User login
For MD-IQ use only
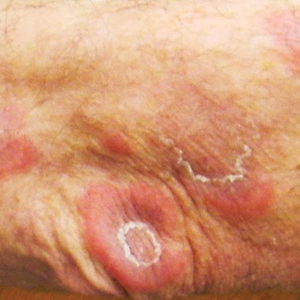
Surgical site infections not increased in immunocompromised patients after Mohs surgery
, suggesting that antibiotic prophylaxis, which is often used for these patients, may not be necessary, according to new research.
The retrospective cohort study found that “immunosuppressed patients had similar infection rates as immunocompetent patients following Mohs micrographic surgery,” first author Tuyet A. Nguyen, MD, of the department of dermatology, Cedars-Sinai Medical Center, Los Angeles, told this news organization.
“Therefore, antibiotic prescribing patterns should not change simply due to immunosuppression. Furthermore, immunosuppressed patients appear to respond well to antibiotics and recover similarly to immunocompetent patients,” she said.
The study was presented at the annual meeting of the American College of Mohs Surgery.
Mohs surgery is increasingly being performed for patients who are immunosuppressed because of the higher incidence of skin cancer in this group of patients and their higher risk of more aggressive skin cancers.
Overall, the rate of surgical site infections following Mohs surgery generally ranges from 0.5% to 2.4%. However, research is lacking on the risk among patients who are immunosuppressed and on how effective the use of prophylactic antibiotics is for these patients.
For the retrospective study, Dr. Nguyen and her colleagues evaluated data on 5,886 patients who underwent Mohs surgery at Cedars-Sinai between October 2014 and August 2021. Among these patients, 741 (12.6%) were immunocompromised.
Causes of immunosuppression in the cohort included the following: immunosuppression after transplant surgery; having HIV, chronic myeloid leukemia, multiple myeloma, or other hematogenous forms of immunosuppression; or immunosuppression related to other conditions, such as chronic inflammatory diseases.
Overall, postprocedural infections occurred in 1.6% (95) of patients, a rate that mirrors that of the general population, Dr. Nguyen noted. No significant differences in surgical site infection rates were observed between immunocompromised patients (2.1%, n = 15) and those who were immunocompetent (1.6%, n = 80; P = .30).
Importantly, among those who were immunocompromised, the rates of infection were not significantly different between those who did receive antibiotics (3.0%, n = 8) and those who did not receive antibiotics (1.5%, n = 7; P = .19).
The lack of a difference in surgical site infection rates among those who did and those who did not receive antibiotics extended to the entire study population (2.0% vs. 1.4%; P = .12).
The study cohort mainly comprised immunosuppressed transplant patients, notably, heart, lung, and kidney transplant patients. However, “even in this population, we did not see a higher rate of infection,” senior author Nima M. Gharavi, MD, PhD, director of dermatologic surgery and Mohs micrographic surgery and associate professor of medicine and pathology and laboratory medicine at Cedars-Sinai Medical Center, said in an interview.
Yet the risk of infection among those patients has been shown to be high and of consequence. Data indicate that infections account for 13%-16% of deaths among kidney and heart transplant patients and up to 21% of deaths among lung transplant patients. The rate of mortality appears to parallel the level of immunosuppression, Dr. Nguyen explained.
Furthermore, up to 25% of patients who undergo heart and lung transplantation develop bacteremia.
In terms of why worse infections or bacteremia surgeries may not occur in association with Mohs, Dr. Nguyen speculated that, as opposed to other surgeries, those involving the skin may benefit from unique defense mechanisms.
“The skin is a complex system in its defense against foreign pathogens and infectious agents,” she explained during her presentation. “There is the physical barrier, the antimicrobial peptides, and an adaptive as well as innate immune response.”
“In immunosuppressed patients, with the decrease in adaptive immunity, it’s possible this loss is less important because the skin has such a robust immune system in general.”
In her presentation, Dr. Nguyen noted that “further studies are necessary to investigate why patients aren’t presenting with greater severity, and we plan to try to investigate whether the unique nature of skin-mediated immunity makes this organ less susceptible to severe or life-threatening infections in patients on immunosuppression.”
Of note, the rate of prophylactic antibiotic prescriptions was no higher for those who were and those who were not immunosuppressed (37.9% vs. 34.1%; P = .14), which Dr. Nguyen said is consistent with recommendations.
“Immunosuppression is not an indication for antibiotic use, and hence, we did not have a higher rate of antibiotics use in this population,” she told this news organization. However, a 2021 ACMS survey found that a high percentage of Mohs surgeons prescribe antibiotics for procedures in which antibiotics are not indicated so as to reduce the risk of infections and that immunosuppression is a common reason for doing so.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, suggesting that antibiotic prophylaxis, which is often used for these patients, may not be necessary, according to new research.
The retrospective cohort study found that “immunosuppressed patients had similar infection rates as immunocompetent patients following Mohs micrographic surgery,” first author Tuyet A. Nguyen, MD, of the department of dermatology, Cedars-Sinai Medical Center, Los Angeles, told this news organization.
“Therefore, antibiotic prescribing patterns should not change simply due to immunosuppression. Furthermore, immunosuppressed patients appear to respond well to antibiotics and recover similarly to immunocompetent patients,” she said.
The study was presented at the annual meeting of the American College of Mohs Surgery.
Mohs surgery is increasingly being performed for patients who are immunosuppressed because of the higher incidence of skin cancer in this group of patients and their higher risk of more aggressive skin cancers.
Overall, the rate of surgical site infections following Mohs surgery generally ranges from 0.5% to 2.4%. However, research is lacking on the risk among patients who are immunosuppressed and on how effective the use of prophylactic antibiotics is for these patients.
For the retrospective study, Dr. Nguyen and her colleagues evaluated data on 5,886 patients who underwent Mohs surgery at Cedars-Sinai between October 2014 and August 2021. Among these patients, 741 (12.6%) were immunocompromised.
Causes of immunosuppression in the cohort included the following: immunosuppression after transplant surgery; having HIV, chronic myeloid leukemia, multiple myeloma, or other hematogenous forms of immunosuppression; or immunosuppression related to other conditions, such as chronic inflammatory diseases.
Overall, postprocedural infections occurred in 1.6% (95) of patients, a rate that mirrors that of the general population, Dr. Nguyen noted. No significant differences in surgical site infection rates were observed between immunocompromised patients (2.1%, n = 15) and those who were immunocompetent (1.6%, n = 80; P = .30).
Importantly, among those who were immunocompromised, the rates of infection were not significantly different between those who did receive antibiotics (3.0%, n = 8) and those who did not receive antibiotics (1.5%, n = 7; P = .19).
The lack of a difference in surgical site infection rates among those who did and those who did not receive antibiotics extended to the entire study population (2.0% vs. 1.4%; P = .12).
The study cohort mainly comprised immunosuppressed transplant patients, notably, heart, lung, and kidney transplant patients. However, “even in this population, we did not see a higher rate of infection,” senior author Nima M. Gharavi, MD, PhD, director of dermatologic surgery and Mohs micrographic surgery and associate professor of medicine and pathology and laboratory medicine at Cedars-Sinai Medical Center, said in an interview.
Yet the risk of infection among those patients has been shown to be high and of consequence. Data indicate that infections account for 13%-16% of deaths among kidney and heart transplant patients and up to 21% of deaths among lung transplant patients. The rate of mortality appears to parallel the level of immunosuppression, Dr. Nguyen explained.
Furthermore, up to 25% of patients who undergo heart and lung transplantation develop bacteremia.
In terms of why worse infections or bacteremia surgeries may not occur in association with Mohs, Dr. Nguyen speculated that, as opposed to other surgeries, those involving the skin may benefit from unique defense mechanisms.
“The skin is a complex system in its defense against foreign pathogens and infectious agents,” she explained during her presentation. “There is the physical barrier, the antimicrobial peptides, and an adaptive as well as innate immune response.”
“In immunosuppressed patients, with the decrease in adaptive immunity, it’s possible this loss is less important because the skin has such a robust immune system in general.”
In her presentation, Dr. Nguyen noted that “further studies are necessary to investigate why patients aren’t presenting with greater severity, and we plan to try to investigate whether the unique nature of skin-mediated immunity makes this organ less susceptible to severe or life-threatening infections in patients on immunosuppression.”
Of note, the rate of prophylactic antibiotic prescriptions was no higher for those who were and those who were not immunosuppressed (37.9% vs. 34.1%; P = .14), which Dr. Nguyen said is consistent with recommendations.
“Immunosuppression is not an indication for antibiotic use, and hence, we did not have a higher rate of antibiotics use in this population,” she told this news organization. However, a 2021 ACMS survey found that a high percentage of Mohs surgeons prescribe antibiotics for procedures in which antibiotics are not indicated so as to reduce the risk of infections and that immunosuppression is a common reason for doing so.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, suggesting that antibiotic prophylaxis, which is often used for these patients, may not be necessary, according to new research.
The retrospective cohort study found that “immunosuppressed patients had similar infection rates as immunocompetent patients following Mohs micrographic surgery,” first author Tuyet A. Nguyen, MD, of the department of dermatology, Cedars-Sinai Medical Center, Los Angeles, told this news organization.
“Therefore, antibiotic prescribing patterns should not change simply due to immunosuppression. Furthermore, immunosuppressed patients appear to respond well to antibiotics and recover similarly to immunocompetent patients,” she said.
The study was presented at the annual meeting of the American College of Mohs Surgery.
Mohs surgery is increasingly being performed for patients who are immunosuppressed because of the higher incidence of skin cancer in this group of patients and their higher risk of more aggressive skin cancers.
Overall, the rate of surgical site infections following Mohs surgery generally ranges from 0.5% to 2.4%. However, research is lacking on the risk among patients who are immunosuppressed and on how effective the use of prophylactic antibiotics is for these patients.
For the retrospective study, Dr. Nguyen and her colleagues evaluated data on 5,886 patients who underwent Mohs surgery at Cedars-Sinai between October 2014 and August 2021. Among these patients, 741 (12.6%) were immunocompromised.
Causes of immunosuppression in the cohort included the following: immunosuppression after transplant surgery; having HIV, chronic myeloid leukemia, multiple myeloma, or other hematogenous forms of immunosuppression; or immunosuppression related to other conditions, such as chronic inflammatory diseases.
Overall, postprocedural infections occurred in 1.6% (95) of patients, a rate that mirrors that of the general population, Dr. Nguyen noted. No significant differences in surgical site infection rates were observed between immunocompromised patients (2.1%, n = 15) and those who were immunocompetent (1.6%, n = 80; P = .30).
Importantly, among those who were immunocompromised, the rates of infection were not significantly different between those who did receive antibiotics (3.0%, n = 8) and those who did not receive antibiotics (1.5%, n = 7; P = .19).
The lack of a difference in surgical site infection rates among those who did and those who did not receive antibiotics extended to the entire study population (2.0% vs. 1.4%; P = .12).
The study cohort mainly comprised immunosuppressed transplant patients, notably, heart, lung, and kidney transplant patients. However, “even in this population, we did not see a higher rate of infection,” senior author Nima M. Gharavi, MD, PhD, director of dermatologic surgery and Mohs micrographic surgery and associate professor of medicine and pathology and laboratory medicine at Cedars-Sinai Medical Center, said in an interview.
Yet the risk of infection among those patients has been shown to be high and of consequence. Data indicate that infections account for 13%-16% of deaths among kidney and heart transplant patients and up to 21% of deaths among lung transplant patients. The rate of mortality appears to parallel the level of immunosuppression, Dr. Nguyen explained.
Furthermore, up to 25% of patients who undergo heart and lung transplantation develop bacteremia.
In terms of why worse infections or bacteremia surgeries may not occur in association with Mohs, Dr. Nguyen speculated that, as opposed to other surgeries, those involving the skin may benefit from unique defense mechanisms.
“The skin is a complex system in its defense against foreign pathogens and infectious agents,” she explained during her presentation. “There is the physical barrier, the antimicrobial peptides, and an adaptive as well as innate immune response.”
“In immunosuppressed patients, with the decrease in adaptive immunity, it’s possible this loss is less important because the skin has such a robust immune system in general.”
In her presentation, Dr. Nguyen noted that “further studies are necessary to investigate why patients aren’t presenting with greater severity, and we plan to try to investigate whether the unique nature of skin-mediated immunity makes this organ less susceptible to severe or life-threatening infections in patients on immunosuppression.”
Of note, the rate of prophylactic antibiotic prescriptions was no higher for those who were and those who were not immunosuppressed (37.9% vs. 34.1%; P = .14), which Dr. Nguyen said is consistent with recommendations.
“Immunosuppression is not an indication for antibiotic use, and hence, we did not have a higher rate of antibiotics use in this population,” she told this news organization. However, a 2021 ACMS survey found that a high percentage of Mohs surgeons prescribe antibiotics for procedures in which antibiotics are not indicated so as to reduce the risk of infections and that immunosuppression is a common reason for doing so.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACMS ANNUAL MEETING
FDA denies petition to disqualify researchers over controversial ketamine studies
The U.S. Food and Drug Administration has declined to take further action against a group of investigators at Hennepin County Medical Center/Hennepin Healthcare (HCMC) who conducted controversial studies involving ketamine and other sedatives on agitated persons without their consent.
A citizen petition filed by Public Citizen, a consumer advocacy group, had asked the FDA to initiate clinical-investigator disqualification proceedings against Jon Cole, MD, and Lauren Klein, MD, along with other researchers who participated in the studies, for “repeatedly and deliberately initiating and conducting clinical investigations of investigational drug products” without having submitted or having in effect the investigational new drug applications (INDs) required by the FDA.
In certain situations, wherein the FDA alleges that a clinical investigator has violated applicable regulations, the agency may initiate clinical investigator disqualification proceedings. The names of the disqualified researchers are then added to a federal database.
The petition, which was filed in November 2021, also requested that the FDA initiate disqualification proceedings against the institutional review board (IRB) at HCMC for repeatedly failing to comply with federal regulations that adversely affected the rights and welfare of the individuals who were enrolled in the study without their consent.
Of note, Public Citizen stated that the FDA should have required the hospital to contact the more than 1,700 patients who “were unwittingly enrolled in unethical experiments” and inform them that their rights had been violated and their health potentially endangered by the research team.
Michael A. Carome, MD, director of Public Citizen’s Health Research Group, told this news organization that it is uncommon for the FDA to disqualify researchers. “It should be more common than it is,” he said. “I think that FDA is just reluctant to take more action.”
The actions of the Hennepin investigators were “repetitive and appeared to be in deliberate violation of regulations,” he added. “The case for the FDA disqualifying the HCMC researchers is overwhelming. The FDA’s slap-on-the-wrist approach to such appalling regulatory and ethical violations risks emboldening other researchers to disregard the rights and welfare of human subjects.”
Carl Elliott, MD, PhD, a bioethicist at the University of Minnesota, Minneapolis, agrees that the researcher from HCMC should be disqualified. “They didn’t just conduct risky, exploitative studies – they conducted them after the FDA had warned them not to proceed,” he said. “The message sent by this slap on the wrist is that investigators can do whatever they want to nonconsenting subjects, and the FDA will look the other way.”
Initial complaint
Public Citizen initially filed a complaint with the FDA in 2018, after learning that researchers affiliated with HCMC were conducting high-risk clinical trials involving ketamine to control agitation outside of the hospital setting. The complaint was cosigned by 64 doctors, bioethicists, and academic researchers and was also submitted to the Office for Human Research Protections.
The FDA typically allows investigational drugs to be used in emergency situation without obtaining informed consent if the therapies are known to carry a minimal risk. The IRB at HCMC had determined that this was the case with ketamine and approved the trials.
But according to Public Citizen’s complaint, prior research had suggested that ketamine could cause more complications and severe adverse events, compared with other sedatives.
The trials were conducted between 2014 and 2018, and in its letter, Public Citizen alleged that the investigators and the IRB had allowed these trials to proceed without obtaining informed consent from patients. The goal was to evaluate how well ketamine worked, compared with other drugs in calming agitated individuals: “The patients were given either ketamine or haloperidol for agitation by paramedics who responded to medical emergencies, and the goal was to see which drug worked faster,” said Dr. Carome. “Patients were only notified afterwards that they had received a sedative. Informed consent had been waived by IRB.”
In the first clinical trial conducted by HCMC, published in 2016, the researchers had hypothesized that 5 mg/kg of intramuscular ketamine would be superior to 10 mg of intramuscular haloperidol for severe prehospital agitation. Time to adequate sedation was the primary outcome measure. The study included 146 people; 64 received ketamine and 82 received haloperidol. They found that ketamine worked far more quickly than haloperidol (5 minutes vs. 17 minutes) but that the risk for complications was much higher. Complications occurred in 49% of patients receiving ketamine, compared with 5%.
“There was a 10-fold risk of adverse events,” said Dr. Carome. “And 39% of patients given ketamine had respiratory problems requiring intubation, compared to 4% who received haloperidol.”
A second study was launched in 2017, wherein ketamine was compared with midazolam in agitated patients. During the first 6-month period of the study, individuals would receive a ketamine-based protocol for prehospital agitation, and during the second 6 months, that would switch to midazolam. However, the study was halted in June 2018 after the local newspaper, the Star Tribune, reported that the city police had encouraged medical personnel to sedate agitated patients. This included individuals who had already been physically restrained.
