User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
OSA patients report sleeping better with dronabinol
in a new study.
A paper published in the January edition of Sleep presents data from a phase 2, blinded, randomized controlled trial of the nonselective cannabinoid 1 and cannabinoid 2 receptor agonist, dronabinol, in 73 adults with moderate or severe obstructive sleep apnea (OSA). No approved drug treatments for OSA exist, and this study provides results “from the largest and longest randomized controlled trial to date of any putative drug treatment for OSA,” the researchers wrote.
Patients were randomized to 2.5 mg dronabinol or 10 mg dronabinol daily for up to 6 weeks, or placebo. At the end of treatment, researchers saw significant increases in the apnea-hypopnea index among the patients on placebo, while those who received dronabinol showed decreases in the number of apnea and hypopnea events per hour. Patients given the 2.5-mg dose of dronabinol had a mean decrease of 10.7 events per hour, and those on the 10-mg dose had a mean decrease of 12.9 events per hour compared with placebo.
The difference between the placebo and treatment arms was significant for both dosages, and the apnea-hypopnea index decreases were similar between the two dosages of dronabinol.
These effects were largely due to reductions in apnea events; the largest reduction was seen in the REM apnea index in patients treated with the 10-mg dose of dronabinol. However, there were few effects on the expression of hypopneas, except in the higher-dose group.
After adjustment for age, race, ethnicity, and baseline apnea-hypopnea index, the increases seen in the placebo group were no longer significant, but the decreases from baseline seen in the treatment arms were greater.
Dronabinol treatment was also associated with significant decreases, compared with placebo, in non-REM apnea-hypopnea index and REM apnea-hypopnea index.
Patients’ self-reported daytime sleepiness, measured by the Epworth Sleepiness Scale, remained similar compared with baseline in those who received placebo and the 2.5-mg/day dose of dronabinol, but decreased significantly by a mean of −2.3 points compared with placebo in those on the higher dose of dronabinol.
There were no significant changes from baseline in objective sleepiness, as measured by the maintenance of wakefulness test, in any of the study groups. Researchers also saw no significant changes in sleep architecture, oxygenation, or the duration of supine sleep in any of the study groups, although the patients on the higher dose of dronabinol showed a slight increase in REM sleep and those on placebo showed a slight decrease.
Younger patients and those with a greater preponderance of REM-related apnea/hypopnea, and shorter average event duration were both more likely to respond to treatment, but apart from these factors there were no other influences on likelihood of patients responding to dronabinol.
David W. Carley, PhD, of the University of Illinois at Chicago, and his coauthors noted that there was a great need for pharmacological treatments for obstructive sleep apnea because positive airway pressure – while effective – has poor long-term adherence rates.
“Based on a series of animal investigations, we proposed that drugs which dampen afferent vagal feedback to the medulla may be effective in stabilizing respiratory pattern generation and increasing activation of upper airway dilating muscles during sleep,” they wrote.
One patient experienced diarrhea and vomiting that required admission to hospital, and which was judged as possibly related to the study medication. There were six other withdrawals due to adverse events including dizziness and vision changes, vertigo, ECG arrhythmias, and headache with dizziness and vomiting. Overall, nearly 90% of patients reported at least one adverse event, but the rates did not differ significantly between the treatment and placebo arms.
The researchers noted that significantly higher satisfaction scores were seen among patients receiving the higher dose of dronabinol.
“All of these observations argue that dronabinol, at doses from 2.5 to 10 mg/day, is safe for use by medically stable patients with moderate or severe OSA,” the authors wrote. “Participants also tolerated and adhered well to daily self-administration of dronabinol.”
The National Institutes of Health, National Heart, Lung, and Blood Institute, and National Center for Advancing Translational Sciences funded the study. One author declared grants from the National Institutes of Health for the study, and patents related to treatment of sleep-related breathing disorders by cannabinoid drugs. He also holds stock in RespireRx Pharmaceuticals, which holds an exclusive license to these and other related patents.
SOURCE: Carley D, et al. Sleep. 2018 Jan 1. doi: 10.1093/sleep/zsx184
This study has found a small overall effect on the apnea-hypopnea index with treatment, but a strong beneficial effect on subjective sleepiness. In addition, participants who received the higher dose of the drug showed significant satisfaction with their therapy. It is therefore intriguing that there was no impact on objective wakefulness or sleep architecture with this treatment.
This suggests that perhaps sleepiness and subjective wellbeing may be improved without necessarily seeing major improvements in the apnea-hypopnea index, which calls into question our use of this index as a primary end-point.
Sigrid C. Veasey, MD, is with the Center for Sleep and Circadian Neurobiology at the Perelman School of Medicine, University of Pennsylvania, Philadelphia. These comments are taken from an accompanying (Sleep 2018 Jan 1. doi: 10.1093/sleep/zsy014). No conflicts of interest were declared.
This study has found a small overall effect on the apnea-hypopnea index with treatment, but a strong beneficial effect on subjective sleepiness. In addition, participants who received the higher dose of the drug showed significant satisfaction with their therapy. It is therefore intriguing that there was no impact on objective wakefulness or sleep architecture with this treatment.
This suggests that perhaps sleepiness and subjective wellbeing may be improved without necessarily seeing major improvements in the apnea-hypopnea index, which calls into question our use of this index as a primary end-point.
Sigrid C. Veasey, MD, is with the Center for Sleep and Circadian Neurobiology at the Perelman School of Medicine, University of Pennsylvania, Philadelphia. These comments are taken from an accompanying (Sleep 2018 Jan 1. doi: 10.1093/sleep/zsy014). No conflicts of interest were declared.
This study has found a small overall effect on the apnea-hypopnea index with treatment, but a strong beneficial effect on subjective sleepiness. In addition, participants who received the higher dose of the drug showed significant satisfaction with their therapy. It is therefore intriguing that there was no impact on objective wakefulness or sleep architecture with this treatment.
This suggests that perhaps sleepiness and subjective wellbeing may be improved without necessarily seeing major improvements in the apnea-hypopnea index, which calls into question our use of this index as a primary end-point.
Sigrid C. Veasey, MD, is with the Center for Sleep and Circadian Neurobiology at the Perelman School of Medicine, University of Pennsylvania, Philadelphia. These comments are taken from an accompanying (Sleep 2018 Jan 1. doi: 10.1093/sleep/zsy014). No conflicts of interest were declared.
in a new study.
A paper published in the January edition of Sleep presents data from a phase 2, blinded, randomized controlled trial of the nonselective cannabinoid 1 and cannabinoid 2 receptor agonist, dronabinol, in 73 adults with moderate or severe obstructive sleep apnea (OSA). No approved drug treatments for OSA exist, and this study provides results “from the largest and longest randomized controlled trial to date of any putative drug treatment for OSA,” the researchers wrote.
Patients were randomized to 2.5 mg dronabinol or 10 mg dronabinol daily for up to 6 weeks, or placebo. At the end of treatment, researchers saw significant increases in the apnea-hypopnea index among the patients on placebo, while those who received dronabinol showed decreases in the number of apnea and hypopnea events per hour. Patients given the 2.5-mg dose of dronabinol had a mean decrease of 10.7 events per hour, and those on the 10-mg dose had a mean decrease of 12.9 events per hour compared with placebo.
The difference between the placebo and treatment arms was significant for both dosages, and the apnea-hypopnea index decreases were similar between the two dosages of dronabinol.
These effects were largely due to reductions in apnea events; the largest reduction was seen in the REM apnea index in patients treated with the 10-mg dose of dronabinol. However, there were few effects on the expression of hypopneas, except in the higher-dose group.
After adjustment for age, race, ethnicity, and baseline apnea-hypopnea index, the increases seen in the placebo group were no longer significant, but the decreases from baseline seen in the treatment arms were greater.
Dronabinol treatment was also associated with significant decreases, compared with placebo, in non-REM apnea-hypopnea index and REM apnea-hypopnea index.
Patients’ self-reported daytime sleepiness, measured by the Epworth Sleepiness Scale, remained similar compared with baseline in those who received placebo and the 2.5-mg/day dose of dronabinol, but decreased significantly by a mean of −2.3 points compared with placebo in those on the higher dose of dronabinol.
There were no significant changes from baseline in objective sleepiness, as measured by the maintenance of wakefulness test, in any of the study groups. Researchers also saw no significant changes in sleep architecture, oxygenation, or the duration of supine sleep in any of the study groups, although the patients on the higher dose of dronabinol showed a slight increase in REM sleep and those on placebo showed a slight decrease.
Younger patients and those with a greater preponderance of REM-related apnea/hypopnea, and shorter average event duration were both more likely to respond to treatment, but apart from these factors there were no other influences on likelihood of patients responding to dronabinol.
David W. Carley, PhD, of the University of Illinois at Chicago, and his coauthors noted that there was a great need for pharmacological treatments for obstructive sleep apnea because positive airway pressure – while effective – has poor long-term adherence rates.
“Based on a series of animal investigations, we proposed that drugs which dampen afferent vagal feedback to the medulla may be effective in stabilizing respiratory pattern generation and increasing activation of upper airway dilating muscles during sleep,” they wrote.
One patient experienced diarrhea and vomiting that required admission to hospital, and which was judged as possibly related to the study medication. There were six other withdrawals due to adverse events including dizziness and vision changes, vertigo, ECG arrhythmias, and headache with dizziness and vomiting. Overall, nearly 90% of patients reported at least one adverse event, but the rates did not differ significantly between the treatment and placebo arms.
The researchers noted that significantly higher satisfaction scores were seen among patients receiving the higher dose of dronabinol.
“All of these observations argue that dronabinol, at doses from 2.5 to 10 mg/day, is safe for use by medically stable patients with moderate or severe OSA,” the authors wrote. “Participants also tolerated and adhered well to daily self-administration of dronabinol.”
The National Institutes of Health, National Heart, Lung, and Blood Institute, and National Center for Advancing Translational Sciences funded the study. One author declared grants from the National Institutes of Health for the study, and patents related to treatment of sleep-related breathing disorders by cannabinoid drugs. He also holds stock in RespireRx Pharmaceuticals, which holds an exclusive license to these and other related patents.
SOURCE: Carley D, et al. Sleep. 2018 Jan 1. doi: 10.1093/sleep/zsx184
in a new study.
A paper published in the January edition of Sleep presents data from a phase 2, blinded, randomized controlled trial of the nonselective cannabinoid 1 and cannabinoid 2 receptor agonist, dronabinol, in 73 adults with moderate or severe obstructive sleep apnea (OSA). No approved drug treatments for OSA exist, and this study provides results “from the largest and longest randomized controlled trial to date of any putative drug treatment for OSA,” the researchers wrote.
Patients were randomized to 2.5 mg dronabinol or 10 mg dronabinol daily for up to 6 weeks, or placebo. At the end of treatment, researchers saw significant increases in the apnea-hypopnea index among the patients on placebo, while those who received dronabinol showed decreases in the number of apnea and hypopnea events per hour. Patients given the 2.5-mg dose of dronabinol had a mean decrease of 10.7 events per hour, and those on the 10-mg dose had a mean decrease of 12.9 events per hour compared with placebo.
The difference between the placebo and treatment arms was significant for both dosages, and the apnea-hypopnea index decreases were similar between the two dosages of dronabinol.
These effects were largely due to reductions in apnea events; the largest reduction was seen in the REM apnea index in patients treated with the 10-mg dose of dronabinol. However, there were few effects on the expression of hypopneas, except in the higher-dose group.
After adjustment for age, race, ethnicity, and baseline apnea-hypopnea index, the increases seen in the placebo group were no longer significant, but the decreases from baseline seen in the treatment arms were greater.
Dronabinol treatment was also associated with significant decreases, compared with placebo, in non-REM apnea-hypopnea index and REM apnea-hypopnea index.
Patients’ self-reported daytime sleepiness, measured by the Epworth Sleepiness Scale, remained similar compared with baseline in those who received placebo and the 2.5-mg/day dose of dronabinol, but decreased significantly by a mean of −2.3 points compared with placebo in those on the higher dose of dronabinol.
There were no significant changes from baseline in objective sleepiness, as measured by the maintenance of wakefulness test, in any of the study groups. Researchers also saw no significant changes in sleep architecture, oxygenation, or the duration of supine sleep in any of the study groups, although the patients on the higher dose of dronabinol showed a slight increase in REM sleep and those on placebo showed a slight decrease.
Younger patients and those with a greater preponderance of REM-related apnea/hypopnea, and shorter average event duration were both more likely to respond to treatment, but apart from these factors there were no other influences on likelihood of patients responding to dronabinol.
David W. Carley, PhD, of the University of Illinois at Chicago, and his coauthors noted that there was a great need for pharmacological treatments for obstructive sleep apnea because positive airway pressure – while effective – has poor long-term adherence rates.
“Based on a series of animal investigations, we proposed that drugs which dampen afferent vagal feedback to the medulla may be effective in stabilizing respiratory pattern generation and increasing activation of upper airway dilating muscles during sleep,” they wrote.
One patient experienced diarrhea and vomiting that required admission to hospital, and which was judged as possibly related to the study medication. There were six other withdrawals due to adverse events including dizziness and vision changes, vertigo, ECG arrhythmias, and headache with dizziness and vomiting. Overall, nearly 90% of patients reported at least one adverse event, but the rates did not differ significantly between the treatment and placebo arms.
The researchers noted that significantly higher satisfaction scores were seen among patients receiving the higher dose of dronabinol.
“All of these observations argue that dronabinol, at doses from 2.5 to 10 mg/day, is safe for use by medically stable patients with moderate or severe OSA,” the authors wrote. “Participants also tolerated and adhered well to daily self-administration of dronabinol.”
The National Institutes of Health, National Heart, Lung, and Blood Institute, and National Center for Advancing Translational Sciences funded the study. One author declared grants from the National Institutes of Health for the study, and patents related to treatment of sleep-related breathing disorders by cannabinoid drugs. He also holds stock in RespireRx Pharmaceuticals, which holds an exclusive license to these and other related patents.
SOURCE: Carley D, et al. Sleep. 2018 Jan 1. doi: 10.1093/sleep/zsx184
FROM SLEEP
Key clinical point: A cannabinoid receptor agonist may significantly reduce apnea and hypopnea events in patients with obstructive sleep apnea.
Major finding: Patients who received either low dose or high-dose dronabinol showed significant decreases in apnea and hypopnea events compared to those on placebo.
Data source: Randomized controlled, blinded phase II trial in 73 patients with obstructive sleep apnea.
Disclosures: The National Institutes of Health, National Heart Lung and Blood Institute, and National Center for Advancing Translational Sciences funded the study. One author declared grants from the National Institutes of Health for the study, and patents related to treatment of sleep-related breathing disorders by cannabinoid drugs. He also holds stock in RespireRx Pharmaceuticals, which holds an exclusive license to these and other related patents.
Source: Carley D, et al. Sleep. 2018 Jan 1. doi: 10.1093/sleep/zsx184.
OSA may provide cardioprotection
, according to researchers.
In a study of 127 patients presenting with acute coronary syndromes (ACS), median peak cardiac troponin-I (cTn-I) values were significantly higher in patients without obstructive sleep apnea, compared with OSA patients (10.7; interquartile range: 1.78-40.1, vs. 3.79; IQR: 0.37-24.3, respectively; P = .04 ). The findings were published Feb. 5 in CHEST.
The study comprised 89 OSA patients and 38 non-OSA patients who were admitted to a hospital for acute coronary syndromes. The OSA group had a median apnea-hypopnea index (AHI) of 32, while the non-OSA group had a median AHI of 4.8. There was no significant difference between the two groups in gender, age, or cardiovascular risk factors such as hypertension, diabetes mellitus, body mass index, dyslipidemia, and smoking.
The cohort was part of the Continuous Positive Airway Pressure (CPAP) in Patients With Acute Coronary Syndrome and Obstructive Sleep Apnea (ISAACC) study, a prior randomized, controlled trial that evaluated the effect of CPAP treatment on new cardiovascular events in patients with an episode of ACS and OSA, reported Alicia Sánchez-de-la-Torre, PhD, of the respiratory department at Hospital Universitari Arnau de Vilanova and Santa Maria in Catalonia, Spain, and her coauthors.
Respiratory polygraphy was performed in the first 24-72 hours after hospital admission, and patients with an AHI of at least 15 events per hour were considered to have OSA. Those with an AHI less than 15 events per hour were included in the non-OSA group.
Blood samples were collected from patients every 6 hours until two consecutive cTn-I measurements showed a decrease, with the highest measurement considered the peak cTn-I value.
Peak cTn-I value was significantly higher in non-OSA patients than in OSA patients. Median infarct size, measured by calculating the area under the cTn-I curve, was significantly different between the two groups (451 for non-OSA patients vs. 143 in OSA patients; P = .049), wrote Dr. Sánchez-de-la-Torre and her colleagues.
As cTn-I levels decreased, there was a trend toward increased OSA severity (P = .058). In the multivariable linear regression model used to assess OSA severity, patients with severe OSA had 61% lower cTn-I levels than non-OSA patients, the authors noted.
“These results suggest that patients with higher AHI are significantly more likely to have low cTn-I levels than patients without evidence of OSA, which could imply that patients with elevated AHI, particularly those with severe OSA, may experience less severe myocardial injury,” the authors said in the report. The findings “suggest that OSA has a protective effect in the context of MI,” they added.
