User login
ACIP Recommends PCV13 for All Adults 65 and Up
All adults who are 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) routinely in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23), according to a new recommendation from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The recommendation appears in the Sept. 19 issue of Morbidity and Mortality Weekly Report.
The ACIP recommendation calls for pneumococcal vaccine-naive adults aged 65 and older to receive one dose of PCV13 vaccine, followed by a dose of PPSV23 6-12 months later (MMWR 2014:63;822-5). Older adults who have previously received only PPSV23 should receive a dose of PCV13 at least 12 months later, wrote Sara Tomczyk of the CDC and her associates.
ACIP has recommended PPSV23 for older adults since 2010. In 2012, the committee made its first recommendation for PCV13, targeting patients 19 years and older who have immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leak, or cochlear implants. The new PCV13 recommendation for all older adults is based on a randomized, placebo-controlled trial of the vaccine in about 85,000 adults aged 65 years and older in the Netherlands who had no prior pneumococcal vaccine exposure. The vaccine showed a moderate level of evidence for efficacy against community-acquired pneumonia in this cohort, ACIP determined. Efficacy against nonbacteremic vaccine-type pneumococcal pneumonia was about 45%, while efficacy against vaccine-type invasive pneumococcal disease was about 75%, the reviewers wrote.
ACIP will reevaluate the recommendations in 2018.
Ms. Tomczyk and her colleagues disclosed no funding sources or conflicts of interest.
All adults who are 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) routinely in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23), according to a new recommendation from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The recommendation appears in the Sept. 19 issue of Morbidity and Mortality Weekly Report.
The ACIP recommendation calls for pneumococcal vaccine-naive adults aged 65 and older to receive one dose of PCV13 vaccine, followed by a dose of PPSV23 6-12 months later (MMWR 2014:63;822-5). Older adults who have previously received only PPSV23 should receive a dose of PCV13 at least 12 months later, wrote Sara Tomczyk of the CDC and her associates.
ACIP has recommended PPSV23 for older adults since 2010. In 2012, the committee made its first recommendation for PCV13, targeting patients 19 years and older who have immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leak, or cochlear implants. The new PCV13 recommendation for all older adults is based on a randomized, placebo-controlled trial of the vaccine in about 85,000 adults aged 65 years and older in the Netherlands who had no prior pneumococcal vaccine exposure. The vaccine showed a moderate level of evidence for efficacy against community-acquired pneumonia in this cohort, ACIP determined. Efficacy against nonbacteremic vaccine-type pneumococcal pneumonia was about 45%, while efficacy against vaccine-type invasive pneumococcal disease was about 75%, the reviewers wrote.
ACIP will reevaluate the recommendations in 2018.
Ms. Tomczyk and her colleagues disclosed no funding sources or conflicts of interest.
All adults who are 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) routinely in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23), according to a new recommendation from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The recommendation appears in the Sept. 19 issue of Morbidity and Mortality Weekly Report.
The ACIP recommendation calls for pneumococcal vaccine-naive adults aged 65 and older to receive one dose of PCV13 vaccine, followed by a dose of PPSV23 6-12 months later (MMWR 2014:63;822-5). Older adults who have previously received only PPSV23 should receive a dose of PCV13 at least 12 months later, wrote Sara Tomczyk of the CDC and her associates.
ACIP has recommended PPSV23 for older adults since 2010. In 2012, the committee made its first recommendation for PCV13, targeting patients 19 years and older who have immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leak, or cochlear implants. The new PCV13 recommendation for all older adults is based on a randomized, placebo-controlled trial of the vaccine in about 85,000 adults aged 65 years and older in the Netherlands who had no prior pneumococcal vaccine exposure. The vaccine showed a moderate level of evidence for efficacy against community-acquired pneumonia in this cohort, ACIP determined. Efficacy against nonbacteremic vaccine-type pneumococcal pneumonia was about 45%, while efficacy against vaccine-type invasive pneumococcal disease was about 75%, the reviewers wrote.
ACIP will reevaluate the recommendations in 2018.
Ms. Tomczyk and her colleagues disclosed no funding sources or conflicts of interest.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Gold and Nickel Lead List of Eyelid Irritants
CHICAGO – Chances are good that if your patient presents with eyelid dermatitis, allergic contact with gold or nickel is the culprit.
"Gold is not thought to be easily released from jewelry, which would be the typical exposure with eyelids, unless it comes into contact with sweat, friction, or abrasives," Dr. Amber Reck Atwater told attendees of a session on facial dermatoses at the American Academy of Dermatology summer meeting.
However, when gold comes into contact with titanium dioxide, a common active ingredient in many cosmetics such as eye shadow, patients are at risk for eyelid irritation.
"If I am wearing a gold ring, and I put this on my eyelids using my finger, the gold will be more easily released, and I will be more likely to get a reaction on my lids," said Dr. Atwater, of the department of dermatology at Duke University, Durham, N.C.
Nickel is another leading cause of eyelid dermatitis, Dr. Atwater said. She warned of the metal’s pernicious tendency to hide in personal care products such as eyelash curlers that do not list it as an active or inactive ingredient.
A simple and relatively inexpensive dimethylglyoxime test, which Dr. Atwater said can be purchased on the consumer market, can help identify items that may contain nickel. Rub a drop of dimethylglyoxime onto an item, such as a house key, with a cotton swab. If the key turns bright pink, then you know it has nickel in it.
"So you can imagine that if I am holding my keys in my hands, I could be transferring nickel from the keys to my eyelids," said Dr. Atwater.
Other potential sources of nickel include faucets, sunglasses with metal frames, barbells, and other weight-lifting equipment.
Other common causes of eyelid dermatitis are products that contain fragrance, including balsam of Peru, neomycin (typically found in antibacterial eye drops), formaldehyde and bronopol (preservatives that are found in certain cosmetics), skin care products, and topicals.
Allergic contact dermatitis in the eyelid can present in an upper, lower, unilateral, or bilateral fashion on the eyelids alone, but typically presents in combination with dermatitis on other areas of the face, or even other areas of the body, according to Dr. Atwater.
"You should be highly suspicious that it’s contact dermatitis when you see eyelid dermatitis with other parts of the body involved," she said.
When the dermatitis presents in the eyelids alone, other factors such as seborrheic dermatitis or aspecific xerotic dermatitis could be the cause.
"Still, a good 30%-50% of our patients will have allergic contact dermatitis when we see them with eyelid dermatitis alone," Dr. Atwater said.
The eyelids are particularly susceptible to irritation in part because the skin is extremely thin – 0.55 mm – compared with other facial areas where the average skin thickness is about 2 mm, Dr. Atwater explained.
"And it’s really easy to transfer substances from our hands to our eyes. People rub their eyes and their faces a lot throughout the day," she noted.
Dr. Atwater had no financial conflicts to disclose.
On Twitter @whitneymcknight
CHICAGO – Chances are good that if your patient presents with eyelid dermatitis, allergic contact with gold or nickel is the culprit.
"Gold is not thought to be easily released from jewelry, which would be the typical exposure with eyelids, unless it comes into contact with sweat, friction, or abrasives," Dr. Amber Reck Atwater told attendees of a session on facial dermatoses at the American Academy of Dermatology summer meeting.
However, when gold comes into contact with titanium dioxide, a common active ingredient in many cosmetics such as eye shadow, patients are at risk for eyelid irritation.
"If I am wearing a gold ring, and I put this on my eyelids using my finger, the gold will be more easily released, and I will be more likely to get a reaction on my lids," said Dr. Atwater, of the department of dermatology at Duke University, Durham, N.C.
Nickel is another leading cause of eyelid dermatitis, Dr. Atwater said. She warned of the metal’s pernicious tendency to hide in personal care products such as eyelash curlers that do not list it as an active or inactive ingredient.
A simple and relatively inexpensive dimethylglyoxime test, which Dr. Atwater said can be purchased on the consumer market, can help identify items that may contain nickel. Rub a drop of dimethylglyoxime onto an item, such as a house key, with a cotton swab. If the key turns bright pink, then you know it has nickel in it.
"So you can imagine that if I am holding my keys in my hands, I could be transferring nickel from the keys to my eyelids," said Dr. Atwater.
Other potential sources of nickel include faucets, sunglasses with metal frames, barbells, and other weight-lifting equipment.
Other common causes of eyelid dermatitis are products that contain fragrance, including balsam of Peru, neomycin (typically found in antibacterial eye drops), formaldehyde and bronopol (preservatives that are found in certain cosmetics), skin care products, and topicals.
Allergic contact dermatitis in the eyelid can present in an upper, lower, unilateral, or bilateral fashion on the eyelids alone, but typically presents in combination with dermatitis on other areas of the face, or even other areas of the body, according to Dr. Atwater.
"You should be highly suspicious that it’s contact dermatitis when you see eyelid dermatitis with other parts of the body involved," she said.
When the dermatitis presents in the eyelids alone, other factors such as seborrheic dermatitis or aspecific xerotic dermatitis could be the cause.
"Still, a good 30%-50% of our patients will have allergic contact dermatitis when we see them with eyelid dermatitis alone," Dr. Atwater said.
The eyelids are particularly susceptible to irritation in part because the skin is extremely thin – 0.55 mm – compared with other facial areas where the average skin thickness is about 2 mm, Dr. Atwater explained.
"And it’s really easy to transfer substances from our hands to our eyes. People rub their eyes and their faces a lot throughout the day," she noted.
Dr. Atwater had no financial conflicts to disclose.
On Twitter @whitneymcknight
CHICAGO – Chances are good that if your patient presents with eyelid dermatitis, allergic contact with gold or nickel is the culprit.
"Gold is not thought to be easily released from jewelry, which would be the typical exposure with eyelids, unless it comes into contact with sweat, friction, or abrasives," Dr. Amber Reck Atwater told attendees of a session on facial dermatoses at the American Academy of Dermatology summer meeting.
