User login
Labor & Delivery: An overlooked entry point for the spread of viral infection
OB hospitalists have a key role to play
A novel coronavirus originating in Wuhan, China, has killed more than 2,800 people and infected more than 81,000 individuals globally. Public health officials around the world and in the United States are working together to contain the outbreak.
There are 57 confirmed cases in the United States, including 18 people evacuated from the Diamond Princess, a cruise ship docked in Yokohama, Japan.1 But the focus on coronavirus, even in early months of the epidemic, serves as an opportunity to revisit the spread of viral disease in hospital settings.
Multiple points of viral entry
In truth, most hospitals are well prepared for the coronavirus, starting with the same place they prepare for most infectious disease epidemics – the emergency department. Patients who seek treatment for early onset symptoms may start with their primary care physicians, but increasing numbers of patients with respiratory concerns and/or infection-related symptoms will first seek medical attention in an emergency care setting.2
Many experts have acknowledged the ED as a viral point of entry, including the American College of Emergency Physicians (ACEP), which produced an excellent guide for management of influenza that details prevention, diagnoses, and treatment protocols in an ED setting.3
But another important, and often forgotten, point of entry in a hospital setting is the obstetrical (OB) Labor & Delivery (L&D) department. Although triage for most patients begins in the main ED, in almost every hospital in the United States, women who present with pregnancy-related issues are sent directly to and triaged in L&D, where – when the proper protocols are not in place – they may transmit viral infection to others.
Pregnancy imparts higher risk
“High risk” is often associated with older, immune-compromised adults. But pregnant women who may appear “healthy” are actually in a state that a 2015 study calls “immunosuppressed” whereby the “… pregnant woman actually undergoes an immunological transformation, where the immune system is necessary to promote and support the pregnancy and growing fetus.”4 Pregnant women, or women with newborns or babies, are at higher risk when exposed to viral infection, with a higher mortality risk than the general population.5 In the best cases, women who contract viral infections are treated carefully and recover fully. In the worst cases, they end up on ventilators and can even die as a result.
Although we are still learning about the Wuhan coronavirus, we already know it is a respiratory illness with a lot of the same characteristics as the influenza virus, and that it is transmitted through droplets (such as a sneeze) or via bodily secretions. Given the extreme vulnerability and physician exposure of women giving birth – in which not one, but two lives are involved – viruses like coronavirus can pose extreme risk. What’s more, public health researchers are still learning about potential transmission of coronavirus from mothers to babies. In the international cases of infant exposure to coronavirus, the newborn showed symptoms within 36 hours of being born, but it is unclear if exposure happened in utero or was vertical transmission after birth.6
Role of OB hospitalists in identifying risk and treating viral infection
Regardless of the type of virus, OB hospitalists are key to screening for viral exposure and care for women, fetuses, and newborns. Given their 24/7 presence and experience with women in L&D, they must champion protocols and precautions that align with those in an ED.
For coronavirus, if a woman presents in L&D with a cough, difficulty breathing, or signs of pneumonia, clinicians should be accustomed to asking about travel to China within the last 14 days and whether the patient has been around someone who has recently traveled to China. If the answer to either question is yes, the woman needs to be immediately placed in a single patient room at negative pressure relative to the surrounding areas, with a minimum of six air changes per hour.
Diagnostic testing should immediately follow. The U.S. Food and Drug Administration just issued Emergency Use Authorization (EUA) for the first commercially-available coronavirus diagnostic test, allowing the use of the test at any lab across the country qualified by the Centers for Disease Control and Prevention.7
If exposure is suspected, containment is paramount until definitive results of diagnostic testing are received. The CDC recommends “Standard Precautions,” which assume that every person is potentially infected or colonized with a pathogen that could be transmitted in the health care setting. These precautions include hand hygiene and personal protective equipment (PPE) to ensure health care workers are not exposed.8
In short, protocols in L&D should mirror those of the ED. But in L&D, clinicians and staff haven’t necessarily been trained to look for or ask for these conditions. Hospitalists can educate their peers and colleagues and advocate for changes at the administrative level.
Biggest current threat: The flu
The coronavirus may eventually present a threat in the United States, but as yet, it is a largely unrealized one. From the perspective of an obstetrician, more immediately concerning is the risk of other viral infections. Although viruses like Ebola and Zika capture headlines, influenza remains the most serious threat to pregnant women in the United States.
According to an article by my colleague, Dr. Mark Simon, “pregnant women and their unborn babies are especially vulnerable to influenza and are more likely to develop serious complications from it … pregnant women who develop the flu are more likely to give birth to children with birth defects of the brain and spine.”9
As of Feb. 1, 2020, the CDC estimates there have been at least 22 million flu illnesses, 210,000 hospitalizations, and 12,000 deaths from flu in the 2019-2020 flu season.10 But the CDC data also suggest that only 54% of pregnant women were vaccinated for influenza in 2019 before or during their pregnancy.11 Hospitalists should ensure that patients diagnosed with flu are quickly and safely treated with antivirals at all stages of their pregnancy to keep them and their babies safe, as well as keep others safe from infection.
Hospitalists can also advocate for across-the-board protocols for the spread of viral illness. The same protocols that protect us from the flu will also protect against coronavirus and viruses that will emerge in the future. Foremost, pregnant women, regardless of trimester, need to receive a flu shot. Women who are pregnant and receive a flu shot can pass on immunity in vitro, and nursing mothers can deliver immunizing agents in their breast milk to their newborn.
Given that hospitalists serve in roles as patient-facing physicians, we should be doing more to protect the public from viral spread, whether coronavirus, influenza, or whatever new viruses the future may hold.
Dr. Dimino is a board-certified ob.gyn. and a Houston-based OB hospitalist with Ob Hospitalist Group. She serves as a faculty member of the TexasAIM Plus Obstetric Hemorrhage Learning Collaborative and currently serves on the Texas Medical Association Council of Science and Public Health.
References
1. The New York Times. Tracking the Coronavirus Map: Tracking the Spread of the Outbreak. Accessed Feb 24, 2020.
2. Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. Accessed Feb 10, 2020.
3. Influenza Emergency Department Best Practices. ACEP Public Health & Injury Prevention Committee, Epidemic Expert Panel, https://www.acep.org/globalassets/uploads/uploaded-files/acep/by-medical-focus/influenza-emergency-department-best-practices.pdf.
4. Silasi M, Cardenas I, Kwon JY, Racicot K, Aldo P, Mor G. Viral infections during pregnancy. Am J Reprod Immunol. 2015;73(3):199-213.
5. Kwon JY, Romero R, Mor G. New insights into the relationship between viral infection and pregnancy complications. Am J Reprod Immunol. 2014;71:387-390.
6. BBC. Coronavirus: Newborn becomes youngest person diagnosed with virus. Accessed Feb 10, 2020.
7. FDA press release. FDA Takes Significant Step in Coronavirus Response Efforts, Issues Emergency Use Authorization for the First 2019 Novel Coronavirus Diagnostic. Feb 4, 2020.
8. CDC. Interim Infection Prevention and Control Recommendations for Patients with Confirmed 2019 Novel Coronavirus (2019-nCoV) or Persons Under Investigation for 2019-nCoV in Healthcare Settings. Accessed Feb 10, 2020.
9. STAT First Opinion. Two-thirds of pregnant women aren’t getting the flu vaccine. That needs to change. Jan 18, 2018.
10. CDC. Weekly U.S. Influenza Surveillance Report, Key Updates for Week 5, ending February 1, 2020.
11. CDC. Vaccinating Pregnant Women Protects Moms and Babies. Accessed Feb 10, 2020.
OB hospitalists have a key role to play
OB hospitalists have a key role to play
A novel coronavirus originating in Wuhan, China, has killed more than 2,800 people and infected more than 81,000 individuals globally. Public health officials around the world and in the United States are working together to contain the outbreak.
There are 57 confirmed cases in the United States, including 18 people evacuated from the Diamond Princess, a cruise ship docked in Yokohama, Japan.1 But the focus on coronavirus, even in early months of the epidemic, serves as an opportunity to revisit the spread of viral disease in hospital settings.
Multiple points of viral entry
In truth, most hospitals are well prepared for the coronavirus, starting with the same place they prepare for most infectious disease epidemics – the emergency department. Patients who seek treatment for early onset symptoms may start with their primary care physicians, but increasing numbers of patients with respiratory concerns and/or infection-related symptoms will first seek medical attention in an emergency care setting.2
Many experts have acknowledged the ED as a viral point of entry, including the American College of Emergency Physicians (ACEP), which produced an excellent guide for management of influenza that details prevention, diagnoses, and treatment protocols in an ED setting.3
But another important, and often forgotten, point of entry in a hospital setting is the obstetrical (OB) Labor & Delivery (L&D) department. Although triage for most patients begins in the main ED, in almost every hospital in the United States, women who present with pregnancy-related issues are sent directly to and triaged in L&D, where – when the proper protocols are not in place – they may transmit viral infection to others.
Pregnancy imparts higher risk
“High risk” is often associated with older, immune-compromised adults. But pregnant women who may appear “healthy” are actually in a state that a 2015 study calls “immunosuppressed” whereby the “… pregnant woman actually undergoes an immunological transformation, where the immune system is necessary to promote and support the pregnancy and growing fetus.”4 Pregnant women, or women with newborns or babies, are at higher risk when exposed to viral infection, with a higher mortality risk than the general population.5 In the best cases, women who contract viral infections are treated carefully and recover fully. In the worst cases, they end up on ventilators and can even die as a result.
Although we are still learning about the Wuhan coronavirus, we already know it is a respiratory illness with a lot of the same characteristics as the influenza virus, and that it is transmitted through droplets (such as a sneeze) or via bodily secretions. Given the extreme vulnerability and physician exposure of women giving birth – in which not one, but two lives are involved – viruses like coronavirus can pose extreme risk. What’s more, public health researchers are still learning about potential transmission of coronavirus from mothers to babies. In the international cases of infant exposure to coronavirus, the newborn showed symptoms within 36 hours of being born, but it is unclear if exposure happened in utero or was vertical transmission after birth.6
Role of OB hospitalists in identifying risk and treating viral infection
Regardless of the type of virus, OB hospitalists are key to screening for viral exposure and care for women, fetuses, and newborns. Given their 24/7 presence and experience with women in L&D, they must champion protocols and precautions that align with those in an ED.
For coronavirus, if a woman presents in L&D with a cough, difficulty breathing, or signs of pneumonia, clinicians should be accustomed to asking about travel to China within the last 14 days and whether the patient has been around someone who has recently traveled to China. If the answer to either question is yes, the woman needs to be immediately placed in a single patient room at negative pressure relative to the surrounding areas, with a minimum of six air changes per hour.
