User login
Literature review: Cryolipolysis safe, effective for reducing submental fat
Cryolipolysis is a safe and effective noninvasive treatment for reducing submental fat, according to a review of several published studies.
In a review of four clinical trials and one case series, which involved a total of 101 patients, , reported Shari R. Lipner, MD, of the department of dermatology at Cornell University, New York.
In 2015, the Food and Drug Administration cleared a cryolipolysis device for use in the submental area.
The literature review was performed in May 2017 using Pubmed, EMBASE, Web of Science, and CINAHL databases, searching for the terms cryolipolysis, submental, and paradoxical adipose hyperplasia. Non-English studies were excluded.
The studies included an open-label prospective multicenter trial of 60 patients who underwent cryolipolysis for submental fat reduction once or twice at –10°C for 60 minutes, which found that in 58 evaluable patients, blinded independent reviewers correctly identified baseline photos in 91.4% of cases (P less than .0001). Ultrasound, used to evaluate 57 patients, determined the mean fat layer reduction was 2.0 mm or 20% (ranging from an increase of 2.0 mm to a reduction of –5.9 mm; P less than .0001). Side effects included erythema, edema, bruising, and numbness, which resolved by week 12. Additionally, 83% of the 60 patients were satisfied with the results.
In another study, a prospective nonrandomized study that evaluated overlapping cryolipolysis applications on different visits in 14 patients, pretreatment and post-treatment photographs were correctly identified by blinded independent reviewers 81% of the time (95% confidence interval, 65.9%-91.4%; P = .02). In addition, the mean fat layer reduction, as measured with skin-fold calipers, was 2.3 mm (95% CI, 1.9-2.7 mm; P less than .001), and 93% of the patients were satisfied with the results.
A prospective nonrandomized single-center open-label study of 15 Hispanic patients evaluated cryolipolysis applied to the submental area at two different temperatures (–12°C for 45 minutes and –15°C for 30 minutes) with treatments given 10 weeks apart. The mean reduction in submental fat, as measured by calipers, was 33%, with no significant difference between the two. Blinded physicians correctly identified pre- and post- treatment photos in 60% of cases, and 80% of the patients said they “were satisfied or very satisfied” with the results, according to Dr. Lipner’s review, published online in the Journal of Cosmetic Dermatology.
All studies reported adverse side effects, most commonly erythema, which resolved within weeks of treatment, she wrote. To date, she noted, no cases of mandibular nerve injury or paradoxical adipose hyperplasia have been reported after cryolipolysis for submental fat.
Dr. Lipner referred to early trials in humans found that cryolipolysis was safe and effective for treatment of the back, arms, and chest. Additional trials found no significant changes in lipids or liver function at 1, 4, and 12 weeks’ follow-up when patients were treated for fat in the flanks and lower abdomen.
The results of the literature review suggest that cryolipolysis is safe and effective for submental fat, Dr. Lipner wrote. Appropriate patient selection is important and “patients should be counseled on clinical improvement in the submental contour, number of sessions necessary, side effects, downtime, and cost,” she noted. Liposuction is still the gold standard for removal of large fat deposits, and although cryolipolysis can reduce submental fat, “it may also worsen the appearance on the neck by making platysmal banding or skin imperfections more obvious,” she added.
Dr. Lipner did not report any relevant disclosures. No funding source was provided.
SOURCE: J Cosmet Dermatol. 2018 Jan 17. doi: 10.1111/jocd.12495.
Cryolipolysis is a safe and effective noninvasive treatment for reducing submental fat, according to a review of several published studies.
In a review of four clinical trials and one case series, which involved a total of 101 patients, , reported Shari R. Lipner, MD, of the department of dermatology at Cornell University, New York.
In 2015, the Food and Drug Administration cleared a cryolipolysis device for use in the submental area.
The literature review was performed in May 2017 using Pubmed, EMBASE, Web of Science, and CINAHL databases, searching for the terms cryolipolysis, submental, and paradoxical adipose hyperplasia. Non-English studies were excluded.
The studies included an open-label prospective multicenter trial of 60 patients who underwent cryolipolysis for submental fat reduction once or twice at –10°C for 60 minutes, which found that in 58 evaluable patients, blinded independent reviewers correctly identified baseline photos in 91.4% of cases (P less than .0001). Ultrasound, used to evaluate 57 patients, determined the mean fat layer reduction was 2.0 mm or 20% (ranging from an increase of 2.0 mm to a reduction of –5.9 mm; P less than .0001). Side effects included erythema, edema, bruising, and numbness, which resolved by week 12. Additionally, 83% of the 60 patients were satisfied with the results.
In another study, a prospective nonrandomized study that evaluated overlapping cryolipolysis applications on different visits in 14 patients, pretreatment and post-treatment photographs were correctly identified by blinded independent reviewers 81% of the time (95% confidence interval, 65.9%-91.4%; P = .02). In addition, the mean fat layer reduction, as measured with skin-fold calipers, was 2.3 mm (95% CI, 1.9-2.7 mm; P less than .001), and 93% of the patients were satisfied with the results.
A prospective nonrandomized single-center open-label study of 15 Hispanic patients evaluated cryolipolysis applied to the submental area at two different temperatures (–12°C for 45 minutes and –15°C for 30 minutes) with treatments given 10 weeks apart. The mean reduction in submental fat, as measured by calipers, was 33%, with no significant difference between the two. Blinded physicians correctly identified pre- and post- treatment photos in 60% of cases, and 80% of the patients said they “were satisfied or very satisfied” with the results, according to Dr. Lipner’s review, published online in the Journal of Cosmetic Dermatology.
All studies reported adverse side effects, most commonly erythema, which resolved within weeks of treatment, she wrote. To date, she noted, no cases of mandibular nerve injury or paradoxical adipose hyperplasia have been reported after cryolipolysis for submental fat.
Dr. Lipner referred to early trials in humans found that cryolipolysis was safe and effective for treatment of the back, arms, and chest. Additional trials found no significant changes in lipids or liver function at 1, 4, and 12 weeks’ follow-up when patients were treated for fat in the flanks and lower abdomen.
The results of the literature review suggest that cryolipolysis is safe and effective for submental fat, Dr. Lipner wrote. Appropriate patient selection is important and “patients should be counseled on clinical improvement in the submental contour, number of sessions necessary, side effects, downtime, and cost,” she noted. Liposuction is still the gold standard for removal of large fat deposits, and although cryolipolysis can reduce submental fat, “it may also worsen the appearance on the neck by making platysmal banding or skin imperfections more obvious,” she added.
Dr. Lipner did not report any relevant disclosures. No funding source was provided.
SOURCE: J Cosmet Dermatol. 2018 Jan 17. doi: 10.1111/jocd.12495.
Cryolipolysis is a safe and effective noninvasive treatment for reducing submental fat, according to a review of several published studies.
In a review of four clinical trials and one case series, which involved a total of 101 patients, , reported Shari R. Lipner, MD, of the department of dermatology at Cornell University, New York.
In 2015, the Food and Drug Administration cleared a cryolipolysis device for use in the submental area.
The literature review was performed in May 2017 using Pubmed, EMBASE, Web of Science, and CINAHL databases, searching for the terms cryolipolysis, submental, and paradoxical adipose hyperplasia. Non-English studies were excluded.
The studies included an open-label prospective multicenter trial of 60 patients who underwent cryolipolysis for submental fat reduction once or twice at –10°C for 60 minutes, which found that in 58 evaluable patients, blinded independent reviewers correctly identified baseline photos in 91.4% of cases (P less than .0001). Ultrasound, used to evaluate 57 patients, determined the mean fat layer reduction was 2.0 mm or 20% (ranging from an increase of 2.0 mm to a reduction of –5.9 mm; P less than .0001). Side effects included erythema, edema, bruising, and numbness, which resolved by week 12. Additionally, 83% of the 60 patients were satisfied with the results.
In another study, a prospective nonrandomized study that evaluated overlapping cryolipolysis applications on different visits in 14 patients, pretreatment and post-treatment photographs were correctly identified by blinded independent reviewers 81% of the time (95% confidence interval, 65.9%-91.4%; P = .02). In addition, the mean fat layer reduction, as measured with skin-fold calipers, was 2.3 mm (95% CI, 1.9-2.7 mm; P less than .001), and 93% of the patients were satisfied with the results.
A prospective nonrandomized single-center open-label study of 15 Hispanic patients evaluated cryolipolysis applied to the submental area at two different temperatures (–12°C for 45 minutes and –15°C for 30 minutes) with treatments given 10 weeks apart. The mean reduction in submental fat, as measured by calipers, was 33%, with no significant difference between the two. Blinded physicians correctly identified pre- and post- treatment photos in 60% of cases, and 80% of the patients said they “were satisfied or very satisfied” with the results, according to Dr. Lipner’s review, published online in the Journal of Cosmetic Dermatology.
All studies reported adverse side effects, most commonly erythema, which resolved within weeks of treatment, she wrote. To date, she noted, no cases of mandibular nerve injury or paradoxical adipose hyperplasia have been reported after cryolipolysis for submental fat.
Dr. Lipner referred to early trials in humans found that cryolipolysis was safe and effective for treatment of the back, arms, and chest. Additional trials found no significant changes in lipids or liver function at 1, 4, and 12 weeks’ follow-up when patients were treated for fat in the flanks and lower abdomen.
The results of the literature review suggest that cryolipolysis is safe and effective for submental fat, Dr. Lipner wrote. Appropriate patient selection is important and “patients should be counseled on clinical improvement in the submental contour, number of sessions necessary, side effects, downtime, and cost,” she noted. Liposuction is still the gold standard for removal of large fat deposits, and although cryolipolysis can reduce submental fat, “it may also worsen the appearance on the neck by making platysmal banding or skin imperfections more obvious,” she added.
Dr. Lipner did not report any relevant disclosures. No funding source was provided.
SOURCE: J Cosmet Dermatol. 2018 Jan 17. doi: 10.1111/jocd.12495.
FROM THE JOURNAL OF COSMETIC DERMATOLOGY
Key clinical point: Cryolipolysis appears to be a safe, effective noninvasive treatment for submental fat.
Major finding: Endpoints evaluating the effects of the cooling technique on submental fat included evaluations of blinded patient photos by blinded reviewers, who, in one study, correctly identified baseline photos in 91.4% of cases (P less than .0001).
Data source: A literature review of four clinical trials and one case series of a total of 101 patients who underwent cryolipolysis for reducing submental fat.
Disclosures: The author did not report any relevant disclosures. No funding source was provided.
Source: Cosmet Dermatol. 2018 Jan 17. doi: 10.1111/jocd.12495.
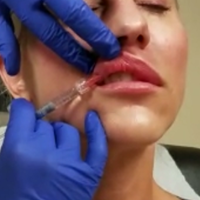
Hyaluronic Acid for Lip Rejuvenation



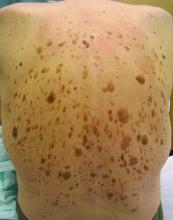
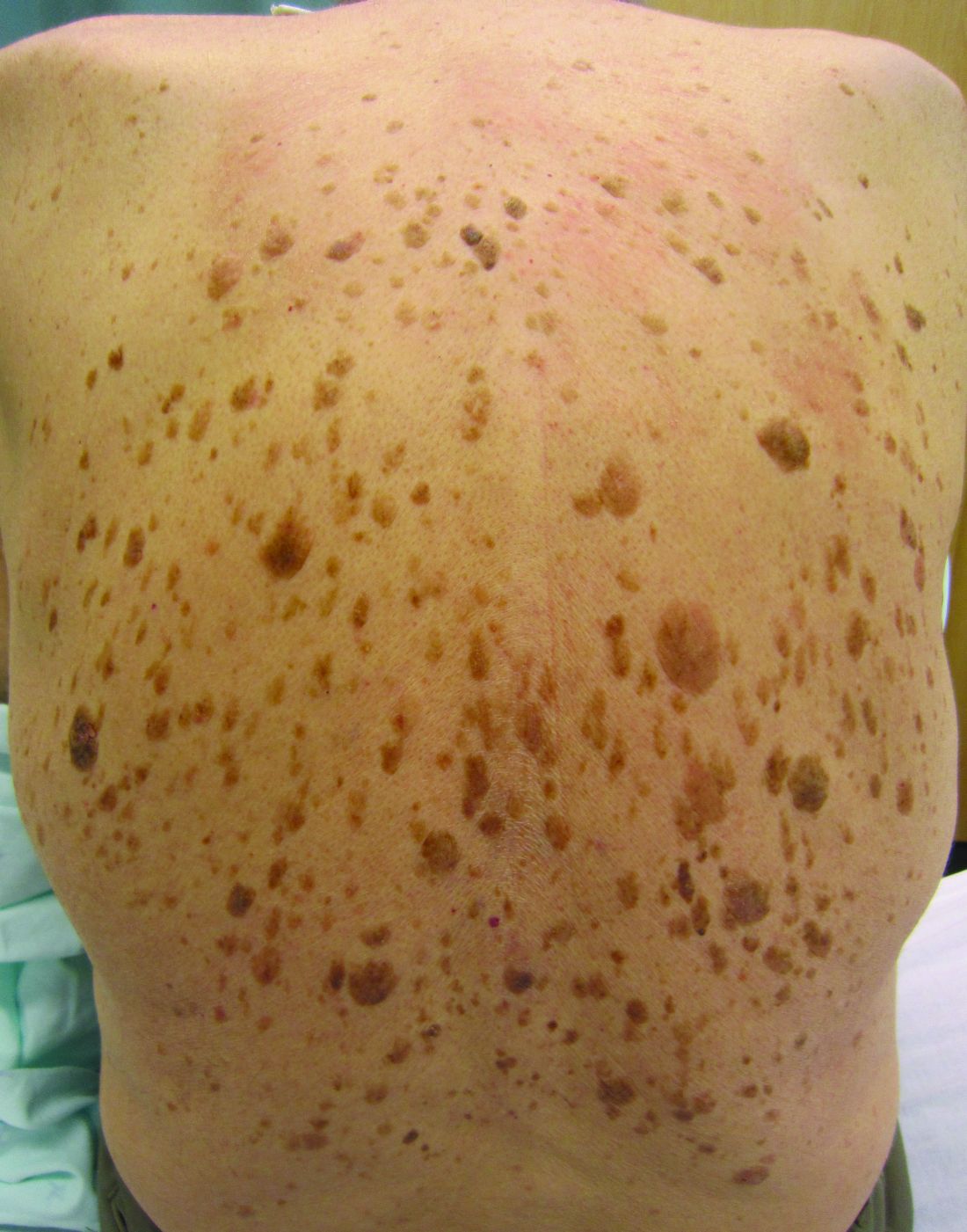
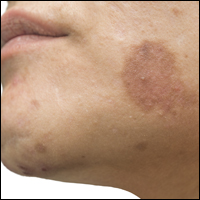
High-dose hydrogen peroxide for SKs
Formerly known as A-101, . It is a 40% hydrogen peroxide topical solution that is applied to raised SKs as an in-office procedure. As previously reported, SKs are composed of hyperadherent senescent cells that are arrested in the G1 phase of the cell cycle. They exhibit decreased apoptotic cell death, compared with normal skin.
Two double-blind vehicle-controlled studies demonstrated that more patients were clear or near clear of SKs after use of the 40% concentration solution than those in the vehicle group, according to the prescribing information. In the studies, patients with four raised SKs of the face, trunk or extremities were treated with Eskata at baseline and 3 weeks later, if necessary, or a vehicle. None of those in the vehicle group were cleared at follow-up (day 106), compared with 4% and 8% of those treated with Eskata, and at least three of the four lesions treated with Eskata had cleared in 13% and 23% at follow-up. Local skin reactions were mostly mild and transient, the most common being itching, stinging, crusting, swelling, redness, and scaling at the application site.