However, when gold comes into contact with titanium dioxide, a common active ingredient in many cosmetics such as eye shadow, patients are at risk for eyelid irritation.
"If I am wearing a gold ring, and I put this on my eyelids using my finger, the gold will be more easily released, and I will be more likely to get a reaction on my lids," said Dr. Atwater, of the department of dermatology at Duke University, Durham, N.C.
Nickel is another leading cause of eyelid dermatitis, Dr. Atwater said. She warned of the metal’s pernicious tendency to hide in personal care products such as eyelash curlers that do not list it as an active or inactive ingredient.
A simple and relatively inexpensive dimethylglyoxime test, which Dr. Atwater said can be purchased on the consumer market, can help identify items that may contain nickel. Rub a drop of dimethylglyoxime onto an item, such as a house key, with a cotton swab. If the key turns bright pink, then you know it has nickel in it.
"So you can imagine that if I am holding my keys in my hands, I could be transferring nickel from the keys to my eyelids," said Dr. Atwater.
Other potential sources of nickel include faucets, sunglasses with metal frames, barbells, and other weight-lifting equipment.
Other common causes of eyelid dermatitis are products that contain fragrance, including balsam of Peru, neomycin (typically found in antibacterial eye drops), formaldehyde and bronopol (preservatives that are found in certain cosmetics), skin care products, and topicals.
Allergic contact dermatitis in the eyelid can present in an upper, lower, unilateral, or bilateral fashion on the eyelids alone, but typically presents in combination with dermatitis on other areas of the face, or even other areas of the body, according to Dr. Atwater.
"You should be highly suspicious that it’s contact dermatitis when you see eyelid dermatitis with other parts of the body involved," she said.
When the dermatitis presents in the eyelids alone, other factors such as seborrheic dermatitis or aspecific xerotic dermatitis could be the cause.
"Still, a good 30%-50% of our patients will have allergic contact dermatitis when we see them with eyelid dermatitis alone," Dr. Atwater said.
The eyelids are particularly susceptible to irritation in part because the skin is extremely thin – 0.55 mm – compared with other facial areas where the average skin thickness is about 2 mm, Dr. Atwater explained.
"And it’s really easy to transfer substances from our hands to our eyes. People rub their eyes and their faces a lot throughout the day," she noted.
Dr. Atwater had no financial conflicts to disclose.
On Twitter @whitneymcknight
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2014
Sleeping on Animal Skins Might Protect Against Childhood Asthma, Hay Fever
Babies who slept on animal skins during their first 3 months of life were almost 40% less likely to have asthma by the time they were 10 years old, according to a population-based cohort study.
Sleeping on animal skins during infancy also was linked to lower odds of wheezing and hay fever, but did not seem to affect eczema or sensitivity to airborne antigens, Dr. Christina Tischer reported at the annual meeting of the European Respiratory Society.
"Early exposure to animal fur could be a simple, cheap, and effective way to resemble an environment with higher microbial exposure," said Dr. Tischer, a researcher at the German Institute for Environmental Health in Neuherberg, Germany. "It might follow similar protective mechanisms in relation to asthma and allergy as it has been observed in farm and rural environments."
The investigators studied 2,441 children in Germany who were up to 10 years old; parents answered a series of questionnaires about asthma and respiratory risk factors and health outcomes. In all, 55% of the children slept on animal skins or animal furs during their first 3 months of life, Dr. Tischer and her associates reported.
By age 10 years, children who slept on animal skins or animal fur as infants had a 25% lower odds of ever having wheezed (adjusted odds ratio, 0.75), a 38% lower odds of having been diagnosed with asthma (aOR, 0.62), and a 35% lower odds of having been diagnosed with hay fever (aOR, 0.65) compared with children who did not sleep on animals skins or furs as infants, the investigators reported.
Funding information for the study was not available. Dr. Tischer reported no conflicts of interest.
Babies who slept on animal skins during their first 3 months of life were almost 40% less likely to have asthma by the time they were 10 years old, according to a population-based cohort study.
Sleeping on animal skins during infancy also was linked to lower odds of wheezing and hay fever, but did not seem to affect eczema or sensitivity to airborne antigens, Dr. Christina Tischer reported at the annual meeting of the European Respiratory Society.
"Early exposure to animal fur could be a simple, cheap, and effective way to resemble an environment with higher microbial exposure," said Dr. Tischer, a researcher at the German Institute for Environmental Health in Neuherberg, Germany. "It might follow similar protective mechanisms in relation to asthma and allergy as it has been observed in farm and rural environments."
The investigators studied 2,441 children in Germany who were up to 10 years old; parents answered a series of questionnaires about asthma and respiratory risk factors and health outcomes. In all, 55% of the children slept on animal skins or animal furs during their first 3 months of life, Dr. Tischer and her associates reported.
By age 10 years, children who slept on animal skins or animal fur as infants had a 25% lower odds of ever having wheezed (adjusted odds ratio, 0.75), a 38% lower odds of having been diagnosed with asthma (aOR, 0.62), and a 35% lower odds of having been diagnosed with hay fever (aOR, 0.65) compared with children who did not sleep on animals skins or furs as infants, the investigators reported.
Funding information for the study was not available. Dr. Tischer reported no conflicts of interest.
Babies who slept on animal skins during their first 3 months of life were almost 40% less likely to have asthma by the time they were 10 years old, according to a population-based cohort study.
Sleeping on animal skins during infancy also was linked to lower odds of wheezing and hay fever, but did not seem to affect eczema or sensitivity to airborne antigens, Dr. Christina Tischer reported at the annual meeting of the European Respiratory Society.
"Early exposure to animal fur could be a simple, cheap, and effective way to resemble an environment with higher microbial exposure," said Dr. Tischer, a researcher at the German Institute for Environmental Health in Neuherberg, Germany. "It might follow similar protective mechanisms in relation to asthma and allergy as it has been observed in farm and rural environments."
The investigators studied 2,441 children in Germany who were up to 10 years old; parents answered a series of questionnaires about asthma and respiratory risk factors and health outcomes. In all, 55% of the children slept on animal skins or animal furs during their first 3 months of life, Dr. Tischer and her associates reported.
By age 10 years, children who slept on animal skins or animal fur as infants had a 25% lower odds of ever having wheezed (adjusted odds ratio, 0.75), a 38% lower odds of having been diagnosed with asthma (aOR, 0.62), and a 35% lower odds of having been diagnosed with hay fever (aOR, 0.65) compared with children who did not sleep on animals skins or furs as infants, the investigators reported.
Funding information for the study was not available. Dr. Tischer reported no conflicts of interest.
FROM THE EUROPEAN RESPIRATORY SOCIETY INTERNATIONAL CONGRESS
Pneumococcal Vaccine Protects Against Cardiac and Cerebrovascular Events
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
AT THE ESC CONGRESS 2014
Itch–Scratch–Itch: Can the Cycle Be Broken?
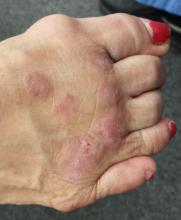
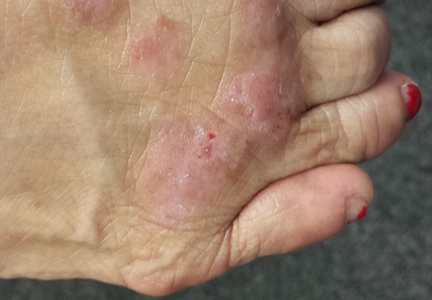
A 67-year-old woman has had a very itchy rash on the dorsa of both feet for almost a year. In addition to consulting her primary care provider, she has presented to a number of medical venues, including urgent care clinics. Different products have been prescribed—including clotrimazole cream, nystatin powder, and OTC hydrocortisone 1% cream—none of which produced any beneficial effect. So the patient finally self-refers to dermatology.
She reports that at one point, she was convinced her shoes were the source of the problem. But trying new shoes and even going entirely barefoot during a two-week vacation at the beach made no difference.
The patient admits that it is “impossible” to leave the lesions alone, because they are so itchy. She knows that “scratching can’t be good,” so she tries to just rub them, often with a wet washcloth.
Aside from the foot rash, her health is excellent. Her only medications are NSAIDs for mild arthritis.
EXAMINATION
The lesions are confined to the forefeet. There are about five on the right foot and three on the left. The lesions are dark purplish round plaques with planar surfaces that are shiny but have a white frosting-like finish. On average, they measure 1.8 cm in diameter. No surrounding inflammation is appreciated. The patient has otherwise unremarkable type IV skin.
What is the diagnosis?
DIAGNOSIS
Punch biopsy confirms the expected diagnosis of lichen planus.
DISCUSSION
Lichen planus (LP) is a very common problem seen in dermatology offices worldwide. LP represents an immune response of unknown origin and may be found in association with other diseases of altered immunity (eg, dermatomyositis, alopecia areata, vitiligo, morphea, and myasthenia gravis). Some studies support the theory that LP is caused by hepatitis C.
The most common forms of LP lend themselves to a useful mnemonic device that uses the letter “P” to describe common features of the disease: purple, papular or plaquish, planar (flat) surfaces, polygonal shapes, pruritic, penile (frequent location), and finally, “puzzling” to the clinician.
In contrast to this particular case, LP commonly affects flexural surfaces, such as the volar wrist. LP can also affect nails (with dystrophy or onycholysis), the scalp, and, most notoriously, the oral mucosae, where it can cause ulcerations and intense burning. Oral lesions often present with a lacy white look on the buccal mucosal surfaces.
LP is only one of a number of skin diseases that “koebnerize” (ie, form along lines of trauma, such as a scratch). The resulting linear collection of planar purple papules—known as the Koebner phenomenon—is useful for diagnosis.
