User login
Coagulopathy outbreak underscores danger of synthetic cannabinoids
Synthetic cannabinoids laced with superwarfarin were behind a recent outbreak of severe coagulopathy in Illinois.
In most cases, vitamin K replacement therapy alleviated symptoms, but four patients died after developing intracranial bleeding, said Amar H. Kelkar, MD, of the University of Illinois at Peoria.
Experts continue to look for how and why superwarfarin ended up in synthetic cannabinoids, whose street names include spice and K2, wrote Dr. Kelkar and his associates. Their report is in the New England Journal of Medicine.
Starting in March 2018, more than 150 patients across Illinois presented to hospitals with bleeding diathesis that involved persistent coagulopathy, the investigators explained. Early inquiries revealed that patients had consumed synthetic cannabinoids. Serum tests identified vitamin K antagonists, including brodifacoum, bromadiolone, and difenacoum. During arrests of suspected distributors, police confiscated synthetic cannabinoids that also tested positive for brodifacoum.
To help characterize the outbreak, the investigators reviewed admissions to Saint Francis Medical Center in Peoria, Ill., between March 28 and April 21, 2018. They identified 34 cases in which patients with vitamin K–dependent factor coagulopathy reported recent exposure to synthetic cannabinoids.
Fifteen of these patients underwent confirmatory anticoagulant testing, which universally confirmed superwarfarin poisoning. Brodifacoum was detected in all patients, difenacoum in five patients, bromadiolone in two patients, and warfarin in one patient.
Common presenting symptoms included gross hematuria (56% of patients) and abdominal pain (47%). Computed tomography identified renal abnormalities in 12 patients.
All patients received oral vitamin K1 (phytonadione). Red cell transfusions, fresh-frozen plasma infusions, and 4-factor prothrombin complex concentrate, or a combination of these treatments, were also used in some patients.
Among the four confirmed deaths in this outbreak, one occurred in a patient in this case series. The patient, a 37-year-old woman presenting to the emergency department with markedly reduced consciousness, was reported by her friends to have recently used synthetic cannabinoids and methamphetamine. She had no personal or family history of coagulopathy.
Computed tomography of the head without contrast material revealed severe acute intraparenchymal hemorrhage of the right basal nuclei and insula with intraventricular extension, a 10-mm left-sided midline shift, and herniation.
She met criteria for brain death 15 hours after hospital admission despite treatment with 10 mg of intravenous vitamin K1, four units of fresh frozen plasma, and 2,300 units of Kcentra.
Treating these patients after hospital discharge was difficult because of a lack of consensus guidelines and access to follow-up care, Dr. Kelkar and his associates noted. Some patients were quoted $24,000 to $34,000 per month for oral vitamin K1 therapy, which also made caring for them difficult and highlighted the need for confirmatory laboratory testing of suspected cases of superwarfarin poisoning.
Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
SOURCE: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
Treating patients who are exposed to synthetic cannabinoid and a superwarfarin such as brodifacoum “requires more than the usual ‘treat ’em and street ’em’ approach,” wrote Jean M. Connors, MD.
“Brodifacoum is a successful rodenticide because of its extremely long half-life (approximately 16-36 days in humans),” Dr. Connors noted.
The drug also is lipophilic, causing tissue sequestration. Once exposed, patients often develop coagulopathy lasting 9 months or longer, she said.
Compared with warfarin poisoning, brodifacoum therefore requires substantially higher-dose and longer-term vitamin K1 therapy. Among the patients in this case series, the maximum outpatient dose was 50 mg, three times daily, and one patient was prescribed 25 mg, twice daily for 270 days, Dr. Connors noted.
“[Dr. Kelkar and his associates] highlight the resources and coordination needed for dealing with a public health crisis that has a prolonged duration of effect,” she added. “Because the synthetic cannabinoid market is lucrative, new products with new toxicity profiles are likely to crop up.”
Dr. Connors is with Brigham and Women’s Hospital, Dana-Farber Cancer Institute, and Harvard Medical School, all in Boston. She reported personal fees from Bristol-Myers Squibb, Portola, Dova Pharmaceuticals, and Unum Therapeutics outside the submitted work. These comments are from her accompanying editorial (N Engl J Med. 2018;379:1275-7).
Treating patients who are exposed to synthetic cannabinoid and a superwarfarin such as brodifacoum “requires more than the usual ‘treat ’em and street ’em’ approach,” wrote Jean M. Connors, MD.
“Brodifacoum is a successful rodenticide because of its extremely long half-life (approximately 16-36 days in humans),” Dr. Connors noted.
The drug also is lipophilic, causing tissue sequestration. Once exposed, patients often develop coagulopathy lasting 9 months or longer, she said.
Compared with warfarin poisoning, brodifacoum therefore requires substantially higher-dose and longer-term vitamin K1 therapy. Among the patients in this case series, the maximum outpatient dose was 50 mg, three times daily, and one patient was prescribed 25 mg, twice daily for 270 days, Dr. Connors noted.
“[Dr. Kelkar and his associates] highlight the resources and coordination needed for dealing with a public health crisis that has a prolonged duration of effect,” she added. “Because the synthetic cannabinoid market is lucrative, new products with new toxicity profiles are likely to crop up.”
Dr. Connors is with Brigham and Women’s Hospital, Dana-Farber Cancer Institute, and Harvard Medical School, all in Boston. She reported personal fees from Bristol-Myers Squibb, Portola, Dova Pharmaceuticals, and Unum Therapeutics outside the submitted work. These comments are from her accompanying editorial (N Engl J Med. 2018;379:1275-7).
Treating patients who are exposed to synthetic cannabinoid and a superwarfarin such as brodifacoum “requires more than the usual ‘treat ’em and street ’em’ approach,” wrote Jean M. Connors, MD.
“Brodifacoum is a successful rodenticide because of its extremely long half-life (approximately 16-36 days in humans),” Dr. Connors noted.
The drug also is lipophilic, causing tissue sequestration. Once exposed, patients often develop coagulopathy lasting 9 months or longer, she said.
Compared with warfarin poisoning, brodifacoum therefore requires substantially higher-dose and longer-term vitamin K1 therapy. Among the patients in this case series, the maximum outpatient dose was 50 mg, three times daily, and one patient was prescribed 25 mg, twice daily for 270 days, Dr. Connors noted.
“[Dr. Kelkar and his associates] highlight the resources and coordination needed for dealing with a public health crisis that has a prolonged duration of effect,” she added. “Because the synthetic cannabinoid market is lucrative, new products with new toxicity profiles are likely to crop up.”
Dr. Connors is with Brigham and Women’s Hospital, Dana-Farber Cancer Institute, and Harvard Medical School, all in Boston. She reported personal fees from Bristol-Myers Squibb, Portola, Dova Pharmaceuticals, and Unum Therapeutics outside the submitted work. These comments are from her accompanying editorial (N Engl J Med. 2018;379:1275-7).
Synthetic cannabinoids laced with superwarfarin were behind a recent outbreak of severe coagulopathy in Illinois.
In most cases, vitamin K replacement therapy alleviated symptoms, but four patients died after developing intracranial bleeding, said Amar H. Kelkar, MD, of the University of Illinois at Peoria.
Experts continue to look for how and why superwarfarin ended up in synthetic cannabinoids, whose street names include spice and K2, wrote Dr. Kelkar and his associates. Their report is in the New England Journal of Medicine.
Starting in March 2018, more than 150 patients across Illinois presented to hospitals with bleeding diathesis that involved persistent coagulopathy, the investigators explained. Early inquiries revealed that patients had consumed synthetic cannabinoids. Serum tests identified vitamin K antagonists, including brodifacoum, bromadiolone, and difenacoum. During arrests of suspected distributors, police confiscated synthetic cannabinoids that also tested positive for brodifacoum.
To help characterize the outbreak, the investigators reviewed admissions to Saint Francis Medical Center in Peoria, Ill., between March 28 and April 21, 2018. They identified 34 cases in which patients with vitamin K–dependent factor coagulopathy reported recent exposure to synthetic cannabinoids.
Fifteen of these patients underwent confirmatory anticoagulant testing, which universally confirmed superwarfarin poisoning. Brodifacoum was detected in all patients, difenacoum in five patients, bromadiolone in two patients, and warfarin in one patient.
Common presenting symptoms included gross hematuria (56% of patients) and abdominal pain (47%). Computed tomography identified renal abnormalities in 12 patients.
All patients received oral vitamin K1 (phytonadione). Red cell transfusions, fresh-frozen plasma infusions, and 4-factor prothrombin complex concentrate, or a combination of these treatments, were also used in some patients.
Among the four confirmed deaths in this outbreak, one occurred in a patient in this case series. The patient, a 37-year-old woman presenting to the emergency department with markedly reduced consciousness, was reported by her friends to have recently used synthetic cannabinoids and methamphetamine. She had no personal or family history of coagulopathy.
Computed tomography of the head without contrast material revealed severe acute intraparenchymal hemorrhage of the right basal nuclei and insula with intraventricular extension, a 10-mm left-sided midline shift, and herniation.
She met criteria for brain death 15 hours after hospital admission despite treatment with 10 mg of intravenous vitamin K1, four units of fresh frozen plasma, and 2,300 units of Kcentra.
Treating these patients after hospital discharge was difficult because of a lack of consensus guidelines and access to follow-up care, Dr. Kelkar and his associates noted. Some patients were quoted $24,000 to $34,000 per month for oral vitamin K1 therapy, which also made caring for them difficult and highlighted the need for confirmatory laboratory testing of suspected cases of superwarfarin poisoning.
Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
SOURCE: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
Synthetic cannabinoids laced with superwarfarin were behind a recent outbreak of severe coagulopathy in Illinois.
In most cases, vitamin K replacement therapy alleviated symptoms, but four patients died after developing intracranial bleeding, said Amar H. Kelkar, MD, of the University of Illinois at Peoria.
Experts continue to look for how and why superwarfarin ended up in synthetic cannabinoids, whose street names include spice and K2, wrote Dr. Kelkar and his associates. Their report is in the New England Journal of Medicine.
Starting in March 2018, more than 150 patients across Illinois presented to hospitals with bleeding diathesis that involved persistent coagulopathy, the investigators explained. Early inquiries revealed that patients had consumed synthetic cannabinoids. Serum tests identified vitamin K antagonists, including brodifacoum, bromadiolone, and difenacoum. During arrests of suspected distributors, police confiscated synthetic cannabinoids that also tested positive for brodifacoum.
To help characterize the outbreak, the investigators reviewed admissions to Saint Francis Medical Center in Peoria, Ill., between March 28 and April 21, 2018. They identified 34 cases in which patients with vitamin K–dependent factor coagulopathy reported recent exposure to synthetic cannabinoids.
Fifteen of these patients underwent confirmatory anticoagulant testing, which universally confirmed superwarfarin poisoning. Brodifacoum was detected in all patients, difenacoum in five patients, bromadiolone in two patients, and warfarin in one patient.
Common presenting symptoms included gross hematuria (56% of patients) and abdominal pain (47%). Computed tomography identified renal abnormalities in 12 patients.
All patients received oral vitamin K1 (phytonadione). Red cell transfusions, fresh-frozen plasma infusions, and 4-factor prothrombin complex concentrate, or a combination of these treatments, were also used in some patients.
Among the four confirmed deaths in this outbreak, one occurred in a patient in this case series. The patient, a 37-year-old woman presenting to the emergency department with markedly reduced consciousness, was reported by her friends to have recently used synthetic cannabinoids and methamphetamine. She had no personal or family history of coagulopathy.
Computed tomography of the head without contrast material revealed severe acute intraparenchymal hemorrhage of the right basal nuclei and insula with intraventricular extension, a 10-mm left-sided midline shift, and herniation.
She met criteria for brain death 15 hours after hospital admission despite treatment with 10 mg of intravenous vitamin K1, four units of fresh frozen plasma, and 2,300 units of Kcentra.
Treating these patients after hospital discharge was difficult because of a lack of consensus guidelines and access to follow-up care, Dr. Kelkar and his associates noted. Some patients were quoted $24,000 to $34,000 per month for oral vitamin K1 therapy, which also made caring for them difficult and highlighted the need for confirmatory laboratory testing of suspected cases of superwarfarin poisoning.
Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
SOURCE: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: There were more than 150 cases in Illinois with four deaths among patients who developed spontaneous intracranial bleeding.
Study details: A single-institution case series of 15 patients.
