User login
A novel method for assessing attractiveness and beauty
While Phi (or the Golden Ratio) and Leonardo da Vinci’s neoclassical canons have been used as traditional mathematical approaches to assess and calculate beauty, there may be more than meets the eye.
This model was created to denote “natural beauty,” both at baseline and after cosmetic procedures, which is what many physicians and patients ideally want to achieve after any aesthetic procedure.
In this model, when all three variables are at a maximum, a desirable attractive appearance is achieved that can be interpreted as “natural.” In his paper introducing this novel model, Dr. Dayan wrote that similar to the time-space dilemma, attractiveness “is relative, dynamic, and highly dependent on the position of the projector and the interpreter.” The 3-D cube of attractiveness “is therefore contained within a fourth dimension that takes into account the perspective of the judger.”
Similarly, in a pilot study,2 Dr. Dayan and colleagues also demonstrated that visually blind individuals can detect beauty. “This study further isolates the nature of beauty as a primal form of messaging that is subconsciously appreciated via embodied senses other than vision,” he and his coauthors wrote.
This observational study consisted of 8 blind and 10 nonblind test subjects and 6 models who were categorized into predetermined beauty categories. Test subjects were blindfolded and unblindfolded during their assessments. All groups rated those models, who were preselected as more beautiful, higher, except for the blindfolded, nonblind group – demonstrating a primal or neural pathway ability to perceive attractiveness in blind individuals. The study, “revealed that beauty is not only detected by visual sense but also through embodied senses other than sight,” the authors commented.
It should be noted that sometimes ethnic features and features that are unique outside of the neoclassical canons or golden ratio can also uniquely make people look more attractive. Ethnic variations in beauty standards exist and need to be further studied and celebrated. There is certainly high expertise and an art required to perceiving aesthetics and performing aesthetic procedures, further exemplified by the complex nature of the different models and mathematical approaches of assessing it. These newer models account for attractiveness that may also start on the inside or beyond purely visual perception.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References:
1. Dayan S, Romero DH. J Cosmet Dermatol. 2018 Oct;17(5):925-30.
2. Dayan SH et al. Dermatol Surg. 2020 Oct;46(10):1317-22.
While Phi (or the Golden Ratio) and Leonardo da Vinci’s neoclassical canons have been used as traditional mathematical approaches to assess and calculate beauty, there may be more than meets the eye.
This model was created to denote “natural beauty,” both at baseline and after cosmetic procedures, which is what many physicians and patients ideally want to achieve after any aesthetic procedure.
In this model, when all three variables are at a maximum, a desirable attractive appearance is achieved that can be interpreted as “natural.” In his paper introducing this novel model, Dr. Dayan wrote that similar to the time-space dilemma, attractiveness “is relative, dynamic, and highly dependent on the position of the projector and the interpreter.” The 3-D cube of attractiveness “is therefore contained within a fourth dimension that takes into account the perspective of the judger.”
Similarly, in a pilot study,2 Dr. Dayan and colleagues also demonstrated that visually blind individuals can detect beauty. “This study further isolates the nature of beauty as a primal form of messaging that is subconsciously appreciated via embodied senses other than vision,” he and his coauthors wrote.
This observational study consisted of 8 blind and 10 nonblind test subjects and 6 models who were categorized into predetermined beauty categories. Test subjects were blindfolded and unblindfolded during their assessments. All groups rated those models, who were preselected as more beautiful, higher, except for the blindfolded, nonblind group – demonstrating a primal or neural pathway ability to perceive attractiveness in blind individuals. The study, “revealed that beauty is not only detected by visual sense but also through embodied senses other than sight,” the authors commented.
It should be noted that sometimes ethnic features and features that are unique outside of the neoclassical canons or golden ratio can also uniquely make people look more attractive. Ethnic variations in beauty standards exist and need to be further studied and celebrated. There is certainly high expertise and an art required to perceiving aesthetics and performing aesthetic procedures, further exemplified by the complex nature of the different models and mathematical approaches of assessing it. These newer models account for attractiveness that may also start on the inside or beyond purely visual perception.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References:
1. Dayan S, Romero DH. J Cosmet Dermatol. 2018 Oct;17(5):925-30.
2. Dayan SH et al. Dermatol Surg. 2020 Oct;46(10):1317-22.
While Phi (or the Golden Ratio) and Leonardo da Vinci’s neoclassical canons have been used as traditional mathematical approaches to assess and calculate beauty, there may be more than meets the eye.
This model was created to denote “natural beauty,” both at baseline and after cosmetic procedures, which is what many physicians and patients ideally want to achieve after any aesthetic procedure.
In this model, when all three variables are at a maximum, a desirable attractive appearance is achieved that can be interpreted as “natural.” In his paper introducing this novel model, Dr. Dayan wrote that similar to the time-space dilemma, attractiveness “is relative, dynamic, and highly dependent on the position of the projector and the interpreter.” The 3-D cube of attractiveness “is therefore contained within a fourth dimension that takes into account the perspective of the judger.”
Similarly, in a pilot study,2 Dr. Dayan and colleagues also demonstrated that visually blind individuals can detect beauty. “This study further isolates the nature of beauty as a primal form of messaging that is subconsciously appreciated via embodied senses other than vision,” he and his coauthors wrote.
This observational study consisted of 8 blind and 10 nonblind test subjects and 6 models who were categorized into predetermined beauty categories. Test subjects were blindfolded and unblindfolded during their assessments. All groups rated those models, who were preselected as more beautiful, higher, except for the blindfolded, nonblind group – demonstrating a primal or neural pathway ability to perceive attractiveness in blind individuals. The study, “revealed that beauty is not only detected by visual sense but also through embodied senses other than sight,” the authors commented.
It should be noted that sometimes ethnic features and features that are unique outside of the neoclassical canons or golden ratio can also uniquely make people look more attractive. Ethnic variations in beauty standards exist and need to be further studied and celebrated. There is certainly high expertise and an art required to perceiving aesthetics and performing aesthetic procedures, further exemplified by the complex nature of the different models and mathematical approaches of assessing it. These newer models account for attractiveness that may also start on the inside or beyond purely visual perception.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References:
1. Dayan S, Romero DH. J Cosmet Dermatol. 2018 Oct;17(5):925-30.
2. Dayan SH et al. Dermatol Surg. 2020 Oct;46(10):1317-22.
The case for a new skin typing system
Even though the popular classification system added skin types V and VI in 1988 to the first iteration established in 1975, it was never intended for categorizing skin color, according to Karen C. Kagha, MD.
“This topic is going to become more relevant in our clinical practices, especially when you look at the current population trends in the U.S.,” Dr. Kagha, MD, a dermatologist and cosmetic and laser fellow in the department of dermatology at Massachusetts General Hospital and a research fellow at Wellman Center for Photomedicine, both in Boston, said during a virtual course on laser and aesthetic skin therapy. “Minority groups continue to increase. According to the U.S. Census Bureau, by the year 2050, we can expect that the majority of the population will be of non-European descent.”
The original intent of the Fitzpatrick Skin Typing (FST) system was to establish a minimal erythema dose, or likelihood to burn for patients receiving phototherapy, she continued. However, a recently published survey of 141 board-certified dermatologists and trainees found that 31% of respondents said that they used the Fitzpatrick Skin Typing System to describe the patient’s race or ethnicity, 47% used it to describe the patients’ constitutive skin color, and 22% used it in both scenarios.
“There also have been inconsistencies reported with the Fitzpatrick Skin Typing System,” Dr. Kagha said during the meeting, which was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. “Some studies show that there are inconsistent correlations between self-reported Fitzpatrick Skin Type and burn risk, and between self-reported FST and physician-reported FST. This means that some patients self-identify differently from how their physicians identify them.” There have also been some inconsistent correlations between race and objective measures of pigmentation, and also between race and self-reported FST.
Several classification systems have surfaced to try to bridge some of these gaps, including the Fanous classification, the Roberts Skin Type Classification System, and the Lancer Ethnicity Scale. Some of these have focused more on expanding the racial and ethnic categories to help predict response to procedures, she said, while others have focused more on hyperpigmentation, photoaging, and risk of scarring. “Others have suggested having different color matching systems to expand on a number of color-matched hues with regard to hyperpigmentation or race,” Dr. Kagha added. “In spite of all these efforts, it seems that the FST system remains the most widely used classification system in dermatology. I think that’s likely because we haven’t established a new consensus on a different system to use.”
She went on to postulate that there is likely “an infinite number of skin colors that are also impacted by geographic and cultural factors. Perhaps we should restructure how we think about skin typing. We need to establish a new consensus on skin typing, one that respects the variability in skin color but also one that’s clear, concise, objective, practical, and can be universally accepted.”
Dr. Kagha concluded her remarks by encouraging dermatologists to become more comfortable with treating all skin types. “This is going to be in line with the current population trends in the U.S., and also in line with the patients that we serve. Finally, I think we as physicians are in a unique position. Our patients’ frustrations and unsolved mysteries can drive our passion and our patient-centered innovation. For me, a common theme and a common source of frustration that I’ve seen in patients with increased melanin in their skin is figuring out how to effectively remove or prevent unwanted marks or unwanted pigment without disturbing the baseline pigment.”
Dr. Kagha reported having no financial disclosures.
Even though the popular classification system added skin types V and VI in 1988 to the first iteration established in 1975, it was never intended for categorizing skin color, according to Karen C. Kagha, MD.
“This topic is going to become more relevant in our clinical practices, especially when you look at the current population trends in the U.S.,” Dr. Kagha, MD, a dermatologist and cosmetic and laser fellow in the department of dermatology at Massachusetts General Hospital and a research fellow at Wellman Center for Photomedicine, both in Boston, said during a virtual course on laser and aesthetic skin therapy. “Minority groups continue to increase. According to the U.S. Census Bureau, by the year 2050, we can expect that the majority of the population will be of non-European descent.”
The original intent of the Fitzpatrick Skin Typing (FST) system was to establish a minimal erythema dose, or likelihood to burn for patients receiving phototherapy, she continued. However, a recently published survey of 141 board-certified dermatologists and trainees found that 31% of respondents said that they used the Fitzpatrick Skin Typing System to describe the patient’s race or ethnicity, 47% used it to describe the patients’ constitutive skin color, and 22% used it in both scenarios.
“There also have been inconsistencies reported with the Fitzpatrick Skin Typing System,” Dr. Kagha said during the meeting, which was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. “Some studies show that there are inconsistent correlations between self-reported Fitzpatrick Skin Type and burn risk, and between self-reported FST and physician-reported FST. This means that some patients self-identify differently from how their physicians identify them.” There have also been some inconsistent correlations between race and objective measures of pigmentation, and also between race and self-reported FST.
Several classification systems have surfaced to try to bridge some of these gaps, including the Fanous classification, the Roberts Skin Type Classification System, and the Lancer Ethnicity Scale. Some of these have focused more on expanding the racial and ethnic categories to help predict response to procedures, she said, while others have focused more on hyperpigmentation, photoaging, and risk of scarring. “Others have suggested having different color matching systems to expand on a number of color-matched hues with regard to hyperpigmentation or race,” Dr. Kagha added. “In spite of all these efforts, it seems that the FST system remains the most widely used classification system in dermatology. I think that’s likely because we haven’t established a new consensus on a different system to use.”
She went on to postulate that there is likely “an infinite number of skin colors that are also impacted by geographic and cultural factors. Perhaps we should restructure how we think about skin typing. We need to establish a new consensus on skin typing, one that respects the variability in skin color but also one that’s clear, concise, objective, practical, and can be universally accepted.”
Dr. Kagha concluded her remarks by encouraging dermatologists to become more comfortable with treating all skin types. “This is going to be in line with the current population trends in the U.S., and also in line with the patients that we serve. Finally, I think we as physicians are in a unique position. Our patients’ frustrations and unsolved mysteries can drive our passion and our patient-centered innovation. For me, a common theme and a common source of frustration that I’ve seen in patients with increased melanin in their skin is figuring out how to effectively remove or prevent unwanted marks or unwanted pigment without disturbing the baseline pigment.”
Dr. Kagha reported having no financial disclosures.
Even though the popular classification system added skin types V and VI in 1988 to the first iteration established in 1975, it was never intended for categorizing skin color, according to Karen C. Kagha, MD.
“This topic is going to become more relevant in our clinical practices, especially when you look at the current population trends in the U.S.,” Dr. Kagha, MD, a dermatologist and cosmetic and laser fellow in the department of dermatology at Massachusetts General Hospital and a research fellow at Wellman Center for Photomedicine, both in Boston, said during a virtual course on laser and aesthetic skin therapy. “Minority groups continue to increase. According to the U.S. Census Bureau, by the year 2050, we can expect that the majority of the population will be of non-European descent.”
The original intent of the Fitzpatrick Skin Typing (FST) system was to establish a minimal erythema dose, or likelihood to burn for patients receiving phototherapy, she continued. However, a recently published survey of 141 board-certified dermatologists and trainees found that 31% of respondents said that they used the Fitzpatrick Skin Typing System to describe the patient’s race or ethnicity, 47% used it to describe the patients’ constitutive skin color, and 22% used it in both scenarios.
“There also have been inconsistencies reported with the Fitzpatrick Skin Typing System,” Dr. Kagha said during the meeting, which was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. “Some studies show that there are inconsistent correlations between self-reported Fitzpatrick Skin Type and burn risk, and between self-reported FST and physician-reported FST. This means that some patients self-identify differently from how their physicians identify them.” There have also been some inconsistent correlations between race and objective measures of pigmentation, and also between race and self-reported FST.
Several classification systems have surfaced to try to bridge some of these gaps, including the Fanous classification, the Roberts Skin Type Classification System, and the Lancer Ethnicity Scale. Some of these have focused more on expanding the racial and ethnic categories to help predict response to procedures, she said, while others have focused more on hyperpigmentation, photoaging, and risk of scarring. “Others have suggested having different color matching systems to expand on a number of color-matched hues with regard to hyperpigmentation or race,” Dr. Kagha added. “In spite of all these efforts, it seems that the FST system remains the most widely used classification system in dermatology. I think that’s likely because we haven’t established a new consensus on a different system to use.”
She went on to postulate that there is likely “an infinite number of skin colors that are also impacted by geographic and cultural factors. Perhaps we should restructure how we think about skin typing. We need to establish a new consensus on skin typing, one that respects the variability in skin color but also one that’s clear, concise, objective, practical, and can be universally accepted.”
Dr. Kagha concluded her remarks by encouraging dermatologists to become more comfortable with treating all skin types. “This is going to be in line with the current population trends in the U.S., and also in line with the patients that we serve. Finally, I think we as physicians are in a unique position. Our patients’ frustrations and unsolved mysteries can drive our passion and our patient-centered innovation. For me, a common theme and a common source of frustration that I’ve seen in patients with increased melanin in their skin is figuring out how to effectively remove or prevent unwanted marks or unwanted pigment without disturbing the baseline pigment.”
Dr. Kagha reported having no financial disclosures.
FROM A LASER & AESTHETIC SKIN THERAPY COURSE
Orbital Granuloma Formation Following Autoinjection of Paraffin Oil: Management Considerations
To the Editor:
Injectable fillers are an increasingly common means of achieving minimally invasive facial rejuvenation. In the hands of well-trained practitioners, these compounds typically are well tolerated, effective, and have a strong safety profile1; however, there have been reports of complications, including vision loss,2 orbital infarction,3 persistent inflammatory nodules,4 and infection.4,5 Paraffin, a derivative of mineral oil, currently is used in cosmetic products and medical ointments.6 In the early 1900s, it often was injected into the body for various medical procedures, such as to create prosthetic testicles, to treat bladder incontinence, and eventually to correct facial contour defects.7,8 Due to adverse effects, injection of paraffin oil was discontinued in the Western medical community around the time of World War I.7 Unfortunately, some patients continue to self-inject paraffin oil for cosmetic purposes today. We present a case of foreign-body granuloma formation mimicking periorbital cellulitis following self-injection of paraffin oil. Our patient developed serious periorbital sequelae that required surgical intervention to restore normal anatomic function.
A 60-year-old woman who was otherwise healthy presented to the emergency department with facial swelling and a rash of 2 weeks’ duration. She reported that she had purchased what she believed was a cosmetic product at a local flea market 2 weeks prior to presentation. Her purchase included needles and a syringe with verbal instructions for injection into the face. She was told the product was used to treat wrinkles and referred to the injectable material as “oil” when providing her history. She reported that she had injected the material into the bilateral lower eyelids, left lateral lip, and left lateral chin. Three days later, she developed tingling and itching with swelling and redness at the injection sites. The patient was evaluated by the emergency department team and was prescribed a 10-day course of clindamycin empirically for suspected facial cellulitis.
The patient returned to the emergency department 12 days later upon completion of the antibiotic course with worsening edema and erythema. Examination revealed indurated, erythematous, and edematous warm plaques on the face that were concentrated around the prior injection sites with substantial periorbital erythema and edema (Figure 1). A consultation with oculoplastic surgery was obtained. Mechanical ptosis of the right eyelid was noted. Visual acuity was 20/30 in both eyes with habitual correction. Intraocular pressure was soft to palpation, and the pupils were round and reactive with no evidence of a relative afferent pupillary defect. Extraocular motility was intact bilaterally. Examination of the conjunctiva and sclera revealed bilateral conjunctival injection with chemosis of the right eye. The remainder of the anterior and posterior segment examination was within normal limits bilaterally.