The report stated that “in many cases, the individual being detained or arrested was not only handcuffed but strapped down on a stretcher in an ambulance before receiving ketamine,” and that it raised a “concerning question” over why these people were given the drug before they were transported to the hospital, “given the immediate effects on breathing and heart function that the drug induces.”
Along with halting the trial, HCMC asked for a review of cases involving its paramedics; an independent investigation led by former U.S. Deputy Attorney General Sally Yates was initiated to assess whether the Minneapolis police had crossed a line and urged paramedics to use ketamine.
“The decision to use ketamine was based on the study’s timeline and not on clinical judgment,” said Dr. Carome.
The FDA acknowledged receipt of the complaint and inspected the IRB records and the clinical trial data. Preliminary reports received by Public Citizen confirmed their allegations. “There were not appropriate protections for vulnerable subjects,” he said. “In 2019, the FDA did further investigations, and those reports had similar findings.”
FDA letters
The FDA had sent warning letters to Dr. Cole and Dr. Klein, citing them for ignoring federal safety laws in experimental research on the public. In their investigations, the FDA cited “objectionable conditions” for the studies led by Dr. Cole and Dr. Klein, according to the letters. Both researchers seemingly ignored FDA regulations and used practices that subjected patients to “significantly increased risk,” and the hospital defended its research with “factually incorrect” statements.
In a letter to Dr. Cole, the FDA noted that he never filed INDs for the trials with the FDA, as required by law, and that he also failed to write appropriate protocols to ensure that children and pregnant women were not enrolled in the research. Individuals under the influence of intoxicants also were not excluded, though the use of ketamine is cautioned in this population.
“Administration of the investigational drugs to these subjects placed them at significantly increased risk of the adverse events associated with the investigational products and decreased the acceptability of those risks,” the FDA said in its letter. “Your failure to exclude, and the lack of any precautions for, subjects under the influence of various intoxicants significantly increased the risks and/or decreased the acceptability of the risks associated with the investigational drugs.”
However, Dr. Cole conducted both studies in the prehospital setting and failed to initiate any specific measures to protect study participants, according to the FDA.
Petition denied
Dr. Carome noted that the researchers had committed repetitive egregious regulatory violations over a 4-year period, which were documented by the FDA in their warning letters to Dr. Cole and Dr. Klein. “We felt that they were so egregious that we need to send a signal to the community that this sort of behavior will not be tolerated,” he said. “The FDA denied our petition, and we think that sends the wrong signal to the research community.”
In their response, the FDA noted that as with judicial enforcement, “the Agency makes decisions regarding whether to pursue administrative enforcement action, including disqualification proceedings, on a case-by-case basis, considering all relevant facts and circumstances.” They added that at this time, they would not be taking further action against Dr. Cole and Dr. Klein.
“However, we intend to continue to consider all the options available to the Agency as we determine whether to pursue additional compliance actions related to this matter,” the FDA concluded.
The FDA declined to comment further on their decision.
Dr. Cole also declined to comment, but Hennepin Healthcare told this news organization that the “decision by the FDA to deny the petition validates the changes we made to strengthen and improve the clinical research program across the institution since the closing of the studies in 2018. We look forward to continuing to work with the FDA to ensure full compliance with the standards in place to protect research subjects.”
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has declined to take further action against a group of investigators at Hennepin County Medical Center/Hennepin Healthcare (HCMC) who conducted controversial studies involving ketamine and other sedatives on agitated persons without their consent.
A citizen petition filed by Public Citizen, a consumer advocacy group, had asked the FDA to initiate clinical-investigator disqualification proceedings against Jon Cole, MD, and Lauren Klein, MD, along with other researchers who participated in the studies, for “repeatedly and deliberately initiating and conducting clinical investigations of investigational drug products” without having submitted or having in effect the investigational new drug applications (INDs) required by the FDA.
In certain situations, wherein the FDA alleges that a clinical investigator has violated applicable regulations, the agency may initiate clinical investigator disqualification proceedings. The names of the disqualified researchers are then added to a federal database.
The petition, which was filed in November 2021, also requested that the FDA initiate disqualification proceedings against the institutional review board (IRB) at HCMC for repeatedly failing to comply with federal regulations that adversely affected the rights and welfare of the individuals who were enrolled in the study without their consent.
Of note, Public Citizen stated that the FDA should have required the hospital to contact the more than 1,700 patients who “were unwittingly enrolled in unethical experiments” and inform them that their rights had been violated and their health potentially endangered by the research team.
Michael A. Carome, MD, director of Public Citizen’s Health Research Group, told this news organization that it is uncommon for the FDA to disqualify researchers. “It should be more common than it is,” he said. “I think that FDA is just reluctant to take more action.”
The actions of the Hennepin investigators were “repetitive and appeared to be in deliberate violation of regulations,” he added. “The case for the FDA disqualifying the HCMC researchers is overwhelming. The FDA’s slap-on-the-wrist approach to such appalling regulatory and ethical violations risks emboldening other researchers to disregard the rights and welfare of human subjects.”
Carl Elliott, MD, PhD, a bioethicist at the University of Minnesota, Minneapolis, agrees that the researcher from HCMC should be disqualified. “They didn’t just conduct risky, exploitative studies – they conducted them after the FDA had warned them not to proceed,” he said. “The message sent by this slap on the wrist is that investigators can do whatever they want to nonconsenting subjects, and the FDA will look the other way.”
Initial complaint
Public Citizen initially filed a complaint with the FDA in 2018, after learning that researchers affiliated with HCMC were conducting high-risk clinical trials involving ketamine to control agitation outside of the hospital setting. The complaint was cosigned by 64 doctors, bioethicists, and academic researchers and was also submitted to the Office for Human Research Protections.
The FDA typically allows investigational drugs to be used in emergency situation without obtaining informed consent if the therapies are known to carry a minimal risk. The IRB at HCMC had determined that this was the case with ketamine and approved the trials.
But according to Public Citizen’s complaint, prior research had suggested that ketamine could cause more complications and severe adverse events, compared with other sedatives.
The trials were conducted between 2014 and 2018, and in its letter, Public Citizen alleged that the investigators and the IRB had allowed these trials to proceed without obtaining informed consent from patients. The goal was to evaluate how well ketamine worked, compared with other drugs in calming agitated individuals: “The patients were given either ketamine or haloperidol for agitation by paramedics who responded to medical emergencies, and the goal was to see which drug worked faster,” said Dr. Carome. “Patients were only notified afterwards that they had received a sedative. Informed consent had been waived by IRB.”
In the first clinical trial conducted by HCMC, published in 2016, the researchers had hypothesized that 5 mg/kg of intramuscular ketamine would be superior to 10 mg of intramuscular haloperidol for severe prehospital agitation. Time to adequate sedation was the primary outcome measure. The study included 146 people; 64 received ketamine and 82 received haloperidol. They found that ketamine worked far more quickly than haloperidol (5 minutes vs. 17 minutes) but that the risk for complications was much higher. Complications occurred in 49% of patients receiving ketamine, compared with 5%.
“There was a 10-fold risk of adverse events,” said Dr. Carome. “And 39% of patients given ketamine had respiratory problems requiring intubation, compared to 4% who received haloperidol.”
A second study was launched in 2017, wherein ketamine was compared with midazolam in agitated patients. During the first 6-month period of the study, individuals would receive a ketamine-based protocol for prehospital agitation, and during the second 6 months, that would switch to midazolam. However, the study was halted in June 2018 after the local newspaper, the Star Tribune, reported that the city police had encouraged medical personnel to sedate agitated patients. This included individuals who had already been physically restrained.
The report stated that “in many cases, the individual being detained or arrested was not only handcuffed but strapped down on a stretcher in an ambulance before receiving ketamine,” and that it raised a “concerning question” over why these people were given the drug before they were transported to the hospital, “given the immediate effects on breathing and heart function that the drug induces.”
Along with halting the trial, HCMC asked for a review of cases involving its paramedics; an independent investigation led by former U.S. Deputy Attorney General Sally Yates was initiated to assess whether the Minneapolis police had crossed a line and urged paramedics to use ketamine.
“The decision to use ketamine was based on the study’s timeline and not on clinical judgment,” said Dr. Carome.
The FDA acknowledged receipt of the complaint and inspected the IRB records and the clinical trial data. Preliminary reports received by Public Citizen confirmed their allegations. “There were not appropriate protections for vulnerable subjects,” he said. “In 2019, the FDA did further investigations, and those reports had similar findings.”
FDA letters
The FDA had sent warning letters to Dr. Cole and Dr. Klein, citing them for ignoring federal safety laws in experimental research on the public. In their investigations, the FDA cited “objectionable conditions” for the studies led by Dr. Cole and Dr. Klein, according to the letters. Both researchers seemingly ignored FDA regulations and used practices that subjected patients to “significantly increased risk,” and the hospital defended its research with “factually incorrect” statements.
In a letter to Dr. Cole, the FDA noted that he never filed INDs for the trials with the FDA, as required by law, and that he also failed to write appropriate protocols to ensure that children and pregnant women were not enrolled in the research. Individuals under the influence of intoxicants also were not excluded, though the use of ketamine is cautioned in this population.
“Administration of the investigational drugs to these subjects placed them at significantly increased risk of the adverse events associated with the investigational products and decreased the acceptability of those risks,” the FDA said in its letter. “Your failure to exclude, and the lack of any precautions for, subjects under the influence of various intoxicants significantly increased the risks and/or decreased the acceptability of the risks associated with the investigational drugs.”
However, Dr. Cole conducted both studies in the prehospital setting and failed to initiate any specific measures to protect study participants, according to the FDA.
Petition denied
Dr. Carome noted that the researchers had committed repetitive egregious regulatory violations over a 4-year period, which were documented by the FDA in their warning letters to Dr. Cole and Dr. Klein. “We felt that they were so egregious that we need to send a signal to the community that this sort of behavior will not be tolerated,” he said. “The FDA denied our petition, and we think that sends the wrong signal to the research community.”
In their response, the FDA noted that as with judicial enforcement, “the Agency makes decisions regarding whether to pursue administrative enforcement action, including disqualification proceedings, on a case-by-case basis, considering all relevant facts and circumstances.” They added that at this time, they would not be taking further action against Dr. Cole and Dr. Klein.
“However, we intend to continue to consider all the options available to the Agency as we determine whether to pursue additional compliance actions related to this matter,” the FDA concluded.
The FDA declined to comment further on their decision.
Dr. Cole also declined to comment, but Hennepin Healthcare told this news organization that the “decision by the FDA to deny the petition validates the changes we made to strengthen and improve the clinical research program across the institution since the closing of the studies in 2018. We look forward to continuing to work with the FDA to ensure full compliance with the standards in place to protect research subjects.”
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has declined to take further action against a group of investigators at Hennepin County Medical Center/Hennepin Healthcare (HCMC) who conducted controversial studies involving ketamine and other sedatives on agitated persons without their consent.
A citizen petition filed by Public Citizen, a consumer advocacy group, had asked the FDA to initiate clinical-investigator disqualification proceedings against Jon Cole, MD, and Lauren Klein, MD, along with other researchers who participated in the studies, for “repeatedly and deliberately initiating and conducting clinical investigations of investigational drug products” without having submitted or having in effect the investigational new drug applications (INDs) required by the FDA.
In certain situations, wherein the FDA alleges that a clinical investigator has violated applicable regulations, the agency may initiate clinical investigator disqualification proceedings. The names of the disqualified researchers are then added to a federal database.
The petition, which was filed in November 2021, also requested that the FDA initiate disqualification proceedings against the institutional review board (IRB) at HCMC for repeatedly failing to comply with federal regulations that adversely affected the rights and welfare of the individuals who were enrolled in the study without their consent.
Of note, Public Citizen stated that the FDA should have required the hospital to contact the more than 1,700 patients who “were unwittingly enrolled in unethical experiments” and inform them that their rights had been violated and their health potentially endangered by the research team.
Michael A. Carome, MD, director of Public Citizen’s Health Research Group, told this news organization that it is uncommon for the FDA to disqualify researchers. “It should be more common than it is,” he said. “I think that FDA is just reluctant to take more action.”
The actions of the Hennepin investigators were “repetitive and appeared to be in deliberate violation of regulations,” he added. “The case for the FDA disqualifying the HCMC researchers is overwhelming. The FDA’s slap-on-the-wrist approach to such appalling regulatory and ethical violations risks emboldening other researchers to disregard the rights and welfare of human subjects.”
Carl Elliott, MD, PhD, a bioethicist at the University of Minnesota, Minneapolis, agrees that the researcher from HCMC should be disqualified. “They didn’t just conduct risky, exploitative studies – they conducted them after the FDA had warned them not to proceed,” he said. “The message sent by this slap on the wrist is that investigators can do whatever they want to nonconsenting subjects, and the FDA will look the other way.”
Initial complaint
Public Citizen initially filed a complaint with the FDA in 2018, after learning that researchers affiliated with HCMC were conducting high-risk clinical trials involving ketamine to control agitation outside of the hospital setting. The complaint was cosigned by 64 doctors, bioethicists, and academic researchers and was also submitted to the Office for Human Research Protections.
The FDA typically allows investigational drugs to be used in emergency situation without obtaining informed consent if the therapies are known to carry a minimal risk. The IRB at HCMC had determined that this was the case with ketamine and approved the trials.
But according to Public Citizen’s complaint, prior research had suggested that ketamine could cause more complications and severe adverse events, compared with other sedatives.
The trials were conducted between 2014 and 2018, and in its letter, Public Citizen alleged that the investigators and the IRB had allowed these trials to proceed without obtaining informed consent from patients. The goal was to evaluate how well ketamine worked, compared with other drugs in calming agitated individuals: “The patients were given either ketamine or haloperidol for agitation by paramedics who responded to medical emergencies, and the goal was to see which drug worked faster,” said Dr. Carome. “Patients were only notified afterwards that they had received a sedative. Informed consent had been waived by IRB.”
In the first clinical trial conducted by HCMC, published in 2016, the researchers had hypothesized that 5 mg/kg of intramuscular ketamine would be superior to 10 mg of intramuscular haloperidol for severe prehospital agitation. Time to adequate sedation was the primary outcome measure. The study included 146 people; 64 received ketamine and 82 received haloperidol. They found that ketamine worked far more quickly than haloperidol (5 minutes vs. 17 minutes) but that the risk for complications was much higher. Complications occurred in 49% of patients receiving ketamine, compared with 5%.
“There was a 10-fold risk of adverse events,” said Dr. Carome. “And 39% of patients given ketamine had respiratory problems requiring intubation, compared to 4% who received haloperidol.”
A second study was launched in 2017, wherein ketamine was compared with midazolam in agitated patients. During the first 6-month period of the study, individuals would receive a ketamine-based protocol for prehospital agitation, and during the second 6 months, that would switch to midazolam. However, the study was halted in June 2018 after the local newspaper, the Star Tribune, reported that the city police had encouraged medical personnel to sedate agitated patients. This included individuals who had already been physically restrained.
The report stated that “in many cases, the individual being detained or arrested was not only handcuffed but strapped down on a stretcher in an ambulance before receiving ketamine,” and that it raised a “concerning question” over why these people were given the drug before they were transported to the hospital, “given the immediate effects on breathing and heart function that the drug induces.”
Along with halting the trial, HCMC asked for a review of cases involving its paramedics; an independent investigation led by former U.S. Deputy Attorney General Sally Yates was initiated to assess whether the Minneapolis police had crossed a line and urged paramedics to use ketamine.
“The decision to use ketamine was based on the study’s timeline and not on clinical judgment,” said Dr. Carome.
The FDA acknowledged receipt of the complaint and inspected the IRB records and the clinical trial data. Preliminary reports received by Public Citizen confirmed their allegations. “There were not appropriate protections for vulnerable subjects,” he said. “In 2019, the FDA did further investigations, and those reports had similar findings.”
FDA letters
The FDA had sent warning letters to Dr. Cole and Dr. Klein, citing them for ignoring federal safety laws in experimental research on the public. In their investigations, the FDA cited “objectionable conditions” for the studies led by Dr. Cole and Dr. Klein, according to the letters. Both researchers seemingly ignored FDA regulations and used practices that subjected patients to “significantly increased risk,” and the hospital defended its research with “factually incorrect” statements.
In a letter to Dr. Cole, the FDA noted that he never filed INDs for the trials with the FDA, as required by law, and that he also failed to write appropriate protocols to ensure that children and pregnant women were not enrolled in the research. Individuals under the influence of intoxicants also were not excluded, though the use of ketamine is cautioned in this population.
“Administration of the investigational drugs to these subjects placed them at significantly increased risk of the adverse events associated with the investigational products and decreased the acceptability of those risks,” the FDA said in its letter. “Your failure to exclude, and the lack of any precautions for, subjects under the influence of various intoxicants significantly increased the risks and/or decreased the acceptability of the risks associated with the investigational drugs.”
However, Dr. Cole conducted both studies in the prehospital setting and failed to initiate any specific measures to protect study participants, according to the FDA.
Petition denied
Dr. Carome noted that the researchers had committed repetitive egregious regulatory violations over a 4-year period, which were documented by the FDA in their warning letters to Dr. Cole and Dr. Klein. “We felt that they were so egregious that we need to send a signal to the community that this sort of behavior will not be tolerated,” he said. “The FDA denied our petition, and we think that sends the wrong signal to the research community.”
