User login
Geriatric-Centered Interdisciplinary Care Pathway Reduces Delirium in Hospitalized Older Adults With Traumatic Injury
Study 1 Overview (Park et al)
Objective: To examine whether implementation of a geriatric trauma clinical pathway is associated with reduced rates of delirium in older adults with traumatic injury.
Design: Retrospective case-control study of electronic health records.
Setting and participants: Eligible patients were persons aged 65 years or older who were admitted to the trauma service and did not undergo an operation. A Geriatric Trauma Care Pathway was developed by a multidisciplinary Stanford Quality Pathways team and formally launched on November 1, 2018. The clinical pathway was designed to incorporate geriatric best practices, which included order sets (eg, age-appropriate nonpharmacological interventions and pharmacological dosages), guidelines (eg, Institute for Healthcare Improvement Age-Friendly Health systems 4M framework), automated consultations (comprehensive geriatric assessment), and escalation pathways executed by a multidisciplinary team (eg, pain, bowel, and sleep regulation). The clinical pathway began with admission to the emergency department (ED) (ie, automatic trigger of geriatric trauma care admission order set), daily multidisciplinary team meetings during acute hospitalization, and a transitional care team consultation for postdischarge follow-up or home visit.
Main outcome measures: The primary outcome was delirium as determined by a positive Confusion Assessment Method (CAM) score or a diagnosis of delirium by the clinical team. The secondary outcome was hospital length of stay (LOS). Process measures for pathway compliance (eg, achieving adequate pain control, early mobilization, advance care planning) were assessed. Outcome measures were compared between patients who underwent the Geriatric Trauma Care Pathway intervention (postimplementation group) vs patients who were treated prior to pathway implementation (baseline pre-implementation group).
Main results: Of the 859 eligible patients, 712 were included in the analysis (442 [62.1%] in the baseline pre-implementation group and 270 [37.9%] in the postimplementation group); mean (SD) age was 81.4 (9.1) years, and 394 (55.3%) were women. The injury mechanism was similar between groups, with falls being the most common cause of injury (247 [55.9%] in the baseline group vs 162 [60.0%] in the postimplementation group; P = .43). Injuries as measured by Injury Severity Score (ISS) were minor or moderate in both groups (261 [59.0%] in baseline group vs 168 [62.2%] in postimplementation group; P = .87). The adjusted odds ratio (OR) for delirium in the postimplementation group was lower compared to the baseline pre-implementation group (OR, 0.54; 95% CI, 0.37-0.80; P < .001). Measures of advance care planning in the postimplementation group improved, including more frequent goals-of-care documentation (53.7% in postimplementation group vs 16.7% in baseline group; P < .001) and a shortened time to first goals-of-care discussion upon presenting to the ED (36 hours in postimplementation group vs 50 hours in baseline group; P = .03).
Conclusion: Implementation of a multidisciplinary geriatric trauma clinical pathway for older adults with traumatic injury at a single level I trauma center was associated with reduced rates of delirium.
Study 2 Overview (Bryant et al)
Objective: To determine whether an interdisciplinary care pathway for frail trauma patients can improve in-hospital mortality, complications, and 30-day readmissions.
Design: Retrospective cohort study of frail patients.
Setting and participants: Eligible patients were persons aged 65 years or older who were admitted to the trauma service and survived more than 24 hours; admitted to and discharged from the trauma unit; and determined to be pre-frail or frail by a geriatrician’s assessment. A Frailty Identification and Care Pathway designed to reduce delirium and complications in frail older trauma patients was developed by a multidisciplinary team and implemented in 2016. The standardized evidence-based interdisciplinary care pathway included utilization of order sets and interventions for delirium prevention, early ambulation, bowel and pain regimens, nutrition and physical therapy consults, medication management, care-goal setting, and geriatric assessments.
Main outcome measures: The main outcomes were delirium as determined by a positive CAM score, major complications as defined by the Trauma Quality Improvement Project, in-hospital mortality, and 30-day hospital readmission. Outcome measures were compared between patients who underwent Frailty Identification and Care Pathway intervention (postintervention group) vs patients who were treated prior to pathway implementation (pre-intervention group).
Main results: A total of 269 frail patients were included in the analysis (125 in pre-intervention group vs 144 in postintervention group). Patient demographic and admission characteristics were similar between the 2 groups: mean age was 83.5 (7.1) years, 60.6% were women, and median ISS was 10 (interquartile range [IQR], 9-14). The injury mechanism was similar between groups, with falls accounting for 92.8% and 86.1% of injuries in the pre-intervention and postintervention groups, respectively (P = .07). In univariate analysis, the Frailty Identification and Care Pathway intervention was associated with a significant reduction in delirium (12.5% vs 21.6%, P = .04) and 30-day hospital readmission (2.7% vs 9.6%, P = .01) compared to patients in the pre-intervention group. However, rates of major complications (28.5% vs 28.0%, P = 0.93) and in-hospital mortality (4.2% vs 7.2%, P = .28) were similar between the pre-intervention and postintervention groups. In multivariate logistic regression models adjusted for patient characteristics (age, sex, race, ISS), patients in the postintervention group had lower delirium (OR, 0.44; 95% CI, 0.22-0.88; P = .02) and 30-day hospital readmission (OR, 0.25; 95% CI, 0.07-0.84; P = .02) rates compared to those in the pre-intervention group.
Conclusion: Implementation of an interdisciplinary care protocol for frail geriatric trauma patients significantly decreased their risks for in-hospital delirium and 30-day hospital readmission.
Commentary
Traumatic injuries in older adults are associated with higher morbidity and mortality compared to younger patients, with falls and motor vehicle accidents accounting for a majority of these injuries. Astoundingly, up to one-third of this vulnerable population presenting to hospitals with an ISS greater than 15 may die during hospitalization.1 As a result, a large number of studies and clinical trials have focused on interventions that are designed to reduce fall risks, and hence reduce adverse consequences of traumatic injuries that may arise after falls.2 However, this emphasis on falls prevention has overshadowed a need to develop effective geriatric-centered clinical interventions that aim to improve outcomes in older adults who present to hospitals with traumatic injuries. Furthermore, frailty—a geriatric syndrome indicative of an increased state of vulnerability and predictive of adverse outcomes such as delirium—is highly prevalent in older patients with traumatic injury.3 Thus, there is an urgent need to develop novel, hospital-based, traumatic injury–targeting strategies that incorporate a thoughtful redesign of the care framework that includes evidence-based interventions for geriatric syndromes such as delirium and frailty.
The study reported by Park et al (Study 1) represents the latest effort to evaluate inpatient management strategies designed to improve outcomes in hospitalized older adults who have sustained traumatic injury. Through the implementation of a novel multidisciplinary Geriatric Trauma Care Pathway that incorporates geriatric best practices, this intervention was found to be associated with a 46% lower risk of in-hospital delirium. Because of the inclusion of all age-eligible patients across all strata of traumatic injuries, rather than preselecting for those at the highest risk for poor clinical outcomes, the benefits of this intervention extend to those with minor or moderate injury severity. Furthermore, the improvement in delirium (ie, the primary outcome) is particularly meaningful given that delirium is one of the most common hospital-associated complications that increase hospital LOS, discharge to an institution, and mortality in older adults. Finally, the study’s observed reduced time to a first goals-of-care discussion and increased frequency of goals-of-care documentation after intervention should not be overlooked. The improvements in these 2 process measures are highly significant given that advanced care planning, an intervention that helps to align patients’ values, goals, and treatments, is completed at substantially lower rates in older adults in the acute hospital setting.4
Similarly, in an earlier published study, Bryant and colleagues (Study 2) also show that a geriatric-focused interdisciplinary trauma care pathway is associated with delirium risk reduction in hospitalized older trauma patients. Much like Study 1, the Frailty Identification and Care Pathway utilized in Study 2 is an evidence-based interdisciplinary care pathway that includes the use of geriatric assessments, order sets, and geriatric best practices. Moreover, its exclusive inclusion of pre-frail and frail older patients (ie, those at higher risk for poor outcomes) with moderate injury severity (median ISS of 10 [IQR, 9-14]) suggests that this type of care pathway benefits hospitalized older trauma patients, who are particularly vulnerable to adverse complications such as delirium. Moreover, the successful utilization of the FRAIL questionnaire, a validated frailty screening tool, by surgical residents in the ED to initiate this care pathway demonstrates the feasibility of its use in expediting frailty screening in older patients in trauma care.
Application for Clinical Practice and System Implementation
Findings from the 2 studies discussed in this review indicate that implementation of interdisciplinary clinical care pathways predicated on evidence-based geriatric principles and best practices is a promising approach to reduce delirium in hospitalized older trauma patients. These studies have helped to lay the groundwork in outlining the roadmaps (eg, processes and infrastructures) needed to create such clinical pathways. These key elements include: (1) integration of a multidisciplinary committee (eg, representation from trauma, emergency, and geriatric medicine, nursing, physical and occupational therapy, pharmacy, social work) in pathway design and implementation; (2) adaption of evidence-based geriatric best practices (eg, the Institute for Healthcare Improvement Age-Friendly Health System 4M framework [medication, mentation, mobility, what matters]) to prioritize interventions and to design a pathway that incorporates these features; (3) incorporation of comprehensive geriatric assessment by interdisciplinary providers; (4) utilization of validated clinical instruments to assess physical and cognitive functions, frailty, delirium, and social determinants of health; (5) modification of electronic health record systems to encompass order sets that incorporate evidence-based, nonpharmacological and pharmacological interventions to manage symptoms (eg, delirium, pain, bowel movement, sleep, immobility, polypharmacy) essential to quality geriatric care; and (6) integration of patient and caregiver preferences via goals-of-care discussions and corresponding documentation and communication of these goals.
Additionally, these 2 studies imparted some strategies that may facilitate the implementation of interdisciplinary clinical care pathways in trauma care. Examples of such facilitators include: (1) collaboration with champions within each specialty to reinforce education and buy-in; (2) creation of automatically triggered order sets upon patient presentation to the ED that unites distinct features of clinical pathways; (3) adaption and reorganization of existing hospital infrastructures and resources to meet the needs of clinical pathways implementation (eg, utilizing information technology resources to develop electronic health record order sets; using quality department to develop clinical pathway guidelines and electronic outcome dashboards); and (4) development of individualized patient and caregiver education materials based on care needs (eg, principles of delirium prevention and preservation of mobility during hospitalization) to prepare and engage these stakeholders in patient care and recovery.
Practice Points
- A geriatric interdisciplinary care model can be effectively applied to the management of acute trauma in older patients.
- Interdisciplinary clinical pathways should incorporate geriatric best practices and guidelines and age-appropriate order sets to prioritize and integrate care.
—Fred Ko, MD, MS
1. Hashmi A, Ibrahim-Zada I, Rhee P, et al. Predictors of mortality in geriatric trauma patients: a systematic review and meta-analysis. J Trauma Acute Care Surg. 2014;76(3):894-901. doi:10.1097/TA.0b013e3182ab0763
2. Hopewell S, Adedire O, Copsey BJ, et al. Multifactorial and multiple component interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2018;7(7):CD012221. doi:10.1002/14651858.CD012221.pub2
3. Joseph B, Pandit V, Zangbar B, et al. Superiority of frailty over age in predicting outcomes among geriatric trauma patients: a prospective analysis. JAMA Surg. 2014;149(8):766-772. doi:10.1001/jamasurg.2014.296
4. Hopkins SA, Bentley A, Phillips V, Barclay S. Advance care plans and hospitalized frail older adults: a systematic review. BMJ Support Palliat Care. 2020;10(2):164-174. doi:10.1136/bmjspcare-2019-002093
Study 1 Overview (Park et al)
Objective: To examine whether implementation of a geriatric trauma clinical pathway is associated with reduced rates of delirium in older adults with traumatic injury.
Design: Retrospective case-control study of electronic health records.
Setting and participants: Eligible patients were persons aged 65 years or older who were admitted to the trauma service and did not undergo an operation. A Geriatric Trauma Care Pathway was developed by a multidisciplinary Stanford Quality Pathways team and formally launched on November 1, 2018. The clinical pathway was designed to incorporate geriatric best practices, which included order sets (eg, age-appropriate nonpharmacological interventions and pharmacological dosages), guidelines (eg, Institute for Healthcare Improvement Age-Friendly Health systems 4M framework), automated consultations (comprehensive geriatric assessment), and escalation pathways executed by a multidisciplinary team (eg, pain, bowel, and sleep regulation). The clinical pathway began with admission to the emergency department (ED) (ie, automatic trigger of geriatric trauma care admission order set), daily multidisciplinary team meetings during acute hospitalization, and a transitional care team consultation for postdischarge follow-up or home visit.
Main outcome measures: The primary outcome was delirium as determined by a positive Confusion Assessment Method (CAM) score or a diagnosis of delirium by the clinical team. The secondary outcome was hospital length of stay (LOS). Process measures for pathway compliance (eg, achieving adequate pain control, early mobilization, advance care planning) were assessed. Outcome measures were compared between patients who underwent the Geriatric Trauma Care Pathway intervention (postimplementation group) vs patients who were treated prior to pathway implementation (baseline pre-implementation group).
Main results: Of the 859 eligible patients, 712 were included in the analysis (442 [62.1%] in the baseline pre-implementation group and 270 [37.9%] in the postimplementation group); mean (SD) age was 81.4 (9.1) years, and 394 (55.3%) were women. The injury mechanism was similar between groups, with falls being the most common cause of injury (247 [55.9%] in the baseline group vs 162 [60.0%] in the postimplementation group; P = .43). Injuries as measured by Injury Severity Score (ISS) were minor or moderate in both groups (261 [59.0%] in baseline group vs 168 [62.2%] in postimplementation group; P = .87). The adjusted odds ratio (OR) for delirium in the postimplementation group was lower compared to the baseline pre-implementation group (OR, 0.54; 95% CI, 0.37-0.80; P < .001). Measures of advance care planning in the postimplementation group improved, including more frequent goals-of-care documentation (53.7% in postimplementation group vs 16.7% in baseline group; P < .001) and a shortened time to first goals-of-care discussion upon presenting to the ED (36 hours in postimplementation group vs 50 hours in baseline group; P = .03).
Conclusion: Implementation of a multidisciplinary geriatric trauma clinical pathway for older adults with traumatic injury at a single level I trauma center was associated with reduced rates of delirium.
Study 2 Overview (Bryant et al)
Objective: To determine whether an interdisciplinary care pathway for frail trauma patients can improve in-hospital mortality, complications, and 30-day readmissions.
Design: Retrospective cohort study of frail patients.
Setting and participants: Eligible patients were persons aged 65 years or older who were admitted to the trauma service and survived more than 24 hours; admitted to and discharged from the trauma unit; and determined to be pre-frail or frail by a geriatrician’s assessment. A Frailty Identification and Care Pathway designed to reduce delirium and complications in frail older trauma patients was developed by a multidisciplinary team and implemented in 2016. The standardized evidence-based interdisciplinary care pathway included utilization of order sets and interventions for delirium prevention, early ambulation, bowel and pain regimens, nutrition and physical therapy consults, medication management, care-goal setting, and geriatric assessments.
Main outcome measures: The main outcomes were delirium as determined by a positive CAM score, major complications as defined by the Trauma Quality Improvement Project, in-hospital mortality, and 30-day hospital readmission. Outcome measures were compared between patients who underwent Frailty Identification and Care Pathway intervention (postintervention group) vs patients who were treated prior to pathway implementation (pre-intervention group).
Main results: A total of 269 frail patients were included in the analysis (125 in pre-intervention group vs 144 in postintervention group). Patient demographic and admission characteristics were similar between the 2 groups: mean age was 83.5 (7.1) years, 60.6% were women, and median ISS was 10 (interquartile range [IQR], 9-14). The injury mechanism was similar between groups, with falls accounting for 92.8% and 86.1% of injuries in the pre-intervention and postintervention groups, respectively (P = .07). In univariate analysis, the Frailty Identification and Care Pathway intervention was associated with a significant reduction in delirium (12.5% vs 21.6%, P = .04) and 30-day hospital readmission (2.7% vs 9.6%, P = .01) compared to patients in the pre-intervention group. However, rates of major complications (28.5% vs 28.0%, P = 0.93) and in-hospital mortality (4.2% vs 7.2%, P = .28) were similar between the pre-intervention and postintervention groups. In multivariate logistic regression models adjusted for patient characteristics (age, sex, race, ISS), patients in the postintervention group had lower delirium (OR, 0.44; 95% CI, 0.22-0.88; P = .02) and 30-day hospital readmission (OR, 0.25; 95% CI, 0.07-0.84; P = .02) rates compared to those in the pre-intervention group.
Conclusion: Implementation of an interdisciplinary care protocol for frail geriatric trauma patients significantly decreased their risks for in-hospital delirium and 30-day hospital readmission.
Commentary
Traumatic injuries in older adults are associated with higher morbidity and mortality compared to younger patients, with falls and motor vehicle accidents accounting for a majority of these injuries. Astoundingly, up to one-third of this vulnerable population presenting to hospitals with an ISS greater than 15 may die during hospitalization.1 As a result, a large number of studies and clinical trials have focused on interventions that are designed to reduce fall risks, and hence reduce adverse consequences of traumatic injuries that may arise after falls.2 However, this emphasis on falls prevention has overshadowed a need to develop effective geriatric-centered clinical interventions that aim to improve outcomes in older adults who present to hospitals with traumatic injuries. Furthermore, frailty—a geriatric syndrome indicative of an increased state of vulnerability and predictive of adverse outcomes such as delirium—is highly prevalent in older patients with traumatic injury.3 Thus, there is an urgent need to develop novel, hospital-based, traumatic injury–targeting strategies that incorporate a thoughtful redesign of the care framework that includes evidence-based interventions for geriatric syndromes such as delirium and frailty.
The study reported by Park et al (Study 1) represents the latest effort to evaluate inpatient management strategies designed to improve outcomes in hospitalized older adults who have sustained traumatic injury. Through the implementation of a novel multidisciplinary Geriatric Trauma Care Pathway that incorporates geriatric best practices, this intervention was found to be associated with a 46% lower risk of in-hospital delirium. Because of the inclusion of all age-eligible patients across all strata of traumatic injuries, rather than preselecting for those at the highest risk for poor clinical outcomes, the benefits of this intervention extend to those with minor or moderate injury severity. Furthermore, the improvement in delirium (ie, the primary outcome) is particularly meaningful given that delirium is one of the most common hospital-associated complications that increase hospital LOS, discharge to an institution, and mortality in older adults. Finally, the study’s observed reduced time to a first goals-of-care discussion and increased frequency of goals-of-care documentation after intervention should not be overlooked. The improvements in these 2 process measures are highly significant given that advanced care planning, an intervention that helps to align patients’ values, goals, and treatments, is completed at substantially lower rates in older adults in the acute hospital setting.4
Similarly, in an earlier published study, Bryant and colleagues (Study 2) also show that a geriatric-focused interdisciplinary trauma care pathway is associated with delirium risk reduction in hospitalized older trauma patients. Much like Study 1, the Frailty Identification and Care Pathway utilized in Study 2 is an evidence-based interdisciplinary care pathway that includes the use of geriatric assessments, order sets, and geriatric best practices. Moreover, its exclusive inclusion of pre-frail and frail older patients (ie, those at higher risk for poor outcomes) with moderate injury severity (median ISS of 10 [IQR, 9-14]) suggests that this type of care pathway benefits hospitalized older trauma patients, who are particularly vulnerable to adverse complications such as delirium. Moreover, the successful utilization of the FRAIL questionnaire, a validated frailty screening tool, by surgical residents in the ED to initiate this care pathway demonstrates the feasibility of its use in expediting frailty screening in older patients in trauma care.
Application for Clinical Practice and System Implementation
Findings from the 2 studies discussed in this review indicate that implementation of interdisciplinary clinical care pathways predicated on evidence-based geriatric principles and best practices is a promising approach to reduce delirium in hospitalized older trauma patients. These studies have helped to lay the groundwork in outlining the roadmaps (eg, processes and infrastructures) needed to create such clinical pathways. These key elements include: (1) integration of a multidisciplinary committee (eg, representation from trauma, emergency, and geriatric medicine, nursing, physical and occupational therapy, pharmacy, social work) in pathway design and implementation; (2) adaption of evidence-based geriatric best practices (eg, the Institute for Healthcare Improvement Age-Friendly Health System 4M framework [medication, mentation, mobility, what matters]) to prioritize interventions and to design a pathway that incorporates these features; (3) incorporation of comprehensive geriatric assessment by interdisciplinary providers; (4) utilization of validated clinical instruments to assess physical and cognitive functions, frailty, delirium, and social determinants of health; (5) modification of electronic health record systems to encompass order sets that incorporate evidence-based, nonpharmacological and pharmacological interventions to manage symptoms (eg, delirium, pain, bowel movement, sleep, immobility, polypharmacy) essential to quality geriatric care; and (6) integration of patient and caregiver preferences via goals-of-care discussions and corresponding documentation and communication of these goals.
Additionally, these 2 studies imparted some strategies that may facilitate the implementation of interdisciplinary clinical care pathways in trauma care. Examples of such facilitators include: (1) collaboration with champions within each specialty to reinforce education and buy-in; (2) creation of automatically triggered order sets upon patient presentation to the ED that unites distinct features of clinical pathways; (3) adaption and reorganization of existing hospital infrastructures and resources to meet the needs of clinical pathways implementation (eg, utilizing information technology resources to develop electronic health record order sets; using quality department to develop clinical pathway guidelines and electronic outcome dashboards); and (4) development of individualized patient and caregiver education materials based on care needs (eg, principles of delirium prevention and preservation of mobility during hospitalization) to prepare and engage these stakeholders in patient care and recovery.
Practice Points
- A geriatric interdisciplinary care model can be effectively applied to the management of acute trauma in older patients.
- Interdisciplinary clinical pathways should incorporate geriatric best practices and guidelines and age-appropriate order sets to prioritize and integrate care.
—Fred Ko, MD, MS
Study 1 Overview (Park et al)
Objective: To examine whether implementation of a geriatric trauma clinical pathway is associated with reduced rates of delirium in older adults with traumatic injury.
Design: Retrospective case-control study of electronic health records.
Setting and participants: Eligible patients were persons aged 65 years or older who were admitted to the trauma service and did not undergo an operation. A Geriatric Trauma Care Pathway was developed by a multidisciplinary Stanford Quality Pathways team and formally launched on November 1, 2018. The clinical pathway was designed to incorporate geriatric best practices, which included order sets (eg, age-appropriate nonpharmacological interventions and pharmacological dosages), guidelines (eg, Institute for Healthcare Improvement Age-Friendly Health systems 4M framework), automated consultations (comprehensive geriatric assessment), and escalation pathways executed by a multidisciplinary team (eg, pain, bowel, and sleep regulation). The clinical pathway began with admission to the emergency department (ED) (ie, automatic trigger of geriatric trauma care admission order set), daily multidisciplinary team meetings during acute hospitalization, and a transitional care team consultation for postdischarge follow-up or home visit.
Main outcome measures: The primary outcome was delirium as determined by a positive Confusion Assessment Method (CAM) score or a diagnosis of delirium by the clinical team. The secondary outcome was hospital length of stay (LOS). Process measures for pathway compliance (eg, achieving adequate pain control, early mobilization, advance care planning) were assessed. Outcome measures were compared between patients who underwent the Geriatric Trauma Care Pathway intervention (postimplementation group) vs patients who were treated prior to pathway implementation (baseline pre-implementation group).
Main results: Of the 859 eligible patients, 712 were included in the analysis (442 [62.1%] in the baseline pre-implementation group and 270 [37.9%] in the postimplementation group); mean (SD) age was 81.4 (9.1) years, and 394 (55.3%) were women. The injury mechanism was similar between groups, with falls being the most common cause of injury (247 [55.9%] in the baseline group vs 162 [60.0%] in the postimplementation group; P = .43). Injuries as measured by Injury Severity Score (ISS) were minor or moderate in both groups (261 [59.0%] in baseline group vs 168 [62.2%] in postimplementation group; P = .87). The adjusted odds ratio (OR) for delirium in the postimplementation group was lower compared to the baseline pre-implementation group (OR, 0.54; 95% CI, 0.37-0.80; P < .001). Measures of advance care planning in the postimplementation group improved, including more frequent goals-of-care documentation (53.7% in postimplementation group vs 16.7% in baseline group; P < .001) and a shortened time to first goals-of-care discussion upon presenting to the ED (36 hours in postimplementation group vs 50 hours in baseline group; P = .03).
Conclusion: Implementation of a multidisciplinary geriatric trauma clinical pathway for older adults with traumatic injury at a single level I trauma center was associated with reduced rates of delirium.
Study 2 Overview (Bryant et al)
Objective: To determine whether an interdisciplinary care pathway for frail trauma patients can improve in-hospital mortality, complications, and 30-day readmissions.
Design: Retrospective cohort study of frail patients.
Setting and participants: Eligible patients were persons aged 65 years or older who were admitted to the trauma service and survived more than 24 hours; admitted to and discharged from the trauma unit; and determined to be pre-frail or frail by a geriatrician’s assessment. A Frailty Identification and Care Pathway designed to reduce delirium and complications in frail older trauma patients was developed by a multidisciplinary team and implemented in 2016. The standardized evidence-based interdisciplinary care pathway included utilization of order sets and interventions for delirium prevention, early ambulation, bowel and pain regimens, nutrition and physical therapy consults, medication management, care-goal setting, and geriatric assessments.
Main outcome measures: The main outcomes were delirium as determined by a positive CAM score, major complications as defined by the Trauma Quality Improvement Project, in-hospital mortality, and 30-day hospital readmission. Outcome measures were compared between patients who underwent Frailty Identification and Care Pathway intervention (postintervention group) vs patients who were treated prior to pathway implementation (pre-intervention group).
Main results: A total of 269 frail patients were included in the analysis (125 in pre-intervention group vs 144 in postintervention group). Patient demographic and admission characteristics were similar between the 2 groups: mean age was 83.5 (7.1) years, 60.6% were women, and median ISS was 10 (interquartile range [IQR], 9-14). The injury mechanism was similar between groups, with falls accounting for 92.8% and 86.1% of injuries in the pre-intervention and postintervention groups, respectively (P = .07). In univariate analysis, the Frailty Identification and Care Pathway intervention was associated with a significant reduction in delirium (12.5% vs 21.6%, P = .04) and 30-day hospital readmission (2.7% vs 9.6%, P = .01) compared to patients in the pre-intervention group. However, rates of major complications (28.5% vs 28.0%, P = 0.93) and in-hospital mortality (4.2% vs 7.2%, P = .28) were similar between the pre-intervention and postintervention groups. In multivariate logistic regression models adjusted for patient characteristics (age, sex, race, ISS), patients in the postintervention group had lower delirium (OR, 0.44; 95% CI, 0.22-0.88; P = .02) and 30-day hospital readmission (OR, 0.25; 95% CI, 0.07-0.84; P = .02) rates compared to those in the pre-intervention group.
Conclusion: Implementation of an interdisciplinary care protocol for frail geriatric trauma patients significantly decreased their risks for in-hospital delirium and 30-day hospital readmission.
Commentary
Traumatic injuries in older adults are associated with higher morbidity and mortality compared to younger patients, with falls and motor vehicle accidents accounting for a majority of these injuries. Astoundingly, up to one-third of this vulnerable population presenting to hospitals with an ISS greater than 15 may die during hospitalization.1 As a result, a large number of studies and clinical trials have focused on interventions that are designed to reduce fall risks, and hence reduce adverse consequences of traumatic injuries that may arise after falls.2 However, this emphasis on falls prevention has overshadowed a need to develop effective geriatric-centered clinical interventions that aim to improve outcomes in older adults who present to hospitals with traumatic injuries. Furthermore, frailty—a geriatric syndrome indicative of an increased state of vulnerability and predictive of adverse outcomes such as delirium—is highly prevalent in older patients with traumatic injury.3 Thus, there is an urgent need to develop novel, hospital-based, traumatic injury–targeting strategies that incorporate a thoughtful redesign of the care framework that includes evidence-based interventions for geriatric syndromes such as delirium and frailty.
The study reported by Park et al (Study 1) represents the latest effort to evaluate inpatient management strategies designed to improve outcomes in hospitalized older adults who have sustained traumatic injury. Through the implementation of a novel multidisciplinary Geriatric Trauma Care Pathway that incorporates geriatric best practices, this intervention was found to be associated with a 46% lower risk of in-hospital delirium. Because of the inclusion of all age-eligible patients across all strata of traumatic injuries, rather than preselecting for those at the highest risk for poor clinical outcomes, the benefits of this intervention extend to those with minor or moderate injury severity. Furthermore, the improvement in delirium (ie, the primary outcome) is particularly meaningful given that delirium is one of the most common hospital-associated complications that increase hospital LOS, discharge to an institution, and mortality in older adults. Finally, the study’s observed reduced time to a first goals-of-care discussion and increased frequency of goals-of-care documentation after intervention should not be overlooked. The improvements in these 2 process measures are highly significant given that advanced care planning, an intervention that helps to align patients’ values, goals, and treatments, is completed at substantially lower rates in older adults in the acute hospital setting.4
Similarly, in an earlier published study, Bryant and colleagues (Study 2) also show that a geriatric-focused interdisciplinary trauma care pathway is associated with delirium risk reduction in hospitalized older trauma patients. Much like Study 1, the Frailty Identification and Care Pathway utilized in Study 2 is an evidence-based interdisciplinary care pathway that includes the use of geriatric assessments, order sets, and geriatric best practices. Moreover, its exclusive inclusion of pre-frail and frail older patients (ie, those at higher risk for poor outcomes) with moderate injury severity (median ISS of 10 [IQR, 9-14]) suggests that this type of care pathway benefits hospitalized older trauma patients, who are particularly vulnerable to adverse complications such as delirium. Moreover, the successful utilization of the FRAIL questionnaire, a validated frailty screening tool, by surgical residents in the ED to initiate this care pathway demonstrates the feasibility of its use in expediting frailty screening in older patients in trauma care.
Application for Clinical Practice and System Implementation
Findings from the 2 studies discussed in this review indicate that implementation of interdisciplinary clinical care pathways predicated on evidence-based geriatric principles and best practices is a promising approach to reduce delirium in hospitalized older trauma patients. These studies have helped to lay the groundwork in outlining the roadmaps (eg, processes and infrastructures) needed to create such clinical pathways. These key elements include: (1) integration of a multidisciplinary committee (eg, representation from trauma, emergency, and geriatric medicine, nursing, physical and occupational therapy, pharmacy, social work) in pathway design and implementation; (2) adaption of evidence-based geriatric best practices (eg, the Institute for Healthcare Improvement Age-Friendly Health System 4M framework [medication, mentation, mobility, what matters]) to prioritize interventions and to design a pathway that incorporates these features; (3) incorporation of comprehensive geriatric assessment by interdisciplinary providers; (4) utilization of validated clinical instruments to assess physical and cognitive functions, frailty, delirium, and social determinants of health; (5) modification of electronic health record systems to encompass order sets that incorporate evidence-based, nonpharmacological and pharmacological interventions to manage symptoms (eg, delirium, pain, bowel movement, sleep, immobility, polypharmacy) essential to quality geriatric care; and (6) integration of patient and caregiver preferences via goals-of-care discussions and corresponding documentation and communication of these goals.
Additionally, these 2 studies imparted some strategies that may facilitate the implementation of interdisciplinary clinical care pathways in trauma care. Examples of such facilitators include: (1) collaboration with champions within each specialty to reinforce education and buy-in; (2) creation of automatically triggered order sets upon patient presentation to the ED that unites distinct features of clinical pathways; (3) adaption and reorganization of existing hospital infrastructures and resources to meet the needs of clinical pathways implementation (eg, utilizing information technology resources to develop electronic health record order sets; using quality department to develop clinical pathway guidelines and electronic outcome dashboards); and (4) development of individualized patient and caregiver education materials based on care needs (eg, principles of delirium prevention and preservation of mobility during hospitalization) to prepare and engage these stakeholders in patient care and recovery.
Practice Points
- A geriatric interdisciplinary care model can be effectively applied to the management of acute trauma in older patients.
- Interdisciplinary clinical pathways should incorporate geriatric best practices and guidelines and age-appropriate order sets to prioritize and integrate care.
