User login
Long COVID could spell kidney troubles down the line
Physicians caring for COVID-19 survivors should routinely check kidney function, which is often damaged by the SARS-CoV-2 virus months after both severe and milder cases, new research indicates.
The largest study to date with the longest follow-up of COVID-19-related kidney outcomes also found that every type of kidney problem, including end-stage kidney disease (ESKD), was far more common in COVID-19 survivors who were admitted to the ICU or experienced acute kidney injury (AKI) while hospitalized.
Researchers analyzed U.S. Veterans Health Administration data from more than 1.7 million patients, including more than 89,000 who tested positive for COVID-19, for the study, which was published online Sept. 1, 2021, in the Journal of the American Society of Nephrology.
The risk of kidney problems “is more robust or pronounced in people who have had severe infection, but present in even asymptomatic and mild disease, which shouldn’t be discounted. Those people represent the majority of those with COVID-19,” said senior author Ziyad Al-Aly, MD, of the Veteran Affairs St. Louis Health Care System.
“That’s why the results are important, because even in people with mild disease to start with, the risk of kidney problems is not trivial,” he told this news organization. “It’s smaller than in people who were in the ICU, but it’s not ... zero.”
Experts aren’t yet certain how COVID-19 can damage the kidneys, hypothesizing that several factors may be at play. The virus may directly infect kidney cells rich in ACE2 receptors, which are key to infection, said nephrologist F. Perry Wilson, MD, of Yale University, New Haven, Conn., and a member of Medscape’s advisory board.
Kidneys might also be particularly vulnerable to the inflammatory cascade or blood clotting often seen in COVID-19, Dr. Al-Aly and Wilson both suggested.
COVID-19 survivors more likely to have kidney damage than controls
“A lot of health systems either have or are establishing post-COVID care clinics, which we think should definitely incorporate a kidney component,” Dr. Al-Aly advised. “They should check patients’ blood and urine for kidney problems.”
This is particularly important because “kidney problems, for the most part, are painless and silent,” he added.
“Realizing 2 years down the road that someone has ESKD, where they need dialysis or a kidney transplant, is what we don’t want. We don’t want this to be unrecognized, uncared for, unattended to,” he said.
Dr. Al-Aly and colleagues evaluated VA health system records, including data from 89,216 patients who tested positive for COVID-19 between March 2020 and March 2021, as well as 1.7 million controls who did not have COVID-19. Over a median follow-up of about 5.5 months, participants’ estimated glomerular filtration rate and serum creatinine levels were tracked to assess kidney health and outcomes according to infection severity.
Results were striking, with COVID-19 survivors about one-third more likely than controls to have kidney damage or significant declines in kidney function between 1 and 6 months after infection. More than 4,700 COVID-19 survivors had lost at least 30% of their kidney function within a year, and these patients were 25% more likely to reach that level of decline than controls.
Additionally, COVID-19 survivors were nearly twice as likely to experience AKI and almost three times as likely to be diagnosed with ESKD as controls.
If your patient had COVID-19, ‘it’s reasonable to check kidney function’
“This information tells us that if your patient was sick with COVID-19 and comes for follow-up visits, it’s reasonable to check their kidney function,” Dr. Wilson, who was not involved with the research, told this news organization.
“Even for patients who were not hospitalized, if they were laid low or dehydrated ... it should be part of the post-COVID care package,” he said.
If just a fraction of the millions of COVID-19 survivors in the United States develop long-term kidney problems, the ripple effect on American health care could be substantial, Dr. Wilson and Dr. Al-Aly agreed.
“We’re still living in a pandemic, so it’s hard to tell the total impact,” Dr. Al-Aly said. “But this ultimately will contribute to a rise in burden of kidney disease. This and other long COVID manifestations are going to alter the landscape of clinical care and health care in the United States for a decade or more.”
Because renal problems can limit a patient’s treatment options for other major diseases, including diabetes and cancer, COVID-related kidney damage can ultimately impact survivability.
“There are a lot of medications you can’t use in people with advanced kidney problems,” Dr. Al-Aly said.
The main study limitation was that patients were mostly older White men (median age, 68 years), although more than 9,000 women were included in the VA data, Dr. Al-Aly noted. Additionally, controls were more likely to be younger, Black, living in long-term care, and have higher rates of chronic health conditions and medication use.
The experts agreed that ongoing research tracking kidney outcomes is crucial for years to come.
“We also need to be following a cohort of these patients as part of a research protocol where they come in every 6 months for a standard set of lab tests to really understand what’s going on with their kidneys,” Dr. Wilson said.
“Lastly – and a much tougher sell – is we need biopsies. It’s very hard to infer what’s going on in complex disease with the kidneys without biopsy tissue,” he added.
The study was funded by the American Society of Nephrology and the Department of Veterans Affairs. Dr. Al-Aly and Dr. Wilson reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians caring for COVID-19 survivors should routinely check kidney function, which is often damaged by the SARS-CoV-2 virus months after both severe and milder cases, new research indicates.
The largest study to date with the longest follow-up of COVID-19-related kidney outcomes also found that every type of kidney problem, including end-stage kidney disease (ESKD), was far more common in COVID-19 survivors who were admitted to the ICU or experienced acute kidney injury (AKI) while hospitalized.
Researchers analyzed U.S. Veterans Health Administration data from more than 1.7 million patients, including more than 89,000 who tested positive for COVID-19, for the study, which was published online Sept. 1, 2021, in the Journal of the American Society of Nephrology.
The risk of kidney problems “is more robust or pronounced in people who have had severe infection, but present in even asymptomatic and mild disease, which shouldn’t be discounted. Those people represent the majority of those with COVID-19,” said senior author Ziyad Al-Aly, MD, of the Veteran Affairs St. Louis Health Care System.
“That’s why the results are important, because even in people with mild disease to start with, the risk of kidney problems is not trivial,” he told this news organization. “It’s smaller than in people who were in the ICU, but it’s not ... zero.”
Experts aren’t yet certain how COVID-19 can damage the kidneys, hypothesizing that several factors may be at play. The virus may directly infect kidney cells rich in ACE2 receptors, which are key to infection, said nephrologist F. Perry Wilson, MD, of Yale University, New Haven, Conn., and a member of Medscape’s advisory board.
Kidneys might also be particularly vulnerable to the inflammatory cascade or blood clotting often seen in COVID-19, Dr. Al-Aly and Wilson both suggested.
COVID-19 survivors more likely to have kidney damage than controls
“A lot of health systems either have or are establishing post-COVID care clinics, which we think should definitely incorporate a kidney component,” Dr. Al-Aly advised. “They should check patients’ blood and urine for kidney problems.”
This is particularly important because “kidney problems, for the most part, are painless and silent,” he added.
“Realizing 2 years down the road that someone has ESKD, where they need dialysis or a kidney transplant, is what we don’t want. We don’t want this to be unrecognized, uncared for, unattended to,” he said.
Dr. Al-Aly and colleagues evaluated VA health system records, including data from 89,216 patients who tested positive for COVID-19 between March 2020 and March 2021, as well as 1.7 million controls who did not have COVID-19. Over a median follow-up of about 5.5 months, participants’ estimated glomerular filtration rate and serum creatinine levels were tracked to assess kidney health and outcomes according to infection severity.
Results were striking, with COVID-19 survivors about one-third more likely than controls to have kidney damage or significant declines in kidney function between 1 and 6 months after infection. More than 4,700 COVID-19 survivors had lost at least 30% of their kidney function within a year, and these patients were 25% more likely to reach that level of decline than controls.
Additionally, COVID-19 survivors were nearly twice as likely to experience AKI and almost three times as likely to be diagnosed with ESKD as controls.
If your patient had COVID-19, ‘it’s reasonable to check kidney function’
“This information tells us that if your patient was sick with COVID-19 and comes for follow-up visits, it’s reasonable to check their kidney function,” Dr. Wilson, who was not involved with the research, told this news organization.
“Even for patients who were not hospitalized, if they were laid low or dehydrated ... it should be part of the post-COVID care package,” he said.
If just a fraction of the millions of COVID-19 survivors in the United States develop long-term kidney problems, the ripple effect on American health care could be substantial, Dr. Wilson and Dr. Al-Aly agreed.
“We’re still living in a pandemic, so it’s hard to tell the total impact,” Dr. Al-Aly said. “But this ultimately will contribute to a rise in burden of kidney disease. This and other long COVID manifestations are going to alter the landscape of clinical care and health care in the United States for a decade or more.”
Because renal problems can limit a patient’s treatment options for other major diseases, including diabetes and cancer, COVID-related kidney damage can ultimately impact survivability.
“There are a lot of medications you can’t use in people with advanced kidney problems,” Dr. Al-Aly said.
The main study limitation was that patients were mostly older White men (median age, 68 years), although more than 9,000 women were included in the VA data, Dr. Al-Aly noted. Additionally, controls were more likely to be younger, Black, living in long-term care, and have higher rates of chronic health conditions and medication use.
The experts agreed that ongoing research tracking kidney outcomes is crucial for years to come.
“We also need to be following a cohort of these patients as part of a research protocol where they come in every 6 months for a standard set of lab tests to really understand what’s going on with their kidneys,” Dr. Wilson said.
“Lastly – and a much tougher sell – is we need biopsies. It’s very hard to infer what’s going on in complex disease with the kidneys without biopsy tissue,” he added.
The study was funded by the American Society of Nephrology and the Department of Veterans Affairs. Dr. Al-Aly and Dr. Wilson reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians caring for COVID-19 survivors should routinely check kidney function, which is often damaged by the SARS-CoV-2 virus months after both severe and milder cases, new research indicates.
The largest study to date with the longest follow-up of COVID-19-related kidney outcomes also found that every type of kidney problem, including end-stage kidney disease (ESKD), was far more common in COVID-19 survivors who were admitted to the ICU or experienced acute kidney injury (AKI) while hospitalized.
Researchers analyzed U.S. Veterans Health Administration data from more than 1.7 million patients, including more than 89,000 who tested positive for COVID-19, for the study, which was published online Sept. 1, 2021, in the Journal of the American Society of Nephrology.
The risk of kidney problems “is more robust or pronounced in people who have had severe infection, but present in even asymptomatic and mild disease, which shouldn’t be discounted. Those people represent the majority of those with COVID-19,” said senior author Ziyad Al-Aly, MD, of the Veteran Affairs St. Louis Health Care System.
“That’s why the results are important, because even in people with mild disease to start with, the risk of kidney problems is not trivial,” he told this news organization. “It’s smaller than in people who were in the ICU, but it’s not ... zero.”
Experts aren’t yet certain how COVID-19 can damage the kidneys, hypothesizing that several factors may be at play. The virus may directly infect kidney cells rich in ACE2 receptors, which are key to infection, said nephrologist F. Perry Wilson, MD, of Yale University, New Haven, Conn., and a member of Medscape’s advisory board.
Kidneys might also be particularly vulnerable to the inflammatory cascade or blood clotting often seen in COVID-19, Dr. Al-Aly and Wilson both suggested.
COVID-19 survivors more likely to have kidney damage than controls
“A lot of health systems either have or are establishing post-COVID care clinics, which we think should definitely incorporate a kidney component,” Dr. Al-Aly advised. “They should check patients’ blood and urine for kidney problems.”
This is particularly important because “kidney problems, for the most part, are painless and silent,” he added.
“Realizing 2 years down the road that someone has ESKD, where they need dialysis or a kidney transplant, is what we don’t want. We don’t want this to be unrecognized, uncared for, unattended to,” he said.
Dr. Al-Aly and colleagues evaluated VA health system records, including data from 89,216 patients who tested positive for COVID-19 between March 2020 and March 2021, as well as 1.7 million controls who did not have COVID-19. Over a median follow-up of about 5.5 months, participants’ estimated glomerular filtration rate and serum creatinine levels were tracked to assess kidney health and outcomes according to infection severity.
Results were striking, with COVID-19 survivors about one-third more likely than controls to have kidney damage or significant declines in kidney function between 1 and 6 months after infection. More than 4,700 COVID-19 survivors had lost at least 30% of their kidney function within a year, and these patients were 25% more likely to reach that level of decline than controls.
Additionally, COVID-19 survivors were nearly twice as likely to experience AKI and almost three times as likely to be diagnosed with ESKD as controls.
If your patient had COVID-19, ‘it’s reasonable to check kidney function’
“This information tells us that if your patient was sick with COVID-19 and comes for follow-up visits, it’s reasonable to check their kidney function,” Dr. Wilson, who was not involved with the research, told this news organization.
“Even for patients who were not hospitalized, if they were laid low or dehydrated ... it should be part of the post-COVID care package,” he said.
If just a fraction of the millions of COVID-19 survivors in the United States develop long-term kidney problems, the ripple effect on American health care could be substantial, Dr. Wilson and Dr. Al-Aly agreed.
“We’re still living in a pandemic, so it’s hard to tell the total impact,” Dr. Al-Aly said. “But this ultimately will contribute to a rise in burden of kidney disease. This and other long COVID manifestations are going to alter the landscape of clinical care and health care in the United States for a decade or more.”
Because renal problems can limit a patient’s treatment options for other major diseases, including diabetes and cancer, COVID-related kidney damage can ultimately impact survivability.
“There are a lot of medications you can’t use in people with advanced kidney problems,” Dr. Al-Aly said.
The main study limitation was that patients were mostly older White men (median age, 68 years), although more than 9,000 women were included in the VA data, Dr. Al-Aly noted. Additionally, controls were more likely to be younger, Black, living in long-term care, and have higher rates of chronic health conditions and medication use.
The experts agreed that ongoing research tracking kidney outcomes is crucial for years to come.
“We also need to be following a cohort of these patients as part of a research protocol where they come in every 6 months for a standard set of lab tests to really understand what’s going on with their kidneys,” Dr. Wilson said.
“Lastly – and a much tougher sell – is we need biopsies. It’s very hard to infer what’s going on in complex disease with the kidneys without biopsy tissue,” he added.
The study was funded by the American Society of Nephrology and the Department of Veterans Affairs. Dr. Al-Aly and Dr. Wilson reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Florida doctor won’t treat unvaccinated patients in person
Linda Marraccini, MD, sent a letter to patients that said those who aren’t vaccinated won’t be allowed to book in-person appointments at her practice now that the Food and Drug Administration has fully approved the Pfizer vaccine for general use, according to NBC Miami.
“This is a public health emergency – the health of the public takes priority over the rights of any given individual in this situation,” she wrote in the letter, which was obtained by NBC Miami.
“It appears that there is a lack of selflessness and concern for the burden on the health and well-being of our society from our encounters,” she wrote.
Dr. Marraccini said the policy is in the best interest of her other patients and doesn’t violate the Hippocratic oath. Patients who are having chemotherapy or who have weak immune systems face higher risks of being infected with the coronavirus.
“It’s not fair for people who are unvaccinated to harm other people,” she told Newsweek Sept. 4.
“The Hippocratic oath is very science based,” she said. “I am following the science. I’m applying this to the benefit of the sick.”
Dr. Marraccini said her new policy goes into effect on Sept. 15, and she will continue to see unvaccinated patients virtually during the next month until they find another health care provider.
She said that the response to her decision has been “99.9% favorable” and that she will make exceptions if patients can’t receive a shot because of hardships. Her office also provides the Johnson & Johnson vaccine.
