartin said. “Hospitalists are available” and are physically present at least once a day, so there is a pragmatic advantage. Also, some data suggest that patients may feel more comfortable having ACP conversations with a hospitalist than with a primary care provider with whom they have a long-standing relationship, Dr. MacMartin added.
Another important advantage of ACP in the hospital setting is that, “as hospitalists, you are the expert on inpatient illness; you know what sick looks like, and you have a unique perspective on prognostication that may be harder to recreate in the outpatient setting,” Dr. MacMartin said.
Barriers to ACP include patient identification, logistics, attitudes
Settings in which ACP is appropriate include those in which a patient is undergoing “sentinel hospitalization,” meaning that the patient is at a transition point in the disease course. Examples are a patient newly diagnosed with metastatic solid cancer, a patient with progressive chronic kidney disease who is considering hemodialysis, or a patient who receives treatment in the ICU for longer than 7 days, Dr. MacMartin said.
Guidelines for identifying patients who might benefit from ACP include the use of the “surprise question” (“would you be surprised if this patient dies in the next year?”) as well as functional status assessments using tools such as the Australia-modified Karnofsky Performance Status or the Eastern Cooperative Oncology Group score, said Dr. MacMartin. Some studies suggest that any hospitalized patient older than 65 years should have an ACP discussion, she added.
Time pressure remains a significant barrier to ACP conversations. Some strategies to overcome this problem include enlisting help from other specialists, particularly social workers, Dr. MacMartin said. Social workers report a higher comfort level for talking to patients about death than any other medical specialty; “this is something they want to be doing,” she said. Also, the possibility of reimbursement may act as a buffer to create more time to have ACP conversations with patients, she noted.
Addressing clinicians’ discomfort with ACP conversations can be “a tougher nut to crack,” Dr. MacMartin acknowledged. Clinicians report that they don’t want to cause their patients distress, and some report that having conversations about end-of-life care is distressing for them as well. Some of these barriers can be overcome with skills training, including use of a prepared guideline or framework to help increase the comfort level for both clinicians and patients, said Dr. MacMartin.
A look ahead: Training strategies and COVID-19 impact
“For hospitalists interested in developing their ACP skills, I highly recommend two resources,” Dr. MacMartin said in an interview. “The Serious Illness Conversation Guide, from Ariadne Labs, is an excellent tool for any clinician to guide discussion about a patient’s goals and values,” she said.
“For clinicians wanting to build or improve their communication, including advance care planning discussions but also topics like responding to patient’s emotions, VitalTalk training offers a deeper dive into core communications skills,” she added.
“If your hospital has a palliative care team, they may also have more local resources available to you. To learn more about billing for ACP discussions, I recommend starting with your institutional billing and coding group, as these practices vary some between practices, and they will be able to provide the best guidance for clinicians. These are new codes that aren’t yet being very widely used so it’s a chance to innovate,” Dr. MacMartin noted.
“The hospital setting is an opportunity for patients to reflect on their health, both present and in the future, with a physician who has expertise in acute illness and prognostication and who is available for discussion on a daily basis during the hospitalization,” Dr. MacMartin emphasized.
As for whether the COVID-19 pandemic has affected ACP in the inpatient setting, the data are limited, but more information is forthcoming, Dr. MacMartin said. “In my personal experience and in talking to colleagues elsewhere, the pandemic has highlighted the need for ACP in some ways, as we have tried to ensure that people who wouldn’t want things like intensive care are identified early,” she said. “I hope that some of the workflows developed to identify patients who should get ACP in the hospital stay in practice and are strengthened over time,” she added.
Dr. MacMartin has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Advance care planning (ACP) is a process that supports adults at any age or stage of health in understanding and sharing their personal values, life goals, and preferences for future medical care, according to Meredith A. MacMartin, MD, director of inpatient palliative care at the Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
ACP “is really about planning for care in advance,” and in many ways, the inpatient setting is uniquely suited to this process, Dr. MacMartin said in a presentation at SHM Converge 2021, the annual conference of the Society of Hospital Medicine. “The key part is the advance part. You want conversations to happen before the care is actually needed,” she said.
Dr. MacMartin emphasized the importance of distinguishing between ACP and advance directives (ADs). ACP is a process, whereas ADs are documentation, “ideally of the content of advance care planning discussions,” she explained. ACP involves discussion about what is important to the patients, their goals, what information is helpful for them, and whether their current care is aligned with their goals, Dr. MacMartin said. ADs might involve a designated power of attorney for health care, a living will, and, in some states, specific clinician-signed orders regarding resuscitation or transport to hospital.
ACP is “more than whether a patient wants CPR [cardiopulmonary resuscitation] or not,” said Dr. MacMartin. ACP matters because it helps ensure that the care a patient receives aligns with the patient’s wishes and values, she said. ACP increases the likelihood that patients will die in their preferred locations, it allows them to discuss their wishes and prepare for decline, and it relieves family members of the burden of decision making, she said. From a hospital perspective, data show that use of an ACP can decrease intensive care unit (ICU) utilization and overall health care costs. “Often, when people are given the opportunity to express their wishes, they get less unnecessary care,” Dr. MacMartin noted.
Although ACP often takes place in an outpatient setting, hospitalists are in a unique position to conduct some ACP conversations with their patients, Dr. MacMartin said. “Hospitalists are available” and are physically present at least once a day, so there is a pragmatic advantage. Also, some data suggest that patients may feel more comfortable having ACP conversations with a hospitalist than with a primary care provider with whom they have a long-standing relationship, Dr. MacMartin added.
Another important advantage of ACP in the hospital setting is that, “as hospitalists, you are the expert on inpatient illness; you know what sick looks like, and you have a unique perspective on prognostication that may be harder to recreate in the outpatient setting,” Dr. MacMartin said.
Barriers to ACP include patient identification, logistics, attitudes
Settings in which ACP is appropriate include those in which a patient is undergoing “sentinel hospitalization,” meaning that the patient is at a transition point in the disease course. Examples are a patient newly diagnosed with metastatic solid cancer, a patient with progressive chronic kidney disease who is considering hemodialysis, or a patient who receives treatment in the ICU for longer than 7 days, Dr. MacMartin said.