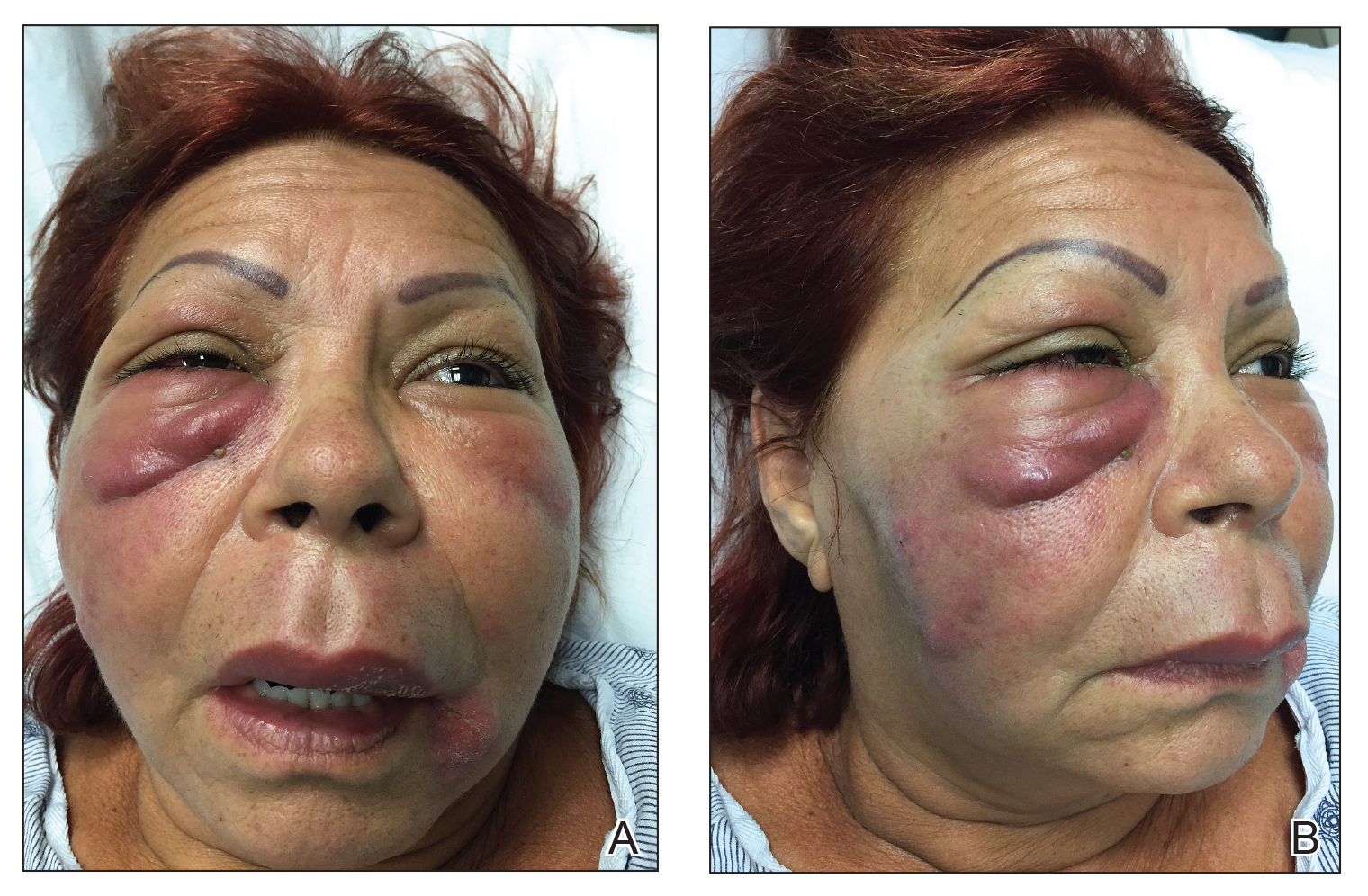
Computed tomography of the face showed extensive facial and periorbital swelling without abscess. A dermatology consultation was obtained. Two 4-mm punch biopsies were obtained from the left lower face and were sent for hematoxylin and eosin stain and tissue culture (bacterial, fungal, and acid-fast bacillus). Given the possibility of facial and periorbital cellulitis, empiric intravenous antibiotic therapy was initiated.
The tissue culture revealed normal skin flora. The biopsy results indicated a foreign-body reaction consistent with paraffin granuloma (Figures 2 and 3). Fite-Faraco, Grocott-Gomori methenamine-silver, and periodic acid–Schiff stains were all negative for infection. A diagnosis of foreign-body granuloma was established. Oral minocycline at a dosage of 100 mg twice daily was started, and the patient was discharged.


After 4 weeks of minocycline therapy, the patient showed no improvement and returned to the emergency department with worsening symptoms. She was readmitted and started on intravenous prednisone (1.5 mg/kg/d). Over the ensuing 5 days, the edema, erythema, conjunctival injection, and chemosis demonstrated notable improvement. She was subsequently discharged on an oral prednisone taper. Unfortunately, she did not respond to a trial of intralesional steroid injections to an area of granuloma formation on the left chin performed in the hospital before she was discharged.
In the ensuing months, she began to develop cicatricial ectropion of the right lower eyelid and mechanical ptosis of the right upper eyelid. Ten months after initial self-injection, staged surgical excision was initiated by an oculoplastic surgeon (I.V.) with the goal of debulking the periorbital region to correct the ectropion and mechanical ptosis. A transconjunctival approach was used to carefully excise the material while still maintaining the architecture of the lower eyelid. The ectropion was surgically corrected concurrently.
One month after excision, serial injections of 5-fluorouracil (5-FU) and triamcinolone acetonide 40 mg/mL were administered to the right lower eyelid and anterior orbit for 3 months. Fifteen weeks after the first surgery, a second surgery was performed to address residual medial right lower eyelid induration, right upper eyelid mechanical ptosis, and left orbital inflammation. During the postoperative period, serial monthly injections of 5-FU and triamcinolone acetonide were again performed beginning at the first postoperative month.
The surgical excisions resulted in notable improvement 3 months following excision (Figure 4). The patient noted improved ocular surface comfort with decreased foreign-body sensation and tearing. She also was pleased with the improved cosmetic outcome.
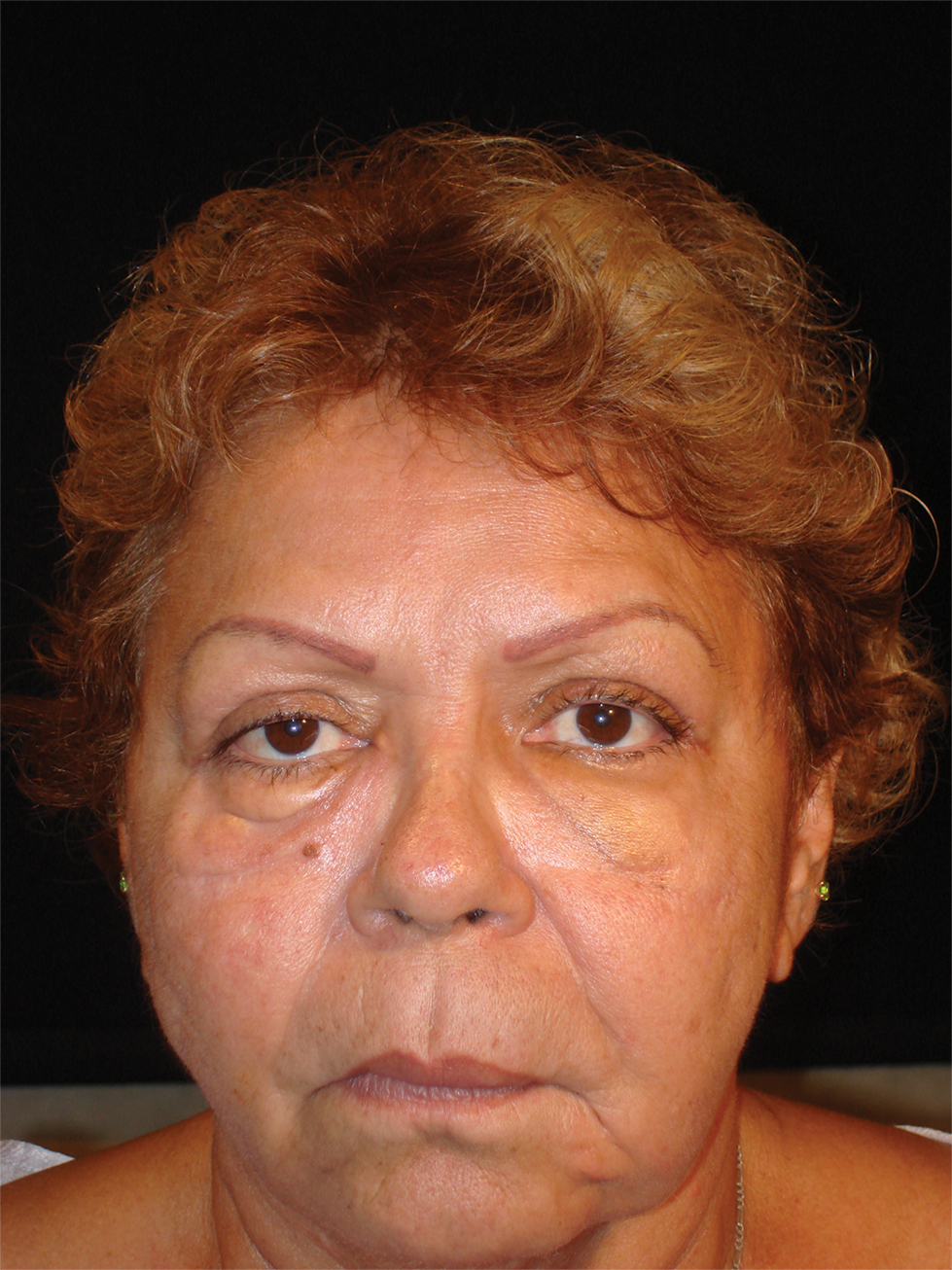
Crude substances such as paraffin, petroleum jelly, and lanolin were used for aesthetic purposes in the late 19th and early 20th centuries, initially with satisfying results; however, long-term adverse effects such as hardening of the skin, swelling, granuloma formation, ulceration, infections, and abscesses have discouraged its use by medical professionals today.5 Since paraffin is resistant to degradation and absorption, foreign-body reactions may occur upon injection. These reactions are characterized by replacement of normal subcutaneous tissue by cystic spaces of paraffin oil and/or calcification, similar to the appearance of Swiss cheese on histology and surrounded by various inflammatory cells and fibrous tissue.9,10
Clinically, there is an acute inflammatory phase followed by a latent phase of chronic granulomatous inflammation that can last for years.10 Our patient presented during the acute phase, with erythematous and edematous warm plaques around the eye mimicking an orbital infection.
The treatment of choice for paraffin granuloma is complete surgical excision to prevent recurrence.6,9 However, intralesional corticosteroids are preferred in the facial area, especially if complete removal is not possible.10 Intralesional corticosteroid injections inhibit fibroblast and macrophage activity as well as the deposition of collagen, leading to reduced pain and swelling in most cases.11 Additionally, combining antimitotic agents such as 5-FU with a corticosteroid might reduce the risk for cortisone skin atrophy.12 In our case, the patient did not respond to combined 5-FU with intralesional steroids and required oral corticosteroids while awaiting serial excisions.
Our case highlights several important points in the management of paraffin granuloma. First, the clinician must perform a thorough patient history, as surreptitious use of non–medical-grade fillers is more common than one might think.13 Second, the initial presentation of these patients can mimic an infectious process. Careful history, testing, and observation can aid in making the appropriate diagnosis. Finally, treatment of these patients is complex. The mainstays of therapy are systemic anti-inflammatory medications, time, and supportive care. In some cases, surgery may be required. When processes such as paraffin granulomas involve the periorbital region, particular care is required to avoid cicatricial lagophthalmos, ectropion, or retraction. Thoughtful surgical manipulation is required to avoid these complications, which indeed may occur even with the most appropriate interventions.
- Duker D, Erdmann R, Hartmann V, et al. The impact of adverse reactions to injectable filler substances on quality of life: results from the Berlin Injectable Filler Safety (IFS)—study. J Eur Acad Dermatol Venereol. 2016;30:1013-1020.
- Prado G, Rodriguez-Feliz J. Ocular pain and impending blindness during facial cosmetic injections: is your office prepared? [published online December 28, 2016]. Aesthetic Plast Surg. 2017;41:199-203.
- Roberts SA, Arthurs BP. Severe visual loss and orbital infarction following periorbital aesthetic poly-(L)-lactic acid (PLLA) injection. Ophthalmic Plast Reconstr Surg. 2012;28:E68-E70.
- Cassuto D, Pignatti M, Pacchioni L, et al. Management of complications caused by permanent fillers in the face: a treatment algorithm. Plast Reconstr Surg. 2016;138:215E-227E.
- Haneke E. Adverse effects of fillers and their histopathology. Facial Plast Surg. 2014;30:599-614.
- Friedrich RE, Zustin J. Paraffinoma of lips and oral mucosa: case report and brief review of literature. GMS Interdiscip Plast Reconstr Surg DGPW. 2014;3:Doc05.
- Matton G, Anseeuw A, De Keyser F. The history of injectable biomaterials and the biology of collagen. Aesthetic Plast Surg. 1985;9:133-140.
- Glicenstein J. Les premiers fillers, Vaseline et paraffine. du miracle a la catastrope. Ann Chir Plast Esthet. 2007;52:157-161.
- Cohen JL, Keoleian CM, Krull EA. Penile paraffinoma: self-injection with mineral oil. J Am Acad Dermatol 2002;47:S251-S253.
- Legaspi-Vicerra ME, Field LM. Paraffin granulomata, “witch’s chin,” and nasal deformities excision and reconstruction with reduction chinplasty and open rhinotomy resection. J Clin Aesthet Dermatol 2010;3:54-58.
- Carlos-Fabuel L, Marzal-Gamarra C, Marti-Alamo S, et al. Foreign body granulomatous reactions to cosmetic fillers. J Clin Exp Dent. 2012;4:E244-E247.
- Lemperle G, Gauthier-Hazan N. Foreign body granulomas after all injectable dermal fillers: part 2. treatment options. Plast Reconstr Surg. 2009;123:1864-1873.
- Seok J, Hong JY, Park KY, et al. Delayed immunologic complications due to injectable fillers by unlicensed practitioners: our experiences and a review of the literature. Dermatol Ther. 2016;29:41-44.
To the Editor:
Injectable fillers are an increasingly common means of achieving minimally invasive facial rejuvenation. In the hands of well-trained practitioners, these compounds typically are well tolerated, effective, and have a strong safety profile1; however, there have been reports of complications, including vision loss,2 orbital infarction,3 persistent inflammatory nodules,4 and infection.4,5 Paraffin, a derivative of mineral oil, currently is used in cosmetic products and medical ointments.6 In the early 1900s, it often was injected into the body for various medical procedures, such as to create prosthetic testicles, to treat bladder incontinence, and eventually to correct facial contour defects.7,8 Due to adverse effects, injection of paraffin oil was discontinued in the Western medical community around the time of World War I.7 Unfortunately, some patients continue to self-inject paraffin oil for cosmetic purposes today. We present a case of foreign-body granuloma formation mimicking periorbital cellulitis following self-injection of paraffin oil. Our patient developed serious periorbital sequelae that required surgical intervention to restore normal anatomic function.
A 60-year-old woman who was otherwise healthy presented to the emergency department with facial swelling and a rash of 2 weeks’ duration. She reported that she had purchased what she believed was a cosmetic product at a local flea market 2 weeks prior to presentation. Her purchase included needles and a syringe with verbal instructions for injection into the face. She was told the product was used to treat wrinkles and referred to the injectable material as “oil” when providing her history. She reported that she had injected the material into the bilateral lower eyelids, left lateral lip, and left lateral chin. Three days later, she developed tingling and itching with swelling and redness at the injection sites. The patient was evaluated by the emergency department team and was prescribed a 10-day course of clindamycin empirically for suspected facial cellulitis.
The patient returned to the emergency department 12 days later upon completion of the antibiotic course with worsening edema and erythema. Examination revealed indurated, erythematous, and edematous warm plaques on the face that were concentrated around the prior injection sites with substantial periorbital erythema and edema (Figure 1). A consultation with oculoplastic surgery was obtained. Mechanical ptosis of the right eyelid was noted. Visual acuity was 20/30 in both eyes with habitual correction. Intraocular pressure was soft to palpation, and the pupils were round and reactive with no evidence of a relative afferent pupillary defect. Extraocular motility was intact bilaterally. Examination of the conjunctiva and sclera revealed bilateral conjunctival injection with chemosis of the right eye. The remainder of the anterior and posterior segment examination was within normal limits bilaterally.

Computed tomography of the face showed extensive facial and periorbital swelling without abscess. A dermatology consultation was obtained. Two 4-mm punch biopsies were obtained from the left lower face and were sent for hematoxylin and eosin stain and tissue culture (bacterial, fungal, and acid-fast bacillus). Given the possibility of facial and periorbital cellulitis, empiric intravenous antibiotic therapy was initiated.
The tissue culture revealed normal skin flora. The biopsy results indicated a foreign-body reaction consistent with paraffin granuloma (Figures 2 and 3). Fite-Faraco, Grocott-Gomori methenamine-silver, and periodic acid–Schiff stains were all negative for infection. A diagnosis of foreign-body granuloma was established. Oral minocycline at a dosage of 100 mg twice daily was started, and the patient was discharged.


After 4 weeks of minocycline therapy, the patient showed no improvement and returned to the emergency department with worsening symptoms. She was readmitted and started on intravenous prednisone (1.5 mg/kg/d). Over the ensuing 5 days, the edema, erythema, conjunctival injection, and chemosis demonstrated notable improvement. She was subsequently discharged on an oral prednisone taper. Unfortunately, she did not respond to a trial of intralesional steroid injections to an area of granuloma formation on the left chin performed in the hospital before she was discharged.
In the ensuing months, she began to develop cicatricial ectropion of the right lower eyelid and mechanical ptosis of the right upper eyelid. Ten months after initial self-injection, staged surgical excision was initiated by an oculoplastic surgeon (I.V.) with the goal of debulking the periorbital region to correct the ectropion and mechanical ptosis. A transconjunctival approach was used to carefully excise the material while still maintaining the architecture of the lower eyelid. The ectropion was surgically corrected concurrently.
One month after excision, serial injections of 5-fluorouracil (5-FU) and triamcinolone acetonide 40 mg/mL were administered to the right lower eyelid and anterior orbit for 3 months. Fifteen weeks after the first surgery, a second surgery was performed to address residual medial right lower eyelid induration, right upper eyelid mechanical ptosis, and left orbital inflammation. During the postoperative period, serial monthly injections of 5-FU and triamcinolone acetonide were again performed beginning at the first postoperative month.
The surgical excisions resulted in notable improvement 3 months following excision (Figure 4). The patient noted improved ocular surface comfort with decreased foreign-body sensation and tearing. She also was pleased with the improved cosmetic outcome.