Limitations of the study include exclusion of patients with severe ACS, exclusion of sleepy subjects, and assessment of myocardial injury using cTn-I as a biomarker, without further data to determine infarct size.
“The possible role of OSA in cardioprotection should be explored in future studies,” the authors concluded.
The authors disclosed relationships with ResMed Inc., Spanish Ministry of Health, Spanish Respiratory Society, Catalonian Cardiology Society, and ALLER. No other disclosures were reported.
SOURCE: Chest. 2018 Feb 5;153[2]:329-38. doi: 10.1016/j.chest.2017.06.046
Although this study cannot definitively establish a clinically meaningful protective effect, it does provide important “preliminary evidence supporting the concept of OSA-induced cardioprotection” and challenges existing research, according to an editorial by Doron Aronson, MD, of the department of cardiology at Rambam Medical Center, Haifa, Israel, and coauthors (CHEST. 2018 Feb 153[2]:295-7. doi: 10.1016/j.chest.2017.07.036).
The results should be interpreted with caution, especially since accurate assessment of infarct size poses a challenge, they wrote.
“Myocardial infarct size is highly variable and is influenced by the duration of coronary occlusion, ST-segment elevation or non–ST elevation myocardial infarction, infarct location, residual antegrade infarct-related artery flow, collateral flow, the presence of non–culprit vessel coronary artery disease and myocardial metabolic demand,” they wrote. “Without accounting for these variables in a small study, results may be affected by variation in the characteristics of the patients.”
Though further study is needed, the findings may have “profound clinical implications regarding our therapeutic approach to patients with sleep apnea” if confirmed, the authors concluded.
Although this study cannot definitively establish a clinically meaningful protective effect, it does provide important “preliminary evidence supporting the concept of OSA-induced cardioprotection” and challenges existing research, according to an editorial by Doron Aronson, MD, of the department of cardiology at Rambam Medical Center, Haifa, Israel, and coauthors (CHEST. 2018 Feb 153[2]:295-7. doi: 10.1016/j.chest.2017.07.036).
The results should be interpreted with caution, especially since accurate assessment of infarct size poses a challenge, they wrote.
“Myocardial infarct size is highly variable and is influenced by the duration of coronary occlusion, ST-segment elevation or non–ST elevation myocardial infarction, infarct location, residual antegrade infarct-related artery flow, collateral flow, the presence of non–culprit vessel coronary artery disease and myocardial metabolic demand,” they wrote. “Without accounting for these variables in a small study, results may be affected by variation in the characteristics of the patients.”
Though further study is needed, the findings may have “profound clinical implications regarding our therapeutic approach to patients with sleep apnea” if confirmed, the authors concluded.
Although this study cannot definitively establish a clinically meaningful protective effect, it does provide important “preliminary evidence supporting the concept of OSA-induced cardioprotection” and challenges existing research, according to an editorial by Doron Aronson, MD, of the department of cardiology at Rambam Medical Center, Haifa, Israel, and coauthors (CHEST. 2018 Feb 153[2]:295-7. doi: 10.1016/j.chest.2017.07.036).
The results should be interpreted with caution, especially since accurate assessment of infarct size poses a challenge, they wrote.
“Myocardial infarct size is highly variable and is influenced by the duration of coronary occlusion, ST-segment elevation or non–ST elevation myocardial infarction, infarct location, residual antegrade infarct-related artery flow, collateral flow, the presence of non–culprit vessel coronary artery disease and myocardial metabolic demand,” they wrote. “Without accounting for these variables in a small study, results may be affected by variation in the characteristics of the patients.”
Though further study is needed, the findings may have “profound clinical implications regarding our therapeutic approach to patients with sleep apnea” if confirmed, the authors concluded.
, according to researchers.
In a study of 127 patients presenting with acute coronary syndromes (ACS), median peak cardiac troponin-I (cTn-I) values were significantly higher in patients without obstructive sleep apnea, compared with OSA patients (10.7; interquartile range: 1.78-40.1, vs. 3.79; IQR: 0.37-24.3, respectively; P = .04 ). The findings were published Feb. 5 in CHEST.
The study comprised 89 OSA patients and 38 non-OSA patients who were admitted to a hospital for acute coronary syndromes. The OSA group had a median apnea-hypopnea index (AHI) of 32, while the non-OSA group had a median AHI of 4.8. There was no significant difference between the two groups in gender, age, or cardiovascular risk factors such as hypertension, diabetes mellitus, body mass index, dyslipidemia, and smoking.
The cohort was part of the Continuous Positive Airway Pressure (CPAP) in Patients With Acute Coronary Syndrome and Obstructive Sleep Apnea (ISAACC) study, a prior randomized, controlled trial that evaluated the effect of CPAP treatment on new cardiovascular events in patients with an episode of ACS and OSA, reported Alicia Sánchez-de-la-Torre, PhD, of the respiratory department at Hospital Universitari Arnau de Vilanova and Santa Maria in Catalonia, Spain, and her coauthors.
Respiratory polygraphy was performed in the first 24-72 hours after hospital admission, and patients with an AHI of at least 15 events per hour were considered to have OSA. Those with an AHI less than 15 events per hour were included in the non-OSA group.
Blood samples were collected from patients every 6 hours until two consecutive cTn-I measurements showed a decrease, with the highest measurement considered the peak cTn-I value.
Peak cTn-I value was significantly higher in non-OSA patients than in OSA patients. Median infarct size, measured by calculating the area under the cTn-I curve, was significantly different between the two groups (451 for non-OSA patients vs. 143 in OSA patients; P = .049), wrote Dr. Sánchez-de-la-Torre and her colleagues.
As cTn-I levels decreased, there was a trend toward increased OSA severity (P = .058). In the multivariable linear regression model used to assess OSA severity, patients with severe OSA had 61% lower cTn-I levels than non-OSA patients, the authors noted.
“These results suggest that patients with higher AHI are significantly more likely to have low cTn-I levels than patients without evidence of OSA, which could imply that patients with elevated AHI, particularly those with severe OSA, may experience less severe myocardial injury,” the authors said in the report. The findings “suggest that OSA has a protective effect in the context of MI,” they added.
Limitations of the study include exclusion of patients with severe ACS, exclusion of sleepy subjects, and assessment of myocardial injury using cTn-I as a biomarker, without further data to determine infarct size.
“The possible role of OSA in cardioprotection should be explored in future studies,” the authors concluded.
The authors disclosed relationships with ResMed Inc., Spanish Ministry of Health, Spanish Respiratory Society, Catalonian Cardiology Society, and ALLER. No other disclosures were reported.
SOURCE: Chest. 2018 Feb 5;153[2]:329-38. doi: 10.1016/j.chest.2017.06.046
, according to researchers.
In a study of 127 patients presenting with acute coronary syndromes (ACS), median peak cardiac troponin-I (cTn-I) values were significantly higher in patients without obstructive sleep apnea, compared with OSA patients (10.7; interquartile range: 1.78-40.1, vs. 3.79; IQR: 0.37-24.3, respectively; P = .04 ). The findings were published Feb. 5 in CHEST.
The study comprised 89 OSA patients and 38 non-OSA patients who were admitted to a hospital for acute coronary syndromes. The OSA group had a median apnea-hypopnea index (AHI) of 32, while the non-OSA group had a median AHI of 4.8. There was no significant difference between the two groups in gender, age, or cardiovascular risk factors such as hypertension, diabetes mellitus, body mass index, dyslipidemia, and smoking.
The cohort was part of the Continuous Positive Airway Pressure (CPAP) in Patients With Acute Coronary Syndrome and Obstructive Sleep Apnea (ISAACC) study, a prior randomized, controlled trial that evaluated the effect of CPAP treatment on new cardiovascular events in patients with an episode of ACS and OSA, reported Alicia Sánchez-de-la-Torre, PhD, of the respiratory department at Hospital Universitari Arnau de Vilanova and Santa Maria in Catalonia, Spain, and her coauthors.
Respiratory polygraphy was performed in the first 24-72 hours after hospital admission, and patients with an AHI of at least 15 events per hour were considered to have OSA. Those with an AHI less than 15 events per hour were included in the non-OSA group.
Blood samples were collected from patients every 6 hours until two consecutive cTn-I measurements showed a decrease, with the highest measurement considered the peak cTn-I value.
Peak cTn-I value was significantly higher in non-OSA patients than in OSA patients. Median infarct size, measured by calculating the area under the cTn-I curve, was significantly different between the two groups (451 for non-OSA patients vs. 143 in OSA patients; P = .049), wrote Dr. Sánchez-de-la-Torre and her colleagues.
As cTn-I levels decreased, there was a trend toward increased OSA severity (P = .058). In the multivariable linear regression model used to assess OSA severity, patients with severe OSA had 61% lower cTn-I levels than non-OSA patients, the authors noted.
“These results suggest that patients with higher AHI are significantly more likely to have low cTn-I levels than patients without evidence of OSA, which could imply that patients with elevated AHI, particularly those with severe OSA, may experience less severe myocardial injury,” the authors said in the report. The findings “suggest that OSA has a protective effect in the context of MI,” they added.
Limitations of the study include exclusion of patients with severe ACS, exclusion of sleepy subjects, and assessment of myocardial injury using cTn-I as a biomarker, without further data to determine infarct size.
“The possible role of OSA in cardioprotection should be explored in future studies,” the authors concluded.
The authors disclosed relationships with ResMed Inc., Spanish Ministry of Health, Spanish Respiratory Society, Catalonian Cardiology Society, and ALLER. No other disclosures were reported.
SOURCE: Chest. 2018 Feb 5;153[2]:329-38. doi: 10.1016/j.chest.2017.06.046
FROM CHEST
Key clinical point: Obstructive sleep apnea may have a protective effect in acute coronary syndromes.
Major finding: Median peak cTn-I value was significantly higher in patients without obstructive sleep apnea than in OSA patients (10.7, interquartile range: 1.78-40.1 vs. 3.79; IQR: 0.37-24.3 respectively; P = .04).
Data source: An observational study of 89 OSA and 38 non-OSA patients admitted for acute coronary syndromes.
Disclosures: The authors disclosed relationships with ResMed Inc., Spanish Ministry of Health, Spanish Respiratory Society, Catalonian Cardiology Society, and ALLER.
Source: Sánchez-de-la-Torre, A et al. CHEST. 2018 Feb 5;153[2]:329-38.
This is what a flu pandemic looks like
For the week ending Feb. 3, 2018, the proportion of outpatient visits for influenza-like illness (ILI) was 7.7%, which would appear to equal the mark of 7.7% set in October of 2009. The earlier 7.7%, however, is rounded down from 7.715%, while the current mark is rounded up from 7.653%, data from the CDC’s Fluview website show.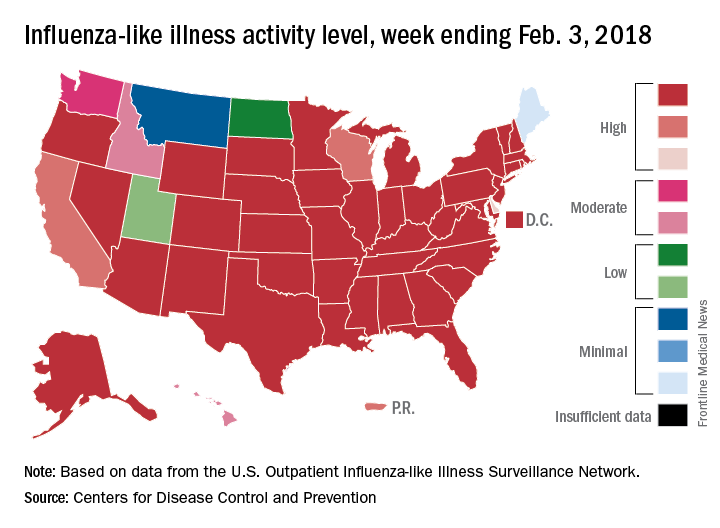
Deaths attributed to pneumonia and influenza were above the epidemic threshold set by the National Center for Health Statistics Mortality Surveillance system, acting CDC director Anne Schuchat, MD, said in a teleconference sponsored by the agency.
ILI activity was at level 10 on the CDC’s 1-10 scale in 41 states, compared with 34 the week before, and was categorized in the “high” range (levels 8-10) in another 3 states and Puerto Rico, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network. In California, which was noted as a possible bright spot last week by Dr. Schuchat because activity there had been decreasing, the ILI level went back up to level 9 after being at 7 the week before.
Flu-related hospitalizations are continuing to rise at a record clip, with the cumulative rate for the week of Feb. 3 at 59.9 per 100,000 population, the CDC reported. A total of 1 in 10 hospital-based deaths last week were related to influenza. At this point in the 2014-2015 flu season – which has the highest number of hospitalizations at 710,000 – the hospitalization rate was only 50.9 per 100,000 population.
There were 10 pediatric deaths reported for the week ending Feb. 3, although 9 occurred in previous weeks. There have been 63 flu-related deaths among children so far during the 2017-2018 season.
Dr. Schuchat continued to recommend members of the public to get a flu shot and to stay home if they are feeling sick.
“What could be mild symptoms for you could be deadly for someone else,” Dr. Schuchat said, adding that antiviral medications remain important. “Physicians do not have to wait for confirmatory flu testing. They should begin treatment with antiviral drugs immediately in they suspect they have a severely ill or a high risk patient.”
“Flu vaccines often have lower effectiveness against H3N1 viruses. However, some protection is better than none. The vaccine’s effectiveness against other flu viruses, like B and H1N1, is better. Because of the ongoing intensity of the flu season and the increasing circulation of influenza B and h1n1, we do continue to recommend vaccination even this late in the season.”
Dr. Schuchat stressed the importance of the pneumococcal pneumonia vaccine. “Flu can make people more vulnerable to secondary infections like bacterial pneumonia. We recommend people aged 65 and over get a pneumococcal pneumonia vaccine,” she said.
For the week ending Feb. 3, 2018, the proportion of outpatient visits for influenza-like illness (ILI) was 7.7%, which would appear to equal the mark of 7.7% set in October of 2009. The earlier 7.7%, however, is rounded down from 7.715%, while the current mark is rounded up from 7.653%, data from the CDC’s Fluview website show.
Deaths attributed to pneumonia and influenza were above the epidemic threshold set by the National Center for Health Statistics Mortality Surveillance system, acting CDC director Anne Schuchat, MD, said in a teleconference sponsored by the agency.
ILI activity was at level 10 on the CDC’s 1-10 scale in 41 states, compared with 34 the week before, and was categorized in the “high” range (levels 8-10) in another 3 states and Puerto Rico, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network. In California, which was noted as a possible bright spot last week by Dr. Schuchat because activity there had been decreasing, the ILI level went back up to level 9 after being at 7 the week before.
Flu-related hospitalizations are continuing to rise at a record clip, with the cumulative rate for the week of Feb. 3 at 59.9 per 100,000 population, the CDC reported. A total of 1 in 10 hospital-based deaths last week were related to influenza. At this point in the 2014-2015 flu season – which has the highest number of hospitalizations at 710,000 – the hospitalization rate was only 50.9 per 100,000 population.
There were 10 pediatric deaths reported for the week ending Feb. 3, although 9 occurred in previous weeks. There have been 63 flu-related deaths among children so far during the 2017-2018 season.
Dr. Schuchat continued to recommend members of the public to get a flu shot and to stay home if they are feeling sick.
“What could be mild symptoms for you could be deadly for someone else,” Dr. Schuchat said, adding that antiviral medications remain important. “Physicians do not have to wait for confirmatory flu testing. They should begin treatment with antiviral drugs immediately in they suspect they have a severely ill or a high risk patient.”
“Flu vaccines often have lower effectiveness against H3N1 viruses. However, some protection is better than none. The vaccine’s effectiveness against other flu viruses, like B and H1N1, is better. Because of the ongoing intensity of the flu season and the increasing circulation of influenza B and h1n1, we do continue to recommend vaccination even this late in the season.”
Dr. Schuchat stressed the importance of the pneumococcal pneumonia vaccine. “Flu can make people more vulnerable to secondary infections like bacterial pneumonia. We recommend people aged 65 and over get a pneumococcal pneumonia vaccine,” she said.
For the week ending Feb. 3, 2018, the proportion of outpatient visits for influenza-like illness (ILI) was 7.7%, which would appear to equal the mark of 7.7% set in October of 2009. The earlier 7.7%, however, is rounded down from 7.715%, while the current mark is rounded up from 7.653%, data from the CDC’s Fluview website show.
Deaths attributed to pneumonia and influenza were above the epidemic threshold set by the National Center for Health Statistics Mortality Surveillance system, acting CDC director Anne Schuchat, MD, said in a teleconference sponsored by the agency.
ILI activity was at level 10 on the CDC’s 1-10 scale in 41 states, compared with 34 the week before, and was categorized in the “high” range (levels 8-10) in another 3 states and Puerto Rico, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network. In California, which was noted as a possible bright spot last week by Dr. Schuchat because activity there had been decreasing, the ILI level went back up to level 9 after being at 7 the week before.
Flu-related hospitalizations are continuing to rise at a record clip, with the cumulative rate for the week of Feb. 3 at 59.9 per 100,000 population, the CDC reported. A total of 1 in 10 hospital-based deaths last week were related to influenza. At this point in the 2014-2015 flu season – which has the highest number of hospitalizations at 710,000 – the hospitalization rate was only 50.9 per 100,000 population.