Guidelines for identifying patients who might benefit from ACP include the use of the “surprise question” (“would you be surprised if this patient dies in the next year?”) as well as functional status assessments using tools such as the Australia-modified Karnofsky Performance Status or the Eastern Cooperative Oncology Group score, said Dr. MacMartin. Some studies suggest that any hospitalized patient older than 65 years should have an ACP discussion, she added.
Time pressure remains a significant barrier to ACP conversations. Some strategies to overcome this problem include enlisting help from other specialists, particularly social workers, Dr. MacMartin said. Social workers report a higher comfort level for talking to patients about death than any other medical specialty; “this is something they want to be doing,” she said. Also, the possibility of reimbursement may act as a buffer to create more time to have ACP conversations with patients, she noted.
Addressing clinicians’ discomfort with ACP conversations can be “a tougher nut to crack,” Dr. MacMartin acknowledged. Clinicians report that they don’t want to cause their patients distress, and some report that having conversations about end-of-life care is distressing for them as well. Some of these barriers can be overcome with skills training, including use of a prepared guideline or framework to help increase the comfort level for both clinicians and patients, said Dr. MacMartin.
A look ahead: Training strategies and COVID-19 impact
“For hospitalists interested in developing their ACP skills, I highly recommend two resources,” Dr. MacMartin said in an interview. “The Serious Illness Conversation Guide, from Ariadne Labs, is an excellent tool for any clinician to guide discussion about a patient’s goals and values,” she said.
“For clinicians wanting to build or improve their communication, including advance care planning discussions but also topics like responding to patient’s emotions, VitalTalk training offers a deeper dive into core communications skills,” she added.
“If your hospital has a palliative care team, they may also have more local resources available to you. To learn more about billing for ACP discussions, I recommend starting with your institutional billing and coding group, as these practices vary some between practices, and they will be able to provide the best guidance for clinicians. These are new codes that aren’t yet being very widely used so it’s a chance to innovate,” Dr. MacMartin noted.
“The hospital setting is an opportunity for patients to reflect on their health, both present and in the future, with a physician who has expertise in acute illness and prognostication and who is available for discussion on a daily basis during the hospitalization,” Dr. MacMartin emphasized.
As for whether the COVID-19 pandemic has affected ACP in the inpatient setting, the data are limited, but more information is forthcoming, Dr. MacMartin said. “In my personal experience and in talking to colleagues elsewhere, the pandemic has highlighted the need for ACP in some ways, as we have tried to ensure that people who wouldn’t want things like intensive care are identified early,” she said. “I hope that some of the workflows developed to identify patients who should get ACP in the hospital stay in practice and are strengthened over time,” she added.
Dr. MacMartin has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM SHM CONVERGE 2021
CDC: New botulism guidelines focus on mass casualty events
Botulinum toxin is said to be the most lethal substance known. Inhaling just 1-3 nanograms of toxin per kilogram of body mass constitutes a lethal dose.
The CDC has been working on these guidelines since 2015, initially establishing a technical development group and steering committee to prioritize topics for review and make recommendations. Since then, the agency published 15 systematic reviews in Clinical Infectious Diseases early in 2018. The reviews addressed the recognition of botulism clinically, treatment with botulinum antitoxin, and complications from that treatment. They also looked at the epidemiology of botulism outbreaks and botulism in the special populations of vulnerable pediatric and pregnant patients.
In 2016, the CDC held two extended forums and convened a workshop with 72 experts. In addition to the more standard topics of diagnosis and treatment, attention was given to crisis standards of care, caring for multiple patients at once, and ethical considerations in management.
Amesh Adalja, MD, senior scholar, Johns Hopkins Center for Health Security, Baltimore, said in an interview that the new guidance “was really specific [and] was meant to address the gap in guidance for mass casualty settings.”
While clinicians are used to focusing on an individual patient, in times of crises, with multiple patients from a food-borne outbreak or a bioterrorism attack, the focus must shift to the population rather than the individual. The workshop explored issues of triaging, adding beds, and caring for patients when a hospital is overwhelmed with an acute influx of severely ill patients.
Such a mass casualty event is similar to the stress encountered this past year with COVID-19 patients swamping the hospitals, which had too little oxygen, too few ventilators, and too few staff members to care for the sudden influx of critically ill patients.
Diagnosis
Leslie Edwards, MHS, BSN, a CDC epidemiologist and botulism expert, said that “botulism is rare and [so] could be difficult to diagnose.” The CDC “wanted to highlight some of those key clinical factors” to speed recognition.
Hospitals and health officials are being urged to develop crisis protocols as part of emergency preparedness plans. And clinicians should be able to recognize four major syndromes: botulism from food, wounds, and inhalation, as well as iatrogenic botulism (from exposure via injection of the neurotoxin).
Botulism has a characteristic and unusual pattern of symptoms, which begin with cranial nerve palsies. Then there is typically a descending, symmetric flaccid paralysis. Symptoms might progress to respiratory failure and death. Other critical clues that implicate botulism include a lack of sensory deficits and the absence of pain.
Symptoms are most likely to be mistaken for myasthenia gravis or Guillain-Barré syndrome, but the latter has an ascending paralysis. Cranial nerve involvement can present as blurred vision, ptosis (drooping lid), diplopia (double vision), ophthalmoplegia (weak eye muscles), or difficulty with speech and swallowing. Shortness of breath and abdominal discomfort can also occur. Respiratory failure may occur from weakness or paralysis of cranial nerves. Cranial nerve signs and symptoms in the absence of fever, along with a descending paralysis, should strongly suggest the diagnosis.
With food-borne botulism, vomiting occurs in half the patients. Improperly sterilized home-canned food is the major risk factor. While the toxin is rapidly destroyed by heat, the bacterial spores are not. Wound botulism is most commonly associated with the injection of drugs, particularly black tar heroin.