—Fred Ko, MD, MS
1. Hashmi A, Ibrahim-Zada I, Rhee P, et al. Predictors of mortality in geriatric trauma patients: a systematic review and meta-analysis. J Trauma Acute Care Surg. 2014;76(3):894-901. doi:10.1097/TA.0b013e3182ab0763
2. Hopewell S, Adedire O, Copsey BJ, et al. Multifactorial and multiple component interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2018;7(7):CD012221. doi:10.1002/14651858.CD012221.pub2
3. Joseph B, Pandit V, Zangbar B, et al. Superiority of frailty over age in predicting outcomes among geriatric trauma patients: a prospective analysis. JAMA Surg. 2014;149(8):766-772. doi:10.1001/jamasurg.2014.296
4. Hopkins SA, Bentley A, Phillips V, Barclay S. Advance care plans and hospitalized frail older adults: a systematic review. BMJ Support Palliat Care. 2020;10(2):164-174. doi:10.1136/bmjspcare-2019-002093
1. Hashmi A, Ibrahim-Zada I, Rhee P, et al. Predictors of mortality in geriatric trauma patients: a systematic review and meta-analysis. J Trauma Acute Care Surg. 2014;76(3):894-901. doi:10.1097/TA.0b013e3182ab0763
2. Hopewell S, Adedire O, Copsey BJ, et al. Multifactorial and multiple component interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2018;7(7):CD012221. doi:10.1002/14651858.CD012221.pub2
3. Joseph B, Pandit V, Zangbar B, et al. Superiority of frailty over age in predicting outcomes among geriatric trauma patients: a prospective analysis. JAMA Surg. 2014;149(8):766-772. doi:10.1001/jamasurg.2014.296
4. Hopkins SA, Bentley A, Phillips V, Barclay S. Advance care plans and hospitalized frail older adults: a systematic review. BMJ Support Palliat Care. 2020;10(2):164-174. doi:10.1136/bmjspcare-2019-002093
Hospital-acquired pneumonia is killing patients, yet there is a simple way to stop it
Four years ago, when Dr. Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside were tissues, bar soap, deodorant, toothpaste, and, without a doubt, the worst toothbrush she’d ever seen.
“I couldn’t believe it. I got a toothbrush with no bristles,” she said. “It must have not gone through the bristle machine. It was just a stick.”
To most patients, a useless hospital toothbrush would be a mild inconvenience. But to Dr. Giuliano, a nursing professor at the University of Massachusetts, Amherst, it was a reminder of a pervasive “blind spot” in U.S. hospitals: the stunning consequences of unbrushed teeth.
Hospital patients not getting their teeth brushed, or not brushing their teeth themselves, is believed to be a leading cause of hundreds of thousands of cases of pneumonia a year in patients who have not been put on a ventilator. Pneumonia is among the most common infections that occur in health care facilities, and a majority of cases are nonventilator hospital-acquired pneumonia, or NVHAP, which kills up to 30% of those infected, Dr. Giuliano and other experts said.
But unlike many infections that strike within hospitals, the federal government doesn’t require hospitals to report cases of NVHAP. As a result, few hospitals understand the origin of the illness, track its occurrence, or actively work to prevent it, the experts said.
, according to a growing body of peer-reviewed research papers. Instead, many hospitals often skip teeth brushing to prioritize other tasks and provide only cheap, ineffective toothbrushes, often unaware of the consequences, said Dr. Dian Baker, a Sacramento (Calif.) State nursing professor who has spent more than a decade studying NVHAP.
“I’ll tell you that today the vast majority of the tens of thousands of nurses in hospitals have no idea that pneumonia comes from germs in the mouth,” Dr. Baker said.
Pneumonia occurs when germs trigger an infection in the lungs. Although NVHAP accounts for most of the cases that occur in hospitals, it historically has not received the same attention as pneumonia tied to ventilators, which is easier to identify and study because it occurs among a narrow subset of patients.
NVHAP, a risk for virtually all hospital patients, is often caused by bacteria from the mouth that gathers in the scummy biofilm on unbrushed teeth and is aspirated into the lungs. Patients face a higher risk if they lie flat or remain immobile for long periods, so NVHAP can also be prevented by elevating their heads and getting them out of bed more often.
According to the National Organization for NV-HAP Prevention, which was founded in 2020, this pneumonia infects about 1 in every 100 hospital patients and kills 15%-30% of them. For those who survive, the illness often extends their hospital stay by up to 15 days and makes it much more likely they will be readmitted within a month or transferred to an intensive care unit.
John McCleary, 83, of Millinocket, Maine, contracted a likely case of NVHAP in 2008 after he fractured his ankle in a fall and spent 12 days in rehabilitation at a hospital, said his daughter, Kathy Day, a retired nurse and advocate with the Patient Safety Action Network.
Mr. McCleary recovered from the fracture but not from pneumonia. Two days after he returned home, the infection in his lungs caused him to be rushed back to the hospital, where he went into sepsis and spent weeks in treatment before moving to an isolation unit in a nursing home.
He died weeks later, emaciated, largely deaf, unable to eat, and often “too weak to get water through a straw,” his daughter said. After contracting pneumonia, he never walked again.
“It was an astounding assault on his body, from him being here visiting me the week before his fall, to his death just a few months later,” Ms. Day said. “And the whole thing was avoidable.”
While experts describe NVHAP as a largely ignored threat, that appears to be changing.
Last year, a group of researchers – including Dr. Giuliano and Dr. Baker, plus officials from the Centers for Disease Control and Prevention, the Veterans Health Administration, and the Joint Commission – published a “call-to-action” research paper hoping to launch “a national health care conversation about NVHAP prevention.”
The Joint Commission, a nonprofit organization whose accreditation can make or break hospitals, is considering broadening the infection control standards to include more ailments, including NVHAP, said Sylvia Garcia-Houchins, its director of infection prevention and control.
Separately, ECRI, a nonprofit focused on health care safety, this year pinpointed NVHAP as one of its top patient safety concerns.
James Davis, an ECRI infection expert, said the prevalence of NVHAP, while already alarming, is likely “underestimated” and probably worsened as hospitals swelled with patients during the coronavirus pandemic.
“We only know what’s reported,” Mr. Davis said. “Could this be the tip of the iceberg? I would say, in my opinion, probably.”
To better measure the condition, some researchers call for a standardized surveillance definition of NVHAP, which could in time open the door for the federal government to mandate reporting of cases or incentivize prevention. With increasing urgency, researchers are pushing for hospitals not to wait for the federal government to act against NVHAP.
Dr. Baker said she has spoken with hundreds of hospitals about how to prevent NVHAP, but thousands more have yet to take up the cause.
“We are not asking for some big, $300,000 piece of equipment,” Dr. Baker said. “The two things that show the best evidence of preventing this harm are things that should be happening in standard care anyway – brushing teeth and getting patients mobilized.”
That evidence comes from a smattering of studies that show those two strategies can lead to sharp reductions in infection rates.
In California, a study at 21 Kaiser Permanente hospitals used a reprioritization of oral care and getting patients out of bed to reduce rates of hospital-acquired pneumonia by around 70%. At Sutter Medical Center in Sacramento, better oral care reduced NVHAP cases by a yearly average of 35%.
At Orlando Regional Medical Center in Florida, a medical unit and a surgical unit where patients received enhanced oral care reduced NVHAP rates by 85% and 56%, respectively, when compared with similar units that received normal care. A similar study is underway at two hospitals in Illinois.
And the most compelling results come from a veterans’ hospital in Salem, Va., where a 2016 oral care pilot program reduced rates of NVHAP by 92% – saving an estimated 13 lives in just 19 months. The program, the HAPPEN Initiative, has been expanded across the Veterans Health Administration, and experts say it could serve as a model for all U.S. hospitals.
Dr. Michelle Lucatorto, a nursing official who leads HAPPEN, said the program trains nurses to most effectively brush patients’ teeth and educates patients and families on the link between oral care and preventing NVHAP. While teeth brushing may not seem to require training, Dr. Lucatorto made comparisons to how the coronavirus revealed many Americans were doing a lackluster job of another routine hygienic practice: washing their hands.
“Sometimes we are searching for the most complicated intervention,” she said. “We are always looking for that new bypass surgery, or some new technical equipment. And sometimes I think we fail to look at the simple things we can do in our practice to save people’s lives.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Four years ago, when Dr. Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside were tissues, bar soap, deodorant, toothpaste, and, without a doubt, the worst toothbrush she’d ever seen.
“I couldn’t believe it. I got a toothbrush with no bristles,” she said. “It must have not gone through the bristle machine. It was just a stick.”
To most patients, a useless hospital toothbrush would be a mild inconvenience. But to Dr. Giuliano, a nursing professor at the University of Massachusetts, Amherst, it was a reminder of a pervasive “blind spot” in U.S. hospitals: the stunning consequences of unbrushed teeth.
Hospital patients not getting their teeth brushed, or not brushing their teeth themselves, is believed to be a leading cause of hundreds of thousands of cases of pneumonia a year in patients who have not been put on a ventilator. Pneumonia is among the most common infections that occur in health care facilities, and a majority of cases are nonventilator hospital-acquired pneumonia, or NVHAP, which kills up to 30% of those infected, Dr. Giuliano and other experts said.
But unlike many infections that strike within hospitals, the federal government doesn’t require hospitals to report cases of NVHAP. As a result, few hospitals understand the origin of the illness, track its occurrence, or actively work to prevent it, the experts said.
, according to a growing body of peer-reviewed research papers. Instead, many hospitals often skip teeth brushing to prioritize other tasks and provide only cheap, ineffective toothbrushes, often unaware of the consequences, said Dr. Dian Baker, a Sacramento (Calif.) State nursing professor who has spent more than a decade studying NVHAP.
“I’ll tell you that today the vast majority of the tens of thousands of nurses in hospitals have no idea that pneumonia comes from germs in the mouth,” Dr. Baker said.
Pneumonia occurs when germs trigger an infection in the lungs. Although NVHAP accounts for most of the cases that occur in hospitals, it historically has not received the same attention as pneumonia tied to ventilators, which is easier to identify and study because it occurs among a narrow subset of patients.
NVHAP, a risk for virtually all hospital patients, is often caused by bacteria from the mouth that gathers in the scummy biofilm on unbrushed teeth and is aspirated into the lungs. Patients face a higher risk if they lie flat or remain immobile for long periods, so NVHAP can also be prevented by elevating their heads and getting them out of bed more often.
According to the National Organization for NV-HAP Prevention, which was founded in 2020, this pneumonia infects about 1 in every 100 hospital patients and kills 15%-30% of them. For those who survive, the illness often extends their hospital stay by up to 15 days and makes it much more likely they will be readmitted within a month or transferred to an intensive care unit.
John McCleary, 83, of Millinocket, Maine, contracted a likely case of NVHAP in 2008 after he fractured his ankle in a fall and spent 12 days in rehabilitation at a hospital, said his daughter, Kathy Day, a retired nurse and advocate with the Patient Safety Action Network.
Mr. McCleary recovered from the fracture but not from pneumonia. Two days after he returned home, the infection in his lungs caused him to be rushed back to the hospital, where he went into sepsis and spent weeks in treatment before moving to an isolation unit in a nursing home.
He died weeks later, emaciated, largely deaf, unable to eat, and often “too weak to get water through a straw,” his daughter said. After contracting pneumonia, he never walked again.
“It was an astounding assault on his body, from him being here visiting me the week before his fall, to his death just a few months later,” Ms. Day said. “And the whole thing was avoidable.”
While experts describe NVHAP as a largely ignored threat, that appears to be changing.
Last year, a group of researchers – including Dr. Giuliano and Dr. Baker, plus officials from the Centers for Disease Control and Prevention, the Veterans Health Administration, and the Joint Commission – published a “call-to-action” research paper hoping to launch “a national health care conversation about NVHAP prevention.”
The Joint Commission, a nonprofit organization whose accreditation can make or break hospitals, is considering broadening the infection control standards to include more ailments, including NVHAP, said Sylvia Garcia-Houchins, its director of infection prevention and control.
Separately, ECRI, a nonprofit focused on health care safety, this year pinpointed NVHAP as one of its top patient safety concerns.
James Davis, an ECRI infection expert, said the prevalence of NVHAP, while already alarming, is likely “underestimated” and probably worsened as hospitals swelled with patients during the coronavirus pandemic.
“We only know what’s reported,” Mr. Davis said. “Could this be the tip of the iceberg? I would say, in my opinion, probably.”
To better measure the condition, some researchers call for a standardized surveillance definition of NVHAP, which could in time open the door for the federal government to mandate reporting of cases or incentivize prevention. With increasing urgency, researchers are pushing for hospitals not to wait for the federal government to act against NVHAP.
Dr. Baker said she has spoken with hundreds of hospitals about how to prevent NVHAP, but thousands more have yet to take up the cause.
“We are not asking for some big, $300,000 piece of equipment,” Dr. Baker said. “The two things that show the best evidence of preventing this harm are things that should be happening in standard care anyway – brushing teeth and getting patients mobilized.”
That evidence comes from a smattering of studies that show those two strategies can lead to sharp reductions in infection rates.
In California, a study at 21 Kaiser Permanente hospitals used a reprioritization of oral care and getting patients out of bed to reduce rates of hospital-acquired pneumonia by around 70%. At Sutter Medical Center in Sacramento, better oral care reduced NVHAP cases by a yearly average of 35%.
At Orlando Regional Medical Center in Florida, a medical unit and a surgical unit where patients received enhanced oral care reduced NVHAP rates by 85% and 56%, respectively, when compared with similar units that received normal care. A similar study is underway at two hospitals in Illinois.
And the most compelling results come from a veterans’ hospital in Salem, Va., where a 2016 oral care pilot program reduced rates of NVHAP by 92% – saving an estimated 13 lives in just 19 months. The program, the HAPPEN Initiative, has been expanded across the Veterans Health Administration, and experts say it could serve as a model for all U.S. hospitals.
Dr. Michelle Lucatorto, a nursing official who leads HAPPEN, said the program trains nurses to most effectively brush patients’ teeth and educates patients and families on the link between oral care and preventing NVHAP. While teeth brushing may not seem to require training, Dr. Lucatorto made comparisons to how the coronavirus revealed many Americans were doing a lackluster job of another routine hygienic practice: washing their hands.
“Sometimes we are searching for the most complicated intervention,” she said. “We are always looking for that new bypass surgery, or some new technical equipment. And sometimes I think we fail to look at the simple things we can do in our practice to save people’s lives.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Four years ago, when Dr. Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside were tissues, bar soap, deodorant, toothpaste, and, without a doubt, the worst toothbrush she’d ever seen.
“I couldn’t believe it. I got a toothbrush with no bristles,” she said. “It must have not gone through the bristle machine. It was just a stick.”
To most patients, a useless hospital toothbrush would be a mild inconvenience. But to Dr. Giuliano, a nursing professor at the University of Massachusetts, Amherst, it was a reminder of a pervasive “blind spot” in U.S. hospitals: the stunning consequences of unbrushed teeth.
Hospital patients not getting their teeth brushed, or not brushing their teeth themselves, is believed to be a leading cause of hundreds of thousands of cases of pneumonia a year in patients who have not been put on a ventilator. Pneumonia is among the most common infections that occur in health care facilities, and a majority of cases are nonventilator hospital-acquired pneumonia, or NVHAP, which kills up to 30% of those infected, Dr. Giuliano and other experts said.
But unlike many infections that strike within hospitals, the federal government doesn’t require hospitals to report cases of NVHAP. As a result, few hospitals understand the origin of the illness, track its occurrence, or actively work to prevent it, the experts said.
, according to a growing body of peer-reviewed research papers. Instead, many hospitals often skip teeth brushing to prioritize other tasks and provide only cheap, ineffective toothbrushes, often unaware of the consequences, said Dr. Dian Baker, a Sacramento (Calif.) State nursing professor who has spent more than a decade studying NVHAP.
“I’ll tell you that today the vast majority of the tens of thousands of nurses in hospitals have no idea that pneumonia comes from germs in the mouth,” Dr. Baker said.
Pneumonia occurs when germs trigger an infection in the lungs. Although NVHAP accounts for most of the cases that occur in hospitals, it historically has not received the same attention as pneumonia tied to ventilators, which is easier to identify and study because it occurs among a narrow subset of patients.
NVHAP, a risk for virtually all hospital patients, is often caused by bacteria from the mouth that gathers in the scummy biofilm on unbrushed teeth and is aspirated into the lungs. Patients face a higher risk if they lie flat or remain immobile for long periods, so NVHAP can also be prevented by elevating their heads and getting them out of bed more often.
According to the National Organization for NV-HAP Prevention, which was founded in 2020, this pneumonia infects about 1 in every 100 hospital patients and kills 15%-30% of them. For those who survive, the illness often extends their hospital stay by up to 15 days and makes it much more likely they will be readmitted within a month or transferred to an intensive care unit.
John McCleary, 83, of Millinocket, Maine, contracted a likely case of NVHAP in 2008 after he fractured his ankle in a fall and spent 12 days in rehabilitation at a hospital, said his daughter, Kathy Day, a retired nurse and advocate with the Patient Safety Action Network.
Mr. McCleary recovered from the fracture but not from pneumonia. Two days after he returned home, the infection in his lungs caused him to be rushed back to the hospital, where he went into sepsis and spent weeks in treatment before moving to an isolation unit in a nursing home.
He died weeks later, emaciated, largely deaf, unable to eat, and often “too weak to get water through a straw,” his daughter said. After contracting pneumonia, he never walked again.
“It was an astounding assault on his body, from him being here visiting me the week before his fall, to his death just a few months later,” Ms. Day said. “And the whole thing was avoidable.”
While experts describe NVHAP as a largely ignored threat, that appears to be changing.
Last year, a group of researchers – including Dr. Giuliano and Dr. Baker, plus officials from the Centers for Disease Control and Prevention, the Veterans Health Administration, and the Joint Commission – published a “call-to-action” research paper hoping to launch “a national health care conversation about NVHAP prevention.”
The Joint Commission, a nonprofit organization whose accreditation can make or break hospitals, is considering broadening the infection control standards to include more ailments, including NVHAP, said Sylvia Garcia-Houchins, its director of infection prevention and control.
Separately, ECRI, a nonprofit focused on health care safety, this year pinpointed NVHAP as one of its top patient safety concerns.
James Davis, an ECRI infection expert, said the prevalence of NVHAP, while already alarming, is likely “underestimated” and probably worsened as hospitals swelled with patients during the coronavirus pandemic.
“We only know what’s reported,” Mr. Davis said. “Could this be the tip of the iceberg? I would say, in my opinion, probably.”
To better measure the condition, some researchers call for a standardized surveillance definition of NVHAP, which could in time open the door for the federal government to mandate reporting of cases or incentivize prevention. With increasing urgency, researchers are pushing for hospitals not to wait for the federal government to act against NVHAP.
Dr. Baker said she has spoken with hundreds of hospitals about how to prevent NVHAP, but thousands more have yet to take up the cause.
“We are not asking for some big, $300,000 piece of equipment,” Dr. Baker said. “The two things that show the best evidence of preventing this harm are things that should be happening in standard care anyway – brushing teeth and getting patients mobilized.”
That evidence comes from a smattering of studies that show those two strategies can lead to sharp reductions in infection rates.
In California, a study at 21 Kaiser Permanente hospitals used a reprioritization of oral care and getting patients out of bed to reduce rates of hospital-acquired pneumonia by around 70%. At Sutter Medical Center in Sacramento, better oral care reduced NVHAP cases by a yearly average of 35%.
At Orlando Regional Medical Center in Florida, a medical unit and a surgical unit where patients received enhanced oral care reduced NVHAP rates by 85% and 56%, respectively, when compared with similar units that received normal care. A similar study is underway at two hospitals in Illinois.
And the most compelling results come from a veterans’ hospital in Salem, Va., where a 2016 oral care pilot program reduced rates of NVHAP by 92% – saving an estimated 13 lives in just 19 months. The program, the HAPPEN Initiative, has been expanded across the Veterans Health Administration, and experts say it could serve as a model for all U.S. hospitals.
Dr. Michelle Lucatorto, a nursing official who leads HAPPEN, said the program trains nurses to most effectively brush patients’ teeth and educates patients and families on the link between oral care and preventing NVHAP. While teeth brushing may not seem to require training, Dr. Lucatorto made comparisons to how the coronavirus revealed many Americans were doing a lackluster job of another routine hygienic practice: washing their hands.
“Sometimes we are searching for the most complicated intervention,” she said. “We are always looking for that new bypass surgery, or some new technical equipment. And sometimes I think we fail to look at the simple things we can do in our practice to save people’s lives.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Effect of Pharmacist Interventions on Hospital Readmissions for Home-Based Primary Care Veterans
Following hospital discharge, patients are often in a vulnerable state due to new medical diagnoses, changes in medications, lack of understanding, and concerns for medical costs. In addition, the discharge process is complex and encompasses decisions regarding the postdischarge site of care, conveying patient instructions, and obtaining supplies and medications. There are several disciplines involved in the transitions of care process that are all essential for ensuring a successful transition and reducing the risk of hospital readmissions. Pharmacists play an integral role in the process.
When pharmacists are provided the opportunity to make therapeutic interventions, medication errors and hospital readmissions decrease and quality of life improves.1 Studies have shown that many older patients return home from the hospital with a limited understanding of their discharge instructions and oftentimes are unable to recall their discharge diagnoses and treatment plan, leaving opportunities for error when patients transition from one level of care to another.2,3 Additionally, high-quality transitional care is especially important for older adults with multiple comorbidities and complex therapeutic regimens as well as for their families and caregivers.4 To prevent hospital readmissions, pharmacists and other health care professionals (HCPs) should work diligently to prevent gaps in care as patients transition between settings. Common factors that lead to increased readmissions include premature discharge, inadequate follow-up, therapeutic errors, and medication-related problems. Furthermore, unintended hospital readmissions are common within the first 30 days following hospital discharge and lead to increased health care costs.2 For these reasons, many health care institutions have developed comprehensive models to improve the discharge process, decrease hospital readmissions, and reduce incidence of adverse events in general medical patients and high-risk populations.5
A study evaluating 693 hospital discharges found that 27.6% of patients were recommended for outpatient workups; however only 9% were actually completed.6 Due to lack of communication regarding discharge summaries, primary care practitioners (PCPs) were unaware of the need for outpatient workups; thus, these patients were lost to follow-up, and appropriate care was not received. Future studies should focus on interventions to improve the quality and dissemination of discharge information to PCPs.6 Fosnight and colleagues assessed a new transitions process focusing on the role of pharmacists. They evaluated medication reconciliations performed and discussed medication adherence barriers, medication recommendations, and time spent performing the interventions.7 After patients received a pharmacy intervention, Fosnight and colleagues reported that readmission rates decreased from 21.0% to 15.3% and mean length of stay decreased from 5.3 to 4.4 days. They also observed greater improvements in patients who received the full pharmacy intervention vs those receiving only parts of the intervention. This study concluded that adding a comprehensive pharmacy intervention to transitions of care resulted in an average of nearly 10 medication recommendations per patient, improved length of stay, and reduced readmission rates. After a review of similar studies, we concluded that a comprehensive discharge model is imperative to improve patient outcomes, along with HCP monitoring of the process to ensure appropriate follow-up.8
At Michael E. DeBakey Veteran Affairs Medical Center (MEDVAMC) in Houston, Texas, 30-day readmissions data were reviewed for veterans 6 months before and 12 months after enrollment in the Home-Based Primary Care (HBPC) service. HBPC is an in-home health care service provided to home-bound veterans with complex health care needs or when routine clinic-based care is not feasible. HBPC programs may differ among various US Department of Veterans Affairs (VA) medical centers. Currently, there are 9 HBPC teams at MEDVAMC and nearly 540 veterans are enrolled in the program. HBPC teams typically consist of PCPs, pharmacists, nurses, psychologists, occupational/physical therapists, social workers, medical support assistants, and dietitians.
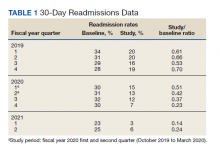
Readmissions data are reviewed quarterly by fiscal year (FY) (Table 1). In FY 2019 quarter (Q) 2, the readmission rate before HBPC enrollment was 31% and decreased to 20% after enrollment. In FY 2019 Q3, the readmission rate was 29% before enrollment and decreased to 16% afterward. In FY 2019 Q4, the readmission rate before HBPC enrollment was 28% and decreased to 19% afterward. Although the readmission rates appeared to be decreasing overall, improvements were needed to decrease these rates further and to ensure readmissions were not rising as there was a slight increase in Q4. After reviewing these data, the HBPC service implemented a streamlined hospital discharge process to lower readmission rates and improve patient outcomes.
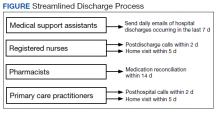
HBPC at MEDVAMC incorporates a team-based approach and the new streamlined discharge process implemented in 2019 highlights the role of each team member (Figure). Medical support assistants send daily emails of hospital discharges occurring in the last 7 days. Registered nurses are responsible for postdischarge calls within 2 days and home visits within 5 days. Pharmacists perform medication reconciliation within 14 days of discharge, review and/or educate on new medications, and change medications. The PCP is responsible for posthospital calls within 2 days and conducts a home visit within 5 days. Because HBPC programs vary among VA medical centers, the streamlined discharge process discussed may be applicable only to MEDVAMC. The primary objective of this quality improvement project was to identify specific pharmacist interventions to improve the HBPC discharge process and improve hospital readmission rates.
Methods
We conducted a Plan-Do-Study-Act quality improvement project. The first step was to conduct a review of veterans enrolled in HBPC at MEDVAMC.9 Patients included were enrolled in HBPC at MEDVAMC from October 2019 to March 2020 (FY 2020 Q1 and Q2). The Computerized Patient Record System was used to access the patients’ electronic health records. Patient information collected included race, age, sex, admission diagnosis, date of discharge, HBPC pharmacist name, PCP notification on the discharge summary, and 30-day readmission rates. Unplanned return to the hospital within 30 days, which was counted as a readmission, was defined as any admission for acute clinical events that required urgent hospital management.10
Next, we identified specific pharmacist interventions, including medication reconciliation completed by an HBPC pharmacist postdischarge; mean time to contact patients postdischarge; correct medications and supplies on discharge; incorrect dose; incorrect medication frequency or route of administration; therapeutic duplications; discontinuation of medications; additional drug therapy recommendations; laboratory test recommendations; maintenance medications not restarted or omitted; new medication education; and medication or formulation changes.
In the third step, we reviewed discharge summaries and clinical pharmacy notes to collect pharmacist intervention data. These data were analyzed to develop a standardized discharge process. Descriptive statistics were used to represent the results of the study.
Results
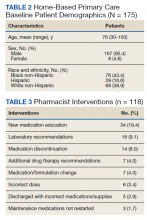
Medication reconciliation was completed postdischarge by an HBPC pharmacist in 118 of 175 study patients (67.4%). The mean age of patients was 76 years, about 95% were male (Table 2). There was a wide variety of admission diagnoses but sepsis, chronic obstructive pulmonary disease, and chronic kidney disease were most common. The PCP was notified on the discharge note for 68 (38.9%) patients. The mean time for HBPC pharmacists to contact patients postdischarge was about 3 days, which was much less than the 14 days allowed in the streamlined discharge process.
Pharmacists made the following interventions during medication reconciliation: New medication education was provided for 34 (19.4%) patients and was the largest intervention completed by HBPC pharmacists. Laboratory tests were recommended for 16 (9.1%) patients, medications were discontinued in 14 (8.0%) patients, and additional drug therapy recommendations were made for 7 (4.0%) patients. Medication or formulation changes were completed in 7 (4.0%) patients, incorrect doses were identified in 6 (3.4%) patients, 5 (2.9%) patients were not discharged with the correct medications or supplies, maintenance medications were not restarted in 3 (1.7%) patients, and there were no therapeutic duplications identified. In total, there were 92 (77.9%) patients with interventions compared with the 118 medication reconciliations completed (Table 3).
Process Improvement
As this was a new streamlined discharge process, it was important to assess the progress of the pharmacist role over time. We evaluated the number of medication reconciliations completed by quarter to determine whether more interventions were completed as the streamlined discharge process was being fully implemented. In FY 2020 Q1, medication reconciliation was completed by an HBPC pharmacist at a rate of 35%, and in FY 2020 Q2, at a rate of 65%.
In addition to assessing interventions completed by an HBPC pharmacist, we noted how many medication reconciliations were completed by an inpatient pharmacist as this may have impacted the results of this study. Of the 175 patients in this study, 49 (28%) received a medication reconciliation by an inpatient clinical pharmacy specialist before discharge. Last, when reviewing the readmissions data for the study period, it was evident that the streamlined discharge process was improving. In FY 2020 Q1, the readmissions rate prior to HBPC enrollment was 30% and decreased to 15% after and in FY 2020 Q2 was 31% before and decreased to 13% after HBPC enrollment. Before the study period in FY 2019 Q4, the readmissions rate after HBPC enrollment was 19%. Therefore, the readmissions rate decreased from 19% before the study period to 13% by the end of the study period.
Discussion
A comparison of the readmissions data from FYs 2019, 2020, and 2021 revealed that the newly implemented discharge process at MEDVAMC had been more effective.
There were 92 interventions made during the study period, which is about 78% of all medication reconciliations completed. Medication doses were changed based on patients’ renal function. Additional laboratory tests were recommended after discharge to ensure safety of therapy. Medications were discontinued if inappropriate or if patients were no longer on them to simplify their medication list and limit polypharmacy. New medication education was provided, including drug name, dose, route of administration, time of administration, frequency, indication, mechanism of action, adverse effect profile, monitoring parameters, and more. The HBPC pharmacists were able to make suitable interventions in a timely fashion as the average time to contact patients postdischarge was 3 days.
Areas for Improvement
The PCP was notified on the discharge note only in 68 (38.9%) patients. This could lead to gaps in care if other mechanisms are not in place to notify the PCP of the patient’s discharge. For this reason, it is imperative not only to implement a streamlined discharge process, but to review it and determine methods for continued improvement.9 The streamlined discharge process implemented by the HBPC team highlights when each team member should contact the patient postdischarge. However, it may be beneficial for each team member to have a list of vital information that should be communicated to the patient postdischarge and to other HCPs. For pharmacists, a standardized discharge note template may aid in the consistency of the medication reconciliation process postdischarge and may also increase interventions from pharmacists. For example, only some HBPC pharmacists inserted a new medication template in their discharge follow-up note. In addition, 23 (13.1%) patients were unreachable, and although a complete medication reconciliation was not feasible, a standardized note to review inpatient and outpatient medications along with the discharge plan may still serve as an asset for HCPs.
As the HBPC team continues to improve the discharge process, it is also important to highlight roles of the inpatient team who may assist with a smoother transition. For example, discharge summaries should be clear, complete, and concise, incorporating key elements from the hospital visit. Methods of communication on discharge should be efficient and understood by both inpatient and outpatient teams. Patients’ health literacy status should be considered when providing discharge instructions. Finally, patients should have a clear understanding of who is included in their primary care team should any questions arise. The potential interventions for HCPs highlighted in this study are critical for preventing adverse outcomes, improving patients’ quality of life, and decreasing hospital readmissions. However, implementing the streamlined discharge process was only step 1. Areas of improvement still exist to provide exceptional patient care.
Our goal is to increase pharmacist-led medication reconciliation after discharge to ≥ 80%. This will be assessed monthly after providing education to the HBPC team regarding the study results. The second goal is to maintain hospital readmission rates to ≤ 10%, which will be assessed with each quarterly review.
Strengths and Limitations
This study was one of the first to evaluate the impact of pharmacist intervention on improving patient outcomes in HBPC veterans. Additionally, only 1 investigator conducted the data collection, which decreased the opportunity for errors.
A notable limitation of this study is that the discharge processes may not be able to be duplicated in other HBPC settings due to variability in programs. Additionally, as this was a new discharge process, there were a few aspects that needed to be worked out in the beginning as it was established. Furthermore, this study did not clarify whether a medication reconciliation was conducted by a physician or nurse after discharge; therefore, this study cannot conclude that the medication interventions were solely attributed to pharmacists. Also this study did not assess readmissions for recurrent events only, which may have impacted the results in a different way from the current results that assessed readmission rates for any hospitalization. Other limitations include the retrospective study design at a single center.
Conclusions
This study outlines several opportunities for interventions to improve patient outcomes and aid in decreasing hospital readmission rates. Using the results from this study, education has been provided for the HBPC Service and its readmission committee. Additionally, the safety concerns identified have been addressed with inpatient and outpatient pharmacy leadership to improve the practices in both settings, prevent delays in patient care, and avoid future adverse outcomes. This project highlights the advantages of having pharmacists involved in transitions of care and demonstrates the benefit of HBPC pharmacists’ role in the streamlined discharge process. This project will be reviewed biannually to further improve the discharge process and quality of care for our veterans.