“We’re not going to leave them out there in the cold,” she told Newsweek.
During the past month, COVID-19 cases have surged in Florida, reaching record-level highs of more than 20,000 cases per day. Cases began rising in the summer because of the more contagious Delta variant.
In late August, a group of doctors in southern Florida urged people to get vaccinated, citing their exhaustion and frustration with unvaccinated patients who make up the large majority of COVID-19 hospitalizations, according to Newsweek.
Other doctors have declined to treat unvaccinated patients in recent weeks. Jason Valentine, MD, a doctor in Mobile, Ala., said he would no longer see unvaccinated patients as of Oct. 1, according to AL.com.
Dr. Marraccini urged people to become informed about COVID-19 vaccines, as well as their role in reducing the surge of cases in the state. The pandemic “did not have to go on this long,” she said.
“Responsibility has to do with each individual,” she told Newsweek. “This is a global health issue, and everyone owns part of that responsibility.”
A version of this article first appeared on WebMD.com.
Linda Marraccini, MD, sent a letter to patients that said those who aren’t vaccinated won’t be allowed to book in-person appointments at her practice now that the Food and Drug Administration has fully approved the Pfizer vaccine for general use, according to NBC Miami.
“This is a public health emergency – the health of the public takes priority over the rights of any given individual in this situation,” she wrote in the letter, which was obtained by NBC Miami.
“It appears that there is a lack of selflessness and concern for the burden on the health and well-being of our society from our encounters,” she wrote.
Dr. Marraccini said the policy is in the best interest of her other patients and doesn’t violate the Hippocratic oath. Patients who are having chemotherapy or who have weak immune systems face higher risks of being infected with the coronavirus.
“It’s not fair for people who are unvaccinated to harm other people,” she told Newsweek Sept. 4.
“The Hippocratic oath is very science based,” she said. “I am following the science. I’m applying this to the benefit of the sick.”
Dr. Marraccini said her new policy goes into effect on Sept. 15, and she will continue to see unvaccinated patients virtually during the next month until they find another health care provider.
She said that the response to her decision has been “99.9% favorable” and that she will make exceptions if patients can’t receive a shot because of hardships. Her office also provides the Johnson & Johnson vaccine.
“We’re not going to leave them out there in the cold,” she told Newsweek.
During the past month, COVID-19 cases have surged in Florida, reaching record-level highs of more than 20,000 cases per day. Cases began rising in the summer because of the more contagious Delta variant.
In late August, a group of doctors in southern Florida urged people to get vaccinated, citing their exhaustion and frustration with unvaccinated patients who make up the large majority of COVID-19 hospitalizations, according to Newsweek.
Other doctors have declined to treat unvaccinated patients in recent weeks. Jason Valentine, MD, a doctor in Mobile, Ala., said he would no longer see unvaccinated patients as of Oct. 1, according to AL.com.
Dr. Marraccini urged people to become informed about COVID-19 vaccines, as well as their role in reducing the surge of cases in the state. The pandemic “did not have to go on this long,” she said.
“Responsibility has to do with each individual,” she told Newsweek. “This is a global health issue, and everyone owns part of that responsibility.”
A version of this article first appeared on WebMD.com.
Linda Marraccini, MD, sent a letter to patients that said those who aren’t vaccinated won’t be allowed to book in-person appointments at her practice now that the Food and Drug Administration has fully approved the Pfizer vaccine for general use, according to NBC Miami.
“This is a public health emergency – the health of the public takes priority over the rights of any given individual in this situation,” she wrote in the letter, which was obtained by NBC Miami.
“It appears that there is a lack of selflessness and concern for the burden on the health and well-being of our society from our encounters,” she wrote.
Dr. Marraccini said the policy is in the best interest of her other patients and doesn’t violate the Hippocratic oath. Patients who are having chemotherapy or who have weak immune systems face higher risks of being infected with the coronavirus.
“It’s not fair for people who are unvaccinated to harm other people,” she told Newsweek Sept. 4.
“The Hippocratic oath is very science based,” she said. “I am following the science. I’m applying this to the benefit of the sick.”
Dr. Marraccini said her new policy goes into effect on Sept. 15, and she will continue to see unvaccinated patients virtually during the next month until they find another health care provider.
She said that the response to her decision has been “99.9% favorable” and that she will make exceptions if patients can’t receive a shot because of hardships. Her office also provides the Johnson & Johnson vaccine.
“We’re not going to leave them out there in the cold,” she told Newsweek.
During the past month, COVID-19 cases have surged in Florida, reaching record-level highs of more than 20,000 cases per day. Cases began rising in the summer because of the more contagious Delta variant.
In late August, a group of doctors in southern Florida urged people to get vaccinated, citing their exhaustion and frustration with unvaccinated patients who make up the large majority of COVID-19 hospitalizations, according to Newsweek.
Other doctors have declined to treat unvaccinated patients in recent weeks. Jason Valentine, MD, a doctor in Mobile, Ala., said he would no longer see unvaccinated patients as of Oct. 1, according to AL.com.
Dr. Marraccini urged people to become informed about COVID-19 vaccines, as well as their role in reducing the surge of cases in the state. The pandemic “did not have to go on this long,” she said.
“Responsibility has to do with each individual,” she told Newsweek. “This is a global health issue, and everyone owns part of that responsibility.”
A version of this article first appeared on WebMD.com.
United States reaches 5 million cases of child COVID
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
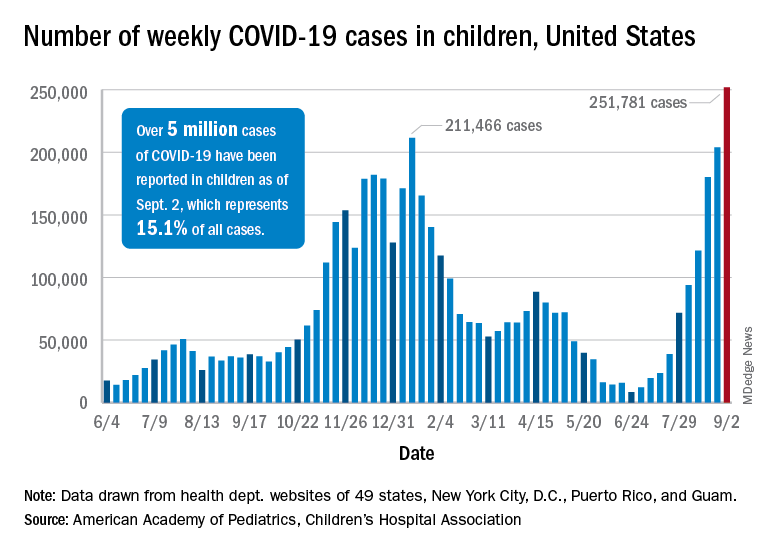
The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Large study affirms what we already know: Masks work to prevent COVID-19
It also shows that surgical masks are more effective than cloth face coverings.
The study, which was published ahead of peer review, demonstrates the power of careful investigation and offers a host of lessons about mask wearing that will be important worldwide. One key finding of the study, for example, is that wearing a mask doesn’t lead people to abandon social distancing, something public health officials had feared might happen if masks gave people a false sense of security.
“What we really were able to achieve is to demonstrate that masks are effective against COVID-19, even under a rigorous and systematic evaluation that was done in the throes of the pandemic,” said Ashley Styczynski, MD, who was an infectious disease fellow at Stanford (Calif.) University when she collaborated on the study with other colleagues at Stanford, Yale, and Innovations for Poverty Action, a large research and policy nonprofit organization that currently works in 22 countries.
“And so, I think people who have been holding out on wearing masks because [they] felt like there wasn’t enough evidence for it, we’re hoping this will really help bridge that gap for them,” she said.
It included more than 600 unions – or local governmental districts in Bangladesh – and roughly 340,000 people.
Half of the districts were given cloth or surgical face masks along with continual reminders to wear them properly; the other half were tracked with no intervention. Blood tests of people who developed symptoms during the study verified their infections.
Compared to villages that didn’t mask, those in which masks of any type were worn had about 9% fewer symptomatic cases of COVID-19. The finding was statistically significant and was unlikely to have occurred by chance alone.
“Somebody could read this study and say, ‘OK, you reduced COVID-19 by 9%. Big deal.’ And what I would respond to that would be that, if anything, we think that that is a substantial underestimate,” Dr. Styczynski said.
One reason they think they underestimated the effectiveness of masks is that they tested only people who were having symptoms, so people who had only very mild or asymptomatic infections were missed.
Another reason is that, among people who had symptoms, only one-third agreed to undergo a blood test. The effect may have been bigger had participation been universal.
Local transmission may have played a role, too. Rates of COVID-19 in Bangladesh were relatively low during the study. Most infections were caused by the B.1.1.7, or Alpha, variant.
Since then, Delta has taken over. Delta is thought to be more transmissible, and some studies have suggested that people infected with Delta shed more viral particles. Masks may be more effective when more virus is circulating.
The investigators also found important differences by age and by the type of mask. Villages in which surgical masks were worn had 11% fewer COVID-19 cases than villages in which masks were not worn. In villages in which cloth masks were worn, on the other hand, infections were reduced by only 5%.
The cloth masks were substantial. Each had three layers – two layers of fabric with an outer layer of polypropylene. On testing, the filtration efficiency of the cloth masks was only about 37%, compared with 95% for the three-layer surgical masks, which were also made of polypropylene.
Masks were most effective for older individuals. People aged 50-60 years who wore surgical masks were 23% less likely to test positive for COVID, compared with their peers who didn’t wear masks. For people older than 60, the reduction in risk was greater – 35%.
Rigorous research
The study took place over a period of 8 weeks in each district. The interventions were rolled out in waves, with the first starting in November 2020 and the last in January 2021.
Investigators gave each household free cloth or surgical face masks and showed families a video about proper mask wearing with promotional messages from the prime minister, a head imam, and a national cricket star. They also handed out free masks.
Previous studies have shown that people aren’t always truthful about wearing masks in public. In Kenya, for example, 88% of people answering a phone survey said that they wore masks regularly, but researchers determined that only 10% of them actually did so.
Investigators in the Bangladesh study didn’t just ask people if they’d worn masks, they stationed themselves in public markets, mosques, tea stalls, and on roads that were the main entrances to the villages and took notes.
They also tested various ways to educate people and to remind them to wear masks. They found that four factors were effective at promoting the wearing of masks, and they gave them an acronym – NORM.
- N for no-cost masks.
- O for offering information through the video and local leaders.
- R for regular reminders to people by investigators who stand in public markets and offer masks or encourage anyone who wasn’t wearing one or wasn’t wearing it correctly.
- M for modeling, in which local leaders, such as imams, wear masks and remind their followers to wear them.
These four measures tripled the wearing of masks in the intervention communities, from a baseline level of 13% to 42%. People continued to wear their masks properly for about 2 weeks after the study ended, indicating that they’d gotten used to wearing them.
Dr. Styczynski said that nothing else – not text message reminders, or signs posted in public places, or local incentives – moved the needle on mask wearing.
Saved lives and money
The study found that the strategy was cost effective, too. Giving masks to a large population and getting people to use them costs about $10,000 per life saved from COVID, on par with the cost of deploying mosquito nets to save people from malaria, Dr. Styczynski said.
“I think that what we’ve been able to show is that this is a really important tool to be used globally, especially as countries have delays in getting access to vaccines and rolling them out,” she said.
Dr. Styczynski said masks will continue to be important even in countries such as the United States, where vaccines aren’t stopping transmission 100% and there are still large portions of the population who are unvaccinated, such as children.
“If we want to reduce COVID-19 here, it’s really important that we consider the ongoing utility of masks, in addition to vaccines, and not really thinking of them as one or the other,” she said.
The study was funded by a grant from GiveWell.org. The funder had no role in the study design, interpretation, or the decision to publish.
A version of this article first appeared on Medscape.com.
It also shows that surgical masks are more effective than cloth face coverings.
The study, which was published ahead of peer review, demonstrates the power of careful investigation and offers a host of lessons about mask wearing that will be important worldwide. One key finding of the study, for example, is that wearing a mask doesn’t lead people to abandon social distancing, something public health officials had feared might happen if masks gave people a false sense of security.
“What we really were able to achieve is to demonstrate that masks are effective against COVID-19, even under a rigorous and systematic evaluation that was done in the throes of the pandemic,” said Ashley Styczynski, MD, who was an infectious disease fellow at Stanford (Calif.) University when she collaborated on the study with other colleagues at Stanford, Yale, and Innovations for Poverty Action, a large research and policy nonprofit organization that currently works in 22 countries.
“And so, I think people who have been holding out on wearing masks because [they] felt like there wasn’t enough evidence for it, we’re hoping this will really help bridge that gap for them,” she said.
It included more than 600 unions – or local governmental districts in Bangladesh – and roughly 340,000 people.
Half of the districts were given cloth or surgical face masks along with continual reminders to wear them properly; the other half were tracked with no intervention. Blood tests of people who developed symptoms during the study verified their infections.
Compared to villages that didn’t mask, those in which masks of any type were worn had about 9% fewer symptomatic cases of COVID-19. The finding was statistically significant and was unlikely to have occurred by chance alone.
“Somebody could read this study and say, ‘OK, you reduced COVID-19 by 9%. Big deal.’ And what I would respond to that would be that, if anything, we think that that is a substantial underestimate,” Dr. Styczynski said.
One reason they think they underestimated the effectiveness of masks is that they tested only people who were having symptoms, so people who had only very mild or asymptomatic infections were missed.
Another reason is that, among people who had symptoms, only one-third agreed to undergo a blood test. The effect may have been bigger had participation been universal.
Local transmission may have played a role, too. Rates of COVID-19 in Bangladesh were relatively low during the study. Most infections were caused by the B.1.1.7, or Alpha, variant.
Since then, Delta has taken over. Delta is thought to be more transmissible, and some studies have suggested that people infected with Delta shed more viral particles. Masks may be more effective when more virus is circulating.
The investigators also found important differences by age and by the type of mask. Villages in which surgical masks were worn had 11% fewer COVID-19 cases than villages in which masks were not worn. In villages in which cloth masks were worn, on the other hand, infections were reduced by only 5%.
The cloth masks were substantial. Each had three layers – two layers of fabric with an outer layer of polypropylene. On testing, the filtration efficiency of the cloth masks was only about 37%, compared with 95% for the three-layer surgical masks, which were also made of polypropylene.
Masks were most effective for older individuals. People aged 50-60 years who wore surgical masks were 23% less likely to test positive for COVID, compared with their peers who didn’t wear masks. For people older than 60, the reduction in risk was greater – 35%.
Rigorous research
The study took place over a period of 8 weeks in each district. The interventions were rolled out in waves, with the first starting in November 2020 and the last in January 2021.
Investigators gave each household free cloth or surgical face masks and showed families a video about proper mask wearing with promotional messages from the prime minister, a head imam, and a national cricket star. They also handed out free masks.
Previous studies have shown that people aren’t always truthful about wearing masks in public. In Kenya, for example, 88% of people answering a phone survey said that they wore masks regularly, but researchers determined that only 10% of them actually did so.
Investigators in the Bangladesh study didn’t just ask people if they’d worn masks, they stationed themselves in public markets, mosques, tea stalls, and on roads that were the main entrances to the villages and took notes.