Diagnostic testing should immediately follow. The U.S. Food and Drug Administration just issued Emergency Use Authorization (EUA) for the first commercially-available coronavirus diagnostic test, allowing the use of the test at any lab across the country qualified by the Centers for Disease Control and Prevention.7
If exposure is suspected, containment is paramount until definitive results of diagnostic testing are received. The CDC recommends “Standard Precautions,” which assume that every person is potentially infected or colonized with a pathogen that could be transmitted in the health care setting. These precautions include hand hygiene and personal protective equipment (PPE) to ensure health care workers are not exposed.8
In short, protocols in L&D should mirror those of the ED. But in L&D, clinicians and staff haven’t necessarily been trained to look for or ask for these conditions. Hospitalists can educate their peers and colleagues and advocate for changes at the administrative level.
Biggest current threat: The flu
The coronavirus may eventually present a threat in the United States, but as yet, it is a largely unrealized one. From the perspective of an obstetrician, more immediately concerning is the risk of other viral infections. Although viruses like Ebola and Zika capture headlines, influenza remains the most serious threat to pregnant women in the United States.
According to an article by my colleague, Dr. Mark Simon, “pregnant women and their unborn babies are especially vulnerable to influenza and are more likely to develop serious complications from it … pregnant women who develop the flu are more likely to give birth to children with birth defects of the brain and spine.”9
As of Feb. 1, 2020, the CDC estimates there have been at least 22 million flu illnesses, 210,000 hospitalizations, and 12,000 deaths from flu in the 2019-2020 flu season.10 But the CDC data also suggest that only 54% of pregnant women were vaccinated for influenza in 2019 before or during their pregnancy.11 Hospitalists should ensure that patients diagnosed with flu are quickly and safely treated with antivirals at all stages of their pregnancy to keep them and their babies safe, as well as keep others safe from infection.
Hospitalists can also advocate for across-the-board protocols for the spread of viral illness. The same protocols that protect us from the flu will also protect against coronavirus and viruses that will emerge in the future. Foremost, pregnant women, regardless of trimester, need to receive a flu shot. Women who are pregnant and receive a flu shot can pass on immunity in vitro, and nursing mothers can deliver immunizing agents in their breast milk to their newborn.
Given that hospitalists serve in roles as patient-facing physicians, we should be doing more to protect the public from viral spread, whether coronavirus, influenza, or whatever new viruses the future may hold.
Dr. Dimino is a board-certified ob.gyn. and a Houston-based OB hospitalist with Ob Hospitalist Group. She serves as a faculty member of the TexasAIM Plus Obstetric Hemorrhage Learning Collaborative and currently serves on the Texas Medical Association Council of Science and Public Health.
References
1. The New York Times. Tracking the Coronavirus Map: Tracking the Spread of the Outbreak. Accessed Feb 24, 2020.
2. Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. Accessed Feb 10, 2020.
3. Influenza Emergency Department Best Practices. ACEP Public Health & Injury Prevention Committee, Epidemic Expert Panel, https://www.acep.org/globalassets/uploads/uploaded-files/acep/by-medical-focus/influenza-emergency-department-best-practices.pdf.
4. Silasi M, Cardenas I, Kwon JY, Racicot K, Aldo P, Mor G. Viral infections during pregnancy. Am J Reprod Immunol. 2015;73(3):199-213.
5. Kwon JY, Romero R, Mor G. New insights into the relationship between viral infection and pregnancy complications. Am J Reprod Immunol. 2014;71:387-390.
6. BBC. Coronavirus: Newborn becomes youngest person diagnosed with virus. Accessed Feb 10, 2020.
7. FDA press release. FDA Takes Significant Step in Coronavirus Response Efforts, Issues Emergency Use Authorization for the First 2019 Novel Coronavirus Diagnostic. Feb 4, 2020.
8. CDC. Interim Infection Prevention and Control Recommendations for Patients with Confirmed 2019 Novel Coronavirus (2019-nCoV) or Persons Under Investigation for 2019-nCoV in Healthcare Settings. Accessed Feb 10, 2020.
9. STAT First Opinion. Two-thirds of pregnant women aren’t getting the flu vaccine. That needs to change. Jan 18, 2018.
10. CDC. Weekly U.S. Influenza Surveillance Report, Key Updates for Week 5, ending February 1, 2020.
11. CDC. Vaccinating Pregnant Women Protects Moms and Babies. Accessed Feb 10, 2020.
A novel coronavirus originating in Wuhan, China, has killed more than 2,800 people and infected more than 81,000 individuals globally. Public health officials around the world and in the United States are working together to contain the outbreak.
There are 57 confirmed cases in the United States, including 18 people evacuated from the Diamond Princess, a cruise ship docked in Yokohama, Japan.1 But the focus on coronavirus, even in early months of the epidemic, serves as an opportunity to revisit the spread of viral disease in hospital settings.
Multiple points of viral entry
In truth, most hospitals are well prepared for the coronavirus, starting with the same place they prepare for most infectious disease epidemics – the emergency department. Patients who seek treatment for early onset symptoms may start with their primary care physicians, but increasing numbers of patients with respiratory concerns and/or infection-related symptoms will first seek medical attention in an emergency care setting.2
Many experts have acknowledged the ED as a viral point of entry, including the American College of Emergency Physicians (ACEP), which produced an excellent guide for management of influenza that details prevention, diagnoses, and treatment protocols in an ED setting.3
But another important, and often forgotten, point of entry in a hospital setting is the obstetrical (OB) Labor & Delivery (L&D) department. Although triage for most patients begins in the main ED, in almost every hospital in the United States, women who present with pregnancy-related issues are sent directly to and triaged in L&D, where – when the proper protocols are not in place – they may transmit viral infection to others.
Pregnancy imparts higher risk
“High risk” is often associated with older, immune-compromised adults. But pregnant women who may appear “healthy” are actually in a state that a 2015 study calls “immunosuppressed” whereby the “… pregnant woman actually undergoes an immunological transformation, where the immune system is necessary to promote and support the pregnancy and growing fetus.”4 Pregnant women, or women with newborns or babies, are at higher risk when exposed to viral infection, with a higher mortality risk than the general population.5 In the best cases, women who contract viral infections are treated carefully and recover fully. In the worst cases, they end up on ventilators and can even die as a result.
Although we are still learning about the Wuhan coronavirus, we already know it is a respiratory illness with a lot of the same characteristics as the influenza virus, and that it is transmitted through droplets (such as a sneeze) or via bodily secretions. Given the extreme vulnerability and physician exposure of women giving birth – in which not one, but two lives are involved – viruses like coronavirus can pose extreme risk. What’s more, public health researchers are still learning about potential transmission of coronavirus from mothers to babies. In the international cases of infant exposure to coronavirus, the newborn showed symptoms within 36 hours of being born, but it is unclear if exposure happened in utero or was vertical transmission after birth.6
Role of OB hospitalists in identifying risk and treating viral infection
Regardless of the type of virus, OB hospitalists are key to screening for viral exposure and care for women, fetuses, and newborns. Given their 24/7 presence and experience with women in L&D, they must champion protocols and precautions that align with those in an ED.
For coronavirus, if a woman presents in L&D with a cough, difficulty breathing, or signs of pneumonia, clinicians should be accustomed to asking about travel to China within the last 14 days and whether the patient has been around someone who has recently traveled to China. If the answer to either question is yes, the woman needs to be immediately placed in a single patient room at negative pressure relative to the surrounding areas, with a minimum of six air changes per hour.
Diagnostic testing should immediately follow. The U.S. Food and Drug Administration just issued Emergency Use Authorization (EUA) for the first commercially-available coronavirus diagnostic test, allowing the use of the test at any lab across the country qualified by the Centers for Disease Control and Prevention.7
If exposure is suspected, containment is paramount until definitive results of diagnostic testing are received. The CDC recommends “Standard Precautions,” which assume that every person is potentially infected or colonized with a pathogen that could be transmitted in the health care setting. These precautions include hand hygiene and personal protective equipment (PPE) to ensure health care workers are not exposed.8
In short, protocols in L&D should mirror those of the ED. But in L&D, clinicians and staff haven’t necessarily been trained to look for or ask for these conditions. Hospitalists can educate their peers and colleagues and advocate for changes at the administrative level.
Biggest current threat: The flu
The coronavirus may eventually present a threat in the United States, but as yet, it is a largely unrealized one. From the perspective of an obstetrician, more immediately concerning is the risk of other viral infections. Although viruses like Ebola and Zika capture headlines, influenza remains the most serious threat to pregnant women in the United States.
According to an article by my colleague, Dr. Mark Simon, “pregnant women and their unborn babies are especially vulnerable to influenza and are more likely to develop serious complications from it … pregnant women who develop the flu are more likely to give birth to children with birth defects of the brain and spine.”9
As of Feb. 1, 2020, the CDC estimates there have been at least 22 million flu illnesses, 210,000 hospitalizations, and 12,000 deaths from flu in the 2019-2020 flu season.10 But the CDC data also suggest that only 54% of pregnant women were vaccinated for influenza in 2019 before or during their pregnancy.11 Hospitalists should ensure that patients diagnosed with flu are quickly and safely treated with antivirals at all stages of their pregnancy to keep them and their babies safe, as well as keep others safe from infection.
Hospitalists can also advocate for across-the-board protocols for the spread of viral illness. The same protocols that protect us from the flu will also protect against coronavirus and viruses that will emerge in the future. Foremost, pregnant women, regardless of trimester, need to receive a flu shot. Women who are pregnant and receive a flu shot can pass on immunity in vitro, and nursing mothers can deliver immunizing agents in their breast milk to their newborn.
Given that hospitalists serve in roles as patient-facing physicians, we should be doing more to protect the public from viral spread, whether coronavirus, influenza, or whatever new viruses the future may hold.
Dr. Dimino is a board-certified ob.gyn. and a Houston-based OB hospitalist with Ob Hospitalist Group. She serves as a faculty member of the TexasAIM Plus Obstetric Hemorrhage Learning Collaborative and currently serves on the Texas Medical Association Council of Science and Public Health.
References
1. The New York Times. Tracking the Coronavirus Map: Tracking the Spread of the Outbreak. Accessed Feb 24, 2020.
2. Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. Accessed Feb 10, 2020.
3. Influenza Emergency Department Best Practices. ACEP Public Health & Injury Prevention Committee, Epidemic Expert Panel, https://www.acep.org/globalassets/uploads/uploaded-files/acep/by-medical-focus/influenza-emergency-department-best-practices.pdf.
4. Silasi M, Cardenas I, Kwon JY, Racicot K, Aldo P, Mor G. Viral infections during pregnancy. Am J Reprod Immunol. 2015;73(3):199-213.
5. Kwon JY, Romero R, Mor G. New insights into the relationship between viral infection and pregnancy complications. Am J Reprod Immunol. 2014;71:387-390.
6. BBC. Coronavirus: Newborn becomes youngest person diagnosed with virus. Accessed Feb 10, 2020.
7. FDA press release. FDA Takes Significant Step in Coronavirus Response Efforts, Issues Emergency Use Authorization for the First 2019 Novel Coronavirus Diagnostic. Feb 4, 2020.
