User login
Staphylococcus aureus: The new adventures of a legendary pathogen
Staphylococcus aureus is rearing its ugly head in new and interesting ways, both in the hospital and in the community.
Rates of invasive infections with methicillin-resistant S aureus (MRSA) have been increasing both in the hospital and in the community, a trend that has attracted considerable interest in the lay media. Curiously, the most common community-associated MRSA strain, which up to now has been distinct from hospital-associated MRSA strains, is invading our hospitals. Alarmingly, vancomycin (Vancocin), the drug of last resort for MRSA infections for the past 40 years, does not seem to be as effective as it used to be.
This paper summarizes the changing epidemiology of S aureus, particularly the emergence of MRSA outside of the hospital; reviews the difficulties associated with S aureus bacteremia and its treatment in view of; some changes in vancomycin susceptibility; and appraises the old and new treatment options.
MRSA IS ON THE RISE IN THE HOSPITAL
S aureus, a gram-positive, coagulase-positive bacterium, is one of the leading nosocomial bloodstream pathogens, second only to coagulase-negative staphylococci.1 And the incidence of S aureus infections is increasing. MRSA in particular is increasingly causing infections throughout hospitals, including intensive care units. As of 2004, nearly two-thirds of isolates of S aureus from intensive care units were MRSA.2
MRSA infections are worse than methicillin-susceptible S aureus (MSSA) infections in terms of the rates of death and other undesirable outcomes.3 Several factors may be responsible: MRSA infection may be a marker of severity of illness (sicker patients may be more likely to have MRSA), our treatment for MRSA may not be as effective as it is for MSSA, and the organism may be inherently more virulent.
METHICILLIN RESISTANCE IS ALSO ON THE RISE IN THE COMMUNITY
Community-associated MRSA began emerging clinically about 10 years ago. It was first described in a cohort of children with necrotizing pneumonia in Minnesota, but soon other populations at risk began to emerge, such as residents of correctional facilities, men who had sex with men, competitive athletes (eg, fencers, wrestlers, and football players), and Alaskan natives and other native populations. A common factor in all these groups was close proximity of the members to each other. Later, it began to spread beyond these traditional risk groups into the community at large.
Community-associated MRSA strains have a characteristic pattern of antimicrobial susceptibility (see below). In the laboratory, they grow somewhat faster than health-care-associated MRSA strains, but not as fast as MSSA. They have a strong association with skin and soft-tissue infections: when you see a skin or soft-tissue infection, be it in an outpatient or an inpatient, think about MRSA. Their virulence varies, but rapid onset and progression of illness are quite common. Their most common strain in the United States at present is USA 300.
Case 1: A young woman with necrotizing fasciitis
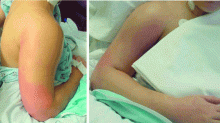
A 21-year-old college student presented to our service in May 2004 with high fever and severe arm pain, which had been worsening for several days. She had been previously healthy, had not had any contact with the health care system, and had not received any antibiotics.
Her blood cultures were positive for MRSA, as were cultures of the deep tissue of the deltoid muscle and fascia when she underwent emergency surgical debridement. The infection required several additional surgical debridements and removal of one head of her deltoid muscle, but she was fortunate: in the past, some patients with this problem might have undergone radical amputation of the arm or even more extensive surgery. This patient continued to have positive blood cultures 4 days postoperatively, but she ultimately recovered, completing 28 days of daptomycin (Cubicin) therapy at a dose of 6 mg/kg every 24 hours. The last 10 days of daptomycin therapy were given at home via a percutaneous intravenous central catheter.
Comment. The epidemiology of MRSA infections is changing. More patients who have no traditional risk factors, specifically health care contact, are getting MRSA infections. A recent report from the US Centers for Disease Control and Prevention (CDC) indicates that the proportion of patients with invasive disease due to MRSA has doubled since 2001–2002.4 Part of the reason undoubtedly is that MRSA, particularly community-associated MRSA, often carries specific virulence factors that make it more invasive. The CDC estimated that in 2005 there were nearly 100,000 cases of invasive MRSA infection in the United States, and nearly a fifth of these infections resulted in death.
Resistance and virulence factors in community-associated MRSA
Most community-associated MRSA strains carry a mobile genetic element called type IV SCCmec (staphylococcal chromosomal cassettemec) that enhances its antimicrobial resistance. This genetic component was probably borrowed from coagulase-negative staphylococci, in which it is quite common but does not cause as much of a problem. It is now present in a wide range of S aureus strains. Most of the S aureus strains that carry type IV SCCmec are MRSA, but a few MSSA strains do carry it as well.
The potent toxin Panton-Valentine leukocidin is an extracellular product that is detected in fewer than 5% of hospital strains but is more common in community-associated strains. It kills leukocytes by forming pores in the cell membrane and causing skin necrosis in cutaneous infections. It is associated with skin abscesses and rapidly progressive necrotizing pneumonia in MSSA or MRSA.
Epidemiologic differences between community- and health-care-associated MRSA
Patients with community-associated MRSA infections tend to be younger than those who traditionally get health-care-associated MRSA infections: in a study from Naimi et al in 2003, the mean ages were 23 vs 68 years.5 A greater proportion of patients with community-associated MRSA strains are nonwhite.4,5
Most community-associated MRSA infections are of the skin and soft tissue (75% in the series from Naimi et al5), but this pathogen causes other infections as well. Bacteremia of unknown origin has been seen, as has necrotizing pneumonia. Most of the skin and soft-tissue infections are relatively superficial, such as folliculitis or furunculosis, but deeper tissue infections such as necrotizing fasciitis and pyomyositis have also been seen.6
The incidence of community-associated MRSA infections varies greatly by geographic region.7 The northeastern United States has so far been relatively spared, but in Atlanta, Houston, and Los Angeles up to 80% of cases of characteristic skin or soft-tissue infections seen in emergency or outpatient departments are due to community-associated MRSA. Physicians at the Texas Children’s Hospital in Houston assume that all skin or soft-tissue infections are due to community-associated MRSA unless proven otherwise.8
Differences in antibiotic susceptibility
Community-associated MRSA is more susceptible to various antibiotics than health-care-associated MRSA,5 but not by much. Strains are usually susceptible to vancomycin, tetracyclines, trimethoprim-sulfamethoxazole (Bactrim, Septra), and rifampin (Rifadin). Unlike hospital strains, a fair number of community-acquired strains are susceptible to clindamycin (Cleocin) in the laboratory, but with a caveat: some of these clindamycin-susceptible strains actually may harbor the tools for inducible resistance. In fact, they can become resistant to clindamycin even without being exposed to it.
The laboratory test for inducible clindamycin resistance is called the D test. After coating an agar plate with S aureus, the technician places erythromycin and clindamycin disks. If the erythromycin induces clindamycin resistance, the plate is clear of growth around the clindamycin disk except for the portion nearest the erythromycin disk, leaving a characteristic D-shaped area of lucency.
Risk factors for MRSA
Moran et al7 analyzed the risk factors for community-associated MRSA in patients with skin or soft-tissue infections seen in the emergency department. The infection was more likely to be due to community-associated MRSA if the patient was black, had used any antibiotic in the past month, had a history of MRSA infection, or had close contact with a person with a similar infection. Many patients interpreted the infections as spider bites because the lesions tended to have a dark center surrounded by a tender area. These infections were not associated with underlying illness. In some cases, community-associated MRSA skin infections have been associated with tattooing and even manicuring.
However, it is very difficult to distinguish between community-associated MRSA and MSSA skin and soft-tissue infections on the basis of clinical and epidemiologic characteristics. Miller et al9 studied a large group of patients in Los Angeles who were hospitalized with community-associated skin and soft-tissue S aureus infections. All the patients were followed up for 30 days after hospital discharge. Regardless of whether they had MRSA or MSSA, they had similar outcomes. Close contacts of the patients also tended to develop infection.
A key point from this and many other studies: patients were more likely to remain infected if they did not undergo incision and drainage. This key intervention is indicated for any patient who has a skin and soft-tissue infection with an undrained focus of infection.
COMMUNITY-ASSOCIATED MRSA IS INVADING THE HOSPITAL
In a new development, community-associated MRSA strains are now appearing in the hospital. This is not only because patients are bacteremic when they come in: patients in the hospital are getting nosocomial infections due to community-associated MRSA strains.
Seybold et al10 analyzed 116 cases of MRSA bloodstream infections in Atlanta, GA. In 9 (8%) of the cases the patient had not had any contact with the health care system within the past year, and these cases were classified as truly community-associated. Of the remaining 107 cases, 49 (42%) were nosocomial, and the USA 300 strain—the predominant community-associated MRSA strain—accounted for 10 (20%) of the nosocomial cases.
In the recent CDC study of invasive MRSA infections, Klevens et al4 reported that nearly a third of cases of bacteremia were due to community-associated MRSA, and these strains accounted for a greater proportion of cases of cellulitis and endocarditis than did health-care-associated strains.
In a study of hospital-associated MRSA, Maree et al11 found that the percentage of cases in which the bacteria carried the SCCmec type IV marker had increased from less than 20% in 1999 to more than 50% in 2004.
Comment. Suffice it to say that we are surrounded by MRSA. Community-associated MRSA is here to stay. It is even invading our hospitals, and we need to consider this very carefully when choosing antimicrobial therapy.
NAGGING QUESTIONS ABOUT VANCOMYCIN
Case 2: Vancomycin-intermediate S aureus (VISA) bacteremia and endocarditis
In December 2006 we saw a very ill 60-year-old woman who was hospitalized with MRSA bacteremia, pacemaker endocarditis, and superior vena cava thrombosis. Although she was treated with vancomycin and rifampin, her condition worsened, she had a stroke, and she developed renal failure. In a difficult operation, the pacemaker was removed, but the bacteremia persisted. In early February 2007 she underwent another difficult operation in which the superior vena cava clot was debrided, a right atrial clot was removed, and her mitral valve was replaced. Less than 2 weeks later, and despite ongoing vancomycin and rifampin therapy, the MRSA bacteremia recurred.
During the approximately 6 weeks that the patient had been receiving these antibiotics, the minimal inhibitory concentration (MIC) of rifampin against the S aureus isolate increased from less than 1 μg/mL (susceptible) to 2 μg/mL (resistant). The MIC of vancomycin went from 2 μg/mL (susceptible) to 4 μg/mL (intermediately susceptible). Vancomycin and rifampin were discontinued, and daptomycin and gentamicin (Garamycin) therapy were started. (Her daptomycin MIC was 0.5 μg/mL). The patient’s condition stabilized, and she was discharged to a long-term nursing facility. She had no relapse of MRSA bacteremia, but she died in early April of that year.
Is vancomycin becoming less effective? Degrees of vancomycin resistance
Vancomycin has been our stalwart for treating MRSA infections for more than 40 years but it is not working as well as it used to, at least in certain situations.
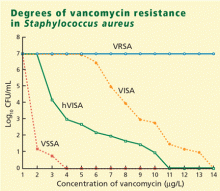
VRSA (vancomycin-resistant S aureus) is rare. These fully resistant strains probably acquired a resistance mechanism (the vanA operon) from vancomycin-resistant enterococci. Infections tend to occur in patients simultaneously infected with both S aureus and vancomycin-resistant enterococci, giving the bacteria an opportunity to exchange genetic material.
VISA (vancomycin-intermediate S aureus) infections tend to occur in patients like the one described above who have had long-term vancomycin therapy. VISA strains appear to overproduce a matrix that captures vancomycin and keeps it from entering the cell. On electron microscopy, these bacteria have a very thick cell wall.13
Vancomycin tolerance is a state in which the bacteria are “stunned” or kept in check but not killed by vancomycin. That is manifested in the laboratory by a ratio of minimum bactericidal concentration to MIC greater than 32.
hVISA (heteroresistant VISA) is new and worrisome. These organisms have an overall MIC in the susceptible range, but within that population are individual isolates with an MIC that is much higher—in the intermediate or perhaps even in the resistant range.14
Reported rates of hVISA vary from less than 2% to as high as 76%, because the methods for detecting it are still very poorly standardized. The usual automated laboratory tests do not detect hVISA.
hVISA is probably clinically relevant, as evidence is emerging both in vitro and in vivo that the higher the MIC for vancomycin, the worse the clinical outcome.15 hVISA has been associated with failures of therapy in several situations, usually in cases of severe invasive or deep infection, endocarditis, and bacteremia with vertebral osteomyelitis where vancomycin concentrations at the site of infection may be suboptimal.16–19 While most hVISA strains that have been described were resistant to methicillin, some were susceptible.
The E test is emerging as the standard test for hVISA. This test uses a plastic strip that contains gradually increasing concentrations of vancomycin along its length. Placed in the culture dish, the strip inhibits growth of the organism at its high-concentration end but not at its low-concentration end. If the sample contains hVISA, the cutoff is not well defined, with a few colonies growing at higher concentrations.
New definition of vancomycin susceptibility
Recognizing that the MICs for vancomycin have been rising in the last few years, the Clinical and Laboratory Standards Institute last year changed the break points between susceptibility and resistance. The new definitions are:
- Susceptible—an MIC of 2.0 μg/mL or less (formerly 4.0 μg/mL or less)
- Intermediate—4.0 to 8.0 μg/mL (formerly 8.0 to 16 μg/mL)
- Resistant—16 μg/mL or greater (formerly 32 μg/mL or greater).
One should pay attention to the MIC numbers on the laboratory reports, not just to the words “susceptible” or “not susceptible.” If the number is, say, 0.5 μg/mL or less, the organism should really be susceptible. If the number is 1 or 2, it is still in the susceptible range, but those are the organisms that may cause problems later on.
Further, even if the vancomycin MIC is in the susceptible range, higher MICs may affect outcomes. The average duration of MRSA bacteremia on therapy is 8 to 9 days, vs 3 to 4 days with MSSA bacteremia.20,21 But Sakoulas et al15 found that, in MRSA bacteremia, the success rate with vancomycin therapy was 56% if the MIC was 0.5 or lower, compared with 10% if the MIC was 1.0 to 2.0 μg/mL. Examined in another way, the success rate was 50% if the logarithm of killing was 6.27 colony-forming units per mL or greater, 23% if 4.71 to 6.26, and zero if less than 4.71.
Case 3: Prolonged MRSA bacteremia
In the summer of 2006, a 66-year-old woman with a history of gastric bypass and cirrhosis underwent a long stay in the surgical intensive care unit because of a recurrent enterocutaneous fistula and chronic renal insufficiency. On November 5th, she had a positive blood culture for MRSA, which was treated appropriately with vancomycin for 4 weeks. She was discharged to subacute care but came back 2 days later, again with MRSA bacteremia. At that time her Hickman catheter, which had been inserted for total parenteral nutrition because of the enterocutaneous fistula, was removed.
Transthoracic echocardiography revealed no vegetations, but her bacteremia persisted. Her mental status was poor this entire time: she was mute and could barely be awakened. We looked for clots and infected clots; duplex ultrasonographic examinations of all four extremities were negative. Finally, magnetic resonance imaging of her back—performed empirically because of the persistent bacteremia—revealed vertebral osteomyelitis at level T12-L1. We also noticed on serial evaluations that the vancomycin MIC for her organism increased from 0.5 to 2.0 μg/mL, so therapy was changed from vancomycin to daptomycin.
Her bacteremia cleared. Follow-up echocardiography was negative, but she had two subsequent relapses of MRSA bacteremia, one in April 2007 and one before she died in the summer of 2007.
Prolonged bacteremia: Is it vancomycin resistance, or something else?
The MRSA isolates that cause prolonged bacteremia seem to have certain characteristics.22 Higher MICs are probably associated with longer periods of bacteremia. But some genetic components within some strains of S aureus give them a survival advantage. They have less susceptibility to the body’s thrombin-induced platelet microbicidal protein. These isolates are not only associated with prolonged bacteremia: they are also associated with osteomyelitis, deep abscesses, endocarditis, recurrent infection, and increased death rate.22 Clinical laboratories do not test for these genetic components. One wonders whether our patient may have had an isolate with these mutations that gave it a survival advantage.
Do not use vancomycin for MSSA
Avoid using vancomycin for MSSA infections. It has been shown time and time again that MSSA infections do not respond as well to vancomycin as they do to beta-lactam antibiotics, specifically to the semisynthetic penicillins such as oxacillin and nafcillin, and even some of the first-generation cephalosporins. Chang et al23 found that patients with MSSA bacteremia had higher rates of persistent infections, relapse, and bacteriologic failure if they received vancomycin than if they received nafcillin.
Do vancomycin trough levels affect toxicity?
The vancomycin trough levels that we aimed for in the past (5 to 10 μg/mL) were probably too low. Today, we aim for trough levels of 15 to 20 μg/mL, and many physicians are aiming for 20 to 25 μg/mL. Part of the reason is that vancomycin MICs are higher than they used to be: in order to keep the vancomycin level above the MIC for a longer period of time, the vancomycin trough level needs to be higher. In theory, keeping the vancomycin levels above the MIC for longer periods should improve outcomes. Yet Fowler et al22 found that vancomycin trough levels among patients who had persistent MRSA bacteremia were actually higher than trough levels among those in whom the bacteremia resolved, although the difference was not statistically significant.
We measure the vancomycin trough level to make sure it is high enough (and give larger doses if it is not); among adults, peak levels need not be monitored on a routine basis because of the predictable pharmacokinetics of vancomycin.
Vancomycin toxicity can be either idiosyncratic or synergistic. Idiosyncratic toxicity occurs when a patient who has been on vancomycin for a long time develops a fixed rash, not associated with infusion. This is an immunologic phenomenon. It is a rare and very serious situation and may require steroid therapy.
Synergistic toxicity occurs when vancomycin is given with other nephrotoxic agents, notably gentamicin. Vancomycin plus gentamicin equals nephrotoxicity. Vancomycin alone is usually not nephrotoxic, but close monitoring of renal function parameters is warranted with the use of higher doses.24
IN UNEXPLAINED BACTEREMIA, LOOK FOR ENDOCARDITIS
In blood cultures from patients with bacteremia, S aureus is never a contaminant. Even if just one blood culture is positive for S aureus, believe that S aureus is the culprit.
Reports in the 1950s suggested that at least half of patients who had S aureus bacteremia had endocarditis,25 leading to recommendations that all patients with S aureus bacteremia without an obvious primary source of infection should be evaluated for endocarditis. Subsequent estimates were lower, in the range of 15% to 25%.26,27 However, throughout the world S aureus endocarditis continues to have a very high mortality rate: at least a third of patients die.28
Clinical criteria (community acquisition, no primary focus, and metastatic sequelae) were developed to try to predict the risk of endocarditis in bacteremic patients.26 However, these criteria did not work very well. The clinical definition of endocarditis has evolved. The criteria of von Reyn et al29 from 1981 did not use echocardiography as part of the definition, but the 1994 Duke criteria,30 which were refined31 in 2000, use both clinical and echocardiographic parameters.
Stratton et al32 performed transthoracic echocardiography in 14 patients with bacteremia and found 1 patient with cryptic tricuspid infective endocarditis. Bayer et al33 subsequently reported that of 72 patients with bacteremia, 6 (18%) of those who had no clinical findings suggestive of infectious endocarditis had findings on echocardiography that led to changes in their regimen. Adding echocardiography to three clinical risk factors increased the sensitivity of diagnosing endocarditis from 70% to 85% with a specificity of 100% and predictive value of 96%.
The Duke criteria call for transesophageal echocardiography, which is not feasible in some patients, eg, those with cirrhosis and esophageal varices.
S aureus endocarditis has changed over the years as our patient population has changed, and MRSA endocarditis tends to hit some of our most vulnerable patients. In a study by Miro et al34 in 2005, MRSA was the leading pathogen in patients who were diagnosed with S aureus endocarditis in 1990 or later. We will only see these numbers go up. Patients with diabetes tend to have more MRSA, and of diabetic patients with MRSA endocarditis, 30% to 40% die in the hospital.
Indications for surgery
Certain conditions are indications for surgery among patients with endocarditis, and no antibiotic will cure the endocarditis if the patient has one of these conditions, eg:
- Persistent bacteremia during antibiotic therapy
- Recurrent emboli
- Heart failure that cannot be controlled
- Perivalvular or myocardial abscesses
- Large vegetations
- Early prosthetic valve infection
- Certain arrhythmias.
How long should S aureus bacteremia be treated?
In cases of bacteremia in which endocarditis has been ruled out and removable foci of infection (eg, intravascular catheters) have been removed, some evidence indicates that treatment for 2 weeks would be as effective as the 4 to 6 weeks that we would use for endocarditis or other severe or invasive infections.35 The issue is controversial. If the patient has had frequent hospitalizations or a chronic medical condition I would hesitate to treat for less than 4 weeks, even if the infection appears to be associated with a removable focus.
Treatment of endocarditis
In the guidelines for treatment of endocarditis from the American Heart Association and Infectious Diseases Society of America,36 all the recommendations are relatively old and many of them are somewhat empiric—they are not based on evidence from randomized clinical trials. Rather, they are best opinions based on clinical experience and some observational studies over the years.
For MSSA. In cases of native-valve endocarditis, oxacillin (Bactocill), nafcillin (Unipen), or another semisynthetic beta-lactam antibiotic is recommended. For penicillin-allergic patients, we have other options, such as cefazolin (Ancef, Kefzol).
Combination therapy is frequently recommended for native valve endocarditis as well as for prosthetic valve endocarditis, with either rifampin or gentamicin along with a primary agent. There is some evidence that one can clear staphylococcal bacteremia a day or two more quickly by use of combination therapy with nafcillin plus an aminoglycoside than with nafcillin alone.37,38 For MSSA-associated endocarditis, vancomycin does not work as well as beta-lactam antibiotics.39,40
Korzeniowski and Sande37 and Chambers et al38 reported that the mean duration of bacteremia was 3.4 days for patients treated with nafcillin alone and 2.9 days for those treated with nafcillin plus an aminoglycoside. These studies led to consideration of a short course of gentamicin to clear the bacteremia quickly.
With MRSA, bacteremia often requires a week or more to clear. Levine et al21 reported a study in 42 patients, mostly injection-drug users, with right-sided native-valve endocarditis. The median duration of bacteremia was 7 days in patients who received vancomycin alone vs 9 days in those who received vancomycin plus rifampin; however, some patients were bacteremic for up to 27 days. Fever persisted for a median of 7 days, probably partly due to septic pulmonary emboli. Three patients died, and three required valve replacement.
NEW ANTIBIOTICS
Several new antibiotics are active against gram-positive cocci.41–44 However, the majority of them have not been prospectively studied for treating bacteremia or endocarditis.
Quinupristin/dalfopristin (Synercid) has not been formally studied for treatment of MRSA bacteremia or endocarditis. There are a few case reports of its use in these conditions.45 Quinupristin/dalfopristin is bacteriostatic, and its use may be associated with phlebitis, myalgias, and arthralgias.46
Linezolid (Zyvox) is approved for treatment of complicated skin and soft-tissue infections and for hospital-acquired pneumonia. There have been no specific studies of linezolid in the treatment of S aureus bacteremia or endocarditis. However, Shorr et al47 retrospectively looked at the bacteremic patients in five previous studies of linezolid vs vancomycin and found 144 cases of S aureus bacteremia, half of which were due to MRSA. Of 53 assessable patients with MRSA bacteremia, the primary infection was cured in 14 (56%) of the linezolid patients and 13 (46%) of the vancomycin patients.
The oral form is 100% bioavailable. One should avoid concomitant use of serotonin-reuptake inhibitors because of the risk of serotonin syndrome. Adverse effects include altered taste sensation and peripheral neuropathy. There are other potential toxicities, including hematologic changes (thrombocytopenia, leukopenia) and metabolic effects (lactic acidosis), so clinical and laboratory monitoring is important.48 The role of linezolid in the treatment of patients with S aureus bacteremia or endocarditis remains to be defined.
Daptomycin is indicated for complicated skin and soft-tissue infections, bacteremia, and right-sided endocarditis due to S aureus. Fowler et al20 found that daptomycin was not inferior to beta-lactam antibiotics for treatment of MSSA bacteremia and right-sided endocarditis, and for MRSA infections it outperformed vancomycin, but the difference was not statistically significant.
The dosing interval should be increased from once every 24 hours to every 48 hours if the creatinine clearance is 30 mL/minute or less. Adverse effects include myalgia, rhabdomyolysis (rare), and elevations in creatine phosphokinase. Reports of rising MICs during daptomycin therapy, in some cases associated with persistent infection,49 suggest that careful attention be paid to dosing and clinical monitoring.
Tigecycline (Tygacil) is indicated for complicated skin and soft-tissue infections and complicated intra-abdominal infections due to susceptible organisms. It is active against both MSSA and MRSA, but clinical experience with its use in invasive infections is somewhat limited.50 The dose of tigecycline should be reduced in advanced cirrhosis. Adverse effects include nausea and vomiting.
Telavancin, dalbavancin, and oritavancin, investigational parenteral antibiotics that are derivatives of vancomycin, are in clinical trials. The pharmacokinetic activity of these agents is of interest: telavancin is being studied with a once-daily dosing interval and dalbavancin’s half-life allows once-weekly dosing. In a limited trial, dalbavancin was found to be safe and effective in the treatment of catheter-related bloodstream infections.51 None of the antibiotics in this group has been studied for treatment of S aureus endocarditis. Telavancin therapy has been associated with rash, hypokalemia, QT prolongation, and creatinine elevations. Gastrointestinal symptoms have been reported with the use of dalbavancin.
Ceftobiprole, another investigational agent, is the only cephalosporin antibiotic that is active against MRSA. It is given every 12 hours. Adverse effects include nausea and taste disturbance.
Iclaprim is a novel diaminopyrimidine and a dihydrofolate reductase inhibitor. In vitro, it is active against gram-positive bacteria, including MRSA, VISA, and VRSA; clinical investigations at this point are limited to the treatment of skin and soft-tissue infections.
- Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 2004; 39:309–371. Erratum in: Clin Infect Dis 2004; 39:1093.
- US Centers for Disease Control and Prevention. National Nosocomial Infections Surveillance (NNIS) System. Campaign to prevent antimicrobial resistance. www.cdc.gov/drugresistance/healthcare/ha/HASlideSet.ppt.
- Blot SI, Vandewoude KH, Hoste EA, Colardyn FA. Outcome and attributable mortality in critically ill patients with bacteremia involving methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Arch Intern Med 2002; 162:2229–2235.
- Klevens RM, Morrison MA, Nadle J, et al; Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA 2007; 298:1763–1771.
- Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003; 290:2976–2984.
- Miller LG, Perdreau-Remington F, Rieg G, et al. Necrotizing fasciitis caused by community-associated methicillin-resistant Staphylococcus aureus in Los Angeles. N Engl J Med 2005; 352:1445–1453.
- Moran GJ, Krishnadasan A, Gorwitz RJ, et al EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006; 355:666–674.
- Mishaan AM, Mason EO, Martinez-Aquilar G, et al. Emergence of a predominant clone of community-acquired Staphylococcus aureus among children in Houston, Texas. Pediatr Infect Dis J 2005; 24:201–206.
- Miller LG, Perdreau-Remington F, Bayer AS, et al. Clinical and epidemiologic characteristics cannot distinguish community-associated methicillin-resistant Staphylococcus aureus infection from methicillin-susceptible S. aureus infection: a prospective investigation. Clin Infect Dis 2007; 44:471–482.
- Seybold U, Kourbatova EV, Johnson JG, et al. Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care-associated blood stream infections. Clin Infect Dis 2006; 42:647–656.
- Maree CL, Daum RS, Boyle-Vavra S, Matayoshi K, Miller LG. Community-associated methicillin-resistant Staphylococcus aureus isolates causing healthcare-associated infections. Emerg Infect Dis 2007; 13:236–242.
- Liu C, Chambers HF. Staphylococcus aureus with heterogeneous resistance to vancomycin: epidemiology, clinical significance, and critical assessment of diagnostic methods. Antimicrob Agents Chemother 2003; 47:3040–3045.
- Sieradzki K, Roberts RB, Haber SW, Tomasz A. The development of vancomycin resistance in a patient with methicillin-resistant Staphylococcus aureus infection. N Engl J Med 1999; 340:517–523.
- Schwaber MJ, Wright SB, Carmeli Y, et al. Clinical implications of varying degrees of vancomycin susceptibility in methicillin-resistant Staphylococcus aureus bacteremia. Emerg Infect Dis 2003; 9:657–664. Erratum in: Emerg Infect Dis 2004; 10:160.
- Sakoulas G, Moise-Broder PA, Schentag J, Forrest A, Moellering RC, Eliopoulos GM. Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin-resistant Staphylococcus aureus bacteremia. J Clin Microbiol 2004; 42:2398–2402.
- Naimi TS, Anderson D, O’Boyle C, et al. Vancomycin-intermediate Staphylococcus aureus with phenotypic susceptibility to methicillin in a patient with recurrent bacteremia. Clin Infect Dis 2003; 36:1609–1612.
- Moore MR, Perdreau-Remington F, Chambers HF. Vancomycin treatment failure associated with heterogeneous vancomycin-intermediate Staphylococcus aureus in a patient with endocarditis and in the rabbit model of endocarditis. Antimicrob Agents Chemother 2003; 47:1262–1266.
- Charles PG, Ward PB, Johnson PD, Howden BP, Grayson ML. Clinical features associated with bacteremia due to heterogenous vancomycin-intermediate Staphylococcus aureus. Clin Infect Dis 2004; 38:448–451.
- Howden BP, Ward PB, Charles PG, et al. Treatment outcomes for serious infections caused by methicillin-resistant Staphylococcus aureus with reduced vancomycin susceptibility. Clin Infect Dis 2004; 38:521–528.
- Fowler VG, Boucher HW, Corey GR, et al. S. aureus Endocarditis and Bacteremia Study Group. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med 2006; 355:653–665.
- Levine DP, Fromm BS, Reddy BR. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann Intern Med 1991; 115:674–680.
- Fowler VG, Sakoulas G, McIntyre LM, et al. Persistent bacteremia due to methicillin-resistant Staphylococcus aureus infection is associated with agr dysfunction and low-level in vitro resistance to thrombin-induced platelet microbicidal protein. J Infect Dis 2004; 190:1140–1149.
- Chang FY, Peacock JE, Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore) 2003; 82:333–339.
- Hidayat LK, Hsu DI, Quist R, Shriner KA, Wong-Beringer A. High-dose vancomycin therapy for methicillin-resistant Staphylococcus aureus infections: efficacy and toxicity. Arch Intern Med 2006; 166:2138–2144.
- Wilson R, Hamburger M. Fifteen years’ experience with staphylococcus septicemia in a large city hospital; analysis of fifty-five cases in the Cincinnati General Hospital 1940 to 1954. Am J Med 1957; 22:437–457.
- Nolan CM, Beaty HN. Staphylococcus aureus bacteremia. Current clinical patterns. Am J Med 1976; 60:495–500.
- Shah M, Watanakunakorn C. Changing patterns of Staphylococcus aureus bacteremia. Am J Med Sci 1979; 278:115–121.
- Fowler VG, Miro JM, Hoen B, et al ICE Investigators. Staphylococcus aureus endocarditis: a consequence of medical progress. JAMA 2005; 293:3012–3021. Erratum in: JAMA 2005; 294:900.
- Von Reyn CF, Levy BS, Arbeit RD, Friedland G, Crumpacker CS. Infective endocarditis: an analysis based on strict case definition. Ann Intern Med 1981; 94:505–518.
- Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med 1994; 96:200–209.
- Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 2000; 30:633–638.
- Stratton JR, Werner JA, Pearlman AS, Janko CL, Kliman S, Jackson MC. Bacteremia and the heart. Serial echocardiographic findings in 80 patients with documented or suspected bacteremia. Am J Med 1982; 73:851–858.
- Bayer AS, Lam K, Ginzton L, Normal DC, Chiu CY, Ward JI. Staphylococcus aureus bacteremia. Clinical, serologic, and echocardiographic findings in patients with and without endocarditis. Arch Intern Med 1987; 147:457–462.
- Miro JM, Anguera I, Cabell CH, et al International Collaboration on Endocarditis Merged Database Study Group. Staphylococcus aureus native valve infective endocarditis: report of 566 episodes from the International Collaboration on Endocarditis Merged Database. Clin Infect Dis 2005; 41:507–514. Erratum in: Clin Infect Dis 2005; 41:1075–1077.
- Jernigan JA, Farr BM. Short-course therapy of catheter-related Staphylococcus aureus bacteremia: a meta-analysis. Ann Intern Med 1993; 119:304–311.
- Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America. Circulation 2005; 111:e394–e434. Erratum in: Circulation 2005; 112:2373. Circulation 2007; 115:e408.
- Korzeniowski O, Sande MA. Combination antimicrobial therapy for Staphylococcus aureus endocarditis in patients addicted to parenteral drugs and in nonaddicts: a prospective study. Ann Intern Med 1982; 97:496–503.
- Chambers HF, Korzeniowski OM, Sande MA. Staphylococcus aureus endocarditis: clinical manifestations in addicts and nonaddicts. Medicine (Baltimore) 1983; 62:170–177.
- Gentry CA, Rodvold KA, Novak RM, Hershow RC, Naderer OJ. Retrospective evaluation of therapies for Staphylococcus aureus endocarditis. Pharmacotherapy 1997; 17:990–997.
- Small PM, Chambers HF. Vancomycin for Staphylococcus aureus endocarditis in intravenous drug users. Antimicrob Agents Chemother 1990; 34:1227–1231.
- Eliopoulos GM. Quinupristin-dalfopristin and linezolid: evidence and opinion. Clin Infect Dis 2003; 36:473–481.
- Rybak MJ. Therapeutic options for Gram-positive infections. J Hosp Infect 2001; 49 suppl A:S25–S32.
- Micek ST. Alternatives to vancomycin for the treatment of methicillin-resistant Staphylococcus aureus infections. Clin Infect Dis 2007; 45 suppl 3:S184–S190.
- Appelbaum PC, Jacobs MR. Recently approved and investigational antibiotics for treatment of severe infections caused by Gram-positive bacteria. Curr Opin Microbiol 2005; 8:510–517.
- Drew RH, Perfect JR, Srinath L, Kirkimilis E, Dowzicky M, Talbot GH for the Synercid Emergency-Use Study Group. Treatment of methicillin-resistant Staphylococcus aureus infections with quinupristin-dalfopristin in patients intolerant of or failing prior therapy. J Antimicrob Chemother 2000; 46:775–784.
- Lamb HM, Figgitt DP, Faulds D. Quinupristin/dalfopristin: a review of its use in the management of serious gram-positive infections. Drugs 1999; 58:1061–1097.
- Shorr AF, Kunkel MJ, Kollef M. Linezolid versus vancomycin for Staphylococcus aureus bacteraemia: pooled analysis of randomized studies. J Antimicrob Chemother 2005; 56:923–929.
- Bishop E, Melvani S, Howden BP, Charles PG, Grayson ML. Good clinical outcomes but high rates of adverse reactions during linezolid therapy for serious infections: a proposed protocol for monitoring therapy in complex patients. Antimicrob Agents Chemother 2006; 50:1599–1602.
- Boucher HW, Sakoulas G. Perspectives on daptomycin resistance, with emphasis on resistance in Staphylococcus aureus. Clin Infect Dis 2007; 45:601–608.
- Munoz-Price LS, Lolans K, Quinn JP. Four cases of invasive methicillin-resistant Staphylococcus aureus (MRSA) infections treated with tigecycline. Scand J Infect Dis 2006; 38:1081–1084.
- Raad I, Darouiche R, Vazquez J, et al. Efficacy and safety of weekly dalbavancin therapy for catheter-related bloodstream infection caused by gram-positive pathogens. Clin Infect Dis 2005; 40:374–80.
Staphylococcus aureus is rearing its ugly head in new and interesting ways, both in the hospital and in the community.
Rates of invasive infections with methicillin-resistant S aureus (MRSA) have been increasing both in the hospital and in the community, a trend that has attracted considerable interest in the lay media. Curiously, the most common community-associated MRSA strain, which up to now has been distinct from hospital-associated MRSA strains, is invading our hospitals. Alarmingly, vancomycin (Vancocin), the drug of last resort for MRSA infections for the past 40 years, does not seem to be as effective as it used to be.
This paper summarizes the changing epidemiology of S aureus, particularly the emergence of MRSA outside of the hospital; reviews the difficulties associated with S aureus bacteremia and its treatment in view of; some changes in vancomycin susceptibility; and appraises the old and new treatment options.
MRSA IS ON THE RISE IN THE HOSPITAL
S aureus, a gram-positive, coagulase-positive bacterium, is one of the leading nosocomial bloodstream pathogens, second only to coagulase-negative staphylococci.1 And the incidence of S aureus infections is increasing. MRSA in particular is increasingly causing infections throughout hospitals, including intensive care units. As of 2004, nearly two-thirds of isolates of S aureus from intensive care units were MRSA.2
MRSA infections are worse than methicillin-susceptible S aureus (MSSA) infections in terms of the rates of death and other undesirable outcomes.3 Several factors may be responsible: MRSA infection may be a marker of severity of illness (sicker patients may be more likely to have MRSA), our treatment for MRSA may not be as effective as it is for MSSA, and the organism may be inherently more virulent.
METHICILLIN RESISTANCE IS ALSO ON THE RISE IN THE COMMUNITY
Community-associated MRSA began emerging clinically about 10 years ago. It was first described in a cohort of children with necrotizing pneumonia in Minnesota, but soon other populations at risk began to emerge, such as residents of correctional facilities, men who had sex with men, competitive athletes (eg, fencers, wrestlers, and football players), and Alaskan natives and other native populations. A common factor in all these groups was close proximity of the members to each other. Later, it began to spread beyond these traditional risk groups into the community at large.
Community-associated MRSA strains have a characteristic pattern of antimicrobial susceptibility (see below). In the laboratory, they grow somewhat faster than health-care-associated MRSA strains, but not as fast as MSSA. They have a strong association with skin and soft-tissue infections: when you see a skin or soft-tissue infection, be it in an outpatient or an inpatient, think about MRSA. Their virulence varies, but rapid onset and progression of illness are quite common. Their most common strain in the United States at present is USA 300.
Case 1: A young woman with necrotizing fasciitis
A 21-year-old college student presented to our service in May 2004 with high fever and severe arm pain, which had been worsening for several days. She had been previously healthy, had not had any contact with the health care system, and had not received any antibiotics.
Her blood cultures were positive for MRSA, as were cultures of the deep tissue of the deltoid muscle and fascia when she underwent emergency surgical debridement. The infection required several additional surgical debridements and removal of one head of her deltoid muscle, but she was fortunate: in the past, some patients with this problem might have undergone radical amputation of the arm or even more extensive surgery. This patient continued to have positive blood cultures 4 days postoperatively, but she ultimately recovered, completing 28 days of daptomycin (Cubicin) therapy at a dose of 6 mg/kg every 24 hours. The last 10 days of daptomycin therapy were given at home via a percutaneous intravenous central catheter.
Comment. The epidemiology of MRSA infections is changing. More patients who have no traditional risk factors, specifically health care contact, are getting MRSA infections. A recent report from the US Centers for Disease Control and Prevention (CDC) indicates that the proportion of patients with invasive disease due to MRSA has doubled since 2001–2002.4 Part of the reason undoubtedly is that MRSA, particularly community-associated MRSA, often carries specific virulence factors that make it more invasive. The CDC estimated that in 2005 there were nearly 100,000 cases of invasive MRSA infection in the United States, and nearly a fifth of these infections resulted in death.
Resistance and virulence factors in community-associated MRSA
Most community-associated MRSA strains carry a mobile genetic element called type IV SCCmec (staphylococcal chromosomal cassettemec) that enhances its antimicrobial resistance. This genetic component was probably borrowed from coagulase-negative staphylococci, in which it is quite common but does not cause as much of a problem. It is now present in a wide range of S aureus strains. Most of the S aureus strains that carry type IV SCCmec are MRSA, but a few MSSA strains do carry it as well.
The potent toxin Panton-Valentine leukocidin is an extracellular product that is detected in fewer than 5% of hospital strains but is more common in community-associated strains. It kills leukocytes by forming pores in the cell membrane and causing skin necrosis in cutaneous infections. It is associated with skin abscesses and rapidly progressive necrotizing pneumonia in MSSA or MRSA.
Epidemiologic differences between community- and health-care-associated MRSA
Patients with community-associated MRSA infections tend to be younger than those who traditionally get health-care-associated MRSA infections: in a study from Naimi et al in 2003, the mean ages were 23 vs 68 years.5 A greater proportion of patients with community-associated MRSA strains are nonwhite.4,5
Most community-associated MRSA infections are of the skin and soft tissue (75% in the series from Naimi et al5), but this pathogen causes other infections as well. Bacteremia of unknown origin has been seen, as has necrotizing pneumonia. Most of the skin and soft-tissue infections are relatively superficial, such as folliculitis or furunculosis, but deeper tissue infections such as necrotizing fasciitis and pyomyositis have also been seen.6
The incidence of community-associated MRSA infections varies greatly by geographic region.7 The northeastern United States has so far been relatively spared, but in Atlanta, Houston, and Los Angeles up to 80% of cases of characteristic skin or soft-tissue infections seen in emergency or outpatient departments are due to community-associated MRSA. Physicians at the Texas Children’s Hospital in Houston assume that all skin or soft-tissue infections are due to community-associated MRSA unless proven otherwise.8
Differences in antibiotic susceptibility
Community-associated MRSA is more susceptible to various antibiotics than health-care-associated MRSA,5 but not by much. Strains are usually susceptible to vancomycin, tetracyclines, trimethoprim-sulfamethoxazole (Bactrim, Septra), and rifampin (Rifadin). Unlike hospital strains, a fair number of community-acquired strains are susceptible to clindamycin (Cleocin) in the laboratory, but with a caveat: some of these clindamycin-susceptible strains actually may harbor the tools for inducible resistance. In fact, they can become resistant to clindamycin even without being exposed to it.
The laboratory test for inducible clindamycin resistance is called the D test. After coating an agar plate with S aureus, the technician places erythromycin and clindamycin disks. If the erythromycin induces clindamycin resistance, the plate is clear of growth around the clindamycin disk except for the portion nearest the erythromycin disk, leaving a characteristic D-shaped area of lucency.
Risk factors for MRSA
Moran et al7 analyzed the risk factors for community-associated MRSA in patients with skin or soft-tissue infections seen in the emergency department. The infection was more likely to be due to community-associated MRSA if the patient was black, had used any antibiotic in the past month, had a history of MRSA infection, or had close contact with a person with a similar infection. Many patients interpreted the infections as spider bites because the lesions tended to have a dark center surrounded by a tender area. These infections were not associated with underlying illness. In some cases, community-associated MRSA skin infections have been associated with tattooing and even manicuring.
However, it is very difficult to distinguish between community-associated MRSA and MSSA skin and soft-tissue infections on the basis of clinical and epidemiologic characteristics. Miller et al9 studied a large group of patients in Los Angeles who were hospitalized with community-associated skin and soft-tissue S aureus infections. All the patients were followed up for 30 days after hospital discharge. Regardless of whether they had MRSA or MSSA, they had similar outcomes. Close contacts of the patients also tended to develop infection.
A key point from this and many other studies: patients were more likely to remain infected if they did not undergo incision and drainage. This key intervention is indicated for any patient who has a skin and soft-tissue infection with an undrained focus of infection.
COMMUNITY-ASSOCIATED MRSA IS INVADING THE HOSPITAL
In a new development, community-associated MRSA strains are now appearing in the hospital. This is not only because patients are bacteremic when they come in: patients in the hospital are getting nosocomial infections due to community-associated MRSA strains.
Seybold et al10 analyzed 116 cases of MRSA bloodstream infections in Atlanta, GA. In 9 (8%) of the cases the patient had not had any contact with the health care system within the past year, and these cases were classified as truly community-associated. Of the remaining 107 cases, 49 (42%) were nosocomial, and the USA 300 strain—the predominant community-associated MRSA strain—accounted for 10 (20%) of the nosocomial cases.
In the recent CDC study of invasive MRSA infections, Klevens et al4 reported that nearly a third of cases of bacteremia were due to community-associated MRSA, and these strains accounted for a greater proportion of cases of cellulitis and endocarditis than did health-care-associated strains.
In a study of hospital-associated MRSA, Maree et al11 found that the percentage of cases in which the bacteria carried the SCCmec type IV marker had increased from less than 20% in 1999 to more than 50% in 2004.
Comment. Suffice it to say that we are surrounded by MRSA. Community-associated MRSA is here to stay. It is even invading our hospitals, and we need to consider this very carefully when choosing antimicrobial therapy.
NAGGING QUESTIONS ABOUT VANCOMYCIN
Case 2: Vancomycin-intermediate S aureus (VISA) bacteremia and endocarditis
In December 2006 we saw a very ill 60-year-old woman who was hospitalized with MRSA bacteremia, pacemaker endocarditis, and superior vena cava thrombosis. Although she was treated with vancomycin and rifampin, her condition worsened, she had a stroke, and she developed renal failure. In a difficult operation, the pacemaker was removed, but the bacteremia persisted. In early February 2007 she underwent another difficult operation in which the superior vena cava clot was debrided, a right atrial clot was removed, and her mitral valve was replaced. Less than 2 weeks later, and despite ongoing vancomycin and rifampin therapy, the MRSA bacteremia recurred.
During the approximately 6 weeks that the patient had been receiving these antibiotics, the minimal inhibitory concentration (MIC) of rifampin against the S aureus isolate increased from less than 1 μg/mL (susceptible) to 2 μg/mL (resistant). The MIC of vancomycin went from 2 μg/mL (susceptible) to 4 μg/mL (intermediately susceptible). Vancomycin and rifampin were discontinued, and daptomycin and gentamicin (Garamycin) therapy were started. (Her daptomycin MIC was 0.5 μg/mL). The patient’s condition stabilized, and she was discharged to a long-term nursing facility. She had no relapse of MRSA bacteremia, but she died in early April of that year.
Is vancomycin becoming less effective? Degrees of vancomycin resistance
Vancomycin has been our stalwart for treating MRSA infections for more than 40 years but it is not working as well as it used to, at least in certain situations.
VRSA (vancomycin-resistant S aureus) is rare. These fully resistant strains probably acquired a resistance mechanism (the vanA operon) from vancomycin-resistant enterococci. Infections tend to occur in patients simultaneously infected with both S aureus and vancomycin-resistant enterococci, giving the bacteria an opportunity to exchange genetic material.
VISA (vancomycin-intermediate S aureus) infections tend to occur in patients like the one described above who have had long-term vancomycin therapy. VISA strains appear to overproduce a matrix that captures vancomycin and keeps it from entering the cell. On electron microscopy, these bacteria have a very thick cell wall.13
Vancomycin tolerance is a state in which the bacteria are “stunned” or kept in check but not killed by vancomycin. That is manifested in the laboratory by a ratio of minimum bactericidal concentration to MIC greater than 32.
hVISA (heteroresistant VISA) is new and worrisome. These organisms have an overall MIC in the susceptible range, but within that population are individual isolates with an MIC that is much higher—in the intermediate or perhaps even in the resistant range.14
Reported rates of hVISA vary from less than 2% to as high as 76%, because the methods for detecting it are still very poorly standardized. The usual automated laboratory tests do not detect hVISA.
hVISA is probably clinically relevant, as evidence is emerging both in vitro and in vivo that the higher the MIC for vancomycin, the worse the clinical outcome.15 hVISA has been associated with failures of therapy in several situations, usually in cases of severe invasive or deep infection, endocarditis, and bacteremia with vertebral osteomyelitis where vancomycin concentrations at the site of infection may be suboptimal.16–19 While most hVISA strains that have been described were resistant to methicillin, some were susceptible.
The E test is emerging as the standard test for hVISA. This test uses a plastic strip that contains gradually increasing concentrations of vancomycin along its length. Placed in the culture dish, the strip inhibits growth of the organism at its high-concentration end but not at its low-concentration end. If the sample contains hVISA, the cutoff is not well defined, with a few colonies growing at higher concentrations.
New definition of vancomycin susceptibility
Recognizing that the MICs for vancomycin have been rising in the last few years, the Clinical and Laboratory Standards Institute last year changed the break points between susceptibility and resistance. The new definitions are:
- Susceptible—an MIC of 2.0 μg/mL or less (formerly 4.0 μg/mL or less)
- Intermediate—4.0 to 8.0 μg/mL (formerly 8.0 to 16 μg/mL)
- Resistant—16 μg/mL or greater (formerly 32 μg/mL or greater).
One should pay attention to the MIC numbers on the laboratory reports, not just to the words “susceptible” or “not susceptible.” If the number is, say, 0.5 μg/mL or less, the organism should really be susceptible. If the number is 1 or 2, it is still in the susceptible range, but those are the organisms that may cause problems later on.
Further, even if the vancomycin MIC is in the susceptible range, higher MICs may affect outcomes. The average duration of MRSA bacteremia on therapy is 8 to 9 days, vs 3 to 4 days with MSSA bacteremia.20,21 But Sakoulas et al15 found that, in MRSA bacteremia, the success rate with vancomycin therapy was 56% if the MIC was 0.5 or lower, compared with 10% if the MIC was 1.0 to 2.0 μg/mL. Examined in another way, the success rate was 50% if the logarithm of killing was 6.27 colony-forming units per mL or greater, 23% if 4.71 to 6.26, and zero if less than 4.71.
Case 3: Prolonged MRSA bacteremia
In the summer of 2006, a 66-year-old woman with a history of gastric bypass and cirrhosis underwent a long stay in the surgical intensive care unit because of a recurrent enterocutaneous fistula and chronic renal insufficiency. On November 5th, she had a positive blood culture for MRSA, which was treated appropriately with vancomycin for 4 weeks. She was discharged to subacute care but came back 2 days later, again with MRSA bacteremia. At that time her Hickman catheter, which had been inserted for total parenteral nutrition because of the enterocutaneous fistula, was removed.
Transthoracic echocardiography revealed no vegetations, but her bacteremia persisted. Her mental status was poor this entire time: she was mute and could barely be awakened. We looked for clots and infected clots; duplex ultrasonographic examinations of all four extremities were negative. Finally, magnetic resonance imaging of her back—performed empirically because of the persistent bacteremia—revealed vertebral osteomyelitis at level T12-L1. We also noticed on serial evaluations that the vancomycin MIC for her organism increased from 0.5 to 2.0 μg/mL, so therapy was changed from vancomycin to daptomycin.
Her bacteremia cleared. Follow-up echocardiography was negative, but she had two subsequent relapses of MRSA bacteremia, one in April 2007 and one before she died in the summer of 2007.
Prolonged bacteremia: Is it vancomycin resistance, or something else?
The MRSA isolates that cause prolonged bacteremia seem to have certain characteristics.22 Higher MICs are probably associated with longer periods of bacteremia. But some genetic components within some strains of S aureus give them a survival advantage. They have less susceptibility to the body’s thrombin-induced platelet microbicidal protein. These isolates are not only associated with prolonged bacteremia: they are also associated with osteomyelitis, deep abscesses, endocarditis, recurrent infection, and increased death rate.22 Clinical laboratories do not test for these genetic components. One wonders whether our patient may have had an isolate with these mutations that gave it a survival advantage.
Do not use vancomycin for MSSA
Avoid using vancomycin for MSSA infections. It has been shown time and time again that MSSA infections do not respond as well to vancomycin as they do to beta-lactam antibiotics, specifically to the semisynthetic penicillins such as oxacillin and nafcillin, and even some of the first-generation cephalosporins. Chang et al23 found that patients with MSSA bacteremia had higher rates of persistent infections, relapse, and bacteriologic failure if they received vancomycin than if they received nafcillin.
Do vancomycin trough levels affect toxicity?
The vancomycin trough levels that we aimed for in the past (5 to 10 μg/mL) were probably too low. Today, we aim for trough levels of 15 to 20 μg/mL, and many physicians are aiming for 20 to 25 μg/mL. Part of the reason is that vancomycin MICs are higher than they used to be: in order to keep the vancomycin level above the MIC for a longer period of time, the vancomycin trough level needs to be higher. In theory, keeping the vancomycin levels above the MIC for longer periods should improve outcomes. Yet Fowler et al22 found that vancomycin trough levels among patients who had persistent MRSA bacteremia were actually higher than trough levels among those in whom the bacteremia resolved, although the difference was not statistically significant.
We measure the vancomycin trough level to make sure it is high enough (and give larger doses if it is not); among adults, peak levels need not be monitored on a routine basis because of the predictable pharmacokinetics of vancomycin.
Vancomycin toxicity can be either idiosyncratic or synergistic. Idiosyncratic toxicity occurs when a patient who has been on vancomycin for a long time develops a fixed rash, not associated with infusion. This is an immunologic phenomenon. It is a rare and very serious situation and may require steroid therapy.
Synergistic toxicity occurs when vancomycin is given with other nephrotoxic agents, notably gentamicin. Vancomycin plus gentamicin equals nephrotoxicity. Vancomycin alone is usually not nephrotoxic, but close monitoring of renal function parameters is warranted with the use of higher doses.24
IN UNEXPLAINED BACTEREMIA, LOOK FOR ENDOCARDITIS
In blood cultures from patients with bacteremia, S aureus is never a contaminant. Even if just one blood culture is positive for S aureus, believe that S aureus is the culprit.
Reports in the 1950s suggested that at least half of patients who had S aureus bacteremia had endocarditis,25 leading to recommendations that all patients with S aureus bacteremia without an obvious primary source of infection should be evaluated for endocarditis. Subsequent estimates were lower, in the range of 15% to 25%.26,27 However, throughout the world S aureus endocarditis continues to have a very high mortality rate: at least a third of patients die.28
Clinical criteria (community acquisition, no primary focus, and metastatic sequelae) were developed to try to predict the risk of endocarditis in bacteremic patients.26 However, these criteria did not work very well. The clinical definition of endocarditis has evolved. The criteria of von Reyn et al29 from 1981 did not use echocardiography as part of the definition, but the 1994 Duke criteria,30 which were refined31 in 2000, use both clinical and echocardiographic parameters.
Stratton et al32 performed transthoracic echocardiography in 14 patients with bacteremia and found 1 patient with cryptic tricuspid infective endocarditis. Bayer et al33 subsequently reported that of 72 patients with bacteremia, 6 (18%) of those who had no clinical findings suggestive of infectious endocarditis had findings on echocardiography that led to changes in their regimen. Adding echocardiography to three clinical risk factors increased the sensitivity of diagnosing endocarditis from 70% to 85% with a specificity of 100% and predictive value of 96%.
The Duke criteria call for transesophageal echocardiography, which is not feasible in some patients, eg, those with cirrhosis and esophageal varices.
S aureus endocarditis has changed over the years as our patient population has changed, and MRSA endocarditis tends to hit some of our most vulnerable patients. In a study by Miro et al34 in 2005, MRSA was the leading pathogen in patients who were diagnosed with S aureus endocarditis in 1990 or later. We will only see these numbers go up. Patients with diabetes tend to have more MRSA, and of diabetic patients with MRSA endocarditis, 30% to 40% die in the hospital.
Indications for surgery
Certain conditions are indications for surgery among patients with endocarditis, and no antibiotic will cure the endocarditis if the patient has one of these conditions, eg:
- Persistent bacteremia during antibiotic therapy
- Recurrent emboli
- Heart failure that cannot be controlled
- Perivalvular or myocardial abscesses
- Large vegetations
- Early prosthetic valve infection
- Certain arrhythmias.
How long should S aureus bacteremia be treated?
In cases of bacteremia in which endocarditis has been ruled out and removable foci of infection (eg, intravascular catheters) have been removed, some evidence indicates that treatment for 2 weeks would be as effective as the 4 to 6 weeks that we would use for endocarditis or other severe or invasive infections.35 The issue is controversial. If the patient has had frequent hospitalizations or a chronic medical condition I would hesitate to treat for less than 4 weeks, even if the infection appears to be associated with a removable focus.
Treatment of endocarditis
In the guidelines for treatment of endocarditis from the American Heart Association and Infectious Diseases Society of America,36 all the recommendations are relatively old and many of them are somewhat empiric—they are not based on evidence from randomized clinical trials. Rather, they are best opinions based on clinical experience and some observational studies over the years.
For MSSA. In cases of native-valve endocarditis, oxacillin (Bactocill), nafcillin (Unipen), or another semisynthetic beta-lactam antibiotic is recommended. For penicillin-allergic patients, we have other options, such as cefazolin (Ancef, Kefzol).
Combination therapy is frequently recommended for native valve endocarditis as well as for prosthetic valve endocarditis, with either rifampin or gentamicin along with a primary agent. There is some evidence that one can clear staphylococcal bacteremia a day or two more quickly by use of combination therapy with nafcillin plus an aminoglycoside than with nafcillin alone.37,38 For MSSA-associated endocarditis, vancomycin does not work as well as beta-lactam antibiotics.39,40
Korzeniowski and Sande37 and Chambers et al38 reported that the mean duration of bacteremia was 3.4 days for patients treated with nafcillin alone and 2.9 days for those treated with nafcillin plus an aminoglycoside. These studies led to consideration of a short course of gentamicin to clear the bacteremia quickly.
With MRSA, bacteremia often requires a week or more to clear. Levine et al21 reported a study in 42 patients, mostly injection-drug users, with right-sided native-valve endocarditis. The median duration of bacteremia was 7 days in patients who received vancomycin alone vs 9 days in those who received vancomycin plus rifampin; however, some patients were bacteremic for up to 27 days. Fever persisted for a median of 7 days, probably partly due to septic pulmonary emboli. Three patients died, and three required valve replacement.
NEW ANTIBIOTICS
Several new antibiotics are active against gram-positive cocci.41–44 However, the majority of them have not been prospectively studied for treating bacteremia or endocarditis.
Quinupristin/dalfopristin (Synercid) has not been formally studied for treatment of MRSA bacteremia or endocarditis. There are a few case reports of its use in these conditions.45 Quinupristin/dalfopristin is bacteriostatic, and its use may be associated with phlebitis, myalgias, and arthralgias.46
Linezolid (Zyvox) is approved for treatment of complicated skin and soft-tissue infections and for hospital-acquired pneumonia. There have been no specific studies of linezolid in the treatment of S aureus bacteremia or endocarditis. However, Shorr et al47 retrospectively looked at the bacteremic patients in five previous studies of linezolid vs vancomycin and found 144 cases of S aureus bacteremia, half of which were due to MRSA. Of 53 assessable patients with MRSA bacteremia, the primary infection was cured in 14 (56%) of the linezolid patients and 13 (46%) of the vancomycin patients.
The oral form is 100% bioavailable. One should avoid concomitant use of serotonin-reuptake inhibitors because of the risk of serotonin syndrome. Adverse effects include altered taste sensation and peripheral neuropathy. There are other potential toxicities, including hematologic changes (thrombocytopenia, leukopenia) and metabolic effects (lactic acidosis), so clinical and laboratory monitoring is important.48 The role of linezolid in the treatment of patients with S aureus bacteremia or endocarditis remains to be defined.
Daptomycin is indicated for complicated skin and soft-tissue infections, bacteremia, and right-sided endocarditis due to S aureus. Fowler et al20 found that daptomycin was not inferior to beta-lactam antibiotics for treatment of MSSA bacteremia and right-sided endocarditis, and for MRSA infections it outperformed vancomycin, but the difference was not statistically significant.
The dosing interval should be increased from once every 24 hours to every 48 hours if the creatinine clearance is 30 mL/minute or less. Adverse effects include myalgia, rhabdomyolysis (rare), and elevations in creatine phosphokinase. Reports of rising MICs during daptomycin therapy, in some cases associated with persistent infection,49 suggest that careful attention be paid to dosing and clinical monitoring.
Tigecycline (Tygacil) is indicated for complicated skin and soft-tissue infections and complicated intra-abdominal infections due to susceptible organisms. It is active against both MSSA and MRSA, but clinical experience with its use in invasive infections is somewhat limited.50 The dose of tigecycline should be reduced in advanced cirrhosis. Adverse effects include nausea and vomiting.
Telavancin, dalbavancin, and oritavancin, investigational parenteral antibiotics that are derivatives of vancomycin, are in clinical trials. The pharmacokinetic activity of these agents is of interest: telavancin is being studied with a once-daily dosing interval and dalbavancin’s half-life allows once-weekly dosing. In a limited trial, dalbavancin was found to be safe and effective in the treatment of catheter-related bloodstream infections.51 None of the antibiotics in this group has been studied for treatment of S aureus endocarditis. Telavancin therapy has been associated with rash, hypokalemia, QT prolongation, and creatinine elevations. Gastrointestinal symptoms have been reported with the use of dalbavancin.
Ceftobiprole, another investigational agent, is the only cephalosporin antibiotic that is active against MRSA. It is given every 12 hours. Adverse effects include nausea and taste disturbance.
Iclaprim is a novel diaminopyrimidine and a dihydrofolate reductase inhibitor. In vitro, it is active against gram-positive bacteria, including MRSA, VISA, and VRSA; clinical investigations at this point are limited to the treatment of skin and soft-tissue infections.
Staphylococcus aureus is rearing its ugly head in new and interesting ways, both in the hospital and in the community.
Rates of invasive infections with methicillin-resistant S aureus (MRSA) have been increasing both in the hospital and in the community, a trend that has attracted considerable interest in the lay media. Curiously, the most common community-associated MRSA strain, which up to now has been distinct from hospital-associated MRSA strains, is invading our hospitals. Alarmingly, vancomycin (Vancocin), the drug of last resort for MRSA infections for the past 40 years, does not seem to be as effective as it used to be.
This paper summarizes the changing epidemiology of S aureus, particularly the emergence of MRSA outside of the hospital; reviews the difficulties associated with S aureus bacteremia and its treatment in view of; some changes in vancomycin susceptibility; and appraises the old and new treatment options.
MRSA IS ON THE RISE IN THE HOSPITAL
S aureus, a gram-positive, coagulase-positive bacterium, is one of the leading nosocomial bloodstream pathogens, second only to coagulase-negative staphylococci.1 And the incidence of S aureus infections is increasing. MRSA in particular is increasingly causing infections throughout hospitals, including intensive care units. As of 2004, nearly two-thirds of isolates of S aureus from intensive care units were MRSA.2
MRSA infections are worse than methicillin-susceptible S aureus (MSSA) infections in terms of the rates of death and other undesirable outcomes.3 Several factors may be responsible: MRSA infection may be a marker of severity of illness (sicker patients may be more likely to have MRSA), our treatment for MRSA may not be as effective as it is for MSSA, and the organism may be inherently more virulent.