There were 10 pediatric deaths reported for the week ending Feb. 3, although 9 occurred in previous weeks. There have been 63 flu-related deaths among children so far during the 2017-2018 season.
Dr. Schuchat continued to recommend members of the public to get a flu shot and to stay home if they are feeling sick.
“What could be mild symptoms for you could be deadly for someone else,” Dr. Schuchat said, adding that antiviral medications remain important. “Physicians do not have to wait for confirmatory flu testing. They should begin treatment with antiviral drugs immediately in they suspect they have a severely ill or a high risk patient.”
“Flu vaccines often have lower effectiveness against H3N1 viruses. However, some protection is better than none. The vaccine’s effectiveness against other flu viruses, like B and H1N1, is better. Because of the ongoing intensity of the flu season and the increasing circulation of influenza B and h1n1, we do continue to recommend vaccination even this late in the season.”
Dr. Schuchat stressed the importance of the pneumococcal pneumonia vaccine. “Flu can make people more vulnerable to secondary infections like bacterial pneumonia. We recommend people aged 65 and over get a pneumococcal pneumonia vaccine,” she said.
FROM A CDC TELECONFERENCE
Pulmonary Fibrosis Foundation offers trial-finding app
The Pulmonary Fibrosis Foundation (PFF) has begun offering a tool to help patients navigate through more than 100 clinical trials aimed at advancing the treatment of pulmonary fibrosis, according to a statement from the Foundation.
The platform includes trials for patients with idiopathic pulmonary fibrosis.
“Before this, it was pretty impossible to search for clinical trials,” noted Bill Burke, a PF patient and support group leader from Williamsburg, Va., in the statement.
The tool, which is available both on the PFF website and as a free app, draws on information from ClinicalTrials.gov.
The Foundation intends for the new tool to give patients “a voice in their care process,” according to the statement.
“We want to empower patients to actively participate in identifying clinical trials,” said Harold R. Collard, MD, senior medical adviser of research and development for the Pulmonary Fibrosis Foundation.
More information about the trial finder is available on the PFF’s website.
The Pulmonary Fibrosis Foundation (PFF) has begun offering a tool to help patients navigate through more than 100 clinical trials aimed at advancing the treatment of pulmonary fibrosis, according to a statement from the Foundation.
The platform includes trials for patients with idiopathic pulmonary fibrosis.
“Before this, it was pretty impossible to search for clinical trials,” noted Bill Burke, a PF patient and support group leader from Williamsburg, Va., in the statement.
The tool, which is available both on the PFF website and as a free app, draws on information from ClinicalTrials.gov.
The Foundation intends for the new tool to give patients “a voice in their care process,” according to the statement.
“We want to empower patients to actively participate in identifying clinical trials,” said Harold R. Collard, MD, senior medical adviser of research and development for the Pulmonary Fibrosis Foundation.
More information about the trial finder is available on the PFF’s website.
The Pulmonary Fibrosis Foundation (PFF) has begun offering a tool to help patients navigate through more than 100 clinical trials aimed at advancing the treatment of pulmonary fibrosis, according to a statement from the Foundation.
The platform includes trials for patients with idiopathic pulmonary fibrosis.
“Before this, it was pretty impossible to search for clinical trials,” noted Bill Burke, a PF patient and support group leader from Williamsburg, Va., in the statement.
The tool, which is available both on the PFF website and as a free app, draws on information from ClinicalTrials.gov.
The Foundation intends for the new tool to give patients “a voice in their care process,” according to the statement.
“We want to empower patients to actively participate in identifying clinical trials,” said Harold R. Collard, MD, senior medical adviser of research and development for the Pulmonary Fibrosis Foundation.
More information about the trial finder is available on the PFF’s website.
FDA’s standards for approving generics are questioned
TORONTO – The Food and Drug Administration’s standards for demonstrating pharmacokinetic bioequivalence between two inhaled products, which allow for single batch comparisons of approved and generic candidate products, need to be revised to address batch to batch variability, suggested a presenter at the CHEST annual meeting.
Marketing approval of a new generic drug in the United States, including orally inhaled products, generally requires a demonstration of pharmacokinetic bioequivalence to a reference listed product. The standard criterion for statistical bioequivalence applied by the FDA requires the pharmacokinetics of the generic to be within about 10% of the branded product.
In early pharmacokinetic bioequivalence studies, Elise Burmeister Getz, PhD, and her colleagues compared single batches of their generic candidate OT329 Solis 100/50 to single batches of Advair Diskus 100/50 in five individual studies and single batches of Advair Diskus 100/50 to single batches of the same drug. They also found Advair Diskus 100/50 batches that were more than 30% different from each other.
“When patients differ from one another, we put many patients in the trial. And when batches differ from one another, we should be putting many batches in the trial,” Dr. Burmeister Getz, director of clinical pharmacology at Oriel Therapeutics, said at the CHEST meeting. “If we want a robust assessment of bioequivalence and not just a check the box exercise, we really need to have product sampling that’s aligned with product variability.”
When the researchers combined the data in a meta-analysis, bioequivalence was demonstrated, but the pooled analysis could not be used for FDA registration because of its retrospective nature.
They later conducted a prospective study with multiple batches of both the generic and branded drugs. This multiple-batch bioequivalence study involved 96 healthy subjects using 16 batches each of Advair Diskus and Oriel’s OT329 Solis 100/50. A single inhalation was administered to healthy adult subjects in a randomized crossover design and blood samples were collected pre dose and up to 48 hours after inhalation.
With the FDA’s definition of bioequivalence, the generic candidate fell within the bioequivalence goalposts, Dr. Burmeister Getz noted.
The issue of pharmacokinetic variance is not unique to Advair Diskus, but she and her colleagues don’t understand why different batches show such wide variability, Dr. Burmeister Getz noted.
“The advantage of this multibatch approach is that the results of the bioequivalence assessment aren’t dependent on the single batch that happened to be chosen for the study. They are generalizable to the product because the product has been robustly represented in the study,” Dr. Burmeister Getz told attendees.
Oriel makes OT329 Solis 100/50, a fully substitutable generic to Advair Diskus 100/50, which is indicated for treating asthma. Both are multidose dry powder oral inhalation products containing fluticasone propionate, to reduce inflammation in the lungs, and salmeterol, to relax muscles in the airways, for the maintenance treatment of asthma. Advair Diskus at higher doses is indicated for asthma and COPD.
An FDA response?
Asked what the FDA makes of the batch-to-batch variability data, Dr. Burmeister Getz answered simply, “We don’t know.” Before she and her colleagues ran the 16 batch per product study, they submitted their protocol to the FDA for review, but 1 year later, they still hadn’t heard any response.
“Sponsors are apparently allowed to simply pick their batch in a careful and, dare I say manipulative way, to gain the result they want. With a single batch study the selection of batch will absolutely determine the outcome of the study.”
In vitro bioequivalence studies are already required to use multiple batches, she noted.
This research was funded by Oriel Therapeutics, an indirect wholly-owned subsidiary of Novartis AG.
TORONTO – The Food and Drug Administration’s standards for demonstrating pharmacokinetic bioequivalence between two inhaled products, which allow for single batch comparisons of approved and generic candidate products, need to be revised to address batch to batch variability, suggested a presenter at the CHEST annual meeting.
Marketing approval of a new generic drug in the United States, including orally inhaled products, generally requires a demonstration of pharmacokinetic bioequivalence to a reference listed product. The standard criterion for statistical bioequivalence applied by the FDA requires the pharmacokinetics of the generic to be within about 10% of the branded product.
In early pharmacokinetic bioequivalence studies, Elise Burmeister Getz, PhD, and her colleagues compared single batches of their generic candidate OT329 Solis 100/50 to single batches of Advair Diskus 100/50 in five individual studies and single batches of Advair Diskus 100/50 to single batches of the same drug. They also found Advair Diskus 100/50 batches that were more than 30% different from each other.
“When patients differ from one another, we put many patients in the trial. And when batches differ from one another, we should be putting many batches in the trial,” Dr. Burmeister Getz, director of clinical pharmacology at Oriel Therapeutics, said at the CHEST meeting. “If we want a robust assessment of bioequivalence and not just a check the box exercise, we really need to have product sampling that’s aligned with product variability.”
When the researchers combined the data in a meta-analysis, bioequivalence was demonstrated, but the pooled analysis could not be used for FDA registration because of its retrospective nature.
They later conducted a prospective study with multiple batches of both the generic and branded drugs. This multiple-batch bioequivalence study involved 96 healthy subjects using 16 batches each of Advair Diskus and Oriel’s OT329 Solis 100/50. A single inhalation was administered to healthy adult subjects in a randomized crossover design and blood samples were collected pre dose and up to 48 hours after inhalation.
With the FDA’s definition of bioequivalence, the generic candidate fell within the bioequivalence goalposts, Dr. Burmeister Getz noted.
The issue of pharmacokinetic variance is not unique to Advair Diskus, but she and her colleagues don’t understand why different batches show such wide variability, Dr. Burmeister Getz noted.
“The advantage of this multibatch approach is that the results of the bioequivalence assessment aren’t dependent on the single batch that happened to be chosen for the study. They are generalizable to the product because the product has been robustly represented in the study,” Dr. Burmeister Getz told attendees.
Oriel makes OT329 Solis 100/50, a fully substitutable generic to Advair Diskus 100/50, which is indicated for treating asthma. Both are multidose dry powder oral inhalation products containing fluticasone propionate, to reduce inflammation in the lungs, and salmeterol, to relax muscles in the airways, for the maintenance treatment of asthma. Advair Diskus at higher doses is indicated for asthma and COPD.
An FDA response?
Asked what the FDA makes of the batch-to-batch variability data, Dr. Burmeister Getz answered simply, “We don’t know.” Before she and her colleagues ran the 16 batch per product study, they submitted their protocol to the FDA for review, but 1 year later, they still hadn’t heard any response.
“Sponsors are apparently allowed to simply pick their batch in a careful and, dare I say manipulative way, to gain the result they want. With a single batch study the selection of batch will absolutely determine the outcome of the study.”
In vitro bioequivalence studies are already required to use multiple batches, she noted.
This research was funded by Oriel Therapeutics, an indirect wholly-owned subsidiary of Novartis AG.
TORONTO – The Food and Drug Administration’s standards for demonstrating pharmacokinetic bioequivalence between two inhaled products, which allow for single batch comparisons of approved and generic candidate products, need to be revised to address batch to batch variability, suggested a presenter at the CHEST annual meeting.
Marketing approval of a new generic drug in the United States, including orally inhaled products, generally requires a demonstration of pharmacokinetic bioequivalence to a reference listed product. The standard criterion for statistical bioequivalence applied by the FDA requires the pharmacokinetics of the generic to be within about 10% of the branded product.
In early pharmacokinetic bioequivalence studies, Elise Burmeister Getz, PhD, and her colleagues compared single batches of their generic candidate OT329 Solis 100/50 to single batches of Advair Diskus 100/50 in five individual studies and single batches of Advair Diskus 100/50 to single batches of the same drug. They also found Advair Diskus 100/50 batches that were more than 30% different from each other.
“When patients differ from one another, we put many patients in the trial. And when batches differ from one another, we should be putting many batches in the trial,” Dr. Burmeister Getz, director of clinical pharmacology at Oriel Therapeutics, said at the CHEST meeting. “If we want a robust assessment of bioequivalence and not just a check the box exercise, we really need to have product sampling that’s aligned with product variability.”
When the researchers combined the data in a meta-analysis, bioequivalence was demonstrated, but the pooled analysis could not be used for FDA registration because of its retrospective nature.
They later conducted a prospective study with multiple batches of both the generic and branded drugs. This multiple-batch bioequivalence study involved 96 healthy subjects using 16 batches each of Advair Diskus and Oriel’s OT329 Solis 100/50. A single inhalation was administered to healthy adult subjects in a randomized crossover design and blood samples were collected pre dose and up to 48 hours after inhalation.
With the FDA’s definition of bioequivalence, the generic candidate fell within the bioequivalence goalposts, Dr. Burmeister Getz noted.
The issue of pharmacokinetic variance is not unique to Advair Diskus, but she and her colleagues don’t understand why different batches show such wide variability, Dr. Burmeister Getz noted.
“The advantage of this multibatch approach is that the results of the bioequivalence assessment aren’t dependent on the single batch that happened to be chosen for the study. They are generalizable to the product because the product has been robustly represented in the study,” Dr. Burmeister Getz told attendees.
Oriel makes OT329 Solis 100/50, a fully substitutable generic to Advair Diskus 100/50, which is indicated for treating asthma. Both are multidose dry powder oral inhalation products containing fluticasone propionate, to reduce inflammation in the lungs, and salmeterol, to relax muscles in the airways, for the maintenance treatment of asthma. Advair Diskus at higher doses is indicated for asthma and COPD.
An FDA response?
Asked what the FDA makes of the batch-to-batch variability data, Dr. Burmeister Getz answered simply, “We don’t know.” Before she and her colleagues ran the 16 batch per product study, they submitted their protocol to the FDA for review, but 1 year later, they still hadn’t heard any response.
“Sponsors are apparently allowed to simply pick their batch in a careful and, dare I say manipulative way, to gain the result they want. With a single batch study the selection of batch will absolutely determine the outcome of the study.”
In vitro bioequivalence studies are already required to use multiple batches, she noted.
This research was funded by Oriel Therapeutics, an indirect wholly-owned subsidiary of Novartis AG.
AT CHEST 2017
Key clinical point: The FDA’s standards for demonstrating pharmacokinetic bioequivalence between two inhaled products need to be revised to address batch to batch variability.
Major finding: Investigators found Advair Diskus 100/50 batches that were more than 30% different from each other.
Data source: Pharmacokinetic bioequivalence studies comparing batches of Advair Diskus 100/50 to each other, and to batches of the generic candidate OT329 Solis 100/50.
Disclosures: This research was funded by Oriel Therapeutics, an indirect wholly-owned subsidiary of Novartis AG. Dr. Burmeister Getz is director of clinical pharmacology at Oriel Therapeutics.
Drug combo indicated for bacterial pneumonia
(Avycaz) to include hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) in adults.
Specifically, the approved indication is for infections caused by certain Gram-negative bacteria – some of which are increasingly resistant to available antibiotics – including, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, Pseudomonas aeruginosa, and Haemophilus influenzae.
There have not been new treatment options for HABP/VABP caused by Gram-negative bacteria in more than 15 years, according to Allergan, the drug’s manufacturer.
This is the third approved indication for ceftazidime/avibactam; the other two indications are for complicated intra-abdominal infections (in combination with metronidazole) and for complicated urinary tract infections.
(Avycaz) to include hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) in adults.
Specifically, the approved indication is for infections caused by certain Gram-negative bacteria – some of which are increasingly resistant to available antibiotics – including, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, Pseudomonas aeruginosa, and Haemophilus influenzae.
There have not been new treatment options for HABP/VABP caused by Gram-negative bacteria in more than 15 years, according to Allergan, the drug’s manufacturer.
This is the third approved indication for ceftazidime/avibactam; the other two indications are for complicated intra-abdominal infections (in combination with metronidazole) and for complicated urinary tract infections.
(Avycaz) to include hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) in adults.
Specifically, the approved indication is for infections caused by certain Gram-negative bacteria – some of which are increasingly resistant to available antibiotics – including, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Serratia marcescens, Proteus mirabilis, Pseudomonas aeruginosa, and Haemophilus influenzae.
There have not been new treatment options for HABP/VABP caused by Gram-negative bacteria in more than 15 years, according to Allergan, the drug’s manufacturer.
This is the third approved indication for ceftazidime/avibactam; the other two indications are for complicated intra-abdominal infections (in combination with metronidazole) and for complicated urinary tract infections.
Hospitals filling as flu season worsens
Through the last full week of January, the cumulative “hospitalization rate is the highest we’ve seen,” acting Centers for Disease Control and Prevention director Anne Schuchat, MD, said. For the current season so far, the hospitalization rate stands at 51.4 per 100,000 population, putting it on pace to top the total of 710,000 flu-related admissions that occurred during the 2014-2015 season, she said in a weekly briefing Feb. 2.
Flu-related pediatric deaths also took a big jump for the week as another 16 were reported, which brings the total for the season to 53. Of the children who have died so far, only 20% were vaccinated, said Dan Jernigan, MD, MPH, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta. He also noted that half of the children who have been hospitalized did not had an underlying condition.
The one bit of good news for the week was that activity in the West seems to be easing up, Dr. Schuchat said. The geographic spread of ILI was reported as widespread in 48 states, which is down from 49 the previous week because Oregon dropped off the list. To go along with that, the ILI activity level in California has dropped 2 weeks in a row and now stands at level 7, the CDC data show.
Through the last full week of January, the cumulative “hospitalization rate is the highest we’ve seen,” acting Centers for Disease Control and Prevention director Anne Schuchat, MD, said. For the current season so far, the hospitalization rate stands at 51.4 per 100,000 population, putting it on pace to top the total of 710,000 flu-related admissions that occurred during the 2014-2015 season, she said in a weekly briefing Feb. 2.

Flu-related pediatric deaths also took a big jump for the week as another 16 were reported, which brings the total for the season to 53. Of the children who have died so far, only 20% were vaccinated, said Dan Jernigan, MD, MPH, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta. He also noted that half of the children who have been hospitalized did not had an underlying condition.