8. CDC. Interim Infection Prevention and Control Recommendations for Patients with Confirmed 2019 Novel Coronavirus (2019-nCoV) or Persons Under Investigation for 2019-nCoV in Healthcare Settings. Accessed Feb 10, 2020.
9. STAT First Opinion. Two-thirds of pregnant women aren’t getting the flu vaccine. That needs to change. Jan 18, 2018.
10. CDC. Weekly U.S. Influenza Surveillance Report, Key Updates for Week 5, ending February 1, 2020.
11. CDC. Vaccinating Pregnant Women Protects Moms and Babies. Accessed Feb 10, 2020.
Pence named COVID-19 point person as CDC reports possible community spread
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
AI algorithm finds diagnostic AFib signatures in normal ECGs
NATIONAL HARBOR, MD. – Researchers have created an artificial intelligence algorithm that can evaluate a 10-second ECG recording of a person in normal sinus rhythm and tell with a sensitivity and specificity of almost 80% whether or not that person ever had atrial fibrillation episodes some time in the past or will have a first arrhythmia episode in the near future.
Although this algorithm – derived from and then validated with a dataset of nearly 650,000 ECG recordings from more than 180,000 patients – still needs prospective validation, it offers the prospect for a potential revolution in screening for atrial fibrillation (AFib), Paul A. Friedman, MD, cautioned at the annual International AF Symposium. If initial clinical findings are confirmed, it would show that a 10-second, 12-lead ECG recording can provide the same screening scope as what otherwise takes weeks of ambulatory ECG recording with a Holter monitor or an implanted device, explained Dr. Friedman, professor of medicine and chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
This finding “could have important implications for atrial fibrillation screening and for the management of patients with unexplained stroke,” Dr. Friedman and his associates noted in the published report of their study (Lancet. 2019 Sep 7;394[10201]:861-7). “We’re still working to define the window of ECG” recording time that provides the optimal assessment for a history of asymptomatic AFib, but the “possibilities this opens are huge,” Dr. Friedman said in his talk at the symposium. This work sprang from the premise that “subtle signatures” in a brief, apparently normal sinus rhythm ECG tracing can harbor reliable clues about AFib history or an imminent episode.
The 2019 report by Dr. Friedman and associates documented that in the validation phase of their study, the trained artificial intelligence (AI) program identified patients with a history of AFib or an impending arrhythmia event from a single, 10-second ECG that to the naked eye seemed to show normal sinus rhythm with a sensitivity of 79.0%, a specificity of 79.5%, and an accuracy of 79.4%. It also showed an area under a receiver operating characteristic curve of 0.87, meaning that screening for AFib by this method compared favorably with the area-under-the-curve (AUC) results tallied by several widely accepted screening tools, including Pap smears for cervical cancer (AUC of 0.70), mammograms for breast cancer (AUC of 0.85), and CHA2DS2-VASc scoring for estimating stroke risk in AFib patients (AUC of 0.57-0.72), Dr. Friedman said.
The researchers developed the AI algorithm with more than 450,000 10-second ECG tracings collected from roughly 126,000 patients who underwent at least one ECG recording as part of their routine care at the Mayo Clinic during 1993-2017. The goal was for the program to find and validate recurring characteristics in the ECG that consistently linked with a history of or an impending AFib episode and that did not appear in ECG recordings from people without any AFib history. The program this effort produced then underwent further adjustment with the use of more than 64,340 ECGs from an additional 18,116 patients, and then the final product underwent validation testing with a further 130,802 ECGs collected from an additional 36,280 people, the study phase that resulted in the reported sensitivity and specificity estimates.
It’s currently unclear to Dr. Friedman and associates what specific features the program uses to classify patients. It’s an important question, but if the results are reproducible and reliable, this uncertainty shouldn’t slow clinical adoption, he said in an interview.
While “this particular algorithm needs prospective vetting,” a similar algorithm developed by Dr. Friedman and the same research team that uses a 10-second ECG to identify patients with a left ventricular ejection fraction of 35% or less is further advanced in development, and a device that uses this algorithm will soon receive Food and Drug Administration review under a fast track designation that the agency approved in late 2019.
The researchers developed this algorithm for estimating left ventricular function using a strategy similar to their development of a tool for diagnosing AFib (Nat Med. 2019 Jan 7;25[1]:70-4), and results from 100 patients prospectively studied with this approach to ECG analysis and reported at the American Heart Association scientific sessions in November 2019 showed that the algorithm identified substantial left ventricular dysfunction with an AUC of 0.906 (Circulation. 2019 Nov 19;140[suppl 1]:A13447). The same team of investigators has developed an AI algorithm that can calculate a person’s physiologic age based on the ECG recording (Circ Arrhythm Electrophysiol. 2019 Sep;12[9]: 10.1161/CIRCEP.119.007284).
The study received no commercial funding, and Dr. Friedman and coauthors had no relevant disclosures. The Mayo Clinic has licensed a related artificial intelligence algorithm to EKO, and Dr. Friedman may benefit financially from this arrangement.
NATIONAL HARBOR, MD. – Researchers have created an artificial intelligence algorithm that can evaluate a 10-second ECG recording of a person in normal sinus rhythm and tell with a sensitivity and specificity of almost 80% whether or not that person ever had atrial fibrillation episodes some time in the past or will have a first arrhythmia episode in the near future.
Although this algorithm – derived from and then validated with a dataset of nearly 650,000 ECG recordings from more than 180,000 patients – still needs prospective validation, it offers the prospect for a potential revolution in screening for atrial fibrillation (AFib), Paul A. Friedman, MD, cautioned at the annual International AF Symposium. If initial clinical findings are confirmed, it would show that a 10-second, 12-lead ECG recording can provide the same screening scope as what otherwise takes weeks of ambulatory ECG recording with a Holter monitor or an implanted device, explained Dr. Friedman, professor of medicine and chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
This finding “could have important implications for atrial fibrillation screening and for the management of patients with unexplained stroke,” Dr. Friedman and his associates noted in the published report of their study (Lancet. 2019 Sep 7;394[10201]:861-7). “We’re still working to define the window of ECG” recording time that provides the optimal assessment for a history of asymptomatic AFib, but the “possibilities this opens are huge,” Dr. Friedman said in his talk at the symposium. This work sprang from the premise that “subtle signatures” in a brief, apparently normal sinus rhythm ECG tracing can harbor reliable clues about AFib history or an imminent episode.
The 2019 report by Dr. Friedman and associates documented that in the validation phase of their study, the trained artificial intelligence (AI) program identified patients with a history of AFib or an impending arrhythmia event from a single, 10-second ECG that to the naked eye seemed to show normal sinus rhythm with a sensitivity of 79.0%, a specificity of 79.5%, and an accuracy of 79.4%. It also showed an area under a receiver operating characteristic curve of 0.87, meaning that screening for AFib by this method compared favorably with the area-under-the-curve (AUC) results tallied by several widely accepted screening tools, including Pap smears for cervical cancer (AUC of 0.70), mammograms for breast cancer (AUC of 0.85), and CHA2DS2-VASc scoring for estimating stroke risk in AFib patients (AUC of 0.57-0.72), Dr. Friedman said.
The researchers developed the AI algorithm with more than 450,000 10-second ECG tracings collected from roughly 126,000 patients who underwent at least one ECG recording as part of their routine care at the Mayo Clinic during 1993-2017. The goal was for the program to find and validate recurring characteristics in the ECG that consistently linked with a history of or an impending AFib episode and that did not appear in ECG recordings from people without any AFib history. The program this effort produced then underwent further adjustment with the use of more than 64,340 ECGs from an additional 18,116 patients, and then the final product underwent validation testing with a further 130,802 ECGs collected from an additional 36,280 people, the study phase that resulted in the reported sensitivity and specificity estimates.
It’s currently unclear to Dr. Friedman and associates what specific features the program uses to classify patients. It’s an important question, but if the results are reproducible and reliable, this uncertainty shouldn’t slow clinical adoption, he said in an interview.
While “this particular algorithm needs prospective vetting,” a similar algorithm developed by Dr. Friedman and the same research team that uses a 10-second ECG to identify patients with a left ventricular ejection fraction of 35% or less is further advanced in development, and a device that uses this algorithm will soon receive Food and Drug Administration review under a fast track designation that the agency approved in late 2019.
The researchers developed this algorithm for estimating left ventricular function using a strategy similar to their development of a tool for diagnosing AFib (Nat Med. 2019 Jan 7;25[1]:70-4), and results from 100 patients prospectively studied with this approach to ECG analysis and reported at the American Heart Association scientific sessions in November 2019 showed that the algorithm identified substantial left ventricular dysfunction with an AUC of 0.906 (Circulation. 2019 Nov 19;140[suppl 1]:A13447). The same team of investigators has developed an AI algorithm that can calculate a person’s physiologic age based on the ECG recording (Circ Arrhythm Electrophysiol. 2019 Sep;12[9]: 10.1161/CIRCEP.119.007284).
The study received no commercial funding, and Dr. Friedman and coauthors had no relevant disclosures. The Mayo Clinic has licensed a related artificial intelligence algorithm to EKO, and Dr. Friedman may benefit financially from this arrangement.
NATIONAL HARBOR, MD. – Researchers have created an artificial intelligence algorithm that can evaluate a 10-second ECG recording of a person in normal sinus rhythm and tell with a sensitivity and specificity of almost 80% whether or not that person ever had atrial fibrillation episodes some time in the past or will have a first arrhythmia episode in the near future.
Although this algorithm – derived from and then validated with a dataset of nearly 650,000 ECG recordings from more than 180,000 patients – still needs prospective validation, it offers the prospect for a potential revolution in screening for atrial fibrillation (AFib), Paul A. Friedman, MD, cautioned at the annual International AF Symposium. If initial clinical findings are confirmed, it would show that a 10-second, 12-lead ECG recording can provide the same screening scope as what otherwise takes weeks of ambulatory ECG recording with a Holter monitor or an implanted device, explained Dr. Friedman, professor of medicine and chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
This finding “could have important implications for atrial fibrillation screening and for the management of patients with unexplained stroke,” Dr. Friedman and his associates noted in the published report of their study (Lancet. 2019 Sep 7;394[10201]:861-7). “We’re still working to define the window of ECG” recording time that provides the optimal assessment for a history of asymptomatic AFib, but the “possibilities this opens are huge,” Dr. Friedman said in his talk at the symposium. This work sprang from the premise that “subtle signatures” in a brief, apparently normal sinus rhythm ECG tracing can harbor reliable clues about AFib history or an imminent episode.