METHICILLIN RESISTANCE IS ALSO ON THE RISE IN THE COMMUNITY
Community-associated MRSA began emerging clinically about 10 years ago. It was first described in a cohort of children with necrotizing pneumonia in Minnesota, but soon other populations at risk began to emerge, such as residents of correctional facilities, men who had sex with men, competitive athletes (eg, fencers, wrestlers, and football players), and Alaskan natives and other native populations. A common factor in all these groups was close proximity of the members to each other. Later, it began to spread beyond these traditional risk groups into the community at large.
Community-associated MRSA strains have a characteristic pattern of antimicrobial susceptibility (see below). In the laboratory, they grow somewhat faster than health-care-associated MRSA strains, but not as fast as MSSA. They have a strong association with skin and soft-tissue infections: when you see a skin or soft-tissue infection, be it in an outpatient or an inpatient, think about MRSA. Their virulence varies, but rapid onset and progression of illness are quite common. Their most common strain in the United States at present is USA 300.
Case 1: A young woman with necrotizing fasciitis
A 21-year-old college student presented to our service in May 2004 with high fever and severe arm pain, which had been worsening for several days. She had been previously healthy, had not had any contact with the health care system, and had not received any antibiotics.
Her blood cultures were positive for MRSA, as were cultures of the deep tissue of the deltoid muscle and fascia when she underwent emergency surgical debridement. The infection required several additional surgical debridements and removal of one head of her deltoid muscle, but she was fortunate: in the past, some patients with this problem might have undergone radical amputation of the arm or even more extensive surgery. This patient continued to have positive blood cultures 4 days postoperatively, but she ultimately recovered, completing 28 days of daptomycin (Cubicin) therapy at a dose of 6 mg/kg every 24 hours. The last 10 days of daptomycin therapy were given at home via a percutaneous intravenous central catheter.
Comment. The epidemiology of MRSA infections is changing. More patients who have no traditional risk factors, specifically health care contact, are getting MRSA infections. A recent report from the US Centers for Disease Control and Prevention (CDC) indicates that the proportion of patients with invasive disease due to MRSA has doubled since 2001–2002.4 Part of the reason undoubtedly is that MRSA, particularly community-associated MRSA, often carries specific virulence factors that make it more invasive. The CDC estimated that in 2005 there were nearly 100,000 cases of invasive MRSA infection in the United States, and nearly a fifth of these infections resulted in death.
Resistance and virulence factors in community-associated MRSA
Most community-associated MRSA strains carry a mobile genetic element called type IV SCCmec (staphylococcal chromosomal cassettemec) that enhances its antimicrobial resistance. This genetic component was probably borrowed from coagulase-negative staphylococci, in which it is quite common but does not cause as much of a problem. It is now present in a wide range of S aureus strains. Most of the S aureus strains that carry type IV SCCmec are MRSA, but a few MSSA strains do carry it as well.
The potent toxin Panton-Valentine leukocidin is an extracellular product that is detected in fewer than 5% of hospital strains but is more common in community-associated strains. It kills leukocytes by forming pores in the cell membrane and causing skin necrosis in cutaneous infections. It is associated with skin abscesses and rapidly progressive necrotizing pneumonia in MSSA or MRSA.
Epidemiologic differences between community- and health-care-associated MRSA
Patients with community-associated MRSA infections tend to be younger than those who traditionally get health-care-associated MRSA infections: in a study from Naimi et al in 2003, the mean ages were 23 vs 68 years.5 A greater proportion of patients with community-associated MRSA strains are nonwhite.4,5
Most community-associated MRSA infections are of the skin and soft tissue (75% in the series from Naimi et al5), but this pathogen causes other infections as well. Bacteremia of unknown origin has been seen, as has necrotizing pneumonia. Most of the skin and soft-tissue infections are relatively superficial, such as folliculitis or furunculosis, but deeper tissue infections such as necrotizing fasciitis and pyomyositis have also been seen.6
The incidence of community-associated MRSA infections varies greatly by geographic region.7 The northeastern United States has so far been relatively spared, but in Atlanta, Houston, and Los Angeles up to 80% of cases of characteristic skin or soft-tissue infections seen in emergency or outpatient departments are due to community-associated MRSA. Physicians at the Texas Children’s Hospital in Houston assume that all skin or soft-tissue infections are due to community-associated MRSA unless proven otherwise.8
Differences in antibiotic susceptibility
Community-associated MRSA is more susceptible to various antibiotics than health-care-associated MRSA,5 but not by much. Strains are usually susceptible to vancomycin, tetracyclines, trimethoprim-sulfamethoxazole (Bactrim, Septra), and rifampin (Rifadin). Unlike hospital strains, a fair number of community-acquired strains are susceptible to clindamycin (Cleocin) in the laboratory, but with a caveat: some of these clindamycin-susceptible strains actually may harbor the tools for inducible resistance. In fact, they can become resistant to clindamycin even without being exposed to it.
The laboratory test for inducible clindamycin resistance is called the D test. After coating an agar plate with S aureus, the technician places erythromycin and clindamycin disks. If the erythromycin induces clindamycin resistance, the plate is clear of growth around the clindamycin disk except for the portion nearest the erythromycin disk, leaving a characteristic D-shaped area of lucency.
Risk factors for MRSA
Moran et al7 analyzed the risk factors for community-associated MRSA in patients with skin or soft-tissue infections seen in the emergency department. The infection was more likely to be due to community-associated MRSA if the patient was black, had used any antibiotic in the past month, had a history of MRSA infection, or had close contact with a person with a similar infection. Many patients interpreted the infections as spider bites because the lesions tended to have a dark center surrounded by a tender area. These infections were not associated with underlying illness. In some cases, community-associated MRSA skin infections have been associated with tattooing and even manicuring.
However, it is very difficult to distinguish between community-associated MRSA and MSSA skin and soft-tissue infections on the basis of clinical and epidemiologic characteristics. Miller et al9 studied a large group of patients in Los Angeles who were hospitalized with community-associated skin and soft-tissue S aureus infections. All the patients were followed up for 30 days after hospital discharge. Regardless of whether they had MRSA or MSSA, they had similar outcomes. Close contacts of the patients also tended to develop infection.
A key point from this and many other studies: patients were more likely to remain infected if they did not undergo incision and drainage. This key intervention is indicated for any patient who has a skin and soft-tissue infection with an undrained focus of infection.
COMMUNITY-ASSOCIATED MRSA IS INVADING THE HOSPITAL
In a new development, community-associated MRSA strains are now appearing in the hospital. This is not only because patients are bacteremic when they come in: patients in the hospital are getting nosocomial infections due to community-associated MRSA strains.
Seybold et al10 analyzed 116 cases of MRSA bloodstream infections in Atlanta, GA. In 9 (8%) of the cases the patient had not had any contact with the health care system within the past year, and these cases were classified as truly community-associated. Of the remaining 107 cases, 49 (42%) were nosocomial, and the USA 300 strain—the predominant community-associated MRSA strain—accounted for 10 (20%) of the nosocomial cases.
In the recent CDC study of invasive MRSA infections, Klevens et al4 reported that nearly a third of cases of bacteremia were due to community-associated MRSA, and these strains accounted for a greater proportion of cases of cellulitis and endocarditis than did health-care-associated strains.
In a study of hospital-associated MRSA, Maree et al11 found that the percentage of cases in which the bacteria carried the SCCmec type IV marker had increased from less than 20% in 1999 to more than 50% in 2004.
Comment. Suffice it to say that we are surrounded by MRSA. Community-associated MRSA is here to stay. It is even invading our hospitals, and we need to consider this very carefully when choosing antimicrobial therapy.
NAGGING QUESTIONS ABOUT VANCOMYCIN
Case 2: Vancomycin-intermediate S aureus (VISA) bacteremia and endocarditis
In December 2006 we saw a very ill 60-year-old woman who was hospitalized with MRSA bacteremia, pacemaker endocarditis, and superior vena cava thrombosis. Although she was treated with vancomycin and rifampin, her condition worsened, she had a stroke, and she developed renal failure. In a difficult operation, the pacemaker was removed, but the bacteremia persisted. In early February 2007 she underwent another difficult operation in which the superior vena cava clot was debrided, a right atrial clot was removed, and her mitral valve was replaced. Less than 2 weeks later, and despite ongoing vancomycin and rifampin therapy, the MRSA bacteremia recurred.
During the approximately 6 weeks that the patient had been receiving these antibiotics, the minimal inhibitory concentration (MIC) of rifampin against the S aureus isolate increased from less than 1 μg/mL (susceptible) to 2 μg/mL (resistant). The MIC of vancomycin went from 2 μg/mL (susceptible) to 4 μg/mL (intermediately susceptible). Vancomycin and rifampin were discontinued, and daptomycin and gentamicin (Garamycin) therapy were started. (Her daptomycin MIC was 0.5 μg/mL). The patient’s condition stabilized, and she was discharged to a long-term nursing facility. She had no relapse of MRSA bacteremia, but she died in early April of that year.
Is vancomycin becoming less effective? Degrees of vancomycin resistance
Vancomycin has been our stalwart for treating MRSA infections for more than 40 years but it is not working as well as it used to, at least in certain situations.
VRSA (vancomycin-resistant S aureus) is rare. These fully resistant strains probably acquired a resistance mechanism (the vanA operon) from vancomycin-resistant enterococci. Infections tend to occur in patients simultaneously infected with both S aureus and vancomycin-resistant enterococci, giving the bacteria an opportunity to exchange genetic material.
VISA (vancomycin-intermediate S aureus) infections tend to occur in patients like the one described above who have had long-term vancomycin therapy. VISA strains appear to overproduce a matrix that captures vancomycin and keeps it from entering the cell. On electron microscopy, these bacteria have a very thick cell wall.13
Vancomycin tolerance is a state in which the bacteria are “stunned” or kept in check but not killed by vancomycin. That is manifested in the laboratory by a ratio of minimum bactericidal concentration to MIC greater than 32.
hVISA (heteroresistant VISA) is new and worrisome. These organisms have an overall MIC in the susceptible range, but within that population are individual isolates with an MIC that is much higher—in the intermediate or perhaps even in the resistant range.14
Reported rates of hVISA vary from less than 2% to as high as 76%, because the methods for detecting it are still very poorly standardized. The usual automated laboratory tests do not detect hVISA.
hVISA is probably clinically relevant, as evidence is emerging both in vitro and in vivo that the higher the MIC for vancomycin, the worse the clinical outcome.15 hVISA has been associated with failures of therapy in several situations, usually in cases of severe invasive or deep infection, endocarditis, and bacteremia with vertebral osteomyelitis where vancomycin concentrations at the site of infection may be suboptimal.16–19 While most hVISA strains that have been described were resistant to methicillin, some were susceptible.
The E test is emerging as the standard test for hVISA. This test uses a plastic strip that contains gradually increasing concentrations of vancomycin along its length. Placed in the culture dish, the strip inhibits growth of the organism at its high-concentration end but not at its low-concentration end. If the sample contains hVISA, the cutoff is not well defined, with a few colonies growing at higher concentrations.
New definition of vancomycin susceptibility
Recognizing that the MICs for vancomycin have been rising in the last few years, the Clinical and Laboratory Standards Institute last year changed the break points between susceptibility and resistance. The new definitions are:
- Susceptible—an MIC of 2.0 μg/mL or less (formerly 4.0 μg/mL or less)
- Intermediate—4.0 to 8.0 μg/mL (formerly 8.0 to 16 μg/mL)
- Resistant—16 μg/mL or greater (formerly 32 μg/mL or greater).
One should pay attention to the MIC numbers on the laboratory reports, not just to the words “susceptible” or “not susceptible.” If the number is, say, 0.5 μg/mL or less, the organism should really be susceptible. If the number is 1 or 2, it is still in the susceptible range, but those are the organisms that may cause problems later on.
Further, even if the vancomycin MIC is in the susceptible range, higher MICs may affect outcomes. The average duration of MRSA bacteremia on therapy is 8 to 9 days, vs 3 to 4 days with MSSA bacteremia.20,21 But Sakoulas et al15 found that, in MRSA bacteremia, the success rate with vancomycin therapy was 56% if the MIC was 0.5 or lower, compared with 10% if the MIC was 1.0 to 2.0 μg/mL. Examined in another way, the success rate was 50% if the logarithm of killing was 6.27 colony-forming units per mL or greater, 23% if 4.71 to 6.26, and zero if less than 4.71.
Case 3: Prolonged MRSA bacteremia
In the summer of 2006, a 66-year-old woman with a history of gastric bypass and cirrhosis underwent a long stay in the surgical intensive care unit because of a recurrent enterocutaneous fistula and chronic renal insufficiency. On November 5th, she had a positive blood culture for MRSA, which was treated appropriately with vancomycin for 4 weeks. She was discharged to subacute care but came back 2 days later, again with MRSA bacteremia. At that time her Hickman catheter, which had been inserted for total parenteral nutrition because of the enterocutaneous fistula, was removed.
Transthoracic echocardiography revealed no vegetations, but her bacteremia persisted. Her mental status was poor this entire time: she was mute and could barely be awakened. We looked for clots and infected clots; duplex ultrasonographic examinations of all four extremities were negative. Finally, magnetic resonance imaging of her back—performed empirically because of the persistent bacteremia—revealed vertebral osteomyelitis at level T12-L1. We also noticed on serial evaluations that the vancomycin MIC for her organism increased from 0.5 to 2.0 μg/mL, so therapy was changed from vancomycin to daptomycin.
Her bacteremia cleared. Follow-up echocardiography was negative, but she had two subsequent relapses of MRSA bacteremia, one in April 2007 and one before she died in the summer of 2007.
Prolonged bacteremia: Is it vancomycin resistance, or something else?
The MRSA isolates that cause prolonged bacteremia seem to have certain characteristics.22 Higher MICs are probably associated with longer periods of bacteremia. But some genetic components within some strains of S aureus give them a survival advantage. They have less susceptibility to the body’s thrombin-induced platelet microbicidal protein. These isolates are not only associated with prolonged bacteremia: they are also associated with osteomyelitis, deep abscesses, endocarditis, recurrent infection, and increased death rate.22 Clinical laboratories do not test for these genetic components. One wonders whether our patient may have had an isolate with these mutations that gave it a survival advantage.
Do not use vancomycin for MSSA
Avoid using vancomycin for MSSA infections. It has been shown time and time again that MSSA infections do not respond as well to vancomycin as they do to beta-lactam antibiotics, specifically to the semisynthetic penicillins such as oxacillin and nafcillin, and even some of the first-generation cephalosporins. Chang et al23 found that patients with MSSA bacteremia had higher rates of persistent infections, relapse, and bacteriologic failure if they received vancomycin than if they received nafcillin.
Do vancomycin trough levels affect toxicity?
The vancomycin trough levels that we aimed for in the past (5 to 10 μg/mL) were probably too low. Today, we aim for trough levels of 15 to 20 μg/mL, and many physicians are aiming for 20 to 25 μg/mL. Part of the reason is that vancomycin MICs are higher than they used to be: in order to keep the vancomycin level above the MIC for a longer period of time, the vancomycin trough level needs to be higher. In theory, keeping the vancomycin levels above the MIC for longer periods should improve outcomes. Yet Fowler et al22 found that vancomycin trough levels among patients who had persistent MRSA bacteremia were actually higher than trough levels among those in whom the bacteremia resolved, although the difference was not statistically significant.
We measure the vancomycin trough level to make sure it is high enough (and give larger doses if it is not); among adults, peak levels need not be monitored on a routine basis because of the predictable pharmacokinetics of vancomycin.
Vancomycin toxicity can be either idiosyncratic or synergistic. Idiosyncratic toxicity occurs when a patient who has been on vancomycin for a long time develops a fixed rash, not associated with infusion. This is an immunologic phenomenon. It is a rare and very serious situation and may require steroid therapy.
Synergistic toxicity occurs when vancomycin is given with other nephrotoxic agents, notably gentamicin. Vancomycin plus gentamicin equals nephrotoxicity. Vancomycin alone is usually not nephrotoxic, but close monitoring of renal function parameters is warranted with the use of higher doses.24
IN UNEXPLAINED BACTEREMIA, LOOK FOR ENDOCARDITIS
In blood cultures from patients with bacteremia, S aureus is never a contaminant. Even if just one blood culture is positive for S aureus, believe that S aureus is the culprit.
Reports in the 1950s suggested that at least half of patients who had S aureus bacteremia had endocarditis,25 leading to recommendations that all patients with S aureus bacteremia without an obvious primary source of infection should be evaluated for endocarditis. Subsequent estimates were lower, in the range of 15% to 25%.26,27 However, throughout the world S aureus endocarditis continues to have a very high mortality rate: at least a third of patients die.28
Clinical criteria (community acquisition, no primary focus, and metastatic sequelae) were developed to try to predict the risk of endocarditis in bacteremic patients.26 However, these criteria did not work very well. The clinical definition of endocarditis has evolved. The criteria of von Reyn et al29 from 1981 did not use echocardiography as part of the definition, but the 1994 Duke criteria,30 which were refined31 in 2000, use both clinical and echocardiographic parameters.
Stratton et al32 performed transthoracic echocardiography in 14 patients with bacteremia and found 1 patient with cryptic tricuspid infective endocarditis. Bayer et al33 subsequently reported that of 72 patients with bacteremia, 6 (18%) of those who had no clinical findings suggestive of infectious endocarditis had findings on echocardiography that led to changes in their regimen. Adding echocardiography to three clinical risk factors increased the sensitivity of diagnosing endocarditis from 70% to 85% with a specificity of 100% and predictive value of 96%.
The Duke criteria call for transesophageal echocardiography, which is not feasible in some patients, eg, those with cirrhosis and esophageal varices.
S aureus endocarditis has changed over the years as our patient population has changed, and MRSA endocarditis tends to hit some of our most vulnerable patients. In a study by Miro et al34 in 2005, MRSA was the leading pathogen in patients who were diagnosed with S aureus endocarditis in 1990 or later. We will only see these numbers go up. Patients with diabetes tend to have more MRSA, and of diabetic patients with MRSA endocarditis, 30% to 40% die in the hospital.
Indications for surgery
Certain conditions are indications for surgery among patients with endocarditis, and no antibiotic will cure the endocarditis if the patient has one of these conditions, eg:
- Persistent bacteremia during antibiotic therapy
- Recurrent emboli
- Heart failure that cannot be controlled
- Perivalvular or myocardial abscesses
- Large vegetations
- Early prosthetic valve infection
- Certain arrhythmias.
How long should S aureus bacteremia be treated?
In cases of bacteremia in which endocarditis has been ruled out and removable foci of infection (eg, intravascular catheters) have been removed, some evidence indicates that treatment for 2 weeks would be as effective as the 4 to 6 weeks that we would use for endocarditis or other severe or invasive infections.35 The issue is controversial. If the patient has had frequent hospitalizations or a chronic medical condition I would hesitate to treat for less than 4 weeks, even if the infection appears to be associated with a removable focus.
Treatment of endocarditis
In the guidelines for treatment of endocarditis from the American Heart Association and Infectious Diseases Society of America,36 all the recommendations are relatively old and many of them are somewhat empiric—they are not based on evidence from randomized clinical trials. Rather, they are best opinions based on clinical experience and some observational studies over the years.
For MSSA. In cases of native-valve endocarditis, oxacillin (Bactocill), nafcillin (Unipen), or another semisynthetic beta-lactam antibiotic is recommended. For penicillin-allergic patients, we have other options, such as cefazolin (Ancef, Kefzol).
Combination therapy is frequently recommended for native valve endocarditis as well as for prosthetic valve endocarditis, with either rifampin or gentamicin along with a primary agent. There is some evidence that one can clear staphylococcal bacteremia a day or two more quickly by use of combination therapy with nafcillin plus an aminoglycoside than with nafcillin alone.37,38 For MSSA-associated endocarditis, vancomycin does not work as well as beta-lactam antibiotics.39,40
Korzeniowski and Sande37 and Chambers et al38 reported that the mean duration of bacteremia was 3.4 days for patients treated with nafcillin alone and 2.9 days for those treated with nafcillin plus an aminoglycoside. These studies led to consideration of a short course of gentamicin to clear the bacteremia quickly.
With MRSA, bacteremia often requires a week or more to clear. Levine et al21 reported a study in 42 patients, mostly injection-drug users, with right-sided native-valve endocarditis. The median duration of bacteremia was 7 days in patients who received vancomycin alone vs 9 days in those who received vancomycin plus rifampin; however, some patients were bacteremic for up to 27 days. Fever persisted for a median of 7 days, probably partly due to septic pulmonary emboli. Three patients died, and three required valve replacement.
NEW ANTIBIOTICS
Several new antibiotics are active against gram-positive cocci.41–44 However, the majority of them have not been prospectively studied for treating bacteremia or endocarditis.
Quinupristin/dalfopristin (Synercid) has not been formally studied for treatment of MRSA bacteremia or endocarditis. There are a few case reports of its use in these conditions.45 Quinupristin/dalfopristin is bacteriostatic, and its use may be associated with phlebitis, myalgias, and arthralgias.46
Linezolid (Zyvox) is approved for treatment of complicated skin and soft-tissue infections and for hospital-acquired pneumonia. There have been no specific studies of linezolid in the treatment of S aureus bacteremia or endocarditis. However, Shorr et al47 retrospectively looked at the bacteremic patients in five previous studies of linezolid vs vancomycin and found 144 cases of S aureus bacteremia, half of which were due to MRSA. Of 53 assessable patients with MRSA bacteremia, the primary infection was cured in 14 (56%) of the linezolid patients and 13 (46%) of the vancomycin patients.
The oral form is 100% bioavailable. One should avoid concomitant use of serotonin-reuptake inhibitors because of the risk of serotonin syndrome. Adverse effects include altered taste sensation and peripheral neuropathy. There are other potential toxicities, including hematologic changes (thrombocytopenia, leukopenia) and metabolic effects (lactic acidosis), so clinical and laboratory monitoring is important.48 The role of linezolid in the treatment of patients with S aureus bacteremia or endocarditis remains to be defined.
Daptomycin is indicated for complicated skin and soft-tissue infections, bacteremia, and right-sided endocarditis due to S aureus. Fowler et al20 found that daptomycin was not inferior to beta-lactam antibiotics for treatment of MSSA bacteremia and right-sided endocarditis, and for MRSA infections it outperformed vancomycin, but the difference was not statistically significant.
The dosing interval should be increased from once every 24 hours to every 48 hours if the creatinine clearance is 30 mL/minute or less. Adverse effects include myalgia, rhabdomyolysis (rare), and elevations in creatine phosphokinase. Reports of rising MICs during daptomycin therapy, in some cases associated with persistent infection,49 suggest that careful attention be paid to dosing and clinical monitoring.
Tigecycline (Tygacil) is indicated for complicated skin and soft-tissue infections and complicated intra-abdominal infections due to susceptible organisms. It is active against both MSSA and MRSA, but clinical experience with its use in invasive infections is somewhat limited.50 The dose of tigecycline should be reduced in advanced cirrhosis. Adverse effects include nausea and vomiting.
Telavancin, dalbavancin, and oritavancin, investigational parenteral antibiotics that are derivatives of vancomycin, are in clinical trials. The pharmacokinetic activity of these agents is of interest: telavancin is being studied with a once-daily dosing interval and dalbavancin’s half-life allows once-weekly dosing. In a limited trial, dalbavancin was found to be safe and effective in the treatment of catheter-related bloodstream infections.51 None of the antibiotics in this group has been studied for treatment of S aureus endocarditis. Telavancin therapy has been associated with rash, hypokalemia, QT prolongation, and creatinine elevations. Gastrointestinal symptoms have been reported with the use of dalbavancin.
Ceftobiprole, another investigational agent, is the only cephalosporin antibiotic that is active against MRSA. It is given every 12 hours. Adverse effects include nausea and taste disturbance.
Iclaprim is a novel diaminopyrimidine and a dihydrofolate reductase inhibitor. In vitro, it is active against gram-positive bacteria, including MRSA, VISA, and VRSA; clinical investigations at this point are limited to the treatment of skin and soft-tissue infections.
- Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 2004; 39:309–371. Erratum in: Clin Infect Dis 2004; 39:1093.
- US Centers for Disease Control and Prevention. National Nosocomial Infections Surveillance (NNIS) System. Campaign to prevent antimicrobial resistance. www.cdc.gov/drugresistance/healthcare/ha/HASlideSet.ppt.
- Blot SI, Vandewoude KH, Hoste EA, Colardyn FA. Outcome and attributable mortality in critically ill patients with bacteremia involving methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Arch Intern Med 2002; 162:2229–2235.
- Klevens RM, Morrison MA, Nadle J, et al; Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA 2007; 298:1763–1771.
- Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003; 290:2976–2984.
- Miller LG, Perdreau-Remington F, Rieg G, et al. Necrotizing fasciitis caused by community-associated methicillin-resistant Staphylococcus aureus in Los Angeles. N Engl J Med 2005; 352:1445–1453.
- Moran GJ, Krishnadasan A, Gorwitz RJ, et al EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006; 355:666–674.
- Mishaan AM, Mason EO, Martinez-Aquilar G, et al. Emergence of a predominant clone of community-acquired Staphylococcus aureus among children in Houston, Texas. Pediatr Infect Dis J 2005; 24:201–206.
- Miller LG, Perdreau-Remington F, Bayer AS, et al. Clinical and epidemiologic characteristics cannot distinguish community-associated methicillin-resistant Staphylococcus aureus infection from methicillin-susceptible S. aureus infection: a prospective investigation. Clin Infect Dis 2007; 44:471–482.
- Seybold U, Kourbatova EV, Johnson JG, et al. Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care-associated blood stream infections. Clin Infect Dis 2006; 42:647–656.
- Maree CL, Daum RS, Boyle-Vavra S, Matayoshi K, Miller LG. Community-associated methicillin-resistant Staphylococcus aureus isolates causing healthcare-associated infections. Emerg Infect Dis 2007; 13:236–242.
- Liu C, Chambers HF. Staphylococcus aureus with heterogeneous resistance to vancomycin: epidemiology, clinical significance, and critical assessment of diagnostic methods. Antimicrob Agents Chemother 2003; 47:3040–3045.
- Sieradzki K, Roberts RB, Haber SW, Tomasz A. The development of vancomycin resistance in a patient with methicillin-resistant Staphylococcus aureus infection. N Engl J Med 1999; 340:517–523.
- Schwaber MJ, Wright SB, Carmeli Y, et al. Clinical implications of varying degrees of vancomycin susceptibility in methicillin-resistant Staphylococcus aureus bacteremia. Emerg Infect Dis 2003; 9:657–664. Erratum in: Emerg Infect Dis 2004; 10:160.
- Sakoulas G, Moise-Broder PA, Schentag J, Forrest A, Moellering RC, Eliopoulos GM. Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin-resistant Staphylococcus aureus bacteremia. J Clin Microbiol 2004; 42:2398–2402.
- Naimi TS, Anderson D, O’Boyle C, et al. Vancomycin-intermediate Staphylococcus aureus with phenotypic susceptibility to methicillin in a patient with recurrent bacteremia. Clin Infect Dis 2003; 36:1609–1612.
- Moore MR, Perdreau-Remington F, Chambers HF. Vancomycin treatment failure associated with heterogeneous vancomycin-intermediate Staphylococcus aureus in a patient with endocarditis and in the rabbit model of endocarditis. Antimicrob Agents Chemother 2003; 47:1262–1266.
- Charles PG, Ward PB, Johnson PD, Howden BP, Grayson ML. Clinical features associated with bacteremia due to heterogenous vancomycin-intermediate Staphylococcus aureus. Clin Infect Dis 2004; 38:448–451.
- Howden BP, Ward PB, Charles PG, et al. Treatment outcomes for serious infections caused by methicillin-resistant Staphylococcus aureus with reduced vancomycin susceptibility. Clin Infect Dis 2004; 38:521–528.
- Fowler VG, Boucher HW, Corey GR, et al. S. aureus Endocarditis and Bacteremia Study Group. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med 2006; 355:653–665.
- Levine DP, Fromm BS, Reddy BR. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann Intern Med 1991; 115:674–680.
- Fowler VG, Sakoulas G, McIntyre LM, et al. Persistent bacteremia due to methicillin-resistant Staphylococcus aureus infection is associated with agr dysfunction and low-level in vitro resistance to thrombin-induced platelet microbicidal protein. J Infect Dis 2004; 190:1140–1149.
- Chang FY, Peacock JE, Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore) 2003; 82:333–339.
- Hidayat LK, Hsu DI, Quist R, Shriner KA, Wong-Beringer A. High-dose vancomycin therapy for methicillin-resistant Staphylococcus aureus infections: efficacy and toxicity. Arch Intern Med 2006; 166:2138–2144.
- Wilson R, Hamburger M. Fifteen years’ experience with staphylococcus septicemia in a large city hospital; analysis of fifty-five cases in the Cincinnati General Hospital 1940 to 1954. Am J Med 1957; 22:437–457.
- Nolan CM, Beaty HN. Staphylococcus aureus bacteremia. Current clinical patterns. Am J Med 1976; 60:495–500.
- Shah M, Watanakunakorn C. Changing patterns of Staphylococcus aureus bacteremia. Am J Med Sci 1979; 278:115–121.
- Fowler VG, Miro JM, Hoen B, et al ICE Investigators. Staphylococcus aureus endocarditis: a consequence of medical progress. JAMA 2005; 293:3012–3021. Erratum in: JAMA 2005; 294:900.
- Von Reyn CF, Levy BS, Arbeit RD, Friedland G, Crumpacker CS. Infective endocarditis: an analysis based on strict case definition. Ann Intern Med 1981; 94:505–518.
- Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med 1994; 96:200–209.
- Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 2000; 30:633–638.
- Stratton JR, Werner JA, Pearlman AS, Janko CL, Kliman S, Jackson MC. Bacteremia and the heart. Serial echocardiographic findings in 80 patients with documented or suspected bacteremia. Am J Med 1982; 73:851–858.
- Bayer AS, Lam K, Ginzton L, Normal DC, Chiu CY, Ward JI. Staphylococcus aureus bacteremia. Clinical, serologic, and echocardiographic findings in patients with and without endocarditis. Arch Intern Med 1987; 147:457–462.
- Miro JM, Anguera I, Cabell CH, et al International Collaboration on Endocarditis Merged Database Study Group. Staphylococcus aureus native valve infective endocarditis: report of 566 episodes from the International Collaboration on Endocarditis Merged Database. Clin Infect Dis 2005; 41:507–514. Erratum in: Clin Infect Dis 2005; 41:1075–1077.
- Jernigan JA, Farr BM. Short-course therapy of catheter-related Staphylococcus aureus bacteremia: a meta-analysis. Ann Intern Med 1993; 119:304–311.
- Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America. Circulation 2005; 111:e394–e434. Erratum in: Circulation 2005; 112:2373. Circulation 2007; 115:e408.
- Korzeniowski O, Sande MA. Combination antimicrobial therapy for Staphylococcus aureus endocarditis in patients addicted to parenteral drugs and in nonaddicts: a prospective study. Ann Intern Med 1982; 97:496–503.
- Chambers HF, Korzeniowski OM, Sande MA. Staphylococcus aureus endocarditis: clinical manifestations in addicts and nonaddicts. Medicine (Baltimore) 1983; 62:170–177.
- Gentry CA, Rodvold KA, Novak RM, Hershow RC, Naderer OJ. Retrospective evaluation of therapies for Staphylococcus aureus endocarditis. Pharmacotherapy 1997; 17:990–997.
