User login
Batten down the hatches for thyroid storm
Thyroid storm is a life-threatening endocrine emergency for which, remarkably, there are no definitive diagnostic tests, and the management of which is supported by a startlingly weak evidence base.
“What’s tricky is there really are no specific biochemical level cutoffs for thyroid storm, and also no unique laboratory abnormalities. So in the end, it’s a clinical diagnosis and a clinical judgment,” Stephanie B. Mayer, MD, MHSc, observed at HM20 Virtual, hosted by the Society of Hospital Medicine.
Moreover, there are no prospective clinical trials addressing the treatment of thyroid storm, and the 2016 American Thyroid Association clinical practice guidelines on the topic are based upon low-quality evidence from case reports and studies dating back to the 1970s and 1980s. UpToDate reached the same conclusion in 2020, noted Dr. Mayer, an endocrinologist at Virginia Commonwealth University, Richmond.
Thinking that perhaps the guideline writing panel had missed something, she asked a university medical research librarian to custom-build a comprehensive search for studies on thyroid storm management. The search proved unrewarding.
“The evidence is, unfortunately, a little disappointing,” Dr. Mayer said.
Thyroid storm is a rare condition, but one that hospitalists must be ready for. She highlighted current best practices in diagnosis and management.
A high-mortality emergency
Thyroid storm is an extreme manifestation of thyrotoxicosis, which is marked by multiorgan dysfunction and rapid decompensation. In a large, first-of-its-kind, national retrospective U.S. study, the incidence of thyroid storm was 0.57-0.76 cases per 100,000 persons per year. Thyroid storm accounted for 16% of the more than 121,000 hospital discharges featuring a primary diagnosis of thyrotoxicosis. The in-hospital mortality rate for patients with thyroid storm was 1.2%-3.6% during the 10-year study period, a rate 12-fold higher than that among patients with thyrotoxicosis without thyroid storm (Thyroid. 2019 Jan;29[1]:36-43).
Dr. Mayer highlighted a multicenter French study that underscored the current hefty morbidity and mortality associated with thyroid storm. Among 92 patients admitted to the ICU for thyroid storm, the in-ICU mortality rate was 17%, and the mortality rate 6 months after admission was 22%. Independent risk factors for in-ICU mortality were multiorgan failure and the occurrence of cardiogenic shock within the first 48 hours in the ICU (Crit Care Med. 2020 Jan;48[1]:83-90).
How to recognize thyroid storm
The most user-friendly system for assistance in diagnosing thyroid storm is the one put forth by the Japan Thyroid Association and the Japan Endocrine Society, in Dr. Mayer’s view. As a prerequisite to the diagnosis a patient must have thyrotoxicosis as evidenced by elevated free thyroxine (free T4) and free or total triiodothyronine (T3), which in the vast majority of cases, is accompanied by low thyroid stimulating hormone (TSH).
The Japanese diagnostic system for thyroid storm relies on five categories of organ system–based clinical features. This approach places greater weight on disturbances of consciousness – restlessness, delirium, agitation, psychosis, lethargy, coma – than the other four components, which consist of fever of at least 100.4° F, tachycardia of 130 or more beats per minute, heart failure signs and symptoms, and gastrointestinal/hepatic involvement as evidenced by nausea, vomiting, hyperdefecation, and/or a total bilirubin level of 3.0 mg/dL or more.
The Japanese approach offers two paths to a definite diagnosis of thyroid storm. One requires at least one CNS manifestation plus symptoms drawn from any one of the other four categories. The other route, for patients without evident CNS symptoms, requires the presence of symptoms from at least three of the other four categories.
A patient is categorized as having suspected rather than definite thyroid storm if the CNS criterion isn’t met but any two of the others are. A patient also qualifies for suspected thyroid storm when CNS manifestations plus symptoms from at least one other category are present, but thyroid hormone levels aren’t available (Endocr J. 2016 Dec 30;63[12]:1025-64).
Management of thyroid storm
There is usually a precipitating event that drives the transition from smoldering thyrotoxicosis to thyroid storm.
“The big thing is to look for and treat the underlying precipitating event,” the endocrinologist stressed.
It’s often a systemic insult: severe infection, trauma, surgery, an acute MI, diabetic ketoacidosis, pulmonary embolism, or perhaps having just gone through labor. Iodine exposure in the form of IV contrast or taking amiodarone, which contains 37% iodine by weight, can also fan thyrotoxicosis into thyroid storm. Abrupt discontinuation of antithyroid medication is another common cause.
Fluid and electrolyte replacement, oxygen if appropriate, cooling blankets, and other supportive measures are also important.
Medical management targets multiple steps in thyroid hormone production and action to quell thyroid storm. The first order of business is to inhibit synthesis of new thyroid hormone by prescribing a thioamide. Dr. Mayer favors propylthiouracil over methimazole for this purpose because, not only does it block the thyroid gland from synthesizing new hormone, it also reduces conversion of T4 to T3. Propylthiouracil is usually given orally as a 500- to 1,000-mg loading dose, then 250 mg every 4 hours. The drug can also be given rectally or by nasogastric tube.
One hour or more after starting the thioamide, inorganic iodine is started to inhibit release of preformed hormone from the thyroid gland. Five drops of saturated solution of potassium iodide given every 6 hours is the recommended dose; it provides 764 mg of iodide per day. Lugol’s solution dosed at four to eight drops every 6-8 hours is an effective alternative.
Simultaneous with starting the patient on inorganic iodine, a low-dose beta blocker is introduced to control adrenergic symptoms.
“Propranolol is first line because it also decreases T4 to T3 conversion and it’s noncardioselective, so it’s better than a cardioselective beta blocker at reducing sympathetic tone-related symptoms, such as agitation, fever, and psychosis,” the endocrinologist explained.
At the same time that propranolol at 60-80 mg is given orally every 4 hours and iodine are started, the patient is placed on glucocorticoids as another means of reducing peripheral conversion of T4 to T3. The options are intravenous hydrocortisone at 100-300 mg/day in divided doses or dexamethasone at 2 mg every 6 hours.
Aspirin and NSAIDs should be avoided as antipyretics because they can actually raise T3 and T4 levels. Acetaminophen is the right fever-lowering agent in the setting of thyroid storm.
Dr. Mayer has occasionally had to reach for one of several backup therapies. Prescribing a bile acid sequestrant – 20-30 g/day of cholestyramine or colestipol – will trap thyroid hormone in the intestine, preventing it from recirculating.
“Be careful to dose it away from the other medications,” she cautioned.
Also, therapeutic plasmapheresis is effective at rapidly removing circulating thyroid hormone in patients who don’t show early clinical improvement in response to multipronged medical therapy.
Dr. Mayer offered a couple of final tips to hospitalists regarding thyroid storm: Know who directs plasmapheresis at your hospital, and keep the American Thyroid Association management guidelines handy (Thyroid. 2016 Oct;26[10]:1343-421).
She reported receiving funding from both NovoNordisk and Astra Zeneca.
Thyroid storm is a life-threatening endocrine emergency for which, remarkably, there are no definitive diagnostic tests, and the management of which is supported by a startlingly weak evidence base.
“What’s tricky is there really are no specific biochemical level cutoffs for thyroid storm, and also no unique laboratory abnormalities. So in the end, it’s a clinical diagnosis and a clinical judgment,” Stephanie B. Mayer, MD, MHSc, observed at HM20 Virtual, hosted by the Society of Hospital Medicine.
Moreover, there are no prospective clinical trials addressing the treatment of thyroid storm, and the 2016 American Thyroid Association clinical practice guidelines on the topic are based upon low-quality evidence from case reports and studies dating back to the 1970s and 1980s. UpToDate reached the same conclusion in 2020, noted Dr. Mayer, an endocrinologist at Virginia Commonwealth University, Richmond.
Thinking that perhaps the guideline writing panel had missed something, she asked a university medical research librarian to custom-build a comprehensive search for studies on thyroid storm management. The search proved unrewarding.
“The evidence is, unfortunately, a little disappointing,” Dr. Mayer said.
Thyroid storm is a rare condition, but one that hospitalists must be ready for. She highlighted current best practices in diagnosis and management.
A high-mortality emergency
Thyroid storm is an extreme manifestation of thyrotoxicosis, which is marked by multiorgan dysfunction and rapid decompensation. In a large, first-of-its-kind, national retrospective U.S. study, the incidence of thyroid storm was 0.57-0.76 cases per 100,000 persons per year. Thyroid storm accounted for 16% of the more than 121,000 hospital discharges featuring a primary diagnosis of thyrotoxicosis. The in-hospital mortality rate for patients with thyroid storm was 1.2%-3.6% during the 10-year study period, a rate 12-fold higher than that among patients with thyrotoxicosis without thyroid storm (Thyroid. 2019 Jan;29[1]:36-43).
Dr. Mayer highlighted a multicenter French study that underscored the current hefty morbidity and mortality associated with thyroid storm. Among 92 patients admitted to the ICU for thyroid storm, the in-ICU mortality rate was 17%, and the mortality rate 6 months after admission was 22%. Independent risk factors for in-ICU mortality were multiorgan failure and the occurrence of cardiogenic shock within the first 48 hours in the ICU (Crit Care Med. 2020 Jan;48[1]:83-90).
How to recognize thyroid storm
The most user-friendly system for assistance in diagnosing thyroid storm is the one put forth by the Japan Thyroid Association and the Japan Endocrine Society, in Dr. Mayer’s view. As a prerequisite to the diagnosis a patient must have thyrotoxicosis as evidenced by elevated free thyroxine (free T4) and free or total triiodothyronine (T3), which in the vast majority of cases, is accompanied by low thyroid stimulating hormone (TSH).
The Japanese diagnostic system for thyroid storm relies on five categories of organ system–based clinical features. This approach places greater weight on disturbances of consciousness – restlessness, delirium, agitation, psychosis, lethargy, coma – than the other four components, which consist of fever of at least 100.4° F, tachycardia of 130 or more beats per minute, heart failure signs and symptoms, and gastrointestinal/hepatic involvement as evidenced by nausea, vomiting, hyperdefecation, and/or a total bilirubin level of 3.0 mg/dL or more.
The Japanese approach offers two paths to a definite diagnosis of thyroid storm. One requires at least one CNS manifestation plus symptoms drawn from any one of the other four categories. The other route, for patients without evident CNS symptoms, requires the presence of symptoms from at least three of the other four categories.
A patient is categorized as having suspected rather than definite thyroid storm if the CNS criterion isn’t met but any two of the others are. A patient also qualifies for suspected thyroid storm when CNS manifestations plus symptoms from at least one other category are present, but thyroid hormone levels aren’t available (Endocr J. 2016 Dec 30;63[12]:1025-64).
Management of thyroid storm
There is usually a precipitating event that drives the transition from smoldering thyrotoxicosis to thyroid storm.
“The big thing is to look for and treat the underlying precipitating event,” the endocrinologist stressed.
It’s often a systemic insult: severe infection, trauma, surgery, an acute MI, diabetic ketoacidosis, pulmonary embolism, or perhaps having just gone through labor. Iodine exposure in the form of IV contrast or taking amiodarone, which contains 37% iodine by weight, can also fan thyrotoxicosis into thyroid storm. Abrupt discontinuation of antithyroid medication is another common cause.
Fluid and electrolyte replacement, oxygen if appropriate, cooling blankets, and other supportive measures are also important.
Medical management targets multiple steps in thyroid hormone production and action to quell thyroid storm. The first order of business is to inhibit synthesis of new thyroid hormone by prescribing a thioamide. Dr. Mayer favors propylthiouracil over methimazole for this purpose because, not only does it block the thyroid gland from synthesizing new hormone, it also reduces conversion of T4 to T3. Propylthiouracil is usually given orally as a 500- to 1,000-mg loading dose, then 250 mg every 4 hours. The drug can also be given rectally or by nasogastric tube.
One hour or more after starting the thioamide, inorganic iodine is started to inhibit release of preformed hormone from the thyroid gland. Five drops of saturated solution of potassium iodide given every 6 hours is the recommended dose; it provides 764 mg of iodide per day. Lugol’s solution dosed at four to eight drops every 6-8 hours is an effective alternative.
Simultaneous with starting the patient on inorganic iodine, a low-dose beta blocker is introduced to control adrenergic symptoms.
“Propranolol is first line because it also decreases T4 to T3 conversion and it’s noncardioselective, so it’s better than a cardioselective beta blocker at reducing sympathetic tone-related symptoms, such as agitation, fever, and psychosis,” the endocrinologist explained.
At the same time that propranolol at 60-80 mg is given orally every 4 hours and iodine are started, the patient is placed on glucocorticoids as another means of reducing peripheral conversion of T4 to T3. The options are intravenous hydrocortisone at 100-300 mg/day in divided doses or dexamethasone at 2 mg every 6 hours.
Aspirin and NSAIDs should be avoided as antipyretics because they can actually raise T3 and T4 levels. Acetaminophen is the right fever-lowering agent in the setting of thyroid storm.
Dr. Mayer has occasionally had to reach for one of several backup therapies. Prescribing a bile acid sequestrant – 20-30 g/day of cholestyramine or colestipol – will trap thyroid hormone in the intestine, preventing it from recirculating.
“Be careful to dose it away from the other medications,” she cautioned.
Also, therapeutic plasmapheresis is effective at rapidly removing circulating thyroid hormone in patients who don’t show early clinical improvement in response to multipronged medical therapy.
Dr. Mayer offered a couple of final tips to hospitalists regarding thyroid storm: Know who directs plasmapheresis at your hospital, and keep the American Thyroid Association management guidelines handy (Thyroid. 2016 Oct;26[10]:1343-421).
She reported receiving funding from both NovoNordisk and Astra Zeneca.
Thyroid storm is a life-threatening endocrine emergency for which, remarkably, there are no definitive diagnostic tests, and the management of which is supported by a startlingly weak evidence base.
“What’s tricky is there really are no specific biochemical level cutoffs for thyroid storm, and also no unique laboratory abnormalities. So in the end, it’s a clinical diagnosis and a clinical judgment,” Stephanie B. Mayer, MD, MHSc, observed at HM20 Virtual, hosted by the Society of Hospital Medicine.
Moreover, there are no prospective clinical trials addressing the treatment of thyroid storm, and the 2016 American Thyroid Association clinical practice guidelines on the topic are based upon low-quality evidence from case reports and studies dating back to the 1970s and 1980s. UpToDate reached the same conclusion in 2020, noted Dr. Mayer, an endocrinologist at Virginia Commonwealth University, Richmond.
Thinking that perhaps the guideline writing panel had missed something, she asked a university medical research librarian to custom-build a comprehensive search for studies on thyroid storm management. The search proved unrewarding.
“The evidence is, unfortunately, a little disappointing,” Dr. Mayer said.
Thyroid storm is a rare condition, but one that hospitalists must be ready for. She highlighted current best practices in diagnosis and management.
A high-mortality emergency
Thyroid storm is an extreme manifestation of thyrotoxicosis, which is marked by multiorgan dysfunction and rapid decompensation. In a large, first-of-its-kind, national retrospective U.S. study, the incidence of thyroid storm was 0.57-0.76 cases per 100,000 persons per year. Thyroid storm accounted for 16% of the more than 121,000 hospital discharges featuring a primary diagnosis of thyrotoxicosis. The in-hospital mortality rate for patients with thyroid storm was 1.2%-3.6% during the 10-year study period, a rate 12-fold higher than that among patients with thyrotoxicosis without thyroid storm (Thyroid. 2019 Jan;29[1]:36-43).
Dr. Mayer highlighted a multicenter French study that underscored the current hefty morbidity and mortality associated with thyroid storm. Among 92 patients admitted to the ICU for thyroid storm, the in-ICU mortality rate was 17%, and the mortality rate 6 months after admission was 22%. Independent risk factors for in-ICU mortality were multiorgan failure and the occurrence of cardiogenic shock within the first 48 hours in the ICU (Crit Care Med. 2020 Jan;48[1]:83-90).
How to recognize thyroid storm
The most user-friendly system for assistance in diagnosing thyroid storm is the one put forth by the Japan Thyroid Association and the Japan Endocrine Society, in Dr. Mayer’s view. As a prerequisite to the diagnosis a patient must have thyrotoxicosis as evidenced by elevated free thyroxine (free T4) and free or total triiodothyronine (T3), which in the vast majority of cases, is accompanied by low thyroid stimulating hormone (TSH).
The Japanese diagnostic system for thyroid storm relies on five categories of organ system–based clinical features. This approach places greater weight on disturbances of consciousness – restlessness, delirium, agitation, psychosis, lethargy, coma – than the other four components, which consist of fever of at least 100.4° F, tachycardia of 130 or more beats per minute, heart failure signs and symptoms, and gastrointestinal/hepatic involvement as evidenced by nausea, vomiting, hyperdefecation, and/or a total bilirubin level of 3.0 mg/dL or more.
The Japanese approach offers two paths to a definite diagnosis of thyroid storm. One requires at least one CNS manifestation plus symptoms drawn from any one of the other four categories. The other route, for patients without evident CNS symptoms, requires the presence of symptoms from at least three of the other four categories.
A patient is categorized as having suspected rather than definite thyroid storm if the CNS criterion isn’t met but any two of the others are. A patient also qualifies for suspected thyroid storm when CNS manifestations plus symptoms from at least one other category are present, but thyroid hormone levels aren’t available (Endocr J. 2016 Dec 30;63[12]:1025-64).
Management of thyroid storm
There is usually a precipitating event that drives the transition from smoldering thyrotoxicosis to thyroid storm.
“The big thing is to look for and treat the underlying precipitating event,” the endocrinologist stressed.
It’s often a systemic insult: severe infection, trauma, surgery, an acute MI, diabetic ketoacidosis, pulmonary embolism, or perhaps having just gone through labor. Iodine exposure in the form of IV contrast or taking amiodarone, which contains 37% iodine by weight, can also fan thyrotoxicosis into thyroid storm. Abrupt discontinuation of antithyroid medication is another common cause.
Fluid and electrolyte replacement, oxygen if appropriate, cooling blankets, and other supportive measures are also important.
Medical management targets multiple steps in thyroid hormone production and action to quell thyroid storm. The first order of business is to inhibit synthesis of new thyroid hormone by prescribing a thioamide. Dr. Mayer favors propylthiouracil over methimazole for this purpose because, not only does it block the thyroid gland from synthesizing new hormone, it also reduces conversion of T4 to T3. Propylthiouracil is usually given orally as a 500- to 1,000-mg loading dose, then 250 mg every 4 hours. The drug can also be given rectally or by nasogastric tube.
One hour or more after starting the thioamide, inorganic iodine is started to inhibit release of preformed hormone from the thyroid gland. Five drops of saturated solution of potassium iodide given every 6 hours is the recommended dose; it provides 764 mg of iodide per day. Lugol’s solution dosed at four to eight drops every 6-8 hours is an effective alternative.