The 2019 report by Dr. Friedman and associates documented that in the validation phase of their study, the trained artificial intelligence (AI) program identified patients with a history of AFib or an impending arrhythmia event from a single, 10-second ECG that to the naked eye seemed to show normal sinus rhythm with a sensitivity of 79.0%, a specificity of 79.5%, and an accuracy of 79.4%. It also showed an area under a receiver operating characteristic curve of 0.87, meaning that screening for AFib by this method compared favorably with the area-under-the-curve (AUC) results tallied by several widely accepted screening tools, including Pap smears for cervical cancer (AUC of 0.70), mammograms for breast cancer (AUC of 0.85), and CHA2DS2-VASc scoring for estimating stroke risk in AFib patients (AUC of 0.57-0.72), Dr. Friedman said.
The researchers developed the AI algorithm with more than 450,000 10-second ECG tracings collected from roughly 126,000 patients who underwent at least one ECG recording as part of their routine care at the Mayo Clinic during 1993-2017. The goal was for the program to find and validate recurring characteristics in the ECG that consistently linked with a history of or an impending AFib episode and that did not appear in ECG recordings from people without any AFib history. The program this effort produced then underwent further adjustment with the use of more than 64,340 ECGs from an additional 18,116 patients, and then the final product underwent validation testing with a further 130,802 ECGs collected from an additional 36,280 people, the study phase that resulted in the reported sensitivity and specificity estimates.
It’s currently unclear to Dr. Friedman and associates what specific features the program uses to classify patients. It’s an important question, but if the results are reproducible and reliable, this uncertainty shouldn’t slow clinical adoption, he said in an interview.
While “this particular algorithm needs prospective vetting,” a similar algorithm developed by Dr. Friedman and the same research team that uses a 10-second ECG to identify patients with a left ventricular ejection fraction of 35% or less is further advanced in development, and a device that uses this algorithm will soon receive Food and Drug Administration review under a fast track designation that the agency approved in late 2019.
The researchers developed this algorithm for estimating left ventricular function using a strategy similar to their development of a tool for diagnosing AFib (Nat Med. 2019 Jan 7;25[1]:70-4), and results from 100 patients prospectively studied with this approach to ECG analysis and reported at the American Heart Association scientific sessions in November 2019 showed that the algorithm identified substantial left ventricular dysfunction with an AUC of 0.906 (Circulation. 2019 Nov 19;140[suppl 1]:A13447). The same team of investigators has developed an AI algorithm that can calculate a person’s physiologic age based on the ECG recording (Circ Arrhythm Electrophysiol. 2019 Sep;12[9]: 10.1161/CIRCEP.119.007284).
The study received no commercial funding, and Dr. Friedman and coauthors had no relevant disclosures. The Mayo Clinic has licensed a related artificial intelligence algorithm to EKO, and Dr. Friedman may benefit financially from this arrangement.
THE AF SYMPOSIUM 2020
COVID-19: Time to ‘take the risk of scaring people’
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
CDC expects eventual community spread of coronavirus in U.S.
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
Guidance defines vaping-related respiratory syndrome
ORLANDO – Knowledge of vaping devices, familiarity with terminology, and the ability to quickly pinpoint individuals at risk of lung injury are just a few skills that can help critical care professionals confronted with patients who may have vaping-associated lung disease, according to a new guidance document.
The guidance offers a risk-stratification system that classifies patients into groups based on exposure, symptoms, and imaging results, and provides specific evaluation needs and management strategies for each. The guidance is designed to help critical care professionals efficiently identify those at high risk of respiratory failure.
Physicians also need to communicate with patients to identify what substances are being vaped and develop effective methods to encourage abstinence, according to the authors, led by Craig M. Lilly, MD, FCCP, professor of medicine, anesthesiology, and surgery at the University of Massachusetts, Worcester.
“I would encourage every intensivist, when they leave their intensive care unit at night, [to ask], ‘have I advised against vaping today?’ ” Dr. Lilly said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
The guidelines, concurrently published as a review article in Critical Care Explorations, propose the term vaping-associated respiratory distress syndrome (VARDS), which the authors say constitutes an acute and progressive respiratory syndrome marked by pathologic changes of lung injury and potentially life-threatening hypoxemic respiratory failure.
They also introduce the three-group Worcester classification system, which is intended to triage vaping-exposed individuals for risk of VARDS based on the presence or absence of vaping-related symptoms and infiltrates, and normal or abnormal oxygen saturation.
“It’s very simple,” said Dr. Lilly, who added that the risk stratification model was developed at the request of Massachusetts public health officials.
Patients with vaping exposure but no symptoms attributable to vaping, such as cough, chest pain, or weight loss, are classified as Worcester Low Risk and testing is not recommended, he said.
By contrast, individuals are considered Worcester Medium Risk if they have vaping exposure, symptoms, and a vaping-associated abnormal pattern on imaging, but no hypoxemia; the presence of hypoxemia would tip the scale toward Worcester High Risk.
“Most patients that have died from vaping have been sent out of emergency rooms when they were noted to be hypoxic,” Dr. Lilly told meeting attendees.
Louella B. Amos, MD, a pediatric pulmonologist at Children’s Hospital of Wisconsin in Milwaukee, said she expects the guidance and risk stratification system will be useful not only for critical care specialists, but for other health care providers as well.
“It’s important to make decisions relatively quickly, depending on the severity of symptoms, and I think this is nice and simple,” Dr. Amos said in an interview.
“We always triage when we see patients, either at the door or in our clinic, or behind that, even in the hospital,” she said. “So I think this can be a great tool for everybody, not only the intensivist, but people who are triaging at the front.”
Management of individuals at low risk of VARDS begins with encouragement of abstinence. “We think that every vaping patient should be advised to quit vaping,” Dr. Lilly said. Patients who are interested in quitting who have not yet worked with someone in their health care team whom they trust can be referred to their primary care physicians for counseling, he added, while those struggling with addiction, unable to quit, and unable to partner with a primary care physician can be referred to an addiction medicine specialist.
For moderate-risk patients, vaping cessation is “absolutely mandatory,” said Dr. Lilly, who recommended monitoring of vaping abstinence, outpatient evaluation based on imaging studies, and adequate follow-up to ensure symptoms resolve, tests normalize, and daily activities bounce back to baseline levels.
The guidance offers more extensive recommendations for the VARDS high-risk group, including supervised vaping abstinence, continuous pulse oximetry, and early intervention with noninvasive ventilation, and mechanical ventilation if required, Dr. Lilly said.
Judging vaping exposure is challenging, requiring clinicians to have a familiarity with the many different devices that are available.
Beyond device type, he added, it’s important to know the various terms for devices and lingo that patients may use to describe them, what solutions are vaped, whether those solutions are commercially prepared or off the street, the dose the device delivers, and a number of other factors, he said.
Clinical evaluation typically comes down to unexplained cough, chest pain, weight loss, fatigue, or dyspnea, though one other clue is whether there are gastrointestinal symptoms: “The same way that aerosols can go down to the lungs, they also go into the GI tract, and when nausea, vomiting, or cramping abdominal pain is tightly associated with vaping exposure, one should assume that the patient has been toxin exposed,” he explained.
Dr. Lilly said he had no financial relationships to disclose.
ORLANDO – Knowledge of vaping devices, familiarity with terminology, and the ability to quickly pinpoint individuals at risk of lung injury are just a few skills that can help critical care professionals confronted with patients who may have vaping-associated lung disease, according to a new guidance document.
The guidance offers a risk-stratification system that classifies patients into groups based on exposure, symptoms, and imaging results, and provides specific evaluation needs and management strategies for each. The guidance is designed to help critical care professionals efficiently identify those at high risk of respiratory failure.
Physicians also need to communicate with patients to identify what substances are being vaped and develop effective methods to encourage abstinence, according to the authors, led by Craig M. Lilly, MD, FCCP, professor of medicine, anesthesiology, and surgery at the University of Massachusetts, Worcester.
“I would encourage every intensivist, when they leave their intensive care unit at night, [to ask], ‘have I advised against vaping today?’ ” Dr. Lilly said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
The guidelines, concurrently published as a review article in Critical Care Explorations, propose the term vaping-associated respiratory distress syndrome (VARDS), which the authors say constitutes an acute and progressive respiratory syndrome marked by pathologic changes of lung injury and potentially life-threatening hypoxemic respiratory failure.
They also introduce the three-group Worcester classification system, which is intended to triage vaping-exposed individuals for risk of VARDS based on the presence or absence of vaping-related symptoms and infiltrates, and normal or abnormal oxygen saturation.
“It’s very simple,” said Dr. Lilly, who added that the risk stratification model was developed at the request of Massachusetts public health officials.
Patients with vaping exposure but no symptoms attributable to vaping, such as cough, chest pain, or weight loss, are classified as Worcester Low Risk and testing is not recommended, he said.
By contrast, individuals are considered Worcester Medium Risk if they have vaping exposure, symptoms, and a vaping-associated abnormal pattern on imaging, but no hypoxemia; the presence of hypoxemia would tip the scale toward Worcester High Risk.
“Most patients that have died from vaping have been sent out of emergency rooms when they were noted to be hypoxic,” Dr. Lilly told meeting attendees.
Louella B. Amos, MD, a pediatric pulmonologist at Children’s Hospital of Wisconsin in Milwaukee, said she expects the guidance and risk stratification system will be useful not only for critical care specialists, but for other health care providers as well.
“It’s important to make decisions relatively quickly, depending on the severity of symptoms, and I think this is nice and simple,” Dr. Amos said in an interview.
“We always triage when we see patients, either at the door or in our clinic, or behind that, even in the hospital,” she said. “So I think this can be a great tool for everybody, not only the intensivist, but people who are triaging at the front.”
Management of individuals at low risk of VARDS begins with encouragement of abstinence. “We think that every vaping patient should be advised to quit vaping,” Dr. Lilly said. Patients who are interested in quitting who have not yet worked with someone in their health care team whom they trust can be referred to their primary care physicians for counseling, he added, while those struggling with addiction, unable to quit, and unable to partner with a primary care physician can be referred to an addiction medicine specialist.
For moderate-risk patients, vaping cessation is “absolutely mandatory,” said Dr. Lilly, who recommended monitoring of vaping abstinence, outpatient evaluation based on imaging studies, and adequate follow-up to ensure symptoms resolve, tests normalize, and daily activities bounce back to baseline levels.
The guidance offers more extensive recommendations for the VARDS high-risk group, including supervised vaping abstinence, continuous pulse oximetry, and early intervention with noninvasive ventilation, and mechanical ventilation if required, Dr. Lilly said.
Judging vaping exposure is challenging, requiring clinicians to have a familiarity with the many different devices that are available.
Beyond device type, he added, it’s important to know the various terms for devices and lingo that patients may use to describe them, what solutions are vaped, whether those solutions are commercially prepared or off the street, the dose the device delivers, and a number of other factors, he said.
Clinical evaluation typically comes down to unexplained cough, chest pain, weight loss, fatigue, or dyspnea, though one other clue is whether there are gastrointestinal symptoms: “The same way that aerosols can go down to the lungs, they also go into the GI tract, and when nausea, vomiting, or cramping abdominal pain is tightly associated with vaping exposure, one should assume that the patient has been toxin exposed,” he explained.