Biopsy is often needed to confirm the diagnosis. It will show hyperkeratotic epidermis with irregular acanthosis. In the upper dermis, there is often an infiltrating band of lymphocytic and histiocytic cells, along with many Langerhans cells, that effectively obliterates the dermo-epidermal junction (a pathognomic finding of LP).
Most cases of LP eventually resolve, usually within months, though some can persist for years. Treatment can be problematic, especially when the disease is widespread or manifests in difficult areas, such as the mouth or scalp. This particular patient’s problem was relatively simple to treat with topical clobetasol cream under occlusion (bid for three weeks). Had that not worked, we could have tried intralesional injection of triamcinolone (10 mg per cc).
This case was typical of LP seen on the legs of older patients with darker skin. In these patients, the lesions tend to become hypertrophic and darker than the usual light pink to purple seen in those with fairer skin.
The differential for LP includes psoriasis, fixed-drug eruption, granuloma annulare, and nummular eczema.
TAKE-HOME LEARNING POINTS
• Lichen planus (LP) is a commonly encountered inflammatory condition that classically affects flexural skin, such as the volar wrist.
• LP lesions can often be seen in a linear configuration, following the line of a scratch or other trauma, a phenomenon known as the Koebner phenomenon (the isomorphic linear response), which can be helpful diagnostically.
• The “Ps” of LP include papular, purple, planar, plaquish, pruritic, penile, polygonal, and puzzling.
• LP can also affect hair follicles, nails, and oral mucosa.
• LP can present with hypertrophic plaques, especially on legs and feet.
A 67-year-old woman has had a very itchy rash on the dorsa of both feet for almost a year. In addition to consulting her primary care provider, she has presented to a number of medical venues, including urgent care clinics. Different products have been prescribed—including clotrimazole cream, nystatin powder, and OTC hydrocortisone 1% cream—none of which produced any beneficial effect. So the patient finally self-refers to dermatology.
She reports that at one point, she was convinced her shoes were the source of the problem. But trying new shoes and even going entirely barefoot during a two-week vacation at the beach made no difference.
The patient admits that it is “impossible” to leave the lesions alone, because they are so itchy. She knows that “scratching can’t be good,” so she tries to just rub them, often with a wet washcloth.
Aside from the foot rash, her health is excellent. Her only medications are NSAIDs for mild arthritis.
EXAMINATION
The lesions are confined to the forefeet. There are about five on the right foot and three on the left. The lesions are dark purplish round plaques with planar surfaces that are shiny but have a white frosting-like finish. On average, they measure 1.8 cm in diameter. No surrounding inflammation is appreciated. The patient has otherwise unremarkable type IV skin.
What is the diagnosis?
DIAGNOSIS
Punch biopsy confirms the expected diagnosis of lichen planus.
DISCUSSION
Lichen planus (LP) is a very common problem seen in dermatology offices worldwide. LP represents an immune response of unknown origin and may be found in association with other diseases of altered immunity (eg, dermatomyositis, alopecia areata, vitiligo, morphea, and myasthenia gravis). Some studies support the theory that LP is caused by hepatitis C.
The most common forms of LP lend themselves to a useful mnemonic device that uses the letter “P” to describe common features of the disease: purple, papular or plaquish, planar (flat) surfaces, polygonal shapes, pruritic, penile (frequent location), and finally, “puzzling” to the clinician.
In contrast to this particular case, LP commonly affects flexural surfaces, such as the volar wrist. LP can also affect nails (with dystrophy or onycholysis), the scalp, and, most notoriously, the oral mucosae, where it can cause ulcerations and intense burning. Oral lesions often present with a lacy white look on the buccal mucosal surfaces.
LP is only one of a number of skin diseases that “koebnerize” (ie, form along lines of trauma, such as a scratch). The resulting linear collection of planar purple papules—known as the Koebner phenomenon—is useful for diagnosis.
Biopsy is often needed to confirm the diagnosis. It will show hyperkeratotic epidermis with irregular acanthosis. In the upper dermis, there is often an infiltrating band of lymphocytic and histiocytic cells, along with many Langerhans cells, that effectively obliterates the dermo-epidermal junction (a pathognomic finding of LP).
Most cases of LP eventually resolve, usually within months, though some can persist for years. Treatment can be problematic, especially when the disease is widespread or manifests in difficult areas, such as the mouth or scalp. This particular patient’s problem was relatively simple to treat with topical clobetasol cream under occlusion (bid for three weeks). Had that not worked, we could have tried intralesional injection of triamcinolone (10 mg per cc).
This case was typical of LP seen on the legs of older patients with darker skin. In these patients, the lesions tend to become hypertrophic and darker than the usual light pink to purple seen in those with fairer skin.
The differential for LP includes psoriasis, fixed-drug eruption, granuloma annulare, and nummular eczema.
TAKE-HOME LEARNING POINTS
• Lichen planus (LP) is a commonly encountered inflammatory condition that classically affects flexural skin, such as the volar wrist.
• LP lesions can often be seen in a linear configuration, following the line of a scratch or other trauma, a phenomenon known as the Koebner phenomenon (the isomorphic linear response), which can be helpful diagnostically.
• The “Ps” of LP include papular, purple, planar, plaquish, pruritic, penile, polygonal, and puzzling.
• LP can also affect hair follicles, nails, and oral mucosa.
• LP can present with hypertrophic plaques, especially on legs and feet.
A 67-year-old woman has had a very itchy rash on the dorsa of both feet for almost a year. In addition to consulting her primary care provider, she has presented to a number of medical venues, including urgent care clinics. Different products have been prescribed—including clotrimazole cream, nystatin powder, and OTC hydrocortisone 1% cream—none of which produced any beneficial effect. So the patient finally self-refers to dermatology.
She reports that at one point, she was convinced her shoes were the source of the problem. But trying new shoes and even going entirely barefoot during a two-week vacation at the beach made no difference.
The patient admits that it is “impossible” to leave the lesions alone, because they are so itchy. She knows that “scratching can’t be good,” so she tries to just rub them, often with a wet washcloth.
Aside from the foot rash, her health is excellent. Her only medications are NSAIDs for mild arthritis.
EXAMINATION
The lesions are confined to the forefeet. There are about five on the right foot and three on the left. The lesions are dark purplish round plaques with planar surfaces that are shiny but have a white frosting-like finish. On average, they measure 1.8 cm in diameter. No surrounding inflammation is appreciated. The patient has otherwise unremarkable type IV skin.
What is the diagnosis?
DIAGNOSIS
Punch biopsy confirms the expected diagnosis of lichen planus.
DISCUSSION
Lichen planus (LP) is a very common problem seen in dermatology offices worldwide. LP represents an immune response of unknown origin and may be found in association with other diseases of altered immunity (eg, dermatomyositis, alopecia areata, vitiligo, morphea, and myasthenia gravis). Some studies support the theory that LP is caused by hepatitis C.
The most common forms of LP lend themselves to a useful mnemonic device that uses the letter “P” to describe common features of the disease: purple, papular or plaquish, planar (flat) surfaces, polygonal shapes, pruritic, penile (frequent location), and finally, “puzzling” to the clinician.
In contrast to this particular case, LP commonly affects flexural surfaces, such as the volar wrist. LP can also affect nails (with dystrophy or onycholysis), the scalp, and, most notoriously, the oral mucosae, where it can cause ulcerations and intense burning. Oral lesions often present with a lacy white look on the buccal mucosal surfaces.
LP is only one of a number of skin diseases that “koebnerize” (ie, form along lines of trauma, such as a scratch). The resulting linear collection of planar purple papules—known as the Koebner phenomenon—is useful for diagnosis.
Biopsy is often needed to confirm the diagnosis. It will show hyperkeratotic epidermis with irregular acanthosis. In the upper dermis, there is often an infiltrating band of lymphocytic and histiocytic cells, along with many Langerhans cells, that effectively obliterates the dermo-epidermal junction (a pathognomic finding of LP).
Most cases of LP eventually resolve, usually within months, though some can persist for years. Treatment can be problematic, especially when the disease is widespread or manifests in difficult areas, such as the mouth or scalp. This particular patient’s problem was relatively simple to treat with topical clobetasol cream under occlusion (bid for three weeks). Had that not worked, we could have tried intralesional injection of triamcinolone (10 mg per cc).
This case was typical of LP seen on the legs of older patients with darker skin. In these patients, the lesions tend to become hypertrophic and darker than the usual light pink to purple seen in those with fairer skin.
The differential for LP includes psoriasis, fixed-drug eruption, granuloma annulare, and nummular eczema.
TAKE-HOME LEARNING POINTS
• Lichen planus (LP) is a commonly encountered inflammatory condition that classically affects flexural skin, such as the volar wrist.
• LP lesions can often be seen in a linear configuration, following the line of a scratch or other trauma, a phenomenon known as the Koebner phenomenon (the isomorphic linear response), which can be helpful diagnostically.
• The “Ps” of LP include papular, purple, planar, plaquish, pruritic, penile, polygonal, and puzzling.
• LP can also affect hair follicles, nails, and oral mucosa.
• LP can present with hypertrophic plaques, especially on legs and feet.
Think Twice About Nebulizers for Asthma Attacks
PRACTICE CHANGER
Stop ordering nebulizers to deliver β-agonists to patients older than 2 who have mild or moderate asthma exacerbations. A metered-dose inhaler (MDI) with a spacer produces the same benefits with fewer adverse effects.1
STRENGTH OF RECOMMENDATION
A: Based on an updated Cochrane meta-analysis of 39 randomized controlled trials (RCTs). 1
ILLUSTRATIVE CASE
A 6-year-old girl with a history of reactive airway disease comes to your office complaining of cough and wheezing. On exam, she has mild retractions, a respiratory rate of 35 breaths/min, and an O2 saturation of 96% on room air. Her lung fields are diffusely wheezy. Her parents would like to keep her out of the hospital. How should you order her albuterol to decrease her wheezing and minimize adverse effects?