In the clinical studies, the solution was applied up to two times, on day 0 and again 3 weeks later on day 22. The lesion should first be cleansed with alcohol, and appropriate measures should be taken to ensure Eskata does not come in contact with the eyes. Nitrile or vinyl examination gloves should be used for application. Eskata is applied to the SKs with a pen-like applicator. The solution is applied uniformly in a circular motion with excess and the surrounding area patted dry with an absorbent wipe. Cotton gauze, tips, paper towels, or tissue should not be used as organic compounds can react with high concentrations of hydrogen peroxide. In a treatment session, one lesion may be treated up to four times, 1 minute apart. The applicator is used only once and may be discarded after lesions are treated. The packages may be stored at controlled room temperature (68° F to 77° F).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Dr. Wesley has served on an advisory board panel for Aclaris, the manufacturer of Eskata. Dr. Talakoub had no related disclosures. Write to them at [email protected].
Formerly known as A-101, . It is a 40% hydrogen peroxide topical solution that is applied to raised SKs as an in-office procedure. As previously reported, SKs are composed of hyperadherent senescent cells that are arrested in the G1 phase of the cell cycle. They exhibit decreased apoptotic cell death, compared with normal skin.
Two double-blind vehicle-controlled studies demonstrated that more patients were clear or near clear of SKs after use of the 40% concentration solution than those in the vehicle group, according to the prescribing information. In the studies, patients with four raised SKs of the face, trunk or extremities were treated with Eskata at baseline and 3 weeks later, if necessary, or a vehicle. None of those in the vehicle group were cleared at follow-up (day 106), compared with 4% and 8% of those treated with Eskata, and at least three of the four lesions treated with Eskata had cleared in 13% and 23% at follow-up. Local skin reactions were mostly mild and transient, the most common being itching, stinging, crusting, swelling, redness, and scaling at the application site.
In the clinical studies, the solution was applied up to two times, on day 0 and again 3 weeks later on day 22. The lesion should first be cleansed with alcohol, and appropriate measures should be taken to ensure Eskata does not come in contact with the eyes. Nitrile or vinyl examination gloves should be used for application. Eskata is applied to the SKs with a pen-like applicator. The solution is applied uniformly in a circular motion with excess and the surrounding area patted dry with an absorbent wipe. Cotton gauze, tips, paper towels, or tissue should not be used as organic compounds can react with high concentrations of hydrogen peroxide. In a treatment session, one lesion may be treated up to four times, 1 minute apart. The applicator is used only once and may be discarded after lesions are treated. The packages may be stored at controlled room temperature (68° F to 77° F).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Dr. Wesley has served on an advisory board panel for Aclaris, the manufacturer of Eskata. Dr. Talakoub had no related disclosures. Write to them at [email protected].
Formerly known as A-101, . It is a 40% hydrogen peroxide topical solution that is applied to raised SKs as an in-office procedure. As previously reported, SKs are composed of hyperadherent senescent cells that are arrested in the G1 phase of the cell cycle. They exhibit decreased apoptotic cell death, compared with normal skin.
Two double-blind vehicle-controlled studies demonstrated that more patients were clear or near clear of SKs after use of the 40% concentration solution than those in the vehicle group, according to the prescribing information. In the studies, patients with four raised SKs of the face, trunk or extremities were treated with Eskata at baseline and 3 weeks later, if necessary, or a vehicle. None of those in the vehicle group were cleared at follow-up (day 106), compared with 4% and 8% of those treated with Eskata, and at least three of the four lesions treated with Eskata had cleared in 13% and 23% at follow-up. Local skin reactions were mostly mild and transient, the most common being itching, stinging, crusting, swelling, redness, and scaling at the application site.
In the clinical studies, the solution was applied up to two times, on day 0 and again 3 weeks later on day 22. The lesion should first be cleansed with alcohol, and appropriate measures should be taken to ensure Eskata does not come in contact with the eyes. Nitrile or vinyl examination gloves should be used for application. Eskata is applied to the SKs with a pen-like applicator. The solution is applied uniformly in a circular motion with excess and the surrounding area patted dry with an absorbent wipe. Cotton gauze, tips, paper towels, or tissue should not be used as organic compounds can react with high concentrations of hydrogen peroxide. In a treatment session, one lesion may be treated up to four times, 1 minute apart. The applicator is used only once and may be discarded after lesions are treated. The packages may be stored at controlled room temperature (68° F to 77° F).
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Dr. Wesley has served on an advisory board panel for Aclaris, the manufacturer of Eskata. Dr. Talakoub had no related disclosures. Write to them at [email protected].
Facial exercises improved appearance in small study of middle-aged women
The role of skin laxity and substructural fat and muscle loss in the appearance of facial aging has been recognized already, and there has been interest within the nonmedical community regarding use of facial exercise to improve appearance, Murad Alam, MD, of Northwestern University, Chicago, and his colleagues wrote in a research letter published in JAMA Dermatology.
The researchers recruited healthy women aged 40-65 years with some photodamage to the face and an interest in facial exercises. After two 90-minute, in-person training sessions with a certified instructor, the participants were asked to perform a 30-minute facial exercise session daily for 8 weeks at home, followed by sessions every other day during weeks 9-20. Sixteen patients completed the full 20-week study.
Two blinded physicians used validated assessment scales to compare photographs of the participants taken at the beginning and end of the 20-week period. Facial exercise was associated with an improved mean upper-cheek fullness score, compared with baseline (1.1 vs. 1.8, respectively; P = .003), and an improved mean lower-cheek fullness score, compared with baseline (0.9 vs. 1.6; P = .003).
In addition, blinded physicians’ estimates of the women’s ages decreased significantly: The estimates dropped from an average of 51 years at baseline to an average of 49 years after the women completed the 20 weeks of facial exercises (P = .002), Dr. Alam and his associates reported.
The study was limited by several factors, including its small sample size, the lack of a control group, and a self-selected population that may have been especially motivated to follow the exercise routine, the researchers noted.
However, the results suggest that the cause for improvements in appearance as a result of the exercises “may be exercise-actuated hypertrophy of cheek and other muscles,” they said. “Further research is warranted to isolate the causes and effects of exercise-related changes and to assess the generalizability of these findings,” Dr. Alam and his associates concluded.
The study was supported by research funds from the department of dermatology at Northwestern University. Dr. Alam disclosed serving as a consultant for Amway and Leo Pharma and has served as an investigator on studies supported in part by Allergan, Medicis Pharmaceutical, BioForm Medical, and Ulthera.
SOURCE: Alam M et al. JAMA Dermatol. 2018 Jan 3. doi: 10.1001/jamadermatol.2017.5142
The role of skin laxity and substructural fat and muscle loss in the appearance of facial aging has been recognized already, and there has been interest within the nonmedical community regarding use of facial exercise to improve appearance, Murad Alam, MD, of Northwestern University, Chicago, and his colleagues wrote in a research letter published in JAMA Dermatology.
The researchers recruited healthy women aged 40-65 years with some photodamage to the face and an interest in facial exercises. After two 90-minute, in-person training sessions with a certified instructor, the participants were asked to perform a 30-minute facial exercise session daily for 8 weeks at home, followed by sessions every other day during weeks 9-20. Sixteen patients completed the full 20-week study.
Two blinded physicians used validated assessment scales to compare photographs of the participants taken at the beginning and end of the 20-week period. Facial exercise was associated with an improved mean upper-cheek fullness score, compared with baseline (1.1 vs. 1.8, respectively; P = .003), and an improved mean lower-cheek fullness score, compared with baseline (0.9 vs. 1.6; P = .003).
In addition, blinded physicians’ estimates of the women’s ages decreased significantly: The estimates dropped from an average of 51 years at baseline to an average of 49 years after the women completed the 20 weeks of facial exercises (P = .002), Dr. Alam and his associates reported.
The study was limited by several factors, including its small sample size, the lack of a control group, and a self-selected population that may have been especially motivated to follow the exercise routine, the researchers noted.
However, the results suggest that the cause for improvements in appearance as a result of the exercises “may be exercise-actuated hypertrophy of cheek and other muscles,” they said. “Further research is warranted to isolate the causes and effects of exercise-related changes and to assess the generalizability of these findings,” Dr. Alam and his associates concluded.
The study was supported by research funds from the department of dermatology at Northwestern University. Dr. Alam disclosed serving as a consultant for Amway and Leo Pharma and has served as an investigator on studies supported in part by Allergan, Medicis Pharmaceutical, BioForm Medical, and Ulthera.
SOURCE: Alam M et al. JAMA Dermatol. 2018 Jan 3. doi: 10.1001/jamadermatol.2017.5142
The role of skin laxity and substructural fat and muscle loss in the appearance of facial aging has been recognized already, and there has been interest within the nonmedical community regarding use of facial exercise to improve appearance, Murad Alam, MD, of Northwestern University, Chicago, and his colleagues wrote in a research letter published in JAMA Dermatology.
The researchers recruited healthy women aged 40-65 years with some photodamage to the face and an interest in facial exercises. After two 90-minute, in-person training sessions with a certified instructor, the participants were asked to perform a 30-minute facial exercise session daily for 8 weeks at home, followed by sessions every other day during weeks 9-20. Sixteen patients completed the full 20-week study.
Two blinded physicians used validated assessment scales to compare photographs of the participants taken at the beginning and end of the 20-week period. Facial exercise was associated with an improved mean upper-cheek fullness score, compared with baseline (1.1 vs. 1.8, respectively; P = .003), and an improved mean lower-cheek fullness score, compared with baseline (0.9 vs. 1.6; P = .003).
In addition, blinded physicians’ estimates of the women’s ages decreased significantly: The estimates dropped from an average of 51 years at baseline to an average of 49 years after the women completed the 20 weeks of facial exercises (P = .002), Dr. Alam and his associates reported.
The study was limited by several factors, including its small sample size, the lack of a control group, and a self-selected population that may have been especially motivated to follow the exercise routine, the researchers noted.
However, the results suggest that the cause for improvements in appearance as a result of the exercises “may be exercise-actuated hypertrophy of cheek and other muscles,” they said. “Further research is warranted to isolate the causes and effects of exercise-related changes and to assess the generalizability of these findings,” Dr. Alam and his associates concluded.
The study was supported by research funds from the department of dermatology at Northwestern University. Dr. Alam disclosed serving as a consultant for Amway and Leo Pharma and has served as an investigator on studies supported in part by Allergan, Medicis Pharmaceutical, BioForm Medical, and Ulthera.
SOURCE: Alam M et al. JAMA Dermatol. 2018 Jan 3. doi: 10.1001/jamadermatol.2017.5142
FROM JAMA DERMATOLOGY
Key clinical point: A 20-week program of facial exercise significantly improved facial fullness and perceived age among women aged 40-65 years.
Major finding: Fullness of the upper and lower cheek significantly improved from baseline after the exercise program, based on a validated scale.
Data source: The data come from a study of 27 women aged 40-65 years.
Disclosures: The study was supported by research funds from the department of dermatology at Northwestern University. Dr. Alam disclosed serving as a consultant for Amway and LEO Pharma and has served as an investigator on studies supported in part by Allergan, Medicis Pharmaceutical, BioForm Medical, and Ulthera.
Source: Alam M et al. JAMA Dermatol. 2018 Jan 3. doi: 10.1001/jamadermatol.2017.5142.
Regenerative Medicine in Cosmetic Dermatology
Regenerative medicine encompasses innovative therapies that allow the body to repair or regenerate aging cells, tissues, and organs. The skin is a particularly attractive organ for the application of novel regenerative therapies due to its easy accessibility. Among these therapies, stem cells and platelet-rich plasma (PRP) have garnered interest based on their therapeutic potential in scar reduction, antiaging effects, and treatment of alopecia.
Stem cells possess the cardinal features of self-renewal and plasticity. Self-renewal refers to symmetric cell division generating daughter cells identical to the parent cell.1 Plasticity is the ability to generate cell types other than the germ line or tissue lineage from which stem cells derive.2 Stem cells can be categorized according to their differentiation potential. Totipotent stem cells may develop into any primary germ cell layer (ectoderm, mesoderm, endoderm) of the embryo, as well as extraembryonic tissue such as the trophoblast, which gives rise to the placenta. Pluripotent stem cells such as embryonic stem cells have the capacity to differentiate into any derivative of the 3 germ cell layers but have lost their ability to differentiate into the trophoblast.3 Adults lack totipotent or pluripotent cells; they have multipotent or unipotent cells. Multipotent stem cells are able to differentiate into multiple cell types from similar lineages; mesenchymal stem cells (MSCs), for example, can differentiate into adipogenic, osteogenic, chondrogenic, and myogenic cells.4 Unipotent stem cells have the lowest differentiation potential and can only self-regenerate. Herein, we review stem cell sources and their therapeutic potential in aesthetic dermatology.
Multipotent Stem Cells
Multipotent stem cells derived from the bone marrow, umbilical cord, adipose tissue, dermis, or hair follicle bulge have various clinical applications in dermatology. Stem cells from these sources are primarily utilized in an autologous manner in which they are processed outside the body and reintroduced into the donor. Autologous multipotent hematopoietic bone marrow cells were first successfully used for the treatment of chronic wounds and show promise for the treatment of atrophic scars.5,6 However, due to the invasive nature of extracting bone marrow stem cells and their declining number with age, other sources of multipotent stem cells have fallen into favor.