1. Coleman EA, Chugh A, Williams MV, et al. Understanding and execution of discharge instructions. Am J Med Qual. 2013;28(5):383-391. doi:10.1177/1062860612472931
2. Hume AL, Kirwin J, Bieber HL, et al. Improving care transitions: current practice and future opportunities for pharmacists. Pharmacotherapy. 2012;32(11):e326-e337. doi:10.1002/phar.1215
3. Milfred-LaForest SK, Gee JA, Pugacz AM, et al. Heart failure transitions of care: a pharmacist-led post discharge pilot experience. Prog Cardiovasc Dis. 2017;60(2):249-258. doi:10.1016/j.pcad.2017.08.005
4. Naylor M, Keating SA. Transitional care: moving patients from one care setting to another. Am J Nurs. 2008;108(suppl 9):58-63. doi:10.1097/01.NAJ.0000336420.34946.3a
5. Rennke S, Nguyen OK, Shoeb MH, Magan Y, Wachter RM, Ranji SR. Hospital-initiated transitional care interventions as a patient safety strategy. Ann Intern Med. 2013;158(5, pt 2):433-440. doi:10.7326/0003-4819-158-5-201303051-00011
6. Moore C, McGinn T, Halm E. Tying up loose ends: discharging patients with unresolved medical issues. Arch Intern Med. 2007;167:1305-1311. doi:10.1001/archinte.167.12.1305
7. Fosnight S, King P, Ewald J, et al. Effects of pharmacy interventions at transitions of care on patient outcomes. Am J Health Syst Pharm. 2020;77(12):943-949. doi:10.1093/ajhp/zxaa081
8. Shull MT, Braitman LE, Stites SD, DeLuca A, Hauser D. Effects of a pharmacist-driven intervention program on hospital readmissions. Am J Health Syst Pharm. 2018;75(9):e221-e230. doi:10.2146/ajhp170287
9. US Department of Health and Human Services, Agency for Healthcare Research and Quality. Plan-Do-Study-Act (PDSA) cycle. February 2015. Accessed June 2, 2022. https://www.ahrq.gov/health-literacy/improve/precautions/tool2b.html10. Horwitz L, Partovian C, Lin Z, et al. Yale New Haven Health Services Corporation/Center for Outcomes Research & Evaluation. Hospital-wide (all-condition) 30-day risk-standardized readmission measure. Updated August 20 2011. Accessed June 2, 2022. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/mms/downloads/mmshospital-wideall-conditionreadmissionrate.pdf
Following hospital discharge, patients are often in a vulnerable state due to new medical diagnoses, changes in medications, lack of understanding, and concerns for medical costs. In addition, the discharge process is complex and encompasses decisions regarding the postdischarge site of care, conveying patient instructions, and obtaining supplies and medications. There are several disciplines involved in the transitions of care process that are all essential for ensuring a successful transition and reducing the risk of hospital readmissions. Pharmacists play an integral role in the process.
When pharmacists are provided the opportunity to make therapeutic interventions, medication errors and hospital readmissions decrease and quality of life improves.1 Studies have shown that many older patients return home from the hospital with a limited understanding of their discharge instructions and oftentimes are unable to recall their discharge diagnoses and treatment plan, leaving opportunities for error when patients transition from one level of care to another.2,3 Additionally, high-quality transitional care is especially important for older adults with multiple comorbidities and complex therapeutic regimens as well as for their families and caregivers.4 To prevent hospital readmissions, pharmacists and other health care professionals (HCPs) should work diligently to prevent gaps in care as patients transition between settings. Common factors that lead to increased readmissions include premature discharge, inadequate follow-up, therapeutic errors, and medication-related problems. Furthermore, unintended hospital readmissions are common within the first 30 days following hospital discharge and lead to increased health care costs.2 For these reasons, many health care institutions have developed comprehensive models to improve the discharge process, decrease hospital readmissions, and reduce incidence of adverse events in general medical patients and high-risk populations.5
A study evaluating 693 hospital discharges found that 27.6% of patients were recommended for outpatient workups; however only 9% were actually completed.6 Due to lack of communication regarding discharge summaries, primary care practitioners (PCPs) were unaware of the need for outpatient workups; thus, these patients were lost to follow-up, and appropriate care was not received. Future studies should focus on interventions to improve the quality and dissemination of discharge information to PCPs.6 Fosnight and colleagues assessed a new transitions process focusing on the role of pharmacists. They evaluated medication reconciliations performed and discussed medication adherence barriers, medication recommendations, and time spent performing the interventions.7 After patients received a pharmacy intervention, Fosnight and colleagues reported that readmission rates decreased from 21.0% to 15.3% and mean length of stay decreased from 5.3 to 4.4 days. They also observed greater improvements in patients who received the full pharmacy intervention vs those receiving only parts of the intervention. This study concluded that adding a comprehensive pharmacy intervention to transitions of care resulted in an average of nearly 10 medication recommendations per patient, improved length of stay, and reduced readmission rates. After a review of similar studies, we concluded that a comprehensive discharge model is imperative to improve patient outcomes, along with HCP monitoring of the process to ensure appropriate follow-up.8
At Michael E. DeBakey Veteran Affairs Medical Center (MEDVAMC) in Houston, Texas, 30-day readmissions data were reviewed for veterans 6 months before and 12 months after enrollment in the Home-Based Primary Care (HBPC) service. HBPC is an in-home health care service provided to home-bound veterans with complex health care needs or when routine clinic-based care is not feasible. HBPC programs may differ among various US Department of Veterans Affairs (VA) medical centers. Currently, there are 9 HBPC teams at MEDVAMC and nearly 540 veterans are enrolled in the program. HBPC teams typically consist of PCPs, pharmacists, nurses, psychologists, occupational/physical therapists, social workers, medical support assistants, and dietitians.
Readmissions data are reviewed quarterly by fiscal year (FY) (Table 1). In FY 2019 quarter (Q) 2, the readmission rate before HBPC enrollment was 31% and decreased to 20% after enrollment. In FY 2019 Q3, the readmission rate was 29% before enrollment and decreased to 16% afterward. In FY 2019 Q4, the readmission rate before HBPC enrollment was 28% and decreased to 19% afterward. Although the readmission rates appeared to be decreasing overall, improvements were needed to decrease these rates further and to ensure readmissions were not rising as there was a slight increase in Q4. After reviewing these data, the HBPC service implemented a streamlined hospital discharge process to lower readmission rates and improve patient outcomes.
HBPC at MEDVAMC incorporates a team-based approach and the new streamlined discharge process implemented in 2019 highlights the role of each team member (Figure). Medical support assistants send daily emails of hospital discharges occurring in the last 7 days. Registered nurses are responsible for postdischarge calls within 2 days and home visits within 5 days. Pharmacists perform medication reconciliation within 14 days of discharge, review and/or educate on new medications, and change medications. The PCP is responsible for posthospital calls within 2 days and conducts a home visit within 5 days. Because HBPC programs vary among VA medical centers, the streamlined discharge process discussed may be applicable only to MEDVAMC. The primary objective of this quality improvement project was to identify specific pharmacist interventions to improve the HBPC discharge process and improve hospital readmission rates.
Methods
We conducted a Plan-Do-Study-Act quality improvement project. The first step was to conduct a review of veterans enrolled in HBPC at MEDVAMC.9 Patients included were enrolled in HBPC at MEDVAMC from October 2019 to March 2020 (FY 2020 Q1 and Q2). The Computerized Patient Record System was used to access the patients’ electronic health records. Patient information collected included race, age, sex, admission diagnosis, date of discharge, HBPC pharmacist name, PCP notification on the discharge summary, and 30-day readmission rates. Unplanned return to the hospital within 30 days, which was counted as a readmission, was defined as any admission for acute clinical events that required urgent hospital management.10
Next, we identified specific pharmacist interventions, including medication reconciliation completed by an HBPC pharmacist postdischarge; mean time to contact patients postdischarge; correct medications and supplies on discharge; incorrect dose; incorrect medication frequency or route of administration; therapeutic duplications; discontinuation of medications; additional drug therapy recommendations; laboratory test recommendations; maintenance medications not restarted or omitted; new medication education; and medication or formulation changes.
In the third step, we reviewed discharge summaries and clinical pharmacy notes to collect pharmacist intervention data. These data were analyzed to develop a standardized discharge process. Descriptive statistics were used to represent the results of the study.
Results
Medication reconciliation was completed postdischarge by an HBPC pharmacist in 118 of 175 study patients (67.4%). The mean age of patients was 76 years, about 95% were male (Table 2). There was a wide variety of admission diagnoses but sepsis, chronic obstructive pulmonary disease, and chronic kidney disease were most common. The PCP was notified on the discharge note for 68 (38.9%) patients. The mean time for HBPC pharmacists to contact patients postdischarge was about 3 days, which was much less than the 14 days allowed in the streamlined discharge process.
Pharmacists made the following interventions during medication reconciliation: New medication education was provided for 34 (19.4%) patients and was the largest intervention completed by HBPC pharmacists. Laboratory tests were recommended for 16 (9.1%) patients, medications were discontinued in 14 (8.0%) patients, and additional drug therapy recommendations were made for 7 (4.0%) patients. Medication or formulation changes were completed in 7 (4.0%) patients, incorrect doses were identified in 6 (3.4%) patients, 5 (2.9%) patients were not discharged with the correct medications or supplies, maintenance medications were not restarted in 3 (1.7%) patients, and there were no therapeutic duplications identified. In total, there were 92 (77.9%) patients with interventions compared with the 118 medication reconciliations completed (Table 3).
Process Improvement
As this was a new streamlined discharge process, it was important to assess the progress of the pharmacist role over time. We evaluated the number of medication reconciliations completed by quarter to determine whether more interventions were completed as the streamlined discharge process was being fully implemented. In FY 2020 Q1, medication reconciliation was completed by an HBPC pharmacist at a rate of 35%, and in FY 2020 Q2, at a rate of 65%.
In addition to assessing interventions completed by an HBPC pharmacist, we noted how many medication reconciliations were completed by an inpatient pharmacist as this may have impacted the results of this study. Of the 175 patients in this study, 49 (28%) received a medication reconciliation by an inpatient clinical pharmacy specialist before discharge. Last, when reviewing the readmissions data for the study period, it was evident that the streamlined discharge process was improving. In FY 2020 Q1, the readmissions rate prior to HBPC enrollment was 30% and decreased to 15% after and in FY 2020 Q2 was 31% before and decreased to 13% after HBPC enrollment. Before the study period in FY 2019 Q4, the readmissions rate after HBPC enrollment was 19%. Therefore, the readmissions rate decreased from 19% before the study period to 13% by the end of the study period.
Discussion
A comparison of the readmissions data from FYs 2019, 2020, and 2021 revealed that the newly implemented discharge process at MEDVAMC had been more effective.
There were 92 interventions made during the study period, which is about 78% of all medication reconciliations completed. Medication doses were changed based on patients’ renal function. Additional laboratory tests were recommended after discharge to ensure safety of therapy. Medications were discontinued if inappropriate or if patients were no longer on them to simplify their medication list and limit polypharmacy. New medication education was provided, including drug name, dose, route of administration, time of administration, frequency, indication, mechanism of action, adverse effect profile, monitoring parameters, and more. The HBPC pharmacists were able to make suitable interventions in a timely fashion as the average time to contact patients postdischarge was 3 days.
Areas for Improvement
The PCP was notified on the discharge note only in 68 (38.9%) patients. This could lead to gaps in care if other mechanisms are not in place to notify the PCP of the patient’s discharge. For this reason, it is imperative not only to implement a streamlined discharge process, but to review it and determine methods for continued improvement.9 The streamlined discharge process implemented by the HBPC team highlights when each team member should contact the patient postdischarge. However, it may be beneficial for each team member to have a list of vital information that should be communicated to the patient postdischarge and to other HCPs. For pharmacists, a standardized discharge note template may aid in the consistency of the medication reconciliation process postdischarge and may also increase interventions from pharmacists. For example, only some HBPC pharmacists inserted a new medication template in their discharge follow-up note. In addition, 23 (13.1%) patients were unreachable, and although a complete medication reconciliation was not feasible, a standardized note to review inpatient and outpatient medications along with the discharge plan may still serve as an asset for HCPs.
As the HBPC team continues to improve the discharge process, it is also important to highlight roles of the inpatient team who may assist with a smoother transition. For example, discharge summaries should be clear, complete, and concise, incorporating key elements from the hospital visit. Methods of communication on discharge should be efficient and understood by both inpatient and outpatient teams. Patients’ health literacy status should be considered when providing discharge instructions. Finally, patients should have a clear understanding of who is included in their primary care team should any questions arise. The potential interventions for HCPs highlighted in this study are critical for preventing adverse outcomes, improving patients’ quality of life, and decreasing hospital readmissions. However, implementing the streamlined discharge process was only step 1. Areas of improvement still exist to provide exceptional patient care.
Our goal is to increase pharmacist-led medication reconciliation after discharge to ≥ 80%. This will be assessed monthly after providing education to the HBPC team regarding the study results. The second goal is to maintain hospital readmission rates to ≤ 10%, which will be assessed with each quarterly review.
Strengths and Limitations
This study was one of the first to evaluate the impact of pharmacist intervention on improving patient outcomes in HBPC veterans. Additionally, only 1 investigator conducted the data collection, which decreased the opportunity for errors.
A notable limitation of this study is that the discharge processes may not be able to be duplicated in other HBPC settings due to variability in programs. Additionally, as this was a new discharge process, there were a few aspects that needed to be worked out in the beginning as it was established. Furthermore, this study did not clarify whether a medication reconciliation was conducted by a physician or nurse after discharge; therefore, this study cannot conclude that the medication interventions were solely attributed to pharmacists. Also this study did not assess readmissions for recurrent events only, which may have impacted the results in a different way from the current results that assessed readmission rates for any hospitalization. Other limitations include the retrospective study design at a single center.
Conclusions
This study outlines several opportunities for interventions to improve patient outcomes and aid in decreasing hospital readmission rates. Using the results from this study, education has been provided for the HBPC Service and its readmission committee. Additionally, the safety concerns identified have been addressed with inpatient and outpatient pharmacy leadership to improve the practices in both settings, prevent delays in patient care, and avoid future adverse outcomes. This project highlights the advantages of having pharmacists involved in transitions of care and demonstrates the benefit of HBPC pharmacists’ role in the streamlined discharge process. This project will be reviewed biannually to further improve the discharge process and quality of care for our veterans.
Following hospital discharge, patients are often in a vulnerable state due to new medical diagnoses, changes in medications, lack of understanding, and concerns for medical costs. In addition, the discharge process is complex and encompasses decisions regarding the postdischarge site of care, conveying patient instructions, and obtaining supplies and medications. There are several disciplines involved in the transitions of care process that are all essential for ensuring a successful transition and reducing the risk of hospital readmissions. Pharmacists play an integral role in the process.
When pharmacists are provided the opportunity to make therapeutic interventions, medication errors and hospital readmissions decrease and quality of life improves.1 Studies have shown that many older patients return home from the hospital with a limited understanding of their discharge instructions and oftentimes are unable to recall their discharge diagnoses and treatment plan, leaving opportunities for error when patients transition from one level of care to another.2,3 Additionally, high-quality transitional care is especially important for older adults with multiple comorbidities and complex therapeutic regimens as well as for their families and caregivers.4 To prevent hospital readmissions, pharmacists and other health care professionals (HCPs) should work diligently to prevent gaps in care as patients transition between settings. Common factors that lead to increased readmissions include premature discharge, inadequate follow-up, therapeutic errors, and medication-related problems. Furthermore, unintended hospital readmissions are common within the first 30 days following hospital discharge and lead to increased health care costs.2 For these reasons, many health care institutions have developed comprehensive models to improve the discharge process, decrease hospital readmissions, and reduce incidence of adverse events in general medical patients and high-risk populations.5
A study evaluating 693 hospital discharges found that 27.6% of patients were recommended for outpatient workups; however only 9% were actually completed.6 Due to lack of communication regarding discharge summaries, primary care practitioners (PCPs) were unaware of the need for outpatient workups; thus, these patients were lost to follow-up, and appropriate care was not received. Future studies should focus on interventions to improve the quality and dissemination of discharge information to PCPs.6 Fosnight and colleagues assessed a new transitions process focusing on the role of pharmacists. They evaluated medication reconciliations performed and discussed medication adherence barriers, medication recommendations, and time spent performing the interventions.7 After patients received a pharmacy intervention, Fosnight and colleagues reported that readmission rates decreased from 21.0% to 15.3% and mean length of stay decreased from 5.3 to 4.4 days. They also observed greater improvements in patients who received the full pharmacy intervention vs those receiving only parts of the intervention. This study concluded that adding a comprehensive pharmacy intervention to transitions of care resulted in an average of nearly 10 medication recommendations per patient, improved length of stay, and reduced readmission rates. After a review of similar studies, we concluded that a comprehensive discharge model is imperative to improve patient outcomes, along with HCP monitoring of the process to ensure appropriate follow-up.8
At Michael E. DeBakey Veteran Affairs Medical Center (MEDVAMC) in Houston, Texas, 30-day readmissions data were reviewed for veterans 6 months before and 12 months after enrollment in the Home-Based Primary Care (HBPC) service. HBPC is an in-home health care service provided to home-bound veterans with complex health care needs or when routine clinic-based care is not feasible. HBPC programs may differ among various US Department of Veterans Affairs (VA) medical centers. Currently, there are 9 HBPC teams at MEDVAMC and nearly 540 veterans are enrolled in the program. HBPC teams typically consist of PCPs, pharmacists, nurses, psychologists, occupational/physical therapists, social workers, medical support assistants, and dietitians.
Readmissions data are reviewed quarterly by fiscal year (FY) (Table 1). In FY 2019 quarter (Q) 2, the readmission rate before HBPC enrollment was 31% and decreased to 20% after enrollment. In FY 2019 Q3, the readmission rate was 29% before enrollment and decreased to 16% afterward. In FY 2019 Q4, the readmission rate before HBPC enrollment was 28% and decreased to 19% afterward. Although the readmission rates appeared to be decreasing overall, improvements were needed to decrease these rates further and to ensure readmissions were not rising as there was a slight increase in Q4. After reviewing these data, the HBPC service implemented a streamlined hospital discharge process to lower readmission rates and improve patient outcomes.
HBPC at MEDVAMC incorporates a team-based approach and the new streamlined discharge process implemented in 2019 highlights the role of each team member (Figure). Medical support assistants send daily emails of hospital discharges occurring in the last 7 days. Registered nurses are responsible for postdischarge calls within 2 days and home visits within 5 days. Pharmacists perform medication reconciliation within 14 days of discharge, review and/or educate on new medications, and change medications. The PCP is responsible for posthospital calls within 2 days and conducts a home visit within 5 days. Because HBPC programs vary among VA medical centers, the streamlined discharge process discussed may be applicable only to MEDVAMC. The primary objective of this quality improvement project was to identify specific pharmacist interventions to improve the HBPC discharge process and improve hospital readmission rates.
Methods
We conducted a Plan-Do-Study-Act quality improvement project. The first step was to conduct a review of veterans enrolled in HBPC at MEDVAMC.9 Patients included were enrolled in HBPC at MEDVAMC from October 2019 to March 2020 (FY 2020 Q1 and Q2). The Computerized Patient Record System was used to access the patients’ electronic health records. Patient information collected included race, age, sex, admission diagnosis, date of discharge, HBPC pharmacist name, PCP notification on the discharge summary, and 30-day readmission rates. Unplanned return to the hospital within 30 days, which was counted as a readmission, was defined as any admission for acute clinical events that required urgent hospital management.10
Next, we identified specific pharmacist interventions, including medication reconciliation completed by an HBPC pharmacist postdischarge; mean time to contact patients postdischarge; correct medications and supplies on discharge; incorrect dose; incorrect medication frequency or route of administration; therapeutic duplications; discontinuation of medications; additional drug therapy recommendations; laboratory test recommendations; maintenance medications not restarted or omitted; new medication education; and medication or formulation changes.
In the third step, we reviewed discharge summaries and clinical pharmacy notes to collect pharmacist intervention data. These data were analyzed to develop a standardized discharge process. Descriptive statistics were used to represent the results of the study.
Results
Medication reconciliation was completed postdischarge by an HBPC pharmacist in 118 of 175 study patients (67.4%). The mean age of patients was 76 years, about 95% were male (Table 2). There was a wide variety of admission diagnoses but sepsis, chronic obstructive pulmonary disease, and chronic kidney disease were most common. The PCP was notified on the discharge note for 68 (38.9%) patients. The mean time for HBPC pharmacists to contact patients postdischarge was about 3 days, which was much less than the 14 days allowed in the streamlined discharge process.
Pharmacists made the following interventions during medication reconciliation: New medication education was provided for 34 (19.4%) patients and was the largest intervention completed by HBPC pharmacists. Laboratory tests were recommended for 16 (9.1%) patients, medications were discontinued in 14 (8.0%) patients, and additional drug therapy recommendations were made for 7 (4.0%) patients. Medication or formulation changes were completed in 7 (4.0%) patients, incorrect doses were identified in 6 (3.4%) patients, 5 (2.9%) patients were not discharged with the correct medications or supplies, maintenance medications were not restarted in 3 (1.7%) patients, and there were no therapeutic duplications identified. In total, there were 92 (77.9%) patients with interventions compared with the 118 medication reconciliations completed (Table 3).
Process Improvement
As this was a new streamlined discharge process, it was important to assess the progress of the pharmacist role over time. We evaluated the number of medication reconciliations completed by quarter to determine whether more interventions were completed as the streamlined discharge process was being fully implemented. In FY 2020 Q1, medication reconciliation was completed by an HBPC pharmacist at a rate of 35%, and in FY 2020 Q2, at a rate of 65%.
In addition to assessing interventions completed by an HBPC pharmacist, we noted how many medication reconciliations were completed by an inpatient pharmacist as this may have impacted the results of this study. Of the 175 patients in this study, 49 (28%) received a medication reconciliation by an inpatient clinical pharmacy specialist before discharge. Last, when reviewing the readmissions data for the study period, it was evident that the streamlined discharge process was improving. In FY 2020 Q1, the readmissions rate prior to HBPC enrollment was 30% and decreased to 15% after and in FY 2020 Q2 was 31% before and decreased to 13% after HBPC enrollment. Before the study period in FY 2019 Q4, the readmissions rate after HBPC enrollment was 19%. Therefore, the readmissions rate decreased from 19% before the study period to 13% by the end of the study period.
Discussion
A comparison of the readmissions data from FYs 2019, 2020, and 2021 revealed that the newly implemented discharge process at MEDVAMC had been more effective.
There were 92 interventions made during the study period, which is about 78% of all medication reconciliations completed. Medication doses were changed based on patients’ renal function. Additional laboratory tests were recommended after discharge to ensure safety of therapy. Medications were discontinued if inappropriate or if patients were no longer on them to simplify their medication list and limit polypharmacy. New medication education was provided, including drug name, dose, route of administration, time of administration, frequency, indication, mechanism of action, adverse effect profile, monitoring parameters, and more. The HBPC pharmacists were able to make suitable interventions in a timely fashion as the average time to contact patients postdischarge was 3 days.
Areas for Improvement
The PCP was notified on the discharge note only in 68 (38.9%) patients. This could lead to gaps in care if other mechanisms are not in place to notify the PCP of the patient’s discharge. For this reason, it is imperative not only to implement a streamlined discharge process, but to review it and determine methods for continued improvement.9 The streamlined discharge process implemented by the HBPC team highlights when each team member should contact the patient postdischarge. However, it may be beneficial for each team member to have a list of vital information that should be communicated to the patient postdischarge and to other HCPs. For pharmacists, a standardized discharge note template may aid in the consistency of the medication reconciliation process postdischarge and may also increase interventions from pharmacists. For example, only some HBPC pharmacists inserted a new medication template in their discharge follow-up note. In addition, 23 (13.1%) patients were unreachable, and although a complete medication reconciliation was not feasible, a standardized note to review inpatient and outpatient medications along with the discharge plan may still serve as an asset for HCPs.
As the HBPC team continues to improve the discharge process, it is also important to highlight roles of the inpatient team who may assist with a smoother transition. For example, discharge summaries should be clear, complete, and concise, incorporating key elements from the hospital visit. Methods of communication on discharge should be efficient and understood by both inpatient and outpatient teams. Patients’ health literacy status should be considered when providing discharge instructions. Finally, patients should have a clear understanding of who is included in their primary care team should any questions arise. The potential interventions for HCPs highlighted in this study are critical for preventing adverse outcomes, improving patients’ quality of life, and decreasing hospital readmissions. However, implementing the streamlined discharge process was only step 1. Areas of improvement still exist to provide exceptional patient care.
Our goal is to increase pharmacist-led medication reconciliation after discharge to ≥ 80%. This will be assessed monthly after providing education to the HBPC team regarding the study results. The second goal is to maintain hospital readmission rates to ≤ 10%, which will be assessed with each quarterly review.
Strengths and Limitations
This study was one of the first to evaluate the impact of pharmacist intervention on improving patient outcomes in HBPC veterans. Additionally, only 1 investigator conducted the data collection, which decreased the opportunity for errors.
A notable limitation of this study is that the discharge processes may not be able to be duplicated in other HBPC settings due to variability in programs. Additionally, as this was a new discharge process, there were a few aspects that needed to be worked out in the beginning as it was established. Furthermore, this study did not clarify whether a medication reconciliation was conducted by a physician or nurse after discharge; therefore, this study cannot conclude that the medication interventions were solely attributed to pharmacists. Also this study did not assess readmissions for recurrent events only, which may have impacted the results in a different way from the current results that assessed readmission rates for any hospitalization. Other limitations include the retrospective study design at a single center.
Conclusions
This study outlines several opportunities for interventions to improve patient outcomes and aid in decreasing hospital readmission rates. Using the results from this study, education has been provided for the HBPC Service and its readmission committee. Additionally, the safety concerns identified have been addressed with inpatient and outpatient pharmacy leadership to improve the practices in both settings, prevent delays in patient care, and avoid future adverse outcomes. This project highlights the advantages of having pharmacists involved in transitions of care and demonstrates the benefit of HBPC pharmacists’ role in the streamlined discharge process. This project will be reviewed biannually to further improve the discharge process and quality of care for our veterans.
1. Coleman EA, Chugh A, Williams MV, et al. Understanding and execution of discharge instructions. Am J Med Qual. 2013;28(5):383-391. doi:10.1177/1062860612472931
2. Hume AL, Kirwin J, Bieber HL, et al. Improving care transitions: current practice and future opportunities for pharmacists. Pharmacotherapy. 2012;32(11):e326-e337. doi:10.1002/phar.1215
3. Milfred-LaForest SK, Gee JA, Pugacz AM, et al. Heart failure transitions of care: a pharmacist-led post discharge pilot experience. Prog Cardiovasc Dis. 2017;60(2):249-258. doi:10.1016/j.pcad.2017.08.005
4. Naylor M, Keating SA. Transitional care: moving patients from one care setting to another. Am J Nurs. 2008;108(suppl 9):58-63. doi:10.1097/01.NAJ.0000336420.34946.3a
5. Rennke S, Nguyen OK, Shoeb MH, Magan Y, Wachter RM, Ranji SR. Hospital-initiated transitional care interventions as a patient safety strategy. Ann Intern Med. 2013;158(5, pt 2):433-440. doi:10.7326/0003-4819-158-5-201303051-00011
6. Moore C, McGinn T, Halm E. Tying up loose ends: discharging patients with unresolved medical issues. Arch Intern Med. 2007;167:1305-1311. doi:10.1001/archinte.167.12.1305
7. Fosnight S, King P, Ewald J, et al. Effects of pharmacy interventions at transitions of care on patient outcomes. Am J Health Syst Pharm. 2020;77(12):943-949. doi:10.1093/ajhp/zxaa081
8. Shull MT, Braitman LE, Stites SD, DeLuca A, Hauser D. Effects of a pharmacist-driven intervention program on hospital readmissions. Am J Health Syst Pharm. 2018;75(9):e221-e230. doi:10.2146/ajhp170287
9. US Department of Health and Human Services, Agency for Healthcare Research and Quality. Plan-Do-Study-Act (PDSA) cycle. February 2015. Accessed June 2, 2022. https://www.ahrq.gov/health-literacy/improve/precautions/tool2b.html10. Horwitz L, Partovian C, Lin Z, et al. Yale New Haven Health Services Corporation/Center for Outcomes Research & Evaluation. Hospital-wide (all-condition) 30-day risk-standardized readmission measure. Updated August 20 2011. Accessed June 2, 2022. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/mms/downloads/mmshospital-wideall-conditionreadmissionrate.pdf
1. Coleman EA, Chugh A, Williams MV, et al. Understanding and execution of discharge instructions. Am J Med Qual. 2013;28(5):383-391. doi:10.1177/1062860612472931
2. Hume AL, Kirwin J, Bieber HL, et al. Improving care transitions: current practice and future opportunities for pharmacists. Pharmacotherapy. 2012;32(11):e326-e337. doi:10.1002/phar.1215
3. Milfred-LaForest SK, Gee JA, Pugacz AM, et al. Heart failure transitions of care: a pharmacist-led post discharge pilot experience. Prog Cardiovasc Dis. 2017;60(2):249-258. doi:10.1016/j.pcad.2017.08.005
4. Naylor M, Keating SA. Transitional care: moving patients from one care setting to another. Am J Nurs. 2008;108(suppl 9):58-63. doi:10.1097/01.NAJ.0000336420.34946.3a
5. Rennke S, Nguyen OK, Shoeb MH, Magan Y, Wachter RM, Ranji SR. Hospital-initiated transitional care interventions as a patient safety strategy. Ann Intern Med. 2013;158(5, pt 2):433-440. doi:10.7326/0003-4819-158-5-201303051-00011
6. Moore C, McGinn T, Halm E. Tying up loose ends: discharging patients with unresolved medical issues. Arch Intern Med. 2007;167:1305-1311. doi:10.1001/archinte.167.12.1305
7. Fosnight S, King P, Ewald J, et al. Effects of pharmacy interventions at transitions of care on patient outcomes. Am J Health Syst Pharm. 2020;77(12):943-949. doi:10.1093/ajhp/zxaa081
8. Shull MT, Braitman LE, Stites SD, DeLuca A, Hauser D. Effects of a pharmacist-driven intervention program on hospital readmissions. Am J Health Syst Pharm. 2018;75(9):e221-e230. doi:10.2146/ajhp170287
9. US Department of Health and Human Services, Agency for Healthcare Research and Quality. Plan-Do-Study-Act (PDSA) cycle. February 2015. Accessed June 2, 2022. https://www.ahrq.gov/health-literacy/improve/precautions/tool2b.html10. Horwitz L, Partovian C, Lin Z, et al. Yale New Haven Health Services Corporation/Center for Outcomes Research & Evaluation. Hospital-wide (all-condition) 30-day risk-standardized readmission measure. Updated August 20 2011. Accessed June 2, 2022. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/mms/downloads/mmshospital-wideall-conditionreadmissionrate.pdf
Registered Dietitian Nutritionists’ Role in Hospital in Home
Hospital in Home (HIH) is the delivery of acute care services in a patient’s home as an alternative to hospitalization.1 Compared with traditional inpatient care, HIH programs have been associated with reduced costs, as well as patient and caregiver satisfaction, diseasespecific outcomes, and mortality rates that were similar or improved compared with inpatient admissions.1-4
The US Department of Veterans Affairs (VA) Veterans Health Administration (VHA) and other hospital systems are increasingly adopting HIH models.2-4 At the time of this writing, there were 12 HIH programs in VHA (personal communication, D. Cooper, 2/28/2022). In addition to physicians and nurses, the interdisciplinary HIH team may include a pharmacist, social worker, and registered dietitian nutritionist (RDN).2,5 HIH programs have been shown to improve nutritional status as measured by the Mini Nutritional Assessment Score, but overall, there is a paucity of published information regarding the provision of nutrition care in HIH.6 The role of the RDN has varied within VHA. Some sites, such as the Sacramento VA Medical Center in California, include a distinct RDN position on the HIH team, whereas others, such as the Spark M. Matsunaga VA Medical Center in Honolulu, Hawaii, and the James A. Haley Veterans’ Hospital in Tampa, Florida, consult clinic RDNs.
Since HIH programs typically treat conditions for which diet is an inherent part of the treatment (eg, congestive heart failure [CHF]), there is a need to precisely define the role of the RDN within the HIH model.2,3,7 Drawing from my experience as an HIH RDN, I will describe how the inclusion of an RDN position within the HIH team is optimal for health care delivery and how HIH practitioners can best utilize RDN services.
RDN Role in HIH Team
Delegating nutrition services to an RDN enhances patient care by empowering HIH team members to function at the highest level of their scope of practice. RDNs have been recognized by physicians as the most qualified health care professionals to help patients with diet-related conditions, such as obesity, and physicians also have reported a desire for additional training in nutrition.8 Although home-health nurses have frequently performed nutrition assessments and interventions, survey results have indicated that many nurses do not feel confident in teaching complex nutritional information.9 In my experience, many HIH patients are nutritionally complex, with more than one condition requiring nutrition intervention. For example, patients may be admitted to HIH for management of CHF, but they may also have diabetes mellitus (DM), obesity, and low socioeconomic status. The HIH RDN can address the nutrition aspects of these conditions, freeing time for physicians and nurses to focus on their respective areas of expertise.9,10 Moreover, the RDN can also provide dietary education to the HIH team to increase their knowledge of nutritional topics and promote consistent messaging to patients.