They also tested various ways to educate people and to remind them to wear masks. They found that four factors were effective at promoting the wearing of masks, and they gave them an acronym – NORM.
- N for no-cost masks.
- O for offering information through the video and local leaders.
- R for regular reminders to people by investigators who stand in public markets and offer masks or encourage anyone who wasn’t wearing one or wasn’t wearing it correctly.
- M for modeling, in which local leaders, such as imams, wear masks and remind their followers to wear them.
These four measures tripled the wearing of masks in the intervention communities, from a baseline level of 13% to 42%. People continued to wear their masks properly for about 2 weeks after the study ended, indicating that they’d gotten used to wearing them.
Dr. Styczynski said that nothing else – not text message reminders, or signs posted in public places, or local incentives – moved the needle on mask wearing.
Saved lives and money
The study found that the strategy was cost effective, too. Giving masks to a large population and getting people to use them costs about $10,000 per life saved from COVID, on par with the cost of deploying mosquito nets to save people from malaria, Dr. Styczynski said.
“I think that what we’ve been able to show is that this is a really important tool to be used globally, especially as countries have delays in getting access to vaccines and rolling them out,” she said.
Dr. Styczynski said masks will continue to be important even in countries such as the United States, where vaccines aren’t stopping transmission 100% and there are still large portions of the population who are unvaccinated, such as children.
“If we want to reduce COVID-19 here, it’s really important that we consider the ongoing utility of masks, in addition to vaccines, and not really thinking of them as one or the other,” she said.
The study was funded by a grant from GiveWell.org. The funder had no role in the study design, interpretation, or the decision to publish.
A version of this article first appeared on Medscape.com.
It also shows that surgical masks are more effective than cloth face coverings.
The study, which was published ahead of peer review, demonstrates the power of careful investigation and offers a host of lessons about mask wearing that will be important worldwide. One key finding of the study, for example, is that wearing a mask doesn’t lead people to abandon social distancing, something public health officials had feared might happen if masks gave people a false sense of security.
“What we really were able to achieve is to demonstrate that masks are effective against COVID-19, even under a rigorous and systematic evaluation that was done in the throes of the pandemic,” said Ashley Styczynski, MD, who was an infectious disease fellow at Stanford (Calif.) University when she collaborated on the study with other colleagues at Stanford, Yale, and Innovations for Poverty Action, a large research and policy nonprofit organization that currently works in 22 countries.
“And so, I think people who have been holding out on wearing masks because [they] felt like there wasn’t enough evidence for it, we’re hoping this will really help bridge that gap for them,” she said.
It included more than 600 unions – or local governmental districts in Bangladesh – and roughly 340,000 people.
Half of the districts were given cloth or surgical face masks along with continual reminders to wear them properly; the other half were tracked with no intervention. Blood tests of people who developed symptoms during the study verified their infections.
Compared to villages that didn’t mask, those in which masks of any type were worn had about 9% fewer symptomatic cases of COVID-19. The finding was statistically significant and was unlikely to have occurred by chance alone.
“Somebody could read this study and say, ‘OK, you reduced COVID-19 by 9%. Big deal.’ And what I would respond to that would be that, if anything, we think that that is a substantial underestimate,” Dr. Styczynski said.
One reason they think they underestimated the effectiveness of masks is that they tested only people who were having symptoms, so people who had only very mild or asymptomatic infections were missed.
Another reason is that, among people who had symptoms, only one-third agreed to undergo a blood test. The effect may have been bigger had participation been universal.
Local transmission may have played a role, too. Rates of COVID-19 in Bangladesh were relatively low during the study. Most infections were caused by the B.1.1.7, or Alpha, variant.
Since then, Delta has taken over. Delta is thought to be more transmissible, and some studies have suggested that people infected with Delta shed more viral particles. Masks may be more effective when more virus is circulating.
The investigators also found important differences by age and by the type of mask. Villages in which surgical masks were worn had 11% fewer COVID-19 cases than villages in which masks were not worn. In villages in which cloth masks were worn, on the other hand, infections were reduced by only 5%.
The cloth masks were substantial. Each had three layers – two layers of fabric with an outer layer of polypropylene. On testing, the filtration efficiency of the cloth masks was only about 37%, compared with 95% for the three-layer surgical masks, which were also made of polypropylene.
Masks were most effective for older individuals. People aged 50-60 years who wore surgical masks were 23% less likely to test positive for COVID, compared with their peers who didn’t wear masks. For people older than 60, the reduction in risk was greater – 35%.
Rigorous research
The study took place over a period of 8 weeks in each district. The interventions were rolled out in waves, with the first starting in November 2020 and the last in January 2021.
Investigators gave each household free cloth or surgical face masks and showed families a video about proper mask wearing with promotional messages from the prime minister, a head imam, and a national cricket star. They also handed out free masks.
Previous studies have shown that people aren’t always truthful about wearing masks in public. In Kenya, for example, 88% of people answering a phone survey said that they wore masks regularly, but researchers determined that only 10% of them actually did so.
Investigators in the Bangladesh study didn’t just ask people if they’d worn masks, they stationed themselves in public markets, mosques, tea stalls, and on roads that were the main entrances to the villages and took notes.
They also tested various ways to educate people and to remind them to wear masks. They found that four factors were effective at promoting the wearing of masks, and they gave them an acronym – NORM.
- N for no-cost masks.
- O for offering information through the video and local leaders.
- R for regular reminders to people by investigators who stand in public markets and offer masks or encourage anyone who wasn’t wearing one or wasn’t wearing it correctly.
- M for modeling, in which local leaders, such as imams, wear masks and remind their followers to wear them.
These four measures tripled the wearing of masks in the intervention communities, from a baseline level of 13% to 42%. People continued to wear their masks properly for about 2 weeks after the study ended, indicating that they’d gotten used to wearing them.
Dr. Styczynski said that nothing else – not text message reminders, or signs posted in public places, or local incentives – moved the needle on mask wearing.
Saved lives and money
The study found that the strategy was cost effective, too. Giving masks to a large population and getting people to use them costs about $10,000 per life saved from COVID, on par with the cost of deploying mosquito nets to save people from malaria, Dr. Styczynski said.
“I think that what we’ve been able to show is that this is a really important tool to be used globally, especially as countries have delays in getting access to vaccines and rolling them out,” she said.
Dr. Styczynski said masks will continue to be important even in countries such as the United States, where vaccines aren’t stopping transmission 100% and there are still large portions of the population who are unvaccinated, such as children.
“If we want to reduce COVID-19 here, it’s really important that we consider the ongoing utility of masks, in addition to vaccines, and not really thinking of them as one or the other,” she said.
The study was funded by a grant from GiveWell.org. The funder had no role in the study design, interpretation, or the decision to publish.
A version of this article first appeared on Medscape.com.
Beta-blocker reduces lung inflammation in critical COVID-19
In a small study, intravenous administration of the beta-blocker metoprolol to critically ill COVID-19 patients with acute respiratory distress syndrome (ARDS) safely blunted lung inflammation associated with the disease.
Metoprolol administration also resulted in better oxygenation and fewer days on intensive mechanical ventilation and in the ICU, compared with no treatment.
These data suggest that metoprolol repurposing for the treatment of ARDS in COVID-19 patients is a safe and inexpensive strategy with the potential to improve outcomes, the researchers said.
“Metoprolol repurposing for the treatment of ARDS associated with COVID-19 is a safe and cheap intervention that can help to alleviate the massive personal and health care burden associated with the pandemic,” they concluded.
The results, from the MADRID-COVID pilot trial from Agustin Clemente-Moragon, BSc, Centro National de Investigaciones Cardiovasculares, Madrid, and colleagues, were published online Aug. 30, 2021, in the Journal of the American College of Cardiology.
In previous work, the researchers showed that metoprolol, but not other clinically available intravenous beta-blockers, abrogates neutrophil-driven exacerbated inflammation, neutrophil-platelet interaction, and formation of neutrophil extracellular traps in a mouse model of acute lung injury.
These results prompted the current pilot trial in 20 patients, ages 18-80 years, with COVID-19–associated ARDS.
Randomization was stratified by age (59 and younger vs. 60 and older), history of hypertension (yes or no), and circulating neutrophil counts (<6,000 vs. ≥6,000). Bronchoalveolar lavage (BAL) fluid and blood samples were obtained from patients at randomization and 24 hours after the third metoprolol dose in the treatment group, and on day 4 in controls.
Because of the cardiovascular effects of metoprolol, patients were monitored invasively and by echocardiography, the authors noted.
As expected, metoprolol significantly reduced heart rate (P < .01) and systolic blood pressure (P < .05), although both remained within the physiological range. Echocardiography showed no deterioration of cardiac function after metoprolol treatment.
To assess the ability of metoprolol to address neutrophil-mediated exacerbated lung inflammation, the researchers analyzed leukocyte populations in BAL samples by flow cytometry at baseline and on day 4.
At baseline, the metoprolol and control groups showed no differences in BAL neutrophil content. But on day 4, after 3 days of treatment with metoprolol, neutrophil content was significantly lower in the metoprolol group (median, 14.3 neutrophils/mcL) than in the control group (median, 397 neutrophils/mcL).
Metoprolol-treated patients also had lower total inflammatory-cell content and lower monocyte/macrophage content. Lymphocytes did not differ between the groups.
The investigators also explored the impact of metoprolol on the chemokine, monocyte chemoattractant protein–1 (MCP-1), as it has been shown to promote pulmonary fibrosis in late-stage ARDS.
They found that MCP-1 was significantly attenuated after 3 days of metoprolol treatment. At baseline, the median MCP-1 level was 298 pg/mL; on day 4 after metoprolol, it was 203 pg/mL (P = .009).
MCP-1 levels remained unchanged in control patients.
An elegant study
In an accompanying editorial, Mourad H. Senussi, MD, assistant professor at Baylor College of Medicine, Houston, wrote: “Although the study has a small sample size, we commend the authors, who attempt to shed light on the important pathophysiological underpinnings that help establish biological plausibility for this inexpensive, safe, and widely available medication.”
In an interview with this news organization, Dr. Senussi added that metoprolol is not itself something primarily used to treat COVID-19 per se. “Rather, the drug blunts the sympathetic-host response. There is a fine balance between that sympathetic surge that is helpful to the body, and then a sympathetic surge that if left unchecked, can lead to significant damage. And so, I think this study really shows that medications like metoprolol can help blunt that initial sympathetic effect.”
A larger study is “absolutely” warranted, he added, “this is a drug that is readily available, safe, and inexpensive. The study design here was simple and most importantly, showed biological plausibility.”
Dr. Senussi also noted that, although the benefit was noted in COVID-19 patients, the study sets the groundwork for further research in the use of beta-blockade in the critically ill. “Further studies are needed to elucidate and identify where along the inflammatory spectrum these critically ill patients lie, which patients would benefit from beta-blockers, and at what time point during their hospital stay.”
The MADRID-COVID authors and Dr. Senussi disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a small study, intravenous administration of the beta-blocker metoprolol to critically ill COVID-19 patients with acute respiratory distress syndrome (ARDS) safely blunted lung inflammation associated with the disease.
Metoprolol administration also resulted in better oxygenation and fewer days on intensive mechanical ventilation and in the ICU, compared with no treatment.
These data suggest that metoprolol repurposing for the treatment of ARDS in COVID-19 patients is a safe and inexpensive strategy with the potential to improve outcomes, the researchers said.
“Metoprolol repurposing for the treatment of ARDS associated with COVID-19 is a safe and cheap intervention that can help to alleviate the massive personal and health care burden associated with the pandemic,” they concluded.
The results, from the MADRID-COVID pilot trial from Agustin Clemente-Moragon, BSc, Centro National de Investigaciones Cardiovasculares, Madrid, and colleagues, were published online Aug. 30, 2021, in the Journal of the American College of Cardiology.
In previous work, the researchers showed that metoprolol, but not other clinically available intravenous beta-blockers, abrogates neutrophil-driven exacerbated inflammation, neutrophil-platelet interaction, and formation of neutrophil extracellular traps in a mouse model of acute lung injury.
These results prompted the current pilot trial in 20 patients, ages 18-80 years, with COVID-19–associated ARDS.
Randomization was stratified by age (59 and younger vs. 60 and older), history of hypertension (yes or no), and circulating neutrophil counts (<6,000 vs. ≥6,000). Bronchoalveolar lavage (BAL) fluid and blood samples were obtained from patients at randomization and 24 hours after the third metoprolol dose in the treatment group, and on day 4 in controls.
Because of the cardiovascular effects of metoprolol, patients were monitored invasively and by echocardiography, the authors noted.
As expected, metoprolol significantly reduced heart rate (P < .01) and systolic blood pressure (P < .05), although both remained within the physiological range. Echocardiography showed no deterioration of cardiac function after metoprolol treatment.
To assess the ability of metoprolol to address neutrophil-mediated exacerbated lung inflammation, the researchers analyzed leukocyte populations in BAL samples by flow cytometry at baseline and on day 4.
At baseline, the metoprolol and control groups showed no differences in BAL neutrophil content. But on day 4, after 3 days of treatment with metoprolol, neutrophil content was significantly lower in the metoprolol group (median, 14.3 neutrophils/mcL) than in the control group (median, 397 neutrophils/mcL).
Metoprolol-treated patients also had lower total inflammatory-cell content and lower monocyte/macrophage content. Lymphocytes did not differ between the groups.
The investigators also explored the impact of metoprolol on the chemokine, monocyte chemoattractant protein–1 (MCP-1), as it has been shown to promote pulmonary fibrosis in late-stage ARDS.
They found that MCP-1 was significantly attenuated after 3 days of metoprolol treatment. At baseline, the median MCP-1 level was 298 pg/mL; on day 4 after metoprolol, it was 203 pg/mL (P = .009).
MCP-1 levels remained unchanged in control patients.
An elegant study
In an accompanying editorial, Mourad H. Senussi, MD, assistant professor at Baylor College of Medicine, Houston, wrote: “Although the study has a small sample size, we commend the authors, who attempt to shed light on the important pathophysiological underpinnings that help establish biological plausibility for this inexpensive, safe, and widely available medication.”
In an interview with this news organization, Dr. Senussi added that metoprolol is not itself something primarily used to treat COVID-19 per se. “Rather, the drug blunts the sympathetic-host response. There is a fine balance between that sympathetic surge that is helpful to the body, and then a sympathetic surge that if left unchecked, can lead to significant damage. And so, I think this study really shows that medications like metoprolol can help blunt that initial sympathetic effect.”
A larger study is “absolutely” warranted, he added, “this is a drug that is readily available, safe, and inexpensive. The study design here was simple and most importantly, showed biological plausibility.”
Dr. Senussi also noted that, although the benefit was noted in COVID-19 patients, the study sets the groundwork for further research in the use of beta-blockade in the critically ill. “Further studies are needed to elucidate and identify where along the inflammatory spectrum these critically ill patients lie, which patients would benefit from beta-blockers, and at what time point during their hospital stay.”
The MADRID-COVID authors and Dr. Senussi disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a small study, intravenous administration of the beta-blocker metoprolol to critically ill COVID-19 patients with acute respiratory distress syndrome (ARDS) safely blunted lung inflammation associated with the disease.