Disclosures: Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
Source: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
Atrial fib guidelines may fall short on oral anticoagulation
Anticoagulation thresholds based on CHA2DS2-VASc risk score varied from population to population, researchers reported in the Annals of Internal Medicine.
After accounting for differing rates of stroke in published studies, the benefit of warfarin anticoagulation varied nearly fourfold, said Sachin J. Shah, MD, of the University of California San Francisco and his associates. They called for guidelines that “better reflect the uncertainty in current thresholds of stroke risk score for recommending anticoagulation.”
Oral anticoagulation markedly reduces risk of ischemic stroke in patients with atrial fibrillation but increases the risk of major bleeding, including intracranial hemorrhage, which often is fatal. Therefore, when deciding whether to recommend oral anticoagulation, physicians must estimate clinical net benefit by quantifying the difference between reduction in stroke risk and increase in major bleeding risk, weighted by the severity of each outcome.
Guidelines on nonvalvular atrial fibrillation from the European Society of Cardiology and joint guidelines from the American Heart Association, American College of Cardiology, and Heart Rhythm Society (AHA/ACC/HRS) recommend oral anticoagulation when CHA2DS2-VASc (congestive heart failure, hypertension, age, diabetes, stroke, and vascular disease) risk score is 2 or greater. These guidelines implicitly assume that a particular CHA2DS2-VASc score denotes the same amount of risk across populations, even though a recent meta-analysis found otherwise, as the researchers noted.
To further test this assumption, they applied an existing Markov model to data from more than 33,000 members of the ATRIA-CVRN cohort. All patients had nonvalvular atrial fibrillation, were members of Kaiser Permanente Northern California, and were diagnosed during 1996-1997. About 81% had a CHA2DS2-VASc score of at least 2. For each patient, the researchers produced four estimates of the net clinical benefit of oral anticoagulation based on ischemic stroke rates from ATRIA, the Swedish AF cohort study, the SPORTIF study, and the Danish National Patient Registry.
Optimal anticoagulation thresholds were a CHA2DS2-VASc score of 3 or more using stroke rates from ATRIA, 2 or more based on Swedish AF rates, 1 or more based on SPORTIF rates, and 0 or more using rates from the Danish National Patient Registry. Oral anticoagulation thresholds were lower but still varied widely after accounting for the lower rates of intracranial hemorrhage associated with non–vitamin K antagonist therapy.
Therefore, current guidelines based on CHA2DS2-VASc score may need revising “in favor of more accurate, individualized assessments of risk for both ischemic stroke and major bleeding,” the investigators wrote. “Until such time, guidelines should better reflect the uncertainty of the current approach in which a patient’s CHA2DS2-VASc score is used as the primary basis for recommending oral anticoagulation.”
The study had no primary funding source. Dr. Shah reported having no conflicts of interest. Three coinvestigators disclosed research support from relevant pharmaceutical or device companies.
SOURCE: Shah SJ et al. Ann Intern Med. 2018 Sep 25. doi: 10.7326/M17-2762
Based on this study, the CHA2DS2-VASc score threshold for anticoagulation might not be a “one-size-fits all approach but rather a starting point for a more tailored assessment,” wrote Jennifer M. Wright, MD, and Craig T. January, MD, PhD, in an editorial accompanying the report.
The CHA2DS2-VASc algorithm uses fixed whole integers and therefore might lack the sensitivity and flexibility needed to accurately reflect the effects of its components, the experts wrote. “For example, female sex now seems to be a risk modifier, and its intensity depends on other risk factors.”
However, CHA2DS2-VASc remains the main way to assess net clinical benefit of oral anticoagulation for patients with anticoagulation, they conceded. “When it comes to the conversation about the risks and benefits of anticoagulation for our patients with atrial fibrillation, we must remember that each patient is an individual and has his or her own ‘score.’ ”
The editorialists are with the University of Wisconsin in Madison. They reported having no relevant conflicts of interest. These comments are based on their editorial (Ann Intern Med. 2018 Sep 25. doi: 10.7326/M18-2355).
Based on this study, the CHA2DS2-VASc score threshold for anticoagulation might not be a “one-size-fits all approach but rather a starting point for a more tailored assessment,” wrote Jennifer M. Wright, MD, and Craig T. January, MD, PhD, in an editorial accompanying the report.
The CHA2DS2-VASc algorithm uses fixed whole integers and therefore might lack the sensitivity and flexibility needed to accurately reflect the effects of its components, the experts wrote. “For example, female sex now seems to be a risk modifier, and its intensity depends on other risk factors.”
However, CHA2DS2-VASc remains the main way to assess net clinical benefit of oral anticoagulation for patients with anticoagulation, they conceded. “When it comes to the conversation about the risks and benefits of anticoagulation for our patients with atrial fibrillation, we must remember that each patient is an individual and has his or her own ‘score.’ ”
The editorialists are with the University of Wisconsin in Madison. They reported having no relevant conflicts of interest. These comments are based on their editorial (Ann Intern Med. 2018 Sep 25. doi: 10.7326/M18-2355).
Based on this study, the CHA2DS2-VASc score threshold for anticoagulation might not be a “one-size-fits all approach but rather a starting point for a more tailored assessment,” wrote Jennifer M. Wright, MD, and Craig T. January, MD, PhD, in an editorial accompanying the report.
The CHA2DS2-VASc algorithm uses fixed whole integers and therefore might lack the sensitivity and flexibility needed to accurately reflect the effects of its components, the experts wrote. “For example, female sex now seems to be a risk modifier, and its intensity depends on other risk factors.”
However, CHA2DS2-VASc remains the main way to assess net clinical benefit of oral anticoagulation for patients with anticoagulation, they conceded. “When it comes to the conversation about the risks and benefits of anticoagulation for our patients with atrial fibrillation, we must remember that each patient is an individual and has his or her own ‘score.’ ”
The editorialists are with the University of Wisconsin in Madison. They reported having no relevant conflicts of interest. These comments are based on their editorial (Ann Intern Med. 2018 Sep 25. doi: 10.7326/M18-2355).
Anticoagulation thresholds based on CHA2DS2-VASc risk score varied from population to population, researchers reported in the Annals of Internal Medicine.
After accounting for differing rates of stroke in published studies, the benefit of warfarin anticoagulation varied nearly fourfold, said Sachin J. Shah, MD, of the University of California San Francisco and his associates. They called for guidelines that “better reflect the uncertainty in current thresholds of stroke risk score for recommending anticoagulation.”
Oral anticoagulation markedly reduces risk of ischemic stroke in patients with atrial fibrillation but increases the risk of major bleeding, including intracranial hemorrhage, which often is fatal. Therefore, when deciding whether to recommend oral anticoagulation, physicians must estimate clinical net benefit by quantifying the difference between reduction in stroke risk and increase in major bleeding risk, weighted by the severity of each outcome.
Guidelines on nonvalvular atrial fibrillation from the European Society of Cardiology and joint guidelines from the American Heart Association, American College of Cardiology, and Heart Rhythm Society (AHA/ACC/HRS) recommend oral anticoagulation when CHA2DS2-VASc (congestive heart failure, hypertension, age, diabetes, stroke, and vascular disease) risk score is 2 or greater. These guidelines implicitly assume that a particular CHA2DS2-VASc score denotes the same amount of risk across populations, even though a recent meta-analysis found otherwise, as the researchers noted.
To further test this assumption, they applied an existing Markov model to data from more than 33,000 members of the ATRIA-CVRN cohort. All patients had nonvalvular atrial fibrillation, were members of Kaiser Permanente Northern California, and were diagnosed during 1996-1997. About 81% had a CHA2DS2-VASc score of at least 2. For each patient, the researchers produced four estimates of the net clinical benefit of oral anticoagulation based on ischemic stroke rates from ATRIA, the Swedish AF cohort study, the SPORTIF study, and the Danish National Patient Registry.
Optimal anticoagulation thresholds were a CHA2DS2-VASc score of 3 or more using stroke rates from ATRIA, 2 or more based on Swedish AF rates, 1 or more based on SPORTIF rates, and 0 or more using rates from the Danish National Patient Registry. Oral anticoagulation thresholds were lower but still varied widely after accounting for the lower rates of intracranial hemorrhage associated with non–vitamin K antagonist therapy.
Therefore, current guidelines based on CHA2DS2-VASc score may need revising “in favor of more accurate, individualized assessments of risk for both ischemic stroke and major bleeding,” the investigators wrote. “Until such time, guidelines should better reflect the uncertainty of the current approach in which a patient’s CHA2DS2-VASc score is used as the primary basis for recommending oral anticoagulation.”
The study had no primary funding source. Dr. Shah reported having no conflicts of interest. Three coinvestigators disclosed research support from relevant pharmaceutical or device companies.
SOURCE: Shah SJ et al. Ann Intern Med. 2018 Sep 25. doi: 10.7326/M17-2762
Anticoagulation thresholds based on CHA2DS2-VASc risk score varied from population to population, researchers reported in the Annals of Internal Medicine.
After accounting for differing rates of stroke in published studies, the benefit of warfarin anticoagulation varied nearly fourfold, said Sachin J. Shah, MD, of the University of California San Francisco and his associates. They called for guidelines that “better reflect the uncertainty in current thresholds of stroke risk score for recommending anticoagulation.”
Oral anticoagulation markedly reduces risk of ischemic stroke in patients with atrial fibrillation but increases the risk of major bleeding, including intracranial hemorrhage, which often is fatal. Therefore, when deciding whether to recommend oral anticoagulation, physicians must estimate clinical net benefit by quantifying the difference between reduction in stroke risk and increase in major bleeding risk, weighted by the severity of each outcome.
Guidelines on nonvalvular atrial fibrillation from the European Society of Cardiology and joint guidelines from the American Heart Association, American College of Cardiology, and Heart Rhythm Society (AHA/ACC/HRS) recommend oral anticoagulation when CHA2DS2-VASc (congestive heart failure, hypertension, age, diabetes, stroke, and vascular disease) risk score is 2 or greater. These guidelines implicitly assume that a particular CHA2DS2-VASc score denotes the same amount of risk across populations, even though a recent meta-analysis found otherwise, as the researchers noted.
To further test this assumption, they applied an existing Markov model to data from more than 33,000 members of the ATRIA-CVRN cohort. All patients had nonvalvular atrial fibrillation, were members of Kaiser Permanente Northern California, and were diagnosed during 1996-1997. About 81% had a CHA2DS2-VASc score of at least 2. For each patient, the researchers produced four estimates of the net clinical benefit of oral anticoagulation based on ischemic stroke rates from ATRIA, the Swedish AF cohort study, the SPORTIF study, and the Danish National Patient Registry.
Optimal anticoagulation thresholds were a CHA2DS2-VASc score of 3 or more using stroke rates from ATRIA, 2 or more based on Swedish AF rates, 1 or more based on SPORTIF rates, and 0 or more using rates from the Danish National Patient Registry. Oral anticoagulation thresholds were lower but still varied widely after accounting for the lower rates of intracranial hemorrhage associated with non–vitamin K antagonist therapy.
Therefore, current guidelines based on CHA2DS2-VASc score may need revising “in favor of more accurate, individualized assessments of risk for both ischemic stroke and major bleeding,” the investigators wrote. “Until such time, guidelines should better reflect the uncertainty of the current approach in which a patient’s CHA2DS2-VASc score is used as the primary basis for recommending oral anticoagulation.”
The study had no primary funding source. Dr. Shah reported having no conflicts of interest. Three coinvestigators disclosed research support from relevant pharmaceutical or device companies.
SOURCE: Shah SJ et al. Ann Intern Med. 2018 Sep 25. doi: 10.7326/M17-2762
FROM THE ANNALS OF INTERNAL MEDICINE
Key clinical point:
Major finding: After accounting for differing rates of stroke in published studies, the benefit of warfarin anticoagulation varied nearly fourfold. Anticoagulation thresholds were lower but still varied widely in a model of non–vitamin K antagonist therapy.
Study details: Markov state-transition model of 33,434 patients with nonvalvular atrial fibrillation.
Disclosures: The study had no primary funding source. Dr. Shah reported having no conflicts of interest. Three coinvestigators disclosed research support from relevant pharmaceutical or device companies.
Source: Shah SJ et al. Ann Intern Med. 2018 Sep 25. doi: 10.7326/M17-2762.