The one bit of good news for the week was that activity in the West seems to be easing up, Dr. Schuchat said. The geographic spread of ILI was reported as widespread in 48 states, which is down from 49 the previous week because Oregon dropped off the list. To go along with that, the ILI activity level in California has dropped 2 weeks in a row and now stands at level 7, the CDC data show.
Through the last full week of January, the cumulative “hospitalization rate is the highest we’ve seen,” acting Centers for Disease Control and Prevention director Anne Schuchat, MD, said. For the current season so far, the hospitalization rate stands at 51.4 per 100,000 population, putting it on pace to top the total of 710,000 flu-related admissions that occurred during the 2014-2015 season, she said in a weekly briefing Feb. 2.

Flu-related pediatric deaths also took a big jump for the week as another 16 were reported, which brings the total for the season to 53. Of the children who have died so far, only 20% were vaccinated, said Dan Jernigan, MD, MPH, director of the influenza division at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta. He also noted that half of the children who have been hospitalized did not had an underlying condition.
The one bit of good news for the week was that activity in the West seems to be easing up, Dr. Schuchat said. The geographic spread of ILI was reported as widespread in 48 states, which is down from 49 the previous week because Oregon dropped off the list. To go along with that, the ILI activity level in California has dropped 2 weeks in a row and now stands at level 7, the CDC data show.
Preoperative exercise lowers postoperative lung resection complications
with a systematic review suggesting it reduces postoperative complications and duration of hospital stay.
The review and meta-analysis, published in the February British Journal of Sports Medicine, looked at the impact of preoperative exercise in patients undergoing surgery for a range of cancers.
Their review of 13 interventional trials, involving 806 patients and six tumor types, found the postoperative benefits of exercise were evident only in patients undergoing lung resection.
Data from five randomized controlled trials and one quasirandomized trial in lung cancer patients showed a significant 48% reduction in postoperative complications, and a significant mean reduction of 2.86 days in hospital stay among patients undergoing lung resection, compared with controls.
“Postoperative complication is a major concern for patients undergoing oncological surgery,” wrote Dr. Daniel Steffens, from the Surgical Outcomes Research Centre at the Royal Prince Alfred Hospital, Sydney, and his coauthors. They suggested the benefits for patients undergoing lung resection were significant enough that exercise before surgery should be considered as standard preoperative care.
“Such findings may also [have impacts] on health care costs and on patients’ quality of life, and consequently, have important implications for patients, health care professionals and policy makers.”
The exercise regimens in the lung cancer studies mostly involved aerobic exercise, such as walking, and breathing exercises to train respiratory muscles, as well as use of an exercise bicycle. The exercises were undertaken in the 1-2 weeks before surgery, with a frequency ranging from three times a week to three times a day.
The authors noted that trials involving a higher frequency of exercise showed a larger effect size, which suggested there was a dose-response relationship.
There was little evidence of benefit in other tumor types. Two studies examined the benefits of preoperative pelvic floor muscle exercises in men undergoing radical prostatectomy and found significant benefits in quality of life, assessed using the International Continence Society Male Short form. However, the authors pointed out that the quality of evidence was very low.
One study investigated the effects of preoperative mouth-opening exercise training in patients undergoing surgery for oral cancer and found enhanced postoperative quality of life in these patients, but the researchers did not report estimates.
For patients undergoing surgery for colon cancer, colorectal liver metastases, and esophageal cancer, there was no benefit of exercise either in postoperative complications or duration of hospital stay. In all these studies, the authors rated the quality of evidence as “very low.”
“Despite the evidence suggesting that exercise improves physical and mental health in patients with cancer, there are only a limited number of trials investigating the effect of preoperative exercise on patients’ quality of life,” the authors wrote. “Therefore, the effect of preoperative exercise on quality of life at short-term and long-term postoperation should be explored in future trials.”
No conflicts of interest were declared.
SOURCE: Steffens D et al. Br J Sports Med. 2018 Feb 1. doi: 10.1136/bjsports-2017-098032
with a systematic review suggesting it reduces postoperative complications and duration of hospital stay.
The review and meta-analysis, published in the February British Journal of Sports Medicine, looked at the impact of preoperative exercise in patients undergoing surgery for a range of cancers.
Their review of 13 interventional trials, involving 806 patients and six tumor types, found the postoperative benefits of exercise were evident only in patients undergoing lung resection.
Data from five randomized controlled trials and one quasirandomized trial in lung cancer patients showed a significant 48% reduction in postoperative complications, and a significant mean reduction of 2.86 days in hospital stay among patients undergoing lung resection, compared with controls.
“Postoperative complication is a major concern for patients undergoing oncological surgery,” wrote Dr. Daniel Steffens, from the Surgical Outcomes Research Centre at the Royal Prince Alfred Hospital, Sydney, and his coauthors. They suggested the benefits for patients undergoing lung resection were significant enough that exercise before surgery should be considered as standard preoperative care.
“Such findings may also [have impacts] on health care costs and on patients’ quality of life, and consequently, have important implications for patients, health care professionals and policy makers.”
The exercise regimens in the lung cancer studies mostly involved aerobic exercise, such as walking, and breathing exercises to train respiratory muscles, as well as use of an exercise bicycle. The exercises were undertaken in the 1-2 weeks before surgery, with a frequency ranging from three times a week to three times a day.
The authors noted that trials involving a higher frequency of exercise showed a larger effect size, which suggested there was a dose-response relationship.
There was little evidence of benefit in other tumor types. Two studies examined the benefits of preoperative pelvic floor muscle exercises in men undergoing radical prostatectomy and found significant benefits in quality of life, assessed using the International Continence Society Male Short form. However, the authors pointed out that the quality of evidence was very low.
One study investigated the effects of preoperative mouth-opening exercise training in patients undergoing surgery for oral cancer and found enhanced postoperative quality of life in these patients, but the researchers did not report estimates.
For patients undergoing surgery for colon cancer, colorectal liver metastases, and esophageal cancer, there was no benefit of exercise either in postoperative complications or duration of hospital stay. In all these studies, the authors rated the quality of evidence as “very low.”
“Despite the evidence suggesting that exercise improves physical and mental health in patients with cancer, there are only a limited number of trials investigating the effect of preoperative exercise on patients’ quality of life,” the authors wrote. “Therefore, the effect of preoperative exercise on quality of life at short-term and long-term postoperation should be explored in future trials.”
No conflicts of interest were declared.
SOURCE: Steffens D et al. Br J Sports Med. 2018 Feb 1. doi: 10.1136/bjsports-2017-098032
with a systematic review suggesting it reduces postoperative complications and duration of hospital stay.
The review and meta-analysis, published in the February British Journal of Sports Medicine, looked at the impact of preoperative exercise in patients undergoing surgery for a range of cancers.
Their review of 13 interventional trials, involving 806 patients and six tumor types, found the postoperative benefits of exercise were evident only in patients undergoing lung resection.
Data from five randomized controlled trials and one quasirandomized trial in lung cancer patients showed a significant 48% reduction in postoperative complications, and a significant mean reduction of 2.86 days in hospital stay among patients undergoing lung resection, compared with controls.
“Postoperative complication is a major concern for patients undergoing oncological surgery,” wrote Dr. Daniel Steffens, from the Surgical Outcomes Research Centre at the Royal Prince Alfred Hospital, Sydney, and his coauthors. They suggested the benefits for patients undergoing lung resection were significant enough that exercise before surgery should be considered as standard preoperative care.
“Such findings may also [have impacts] on health care costs and on patients’ quality of life, and consequently, have important implications for patients, health care professionals and policy makers.”
The exercise regimens in the lung cancer studies mostly involved aerobic exercise, such as walking, and breathing exercises to train respiratory muscles, as well as use of an exercise bicycle. The exercises were undertaken in the 1-2 weeks before surgery, with a frequency ranging from three times a week to three times a day.
The authors noted that trials involving a higher frequency of exercise showed a larger effect size, which suggested there was a dose-response relationship.
There was little evidence of benefit in other tumor types. Two studies examined the benefits of preoperative pelvic floor muscle exercises in men undergoing radical prostatectomy and found significant benefits in quality of life, assessed using the International Continence Society Male Short form. However, the authors pointed out that the quality of evidence was very low.
One study investigated the effects of preoperative mouth-opening exercise training in patients undergoing surgery for oral cancer and found enhanced postoperative quality of life in these patients, but the researchers did not report estimates.
For patients undergoing surgery for colon cancer, colorectal liver metastases, and esophageal cancer, there was no benefit of exercise either in postoperative complications or duration of hospital stay. In all these studies, the authors rated the quality of evidence as “very low.”
“Despite the evidence suggesting that exercise improves physical and mental health in patients with cancer, there are only a limited number of trials investigating the effect of preoperative exercise on patients’ quality of life,” the authors wrote. “Therefore, the effect of preoperative exercise on quality of life at short-term and long-term postoperation should be explored in future trials.”
No conflicts of interest were declared.
SOURCE: Steffens D et al. Br J Sports Med. 2018 Feb 1. doi: 10.1136/bjsports-2017-098032
FROM THE BRITISH JOURNAL OF SPORTS MEDICINE
Key clinical point: Exercising before oncologic surgery appears to lower the risk of postoperative complications and reduce hospital stay for lung cancer patients.
Major finding: Patients who participated in preoperative exercise before lung cancer surgery had a 48% reduction in postoperative complications, compared with controls.
Data source: Systematic review and meta-analysis of 13 interventional trials involving 806 patients.
Disclosures: No conflicts of interest were declared.
Source: Steffens D et al. Br J Sports Med. 2018, Feb 1. doi: 10.1136/bjsports-2017-098032
FDA approves implantable therapy for PAH
to treat adult patients with New York Heart Association (NYHA) Class I, II and III pulmonary arterial hypertension.
This infusion system is implanted into a patient for intravenous delivery of treprostinil (Remodulin) and is designed to help supply blood to the lungs and keep a patient’s blood pressure within a healthy range. The system comprises three parts: the pump, the programmer, and the catheter.
The implant should not be used for patients with NYHA Class IV heart failure, a known or suspected infection, bacteremia, or sepsis requiring antibiotics; vasculature that is inadequate for an 8 French introducer or catheter advancement without stylet guidance; implanted leads or catheters (active or abandoned) in the superior vena cava that cannot be removed prior to or at system implant; a body size not sufficient to accept the pump; or skin or soft tissue that would heal poorly or increase susceptibility to infections. Patients who are unable to tolerate a sudden cessation of treprostinil therapy also would not be able to receive the implantable device.
Read the full approval on the FDA’s website.
to treat adult patients with New York Heart Association (NYHA) Class I, II and III pulmonary arterial hypertension.
This infusion system is implanted into a patient for intravenous delivery of treprostinil (Remodulin) and is designed to help supply blood to the lungs and keep a patient’s blood pressure within a healthy range. The system comprises three parts: the pump, the programmer, and the catheter.
The implant should not be used for patients with NYHA Class IV heart failure, a known or suspected infection, bacteremia, or sepsis requiring antibiotics; vasculature that is inadequate for an 8 French introducer or catheter advancement without stylet guidance; implanted leads or catheters (active or abandoned) in the superior vena cava that cannot be removed prior to or at system implant; a body size not sufficient to accept the pump; or skin or soft tissue that would heal poorly or increase susceptibility to infections. Patients who are unable to tolerate a sudden cessation of treprostinil therapy also would not be able to receive the implantable device.
Read the full approval on the FDA’s website.
to treat adult patients with New York Heart Association (NYHA) Class I, II and III pulmonary arterial hypertension.
This infusion system is implanted into a patient for intravenous delivery of treprostinil (Remodulin) and is designed to help supply blood to the lungs and keep a patient’s blood pressure within a healthy range. The system comprises three parts: the pump, the programmer, and the catheter.
The implant should not be used for patients with NYHA Class IV heart failure, a known or suspected infection, bacteremia, or sepsis requiring antibiotics; vasculature that is inadequate for an 8 French introducer or catheter advancement without stylet guidance; implanted leads or catheters (active or abandoned) in the superior vena cava that cannot be removed prior to or at system implant; a body size not sufficient to accept the pump; or skin or soft tissue that would heal poorly or increase susceptibility to infections. Patients who are unable to tolerate a sudden cessation of treprostinil therapy also would not be able to receive the implantable device.
Read the full approval on the FDA’s website.
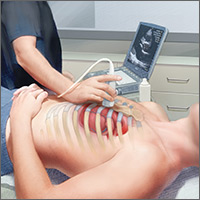
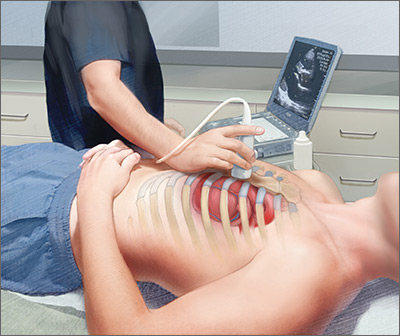
Point-of-care ultrasound: Coming soon to primary care?
Point-of-care ultrasound (POCUS) has been gaining greater traction in recent years as a way to quickly (and cost-effectively) assess for conditions including systolic dysfunction, pleural effusion, abdominal aortic aneurysms (AAAs), and deep vein thrombosis (DVT). It involves limited and specific ultrasound protocols performed at the bedside by the health care provider who is trying to answer a specific question and, thus, help guide treatment of the patient.
POCUS was first widely used by emergency physicians starting in the early 1990s with the widespread adoption of the Focused Assessment with Sonography in Trauma (FAST) scan.1,2 Since that time, POCUS has expanded beyond trauma applications and into family medicine.
One study assessed physicians’ perceptions of POCUS after its integration into a military family medicine clinic. The study showed that physicians perceived POCUS to be relatively easy to use, not overly time consuming, and of high value to the practice.3 In fact, the literature tells us that POCUS can help decrease the cost of health care and improve outcomes,4-7 while requiring a relatively brief training period.
If residencies are any indication, POCUS may be headed your way
Ultrasound units are becoming smaller and more affordable, and medical schools are increasingly incorporating ultrasound curricula into medical student training.8 As of 2016, only 6% of practicing FPs reported using non-obstetric POCUS in their practices.9 Similarly, a survey from 2015 reported that only 2% of family medicine residency programs had established POCUS curricula.10 However, 50% of respondents in the 2015 survey reported early-stage development or interest in developing a POCUS curriculum.
Since then a validated family medicine residency curriculum has been published,11 and the American Academy of Family Physicians (AAFP) recently released a POCUS Curriculum Guideline for residencies (https://www.aafp.org/dam/AAFP/documents/medical_education_residency/program_directors/Reprint290D_POCUS.pdf).