Umbilical cord blood is a source of multipotent hematopoietic stem cells for which surgical intervention is not necessary because they are retrieved after umbilical cord clamping.7 Advantages of sourcing stem cells from umbilical cord blood includes high regenerative power compared to a newborn’s skin and low immunogenicity given that the newborn is immunologically immature.8
Another popular source for autologous stem cells is adipose tissue due to its ease of accessibility and relative abundance. Given that adipose tissue–derived stem cells (ASCs) are capable of differentiating into adipocytes that help maintain volume over time, they are being used for midface contouring, lip augmentation, facial rejuvenation, facial scarring, lipodystrophy, penile girth enhancement, and vaginal augmentation. Adipose tissue–derived stem cells also are capable of differentiating into other types of tissue, including cartilage and bone. Thus, they have been successfully harnessed in the treatment of patients affected by systemic sclerosis and Parry-Romberg syndrome as well in the functional and aesthetic reconstruction of various military combat–related deformities.9,10
Adipose tissue–derived stem cells are commonly harvested from lipoaspirate of the abdomen and are combined with supportive mechanical scaffolds such as hydrogels. Lipoaspirate itself can serve as a scaffold for ASCs. Accordingly, ASCs also are being utilized as a scaffold for autologous fat transfer procedures in an effort to increase the viability of transplanted donor tissue, a process known as cell-assisted lipotransfer (CAL). In CAL, a fraction of the aspirated fat is processed for isolation of ASCs, which are then recombined with the remainder of the aspirated fat prior to grafting.11 However, there is conflicting evidence as to whether CAL leads to improved graft success relative to conventional autologous fat transfer.12,13
The skin also serves as an easily accessible and abundant autologous source of stem cells. A subtype of dermal fibroblasts has been proven to have multipotent potential.14,15 These dermal fibroblasts are harvested from one area of the skin using punch biopsy and are processed and reinjected into another desired area of the skin.16 Autologous human fibroblasts have proven to be effective for the treatment of wrinkles, rhytides, and acne scars.17 In June 2011, the US Food and Drug Administration approved azficel-T, an autologous cellular product created by harvesting fibroblasts from a patient’s own postauricular skin, culture-expanding them in vitro for 3 months, and reinjecting the cells into the desired area of dermis in a series of treatments. This product was the first personalized cell therapy approved by the US Food and Drug Administration for aesthetic uses, specifically for the improvement of nasolabial fold wrinkles.18
In adults, hair follicles contain an area known as the bulge, which is a site rich in epithelial and melanocytic stem cells. Bulge stem cells have the ability to reproduce the interfollicular epidermis, hair follicle structures, and sebaceous glands, and they have been used to construct entirely new hair follicles in an artificial in vivo system.19 Sugiyama-Nakagiri et al20 demonstrated that an entire hair follicle epithelium and interfollicular epidermis can be regenerated using cultured bulge stem cells. The cultured bulge stem cells were mixed with dermal papilla cells from neonatal rat vibrissae and engrafted into a silicone chamber implanted on the backs of severe combined immune deficient (SCID) mice. The grafts exhibited tufts of hair as well as a complete interfollicular epidermis at 4 weeks after transplantation.20 Thus, these bulge stem cells have the potential to treat male androgenic alopecia and female pattern hair loss. Bulge stem cells also have been shown to accelerate wound healing.21 Additionally, autologous melanocytic stem cells located at the hair follicle bulge are effective for treating vitiligo and are being investigated for the treatment of hair graying.22
Induced Pluripotent Stem Cells
Given the ethical concerns that surround the procurement and use of embryonic stem cells, efforts have been made to retrieve pluripotent stem cells from adults. A major breakthrough occurred in 2006 when researchers altered the genes of specialized adult mouse cells to cause dedifferentiation and the return to an embryoniclike stem cell state.23 Mouse somatic cells were reprogrammed through the activation of a combination of transcription factors. The resulting cells were termed induced pluripotent stem cells (iPSCs) and have since been recreated in human cell lines. The discovery of iPSCs precipitated a translational science revolution. Physician-scientists sought ways to apply the reprogrammed cells to the pathophysiology of obscure diseases, examination of drug targets, and regeneration of human tissue.24 Tissue regeneration via induced naïve somatic cells has shown promise as a future method to treat neurologic, cardiovascular, and ophthalmologic diseases.25
As the technology of cultivating and identifying optimal sources of iPSCs continues to advance, stem cell–based treatments have evolved as leading prospects in the field of biogerontology.26-29 Although much of the research in antiaging medicine has utilized iPSCs to reprogram cell senescence, the altering of iPSCs at a cellular level also allows for the stimulation of collagen synthesis. This potential for collagen generation may have direct applicability in dermatologic practice, particularly for aesthetic treatments.
Much of the research into iPSC-derived collagen has focused on genodermatoses. Itoh et al30 examined the creation of collagen through iPSCs to identify possible treatments for recessive dystrophic epidermolysis bullosa (DEB). Recessive DEB is characterized by mutations in the COL7A1 gene, which encodes type VII collagen, a basement membrane protein and component of the anchoring fibrils essential for skin integrity.31 Itoh et al30 began with source cells obtained from a skin biopsy. The cells were dedifferentiated to iPSCs and then induced into dermal fibroblasts according to the methods established in prior studies of embryonic stem cells, namely with the use of ascorbic acid and transforming growth factor b. The newly formed fibroblasts were determined to be functional based on their ability to synthesize mature type VII collagen.30 Once the viability of the iPSC-derived fibroblasts was confirmed in vitro, the cells were further tested through combination with human keratinocytes on SCID mice. The human keratinocytes grew together with the iPSC-derived fibroblasts, producing type VII collagen in the basement membrane zone and creating an epidermis with the normal markers.30 Similarly, Robbins et al32 utilized SCID mice to successfully demonstrate that the transfection of keratinocytes from patients with junctional epidermolysis bullosa into SCID mice produced phenotypically normal skin.
Sebastiano et al33 combined the concepts of iPSCs and genome editing in another study of recessive DEB. The investigators first cultured iPSCs from biopsies of affected patients. After deriving iPSCs and correcting their mutation via adenovirus-associated viral gene editing, the COL7A1 mutation-free cells were differentiated into keratinocytes. These iPSC-derived keratinocytes were subsequently grafted onto mice, which led to the production of wild-type collagen VII and a stratified epidermis. Despite this successful outcome, the grafts of iPSC-derived epidermis did not survive longer than 1 month.33
One of the many obstacles facing the practical use of stem cells is their successful incorporation into human tissue. A possible solution was uncovered by Zhang et al34 who examined iPSC-derived MSCs. Mesenchymal stem cells communicate via paracrine mechanisms, whereby exosomes containing RNA and proteins are released to potentiate a regenerative effect.35 Zhang et al34 found that injecting exosomes from human iPSC-derived MSCs into the wound sites of rats stimulated the production of type I collagen, type III collagen, and elastin. The wound sites demonstrated accelerated closure, narrower scar widths, and increased collagen maturity.
Understanding the role that local environment plays in stem cell differentiation, Xu et al36 aimed to create an extracellular scaffold to induce fibroblast behavior from iPSCs. The authors engineered a framework similar to the normal extracellular membrane using proteoglycans, glycosaminoglycans, fibrinogen, and connective tissue growth factor. The iPSCs were then applied to the scaffolding, which led to successful fibroblast differentiation and type I collagen synthesis.36 This use of local biosignaling cues holds important ramifications for controlling the fate of stem cells that have been introduced into a new environment.
Although the application of iPSCs in clinical dermatology has yet to be achieved, progress in the field is moving at a rapid pace. Several logistical elements require further mastery before therapeutics can be delivered. These areas include the optimal environment for iPSC differentiation, methods for maximization of graft survival, and different modes of transplanting iPSC-derived cells into patients. In cosmetic practice, success will depend on intradermal injections of collagen-producing iPSC-derived cells that possess long-term proliferative potential. Current research in mice models has demonstrated viability up to 16 weeks after intradermal injection of such cells.37
Plant Stem Cells
In discussing the dermatologic applications of stem cell technology, clinicians should be aware of the plant stem cell products that have become a popular cosmeceutical trend. Companies advertise plant cells as a natural source of regenerative cells that can induce rejuvenation in human skin; however, there are no significant data to indicate that plant stem cells encourage or activate cellular growth in humans. Indeed, for stem cells to differentiate and produce viable components, the cells must first be incorporated as living components in the host tissue. Because plant stem cells do not survive in human tissue and plant cell cytokines fail to interact with the receptors on human cells, their current value in cosmeceuticals may be overstated.
Platelet-Rich Plasma
Platelet-rich plasma also is commonly associated with stem cell therapy, as PRP is known to potentiate stem cell proliferation, migration, and differentiation. However, PRP does not contain stem cells and is instead autologous plasma concentrated with platelets. In fact, platelets cannot even be classified as cells given that they lack a nucleus; platelets are considered cell fragments. The regenerative potential of PRP can be attributed to the growth factors released from platelets, which play an important role in tissue regeneration and repair. Platelet-rich plasma currently is being used in dermatology for skin rejuvenation (reduction of wrinkles and furrows) and treatment of acne scars.38 There also is evidence supporting the effectiveness of PRP for alopecia and wound therapy, as growth factors play a vital role in hair growth and wound healing.38 Apart from the use of PRP on its own, it can be used as a supplement to enhance the effects of antiaging procedures such as microneedling.39
Future Directions
Multipotent stem cells and iPSCs discussed herein provide much promise in the field of regenerative dermatology. They are increasingly accessible and circumvent the use of ethically questionable embryonic stem cells. Although there is a general consensus on the great potential of stem cells for treating aesthetic skin conditions, high-quality randomized controlled trials remain scarce within the literature. Recognizing and optimizing these opportunities remains the next step in the future delivery of evidence-based care in regenerative dermatology.
- Thomas ED, Lochte HL, Lu WC, et al. Intravenous infusion of bone marrow in patients receiving radiation and chemotherapy. N Engl J Med. 1957;257:491-496.
- Ogliari KS, Marinowic D, Brum DE, et al. Stem cells in dermatology. An Bras Dermatol. 2014;89:286-291.
- Xu C, Inokuma MS, Denham J, et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol. 2001;19:971-974.
- Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7:211-228.
- Badiavas EV, Falanga V. Treatment of chronic wounds with bone marrow-derived cells. Arch Dermatol. 2003;139:510-516.
- Ibrahim ZA, Eltatawy RA, Ghaly NR, et al. Autologous bone marrow stem cells in atrophic acne scars: a pilot study. J Dermatolog Treat. 2015;26:260-265.
- Broxmeyer HE, Douglas GW, Hangoc G, et al. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc Natl Acad Sci U S A. 1989;86:3828-3832.
- Gluckman E, Rocha V, Boyer-Chammard A, et al. Outcome of cord-blood transplantation from related and unrelated donors. Eurocord Transplant Group and the European Blood and Marrow Transplantation Group. N Engl J Med. 1997;337:373-381.
- Valerio IL, Sabino JM, Dearth CL. Plastic surgery challenges in war wounded II: regenerative medicine. Adv Wound Care (New Rochelle). 2016;5:412-419.
- Vescarelli E, D’Amici S, Onesti MG, et al. Adipose-derived stem cell: an innovative therapeutic approach in systemic sclerosis and Parry-Romberg syndrome. CellR4. 2014;2:E791-E797.
- Yoshimura K, Sato K, Aoi N, et al. Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg. 2008;32:48-55.
- Grabin S, Antes G, Stark GB, et al. Cell-assisted lipotransfer: a critical appraisal of the evidence. Dtsch Arztebl Int. 2015;112:255.
- Zhou Y, Wang J, Li H, et al. Efficacy and safety of cell-assisted lipotransfer: a systematic review and meta-analysis. Plast Reconstr Surg. 2016;137:E44-E57.
- Toma JG, Akhavan M, Fernandes KJL, et al. Isolation of multipotent adult stem cells from the dermis of mammalian skin. Nat Cell Biol. 2001;3:778-784.
- Toma JG, McKenzie IA, Bagli D, et al. Isolation and characterization of multipotent skin-derived precursors from human skin. Stem Cells. 2005;23:727-737.
- Homicz MR, Watson D. Review of injectable materials for soft tissue augmentation. Facial Plast Surg. 2004;20:21-29.
- Kumar S, Mahajan BB, Kaur S, et al. Autologous therapies in dermatology. J Clin Aesthet Dermatol. 2014;7:38-45.
- Schmidt C. FDA approves first cell therapy for wrinkle-free visage. Nat Biotech. 2011;29:674-675.
- Gentile P, Scioli MG, Bielli A, et al. Stem cells from human hair follicles: first mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017;4:58.
- Sugiyama-Nakagiri Y, Akiyama M, Shimizu H. Hair follicle stem cell-targeted gene transfer and reconstitution system. Gene Ther. 2006;13:732-737.
- Heidari F, Yari A, Rasoolijazi H, et al. Bulge hair follicle stem cells accelerate cutaneous wound healing in rats. Wounds. 2016;28:132-141.
- Lee JH, Fisher DE. Melanocyte stem cells as potential therapeutics in skin disorders. Expert Opin Biol Ther. 2014;14:1-11.
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676.
- Singh VK, Kalsan M, Kumar N, et al. Induced pluripotent stem cells: applications in regenerative medicine, disease modeling, and drug discovery. Front Cell Dev Biol. 2015;3:2.
- Aoi T. 10th anniversary of iPS cells: The challenges that lie ahead. J Biochem. 2016;160:121-129.
- Lowry WE, Plath K. The many ways to make an iPS cell. Nat Biotechnol. 2008;26:1246-1248.
- Kim K, Doi A, Wen B, et al. Epigenetic memory in induced pluripotent stem cells. Nature. 2010;467:285-290.
- Gafni O, Weinberger L, Mansour AA, et al. Derivation of novel human ground state naive pluripotent stem cells. Nature. 2013;504:282-286.
- Pareja-Galeano H, Sanchis-Gomar F, Pérez LM, et al. IPSCs-based anti-aging therapies: Recent discoveries and future challenges. Ageing Res Rev. 2016;27:37-41.
- Itoh M, Umegaki-Arao N, Guo Z, et al. Generation of 3D skin equivalents fully reconstituted from human induced pluripotent stem cells (iPSCs). PLoS One. 2013;8:e77673.
- Nyström A, Velati D, Mittapalli VR, et al. Collagen VII plays a dual role in wound healing. J Clin Invest. 2013;123:3498-3509.
- Robbins PB, Lin Q, Goodnough JB, et al. In vivo restoration of laminin 5 β3 expression and function in junctional epidermolysis bullosa. Proc Natl Acad Sci. 2001;98:5193-5198.
- Sebastiano V, Zhen HH, Haddad B, et al. Human COL7A1-corrected induced pluripotent stem cells for the treatment of recessive dystrophic epidermolysis bullosa. Sci Transl Med. 2014;6:264ra163.
- Zhang J, Guan J, Niu X, et al. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J Transl Med. 2015;13:49.
- Pap E, Pállinger É, Pásztói M, et al. Highlights of a new type of intercellular communication: microvesicle-based information transfer. Inflamm Res. 2009;58:1-8.
- Xu R, Taskin MB, Rubert M, et al. hiPS-MSCs differentiation towards fibroblasts on a 3D ECM mimicking scaffold. Sci Rep. 2015;5:8480.
- Wenzel D, Bayerl J, Nyström A, et al. Genetically corrected iPSCs as cell therapy for recessive dystrophic epidermolysis bullosa. Sci Transl Med. 2014;6:264ra165.
- Bednarska K, Kieszek R, Domagała P, et al. The use of platelet-rich-plasma in aesthetic and regenerative medicine. MEDtube Science. 2015;2:8-15.
- Hashim PW, Levy Z, Cohen JL, et al. Microneedling therapy with and without platelet-rich plasma. Cutis. 2017;99:239-242.
Regenerative medicine encompasses innovative therapies that allow the body to repair or regenerate aging cells, tissues, and organs. The skin is a particularly attractive organ for the application of novel regenerative therapies due to its easy accessibility. Among these therapies, stem cells and platelet-rich plasma (PRP) have garnered interest based on their therapeutic potential in scar reduction, antiaging effects, and treatment of alopecia.
Stem cells possess the cardinal features of self-renewal and plasticity. Self-renewal refers to symmetric cell division generating daughter cells identical to the parent cell.1 Plasticity is the ability to generate cell types other than the germ line or tissue lineage from which stem cells derive.2 Stem cells can be categorized according to their differentiation potential. Totipotent stem cells may develop into any primary germ cell layer (ectoderm, mesoderm, endoderm) of the embryo, as well as extraembryonic tissue such as the trophoblast, which gives rise to the placenta. Pluripotent stem cells such as embryonic stem cells have the capacity to differentiate into any derivative of the 3 germ cell layers but have lost their ability to differentiate into the trophoblast.3 Adults lack totipotent or pluripotent cells; they have multipotent or unipotent cells. Multipotent stem cells are able to differentiate into multiple cell types from similar lineages; mesenchymal stem cells (MSCs), for example, can differentiate into adipogenic, osteogenic, chondrogenic, and myogenic cells.4 Unipotent stem cells have the lowest differentiation potential and can only self-regenerate. Herein, we review stem cell sources and their therapeutic potential in aesthetic dermatology.
Multipotent Stem Cells
Multipotent stem cells derived from the bone marrow, umbilical cord, adipose tissue, dermis, or hair follicle bulge have various clinical applications in dermatology. Stem cells from these sources are primarily utilized in an autologous manner in which they are processed outside the body and reintroduced into the donor. Autologous multipotent hematopoietic bone marrow cells were first successfully used for the treatment of chronic wounds and show promise for the treatment of atrophic scars.5,6 However, due to the invasive nature of extracting bone marrow stem cells and their declining number with age, other sources of multipotent stem cells have fallen into favor.