Five-year follow-up confirms safety of antibiotics for uncomplicated appendicitis
Longer-term outcomes of treating uncomplicated acute appendicitis with antibiotics suggest it is a feasible alternative to appendectomy.
Researchers have presented the 5-year follow-up data from the Appendicitis Acuta (APPAC) multicenter randomized clinical trial comparing appendectomy with antibiotic therapy in 530 patients.
They found that 39.1% (100) of the 257 patients randomized to antibiotic therapy – 3 days of intravenous ertapenem followed by 7 days of oral levofloxacin and metronidazole – experienced a recurrence of appendicitis within 5 years and subsequently had surgery.
However the authors noted that seven of these patients were later found not to have appendicitis, so the true success rate for antibiotic treatment was actually 63.7%.
Seventy of the patients who experienced a recurrence underwent surgery in the first year after randomization, 17 in the second year, 3 in the third year, 5 in the fourth year, and the remaining 5 patients in the fifth year, the authors wrote in an article published in JAMA.
However the overall complication rate was similar in patients who were randomized to undergo appendectomy and in those who were initially randomized to the antibiotic group but later experienced a recurrence and underwent surgery.
“No patient initially treated with antibiotics, who ultimately developed recurrent appendicitis, had any complications related to the delay in surgery,” wrote Paulina Salminen, MD, from Turku (Finland) University Hospital and coauthors. “Nearly two-thirds of all patients who initially presented with uncomplicated appendicitis were successfully treated with antibiotics alone, and those who ultimately developed recurrent disease did not experience any adverse outcomes related to the delay in appendectomy.”
Of the 100 patients randomized to antibiotics who underwent appendectomy after a recurrence, 15 were operated on when they were first hospitalized at study admission.
The authors commented that the study design allowed for surgeons to exercise their clinical judgment in choosing when to perform an appendectomy on patients in the antibiotic group, because antibiotics alone was not considered acceptable treatment for appendicitis.
“This led to some patients undergoing appendectomy who did not have appendicitis or who might have been successfully treated with antibiotics or an another course of antibiotics,” they wrote. “Future studies should investigate protocols for further imaging or antibiotic treatment for patients who develop recurrent appendicitis after they were initially treated with antibiotics.”
In the recurrence group, the majority were found to have uncomplicated appendicitis, but complicated appendicitis was seen in two patients between 2 and 5 years after the index admission.
There was a significant 17.9% higher complication rate in the appendectomy group, compared with the antibiotic group – 24.4% versus 6.5% – at 5 years and two patients in the appendectomy group had severe complications requiring reoperation.
They suggested that the higher complication rate with surgery, which was mostly attributable to infections, could be reduced by the use of laparoscopic appendectomy, which is also associated with faster recovery.
The median length of hospital stay was 3 days for both the appendectomy group and the antibiotics-only group, but patients randomized to appendectomy took a median of 22 days of sick leave, compared with 11 days for those randomized to antibiotics (P less than .001).
In the absence of standard protocol on treating appendicitis with antibiotics, the authors noted that they took a conservative approach, using broad-spectrum antibiotics and keeping patients in hospital for 3 days for observation.
“The success of antibiotic treatment for appendicitis calls into question prior beliefs that appendicitis inevitably results in serious intra-abdominal infection if appendectomy is not performed.”
The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
SOURCE: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
“When the earlier results of the APPAC trial were published, showing that 73% of patients with uncomplicated acute appendicitis did not require surgery at 1 year of follow-up, critics raised concerns that many more of these patients would eventually require surgery,” wrote Edward H. Livingston, MD, deputy editor of JAMA in an accompanying editorial (JAMA 2018;320:1245-46).Surgeons have since been waiting until longer-term outcomes were known. These 5-year results are supportive of the antibiotics approach. “They show no increase in major complications in patients who experienced a recurrence and underwent appendectomy after initially being randomized to antibiotic therapy. They challenge the notion that uncomplicated acute appendicitis is a surgical emergency and show that nonsurgical treatment is a reasonable option,” Dr Livingston wrote.
He declared no conflicts of interest.
“When the earlier results of the APPAC trial were published, showing that 73% of patients with uncomplicated acute appendicitis did not require surgery at 1 year of follow-up, critics raised concerns that many more of these patients would eventually require surgery,” wrote Edward H. Livingston, MD, deputy editor of JAMA in an accompanying editorial (JAMA 2018;320:1245-46).Surgeons have since been waiting until longer-term outcomes were known. These 5-year results are supportive of the antibiotics approach. “They show no increase in major complications in patients who experienced a recurrence and underwent appendectomy after initially being randomized to antibiotic therapy. They challenge the notion that uncomplicated acute appendicitis is a surgical emergency and show that nonsurgical treatment is a reasonable option,” Dr Livingston wrote.
He declared no conflicts of interest.
“When the earlier results of the APPAC trial were published, showing that 73% of patients with uncomplicated acute appendicitis did not require surgery at 1 year of follow-up, critics raised concerns that many more of these patients would eventually require surgery,” wrote Edward H. Livingston, MD, deputy editor of JAMA in an accompanying editorial (JAMA 2018;320:1245-46).Surgeons have since been waiting until longer-term outcomes were known. These 5-year results are supportive of the antibiotics approach. “They show no increase in major complications in patients who experienced a recurrence and underwent appendectomy after initially being randomized to antibiotic therapy. They challenge the notion that uncomplicated acute appendicitis is a surgical emergency and show that nonsurgical treatment is a reasonable option,” Dr Livingston wrote.
He declared no conflicts of interest.
Longer-term outcomes of treating uncomplicated acute appendicitis with antibiotics suggest it is a feasible alternative to appendectomy.
Researchers have presented the 5-year follow-up data from the Appendicitis Acuta (APPAC) multicenter randomized clinical trial comparing appendectomy with antibiotic therapy in 530 patients.
They found that 39.1% (100) of the 257 patients randomized to antibiotic therapy – 3 days of intravenous ertapenem followed by 7 days of oral levofloxacin and metronidazole – experienced a recurrence of appendicitis within 5 years and subsequently had surgery.
However the authors noted that seven of these patients were later found not to have appendicitis, so the true success rate for antibiotic treatment was actually 63.7%.
Seventy of the patients who experienced a recurrence underwent surgery in the first year after randomization, 17 in the second year, 3 in the third year, 5 in the fourth year, and the remaining 5 patients in the fifth year, the authors wrote in an article published in JAMA.
However the overall complication rate was similar in patients who were randomized to undergo appendectomy and in those who were initially randomized to the antibiotic group but later experienced a recurrence and underwent surgery.
“No patient initially treated with antibiotics, who ultimately developed recurrent appendicitis, had any complications related to the delay in surgery,” wrote Paulina Salminen, MD, from Turku (Finland) University Hospital and coauthors. “Nearly two-thirds of all patients who initially presented with uncomplicated appendicitis were successfully treated with antibiotics alone, and those who ultimately developed recurrent disease did not experience any adverse outcomes related to the delay in appendectomy.”
Of the 100 patients randomized to antibiotics who underwent appendectomy after a recurrence, 15 were operated on when they were first hospitalized at study admission.
The authors commented that the study design allowed for surgeons to exercise their clinical judgment in choosing when to perform an appendectomy on patients in the antibiotic group, because antibiotics alone was not considered acceptable treatment for appendicitis.
“This led to some patients undergoing appendectomy who did not have appendicitis or who might have been successfully treated with antibiotics or an another course of antibiotics,” they wrote. “Future studies should investigate protocols for further imaging or antibiotic treatment for patients who develop recurrent appendicitis after they were initially treated with antibiotics.”
In the recurrence group, the majority were found to have uncomplicated appendicitis, but complicated appendicitis was seen in two patients between 2 and 5 years after the index admission.
There was a significant 17.9% higher complication rate in the appendectomy group, compared with the antibiotic group – 24.4% versus 6.5% – at 5 years and two patients in the appendectomy group had severe complications requiring reoperation.
They suggested that the higher complication rate with surgery, which was mostly attributable to infections, could be reduced by the use of laparoscopic appendectomy, which is also associated with faster recovery.
The median length of hospital stay was 3 days for both the appendectomy group and the antibiotics-only group, but patients randomized to appendectomy took a median of 22 days of sick leave, compared with 11 days for those randomized to antibiotics (P less than .001).
In the absence of standard protocol on treating appendicitis with antibiotics, the authors noted that they took a conservative approach, using broad-spectrum antibiotics and keeping patients in hospital for 3 days for observation.
“The success of antibiotic treatment for appendicitis calls into question prior beliefs that appendicitis inevitably results in serious intra-abdominal infection if appendectomy is not performed.”
The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
SOURCE: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
Longer-term outcomes of treating uncomplicated acute appendicitis with antibiotics suggest it is a feasible alternative to appendectomy.
Researchers have presented the 5-year follow-up data from the Appendicitis Acuta (APPAC) multicenter randomized clinical trial comparing appendectomy with antibiotic therapy in 530 patients.
They found that 39.1% (100) of the 257 patients randomized to antibiotic therapy – 3 days of intravenous ertapenem followed by 7 days of oral levofloxacin and metronidazole – experienced a recurrence of appendicitis within 5 years and subsequently had surgery.
However the authors noted that seven of these patients were later found not to have appendicitis, so the true success rate for antibiotic treatment was actually 63.7%.
Seventy of the patients who experienced a recurrence underwent surgery in the first year after randomization, 17 in the second year, 3 in the third year, 5 in the fourth year, and the remaining 5 patients in the fifth year, the authors wrote in an article published in JAMA.
However the overall complication rate was similar in patients who were randomized to undergo appendectomy and in those who were initially randomized to the antibiotic group but later experienced a recurrence and underwent surgery.
“No patient initially treated with antibiotics, who ultimately developed recurrent appendicitis, had any complications related to the delay in surgery,” wrote Paulina Salminen, MD, from Turku (Finland) University Hospital and coauthors. “Nearly two-thirds of all patients who initially presented with uncomplicated appendicitis were successfully treated with antibiotics alone, and those who ultimately developed recurrent disease did not experience any adverse outcomes related to the delay in appendectomy.”
Of the 100 patients randomized to antibiotics who underwent appendectomy after a recurrence, 15 were operated on when they were first hospitalized at study admission.
The authors commented that the study design allowed for surgeons to exercise their clinical judgment in choosing when to perform an appendectomy on patients in the antibiotic group, because antibiotics alone was not considered acceptable treatment for appendicitis.
“This led to some patients undergoing appendectomy who did not have appendicitis or who might have been successfully treated with antibiotics or an another course of antibiotics,” they wrote. “Future studies should investigate protocols for further imaging or antibiotic treatment for patients who develop recurrent appendicitis after they were initially treated with antibiotics.”
In the recurrence group, the majority were found to have uncomplicated appendicitis, but complicated appendicitis was seen in two patients between 2 and 5 years after the index admission.
There was a significant 17.9% higher complication rate in the appendectomy group, compared with the antibiotic group – 24.4% versus 6.5% – at 5 years and two patients in the appendectomy group had severe complications requiring reoperation.
They suggested that the higher complication rate with surgery, which was mostly attributable to infections, could be reduced by the use of laparoscopic appendectomy, which is also associated with faster recovery.
The median length of hospital stay was 3 days for both the appendectomy group and the antibiotics-only group, but patients randomized to appendectomy took a median of 22 days of sick leave, compared with 11 days for those randomized to antibiotics (P less than .001).
In the absence of standard protocol on treating appendicitis with antibiotics, the authors noted that they took a conservative approach, using broad-spectrum antibiotics and keeping patients in hospital for 3 days for observation.
“The success of antibiotic treatment for appendicitis calls into question prior beliefs that appendicitis inevitably results in serious intra-abdominal infection if appendectomy is not performed.”
The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
SOURCE: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
FROM JAMA
Key clinical point: Antibiotics could be used as an alternative to surgery for uncomplicated acute appendicitis.
Major finding: Around two-thirds of patients with acute appendicitis treated with antibiotics only did not experience a recurrence in 5 years.
Study details: Randomized controlled study in 530 patients with uncomplicated acute appendicitis.
Disclosures: The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
Source: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
Signs point to growing abuse of gabapentinoids in the U.S.
LAS VEGAS – Up to 22% of opioid abusers also abuse gabapentin, taking high doses of the antiepileptic for its psychoactive effects, or to potentiate the effect of opioids.