Crude substances such as paraffin, petroleum jelly, and lanolin were used for aesthetic purposes in the late 19th and early 20th centuries, initially with satisfying results; however, long-term adverse effects such as hardening of the skin, swelling, granuloma formation, ulceration, infections, and abscesses have discouraged its use by medical professionals today.5 Since paraffin is resistant to degradation and absorption, foreign-body reactions may occur upon injection. These reactions are characterized by replacement of normal subcutaneous tissue by cystic spaces of paraffin oil and/or calcification, similar to the appearance of Swiss cheese on histology and surrounded by various inflammatory cells and fibrous tissue.9,10
Clinically, there is an acute inflammatory phase followed by a latent phase of chronic granulomatous inflammation that can last for years.10 Our patient presented during the acute phase, with erythematous and edematous warm plaques around the eye mimicking an orbital infection.
The treatment of choice for paraffin granuloma is complete surgical excision to prevent recurrence.6,9 However, intralesional corticosteroids are preferred in the facial area, especially if complete removal is not possible.10 Intralesional corticosteroid injections inhibit fibroblast and macrophage activity as well as the deposition of collagen, leading to reduced pain and swelling in most cases.11 Additionally, combining antimitotic agents such as 5-FU with a corticosteroid might reduce the risk for cortisone skin atrophy.12 In our case, the patient did not respond to combined 5-FU with intralesional steroids and required oral corticosteroids while awaiting serial excisions.
Our case highlights several important points in the management of paraffin granuloma. First, the clinician must perform a thorough patient history, as surreptitious use of non–medical-grade fillers is more common than one might think.13 Second, the initial presentation of these patients can mimic an infectious process. Careful history, testing, and observation can aid in making the appropriate diagnosis. Finally, treatment of these patients is complex. The mainstays of therapy are systemic anti-inflammatory medications, time, and supportive care. In some cases, surgery may be required. When processes such as paraffin granulomas involve the periorbital region, particular care is required to avoid cicatricial lagophthalmos, ectropion, or retraction. Thoughtful surgical manipulation is required to avoid these complications, which indeed may occur even with the most appropriate interventions.
To the Editor:
Injectable fillers are an increasingly common means of achieving minimally invasive facial rejuvenation. In the hands of well-trained practitioners, these compounds typically are well tolerated, effective, and have a strong safety profile1; however, there have been reports of complications, including vision loss,2 orbital infarction,3 persistent inflammatory nodules,4 and infection.4,5 Paraffin, a derivative of mineral oil, currently is used in cosmetic products and medical ointments.6 In the early 1900s, it often was injected into the body for various medical procedures, such as to create prosthetic testicles, to treat bladder incontinence, and eventually to correct facial contour defects.7,8 Due to adverse effects, injection of paraffin oil was discontinued in the Western medical community around the time of World War I.7 Unfortunately, some patients continue to self-inject paraffin oil for cosmetic purposes today. We present a case of foreign-body granuloma formation mimicking periorbital cellulitis following self-injection of paraffin oil. Our patient developed serious periorbital sequelae that required surgical intervention to restore normal anatomic function.
A 60-year-old woman who was otherwise healthy presented to the emergency department with facial swelling and a rash of 2 weeks’ duration. She reported that she had purchased what she believed was a cosmetic product at a local flea market 2 weeks prior to presentation. Her purchase included needles and a syringe with verbal instructions for injection into the face. She was told the product was used to treat wrinkles and referred to the injectable material as “oil” when providing her history. She reported that she had injected the material into the bilateral lower eyelids, left lateral lip, and left lateral chin. Three days later, she developed tingling and itching with swelling and redness at the injection sites. The patient was evaluated by the emergency department team and was prescribed a 10-day course of clindamycin empirically for suspected facial cellulitis.
The patient returned to the emergency department 12 days later upon completion of the antibiotic course with worsening edema and erythema. Examination revealed indurated, erythematous, and edematous warm plaques on the face that were concentrated around the prior injection sites with substantial periorbital erythema and edema (Figure 1). A consultation with oculoplastic surgery was obtained. Mechanical ptosis of the right eyelid was noted. Visual acuity was 20/30 in both eyes with habitual correction. Intraocular pressure was soft to palpation, and the pupils were round and reactive with no evidence of a relative afferent pupillary defect. Extraocular motility was intact bilaterally. Examination of the conjunctiva and sclera revealed bilateral conjunctival injection with chemosis of the right eye. The remainder of the anterior and posterior segment examination was within normal limits bilaterally.

Computed tomography of the face showed extensive facial and periorbital swelling without abscess. A dermatology consultation was obtained. Two 4-mm punch biopsies were obtained from the left lower face and were sent for hematoxylin and eosin stain and tissue culture (bacterial, fungal, and acid-fast bacillus). Given the possibility of facial and periorbital cellulitis, empiric intravenous antibiotic therapy was initiated.
The tissue culture revealed normal skin flora. The biopsy results indicated a foreign-body reaction consistent with paraffin granuloma (Figures 2 and 3). Fite-Faraco, Grocott-Gomori methenamine-silver, and periodic acid–Schiff stains were all negative for infection. A diagnosis of foreign-body granuloma was established. Oral minocycline at a dosage of 100 mg twice daily was started, and the patient was discharged.


After 4 weeks of minocycline therapy, the patient showed no improvement and returned to the emergency department with worsening symptoms. She was readmitted and started on intravenous prednisone (1.5 mg/kg/d). Over the ensuing 5 days, the edema, erythema, conjunctival injection, and chemosis demonstrated notable improvement. She was subsequently discharged on an oral prednisone taper. Unfortunately, she did not respond to a trial of intralesional steroid injections to an area of granuloma formation on the left chin performed in the hospital before she was discharged.
In the ensuing months, she began to develop cicatricial ectropion of the right lower eyelid and mechanical ptosis of the right upper eyelid. Ten months after initial self-injection, staged surgical excision was initiated by an oculoplastic surgeon (I.V.) with the goal of debulking the periorbital region to correct the ectropion and mechanical ptosis. A transconjunctival approach was used to carefully excise the material while still maintaining the architecture of the lower eyelid. The ectropion was surgically corrected concurrently.
One month after excision, serial injections of 5-fluorouracil (5-FU) and triamcinolone acetonide 40 mg/mL were administered to the right lower eyelid and anterior orbit for 3 months. Fifteen weeks after the first surgery, a second surgery was performed to address residual medial right lower eyelid induration, right upper eyelid mechanical ptosis, and left orbital inflammation. During the postoperative period, serial monthly injections of 5-FU and triamcinolone acetonide were again performed beginning at the first postoperative month.
The surgical excisions resulted in notable improvement 3 months following excision (Figure 4). The patient noted improved ocular surface comfort with decreased foreign-body sensation and tearing. She also was pleased with the improved cosmetic outcome.

Crude substances such as paraffin, petroleum jelly, and lanolin were used for aesthetic purposes in the late 19th and early 20th centuries, initially with satisfying results; however, long-term adverse effects such as hardening of the skin, swelling, granuloma formation, ulceration, infections, and abscesses have discouraged its use by medical professionals today.5 Since paraffin is resistant to degradation and absorption, foreign-body reactions may occur upon injection. These reactions are characterized by replacement of normal subcutaneous tissue by cystic spaces of paraffin oil and/or calcification, similar to the appearance of Swiss cheese on histology and surrounded by various inflammatory cells and fibrous tissue.9,10
Clinically, there is an acute inflammatory phase followed by a latent phase of chronic granulomatous inflammation that can last for years.10 Our patient presented during the acute phase, with erythematous and edematous warm plaques around the eye mimicking an orbital infection.
The treatment of choice for paraffin granuloma is complete surgical excision to prevent recurrence.6,9 However, intralesional corticosteroids are preferred in the facial area, especially if complete removal is not possible.10 Intralesional corticosteroid injections inhibit fibroblast and macrophage activity as well as the deposition of collagen, leading to reduced pain and swelling in most cases.11 Additionally, combining antimitotic agents such as 5-FU with a corticosteroid might reduce the risk for cortisone skin atrophy.12 In our case, the patient did not respond to combined 5-FU with intralesional steroids and required oral corticosteroids while awaiting serial excisions.
Our case highlights several important points in the management of paraffin granuloma. First, the clinician must perform a thorough patient history, as surreptitious use of non–medical-grade fillers is more common than one might think.13 Second, the initial presentation of these patients can mimic an infectious process. Careful history, testing, and observation can aid in making the appropriate diagnosis. Finally, treatment of these patients is complex. The mainstays of therapy are systemic anti-inflammatory medications, time, and supportive care. In some cases, surgery may be required. When processes such as paraffin granulomas involve the periorbital region, particular care is required to avoid cicatricial lagophthalmos, ectropion, or retraction. Thoughtful surgical manipulation is required to avoid these complications, which indeed may occur even with the most appropriate interventions.
- Duker D, Erdmann R, Hartmann V, et al. The impact of adverse reactions to injectable filler substances on quality of life: results from the Berlin Injectable Filler Safety (IFS)—study. J Eur Acad Dermatol Venereol. 2016;30:1013-1020.
- Prado G, Rodriguez-Feliz J. Ocular pain and impending blindness during facial cosmetic injections: is your office prepared? [published online December 28, 2016]. Aesthetic Plast Surg. 2017;41:199-203.
- Roberts SA, Arthurs BP. Severe visual loss and orbital infarction following periorbital aesthetic poly-(L)-lactic acid (PLLA) injection. Ophthalmic Plast Reconstr Surg. 2012;28:E68-E70.
- Cassuto D, Pignatti M, Pacchioni L, et al. Management of complications caused by permanent fillers in the face: a treatment algorithm. Plast Reconstr Surg. 2016;138:215E-227E.
- Haneke E. Adverse effects of fillers and their histopathology. Facial Plast Surg. 2014;30:599-614.
- Friedrich RE, Zustin J. Paraffinoma of lips and oral mucosa: case report and brief review of literature. GMS Interdiscip Plast Reconstr Surg DGPW. 2014;3:Doc05.
- Matton G, Anseeuw A, De Keyser F. The history of injectable biomaterials and the biology of collagen. Aesthetic Plast Surg. 1985;9:133-140.
- Glicenstein J. Les premiers fillers, Vaseline et paraffine. du miracle a la catastrope. Ann Chir Plast Esthet. 2007;52:157-161.
- Cohen JL, Keoleian CM, Krull EA. Penile paraffinoma: self-injection with mineral oil. J Am Acad Dermatol 2002;47:S251-S253.
- Legaspi-Vicerra ME, Field LM. Paraffin granulomata, “witch’s chin,” and nasal deformities excision and reconstruction with reduction chinplasty and open rhinotomy resection. J Clin Aesthet Dermatol 2010;3:54-58.
- Carlos-Fabuel L, Marzal-Gamarra C, Marti-Alamo S, et al. Foreign body granulomatous reactions to cosmetic fillers. J Clin Exp Dent. 2012;4:E244-E247.
- Lemperle G, Gauthier-Hazan N. Foreign body granulomas after all injectable dermal fillers: part 2. treatment options. Plast Reconstr Surg. 2009;123:1864-1873.
- Seok J, Hong JY, Park KY, et al. Delayed immunologic complications due to injectable fillers by unlicensed practitioners: our experiences and a review of the literature. Dermatol Ther. 2016;29:41-44.
- Duker D, Erdmann R, Hartmann V, et al. The impact of adverse reactions to injectable filler substances on quality of life: results from the Berlin Injectable Filler Safety (IFS)—study. J Eur Acad Dermatol Venereol. 2016;30:1013-1020.
- Prado G, Rodriguez-Feliz J. Ocular pain and impending blindness during facial cosmetic injections: is your office prepared? [published online December 28, 2016]. Aesthetic Plast Surg. 2017;41:199-203.
- Roberts SA, Arthurs BP. Severe visual loss and orbital infarction following periorbital aesthetic poly-(L)-lactic acid (PLLA) injection. Ophthalmic Plast Reconstr Surg. 2012;28:E68-E70.
- Cassuto D, Pignatti M, Pacchioni L, et al. Management of complications caused by permanent fillers in the face: a treatment algorithm. Plast Reconstr Surg. 2016;138:215E-227E.
- Haneke E. Adverse effects of fillers and their histopathology. Facial Plast Surg. 2014;30:599-614.
- Friedrich RE, Zustin J. Paraffinoma of lips and oral mucosa: case report and brief review of literature. GMS Interdiscip Plast Reconstr Surg DGPW. 2014;3:Doc05.
- Matton G, Anseeuw A, De Keyser F. The history of injectable biomaterials and the biology of collagen. Aesthetic Plast Surg. 1985;9:133-140.
- Glicenstein J. Les premiers fillers, Vaseline et paraffine. du miracle a la catastrope. Ann Chir Plast Esthet. 2007;52:157-161.
- Cohen JL, Keoleian CM, Krull EA. Penile paraffinoma: self-injection with mineral oil. J Am Acad Dermatol 2002;47:S251-S253.
- Legaspi-Vicerra ME, Field LM. Paraffin granulomata, “witch’s chin,” and nasal deformities excision and reconstruction with reduction chinplasty and open rhinotomy resection. J Clin Aesthet Dermatol 2010;3:54-58.
- Carlos-Fabuel L, Marzal-Gamarra C, Marti-Alamo S, et al. Foreign body granulomatous reactions to cosmetic fillers. J Clin Exp Dent. 2012;4:E244-E247.
- Lemperle G, Gauthier-Hazan N. Foreign body granulomas after all injectable dermal fillers: part 2. treatment options. Plast Reconstr Surg. 2009;123:1864-1873.
- Seok J, Hong JY, Park KY, et al. Delayed immunologic complications due to injectable fillers by unlicensed practitioners: our experiences and a review of the literature. Dermatol Ther. 2016;29:41-44.
Practice Points
- The initial presentation of a foreign-body granulomatous process in a patient with surreptitious use of nonmedical filler can mimic infection; thus, careful history and diagnostic measures are paramount.
- Treatment of paraffin oil granuloma can be multifactorial and involves supportive care, systemic anti-inflammatory medications, time, and surgery.
- When a paraffin granuloma involves the orbital region, particular care is required to avoid long-term complications including cicatricial lagophthalmos, ectropion, or retractions, which can be mitigated with the help of oculoplastic surgery.
Subscription services a consideration for aesthetic patients
According to W. Grant Stevens, MD, an estimated 73% of aesthetic patients fall short when it comes to compliance with recommended treatment intervals for toxins, fillers, and other procedures.
“When we talk about how often the average patient should be treated with Botox, for instance, we say every 3-4 months,” Dr. Stevens, founder and CEO of Marina Plastic Surgery in Marina Del Rey, Calif., said during the virtual annual Masters of Aesthetics Symposium. But in reality, he added, “it’s more like every 7 months.” A 2015 survey of 23 Bay Area aesthetic practices conducted by HintMD found that 73% of patients were noncompliant and that they came in fewer than 3-4 times per year for treatments. “Not only did they come in infrequently, but they oftentimes were undercorrected and the revenue was being left on the table because of discounting and undercorrection,” said Dr. Stevens, who is also a professor of surgery in the division of plastic surgery at the University of Southern California, Los Angeles.
On average, each patient from the 23 practices surveyed spent $601.88 on treatments 1.44 times per year, yet the industry standard for neuromodulators is 3-4 times per year and every 2 months for HydraFacials and med spa facials. “What’s the problem?” he asked “Why are we falling off? For our practices, noncompliance leads to unhappy, undertreated patients, so they may write negative reviews. In addition to that, we lose revenue.” He cited results from a 2016 focus group of aesthetic patients who were asked about the perceived barriers to treatment compliance. More than two-thirds (68%) said cost was the issue, followed by the number of treatments required (43%) and effectiveness (16%).
Three years ago, Dr. Stevens used the HintMD platform to implement a treatment plan subscription service to 472 active members of his practice. Prior to implementation, patients were coming in for treatment with toxins an average of 1.8 times per year. After implementation, that rose to an average of 3.1 times per year. “That was almost an $800 incremental average increase spent on toxins alone,” Dr. Stevens said. “More importantly, the patients were therapeutic all year long.” With toxin and filler services combined, the average increased income grew to more than $1,100 per patient, which translated into increased annual revenue of $519,200.
Dr. Stevens said that many of his patients favor subscription services because most use them in other aspects of their lives, such as with Amazon Prime, Blue Apron, and Netflix. “They like it because it is personalized and customized,” he said. “If we want to adjust the amount of toxin or filler, we can do it that very day, and it’s customized for them. It’s not a one-size-fits-all program. It also allows them to have convenient, smaller monthly payments. That’s the key. That way, they budget. So, if they’re spending $200 a month or $500 a month or $1,000 a month, it’s a convenient monthly payment.”
Dr. Stevens disclosed that he is an adviser to Viveve, Venus, Aesthetics Biomedical, Alastin, Cypris Medical, Allergan, CoolSculpting, HydraFacial, Revance, Ampersand, and HintMD.
According to W. Grant Stevens, MD, an estimated 73% of aesthetic patients fall short when it comes to compliance with recommended treatment intervals for toxins, fillers, and other procedures.
“When we talk about how often the average patient should be treated with Botox, for instance, we say every 3-4 months,” Dr. Stevens, founder and CEO of Marina Plastic Surgery in Marina Del Rey, Calif., said during the virtual annual Masters of Aesthetics Symposium. But in reality, he added, “it’s more like every 7 months.” A 2015 survey of 23 Bay Area aesthetic practices conducted by HintMD found that 73% of patients were noncompliant and that they came in fewer than 3-4 times per year for treatments. “Not only did they come in infrequently, but they oftentimes were undercorrected and the revenue was being left on the table because of discounting and undercorrection,” said Dr. Stevens, who is also a professor of surgery in the division of plastic surgery at the University of Southern California, Los Angeles.
On average, each patient from the 23 practices surveyed spent $601.88 on treatments 1.44 times per year, yet the industry standard for neuromodulators is 3-4 times per year and every 2 months for HydraFacials and med spa facials. “What’s the problem?” he asked “Why are we falling off? For our practices, noncompliance leads to unhappy, undertreated patients, so they may write negative reviews. In addition to that, we lose revenue.” He cited results from a 2016 focus group of aesthetic patients who were asked about the perceived barriers to treatment compliance. More than two-thirds (68%) said cost was the issue, followed by the number of treatments required (43%) and effectiveness (16%).