Including an RDN on the HIH team enables patients to have comprehensive, personalized nutrition care. Rather than merely offering generalized nutrition education, RDNs are trained to provide medical nutrition therapy (MNT), which has been shown to improve health outcomes and be cost-effective for conditions such as type 2 DM, chronic kidney disease, hypertension, and obesity.10,11 In MNT, RDNs use the standardized 4-stepnutrition care process (NCP).12 The Table shows examples of how the NCP can be applied in HIH settings. Furthermore, in my experience, MNT from an RDN also contributes to patient satisfaction. Subjective observations from my team have indicated that patients often express more confidence in managing their diets by the time of HIH discharge.
RDNs can guide physicians and pharmacists in ordering oral nutrition supplements (ONS). Within the VHA, a “food first” approach is preferred to increase caloric intake, and patients must meet specific criteria for prescription of an ONS.13 Furthermore, ONS designed for specific medical conditions (eg, chronic kidney disease) are considered nonformulary and require an RDN evaluation.13 Including an RDN on the HIH team allows this evaluation process to begin early in the patient’s admission to the program and ensures that provision of ONS is clinically appropriate and cost-effective.
Care Coordination
HIH is highly interdisciplinary. Team members perform their respective roles and communicate with the team throughout the day. RDNs can help monitor patients and alert physicians for changes in blood glucose, gastrointestinal concerns, and weight. This is especially helpful for patients who do not have a planned nursing visit on the day of an RDN evaluation. The HIH RDN can also collaborate with other team members to address patient needs. For example, for patients with limited financial resources, the HIH RDN can provide nutrition education regarding cooking on a budget, and the HIH social worker can arrange free or low-cost meal services.
Tips
When hiring an HIH RDN, seek candidates with experience in inpatient, outpatient, and home care settings. As a hybrid of these 3 areas, the HIH RDN position requires a unique combination of acute care skills and health coaching. Additionally, in my experience, the HIH RDN interacts more frequently with the HIH team than other RDN colleagues, so it is important that candidates can work independently and take initiative. This type of position would not be suitable for entry-level RDNs.
Stagger HIH team visits to prevent overwhelming the patient and caregivers. Early in our program, my team quickly learned that patients and caregivers can feel overwhelmed with too many home visits upon admission to HIH. After seeing multiple HIH team members the same day, they were often too tired to focus well on diet education during my visit. Staggering visits (eg, completing the initial nutrition assessment 1 day to 1 week after the initial medical and pharmacy visits) has been an effective strategy to address this problem. Furthermore, some patients prefer that the initial RDN appointment is conducted by telephone, with an inperson reassessment the following week. In my experience, HIH workflow is dynamic by nature, so it is crucial to remain flexible and accommodate individual patient needs as much as possible.
Dietary behavior change is a long-term process, and restrictive hospital diets can be challenging to replicate at home. In a hospital setting, clinicians can order a specialized diet (eg, low sodium with fluid restriction for CHF patients), whereas efforts to implement these restrictions in the home setting can be cumbersome and negatively impact quality of life.7,14 Nevertheless, the effectiveness of medical treatment is compromised when patients do not adhere to dietary recommendations. Meal delivery services that offer specialized diets can be a useful resource for patients and caregivers who are unable to cook, and the HIH RDN can assist patients in ordering these services.
HIH patients may vary in terms of readiness to make dietary changes, and in addition to nutrition education, nutrition counseling is usually needed to effect behavior change. My team has found that consideration of the transtheoretical/ stages of change model can be a helpful approach. 15 The HIH RDN can tailor nutrition interventions to the patient’s stage of change. For example, for patients in the precontemplation stage, the HIH RDN would focus on providing information and addressing emotional aspects of dietary change. In contrast, for patients in the action stage of change, the HIH RDN might emphasize behavioral skill training and social support.15 Particularly for patients in the early stages of change, it may be unrealistic to expect full adoption of the recommended diet within the 30 days of the HIH program. However, by acknowledging the reality of the patient’s stage of change, the HIH RDN and team can then collaborate to support the patient in moving toward the next stage. Patients who are not ready for dietary behavior change during the 30 days of HIH may benefit from longer-term support, and the HIH RDN can arrange followup care with an outpatient RDN.
Conclusions
As the HIH model continues to be adopted across the VHA and other health care systems, it is crucial to consider the value and expertise of an RDN for guiding nutrition care in the HIH setting. The HIH RDN contributes to optimal health care delivery by leading nutritional aspects of patient care, offering personalized MNT, and coordinating and collaborating with team members to meet individual patient needs. An RDN can serve as a valuable resource for nutrition information and enhance the team’s overall services, with the potential to impact clinical outcomes and patient satisfaction.
1. Levine DM, Ouchi K, Blanchfield B, et al. Hospitallevel care at home for acutely ill adults: a randomized controlled trial. Ann Intern Med. 2020;172(2):77-85. doi:10.7326/M19-0600
2. Cai S, Grubbs A, Makineni R, Kinosian B, Phibbs CS, Intrator O. Evaluation of the Cincinnati Veterans Affairs medical center hospital-in-home program. J Am Geriatr Soc. 2018;66(7):1392-1398. doi:10.1111/jgs.15382
3. Cai S, Laurel PA, Makineni R, Marks ML. Evaluation of a hospital-in-home program implemented among veterans. Am J Manag Care. 2017;23(8):482-487.
4. Conley J, O’Brien CW, Leff BA, Bolen S, Zulman D. Alternative strategies to inpatient hospitalization for acute medical conditions: a systematic review. JAMA Intern Med. 2016;176(11):1693-1702. doi:10.1001/jamainternmed.2016.5974
5. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1144: Hospital in Home program, Appendix A, Hospital in Home program standards. January 19, 2021. Accessed May 5, 2022. https://www .va.gov/VHApublications/ViewPublication.asp?pub _ID=9157
6. Tibaldi V, Isaia G, Scarafiotti C, et al. Hospital at home for elderly patients with acute decompensation of chronic heart failure: a prospective randomized controlled trial. Arch Intern Med. 2009;169(17):1569-1575. doi:10.1001/archinternmed.2009.267
7. Abshire M, Xu J, Baptiste D, et al. Nutritional interventions in heart failure: a systematic review of the literature. J Card Fail. 2015;21(12):989-999. doi:10.1016/j.cardfail.2015.10.004
8. Bleich SN, Bennett WL, Gudzune KA, Cooper LA. National survey of US primary care physicians’ perspectives about causes of obesity and solutions to improve care. BMJ Open. 2012;2(6):e001871. Published 2012 Dec 20. doi:10.1136/bmjopen-2012-001871
9. Sousa AM. Benefits of dietitian home visits. J Am Diet Assoc. 1994;94(10):1149-1151. doi:10.1016/0002-8223(94)91136-3
10. Casas-Agustench P, Megías-Rangil I, Babio N. Economic benefit of dietetic-nutritional treatment in the multidisciplinary primary care team. Beneficio económico del tratamiento dietético-nutricional en el equipo multidisciplinario de atención primaria. Nutr Hosp. 2020;37(4):863-874. doi:10.20960/nh.03025
11. Lee J, Briggs Early K, Kovesdy CP, Lancaster K, Brown N, Steiber AL. The impact of RDNs on non-communicable diseases: proceedings from The State of Food and Nutrition Series Forum. J Acad Nutr Diet. 2022;122(1):166-174. doi:10.1016/j.jand.2021.02.021
12. Academy of Nutrition and Dietetics. Evidence analysis library, nutrition care process. Accessed May 5, 2022. https://www.andeal.org/ncp
13. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1438, clinical nutrition management and therapy. Appendix A, nutrition support therapy. September 19, 2019. Accessed January 20, 2022. https://www.va.gov/VHAPUBLICATIONS/ViewPublication .asp?pub_ID=8512
14. Vogelzang JL. Fifteen ways to enhance client outcomes by using your registered dietitian. Home Healthc Nurse. 2002;20(4):227-229. doi:10.1097/00004045-200204000-00005
15. Kristal AR, Glanz K, Curry SJ, Patterson RE. How can stages of change be best used in dietary interventions?. J Am Diet Assoc. 1999;99(6):679-684. doi:10.1016/S0002-8223(99)00165-0
Hospital in Home (HIH) is the delivery of acute care services in a patient’s home as an alternative to hospitalization.1 Compared with traditional inpatient care, HIH programs have been associated with reduced costs, as well as patient and caregiver satisfaction, diseasespecific outcomes, and mortality rates that were similar or improved compared with inpatient admissions.1-4
The US Department of Veterans Affairs (VA) Veterans Health Administration (VHA) and other hospital systems are increasingly adopting HIH models.2-4 At the time of this writing, there were 12 HIH programs in VHA (personal communication, D. Cooper, 2/28/2022). In addition to physicians and nurses, the interdisciplinary HIH team may include a pharmacist, social worker, and registered dietitian nutritionist (RDN).2,5 HIH programs have been shown to improve nutritional status as measured by the Mini Nutritional Assessment Score, but overall, there is a paucity of published information regarding the provision of nutrition care in HIH.6 The role of the RDN has varied within VHA. Some sites, such as the Sacramento VA Medical Center in California, include a distinct RDN position on the HIH team, whereas others, such as the Spark M. Matsunaga VA Medical Center in Honolulu, Hawaii, and the James A. Haley Veterans’ Hospital in Tampa, Florida, consult clinic RDNs.
Since HIH programs typically treat conditions for which diet is an inherent part of the treatment (eg, congestive heart failure [CHF]), there is a need to precisely define the role of the RDN within the HIH model.2,3,7 Drawing from my experience as an HIH RDN, I will describe how the inclusion of an RDN position within the HIH team is optimal for health care delivery and how HIH practitioners can best utilize RDN services.
RDN Role in HIH Team
Delegating nutrition services to an RDN enhances patient care by empowering HIH team members to function at the highest level of their scope of practice. RDNs have been recognized by physicians as the most qualified health care professionals to help patients with diet-related conditions, such as obesity, and physicians also have reported a desire for additional training in nutrition.8 Although home-health nurses have frequently performed nutrition assessments and interventions, survey results have indicated that many nurses do not feel confident in teaching complex nutritional information.9 In my experience, many HIH patients are nutritionally complex, with more than one condition requiring nutrition intervention. For example, patients may be admitted to HIH for management of CHF, but they may also have diabetes mellitus (DM), obesity, and low socioeconomic status. The HIH RDN can address the nutrition aspects of these conditions, freeing time for physicians and nurses to focus on their respective areas of expertise.9,10 Moreover, the RDN can also provide dietary education to the HIH team to increase their knowledge of nutritional topics and promote consistent messaging to patients.
Including an RDN on the HIH team enables patients to have comprehensive, personalized nutrition care. Rather than merely offering generalized nutrition education, RDNs are trained to provide medical nutrition therapy (MNT), which has been shown to improve health outcomes and be cost-effective for conditions such as type 2 DM, chronic kidney disease, hypertension, and obesity.10,11 In MNT, RDNs use the standardized 4-stepnutrition care process (NCP).12 The Table shows examples of how the NCP can be applied in HIH settings. Furthermore, in my experience, MNT from an RDN also contributes to patient satisfaction. Subjective observations from my team have indicated that patients often express more confidence in managing their diets by the time of HIH discharge.
RDNs can guide physicians and pharmacists in ordering oral nutrition supplements (ONS). Within the VHA, a “food first” approach is preferred to increase caloric intake, and patients must meet specific criteria for prescription of an ONS.13 Furthermore, ONS designed for specific medical conditions (eg, chronic kidney disease) are considered nonformulary and require an RDN evaluation.13 Including an RDN on the HIH team allows this evaluation process to begin early in the patient’s admission to the program and ensures that provision of ONS is clinically appropriate and cost-effective.
Care Coordination
HIH is highly interdisciplinary. Team members perform their respective roles and communicate with the team throughout the day. RDNs can help monitor patients and alert physicians for changes in blood glucose, gastrointestinal concerns, and weight. This is especially helpful for patients who do not have a planned nursing visit on the day of an RDN evaluation. The HIH RDN can also collaborate with other team members to address patient needs. For example, for patients with limited financial resources, the HIH RDN can provide nutrition education regarding cooking on a budget, and the HIH social worker can arrange free or low-cost meal services.
Tips
When hiring an HIH RDN, seek candidates with experience in inpatient, outpatient, and home care settings. As a hybrid of these 3 areas, the HIH RDN position requires a unique combination of acute care skills and health coaching. Additionally, in my experience, the HIH RDN interacts more frequently with the HIH team than other RDN colleagues, so it is important that candidates can work independently and take initiative. This type of position would not be suitable for entry-level RDNs.
Stagger HIH team visits to prevent overwhelming the patient and caregivers. Early in our program, my team quickly learned that patients and caregivers can feel overwhelmed with too many home visits upon admission to HIH. After seeing multiple HIH team members the same day, they were often too tired to focus well on diet education during my visit. Staggering visits (eg, completing the initial nutrition assessment 1 day to 1 week after the initial medical and pharmacy visits) has been an effective strategy to address this problem. Furthermore, some patients prefer that the initial RDN appointment is conducted by telephone, with an inperson reassessment the following week. In my experience, HIH workflow is dynamic by nature, so it is crucial to remain flexible and accommodate individual patient needs as much as possible.
Dietary behavior change is a long-term process, and restrictive hospital diets can be challenging to replicate at home. In a hospital setting, clinicians can order a specialized diet (eg, low sodium with fluid restriction for CHF patients), whereas efforts to implement these restrictions in the home setting can be cumbersome and negatively impact quality of life.7,14 Nevertheless, the effectiveness of medical treatment is compromised when patients do not adhere to dietary recommendations. Meal delivery services that offer specialized diets can be a useful resource for patients and caregivers who are unable to cook, and the HIH RDN can assist patients in ordering these services.
HIH patients may vary in terms of readiness to make dietary changes, and in addition to nutrition education, nutrition counseling is usually needed to effect behavior change. My team has found that consideration of the transtheoretical/ stages of change model can be a helpful approach. 15 The HIH RDN can tailor nutrition interventions to the patient’s stage of change. For example, for patients in the precontemplation stage, the HIH RDN would focus on providing information and addressing emotional aspects of dietary change. In contrast, for patients in the action stage of change, the HIH RDN might emphasize behavioral skill training and social support.15 Particularly for patients in the early stages of change, it may be unrealistic to expect full adoption of the recommended diet within the 30 days of the HIH program. However, by acknowledging the reality of the patient’s stage of change, the HIH RDN and team can then collaborate to support the patient in moving toward the next stage. Patients who are not ready for dietary behavior change during the 30 days of HIH may benefit from longer-term support, and the HIH RDN can arrange followup care with an outpatient RDN.
Conclusions
As the HIH model continues to be adopted across the VHA and other health care systems, it is crucial to consider the value and expertise of an RDN for guiding nutrition care in the HIH setting. The HIH RDN contributes to optimal health care delivery by leading nutritional aspects of patient care, offering personalized MNT, and coordinating and collaborating with team members to meet individual patient needs. An RDN can serve as a valuable resource for nutrition information and enhance the team’s overall services, with the potential to impact clinical outcomes and patient satisfaction.
Hospital in Home (HIH) is the delivery of acute care services in a patient’s home as an alternative to hospitalization.1 Compared with traditional inpatient care, HIH programs have been associated with reduced costs, as well as patient and caregiver satisfaction, diseasespecific outcomes, and mortality rates that were similar or improved compared with inpatient admissions.1-4
The US Department of Veterans Affairs (VA) Veterans Health Administration (VHA) and other hospital systems are increasingly adopting HIH models.2-4 At the time of this writing, there were 12 HIH programs in VHA (personal communication, D. Cooper, 2/28/2022). In addition to physicians and nurses, the interdisciplinary HIH team may include a pharmacist, social worker, and registered dietitian nutritionist (RDN).2,5 HIH programs have been shown to improve nutritional status as measured by the Mini Nutritional Assessment Score, but overall, there is a paucity of published information regarding the provision of nutrition care in HIH.6 The role of the RDN has varied within VHA. Some sites, such as the Sacramento VA Medical Center in California, include a distinct RDN position on the HIH team, whereas others, such as the Spark M. Matsunaga VA Medical Center in Honolulu, Hawaii, and the James A. Haley Veterans’ Hospital in Tampa, Florida, consult clinic RDNs.
Since HIH programs typically treat conditions for which diet is an inherent part of the treatment (eg, congestive heart failure [CHF]), there is a need to precisely define the role of the RDN within the HIH model.2,3,7 Drawing from my experience as an HIH RDN, I will describe how the inclusion of an RDN position within the HIH team is optimal for health care delivery and how HIH practitioners can best utilize RDN services.
RDN Role in HIH Team
Delegating nutrition services to an RDN enhances patient care by empowering HIH team members to function at the highest level of their scope of practice. RDNs have been recognized by physicians as the most qualified health care professionals to help patients with diet-related conditions, such as obesity, and physicians also have reported a desire for additional training in nutrition.8 Although home-health nurses have frequently performed nutrition assessments and interventions, survey results have indicated that many nurses do not feel confident in teaching complex nutritional information.9 In my experience, many HIH patients are nutritionally complex, with more than one condition requiring nutrition intervention. For example, patients may be admitted to HIH for management of CHF, but they may also have diabetes mellitus (DM), obesity, and low socioeconomic status. The HIH RDN can address the nutrition aspects of these conditions, freeing time for physicians and nurses to focus on their respective areas of expertise.9,10 Moreover, the RDN can also provide dietary education to the HIH team to increase their knowledge of nutritional topics and promote consistent messaging to patients.
Including an RDN on the HIH team enables patients to have comprehensive, personalized nutrition care. Rather than merely offering generalized nutrition education, RDNs are trained to provide medical nutrition therapy (MNT), which has been shown to improve health outcomes and be cost-effective for conditions such as type 2 DM, chronic kidney disease, hypertension, and obesity.10,11 In MNT, RDNs use the standardized 4-stepnutrition care process (NCP).12 The Table shows examples of how the NCP can be applied in HIH settings. Furthermore, in my experience, MNT from an RDN also contributes to patient satisfaction. Subjective observations from my team have indicated that patients often express more confidence in managing their diets by the time of HIH discharge.
RDNs can guide physicians and pharmacists in ordering oral nutrition supplements (ONS). Within the VHA, a “food first” approach is preferred to increase caloric intake, and patients must meet specific criteria for prescription of an ONS.13 Furthermore, ONS designed for specific medical conditions (eg, chronic kidney disease) are considered nonformulary and require an RDN evaluation.13 Including an RDN on the HIH team allows this evaluation process to begin early in the patient’s admission to the program and ensures that provision of ONS is clinically appropriate and cost-effective.
Care Coordination
HIH is highly interdisciplinary. Team members perform their respective roles and communicate with the team throughout the day. RDNs can help monitor patients and alert physicians for changes in blood glucose, gastrointestinal concerns, and weight. This is especially helpful for patients who do not have a planned nursing visit on the day of an RDN evaluation. The HIH RDN can also collaborate with other team members to address patient needs. For example, for patients with limited financial resources, the HIH RDN can provide nutrition education regarding cooking on a budget, and the HIH social worker can arrange free or low-cost meal services.
Tips
When hiring an HIH RDN, seek candidates with experience in inpatient, outpatient, and home care settings. As a hybrid of these 3 areas, the HIH RDN position requires a unique combination of acute care skills and health coaching. Additionally, in my experience, the HIH RDN interacts more frequently with the HIH team than other RDN colleagues, so it is important that candidates can work independently and take initiative. This type of position would not be suitable for entry-level RDNs.
Stagger HIH team visits to prevent overwhelming the patient and caregivers. Early in our program, my team quickly learned that patients and caregivers can feel overwhelmed with too many home visits upon admission to HIH. After seeing multiple HIH team members the same day, they were often too tired to focus well on diet education during my visit. Staggering visits (eg, completing the initial nutrition assessment 1 day to 1 week after the initial medical and pharmacy visits) has been an effective strategy to address this problem. Furthermore, some patients prefer that the initial RDN appointment is conducted by telephone, with an inperson reassessment the following week. In my experience, HIH workflow is dynamic by nature, so it is crucial to remain flexible and accommodate individual patient needs as much as possible.
Dietary behavior change is a long-term process, and restrictive hospital diets can be challenging to replicate at home. In a hospital setting, clinicians can order a specialized diet (eg, low sodium with fluid restriction for CHF patients), whereas efforts to implement these restrictions in the home setting can be cumbersome and negatively impact quality of life.7,14 Nevertheless, the effectiveness of medical treatment is compromised when patients do not adhere to dietary recommendations. Meal delivery services that offer specialized diets can be a useful resource for patients and caregivers who are unable to cook, and the HIH RDN can assist patients in ordering these services.
HIH patients may vary in terms of readiness to make dietary changes, and in addition to nutrition education, nutrition counseling is usually needed to effect behavior change. My team has found that consideration of the transtheoretical/ stages of change model can be a helpful approach. 15 The HIH RDN can tailor nutrition interventions to the patient’s stage of change. For example, for patients in the precontemplation stage, the HIH RDN would focus on providing information and addressing emotional aspects of dietary change. In contrast, for patients in the action stage of change, the HIH RDN might emphasize behavioral skill training and social support.15 Particularly for patients in the early stages of change, it may be unrealistic to expect full adoption of the recommended diet within the 30 days of the HIH program. However, by acknowledging the reality of the patient’s stage of change, the HIH RDN and team can then collaborate to support the patient in moving toward the next stage. Patients who are not ready for dietary behavior change during the 30 days of HIH may benefit from longer-term support, and the HIH RDN can arrange followup care with an outpatient RDN.
Conclusions
As the HIH model continues to be adopted across the VHA and other health care systems, it is crucial to consider the value and expertise of an RDN for guiding nutrition care in the HIH setting. The HIH RDN contributes to optimal health care delivery by leading nutritional aspects of patient care, offering personalized MNT, and coordinating and collaborating with team members to meet individual patient needs. An RDN can serve as a valuable resource for nutrition information and enhance the team’s overall services, with the potential to impact clinical outcomes and patient satisfaction.
1. Levine DM, Ouchi K, Blanchfield B, et al. Hospitallevel care at home for acutely ill adults: a randomized controlled trial. Ann Intern Med. 2020;172(2):77-85. doi:10.7326/M19-0600
2. Cai S, Grubbs A, Makineni R, Kinosian B, Phibbs CS, Intrator O. Evaluation of the Cincinnati Veterans Affairs medical center hospital-in-home program. J Am Geriatr Soc. 2018;66(7):1392-1398. doi:10.1111/jgs.15382
3. Cai S, Laurel PA, Makineni R, Marks ML. Evaluation of a hospital-in-home program implemented among veterans. Am J Manag Care. 2017;23(8):482-487.
4. Conley J, O’Brien CW, Leff BA, Bolen S, Zulman D. Alternative strategies to inpatient hospitalization for acute medical conditions: a systematic review. JAMA Intern Med. 2016;176(11):1693-1702. doi:10.1001/jamainternmed.2016.5974
5. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1144: Hospital in Home program, Appendix A, Hospital in Home program standards. January 19, 2021. Accessed May 5, 2022. https://www .va.gov/VHApublications/ViewPublication.asp?pub _ID=9157
6. Tibaldi V, Isaia G, Scarafiotti C, et al. Hospital at home for elderly patients with acute decompensation of chronic heart failure: a prospective randomized controlled trial. Arch Intern Med. 2009;169(17):1569-1575. doi:10.1001/archinternmed.2009.267
7. Abshire M, Xu J, Baptiste D, et al. Nutritional interventions in heart failure: a systematic review of the literature. J Card Fail. 2015;21(12):989-999. doi:10.1016/j.cardfail.2015.10.004
8. Bleich SN, Bennett WL, Gudzune KA, Cooper LA. National survey of US primary care physicians’ perspectives about causes of obesity and solutions to improve care. BMJ Open. 2012;2(6):e001871. Published 2012 Dec 20. doi:10.1136/bmjopen-2012-001871
9. Sousa AM. Benefits of dietitian home visits. J Am Diet Assoc. 1994;94(10):1149-1151. doi:10.1016/0002-8223(94)91136-3
10. Casas-Agustench P, Megías-Rangil I, Babio N. Economic benefit of dietetic-nutritional treatment in the multidisciplinary primary care team. Beneficio económico del tratamiento dietético-nutricional en el equipo multidisciplinario de atención primaria. Nutr Hosp. 2020;37(4):863-874. doi:10.20960/nh.03025
11. Lee J, Briggs Early K, Kovesdy CP, Lancaster K, Brown N, Steiber AL. The impact of RDNs on non-communicable diseases: proceedings from The State of Food and Nutrition Series Forum. J Acad Nutr Diet. 2022;122(1):166-174. doi:10.1016/j.jand.2021.02.021
12. Academy of Nutrition and Dietetics. Evidence analysis library, nutrition care process. Accessed May 5, 2022. https://www.andeal.org/ncp
13. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1438, clinical nutrition management and therapy. Appendix A, nutrition support therapy. September 19, 2019. Accessed January 20, 2022. https://www.va.gov/VHAPUBLICATIONS/ViewPublication .asp?pub_ID=8512
14. Vogelzang JL. Fifteen ways to enhance client outcomes by using your registered dietitian. Home Healthc Nurse. 2002;20(4):227-229. doi:10.1097/00004045-200204000-00005
15. Kristal AR, Glanz K, Curry SJ, Patterson RE. How can stages of change be best used in dietary interventions?. J Am Diet Assoc. 1999;99(6):679-684. doi:10.1016/S0002-8223(99)00165-0
1. Levine DM, Ouchi K, Blanchfield B, et al. Hospitallevel care at home for acutely ill adults: a randomized controlled trial. Ann Intern Med. 2020;172(2):77-85. doi:10.7326/M19-0600
2. Cai S, Grubbs A, Makineni R, Kinosian B, Phibbs CS, Intrator O. Evaluation of the Cincinnati Veterans Affairs medical center hospital-in-home program. J Am Geriatr Soc. 2018;66(7):1392-1398. doi:10.1111/jgs.15382
3. Cai S, Laurel PA, Makineni R, Marks ML. Evaluation of a hospital-in-home program implemented among veterans. Am J Manag Care. 2017;23(8):482-487.
4. Conley J, O’Brien CW, Leff BA, Bolen S, Zulman D. Alternative strategies to inpatient hospitalization for acute medical conditions: a systematic review. JAMA Intern Med. 2016;176(11):1693-1702. doi:10.1001/jamainternmed.2016.5974
5. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1144: Hospital in Home program, Appendix A, Hospital in Home program standards. January 19, 2021. Accessed May 5, 2022. https://www .va.gov/VHApublications/ViewPublication.asp?pub _ID=9157
6. Tibaldi V, Isaia G, Scarafiotti C, et al. Hospital at home for elderly patients with acute decompensation of chronic heart failure: a prospective randomized controlled trial. Arch Intern Med. 2009;169(17):1569-1575. doi:10.1001/archinternmed.2009.267
7. Abshire M, Xu J, Baptiste D, et al. Nutritional interventions in heart failure: a systematic review of the literature. J Card Fail. 2015;21(12):989-999. doi:10.1016/j.cardfail.2015.10.004
8. Bleich SN, Bennett WL, Gudzune KA, Cooper LA. National survey of US primary care physicians’ perspectives about causes of obesity and solutions to improve care. BMJ Open. 2012;2(6):e001871. Published 2012 Dec 20. doi:10.1136/bmjopen-2012-001871
9. Sousa AM. Benefits of dietitian home visits. J Am Diet Assoc. 1994;94(10):1149-1151. doi:10.1016/0002-8223(94)91136-3
10. Casas-Agustench P, Megías-Rangil I, Babio N. Economic benefit of dietetic-nutritional treatment in the multidisciplinary primary care team. Beneficio económico del tratamiento dietético-nutricional en el equipo multidisciplinario de atención primaria. Nutr Hosp. 2020;37(4):863-874. doi:10.20960/nh.03025
11. Lee J, Briggs Early K, Kovesdy CP, Lancaster K, Brown N, Steiber AL. The impact of RDNs on non-communicable diseases: proceedings from The State of Food and Nutrition Series Forum. J Acad Nutr Diet. 2022;122(1):166-174. doi:10.1016/j.jand.2021.02.021
12. Academy of Nutrition and Dietetics. Evidence analysis library, nutrition care process. Accessed May 5, 2022. https://www.andeal.org/ncp
13. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1438, clinical nutrition management and therapy. Appendix A, nutrition support therapy. September 19, 2019. Accessed January 20, 2022. https://www.va.gov/VHAPUBLICATIONS/ViewPublication .asp?pub_ID=8512
14. Vogelzang JL. Fifteen ways to enhance client outcomes by using your registered dietitian. Home Healthc Nurse. 2002;20(4):227-229. doi:10.1097/00004045-200204000-00005
15. Kristal AR, Glanz K, Curry SJ, Patterson RE. How can stages of change be best used in dietary interventions?. J Am Diet Assoc. 1999;99(6):679-684. doi:10.1016/S0002-8223(99)00165-0
New guideline for in-hospital care of diabetes says use CGMs
Goal-directed glycemic management – which may include new technologies for glucose monitoring – for non–critically ill hospitalized patients who have diabetes or newly recognized hyperglycemia can improve outcomes, according to a new practice guideline from the Endocrine Society.
Even though roughly 35% of hospitalized patients have diabetes or newly discovered hyperglycemia, there is “wide variability in glycemic management in clinical practice,” writing panel chair Mary Korytkowski, MD, from the University of Pittsburgh, said at the annual meeting of the Endocrine Society. “These patients get admitted to every patient service in the hospital, meaning that every clinical service will encounter this group of patients, and their glycemic management can have a major effect on their outcomes. Both short term and long term.”
This guideline provides strategies “to achieve previously recommended glycemic goals while also reducing the risk for hypoglycemia, and this includes inpatient use of insulin pump therapy or continuous glucose monitoring [CGM] devices, among others,” she said.
It also includes “recommendations for preoperative glycemic goals as well as when the use of correctional insulin – well known as sliding scale insulin – may be appropriate” and when it is not.
The document, which replaces a 2012 guideline, was published online in the Journal of Clinical Endocrinology & Metabolism.
A multidisciplinary panel developed the document over the last 3 years to answer 10 clinical practice questions related to management of non–critically ill hospitalized patients with diabetes or newly discovered hyperglycemia.
Use of CGM devices in hospital
The first recommendation is: “In adults with insulin-treated diabetes hospitalized for noncritical illness who are at high risk of hypoglycemia, we suggest the use of real-time [CGM] with confirmatory bedside point-of-care blood glucose monitoring for adjustments in insulin dosing rather than point-of-care blood glucose rather than testing alone in hospital settings where resources and training are available.” (Conditional recommendation. Low certainty of evidence).
“We were actually very careful in terms of looking at the data” for use of CGMs, Dr. Korytkowski said in an interview.
Although CGMs are approved by the Food and Drug Administration in the outpatient setting, and that’s becoming the standard of care there, they are not yet approved for in-hospital use.
However, the FDA granted an emergency allowance for use of CGMs in hospitals during the COVID-19 pandemic.
That was “when everyone was scrambling for what to do,” Dr. Korytkowski noted. “There was a shortage of personal protective equipment and a real interest in trying to limit the amount of exposure of healthcare personnel in some of these really critically ill patients for whom intravenous insulin therapy was used to control their glucose level.”
On March 1, the FDA granted Breakthrough Devices Designation for Dexcom CGM use in the hospital setting.
The new guideline suggests CGM be used to detect trends in glycemic management, with insulin dosing decisions made with point-of-care glucose measure (the standard of care).
To implement CGM for glycemic management in hospitals, Dr. Korytkowski said, would require “extensive staff and nursing education to have people with expertise available to provide support to nursing personnel who are both placing these devices, changing these devices, looking at trends, and then knowing when to remove them for certain procedures such as MRI or radiologic procedures.”
“We know that not all hospitals may be readily available to use these devices,” she said. “It is an area of active research. But the use of these devices during the pandemic, in both critical care and non–critical care setting has really provided us with a lot of information that was used to formulate this suggestion in the guideline.”
The document addresses the following areas: CGM, continuous subcutaneous insulin infusion pump therapy, inpatient diabetes education, prespecified preoperative glycemic targets, use of neutral protamine Hagedorn insulin for glucocorticoid or enteral nutrition-associated hyperglycemia, noninsulin therapies, preoperative carbohydrate-containing oral fluids, carbohydrate counting for prandial (mealtime) insulin dosing, and correctional and scheduled (basal or basal bolus) insulin therapies.
Nine key recommendations
Dr. Korytkowski identified nine key recommendations:
- CGM systems can help guide glycemic management with reduced risk for hypoglycemia.
- Patients experiencing glucocorticoid- or enteral nutrition–associated hyperglycemia require scheduled insulin therapy to address anticipated glucose excursions.
- Selected patients using insulin pump therapy prior to a hospital admission can continue to use these devices in the hospital if they have the mental and physical capacity to do so with knowledgeable hospital personnel.