The drug – the 10th most commonly prescribed in the United States – is increasingly implicated in overdose deaths. In response, several states have recently reclassified the antiepileptic as a Schedule V controlled substance. Other states have declined to go that far, but have added gabapentin (Neurontin and others) to the drugs that must be reported to state prescription monitoring programs.
“Gabapentin and pregabalin [Lyrica] are versatile and important drugs that are widely prescribed and used off label for a number of conditions from seizure disorder to fibromyalgia,” said Joseph Pergolizzi Jr., MD, who discussed the issue during the annual PAINWeek. “Abuse patterns differ somewhat from abuse patterns with prescription opioids. People who misuse gabapentinoids tend to already use other drugs inappropriately. It’s rare to find a person who only takes them recreationally, but it is increasingly common to find polydrug abusers who take gabapentinoids.”
Gabapentin abuse appears to be more common in Europe than in the United States, where it’s just beginning to emerge, Dr. Pergolizzi said. And the picture is more nuanced than it might first appear: Many of those who are misusing the drug are using it as a “do-it-yourself” opioid withdrawal aid, he said in an interview.
At the meeting, he presented a literature review comprising 46 papers on pregabalin abuse and 263 on gabapentin abuse. Several important themes arose from these papers, said Dr. Pergolizzi, cofounder and chief operating officer of NEMA Research Inc., Naples, Fla.:
- Gabapentin and pregabalin are being prescribed off label for numerous conditions, including bipolar disorder, neuropathic pain, diabetic neuropathy, complex regional pain syndrome, attention deficit disorder, restless leg syndrome, trigeminal neuralgia, periodic limb movement disorder of sleep, migraine, drug and alcohol withdrawal seizures, chronic low back pain, and even menopausal symptoms.
- About a one-third of users experience withdrawal symptoms with sudden discontinuation. Symptoms include disorientation, anxiety, insomnia, heart palpitations, diaphoresis, and abdominal cramps.
- Risk factors for abuse are emerging. These include opioid use disorders, mental illnesses, and a history of taking supratherapeutic doses of the drugs. Age and sex don’t seem to be a consistent risk factor for abuse.
- Abusers can obtain nonprescription gabapentinoids more easily each year, including street sales and online orders, Dr. Pergolizzi said. “A Google search conducted in July for ‘buy gabapentin without a prescription’ yielded 4.48 million results and ‘buy pregabalin without a prescription,’ 622,000. A similar search conducted in September 2017 yielded 1.19 million and 352,000 results, respectively.”
- Few urine drug assays screen for gabapentinoids, which makes them easy to conceal in random drug testing.
Reports of gabapentin-involved drug overdoses and even deaths have recently emerged in the United States, particularly in the opioid abuse-plagued Appalachian states. An article in May in Drug and Alcohol Dependence examined the prevalence of gabapentin in postmortem toxicology in drug overdose deaths in four Appalachian states in 2015 (Drug Alcohol Depend. 2018;186:80-5). Rates were 4% in eastern Tennessee, 15% in West Virginia, 20% in North Carolina, and 41% in Kentucky.
Three states have now added gabapentin to their list of Schedule V controlled substances: Kentucky in 2017, West Virginia this May, and Tennessee in July.
Ohio, Minnesota, Virginia, and Massachusetts have taken a different tack to controlling dispensing. In those states, all pharmacies, prescribers, and wholesalers must report all dispensing and sales of gabapentin to their prescription monitoring databases.
Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
LAS VEGAS – Up to 22% of opioid abusers also abuse gabapentin, taking high doses of the antiepileptic for its psychoactive effects, or to potentiate the effect of opioids.
The drug – the 10th most commonly prescribed in the United States – is increasingly implicated in overdose deaths. In response, several states have recently reclassified the antiepileptic as a Schedule V controlled substance. Other states have declined to go that far, but have added gabapentin (Neurontin and others) to the drugs that must be reported to state prescription monitoring programs.
“Gabapentin and pregabalin [Lyrica] are versatile and important drugs that are widely prescribed and used off label for a number of conditions from seizure disorder to fibromyalgia,” said Joseph Pergolizzi Jr., MD, who discussed the issue during the annual PAINWeek. “Abuse patterns differ somewhat from abuse patterns with prescription opioids. People who misuse gabapentinoids tend to already use other drugs inappropriately. It’s rare to find a person who only takes them recreationally, but it is increasingly common to find polydrug abusers who take gabapentinoids.”
Gabapentin abuse appears to be more common in Europe than in the United States, where it’s just beginning to emerge, Dr. Pergolizzi said. And the picture is more nuanced than it might first appear: Many of those who are misusing the drug are using it as a “do-it-yourself” opioid withdrawal aid, he said in an interview.
At the meeting, he presented a literature review comprising 46 papers on pregabalin abuse and 263 on gabapentin abuse. Several important themes arose from these papers, said Dr. Pergolizzi, cofounder and chief operating officer of NEMA Research Inc., Naples, Fla.:
- Gabapentin and pregabalin are being prescribed off label for numerous conditions, including bipolar disorder, neuropathic pain, diabetic neuropathy, complex regional pain syndrome, attention deficit disorder, restless leg syndrome, trigeminal neuralgia, periodic limb movement disorder of sleep, migraine, drug and alcohol withdrawal seizures, chronic low back pain, and even menopausal symptoms.
- About a one-third of users experience withdrawal symptoms with sudden discontinuation. Symptoms include disorientation, anxiety, insomnia, heart palpitations, diaphoresis, and abdominal cramps.
- Risk factors for abuse are emerging. These include opioid use disorders, mental illnesses, and a history of taking supratherapeutic doses of the drugs. Age and sex don’t seem to be a consistent risk factor for abuse.
- Abusers can obtain nonprescription gabapentinoids more easily each year, including street sales and online orders, Dr. Pergolizzi said. “A Google search conducted in July for ‘buy gabapentin without a prescription’ yielded 4.48 million results and ‘buy pregabalin without a prescription,’ 622,000. A similar search conducted in September 2017 yielded 1.19 million and 352,000 results, respectively.”
- Few urine drug assays screen for gabapentinoids, which makes them easy to conceal in random drug testing.
Reports of gabapentin-involved drug overdoses and even deaths have recently emerged in the United States, particularly in the opioid abuse-plagued Appalachian states. An article in May in Drug and Alcohol Dependence examined the prevalence of gabapentin in postmortem toxicology in drug overdose deaths in four Appalachian states in 2015 (Drug Alcohol Depend. 2018;186:80-5). Rates were 4% in eastern Tennessee, 15% in West Virginia, 20% in North Carolina, and 41% in Kentucky.
Three states have now added gabapentin to their list of Schedule V controlled substances: Kentucky in 2017, West Virginia this May, and Tennessee in July.
Ohio, Minnesota, Virginia, and Massachusetts have taken a different tack to controlling dispensing. In those states, all pharmacies, prescribers, and wholesalers must report all dispensing and sales of gabapentin to their prescription monitoring databases.
Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
LAS VEGAS – Up to 22% of opioid abusers also abuse gabapentin, taking high doses of the antiepileptic for its psychoactive effects, or to potentiate the effect of opioids.
The drug – the 10th most commonly prescribed in the United States – is increasingly implicated in overdose deaths. In response, several states have recently reclassified the antiepileptic as a Schedule V controlled substance. Other states have declined to go that far, but have added gabapentin (Neurontin and others) to the drugs that must be reported to state prescription monitoring programs.
“Gabapentin and pregabalin [Lyrica] are versatile and important drugs that are widely prescribed and used off label for a number of conditions from seizure disorder to fibromyalgia,” said Joseph Pergolizzi Jr., MD, who discussed the issue during the annual PAINWeek. “Abuse patterns differ somewhat from abuse patterns with prescription opioids. People who misuse gabapentinoids tend to already use other drugs inappropriately. It’s rare to find a person who only takes them recreationally, but it is increasingly common to find polydrug abusers who take gabapentinoids.”
Gabapentin abuse appears to be more common in Europe than in the United States, where it’s just beginning to emerge, Dr. Pergolizzi said. And the picture is more nuanced than it might first appear: Many of those who are misusing the drug are using it as a “do-it-yourself” opioid withdrawal aid, he said in an interview.
At the meeting, he presented a literature review comprising 46 papers on pregabalin abuse and 263 on gabapentin abuse. Several important themes arose from these papers, said Dr. Pergolizzi, cofounder and chief operating officer of NEMA Research Inc., Naples, Fla.:
- Gabapentin and pregabalin are being prescribed off label for numerous conditions, including bipolar disorder, neuropathic pain, diabetic neuropathy, complex regional pain syndrome, attention deficit disorder, restless leg syndrome, trigeminal neuralgia, periodic limb movement disorder of sleep, migraine, drug and alcohol withdrawal seizures, chronic low back pain, and even menopausal symptoms.
- About a one-third of users experience withdrawal symptoms with sudden discontinuation. Symptoms include disorientation, anxiety, insomnia, heart palpitations, diaphoresis, and abdominal cramps.
- Risk factors for abuse are emerging. These include opioid use disorders, mental illnesses, and a history of taking supratherapeutic doses of the drugs. Age and sex don’t seem to be a consistent risk factor for abuse.
- Abusers can obtain nonprescription gabapentinoids more easily each year, including street sales and online orders, Dr. Pergolizzi said. “A Google search conducted in July for ‘buy gabapentin without a prescription’ yielded 4.48 million results and ‘buy pregabalin without a prescription,’ 622,000. A similar search conducted in September 2017 yielded 1.19 million and 352,000 results, respectively.”
- Few urine drug assays screen for gabapentinoids, which makes them easy to conceal in random drug testing.
Reports of gabapentin-involved drug overdoses and even deaths have recently emerged in the United States, particularly in the opioid abuse-plagued Appalachian states. An article in May in Drug and Alcohol Dependence examined the prevalence of gabapentin in postmortem toxicology in drug overdose deaths in four Appalachian states in 2015 (Drug Alcohol Depend. 2018;186:80-5). Rates were 4% in eastern Tennessee, 15% in West Virginia, 20% in North Carolina, and 41% in Kentucky.
Three states have now added gabapentin to their list of Schedule V controlled substances: Kentucky in 2017, West Virginia this May, and Tennessee in July.
Ohio, Minnesota, Virginia, and Massachusetts have taken a different tack to controlling dispensing. In those states, all pharmacies, prescribers, and wholesalers must report all dispensing and sales of gabapentin to their prescription monitoring databases.
Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
REPORTING FROM PAINWEEK 2018
Key clinical point:
Major finding: Up to 22% of opioid users are also using gabapentin.
Study details: A literature review of 46 papers on pregabalin abuse and 263 on gabapentin abuse.
Disclosures: Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
Source: Pergolizzi J et al. PAINWeek 2018, Poster 55.
Residents curb IV antibiotic overuse in children
ATLANTA – It took less than a year to curb overuse of intravenous antibiotics at the Cincinnati Children’s Hospital, according to a report given at the Pediatric Hospital Medicine meeting.
Overuse of IV antibiotics – continuing IV formulations when oral formulations would work just as well – is a widespread concern in hospital medicine. Patients can often be switched to an oral antibiotic after an initial IV course. It lowers costs, lessens the risk of antimicrobial resistance, and reduces IV complications, but timely transitions don’t always happen.
They certainly weren’t happening at Cincinnati Children’s. “Despite a strong antimicrobial stewardship program, we identified a problem with overuse of IV antibiotics. The majority of pediatric patients admitted to an in-hospital service were started on IV antibiotics regardless of diagnosis or condition. Conversion to enteral antibiotics was often not considered until the day of discharge, even if patients were taking other enteral medications earlier in the admission,” said project leader Sonya Girdwood, MD, a research fellow at the hospital.
To get a handle on the problem, her team focused on two common IV antibiotics, ampicillin and clindamycin, that have oral equivalents with equal bioavailability: amoxicillin in the case of ampicillin, and oral clindamycin. To further define the project, they zeroed in on two common indications: clindamycin for uncomplicated skin and soft-tissue infections, and ampicillin for community-acquired pneumonia, in children over 2 months old.
The team figured that, if patients were able to take other oral medications, they should also be able to take oral antibiotics, so the goal of the project was to increase the rate of antibiotics given orally in children who were taking other enteral medications.
That percentage was 44% at baseline, and increased to 80% by month 8, saving an estimated $30,000 annually. There was no increase in 30-day readmissions. Length of stay held steady overall at about a day and half, but Dr. Girdwood suspected it might have been reduced for cellulitis.