In their response, the FDA noted that as with judicial enforcement, “the Agency makes decisions regarding whether to pursue administrative enforcement action, including disqualification proceedings, on a case-by-case basis, considering all relevant facts and circumstances.” They added that at this time, they would not be taking further action against Dr. Cole and Dr. Klein.
“However, we intend to continue to consider all the options available to the Agency as we determine whether to pursue additional compliance actions related to this matter,” the FDA concluded.
The FDA declined to comment further on their decision.
Dr. Cole also declined to comment, but Hennepin Healthcare told this news organization that the “decision by the FDA to deny the petition validates the changes we made to strengthen and improve the clinical research program across the institution since the closing of the studies in 2018. We look forward to continuing to work with the FDA to ensure full compliance with the standards in place to protect research subjects.”
A version of this article first appeared on Medscape.com.
TNF placental transfer makes little difference in offspring infections
COPENHAGEN – Here’s reassuring news for pregnant women with rheumatic diseases treated with tumor necrosis factor (TNF)–alpha inhibitors: Although the drugs vary widely in their transmissibility across the placenta, there appears to be no excess risk for serious infections in children exposed in utero to TNF inhibitors with high, compared with low, placental transfer.
That’s according to investigators at McGill University in Montreal, who studied outcomes for nearly 3,000 infants who were exposed to TNF inhibitors during gestation.
“Our data are reassuring as we saw no strong signal, which suggests that there is no need to switch the mother’s drugs. More studies are needed, but this is a step in the right direction to reduce maternal stress and reassure physicians,” said Leah K. Flatman, MSc, a PhD candidate in epidemiology at McGill.
Ms. Flatman presented the findings in an oral abstract session at the annual European Congress of Rheumatology.
Not without risks
Approximately 20% of pregnant women with chronic inflammatory diseases are prescribed a TNF inhibitor, a class of drug that is effective for disease control but also increases risk for infection because of immunosuppressive effects.
“Similarly, offspring exposed in utero to TNF inhibitors may also experience immunosuppression and subsequent serious infections in their first year of life. This is the result of the TNF inhibitor entering the fetal bloodstream at different concentrations,» Ms. Flatman said.
Anti-TNF monoclonal immunoglobulins, such as infliximab (Remicade and biosimilars), adalimumab (Humira and biosimilars), and golimumab (Simponi) have the highest placental transfer, reaching higher levels in fetal circulation than in maternal circulation, she noted.
In contrast, certolizumab (Cimzia), a pegylated humanized antigen-binding fragment, and etanercept (Enbrel and biosimilars), a fusion protein, have the lowest placental penetration, Ms. Flatman said.
Population study
The investigators conducted a population cohort study using the IBM MarketScan database of commercial claims from employer-provided health insurance plans in the United States.
They looked at data on offspring of mothers with rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis and/or inflammatory bowel diseases (IBD; Crohn’s disease, and ulcerative colitis). The children were born from Jan. 1, 2011 through Dec. 31, 2019.
The exposure was at least one filled prescription and/or infusion procedure claim for TNF inhibitors in the 6 months before delivery. The exposures were divided into high and low placental-transfer categories.
A total of 26,088 offspring were identified, of whom 2,902 (11.1%) were exposed to a TNF inhibitor in utero. A little more than half of these children were born to mothers treated with TNF inhibitors for IBD.
For the primary outcome of serious infections (based on at least one hospitalization with infection in the first year of life), the investigators plotted Kaplan-Meier curves, which showed that the survival probability of serious infections in the high and low groups overlapped, indicating no large differences.
Of 2,105 offspring of mothers treated with a high–placental-transfer drug, 38 (1.8%) had serious infections, compared with 10 of 797 offspring (1.3%) of mothers who received low–placental-transfer drugs.
In multivariable analysis that controlled for maternal age at delivery, any RA diagnosis without an IBD diagnosis, and IBD diagnosis, gestational or pregestational diabetes, maternal asthma, preterm delivery, corticosteroid use, and disease-modifying antirheumatic drug use, the investigators saw that the hazard ratio for risk for serious infection in the high–, compared with the low–placental-transfer group was 1.20, with a confidence interval crossing 1, indicating nonsignificance.
Similar results reported
Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented, told this news organization that the findings were in line with those of a recent meta-analysis looking at the safety of biologic agents in pregnant women with IBD.
She added, however, that although the meta-analysis also showed little difference in outcomes for the children of women treated with high– compared with low–placental-transfer drugs, “we need more data to be sure about this.”
Comoderator Gabriela Riemekasten, MD, director of the clinic for rheumatology and clinical immunology at University Hospital in Lübeck, Germany, told this news organization that she was surprised to see that more women received high– than low–placental-transfer drugs.
Although there was a 20% difference between the groups, the numbers were relatively low, and “I would consider this in my practice and give my patients the advice of these data,” she said.
The study was supported by an Arthritis Society PhD Salary Award, and a Canadian Institutes of Health Project grant. Ms. Flatman, Dr. Förger, and Dr. Riemekasten reported having no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
COPENHAGEN – Here’s reassuring news for pregnant women with rheumatic diseases treated with tumor necrosis factor (TNF)–alpha inhibitors: Although the drugs vary widely in their transmissibility across the placenta, there appears to be no excess risk for serious infections in children exposed in utero to TNF inhibitors with high, compared with low, placental transfer.
That’s according to investigators at McGill University in Montreal, who studied outcomes for nearly 3,000 infants who were exposed to TNF inhibitors during gestation.
“Our data are reassuring as we saw no strong signal, which suggests that there is no need to switch the mother’s drugs. More studies are needed, but this is a step in the right direction to reduce maternal stress and reassure physicians,” said Leah K. Flatman, MSc, a PhD candidate in epidemiology at McGill.
Ms. Flatman presented the findings in an oral abstract session at the annual European Congress of Rheumatology.
Not without risks
Approximately 20% of pregnant women with chronic inflammatory diseases are prescribed a TNF inhibitor, a class of drug that is effective for disease control but also increases risk for infection because of immunosuppressive effects.
“Similarly, offspring exposed in utero to TNF inhibitors may also experience immunosuppression and subsequent serious infections in their first year of life. This is the result of the TNF inhibitor entering the fetal bloodstream at different concentrations,» Ms. Flatman said.
Anti-TNF monoclonal immunoglobulins, such as infliximab (Remicade and biosimilars), adalimumab (Humira and biosimilars), and golimumab (Simponi) have the highest placental transfer, reaching higher levels in fetal circulation than in maternal circulation, she noted.
In contrast, certolizumab (Cimzia), a pegylated humanized antigen-binding fragment, and etanercept (Enbrel and biosimilars), a fusion protein, have the lowest placental penetration, Ms. Flatman said.
Population study
The investigators conducted a population cohort study using the IBM MarketScan database of commercial claims from employer-provided health insurance plans in the United States.
They looked at data on offspring of mothers with rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis and/or inflammatory bowel diseases (IBD; Crohn’s disease, and ulcerative colitis). The children were born from Jan. 1, 2011 through Dec. 31, 2019.
The exposure was at least one filled prescription and/or infusion procedure claim for TNF inhibitors in the 6 months before delivery. The exposures were divided into high and low placental-transfer categories.
A total of 26,088 offspring were identified, of whom 2,902 (11.1%) were exposed to a TNF inhibitor in utero. A little more than half of these children were born to mothers treated with TNF inhibitors for IBD.
For the primary outcome of serious infections (based on at least one hospitalization with infection in the first year of life), the investigators plotted Kaplan-Meier curves, which showed that the survival probability of serious infections in the high and low groups overlapped, indicating no large differences.
Of 2,105 offspring of mothers treated with a high–placental-transfer drug, 38 (1.8%) had serious infections, compared with 10 of 797 offspring (1.3%) of mothers who received low–placental-transfer drugs.
In multivariable analysis that controlled for maternal age at delivery, any RA diagnosis without an IBD diagnosis, and IBD diagnosis, gestational or pregestational diabetes, maternal asthma, preterm delivery, corticosteroid use, and disease-modifying antirheumatic drug use, the investigators saw that the hazard ratio for risk for serious infection in the high–, compared with the low–placental-transfer group was 1.20, with a confidence interval crossing 1, indicating nonsignificance.
Similar results reported
Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented, told this news organization that the findings were in line with those of a recent meta-analysis looking at the safety of biologic agents in pregnant women with IBD.
She added, however, that although the meta-analysis also showed little difference in outcomes for the children of women treated with high– compared with low–placental-transfer drugs, “we need more data to be sure about this.”
Comoderator Gabriela Riemekasten, MD, director of the clinic for rheumatology and clinical immunology at University Hospital in Lübeck, Germany, told this news organization that she was surprised to see that more women received high– than low–placental-transfer drugs.
Although there was a 20% difference between the groups, the numbers were relatively low, and “I would consider this in my practice and give my patients the advice of these data,” she said.
The study was supported by an Arthritis Society PhD Salary Award, and a Canadian Institutes of Health Project grant. Ms. Flatman, Dr. Förger, and Dr. Riemekasten reported having no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
COPENHAGEN – Here’s reassuring news for pregnant women with rheumatic diseases treated with tumor necrosis factor (TNF)–alpha inhibitors: Although the drugs vary widely in their transmissibility across the placenta, there appears to be no excess risk for serious infections in children exposed in utero to TNF inhibitors with high, compared with low, placental transfer.
That’s according to investigators at McGill University in Montreal, who studied outcomes for nearly 3,000 infants who were exposed to TNF inhibitors during gestation.
“Our data are reassuring as we saw no strong signal, which suggests that there is no need to switch the mother’s drugs. More studies are needed, but this is a step in the right direction to reduce maternal stress and reassure physicians,” said Leah K. Flatman, MSc, a PhD candidate in epidemiology at McGill.
Ms. Flatman presented the findings in an oral abstract session at the annual European Congress of Rheumatology.
Not without risks
Approximately 20% of pregnant women with chronic inflammatory diseases are prescribed a TNF inhibitor, a class of drug that is effective for disease control but also increases risk for infection because of immunosuppressive effects.
“Similarly, offspring exposed in utero to TNF inhibitors may also experience immunosuppression and subsequent serious infections in their first year of life. This is the result of the TNF inhibitor entering the fetal bloodstream at different concentrations,» Ms. Flatman said.
Anti-TNF monoclonal immunoglobulins, such as infliximab (Remicade and biosimilars), adalimumab (Humira and biosimilars), and golimumab (Simponi) have the highest placental transfer, reaching higher levels in fetal circulation than in maternal circulation, she noted.
In contrast, certolizumab (Cimzia), a pegylated humanized antigen-binding fragment, and etanercept (Enbrel and biosimilars), a fusion protein, have the lowest placental penetration, Ms. Flatman said.
Population study
The investigators conducted a population cohort study using the IBM MarketScan database of commercial claims from employer-provided health insurance plans in the United States.
They looked at data on offspring of mothers with rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis and/or inflammatory bowel diseases (IBD; Crohn’s disease, and ulcerative colitis). The children were born from Jan. 1, 2011 through Dec. 31, 2019.
The exposure was at least one filled prescription and/or infusion procedure claim for TNF inhibitors in the 6 months before delivery. The exposures were divided into high and low placental-transfer categories.
A total of 26,088 offspring were identified, of whom 2,902 (11.1%) were exposed to a TNF inhibitor in utero. A little more than half of these children were born to mothers treated with TNF inhibitors for IBD.
For the primary outcome of serious infections (based on at least one hospitalization with infection in the first year of life), the investigators plotted Kaplan-Meier curves, which showed that the survival probability of serious infections in the high and low groups overlapped, indicating no large differences.
Of 2,105 offspring of mothers treated with a high–placental-transfer drug, 38 (1.8%) had serious infections, compared with 10 of 797 offspring (1.3%) of mothers who received low–placental-transfer drugs.
In multivariable analysis that controlled for maternal age at delivery, any RA diagnosis without an IBD diagnosis, and IBD diagnosis, gestational or pregestational diabetes, maternal asthma, preterm delivery, corticosteroid use, and disease-modifying antirheumatic drug use, the investigators saw that the hazard ratio for risk for serious infection in the high–, compared with the low–placental-transfer group was 1.20, with a confidence interval crossing 1, indicating nonsignificance.
Similar results reported
Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented, told this news organization that the findings were in line with those of a recent meta-analysis looking at the safety of biologic agents in pregnant women with IBD.
She added, however, that although the meta-analysis also showed little difference in outcomes for the children of women treated with high– compared with low–placental-transfer drugs, “we need more data to be sure about this.”
Comoderator Gabriela Riemekasten, MD, director of the clinic for rheumatology and clinical immunology at University Hospital in Lübeck, Germany, told this news organization that she was surprised to see that more women received high– than low–placental-transfer drugs.
Although there was a 20% difference between the groups, the numbers were relatively low, and “I would consider this in my practice and give my patients the advice of these data,” she said.
The study was supported by an Arthritis Society PhD Salary Award, and a Canadian Institutes of Health Project grant. Ms. Flatman, Dr. Förger, and Dr. Riemekasten reported having no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
THE EULAR 2022 CONGRESS
‘Encouraging’ results of baricitinib in juvenile idiopathic arthritis
COPENHAGEN – Baricitinib (Olumiant), a Janus kinase (JAK) inhibitor, significantly increases time to disease flare and decreases frequency of flares in patients with juvenile idiopathic arthritis (JIA), according to the results of a phase 3, placebo-controlled study.
The results support use of baricitinib when biologic or conventional synthetic disease-modifying antirheumatic drugs (DMARDs) fail.
The difference in the proportion of patients who flared between baricitinib and placebo was seen as soon as 4 weeks after half of the patients switched from active drug to placebo, at 3.7% versus 23.5% respectively, reported Athimalaipet Ramanan, MD, from the University of Bristol (England) who presented the findings of the withdrawal, efficacy, and safety study at the annual European Congress of Rheumatology.
“Our patients and parents have been waiting for alternative drugs for JIA, so JAK inhibitors have come at the right time,” he said. “These are really very encouraging findings for families, caregivers, and patients with JIA, to have an effective oral JAK inhibitor for managing these children.”
In reporting the key findings, Dr. Ramanan added that the majority of patients (76%) achieved a JIA-ACR (American College of Rheumatology) 30 score during the 12-week open-label phase and went on to enter the double-blind withdrawal phase of the trial.
Baricitinib 2-mg tablets are already Food and Drug Administration approved for the treatment of adults with moderately to severely active rheumatoid arthritis. This study, sponsored by the drug manufacturer Eli Lilly, aimed to investigate the efficacy and safety in pediatric patients with JIA who have shown an inadequate response to conventional synthetic or biologic DMARDs.
“For juvenile patients we need to make a dose adjustment [from the adult dosing], especially because we don’t have long-term safety data from JAK inhibitors in general,” said Osama Elfayad, MD, rheumatologist from Mouwasat Hospital, Dammam, Saudi Arabia who attended the presentation and commented on the findings.
He emphasized that safety was of primary concern in the pediatric population who have a long life expectancy. “For me it is essential to have good long-term safety data in juvenile patients. If we start with 4 mg and if the patient is controlled, we should shift to 2 mg which will be much better. I understand some clinicians are asking for 1 mg.”
Study details
The study population included patients aged from 2 to 17 years old with extended oligo- or polyarticular JIA, enthesitis-related juvenile idiopathic arthritis (ERA) and juvenile psoriatic arthritis.
The trial was divided into three periods: a 2-week safety assessment, a 12-week open-label lead-in phase, and an up-to 32-week double-blind withdrawal phase. After confirmation of dose and safety, children were enrolled in the open-label phase receiving age-based, oral, once daily doses of baricitinib.
“The primary endpoint is really concerned with the next phase of the study [double-blind withdrawal phase] looking at the proportion of patients who have shown a response at week 12 [achieved JIA-ACR30] but when switched from active drug to placebo have a flare,” explained Dr. Ramanan.
Patients were randomized 1:1 to continuing baricitinib or newly starting placebo until disease flare or up to week 32. The time to flare during the double-blind phase was the primary endpoint, while secondary endpoints included JIA-ACR30/50/70/90 response rates at week 12, and the proportion of patients with a flare during the double-blind phase.
“These secondary endpoints are more relevant to the clinic,” noted Dr. Ramanan.
A total of 219 patients entered the open-label phase, and of these, 163 achieved a JIA-ACR 30. These 163 children entered the double-blind stage and were randomized to baricitinib four times a day (56 completed), or placebo (32 completed).
Two-thirds of patients were female, which is typical of the disease, explained Dr. Ramanan, and over two-thirds were White. “Most patients had had disease for around 4 years, and about half had had prior biologic therapy. About half were on baseline methotrexate and almost one-third had used corticosteroids although at doses of under 0.2mg/kg.
“It’s gratifying to see that over 75% achieved a JIA-ACR 30 [76.3%]. More importantly, two-thirds of the patients have a JIA-ACR 50 [63.5%], and almost half of the patients have a JIA-ACR 70 [46.1%]. This is pretty significant at 12 weeks only,” he remarked.
The key finding, however, was in the withdrawal phase, said Dr. Ramanan. “We see that those patients who had a response at week 12 and were then switched to placebo, about half [50.6%] flared on placebo, compared to only 17% of those who continued with baricitinib. So not only do those who switch to placebo have a higher frequency of flares but they are more likely to flare quickly, as early as 4 weeks.”