Dr. Lilly said he had no financial relationships to disclose.
ORLANDO – Knowledge of vaping devices, familiarity with terminology, and the ability to quickly pinpoint individuals at risk of lung injury are just a few skills that can help critical care professionals confronted with patients who may have vaping-associated lung disease, according to a new guidance document.
The guidance offers a risk-stratification system that classifies patients into groups based on exposure, symptoms, and imaging results, and provides specific evaluation needs and management strategies for each. The guidance is designed to help critical care professionals efficiently identify those at high risk of respiratory failure.
Physicians also need to communicate with patients to identify what substances are being vaped and develop effective methods to encourage abstinence, according to the authors, led by Craig M. Lilly, MD, FCCP, professor of medicine, anesthesiology, and surgery at the University of Massachusetts, Worcester.
“I would encourage every intensivist, when they leave their intensive care unit at night, [to ask], ‘have I advised against vaping today?’ ” Dr. Lilly said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
The guidelines, concurrently published as a review article in Critical Care Explorations, propose the term vaping-associated respiratory distress syndrome (VARDS), which the authors say constitutes an acute and progressive respiratory syndrome marked by pathologic changes of lung injury and potentially life-threatening hypoxemic respiratory failure.
They also introduce the three-group Worcester classification system, which is intended to triage vaping-exposed individuals for risk of VARDS based on the presence or absence of vaping-related symptoms and infiltrates, and normal or abnormal oxygen saturation.
“It’s very simple,” said Dr. Lilly, who added that the risk stratification model was developed at the request of Massachusetts public health officials.
Patients with vaping exposure but no symptoms attributable to vaping, such as cough, chest pain, or weight loss, are classified as Worcester Low Risk and testing is not recommended, he said.
By contrast, individuals are considered Worcester Medium Risk if they have vaping exposure, symptoms, and a vaping-associated abnormal pattern on imaging, but no hypoxemia; the presence of hypoxemia would tip the scale toward Worcester High Risk.
“Most patients that have died from vaping have been sent out of emergency rooms when they were noted to be hypoxic,” Dr. Lilly told meeting attendees.
Louella B. Amos, MD, a pediatric pulmonologist at Children’s Hospital of Wisconsin in Milwaukee, said she expects the guidance and risk stratification system will be useful not only for critical care specialists, but for other health care providers as well.
“It’s important to make decisions relatively quickly, depending on the severity of symptoms, and I think this is nice and simple,” Dr. Amos said in an interview.
“We always triage when we see patients, either at the door or in our clinic, or behind that, even in the hospital,” she said. “So I think this can be a great tool for everybody, not only the intensivist, but people who are triaging at the front.”
Management of individuals at low risk of VARDS begins with encouragement of abstinence. “We think that every vaping patient should be advised to quit vaping,” Dr. Lilly said. Patients who are interested in quitting who have not yet worked with someone in their health care team whom they trust can be referred to their primary care physicians for counseling, he added, while those struggling with addiction, unable to quit, and unable to partner with a primary care physician can be referred to an addiction medicine specialist.
For moderate-risk patients, vaping cessation is “absolutely mandatory,” said Dr. Lilly, who recommended monitoring of vaping abstinence, outpatient evaluation based on imaging studies, and adequate follow-up to ensure symptoms resolve, tests normalize, and daily activities bounce back to baseline levels.
The guidance offers more extensive recommendations for the VARDS high-risk group, including supervised vaping abstinence, continuous pulse oximetry, and early intervention with noninvasive ventilation, and mechanical ventilation if required, Dr. Lilly said.
Judging vaping exposure is challenging, requiring clinicians to have a familiarity with the many different devices that are available.
Beyond device type, he added, it’s important to know the various terms for devices and lingo that patients may use to describe them, what solutions are vaped, whether those solutions are commercially prepared or off the street, the dose the device delivers, and a number of other factors, he said.
Clinical evaluation typically comes down to unexplained cough, chest pain, weight loss, fatigue, or dyspnea, though one other clue is whether there are gastrointestinal symptoms: “The same way that aerosols can go down to the lungs, they also go into the GI tract, and when nausea, vomiting, or cramping abdominal pain is tightly associated with vaping exposure, one should assume that the patient has been toxin exposed,” he explained.
Dr. Lilly said he had no financial relationships to disclose.
REPORTING FROM CCC49
Medicare beneficiaries get few home health visits after ICU stay
ORLANDO – , an analysis of hospital and home health claims data suggests.
The beneficiaries, all discharged directly to home health after an intensive care unit stay, received an average of less than one visit per week in the ensuing month, while a full third received no visits at all, according to authors of the analysis, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
Living alone and living in a rural area were associated with significantly fewer home health rehabilitation visits, according to investigator Jason Raymond Falvey, PT, DPT, PhD, of Yale University, New Haven, Conn.
“We identified that these patients are receiving low doses of rehabilitation in home health care settings, and perhaps doses low enough to not be physiologically adequate to overcome the deconditioning and aerobic capacity concerns that these patients have,” Dr. Falvey said.
These findings reflect an “underrecognition” of the importance of rehabilitation both outside and inside the hospital setting, according to Patricia J. Posa, RN, of Saint Joseph Mercy Hospital, Northville, Mich.
“We even struggle to provide sufficient rehabilitation while they’re in the hospital,” Ms. Posa said in an interview. “So I think that we still have a major gap in providing rehab services across the continuum, and part of that is recognizing the deficits that patients, especially our elderly patients, might be leaving the hospital with.”
Medicare beneficiaries who survive a critical illness are often discharged with referrals for physical, occupational, or speech therapy, yet there are not much data on the delivery of that care or how many visits actually take place, according to Dr. Falvey.
He and coinvestigators analyzed data on 3,176 Medicare beneficiaries discharged to home health right after an acute hospitalization with an ICU stay of at least 24 hours. To do this, they linked 2012 Medicare hospital and home health claims data with Medicare demographic and patient assessment data.
They found that the beneficiaries received just 3.5 home rehabilitation visits in 30 days, while 33% had no visits on record.
The factors most strongly associated with receiving fewer rehabilitation visits, in adjusted models, included living in a rural setting, with a rate ratio (RR) of 0.87 and living alone, with an RR of 0.88.
Higher comorbidity count also was associated with fewer visits (RR, 0.98), according to the investigators.
On the other hand, Medicare beneficiaries who received more visits were more likely to be older (RR, 1.03; 1.01-1.04; for every 5 years), more likely to have higher disability scores (RR, 1.03; 1.02-1.04; per point on the Elixhauser Comorbidity Index), and more likely to have reported severe dyspnea (RR, 1.12; 1.04-1.21), according to the report.
More research will be needed to determine the appropriate number of home health rehabilitation visits for older hospitalized patients, according to Ms. Pena, a member of the Society of Critical Care Medicine’s ICU Liberation initiative, which aims to free patients from the harmful effects of pain, agitation/sedation, delirium, immobility, and sleep disruption in the ICU, as well as improve patient outcomes after an ICU stay.
The literature is already fairly robust, she said, on how frequently visits are warranted following specific scenarios such as postsurgical hip or knee replacement or stroke.
“For the general hospitalized patients that are just losing function because they were sick and didn’t get out of bed enough, we don’t really have good data to say, ‘you know, they need three visits a week, or they need two visits a week for an hour in order to improve,’ ” she said, “so the science is still not caught up with the frequency.”
In the absence of data, the number of visits may be left up to an individual clinician’s knowledge and past experience as well as what insurance will pay for, Ms. Pena said.
Dr. Falvey reported royalties related to an online continuing education course on hospital readmissions. No other disclosures were reported.
SOURCE: Falvey J et al. Crit Care Med. 2020 Jan;48(1):28.
ORLANDO – , an analysis of hospital and home health claims data suggests.
The beneficiaries, all discharged directly to home health after an intensive care unit stay, received an average of less than one visit per week in the ensuing month, while a full third received no visits at all, according to authors of the analysis, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
Living alone and living in a rural area were associated with significantly fewer home health rehabilitation visits, according to investigator Jason Raymond Falvey, PT, DPT, PhD, of Yale University, New Haven, Conn.
“We identified that these patients are receiving low doses of rehabilitation in home health care settings, and perhaps doses low enough to not be physiologically adequate to overcome the deconditioning and aerobic capacity concerns that these patients have,” Dr. Falvey said.
These findings reflect an “underrecognition” of the importance of rehabilitation both outside and inside the hospital setting, according to Patricia J. Posa, RN, of Saint Joseph Mercy Hospital, Northville, Mich.
“We even struggle to provide sufficient rehabilitation while they’re in the hospital,” Ms. Posa said in an interview. “So I think that we still have a major gap in providing rehab services across the continuum, and part of that is recognizing the deficits that patients, especially our elderly patients, might be leaving the hospital with.”
Medicare beneficiaries who survive a critical illness are often discharged with referrals for physical, occupational, or speech therapy, yet there are not much data on the delivery of that care or how many visits actually take place, according to Dr. Falvey.
He and coinvestigators analyzed data on 3,176 Medicare beneficiaries discharged to home health right after an acute hospitalization with an ICU stay of at least 24 hours. To do this, they linked 2012 Medicare hospital and home health claims data with Medicare demographic and patient assessment data.
They found that the beneficiaries received just 3.5 home rehabilitation visits in 30 days, while 33% had no visits on record.
The factors most strongly associated with receiving fewer rehabilitation visits, in adjusted models, included living in a rural setting, with a rate ratio (RR) of 0.87 and living alone, with an RR of 0.88.
Higher comorbidity count also was associated with fewer visits (RR, 0.98), according to the investigators.
On the other hand, Medicare beneficiaries who received more visits were more likely to be older (RR, 1.03; 1.01-1.04; for every 5 years), more likely to have higher disability scores (RR, 1.03; 1.02-1.04; per point on the Elixhauser Comorbidity Index), and more likely to have reported severe dyspnea (RR, 1.12; 1.04-1.21), according to the report.
More research will be needed to determine the appropriate number of home health rehabilitation visits for older hospitalized patients, according to Ms. Pena, a member of the Society of Critical Care Medicine’s ICU Liberation initiative, which aims to free patients from the harmful effects of pain, agitation/sedation, delirium, immobility, and sleep disruption in the ICU, as well as improve patient outcomes after an ICU stay.
The literature is already fairly robust, she said, on how frequently visits are warranted following specific scenarios such as postsurgical hip or knee replacement or stroke.
“For the general hospitalized patients that are just losing function because they were sick and didn’t get out of bed enough, we don’t really have good data to say, ‘you know, they need three visits a week, or they need two visits a week for an hour in order to improve,’ ” she said, “so the science is still not caught up with the frequency.”
In the absence of data, the number of visits may be left up to an individual clinician’s knowledge and past experience as well as what insurance will pay for, Ms. Pena said.
Dr. Falvey reported royalties related to an online continuing education course on hospital readmissions. No other disclosures were reported.
