User login
Bacterial contamination behind most cosmetics recalls
Most of the 313 cosmetic and personal care products recalled from 2002 to 2016 had problems with bacterial contamination, according to data obtained from the Food and Drug Administration.
, said Timothy M. Janetos, MD, and his associates at Northwestern University in Chicago. Bacterial contamination was by far the most common reason – 76% of the recalls over that period (11 class I, 217 class II, and 9 class II) – with unapproved ingredients and labeling problems well behind at 6%.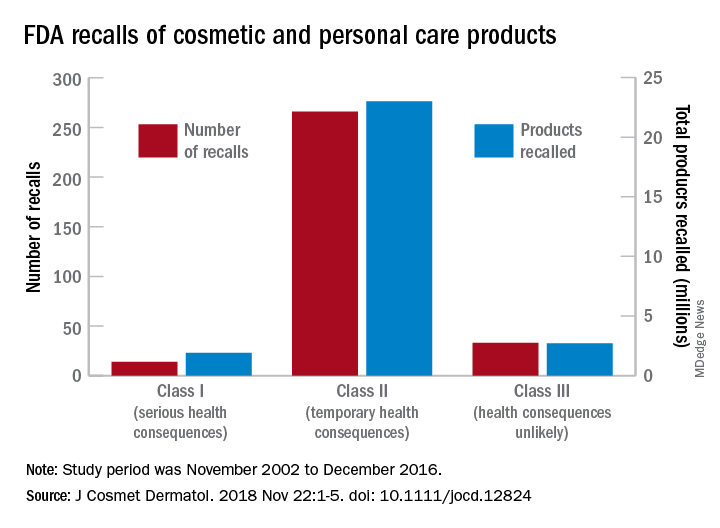
Recalls are classified by the FDA according to risk to patient safety: Class I means there is “reasonable probability of causing serious adverse health outcomes or death,” class II defines the risk as “temporary or reversible,” and class III recalls are “unlikely to cause an adverse health consequence,” they explained.
“While the number of total recalls per year was low in the context of the industry’s size and the ubiquity of cosmetic use by consumers (median: 17/year), these events involved millions of products distributed worldwide,” Dr. Janetos and his associates wrote. The class I recalls covered over 1.9 million products in distribution, the class II recalls accounted for 23 million products, and class II recalls involved over 2.7 million products.
Baby products were the category most likely to be affected, accounting for 76 (24%) of all recalls, the investigators said, with 30 involving one manufacturer of cleansing kits intended for hospital use. All but 3 of the 76 recalls resulted from bacterial contamination.
“The FDA currently has no authority to order a cosmetics manufacturer to recall a product,” they wrote, and “inspectors are only capable of inspecting 0.3% of foreign-imported products yearly,” so underreporting of such problems is likely. “Dermatologists are often the first to encounter [adverse events] related to cosmetic products and can help strengthen public safety by actively reporting these events and advocating for recalls,” said Dr. Janetos and his associates, who did not declare any conflicts of interest.
Information on reporting cosmetic-related complaints to the FDA is available on the FDA website at: https://www.fda.gov/Cosmetics/ComplianceEnforcement/AdverseEventReporting/default.htm.
[email protected]
SOURCE: Janetos TM et al. J Cosmet Dermatol. 2018 Nov 22:1-5. doi: 10.1111/jocd.12824.
Most of the 313 cosmetic and personal care products recalled from 2002 to 2016 had problems with bacterial contamination, according to data obtained from the Food and Drug Administration.
, said Timothy M. Janetos, MD, and his associates at Northwestern University in Chicago. Bacterial contamination was by far the most common reason – 76% of the recalls over that period (11 class I, 217 class II, and 9 class II) – with unapproved ingredients and labeling problems well behind at 6%.
Recalls are classified by the FDA according to risk to patient safety: Class I means there is “reasonable probability of causing serious adverse health outcomes or death,” class II defines the risk as “temporary or reversible,” and class III recalls are “unlikely to cause an adverse health consequence,” they explained.
“While the number of total recalls per year was low in the context of the industry’s size and the ubiquity of cosmetic use by consumers (median: 17/year), these events involved millions of products distributed worldwide,” Dr. Janetos and his associates wrote. The class I recalls covered over 1.9 million products in distribution, the class II recalls accounted for 23 million products, and class II recalls involved over 2.7 million products.
Baby products were the category most likely to be affected, accounting for 76 (24%) of all recalls, the investigators said, with 30 involving one manufacturer of cleansing kits intended for hospital use. All but 3 of the 76 recalls resulted from bacterial contamination.
“The FDA currently has no authority to order a cosmetics manufacturer to recall a product,” they wrote, and “inspectors are only capable of inspecting 0.3% of foreign-imported products yearly,” so underreporting of such problems is likely. “Dermatologists are often the first to encounter [adverse events] related to cosmetic products and can help strengthen public safety by actively reporting these events and advocating for recalls,” said Dr. Janetos and his associates, who did not declare any conflicts of interest.
Information on reporting cosmetic-related complaints to the FDA is available on the FDA website at: https://www.fda.gov/Cosmetics/ComplianceEnforcement/AdverseEventReporting/default.htm.
[email protected]
SOURCE: Janetos TM et al. J Cosmet Dermatol. 2018 Nov 22:1-5. doi: 10.1111/jocd.12824.
Most of the 313 cosmetic and personal care products recalled from 2002 to 2016 had problems with bacterial contamination, according to data obtained from the Food and Drug Administration.
, said Timothy M. Janetos, MD, and his associates at Northwestern University in Chicago. Bacterial contamination was by far the most common reason – 76% of the recalls over that period (11 class I, 217 class II, and 9 class II) – with unapproved ingredients and labeling problems well behind at 6%.
Recalls are classified by the FDA according to risk to patient safety: Class I means there is “reasonable probability of causing serious adverse health outcomes or death,” class II defines the risk as “temporary or reversible,” and class III recalls are “unlikely to cause an adverse health consequence,” they explained.
“While the number of total recalls per year was low in the context of the industry’s size and the ubiquity of cosmetic use by consumers (median: 17/year), these events involved millions of products distributed worldwide,” Dr. Janetos and his associates wrote. The class I recalls covered over 1.9 million products in distribution, the class II recalls accounted for 23 million products, and class II recalls involved over 2.7 million products.
Baby products were the category most likely to be affected, accounting for 76 (24%) of all recalls, the investigators said, with 30 involving one manufacturer of cleansing kits intended for hospital use. All but 3 of the 76 recalls resulted from bacterial contamination.
“The FDA currently has no authority to order a cosmetics manufacturer to recall a product,” they wrote, and “inspectors are only capable of inspecting 0.3% of foreign-imported products yearly,” so underreporting of such problems is likely. “Dermatologists are often the first to encounter [adverse events] related to cosmetic products and can help strengthen public safety by actively reporting these events and advocating for recalls,” said Dr. Janetos and his associates, who did not declare any conflicts of interest.
Information on reporting cosmetic-related complaints to the FDA is available on the FDA website at: https://www.fda.gov/Cosmetics/ComplianceEnforcement/AdverseEventReporting/default.htm.
[email protected]
SOURCE: Janetos TM et al. J Cosmet Dermatol. 2018 Nov 22:1-5. doi: 10.1111/jocd.12824.
FROM THE JOURNAL OF COSMETIC DERMATOLOGY
Fewer insured may have helped slow health spending growth in 2017
Health care spending as a percentage of gross domestic product remained relatively stable in 2017, despite a slowdown in the growth of spending.
Total health care spending in the United States was $3.5 trillion in 2017, an increase of 3.9% from 2016, according to data released Dec. 6 by the Centers for Medicare & Medicaid Services.
The growth rate was down from that of 2016 (4.8%) but similar to growth rates experienced during 2008-2013, according to the research article in Health Affairs.
“The slower growth in health care spending in 2017 resulted primarily from slower growth in hospital care, physician and clinical services, and retail prescription drugs, with residual use and intensity of these goods and services contributing substantially to the trend,” Anne B. Martin, an economist in the CMS Office of the Actuary’s National Health Statistics Group, and her colleagues wrote.
The report notes that slower growth in the use and intensity of health care goods and services in 2017 “may have been affected by slower growth in overall health insurance enrollment, as the insured share of the population fell from 91.1% in 2016 to 90.9% in 2017.”
Spending on hospital care increased 4.6% to $1.1 trillion in 2017 and accounted for 33% of total health care spending; however, growth was slower than in the previous year (5.6%). Ms. Martin and her colleagues noted that growth in outpatient visits slowed while growth in inpatient days increased at about the same rate and prices in hospital care grew in 2017 to 1.7% from 1.2% in the previous year.
Spending on physician and clinical services grew 4.2% in 2017 to $694.3 billion and accounted for 20% of total health care spending. The growth rate is down from the previous year (5.6%) and a recent peak of 6% in 2015.
“Although spending growth for both physician services and clinical services slowed in 2017, the growth rate for the latter (5.0%) continued to out pace the rate for the former (3.9%), as spending for most types of outpatient care centers contributed to the stronger growth in spending for clinical services,” Ms. Martin and her colleagues reported.
They attributed the slowdown to non-price factors, such as slower growth in the use and intensity of physician and clinical services, although price growth for physician and clinical services increased 0.4% in 2017, up from 0.2% in 2016.
Spending on retail prescription drugs grew 0.4% in 2017 to $333.4 billion and accounted for 10% of total national health spending. It is the slowest growth rate increase since 2012, a year that saw a number of blockbuster drugs lose patent protection. This was down from a growth rate of 2.3% in 2016 and down from recent rates of 12.4% in 2014 and 8.9% in 2015.
“Slower growth in non-price factors, such as the use and mix of retail prescription drugs – and, to a lesser extent, in retail prescription drug prices – contributed to the slower overall growth in retail prescription drug spending in 2017,” according to the authors. Key factors included slower growth in the number of prescriptions dispensed, the continued shift to lower-cost generics, and slower growth in the volume of high-cost drugs, particularly those used to treat hepatitis C. Price decreases in generics and lower increases for existing brand-name drugs also contributed to the lower spending growth in 2017.
Ms. Martin and her colleagues highlighted the slower growth rate in the number of prescriptions (1.8% in 2017, down from 2.3% in 2016) “resulted in large part from a decline in the number of prescriptions dispensed for drugs used to treat pain.”
Medicare spending, which represents 20% of all national health care spending in 2017 ($705.9 billion), grew 4.2%, a slight decline from the 4.3% growth in 2016. Enrollment growth slowed slightly to 2.5% in 2017 from 2.7% in the previous year, while in the same time frame, per-enrollee expenditures increased slightly to 1.7% from 1.6%. Slower growth in fee-for-service Medicare spending was offset by faster growth in spending by Medicare private health plans.
Medicaid spending reached $581.9 billion (17% of national health care spending), and the growth rate slowed for the third straight year, increasing 2.9% in 2017 versus 4.2% in 2016. The slower growth “was influenced by a deceleration in enrollment growth and a reduction in the Medicaid net cost of health insurance as the federal government recovered payments from managed care organizations based on their favorable prior-period experience,” the authors stated. Enrollment growth has been decelerating following a peak of growth of 11.9% in 2014 because of states that elected to expand Medicaid eligibility, which was followed by 3 years of slower growth rates of 4.9%, 3.0% and 2.0% in 2015, 2016, and 2017, respectively. Per-enrollee spending also slowed to 0.9% growth in 2017 from a rate of 1.2% in 2016, attributed to “the decline in government administration and the net cost of insurance.”
SOURCE: Martin A et al. Health Aff. 2018. doi: 10.1377/hlthaff.2018.05085.
Health care spending as a percentage of gross domestic product remained relatively stable in 2017, despite a slowdown in the growth of spending.
Total health care spending in the United States was $3.5 trillion in 2017, an increase of 3.9% from 2016, according to data released Dec. 6 by the Centers for Medicare & Medicaid Services.
The growth rate was down from that of 2016 (4.8%) but similar to growth rates experienced during 2008-2013, according to the research article in Health Affairs.
“The slower growth in health care spending in 2017 resulted primarily from slower growth in hospital care, physician and clinical services, and retail prescription drugs, with residual use and intensity of these goods and services contributing substantially to the trend,” Anne B. Martin, an economist in the CMS Office of the Actuary’s National Health Statistics Group, and her colleagues wrote.
The report notes that slower growth in the use and intensity of health care goods and services in 2017 “may have been affected by slower growth in overall health insurance enrollment, as the insured share of the population fell from 91.1% in 2016 to 90.9% in 2017.”
Spending on hospital care increased 4.6% to $1.1 trillion in 2017 and accounted for 33% of total health care spending; however, growth was slower than in the previous year (5.6%). Ms. Martin and her colleagues noted that growth in outpatient visits slowed while growth in inpatient days increased at about the same rate and prices in hospital care grew in 2017 to 1.7% from 1.2% in the previous year.
Spending on physician and clinical services grew 4.2% in 2017 to $694.3 billion and accounted for 20% of total health care spending. The growth rate is down from the previous year (5.6%) and a recent peak of 6% in 2015.
“Although spending growth for both physician services and clinical services slowed in 2017, the growth rate for the latter (5.0%) continued to out pace the rate for the former (3.9%), as spending for most types of outpatient care centers contributed to the stronger growth in spending for clinical services,” Ms. Martin and her colleagues reported.
They attributed the slowdown to non-price factors, such as slower growth in the use and intensity of physician and clinical services, although price growth for physician and clinical services increased 0.4% in 2017, up from 0.2% in 2016.
Spending on retail prescription drugs grew 0.4% in 2017 to $333.4 billion and accounted for 10% of total national health spending. It is the slowest growth rate increase since 2012, a year that saw a number of blockbuster drugs lose patent protection. This was down from a growth rate of 2.3% in 2016 and down from recent rates of 12.4% in 2014 and 8.9% in 2015.
“Slower growth in non-price factors, such as the use and mix of retail prescription drugs – and, to a lesser extent, in retail prescription drug prices – contributed to the slower overall growth in retail prescription drug spending in 2017,” according to the authors. Key factors included slower growth in the number of prescriptions dispensed, the continued shift to lower-cost generics, and slower growth in the volume of high-cost drugs, particularly those used to treat hepatitis C. Price decreases in generics and lower increases for existing brand-name drugs also contributed to the lower spending growth in 2017.
Ms. Martin and her colleagues highlighted the slower growth rate in the number of prescriptions (1.8% in 2017, down from 2.3% in 2016) “resulted in large part from a decline in the number of prescriptions dispensed for drugs used to treat pain.”