- Diabetes self-management education provided to hospitalized patients can promote improved glycemic control following discharge with reductions in the risk for hospital readmission. “We know that is recommended for patients in the outpatient setting but often they do not get this,” she said. “We were able to observe that this can also impact long-term outcomes “
- Patients with diabetes scheduled for elective surgery may have improved postoperative outcomes when preoperative hemoglobin A1c is 8% or less and preoperative blood glucose is less than 180 mg/dL. “This recommendation answers the question: ‘Where should glycemic goals be for people who are undergoing surgery?’ ”
- Providing preoperative carbohydrate-containing beverages to patients with known diabetes is not recommended.
- Patients with newly recognized hyperglycemia or well-managed diabetes on noninsulin therapy may be treated with correctional insulin alone as initial therapy at hospital admission.
- Some noninsulin diabetes therapies can be used in combination with correction insulin for patients with type 2 diabetes who have mild hyperglycemia.
- Correctional insulin – “otherwise known as sliding-scale insulin” – can be used as initial therapy for patients with newly recognized hyperglycemia or type 2 diabetes treated with noninsulin therapy prior to hospital admission.
- Scheduled insulin therapy is preferred for patients experiencing persistent blood glucose values greater than 180 mg/dL and is recommended for patients using insulin therapy prior to admission.
The guideline writers’ hopes
“We hope that this guideline will resolve debates” about appropriate preoperative glycemic management and when sliding-scale insulin can be used and should not be used, said Dr. Korytkowski.
The authors also hope that “it will stimulate research funding for this very important aspect of diabetes care, and that hospitals will recognize the importance of having access to knowledgeable diabetes care and education specialists who can provide staff education regarding inpatient glycemic management, provide oversight for patients using insulin pump therapy or CGM devices, and empower hospital nurses to provide diabetes [self-management] education prior to patient discharge.”
Claire Pegg, the patient representative on the panel, hopes “that this guideline serves as the beginning of a conversation that will allow inpatient caregivers to provide individualized care to patients – some of whom may be self-sufficient with their glycemic management and others who need additional assistance.”
Development of the guideline was funded by the Endocrine Society. Dr. Korytkowski has reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Goal-directed glycemic management – which may include new technologies for glucose monitoring – for non–critically ill hospitalized patients who have diabetes or newly recognized hyperglycemia can improve outcomes, according to a new practice guideline from the Endocrine Society.
Even though roughly 35% of hospitalized patients have diabetes or newly discovered hyperglycemia, there is “wide variability in glycemic management in clinical practice,” writing panel chair Mary Korytkowski, MD, from the University of Pittsburgh, said at the annual meeting of the Endocrine Society. “These patients get admitted to every patient service in the hospital, meaning that every clinical service will encounter this group of patients, and their glycemic management can have a major effect on their outcomes. Both short term and long term.”
This guideline provides strategies “to achieve previously recommended glycemic goals while also reducing the risk for hypoglycemia, and this includes inpatient use of insulin pump therapy or continuous glucose monitoring [CGM] devices, among others,” she said.
It also includes “recommendations for preoperative glycemic goals as well as when the use of correctional insulin – well known as sliding scale insulin – may be appropriate” and when it is not.
The document, which replaces a 2012 guideline, was published online in the Journal of Clinical Endocrinology & Metabolism.
A multidisciplinary panel developed the document over the last 3 years to answer 10 clinical practice questions related to management of non–critically ill hospitalized patients with diabetes or newly discovered hyperglycemia.
Use of CGM devices in hospital
The first recommendation is: “In adults with insulin-treated diabetes hospitalized for noncritical illness who are at high risk of hypoglycemia, we suggest the use of real-time [CGM] with confirmatory bedside point-of-care blood glucose monitoring for adjustments in insulin dosing rather than point-of-care blood glucose rather than testing alone in hospital settings where resources and training are available.” (Conditional recommendation. Low certainty of evidence).
“We were actually very careful in terms of looking at the data” for use of CGMs, Dr. Korytkowski said in an interview.
Although CGMs are approved by the Food and Drug Administration in the outpatient setting, and that’s becoming the standard of care there, they are not yet approved for in-hospital use.
However, the FDA granted an emergency allowance for use of CGMs in hospitals during the COVID-19 pandemic.
That was “when everyone was scrambling for what to do,” Dr. Korytkowski noted. “There was a shortage of personal protective equipment and a real interest in trying to limit the amount of exposure of healthcare personnel in some of these really critically ill patients for whom intravenous insulin therapy was used to control their glucose level.”
On March 1, the FDA granted Breakthrough Devices Designation for Dexcom CGM use in the hospital setting.
The new guideline suggests CGM be used to detect trends in glycemic management, with insulin dosing decisions made with point-of-care glucose measure (the standard of care).
To implement CGM for glycemic management in hospitals, Dr. Korytkowski said, would require “extensive staff and nursing education to have people with expertise available to provide support to nursing personnel who are both placing these devices, changing these devices, looking at trends, and then knowing when to remove them for certain procedures such as MRI or radiologic procedures.”
“We know that not all hospitals may be readily available to use these devices,” she said. “It is an area of active research. But the use of these devices during the pandemic, in both critical care and non–critical care setting has really provided us with a lot of information that was used to formulate this suggestion in the guideline.”
The document addresses the following areas: CGM, continuous subcutaneous insulin infusion pump therapy, inpatient diabetes education, prespecified preoperative glycemic targets, use of neutral protamine Hagedorn insulin for glucocorticoid or enteral nutrition-associated hyperglycemia, noninsulin therapies, preoperative carbohydrate-containing oral fluids, carbohydrate counting for prandial (mealtime) insulin dosing, and correctional and scheduled (basal or basal bolus) insulin therapies.
Nine key recommendations
Dr. Korytkowski identified nine key recommendations:
- CGM systems can help guide glycemic management with reduced risk for hypoglycemia.
- Patients experiencing glucocorticoid- or enteral nutrition–associated hyperglycemia require scheduled insulin therapy to address anticipated glucose excursions.
- Selected patients using insulin pump therapy prior to a hospital admission can continue to use these devices in the hospital if they have the mental and physical capacity to do so with knowledgeable hospital personnel.
- Diabetes self-management education provided to hospitalized patients can promote improved glycemic control following discharge with reductions in the risk for hospital readmission. “We know that is recommended for patients in the outpatient setting but often they do not get this,” she said. “We were able to observe that this can also impact long-term outcomes “
- Patients with diabetes scheduled for elective surgery may have improved postoperative outcomes when preoperative hemoglobin A1c is 8% or less and preoperative blood glucose is less than 180 mg/dL. “This recommendation answers the question: ‘Where should glycemic goals be for people who are undergoing surgery?’ ”
- Providing preoperative carbohydrate-containing beverages to patients with known diabetes is not recommended.
- Patients with newly recognized hyperglycemia or well-managed diabetes on noninsulin therapy may be treated with correctional insulin alone as initial therapy at hospital admission.
- Some noninsulin diabetes therapies can be used in combination with correction insulin for patients with type 2 diabetes who have mild hyperglycemia.
- Correctional insulin – “otherwise known as sliding-scale insulin” – can be used as initial therapy for patients with newly recognized hyperglycemia or type 2 diabetes treated with noninsulin therapy prior to hospital admission.
- Scheduled insulin therapy is preferred for patients experiencing persistent blood glucose values greater than 180 mg/dL and is recommended for patients using insulin therapy prior to admission.
The guideline writers’ hopes
“We hope that this guideline will resolve debates” about appropriate preoperative glycemic management and when sliding-scale insulin can be used and should not be used, said Dr. Korytkowski.
The authors also hope that “it will stimulate research funding for this very important aspect of diabetes care, and that hospitals will recognize the importance of having access to knowledgeable diabetes care and education specialists who can provide staff education regarding inpatient glycemic management, provide oversight for patients using insulin pump therapy or CGM devices, and empower hospital nurses to provide diabetes [self-management] education prior to patient discharge.”
Claire Pegg, the patient representative on the panel, hopes “that this guideline serves as the beginning of a conversation that will allow inpatient caregivers to provide individualized care to patients – some of whom may be self-sufficient with their glycemic management and others who need additional assistance.”
Development of the guideline was funded by the Endocrine Society. Dr. Korytkowski has reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Goal-directed glycemic management – which may include new technologies for glucose monitoring – for non–critically ill hospitalized patients who have diabetes or newly recognized hyperglycemia can improve outcomes, according to a new practice guideline from the Endocrine Society.
Even though roughly 35% of hospitalized patients have diabetes or newly discovered hyperglycemia, there is “wide variability in glycemic management in clinical practice,” writing panel chair Mary Korytkowski, MD, from the University of Pittsburgh, said at the annual meeting of the Endocrine Society. “These patients get admitted to every patient service in the hospital, meaning that every clinical service will encounter this group of patients, and their glycemic management can have a major effect on their outcomes. Both short term and long term.”
This guideline provides strategies “to achieve previously recommended glycemic goals while also reducing the risk for hypoglycemia, and this includes inpatient use of insulin pump therapy or continuous glucose monitoring [CGM] devices, among others,” she said.
It also includes “recommendations for preoperative glycemic goals as well as when the use of correctional insulin – well known as sliding scale insulin – may be appropriate” and when it is not.
The document, which replaces a 2012 guideline, was published online in the Journal of Clinical Endocrinology & Metabolism.
A multidisciplinary panel developed the document over the last 3 years to answer 10 clinical practice questions related to management of non–critically ill hospitalized patients with diabetes or newly discovered hyperglycemia.
Use of CGM devices in hospital
The first recommendation is: “In adults with insulin-treated diabetes hospitalized for noncritical illness who are at high risk of hypoglycemia, we suggest the use of real-time [CGM] with confirmatory bedside point-of-care blood glucose monitoring for adjustments in insulin dosing rather than point-of-care blood glucose rather than testing alone in hospital settings where resources and training are available.” (Conditional recommendation. Low certainty of evidence).
“We were actually very careful in terms of looking at the data” for use of CGMs, Dr. Korytkowski said in an interview.
Although CGMs are approved by the Food and Drug Administration in the outpatient setting, and that’s becoming the standard of care there, they are not yet approved for in-hospital use.
However, the FDA granted an emergency allowance for use of CGMs in hospitals during the COVID-19 pandemic.
That was “when everyone was scrambling for what to do,” Dr. Korytkowski noted. “There was a shortage of personal protective equipment and a real interest in trying to limit the amount of exposure of healthcare personnel in some of these really critically ill patients for whom intravenous insulin therapy was used to control their glucose level.”
On March 1, the FDA granted Breakthrough Devices Designation for Dexcom CGM use in the hospital setting.
The new guideline suggests CGM be used to detect trends in glycemic management, with insulin dosing decisions made with point-of-care glucose measure (the standard of care).
To implement CGM for glycemic management in hospitals, Dr. Korytkowski said, would require “extensive staff and nursing education to have people with expertise available to provide support to nursing personnel who are both placing these devices, changing these devices, looking at trends, and then knowing when to remove them for certain procedures such as MRI or radiologic procedures.”
“We know that not all hospitals may be readily available to use these devices,” she said. “It is an area of active research. But the use of these devices during the pandemic, in both critical care and non–critical care setting has really provided us with a lot of information that was used to formulate this suggestion in the guideline.”
The document addresses the following areas: CGM, continuous subcutaneous insulin infusion pump therapy, inpatient diabetes education, prespecified preoperative glycemic targets, use of neutral protamine Hagedorn insulin for glucocorticoid or enteral nutrition-associated hyperglycemia, noninsulin therapies, preoperative carbohydrate-containing oral fluids, carbohydrate counting for prandial (mealtime) insulin dosing, and correctional and scheduled (basal or basal bolus) insulin therapies.
Nine key recommendations
Dr. Korytkowski identified nine key recommendations:
- CGM systems can help guide glycemic management with reduced risk for hypoglycemia.
- Patients experiencing glucocorticoid- or enteral nutrition–associated hyperglycemia require scheduled insulin therapy to address anticipated glucose excursions.
- Selected patients using insulin pump therapy prior to a hospital admission can continue to use these devices in the hospital if they have the mental and physical capacity to do so with knowledgeable hospital personnel.
- Diabetes self-management education provided to hospitalized patients can promote improved glycemic control following discharge with reductions in the risk for hospital readmission. “We know that is recommended for patients in the outpatient setting but often they do not get this,” she said. “We were able to observe that this can also impact long-term outcomes “
- Patients with diabetes scheduled for elective surgery may have improved postoperative outcomes when preoperative hemoglobin A1c is 8% or less and preoperative blood glucose is less than 180 mg/dL. “This recommendation answers the question: ‘Where should glycemic goals be for people who are undergoing surgery?’ ”
- Providing preoperative carbohydrate-containing beverages to patients with known diabetes is not recommended.
- Patients with newly recognized hyperglycemia or well-managed diabetes on noninsulin therapy may be treated with correctional insulin alone as initial therapy at hospital admission.
- Some noninsulin diabetes therapies can be used in combination with correction insulin for patients with type 2 diabetes who have mild hyperglycemia.
- Correctional insulin – “otherwise known as sliding-scale insulin” – can be used as initial therapy for patients with newly recognized hyperglycemia or type 2 diabetes treated with noninsulin therapy prior to hospital admission.
- Scheduled insulin therapy is preferred for patients experiencing persistent blood glucose values greater than 180 mg/dL and is recommended for patients using insulin therapy prior to admission.
The guideline writers’ hopes
“We hope that this guideline will resolve debates” about appropriate preoperative glycemic management and when sliding-scale insulin can be used and should not be used, said Dr. Korytkowski.
The authors also hope that “it will stimulate research funding for this very important aspect of diabetes care, and that hospitals will recognize the importance of having access to knowledgeable diabetes care and education specialists who can provide staff education regarding inpatient glycemic management, provide oversight for patients using insulin pump therapy or CGM devices, and empower hospital nurses to provide diabetes [self-management] education prior to patient discharge.”
Claire Pegg, the patient representative on the panel, hopes “that this guideline serves as the beginning of a conversation that will allow inpatient caregivers to provide individualized care to patients – some of whom may be self-sufficient with their glycemic management and others who need additional assistance.”
Development of the guideline was funded by the Endocrine Society. Dr. Korytkowski has reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM ENDO 2022
FDA: Urgent device correction, recall for Philips ventilator
The U.S. Food and Drug Administration has announced a Class I recall for Philips Respironics V60 and V60 Plus ventilators, citing a power failure leading to potential oxygen deprivation. Class I recalls, the most severe, are reserved for devices that may cause serious injury or death, as noted in the FDA’s announcement. As of April 14, one death and four injuries have been associated with this device failure.
These ventilators are commonly used in hospitals or under medical supervision for patients who have difficulty regulating breathing on their own. Normally, if oxygen flow is interrupted, the device sounds alarms, alerting supervisors. The failure comes when a power fluctuation causes the device to randomly shut down, which forces the alarm system to reboot. This internal disruption is the reason for the recall.
When the device shuts down out of the blue, it may or may not sound the requisite alarm that would allow providers to intervene. If the device does not sound the alarm, patients may lose oxygen for an extended period, without a provider even knowing.
Philips was notified of these problems and began the recall process on March 10. Currently, it is estimated that 56,671 devices have been distributed throughout the United States. The FDA and Philips Respironics advise that if providers are already using these ventilators, they may continue to do so in accordance with extra set of instructions.
First, customers should connect the device to an external alarm or nurse call system. Second, they should use an external oxygen monitor and a pulse oximeter to keep track of air flow. Finally, if one is available, there should be a backup ventilator on the premises. That way, if there is an interruption in oxygen flow, someone will be alerted and can quickly intervene.
If there is a problem, the patient should be removed from the Philips ventilator and immediately placed on an alternate device. The FDA instructs customers who have experienced problems to report them to its MedWatch database.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has announced a Class I recall for Philips Respironics V60 and V60 Plus ventilators, citing a power failure leading to potential oxygen deprivation. Class I recalls, the most severe, are reserved for devices that may cause serious injury or death, as noted in the FDA’s announcement. As of April 14, one death and four injuries have been associated with this device failure.
These ventilators are commonly used in hospitals or under medical supervision for patients who have difficulty regulating breathing on their own. Normally, if oxygen flow is interrupted, the device sounds alarms, alerting supervisors. The failure comes when a power fluctuation causes the device to randomly shut down, which forces the alarm system to reboot. This internal disruption is the reason for the recall.
When the device shuts down out of the blue, it may or may not sound the requisite alarm that would allow providers to intervene. If the device does not sound the alarm, patients may lose oxygen for an extended period, without a provider even knowing.
Philips was notified of these problems and began the recall process on March 10. Currently, it is estimated that 56,671 devices have been distributed throughout the United States. The FDA and Philips Respironics advise that if providers are already using these ventilators, they may continue to do so in accordance with extra set of instructions.
First, customers should connect the device to an external alarm or nurse call system. Second, they should use an external oxygen monitor and a pulse oximeter to keep track of air flow. Finally, if one is available, there should be a backup ventilator on the premises. That way, if there is an interruption in oxygen flow, someone will be alerted and can quickly intervene.
If there is a problem, the patient should be removed from the Philips ventilator and immediately placed on an alternate device. The FDA instructs customers who have experienced problems to report them to its MedWatch database.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has announced a Class I recall for Philips Respironics V60 and V60 Plus ventilators, citing a power failure leading to potential oxygen deprivation. Class I recalls, the most severe, are reserved for devices that may cause serious injury or death, as noted in the FDA’s announcement. As of April 14, one death and four injuries have been associated with this device failure.
These ventilators are commonly used in hospitals or under medical supervision for patients who have difficulty regulating breathing on their own. Normally, if oxygen flow is interrupted, the device sounds alarms, alerting supervisors. The failure comes when a power fluctuation causes the device to randomly shut down, which forces the alarm system to reboot. This internal disruption is the reason for the recall.
When the device shuts down out of the blue, it may or may not sound the requisite alarm that would allow providers to intervene. If the device does not sound the alarm, patients may lose oxygen for an extended period, without a provider even knowing.
Philips was notified of these problems and began the recall process on March 10. Currently, it is estimated that 56,671 devices have been distributed throughout the United States. The FDA and Philips Respironics advise that if providers are already using these ventilators, they may continue to do so in accordance with extra set of instructions.
First, customers should connect the device to an external alarm or nurse call system. Second, they should use an external oxygen monitor and a pulse oximeter to keep track of air flow. Finally, if one is available, there should be a backup ventilator on the premises. That way, if there is an interruption in oxygen flow, someone will be alerted and can quickly intervene.
If there is a problem, the patient should be removed from the Philips ventilator and immediately placed on an alternate device. The FDA instructs customers who have experienced problems to report them to its MedWatch database.
A version of this article first appeared on Medscape.com.
Hospital medicine gains popularity among newly minted physicians
In a new study, published in Annals of Internal Medicine, researchers from ABIM reviewed certification data from 67,902 general internists, accounting for 80% of all general internists certified in the United States from 1990 to 2017.
The researchers also used data from Medicare fee-for-service claims from 2008-2018 to measure and categorize practice setting types. The claims were from patients aged 65 years or older with at least 20 evaluation and management visits each year. Practice settings were categorized as hospitalist, outpatient, or mixed.
“ABIM is always working to understand the real-life experience of physicians, and this project grew out of that sort of analysis,” lead author Bradley M. Gray, PhD, a health services researcher at ABIM in Philadelphia, said in an interview. “We wanted to better understand practice setting, because that relates to the kinds of questions that we ask on our certifying exams. When we did this, we noticed a trend toward hospital medicine.”
Overall, the percentages of general internists in hospitalist practice and outpatient-only practice increased during the study period, from 25% to 40% and from 23% to 38%, respectively. By contrast, the percentage of general internists in a mixed-practice setting decreased from 52% to 23%, a 56% decline. Most of the physicians who left the mixed practice setting switched to outpatient-only practices.
Among the internists certified in 2017, 71% practiced as hospitalists, compared with 8% practicing as outpatient-only physicians. Most physicians remained in their original choice of practice setting. For physicians certified in 1999 and 2012, 86% and 85%, respectively, of those who chose hospitalist medicine remained in the hospital setting 5 years later, as did 95% of outpatient physicians, but only 57% of mixed-practice physicians.
The shift to outpatient practice among senior physicians offset the potential decline in outpatient primary care resulting from the increased choice of hospitalist medicine by new internists, the researchers noted.
The study findings were limited by several factors, including the reliance on Medicare fee-for-service claims, the researchers noted.
“We were surprised by both the dramatic shift toward hospital medicine by new physicians and the shift to outpatient only (an extreme category) for more senior physicians,” Dr. Gray said in an interview.
The shift toward outpatient practice among older physicians may be driven by convenience, said Dr. Gray. “I suspect that it is more efficient to specialize in terms of practice setting. Only seeing patients in the outpatient setting means that you don’t have to travel to the hospital, which can be time consuming.
“Also, with fewer new physicians going into primary care, older physicians need to focus on outpatient visits. This could be problematic in the future as more senior physicians retire and are replaced by new physicians who focus on hospital care,” which could lead to more shortages in primary care physicians, he explained.
The trend toward hospital medicine as a career has been going on since before the pandemic, said Dr. Gray. “I don’t think the pandemic will ultimately impact this trend. That said, at least in the short run, there may have been a decreased demand for primary care, but that is just my speculation. As more data flow in we will be able to answer this question more directly.”
Next steps for research included digging deeper into the data to understand the nature of conditions facing hospitalists, Dr. Gray said.
Implications for primary care
“This study provides an updated snapshot of the popularity of hospital medicine,” said Bradley A. Sharpe, MD, of the division of hospital medicine at the University of California, San Francisco. “It is also important to conduct this study now as health systems think about the challenge of providing high-quality primary care with a rapidly decreasing number of internists choosing to practice outpatient medicine.” Dr. Sharpe was not involved in the study.
“The most surprising finding to me was not the increase in general internists focusing on hospital medicine, but the amount of the increase; it is remarkable that nearly three quarters of general internists are choosing to practice as hospitalists,” Dr. Sharpe noted.
“I think there are a number of key factors at play,” he said. “First, as hospital medicine as a field is now more than 25 years old, hospitals and health systems have evolved to create hospital medicine jobs that are interesting, engaging, rewarding (financially and otherwise), doable, and sustainable. Second, being an outpatient internist is incredibly challenging; multiple studies have shown that it is essentially impossible to complete the evidence-based preventive care for a panel of patients on top of everything else. We know burnout rates are often higher among primary care and family medicine providers. On top of that, the expansion of electronic health records and patient access has led to a massive increase in messages to providers; this has been shown to be associated with burnout.”
The potential impact of the pandemic on physicians’ choices and the trend toward hospital medicine is an interested question, Dr. Sharpe said. The current study showed only trends through 2017.
“To be honest, I think it is difficult to predict,” he said. “Hospitalists shouldered much of the burden of COVID care nationally and burnout rates are high. One could imagine the extra work (as well as concern for personal safety) could lead to fewer providers choosing hospital medicine.
“At the same time, the pandemic has driven many of us to reflect on life and our values and what is important and, through that lens, providers might choose hospital medicine as a more sustainable, do-able, rewarding, and enjoyable career choice,” Dr. Sharpe emphasized.
“Additional research could explore the drivers of this clear trend toward hospital medicine. Determining what is motivating this trend could help hospitals and health systems ensure they have the right workforce for the future and, in particular, how to create outpatient positions that are attractive and rewarding,” he said.
The study received no outside funding. The researchers and Dr. Sharpe disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a new study, published in Annals of Internal Medicine, researchers from ABIM reviewed certification data from 67,902 general internists, accounting for 80% of all general internists certified in the United States from 1990 to 2017.
The researchers also used data from Medicare fee-for-service claims from 2008-2018 to measure and categorize practice setting types. The claims were from patients aged 65 years or older with at least 20 evaluation and management visits each year. Practice settings were categorized as hospitalist, outpatient, or mixed.
“ABIM is always working to understand the real-life experience of physicians, and this project grew out of that sort of analysis,” lead author Bradley M. Gray, PhD, a health services researcher at ABIM in Philadelphia, said in an interview. “We wanted to better understand practice setting, because that relates to the kinds of questions that we ask on our certifying exams. When we did this, we noticed a trend toward hospital medicine.”
Overall, the percentages of general internists in hospitalist practice and outpatient-only practice increased during the study period, from 25% to 40% and from 23% to 38%, respectively. By contrast, the percentage of general internists in a mixed-practice setting decreased from 52% to 23%, a 56% decline. Most of the physicians who left the mixed practice setting switched to outpatient-only practices.
Among the internists certified in 2017, 71% practiced as hospitalists, compared with 8% practicing as outpatient-only physicians. Most physicians remained in their original choice of practice setting. For physicians certified in 1999 and 2012, 86% and 85%, respectively, of those who chose hospitalist medicine remained in the hospital setting 5 years later, as did 95% of outpatient physicians, but only 57% of mixed-practice physicians.
The shift to outpatient practice among senior physicians offset the potential decline in outpatient primary care resulting from the increased choice of hospitalist medicine by new internists, the researchers noted.
The study findings were limited by several factors, including the reliance on Medicare fee-for-service claims, the researchers noted.
“We were surprised by both the dramatic shift toward hospital medicine by new physicians and the shift to outpatient only (an extreme category) for more senior physicians,” Dr. Gray said in an interview.
The shift toward outpatient practice among older physicians may be driven by convenience, said Dr. Gray. “I suspect that it is more efficient to specialize in terms of practice setting. Only seeing patients in the outpatient setting means that you don’t have to travel to the hospital, which can be time consuming.
“Also, with fewer new physicians going into primary care, older physicians need to focus on outpatient visits. This could be problematic in the future as more senior physicians retire and are replaced by new physicians who focus on hospital care,” which could lead to more shortages in primary care physicians, he explained.
The trend toward hospital medicine as a career has been going on since before the pandemic, said Dr. Gray. “I don’t think the pandemic will ultimately impact this trend. That said, at least in the short run, there may have been a decreased demand for primary care, but that is just my speculation. As more data flow in we will be able to answer this question more directly.”
Next steps for research included digging deeper into the data to understand the nature of conditions facing hospitalists, Dr. Gray said.
Implications for primary care
“This study provides an updated snapshot of the popularity of hospital medicine,” said Bradley A. Sharpe, MD, of the division of hospital medicine at the University of California, San Francisco. “It is also important to conduct this study now as health systems think about the challenge of providing high-quality primary care with a rapidly decreasing number of internists choosing to practice outpatient medicine.” Dr. Sharpe was not involved in the study.
“The most surprising finding to me was not the increase in general internists focusing on hospital medicine, but the amount of the increase; it is remarkable that nearly three quarters of general internists are choosing to practice as hospitalists,” Dr. Sharpe noted.
“I think there are a number of key factors at play,” he said. “First, as hospital medicine as a field is now more than 25 years old, hospitals and health systems have evolved to create hospital medicine jobs that are interesting, engaging, rewarding (financially and otherwise), doable, and sustainable. Second, being an outpatient internist is incredibly challenging; multiple studies have shown that it is essentially impossible to complete the evidence-based preventive care for a panel of patients on top of everything else. We know burnout rates are often higher among primary care and family medicine providers. On top of that, the expansion of electronic health records and patient access has led to a massive increase in messages to providers; this has been shown to be associated with burnout.”
The potential impact of the pandemic on physicians’ choices and the trend toward hospital medicine is an interested question, Dr. Sharpe said. The current study showed only trends through 2017.
“To be honest, I think it is difficult to predict,” he said. “Hospitalists shouldered much of the burden of COVID care nationally and burnout rates are high. One could imagine the extra work (as well as concern for personal safety) could lead to fewer providers choosing hospital medicine.
“At the same time, the pandemic has driven many of us to reflect on life and our values and what is important and, through that lens, providers might choose hospital medicine as a more sustainable, do-able, rewarding, and enjoyable career choice,” Dr. Sharpe emphasized.
“Additional research could explore the drivers of this clear trend toward hospital medicine. Determining what is motivating this trend could help hospitals and health systems ensure they have the right workforce for the future and, in particular, how to create outpatient positions that are attractive and rewarding,” he said.
The study received no outside funding. The researchers and Dr. Sharpe disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a new study, published in Annals of Internal Medicine, researchers from ABIM reviewed certification data from 67,902 general internists, accounting for 80% of all general internists certified in the United States from 1990 to 2017.
The researchers also used data from Medicare fee-for-service claims from 2008-2018 to measure and categorize practice setting types. The claims were from patients aged 65 years or older with at least 20 evaluation and management visits each year. Practice settings were categorized as hospitalist, outpatient, or mixed.
“ABIM is always working to understand the real-life experience of physicians, and this project grew out of that sort of analysis,” lead author Bradley M. Gray, PhD, a health services researcher at ABIM in Philadelphia, said in an interview. “We wanted to better understand practice setting, because that relates to the kinds of questions that we ask on our certifying exams. When we did this, we noticed a trend toward hospital medicine.”
Overall, the percentages of general internists in hospitalist practice and outpatient-only practice increased during the study period, from 25% to 40% and from 23% to 38%, respectively. By contrast, the percentage of general internists in a mixed-practice setting decreased from 52% to 23%, a 56% decline. Most of the physicians who left the mixed practice setting switched to outpatient-only practices.
Among the internists certified in 2017, 71% practiced as hospitalists, compared with 8% practicing as outpatient-only physicians. Most physicians remained in their original choice of practice setting. For physicians certified in 1999 and 2012, 86% and 85%, respectively, of those who chose hospitalist medicine remained in the hospital setting 5 years later, as did 95% of outpatient physicians, but only 57% of mixed-practice physicians.
The shift to outpatient practice among senior physicians offset the potential decline in outpatient primary care resulting from the increased choice of hospitalist medicine by new internists, the researchers noted.
The study findings were limited by several factors, including the reliance on Medicare fee-for-service claims, the researchers noted.
“We were surprised by both the dramatic shift toward hospital medicine by new physicians and the shift to outpatient only (an extreme category) for more senior physicians,” Dr. Gray said in an interview.
The shift toward outpatient practice among older physicians may be driven by convenience, said Dr. Gray. “I suspect that it is more efficient to specialize in terms of practice setting. Only seeing patients in the outpatient setting means that you don’t have to travel to the hospital, which can be time consuming.
“Also, with fewer new physicians going into primary care, older physicians need to focus on outpatient visits. This could be problematic in the future as more senior physicians retire and are replaced by new physicians who focus on hospital care,” which could lead to more shortages in primary care physicians, he explained.
The trend toward hospital medicine as a career has been going on since before the pandemic, said Dr. Gray. “I don’t think the pandemic will ultimately impact this trend. That said, at least in the short run, there may have been a decreased demand for primary care, but that is just my speculation. As more data flow in we will be able to answer this question more directly.”
Next steps for research included digging deeper into the data to understand the nature of conditions facing hospitalists, Dr. Gray said.
Implications for primary care
“This study provides an updated snapshot of the popularity of hospital medicine,” said Bradley A. Sharpe, MD, of the division of hospital medicine at the University of California, San Francisco. “It is also important to conduct this study now as health systems think about the challenge of providing high-quality primary care with a rapidly decreasing number of internists choosing to practice outpatient medicine.” Dr. Sharpe was not involved in the study.
“The most surprising finding to me was not the increase in general internists focusing on hospital medicine, but the amount of the increase; it is remarkable that nearly three quarters of general internists are choosing to practice as hospitalists,” Dr. Sharpe noted.
“I think there are a number of key factors at play,” he said. “First, as hospital medicine as a field is now more than 25 years old, hospitals and health systems have evolved to create hospital medicine jobs that are interesting, engaging, rewarding (financially and otherwise), doable, and sustainable. Second, being an outpatient internist is incredibly challenging; multiple studies have shown that it is essentially impossible to complete the evidence-based preventive care for a panel of patients on top of everything else. We know burnout rates are often higher among primary care and family medicine providers. On top of that, the expansion of electronic health records and patient access has led to a massive increase in messages to providers; this has been shown to be associated with burnout.”
The potential impact of the pandemic on physicians’ choices and the trend toward hospital medicine is an interested question, Dr. Sharpe said. The current study showed only trends through 2017.
“To be honest, I think it is difficult to predict,” he said. “Hospitalists shouldered much of the burden of COVID care nationally and burnout rates are high. One could imagine the extra work (as well as concern for personal safety) could lead to fewer providers choosing hospital medicine.
“At the same time, the pandemic has driven many of us to reflect on life and our values and what is important and, through that lens, providers might choose hospital medicine as a more sustainable, do-able, rewarding, and enjoyable career choice,” Dr. Sharpe emphasized.
“Additional research could explore the drivers of this clear trend toward hospital medicine. Determining what is motivating this trend could help hospitals and health systems ensure they have the right workforce for the future and, in particular, how to create outpatient positions that are attractive and rewarding,” he said.