[polldaddy:9928416]
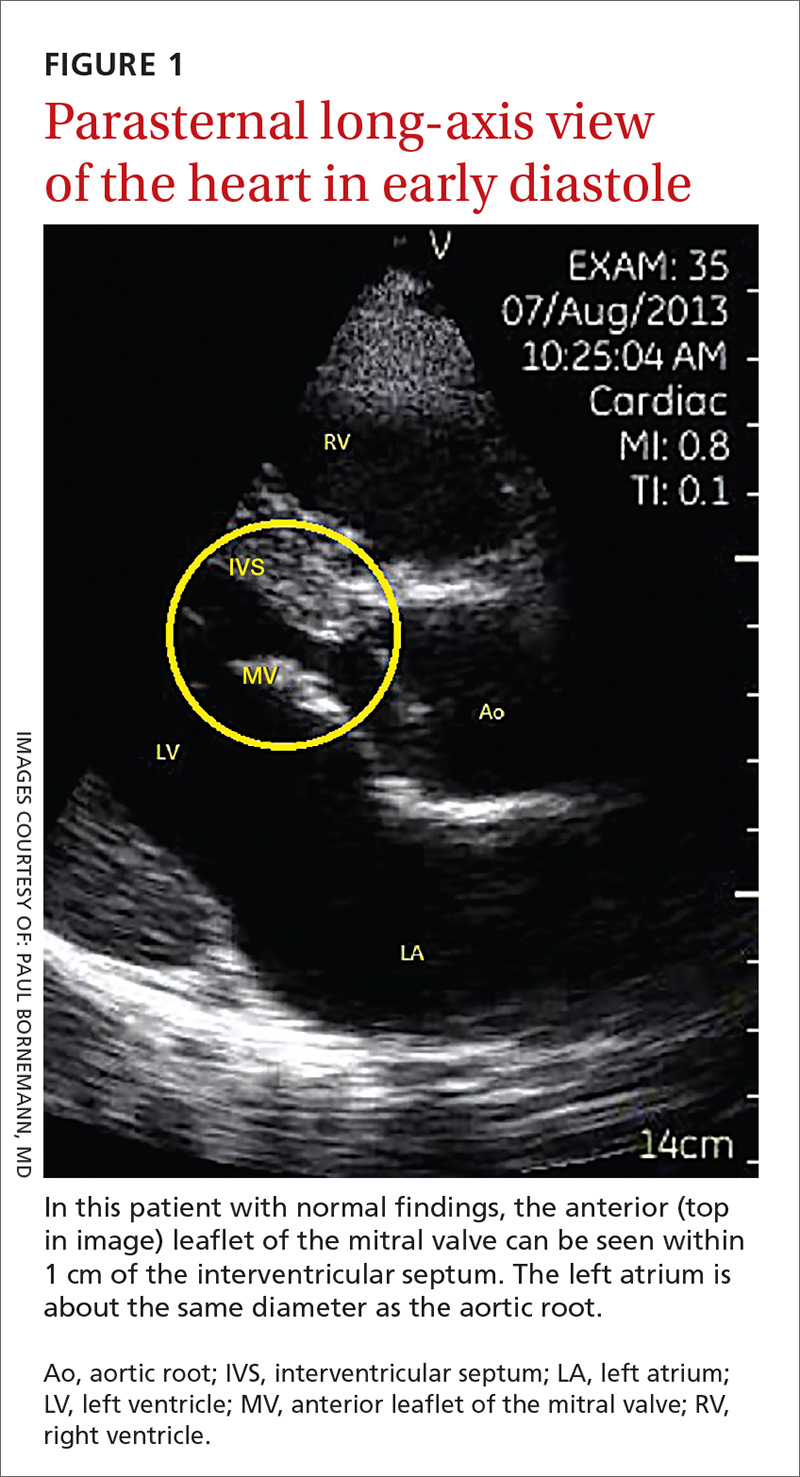
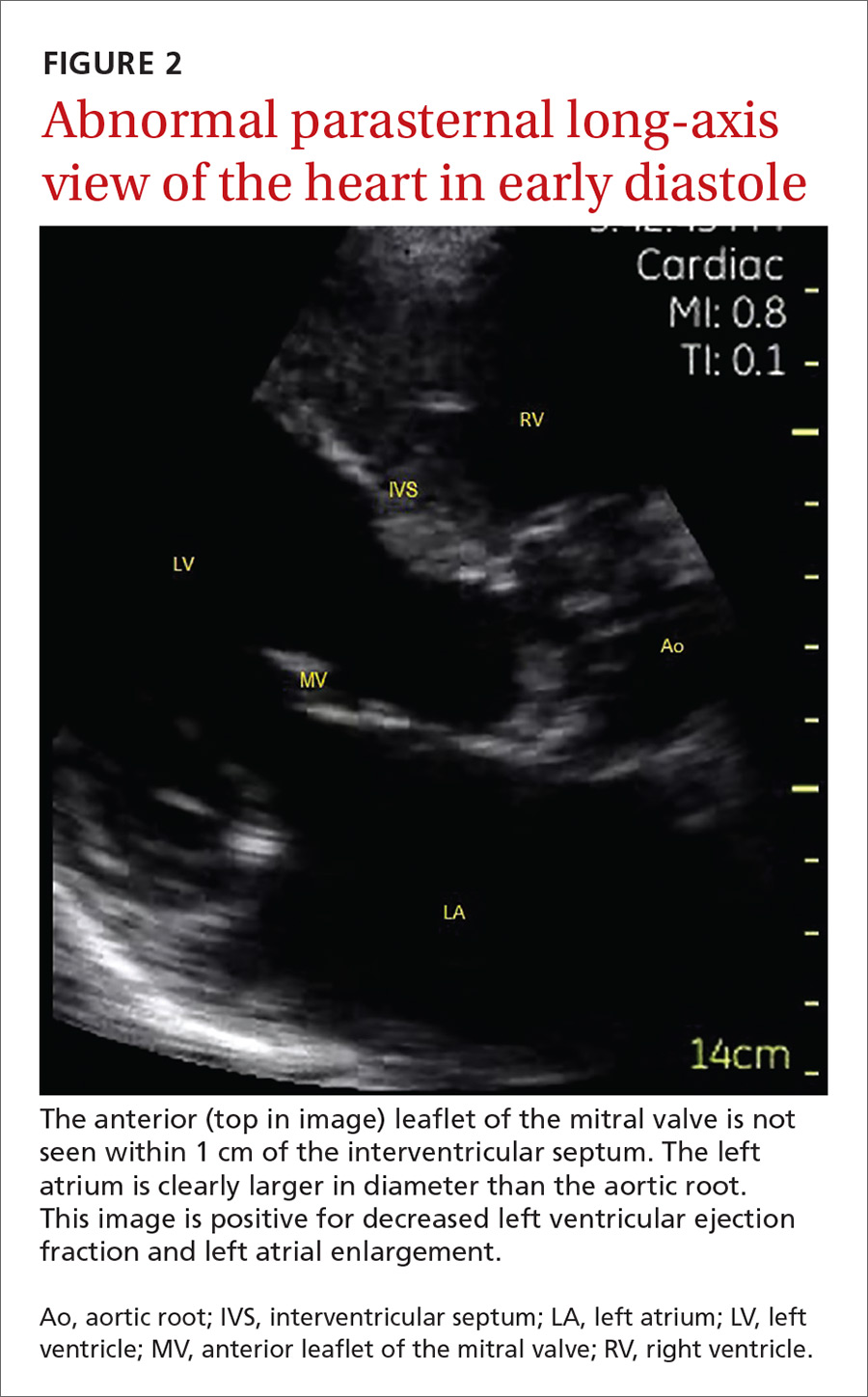
The potential applications of POCUS in family medicine are numerous and have been reviewed in several recent publications.12,13 In this article, we will review the evidence for the use of POCUS in 4 areas: the cardiovascular exam (FIGURES 1 and 2), the lung exam (FIGURES 3-6), the screening exam for AAAs (FIGURE 7), and the evaluation for DVT (FIGURES 8 and 9). (Obstetric and musculoskeletal applications have been sufficiently covered elsewhere.14-17) For all of these applications, POCUS is safe, accurate, and beneficial and can be performed with a relatively small amount of training by non-radiology specialists, including FPs (TABLEs 1 and 2).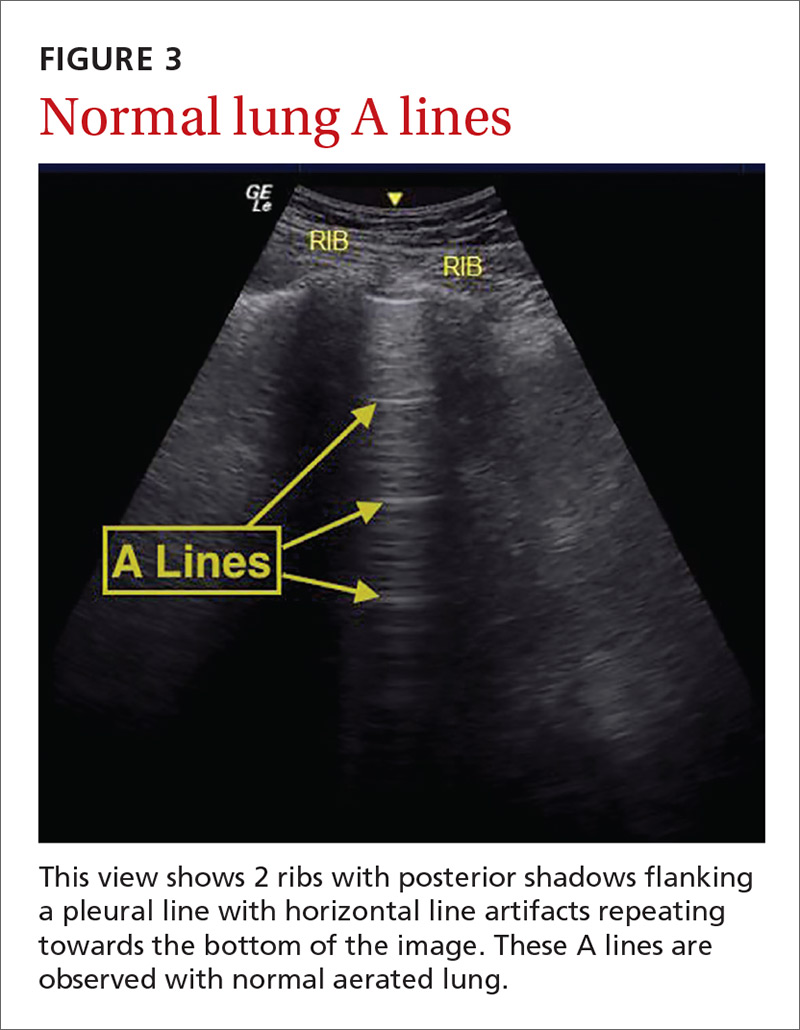
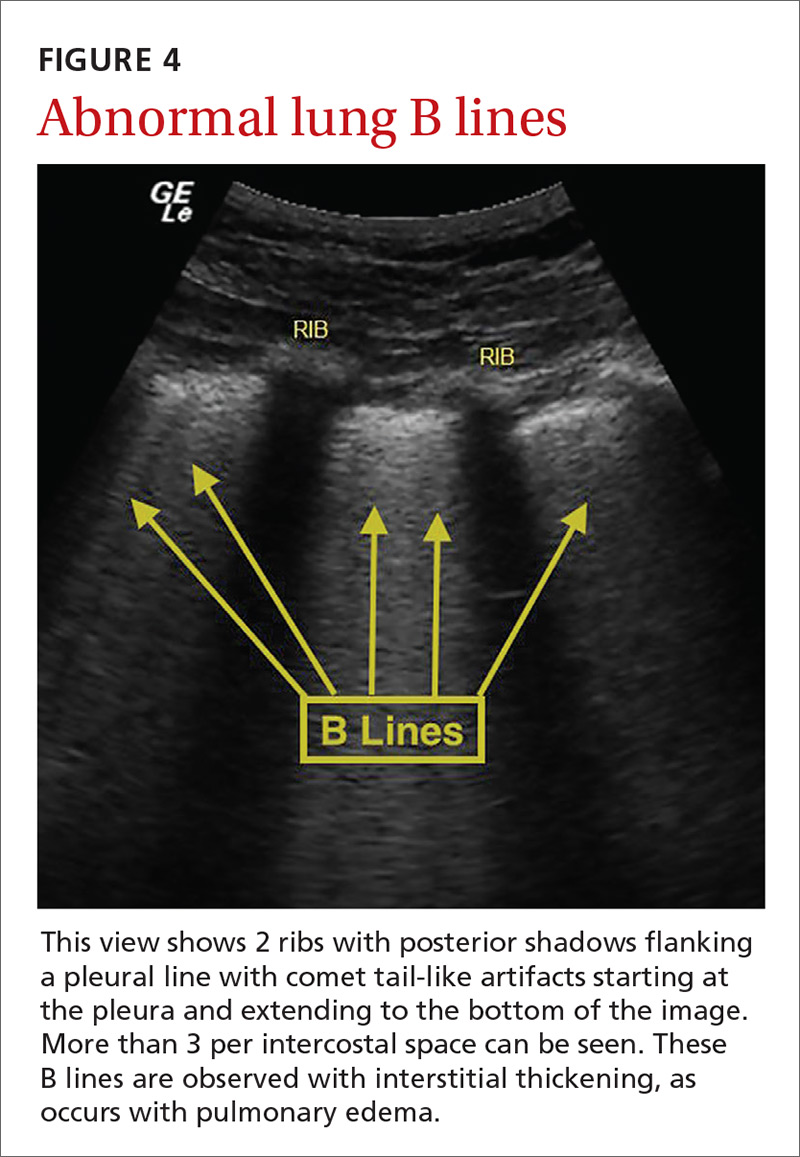
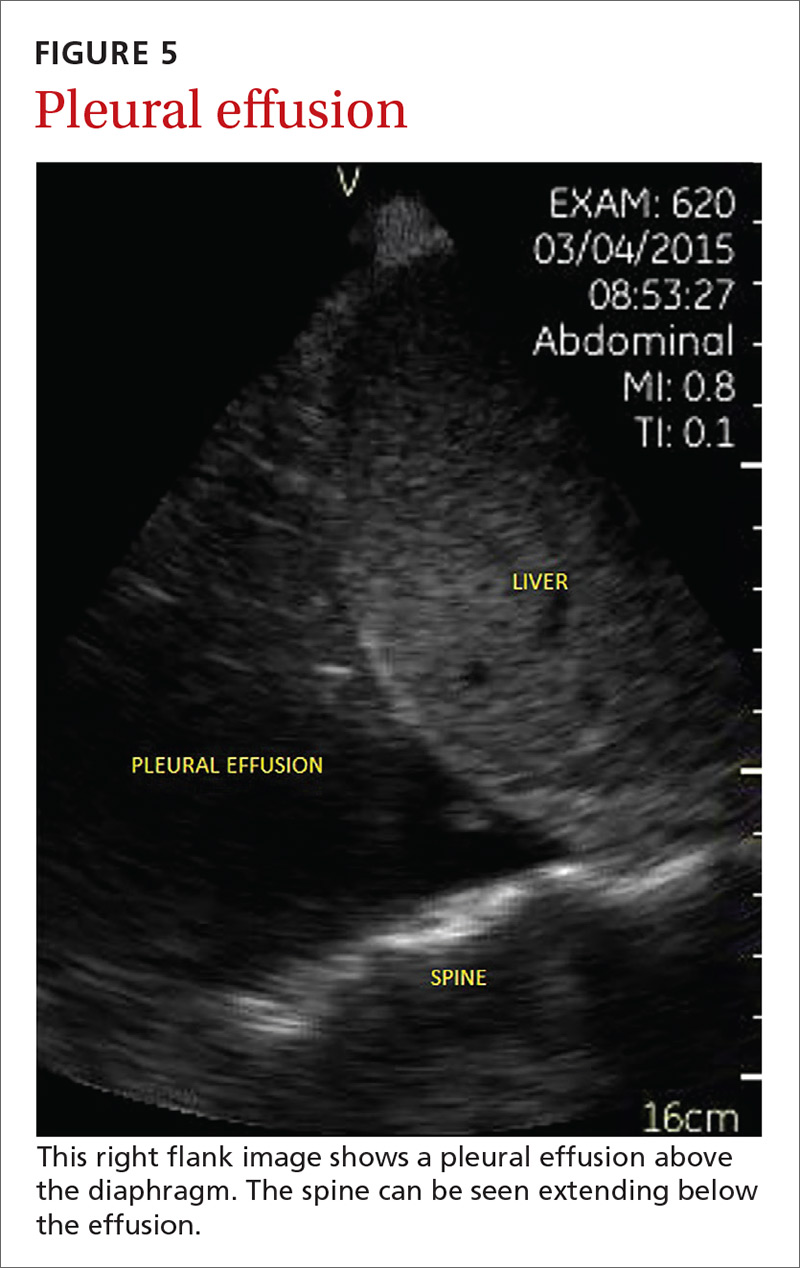
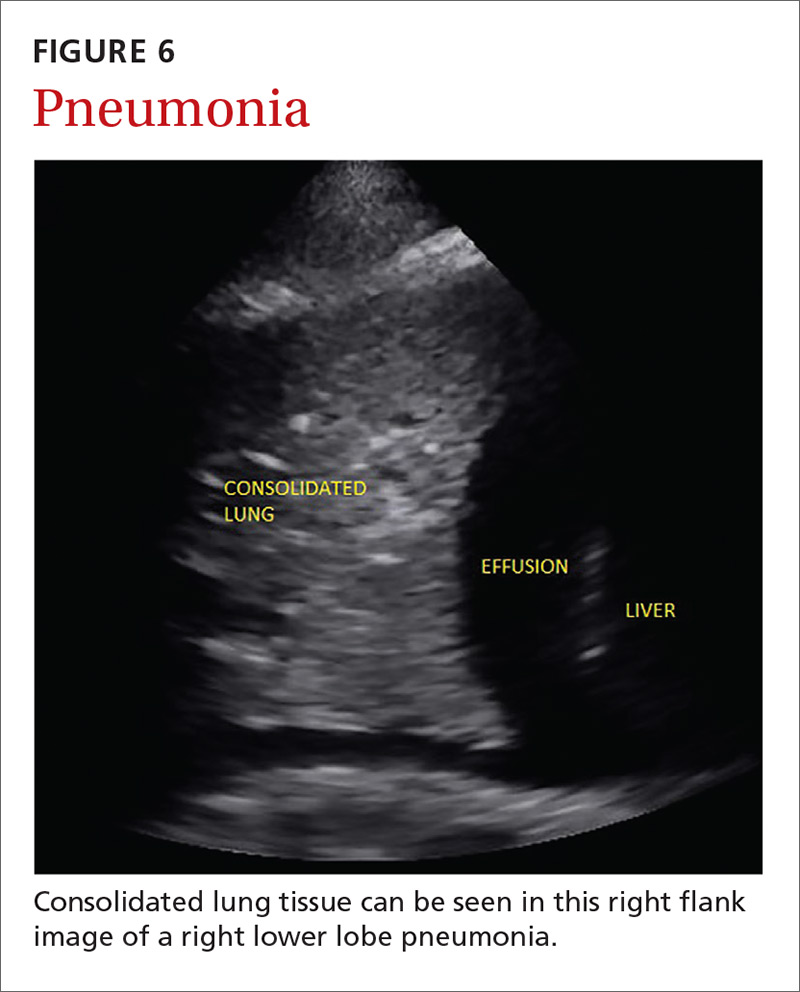
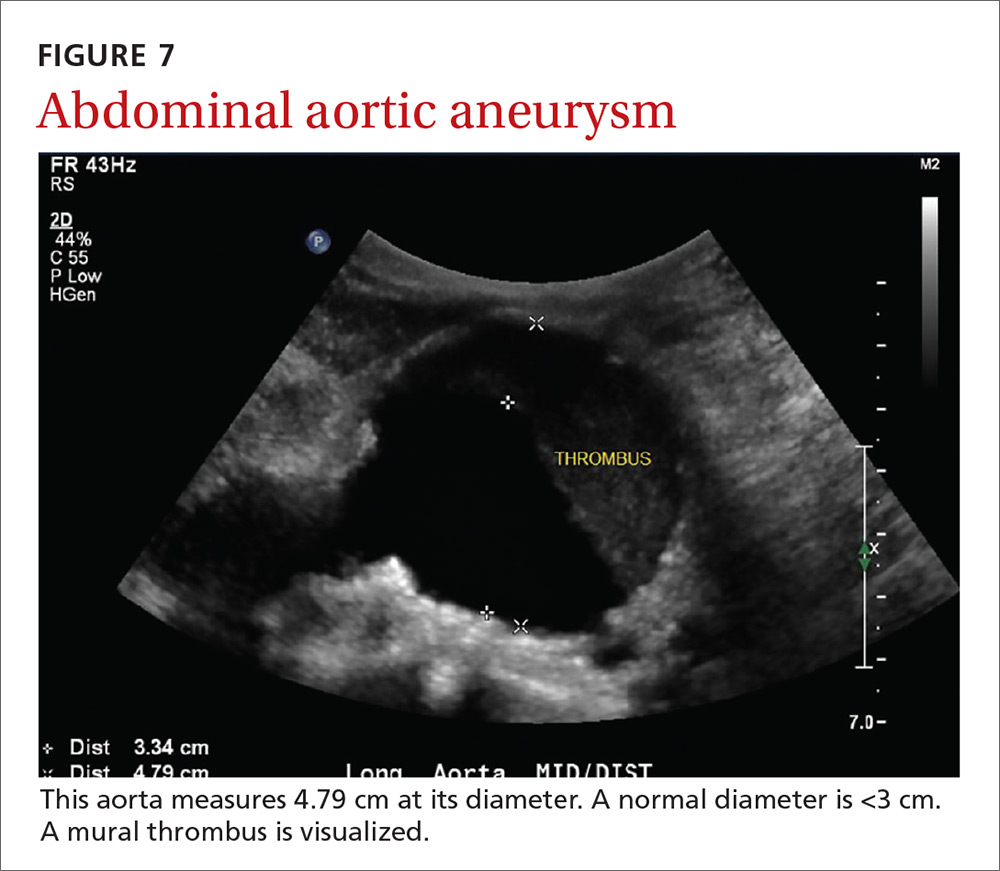
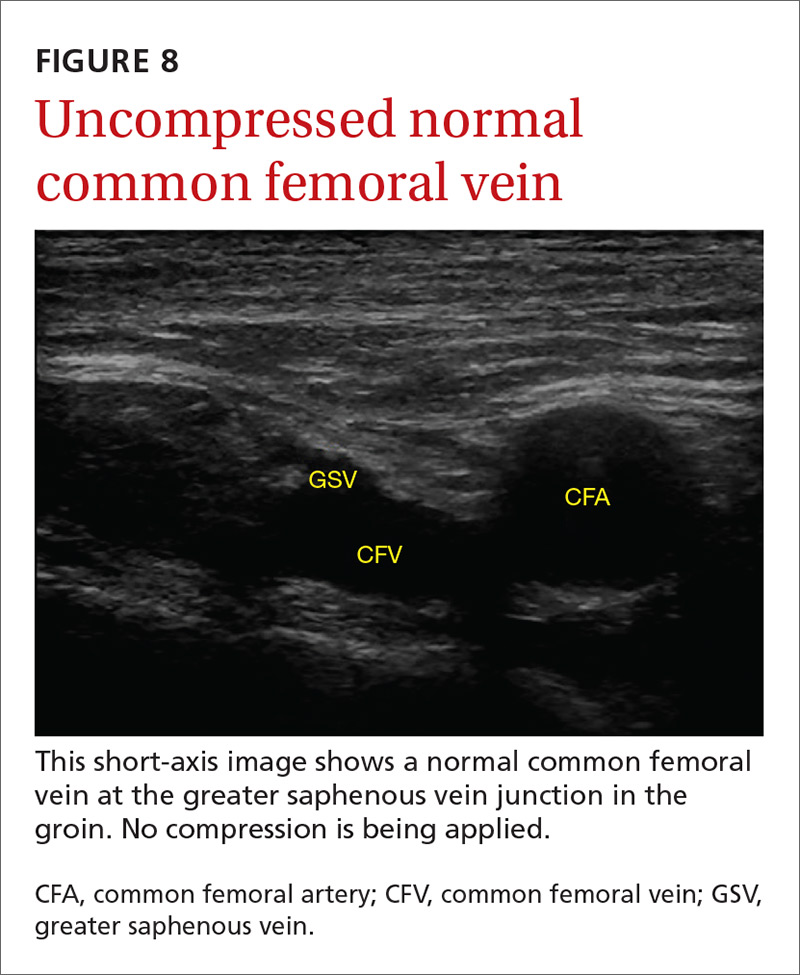
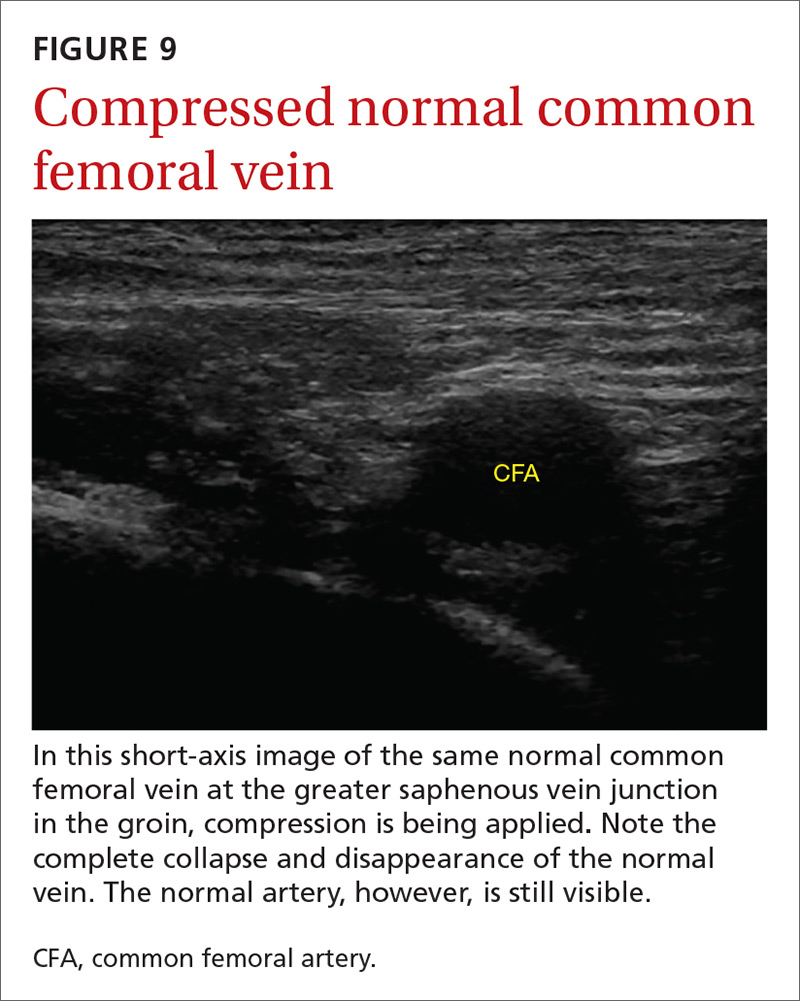
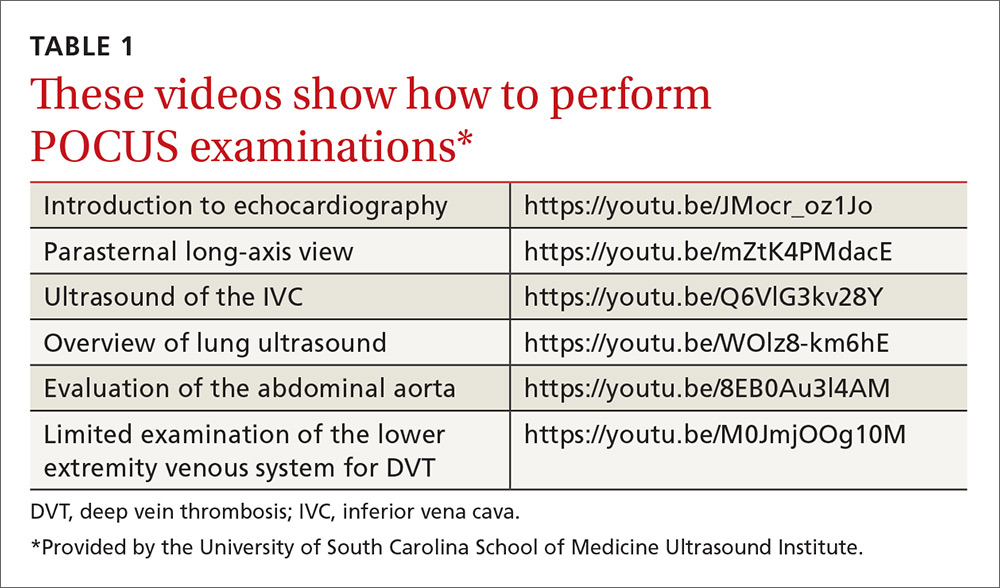
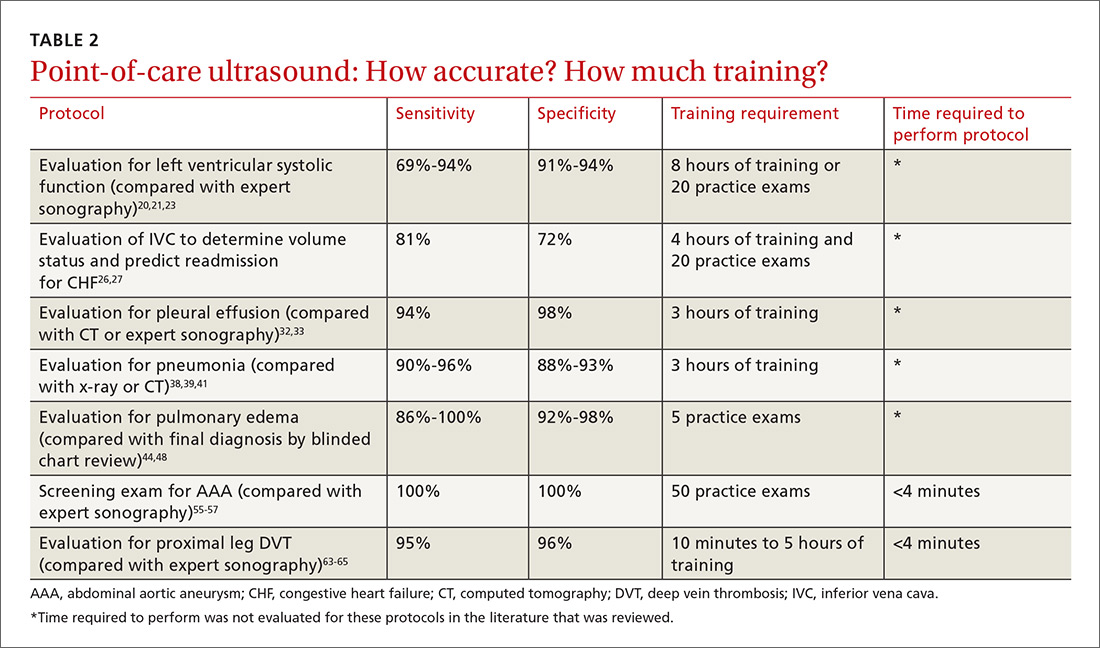
Just 2 hours of cardio POCUS training enhanced Dx accuracy
The American Society of Echocardiography (ASE) issued an expert consensus statement for focused cardiac ultrasound in 2013.18 The guideline supports non-cardiologists utilizing POCUS to assess for pericardial effusion and right and left ventricular enlargement, as well as to review global cardiac systolic function and intravascular volume status. Cardiovascular POCUS protocols are relatively easy to learn; even small amounts of training and practice can yield competency.
For example, a 2013 study showed that after 2 hours of training with a pocket ultrasound device, medical students and junior physicians inexperienced with POCUS were able to improve their diagnostic accuracy for heart failure from 50% to 75%.19 In another study, internal medicine residents with limited cardiac ultrasound training (ie, 20 practice exams) were able to detect decreased left ventricular ejection fraction using a handheld ultrasound device with 94% sensitivity and specificity in patients admitted to the hospital with acute decompensated heart failure.20 Similarly, after only 8 hours of training, a group of Norwegian general practitioners were able to obtain measurements of systolic function with a pocket ultrasound device that were not statistically different from a cardiologist’s measurements.21