SOURCE: Falvey J et al. Crit Care Med. 2020 Jan;48(1):28.
ORLANDO – , an analysis of hospital and home health claims data suggests.
The beneficiaries, all discharged directly to home health after an intensive care unit stay, received an average of less than one visit per week in the ensuing month, while a full third received no visits at all, according to authors of the analysis, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
Living alone and living in a rural area were associated with significantly fewer home health rehabilitation visits, according to investigator Jason Raymond Falvey, PT, DPT, PhD, of Yale University, New Haven, Conn.
“We identified that these patients are receiving low doses of rehabilitation in home health care settings, and perhaps doses low enough to not be physiologically adequate to overcome the deconditioning and aerobic capacity concerns that these patients have,” Dr. Falvey said.
These findings reflect an “underrecognition” of the importance of rehabilitation both outside and inside the hospital setting, according to Patricia J. Posa, RN, of Saint Joseph Mercy Hospital, Northville, Mich.
“We even struggle to provide sufficient rehabilitation while they’re in the hospital,” Ms. Posa said in an interview. “So I think that we still have a major gap in providing rehab services across the continuum, and part of that is recognizing the deficits that patients, especially our elderly patients, might be leaving the hospital with.”
Medicare beneficiaries who survive a critical illness are often discharged with referrals for physical, occupational, or speech therapy, yet there are not much data on the delivery of that care or how many visits actually take place, according to Dr. Falvey.
He and coinvestigators analyzed data on 3,176 Medicare beneficiaries discharged to home health right after an acute hospitalization with an ICU stay of at least 24 hours. To do this, they linked 2012 Medicare hospital and home health claims data with Medicare demographic and patient assessment data.
They found that the beneficiaries received just 3.5 home rehabilitation visits in 30 days, while 33% had no visits on record.
The factors most strongly associated with receiving fewer rehabilitation visits, in adjusted models, included living in a rural setting, with a rate ratio (RR) of 0.87 and living alone, with an RR of 0.88.
Higher comorbidity count also was associated with fewer visits (RR, 0.98), according to the investigators.
On the other hand, Medicare beneficiaries who received more visits were more likely to be older (RR, 1.03; 1.01-1.04; for every 5 years), more likely to have higher disability scores (RR, 1.03; 1.02-1.04; per point on the Elixhauser Comorbidity Index), and more likely to have reported severe dyspnea (RR, 1.12; 1.04-1.21), according to the report.
More research will be needed to determine the appropriate number of home health rehabilitation visits for older hospitalized patients, according to Ms. Pena, a member of the Society of Critical Care Medicine’s ICU Liberation initiative, which aims to free patients from the harmful effects of pain, agitation/sedation, delirium, immobility, and sleep disruption in the ICU, as well as improve patient outcomes after an ICU stay.
The literature is already fairly robust, she said, on how frequently visits are warranted following specific scenarios such as postsurgical hip or knee replacement or stroke.
“For the general hospitalized patients that are just losing function because they were sick and didn’t get out of bed enough, we don’t really have good data to say, ‘you know, they need three visits a week, or they need two visits a week for an hour in order to improve,’ ” she said, “so the science is still not caught up with the frequency.”
In the absence of data, the number of visits may be left up to an individual clinician’s knowledge and past experience as well as what insurance will pay for, Ms. Pena said.
Dr. Falvey reported royalties related to an online continuing education course on hospital readmissions. No other disclosures were reported.
SOURCE: Falvey J et al. Crit Care Med. 2020 Jan;48(1):28.
REPORTING FROM CCC49
Opioid use disorder up in sepsis hospitalizations
ORLANDO –
The prevalence of opioid use disorder (OUD) has significantly increased over the past 15 years, the analysis further shows.
Results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, further suggested that OUD disproportionately contributes to sepsis deaths in younger, healthier patients.
Together, these findings underscore the importance of ongoing efforts to address the opioid epidemic in the United States, according to researcher Mohammad Alrawashdeh, PhD, MSN, a postdoctoral research fellow with Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston.
“In addition to ongoing efforts to combat the opioid crisis, future public health interventions should focus on increasing awareness, recognition, and aggressive treatment of sepsis in this population,” Dr. Alrawashdeh said in an oral presentation of the study.
This study fills an important knowledge gap regarding the connection between OUD and sepsis, according to Greg S. Martin, MD, MS, FCCM, professor of medicine in pulmonary critical care at Emory University, Atlanta, and secretary for the Society of Critical Care Medicine.
“We’ve not really ever been able to piece together the relationship between opioid use disorders and sepsis,” Dr. Martin said in an interview. “It’s not that people wouldn’t suspect that there’s a connection – it’s more that we have simply not been able to get the kind of data that you can use, like they’ve done here, that really helps you to answer that question.”
The study suggests not only that OUD and sepsis are linked, Dr. Martin added, but that health care providers need to be prepared to potentially see further increases in the number of patients with OUD seen in the intensive care unit.
“Both of those are things that we certainly need to be aware of, both from the individual practitioner perspective and also the public health planning perspective,” he said.
The retrospective study by Dr. Alrawashdeh and coinvestigators focused on electronic health record data for adults admitted to 373 hospitals in the United States between 2009 and 2015, including 375,479 who had sepsis.
Over time, there was a significant increase in the prevalence of OUD among those hospitalized for sepsis, from less than 2.0% in 2009 to more than 3% in 2015, representing a significant 77.3% increase. In general, the prevalence of sepsis was significantly higher among hospitalized patients with OUD compared with patients without the disorder, at 7.2% and 5.6%, respectively.
The sepsis patients with OUD tended to be younger, healthier, and more likely to be white compared with patients without OUD, according to the report. Moreover, the sepsis patients with OUD more often had endocarditis and gram-positive and fungal bloodstream infections. They also required more mechanical ventilation and had more ICU admissions, with longer stays in both the ICU and hospital.
The OUD patients accounted for 2.1% of sepsis-associated deaths overall, but 3.3% of those deaths in healthy patients, and 7.1% of deaths among younger patients, according to the report.
Those findings provide some clues that could help guide clinical practice, according to Dr. Martin. For example, the data show a nearly fivefold increased risk of endocarditis with OUD (3.9% versus 0.7%), which may inform screening practices.
“While we don’t necessarily screen every sepsis patient for endocarditis, if it’s an opioid use disorder patient – particularly one with a bloodstream infection – then that’s almost certainly something you should be doing,” Dr. Martin said.
The data suggest gram-positive bacterial and fungal infections will more likely be encountered among these patients, which could guide empiric treatment, he said.
Providers specializing in OUD should have a heightened awareness of the potential for infection and sepsis among those patients, and perhaps be more attuned to fever and other signs of infection that might warrant a referral or additional care, Dr. Martin added.
Dr. Alrawashdeh reported no disclosures related to the study.
SOURCE: Alrawashdeh M et al. Crit Care Med. 2020 Jan;48(1):28. Abstract 56.
ORLANDO –
The prevalence of opioid use disorder (OUD) has significantly increased over the past 15 years, the analysis further shows.
Results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, further suggested that OUD disproportionately contributes to sepsis deaths in younger, healthier patients.
Together, these findings underscore the importance of ongoing efforts to address the opioid epidemic in the United States, according to researcher Mohammad Alrawashdeh, PhD, MSN, a postdoctoral research fellow with Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston.
“In addition to ongoing efforts to combat the opioid crisis, future public health interventions should focus on increasing awareness, recognition, and aggressive treatment of sepsis in this population,” Dr. Alrawashdeh said in an oral presentation of the study.
This study fills an important knowledge gap regarding the connection between OUD and sepsis, according to Greg S. Martin, MD, MS, FCCM, professor of medicine in pulmonary critical care at Emory University, Atlanta, and secretary for the Society of Critical Care Medicine.
“We’ve not really ever been able to piece together the relationship between opioid use disorders and sepsis,” Dr. Martin said in an interview. “It’s not that people wouldn’t suspect that there’s a connection – it’s more that we have simply not been able to get the kind of data that you can use, like they’ve done here, that really helps you to answer that question.”
The study suggests not only that OUD and sepsis are linked, Dr. Martin added, but that health care providers need to be prepared to potentially see further increases in the number of patients with OUD seen in the intensive care unit.
“Both of those are things that we certainly need to be aware of, both from the individual practitioner perspective and also the public health planning perspective,” he said.
The retrospective study by Dr. Alrawashdeh and coinvestigators focused on electronic health record data for adults admitted to 373 hospitals in the United States between 2009 and 2015, including 375,479 who had sepsis.
Over time, there was a significant increase in the prevalence of OUD among those hospitalized for sepsis, from less than 2.0% in 2009 to more than 3% in 2015, representing a significant 77.3% increase. In general, the prevalence of sepsis was significantly higher among hospitalized patients with OUD compared with patients without the disorder, at 7.2% and 5.6%, respectively.
The sepsis patients with OUD tended to be younger, healthier, and more likely to be white compared with patients without OUD, according to the report. Moreover, the sepsis patients with OUD more often had endocarditis and gram-positive and fungal bloodstream infections. They also required more mechanical ventilation and had more ICU admissions, with longer stays in both the ICU and hospital.
The OUD patients accounted for 2.1% of sepsis-associated deaths overall, but 3.3% of those deaths in healthy patients, and 7.1% of deaths among younger patients, according to the report.
Those findings provide some clues that could help guide clinical practice, according to Dr. Martin. For example, the data show a nearly fivefold increased risk of endocarditis with OUD (3.9% versus 0.7%), which may inform screening practices.
“While we don’t necessarily screen every sepsis patient for endocarditis, if it’s an opioid use disorder patient – particularly one with a bloodstream infection – then that’s almost certainly something you should be doing,” Dr. Martin said.
The data suggest gram-positive bacterial and fungal infections will more likely be encountered among these patients, which could guide empiric treatment, he said.
Providers specializing in OUD should have a heightened awareness of the potential for infection and sepsis among those patients, and perhaps be more attuned to fever and other signs of infection that might warrant a referral or additional care, Dr. Martin added.
Dr. Alrawashdeh reported no disclosures related to the study.
SOURCE: Alrawashdeh M et al. Crit Care Med. 2020 Jan;48(1):28. Abstract 56.
ORLANDO –
The prevalence of opioid use disorder (OUD) has significantly increased over the past 15 years, the analysis further shows.
Results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, further suggested that OUD disproportionately contributes to sepsis deaths in younger, healthier patients.
Together, these findings underscore the importance of ongoing efforts to address the opioid epidemic in the United States, according to researcher Mohammad Alrawashdeh, PhD, MSN, a postdoctoral research fellow with Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston.
“In addition to ongoing efforts to combat the opioid crisis, future public health interventions should focus on increasing awareness, recognition, and aggressive treatment of sepsis in this population,” Dr. Alrawashdeh said in an oral presentation of the study.