Umbilical cord blood is a source of multipotent hematopoietic stem cells for which surgical intervention is not necessary because they are retrieved after umbilical cord clamping.7 Advantages of sourcing stem cells from umbilical cord blood includes high regenerative power compared to a newborn’s skin and low immunogenicity given that the newborn is immunologically immature.8
Another popular source for autologous stem cells is adipose tissue due to its ease of accessibility and relative abundance. Given that adipose tissue–derived stem cells (ASCs) are capable of differentiating into adipocytes that help maintain volume over time, they are being used for midface contouring, lip augmentation, facial rejuvenation, facial scarring, lipodystrophy, penile girth enhancement, and vaginal augmentation. Adipose tissue–derived stem cells also are capable of differentiating into other types of tissue, including cartilage and bone. Thus, they have been successfully harnessed in the treatment of patients affected by systemic sclerosis and Parry-Romberg syndrome as well in the functional and aesthetic reconstruction of various military combat–related deformities.9,10
Adipose tissue–derived stem cells are commonly harvested from lipoaspirate of the abdomen and are combined with supportive mechanical scaffolds such as hydrogels. Lipoaspirate itself can serve as a scaffold for ASCs. Accordingly, ASCs also are being utilized as a scaffold for autologous fat transfer procedures in an effort to increase the viability of transplanted donor tissue, a process known as cell-assisted lipotransfer (CAL). In CAL, a fraction of the aspirated fat is processed for isolation of ASCs, which are then recombined with the remainder of the aspirated fat prior to grafting.11 However, there is conflicting evidence as to whether CAL leads to improved graft success relative to conventional autologous fat transfer.12,13
The skin also serves as an easily accessible and abundant autologous source of stem cells. A subtype of dermal fibroblasts has been proven to have multipotent potential.14,15 These dermal fibroblasts are harvested from one area of the skin using punch biopsy and are processed and reinjected into another desired area of the skin.16 Autologous human fibroblasts have proven to be effective for the treatment of wrinkles, rhytides, and acne scars.17 In June 2011, the US Food and Drug Administration approved azficel-T, an autologous cellular product created by harvesting fibroblasts from a patient’s own postauricular skin, culture-expanding them in vitro for 3 months, and reinjecting the cells into the desired area of dermis in a series of treatments. This product was the first personalized cell therapy approved by the US Food and Drug Administration for aesthetic uses, specifically for the improvement of nasolabial fold wrinkles.18
In adults, hair follicles contain an area known as the bulge, which is a site rich in epithelial and melanocytic stem cells. Bulge stem cells have the ability to reproduce the interfollicular epidermis, hair follicle structures, and sebaceous glands, and they have been used to construct entirely new hair follicles in an artificial in vivo system.19 Sugiyama-Nakagiri et al20 demonstrated that an entire hair follicle epithelium and interfollicular epidermis can be regenerated using cultured bulge stem cells. The cultured bulge stem cells were mixed with dermal papilla cells from neonatal rat vibrissae and engrafted into a silicone chamber implanted on the backs of severe combined immune deficient (SCID) mice. The grafts exhibited tufts of hair as well as a complete interfollicular epidermis at 4 weeks after transplantation.20 Thus, these bulge stem cells have the potential to treat male androgenic alopecia and female pattern hair loss. Bulge stem cells also have been shown to accelerate wound healing.21 Additionally, autologous melanocytic stem cells located at the hair follicle bulge are effective for treating vitiligo and are being investigated for the treatment of hair graying.22
Induced Pluripotent Stem Cells
Given the ethical concerns that surround the procurement and use of embryonic stem cells, efforts have been made to retrieve pluripotent stem cells from adults. A major breakthrough occurred in 2006 when researchers altered the genes of specialized adult mouse cells to cause dedifferentiation and the return to an embryoniclike stem cell state.23 Mouse somatic cells were reprogrammed through the activation of a combination of transcription factors. The resulting cells were termed induced pluripotent stem cells (iPSCs) and have since been recreated in human cell lines. The discovery of iPSCs precipitated a translational science revolution. Physician-scientists sought ways to apply the reprogrammed cells to the pathophysiology of obscure diseases, examination of drug targets, and regeneration of human tissue.24 Tissue regeneration via induced naïve somatic cells has shown promise as a future method to treat neurologic, cardiovascular, and ophthalmologic diseases.25
As the technology of cultivating and identifying optimal sources of iPSCs continues to advance, stem cell–based treatments have evolved as leading prospects in the field of biogerontology.26-29 Although much of the research in antiaging medicine has utilized iPSCs to reprogram cell senescence, the altering of iPSCs at a cellular level also allows for the stimulation of collagen synthesis. This potential for collagen generation may have direct applicability in dermatologic practice, particularly for aesthetic treatments.
Much of the research into iPSC-derived collagen has focused on genodermatoses. Itoh et al30 examined the creation of collagen through iPSCs to identify possible treatments for recessive dystrophic epidermolysis bullosa (DEB). Recessive DEB is characterized by mutations in the COL7A1 gene, which encodes type VII collagen, a basement membrane protein and component of the anchoring fibrils essential for skin integrity.31 Itoh et al30 began with source cells obtained from a skin biopsy. The cells were dedifferentiated to iPSCs and then induced into dermal fibroblasts according to the methods established in prior studies of embryonic stem cells, namely with the use of ascorbic acid and transforming growth factor b. The newly formed fibroblasts were determined to be functional based on their ability to synthesize mature type VII collagen.30 Once the viability of the iPSC-derived fibroblasts was confirmed in vitro, the cells were further tested through combination with human keratinocytes on SCID mice. The human keratinocytes grew together with the iPSC-derived fibroblasts, producing type VII collagen in the basement membrane zone and creating an epidermis with the normal markers.30 Similarly, Robbins et al32 utilized SCID mice to successfully demonstrate that the transfection of keratinocytes from patients with junctional epidermolysis bullosa into SCID mice produced phenotypically normal skin.
Sebastiano et al33 combined the concepts of iPSCs and genome editing in another study of recessive DEB. The investigators first cultured iPSCs from biopsies of affected patients. After deriving iPSCs and correcting their mutation via adenovirus-associated viral gene editing, the COL7A1 mutation-free cells were differentiated into keratinocytes. These iPSC-derived keratinocytes were subsequently grafted onto mice, which led to the production of wild-type collagen VII and a stratified epidermis. Despite this successful outcome, the grafts of iPSC-derived epidermis did not survive longer than 1 month.33
One of the many obstacles facing the practical use of stem cells is their successful incorporation into human tissue. A possible solution was uncovered by Zhang et al34 who examined iPSC-derived MSCs. Mesenchymal stem cells communicate via paracrine mechanisms, whereby exosomes containing RNA and proteins are released to potentiate a regenerative effect.35 Zhang et al34 found that injecting exosomes from human iPSC-derived MSCs into the wound sites of rats stimulated the production of type I collagen, type III collagen, and elastin. The wound sites demonstrated accelerated closure, narrower scar widths, and increased collagen maturity.
Understanding the role that local environment plays in stem cell differentiation, Xu et al36 aimed to create an extracellular scaffold to induce fibroblast behavior from iPSCs. The authors engineered a framework similar to the normal extracellular membrane using proteoglycans, glycosaminoglycans, fibrinogen, and connective tissue growth factor. The iPSCs were then applied to the scaffolding, which led to successful fibroblast differentiation and type I collagen synthesis.36 This use of local biosignaling cues holds important ramifications for controlling the fate of stem cells that have been introduced into a new environment.
Although the application of iPSCs in clinical dermatology has yet to be achieved, progress in the field is moving at a rapid pace. Several logistical elements require further mastery before therapeutics can be delivered. These areas include the optimal environment for iPSC differentiation, methods for maximization of graft survival, and different modes of transplanting iPSC-derived cells into patients. In cosmetic practice, success will depend on intradermal injections of collagen-producing iPSC-derived cells that possess long-term proliferative potential. Current research in mice models has demonstrated viability up to 16 weeks after intradermal injection of such cells.37
Plant Stem Cells
In discussing the dermatologic applications of stem cell technology, clinicians should be aware of the plant stem cell products that have become a popular cosmeceutical trend. Companies advertise plant cells as a natural source of regenerative cells that can induce rejuvenation in human skin; however, there are no significant data to indicate that plant stem cells encourage or activate cellular growth in humans. Indeed, for stem cells to differentiate and produce viable components, the cells must first be incorporated as living components in the host tissue. Because plant stem cells do not survive in human tissue and plant cell cytokines fail to interact with the receptors on human cells, their current value in cosmeceuticals may be overstated.
Platelet-Rich Plasma
Platelet-rich plasma also is commonly associated with stem cell therapy, as PRP is known to potentiate stem cell proliferation, migration, and differentiation. However, PRP does not contain stem cells and is instead autologous plasma concentrated with platelets. In fact, platelets cannot even be classified as cells given that they lack a nucleus; platelets are considered cell fragments. The regenerative potential of PRP can be attributed to the growth factors released from platelets, which play an important role in tissue regeneration and repair. Platelet-rich plasma currently is being used in dermatology for skin rejuvenation (reduction of wrinkles and furrows) and treatment of acne scars.38 There also is evidence supporting the effectiveness of PRP for alopecia and wound therapy, as growth factors play a vital role in hair growth and wound healing.38 Apart from the use of PRP on its own, it can be used as a supplement to enhance the effects of antiaging procedures such as microneedling.39
Future Directions
Multipotent stem cells and iPSCs discussed herein provide much promise in the field of regenerative dermatology. They are increasingly accessible and circumvent the use of ethically questionable embryonic stem cells. Although there is a general consensus on the great potential of stem cells for treating aesthetic skin conditions, high-quality randomized controlled trials remain scarce within the literature. Recognizing and optimizing these opportunities remains the next step in the future delivery of evidence-based care in regenerative dermatology.
Regenerative medicine encompasses innovative therapies that allow the body to repair or regenerate aging cells, tissues, and organs. The skin is a particularly attractive organ for the application of novel regenerative therapies due to its easy accessibility. Among these therapies, stem cells and platelet-rich plasma (PRP) have garnered interest based on their therapeutic potential in scar reduction, antiaging effects, and treatment of alopecia.
Stem cells possess the cardinal features of self-renewal and plasticity. Self-renewal refers to symmetric cell division generating daughter cells identical to the parent cell.1 Plasticity is the ability to generate cell types other than the germ line or tissue lineage from which stem cells derive.2 Stem cells can be categorized according to their differentiation potential. Totipotent stem cells may develop into any primary germ cell layer (ectoderm, mesoderm, endoderm) of the embryo, as well as extraembryonic tissue such as the trophoblast, which gives rise to the placenta. Pluripotent stem cells such as embryonic stem cells have the capacity to differentiate into any derivative of the 3 germ cell layers but have lost their ability to differentiate into the trophoblast.3 Adults lack totipotent or pluripotent cells; they have multipotent or unipotent cells. Multipotent stem cells are able to differentiate into multiple cell types from similar lineages; mesenchymal stem cells (MSCs), for example, can differentiate into adipogenic, osteogenic, chondrogenic, and myogenic cells.4 Unipotent stem cells have the lowest differentiation potential and can only self-regenerate. Herein, we review stem cell sources and their therapeutic potential in aesthetic dermatology.
Multipotent Stem Cells
Multipotent stem cells derived from the bone marrow, umbilical cord, adipose tissue, dermis, or hair follicle bulge have various clinical applications in dermatology. Stem cells from these sources are primarily utilized in an autologous manner in which they are processed outside the body and reintroduced into the donor. Autologous multipotent hematopoietic bone marrow cells were first successfully used for the treatment of chronic wounds and show promise for the treatment of atrophic scars.5,6 However, due to the invasive nature of extracting bone marrow stem cells and their declining number with age, other sources of multipotent stem cells have fallen into favor.
Umbilical cord blood is a source of multipotent hematopoietic stem cells for which surgical intervention is not necessary because they are retrieved after umbilical cord clamping.7 Advantages of sourcing stem cells from umbilical cord blood includes high regenerative power compared to a newborn’s skin and low immunogenicity given that the newborn is immunologically immature.8
Another popular source for autologous stem cells is adipose tissue due to its ease of accessibility and relative abundance. Given that adipose tissue–derived stem cells (ASCs) are capable of differentiating into adipocytes that help maintain volume over time, they are being used for midface contouring, lip augmentation, facial rejuvenation, facial scarring, lipodystrophy, penile girth enhancement, and vaginal augmentation. Adipose tissue–derived stem cells also are capable of differentiating into other types of tissue, including cartilage and bone. Thus, they have been successfully harnessed in the treatment of patients affected by systemic sclerosis and Parry-Romberg syndrome as well in the functional and aesthetic reconstruction of various military combat–related deformities.9,10
Adipose tissue–derived stem cells are commonly harvested from lipoaspirate of the abdomen and are combined with supportive mechanical scaffolds such as hydrogels. Lipoaspirate itself can serve as a scaffold for ASCs. Accordingly, ASCs also are being utilized as a scaffold for autologous fat transfer procedures in an effort to increase the viability of transplanted donor tissue, a process known as cell-assisted lipotransfer (CAL). In CAL, a fraction of the aspirated fat is processed for isolation of ASCs, which are then recombined with the remainder of the aspirated fat prior to grafting.11 However, there is conflicting evidence as to whether CAL leads to improved graft success relative to conventional autologous fat transfer.12,13
The skin also serves as an easily accessible and abundant autologous source of stem cells. A subtype of dermal fibroblasts has been proven to have multipotent potential.14,15 These dermal fibroblasts are harvested from one area of the skin using punch biopsy and are processed and reinjected into another desired area of the skin.16 Autologous human fibroblasts have proven to be effective for the treatment of wrinkles, rhytides, and acne scars.17 In June 2011, the US Food and Drug Administration approved azficel-T, an autologous cellular product created by harvesting fibroblasts from a patient’s own postauricular skin, culture-expanding them in vitro for 3 months, and reinjecting the cells into the desired area of dermis in a series of treatments. This product was the first personalized cell therapy approved by the US Food and Drug Administration for aesthetic uses, specifically for the improvement of nasolabial fold wrinkles.18
In adults, hair follicles contain an area known as the bulge, which is a site rich in epithelial and melanocytic stem cells. Bulge stem cells have the ability to reproduce the interfollicular epidermis, hair follicle structures, and sebaceous glands, and they have been used to construct entirely new hair follicles in an artificial in vivo system.19 Sugiyama-Nakagiri et al20 demonstrated that an entire hair follicle epithelium and interfollicular epidermis can be regenerated using cultured bulge stem cells. The cultured bulge stem cells were mixed with dermal papilla cells from neonatal rat vibrissae and engrafted into a silicone chamber implanted on the backs of severe combined immune deficient (SCID) mice. The grafts exhibited tufts of hair as well as a complete interfollicular epidermis at 4 weeks after transplantation.20 Thus, these bulge stem cells have the potential to treat male androgenic alopecia and female pattern hair loss. Bulge stem cells also have been shown to accelerate wound healing.21 Additionally, autologous melanocytic stem cells located at the hair follicle bulge are effective for treating vitiligo and are being investigated for the treatment of hair graying.22
Induced Pluripotent Stem Cells
Given the ethical concerns that surround the procurement and use of embryonic stem cells, efforts have been made to retrieve pluripotent stem cells from adults. A major breakthrough occurred in 2006 when researchers altered the genes of specialized adult mouse cells to cause dedifferentiation and the return to an embryoniclike stem cell state.23 Mouse somatic cells were reprogrammed through the activation of a combination of transcription factors. The resulting cells were termed induced pluripotent stem cells (iPSCs) and have since been recreated in human cell lines. The discovery of iPSCs precipitated a translational science revolution. Physician-scientists sought ways to apply the reprogrammed cells to the pathophysiology of obscure diseases, examination of drug targets, and regeneration of human tissue.24 Tissue regeneration via induced naïve somatic cells has shown promise as a future method to treat neurologic, cardiovascular, and ophthalmologic diseases.25
As the technology of cultivating and identifying optimal sources of iPSCs continues to advance, stem cell–based treatments have evolved as leading prospects in the field of biogerontology.26-29 Although much of the research in antiaging medicine has utilized iPSCs to reprogram cell senescence, the altering of iPSCs at a cellular level also allows for the stimulation of collagen synthesis. This potential for collagen generation may have direct applicability in dermatologic practice, particularly for aesthetic treatments.