Improvement efforts focused on residents and started in January 2017. Among the first lessons was that IV ampicillin is about 21 times more expensive than amoxicillin and that IV clindamycin is about twice as expensive as its oral formulation.
Residents were tasked with forming a plan at admission to transition children to oral antibiotics as soon as possible and to discuss those plans with attending physicians in preround huddles. Often, “this led to [transition] orders being placed even before rounds started,” Dr. Girdwood said.
A time was set up during evening huddles – 10 p.m. – for residents working overnight to discuss transition timing with attending. Failures – patients still on IV clindamycin or ampicillin when they were taking oral meds – were identified and shared with resident teams.
The gains have been maintained for almost a year with little backsliding; residents are reminded weekly of transition goals.
Children with skin and soft-tissue infections with bone or eye involvement were excluded from the project, along with pneumonia patients with chest tubes or complex or loculated effusions requiring a surgery consult.
There was no external funding, and the investigators had no disclosures.
ATLANTA – It took less than a year to curb overuse of intravenous antibiotics at the Cincinnati Children’s Hospital, according to a report given at the Pediatric Hospital Medicine meeting.
Overuse of IV antibiotics – continuing IV formulations when oral formulations would work just as well – is a widespread concern in hospital medicine. Patients can often be switched to an oral antibiotic after an initial IV course. It lowers costs, lessens the risk of antimicrobial resistance, and reduces IV complications, but timely transitions don’t always happen.
They certainly weren’t happening at Cincinnati Children’s. “Despite a strong antimicrobial stewardship program, we identified a problem with overuse of IV antibiotics. The majority of pediatric patients admitted to an in-hospital service were started on IV antibiotics regardless of diagnosis or condition. Conversion to enteral antibiotics was often not considered until the day of discharge, even if patients were taking other enteral medications earlier in the admission,” said project leader Sonya Girdwood, MD, a research fellow at the hospital.
To get a handle on the problem, her team focused on two common IV antibiotics, ampicillin and clindamycin, that have oral equivalents with equal bioavailability: amoxicillin in the case of ampicillin, and oral clindamycin. To further define the project, they zeroed in on two common indications: clindamycin for uncomplicated skin and soft-tissue infections, and ampicillin for community-acquired pneumonia, in children over 2 months old.
The team figured that, if patients were able to take other oral medications, they should also be able to take oral antibiotics, so the goal of the project was to increase the rate of antibiotics given orally in children who were taking other enteral medications.
That percentage was 44% at baseline, and increased to 80% by month 8, saving an estimated $30,000 annually. There was no increase in 30-day readmissions. Length of stay held steady overall at about a day and half, but Dr. Girdwood suspected it might have been reduced for cellulitis.
Improvement efforts focused on residents and started in January 2017. Among the first lessons was that IV ampicillin is about 21 times more expensive than amoxicillin and that IV clindamycin is about twice as expensive as its oral formulation.
Residents were tasked with forming a plan at admission to transition children to oral antibiotics as soon as possible and to discuss those plans with attending physicians in preround huddles. Often, “this led to [transition] orders being placed even before rounds started,” Dr. Girdwood said.
A time was set up during evening huddles – 10 p.m. – for residents working overnight to discuss transition timing with attending. Failures – patients still on IV clindamycin or ampicillin when they were taking oral meds – were identified and shared with resident teams.
The gains have been maintained for almost a year with little backsliding; residents are reminded weekly of transition goals.
Children with skin and soft-tissue infections with bone or eye involvement were excluded from the project, along with pneumonia patients with chest tubes or complex or loculated effusions requiring a surgery consult.
There was no external funding, and the investigators had no disclosures.
ATLANTA – It took less than a year to curb overuse of intravenous antibiotics at the Cincinnati Children’s Hospital, according to a report given at the Pediatric Hospital Medicine meeting.
Overuse of IV antibiotics – continuing IV formulations when oral formulations would work just as well – is a widespread concern in hospital medicine. Patients can often be switched to an oral antibiotic after an initial IV course. It lowers costs, lessens the risk of antimicrobial resistance, and reduces IV complications, but timely transitions don’t always happen.
They certainly weren’t happening at Cincinnati Children’s. “Despite a strong antimicrobial stewardship program, we identified a problem with overuse of IV antibiotics. The majority of pediatric patients admitted to an in-hospital service were started on IV antibiotics regardless of diagnosis or condition. Conversion to enteral antibiotics was often not considered until the day of discharge, even if patients were taking other enteral medications earlier in the admission,” said project leader Sonya Girdwood, MD, a research fellow at the hospital.
To get a handle on the problem, her team focused on two common IV antibiotics, ampicillin and clindamycin, that have oral equivalents with equal bioavailability: amoxicillin in the case of ampicillin, and oral clindamycin. To further define the project, they zeroed in on two common indications: clindamycin for uncomplicated skin and soft-tissue infections, and ampicillin for community-acquired pneumonia, in children over 2 months old.
The team figured that, if patients were able to take other oral medications, they should also be able to take oral antibiotics, so the goal of the project was to increase the rate of antibiotics given orally in children who were taking other enteral medications.
That percentage was 44% at baseline, and increased to 80% by month 8, saving an estimated $30,000 annually. There was no increase in 30-day readmissions. Length of stay held steady overall at about a day and half, but Dr. Girdwood suspected it might have been reduced for cellulitis.
Improvement efforts focused on residents and started in January 2017. Among the first lessons was that IV ampicillin is about 21 times more expensive than amoxicillin and that IV clindamycin is about twice as expensive as its oral formulation.
Residents were tasked with forming a plan at admission to transition children to oral antibiotics as soon as possible and to discuss those plans with attending physicians in preround huddles. Often, “this led to [transition] orders being placed even before rounds started,” Dr. Girdwood said.
A time was set up during evening huddles – 10 p.m. – for residents working overnight to discuss transition timing with attending. Failures – patients still on IV clindamycin or ampicillin when they were taking oral meds – were identified and shared with resident teams.
The gains have been maintained for almost a year with little backsliding; residents are reminded weekly of transition goals.
Children with skin and soft-tissue infections with bone or eye involvement were excluded from the project, along with pneumonia patients with chest tubes or complex or loculated effusions requiring a surgery consult.
There was no external funding, and the investigators had no disclosures.
REPORTING FROM PHM 2018
Key clinical point:
Major finding: The percentage of antibiotics given orally to children who were taking other enteral medications rose from 44% to 80% over 8 months, saving an estimated $30,000 annually.
Study details: Quality improvement project
Disclosures: There was no external funding, and the investigators didn’t have any disclosures.
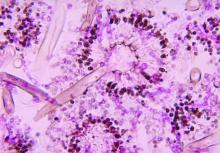
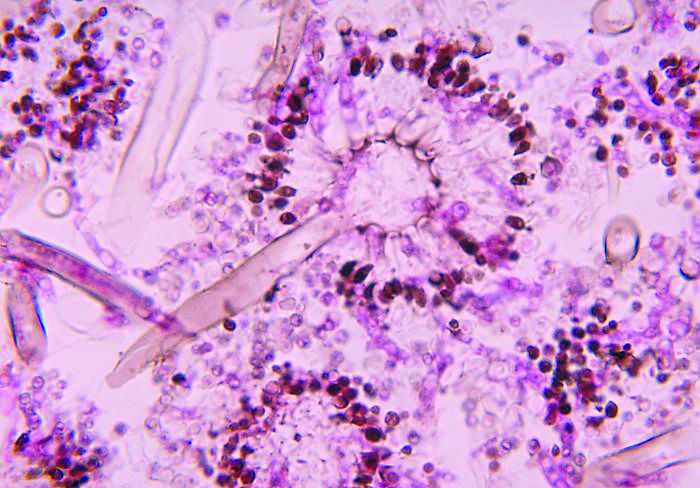
Severe influenza increases risk of invasive pulmonary aspergillosis in the ICU
Severe influenza is an independent risk factor for invasive pulmonary aspergillosis with an accompanying increased mortality in the ICU, according to a multicenter retrospective cohort study at seven tertiary centers in Belgium and the Netherlands.
Data was collected from criteria-meeting adult patients admitted to the ICU for more than 24 hours with acute respiratory failure during the 2009-2016 influenza seasons. The included cohort of 432 patients was composed of 56% men and had a median age of 59 years; all participants were diagnosed as having severe type A or type B influenza infection according to positive airway RT-PCR results.
The full cohort was subcategorized into 117 immunocompromised and 315 as nonimmunocompromised individuals using criteria established by the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study group (EORTC/MSG) . To assess influenza as an independent variable in the development of invasive pulmonary aspergillosis, the 315 nonimmunocompromised influenza positive individuals were compared to an influenza-negative control group of 315 nonimmunocompromised patients admitted to the ICU that presented similar respiratory insufficiency symptoms with community-acquired pneumonia.
Determination of other independent risk factors for incidence of invasive pulmonary aspergillosis was achieved by multivariate analysis of factors such as sex, diabetes status, prednisone use, age, and acute physiology and chronic health evaluation (APACHE) II score. The mean APACHE II score was 22, with the majority of patients requiring intubation for mechanical ventilation for a median duration of 11 days.
Influenza is not considered a host factor for invasive pulmonary aspergillosis and will often miss being diagnosed when using strict interpretation of the current EORTC/MSG or AspICU algorithm criteria, according to the researchers. Consequently for patients with influenza and the noninfluenza control group with community-acquired pneumonia, the definition of invasive pulmonary aspergillosis was modified from the AspICU algorithm. Stringent mycological criteria, including bronchoaveolar lavage (BAL) culture, a positive Aspergillus culture, positive galactomannan test, and/or positive serum galactomannan tests, provided supporting diagnostics for an invasive pulmonary aspergillosis determination.
At a median of 3 days following admission to the ICU, a diagnosis of invasive pulmonary aspergillosis was determined for 19% of the 432 influenza patients. Similar incident percentages of invasive pulmonary aspergillosis occurring for type A and type B, 71/355 (20%) and 12/77 (16%) patients respectively, showed that there was no clear association of the disease development with influenza subtypes that occurred during different annual seasons.
AspICU or EORTC/MSG criteria characterized only 43% and 58% of cases as proven or possible aspergillosis, respectively. On the other hand, stringent mycological tests yielded better invasive pulmonary aspergillosis classification, with 63% of BAL cultures being positive for Aspergillus, 88% of BAL galactomannan tests being positive, and 65% of serum galactomannan tests being positive in the 81/83 patients tested.
The study found that, for influenza patients, being immunocompromised more than doubled the incidence of invasive pulmonary aspergillosis, at 32% versus the 14% of those patients who were nonimmunocompromised. In contrast only 5% in the control group developed invasive pulmonary aspergillosis.
Influenza patients who developed invasive pulmonary aspergillosis in the ICU tended to have their stays significantly lengthened from 9 days (interquartile range, 5-20 days) for those without it to 19 days (IQR, 12-38 days) for those infected (P less than .0001). Likewise, 90-day mortality significantly rose from 28% for those influenza patients without invasive pulmonary aspergillosis to 51% for those with it (P = .0001).
The authors concluded that influenza was “independently associated with invasive pulmonary aspergillosis (adjusted odds ratio, 5.19; P less than.0001) along with a higher APACHE II score, male sex, and use of corticosteroids.”
Furthermore, as influenza appears to be an independent risk factor for invasive pulmonary aspergillosis and its associated high mortality, the authors suggested that “future studies should assess whether a faster diagnosis or antifungal prophylaxis could improve the outcome of influenza-associated aspergillosis.”
The authors reported that they had no conflicts of interest.
SOURCE: Schauwvlieghe AFAD et al. Lancet Respir Med. 2018 Jul 31. doi: 10.1016/S2213-2600(18)30274-1
Severe influenza is an independent risk factor for invasive pulmonary aspergillosis with an accompanying increased mortality in the ICU, according to a multicenter retrospective cohort study at seven tertiary centers in Belgium and the Netherlands.
Data was collected from criteria-meeting adult patients admitted to the ICU for more than 24 hours with acute respiratory failure during the 2009-2016 influenza seasons. The included cohort of 432 patients was composed of 56% men and had a median age of 59 years; all participants were diagnosed as having severe type A or type B influenza infection according to positive airway RT-PCR results.