- Small PM, Chambers HF. Vancomycin for Staphylococcus aureus endocarditis in intravenous drug users. Antimicrob Agents Chemother 1990; 34:1227–1231.
- Eliopoulos GM. Quinupristin-dalfopristin and linezolid: evidence and opinion. Clin Infect Dis 2003; 36:473–481.
- Rybak MJ. Therapeutic options for Gram-positive infections. J Hosp Infect 2001; 49 suppl A:S25–S32.
- Micek ST. Alternatives to vancomycin for the treatment of methicillin-resistant Staphylococcus aureus infections. Clin Infect Dis 2007; 45 suppl 3:S184–S190.
- Appelbaum PC, Jacobs MR. Recently approved and investigational antibiotics for treatment of severe infections caused by Gram-positive bacteria. Curr Opin Microbiol 2005; 8:510–517.
- Drew RH, Perfect JR, Srinath L, Kirkimilis E, Dowzicky M, Talbot GH for the Synercid Emergency-Use Study Group. Treatment of methicillin-resistant Staphylococcus aureus infections with quinupristin-dalfopristin in patients intolerant of or failing prior therapy. J Antimicrob Chemother 2000; 46:775–784.
- Lamb HM, Figgitt DP, Faulds D. Quinupristin/dalfopristin: a review of its use in the management of serious gram-positive infections. Drugs 1999; 58:1061–1097.
- Shorr AF, Kunkel MJ, Kollef M. Linezolid versus vancomycin for Staphylococcus aureus bacteraemia: pooled analysis of randomized studies. J Antimicrob Chemother 2005; 56:923–929.
- Bishop E, Melvani S, Howden BP, Charles PG, Grayson ML. Good clinical outcomes but high rates of adverse reactions during linezolid therapy for serious infections: a proposed protocol for monitoring therapy in complex patients. Antimicrob Agents Chemother 2006; 50:1599–1602.
- Boucher HW, Sakoulas G. Perspectives on daptomycin resistance, with emphasis on resistance in Staphylococcus aureus. Clin Infect Dis 2007; 45:601–608.
- Munoz-Price LS, Lolans K, Quinn JP. Four cases of invasive methicillin-resistant Staphylococcus aureus (MRSA) infections treated with tigecycline. Scand J Infect Dis 2006; 38:1081–1084.
- Raad I, Darouiche R, Vazquez J, et al. Efficacy and safety of weekly dalbavancin therapy for catheter-related bloodstream infection caused by gram-positive pathogens. Clin Infect Dis 2005; 40:374–80.
- Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 2004; 39:309–371. Erratum in: Clin Infect Dis 2004; 39:1093.
- US Centers for Disease Control and Prevention. National Nosocomial Infections Surveillance (NNIS) System. Campaign to prevent antimicrobial resistance. www.cdc.gov/drugresistance/healthcare/ha/HASlideSet.ppt.
- Blot SI, Vandewoude KH, Hoste EA, Colardyn FA. Outcome and attributable mortality in critically ill patients with bacteremia involving methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Arch Intern Med 2002; 162:2229–2235.
- Klevens RM, Morrison MA, Nadle J, et al; Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA 2007; 298:1763–1771.
- Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003; 290:2976–2984.
- Miller LG, Perdreau-Remington F, Rieg G, et al. Necrotizing fasciitis caused by community-associated methicillin-resistant Staphylococcus aureus in Los Angeles. N Engl J Med 2005; 352:1445–1453.
- Moran GJ, Krishnadasan A, Gorwitz RJ, et al EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006; 355:666–674.
- Mishaan AM, Mason EO, Martinez-Aquilar G, et al. Emergence of a predominant clone of community-acquired Staphylococcus aureus among children in Houston, Texas. Pediatr Infect Dis J 2005; 24:201–206.
- Miller LG, Perdreau-Remington F, Bayer AS, et al. Clinical and epidemiologic characteristics cannot distinguish community-associated methicillin-resistant Staphylococcus aureus infection from methicillin-susceptible S. aureus infection: a prospective investigation. Clin Infect Dis 2007; 44:471–482.
- Seybold U, Kourbatova EV, Johnson JG, et al. Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care-associated blood stream infections. Clin Infect Dis 2006; 42:647–656.
- Maree CL, Daum RS, Boyle-Vavra S, Matayoshi K, Miller LG. Community-associated methicillin-resistant Staphylococcus aureus isolates causing healthcare-associated infections. Emerg Infect Dis 2007; 13:236–242.
- Liu C, Chambers HF. Staphylococcus aureus with heterogeneous resistance to vancomycin: epidemiology, clinical significance, and critical assessment of diagnostic methods. Antimicrob Agents Chemother 2003; 47:3040–3045.
- Sieradzki K, Roberts RB, Haber SW, Tomasz A. The development of vancomycin resistance in a patient with methicillin-resistant Staphylococcus aureus infection. N Engl J Med 1999; 340:517–523.
- Schwaber MJ, Wright SB, Carmeli Y, et al. Clinical implications of varying degrees of vancomycin susceptibility in methicillin-resistant Staphylococcus aureus bacteremia. Emerg Infect Dis 2003; 9:657–664. Erratum in: Emerg Infect Dis 2004; 10:160.
- Sakoulas G, Moise-Broder PA, Schentag J, Forrest A, Moellering RC, Eliopoulos GM. Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin-resistant Staphylococcus aureus bacteremia. J Clin Microbiol 2004; 42:2398–2402.
- Naimi TS, Anderson D, O’Boyle C, et al. Vancomycin-intermediate Staphylococcus aureus with phenotypic susceptibility to methicillin in a patient with recurrent bacteremia. Clin Infect Dis 2003; 36:1609–1612.
- Moore MR, Perdreau-Remington F, Chambers HF. Vancomycin treatment failure associated with heterogeneous vancomycin-intermediate Staphylococcus aureus in a patient with endocarditis and in the rabbit model of endocarditis. Antimicrob Agents Chemother 2003; 47:1262–1266.
- Charles PG, Ward PB, Johnson PD, Howden BP, Grayson ML. Clinical features associated with bacteremia due to heterogenous vancomycin-intermediate Staphylococcus aureus. Clin Infect Dis 2004; 38:448–451.
- Howden BP, Ward PB, Charles PG, et al. Treatment outcomes for serious infections caused by methicillin-resistant Staphylococcus aureus with reduced vancomycin susceptibility. Clin Infect Dis 2004; 38:521–528.
- Fowler VG, Boucher HW, Corey GR, et al. S. aureus Endocarditis and Bacteremia Study Group. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med 2006; 355:653–665.
- Levine DP, Fromm BS, Reddy BR. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann Intern Med 1991; 115:674–680.
- Fowler VG, Sakoulas G, McIntyre LM, et al. Persistent bacteremia due to methicillin-resistant Staphylococcus aureus infection is associated with agr dysfunction and low-level in vitro resistance to thrombin-induced platelet microbicidal protein. J Infect Dis 2004; 190:1140–1149.
- Chang FY, Peacock JE, Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore) 2003; 82:333–339.
- Hidayat LK, Hsu DI, Quist R, Shriner KA, Wong-Beringer A. High-dose vancomycin therapy for methicillin-resistant Staphylococcus aureus infections: efficacy and toxicity. Arch Intern Med 2006; 166:2138–2144.
- Wilson R, Hamburger M. Fifteen years’ experience with staphylococcus septicemia in a large city hospital; analysis of fifty-five cases in the Cincinnati General Hospital 1940 to 1954. Am J Med 1957; 22:437–457.
- Nolan CM, Beaty HN. Staphylococcus aureus bacteremia. Current clinical patterns. Am J Med 1976; 60:495–500.
- Shah M, Watanakunakorn C. Changing patterns of Staphylococcus aureus bacteremia. Am J Med Sci 1979; 278:115–121.
- Fowler VG, Miro JM, Hoen B, et al ICE Investigators. Staphylococcus aureus endocarditis: a consequence of medical progress. JAMA 2005; 293:3012–3021. Erratum in: JAMA 2005; 294:900.
- Von Reyn CF, Levy BS, Arbeit RD, Friedland G, Crumpacker CS. Infective endocarditis: an analysis based on strict case definition. Ann Intern Med 1981; 94:505–518.
- Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med 1994; 96:200–209.
- Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 2000; 30:633–638.
- Stratton JR, Werner JA, Pearlman AS, Janko CL, Kliman S, Jackson MC. Bacteremia and the heart. Serial echocardiographic findings in 80 patients with documented or suspected bacteremia. Am J Med 1982; 73:851–858.
- Bayer AS, Lam K, Ginzton L, Normal DC, Chiu CY, Ward JI. Staphylococcus aureus bacteremia. Clinical, serologic, and echocardiographic findings in patients with and without endocarditis. Arch Intern Med 1987; 147:457–462.
- Miro JM, Anguera I, Cabell CH, et al International Collaboration on Endocarditis Merged Database Study Group. Staphylococcus aureus native valve infective endocarditis: report of 566 episodes from the International Collaboration on Endocarditis Merged Database. Clin Infect Dis 2005; 41:507–514. Erratum in: Clin Infect Dis 2005; 41:1075–1077.
- Jernigan JA, Farr BM. Short-course therapy of catheter-related Staphylococcus aureus bacteremia: a meta-analysis. Ann Intern Med 1993; 119:304–311.
- Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America. Circulation 2005; 111:e394–e434. Erratum in: Circulation 2005; 112:2373. Circulation 2007; 115:e408.
- Korzeniowski O, Sande MA. Combination antimicrobial therapy for Staphylococcus aureus endocarditis in patients addicted to parenteral drugs and in nonaddicts: a prospective study. Ann Intern Med 1982; 97:496–503.
- Chambers HF, Korzeniowski OM, Sande MA. Staphylococcus aureus endocarditis: clinical manifestations in addicts and nonaddicts. Medicine (Baltimore) 1983; 62:170–177.
- Gentry CA, Rodvold KA, Novak RM, Hershow RC, Naderer OJ. Retrospective evaluation of therapies for Staphylococcus aureus endocarditis. Pharmacotherapy 1997; 17:990–997.
- Small PM, Chambers HF. Vancomycin for Staphylococcus aureus endocarditis in intravenous drug users. Antimicrob Agents Chemother 1990; 34:1227–1231.
- Eliopoulos GM. Quinupristin-dalfopristin and linezolid: evidence and opinion. Clin Infect Dis 2003; 36:473–481.
- Rybak MJ. Therapeutic options for Gram-positive infections. J Hosp Infect 2001; 49 suppl A:S25–S32.
- Micek ST. Alternatives to vancomycin for the treatment of methicillin-resistant Staphylococcus aureus infections. Clin Infect Dis 2007; 45 suppl 3:S184–S190.
- Appelbaum PC, Jacobs MR. Recently approved and investigational antibiotics for treatment of severe infections caused by Gram-positive bacteria. Curr Opin Microbiol 2005; 8:510–517.
- Drew RH, Perfect JR, Srinath L, Kirkimilis E, Dowzicky M, Talbot GH for the Synercid Emergency-Use Study Group. Treatment of methicillin-resistant Staphylococcus aureus infections with quinupristin-dalfopristin in patients intolerant of or failing prior therapy. J Antimicrob Chemother 2000; 46:775–784.
- Lamb HM, Figgitt DP, Faulds D. Quinupristin/dalfopristin: a review of its use in the management of serious gram-positive infections. Drugs 1999; 58:1061–1097.
- Shorr AF, Kunkel MJ, Kollef M. Linezolid versus vancomycin for Staphylococcus aureus bacteraemia: pooled analysis of randomized studies. J Antimicrob Chemother 2005; 56:923–929.
- Bishop E, Melvani S, Howden BP, Charles PG, Grayson ML. Good clinical outcomes but high rates of adverse reactions during linezolid therapy for serious infections: a proposed protocol for monitoring therapy in complex patients. Antimicrob Agents Chemother 2006; 50:1599–1602.
- Boucher HW, Sakoulas G. Perspectives on daptomycin resistance, with emphasis on resistance in Staphylococcus aureus. Clin Infect Dis 2007; 45:601–608.
- Munoz-Price LS, Lolans K, Quinn JP. Four cases of invasive methicillin-resistant Staphylococcus aureus (MRSA) infections treated with tigecycline. Scand J Infect Dis 2006; 38:1081–1084.
- Raad I, Darouiche R, Vazquez J, et al. Efficacy and safety of weekly dalbavancin therapy for catheter-related bloodstream infection caused by gram-positive pathogens. Clin Infect Dis 2005; 40:374–80.
KEY POINTS
- Community-associated MRSA infections tend to affect patients younger than those who traditionally get hospital-associated MRSA infections. Most of these infections are of the skin and soft tissues, but this pathogen can also affect deeper tissues, and bacteremia and necrotizing pneumonia have been reported.
- For patients with skin and soft-tissue infections due to MRSA, incision and drainage rather than antibiotic therapy is often the key intervention.
- Vancomycin has been our stalwart for treating MRSA infections for more than 40 years, but it is not working as well as it used to, at least in certain situations. Vancomycin should not be used to treat infections due to methicillin-susceptible S aureus.
- Needed are better understanding of the factors that influence persistent S aureus bacteremia, well-controlled, prospective studies, and continued antibiotic development.
Fluid restriction is superior in acute lung injury and ARDS
Although most clinicians tend to manage acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) by giving more rather than less fluid,1,2 patients may actually fare better under a strategy of limited fluid intake and increased fluid excretion.
According to the results of the Fluids and Catheters Treatment Trial (FACTT),3 patients managed with fluid restriction (the “dry” or conservative strategy) spent significantly less time in the intensive care unit (ICU) and on mechanical ventilation than did patients who received a high fluid intake (the “wet” or liberal strategy). These benefits of the conservative strategy were attained without any increase in the mortality rate at 60 days or in nonpulmonary organ failure at 28 days.
In this article, I discuss the basis for the FACTT researchers’ conclusion that a conservative fluid strategy is preferable to a liberal fluid strategy in ALI/ARDS.
STUDY RATIONALE
One of the more enduring questions in critical care medicine is which fluid-management strategy is best for patients with ALI/ARDS.
The conservative strategy results in a lower vascular filling pressure, which in turn reduces pulmonary edema and improves gas exchange. The drawback to this strategy is that it may have a negative effect on cardiac output and nonpulmonary organ function.
The liberal strategy results in a higher vascular filling pressure, which may be beneficial in terms of cardiac output and nonpulmonary organ perfusion. However, this strategy does not reduce lung edema.
The evidence accumulated before FACTT did not favor one strategy over the other. However, most deaths among patients with ALI/ARDS are attributable to the failure of organs other than the lungs.4,5 As a result, aggressive fluid restriction has not been a common approach in hospitals throughout the United States.1,2
In an effort to resolve the controversy surrounding the management of ALI/ARDS and to broaden the scope of what we know about fluid balance, we undertook this multicenter, randomized, prospective clinical comparison of the two strategies. This study was conducted under the auspices of the National Heart, Lung, and Blood Institute’s Acute Respiratory Distress Syndrome Clinical Trials Network (ARDSnet).
STUDY DESIGN
Between June 8, 2000, and October 3, 2005, we screened more than 11,000 patients with ALI/ARDS at 20 centers in North America.
Eligibility
Eligible patients had experienced ALI/ARDS within the previous 48 hours, had been intubated for positive-pressure ventilation, had a ratio of partial pressure of arterial oxygen (PaO2) to the fraction of inspired oxygen (FIO2) of less than 300, and exhibited bilateral infiltrates on chest radiography that were consistent with the presence of pulmonary edema without evidence of left atrial hypertension.6
Major exclusion criteria included the placement of a pulmonary artery catheter prior to randomization and the presence of certain illnesses that might have compromised the study results.
Patient population
The target enrollment of 1,000 patients was reached. These patients were randomized into one of four roughly equal groups based on the type of fluid-management strategy—conservative or liberal—and the type of catheter that was placed—pulmonary artery or central venous. (The ARDSnet researchers published the results of the catheter comparison in a separate article.7 Those results are not discussed here except to note that there were no statistically significant differences in outcomes between the two catheter groups.)
There were no statistically significant differences between the two groups with respect to baseline demographic characteristics. The conservative-strategy group consisted of 503 patients, of whom 52% were male and 65% were white; the mean age was 50.1 years. The liberal-strategy group consisted of 497 patients, of whom 55% were male and 63% were white; mean age was 49.5 years.
Management
Ventilation according to a low tidal volume strategy (6 mg/kg) was initiated within 1 hour after randomization. The pulmonary artery catheter or central venous catheter was inserted within 4 hours of randomization, and fluid management was started within 2 hours after catheter insertion. Fluid management was continued for 7 days or until 12 hours after extubation in patients who became able to breathe without assistance, whichever occurred first.
Target filling pressures. In the conservative-strategy group, the target filling pressures were low—a pulmonary artery occlusion pressure less than 8 mm Hg for those randomized to receive a pulmonary artery catheter, and a central venous pressure less than 4 mm Hg for those randomized to receive a central venous catheter. Barring adverse effects, patients were to undergo diuresis with furosemide (Lasix) until their goal was achieved, and then they would be maintained on that dosage through day 7. If we experienced difficulty in safely reaching these goals—say, if a patient developed hypoxemia, oliguria, or hypotension—we backed off the diuresis until the patient stabilized, and then we tried again. An inability to reach these filling pressure targets was not considered to be a treatment failure; our actual aim was to get as close to the target as possible as long as the patient tolerated the treatment.
In the liberal-strategy group, the target pressures were in the high-to-normal range—14 to 18 mm Hg for those with a pulmonary artery catheter and 10 to 14 mm Hg for those with a central venous catheter.
Patients with a pulmonary artery catheter who were hemodynamically stable after 3 days could be switched to a central venous catheter at the discretion of the clinician.
Monitoring. Patients were monitored once every 4 hours—more often if the clinician felt it necessary—for four variables:
- Pulmonary artery occlusion pressure or central venous pressure, depending on the type of catheter
- Shock, indicated by a mean arterial pressure of less than 60 mm Hg or the need for a vasopressor
- Oliguria, indicated by a urine output of less than 0.5 mL/kg/hour
- Ineffective circulation, represented by a cardiac index of less than 2.5 L/minute/cm2 in the pulmonary artery catheter group and by the presence of cold, mottled skin and a capillary-refilling time of more than 2 seconds in the central venous catheter group.
Depending on what the clinician found during monitoring, patients could receive a fluid bolus (if the filling pressure was too low), furosemide (if the filling pressure was too high), dobutamine (in certain rare circumstances), or nothing.
We monitored compliance with the protocol instructions twice each day—at a set time each morning and later in the day at a randomly selected time. An important aspect of this study is that we had no protocol instructions for managing shock. Individual clinicians were free to treat shock however they deemed best. In essence, then, our study was a comparison of liberal and conservative strategies during the nonshock phase of ALI/ARDS.
End points
The primary end point was the mortality rate at 60 days. Patients who were discharged earlier were assumed to be alive at 60 days.
The secondary end points were the number of ICU-free and ventilator-free days and the number of organ-failure-free days at day 28. Other end points included various indicators of lung physiology.
Statistical analysis
This intention-to-treat analysis was powered so that we had a 90% chance of detecting a 10% difference in mortality rate at day 60 (statistical significance: P < .05).
Protocol safeguards
Prior to treatment, we knew that some patients in the liberal-strategy group would not reach their filling-pressure targets despite the infusion of large amounts of fluid. To avoid “overdosing” these patients, we limited all patients to a maximum of three fluid boluses per 24 hours. Also, we withheld fluid boluses if a patient’s FIO2 level reached or exceeded 0.7 or if the cardiac index rose to 4.5 L/minute/cm2 or higher.
Diuretics were withheld when a patient had received a vasopressor or had emerged from shock within the preceding 12 hours. Also, diuretics were not given to any patient who had received a fluid bolus within the preceding 12 hours or when renal failure was present (these patients were given renal support therapy).
Finally, physicians and coordinators were instructed to assess each protocol instruction for safety and clinical validity before implementing the particular instruction. If, in their medical judgment, a particular protocol instruction should not be implemented, they were authorized to override the instruction and record the reason for doing so in the case report form.
RESULTS
Protocol compliance
Clinicians adhered to the protocol instructions during approximately 90% of the time.
Diuretic administration. In response to high filling pressures, patients in the conservative-strategy and liberal-strategy groups received furosemide during 41% and 10% of assessment periods, respectively (P < .0001). By day 7, the average patient in the conservative-strategy group had received a cumulative dose of approximately 1,000 mg of furosemide, while the average patient in the liberal-strategy group had received 500 mg.
Fluid administration. Low filling pressure prompted the administration of a fluid bolus to the liberal-strategy group during 15% of the assessment periods, compared with 6% in the conservative-strategy group (P < .0001).
The conservative-strategy patients who were in shock at study entry had a net gain of approximately 3 L of fluid by day 7, while the liberal-strategy group had a gain of approximately 10 L. Among the patients who were shock-free at baseline, the conservative-strategy group had a net loss of almost 2 L at day 7 while the liberal-strategy group had a net gain of about 5 L.
The pulmonary artery occlusion pressure fell from 15.6 mm Hg to just below 13 mm Hg in the conservative-strategy group by day 7, although there was a wide variation among individual patients. The pressure in the liberal-strategy group (15.7 mm Hg at baseline) was unchanged at day 7 (Figure 2).
Primary end point
Secondary end points
Through day 7, the average patient in the conservative-strategy group experienced significantly more ICU-free days (0.9 vs 0.6; P <.001) and more days free of central nervous system (CNS) failure (3.4 vs 2.9; P = .02). No significant differences were observed in the number of days free from coagulation abnormalities and renal or hepatic failure at day 28.
Through day 28, the average patient in the conservative-strategy group experienced significantly more ventilator-free days (14.6 vs 12.1; P < .001). The other 7-day results held up after 28 days, as the average conservative-strategy patient continued to experience more ICU-free days (13.4 vs 11.2; P < .001) and more days free of CNS failure (18.8 vs 17.2; P = .03). Again, no significant differences were observed in the number of days free of coagulation abnormalities and cardiovascular, renal, or hepatic failure.
It is not clear if the conservative strategy’s advantage in terms of more CNS-failure-free days was actually the result of the strategy itself or due to the fact that these patients were weaned off ventilation earlier and therefore received less sedation.
Other outcomes
Shock. One concern we had with the conservative strategy was that it might induce shock more frequently, but this did not occur. The percentage of patients who developed shock at least once during the 7-day treatment protocol was quite similar in the two groups. Also, it is interesting that patients who presented with no baseline shock had only about a 30% chance of developing shock during therapy. There was no significant difference in vasopressor use between the two groups.
Lung function. The conservative-strategy group had a significantly better Murray lung injury score at day 7: 2.03 vs 2.27 (P < .001).
Tidal-volume scores (7.4 mL/kg in both groups at baseline) dropped at an equal rate and were virtually identical at day 7 (6.36 mL/kg in the conservative-strategy group and 6.34 in the liberal-strategy group), as expected.The plateau pressure, positive end-expiraory pressure, PaO2–FIO2 ratio, and oxygenation index were slightly but not significantly better in the conservative-strategy group at day 7.
Overall, lung function was considerably better in the conservative-strategy group.
Cardiovascular function. The mean arterial pressure was significantly lower in the conservative-strategy group at day 7 (81.00 vs 84.36 mm Hg; P = .03). It is interesting that both levels were higher than the baseline levels (77.1 and 77.2, respectively; not significant).
The stroke volume index and the cardiac index were slightly lower in the conservative-strategy group at day 7, but not significantly so. No differences were seen in heart rate and venous oxygen saturation levels.
Renal and metabolic function. At day 7, the conservative-strategy group had a significantly higher blood urea nitrogen level (33.62 vs 28.44 mg/dL; P = .009). No significant differences were seen between the groups in creatinine levels at day 7 and day 28.
At day 60, dialysis was needed by 10% of the conservative-strategy group and 14% of the liberal-strategy group (P = .06). The important finding here is that there was no trend toward a more frequent need for dialysis in the conservative-strategy group. Also, the average number of days on dialysis in the two groups was essentially the same (11.0 and 10.9, respectively).
Again, there was no difference in the number of renal-failure-free days at either day 7 or day 28.
Hematologic factors. At day 7, the conservative-strategy group had significantly higher hemoglobin (10.22 vs 9.65 g/dL) and albumin (2.30 vs 2.11 g/dL) levels and capillary osmotic pressure (19.18 vs 17.39 mm Hg), even though significantly more patients in the liberal-strategy group received transfusions through day 7 (39% vs 29%; P = .0007).
Safety. Although the number of adverse events—particularly, metabolic alkalosis and electrolyte imbalance—was significantly higher in the conservative-strategy group (42 vs 19; P = .001), the overall incidence was low. No adverse event was associated with arrhythmia.
CONCLUSION
The two fluid-management protocols used in this study were designed to be prudent yet distinctly different. While designing our protocol, we were concerned on the one hand that despite our best efforts fluid balance would turn out to be very similar in the two groups; this did not happen. On the other hand, we were also worried that the fluid level in one of the two groups might turn out to be so bizarre that it would invalidate our study; this too did not occur. Therefore, we are pleased with the way the study was designed and conducted, and we are satisfied that the two protocols were legitimate.
As we went into our study, the literature contained only one other prospective trial that was in some way similar to ours. Mitchell et al9 conducted a randomized, prospective study of 101 critically ill patients, including 89 with pulmonary edema. A group of 52 patients were managed with a conservative strategy intended to reduce the amount of extravascular lung water; the other 49 patients were managed with a strategy similar to the liberal strategy used in our study. At the study’s end, the patients in the conservative-strategy group had a significantly lower amount of extravascular lung water and spent significantly fewer days on ventilation and in the ICU. No clinically significant adverse effects were associated with the conservative strategy. This small study was not highly powered, but it did show that aggressive fluid restriction conferred some benefit.
In our study, the conservative strategy improved lung function and shortened the duration of mechanical ventilation and ICU stay without increasing nonpulmonary organ failures or increasing the risk of death within 60 days. Therefore, we recommend the conservative strategy for patients with ALI/ARDS.
- The Acute Respiratory Distress Syndrome Network.Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 2000; 342:1301–1308.
- Brower RG, Lanken PN, MacIntyre N, et al; National Heart, Lung, and Blood Institute ARDS Clinical Trials Network. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med 2004; 351:327–336.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354:2564–2575.
- Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000; 342:1334–1349.
- Montgomery AB, Stager MA, Carrico CJ, Hudson LD. Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis 1985; 132:485–489.
- Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149:818–824.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wheeler AP, Bernard GR, Thompson BT, et al. Pulmonary-artery versus central venous catheter to guide treatment of acute lung injury. N Engl J Med 2006; 354:2213–2224.
- The National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical TrialsNetwork. Supplementary appendix.http://content.nejm.org/cgi/data/NEJMoa062200/DC1/1.Accessed August 3, 2007.
- Mitchell JP, Schuller D, Calandrino FS, Schuster DP.Improved outcome based on fluid management in criticallyill patients requiring pulmonary artery catheterization.Am Rev Respir Dis 1992; 145:990–998.
Although most clinicians tend to manage acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) by giving more rather than less fluid,1,2 patients may actually fare better under a strategy of limited fluid intake and increased fluid excretion.
According to the results of the Fluids and Catheters Treatment Trial (FACTT),3 patients managed with fluid restriction (the “dry” or conservative strategy) spent significantly less time in the intensive care unit (ICU) and on mechanical ventilation than did patients who received a high fluid intake (the “wet” or liberal strategy). These benefits of the conservative strategy were attained without any increase in the mortality rate at 60 days or in nonpulmonary organ failure at 28 days.
In this article, I discuss the basis for the FACTT researchers’ conclusion that a conservative fluid strategy is preferable to a liberal fluid strategy in ALI/ARDS.
STUDY RATIONALE
One of the more enduring questions in critical care medicine is which fluid-management strategy is best for patients with ALI/ARDS.
The conservative strategy results in a lower vascular filling pressure, which in turn reduces pulmonary edema and improves gas exchange. The drawback to this strategy is that it may have a negative effect on cardiac output and nonpulmonary organ function.
The liberal strategy results in a higher vascular filling pressure, which may be beneficial in terms of cardiac output and nonpulmonary organ perfusion. However, this strategy does not reduce lung edema.
The evidence accumulated before FACTT did not favor one strategy over the other. However, most deaths among patients with ALI/ARDS are attributable to the failure of organs other than the lungs.4,5 As a result, aggressive fluid restriction has not been a common approach in hospitals throughout the United States.1,2
In an effort to resolve the controversy surrounding the management of ALI/ARDS and to broaden the scope of what we know about fluid balance, we undertook this multicenter, randomized, prospective clinical comparison of the two strategies. This study was conducted under the auspices of the National Heart, Lung, and Blood Institute’s Acute Respiratory Distress Syndrome Clinical Trials Network (ARDSnet).
STUDY DESIGN
Between June 8, 2000, and October 3, 2005, we screened more than 11,000 patients with ALI/ARDS at 20 centers in North America.
Eligibility
Eligible patients had experienced ALI/ARDS within the previous 48 hours, had been intubated for positive-pressure ventilation, had a ratio of partial pressure of arterial oxygen (PaO2) to the fraction of inspired oxygen (FIO2) of less than 300, and exhibited bilateral infiltrates on chest radiography that were consistent with the presence of pulmonary edema without evidence of left atrial hypertension.6
Major exclusion criteria included the placement of a pulmonary artery catheter prior to randomization and the presence of certain illnesses that might have compromised the study results.
Patient population
The target enrollment of 1,000 patients was reached. These patients were randomized into one of four roughly equal groups based on the type of fluid-management strategy—conservative or liberal—and the type of catheter that was placed—pulmonary artery or central venous. (The ARDSnet researchers published the results of the catheter comparison in a separate article.7 Those results are not discussed here except to note that there were no statistically significant differences in outcomes between the two catheter groups.)
There were no statistically significant differences between the two groups with respect to baseline demographic characteristics. The conservative-strategy group consisted of 503 patients, of whom 52% were male and 65% were white; the mean age was 50.1 years. The liberal-strategy group consisted of 497 patients, of whom 55% were male and 63% were white; mean age was 49.5 years.
Management
Ventilation according to a low tidal volume strategy (6 mg/kg) was initiated within 1 hour after randomization. The pulmonary artery catheter or central venous catheter was inserted within 4 hours of randomization, and fluid management was started within 2 hours after catheter insertion. Fluid management was continued for 7 days or until 12 hours after extubation in patients who became able to breathe without assistance, whichever occurred first.
Target filling pressures. In the conservative-strategy group, the target filling pressures were low—a pulmonary artery occlusion pressure less than 8 mm Hg for those randomized to receive a pulmonary artery catheter, and a central venous pressure less than 4 mm Hg for those randomized to receive a central venous catheter. Barring adverse effects, patients were to undergo diuresis with furosemide (Lasix) until their goal was achieved, and then they would be maintained on that dosage through day 7. If we experienced difficulty in safely reaching these goals—say, if a patient developed hypoxemia, oliguria, or hypotension—we backed off the diuresis until the patient stabilized, and then we tried again. An inability to reach these filling pressure targets was not considered to be a treatment failure; our actual aim was to get as close to the target as possible as long as the patient tolerated the treatment.
In the liberal-strategy group, the target pressures were in the high-to-normal range—14 to 18 mm Hg for those with a pulmonary artery catheter and 10 to 14 mm Hg for those with a central venous catheter.