Much of the research into iPSC-derived collagen has focused on genodermatoses. Itoh et al30 examined the creation of collagen through iPSCs to identify possible treatments for recessive dystrophic epidermolysis bullosa (DEB). Recessive DEB is characterized by mutations in the COL7A1 gene, which encodes type VII collagen, a basement membrane protein and component of the anchoring fibrils essential for skin integrity.31 Itoh et al30 began with source cells obtained from a skin biopsy. The cells were dedifferentiated to iPSCs and then induced into dermal fibroblasts according to the methods established in prior studies of embryonic stem cells, namely with the use of ascorbic acid and transforming growth factor b. The newly formed fibroblasts were determined to be functional based on their ability to synthesize mature type VII collagen.30 Once the viability of the iPSC-derived fibroblasts was confirmed in vitro, the cells were further tested through combination with human keratinocytes on SCID mice. The human keratinocytes grew together with the iPSC-derived fibroblasts, producing type VII collagen in the basement membrane zone and creating an epidermis with the normal markers.30 Similarly, Robbins et al32 utilized SCID mice to successfully demonstrate that the transfection of keratinocytes from patients with junctional epidermolysis bullosa into SCID mice produced phenotypically normal skin.
Sebastiano et al33 combined the concepts of iPSCs and genome editing in another study of recessive DEB. The investigators first cultured iPSCs from biopsies of affected patients. After deriving iPSCs and correcting their mutation via adenovirus-associated viral gene editing, the COL7A1 mutation-free cells were differentiated into keratinocytes. These iPSC-derived keratinocytes were subsequently grafted onto mice, which led to the production of wild-type collagen VII and a stratified epidermis. Despite this successful outcome, the grafts of iPSC-derived epidermis did not survive longer than 1 month.33
One of the many obstacles facing the practical use of stem cells is their successful incorporation into human tissue. A possible solution was uncovered by Zhang et al34 who examined iPSC-derived MSCs. Mesenchymal stem cells communicate via paracrine mechanisms, whereby exosomes containing RNA and proteins are released to potentiate a regenerative effect.35 Zhang et al34 found that injecting exosomes from human iPSC-derived MSCs into the wound sites of rats stimulated the production of type I collagen, type III collagen, and elastin. The wound sites demonstrated accelerated closure, narrower scar widths, and increased collagen maturity.
Understanding the role that local environment plays in stem cell differentiation, Xu et al36 aimed to create an extracellular scaffold to induce fibroblast behavior from iPSCs. The authors engineered a framework similar to the normal extracellular membrane using proteoglycans, glycosaminoglycans, fibrinogen, and connective tissue growth factor. The iPSCs were then applied to the scaffolding, which led to successful fibroblast differentiation and type I collagen synthesis.36 This use of local biosignaling cues holds important ramifications for controlling the fate of stem cells that have been introduced into a new environment.
Although the application of iPSCs in clinical dermatology has yet to be achieved, progress in the field is moving at a rapid pace. Several logistical elements require further mastery before therapeutics can be delivered. These areas include the optimal environment for iPSC differentiation, methods for maximization of graft survival, and different modes of transplanting iPSC-derived cells into patients. In cosmetic practice, success will depend on intradermal injections of collagen-producing iPSC-derived cells that possess long-term proliferative potential. Current research in mice models has demonstrated viability up to 16 weeks after intradermal injection of such cells.37
Plant Stem Cells
In discussing the dermatologic applications of stem cell technology, clinicians should be aware of the plant stem cell products that have become a popular cosmeceutical trend. Companies advertise plant cells as a natural source of regenerative cells that can induce rejuvenation in human skin; however, there are no significant data to indicate that plant stem cells encourage or activate cellular growth in humans. Indeed, for stem cells to differentiate and produce viable components, the cells must first be incorporated as living components in the host tissue. Because plant stem cells do not survive in human tissue and plant cell cytokines fail to interact with the receptors on human cells, their current value in cosmeceuticals may be overstated.
Platelet-Rich Plasma
Platelet-rich plasma also is commonly associated with stem cell therapy, as PRP is known to potentiate stem cell proliferation, migration, and differentiation. However, PRP does not contain stem cells and is instead autologous plasma concentrated with platelets. In fact, platelets cannot even be classified as cells given that they lack a nucleus; platelets are considered cell fragments. The regenerative potential of PRP can be attributed to the growth factors released from platelets, which play an important role in tissue regeneration and repair. Platelet-rich plasma currently is being used in dermatology for skin rejuvenation (reduction of wrinkles and furrows) and treatment of acne scars.38 There also is evidence supporting the effectiveness of PRP for alopecia and wound therapy, as growth factors play a vital role in hair growth and wound healing.38 Apart from the use of PRP on its own, it can be used as a supplement to enhance the effects of antiaging procedures such as microneedling.39
Future Directions
Multipotent stem cells and iPSCs discussed herein provide much promise in the field of regenerative dermatology. They are increasingly accessible and circumvent the use of ethically questionable embryonic stem cells. Although there is a general consensus on the great potential of stem cells for treating aesthetic skin conditions, high-quality randomized controlled trials remain scarce within the literature. Recognizing and optimizing these opportunities remains the next step in the future delivery of evidence-based care in regenerative dermatology.
- Thomas ED, Lochte HL, Lu WC, et al. Intravenous infusion of bone marrow in patients receiving radiation and chemotherapy. N Engl J Med. 1957;257:491-496.
- Ogliari KS, Marinowic D, Brum DE, et al. Stem cells in dermatology. An Bras Dermatol. 2014;89:286-291.
- Xu C, Inokuma MS, Denham J, et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol. 2001;19:971-974.
- Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7:211-228.
- Badiavas EV, Falanga V. Treatment of chronic wounds with bone marrow-derived cells. Arch Dermatol. 2003;139:510-516.
- Ibrahim ZA, Eltatawy RA, Ghaly NR, et al. Autologous bone marrow stem cells in atrophic acne scars: a pilot study. J Dermatolog Treat. 2015;26:260-265.
- Broxmeyer HE, Douglas GW, Hangoc G, et al. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc Natl Acad Sci U S A. 1989;86:3828-3832.
- Gluckman E, Rocha V, Boyer-Chammard A, et al. Outcome of cord-blood transplantation from related and unrelated donors. Eurocord Transplant Group and the European Blood and Marrow Transplantation Group. N Engl J Med. 1997;337:373-381.
- Valerio IL, Sabino JM, Dearth CL. Plastic surgery challenges in war wounded II: regenerative medicine. Adv Wound Care (New Rochelle). 2016;5:412-419.
- Vescarelli E, D’Amici S, Onesti MG, et al. Adipose-derived stem cell: an innovative therapeutic approach in systemic sclerosis and Parry-Romberg syndrome. CellR4. 2014;2:E791-E797.
- Yoshimura K, Sato K, Aoi N, et al. Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg. 2008;32:48-55.
- Grabin S, Antes G, Stark GB, et al. Cell-assisted lipotransfer: a critical appraisal of the evidence. Dtsch Arztebl Int. 2015;112:255.
- Zhou Y, Wang J, Li H, et al. Efficacy and safety of cell-assisted lipotransfer: a systematic review and meta-analysis. Plast Reconstr Surg. 2016;137:E44-E57.
- Toma JG, Akhavan M, Fernandes KJL, et al. Isolation of multipotent adult stem cells from the dermis of mammalian skin. Nat Cell Biol. 2001;3:778-784.
- Toma JG, McKenzie IA, Bagli D, et al. Isolation and characterization of multipotent skin-derived precursors from human skin. Stem Cells. 2005;23:727-737.
- Homicz MR, Watson D. Review of injectable materials for soft tissue augmentation. Facial Plast Surg. 2004;20:21-29.
- Kumar S, Mahajan BB, Kaur S, et al. Autologous therapies in dermatology. J Clin Aesthet Dermatol. 2014;7:38-45.
- Schmidt C. FDA approves first cell therapy for wrinkle-free visage. Nat Biotech. 2011;29:674-675.
- Gentile P, Scioli MG, Bielli A, et al. Stem cells from human hair follicles: first mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017;4:58.
- Sugiyama-Nakagiri Y, Akiyama M, Shimizu H. Hair follicle stem cell-targeted gene transfer and reconstitution system. Gene Ther. 2006;13:732-737.
- Heidari F, Yari A, Rasoolijazi H, et al. Bulge hair follicle stem cells accelerate cutaneous wound healing in rats. Wounds. 2016;28:132-141.
- Lee JH, Fisher DE. Melanocyte stem cells as potential therapeutics in skin disorders. Expert Opin Biol Ther. 2014;14:1-11.
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676.
- Singh VK, Kalsan M, Kumar N, et al. Induced pluripotent stem cells: applications in regenerative medicine, disease modeling, and drug discovery. Front Cell Dev Biol. 2015;3:2.
- Aoi T. 10th anniversary of iPS cells: The challenges that lie ahead. J Biochem. 2016;160:121-129.
- Lowry WE, Plath K. The many ways to make an iPS cell. Nat Biotechnol. 2008;26:1246-1248.
- Kim K, Doi A, Wen B, et al. Epigenetic memory in induced pluripotent stem cells. Nature. 2010;467:285-290.
- Gafni O, Weinberger L, Mansour AA, et al. Derivation of novel human ground state naive pluripotent stem cells. Nature. 2013;504:282-286.
- Pareja-Galeano H, Sanchis-Gomar F, Pérez LM, et al. IPSCs-based anti-aging therapies: Recent discoveries and future challenges. Ageing Res Rev. 2016;27:37-41.
- Itoh M, Umegaki-Arao N, Guo Z, et al. Generation of 3D skin equivalents fully reconstituted from human induced pluripotent stem cells (iPSCs). PLoS One. 2013;8:e77673.
- Nyström A, Velati D, Mittapalli VR, et al. Collagen VII plays a dual role in wound healing. J Clin Invest. 2013;123:3498-3509.
- Robbins PB, Lin Q, Goodnough JB, et al. In vivo restoration of laminin 5 β3 expression and function in junctional epidermolysis bullosa. Proc Natl Acad Sci. 2001;98:5193-5198.
- Sebastiano V, Zhen HH, Haddad B, et al. Human COL7A1-corrected induced pluripotent stem cells for the treatment of recessive dystrophic epidermolysis bullosa. Sci Transl Med. 2014;6:264ra163.
- Zhang J, Guan J, Niu X, et al. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J Transl Med. 2015;13:49.
- Pap E, Pállinger É, Pásztói M, et al. Highlights of a new type of intercellular communication: microvesicle-based information transfer. Inflamm Res. 2009;58:1-8.
- Xu R, Taskin MB, Rubert M, et al. hiPS-MSCs differentiation towards fibroblasts on a 3D ECM mimicking scaffold. Sci Rep. 2015;5:8480.
- Wenzel D, Bayerl J, Nyström A, et al. Genetically corrected iPSCs as cell therapy for recessive dystrophic epidermolysis bullosa. Sci Transl Med. 2014;6:264ra165.
- Bednarska K, Kieszek R, Domagała P, et al. The use of platelet-rich-plasma in aesthetic and regenerative medicine. MEDtube Science. 2015;2:8-15.
- Hashim PW, Levy Z, Cohen JL, et al. Microneedling therapy with and without platelet-rich plasma. Cutis. 2017;99:239-242.
- Thomas ED, Lochte HL, Lu WC, et al. Intravenous infusion of bone marrow in patients receiving radiation and chemotherapy. N Engl J Med. 1957;257:491-496.
- Ogliari KS, Marinowic D, Brum DE, et al. Stem cells in dermatology. An Bras Dermatol. 2014;89:286-291.
- Xu C, Inokuma MS, Denham J, et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol. 2001;19:971-974.
- Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7:211-228.
- Badiavas EV, Falanga V. Treatment of chronic wounds with bone marrow-derived cells. Arch Dermatol. 2003;139:510-516.
- Ibrahim ZA, Eltatawy RA, Ghaly NR, et al. Autologous bone marrow stem cells in atrophic acne scars: a pilot study. J Dermatolog Treat. 2015;26:260-265.
- Broxmeyer HE, Douglas GW, Hangoc G, et al. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc Natl Acad Sci U S A. 1989;86:3828-3832.
- Gluckman E, Rocha V, Boyer-Chammard A, et al. Outcome of cord-blood transplantation from related and unrelated donors. Eurocord Transplant Group and the European Blood and Marrow Transplantation Group. N Engl J Med. 1997;337:373-381.
- Valerio IL, Sabino JM, Dearth CL. Plastic surgery challenges in war wounded II: regenerative medicine. Adv Wound Care (New Rochelle). 2016;5:412-419.
- Vescarelli E, D’Amici S, Onesti MG, et al. Adipose-derived stem cell: an innovative therapeutic approach in systemic sclerosis and Parry-Romberg syndrome. CellR4. 2014;2:E791-E797.
- Yoshimura K, Sato K, Aoi N, et al. Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg. 2008;32:48-55.
- Grabin S, Antes G, Stark GB, et al. Cell-assisted lipotransfer: a critical appraisal of the evidence. Dtsch Arztebl Int. 2015;112:255.
- Zhou Y, Wang J, Li H, et al. Efficacy and safety of cell-assisted lipotransfer: a systematic review and meta-analysis. Plast Reconstr Surg. 2016;137:E44-E57.
- Toma JG, Akhavan M, Fernandes KJL, et al. Isolation of multipotent adult stem cells from the dermis of mammalian skin. Nat Cell Biol. 2001;3:778-784.
- Toma JG, McKenzie IA, Bagli D, et al. Isolation and characterization of multipotent skin-derived precursors from human skin. Stem Cells. 2005;23:727-737.
- Homicz MR, Watson D. Review of injectable materials for soft tissue augmentation. Facial Plast Surg. 2004;20:21-29.
- Kumar S, Mahajan BB, Kaur S, et al. Autologous therapies in dermatology. J Clin Aesthet Dermatol. 2014;7:38-45.
- Schmidt C. FDA approves first cell therapy for wrinkle-free visage. Nat Biotech. 2011;29:674-675.
- Gentile P, Scioli MG, Bielli A, et al. Stem cells from human hair follicles: first mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017;4:58.
- Sugiyama-Nakagiri Y, Akiyama M, Shimizu H. Hair follicle stem cell-targeted gene transfer and reconstitution system. Gene Ther. 2006;13:732-737.
- Heidari F, Yari A, Rasoolijazi H, et al. Bulge hair follicle stem cells accelerate cutaneous wound healing in rats. Wounds. 2016;28:132-141.
- Lee JH, Fisher DE. Melanocyte stem cells as potential therapeutics in skin disorders. Expert Opin Biol Ther. 2014;14:1-11.