With respect to safety, he said: “This shows short-term safety, but what we really need is medium and long-term safety data. It is no surprise that most of the events seen were as expected in children including nasopharyngitis, upper respiratory tract infections, and nausea.”
In the baricitinib versus placebo phase, 4.9% had serious adverse events in the baricitinib group compared to 3.7% in the placebo group. “There was nothing we didn’t expect to see which was mainly infection,” said Dr. Ramanan.
Dr. Elfayad has no disclosures. Professor Ramanan is a consultant for Eli Lilly, Abbvie, Roche, UCB, Novartis, Pfizer, and Sobi. He has received grant/research support from Eli Lilly.
COPENHAGEN – Baricitinib (Olumiant), a Janus kinase (JAK) inhibitor, significantly increases time to disease flare and decreases frequency of flares in patients with juvenile idiopathic arthritis (JIA), according to the results of a phase 3, placebo-controlled study.
The results support use of baricitinib when biologic or conventional synthetic disease-modifying antirheumatic drugs (DMARDs) fail.
The difference in the proportion of patients who flared between baricitinib and placebo was seen as soon as 4 weeks after half of the patients switched from active drug to placebo, at 3.7% versus 23.5% respectively, reported Athimalaipet Ramanan, MD, from the University of Bristol (England) who presented the findings of the withdrawal, efficacy, and safety study at the annual European Congress of Rheumatology.
“Our patients and parents have been waiting for alternative drugs for JIA, so JAK inhibitors have come at the right time,” he said. “These are really very encouraging findings for families, caregivers, and patients with JIA, to have an effective oral JAK inhibitor for managing these children.”
In reporting the key findings, Dr. Ramanan added that the majority of patients (76%) achieved a JIA-ACR (American College of Rheumatology) 30 score during the 12-week open-label phase and went on to enter the double-blind withdrawal phase of the trial.
Baricitinib 2-mg tablets are already Food and Drug Administration approved for the treatment of adults with moderately to severely active rheumatoid arthritis. This study, sponsored by the drug manufacturer Eli Lilly, aimed to investigate the efficacy and safety in pediatric patients with JIA who have shown an inadequate response to conventional synthetic or biologic DMARDs.
“For juvenile patients we need to make a dose adjustment [from the adult dosing], especially because we don’t have long-term safety data from JAK inhibitors in general,” said Osama Elfayad, MD, rheumatologist from Mouwasat Hospital, Dammam, Saudi Arabia who attended the presentation and commented on the findings.
He emphasized that safety was of primary concern in the pediatric population who have a long life expectancy. “For me it is essential to have good long-term safety data in juvenile patients. If we start with 4 mg and if the patient is controlled, we should shift to 2 mg which will be much better. I understand some clinicians are asking for 1 mg.”
Study details
The study population included patients aged from 2 to 17 years old with extended oligo- or polyarticular JIA, enthesitis-related juvenile idiopathic arthritis (ERA) and juvenile psoriatic arthritis.
The trial was divided into three periods: a 2-week safety assessment, a 12-week open-label lead-in phase, and an up-to 32-week double-blind withdrawal phase. After confirmation of dose and safety, children were enrolled in the open-label phase receiving age-based, oral, once daily doses of baricitinib.
“The primary endpoint is really concerned with the next phase of the study [double-blind withdrawal phase] looking at the proportion of patients who have shown a response at week 12 [achieved JIA-ACR30] but when switched from active drug to placebo have a flare,” explained Dr. Ramanan.
Patients were randomized 1:1 to continuing baricitinib or newly starting placebo until disease flare or up to week 32. The time to flare during the double-blind phase was the primary endpoint, while secondary endpoints included JIA-ACR30/50/70/90 response rates at week 12, and the proportion of patients with a flare during the double-blind phase.
“These secondary endpoints are more relevant to the clinic,” noted Dr. Ramanan.
A total of 219 patients entered the open-label phase, and of these, 163 achieved a JIA-ACR 30. These 163 children entered the double-blind stage and were randomized to baricitinib four times a day (56 completed), or placebo (32 completed).
Two-thirds of patients were female, which is typical of the disease, explained Dr. Ramanan, and over two-thirds were White. “Most patients had had disease for around 4 years, and about half had had prior biologic therapy. About half were on baseline methotrexate and almost one-third had used corticosteroids although at doses of under 0.2mg/kg.
“It’s gratifying to see that over 75% achieved a JIA-ACR 30 [76.3%]. More importantly, two-thirds of the patients have a JIA-ACR 50 [63.5%], and almost half of the patients have a JIA-ACR 70 [46.1%]. This is pretty significant at 12 weeks only,” he remarked.
The key finding, however, was in the withdrawal phase, said Dr. Ramanan. “We see that those patients who had a response at week 12 and were then switched to placebo, about half [50.6%] flared on placebo, compared to only 17% of those who continued with baricitinib. So not only do those who switch to placebo have a higher frequency of flares but they are more likely to flare quickly, as early as 4 weeks.”
With respect to safety, he said: “This shows short-term safety, but what we really need is medium and long-term safety data. It is no surprise that most of the events seen were as expected in children including nasopharyngitis, upper respiratory tract infections, and nausea.”
In the baricitinib versus placebo phase, 4.9% had serious adverse events in the baricitinib group compared to 3.7% in the placebo group. “There was nothing we didn’t expect to see which was mainly infection,” said Dr. Ramanan.
Dr. Elfayad has no disclosures. Professor Ramanan is a consultant for Eli Lilly, Abbvie, Roche, UCB, Novartis, Pfizer, and Sobi. He has received grant/research support from Eli Lilly.
COPENHAGEN – Baricitinib (Olumiant), a Janus kinase (JAK) inhibitor, significantly increases time to disease flare and decreases frequency of flares in patients with juvenile idiopathic arthritis (JIA), according to the results of a phase 3, placebo-controlled study.
The results support use of baricitinib when biologic or conventional synthetic disease-modifying antirheumatic drugs (DMARDs) fail.
The difference in the proportion of patients who flared between baricitinib and placebo was seen as soon as 4 weeks after half of the patients switched from active drug to placebo, at 3.7% versus 23.5% respectively, reported Athimalaipet Ramanan, MD, from the University of Bristol (England) who presented the findings of the withdrawal, efficacy, and safety study at the annual European Congress of Rheumatology.
“Our patients and parents have been waiting for alternative drugs for JIA, so JAK inhibitors have come at the right time,” he said. “These are really very encouraging findings for families, caregivers, and patients with JIA, to have an effective oral JAK inhibitor for managing these children.”
In reporting the key findings, Dr. Ramanan added that the majority of patients (76%) achieved a JIA-ACR (American College of Rheumatology) 30 score during the 12-week open-label phase and went on to enter the double-blind withdrawal phase of the trial.
Baricitinib 2-mg tablets are already Food and Drug Administration approved for the treatment of adults with moderately to severely active rheumatoid arthritis. This study, sponsored by the drug manufacturer Eli Lilly, aimed to investigate the efficacy and safety in pediatric patients with JIA who have shown an inadequate response to conventional synthetic or biologic DMARDs.
“For juvenile patients we need to make a dose adjustment [from the adult dosing], especially because we don’t have long-term safety data from JAK inhibitors in general,” said Osama Elfayad, MD, rheumatologist from Mouwasat Hospital, Dammam, Saudi Arabia who attended the presentation and commented on the findings.
He emphasized that safety was of primary concern in the pediatric population who have a long life expectancy. “For me it is essential to have good long-term safety data in juvenile patients. If we start with 4 mg and if the patient is controlled, we should shift to 2 mg which will be much better. I understand some clinicians are asking for 1 mg.”
Study details
The study population included patients aged from 2 to 17 years old with extended oligo- or polyarticular JIA, enthesitis-related juvenile idiopathic arthritis (ERA) and juvenile psoriatic arthritis.
The trial was divided into three periods: a 2-week safety assessment, a 12-week open-label lead-in phase, and an up-to 32-week double-blind withdrawal phase. After confirmation of dose and safety, children were enrolled in the open-label phase receiving age-based, oral, once daily doses of baricitinib.
“The primary endpoint is really concerned with the next phase of the study [double-blind withdrawal phase] looking at the proportion of patients who have shown a response at week 12 [achieved JIA-ACR30] but when switched from active drug to placebo have a flare,” explained Dr. Ramanan.
Patients were randomized 1:1 to continuing baricitinib or newly starting placebo until disease flare or up to week 32. The time to flare during the double-blind phase was the primary endpoint, while secondary endpoints included JIA-ACR30/50/70/90 response rates at week 12, and the proportion of patients with a flare during the double-blind phase.
“These secondary endpoints are more relevant to the clinic,” noted Dr. Ramanan.
A total of 219 patients entered the open-label phase, and of these, 163 achieved a JIA-ACR 30. These 163 children entered the double-blind stage and were randomized to baricitinib four times a day (56 completed), or placebo (32 completed).
Two-thirds of patients were female, which is typical of the disease, explained Dr. Ramanan, and over two-thirds were White. “Most patients had had disease for around 4 years, and about half had had prior biologic therapy. About half were on baseline methotrexate and almost one-third had used corticosteroids although at doses of under 0.2mg/kg.
“It’s gratifying to see that over 75% achieved a JIA-ACR 30 [76.3%]. More importantly, two-thirds of the patients have a JIA-ACR 50 [63.5%], and almost half of the patients have a JIA-ACR 70 [46.1%]. This is pretty significant at 12 weeks only,” he remarked.
The key finding, however, was in the withdrawal phase, said Dr. Ramanan. “We see that those patients who had a response at week 12 and were then switched to placebo, about half [50.6%] flared on placebo, compared to only 17% of those who continued with baricitinib. So not only do those who switch to placebo have a higher frequency of flares but they are more likely to flare quickly, as early as 4 weeks.”
With respect to safety, he said: “This shows short-term safety, but what we really need is medium and long-term safety data. It is no surprise that most of the events seen were as expected in children including nasopharyngitis, upper respiratory tract infections, and nausea.”
In the baricitinib versus placebo phase, 4.9% had serious adverse events in the baricitinib group compared to 3.7% in the placebo group. “There was nothing we didn’t expect to see which was mainly infection,” said Dr. Ramanan.
Dr. Elfayad has no disclosures. Professor Ramanan is a consultant for Eli Lilly, Abbvie, Roche, UCB, Novartis, Pfizer, and Sobi. He has received grant/research support from Eli Lilly.
AT THE EULAR 2022 CONGRESS
Think it’s ILD? Tell it to the machines
SAN FRANCISCO – Interstitial lung disease is a difficult diagnosis to make, but a combination of artificial intelligence (AI) techniques and automated language processing could help clinicians identify the early signs of ILD and start patients on therapy, investigators say.
For example, applying an AI algorithm to spirometry readings taken from patients whose data were registered in the UK Biobank identified 27% as having ILD, and of this group, 66% had ostensibly normal lung function on spirometry but were later diagnosed with ILD, reported Marko Topalovic, PhD, from the AI company ArtiQ in Leuven, Belgium, at the American Thoracic Society’s international conference.
“A diagnosis of ILD is very challenging, so you have patients who are going to be misdiagnosed or have a very late diagnosis, so we aimed to apply our AI algorithm on spirometry to see whether we could detect ILD much earlier,” he said in an interview conducted during a poster discussion session.
AI detected ILD up to 6.8 years before a clinician’s diagnosis, Dr. Topalovic said.
Reading between the lines
In a separate study, investigators at the University of California, Davis, used language analysis software to scour electronic health records for words indicative of early ILD, and found that the technique dramatically shortened the median time to a pulmonary referral, compared with historical controls.
“This is a language processing program that can essentially look through the radiology reports and look for the key words that often describe interstitial lung disease, like traction, honeycomb, fibrotic, etc. With those studies being flagged, an actual pulmonologist will then further review the scan, and see whether it meets criteria for one of the interstitial lung diseases,” lead author William Leon, MD, a resident in the department of internal medicine at the University of California, Davis, said in an interview.
“We then sent the primary care doctor a message to say: ‘Hey, this patient has ILD. You need to send them to a pulmonologist,’ ” he added.
Putting it together
Philip L. Molyneaux, MRCP (UK), MBBS, BS (Hons), from Imperial College London, who comoderated the session but was not involved in the studies, speculated that combining these and other, nontechnical interventions also discussed could help to improve diagnosis of ILD and allow clinicians to prescribe therapy earlier in the disease course.
“What’s going to give you the biggest impact for patients? Everyone working individually is coming up with great advances, and if you put them all together it’s going to provide much greater benefit for our patients,” he said in an interview.
AI Spirometry details
In collaboration with colleagues at the Laboratory of Respiratory Disease at University Hospital in Leuven, Dr. Topalovic applied AI to results of spirometry performed prior to diagnosis of ILD among 109 patients registered in the UK Biobank, a repository of information on more than 500,000 volunteers.
The patients selected had ILD listed as their cause of death, had spirometry performed up to 7 years before their deaths, and did not receive a diagnosis of ILD on the day of the index spirometry.
In all 73% of patients were men, 27% women, with an average age of 64.6 years. A large majority of the sample (77.15%) had a history of smoking, and 60 of the patients (55%) died within one year of an ILD diagnosis.
The investigators plugged the spirometry data and each patients demographic information – including gender, age, height, weight, race, and smoking status – into the AI clinical decision support program, which yielded a statistical probability for each subject of having normal lung function, asthma, COPD, ILD, another obstructive disease, or another unidentifiable respiratory disease.
In 29 patients (27%) the software listed ILD as the highest probability, and of this group 19 patients (66%) had normal lung function according to standard interpretation guidelines.
Spirometry parameters among patients identified as having probable ILD were different from those where ILD was not detected. For example, forced vital capacity (FVC) was 76% of predicted among patients with likely ILD versus 87% of predicted in those who had a diagnosis later (P = .003). Similar differences were seen in the forced expiratory volume in 1 second to FVC ratio, at 0.82 vs. 0.75, respectively (P = .007).
There were no differences in mortality or in median time between spirometry and clinician diagnosis between the groups.
Language processing details
Dr. Leon and colleagues used a language analysis software package to review CT chest reports. Reports were flagged if they contained the words traction, honeycomb, fibrotic, fibrosis, reticular, or reticulation.
The CT scan accompanying each flagged reported was reviewed by a pulmonologist for the presence of ILD, and scans with ILD identified were referred to pulmonary specialists. The results of 2,198 prospective scans followed by prospective screening were compared with those of 1,690 historical controls seen in 2015 and 2016.
The investigators found that 85 incident cases of ILD were identified in the historical controls, compared with 143 in the prospective cohort, leading to 38 and 120 pulmonary referrals, respectively.
For the primary outcome of median time from CT to pulmonary referral, the authors found that it was 1.27 months for the prospective cohort, compared with not reached (censored after 18 months) in historical controls.
The hazard ratio for a pulmonary referral in the prospective versus historical cohort was 2.79, an association that was strengthened after adjusting for sex, age, race, smoking pack-years, cough, crackles, and dyspnea (HR, 4.54; both comparisons significant according to confidence intervals).
The studies were internally funded. Dr. Topalovic is CEO and cofounder of ArtiQ. Dr. Leon and Dr. Molyneaux reported no relevant conflicts of interest.
SAN FRANCISCO – Interstitial lung disease is a difficult diagnosis to make, but a combination of artificial intelligence (AI) techniques and automated language processing could help clinicians identify the early signs of ILD and start patients on therapy, investigators say.
For example, applying an AI algorithm to spirometry readings taken from patients whose data were registered in the UK Biobank identified 27% as having ILD, and of this group, 66% had ostensibly normal lung function on spirometry but were later diagnosed with ILD, reported Marko Topalovic, PhD, from the AI company ArtiQ in Leuven, Belgium, at the American Thoracic Society’s international conference.
“A diagnosis of ILD is very challenging, so you have patients who are going to be misdiagnosed or have a very late diagnosis, so we aimed to apply our AI algorithm on spirometry to see whether we could detect ILD much earlier,” he said in an interview conducted during a poster discussion session.
AI detected ILD up to 6.8 years before a clinician’s diagnosis, Dr. Topalovic said.
Reading between the lines
In a separate study, investigators at the University of California, Davis, used language analysis software to scour electronic health records for words indicative of early ILD, and found that the technique dramatically shortened the median time to a pulmonary referral, compared with historical controls.
“This is a language processing program that can essentially look through the radiology reports and look for the key words that often describe interstitial lung disease, like traction, honeycomb, fibrotic, etc. With those studies being flagged, an actual pulmonologist will then further review the scan, and see whether it meets criteria for one of the interstitial lung diseases,” lead author William Leon, MD, a resident in the department of internal medicine at the University of California, Davis, said in an interview.
“We then sent the primary care doctor a message to say: ‘Hey, this patient has ILD. You need to send them to a pulmonologist,’ ” he added.
Putting it together
Philip L. Molyneaux, MRCP (UK), MBBS, BS (Hons), from Imperial College London, who comoderated the session but was not involved in the studies, speculated that combining these and other, nontechnical interventions also discussed could help to improve diagnosis of ILD and allow clinicians to prescribe therapy earlier in the disease course.
“What’s going to give you the biggest impact for patients? Everyone working individually is coming up with great advances, and if you put them all together it’s going to provide much greater benefit for our patients,” he said in an interview.