Dr. Edwards stressed that “time is of the essence when it comes to botulism diagnostics and treating. Timely administration of the botulism antitoxin early in the course of illness can arrest the progression of paralysis and possibly avert the need for intubation or ventilation.”
It’s essential to note that botulism is an urgent diagnosis that has to be made on clinical grounds. Lab assays for botulinum neurotoxins take too long and are only conducted in public health laboratories. The decision to use antitoxin must not be delayed to wait for confirmation.
Clinicians should immediately contact the local or state health department’s emergency on-call team if botulism is suspected. They will arrange for expert consultation.
Treatment
Botulinum antitoxin is the only specific therapy for this infection. If given early – preferably within 24-48 hours of symptom onset – it can stop the progression of paralysis. But antitoxin will not reverse existing paralysis. If paralysis is still progressing outside of that 24- to 48-hour window, the antitoxin should still provide benefit. The antitoxin is available only through state health departments and a request to the CDC.
Botulism antitoxin is made from horse serum and therefore may cause a variety of allergic reactions. The risk for anaphylaxis is less than 2%, far lower than the mortality from untreated botulism.
While these guidelines have an important focus on triaging and treating mass casualties from botulism, it’s important to note that food-borne outbreaks and prevention issues are covered elsewhere on the CDC site.
Dr. Edwards has disclosed no relevant financial relationships. Dr. Adalja is a consultant for Emergent BioSolutions, which makes the heptavalent botulism antitoxin.
Dr. Stone is an infectious disease specialist and author of “Resilience: One Family’s Story of Hope and Triumph Over Evil” and of “Conducting Clinical Research,” the essential guide to the topic. You can find her at drjudystone.com or on Twitter @drjudystone.
A version of this article first appeared on Medscape.com.
Botulinum toxin is said to be the most lethal substance known. Inhaling just 1-3 nanograms of toxin per kilogram of body mass constitutes a lethal dose.
The CDC has been working on these guidelines since 2015, initially establishing a technical development group and steering committee to prioritize topics for review and make recommendations. Since then, the agency published 15 systematic reviews in Clinical Infectious Diseases early in 2018. The reviews addressed the recognition of botulism clinically, treatment with botulinum antitoxin, and complications from that treatment. They also looked at the epidemiology of botulism outbreaks and botulism in the special populations of vulnerable pediatric and pregnant patients.
In 2016, the CDC held two extended forums and convened a workshop with 72 experts. In addition to the more standard topics of diagnosis and treatment, attention was given to crisis standards of care, caring for multiple patients at once, and ethical considerations in management.
Amesh Adalja, MD, senior scholar, Johns Hopkins Center for Health Security, Baltimore, said in an interview that the new guidance “was really specific [and] was meant to address the gap in guidance for mass casualty settings.”
While clinicians are used to focusing on an individual patient, in times of crises, with multiple patients from a food-borne outbreak or a bioterrorism attack, the focus must shift to the population rather than the individual. The workshop explored issues of triaging, adding beds, and caring for patients when a hospital is overwhelmed with an acute influx of severely ill patients.
Such a mass casualty event is similar to the stress encountered this past year with COVID-19 patients swamping the hospitals, which had too little oxygen, too few ventilators, and too few staff members to care for the sudden influx of critically ill patients.
Diagnosis
Leslie Edwards, MHS, BSN, a CDC epidemiologist and botulism expert, said that “botulism is rare and [so] could be difficult to diagnose.” The CDC “wanted to highlight some of those key clinical factors” to speed recognition.
Hospitals and health officials are being urged to develop crisis protocols as part of emergency preparedness plans. And clinicians should be able to recognize four major syndromes: botulism from food, wounds, and inhalation, as well as iatrogenic botulism (from exposure via injection of the neurotoxin).
Botulism has a characteristic and unusual pattern of symptoms, which begin with cranial nerve palsies. Then there is typically a descending, symmetric flaccid paralysis. Symptoms might progress to respiratory failure and death. Other critical clues that implicate botulism include a lack of sensory deficits and the absence of pain.
Symptoms are most likely to be mistaken for myasthenia gravis or Guillain-Barré syndrome, but the latter has an ascending paralysis. Cranial nerve involvement can present as blurred vision, ptosis (drooping lid), diplopia (double vision), ophthalmoplegia (weak eye muscles), or difficulty with speech and swallowing. Shortness of breath and abdominal discomfort can also occur. Respiratory failure may occur from weakness or paralysis of cranial nerves. Cranial nerve signs and symptoms in the absence of fever, along with a descending paralysis, should strongly suggest the diagnosis.
With food-borne botulism, vomiting occurs in half the patients. Improperly sterilized home-canned food is the major risk factor. While the toxin is rapidly destroyed by heat, the bacterial spores are not. Wound botulism is most commonly associated with the injection of drugs, particularly black tar heroin.
Dr. Edwards stressed that “time is of the essence when it comes to botulism diagnostics and treating. Timely administration of the botulism antitoxin early in the course of illness can arrest the progression of paralysis and possibly avert the need for intubation or ventilation.”
It’s essential to note that botulism is an urgent diagnosis that has to be made on clinical grounds. Lab assays for botulinum neurotoxins take too long and are only conducted in public health laboratories. The decision to use antitoxin must not be delayed to wait for confirmation.
Clinicians should immediately contact the local or state health department’s emergency on-call team if botulism is suspected. They will arrange for expert consultation.
Treatment
Botulinum antitoxin is the only specific therapy for this infection. If given early – preferably within 24-48 hours of symptom onset – it can stop the progression of paralysis. But antitoxin will not reverse existing paralysis. If paralysis is still progressing outside of that 24- to 48-hour window, the antitoxin should still provide benefit. The antitoxin is available only through state health departments and a request to the CDC.
Botulism antitoxin is made from horse serum and therefore may cause a variety of allergic reactions. The risk for anaphylaxis is less than 2%, far lower than the mortality from untreated botulism.
While these guidelines have an important focus on triaging and treating mass casualties from botulism, it’s important to note that food-borne outbreaks and prevention issues are covered elsewhere on the CDC site.