The study received no outside funding. The researchers and Dr. Sharpe disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Where Does the Hospital Belong? Perspectives on Hospital at Home in the 21st Century
From Medically Home Group, Boston, MA.
Brick-and-mortar hospitals in the United States have historically been considered the dominant setting for providing care to patients. The coordination and delivery of care has previously been bound to physical hospitals largely because multidisciplinary services were only accessible in an individual location. While the fundamental make-up of these services remains unchanged, these services are now available in alternate settings. Some of these services include access to a patient care team, supplies, diagnostics, pharmacy, and advanced therapeutic interventions. Presently, the physical environment is becoming increasingly irrelevant as the core of what makes the traditional hospital—the professional staff, collaborative work processes, and the dynamics of the space—have all been translated into a modern digitally integrated environment. The elements necessary to providing safe, effective care in a physical hospital setting are now available in a patient’s home.
Impetus for the Model
As hospitals reconsider how and where they deliver patient care because of limited resources, the hospital-at-home model has gained significant momentum and interest. This model transforms a home into a hospital. The inpatient acute care episode is entirely substituted with an intensive at-home hospital admission enabled by technology, multidisciplinary teams, and ancillary services. Furthermore, patients requiring post-acute support can be transitioned to their next phase of care seamlessly. Given the nationwide nursing shortage, aging population, challenges uncovered by the COVID-19 pandemic, rising hospital costs, nurse/provider burnout related to challenging work environments, and capacity constraints, a shift toward the combination of virtual and in-home care is imperative. The hospital-at-home model has been associated with superior patient outcomes, including reduced risks of delirium, improved functional status, improved patient and family member satisfaction, reduced mortality, reduced readmissions, and significantly lower costs.1 COVID-19 alone has unmasked major facility-based deficiencies and limitations of our health care system. While the pandemic is not the impetus for the hospital-at-home model, the extended stress of this event has created a unique opportunity to reimagine and transform our health care delivery system so that it is less fragmented and more flexible.
Nursing in the Model
Nursing is central to the hospital-at-home model. Virtual nurses provide meticulous care plan oversight, assessment, and documentation across in-home service providers, to ensure holistic, safe, transparent, and continuous progression toward care plan milestones. The virtual nurse monitors patients using in-home technology that is set up at the time of admission. Connecting with patients to verify social and medical needs, the virtual nurse advocates for their patients and uses these technologies to care and deploy on-demand hands-on services to the patient. Service providers such as paramedics, infusion nurses, or home health nurses may be deployed to provide services in the patient’s home. By bringing in supplies, therapeutics, and interdisciplinary team members, the capabilities of a brick-and-mortar hospital are replicated in the home. All actions that occur wherever the patient is receiving care are overseen by professional nursing staff; in short, virtual nurses are the equivalent of bedside nurses in the brick-and-mortar health care facilities.
Potential Benefits
There are many benefits to the hospital-at-home model (Table). This health care model can be particularly helpful for patients who require frequent admission to acute care facilities, and is well suited for patients with a range of conditions, including those with COVID-19, pneumonia, cellulitis, or congestive heart failure. This care model helps eliminate some of the stressors for patients who have chronic illnesses or other conditions that require frequent hospital admissions. Patients can independently recover at home and can also be surrounded by their loved ones and pets while recovering. This care approach additionally eliminates the risk of hospital-acquired infections and injuries. The hospital-at-home model allows for increased mobility,2 as patients are familiar with their surroundings, resulting in reduced onset of delirium. Additionally, patients with improved mobility performance are less likely to experience negative health outcomes.3 There is less chance of sleep disruption as the patient is sleeping in their own bed—no unfamiliar roommate, no call bells or health care personnel frequently coming into the room. The in-home technology set up for remote patient monitoring is designed with the user in mind. Ease of use empowers the patient to collaborate with their care team on their own terms and center the priorities of themselves and their families.

Positive Outcomes
The hospital-at-home model is associated with positive outcomes. The authors of a systematic review identified 10 randomized controlled trials of hospital-at-home programs (with a total of 1372 patients), but were able to obtain data for only 5 of these trials (with a total of 844 patients).4 They found a 38% reduction in 6-month mortality for patients who received hospital care at home, as well as significantly higher patient satisfaction across a range of medical conditions, including patients with cellulitis and community-acquired pneumonia, as well as elderly patients with multiple medical conditions. The authors concluded that hospital care at home was less expensive than admission to an acute care hospital.4 Similarly, a meta-analysis done by Caplan et al5 that included 61 randomized controlled trials concluded that hospital at home is associated with reductions in mortality, readmission rates, and cost, and increases in patient and caregiver satisfaction. Levine et al2 found reduced costs and utilization with home hospitalization compared to in-hospital care, as well as improved patient mobility status.
The home is the ideal place to empower patients and caregivers to engage in self-management.2 Receiving hospital care at home eliminates the need for dealing with transportation arrangements, traffic, road tolls, and time/scheduling constraints, or finding care for a dependent family member, some of the many stressors that may be experienced by patients who require frequent trips to the hospital. For patients who may not be clinically suitable candidates for hospital at home, such as those requiring critical care intervention and support, the brick-and-mortar hospital is still the appropriate site of care. The hospital-at-home model helps prevent bed shortages in brick-and-mortar hospital settings by allowing hospital care at home for patients who meet preset criteria. These patients can be hospitalized in alternative locations such as their own homes or the residence of a friend. This helps increase health system capacity as well as resiliency.
In addition to expanding safe and appropriate treatment spaces, the hospital-at-home model helps increase access to care for patients during nonstandard hours, including weekends, holidays, or when the waiting time in the emergency room is painfully long. Furthermore, providing care in the home gives the clinical team valuable insight into the patient’s daily life and routine. Performing medication reconciliation with the medicine cabinet in sight and dietary education in a patient’s kitchen are powerful touch points.2 For example, a patient with congestive heart failure who must undergo diuresis is much more likely to meet their care goals when their home diet is aligned with the treatment goal. By being able to see exactly what is in a patient’s pantry and fridge, the care team can create a much more tailored approach to sodium intake and fluid management. Providers can create and execute true patient-centric care as they gain direct insight into the patient’s lifestyle, which is clearly valuable when creating care plans for complex chronic health issues.
Challenges to Implementation and Scaling
Although there are clear benefits to hospital at home, how to best implement and scale this model presents a challenge. In addition to educating patients and families about this model of care, health care systems must expand their hospital-at-home programs and provide education about this model to clinical staff and trainees, and insurers must create reimbursement paradigms. Patients meeting eligibility criteria to enroll in hospital at home is the easiest hurdle, as hospital-at-home programs function best when they enroll and service as many patients as possible, including underserved populations.
Upfront Costs and Cost Savings
While there are upfront costs to set up technology and coordinate services, hospital at home also provides significant total cost savings when compared to coordination associated with brick-and-mortar admission. Hospital care accounts for about one-third of total medical expenditures and is a leading cause of debt.2 Eliminating fixed hospital costs such as facility, overhead, and equipment costs through adoption of the hospital-at-home model can lead to a reduction in expenditures. It has been found that fewer laboratory and diagnostic tests are ordered for hospital-at-home patients when compared to similar patients in brick-and-mortar hospital settings, with comparable or better clinical patient outcomes.6 Furthermore, it is estimated that there are cost savings of 19% to 30% when compared to traditional inpatient care.6 Without legislative action, upon the end of the current COVID-19 public health emergency, the Centers for Medicare & Medicaid Service’s Acute Hospital Care at Home waiver will terminate. This could slow down scaling of the model.However, over the past 2 years there has been enough buy-in from major health systems and patients to continue the momentum of the model’s growth. When setting up a hospital-at-home program, it would be wise to consider a few factors: where in the hospital or health system entity structure the hospital-at-home program will reside, which existing resources can be leveraged within the hospital or health system, and what are the state or federal regulatory requirements for such a program. This type of program continues to fill gaps within the US health care system, meeting the needs of widely overlooked populations and increasing access to essential ancillary services.
Conclusion
It is time to consider our bias toward hospital-first options when managing the care needs of our patients. Health care providers have the option to advocate for holistic care, better experience, and better outcomes. Home-based options are safe, equitable, and patient-centric. Increased costs, consumerism, and technology have pushed us to think about alternative approaches to patient care delivery, and the pandemic created a unique opportunity to see just how far the health care system could stretch itself with capacity constraints, insufficient resources, and staff shortages. In light of new possibilities, it is time to reimagine and transform our health care delivery system so that it is unified, seamless, cohesive, and flexible.
Corresponding author: Payal Sharma, DNP, MSN, RN, FNP-BC, CBN; [email protected].
Disclosures: None reported.
1. Cai S, Laurel PA, Makineni R, Marks ML. Evaluation of a hospital-in-home program implemented among veterans. Am J Manag Care. 2017;23(8):482-487.
2. Levine DM, Ouchi K, Blanchfield B, et al. Hospital-level care at home for acutely ill adults: a pilot randomized controlled trial. J Gen Intern Med. 2018;33(5):729-736. doi:10.1007/s11606-018-4307-z
3. Shuman V, Coyle PC, Perera S,et al. Association between improved mobility and distal health outcomes. J Gerontol A Biol Sci Med Sci. 2020;75(12):2412-2417. doi:10.1093/gerona/glaa086
4. Shepperd S, Doll H, Angus RM, et al. Avoiding hospital admission through provision of hospital care at home: a systematic review and meta-analysis of individual patient data. CMAJ. 2009;180(2):175-182. doi:10.1503/cmaj.081491
5. Caplan GA, Sulaiman NS, Mangin DA, et al. A meta-analysis of “hospital in the home”. Med J Aust. 2012;197(9):512-519. doi:10.5694/mja12.10480
6. Hospital at Home. Johns Hopkins Medicine. Healthcare Solutions. Accessed May 20, 2022. https://www.johnshopkinssolutions.com/solution/hospital-at-home/
From Medically Home Group, Boston, MA.
Brick-and-mortar hospitals in the United States have historically been considered the dominant setting for providing care to patients. The coordination and delivery of care has previously been bound to physical hospitals largely because multidisciplinary services were only accessible in an individual location. While the fundamental make-up of these services remains unchanged, these services are now available in alternate settings. Some of these services include access to a patient care team, supplies, diagnostics, pharmacy, and advanced therapeutic interventions. Presently, the physical environment is becoming increasingly irrelevant as the core of what makes the traditional hospital—the professional staff, collaborative work processes, and the dynamics of the space—have all been translated into a modern digitally integrated environment. The elements necessary to providing safe, effective care in a physical hospital setting are now available in a patient’s home.
Impetus for the Model
As hospitals reconsider how and where they deliver patient care because of limited resources, the hospital-at-home model has gained significant momentum and interest. This model transforms a home into a hospital. The inpatient acute care episode is entirely substituted with an intensive at-home hospital admission enabled by technology, multidisciplinary teams, and ancillary services. Furthermore, patients requiring post-acute support can be transitioned to their next phase of care seamlessly. Given the nationwide nursing shortage, aging population, challenges uncovered by the COVID-19 pandemic, rising hospital costs, nurse/provider burnout related to challenging work environments, and capacity constraints, a shift toward the combination of virtual and in-home care is imperative. The hospital-at-home model has been associated with superior patient outcomes, including reduced risks of delirium, improved functional status, improved patient and family member satisfaction, reduced mortality, reduced readmissions, and significantly lower costs.1 COVID-19 alone has unmasked major facility-based deficiencies and limitations of our health care system. While the pandemic is not the impetus for the hospital-at-home model, the extended stress of this event has created a unique opportunity to reimagine and transform our health care delivery system so that it is less fragmented and more flexible.
Nursing in the Model
Nursing is central to the hospital-at-home model. Virtual nurses provide meticulous care plan oversight, assessment, and documentation across in-home service providers, to ensure holistic, safe, transparent, and continuous progression toward care plan milestones. The virtual nurse monitors patients using in-home technology that is set up at the time of admission. Connecting with patients to verify social and medical needs, the virtual nurse advocates for their patients and uses these technologies to care and deploy on-demand hands-on services to the patient. Service providers such as paramedics, infusion nurses, or home health nurses may be deployed to provide services in the patient’s home. By bringing in supplies, therapeutics, and interdisciplinary team members, the capabilities of a brick-and-mortar hospital are replicated in the home. All actions that occur wherever the patient is receiving care are overseen by professional nursing staff; in short, virtual nurses are the equivalent of bedside nurses in the brick-and-mortar health care facilities.
Potential Benefits
There are many benefits to the hospital-at-home model (Table). This health care model can be particularly helpful for patients who require frequent admission to acute care facilities, and is well suited for patients with a range of conditions, including those with COVID-19, pneumonia, cellulitis, or congestive heart failure. This care model helps eliminate some of the stressors for patients who have chronic illnesses or other conditions that require frequent hospital admissions. Patients can independently recover at home and can also be surrounded by their loved ones and pets while recovering. This care approach additionally eliminates the risk of hospital-acquired infections and injuries. The hospital-at-home model allows for increased mobility,2 as patients are familiar with their surroundings, resulting in reduced onset of delirium. Additionally, patients with improved mobility performance are less likely to experience negative health outcomes.3 There is less chance of sleep disruption as the patient is sleeping in their own bed—no unfamiliar roommate, no call bells or health care personnel frequently coming into the room. The in-home technology set up for remote patient monitoring is designed with the user in mind. Ease of use empowers the patient to collaborate with their care team on their own terms and center the priorities of themselves and their families.

Positive Outcomes
The hospital-at-home model is associated with positive outcomes. The authors of a systematic review identified 10 randomized controlled trials of hospital-at-home programs (with a total of 1372 patients), but were able to obtain data for only 5 of these trials (with a total of 844 patients).4 They found a 38% reduction in 6-month mortality for patients who received hospital care at home, as well as significantly higher patient satisfaction across a range of medical conditions, including patients with cellulitis and community-acquired pneumonia, as well as elderly patients with multiple medical conditions. The authors concluded that hospital care at home was less expensive than admission to an acute care hospital.4 Similarly, a meta-analysis done by Caplan et al5 that included 61 randomized controlled trials concluded that hospital at home is associated with reductions in mortality, readmission rates, and cost, and increases in patient and caregiver satisfaction. Levine et al2 found reduced costs and utilization with home hospitalization compared to in-hospital care, as well as improved patient mobility status.
The home is the ideal place to empower patients and caregivers to engage in self-management.2 Receiving hospital care at home eliminates the need for dealing with transportation arrangements, traffic, road tolls, and time/scheduling constraints, or finding care for a dependent family member, some of the many stressors that may be experienced by patients who require frequent trips to the hospital. For patients who may not be clinically suitable candidates for hospital at home, such as those requiring critical care intervention and support, the brick-and-mortar hospital is still the appropriate site of care. The hospital-at-home model helps prevent bed shortages in brick-and-mortar hospital settings by allowing hospital care at home for patients who meet preset criteria. These patients can be hospitalized in alternative locations such as their own homes or the residence of a friend. This helps increase health system capacity as well as resiliency.
In addition to expanding safe and appropriate treatment spaces, the hospital-at-home model helps increase access to care for patients during nonstandard hours, including weekends, holidays, or when the waiting time in the emergency room is painfully long. Furthermore, providing care in the home gives the clinical team valuable insight into the patient’s daily life and routine. Performing medication reconciliation with the medicine cabinet in sight and dietary education in a patient’s kitchen are powerful touch points.2 For example, a patient with congestive heart failure who must undergo diuresis is much more likely to meet their care goals when their home diet is aligned with the treatment goal. By being able to see exactly what is in a patient’s pantry and fridge, the care team can create a much more tailored approach to sodium intake and fluid management. Providers can create and execute true patient-centric care as they gain direct insight into the patient’s lifestyle, which is clearly valuable when creating care plans for complex chronic health issues.
Challenges to Implementation and Scaling
Although there are clear benefits to hospital at home, how to best implement and scale this model presents a challenge. In addition to educating patients and families about this model of care, health care systems must expand their hospital-at-home programs and provide education about this model to clinical staff and trainees, and insurers must create reimbursement paradigms. Patients meeting eligibility criteria to enroll in hospital at home is the easiest hurdle, as hospital-at-home programs function best when they enroll and service as many patients as possible, including underserved populations.
Upfront Costs and Cost Savings
While there are upfront costs to set up technology and coordinate services, hospital at home also provides significant total cost savings when compared to coordination associated with brick-and-mortar admission. Hospital care accounts for about one-third of total medical expenditures and is a leading cause of debt.2 Eliminating fixed hospital costs such as facility, overhead, and equipment costs through adoption of the hospital-at-home model can lead to a reduction in expenditures. It has been found that fewer laboratory and diagnostic tests are ordered for hospital-at-home patients when compared to similar patients in brick-and-mortar hospital settings, with comparable or better clinical patient outcomes.6 Furthermore, it is estimated that there are cost savings of 19% to 30% when compared to traditional inpatient care.6 Without legislative action, upon the end of the current COVID-19 public health emergency, the Centers for Medicare & Medicaid Service’s Acute Hospital Care at Home waiver will terminate. This could slow down scaling of the model.However, over the past 2 years there has been enough buy-in from major health systems and patients to continue the momentum of the model’s growth. When setting up a hospital-at-home program, it would be wise to consider a few factors: where in the hospital or health system entity structure the hospital-at-home program will reside, which existing resources can be leveraged within the hospital or health system, and what are the state or federal regulatory requirements for such a program. This type of program continues to fill gaps within the US health care system, meeting the needs of widely overlooked populations and increasing access to essential ancillary services.
Conclusion
It is time to consider our bias toward hospital-first options when managing the care needs of our patients. Health care providers have the option to advocate for holistic care, better experience, and better outcomes. Home-based options are safe, equitable, and patient-centric. Increased costs, consumerism, and technology have pushed us to think about alternative approaches to patient care delivery, and the pandemic created a unique opportunity to see just how far the health care system could stretch itself with capacity constraints, insufficient resources, and staff shortages. In light of new possibilities, it is time to reimagine and transform our health care delivery system so that it is unified, seamless, cohesive, and flexible.
Corresponding author: Payal Sharma, DNP, MSN, RN, FNP-BC, CBN; [email protected].
Disclosures: None reported.
From Medically Home Group, Boston, MA.
Brick-and-mortar hospitals in the United States have historically been considered the dominant setting for providing care to patients. The coordination and delivery of care has previously been bound to physical hospitals largely because multidisciplinary services were only accessible in an individual location. While the fundamental make-up of these services remains unchanged, these services are now available in alternate settings. Some of these services include access to a patient care team, supplies, diagnostics, pharmacy, and advanced therapeutic interventions. Presently, the physical environment is becoming increasingly irrelevant as the core of what makes the traditional hospital—the professional staff, collaborative work processes, and the dynamics of the space—have all been translated into a modern digitally integrated environment. The elements necessary to providing safe, effective care in a physical hospital setting are now available in a patient’s home.
Impetus for the Model
As hospitals reconsider how and where they deliver patient care because of limited resources, the hospital-at-home model has gained significant momentum and interest. This model transforms a home into a hospital. The inpatient acute care episode is entirely substituted with an intensive at-home hospital admission enabled by technology, multidisciplinary teams, and ancillary services. Furthermore, patients requiring post-acute support can be transitioned to their next phase of care seamlessly. Given the nationwide nursing shortage, aging population, challenges uncovered by the COVID-19 pandemic, rising hospital costs, nurse/provider burnout related to challenging work environments, and capacity constraints, a shift toward the combination of virtual and in-home care is imperative. The hospital-at-home model has been associated with superior patient outcomes, including reduced risks of delirium, improved functional status, improved patient and family member satisfaction, reduced mortality, reduced readmissions, and significantly lower costs.1 COVID-19 alone has unmasked major facility-based deficiencies and limitations of our health care system. While the pandemic is not the impetus for the hospital-at-home model, the extended stress of this event has created a unique opportunity to reimagine and transform our health care delivery system so that it is less fragmented and more flexible.
Nursing in the Model
Nursing is central to the hospital-at-home model. Virtual nurses provide meticulous care plan oversight, assessment, and documentation across in-home service providers, to ensure holistic, safe, transparent, and continuous progression toward care plan milestones. The virtual nurse monitors patients using in-home technology that is set up at the time of admission. Connecting with patients to verify social and medical needs, the virtual nurse advocates for their patients and uses these technologies to care and deploy on-demand hands-on services to the patient. Service providers such as paramedics, infusion nurses, or home health nurses may be deployed to provide services in the patient’s home. By bringing in supplies, therapeutics, and interdisciplinary team members, the capabilities of a brick-and-mortar hospital are replicated in the home. All actions that occur wherever the patient is receiving care are overseen by professional nursing staff; in short, virtual nurses are the equivalent of bedside nurses in the brick-and-mortar health care facilities.
Potential Benefits
There are many benefits to the hospital-at-home model (Table). This health care model can be particularly helpful for patients who require frequent admission to acute care facilities, and is well suited for patients with a range of conditions, including those with COVID-19, pneumonia, cellulitis, or congestive heart failure. This care model helps eliminate some of the stressors for patients who have chronic illnesses or other conditions that require frequent hospital admissions. Patients can independently recover at home and can also be surrounded by their loved ones and pets while recovering. This care approach additionally eliminates the risk of hospital-acquired infections and injuries. The hospital-at-home model allows for increased mobility,2 as patients are familiar with their surroundings, resulting in reduced onset of delirium. Additionally, patients with improved mobility performance are less likely to experience negative health outcomes.3 There is less chance of sleep disruption as the patient is sleeping in their own bed—no unfamiliar roommate, no call bells or health care personnel frequently coming into the room. The in-home technology set up for remote patient monitoring is designed with the user in mind. Ease of use empowers the patient to collaborate with their care team on their own terms and center the priorities of themselves and their families.

Positive Outcomes
The hospital-at-home model is associated with positive outcomes. The authors of a systematic review identified 10 randomized controlled trials of hospital-at-home programs (with a total of 1372 patients), but were able to obtain data for only 5 of these trials (with a total of 844 patients).4 They found a 38% reduction in 6-month mortality for patients who received hospital care at home, as well as significantly higher patient satisfaction across a range of medical conditions, including patients with cellulitis and community-acquired pneumonia, as well as elderly patients with multiple medical conditions. The authors concluded that hospital care at home was less expensive than admission to an acute care hospital.4 Similarly, a meta-analysis done by Caplan et al5 that included 61 randomized controlled trials concluded that hospital at home is associated with reductions in mortality, readmission rates, and cost, and increases in patient and caregiver satisfaction. Levine et al2 found reduced costs and utilization with home hospitalization compared to in-hospital care, as well as improved patient mobility status.
The home is the ideal place to empower patients and caregivers to engage in self-management.2 Receiving hospital care at home eliminates the need for dealing with transportation arrangements, traffic, road tolls, and time/scheduling constraints, or finding care for a dependent family member, some of the many stressors that may be experienced by patients who require frequent trips to the hospital. For patients who may not be clinically suitable candidates for hospital at home, such as those requiring critical care intervention and support, the brick-and-mortar hospital is still the appropriate site of care. The hospital-at-home model helps prevent bed shortages in brick-and-mortar hospital settings by allowing hospital care at home for patients who meet preset criteria. These patients can be hospitalized in alternative locations such as their own homes or the residence of a friend. This helps increase health system capacity as well as resiliency.
In addition to expanding safe and appropriate treatment spaces, the hospital-at-home model helps increase access to care for patients during nonstandard hours, including weekends, holidays, or when the waiting time in the emergency room is painfully long. Furthermore, providing care in the home gives the clinical team valuable insight into the patient’s daily life and routine. Performing medication reconciliation with the medicine cabinet in sight and dietary education in a patient’s kitchen are powerful touch points.2 For example, a patient with congestive heart failure who must undergo diuresis is much more likely to meet their care goals when their home diet is aligned with the treatment goal. By being able to see exactly what is in a patient’s pantry and fridge, the care team can create a much more tailored approach to sodium intake and fluid management. Providers can create and execute true patient-centric care as they gain direct insight into the patient’s lifestyle, which is clearly valuable when creating care plans for complex chronic health issues.
Challenges to Implementation and Scaling
Although there are clear benefits to hospital at home, how to best implement and scale this model presents a challenge. In addition to educating patients and families about this model of care, health care systems must expand their hospital-at-home programs and provide education about this model to clinical staff and trainees, and insurers must create reimbursement paradigms. Patients meeting eligibility criteria to enroll in hospital at home is the easiest hurdle, as hospital-at-home programs function best when they enroll and service as many patients as possible, including underserved populations.
Upfront Costs and Cost Savings
While there are upfront costs to set up technology and coordinate services, hospital at home also provides significant total cost savings when compared to coordination associated with brick-and-mortar admission. Hospital care accounts for about one-third of total medical expenditures and is a leading cause of debt.2 Eliminating fixed hospital costs such as facility, overhead, and equipment costs through adoption of the hospital-at-home model can lead to a reduction in expenditures. It has been found that fewer laboratory and diagnostic tests are ordered for hospital-at-home patients when compared to similar patients in brick-and-mortar hospital settings, with comparable or better clinical patient outcomes.6 Furthermore, it is estimated that there are cost savings of 19% to 30% when compared to traditional inpatient care.6 Without legislative action, upon the end of the current COVID-19 public health emergency, the Centers for Medicare & Medicaid Service’s Acute Hospital Care at Home waiver will terminate. This could slow down scaling of the model.However, over the past 2 years there has been enough buy-in from major health systems and patients to continue the momentum of the model’s growth. When setting up a hospital-at-home program, it would be wise to consider a few factors: where in the hospital or health system entity structure the hospital-at-home program will reside, which existing resources can be leveraged within the hospital or health system, and what are the state or federal regulatory requirements for such a program. This type of program continues to fill gaps within the US health care system, meeting the needs of widely overlooked populations and increasing access to essential ancillary services.
Conclusion
It is time to consider our bias toward hospital-first options when managing the care needs of our patients. Health care providers have the option to advocate for holistic care, better experience, and better outcomes. Home-based options are safe, equitable, and patient-centric. Increased costs, consumerism, and technology have pushed us to think about alternative approaches to patient care delivery, and the pandemic created a unique opportunity to see just how far the health care system could stretch itself with capacity constraints, insufficient resources, and staff shortages. In light of new possibilities, it is time to reimagine and transform our health care delivery system so that it is unified, seamless, cohesive, and flexible.
Corresponding author: Payal Sharma, DNP, MSN, RN, FNP-BC, CBN; [email protected].
Disclosures: None reported.
1. Cai S, Laurel PA, Makineni R, Marks ML. Evaluation of a hospital-in-home program implemented among veterans. Am J Manag Care. 2017;23(8):482-487.
2. Levine DM, Ouchi K, Blanchfield B, et al. Hospital-level care at home for acutely ill adults: a pilot randomized controlled trial. J Gen Intern Med. 2018;33(5):729-736. doi:10.1007/s11606-018-4307-z
3. Shuman V, Coyle PC, Perera S,et al. Association between improved mobility and distal health outcomes. J Gerontol A Biol Sci Med Sci. 2020;75(12):2412-2417. doi:10.1093/gerona/glaa086
4. Shepperd S, Doll H, Angus RM, et al. Avoiding hospital admission through provision of hospital care at home: a systematic review and meta-analysis of individual patient data. CMAJ. 2009;180(2):175-182. doi:10.1503/cmaj.081491
5. Caplan GA, Sulaiman NS, Mangin DA, et al. A meta-analysis of “hospital in the home”. Med J Aust. 2012;197(9):512-519. doi:10.5694/mja12.10480
6. Hospital at Home. Johns Hopkins Medicine. Healthcare Solutions. Accessed May 20, 2022. https://www.johnshopkinssolutions.com/solution/hospital-at-home/
1. Cai S, Laurel PA, Makineni R, Marks ML. Evaluation of a hospital-in-home program implemented among veterans. Am J Manag Care. 2017;23(8):482-487.
2. Levine DM, Ouchi K, Blanchfield B, et al. Hospital-level care at home for acutely ill adults: a pilot randomized controlled trial. J Gen Intern Med. 2018;33(5):729-736. doi:10.1007/s11606-018-4307-z
3. Shuman V, Coyle PC, Perera S,et al. Association between improved mobility and distal health outcomes. J Gerontol A Biol Sci Med Sci. 2020;75(12):2412-2417. doi:10.1093/gerona/glaa086
4. Shepperd S, Doll H, Angus RM, et al. Avoiding hospital admission through provision of hospital care at home: a systematic review and meta-analysis of individual patient data. CMAJ. 2009;180(2):175-182. doi:10.1503/cmaj.081491
5. Caplan GA, Sulaiman NS, Mangin DA, et al. A meta-analysis of “hospital in the home”. Med J Aust. 2012;197(9):512-519. doi:10.5694/mja12.10480
6. Hospital at Home. Johns Hopkins Medicine. Healthcare Solutions. Accessed May 20, 2022. https://www.johnshopkinssolutions.com/solution/hospital-at-home/
The Intersection of Clinical Quality Improvement Research and Implementation Science
The Institute of Medicine brought much-needed attention to the need for process improvement in medicine with its seminal report To Err Is Human: Building a Safer Health System, which was issued in 1999, leading to the quality movement’s call to close health care performance gaps in Crossing the Quality Chasm: A New Health System for the 21st Century.1,2 Quality improvement science in medicine has evolved over the past 2 decades to include a broad spectrum of approaches, from agile improvement to continuous learning and improvement. Current efforts focus on Lean-based process improvement along with a reduction in variation in clinical practice to align practice with the principles of evidence-based medicine in a patient-centered approach.3 Further, the definition of quality improvement under the Affordable Care Act was framed as an equitable, timely, value-based, patient-centered approach to achieving population-level health goals.4 Thus, the science of quality improvement drives the core principles of care delivery improvement, and the rigorous evidence needed to expand innovation is embedded within the same framework.5,6 In clinical practice, quality improvement projects aim to define gaps and then specific steps are undertaken to improve the evidence-based practice of a specific process. The overarching goal is to enhance the efficacy of the practice by reducing waste within a particular domain. Thus, quality improvement and implementation research eventually unify how clinical practice is advanced concurrently to bridge identified gaps.7
System redesign through a patient-centered framework forms the core of an overarching strategy to support system-level processes. Both require a deep understanding of the fields of quality improvement science and implementation science.8 Furthermore, aligning clinical research needs, system aims, patients’ values, and clinical care give the new design a clear path forward. Patient-centered improvement includes the essential elements of system redesign around human factors, including communication, physical resources, and updated information during episodes of care. The patient-centered improvement design is juxtaposed with care planning and establishing continuum of care processes.9 It is essential to note that safety is rooted within the quality domain as a top priority in medicine.10 The best implementation methods and approaches are discussed and debated, and the improvement progress continues on multiple fronts.11 Patient safety systems are implemented simultaneously during the redesign phase. Moreover, identifying and testing the health care delivery methods in the era of competing strategic priorities to achieve the desirable clinical outcomes highlights the importance of implementation, while contemplating the methods of dissemination, scalability, and sustainability of the best evidence-based clinical practice.
The cycle of quality improvement research completes the system implementation efforts. The conceptual framework of quality improvement includes multiple areas of care and transition, along with applying the best clinical practices in a culture that emphasizes continuous improvement and learning. At the same time, the operating principles should include continuous improvement in a simple and continuous system of learning as a core concept. Our proposed implementation approach involves taking simple and practical steps while separating the process from the outcomes measures, extracting effectiveness throughout the process. It is essential to keep in mind that building a proactive and systematic improvement environment requires a framework for safety, reliability, and effective care, as well as the alignment of the physical system, communication, and professional environment and culture (Figure).
In summary, system design for quality improvement research should incorporate the principles and conceptual framework that embody effective implementation strategies, with a focus on operational and practical steps. Continuous improvement will be reached through the multidimensional development of current health care system metrics and the incorporation of implementation science methods.
Corresponding author: Ebrahim Barkoudah, MD, MPH, Department of Medicine, Brigham and Women’s Hospital, Boston, MA; [email protected]
Disclosures: None reported.
1. Institute of Medicine (US) Committee on Quality of Health Care in America. To Err is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, editors. Washington (DC): National Academies Press (US); 2000.
2. Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington (DC): National Academies Press (US); 2001.