Three years ago, Dr. Stevens used the HintMD platform to implement a treatment plan subscription service to 472 active members of his practice. Prior to implementation, patients were coming in for treatment with toxins an average of 1.8 times per year. After implementation, that rose to an average of 3.1 times per year. “That was almost an $800 incremental average increase spent on toxins alone,” Dr. Stevens said. “More importantly, the patients were therapeutic all year long.” With toxin and filler services combined, the average increased income grew to more than $1,100 per patient, which translated into increased annual revenue of $519,200.
Dr. Stevens said that many of his patients favor subscription services because most use them in other aspects of their lives, such as with Amazon Prime, Blue Apron, and Netflix. “They like it because it is personalized and customized,” he said. “If we want to adjust the amount of toxin or filler, we can do it that very day, and it’s customized for them. It’s not a one-size-fits-all program. It also allows them to have convenient, smaller monthly payments. That’s the key. That way, they budget. So, if they’re spending $200 a month or $500 a month or $1,000 a month, it’s a convenient monthly payment.”
Dr. Stevens disclosed that he is an adviser to Viveve, Venus, Aesthetics Biomedical, Alastin, Cypris Medical, Allergan, CoolSculpting, HydraFacial, Revance, Ampersand, and HintMD.
According to W. Grant Stevens, MD, an estimated 73% of aesthetic patients fall short when it comes to compliance with recommended treatment intervals for toxins, fillers, and other procedures.
“When we talk about how often the average patient should be treated with Botox, for instance, we say every 3-4 months,” Dr. Stevens, founder and CEO of Marina Plastic Surgery in Marina Del Rey, Calif., said during the virtual annual Masters of Aesthetics Symposium. But in reality, he added, “it’s more like every 7 months.” A 2015 survey of 23 Bay Area aesthetic practices conducted by HintMD found that 73% of patients were noncompliant and that they came in fewer than 3-4 times per year for treatments. “Not only did they come in infrequently, but they oftentimes were undercorrected and the revenue was being left on the table because of discounting and undercorrection,” said Dr. Stevens, who is also a professor of surgery in the division of plastic surgery at the University of Southern California, Los Angeles.
On average, each patient from the 23 practices surveyed spent $601.88 on treatments 1.44 times per year, yet the industry standard for neuromodulators is 3-4 times per year and every 2 months for HydraFacials and med spa facials. “What’s the problem?” he asked “Why are we falling off? For our practices, noncompliance leads to unhappy, undertreated patients, so they may write negative reviews. In addition to that, we lose revenue.” He cited results from a 2016 focus group of aesthetic patients who were asked about the perceived barriers to treatment compliance. More than two-thirds (68%) said cost was the issue, followed by the number of treatments required (43%) and effectiveness (16%).
Three years ago, Dr. Stevens used the HintMD platform to implement a treatment plan subscription service to 472 active members of his practice. Prior to implementation, patients were coming in for treatment with toxins an average of 1.8 times per year. After implementation, that rose to an average of 3.1 times per year. “That was almost an $800 incremental average increase spent on toxins alone,” Dr. Stevens said. “More importantly, the patients were therapeutic all year long.” With toxin and filler services combined, the average increased income grew to more than $1,100 per patient, which translated into increased annual revenue of $519,200.
Dr. Stevens said that many of his patients favor subscription services because most use them in other aspects of their lives, such as with Amazon Prime, Blue Apron, and Netflix. “They like it because it is personalized and customized,” he said. “If we want to adjust the amount of toxin or filler, we can do it that very day, and it’s customized for them. It’s not a one-size-fits-all program. It also allows them to have convenient, smaller monthly payments. That’s the key. That way, they budget. So, if they’re spending $200 a month or $500 a month or $1,000 a month, it’s a convenient monthly payment.”
Dr. Stevens disclosed that he is an adviser to Viveve, Venus, Aesthetics Biomedical, Alastin, Cypris Medical, Allergan, CoolSculpting, HydraFacial, Revance, Ampersand, and HintMD.
FROM MOA 2020
Dermatologists’ role in the development of the skin care industry
This is the third in a series of columns discussing the important roles that dermatologists have played in the skin care industry. with the cosmetic industry, rather than developing their own skin care lines.
Norman Orentreich, MD
Dr. Orentreich was a successful New York City dermatologist and the first to perform hair transplants. This new technique brought him fame and notoriety and arguably made him the first “celebrity dermatologist.” (He was also a member of the original advisory board of Dermatology News, at that time Skin & Allergy News, in January 1970.) Dr. Orentreich was a seminal figure in the trend to link the cosmetic industry and dermatology. In August 1967, Vogue magazine1 published an article on him, titled “Can Great Skin be Created?” This popular article caught the attention of Leonard Lauder, of Estée Lauder, who recruited Dr. Orentreich to help create the skin care line Clinique. Clinique was intended to be a brand with a medical look that promoted its products as “allergy tested,” with packaging that has an antiseptic look and beauty counter salespeople wearing white coats.
Dr. Orentreich’s input into the development of a skin type–based skin care line was fundamental to the development of this brand. The four-question questionnaire with an iconic plastic lever that customers slide left or right instantly provided them with an assessment of their skin type at the beauty counter, with one of four skin types: Very Dry to Dry Skin (Skin Type 1), Dry Combination (Skin Type 2), Combination Oily (Skin Type 3), and Oily (Skin Type 4).
Although this skin-typing system was not scientifically accurate (there is no scientific definition of combination skin), it was reminiscent of the system developed by cosmetic company tycoon Helena Rubinstein in the 1940s that classified people into four skin types: oily, dry, combination, and sensitive. Clinique became a blockbuster skin care brand and was one of the first developed by a dermatologist – although Dr. Orentreich did not put his name on it.
In 1972, Dr. Orentreich filed a patent2 for an exfoliating pad for the skin that later became known as the “Buf-Puf.” I heard years ago that he got the idea from the machines used to buff the floors in the hospital. The buffing pad had a hole in the center where the machine attached. Dr. Orentreich purportedly thought “I wonder what they do with the cut-out centers?” He looked into this, and subsequently used the centers to create the Buf-Puf. I cannot find a reference for this, but I love this story and hope it’s true. If any readers have any knowledge of this, please let me know, so I can amend my story if it is incorrect.
Almay
Almay, an amalgamation of the founders’ names, Alfred and Fanny May Woititz, was the first hypoallergenic brand, established in 1931, and the first to provide hypoallergenic cosmetics, long before Clinique. In addition, the company was the first skin care brand to become available by prescription only (as it was initially), fully disclose all individual ingredients in its products (well before this became mandatory in 1976), provide totally fragrance-free products, develop a hypoallergenic fragrance – and provide patch tests and other materials to physicians to identify contact allergens.
Over 90 years, the company was also the first among skin care brands to do the following:
- Provide custom formulations to individuals proven to be allergic to a specific ingredient, through their physicians.
- Perform a full range of premarket safety testing on all products for allergy and irritation, and test all its products for comedogenicity.
- Formulate cosmetics for use around the eye area (eye shadows and eyeliners) specifically for contact lens wearers.
- Formulate hypoallergenic regimens for specific skin types in the mass market.
- Provide a specific cosmetic regimen for acne-prone women, including a silicone-based makeup and active ingredients for treatment in cosmetics and skin care.
I recently interviewed Stanley Levy, MD, who was one of the consultants to Almay, and practices in Chapel Hill, N.C., where he has an academic niche related to skin care formulation and safety. He told me how Almay provided patch test materials to dermatologists to help identify contact dermatitis to cosmetic ingredients, and described Almay’s relationship with the dermatology field as follows: “From the outset, Almay was linked to dermatology. In 1930, a chemist and pharmacist in New York City, Al Woititz, was looking to compound cosmetics for his wife suffering from cosmetic allergies, Fannie May. He enlisted the counsel of the preeminent dermatologic expert in contact dermatitis at the time, Dr. Marion Sulzberger, to suggest ingredients to avoid. [Dr. Sulzberger was also a member of the original Dermatology News editorial advisory board.] Soon, dermatologists around New York City were recommending these formulations. This led to a product line free of the known allergens and a fledgling company trademarked as Almay. For the past 90 years, [the company] has kept a close relationship with dermatologists, well before that was the norm.”
The Almay research overseen by Dr. Levy and others contributed greatly to our understanding of the allergenicity of skin care.
Albert Kligman, MD
The turning point for the interface of dermatology with the cosmetic industry was the shift from a safety-based approach (hypoallergenic and noncomedogenic) to an emphasis on efficacy claims in the 1980s. Part of the impetus for this was the Dr. Kligman’s observation that retinoids could improve photoaging.
Dr. Kligman, a well-known dermatologist at the University of Pennsylvania, Philadelphia, showed that retinoids were an effective treatment for acne. For more about this, listen to my interview on the Dermatology Weekly podcast, with James Leyden, MD, about his work at the University of Pennsylvania with Dr. Kligman on the development of oral and topical retinoids. During Dr. Kligman’s research on acne, he noticed that wrinkles improved after treatment with tretinoin, and in 1986, he and Dr. Leyden (and several other authors) published the first article about tretinoin’s use for photoaged skin.3 This led to a double-blind study4 conducted by John J. Voorhees, MD, University of Michigan, Ann Arbor, and coauthors that showed statistically significant improvement of photoaged skin when treated with topical tretinoin. Dr. Voorhees and his group did many more studies on retinoids5,6 and photoaging7 – so many that, at one time, he was (and maybe still is) the most widely published dermatologist in the United States. These studies showed that, not only did prescription tretinoin improve the appearance of wrinkles, but so did over-the-counter retinol.8 Retinoids remain the most efficacious prescription and cosmeceutical ingredients to treat wrinkled skin.
When studies conducted by Dr. Kligman, Dr. Voorhees, and by Barbara Gilcrest, MD, 9,10 showed that retinoids improved wrinkles, a major change in the focus in the skin care industry occurred.
During the same time period, the studies on alpha hydroxy acids by Chérie Ditre, MD, Eugene Van Scott, MD, and colleages11,12; and studies by Sheldon Pinnell, MD, on Vitamin C (see part 1 of this series) all demonstrated the efficacy of cosmetic ingredients on photoaged skin. This triggered a major change in how skin care products were marketed, with an efficacy approach rather than a safety approach.
With the shift from safety (hypoallergenic and noncomedogenic issues) to efficacy claims in the 1980s, and as nondrug active ingredients like retinol were shown to have biologic effects, the lines between the Food and Drug Administration’s definition of a drug versus a cosmetic became blurred. In 1984, Dr. Kligman suggested a new classification for the ingredients that fell in the middle, proposing the term “cosmeceutical” and thus, the concept of a cosmeceutical was introduced. To this day, cosmeceutical is not an official definition and the FDA has yet to deal with it as a quasi-drug category. FDA regulations as to what constitutes a drug versus a cosmetic date back to the 1938 Food, Drug and Cosmetic Act.
Once marketing focused on efficacy, many companies made outrageous claims. During the second half of the 1980s, the FDA issued some warning letters to some companies in an effort to control these claims.
Now efficacy claims abound and we, as dermatologists, should be the experts who back up these claims with scientific data. As the cosmeceutical market has evolved and grown, consumers are bewildered by the myriad of active ingredients being promoted and the number of products in the marketplace. As dermatologic innovation has led to more efficacious active ingredients, our patients look to us as knowledgeable and credible sources of information and for recommendations about the best skin care routines for their skin issues. This is all reflected in the fact that physician-dispensed skin care is becoming the fastest growing segment in this market. It is incumbent upon dermatologists to be knowledgeable and conversant about skin care products and skin care routines, and is particularly true for those of us who sell skin care products in our offices.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Burt’s Bees, Evolus, Galderma, and Revance. She is the CEO of Skin Type Solutions, a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Vogue Magazine, 1967 Aug 15. “Can Great Skin be Created?”
2. https://patents.google.com/patent/US3910284.
3. Kligman AM et al. J Am Acad Dermatol. 1986 Oct;15(4 Pt 2):836-59.
4. Weiss JS et al. JAMA. 1988 Jan 22-29;259(4):527-32.
5. Goldfarb MT et al. J Am Acad Dermatol. 1989 Sep;21(3 Pt 2):645-50.
6. Ellis CN et al. J Am Acad Dermatol. 1990 Oct;23(4 Pt 1):629-37.
7. Kang S; Voorhees JJ. J Am Acad Dermatol. 1998 Aug;39(2 Pt 3):S55-61.
8. Kafi R et al. Arch Dermatol. 2007 May;143(5):606-12.
9. Gilchrest BA. J Am Acad Dermatol. 1989 Sep;21(3 Pt 2):610-3.
10. Bhawan J et al. Arch Dermatol. 1991 May;127(5):666-72.
11. Griffin TD et al. J Am Acad Dermatol. 1996 Feb;34(2 Pt 1):196-203.
12. Ditre CM et al. J Am Acad Dermatol. 1996 Feb;34(2 Pt 1):187-95.
This is the third in a series of columns discussing the important roles that dermatologists have played in the skin care industry. with the cosmetic industry, rather than developing their own skin care lines.
Norman Orentreich, MD
Dr. Orentreich was a successful New York City dermatologist and the first to perform hair transplants. This new technique brought him fame and notoriety and arguably made him the first “celebrity dermatologist.” (He was also a member of the original advisory board of Dermatology News, at that time Skin & Allergy News, in January 1970.) Dr. Orentreich was a seminal figure in the trend to link the cosmetic industry and dermatology. In August 1967, Vogue magazine1 published an article on him, titled “Can Great Skin be Created?” This popular article caught the attention of Leonard Lauder, of Estée Lauder, who recruited Dr. Orentreich to help create the skin care line Clinique. Clinique was intended to be a brand with a medical look that promoted its products as “allergy tested,” with packaging that has an antiseptic look and beauty counter salespeople wearing white coats.
Dr. Orentreich’s input into the development of a skin type–based skin care line was fundamental to the development of this brand. The four-question questionnaire with an iconic plastic lever that customers slide left or right instantly provided them with an assessment of their skin type at the beauty counter, with one of four skin types: Very Dry to Dry Skin (Skin Type 1), Dry Combination (Skin Type 2), Combination Oily (Skin Type 3), and Oily (Skin Type 4).
Although this skin-typing system was not scientifically accurate (there is no scientific definition of combination skin), it was reminiscent of the system developed by cosmetic company tycoon Helena Rubinstein in the 1940s that classified people into four skin types: oily, dry, combination, and sensitive. Clinique became a blockbuster skin care brand and was one of the first developed by a dermatologist – although Dr. Orentreich did not put his name on it.
In 1972, Dr. Orentreich filed a patent2 for an exfoliating pad for the skin that later became known as the “Buf-Puf.” I heard years ago that he got the idea from the machines used to buff the floors in the hospital. The buffing pad had a hole in the center where the machine attached. Dr. Orentreich purportedly thought “I wonder what they do with the cut-out centers?” He looked into this, and subsequently used the centers to create the Buf-Puf. I cannot find a reference for this, but I love this story and hope it’s true. If any readers have any knowledge of this, please let me know, so I can amend my story if it is incorrect.
Almay
Almay, an amalgamation of the founders’ names, Alfred and Fanny May Woititz, was the first hypoallergenic brand, established in 1931, and the first to provide hypoallergenic cosmetics, long before Clinique. In addition, the company was the first skin care brand to become available by prescription only (as it was initially), fully disclose all individual ingredients in its products (well before this became mandatory in 1976), provide totally fragrance-free products, develop a hypoallergenic fragrance – and provide patch tests and other materials to physicians to identify contact allergens.
Over 90 years, the company was also the first among skin care brands to do the following:
- Provide custom formulations to individuals proven to be allergic to a specific ingredient, through their physicians.
- Perform a full range of premarket safety testing on all products for allergy and irritation, and test all its products for comedogenicity.
- Formulate cosmetics for use around the eye area (eye shadows and eyeliners) specifically for contact lens wearers.
- Formulate hypoallergenic regimens for specific skin types in the mass market.
- Provide a specific cosmetic regimen for acne-prone women, including a silicone-based makeup and active ingredients for treatment in cosmetics and skin care.
I recently interviewed Stanley Levy, MD, who was one of the consultants to Almay, and practices in Chapel Hill, N.C., where he has an academic niche related to skin care formulation and safety. He told me how Almay provided patch test materials to dermatologists to help identify contact dermatitis to cosmetic ingredients, and described Almay’s relationship with the dermatology field as follows: “From the outset, Almay was linked to dermatology. In 1930, a chemist and pharmacist in New York City, Al Woititz, was looking to compound cosmetics for his wife suffering from cosmetic allergies, Fannie May. He enlisted the counsel of the preeminent dermatologic expert in contact dermatitis at the time, Dr. Marion Sulzberger, to suggest ingredients to avoid. [Dr. Sulzberger was also a member of the original Dermatology News editorial advisory board.] Soon, dermatologists around New York City were recommending these formulations. This led to a product line free of the known allergens and a fledgling company trademarked as Almay. For the past 90 years, [the company] has kept a close relationship with dermatologists, well before that was the norm.”
The Almay research overseen by Dr. Levy and others contributed greatly to our understanding of the allergenicity of skin care.
Albert Kligman, MD
The turning point for the interface of dermatology with the cosmetic industry was the shift from a safety-based approach (hypoallergenic and noncomedogenic) to an emphasis on efficacy claims in the 1980s. Part of the impetus for this was the Dr. Kligman’s observation that retinoids could improve photoaging.
Dr. Kligman, a well-known dermatologist at the University of Pennsylvania, Philadelphia, showed that retinoids were an effective treatment for acne. For more about this, listen to my interview on the Dermatology Weekly podcast, with James Leyden, MD, about his work at the University of Pennsylvania with Dr. Kligman on the development of oral and topical retinoids. During Dr. Kligman’s research on acne, he noticed that wrinkles improved after treatment with tretinoin, and in 1986, he and Dr. Leyden (and several other authors) published the first article about tretinoin’s use for photoaged skin.3 This led to a double-blind study4 conducted by John J. Voorhees, MD, University of Michigan, Ann Arbor, and coauthors that showed statistically significant improvement of photoaged skin when treated with topical tretinoin. Dr. Voorhees and his group did many more studies on retinoids5,6 and photoaging7 – so many that, at one time, he was (and maybe still is) the most widely published dermatologist in the United States. These studies showed that, not only did prescription tretinoin improve the appearance of wrinkles, but so did over-the-counter retinol.8 Retinoids remain the most efficacious prescription and cosmeceutical ingredients to treat wrinkled skin.