Medicare spending, which represents 20% of all national health care spending in 2017 ($705.9 billion), grew 4.2%, a slight decline from the 4.3% growth in 2016. Enrollment growth slowed slightly to 2.5% in 2017 from 2.7% in the previous year, while in the same time frame, per-enrollee expenditures increased slightly to 1.7% from 1.6%. Slower growth in fee-for-service Medicare spending was offset by faster growth in spending by Medicare private health plans.
Medicaid spending reached $581.9 billion (17% of national health care spending), and the growth rate slowed for the third straight year, increasing 2.9% in 2017 versus 4.2% in 2016. The slower growth “was influenced by a deceleration in enrollment growth and a reduction in the Medicaid net cost of health insurance as the federal government recovered payments from managed care organizations based on their favorable prior-period experience,” the authors stated. Enrollment growth has been decelerating following a peak of growth of 11.9% in 2014 because of states that elected to expand Medicaid eligibility, which was followed by 3 years of slower growth rates of 4.9%, 3.0% and 2.0% in 2015, 2016, and 2017, respectively. Per-enrollee spending also slowed to 0.9% growth in 2017 from a rate of 1.2% in 2016, attributed to “the decline in government administration and the net cost of insurance.”
SOURCE: Martin A et al. Health Aff. 2018. doi: 10.1377/hlthaff.2018.05085.
Health care spending as a percentage of gross domestic product remained relatively stable in 2017, despite a slowdown in the growth of spending.
Total health care spending in the United States was $3.5 trillion in 2017, an increase of 3.9% from 2016, according to data released Dec. 6 by the Centers for Medicare & Medicaid Services.
The growth rate was down from that of 2016 (4.8%) but similar to growth rates experienced during 2008-2013, according to the research article in Health Affairs.
“The slower growth in health care spending in 2017 resulted primarily from slower growth in hospital care, physician and clinical services, and retail prescription drugs, with residual use and intensity of these goods and services contributing substantially to the trend,” Anne B. Martin, an economist in the CMS Office of the Actuary’s National Health Statistics Group, and her colleagues wrote.
The report notes that slower growth in the use and intensity of health care goods and services in 2017 “may have been affected by slower growth in overall health insurance enrollment, as the insured share of the population fell from 91.1% in 2016 to 90.9% in 2017.”
Spending on hospital care increased 4.6% to $1.1 trillion in 2017 and accounted for 33% of total health care spending; however, growth was slower than in the previous year (5.6%). Ms. Martin and her colleagues noted that growth in outpatient visits slowed while growth in inpatient days increased at about the same rate and prices in hospital care grew in 2017 to 1.7% from 1.2% in the previous year.
Spending on physician and clinical services grew 4.2% in 2017 to $694.3 billion and accounted for 20% of total health care spending. The growth rate is down from the previous year (5.6%) and a recent peak of 6% in 2015.
“Although spending growth for both physician services and clinical services slowed in 2017, the growth rate for the latter (5.0%) continued to out pace the rate for the former (3.9%), as spending for most types of outpatient care centers contributed to the stronger growth in spending for clinical services,” Ms. Martin and her colleagues reported.
They attributed the slowdown to non-price factors, such as slower growth in the use and intensity of physician and clinical services, although price growth for physician and clinical services increased 0.4% in 2017, up from 0.2% in 2016.
Spending on retail prescription drugs grew 0.4% in 2017 to $333.4 billion and accounted for 10% of total national health spending. It is the slowest growth rate increase since 2012, a year that saw a number of blockbuster drugs lose patent protection. This was down from a growth rate of 2.3% in 2016 and down from recent rates of 12.4% in 2014 and 8.9% in 2015.
“Slower growth in non-price factors, such as the use and mix of retail prescription drugs – and, to a lesser extent, in retail prescription drug prices – contributed to the slower overall growth in retail prescription drug spending in 2017,” according to the authors. Key factors included slower growth in the number of prescriptions dispensed, the continued shift to lower-cost generics, and slower growth in the volume of high-cost drugs, particularly those used to treat hepatitis C. Price decreases in generics and lower increases for existing brand-name drugs also contributed to the lower spending growth in 2017.
Ms. Martin and her colleagues highlighted the slower growth rate in the number of prescriptions (1.8% in 2017, down from 2.3% in 2016) “resulted in large part from a decline in the number of prescriptions dispensed for drugs used to treat pain.”
Medicare spending, which represents 20% of all national health care spending in 2017 ($705.9 billion), grew 4.2%, a slight decline from the 4.3% growth in 2016. Enrollment growth slowed slightly to 2.5% in 2017 from 2.7% in the previous year, while in the same time frame, per-enrollee expenditures increased slightly to 1.7% from 1.6%. Slower growth in fee-for-service Medicare spending was offset by faster growth in spending by Medicare private health plans.
Medicaid spending reached $581.9 billion (17% of national health care spending), and the growth rate slowed for the third straight year, increasing 2.9% in 2017 versus 4.2% in 2016. The slower growth “was influenced by a deceleration in enrollment growth and a reduction in the Medicaid net cost of health insurance as the federal government recovered payments from managed care organizations based on their favorable prior-period experience,” the authors stated. Enrollment growth has been decelerating following a peak of growth of 11.9% in 2014 because of states that elected to expand Medicaid eligibility, which was followed by 3 years of slower growth rates of 4.9%, 3.0% and 2.0% in 2015, 2016, and 2017, respectively. Per-enrollee spending also slowed to 0.9% growth in 2017 from a rate of 1.2% in 2016, attributed to “the decline in government administration and the net cost of insurance.”
SOURCE: Martin A et al. Health Aff. 2018. doi: 10.1377/hlthaff.2018.05085.
FROM HEALTH AFFAIRS
Key clinical point: National health care spending growth slowed to 3.9% in 2017.
Major finding: The $3.5 trillion in national health care spending represents 17.9% of GDP.
Study details: Annual analysis of national health expenditures conducted by federal actuaries.
Disclosures: Analysis conducted by the Centers for Medicaid & Medicare Services Office of the Actuary; the authors have no relevant financial conflicts of interest.
Source: Martin A et al. Health Affairs. 2018. doi: 10.1377/hlthaff.2018.05085.
Action on HealthCare.gov picked up during week 5
Activity during week 5 of open enrollment on HealthCare.gov was up by more than 50% over the previous week, but the total number of plans selected for the 2019 coverage year remains lower than it was last year, according to the Centers for Medicare & Medicaid Services.
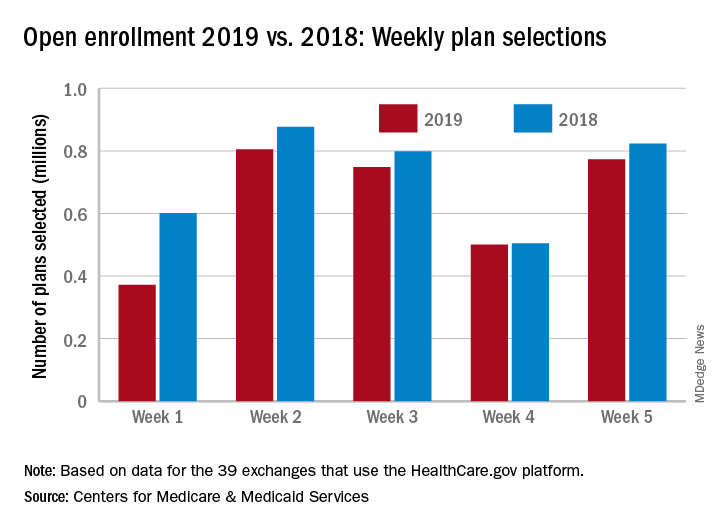
The 773,000 plans selected during week 5 (Nov. 25 – Dec. 1) of the 2019 open enrollment season were an increase of 54% over week 4, CMS data show for the 39 states that use the HealthCare.gov platform, with the cumulative total now at 3.2 million. By comparison, week-5 selections in last year’s open enrollment totaled 823,000, and the cumulative figure was 3.6 million.
The deadline for applying for 2019 coverage on HealthCare.gov is Dec. 15.
Activity during week 5 of open enrollment on HealthCare.gov was up by more than 50% over the previous week, but the total number of plans selected for the 2019 coverage year remains lower than it was last year, according to the Centers for Medicare & Medicaid Services.

The 773,000 plans selected during week 5 (Nov. 25 – Dec. 1) of the 2019 open enrollment season were an increase of 54% over week 4, CMS data show for the 39 states that use the HealthCare.gov platform, with the cumulative total now at 3.2 million. By comparison, week-5 selections in last year’s open enrollment totaled 823,000, and the cumulative figure was 3.6 million.
The deadline for applying for 2019 coverage on HealthCare.gov is Dec. 15.
Activity during week 5 of open enrollment on HealthCare.gov was up by more than 50% over the previous week, but the total number of plans selected for the 2019 coverage year remains lower than it was last year, according to the Centers for Medicare & Medicaid Services.

The 773,000 plans selected during week 5 (Nov. 25 – Dec. 1) of the 2019 open enrollment season were an increase of 54% over week 4, CMS data show for the 39 states that use the HealthCare.gov platform, with the cumulative total now at 3.2 million. By comparison, week-5 selections in last year’s open enrollment totaled 823,000, and the cumulative figure was 3.6 million.
The deadline for applying for 2019 coverage on HealthCare.gov is Dec. 15.
The Gift and the Thought Both Count
It is that time of year when federal compliance officers, clinical ethicists, and staff counsels are flooded with queries about the legal and ethical acceptability of gifts. And no wonder, all the winter holidays often involve giving gifts. The simple and spontaneous acts of giving and receiving gifts become more complicated and deliberative in the federal health care system. Both legal rules and ethical values bear upon who can offer and accept what gift to whom upon what occasion and in what amount. The “Standards of Ethical Conduct for Employees of the Executive Branch” devotes 2 entire subparts to the subject of gifts.1 We will examine a small section of the document that can become a big issue for federal practitioners during that holiday—gifts from patients.
First, veterans (patients) are “prohibited sources” in section 5 CFR §2635.203 (d).1 And since VA employees are subject to restrictions on accepting gifts from sources outside the government, unless an exception applies, federal employees may not accept a gift because of their official position (eg, Federal Practitioner, editor-in-chief) or a gift from a patient (prohibited source; [5 CFR §2635.201]).
It might seem like this is going to be a very short column this month, as gifts from patients are forbidden. Yet, a Christmas card or homemade fudge isn’t really a gift, is it?
5 CFR §2635.203 (b) defines what is and is not a gift: For example, minor items of food or items like a thank-you card are specifically excluded in section (b) 1-10.
Is Christmas an exception or are just types of gifts excluded?
There are exceptions to the general regulation about accepting gifts from prohibited sources. Many staff will recall hearing about the “$20 rule,” which is actually the “$20-50” rule stating that a federal employee may accept a gift from a patient (prohibited source) if the value of said gift is under $20 and the employee does not accept more than $50 from any single source in a calendar year. §2635.204 lists the exceptions. However, starting in 2017, the regulations changed to require that federal employees also consider not just whether they could accept a gift but—ethically—they should take the gift even if it was permitted under the law. The Office of Government Ethics made this change because it wanted to emphasize the importance of considering not just how things are but how things appear to be. The regulations contain detailed descriptions of what employees should think about and stipulates that the decisions will not be further scrutinized (5 CFR §2635.201[b]).1
Based on this new emphasis on appearances, ethically, no doctor or nurse should accept the keys to a new BMW from a patient who owns a luxury car dealership. But what about more prosaic and probable presents: the holiday cookies a single father made with his children for the nurse practitioner who has been his primary care practitioner for years; the birdhouse a Vietnam veteran made in her crafts class for the surgeon who removed her gallbladder; or even the store-bought but no less heartfelt tin of popcorn from an elderly veteran for the hospitalist who saw him through a rough bought of pneumonia?
Related: Happy Federal New Year
The rules about practitioners accepting patient gifts are rational and unambiguous: It is the values conflict surrounding patient gifts that is often emotion driven and muddled; it may be easier and safer to adopt the “just say no” policy. And yet, while this might seem the most unassailable position to avoid a conflict of interest, could this possibly be a more practitioner- than patient-centered standard? Authoritative sources in the ethics literature are equally divided and ambivalent on the question.2,3 The American College of Physicians Ethics Manual states: “In deciding whether to accept a gift from a patient, the physician should consider the nature of the gift and its value to the patient, the potential implications for the patient-physician relationship of accepting or refusing it, and the patient’s probable intention and expectations.”4 A small gift as a token of appreciation is not ethically problematic. Favored treatment as a result of acceptance of any gift is problematic, undermines professionalism, and may interfere with objectivity in the care of the patient.4
Related: Am I My Brother’s/Sister’s Keeper?
Many an ethics commentator have cautioned, “beware of patients bearing gifts.”5 In making an ethical assessment of whether or not to accept the gift, a key question a practitioner needs to ask him or herself is about the patient’s motive. Even the patient may be unaware of the reasons behind their giving, and the wise practitioner will take a mindful moment to think about the context and timing of the gift and the nature of his or her relationship to the patient. Sadly, many of our patients are lonely especially at this family time of year and on some level may hope the gift will help slide the professional relationship toward a more personal one. Some patients may think a gift might earn them preferential treatment. Finally, a few patients may have a romantic or sexual attraction toward a clinician.
Often in the latter 2 cases, a pattern will develop that discloses the patient’s true intent. Very expensive gifts, monetary gifts, excessively personal gifts, or frequent gifts should alert the practitioner that more may be going on. A kind reminder to the patient that providing good care is the only reward needed may be sufficient. For practitioners whose ethical code does not permit them to accept gifts, then a genuine thank you and an explanation of the rules and/or values behind the refusal may be necessary. There are other times when the practitioner or a supervisor/advisor may need to reset the boundaries or even to transfer the patient to another practitioner. The norms in mental health care and psychotherapy are more stringent because of the intimacy of the relationship and the potential vulnerability of patients.6
For gifts that seem genuine and generous or cost a trivial amount, then there is an ethical argument to be made for accepting them with gratitude. In many cultures, hospitality is a tradition, and expressing appreciation a virtue, so when a practitioner refuses to graciously take a small gift, they risk offending the patient. Rejection of a gift can be seen as disrespectful and could cause a rupture in an otherwise sound practitioner-patient relationship. Other patients experience strong feelings of gratitude and admiration for their practitioners, stronger than most of us recognize. The ability for a patient to give their practitioner a holiday gift, particularly one they invested time and energy in creating or choosing, can enhance their sense of self-worth and individual agency. These gifts are not so much an attempt to diminish the professional power differential but to close the gap between 2 human beings in an unequal relationship that is yet one of shared decision making. All practitioners should be aware of the often underappreciated social power of a gift to influence decisions.