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676.
- Singh VK, Kalsan M, Kumar N, et al. Induced pluripotent stem cells: applications in regenerative medicine, disease modeling, and drug discovery. Front Cell Dev Biol. 2015;3:2.
- Aoi T. 10th anniversary of iPS cells: The challenges that lie ahead. J Biochem. 2016;160:121-129.
- Lowry WE, Plath K. The many ways to make an iPS cell. Nat Biotechnol. 2008;26:1246-1248.
- Kim K, Doi A, Wen B, et al. Epigenetic memory in induced pluripotent stem cells. Nature. 2010;467:285-290.
- Gafni O, Weinberger L, Mansour AA, et al. Derivation of novel human ground state naive pluripotent stem cells. Nature. 2013;504:282-286.
- Pareja-Galeano H, Sanchis-Gomar F, Pérez LM, et al. IPSCs-based anti-aging therapies: Recent discoveries and future challenges. Ageing Res Rev. 2016;27:37-41.
- Itoh M, Umegaki-Arao N, Guo Z, et al. Generation of 3D skin equivalents fully reconstituted from human induced pluripotent stem cells (iPSCs). PLoS One. 2013;8:e77673.
- Nyström A, Velati D, Mittapalli VR, et al. Collagen VII plays a dual role in wound healing. J Clin Invest. 2013;123:3498-3509.
- Robbins PB, Lin Q, Goodnough JB, et al. In vivo restoration of laminin 5 β3 expression and function in junctional epidermolysis bullosa. Proc Natl Acad Sci. 2001;98:5193-5198.
- Sebastiano V, Zhen HH, Haddad B, et al. Human COL7A1-corrected induced pluripotent stem cells for the treatment of recessive dystrophic epidermolysis bullosa. Sci Transl Med. 2014;6:264ra163.
- Zhang J, Guan J, Niu X, et al. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J Transl Med. 2015;13:49.
- Pap E, Pállinger É, Pásztói M, et al. Highlights of a new type of intercellular communication: microvesicle-based information transfer. Inflamm Res. 2009;58:1-8.
- Xu R, Taskin MB, Rubert M, et al. hiPS-MSCs differentiation towards fibroblasts on a 3D ECM mimicking scaffold. Sci Rep. 2015;5:8480.
- Wenzel D, Bayerl J, Nyström A, et al. Genetically corrected iPSCs as cell therapy for recessive dystrophic epidermolysis bullosa. Sci Transl Med. 2014;6:264ra165.
- Bednarska K, Kieszek R, Domagała P, et al. The use of platelet-rich-plasma in aesthetic and regenerative medicine. MEDtube Science. 2015;2:8-15.
- Hashim PW, Levy Z, Cohen JL, et al. Microneedling therapy with and without platelet-rich plasma. Cutis. 2017;99:239-242.
Practice Points
- Multipotent stem cells derived from the bone marrow, umbilical cord, adipose tissue, dermis, and hair follicle bulge show promise in tissue regeneration for various dermatologic conditions and aesthetic applications.
- Induced pluripotent stem cells, progenitor cells that result from the dedifferentiation of specialized adult cells, have potential for collagen generation.
Cosmetic Corner: Dermatologists Weigh in on Pigment Correctors
To improve patient care and outcomes, leading dermatologists offered their recommendations on pigment correctors. Consideration must be given to:
- dEp Patch Full Face Mask
Activaderm, Inc
“This product uses microcurrent to push vitamin C into the skin. Vitamin C, a known antioxidant that usually has a difficult time passing through the stratum corneum, corrects pigmentary abnormalities. The product also comes with a botanical pigment corrector.”—Gary Goldenberg, MD, New York, New York
- De-Spot Skin Brightening Corrector
Peter Thomas Roth Labs LLC
“This product is a useful over-the-counter adjunct to prescription-strength hydroquinone, with niacinamide as one of the active ingredients.”—Shari Lipner, MD, PhD, New York, New York
- Glytone Dark Spot Corrector
Pierre Fabre Laboratories
“With 2% hydroquinone, glycolic acid, and kojic acid, you have a highly effective combination of ingredients that work synergistically to lighten areas of skin discoloration.”—Jeannette Graf, MD, Great Neck, New York
Cutis invites readers to send us their recommendations. Bar soap, lip plumper, and night cream will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on pigment correctors. Consideration must be given to:
- dEp Patch Full Face Mask
Activaderm, Inc
“This product uses microcurrent to push vitamin C into the skin. Vitamin C, a known antioxidant that usually has a difficult time passing through the stratum corneum, corrects pigmentary abnormalities. The product also comes with a botanical pigment corrector.”—Gary Goldenberg, MD, New York, New York
- De-Spot Skin Brightening Corrector
Peter Thomas Roth Labs LLC
“This product is a useful over-the-counter adjunct to prescription-strength hydroquinone, with niacinamide as one of the active ingredients.”—Shari Lipner, MD, PhD, New York, New York
- Glytone Dark Spot Corrector
Pierre Fabre Laboratories
“With 2% hydroquinone, glycolic acid, and kojic acid, you have a highly effective combination of ingredients that work synergistically to lighten areas of skin discoloration.”—Jeannette Graf, MD, Great Neck, New York
Cutis invites readers to send us their recommendations. Bar soap, lip plumper, and night cream will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on pigment correctors. Consideration must be given to:
- dEp Patch Full Face Mask
Activaderm, Inc
“This product uses microcurrent to push vitamin C into the skin. Vitamin C, a known antioxidant that usually has a difficult time passing through the stratum corneum, corrects pigmentary abnormalities. The product also comes with a botanical pigment corrector.”—Gary Goldenberg, MD, New York, New York
- De-Spot Skin Brightening Corrector
Peter Thomas Roth Labs LLC
“This product is a useful over-the-counter adjunct to prescription-strength hydroquinone, with niacinamide as one of the active ingredients.”—Shari Lipner, MD, PhD, New York, New York
- Glytone Dark Spot Corrector
Pierre Fabre Laboratories
“With 2% hydroquinone, glycolic acid, and kojic acid, you have a highly effective combination of ingredients that work synergistically to lighten areas of skin discoloration.”—Jeannette Graf, MD, Great Neck, New York
Cutis invites readers to send us their recommendations. Bar soap, lip plumper, and night cream will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Juvéderm Voluma for Cheek Rejuvenation



Thread lifts: A face-lift alternative? Or not?
The rise of noninvasive procedures has shifted the aesthetic culture. Patients now are asking for less invasive, less painful, less expensive procedures with short recovery times. Thread-lifts are one of the newest approaches to nonsurgical facial tightening. However, are they of value? Where, and for whom?
The thread-lifts initially came onto the market in the late 1990s but were difficult to use. The nonabsorbable threads had to be anchored into the scalp, temple, and brow region. The anchoring knots were carefully tied and were permanent. The newest technology threads – NovaThreads and the Silhouette Instalift – have recently received Food and Drug Administration clearance and grown in popularity because of their “lunchtime” appeal and their ease of use. Primarily marketed for the neck, jowls, and lower face, these threads – available in various sizes, lengths, and diameters – can be used almost anywhere. The sutures dissolve over time and do not need any anchoring, making it a very simple in-office procedure.
Side effects include mild procedural pain, edema, erythema, bruising, and rarely, suture granuloma formation; and they may need to be replaced. If not done properly, buckling of the skin can occur and superficially placed sutures can be visible.
Similar to fillers that provide a “liquid face-lift,” the down time is minimal. Common side effects include bruising, and patients should refrain from heavy exercise and opening their mouth wide with chewing for 5-7 days (such as eating a large apple). Soreness, particularly in or near the hairline or jaw line, can occur and can last up to 2 weeks. Dimpling in the skin can occur and usually resolves on its own; however, if threads are placed incorrectly, dimpling can cause some disfigurement.
Results can vary based on the tissue laxity, and the type, amount, and location of the threads used. While results have been reported to last 18 months to 2 years, the procedure is not a replacement for fillers. Facial aging is caused by a combination of skeletal, soft tissue, and skin changes that lead to soft tissue laxity and volume loss. Fillers are essential in restoring lost volume in the aging face and are particularly helpful in combination with tissue tightening lasers, face-lifts and the thread-lift procedures. Fillers used in combination with thread-lifts also increase the longevity of the thread-lift because of additional collagen stimulation.
As the procedure is not indicated for severe laxity, thread-lifts also do not replace the traditional face-lift. Tissue is not released from its underlying attachments, and skin contraction and gravitational pull limit its extent of improvement and its longevity.
Long-term success of the thread-lift procedure for facial rejuvenation was evaluated in a retrospective review of 33 patients who underwent the traditional thread-lift procedure alone or in combination with other facial rejuvenation procedures to the brow, mid-face, jowl, and neck published in 2009.1 The study compared results in 10 patients who had a thread-lift alone, 23 who had thread-lifts combined with other procedures, and controls, who were 10 ten patients who had non–thread-lift rejuvenation procedures, which included lipotransfer, chemical peels, and rhytidectomies. Independent, blinded, board-certified facial plastic surgeons evaluated pre- and postoperative photos. Patients were followed-up for a mean of 21 months.
While thread-lifts are a beneficial addition to our armamentarium of noninvasive aesthetic procedures, they have better outcomes and higher patient satisfaction when used in combination with fillers, radiofrequency, and fractional lasers and neuromodulators.
Reference
1. Abraham RF et al. Arch Facial Plast Surg. 2009 May-Jun;11(3):178-83.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
The rise of noninvasive procedures has shifted the aesthetic culture. Patients now are asking for less invasive, less painful, less expensive procedures with short recovery times. Thread-lifts are one of the newest approaches to nonsurgical facial tightening. However, are they of value? Where, and for whom?
The thread-lifts initially came onto the market in the late 1990s but were difficult to use. The nonabsorbable threads had to be anchored into the scalp, temple, and brow region. The anchoring knots were carefully tied and were permanent. The newest technology threads – NovaThreads and the Silhouette Instalift – have recently received Food and Drug Administration clearance and grown in popularity because of their “lunchtime” appeal and their ease of use. Primarily marketed for the neck, jowls, and lower face, these threads – available in various sizes, lengths, and diameters – can be used almost anywhere. The sutures dissolve over time and do not need any anchoring, making it a very simple in-office procedure.
Side effects include mild procedural pain, edema, erythema, bruising, and rarely, suture granuloma formation; and they may need to be replaced. If not done properly, buckling of the skin can occur and superficially placed sutures can be visible.
Similar to fillers that provide a “liquid face-lift,” the down time is minimal. Common side effects include bruising, and patients should refrain from heavy exercise and opening their mouth wide with chewing for 5-7 days (such as eating a large apple). Soreness, particularly in or near the hairline or jaw line, can occur and can last up to 2 weeks. Dimpling in the skin can occur and usually resolves on its own; however, if threads are placed incorrectly, dimpling can cause some disfigurement.
Results can vary based on the tissue laxity, and the type, amount, and location of the threads used. While results have been reported to last 18 months to 2 years, the procedure is not a replacement for fillers. Facial aging is caused by a combination of skeletal, soft tissue, and skin changes that lead to soft tissue laxity and volume loss. Fillers are essential in restoring lost volume in the aging face and are particularly helpful in combination with tissue tightening lasers, face-lifts and the thread-lift procedures. Fillers used in combination with thread-lifts also increase the longevity of the thread-lift because of additional collagen stimulation.
As the procedure is not indicated for severe laxity, thread-lifts also do not replace the traditional face-lift. Tissue is not released from its underlying attachments, and skin contraction and gravitational pull limit its extent of improvement and its longevity.
Long-term success of the thread-lift procedure for facial rejuvenation was evaluated in a retrospective review of 33 patients who underwent the traditional thread-lift procedure alone or in combination with other facial rejuvenation procedures to the brow, mid-face, jowl, and neck published in 2009.1 The study compared results in 10 patients who had a thread-lift alone, 23 who had thread-lifts combined with other procedures, and controls, who were 10 ten patients who had non–thread-lift rejuvenation procedures, which included lipotransfer, chemical peels, and rhytidectomies. Independent, blinded, board-certified facial plastic surgeons evaluated pre- and postoperative photos. Patients were followed-up for a mean of 21 months.
While thread-lifts are a beneficial addition to our armamentarium of noninvasive aesthetic procedures, they have better outcomes and higher patient satisfaction when used in combination with fillers, radiofrequency, and fractional lasers and neuromodulators.
Reference
1. Abraham RF et al. Arch Facial Plast Surg. 2009 May-Jun;11(3):178-83.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
The rise of noninvasive procedures has shifted the aesthetic culture. Patients now are asking for less invasive, less painful, less expensive procedures with short recovery times. Thread-lifts are one of the newest approaches to nonsurgical facial tightening. However, are they of value? Where, and for whom?
The thread-lifts initially came onto the market in the late 1990s but were difficult to use. The nonabsorbable threads had to be anchored into the scalp, temple, and brow region. The anchoring knots were carefully tied and were permanent. The newest technology threads – NovaThreads and the Silhouette Instalift – have recently received Food and Drug Administration clearance and grown in popularity because of their “lunchtime” appeal and their ease of use. Primarily marketed for the neck, jowls, and lower face, these threads – available in various sizes, lengths, and diameters – can be used almost anywhere. The sutures dissolve over time and do not need any anchoring, making it a very simple in-office procedure.
Side effects include mild procedural pain, edema, erythema, bruising, and rarely, suture granuloma formation; and they may need to be replaced. If not done properly, buckling of the skin can occur and superficially placed sutures can be visible.
Similar to fillers that provide a “liquid face-lift,” the down time is minimal. Common side effects include bruising, and patients should refrain from heavy exercise and opening their mouth wide with chewing for 5-7 days (such as eating a large apple). Soreness, particularly in or near the hairline or jaw line, can occur and can last up to 2 weeks. Dimpling in the skin can occur and usually resolves on its own; however, if threads are placed incorrectly, dimpling can cause some disfigurement.
Results can vary based on the tissue laxity, and the type, amount, and location of the threads used. While results have been reported to last 18 months to 2 years, the procedure is not a replacement for fillers. Facial aging is caused by a combination of skeletal, soft tissue, and skin changes that lead to soft tissue laxity and volume loss. Fillers are essential in restoring lost volume in the aging face and are particularly helpful in combination with tissue tightening lasers, face-lifts and the thread-lift procedures. Fillers used in combination with thread-lifts also increase the longevity of the thread-lift because of additional collagen stimulation.
As the procedure is not indicated for severe laxity, thread-lifts also do not replace the traditional face-lift. Tissue is not released from its underlying attachments, and skin contraction and gravitational pull limit its extent of improvement and its longevity.
Long-term success of the thread-lift procedure for facial rejuvenation was evaluated in a retrospective review of 33 patients who underwent the traditional thread-lift procedure alone or in combination with other facial rejuvenation procedures to the brow, mid-face, jowl, and neck published in 2009.1 The study compared results in 10 patients who had a thread-lift alone, 23 who had thread-lifts combined with other procedures, and controls, who were 10 ten patients who had non–thread-lift rejuvenation procedures, which included lipotransfer, chemical peels, and rhytidectomies. Independent, blinded, board-certified facial plastic surgeons evaluated pre- and postoperative photos. Patients were followed-up for a mean of 21 months.
While thread-lifts are a beneficial addition to our armamentarium of noninvasive aesthetic procedures, they have better outcomes and higher patient satisfaction when used in combination with fillers, radiofrequency, and fractional lasers and neuromodulators.