When studies conducted by Dr. Kligman, Dr. Voorhees, and by Barbara Gilcrest, MD, 9,10 showed that retinoids improved wrinkles, a major change in the focus in the skin care industry occurred.
During the same time period, the studies on alpha hydroxy acids by Chérie Ditre, MD, Eugene Van Scott, MD, and colleages11,12; and studies by Sheldon Pinnell, MD, on Vitamin C (see part 1 of this series) all demonstrated the efficacy of cosmetic ingredients on photoaged skin. This triggered a major change in how skin care products were marketed, with an efficacy approach rather than a safety approach.
With the shift from safety (hypoallergenic and noncomedogenic issues) to efficacy claims in the 1980s, and as nondrug active ingredients like retinol were shown to have biologic effects, the lines between the Food and Drug Administration’s definition of a drug versus a cosmetic became blurred. In 1984, Dr. Kligman suggested a new classification for the ingredients that fell in the middle, proposing the term “cosmeceutical” and thus, the concept of a cosmeceutical was introduced. To this day, cosmeceutical is not an official definition and the FDA has yet to deal with it as a quasi-drug category. FDA regulations as to what constitutes a drug versus a cosmetic date back to the 1938 Food, Drug and Cosmetic Act.
Once marketing focused on efficacy, many companies made outrageous claims. During the second half of the 1980s, the FDA issued some warning letters to some companies in an effort to control these claims.
Now efficacy claims abound and we, as dermatologists, should be the experts who back up these claims with scientific data. As the cosmeceutical market has evolved and grown, consumers are bewildered by the myriad of active ingredients being promoted and the number of products in the marketplace. As dermatologic innovation has led to more efficacious active ingredients, our patients look to us as knowledgeable and credible sources of information and for recommendations about the best skin care routines for their skin issues. This is all reflected in the fact that physician-dispensed skin care is becoming the fastest growing segment in this market. It is incumbent upon dermatologists to be knowledgeable and conversant about skin care products and skin care routines, and is particularly true for those of us who sell skin care products in our offices.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Burt’s Bees, Evolus, Galderma, and Revance. She is the CEO of Skin Type Solutions, a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Vogue Magazine, 1967 Aug 15. “Can Great Skin be Created?”
2. https://patents.google.com/patent/US3910284.
3. Kligman AM et al. J Am Acad Dermatol. 1986 Oct;15(4 Pt 2):836-59.
4. Weiss JS et al. JAMA. 1988 Jan 22-29;259(4):527-32.
5. Goldfarb MT et al. J Am Acad Dermatol. 1989 Sep;21(3 Pt 2):645-50.
6. Ellis CN et al. J Am Acad Dermatol. 1990 Oct;23(4 Pt 1):629-37.
7. Kang S; Voorhees JJ. J Am Acad Dermatol. 1998 Aug;39(2 Pt 3):S55-61.
8. Kafi R et al. Arch Dermatol. 2007 May;143(5):606-12.
9. Gilchrest BA. J Am Acad Dermatol. 1989 Sep;21(3 Pt 2):610-3.
10. Bhawan J et al. Arch Dermatol. 1991 May;127(5):666-72.
11. Griffin TD et al. J Am Acad Dermatol. 1996 Feb;34(2 Pt 1):196-203.
12. Ditre CM et al. J Am Acad Dermatol. 1996 Feb;34(2 Pt 1):187-95.
This is the third in a series of columns discussing the important roles that dermatologists have played in the skin care industry. with the cosmetic industry, rather than developing their own skin care lines.
Norman Orentreich, MD
Dr. Orentreich was a successful New York City dermatologist and the first to perform hair transplants. This new technique brought him fame and notoriety and arguably made him the first “celebrity dermatologist.” (He was also a member of the original advisory board of Dermatology News, at that time Skin & Allergy News, in January 1970.) Dr. Orentreich was a seminal figure in the trend to link the cosmetic industry and dermatology. In August 1967, Vogue magazine1 published an article on him, titled “Can Great Skin be Created?” This popular article caught the attention of Leonard Lauder, of Estée Lauder, who recruited Dr. Orentreich to help create the skin care line Clinique. Clinique was intended to be a brand with a medical look that promoted its products as “allergy tested,” with packaging that has an antiseptic look and beauty counter salespeople wearing white coats.
Dr. Orentreich’s input into the development of a skin type–based skin care line was fundamental to the development of this brand. The four-question questionnaire with an iconic plastic lever that customers slide left or right instantly provided them with an assessment of their skin type at the beauty counter, with one of four skin types: Very Dry to Dry Skin (Skin Type 1), Dry Combination (Skin Type 2), Combination Oily (Skin Type 3), and Oily (Skin Type 4).
Although this skin-typing system was not scientifically accurate (there is no scientific definition of combination skin), it was reminiscent of the system developed by cosmetic company tycoon Helena Rubinstein in the 1940s that classified people into four skin types: oily, dry, combination, and sensitive. Clinique became a blockbuster skin care brand and was one of the first developed by a dermatologist – although Dr. Orentreich did not put his name on it.
In 1972, Dr. Orentreich filed a patent2 for an exfoliating pad for the skin that later became known as the “Buf-Puf.” I heard years ago that he got the idea from the machines used to buff the floors in the hospital. The buffing pad had a hole in the center where the machine attached. Dr. Orentreich purportedly thought “I wonder what they do with the cut-out centers?” He looked into this, and subsequently used the centers to create the Buf-Puf. I cannot find a reference for this, but I love this story and hope it’s true. If any readers have any knowledge of this, please let me know, so I can amend my story if it is incorrect.
Almay
Almay, an amalgamation of the founders’ names, Alfred and Fanny May Woititz, was the first hypoallergenic brand, established in 1931, and the first to provide hypoallergenic cosmetics, long before Clinique. In addition, the company was the first skin care brand to become available by prescription only (as it was initially), fully disclose all individual ingredients in its products (well before this became mandatory in 1976), provide totally fragrance-free products, develop a hypoallergenic fragrance – and provide patch tests and other materials to physicians to identify contact allergens.
Over 90 years, the company was also the first among skin care brands to do the following:
- Provide custom formulations to individuals proven to be allergic to a specific ingredient, through their physicians.
- Perform a full range of premarket safety testing on all products for allergy and irritation, and test all its products for comedogenicity.
- Formulate cosmetics for use around the eye area (eye shadows and eyeliners) specifically for contact lens wearers.
- Formulate hypoallergenic regimens for specific skin types in the mass market.
- Provide a specific cosmetic regimen for acne-prone women, including a silicone-based makeup and active ingredients for treatment in cosmetics and skin care.
I recently interviewed Stanley Levy, MD, who was one of the consultants to Almay, and practices in Chapel Hill, N.C., where he has an academic niche related to skin care formulation and safety. He told me how Almay provided patch test materials to dermatologists to help identify contact dermatitis to cosmetic ingredients, and described Almay’s relationship with the dermatology field as follows: “From the outset, Almay was linked to dermatology. In 1930, a chemist and pharmacist in New York City, Al Woititz, was looking to compound cosmetics for his wife suffering from cosmetic allergies, Fannie May. He enlisted the counsel of the preeminent dermatologic expert in contact dermatitis at the time, Dr. Marion Sulzberger, to suggest ingredients to avoid. [Dr. Sulzberger was also a member of the original Dermatology News editorial advisory board.] Soon, dermatologists around New York City were recommending these formulations. This led to a product line free of the known allergens and a fledgling company trademarked as Almay. For the past 90 years, [the company] has kept a close relationship with dermatologists, well before that was the norm.”
The Almay research overseen by Dr. Levy and others contributed greatly to our understanding of the allergenicity of skin care.
Albert Kligman, MD
The turning point for the interface of dermatology with the cosmetic industry was the shift from a safety-based approach (hypoallergenic and noncomedogenic) to an emphasis on efficacy claims in the 1980s. Part of the impetus for this was the Dr. Kligman’s observation that retinoids could improve photoaging.
Dr. Kligman, a well-known dermatologist at the University of Pennsylvania, Philadelphia, showed that retinoids were an effective treatment for acne. For more about this, listen to my interview on the Dermatology Weekly podcast, with James Leyden, MD, about his work at the University of Pennsylvania with Dr. Kligman on the development of oral and topical retinoids. During Dr. Kligman’s research on acne, he noticed that wrinkles improved after treatment with tretinoin, and in 1986, he and Dr. Leyden (and several other authors) published the first article about tretinoin’s use for photoaged skin.3 This led to a double-blind study4 conducted by John J. Voorhees, MD, University of Michigan, Ann Arbor, and coauthors that showed statistically significant improvement of photoaged skin when treated with topical tretinoin. Dr. Voorhees and his group did many more studies on retinoids5,6 and photoaging7 – so many that, at one time, he was (and maybe still is) the most widely published dermatologist in the United States. These studies showed that, not only did prescription tretinoin improve the appearance of wrinkles, but so did over-the-counter retinol.8 Retinoids remain the most efficacious prescription and cosmeceutical ingredients to treat wrinkled skin.
When studies conducted by Dr. Kligman, Dr. Voorhees, and by Barbara Gilcrest, MD, 9,10 showed that retinoids improved wrinkles, a major change in the focus in the skin care industry occurred.
During the same time period, the studies on alpha hydroxy acids by Chérie Ditre, MD, Eugene Van Scott, MD, and colleages11,12; and studies by Sheldon Pinnell, MD, on Vitamin C (see part 1 of this series) all demonstrated the efficacy of cosmetic ingredients on photoaged skin. This triggered a major change in how skin care products were marketed, with an efficacy approach rather than a safety approach.
With the shift from safety (hypoallergenic and noncomedogenic issues) to efficacy claims in the 1980s, and as nondrug active ingredients like retinol were shown to have biologic effects, the lines between the Food and Drug Administration’s definition of a drug versus a cosmetic became blurred. In 1984, Dr. Kligman suggested a new classification for the ingredients that fell in the middle, proposing the term “cosmeceutical” and thus, the concept of a cosmeceutical was introduced. To this day, cosmeceutical is not an official definition and the FDA has yet to deal with it as a quasi-drug category. FDA regulations as to what constitutes a drug versus a cosmetic date back to the 1938 Food, Drug and Cosmetic Act.
Once marketing focused on efficacy, many companies made outrageous claims. During the second half of the 1980s, the FDA issued some warning letters to some companies in an effort to control these claims.
Now efficacy claims abound and we, as dermatologists, should be the experts who back up these claims with scientific data. As the cosmeceutical market has evolved and grown, consumers are bewildered by the myriad of active ingredients being promoted and the number of products in the marketplace. As dermatologic innovation has led to more efficacious active ingredients, our patients look to us as knowledgeable and credible sources of information and for recommendations about the best skin care routines for their skin issues. This is all reflected in the fact that physician-dispensed skin care is becoming the fastest growing segment in this market. It is incumbent upon dermatologists to be knowledgeable and conversant about skin care products and skin care routines, and is particularly true for those of us who sell skin care products in our offices.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Burt’s Bees, Evolus, Galderma, and Revance. She is the CEO of Skin Type Solutions, a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Vogue Magazine, 1967 Aug 15. “Can Great Skin be Created?”
2. https://patents.google.com/patent/US3910284.
3. Kligman AM et al. J Am Acad Dermatol. 1986 Oct;15(4 Pt 2):836-59.
4. Weiss JS et al. JAMA. 1988 Jan 22-29;259(4):527-32.
5. Goldfarb MT et al. J Am Acad Dermatol. 1989 Sep;21(3 Pt 2):645-50.
6. Ellis CN et al. J Am Acad Dermatol. 1990 Oct;23(4 Pt 1):629-37.
7. Kang S; Voorhees JJ. J Am Acad Dermatol. 1998 Aug;39(2 Pt 3):S55-61.
8. Kafi R et al. Arch Dermatol. 2007 May;143(5):606-12.
9. Gilchrest BA. J Am Acad Dermatol. 1989 Sep;21(3 Pt 2):610-3.
10. Bhawan J et al. Arch Dermatol. 1991 May;127(5):666-72.
11. Griffin TD et al. J Am Acad Dermatol. 1996 Feb;34(2 Pt 1):196-203.
12. Ditre CM et al. J Am Acad Dermatol. 1996 Feb;34(2 Pt 1):187-95.
Treating the jowl fat overhang with deoxycholic acid
Rejuvenation of the lower face often involves treatment of the submentum and the jowls. Energy-based devices such as lasers, radiofrequency, radiofrequency microneedling, CoolSculpting, and ultrasound have been used in the tightening of the neck and jowls.
However, the only noninvasive injectable treatment approved for the reduction of submental fat is deoxycholic acid (Kybella). The mechanism of action of deoxycholic acid has been documented as adipocyte lysis, followed by a local tissue response with neutrophil infiltration, septal thickening, neocollagenesis, and neovascularization within the subcutaneous layer, with no adverse changes in the dermis or epidermis. This treatment, which has a dose-dependent response, is highly effective for submental fat reduction and jaw contouring.
In my practice, I have found that multiple consecutive treatments with deoxycholic acid (an off-label use) are effective in permanently reducing the jowl overhang with minimal adverse effects.
Jowl fat is a common cause of sagging of the jowls, and there are few alternatives to treatment with surgery or liposuction. Jowl overhang results from multiple factors related to aging, including skeletal resorption, subcutaneous atrophy, superior and inferior fat pad compartment displacement, or mandibular septum dehiscence, which allows for the accumulation of fat pockets to migrate into the neck.
A prospective study published earlier this year describes results in 66 adults with excess jowl fat, who were treated with 2 mg/cm2 of deoxycholic acid. Injections were done in patients with “pinchable fat on the jawline” and “relatively” minimal skin laxity of 0.2 mL spaced approximately 1 cm apart or 0.1 mL spaced 0.5 cm-0.75 cm apart; the mean injection volume was 0.8 mL. After 6 months, 98% of the patients experienced improvement with a mean of 1.8 treatments. Common injection site adverse events included edema, numbness, tenderness, and bruising.
In my experience, injection volumes from 1.0 mL to 1.5 mL of deoxycholic acid can be used in each jowl with minimal adverse events if proper landmarks are followed. It is crucial that the correct patient is selected (one with minimal skin laxity), and that during injection, the fat and skin are pinched away from the underlying musculature and neurovascular structures to avoid injection near the marginal mandibular nerve. Volumes less than 1.0 mL have minimal visible improvements and will require more than 3-4 treatment sessions for optimal results.
. I often see a marked improvement in patients who present prominent marionette lines who have been unhappy with fillers in the lower face. Often, the marionette lines are a result of significant overhang from jowl fat and hyaluronic acid fillers are a temporary and often unsatisfactory treatment option. The use of deoxycholic acid in the treatment of the jowl fat is a highly effective option to minimize the appearance of marionette lines caused by displaced fat pockets in the aging lower face.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected] . They had no relevant disclosures.
Rejuvenation of the lower face often involves treatment of the submentum and the jowls. Energy-based devices such as lasers, radiofrequency, radiofrequency microneedling, CoolSculpting, and ultrasound have been used in the tightening of the neck and jowls.
However, the only noninvasive injectable treatment approved for the reduction of submental fat is deoxycholic acid (Kybella). The mechanism of action of deoxycholic acid has been documented as adipocyte lysis, followed by a local tissue response with neutrophil infiltration, septal thickening, neocollagenesis, and neovascularization within the subcutaneous layer, with no adverse changes in the dermis or epidermis. This treatment, which has a dose-dependent response, is highly effective for submental fat reduction and jaw contouring.
In my practice, I have found that multiple consecutive treatments with deoxycholic acid (an off-label use) are effective in permanently reducing the jowl overhang with minimal adverse effects.
Jowl fat is a common cause of sagging of the jowls, and there are few alternatives to treatment with surgery or liposuction. Jowl overhang results from multiple factors related to aging, including skeletal resorption, subcutaneous atrophy, superior and inferior fat pad compartment displacement, or mandibular septum dehiscence, which allows for the accumulation of fat pockets to migrate into the neck.
A prospective study published earlier this year describes results in 66 adults with excess jowl fat, who were treated with 2 mg/cm2 of deoxycholic acid. Injections were done in patients with “pinchable fat on the jawline” and “relatively” minimal skin laxity of 0.2 mL spaced approximately 1 cm apart or 0.1 mL spaced 0.5 cm-0.75 cm apart; the mean injection volume was 0.8 mL. After 6 months, 98% of the patients experienced improvement with a mean of 1.8 treatments. Common injection site adverse events included edema, numbness, tenderness, and bruising.
In my experience, injection volumes from 1.0 mL to 1.5 mL of deoxycholic acid can be used in each jowl with minimal adverse events if proper landmarks are followed. It is crucial that the correct patient is selected (one with minimal skin laxity), and that during injection, the fat and skin are pinched away from the underlying musculature and neurovascular structures to avoid injection near the marginal mandibular nerve. Volumes less than 1.0 mL have minimal visible improvements and will require more than 3-4 treatment sessions for optimal results.
. I often see a marked improvement in patients who present prominent marionette lines who have been unhappy with fillers in the lower face. Often, the marionette lines are a result of significant overhang from jowl fat and hyaluronic acid fillers are a temporary and often unsatisfactory treatment option. The use of deoxycholic acid in the treatment of the jowl fat is a highly effective option to minimize the appearance of marionette lines caused by displaced fat pockets in the aging lower face.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected] . They had no relevant disclosures.
Rejuvenation of the lower face often involves treatment of the submentum and the jowls. Energy-based devices such as lasers, radiofrequency, radiofrequency microneedling, CoolSculpting, and ultrasound have been used in the tightening of the neck and jowls.
However, the only noninvasive injectable treatment approved for the reduction of submental fat is deoxycholic acid (Kybella). The mechanism of action of deoxycholic acid has been documented as adipocyte lysis, followed by a local tissue response with neutrophil infiltration, septal thickening, neocollagenesis, and neovascularization within the subcutaneous layer, with no adverse changes in the dermis or epidermis. This treatment, which has a dose-dependent response, is highly effective for submental fat reduction and jaw contouring.