Metoprolol administration also resulted in better oxygenation and fewer days on intensive mechanical ventilation and in the ICU, compared with no treatment.
These data suggest that metoprolol repurposing for the treatment of ARDS in COVID-19 patients is a safe and inexpensive strategy with the potential to improve outcomes, the researchers said.
“Metoprolol repurposing for the treatment of ARDS associated with COVID-19 is a safe and cheap intervention that can help to alleviate the massive personal and health care burden associated with the pandemic,” they concluded.
The results, from the MADRID-COVID pilot trial from Agustin Clemente-Moragon, BSc, Centro National de Investigaciones Cardiovasculares, Madrid, and colleagues, were published online Aug. 30, 2021, in the Journal of the American College of Cardiology.
In previous work, the researchers showed that metoprolol, but not other clinically available intravenous beta-blockers, abrogates neutrophil-driven exacerbated inflammation, neutrophil-platelet interaction, and formation of neutrophil extracellular traps in a mouse model of acute lung injury.
These results prompted the current pilot trial in 20 patients, ages 18-80 years, with COVID-19–associated ARDS.
Randomization was stratified by age (59 and younger vs. 60 and older), history of hypertension (yes or no), and circulating neutrophil counts (<6,000 vs. ≥6,000). Bronchoalveolar lavage (BAL) fluid and blood samples were obtained from patients at randomization and 24 hours after the third metoprolol dose in the treatment group, and on day 4 in controls.
Because of the cardiovascular effects of metoprolol, patients were monitored invasively and by echocardiography, the authors noted.
As expected, metoprolol significantly reduced heart rate (P < .01) and systolic blood pressure (P < .05), although both remained within the physiological range. Echocardiography showed no deterioration of cardiac function after metoprolol treatment.
To assess the ability of metoprolol to address neutrophil-mediated exacerbated lung inflammation, the researchers analyzed leukocyte populations in BAL samples by flow cytometry at baseline and on day 4.
At baseline, the metoprolol and control groups showed no differences in BAL neutrophil content. But on day 4, after 3 days of treatment with metoprolol, neutrophil content was significantly lower in the metoprolol group (median, 14.3 neutrophils/mcL) than in the control group (median, 397 neutrophils/mcL).
Metoprolol-treated patients also had lower total inflammatory-cell content and lower monocyte/macrophage content. Lymphocytes did not differ between the groups.
The investigators also explored the impact of metoprolol on the chemokine, monocyte chemoattractant protein–1 (MCP-1), as it has been shown to promote pulmonary fibrosis in late-stage ARDS.
They found that MCP-1 was significantly attenuated after 3 days of metoprolol treatment. At baseline, the median MCP-1 level was 298 pg/mL; on day 4 after metoprolol, it was 203 pg/mL (P = .009).
MCP-1 levels remained unchanged in control patients.
An elegant study
In an accompanying editorial, Mourad H. Senussi, MD, assistant professor at Baylor College of Medicine, Houston, wrote: “Although the study has a small sample size, we commend the authors, who attempt to shed light on the important pathophysiological underpinnings that help establish biological plausibility for this inexpensive, safe, and widely available medication.”
In an interview with this news organization, Dr. Senussi added that metoprolol is not itself something primarily used to treat COVID-19 per se. “Rather, the drug blunts the sympathetic-host response. There is a fine balance between that sympathetic surge that is helpful to the body, and then a sympathetic surge that if left unchecked, can lead to significant damage. And so, I think this study really shows that medications like metoprolol can help blunt that initial sympathetic effect.”
A larger study is “absolutely” warranted, he added, “this is a drug that is readily available, safe, and inexpensive. The study design here was simple and most importantly, showed biological plausibility.”
Dr. Senussi also noted that, although the benefit was noted in COVID-19 patients, the study sets the groundwork for further research in the use of beta-blockade in the critically ill. “Further studies are needed to elucidate and identify where along the inflammatory spectrum these critically ill patients lie, which patients would benefit from beta-blockers, and at what time point during their hospital stay.”
The MADRID-COVID authors and Dr. Senussi disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NCCN recommends third COVID-19 dose for patients with cancer
Experts at the National Comprehensive Cancer Network have now issued an updated recommendation for COVID-19 vaccination in people with cancer. The panel calls for these patients to be among the highest-priority group to be vaccinated against COVID-19 and to receive the newly approved third dose of vaccine.
The NCCN has recommended in February that all patients receiving active cancer treatment should receive a COVID-19 vaccine and should be prioritized for vaccination. In August, the FDA authorized a third dose of either the Pfizer or Moderna COVID-19 vaccines for people with compromised immune systems. Those eligible for a third dose include solid organ transplant recipients, those undergoing cancer treatments, and people with autoimmune diseases that suppress their immune systems
The new NCCN recommendations state that the following groups should be considered eligible for a third dose of the mRNA COVID-19 vaccine immediately, based on the latest decisions from the Food and Drug Administration and the Centers for Disease Control and Prevention:
- Patients with solid tumors (either new or recurring) receiving treatment within 1 year of their initial vaccine dose, regardless of their type of cancer therapy.
- Patients with active hematologic malignancies regardless of whether they are currently receiving cancer therapy.
- Anyone who received a stem cell transplant (SCT) or engineered cellular therapy (for example, chimeric antigen receptor T cells), especially within the past 2 years.
- Any recipients of allogeneic SCT on immunosuppressive therapy or with a history of graft-versus-host disease regardless of the time of transplant.
- Anyone with an additional immunosuppressive condition (for example, HIV) or being treated with immunosuppressive agents unrelated to their cancer therapy.
Cancer patients at high risk of complications
As previously reported by this news organization, infection with COVID-19 in people with cancer can severely impact survival. One study published in 2020 found that patients with both COVID-19 infection and progressing cancer had a fivefold increase in the risk of 30-day mortality, compared with COVID-19–positive cancer patients who were in remission or had no evidence of cancer.
Another study found that cancer type, stage, and recent treatment could affect outcomes of COVID-19 in patients with cancer. Patients with hematologic malignancies and metastatic cancers had higher risks of developing severe or critical COVID-19 symptoms, being admitted to the ICU, requiring ventilation, and dying. Conversely, those with nonmetastatic disease had outcomes that were comparable with persons without cancer and a COVID-19 infection. This study also found that having undergone recent surgery or receiving immunotherapy also put patients at a higher risk of poor outcomes, although patients with cancer who were treated with radiotherapy had outcomes similar to those of noncancer COVID-19 patients.
“COVID-19 can be very dangerous, especially for people living with cancer, which is why we’re so grateful for safe and effective vaccines that are saving lives,” Robert W. Carlson, MD, CEO of NCCN, said in a statement.
Right timing and location
The current NCCN update also recommends that individuals wait at least 4 weeks between the second and third doses, and those who are infected with COVID-19 after being vaccinated should wait until they have documented clearance of the virus before receiving a third dose.
It also recommends that people who live in the same household with immunocompromised individuals should also get a third dose once it becomes available, and that it is best to have a third dose of the same type of vaccine as the first two doses. However, a different mRNA vaccine is also acceptable.
Immunocompromised individuals should try to receive their third dose in a health care delivery setting, as opposed to a pharmacy or public vaccination clinic if possible, as it would limit their risk of exposure to the general population.
Steve Pergam, MD, MPH, associate professor, vaccine and infectious disease division, Fred Hutchinson Cancer Research Center, Seattle, commented that it is still necessary to take precautions, even after getting the booster dose.
“That means, even after a third dose of vaccine, we still recommend immunocompromised people, such as those undergoing cancer treatment, continue to be cautious, wear masks, and avoid large group gatherings, particularly around those who are unvaccinated,” said Dr. Pergam, who is also coleader of the NCCN COVID-19 Vaccination Advisory Committee. “All of us should do our part to reduce the spread of COVID-19 and get vaccinated to protect those around us from preventable suffering.”
A version of this article first appeared on Medscape.com.
Experts at the National Comprehensive Cancer Network have now issued an updated recommendation for COVID-19 vaccination in people with cancer. The panel calls for these patients to be among the highest-priority group to be vaccinated against COVID-19 and to receive the newly approved third dose of vaccine.
The NCCN has recommended in February that all patients receiving active cancer treatment should receive a COVID-19 vaccine and should be prioritized for vaccination. In August, the FDA authorized a third dose of either the Pfizer or Moderna COVID-19 vaccines for people with compromised immune systems. Those eligible for a third dose include solid organ transplant recipients, those undergoing cancer treatments, and people with autoimmune diseases that suppress their immune systems
The new NCCN recommendations state that the following groups should be considered eligible for a third dose of the mRNA COVID-19 vaccine immediately, based on the latest decisions from the Food and Drug Administration and the Centers for Disease Control and Prevention:
- Patients with solid tumors (either new or recurring) receiving treatment within 1 year of their initial vaccine dose, regardless of their type of cancer therapy.
- Patients with active hematologic malignancies regardless of whether they are currently receiving cancer therapy.
- Anyone who received a stem cell transplant (SCT) or engineered cellular therapy (for example, chimeric antigen receptor T cells), especially within the past 2 years.
- Any recipients of allogeneic SCT on immunosuppressive therapy or with a history of graft-versus-host disease regardless of the time of transplant.
- Anyone with an additional immunosuppressive condition (for example, HIV) or being treated with immunosuppressive agents unrelated to their cancer therapy.
Cancer patients at high risk of complications
As previously reported by this news organization, infection with COVID-19 in people with cancer can severely impact survival. One study published in 2020 found that patients with both COVID-19 infection and progressing cancer had a fivefold increase in the risk of 30-day mortality, compared with COVID-19–positive cancer patients who were in remission or had no evidence of cancer.
Another study found that cancer type, stage, and recent treatment could affect outcomes of COVID-19 in patients with cancer. Patients with hematologic malignancies and metastatic cancers had higher risks of developing severe or critical COVID-19 symptoms, being admitted to the ICU, requiring ventilation, and dying. Conversely, those with nonmetastatic disease had outcomes that were comparable with persons without cancer and a COVID-19 infection. This study also found that having undergone recent surgery or receiving immunotherapy also put patients at a higher risk of poor outcomes, although patients with cancer who were treated with radiotherapy had outcomes similar to those of noncancer COVID-19 patients.
“COVID-19 can be very dangerous, especially for people living with cancer, which is why we’re so grateful for safe and effective vaccines that are saving lives,” Robert W. Carlson, MD, CEO of NCCN, said in a statement.
Right timing and location
The current NCCN update also recommends that individuals wait at least 4 weeks between the second and third doses, and those who are infected with COVID-19 after being vaccinated should wait until they have documented clearance of the virus before receiving a third dose.
It also recommends that people who live in the same household with immunocompromised individuals should also get a third dose once it becomes available, and that it is best to have a third dose of the same type of vaccine as the first two doses. However, a different mRNA vaccine is also acceptable.
Immunocompromised individuals should try to receive their third dose in a health care delivery setting, as opposed to a pharmacy or public vaccination clinic if possible, as it would limit their risk of exposure to the general population.
Steve Pergam, MD, MPH, associate professor, vaccine and infectious disease division, Fred Hutchinson Cancer Research Center, Seattle, commented that it is still necessary to take precautions, even after getting the booster dose.
“That means, even after a third dose of vaccine, we still recommend immunocompromised people, such as those undergoing cancer treatment, continue to be cautious, wear masks, and avoid large group gatherings, particularly around those who are unvaccinated,” said Dr. Pergam, who is also coleader of the NCCN COVID-19 Vaccination Advisory Committee. “All of us should do our part to reduce the spread of COVID-19 and get vaccinated to protect those around us from preventable suffering.”
A version of this article first appeared on Medscape.com.
Experts at the National Comprehensive Cancer Network have now issued an updated recommendation for COVID-19 vaccination in people with cancer. The panel calls for these patients to be among the highest-priority group to be vaccinated against COVID-19 and to receive the newly approved third dose of vaccine.
The NCCN has recommended in February that all patients receiving active cancer treatment should receive a COVID-19 vaccine and should be prioritized for vaccination. In August, the FDA authorized a third dose of either the Pfizer or Moderna COVID-19 vaccines for people with compromised immune systems. Those eligible for a third dose include solid organ transplant recipients, those undergoing cancer treatments, and people with autoimmune diseases that suppress their immune systems
The new NCCN recommendations state that the following groups should be considered eligible for a third dose of the mRNA COVID-19 vaccine immediately, based on the latest decisions from the Food and Drug Administration and the Centers for Disease Control and Prevention:
- Patients with solid tumors (either new or recurring) receiving treatment within 1 year of their initial vaccine dose, regardless of their type of cancer therapy.
- Patients with active hematologic malignancies regardless of whether they are currently receiving cancer therapy.
- Anyone who received a stem cell transplant (SCT) or engineered cellular therapy (for example, chimeric antigen receptor T cells), especially within the past 2 years.
- Any recipients of allogeneic SCT on immunosuppressive therapy or with a history of graft-versus-host disease regardless of the time of transplant.
- Anyone with an additional immunosuppressive condition (for example, HIV) or being treated with immunosuppressive agents unrelated to their cancer therapy.
Cancer patients at high risk of complications
As previously reported by this news organization, infection with COVID-19 in people with cancer can severely impact survival. One study published in 2020 found that patients with both COVID-19 infection and progressing cancer had a fivefold increase in the risk of 30-day mortality, compared with COVID-19–positive cancer patients who were in remission or had no evidence of cancer.
Another study found that cancer type, stage, and recent treatment could affect outcomes of COVID-19 in patients with cancer. Patients with hematologic malignancies and metastatic cancers had higher risks of developing severe or critical COVID-19 symptoms, being admitted to the ICU, requiring ventilation, and dying. Conversely, those with nonmetastatic disease had outcomes that were comparable with persons without cancer and a COVID-19 infection. This study also found that having undergone recent surgery or receiving immunotherapy also put patients at a higher risk of poor outcomes, although patients with cancer who were treated with radiotherapy had outcomes similar to those of noncancer COVID-19 patients.
“COVID-19 can be very dangerous, especially for people living with cancer, which is why we’re so grateful for safe and effective vaccines that are saving lives,” Robert W. Carlson, MD, CEO of NCCN, said in a statement.
Right timing and location
The current NCCN update also recommends that individuals wait at least 4 weeks between the second and third doses, and those who are infected with COVID-19 after being vaccinated should wait until they have documented clearance of the virus before receiving a third dose.
It also recommends that people who live in the same household with immunocompromised individuals should also get a third dose once it becomes available, and that it is best to have a third dose of the same type of vaccine as the first two doses. However, a different mRNA vaccine is also acceptable.
Immunocompromised individuals should try to receive their third dose in a health care delivery setting, as opposed to a pharmacy or public vaccination clinic if possible, as it would limit their risk of exposure to the general population.
Steve Pergam, MD, MPH, associate professor, vaccine and infectious disease division, Fred Hutchinson Cancer Research Center, Seattle, commented that it is still necessary to take precautions, even after getting the booster dose.
“That means, even after a third dose of vaccine, we still recommend immunocompromised people, such as those undergoing cancer treatment, continue to be cautious, wear masks, and avoid large group gatherings, particularly around those who are unvaccinated,” said Dr. Pergam, who is also coleader of the NCCN COVID-19 Vaccination Advisory Committee. “All of us should do our part to reduce the spread of COVID-19 and get vaccinated to protect those around us from preventable suffering.”