Related: Caring Under a Microscope
At the same time, clinicians can strive to be sensitively attuned to the reality that, sometimes the cookie is just a cookie so eat and enjoy, just remember to share with your group. External judgments are often cited as practical rules of thumb for determining the ethical acceptability of a gift: Would you want your mother, newspaper, or colleague to know you took the present? I prefer an internal moral compass that steers always to the true north of is accepting this gift really in the patient’s best interest?
1. Standards of ethical conduct for employees of the executive branch. Fed Regist. 2016;81(223):81641-81657. To be codified at 5 CFR §2635.
2. Spence SA. Patients bearing gifts: are there strings attached. BMJ. 2005;331.
3. American Medical Association. American Medical Association Code of Medical Ethics Opinion 1.2.8. https://www.ama-assn.org/delivering-care/code-medical-ethics-patient-physician-relationships. Accessed November 27, 2018.
4. Snyder L; Ethics, Professionalism, and Human Rights Committee. American College of Physicians Ethics Manual, 6th ed. Ann Intern Med. 2012;156(1, Part 2):73-104.
5. Levine A, Valeriote T. Beware the patient bearing gifts. http://epmonthly.com/article/beware-patient-bearing-gifts. Published December 14, 2016. Accessed November 27, 2018.
6. Brendel DH, Chu J, Radden J, et al. The price of a gift: an approach to receiving gifts from psychiatric patients. Harv Rev Psychiatry. 2007;15(2):43-51.
It is that time of year when federal compliance officers, clinical ethicists, and staff counsels are flooded with queries about the legal and ethical acceptability of gifts. And no wonder, all the winter holidays often involve giving gifts. The simple and spontaneous acts of giving and receiving gifts become more complicated and deliberative in the federal health care system. Both legal rules and ethical values bear upon who can offer and accept what gift to whom upon what occasion and in what amount. The “Standards of Ethical Conduct for Employees of the Executive Branch” devotes 2 entire subparts to the subject of gifts.1 We will examine a small section of the document that can become a big issue for federal practitioners during that holiday—gifts from patients.
First, veterans (patients) are “prohibited sources” in section 5 CFR §2635.203 (d).1 And since VA employees are subject to restrictions on accepting gifts from sources outside the government, unless an exception applies, federal employees may not accept a gift because of their official position (eg, Federal Practitioner, editor-in-chief) or a gift from a patient (prohibited source; [5 CFR §2635.201]).
It might seem like this is going to be a very short column this month, as gifts from patients are forbidden. Yet, a Christmas card or homemade fudge isn’t really a gift, is it?
5 CFR §2635.203 (b) defines what is and is not a gift: For example, minor items of food or items like a thank-you card are specifically excluded in section (b) 1-10.
Is Christmas an exception or are just types of gifts excluded?
There are exceptions to the general regulation about accepting gifts from prohibited sources. Many staff will recall hearing about the “$20 rule,” which is actually the “$20-50” rule stating that a federal employee may accept a gift from a patient (prohibited source) if the value of said gift is under $20 and the employee does not accept more than $50 from any single source in a calendar year. §2635.204 lists the exceptions. However, starting in 2017, the regulations changed to require that federal employees also consider not just whether they could accept a gift but—ethically—they should take the gift even if it was permitted under the law. The Office of Government Ethics made this change because it wanted to emphasize the importance of considering not just how things are but how things appear to be. The regulations contain detailed descriptions of what employees should think about and stipulates that the decisions will not be further scrutinized (5 CFR §2635.201[b]).1
Based on this new emphasis on appearances, ethically, no doctor or nurse should accept the keys to a new BMW from a patient who owns a luxury car dealership. But what about more prosaic and probable presents: the holiday cookies a single father made with his children for the nurse practitioner who has been his primary care practitioner for years; the birdhouse a Vietnam veteran made in her crafts class for the surgeon who removed her gallbladder; or even the store-bought but no less heartfelt tin of popcorn from an elderly veteran for the hospitalist who saw him through a rough bought of pneumonia?
Related: Happy Federal New Year
The rules about practitioners accepting patient gifts are rational and unambiguous: It is the values conflict surrounding patient gifts that is often emotion driven and muddled; it may be easier and safer to adopt the “just say no” policy. And yet, while this might seem the most unassailable position to avoid a conflict of interest, could this possibly be a more practitioner- than patient-centered standard? Authoritative sources in the ethics literature are equally divided and ambivalent on the question.2,3 The American College of Physicians Ethics Manual states: “In deciding whether to accept a gift from a patient, the physician should consider the nature of the gift and its value to the patient, the potential implications for the patient-physician relationship of accepting or refusing it, and the patient’s probable intention and expectations.”4 A small gift as a token of appreciation is not ethically problematic. Favored treatment as a result of acceptance of any gift is problematic, undermines professionalism, and may interfere with objectivity in the care of the patient.4
Related: Am I My Brother’s/Sister’s Keeper?
Many an ethics commentator have cautioned, “beware of patients bearing gifts.”5 In making an ethical assessment of whether or not to accept the gift, a key question a practitioner needs to ask him or herself is about the patient’s motive. Even the patient may be unaware of the reasons behind their giving, and the wise practitioner will take a mindful moment to think about the context and timing of the gift and the nature of his or her relationship to the patient. Sadly, many of our patients are lonely especially at this family time of year and on some level may hope the gift will help slide the professional relationship toward a more personal one. Some patients may think a gift might earn them preferential treatment. Finally, a few patients may have a romantic or sexual attraction toward a clinician.
Often in the latter 2 cases, a pattern will develop that discloses the patient’s true intent. Very expensive gifts, monetary gifts, excessively personal gifts, or frequent gifts should alert the practitioner that more may be going on. A kind reminder to the patient that providing good care is the only reward needed may be sufficient. For practitioners whose ethical code does not permit them to accept gifts, then a genuine thank you and an explanation of the rules and/or values behind the refusal may be necessary. There are other times when the practitioner or a supervisor/advisor may need to reset the boundaries or even to transfer the patient to another practitioner. The norms in mental health care and psychotherapy are more stringent because of the intimacy of the relationship and the potential vulnerability of patients.6
For gifts that seem genuine and generous or cost a trivial amount, then there is an ethical argument to be made for accepting them with gratitude. In many cultures, hospitality is a tradition, and expressing appreciation a virtue, so when a practitioner refuses to graciously take a small gift, they risk offending the patient. Rejection of a gift can be seen as disrespectful and could cause a rupture in an otherwise sound practitioner-patient relationship. Other patients experience strong feelings of gratitude and admiration for their practitioners, stronger than most of us recognize. The ability for a patient to give their practitioner a holiday gift, particularly one they invested time and energy in creating or choosing, can enhance their sense of self-worth and individual agency. These gifts are not so much an attempt to diminish the professional power differential but to close the gap between 2 human beings in an unequal relationship that is yet one of shared decision making. All practitioners should be aware of the often underappreciated social power of a gift to influence decisions.
Related: Caring Under a Microscope
At the same time, clinicians can strive to be sensitively attuned to the reality that, sometimes the cookie is just a cookie so eat and enjoy, just remember to share with your group. External judgments are often cited as practical rules of thumb for determining the ethical acceptability of a gift: Would you want your mother, newspaper, or colleague to know you took the present? I prefer an internal moral compass that steers always to the true north of is accepting this gift really in the patient’s best interest?
It is that time of year when federal compliance officers, clinical ethicists, and staff counsels are flooded with queries about the legal and ethical acceptability of gifts. And no wonder, all the winter holidays often involve giving gifts. The simple and spontaneous acts of giving and receiving gifts become more complicated and deliberative in the federal health care system. Both legal rules and ethical values bear upon who can offer and accept what gift to whom upon what occasion and in what amount. The “Standards of Ethical Conduct for Employees of the Executive Branch” devotes 2 entire subparts to the subject of gifts.1 We will examine a small section of the document that can become a big issue for federal practitioners during that holiday—gifts from patients.
First, veterans (patients) are “prohibited sources” in section 5 CFR §2635.203 (d).1 And since VA employees are subject to restrictions on accepting gifts from sources outside the government, unless an exception applies, federal employees may not accept a gift because of their official position (eg, Federal Practitioner, editor-in-chief) or a gift from a patient (prohibited source; [5 CFR §2635.201]).
It might seem like this is going to be a very short column this month, as gifts from patients are forbidden. Yet, a Christmas card or homemade fudge isn’t really a gift, is it?
5 CFR §2635.203 (b) defines what is and is not a gift: For example, minor items of food or items like a thank-you card are specifically excluded in section (b) 1-10.
Is Christmas an exception or are just types of gifts excluded?
There are exceptions to the general regulation about accepting gifts from prohibited sources. Many staff will recall hearing about the “$20 rule,” which is actually the “$20-50” rule stating that a federal employee may accept a gift from a patient (prohibited source) if the value of said gift is under $20 and the employee does not accept more than $50 from any single source in a calendar year. §2635.204 lists the exceptions. However, starting in 2017, the regulations changed to require that federal employees also consider not just whether they could accept a gift but—ethically—they should take the gift even if it was permitted under the law. The Office of Government Ethics made this change because it wanted to emphasize the importance of considering not just how things are but how things appear to be. The regulations contain detailed descriptions of what employees should think about and stipulates that the decisions will not be further scrutinized (5 CFR §2635.201[b]).1
Based on this new emphasis on appearances, ethically, no doctor or nurse should accept the keys to a new BMW from a patient who owns a luxury car dealership. But what about more prosaic and probable presents: the holiday cookies a single father made with his children for the nurse practitioner who has been his primary care practitioner for years; the birdhouse a Vietnam veteran made in her crafts class for the surgeon who removed her gallbladder; or even the store-bought but no less heartfelt tin of popcorn from an elderly veteran for the hospitalist who saw him through a rough bought of pneumonia?
Related: Happy Federal New Year
The rules about practitioners accepting patient gifts are rational and unambiguous: It is the values conflict surrounding patient gifts that is often emotion driven and muddled; it may be easier and safer to adopt the “just say no” policy. And yet, while this might seem the most unassailable position to avoid a conflict of interest, could this possibly be a more practitioner- than patient-centered standard? Authoritative sources in the ethics literature are equally divided and ambivalent on the question.2,3 The American College of Physicians Ethics Manual states: “In deciding whether to accept a gift from a patient, the physician should consider the nature of the gift and its value to the patient, the potential implications for the patient-physician relationship of accepting or refusing it, and the patient’s probable intention and expectations.”4 A small gift as a token of appreciation is not ethically problematic. Favored treatment as a result of acceptance of any gift is problematic, undermines professionalism, and may interfere with objectivity in the care of the patient.4
Related: Am I My Brother’s/Sister’s Keeper?
Many an ethics commentator have cautioned, “beware of patients bearing gifts.”5 In making an ethical assessment of whether or not to accept the gift, a key question a practitioner needs to ask him or herself is about the patient’s motive. Even the patient may be unaware of the reasons behind their giving, and the wise practitioner will take a mindful moment to think about the context and timing of the gift and the nature of his or her relationship to the patient. Sadly, many of our patients are lonely especially at this family time of year and on some level may hope the gift will help slide the professional relationship toward a more personal one. Some patients may think a gift might earn them preferential treatment. Finally, a few patients may have a romantic or sexual attraction toward a clinician.
Often in the latter 2 cases, a pattern will develop that discloses the patient’s true intent. Very expensive gifts, monetary gifts, excessively personal gifts, or frequent gifts should alert the practitioner that more may be going on. A kind reminder to the patient that providing good care is the only reward needed may be sufficient. For practitioners whose ethical code does not permit them to accept gifts, then a genuine thank you and an explanation of the rules and/or values behind the refusal may be necessary. There are other times when the practitioner or a supervisor/advisor may need to reset the boundaries or even to transfer the patient to another practitioner. The norms in mental health care and psychotherapy are more stringent because of the intimacy of the relationship and the potential vulnerability of patients.6
For gifts that seem genuine and generous or cost a trivial amount, then there is an ethical argument to be made for accepting them with gratitude. In many cultures, hospitality is a tradition, and expressing appreciation a virtue, so when a practitioner refuses to graciously take a small gift, they risk offending the patient. Rejection of a gift can be seen as disrespectful and could cause a rupture in an otherwise sound practitioner-patient relationship. Other patients experience strong feelings of gratitude and admiration for their practitioners, stronger than most of us recognize. The ability for a patient to give their practitioner a holiday gift, particularly one they invested time and energy in creating or choosing, can enhance their sense of self-worth and individual agency. These gifts are not so much an attempt to diminish the professional power differential but to close the gap between 2 human beings in an unequal relationship that is yet one of shared decision making. All practitioners should be aware of the often underappreciated social power of a gift to influence decisions.
Related: Caring Under a Microscope
At the same time, clinicians can strive to be sensitively attuned to the reality that, sometimes the cookie is just a cookie so eat and enjoy, just remember to share with your group. External judgments are often cited as practical rules of thumb for determining the ethical acceptability of a gift: Would you want your mother, newspaper, or colleague to know you took the present? I prefer an internal moral compass that steers always to the true north of is accepting this gift really in the patient’s best interest?
1. Standards of ethical conduct for employees of the executive branch. Fed Regist. 2016;81(223):81641-81657. To be codified at 5 CFR §2635.
2. Spence SA. Patients bearing gifts: are there strings attached. BMJ. 2005;331.
3. American Medical Association. American Medical Association Code of Medical Ethics Opinion 1.2.8. https://www.ama-assn.org/delivering-care/code-medical-ethics-patient-physician-relationships. Accessed November 27, 2018.
4. Snyder L; Ethics, Professionalism, and Human Rights Committee. American College of Physicians Ethics Manual, 6th ed. Ann Intern Med. 2012;156(1, Part 2):73-104.
5. Levine A, Valeriote T. Beware the patient bearing gifts. http://epmonthly.com/article/beware-patient-bearing-gifts. Published December 14, 2016. Accessed November 27, 2018.