Simultaneous with starting the patient on inorganic iodine, a low-dose beta blocker is introduced to control adrenergic symptoms.
“Propranolol is first line because it also decreases T4 to T3 conversion and it’s noncardioselective, so it’s better than a cardioselective beta blocker at reducing sympathetic tone-related symptoms, such as agitation, fever, and psychosis,” the endocrinologist explained.
At the same time that propranolol at 60-80 mg is given orally every 4 hours and iodine are started, the patient is placed on glucocorticoids as another means of reducing peripheral conversion of T4 to T3. The options are intravenous hydrocortisone at 100-300 mg/day in divided doses or dexamethasone at 2 mg every 6 hours.
Aspirin and NSAIDs should be avoided as antipyretics because they can actually raise T3 and T4 levels. Acetaminophen is the right fever-lowering agent in the setting of thyroid storm.
Dr. Mayer has occasionally had to reach for one of several backup therapies. Prescribing a bile acid sequestrant – 20-30 g/day of cholestyramine or colestipol – will trap thyroid hormone in the intestine, preventing it from recirculating.
“Be careful to dose it away from the other medications,” she cautioned.
Also, therapeutic plasmapheresis is effective at rapidly removing circulating thyroid hormone in patients who don’t show early clinical improvement in response to multipronged medical therapy.
Dr. Mayer offered a couple of final tips to hospitalists regarding thyroid storm: Know who directs plasmapheresis at your hospital, and keep the American Thyroid Association management guidelines handy (Thyroid. 2016 Oct;26[10]:1343-421).
She reported receiving funding from both NovoNordisk and Astra Zeneca.
FROM HM20 VIRTUAL
Drug allergy in the chart? Ask patients for specifics
Paige Wickner, MD, MPH, medical director for quality and safety at Brigham and Women’s Hospital and assistant professor at Harvard Medical School, both in Boston, described a scenario that might sound familiar to hospitalists.
A 72-year-old man is admitted to the hospital for a lung transplant, and has a listed allergy to “sulfa,” contained in antibiotics and other medications. His medical records say his reaction was “rash.”
What do you do?
The answer, Dr. Wickner said, speaking at HM20 Virtual, sponsored by the Society of Hospital Medicine, is to first ask more questions for clarification. How bad was the rash? Was it blistering? To what type of sulfa did the patient have a reaction?
These questions can help determine the next steps. For sulfa-based antibiotics, hospitalists can often desensitize patients with certain reaction characteristics using widely studied protocols to allow the patient to temporarily take a sulfa-containing medication.
The dominant message of Dr. Wickner’s talk on drug allergies was to get clear details on the allergic reaction –
“Please, for all of your patients, take an allergy history on every listed medication; often you will be able to remove or clarify the medical record and the changes can be life saving,” she said.
For instance, desensitization to sulfa for patients who’ve had a morbilliform rash without a fever can be done on an outpatient basis. But if the patient had hives, or became short of breath or anaphylactic, it needs to be done as an inpatient by an allergist, she said.
The question of drug allergies is substantial. About 35% of patients have at least one listed drug allergy, with penicillin, NSAIDs, and CT contrast agents topping the list, Dr. Wickner said. Although 10% of the general population and 15% of inpatients have a listed penicillin allergy, more than 90% of listed penicillin allergies turn out not to actually be allergic, in part because penicillin allergies are often diagnosed in childhood and are frequently outgrown over time. Having a listed allergy can impact treatment, Dr. Wickner said, with alternatives not always clear-cut.
She described one patient she saw who had 62 listed drug allergies, prompting her clinicians to wonder, “what can I safely give this patient?” Physicians, she said, subject to drug allergy “alert fatigue,” tend to override about 80% of allergy alerts, but this can sometimes have serious consequences.
“The best time to clarify is when a patient is healthy and well,” said Dr. Wickner, not when they are an inpatient and sick. It is much more difficult to test for an allergy, and to treat an allergic reaction, than when someone’s health is quickly declining.
She urged physicians to ask patients to be specific about the name of a drug they suspect they’re allergic to, about the indications, the symptoms, and the timing – an immediate reaction is much different than a symptom that showed up days later.
“Sometimes they’ll say they’re allergic to penicillin, but will tell you they’ve taken Augmentin or amoxicillin, so you can take that allergy off the list,” Dr. Wickner said.
At Brigham and Women’s Hospital and Massachusetts General Hospital, Boston, physicians have developed protocols for assessing and managing suspected allergies to penicillin, aspirin and NSAIDs, and trimethoprim/sulfamethoxazole – helpful tools, she said, because the nature and context of the reaction can matter a great deal in how to respond to the listed allergy.
If someone has a reaction, and you think it might be anaphylaxis, don’t spend time pondering it, Dr. Wickner said. “If that thought crosses your mind, treat it like anaphylaxis, then analyze after the fact.” Most patients with anaphylaxis have some cutaneous sign, even if it’s just flushing.
Dr. Wickner said that, if an allergist is available, take advantage of that. “If allergy is available in-house, utilize them. Often, skin testing and/or challenge can help patients receive first-line therapy.”
In a question-and-answer session after her presentation, Dr. Wickner said that hospitalists “have a huge role to play” in drug allergy delabeling.
“We would love to have a more standard practice of allergy reconciliation, just like we do with medication reconciliation,” she said. Asking questions to get more specifics is essential – and simply asking directly about each listed allergen is “step one, and you’ll really find it’s going to broaden the things that you can do for your patients.”
Asked about whether reactions listed as allergies are frequently just adverse effects, Dr. Wickner said that patients who say they are frequently nauseous – rather than breaking out into a rash, for example – might not be having a true allergic reaction. After careful consideration, they might be better managed with antinausea medication than avoidance of the drug.
Dr. Wickner reported no relevant financial disclosures.
Paige Wickner, MD, MPH, medical director for quality and safety at Brigham and Women’s Hospital and assistant professor at Harvard Medical School, both in Boston, described a scenario that might sound familiar to hospitalists.
A 72-year-old man is admitted to the hospital for a lung transplant, and has a listed allergy to “sulfa,” contained in antibiotics and other medications. His medical records say his reaction was “rash.”
What do you do?
The answer, Dr. Wickner said, speaking at HM20 Virtual, sponsored by the Society of Hospital Medicine, is to first ask more questions for clarification. How bad was the rash? Was it blistering? To what type of sulfa did the patient have a reaction?
These questions can help determine the next steps. For sulfa-based antibiotics, hospitalists can often desensitize patients with certain reaction characteristics using widely studied protocols to allow the patient to temporarily take a sulfa-containing medication.
The dominant message of Dr. Wickner’s talk on drug allergies was to get clear details on the allergic reaction –
“Please, for all of your patients, take an allergy history on every listed medication; often you will be able to remove or clarify the medical record and the changes can be life saving,” she said.
For instance, desensitization to sulfa for patients who’ve had a morbilliform rash without a fever can be done on an outpatient basis. But if the patient had hives, or became short of breath or anaphylactic, it needs to be done as an inpatient by an allergist, she said.
The question of drug allergies is substantial. About 35% of patients have at least one listed drug allergy, with penicillin, NSAIDs, and CT contrast agents topping the list, Dr. Wickner said. Although 10% of the general population and 15% of inpatients have a listed penicillin allergy, more than 90% of listed penicillin allergies turn out not to actually be allergic, in part because penicillin allergies are often diagnosed in childhood and are frequently outgrown over time. Having a listed allergy can impact treatment, Dr. Wickner said, with alternatives not always clear-cut.
She described one patient she saw who had 62 listed drug allergies, prompting her clinicians to wonder, “what can I safely give this patient?” Physicians, she said, subject to drug allergy “alert fatigue,” tend to override about 80% of allergy alerts, but this can sometimes have serious consequences.
“The best time to clarify is when a patient is healthy and well,” said Dr. Wickner, not when they are an inpatient and sick. It is much more difficult to test for an allergy, and to treat an allergic reaction, than when someone’s health is quickly declining.
She urged physicians to ask patients to be specific about the name of a drug they suspect they’re allergic to, about the indications, the symptoms, and the timing – an immediate reaction is much different than a symptom that showed up days later.
“Sometimes they’ll say they’re allergic to penicillin, but will tell you they’ve taken Augmentin or amoxicillin, so you can take that allergy off the list,” Dr. Wickner said.
At Brigham and Women’s Hospital and Massachusetts General Hospital, Boston, physicians have developed protocols for assessing and managing suspected allergies to penicillin, aspirin and NSAIDs, and trimethoprim/sulfamethoxazole – helpful tools, she said, because the nature and context of the reaction can matter a great deal in how to respond to the listed allergy.
If someone has a reaction, and you think it might be anaphylaxis, don’t spend time pondering it, Dr. Wickner said. “If that thought crosses your mind, treat it like anaphylaxis, then analyze after the fact.” Most patients with anaphylaxis have some cutaneous sign, even if it’s just flushing.
Dr. Wickner said that, if an allergist is available, take advantage of that. “If allergy is available in-house, utilize them. Often, skin testing and/or challenge can help patients receive first-line therapy.”
In a question-and-answer session after her presentation, Dr. Wickner said that hospitalists “have a huge role to play” in drug allergy delabeling.
“We would love to have a more standard practice of allergy reconciliation, just like we do with medication reconciliation,” she said. Asking questions to get more specifics is essential – and simply asking directly about each listed allergen is “step one, and you’ll really find it’s going to broaden the things that you can do for your patients.”
Asked about whether reactions listed as allergies are frequently just adverse effects, Dr. Wickner said that patients who say they are frequently nauseous – rather than breaking out into a rash, for example – might not be having a true allergic reaction. After careful consideration, they might be better managed with antinausea medication than avoidance of the drug.
Dr. Wickner reported no relevant financial disclosures.
Paige Wickner, MD, MPH, medical director for quality and safety at Brigham and Women’s Hospital and assistant professor at Harvard Medical School, both in Boston, described a scenario that might sound familiar to hospitalists.
A 72-year-old man is admitted to the hospital for a lung transplant, and has a listed allergy to “sulfa,” contained in antibiotics and other medications. His medical records say his reaction was “rash.”
What do you do?
The answer, Dr. Wickner said, speaking at HM20 Virtual, sponsored by the Society of Hospital Medicine, is to first ask more questions for clarification. How bad was the rash? Was it blistering? To what type of sulfa did the patient have a reaction?
These questions can help determine the next steps. For sulfa-based antibiotics, hospitalists can often desensitize patients with certain reaction characteristics using widely studied protocols to allow the patient to temporarily take a sulfa-containing medication.
The dominant message of Dr. Wickner’s talk on drug allergies was to get clear details on the allergic reaction –
“Please, for all of your patients, take an allergy history on every listed medication; often you will be able to remove or clarify the medical record and the changes can be life saving,” she said.
For instance, desensitization to sulfa for patients who’ve had a morbilliform rash without a fever can be done on an outpatient basis. But if the patient had hives, or became short of breath or anaphylactic, it needs to be done as an inpatient by an allergist, she said.
The question of drug allergies is substantial. About 35% of patients have at least one listed drug allergy, with penicillin, NSAIDs, and CT contrast agents topping the list, Dr. Wickner said. Although 10% of the general population and 15% of inpatients have a listed penicillin allergy, more than 90% of listed penicillin allergies turn out not to actually be allergic, in part because penicillin allergies are often diagnosed in childhood and are frequently outgrown over time. Having a listed allergy can impact treatment, Dr. Wickner said, with alternatives not always clear-cut.
She described one patient she saw who had 62 listed drug allergies, prompting her clinicians to wonder, “what can I safely give this patient?” Physicians, she said, subject to drug allergy “alert fatigue,” tend to override about 80% of allergy alerts, but this can sometimes have serious consequences.
“The best time to clarify is when a patient is healthy and well,” said Dr. Wickner, not when they are an inpatient and sick. It is much more difficult to test for an allergy, and to treat an allergic reaction, than when someone’s health is quickly declining.
She urged physicians to ask patients to be specific about the name of a drug they suspect they’re allergic to, about the indications, the symptoms, and the timing – an immediate reaction is much different than a symptom that showed up days later.
“Sometimes they’ll say they’re allergic to penicillin, but will tell you they’ve taken Augmentin or amoxicillin, so you can take that allergy off the list,” Dr. Wickner said.
At Brigham and Women’s Hospital and Massachusetts General Hospital, Boston, physicians have developed protocols for assessing and managing suspected allergies to penicillin, aspirin and NSAIDs, and trimethoprim/sulfamethoxazole – helpful tools, she said, because the nature and context of the reaction can matter a great deal in how to respond to the listed allergy.
If someone has a reaction, and you think it might be anaphylaxis, don’t spend time pondering it, Dr. Wickner said. “If that thought crosses your mind, treat it like anaphylaxis, then analyze after the fact.” Most patients with anaphylaxis have some cutaneous sign, even if it’s just flushing.
Dr. Wickner said that, if an allergist is available, take advantage of that. “If allergy is available in-house, utilize them. Often, skin testing and/or challenge can help patients receive first-line therapy.”
In a question-and-answer session after her presentation, Dr. Wickner said that hospitalists “have a huge role to play” in drug allergy delabeling.
“We would love to have a more standard practice of allergy reconciliation, just like we do with medication reconciliation,” she said. Asking questions to get more specifics is essential – and simply asking directly about each listed allergen is “step one, and you’ll really find it’s going to broaden the things that you can do for your patients.”
Asked about whether reactions listed as allergies are frequently just adverse effects, Dr. Wickner said that patients who say they are frequently nauseous – rather than breaking out into a rash, for example – might not be having a true allergic reaction. After careful consideration, they might be better managed with antinausea medication than avoidance of the drug.
Dr. Wickner reported no relevant financial disclosures.
FROM HM20 VIRTUAL
FDA approves point-of-care COVID-19 antigen test
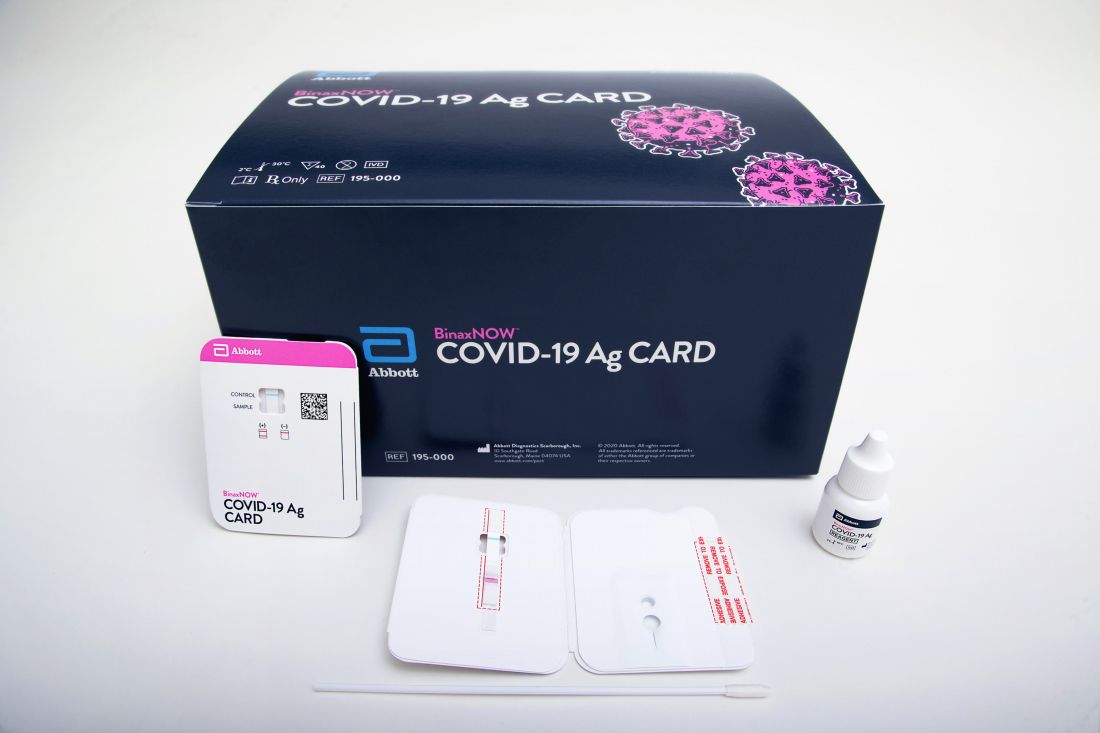
The BinaxNOW COVID-19 Ag Card (Abbott) is similar in some ways to a home pregnancy test. Clinicians read results on a card – one line for a negative result, two lines for positive.
A health care provider swabs a symptomatic patient’s nose, twirls the sample on a test card with a reagent, and waits approximately 15 minutes for results. No additional equipment is required.
Abbott expects the test to cost about $5.00, the company announced.
Office-based physicians, ED physicians, and school nurses could potentially use the product as a point-of-care test. The FDA granted the test emergency use authorization. It is approved for people suspected of having COVID-19 who are within 7 days of symptom onset.
“This new COVID-19 antigen test is an important addition to available tests because the results can be read in minutes, right off the testing card,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, wrote in a news release. “This means people will know if they have the virus in almost real time.”
“This fits into the testing landscape as a simple, inexpensive test that does not require additional equipment,” Marcus Lynch, PhD, assistant manager of the Health Care Horizon Scanning program at ECRI, told Medscape Medical News when asked to comment. ECRI is an independent, nonprofit organization that reviews and analyses COVID-19 therapeutics and diagnostics.
The test could help with early triage of patients who test positive, perhaps alerting physicians to the need to start COVID-19 therapy, added Lynch, who specializes in immunology and vaccine development. The test also could be useful in low-resource settings.
The FDA included a caveat: antigen tests are generally less sensitive than molecular assays. “Due to the potential for decreased sensitivity compared to molecular assays, negative results from an antigen test may need to be confirmed with a molecular test prior to making treatment decisions,” the agency noted.
Lynch agreed and said that when a patient tests negative, physicians still need to use their clinical judgment on the basis of symptoms and other factors. The test is not designed for population-based screening of asymptomatic people, he added.
Abbott announced plans to make up to 50 million tests available per month in the United States starting in October. The product comes with a free smartphone app that people can use to share results with an employer or with others as needed.
This article first appeared on Medscape.com.

The BinaxNOW COVID-19 Ag Card (Abbott) is similar in some ways to a home pregnancy test. Clinicians read results on a card – one line for a negative result, two lines for positive.
A health care provider swabs a symptomatic patient’s nose, twirls the sample on a test card with a reagent, and waits approximately 15 minutes for results. No additional equipment is required.
Abbott expects the test to cost about $5.00, the company announced.
Office-based physicians, ED physicians, and school nurses could potentially use the product as a point-of-care test. The FDA granted the test emergency use authorization. It is approved for people suspected of having COVID-19 who are within 7 days of symptom onset.