Patients with a pulmonary artery catheter who were hemodynamically stable after 3 days could be switched to a central venous catheter at the discretion of the clinician.
Monitoring. Patients were monitored once every 4 hours—more often if the clinician felt it necessary—for four variables:
- Pulmonary artery occlusion pressure or central venous pressure, depending on the type of catheter
- Shock, indicated by a mean arterial pressure of less than 60 mm Hg or the need for a vasopressor
- Oliguria, indicated by a urine output of less than 0.5 mL/kg/hour
- Ineffective circulation, represented by a cardiac index of less than 2.5 L/minute/cm2 in the pulmonary artery catheter group and by the presence of cold, mottled skin and a capillary-refilling time of more than 2 seconds in the central venous catheter group.
Depending on what the clinician found during monitoring, patients could receive a fluid bolus (if the filling pressure was too low), furosemide (if the filling pressure was too high), dobutamine (in certain rare circumstances), or nothing.
We monitored compliance with the protocol instructions twice each day—at a set time each morning and later in the day at a randomly selected time. An important aspect of this study is that we had no protocol instructions for managing shock. Individual clinicians were free to treat shock however they deemed best. In essence, then, our study was a comparison of liberal and conservative strategies during the nonshock phase of ALI/ARDS.
End points
The primary end point was the mortality rate at 60 days. Patients who were discharged earlier were assumed to be alive at 60 days.
The secondary end points were the number of ICU-free and ventilator-free days and the number of organ-failure-free days at day 28. Other end points included various indicators of lung physiology.
Statistical analysis
This intention-to-treat analysis was powered so that we had a 90% chance of detecting a 10% difference in mortality rate at day 60 (statistical significance: P < .05).
Protocol safeguards
Prior to treatment, we knew that some patients in the liberal-strategy group would not reach their filling-pressure targets despite the infusion of large amounts of fluid. To avoid “overdosing” these patients, we limited all patients to a maximum of three fluid boluses per 24 hours. Also, we withheld fluid boluses if a patient’s FIO2 level reached or exceeded 0.7 or if the cardiac index rose to 4.5 L/minute/cm2 or higher.
Diuretics were withheld when a patient had received a vasopressor or had emerged from shock within the preceding 12 hours. Also, diuretics were not given to any patient who had received a fluid bolus within the preceding 12 hours or when renal failure was present (these patients were given renal support therapy).
Finally, physicians and coordinators were instructed to assess each protocol instruction for safety and clinical validity before implementing the particular instruction. If, in their medical judgment, a particular protocol instruction should not be implemented, they were authorized to override the instruction and record the reason for doing so in the case report form.
RESULTS
Protocol compliance
Clinicians adhered to the protocol instructions during approximately 90% of the time.
Diuretic administration. In response to high filling pressures, patients in the conservative-strategy and liberal-strategy groups received furosemide during 41% and 10% of assessment periods, respectively (P < .0001). By day 7, the average patient in the conservative-strategy group had received a cumulative dose of approximately 1,000 mg of furosemide, while the average patient in the liberal-strategy group had received 500 mg.
Fluid administration. Low filling pressure prompted the administration of a fluid bolus to the liberal-strategy group during 15% of the assessment periods, compared with 6% in the conservative-strategy group (P < .0001).
The conservative-strategy patients who were in shock at study entry had a net gain of approximately 3 L of fluid by day 7, while the liberal-strategy group had a gain of approximately 10 L. Among the patients who were shock-free at baseline, the conservative-strategy group had a net loss of almost 2 L at day 7 while the liberal-strategy group had a net gain of about 5 L.
The pulmonary artery occlusion pressure fell from 15.6 mm Hg to just below 13 mm Hg in the conservative-strategy group by day 7, although there was a wide variation among individual patients. The pressure in the liberal-strategy group (15.7 mm Hg at baseline) was unchanged at day 7 (Figure 2).
Primary end point
Secondary end points
Through day 7, the average patient in the conservative-strategy group experienced significantly more ICU-free days (0.9 vs 0.6; P <.001) and more days free of central nervous system (CNS) failure (3.4 vs 2.9; P = .02). No significant differences were observed in the number of days free from coagulation abnormalities and renal or hepatic failure at day 28.
Through day 28, the average patient in the conservative-strategy group experienced significantly more ventilator-free days (14.6 vs 12.1; P < .001). The other 7-day results held up after 28 days, as the average conservative-strategy patient continued to experience more ICU-free days (13.4 vs 11.2; P < .001) and more days free of CNS failure (18.8 vs 17.2; P = .03). Again, no significant differences were observed in the number of days free of coagulation abnormalities and cardiovascular, renal, or hepatic failure.
It is not clear if the conservative strategy’s advantage in terms of more CNS-failure-free days was actually the result of the strategy itself or due to the fact that these patients were weaned off ventilation earlier and therefore received less sedation.
Other outcomes
Shock. One concern we had with the conservative strategy was that it might induce shock more frequently, but this did not occur. The percentage of patients who developed shock at least once during the 7-day treatment protocol was quite similar in the two groups. Also, it is interesting that patients who presented with no baseline shock had only about a 30% chance of developing shock during therapy. There was no significant difference in vasopressor use between the two groups.
Lung function. The conservative-strategy group had a significantly better Murray lung injury score at day 7: 2.03 vs 2.27 (P < .001).
Tidal-volume scores (7.4 mL/kg in both groups at baseline) dropped at an equal rate and were virtually identical at day 7 (6.36 mL/kg in the conservative-strategy group and 6.34 in the liberal-strategy group), as expected.The plateau pressure, positive end-expiraory pressure, PaO2–FIO2 ratio, and oxygenation index were slightly but not significantly better in the conservative-strategy group at day 7.
Overall, lung function was considerably better in the conservative-strategy group.
Cardiovascular function. The mean arterial pressure was significantly lower in the conservative-strategy group at day 7 (81.00 vs 84.36 mm Hg; P = .03). It is interesting that both levels were higher than the baseline levels (77.1 and 77.2, respectively; not significant).
The stroke volume index and the cardiac index were slightly lower in the conservative-strategy group at day 7, but not significantly so. No differences were seen in heart rate and venous oxygen saturation levels.
Renal and metabolic function. At day 7, the conservative-strategy group had a significantly higher blood urea nitrogen level (33.62 vs 28.44 mg/dL; P = .009). No significant differences were seen between the groups in creatinine levels at day 7 and day 28.
At day 60, dialysis was needed by 10% of the conservative-strategy group and 14% of the liberal-strategy group (P = .06). The important finding here is that there was no trend toward a more frequent need for dialysis in the conservative-strategy group. Also, the average number of days on dialysis in the two groups was essentially the same (11.0 and 10.9, respectively).
Again, there was no difference in the number of renal-failure-free days at either day 7 or day 28.
Hematologic factors. At day 7, the conservative-strategy group had significantly higher hemoglobin (10.22 vs 9.65 g/dL) and albumin (2.30 vs 2.11 g/dL) levels and capillary osmotic pressure (19.18 vs 17.39 mm Hg), even though significantly more patients in the liberal-strategy group received transfusions through day 7 (39% vs 29%; P = .0007).
Safety. Although the number of adverse events—particularly, metabolic alkalosis and electrolyte imbalance—was significantly higher in the conservative-strategy group (42 vs 19; P = .001), the overall incidence was low. No adverse event was associated with arrhythmia.
CONCLUSION
The two fluid-management protocols used in this study were designed to be prudent yet distinctly different. While designing our protocol, we were concerned on the one hand that despite our best efforts fluid balance would turn out to be very similar in the two groups; this did not happen. On the other hand, we were also worried that the fluid level in one of the two groups might turn out to be so bizarre that it would invalidate our study; this too did not occur. Therefore, we are pleased with the way the study was designed and conducted, and we are satisfied that the two protocols were legitimate.
As we went into our study, the literature contained only one other prospective trial that was in some way similar to ours. Mitchell et al9 conducted a randomized, prospective study of 101 critically ill patients, including 89 with pulmonary edema. A group of 52 patients were managed with a conservative strategy intended to reduce the amount of extravascular lung water; the other 49 patients were managed with a strategy similar to the liberal strategy used in our study. At the study’s end, the patients in the conservative-strategy group had a significantly lower amount of extravascular lung water and spent significantly fewer days on ventilation and in the ICU. No clinically significant adverse effects were associated with the conservative strategy. This small study was not highly powered, but it did show that aggressive fluid restriction conferred some benefit.
In our study, the conservative strategy improved lung function and shortened the duration of mechanical ventilation and ICU stay without increasing nonpulmonary organ failures or increasing the risk of death within 60 days. Therefore, we recommend the conservative strategy for patients with ALI/ARDS.
Although most clinicians tend to manage acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) by giving more rather than less fluid,1,2 patients may actually fare better under a strategy of limited fluid intake and increased fluid excretion.
According to the results of the Fluids and Catheters Treatment Trial (FACTT),3 patients managed with fluid restriction (the “dry” or conservative strategy) spent significantly less time in the intensive care unit (ICU) and on mechanical ventilation than did patients who received a high fluid intake (the “wet” or liberal strategy). These benefits of the conservative strategy were attained without any increase in the mortality rate at 60 days or in nonpulmonary organ failure at 28 days.
In this article, I discuss the basis for the FACTT researchers’ conclusion that a conservative fluid strategy is preferable to a liberal fluid strategy in ALI/ARDS.
STUDY RATIONALE
One of the more enduring questions in critical care medicine is which fluid-management strategy is best for patients with ALI/ARDS.
The conservative strategy results in a lower vascular filling pressure, which in turn reduces pulmonary edema and improves gas exchange. The drawback to this strategy is that it may have a negative effect on cardiac output and nonpulmonary organ function.
The liberal strategy results in a higher vascular filling pressure, which may be beneficial in terms of cardiac output and nonpulmonary organ perfusion. However, this strategy does not reduce lung edema.
The evidence accumulated before FACTT did not favor one strategy over the other. However, most deaths among patients with ALI/ARDS are attributable to the failure of organs other than the lungs.4,5 As a result, aggressive fluid restriction has not been a common approach in hospitals throughout the United States.1,2
In an effort to resolve the controversy surrounding the management of ALI/ARDS and to broaden the scope of what we know about fluid balance, we undertook this multicenter, randomized, prospective clinical comparison of the two strategies. This study was conducted under the auspices of the National Heart, Lung, and Blood Institute’s Acute Respiratory Distress Syndrome Clinical Trials Network (ARDSnet).
STUDY DESIGN
Between June 8, 2000, and October 3, 2005, we screened more than 11,000 patients with ALI/ARDS at 20 centers in North America.
Eligibility
Eligible patients had experienced ALI/ARDS within the previous 48 hours, had been intubated for positive-pressure ventilation, had a ratio of partial pressure of arterial oxygen (PaO2) to the fraction of inspired oxygen (FIO2) of less than 300, and exhibited bilateral infiltrates on chest radiography that were consistent with the presence of pulmonary edema without evidence of left atrial hypertension.6
Major exclusion criteria included the placement of a pulmonary artery catheter prior to randomization and the presence of certain illnesses that might have compromised the study results.
Patient population
The target enrollment of 1,000 patients was reached. These patients were randomized into one of four roughly equal groups based on the type of fluid-management strategy—conservative or liberal—and the type of catheter that was placed—pulmonary artery or central venous. (The ARDSnet researchers published the results of the catheter comparison in a separate article.7 Those results are not discussed here except to note that there were no statistically significant differences in outcomes between the two catheter groups.)
There were no statistically significant differences between the two groups with respect to baseline demographic characteristics. The conservative-strategy group consisted of 503 patients, of whom 52% were male and 65% were white; the mean age was 50.1 years. The liberal-strategy group consisted of 497 patients, of whom 55% were male and 63% were white; mean age was 49.5 years.
Management
Ventilation according to a low tidal volume strategy (6 mg/kg) was initiated within 1 hour after randomization. The pulmonary artery catheter or central venous catheter was inserted within 4 hours of randomization, and fluid management was started within 2 hours after catheter insertion. Fluid management was continued for 7 days or until 12 hours after extubation in patients who became able to breathe without assistance, whichever occurred first.
Target filling pressures. In the conservative-strategy group, the target filling pressures were low—a pulmonary artery occlusion pressure less than 8 mm Hg for those randomized to receive a pulmonary artery catheter, and a central venous pressure less than 4 mm Hg for those randomized to receive a central venous catheter. Barring adverse effects, patients were to undergo diuresis with furosemide (Lasix) until their goal was achieved, and then they would be maintained on that dosage through day 7. If we experienced difficulty in safely reaching these goals—say, if a patient developed hypoxemia, oliguria, or hypotension—we backed off the diuresis until the patient stabilized, and then we tried again. An inability to reach these filling pressure targets was not considered to be a treatment failure; our actual aim was to get as close to the target as possible as long as the patient tolerated the treatment.
In the liberal-strategy group, the target pressures were in the high-to-normal range—14 to 18 mm Hg for those with a pulmonary artery catheter and 10 to 14 mm Hg for those with a central venous catheter.
Patients with a pulmonary artery catheter who were hemodynamically stable after 3 days could be switched to a central venous catheter at the discretion of the clinician.
Monitoring. Patients were monitored once every 4 hours—more often if the clinician felt it necessary—for four variables:
- Pulmonary artery occlusion pressure or central venous pressure, depending on the type of catheter
- Shock, indicated by a mean arterial pressure of less than 60 mm Hg or the need for a vasopressor
- Oliguria, indicated by a urine output of less than 0.5 mL/kg/hour
- Ineffective circulation, represented by a cardiac index of less than 2.5 L/minute/cm2 in the pulmonary artery catheter group and by the presence of cold, mottled skin and a capillary-refilling time of more than 2 seconds in the central venous catheter group.
Depending on what the clinician found during monitoring, patients could receive a fluid bolus (if the filling pressure was too low), furosemide (if the filling pressure was too high), dobutamine (in certain rare circumstances), or nothing.
We monitored compliance with the protocol instructions twice each day—at a set time each morning and later in the day at a randomly selected time. An important aspect of this study is that we had no protocol instructions for managing shock. Individual clinicians were free to treat shock however they deemed best. In essence, then, our study was a comparison of liberal and conservative strategies during the nonshock phase of ALI/ARDS.
End points
The primary end point was the mortality rate at 60 days. Patients who were discharged earlier were assumed to be alive at 60 days.
The secondary end points were the number of ICU-free and ventilator-free days and the number of organ-failure-free days at day 28. Other end points included various indicators of lung physiology.
Statistical analysis
This intention-to-treat analysis was powered so that we had a 90% chance of detecting a 10% difference in mortality rate at day 60 (statistical significance: P < .05).
Protocol safeguards
Prior to treatment, we knew that some patients in the liberal-strategy group would not reach their filling-pressure targets despite the infusion of large amounts of fluid. To avoid “overdosing” these patients, we limited all patients to a maximum of three fluid boluses per 24 hours. Also, we withheld fluid boluses if a patient’s FIO2 level reached or exceeded 0.7 or if the cardiac index rose to 4.5 L/minute/cm2 or higher.
Diuretics were withheld when a patient had received a vasopressor or had emerged from shock within the preceding 12 hours. Also, diuretics were not given to any patient who had received a fluid bolus within the preceding 12 hours or when renal failure was present (these patients were given renal support therapy).
Finally, physicians and coordinators were instructed to assess each protocol instruction for safety and clinical validity before implementing the particular instruction. If, in their medical judgment, a particular protocol instruction should not be implemented, they were authorized to override the instruction and record the reason for doing so in the case report form.
RESULTS
Protocol compliance
Clinicians adhered to the protocol instructions during approximately 90% of the time.
Diuretic administration. In response to high filling pressures, patients in the conservative-strategy and liberal-strategy groups received furosemide during 41% and 10% of assessment periods, respectively (P < .0001). By day 7, the average patient in the conservative-strategy group had received a cumulative dose of approximately 1,000 mg of furosemide, while the average patient in the liberal-strategy group had received 500 mg.
Fluid administration. Low filling pressure prompted the administration of a fluid bolus to the liberal-strategy group during 15% of the assessment periods, compared with 6% in the conservative-strategy group (P < .0001).
The conservative-strategy patients who were in shock at study entry had a net gain of approximately 3 L of fluid by day 7, while the liberal-strategy group had a gain of approximately 10 L. Among the patients who were shock-free at baseline, the conservative-strategy group had a net loss of almost 2 L at day 7 while the liberal-strategy group had a net gain of about 5 L.
The pulmonary artery occlusion pressure fell from 15.6 mm Hg to just below 13 mm Hg in the conservative-strategy group by day 7, although there was a wide variation among individual patients. The pressure in the liberal-strategy group (15.7 mm Hg at baseline) was unchanged at day 7 (Figure 2).
Primary end point
Secondary end points
Through day 7, the average patient in the conservative-strategy group experienced significantly more ICU-free days (0.9 vs 0.6; P <.001) and more days free of central nervous system (CNS) failure (3.4 vs 2.9; P = .02). No significant differences were observed in the number of days free from coagulation abnormalities and renal or hepatic failure at day 28.
Through day 28, the average patient in the conservative-strategy group experienced significantly more ventilator-free days (14.6 vs 12.1; P < .001). The other 7-day results held up after 28 days, as the average conservative-strategy patient continued to experience more ICU-free days (13.4 vs 11.2; P < .001) and more days free of CNS failure (18.8 vs 17.2; P = .03). Again, no significant differences were observed in the number of days free of coagulation abnormalities and cardiovascular, renal, or hepatic failure.
It is not clear if the conservative strategy’s advantage in terms of more CNS-failure-free days was actually the result of the strategy itself or due to the fact that these patients were weaned off ventilation earlier and therefore received less sedation.
Other outcomes
Shock. One concern we had with the conservative strategy was that it might induce shock more frequently, but this did not occur. The percentage of patients who developed shock at least once during the 7-day treatment protocol was quite similar in the two groups. Also, it is interesting that patients who presented with no baseline shock had only about a 30% chance of developing shock during therapy. There was no significant difference in vasopressor use between the two groups.
Lung function. The conservative-strategy group had a significantly better Murray lung injury score at day 7: 2.03 vs 2.27 (P < .001).
Tidal-volume scores (7.4 mL/kg in both groups at baseline) dropped at an equal rate and were virtually identical at day 7 (6.36 mL/kg in the conservative-strategy group and 6.34 in the liberal-strategy group), as expected.The plateau pressure, positive end-expiraory pressure, PaO2–FIO2 ratio, and oxygenation index were slightly but not significantly better in the conservative-strategy group at day 7.
Overall, lung function was considerably better in the conservative-strategy group.
Cardiovascular function. The mean arterial pressure was significantly lower in the conservative-strategy group at day 7 (81.00 vs 84.36 mm Hg; P = .03). It is interesting that both levels were higher than the baseline levels (77.1 and 77.2, respectively; not significant).
The stroke volume index and the cardiac index were slightly lower in the conservative-strategy group at day 7, but not significantly so. No differences were seen in heart rate and venous oxygen saturation levels.
Renal and metabolic function. At day 7, the conservative-strategy group had a significantly higher blood urea nitrogen level (33.62 vs 28.44 mg/dL; P = .009). No significant differences were seen between the groups in creatinine levels at day 7 and day 28.
At day 60, dialysis was needed by 10% of the conservative-strategy group and 14% of the liberal-strategy group (P = .06). The important finding here is that there was no trend toward a more frequent need for dialysis in the conservative-strategy group. Also, the average number of days on dialysis in the two groups was essentially the same (11.0 and 10.9, respectively).
Again, there was no difference in the number of renal-failure-free days at either day 7 or day 28.
Hematologic factors. At day 7, the conservative-strategy group had significantly higher hemoglobin (10.22 vs 9.65 g/dL) and albumin (2.30 vs 2.11 g/dL) levels and capillary osmotic pressure (19.18 vs 17.39 mm Hg), even though significantly more patients in the liberal-strategy group received transfusions through day 7 (39% vs 29%; P = .0007).
Safety. Although the number of adverse events—particularly, metabolic alkalosis and electrolyte imbalance—was significantly higher in the conservative-strategy group (42 vs 19; P = .001), the overall incidence was low. No adverse event was associated with arrhythmia.
CONCLUSION
The two fluid-management protocols used in this study were designed to be prudent yet distinctly different. While designing our protocol, we were concerned on the one hand that despite our best efforts fluid balance would turn out to be very similar in the two groups; this did not happen. On the other hand, we were also worried that the fluid level in one of the two groups might turn out to be so bizarre that it would invalidate our study; this too did not occur. Therefore, we are pleased with the way the study was designed and conducted, and we are satisfied that the two protocols were legitimate.
As we went into our study, the literature contained only one other prospective trial that was in some way similar to ours. Mitchell et al9 conducted a randomized, prospective study of 101 critically ill patients, including 89 with pulmonary edema. A group of 52 patients were managed with a conservative strategy intended to reduce the amount of extravascular lung water; the other 49 patients were managed with a strategy similar to the liberal strategy used in our study. At the study’s end, the patients in the conservative-strategy group had a significantly lower amount of extravascular lung water and spent significantly fewer days on ventilation and in the ICU. No clinically significant adverse effects were associated with the conservative strategy. This small study was not highly powered, but it did show that aggressive fluid restriction conferred some benefit.
In our study, the conservative strategy improved lung function and shortened the duration of mechanical ventilation and ICU stay without increasing nonpulmonary organ failures or increasing the risk of death within 60 days. Therefore, we recommend the conservative strategy for patients with ALI/ARDS.
- The Acute Respiratory Distress Syndrome Network.Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 2000; 342:1301–1308.
- Brower RG, Lanken PN, MacIntyre N, et al; National Heart, Lung, and Blood Institute ARDS Clinical Trials Network. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med 2004; 351:327–336.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354:2564–2575.
- Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000; 342:1334–1349.
- Montgomery AB, Stager MA, Carrico CJ, Hudson LD. Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis 1985; 132:485–489.
- Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149:818–824.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wheeler AP, Bernard GR, Thompson BT, et al. Pulmonary-artery versus central venous catheter to guide treatment of acute lung injury. N Engl J Med 2006; 354:2213–2224.
- The National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical TrialsNetwork. Supplementary appendix.http://content.nejm.org/cgi/data/NEJMoa062200/DC1/1.Accessed August 3, 2007.
- Mitchell JP, Schuller D, Calandrino FS, Schuster DP.Improved outcome based on fluid management in criticallyill patients requiring pulmonary artery catheterization.Am Rev Respir Dis 1992; 145:990–998.
- The Acute Respiratory Distress Syndrome Network.Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 2000; 342:1301–1308.
- Brower RG, Lanken PN, MacIntyre N, et al; National Heart, Lung, and Blood Institute ARDS Clinical Trials Network. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med 2004; 351:327–336.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354:2564–2575.
- Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000; 342:1334–1349.
- Montgomery AB, Stager MA, Carrico CJ, Hudson LD. Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis 1985; 132:485–489.
- Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149:818–824.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wheeler AP, Bernard GR, Thompson BT, et al. Pulmonary-artery versus central venous catheter to guide treatment of acute lung injury. N Engl J Med 2006; 354:2213–2224.
- The National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical TrialsNetwork. Supplementary appendix.http://content.nejm.org/cgi/data/NEJMoa062200/DC1/1.Accessed August 3, 2007.
- Mitchell JP, Schuller D, Calandrino FS, Schuster DP.Improved outcome based on fluid management in criticallyill patients requiring pulmonary artery catheterization.Am Rev Respir Dis 1992; 145:990–998.
KEY POINTS
- In the conservative-strategy group, the target filling pressures were a pulmonary artery occlusion pressure less than 8 mm Hg for those with a pulmonary artery catheter and a central venous pressure less than 4 mm Hg for those with only a central venous catheter. Pressures were brought into these ranges by diuresis.
- The conservative-strategy group did not experience more frequent need for dialysis or more shock.
- Although the number of adverse events—particularly ,metabolic alkalosis and electrolyte imbalance—was significantly higher in the conservative-strategy group, the overall incidence was low.
When should a methacholine challenge be ordered for a patient with suspected asthma?
The methacholine challenge test isused in several situations:
If the diagnosis of asthma is in question, eg, if the patient has symptoms that suggest asthma (either typical symptoms such as coughing, wheezing, and dyspnea or atypical symptoms) but normal results on regular spirometric testing and no response to a bronchodilator. Because the test has a high negative predictive value, it is more useful in ruling out asthma (if the result is negative) than in ruling it in (if the result is positive).1,2 A negative methacholine challenge test nearly rules out asthma; however, a positive test result needs to be interpreted cautiously if the patient is not experiencing symptoms.
In establishing a diagnosis of occupational asthma. For patients with remitting and relapsing symptoms suggestive of asthma associated with a particular work environment, a detailed history, physical examination, and methacholine challenge test can establish the diagnosis. Specific bronchial challenge testing with the suspected offending agent is possible, although this is more frequently used in research and in situations with significant legal or financial implications for the patient, such as workers’ compensation cases.3
Possibly, in managing asthma. In several clinical trials,4,5 outcomes were better when asthma management decisions were based on airway hyper responsiveness combined with conventional factors (symptoms and lung function) than with management based on conventional factors alone. These findings suggest that asthma management based on serial measurement of airway hyperresponsiveness may be useful in optimizing outcomes of care; however, adjustment in treatment according to response to serial methacholine challenge tests is currently not recommended for routine management of asthma.
In clinical research.
OBSTRUCTION CAN BE IMPROVED OR PROVOKED
Asthma is a chronic inflammatory disorder of the airways associated with characteristic clinical symptoms of wheezing, chest tightness, breathlessness, and cough. These symptoms may be associated with airflow limitation that is at least partially reversible, either spontaneously or with treatment.
Spirometry can confirm the diagnosis of asthma if lung function improves after a bronchodilator is given, as reflected by an increase in forced expiratory volume in 1 second (FEV1) of more than 12% and more than 0.2 L.6,7
Conversely, during bronchoprovocation testing, airflow obstruction is provoked by a stimulus known to elicit airway narrowing, such as inhaled methacholine. Bronchial hyperresponsiveness can reliably distinguish patients with asthma from those without asthma.
HOW THE TEST IS DONE
During the test, the patient inhales methacholine aerosols in increasing concentrations; various protocols can be used. Spirometry is performed before and after each dose, and the results are reported as a percent decrease in FEV1 from baseline for each step of the protocol.
A positive reaction is a 20% fall in FEV1, and the provocative concentration that causes a positive reaction (the PC20) is used to indicate the level of airway hyperresponsiveness. If the FEV1 does not fall by at least 20% with the highest concentration of methacholine, the testis interpreted as negative and the PC20 is reported as “more than 16 mg/mL” or “more than 25 mg/mL,” depending on the highest dose given.
The maximum dose of methacholine varies among pulmonary function testing laboratories and asthma specialists; final doses of 16, 25, and 32 mg/mL are commonly used. Studies have defined a range of 8 to 16 mg/mL as an optimal cutoff point to separate patients with asthma from those without asthma.2,6,7
The response to methacholine can also be expressed in terms of specific airway conductance;however, this is more complicated and requires body plethysmography.
Other stimuli that can be used as bronchoprovocation challenges to diagnose asthma include inhaled histamine, exposure to cold air, or eucapneic hyperventilation.Compared with these alternative stimuli, methacholine is the most feasible as it does not require extensive equipment and is better tolerated than histamine.8
POTENTIAL COMPLICATIONS
Methacholine elicits airway narrowing in susceptible people and can cause severe bronchoconstriction, hyperinflation, or severe coughing. However, this procedure is generally well tolerated, and respiratory symptoms inpatients who react to methacholine typically reverse promptly in response to bronchodilators.
Nevertheless, the test should be performed in a pulmonary function laboratory or doctor’s office with available personnel trained to treat acute bronchospasm and to use resuscitation equipment if needed. Informed consent should be obtained and recorded in the medical record after a detailed explanation of the risks and benefits of this procedure and alternatives to it.
CONTRAINDICATIONS
Baseline obstruction. A ratio of FEV1 to forced vital capacity less than 70% on baseline spirometry defines airway obstruction, and methacholine challenge for diagnostic purposes would not be indicated.
Furthermore, patients with low baseline lung function, who may not be able to compensate for a further decline in lung function due to methacholine-induced bronchospasm, are at increased risk of a serious respiratory reaction. For this reason, an FEV1 less than 50% of predicted or less than 1.0 L is an absolute contraindication to methacholine challenge testing, and an FEV1 less than 60% of predicted or less than 1.5 L must be evaluated on an individual basis.9
Myocardial infarction or stroke within the previous 3 months, uncontrolled hypertension, and aortic or cerebral aneurysm are absolute contraindications to this procedure, since induced bronchospasm may cause ventilation-perfusion mismatching resulting in arterial hypoxemia and compensatory changes in blood pressure, cardiac output, and heart rate. There is no increased risk of cardiac arrhythmia during methacholine challenge.10
Pregnancy is a relative contraindication to methacholine challenge testing; metha- choline is classified in pregnancy category C.
Inability to perform spirometry correctly is also a relative contraindication, and therefore this test is not recommended for preschool-age children.
SOME DRUGS SHOULD BE HELD
Other factors that can confound the results include smoking,16 respiratory infection, exercise, and consumption of caffeine (coffee, tea, chocolate, or cola drinks) on the day of the test. Airway responsiveness may worsen due to exposure to allergen or upper airway viral infections. Vigorous exercise could induce bronchoconstriction; therefore, performing other bronchial challenge procedures or exercise testing immediately before methacholine challenge may affect the results.17,18
Bronchial hyperresponsiveness is seen in a variety of disorders other than asthma, such as smoking-induced chronic airflow limitation, congestive heart failure, sarcoidosis, cysticfibrosis, and bronchiectasis, as well as in siblings of asthmatics and in people with allergic rhinitis.19 In these situations, the methacholine test can be falsely positive, and one should interpret the results in the context of the clinical history.
- Gilbert R, Auchincloss JH. Post-test probability of asthma following methacholine challenge. Chest 1990; 97:562–565.
- Perpina M, Pellicer C, de Diego A, Compte L, Macian V. Diagnostic value of the bronchial provocation test with methacholine in asthma: a Bayesian analysis approach. Chest 1993; 104:149–154.
- Tan RA, Spector SL. Provocation studies in the diagnosis of occupational asthma. Immunol Allergy Clin North Am 2003; 23:251–267.
- Sont JK, Willems LN, Bel EH, van Krieken JH, Vandenbroucke JP, Sterk PJ. Clinical control and histopathologic outcome of asthma when using airway hyperresponsiveness as an additional guide to long-term treatment. The AMPUL Study Group. Am J Respir Crit Care Med 1999; 159:1043–1051.
- Green RH, Brightling CE, McKenna S, et al. Asthma exacerbations and sputum eosinophil counts: a randomized controlled trial. Lancet 2002; 360:1715–1721.
- Crapo RO, Casaburi R, Coates AL, et al. Guidelines for methacholine and exercise challenge testing—1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med 2000; 161:309–329.
- Miller MR, Hankinson J, Brusasco V, et al; ATS/ERS Task Force. Standardisation of spirometry. Eur Respir J 2005; 26:319–338.
- Fish JE, Kelly JF. Measurements of responsiveness in bronchoprovocation testing. J Allergy Clin Immunol 1979; 64:592–596.
- Martin RJ, Wanger JS, Irwin CG, Bucher Bartelson B, Cherniac RM. Methacholine challenge testing: safety of low starting FEV1. Asthma Clinical Research Network (ACRN). Chest 1997; 112:53–56.