AI Spirometry details
In collaboration with colleagues at the Laboratory of Respiratory Disease at University Hospital in Leuven, Dr. Topalovic applied AI to results of spirometry performed prior to diagnosis of ILD among 109 patients registered in the UK Biobank, a repository of information on more than 500,000 volunteers.
The patients selected had ILD listed as their cause of death, had spirometry performed up to 7 years before their deaths, and did not receive a diagnosis of ILD on the day of the index spirometry.
In all 73% of patients were men, 27% women, with an average age of 64.6 years. A large majority of the sample (77.15%) had a history of smoking, and 60 of the patients (55%) died within one year of an ILD diagnosis.
The investigators plugged the spirometry data and each patients demographic information – including gender, age, height, weight, race, and smoking status – into the AI clinical decision support program, which yielded a statistical probability for each subject of having normal lung function, asthma, COPD, ILD, another obstructive disease, or another unidentifiable respiratory disease.
In 29 patients (27%) the software listed ILD as the highest probability, and of this group 19 patients (66%) had normal lung function according to standard interpretation guidelines.
Spirometry parameters among patients identified as having probable ILD were different from those where ILD was not detected. For example, forced vital capacity (FVC) was 76% of predicted among patients with likely ILD versus 87% of predicted in those who had a diagnosis later (P = .003). Similar differences were seen in the forced expiratory volume in 1 second to FVC ratio, at 0.82 vs. 0.75, respectively (P = .007).
There were no differences in mortality or in median time between spirometry and clinician diagnosis between the groups.
Language processing details
Dr. Leon and colleagues used a language analysis software package to review CT chest reports. Reports were flagged if they contained the words traction, honeycomb, fibrotic, fibrosis, reticular, or reticulation.
The CT scan accompanying each flagged reported was reviewed by a pulmonologist for the presence of ILD, and scans with ILD identified were referred to pulmonary specialists. The results of 2,198 prospective scans followed by prospective screening were compared with those of 1,690 historical controls seen in 2015 and 2016.
The investigators found that 85 incident cases of ILD were identified in the historical controls, compared with 143 in the prospective cohort, leading to 38 and 120 pulmonary referrals, respectively.
For the primary outcome of median time from CT to pulmonary referral, the authors found that it was 1.27 months for the prospective cohort, compared with not reached (censored after 18 months) in historical controls.
The hazard ratio for a pulmonary referral in the prospective versus historical cohort was 2.79, an association that was strengthened after adjusting for sex, age, race, smoking pack-years, cough, crackles, and dyspnea (HR, 4.54; both comparisons significant according to confidence intervals).
The studies were internally funded. Dr. Topalovic is CEO and cofounder of ArtiQ. Dr. Leon and Dr. Molyneaux reported no relevant conflicts of interest.
SAN FRANCISCO – Interstitial lung disease is a difficult diagnosis to make, but a combination of artificial intelligence (AI) techniques and automated language processing could help clinicians identify the early signs of ILD and start patients on therapy, investigators say.
For example, applying an AI algorithm to spirometry readings taken from patients whose data were registered in the UK Biobank identified 27% as having ILD, and of this group, 66% had ostensibly normal lung function on spirometry but were later diagnosed with ILD, reported Marko Topalovic, PhD, from the AI company ArtiQ in Leuven, Belgium, at the American Thoracic Society’s international conference.
“A diagnosis of ILD is very challenging, so you have patients who are going to be misdiagnosed or have a very late diagnosis, so we aimed to apply our AI algorithm on spirometry to see whether we could detect ILD much earlier,” he said in an interview conducted during a poster discussion session.
AI detected ILD up to 6.8 years before a clinician’s diagnosis, Dr. Topalovic said.
Reading between the lines
In a separate study, investigators at the University of California, Davis, used language analysis software to scour electronic health records for words indicative of early ILD, and found that the technique dramatically shortened the median time to a pulmonary referral, compared with historical controls.
“This is a language processing program that can essentially look through the radiology reports and look for the key words that often describe interstitial lung disease, like traction, honeycomb, fibrotic, etc. With those studies being flagged, an actual pulmonologist will then further review the scan, and see whether it meets criteria for one of the interstitial lung diseases,” lead author William Leon, MD, a resident in the department of internal medicine at the University of California, Davis, said in an interview.
“We then sent the primary care doctor a message to say: ‘Hey, this patient has ILD. You need to send them to a pulmonologist,’ ” he added.
Putting it together
Philip L. Molyneaux, MRCP (UK), MBBS, BS (Hons), from Imperial College London, who comoderated the session but was not involved in the studies, speculated that combining these and other, nontechnical interventions also discussed could help to improve diagnosis of ILD and allow clinicians to prescribe therapy earlier in the disease course.
“What’s going to give you the biggest impact for patients? Everyone working individually is coming up with great advances, and if you put them all together it’s going to provide much greater benefit for our patients,” he said in an interview.
AI Spirometry details
In collaboration with colleagues at the Laboratory of Respiratory Disease at University Hospital in Leuven, Dr. Topalovic applied AI to results of spirometry performed prior to diagnosis of ILD among 109 patients registered in the UK Biobank, a repository of information on more than 500,000 volunteers.
The patients selected had ILD listed as their cause of death, had spirometry performed up to 7 years before their deaths, and did not receive a diagnosis of ILD on the day of the index spirometry.
In all 73% of patients were men, 27% women, with an average age of 64.6 years. A large majority of the sample (77.15%) had a history of smoking, and 60 of the patients (55%) died within one year of an ILD diagnosis.
The investigators plugged the spirometry data and each patients demographic information – including gender, age, height, weight, race, and smoking status – into the AI clinical decision support program, which yielded a statistical probability for each subject of having normal lung function, asthma, COPD, ILD, another obstructive disease, or another unidentifiable respiratory disease.
In 29 patients (27%) the software listed ILD as the highest probability, and of this group 19 patients (66%) had normal lung function according to standard interpretation guidelines.
Spirometry parameters among patients identified as having probable ILD were different from those where ILD was not detected. For example, forced vital capacity (FVC) was 76% of predicted among patients with likely ILD versus 87% of predicted in those who had a diagnosis later (P = .003). Similar differences were seen in the forced expiratory volume in 1 second to FVC ratio, at 0.82 vs. 0.75, respectively (P = .007).
There were no differences in mortality or in median time between spirometry and clinician diagnosis between the groups.
Language processing details
Dr. Leon and colleagues used a language analysis software package to review CT chest reports. Reports were flagged if they contained the words traction, honeycomb, fibrotic, fibrosis, reticular, or reticulation.
The CT scan accompanying each flagged reported was reviewed by a pulmonologist for the presence of ILD, and scans with ILD identified were referred to pulmonary specialists. The results of 2,198 prospective scans followed by prospective screening were compared with those of 1,690 historical controls seen in 2015 and 2016.
The investigators found that 85 incident cases of ILD were identified in the historical controls, compared with 143 in the prospective cohort, leading to 38 and 120 pulmonary referrals, respectively.
For the primary outcome of median time from CT to pulmonary referral, the authors found that it was 1.27 months for the prospective cohort, compared with not reached (censored after 18 months) in historical controls.
The hazard ratio for a pulmonary referral in the prospective versus historical cohort was 2.79, an association that was strengthened after adjusting for sex, age, race, smoking pack-years, cough, crackles, and dyspnea (HR, 4.54; both comparisons significant according to confidence intervals).
The studies were internally funded. Dr. Topalovic is CEO and cofounder of ArtiQ. Dr. Leon and Dr. Molyneaux reported no relevant conflicts of interest.
AT ATS 2022
Surgeons, who see it up close, offer ways to stop gun violence
Their strategies can work regardless of where you stand on the Second Amendment of the Constitution, said Patricia Turner, MD. “Our proposals are embraced by both gun owners and non–gun owners alike, and we are unique in that regard.”
These “implementable solutions” could prevent the next massacre, Dr. Turner, executive director of the American College of Surgeons, said during a news briefing the group sponsored on June 2.
“Our future – indeed all of our futures – depend on our ability to find durable, actionable steps that we can implement tomorrow to save lives,” she said.
Firsthand perspective
“Sadly I’m here today as a trauma surgeon who has cared for two of the largest mass shootings in modern U.S. history,” said Ronald Stewart, MD, chair of the department of surgery at University Hospital in San Antonio, Texas.
Dr. Stewart treated victims of the 2017 Sutherland Springs First Baptist Church shooting – where 27 people died, including the shooter – and the recent Uvalde school shooting, both in Texas.
“The injuries inflicted by high-velocity weapons used at both of these attacks are horrific. A high-capacity, magazine-fed automatic rifle such as the AR-15 causes extremely destructive tissue wounds,” he said.
One of the group’s proposals is to increase the regulation of high-velocity weapons, including AR-15s.
“These wounds are horribly lethal at close range, and sadly, most victims do not survive long enough to make it to a trauma center,” Dr. Stewart said.
On a positive note, “all of our current [Uvalde] patients are improving, which really brings us joy in this dark time,” he said. “But all of them have a long road to deal with recovery with both the physical and emotional impact of their injuries.”
Jeffrey Kerby, MD, agreed.
“Trauma surgeons see the short-term physical effects of these injuries and watch patients struggle with the long-term impact of these wounds,” said Dr. Kerby, director of trauma and acute care surgery at the University of Alabama at Birmingham.
Surgeons feel ‘profound impact’ of shootings
“Firearm violence has a profound impact on surgeons, and we are the undisputed subject matter experts in treating the tragic results,” said Patrick Bailey, MD, medical director for advocacy at the American College of Surgeons.
“This impacts surgeons as well,” said Dr. Kerby, chair of the Committee on Trauma for the surgeons’ group. “We are human, and we can’t help but share in the grief, the pain, and the suffering that our patients endure.
“As a pediatric surgeon ... I have too often witnessed the impact of firearm violence, and obviously, the devastation extends beyond the victims to their families,” he said. “To put it succinctly, in our culture, parents are not supposed to be put in a position of burying their children.”
A public health crisis
“It’s important to recognize that we’ve been talking about a public health approach,” said Eileen Bulger, MD, acting chief of the trauma division at the University of Washington in Seattle. That strategy is important for engaging both firearm owners and communities that have a higher risk for firearm violence, she said.
A committee of the American College of Surgeons developed specific recommendations in 2018, which are still valid today. The group brought together surgeons from across the U.S. including “passionate firearm owners and experts in firearm safety,” Dr. Bulger said.
The committee, for example, agreed on 10 specific recommendations “that we believe are bipartisan and could have an immediate impact in saving lives.”
“I’m a lifelong gun owner,” Dr. Bailey said, emphasizing that the team’s process included participation and perspective from other surgeons “who, like me, are also gun owners, but gun owners who also seek to reduce the impact of firearm violence in our country.”
The recommendations address these areas:
- Gun ownership
- Firearm registration
- Licensure
- Education and training
- Ownership responsibilities
- Mandatory reporting and risk reduction
- Safety innovation and technology
- Research
- The culture of violence
- Social isolation and mental health
For example, “we currently have certain classes of weapons with significant offensive capability,” Dr. Bulger said, “that are appropriately restricted and regulated under the National Firearms Act as Class 3 weapons.”
This group includes fully automatic machine guns, explosive devices, and short-barrel shotguns.
“We recommend a formal reassessment of the firearms designated within each of these national firearms classifications,” Dr. Bulger said.
For example, high-capacity, magazine-fed semiautomatic rifles, such as the AR-15, should be considered for reclassification as NFA Class 3 firearms, or they should get a new designation with tighter regulation.
The ACS endorses formal firearm safety training for all new gun owners. Also, owners who do not provide reasonably safe firearm storage should be held responsible for events related to the discharge of their firearms, Dr. Bulger said. And people who are deemed an imminent threat to themselves or others through firearm ownership should be temporarily or permanently restricted, with due process.
Research and reporting reforms
The ACS is also calling for research on firearm injuries and firearm injury prevention to be federally funded, Dr. Bulger said. The research should be done in a nonpartisan manner, she said.
“We have concerns that the manner and tone in which information is released to the public may lead to copycat mass killers,” she said. “The ACS recommends that law enforcement officials and the press take steps to eliminate the notoriety of the shooter, for example.”
Dr. Bulger also addressed the mental health angle. “We encourage recognition of mental health warning signs and social isolation by teachers, counselors, peers, and parents.” When identified, immediate referral to professionals is needed.
In addition to these recommendations, another team from the American College of Surgeons has published an overview of ways to address the inequities that contribute to violence. “We advocate for federal funding to support the development of hospital-based and community programs for violence intervention and prevention,” Dr. Bulger said.
Dr. Bailey said that as a gun owner himself, he thinks other gun owners would support these recommendations.
“I do not believe that the steps recommended ... pose undue burden on the rights of individual gun owners,” he said.
The time is now
Most firearm injuries are not from mass shooting events, Dr. Kerby said.
“My own trauma center has seen a 40% increase in the number of firearm injuries just in the last 2 years,” he added, “and these numbers continue to grow.”
A version of this article first appeared on WebMD.com.
Their strategies can work regardless of where you stand on the Second Amendment of the Constitution, said Patricia Turner, MD. “Our proposals are embraced by both gun owners and non–gun owners alike, and we are unique in that regard.”
These “implementable solutions” could prevent the next massacre, Dr. Turner, executive director of the American College of Surgeons, said during a news briefing the group sponsored on June 2.
“Our future – indeed all of our futures – depend on our ability to find durable, actionable steps that we can implement tomorrow to save lives,” she said.
Firsthand perspective
“Sadly I’m here today as a trauma surgeon who has cared for two of the largest mass shootings in modern U.S. history,” said Ronald Stewart, MD, chair of the department of surgery at University Hospital in San Antonio, Texas.
Dr. Stewart treated victims of the 2017 Sutherland Springs First Baptist Church shooting – where 27 people died, including the shooter – and the recent Uvalde school shooting, both in Texas.
“The injuries inflicted by high-velocity weapons used at both of these attacks are horrific. A high-capacity, magazine-fed automatic rifle such as the AR-15 causes extremely destructive tissue wounds,” he said.
One of the group’s proposals is to increase the regulation of high-velocity weapons, including AR-15s.
“These wounds are horribly lethal at close range, and sadly, most victims do not survive long enough to make it to a trauma center,” Dr. Stewart said.
On a positive note, “all of our current [Uvalde] patients are improving, which really brings us joy in this dark time,” he said. “But all of them have a long road to deal with recovery with both the physical and emotional impact of their injuries.”
Jeffrey Kerby, MD, agreed.
“Trauma surgeons see the short-term physical effects of these injuries and watch patients struggle with the long-term impact of these wounds,” said Dr. Kerby, director of trauma and acute care surgery at the University of Alabama at Birmingham.
Surgeons feel ‘profound impact’ of shootings
“Firearm violence has a profound impact on surgeons, and we are the undisputed subject matter experts in treating the tragic results,” said Patrick Bailey, MD, medical director for advocacy at the American College of Surgeons.
“This impacts surgeons as well,” said Dr. Kerby, chair of the Committee on Trauma for the surgeons’ group. “We are human, and we can’t help but share in the grief, the pain, and the suffering that our patients endure.
“As a pediatric surgeon ... I have too often witnessed the impact of firearm violence, and obviously, the devastation extends beyond the victims to their families,” he said. “To put it succinctly, in our culture, parents are not supposed to be put in a position of burying their children.”
A public health crisis
“It’s important to recognize that we’ve been talking about a public health approach,” said Eileen Bulger, MD, acting chief of the trauma division at the University of Washington in Seattle. That strategy is important for engaging both firearm owners and communities that have a higher risk for firearm violence, she said.
A committee of the American College of Surgeons developed specific recommendations in 2018, which are still valid today. The group brought together surgeons from across the U.S. including “passionate firearm owners and experts in firearm safety,” Dr. Bulger said.
The committee, for example, agreed on 10 specific recommendations “that we believe are bipartisan and could have an immediate impact in saving lives.”
“I’m a lifelong gun owner,” Dr. Bailey said, emphasizing that the team’s process included participation and perspective from other surgeons “who, like me, are also gun owners, but gun owners who also seek to reduce the impact of firearm violence in our country.”
The recommendations address these areas:
- Gun ownership
- Firearm registration
- Licensure
- Education and training
- Ownership responsibilities
- Mandatory reporting and risk reduction
- Safety innovation and technology
- Research
- The culture of violence
- Social isolation and mental health
For example, “we currently have certain classes of weapons with significant offensive capability,” Dr. Bulger said, “that are appropriately restricted and regulated under the National Firearms Act as Class 3 weapons.”
This group includes fully automatic machine guns, explosive devices, and short-barrel shotguns.
“We recommend a formal reassessment of the firearms designated within each of these national firearms classifications,” Dr. Bulger said.
For example, high-capacity, magazine-fed semiautomatic rifles, such as the AR-15, should be considered for reclassification as NFA Class 3 firearms, or they should get a new designation with tighter regulation.
The ACS endorses formal firearm safety training for all new gun owners. Also, owners who do not provide reasonably safe firearm storage should be held responsible for events related to the discharge of their firearms, Dr. Bulger said. And people who are deemed an imminent threat to themselves or others through firearm ownership should be temporarily or permanently restricted, with due process.