“This new COVID-19 antigen test is an important addition to available tests because the results can be read in minutes, right off the testing card,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, wrote in a news release. “This means people will know if they have the virus in almost real time.”
“This fits into the testing landscape as a simple, inexpensive test that does not require additional equipment,” Marcus Lynch, PhD, assistant manager of the Health Care Horizon Scanning program at ECRI, told Medscape Medical News when asked to comment. ECRI is an independent, nonprofit organization that reviews and analyses COVID-19 therapeutics and diagnostics.
The test could help with early triage of patients who test positive, perhaps alerting physicians to the need to start COVID-19 therapy, added Lynch, who specializes in immunology and vaccine development. The test also could be useful in low-resource settings.
The FDA included a caveat: antigen tests are generally less sensitive than molecular assays. “Due to the potential for decreased sensitivity compared to molecular assays, negative results from an antigen test may need to be confirmed with a molecular test prior to making treatment decisions,” the agency noted.
Lynch agreed and said that when a patient tests negative, physicians still need to use their clinical judgment on the basis of symptoms and other factors. The test is not designed for population-based screening of asymptomatic people, he added.
Abbott announced plans to make up to 50 million tests available per month in the United States starting in October. The product comes with a free smartphone app that people can use to share results with an employer or with others as needed.
This article first appeared on Medscape.com.

The BinaxNOW COVID-19 Ag Card (Abbott) is similar in some ways to a home pregnancy test. Clinicians read results on a card – one line for a negative result, two lines for positive.
A health care provider swabs a symptomatic patient’s nose, twirls the sample on a test card with a reagent, and waits approximately 15 minutes for results. No additional equipment is required.
Abbott expects the test to cost about $5.00, the company announced.
Office-based physicians, ED physicians, and school nurses could potentially use the product as a point-of-care test. The FDA granted the test emergency use authorization. It is approved for people suspected of having COVID-19 who are within 7 days of symptom onset.
“This new COVID-19 antigen test is an important addition to available tests because the results can be read in minutes, right off the testing card,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, wrote in a news release. “This means people will know if they have the virus in almost real time.”
“This fits into the testing landscape as a simple, inexpensive test that does not require additional equipment,” Marcus Lynch, PhD, assistant manager of the Health Care Horizon Scanning program at ECRI, told Medscape Medical News when asked to comment. ECRI is an independent, nonprofit organization that reviews and analyses COVID-19 therapeutics and diagnostics.
The test could help with early triage of patients who test positive, perhaps alerting physicians to the need to start COVID-19 therapy, added Lynch, who specializes in immunology and vaccine development. The test also could be useful in low-resource settings.
The FDA included a caveat: antigen tests are generally less sensitive than molecular assays. “Due to the potential for decreased sensitivity compared to molecular assays, negative results from an antigen test may need to be confirmed with a molecular test prior to making treatment decisions,” the agency noted.
Lynch agreed and said that when a patient tests negative, physicians still need to use their clinical judgment on the basis of symptoms and other factors. The test is not designed for population-based screening of asymptomatic people, he added.
Abbott announced plans to make up to 50 million tests available per month in the United States starting in October. The product comes with a free smartphone app that people can use to share results with an employer or with others as needed.
This article first appeared on Medscape.com.
COVID-19 vaccine supply will be limited at first, ACIP says
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) yesterday held its third meeting this summer to discuss the vaccines and plan how initial vaccines will be allocated, inasmuch as supplies will likely be limited at first. Vaccines are expected to be more available as production ramps up and as more than one vaccine become available, but vaccine allocation initially will need to take place in phases.
Considerations include first getting the vaccine to individuals who need it the most, such as healthcare personnel and essential workers, as well as those at higher risk for severe illness or death, including the elderly, those with underlying conditions, and certain racial and ethnic minorities. Other factors include storage requirements that might be difficult to meet in certain settings and the fact that both vaccines must be given in two doses.
Vaccine allocation models
The group presented two possible models for allocating initial vaccine supplies.
The first population model considers risk status within each age group on the basis of underlying health conditions and occupational group, with priority given to healthcare personnel (paid or unpaid) and essential workers. The model considers partial reopening and social distancing, expected vaccine efficacy, prevaccination immunity, mortality, and the direct and indirect benefits of vaccination.
In this model, COVID-19 infections and deaths were reduced when healthcare personnel, essential workers, or adults with underlying conditions were vaccinated. There were smaller differences between the groups with respect to the impact of vaccination. Declines in infections were “more modest” and declines in deaths were greater when adults aged 65 years and older were vaccinated in comparison with other age groups.
The second model focused on vaccination of nursing home healthcare personnel and residents. Vaccinating nursing home healthcare personnel reduced infections and deaths more than vaccinating nursing home residents.
In settings such as long-term care facilities and correction facilities, where people gather in groups, cases increase first among staff. The vaccine working group suggests that vaccinating staff may also benefit individuals living in those facilities.
The working group expects that from 15 to 45 million doses of vaccine will be available by the end of December, depending on which vaccine is approved by then or whether both are approved.
Supplies won’t be nearly enough to vaccinate everyone: There are approximately 17 to 20 million healthcare workers in the United States and 60 to 80 million essential workers who do not work in healthcare. More than 100 million adults have underlying medical conditions that put them at higher risk for hospitalization and death, such as obesity, cardiovascular disease, diabetes, and chronic obstructive pulmonary disease. And approximately 53 million adults are aged 65 years or older.
The group reviewed promising early data for two vaccines under development.
The mRNA-1273 vaccine, made by Moderna with support from two federal agencies, is moving into phase 3 clinical trials – enrollment into the COVID-19 Efficacy and Safety (COVE) study is ongoing, according to Jacqueline M. Miller, MD, senior vice president and therapeutic area head of infectious diseases. The study’s primary objective will be to determine whether two doses can prevent symptomatic COVID-19, according to an NIH news release.
A second mRNA vaccine, BNT 162b2, made by Pfizer and BioNTech, is entering phase 2/3 trials. Nearly 20% of people enrolled are Black or Hispanic persons, and 4% are Asian persons. The team is also trying to recruit Native American participants, Nicholas Kitchin, MD, senior director in Pfizer’s vaccine clinical research and development group, said in a presentation to the advisory committee.
‘Ultra-cold’ temperatures required for storage
Both vaccines require storage at lower temperatures than is usually needed for vaccines. One vaccine must be distributed and stored at -20° C, and the other must be stored, distributed, and handled at -70° C.
This issue stands out most to ACIP Chair Jose Romero, MD. He says the “ultra-cold” temperatures required for storage and transportation of the vaccines will be a “significant problem” for those in rural areas.
High-risk populations such as meat processors and agricultural workers “may have to wait until we have a more stable vaccine that can be transported and delivered more or less at room temperature,” Romero explained. He is the chief medical officer at the Arkansas Department of Health and is a professor of pediatrics and pediatric infectious diseases at the University of Arkansas for Medical Sciences, both in Little Rock.
The advisory committee will meet again on September 22. At that time, they’ll vote on an interim plan for prioritization of the first COVID-19 vaccine.
This article first appeared on Medscape.com.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) yesterday held its third meeting this summer to discuss the vaccines and plan how initial vaccines will be allocated, inasmuch as supplies will likely be limited at first. Vaccines are expected to be more available as production ramps up and as more than one vaccine become available, but vaccine allocation initially will need to take place in phases.
Considerations include first getting the vaccine to individuals who need it the most, such as healthcare personnel and essential workers, as well as those at higher risk for severe illness or death, including the elderly, those with underlying conditions, and certain racial and ethnic minorities. Other factors include storage requirements that might be difficult to meet in certain settings and the fact that both vaccines must be given in two doses.
Vaccine allocation models
The group presented two possible models for allocating initial vaccine supplies.
The first population model considers risk status within each age group on the basis of underlying health conditions and occupational group, with priority given to healthcare personnel (paid or unpaid) and essential workers. The model considers partial reopening and social distancing, expected vaccine efficacy, prevaccination immunity, mortality, and the direct and indirect benefits of vaccination.
In this model, COVID-19 infections and deaths were reduced when healthcare personnel, essential workers, or adults with underlying conditions were vaccinated. There were smaller differences between the groups with respect to the impact of vaccination. Declines in infections were “more modest” and declines in deaths were greater when adults aged 65 years and older were vaccinated in comparison with other age groups.
The second model focused on vaccination of nursing home healthcare personnel and residents. Vaccinating nursing home healthcare personnel reduced infections and deaths more than vaccinating nursing home residents.
In settings such as long-term care facilities and correction facilities, where people gather in groups, cases increase first among staff. The vaccine working group suggests that vaccinating staff may also benefit individuals living in those facilities.
The working group expects that from 15 to 45 million doses of vaccine will be available by the end of December, depending on which vaccine is approved by then or whether both are approved.
Supplies won’t be nearly enough to vaccinate everyone: There are approximately 17 to 20 million healthcare workers in the United States and 60 to 80 million essential workers who do not work in healthcare. More than 100 million adults have underlying medical conditions that put them at higher risk for hospitalization and death, such as obesity, cardiovascular disease, diabetes, and chronic obstructive pulmonary disease. And approximately 53 million adults are aged 65 years or older.
The group reviewed promising early data for two vaccines under development.
The mRNA-1273 vaccine, made by Moderna with support from two federal agencies, is moving into phase 3 clinical trials – enrollment into the COVID-19 Efficacy and Safety (COVE) study is ongoing, according to Jacqueline M. Miller, MD, senior vice president and therapeutic area head of infectious diseases. The study’s primary objective will be to determine whether two doses can prevent symptomatic COVID-19, according to an NIH news release.
A second mRNA vaccine, BNT 162b2, made by Pfizer and BioNTech, is entering phase 2/3 trials. Nearly 20% of people enrolled are Black or Hispanic persons, and 4% are Asian persons. The team is also trying to recruit Native American participants, Nicholas Kitchin, MD, senior director in Pfizer’s vaccine clinical research and development group, said in a presentation to the advisory committee.
‘Ultra-cold’ temperatures required for storage
Both vaccines require storage at lower temperatures than is usually needed for vaccines. One vaccine must be distributed and stored at -20° C, and the other must be stored, distributed, and handled at -70° C.
This issue stands out most to ACIP Chair Jose Romero, MD. He says the “ultra-cold” temperatures required for storage and transportation of the vaccines will be a “significant problem” for those in rural areas.
High-risk populations such as meat processors and agricultural workers “may have to wait until we have a more stable vaccine that can be transported and delivered more or less at room temperature,” Romero explained. He is the chief medical officer at the Arkansas Department of Health and is a professor of pediatrics and pediatric infectious diseases at the University of Arkansas for Medical Sciences, both in Little Rock.
The advisory committee will meet again on September 22. At that time, they’ll vote on an interim plan for prioritization of the first COVID-19 vaccine.
This article first appeared on Medscape.com.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) yesterday held its third meeting this summer to discuss the vaccines and plan how initial vaccines will be allocated, inasmuch as supplies will likely be limited at first. Vaccines are expected to be more available as production ramps up and as more than one vaccine become available, but vaccine allocation initially will need to take place in phases.
Considerations include first getting the vaccine to individuals who need it the most, such as healthcare personnel and essential workers, as well as those at higher risk for severe illness or death, including the elderly, those with underlying conditions, and certain racial and ethnic minorities. Other factors include storage requirements that might be difficult to meet in certain settings and the fact that both vaccines must be given in two doses.
Vaccine allocation models
The group presented two possible models for allocating initial vaccine supplies.
The first population model considers risk status within each age group on the basis of underlying health conditions and occupational group, with priority given to healthcare personnel (paid or unpaid) and essential workers. The model considers partial reopening and social distancing, expected vaccine efficacy, prevaccination immunity, mortality, and the direct and indirect benefits of vaccination.
In this model, COVID-19 infections and deaths were reduced when healthcare personnel, essential workers, or adults with underlying conditions were vaccinated. There were smaller differences between the groups with respect to the impact of vaccination. Declines in infections were “more modest” and declines in deaths were greater when adults aged 65 years and older were vaccinated in comparison with other age groups.
The second model focused on vaccination of nursing home healthcare personnel and residents. Vaccinating nursing home healthcare personnel reduced infections and deaths more than vaccinating nursing home residents.
In settings such as long-term care facilities and correction facilities, where people gather in groups, cases increase first among staff. The vaccine working group suggests that vaccinating staff may also benefit individuals living in those facilities.
The working group expects that from 15 to 45 million doses of vaccine will be available by the end of December, depending on which vaccine is approved by then or whether both are approved.
Supplies won’t be nearly enough to vaccinate everyone: There are approximately 17 to 20 million healthcare workers in the United States and 60 to 80 million essential workers who do not work in healthcare. More than 100 million adults have underlying medical conditions that put them at higher risk for hospitalization and death, such as obesity, cardiovascular disease, diabetes, and chronic obstructive pulmonary disease. And approximately 53 million adults are aged 65 years or older.
The group reviewed promising early data for two vaccines under development.
The mRNA-1273 vaccine, made by Moderna with support from two federal agencies, is moving into phase 3 clinical trials – enrollment into the COVID-19 Efficacy and Safety (COVE) study is ongoing, according to Jacqueline M. Miller, MD, senior vice president and therapeutic area head of infectious diseases. The study’s primary objective will be to determine whether two doses can prevent symptomatic COVID-19, according to an NIH news release.
A second mRNA vaccine, BNT 162b2, made by Pfizer and BioNTech, is entering phase 2/3 trials. Nearly 20% of people enrolled are Black or Hispanic persons, and 4% are Asian persons. The team is also trying to recruit Native American participants, Nicholas Kitchin, MD, senior director in Pfizer’s vaccine clinical research and development group, said in a presentation to the advisory committee.
‘Ultra-cold’ temperatures required for storage
Both vaccines require storage at lower temperatures than is usually needed for vaccines. One vaccine must be distributed and stored at -20° C, and the other must be stored, distributed, and handled at -70° C.
This issue stands out most to ACIP Chair Jose Romero, MD. He says the “ultra-cold” temperatures required for storage and transportation of the vaccines will be a “significant problem” for those in rural areas.
High-risk populations such as meat processors and agricultural workers “may have to wait until we have a more stable vaccine that can be transported and delivered more or less at room temperature,” Romero explained. He is the chief medical officer at the Arkansas Department of Health and is a professor of pediatrics and pediatric infectious diseases at the University of Arkansas for Medical Sciences, both in Little Rock.
The advisory committee will meet again on September 22. At that time, they’ll vote on an interim plan for prioritization of the first COVID-19 vaccine.
This article first appeared on Medscape.com.
Asymptomatic SARS-CoV-2 infections in kids tied to local rates
As communities wrestle with the decision to send children back to school or opt for distance learning, a key question is how many children are likely to have asymptomatic SARS-CoV-2 infections.
“The strong association between prevalence of SARS-CoV-2 in children who are asymptomatic and contemporaneous weekly incidence of COVID-19 in the general population ... provides a simple means for institutions to estimate local pediatric asymptomatic prevalence from the publicly available Johns Hopkins University database,” researchers say in an article published online August 25 in JAMA Pediatrics.
Ana Marija Sola, BS, a researcher at the University of California, San Francisco, and colleagues examined the prevalence of SARS-CoV-2 infection among 33,041 children who underwent routine testing in April and May when hospitals resumed elective medical and surgical care. The hospitals performed reverse transcription–polymerase chain reaction tests for SARS-CoV-2 RNA before surgery, clinic visits, or hospital admissions. Pediatric otolaryngologists reported the prevalence data through May 29 as part of a quality improvement project.
In all, 250 patients tested positive for the virus, for an overall prevalence of 0.65%. Across 25 geographic areas, the prevalence ranged from 0% to 2.2%. By region, prevalence was highest in the Northeast, at 0.90%, and the Midwest, at 0.87%; prevalence was lower in the West, at 0.59%, and the South, at 0.52%.
To get a sense of how those rates compared with overall rates in the same geographic areas, the researchers used the Johns Hopkins University confirmed cases database to calculate the average weekly incidence of COVID-19 for the entire population for each geographic area.
“Asymptomatic pediatric prevalence was significantly associated with weekly incidence of COVID-19 in the general population during the 6-week period over which most testing of individuals without symptoms occurred,” Ms. Sola and colleagues reported. An analysis using additional data from 11 geographic areas demonstrated that this association persisted at a later time point.
The study provides “another window on the question of how likely is it that an asymptomatic child will be carrying coronavirus,” said Susan E. Coffin, MD, MPH, an attending physician for the division of infectious diseases at Children’s Hospital of Philadelphia. However, important related questions remain, said Dr. Coffin, who was not involved with the study.
For one, it is unclear how many children remain asymptomatic in comparison with those who were in a presymptomatic phase at the time of testing. And importantly, “what proportion of these children are infectious?” said Dr. Coffin. “There is some data to suggest that children with asymptomatic infection may be less infectious than children with symptomatic infection.”
It also could be that patients seen at children’s hospitals differ from the general pediatric population. “What does this look like if you do the exact same study in a group of randomly selected children, not children who are queueing up to have a procedure? ... And what do these numbers look like now that stay-at-home orders have been lifted?” Dr. Coffin asked.
Further studies are needed to establish that detection of COVID-19 in the general population is predictive of the prevalence of SARS-CoV-2 infection in asymptomatic children, Dr. Coffin said.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
As communities wrestle with the decision to send children back to school or opt for distance learning, a key question is how many children are likely to have asymptomatic SARS-CoV-2 infections.
“The strong association between prevalence of SARS-CoV-2 in children who are asymptomatic and contemporaneous weekly incidence of COVID-19 in the general population ... provides a simple means for institutions to estimate local pediatric asymptomatic prevalence from the publicly available Johns Hopkins University database,” researchers say in an article published online August 25 in JAMA Pediatrics.
Ana Marija Sola, BS, a researcher at the University of California, San Francisco, and colleagues examined the prevalence of SARS-CoV-2 infection among 33,041 children who underwent routine testing in April and May when hospitals resumed elective medical and surgical care. The hospitals performed reverse transcription–polymerase chain reaction tests for SARS-CoV-2 RNA before surgery, clinic visits, or hospital admissions. Pediatric otolaryngologists reported the prevalence data through May 29 as part of a quality improvement project.
In all, 250 patients tested positive for the virus, for an overall prevalence of 0.65%. Across 25 geographic areas, the prevalence ranged from 0% to 2.2%. By region, prevalence was highest in the Northeast, at 0.90%, and the Midwest, at 0.87%; prevalence was lower in the West, at 0.59%, and the South, at 0.52%.
To get a sense of how those rates compared with overall rates in the same geographic areas, the researchers used the Johns Hopkins University confirmed cases database to calculate the average weekly incidence of COVID-19 for the entire population for each geographic area.