This study fills an important knowledge gap regarding the connection between OUD and sepsis, according to Greg S. Martin, MD, MS, FCCM, professor of medicine in pulmonary critical care at Emory University, Atlanta, and secretary for the Society of Critical Care Medicine.
“We’ve not really ever been able to piece together the relationship between opioid use disorders and sepsis,” Dr. Martin said in an interview. “It’s not that people wouldn’t suspect that there’s a connection – it’s more that we have simply not been able to get the kind of data that you can use, like they’ve done here, that really helps you to answer that question.”
The study suggests not only that OUD and sepsis are linked, Dr. Martin added, but that health care providers need to be prepared to potentially see further increases in the number of patients with OUD seen in the intensive care unit.
“Both of those are things that we certainly need to be aware of, both from the individual practitioner perspective and also the public health planning perspective,” he said.
The retrospective study by Dr. Alrawashdeh and coinvestigators focused on electronic health record data for adults admitted to 373 hospitals in the United States between 2009 and 2015, including 375,479 who had sepsis.
Over time, there was a significant increase in the prevalence of OUD among those hospitalized for sepsis, from less than 2.0% in 2009 to more than 3% in 2015, representing a significant 77.3% increase. In general, the prevalence of sepsis was significantly higher among hospitalized patients with OUD compared with patients without the disorder, at 7.2% and 5.6%, respectively.
The sepsis patients with OUD tended to be younger, healthier, and more likely to be white compared with patients without OUD, according to the report. Moreover, the sepsis patients with OUD more often had endocarditis and gram-positive and fungal bloodstream infections. They also required more mechanical ventilation and had more ICU admissions, with longer stays in both the ICU and hospital.
The OUD patients accounted for 2.1% of sepsis-associated deaths overall, but 3.3% of those deaths in healthy patients, and 7.1% of deaths among younger patients, according to the report.
Those findings provide some clues that could help guide clinical practice, according to Dr. Martin. For example, the data show a nearly fivefold increased risk of endocarditis with OUD (3.9% versus 0.7%), which may inform screening practices.
“While we don’t necessarily screen every sepsis patient for endocarditis, if it’s an opioid use disorder patient – particularly one with a bloodstream infection – then that’s almost certainly something you should be doing,” Dr. Martin said.
The data suggest gram-positive bacterial and fungal infections will more likely be encountered among these patients, which could guide empiric treatment, he said.
Providers specializing in OUD should have a heightened awareness of the potential for infection and sepsis among those patients, and perhaps be more attuned to fever and other signs of infection that might warrant a referral or additional care, Dr. Martin added.
Dr. Alrawashdeh reported no disclosures related to the study.
SOURCE: Alrawashdeh M et al. Crit Care Med. 2020 Jan;48(1):28. Abstract 56.
REPORTING FROM CCC49
Vitamin E acetate found in more vapers’ lung fluid
Analysis of additional lung fluid samples confirms the presence of vitamin E acetate in patients with electronic-cigarette, or vaping, product use–associated lung injury, according to a report on 51 patients in 16 states.
The average age of the patients was 23 years; 69% were male.
The report extends previous work by the Centers for Disease Control and Prevention to test for harmful substances in bronchoalveolar-lavage (BAL) fluid obtained from patients with electronic-cigarette, or vaping, product use–associated lung injury (EVALI) as part of a strategy to understand and manage the recent outbreak of EVALI cases in the United States, wrote Benjamin C. Blount, PhD, of the Division of Laboratory Sciences at the CDC’s National Center for Environmental Health, and colleagues.
“CDC was addressing a serious outbreak of lung injury that was sometimes lethal; but after the first 10 weeks of the outbreak investigation, the cause was still unknown,” Dr. Blount said in an interview. “Possible theories could not be evaluated unless the laboratory could develop tests that could confidently connect exposure to lung injury. Detection of toxicants in bronchoalveolar-lavage fluid from patients with EVALI can provide direct information on exposure within the lung.”
In a study published in the New England Journal of Medicine, the researchers examined the BAL of 51 cases of EVALI from 16 states. They analyzed the samples for multiple toxicants, including vitamin E acetate, plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and diluent terpenes.
Overall, 77% of the patients reported using products containing THC, 67% reported using products containing nicotine, and 51% reported using both types.
Researchers found vitamin E acetate in 48 of the 51 patients (94%); no vitamin E acetate was found in the BAL of healthy controls. Coconut oil and limonene were found in one patient each, but none of the other toxicants was found in the samples from the patients or controls.
In addition, 47 of the 50 patients for whom data were available either had detectable tetrahydrocannabinol (THC) or its metabolites in their BAL fluid samples, or they reported vaping THC products within 90 days before they became ill. Nicotine or its metabolites were found in 30 of 47 patients (64%).
The study findings were limited by several factors, including the potential role of vitamin E acetate as a marker for exposure to other toxicants, the uncertainty of the role of aerosolized constituents formed when vitamin E acetate is heated, and the lack of data on the timing and burden of toxicant exposure, the investigators noted.
As for the next steps in research, “additional studies are needed to examine the respiratory effects of inhaling aerosolized vitamin E acetate and provide information on whether vitamin E acetate in isolation causes lung injury,” Dr. Blount explained. Analysis of the aerosol and gases generated by case-associated product fluids is ongoing.
“When CDC developed the BAL study for this response, we considered several possible toxicants in this investigation to find a possible cause of the outbreak,” Dr. Blount noted. “To accomplish the study, CDC’s Environmental Health Laboratory developed 12 analytical methods and validated them in less than 3 weeks because of the urgent nature of the emergency.”
Dr. Blount said he would advise clinicians to “continue to reference CDC guidance on treating suspected or EVALI patients.” In December, the CDC published updated guidance for clinicians on hospitalized EVALI patients. “Following this guidance and other recommendations could reduce EVALI-associated morbidity and mortality,” Dr. Blount said.
The study was supported in part by the National Cancer Institute, the FDA Center for Tobacco Products, and Ohio State University Pelotonia Intramural Research. The researchers had no financial conflicts to disclose.
SOURCE: Blount BC et al. N Engl J Med. 2020 Feb 20. doi: 10.1056/NEJMoa1916433.
Analysis of additional lung fluid samples confirms the presence of vitamin E acetate in patients with electronic-cigarette, or vaping, product use–associated lung injury, according to a report on 51 patients in 16 states.
The average age of the patients was 23 years; 69% were male.
The report extends previous work by the Centers for Disease Control and Prevention to test for harmful substances in bronchoalveolar-lavage (BAL) fluid obtained from patients with electronic-cigarette, or vaping, product use–associated lung injury (EVALI) as part of a strategy to understand and manage the recent outbreak of EVALI cases in the United States, wrote Benjamin C. Blount, PhD, of the Division of Laboratory Sciences at the CDC’s National Center for Environmental Health, and colleagues.
“CDC was addressing a serious outbreak of lung injury that was sometimes lethal; but after the first 10 weeks of the outbreak investigation, the cause was still unknown,” Dr. Blount said in an interview. “Possible theories could not be evaluated unless the laboratory could develop tests that could confidently connect exposure to lung injury. Detection of toxicants in bronchoalveolar-lavage fluid from patients with EVALI can provide direct information on exposure within the lung.”
In a study published in the New England Journal of Medicine, the researchers examined the BAL of 51 cases of EVALI from 16 states. They analyzed the samples for multiple toxicants, including vitamin E acetate, plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and diluent terpenes.
Overall, 77% of the patients reported using products containing THC, 67% reported using products containing nicotine, and 51% reported using both types.
Researchers found vitamin E acetate in 48 of the 51 patients (94%); no vitamin E acetate was found in the BAL of healthy controls. Coconut oil and limonene were found in one patient each, but none of the other toxicants was found in the samples from the patients or controls.
In addition, 47 of the 50 patients for whom data were available either had detectable tetrahydrocannabinol (THC) or its metabolites in their BAL fluid samples, or they reported vaping THC products within 90 days before they became ill. Nicotine or its metabolites were found in 30 of 47 patients (64%).
The study findings were limited by several factors, including the potential role of vitamin E acetate as a marker for exposure to other toxicants, the uncertainty of the role of aerosolized constituents formed when vitamin E acetate is heated, and the lack of data on the timing and burden of toxicant exposure, the investigators noted.
As for the next steps in research, “additional studies are needed to examine the respiratory effects of inhaling aerosolized vitamin E acetate and provide information on whether vitamin E acetate in isolation causes lung injury,” Dr. Blount explained. Analysis of the aerosol and gases generated by case-associated product fluids is ongoing.
“When CDC developed the BAL study for this response, we considered several possible toxicants in this investigation to find a possible cause of the outbreak,” Dr. Blount noted. “To accomplish the study, CDC’s Environmental Health Laboratory developed 12 analytical methods and validated them in less than 3 weeks because of the urgent nature of the emergency.”
Dr. Blount said he would advise clinicians to “continue to reference CDC guidance on treating suspected or EVALI patients.” In December, the CDC published updated guidance for clinicians on hospitalized EVALI patients. “Following this guidance and other recommendations could reduce EVALI-associated morbidity and mortality,” Dr. Blount said.
The study was supported in part by the National Cancer Institute, the FDA Center for Tobacco Products, and Ohio State University Pelotonia Intramural Research. The researchers had no financial conflicts to disclose.
SOURCE: Blount BC et al. N Engl J Med. 2020 Feb 20. doi: 10.1056/NEJMoa1916433.
Analysis of additional lung fluid samples confirms the presence of vitamin E acetate in patients with electronic-cigarette, or vaping, product use–associated lung injury, according to a report on 51 patients in 16 states.
The average age of the patients was 23 years; 69% were male.
The report extends previous work by the Centers for Disease Control and Prevention to test for harmful substances in bronchoalveolar-lavage (BAL) fluid obtained from patients with electronic-cigarette, or vaping, product use–associated lung injury (EVALI) as part of a strategy to understand and manage the recent outbreak of EVALI cases in the United States, wrote Benjamin C. Blount, PhD, of the Division of Laboratory Sciences at the CDC’s National Center for Environmental Health, and colleagues.
“CDC was addressing a serious outbreak of lung injury that was sometimes lethal; but after the first 10 weeks of the outbreak investigation, the cause was still unknown,” Dr. Blount said in an interview. “Possible theories could not be evaluated unless the laboratory could develop tests that could confidently connect exposure to lung injury. Detection of toxicants in bronchoalveolar-lavage fluid from patients with EVALI can provide direct information on exposure within the lung.”
In a study published in the New England Journal of Medicine, the researchers examined the BAL of 51 cases of EVALI from 16 states. They analyzed the samples for multiple toxicants, including vitamin E acetate, plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and diluent terpenes.
Overall, 77% of the patients reported using products containing THC, 67% reported using products containing nicotine, and 51% reported using both types.
Researchers found vitamin E acetate in 48 of the 51 patients (94%); no vitamin E acetate was found in the BAL of healthy controls. Coconut oil and limonene were found in one patient each, but none of the other toxicants was found in the samples from the patients or controls.