In my practice, I have found that multiple consecutive treatments with deoxycholic acid (an off-label use) are effective in permanently reducing the jowl overhang with minimal adverse effects.
Jowl fat is a common cause of sagging of the jowls, and there are few alternatives to treatment with surgery or liposuction. Jowl overhang results from multiple factors related to aging, including skeletal resorption, subcutaneous atrophy, superior and inferior fat pad compartment displacement, or mandibular septum dehiscence, which allows for the accumulation of fat pockets to migrate into the neck.
A prospective study published earlier this year describes results in 66 adults with excess jowl fat, who were treated with 2 mg/cm2 of deoxycholic acid. Injections were done in patients with “pinchable fat on the jawline” and “relatively” minimal skin laxity of 0.2 mL spaced approximately 1 cm apart or 0.1 mL spaced 0.5 cm-0.75 cm apart; the mean injection volume was 0.8 mL. After 6 months, 98% of the patients experienced improvement with a mean of 1.8 treatments. Common injection site adverse events included edema, numbness, tenderness, and bruising.
In my experience, injection volumes from 1.0 mL to 1.5 mL of deoxycholic acid can be used in each jowl with minimal adverse events if proper landmarks are followed. It is crucial that the correct patient is selected (one with minimal skin laxity), and that during injection, the fat and skin are pinched away from the underlying musculature and neurovascular structures to avoid injection near the marginal mandibular nerve. Volumes less than 1.0 mL have minimal visible improvements and will require more than 3-4 treatment sessions for optimal results.
. I often see a marked improvement in patients who present prominent marionette lines who have been unhappy with fillers in the lower face. Often, the marionette lines are a result of significant overhang from jowl fat and hyaluronic acid fillers are a temporary and often unsatisfactory treatment option. The use of deoxycholic acid in the treatment of the jowl fat is a highly effective option to minimize the appearance of marionette lines caused by displaced fat pockets in the aging lower face.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected] . They had no relevant disclosures.
New technologies show promise for treating pigmented lesions
carry a higher risk for postinflammatory hyperpigmentation than intense pulsed light or the long-pulsed laser, according to Mathew M. Avram, MD, JD.
For treating melanosomes with selective photothermolysis, some of the peak wavelengths include 532 nm, 694 nm, 755 nm, and 1064 nm, Dr. Avram, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital, Boston, said during the virtual annual Masters of Aesthetics Symposium. “The ideal target is fair skin with a dark, pigmented lesion,” he said. “That way you’re going to get energy focused to the melanin that’s in the lesion itself.”
Q-switched and picosecond lasers are effective for pigmented lesions. These employ as much energy as the city of Boston for 20-30 billionths of a second, or 750 picoseconds. “This raises the temperature to 1,000° C in that time, which produces the characteristic epidermal whitening,” he said. “This targets pigment cells only, whether it’s exogenous or endogenous pigment.”
Benign pigmented lesions amenable to the Q-switched nanosecond and picosecond laser include lentigines and nevus of Ota/Ito. The mechanism of action for clinical lightening is fragmentation and release of melanin-laden cells and the gradual uptake and removal of fragments by activated macrophages into lymphatic vessels. “For effective results, do not blindly memorize settings or replicate recommended settings from a colleague or a device manufacturer,” advised Dr. Avram, who practiced law prior to becoming a physician. “Some lasers are not externally calibrated, so what you have to do is pay attention to the laser endpoint, which in this case is epidermal whitening. Tissue ‘splatter’ is an unsafe endpoint and may lead to scarring. Safe and unsafe laser endpoints and close clinical observation are the best means to avoid complications and get the best results for your patients. The key finding is the endpoint, not the energy settings.”
Pigmented lesions that should not be treated with a laser include atypical nevi, lentigo maligna, and other forms of melanoma. “When in doubt, perform a biopsy,” he said. “Regardless of who referred the case, you are liable if you treat a melanoma with a laser. This is not only misdiagnosis but it probably delays diagnosis as well. If you cannot recognize basis pigmented lesion morphology, do not treat pigmented lesions. At some point, it’s going to catch up with you.”
Patients with more pigment to their skin face a higher risk for postinflammatory hyperpigmentation, Dr. Avram continued. While longer pulsed lasers produce less hyperpigmentation, they’re also less effective at getting rid of lesions. “You can combine a long-pulsed laser with fractional resurfacing or IPL [intense pulsed light] to optimize improvement,” he said. “If you don’t have two lasers to use, you can just use a longer-pulsed laser. The desired treatment endpoint for this approach is an ashen gray appearance.” Options include a 532-nm Nd:YAG laser with or without cooling, a 595-nm pulsed dye laser without cooling, and a 755-nm alexandrite laser without cooling.
One advance in the treatment of seborrheic keratoses is Nano-Pulse Stimulation (NPS), a novel technology being developed by Pulse Biosciences. With this approach, nanosecond electrical energy pulses cause internal organelle disruption, which leads to regulated cell death. “The cell-specific effect is nonthermal, as a typical nano-pulse delivers 0.1 joules of energy distributed in a volume of tissue,” Dr. Avram said. Early human studies established safe doses and validation of mechanism hypothesis for benign-lesion efficacy. “What you have are tiny nanopores that allow calcium ions to flow into the cell,” he explained. “The nanopores in the endoplasmic reticulum allow calcium ions to flow out of the endoplasmic reticulum, stressing it. These nanopores in the mitochondria disrupt the ability to generate energy, and the cell dies.”
Histology has revealed that within days the procedure causes regulated cell death with no thermal effects. The ability of NPS energy to clear seborrheic keratoses (SK) was confirmed in a study of 58 subjects who had 174 SK lesions treated. The majority of SKs (82%) were rated as clear or mostly clear 106 days post treatment. All results reflected a single treatment session.
Another novel treatment, “cryomodulation,” a technology being developed by R. Rox Anderson, MD, Dieter Manstein, MD, PhD, and Henry Chan, MD, PhD, expresses cold-induced change to the skin as a way to pause melanin production. “You get melanin production paused but melanocyte function is preserved,” Dr. Avram explained. “There is a normal epidermal barrier and no persistent inflammatory response, so there’s no hyperpigmentation.” He characterized it as an ease-of-use clinical procedure for treating benign lesions in all skin types. A mask is applied to confine freezing to the desired treatment area, and hydrated gauze is used to help facilitate ice crystal propagation. A prototype of the device features a parameter selection based on lesion type, anatomical location, and skin type. “It uses between 107 and 166 kJ/m2 of extracted energy, and you take photos at baseline and follow-up,” he said. “You get 2-3 days of redness, darkening, and swelling. It’s well tolerated, with minimal discomfort. There’s no long-term dyschromia. This is nice, because patients have little, if any, downtime.”
Dr. Avram disclosed that he has received consulting fees from Allergan, Merz, Sciton, and Soliton. He also reported having ownership and/or shareholder interest in Cytrellis.
carry a higher risk for postinflammatory hyperpigmentation than intense pulsed light or the long-pulsed laser, according to Mathew M. Avram, MD, JD.
For treating melanosomes with selective photothermolysis, some of the peak wavelengths include 532 nm, 694 nm, 755 nm, and 1064 nm, Dr. Avram, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital, Boston, said during the virtual annual Masters of Aesthetics Symposium. “The ideal target is fair skin with a dark, pigmented lesion,” he said. “That way you’re going to get energy focused to the melanin that’s in the lesion itself.”
Q-switched and picosecond lasers are effective for pigmented lesions. These employ as much energy as the city of Boston for 20-30 billionths of a second, or 750 picoseconds. “This raises the temperature to 1,000° C in that time, which produces the characteristic epidermal whitening,” he said. “This targets pigment cells only, whether it’s exogenous or endogenous pigment.”
Benign pigmented lesions amenable to the Q-switched nanosecond and picosecond laser include lentigines and nevus of Ota/Ito. The mechanism of action for clinical lightening is fragmentation and release of melanin-laden cells and the gradual uptake and removal of fragments by activated macrophages into lymphatic vessels. “For effective results, do not blindly memorize settings or replicate recommended settings from a colleague or a device manufacturer,” advised Dr. Avram, who practiced law prior to becoming a physician. “Some lasers are not externally calibrated, so what you have to do is pay attention to the laser endpoint, which in this case is epidermal whitening. Tissue ‘splatter’ is an unsafe endpoint and may lead to scarring. Safe and unsafe laser endpoints and close clinical observation are the best means to avoid complications and get the best results for your patients. The key finding is the endpoint, not the energy settings.”
Pigmented lesions that should not be treated with a laser include atypical nevi, lentigo maligna, and other forms of melanoma. “When in doubt, perform a biopsy,” he said. “Regardless of who referred the case, you are liable if you treat a melanoma with a laser. This is not only misdiagnosis but it probably delays diagnosis as well. If you cannot recognize basis pigmented lesion morphology, do not treat pigmented lesions. At some point, it’s going to catch up with you.”
Patients with more pigment to their skin face a higher risk for postinflammatory hyperpigmentation, Dr. Avram continued. While longer pulsed lasers produce less hyperpigmentation, they’re also less effective at getting rid of lesions. “You can combine a long-pulsed laser with fractional resurfacing or IPL [intense pulsed light] to optimize improvement,” he said. “If you don’t have two lasers to use, you can just use a longer-pulsed laser. The desired treatment endpoint for this approach is an ashen gray appearance.” Options include a 532-nm Nd:YAG laser with or without cooling, a 595-nm pulsed dye laser without cooling, and a 755-nm alexandrite laser without cooling.
One advance in the treatment of seborrheic keratoses is Nano-Pulse Stimulation (NPS), a novel technology being developed by Pulse Biosciences. With this approach, nanosecond electrical energy pulses cause internal organelle disruption, which leads to regulated cell death. “The cell-specific effect is nonthermal, as a typical nano-pulse delivers 0.1 joules of energy distributed in a volume of tissue,” Dr. Avram said. Early human studies established safe doses and validation of mechanism hypothesis for benign-lesion efficacy. “What you have are tiny nanopores that allow calcium ions to flow into the cell,” he explained. “The nanopores in the endoplasmic reticulum allow calcium ions to flow out of the endoplasmic reticulum, stressing it. These nanopores in the mitochondria disrupt the ability to generate energy, and the cell dies.”
Histology has revealed that within days the procedure causes regulated cell death with no thermal effects. The ability of NPS energy to clear seborrheic keratoses (SK) was confirmed in a study of 58 subjects who had 174 SK lesions treated. The majority of SKs (82%) were rated as clear or mostly clear 106 days post treatment. All results reflected a single treatment session.
Another novel treatment, “cryomodulation,” a technology being developed by R. Rox Anderson, MD, Dieter Manstein, MD, PhD, and Henry Chan, MD, PhD, expresses cold-induced change to the skin as a way to pause melanin production. “You get melanin production paused but melanocyte function is preserved,” Dr. Avram explained. “There is a normal epidermal barrier and no persistent inflammatory response, so there’s no hyperpigmentation.” He characterized it as an ease-of-use clinical procedure for treating benign lesions in all skin types. A mask is applied to confine freezing to the desired treatment area, and hydrated gauze is used to help facilitate ice crystal propagation. A prototype of the device features a parameter selection based on lesion type, anatomical location, and skin type. “It uses between 107 and 166 kJ/m2 of extracted energy, and you take photos at baseline and follow-up,” he said. “You get 2-3 days of redness, darkening, and swelling. It’s well tolerated, with minimal discomfort. There’s no long-term dyschromia. This is nice, because patients have little, if any, downtime.”
Dr. Avram disclosed that he has received consulting fees from Allergan, Merz, Sciton, and Soliton. He also reported having ownership and/or shareholder interest in Cytrellis.
carry a higher risk for postinflammatory hyperpigmentation than intense pulsed light or the long-pulsed laser, according to Mathew M. Avram, MD, JD.
For treating melanosomes with selective photothermolysis, some of the peak wavelengths include 532 nm, 694 nm, 755 nm, and 1064 nm, Dr. Avram, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital, Boston, said during the virtual annual Masters of Aesthetics Symposium. “The ideal target is fair skin with a dark, pigmented lesion,” he said. “That way you’re going to get energy focused to the melanin that’s in the lesion itself.”
Q-switched and picosecond lasers are effective for pigmented lesions. These employ as much energy as the city of Boston for 20-30 billionths of a second, or 750 picoseconds. “This raises the temperature to 1,000° C in that time, which produces the characteristic epidermal whitening,” he said. “This targets pigment cells only, whether it’s exogenous or endogenous pigment.”
Benign pigmented lesions amenable to the Q-switched nanosecond and picosecond laser include lentigines and nevus of Ota/Ito. The mechanism of action for clinical lightening is fragmentation and release of melanin-laden cells and the gradual uptake and removal of fragments by activated macrophages into lymphatic vessels. “For effective results, do not blindly memorize settings or replicate recommended settings from a colleague or a device manufacturer,” advised Dr. Avram, who practiced law prior to becoming a physician. “Some lasers are not externally calibrated, so what you have to do is pay attention to the laser endpoint, which in this case is epidermal whitening. Tissue ‘splatter’ is an unsafe endpoint and may lead to scarring. Safe and unsafe laser endpoints and close clinical observation are the best means to avoid complications and get the best results for your patients. The key finding is the endpoint, not the energy settings.”
Pigmented lesions that should not be treated with a laser include atypical nevi, lentigo maligna, and other forms of melanoma. “When in doubt, perform a biopsy,” he said. “Regardless of who referred the case, you are liable if you treat a melanoma with a laser. This is not only misdiagnosis but it probably delays diagnosis as well. If you cannot recognize basis pigmented lesion morphology, do not treat pigmented lesions. At some point, it’s going to catch up with you.”
Patients with more pigment to their skin face a higher risk for postinflammatory hyperpigmentation, Dr. Avram continued. While longer pulsed lasers produce less hyperpigmentation, they’re also less effective at getting rid of lesions. “You can combine a long-pulsed laser with fractional resurfacing or IPL [intense pulsed light] to optimize improvement,” he said. “If you don’t have two lasers to use, you can just use a longer-pulsed laser. The desired treatment endpoint for this approach is an ashen gray appearance.” Options include a 532-nm Nd:YAG laser with or without cooling, a 595-nm pulsed dye laser without cooling, and a 755-nm alexandrite laser without cooling.
One advance in the treatment of seborrheic keratoses is Nano-Pulse Stimulation (NPS), a novel technology being developed by Pulse Biosciences. With this approach, nanosecond electrical energy pulses cause internal organelle disruption, which leads to regulated cell death. “The cell-specific effect is nonthermal, as a typical nano-pulse delivers 0.1 joules of energy distributed in a volume of tissue,” Dr. Avram said. Early human studies established safe doses and validation of mechanism hypothesis for benign-lesion efficacy. “What you have are tiny nanopores that allow calcium ions to flow into the cell,” he explained. “The nanopores in the endoplasmic reticulum allow calcium ions to flow out of the endoplasmic reticulum, stressing it. These nanopores in the mitochondria disrupt the ability to generate energy, and the cell dies.”
Histology has revealed that within days the procedure causes regulated cell death with no thermal effects. The ability of NPS energy to clear seborrheic keratoses (SK) was confirmed in a study of 58 subjects who had 174 SK lesions treated. The majority of SKs (82%) were rated as clear or mostly clear 106 days post treatment. All results reflected a single treatment session.
Another novel treatment, “cryomodulation,” a technology being developed by R. Rox Anderson, MD, Dieter Manstein, MD, PhD, and Henry Chan, MD, PhD, expresses cold-induced change to the skin as a way to pause melanin production. “You get melanin production paused but melanocyte function is preserved,” Dr. Avram explained. “There is a normal epidermal barrier and no persistent inflammatory response, so there’s no hyperpigmentation.” He characterized it as an ease-of-use clinical procedure for treating benign lesions in all skin types. A mask is applied to confine freezing to the desired treatment area, and hydrated gauze is used to help facilitate ice crystal propagation. A prototype of the device features a parameter selection based on lesion type, anatomical location, and skin type. “It uses between 107 and 166 kJ/m2 of extracted energy, and you take photos at baseline and follow-up,” he said. “You get 2-3 days of redness, darkening, and swelling. It’s well tolerated, with minimal discomfort. There’s no long-term dyschromia. This is nice, because patients have little, if any, downtime.”
Dr. Avram disclosed that he has received consulting fees from Allergan, Merz, Sciton, and Soliton. He also reported having ownership and/or shareholder interest in Cytrellis.
FROM MOA 2020
Expert offers tips for combining lasers and injectables on the same day
While if it involves the same area.
“Swelling from the laser can potentially make the toxin migrate and cause ptosis,” Arisa E. Ortiz, MD, said at the virtual annual Masters of Aesthetics Symposium. “Even though this is temporary, your patient’s not going to be very happy with you. I would separate these at least 1 day apart, and then you should be OK.”
When using a filler on the same day as a laser treatment, Dr. Ortiz, who is director of laser and cosmetic dermatology at the University of California, San Diego, performs the laser procedure after injecting the filler, “because you may get some swelling, which can distort your need for filler,” she said. “I like to do the filler first to make sure I can assess how much volume loss they have. Then I’ll do the laser procedure right after.”
Another general rule of thumb is that, when combining lasers on the same day, consider lowering the device settings, “because it’s going to be a more aggressive treatment when you’re combining various laser procedures,” she said. “Treat vascular lesions first to not exacerbate nonspecific erythema. Then treat pigment, then resurfacing, followed by liquid nitrogen if needed to treat seborrheic keratoses.”
For periorbital rejuvenation, Dr. Ortiz likes to use a neurotoxin 1 week before performing the laser-resurfacing or skin-tightening procedure, followed by injection of a filler. “This augments your results,” she said. “Studies have shown that, if you start with a neuromodulator, you can get more improvement with your resurfacing procedure,” she said. “That makes sense, because you’re not contracting the muscle while you’re healing from the laser, so you get more effective collagen remodeling.”
When using a neuromodulator for dynamic periorbital rhytides, place it superficially to avoid bruising and stay superior to the maxillary prominence to avoid the zygomaticus major “so you don’t get a droopy smile,” she said. “The approved dosing is 24 units, 12 on each side. Less may be required for younger patients and more for more severe rhytides.”