3. Berwick DM. The science of improvement. JAMA. 2008;299(10):1182-1184. doi:10.1001/jama.299.10.1182
4. Mazurenko O, Balio CP, Agarwal R, Carroll AE, Menachemi N. The effects of Medicaid expansion under the ACA: a systematic review. Health Affairs. 2018;37(6):944-950. doi: 10.1377/hlthaff.2017.1491
5. Fan E, Needham DM. The science of quality improvement. JAMA. 2008;300(4):390-391. doi:10.1001/jama.300.4.390-b
6. Alexander JA, Hearld LR. The science of quality improvement implementation: developing capacity to make a difference. Med Care. 2011:S6-20. doi:10.1097/MLR.0b013e3181e1709c
7. Rohweder C, Wangen M, Black M, et al. Understanding quality improvement collaboratives through an implementation science lens. Prev Med. 2019;129:105859. doi: 10.1016/j.ypmed.2019.105859
8. Bergeson SC, Dean JD. A systems approach to patient-centered care. JAMA. 2006;296(23):2848-2851. doi:10.1001/jama.296.23.2848
9. Leonard M, Graham S, Bonacum D. The human factor: the critical importance of effective teamwork and communication in providing safe care. Qual Saf Health Care. 2004;13 Suppl 1(Suppl 1):i85-90. doi:10.1136/qhc.13.suppl_1.i85
10. Leape LL, Berwick DM, Bates DW. What practices will most improve safety? Evidence-based medicine meets patient safety. JAMA. 2002;288(4):501-507. doi:10.1001/jama.288.4.501
11. Auerbach AD, Landefeld CS, Shojania KG. The tension between needing to improve care and knowing how to do it. N Engl J Med. 2007;357(6):608-613. doi:10.1056/NEJMsb070738
The Institute of Medicine brought much-needed attention to the need for process improvement in medicine with its seminal report To Err Is Human: Building a Safer Health System, which was issued in 1999, leading to the quality movement’s call to close health care performance gaps in Crossing the Quality Chasm: A New Health System for the 21st Century.1,2 Quality improvement science in medicine has evolved over the past 2 decades to include a broad spectrum of approaches, from agile improvement to continuous learning and improvement. Current efforts focus on Lean-based process improvement along with a reduction in variation in clinical practice to align practice with the principles of evidence-based medicine in a patient-centered approach.3 Further, the definition of quality improvement under the Affordable Care Act was framed as an equitable, timely, value-based, patient-centered approach to achieving population-level health goals.4 Thus, the science of quality improvement drives the core principles of care delivery improvement, and the rigorous evidence needed to expand innovation is embedded within the same framework.5,6 In clinical practice, quality improvement projects aim to define gaps and then specific steps are undertaken to improve the evidence-based practice of a specific process. The overarching goal is to enhance the efficacy of the practice by reducing waste within a particular domain. Thus, quality improvement and implementation research eventually unify how clinical practice is advanced concurrently to bridge identified gaps.7
System redesign through a patient-centered framework forms the core of an overarching strategy to support system-level processes. Both require a deep understanding of the fields of quality improvement science and implementation science.8 Furthermore, aligning clinical research needs, system aims, patients’ values, and clinical care give the new design a clear path forward. Patient-centered improvement includes the essential elements of system redesign around human factors, including communication, physical resources, and updated information during episodes of care. The patient-centered improvement design is juxtaposed with care planning and establishing continuum of care processes.9 It is essential to note that safety is rooted within the quality domain as a top priority in medicine.10 The best implementation methods and approaches are discussed and debated, and the improvement progress continues on multiple fronts.11 Patient safety systems are implemented simultaneously during the redesign phase. Moreover, identifying and testing the health care delivery methods in the era of competing strategic priorities to achieve the desirable clinical outcomes highlights the importance of implementation, while contemplating the methods of dissemination, scalability, and sustainability of the best evidence-based clinical practice.
The cycle of quality improvement research completes the system implementation efforts. The conceptual framework of quality improvement includes multiple areas of care and transition, along with applying the best clinical practices in a culture that emphasizes continuous improvement and learning. At the same time, the operating principles should include continuous improvement in a simple and continuous system of learning as a core concept. Our proposed implementation approach involves taking simple and practical steps while separating the process from the outcomes measures, extracting effectiveness throughout the process. It is essential to keep in mind that building a proactive and systematic improvement environment requires a framework for safety, reliability, and effective care, as well as the alignment of the physical system, communication, and professional environment and culture (Figure).

In summary, system design for quality improvement research should incorporate the principles and conceptual framework that embody effective implementation strategies, with a focus on operational and practical steps. Continuous improvement will be reached through the multidimensional development of current health care system metrics and the incorporation of implementation science methods.
Corresponding author: Ebrahim Barkoudah, MD, MPH, Department of Medicine, Brigham and Women’s Hospital, Boston, MA; [email protected]
Disclosures: None reported.
The Institute of Medicine brought much-needed attention to the need for process improvement in medicine with its seminal report To Err Is Human: Building a Safer Health System, which was issued in 1999, leading to the quality movement’s call to close health care performance gaps in Crossing the Quality Chasm: A New Health System for the 21st Century.1,2 Quality improvement science in medicine has evolved over the past 2 decades to include a broad spectrum of approaches, from agile improvement to continuous learning and improvement. Current efforts focus on Lean-based process improvement along with a reduction in variation in clinical practice to align practice with the principles of evidence-based medicine in a patient-centered approach.3 Further, the definition of quality improvement under the Affordable Care Act was framed as an equitable, timely, value-based, patient-centered approach to achieving population-level health goals.4 Thus, the science of quality improvement drives the core principles of care delivery improvement, and the rigorous evidence needed to expand innovation is embedded within the same framework.5,6 In clinical practice, quality improvement projects aim to define gaps and then specific steps are undertaken to improve the evidence-based practice of a specific process. The overarching goal is to enhance the efficacy of the practice by reducing waste within a particular domain. Thus, quality improvement and implementation research eventually unify how clinical practice is advanced concurrently to bridge identified gaps.7
System redesign through a patient-centered framework forms the core of an overarching strategy to support system-level processes. Both require a deep understanding of the fields of quality improvement science and implementation science.8 Furthermore, aligning clinical research needs, system aims, patients’ values, and clinical care give the new design a clear path forward. Patient-centered improvement includes the essential elements of system redesign around human factors, including communication, physical resources, and updated information during episodes of care. The patient-centered improvement design is juxtaposed with care planning and establishing continuum of care processes.9 It is essential to note that safety is rooted within the quality domain as a top priority in medicine.10 The best implementation methods and approaches are discussed and debated, and the improvement progress continues on multiple fronts.11 Patient safety systems are implemented simultaneously during the redesign phase. Moreover, identifying and testing the health care delivery methods in the era of competing strategic priorities to achieve the desirable clinical outcomes highlights the importance of implementation, while contemplating the methods of dissemination, scalability, and sustainability of the best evidence-based clinical practice.
The cycle of quality improvement research completes the system implementation efforts. The conceptual framework of quality improvement includes multiple areas of care and transition, along with applying the best clinical practices in a culture that emphasizes continuous improvement and learning. At the same time, the operating principles should include continuous improvement in a simple and continuous system of learning as a core concept. Our proposed implementation approach involves taking simple and practical steps while separating the process from the outcomes measures, extracting effectiveness throughout the process. It is essential to keep in mind that building a proactive and systematic improvement environment requires a framework for safety, reliability, and effective care, as well as the alignment of the physical system, communication, and professional environment and culture (Figure).

In summary, system design for quality improvement research should incorporate the principles and conceptual framework that embody effective implementation strategies, with a focus on operational and practical steps. Continuous improvement will be reached through the multidimensional development of current health care system metrics and the incorporation of implementation science methods.
Corresponding author: Ebrahim Barkoudah, MD, MPH, Department of Medicine, Brigham and Women’s Hospital, Boston, MA; [email protected]
Disclosures: None reported.
1. Institute of Medicine (US) Committee on Quality of Health Care in America. To Err is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, editors. Washington (DC): National Academies Press (US); 2000.
2. Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington (DC): National Academies Press (US); 2001.
3. Berwick DM. The science of improvement. JAMA. 2008;299(10):1182-1184. doi:10.1001/jama.299.10.1182
4. Mazurenko O, Balio CP, Agarwal R, Carroll AE, Menachemi N. The effects of Medicaid expansion under the ACA: a systematic review. Health Affairs. 2018;37(6):944-950. doi: 10.1377/hlthaff.2017.1491
5. Fan E, Needham DM. The science of quality improvement. JAMA. 2008;300(4):390-391. doi:10.1001/jama.300.4.390-b
6. Alexander JA, Hearld LR. The science of quality improvement implementation: developing capacity to make a difference. Med Care. 2011:S6-20. doi:10.1097/MLR.0b013e3181e1709c
7. Rohweder C, Wangen M, Black M, et al. Understanding quality improvement collaboratives through an implementation science lens. Prev Med. 2019;129:105859. doi: 10.1016/j.ypmed.2019.105859
8. Bergeson SC, Dean JD. A systems approach to patient-centered care. JAMA. 2006;296(23):2848-2851. doi:10.1001/jama.296.23.2848
9. Leonard M, Graham S, Bonacum D. The human factor: the critical importance of effective teamwork and communication in providing safe care. Qual Saf Health Care. 2004;13 Suppl 1(Suppl 1):i85-90. doi:10.1136/qhc.13.suppl_1.i85
10. Leape LL, Berwick DM, Bates DW. What practices will most improve safety? Evidence-based medicine meets patient safety. JAMA. 2002;288(4):501-507. doi:10.1001/jama.288.4.501
11. Auerbach AD, Landefeld CS, Shojania KG. The tension between needing to improve care and knowing how to do it. N Engl J Med. 2007;357(6):608-613. doi:10.1056/NEJMsb070738
1. Institute of Medicine (US) Committee on Quality of Health Care in America. To Err is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, editors. Washington (DC): National Academies Press (US); 2000.
2. Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington (DC): National Academies Press (US); 2001.
3. Berwick DM. The science of improvement. JAMA. 2008;299(10):1182-1184. doi:10.1001/jama.299.10.1182
4. Mazurenko O, Balio CP, Agarwal R, Carroll AE, Menachemi N. The effects of Medicaid expansion under the ACA: a systematic review. Health Affairs. 2018;37(6):944-950. doi: 10.1377/hlthaff.2017.1491
5. Fan E, Needham DM. The science of quality improvement. JAMA. 2008;300(4):390-391. doi:10.1001/jama.300.4.390-b
6. Alexander JA, Hearld LR. The science of quality improvement implementation: developing capacity to make a difference. Med Care. 2011:S6-20. doi:10.1097/MLR.0b013e3181e1709c
7. Rohweder C, Wangen M, Black M, et al. Understanding quality improvement collaboratives through an implementation science lens. Prev Med. 2019;129:105859. doi: 10.1016/j.ypmed.2019.105859
8. Bergeson SC, Dean JD. A systems approach to patient-centered care. JAMA. 2006;296(23):2848-2851. doi:10.1001/jama.296.23.2848
9. Leonard M, Graham S, Bonacum D. The human factor: the critical importance of effective teamwork and communication in providing safe care. Qual Saf Health Care. 2004;13 Suppl 1(Suppl 1):i85-90. doi:10.1136/qhc.13.suppl_1.i85
10. Leape LL, Berwick DM, Bates DW. What practices will most improve safety? Evidence-based medicine meets patient safety. JAMA. 2002;288(4):501-507. doi:10.1001/jama.288.4.501
11. Auerbach AD, Landefeld CS, Shojania KG. The tension between needing to improve care and knowing how to do it. N Engl J Med. 2007;357(6):608-613. doi:10.1056/NEJMsb070738
A Quantification Method to Compare the Value of Surgery and Palliative Care in Patients With Complex Cardiac Disease: A Concept
From the Department of Cardiothoracic Surgery, Stanford University, Stanford, CA.
Abstract
Complex cardiac patients are often referred for surgery or palliative care based on the risk of perioperative mortality. This decision ignores factors such as quality of life or duration of life in either surgery or the palliative path. Here, we propose a model to numerically assess and compare the value of surgery vs palliation. This model includes quality and duration of life, as well as risk of perioperative mortality, and involves a patient’s preferences in the decision-making process.
For each pathway, surgery or palliative care, a value is calculated and compared to a normal life value (no disease symptoms and normal life expectancy). The formula is adjusted for the risk of operative mortality. The model produces a ratio of the value of surgery to the value of palliative care that signifies the superiority of one or another. This model calculation presents an objective estimated numerical value to compare the value of surgery and palliative care. It can be applied to every decision-making process before surgery. In general, if a procedure has the potential to significantly extend life in a patient who otherwise has a very short life expectancy with palliation only, performing high-risk surgery would be a reasonable option. A model that provides a numerical value for surgery vs palliative care and includes quality and duration of life in each pathway could be a useful tool for cardiac surgeons in decision making regarding high-risk surgery.
Keywords: high-risk surgery, palliative care, quality of life, life expectancy.
Patients with complex cardiovascular disease are occasionally considered inoperable due to the high risk of surgical mortality. When the risk of perioperative mortality (POM) is predicted to be too high, surgical intervention is denied, and patients are often referred to palliative care. The risk of POM in cardiac surgery is often calculated using large-scale databases, such as the Society of Thoracic Surgeons (STS) records. The STS risk models, which are regularly updated, are based on large data sets and incorporate precise statistical methods for risk adjustment.1 In general, these calculators provide a percentage value that defines the magnitude of the risk of death, and then an arbitrary range is selected to categorize the procedure as low, medium, or high risk or inoperable status. The STS database does not set a cutoff point or range to define “operability.” Assigning inoperable status to a certain risk rate is problematic, with many ethical, legal, and moral implications, and for this reason, it has mostly remained undefined. In contrast, the low- and medium-risk ranges are easier to define. Another limitation encountered in the STS database is the lack of risk data for less common but very high-risk procedures, such as a triple valve replacement.
A common example where risk classification has been defined is in patients who are candidates for surgical vs transcatheter aortic valve replacement. Some groups have described a risk of <4% as low risk, 4% to 8% as intermediate risk, >8% as high risk, and >15% as inoperable2; for some other groups, a risk of POM >50% is considered extreme risk or inoperable.3,4 This procedure-specific classification is a useful decision-making tool and helps the surgeon perform an initial risk assessment to allocate a specific patient to a group—operable or nonoperable—only by calculating the risk of surgical death. However, this allocation method does not provide any information on how and when death occurs in either group. These 2 parameters of how and when death occurs define the quality of life (QOL) and the duration of life (DOL), respectively, and together could be considered as the value of life in each pathway. A survivor of a high-risk surgery may benefit from good quality and extended life (a high value), or, on the other end of the spectrum, a high-risk patient who does not undergo surgery is spared the mortality risk of the surgery but dies sooner (low value) with symptoms due to the natural course of the untreated disease.
The central question is, if a surgery is high risk but has the potential of providing a good value (for those who survive it), what QOL and DOL values are acceptable to risk or to justify accepting and proceeding with a risky surgery? Or how high a POM risk is justified to proceed with surgery rather than the alternative palliative care with a certain quality and duration? It is obvious that a decision-making process that is based on POM cannot compare the value of surgery (Vs) and the value of palliation (Vp). Furthermore, it ignores patient preferences and their input, as these are excluded from this decision-making process.
To be able to include QOL and DOL in any decision making, one must precisely describe these parameters. Both QOL and DOL are used for estimation of disease burden by health care administrators, public health experts, insurance agencies, and others. Multiple models have been proposed and used to estimate the overall burden of the disease. Most of the models for this purpose are created for large-scale economic purposes and not for decision making in individual cases.
An important measure is the quality-adjusted life year (QALY). This is an important parameter since it includes both measures of quality and quantity of life.5,6 QALY is a simplified measure to assess the value of health outcomes, and it has been used in economic calculations to assess mainly the cost-effectiveness of various interventions. We sought to evaluate the utility of a similar method in adding further insight into the surgical decision-making process. In this article, we propose a simple model to compare the value of surgery vs palliative care, similar to QALY. This model includes and adjusts for the quality and the quantity of life, in addition to the risk of POM, in the decision-making process for high-risk patients.
The Model
The 2 decision pathways, surgery and palliative care, are compared for their value. We define the value as the product of QOL and DOL in each pathway and use the severity of the symptoms as a surrogate for QOL. If duration and quality were depicted on the x and y axes of a graph (Figure 1), then the area under the curve would represent the collective value in each situation. Figure 2 shows the timeline and the different pathways with each decision. The value in each situation is calculated in relation to the full value, which is represented as the value of normal life (Vn), that is, life without disease and with normal life expectancy. The values of each decision pathway, the value of surgery (Vs) and the value of palliation (Vp), are then compared to define the benefit for each decision as follows:
If Vs/Vp > 1, the benefit is toward surgery;
If Vs/Vp < 1, the benefit is for palliative care.
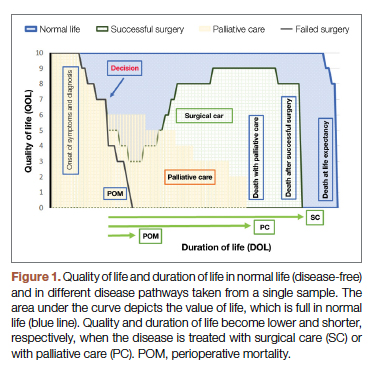
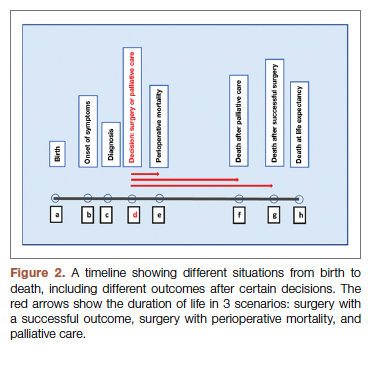
Definitions
Both quality and duration of life are presented on a 1-10 scale, 1 being the lowest and 10 the highest value, to yield a product with a value of 100 in normal, disease-free life. Any lower value is presented as a percentage to represent the comparison to the full value. QOL is determined by degradation of full quality with the average level of symptoms. DOL is calculated as a lost time (

For the DOL under any condition, a 10-year survival rate could be used as a surrogate in this formula. Compared to life expectancy value, using the 10-year survival rate simplifies the calculation since cardiac diseases are more prevalent in older age, close to or beyond the average life expectancy value.
Using the time intervals from the timeline in Figure 2:
dh = time interval from diagnosis to death at life expectancy
dg = time interval from diagnosis to death after successful surgery
df = time interval from diagnosis to death after palliative care
Duration for palliative care:
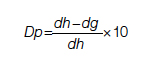
Duration for surgery:
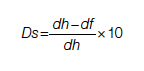
Adjustment: This value is calculated for those who survive the surgery. To adjust for the POM, it is multiplied by the 100 − POM risk.
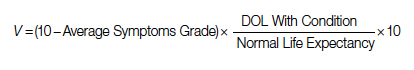
Since value is the base for comparison in this model, and it is the product of 2 equally important factors in the formula (
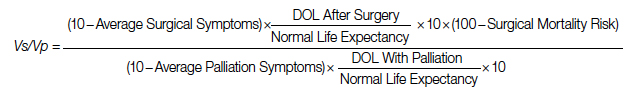
After elimination of normal life expectancy, form the numerator and denominator:
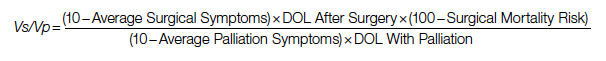
To adjust for surgical outcomes in special circumstances where less than optimal or standard surgical results are expected (eg, in very rare surgeries, limited resource institutions, or suboptimal postoperative surgical care), an optional coefficient R can be added to the numerator (surgical value). This optional coefficient, with values such as 0.8, 0.9 (to degrade the value of surgery) or 1 (standard surgical outcome), adjusts for variability in interinstitutional surgical results or surgeon variability. No coefficient is added to the denominator since palliative care provides minimal differences between clinicians and hospitals. Thus, the final adjusted formula would be as follows:
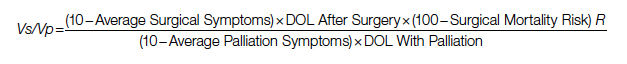
Example
A 60-year-old patient with a 10% POM risk needs to be allocated to surgical or palliative care. With palliative care, if this patient lived 6 years with average symptoms grade 4, the Vp would be 20; that is, 20% of the normal life value (if he lived 18 years instead without the disease).
Using the formula for calculation of value in each pathway:
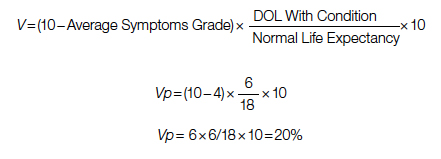
If the same patient undergoes a surgery with a 10% risk of POM, with an average grade 2 related to surgical recovery symptoms for 1 year and then is symptom-free and lives 12 years (instead of 18 years [life expectancy]), his Vs would be 53, or 53% out of the normal life value that is saved if the surgery is 100% successful; adjusted Vs with (chance of survival of 90%) would be 53 × 90% = 48%.
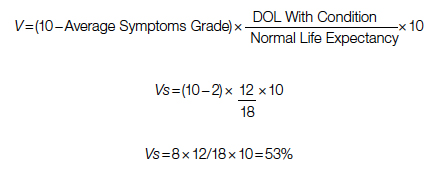
With adjustment of 90% survival chance in surgery, 53 × 90% = 48%. In this example, Vs/Vp = 48/20 = 2.4, showing a significant benefit for surgical care. Notably, the unknown value of normal life expectancy is not needed for the calculation of Vs/Vp, since it is the same in both pathways and it is eliminated by calculation in fraction.
Based on this formula, since the duration of surgical symptoms is short, no matter how severe these are, if the potential duration of life after surgery is high (represented by smaller area under the curve in Figure 1), the numerator becomes larger and the value of the surgery grows. For example, if a patient with a 15% risk of POM, which is generally considered inoperable, lives 5 years, as opposed to 2 years with palliative care with mild symptoms (eg 3/10), Vs/Vp would be 2.7, still showing a significant benefit for surgical care.
Discussion
Any surgical intervention is offered with 2 goals in mind, improving QOL and extending DOL. In a high-risk patient, surgery might be declined due to a high risk of POM, and the patient is offered palliative care, which other than providing symptom relief does not change the course of disease and eventually the patient will die due to the untreated disease. In this decision-making method, mostly completed by a care team only, a potential risk of death due to surgery which possibly could cure the patient is traded for immediate survival; however, the symptomatic course ensues until death. This mostly unilateral decision-making process by a care team, which incorporates minimal input from the patient or ignores patient preferences altogether, is based only on POM risk, and roughly includes a single parameter: years of potential life lost (YPLL). YPLL is a measure of premature mortality, and in the setting of surgical intervention, YPLL is the number of years a patient would lose unless a successful surgery were undertaken. Obviously, patients would live longer if a surgery that was intended to save them failed.
In this article, we proposed a simple method to quantify each decision to decide whether to operate or choose surgical care vs palliative care. Since quality and duration of life are both end factors clinicians and patients aspire to in each decision, they can be considered together as the value of each decision. We believe a numerical framework would provide an objective way to assist both the patient at high risk and the care team in the decision-making process.
The 2 parameters we consider are DOL and QOL. DOL, or survival, can be extracted from large-scale data using statistical methods that have been developed to predict survival under various conditions, such as Kaplan-Meier curves. These methods present the chance of survival in percentages in a defined time frame, such as a 5- or 10-year period.
While the DOL is a numerical parameter and quantifiable, the QOL is a more complex entity. This subjective parameter bears multiple definitions, aspects, and categories, and therefore multiple scales for quantification of QOL have been proposed. These scales have been used extensively for the purpose of health determination in health care policy and economic planning. Most scales acknowledge that QOL is multifactorial and includes interrelated aspects such as mental and socioeconomic factors. We have also noticed that QOL is better determined by the palliative care team than surgeons, so including these care providers in the decision-making process might reduce surgeon bias.
Since our purpose here is only to assist with the decision on medical intervention, we focus on physical QOL. Multiple scales are used to assess health-related QOL, such as the Assessment of Quality of Life (AQoL)-8D,7 EuroQol-5 Dimension (EQ-5D),8 15D,9 and the 36-Item Short Form Survey (SF-36).10 These complex scales are built for systematic reviews, and they are not practical for a clinical user. To simplify and keep this practical, we define QOL by using the severity or grade of symptoms related to the disease the patient has on a scale of 0 to 10. The severity of symptoms can be easily determined using available scales. An applicable scale for this purpose is the Edmonton Symptom Assessment Scale (ESAS), which has been in use for years and has evolved as a useful tool in the medical field.11
Once DOL and QOL are determined on a 1-10 scale, the multiplied value then provides a product that we consider a value. The highest value hoped for in each decision is the achievement of the best QOL and DOL, a value of 100. In Figure 1, a graphic presentation of value in each decision is best seen as the area under the curve. As shown, a successful surgery, even when accompanied by significant symptoms during initial recovery, has a chance (100 – risk of POM%) to gain a larger area under curve (value) by achieving a longer life with no or fewer symptoms. However, in palliative care, progressing disease and even palliated symptoms with a shorter life expectancy impose a large burden on the patient and a much lower value. Note that in this calculation, life expectancy, which is an important but unpredictable factor, is initially included; however, by ratio comparison, it is eliminated, simplifying the calculation further.
Using this formula in different settings reveals that high-risk surgery has a greater potential to reduce YPLL in the general population. Based on this formula, compared to a surgery with potential to significantly extend DOL, a definite shorter and symptomatic life course with palliative care makes it a significantly less favorable option. In fact, in the cardiovascular field, palliative care has minimal or no effect on natural history, as the mechanism of illness is mechanical, such as occlusion of coronary arteries or valve dysfunction, leading eventually to heart failure and death. In a study by Xu et al, although palliative care reduced readmission rates and improved symptoms on a variety of scales, there was no effect on mortality and QOL in patients with heart failure.12
No model in this field has proven to be ideal, and this model bears multiple limitations as well. We have used severity of symptoms as a surrogate for QOL based on the fact that cardiac patients with different pathologies who are untreated will have a common final pathway with development of heart failure symptoms that dictate their QOL. Also, grading QOL is a difficult task at times. Even a model such as QALY, which is one of the most used, is not a perfect model and is not free of problems.6 The difference in surgical results and life expectancy between sexes and ethnic groups might be a source of bias in this formula. Also, multiple factors directly and indirectly affect QOL and DOL and create inaccuracies; therefore, making an exact science from an inexact one naturally relies on multiple assumptions. Although it has previously been shown that most POM occurs in a short period of time after cardiac surgery,13 long-term complications that potentially degrade QOL are not included in this model. By applying this model, one must assume indefinite economic resources. Moreover, applying a single mathematical model in a biologic system and in the general population has intrinsic shortcomings, and it must overlook many other factors (eg, ethical, legal). For example, it will be hard to justify a failed surgery with 15% risk of POM undertaken to eliminate the severe long-lasting symptoms of a disease, while the outcome of a successful surgery with a 20% risk of POM that adds life and quality would be ignored in the current health care system. Thus, regardless of the significant potential, most surgeons would waive a surgery based solely on the percentage rate of POM, perhaps using other terms such as ”peri-nonoperative mortality.”
Conclusion
We have proposed a simple and practical formula for decision making regarding surgical vs palliative care in high-risk patients. By assigning a value that is composed of QOL and DOL in each pathway and including the risk of POM, a ratio of values provides a numerical estimation that can be used to show preference over a specific decision. An advantage of this formula, in addition to presenting an arithmetic value that is easier to understand, is that it can be used in shared decision making with patients. We emphasize that this model is only a preliminary concept at this time and has not been tested or validated for clinical use. Validation of such a model will require extensive work and testing within a large-scale population. We hope that this article will serve as a starting point for the development of other models, and that this formula will become more sophisticated with fewer limitations through larger multidisciplinary efforts in the future.
Corresponding author: Rabin Gerrah, MD, Good Samaritan Regional Medical Center, 3640 NW Samaritan Drive, Suite 100B, Corvallis, OR 97330; [email protected].
Disclosures: None reported.
1. O’Brien SM, Feng L, He X, et al. The Society of Thoracic Surgeons 2018 Adult Cardiac Surgery Risk Models: Part 2-statistical methods and results. Ann Thorac Surg. 2018;105(5):1419-1428. doi: 10.1016/j.athoracsur.2018.03.003
2. Hurtado Rendón IS, Bittenbender P, Dunn JM, Firstenberg MS. Chapter 8: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines. In: Transcatheter Heart Valve Handbook: A Surgeons’ and Interventional Council Review. Akron City Hospital, Summa Health System, Akron, OH.
3. Herrmann HC, Thourani VH, Kodali SK, et al; PARTNER Investigators. One-year clinical outcomes with SAPIEN 3 transcatheter aortic valve replacement in high-risk and inoperable patients with severe aortic stenosis. Circulation. 2016;134:130-140. doi:10.1161/CIRCULATIONAHA
4. Ho C, Argáez C. Transcatheter Aortic Valve Implantation for Patients with Severe Aortic Stenosis at Various Levels of Surgical Risk: A Review of Clinical Effectiveness. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; March 19, 2018.
5. Rios-Diaz AJ, Lam J, Ramos MS, et al. Global patterns of QALY and DALY use in surgical cost-utility analyses: a systematic review. PLoS One. 2016:10;11:e0148304. doi:10.1371/journal.pone.0148304
6. Prieto L, Sacristán JA. Health, Problems and solutions in calculating quality-adjusted life years (QALYs). Qual Life Outcomes. 2003:19;1:80.
7. Centre for Health Economics. Assessment of Quality of Life. 2014. Accessed May 13, 2022. http://www.aqol.com.au/
8. EuroQol Research Foundation. EQ-5D. Accessed May 13, 2022. https://euroqol.org/
9. 15D Instrument. Accessed May 13, 2022. http://www.15d-instrument.net/15d/
10. Rand Corporation. 36-Item Short Form Survey (SF-36).Accessed May 12, 2022. https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form.html
11. Hui D, Bruera E. The Edmonton Symptom Assessment System 25 years later: past, present, and future developments. J Pain Symptom Manage. 2017:53:630-643. doi:10.1016/j.jpainsymman.2016
12. Xu Z, Chen L, Jin S, Yang B, Chen X, Wu Z. Effect of palliative care for patients with heart failure. Int Heart J. 2018:30;59:503-509. doi:10.1536/ihj.17-289
13. Mazzeffi M, Zivot J, Buchman T, Halkos M. In-hospital mortality after cardiac surgery: patient characteristics, timing, and association with postoperative length of intensive care unit and hospital stay. Ann Thorac Surg. 2014;97:1220-1225. doi:10.1016/j.athoracsur.2013.10.040
From the Department of Cardiothoracic Surgery, Stanford University, Stanford, CA.
Abstract
Complex cardiac patients are often referred for surgery or palliative care based on the risk of perioperative mortality. This decision ignores factors such as quality of life or duration of life in either surgery or the palliative path. Here, we propose a model to numerically assess and compare the value of surgery vs palliation. This model includes quality and duration of life, as well as risk of perioperative mortality, and involves a patient’s preferences in the decision-making process.
For each pathway, surgery or palliative care, a value is calculated and compared to a normal life value (no disease symptoms and normal life expectancy). The formula is adjusted for the risk of operative mortality. The model produces a ratio of the value of surgery to the value of palliative care that signifies the superiority of one or another. This model calculation presents an objective estimated numerical value to compare the value of surgery and palliative care. It can be applied to every decision-making process before surgery. In general, if a procedure has the potential to significantly extend life in a patient who otherwise has a very short life expectancy with palliation only, performing high-risk surgery would be a reasonable option. A model that provides a numerical value for surgery vs palliative care and includes quality and duration of life in each pathway could be a useful tool for cardiac surgeons in decision making regarding high-risk surgery.
Keywords: high-risk surgery, palliative care, quality of life, life expectancy.
Patients with complex cardiovascular disease are occasionally considered inoperable due to the high risk of surgical mortality. When the risk of perioperative mortality (POM) is predicted to be too high, surgical intervention is denied, and patients are often referred to palliative care. The risk of POM in cardiac surgery is often calculated using large-scale databases, such as the Society of Thoracic Surgeons (STS) records. The STS risk models, which are regularly updated, are based on large data sets and incorporate precise statistical methods for risk adjustment.1 In general, these calculators provide a percentage value that defines the magnitude of the risk of death, and then an arbitrary range is selected to categorize the procedure as low, medium, or high risk or inoperable status. The STS database does not set a cutoff point or range to define “operability.” Assigning inoperable status to a certain risk rate is problematic, with many ethical, legal, and moral implications, and for this reason, it has mostly remained undefined. In contrast, the low- and medium-risk ranges are easier to define. Another limitation encountered in the STS database is the lack of risk data for less common but very high-risk procedures, such as a triple valve replacement.
A common example where risk classification has been defined is in patients who are candidates for surgical vs transcatheter aortic valve replacement. Some groups have described a risk of <4% as low risk, 4% to 8% as intermediate risk, >8% as high risk, and >15% as inoperable2; for some other groups, a risk of POM >50% is considered extreme risk or inoperable.3,4 This procedure-specific classification is a useful decision-making tool and helps the surgeon perform an initial risk assessment to allocate a specific patient to a group—operable or nonoperable—only by calculating the risk of surgical death. However, this allocation method does not provide any information on how and when death occurs in either group. These 2 parameters of how and when death occurs define the quality of life (QOL) and the duration of life (DOL), respectively, and together could be considered as the value of life in each pathway. A survivor of a high-risk surgery may benefit from good quality and extended life (a high value), or, on the other end of the spectrum, a high-risk patient who does not undergo surgery is spared the mortality risk of the surgery but dies sooner (low value) with symptoms due to the natural course of the untreated disease.