A version of this article first appeared on Medscape.com.
Changing minds: What moves the needle for the unvaccinated?
Not so long ago, Heather Simpson of Dallas was known as the anti-vaccine mom who dressed as “the measles” for Halloween. She painted red spots on her face and posted her photo on Facebook, joking: “Was trying to think of the least scary thing I could be for Halloween … so I became the measles.” It went viral with the anti-vaccine crowd.
But between that Halloween and today, a series of “aha” moments transformed Ms. Simpson’s attitudes toward vaccines.
In January 2021, one of those moments involved her daughter, now 4, who was scratched by a feral cat, raising concerns about tetanus. Her daughter had been bitten by a dog when she was just 1, and Ms. Simpson turned down advice then to get a tetanus shot. “I was convinced the tetanus shot would kill her faster than the tetanus.”
After the cat incident, the anxiety was so exhausting, she listened to the nurse practitioner at the clinic, whom she trusted. The nurse gently reassured Ms. Simpson that the shot was less risky than the possibility of tetanus – but did not bombard her with statistics – and that won over Ms. Simpson and triggered an overall rethinking of her vaccine stance.
Fast-forward to February, and that “aha” turned into action when Ms. Simpson launched a “Back to the Vax” effort with a fellow former vaccine opponent. Through their website, Facebook page, and podcasts, they now encourage people to get the COVID-19 vaccine, as well as other immunizations.
Challenge: Reaching the rest
With just over 52% of those eligible in the United States fully vaccinated as of Sept. 1,
Recent data and a poll show some movement in the right direction, as immunizations are increasing and hesitancy is declining among certain groups. According to federal officials, about 14 million people in the United States got their first dose in August, an increase of 4 million, compared to the numbers who got it in July.
And a new poll from the Axios-IPSOS Coronavirus Index found only one in five Americans, or 20%, say they are not likely to get the vaccine, while “hard opposition,” those not at all likely, has dropped to 14% of those adults.
But there is still a lot of work to do. So, how do medical professionals or concerned citizens reach those who haven’t gotten vaccinated yet, whatever their reason?
Many experts in communication and persuasion that this news organization talked to agree that throwing statistics at people hesitant to get the COVID-19 vaccine is generally useless and often backfires.
So what does work, according to these experts?
- Emphasizing the trends of more people getting vaccinated.
- Focusing on everyone’s freedom of choice.
- Listening to concerns without judgment.
- Offering credible information.
- Correcting myths when necessary.
- Helping them fit vaccination into their “world view.”
Stories over statistics
Talking about the trends of vaccinations can definitely change minds about getting vaccinated, said Robert Cialdini, PhD, regents professor emeritus of psychology and marketing at Arizona State University, Tempe, and author of the recently updated book, “Influence: The Psychology of Persuasion,” which has sold over 5 million copies since it was first published in 1984.
Face-to-face with a hesitant patient, a doctor can say: “More and more people are being vaccinated every day,” Dr. Cialdini says. “The reason you say more and more is [that] it conveys a trend. When people see a trend, they project it into the future that it is going to get even larger.”
A focus on choice can also help people change their minds and accept the vaccine, he says. “A lot of conspiracy theorists claim they don’t want to do it because they are being pushed or forced by the government, and they are resisting that.”
If that’s the case, presenting people with new information, such as the increased infectiousness of the Delta variant, and suggesting that a decision be made based on the new information, can work, Dr. Cialdini says, but be sure to end with: “It’s completely up to you.”
“This removes all their sense of being pushed. It says, ‘Here is all the evidence.’ ” At this point, a doctor’s personal recommendation with a patient who trusts him or her may sway them, Dr. Cialdini said. “I think you have to personalize the communication in both directions. That is, to say, ‘For someone in your situation, I would personally recommend that you get the vaccine.’ ” A health care professional’s authority and expertise can carry the day, he says, although “not always.”
This approach worked, Dr. Cialdini says, with a friend of the family hesitant about the COVID-19 vaccine. “I told him: ‘We have gotten it. You trust us, right?’ ” He waited for the person to say yes.
Then: “For someone in your position, my personal recommendation is to get vaccinated. There is new information about the vaccine, and more and more people are getting vaccinated. And of course, it is completely up to you.”
The person decided to get the vaccine.
‘Live in that space’
“People develop negative attitudes [about vaccines] by accessing alternative sources of information, anecdotes, and personal stories,” said Matthew Seeger, PhD, dean of the College of Fine, Performing, and Communication Arts and codirector of the Center for Emerging Infectious Diseases at Wayne State University in Detroit.
“If we are going to change their opinion, we need to live in that space.” That means listening first, he says. Ask: “Where did you get that information? How credible do you think the sources are? What do you mean about the vaccine changing DNA?”
Then, you might respond, he said, by addressing that specific information, such as, “We have no cases of DNA being changed.”
Dr. Seeger recalls that his mother would simply talk louder when she couldn’t understand someone who wasn’t a native English speaker. “That’s what we are trying to do with the vaccine-hesitant,” he says. “In some cases, we are yelling at them.” Instead, he says, probe their sources of information.
For some who are vaccine-hesitant, Dr. Seeger said, it is not just about the vaccine. The attitude about vaccines is tied in, often, with a distrust of government and feelings about personal freedom. “That’s one reason it’s so hard to change the attitude.” For some, getting the vaccine in a family against the vaccine might also disrupt their social structure or even get them ostracized.
For these people, a health care provider might give opportunities to get the vaccine without affecting either what they see as their political stance or upsetting family harmony. “There are places you can go, make an appointment, get a vaccine, and nobody knows,” Dr. Seeger said.
One Missouri doctor told CNN that some people calling for a vaccine appointment do request privacy, such as going through a drive-thru or having the shot as they sit in their cars. She said the hospital tries to accommodate them, reasoning that every additional vaccine shot is a win.
Dr. Seeger agrees. “Of course there are still public records,” he says, “but you can still claim you are a vaccine denier. It’s very difficult to persuade people to give up their whole world. Vaccine denial is part of that world. At this point, we need to do whatever we can to get people vaccinated.”
From peer to peer
A theme that runs through many of these persuasion techniques is peer pressure.
One example, while a bit more profane and confrontational than some groups, is COVIDAteMyFace, a subgroup, or “subreddit,” of the popular online site Reddit, which hosts numerous forums inviting users to share news and comments on a variety of topics. The subreddit has over 20,000 members. Its purpose, says the sub’s creator, “was to document the folks who denied COVID, then got bitten in the ass by it.” Reports are of actual cases.
“It’s interesting and powerful that Reddit users are taking this on,” Dr. Seeger said. And this kind of peer pressure, or peer-to-peer information, can be persuasive, he says. “We often seek consensual validation from peers about risk messages and risk behaviors.”
For instance, hurricane evacuation notices are more effective, he said, when people learn their neighbors are leaving.
Peer information – “the number of others who are doing or believing or responding to something – definitely persuades people,” agreed Dr. Cialdini. “When a lot of others are responding in a particular way – for example, getting vaccinated – people follow for three reasons: The action seems more appropriate or correct, it appears more feasible to perform, and it avoids social disapproval from those others.”
Let them talk, give them time
Gladys Jimenez is a contact tracer and “vaccine ambassador” for Tracing Health, a partnership between the Oregon Public Health Institute and the Public Health Institute that has nearly 300 bilingual contract tracers who serve the ethnic communities they’re from. During a typical week, she talks to 50 people or more, and promoting the vaccine is top of mind.
The conversations, Ms. Jimenez said, are like a dance. She presents information, then steps back and lets them talk. “I want to hear the person talk, where they are coming from, where they are at.” Depending on what they say, she gives them more information or corrects their misinformation. “They often will say, ‘Oh, I didn’t know that.’ ”
It’s rarely one conversation that convinces hesitant people, she said. “I’m planting this seed in their brain. ... people want someone to listen to them ... they want to vent.”
Once you let them do that, Ms. Jimenez said, “I can tell the person is in a different state of mind.” She also knows that people “will make the decision in their own time.”
With time, people can change their minds, as a Southern California woman who resisted at first (and asked to remain anonymous) can attest. “When the vaccine first came out, I remember thinking [that] it was a quick fix to a very big problem,” she said. The lack of full FDA approval, which has since been granted, was also an issue. She doesn’t oppose vaccines, she said, but was leery just of the COVID-19 vaccine.
When her longtime partner got his vaccine, he urged her to go right away for hers. She stalled. He got his second dose and grew impatient with her hesitancy. It began to wear on the relationship. Finally, the woman talked to two health care professionals she knew socially. They both follow the science, and “they both could explain vaccination to me in a way that resonated. The information was coming from sources I already trusted.”
Those conversations are what convinced her to get vaccinated this summer.
Simpson’s transformation
Ms. Simpson of Back to the Vax got her first COVID-19 immunization April 16. She had an allergic reaction, including severe itchiness and a bad headache, and needed emergency care, she said. Even so, she scheduled her second shot appointment.
Like many who turned against vaccines as adults, Ms. Simpson had all her childhood vaccines, but she developed a distrust after watching a lengthy documentary series that warned of vaccine dangers as an adult.
Looking back at that documentary, she thought about how it seems to blame everything – childhood cancer, ADHD, autism, allergies – on vaccinations. That suddenly seemed like sketchy science to her.
So did the claim from a family friend who said she knew someone who got the flu shot and began walking backward. She researched on her own, and with time, she decided to be pro-vaccines.
These days, she continues to find that stories, not statistics, are changing the minds of many who decide to get vaccinated. If the nurse practitioner urging the tetanus shot for her daughter had told her that the tetanus shot is linked with problems in one of a specific number of people who get it, no matter how large that second number was, Ms. Simpson said she would have thought: “What if she is that one?”
So she relies on stories that point out how universally vulnerable people are to COVID-19 first, facts next.
“Facts help once you are already moved,” Ms. Simpson said.
A version of this article first appeared on WebMD.com.
Not so long ago, Heather Simpson of Dallas was known as the anti-vaccine mom who dressed as “the measles” for Halloween. She painted red spots on her face and posted her photo on Facebook, joking: “Was trying to think of the least scary thing I could be for Halloween … so I became the measles.” It went viral with the anti-vaccine crowd.
But between that Halloween and today, a series of “aha” moments transformed Ms. Simpson’s attitudes toward vaccines.
In January 2021, one of those moments involved her daughter, now 4, who was scratched by a feral cat, raising concerns about tetanus. Her daughter had been bitten by a dog when she was just 1, and Ms. Simpson turned down advice then to get a tetanus shot. “I was convinced the tetanus shot would kill her faster than the tetanus.”
After the cat incident, the anxiety was so exhausting, she listened to the nurse practitioner at the clinic, whom she trusted. The nurse gently reassured Ms. Simpson that the shot was less risky than the possibility of tetanus – but did not bombard her with statistics – and that won over Ms. Simpson and triggered an overall rethinking of her vaccine stance.
Fast-forward to February, and that “aha” turned into action when Ms. Simpson launched a “Back to the Vax” effort with a fellow former vaccine opponent. Through their website, Facebook page, and podcasts, they now encourage people to get the COVID-19 vaccine, as well as other immunizations.
Challenge: Reaching the rest
With just over 52% of those eligible in the United States fully vaccinated as of Sept. 1,
Recent data and a poll show some movement in the right direction, as immunizations are increasing and hesitancy is declining among certain groups. According to federal officials, about 14 million people in the United States got their first dose in August, an increase of 4 million, compared to the numbers who got it in July.
And a new poll from the Axios-IPSOS Coronavirus Index found only one in five Americans, or 20%, say they are not likely to get the vaccine, while “hard opposition,” those not at all likely, has dropped to 14% of those adults.
But there is still a lot of work to do. So, how do medical professionals or concerned citizens reach those who haven’t gotten vaccinated yet, whatever their reason?
Many experts in communication and persuasion that this news organization talked to agree that throwing statistics at people hesitant to get the COVID-19 vaccine is generally useless and often backfires.
So what does work, according to these experts?
- Emphasizing the trends of more people getting vaccinated.
- Focusing on everyone’s freedom of choice.
- Listening to concerns without judgment.
- Offering credible information.
- Correcting myths when necessary.
- Helping them fit vaccination into their “world view.”
Stories over statistics
Talking about the trends of vaccinations can definitely change minds about getting vaccinated, said Robert Cialdini, PhD, regents professor emeritus of psychology and marketing at Arizona State University, Tempe, and author of the recently updated book, “Influence: The Psychology of Persuasion,” which has sold over 5 million copies since it was first published in 1984.
Face-to-face with a hesitant patient, a doctor can say: “More and more people are being vaccinated every day,” Dr. Cialdini says. “The reason you say more and more is [that] it conveys a trend. When people see a trend, they project it into the future that it is going to get even larger.”
A focus on choice can also help people change their minds and accept the vaccine, he says. “A lot of conspiracy theorists claim they don’t want to do it because they are being pushed or forced by the government, and they are resisting that.”
If that’s the case, presenting people with new information, such as the increased infectiousness of the Delta variant, and suggesting that a decision be made based on the new information, can work, Dr. Cialdini says, but be sure to end with: “It’s completely up to you.”
“This removes all their sense of being pushed. It says, ‘Here is all the evidence.’ ” At this point, a doctor’s personal recommendation with a patient who trusts him or her may sway them, Dr. Cialdini said. “I think you have to personalize the communication in both directions. That is, to say, ‘For someone in your situation, I would personally recommend that you get the vaccine.’ ” A health care professional’s authority and expertise can carry the day, he says, although “not always.”
This approach worked, Dr. Cialdini says, with a friend of the family hesitant about the COVID-19 vaccine. “I told him: ‘We have gotten it. You trust us, right?’ ” He waited for the person to say yes.
Then: “For someone in your position, my personal recommendation is to get vaccinated. There is new information about the vaccine, and more and more people are getting vaccinated. And of course, it is completely up to you.”
The person decided to get the vaccine.
‘Live in that space’
“People develop negative attitudes [about vaccines] by accessing alternative sources of information, anecdotes, and personal stories,” said Matthew Seeger, PhD, dean of the College of Fine, Performing, and Communication Arts and codirector of the Center for Emerging Infectious Diseases at Wayne State University in Detroit.
“If we are going to change their opinion, we need to live in that space.” That means listening first, he says. Ask: “Where did you get that information? How credible do you think the sources are? What do you mean about the vaccine changing DNA?”
Then, you might respond, he said, by addressing that specific information, such as, “We have no cases of DNA being changed.”
Dr. Seeger recalls that his mother would simply talk louder when she couldn’t understand someone who wasn’t a native English speaker. “That’s what we are trying to do with the vaccine-hesitant,” he says. “In some cases, we are yelling at them.” Instead, he says, probe their sources of information.
For some who are vaccine-hesitant, Dr. Seeger said, it is not just about the vaccine. The attitude about vaccines is tied in, often, with a distrust of government and feelings about personal freedom. “That’s one reason it’s so hard to change the attitude.” For some, getting the vaccine in a family against the vaccine might also disrupt their social structure or even get them ostracized.
For these people, a health care provider might give opportunities to get the vaccine without affecting either what they see as their political stance or upsetting family harmony. “There are places you can go, make an appointment, get a vaccine, and nobody knows,” Dr. Seeger said.