- Malerba M, Radaeli A, Politi A, Ceriani L, Zulli R, Grassi V. Cardiac arrhythmia monitoring during bronchial provocation test with methacholine. Chest 2003; 124:813–818.
- Cockcroft DW, Swystun VA, Bhagat R. Interaction of inhaled beta 2 agonist and inhaled corticosteroid on airway responsiveness to allergen and methacholine. Am J Respir Crit Care Med 1995; 152:1485–1489.
- Reid JK, Davis BE, Cockcroft DW. The effect of ipratropium nasal spray on bronchial methacholine challenge. Chest 2005; 128:1245–1247.
- O’Connor BJ, Towse LJ, Barnes PJ. Prolonged effect of tiotropium bromide on methacholine-induced bronchoconstriction in asthma. Am J Respir Crit Care Med 1996; 154:876–880.
- Juniper EF, Kline PA, Vanzieleghem MA, Ramsdale EH, O’Byrne PM, Hargreave FE. Effect of long-term treatment with an inhaled corticosteroid (budesonide) on airway hyperresponsiveness and clinical asthma in nonsteroid-dependent asthmatics. Am Rev Respir Dis 1990; 142:832–836.
- Freezer NJ, Croasdell H, Doull IJ, Holgate ST. Effect of regular inhaled beclomethasone on exercise and methacholine airway responses in school children with recurrent wheeze. Eur Respir J 1995; 8:1488–1493.
- Jensen EJ, Dahl R, Steffensen F. Bronchial reactivity to cigarette smoke in smokers: repeatability, relationship to methacholine reactivity, smoking and atopy. Eu rRespir J 1998; 11:670–676.
- Cheung D, Dick EC, Timmers MC, de Klerk EP, Spaan WJ, Sterk PJ. Rhinovirus inhalation causes longlasting excessive airway narrowing in response to methacholine in asthmatic subjects in vivo. Am J Respir Crit Care Med 1995; 152:1490–1496.
- Dinh Xuan AT, Lockart A. Use of non-specific bronchial challenges in the assessment of anti-asthmatic drugs. Eur Respir Rev 1991; 1:19–24.
- Ramsdell JW, Nachtwey FJ, Moser KM. Bronchia lhyperreactivity in chronic obstructive bronchitis. Am Rev Respir Dis 1982; 126:829–832.
The methacholine challenge test isused in several situations:
If the diagnosis of asthma is in question, eg, if the patient has symptoms that suggest asthma (either typical symptoms such as coughing, wheezing, and dyspnea or atypical symptoms) but normal results on regular spirometric testing and no response to a bronchodilator. Because the test has a high negative predictive value, it is more useful in ruling out asthma (if the result is negative) than in ruling it in (if the result is positive).1,2 A negative methacholine challenge test nearly rules out asthma; however, a positive test result needs to be interpreted cautiously if the patient is not experiencing symptoms.
In establishing a diagnosis of occupational asthma. For patients with remitting and relapsing symptoms suggestive of asthma associated with a particular work environment, a detailed history, physical examination, and methacholine challenge test can establish the diagnosis. Specific bronchial challenge testing with the suspected offending agent is possible, although this is more frequently used in research and in situations with significant legal or financial implications for the patient, such as workers’ compensation cases.3
Possibly, in managing asthma. In several clinical trials,4,5 outcomes were better when asthma management decisions were based on airway hyper responsiveness combined with conventional factors (symptoms and lung function) than with management based on conventional factors alone. These findings suggest that asthma management based on serial measurement of airway hyperresponsiveness may be useful in optimizing outcomes of care; however, adjustment in treatment according to response to serial methacholine challenge tests is currently not recommended for routine management of asthma.
In clinical research.
OBSTRUCTION CAN BE IMPROVED OR PROVOKED
Asthma is a chronic inflammatory disorder of the airways associated with characteristic clinical symptoms of wheezing, chest tightness, breathlessness, and cough. These symptoms may be associated with airflow limitation that is at least partially reversible, either spontaneously or with treatment.
Spirometry can confirm the diagnosis of asthma if lung function improves after a bronchodilator is given, as reflected by an increase in forced expiratory volume in 1 second (FEV1) of more than 12% and more than 0.2 L.6,7
Conversely, during bronchoprovocation testing, airflow obstruction is provoked by a stimulus known to elicit airway narrowing, such as inhaled methacholine. Bronchial hyperresponsiveness can reliably distinguish patients with asthma from those without asthma.
HOW THE TEST IS DONE
During the test, the patient inhales methacholine aerosols in increasing concentrations; various protocols can be used. Spirometry is performed before and after each dose, and the results are reported as a percent decrease in FEV1 from baseline for each step of the protocol.
A positive reaction is a 20% fall in FEV1, and the provocative concentration that causes a positive reaction (the PC20) is used to indicate the level of airway hyperresponsiveness. If the FEV1 does not fall by at least 20% with the highest concentration of methacholine, the testis interpreted as negative and the PC20 is reported as “more than 16 mg/mL” or “more than 25 mg/mL,” depending on the highest dose given.
The maximum dose of methacholine varies among pulmonary function testing laboratories and asthma specialists; final doses of 16, 25, and 32 mg/mL are commonly used. Studies have defined a range of 8 to 16 mg/mL as an optimal cutoff point to separate patients with asthma from those without asthma.2,6,7
The response to methacholine can also be expressed in terms of specific airway conductance;however, this is more complicated and requires body plethysmography.
Other stimuli that can be used as bronchoprovocation challenges to diagnose asthma include inhaled histamine, exposure to cold air, or eucapneic hyperventilation.Compared with these alternative stimuli, methacholine is the most feasible as it does not require extensive equipment and is better tolerated than histamine.8
POTENTIAL COMPLICATIONS
Methacholine elicits airway narrowing in susceptible people and can cause severe bronchoconstriction, hyperinflation, or severe coughing. However, this procedure is generally well tolerated, and respiratory symptoms inpatients who react to methacholine typically reverse promptly in response to bronchodilators.
Nevertheless, the test should be performed in a pulmonary function laboratory or doctor’s office with available personnel trained to treat acute bronchospasm and to use resuscitation equipment if needed. Informed consent should be obtained and recorded in the medical record after a detailed explanation of the risks and benefits of this procedure and alternatives to it.
CONTRAINDICATIONS
Baseline obstruction. A ratio of FEV1 to forced vital capacity less than 70% on baseline spirometry defines airway obstruction, and methacholine challenge for diagnostic purposes would not be indicated.
Furthermore, patients with low baseline lung function, who may not be able to compensate for a further decline in lung function due to methacholine-induced bronchospasm, are at increased risk of a serious respiratory reaction. For this reason, an FEV1 less than 50% of predicted or less than 1.0 L is an absolute contraindication to methacholine challenge testing, and an FEV1 less than 60% of predicted or less than 1.5 L must be evaluated on an individual basis.9
Myocardial infarction or stroke within the previous 3 months, uncontrolled hypertension, and aortic or cerebral aneurysm are absolute contraindications to this procedure, since induced bronchospasm may cause ventilation-perfusion mismatching resulting in arterial hypoxemia and compensatory changes in blood pressure, cardiac output, and heart rate. There is no increased risk of cardiac arrhythmia during methacholine challenge.10
Pregnancy is a relative contraindication to methacholine challenge testing; metha- choline is classified in pregnancy category C.
Inability to perform spirometry correctly is also a relative contraindication, and therefore this test is not recommended for preschool-age children.
SOME DRUGS SHOULD BE HELD
Other factors that can confound the results include smoking,16 respiratory infection, exercise, and consumption of caffeine (coffee, tea, chocolate, or cola drinks) on the day of the test. Airway responsiveness may worsen due to exposure to allergen or upper airway viral infections. Vigorous exercise could induce bronchoconstriction; therefore, performing other bronchial challenge procedures or exercise testing immediately before methacholine challenge may affect the results.17,18
Bronchial hyperresponsiveness is seen in a variety of disorders other than asthma, such as smoking-induced chronic airflow limitation, congestive heart failure, sarcoidosis, cysticfibrosis, and bronchiectasis, as well as in siblings of asthmatics and in people with allergic rhinitis.19 In these situations, the methacholine test can be falsely positive, and one should interpret the results in the context of the clinical history.
The methacholine challenge test isused in several situations:
If the diagnosis of asthma is in question, eg, if the patient has symptoms that suggest asthma (either typical symptoms such as coughing, wheezing, and dyspnea or atypical symptoms) but normal results on regular spirometric testing and no response to a bronchodilator. Because the test has a high negative predictive value, it is more useful in ruling out asthma (if the result is negative) than in ruling it in (if the result is positive).1,2 A negative methacholine challenge test nearly rules out asthma; however, a positive test result needs to be interpreted cautiously if the patient is not experiencing symptoms.
In establishing a diagnosis of occupational asthma. For patients with remitting and relapsing symptoms suggestive of asthma associated with a particular work environment, a detailed history, physical examination, and methacholine challenge test can establish the diagnosis. Specific bronchial challenge testing with the suspected offending agent is possible, although this is more frequently used in research and in situations with significant legal or financial implications for the patient, such as workers’ compensation cases.3
Possibly, in managing asthma. In several clinical trials,4,5 outcomes were better when asthma management decisions were based on airway hyper responsiveness combined with conventional factors (symptoms and lung function) than with management based on conventional factors alone. These findings suggest that asthma management based on serial measurement of airway hyperresponsiveness may be useful in optimizing outcomes of care; however, adjustment in treatment according to response to serial methacholine challenge tests is currently not recommended for routine management of asthma.
In clinical research.
OBSTRUCTION CAN BE IMPROVED OR PROVOKED
Asthma is a chronic inflammatory disorder of the airways associated with characteristic clinical symptoms of wheezing, chest tightness, breathlessness, and cough. These symptoms may be associated with airflow limitation that is at least partially reversible, either spontaneously or with treatment.
Spirometry can confirm the diagnosis of asthma if lung function improves after a bronchodilator is given, as reflected by an increase in forced expiratory volume in 1 second (FEV1) of more than 12% and more than 0.2 L.6,7
Conversely, during bronchoprovocation testing, airflow obstruction is provoked by a stimulus known to elicit airway narrowing, such as inhaled methacholine. Bronchial hyperresponsiveness can reliably distinguish patients with asthma from those without asthma.
HOW THE TEST IS DONE
During the test, the patient inhales methacholine aerosols in increasing concentrations; various protocols can be used. Spirometry is performed before and after each dose, and the results are reported as a percent decrease in FEV1 from baseline for each step of the protocol.
A positive reaction is a 20% fall in FEV1, and the provocative concentration that causes a positive reaction (the PC20) is used to indicate the level of airway hyperresponsiveness. If the FEV1 does not fall by at least 20% with the highest concentration of methacholine, the testis interpreted as negative and the PC20 is reported as “more than 16 mg/mL” or “more than 25 mg/mL,” depending on the highest dose given.
The maximum dose of methacholine varies among pulmonary function testing laboratories and asthma specialists; final doses of 16, 25, and 32 mg/mL are commonly used. Studies have defined a range of 8 to 16 mg/mL as an optimal cutoff point to separate patients with asthma from those without asthma.2,6,7
The response to methacholine can also be expressed in terms of specific airway conductance;however, this is more complicated and requires body plethysmography.
Other stimuli that can be used as bronchoprovocation challenges to diagnose asthma include inhaled histamine, exposure to cold air, or eucapneic hyperventilation.Compared with these alternative stimuli, methacholine is the most feasible as it does not require extensive equipment and is better tolerated than histamine.8
POTENTIAL COMPLICATIONS
Methacholine elicits airway narrowing in susceptible people and can cause severe bronchoconstriction, hyperinflation, or severe coughing. However, this procedure is generally well tolerated, and respiratory symptoms inpatients who react to methacholine typically reverse promptly in response to bronchodilators.
Nevertheless, the test should be performed in a pulmonary function laboratory or doctor’s office with available personnel trained to treat acute bronchospasm and to use resuscitation equipment if needed. Informed consent should be obtained and recorded in the medical record after a detailed explanation of the risks and benefits of this procedure and alternatives to it.
CONTRAINDICATIONS
Baseline obstruction. A ratio of FEV1 to forced vital capacity less than 70% on baseline spirometry defines airway obstruction, and methacholine challenge for diagnostic purposes would not be indicated.
Furthermore, patients with low baseline lung function, who may not be able to compensate for a further decline in lung function due to methacholine-induced bronchospasm, are at increased risk of a serious respiratory reaction. For this reason, an FEV1 less than 50% of predicted or less than 1.0 L is an absolute contraindication to methacholine challenge testing, and an FEV1 less than 60% of predicted or less than 1.5 L must be evaluated on an individual basis.9
Myocardial infarction or stroke within the previous 3 months, uncontrolled hypertension, and aortic or cerebral aneurysm are absolute contraindications to this procedure, since induced bronchospasm may cause ventilation-perfusion mismatching resulting in arterial hypoxemia and compensatory changes in blood pressure, cardiac output, and heart rate. There is no increased risk of cardiac arrhythmia during methacholine challenge.10
Pregnancy is a relative contraindication to methacholine challenge testing; metha- choline is classified in pregnancy category C.
Inability to perform spirometry correctly is also a relative contraindication, and therefore this test is not recommended for preschool-age children.
SOME DRUGS SHOULD BE HELD
Other factors that can confound the results include smoking,16 respiratory infection, exercise, and consumption of caffeine (coffee, tea, chocolate, or cola drinks) on the day of the test. Airway responsiveness may worsen due to exposure to allergen or upper airway viral infections. Vigorous exercise could induce bronchoconstriction; therefore, performing other bronchial challenge procedures or exercise testing immediately before methacholine challenge may affect the results.17,18
Bronchial hyperresponsiveness is seen in a variety of disorders other than asthma, such as smoking-induced chronic airflow limitation, congestive heart failure, sarcoidosis, cysticfibrosis, and bronchiectasis, as well as in siblings of asthmatics and in people with allergic rhinitis.19 In these situations, the methacholine test can be falsely positive, and one should interpret the results in the context of the clinical history.
- Gilbert R, Auchincloss JH. Post-test probability of asthma following methacholine challenge. Chest 1990; 97:562–565.
- Perpina M, Pellicer C, de Diego A, Compte L, Macian V. Diagnostic value of the bronchial provocation test with methacholine in asthma: a Bayesian analysis approach. Chest 1993; 104:149–154.
- Tan RA, Spector SL. Provocation studies in the diagnosis of occupational asthma. Immunol Allergy Clin North Am 2003; 23:251–267.
- Sont JK, Willems LN, Bel EH, van Krieken JH, Vandenbroucke JP, Sterk PJ. Clinical control and histopathologic outcome of asthma when using airway hyperresponsiveness as an additional guide to long-term treatment. The AMPUL Study Group. Am J Respir Crit Care Med 1999; 159:1043–1051.
- Green RH, Brightling CE, McKenna S, et al. Asthma exacerbations and sputum eosinophil counts: a randomized controlled trial. Lancet 2002; 360:1715–1721.
- Crapo RO, Casaburi R, Coates AL, et al. Guidelines for methacholine and exercise challenge testing—1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med 2000; 161:309–329.
- Miller MR, Hankinson J, Brusasco V, et al; ATS/ERS Task Force. Standardisation of spirometry. Eur Respir J 2005; 26:319–338.
- Fish JE, Kelly JF. Measurements of responsiveness in bronchoprovocation testing. J Allergy Clin Immunol 1979; 64:592–596.
- Martin RJ, Wanger JS, Irwin CG, Bucher Bartelson B, Cherniac RM. Methacholine challenge testing: safety of low starting FEV1. Asthma Clinical Research Network (ACRN). Chest 1997; 112:53–56.
- Malerba M, Radaeli A, Politi A, Ceriani L, Zulli R, Grassi V. Cardiac arrhythmia monitoring during bronchial provocation test with methacholine. Chest 2003; 124:813–818.
- Cockcroft DW, Swystun VA, Bhagat R. Interaction of inhaled beta 2 agonist and inhaled corticosteroid on airway responsiveness to allergen and methacholine. Am J Respir Crit Care Med 1995; 152:1485–1489.
- Reid JK, Davis BE, Cockcroft DW. The effect of ipratropium nasal spray on bronchial methacholine challenge. Chest 2005; 128:1245–1247.
- O’Connor BJ, Towse LJ, Barnes PJ. Prolonged effect of tiotropium bromide on methacholine-induced bronchoconstriction in asthma. Am J Respir Crit Care Med 1996; 154:876–880.
- Juniper EF, Kline PA, Vanzieleghem MA, Ramsdale EH, O’Byrne PM, Hargreave FE. Effect of long-term treatment with an inhaled corticosteroid (budesonide) on airway hyperresponsiveness and clinical asthma in nonsteroid-dependent asthmatics. Am Rev Respir Dis 1990; 142:832–836.
- Freezer NJ, Croasdell H, Doull IJ, Holgate ST. Effect of regular inhaled beclomethasone on exercise and methacholine airway responses in school children with recurrent wheeze. Eur Respir J 1995; 8:1488–1493.
- Jensen EJ, Dahl R, Steffensen F. Bronchial reactivity to cigarette smoke in smokers: repeatability, relationship to methacholine reactivity, smoking and atopy. Eu rRespir J 1998; 11:670–676.
- Cheung D, Dick EC, Timmers MC, de Klerk EP, Spaan WJ, Sterk PJ. Rhinovirus inhalation causes longlasting excessive airway narrowing in response to methacholine in asthmatic subjects in vivo. Am J Respir Crit Care Med 1995; 152:1490–1496.
- Dinh Xuan AT, Lockart A. Use of non-specific bronchial challenges in the assessment of anti-asthmatic drugs. Eur Respir Rev 1991; 1:19–24.
- Ramsdell JW, Nachtwey FJ, Moser KM. Bronchia lhyperreactivity in chronic obstructive bronchitis. Am Rev Respir Dis 1982; 126:829–832.
- Gilbert R, Auchincloss JH. Post-test probability of asthma following methacholine challenge. Chest 1990; 97:562–565.
- Perpina M, Pellicer C, de Diego A, Compte L, Macian V. Diagnostic value of the bronchial provocation test with methacholine in asthma: a Bayesian analysis approach. Chest 1993; 104:149–154.
- Tan RA, Spector SL. Provocation studies in the diagnosis of occupational asthma. Immunol Allergy Clin North Am 2003; 23:251–267.
- Sont JK, Willems LN, Bel EH, van Krieken JH, Vandenbroucke JP, Sterk PJ. Clinical control and histopathologic outcome of asthma when using airway hyperresponsiveness as an additional guide to long-term treatment. The AMPUL Study Group. Am J Respir Crit Care Med 1999; 159:1043–1051.
- Green RH, Brightling CE, McKenna S, et al. Asthma exacerbations and sputum eosinophil counts: a randomized controlled trial. Lancet 2002; 360:1715–1721.
- Crapo RO, Casaburi R, Coates AL, et al. Guidelines for methacholine and exercise challenge testing—1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med 2000; 161:309–329.
- Miller MR, Hankinson J, Brusasco V, et al; ATS/ERS Task Force. Standardisation of spirometry. Eur Respir J 2005; 26:319–338.
- Fish JE, Kelly JF. Measurements of responsiveness in bronchoprovocation testing. J Allergy Clin Immunol 1979; 64:592–596.
- Martin RJ, Wanger JS, Irwin CG, Bucher Bartelson B, Cherniac RM. Methacholine challenge testing: safety of low starting FEV1. Asthma Clinical Research Network (ACRN). Chest 1997; 112:53–56.
- Malerba M, Radaeli A, Politi A, Ceriani L, Zulli R, Grassi V. Cardiac arrhythmia monitoring during bronchial provocation test with methacholine. Chest 2003; 124:813–818.
- Cockcroft DW, Swystun VA, Bhagat R. Interaction of inhaled beta 2 agonist and inhaled corticosteroid on airway responsiveness to allergen and methacholine. Am J Respir Crit Care Med 1995; 152:1485–1489.
- Reid JK, Davis BE, Cockcroft DW. The effect of ipratropium nasal spray on bronchial methacholine challenge. Chest 2005; 128:1245–1247.
- O’Connor BJ, Towse LJ, Barnes PJ. Prolonged effect of tiotropium bromide on methacholine-induced bronchoconstriction in asthma. Am J Respir Crit Care Med 1996; 154:876–880.
- Juniper EF, Kline PA, Vanzieleghem MA, Ramsdale EH, O’Byrne PM, Hargreave FE. Effect of long-term treatment with an inhaled corticosteroid (budesonide) on airway hyperresponsiveness and clinical asthma in nonsteroid-dependent asthmatics. Am Rev Respir Dis 1990; 142:832–836.
- Freezer NJ, Croasdell H, Doull IJ, Holgate ST. Effect of regular inhaled beclomethasone on exercise and methacholine airway responses in school children with recurrent wheeze. Eur Respir J 1995; 8:1488–1493.
- Jensen EJ, Dahl R, Steffensen F. Bronchial reactivity to cigarette smoke in smokers: repeatability, relationship to methacholine reactivity, smoking and atopy. Eu rRespir J 1998; 11:670–676.
- Cheung D, Dick EC, Timmers MC, de Klerk EP, Spaan WJ, Sterk PJ. Rhinovirus inhalation causes longlasting excessive airway narrowing in response to methacholine in asthmatic subjects in vivo. Am J Respir Crit Care Med 1995; 152:1490–1496.
- Dinh Xuan AT, Lockart A. Use of non-specific bronchial challenges in the assessment of anti-asthmatic drugs. Eur Respir Rev 1991; 1:19–24.
- Ramsdell JW, Nachtwey FJ, Moser KM. Bronchia lhyperreactivity in chronic obstructive bronchitis. Am Rev Respir Dis 1982; 126:829–832.
Acute aortic syndromes: Time to talk of many things
“The time has come,” the Walrus said,
“To talk of many things:
Of shoes—and ships—and sealing-wax—
Of cabbages—and kings—
And why the sea is boiling hot
And whether pigs have wings.”
—Lewis Carroll, The Walrus and the Carpenter (from Through the Looking-Glass and What Alice Found There, 1872).
Lewis Carroll's poem of 1872 is a useful starting point for identifying issues resulting from confusion over the variously described acute aortic syndromes—and, for oysters, the dangers of listening to walruses.
TALK OF MANY THINGS
In cases of aortic dissection (splitting or separation of the layers of the aortic wall), it is important to establish the type (ie, the location and extent) and class (ie, the structure) of the dissection, because these distinctions determine the treatment.1 Similarly, in cases of painful or leaking degenerative aneurysms, we need to know the location of the aneurysm and whether the presenting pain is from compression of surrounding tissue, particularly of the vertebral bodies, or from leakage.
The location and extent of an aortic dissection can be classified in three ways (see Figure 3 in Smith and Schoenhagen’s excellent review of the use of computed tomography [CT] in acute aortic syndromes in this issue of the Cleveland Clinic Journal of Medicine2):
- The DeBakey system (type I, II, or III)
- The Stanford system (type A or B)
- Distal or proximal to the left subclavian artery.
Of note, the DeBakey system does not include tears in the arch that extend distally without ascending involvement. The original Stanford system included arch tears with distal extension in type B; hence, type B excluded all patients without ascending involvement.
The importance of the extent of dissection is that most patients with Stanford type A or DeBakey type I or II dissections should undergo immediate surgery, as most of them would die without it. Surgery is also indicated for arch tears (non-DeBakey, original Stanford type B).
Because these classifications are somewhat confusing, the simplest approach is to note whether the dissection extends proximal or distal to the left subclavian artery, because proximal dissections need surgery and distal ones are first managed medically.
The classes of dissection also have bearing on treatment.1 These are:
- Class I—classic aortic dissection in the media with two lumens separated by a “flap” or septum
- Class II—intramural hematoma in the aortic wall from dissection in which the intimal tear cannot be imaged (these are nearly always found duringsurgery or autopsy)
- Class III—localized confined intimal tears without extensive undermining of the intima or flap formation. These are often seen with Marfan syndrome and can rupture or cause tamponade, as can any type of proximal dissection. The typical appearance is of a bulging bubble in the aortic wall.
- Class IV—penetrating atherosclerotic ulcers with localized dissections or wall hematomas, often with calcium at the base of a mushroom-shaped area of extraluminal contrast. Of note, the plane of dissection is often between the media and adventitia.
- Class V—iatrogenic or posttraumatic dissection.
All class I to class IV tears of the proximal aorta require surgery, whereas distal class IV and V tears may require either open or endovascular surgical intervention. Surgery is also indicated for patients with distal dissections who have severe narrowing of the true lumen, distal ischemia, uncontrolled pain, severe hypertension, or evidence of leaking, particularly with class IV tears.
In distal dissections that are subacute (2–6 week sold), the Investigation of Stent grafts in Patients With Type B Aortic Dissection (INSTEAD) trial found that inserting a stent prophylactically provided no benefit. Further-more, there is no proof that stenting is beneficial if the aortic dissection is chronic, ie, more than 6 weeks old.1,3–5
WHICH SHOE FITS?
There is no ideal procedure to detect dissection, although the trend is towards CT angiography, as Smith and Schoenhagen report.2 Although some investigators have optimistically estimated CT’s sensitivity and specificity as 100%, cardiovascular surgeons are well aware of both false-positive and false-negative CT studies. Thus, for emergency repairs of proximal dissections, transesophageal echocardiography should be done after intubation and before opening a patient’s chest if time allows. Magnetic resonance imaging, CT, and transesophageal echocardiography may all miss class III tears, but these are frequently evidenced by eccentric “bubbles”or “ballooning.”1
SHIPS
Patients with either acute aortic dissection or severe pain associated with degenerative aneurysms need to be “shipped” promptly to a tertiary medical center after diagnosis, since larger volumes of procedures appear to be associated with better outcomes.
SEALING WAX
Using current surgical methods, the aortic valve can be preserved during aortic dissection repair unless the valve is bicuspid or the patient has Marfan syndrome.1,3,4,6–8
Sealing wax in the form of biological glues, rather than for letters, is a new innovation. A caveat remains, however: we have seen patients who have required reoperation for false aneurysms or infection. Hence, glues should be used with caution.
CABBAGES
A dilemma is whether patients should undergo coronary catheterization (or CT angiography—a separate question) and subsequent coronary artery bypass grafting (CABG), if needed, at the time of aortic dissection repair. The problem is that approximately one-third of patients have coronary artery disease that may require CABG, but the delay for catheterization increases the risk of rupture or tamponade before surgery.
Indeed, 40% of patients with proximal dissections die immediately, and 1% to 3% die in the hour before surgery. The short-term (in-hospital and 30-day) mortality rates range from 3.4% (Cleveland Clinic 2006 data) to 25%, and of the survivors only about 50% area live 5 years after surgery.
Though dismal, the prognosis is improving. In 162 patients with aortic dissection and Marfan syndrome or connective tissue disorders who underwent surgery at Cleveland Clinic in the years 1978–2003, the 5-year survival rate in those with aortic dissection was 75% and the 10-year rate was 55%.7 In those without dissection, the 10-year survival rate was approximately 90% (P < .001).
KINGS
Noted personalities who have had aortic dissection include King George II of England (who died in 1760), Lucille Ball, Conway Twitty, Jan Larson, and most recently John Ritter. None of these famous people survived their aortic dissections. Indeed, dissection and diseases of the aorta or its branches cause between 43,000 and 47,000 deaths annually,9 more than from breast cancer, murders, or motor vehicle accidents. The main reason for these dismal statistics is that the disease is often misdiagnosed at the time of presentation.
BOILING SEA
Careful studies from Olmsted County, Minnesota,10 have shown a tripling of the incidence of aortic disease, particularly in women, even though the rate of deaths from coronary artery disease has been decreasing. Furthermore, Olsson et al11 report that the incidence of aortic dissection in men in Sweden increased to approximately16 per 100,000 per year from 1987 to 2002, a 52% increase. The aging of the population must play a large role, but other factors may exist that are not well understood or defined and require further research.
PIGS HAVE WINGS
Will it be possible to overcome this rising problem? The answer is a definite yes. The results of aortic surgery have never been better. Many new innovations are available, such as aortic root preservation and endovascular stenting procedures. It may be possible to slow the growth of or prevent some aneurysms and aortic dissections, particularly with beta-blockers and, potentially, with losartan (Cozaar) for Marfan syndrome patients.
One of the keys to preventing aortic catastrophes and aortic dissection is to repair aortic aneurysms. The threshold for surgery, however, depends on a surgeon’s experience and results, the underlying pathology, and the aortic size.
We observed that 12.5% of dissections in patients with bicuspid valves and 15% of those in patients with Marfan syndrome were in aortas smaller than 5.0 cm in diameter, that aortic dissection occurred at smaller diameters in shorter patients, and that the risk of dissection increased exponentially with the size of the aorta. Subsequently, we found that a better measure of risk is the maximal aortic cross-sectional area in cm2 divided by the patient’s height in meters; if this ratio exceeds 10, then surgery is recommended.12
Results of surgery are good in experienced hands. In patients who undergo surgical repair of bicuspid aortic valves with or without concurrent repair of the ascending aorta (mostly in patients with an aortic cross-section-to-height ratio > 10), the perioperative mortality rate is about 1.0% for both groups, and at 10 years about 98% of patients are free from re-operation on the aorta and more than 90% are free from re-operation on the aortic valve.8 This is important because these are typically young patients who would do better without biological replacement valves (which are not very durable) or mechanical valves (which necessitate lifelong anticoagulation). Results are also good in surgery of the aortic arch and even better in patients with tricuspid aortic valves.4,6,8
Increasingly, in patients at high risk, we are inserting thoracic, abdominal, and thoracoabdominal stent grafts, with excellent early results. An even newer innovation is to replace the aortic valve in high-risk patients via a transcatheter balloon-expandable valve stent inserted through the groin or left ventricular apex.
These treatment innovations have been big strides, but aortic disease continues to increase. Indeed, our volume of thoracic aortic surgery at Cleveland Clinic increased from 190 procedures in 1999 to 717 in 2006. Early detection—before acute emergency surgery is required, with its concomitant high risk of death—is the key to successful surgical outcome and long-term survival.
- Svensson LG, Labib SB, Eisenhauer AC, Butterly JR. Intimal tear without hematoma: an important variant of aortic dissection that can elude current imaging techniques. Circulation 1999; 99:1331–1336.
- Smith AD, Schoenhagen P. CT imaging for acute aortic syndrome. Cleve Clin J Med 2008; 75:7–24.
- Svensson LG, Nadolny EM, Kimmel WA. Multimodal protocol influence on stroke and neurocognitive deficit prevention after ascending/arch aortic operations. Ann Thorac Surg 2002; 74:2040–2046.
- Svensson LG, Kim KH, Blackstone EH, et al. Elephant trunk procedure: newer indications and uses. Ann Thorac Surg 2004; 78:109–116.
- Greenberg RK, Haddad F, Svensson L, et al. Hybrid approaches to thoracic aortic aneurysms: the role of endovascular elephant trunk completion. Circulation 2005; 112:2619–2626.
- Svensson LG. Sizing for modified David’s reimplantation procedure. Ann Thorac Surg 2003; 76:1751–1753.
- Svensson LG, Blackstone EH, Feng J, et al. Are Marfan syndrome and marfanoid patients distinguishable on long-term follow-up? Ann Thorac Surg 2007; 83:1067–1074.