In another study, rural FPs attended a 4-day course and then performed focused cardiac ultrasounds on primary care patients with a clinical indication for an echocardiogram.22 The scans were uploaded to a Web-based program for remote interpretation by a cardiologist. There was high concordance between the FPs’ interpretations of the focused cardiac ultrasounds and the cardiologist’s interpretations. Only 32% of the patients in the study group required a formal follow-up echocardiogram.
Kimura et al published a POCUS protocol for the rapid assessment of patients with heart failure, called the Cardiopulmonary Limited Ultrasound Exam (CLUE).23 The CLUE protocol utilizes 4 views to assess left ventricular systolic and diastolic function along with signs of pulmonary edema or systemic volume overload (TABLE 323). The presence of pulmonary edema or a plethoric inferior vena cava (IVC) was highly prognostic of in-hospital mortality. The CLUE protocol has been successfully used by novices including internal medicine residents after brief training (ie, up to 60 supervised scans) and can be performed in less than 5 minutes.24,25 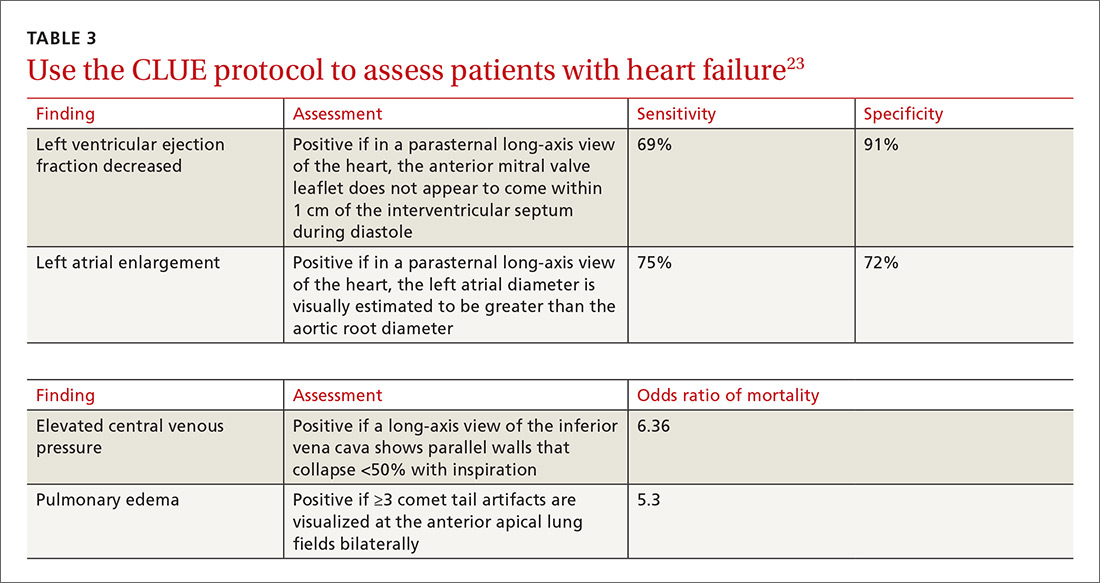
Inpatient use. In addition to its use as an outpatient diagnostic tool, POCUS may be able to help guide therapy in patients admitted to the hospital with heart failure. Increasing collapse of the IVC directly correlates with the amount of fluid volume removed during hemodialysis.26 Goonewardena et al showed that IVC collapsibility was an independent predictor of 30-day hospital readmission even when demographics, signs and symptoms, and volume of diuresis were otherwise equal.27 However, whether the use of IVC collapsibility to guide management improves outcomes in heart failure remains to be validated in a prospective trial.
More sensitive, specific than x-rays for pulmonary diagnoses
The chest x-ray has traditionally been the imaging modality of choice to evaluate primary care pulmonary complaints. However, POCUS can be more sensitive and specific than a chest x-ray for evaluating several pulmonary diagnoses including pleural effusion, pneumonia, and pulmonary edema.