Research and reporting reforms
The ACS is also calling for research on firearm injuries and firearm injury prevention to be federally funded, Dr. Bulger said. The research should be done in a nonpartisan manner, she said.
“We have concerns that the manner and tone in which information is released to the public may lead to copycat mass killers,” she said. “The ACS recommends that law enforcement officials and the press take steps to eliminate the notoriety of the shooter, for example.”
Dr. Bulger also addressed the mental health angle. “We encourage recognition of mental health warning signs and social isolation by teachers, counselors, peers, and parents.” When identified, immediate referral to professionals is needed.
In addition to these recommendations, another team from the American College of Surgeons has published an overview of ways to address the inequities that contribute to violence. “We advocate for federal funding to support the development of hospital-based and community programs for violence intervention and prevention,” Dr. Bulger said.
Dr. Bailey said that as a gun owner himself, he thinks other gun owners would support these recommendations.
“I do not believe that the steps recommended ... pose undue burden on the rights of individual gun owners,” he said.
The time is now
Most firearm injuries are not from mass shooting events, Dr. Kerby said.
“My own trauma center has seen a 40% increase in the number of firearm injuries just in the last 2 years,” he added, “and these numbers continue to grow.”
A version of this article first appeared on WebMD.com.
Their strategies can work regardless of where you stand on the Second Amendment of the Constitution, said Patricia Turner, MD. “Our proposals are embraced by both gun owners and non–gun owners alike, and we are unique in that regard.”
These “implementable solutions” could prevent the next massacre, Dr. Turner, executive director of the American College of Surgeons, said during a news briefing the group sponsored on June 2.
“Our future – indeed all of our futures – depend on our ability to find durable, actionable steps that we can implement tomorrow to save lives,” she said.
Firsthand perspective
“Sadly I’m here today as a trauma surgeon who has cared for two of the largest mass shootings in modern U.S. history,” said Ronald Stewart, MD, chair of the department of surgery at University Hospital in San Antonio, Texas.
Dr. Stewart treated victims of the 2017 Sutherland Springs First Baptist Church shooting – where 27 people died, including the shooter – and the recent Uvalde school shooting, both in Texas.
“The injuries inflicted by high-velocity weapons used at both of these attacks are horrific. A high-capacity, magazine-fed automatic rifle such as the AR-15 causes extremely destructive tissue wounds,” he said.
One of the group’s proposals is to increase the regulation of high-velocity weapons, including AR-15s.
“These wounds are horribly lethal at close range, and sadly, most victims do not survive long enough to make it to a trauma center,” Dr. Stewart said.
On a positive note, “all of our current [Uvalde] patients are improving, which really brings us joy in this dark time,” he said. “But all of them have a long road to deal with recovery with both the physical and emotional impact of their injuries.”
Jeffrey Kerby, MD, agreed.
“Trauma surgeons see the short-term physical effects of these injuries and watch patients struggle with the long-term impact of these wounds,” said Dr. Kerby, director of trauma and acute care surgery at the University of Alabama at Birmingham.
Surgeons feel ‘profound impact’ of shootings
“Firearm violence has a profound impact on surgeons, and we are the undisputed subject matter experts in treating the tragic results,” said Patrick Bailey, MD, medical director for advocacy at the American College of Surgeons.
“This impacts surgeons as well,” said Dr. Kerby, chair of the Committee on Trauma for the surgeons’ group. “We are human, and we can’t help but share in the grief, the pain, and the suffering that our patients endure.
“As a pediatric surgeon ... I have too often witnessed the impact of firearm violence, and obviously, the devastation extends beyond the victims to their families,” he said. “To put it succinctly, in our culture, parents are not supposed to be put in a position of burying their children.”
A public health crisis
“It’s important to recognize that we’ve been talking about a public health approach,” said Eileen Bulger, MD, acting chief of the trauma division at the University of Washington in Seattle. That strategy is important for engaging both firearm owners and communities that have a higher risk for firearm violence, she said.
A committee of the American College of Surgeons developed specific recommendations in 2018, which are still valid today. The group brought together surgeons from across the U.S. including “passionate firearm owners and experts in firearm safety,” Dr. Bulger said.
The committee, for example, agreed on 10 specific recommendations “that we believe are bipartisan and could have an immediate impact in saving lives.”
“I’m a lifelong gun owner,” Dr. Bailey said, emphasizing that the team’s process included participation and perspective from other surgeons “who, like me, are also gun owners, but gun owners who also seek to reduce the impact of firearm violence in our country.”
The recommendations address these areas:
- Gun ownership
- Firearm registration
- Licensure
- Education and training
- Ownership responsibilities
- Mandatory reporting and risk reduction
- Safety innovation and technology
- Research
- The culture of violence
- Social isolation and mental health
For example, “we currently have certain classes of weapons with significant offensive capability,” Dr. Bulger said, “that are appropriately restricted and regulated under the National Firearms Act as Class 3 weapons.”
This group includes fully automatic machine guns, explosive devices, and short-barrel shotguns.
“We recommend a formal reassessment of the firearms designated within each of these national firearms classifications,” Dr. Bulger said.
For example, high-capacity, magazine-fed semiautomatic rifles, such as the AR-15, should be considered for reclassification as NFA Class 3 firearms, or they should get a new designation with tighter regulation.
The ACS endorses formal firearm safety training for all new gun owners. Also, owners who do not provide reasonably safe firearm storage should be held responsible for events related to the discharge of their firearms, Dr. Bulger said. And people who are deemed an imminent threat to themselves or others through firearm ownership should be temporarily or permanently restricted, with due process.
Research and reporting reforms
The ACS is also calling for research on firearm injuries and firearm injury prevention to be federally funded, Dr. Bulger said. The research should be done in a nonpartisan manner, she said.
“We have concerns that the manner and tone in which information is released to the public may lead to copycat mass killers,” she said. “The ACS recommends that law enforcement officials and the press take steps to eliminate the notoriety of the shooter, for example.”
Dr. Bulger also addressed the mental health angle. “We encourage recognition of mental health warning signs and social isolation by teachers, counselors, peers, and parents.” When identified, immediate referral to professionals is needed.
In addition to these recommendations, another team from the American College of Surgeons has published an overview of ways to address the inequities that contribute to violence. “We advocate for federal funding to support the development of hospital-based and community programs for violence intervention and prevention,” Dr. Bulger said.
Dr. Bailey said that as a gun owner himself, he thinks other gun owners would support these recommendations.
“I do not believe that the steps recommended ... pose undue burden on the rights of individual gun owners,” he said.
The time is now
Most firearm injuries are not from mass shooting events, Dr. Kerby said.
“My own trauma center has seen a 40% increase in the number of firearm injuries just in the last 2 years,” he added, “and these numbers continue to grow.”
A version of this article first appeared on WebMD.com.
mTOR inhibitor shows early promise in endometrial cancer
In an open-label, phase 1/2, randomized clinical trial (VICTORIA) performed at 12 cancer centers in France, with good tolerability.
The study was published in JAMA Oncology.
Treatment of endometrial cancer involves a combination of surgery, radiation, and chemotherapy, but about 20% of patients relapse, usually within 5 years. HR+ endometrial cancer represents about 65% of endometrial cancers. It is usually endometrioid, and about 80% have phosphatase and tensin homologue (PTEN) mutations, while 36-52% have a mutation in the phosphoinositide 3-kinases/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) pathway.
Endocrine therapy alone elicits a response rate of 15%-30% in HR+ endometrial cancer, generally in low-grade endometrioid subtypes. Most responses are short in duration. Aromatase inhibitors like anastrozole are used for more often than progestogens because they are better tolerated and they have a lower thromboembolic risk in this patient population. Previously, the phase 2 PARAGON trial showed a response rate of just 7% with anastrozole monotherapy, but 44% in women with recurrent HR+ endometrial cancer gained a clinical benefit.
Deregulation in the PI3K/AKT/mTOR pathway can also lead to hormone resistance, suggesting that combination of an mTOR inhibitor with endocrine therapy might have a synergistic effect.
mTOR inhibition alone or in combination with endocrine treatment has been investigated in some single-arm studies, with some encouraging progression-free survival results, but no clear objective response rate or overall survival benefit.
The new study included just 73 patients with a median age of 69.5 years: 49 received 125 mg vistusertib 2 days per week and 1 mg anastrozole daily and 24 received anastrozole only. The 8-week progression-free rate was 67.3% (unilateral 95% confidence interval, 54.7%) in the combination arm versus 39.1% (unilateral 95% CI, 22.2%) in the anastrozole-only arm.
Among 6 patients in the safety run-in period of the combination arm, there were no serious adverse events. Overall response rate was 24.5% (95% CI, 13.3-38.9%) in the combination arm and 17.4% (95% CI, 5.0-38.8%) in the anastrozole-only arm.
Over a median follow-up of 27.7 months, progression-free survival was 5.2 months in the combination arm (95% CI, 3.4-8.9 months) and 1.9 months (95% CI, 1.6-8.9) In the anastrozole-only arm. Common grade 2 or higher side effects linked to vistusertib included fatigue, lymphopenia, hyperglycemia, and diarrhea.
Although low tumor grade, endometrioid subtype, and HR+ status are associated with response to endocrine therapy, the low overall response and progression-free survival in the anastrozole arm suggest that better patient selection is needed. “The choice of treatment according to the histologic characteristics is not sufficient, and highly selected molecular criteria are necessary,” the authors wrote.
The study is limited by its small size and a lack of data on expression level of hormone receptors.
The study was funded by the National Cancer Institute of France.
In an open-label, phase 1/2, randomized clinical trial (VICTORIA) performed at 12 cancer centers in France, with good tolerability.
The study was published in JAMA Oncology.
Treatment of endometrial cancer involves a combination of surgery, radiation, and chemotherapy, but about 20% of patients relapse, usually within 5 years. HR+ endometrial cancer represents about 65% of endometrial cancers. It is usually endometrioid, and about 80% have phosphatase and tensin homologue (PTEN) mutations, while 36-52% have a mutation in the phosphoinositide 3-kinases/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) pathway.
Endocrine therapy alone elicits a response rate of 15%-30% in HR+ endometrial cancer, generally in low-grade endometrioid subtypes. Most responses are short in duration. Aromatase inhibitors like anastrozole are used for more often than progestogens because they are better tolerated and they have a lower thromboembolic risk in this patient population. Previously, the phase 2 PARAGON trial showed a response rate of just 7% with anastrozole monotherapy, but 44% in women with recurrent HR+ endometrial cancer gained a clinical benefit.
Deregulation in the PI3K/AKT/mTOR pathway can also lead to hormone resistance, suggesting that combination of an mTOR inhibitor with endocrine therapy might have a synergistic effect.
mTOR inhibition alone or in combination with endocrine treatment has been investigated in some single-arm studies, with some encouraging progression-free survival results, but no clear objective response rate or overall survival benefit.
The new study included just 73 patients with a median age of 69.5 years: 49 received 125 mg vistusertib 2 days per week and 1 mg anastrozole daily and 24 received anastrozole only. The 8-week progression-free rate was 67.3% (unilateral 95% confidence interval, 54.7%) in the combination arm versus 39.1% (unilateral 95% CI, 22.2%) in the anastrozole-only arm.
Among 6 patients in the safety run-in period of the combination arm, there were no serious adverse events. Overall response rate was 24.5% (95% CI, 13.3-38.9%) in the combination arm and 17.4% (95% CI, 5.0-38.8%) in the anastrozole-only arm.
Over a median follow-up of 27.7 months, progression-free survival was 5.2 months in the combination arm (95% CI, 3.4-8.9 months) and 1.9 months (95% CI, 1.6-8.9) In the anastrozole-only arm. Common grade 2 or higher side effects linked to vistusertib included fatigue, lymphopenia, hyperglycemia, and diarrhea.
Although low tumor grade, endometrioid subtype, and HR+ status are associated with response to endocrine therapy, the low overall response and progression-free survival in the anastrozole arm suggest that better patient selection is needed. “The choice of treatment according to the histologic characteristics is not sufficient, and highly selected molecular criteria are necessary,” the authors wrote.
The study is limited by its small size and a lack of data on expression level of hormone receptors.
The study was funded by the National Cancer Institute of France.
In an open-label, phase 1/2, randomized clinical trial (VICTORIA) performed at 12 cancer centers in France, with good tolerability.
The study was published in JAMA Oncology.
Treatment of endometrial cancer involves a combination of surgery, radiation, and chemotherapy, but about 20% of patients relapse, usually within 5 years. HR+ endometrial cancer represents about 65% of endometrial cancers. It is usually endometrioid, and about 80% have phosphatase and tensin homologue (PTEN) mutations, while 36-52% have a mutation in the phosphoinositide 3-kinases/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) pathway.
Endocrine therapy alone elicits a response rate of 15%-30% in HR+ endometrial cancer, generally in low-grade endometrioid subtypes. Most responses are short in duration. Aromatase inhibitors like anastrozole are used for more often than progestogens because they are better tolerated and they have a lower thromboembolic risk in this patient population. Previously, the phase 2 PARAGON trial showed a response rate of just 7% with anastrozole monotherapy, but 44% in women with recurrent HR+ endometrial cancer gained a clinical benefit.
Deregulation in the PI3K/AKT/mTOR pathway can also lead to hormone resistance, suggesting that combination of an mTOR inhibitor with endocrine therapy might have a synergistic effect.
mTOR inhibition alone or in combination with endocrine treatment has been investigated in some single-arm studies, with some encouraging progression-free survival results, but no clear objective response rate or overall survival benefit.
The new study included just 73 patients with a median age of 69.5 years: 49 received 125 mg vistusertib 2 days per week and 1 mg anastrozole daily and 24 received anastrozole only. The 8-week progression-free rate was 67.3% (unilateral 95% confidence interval, 54.7%) in the combination arm versus 39.1% (unilateral 95% CI, 22.2%) in the anastrozole-only arm.
Among 6 patients in the safety run-in period of the combination arm, there were no serious adverse events. Overall response rate was 24.5% (95% CI, 13.3-38.9%) in the combination arm and 17.4% (95% CI, 5.0-38.8%) in the anastrozole-only arm.
Over a median follow-up of 27.7 months, progression-free survival was 5.2 months in the combination arm (95% CI, 3.4-8.9 months) and 1.9 months (95% CI, 1.6-8.9) In the anastrozole-only arm. Common grade 2 or higher side effects linked to vistusertib included fatigue, lymphopenia, hyperglycemia, and diarrhea.
Although low tumor grade, endometrioid subtype, and HR+ status are associated with response to endocrine therapy, the low overall response and progression-free survival in the anastrozole arm suggest that better patient selection is needed. “The choice of treatment according to the histologic characteristics is not sufficient, and highly selected molecular criteria are necessary,” the authors wrote.
The study is limited by its small size and a lack of data on expression level of hormone receptors.
The study was funded by the National Cancer Institute of France.
FROM JAMA ONCOLOGY
Dogs can be protective, even against Crohn’s disease
Sorry, cat people and only children: Having a dog as a toddler and growing up in a large family are two things linked to a significantly lower chance of getting Crohn’s disease later in life, according to a new study.
Children who lived with a dog between the ages of 2 years and 4 years were 37% less likely to have Crohn’s disease, the study says. And those who lived with at least three other family members during the first year of life were 64% less likely to have this form of inflammatory bowel disease (IBD).
“In this study, we’re interested in environmental exposures and which ones are associated with Crohn’s disease onset,” Williams Turpin, PhD, said in a media interview May 23 at the annual Digestive Disease Week® (DDW).
Dr. Turpin and colleagues looked at other things in the environment – including living on a farm, drinking unpasteurized milk or well water, and growing up with a cat – but they did not have a significant link to a higher risk.
Two other things were associated with a slight increase in risk: having a sibling with Crohn’s disease and living with a bird at time of the study. But the number of bird owners was small; only a few people in the study had a pet bird when they enrolled.
The link to living with a dog as a toddler “was more robust,” said Dr. Turpin, a project manager at Mount Sinai Hospital in Toronto.
The study included 4,289 healthy first-degree relatives of people diagnosed with Crohn’s disease. They provided urine, blood, and stool samples and did surveys about environmental exposures at different stages of life.
Investigators followed them an average of 5.6 years, during which time 86 people got Crohn’s disease.
Gut instinct
Living with a dog early in life likely means more exposure to different microbes, boosting the strength of a person’s immune system against later challenges. This theory was supported in the study comparing the gut microbiome in people who did and not have a dog in the home early in life.
Dr. Turpin and colleagues genetically sequenced the gut microbiome of the people in the study and found differences in bacteria between groups.
“Our study also shows that just by living with a dog, it impacts your gut microbiome composition, which may have an impact on the immune response later in life,” Dr. Turpin said.
The researchers also looked at the health of the gut by measuring certain factors in the urine. One factor was higher in people who did not live with a dog at any point.
Mediated by the microbiome?
Living with a dog between the ages of 2 and 4 years and a large family size (more than three people) in the first year were significantly associated with a lower risk of Crohn’s disease onset.
It is unknown if the results apply to other populations; the researchers studied first-degree relatives of people with Crohn’s disease.
“The study needs to be replicated and validated,” Dr. Turpin said.
Future research could evaluate people who never had a dog and look for changes in their microbiome after they get one.
‘Well-crafted’ study
“It’s a really interesting study from a good group. It’s novel in terms of getting at what really drives environmental risk factors,” said Brigid Boland, MD, a gastroenterologist at UC San Diego Health, who was not affiliated with the study.