“Asymptomatic pediatric prevalence was significantly associated with weekly incidence of COVID-19 in the general population during the 6-week period over which most testing of individuals without symptoms occurred,” Ms. Sola and colleagues reported. An analysis using additional data from 11 geographic areas demonstrated that this association persisted at a later time point.
The study provides “another window on the question of how likely is it that an asymptomatic child will be carrying coronavirus,” said Susan E. Coffin, MD, MPH, an attending physician for the division of infectious diseases at Children’s Hospital of Philadelphia. However, important related questions remain, said Dr. Coffin, who was not involved with the study.
For one, it is unclear how many children remain asymptomatic in comparison with those who were in a presymptomatic phase at the time of testing. And importantly, “what proportion of these children are infectious?” said Dr. Coffin. “There is some data to suggest that children with asymptomatic infection may be less infectious than children with symptomatic infection.”
It also could be that patients seen at children’s hospitals differ from the general pediatric population. “What does this look like if you do the exact same study in a group of randomly selected children, not children who are queueing up to have a procedure? ... And what do these numbers look like now that stay-at-home orders have been lifted?” Dr. Coffin asked.
Further studies are needed to establish that detection of COVID-19 in the general population is predictive of the prevalence of SARS-CoV-2 infection in asymptomatic children, Dr. Coffin said.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
As communities wrestle with the decision to send children back to school or opt for distance learning, a key question is how many children are likely to have asymptomatic SARS-CoV-2 infections.
“The strong association between prevalence of SARS-CoV-2 in children who are asymptomatic and contemporaneous weekly incidence of COVID-19 in the general population ... provides a simple means for institutions to estimate local pediatric asymptomatic prevalence from the publicly available Johns Hopkins University database,” researchers say in an article published online August 25 in JAMA Pediatrics.
Ana Marija Sola, BS, a researcher at the University of California, San Francisco, and colleagues examined the prevalence of SARS-CoV-2 infection among 33,041 children who underwent routine testing in April and May when hospitals resumed elective medical and surgical care. The hospitals performed reverse transcription–polymerase chain reaction tests for SARS-CoV-2 RNA before surgery, clinic visits, or hospital admissions. Pediatric otolaryngologists reported the prevalence data through May 29 as part of a quality improvement project.
In all, 250 patients tested positive for the virus, for an overall prevalence of 0.65%. Across 25 geographic areas, the prevalence ranged from 0% to 2.2%. By region, prevalence was highest in the Northeast, at 0.90%, and the Midwest, at 0.87%; prevalence was lower in the West, at 0.59%, and the South, at 0.52%.
To get a sense of how those rates compared with overall rates in the same geographic areas, the researchers used the Johns Hopkins University confirmed cases database to calculate the average weekly incidence of COVID-19 for the entire population for each geographic area.
“Asymptomatic pediatric prevalence was significantly associated with weekly incidence of COVID-19 in the general population during the 6-week period over which most testing of individuals without symptoms occurred,” Ms. Sola and colleagues reported. An analysis using additional data from 11 geographic areas demonstrated that this association persisted at a later time point.
The study provides “another window on the question of how likely is it that an asymptomatic child will be carrying coronavirus,” said Susan E. Coffin, MD, MPH, an attending physician for the division of infectious diseases at Children’s Hospital of Philadelphia. However, important related questions remain, said Dr. Coffin, who was not involved with the study.
For one, it is unclear how many children remain asymptomatic in comparison with those who were in a presymptomatic phase at the time of testing. And importantly, “what proportion of these children are infectious?” said Dr. Coffin. “There is some data to suggest that children with asymptomatic infection may be less infectious than children with symptomatic infection.”
It also could be that patients seen at children’s hospitals differ from the general pediatric population. “What does this look like if you do the exact same study in a group of randomly selected children, not children who are queueing up to have a procedure? ... And what do these numbers look like now that stay-at-home orders have been lifted?” Dr. Coffin asked.
Further studies are needed to establish that detection of COVID-19 in the general population is predictive of the prevalence of SARS-CoV-2 infection in asymptomatic children, Dr. Coffin said.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Heart failure: Practice-changing developments for hospitalists
A recently validated, easy-to-use calculator of predicted 7-day mortality risk in patients presenting with acute decompensated heart failure is well worth incorporating into hospitalist clinical practice, Dustin T. Smith, MD, said at HM20 Virtual, hosted by the Society of Hospital Medicine.
In addition to the EHMRG, other highlights of his wide-ranging update on recent practice-changing developments in heart failure directly relevant to hospitalists included the introduction of a simple, evidence-based tool for differentiating heart failure with preserved ejection fraction from other potential causes of unexplained dyspnea on exertion in euvolemic patients, and a study debunking what has been called the potassium repletion reflex in patients with acute heart failure undergoing diuresis.
The ACUTE study
Heart failure is an area of special interest for Dr. Smith. He has been surprised to find that virtually no hospitalists, emergency medicine physicians, or cardiologists he has spoken with have heard of the EHMRG or its validation in the ACUTE (Acute Congestive Heart Failure Urgent Care Evaluation) study. Yet this is a very handy tool for hospitalists, he observed.
The EHMRG algorithm utilizes nine variables for which data is readily available for every patient who arrives at the emergency department with acute heart failure. The variables are age, arrival by ambulance, heart rate, systolic blood pressure, potassium level, oxygen saturation, troponin, serum creatine, and presence or absence of active cancer. The information is entered into a cell phone app, which spits out the patient’s estimated 7-day mortality risk. The algorithm divides patients into one of five risk groups ranging from very low to very high. With the addition of data input as to the presence or absence of ST-segment depression on the 12-lead ECG, the weighted algorithm will simultaneously generate an estimated 30-day mortality risk.
ACUTE was a prospective, observational, real-world validation study of EHMRG involving 1,983 patients seeking emergency department care for acute heart failure at nine Canadian hospitals. The actual 7-day mortality rate was 0% in the very-low-risk group, 0% in the low-risk group, 0.6% with an intermediate-risk EHMRG, 1.9% with high risk, and 3.9% in the very-high-risk group. The corresponding 30-day mortality rates were 0%, 1.9%, 3.9%, 5.9%, and 14.3%.
The University of Toronto investigators also asked participating physicians for their clinical estimates of 7-day mortality risk while blinded to the EHMRG predictions. The algorithm proved more accurate than physician predictions across the board. Indeed, physicians consistently overestimated the mortality risk for all categories except the very-high-risk one, where they underestimated the true risk (Circulation. 2019 Feb 26;139[9]:1146-56).
Given that heart failure remains year after year at the top of the list of most frequent causes for hospital admission, and that there is compelling evidence that many low-risk patients get hospitalized while potentially unsafe early discharges also occur, the EHMRG score fills an important unmet need.
“I think this can help inform us as to who with acute heart failure potentially needs to come into the hospital and who doesn’t,” Dr. Smith said. “I think the sweet spot here is that if you’re in the low- or very-low-risk category, your 7-day mortality is less than 1%; in fact, in this study it’s zero. But once you get to category 3 – the intermediate category – you’re talking about a 7-day mortality of 1%-2%, which I think is high enough to warrant hospital admission for treatment and to watch them, not just send them home.”
The H2FPEF score
Diagnosis of heart failure with preserved ejection fraction (HFpEF) is a challenge in euvolemic patients with clear lungs and dyspnea on exertion. Investigators at the Mayo Clinic have developed and subsequently validated a weighted score known as the H2FPEF score that’s of great assistance in this task. The score is based upon a set of six simple variables universally available in patients undergoing diagnostic workup for the numerous potential causes for dyspnea on exertion. Together these six variables comprise the acronym H2FPEF:
- Heavy: One point for a BMI greater than 30 kg/m2.
- Hypertension: One point for being on two or more antihypertensive drugs.
- Atrial fibrillation: Three points for paroxysmal or persistent AF.
- Pulmonary hypertension: One point for having a Doppler echocardiographic estimated pulmonary artery systolic pressure greater than 35 mm Hg.
- Elder: One point for age greater than 60 years.
- Filling pressure: One point for a Doppler echocardiographic E/e’ ratio above 9.
The total score can range from 0 to 9. (Circulation. 2018 Aug 28;138[9]:861-70).
Each 1-point increase in the score essentially doubled a patient’s risk of having HFpEF as opposed to pulmonary embolism or some other cause for the dyspnea.
“I really like this H2FPEF score. The score works very, very well. Once you get to a score of 6 or above, the probability of HFpEF is more than 90%, which is pretty powerful. I think this is worthwhile,” Dr. Smith said.
In their derivation and validation cohorts, the Mayo Clinic investigators used as their gold standard for diagnosis of HFpEF invasive hemodynamic exercise testing with a pulmonary artery catheter in place to measure pressures. A score that enables hospitalists to lessen the need for that kind of costly invasive testing is most welcome.
“Here’s how I’d use this score: With an H2FPEF score of 0-1, HFpEF is unlikely. With an intermediate score of 2-5, additional testing is warranted. If the score is high, 6-9, I think HFpEF is likely,” the hospitalist said.
Dr. Smith isn’t the only big fan of the H2FPEF score. In an editorial accompanying publication of the score’s validation study, Walter J. Paulus, MD, PhD, hailed the H2FPEF score as “a unique tour de force” which constitutes a major advance beyond the confusing diagnostic recommendations for HFpEF issued by the European Society of Cardiology and the American Society of Echocardiography, which he said have been “met by skepticism qualifying them as overcomplicated and even triggered disbelief in the existence of HFpEF.”
Particularly interesting were the variables rejected for inclusion in the H2FPEF score because they failed to achieve statistical significance as predictors, even though they’re often considered important in defining HFpEF, he noted. These included left atrial volume index, sex, and levels of circulating N-terminal probrain natriuretic peptide, wrote Dr. Paulus, professor of cardiac pathophysiology at VU University, Amsterdam.
Debunking the potassium repletion reflex
Longstanding conventional wisdom holds that patients hospitalized for heart failure need to maintain a serum potassium above 4.0 mEq/L.
“I’m sure you’ve all written orders to keep the potassium greater than 4.0 mEq/L and the magnesium above 2mEq/L about a million times, like I have,” Dr. Smith said.
But it turns out this traditional practice, which involves a huge cost in terms of time, money, and health care resources, is supported by weak evidence – and an important recent study has now debunked what the investigators termed the potassium “repletion reflex.”
The investigators at the University of Massachusetts identified 4,995 patients admitted with exacerbation of acute heart failure and a normal admission serum potassium level of 3.5-5.0 mEq/L. More than 70% received potassium repletion at least once within a 72-hour observation window, during which 2,080 patients maintained a low-normal serum potassium below 4.0 mEq/L, 2,326 had a mid-normal level of 4.0-4.5 mEq/L, and 589 had a high-normal level of more than 4.5 mEq/L but not more than 5.0 mEq/L.
The study had three endpoints: in-hospital mortality, transfer to the intensive care unit, and hospital length of stay. After statistical adjustment for comorbidities, demographics, and severity at admission, there was no difference between the low- and mid-normal serum potassium groups in any of the three endpoints. In contrast, the high-normal potassium group had a significantly longer length of stay, by a median of 0.6 extra days. The high-normal group also had a 78% increased likelihood of ICU transfer and a 51% increased risk of in-hospital mortality, although neither of these differences reached statistical significance (J Hosp Med. 2019 Dec 1;14[12]:729-36).
“A potassium greater than 4.5 mEq/L may be associated with increased risk of worse outcomes,” Dr. Smith observed. “I think the sweet spot may be 3.5-4.5 mEq/L based on this study.”
He reported having no financial conflicts regarding his presentation.
A recently validated, easy-to-use calculator of predicted 7-day mortality risk in patients presenting with acute decompensated heart failure is well worth incorporating into hospitalist clinical practice, Dustin T. Smith, MD, said at HM20 Virtual, hosted by the Society of Hospital Medicine.
In addition to the EHMRG, other highlights of his wide-ranging update on recent practice-changing developments in heart failure directly relevant to hospitalists included the introduction of a simple, evidence-based tool for differentiating heart failure with preserved ejection fraction from other potential causes of unexplained dyspnea on exertion in euvolemic patients, and a study debunking what has been called the potassium repletion reflex in patients with acute heart failure undergoing diuresis.
The ACUTE study
Heart failure is an area of special interest for Dr. Smith. He has been surprised to find that virtually no hospitalists, emergency medicine physicians, or cardiologists he has spoken with have heard of the EHMRG or its validation in the ACUTE (Acute Congestive Heart Failure Urgent Care Evaluation) study. Yet this is a very handy tool for hospitalists, he observed.
The EHMRG algorithm utilizes nine variables for which data is readily available for every patient who arrives at the emergency department with acute heart failure. The variables are age, arrival by ambulance, heart rate, systolic blood pressure, potassium level, oxygen saturation, troponin, serum creatine, and presence or absence of active cancer. The information is entered into a cell phone app, which spits out the patient’s estimated 7-day mortality risk. The algorithm divides patients into one of five risk groups ranging from very low to very high. With the addition of data input as to the presence or absence of ST-segment depression on the 12-lead ECG, the weighted algorithm will simultaneously generate an estimated 30-day mortality risk.
ACUTE was a prospective, observational, real-world validation study of EHMRG involving 1,983 patients seeking emergency department care for acute heart failure at nine Canadian hospitals. The actual 7-day mortality rate was 0% in the very-low-risk group, 0% in the low-risk group, 0.6% with an intermediate-risk EHMRG, 1.9% with high risk, and 3.9% in the very-high-risk group. The corresponding 30-day mortality rates were 0%, 1.9%, 3.9%, 5.9%, and 14.3%.
The University of Toronto investigators also asked participating physicians for their clinical estimates of 7-day mortality risk while blinded to the EHMRG predictions. The algorithm proved more accurate than physician predictions across the board. Indeed, physicians consistently overestimated the mortality risk for all categories except the very-high-risk one, where they underestimated the true risk (Circulation. 2019 Feb 26;139[9]:1146-56).
Given that heart failure remains year after year at the top of the list of most frequent causes for hospital admission, and that there is compelling evidence that many low-risk patients get hospitalized while potentially unsafe early discharges also occur, the EHMRG score fills an important unmet need.
“I think this can help inform us as to who with acute heart failure potentially needs to come into the hospital and who doesn’t,” Dr. Smith said. “I think the sweet spot here is that if you’re in the low- or very-low-risk category, your 7-day mortality is less than 1%; in fact, in this study it’s zero. But once you get to category 3 – the intermediate category – you’re talking about a 7-day mortality of 1%-2%, which I think is high enough to warrant hospital admission for treatment and to watch them, not just send them home.”
The H2FPEF score
Diagnosis of heart failure with preserved ejection fraction (HFpEF) is a challenge in euvolemic patients with clear lungs and dyspnea on exertion. Investigators at the Mayo Clinic have developed and subsequently validated a weighted score known as the H2FPEF score that’s of great assistance in this task. The score is based upon a set of six simple variables universally available in patients undergoing diagnostic workup for the numerous potential causes for dyspnea on exertion. Together these six variables comprise the acronym H2FPEF:
- Heavy: One point for a BMI greater than 30 kg/m2.
- Hypertension: One point for being on two or more antihypertensive drugs.
- Atrial fibrillation: Three points for paroxysmal or persistent AF.
- Pulmonary hypertension: One point for having a Doppler echocardiographic estimated pulmonary artery systolic pressure greater than 35 mm Hg.
- Elder: One point for age greater than 60 years.
- Filling pressure: One point for a Doppler echocardiographic E/e’ ratio above 9.
The total score can range from 0 to 9. (Circulation. 2018 Aug 28;138[9]:861-70).
Each 1-point increase in the score essentially doubled a patient’s risk of having HFpEF as opposed to pulmonary embolism or some other cause for the dyspnea.
“I really like this H2FPEF score. The score works very, very well. Once you get to a score of 6 or above, the probability of HFpEF is more than 90%, which is pretty powerful. I think this is worthwhile,” Dr. Smith said.
In their derivation and validation cohorts, the Mayo Clinic investigators used as their gold standard for diagnosis of HFpEF invasive hemodynamic exercise testing with a pulmonary artery catheter in place to measure pressures. A score that enables hospitalists to lessen the need for that kind of costly invasive testing is most welcome.
“Here’s how I’d use this score: With an H2FPEF score of 0-1, HFpEF is unlikely. With an intermediate score of 2-5, additional testing is warranted. If the score is high, 6-9, I think HFpEF is likely,” the hospitalist said.
Dr. Smith isn’t the only big fan of the H2FPEF score. In an editorial accompanying publication of the score’s validation study, Walter J. Paulus, MD, PhD, hailed the H2FPEF score as “a unique tour de force” which constitutes a major advance beyond the confusing diagnostic recommendations for HFpEF issued by the European Society of Cardiology and the American Society of Echocardiography, which he said have been “met by skepticism qualifying them as overcomplicated and even triggered disbelief in the existence of HFpEF.”
Particularly interesting were the variables rejected for inclusion in the H2FPEF score because they failed to achieve statistical significance as predictors, even though they’re often considered important in defining HFpEF, he noted. These included left atrial volume index, sex, and levels of circulating N-terminal probrain natriuretic peptide, wrote Dr. Paulus, professor of cardiac pathophysiology at VU University, Amsterdam.
Debunking the potassium repletion reflex
Longstanding conventional wisdom holds that patients hospitalized for heart failure need to maintain a serum potassium above 4.0 mEq/L.
“I’m sure you’ve all written orders to keep the potassium greater than 4.0 mEq/L and the magnesium above 2mEq/L about a million times, like I have,” Dr. Smith said.
But it turns out this traditional practice, which involves a huge cost in terms of time, money, and health care resources, is supported by weak evidence – and an important recent study has now debunked what the investigators termed the potassium “repletion reflex.”
The investigators at the University of Massachusetts identified 4,995 patients admitted with exacerbation of acute heart failure and a normal admission serum potassium level of 3.5-5.0 mEq/L. More than 70% received potassium repletion at least once within a 72-hour observation window, during which 2,080 patients maintained a low-normal serum potassium below 4.0 mEq/L, 2,326 had a mid-normal level of 4.0-4.5 mEq/L, and 589 had a high-normal level of more than 4.5 mEq/L but not more than 5.0 mEq/L.
The study had three endpoints: in-hospital mortality, transfer to the intensive care unit, and hospital length of stay. After statistical adjustment for comorbidities, demographics, and severity at admission, there was no difference between the low- and mid-normal serum potassium groups in any of the three endpoints. In contrast, the high-normal potassium group had a significantly longer length of stay, by a median of 0.6 extra days. The high-normal group also had a 78% increased likelihood of ICU transfer and a 51% increased risk of in-hospital mortality, although neither of these differences reached statistical significance (J Hosp Med. 2019 Dec 1;14[12]:729-36).