6. Brendel DH, Chu J, Radden J, et al. The price of a gift: an approach to receiving gifts from psychiatric patients. Harv Rev Psychiatry. 2007;15(2):43-51.
1. Standards of ethical conduct for employees of the executive branch. Fed Regist. 2016;81(223):81641-81657. To be codified at 5 CFR §2635.
2. Spence SA. Patients bearing gifts: are there strings attached. BMJ. 2005;331.
3. American Medical Association. American Medical Association Code of Medical Ethics Opinion 1.2.8. https://www.ama-assn.org/delivering-care/code-medical-ethics-patient-physician-relationships. Accessed November 27, 2018.
4. Snyder L; Ethics, Professionalism, and Human Rights Committee. American College of Physicians Ethics Manual, 6th ed. Ann Intern Med. 2012;156(1, Part 2):73-104.
5. Levine A, Valeriote T. Beware the patient bearing gifts. http://epmonthly.com/article/beware-patient-bearing-gifts. Published December 14, 2016. Accessed November 27, 2018.
6. Brendel DH, Chu J, Radden J, et al. The price of a gift: an approach to receiving gifts from psychiatric patients. Harv Rev Psychiatry. 2007;15(2):43-51.
Jack Drescher: Sexual Conversion Therapy
Dr. Drescher is a psychiatrist and psychoanalyst in private practice in New York City. In this episode he explains issues with the therapy as well as how to treat patients who have undergone this “therapy.”
In 2016, Dr. Drescher and his colleagues authored a report that reviews the history of conversion therapy and offers guidelines for government regulations as well as for other regulatory bodies.
He and his colleagues wrote that “peer-reviewed literature from multiple professional organizations, including the American Psychiatric Association and the American Psychological Association, have found no evidence that conversion therapy treatments result in changes in sexual orientation.” They also noted that there is evidence suggesting that these treatments are harmful.
Dr. Drescher is a psychiatrist and psychoanalyst in private practice in New York City. In this episode he explains issues with the therapy as well as how to treat patients who have undergone this “therapy.”
In 2016, Dr. Drescher and his colleagues authored a report that reviews the history of conversion therapy and offers guidelines for government regulations as well as for other regulatory bodies.
He and his colleagues wrote that “peer-reviewed literature from multiple professional organizations, including the American Psychiatric Association and the American Psychological Association, have found no evidence that conversion therapy treatments result in changes in sexual orientation.” They also noted that there is evidence suggesting that these treatments are harmful.
Dr. Drescher is a psychiatrist and psychoanalyst in private practice in New York City. In this episode he explains issues with the therapy as well as how to treat patients who have undergone this “therapy.”
In 2016, Dr. Drescher and his colleagues authored a report that reviews the history of conversion therapy and offers guidelines for government regulations as well as for other regulatory bodies.
He and his colleagues wrote that “peer-reviewed literature from multiple professional organizations, including the American Psychiatric Association and the American Psychological Association, have found no evidence that conversion therapy treatments result in changes in sexual orientation.” They also noted that there is evidence suggesting that these treatments are harmful.
Overemphasizing Communities in the National Strategy for Preventing Veteran Suicide Could Undercut VA Successes
In June 2018, the US Department of Veterans Affairs (VA) issued its National Strategy for Preventing Veteran Suicide, 2018-2028. Its 14 goals—many highly innovative—are “to provide a framework for identifying priorities, organizing efforts, and contributing to a national focus on Veteran suicide prevention.”1
The National Strategy recognizes that suicide prevention requires a 3-pronged approach that includes universal, selective, and targeted strategies because “suicide cannot be prevented by any single strategy.”1 Even so, the National Strategy does not heed this core tenet. It focuses exclusively on universal, non-VA community-based priorities and efforts. That focus causes a problem because it neglects the other strategies. It also is precarious because in the current era of VA zero sum budgets, increases in 1 domain come from decreases in another. Thus, sole prioritizing of universal community components could divert funds from extant effective VA suicide prevention programs.
Community-based engagement is unquestionably necessary to prevent suicide among all veterans. Even so, a 10-year prospective strategy should build up, not compromise, VA initiatives. The plan would be improved by explicitly bolstering VA programs that are making a vital difference.
Undercutting VA Suicide Prevention
As my recent review in Federal Practitioner documented, VA’s multiple levels of evidenced-based suicide prevention practices are pre-eminent in the field.2 The VA’s innovative use of predictive analytics to identify and intervene with at-risk individuals is more advanced than anything available in the community. For older veterans who constitute the majority of veterans and the majority of veteran suicides, the VA has more comprehensive and integrated mental health care services than those found in community-based care systems. The embedding of suicide prevention coordinators at every VA facility is unparalleled.
But one would never know about such quality from the National Strategy document: The VA is barely mentioned. The report never advocates for strengthening—or even maintaining—VA’s resources, programs, and efforts. It never recommends that eligible veterans be connected to VA mental health services.
The strategy observes that employment and housing are keys that protect against suicide risk. It does not, however, call for boosting and resourcing VA’s integrated approach that wraps in social services better than does any other program. Similarly, it acknowledges the role of family involvement in mitigating risk but does not propose expanding VA treatments to improve relationship well-being, leaving these services to the private sector.
The National Strategy expands on the recent suicide prevention executive order (EO) for supporting veterans during their transition from military to civilian life. Yet the EO has no funding allocated to this critical initiative. The National Strategy has the same shortcoming. In failing to advocate for more funds to pay for vastly enhanced outreach and intervention, the plan could drain the VA of existing resources needed to maintain its high-quality, suicide prevention services.
First Step: Define the Problem
The National Strategy wisely specifies that the initial step in any suicide prevention effort should be to “define the problem. This involves collecting data to determine the ‘who,’ ‘what,’ ‘where,’ ‘when,’ and ‘how’ of suicide deaths.” Then, “identify risk and protective factors.”
Yet the report doesn’t follow its own advice. Although little is known about the 14 of 20 veterans who die by suicide daily who are not recent users of VA health services, the National Strategy foregoes the necessity of first ascertaining crucial factors, including whether those veterans were (a) eligible for VA care; (b) receiving any mental health or substance use treatment; and (c) going through life crises, etc. What’s needed before reallocating funds to community-based programs is for Congress to finance a post-suicide, case-by-case study of these veteran decedents who did not use VA.
Proceeding in this manner has 2 benefits. First, it would allow initiatives to be targeted. Second, it could preserve funds for successful VA programs that otherwise might be cut to pay for private sector programs.
A Positive Starting Point
There are many positive components of the National Strategy for Preventing Veteran Suicide that will make a difference. That said, they fall short of their potential. The following are suggestions that could strengthen the VA’s plan.
First, given the overwhelming use of firearms by veterans who die by suicide, the National Strategy acknowledges that an effective prevention policy must attend to this factor. It prudently calls for expansion of firearm safety/suicide prevention collaboration with firearm owners, firearm dealers, shooting clubs, and gun/hunting organizations. This will help ensure that lethal means safety counseling is culturally relevant, comes from a trusted source, and has no antifirearm bias.
Nothing would be more useful in diminishing suicide than correcting the false belief among many veterans that “the VA wants to take away our guns.” If that misperception were replaced with an accurate message, not only would more at-risk veterans seek out VA mental health care, more veterans/families/friends would adopt a new cultural norm akin to buddies talk to vets in crisis about safely storing guns. Establishing a workgroup with gun constituency collaborators could spearhead such a shift.
Second, although, the National Strategy emphasizes the benefits of using peer supports, peers currently express qualms that they have too little expertise intervening with this vulnerable population. Peers could be given extensive training and continued supervision in suicide prevention techniques.
Third, the National Strategy calls for expanded use of big data predictive analytics, whose initial implementation has shown great promise. However, it fails to mention that this approach depends on linked electronic health records and therefore best succeeds for at-risk veterans within VA but not in insulated community care.
Fourth, the National Strategy recognizes that reshaping media and entertainment portrayals could help prevent veteran suicide. Yet it ignores the importance of correcting the sullied narrative about the VA. The disproportionate negative image contributes to veterans’ reticence to seek VA health care. One simple solution would be to require that service members readying to transition to civilian life be informed about the superior nature of VA mental health care. Another is to provide the media with positive VA stories more routinely.
Fifth, the National Strategy suggests that enhanced community care guidelines be developed, but it never recommends that community partners should equal VA’s standards. Those providers should be mandated to conduct the same root cause analyses and comprehensive documentation of suicide risk assessments that VA does.
Conclusion
With zero sum department budgets, the National Strategy’s exclusive priority on public health, community-based initiatives could undercut VA successes. An amended plan that explicitly supports and further strengthens successful VA suicide prevention programs is warranted.
1. US Department of Veterans Affairs. National Strategy for Preventing Veteran Suicide, 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf Published June 2018. Accessed November 6, 2018.
2. Lemle RB. Choice program expansion jeopardizes high-quality VHA mental health Services. Fed Pract. 2018;35(3):18-24.
In June 2018, the US Department of Veterans Affairs (VA) issued its National Strategy for Preventing Veteran Suicide, 2018-2028. Its 14 goals—many highly innovative—are “to provide a framework for identifying priorities, organizing efforts, and contributing to a national focus on Veteran suicide prevention.”1
The National Strategy recognizes that suicide prevention requires a 3-pronged approach that includes universal, selective, and targeted strategies because “suicide cannot be prevented by any single strategy.”1 Even so, the National Strategy does not heed this core tenet. It focuses exclusively on universal, non-VA community-based priorities and efforts. That focus causes a problem because it neglects the other strategies. It also is precarious because in the current era of VA zero sum budgets, increases in 1 domain come from decreases in another. Thus, sole prioritizing of universal community components could divert funds from extant effective VA suicide prevention programs.
Community-based engagement is unquestionably necessary to prevent suicide among all veterans. Even so, a 10-year prospective strategy should build up, not compromise, VA initiatives. The plan would be improved by explicitly bolstering VA programs that are making a vital difference.
Undercutting VA Suicide Prevention
As my recent review in Federal Practitioner documented, VA’s multiple levels of evidenced-based suicide prevention practices are pre-eminent in the field.2 The VA’s innovative use of predictive analytics to identify and intervene with at-risk individuals is more advanced than anything available in the community. For older veterans who constitute the majority of veterans and the majority of veteran suicides, the VA has more comprehensive and integrated mental health care services than those found in community-based care systems. The embedding of suicide prevention coordinators at every VA facility is unparalleled.
But one would never know about such quality from the National Strategy document: The VA is barely mentioned. The report never advocates for strengthening—or even maintaining—VA’s resources, programs, and efforts. It never recommends that eligible veterans be connected to VA mental health services.
The strategy observes that employment and housing are keys that protect against suicide risk. It does not, however, call for boosting and resourcing VA’s integrated approach that wraps in social services better than does any other program. Similarly, it acknowledges the role of family involvement in mitigating risk but does not propose expanding VA treatments to improve relationship well-being, leaving these services to the private sector.
The National Strategy expands on the recent suicide prevention executive order (EO) for supporting veterans during their transition from military to civilian life. Yet the EO has no funding allocated to this critical initiative. The National Strategy has the same shortcoming. In failing to advocate for more funds to pay for vastly enhanced outreach and intervention, the plan could drain the VA of existing resources needed to maintain its high-quality, suicide prevention services.
First Step: Define the Problem
The National Strategy wisely specifies that the initial step in any suicide prevention effort should be to “define the problem. This involves collecting data to determine the ‘who,’ ‘what,’ ‘where,’ ‘when,’ and ‘how’ of suicide deaths.” Then, “identify risk and protective factors.”
Yet the report doesn’t follow its own advice. Although little is known about the 14 of 20 veterans who die by suicide daily who are not recent users of VA health services, the National Strategy foregoes the necessity of first ascertaining crucial factors, including whether those veterans were (a) eligible for VA care; (b) receiving any mental health or substance use treatment; and (c) going through life crises, etc. What’s needed before reallocating funds to community-based programs is for Congress to finance a post-suicide, case-by-case study of these veteran decedents who did not use VA.
Proceeding in this manner has 2 benefits. First, it would allow initiatives to be targeted. Second, it could preserve funds for successful VA programs that otherwise might be cut to pay for private sector programs.
A Positive Starting Point
There are many positive components of the National Strategy for Preventing Veteran Suicide that will make a difference. That said, they fall short of their potential. The following are suggestions that could strengthen the VA’s plan.
First, given the overwhelming use of firearms by veterans who die by suicide, the National Strategy acknowledges that an effective prevention policy must attend to this factor. It prudently calls for expansion of firearm safety/suicide prevention collaboration with firearm owners, firearm dealers, shooting clubs, and gun/hunting organizations. This will help ensure that lethal means safety counseling is culturally relevant, comes from a trusted source, and has no antifirearm bias.
Nothing would be more useful in diminishing suicide than correcting the false belief among many veterans that “the VA wants to take away our guns.” If that misperception were replaced with an accurate message, not only would more at-risk veterans seek out VA mental health care, more veterans/families/friends would adopt a new cultural norm akin to buddies talk to vets in crisis about safely storing guns. Establishing a workgroup with gun constituency collaborators could spearhead such a shift.
Second, although, the National Strategy emphasizes the benefits of using peer supports, peers currently express qualms that they have too little expertise intervening with this vulnerable population. Peers could be given extensive training and continued supervision in suicide prevention techniques.
Third, the National Strategy calls for expanded use of big data predictive analytics, whose initial implementation has shown great promise. However, it fails to mention that this approach depends on linked electronic health records and therefore best succeeds for at-risk veterans within VA but not in insulated community care.
Fourth, the National Strategy recognizes that reshaping media and entertainment portrayals could help prevent veteran suicide. Yet it ignores the importance of correcting the sullied narrative about the VA. The disproportionate negative image contributes to veterans’ reticence to seek VA health care. One simple solution would be to require that service members readying to transition to civilian life be informed about the superior nature of VA mental health care. Another is to provide the media with positive VA stories more routinely.
Fifth, the National Strategy suggests that enhanced community care guidelines be developed, but it never recommends that community partners should equal VA’s standards. Those providers should be mandated to conduct the same root cause analyses and comprehensive documentation of suicide risk assessments that VA does.
Conclusion
With zero sum department budgets, the National Strategy’s exclusive priority on public health, community-based initiatives could undercut VA successes. An amended plan that explicitly supports and further strengthens successful VA suicide prevention programs is warranted.