Asthma affects nearly 19 million adults and 7 million children in the United States.2 Asthma exacerbations are the third most common reason for hospitalization in children.2,3 Treatment usually requires multiple agents, including inhaled β-agonists. These are most effective when delivered to the peripheral airways, which is a challenge during an asthma exacerbation because of airway swelling and rapid breathing. Two devices have been developed to effectively deliver medication to the peripheral airways: nebulizers and MDIs with a holding chamber (spacer).1
Several studies have demonstrated that for mild to moderate asthma exacerbations, administering a β-agonist via an MDI with a spacer is as effective as using a nebulizer.4,5 Asthma treatment guidelines also state that spacers are either comparable or preferable to nebulizers for β-agonist administration in children and adults.6,7 However, based on our experience, clinicians still frequently order nebulizer treatments for patients with asthma exacerbations, despite several advantages of MDIs with spacers. Notably, they cost less and don’t require maintenance or a power source. Clinicians administered nebulizer therapy at more than 3.6 million emergency department (ED) visits in 2006.8
In this latest Cochrane review, Cates et al1 added four new studies to those included in their earlier Cochrane meta-analysis and evaluated what, if any, effect these studies had on our understanding of nebulizers versus MDIs with spacers.
STUDY SUMMARY
Outcomes with nebulizers are no better than those with spacers
This systematic review and meta-analysis pooled the results of RCTs comparing spacers to nebulizers for administering β-agonists during acute, non–life-threatening asthma exacerbations.1 The authors reviewed studies conducted in EDs, hospitals, and outpatient settings that included children and adults. The primary outcomes were hospital admission rates and duration of hospital stay. Secondary outcomes included time spent in the ED, change in pulse rate, and incidence of tremor.
Cates et al1 analyzed 39 trials that included 1,897 children and 729 adults and were conducted primarily in an ED or outpatient setting. The four new studies added 295 children and 58 adults to the researchers’ earlier meta-analysis. Studies involving adults and children were pooled separately. Most patients received multiple treatments with β-agonists titrated to the individual’s response.
No differences in hospitalizations. Rates of hospital admissions did not differ between patients receiving β-agonists via a spacer compared to a nebulizer in both adults (relative risk [RR] = 0.94) and children (RR = 0.71). Duration of hospital stay did not differ between the two delivery methods in adults (mean difference [MD] = –0.60 d) and children (MD = 0.33 d).
For kids, spacers meant less time in the ED. Duration in the ED was approximately half an hour shorter for children using spacers (MD = –33.48 min). There was no difference observed in adults (MD = 1.75 min). The rate of tremor was lower in children using spacers (RR = 0.64) and was similar in adults (RR = 1.12). The rise in pulse rate was lower in children using spacers
(MD = –5.41% change from baseline) and was similar in adults (MD = –1.23%).
On the next page: What's new and challenges to implementation >>
WHAT’S NEW
Additional evidence that spacers are as effective as nebulizers
This meta-analysis, which included four new studies, should finally dispel the myth that nebulizers deliver β-agonists more effectively than MDIs with spacers. Additionally, in children, spacers are associated with lower rates of adverse effects, including tremor and elevated pulse rate.
CAVEATS
Most studies involving children were open label
Although most of the adult trials in this meta-analysis involved a double-dummy design, which allows for effective participant blinding, most of the studies involving children were open label. This open-label design might have been a source of reporting bias for symptom-related outcomes but should not have affected hospital admission rates or duration of hospital stay.
In the double-dummy studies, adults received both a nebulizer and a spacer, which likely explains the similar time spent in the ED by the treatment and control groups.
CHALLENGES TO IMPLEMENTATION
Old habits are hard to break
Clinicians may think that patients view nebulizers as more potent or more effective than spacers and thus be more likely to order them. Some patients may prefer nebulizers because of convenience or other factors.
REFERENCES
1. Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2013;9: CD000052.
2. Barrett ML, Wier LM, Washington R. Trends in pediatric and adult hospital stays for asthma, 2000-2010. HCUP Statistical Brief #169. www.hcup-us.ahrq.gov/reports/stat briefs/sb169-Asthma-Trends-Hospital-Stays.pdf. Accessed June 16, 2014.
3. Pfuntner A, Wier LM, Stocks C. Most frequent conditions in US hospitals, 2011. HCUP Statistical Brief #162. www.hcup-us.ahrq.gov/reports/statbriefs/sb162.pdf. Accessed June 16, 2014.
4. Cates CJ, Crilly JA, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2006;(2): CD000052.
5. Turner MO, Patel A, Ginsburg S, et al. Bronchodilator delivery in acute airflow obstruction: a meta-analysis. Arch Intern Med. 1997;157:1736-1744.
6. National Heart, Lung, and Blood Institute Expert Panel Report 3 (EPR3): Guidelines for the diagnosis and management of asthma. www.nhlbi.nih.gov/guidelines/asthma/asth gdln.htm. Accessed June 16, 2014.
7. British Thoracic Society. British guideline of the management of asthma: a national clinical guideline. www.brit-thoracic.org.uk/document-library/clinical-information/asth ma/btssign-guideline-on-the-management-of-asthma/. Accessed June 16, 2014.
8. Pitts SR, Niska RW, Xu J, et al. National Hospital Ambulatory Medical Care Survey: 2006 emergency department summary. www.cdc.gov/nchs/data/nhsr/nhsr007.pdf. Accessed June 16, 2014.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Copyright © 2014. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice. 2014;63(6):321-322, 346.
PRACTICE CHANGER
Stop ordering nebulizers to deliver β-agonists to patients older than 2 who have mild or moderate asthma exacerbations. A metered-dose inhaler (MDI) with a spacer produces the same benefits with fewer adverse effects.1
STRENGTH OF RECOMMENDATION
A: Based on an updated Cochrane meta-analysis of 39 randomized controlled trials (RCTs). 1
ILLUSTRATIVE CASE
A 6-year-old girl with a history of reactive airway disease comes to your office complaining of cough and wheezing. On exam, she has mild retractions, a respiratory rate of 35 breaths/min, and an O2 saturation of 96% on room air. Her lung fields are diffusely wheezy. Her parents would like to keep her out of the hospital. How should you order her albuterol to decrease her wheezing and minimize adverse effects?
Asthma affects nearly 19 million adults and 7 million children in the United States.2 Asthma exacerbations are the third most common reason for hospitalization in children.2,3 Treatment usually requires multiple agents, including inhaled β-agonists. These are most effective when delivered to the peripheral airways, which is a challenge during an asthma exacerbation because of airway swelling and rapid breathing. Two devices have been developed to effectively deliver medication to the peripheral airways: nebulizers and MDIs with a holding chamber (spacer).1
Several studies have demonstrated that for mild to moderate asthma exacerbations, administering a β-agonist via an MDI with a spacer is as effective as using a nebulizer.4,5 Asthma treatment guidelines also state that spacers are either comparable or preferable to nebulizers for β-agonist administration in children and adults.6,7 However, based on our experience, clinicians still frequently order nebulizer treatments for patients with asthma exacerbations, despite several advantages of MDIs with spacers. Notably, they cost less and don’t require maintenance or a power source. Clinicians administered nebulizer therapy at more than 3.6 million emergency department (ED) visits in 2006.8
In this latest Cochrane review, Cates et al1 added four new studies to those included in their earlier Cochrane meta-analysis and evaluated what, if any, effect these studies had on our understanding of nebulizers versus MDIs with spacers.
STUDY SUMMARY
Outcomes with nebulizers are no better than those with spacers
This systematic review and meta-analysis pooled the results of RCTs comparing spacers to nebulizers for administering β-agonists during acute, non–life-threatening asthma exacerbations.1 The authors reviewed studies conducted in EDs, hospitals, and outpatient settings that included children and adults. The primary outcomes were hospital admission rates and duration of hospital stay. Secondary outcomes included time spent in the ED, change in pulse rate, and incidence of tremor.
Cates et al1 analyzed 39 trials that included 1,897 children and 729 adults and were conducted primarily in an ED or outpatient setting. The four new studies added 295 children and 58 adults to the researchers’ earlier meta-analysis. Studies involving adults and children were pooled separately. Most patients received multiple treatments with β-agonists titrated to the individual’s response.
No differences in hospitalizations. Rates of hospital admissions did not differ between patients receiving β-agonists via a spacer compared to a nebulizer in both adults (relative risk [RR] = 0.94) and children (RR = 0.71). Duration of hospital stay did not differ between the two delivery methods in adults (mean difference [MD] = –0.60 d) and children (MD = 0.33 d).
For kids, spacers meant less time in the ED. Duration in the ED was approximately half an hour shorter for children using spacers (MD = –33.48 min). There was no difference observed in adults (MD = 1.75 min). The rate of tremor was lower in children using spacers (RR = 0.64) and was similar in adults (RR = 1.12). The rise in pulse rate was lower in children using spacers
(MD = –5.41% change from baseline) and was similar in adults (MD = –1.23%).
On the next page: What's new and challenges to implementation >>
WHAT’S NEW
Additional evidence that spacers are as effective as nebulizers
This meta-analysis, which included four new studies, should finally dispel the myth that nebulizers deliver β-agonists more effectively than MDIs with spacers. Additionally, in children, spacers are associated with lower rates of adverse effects, including tremor and elevated pulse rate.
CAVEATS
Most studies involving children were open label
Although most of the adult trials in this meta-analysis involved a double-dummy design, which allows for effective participant blinding, most of the studies involving children were open label. This open-label design might have been a source of reporting bias for symptom-related outcomes but should not have affected hospital admission rates or duration of hospital stay.