The central question is, if a surgery is high risk but has the potential of providing a good value (for those who survive it), what QOL and DOL values are acceptable to risk or to justify accepting and proceeding with a risky surgery? Or how high a POM risk is justified to proceed with surgery rather than the alternative palliative care with a certain quality and duration? It is obvious that a decision-making process that is based on POM cannot compare the value of surgery (Vs) and the value of palliation (Vp). Furthermore, it ignores patient preferences and their input, as these are excluded from this decision-making process.
To be able to include QOL and DOL in any decision making, one must precisely describe these parameters. Both QOL and DOL are used for estimation of disease burden by health care administrators, public health experts, insurance agencies, and others. Multiple models have been proposed and used to estimate the overall burden of the disease. Most of the models for this purpose are created for large-scale economic purposes and not for decision making in individual cases.
An important measure is the quality-adjusted life year (QALY). This is an important parameter since it includes both measures of quality and quantity of life.5,6 QALY is a simplified measure to assess the value of health outcomes, and it has been used in economic calculations to assess mainly the cost-effectiveness of various interventions. We sought to evaluate the utility of a similar method in adding further insight into the surgical decision-making process. In this article, we propose a simple model to compare the value of surgery vs palliative care, similar to QALY. This model includes and adjusts for the quality and the quantity of life, in addition to the risk of POM, in the decision-making process for high-risk patients.
The Model
The 2 decision pathways, surgery and palliative care, are compared for their value. We define the value as the product of QOL and DOL in each pathway and use the severity of the symptoms as a surrogate for QOL. If duration and quality were depicted on the x and y axes of a graph (Figure 1), then the area under the curve would represent the collective value in each situation. Figure 2 shows the timeline and the different pathways with each decision. The value in each situation is calculated in relation to the full value, which is represented as the value of normal life (Vn), that is, life without disease and with normal life expectancy. The values of each decision pathway, the value of surgery (Vs) and the value of palliation (Vp), are then compared to define the benefit for each decision as follows:
If Vs/Vp > 1, the benefit is toward surgery;
If Vs/Vp < 1, the benefit is for palliative care.


Definitions
Both quality and duration of life are presented on a 1-10 scale, 1 being the lowest and 10 the highest value, to yield a product with a value of 100 in normal, disease-free life. Any lower value is presented as a percentage to represent the comparison to the full value. QOL is determined by degradation of full quality with the average level of symptoms. DOL is calculated as a lost time (

For the DOL under any condition, a 10-year survival rate could be used as a surrogate in this formula. Compared to life expectancy value, using the 10-year survival rate simplifies the calculation since cardiac diseases are more prevalent in older age, close to or beyond the average life expectancy value.
Using the time intervals from the timeline in Figure 2:
dh = time interval from diagnosis to death at life expectancy
dg = time interval from diagnosis to death after successful surgery
df = time interval from diagnosis to death after palliative care
Duration for palliative care:
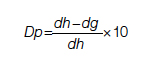
Duration for surgery:
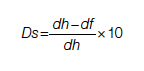
Adjustment: This value is calculated for those who survive the surgery. To adjust for the POM, it is multiplied by the 100 − POM risk.
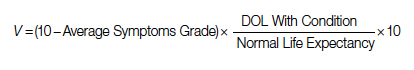
Since value is the base for comparison in this model, and it is the product of 2 equally important factors in the formula (

After elimination of normal life expectancy, form the numerator and denominator:
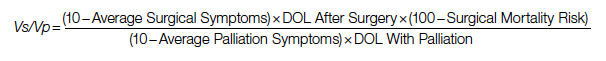
To adjust for surgical outcomes in special circumstances where less than optimal or standard surgical results are expected (eg, in very rare surgeries, limited resource institutions, or suboptimal postoperative surgical care), an optional coefficient R can be added to the numerator (surgical value). This optional coefficient, with values such as 0.8, 0.9 (to degrade the value of surgery) or 1 (standard surgical outcome), adjusts for variability in interinstitutional surgical results or surgeon variability. No coefficient is added to the denominator since palliative care provides minimal differences between clinicians and hospitals. Thus, the final adjusted formula would be as follows:
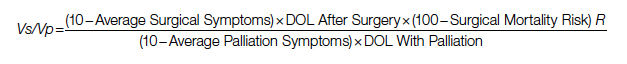
Example
A 60-year-old patient with a 10% POM risk needs to be allocated to surgical or palliative care. With palliative care, if this patient lived 6 years with average symptoms grade 4, the Vp would be 20; that is, 20% of the normal life value (if he lived 18 years instead without the disease).
Using the formula for calculation of value in each pathway:
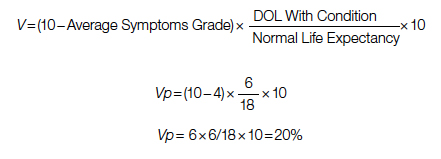
If the same patient undergoes a surgery with a 10% risk of POM, with an average grade 2 related to surgical recovery symptoms for 1 year and then is symptom-free and lives 12 years (instead of 18 years [life expectancy]), his Vs would be 53, or 53% out of the normal life value that is saved if the surgery is 100% successful; adjusted Vs with (chance of survival of 90%) would be 53 × 90% = 48%.
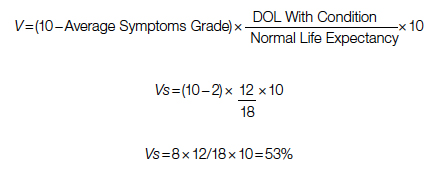
With adjustment of 90% survival chance in surgery, 53 × 90% = 48%. In this example, Vs/Vp = 48/20 = 2.4, showing a significant benefit for surgical care. Notably, the unknown value of normal life expectancy is not needed for the calculation of Vs/Vp, since it is the same in both pathways and it is eliminated by calculation in fraction.
Based on this formula, since the duration of surgical symptoms is short, no matter how severe these are, if the potential duration of life after surgery is high (represented by smaller area under the curve in Figure 1), the numerator becomes larger and the value of the surgery grows. For example, if a patient with a 15% risk of POM, which is generally considered inoperable, lives 5 years, as opposed to 2 years with palliative care with mild symptoms (eg 3/10), Vs/Vp would be 2.7, still showing a significant benefit for surgical care.
Discussion
Any surgical intervention is offered with 2 goals in mind, improving QOL and extending DOL. In a high-risk patient, surgery might be declined due to a high risk of POM, and the patient is offered palliative care, which other than providing symptom relief does not change the course of disease and eventually the patient will die due to the untreated disease. In this decision-making method, mostly completed by a care team only, a potential risk of death due to surgery which possibly could cure the patient is traded for immediate survival; however, the symptomatic course ensues until death. This mostly unilateral decision-making process by a care team, which incorporates minimal input from the patient or ignores patient preferences altogether, is based only on POM risk, and roughly includes a single parameter: years of potential life lost (YPLL). YPLL is a measure of premature mortality, and in the setting of surgical intervention, YPLL is the number of years a patient would lose unless a successful surgery were undertaken. Obviously, patients would live longer if a surgery that was intended to save them failed.
In this article, we proposed a simple method to quantify each decision to decide whether to operate or choose surgical care vs palliative care. Since quality and duration of life are both end factors clinicians and patients aspire to in each decision, they can be considered together as the value of each decision. We believe a numerical framework would provide an objective way to assist both the patient at high risk and the care team in the decision-making process.
The 2 parameters we consider are DOL and QOL. DOL, or survival, can be extracted from large-scale data using statistical methods that have been developed to predict survival under various conditions, such as Kaplan-Meier curves. These methods present the chance of survival in percentages in a defined time frame, such as a 5- or 10-year period.
While the DOL is a numerical parameter and quantifiable, the QOL is a more complex entity. This subjective parameter bears multiple definitions, aspects, and categories, and therefore multiple scales for quantification of QOL have been proposed. These scales have been used extensively for the purpose of health determination in health care policy and economic planning. Most scales acknowledge that QOL is multifactorial and includes interrelated aspects such as mental and socioeconomic factors. We have also noticed that QOL is better determined by the palliative care team than surgeons, so including these care providers in the decision-making process might reduce surgeon bias.
Since our purpose here is only to assist with the decision on medical intervention, we focus on physical QOL. Multiple scales are used to assess health-related QOL, such as the Assessment of Quality of Life (AQoL)-8D,7 EuroQol-5 Dimension (EQ-5D),8 15D,9 and the 36-Item Short Form Survey (SF-36).10 These complex scales are built for systematic reviews, and they are not practical for a clinical user. To simplify and keep this practical, we define QOL by using the severity or grade of symptoms related to the disease the patient has on a scale of 0 to 10. The severity of symptoms can be easily determined using available scales. An applicable scale for this purpose is the Edmonton Symptom Assessment Scale (ESAS), which has been in use for years and has evolved as a useful tool in the medical field.11
Once DOL and QOL are determined on a 1-10 scale, the multiplied value then provides a product that we consider a value. The highest value hoped for in each decision is the achievement of the best QOL and DOL, a value of 100. In Figure 1, a graphic presentation of value in each decision is best seen as the area under the curve. As shown, a successful surgery, even when accompanied by significant symptoms during initial recovery, has a chance (100 – risk of POM%) to gain a larger area under curve (value) by achieving a longer life with no or fewer symptoms. However, in palliative care, progressing disease and even palliated symptoms with a shorter life expectancy impose a large burden on the patient and a much lower value. Note that in this calculation, life expectancy, which is an important but unpredictable factor, is initially included; however, by ratio comparison, it is eliminated, simplifying the calculation further.
Using this formula in different settings reveals that high-risk surgery has a greater potential to reduce YPLL in the general population. Based on this formula, compared to a surgery with potential to significantly extend DOL, a definite shorter and symptomatic life course with palliative care makes it a significantly less favorable option. In fact, in the cardiovascular field, palliative care has minimal or no effect on natural history, as the mechanism of illness is mechanical, such as occlusion of coronary arteries or valve dysfunction, leading eventually to heart failure and death. In a study by Xu et al, although palliative care reduced readmission rates and improved symptoms on a variety of scales, there was no effect on mortality and QOL in patients with heart failure.12
No model in this field has proven to be ideal, and this model bears multiple limitations as well. We have used severity of symptoms as a surrogate for QOL based on the fact that cardiac patients with different pathologies who are untreated will have a common final pathway with development of heart failure symptoms that dictate their QOL. Also, grading QOL is a difficult task at times. Even a model such as QALY, which is one of the most used, is not a perfect model and is not free of problems.6 The difference in surgical results and life expectancy between sexes and ethnic groups might be a source of bias in this formula. Also, multiple factors directly and indirectly affect QOL and DOL and create inaccuracies; therefore, making an exact science from an inexact one naturally relies on multiple assumptions. Although it has previously been shown that most POM occurs in a short period of time after cardiac surgery,13 long-term complications that potentially degrade QOL are not included in this model. By applying this model, one must assume indefinite economic resources. Moreover, applying a single mathematical model in a biologic system and in the general population has intrinsic shortcomings, and it must overlook many other factors (eg, ethical, legal). For example, it will be hard to justify a failed surgery with 15% risk of POM undertaken to eliminate the severe long-lasting symptoms of a disease, while the outcome of a successful surgery with a 20% risk of POM that adds life and quality would be ignored in the current health care system. Thus, regardless of the significant potential, most surgeons would waive a surgery based solely on the percentage rate of POM, perhaps using other terms such as ”peri-nonoperative mortality.”
Conclusion
We have proposed a simple and practical formula for decision making regarding surgical vs palliative care in high-risk patients. By assigning a value that is composed of QOL and DOL in each pathway and including the risk of POM, a ratio of values provides a numerical estimation that can be used to show preference over a specific decision. An advantage of this formula, in addition to presenting an arithmetic value that is easier to understand, is that it can be used in shared decision making with patients. We emphasize that this model is only a preliminary concept at this time and has not been tested or validated for clinical use. Validation of such a model will require extensive work and testing within a large-scale population. We hope that this article will serve as a starting point for the development of other models, and that this formula will become more sophisticated with fewer limitations through larger multidisciplinary efforts in the future.
Corresponding author: Rabin Gerrah, MD, Good Samaritan Regional Medical Center, 3640 NW Samaritan Drive, Suite 100B, Corvallis, OR 97330; [email protected].
Disclosures: None reported.
From the Department of Cardiothoracic Surgery, Stanford University, Stanford, CA.
Abstract
Complex cardiac patients are often referred for surgery or palliative care based on the risk of perioperative mortality. This decision ignores factors such as quality of life or duration of life in either surgery or the palliative path. Here, we propose a model to numerically assess and compare the value of surgery vs palliation. This model includes quality and duration of life, as well as risk of perioperative mortality, and involves a patient’s preferences in the decision-making process.
For each pathway, surgery or palliative care, a value is calculated and compared to a normal life value (no disease symptoms and normal life expectancy). The formula is adjusted for the risk of operative mortality. The model produces a ratio of the value of surgery to the value of palliative care that signifies the superiority of one or another. This model calculation presents an objective estimated numerical value to compare the value of surgery and palliative care. It can be applied to every decision-making process before surgery. In general, if a procedure has the potential to significantly extend life in a patient who otherwise has a very short life expectancy with palliation only, performing high-risk surgery would be a reasonable option. A model that provides a numerical value for surgery vs palliative care and includes quality and duration of life in each pathway could be a useful tool for cardiac surgeons in decision making regarding high-risk surgery.
Keywords: high-risk surgery, palliative care, quality of life, life expectancy.
Patients with complex cardiovascular disease are occasionally considered inoperable due to the high risk of surgical mortality. When the risk of perioperative mortality (POM) is predicted to be too high, surgical intervention is denied, and patients are often referred to palliative care. The risk of POM in cardiac surgery is often calculated using large-scale databases, such as the Society of Thoracic Surgeons (STS) records. The STS risk models, which are regularly updated, are based on large data sets and incorporate precise statistical methods for risk adjustment.1 In general, these calculators provide a percentage value that defines the magnitude of the risk of death, and then an arbitrary range is selected to categorize the procedure as low, medium, or high risk or inoperable status. The STS database does not set a cutoff point or range to define “operability.” Assigning inoperable status to a certain risk rate is problematic, with many ethical, legal, and moral implications, and for this reason, it has mostly remained undefined. In contrast, the low- and medium-risk ranges are easier to define. Another limitation encountered in the STS database is the lack of risk data for less common but very high-risk procedures, such as a triple valve replacement.
A common example where risk classification has been defined is in patients who are candidates for surgical vs transcatheter aortic valve replacement. Some groups have described a risk of <4% as low risk, 4% to 8% as intermediate risk, >8% as high risk, and >15% as inoperable2; for some other groups, a risk of POM >50% is considered extreme risk or inoperable.3,4 This procedure-specific classification is a useful decision-making tool and helps the surgeon perform an initial risk assessment to allocate a specific patient to a group—operable or nonoperable—only by calculating the risk of surgical death. However, this allocation method does not provide any information on how and when death occurs in either group. These 2 parameters of how and when death occurs define the quality of life (QOL) and the duration of life (DOL), respectively, and together could be considered as the value of life in each pathway. A survivor of a high-risk surgery may benefit from good quality and extended life (a high value), or, on the other end of the spectrum, a high-risk patient who does not undergo surgery is spared the mortality risk of the surgery but dies sooner (low value) with symptoms due to the natural course of the untreated disease.
The central question is, if a surgery is high risk but has the potential of providing a good value (for those who survive it), what QOL and DOL values are acceptable to risk or to justify accepting and proceeding with a risky surgery? Or how high a POM risk is justified to proceed with surgery rather than the alternative palliative care with a certain quality and duration? It is obvious that a decision-making process that is based on POM cannot compare the value of surgery (Vs) and the value of palliation (Vp). Furthermore, it ignores patient preferences and their input, as these are excluded from this decision-making process.
To be able to include QOL and DOL in any decision making, one must precisely describe these parameters. Both QOL and DOL are used for estimation of disease burden by health care administrators, public health experts, insurance agencies, and others. Multiple models have been proposed and used to estimate the overall burden of the disease. Most of the models for this purpose are created for large-scale economic purposes and not for decision making in individual cases.
An important measure is the quality-adjusted life year (QALY). This is an important parameter since it includes both measures of quality and quantity of life.5,6 QALY is a simplified measure to assess the value of health outcomes, and it has been used in economic calculations to assess mainly the cost-effectiveness of various interventions. We sought to evaluate the utility of a similar method in adding further insight into the surgical decision-making process. In this article, we propose a simple model to compare the value of surgery vs palliative care, similar to QALY. This model includes and adjusts for the quality and the quantity of life, in addition to the risk of POM, in the decision-making process for high-risk patients.
The Model
The 2 decision pathways, surgery and palliative care, are compared for their value. We define the value as the product of QOL and DOL in each pathway and use the severity of the symptoms as a surrogate for QOL. If duration and quality were depicted on the x and y axes of a graph (Figure 1), then the area under the curve would represent the collective value in each situation. Figure 2 shows the timeline and the different pathways with each decision. The value in each situation is calculated in relation to the full value, which is represented as the value of normal life (Vn), that is, life without disease and with normal life expectancy. The values of each decision pathway, the value of surgery (Vs) and the value of palliation (Vp), are then compared to define the benefit for each decision as follows:
If Vs/Vp > 1, the benefit is toward surgery;
If Vs/Vp < 1, the benefit is for palliative care.


Definitions
Both quality and duration of life are presented on a 1-10 scale, 1 being the lowest and 10 the highest value, to yield a product with a value of 100 in normal, disease-free life. Any lower value is presented as a percentage to represent the comparison to the full value. QOL is determined by degradation of full quality with the average level of symptoms. DOL is calculated as a lost time (

For the DOL under any condition, a 10-year survival rate could be used as a surrogate in this formula. Compared to life expectancy value, using the 10-year survival rate simplifies the calculation since cardiac diseases are more prevalent in older age, close to or beyond the average life expectancy value.
Using the time intervals from the timeline in Figure 2:
dh = time interval from diagnosis to death at life expectancy
dg = time interval from diagnosis to death after successful surgery
df = time interval from diagnosis to death after palliative care
Duration for palliative care:
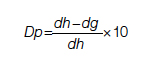
Duration for surgery:
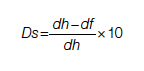
Adjustment: This value is calculated for those who survive the surgery. To adjust for the POM, it is multiplied by the 100 − POM risk.
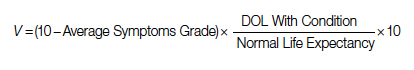
Since value is the base for comparison in this model, and it is the product of 2 equally important factors in the formula (

After elimination of normal life expectancy, form the numerator and denominator:
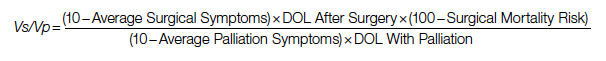
To adjust for surgical outcomes in special circumstances where less than optimal or standard surgical results are expected (eg, in very rare surgeries, limited resource institutions, or suboptimal postoperative surgical care), an optional coefficient R can be added to the numerator (surgical value). This optional coefficient, with values such as 0.8, 0.9 (to degrade the value of surgery) or 1 (standard surgical outcome), adjusts for variability in interinstitutional surgical results or surgeon variability. No coefficient is added to the denominator since palliative care provides minimal differences between clinicians and hospitals. Thus, the final adjusted formula would be as follows:
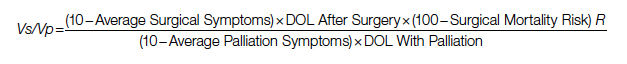
Example
A 60-year-old patient with a 10% POM risk needs to be allocated to surgical or palliative care. With palliative care, if this patient lived 6 years with average symptoms grade 4, the Vp would be 20; that is, 20% of the normal life value (if he lived 18 years instead without the disease).
Using the formula for calculation of value in each pathway:
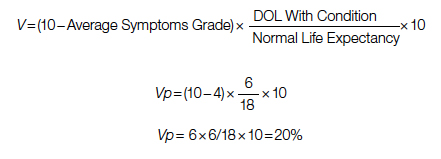
If the same patient undergoes a surgery with a 10% risk of POM, with an average grade 2 related to surgical recovery symptoms for 1 year and then is symptom-free and lives 12 years (instead of 18 years [life expectancy]), his Vs would be 53, or 53% out of the normal life value that is saved if the surgery is 100% successful; adjusted Vs with (chance of survival of 90%) would be 53 × 90% = 48%.
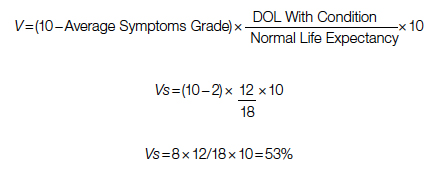
With adjustment of 90% survival chance in surgery, 53 × 90% = 48%. In this example, Vs/Vp = 48/20 = 2.4, showing a significant benefit for surgical care. Notably, the unknown value of normal life expectancy is not needed for the calculation of Vs/Vp, since it is the same in both pathways and it is eliminated by calculation in fraction.
Based on this formula, since the duration of surgical symptoms is short, no matter how severe these are, if the potential duration of life after surgery is high (represented by smaller area under the curve in Figure 1), the numerator becomes larger and the value of the surgery grows. For example, if a patient with a 15% risk of POM, which is generally considered inoperable, lives 5 years, as opposed to 2 years with palliative care with mild symptoms (eg 3/10), Vs/Vp would be 2.7, still showing a significant benefit for surgical care.
Discussion
Any surgical intervention is offered with 2 goals in mind, improving QOL and extending DOL. In a high-risk patient, surgery might be declined due to a high risk of POM, and the patient is offered palliative care, which other than providing symptom relief does not change the course of disease and eventually the patient will die due to the untreated disease. In this decision-making method, mostly completed by a care team only, a potential risk of death due to surgery which possibly could cure the patient is traded for immediate survival; however, the symptomatic course ensues until death. This mostly unilateral decision-making process by a care team, which incorporates minimal input from the patient or ignores patient preferences altogether, is based only on POM risk, and roughly includes a single parameter: years of potential life lost (YPLL). YPLL is a measure of premature mortality, and in the setting of surgical intervention, YPLL is the number of years a patient would lose unless a successful surgery were undertaken. Obviously, patients would live longer if a surgery that was intended to save them failed.
In this article, we proposed a simple method to quantify each decision to decide whether to operate or choose surgical care vs palliative care. Since quality and duration of life are both end factors clinicians and patients aspire to in each decision, they can be considered together as the value of each decision. We believe a numerical framework would provide an objective way to assist both the patient at high risk and the care team in the decision-making process.
The 2 parameters we consider are DOL and QOL. DOL, or survival, can be extracted from large-scale data using statistical methods that have been developed to predict survival under various conditions, such as Kaplan-Meier curves. These methods present the chance of survival in percentages in a defined time frame, such as a 5- or 10-year period.
While the DOL is a numerical parameter and quantifiable, the QOL is a more complex entity. This subjective parameter bears multiple definitions, aspects, and categories, and therefore multiple scales for quantification of QOL have been proposed. These scales have been used extensively for the purpose of health determination in health care policy and economic planning. Most scales acknowledge that QOL is multifactorial and includes interrelated aspects such as mental and socioeconomic factors. We have also noticed that QOL is better determined by the palliative care team than surgeons, so including these care providers in the decision-making process might reduce surgeon bias.
Since our purpose here is only to assist with the decision on medical intervention, we focus on physical QOL. Multiple scales are used to assess health-related QOL, such as the Assessment of Quality of Life (AQoL)-8D,7 EuroQol-5 Dimension (EQ-5D),8 15D,9 and the 36-Item Short Form Survey (SF-36).10 These complex scales are built for systematic reviews, and they are not practical for a clinical user. To simplify and keep this practical, we define QOL by using the severity or grade of symptoms related to the disease the patient has on a scale of 0 to 10. The severity of symptoms can be easily determined using available scales. An applicable scale for this purpose is the Edmonton Symptom Assessment Scale (ESAS), which has been in use for years and has evolved as a useful tool in the medical field.11
Once DOL and QOL are determined on a 1-10 scale, the multiplied value then provides a product that we consider a value. The highest value hoped for in each decision is the achievement of the best QOL and DOL, a value of 100. In Figure 1, a graphic presentation of value in each decision is best seen as the area under the curve. As shown, a successful surgery, even when accompanied by significant symptoms during initial recovery, has a chance (100 – risk of POM%) to gain a larger area under curve (value) by achieving a longer life with no or fewer symptoms. However, in palliative care, progressing disease and even palliated symptoms with a shorter life expectancy impose a large burden on the patient and a much lower value. Note that in this calculation, life expectancy, which is an important but unpredictable factor, is initially included; however, by ratio comparison, it is eliminated, simplifying the calculation further.
Using this formula in different settings reveals that high-risk surgery has a greater potential to reduce YPLL in the general population. Based on this formula, compared to a surgery with potential to significantly extend DOL, a definite shorter and symptomatic life course with palliative care makes it a significantly less favorable option. In fact, in the cardiovascular field, palliative care has minimal or no effect on natural history, as the mechanism of illness is mechanical, such as occlusion of coronary arteries or valve dysfunction, leading eventually to heart failure and death. In a study by Xu et al, although palliative care reduced readmission rates and improved symptoms on a variety of scales, there was no effect on mortality and QOL in patients with heart failure.12
No model in this field has proven to be ideal, and this model bears multiple limitations as well. We have used severity of symptoms as a surrogate for QOL based on the fact that cardiac patients with different pathologies who are untreated will have a common final pathway with development of heart failure symptoms that dictate their QOL. Also, grading QOL is a difficult task at times. Even a model such as QALY, which is one of the most used, is not a perfect model and is not free of problems.6 The difference in surgical results and life expectancy between sexes and ethnic groups might be a source of bias in this formula. Also, multiple factors directly and indirectly affect QOL and DOL and create inaccuracies; therefore, making an exact science from an inexact one naturally relies on multiple assumptions. Although it has previously been shown that most POM occurs in a short period of time after cardiac surgery,13 long-term complications that potentially degrade QOL are not included in this model. By applying this model, one must assume indefinite economic resources. Moreover, applying a single mathematical model in a biologic system and in the general population has intrinsic shortcomings, and it must overlook many other factors (eg, ethical, legal). For example, it will be hard to justify a failed surgery with 15% risk of POM undertaken to eliminate the severe long-lasting symptoms of a disease, while the outcome of a successful surgery with a 20% risk of POM that adds life and quality would be ignored in the current health care system. Thus, regardless of the significant potential, most surgeons would waive a surgery based solely on the percentage rate of POM, perhaps using other terms such as ”peri-nonoperative mortality.”
Conclusion
We have proposed a simple and practical formula for decision making regarding surgical vs palliative care in high-risk patients. By assigning a value that is composed of QOL and DOL in each pathway and including the risk of POM, a ratio of values provides a numerical estimation that can be used to show preference over a specific decision. An advantage of this formula, in addition to presenting an arithmetic value that is easier to understand, is that it can be used in shared decision making with patients. We emphasize that this model is only a preliminary concept at this time and has not been tested or validated for clinical use. Validation of such a model will require extensive work and testing within a large-scale population. We hope that this article will serve as a starting point for the development of other models, and that this formula will become more sophisticated with fewer limitations through larger multidisciplinary efforts in the future.
Corresponding author: Rabin Gerrah, MD, Good Samaritan Regional Medical Center, 3640 NW Samaritan Drive, Suite 100B, Corvallis, OR 97330; [email protected].
Disclosures: None reported.
1. O’Brien SM, Feng L, He X, et al. The Society of Thoracic Surgeons 2018 Adult Cardiac Surgery Risk Models: Part 2-statistical methods and results. Ann Thorac Surg. 2018;105(5):1419-1428. doi: 10.1016/j.athoracsur.2018.03.003
2. Hurtado Rendón IS, Bittenbender P, Dunn JM, Firstenberg MS. Chapter 8: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines. In: Transcatheter Heart Valve Handbook: A Surgeons’ and Interventional Council Review. Akron City Hospital, Summa Health System, Akron, OH.
3. Herrmann HC, Thourani VH, Kodali SK, et al; PARTNER Investigators. One-year clinical outcomes with SAPIEN 3 transcatheter aortic valve replacement in high-risk and inoperable patients with severe aortic stenosis. Circulation. 2016;134:130-140. doi:10.1161/CIRCULATIONAHA
4. Ho C, Argáez C. Transcatheter Aortic Valve Implantation for Patients with Severe Aortic Stenosis at Various Levels of Surgical Risk: A Review of Clinical Effectiveness. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; March 19, 2018.
5. Rios-Diaz AJ, Lam J, Ramos MS, et al. Global patterns of QALY and DALY use in surgical cost-utility analyses: a systematic review. PLoS One. 2016:10;11:e0148304. doi:10.1371/journal.pone.0148304
6. Prieto L, Sacristán JA. Health, Problems and solutions in calculating quality-adjusted life years (QALYs). Qual Life Outcomes. 2003:19;1:80.
7. Centre for Health Economics. Assessment of Quality of Life. 2014. Accessed May 13, 2022. http://www.aqol.com.au/
8. EuroQol Research Foundation. EQ-5D. Accessed May 13, 2022. https://euroqol.org/
9. 15D Instrument. Accessed May 13, 2022. http://www.15d-instrument.net/15d/
10. Rand Corporation. 36-Item Short Form Survey (SF-36).Accessed May 12, 2022. https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form.html
11. Hui D, Bruera E. The Edmonton Symptom Assessment System 25 years later: past, present, and future developments. J Pain Symptom Manage. 2017:53:630-643. doi:10.1016/j.jpainsymman.2016
12. Xu Z, Chen L, Jin S, Yang B, Chen X, Wu Z. Effect of palliative care for patients with heart failure. Int Heart J. 2018:30;59:503-509. doi:10.1536/ihj.17-289
13. Mazzeffi M, Zivot J, Buchman T, Halkos M. In-hospital mortality after cardiac surgery: patient characteristics, timing, and association with postoperative length of intensive care unit and hospital stay. Ann Thorac Surg. 2014;97:1220-1225. doi:10.1016/j.athoracsur.2013.10.040
1. O’Brien SM, Feng L, He X, et al. The Society of Thoracic Surgeons 2018 Adult Cardiac Surgery Risk Models: Part 2-statistical methods and results. Ann Thorac Surg. 2018;105(5):1419-1428. doi: 10.1016/j.athoracsur.2018.03.003
2. Hurtado Rendón IS, Bittenbender P, Dunn JM, Firstenberg MS. Chapter 8: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines. In: Transcatheter Heart Valve Handbook: A Surgeons’ and Interventional Council Review. Akron City Hospital, Summa Health System, Akron, OH.
3. Herrmann HC, Thourani VH, Kodali SK, et al; PARTNER Investigators. One-year clinical outcomes with SAPIEN 3 transcatheter aortic valve replacement in high-risk and inoperable patients with severe aortic stenosis. Circulation. 2016;134:130-140. doi:10.1161/CIRCULATIONAHA
4. Ho C, Argáez C. Transcatheter Aortic Valve Implantation for Patients with Severe Aortic Stenosis at Various Levels of Surgical Risk: A Review of Clinical Effectiveness. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; March 19, 2018.
5. Rios-Diaz AJ, Lam J, Ramos MS, et al. Global patterns of QALY and DALY use in surgical cost-utility analyses: a systematic review. PLoS One. 2016:10;11:e0148304. doi:10.1371/journal.pone.0148304
6. Prieto L, Sacristán JA. Health, Problems and solutions in calculating quality-adjusted life years (QALYs). Qual Life Outcomes. 2003:19;1:80.
7. Centre for Health Economics. Assessment of Quality of Life. 2014. Accessed May 13, 2022. http://www.aqol.com.au/
8. EuroQol Research Foundation. EQ-5D. Accessed May 13, 2022. https://euroqol.org/
9. 15D Instrument. Accessed May 13, 2022. http://www.15d-instrument.net/15d/
10. Rand Corporation. 36-Item Short Form Survey (SF-36).Accessed May 12, 2022. https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form.html
11. Hui D, Bruera E. The Edmonton Symptom Assessment System 25 years later: past, present, and future developments. J Pain Symptom Manage. 2017:53:630-643. doi:10.1016/j.jpainsymman.2016
12. Xu Z, Chen L, Jin S, Yang B, Chen X, Wu Z. Effect of palliative care for patients with heart failure. Int Heart J. 2018:30;59:503-509. doi:10.1536/ihj.17-289
13. Mazzeffi M, Zivot J, Buchman T, Halkos M. In-hospital mortality after cardiac surgery: patient characteristics, timing, and association with postoperative length of intensive care unit and hospital stay. Ann Thorac Surg. 2014;97:1220-1225. doi:10.1016/j.athoracsur.2013.10.040