- Svensson LG, Blackstone EH, Cosgrove DM 3rd. Surgical options in young adults with aortic valve disease. Curr Probl Cardiol 2003; 28:417–480.
- Svensson LG, Rodriguez ER. Aortic organ disease epidemic, and why do balloons pop? Circulation 2005; 112:1082–1084.
- Clouse WD, Hallett JW Jr, Schaff HV, Gayari MM, Ilstrup DM, Melton LJ 3rd. Improved prognosis of thoracic aortic aneurysms: a population-based study. JAMA 1998; 280:1926–1929.
- Olsson C, Thelin S, Ståhle E, Ekbom A, Granath F. Thoracic aortic aneurysm and dissection: increasing prevalence and improved outcomes reported in a nationwide population-based study of more than 14,000 cases from 1987 to 2002. Circulation 2006; 114:2611–2618.
- Svensson LG, Kim KH, Lytle BW, Cosgrove DM. Relationship of aortic cross-sectional area to height ratio and the risk of aortic dissection inpatients with bicuspid aortic valves. J Thorac Cardiovasc Surg 2003;126:892–893.
“The time has come,” the Walrus said,
“To talk of many things:
Of shoes—and ships—and sealing-wax—
Of cabbages—and kings—
And why the sea is boiling hot
And whether pigs have wings.”
—Lewis Carroll, The Walrus and the Carpenter (from Through the Looking-Glass and What Alice Found There, 1872).
Lewis Carroll's poem of 1872 is a useful starting point for identifying issues resulting from confusion over the variously described acute aortic syndromes—and, for oysters, the dangers of listening to walruses.
TALK OF MANY THINGS
In cases of aortic dissection (splitting or separation of the layers of the aortic wall), it is important to establish the type (ie, the location and extent) and class (ie, the structure) of the dissection, because these distinctions determine the treatment.1 Similarly, in cases of painful or leaking degenerative aneurysms, we need to know the location of the aneurysm and whether the presenting pain is from compression of surrounding tissue, particularly of the vertebral bodies, or from leakage.
The location and extent of an aortic dissection can be classified in three ways (see Figure 3 in Smith and Schoenhagen’s excellent review of the use of computed tomography [CT] in acute aortic syndromes in this issue of the Cleveland Clinic Journal of Medicine2):
- The DeBakey system (type I, II, or III)
- The Stanford system (type A or B)
- Distal or proximal to the left subclavian artery.
Of note, the DeBakey system does not include tears in the arch that extend distally without ascending involvement. The original Stanford system included arch tears with distal extension in type B; hence, type B excluded all patients without ascending involvement.
The importance of the extent of dissection is that most patients with Stanford type A or DeBakey type I or II dissections should undergo immediate surgery, as most of them would die without it. Surgery is also indicated for arch tears (non-DeBakey, original Stanford type B).
Because these classifications are somewhat confusing, the simplest approach is to note whether the dissection extends proximal or distal to the left subclavian artery, because proximal dissections need surgery and distal ones are first managed medically.
The classes of dissection also have bearing on treatment.1 These are:
- Class I—classic aortic dissection in the media with two lumens separated by a “flap” or septum
- Class II—intramural hematoma in the aortic wall from dissection in which the intimal tear cannot be imaged (these are nearly always found duringsurgery or autopsy)
- Class III—localized confined intimal tears without extensive undermining of the intima or flap formation. These are often seen with Marfan syndrome and can rupture or cause tamponade, as can any type of proximal dissection. The typical appearance is of a bulging bubble in the aortic wall.
- Class IV—penetrating atherosclerotic ulcers with localized dissections or wall hematomas, often with calcium at the base of a mushroom-shaped area of extraluminal contrast. Of note, the plane of dissection is often between the media and adventitia.
- Class V—iatrogenic or posttraumatic dissection.
All class I to class IV tears of the proximal aorta require surgery, whereas distal class IV and V tears may require either open or endovascular surgical intervention. Surgery is also indicated for patients with distal dissections who have severe narrowing of the true lumen, distal ischemia, uncontrolled pain, severe hypertension, or evidence of leaking, particularly with class IV tears.
In distal dissections that are subacute (2–6 week sold), the Investigation of Stent grafts in Patients With Type B Aortic Dissection (INSTEAD) trial found that inserting a stent prophylactically provided no benefit. Further-more, there is no proof that stenting is beneficial if the aortic dissection is chronic, ie, more than 6 weeks old.1,3–5
WHICH SHOE FITS?
There is no ideal procedure to detect dissection, although the trend is towards CT angiography, as Smith and Schoenhagen report.2 Although some investigators have optimistically estimated CT’s sensitivity and specificity as 100%, cardiovascular surgeons are well aware of both false-positive and false-negative CT studies. Thus, for emergency repairs of proximal dissections, transesophageal echocardiography should be done after intubation and before opening a patient’s chest if time allows. Magnetic resonance imaging, CT, and transesophageal echocardiography may all miss class III tears, but these are frequently evidenced by eccentric “bubbles”or “ballooning.”1
SHIPS
Patients with either acute aortic dissection or severe pain associated with degenerative aneurysms need to be “shipped” promptly to a tertiary medical center after diagnosis, since larger volumes of procedures appear to be associated with better outcomes.
SEALING WAX
Using current surgical methods, the aortic valve can be preserved during aortic dissection repair unless the valve is bicuspid or the patient has Marfan syndrome.1,3,4,6–8
Sealing wax in the form of biological glues, rather than for letters, is a new innovation. A caveat remains, however: we have seen patients who have required reoperation for false aneurysms or infection. Hence, glues should be used with caution.
CABBAGES
A dilemma is whether patients should undergo coronary catheterization (or CT angiography—a separate question) and subsequent coronary artery bypass grafting (CABG), if needed, at the time of aortic dissection repair. The problem is that approximately one-third of patients have coronary artery disease that may require CABG, but the delay for catheterization increases the risk of rupture or tamponade before surgery.
Indeed, 40% of patients with proximal dissections die immediately, and 1% to 3% die in the hour before surgery. The short-term (in-hospital and 30-day) mortality rates range from 3.4% (Cleveland Clinic 2006 data) to 25%, and of the survivors only about 50% area live 5 years after surgery.
Though dismal, the prognosis is improving. In 162 patients with aortic dissection and Marfan syndrome or connective tissue disorders who underwent surgery at Cleveland Clinic in the years 1978–2003, the 5-year survival rate in those with aortic dissection was 75% and the 10-year rate was 55%.7 In those without dissection, the 10-year survival rate was approximately 90% (P < .001).
KINGS
Noted personalities who have had aortic dissection include King George II of England (who died in 1760), Lucille Ball, Conway Twitty, Jan Larson, and most recently John Ritter. None of these famous people survived their aortic dissections. Indeed, dissection and diseases of the aorta or its branches cause between 43,000 and 47,000 deaths annually,9 more than from breast cancer, murders, or motor vehicle accidents. The main reason for these dismal statistics is that the disease is often misdiagnosed at the time of presentation.
BOILING SEA
Careful studies from Olmsted County, Minnesota,10 have shown a tripling of the incidence of aortic disease, particularly in women, even though the rate of deaths from coronary artery disease has been decreasing. Furthermore, Olsson et al11 report that the incidence of aortic dissection in men in Sweden increased to approximately16 per 100,000 per year from 1987 to 2002, a 52% increase. The aging of the population must play a large role, but other factors may exist that are not well understood or defined and require further research.
PIGS HAVE WINGS
Will it be possible to overcome this rising problem? The answer is a definite yes. The results of aortic surgery have never been better. Many new innovations are available, such as aortic root preservation and endovascular stenting procedures. It may be possible to slow the growth of or prevent some aneurysms and aortic dissections, particularly with beta-blockers and, potentially, with losartan (Cozaar) for Marfan syndrome patients.
One of the keys to preventing aortic catastrophes and aortic dissection is to repair aortic aneurysms. The threshold for surgery, however, depends on a surgeon’s experience and results, the underlying pathology, and the aortic size.
We observed that 12.5% of dissections in patients with bicuspid valves and 15% of those in patients with Marfan syndrome were in aortas smaller than 5.0 cm in diameter, that aortic dissection occurred at smaller diameters in shorter patients, and that the risk of dissection increased exponentially with the size of the aorta. Subsequently, we found that a better measure of risk is the maximal aortic cross-sectional area in cm2 divided by the patient’s height in meters; if this ratio exceeds 10, then surgery is recommended.12
Results of surgery are good in experienced hands. In patients who undergo surgical repair of bicuspid aortic valves with or without concurrent repair of the ascending aorta (mostly in patients with an aortic cross-section-to-height ratio > 10), the perioperative mortality rate is about 1.0% for both groups, and at 10 years about 98% of patients are free from re-operation on the aorta and more than 90% are free from re-operation on the aortic valve.8 This is important because these are typically young patients who would do better without biological replacement valves (which are not very durable) or mechanical valves (which necessitate lifelong anticoagulation). Results are also good in surgery of the aortic arch and even better in patients with tricuspid aortic valves.4,6,8
Increasingly, in patients at high risk, we are inserting thoracic, abdominal, and thoracoabdominal stent grafts, with excellent early results. An even newer innovation is to replace the aortic valve in high-risk patients via a transcatheter balloon-expandable valve stent inserted through the groin or left ventricular apex.
These treatment innovations have been big strides, but aortic disease continues to increase. Indeed, our volume of thoracic aortic surgery at Cleveland Clinic increased from 190 procedures in 1999 to 717 in 2006. Early detection—before acute emergency surgery is required, with its concomitant high risk of death—is the key to successful surgical outcome and long-term survival.
“The time has come,” the Walrus said,
“To talk of many things:
Of shoes—and ships—and sealing-wax—
Of cabbages—and kings—
And why the sea is boiling hot
And whether pigs have wings.”
—Lewis Carroll, The Walrus and the Carpenter (from Through the Looking-Glass and What Alice Found There, 1872).
Lewis Carroll's poem of 1872 is a useful starting point for identifying issues resulting from confusion over the variously described acute aortic syndromes—and, for oysters, the dangers of listening to walruses.
TALK OF MANY THINGS
In cases of aortic dissection (splitting or separation of the layers of the aortic wall), it is important to establish the type (ie, the location and extent) and class (ie, the structure) of the dissection, because these distinctions determine the treatment.1 Similarly, in cases of painful or leaking degenerative aneurysms, we need to know the location of the aneurysm and whether the presenting pain is from compression of surrounding tissue, particularly of the vertebral bodies, or from leakage.
The location and extent of an aortic dissection can be classified in three ways (see Figure 3 in Smith and Schoenhagen’s excellent review of the use of computed tomography [CT] in acute aortic syndromes in this issue of the Cleveland Clinic Journal of Medicine2):
- The DeBakey system (type I, II, or III)
- The Stanford system (type A or B)
- Distal or proximal to the left subclavian artery.
Of note, the DeBakey system does not include tears in the arch that extend distally without ascending involvement. The original Stanford system included arch tears with distal extension in type B; hence, type B excluded all patients without ascending involvement.
The importance of the extent of dissection is that most patients with Stanford type A or DeBakey type I or II dissections should undergo immediate surgery, as most of them would die without it. Surgery is also indicated for arch tears (non-DeBakey, original Stanford type B).
Because these classifications are somewhat confusing, the simplest approach is to note whether the dissection extends proximal or distal to the left subclavian artery, because proximal dissections need surgery and distal ones are first managed medically.
The classes of dissection also have bearing on treatment.1 These are:
- Class I—classic aortic dissection in the media with two lumens separated by a “flap” or septum
- Class II—intramural hematoma in the aortic wall from dissection in which the intimal tear cannot be imaged (these are nearly always found duringsurgery or autopsy)
- Class III—localized confined intimal tears without extensive undermining of the intima or flap formation. These are often seen with Marfan syndrome and can rupture or cause tamponade, as can any type of proximal dissection. The typical appearance is of a bulging bubble in the aortic wall.
- Class IV—penetrating atherosclerotic ulcers with localized dissections or wall hematomas, often with calcium at the base of a mushroom-shaped area of extraluminal contrast. Of note, the plane of dissection is often between the media and adventitia.
- Class V—iatrogenic or posttraumatic dissection.
All class I to class IV tears of the proximal aorta require surgery, whereas distal class IV and V tears may require either open or endovascular surgical intervention. Surgery is also indicated for patients with distal dissections who have severe narrowing of the true lumen, distal ischemia, uncontrolled pain, severe hypertension, or evidence of leaking, particularly with class IV tears.
In distal dissections that are subacute (2–6 week sold), the Investigation of Stent grafts in Patients With Type B Aortic Dissection (INSTEAD) trial found that inserting a stent prophylactically provided no benefit. Further-more, there is no proof that stenting is beneficial if the aortic dissection is chronic, ie, more than 6 weeks old.1,3–5
WHICH SHOE FITS?
There is no ideal procedure to detect dissection, although the trend is towards CT angiography, as Smith and Schoenhagen report.2 Although some investigators have optimistically estimated CT’s sensitivity and specificity as 100%, cardiovascular surgeons are well aware of both false-positive and false-negative CT studies. Thus, for emergency repairs of proximal dissections, transesophageal echocardiography should be done after intubation and before opening a patient’s chest if time allows. Magnetic resonance imaging, CT, and transesophageal echocardiography may all miss class III tears, but these are frequently evidenced by eccentric “bubbles”or “ballooning.”1
SHIPS
Patients with either acute aortic dissection or severe pain associated with degenerative aneurysms need to be “shipped” promptly to a tertiary medical center after diagnosis, since larger volumes of procedures appear to be associated with better outcomes.
SEALING WAX
Using current surgical methods, the aortic valve can be preserved during aortic dissection repair unless the valve is bicuspid or the patient has Marfan syndrome.1,3,4,6–8
Sealing wax in the form of biological glues, rather than for letters, is a new innovation. A caveat remains, however: we have seen patients who have required reoperation for false aneurysms or infection. Hence, glues should be used with caution.
CABBAGES
A dilemma is whether patients should undergo coronary catheterization (or CT angiography—a separate question) and subsequent coronary artery bypass grafting (CABG), if needed, at the time of aortic dissection repair. The problem is that approximately one-third of patients have coronary artery disease that may require CABG, but the delay for catheterization increases the risk of rupture or tamponade before surgery.
Indeed, 40% of patients with proximal dissections die immediately, and 1% to 3% die in the hour before surgery. The short-term (in-hospital and 30-day) mortality rates range from 3.4% (Cleveland Clinic 2006 data) to 25%, and of the survivors only about 50% area live 5 years after surgery.
Though dismal, the prognosis is improving. In 162 patients with aortic dissection and Marfan syndrome or connective tissue disorders who underwent surgery at Cleveland Clinic in the years 1978–2003, the 5-year survival rate in those with aortic dissection was 75% and the 10-year rate was 55%.7 In those without dissection, the 10-year survival rate was approximately 90% (P < .001).
KINGS
Noted personalities who have had aortic dissection include King George II of England (who died in 1760), Lucille Ball, Conway Twitty, Jan Larson, and most recently John Ritter. None of these famous people survived their aortic dissections. Indeed, dissection and diseases of the aorta or its branches cause between 43,000 and 47,000 deaths annually,9 more than from breast cancer, murders, or motor vehicle accidents. The main reason for these dismal statistics is that the disease is often misdiagnosed at the time of presentation.
BOILING SEA
Careful studies from Olmsted County, Minnesota,10 have shown a tripling of the incidence of aortic disease, particularly in women, even though the rate of deaths from coronary artery disease has been decreasing. Furthermore, Olsson et al11 report that the incidence of aortic dissection in men in Sweden increased to approximately16 per 100,000 per year from 1987 to 2002, a 52% increase. The aging of the population must play a large role, but other factors may exist that are not well understood or defined and require further research.
PIGS HAVE WINGS
Will it be possible to overcome this rising problem? The answer is a definite yes. The results of aortic surgery have never been better. Many new innovations are available, such as aortic root preservation and endovascular stenting procedures. It may be possible to slow the growth of or prevent some aneurysms and aortic dissections, particularly with beta-blockers and, potentially, with losartan (Cozaar) for Marfan syndrome patients.
One of the keys to preventing aortic catastrophes and aortic dissection is to repair aortic aneurysms. The threshold for surgery, however, depends on a surgeon’s experience and results, the underlying pathology, and the aortic size.
We observed that 12.5% of dissections in patients with bicuspid valves and 15% of those in patients with Marfan syndrome were in aortas smaller than 5.0 cm in diameter, that aortic dissection occurred at smaller diameters in shorter patients, and that the risk of dissection increased exponentially with the size of the aorta. Subsequently, we found that a better measure of risk is the maximal aortic cross-sectional area in cm2 divided by the patient’s height in meters; if this ratio exceeds 10, then surgery is recommended.12
Results of surgery are good in experienced hands. In patients who undergo surgical repair of bicuspid aortic valves with or without concurrent repair of the ascending aorta (mostly in patients with an aortic cross-section-to-height ratio > 10), the perioperative mortality rate is about 1.0% for both groups, and at 10 years about 98% of patients are free from re-operation on the aorta and more than 90% are free from re-operation on the aortic valve.8 This is important because these are typically young patients who would do better without biological replacement valves (which are not very durable) or mechanical valves (which necessitate lifelong anticoagulation). Results are also good in surgery of the aortic arch and even better in patients with tricuspid aortic valves.4,6,8
Increasingly, in patients at high risk, we are inserting thoracic, abdominal, and thoracoabdominal stent grafts, with excellent early results. An even newer innovation is to replace the aortic valve in high-risk patients via a transcatheter balloon-expandable valve stent inserted through the groin or left ventricular apex.
These treatment innovations have been big strides, but aortic disease continues to increase. Indeed, our volume of thoracic aortic surgery at Cleveland Clinic increased from 190 procedures in 1999 to 717 in 2006. Early detection—before acute emergency surgery is required, with its concomitant high risk of death—is the key to successful surgical outcome and long-term survival.
- Svensson LG, Labib SB, Eisenhauer AC, Butterly JR. Intimal tear without hematoma: an important variant of aortic dissection that can elude current imaging techniques. Circulation 1999; 99:1331–1336.
- Smith AD, Schoenhagen P. CT imaging for acute aortic syndrome. Cleve Clin J Med 2008; 75:7–24.
- Svensson LG, Nadolny EM, Kimmel WA. Multimodal protocol influence on stroke and neurocognitive deficit prevention after ascending/arch aortic operations. Ann Thorac Surg 2002; 74:2040–2046.
- Svensson LG, Kim KH, Blackstone EH, et al. Elephant trunk procedure: newer indications and uses. Ann Thorac Surg 2004; 78:109–116.
- Greenberg RK, Haddad F, Svensson L, et al. Hybrid approaches to thoracic aortic aneurysms: the role of endovascular elephant trunk completion. Circulation 2005; 112:2619–2626.
- Svensson LG. Sizing for modified David’s reimplantation procedure. Ann Thorac Surg 2003; 76:1751–1753.
- Svensson LG, Blackstone EH, Feng J, et al. Are Marfan syndrome and marfanoid patients distinguishable on long-term follow-up? Ann Thorac Surg 2007; 83:1067–1074.
- Svensson LG, Blackstone EH, Cosgrove DM 3rd. Surgical options in young adults with aortic valve disease. Curr Probl Cardiol 2003; 28:417–480.
- Svensson LG, Rodriguez ER. Aortic organ disease epidemic, and why do balloons pop? Circulation 2005; 112:1082–1084.
- Clouse WD, Hallett JW Jr, Schaff HV, Gayari MM, Ilstrup DM, Melton LJ 3rd. Improved prognosis of thoracic aortic aneurysms: a population-based study. JAMA 1998; 280:1926–1929.
- Olsson C, Thelin S, Ståhle E, Ekbom A, Granath F. Thoracic aortic aneurysm and dissection: increasing prevalence and improved outcomes reported in a nationwide population-based study of more than 14,000 cases from 1987 to 2002. Circulation 2006; 114:2611–2618.
- Svensson LG, Kim KH, Lytle BW, Cosgrove DM. Relationship of aortic cross-sectional area to height ratio and the risk of aortic dissection inpatients with bicuspid aortic valves. J Thorac Cardiovasc Surg 2003;126:892–893.
- Svensson LG, Labib SB, Eisenhauer AC, Butterly JR. Intimal tear without hematoma: an important variant of aortic dissection that can elude current imaging techniques. Circulation 1999; 99:1331–1336.
- Smith AD, Schoenhagen P. CT imaging for acute aortic syndrome. Cleve Clin J Med 2008; 75:7–24.
- Svensson LG, Nadolny EM, Kimmel WA. Multimodal protocol influence on stroke and neurocognitive deficit prevention after ascending/arch aortic operations. Ann Thorac Surg 2002; 74:2040–2046.
- Svensson LG, Kim KH, Blackstone EH, et al. Elephant trunk procedure: newer indications and uses. Ann Thorac Surg 2004; 78:109–116.
- Greenberg RK, Haddad F, Svensson L, et al. Hybrid approaches to thoracic aortic aneurysms: the role of endovascular elephant trunk completion. Circulation 2005; 112:2619–2626.
- Svensson LG. Sizing for modified David’s reimplantation procedure. Ann Thorac Surg 2003; 76:1751–1753.
- Svensson LG, Blackstone EH, Feng J, et al. Are Marfan syndrome and marfanoid patients distinguishable on long-term follow-up? Ann Thorac Surg 2007; 83:1067–1074.
- Svensson LG, Blackstone EH, Cosgrove DM 3rd. Surgical options in young adults with aortic valve disease. Curr Probl Cardiol 2003; 28:417–480.
- Svensson LG, Rodriguez ER. Aortic organ disease epidemic, and why do balloons pop? Circulation 2005; 112:1082–1084.
- Clouse WD, Hallett JW Jr, Schaff HV, Gayari MM, Ilstrup DM, Melton LJ 3rd. Improved prognosis of thoracic aortic aneurysms: a population-based study. JAMA 1998; 280:1926–1929.
- Olsson C, Thelin S, Ståhle E, Ekbom A, Granath F. Thoracic aortic aneurysm and dissection: increasing prevalence and improved outcomes reported in a nationwide population-based study of more than 14,000 cases from 1987 to 2002. Circulation 2006; 114:2611–2618.
- Svensson LG, Kim KH, Lytle BW, Cosgrove DM. Relationship of aortic cross-sectional area to height ratio and the risk of aortic dissection inpatients with bicuspid aortic valves. J Thorac Cardiovasc Surg 2003;126:892–893.
Proceedings of the 3rd Annual Perioperative Medicine Summit
Supplement Co-Editors and Supplement Co-Directors:
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Co-Directors:
Angela M. Bader, MD, MPH, and Raymond Borkowski, MD
Contents
Forword: New topics, returning features, tools for enduring challenges
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Faculty
Summit Program
IMPACT Consults
Are routine preoperative chest radiographs necessary in asymptomatic patients undergoing noncardiothoracic surgery?
Anitha Rajamanickam, MD, Preethi Patel, MD, and Ali Usmani, MD
Do preoperative nutritional interventions improve outcomes in malnourished patients undergoing elective surgery?
Ramnath Hebbar, MD, and Brian Harte, MD
Do all patients undergoing bariatric surgery need polysomnography to evaluate for obstructive sleep apnea?
Roop Kaw, MD, Vesselin Dimov, MD, and Charles Bae, MD
Can brain natriuretic peptide identify noncardiac surgery patients at high risk for cardiac events?
Ali Usmani, MD, Priyanka Sharma, MD, and Ashish Aneja, MD
What is the significance of an isolated elevated activated partial thromboplastin time in the preoperative setting?
William H. Morris, MD, and Ajay Kumar, MD
Does unrecognized diabetes in the preoperative period worsen postoperative outcomes?
Krista Andersen-Harris, DO, and Christopher Whinney, MD
Should an asymptomatic patient with an abnormal urinalysis (bacteriuria or pyuria) be treated with antibiotics prior to major joint replacement surgery?
Anitha Rajamanickam, MD, Saira Noor, MD, and Ali Usmani, MD
Does a carotid bruit predict cerebrovascular complications following noncardiac surgery in asymptomatic patients?
Robert Mayock, MD
What risks does a history of pulmonary hypertension present for patients undergoing noncardiac surgery?
Roop Kaw, MD, Priyanka Sharma, MD, and Omar A. Minai, MD
Does a systolic murmur heard in the aortic area need to be further evaluated prior to elective surgery?
Thadeo Catacutan, MD, Ali Usmani, MD, and Ashish Aneja, MD
Abstracts
Oral Abstracts
Preoperative electrocardiograms: Patient factors predictive of abnormalities
Darin Correll, David Hepner, Lawrence Tsen, Candace Chang, Angela Bader
Impact of combination medical therapy on mortality in vascular surgery patients
Thomas Barrett, Motomi Mori, Caroline Koudelka
Do large electronic medical record databases permit collection of reliable and valid data for quality improvement purposes?
Ashish Aneja, Eric Hixson, Brian Harte, Vesselin Dimov, Amir Jaffer
Poster Abstracts
Innovations in Perioperative Medicine
Abstract 1: PONV: 'An ounce of prevention is worth a pound of cure'
Catherine Capitula, Shari Duguay
Abstract 2: Optimization of perioperative processes through innovation and technology for the orthopaedic operating room of the future
J.H. James Choi, Jennifer Blueter, Barbara Fahey, James Leonard, Ted Omilanowski, Vincent Riley, Mark Schauer, Timothy Sullivan, Viktor Krebs, Jonathan Schaffer
Abstract 3: A systematic approach to interpreting electrocardiograms by using two mnemonics
Vesselin Dimov, Kalina Uzunova-Dimova, Ajay Kumar, Ashish Aneja
Abstract 4: Improving and standardizing medicine consultation
Benny Gavi, Lisa Shieh, Keith Posley, Shahram Sepehri, Phil Pang
Abstract 5: Medical students' assessment of a required rotation in perioperative medicine and pain
Amir Jaffer, Samuel Irefin, John Tetzlaff, J. Harry Isaacson
Abstract 6: Improving safety for adult surgical patients with obstructive sleep apnea
Karen Watkins
Abstract 7: A multidisciplinary approach to improving the safety of high-risk spine surgery: The complex spine protocol
Peter Kallas, Anjali Desai, Andrew Naidech, Tyler Koski, Steve Ondra, Mary Lou Green
Abstract 8: The nurse practitioner role in evidence-based medication strategies
Patricia Kidik, Kathleen Holbrook
Abstract 9: Use of the motivator/hygiene theory of motivation to guide quality efforts
Ronald Kratz
Abstract 10: A novel care model coordinating inpatient and outpatient perioperative care, utilizing a computerized patient tracking system
Diane Levitan, Dominic Reilly, Christopher Wong, Kara Mitchell, Philip Vedovatti, Nason Hamlin
Abstract 11: The development of an admitting team
Kathleen McGrath, Janet Piatek, Jeanne Lanchester
Abstract 12: Improve communication among caregivers: Eliminating unauthorized abbreviations on hospital medical records
Magdalena G. Smith, Maura Walsh, Laurie Walsh, Marjorie Guglin, Dio Sumaygaysay, Evangelina Sapalasan, Frances Haug, Olivia Voellmicke, Mahin Sanjari, Nancy Cimitile, Mariya Chernyatskaya
Abstract 13: Improve preadmission testing process
Magdalena G. Smith, Tak Tam, Rita Medrozo, Maura Walsh, Laurie Walsh, Marjorie Guglin
Perioperative Clinical Vignettes
Abstract 14: Chronic renal insufficiency: An oft-forgotten component of the revised cardiac risk index
Vesselin Dimov, Ashish Aneja, Kalina Uzunova-Dimova
Abstract 15: When is a stress test indicated in patients with chronic kidney disease evaluated for noncardiac surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Saira Noor
Abstract 16: When to correct hyperkalemia in patients with chronic kidney disease prior to noncardiac surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Ajay Kumar, Anitha Rajamanickam, Mitko Badov
Abstract 17: What is the optimal time frame for performing hemodialysis in patients with end-stage renal disease prior to surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Ajay Kumar
Abstract 18: A recent vascular graft in a patient with end-stage renal disease on hemodialysis and the need for preoperative antibiotic prophylaxis
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Ajay Kumar
Abstract 19: Postoperative risk of acute kidney injury in patients with chronic kidney disease
Vesselin Dimov, Kalina Uzunova-Dimova, Ali Usmani, Ajay Kumar
Abstract 20: Preoperative hypoglycemia in a patient on detemir insulin
Ronad P. Olson, M. Angelyn Bethel, Lillian F. Lien
Abstract 21: Evaluation of Mobitz I atrioventricular block in a preoperative patient
Margaret Pothier
Abstract 22: Perioperative cardiac arrest in a patient with aortic stenosis: Is it preventable?
Zdravka Zafirova, Bobbie Sweitzer
Abstract 23: Antiplatelet therapy interruption and perioperative stent thrombosis: Too much, too early
Zdravka Zafirova, Bobbie Sweitzer
Research in Perioperative Medicine
Abstract 24: Use of an at-home internet-based patient evaluation tool for preoperative assessment
Margaret Pothier, David Hepner, Darrin Correll, Thomas Ho, Alina Lazar, Angela Bader
Abstract 25: The utility of a preoperative clinic questionnaire to predict postoperative delirium risk
David Hepner, Darin Correll, Thomas Ho, Juergen Bludau, Jhoanna Santos, Angela Bader
Abstract 26: A drug by any other name: Preoperative insulin regimens
Carlee Clark, Vivek Moitra, Bobbie Jean Sweitzer
Abstract 27: Preoperative cardiovascular risk factor assessment in morbidly obese patients with an abnormal electrocardiogram
Girish Mood, Roomana Akhtar, Rajagopal Reddy Edula, Gunjana Bhandari, Vishal Gupta, Michael Koch
Abstract 28: Cardiac testing prior to nonvascular surgery: The results from a newly formed preoperative clinic
Sheela Pai, Giang Tran, Alvin Blaustein, Prasad Atluri, Salwa Shenaq
Abstract 29: Which is better—half-dose or no insulin on day of surgery?
Kirk Smith, Vivek Moitra, Melinda Drum, Bobbie Jean Sweitzer
Index of Authors
Supplement Co-Editors and Supplement Co-Directors:
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Co-Directors:
Angela M. Bader, MD, MPH, and Raymond Borkowski, MD
Contents
Forword: New topics, returning features, tools for enduring challenges
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Faculty
Summit Program
IMPACT Consults
Are routine preoperative chest radiographs necessary in asymptomatic patients undergoing noncardiothoracic surgery?
Anitha Rajamanickam, MD, Preethi Patel, MD, and Ali Usmani, MD
Do preoperative nutritional interventions improve outcomes in malnourished patients undergoing elective surgery?
Ramnath Hebbar, MD, and Brian Harte, MD
Do all patients undergoing bariatric surgery need polysomnography to evaluate for obstructive sleep apnea?
Roop Kaw, MD, Vesselin Dimov, MD, and Charles Bae, MD
Can brain natriuretic peptide identify noncardiac surgery patients at high risk for cardiac events?
Ali Usmani, MD, Priyanka Sharma, MD, and Ashish Aneja, MD
What is the significance of an isolated elevated activated partial thromboplastin time in the preoperative setting?
William H. Morris, MD, and Ajay Kumar, MD
Does unrecognized diabetes in the preoperative period worsen postoperative outcomes?
Krista Andersen-Harris, DO, and Christopher Whinney, MD
Should an asymptomatic patient with an abnormal urinalysis (bacteriuria or pyuria) be treated with antibiotics prior to major joint replacement surgery?