The full cohort was subcategorized into 117 immunocompromised and 315 as nonimmunocompromised individuals using criteria established by the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study group (EORTC/MSG) . To assess influenza as an independent variable in the development of invasive pulmonary aspergillosis, the 315 nonimmunocompromised influenza positive individuals were compared to an influenza-negative control group of 315 nonimmunocompromised patients admitted to the ICU that presented similar respiratory insufficiency symptoms with community-acquired pneumonia.
Determination of other independent risk factors for incidence of invasive pulmonary aspergillosis was achieved by multivariate analysis of factors such as sex, diabetes status, prednisone use, age, and acute physiology and chronic health evaluation (APACHE) II score. The mean APACHE II score was 22, with the majority of patients requiring intubation for mechanical ventilation for a median duration of 11 days.
Influenza is not considered a host factor for invasive pulmonary aspergillosis and will often miss being diagnosed when using strict interpretation of the current EORTC/MSG or AspICU algorithm criteria, according to the researchers. Consequently for patients with influenza and the noninfluenza control group with community-acquired pneumonia, the definition of invasive pulmonary aspergillosis was modified from the AspICU algorithm. Stringent mycological criteria, including bronchoaveolar lavage (BAL) culture, a positive Aspergillus culture, positive galactomannan test, and/or positive serum galactomannan tests, provided supporting diagnostics for an invasive pulmonary aspergillosis determination.
At a median of 3 days following admission to the ICU, a diagnosis of invasive pulmonary aspergillosis was determined for 19% of the 432 influenza patients. Similar incident percentages of invasive pulmonary aspergillosis occurring for type A and type B, 71/355 (20%) and 12/77 (16%) patients respectively, showed that there was no clear association of the disease development with influenza subtypes that occurred during different annual seasons.
AspICU or EORTC/MSG criteria characterized only 43% and 58% of cases as proven or possible aspergillosis, respectively. On the other hand, stringent mycological tests yielded better invasive pulmonary aspergillosis classification, with 63% of BAL cultures being positive for Aspergillus, 88% of BAL galactomannan tests being positive, and 65% of serum galactomannan tests being positive in the 81/83 patients tested.
The study found that, for influenza patients, being immunocompromised more than doubled the incidence of invasive pulmonary aspergillosis, at 32% versus the 14% of those patients who were nonimmunocompromised. In contrast only 5% in the control group developed invasive pulmonary aspergillosis.
Influenza patients who developed invasive pulmonary aspergillosis in the ICU tended to have their stays significantly lengthened from 9 days (interquartile range, 5-20 days) for those without it to 19 days (IQR, 12-38 days) for those infected (P less than .0001). Likewise, 90-day mortality significantly rose from 28% for those influenza patients without invasive pulmonary aspergillosis to 51% for those with it (P = .0001).
The authors concluded that influenza was “independently associated with invasive pulmonary aspergillosis (adjusted odds ratio, 5.19; P less than.0001) along with a higher APACHE II score, male sex, and use of corticosteroids.”
Furthermore, as influenza appears to be an independent risk factor for invasive pulmonary aspergillosis and its associated high mortality, the authors suggested that “future studies should assess whether a faster diagnosis or antifungal prophylaxis could improve the outcome of influenza-associated aspergillosis.”
The authors reported that they had no conflicts of interest.
SOURCE: Schauwvlieghe AFAD et al. Lancet Respir Med. 2018 Jul 31. doi: 10.1016/S2213-2600(18)30274-1
Severe influenza is an independent risk factor for invasive pulmonary aspergillosis with an accompanying increased mortality in the ICU, according to a multicenter retrospective cohort study at seven tertiary centers in Belgium and the Netherlands.
Data was collected from criteria-meeting adult patients admitted to the ICU for more than 24 hours with acute respiratory failure during the 2009-2016 influenza seasons. The included cohort of 432 patients was composed of 56% men and had a median age of 59 years; all participants were diagnosed as having severe type A or type B influenza infection according to positive airway RT-PCR results.
The full cohort was subcategorized into 117 immunocompromised and 315 as nonimmunocompromised individuals using criteria established by the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study group (EORTC/MSG) . To assess influenza as an independent variable in the development of invasive pulmonary aspergillosis, the 315 nonimmunocompromised influenza positive individuals were compared to an influenza-negative control group of 315 nonimmunocompromised patients admitted to the ICU that presented similar respiratory insufficiency symptoms with community-acquired pneumonia.
Determination of other independent risk factors for incidence of invasive pulmonary aspergillosis was achieved by multivariate analysis of factors such as sex, diabetes status, prednisone use, age, and acute physiology and chronic health evaluation (APACHE) II score. The mean APACHE II score was 22, with the majority of patients requiring intubation for mechanical ventilation for a median duration of 11 days.
Influenza is not considered a host factor for invasive pulmonary aspergillosis and will often miss being diagnosed when using strict interpretation of the current EORTC/MSG or AspICU algorithm criteria, according to the researchers. Consequently for patients with influenza and the noninfluenza control group with community-acquired pneumonia, the definition of invasive pulmonary aspergillosis was modified from the AspICU algorithm. Stringent mycological criteria, including bronchoaveolar lavage (BAL) culture, a positive Aspergillus culture, positive galactomannan test, and/or positive serum galactomannan tests, provided supporting diagnostics for an invasive pulmonary aspergillosis determination.
At a median of 3 days following admission to the ICU, a diagnosis of invasive pulmonary aspergillosis was determined for 19% of the 432 influenza patients. Similar incident percentages of invasive pulmonary aspergillosis occurring for type A and type B, 71/355 (20%) and 12/77 (16%) patients respectively, showed that there was no clear association of the disease development with influenza subtypes that occurred during different annual seasons.
AspICU or EORTC/MSG criteria characterized only 43% and 58% of cases as proven or possible aspergillosis, respectively. On the other hand, stringent mycological tests yielded better invasive pulmonary aspergillosis classification, with 63% of BAL cultures being positive for Aspergillus, 88% of BAL galactomannan tests being positive, and 65% of serum galactomannan tests being positive in the 81/83 patients tested.
The study found that, for influenza patients, being immunocompromised more than doubled the incidence of invasive pulmonary aspergillosis, at 32% versus the 14% of those patients who were nonimmunocompromised. In contrast only 5% in the control group developed invasive pulmonary aspergillosis.
Influenza patients who developed invasive pulmonary aspergillosis in the ICU tended to have their stays significantly lengthened from 9 days (interquartile range, 5-20 days) for those without it to 19 days (IQR, 12-38 days) for those infected (P less than .0001). Likewise, 90-day mortality significantly rose from 28% for those influenza patients without invasive pulmonary aspergillosis to 51% for those with it (P = .0001).
The authors concluded that influenza was “independently associated with invasive pulmonary aspergillosis (adjusted odds ratio, 5.19; P less than.0001) along with a higher APACHE II score, male sex, and use of corticosteroids.”
Furthermore, as influenza appears to be an independent risk factor for invasive pulmonary aspergillosis and its associated high mortality, the authors suggested that “future studies should assess whether a faster diagnosis or antifungal prophylaxis could improve the outcome of influenza-associated aspergillosis.”
The authors reported that they had no conflicts of interest.
SOURCE: Schauwvlieghe AFAD et al. Lancet Respir Med. 2018 Jul 31. doi: 10.1016/S2213-2600(18)30274-1
FROM THE LANCET RESPIRATORY MEDICINE
Key clinical point: ICU admission for severe influenza as significant a risk factor should be included in the existing diagnostic criteria for predicting incidence of invasive pulmonary aspergillosis.
Major finding: Influenza is an independent risk factor associated with invasive pulmonary aspergillosis, with 90-day mortality rising from 28% to 51% when this fungal infection occurs.
Study details: Multicenter retrospective study of 432 adult patients with confirmed severe influenza admitted to the ICU with acute respiratory failure.
Disclosures: The authors reported that they had no conflicts of interest.
Source: Schauwvlieghe AFAD et al. Lancet Respir Med. 2018 Jul 31. doi: 10.1016/S2213-2600(18)30274-1.
Troponin I: Powerful all-cause mortality risk marker in COPD
PARIS – High relative even after researchers adjusted for all major cardiovascular and COPD prognostic indicators, according to a late-breaker presentation at the annual congress of the European Respiratory Society.
Troponin I is detectable in the plasma of most patients with COPD, but relative increases in troponin I correlate with greater relative increases in most cardiovascular and COPD risk factors, according to Benjamin Waschki, MD, Pulmonary Research Institute, LungenClinic, Grosshansdorf, Germany.
The relationship between increased troponin I and increased all-cause mortality was observed in an on-going prospective multicenter cohort of COPD patients followed at 31 centers in Germany. The cohort is called COSYCONET and it began in 2010. The current analysis evaluated 2,020 COPD patients without regard to stage of disease.
There were 136 deaths over the course of follow-up. Without adjustment, the hazard ratio (HR) for death was more than twofold higher in the highest quartile of troponin I (equal to or greater than 6.6 ng/mL), when compared with the lowest (under 2.5 ng/mL) (HR, 2.42; P less than .001). Graphically, the mortality curves for each of the quartiles began to separate at about 12 months, widening in a stepwise manner for greater likelihood of death from the lowest to highest quartiles.
The risk of death from any cause remained elevated for the highest relative to lowest troponin I quartiles after adjusting for cardiovascular risk factors and after adjusting for COPD severity. Again, there was a distinct stepwise separation of the mortality curves for each higher troponin quartile,
Of particular importance, troponin I remained predictive beyond the BODE index, which is a currently employed prognostic mortality predictor in COPD, according to Dr. Waschki. When defining elevated troponin as greater than 6 ng/ML and a high BODE score as greater than 4, mortality was higher for those with a high BODE and low troponin than a high troponin and low BODE, (P less than .001), but a high troponin I was associated with a higher risk of mortality when BODE was low (P less than .001). Moreover, when both troponin I and BODE were elevated, all-cause mortality was more than doubled, relative to those without either risk factor (HR, 2.56; P = .003), Dr. Waschki reported.
After researchers adjusted for major cardiovascular risk factors, such as history of MI and renal impairment, and for major COPD risk factors, such as 6-minute walk test and BODE index, those in the highest quartile had a more than 50% greater risk of death relative to those in the lower quartile over the 3 years of follow-up (HR, 1.69; P = .007), according to Dr. Waschki.
Although troponin I is best known for its diagnostic role in MI, it is now being evaluated as a risk stratifier for many chronic diseases, such as heart failure and chronic kidney disease, explained Dr. Waschki in providing background for this study. He reported that many groups are looking at this as a marker of risk in a variety of chronic diseases.
In fact, a group working independently published a study in COPD just weeks before the ERS Congress that was complementary to those presented by Dr. Waschki. In this study, the goal was to evaluate troponin I as a predictor of cardiovascular events and cardiovascular death (Adamson PD et al. J Am Coll Cardiol 2018;72:1126-37). Performed as a subgroup analysis of 1,599 COPD patients participating in a large treatment trial, there was an almost fourfold increase in the risk of cardiovascular events (HR, 3.7; P = .012) when those in the highest quintile of troponin I (greater than 7.7 ng/ML) were compared with those in the lowest quintile (less than 2.3 ng/mL).
When compared for cardiovascular death, the highest quintile, relative to the lowest quintile, had a more than 20-fold increased risk of cardiovascular death (HR 20.1; P = .005). In the Adamson et al. study, which evaluated inhaled therapies for COPD, treatment response had no impact on troponin I levels or on the risk of cardiovascular events or death.
Based on this study and his own data, Dr. Waschki believes troponin I, which is readily ordered laboratory value, appears to be a useful tool for identifying COPD patients at high risk of death.
“The major message is that after adjusting for all known COPD and cardiovascular risk factors, troponin I remains a significant independent predictor of mortality,” he said.
Dr. Waschki reports no relevant conflicts of interest.
PARIS – High relative even after researchers adjusted for all major cardiovascular and COPD prognostic indicators, according to a late-breaker presentation at the annual congress of the European Respiratory Society.
Troponin I is detectable in the plasma of most patients with COPD, but relative increases in troponin I correlate with greater relative increases in most cardiovascular and COPD risk factors, according to Benjamin Waschki, MD, Pulmonary Research Institute, LungenClinic, Grosshansdorf, Germany.
The relationship between increased troponin I and increased all-cause mortality was observed in an on-going prospective multicenter cohort of COPD patients followed at 31 centers in Germany. The cohort is called COSYCONET and it began in 2010. The current analysis evaluated 2,020 COPD patients without regard to stage of disease.