In the double-dummy studies, adults received both a nebulizer and a spacer, which likely explains the similar time spent in the ED by the treatment and control groups.
CHALLENGES TO IMPLEMENTATION
Old habits are hard to break
Clinicians may think that patients view nebulizers as more potent or more effective than spacers and thus be more likely to order them. Some patients may prefer nebulizers because of convenience or other factors.
REFERENCES
1. Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2013;9: CD000052.
2. Barrett ML, Wier LM, Washington R. Trends in pediatric and adult hospital stays for asthma, 2000-2010. HCUP Statistical Brief #169. www.hcup-us.ahrq.gov/reports/stat briefs/sb169-Asthma-Trends-Hospital-Stays.pdf. Accessed June 16, 2014.
3. Pfuntner A, Wier LM, Stocks C. Most frequent conditions in US hospitals, 2011. HCUP Statistical Brief #162. www.hcup-us.ahrq.gov/reports/statbriefs/sb162.pdf. Accessed June 16, 2014.
4. Cates CJ, Crilly JA, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2006;(2): CD000052.
5. Turner MO, Patel A, Ginsburg S, et al. Bronchodilator delivery in acute airflow obstruction: a meta-analysis. Arch Intern Med. 1997;157:1736-1744.
6. National Heart, Lung, and Blood Institute Expert Panel Report 3 (EPR3): Guidelines for the diagnosis and management of asthma. www.nhlbi.nih.gov/guidelines/asthma/asth gdln.htm. Accessed June 16, 2014.
7. British Thoracic Society. British guideline of the management of asthma: a national clinical guideline. www.brit-thoracic.org.uk/document-library/clinical-information/asth ma/btssign-guideline-on-the-management-of-asthma/. Accessed June 16, 2014.
8. Pitts SR, Niska RW, Xu J, et al. National Hospital Ambulatory Medical Care Survey: 2006 emergency department summary. www.cdc.gov/nchs/data/nhsr/nhsr007.pdf. Accessed June 16, 2014.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Copyright © 2014. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice. 2014;63(6):321-322, 346.
PRACTICE CHANGER
Stop ordering nebulizers to deliver β-agonists to patients older than 2 who have mild or moderate asthma exacerbations. A metered-dose inhaler (MDI) with a spacer produces the same benefits with fewer adverse effects.1
STRENGTH OF RECOMMENDATION
A: Based on an updated Cochrane meta-analysis of 39 randomized controlled trials (RCTs). 1
ILLUSTRATIVE CASE
A 6-year-old girl with a history of reactive airway disease comes to your office complaining of cough and wheezing. On exam, she has mild retractions, a respiratory rate of 35 breaths/min, and an O2 saturation of 96% on room air. Her lung fields are diffusely wheezy. Her parents would like to keep her out of the hospital. How should you order her albuterol to decrease her wheezing and minimize adverse effects?
Asthma affects nearly 19 million adults and 7 million children in the United States.2 Asthma exacerbations are the third most common reason for hospitalization in children.2,3 Treatment usually requires multiple agents, including inhaled β-agonists. These are most effective when delivered to the peripheral airways, which is a challenge during an asthma exacerbation because of airway swelling and rapid breathing. Two devices have been developed to effectively deliver medication to the peripheral airways: nebulizers and MDIs with a holding chamber (spacer).1
Several studies have demonstrated that for mild to moderate asthma exacerbations, administering a β-agonist via an MDI with a spacer is as effective as using a nebulizer.4,5 Asthma treatment guidelines also state that spacers are either comparable or preferable to nebulizers for β-agonist administration in children and adults.6,7 However, based on our experience, clinicians still frequently order nebulizer treatments for patients with asthma exacerbations, despite several advantages of MDIs with spacers. Notably, they cost less and don’t require maintenance or a power source. Clinicians administered nebulizer therapy at more than 3.6 million emergency department (ED) visits in 2006.8
In this latest Cochrane review, Cates et al1 added four new studies to those included in their earlier Cochrane meta-analysis and evaluated what, if any, effect these studies had on our understanding of nebulizers versus MDIs with spacers.
STUDY SUMMARY
Outcomes with nebulizers are no better than those with spacers
This systematic review and meta-analysis pooled the results of RCTs comparing spacers to nebulizers for administering β-agonists during acute, non–life-threatening asthma exacerbations.1 The authors reviewed studies conducted in EDs, hospitals, and outpatient settings that included children and adults. The primary outcomes were hospital admission rates and duration of hospital stay. Secondary outcomes included time spent in the ED, change in pulse rate, and incidence of tremor.
Cates et al1 analyzed 39 trials that included 1,897 children and 729 adults and were conducted primarily in an ED or outpatient setting. The four new studies added 295 children and 58 adults to the researchers’ earlier meta-analysis. Studies involving adults and children were pooled separately. Most patients received multiple treatments with β-agonists titrated to the individual’s response.
No differences in hospitalizations. Rates of hospital admissions did not differ between patients receiving β-agonists via a spacer compared to a nebulizer in both adults (relative risk [RR] = 0.94) and children (RR = 0.71). Duration of hospital stay did not differ between the two delivery methods in adults (mean difference [MD] = –0.60 d) and children (MD = 0.33 d).
For kids, spacers meant less time in the ED. Duration in the ED was approximately half an hour shorter for children using spacers (MD = –33.48 min). There was no difference observed in adults (MD = 1.75 min). The rate of tremor was lower in children using spacers (RR = 0.64) and was similar in adults (RR = 1.12). The rise in pulse rate was lower in children using spacers
(MD = –5.41% change from baseline) and was similar in adults (MD = –1.23%).
On the next page: What's new and challenges to implementation >>
WHAT’S NEW
Additional evidence that spacers are as effective as nebulizers
This meta-analysis, which included four new studies, should finally dispel the myth that nebulizers deliver β-agonists more effectively than MDIs with spacers. Additionally, in children, spacers are associated with lower rates of adverse effects, including tremor and elevated pulse rate.
CAVEATS
Most studies involving children were open label
Although most of the adult trials in this meta-analysis involved a double-dummy design, which allows for effective participant blinding, most of the studies involving children were open label. This open-label design might have been a source of reporting bias for symptom-related outcomes but should not have affected hospital admission rates or duration of hospital stay.
In the double-dummy studies, adults received both a nebulizer and a spacer, which likely explains the similar time spent in the ED by the treatment and control groups.
CHALLENGES TO IMPLEMENTATION
Old habits are hard to break
Clinicians may think that patients view nebulizers as more potent or more effective than spacers and thus be more likely to order them. Some patients may prefer nebulizers because of convenience or other factors.
REFERENCES
1. Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2013;9: CD000052.
2. Barrett ML, Wier LM, Washington R. Trends in pediatric and adult hospital stays for asthma, 2000-2010. HCUP Statistical Brief #169. www.hcup-us.ahrq.gov/reports/stat briefs/sb169-Asthma-Trends-Hospital-Stays.pdf. Accessed June 16, 2014.
3. Pfuntner A, Wier LM, Stocks C. Most frequent conditions in US hospitals, 2011. HCUP Statistical Brief #162. www.hcup-us.ahrq.gov/reports/statbriefs/sb162.pdf. Accessed June 16, 2014.
4. Cates CJ, Crilly JA, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2006;(2): CD000052.
5. Turner MO, Patel A, Ginsburg S, et al. Bronchodilator delivery in acute airflow obstruction: a meta-analysis. Arch Intern Med. 1997;157:1736-1744.
6. National Heart, Lung, and Blood Institute Expert Panel Report 3 (EPR3): Guidelines for the diagnosis and management of asthma. www.nhlbi.nih.gov/guidelines/asthma/asth gdln.htm. Accessed June 16, 2014.
7. British Thoracic Society. British guideline of the management of asthma: a national clinical guideline. www.brit-thoracic.org.uk/document-library/clinical-information/asth ma/btssign-guideline-on-the-management-of-asthma/. Accessed June 16, 2014.
8. Pitts SR, Niska RW, Xu J, et al. National Hospital Ambulatory Medical Care Survey: 2006 emergency department summary. www.cdc.gov/nchs/data/nhsr/nhsr007.pdf. Accessed June 16, 2014.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Copyright © 2014. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice. 2014;63(6):321-322, 346.
Tips for Vaccinating Multiple Sclerosis Patients
DALLAS – Vaccinations are beneficial for individuals with multiple sclerosis because they help avoid infections, which in turn lower the risk of relapse.
But while inactive vaccines are considered safe for MS patients, the use of live vaccines such as Varivax, Zostavax, or yellow fever vaccine is more nuanced, especially as more disease-modifying therapies (DMT) become available. There are limited data available about the effect of live vaccines on patients who are on DMTs, and the final decision comes down to the risk-benefit ratio, said, Dr. Patricia K. Coyle, professor of neurology and director of the MS Comprehensive Care Center at Stony Brook (N.Y.) University Medical Center.
In a video interview at a meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis, Dr. Coyle summarizes what’s known so far about vaccines and MS, and provides tips to clinicians.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
DALLAS – Vaccinations are beneficial for individuals with multiple sclerosis because they help avoid infections, which in turn lower the risk of relapse.
But while inactive vaccines are considered safe for MS patients, the use of live vaccines such as Varivax, Zostavax, or yellow fever vaccine is more nuanced, especially as more disease-modifying therapies (DMT) become available. There are limited data available about the effect of live vaccines on patients who are on DMTs, and the final decision comes down to the risk-benefit ratio, said, Dr. Patricia K. Coyle, professor of neurology and director of the MS Comprehensive Care Center at Stony Brook (N.Y.) University Medical Center.