Anitha Rajamanickam, MD, Saira Noor, MD, and Ali Usmani, MD
Does a carotid bruit predict cerebrovascular complications following noncardiac surgery in asymptomatic patients?
Robert Mayock, MD
What risks does a history of pulmonary hypertension present for patients undergoing noncardiac surgery?
Roop Kaw, MD, Priyanka Sharma, MD, and Omar A. Minai, MD
Does a systolic murmur heard in the aortic area need to be further evaluated prior to elective surgery?
Thadeo Catacutan, MD, Ali Usmani, MD, and Ashish Aneja, MD
Abstracts
Oral Abstracts
Preoperative electrocardiograms: Patient factors predictive of abnormalities
Darin Correll, David Hepner, Lawrence Tsen, Candace Chang, Angela Bader
Impact of combination medical therapy on mortality in vascular surgery patients
Thomas Barrett, Motomi Mori, Caroline Koudelka
Do large electronic medical record databases permit collection of reliable and valid data for quality improvement purposes?
Ashish Aneja, Eric Hixson, Brian Harte, Vesselin Dimov, Amir Jaffer
Poster Abstracts
Innovations in Perioperative Medicine
Abstract 1: PONV: 'An ounce of prevention is worth a pound of cure'
Catherine Capitula, Shari Duguay
Abstract 2: Optimization of perioperative processes through innovation and technology for the orthopaedic operating room of the future
J.H. James Choi, Jennifer Blueter, Barbara Fahey, James Leonard, Ted Omilanowski, Vincent Riley, Mark Schauer, Timothy Sullivan, Viktor Krebs, Jonathan Schaffer
Abstract 3: A systematic approach to interpreting electrocardiograms by using two mnemonics
Vesselin Dimov, Kalina Uzunova-Dimova, Ajay Kumar, Ashish Aneja
Abstract 4: Improving and standardizing medicine consultation
Benny Gavi, Lisa Shieh, Keith Posley, Shahram Sepehri, Phil Pang
Abstract 5: Medical students' assessment of a required rotation in perioperative medicine and pain
Amir Jaffer, Samuel Irefin, John Tetzlaff, J. Harry Isaacson
Abstract 6: Improving safety for adult surgical patients with obstructive sleep apnea
Karen Watkins
Abstract 7: A multidisciplinary approach to improving the safety of high-risk spine surgery: The complex spine protocol
Peter Kallas, Anjali Desai, Andrew Naidech, Tyler Koski, Steve Ondra, Mary Lou Green
Abstract 8: The nurse practitioner role in evidence-based medication strategies
Patricia Kidik, Kathleen Holbrook
Abstract 9: Use of the motivator/hygiene theory of motivation to guide quality efforts
Ronald Kratz
Abstract 10: A novel care model coordinating inpatient and outpatient perioperative care, utilizing a computerized patient tracking system
Diane Levitan, Dominic Reilly, Christopher Wong, Kara Mitchell, Philip Vedovatti, Nason Hamlin
Abstract 11: The development of an admitting team
Kathleen McGrath, Janet Piatek, Jeanne Lanchester
Abstract 12: Improve communication among caregivers: Eliminating unauthorized abbreviations on hospital medical records
Magdalena G. Smith, Maura Walsh, Laurie Walsh, Marjorie Guglin, Dio Sumaygaysay, Evangelina Sapalasan, Frances Haug, Olivia Voellmicke, Mahin Sanjari, Nancy Cimitile, Mariya Chernyatskaya
Abstract 13: Improve preadmission testing process
Magdalena G. Smith, Tak Tam, Rita Medrozo, Maura Walsh, Laurie Walsh, Marjorie Guglin
Perioperative Clinical Vignettes
Abstract 14: Chronic renal insufficiency: An oft-forgotten component of the revised cardiac risk index
Vesselin Dimov, Ashish Aneja, Kalina Uzunova-Dimova
Abstract 15: When is a stress test indicated in patients with chronic kidney disease evaluated for noncardiac surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Saira Noor
Abstract 16: When to correct hyperkalemia in patients with chronic kidney disease prior to noncardiac surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Ajay Kumar, Anitha Rajamanickam, Mitko Badov
Abstract 17: What is the optimal time frame for performing hemodialysis in patients with end-stage renal disease prior to surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Ajay Kumar
Abstract 18: A recent vascular graft in a patient with end-stage renal disease on hemodialysis and the need for preoperative antibiotic prophylaxis
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Ajay Kumar
Abstract 19: Postoperative risk of acute kidney injury in patients with chronic kidney disease
Vesselin Dimov, Kalina Uzunova-Dimova, Ali Usmani, Ajay Kumar
Abstract 20: Preoperative hypoglycemia in a patient on detemir insulin
Ronad P. Olson, M. Angelyn Bethel, Lillian F. Lien
Abstract 21: Evaluation of Mobitz I atrioventricular block in a preoperative patient
Margaret Pothier
Abstract 22: Perioperative cardiac arrest in a patient with aortic stenosis: Is it preventable?
Zdravka Zafirova, Bobbie Sweitzer
Abstract 23: Antiplatelet therapy interruption and perioperative stent thrombosis: Too much, too early
Zdravka Zafirova, Bobbie Sweitzer
Research in Perioperative Medicine
Abstract 24: Use of an at-home internet-based patient evaluation tool for preoperative assessment
Margaret Pothier, David Hepner, Darrin Correll, Thomas Ho, Alina Lazar, Angela Bader
Abstract 25: The utility of a preoperative clinic questionnaire to predict postoperative delirium risk
David Hepner, Darin Correll, Thomas Ho, Juergen Bludau, Jhoanna Santos, Angela Bader
Abstract 26: A drug by any other name: Preoperative insulin regimens
Carlee Clark, Vivek Moitra, Bobbie Jean Sweitzer
Abstract 27: Preoperative cardiovascular risk factor assessment in morbidly obese patients with an abnormal electrocardiogram
Girish Mood, Roomana Akhtar, Rajagopal Reddy Edula, Gunjana Bhandari, Vishal Gupta, Michael Koch
Abstract 28: Cardiac testing prior to nonvascular surgery: The results from a newly formed preoperative clinic
Sheela Pai, Giang Tran, Alvin Blaustein, Prasad Atluri, Salwa Shenaq
Abstract 29: Which is better—half-dose or no insulin on day of surgery?
Kirk Smith, Vivek Moitra, Melinda Drum, Bobbie Jean Sweitzer
Index of Authors
Supplement Co-Editors and Supplement Co-Directors:
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Co-Directors:
Angela M. Bader, MD, MPH, and Raymond Borkowski, MD
Contents
Forword: New topics, returning features, tools for enduring challenges
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Faculty
Summit Program
IMPACT Consults
Are routine preoperative chest radiographs necessary in asymptomatic patients undergoing noncardiothoracic surgery?
Anitha Rajamanickam, MD, Preethi Patel, MD, and Ali Usmani, MD
Do preoperative nutritional interventions improve outcomes in malnourished patients undergoing elective surgery?
Ramnath Hebbar, MD, and Brian Harte, MD
Do all patients undergoing bariatric surgery need polysomnography to evaluate for obstructive sleep apnea?
Roop Kaw, MD, Vesselin Dimov, MD, and Charles Bae, MD
Can brain natriuretic peptide identify noncardiac surgery patients at high risk for cardiac events?
Ali Usmani, MD, Priyanka Sharma, MD, and Ashish Aneja, MD
What is the significance of an isolated elevated activated partial thromboplastin time in the preoperative setting?
William H. Morris, MD, and Ajay Kumar, MD
Does unrecognized diabetes in the preoperative period worsen postoperative outcomes?
Krista Andersen-Harris, DO, and Christopher Whinney, MD
Should an asymptomatic patient with an abnormal urinalysis (bacteriuria or pyuria) be treated with antibiotics prior to major joint replacement surgery?
Anitha Rajamanickam, MD, Saira Noor, MD, and Ali Usmani, MD
Does a carotid bruit predict cerebrovascular complications following noncardiac surgery in asymptomatic patients?
Robert Mayock, MD
What risks does a history of pulmonary hypertension present for patients undergoing noncardiac surgery?
Roop Kaw, MD, Priyanka Sharma, MD, and Omar A. Minai, MD
Does a systolic murmur heard in the aortic area need to be further evaluated prior to elective surgery?
Thadeo Catacutan, MD, Ali Usmani, MD, and Ashish Aneja, MD
Abstracts
Oral Abstracts
Preoperative electrocardiograms: Patient factors predictive of abnormalities
Darin Correll, David Hepner, Lawrence Tsen, Candace Chang, Angela Bader
Impact of combination medical therapy on mortality in vascular surgery patients
Thomas Barrett, Motomi Mori, Caroline Koudelka
Do large electronic medical record databases permit collection of reliable and valid data for quality improvement purposes?
Ashish Aneja, Eric Hixson, Brian Harte, Vesselin Dimov, Amir Jaffer
Poster Abstracts
Innovations in Perioperative Medicine
Abstract 1: PONV: 'An ounce of prevention is worth a pound of cure'
Catherine Capitula, Shari Duguay
Abstract 2: Optimization of perioperative processes through innovation and technology for the orthopaedic operating room of the future
J.H. James Choi, Jennifer Blueter, Barbara Fahey, James Leonard, Ted Omilanowski, Vincent Riley, Mark Schauer, Timothy Sullivan, Viktor Krebs, Jonathan Schaffer
Abstract 3: A systematic approach to interpreting electrocardiograms by using two mnemonics
Vesselin Dimov, Kalina Uzunova-Dimova, Ajay Kumar, Ashish Aneja
Abstract 4: Improving and standardizing medicine consultation
Benny Gavi, Lisa Shieh, Keith Posley, Shahram Sepehri, Phil Pang
Abstract 5: Medical students' assessment of a required rotation in perioperative medicine and pain
Amir Jaffer, Samuel Irefin, John Tetzlaff, J. Harry Isaacson
Abstract 6: Improving safety for adult surgical patients with obstructive sleep apnea
Karen Watkins
Abstract 7: A multidisciplinary approach to improving the safety of high-risk spine surgery: The complex spine protocol
Peter Kallas, Anjali Desai, Andrew Naidech, Tyler Koski, Steve Ondra, Mary Lou Green
Abstract 8: The nurse practitioner role in evidence-based medication strategies
Patricia Kidik, Kathleen Holbrook
Abstract 9: Use of the motivator/hygiene theory of motivation to guide quality efforts
Ronald Kratz
Abstract 10: A novel care model coordinating inpatient and outpatient perioperative care, utilizing a computerized patient tracking system
Diane Levitan, Dominic Reilly, Christopher Wong, Kara Mitchell, Philip Vedovatti, Nason Hamlin
Abstract 11: The development of an admitting team
Kathleen McGrath, Janet Piatek, Jeanne Lanchester
Abstract 12: Improve communication among caregivers: Eliminating unauthorized abbreviations on hospital medical records
Magdalena G. Smith, Maura Walsh, Laurie Walsh, Marjorie Guglin, Dio Sumaygaysay, Evangelina Sapalasan, Frances Haug, Olivia Voellmicke, Mahin Sanjari, Nancy Cimitile, Mariya Chernyatskaya
Abstract 13: Improve preadmission testing process
Magdalena G. Smith, Tak Tam, Rita Medrozo, Maura Walsh, Laurie Walsh, Marjorie Guglin
Perioperative Clinical Vignettes
Abstract 14: Chronic renal insufficiency: An oft-forgotten component of the revised cardiac risk index
Vesselin Dimov, Ashish Aneja, Kalina Uzunova-Dimova
Abstract 15: When is a stress test indicated in patients with chronic kidney disease evaluated for noncardiac surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Saira Noor
Abstract 16: When to correct hyperkalemia in patients with chronic kidney disease prior to noncardiac surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Ajay Kumar, Anitha Rajamanickam, Mitko Badov
Abstract 17: What is the optimal time frame for performing hemodialysis in patients with end-stage renal disease prior to surgery?
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Ajay Kumar
Abstract 18: A recent vascular graft in a patient with end-stage renal disease on hemodialysis and the need for preoperative antibiotic prophylaxis
Vesselin Dimov, Kalina Uzunova-Dimova, Mitko Badov, Ajay Kumar
Abstract 19: Postoperative risk of acute kidney injury in patients with chronic kidney disease
Vesselin Dimov, Kalina Uzunova-Dimova, Ali Usmani, Ajay Kumar
Abstract 20: Preoperative hypoglycemia in a patient on detemir insulin
Ronad P. Olson, M. Angelyn Bethel, Lillian F. Lien
Abstract 21: Evaluation of Mobitz I atrioventricular block in a preoperative patient
Margaret Pothier
Abstract 22: Perioperative cardiac arrest in a patient with aortic stenosis: Is it preventable?
Zdravka Zafirova, Bobbie Sweitzer
Abstract 23: Antiplatelet therapy interruption and perioperative stent thrombosis: Too much, too early
Zdravka Zafirova, Bobbie Sweitzer
Research in Perioperative Medicine
Abstract 24: Use of an at-home internet-based patient evaluation tool for preoperative assessment
Margaret Pothier, David Hepner, Darrin Correll, Thomas Ho, Alina Lazar, Angela Bader
Abstract 25: The utility of a preoperative clinic questionnaire to predict postoperative delirium risk
David Hepner, Darin Correll, Thomas Ho, Juergen Bludau, Jhoanna Santos, Angela Bader
Abstract 26: A drug by any other name: Preoperative insulin regimens
Carlee Clark, Vivek Moitra, Bobbie Jean Sweitzer
Abstract 27: Preoperative cardiovascular risk factor assessment in morbidly obese patients with an abnormal electrocardiogram
Girish Mood, Roomana Akhtar, Rajagopal Reddy Edula, Gunjana Bhandari, Vishal Gupta, Michael Koch
Abstract 28: Cardiac testing prior to nonvascular surgery: The results from a newly formed preoperative clinic
Sheela Pai, Giang Tran, Alvin Blaustein, Prasad Atluri, Salwa Shenaq
Abstract 29: Which is better—half-dose or no insulin on day of surgery?
Kirk Smith, Vivek Moitra, Melinda Drum, Bobbie Jean Sweitzer
Index of Authors
Infections in Hospitalized Patients
Supplement Editors:
Susan J. Rehm, MD, and Alpesh N. Amin, MD, MBA
Contents
Infections in hospitalized patients: What is happening and who can help?
Alpesh N. Amin, MD, MBA, and Susan J. Rehm, MD
Impact of community-acquired methicillin-resistant Staphylococcus aureus in the hospital setting
Thomas M. File, Jr., MD
Emerging issues in the management of infections caused by multidrug-resistant gram-negative bacteria
Louis B. Rice, MD
Complicated skin and soft-tissue infections: Diagnostic approach and empiric treatment options
James I. Merlino, MD, and Mark A. Malangoni, MD
Empiric treatment options in the management of complicated intra-abdominal infections
John A. Weigelt, MD
Antibacterial treatment strategies in hospitalized patients: What role for pharmacoeconomics?
Morton P. Goldman, PharmD, BCPS, and Radhika Nair, PhD
Supplement Editors:
Susan J. Rehm, MD, and Alpesh N. Amin, MD, MBA
Contents
Infections in hospitalized patients: What is happening and who can help?
Alpesh N. Amin, MD, MBA, and Susan J. Rehm, MD
Impact of community-acquired methicillin-resistant Staphylococcus aureus in the hospital setting
Thomas M. File, Jr., MD
Emerging issues in the management of infections caused by multidrug-resistant gram-negative bacteria
Louis B. Rice, MD
Complicated skin and soft-tissue infections: Diagnostic approach and empiric treatment options
James I. Merlino, MD, and Mark A. Malangoni, MD
Empiric treatment options in the management of complicated intra-abdominal infections
John A. Weigelt, MD
Antibacterial treatment strategies in hospitalized patients: What role for pharmacoeconomics?
Morton P. Goldman, PharmD, BCPS, and Radhika Nair, PhD
Supplement Editors:
Susan J. Rehm, MD, and Alpesh N. Amin, MD, MBA
Contents
Infections in hospitalized patients: What is happening and who can help?
Alpesh N. Amin, MD, MBA, and Susan J. Rehm, MD
Impact of community-acquired methicillin-resistant Staphylococcus aureus in the hospital setting
Thomas M. File, Jr., MD
Emerging issues in the management of infections caused by multidrug-resistant gram-negative bacteria
Louis B. Rice, MD
Complicated skin and soft-tissue infections: Diagnostic approach and empiric treatment options
James I. Merlino, MD, and Mark A. Malangoni, MD
Empiric treatment options in the management of complicated intra-abdominal infections
John A. Weigelt, MD
Antibacterial treatment strategies in hospitalized patients: What role for pharmacoeconomics?
Morton P. Goldman, PharmD, BCPS, and Radhika Nair, PhD
Hypotension in the intensive care unit
Risks and benefits of bariatric surgery: Current evidence
When can patients with acute deep vein thrombosis be allowed to get up and walk?
Proceedings of the 2nd Annual Perioperative Medicine Summit
Supplement Co-Editors and Summit Co-Directors:
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Co-Directors:
Angela M. Bader, MD, and Raymond Borkowski, MD
Contents
IMPACT Consults
Does elevated blood pressure at the time of surgery increase perioperative cardiac risk?
Collin Kroen, MD
When is it appropriate to stop antiplatelet therapy in a patient with a drug-eluting stent prior to noncardiac surgery?
Anitha Rajamanickam, MD; Vaishali Singh, MD, MPH, MBA; and Ashish Aneja, MD
Should statins be discontinued preoperatively?
Paul J. Grant, MD, and Navin Kedia, DO
What is the appropriate means of perioperative risk assessment for patients with cirrhosis?
Brian Harte, MD
Who is at risk for developing acute renal failure after surgery?
Vesselin Dimov, MD; Ali Usmani, MD; Saira Noor, MD; and Ajay Kumar, MD
Why treat anemia in the preoperative period of joint replacement surgery with erythropoietin?
Ajay Kumar, MD, and Vesselin Dimov, MD
Obstructive sleep apnea: What to do in the surgical patient?
Roop Kaw, MD, and Joseph Golish, MD
What is the optimal venous thromboembolism prophylaxis for patients undergoing bariatric surgery?
David V. Gugliotti, MD
Do hip fractures need to be repaired withing 24 hours of injury?
Christopher M. Whinney, MD
Is postoperative atrial fibrillation in patients undergoing noncardiothoracic surgery an important problem?
Ashish Aneja, MD, and Wassim H. Fares, MD
How can postoperative ileus be prevented and treated?
Vaishali Singh, MD, MPH, MBA
Abstracts
Oral Abstracts
Is discontinuation of antiplatelet therapy after 6 months safe in patients with drug-eluting stents undergoing noncardiac surgery?
Mihir Bakhru, Wael Saber, Daniel Brotman, Deepak Bhatt, Ashish Aneja, Katherina Tillan-Martinez, and Amir Jaffer
Initiating a preoperative cardiac risk assessment quality improvement program: The hurdles to changing traditional paradigms
Eric Hixson, Karl McCleary, Vikram Kashyap, Vaishali Singh, Brian Harte, Ashish Aneja, Brian Parker, Raymond Borkowski, Walter Maurer, Venkatesh Krishnamurthi, Sue Vitagliano, Jacqueline Matthews, Linda Vopat, Michael Henderson, and Amir Jaffer
Impact of a preoperative medical clinic on operating room cancellation rates in orthopedic surgery
Peter Kallas, Anjali Desai, and Jeanette Bauer
Poster Abstracts
Innovations in Perioperative Medicine
Abstract 1: Best safety practices to prevent postoperative myocardial infarction
Abstract 2: Blog web site as a new educational and promotional medium in perioperative medicine
Abstract 3: Development of a validated questionnaire: The satisfaction with general anesthesia scale
Abstract 4: Perioperative medicine and pain: A required advanced core clerkship for third-year medical students
Abstract 5: Optimal administration of perioperative antibiotics using system redesign
Abstract 6: Blood conservation protocol with erythropoietin in the preoperative period of joint replacement surgery
Abstract 7: Evolution of the nurse practitioner (NP) role in the Center for Preoperative Evaluation (CPE) at Brigham and Women's Hospital
Abstract 8: Development and implementation of a web site for the Center for Preoperative Evaluation (CPE)
Abstract 9: Patient education tool for the preoperative process and the role of the medical consultant
Abstract 10: The internal medicine perioperative assessment center: An innovation in the perioperative management of medical comorbidities at a comprehensive cancer center
Abstract 11: PAC collaborative practice model
Abstract 12: Development and implementation of beta-blocker recommendation
Abstract 13: Development of pre-procedure consult services
Perioperative Clinical Vignettes
Abstract 14: Isolated left bundle branch block in a patient undergoing elective noncardiac surgery
Abstract 15: Avoiding delirium
Abstract 16: Cardiac sarcoma—the role of multimodality cardiovascular imaging
Abstract 17: Asymptomatic bacteriuria before nonprosthetic joint surgery
Abstract 18: Negative T waves on the preoperative electrocardiogram—a cause for worry?
Abstract 19: Preoperative hypokalemia
Abstract 20: Preoperative evaluation can aid in the diagnosis of CAD and risk assessment and management
Research in Perioperative Medicine
Abstract 21: Needs analysis for the development of a preoperative clinic protocol for perioperative beta-blockade
Abstract 22: Improving efficiency in a preoperative clinic
Abstract 23: Formalized preoperative assessment for noncardiac surgery at a large tertiary care medical center leads to higher rates of perioperative beta-blocker use
Abstract 24: Insulin errors in hospitalized patients
Abstract 25: A survey of perioperative beta-blockade at a comprehensive cancer center
Abstract 26: Risk factors for long-term mortality among heart failure patients after elective major noncardiac surgery
Index of Authors and Presenters
Supplement Co-Editors and Summit Co-Directors:
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Co-Directors:
Angela M. Bader, MD, and Raymond Borkowski, MD
Contents
IMPACT Consults
Does elevated blood pressure at the time of surgery increase perioperative cardiac risk?
Collin Kroen, MD
When is it appropriate to stop antiplatelet therapy in a patient with a drug-eluting stent prior to noncardiac surgery?
Anitha Rajamanickam, MD; Vaishali Singh, MD, MPH, MBA; and Ashish Aneja, MD
Should statins be discontinued preoperatively?
Paul J. Grant, MD, and Navin Kedia, DO
What is the appropriate means of perioperative risk assessment for patients with cirrhosis?
Brian Harte, MD
Who is at risk for developing acute renal failure after surgery?
Vesselin Dimov, MD; Ali Usmani, MD; Saira Noor, MD; and Ajay Kumar, MD
Why treat anemia in the preoperative period of joint replacement surgery with erythropoietin?
Ajay Kumar, MD, and Vesselin Dimov, MD
Obstructive sleep apnea: What to do in the surgical patient?
Roop Kaw, MD, and Joseph Golish, MD
What is the optimal venous thromboembolism prophylaxis for patients undergoing bariatric surgery?
David V. Gugliotti, MD
Do hip fractures need to be repaired withing 24 hours of injury?
Christopher M. Whinney, MD
Is postoperative atrial fibrillation in patients undergoing noncardiothoracic surgery an important problem?
Ashish Aneja, MD, and Wassim H. Fares, MD
How can postoperative ileus be prevented and treated?
Vaishali Singh, MD, MPH, MBA
Abstracts
Oral Abstracts
Is discontinuation of antiplatelet therapy after 6 months safe in patients with drug-eluting stents undergoing noncardiac surgery?
Mihir Bakhru, Wael Saber, Daniel Brotman, Deepak Bhatt, Ashish Aneja, Katherina Tillan-Martinez, and Amir Jaffer
Initiating a preoperative cardiac risk assessment quality improvement program: The hurdles to changing traditional paradigms
Eric Hixson, Karl McCleary, Vikram Kashyap, Vaishali Singh, Brian Harte, Ashish Aneja, Brian Parker, Raymond Borkowski, Walter Maurer, Venkatesh Krishnamurthi, Sue Vitagliano, Jacqueline Matthews, Linda Vopat, Michael Henderson, and Amir Jaffer
Impact of a preoperative medical clinic on operating room cancellation rates in orthopedic surgery
Peter Kallas, Anjali Desai, and Jeanette Bauer
Poster Abstracts
Innovations in Perioperative Medicine
Abstract 1: Best safety practices to prevent postoperative myocardial infarction
Abstract 2: Blog web site as a new educational and promotional medium in perioperative medicine
Abstract 3: Development of a validated questionnaire: The satisfaction with general anesthesia scale
Abstract 4: Perioperative medicine and pain: A required advanced core clerkship for third-year medical students
Abstract 5: Optimal administration of perioperative antibiotics using system redesign
Abstract 6: Blood conservation protocol with erythropoietin in the preoperative period of joint replacement surgery
Abstract 7: Evolution of the nurse practitioner (NP) role in the Center for Preoperative Evaluation (CPE) at Brigham and Women's Hospital
Abstract 8: Development and implementation of a web site for the Center for Preoperative Evaluation (CPE)
Abstract 9: Patient education tool for the preoperative process and the role of the medical consultant
Abstract 10: The internal medicine perioperative assessment center: An innovation in the perioperative management of medical comorbidities at a comprehensive cancer center
Abstract 11: PAC collaborative practice model
Abstract 12: Development and implementation of beta-blocker recommendation
Abstract 13: Development of pre-procedure consult services
Perioperative Clinical Vignettes
Abstract 14: Isolated left bundle branch block in a patient undergoing elective noncardiac surgery
Abstract 15: Avoiding delirium
Abstract 16: Cardiac sarcoma—the role of multimodality cardiovascular imaging
Abstract 17: Asymptomatic bacteriuria before nonprosthetic joint surgery
Abstract 18: Negative T waves on the preoperative electrocardiogram—a cause for worry?
Abstract 19: Preoperative hypokalemia
Abstract 20: Preoperative evaluation can aid in the diagnosis of CAD and risk assessment and management
Research in Perioperative Medicine
Abstract 21: Needs analysis for the development of a preoperative clinic protocol for perioperative beta-blockade
Abstract 22: Improving efficiency in a preoperative clinic
Abstract 23: Formalized preoperative assessment for noncardiac surgery at a large tertiary care medical center leads to higher rates of perioperative beta-blocker use
Abstract 24: Insulin errors in hospitalized patients
Abstract 25: A survey of perioperative beta-blockade at a comprehensive cancer center
Abstract 26: Risk factors for long-term mortality among heart failure patients after elective major noncardiac surgery
Index of Authors and Presenters
Supplement Co-Editors and Summit Co-Directors:
Amir K. Jaffer, MD, and Franklin A. Michota, Jr., MD
Summit Co-Directors:
Angela M. Bader, MD, and Raymond Borkowski, MD
Contents
IMPACT Consults
Does elevated blood pressure at the time of surgery increase perioperative cardiac risk?
Collin Kroen, MD
When is it appropriate to stop antiplatelet therapy in a patient with a drug-eluting stent prior to noncardiac surgery?
Anitha Rajamanickam, MD; Vaishali Singh, MD, MPH, MBA; and Ashish Aneja, MD
Should statins be discontinued preoperatively?
Paul J. Grant, MD, and Navin Kedia, DO
What is the appropriate means of perioperative risk assessment for patients with cirrhosis?
Brian Harte, MD
Who is at risk for developing acute renal failure after surgery?
Vesselin Dimov, MD; Ali Usmani, MD; Saira Noor, MD; and Ajay Kumar, MD
Why treat anemia in the preoperative period of joint replacement surgery with erythropoietin?
Ajay Kumar, MD, and Vesselin Dimov, MD
Obstructive sleep apnea: What to do in the surgical patient?
Roop Kaw, MD, and Joseph Golish, MD
What is the optimal venous thromboembolism prophylaxis for patients undergoing bariatric surgery?
David V. Gugliotti, MD
Do hip fractures need to be repaired withing 24 hours of injury?
Christopher M. Whinney, MD
Is postoperative atrial fibrillation in patients undergoing noncardiothoracic surgery an important problem?
Ashish Aneja, MD, and Wassim H. Fares, MD
How can postoperative ileus be prevented and treated?
Vaishali Singh, MD, MPH, MBA
Abstracts
Oral Abstracts
Is discontinuation of antiplatelet therapy after 6 months safe in patients with drug-eluting stents undergoing noncardiac surgery?
Mihir Bakhru, Wael Saber, Daniel Brotman, Deepak Bhatt, Ashish Aneja, Katherina Tillan-Martinez, and Amir Jaffer
Initiating a preoperative cardiac risk assessment quality improvement program: The hurdles to changing traditional paradigms
Eric Hixson, Karl McCleary, Vikram Kashyap, Vaishali Singh, Brian Harte, Ashish Aneja, Brian Parker, Raymond Borkowski, Walter Maurer, Venkatesh Krishnamurthi, Sue Vitagliano, Jacqueline Matthews, Linda Vopat, Michael Henderson, and Amir Jaffer
Impact of a preoperative medical clinic on operating room cancellation rates in orthopedic surgery
Peter Kallas, Anjali Desai, and Jeanette Bauer
Poster Abstracts
Innovations in Perioperative Medicine
Abstract 1: Best safety practices to prevent postoperative myocardial infarction
Abstract 2: Blog web site as a new educational and promotional medium in perioperative medicine
Abstract 3: Development of a validated questionnaire: The satisfaction with general anesthesia scale
Abstract 4: Perioperative medicine and pain: A required advanced core clerkship for third-year medical students
Abstract 5: Optimal administration of perioperative antibiotics using system redesign
Abstract 6: Blood conservation protocol with erythropoietin in the preoperative period of joint replacement surgery
Abstract 7: Evolution of the nurse practitioner (NP) role in the Center for Preoperative Evaluation (CPE) at Brigham and Women's Hospital
Abstract 8: Development and implementation of a web site for the Center for Preoperative Evaluation (CPE)
Abstract 9: Patient education tool for the preoperative process and the role of the medical consultant
Abstract 10: The internal medicine perioperative assessment center: An innovation in the perioperative management of medical comorbidities at a comprehensive cancer center
Abstract 11: PAC collaborative practice model
Abstract 12: Development and implementation of beta-blocker recommendation
Abstract 13: Development of pre-procedure consult services
Perioperative Clinical Vignettes
Abstract 14: Isolated left bundle branch block in a patient undergoing elective noncardiac surgery
Abstract 15: Avoiding delirium
Abstract 16: Cardiac sarcoma—the role of multimodality cardiovascular imaging
Abstract 17: Asymptomatic bacteriuria before nonprosthetic joint surgery
Abstract 18: Negative T waves on the preoperative electrocardiogram—a cause for worry?
Abstract 19: Preoperative hypokalemia
Abstract 20: Preoperative evaluation can aid in the diagnosis of CAD and risk assessment and management
Research in Perioperative Medicine
Abstract 21: Needs analysis for the development of a preoperative clinic protocol for perioperative beta-blockade
Abstract 22: Improving efficiency in a preoperative clinic
Abstract 23: Formalized preoperative assessment for noncardiac surgery at a large tertiary care medical center leads to higher rates of perioperative beta-blocker use
Abstract 24: Insulin errors in hospitalized patients
Abstract 25: A survey of perioperative beta-blockade at a comprehensive cancer center
Abstract 26: Risk factors for long-term mortality among heart failure patients after elective major noncardiac surgery
Index of Authors and Presenters