Reference
1. Abraham RF et al. Arch Facial Plast Surg. 2009 May-Jun;11(3):178-83.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
Using oral and topical cosmeceuticals to prevent and treat skin aging, Part I
It is important for dermatologists to recognize patients at an increased risk of skin aging early enough to initiate countermeasures. “Wrinkle-prone” skin types can be identified easily through use of the Baumann Skin Type Indicator Questionnaire.1 The wrinkle-prone Baumann skin type is associated with age or with lifestyle factors that increase the risk for promoting skin aging.2 Prevention and treatment of numerous signs of cutaneous aging can be achieved through consistent daily use of oral and topical products suited to the identified specifically wrinkle-prone Baumann skin type.
Skin aging
The numerous causes of skin aging can be divided into two broad categories: intrinsic and extrinsic. Intrinsic aging results from cellular processes that occur over time and is influenced by genetics. Such aging is characterized by decreased function of keratinocytes and fibroblasts, intra- and extracellular accumulation of by-products, reduced function of sirtuins (proteins that regulate cell metabolism and aging), mitochondrial damage, and loss of telomeres.
Extrinsic aging results from environmental exposures that engender cell damage, including UV light, infrared and radiation exposure, air pollution, smoking, tanning beds, alcohol and drug usage, stress, and poor diet. Extrinsic aging occurs as a result of intersecting processes caused by free radicals, DNA damage, glycation, inflammation, and other actions by the immune system. Generally, these factors can be partially mitigated through behavioral change. As much as 80% of facial aging can be ascribed to sun exposure.4 Several mechanisms through which sun exposure promotes aging have been well characterized. DNA damage results when UV light induces covalent bonds between nucleic acid base pairs and forms thymine dimers, which can alter tumor suppressor gene p53 function, thereby increasing the risk of cutaneous cancers and aging.5 UV exposure also yields free radicals that create damaging oxidative stress,6 which can activate the arachidonic acid pathway resulting in inflammation.7 Other skin aging mechanisms are not as well understood.
The cellular role in aging: Keratinocytes and fibroblasts
Keratinocyte cells found in layers that resemble the brick-and-mortar structure of a brick wall compose the epidermis. Each epidermal layer exhibits specific functional roles and characteristics. The top layer of the epidermis, known as the stratum corneum, is notable because it forms the skin barrier. This protective barrier contains cross-linked proteins for strength, antioxidants to protect the cells from free radicals, a bilayer lipid membrane layer to prevent water evaporation from the cells surface, immune cells, antimicrobial peptides, and a natural microbiome. Damage to any layer of the epidermis can unleash a cascade of events that can lead to increased cutaneous aging.
The dermis is composed of fibroblast cells, which synthesize collagen, elastin, hyaluronic acid, heparan sulfate, and other glycosaminoglycans that keep the skin smooth, strong, and healthy. Collagen confers strength, elastin provides elasticity, and the glycosaminoglycans such as hyaluronic acid, heparan sulfate, and dermatan sulfate bind water, impart volume to the skin, and provide support for important cell-to-cell communication.
When keratinocytes and fibroblasts age, they may no longer respond to cellular signals such as growth factors. The primary aim of any antiaging skin care regimen is to protect and rejuvenate these key skin cells.
Cellular damage that contributes to skin aging
The accumulated damage from intrinsic and extrinsic factors yields keratinocytes and fibroblasts that fail to produce important cellular components as well as they did when they were younger. Cellular factors that age cells include nuclear DNA damage, mitochondrial DNA damage, diminished lysosomal function, structural impairment of proteins, and damage to cell membranes. This harm occurs because of the direct effects of UV radiation, pollution, toxins, free radicals (oxidation), glycation, and inflammation.
Preventing and treating DNA damage
DNA damage presents as thymine-thymine dimers, pyrimidine-pyrimidine dimers, impaired telomeres, or other mutations. Broad-spectrum sunscreens and sun avoidance are important steps in preventing DNA damage induced from exposure to UV radiation. Other cosmeceutical agents have been designed to hinder the effects of UV radiation or to foster DNA repair. Besides sunscreen, the key members of the dermatologic armamentarium against DNA damage are various antioxidants. Data have been gathered over the last few decades that support the protective effects of antioxidants such as polypodium leucotomos,ascorbic acid, and green tea. Other antioxidants are associated with less data, but hypothetically should deliver similar benefits.
Polypodium leucotomos (PL), an oral extract derived from ferns, has been demonstrated to display photoprotective effects at an oral dose of 7.5 mg. PL has consistently exhibited antitumor and skin protective effects.8 A 2004 study in humans revealed that two oral doses of PL contributed to a significant reduction in DNA damage after UV exposure,9 and a 2017 study showed that PL protected skin DNA from UVB.10 Although PL has been linked to topical benefits, it is the oral form that is most often used to protect skin.
Ascorbic acid, also known as vitamin C, has been amply demonstrated to confer benefits when given both orally and topically. An acidic environment is necessary for optimal absorption. Topical application of ascorbic acid, along with vitamin E and ferulic acid, has been demonstrated to decrease the formation of thymine dimers.11 Unlike other antioxidants, ascorbic acid also stimulates procollagen genes in fibroblasts to increase collagen synthesis.12
EpiGalloCatechin-3-O-Gallate (also known as EGCG), the primary active constituent of green tea, has been demonstrated to induce IL-12 to increase the production of enzymes that repair UV-induced DNA damage.15 The proven photoprotective effects of topical and oral green tea include reducing UV-induced erythema, decreasing sunburn cell formation, and attenuating DNA damage.16
Preventing and treating mitochondrial DNA damage
UV radiation elicits mitochondrial DNA damage known as the “common deletion.”17 Damaged mitochondria produce harmful free radicals known as reactive oxygen species. Mitochondria damage caused by ROS decreases the mitochondria’s ability to generate ATP energy, which is necessary for DNA repair and other cellular processes.
Free radicals and UV radiation damage mitochondria, as does normal cellular metabolism. The range of damage includes mitochondrial DNA impairment, loss of mitochondrial enzymes, and decreased ATP production. This leads to less energy for DNA repair and other reparative processes. While there is no established way to reduce mitochondrial damage once it has occurred, several research initiatives to achieve this end are underway. Currently, protecting the mitochondria from harm with sunscreens and antioxidants is the best option.
Antioxidants are effective in preventing the damaging effects of free radicals on vulnerable mitochondria. As a component of the mitochondrial respiratory chain and an antioxidant itself, coenzyme Q10 is particularly useful in this role. CoQ10 is available in both oral and topical formulations. Oral forms should be taken only in the morning because of a caffeine-like effect. Topical forms of CoQ10 have a dark yellow color that may be unappealing to patients. Polypodium leucotomos has been shown to lower the number of common deletions found in the mitochondria of irradiated keratinocytes and fibroblasts.18 The oral form is recommended. Another potent antioxidant, curcumin, is being studied for mitochondrial protective properties.19 Its strong yellow color and smell render it better suited for oral use although many companies are trying to develop cosmetically elegant topical formulations.
Scavenging free radicals
Ultraviolet light, pollution, and other insults engender free radical formation. Even sunscreen use has been linked to increased production of free radicals. Free radicals, also known as reactive oxygen species, harm cells in many ways including mitochondrial damage, DNA mutations, glycation, lysosomal damage, and oxidation of important lipids and other cellular components such as proteins. Antioxidants present various beneficial effects including scavenging free radicals, decreasing activation of mitogen-activated protein kinases, chelation of copper required by tyrosinase, and suppression of inflammatory factors, such as nuclear factor (NF)-
In summary, skin aging has many causes. Although they are not all understood, some of the processes have been elucidated. Next month, this column will focus on the prevention and treatment of inflammation and glycation, as well as reversing the effects of aging on skin cells.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014). She also wrote a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems LLC.
References
1. Baumann, Leslie S. “Cosmeceuticals and cosmetic ingredients” (New York: McGraw-Hill Education / Medical, 2014).
2. Baumann, Leslie S. The Baumann Skin Typing System in “Textbook of Aging Skin” (New York: Springer-Verlag Berlin Heidelberg, 2017). pp. 1579-94.
3. Storm A et al. J Am Acad Dermatol. 2008 Dec;59(6):975-80.
4. Uitto J. N Engl J Med. 1997 Nov 13;337(20):1463-5.
5. Tornaletti S et al. Science. 1994;263(5152):1436-8.
6. Bickers D et al. J. Investig. Dermatol. 2006;126(12):2565-75.
7. Yaar M et al. Br J Dermatol. 2007 Nov;157(5):874-87.
8. Parrado C et al. Int J Mol Sci. 2016 Jun 29;17(7). pii: E1026.
9. Middelkamp-Hup MA et al. J Am Acad Dermatol. 2004 Dec;51(6):910-8.
10. Kohli I et al. J Am Acad Dermatol. 2017 Jul;77(1):33-41.
11. Murray JC et al. J Am Acad Dermatol. 2008;59(3):418-25.
12. Geesin JC et al. J Invest Dermatol. 1988 Apr;90(4):420-4.
13. Thompson BC et al. PLoS One. 2015 Feb 6;10(2):e0117491.
14. Surjana D et al. Carcinogenesis. 2013 May;34(5):1144-9.
15. Meeran SM et al. Cancer Res. 2006 May 15;66(10):5512-20.
16. Elmets CA et al. J Am Acad Dermatol. 2001 Mar;44(3):425-32.
17. Berneburg M et al. J Invest Dermatol. 2004 May;122(5):1277-83.
18. Villa A et al. J Am Acad Dermatol. 2010 Mar;62(3):511-3.
19. Trujillo J et al. Arch Pharm Chem Life Sci. 2014. doi: 10.1002/ardp.2014002662014.
20. Muthusam V et al. Arch Dermatol Res. 2010 Jan;302(1):5-17.
It is important for dermatologists to recognize patients at an increased risk of skin aging early enough to initiate countermeasures. “Wrinkle-prone” skin types can be identified easily through use of the Baumann Skin Type Indicator Questionnaire.1 The wrinkle-prone Baumann skin type is associated with age or with lifestyle factors that increase the risk for promoting skin aging.2 Prevention and treatment of numerous signs of cutaneous aging can be achieved through consistent daily use of oral and topical products suited to the identified specifically wrinkle-prone Baumann skin type.
Skin aging
The numerous causes of skin aging can be divided into two broad categories: intrinsic and extrinsic. Intrinsic aging results from cellular processes that occur over time and is influenced by genetics. Such aging is characterized by decreased function of keratinocytes and fibroblasts, intra- and extracellular accumulation of by-products, reduced function of sirtuins (proteins that regulate cell metabolism and aging), mitochondrial damage, and loss of telomeres.
Extrinsic aging results from environmental exposures that engender cell damage, including UV light, infrared and radiation exposure, air pollution, smoking, tanning beds, alcohol and drug usage, stress, and poor diet. Extrinsic aging occurs as a result of intersecting processes caused by free radicals, DNA damage, glycation, inflammation, and other actions by the immune system. Generally, these factors can be partially mitigated through behavioral change. As much as 80% of facial aging can be ascribed to sun exposure.4 Several mechanisms through which sun exposure promotes aging have been well characterized. DNA damage results when UV light induces covalent bonds between nucleic acid base pairs and forms thymine dimers, which can alter tumor suppressor gene p53 function, thereby increasing the risk of cutaneous cancers and aging.5 UV exposure also yields free radicals that create damaging oxidative stress,6 which can activate the arachidonic acid pathway resulting in inflammation.7 Other skin aging mechanisms are not as well understood.
The cellular role in aging: Keratinocytes and fibroblasts
Keratinocyte cells found in layers that resemble the brick-and-mortar structure of a brick wall compose the epidermis. Each epidermal layer exhibits specific functional roles and characteristics. The top layer of the epidermis, known as the stratum corneum, is notable because it forms the skin barrier. This protective barrier contains cross-linked proteins for strength, antioxidants to protect the cells from free radicals, a bilayer lipid membrane layer to prevent water evaporation from the cells surface, immune cells, antimicrobial peptides, and a natural microbiome. Damage to any layer of the epidermis can unleash a cascade of events that can lead to increased cutaneous aging.
The dermis is composed of fibroblast cells, which synthesize collagen, elastin, hyaluronic acid, heparan sulfate, and other glycosaminoglycans that keep the skin smooth, strong, and healthy. Collagen confers strength, elastin provides elasticity, and the glycosaminoglycans such as hyaluronic acid, heparan sulfate, and dermatan sulfate bind water, impart volume to the skin, and provide support for important cell-to-cell communication.
When keratinocytes and fibroblasts age, they may no longer respond to cellular signals such as growth factors. The primary aim of any antiaging skin care regimen is to protect and rejuvenate these key skin cells.
Cellular damage that contributes to skin aging
The accumulated damage from intrinsic and extrinsic factors yields keratinocytes and fibroblasts that fail to produce important cellular components as well as they did when they were younger. Cellular factors that age cells include nuclear DNA damage, mitochondrial DNA damage, diminished lysosomal function, structural impairment of proteins, and damage to cell membranes. This harm occurs because of the direct effects of UV radiation, pollution, toxins, free radicals (oxidation), glycation, and inflammation.
Preventing and treating DNA damage
DNA damage presents as thymine-thymine dimers, pyrimidine-pyrimidine dimers, impaired telomeres, or other mutations. Broad-spectrum sunscreens and sun avoidance are important steps in preventing DNA damage induced from exposure to UV radiation. Other cosmeceutical agents have been designed to hinder the effects of UV radiation or to foster DNA repair. Besides sunscreen, the key members of the dermatologic armamentarium against DNA damage are various antioxidants. Data have been gathered over the last few decades that support the protective effects of antioxidants such as polypodium leucotomos,ascorbic acid, and green tea. Other antioxidants are associated with less data, but hypothetically should deliver similar benefits.
Polypodium leucotomos (PL), an oral extract derived from ferns, has been demonstrated to display photoprotective effects at an oral dose of 7.5 mg. PL has consistently exhibited antitumor and skin protective effects.8 A 2004 study in humans revealed that two oral doses of PL contributed to a significant reduction in DNA damage after UV exposure,9 and a 2017 study showed that PL protected skin DNA from UVB.10 Although PL has been linked to topical benefits, it is the oral form that is most often used to protect skin.
Ascorbic acid, also known as vitamin C, has been amply demonstrated to confer benefits when given both orally and topically. An acidic environment is necessary for optimal absorption. Topical application of ascorbic acid, along with vitamin E and ferulic acid, has been demonstrated to decrease the formation of thymine dimers.11 Unlike other antioxidants, ascorbic acid also stimulates procollagen genes in fibroblasts to increase collagen synthesis.12
EpiGalloCatechin-3-O-Gallate (also known as EGCG), the primary active constituent of green tea, has been demonstrated to induce IL-12 to increase the production of enzymes that repair UV-induced DNA damage.15 The proven photoprotective effects of topical and oral green tea include reducing UV-induced erythema, decreasing sunburn cell formation, and attenuating DNA damage.16
Preventing and treating mitochondrial DNA damage
UV radiation elicits mitochondrial DNA damage known as the “common deletion.”17 Damaged mitochondria produce harmful free radicals known as reactive oxygen species. Mitochondria damage caused by ROS decreases the mitochondria’s ability to generate ATP energy, which is necessary for DNA repair and other cellular processes.
Free radicals and UV radiation damage mitochondria, as does normal cellular metabolism. The range of damage includes mitochondrial DNA impairment, loss of mitochondrial enzymes, and decreased ATP production. This leads to less energy for DNA repair and other reparative processes. While there is no established way to reduce mitochondrial damage once it has occurred, several research initiatives to achieve this end are underway. Currently, protecting the mitochondria from harm with sunscreens and antioxidants is the best option.