In a video interview at a meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis, Dr. Coyle summarizes what’s known so far about vaccines and MS, and provides tips to clinicians.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
DALLAS – Vaccinations are beneficial for individuals with multiple sclerosis because they help avoid infections, which in turn lower the risk of relapse.
But while inactive vaccines are considered safe for MS patients, the use of live vaccines such as Varivax, Zostavax, or yellow fever vaccine is more nuanced, especially as more disease-modifying therapies (DMT) become available. There are limited data available about the effect of live vaccines on patients who are on DMTs, and the final decision comes down to the risk-benefit ratio, said, Dr. Patricia K. Coyle, professor of neurology and director of the MS Comprehensive Care Center at Stony Brook (N.Y.) University Medical Center.
In a video interview at a meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis, Dr. Coyle summarizes what’s known so far about vaccines and MS, and provides tips to clinicians.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
AT THE CMSC/ACTRIMS ANNUAL MEETING
No Relief From Persistent Itchy Rash
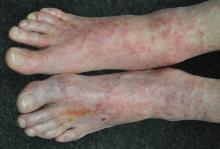
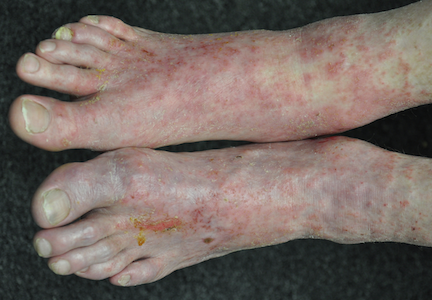
It’s somewhat unusual to see a patient with an eight-year history of the same problem, but this is what happens when a 51-year-old man presents to dermatology. Specifically, the problem is a very itchy foot rash, for which the patient has tried many OTC products without success. He has also used halobetasol cream, prescribed by a dermatologist he saw several years ago.
The patient denies having a rash anywhere else. He does, however, have a markedly atopic history, significant for seasonal allergies, asthma, and very sensitive skin.
For the past several years, when the rash has been particularly unbearable, he admits to pouring rubbing alcohol on his feet. This burned terribly, but his feet felt better afterward.
Asked what else happened eight years ago, the patient recalls starting his current job as a lineman for a power company—an occupation that requires him to wear steel-toed leather boots for hours at a time.
EXAMINATION
A dense, red, papulovesicular rash covers both feet in a stocking pattern. The rash stops abruptly at the same place on both lower legs, completely sparing the soles and interdigital skin. Focal areas of scaling and broken skin are seen on the tops and sides of both feet.
Continue for Joe Monroe's diagnosis and discussion >>
DISCUSSION
In the ’90s, a commercial for sneakers utilized the catchphrase, "It's the shoes!" And so it is occasionally with rashes on the feet. In this case, the patient was allergic to the leather on the inside of his work boots.
A true allergy such as this would be expected to itch and to manifest as a papulovesicular rash limited to areas touched by the leather on the upper portions of his shoes. And it would persist, despite the quantity of topical medications tried, because the patient wore the shoes for eight to 12 hours a day, five or six days per week, continually re-exposing his skin to the offending material. He had never taken any significant time off work and therefore hadn’t refrained from wearing the shoes long enough to allow the rash to abate.
Often, frustrated and uncomfortable patients take matters into their own hands, which can ultimately compound the problem. This patient didn't do his skin any favors with the rubbing alcohol and other products he'd tried (the most common offender being triple-antibiotic ointment, although this patient used it only on rare occasions). Another strategy these patients often employ is to soak their feet in watered-down bleach. Fortunately, this patient had considered this option but thought better of it. A more benign, but just as ineffective, attempt at self-treatment had been to change laundry detergents, which of course did nothing to resolve the rash. (His first clue should have been that laundry detergent would not affect the tops of his feet while sparing the rest of his body.)
The insides of leather shoes are usually tanned with potassium dichromate, a chemical known to provoke this kind of reaction. Even after the source of the rash was identified, however, getting some distance between the patient and his shoes wasn’t easy. I had to write him a note for work, requesting that he be allowed to refrain from wearing his boots for about two weeks.
During that time, he started a week-long course of cephalexin 500 mg tid and applied clobetasol foam twice a day. In dermatology, we assume that any longstanding wet rash on the feet will become secondarily infected or at least colonized with gram-positive bacteria. Of course, giving him the steroid foam meant we were utilizing a class 1 corticosteroid in a very drying vehicle.
Within a week, he was a new man, with almost totally clear foot skin. This still left him with the problem of the work boots and the job—but one problem at a time.
TAKE-HOME LEARNING POINTS
• The areas spared by a rash are often just as important as those that are affected.
• Laundry detergents, often blamed for rashes, are seldom the culprit.
• Fungal infections rarely affect the dorsum of the foot while sparing interdigital and plantar surfaces.
• The patient’s atopic state will likely render him/her more susceptible to allergens.
• The vehicle (cream, gel, ointment, solution, foam) and strength of topical steroids both matter.
• Patients can become sensitized to the preservatives or other chemicals in OTC or prescription corticosteroid creams.
It’s somewhat unusual to see a patient with an eight-year history of the same problem, but this is what happens when a 51-year-old man presents to dermatology. Specifically, the problem is a very itchy foot rash, for which the patient has tried many OTC products without success. He has also used halobetasol cream, prescribed by a dermatologist he saw several years ago.
The patient denies having a rash anywhere else. He does, however, have a markedly atopic history, significant for seasonal allergies, asthma, and very sensitive skin.
For the past several years, when the rash has been particularly unbearable, he admits to pouring rubbing alcohol on his feet. This burned terribly, but his feet felt better afterward.
Asked what else happened eight years ago, the patient recalls starting his current job as a lineman for a power company—an occupation that requires him to wear steel-toed leather boots for hours at a time.
EXAMINATION
A dense, red, papulovesicular rash covers both feet in a stocking pattern. The rash stops abruptly at the same place on both lower legs, completely sparing the soles and interdigital skin. Focal areas of scaling and broken skin are seen on the tops and sides of both feet.
Continue for Joe Monroe's diagnosis and discussion >>
DISCUSSION
In the ’90s, a commercial for sneakers utilized the catchphrase, "It's the shoes!" And so it is occasionally with rashes on the feet. In this case, the patient was allergic to the leather on the inside of his work boots.
A true allergy such as this would be expected to itch and to manifest as a papulovesicular rash limited to areas touched by the leather on the upper portions of his shoes. And it would persist, despite the quantity of topical medications tried, because the patient wore the shoes for eight to 12 hours a day, five or six days per week, continually re-exposing his skin to the offending material. He had never taken any significant time off work and therefore hadn’t refrained from wearing the shoes long enough to allow the rash to abate.
Often, frustrated and uncomfortable patients take matters into their own hands, which can ultimately compound the problem. This patient didn't do his skin any favors with the rubbing alcohol and other products he'd tried (the most common offender being triple-antibiotic ointment, although this patient used it only on rare occasions). Another strategy these patients often employ is to soak their feet in watered-down bleach. Fortunately, this patient had considered this option but thought better of it. A more benign, but just as ineffective, attempt at self-treatment had been to change laundry detergents, which of course did nothing to resolve the rash. (His first clue should have been that laundry detergent would not affect the tops of his feet while sparing the rest of his body.)
The insides of leather shoes are usually tanned with potassium dichromate, a chemical known to provoke this kind of reaction. Even after the source of the rash was identified, however, getting some distance between the patient and his shoes wasn’t easy. I had to write him a note for work, requesting that he be allowed to refrain from wearing his boots for about two weeks.
During that time, he started a week-long course of cephalexin 500 mg tid and applied clobetasol foam twice a day. In dermatology, we assume that any longstanding wet rash on the feet will become secondarily infected or at least colonized with gram-positive bacteria. Of course, giving him the steroid foam meant we were utilizing a class 1 corticosteroid in a very drying vehicle.
Within a week, he was a new man, with almost totally clear foot skin. This still left him with the problem of the work boots and the job—but one problem at a time.
TAKE-HOME LEARNING POINTS
• The areas spared by a rash are often just as important as those that are affected.
• Laundry detergents, often blamed for rashes, are seldom the culprit.
• Fungal infections rarely affect the dorsum of the foot while sparing interdigital and plantar surfaces.
• The patient’s atopic state will likely render him/her more susceptible to allergens.
• The vehicle (cream, gel, ointment, solution, foam) and strength of topical steroids both matter.
• Patients can become sensitized to the preservatives or other chemicals in OTC or prescription corticosteroid creams.
It’s somewhat unusual to see a patient with an eight-year history of the same problem, but this is what happens when a 51-year-old man presents to dermatology. Specifically, the problem is a very itchy foot rash, for which the patient has tried many OTC products without success. He has also used halobetasol cream, prescribed by a dermatologist he saw several years ago.
The patient denies having a rash anywhere else. He does, however, have a markedly atopic history, significant for seasonal allergies, asthma, and very sensitive skin.
For the past several years, when the rash has been particularly unbearable, he admits to pouring rubbing alcohol on his feet. This burned terribly, but his feet felt better afterward.
Asked what else happened eight years ago, the patient recalls starting his current job as a lineman for a power company—an occupation that requires him to wear steel-toed leather boots for hours at a time.
EXAMINATION
A dense, red, papulovesicular rash covers both feet in a stocking pattern. The rash stops abruptly at the same place on both lower legs, completely sparing the soles and interdigital skin. Focal areas of scaling and broken skin are seen on the tops and sides of both feet.
Continue for Joe Monroe's diagnosis and discussion >>
DISCUSSION
In the ’90s, a commercial for sneakers utilized the catchphrase, "It's the shoes!" And so it is occasionally with rashes on the feet. In this case, the patient was allergic to the leather on the inside of his work boots.