“A potassium greater than 4.5 mEq/L may be associated with increased risk of worse outcomes,” Dr. Smith observed. “I think the sweet spot may be 3.5-4.5 mEq/L based on this study.”
He reported having no financial conflicts regarding his presentation.
A recently validated, easy-to-use calculator of predicted 7-day mortality risk in patients presenting with acute decompensated heart failure is well worth incorporating into hospitalist clinical practice, Dustin T. Smith, MD, said at HM20 Virtual, hosted by the Society of Hospital Medicine.
In addition to the EHMRG, other highlights of his wide-ranging update on recent practice-changing developments in heart failure directly relevant to hospitalists included the introduction of a simple, evidence-based tool for differentiating heart failure with preserved ejection fraction from other potential causes of unexplained dyspnea on exertion in euvolemic patients, and a study debunking what has been called the potassium repletion reflex in patients with acute heart failure undergoing diuresis.
The ACUTE study
Heart failure is an area of special interest for Dr. Smith. He has been surprised to find that virtually no hospitalists, emergency medicine physicians, or cardiologists he has spoken with have heard of the EHMRG or its validation in the ACUTE (Acute Congestive Heart Failure Urgent Care Evaluation) study. Yet this is a very handy tool for hospitalists, he observed.
The EHMRG algorithm utilizes nine variables for which data is readily available for every patient who arrives at the emergency department with acute heart failure. The variables are age, arrival by ambulance, heart rate, systolic blood pressure, potassium level, oxygen saturation, troponin, serum creatine, and presence or absence of active cancer. The information is entered into a cell phone app, which spits out the patient’s estimated 7-day mortality risk. The algorithm divides patients into one of five risk groups ranging from very low to very high. With the addition of data input as to the presence or absence of ST-segment depression on the 12-lead ECG, the weighted algorithm will simultaneously generate an estimated 30-day mortality risk.
ACUTE was a prospective, observational, real-world validation study of EHMRG involving 1,983 patients seeking emergency department care for acute heart failure at nine Canadian hospitals. The actual 7-day mortality rate was 0% in the very-low-risk group, 0% in the low-risk group, 0.6% with an intermediate-risk EHMRG, 1.9% with high risk, and 3.9% in the very-high-risk group. The corresponding 30-day mortality rates were 0%, 1.9%, 3.9%, 5.9%, and 14.3%.
The University of Toronto investigators also asked participating physicians for their clinical estimates of 7-day mortality risk while blinded to the EHMRG predictions. The algorithm proved more accurate than physician predictions across the board. Indeed, physicians consistently overestimated the mortality risk for all categories except the very-high-risk one, where they underestimated the true risk (Circulation. 2019 Feb 26;139[9]:1146-56).
Given that heart failure remains year after year at the top of the list of most frequent causes for hospital admission, and that there is compelling evidence that many low-risk patients get hospitalized while potentially unsafe early discharges also occur, the EHMRG score fills an important unmet need.
“I think this can help inform us as to who with acute heart failure potentially needs to come into the hospital and who doesn’t,” Dr. Smith said. “I think the sweet spot here is that if you’re in the low- or very-low-risk category, your 7-day mortality is less than 1%; in fact, in this study it’s zero. But once you get to category 3 – the intermediate category – you’re talking about a 7-day mortality of 1%-2%, which I think is high enough to warrant hospital admission for treatment and to watch them, not just send them home.”
The H2FPEF score
Diagnosis of heart failure with preserved ejection fraction (HFpEF) is a challenge in euvolemic patients with clear lungs and dyspnea on exertion. Investigators at the Mayo Clinic have developed and subsequently validated a weighted score known as the H2FPEF score that’s of great assistance in this task. The score is based upon a set of six simple variables universally available in patients undergoing diagnostic workup for the numerous potential causes for dyspnea on exertion. Together these six variables comprise the acronym H2FPEF:
- Heavy: One point for a BMI greater than 30 kg/m2.
- Hypertension: One point for being on two or more antihypertensive drugs.
- Atrial fibrillation: Three points for paroxysmal or persistent AF.
- Pulmonary hypertension: One point for having a Doppler echocardiographic estimated pulmonary artery systolic pressure greater than 35 mm Hg.
- Elder: One point for age greater than 60 years.
- Filling pressure: One point for a Doppler echocardiographic E/e’ ratio above 9.
The total score can range from 0 to 9. (Circulation. 2018 Aug 28;138[9]:861-70).
Each 1-point increase in the score essentially doubled a patient’s risk of having HFpEF as opposed to pulmonary embolism or some other cause for the dyspnea.
“I really like this H2FPEF score. The score works very, very well. Once you get to a score of 6 or above, the probability of HFpEF is more than 90%, which is pretty powerful. I think this is worthwhile,” Dr. Smith said.
In their derivation and validation cohorts, the Mayo Clinic investigators used as their gold standard for diagnosis of HFpEF invasive hemodynamic exercise testing with a pulmonary artery catheter in place to measure pressures. A score that enables hospitalists to lessen the need for that kind of costly invasive testing is most welcome.
“Here’s how I’d use this score: With an H2FPEF score of 0-1, HFpEF is unlikely. With an intermediate score of 2-5, additional testing is warranted. If the score is high, 6-9, I think HFpEF is likely,” the hospitalist said.
Dr. Smith isn’t the only big fan of the H2FPEF score. In an editorial accompanying publication of the score’s validation study, Walter J. Paulus, MD, PhD, hailed the H2FPEF score as “a unique tour de force” which constitutes a major advance beyond the confusing diagnostic recommendations for HFpEF issued by the European Society of Cardiology and the American Society of Echocardiography, which he said have been “met by skepticism qualifying them as overcomplicated and even triggered disbelief in the existence of HFpEF.”
Particularly interesting were the variables rejected for inclusion in the H2FPEF score because they failed to achieve statistical significance as predictors, even though they’re often considered important in defining HFpEF, he noted. These included left atrial volume index, sex, and levels of circulating N-terminal probrain natriuretic peptide, wrote Dr. Paulus, professor of cardiac pathophysiology at VU University, Amsterdam.
Debunking the potassium repletion reflex
Longstanding conventional wisdom holds that patients hospitalized for heart failure need to maintain a serum potassium above 4.0 mEq/L.
“I’m sure you’ve all written orders to keep the potassium greater than 4.0 mEq/L and the magnesium above 2mEq/L about a million times, like I have,” Dr. Smith said.
But it turns out this traditional practice, which involves a huge cost in terms of time, money, and health care resources, is supported by weak evidence – and an important recent study has now debunked what the investigators termed the potassium “repletion reflex.”
The investigators at the University of Massachusetts identified 4,995 patients admitted with exacerbation of acute heart failure and a normal admission serum potassium level of 3.5-5.0 mEq/L. More than 70% received potassium repletion at least once within a 72-hour observation window, during which 2,080 patients maintained a low-normal serum potassium below 4.0 mEq/L, 2,326 had a mid-normal level of 4.0-4.5 mEq/L, and 589 had a high-normal level of more than 4.5 mEq/L but not more than 5.0 mEq/L.
The study had three endpoints: in-hospital mortality, transfer to the intensive care unit, and hospital length of stay. After statistical adjustment for comorbidities, demographics, and severity at admission, there was no difference between the low- and mid-normal serum potassium groups in any of the three endpoints. In contrast, the high-normal potassium group had a significantly longer length of stay, by a median of 0.6 extra days. The high-normal group also had a 78% increased likelihood of ICU transfer and a 51% increased risk of in-hospital mortality, although neither of these differences reached statistical significance (J Hosp Med. 2019 Dec 1;14[12]:729-36).
“A potassium greater than 4.5 mEq/L may be associated with increased risk of worse outcomes,” Dr. Smith observed. “I think the sweet spot may be 3.5-4.5 mEq/L based on this study.”
He reported having no financial conflicts regarding his presentation.
FROM HM20 VIRTUAL
Convalescent plasma actions spark trial recruitment concerns
The agency’s move took many investigators by surprise. The EUA was announced at the White House the day after President Donald J. Trump accused the FDA of delaying approval of therapeutics to hurt his re-election chances.
In a memo describing the decision, the FDA cited data from some controlled and uncontrolled studies and, primarily, data from an open-label expanded-access protocol overseen by the Mayo Clinic.
At the White House, FDA Commissioner Stephen Hahn, MD, said that plasma had been found to save the lives of 35 out of every 100 who were treated. That figure was later found to have been erroneous, and many experts pointed out that Hahn had conflated an absolute risk reduction with a relative reduction. After a firestorm of criticism, Hahn issued an apology.
“The criticism is entirely justified,” he tweeted. “What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
About 15 randomized controlled trials – out of 54 total studies involving convalescent plasma – are underway in the United States, according to ClinicalTrials.gov. The FDA’s Aug. 23 emergency authorization gave clinicians wide leeway to employ convalescent plasma in patients hospitalized with COVID-19.
The agency noted, however, that “adequate and well-controlled randomized trials remain necessary for a definitive demonstration of COVID-19 convalescent plasma efficacy and to determine the optimal product attributes and appropriate patient populations for its use.”
But it’s not clear that people with COVID-19, especially those who are severely ill and hospitalized, will choose to enlist in a clinical trial – where they could receive a placebo – when they instead could get plasma.
“I’ve been asked repeatedly whether the EUA will affect our ability to recruit people into our hospitalized patient trial,” said Liise-anne Pirofski, MD, FIDSA, chief of the department of medicine, infectious diseases division at Albert Einstein College of Medicine and Montefiore Medical Center in the Bronx, New York. “I do not know,” she said, on a call with reporters organized by the Infectious Diseases Society of America.
“But,” she said, “I do know that the trial will continue and that we will discuss the evidence that we have with our patients and give them all that we can to help them weigh the evidence and make up their minds.”
Pirofski said the study being conducted at Montefiore and four other sites has since late April enrolled 190 patients out of a hoped-for 300.
When the study – which compares convalescent plasma to saline in hospitalized patients – was first designed, “there was not any funding for our trial and honestly not a whole lot of interest,” Pirofski told reporters. Individual donors helped support the initial rollout in late April and the trial quickly enrolled 150 patients as the pandemic peaked in the New York City area.
The National Institutes of Health has since given funding, which allowed the study to expand to New York University, Yale University, the University of Miami, and the University of Texas at Houston.
Hopeful, but a long way to go
Shmuel Shoham, MD, FIDSA, associate director of the transplant and oncology infectious diseases center at Johns Hopkins University School of Medicine in Baltimore, said that he’s hopeful that people will continue to enroll in his trial, which is seeking to determine if plasma can prevent COVID-19 in those who’ve been recently exposed.
“Volunteers joining the study is the only way that we’re going to get to know whether this stuff works for prevention and treatment,” Shoham said on the call. He urged physicians and other healthcare workers to talk with patients about considering trial participation.
Shoham’s study is being conducted at 30 US sites and one at the Navajo Nation. It has enrolled 25 out of a hoped-for 500 participants. “We have a long way to go,” said Shoham.
Another Hopkins study to determine whether plasma is helpful in shortening illness in nonhospitalized patients, which is being conducted at the same 31 sites, has enrolled 50 out of 600.
Shoham said recruiting patients with COVID for any study had proven to be difficult. “The vast majority of people that have coronavirus do not come to centers that do clinical trials or interventional trials,” he said, adding that, in addition, most of those “who have coronavirus don’t want to be in a trial. They just want to have coronavirus and get it over with.”
But it’s important to understand how to conduct trials in a pandemic – in part to get answers quickly, he said. Researchers have been looking at convalescent plasma for months, said Shoham. “Why don’t we have the randomized clinical trial data that we want?”
Pirofski noted that trials have also been hobbled in part by “the shifting areas of the pandemic.” Fewer cases make for fewer potential plasma donors.
Both Shoham and Pirofski also said that more needed to be done to encourage plasma donors to participate.
The US Department of Health & Human Services clarified in August that hospitals, physicians, health plans, and other health care workers could contact individuals who had recovered from COVID-19 without violating the HIPAA privacy rule.
Pirofski said she believes that trial investigators know it is legal to reach out to patients. But, she said, “it probably could be better known.”
This article first appeared on Medscape.com.
The agency’s move took many investigators by surprise. The EUA was announced at the White House the day after President Donald J. Trump accused the FDA of delaying approval of therapeutics to hurt his re-election chances.
In a memo describing the decision, the FDA cited data from some controlled and uncontrolled studies and, primarily, data from an open-label expanded-access protocol overseen by the Mayo Clinic.
At the White House, FDA Commissioner Stephen Hahn, MD, said that plasma had been found to save the lives of 35 out of every 100 who were treated. That figure was later found to have been erroneous, and many experts pointed out that Hahn had conflated an absolute risk reduction with a relative reduction. After a firestorm of criticism, Hahn issued an apology.
“The criticism is entirely justified,” he tweeted. “What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
About 15 randomized controlled trials – out of 54 total studies involving convalescent plasma – are underway in the United States, according to ClinicalTrials.gov. The FDA’s Aug. 23 emergency authorization gave clinicians wide leeway to employ convalescent plasma in patients hospitalized with COVID-19.
The agency noted, however, that “adequate and well-controlled randomized trials remain necessary for a definitive demonstration of COVID-19 convalescent plasma efficacy and to determine the optimal product attributes and appropriate patient populations for its use.”
But it’s not clear that people with COVID-19, especially those who are severely ill and hospitalized, will choose to enlist in a clinical trial – where they could receive a placebo – when they instead could get plasma.
“I’ve been asked repeatedly whether the EUA will affect our ability to recruit people into our hospitalized patient trial,” said Liise-anne Pirofski, MD, FIDSA, chief of the department of medicine, infectious diseases division at Albert Einstein College of Medicine and Montefiore Medical Center in the Bronx, New York. “I do not know,” she said, on a call with reporters organized by the Infectious Diseases Society of America.
“But,” she said, “I do know that the trial will continue and that we will discuss the evidence that we have with our patients and give them all that we can to help them weigh the evidence and make up their minds.”
Pirofski said the study being conducted at Montefiore and four other sites has since late April enrolled 190 patients out of a hoped-for 300.
When the study – which compares convalescent plasma to saline in hospitalized patients – was first designed, “there was not any funding for our trial and honestly not a whole lot of interest,” Pirofski told reporters. Individual donors helped support the initial rollout in late April and the trial quickly enrolled 150 patients as the pandemic peaked in the New York City area.
The National Institutes of Health has since given funding, which allowed the study to expand to New York University, Yale University, the University of Miami, and the University of Texas at Houston.
Hopeful, but a long way to go
Shmuel Shoham, MD, FIDSA, associate director of the transplant and oncology infectious diseases center at Johns Hopkins University School of Medicine in Baltimore, said that he’s hopeful that people will continue to enroll in his trial, which is seeking to determine if plasma can prevent COVID-19 in those who’ve been recently exposed.
“Volunteers joining the study is the only way that we’re going to get to know whether this stuff works for prevention and treatment,” Shoham said on the call. He urged physicians and other healthcare workers to talk with patients about considering trial participation.
Shoham’s study is being conducted at 30 US sites and one at the Navajo Nation. It has enrolled 25 out of a hoped-for 500 participants. “We have a long way to go,” said Shoham.
Another Hopkins study to determine whether plasma is helpful in shortening illness in nonhospitalized patients, which is being conducted at the same 31 sites, has enrolled 50 out of 600.
Shoham said recruiting patients with COVID for any study had proven to be difficult. “The vast majority of people that have coronavirus do not come to centers that do clinical trials or interventional trials,” he said, adding that, in addition, most of those “who have coronavirus don’t want to be in a trial. They just want to have coronavirus and get it over with.”
But it’s important to understand how to conduct trials in a pandemic – in part to get answers quickly, he said. Researchers have been looking at convalescent plasma for months, said Shoham. “Why don’t we have the randomized clinical trial data that we want?”
Pirofski noted that trials have also been hobbled in part by “the shifting areas of the pandemic.” Fewer cases make for fewer potential plasma donors.
Both Shoham and Pirofski also said that more needed to be done to encourage plasma donors to participate.
The US Department of Health & Human Services clarified in August that hospitals, physicians, health plans, and other health care workers could contact individuals who had recovered from COVID-19 without violating the HIPAA privacy rule.
Pirofski said she believes that trial investigators know it is legal to reach out to patients. But, she said, “it probably could be better known.”
This article first appeared on Medscape.com.
The agency’s move took many investigators by surprise. The EUA was announced at the White House the day after President Donald J. Trump accused the FDA of delaying approval of therapeutics to hurt his re-election chances.
In a memo describing the decision, the FDA cited data from some controlled and uncontrolled studies and, primarily, data from an open-label expanded-access protocol overseen by the Mayo Clinic.
At the White House, FDA Commissioner Stephen Hahn, MD, said that plasma had been found to save the lives of 35 out of every 100 who were treated. That figure was later found to have been erroneous, and many experts pointed out that Hahn had conflated an absolute risk reduction with a relative reduction. After a firestorm of criticism, Hahn issued an apology.
“The criticism is entirely justified,” he tweeted. “What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
About 15 randomized controlled trials – out of 54 total studies involving convalescent plasma – are underway in the United States, according to ClinicalTrials.gov. The FDA’s Aug. 23 emergency authorization gave clinicians wide leeway to employ convalescent plasma in patients hospitalized with COVID-19.
The agency noted, however, that “adequate and well-controlled randomized trials remain necessary for a definitive demonstration of COVID-19 convalescent plasma efficacy and to determine the optimal product attributes and appropriate patient populations for its use.”
But it’s not clear that people with COVID-19, especially those who are severely ill and hospitalized, will choose to enlist in a clinical trial – where they could receive a placebo – when they instead could get plasma.
“I’ve been asked repeatedly whether the EUA will affect our ability to recruit people into our hospitalized patient trial,” said Liise-anne Pirofski, MD, FIDSA, chief of the department of medicine, infectious diseases division at Albert Einstein College of Medicine and Montefiore Medical Center in the Bronx, New York. “I do not know,” she said, on a call with reporters organized by the Infectious Diseases Society of America.
“But,” she said, “I do know that the trial will continue and that we will discuss the evidence that we have with our patients and give them all that we can to help them weigh the evidence and make up their minds.”
Pirofski said the study being conducted at Montefiore and four other sites has since late April enrolled 190 patients out of a hoped-for 300.
When the study – which compares convalescent plasma to saline in hospitalized patients – was first designed, “there was not any funding for our trial and honestly not a whole lot of interest,” Pirofski told reporters. Individual donors helped support the initial rollout in late April and the trial quickly enrolled 150 patients as the pandemic peaked in the New York City area.
The National Institutes of Health has since given funding, which allowed the study to expand to New York University, Yale University, the University of Miami, and the University of Texas at Houston.