Antioxidants are effective in preventing the damaging effects of free radicals on vulnerable mitochondria. As a component of the mitochondrial respiratory chain and an antioxidant itself, coenzyme Q10 is particularly useful in this role. CoQ10 is available in both oral and topical formulations. Oral forms should be taken only in the morning because of a caffeine-like effect. Topical forms of CoQ10 have a dark yellow color that may be unappealing to patients. Polypodium leucotomos has been shown to lower the number of common deletions found in the mitochondria of irradiated keratinocytes and fibroblasts.18 The oral form is recommended. Another potent antioxidant, curcumin, is being studied for mitochondrial protective properties.19 Its strong yellow color and smell render it better suited for oral use although many companies are trying to develop cosmetically elegant topical formulations.
Scavenging free radicals
Ultraviolet light, pollution, and other insults engender free radical formation. Even sunscreen use has been linked to increased production of free radicals. Free radicals, also known as reactive oxygen species, harm cells in many ways including mitochondrial damage, DNA mutations, glycation, lysosomal damage, and oxidation of important lipids and other cellular components such as proteins. Antioxidants present various beneficial effects including scavenging free radicals, decreasing activation of mitogen-activated protein kinases, chelation of copper required by tyrosinase, and suppression of inflammatory factors, such as nuclear factor (NF)-
In summary, skin aging has many causes. Although they are not all understood, some of the processes have been elucidated. Next month, this column will focus on the prevention and treatment of inflammation and glycation, as well as reversing the effects of aging on skin cells.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014). She also wrote a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems LLC.
References
1. Baumann, Leslie S. “Cosmeceuticals and cosmetic ingredients” (New York: McGraw-Hill Education / Medical, 2014).
2. Baumann, Leslie S. The Baumann Skin Typing System in “Textbook of Aging Skin” (New York: Springer-Verlag Berlin Heidelberg, 2017). pp. 1579-94.
3. Storm A et al. J Am Acad Dermatol. 2008 Dec;59(6):975-80.
4. Uitto J. N Engl J Med. 1997 Nov 13;337(20):1463-5.
5. Tornaletti S et al. Science. 1994;263(5152):1436-8.
6. Bickers D et al. J. Investig. Dermatol. 2006;126(12):2565-75.
7. Yaar M et al. Br J Dermatol. 2007 Nov;157(5):874-87.
8. Parrado C et al. Int J Mol Sci. 2016 Jun 29;17(7). pii: E1026.
9. Middelkamp-Hup MA et al. J Am Acad Dermatol. 2004 Dec;51(6):910-8.
10. Kohli I et al. J Am Acad Dermatol. 2017 Jul;77(1):33-41.
11. Murray JC et al. J Am Acad Dermatol. 2008;59(3):418-25.
12. Geesin JC et al. J Invest Dermatol. 1988 Apr;90(4):420-4.
13. Thompson BC et al. PLoS One. 2015 Feb 6;10(2):e0117491.
14. Surjana D et al. Carcinogenesis. 2013 May;34(5):1144-9.
15. Meeran SM et al. Cancer Res. 2006 May 15;66(10):5512-20.
16. Elmets CA et al. J Am Acad Dermatol. 2001 Mar;44(3):425-32.
17. Berneburg M et al. J Invest Dermatol. 2004 May;122(5):1277-83.
18. Villa A et al. J Am Acad Dermatol. 2010 Mar;62(3):511-3.
19. Trujillo J et al. Arch Pharm Chem Life Sci. 2014. doi: 10.1002/ardp.2014002662014.
20. Muthusam V et al. Arch Dermatol Res. 2010 Jan;302(1):5-17.
It is important for dermatologists to recognize patients at an increased risk of skin aging early enough to initiate countermeasures. “Wrinkle-prone” skin types can be identified easily through use of the Baumann Skin Type Indicator Questionnaire.1 The wrinkle-prone Baumann skin type is associated with age or with lifestyle factors that increase the risk for promoting skin aging.2 Prevention and treatment of numerous signs of cutaneous aging can be achieved through consistent daily use of oral and topical products suited to the identified specifically wrinkle-prone Baumann skin type.
Skin aging
The numerous causes of skin aging can be divided into two broad categories: intrinsic and extrinsic. Intrinsic aging results from cellular processes that occur over time and is influenced by genetics. Such aging is characterized by decreased function of keratinocytes and fibroblasts, intra- and extracellular accumulation of by-products, reduced function of sirtuins (proteins that regulate cell metabolism and aging), mitochondrial damage, and loss of telomeres.
Extrinsic aging results from environmental exposures that engender cell damage, including UV light, infrared and radiation exposure, air pollution, smoking, tanning beds, alcohol and drug usage, stress, and poor diet. Extrinsic aging occurs as a result of intersecting processes caused by free radicals, DNA damage, glycation, inflammation, and other actions by the immune system. Generally, these factors can be partially mitigated through behavioral change. As much as 80% of facial aging can be ascribed to sun exposure.4 Several mechanisms through which sun exposure promotes aging have been well characterized. DNA damage results when UV light induces covalent bonds between nucleic acid base pairs and forms thymine dimers, which can alter tumor suppressor gene p53 function, thereby increasing the risk of cutaneous cancers and aging.5 UV exposure also yields free radicals that create damaging oxidative stress,6 which can activate the arachidonic acid pathway resulting in inflammation.7 Other skin aging mechanisms are not as well understood.
The cellular role in aging: Keratinocytes and fibroblasts
Keratinocyte cells found in layers that resemble the brick-and-mortar structure of a brick wall compose the epidermis. Each epidermal layer exhibits specific functional roles and characteristics. The top layer of the epidermis, known as the stratum corneum, is notable because it forms the skin barrier. This protective barrier contains cross-linked proteins for strength, antioxidants to protect the cells from free radicals, a bilayer lipid membrane layer to prevent water evaporation from the cells surface, immune cells, antimicrobial peptides, and a natural microbiome. Damage to any layer of the epidermis can unleash a cascade of events that can lead to increased cutaneous aging.
The dermis is composed of fibroblast cells, which synthesize collagen, elastin, hyaluronic acid, heparan sulfate, and other glycosaminoglycans that keep the skin smooth, strong, and healthy. Collagen confers strength, elastin provides elasticity, and the glycosaminoglycans such as hyaluronic acid, heparan sulfate, and dermatan sulfate bind water, impart volume to the skin, and provide support for important cell-to-cell communication.
When keratinocytes and fibroblasts age, they may no longer respond to cellular signals such as growth factors. The primary aim of any antiaging skin care regimen is to protect and rejuvenate these key skin cells.
Cellular damage that contributes to skin aging
The accumulated damage from intrinsic and extrinsic factors yields keratinocytes and fibroblasts that fail to produce important cellular components as well as they did when they were younger. Cellular factors that age cells include nuclear DNA damage, mitochondrial DNA damage, diminished lysosomal function, structural impairment of proteins, and damage to cell membranes. This harm occurs because of the direct effects of UV radiation, pollution, toxins, free radicals (oxidation), glycation, and inflammation.
Preventing and treating DNA damage
DNA damage presents as thymine-thymine dimers, pyrimidine-pyrimidine dimers, impaired telomeres, or other mutations. Broad-spectrum sunscreens and sun avoidance are important steps in preventing DNA damage induced from exposure to UV radiation. Other cosmeceutical agents have been designed to hinder the effects of UV radiation or to foster DNA repair. Besides sunscreen, the key members of the dermatologic armamentarium against DNA damage are various antioxidants. Data have been gathered over the last few decades that support the protective effects of antioxidants such as polypodium leucotomos,ascorbic acid, and green tea. Other antioxidants are associated with less data, but hypothetically should deliver similar benefits.
Polypodium leucotomos (PL), an oral extract derived from ferns, has been demonstrated to display photoprotective effects at an oral dose of 7.5 mg. PL has consistently exhibited antitumor and skin protective effects.8 A 2004 study in humans revealed that two oral doses of PL contributed to a significant reduction in DNA damage after UV exposure,9 and a 2017 study showed that PL protected skin DNA from UVB.10 Although PL has been linked to topical benefits, it is the oral form that is most often used to protect skin.
Ascorbic acid, also known as vitamin C, has been amply demonstrated to confer benefits when given both orally and topically. An acidic environment is necessary for optimal absorption. Topical application of ascorbic acid, along with vitamin E and ferulic acid, has been demonstrated to decrease the formation of thymine dimers.11 Unlike other antioxidants, ascorbic acid also stimulates procollagen genes in fibroblasts to increase collagen synthesis.12
EpiGalloCatechin-3-O-Gallate (also known as EGCG), the primary active constituent of green tea, has been demonstrated to induce IL-12 to increase the production of enzymes that repair UV-induced DNA damage.15 The proven photoprotective effects of topical and oral green tea include reducing UV-induced erythema, decreasing sunburn cell formation, and attenuating DNA damage.16
Preventing and treating mitochondrial DNA damage
UV radiation elicits mitochondrial DNA damage known as the “common deletion.”17 Damaged mitochondria produce harmful free radicals known as reactive oxygen species. Mitochondria damage caused by ROS decreases the mitochondria’s ability to generate ATP energy, which is necessary for DNA repair and other cellular processes.
Free radicals and UV radiation damage mitochondria, as does normal cellular metabolism. The range of damage includes mitochondrial DNA impairment, loss of mitochondrial enzymes, and decreased ATP production. This leads to less energy for DNA repair and other reparative processes. While there is no established way to reduce mitochondrial damage once it has occurred, several research initiatives to achieve this end are underway. Currently, protecting the mitochondria from harm with sunscreens and antioxidants is the best option.
Antioxidants are effective in preventing the damaging effects of free radicals on vulnerable mitochondria. As a component of the mitochondrial respiratory chain and an antioxidant itself, coenzyme Q10 is particularly useful in this role. CoQ10 is available in both oral and topical formulations. Oral forms should be taken only in the morning because of a caffeine-like effect. Topical forms of CoQ10 have a dark yellow color that may be unappealing to patients. Polypodium leucotomos has been shown to lower the number of common deletions found in the mitochondria of irradiated keratinocytes and fibroblasts.18 The oral form is recommended. Another potent antioxidant, curcumin, is being studied for mitochondrial protective properties.19 Its strong yellow color and smell render it better suited for oral use although many companies are trying to develop cosmetically elegant topical formulations.
Scavenging free radicals
Ultraviolet light, pollution, and other insults engender free radical formation. Even sunscreen use has been linked to increased production of free radicals. Free radicals, also known as reactive oxygen species, harm cells in many ways including mitochondrial damage, DNA mutations, glycation, lysosomal damage, and oxidation of important lipids and other cellular components such as proteins. Antioxidants present various beneficial effects including scavenging free radicals, decreasing activation of mitogen-activated protein kinases, chelation of copper required by tyrosinase, and suppression of inflammatory factors, such as nuclear factor (NF)-
In summary, skin aging has many causes. Although they are not all understood, some of the processes have been elucidated. Next month, this column will focus on the prevention and treatment of inflammation and glycation, as well as reversing the effects of aging on skin cells.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014). She also wrote a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems LLC.
References
1. Baumann, Leslie S. “Cosmeceuticals and cosmetic ingredients” (New York: McGraw-Hill Education / Medical, 2014).
2. Baumann, Leslie S. The Baumann Skin Typing System in “Textbook of Aging Skin” (New York: Springer-Verlag Berlin Heidelberg, 2017). pp. 1579-94.
3. Storm A et al. J Am Acad Dermatol. 2008 Dec;59(6):975-80.
4. Uitto J. N Engl J Med. 1997 Nov 13;337(20):1463-5.
5. Tornaletti S et al. Science. 1994;263(5152):1436-8.
6. Bickers D et al. J. Investig. Dermatol. 2006;126(12):2565-75.
7. Yaar M et al. Br J Dermatol. 2007 Nov;157(5):874-87.
8. Parrado C et al. Int J Mol Sci. 2016 Jun 29;17(7). pii: E1026.
9. Middelkamp-Hup MA et al. J Am Acad Dermatol. 2004 Dec;51(6):910-8.
10. Kohli I et al. J Am Acad Dermatol. 2017 Jul;77(1):33-41.
11. Murray JC et al. J Am Acad Dermatol. 2008;59(3):418-25.
12. Geesin JC et al. J Invest Dermatol. 1988 Apr;90(4):420-4.
13. Thompson BC et al. PLoS One. 2015 Feb 6;10(2):e0117491.
14. Surjana D et al. Carcinogenesis. 2013 May;34(5):1144-9.
15. Meeran SM et al. Cancer Res. 2006 May 15;66(10):5512-20.
16. Elmets CA et al. J Am Acad Dermatol. 2001 Mar;44(3):425-32.
17. Berneburg M et al. J Invest Dermatol. 2004 May;122(5):1277-83.
18. Villa A et al. J Am Acad Dermatol. 2010 Mar;62(3):511-3.
19. Trujillo J et al. Arch Pharm Chem Life Sci. 2014. doi: 10.1002/ardp.2014002662014.
20. Muthusam V et al. Arch Dermatol Res. 2010 Jan;302(1):5-17.
Cosmetic Corner: Dermatologists Weigh in on Wet Skin Moisturizers
To improve patient care and outcomes, leading dermatologists offered their recommendations on wet skin moisturizers. Consideration must be given to:
- Eucerin In-Shower Body Lotion
Beiersdorf
“This product is inexpensive, hypoallergenic, and fragrance free.”—Gary Goldenberg, MD, New York, New York
- Jergens Wet Skin Moisturizer With Refreshing Coconut Oil
Kao USA Inc
“I like how quickly the skin absorbs this product, and coconut oil is an excellent moisturizing ingredient. You simply apply to wet skin straight out of the shower or bath, and gently pat dry.”—Jeannette Graf, MD, Great Neck, New York
- Olay Ultra Moisture In-Shower Body Lotion
Procter & Gamble
“This is a time-saving and nongreasy moisturizing product for patients who are noncompliant with regular moisturizers.”—Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Bar soaps, lip plumpers, and pigment correctors will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on wet skin moisturizers. Consideration must be given to:
- Eucerin In-Shower Body Lotion
Beiersdorf
“This product is inexpensive, hypoallergenic, and fragrance free.”—Gary Goldenberg, MD, New York, New York
- Jergens Wet Skin Moisturizer With Refreshing Coconut Oil
Kao USA Inc
“I like how quickly the skin absorbs this product, and coconut oil is an excellent moisturizing ingredient. You simply apply to wet skin straight out of the shower or bath, and gently pat dry.”—Jeannette Graf, MD, Great Neck, New York
- Olay Ultra Moisture In-Shower Body Lotion
Procter & Gamble
“This is a time-saving and nongreasy moisturizing product for patients who are noncompliant with regular moisturizers.”—Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Bar soaps, lip plumpers, and pigment correctors will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on wet skin moisturizers. Consideration must be given to:
- Eucerin In-Shower Body Lotion
Beiersdorf
“This product is inexpensive, hypoallergenic, and fragrance free.”—Gary Goldenberg, MD, New York, New York
- Jergens Wet Skin Moisturizer With Refreshing Coconut Oil
Kao USA Inc
“I like how quickly the skin absorbs this product, and coconut oil is an excellent moisturizing ingredient. You simply apply to wet skin straight out of the shower or bath, and gently pat dry.”—Jeannette Graf, MD, Great Neck, New York
- Olay Ultra Moisture In-Shower Body Lotion
Procter & Gamble
“This is a time-saving and nongreasy moisturizing product for patients who are noncompliant with regular moisturizers.”—Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Bar soaps, lip plumpers, and pigment correctors will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.