Dr. Edwards has disclosed no relevant financial relationships. Dr. Adalja is a consultant for Emergent BioSolutions, which makes the heptavalent botulism antitoxin.
Dr. Stone is an infectious disease specialist and author of “Resilience: One Family’s Story of Hope and Triumph Over Evil” and of “Conducting Clinical Research,” the essential guide to the topic. You can find her at drjudystone.com or on Twitter @drjudystone.
A version of this article first appeared on Medscape.com.
Botulinum toxin is said to be the most lethal substance known. Inhaling just 1-3 nanograms of toxin per kilogram of body mass constitutes a lethal dose.
The CDC has been working on these guidelines since 2015, initially establishing a technical development group and steering committee to prioritize topics for review and make recommendations. Since then, the agency published 15 systematic reviews in Clinical Infectious Diseases early in 2018. The reviews addressed the recognition of botulism clinically, treatment with botulinum antitoxin, and complications from that treatment. They also looked at the epidemiology of botulism outbreaks and botulism in the special populations of vulnerable pediatric and pregnant patients.
In 2016, the CDC held two extended forums and convened a workshop with 72 experts. In addition to the more standard topics of diagnosis and treatment, attention was given to crisis standards of care, caring for multiple patients at once, and ethical considerations in management.
Amesh Adalja, MD, senior scholar, Johns Hopkins Center for Health Security, Baltimore, said in an interview that the new guidance “was really specific [and] was meant to address the gap in guidance for mass casualty settings.”
While clinicians are used to focusing on an individual patient, in times of crises, with multiple patients from a food-borne outbreak or a bioterrorism attack, the focus must shift to the population rather than the individual. The workshop explored issues of triaging, adding beds, and caring for patients when a hospital is overwhelmed with an acute influx of severely ill patients.
Such a mass casualty event is similar to the stress encountered this past year with COVID-19 patients swamping the hospitals, which had too little oxygen, too few ventilators, and too few staff members to care for the sudden influx of critically ill patients.
Diagnosis
Leslie Edwards, MHS, BSN, a CDC epidemiologist and botulism expert, said that “botulism is rare and [so] could be difficult to diagnose.” The CDC “wanted to highlight some of those key clinical factors” to speed recognition.
Hospitals and health officials are being urged to develop crisis protocols as part of emergency preparedness plans. And clinicians should be able to recognize four major syndromes: botulism from food, wounds, and inhalation, as well as iatrogenic botulism (from exposure via injection of the neurotoxin).
Botulism has a characteristic and unusual pattern of symptoms, which begin with cranial nerve palsies. Then there is typically a descending, symmetric flaccid paralysis. Symptoms might progress to respiratory failure and death. Other critical clues that implicate botulism include a lack of sensory deficits and the absence of pain.
Symptoms are most likely to be mistaken for myasthenia gravis or Guillain-Barré syndrome, but the latter has an ascending paralysis. Cranial nerve involvement can present as blurred vision, ptosis (drooping lid), diplopia (double vision), ophthalmoplegia (weak eye muscles), or difficulty with speech and swallowing. Shortness of breath and abdominal discomfort can also occur. Respiratory failure may occur from weakness or paralysis of cranial nerves. Cranial nerve signs and symptoms in the absence of fever, along with a descending paralysis, should strongly suggest the diagnosis.
With food-borne botulism, vomiting occurs in half the patients. Improperly sterilized home-canned food is the major risk factor. While the toxin is rapidly destroyed by heat, the bacterial spores are not. Wound botulism is most commonly associated with the injection of drugs, particularly black tar heroin.
Dr. Edwards stressed that “time is of the essence when it comes to botulism diagnostics and treating. Timely administration of the botulism antitoxin early in the course of illness can arrest the progression of paralysis and possibly avert the need for intubation or ventilation.”
It’s essential to note that botulism is an urgent diagnosis that has to be made on clinical grounds. Lab assays for botulinum neurotoxins take too long and are only conducted in public health laboratories. The decision to use antitoxin must not be delayed to wait for confirmation.
Clinicians should immediately contact the local or state health department’s emergency on-call team if botulism is suspected. They will arrange for expert consultation.
Treatment
Botulinum antitoxin is the only specific therapy for this infection. If given early – preferably within 24-48 hours of symptom onset – it can stop the progression of paralysis. But antitoxin will not reverse existing paralysis. If paralysis is still progressing outside of that 24- to 48-hour window, the antitoxin should still provide benefit. The antitoxin is available only through state health departments and a request to the CDC.
Botulism antitoxin is made from horse serum and therefore may cause a variety of allergic reactions. The risk for anaphylaxis is less than 2%, far lower than the mortality from untreated botulism.
While these guidelines have an important focus on triaging and treating mass casualties from botulism, it’s important to note that food-borne outbreaks and prevention issues are covered elsewhere on the CDC site.
Dr. Edwards has disclosed no relevant financial relationships. Dr. Adalja is a consultant for Emergent BioSolutions, which makes the heptavalent botulism antitoxin.
Dr. Stone is an infectious disease specialist and author of “Resilience: One Family’s Story of Hope and Triumph Over Evil” and of “Conducting Clinical Research,” the essential guide to the topic. You can find her at drjudystone.com or on Twitter @drjudystone.
A version of this article first appeared on Medscape.com.
‘Smart toilet’ with AI automatically scans stool for blood and consistency
A “smart toilet” in development uses artificial intelligence (AI) to scan stool for consistency and presence of blood – and early evidence suggests it is more accurate than patient self-reporting, a study reveals.
The remote, automated, real-time analysis and reporting increase the likelihood of physicians detecting gastrointestinal issues earlier, investigators reported.
In a proof-of-concept study, the smart toilet was 85% accurate in categorizing stool consistency as loose, normal, or constipated. The findings were presented at the annual Digestive Disease Week® (DDW).
“This study highlights a very innovative and practical tool that could have major implications for patients and clinicians alike,” Andrea Shin, MD, who was not affiliated with the research, said in an interview.