In June 2018, the US Department of Veterans Affairs (VA) issued its National Strategy for Preventing Veteran Suicide, 2018-2028. Its 14 goals—many highly innovative—are “to provide a framework for identifying priorities, organizing efforts, and contributing to a national focus on Veteran suicide prevention.”1
The National Strategy recognizes that suicide prevention requires a 3-pronged approach that includes universal, selective, and targeted strategies because “suicide cannot be prevented by any single strategy.”1 Even so, the National Strategy does not heed this core tenet. It focuses exclusively on universal, non-VA community-based priorities and efforts. That focus causes a problem because it neglects the other strategies. It also is precarious because in the current era of VA zero sum budgets, increases in 1 domain come from decreases in another. Thus, sole prioritizing of universal community components could divert funds from extant effective VA suicide prevention programs.
Community-based engagement is unquestionably necessary to prevent suicide among all veterans. Even so, a 10-year prospective strategy should build up, not compromise, VA initiatives. The plan would be improved by explicitly bolstering VA programs that are making a vital difference.
Undercutting VA Suicide Prevention
As my recent review in Federal Practitioner documented, VA’s multiple levels of evidenced-based suicide prevention practices are pre-eminent in the field.2 The VA’s innovative use of predictive analytics to identify and intervene with at-risk individuals is more advanced than anything available in the community. For older veterans who constitute the majority of veterans and the majority of veteran suicides, the VA has more comprehensive and integrated mental health care services than those found in community-based care systems. The embedding of suicide prevention coordinators at every VA facility is unparalleled.
But one would never know about such quality from the National Strategy document: The VA is barely mentioned. The report never advocates for strengthening—or even maintaining—VA’s resources, programs, and efforts. It never recommends that eligible veterans be connected to VA mental health services.
The strategy observes that employment and housing are keys that protect against suicide risk. It does not, however, call for boosting and resourcing VA’s integrated approach that wraps in social services better than does any other program. Similarly, it acknowledges the role of family involvement in mitigating risk but does not propose expanding VA treatments to improve relationship well-being, leaving these services to the private sector.
The National Strategy expands on the recent suicide prevention executive order (EO) for supporting veterans during their transition from military to civilian life. Yet the EO has no funding allocated to this critical initiative. The National Strategy has the same shortcoming. In failing to advocate for more funds to pay for vastly enhanced outreach and intervention, the plan could drain the VA of existing resources needed to maintain its high-quality, suicide prevention services.
First Step: Define the Problem
The National Strategy wisely specifies that the initial step in any suicide prevention effort should be to “define the problem. This involves collecting data to determine the ‘who,’ ‘what,’ ‘where,’ ‘when,’ and ‘how’ of suicide deaths.” Then, “identify risk and protective factors.”
Yet the report doesn’t follow its own advice. Although little is known about the 14 of 20 veterans who die by suicide daily who are not recent users of VA health services, the National Strategy foregoes the necessity of first ascertaining crucial factors, including whether those veterans were (a) eligible for VA care; (b) receiving any mental health or substance use treatment; and (c) going through life crises, etc. What’s needed before reallocating funds to community-based programs is for Congress to finance a post-suicide, case-by-case study of these veteran decedents who did not use VA.
Proceeding in this manner has 2 benefits. First, it would allow initiatives to be targeted. Second, it could preserve funds for successful VA programs that otherwise might be cut to pay for private sector programs.
A Positive Starting Point
There are many positive components of the National Strategy for Preventing Veteran Suicide that will make a difference. That said, they fall short of their potential. The following are suggestions that could strengthen the VA’s plan.
First, given the overwhelming use of firearms by veterans who die by suicide, the National Strategy acknowledges that an effective prevention policy must attend to this factor. It prudently calls for expansion of firearm safety/suicide prevention collaboration with firearm owners, firearm dealers, shooting clubs, and gun/hunting organizations. This will help ensure that lethal means safety counseling is culturally relevant, comes from a trusted source, and has no antifirearm bias.
Nothing would be more useful in diminishing suicide than correcting the false belief among many veterans that “the VA wants to take away our guns.” If that misperception were replaced with an accurate message, not only would more at-risk veterans seek out VA mental health care, more veterans/families/friends would adopt a new cultural norm akin to buddies talk to vets in crisis about safely storing guns. Establishing a workgroup with gun constituency collaborators could spearhead such a shift.
Second, although, the National Strategy emphasizes the benefits of using peer supports, peers currently express qualms that they have too little expertise intervening with this vulnerable population. Peers could be given extensive training and continued supervision in suicide prevention techniques.
Third, the National Strategy calls for expanded use of big data predictive analytics, whose initial implementation has shown great promise. However, it fails to mention that this approach depends on linked electronic health records and therefore best succeeds for at-risk veterans within VA but not in insulated community care.
Fourth, the National Strategy recognizes that reshaping media and entertainment portrayals could help prevent veteran suicide. Yet it ignores the importance of correcting the sullied narrative about the VA. The disproportionate negative image contributes to veterans’ reticence to seek VA health care. One simple solution would be to require that service members readying to transition to civilian life be informed about the superior nature of VA mental health care. Another is to provide the media with positive VA stories more routinely.
Fifth, the National Strategy suggests that enhanced community care guidelines be developed, but it never recommends that community partners should equal VA’s standards. Those providers should be mandated to conduct the same root cause analyses and comprehensive documentation of suicide risk assessments that VA does.
Conclusion
With zero sum department budgets, the National Strategy’s exclusive priority on public health, community-based initiatives could undercut VA successes. An amended plan that explicitly supports and further strengthens successful VA suicide prevention programs is warranted.
1. US Department of Veterans Affairs. National Strategy for Preventing Veteran Suicide, 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf Published June 2018. Accessed November 6, 2018.
2. Lemle RB. Choice program expansion jeopardizes high-quality VHA mental health Services. Fed Pract. 2018;35(3):18-24.
1. US Department of Veterans Affairs. National Strategy for Preventing Veteran Suicide, 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf Published June 2018. Accessed November 6, 2018.
2. Lemle RB. Choice program expansion jeopardizes high-quality VHA mental health Services. Fed Pract. 2018;35(3):18-24.
Are We Overproducing NPs and PAs?
Recently, the Association of American Medical Colleges (AAMC) reiterated its projection of a physician shortage in the United States, predicting a shortfall of up to 121,300 physicians by 2030. The shortage would affect primary care as well as medical and surgical specialties. These projections are consistent with prior estimates and, AAMC says, take into account both utilization of NPs and PAs and future changes in how care is delivered.1
However, other entities have suggested we are misinterpreting the situation. The Institute of Medicine (IOM) has argued that there is no physician shortage. According to their analysis, the health care system isn’t undermanned—rather, it’s inefficient and relies too heavily on physicians and not enough on advanced practice providers. Furthermore, the IOM posits that many of the studies upon which physician workforce projections have been based fail to account for advances in medicine and technology that impact care delivery: telehealth, new medications, and medical devices that give patients a more active role in their health maintenance.2
Who’s right? You might say, “Who cares?” but this isn’t simply a matter of institutional reputation; the data have informed action plans for offsetting the projected shortage. Since 2002, medical schools have increased class sizes by 30% and are working to ensure that the supply of physicians will be sufficient to meet future needs—even though funding for residency training has been frozen since 1997. At the same time, many thought leaders—including the IOM—have recognized NPs and PAs as significant contributors to the health care workforce. In 2007, for example, Cooper called on the NP and PA professions to expand their training capacity, predicting that neither would have a supply of practitioners to meet needs in the event of a physician shortage.3
Both professions took that message to heart. There are now more than 123,000 certified PAs (70% of whom work in specialty practice) and 248,000 licensed NPs (87% in primary care) in the United States.4,5 There are 239 accredited PA programs (including those with provisional or probationary status), with the number of new graduates per year expected to reach 18,000 by 2026 (compared to 9,000 in 2018).6,7 There are about 400 academic institutions in the US that have an NP program; in 2016-2017, more than 26,000 new graduates completed their training.5,8 Overall, the Bureau of Labor Statistics projects that by 2024 the NP profession will have grown by 36%, the PA profession by 37%, and the physician population by 13% (excluding anesthesiologists and surgeons).9
There is no argument that NPs and PAs are making an enormous impact on the quality and accessibility of health care in this country. But I am starting to hear rumblings that we may be educating too many NPs and PAs—especially if the physician shortage is not as dire as predicted.
This entire conversation takes me back to the 1970s, when the Graduate Medical Education National Advisory Committee (GMENAC) projected a surplus of physicians, and a moratorium was placed on medical school enrollment. Those projections were validated and repeated through the 1990s; in fact, the aforementioned Cooper was among the first to flip the message around, using new calculations and considerations to project a physician shortage.10
GMENAC is a classic example of what happens when people and entities overreact to a projection of some kind. If there is a physician shortage today, GMENAC is probably partly responsible because their prediction of an oversupply triggered an arguably over-the-top response. Everyone worked so hard to avoid a surplus that they are creating a deficit!
Continue to: As I listen to...
As I listen to those rumblings of “too many NPs and PAs,” I wonder if this is a mirror to that GMENAC response. Have the NP and PA professions worked so hard to offset the physician shortage (real or imagined) that we may face a glut of NPs and PAs? If so, the concern is that within five to 10 years, we won’t have employment opportunities for all of them. That’s the fear driving these whispers, isn’t it?
As far back as 2000—when this conversation was in its infancy—Dehn and Cawley discussed the consequences of expanding the supply of NPs and PAs. They questioned how the number of, and demand for, NPs and PAs would be balanced in America’s future health care marketplace and wondered if a sharp growth in NP and PA graduates (in conjunction with similar increases in other health professions) could surpass demand and prompt an oversupply, resulting in underemployment and market saturation.11
So, is it time to pause and take another look at the numbers and needs? Maybe. But I believe one of the aspects we must continue to focus on is the quality of our professions. In the wake of the projected physician shortage, the NP profession developed its Doctor of Nursing Practice and the PA profession added postgraduate training opportunities in specific specialties. These not only enhance NPs’ and PAs’ professional credentials—they equip us to provide better patient care. At the end of the day, our ability to care for patients will be the rubric upon which we are judged.
We’ve already been making the case for our professions and gaining recognition throughout this process. When Salsberg wrote about the physician shortage in Health Affairs (2015), he reminded us that a critical factor is the supply and availability of clinicians other than physicians (NPs, PAs, midwives) who can make a significant contribution to access and efficiency of health care. He called for NPs and PAs to be fully integrated into the delivery system and to be allowed scope of practice consistent with their education and training.7
Continue to: Both NPs and PAs have become...
Both NPs and PAs have become participants in dialogues on health policy and health care reform. Both professions are spending increasing dollars on national advertising to raise awareness of their critical role in expanding access to primary care for millions of Americans. In fact, Princeton University Professor of Economics Uwe E. Reinhardt told the New York Times, “The doctors are fighting a losing battle. The nurses are like insurgents. They are occasionally beaten back, but they’ll win in the long run. They have economics and common sense on their side.”12 Some suggest that PAs need to fight a similar battle.
So, dear reader, what do you think? Should we be concerned that we are educating too many NPs and PAs? Does that argument become somewhat irrelevant if we can firmly establish a substantial role for ourselves in the future of health care? I would love to hear your thoughts; email me at [email protected].
1. Association of American Medical Colleges. The Complexities of Physician Supply and Demand: Projections from 2016 to 2030—2018 Update. Final Report. March 2018. https://aamc-black.global.ssl.fastly.net/production/media/filer_public/85/d7/85d7b689-f417-4ef0-97fb-ecc129836829/aamc_2018_workforce_projections_update_april_11_2018.pdf
2. Institute of Medicine. Graduate Medical Education That Meets the Nation’s Health Needs. Washington, DC: The National Academies Press; 2014.
3. Cooper RA. New directions for nurse practitioners and physician assistants in the era of physician shortages. Acad Med. 2007;82(9):827-828.
4. American Academy of PAs. What is a PA? Frequently asked questions. www.aapa.org/wp-content/uploads/2018/06/Frequently_Asked_Questions_4.3_FINAL.pdf. Accessed November 14, 2018.
5. American Association of Nurse Practitioners. NP Fact Sheet. www.aanp.org/about/all-about-nps/np-fact-sheet. Accessed November 14, 2018.
6. Accreditation Review Commission on Education for the Physician Assistant. Accredited programs. www.arc-pa.org/accreditation/accredited-programs/. Accessed November 14, 2018.
7. Salsberg E. The nurse practitioner, physician assistant, and pharmacist pipelines: continued growth. Health Affairs. May 26, 2015. www.healthaffairs.org/do/10.1377/hblog20150526.047896/full/. Accessed November 14, 2018.
8. American Association of Nurse Practitioners. Planning your NP education. www.aanp.org/student-resources/planning-your-np-education. Accessed November 14, 2018.
9. United States Department of Labor, Bureau of Labor Statistics. Occupational outlook handbook: healthcare. www.bls.gov/ooh/healthcare/home.htm. Accessed November 14, 2018.
10. Dalen JE. The moratorium on US medical school enrollment, from 1980 to 2005: what were we thinking? Am J Med. 2008;121(2):e1-e2.
11. Dehn RW, Cawley JF. Looking into tomorrow: health workforce issues confronting physician assistants. JAAPA. 2000;13(11):29-32, 35-38, 43-46.
12. Tavernise S. Doctoring, without the doctor. New York Times. May 25, 2015. www.nytimes.com/2015/05/26/health/rural-nebraska-offers-stark-view-of-nursing-autonomy-debate.html. Accessed November 14, 2018.