A true allergy such as this would be expected to itch and to manifest as a papulovesicular rash limited to areas touched by the leather on the upper portions of his shoes. And it would persist, despite the quantity of topical medications tried, because the patient wore the shoes for eight to 12 hours a day, five or six days per week, continually re-exposing his skin to the offending material. He had never taken any significant time off work and therefore hadn’t refrained from wearing the shoes long enough to allow the rash to abate.
Often, frustrated and uncomfortable patients take matters into their own hands, which can ultimately compound the problem. This patient didn't do his skin any favors with the rubbing alcohol and other products he'd tried (the most common offender being triple-antibiotic ointment, although this patient used it only on rare occasions). Another strategy these patients often employ is to soak their feet in watered-down bleach. Fortunately, this patient had considered this option but thought better of it. A more benign, but just as ineffective, attempt at self-treatment had been to change laundry detergents, which of course did nothing to resolve the rash. (His first clue should have been that laundry detergent would not affect the tops of his feet while sparing the rest of his body.)
The insides of leather shoes are usually tanned with potassium dichromate, a chemical known to provoke this kind of reaction. Even after the source of the rash was identified, however, getting some distance between the patient and his shoes wasn’t easy. I had to write him a note for work, requesting that he be allowed to refrain from wearing his boots for about two weeks.
During that time, he started a week-long course of cephalexin 500 mg tid and applied clobetasol foam twice a day. In dermatology, we assume that any longstanding wet rash on the feet will become secondarily infected or at least colonized with gram-positive bacteria. Of course, giving him the steroid foam meant we were utilizing a class 1 corticosteroid in a very drying vehicle.
Within a week, he was a new man, with almost totally clear foot skin. This still left him with the problem of the work boots and the job—but one problem at a time.
TAKE-HOME LEARNING POINTS
• The areas spared by a rash are often just as important as those that are affected.
• Laundry detergents, often blamed for rashes, are seldom the culprit.
• Fungal infections rarely affect the dorsum of the foot while sparing interdigital and plantar surfaces.
• The patient’s atopic state will likely render him/her more susceptible to allergens.
• The vehicle (cream, gel, ointment, solution, foam) and strength of topical steroids both matter.
• Patients can become sensitized to the preservatives or other chemicals in OTC or prescription corticosteroid creams.
In chronic sinusitis, Pneumovax screen may fuel IVIG overuse
SAN DIEGO – Using chronic sinusitis patients’ response to Pneumovax to screen them for specific antibody deficiency may lead to overtreatment with intravenous immunoglobulin, a study showed.
In addition to its traditional use to prevent pneumonia and other infections, Pneumovax (pneumococcal vaccine polyvalent) has found a new role recently: as a screening tool in chronic rhinosinusitis (CRS) patients for specific antibody deficiency (SAD), a type of immune deficiency. If patients have a poor antibody response to the Pneumovax shot, they are thought to have SAD and are placed on intravenous immunoglobulin (IVIG) – sometimes without much regard for their clinical history. The practice has been fueled by the growing suspicion that chronic rhinosinusitis might be driven by a faulty immune system.
However, the severity of SAD does not correlate neatly with the response to Pneumovax, Northwestern University investigators found in a study of 595 adult CRS patients. So, treating patients with IVIG based primarily on how they respond to the vaccine might be overkill. To prevent overuse of IVIG, it’s probably best to limit Pneumovax screening to patients with clinically worse disease, according to lead investigator Dr. Anjeni Keswani, formerly of Northwestern University in Chicago but now an allergist/immunologist at Duke University in Durham, N.C.
Overall, the proper role of Pneumovax screening still needs to be worked out. At least for now, SAD "just can’t be a laboratory diagnosis. It should include clinical history before you make a decision on IVIG therapy," Dr. Keswani said.
In the Lund-MacKay staging system for CRS, higher scores mean worse disease. The 95 patients in the study who had mild SAD (defined as 7-9 protective Streptococcus pneumoniae antibody titers after vaccination, out of 14 serotypes measured) and the 120 patients with moderate SAD (3-6 protective titers after vaccination) had significantly higher Lund-MacKay scores than the 24 patients with severe SAD. Severe SAD was defined as 2 or fewer protective titers following the vaccine.
Mild and moderate SAD patients also had significantly higher rates of asthma and higher, although not significantly higher, rates of allergic rhinitis and nasal polyps, Dr. Keswani said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Meanwhile, patients with more severe SAD were significantly more likely than mild SAD patients to have pneumonia histories and, when present, more severe asthma. And they used significantly more antibiotics in the 2 years following vaccination.
Asthma severity, antibiotic use, and pneumonia history in mild SAD patients were, in fact, similar to rates in CRS patients without immune deficiency. That raises the possibility that what’s now considered mild SAD might not even be an immunodeficiency, hence the notion of limiting Pneumovax screening to patients who are worse off.
As it all gets sorted out, "we don’t want patients diagnosed with SAD to automatically be placed on immunoglobulin replacement," Dr. Keswani said. "I don’t think that’s useful for the majority of patients. There is a proportion who mount a good response to infection; they may have an impaired response to the vaccine, but we can manage them with antibiotic prophylaxis and early assessment.
"We are trying to come up with guidelines to better help determine who should be screened," she added. "If they don’t have evidence of infection, I don’t think we can actually call them SAD."
Dr. Keswani said she had no relevant disclosures, and there was no outside funding involved in the project.
SAN DIEGO – Using chronic sinusitis patients’ response to Pneumovax to screen them for specific antibody deficiency may lead to overtreatment with intravenous immunoglobulin, a study showed.
In addition to its traditional use to prevent pneumonia and other infections, Pneumovax (pneumococcal vaccine polyvalent) has found a new role recently: as a screening tool in chronic rhinosinusitis (CRS) patients for specific antibody deficiency (SAD), a type of immune deficiency. If patients have a poor antibody response to the Pneumovax shot, they are thought to have SAD and are placed on intravenous immunoglobulin (IVIG) – sometimes without much regard for their clinical history. The practice has been fueled by the growing suspicion that chronic rhinosinusitis might be driven by a faulty immune system.
However, the severity of SAD does not correlate neatly with the response to Pneumovax, Northwestern University investigators found in a study of 595 adult CRS patients. So, treating patients with IVIG based primarily on how they respond to the vaccine might be overkill. To prevent overuse of IVIG, it’s probably best to limit Pneumovax screening to patients with clinically worse disease, according to lead investigator Dr. Anjeni Keswani, formerly of Northwestern University in Chicago but now an allergist/immunologist at Duke University in Durham, N.C.
Overall, the proper role of Pneumovax screening still needs to be worked out. At least for now, SAD "just can’t be a laboratory diagnosis. It should include clinical history before you make a decision on IVIG therapy," Dr. Keswani said.
In the Lund-MacKay staging system for CRS, higher scores mean worse disease. The 95 patients in the study who had mild SAD (defined as 7-9 protective Streptococcus pneumoniae antibody titers after vaccination, out of 14 serotypes measured) and the 120 patients with moderate SAD (3-6 protective titers after vaccination) had significantly higher Lund-MacKay scores than the 24 patients with severe SAD. Severe SAD was defined as 2 or fewer protective titers following the vaccine.
Mild and moderate SAD patients also had significantly higher rates of asthma and higher, although not significantly higher, rates of allergic rhinitis and nasal polyps, Dr. Keswani said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Meanwhile, patients with more severe SAD were significantly more likely than mild SAD patients to have pneumonia histories and, when present, more severe asthma. And they used significantly more antibiotics in the 2 years following vaccination.
Asthma severity, antibiotic use, and pneumonia history in mild SAD patients were, in fact, similar to rates in CRS patients without immune deficiency. That raises the possibility that what’s now considered mild SAD might not even be an immunodeficiency, hence the notion of limiting Pneumovax screening to patients who are worse off.
As it all gets sorted out, "we don’t want patients diagnosed with SAD to automatically be placed on immunoglobulin replacement," Dr. Keswani said. "I don’t think that’s useful for the majority of patients. There is a proportion who mount a good response to infection; they may have an impaired response to the vaccine, but we can manage them with antibiotic prophylaxis and early assessment.
"We are trying to come up with guidelines to better help determine who should be screened," she added. "If they don’t have evidence of infection, I don’t think we can actually call them SAD."
Dr. Keswani said she had no relevant disclosures, and there was no outside funding involved in the project.
SAN DIEGO – Using chronic sinusitis patients’ response to Pneumovax to screen them for specific antibody deficiency may lead to overtreatment with intravenous immunoglobulin, a study showed.
In addition to its traditional use to prevent pneumonia and other infections, Pneumovax (pneumococcal vaccine polyvalent) has found a new role recently: as a screening tool in chronic rhinosinusitis (CRS) patients for specific antibody deficiency (SAD), a type of immune deficiency. If patients have a poor antibody response to the Pneumovax shot, they are thought to have SAD and are placed on intravenous immunoglobulin (IVIG) – sometimes without much regard for their clinical history. The practice has been fueled by the growing suspicion that chronic rhinosinusitis might be driven by a faulty immune system.
However, the severity of SAD does not correlate neatly with the response to Pneumovax, Northwestern University investigators found in a study of 595 adult CRS patients. So, treating patients with IVIG based primarily on how they respond to the vaccine might be overkill. To prevent overuse of IVIG, it’s probably best to limit Pneumovax screening to patients with clinically worse disease, according to lead investigator Dr. Anjeni Keswani, formerly of Northwestern University in Chicago but now an allergist/immunologist at Duke University in Durham, N.C.
Overall, the proper role of Pneumovax screening still needs to be worked out. At least for now, SAD "just can’t be a laboratory diagnosis. It should include clinical history before you make a decision on IVIG therapy," Dr. Keswani said.