Pleural effusion can be difficult to detect with a physical exam alone. A systematic review showed that the physical exam is not sensitive for effusions <300 mL and can have even lower utility in obese patients.28 While an upright lateral chest x-ray can accurately detect effusions as small as 50 mL, portable x-rays have sensitivities of only 53% to 71% for small- or moderate-sized effusions.29,30 Ultrasound, however, has a sensitivity of 97% for small effusions.31
A 2016 meta-analysis showed that POCUS had a pooled sensitivity and specificity of 94% and 98%, respectively, for pleural effusions, while chest x-ray had a pooled sensitivity and specificity of 51% and 91%, respectively, when compared with computed tomography (CT) and expert sonography.32 POCUS evaluation for pleural effusion is technically simple, and at least one study showed that even novice users can achieve high diagnostic accuracy after only 3 hours of training.33
Pneumonia is the eighth leading cause of death in the United States and the single leading cause of infectious disease death in children worldwide.34-36 Pneumonia is a difficult diagnosis to make based on a history and physical examination alone, and the Infectious Diseases Society of America recommends diagnostic imaging to make the diagnosis.37
The adult and pediatric literature clearly demonstrate that lung ultrasound is accurate at diagnosing pneumonia. In a 2015 meta-analysis of the pediatric literature, lung ultrasound had a sensitivity of 96% and a specificity of 93% and positive and negative likelihood ratios of 15.3 and 0.06, respectively.38 In adults, a 2016 meta-analysis of lung ultrasound showed a pooled sensitivity and specificity of 90% and 88%, respectively, with positive and negative likelihood ratios of 6.6 and 0.08, respectively.39
In 2015, a prospective study compared the accuracy of lung ultrasound and chest x-ray using CT as the gold standard.40 Lung ultrasound had a significantly better sensitivity of 82% compared to a sensitivity of 64% for chest x-ray. Specificities were comparable at 94% for ultrasound and 90% for chest x-ray.40
At least one study found novice sonographers to be accurate with lung POCUS for the diagnosis of pneumonia after only two 90-minute training sessions.41 Moreover, ultrasound has a more favorable safety profile, greater portability, and lower cost compared with chest x-ray and CT.
Pulmonary edema. Lung ultrasound can identify interstitial pulmonary edema via artifacts called B lines, which are produced by the reverberation of sound waves from the pleura due to the widening of the fluid-filled interlobular septa. These are distinctly different from the A-line pattern of repeating horizontal lines that is seen with normal lungs, making lung ultrasound more accurate than chest x-ray for identification of pulmonary edema.42,43 When final diagnosis via blinded chart review is used as the reference standard, bilateral B lines on a lung ultrasound image have a sensitivity of 86% to 100% and a specificity of 92% to 98% for the diagnosis of pulmonary edema compared to chest x-ray’s sensitivity of 56.9% and specificity of 89.2%.44 There is also a linear correlation between the number of B lines present and the extent of pulmonary edema.42,45,46 The number of B lines decreases in real time as volume is removed in dialysis patients.47
POCUS evaluation for B lines can be learned very quickly. Exams of novices who have performed only 5 prior exams correlate highly with those of experts who have performed more than 100 exams.48
Simple, efficient screening method for abdominal aortic aneurysm
AAAs are present in up to 7% of men over the age of 50.49 The mortality rate of a ruptured AAA is as high as 80% to 95%.50 There is, however, a long prodromal period when interventions can make a significant difference, which is why accurate screening is so important.
AAA screening with ultrasound has been shown to decrease mortality.51 The current recommendation of the US Preventive Services Task Force (USPSTF) is a one-time AAA screening for all men ages 65 to 75 years who have ever smoked (Grade B).52 Despite the recommendations of the USPSTF, screening rates are low. One study found that only 9% of eligible patients in primary care practices received appropriate screening.51
Ultrasound performed by specialists is known to be an excellent screening test for AAA with a sensitivity of 98.9% and a specificity of 99.9%.53 POCUS use by emergency medicine physicians for the evaluation of symptomatic AAA is well established in the literature. A meta-analysis including 7 studies and 655 patients showed a pooled sensitivity of 99% and a specificity of 98%.54 Multiple studies also support primary care physicians performing POCUS AAA screening in the clinic setting.
For example, a 2012 prospective, observational study performed in Canada compared office-based ultrasound screening exams performed by a rural FP to scans performed in the hospital on the same patients.55 The physician completed 50 training examinations. The average discrepancy in aorta diameters between the 2 was only 2 mm, which is clinically insignificant, and the office-based scans had a sensitivity and specificity of 100%.
Similarly, a second FP study performed in Barcelona, showed that an FP who performed POCUS AAA screening had 100% concordance with a radiologist.56 Additionally, POCUS screening for AAA was not time consuming; it was performed in under 4 minutes per patient.55,57
Ruling out DVT
DVT is a relatively rare occurrence in the ambulatory setting. However, patients who present with a painful, swollen lower extremity are much more common, and DVT must be considered and ruled out in these situations.
Although isolated distal DVTs that occur in the calf veins are usually self-limited and have a very low risk of embolization, they can progress to proximal DVTs of the thigh veins up to 20% of time.58,59 Similarly, thrombophlebitis of the superficial lower extremity veins rarely embolizes, but can progress to a proximal DVT, especially if large segments are involved or if the segments are within 5 cm of the junction to the deep venous system.59 The risk of missing a proximal leg DVT is high because embolization occurs up to 60% of the time if the DVT is left untreated.60
The current standard for diagnosis of DVT is the lower extremity Doppler ultrasound examination, but obtaining same-day Doppler evaluations can be difficult in the ambulatory setting. In these instances, the American College of Chest Physicians (ACCP) recommends that even low-risk patients receive anticoagulation pending the evaluation if it cannot be obtained in the first 24 hours.59 This approach not only increases the cost of care, but also exposes patients—many of whom will not be diagnosed with thrombosis in the end—to the risks of anticoagulation.
D-dimer blood tests have drawbacks, too. While a negative high-sensitivity D-dimer blood test in a patient with a low pre-test probability of DVT can effectively rule out a DVT, laboratory testing is not always immediately available in the ambulatory setting either.61 Additionally, false-positive rates are high, and positive D-dimer exams still require evaluation by Doppler ultrasound.
Given these limitations, performing an ultrasound at the bedside or in the exam room can allow for more timely and cost-effective care. In fact, research shows that a limited ultrasound, called the 2-region compression exam, which follows along the course of the common femoral vein and popliteal vein only, ignoring the femoral and calf veins, is highly accurate in assessing for proximal leg DVTs. As such, it has been adopted for POCUS use by emergency medicine physicians.62
Multiple studies show that physicians with minimal training can perform the 2-region compression exam with a high degree of accuracy when full-leg Doppler ultrasound was used as the gold standard.63,64 In these studies, hands-on training times ranged from only 10 minutes to 5 hours, and the exam could be performed in less than 4 minutes. A systematic review of 6 studies comparing emergency physician-performed ultrasound with radiology-performed ultrasound calculated an overall sensitivity of 0.95 (95% CI, 0.87-0.99) and specificity of 0.96 (95% CI, 0.87-0.99) for those performed by emergency physicians.65
The main concern with the 2-region compression exam is that it can miss a distal leg DVT. As stated earlier, distal DVTs are relatively benign and tend to resolve without treatment; however, up to 20% can progress to become a dangerous proximal leg DVT.58 Researchers have validated several methods by prospective trials to address this limitation.
Specifically, researchers have demonstrated that patients with a low pre-test probability of DVT per the Wells scoring system could have DVT effectively ruled out with a single 2-region compression ultrasound without further evaluation.66 In another study, researchers evaluated all patients (regardless of pretest probability) with a 2-point compression exam and found that those with negative exams could be followed with a second exam in 7 to 10 days without initiating anticoagulation. If the second one was negative, no further evaluation was needed.67,68
And finally, researchers demonstrated that a negative 2-point compression ultrasound in combination with a concurrent negative D-dimer test was effective at ruling out DVT, regardless of pre-test probability.69,70
A preferred approach
Given this data and the fact that in the ambulatory setting it is often easier and faster to perform a 2-region compression examination than to obtain a D-dimer laboratory test or a formal full-leg Doppler ultrasound, what follows is our preferred approach to a patient with suspected DVT in the outpatient setting (FIGURE 10).
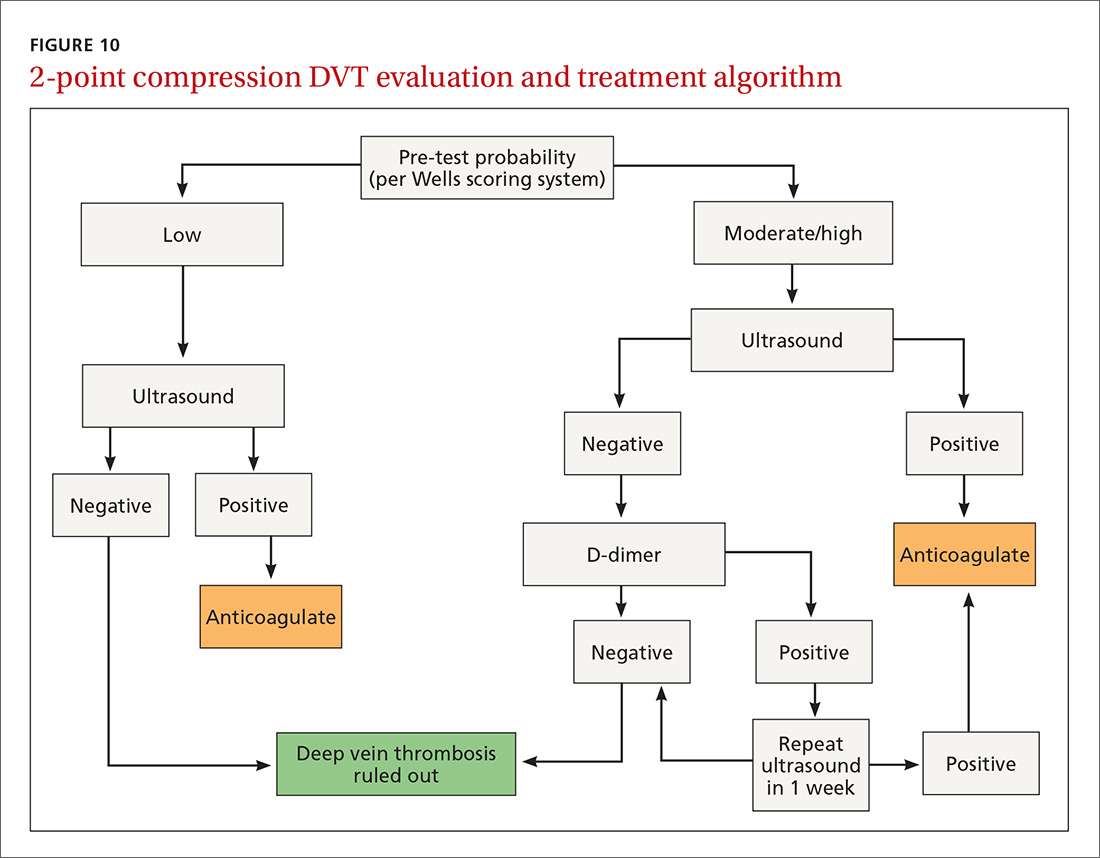
We first assess pre-test probability using the Wells scoring system. We then perform the 2-region compression ultrasound. If the patient has low pre-test risk according to the Wells score, we rule out DVT. If the patient has moderate or high risk with a negative 2-region compression ultrasound, the patient gets a D-dimer test. If the D-dimer test is negative, we rule out DVT. If the D-dimer test is positive, we schedule the patient for a repeat 2-region compression ultrasound in 7 to 10 days. If at any time the 2-region compression evaluation is positive, we treat the patient for DVT.
CORRESPONDENCE
Paul Bornemann, MD, Palmetto Health Family Medicine Residency, Department of Family and Preventive Medicine, University of South Carolina School of Medicine, 3209 Colonial Drive, Columbia, SC 29203; [email protected].
1. Hahn RG, Davies TC, Rodney WM. Diagnostic ultrasound in general practice. Fam Pract. 1988;5:129-135.
2. Deutchman ME, Hahn RG, Rodney WMM. Diagnostic ultrasound imaging by physicians of first contact: extending the family medicine experience into emergency medicine. Ann Emerg Med. 1993;22:594-596.
3. Bornemann P, Bornemann G. Military family physicians’ perceptions of a pocket point-of-care ultrasound device in clinical practice. Mil Med. 2014;179:1474-1477.
4. Smith-Bindman R, Aubin C, Bailitz J, et al. Ultrasonography versus computed tomography for suspected nephrolithiasis. N Engl J Med. 2014;371:1100-1110.
5. Parker L, Nazarian LN, Carrino JA, et al. Musculoskeletal imaging: medicare use, costs, and potential for cost substitution. J Am Coll Radiol. 2008;5:182-188.
6. Gordon CE, Feller-Kopman D, Balk EM, et al. Pneumothorax following thoracentesis: a systematic review and meta-analysis. Arch Intern Med. 2010;170:332-339.
7. Calvert N, Hind D, McWilliams RG, et al. The effectiveness and cost-effectiveness of ultrasound locating devices for central venous access: a systematic review and economic evaluation. Health Technol Assess. 2003;7:1-84.
8. Hoppmann RA, Rao VV, Bell F, et al. The evolution of an integrated ultrasound curriculum (iUSC) for medical students: 9-year experience. Crit Ultrasound J. 2015;7:18.
9. Clinical procedures performed by physicians at their practice. American Academy of Family Physicians Member Census, December 31, 2016. Available at: http://www.aafp.org/about/the-aafp/family-medicine-facts/table-12(rev).html. Accessed June 26, 2017.
10. Hall JW, Holman H, Bornemann P, et al. Point of care ultrasound in family medicine residency programs: a CERA study. Fam Med. 2015;47:706-711.
11. Bornemann P. Assessment of a novel point-of-care ultrasound curriculum’s effect on competency measures in family medicine graduate medical education. J Ultrasound Med. 2017;36:1205-1211.
12. Steinmetz P, Oleskevich S. The benefits of doing ultrasound exams in your office. J Fam Pract. 2016;65:517-523.
13. Flick D. Bedside ultrasound education in family medicine. J Ultrasound Med. 2016;35:1369-1371.
14. Dresang LT, Rodney WM, Rodney KM. Prenatal ultrasound: a tale of two cities. J Natl Med Assoc. 2006;98:167-171.
15. Dresang LT, Rodney WM, Dees J. Teaching prenatal ultrasound to family medicine residents. Fam Med. 2004;36:98-107.
16. Rodney WM, Deutchman ME, Hartman KJ, et al. Obstetric ultrasound by family physicians. J Fam Pract. 1992;34:186-194.
17. Broadhurst NA, Simmons N. Musculoskeletal ultrasound - used to best advantage. Aust Fam Physician. 2007;36:430-432.
18. Spencer KT, Kimura BJ, Korcarz CE, et al. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:567-581.
19. Panoulas VF, Daigeler AL, Malaweera AS, et al. Pocket-size hand-held cardiac ultrasound as an adjunct to clinical examination in the hands of medical students and junior doctors. Eur Heart J Cardiovasc Imaging. 2013;14:323-330.
20. Razi R, Estrada JR, Doll J, et al. Bedside hand-carried ultrasound by internal medicine residents versus traditional clinical assessment for the identification of systolic dysfunction in patients admitted with decompensated heart failure. J Am Soc Echocardiogr. 2011;24:1319-1324.
21. Mjølstad OC, Snare SR, Folkvord L, et al. Assessment of left ventricular function by GPs using pocket-sized ultrasound. Fam Pract. 2012;29:534-540.
22. Evangelista A, Galuppo V, Méndez J, et al. Hand-held cardiac ultrasound screening performed by family doctors with remote expert support interpretation. Heart. 2016;102:376-382.
23. Kimura BJ, Yogo N, O’Connell CW, et al. Cardiopulmonary limited ultrasound examination for “quick-look” bedside application. Am J Cardiol. 2011;108:586-590.
24. Kimura BJ, Amundson SA, Phan JN, et al. Observations during development of an internal medicine residency training program in cardiovascular limited ultrasound examination. J Hosp Med. 2012;7:537-542.
25. Kimura BJ, Shaw DJ, Amundson SA, et al. Cardiac limited ultrasound examination techniques to augment the bedside cardiac physical examination. J Ultrasound Med. 2015;34:1683-1690.
26. Brennan JM, Ronan A, Goonewardena S, et al. Handcarried ultrasound measurement of the inferior vena cava for assessment of intravascular volume status in the outpatient hemodialysis clinic. Clin J Am Soc Nephrol. 2006;1:749-753.
27. Goonewardena SN, Gemignani A, Ronan A, et al. Comparison of hand-carried ultrasound assessment of the inferior vena cava and N-terminal pro-brain natriuretic peptide for predicting readmission after hospitalization for acute decompensated heart failure. JACC Cardiovasc Imaging. 2008;1:595-601.
28. Wong CL, Holroyd-Leduc J, Straus SE. Does this patient have a pleural effusion? JAMA. 2009;301:309-317.
29. Blackmore CC, Black WC, Dallas RV, et al. Pleural fluid volume estimation: a chest radiograph prediction rule. Acad Radiol. 1996;3:103-109.
30. Kitazono MT, Lau CT, Parada AN, et al. Differentiation of pleural effusions from parenchymal opacities: accuracy of bedside chest radiography. Am J Roentgenol. 2010;194:407-412.
31. Kalokairinou-Motogna M, Maratou K, Paianid I, et al. Application of color Doppler ultrasound in the study of small pleural effusion. Med Ultrason. 2010;12:12-16.
32. Yousefifard M, Baikpour M, Ghelichkhani P, et al. Screening performance characteristic of ultrasonography and radiography in detection of pleural effusion; a meta-analysis. Emerg (Tehran, Iran). 2016;4:1-10.