“Stool form or consistency and signs of bleeding are some of the most important pieces of clinical history when it comes to GI or bowel symptoms,” added Dr. Shin, assistant professor of medicine in the department of gastroenterology and hepatology at Indiana University, Indianapolis.
Image analysis
The researchers tested their AI algorithm on 3,328 images. They assessed photos from the Internet and some submitted anonymously by participants in the study.
Two gastroenterologists also rated a subset of 552 images. The physicians showed “satisfactory agreement” on interrater reliability (the extent to which two or more “raters” [for example, observers, examiners] agree), the investigators noted.
The smart toilet also was 76% accurate for gross blood detection.
“It’s objective and more accurate,” study author Sonia Grego, PhD, said in an interview. In contrast to asking patients to keep a bowel movement diary or recall the frequency and consistency of their stool over time, “the system does it for you,” she added.
“Our technology – by automating the image acquisition – removes the burden of having to track your pattern for weeks or months,” added Dr. Grego, founding director of the Duke Smart Toilet Lab at Duke University in Durham, N.C.
Information provided by patients “can have a big impact on decision-making,” Dr. Shin said. “For example, if I am talking to an individual who suffers from irritable bowel syndrome [IBS], I commonly ask them about how loose or watery and hard or formed their stool is, because this information gives me clues as to the underlying problems that may be driving their symptoms.”
Dr. Shin agreed it can be challenging for people to know what is important to report to their doctor. “This tool has the potential to relieve patient burden and facilitate communication between a patient and their clinician. It’s a great example of how technology can be leveraged to enhance care.”
Working behind the scenes
Together with gastroenterologist Deborah Anne Fisher, MD, an associate professor of medicine at Duke, Dr. Grego and colleagues devised a prototype that positions the image analyzer in the pipes behind the toilet. So the analysis is done post flush.
“We are experts of toilets and toilet technology,” Dr. Grego said. “We have learned that people really don’t like to see anything weird around the toilet bowl.”
The smart toilet system is designed for multiple users in a residential or commercial setting. The technology could be used in hospitals or long-term care facilities, for example. A fingerprint scanner on the flush mechanism tracks each individual user.
Mixed reactions
Dr. Grego gets a range of reactions when she tells people she is developing smart toilet technology.
“Friends and family laugh about the concept of the smart toilet,” she said, “so all the possible jokes that have been done on poops, we know.”
In fact, the researchers also are collecting the jokes they hear. “We’re being very systematic.”
In contrast, gastroenterologists who learn of the technology in development are more enthusiastic, Dr. Grego said. “There is such a need for removing the uncertainty of the patient recall about bowel movement frequency and appearance.
“We are seeking to expand through collaboration with additional GI doctors. We want to develop a more advanced prototype and do further validation studies,” Dr. Grego said.
Digital health tool
There is an aversion among patients to handling stool “or even talking about it,” Dr. Grego said. Colleagues tell her that people are more willing to provide a blood sample, which requires a needle, than a stool sample.
“But a lot of health data is there [in the stool],” she added. “We think this will empower a lot of research as well as consumer data gathering.”
For example, Dr. Grego envisions pharmaceutical companies using the technology to detect or monitor any changes in stool or gut health based on a treatment in development during clinical trials.
Furthermore, the technology might empower health-conscious consumers who want to track their own gut health. “This technology will be a whole new entry in the digital health toolkit,” Dr. Grego said.
Although not included in the research presented at this year’s DDW, the developers plan to add a sampling capability. Biochemic analysis of stool samples could provide “metabolically relevant information,” including stool biomarkers and microbiome composition.
“We have demonstrated it in the laboratory. It will be part of the technology when developed into a product,” Dr. Grego said.
This proof-of-concept study “is the first step in a path we are aggressively pursuing,” Dr. Grego said. She estimated it will take about 12-18 months to develop a prototype for use with patients. “We hope to move to a product soon after that.”
“I’m looking forward to seeing future iterations of this tool,” Dr. Shin said. “It could have a role in monitoring important GI diseases and disorders, including IBS and inflammatory bowel disease, or even for the detection of ‘alarm symptoms’ that shouldn’t be ignored.
“I could even see it having a role in preventative health in the future,” Dr. Shin added.
The technology has been licensed to the spin-off company Coprata to develop the product further.
“We hope to have an impact on people’s health very soon,” Dr. Grego said.
Duke University funded the study. Dr. Grego holds a management position at Coprata. Dr. Shin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A “smart toilet” in development uses artificial intelligence (AI) to scan stool for consistency and presence of blood – and early evidence suggests it is more accurate than patient self-reporting, a study reveals.
The remote, automated, real-time analysis and reporting increase the likelihood of physicians detecting gastrointestinal issues earlier, investigators reported.
In a proof-of-concept study, the smart toilet was 85% accurate in categorizing stool consistency as loose, normal, or constipated. The findings were presented at the annual Digestive Disease Week® (DDW).
“This study highlights a very innovative and practical tool that could have major implications for patients and clinicians alike,” Andrea Shin, MD, who was not affiliated with the research, said in an interview.
“Stool form or consistency and signs of bleeding are some of the most important pieces of clinical history when it comes to GI or bowel symptoms,” added Dr. Shin, assistant professor of medicine in the department of gastroenterology and hepatology at Indiana University, Indianapolis.
Image analysis
The researchers tested their AI algorithm on 3,328 images. They assessed photos from the Internet and some submitted anonymously by participants in the study.
Two gastroenterologists also rated a subset of 552 images. The physicians showed “satisfactory agreement” on interrater reliability (the extent to which two or more “raters” [for example, observers, examiners] agree), the investigators noted.
The smart toilet also was 76% accurate for gross blood detection.
“It’s objective and more accurate,” study author Sonia Grego, PhD, said in an interview. In contrast to asking patients to keep a bowel movement diary or recall the frequency and consistency of their stool over time, “the system does it for you,” she added.