In the Lund-MacKay staging system for CRS, higher scores mean worse disease. The 95 patients in the study who had mild SAD (defined as 7-9 protective Streptococcus pneumoniae antibody titers after vaccination, out of 14 serotypes measured) and the 120 patients with moderate SAD (3-6 protective titers after vaccination) had significantly higher Lund-MacKay scores than the 24 patients with severe SAD. Severe SAD was defined as 2 or fewer protective titers following the vaccine.
Mild and moderate SAD patients also had significantly higher rates of asthma and higher, although not significantly higher, rates of allergic rhinitis and nasal polyps, Dr. Keswani said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Meanwhile, patients with more severe SAD were significantly more likely than mild SAD patients to have pneumonia histories and, when present, more severe asthma. And they used significantly more antibiotics in the 2 years following vaccination.
Asthma severity, antibiotic use, and pneumonia history in mild SAD patients were, in fact, similar to rates in CRS patients without immune deficiency. That raises the possibility that what’s now considered mild SAD might not even be an immunodeficiency, hence the notion of limiting Pneumovax screening to patients who are worse off.
As it all gets sorted out, "we don’t want patients diagnosed with SAD to automatically be placed on immunoglobulin replacement," Dr. Keswani said. "I don’t think that’s useful for the majority of patients. There is a proportion who mount a good response to infection; they may have an impaired response to the vaccine, but we can manage them with antibiotic prophylaxis and early assessment.
"We are trying to come up with guidelines to better help determine who should be screened," she added. "If they don’t have evidence of infection, I don’t think we can actually call them SAD."
Dr. Keswani said she had no relevant disclosures, and there was no outside funding involved in the project.
AT THE 2014 AAAAI ANNUAL MEETING
Major finding: In patients with chronic rhinosinusitis, the severity of specific antibody deficiency did not correlate neatly with patients’ response to Pneumovax vaccination.
Data Source: Retrospective, electronic medical record review of 595 patients.
Disclosures: Dr. Anjeni Keswani said she had no disclosures, and the study received no outside funding.
Peanut Reactivity After Immunotherapy
SAN DIEGO – Waiting 3 months after successful oral immunotherapy for peanut allergy before exposure to peanuts significantly increased the odds of reactivity returning in a prospective study of 20 children.
All 20 patients became desensitized to peanuts while on daily oral immunotherapy and successfully passed double-blind, placebo-controlled food challenges at the end of immunotherapy. All 16 patients who then avoided peanuts for 1 month passed a second food challenge, but only 1 of 4 patients who avoided peanuts for 3 months after immunotherapy passed a second one.
"If you wait long enough, the desensitization effect may well wear off," Dr. Brian P. Vickery said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
He and his associates looked at levels of basophil activation and conducted skin prick tests to peanut to get a better sense of why this was happening. Basophil activation to peanut antigen and anti-IgE stimulation increased significantly between the times of the first and second food challenges in the patients who avoided peanuts for 3 months but not in those who avoided the nuts for only 1 month.
The basophil activation levels had been similar between groups at the end of immunotherapy, suggesting that desensitization succeeded in all patients, but the length of peanut avoidance affected basophil responses, leading to revival of clinical reactivity.
Skin prick test results returned to baseline levels in three of the four patients who avoided peanuts for 3 months after the end of immunotherapy.
The amount of time on oral immunotherapy did not seem to be a key factor in the likelihood of sustained suppression of allergic disease, reported Dr. Vickery of the department of pediatrics at the University of North Carolina at Chapel Hill. Patients who waited 1 month before exposure to peanut had been on approximately 20-50 months of oral immunotherapy, and patients who waited 3 months before exposure had been on approximately 60 months of immunotherapy.
The lead author of the study was Michael D. Kulis Jr., Ph.D., also of the university.
Prolonged avoidance of peanut after peanut oral immunotherapy appears to be detrimental and may reverse the effects of the treatment, Dr. A. Wesley Burks said at a press briefing.
The findings may be applicable to other food allergies, but "we don’t know that because there have not been enough studies that long with other foods," said Dr. Burks, a coinvestigator in the study and chairman of the department of pediatrics at the university. "I wouldn’t anticipate that it would be different."
The average age of children in the study was 6 years.
This exploratory study was not controlled and was limited by its small size. The Peanut Oral Immunotherapy in Children (IMPACT) study is underway and should answer the question of how long the clinical effects of oral immunotherapy last, Dr. Vickery said. The study is being sponsored by the Immune Tolerance Network and the National Institute of Allergy and Infectious Diseases.
Dr. Vickery, Dr. Kulis, and Dr. Burks reported having no financial disclosures. The National Institutes of Health funded the study.
On Twitter @sherryboschert
SAN DIEGO – Waiting 3 months after successful oral immunotherapy for peanut allergy before exposure to peanuts significantly increased the odds of reactivity returning in a prospective study of 20 children.
All 20 patients became desensitized to peanuts while on daily oral immunotherapy and successfully passed double-blind, placebo-controlled food challenges at the end of immunotherapy. All 16 patients who then avoided peanuts for 1 month passed a second food challenge, but only 1 of 4 patients who avoided peanuts for 3 months after immunotherapy passed a second one.
"If you wait long enough, the desensitization effect may well wear off," Dr. Brian P. Vickery said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
He and his associates looked at levels of basophil activation and conducted skin prick tests to peanut to get a better sense of why this was happening. Basophil activation to peanut antigen and anti-IgE stimulation increased significantly between the times of the first and second food challenges in the patients who avoided peanuts for 3 months but not in those who avoided the nuts for only 1 month.
The basophil activation levels had been similar between groups at the end of immunotherapy, suggesting that desensitization succeeded in all patients, but the length of peanut avoidance affected basophil responses, leading to revival of clinical reactivity.
Skin prick test results returned to baseline levels in three of the four patients who avoided peanuts for 3 months after the end of immunotherapy.
The amount of time on oral immunotherapy did not seem to be a key factor in the likelihood of sustained suppression of allergic disease, reported Dr. Vickery of the department of pediatrics at the University of North Carolina at Chapel Hill. Patients who waited 1 month before exposure to peanut had been on approximately 20-50 months of oral immunotherapy, and patients who waited 3 months before exposure had been on approximately 60 months of immunotherapy.
The lead author of the study was Michael D. Kulis Jr., Ph.D., also of the university.
Prolonged avoidance of peanut after peanut oral immunotherapy appears to be detrimental and may reverse the effects of the treatment, Dr. A. Wesley Burks said at a press briefing.
The findings may be applicable to other food allergies, but "we don’t know that because there have not been enough studies that long with other foods," said Dr. Burks, a coinvestigator in the study and chairman of the department of pediatrics at the university. "I wouldn’t anticipate that it would be different."
The average age of children in the study was 6 years.
This exploratory study was not controlled and was limited by its small size. The Peanut Oral Immunotherapy in Children (IMPACT) study is underway and should answer the question of how long the clinical effects of oral immunotherapy last, Dr. Vickery said. The study is being sponsored by the Immune Tolerance Network and the National Institute of Allergy and Infectious Diseases.
Dr. Vickery, Dr. Kulis, and Dr. Burks reported having no financial disclosures. The National Institutes of Health funded the study.
On Twitter @sherryboschert
SAN DIEGO – Waiting 3 months after successful oral immunotherapy for peanut allergy before exposure to peanuts significantly increased the odds of reactivity returning in a prospective study of 20 children.
All 20 patients became desensitized to peanuts while on daily oral immunotherapy and successfully passed double-blind, placebo-controlled food challenges at the end of immunotherapy. All 16 patients who then avoided peanuts for 1 month passed a second food challenge, but only 1 of 4 patients who avoided peanuts for 3 months after immunotherapy passed a second one.
"If you wait long enough, the desensitization effect may well wear off," Dr. Brian P. Vickery said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
He and his associates looked at levels of basophil activation and conducted skin prick tests to peanut to get a better sense of why this was happening. Basophil activation to peanut antigen and anti-IgE stimulation increased significantly between the times of the first and second food challenges in the patients who avoided peanuts for 3 months but not in those who avoided the nuts for only 1 month.
The basophil activation levels had been similar between groups at the end of immunotherapy, suggesting that desensitization succeeded in all patients, but the length of peanut avoidance affected basophil responses, leading to revival of clinical reactivity.
Skin prick test results returned to baseline levels in three of the four patients who avoided peanuts for 3 months after the end of immunotherapy.
The amount of time on oral immunotherapy did not seem to be a key factor in the likelihood of sustained suppression of allergic disease, reported Dr. Vickery of the department of pediatrics at the University of North Carolina at Chapel Hill. Patients who waited 1 month before exposure to peanut had been on approximately 20-50 months of oral immunotherapy, and patients who waited 3 months before exposure had been on approximately 60 months of immunotherapy.
The lead author of the study was Michael D. Kulis Jr., Ph.D., also of the university.
Prolonged avoidance of peanut after peanut oral immunotherapy appears to be detrimental and may reverse the effects of the treatment, Dr. A. Wesley Burks said at a press briefing.
The findings may be applicable to other food allergies, but "we don’t know that because there have not been enough studies that long with other foods," said Dr. Burks, a coinvestigator in the study and chairman of the department of pediatrics at the university. "I wouldn’t anticipate that it would be different."
The average age of children in the study was 6 years.
This exploratory study was not controlled and was limited by its small size. The Peanut Oral Immunotherapy in Children (IMPACT) study is underway and should answer the question of how long the clinical effects of oral immunotherapy last, Dr. Vickery said. The study is being sponsored by the Immune Tolerance Network and the National Institute of Allergy and Infectious Diseases.
Dr. Vickery, Dr. Kulis, and Dr. Burks reported having no financial disclosures. The National Institutes of Health funded the study.
On Twitter @sherryboschert
AT 2014 AAAAI ANNUAL MEETING