There were 136 deaths over the course of follow-up. Without adjustment, the hazard ratio (HR) for death was more than twofold higher in the highest quartile of troponin I (equal to or greater than 6.6 ng/mL), when compared with the lowest (under 2.5 ng/mL) (HR, 2.42; P less than .001). Graphically, the mortality curves for each of the quartiles began to separate at about 12 months, widening in a stepwise manner for greater likelihood of death from the lowest to highest quartiles.
The risk of death from any cause remained elevated for the highest relative to lowest troponin I quartiles after adjusting for cardiovascular risk factors and after adjusting for COPD severity. Again, there was a distinct stepwise separation of the mortality curves for each higher troponin quartile,
Of particular importance, troponin I remained predictive beyond the BODE index, which is a currently employed prognostic mortality predictor in COPD, according to Dr. Waschki. When defining elevated troponin as greater than 6 ng/ML and a high BODE score as greater than 4, mortality was higher for those with a high BODE and low troponin than a high troponin and low BODE, (P less than .001), but a high troponin I was associated with a higher risk of mortality when BODE was low (P less than .001). Moreover, when both troponin I and BODE were elevated, all-cause mortality was more than doubled, relative to those without either risk factor (HR, 2.56; P = .003), Dr. Waschki reported.
After researchers adjusted for major cardiovascular risk factors, such as history of MI and renal impairment, and for major COPD risk factors, such as 6-minute walk test and BODE index, those in the highest quartile had a more than 50% greater risk of death relative to those in the lower quartile over the 3 years of follow-up (HR, 1.69; P = .007), according to Dr. Waschki.
Although troponin I is best known for its diagnostic role in MI, it is now being evaluated as a risk stratifier for many chronic diseases, such as heart failure and chronic kidney disease, explained Dr. Waschki in providing background for this study. He reported that many groups are looking at this as a marker of risk in a variety of chronic diseases.
In fact, a group working independently published a study in COPD just weeks before the ERS Congress that was complementary to those presented by Dr. Waschki. In this study, the goal was to evaluate troponin I as a predictor of cardiovascular events and cardiovascular death (Adamson PD et al. J Am Coll Cardiol 2018;72:1126-37). Performed as a subgroup analysis of 1,599 COPD patients participating in a large treatment trial, there was an almost fourfold increase in the risk of cardiovascular events (HR, 3.7; P = .012) when those in the highest quintile of troponin I (greater than 7.7 ng/ML) were compared with those in the lowest quintile (less than 2.3 ng/mL).
When compared for cardiovascular death, the highest quintile, relative to the lowest quintile, had a more than 20-fold increased risk of cardiovascular death (HR 20.1; P = .005). In the Adamson et al. study, which evaluated inhaled therapies for COPD, treatment response had no impact on troponin I levels or on the risk of cardiovascular events or death.
Based on this study and his own data, Dr. Waschki believes troponin I, which is readily ordered laboratory value, appears to be a useful tool for identifying COPD patients at high risk of death.
“The major message is that after adjusting for all known COPD and cardiovascular risk factors, troponin I remains a significant independent predictor of mortality,” he said.
Dr. Waschki reports no relevant conflicts of interest.
PARIS – High relative even after researchers adjusted for all major cardiovascular and COPD prognostic indicators, according to a late-breaker presentation at the annual congress of the European Respiratory Society.
Troponin I is detectable in the plasma of most patients with COPD, but relative increases in troponin I correlate with greater relative increases in most cardiovascular and COPD risk factors, according to Benjamin Waschki, MD, Pulmonary Research Institute, LungenClinic, Grosshansdorf, Germany.
The relationship between increased troponin I and increased all-cause mortality was observed in an on-going prospective multicenter cohort of COPD patients followed at 31 centers in Germany. The cohort is called COSYCONET and it began in 2010. The current analysis evaluated 2,020 COPD patients without regard to stage of disease.
There were 136 deaths over the course of follow-up. Without adjustment, the hazard ratio (HR) for death was more than twofold higher in the highest quartile of troponin I (equal to or greater than 6.6 ng/mL), when compared with the lowest (under 2.5 ng/mL) (HR, 2.42; P less than .001). Graphically, the mortality curves for each of the quartiles began to separate at about 12 months, widening in a stepwise manner for greater likelihood of death from the lowest to highest quartiles.
The risk of death from any cause remained elevated for the highest relative to lowest troponin I quartiles after adjusting for cardiovascular risk factors and after adjusting for COPD severity. Again, there was a distinct stepwise separation of the mortality curves for each higher troponin quartile,
Of particular importance, troponin I remained predictive beyond the BODE index, which is a currently employed prognostic mortality predictor in COPD, according to Dr. Waschki. When defining elevated troponin as greater than 6 ng/ML and a high BODE score as greater than 4, mortality was higher for those with a high BODE and low troponin than a high troponin and low BODE, (P less than .001), but a high troponin I was associated with a higher risk of mortality when BODE was low (P less than .001). Moreover, when both troponin I and BODE were elevated, all-cause mortality was more than doubled, relative to those without either risk factor (HR, 2.56; P = .003), Dr. Waschki reported.
After researchers adjusted for major cardiovascular risk factors, such as history of MI and renal impairment, and for major COPD risk factors, such as 6-minute walk test and BODE index, those in the highest quartile had a more than 50% greater risk of death relative to those in the lower quartile over the 3 years of follow-up (HR, 1.69; P = .007), according to Dr. Waschki.
Although troponin I is best known for its diagnostic role in MI, it is now being evaluated as a risk stratifier for many chronic diseases, such as heart failure and chronic kidney disease, explained Dr. Waschki in providing background for this study. He reported that many groups are looking at this as a marker of risk in a variety of chronic diseases.
In fact, a group working independently published a study in COPD just weeks before the ERS Congress that was complementary to those presented by Dr. Waschki. In this study, the goal was to evaluate troponin I as a predictor of cardiovascular events and cardiovascular death (Adamson PD et al. J Am Coll Cardiol 2018;72:1126-37). Performed as a subgroup analysis of 1,599 COPD patients participating in a large treatment trial, there was an almost fourfold increase in the risk of cardiovascular events (HR, 3.7; P = .012) when those in the highest quintile of troponin I (greater than 7.7 ng/ML) were compared with those in the lowest quintile (less than 2.3 ng/mL).
When compared for cardiovascular death, the highest quintile, relative to the lowest quintile, had a more than 20-fold increased risk of cardiovascular death (HR 20.1; P = .005). In the Adamson et al. study, which evaluated inhaled therapies for COPD, treatment response had no impact on troponin I levels or on the risk of cardiovascular events or death.
Based on this study and his own data, Dr. Waschki believes troponin I, which is readily ordered laboratory value, appears to be a useful tool for identifying COPD patients at high risk of death.
“The major message is that after adjusting for all known COPD and cardiovascular risk factors, troponin I remains a significant independent predictor of mortality,” he said.
Dr. Waschki reports no relevant conflicts of interest.
REPORTING FROM ERS CONGRESS 2018
Key clinical point: Elevated troponin I identifies COPD patients with increased mortality risk independent of all other clinical risk markers.
Major finding: With high troponin I levels, all-cause mortality was increased 69% after researchers adjusted for other risk markers.
Study details: Analysis drawn from on-going multicenter cohort study
Disclosures: Dr. Waschki reports no relevant conflicts of interest.
Most in-hospital pneumonia deaths may not be preventable
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
FROM CHEST
Key clinical point: Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy.
Major finding: Two out of 52 patients who died in-hospital from community-acquired pneumonia (CAP) who were undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care that potentially contributed to their death.
Study details: A secondary analysis of the prospective multicenter Etiology of Pneumonia in the Community (EPIC) study involving 2,320 adults with radiographically confirmed CAP.
Disclosures: Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
Source: Waterer G. et al. CHEST 2018;154(3):628-35.
Confirmed: Growth in overdose deaths is exponential
Overdose death rates for individual drugs show no particular patterns since the turn of the century, but the exponential growth of overall drug mortality actually started before the opioid epidemic, according to an analysis of almost 600,000 unintentional overdose deaths since 1979.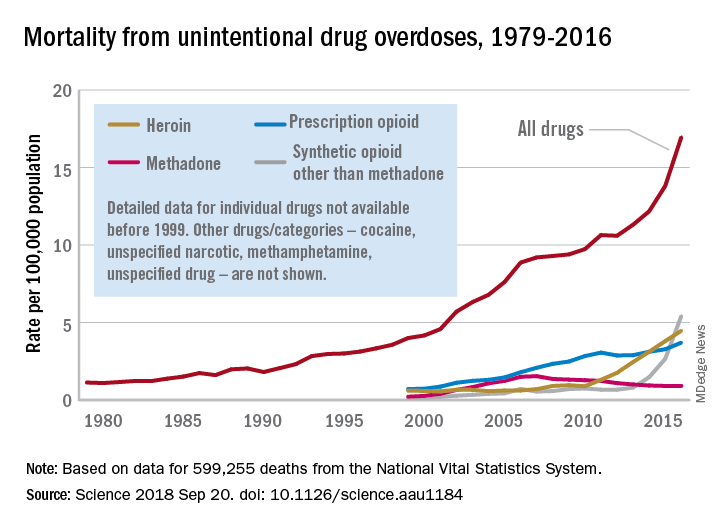
“The current epidemic of overdose deaths due to prescription opioids, heroin, and fentanyl appears to be the most recent manifestation of a more fundamental, longer-term process,” senior author Donald S. Burke, MD, of the University of Pittsburgh, said in a written statement.
Overdose mortality from all types of drugs rose from 1.13 per 100,000 population in 1979 to 16.96 per 100,000 in 2016, based on data for 599,255 deaths from unintentional drug overdoses in the National Vital Statistics System, they reported in Science.
When the investigators plotted annual drug overdose mortality over that 38-year period, they saw a smooth upward exponential curve with a doubling time of about 9 years. “This remarkably smooth, long-term epidemic growth pattern really caught our attention,” Dr. Burke said. “If we can figure it out, we should be able to bend that curve downward.”
The individual drug types that make up the whole, however, are a different story. “There is no regular or predictable pattern to the overdose rates for any of these drugs. Cocaine overdose death rates curved down and up and down and back up over the past 20 years. Methadone deaths have been on a downturn since the mid-2000s. Prescription opioids have been on a fairly steady, steep climb. Heroin deaths shot up in 2010, followed in 2013 by synthetic opioids, such as fentanyl,” lead author Hawre Jalal, MD, PhD, also of the university, said in the statement.
Geographic and demographic analyses produced the same absence of patterns. the researchers wrote.
The study was supported by grants from the Centers for Disease Control and Prevention and the Robert Wood Johnson Foundation. The investigators said they have no competing interests.
SOURCE: Jalal H et al. Science 2018 Sep 20. doi: 10.1126/science.aau1184.
Overdose death rates for individual drugs show no particular patterns since the turn of the century, but the exponential growth of overall drug mortality actually started before the opioid epidemic, according to an analysis of almost 600,000 unintentional overdose deaths since 1979.
“The current epidemic of overdose deaths due to prescription opioids, heroin, and fentanyl appears to be the most recent manifestation of a more fundamental, longer-term process,” senior author Donald S. Burke, MD, of the University of Pittsburgh, said in a written statement.
Overdose mortality from all types of drugs rose from 1.13 per 100,000 population in 1979 to 16.96 per 100,000 in 2016, based on data for 599,255 deaths from unintentional drug overdoses in the National Vital Statistics System, they reported in Science.
When the investigators plotted annual drug overdose mortality over that 38-year period, they saw a smooth upward exponential curve with a doubling time of about 9 years. “This remarkably smooth, long-term epidemic growth pattern really caught our attention,” Dr. Burke said. “If we can figure it out, we should be able to bend that curve downward.”
The individual drug types that make up the whole, however, are a different story. “There is no regular or predictable pattern to the overdose rates for any of these drugs. Cocaine overdose death rates curved down and up and down and back up over the past 20 years. Methadone deaths have been on a downturn since the mid-2000s. Prescription opioids have been on a fairly steady, steep climb. Heroin deaths shot up in 2010, followed in 2013 by synthetic opioids, such as fentanyl,” lead author Hawre Jalal, MD, PhD, also of the university, said in the statement.
Geographic and demographic analyses produced the same absence of patterns. the researchers wrote.