Hopeful, but a long way to go
Shmuel Shoham, MD, FIDSA, associate director of the transplant and oncology infectious diseases center at Johns Hopkins University School of Medicine in Baltimore, said that he’s hopeful that people will continue to enroll in his trial, which is seeking to determine if plasma can prevent COVID-19 in those who’ve been recently exposed.
“Volunteers joining the study is the only way that we’re going to get to know whether this stuff works for prevention and treatment,” Shoham said on the call. He urged physicians and other healthcare workers to talk with patients about considering trial participation.
Shoham’s study is being conducted at 30 US sites and one at the Navajo Nation. It has enrolled 25 out of a hoped-for 500 participants. “We have a long way to go,” said Shoham.
Another Hopkins study to determine whether plasma is helpful in shortening illness in nonhospitalized patients, which is being conducted at the same 31 sites, has enrolled 50 out of 600.
Shoham said recruiting patients with COVID for any study had proven to be difficult. “The vast majority of people that have coronavirus do not come to centers that do clinical trials or interventional trials,” he said, adding that, in addition, most of those “who have coronavirus don’t want to be in a trial. They just want to have coronavirus and get it over with.”
But it’s important to understand how to conduct trials in a pandemic – in part to get answers quickly, he said. Researchers have been looking at convalescent plasma for months, said Shoham. “Why don’t we have the randomized clinical trial data that we want?”
Pirofski noted that trials have also been hobbled in part by “the shifting areas of the pandemic.” Fewer cases make for fewer potential plasma donors.
Both Shoham and Pirofski also said that more needed to be done to encourage plasma donors to participate.
The US Department of Health & Human Services clarified in August that hospitals, physicians, health plans, and other health care workers could contact individuals who had recovered from COVID-19 without violating the HIPAA privacy rule.
Pirofski said she believes that trial investigators know it is legal to reach out to patients. But, she said, “it probably could be better known.”
This article first appeared on Medscape.com.
Prognosis for rural hospitals worsens with pandemic
Jerome Antone said he is one of the lucky ones.
After becoming ill with COVID-19, Mr. Antone was hospitalized only 65 miles away from his small Alabama town. He is the mayor of Georgiana – population 1,700.
“It hit our rural community so rabid,” Mr. Antone said. The town’s hospital closed last year. If hospitals in nearby communities don’t have beds available, “you may have to go 4 or 5 hours away.”
Eighteen rural hospitals closed last year and the first 3 months of 2020 were “really big months,” said Mark Holmes, PhD, director of the Cecil G. Sheps Center for Health Services Research at the University of North Carolina at Chapel Hill. Many of the losses are in Southern states like Florida and Texas. More than 170 rural hospitals have closed nationwide since 2005, according to data collected by the Sheps Center.
It’s a dangerous scenario. “We know that a closure leads to higher mortality pretty quickly” among the populations served, said Dr. Holmes, who is also a professor at UNC Gillings School of Global Public Health. “That’s pretty clear.”
One 2019 study found that death rates in the surrounding communities increase nearly 6% after a rural hospital closes – and that’s when there’s not a pandemic.
Add to that what is known about the coronavirus: People who are obese or live with diabetes, hypertension, asthma, and other underlying health issues are more susceptible to COVID-19. Rural areas tend to have higher rates of these conditions. And rural residents are more likely to be older, sicker and poorer than those in urban areas. All this leaves rural communities particularly vulnerable to the coronavirus.
Congress approved billions in federal relief funds for health care providers. Initially, federal officials based what a hospital would get on its Medicare payments, but by late April they heeded criticism and carved out funds for rural hospitals and COVID-19 hot spots. Rural hospitals leapt at the chance to shore up already-negative budgets and prepare for the pandemic.
The funds “helped rural hospitals with the immediate storm,” said Don Williamson, MD, president of the Alabama Hospital Association. Nearly 80% of Alabama’s rural hospitals began the year with negative balance sheets and about 8 days’ worth of cash on hand.
Before the pandemic hit this year, hundreds of rural hospitals “were just trying to keep their doors open,” said Maggie Elehwany, vice president of government affairs with the National Rural Health Association. Then an estimated 70% of their income stopped as patients avoided the emergency room, doctor’s appointments, and elective surgeries.
“It was devastating,” Ms. Elehwany said.
Paul Taylor, chief executive of a 25-bed critical-access hospital and outpatient clinics in northwestern Arkansas, accepted millions in grants and loan money Congress approved this spring, largely through the CARES (Coronavirus Aid, Relief, and Economic Security) Act.
“For us, this was survival money and we spent it already,” Mr. Taylor said. With those funds, Ozarks Community Hospital increased surge capacity, expanding from 25 beds to 50 beds, adding negative pressure rooms and buying six ventilators. Taylor also ramped up COVID-19 testing at his hospital and clinics, located near some meat-processing plants.
Throughout June and July, Ozarks tested 1,000 patients a day and reported a 20% positive rate. The rate dropped to 16.9% in late July. But patients continue to avoid routine care.
Mr. Taylor said revenue is still constrained and he does not know how he will pay back $8 million that he borrowed from Medicare. The program allowed hospitals to borrow against future payments from the federal government, but stipulated that repayment would begin within 120 days.
For Mr. Taylor, this seems impossible. Medicare makes up 40% of Ozarks’ income. And he has to pay the loan back before he gets any more payments from Medicare. He’s hoping to refinance the hospital’s mortgage.
“If I get no relief and they take the money ... we won’t still be open,” Mr. Taylor said. Ozarks provides 625 jobs and serves an area with a population of about 75,000.
There are 1,300 small critical-access hospitals like Ozarks in rural America, and of those, 859 took advantage of the Medicare loans, sending about $3.1 billion into the local communities. But many rural communities have not yet experienced a surge in coronavirus cases – national leaders fear it will come as part of a new phase.
“There are pockets of rural America who say, ‘We haven’t seen a single COVID patient yet and we do not believe it’s real,’ ” Mr. Taylor said. “They will get hit sooner or later.”
Across the country, the reduced patient numbers and increased spending required to fight and prepare for the coronavirus was “like a knife cutting into a hospital’s blood supply,” said Ge Bai, PhD, associate professor of health policy and management at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Dr. Bai said the way the federal government reimbursed small rural hospitals through federal programs like Medicare before the pandemic was faulty and inefficient. “They are too weak to survive,” she said.
In rural Texas, about 2 hours from Dallas, Titus Regional Medical Center chief executive officer Terry Scoggin cut staff and furloughed workers even as his rural hospital faced down the pandemic. Titus Regional lost about $4 million last fiscal year and broke even each of the three years before that.
Mr. Scoggin said he did not cut from his clinical staff, though. Titus is now facing its second surge of the virus in the community. “The last 7 days, we’ve been testing 30% positive,” he said, including the case of his father, who contracted it at a nursing home and survived.
“It’s personal and this is real,” Mr. Scoggin said. “You know the people who are infected. You know the people who are passing away.”
Of his roughly 700 employees, 48 have tested positive for the virus and 1 has died. They are short on testing kits, medication, and supplies.
“Right now the staff is strained,” Mr. Scoggin said. “I’ve been blown away by their selflessness and unbelievable spirit. We’re resilient, we’re nimble, and we will make it. We don’t have a choice.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Jerome Antone said he is one of the lucky ones.
After becoming ill with COVID-19, Mr. Antone was hospitalized only 65 miles away from his small Alabama town. He is the mayor of Georgiana – population 1,700.
“It hit our rural community so rabid,” Mr. Antone said. The town’s hospital closed last year. If hospitals in nearby communities don’t have beds available, “you may have to go 4 or 5 hours away.”
Eighteen rural hospitals closed last year and the first 3 months of 2020 were “really big months,” said Mark Holmes, PhD, director of the Cecil G. Sheps Center for Health Services Research at the University of North Carolina at Chapel Hill. Many of the losses are in Southern states like Florida and Texas. More than 170 rural hospitals have closed nationwide since 2005, according to data collected by the Sheps Center.
It’s a dangerous scenario. “We know that a closure leads to higher mortality pretty quickly” among the populations served, said Dr. Holmes, who is also a professor at UNC Gillings School of Global Public Health. “That’s pretty clear.”
One 2019 study found that death rates in the surrounding communities increase nearly 6% after a rural hospital closes – and that’s when there’s not a pandemic.
Add to that what is known about the coronavirus: People who are obese or live with diabetes, hypertension, asthma, and other underlying health issues are more susceptible to COVID-19. Rural areas tend to have higher rates of these conditions. And rural residents are more likely to be older, sicker and poorer than those in urban areas. All this leaves rural communities particularly vulnerable to the coronavirus.
Congress approved billions in federal relief funds for health care providers. Initially, federal officials based what a hospital would get on its Medicare payments, but by late April they heeded criticism and carved out funds for rural hospitals and COVID-19 hot spots. Rural hospitals leapt at the chance to shore up already-negative budgets and prepare for the pandemic.
The funds “helped rural hospitals with the immediate storm,” said Don Williamson, MD, president of the Alabama Hospital Association. Nearly 80% of Alabama’s rural hospitals began the year with negative balance sheets and about 8 days’ worth of cash on hand.
Before the pandemic hit this year, hundreds of rural hospitals “were just trying to keep their doors open,” said Maggie Elehwany, vice president of government affairs with the National Rural Health Association. Then an estimated 70% of their income stopped as patients avoided the emergency room, doctor’s appointments, and elective surgeries.
“It was devastating,” Ms. Elehwany said.
Paul Taylor, chief executive of a 25-bed critical-access hospital and outpatient clinics in northwestern Arkansas, accepted millions in grants and loan money Congress approved this spring, largely through the CARES (Coronavirus Aid, Relief, and Economic Security) Act.
“For us, this was survival money and we spent it already,” Mr. Taylor said. With those funds, Ozarks Community Hospital increased surge capacity, expanding from 25 beds to 50 beds, adding negative pressure rooms and buying six ventilators. Taylor also ramped up COVID-19 testing at his hospital and clinics, located near some meat-processing plants.
Throughout June and July, Ozarks tested 1,000 patients a day and reported a 20% positive rate. The rate dropped to 16.9% in late July. But patients continue to avoid routine care.
Mr. Taylor said revenue is still constrained and he does not know how he will pay back $8 million that he borrowed from Medicare. The program allowed hospitals to borrow against future payments from the federal government, but stipulated that repayment would begin within 120 days.
For Mr. Taylor, this seems impossible. Medicare makes up 40% of Ozarks’ income. And he has to pay the loan back before he gets any more payments from Medicare. He’s hoping to refinance the hospital’s mortgage.
“If I get no relief and they take the money ... we won’t still be open,” Mr. Taylor said. Ozarks provides 625 jobs and serves an area with a population of about 75,000.
There are 1,300 small critical-access hospitals like Ozarks in rural America, and of those, 859 took advantage of the Medicare loans, sending about $3.1 billion into the local communities. But many rural communities have not yet experienced a surge in coronavirus cases – national leaders fear it will come as part of a new phase.
“There are pockets of rural America who say, ‘We haven’t seen a single COVID patient yet and we do not believe it’s real,’ ” Mr. Taylor said. “They will get hit sooner or later.”
Across the country, the reduced patient numbers and increased spending required to fight and prepare for the coronavirus was “like a knife cutting into a hospital’s blood supply,” said Ge Bai, PhD, associate professor of health policy and management at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Dr. Bai said the way the federal government reimbursed small rural hospitals through federal programs like Medicare before the pandemic was faulty and inefficient. “They are too weak to survive,” she said.
In rural Texas, about 2 hours from Dallas, Titus Regional Medical Center chief executive officer Terry Scoggin cut staff and furloughed workers even as his rural hospital faced down the pandemic. Titus Regional lost about $4 million last fiscal year and broke even each of the three years before that.
Mr. Scoggin said he did not cut from his clinical staff, though. Titus is now facing its second surge of the virus in the community. “The last 7 days, we’ve been testing 30% positive,” he said, including the case of his father, who contracted it at a nursing home and survived.
“It’s personal and this is real,” Mr. Scoggin said. “You know the people who are infected. You know the people who are passing away.”
Of his roughly 700 employees, 48 have tested positive for the virus and 1 has died. They are short on testing kits, medication, and supplies.
“Right now the staff is strained,” Mr. Scoggin said. “I’ve been blown away by their selflessness and unbelievable spirit. We’re resilient, we’re nimble, and we will make it. We don’t have a choice.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Jerome Antone said he is one of the lucky ones.
After becoming ill with COVID-19, Mr. Antone was hospitalized only 65 miles away from his small Alabama town. He is the mayor of Georgiana – population 1,700.
“It hit our rural community so rabid,” Mr. Antone said. The town’s hospital closed last year. If hospitals in nearby communities don’t have beds available, “you may have to go 4 or 5 hours away.”
Eighteen rural hospitals closed last year and the first 3 months of 2020 were “really big months,” said Mark Holmes, PhD, director of the Cecil G. Sheps Center for Health Services Research at the University of North Carolina at Chapel Hill. Many of the losses are in Southern states like Florida and Texas. More than 170 rural hospitals have closed nationwide since 2005, according to data collected by the Sheps Center.
It’s a dangerous scenario. “We know that a closure leads to higher mortality pretty quickly” among the populations served, said Dr. Holmes, who is also a professor at UNC Gillings School of Global Public Health. “That’s pretty clear.”
One 2019 study found that death rates in the surrounding communities increase nearly 6% after a rural hospital closes – and that’s when there’s not a pandemic.
Add to that what is known about the coronavirus: People who are obese or live with diabetes, hypertension, asthma, and other underlying health issues are more susceptible to COVID-19. Rural areas tend to have higher rates of these conditions. And rural residents are more likely to be older, sicker and poorer than those in urban areas. All this leaves rural communities particularly vulnerable to the coronavirus.
Congress approved billions in federal relief funds for health care providers. Initially, federal officials based what a hospital would get on its Medicare payments, but by late April they heeded criticism and carved out funds for rural hospitals and COVID-19 hot spots. Rural hospitals leapt at the chance to shore up already-negative budgets and prepare for the pandemic.
The funds “helped rural hospitals with the immediate storm,” said Don Williamson, MD, president of the Alabama Hospital Association. Nearly 80% of Alabama’s rural hospitals began the year with negative balance sheets and about 8 days’ worth of cash on hand.
Before the pandemic hit this year, hundreds of rural hospitals “were just trying to keep their doors open,” said Maggie Elehwany, vice president of government affairs with the National Rural Health Association. Then an estimated 70% of their income stopped as patients avoided the emergency room, doctor’s appointments, and elective surgeries.
“It was devastating,” Ms. Elehwany said.
Paul Taylor, chief executive of a 25-bed critical-access hospital and outpatient clinics in northwestern Arkansas, accepted millions in grants and loan money Congress approved this spring, largely through the CARES (Coronavirus Aid, Relief, and Economic Security) Act.
“For us, this was survival money and we spent it already,” Mr. Taylor said. With those funds, Ozarks Community Hospital increased surge capacity, expanding from 25 beds to 50 beds, adding negative pressure rooms and buying six ventilators. Taylor also ramped up COVID-19 testing at his hospital and clinics, located near some meat-processing plants.
Throughout June and July, Ozarks tested 1,000 patients a day and reported a 20% positive rate. The rate dropped to 16.9% in late July. But patients continue to avoid routine care.
Mr. Taylor said revenue is still constrained and he does not know how he will pay back $8 million that he borrowed from Medicare. The program allowed hospitals to borrow against future payments from the federal government, but stipulated that repayment would begin within 120 days.
For Mr. Taylor, this seems impossible. Medicare makes up 40% of Ozarks’ income. And he has to pay the loan back before he gets any more payments from Medicare. He’s hoping to refinance the hospital’s mortgage.
“If I get no relief and they take the money ... we won’t still be open,” Mr. Taylor said. Ozarks provides 625 jobs and serves an area with a population of about 75,000.
There are 1,300 small critical-access hospitals like Ozarks in rural America, and of those, 859 took advantage of the Medicare loans, sending about $3.1 billion into the local communities. But many rural communities have not yet experienced a surge in coronavirus cases – national leaders fear it will come as part of a new phase.
“There are pockets of rural America who say, ‘We haven’t seen a single COVID patient yet and we do not believe it’s real,’ ” Mr. Taylor said. “They will get hit sooner or later.”
Across the country, the reduced patient numbers and increased spending required to fight and prepare for the coronavirus was “like a knife cutting into a hospital’s blood supply,” said Ge Bai, PhD, associate professor of health policy and management at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Dr. Bai said the way the federal government reimbursed small rural hospitals through federal programs like Medicare before the pandemic was faulty and inefficient. “They are too weak to survive,” she said.
In rural Texas, about 2 hours from Dallas, Titus Regional Medical Center chief executive officer Terry Scoggin cut staff and furloughed workers even as his rural hospital faced down the pandemic. Titus Regional lost about $4 million last fiscal year and broke even each of the three years before that.
Mr. Scoggin said he did not cut from his clinical staff, though. Titus is now facing its second surge of the virus in the community. “The last 7 days, we’ve been testing 30% positive,” he said, including the case of his father, who contracted it at a nursing home and survived.
“It’s personal and this is real,” Mr. Scoggin said. “You know the people who are infected. You know the people who are passing away.”
Of his roughly 700 employees, 48 have tested positive for the virus and 1 has died. They are short on testing kits, medication, and supplies.
“Right now the staff is strained,” Mr. Scoggin said. “I’ve been blown away by their selflessness and unbelievable spirit. We’re resilient, we’re nimble, and we will make it. We don’t have a choice.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Being a pediatric hospitalist during the COVID-19 pandemic
“Times of great calamity and confusion have been productive for the greatest minds. The purest ore is produced from the hottest furnace. The brightest thunderbolt is elicited from the darkest storm.” – Charles Caleb Colton
I walk inside the pediatric unit of our hospital, only to be welcomed by an eerie silence. There are a handful of nurses at the nursing station, faces covered with masks sitting 6 feet apart and quietly working on their computers. The resident work lounge also depicts a similar picture of emptiness. Just over a month ago, these halls were bustling with children, parents, consultants, and a host of ancillary staff. I recall times in which I was running around from one patient room to another talking to families and attending to patient needs. For the past 2 months I have often spent hours alone in my office waiting to see a patient. This is the new norm for many of us.
Across the board in hospitals, pediatric census has dropped since the start of the COVID-19 pandemic. Reasons for these are nonspecific but may include fear among parents of “exposure” to the virus by bringing their sick children to the hospital to get evaluated for other concerns. A few patients that we have seen in our hospital are sicker when they have arrived because their parents avoided seeking medical care earlier, plagued by the same fear. Social distancing and school closure have also limited the amount of infectious diseases going around, which are responsible for a bulk of pediatric admissions.
While many of us are still coming in to see the limited number of patients we have, we are not in the true sense frontline providers during this pandemic. There have been limited cases of COVID-19 in children, most of which – fortunately – present with mild symptoms. Although multisystem inflammatory syndrome in children (MIS-C) is a new disease that COVID-19 has brought us closer to, many of us have yet to see our first case because of its rarity.