Recently, the Association of American Medical Colleges (AAMC) reiterated its projection of a physician shortage in the United States, predicting a shortfall of up to 121,300 physicians by 2030. The shortage would affect primary care as well as medical and surgical specialties. These projections are consistent with prior estimates and, AAMC says, take into account both utilization of NPs and PAs and future changes in how care is delivered.1
However, other entities have suggested we are misinterpreting the situation. The Institute of Medicine (IOM) has argued that there is no physician shortage. According to their analysis, the health care system isn’t undermanned—rather, it’s inefficient and relies too heavily on physicians and not enough on advanced practice providers. Furthermore, the IOM posits that many of the studies upon which physician workforce projections have been based fail to account for advances in medicine and technology that impact care delivery: telehealth, new medications, and medical devices that give patients a more active role in their health maintenance.2
Who’s right? You might say, “Who cares?” but this isn’t simply a matter of institutional reputation; the data have informed action plans for offsetting the projected shortage. Since 2002, medical schools have increased class sizes by 30% and are working to ensure that the supply of physicians will be sufficient to meet future needs—even though funding for residency training has been frozen since 1997. At the same time, many thought leaders—including the IOM—have recognized NPs and PAs as significant contributors to the health care workforce. In 2007, for example, Cooper called on the NP and PA professions to expand their training capacity, predicting that neither would have a supply of practitioners to meet needs in the event of a physician shortage.3
Both professions took that message to heart. There are now more than 123,000 certified PAs (70% of whom work in specialty practice) and 248,000 licensed NPs (87% in primary care) in the United States.4,5 There are 239 accredited PA programs (including those with provisional or probationary status), with the number of new graduates per year expected to reach 18,000 by 2026 (compared to 9,000 in 2018).6,7 There are about 400 academic institutions in the US that have an NP program; in 2016-2017, more than 26,000 new graduates completed their training.5,8 Overall, the Bureau of Labor Statistics projects that by 2024 the NP profession will have grown by 36%, the PA profession by 37%, and the physician population by 13% (excluding anesthesiologists and surgeons).9
There is no argument that NPs and PAs are making an enormous impact on the quality and accessibility of health care in this country. But I am starting to hear rumblings that we may be educating too many NPs and PAs—especially if the physician shortage is not as dire as predicted.
This entire conversation takes me back to the 1970s, when the Graduate Medical Education National Advisory Committee (GMENAC) projected a surplus of physicians, and a moratorium was placed on medical school enrollment. Those projections were validated and repeated through the 1990s; in fact, the aforementioned Cooper was among the first to flip the message around, using new calculations and considerations to project a physician shortage.10
GMENAC is a classic example of what happens when people and entities overreact to a projection of some kind. If there is a physician shortage today, GMENAC is probably partly responsible because their prediction of an oversupply triggered an arguably over-the-top response. Everyone worked so hard to avoid a surplus that they are creating a deficit!
Continue to: As I listen to...
As I listen to those rumblings of “too many NPs and PAs,” I wonder if this is a mirror to that GMENAC response. Have the NP and PA professions worked so hard to offset the physician shortage (real or imagined) that we may face a glut of NPs and PAs? If so, the concern is that within five to 10 years, we won’t have employment opportunities for all of them. That’s the fear driving these whispers, isn’t it?
As far back as 2000—when this conversation was in its infancy—Dehn and Cawley discussed the consequences of expanding the supply of NPs and PAs. They questioned how the number of, and demand for, NPs and PAs would be balanced in America’s future health care marketplace and wondered if a sharp growth in NP and PA graduates (in conjunction with similar increases in other health professions) could surpass demand and prompt an oversupply, resulting in underemployment and market saturation.11
So, is it time to pause and take another look at the numbers and needs? Maybe. But I believe one of the aspects we must continue to focus on is the quality of our professions. In the wake of the projected physician shortage, the NP profession developed its Doctor of Nursing Practice and the PA profession added postgraduate training opportunities in specific specialties. These not only enhance NPs’ and PAs’ professional credentials—they equip us to provide better patient care. At the end of the day, our ability to care for patients will be the rubric upon which we are judged.
We’ve already been making the case for our professions and gaining recognition throughout this process. When Salsberg wrote about the physician shortage in Health Affairs (2015), he reminded us that a critical factor is the supply and availability of clinicians other than physicians (NPs, PAs, midwives) who can make a significant contribution to access and efficiency of health care. He called for NPs and PAs to be fully integrated into the delivery system and to be allowed scope of practice consistent with their education and training.7
Continue to: Both NPs and PAs have become...
Both NPs and PAs have become participants in dialogues on health policy and health care reform. Both professions are spending increasing dollars on national advertising to raise awareness of their critical role in expanding access to primary care for millions of Americans. In fact, Princeton University Professor of Economics Uwe E. Reinhardt told the New York Times, “The doctors are fighting a losing battle. The nurses are like insurgents. They are occasionally beaten back, but they’ll win in the long run. They have economics and common sense on their side.”12 Some suggest that PAs need to fight a similar battle.
So, dear reader, what do you think? Should we be concerned that we are educating too many NPs and PAs? Does that argument become somewhat irrelevant if we can firmly establish a substantial role for ourselves in the future of health care? I would love to hear your thoughts; email me at [email protected].
Recently, the Association of American Medical Colleges (AAMC) reiterated its projection of a physician shortage in the United States, predicting a shortfall of up to 121,300 physicians by 2030. The shortage would affect primary care as well as medical and surgical specialties. These projections are consistent with prior estimates and, AAMC says, take into account both utilization of NPs and PAs and future changes in how care is delivered.1
However, other entities have suggested we are misinterpreting the situation. The Institute of Medicine (IOM) has argued that there is no physician shortage. According to their analysis, the health care system isn’t undermanned—rather, it’s inefficient and relies too heavily on physicians and not enough on advanced practice providers. Furthermore, the IOM posits that many of the studies upon which physician workforce projections have been based fail to account for advances in medicine and technology that impact care delivery: telehealth, new medications, and medical devices that give patients a more active role in their health maintenance.2
Who’s right? You might say, “Who cares?” but this isn’t simply a matter of institutional reputation; the data have informed action plans for offsetting the projected shortage. Since 2002, medical schools have increased class sizes by 30% and are working to ensure that the supply of physicians will be sufficient to meet future needs—even though funding for residency training has been frozen since 1997. At the same time, many thought leaders—including the IOM—have recognized NPs and PAs as significant contributors to the health care workforce. In 2007, for example, Cooper called on the NP and PA professions to expand their training capacity, predicting that neither would have a supply of practitioners to meet needs in the event of a physician shortage.3
Both professions took that message to heart. There are now more than 123,000 certified PAs (70% of whom work in specialty practice) and 248,000 licensed NPs (87% in primary care) in the United States.4,5 There are 239 accredited PA programs (including those with provisional or probationary status), with the number of new graduates per year expected to reach 18,000 by 2026 (compared to 9,000 in 2018).6,7 There are about 400 academic institutions in the US that have an NP program; in 2016-2017, more than 26,000 new graduates completed their training.5,8 Overall, the Bureau of Labor Statistics projects that by 2024 the NP profession will have grown by 36%, the PA profession by 37%, and the physician population by 13% (excluding anesthesiologists and surgeons).9
There is no argument that NPs and PAs are making an enormous impact on the quality and accessibility of health care in this country. But I am starting to hear rumblings that we may be educating too many NPs and PAs—especially if the physician shortage is not as dire as predicted.
This entire conversation takes me back to the 1970s, when the Graduate Medical Education National Advisory Committee (GMENAC) projected a surplus of physicians, and a moratorium was placed on medical school enrollment. Those projections were validated and repeated through the 1990s; in fact, the aforementioned Cooper was among the first to flip the message around, using new calculations and considerations to project a physician shortage.10
GMENAC is a classic example of what happens when people and entities overreact to a projection of some kind. If there is a physician shortage today, GMENAC is probably partly responsible because their prediction of an oversupply triggered an arguably over-the-top response. Everyone worked so hard to avoid a surplus that they are creating a deficit!
Continue to: As I listen to...
As I listen to those rumblings of “too many NPs and PAs,” I wonder if this is a mirror to that GMENAC response. Have the NP and PA professions worked so hard to offset the physician shortage (real or imagined) that we may face a glut of NPs and PAs? If so, the concern is that within five to 10 years, we won’t have employment opportunities for all of them. That’s the fear driving these whispers, isn’t it?
As far back as 2000—when this conversation was in its infancy—Dehn and Cawley discussed the consequences of expanding the supply of NPs and PAs. They questioned how the number of, and demand for, NPs and PAs would be balanced in America’s future health care marketplace and wondered if a sharp growth in NP and PA graduates (in conjunction with similar increases in other health professions) could surpass demand and prompt an oversupply, resulting in underemployment and market saturation.11
So, is it time to pause and take another look at the numbers and needs? Maybe. But I believe one of the aspects we must continue to focus on is the quality of our professions. In the wake of the projected physician shortage, the NP profession developed its Doctor of Nursing Practice and the PA profession added postgraduate training opportunities in specific specialties. These not only enhance NPs’ and PAs’ professional credentials—they equip us to provide better patient care. At the end of the day, our ability to care for patients will be the rubric upon which we are judged.
We’ve already been making the case for our professions and gaining recognition throughout this process. When Salsberg wrote about the physician shortage in Health Affairs (2015), he reminded us that a critical factor is the supply and availability of clinicians other than physicians (NPs, PAs, midwives) who can make a significant contribution to access and efficiency of health care. He called for NPs and PAs to be fully integrated into the delivery system and to be allowed scope of practice consistent with their education and training.7
Continue to: Both NPs and PAs have become...
Both NPs and PAs have become participants in dialogues on health policy and health care reform. Both professions are spending increasing dollars on national advertising to raise awareness of their critical role in expanding access to primary care for millions of Americans. In fact, Princeton University Professor of Economics Uwe E. Reinhardt told the New York Times, “The doctors are fighting a losing battle. The nurses are like insurgents. They are occasionally beaten back, but they’ll win in the long run. They have economics and common sense on their side.”12 Some suggest that PAs need to fight a similar battle.
So, dear reader, what do you think? Should we be concerned that we are educating too many NPs and PAs? Does that argument become somewhat irrelevant if we can firmly establish a substantial role for ourselves in the future of health care? I would love to hear your thoughts; email me at [email protected].
1. Association of American Medical Colleges. The Complexities of Physician Supply and Demand: Projections from 2016 to 2030—2018 Update. Final Report. March 2018. https://aamc-black.global.ssl.fastly.net/production/media/filer_public/85/d7/85d7b689-f417-4ef0-97fb-ecc129836829/aamc_2018_workforce_projections_update_april_11_2018.pdf
2. Institute of Medicine. Graduate Medical Education That Meets the Nation’s Health Needs. Washington, DC: The National Academies Press; 2014.
3. Cooper RA. New directions for nurse practitioners and physician assistants in the era of physician shortages. Acad Med. 2007;82(9):827-828.
4. American Academy of PAs. What is a PA? Frequently asked questions. www.aapa.org/wp-content/uploads/2018/06/Frequently_Asked_Questions_4.3_FINAL.pdf. Accessed November 14, 2018.
5. American Association of Nurse Practitioners. NP Fact Sheet. www.aanp.org/about/all-about-nps/np-fact-sheet. Accessed November 14, 2018.
6. Accreditation Review Commission on Education for the Physician Assistant. Accredited programs. www.arc-pa.org/accreditation/accredited-programs/. Accessed November 14, 2018.
7. Salsberg E. The nurse practitioner, physician assistant, and pharmacist pipelines: continued growth. Health Affairs. May 26, 2015. www.healthaffairs.org/do/10.1377/hblog20150526.047896/full/. Accessed November 14, 2018.
8. American Association of Nurse Practitioners. Planning your NP education. www.aanp.org/student-resources/planning-your-np-education. Accessed November 14, 2018.
9. United States Department of Labor, Bureau of Labor Statistics. Occupational outlook handbook: healthcare. www.bls.gov/ooh/healthcare/home.htm. Accessed November 14, 2018.
10. Dalen JE. The moratorium on US medical school enrollment, from 1980 to 2005: what were we thinking? Am J Med. 2008;121(2):e1-e2.
11. Dehn RW, Cawley JF. Looking into tomorrow: health workforce issues confronting physician assistants. JAAPA. 2000;13(11):29-32, 35-38, 43-46.
12. Tavernise S. Doctoring, without the doctor. New York Times. May 25, 2015. www.nytimes.com/2015/05/26/health/rural-nebraska-offers-stark-view-of-nursing-autonomy-debate.html. Accessed November 14, 2018.
1. Association of American Medical Colleges. The Complexities of Physician Supply and Demand: Projections from 2016 to 2030—2018 Update. Final Report. March 2018. https://aamc-black.global.ssl.fastly.net/production/media/filer_public/85/d7/85d7b689-f417-4ef0-97fb-ecc129836829/aamc_2018_workforce_projections_update_april_11_2018.pdf
2. Institute of Medicine. Graduate Medical Education That Meets the Nation’s Health Needs. Washington, DC: The National Academies Press; 2014.
3. Cooper RA. New directions for nurse practitioners and physician assistants in the era of physician shortages. Acad Med. 2007;82(9):827-828.
4. American Academy of PAs. What is a PA? Frequently asked questions. www.aapa.org/wp-content/uploads/2018/06/Frequently_Asked_Questions_4.3_FINAL.pdf. Accessed November 14, 2018.
5. American Association of Nurse Practitioners. NP Fact Sheet. www.aanp.org/about/all-about-nps/np-fact-sheet. Accessed November 14, 2018.
6. Accreditation Review Commission on Education for the Physician Assistant. Accredited programs. www.arc-pa.org/accreditation/accredited-programs/. Accessed November 14, 2018.
7. Salsberg E. The nurse practitioner, physician assistant, and pharmacist pipelines: continued growth. Health Affairs. May 26, 2015. www.healthaffairs.org/do/10.1377/hblog20150526.047896/full/. Accessed November 14, 2018.
8. American Association of Nurse Practitioners. Planning your NP education. www.aanp.org/student-resources/planning-your-np-education. Accessed November 14, 2018.
9. United States Department of Labor, Bureau of Labor Statistics. Occupational outlook handbook: healthcare. www.bls.gov/ooh/healthcare/home.htm. Accessed November 14, 2018.
10. Dalen JE. The moratorium on US medical school enrollment, from 1980 to 2005: what were we thinking? Am J Med. 2008;121(2):e1-e2.
11. Dehn RW, Cawley JF. Looking into tomorrow: health workforce issues confronting physician assistants. JAAPA. 2000;13(11):29-32, 35-38, 43-46.
12. Tavernise S. Doctoring, without the doctor. New York Times. May 25, 2015. www.nytimes.com/2015/05/26/health/rural-nebraska-offers-stark-view-of-nursing-autonomy-debate.html. Accessed November 14, 2018.
HIV prevention: Mandating insurance coverage of PrEP
Pre-exposure prophylaxis (PrEP) for HIV is valuable enough for the federal government to mandate insurance coverage, a group of experts told the personal finance website WalletHub, but individuals who are at risk for infection may be missing out for other reasons.