Autoimmune diseases are really complicated, in part because the risk of getting an autoimmune disease is low, and you’re going back in time to look at what put people at risk.
“The study was well crafted in choosing siblings and family members of people with IBD,” Dr. Boland said, agreeing with Dr. Turpin that more research is needed to understand this.
A version of this article first appeared on WebMD.com.
Sorry, cat people and only children: Having a dog as a toddler and growing up in a large family are two things linked to a significantly lower chance of getting Crohn’s disease later in life, according to a new study.
Children who lived with a dog between the ages of 2 years and 4 years were 37% less likely to have Crohn’s disease, the study says. And those who lived with at least three other family members during the first year of life were 64% less likely to have this form of inflammatory bowel disease (IBD).
“In this study, we’re interested in environmental exposures and which ones are associated with Crohn’s disease onset,” Williams Turpin, PhD, said in a media interview May 23 at the annual Digestive Disease Week® (DDW).
Dr. Turpin and colleagues looked at other things in the environment – including living on a farm, drinking unpasteurized milk or well water, and growing up with a cat – but they did not have a significant link to a higher risk.
Two other things were associated with a slight increase in risk: having a sibling with Crohn’s disease and living with a bird at time of the study. But the number of bird owners was small; only a few people in the study had a pet bird when they enrolled.
The link to living with a dog as a toddler “was more robust,” said Dr. Turpin, a project manager at Mount Sinai Hospital in Toronto.
The study included 4,289 healthy first-degree relatives of people diagnosed with Crohn’s disease. They provided urine, blood, and stool samples and did surveys about environmental exposures at different stages of life.
Investigators followed them an average of 5.6 years, during which time 86 people got Crohn’s disease.
Gut instinct
Living with a dog early in life likely means more exposure to different microbes, boosting the strength of a person’s immune system against later challenges. This theory was supported in the study comparing the gut microbiome in people who did and not have a dog in the home early in life.
Dr. Turpin and colleagues genetically sequenced the gut microbiome of the people in the study and found differences in bacteria between groups.
“Our study also shows that just by living with a dog, it impacts your gut microbiome composition, which may have an impact on the immune response later in life,” Dr. Turpin said.
The researchers also looked at the health of the gut by measuring certain factors in the urine. One factor was higher in people who did not live with a dog at any point.
Mediated by the microbiome?
Living with a dog between the ages of 2 and 4 years and a large family size (more than three people) in the first year were significantly associated with a lower risk of Crohn’s disease onset.
It is unknown if the results apply to other populations; the researchers studied first-degree relatives of people with Crohn’s disease.
“The study needs to be replicated and validated,” Dr. Turpin said.
Future research could evaluate people who never had a dog and look for changes in their microbiome after they get one.
‘Well-crafted’ study
“It’s a really interesting study from a good group. It’s novel in terms of getting at what really drives environmental risk factors,” said Brigid Boland, MD, a gastroenterologist at UC San Diego Health, who was not affiliated with the study.
Autoimmune diseases are really complicated, in part because the risk of getting an autoimmune disease is low, and you’re going back in time to look at what put people at risk.
“The study was well crafted in choosing siblings and family members of people with IBD,” Dr. Boland said, agreeing with Dr. Turpin that more research is needed to understand this.
A version of this article first appeared on WebMD.com.
Sorry, cat people and only children: Having a dog as a toddler and growing up in a large family are two things linked to a significantly lower chance of getting Crohn’s disease later in life, according to a new study.
Children who lived with a dog between the ages of 2 years and 4 years were 37% less likely to have Crohn’s disease, the study says. And those who lived with at least three other family members during the first year of life were 64% less likely to have this form of inflammatory bowel disease (IBD).
“In this study, we’re interested in environmental exposures and which ones are associated with Crohn’s disease onset,” Williams Turpin, PhD, said in a media interview May 23 at the annual Digestive Disease Week® (DDW).
Dr. Turpin and colleagues looked at other things in the environment – including living on a farm, drinking unpasteurized milk or well water, and growing up with a cat – but they did not have a significant link to a higher risk.
Two other things were associated with a slight increase in risk: having a sibling with Crohn’s disease and living with a bird at time of the study. But the number of bird owners was small; only a few people in the study had a pet bird when they enrolled.
The link to living with a dog as a toddler “was more robust,” said Dr. Turpin, a project manager at Mount Sinai Hospital in Toronto.
The study included 4,289 healthy first-degree relatives of people diagnosed with Crohn’s disease. They provided urine, blood, and stool samples and did surveys about environmental exposures at different stages of life.
Investigators followed them an average of 5.6 years, during which time 86 people got Crohn’s disease.
Gut instinct
Living with a dog early in life likely means more exposure to different microbes, boosting the strength of a person’s immune system against later challenges. This theory was supported in the study comparing the gut microbiome in people who did and not have a dog in the home early in life.
Dr. Turpin and colleagues genetically sequenced the gut microbiome of the people in the study and found differences in bacteria between groups.
“Our study also shows that just by living with a dog, it impacts your gut microbiome composition, which may have an impact on the immune response later in life,” Dr. Turpin said.
The researchers also looked at the health of the gut by measuring certain factors in the urine. One factor was higher in people who did not live with a dog at any point.
Mediated by the microbiome?
Living with a dog between the ages of 2 and 4 years and a large family size (more than three people) in the first year were significantly associated with a lower risk of Crohn’s disease onset.
It is unknown if the results apply to other populations; the researchers studied first-degree relatives of people with Crohn’s disease.
“The study needs to be replicated and validated,” Dr. Turpin said.
Future research could evaluate people who never had a dog and look for changes in their microbiome after they get one.
‘Well-crafted’ study
“It’s a really interesting study from a good group. It’s novel in terms of getting at what really drives environmental risk factors,” said Brigid Boland, MD, a gastroenterologist at UC San Diego Health, who was not affiliated with the study.
Autoimmune diseases are really complicated, in part because the risk of getting an autoimmune disease is low, and you’re going back in time to look at what put people at risk.
“The study was well crafted in choosing siblings and family members of people with IBD,” Dr. Boland said, agreeing with Dr. Turpin that more research is needed to understand this.
A version of this article first appeared on WebMD.com.
FROM DDW 2022
Can lasers be used to measure nerve sensitivity in the skin?
SAN DIEGO – In a 2006 report of complications from laser dermatologic surgery, one of the authors, Dieter Manstein, MD, PhD, who had subjected his forearm to treatment with a fractional laser skin resurfacing prototype device, was included as 1 of the 19 featured cases.
Dr. Manstein, of the Cutaneous Biology Research Center in the department of dermatology at Massachusetts General Hospital, Boston, was exposed to three test spots in the evaluation of the effects of different microscopic thermal zone densities for the prototype device, emitting at 1,450 nm and an energy per MTZ of 3 mJ.
Two years later, hypopigmentation persisted at the test site treated with the highest MTZ density, while two other sites treated with the lower MTZ densities did not show any dyspigmentation. But he noticed something else during the experiment: He felt minimal to no pain as each test site was being treated.
“It took 7 minutes without any cooling or anesthesia,” Dr. Manstein recalled at the annual meeting of the American Society for Laser Medicine and Surgery. “It was not completely painless, but each time the laser was applied, sometimes I felt a little prick, sometimes I felt nothing.” Essentially, he added, “we created cell injury with a focused laser beam without anesthesia,” but this could also indicate that if skin is treated with a fractional laser very slowly, anesthesia is not needed. “Current devices are meant to treat very quickly, but if we [treat] slowly, maybe you could remove lesions painlessly without anesthesia.”
The observation from that experiment also led Dr. Manstein and colleagues to wonder: Could a focused laser beam pattern be used to assess cutaneous innervation? If so, they postulated, perhaps it could be used to not only assess nerve sensitivity of candidates for dermatologic surgery, but as a tool to help diagnose small fiber neuropathies such as diabetic neuropathy, and neuropathies in patients with HIV and sarcoidosis.
The current gold standard for making these diagnoses involves a skin biopsy, immunohistochemical analysis, and nerve fiber quantification, which is not widely available. It also requires strict histologic processing and nerve counting rules. Confocal microscopy of nerve fibers in the cornea is another approach, but is very difficult to perform, “so it would be nice if there was a simple way” to determine nerve fiber density in the skin using a focused laser beam, Dr. Manstein said.
With help from Payal Patel, MD, a dermatology research fellow at MGH, records each subject’s perception of a stimulus, and maps the areas of stimulus response. Current diameters being studied range from 0.076-1.15 mm and depths less than 0.71 mm. “We can focus the laser beam, preset the beam diameter, and very slowly, in a controlled manner, make a rectangular pattern, and after each time, inquire if the subject felt the pulse or not,” Dr. Manstein explained.
“This laser could become a new method for diagnosing nerve fiber neuropathies. If this works well, I think we can miniaturize the device,” he added.
Dr. Manstein disclosed that he is a consultant for Blossom Innovations, R2 Dermatology, and AVAVA. He is also a member of the advisory board for Blossom Innovations.
SAN DIEGO – In a 2006 report of complications from laser dermatologic surgery, one of the authors, Dieter Manstein, MD, PhD, who had subjected his forearm to treatment with a fractional laser skin resurfacing prototype device, was included as 1 of the 19 featured cases.
Dr. Manstein, of the Cutaneous Biology Research Center in the department of dermatology at Massachusetts General Hospital, Boston, was exposed to three test spots in the evaluation of the effects of different microscopic thermal zone densities for the prototype device, emitting at 1,450 nm and an energy per MTZ of 3 mJ.
Two years later, hypopigmentation persisted at the test site treated with the highest MTZ density, while two other sites treated with the lower MTZ densities did not show any dyspigmentation. But he noticed something else during the experiment: He felt minimal to no pain as each test site was being treated.
“It took 7 minutes without any cooling or anesthesia,” Dr. Manstein recalled at the annual meeting of the American Society for Laser Medicine and Surgery. “It was not completely painless, but each time the laser was applied, sometimes I felt a little prick, sometimes I felt nothing.” Essentially, he added, “we created cell injury with a focused laser beam without anesthesia,” but this could also indicate that if skin is treated with a fractional laser very slowly, anesthesia is not needed. “Current devices are meant to treat very quickly, but if we [treat] slowly, maybe you could remove lesions painlessly without anesthesia.”
The observation from that experiment also led Dr. Manstein and colleagues to wonder: Could a focused laser beam pattern be used to assess cutaneous innervation? If so, they postulated, perhaps it could be used to not only assess nerve sensitivity of candidates for dermatologic surgery, but as a tool to help diagnose small fiber neuropathies such as diabetic neuropathy, and neuropathies in patients with HIV and sarcoidosis.
The current gold standard for making these diagnoses involves a skin biopsy, immunohistochemical analysis, and nerve fiber quantification, which is not widely available. It also requires strict histologic processing and nerve counting rules. Confocal microscopy of nerve fibers in the cornea is another approach, but is very difficult to perform, “so it would be nice if there was a simple way” to determine nerve fiber density in the skin using a focused laser beam, Dr. Manstein said.
With help from Payal Patel, MD, a dermatology research fellow at MGH, records each subject’s perception of a stimulus, and maps the areas of stimulus response. Current diameters being studied range from 0.076-1.15 mm and depths less than 0.71 mm. “We can focus the laser beam, preset the beam diameter, and very slowly, in a controlled manner, make a rectangular pattern, and after each time, inquire if the subject felt the pulse or not,” Dr. Manstein explained.
“This laser could become a new method for diagnosing nerve fiber neuropathies. If this works well, I think we can miniaturize the device,” he added.
Dr. Manstein disclosed that he is a consultant for Blossom Innovations, R2 Dermatology, and AVAVA. He is also a member of the advisory board for Blossom Innovations.
SAN DIEGO – In a 2006 report of complications from laser dermatologic surgery, one of the authors, Dieter Manstein, MD, PhD, who had subjected his forearm to treatment with a fractional laser skin resurfacing prototype device, was included as 1 of the 19 featured cases.
Dr. Manstein, of the Cutaneous Biology Research Center in the department of dermatology at Massachusetts General Hospital, Boston, was exposed to three test spots in the evaluation of the effects of different microscopic thermal zone densities for the prototype device, emitting at 1,450 nm and an energy per MTZ of 3 mJ.
Two years later, hypopigmentation persisted at the test site treated with the highest MTZ density, while two other sites treated with the lower MTZ densities did not show any dyspigmentation. But he noticed something else during the experiment: He felt minimal to no pain as each test site was being treated.
“It took 7 minutes without any cooling or anesthesia,” Dr. Manstein recalled at the annual meeting of the American Society for Laser Medicine and Surgery. “It was not completely painless, but each time the laser was applied, sometimes I felt a little prick, sometimes I felt nothing.” Essentially, he added, “we created cell injury with a focused laser beam without anesthesia,” but this could also indicate that if skin is treated with a fractional laser very slowly, anesthesia is not needed. “Current devices are meant to treat very quickly, but if we [treat] slowly, maybe you could remove lesions painlessly without anesthesia.”
The observation from that experiment also led Dr. Manstein and colleagues to wonder: Could a focused laser beam pattern be used to assess cutaneous innervation? If so, they postulated, perhaps it could be used to not only assess nerve sensitivity of candidates for dermatologic surgery, but as a tool to help diagnose small fiber neuropathies such as diabetic neuropathy, and neuropathies in patients with HIV and sarcoidosis.
The current gold standard for making these diagnoses involves a skin biopsy, immunohistochemical analysis, and nerve fiber quantification, which is not widely available. It also requires strict histologic processing and nerve counting rules. Confocal microscopy of nerve fibers in the cornea is another approach, but is very difficult to perform, “so it would be nice if there was a simple way” to determine nerve fiber density in the skin using a focused laser beam, Dr. Manstein said.
With help from Payal Patel, MD, a dermatology research fellow at MGH, records each subject’s perception of a stimulus, and maps the areas of stimulus response. Current diameters being studied range from 0.076-1.15 mm and depths less than 0.71 mm. “We can focus the laser beam, preset the beam diameter, and very slowly, in a controlled manner, make a rectangular pattern, and after each time, inquire if the subject felt the pulse or not,” Dr. Manstein explained.
“This laser could become a new method for diagnosing nerve fiber neuropathies. If this works well, I think we can miniaturize the device,” he added.
Dr. Manstein disclosed that he is a consultant for Blossom Innovations, R2 Dermatology, and AVAVA. He is also a member of the advisory board for Blossom Innovations.
AT ASLMS 2022
Sweet Syndrome With Pulmonary Involvement Preceding the Development of Myelodysplastic Syndrome
To the Editor:
A 59-year-old man was referred to our clinic for a rash, fever, and night sweats following treatment for metastatic seminoma with cisplatin and etoposide. Physical examination revealed indurated erythematous papules and plaques on the trunk and upper and lower extremities, some with annular or arcuate configuration with trailing scale (Figure, A). A skin biopsy demonstrated mild papillary dermal edema with a mixed infiltrate of mononuclear cells, neutrophils, eosinophils, mast cells, lymphocytes, and karyorrhectic debris without evidence of leukocytoclastic vasculitis. The histopathologic differential diagnosis included a histiocytoid variant of Sweet syndrome (SS), and our patient’s rapid clinical response to corticosteroids supported this diagnosis.
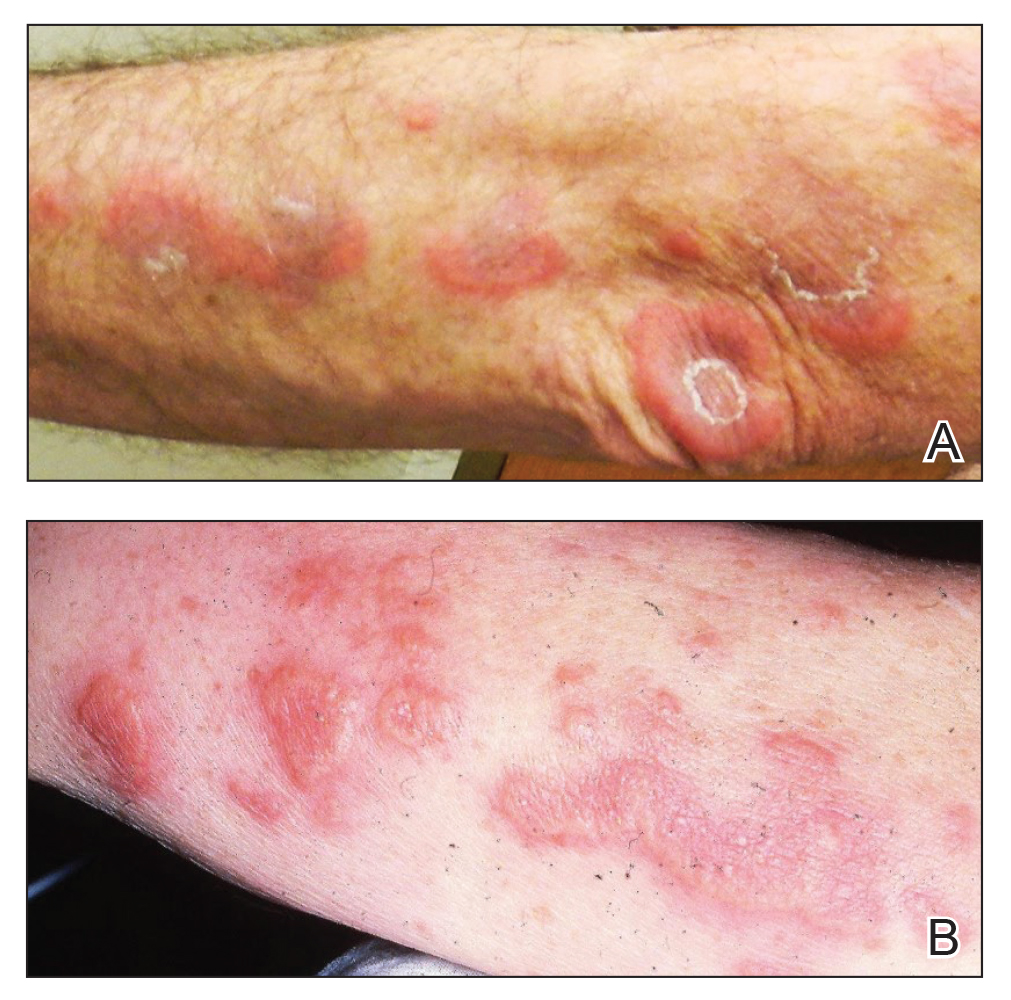
With a relapsing and remitting course over 3 years, the rash eventually evolved into more edematous papules and plaques (Figure, B), and a repeat biopsy 3 years later was consistent with classic SS. Although the patient's condition improved with prednisone, attempts to taper prednisone invariably resulted in relapse. Multiple steroid-sparing agents were trialed over the course of 3 years including dapsone and mycophenolate mofetil, both of which resulted in hypersensitivity drug eruptions. Colchicine and methotrexate were ineffective. Thalidomide strongly was considered but ultimately was avoided due to substantial existing neuropathy associated with his prior chemotherapy for metastatic seminoma.