The study was supported by grants from the Centers for Disease Control and Prevention and the Robert Wood Johnson Foundation. The investigators said they have no competing interests.
SOURCE: Jalal H et al. Science 2018 Sep 20. doi: 10.1126/science.aau1184.
Overdose death rates for individual drugs show no particular patterns since the turn of the century, but the exponential growth of overall drug mortality actually started before the opioid epidemic, according to an analysis of almost 600,000 unintentional overdose deaths since 1979.
“The current epidemic of overdose deaths due to prescription opioids, heroin, and fentanyl appears to be the most recent manifestation of a more fundamental, longer-term process,” senior author Donald S. Burke, MD, of the University of Pittsburgh, said in a written statement.
Overdose mortality from all types of drugs rose from 1.13 per 100,000 population in 1979 to 16.96 per 100,000 in 2016, based on data for 599,255 deaths from unintentional drug overdoses in the National Vital Statistics System, they reported in Science.
When the investigators plotted annual drug overdose mortality over that 38-year period, they saw a smooth upward exponential curve with a doubling time of about 9 years. “This remarkably smooth, long-term epidemic growth pattern really caught our attention,” Dr. Burke said. “If we can figure it out, we should be able to bend that curve downward.”
The individual drug types that make up the whole, however, are a different story. “There is no regular or predictable pattern to the overdose rates for any of these drugs. Cocaine overdose death rates curved down and up and down and back up over the past 20 years. Methadone deaths have been on a downturn since the mid-2000s. Prescription opioids have been on a fairly steady, steep climb. Heroin deaths shot up in 2010, followed in 2013 by synthetic opioids, such as fentanyl,” lead author Hawre Jalal, MD, PhD, also of the university, said in the statement.
Geographic and demographic analyses produced the same absence of patterns. the researchers wrote.
The study was supported by grants from the Centers for Disease Control and Prevention and the Robert Wood Johnson Foundation. The investigators said they have no competing interests.
SOURCE: Jalal H et al. Science 2018 Sep 20. doi: 10.1126/science.aau1184.
FROM SCIENCE
Guideline offers comprehensive approach for ICU clinicians
A new (PADIS).
The guideline builds upon the 2013 Clinical Practice Guidelines for the Management of Pain, Agitation, and Delirium (PAD) in Adult Patients in the ICU. Given the comprehensive nature of the PADIS guideline, an accompanying commentary was published simultaneously to help with implementation and interpretation. Both papers are the result of a large-scale, multicenter collaboration and were published in Critical Care Medicine.
A panel of 32 international experts, four methodologists, and four survivors of critical illness used the Grading of Recommendations Assessment, Development and Evaluation approach to develop the PADIS guideline.
“Thousands of hours were invested by these guidelines’ authors, who were in turn were supported by formal and informal collaborators, over the 3.5 years it took to produce this effort,” reported lead author John W. Devlin, PharmD, of the department of pharmacy and health systems sciences, Bouvé College of Health Sciences at Northeastern University, Boston, and his colleagues.
Compared with the 2013 PAD guideline, the PADIS guideline includes new sections regarding rehabilitation/mobility and sleep. “We sought to clarify conceptual definitions within these relatively new critical care research domains,” the panel wrote. “The recommendation rationales, fueled by debate and discussion, circled back to the bedside experience – and the perspective of what was best for patients – held by all panelists and methodology experts.”
The result is extensive and comprehensive, consisting of both broad and specific descriptions of current ICU practices and associated evidence; the guideline includes 37 recommendations, 32 ungraded, nonactionable statements, and two good practice statements. Of note, conditional recommendations far outnumber strong recommendations (34 vs. 3). Reasons for conditional rather than strong recommendations are discussed in rationale sections within the guideline and in the accompanying paper.
“Although our goal was to provide specific recommendations for each question, we suspect some guideline readers may be discouraged by the conditional nature of many recommendations and daunted by the breadth of topics discussed,” wrote Michele C. Balas, PhD, of the Ohio State University College of Nursing in Columbus, and her colleagues. Dr. Balas was on the guideline panel and is the lead author of the accompanying article intended to facilitate implementation and interpretation.
One of the more challenging recommendations surrounds the use of antipsychotics for delirious patients. Although this intervention has become relatively common, the guideline stands against it.
“It should be emphasized that there are few supportive data on ICU antipsychotic use and that the initiation of psychoactive medications during critical illness often results in their inappropriate continuation after ICU discharge,” wrote Dr. Balas and her coauthors.
Along with a hard look at existing practices, the panel actively sought to expand upon the 2013 guideline with new interventions. Discussions ranged from the less conventional, such as aromatherapy, to the more established, such as polypharmacy. Questions, recommendations, and rationale are clearly described for each topic, with clear supporting evidence. Where evidence is missing, the panel recommends future research possibilities.
“One example is the consideration of multiple pharmacologic and nonpharmacologic coanalgesic approaches to the ICU patient,” wrote Dr. Devlin and his coauthors. “When the published evidence was insufficient, limited to a narrow population or specific intervention (e.g., for procedural analgesia), or outright absent to answer the questions we posed, we structured evidence gap descriptors to inform clinicians where the uncertainty lay, and intended to provide sufficient information to apprise and invite researchers to address these gaps.”
The authors disclosed funding from AstraZeneca, Baxter, Covidien, and others.
SOURCE: Devlin JW et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003299; Balas MC et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003307.
A new (PADIS).
The guideline builds upon the 2013 Clinical Practice Guidelines for the Management of Pain, Agitation, and Delirium (PAD) in Adult Patients in the ICU. Given the comprehensive nature of the PADIS guideline, an accompanying commentary was published simultaneously to help with implementation and interpretation. Both papers are the result of a large-scale, multicenter collaboration and were published in Critical Care Medicine.
A panel of 32 international experts, four methodologists, and four survivors of critical illness used the Grading of Recommendations Assessment, Development and Evaluation approach to develop the PADIS guideline.
“Thousands of hours were invested by these guidelines’ authors, who were in turn were supported by formal and informal collaborators, over the 3.5 years it took to produce this effort,” reported lead author John W. Devlin, PharmD, of the department of pharmacy and health systems sciences, Bouvé College of Health Sciences at Northeastern University, Boston, and his colleagues.
Compared with the 2013 PAD guideline, the PADIS guideline includes new sections regarding rehabilitation/mobility and sleep. “We sought to clarify conceptual definitions within these relatively new critical care research domains,” the panel wrote. “The recommendation rationales, fueled by debate and discussion, circled back to the bedside experience – and the perspective of what was best for patients – held by all panelists and methodology experts.”
The result is extensive and comprehensive, consisting of both broad and specific descriptions of current ICU practices and associated evidence; the guideline includes 37 recommendations, 32 ungraded, nonactionable statements, and two good practice statements. Of note, conditional recommendations far outnumber strong recommendations (34 vs. 3). Reasons for conditional rather than strong recommendations are discussed in rationale sections within the guideline and in the accompanying paper.
“Although our goal was to provide specific recommendations for each question, we suspect some guideline readers may be discouraged by the conditional nature of many recommendations and daunted by the breadth of topics discussed,” wrote Michele C. Balas, PhD, of the Ohio State University College of Nursing in Columbus, and her colleagues. Dr. Balas was on the guideline panel and is the lead author of the accompanying article intended to facilitate implementation and interpretation.
One of the more challenging recommendations surrounds the use of antipsychotics for delirious patients. Although this intervention has become relatively common, the guideline stands against it.
“It should be emphasized that there are few supportive data on ICU antipsychotic use and that the initiation of psychoactive medications during critical illness often results in their inappropriate continuation after ICU discharge,” wrote Dr. Balas and her coauthors.
Along with a hard look at existing practices, the panel actively sought to expand upon the 2013 guideline with new interventions. Discussions ranged from the less conventional, such as aromatherapy, to the more established, such as polypharmacy. Questions, recommendations, and rationale are clearly described for each topic, with clear supporting evidence. Where evidence is missing, the panel recommends future research possibilities.
“One example is the consideration of multiple pharmacologic and nonpharmacologic coanalgesic approaches to the ICU patient,” wrote Dr. Devlin and his coauthors. “When the published evidence was insufficient, limited to a narrow population or specific intervention (e.g., for procedural analgesia), or outright absent to answer the questions we posed, we structured evidence gap descriptors to inform clinicians where the uncertainty lay, and intended to provide sufficient information to apprise and invite researchers to address these gaps.”
The authors disclosed funding from AstraZeneca, Baxter, Covidien, and others.
SOURCE: Devlin JW et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003299; Balas MC et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003307.
A new (PADIS).
The guideline builds upon the 2013 Clinical Practice Guidelines for the Management of Pain, Agitation, and Delirium (PAD) in Adult Patients in the ICU. Given the comprehensive nature of the PADIS guideline, an accompanying commentary was published simultaneously to help with implementation and interpretation. Both papers are the result of a large-scale, multicenter collaboration and were published in Critical Care Medicine.
A panel of 32 international experts, four methodologists, and four survivors of critical illness used the Grading of Recommendations Assessment, Development and Evaluation approach to develop the PADIS guideline.
“Thousands of hours were invested by these guidelines’ authors, who were in turn were supported by formal and informal collaborators, over the 3.5 years it took to produce this effort,” reported lead author John W. Devlin, PharmD, of the department of pharmacy and health systems sciences, Bouvé College of Health Sciences at Northeastern University, Boston, and his colleagues.
Compared with the 2013 PAD guideline, the PADIS guideline includes new sections regarding rehabilitation/mobility and sleep. “We sought to clarify conceptual definitions within these relatively new critical care research domains,” the panel wrote. “The recommendation rationales, fueled by debate and discussion, circled back to the bedside experience – and the perspective of what was best for patients – held by all panelists and methodology experts.”
The result is extensive and comprehensive, consisting of both broad and specific descriptions of current ICU practices and associated evidence; the guideline includes 37 recommendations, 32 ungraded, nonactionable statements, and two good practice statements. Of note, conditional recommendations far outnumber strong recommendations (34 vs. 3). Reasons for conditional rather than strong recommendations are discussed in rationale sections within the guideline and in the accompanying paper.
“Although our goal was to provide specific recommendations for each question, we suspect some guideline readers may be discouraged by the conditional nature of many recommendations and daunted by the breadth of topics discussed,” wrote Michele C. Balas, PhD, of the Ohio State University College of Nursing in Columbus, and her colleagues. Dr. Balas was on the guideline panel and is the lead author of the accompanying article intended to facilitate implementation and interpretation.
One of the more challenging recommendations surrounds the use of antipsychotics for delirious patients. Although this intervention has become relatively common, the guideline stands against it.
“It should be emphasized that there are few supportive data on ICU antipsychotic use and that the initiation of psychoactive medications during critical illness often results in their inappropriate continuation after ICU discharge,” wrote Dr. Balas and her coauthors.
Along with a hard look at existing practices, the panel actively sought to expand upon the 2013 guideline with new interventions. Discussions ranged from the less conventional, such as aromatherapy, to the more established, such as polypharmacy. Questions, recommendations, and rationale are clearly described for each topic, with clear supporting evidence. Where evidence is missing, the panel recommends future research possibilities.
“One example is the consideration of multiple pharmacologic and nonpharmacologic coanalgesic approaches to the ICU patient,” wrote Dr. Devlin and his coauthors. “When the published evidence was insufficient, limited to a narrow population or specific intervention (e.g., for procedural analgesia), or outright absent to answer the questions we posed, we structured evidence gap descriptors to inform clinicians where the uncertainty lay, and intended to provide sufficient information to apprise and invite researchers to address these gaps.”
The authors disclosed funding from AstraZeneca, Baxter, Covidien, and others.
SOURCE: Devlin JW et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003299; Balas MC et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003307.
FROM CRITICAL CARE MEDICINE
Key clinical point: The 2018 PADIS guideline recommends intervention strategies for adult ICU patients with pain, agitation/sedation, delirium, immobility, and sleep disruption.
Major finding: The guideline includes 37 recommendations; 32 ungraded, nonactionable statements; and two good practice statements.
Study details: A clinical practice guideline was created by 32 international experts, four methodologists, and four survivors of critical illness.
Disclosures: The authors declared funding from AstraZeneca, Baxter, Covidien, and others.
Sources: Devlin JW et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003299; Balas MC et al. Crit Care Med. 2018 Sep 1. doi: 10.1097/CCM.0000000000003307.