I have read through the news daily in the past few months to find many adult provider physicians succumbing to COVID-19 and felt a pang of guilt. My social media is full of heartbreaking stories as adult hospitalists are having difficult conversations with families and supporting them through this unknown territory, often sacrificing their own safety. I feel so proud of them and my profession. My heart tells me, though, I personally may not be living up to the true calling I was expected to have as a physician.
As pediatric hospitalists, while we sit and wait for this pandemic to pass, we have been ruminating on and anxious about our future. As census drops, there is a financial strain that many of us are feeling. Job cuts and furloughing of health care workers in our surroundings leave us with a sense of insecurity and low morale. Many small inpatient pediatric units have had to be shut down temporarily either so they could be used for adult patients or because of lack of pediatric patients. Limiting staffing to avoid exposure and cohorting providers has also been a challenge.
A big question that has risen in these times is how to ensure productivity and stay useful while at the same time being prepared for the unknown that lies ahead. The economics of medicine is staring hard at our years of hard work, questioning the need for our specialty in the first place.
In smaller community settings, the closure of pediatric units has put an additional strain on the overall framework of the community, parents, and referring primary care providers. With the absence of local resources, children who have needed care have had to be transferred to bigger referral centers that are still taking care of pediatric patients. On one end of the spectrum there is concern for pediatric inpatient units not being productive enough for the hospital, but that coexists with a worry that, as we pass through this pandemic, we could see more hospitalizations for vaccine-preventable illnesses, child abuse/medical neglect, and respiratory syncytial virus plus COVID.
The question remains about how best to cope and use this time of uncertainty to be productive and prepare for the worst. A few solutions and suggestions are highlighted below.
- Helping adult providers: Many pediatric hospitalist colleagues in highly affected states have filled the increasing need for clinicians and taken care of adult patients. As pediatric units have closed, providers have continued to offer care where it is needed. Pediatric hospitalists have used this time to take urgent refresher courses in advanced cardiac life support and adult critical care. In states that are not as severely hit, many pediatric hospitalists have utilized this time to plan and prepare protocols for the future as information continues coming in regarding MIS-C and COVID-19 in pediatric patients.
- Use of telemedicine: With the ease in restrictions for use of telemedicine in many states, pediatric hospitalists can consider using it to restructure their staffing model whenever feasible. This can help in cohorting and allowing high risk and quarantined providers to work from home. This model simultaneously provides opportunities for pediatric hospitalists to continue providing their services, while at the same time decreasing financial burden on their institution.
- Reaching out to the community: Engaging with the community during these times can help ensure services and options remain available to our referral providers and patients for pediatric services. Information about COVID-19 can be widely disseminated. We can also play our part by continuing to encourage parents in our maximum capacity to obtain care for their children when needed and to not avoid the hospital because of fears of exposure.
- Supporting each other: There is no doubt that these times are unsettling for the pediatric hospitalist community, and the uncertainty that surrounds us can feel crippling. Strong team building is imperative in these times. While we may not be frequently meeting in work lounges and sharing meals, a good sense of support and camaraderie will go a long way in building morale for the future. Seeking mental health resources if needed is essential for us and should not be looked at with shame or guilt. This is something that many of us have never seen before, and it is okay to ask for help. Seeking help is and always will be a sign of strength.
Today, as I envision myself walking in the hospital on the other side of this pandemic I see a cheerful pediatric unit, smiling faces without masks, my 3-year-old patient cruising around the hallways in a toy car, our therapy dog walking around bringing joy to many, and many healthy patients feeling better and ready to go home. A time when we are not scared to hug each other, shake hands, or share emotion. When our teams are stronger and more well bonded. A time when parents are not scared to bring their sick children to the hospital. Will it be many months before this happens? I don’t know. But I do know that the children I take care of are known for their resilience. I will live up to them today by practicing the same.
Dr. Fatima is a pediatric hospitalist at Wesley Children’s Hospital and assistant professor of pediatrics at Kansas University School of Medicine, both in Wichita. Her research interests include medical errors, medical education, and high-value care.
“Times of great calamity and confusion have been productive for the greatest minds. The purest ore is produced from the hottest furnace. The brightest thunderbolt is elicited from the darkest storm.” – Charles Caleb Colton
I walk inside the pediatric unit of our hospital, only to be welcomed by an eerie silence. There are a handful of nurses at the nursing station, faces covered with masks sitting 6 feet apart and quietly working on their computers. The resident work lounge also depicts a similar picture of emptiness. Just over a month ago, these halls were bustling with children, parents, consultants, and a host of ancillary staff. I recall times in which I was running around from one patient room to another talking to families and attending to patient needs. For the past 2 months I have often spent hours alone in my office waiting to see a patient. This is the new norm for many of us.
Across the board in hospitals, pediatric census has dropped since the start of the COVID-19 pandemic. Reasons for these are nonspecific but may include fear among parents of “exposure” to the virus by bringing their sick children to the hospital to get evaluated for other concerns. A few patients that we have seen in our hospital are sicker when they have arrived because their parents avoided seeking medical care earlier, plagued by the same fear. Social distancing and school closure have also limited the amount of infectious diseases going around, which are responsible for a bulk of pediatric admissions.
While many of us are still coming in to see the limited number of patients we have, we are not in the true sense frontline providers during this pandemic. There have been limited cases of COVID-19 in children, most of which – fortunately – present with mild symptoms. Although multisystem inflammatory syndrome in children (MIS-C) is a new disease that COVID-19 has brought us closer to, many of us have yet to see our first case because of its rarity.
I have read through the news daily in the past few months to find many adult provider physicians succumbing to COVID-19 and felt a pang of guilt. My social media is full of heartbreaking stories as adult hospitalists are having difficult conversations with families and supporting them through this unknown territory, often sacrificing their own safety. I feel so proud of them and my profession. My heart tells me, though, I personally may not be living up to the true calling I was expected to have as a physician.
As pediatric hospitalists, while we sit and wait for this pandemic to pass, we have been ruminating on and anxious about our future. As census drops, there is a financial strain that many of us are feeling. Job cuts and furloughing of health care workers in our surroundings leave us with a sense of insecurity and low morale. Many small inpatient pediatric units have had to be shut down temporarily either so they could be used for adult patients or because of lack of pediatric patients. Limiting staffing to avoid exposure and cohorting providers has also been a challenge.
A big question that has risen in these times is how to ensure productivity and stay useful while at the same time being prepared for the unknown that lies ahead. The economics of medicine is staring hard at our years of hard work, questioning the need for our specialty in the first place.
In smaller community settings, the closure of pediatric units has put an additional strain on the overall framework of the community, parents, and referring primary care providers. With the absence of local resources, children who have needed care have had to be transferred to bigger referral centers that are still taking care of pediatric patients. On one end of the spectrum there is concern for pediatric inpatient units not being productive enough for the hospital, but that coexists with a worry that, as we pass through this pandemic, we could see more hospitalizations for vaccine-preventable illnesses, child abuse/medical neglect, and respiratory syncytial virus plus COVID.
The question remains about how best to cope and use this time of uncertainty to be productive and prepare for the worst. A few solutions and suggestions are highlighted below.
- Helping adult providers: Many pediatric hospitalist colleagues in highly affected states have filled the increasing need for clinicians and taken care of adult patients. As pediatric units have closed, providers have continued to offer care where it is needed. Pediatric hospitalists have used this time to take urgent refresher courses in advanced cardiac life support and adult critical care. In states that are not as severely hit, many pediatric hospitalists have utilized this time to plan and prepare protocols for the future as information continues coming in regarding MIS-C and COVID-19 in pediatric patients.
- Use of telemedicine: With the ease in restrictions for use of telemedicine in many states, pediatric hospitalists can consider using it to restructure their staffing model whenever feasible. This can help in cohorting and allowing high risk and quarantined providers to work from home. This model simultaneously provides opportunities for pediatric hospitalists to continue providing their services, while at the same time decreasing financial burden on their institution.
- Reaching out to the community: Engaging with the community during these times can help ensure services and options remain available to our referral providers and patients for pediatric services. Information about COVID-19 can be widely disseminated. We can also play our part by continuing to encourage parents in our maximum capacity to obtain care for their children when needed and to not avoid the hospital because of fears of exposure.
- Supporting each other: There is no doubt that these times are unsettling for the pediatric hospitalist community, and the uncertainty that surrounds us can feel crippling. Strong team building is imperative in these times. While we may not be frequently meeting in work lounges and sharing meals, a good sense of support and camaraderie will go a long way in building morale for the future. Seeking mental health resources if needed is essential for us and should not be looked at with shame or guilt. This is something that many of us have never seen before, and it is okay to ask for help. Seeking help is and always will be a sign of strength.
Today, as I envision myself walking in the hospital on the other side of this pandemic I see a cheerful pediatric unit, smiling faces without masks, my 3-year-old patient cruising around the hallways in a toy car, our therapy dog walking around bringing joy to many, and many healthy patients feeling better and ready to go home. A time when we are not scared to hug each other, shake hands, or share emotion. When our teams are stronger and more well bonded. A time when parents are not scared to bring their sick children to the hospital. Will it be many months before this happens? I don’t know. But I do know that the children I take care of are known for their resilience. I will live up to them today by practicing the same.
Dr. Fatima is a pediatric hospitalist at Wesley Children’s Hospital and assistant professor of pediatrics at Kansas University School of Medicine, both in Wichita. Her research interests include medical errors, medical education, and high-value care.
“Times of great calamity and confusion have been productive for the greatest minds. The purest ore is produced from the hottest furnace. The brightest thunderbolt is elicited from the darkest storm.” – Charles Caleb Colton
I walk inside the pediatric unit of our hospital, only to be welcomed by an eerie silence. There are a handful of nurses at the nursing station, faces covered with masks sitting 6 feet apart and quietly working on their computers. The resident work lounge also depicts a similar picture of emptiness. Just over a month ago, these halls were bustling with children, parents, consultants, and a host of ancillary staff. I recall times in which I was running around from one patient room to another talking to families and attending to patient needs. For the past 2 months I have often spent hours alone in my office waiting to see a patient. This is the new norm for many of us.
Across the board in hospitals, pediatric census has dropped since the start of the COVID-19 pandemic. Reasons for these are nonspecific but may include fear among parents of “exposure” to the virus by bringing their sick children to the hospital to get evaluated for other concerns. A few patients that we have seen in our hospital are sicker when they have arrived because their parents avoided seeking medical care earlier, plagued by the same fear. Social distancing and school closure have also limited the amount of infectious diseases going around, which are responsible for a bulk of pediatric admissions.
While many of us are still coming in to see the limited number of patients we have, we are not in the true sense frontline providers during this pandemic. There have been limited cases of COVID-19 in children, most of which – fortunately – present with mild symptoms. Although multisystem inflammatory syndrome in children (MIS-C) is a new disease that COVID-19 has brought us closer to, many of us have yet to see our first case because of its rarity.
I have read through the news daily in the past few months to find many adult provider physicians succumbing to COVID-19 and felt a pang of guilt. My social media is full of heartbreaking stories as adult hospitalists are having difficult conversations with families and supporting them through this unknown territory, often sacrificing their own safety. I feel so proud of them and my profession. My heart tells me, though, I personally may not be living up to the true calling I was expected to have as a physician.
As pediatric hospitalists, while we sit and wait for this pandemic to pass, we have been ruminating on and anxious about our future. As census drops, there is a financial strain that many of us are feeling. Job cuts and furloughing of health care workers in our surroundings leave us with a sense of insecurity and low morale. Many small inpatient pediatric units have had to be shut down temporarily either so they could be used for adult patients or because of lack of pediatric patients. Limiting staffing to avoid exposure and cohorting providers has also been a challenge.
A big question that has risen in these times is how to ensure productivity and stay useful while at the same time being prepared for the unknown that lies ahead. The economics of medicine is staring hard at our years of hard work, questioning the need for our specialty in the first place.
In smaller community settings, the closure of pediatric units has put an additional strain on the overall framework of the community, parents, and referring primary care providers. With the absence of local resources, children who have needed care have had to be transferred to bigger referral centers that are still taking care of pediatric patients. On one end of the spectrum there is concern for pediatric inpatient units not being productive enough for the hospital, but that coexists with a worry that, as we pass through this pandemic, we could see more hospitalizations for vaccine-preventable illnesses, child abuse/medical neglect, and respiratory syncytial virus plus COVID.
The question remains about how best to cope and use this time of uncertainty to be productive and prepare for the worst. A few solutions and suggestions are highlighted below.
- Helping adult providers: Many pediatric hospitalist colleagues in highly affected states have filled the increasing need for clinicians and taken care of adult patients. As pediatric units have closed, providers have continued to offer care where it is needed. Pediatric hospitalists have used this time to take urgent refresher courses in advanced cardiac life support and adult critical care. In states that are not as severely hit, many pediatric hospitalists have utilized this time to plan and prepare protocols for the future as information continues coming in regarding MIS-C and COVID-19 in pediatric patients.
- Use of telemedicine: With the ease in restrictions for use of telemedicine in many states, pediatric hospitalists can consider using it to restructure their staffing model whenever feasible. This can help in cohorting and allowing high risk and quarantined providers to work from home. This model simultaneously provides opportunities for pediatric hospitalists to continue providing their services, while at the same time decreasing financial burden on their institution.
- Reaching out to the community: Engaging with the community during these times can help ensure services and options remain available to our referral providers and patients for pediatric services. Information about COVID-19 can be widely disseminated. We can also play our part by continuing to encourage parents in our maximum capacity to obtain care for their children when needed and to not avoid the hospital because of fears of exposure.
- Supporting each other: There is no doubt that these times are unsettling for the pediatric hospitalist community, and the uncertainty that surrounds us can feel crippling. Strong team building is imperative in these times. While we may not be frequently meeting in work lounges and sharing meals, a good sense of support and camaraderie will go a long way in building morale for the future. Seeking mental health resources if needed is essential for us and should not be looked at with shame or guilt. This is something that many of us have never seen before, and it is okay to ask for help. Seeking help is and always will be a sign of strength.
Today, as I envision myself walking in the hospital on the other side of this pandemic I see a cheerful pediatric unit, smiling faces without masks, my 3-year-old patient cruising around the hallways in a toy car, our therapy dog walking around bringing joy to many, and many healthy patients feeling better and ready to go home. A time when we are not scared to hug each other, shake hands, or share emotion. When our teams are stronger and more well bonded. A time when parents are not scared to bring their sick children to the hospital. Will it be many months before this happens? I don’t know. But I do know that the children I take care of are known for their resilience. I will live up to them today by practicing the same.
Dr. Fatima is a pediatric hospitalist at Wesley Children’s Hospital and assistant professor of pediatrics at Kansas University School of Medicine, both in Wichita. Her research interests include medical errors, medical education, and high-value care.
When viruses collide: Flu season during pandemic
The medical community is about to find out how prepared it is for the double whammy of influenza and COVID-19 that has been predicted for the fall of 2020. The complexities of diagnosis, management of vulnerable patients, and overflowing medical centers that have made the COVID-19 crisis so brutal may all be exacerbated by the arrival of seasonal influenza.
Lewis Jay Kaplan, MD, FCCP, a critical care surgeon at the University of Pennsylvania, Philadelphia, has seen his share of critically ill COVID-19 patients in the surgical ICU that he oversees. He’s approaching the upcoming flu season, poised to collide with the ongoing COVID-19 pandemic, ready to listen to each patient’s story to distinguish one from the other and determine treatment.
“The patients that have underlying comorbidities all have a story, and it’s up to you to figure out which chapter you’re in and how far along you happen to be,” he said. “It’s a very interesting approach to care, medical storytelling.”
With flu season closing in, pulmonologists are ruminating about how they’ll distinguish symptoms of COVID-19 and traditional influenza and how they’ll manage the most vulnerable patients, namely those with underlying respiratory disease and children. Influenza kills 12,000-61,000 people a year, according to the Centers for Disease Control, and results in 140,000-810,00 hospitalizations. Having a flu season in the midst of a pandemic of a disease with multiple overlapping symptoms threatens to overwhelm practitioners, hospitals, and the health system.
Dr. Kaplan said each patient’s story can point to the correct clinical approach. “Instead of just sharing data when you are on rounds, you’re really telling someone’s story.” It arises from a series of questions about how the disease has impacted them, specifics of their presentation, how their signs and symptoms differ from the usual, and how they responded to treatment. “It also helps you to then take what you’re doing, which can seem very, very complicated to individuals who are not medically sophisticated, and then help them to understand why you’re doing what you’re doing at this point.”
That can help get through to a patient with respiratory disease who insists he or she has or doesn’t have COVID-19 rather than the flu. “They form a different group that brings with them different fears and concerns, and you have to help them navigate that, too: all of this data and your decision-making around testing and admissions, and what you can omit doing and what you must do help them to navigate their own story,” Dr. Kaplan said.
Benjamin D. Singer, MD, a pulmonologist at Northwestern University, Chicago, authored an editorial in Science Advances that addressed four factors that will determine the scope of flu spread in the upcoming season: rate of transmission; vaccination rates; coinfection rates; and health disparities in minority populations, which are prone to higher rates of flu as well as COVID-19.
Flu vaccine ‘extra important’
The convergence of COVID-19 and influenza has the potential to overwhelm the health system, said Daniel A. Solomon, MD, of Brigham and Women’s in Boston. He coauthored a JAMA Insights clinical update on flu season during the COVID-19 pandemic that lists distinguishing and overlapping signs and symptoms of the two diseases.
The flu vaccine, he said, is “extra important this year,” especially in patients with existing respiratory disease, but COVID-19 has thrown up barriers to vaccination. Telemedicine has supplanted office visits. “People may miss that easy-touch opportunity to get the flu vaccine, so we have to be creative about making the flu vaccine highly accessible, maybe in nontraditional ways,” Dr. Solomon said. Some ideas he offered are pop-up vaccine fairs at schools and churches.
But just as COVID-19 may hinder flu vaccines, it may also be helping to mitigate flu transmission. “The interesting thing about transmission of the flu is that it’s transmitted the same way COVID is, so if we actually know how to decrease transmission of COVID, which we do – we’ve done it – we can actually decrease transmission of influenza as well,” Dr. Solomon said. Studies out of Hong Kong and Japan have reported a reduction in influenza cases during COVID-19 outbreaks in those places (Lancet Public Health. 2020;5:e279-88; JAMA. 2020;323:1969-71).
Risks of coinfection
About one in four COVID-19 patients have been diagnosed with an additional respiratory infection, including influenza (JAMA. 2020:323:2085-6). Pulmonologists must keep that in mind when managing COVID-19 suspects, said Dr. Singer.