“Our technology – by automating the image acquisition – removes the burden of having to track your pattern for weeks or months,” added Dr. Grego, founding director of the Duke Smart Toilet Lab at Duke University in Durham, N.C.
Information provided by patients “can have a big impact on decision-making,” Dr. Shin said. “For example, if I am talking to an individual who suffers from irritable bowel syndrome [IBS], I commonly ask them about how loose or watery and hard or formed their stool is, because this information gives me clues as to the underlying problems that may be driving their symptoms.”
Dr. Shin agreed it can be challenging for people to know what is important to report to their doctor. “This tool has the potential to relieve patient burden and facilitate communication between a patient and their clinician. It’s a great example of how technology can be leveraged to enhance care.”
Working behind the scenes
Together with gastroenterologist Deborah Anne Fisher, MD, an associate professor of medicine at Duke, Dr. Grego and colleagues devised a prototype that positions the image analyzer in the pipes behind the toilet. So the analysis is done post flush.
“We are experts of toilets and toilet technology,” Dr. Grego said. “We have learned that people really don’t like to see anything weird around the toilet bowl.”
The smart toilet system is designed for multiple users in a residential or commercial setting. The technology could be used in hospitals or long-term care facilities, for example. A fingerprint scanner on the flush mechanism tracks each individual user.
Mixed reactions
Dr. Grego gets a range of reactions when she tells people she is developing smart toilet technology.
“Friends and family laugh about the concept of the smart toilet,” she said, “so all the possible jokes that have been done on poops, we know.”
In fact, the researchers also are collecting the jokes they hear. “We’re being very systematic.”
In contrast, gastroenterologists who learn of the technology in development are more enthusiastic, Dr. Grego said. “There is such a need for removing the uncertainty of the patient recall about bowel movement frequency and appearance.
“We are seeking to expand through collaboration with additional GI doctors. We want to develop a more advanced prototype and do further validation studies,” Dr. Grego said.
Digital health tool
There is an aversion among patients to handling stool “or even talking about it,” Dr. Grego said. Colleagues tell her that people are more willing to provide a blood sample, which requires a needle, than a stool sample.
“But a lot of health data is there [in the stool],” she added. “We think this will empower a lot of research as well as consumer data gathering.”
For example, Dr. Grego envisions pharmaceutical companies using the technology to detect or monitor any changes in stool or gut health based on a treatment in development during clinical trials.
Furthermore, the technology might empower health-conscious consumers who want to track their own gut health. “This technology will be a whole new entry in the digital health toolkit,” Dr. Grego said.
Although not included in the research presented at this year’s DDW, the developers plan to add a sampling capability. Biochemic analysis of stool samples could provide “metabolically relevant information,” including stool biomarkers and microbiome composition.
“We have demonstrated it in the laboratory. It will be part of the technology when developed into a product,” Dr. Grego said.
This proof-of-concept study “is the first step in a path we are aggressively pursuing,” Dr. Grego said. She estimated it will take about 12-18 months to develop a prototype for use with patients. “We hope to move to a product soon after that.”
“I’m looking forward to seeing future iterations of this tool,” Dr. Shin said. “It could have a role in monitoring important GI diseases and disorders, including IBS and inflammatory bowel disease, or even for the detection of ‘alarm symptoms’ that shouldn’t be ignored.
“I could even see it having a role in preventative health in the future,” Dr. Shin added.
The technology has been licensed to the spin-off company Coprata to develop the product further.
“We hope to have an impact on people’s health very soon,” Dr. Grego said.
Duke University funded the study. Dr. Grego holds a management position at Coprata. Dr. Shin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A “smart toilet” in development uses artificial intelligence (AI) to scan stool for consistency and presence of blood – and early evidence suggests it is more accurate than patient self-reporting, a study reveals.
The remote, automated, real-time analysis and reporting increase the likelihood of physicians detecting gastrointestinal issues earlier, investigators reported.
In a proof-of-concept study, the smart toilet was 85% accurate in categorizing stool consistency as loose, normal, or constipated. The findings were presented at the annual Digestive Disease Week® (DDW).
“This study highlights a very innovative and practical tool that could have major implications for patients and clinicians alike,” Andrea Shin, MD, who was not affiliated with the research, said in an interview.
“Stool form or consistency and signs of bleeding are some of the most important pieces of clinical history when it comes to GI or bowel symptoms,” added Dr. Shin, assistant professor of medicine in the department of gastroenterology and hepatology at Indiana University, Indianapolis.
Image analysis
The researchers tested their AI algorithm on 3,328 images. They assessed photos from the Internet and some submitted anonymously by participants in the study.
Two gastroenterologists also rated a subset of 552 images. The physicians showed “satisfactory agreement” on interrater reliability (the extent to which two or more “raters” [for example, observers, examiners] agree), the investigators noted.
The smart toilet also was 76% accurate for gross blood detection.
“It’s objective and more accurate,” study author Sonia Grego, PhD, said in an interview. In contrast to asking patients to keep a bowel movement diary or recall the frequency and consistency of their stool over time, “the system does it for you,” she added.
“Our technology – by automating the image acquisition – removes the burden of having to track your pattern for weeks or months,” added Dr. Grego, founding director of the Duke Smart Toilet Lab at Duke University in Durham, N.C.
Information provided by patients “can have a big impact on decision-making,” Dr. Shin said. “For example, if I am talking to an individual who suffers from irritable bowel syndrome [IBS], I commonly ask them about how loose or watery and hard or formed their stool is, because this information gives me clues as to the underlying problems that may be driving their symptoms.”
Dr. Shin agreed it can be challenging for people to know what is important to report to their doctor. “This tool has the potential to relieve patient burden and facilitate communication between a patient and their clinician. It’s a great example of how technology can be leveraged to enhance care.”
Working behind the scenes
Together with gastroenterologist Deborah Anne Fisher, MD, an associate professor of medicine at Duke, Dr. Grego and colleagues devised a prototype that positions the image analyzer in the pipes behind the toilet. So the analysis is done post flush.