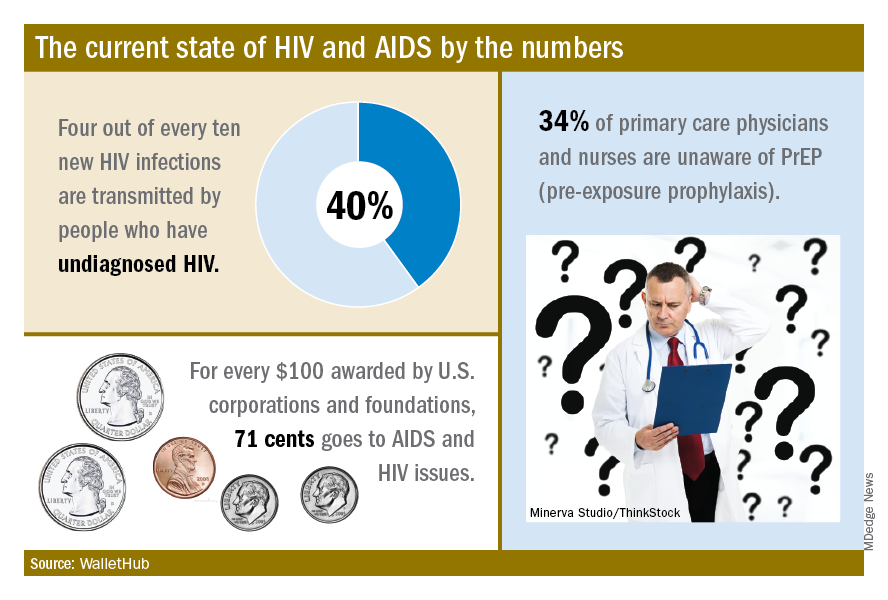
The effectiveness of PrEP is clear, those experts said, but 34% of primary care physicians and nurses in the United States are unaware of the preventive regimen, according to the WalletHub report, which also noted that the majority of Americans with AIDS (61%) are not seeing a specialist.
“Even among [men who have sex with men] in the U.S., coverage is only about 10%, which is abysmal. We can and need to do better. If we don’t pay now, we’ll pay later,” Steffanie Strathdee, PhD, associate dean of global health sciences and Harold Simon Professor at the University of California, San Diego, told WalletHub.
Those taking PrEP have a 90% chance of avoiding HIV infection, the report noted.
“Making PrEP available to all is a giant step forward in the fight against HIV. Mandating this critical prevention be covered by all insurance plans makes it part of mainstream medicine and will only increase its use and help prevent HIV acquisition in exposed populations. I can’t think of other low-risk, high-reward prophylaxis for a lifelong disease,” said Sharon Nachman, MD, professor of pediatrics and associate dean for research at the State University of New York at Stony Brook.
To get PrEP covered, the U.S. Preventive Services Task Force needs to act, explained Gerald M. Oppenheimer, PhD, MPH, of the department of health policy and management at the City University of New York.
“Under the Affordable Care Act, if the [USPSTF] finds that PrEP serves as an effective prevention to disease and gives it a grade of A or B, all insurers must offer it free. That, of course, may lead to an increase in premiums. This is another example of pharmaceutical companies charging high prices in the U.S., compared to what other countries pay, and cries out for an amendment to Medicare Part D, allowing the federal government to negotiate lower drug prices,” he said.
Pre-exposure prophylaxis (PrEP) for HIV is valuable enough for the federal government to mandate insurance coverage, a group of experts told the personal finance website WalletHub, but individuals who are at risk for infection may be missing out for other reasons.

The effectiveness of PrEP is clear, those experts said, but 34% of primary care physicians and nurses in the United States are unaware of the preventive regimen, according to the WalletHub report, which also noted that the majority of Americans with AIDS (61%) are not seeing a specialist.
“Even among [men who have sex with men] in the U.S., coverage is only about 10%, which is abysmal. We can and need to do better. If we don’t pay now, we’ll pay later,” Steffanie Strathdee, PhD, associate dean of global health sciences and Harold Simon Professor at the University of California, San Diego, told WalletHub.
Those taking PrEP have a 90% chance of avoiding HIV infection, the report noted.
“Making PrEP available to all is a giant step forward in the fight against HIV. Mandating this critical prevention be covered by all insurance plans makes it part of mainstream medicine and will only increase its use and help prevent HIV acquisition in exposed populations. I can’t think of other low-risk, high-reward prophylaxis for a lifelong disease,” said Sharon Nachman, MD, professor of pediatrics and associate dean for research at the State University of New York at Stony Brook.
To get PrEP covered, the U.S. Preventive Services Task Force needs to act, explained Gerald M. Oppenheimer, PhD, MPH, of the department of health policy and management at the City University of New York.
“Under the Affordable Care Act, if the [USPSTF] finds that PrEP serves as an effective prevention to disease and gives it a grade of A or B, all insurers must offer it free. That, of course, may lead to an increase in premiums. This is another example of pharmaceutical companies charging high prices in the U.S., compared to what other countries pay, and cries out for an amendment to Medicare Part D, allowing the federal government to negotiate lower drug prices,” he said.
Pre-exposure prophylaxis (PrEP) for HIV is valuable enough for the federal government to mandate insurance coverage, a group of experts told the personal finance website WalletHub, but individuals who are at risk for infection may be missing out for other reasons.

The effectiveness of PrEP is clear, those experts said, but 34% of primary care physicians and nurses in the United States are unaware of the preventive regimen, according to the WalletHub report, which also noted that the majority of Americans with AIDS (61%) are not seeing a specialist.
“Even among [men who have sex with men] in the U.S., coverage is only about 10%, which is abysmal. We can and need to do better. If we don’t pay now, we’ll pay later,” Steffanie Strathdee, PhD, associate dean of global health sciences and Harold Simon Professor at the University of California, San Diego, told WalletHub.
Those taking PrEP have a 90% chance of avoiding HIV infection, the report noted.
“Making PrEP available to all is a giant step forward in the fight against HIV. Mandating this critical prevention be covered by all insurance plans makes it part of mainstream medicine and will only increase its use and help prevent HIV acquisition in exposed populations. I can’t think of other low-risk, high-reward prophylaxis for a lifelong disease,” said Sharon Nachman, MD, professor of pediatrics and associate dean for research at the State University of New York at Stony Brook.
To get PrEP covered, the U.S. Preventive Services Task Force needs to act, explained Gerald M. Oppenheimer, PhD, MPH, of the department of health policy and management at the City University of New York.
“Under the Affordable Care Act, if the [USPSTF] finds that PrEP serves as an effective prevention to disease and gives it a grade of A or B, all insurers must offer it free. That, of course, may lead to an increase in premiums. This is another example of pharmaceutical companies charging high prices in the U.S., compared to what other countries pay, and cries out for an amendment to Medicare Part D, allowing the federal government to negotiate lower drug prices,” he said.
Open enrollment: Weekly volume down again
Plan selections at HealthCare.gov fell for the second week in a row as overall volume for open enrollment 2019 continues to lag behind last year, according to the Centers for Medicare & Medicaid Services.
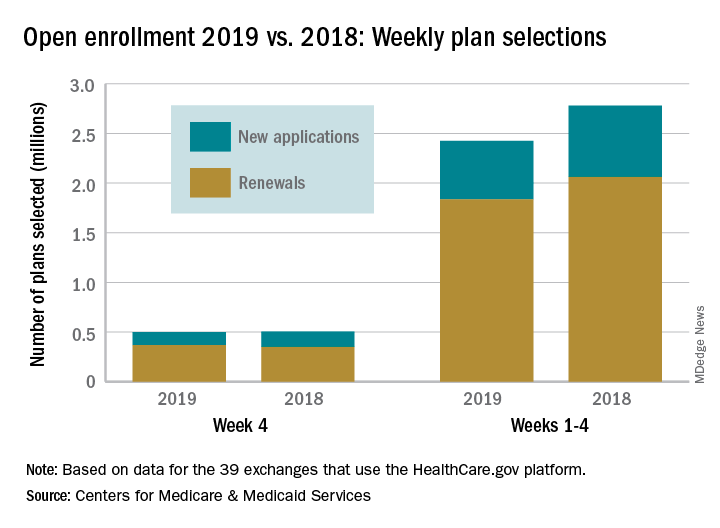
Just over 500,000 plans – 369,000 renewals and 131,000 new applications – were selected during week 4 (Nov. 18-24) for the 2019 coverage year in the 39 states that use the HealthCare.gov platform, which is down from 748,000 for week 3 and 805,000 for week 2. A similar pattern of decreases in weeks 3 and 4 was seen during last year’s open-enrollment period.
For the entire open enrollment so far this year, a little over 2.42 million plans have been selected, which is down by 12.8% from last year’s 4-week total of 2.78 million selections, the CMS data show.
Plan selections at HealthCare.gov fell for the second week in a row as overall volume for open enrollment 2019 continues to lag behind last year, according to the Centers for Medicare & Medicaid Services.

Just over 500,000 plans – 369,000 renewals and 131,000 new applications – were selected during week 4 (Nov. 18-24) for the 2019 coverage year in the 39 states that use the HealthCare.gov platform, which is down from 748,000 for week 3 and 805,000 for week 2. A similar pattern of decreases in weeks 3 and 4 was seen during last year’s open-enrollment period.
For the entire open enrollment so far this year, a little over 2.42 million plans have been selected, which is down by 12.8% from last year’s 4-week total of 2.78 million selections, the CMS data show.
Plan selections at HealthCare.gov fell for the second week in a row as overall volume for open enrollment 2019 continues to lag behind last year, according to the Centers for Medicare & Medicaid Services.

Just over 500,000 plans – 369,000 renewals and 131,000 new applications – were selected during week 4 (Nov. 18-24) for the 2019 coverage year in the 39 states that use the HealthCare.gov platform, which is down from 748,000 for week 3 and 805,000 for week 2. A similar pattern of decreases in weeks 3 and 4 was seen during last year’s open-enrollment period.
For the entire open enrollment so far this year, a little over 2.42 million plans have been selected, which is down by 12.8% from last year’s 4-week total of 2.78 million selections, the CMS data show.
Medical liability at sea
Question: Regarding medical care aboard cruise ships, which of the following is incorrect?
A. It is difficult to prove negligence because of jurisdictional issues.
B. The American College of Emergency Physicians has published practice guidelines.
C. Cruise line owners are immune from liability under the Barbetta rule.
D. The Franza decision may be a game changer.
E. Lawsuits are on the increase.
Answer: A. More and more people are cruising, with the number of passengers on North American lines reaching nearly 18 million in 2017, about one-third out of Florida. Medical illnesses and accidents predictably occur at sea, and unfortunately, substandard and negligent care occasionally follows some of these mishaps.
However, up until recently, courts have immunized cruise line owners from legal liability by relying on the so-called Barbetta rule, which is based on historical notions of limited resources at sea and the impossibility of exerting control over the conduct of a ship’s health care providers.
A 2007 Florida case is illustrative. The doctor aboard a Carnival cruise ship failed to diagnose acute appendicitis in a 14-year-old girl who had complained of several days of abdominal symptoms. As a result, the patient ruptured her appendix, and this eventually resulted in sterility. The parents sued the cruise line, which denied liability because the doctor was not an employee, a fact specifically disclosed in the cruise ticket.
Although the doctor’s contract stated that he was an independent contractor, the District Court of Appeal of Florida reasoned that, in a claim based on agency, it is the right of control rather than actual control itself that matters.
It therefore held that: “for purposes of fulfilling cruise line’s duty to exercise reasonable care, the ship’s doctor is an agent of cruise line whose negligence should be imputed to cruise line ... regardless of the contractual status ascribed to the doctor” and to the extent cruise ticket sought to limit cruise line’s liability for negligence of doctor, it was invalid.
However, the Florida Supreme Court quashed this decision, because federal maritime law has historically protected shipowners from liability flowing from the medical negligence of shipboard physicians.1
The Barbetta rule was named after a 1988 case in which Florence L. Barbetta suffered serious medical complications during a Mexican cruise out of Florida. The ship’s doctor was alleged to have been negligent for his failure to diagnose diabetes.2 The lower court dismissed the case, and the appellate court affirmed.
The appellate court noted that an impressive number of courts from many jurisdictions have, for almost 100 years, followed the same basic rule: If the doctor is negligent in treating a passenger, that negligence will not be imputed to the carrier unless the carrier itself was negligent in hiring the doctor.
Citing approvingly from another case, the appellate court noted: “[A] shipping company is not in the business of providing medical services to passengers; it does not possess the expertise requisite to supervise a physician or surgeon carried on board a ship as a convenience to passengers. A ship is not a floating hospital; a ship’s physician is an independent medical expert engaged on the basis of his professional qualifications and carried on board a ship for the convenience of passengers, who are free to contract with him for any medical services they may require.”
However, courts now appear ready to jettison this rule.
In the 2014 case of Franza v. Royal Caribbean Cruises, Ltd., a passenger fell and hit his head while his ship was at port in Bermuda.3 He died several days later, allegedly because of negligence and delay by the ship’s medical staff. The lower courts barred the lawsuit, based on Barbetta, but on appeal, the 11th Circuit Court reversed. It rejected the historical justifications for immunizing shipowners from liability, concluding that past reasons were no longer applicable to modern-day cruise ships.
The court wrote: “Here, the roots of the Barbetta rule snake back to a wholly different world. Instead of 19th-century steamships, we now confront state-of-the-art cruise ships that house thousands of people and operate as floating cities complete with well-stocked modern infirmaries and urgent-care centers. In place of independent doctors and nurses, we must now acknowledge that medical professionals routinely work for corporate masters. And whereas ships historically went ‘off the grid’ when they set sail, modern technology enables distant ships to communicate instantaneously with the mainland in meaningful ways.”
However, the injured person must still prove that the doctor or nurse was acting as a ship employee rather than an independent contractor. Some of the factors to be considered include whether the cruise line advertised its medical center or medical staff to passengers, whether it retained the right to hire and fire medical staff, and methods of payment.
This 2014 11th Circuit Court ruling has particular impact on Florida, because the state’s federal court system falls within its jurisdiction, and many cruise lines – such as Carnival, Celebrity, Disney, Norwegian, Royal Caribbean, and Silversea Cruises – are headquartered in Florida. Because most ocean liners hire medical professionals who are foreign nationals, seeking legal remedy against the individuals can be difficult.
As a result, suing the cruise line directly can serve as the simplest way to obtain compensation. Some major cruise lines have already seen an increase in personal injury lawsuits. According to Bloomberg Law, there were 164 such federal injury suits in 2016, 188 in 2017, and 83 cases in just the first 3 months of 2018.