One Missouri doctor told CNN that some people calling for a vaccine appointment do request privacy, such as going through a drive-thru or having the shot as they sit in their cars. She said the hospital tries to accommodate them, reasoning that every additional vaccine shot is a win.
Dr. Seeger agrees. “Of course there are still public records,” he says, “but you can still claim you are a vaccine denier. It’s very difficult to persuade people to give up their whole world. Vaccine denial is part of that world. At this point, we need to do whatever we can to get people vaccinated.”
From peer to peer
A theme that runs through many of these persuasion techniques is peer pressure.
One example, while a bit more profane and confrontational than some groups, is COVIDAteMyFace, a subgroup, or “subreddit,” of the popular online site Reddit, which hosts numerous forums inviting users to share news and comments on a variety of topics. The subreddit has over 20,000 members. Its purpose, says the sub’s creator, “was to document the folks who denied COVID, then got bitten in the ass by it.” Reports are of actual cases.
“It’s interesting and powerful that Reddit users are taking this on,” Dr. Seeger said. And this kind of peer pressure, or peer-to-peer information, can be persuasive, he says. “We often seek consensual validation from peers about risk messages and risk behaviors.”
For instance, hurricane evacuation notices are more effective, he said, when people learn their neighbors are leaving.
Peer information – “the number of others who are doing or believing or responding to something – definitely persuades people,” agreed Dr. Cialdini. “When a lot of others are responding in a particular way – for example, getting vaccinated – people follow for three reasons: The action seems more appropriate or correct, it appears more feasible to perform, and it avoids social disapproval from those others.”
Let them talk, give them time
Gladys Jimenez is a contact tracer and “vaccine ambassador” for Tracing Health, a partnership between the Oregon Public Health Institute and the Public Health Institute that has nearly 300 bilingual contract tracers who serve the ethnic communities they’re from. During a typical week, she talks to 50 people or more, and promoting the vaccine is top of mind.
The conversations, Ms. Jimenez said, are like a dance. She presents information, then steps back and lets them talk. “I want to hear the person talk, where they are coming from, where they are at.” Depending on what they say, she gives them more information or corrects their misinformation. “They often will say, ‘Oh, I didn’t know that.’ ”
It’s rarely one conversation that convinces hesitant people, she said. “I’m planting this seed in their brain. ... people want someone to listen to them ... they want to vent.”
Once you let them do that, Ms. Jimenez said, “I can tell the person is in a different state of mind.” She also knows that people “will make the decision in their own time.”
With time, people can change their minds, as a Southern California woman who resisted at first (and asked to remain anonymous) can attest. “When the vaccine first came out, I remember thinking [that] it was a quick fix to a very big problem,” she said. The lack of full FDA approval, which has since been granted, was also an issue. She doesn’t oppose vaccines, she said, but was leery just of the COVID-19 vaccine.
When her longtime partner got his vaccine, he urged her to go right away for hers. She stalled. He got his second dose and grew impatient with her hesitancy. It began to wear on the relationship. Finally, the woman talked to two health care professionals she knew socially. They both follow the science, and “they both could explain vaccination to me in a way that resonated. The information was coming from sources I already trusted.”
Those conversations are what convinced her to get vaccinated this summer.
Simpson’s transformation
Ms. Simpson of Back to the Vax got her first COVID-19 immunization April 16. She had an allergic reaction, including severe itchiness and a bad headache, and needed emergency care, she said. Even so, she scheduled her second shot appointment.
Like many who turned against vaccines as adults, Ms. Simpson had all her childhood vaccines, but she developed a distrust after watching a lengthy documentary series that warned of vaccine dangers as an adult.
Looking back at that documentary, she thought about how it seems to blame everything – childhood cancer, ADHD, autism, allergies – on vaccinations. That suddenly seemed like sketchy science to her.
So did the claim from a family friend who said she knew someone who got the flu shot and began walking backward. She researched on her own, and with time, she decided to be pro-vaccines.
These days, she continues to find that stories, not statistics, are changing the minds of many who decide to get vaccinated. If the nurse practitioner urging the tetanus shot for her daughter had told her that the tetanus shot is linked with problems in one of a specific number of people who get it, no matter how large that second number was, Ms. Simpson said she would have thought: “What if she is that one?”
So she relies on stories that point out how universally vulnerable people are to COVID-19 first, facts next.
“Facts help once you are already moved,” Ms. Simpson said.
A version of this article first appeared on WebMD.com.
Not so long ago, Heather Simpson of Dallas was known as the anti-vaccine mom who dressed as “the measles” for Halloween. She painted red spots on her face and posted her photo on Facebook, joking: “Was trying to think of the least scary thing I could be for Halloween … so I became the measles.” It went viral with the anti-vaccine crowd.
But between that Halloween and today, a series of “aha” moments transformed Ms. Simpson’s attitudes toward vaccines.
In January 2021, one of those moments involved her daughter, now 4, who was scratched by a feral cat, raising concerns about tetanus. Her daughter had been bitten by a dog when she was just 1, and Ms. Simpson turned down advice then to get a tetanus shot. “I was convinced the tetanus shot would kill her faster than the tetanus.”
After the cat incident, the anxiety was so exhausting, she listened to the nurse practitioner at the clinic, whom she trusted. The nurse gently reassured Ms. Simpson that the shot was less risky than the possibility of tetanus – but did not bombard her with statistics – and that won over Ms. Simpson and triggered an overall rethinking of her vaccine stance.
Fast-forward to February, and that “aha” turned into action when Ms. Simpson launched a “Back to the Vax” effort with a fellow former vaccine opponent. Through their website, Facebook page, and podcasts, they now encourage people to get the COVID-19 vaccine, as well as other immunizations.
Challenge: Reaching the rest
With just over 52% of those eligible in the United States fully vaccinated as of Sept. 1,
Recent data and a poll show some movement in the right direction, as immunizations are increasing and hesitancy is declining among certain groups. According to federal officials, about 14 million people in the United States got their first dose in August, an increase of 4 million, compared to the numbers who got it in July.
And a new poll from the Axios-IPSOS Coronavirus Index found only one in five Americans, or 20%, say they are not likely to get the vaccine, while “hard opposition,” those not at all likely, has dropped to 14% of those adults.
But there is still a lot of work to do. So, how do medical professionals or concerned citizens reach those who haven’t gotten vaccinated yet, whatever their reason?
Many experts in communication and persuasion that this news organization talked to agree that throwing statistics at people hesitant to get the COVID-19 vaccine is generally useless and often backfires.
So what does work, according to these experts?
- Emphasizing the trends of more people getting vaccinated.
- Focusing on everyone’s freedom of choice.
- Listening to concerns without judgment.
- Offering credible information.
- Correcting myths when necessary.
- Helping them fit vaccination into their “world view.”
Stories over statistics
Talking about the trends of vaccinations can definitely change minds about getting vaccinated, said Robert Cialdini, PhD, regents professor emeritus of psychology and marketing at Arizona State University, Tempe, and author of the recently updated book, “Influence: The Psychology of Persuasion,” which has sold over 5 million copies since it was first published in 1984.
Face-to-face with a hesitant patient, a doctor can say: “More and more people are being vaccinated every day,” Dr. Cialdini says. “The reason you say more and more is [that] it conveys a trend. When people see a trend, they project it into the future that it is going to get even larger.”
A focus on choice can also help people change their minds and accept the vaccine, he says. “A lot of conspiracy theorists claim they don’t want to do it because they are being pushed or forced by the government, and they are resisting that.”
If that’s the case, presenting people with new information, such as the increased infectiousness of the Delta variant, and suggesting that a decision be made based on the new information, can work, Dr. Cialdini says, but be sure to end with: “It’s completely up to you.”
“This removes all their sense of being pushed. It says, ‘Here is all the evidence.’ ” At this point, a doctor’s personal recommendation with a patient who trusts him or her may sway them, Dr. Cialdini said. “I think you have to personalize the communication in both directions. That is, to say, ‘For someone in your situation, I would personally recommend that you get the vaccine.’ ” A health care professional’s authority and expertise can carry the day, he says, although “not always.”
This approach worked, Dr. Cialdini says, with a friend of the family hesitant about the COVID-19 vaccine. “I told him: ‘We have gotten it. You trust us, right?’ ” He waited for the person to say yes.
Then: “For someone in your position, my personal recommendation is to get vaccinated. There is new information about the vaccine, and more and more people are getting vaccinated. And of course, it is completely up to you.”
The person decided to get the vaccine.
‘Live in that space’
“People develop negative attitudes [about vaccines] by accessing alternative sources of information, anecdotes, and personal stories,” said Matthew Seeger, PhD, dean of the College of Fine, Performing, and Communication Arts and codirector of the Center for Emerging Infectious Diseases at Wayne State University in Detroit.
“If we are going to change their opinion, we need to live in that space.” That means listening first, he says. Ask: “Where did you get that information? How credible do you think the sources are? What do you mean about the vaccine changing DNA?”
Then, you might respond, he said, by addressing that specific information, such as, “We have no cases of DNA being changed.”
Dr. Seeger recalls that his mother would simply talk louder when she couldn’t understand someone who wasn’t a native English speaker. “That’s what we are trying to do with the vaccine-hesitant,” he says. “In some cases, we are yelling at them.” Instead, he says, probe their sources of information.
For some who are vaccine-hesitant, Dr. Seeger said, it is not just about the vaccine. The attitude about vaccines is tied in, often, with a distrust of government and feelings about personal freedom. “That’s one reason it’s so hard to change the attitude.” For some, getting the vaccine in a family against the vaccine might also disrupt their social structure or even get them ostracized.
For these people, a health care provider might give opportunities to get the vaccine without affecting either what they see as their political stance or upsetting family harmony. “There are places you can go, make an appointment, get a vaccine, and nobody knows,” Dr. Seeger said.
One Missouri doctor told CNN that some people calling for a vaccine appointment do request privacy, such as going through a drive-thru or having the shot as they sit in their cars. She said the hospital tries to accommodate them, reasoning that every additional vaccine shot is a win.
Dr. Seeger agrees. “Of course there are still public records,” he says, “but you can still claim you are a vaccine denier. It’s very difficult to persuade people to give up their whole world. Vaccine denial is part of that world. At this point, we need to do whatever we can to get people vaccinated.”
From peer to peer
A theme that runs through many of these persuasion techniques is peer pressure.
One example, while a bit more profane and confrontational than some groups, is COVIDAteMyFace, a subgroup, or “subreddit,” of the popular online site Reddit, which hosts numerous forums inviting users to share news and comments on a variety of topics. The subreddit has over 20,000 members. Its purpose, says the sub’s creator, “was to document the folks who denied COVID, then got bitten in the ass by it.” Reports are of actual cases.
“It’s interesting and powerful that Reddit users are taking this on,” Dr. Seeger said. And this kind of peer pressure, or peer-to-peer information, can be persuasive, he says. “We often seek consensual validation from peers about risk messages and risk behaviors.”
For instance, hurricane evacuation notices are more effective, he said, when people learn their neighbors are leaving.
Peer information – “the number of others who are doing or believing or responding to something – definitely persuades people,” agreed Dr. Cialdini. “When a lot of others are responding in a particular way – for example, getting vaccinated – people follow for three reasons: The action seems more appropriate or correct, it appears more feasible to perform, and it avoids social disapproval from those others.”
Let them talk, give them time
Gladys Jimenez is a contact tracer and “vaccine ambassador” for Tracing Health, a partnership between the Oregon Public Health Institute and the Public Health Institute that has nearly 300 bilingual contract tracers who serve the ethnic communities they’re from. During a typical week, she talks to 50 people or more, and promoting the vaccine is top of mind.
The conversations, Ms. Jimenez said, are like a dance. She presents information, then steps back and lets them talk. “I want to hear the person talk, where they are coming from, where they are at.” Depending on what they say, she gives them more information or corrects their misinformation. “They often will say, ‘Oh, I didn’t know that.’ ”
It’s rarely one conversation that convinces hesitant people, she said. “I’m planting this seed in their brain. ... people want someone to listen to them ... they want to vent.”
Once you let them do that, Ms. Jimenez said, “I can tell the person is in a different state of mind.” She also knows that people “will make the decision in their own time.”
With time, people can change their minds, as a Southern California woman who resisted at first (and asked to remain anonymous) can attest. “When the vaccine first came out, I remember thinking [that] it was a quick fix to a very big problem,” she said. The lack of full FDA approval, which has since been granted, was also an issue. She doesn’t oppose vaccines, she said, but was leery just of the COVID-19 vaccine.
When her longtime partner got his vaccine, he urged her to go right away for hers. She stalled. He got his second dose and grew impatient with her hesitancy. It began to wear on the relationship. Finally, the woman talked to two health care professionals she knew socially. They both follow the science, and “they both could explain vaccination to me in a way that resonated. The information was coming from sources I already trusted.”
Those conversations are what convinced her to get vaccinated this summer.
Simpson’s transformation
Ms. Simpson of Back to the Vax got her first COVID-19 immunization April 16. She had an allergic reaction, including severe itchiness and a bad headache, and needed emergency care, she said. Even so, she scheduled her second shot appointment.
Like many who turned against vaccines as adults, Ms. Simpson had all her childhood vaccines, but she developed a distrust after watching a lengthy documentary series that warned of vaccine dangers as an adult.
Looking back at that documentary, she thought about how it seems to blame everything – childhood cancer, ADHD, autism, allergies – on vaccinations. That suddenly seemed like sketchy science to her.
So did the claim from a family friend who said she knew someone who got the flu shot and began walking backward. She researched on her own, and with time, she decided to be pro-vaccines.
These days, she continues to find that stories, not statistics, are changing the minds of many who decide to get vaccinated. If the nurse practitioner urging the tetanus shot for her daughter had told her that the tetanus shot is linked with problems in one of a specific number of people who get it, no matter how large that second number was, Ms. Simpson said she would have thought: “What if she is that one?”
So she relies on stories that point out how universally vulnerable people are to COVID-19 first, facts next.
“Facts help once you are already moved,” Ms. Simpson said.
A version of this article first appeared on WebMD.com.
Politics or protection? What’s behind the push for boosters?
That plan, which was first announced on Aug. 18, has raised eyebrows because it comes in advance of regulatory reviews by the Food and Drug Administration and recommendations from the Centers for Disease Control and Prevention. Those reviews are needed to determine whether third doses of these vaccines are effective or even safe. The move could have important legal ramifications for doctors and patients, too.
On Aug. 31, two high-level officials in the FDA’s Office of Vaccines Research and Review abruptly resigned amid reports that they were angry that the Biden administration was making decisions that should be left up to that agency.
So far, data show that the vaccines are highly effective at preventing the most severe consequences of COVID-19 – hospitalization and death – even regarding the Delta variant. The World Health Organization has urged wealthy nations such as the United States not to offer boosters so that the limited supply of vaccines can be directed to countries with fewer resources.
White House supports boosters
In a recent press briefing, Jeff Zients, the White House COVID-19 response coordinator, defended the move.
“You know, the booster decision, which you referenced ... was made by and announced by the nation’s leading public health officials, including Dr. Walensky; Dr. Fauci; Surgeon General Vivek Murthy; Dr. Janet Woodcock; the FDA acting commissioner, Dr. Francis Collins; Dr. Kessler; and others,” Mr. Zients said.