“We are experts of toilets and toilet technology,” Dr. Grego said. “We have learned that people really don’t like to see anything weird around the toilet bowl.”
The smart toilet system is designed for multiple users in a residential or commercial setting. The technology could be used in hospitals or long-term care facilities, for example. A fingerprint scanner on the flush mechanism tracks each individual user.
Mixed reactions
Dr. Grego gets a range of reactions when she tells people she is developing smart toilet technology.
“Friends and family laugh about the concept of the smart toilet,” she said, “so all the possible jokes that have been done on poops, we know.”
In fact, the researchers also are collecting the jokes they hear. “We’re being very systematic.”
In contrast, gastroenterologists who learn of the technology in development are more enthusiastic, Dr. Grego said. “There is such a need for removing the uncertainty of the patient recall about bowel movement frequency and appearance.
“We are seeking to expand through collaboration with additional GI doctors. We want to develop a more advanced prototype and do further validation studies,” Dr. Grego said.
Digital health tool
There is an aversion among patients to handling stool “or even talking about it,” Dr. Grego said. Colleagues tell her that people are more willing to provide a blood sample, which requires a needle, than a stool sample.
“But a lot of health data is there [in the stool],” she added. “We think this will empower a lot of research as well as consumer data gathering.”
For example, Dr. Grego envisions pharmaceutical companies using the technology to detect or monitor any changes in stool or gut health based on a treatment in development during clinical trials.
Furthermore, the technology might empower health-conscious consumers who want to track their own gut health. “This technology will be a whole new entry in the digital health toolkit,” Dr. Grego said.
Although not included in the research presented at this year’s DDW, the developers plan to add a sampling capability. Biochemic analysis of stool samples could provide “metabolically relevant information,” including stool biomarkers and microbiome composition.
“We have demonstrated it in the laboratory. It will be part of the technology when developed into a product,” Dr. Grego said.