“While it is true that most of the time COVID-19 travels alone, we have numerous examples in the literature and in our own experience that COVID-19 is accompanied by either another virus or another bacterial infection, including influenza,” Dr. Singer said. “The distinction is important. One is just for diagnostic reasons and public reporting reasons, but also because flu and COVID-19 have different requirements for how you care for patients in terms of the health system.”
Clinical suspicion for coinfection should remain high if the community spread of both COVID-19 and influenza is high, said Megan Conroy, MD, chief pulmonary and critical care fellow at Ohio State University, Columbus. “As the coronavirus first took hold in the United States in March 2020, we were at the tail end of influenza season, so it’s hard to predict what the upcoming influenza season will really look like with regards to coinfection.”
Distinguishing COVID-19 from flu
Multiple signs and symptoms between COVID-19 and the flu overlap. They include fever, chills, headache, myalgia, cough, and fatigue. Nasal congestion and sore throat are characteristic of the flu; shortness of breath and loss of the sense of smell have been widely reported in COVID-19. “While many upper respiratory infections can result in loss of smell, this may be more prevalent in COVID-19,” Dr. Conroy said. Other symptoms unique to COVID-19 are GI symptoms such as diarrhea and skin rashes such as acral ischemia.
Testing, however, is the cornerstone of the differential diagnosis. “You can’t confidently distinguish between them on symptoms alone,” Dr. Conroy added.
“I think the challenge we’ll face as clinicians, is caring for people with nonspecific symptoms of a respiratory viral illness, especially in the early phase of the illness,” said Dr. Solomon.
But even after that, symptoms can be difficult to distinguish.
“Later in the illness, COVID is more associated with a hypercoagulable state,” he said. “It is more associated with viral pneumonia on chest imaging, like the diffuse ground-glass infiltrates that we’ve all gotten used to seeing – but flu can do both of those things as well. So, without a test, it’s impossible to distinguish between the two infections in the clinic.”
But testing can have its shortcomings when flu season clashes with the COVID-19 pandemic. “Getting the test is not the same as getting the test results,” Dr. Solomon added. “Though a lot of people can get a test, if it takes 7 or 8 days to get the test result back, the result is useless.”
Widespread, rapid testing also depends on having adequate supplies of viral media transport and swabs. “I think that this is what we should be focusing on now: scaling up access to rapid turnaround testing,” he said. Distinguishing between the two is also important to preserve hospital resources. COVID-19 has more rigorous standards than flu for personal protective equipment and isolation of patients within the hospital.
Having chronic lung disease isn’t necessarily a risk factor for contracting COVID-19 or the flu, or both, Dr. Solomon said. “It’s a risk factor for having severe disease.” Again, he noted that flu vaccines are still necessary in these patients, as well as patients of advanced age and underlying medical conditions such as heart disease, diabetes, and obesity.
In managing children, it’s important to keep in mind that they communicate differently about their illnesses than adults, said Dr. Kaplan. “They may not have the words to tell you the same kind of thing that the adult tells you.” That’s where family members can help to flesh out the history. “They may present with an initially much milder form, if you will, where they’re not as critical up front, but then that small proportion of them comes back with the multi-inflammatory syndrome and then they are profoundly ill.”
Younger people make up a larger share of COVID-19 patients now, compared with the initial wave that hit the Northeast in the spring, Dr. Kaplan said. “We don’t know if that’s because the virus is a little different or the people that are getting sick are a little bit different.”
The COVID-19 strain now emerging may be less virulent than the strain that hit in early spring, he said. “That doesn’t mean that there aren’t still profoundly critical ill people with COVID of many different age ranges, that is true, but there are a lot of people that we now see will test positive, but aren’t really as profoundly ill as when it first landed here in the United States.”
That may be somewhat welcome as flu season arrives.
The physicians interviewed have no relevant disclosures.
The medical community is about to find out how prepared it is for the double whammy of influenza and COVID-19 that has been predicted for the fall of 2020. The complexities of diagnosis, management of vulnerable patients, and overflowing medical centers that have made the COVID-19 crisis so brutal may all be exacerbated by the arrival of seasonal influenza.
Lewis Jay Kaplan, MD, FCCP, a critical care surgeon at the University of Pennsylvania, Philadelphia, has seen his share of critically ill COVID-19 patients in the surgical ICU that he oversees. He’s approaching the upcoming flu season, poised to collide with the ongoing COVID-19 pandemic, ready to listen to each patient’s story to distinguish one from the other and determine treatment.
“The patients that have underlying comorbidities all have a story, and it’s up to you to figure out which chapter you’re in and how far along you happen to be,” he said. “It’s a very interesting approach to care, medical storytelling.”
With flu season closing in, pulmonologists are ruminating about how they’ll distinguish symptoms of COVID-19 and traditional influenza and how they’ll manage the most vulnerable patients, namely those with underlying respiratory disease and children. Influenza kills 12,000-61,000 people a year, according to the Centers for Disease Control, and results in 140,000-810,00 hospitalizations. Having a flu season in the midst of a pandemic of a disease with multiple overlapping symptoms threatens to overwhelm practitioners, hospitals, and the health system.
Dr. Kaplan said each patient’s story can point to the correct clinical approach. “Instead of just sharing data when you are on rounds, you’re really telling someone’s story.” It arises from a series of questions about how the disease has impacted them, specifics of their presentation, how their signs and symptoms differ from the usual, and how they responded to treatment. “It also helps you to then take what you’re doing, which can seem very, very complicated to individuals who are not medically sophisticated, and then help them to understand why you’re doing what you’re doing at this point.”
That can help get through to a patient with respiratory disease who insists he or she has or doesn’t have COVID-19 rather than the flu. “They form a different group that brings with them different fears and concerns, and you have to help them navigate that, too: all of this data and your decision-making around testing and admissions, and what you can omit doing and what you must do help them to navigate their own story,” Dr. Kaplan said.
Benjamin D. Singer, MD, a pulmonologist at Northwestern University, Chicago, authored an editorial in Science Advances that addressed four factors that will determine the scope of flu spread in the upcoming season: rate of transmission; vaccination rates; coinfection rates; and health disparities in minority populations, which are prone to higher rates of flu as well as COVID-19.
Flu vaccine ‘extra important’
The convergence of COVID-19 and influenza has the potential to overwhelm the health system, said Daniel A. Solomon, MD, of Brigham and Women’s in Boston. He coauthored a JAMA Insights clinical update on flu season during the COVID-19 pandemic that lists distinguishing and overlapping signs and symptoms of the two diseases.
The flu vaccine, he said, is “extra important this year,” especially in patients with existing respiratory disease, but COVID-19 has thrown up barriers to vaccination. Telemedicine has supplanted office visits. “People may miss that easy-touch opportunity to get the flu vaccine, so we have to be creative about making the flu vaccine highly accessible, maybe in nontraditional ways,” Dr. Solomon said. Some ideas he offered are pop-up vaccine fairs at schools and churches.
But just as COVID-19 may hinder flu vaccines, it may also be helping to mitigate flu transmission. “The interesting thing about transmission of the flu is that it’s transmitted the same way COVID is, so if we actually know how to decrease transmission of COVID, which we do – we’ve done it – we can actually decrease transmission of influenza as well,” Dr. Solomon said. Studies out of Hong Kong and Japan have reported a reduction in influenza cases during COVID-19 outbreaks in those places (Lancet Public Health. 2020;5:e279-88; JAMA. 2020;323:1969-71).
Risks of coinfection
About one in four COVID-19 patients have been diagnosed with an additional respiratory infection, including influenza (JAMA. 2020:323:2085-6). Pulmonologists must keep that in mind when managing COVID-19 suspects, said Dr. Singer.
“While it is true that most of the time COVID-19 travels alone, we have numerous examples in the literature and in our own experience that COVID-19 is accompanied by either another virus or another bacterial infection, including influenza,” Dr. Singer said. “The distinction is important. One is just for diagnostic reasons and public reporting reasons, but also because flu and COVID-19 have different requirements for how you care for patients in terms of the health system.”
Clinical suspicion for coinfection should remain high if the community spread of both COVID-19 and influenza is high, said Megan Conroy, MD, chief pulmonary and critical care fellow at Ohio State University, Columbus. “As the coronavirus first took hold in the United States in March 2020, we were at the tail end of influenza season, so it’s hard to predict what the upcoming influenza season will really look like with regards to coinfection.”
Distinguishing COVID-19 from flu
Multiple signs and symptoms between COVID-19 and the flu overlap. They include fever, chills, headache, myalgia, cough, and fatigue. Nasal congestion and sore throat are characteristic of the flu; shortness of breath and loss of the sense of smell have been widely reported in COVID-19. “While many upper respiratory infections can result in loss of smell, this may be more prevalent in COVID-19,” Dr. Conroy said. Other symptoms unique to COVID-19 are GI symptoms such as diarrhea and skin rashes such as acral ischemia.
Testing, however, is the cornerstone of the differential diagnosis. “You can’t confidently distinguish between them on symptoms alone,” Dr. Conroy added.
“I think the challenge we’ll face as clinicians, is caring for people with nonspecific symptoms of a respiratory viral illness, especially in the early phase of the illness,” said Dr. Solomon.
But even after that, symptoms can be difficult to distinguish.
“Later in the illness, COVID is more associated with a hypercoagulable state,” he said. “It is more associated with viral pneumonia on chest imaging, like the diffuse ground-glass infiltrates that we’ve all gotten used to seeing – but flu can do both of those things as well. So, without a test, it’s impossible to distinguish between the two infections in the clinic.”
But testing can have its shortcomings when flu season clashes with the COVID-19 pandemic. “Getting the test is not the same as getting the test results,” Dr. Solomon added. “Though a lot of people can get a test, if it takes 7 or 8 days to get the test result back, the result is useless.”
Widespread, rapid testing also depends on having adequate supplies of viral media transport and swabs. “I think that this is what we should be focusing on now: scaling up access to rapid turnaround testing,” he said. Distinguishing between the two is also important to preserve hospital resources. COVID-19 has more rigorous standards than flu for personal protective equipment and isolation of patients within the hospital.
Having chronic lung disease isn’t necessarily a risk factor for contracting COVID-19 or the flu, or both, Dr. Solomon said. “It’s a risk factor for having severe disease.” Again, he noted that flu vaccines are still necessary in these patients, as well as patients of advanced age and underlying medical conditions such as heart disease, diabetes, and obesity.
In managing children, it’s important to keep in mind that they communicate differently about their illnesses than adults, said Dr. Kaplan. “They may not have the words to tell you the same kind of thing that the adult tells you.” That’s where family members can help to flesh out the history. “They may present with an initially much milder form, if you will, where they’re not as critical up front, but then that small proportion of them comes back with the multi-inflammatory syndrome and then they are profoundly ill.”
Younger people make up a larger share of COVID-19 patients now, compared with the initial wave that hit the Northeast in the spring, Dr. Kaplan said. “We don’t know if that’s because the virus is a little different or the people that are getting sick are a little bit different.”
The COVID-19 strain now emerging may be less virulent than the strain that hit in early spring, he said. “That doesn’t mean that there aren’t still profoundly critical ill people with COVID of many different age ranges, that is true, but there are a lot of people that we now see will test positive, but aren’t really as profoundly ill as when it first landed here in the United States.”
That may be somewhat welcome as flu season arrives.
The physicians interviewed have no relevant disclosures.
The medical community is about to find out how prepared it is for the double whammy of influenza and COVID-19 that has been predicted for the fall of 2020. The complexities of diagnosis, management of vulnerable patients, and overflowing medical centers that have made the COVID-19 crisis so brutal may all be exacerbated by the arrival of seasonal influenza.
Lewis Jay Kaplan, MD, FCCP, a critical care surgeon at the University of Pennsylvania, Philadelphia, has seen his share of critically ill COVID-19 patients in the surgical ICU that he oversees. He’s approaching the upcoming flu season, poised to collide with the ongoing COVID-19 pandemic, ready to listen to each patient’s story to distinguish one from the other and determine treatment.
“The patients that have underlying comorbidities all have a story, and it’s up to you to figure out which chapter you’re in and how far along you happen to be,” he said. “It’s a very interesting approach to care, medical storytelling.”
With flu season closing in, pulmonologists are ruminating about how they’ll distinguish symptoms of COVID-19 and traditional influenza and how they’ll manage the most vulnerable patients, namely those with underlying respiratory disease and children. Influenza kills 12,000-61,000 people a year, according to the Centers for Disease Control, and results in 140,000-810,00 hospitalizations. Having a flu season in the midst of a pandemic of a disease with multiple overlapping symptoms threatens to overwhelm practitioners, hospitals, and the health system.
Dr. Kaplan said each patient’s story can point to the correct clinical approach. “Instead of just sharing data when you are on rounds, you’re really telling someone’s story.” It arises from a series of questions about how the disease has impacted them, specifics of their presentation, how their signs and symptoms differ from the usual, and how they responded to treatment. “It also helps you to then take what you’re doing, which can seem very, very complicated to individuals who are not medically sophisticated, and then help them to understand why you’re doing what you’re doing at this point.”
That can help get through to a patient with respiratory disease who insists he or she has or doesn’t have COVID-19 rather than the flu. “They form a different group that brings with them different fears and concerns, and you have to help them navigate that, too: all of this data and your decision-making around testing and admissions, and what you can omit doing and what you must do help them to navigate their own story,” Dr. Kaplan said.
Benjamin D. Singer, MD, a pulmonologist at Northwestern University, Chicago, authored an editorial in Science Advances that addressed four factors that will determine the scope of flu spread in the upcoming season: rate of transmission; vaccination rates; coinfection rates; and health disparities in minority populations, which are prone to higher rates of flu as well as COVID-19.
Flu vaccine ‘extra important’
The convergence of COVID-19 and influenza has the potential to overwhelm the health system, said Daniel A. Solomon, MD, of Brigham and Women’s in Boston. He coauthored a JAMA Insights clinical update on flu season during the COVID-19 pandemic that lists distinguishing and overlapping signs and symptoms of the two diseases.
The flu vaccine, he said, is “extra important this year,” especially in patients with existing respiratory disease, but COVID-19 has thrown up barriers to vaccination. Telemedicine has supplanted office visits. “People may miss that easy-touch opportunity to get the flu vaccine, so we have to be creative about making the flu vaccine highly accessible, maybe in nontraditional ways,” Dr. Solomon said. Some ideas he offered are pop-up vaccine fairs at schools and churches.
But just as COVID-19 may hinder flu vaccines, it may also be helping to mitigate flu transmission. “The interesting thing about transmission of the flu is that it’s transmitted the same way COVID is, so if we actually know how to decrease transmission of COVID, which we do – we’ve done it – we can actually decrease transmission of influenza as well,” Dr. Solomon said. Studies out of Hong Kong and Japan have reported a reduction in influenza cases during COVID-19 outbreaks in those places (Lancet Public Health. 2020;5:e279-88; JAMA. 2020;323:1969-71).
Risks of coinfection
About one in four COVID-19 patients have been diagnosed with an additional respiratory infection, including influenza (JAMA. 2020:323:2085-6). Pulmonologists must keep that in mind when managing COVID-19 suspects, said Dr. Singer.
“While it is true that most of the time COVID-19 travels alone, we have numerous examples in the literature and in our own experience that COVID-19 is accompanied by either another virus or another bacterial infection, including influenza,” Dr. Singer said. “The distinction is important. One is just for diagnostic reasons and public reporting reasons, but also because flu and COVID-19 have different requirements for how you care for patients in terms of the health system.”
Clinical suspicion for coinfection should remain high if the community spread of both COVID-19 and influenza is high, said Megan Conroy, MD, chief pulmonary and critical care fellow at Ohio State University, Columbus. “As the coronavirus first took hold in the United States in March 2020, we were at the tail end of influenza season, so it’s hard to predict what the upcoming influenza season will really look like with regards to coinfection.”
Distinguishing COVID-19 from flu
Multiple signs and symptoms between COVID-19 and the flu overlap. They include fever, chills, headache, myalgia, cough, and fatigue. Nasal congestion and sore throat are characteristic of the flu; shortness of breath and loss of the sense of smell have been widely reported in COVID-19. “While many upper respiratory infections can result in loss of smell, this may be more prevalent in COVID-19,” Dr. Conroy said. Other symptoms unique to COVID-19 are GI symptoms such as diarrhea and skin rashes such as acral ischemia.
Testing, however, is the cornerstone of the differential diagnosis. “You can’t confidently distinguish between them on symptoms alone,” Dr. Conroy added.
“I think the challenge we’ll face as clinicians, is caring for people with nonspecific symptoms of a respiratory viral illness, especially in the early phase of the illness,” said Dr. Solomon.
But even after that, symptoms can be difficult to distinguish.
“Later in the illness, COVID is more associated with a hypercoagulable state,” he said. “It is more associated with viral pneumonia on chest imaging, like the diffuse ground-glass infiltrates that we’ve all gotten used to seeing – but flu can do both of those things as well. So, without a test, it’s impossible to distinguish between the two infections in the clinic.”
But testing can have its shortcomings when flu season clashes with the COVID-19 pandemic. “Getting the test is not the same as getting the test results,” Dr. Solomon added. “Though a lot of people can get a test, if it takes 7 or 8 days to get the test result back, the result is useless.”
Widespread, rapid testing also depends on having adequate supplies of viral media transport and swabs. “I think that this is what we should be focusing on now: scaling up access to rapid turnaround testing,” he said. Distinguishing between the two is also important to preserve hospital resources. COVID-19 has more rigorous standards than flu for personal protective equipment and isolation of patients within the hospital.
Having chronic lung disease isn’t necessarily a risk factor for contracting COVID-19 or the flu, or both, Dr. Solomon said. “It’s a risk factor for having severe disease.” Again, he noted that flu vaccines are still necessary in these patients, as well as patients of advanced age and underlying medical conditions such as heart disease, diabetes, and obesity.
In managing children, it’s important to keep in mind that they communicate differently about their illnesses than adults, said Dr. Kaplan. “They may not have the words to tell you the same kind of thing that the adult tells you.” That’s where family members can help to flesh out the history. “They may present with an initially much milder form, if you will, where they’re not as critical up front, but then that small proportion of them comes back with the multi-inflammatory syndrome and then they are profoundly ill.”
Younger people make up a larger share of COVID-19 patients now, compared with the initial wave that hit the Northeast in the spring, Dr. Kaplan said. “We don’t know if that’s because the virus is a little different or the people that are getting sick are a little bit different.”
The COVID-19 strain now emerging may be less virulent than the strain that hit in early spring, he said. “That doesn’t mean that there aren’t still profoundly critical ill people with COVID of many different age ranges, that is true, but there are a lot of people that we now see will test positive, but aren’t really as profoundly ill as when it first landed here in the United States.”
That may be somewhat welcome as flu season arrives.
The physicians interviewed have no relevant disclosures.