“And as our medical experts laid out, having reviewed all of the available data, it is in their clinical judgment that it is time to prepare Americans for a booster shot.”
He said a target date of Sept. 20 was announced so as to give states and practitioners time to prepare. He also said the move to give boosters was meant to help the United States stay ahead of a rapidly changing virus. Mr. Zients added that whether boosters will be administered starting on Sept. 20 depends on the FDA’s and CDC’s giving the go-ahead.
“Booster doses are going to be handled the same way all vaccines are handled,” said Kristen Nordlund, a CDC spokesperson. “Companies will have to provide data to FDA. FDA will have to make a decision and authorize the use of those, and ACIP [the Advisory Committee on Immunization Practices] will have to look at the evidence as well and make recommendations on top of FDA’s regulatory action,” she said.
Ms. Nordlund agreed that the planned Sept. 20 start date for boosters was something to which they aspired and was not necessarily set.
Historically, the FDA has needed at least 4 months to review a change to a vaccine’s approval, even on an accelerated schedule. Reviewers use that time to assess data regarding individual patients in a study, to review raw data, and essentially to check a drug company’s math and conclusions. The Biden administration’s timeline would shorten that review period from months to just a few weeks.
‘FDA in a very difficult position’
After the FDA approves, the ACIP of the CDC must meet to review the evidence and make recommendations on the use of the boosters in the United States.
Pfizer says it completed its submission for a supplemental biologics license application to the FDA on Aug. 27. To meet a Sept. 20 timeline, the entire process would have to be completed within 3 weeks.
“I don’t think that was handled, you know, ideally,” said Peter Lurie, MD, president of the Center for Science in the Public Interest and former associate commissioner of public health strategy and analysis at the FDA.
“It puts FDA in a very difficult position,” Dr. Lurie said. “It’s almost as if the decision has been made and they’re just checking a box, and that is, you know, contrary to the what FDA – at least the internal people at FDA – have been trying to do for ages.”
He said the agency took great pains with the emergency use authorizations and the full approvals of the vaccines to work as rapidly but thoroughly as possible. They did not skip steps.
“I think all of that reflected very well on the agency,” Dr. Lurie said. “And I think it worked out well in terms of trust in the vaccines.”
Although additional doses of vaccine are expected to be safe, little is known about side effects or adverse events after a third dose.
“It’s critical to wait for additional data and regulatory allowance for booster doses,” Sara Oliver, MD, a member of the CDC’s epidemic intelligence service, said in an Aug. 30 presentation to the ACIP, which is charged with making recommendations for use of all vaccines in the United States.
Boosters already being given
But after the White House announced that boosters were on the way, many people are not waiting.
Many health care practitioners and pharmacies have already been giving people third doses of vaccines, even if they are not among the immunocompromised – the group for which the shots are currently approved.
“You can walk into a pharmacy and ask for a third dose. Depending on which pharmacy you go to, you may get it,” said Helen Talbot, MD, associate professor of medicine at Vanderbilt University, Nashville, Tenn., and a member of the ACIP.
She says she has a friend who recently went for a checkup and was offered a third dose. His physician is already giving extra doses to everyone who is older than 65.
Dr. Talbot said that in fairness, pharmacies in the United States are throwing away doses of vaccine because they are expiring before they get used.
“Many of us may or may not be ready to give a third dose but would rather give someone a third dose than throw a vaccine away,” she said.
Consequences of a third shot
But giving or getting a third dose before approval by the FDA may have legal consequences.
In the ACIP meeting on Aug. 30, Demetre Daskalakis, MD, who leads vaccine equity efforts at the CDC, cautioned that physicians who give extra doses of the vaccine before the FDA and CDC have signed off may be in violation of practitioner agreements with the federal government and might not be covered by the federal PREP Act. The PREP Act provides immunity from lawsuits for people who administer COVID-19 vaccines and compensates patients in the event of injury. Patients who get a vaccine and suffer a rare but serious side effect may lose the ability to claim compensation offered by the act.
“Many of us gasped when he said that,” Dr. Talbot said, “because that’s a big deal.”
The ACIP signaled that it is considering recommending boosters for a much narrower slice of the American population than the Biden administration has suggested.
They said that so far, the data point only to the need for boosters for seniors, who are the patients most likely to experience breakthrough infections that require hospitalization, and health care workers, who are needed now more than ever and cannot work if they’re sick.
In a White House news briefing Aug. 31, CDC Director Rochelle Walensky, MD, was asked about the ACIP’s conclusions and whether she believed there were enough data to recommend booster shots for most Americans 8 months after their last dose.
“The ACIP did not review international data that actually has led us to be even more concerned about increased risk of vaccine effectiveness waning against hospitalization, severe disease, and death. They will be reviewing that as well,” she said.
A version of this article first appeared on Medscape.com.
That plan, which was first announced on Aug. 18, has raised eyebrows because it comes in advance of regulatory reviews by the Food and Drug Administration and recommendations from the Centers for Disease Control and Prevention. Those reviews are needed to determine whether third doses of these vaccines are effective or even safe. The move could have important legal ramifications for doctors and patients, too.
On Aug. 31, two high-level officials in the FDA’s Office of Vaccines Research and Review abruptly resigned amid reports that they were angry that the Biden administration was making decisions that should be left up to that agency.
So far, data show that the vaccines are highly effective at preventing the most severe consequences of COVID-19 – hospitalization and death – even regarding the Delta variant. The World Health Organization has urged wealthy nations such as the United States not to offer boosters so that the limited supply of vaccines can be directed to countries with fewer resources.
White House supports boosters
In a recent press briefing, Jeff Zients, the White House COVID-19 response coordinator, defended the move.
“You know, the booster decision, which you referenced ... was made by and announced by the nation’s leading public health officials, including Dr. Walensky; Dr. Fauci; Surgeon General Vivek Murthy; Dr. Janet Woodcock; the FDA acting commissioner, Dr. Francis Collins; Dr. Kessler; and others,” Mr. Zients said.
“And as our medical experts laid out, having reviewed all of the available data, it is in their clinical judgment that it is time to prepare Americans for a booster shot.”
He said a target date of Sept. 20 was announced so as to give states and practitioners time to prepare. He also said the move to give boosters was meant to help the United States stay ahead of a rapidly changing virus. Mr. Zients added that whether boosters will be administered starting on Sept. 20 depends on the FDA’s and CDC’s giving the go-ahead.
“Booster doses are going to be handled the same way all vaccines are handled,” said Kristen Nordlund, a CDC spokesperson. “Companies will have to provide data to FDA. FDA will have to make a decision and authorize the use of those, and ACIP [the Advisory Committee on Immunization Practices] will have to look at the evidence as well and make recommendations on top of FDA’s regulatory action,” she said.
Ms. Nordlund agreed that the planned Sept. 20 start date for boosters was something to which they aspired and was not necessarily set.
Historically, the FDA has needed at least 4 months to review a change to a vaccine’s approval, even on an accelerated schedule. Reviewers use that time to assess data regarding individual patients in a study, to review raw data, and essentially to check a drug company’s math and conclusions. The Biden administration’s timeline would shorten that review period from months to just a few weeks.
‘FDA in a very difficult position’
After the FDA approves, the ACIP of the CDC must meet to review the evidence and make recommendations on the use of the boosters in the United States.
Pfizer says it completed its submission for a supplemental biologics license application to the FDA on Aug. 27. To meet a Sept. 20 timeline, the entire process would have to be completed within 3 weeks.
“I don’t think that was handled, you know, ideally,” said Peter Lurie, MD, president of the Center for Science in the Public Interest and former associate commissioner of public health strategy and analysis at the FDA.
“It puts FDA in a very difficult position,” Dr. Lurie said. “It’s almost as if the decision has been made and they’re just checking a box, and that is, you know, contrary to the what FDA – at least the internal people at FDA – have been trying to do for ages.”
He said the agency took great pains with the emergency use authorizations and the full approvals of the vaccines to work as rapidly but thoroughly as possible. They did not skip steps.
“I think all of that reflected very well on the agency,” Dr. Lurie said. “And I think it worked out well in terms of trust in the vaccines.”
Although additional doses of vaccine are expected to be safe, little is known about side effects or adverse events after a third dose.
“It’s critical to wait for additional data and regulatory allowance for booster doses,” Sara Oliver, MD, a member of the CDC’s epidemic intelligence service, said in an Aug. 30 presentation to the ACIP, which is charged with making recommendations for use of all vaccines in the United States.
Boosters already being given
But after the White House announced that boosters were on the way, many people are not waiting.
Many health care practitioners and pharmacies have already been giving people third doses of vaccines, even if they are not among the immunocompromised – the group for which the shots are currently approved.
“You can walk into a pharmacy and ask for a third dose. Depending on which pharmacy you go to, you may get it,” said Helen Talbot, MD, associate professor of medicine at Vanderbilt University, Nashville, Tenn., and a member of the ACIP.
She says she has a friend who recently went for a checkup and was offered a third dose. His physician is already giving extra doses to everyone who is older than 65.
Dr. Talbot said that in fairness, pharmacies in the United States are throwing away doses of vaccine because they are expiring before they get used.
“Many of us may or may not be ready to give a third dose but would rather give someone a third dose than throw a vaccine away,” she said.
Consequences of a third shot
But giving or getting a third dose before approval by the FDA may have legal consequences.
In the ACIP meeting on Aug. 30, Demetre Daskalakis, MD, who leads vaccine equity efforts at the CDC, cautioned that physicians who give extra doses of the vaccine before the FDA and CDC have signed off may be in violation of practitioner agreements with the federal government and might not be covered by the federal PREP Act. The PREP Act provides immunity from lawsuits for people who administer COVID-19 vaccines and compensates patients in the event of injury. Patients who get a vaccine and suffer a rare but serious side effect may lose the ability to claim compensation offered by the act.
“Many of us gasped when he said that,” Dr. Talbot said, “because that’s a big deal.”
The ACIP signaled that it is considering recommending boosters for a much narrower slice of the American population than the Biden administration has suggested.
They said that so far, the data point only to the need for boosters for seniors, who are the patients most likely to experience breakthrough infections that require hospitalization, and health care workers, who are needed now more than ever and cannot work if they’re sick.
In a White House news briefing Aug. 31, CDC Director Rochelle Walensky, MD, was asked about the ACIP’s conclusions and whether she believed there were enough data to recommend booster shots for most Americans 8 months after their last dose.
“The ACIP did not review international data that actually has led us to be even more concerned about increased risk of vaccine effectiveness waning against hospitalization, severe disease, and death. They will be reviewing that as well,” she said.
A version of this article first appeared on Medscape.com.
That plan, which was first announced on Aug. 18, has raised eyebrows because it comes in advance of regulatory reviews by the Food and Drug Administration and recommendations from the Centers for Disease Control and Prevention. Those reviews are needed to determine whether third doses of these vaccines are effective or even safe. The move could have important legal ramifications for doctors and patients, too.
On Aug. 31, two high-level officials in the FDA’s Office of Vaccines Research and Review abruptly resigned amid reports that they were angry that the Biden administration was making decisions that should be left up to that agency.
So far, data show that the vaccines are highly effective at preventing the most severe consequences of COVID-19 – hospitalization and death – even regarding the Delta variant. The World Health Organization has urged wealthy nations such as the United States not to offer boosters so that the limited supply of vaccines can be directed to countries with fewer resources.
White House supports boosters
In a recent press briefing, Jeff Zients, the White House COVID-19 response coordinator, defended the move.
“You know, the booster decision, which you referenced ... was made by and announced by the nation’s leading public health officials, including Dr. Walensky; Dr. Fauci; Surgeon General Vivek Murthy; Dr. Janet Woodcock; the FDA acting commissioner, Dr. Francis Collins; Dr. Kessler; and others,” Mr. Zients said.
“And as our medical experts laid out, having reviewed all of the available data, it is in their clinical judgment that it is time to prepare Americans for a booster shot.”
He said a target date of Sept. 20 was announced so as to give states and practitioners time to prepare. He also said the move to give boosters was meant to help the United States stay ahead of a rapidly changing virus. Mr. Zients added that whether boosters will be administered starting on Sept. 20 depends on the FDA’s and CDC’s giving the go-ahead.
“Booster doses are going to be handled the same way all vaccines are handled,” said Kristen Nordlund, a CDC spokesperson. “Companies will have to provide data to FDA. FDA will have to make a decision and authorize the use of those, and ACIP [the Advisory Committee on Immunization Practices] will have to look at the evidence as well and make recommendations on top of FDA’s regulatory action,” she said.
Ms. Nordlund agreed that the planned Sept. 20 start date for boosters was something to which they aspired and was not necessarily set.
Historically, the FDA has needed at least 4 months to review a change to a vaccine’s approval, even on an accelerated schedule. Reviewers use that time to assess data regarding individual patients in a study, to review raw data, and essentially to check a drug company’s math and conclusions. The Biden administration’s timeline would shorten that review period from months to just a few weeks.
‘FDA in a very difficult position’
After the FDA approves, the ACIP of the CDC must meet to review the evidence and make recommendations on the use of the boosters in the United States.
Pfizer says it completed its submission for a supplemental biologics license application to the FDA on Aug. 27. To meet a Sept. 20 timeline, the entire process would have to be completed within 3 weeks.
“I don’t think that was handled, you know, ideally,” said Peter Lurie, MD, president of the Center for Science in the Public Interest and former associate commissioner of public health strategy and analysis at the FDA.
“It puts FDA in a very difficult position,” Dr. Lurie said. “It’s almost as if the decision has been made and they’re just checking a box, and that is, you know, contrary to the what FDA – at least the internal people at FDA – have been trying to do for ages.”
He said the agency took great pains with the emergency use authorizations and the full approvals of the vaccines to work as rapidly but thoroughly as possible. They did not skip steps.
“I think all of that reflected very well on the agency,” Dr. Lurie said. “And I think it worked out well in terms of trust in the vaccines.”
Although additional doses of vaccine are expected to be safe, little is known about side effects or adverse events after a third dose.
“It’s critical to wait for additional data and regulatory allowance for booster doses,” Sara Oliver, MD, a member of the CDC’s epidemic intelligence service, said in an Aug. 30 presentation to the ACIP, which is charged with making recommendations for use of all vaccines in the United States.
Boosters already being given
But after the White House announced that boosters were on the way, many people are not waiting.
Many health care practitioners and pharmacies have already been giving people third doses of vaccines, even if they are not among the immunocompromised – the group for which the shots are currently approved.
“You can walk into a pharmacy and ask for a third dose. Depending on which pharmacy you go to, you may get it,” said Helen Talbot, MD, associate professor of medicine at Vanderbilt University, Nashville, Tenn., and a member of the ACIP.
She says she has a friend who recently went for a checkup and was offered a third dose. His physician is already giving extra doses to everyone who is older than 65.
Dr. Talbot said that in fairness, pharmacies in the United States are throwing away doses of vaccine because they are expiring before they get used.
“Many of us may or may not be ready to give a third dose but would rather give someone a third dose than throw a vaccine away,” she said.
Consequences of a third shot
But giving or getting a third dose before approval by the FDA may have legal consequences.