For static rhytides, fractional resurfacing procedures will provide a more modest result with less downtime, while fully ablative laser resurfacing procedures will provide more dramatic improvement with more downtime. “You’re really going to tailor your treatment to what the patient is looking for,” Dr. Ortiz said. “If you use a fractional device you may need multiple treatments. Using a corneal shield when you’re resurfacing within the periorbital rim is a must, so you need to know how to place these if you’re going to be resurfacing in that area.”
For anesthesia, Dr. Ortiz likes to use injectable lidocaine, “because if you use a topical it can creep into the eye, and then you get a chemical corneal abrasion. This resolves after a few days but it’s really painful and your patient won’t be very happy.”
For tear troughs, use a hyaluronic acid filler with a low G prime. “If you use a thicker filler it can look lumpy or too full,” she said. While some clinicians use a needle to administer the filler, Dr. Ortiz prefers to use a blunt-tipped cannula. “It’s less painful and there’s less risk of bruising or swelling,” she said. “There’s also less risk of cannulizing a vessel. This is not zero risk. It’s been shown that the 27-gauge can actually cannulize the vessel, so it shouldn’t give you a false sense of security, but there is less risk, compared with using a needle. You can use the cannula to thread. If you’re using a needle you can inject a bolus and then massage it in, or you can use the microdroplet technique.”
With the cannula technique, bruising or swelling can occur even in the most experienced hands, “so make sure your patients don’t have an important event coming up,” Dr. Ortiz said. “With filler, not only do you improve the volume loss, but sometimes you improve the dark circles. I tend to see this more in lighter-skinned patients. In darker-skinned patients, the dark circles can be caused by racial pigmentation. That’s hard to fix, so I never promise that we can improve dark circles, but sometimes it does improve.”
For dynamic perioral rhytides, Dr. Ortiz generally treats with a neuromodulator 1 week in advance of laser resurfacing, followed by a filler for any etched-in lines. Use of a neuromodulator in the perioral region of musicians or singers is contraindicated “because it can affect their phonation,” she said. “Also, older patients might complain that it’s difficult for them to pucker their lips when they’re putting on a lip liner or lipstick. There are four injection sites on the upper lip and two on the lower lip. I do 1 unit at each injection site, with a max of 6-8 units. Any more than that and they’ll have difficulty puckering.”
Two main options for treating submental fullness include cryolipolysis or deoxycholic acid. “If you have a lot of volume, you want to use cryolipolysis,” Dr. Ortiz said. “The general rule is, if it fits in the cup [of the applicator], hook them up.” Use deoxycholic acid for areas of smaller volume, or to fine-tune, she added.
For platysmal bands, Dr. Ortiz favors injecting 2 units of botulinum toxin at three to four sites along the band. She pulls away and injects superficially and limits the treatment dose to 40 units in one session “because excessive doses can cause dysphagia,” she said. “If they need additional units, I’ll have them come back in 2 weeks.”
The Nefertiti lift combines the treatment of the platysma with the insertion point of the platysma along the jawline. Treatment of the patient along the lateral jawline with 2 units of botulinum toxin every centimeter or so can actually improve the definition of the jawline, “because your platysma is pulling down on your lower face,” Dr. Ortiz explained. “So, if you relax that, it can help to define the jawline. By treating the platysma, you can also prevent or soften the horizontal bands that occur across the neck.”
For necklace creases, she likes to inject 1-2 units of a low-HA filler along the crease – evenly spaced all along. “I’ll dilute it even further with 0.5 cc of lidocaine with epinephrine,” she said. “Then you can do serial punctures or you can thread along that line.”
For treating static rhytides on the neck, laser-resurfacing procedures work best, but at low settings. “Because there are fewer adnexal structures, the neck is at increased risk for scarring,” Dr. Ortiz said. “You want to use a lower fluence because your neck skin is thin. Your fluence determines your depth with resurfacing. Most importantly, use a lower density for a more conservative setting”
Options for treating poikiloderma of Civatte include the vascular laser, an IPL [intense pulsed light device], or a 1927-nm thulium laser. To avoid footprinting, or a “chicken wire” appearance to the treated area, Dr. Ortiz recommends using a large spot size with the pulsed dye laser or the IPL.
She concluded her presentation by underscoring the importance of communicating realistic expectations with patients. “There is some delayed gratification here,” she said. “For procedures that take time to see results, consider adding another procedure that will give them immediate results.”
Dr. Ortiz disclosed having financial relationships with numerous pharmaceutical and device companies. She is also cochair of the MOA.
While if it involves the same area.
“Swelling from the laser can potentially make the toxin migrate and cause ptosis,” Arisa E. Ortiz, MD, said at the virtual annual Masters of Aesthetics Symposium. “Even though this is temporary, your patient’s not going to be very happy with you. I would separate these at least 1 day apart, and then you should be OK.”
When using a filler on the same day as a laser treatment, Dr. Ortiz, who is director of laser and cosmetic dermatology at the University of California, San Diego, performs the laser procedure after injecting the filler, “because you may get some swelling, which can distort your need for filler,” she said. “I like to do the filler first to make sure I can assess how much volume loss they have. Then I’ll do the laser procedure right after.”
Another general rule of thumb is that, when combining lasers on the same day, consider lowering the device settings, “because it’s going to be a more aggressive treatment when you’re combining various laser procedures,” she said. “Treat vascular lesions first to not exacerbate nonspecific erythema. Then treat pigment, then resurfacing, followed by liquid nitrogen if needed to treat seborrheic keratoses.”
For periorbital rejuvenation, Dr. Ortiz likes to use a neurotoxin 1 week before performing the laser-resurfacing or skin-tightening procedure, followed by injection of a filler. “This augments your results,” she said. “Studies have shown that, if you start with a neuromodulator, you can get more improvement with your resurfacing procedure,” she said. “That makes sense, because you’re not contracting the muscle while you’re healing from the laser, so you get more effective collagen remodeling.”
When using a neuromodulator for dynamic periorbital rhytides, place it superficially to avoid bruising and stay superior to the maxillary prominence to avoid the zygomaticus major “so you don’t get a droopy smile,” she said. “The approved dosing is 24 units, 12 on each side. Less may be required for younger patients and more for more severe rhytides.”
For static rhytides, fractional resurfacing procedures will provide a more modest result with less downtime, while fully ablative laser resurfacing procedures will provide more dramatic improvement with more downtime. “You’re really going to tailor your treatment to what the patient is looking for,” Dr. Ortiz said. “If you use a fractional device you may need multiple treatments. Using a corneal shield when you’re resurfacing within the periorbital rim is a must, so you need to know how to place these if you’re going to be resurfacing in that area.”
For anesthesia, Dr. Ortiz likes to use injectable lidocaine, “because if you use a topical it can creep into the eye, and then you get a chemical corneal abrasion. This resolves after a few days but it’s really painful and your patient won’t be very happy.”
For tear troughs, use a hyaluronic acid filler with a low G prime. “If you use a thicker filler it can look lumpy or too full,” she said. While some clinicians use a needle to administer the filler, Dr. Ortiz prefers to use a blunt-tipped cannula. “It’s less painful and there’s less risk of bruising or swelling,” she said. “There’s also less risk of cannulizing a vessel. This is not zero risk. It’s been shown that the 27-gauge can actually cannulize the vessel, so it shouldn’t give you a false sense of security, but there is less risk, compared with using a needle. You can use the cannula to thread. If you’re using a needle you can inject a bolus and then massage it in, or you can use the microdroplet technique.”
With the cannula technique, bruising or swelling can occur even in the most experienced hands, “so make sure your patients don’t have an important event coming up,” Dr. Ortiz said. “With filler, not only do you improve the volume loss, but sometimes you improve the dark circles. I tend to see this more in lighter-skinned patients. In darker-skinned patients, the dark circles can be caused by racial pigmentation. That’s hard to fix, so I never promise that we can improve dark circles, but sometimes it does improve.”
For dynamic perioral rhytides, Dr. Ortiz generally treats with a neuromodulator 1 week in advance of laser resurfacing, followed by a filler for any etched-in lines. Use of a neuromodulator in the perioral region of musicians or singers is contraindicated “because it can affect their phonation,” she said. “Also, older patients might complain that it’s difficult for them to pucker their lips when they’re putting on a lip liner or lipstick. There are four injection sites on the upper lip and two on the lower lip. I do 1 unit at each injection site, with a max of 6-8 units. Any more than that and they’ll have difficulty puckering.”
Two main options for treating submental fullness include cryolipolysis or deoxycholic acid. “If you have a lot of volume, you want to use cryolipolysis,” Dr. Ortiz said. “The general rule is, if it fits in the cup [of the applicator], hook them up.” Use deoxycholic acid for areas of smaller volume, or to fine-tune, she added.
For platysmal bands, Dr. Ortiz favors injecting 2 units of botulinum toxin at three to four sites along the band. She pulls away and injects superficially and limits the treatment dose to 40 units in one session “because excessive doses can cause dysphagia,” she said. “If they need additional units, I’ll have them come back in 2 weeks.”
The Nefertiti lift combines the treatment of the platysma with the insertion point of the platysma along the jawline. Treatment of the patient along the lateral jawline with 2 units of botulinum toxin every centimeter or so can actually improve the definition of the jawline, “because your platysma is pulling down on your lower face,” Dr. Ortiz explained. “So, if you relax that, it can help to define the jawline. By treating the platysma, you can also prevent or soften the horizontal bands that occur across the neck.”
For necklace creases, she likes to inject 1-2 units of a low-HA filler along the crease – evenly spaced all along. “I’ll dilute it even further with 0.5 cc of lidocaine with epinephrine,” she said. “Then you can do serial punctures or you can thread along that line.”
For treating static rhytides on the neck, laser-resurfacing procedures work best, but at low settings. “Because there are fewer adnexal structures, the neck is at increased risk for scarring,” Dr. Ortiz said. “You want to use a lower fluence because your neck skin is thin. Your fluence determines your depth with resurfacing. Most importantly, use a lower density for a more conservative setting”
Options for treating poikiloderma of Civatte include the vascular laser, an IPL [intense pulsed light device], or a 1927-nm thulium laser. To avoid footprinting, or a “chicken wire” appearance to the treated area, Dr. Ortiz recommends using a large spot size with the pulsed dye laser or the IPL.
She concluded her presentation by underscoring the importance of communicating realistic expectations with patients. “There is some delayed gratification here,” she said. “For procedures that take time to see results, consider adding another procedure that will give them immediate results.”
Dr. Ortiz disclosed having financial relationships with numerous pharmaceutical and device companies. She is also cochair of the MOA.
While if it involves the same area.
“Swelling from the laser can potentially make the toxin migrate and cause ptosis,” Arisa E. Ortiz, MD, said at the virtual annual Masters of Aesthetics Symposium. “Even though this is temporary, your patient’s not going to be very happy with you. I would separate these at least 1 day apart, and then you should be OK.”
When using a filler on the same day as a laser treatment, Dr. Ortiz, who is director of laser and cosmetic dermatology at the University of California, San Diego, performs the laser procedure after injecting the filler, “because you may get some swelling, which can distort your need for filler,” she said. “I like to do the filler first to make sure I can assess how much volume loss they have. Then I’ll do the laser procedure right after.”
Another general rule of thumb is that, when combining lasers on the same day, consider lowering the device settings, “because it’s going to be a more aggressive treatment when you’re combining various laser procedures,” she said. “Treat vascular lesions first to not exacerbate nonspecific erythema. Then treat pigment, then resurfacing, followed by liquid nitrogen if needed to treat seborrheic keratoses.”
For periorbital rejuvenation, Dr. Ortiz likes to use a neurotoxin 1 week before performing the laser-resurfacing or skin-tightening procedure, followed by injection of a filler. “This augments your results,” she said. “Studies have shown that, if you start with a neuromodulator, you can get more improvement with your resurfacing procedure,” she said. “That makes sense, because you’re not contracting the muscle while you’re healing from the laser, so you get more effective collagen remodeling.”
When using a neuromodulator for dynamic periorbital rhytides, place it superficially to avoid bruising and stay superior to the maxillary prominence to avoid the zygomaticus major “so you don’t get a droopy smile,” she said. “The approved dosing is 24 units, 12 on each side. Less may be required for younger patients and more for more severe rhytides.”
For static rhytides, fractional resurfacing procedures will provide a more modest result with less downtime, while fully ablative laser resurfacing procedures will provide more dramatic improvement with more downtime. “You’re really going to tailor your treatment to what the patient is looking for,” Dr. Ortiz said. “If you use a fractional device you may need multiple treatments. Using a corneal shield when you’re resurfacing within the periorbital rim is a must, so you need to know how to place these if you’re going to be resurfacing in that area.”
For anesthesia, Dr. Ortiz likes to use injectable lidocaine, “because if you use a topical it can creep into the eye, and then you get a chemical corneal abrasion. This resolves after a few days but it’s really painful and your patient won’t be very happy.”
For tear troughs, use a hyaluronic acid filler with a low G prime. “If you use a thicker filler it can look lumpy or too full,” she said. While some clinicians use a needle to administer the filler, Dr. Ortiz prefers to use a blunt-tipped cannula. “It’s less painful and there’s less risk of bruising or swelling,” she said. “There’s also less risk of cannulizing a vessel. This is not zero risk. It’s been shown that the 27-gauge can actually cannulize the vessel, so it shouldn’t give you a false sense of security, but there is less risk, compared with using a needle. You can use the cannula to thread. If you’re using a needle you can inject a bolus and then massage it in, or you can use the microdroplet technique.”
With the cannula technique, bruising or swelling can occur even in the most experienced hands, “so make sure your patients don’t have an important event coming up,” Dr. Ortiz said. “With filler, not only do you improve the volume loss, but sometimes you improve the dark circles. I tend to see this more in lighter-skinned patients. In darker-skinned patients, the dark circles can be caused by racial pigmentation. That’s hard to fix, so I never promise that we can improve dark circles, but sometimes it does improve.”
For dynamic perioral rhytides, Dr. Ortiz generally treats with a neuromodulator 1 week in advance of laser resurfacing, followed by a filler for any etched-in lines. Use of a neuromodulator in the perioral region of musicians or singers is contraindicated “because it can affect their phonation,” she said. “Also, older patients might complain that it’s difficult for them to pucker their lips when they’re putting on a lip liner or lipstick. There are four injection sites on the upper lip and two on the lower lip. I do 1 unit at each injection site, with a max of 6-8 units. Any more than that and they’ll have difficulty puckering.”
Two main options for treating submental fullness include cryolipolysis or deoxycholic acid. “If you have a lot of volume, you want to use cryolipolysis,” Dr. Ortiz said. “The general rule is, if it fits in the cup [of the applicator], hook them up.” Use deoxycholic acid for areas of smaller volume, or to fine-tune, she added.
For platysmal bands, Dr. Ortiz favors injecting 2 units of botulinum toxin at three to four sites along the band. She pulls away and injects superficially and limits the treatment dose to 40 units in one session “because excessive doses can cause dysphagia,” she said. “If they need additional units, I’ll have them come back in 2 weeks.”
The Nefertiti lift combines the treatment of the platysma with the insertion point of the platysma along the jawline. Treatment of the patient along the lateral jawline with 2 units of botulinum toxin every centimeter or so can actually improve the definition of the jawline, “because your platysma is pulling down on your lower face,” Dr. Ortiz explained. “So, if you relax that, it can help to define the jawline. By treating the platysma, you can also prevent or soften the horizontal bands that occur across the neck.”
For necklace creases, she likes to inject 1-2 units of a low-HA filler along the crease – evenly spaced all along. “I’ll dilute it even further with 0.5 cc of lidocaine with epinephrine,” she said. “Then you can do serial punctures or you can thread along that line.”
For treating static rhytides on the neck, laser-resurfacing procedures work best, but at low settings. “Because there are fewer adnexal structures, the neck is at increased risk for scarring,” Dr. Ortiz said. “You want to use a lower fluence because your neck skin is thin. Your fluence determines your depth with resurfacing. Most importantly, use a lower density for a more conservative setting”
Options for treating poikiloderma of Civatte include the vascular laser, an IPL [intense pulsed light device], or a 1927-nm thulium laser. To avoid footprinting, or a “chicken wire” appearance to the treated area, Dr. Ortiz recommends using a large spot size with the pulsed dye laser or the IPL.
She concluded her presentation by underscoring the importance of communicating realistic expectations with patients. “There is some delayed gratification here,” she said. “For procedures that take time to see results, consider adding another procedure that will give them immediate results.”
Dr. Ortiz disclosed having financial relationships with numerous pharmaceutical and device companies. She is also cochair of the MOA.
EXPERT ANALYSIS FROM MOA 2020
Counterfeits: An ugly truth in aesthetic medicine
according to the results of two recent surveys of such providers.

“Counterfeit medical devices and injectables may be more prevalent in aesthetic medicine than most practitioners might estimate,” Jordan V. Wang, MD, of Sidney Kimmel Medical College, Philadelphia, and associates wrote in Dermatologic Surgery, even though “the vast majority [believe] that they are inferior and even potentially harmful.”
In one of the online surveys, conducted among members of the American Society of Dermatologic Surgery, 41.1% of the 616 respondents said they had encountered counterfeit injectables, more than half (56.5%) of whom were solicited to buy such products. Just over 10% had purchased counterfeit injectables, although nearly 80% did so unknowingly, the investigators said.
In the second survey, 37.4% of the 765 respondents (members of the ASDS as well as the American Society for Laser Medicine and Surgery) said that they had encountered counterfeit medical devices, and nearly half had been approached to purchase such devices. Of those who were approached, 4.6% had actually purchased a counterfeit, but only 6.1% did so unknowingly, Dr. Wang and associates reported.
In the medical device survey, almost a quarter (24.2%) acknowledged that they know of other providers using them, while 29.3% of those surveyed about injectables know of others who use counterfeits, they said.
Over 90% of practitioners in each survey agreed that counterfeits are worse in terms of safety, reliability, and effectiveness, but the proportions were smaller when they were asked if counterfeits were either very or extremely endangering to patient safety: 70.5% for injectables and 59.2% for devices, the investigators said.
Experience with adverse events from counterfeits in patients was reported by 39.7% of respondents to the injectables survey and by 20.1% of those in the device survey, they added.