The popular press recently highlighted the latest example of medical malpractice at sea. A cruise worker was awarded $3.34 million after a young, inexperienced doctor prescribed a large dose of intravenous promethazine for nausea. Instead of using 6.25 mg, the usual dose, he prescribed 25 mg, which was inadvertently injected into the ulnar artery. The caustic drug, known to injure vascular walls, caused severe damage with extravasation, and tissue swelling, ending in compartment syndrome.
Furthermore, he had to wait 24 hours before arriving in port for treatment. By then, his arm was gangrenous and had to be amputated. The defense had argued unsuccessfully that the plaintiff had a venous anomaly to account for the injury.4
These changes in the law augur well for the cruising public. The Franza decision puts cruise line owners on notice that they no longer enjoy blanket immunity, and will be held responsible for the negligence of their health care providers. Hopefully, this will ensure a more uniform and adequate standard of care.
The American College of Emergency Physicians has published specific guidelines regarding medical standards aboard cruise ships. In addition to having established medical policies and procedures, and a dedicated medical emergency telephone number, they must have at least one doctor available 24/7 to provide emergency medical care and maintain certain equipment on board, such as pulse oximeters, cardiac monitors, defibrillators, an EKG device, as well as a laboratory.
Norwegian Cruise Line has taken a step further: It recently installed the capability to consult in real time with the Cleveland Clinic for diagnostic and treatment advice.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Carlisle v. Carnival Corporation, et al., 953 So.2d 461, 2007.
2. Barbetta v. S/S Bermuda Star, 848 F.2d 1364 (5th Cir. 1988).
3. Franza v. Royal Caribbean Cruises Ltd., 772 F.3d 1225 (11th Cir. 2014).
4. Loncar v. NCL Bahamas, International Center for Dispute Resolution, Case # 01-16-0004-3640, June 13, 2018.
Question: Regarding medical care aboard cruise ships, which of the following is incorrect?
A. It is difficult to prove negligence because of jurisdictional issues.
B. The American College of Emergency Physicians has published practice guidelines.
C. Cruise line owners are immune from liability under the Barbetta rule.
D. The Franza decision may be a game changer.
E. Lawsuits are on the increase.
Answer: A. More and more people are cruising, with the number of passengers on North American lines reaching nearly 18 million in 2017, about one-third out of Florida. Medical illnesses and accidents predictably occur at sea, and unfortunately, substandard and negligent care occasionally follows some of these mishaps.
However, up until recently, courts have immunized cruise line owners from legal liability by relying on the so-called Barbetta rule, which is based on historical notions of limited resources at sea and the impossibility of exerting control over the conduct of a ship’s health care providers.
A 2007 Florida case is illustrative. The doctor aboard a Carnival cruise ship failed to diagnose acute appendicitis in a 14-year-old girl who had complained of several days of abdominal symptoms. As a result, the patient ruptured her appendix, and this eventually resulted in sterility. The parents sued the cruise line, which denied liability because the doctor was not an employee, a fact specifically disclosed in the cruise ticket.
Although the doctor’s contract stated that he was an independent contractor, the District Court of Appeal of Florida reasoned that, in a claim based on agency, it is the right of control rather than actual control itself that matters.
It therefore held that: “for purposes of fulfilling cruise line’s duty to exercise reasonable care, the ship’s doctor is an agent of cruise line whose negligence should be imputed to cruise line ... regardless of the contractual status ascribed to the doctor” and to the extent cruise ticket sought to limit cruise line’s liability for negligence of doctor, it was invalid.
However, the Florida Supreme Court quashed this decision, because federal maritime law has historically protected shipowners from liability flowing from the medical negligence of shipboard physicians.1
The Barbetta rule was named after a 1988 case in which Florence L. Barbetta suffered serious medical complications during a Mexican cruise out of Florida. The ship’s doctor was alleged to have been negligent for his failure to diagnose diabetes.2 The lower court dismissed the case, and the appellate court affirmed.
The appellate court noted that an impressive number of courts from many jurisdictions have, for almost 100 years, followed the same basic rule: If the doctor is negligent in treating a passenger, that negligence will not be imputed to the carrier unless the carrier itself was negligent in hiring the doctor.
Citing approvingly from another case, the appellate court noted: “[A] shipping company is not in the business of providing medical services to passengers; it does not possess the expertise requisite to supervise a physician or surgeon carried on board a ship as a convenience to passengers. A ship is not a floating hospital; a ship’s physician is an independent medical expert engaged on the basis of his professional qualifications and carried on board a ship for the convenience of passengers, who are free to contract with him for any medical services they may require.”
However, courts now appear ready to jettison this rule.
In the 2014 case of Franza v. Royal Caribbean Cruises, Ltd., a passenger fell and hit his head while his ship was at port in Bermuda.3 He died several days later, allegedly because of negligence and delay by the ship’s medical staff. The lower courts barred the lawsuit, based on Barbetta, but on appeal, the 11th Circuit Court reversed. It rejected the historical justifications for immunizing shipowners from liability, concluding that past reasons were no longer applicable to modern-day cruise ships.
The court wrote: “Here, the roots of the Barbetta rule snake back to a wholly different world. Instead of 19th-century steamships, we now confront state-of-the-art cruise ships that house thousands of people and operate as floating cities complete with well-stocked modern infirmaries and urgent-care centers. In place of independent doctors and nurses, we must now acknowledge that medical professionals routinely work for corporate masters. And whereas ships historically went ‘off the grid’ when they set sail, modern technology enables distant ships to communicate instantaneously with the mainland in meaningful ways.”
However, the injured person must still prove that the doctor or nurse was acting as a ship employee rather than an independent contractor. Some of the factors to be considered include whether the cruise line advertised its medical center or medical staff to passengers, whether it retained the right to hire and fire medical staff, and methods of payment.
This 2014 11th Circuit Court ruling has particular impact on Florida, because the state’s federal court system falls within its jurisdiction, and many cruise lines – such as Carnival, Celebrity, Disney, Norwegian, Royal Caribbean, and Silversea Cruises – are headquartered in Florida. Because most ocean liners hire medical professionals who are foreign nationals, seeking legal remedy against the individuals can be difficult.
As a result, suing the cruise line directly can serve as the simplest way to obtain compensation. Some major cruise lines have already seen an increase in personal injury lawsuits. According to Bloomberg Law, there were 164 such federal injury suits in 2016, 188 in 2017, and 83 cases in just the first 3 months of 2018.
The popular press recently highlighted the latest example of medical malpractice at sea. A cruise worker was awarded $3.34 million after a young, inexperienced doctor prescribed a large dose of intravenous promethazine for nausea. Instead of using 6.25 mg, the usual dose, he prescribed 25 mg, which was inadvertently injected into the ulnar artery. The caustic drug, known to injure vascular walls, caused severe damage with extravasation, and tissue swelling, ending in compartment syndrome.
Furthermore, he had to wait 24 hours before arriving in port for treatment. By then, his arm was gangrenous and had to be amputated. The defense had argued unsuccessfully that the plaintiff had a venous anomaly to account for the injury.4
These changes in the law augur well for the cruising public. The Franza decision puts cruise line owners on notice that they no longer enjoy blanket immunity, and will be held responsible for the negligence of their health care providers. Hopefully, this will ensure a more uniform and adequate standard of care.
The American College of Emergency Physicians has published specific guidelines regarding medical standards aboard cruise ships. In addition to having established medical policies and procedures, and a dedicated medical emergency telephone number, they must have at least one doctor available 24/7 to provide emergency medical care and maintain certain equipment on board, such as pulse oximeters, cardiac monitors, defibrillators, an EKG device, as well as a laboratory.
Norwegian Cruise Line has taken a step further: It recently installed the capability to consult in real time with the Cleveland Clinic for diagnostic and treatment advice.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Carlisle v. Carnival Corporation, et al., 953 So.2d 461, 2007.
2. Barbetta v. S/S Bermuda Star, 848 F.2d 1364 (5th Cir. 1988).
3. Franza v. Royal Caribbean Cruises Ltd., 772 F.3d 1225 (11th Cir. 2014).
4. Loncar v. NCL Bahamas, International Center for Dispute Resolution, Case # 01-16-0004-3640, June 13, 2018.
Question: Regarding medical care aboard cruise ships, which of the following is incorrect?
A. It is difficult to prove negligence because of jurisdictional issues.
B. The American College of Emergency Physicians has published practice guidelines.
C. Cruise line owners are immune from liability under the Barbetta rule.
D. The Franza decision may be a game changer.
E. Lawsuits are on the increase.
Answer: A. More and more people are cruising, with the number of passengers on North American lines reaching nearly 18 million in 2017, about one-third out of Florida. Medical illnesses and accidents predictably occur at sea, and unfortunately, substandard and negligent care occasionally follows some of these mishaps.
However, up until recently, courts have immunized cruise line owners from legal liability by relying on the so-called Barbetta rule, which is based on historical notions of limited resources at sea and the impossibility of exerting control over the conduct of a ship’s health care providers.
A 2007 Florida case is illustrative. The doctor aboard a Carnival cruise ship failed to diagnose acute appendicitis in a 14-year-old girl who had complained of several days of abdominal symptoms. As a result, the patient ruptured her appendix, and this eventually resulted in sterility. The parents sued the cruise line, which denied liability because the doctor was not an employee, a fact specifically disclosed in the cruise ticket.
Although the doctor’s contract stated that he was an independent contractor, the District Court of Appeal of Florida reasoned that, in a claim based on agency, it is the right of control rather than actual control itself that matters.
It therefore held that: “for purposes of fulfilling cruise line’s duty to exercise reasonable care, the ship’s doctor is an agent of cruise line whose negligence should be imputed to cruise line ... regardless of the contractual status ascribed to the doctor” and to the extent cruise ticket sought to limit cruise line’s liability for negligence of doctor, it was invalid.
However, the Florida Supreme Court quashed this decision, because federal maritime law has historically protected shipowners from liability flowing from the medical negligence of shipboard physicians.1
The Barbetta rule was named after a 1988 case in which Florence L. Barbetta suffered serious medical complications during a Mexican cruise out of Florida. The ship’s doctor was alleged to have been negligent for his failure to diagnose diabetes.2 The lower court dismissed the case, and the appellate court affirmed.
The appellate court noted that an impressive number of courts from many jurisdictions have, for almost 100 years, followed the same basic rule: If the doctor is negligent in treating a passenger, that negligence will not be imputed to the carrier unless the carrier itself was negligent in hiring the doctor.
Citing approvingly from another case, the appellate court noted: “[A] shipping company is not in the business of providing medical services to passengers; it does not possess the expertise requisite to supervise a physician or surgeon carried on board a ship as a convenience to passengers. A ship is not a floating hospital; a ship’s physician is an independent medical expert engaged on the basis of his professional qualifications and carried on board a ship for the convenience of passengers, who are free to contract with him for any medical services they may require.”
However, courts now appear ready to jettison this rule.
In the 2014 case of Franza v. Royal Caribbean Cruises, Ltd., a passenger fell and hit his head while his ship was at port in Bermuda.3 He died several days later, allegedly because of negligence and delay by the ship’s medical staff. The lower courts barred the lawsuit, based on Barbetta, but on appeal, the 11th Circuit Court reversed. It rejected the historical justifications for immunizing shipowners from liability, concluding that past reasons were no longer applicable to modern-day cruise ships.
The court wrote: “Here, the roots of the Barbetta rule snake back to a wholly different world. Instead of 19th-century steamships, we now confront state-of-the-art cruise ships that house thousands of people and operate as floating cities complete with well-stocked modern infirmaries and urgent-care centers. In place of independent doctors and nurses, we must now acknowledge that medical professionals routinely work for corporate masters. And whereas ships historically went ‘off the grid’ when they set sail, modern technology enables distant ships to communicate instantaneously with the mainland in meaningful ways.”
However, the injured person must still prove that the doctor or nurse was acting as a ship employee rather than an independent contractor. Some of the factors to be considered include whether the cruise line advertised its medical center or medical staff to passengers, whether it retained the right to hire and fire medical staff, and methods of payment.
This 2014 11th Circuit Court ruling has particular impact on Florida, because the state’s federal court system falls within its jurisdiction, and many cruise lines – such as Carnival, Celebrity, Disney, Norwegian, Royal Caribbean, and Silversea Cruises – are headquartered in Florida. Because most ocean liners hire medical professionals who are foreign nationals, seeking legal remedy against the individuals can be difficult.
As a result, suing the cruise line directly can serve as the simplest way to obtain compensation. Some major cruise lines have already seen an increase in personal injury lawsuits. According to Bloomberg Law, there were 164 such federal injury suits in 2016, 188 in 2017, and 83 cases in just the first 3 months of 2018.
The popular press recently highlighted the latest example of medical malpractice at sea. A cruise worker was awarded $3.34 million after a young, inexperienced doctor prescribed a large dose of intravenous promethazine for nausea. Instead of using 6.25 mg, the usual dose, he prescribed 25 mg, which was inadvertently injected into the ulnar artery. The caustic drug, known to injure vascular walls, caused severe damage with extravasation, and tissue swelling, ending in compartment syndrome.
Furthermore, he had to wait 24 hours before arriving in port for treatment. By then, his arm was gangrenous and had to be amputated. The defense had argued unsuccessfully that the plaintiff had a venous anomaly to account for the injury.4
These changes in the law augur well for the cruising public. The Franza decision puts cruise line owners on notice that they no longer enjoy blanket immunity, and will be held responsible for the negligence of their health care providers. Hopefully, this will ensure a more uniform and adequate standard of care.
The American College of Emergency Physicians has published specific guidelines regarding medical standards aboard cruise ships. In addition to having established medical policies and procedures, and a dedicated medical emergency telephone number, they must have at least one doctor available 24/7 to provide emergency medical care and maintain certain equipment on board, such as pulse oximeters, cardiac monitors, defibrillators, an EKG device, as well as a laboratory.
Norwegian Cruise Line has taken a step further: It recently installed the capability to consult in real time with the Cleveland Clinic for diagnostic and treatment advice.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Carlisle v. Carnival Corporation, et al., 953 So.2d 461, 2007.
2. Barbetta v. S/S Bermuda Star, 848 F.2d 1364 (5th Cir. 1988).
3. Franza v. Royal Caribbean Cruises Ltd., 772 F.3d 1225 (11th Cir. 2014).
4. Loncar v. NCL Bahamas, International Center for Dispute Resolution, Case # 01-16-0004-3640, June 13, 2018.