In the ACIP meeting on Aug. 30, Demetre Daskalakis, MD, who leads vaccine equity efforts at the CDC, cautioned that physicians who give extra doses of the vaccine before the FDA and CDC have signed off may be in violation of practitioner agreements with the federal government and might not be covered by the federal PREP Act. The PREP Act provides immunity from lawsuits for people who administer COVID-19 vaccines and compensates patients in the event of injury. Patients who get a vaccine and suffer a rare but serious side effect may lose the ability to claim compensation offered by the act.
“Many of us gasped when he said that,” Dr. Talbot said, “because that’s a big deal.”
The ACIP signaled that it is considering recommending boosters for a much narrower slice of the American population than the Biden administration has suggested.
They said that so far, the data point only to the need for boosters for seniors, who are the patients most likely to experience breakthrough infections that require hospitalization, and health care workers, who are needed now more than ever and cannot work if they’re sick.
In a White House news briefing Aug. 31, CDC Director Rochelle Walensky, MD, was asked about the ACIP’s conclusions and whether she believed there were enough data to recommend booster shots for most Americans 8 months after their last dose.
“The ACIP did not review international data that actually has led us to be even more concerned about increased risk of vaccine effectiveness waning against hospitalization, severe disease, and death. They will be reviewing that as well,” she said.
A version of this article first appeared on Medscape.com.
WHO tracking new COVID-19 variant called Mu
The variant, also known as B.1.621, was first identified in Colombia in January. It has now been detected in 43 countries and was added to the WHO’s “variant of interest” list Aug. 30.
“The Mu variant has a constellation of mutations that indicate potential properties of immune escape,” the WHO wrote in its weekly COVID-19 update on Aug 31.
Preliminary data suggests that the Mu variant may be able to evade antibodies at levels similar to the Beta variant, the WHO wrote, though more studies are needed. The Beta variant, also known as B.1.351, was first detected in South Africa and has shown some ability to evade vaccines.
As of Aug. 29, the global prevalence of the Mu variant appears to be less than 0.1%. But its prevalence in South America has “consistently increased,” the WHO wrote, now making up 39% of cases in Colombia and 13% of cases in Ecuador.
More than 4,700 cases of the Mu variant have been identified worldwide through genomic sequencing, according to Outbreak.info, an open-source database operated by Scripps Research. The United States has identified 2,011 of these cases, with 348 in California. As of Sept. 2, only one state -- Nebraska -- had not yet reported a Mu case.
“At the moment, it looks like there’s genuine cause for concern in USA, Central America, and South America, but as we saw with Delta, a potent variant can traverse the globe in the blink of an eye,” Danny Altmann, PhD, an immunologist at Imperial College London, told The Telegraph.
The WHO is monitoring nine variants with genetic mutations that could make them more transmissible, lead to more severe disease, and help them evade vaccines. The Delta variant, which is now a dominant form of the virus in the United States and worldwide, has led to a surge in cases and hospitalizations this summer.
In its report, the WHO said it would monitor the Mu variant for changes, “particularly with the co-circulation of the Delta variant.”
“Mu looks potentially good at immune evasion,” Dr. Altmann told The Telegraph. “For my taste, it’s a stark reminder that this isn’t by any means over. On a planet of 4.4 million-plus new infections per week, there are new variants popping up all the time, and little reason to feel complacent.”
A version of this article first appeared on WebMD.com.
The variant, also known as B.1.621, was first identified in Colombia in January. It has now been detected in 43 countries and was added to the WHO’s “variant of interest” list Aug. 30.
“The Mu variant has a constellation of mutations that indicate potential properties of immune escape,” the WHO wrote in its weekly COVID-19 update on Aug 31.
Preliminary data suggests that the Mu variant may be able to evade antibodies at levels similar to the Beta variant, the WHO wrote, though more studies are needed. The Beta variant, also known as B.1.351, was first detected in South Africa and has shown some ability to evade vaccines.
As of Aug. 29, the global prevalence of the Mu variant appears to be less than 0.1%. But its prevalence in South America has “consistently increased,” the WHO wrote, now making up 39% of cases in Colombia and 13% of cases in Ecuador.
More than 4,700 cases of the Mu variant have been identified worldwide through genomic sequencing, according to Outbreak.info, an open-source database operated by Scripps Research. The United States has identified 2,011 of these cases, with 348 in California. As of Sept. 2, only one state -- Nebraska -- had not yet reported a Mu case.
“At the moment, it looks like there’s genuine cause for concern in USA, Central America, and South America, but as we saw with Delta, a potent variant can traverse the globe in the blink of an eye,” Danny Altmann, PhD, an immunologist at Imperial College London, told The Telegraph.
The WHO is monitoring nine variants with genetic mutations that could make them more transmissible, lead to more severe disease, and help them evade vaccines. The Delta variant, which is now a dominant form of the virus in the United States and worldwide, has led to a surge in cases and hospitalizations this summer.
In its report, the WHO said it would monitor the Mu variant for changes, “particularly with the co-circulation of the Delta variant.”
“Mu looks potentially good at immune evasion,” Dr. Altmann told The Telegraph. “For my taste, it’s a stark reminder that this isn’t by any means over. On a planet of 4.4 million-plus new infections per week, there are new variants popping up all the time, and little reason to feel complacent.”
A version of this article first appeared on WebMD.com.
The variant, also known as B.1.621, was first identified in Colombia in January. It has now been detected in 43 countries and was added to the WHO’s “variant of interest” list Aug. 30.
“The Mu variant has a constellation of mutations that indicate potential properties of immune escape,” the WHO wrote in its weekly COVID-19 update on Aug 31.
Preliminary data suggests that the Mu variant may be able to evade antibodies at levels similar to the Beta variant, the WHO wrote, though more studies are needed. The Beta variant, also known as B.1.351, was first detected in South Africa and has shown some ability to evade vaccines.
As of Aug. 29, the global prevalence of the Mu variant appears to be less than 0.1%. But its prevalence in South America has “consistently increased,” the WHO wrote, now making up 39% of cases in Colombia and 13% of cases in Ecuador.
More than 4,700 cases of the Mu variant have been identified worldwide through genomic sequencing, according to Outbreak.info, an open-source database operated by Scripps Research. The United States has identified 2,011 of these cases, with 348 in California. As of Sept. 2, only one state -- Nebraska -- had not yet reported a Mu case.
“At the moment, it looks like there’s genuine cause for concern in USA, Central America, and South America, but as we saw with Delta, a potent variant can traverse the globe in the blink of an eye,” Danny Altmann, PhD, an immunologist at Imperial College London, told The Telegraph.
The WHO is monitoring nine variants with genetic mutations that could make them more transmissible, lead to more severe disease, and help them evade vaccines. The Delta variant, which is now a dominant form of the virus in the United States and worldwide, has led to a surge in cases and hospitalizations this summer.
In its report, the WHO said it would monitor the Mu variant for changes, “particularly with the co-circulation of the Delta variant.”
“Mu looks potentially good at immune evasion,” Dr. Altmann told The Telegraph. “For my taste, it’s a stark reminder that this isn’t by any means over. On a planet of 4.4 million-plus new infections per week, there are new variants popping up all the time, and little reason to feel complacent.”
A version of this article first appeared on WebMD.com.
A long look at long haulers
With the number of pediatric infections with SARS-CoV-2 rising it is not surprising that children with persistent symptoms are beginning to accumulate. Who are these pediatric “long haulers” and do they differ from their adult counterparts? The answer is far from clear because the terms “long COVID” and “long hauler” are not well defined. But, I suspect we will find that they will be similar in most respects.
In a recent Guest Essay in the New York Times, two medical school professors attempt to inject some common sense into the long hauler phenomenon. (“The Truth About Long Covid is Complicated. Better Treatment Isn’t,” Adam Gaffney and Zackary Berger, The New York Times, Aug. 18, 2021).
The authors divide the patients with long COVID into three categories. The first includes those who are complaining of persistent cough and fatigue for up to 3 months, a not unexpected course for patients recovering from a significant respiratory illness like pneumonia.
The second group comprises patients who developed acute respiratory distress syndrome during the course of their SARS-CoV-2 infection. These unfortunate individuals likely incurred lung damage that may have triggered renal damage and delirium and may never regain full function.
The third group of patients reports a wide variety of less specific symptoms including, but not limited to, severe fatigue, brain fog, shortness of breath, gastrointestinal symptoms, chronic pain, and palpitations.
The authors of the essay refer to several studies in which there was little if any correlation between these patients’ complaints and their antibody levels. In fact, one study of adolescents found that in a group with similar symptoms many of the individuals had no serologic evidence of SARS-CoV-2 infection.
Unfortunately, the lay public, the media, and some physicians make no distinction between these three groups and lump them all under the same long COVID umbrella. The resulting confusion seeds unwarranted anxiety among the first and third groups and may prevent some individuals from receiving the appropriate attention they deserve.
I suspect that like me, many of you see some similarities between this third group of long COVID patients and adolescents whose persistent symptoms don’t quite fit with their primary illness. Patients labeled as having post-concussion syndrome or “chronic Lyme disease” come immediately to mind. In both conditions, many of the patients had little if any evidence of severe insult from the initial event but continue to complain about a variety of symptoms including severe fatigue and brain fog.
We have done a very poor job of properly managing these patients. And there are a lot of them. A large part of the problem is labeling. In the old days one might have said these patients were having “psychosomatic” symptoms. But, while it may be an accurate description, like the term “retardation” it has been permanently tarnished. Fortunately, most of us are smart enough to avoid telling these patients that it is all in their heads.
However, convincing an individual that many of his symptoms may be the result of the psychological insult from the original disease compounded by other stresses and lifestyle factors can be a difficult sell. The task is made particularly difficult when there continue to be physicians who will miss or ignore the obvious and embark on therapeutic endeavors that are not only ineffective but can serve as a distraction from the real work of listening to and engaging these patients whose suffering may be just as real as that of those long haulers with structural damage.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
With the number of pediatric infections with SARS-CoV-2 rising it is not surprising that children with persistent symptoms are beginning to accumulate. Who are these pediatric “long haulers” and do they differ from their adult counterparts? The answer is far from clear because the terms “long COVID” and “long hauler” are not well defined. But, I suspect we will find that they will be similar in most respects.
In a recent Guest Essay in the New York Times, two medical school professors attempt to inject some common sense into the long hauler phenomenon. (“The Truth About Long Covid is Complicated. Better Treatment Isn’t,” Adam Gaffney and Zackary Berger, The New York Times, Aug. 18, 2021).
The authors divide the patients with long COVID into three categories. The first includes those who are complaining of persistent cough and fatigue for up to 3 months, a not unexpected course for patients recovering from a significant respiratory illness like pneumonia.
The second group comprises patients who developed acute respiratory distress syndrome during the course of their SARS-CoV-2 infection. These unfortunate individuals likely incurred lung damage that may have triggered renal damage and delirium and may never regain full function.
The third group of patients reports a wide variety of less specific symptoms including, but not limited to, severe fatigue, brain fog, shortness of breath, gastrointestinal symptoms, chronic pain, and palpitations.
The authors of the essay refer to several studies in which there was little if any correlation between these patients’ complaints and their antibody levels. In fact, one study of adolescents found that in a group with similar symptoms many of the individuals had no serologic evidence of SARS-CoV-2 infection.
Unfortunately, the lay public, the media, and some physicians make no distinction between these three groups and lump them all under the same long COVID umbrella. The resulting confusion seeds unwarranted anxiety among the first and third groups and may prevent some individuals from receiving the appropriate attention they deserve.
I suspect that like me, many of you see some similarities between this third group of long COVID patients and adolescents whose persistent symptoms don’t quite fit with their primary illness. Patients labeled as having post-concussion syndrome or “chronic Lyme disease” come immediately to mind. In both conditions, many of the patients had little if any evidence of severe insult from the initial event but continue to complain about a variety of symptoms including severe fatigue and brain fog.
We have done a very poor job of properly managing these patients. And there are a lot of them. A large part of the problem is labeling. In the old days one might have said these patients were having “psychosomatic” symptoms. But, while it may be an accurate description, like the term “retardation” it has been permanently tarnished. Fortunately, most of us are smart enough to avoid telling these patients that it is all in their heads.
However, convincing an individual that many of his symptoms may be the result of the psychological insult from the original disease compounded by other stresses and lifestyle factors can be a difficult sell. The task is made particularly difficult when there continue to be physicians who will miss or ignore the obvious and embark on therapeutic endeavors that are not only ineffective but can serve as a distraction from the real work of listening to and engaging these patients whose suffering may be just as real as that of those long haulers with structural damage.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
With the number of pediatric infections with SARS-CoV-2 rising it is not surprising that children with persistent symptoms are beginning to accumulate. Who are these pediatric “long haulers” and do they differ from their adult counterparts? The answer is far from clear because the terms “long COVID” and “long hauler” are not well defined. But, I suspect we will find that they will be similar in most respects.
In a recent Guest Essay in the New York Times, two medical school professors attempt to inject some common sense into the long hauler phenomenon. (“The Truth About Long Covid is Complicated. Better Treatment Isn’t,” Adam Gaffney and Zackary Berger, The New York Times, Aug. 18, 2021).
The authors divide the patients with long COVID into three categories. The first includes those who are complaining of persistent cough and fatigue for up to 3 months, a not unexpected course for patients recovering from a significant respiratory illness like pneumonia.
The second group comprises patients who developed acute respiratory distress syndrome during the course of their SARS-CoV-2 infection. These unfortunate individuals likely incurred lung damage that may have triggered renal damage and delirium and may never regain full function.
The third group of patients reports a wide variety of less specific symptoms including, but not limited to, severe fatigue, brain fog, shortness of breath, gastrointestinal symptoms, chronic pain, and palpitations.
The authors of the essay refer to several studies in which there was little if any correlation between these patients’ complaints and their antibody levels. In fact, one study of adolescents found that in a group with similar symptoms many of the individuals had no serologic evidence of SARS-CoV-2 infection.
Unfortunately, the lay public, the media, and some physicians make no distinction between these three groups and lump them all under the same long COVID umbrella. The resulting confusion seeds unwarranted anxiety among the first and third groups and may prevent some individuals from receiving the appropriate attention they deserve.
I suspect that like me, many of you see some similarities between this third group of long COVID patients and adolescents whose persistent symptoms don’t quite fit with their primary illness. Patients labeled as having post-concussion syndrome or “chronic Lyme disease” come immediately to mind. In both conditions, many of the patients had little if any evidence of severe insult from the initial event but continue to complain about a variety of symptoms including severe fatigue and brain fog.
We have done a very poor job of properly managing these patients. And there are a lot of them. A large part of the problem is labeling. In the old days one might have said these patients were having “psychosomatic” symptoms. But, while it may be an accurate description, like the term “retardation” it has been permanently tarnished. Fortunately, most of us are smart enough to avoid telling these patients that it is all in their heads.
However, convincing an individual that many of his symptoms may be the result of the psychological insult from the original disease compounded by other stresses and lifestyle factors can be a difficult sell. The task is made particularly difficult when there continue to be physicians who will miss or ignore the obvious and embark on therapeutic endeavors that are not only ineffective but can serve as a distraction from the real work of listening to and engaging these patients whose suffering may be just as real as that of those long haulers with structural damage.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].