In addition, 47 of the 50 patients for whom data were available either had detectable tetrahydrocannabinol (THC) or its metabolites in their BAL fluid samples, or they reported vaping THC products within 90 days before they became ill. Nicotine or its metabolites were found in 30 of 47 patients (64%).
The study findings were limited by several factors, including the potential role of vitamin E acetate as a marker for exposure to other toxicants, the uncertainty of the role of aerosolized constituents formed when vitamin E acetate is heated, and the lack of data on the timing and burden of toxicant exposure, the investigators noted.
As for the next steps in research, “additional studies are needed to examine the respiratory effects of inhaling aerosolized vitamin E acetate and provide information on whether vitamin E acetate in isolation causes lung injury,” Dr. Blount explained. Analysis of the aerosol and gases generated by case-associated product fluids is ongoing.
“When CDC developed the BAL study for this response, we considered several possible toxicants in this investigation to find a possible cause of the outbreak,” Dr. Blount noted. “To accomplish the study, CDC’s Environmental Health Laboratory developed 12 analytical methods and validated them in less than 3 weeks because of the urgent nature of the emergency.”
Dr. Blount said he would advise clinicians to “continue to reference CDC guidance on treating suspected or EVALI patients.” In December, the CDC published updated guidance for clinicians on hospitalized EVALI patients. “Following this guidance and other recommendations could reduce EVALI-associated morbidity and mortality,” Dr. Blount said.
The study was supported in part by the National Cancer Institute, the FDA Center for Tobacco Products, and Ohio State University Pelotonia Intramural Research. The researchers had no financial conflicts to disclose.
SOURCE: Blount BC et al. N Engl J Med. 2020 Feb 20. doi: 10.1056/NEJMoa1916433.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Drop in flu activity suggests season may have peaked
Influenza activity dropped during the week ending Feb. 15, according to the Centers for Disease Control and Prevention. That decline, along with revised data from the 2 previous weeks, suggests that the 2019-2020 season has peaked for the second time. The rate of outpatient visits for influenza-like illness (ILI) came in at 6.1% for the week ending Feb. 15, after two straight weeks at 6.7%, the CDC’s influenza division reported Feb. 21.
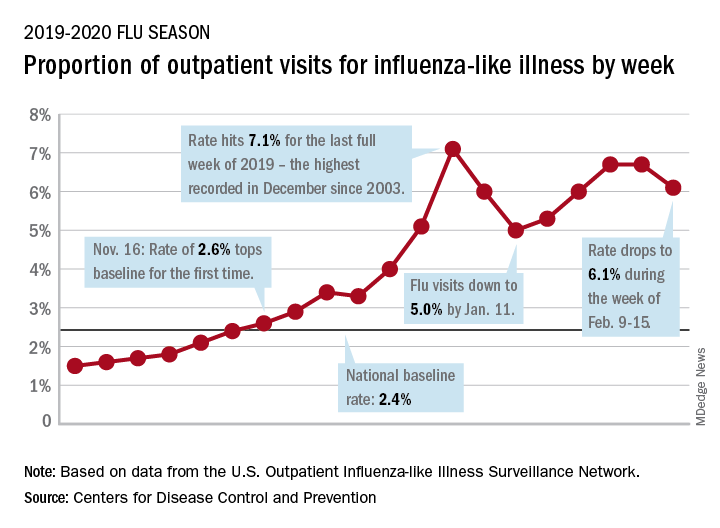
The rates for those 2 earlier weeks had previously been reported at 6.8% (Feb. 8) and 6.6% (Feb. 1), which means that there have now been 2 consecutive weeks without an increase in national ILI activity.
State-level activity was down slightly as well. For the week ending Feb. 15, there were 39 states and Puerto Rico at the highest level of activity on the CDC’s 1-10 scale, compared with 41 states and Puerto Rico the week before. The number of states in the “high” range, which includes levels 8 and 9, went from 44 to 45, however, CDC data show.
Laboratory measures also dropped a bit. For the week, 29.6% of respiratory specimens tested positive for influenza, compared with 30.3% the previous week. The predominance of influenza A continued to increase, as type A went from 59.4% to 63.5% of positive specimens and type B dropped from 40.6% to 36.5%, the influenza division said.
In a separate report, the CDC announced interim flu vaccine effectiveness estimates.For the 2019-2020 season so far, “flu vaccines are reducing doctor’s visits for flu illness by almost half (45%). This is consistent with estimates of flu vaccine effectiveness (VE) from previous flu seasons that ranged from 40% to 60% when flu vaccine viruses were similar to circulating influenza viruses,” the CDC said.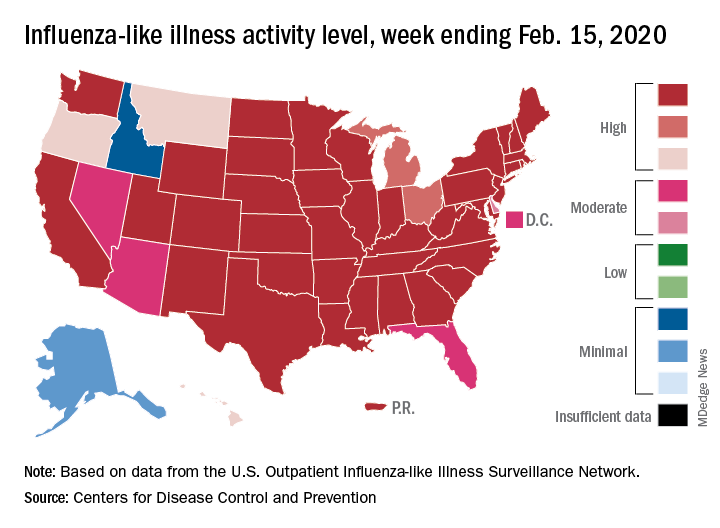
Although VE among children aged 6 months to 17 years is even higher, at 55%, this season “has been especially bad for children. Flu hospitalization rates among children are higher than at this time in other recent seasons, including the 2017-18 season,” the CDC noted.
The number of pediatric flu deaths for 2019-2020 – now up to 105 – is “higher for the same time period than in every season since reporting began in 2004-05, with the exception of the 2009 pandemic,” the CDC added.
Interim VE estimates for other age groups are 25% for adults aged 18-49 and 43% for those 50 years and older. “The lower VE point estimates observed among adults 18-49 years appear to be associated with a trend suggesting lower VE in this age group against A(H1N1)pdm09 viruses,” the CDC said.
Influenza activity dropped during the week ending Feb. 15, according to the Centers for Disease Control and Prevention. That decline, along with revised data from the 2 previous weeks, suggests that the 2019-2020 season has peaked for the second time. The rate of outpatient visits for influenza-like illness (ILI) came in at 6.1% for the week ending Feb. 15, after two straight weeks at 6.7%, the CDC’s influenza division reported Feb. 21.

The rates for those 2 earlier weeks had previously been reported at 6.8% (Feb. 8) and 6.6% (Feb. 1), which means that there have now been 2 consecutive weeks without an increase in national ILI activity.
State-level activity was down slightly as well. For the week ending Feb. 15, there were 39 states and Puerto Rico at the highest level of activity on the CDC’s 1-10 scale, compared with 41 states and Puerto Rico the week before. The number of states in the “high” range, which includes levels 8 and 9, went from 44 to 45, however, CDC data show.
Laboratory measures also dropped a bit. For the week, 29.6% of respiratory specimens tested positive for influenza, compared with 30.3% the previous week. The predominance of influenza A continued to increase, as type A went from 59.4% to 63.5% of positive specimens and type B dropped from 40.6% to 36.5%, the influenza division said.
In a separate report, the CDC announced interim flu vaccine effectiveness estimates.For the 2019-2020 season so far, “flu vaccines are reducing doctor’s visits for flu illness by almost half (45%). This is consistent with estimates of flu vaccine effectiveness (VE) from previous flu seasons that ranged from 40% to 60% when flu vaccine viruses were similar to circulating influenza viruses,” the CDC said.
Although VE among children aged 6 months to 17 years is even higher, at 55%, this season “has been especially bad for children. Flu hospitalization rates among children are higher than at this time in other recent seasons, including the 2017-18 season,” the CDC noted.
The number of pediatric flu deaths for 2019-2020 – now up to 105 – is “higher for the same time period than in every season since reporting began in 2004-05, with the exception of the 2009 pandemic,” the CDC added.
Interim VE estimates for other age groups are 25% for adults aged 18-49 and 43% for those 50 years and older. “The lower VE point estimates observed among adults 18-49 years appear to be associated with a trend suggesting lower VE in this age group against A(H1N1)pdm09 viruses,” the CDC said.
Influenza activity dropped during the week ending Feb. 15, according to the Centers for Disease Control and Prevention. That decline, along with revised data from the 2 previous weeks, suggests that the 2019-2020 season has peaked for the second time. The rate of outpatient visits for influenza-like illness (ILI) came in at 6.1% for the week ending Feb. 15, after two straight weeks at 6.7%, the CDC’s influenza division reported Feb. 21.

The rates for those 2 earlier weeks had previously been reported at 6.8% (Feb. 8) and 6.6% (Feb. 1), which means that there have now been 2 consecutive weeks without an increase in national ILI activity.
State-level activity was down slightly as well. For the week ending Feb. 15, there were 39 states and Puerto Rico at the highest level of activity on the CDC’s 1-10 scale, compared with 41 states and Puerto Rico the week before. The number of states in the “high” range, which includes levels 8 and 9, went from 44 to 45, however, CDC data show.
Laboratory measures also dropped a bit. For the week, 29.6% of respiratory specimens tested positive for influenza, compared with 30.3% the previous week. The predominance of influenza A continued to increase, as type A went from 59.4% to 63.5% of positive specimens and type B dropped from 40.6% to 36.5%, the influenza division said.
In a separate report, the CDC announced interim flu vaccine effectiveness estimates.For the 2019-2020 season so far, “flu vaccines are reducing doctor’s visits for flu illness by almost half (45%). This is consistent with estimates of flu vaccine effectiveness (VE) from previous flu seasons that ranged from 40% to 60% when flu vaccine viruses were similar to circulating influenza viruses,” the CDC said.
Although VE among children aged 6 months to 17 years is even higher, at 55%, this season “has been especially bad for children. Flu hospitalization rates among children are higher than at this time in other recent seasons, including the 2017-18 season,” the CDC noted.
The number of pediatric flu deaths for 2019-2020 – now up to 105 – is “higher for the same time period than in every season since reporting began in 2004-05, with the exception of the 2009 pandemic,” the CDC added.
Interim VE estimates for other age groups are 25% for adults aged 18-49 and 43% for those 50 years and older. “The lower VE point estimates observed among adults 18-49 years appear to be associated with a trend suggesting lower VE in this age group against A(H1N1)pdm09 viruses,” the CDC said.
FROM THE CDC