This proof-of-concept study “is the first step in a path we are aggressively pursuing,” Dr. Grego said. She estimated it will take about 12-18 months to develop a prototype for use with patients. “We hope to move to a product soon after that.”
“I’m looking forward to seeing future iterations of this tool,” Dr. Shin said. “It could have a role in monitoring important GI diseases and disorders, including IBS and inflammatory bowel disease, or even for the detection of ‘alarm symptoms’ that shouldn’t be ignored.
“I could even see it having a role in preventative health in the future,” Dr. Shin added.
The technology has been licensed to the spin-off company Coprata to develop the product further.
“We hope to have an impact on people’s health very soon,” Dr. Grego said.
Duke University funded the study. Dr. Grego holds a management position at Coprata. Dr. Shin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Multiple Crusted Swellings on the Chin
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).
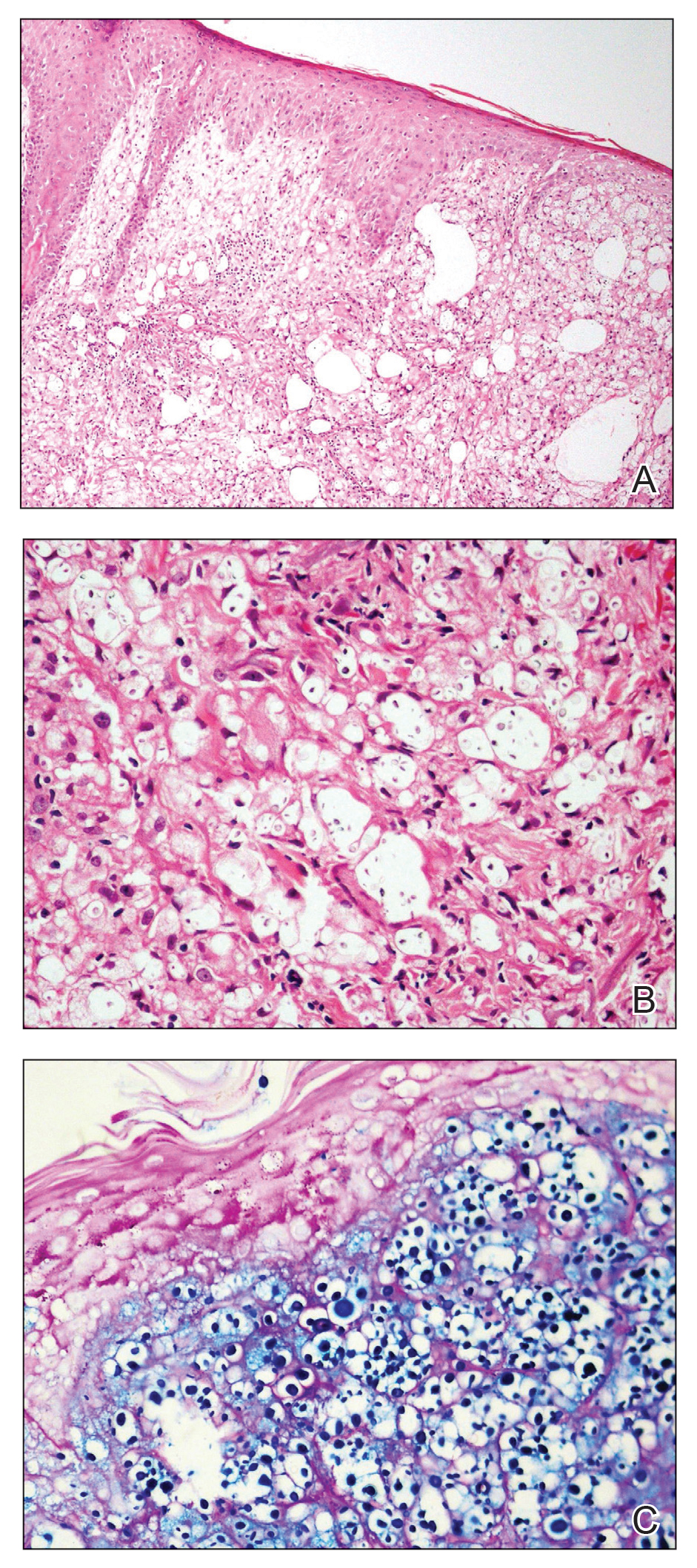
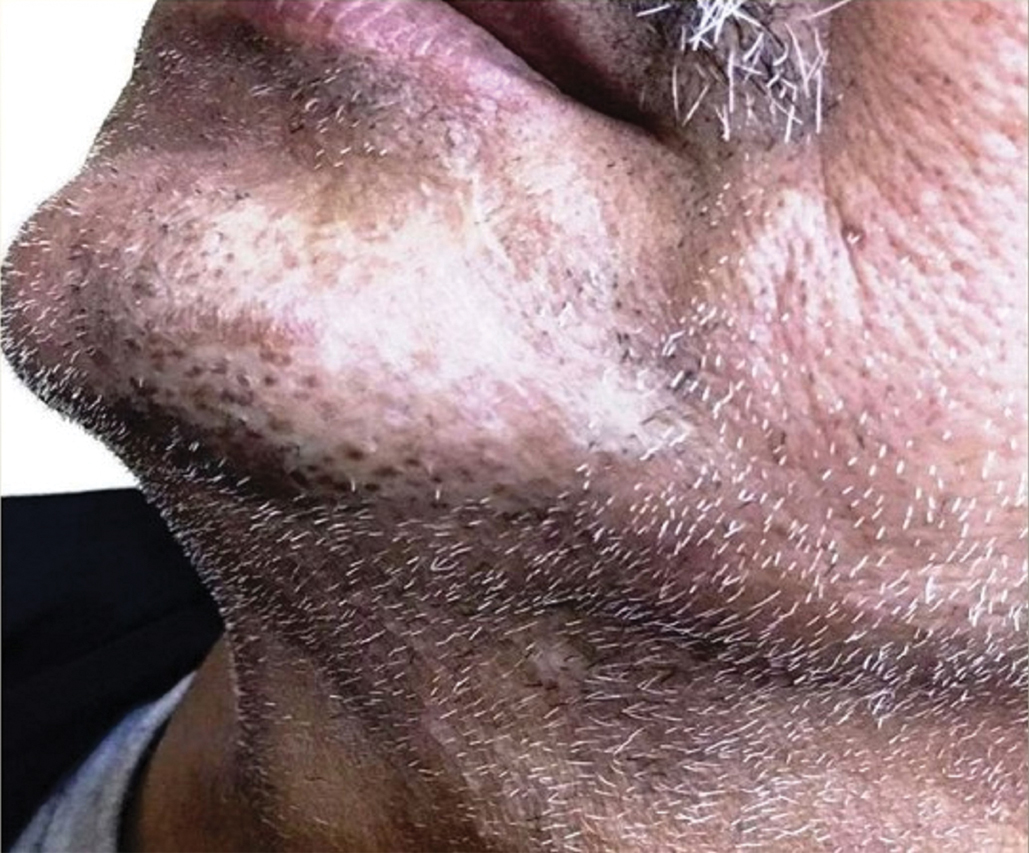
Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).


Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).


Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
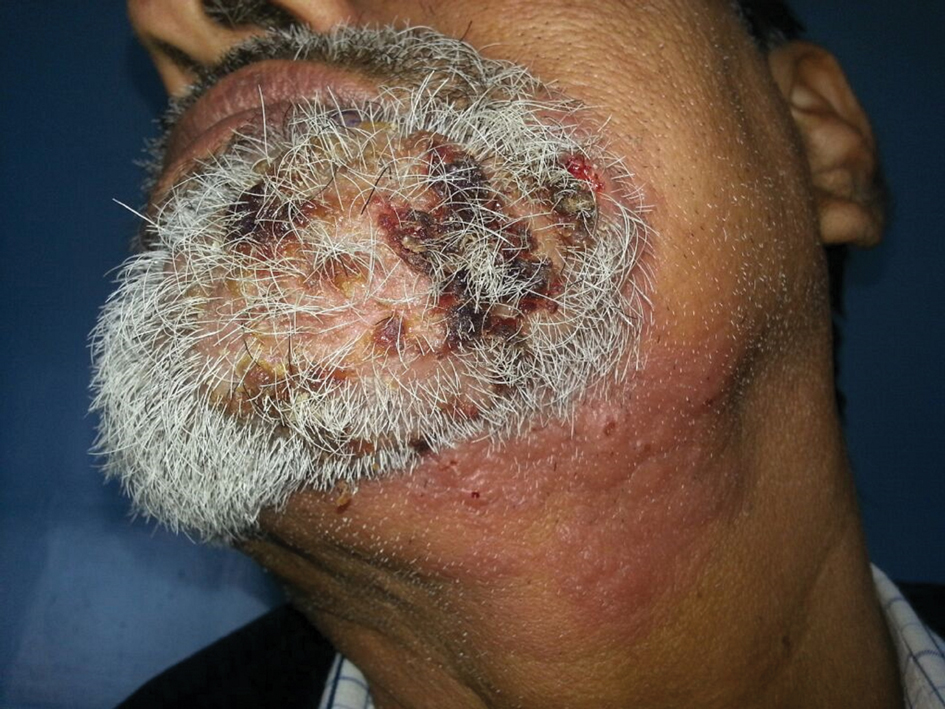
A 54-year-old man with no comorbidities presented with multiple painless swellings on the left side of the chin of 1 month’s duration that progressively were increasing, both in size and number. He denied any discharge of pus or grains from the lesion, facial trauma, insect bites, or dental procedures. The patient was treated with oral antibiotics for 15 days with no relief at an outside hospital. All routine blood and serologic investigations including viral markers and chest radiography were normal. Bacterial and fungal cultures as well as an acid-fast bacilli culture were negative. Systemic examination was normal, and vitals were within reference range. Mucocutaneous examination revealed multiple nontender small nodules and plaques with yellow-brown to dark brown hemorrhagic crusts with mild perilesional erythema on the left side of the chin extending to the adjacent neck. All mucosal sites were normal, and a biopsy was performed.