Majorities in both surveys – 73.7% for injectables and 68.9% for devices – also said that they were either not familiar or only somewhat familiar with the Food and Drug Administration’s regulations on counterfeits. “This is especially problematic considering the potentially severe adverse events and steep punishments,” Dr. Wang and associates wrote.
The authors disclosed that they had no significant interest with commercial supporters. Dr. Wang is now a fellow at the Laser & Skin Surgery Center of New York.
SOURCE: Wang JV et al. Dermatol. Surg. 2020 Oct;46(10):1323-6.
according to the results of two recent surveys of such providers.

“Counterfeit medical devices and injectables may be more prevalent in aesthetic medicine than most practitioners might estimate,” Jordan V. Wang, MD, of Sidney Kimmel Medical College, Philadelphia, and associates wrote in Dermatologic Surgery, even though “the vast majority [believe] that they are inferior and even potentially harmful.”
In one of the online surveys, conducted among members of the American Society of Dermatologic Surgery, 41.1% of the 616 respondents said they had encountered counterfeit injectables, more than half (56.5%) of whom were solicited to buy such products. Just over 10% had purchased counterfeit injectables, although nearly 80% did so unknowingly, the investigators said.
In the second survey, 37.4% of the 765 respondents (members of the ASDS as well as the American Society for Laser Medicine and Surgery) said that they had encountered counterfeit medical devices, and nearly half had been approached to purchase such devices. Of those who were approached, 4.6% had actually purchased a counterfeit, but only 6.1% did so unknowingly, Dr. Wang and associates reported.
In the medical device survey, almost a quarter (24.2%) acknowledged that they know of other providers using them, while 29.3% of those surveyed about injectables know of others who use counterfeits, they said.
Over 90% of practitioners in each survey agreed that counterfeits are worse in terms of safety, reliability, and effectiveness, but the proportions were smaller when they were asked if counterfeits were either very or extremely endangering to patient safety: 70.5% for injectables and 59.2% for devices, the investigators said.
Experience with adverse events from counterfeits in patients was reported by 39.7% of respondents to the injectables survey and by 20.1% of those in the device survey, they added.
Majorities in both surveys – 73.7% for injectables and 68.9% for devices – also said that they were either not familiar or only somewhat familiar with the Food and Drug Administration’s regulations on counterfeits. “This is especially problematic considering the potentially severe adverse events and steep punishments,” Dr. Wang and associates wrote.
The authors disclosed that they had no significant interest with commercial supporters. Dr. Wang is now a fellow at the Laser & Skin Surgery Center of New York.
SOURCE: Wang JV et al. Dermatol. Surg. 2020 Oct;46(10):1323-6.
according to the results of two recent surveys of such providers.

“Counterfeit medical devices and injectables may be more prevalent in aesthetic medicine than most practitioners might estimate,” Jordan V. Wang, MD, of Sidney Kimmel Medical College, Philadelphia, and associates wrote in Dermatologic Surgery, even though “the vast majority [believe] that they are inferior and even potentially harmful.”
In one of the online surveys, conducted among members of the American Society of Dermatologic Surgery, 41.1% of the 616 respondents said they had encountered counterfeit injectables, more than half (56.5%) of whom were solicited to buy such products. Just over 10% had purchased counterfeit injectables, although nearly 80% did so unknowingly, the investigators said.
In the second survey, 37.4% of the 765 respondents (members of the ASDS as well as the American Society for Laser Medicine and Surgery) said that they had encountered counterfeit medical devices, and nearly half had been approached to purchase such devices. Of those who were approached, 4.6% had actually purchased a counterfeit, but only 6.1% did so unknowingly, Dr. Wang and associates reported.
In the medical device survey, almost a quarter (24.2%) acknowledged that they know of other providers using them, while 29.3% of those surveyed about injectables know of others who use counterfeits, they said.
Over 90% of practitioners in each survey agreed that counterfeits are worse in terms of safety, reliability, and effectiveness, but the proportions were smaller when they were asked if counterfeits were either very or extremely endangering to patient safety: 70.5% for injectables and 59.2% for devices, the investigators said.
Experience with adverse events from counterfeits in patients was reported by 39.7% of respondents to the injectables survey and by 20.1% of those in the device survey, they added.
Majorities in both surveys – 73.7% for injectables and 68.9% for devices – also said that they were either not familiar or only somewhat familiar with the Food and Drug Administration’s regulations on counterfeits. “This is especially problematic considering the potentially severe adverse events and steep punishments,” Dr. Wang and associates wrote.
The authors disclosed that they had no significant interest with commercial supporters. Dr. Wang is now a fellow at the Laser & Skin Surgery Center of New York.
SOURCE: Wang JV et al. Dermatol. Surg. 2020 Oct;46(10):1323-6.
FROM DERMATOLOGIC SURGERY
Tailoring cosmetic procedures for skin of color patients minimize risks
Based on the fact that hyperpigmentation and other adverse events associated with cosmetic dermatologic procedures are relevant to skin type, not racial identification, individualized strategies to minimize the risk of potential adverse events are always appropriate, according to an expert speaking at the virtual Skin of Color Update 2020.
There are many highly effective interventions that employ lasers, chemical peels, and topical agents to achieve excellent cosmetic results in darker skin, but results are highly dependent on first understanding the relative risks and treatment goals, Cheryl Burgess, MD, president and founder of the Center for Dermatology and Dermatologic Surgery, Washington, D.C., said at the meeting.
She stressed the importance of educating patients that “all cosmetic procedures are not for skin of color.” Her approach is to engage patients on what they are trying to accomplish and then seeking a solution that tailors treatment to skin type based on the Fitzpatrick scale, the Roberts Hyperpigmentation Scale, or other guidance.
“There are so many different methods that we can use, and these are not necessarily the ones that patients have read about in a magazine,” Dr. Burgess said.
Intense pulsed laser (IPL) for hair removal is an example. This technique is not appropriate in patients with Fitzpatrick skin type IV or higher, according to Dr. Burgess, who presented a case example of a bad outcome. In this case, a patient came to her for treatment after exposure to IPL resulted in first- and second-degree burns complicated by extensive hypopigmentation.
Ultimately, the solution in this case involved more laser therapy, but this time a strategy was selected appropriate to skin of color.
“It is hard to suggest to a patient that we are going to use a laser device” when the problem was caused by a laser, Dr. Burgess observed, but properly selected lasers are effective and should be considered in patients with dark skin.
In this case, triple cream containing 6% hydroquinone was the first step towards resolving the hyperpigmentation. Jessner’s peel was also applied to increase penetration.
Laser treatment using two different types of devices was also employed: A 1,927-nm thulium-fractionated erbium glass laser and a 650-microsecond 1,064-nm Nd:YAG laser. The excellent resolution of the hyperpigmentation demonstrates that lasers are effective in dark skin when used appropriately, she noted.
Dr. Burgess emphasized that tailored therapy is not just relevant to Black patients. She cited data indicating that the proportion of multiracial individuals in the United States is increasing, and when tailoring cosmetic procedures, she recommended considering skin characteristics, not just skin color.
Relative to white skin, pigmented skin typically has greater elasticity, greater amounts of collagen, and greater oil content. Importantly, darker skin has a greater propensity to darkening as a result of injury, she said.
In a review of the hyperpigmentation process that follows injury or other insults, Dr. Burgess reported that only three occur inside the melanocyte. There are now topically applied agents to intervene at many of these steps, including hydroquinone to reduce up-regulation of tyrosinase enzymes, and cysteamine to inhibit conversion of DOPA to dopaquinone. All of these, often used in combination, offer potential benefit in skin of color.
However, “you must understand skin of color,” Dr. Burgess emphasized. For example, most hyaluronic acid dermal fillers can be considered in patients with Fitzpatrick skin types IV or higher with low risks for hypo- or hyperpigmentation, scarring, or keloid formation, but technique is important.
“There is more postinflammatory hyperpigmentation with serial or multiple puncture injection techniques” in dark skin relative to lighter skin, according to Dr. Burgess. She recommended reducing this risk with relatively slow injection times.
When in doubt about the result with any cosmetic procedure, test spots are a reasonable strategy, Dr. Burgess said. When there is concern about risk for adverse events, she recommended using low doses and longer intervals between treatments than might otherwise be considered. Patients should participate in understanding the rationale for selecting one approach over another.
It helps for patients to know that “the desired outcome may take many more sessions than what they read about in that we might have to consider conservative measures in order to ensure that we accomplish the cosmetic effect than they want,” she said.
It is critical that clinicians who perform laser or other cosmetic procedures on darker skin be aware of these precautions, agreed Eliot F. Battle Jr., MD, CEO and cofounder of Cultura Dermatology and Laser Center, Washington, D.C. “Over the past 20 years, we have improved lasers that can safely and effectively treat patients with skin of color, but we still have a way to go,” he said at the meeting. Darker skin behaves differently in response to this energy.
“The pigment in the skin of patients of color competes for the laser light, which can cause heat-related side effects, like blistering and pigmentary changes. Skin of color also has an increased incidence of scarring and unwanted pigmentary changes from laser treatments that create irritation and inflammation,” he explained.
It is important to be aware of these differences, but practitioners also “need to treat conservatively to minimize these unwanted side effects,” Dr. Battle said.
Dr. Burgess reported financial relationships with Allergan, Merz Aesthetics, Revance Therapeutics, and Galderma. Dr. Battle had no commercial disclosures.
Based on the fact that hyperpigmentation and other adverse events associated with cosmetic dermatologic procedures are relevant to skin type, not racial identification, individualized strategies to minimize the risk of potential adverse events are always appropriate, according to an expert speaking at the virtual Skin of Color Update 2020.
There are many highly effective interventions that employ lasers, chemical peels, and topical agents to achieve excellent cosmetic results in darker skin, but results are highly dependent on first understanding the relative risks and treatment goals, Cheryl Burgess, MD, president and founder of the Center for Dermatology and Dermatologic Surgery, Washington, D.C., said at the meeting.
She stressed the importance of educating patients that “all cosmetic procedures are not for skin of color.” Her approach is to engage patients on what they are trying to accomplish and then seeking a solution that tailors treatment to skin type based on the Fitzpatrick scale, the Roberts Hyperpigmentation Scale, or other guidance.
“There are so many different methods that we can use, and these are not necessarily the ones that patients have read about in a magazine,” Dr. Burgess said.
Intense pulsed laser (IPL) for hair removal is an example. This technique is not appropriate in patients with Fitzpatrick skin type IV or higher, according to Dr. Burgess, who presented a case example of a bad outcome. In this case, a patient came to her for treatment after exposure to IPL resulted in first- and second-degree burns complicated by extensive hypopigmentation.
Ultimately, the solution in this case involved more laser therapy, but this time a strategy was selected appropriate to skin of color.
“It is hard to suggest to a patient that we are going to use a laser device” when the problem was caused by a laser, Dr. Burgess observed, but properly selected lasers are effective and should be considered in patients with dark skin.
In this case, triple cream containing 6% hydroquinone was the first step towards resolving the hyperpigmentation. Jessner’s peel was also applied to increase penetration.
Laser treatment using two different types of devices was also employed: A 1,927-nm thulium-fractionated erbium glass laser and a 650-microsecond 1,064-nm Nd:YAG laser. The excellent resolution of the hyperpigmentation demonstrates that lasers are effective in dark skin when used appropriately, she noted.
Dr. Burgess emphasized that tailored therapy is not just relevant to Black patients. She cited data indicating that the proportion of multiracial individuals in the United States is increasing, and when tailoring cosmetic procedures, she recommended considering skin characteristics, not just skin color.
Relative to white skin, pigmented skin typically has greater elasticity, greater amounts of collagen, and greater oil content. Importantly, darker skin has a greater propensity to darkening as a result of injury, she said.
In a review of the hyperpigmentation process that follows injury or other insults, Dr. Burgess reported that only three occur inside the melanocyte. There are now topically applied agents to intervene at many of these steps, including hydroquinone to reduce up-regulation of tyrosinase enzymes, and cysteamine to inhibit conversion of DOPA to dopaquinone. All of these, often used in combination, offer potential benefit in skin of color.
However, “you must understand skin of color,” Dr. Burgess emphasized. For example, most hyaluronic acid dermal fillers can be considered in patients with Fitzpatrick skin types IV or higher with low risks for hypo- or hyperpigmentation, scarring, or keloid formation, but technique is important.
“There is more postinflammatory hyperpigmentation with serial or multiple puncture injection techniques” in dark skin relative to lighter skin, according to Dr. Burgess. She recommended reducing this risk with relatively slow injection times.
When in doubt about the result with any cosmetic procedure, test spots are a reasonable strategy, Dr. Burgess said. When there is concern about risk for adverse events, she recommended using low doses and longer intervals between treatments than might otherwise be considered. Patients should participate in understanding the rationale for selecting one approach over another.
It helps for patients to know that “the desired outcome may take many more sessions than what they read about in that we might have to consider conservative measures in order to ensure that we accomplish the cosmetic effect than they want,” she said.
It is critical that clinicians who perform laser or other cosmetic procedures on darker skin be aware of these precautions, agreed Eliot F. Battle Jr., MD, CEO and cofounder of Cultura Dermatology and Laser Center, Washington, D.C. “Over the past 20 years, we have improved lasers that can safely and effectively treat patients with skin of color, but we still have a way to go,” he said at the meeting. Darker skin behaves differently in response to this energy.
“The pigment in the skin of patients of color competes for the laser light, which can cause heat-related side effects, like blistering and pigmentary changes. Skin of color also has an increased incidence of scarring and unwanted pigmentary changes from laser treatments that create irritation and inflammation,” he explained.
It is important to be aware of these differences, but practitioners also “need to treat conservatively to minimize these unwanted side effects,” Dr. Battle said.
Dr. Burgess reported financial relationships with Allergan, Merz Aesthetics, Revance Therapeutics, and Galderma. Dr. Battle had no commercial disclosures.
Based on the fact that hyperpigmentation and other adverse events associated with cosmetic dermatologic procedures are relevant to skin type, not racial identification, individualized strategies to minimize the risk of potential adverse events are always appropriate, according to an expert speaking at the virtual Skin of Color Update 2020.
There are many highly effective interventions that employ lasers, chemical peels, and topical agents to achieve excellent cosmetic results in darker skin, but results are highly dependent on first understanding the relative risks and treatment goals, Cheryl Burgess, MD, president and founder of the Center for Dermatology and Dermatologic Surgery, Washington, D.C., said at the meeting.
She stressed the importance of educating patients that “all cosmetic procedures are not for skin of color.” Her approach is to engage patients on what they are trying to accomplish and then seeking a solution that tailors treatment to skin type based on the Fitzpatrick scale, the Roberts Hyperpigmentation Scale, or other guidance.
“There are so many different methods that we can use, and these are not necessarily the ones that patients have read about in a magazine,” Dr. Burgess said.
Intense pulsed laser (IPL) for hair removal is an example. This technique is not appropriate in patients with Fitzpatrick skin type IV or higher, according to Dr. Burgess, who presented a case example of a bad outcome. In this case, a patient came to her for treatment after exposure to IPL resulted in first- and second-degree burns complicated by extensive hypopigmentation.
Ultimately, the solution in this case involved more laser therapy, but this time a strategy was selected appropriate to skin of color.
“It is hard to suggest to a patient that we are going to use a laser device” when the problem was caused by a laser, Dr. Burgess observed, but properly selected lasers are effective and should be considered in patients with dark skin.
In this case, triple cream containing 6% hydroquinone was the first step towards resolving the hyperpigmentation. Jessner’s peel was also applied to increase penetration.
Laser treatment using two different types of devices was also employed: A 1,927-nm thulium-fractionated erbium glass laser and a 650-microsecond 1,064-nm Nd:YAG laser. The excellent resolution of the hyperpigmentation demonstrates that lasers are effective in dark skin when used appropriately, she noted.
Dr. Burgess emphasized that tailored therapy is not just relevant to Black patients. She cited data indicating that the proportion of multiracial individuals in the United States is increasing, and when tailoring cosmetic procedures, she recommended considering skin characteristics, not just skin color.
Relative to white skin, pigmented skin typically has greater elasticity, greater amounts of collagen, and greater oil content. Importantly, darker skin has a greater propensity to darkening as a result of injury, she said.
In a review of the hyperpigmentation process that follows injury or other insults, Dr. Burgess reported that only three occur inside the melanocyte. There are now topically applied agents to intervene at many of these steps, including hydroquinone to reduce up-regulation of tyrosinase enzymes, and cysteamine to inhibit conversion of DOPA to dopaquinone. All of these, often used in combination, offer potential benefit in skin of color.
However, “you must understand skin of color,” Dr. Burgess emphasized. For example, most hyaluronic acid dermal fillers can be considered in patients with Fitzpatrick skin types IV or higher with low risks for hypo- or hyperpigmentation, scarring, or keloid formation, but technique is important.
“There is more postinflammatory hyperpigmentation with serial or multiple puncture injection techniques” in dark skin relative to lighter skin, according to Dr. Burgess. She recommended reducing this risk with relatively slow injection times.
When in doubt about the result with any cosmetic procedure, test spots are a reasonable strategy, Dr. Burgess said. When there is concern about risk for adverse events, she recommended using low doses and longer intervals between treatments than might otherwise be considered. Patients should participate in understanding the rationale for selecting one approach over another.
It helps for patients to know that “the desired outcome may take many more sessions than what they read about in that we might have to consider conservative measures in order to ensure that we accomplish the cosmetic effect than they want,” she said.
It is critical that clinicians who perform laser or other cosmetic procedures on darker skin be aware of these precautions, agreed Eliot F. Battle Jr., MD, CEO and cofounder of Cultura Dermatology and Laser Center, Washington, D.C. “Over the past 20 years, we have improved lasers that can safely and effectively treat patients with skin of color, but we still have a way to go,” he said at the meeting. Darker skin behaves differently in response to this energy.
“The pigment in the skin of patients of color competes for the laser light, which can cause heat-related side effects, like blistering and pigmentary changes. Skin of color also has an increased incidence of scarring and unwanted pigmentary changes from laser treatments that create irritation and inflammation,” he explained.
It is important to be aware of these differences, but practitioners also “need to treat conservatively to minimize these unwanted side effects,” Dr. Battle said.
Dr. Burgess reported financial relationships with Allergan, Merz Aesthetics, Revance Therapeutics, and Galderma. Dr. Battle had no commercial disclosures.
FROM SOC 2020