Four years after the initial diagnosis of SS, our patient presented with dyspnea and weight loss. Computed tomography revealed a nearly confluent miliary pattern of nodularity in the lungs. A wedge biopsy demonstrated pneumonitis with intra-alveolar fibrin and neutrophils with a notable absence of granulomatous inflammation. Fungal and acid-fast bacilli staining as well as tissue cultures were negative. He had a history of Mycobacterium kansasii pulmonary infection treated 18 months prior; however, in this instance, the histopathology, negative microbial cultures, and rapid steroid responsiveness were consistent with pulmonary involvement of SS. Over the ensuing 2 years, the patient developed worsening of his chronic anemia. He was diagnosed with myelodysplastic syndrome (MDS) by bone marrow biopsy, despite having a normal bone marrow biopsy more than 3 years prior to evaluate his anemia. At this time, thalidomide was initiated at 50 mg daily leading to notable improvement in his SS symptoms; however, he developed worsening neuropathy resulting in the discontinuation of this treatment 2 months later. An investigational combination of vosaroxin and azacytidine was used to treat his MDS, resulting in normalization of blood counts and remission from SS.
Sweet syndrome may occur in the setting of undiagnosed cancer or may signal the return of a previously treated malignancy. The first description of SS associated with solid tumors was in a patient with testicular cancer,1 which prompted continuous surveillance for recurrent seminoma in our patient, though none was found. Hematologic malignancies, as well as MDS, often are associated with SS.2 In our patient, multiple atypical features linked the development of SS to the ultimate presentation of MDS. The initial finding of a histiocytoid variant has been described in a case series of 9 patients with chronic relapsing SS who eventually developed MDS with latency of up to 7 years. The histopathology in these cases evolved over time to that of classic neutrophilic SS.3 Pulmonary involvement of SS is another interesting aspect of our case. In one analysis, 18 of 34 (53%) cases with pulmonary involvement featured hematologic pathology, including myelodysplasia and acute leukemia.4
In our patient, SS preceded the clinical manifestation of MDS by 6 years. A similar phenomenon has been described in a patient with SS that preceded myelodysplasia by 30 months and was recalcitrant to numerous steroid-sparing therapies except thalidomide, despite the persistence of myelodysplasia. Tapering thalidomide, however, resulted in recurrence of SS lesions in that patient.5 In another case, resolution of myelodysplasia from azacytidine treatment was associated with remission from SS.6
Our case represents a confluence of atypical features that seem to define myelodysplasia-associated SS, including the initial presentation with a clinically atypical histiocytoid variant, chronic relapsing and remitting course, and extracutaneous involvement of the lungs. These findings should prompt surveillance for hematologic malignancy or myelodysplasia. Serial bone marrow biopsies were required to evaluate persistent anemia before the histopathologic findings of MDS became apparent in our patient. Thalidomide was an effective treatment for the cutaneous manifestations in our patient and should be considered as a steroid-sparing agent in the treatment of recalcitrant SS. Despite the discontinuation of thalidomide therapy, effective control of our patient’s myelodysplasia with chemotherapy has kept him in remission from SS for more than 7 years of follow-up, suggesting a causal relationship between these disorders.
- Shapiro L, Baraf CS, Richheimer LL. Sweet’s syndrome (acute febrile neutrophilic dermatosis): report of a case. Arch Dermatol. 1971;103:81-84.
- Cohen PR. Sweet’s syndrome—a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
- Vignon-Pennamen MD, Juillard C, Rybojad M, et al. Chronic recurrent lymphocytic Sweet syndrome as a predictive marker of myelodysplasia. Arch Dermatol. 2006;142:1170-1176.
- Fernandez-Bussy S, Labarca G, Cabello F, et al. Sweet’s syndrome with pulmonary involvement: case report and literature review. Respir Med Case Rep. 2012;6:16-19.
- Browning CE, Dixon DE, Malone JC, et al. Thalidomide in the treatment of recalcitrant Sweet’s syndrome associated with myelodysplasia. J Am Acad Dermatol. 2005;53(2 suppl 1):S135-S138.
- Martinelli S, Rigolin GM, Leo G, et al. Complete remission Sweet’s syndrome after azacytidine treatment for concomitant myelodysplastic syndrome. Int J Hematol. 2014;99:663-667.
To the Editor:
A 59-year-old man was referred to our clinic for a rash, fever, and night sweats following treatment for metastatic seminoma with cisplatin and etoposide. Physical examination revealed indurated erythematous papules and plaques on the trunk and upper and lower extremities, some with annular or arcuate configuration with trailing scale (Figure, A). A skin biopsy demonstrated mild papillary dermal edema with a mixed infiltrate of mononuclear cells, neutrophils, eosinophils, mast cells, lymphocytes, and karyorrhectic debris without evidence of leukocytoclastic vasculitis. The histopathologic differential diagnosis included a histiocytoid variant of Sweet syndrome (SS), and our patient’s rapid clinical response to corticosteroids supported this diagnosis.

With a relapsing and remitting course over 3 years, the rash eventually evolved into more edematous papules and plaques (Figure, B), and a repeat biopsy 3 years later was consistent with classic SS. Although the patient's condition improved with prednisone, attempts to taper prednisone invariably resulted in relapse. Multiple steroid-sparing agents were trialed over the course of 3 years including dapsone and mycophenolate mofetil, both of which resulted in hypersensitivity drug eruptions. Colchicine and methotrexate were ineffective. Thalidomide strongly was considered but ultimately was avoided due to substantial existing neuropathy associated with his prior chemotherapy for metastatic seminoma.
Four years after the initial diagnosis of SS, our patient presented with dyspnea and weight loss. Computed tomography revealed a nearly confluent miliary pattern of nodularity in the lungs. A wedge biopsy demonstrated pneumonitis with intra-alveolar fibrin and neutrophils with a notable absence of granulomatous inflammation. Fungal and acid-fast bacilli staining as well as tissue cultures were negative. He had a history of Mycobacterium kansasii pulmonary infection treated 18 months prior; however, in this instance, the histopathology, negative microbial cultures, and rapid steroid responsiveness were consistent with pulmonary involvement of SS. Over the ensuing 2 years, the patient developed worsening of his chronic anemia. He was diagnosed with myelodysplastic syndrome (MDS) by bone marrow biopsy, despite having a normal bone marrow biopsy more than 3 years prior to evaluate his anemia. At this time, thalidomide was initiated at 50 mg daily leading to notable improvement in his SS symptoms; however, he developed worsening neuropathy resulting in the discontinuation of this treatment 2 months later. An investigational combination of vosaroxin and azacytidine was used to treat his MDS, resulting in normalization of blood counts and remission from SS.
Sweet syndrome may occur in the setting of undiagnosed cancer or may signal the return of a previously treated malignancy. The first description of SS associated with solid tumors was in a patient with testicular cancer,1 which prompted continuous surveillance for recurrent seminoma in our patient, though none was found. Hematologic malignancies, as well as MDS, often are associated with SS.2 In our patient, multiple atypical features linked the development of SS to the ultimate presentation of MDS. The initial finding of a histiocytoid variant has been described in a case series of 9 patients with chronic relapsing SS who eventually developed MDS with latency of up to 7 years. The histopathology in these cases evolved over time to that of classic neutrophilic SS.3 Pulmonary involvement of SS is another interesting aspect of our case. In one analysis, 18 of 34 (53%) cases with pulmonary involvement featured hematologic pathology, including myelodysplasia and acute leukemia.4
In our patient, SS preceded the clinical manifestation of MDS by 6 years. A similar phenomenon has been described in a patient with SS that preceded myelodysplasia by 30 months and was recalcitrant to numerous steroid-sparing therapies except thalidomide, despite the persistence of myelodysplasia. Tapering thalidomide, however, resulted in recurrence of SS lesions in that patient.5 In another case, resolution of myelodysplasia from azacytidine treatment was associated with remission from SS.6
Our case represents a confluence of atypical features that seem to define myelodysplasia-associated SS, including the initial presentation with a clinically atypical histiocytoid variant, chronic relapsing and remitting course, and extracutaneous involvement of the lungs. These findings should prompt surveillance for hematologic malignancy or myelodysplasia. Serial bone marrow biopsies were required to evaluate persistent anemia before the histopathologic findings of MDS became apparent in our patient. Thalidomide was an effective treatment for the cutaneous manifestations in our patient and should be considered as a steroid-sparing agent in the treatment of recalcitrant SS. Despite the discontinuation of thalidomide therapy, effective control of our patient’s myelodysplasia with chemotherapy has kept him in remission from SS for more than 7 years of follow-up, suggesting a causal relationship between these disorders.
To the Editor:
A 59-year-old man was referred to our clinic for a rash, fever, and night sweats following treatment for metastatic seminoma with cisplatin and etoposide. Physical examination revealed indurated erythematous papules and plaques on the trunk and upper and lower extremities, some with annular or arcuate configuration with trailing scale (Figure, A). A skin biopsy demonstrated mild papillary dermal edema with a mixed infiltrate of mononuclear cells, neutrophils, eosinophils, mast cells, lymphocytes, and karyorrhectic debris without evidence of leukocytoclastic vasculitis. The histopathologic differential diagnosis included a histiocytoid variant of Sweet syndrome (SS), and our patient’s rapid clinical response to corticosteroids supported this diagnosis.

With a relapsing and remitting course over 3 years, the rash eventually evolved into more edematous papules and plaques (Figure, B), and a repeat biopsy 3 years later was consistent with classic SS. Although the patient's condition improved with prednisone, attempts to taper prednisone invariably resulted in relapse. Multiple steroid-sparing agents were trialed over the course of 3 years including dapsone and mycophenolate mofetil, both of which resulted in hypersensitivity drug eruptions. Colchicine and methotrexate were ineffective. Thalidomide strongly was considered but ultimately was avoided due to substantial existing neuropathy associated with his prior chemotherapy for metastatic seminoma.
Four years after the initial diagnosis of SS, our patient presented with dyspnea and weight loss. Computed tomography revealed a nearly confluent miliary pattern of nodularity in the lungs. A wedge biopsy demonstrated pneumonitis with intra-alveolar fibrin and neutrophils with a notable absence of granulomatous inflammation. Fungal and acid-fast bacilli staining as well as tissue cultures were negative. He had a history of Mycobacterium kansasii pulmonary infection treated 18 months prior; however, in this instance, the histopathology, negative microbial cultures, and rapid steroid responsiveness were consistent with pulmonary involvement of SS. Over the ensuing 2 years, the patient developed worsening of his chronic anemia. He was diagnosed with myelodysplastic syndrome (MDS) by bone marrow biopsy, despite having a normal bone marrow biopsy more than 3 years prior to evaluate his anemia. At this time, thalidomide was initiated at 50 mg daily leading to notable improvement in his SS symptoms; however, he developed worsening neuropathy resulting in the discontinuation of this treatment 2 months later. An investigational combination of vosaroxin and azacytidine was used to treat his MDS, resulting in normalization of blood counts and remission from SS.
Sweet syndrome may occur in the setting of undiagnosed cancer or may signal the return of a previously treated malignancy. The first description of SS associated with solid tumors was in a patient with testicular cancer,1 which prompted continuous surveillance for recurrent seminoma in our patient, though none was found. Hematologic malignancies, as well as MDS, often are associated with SS.2 In our patient, multiple atypical features linked the development of SS to the ultimate presentation of MDS. The initial finding of a histiocytoid variant has been described in a case series of 9 patients with chronic relapsing SS who eventually developed MDS with latency of up to 7 years. The histopathology in these cases evolved over time to that of classic neutrophilic SS.3 Pulmonary involvement of SS is another interesting aspect of our case. In one analysis, 18 of 34 (53%) cases with pulmonary involvement featured hematologic pathology, including myelodysplasia and acute leukemia.4
In our patient, SS preceded the clinical manifestation of MDS by 6 years. A similar phenomenon has been described in a patient with SS that preceded myelodysplasia by 30 months and was recalcitrant to numerous steroid-sparing therapies except thalidomide, despite the persistence of myelodysplasia. Tapering thalidomide, however, resulted in recurrence of SS lesions in that patient.5 In another case, resolution of myelodysplasia from azacytidine treatment was associated with remission from SS.6
Our case represents a confluence of atypical features that seem to define myelodysplasia-associated SS, including the initial presentation with a clinically atypical histiocytoid variant, chronic relapsing and remitting course, and extracutaneous involvement of the lungs. These findings should prompt surveillance for hematologic malignancy or myelodysplasia. Serial bone marrow biopsies were required to evaluate persistent anemia before the histopathologic findings of MDS became apparent in our patient. Thalidomide was an effective treatment for the cutaneous manifestations in our patient and should be considered as a steroid-sparing agent in the treatment of recalcitrant SS. Despite the discontinuation of thalidomide therapy, effective control of our patient’s myelodysplasia with chemotherapy has kept him in remission from SS for more than 7 years of follow-up, suggesting a causal relationship between these disorders.
- Shapiro L, Baraf CS, Richheimer LL. Sweet’s syndrome (acute febrile neutrophilic dermatosis): report of a case. Arch Dermatol. 1971;103:81-84.
- Cohen PR. Sweet’s syndrome—a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
- Vignon-Pennamen MD, Juillard C, Rybojad M, et al. Chronic recurrent lymphocytic Sweet syndrome as a predictive marker of myelodysplasia. Arch Dermatol. 2006;142:1170-1176.
- Fernandez-Bussy S, Labarca G, Cabello F, et al. Sweet’s syndrome with pulmonary involvement: case report and literature review. Respir Med Case Rep. 2012;6:16-19.
- Browning CE, Dixon DE, Malone JC, et al. Thalidomide in the treatment of recalcitrant Sweet’s syndrome associated with myelodysplasia. J Am Acad Dermatol. 2005;53(2 suppl 1):S135-S138.
- Martinelli S, Rigolin GM, Leo G, et al. Complete remission Sweet’s syndrome after azacytidine treatment for concomitant myelodysplastic syndrome. Int J Hematol. 2014;99:663-667.
- Shapiro L, Baraf CS, Richheimer LL. Sweet’s syndrome (acute febrile neutrophilic dermatosis): report of a case. Arch Dermatol. 1971;103:81-84.
- Cohen PR. Sweet’s syndrome—a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
- Vignon-Pennamen MD, Juillard C, Rybojad M, et al. Chronic recurrent lymphocytic Sweet syndrome as a predictive marker of myelodysplasia. Arch Dermatol. 2006;142:1170-1176.
- Fernandez-Bussy S, Labarca G, Cabello F, et al. Sweet’s syndrome with pulmonary involvement: case report and literature review. Respir Med Case Rep. 2012;6:16-19.
- Browning CE, Dixon DE, Malone JC, et al. Thalidomide in the treatment of recalcitrant Sweet’s syndrome associated with myelodysplasia. J Am Acad Dermatol. 2005;53(2 suppl 1):S135-S138.
- Martinelli S, Rigolin GM, Leo G, et al. Complete remission Sweet’s syndrome after azacytidine treatment for concomitant myelodysplastic syndrome. Int J Hematol. 2014;99:663-667.
Practice Points
- Sweet syndrome is characterized by the clinical constellation of fever, a skin eruption of tender erythematous papules or plaques, and response to corticosteroids.
- Skin biopsy characteristically demonstrates marked papillary dermal edema with a dense infiltrate of mature neutrophils without leukocytoclasia.
- Sweet syndrome often is idiopathic, though it has been associated with infection, autoimmunity, medication, and malignancy.