33. Begot E, Grumann A, Duvoid T, et al. Ultrasonographic identification and semiquantitative assessment of unloculated pleural effusions in critically ill patients by residents after a focused training. Intensive Care Med. 2014;40:1475-1480.
34. World Health Organization. Pneumonia. Fact Sheet No. 331. Available at: http://www.who.int/mediacentre/factsheets/fs331/en/. Accessed June 26, 2017.
35. Gereige RS, Laufer PM. Pneumonia. Pediatr Rev. 2013;34:438-456.
36. National Center for Health Statistics. Leading causes of death. https://www.cdc.gov/nchs/fastats/leading-causes-of-death.htm. Accessed July 2, 2017.
37. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-S72.
38. Pereda MA, Chavez MA, Hooper-Miele CC, et al. Lung ultrasound for the diagnosis of pneumonia in children: a meta-analysis. Pediatrics. 2015;135:714-722.
39. Xia Y, Ying Y, Wang S, et al. Effectiveness of lung ultrasonography for diagnosis of pneumonia in adults: a systematic review and meta-analysis. J Thorac Dis. 2016;8:2822-2831.
40. Nazerian P, Volpicelli G, Vanni S, et al. Accuracy of lung ultrasound for the diagnosis of consolidations when compared to chest computed tomography. Am J Emerg Med. 2015;33:620-625.
41. Filopei J, Siedenburg H, Rattner P, et al. Impact of pocket ultrasound use by internal medicine housestaff in the diagnosis of dyspnea. J Hosp Med. 2014;9:594-597.
42. Lichtenstein D, Mezière G. A lung ultrasound sign allowing bedside distinction between pulmonary edema and COPD: the comet-tail artifact. Intensive Care Med. 1998;24:1331-1334.
43. Gargani L, Volpicelli G. How I do it: lung ultrasound. Cardiovasc Ultrasound. 2014;12:25.
44. Martindale JL, Wakai A, Collins SP, et al. Diagnosing acute heart failure in the emergency department: a systematic review and meta-analysis. Acad Emerg Med. 2016;23:223-242.
45. Volpicelli G, Mussa A, Garofalo G, et al. Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. Am J Emerg Med. 2006;24:689-696.
46. Picano E, Frassi F, Agricola E, et al. Ultrasound lung comets: a clinically useful sign of extravascular lung water. J Am Soc Echocardiogr. 2006;19:356-363.
47. Noble VE, Murray AF, Capp R, et al. Ultrasound assessment for extravascular lung water in patients undergoing hemodialysis: time course for resolution. Chest. 2009;135:1433-1439.
48. Gullett J, Donnelly JP, Sinert R, et al. Interobserver agreement in the evaluation of B-lines using bedside ultrasound. J Crit Care. 2015;30:1395-1399.
49. Guirguis-Blake JM, Beil TL, Sun X, et al. Primary Care Screening for Abdominal Aortic Aneurysm: A Systematic Evidence Review for the U.S. Preventive Services Task Force. Evidence Syntheses No. 109. Rockville, MD; 2014.
50. Metcalfe D, Holt PJE, Thompson MM. The management of abdominal aortic aneurysms. BMJ. 2011;342:d1384.
51. Thompson SG, Ashton HA, Gao L, et al. Final follow-up of the Multicentre Aneurysm Screening Study (MASS) randomized trial of abdominal aortic aneurysm screening. Brit J Surg. 2012;99:1649-1656.
52. LeFevre ML. Screening for abdominal aortic aneurysm: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:281-290.
53. Lindholt JS, Vammen S, Juul S, et al. The validity of ultrasonographic scanning as screening method for abdominal aortic aneurysm. Eur J Vasc Endovasc Surg. 1999;17:472-475.
54. Rubano E, Mehta N, Caputo W, et al. Systematic review: emergency department bedside ultrasonography for diagnosing suspected abdominal aortic aneurysm. Acad Emerg Med. 2013;20:128-138.
55. Blois B. Office-based ultrasound screening for abdominal aortic aneurysm. Can Fam Physician. 2012;58:e172-e178.
56. Sisó-Almirall A, Gilabert Solé R, Bru Saumell C, et al. Feasibility of hand-held-ultrasonography in the screening of abdominal aortic aneurysms and abdominal aortic atherosclerosis. Med Clin (Barc). 2013;141:417-422.
57. Sisó-Almirall A, Kostov B, Navarro González M, et al. Abdominal aortic aneurysm screening program using hand-held ultrasound in primary healthcare. PLoS One. 2017;12:e0176877.
58. Philbrick JT, Becker DM. Calf deep venous thrombosis: a wolf in sheep’s clothing? Arch Intern Med. 1988;148:2131-2138.
59. Bates SM, Jaeschke R, Stevens SM, et al. Diagnosis of DVT: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e351S-418S.
60. Cushman M, Tsai AW, White RH, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19-25.
61. Wells PS, Anderson DR, Rodger M, et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 2003;349:1227-1235.
62. Lensing AW, Prandoni P, Brandjes D, et al. Detection of deep-vein thrombosis by real-time B-mode ultrasonography. N Engl J Med. 1989;320:342-345.
63. Crisp JG, Lovato LM, Jang TB. Compression ultrasonography of the lower extremity with portable vascular ultrasonography can accurately detect deep venous thrombosis in the emergency department. Ann Emerg Med. 2010;56:601-610.
64. Blaivas M, Lambert MJ, Harwood RA, et al. Lower-extremity doppler for deep venous thrombosis—can emergency physicians be accurate and fast? Acad Emerg Med. 2000;7:120-126.
65. Burnside PR, Brown MD, Kline JA. Systematic review of emergency physician-performed ultrasonography for lower-extremity deep vein thrombosis. Acad Emerg Med. 2008;15:493-498.
66. Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350:1795-1798.
67. Birdwell BG, Raskob GE, Whitsett TL, et al. The clinical validity of normal compression ultrasonography in outpatients suspected of having deep venous thrombosis. Ann Intern Med. 1998;128:1-7.
68. Cogo A, Lensing AW, Koopman MM, et al. Compression ultrasonography for diagnostic management of patients with clinically suspected deep vein thrombosis: prospective cohort study. BMJ. 1998;316:17-20.
69. Tick LW, Ton E, Van Voorthuizen T, et al. Practical diagnostic management of patients with clinically suspected deep vein thrombosis by clinical probability test, compression ultrasonography, and D-dimer test. Am J Med. 2002;113:630-635.
70. Stevens
Point-of-care ultrasound (POCUS) has been gaining greater traction in recent years as a way to quickly (and cost-effectively) assess for conditions including systolic dysfunction, pleural effusion, abdominal aortic aneurysms (AAAs), and deep vein thrombosis (DVT). It involves limited and specific ultrasound protocols performed at the bedside by the health care provider who is trying to answer a specific question and, thus, help guide treatment of the patient.
POCUS was first widely used by emergency physicians starting in the early 1990s with the widespread adoption of the Focused Assessment with Sonography in Trauma (FAST) scan.1,2 Since that time, POCUS has expanded beyond trauma applications and into family medicine.
One study assessed physicians’ perceptions of POCUS after its integration into a military family medicine clinic. The study showed that physicians perceived POCUS to be relatively easy to use, not overly time consuming, and of high value to the practice.3 In fact, the literature tells us that POCUS can help decrease the cost of health care and improve outcomes,4-7 while requiring a relatively brief training period.

If residencies are any indication, POCUS may be headed your way
Ultrasound units are becoming smaller and more affordable, and medical schools are increasingly incorporating ultrasound curricula into medical student training.8 As of 2016, only 6% of practicing FPs reported using non-obstetric POCUS in their practices.9 Similarly, a survey from 2015 reported that only 2% of family medicine residency programs had established POCUS curricula.10 However, 50% of respondents in the 2015 survey reported early-stage development or interest in developing a POCUS curriculum.
Since then a validated family medicine residency curriculum has been published,11 and the American Academy of Family Physicians (AAFP) recently released a POCUS Curriculum Guideline for residencies (https://www.aafp.org/dam/AAFP/documents/medical_education_residency/program_directors/Reprint290D_POCUS.pdf).
[polldaddy:9928416]


The potential applications of POCUS in family medicine are numerous and have been reviewed in several recent publications.12,13 In this article, we will review the evidence for the use of POCUS in 4 areas: the cardiovascular exam (FIGURES 1 and 2), the lung exam (FIGURES 3-6), the screening exam for AAAs (FIGURE 7), and the evaluation for DVT (FIGURES 8 and 9). (Obstetric and musculoskeletal applications have been sufficiently covered elsewhere.14-17) For all of these applications, POCUS is safe, accurate, and beneficial and can be performed with a relatively small amount of training by non-radiology specialists, including FPs (TABLEs 1 and 2).








Just 2 hours of cardio POCUS training enhanced Dx accuracy
The American Society of Echocardiography (ASE) issued an expert consensus statement for focused cardiac ultrasound in 2013.18 The guideline supports non-cardiologists utilizing POCUS to assess for pericardial effusion and right and left ventricular enlargement, as well as to review global cardiac systolic function and intravascular volume status. Cardiovascular POCUS protocols are relatively easy to learn; even small amounts of training and practice can yield competency.
For example, a 2013 study showed that after 2 hours of training with a pocket ultrasound device, medical students and junior physicians inexperienced with POCUS were able to improve their diagnostic accuracy for heart failure from 50% to 75%.19 In another study, internal medicine residents with limited cardiac ultrasound training (ie, 20 practice exams) were able to detect decreased left ventricular ejection fraction using a handheld ultrasound device with 94% sensitivity and specificity in patients admitted to the hospital with acute decompensated heart failure.20 Similarly, after only 8 hours of training, a group of Norwegian general practitioners were able to obtain measurements of systolic function with a pocket ultrasound device that were not statistically different from a cardiologist’s measurements.21
In another study, rural FPs attended a 4-day course and then performed focused cardiac ultrasounds on primary care patients with a clinical indication for an echocardiogram.22 The scans were uploaded to a Web-based program for remote interpretation by a cardiologist. There was high concordance between the FPs’ interpretations of the focused cardiac ultrasounds and the cardiologist’s interpretations. Only 32% of the patients in the study group required a formal follow-up echocardiogram.
Kimura et al published a POCUS protocol for the rapid assessment of patients with heart failure, called the Cardiopulmonary Limited Ultrasound Exam (CLUE).23 The CLUE protocol utilizes 4 views to assess left ventricular systolic and diastolic function along with signs of pulmonary edema or systemic volume overload (TABLE 323). The presence of pulmonary edema or a plethoric inferior vena cava (IVC) was highly prognostic of in-hospital mortality. The CLUE protocol has been successfully used by novices including internal medicine residents after brief training (ie, up to 60 supervised scans) and can be performed in less than 5 minutes.24,25 
Inpatient use. In addition to its use as an outpatient diagnostic tool, POCUS may be able to help guide therapy in patients admitted to the hospital with heart failure. Increasing collapse of the IVC directly correlates with the amount of fluid volume removed during hemodialysis.26 Goonewardena et al showed that IVC collapsibility was an independent predictor of 30-day hospital readmission even when demographics, signs and symptoms, and volume of diuresis were otherwise equal.27 However, whether the use of IVC collapsibility to guide management improves outcomes in heart failure remains to be validated in a prospective trial.
More sensitive, specific than x-rays for pulmonary diagnoses
The chest x-ray has traditionally been the imaging modality of choice to evaluate primary care pulmonary complaints. However, POCUS can be more sensitive and specific than a chest x-ray for evaluating several pulmonary diagnoses including pleural effusion, pneumonia, and pulmonary edema.
Pleural effusion can be difficult to detect with a physical exam alone. A systematic review showed that the physical exam is not sensitive for effusions <300 mL and can have even lower utility in obese patients.28 While an upright lateral chest x-ray can accurately detect effusions as small as 50 mL, portable x-rays have sensitivities of only 53% to 71% for small- or moderate-sized effusions.29,30 Ultrasound, however, has a sensitivity of 97% for small effusions.31
A 2016 meta-analysis showed that POCUS had a pooled sensitivity and specificity of 94% and 98%, respectively, for pleural effusions, while chest x-ray had a pooled sensitivity and specificity of 51% and 91%, respectively, when compared with computed tomography (CT) and expert sonography.32 POCUS evaluation for pleural effusion is technically simple, and at least one study showed that even novice users can achieve high diagnostic accuracy after only 3 hours of training.33
Pneumonia is the eighth leading cause of death in the United States and the single leading cause of infectious disease death in children worldwide.34-36 Pneumonia is a difficult diagnosis to make based on a history and physical examination alone, and the Infectious Diseases Society of America recommends diagnostic imaging to make the diagnosis.37
The adult and pediatric literature clearly demonstrate that lung ultrasound is accurate at diagnosing pneumonia. In a 2015 meta-analysis of the pediatric literature, lung ultrasound had a sensitivity of 96% and a specificity of 93% and positive and negative likelihood ratios of 15.3 and 0.06, respectively.38 In adults, a 2016 meta-analysis of lung ultrasound showed a pooled sensitivity and specificity of 90% and 88%, respectively, with positive and negative likelihood ratios of 6.6 and 0.08, respectively.39
In 2015, a prospective study compared the accuracy of lung ultrasound and chest x-ray using CT as the gold standard.40 Lung ultrasound had a significantly better sensitivity of 82% compared to a sensitivity of 64% for chest x-ray. Specificities were comparable at 94% for ultrasound and 90% for chest x-ray.40
At least one study found novice sonographers to be accurate with lung POCUS for the diagnosis of pneumonia after only two 90-minute training sessions.41 Moreover, ultrasound has a more favorable safety profile, greater portability, and lower cost compared with chest x-ray and CT.
Pulmonary edema. Lung ultrasound can identify interstitial pulmonary edema via artifacts called B lines, which are produced by the reverberation of sound waves from the pleura due to the widening of the fluid-filled interlobular septa. These are distinctly different from the A-line pattern of repeating horizontal lines that is seen with normal lungs, making lung ultrasound more accurate than chest x-ray for identification of pulmonary edema.42,43 When final diagnosis via blinded chart review is used as the reference standard, bilateral B lines on a lung ultrasound image have a sensitivity of 86% to 100% and a specificity of 92% to 98% for the diagnosis of pulmonary edema compared to chest x-ray’s sensitivity of 56.9% and specificity of 89.2%.44 There is also a linear correlation between the number of B lines present and the extent of pulmonary edema.42,45,46 The number of B lines decreases in real time as volume is removed in dialysis patients.47
POCUS evaluation for B lines can be learned very quickly. Exams of novices who have performed only 5 prior exams correlate highly with those of experts who have performed more than 100 exams.48
Simple, efficient screening method for abdominal aortic aneurysm
AAAs are present in up to 7% of men over the age of 50.49 The mortality rate of a ruptured AAA is as high as 80% to 95%.50 There is, however, a long prodromal period when interventions can make a significant difference, which is why accurate screening is so important.
AAA screening with ultrasound has been shown to decrease mortality.51 The current recommendation of the US Preventive Services Task Force (USPSTF) is a one-time AAA screening for all men ages 65 to 75 years who have ever smoked (Grade B).52 Despite the recommendations of the USPSTF, screening rates are low. One study found that only 9% of eligible patients in primary care practices received appropriate screening.51
Ultrasound performed by specialists is known to be an excellent screening test for AAA with a sensitivity of 98.9% and a specificity of 99.9%.53 POCUS use by emergency medicine physicians for the evaluation of symptomatic AAA is well established in the literature. A meta-analysis including 7 studies and 655 patients showed a pooled sensitivity of 99% and a specificity of 98%.54 Multiple studies also support primary care physicians performing POCUS AAA screening in the clinic setting.
For example, a 2012 prospective, observational study performed in Canada compared office-based ultrasound screening exams performed by a rural FP to scans performed in the hospital on the same patients.55 The physician completed 50 training examinations. The average discrepancy in aorta diameters between the 2 was only 2 mm, which is clinically insignificant, and the office-based scans had a sensitivity and specificity of 100%.
Similarly, a second FP study performed in Barcelona, showed that an FP who performed POCUS AAA screening had 100% concordance with a radiologist.56 Additionally, POCUS screening for AAA was not time consuming; it was performed in under 4 minutes per patient.55,57
Ruling out DVT
DVT is a relatively rare occurrence in the ambulatory setting. However, patients who present with a painful, swollen lower extremity are much more common, and DVT must be considered and ruled out in these situations.
Although isolated distal DVTs that occur in the calf veins are usually self-limited and have a very low risk of embolization, they can progress to proximal DVTs of the thigh veins up to 20% of time.58,59 Similarly, thrombophlebitis of the superficial lower extremity veins rarely embolizes, but can progress to a proximal DVT, especially if large segments are involved or if the segments are within 5 cm of the junction to the deep venous system.59 The risk of missing a proximal leg DVT is high because embolization occurs up to 60% of the time if the DVT is left untreated.60
The current standard for diagnosis of DVT is the lower extremity Doppler ultrasound examination, but obtaining same-day Doppler evaluations can be difficult in the ambulatory setting. In these instances, the American College of Chest Physicians (ACCP) recommends that even low-risk patients receive anticoagulation pending the evaluation if it cannot be obtained in the first 24 hours.59 This approach not only increases the cost of care, but also exposes patients—many of whom will not be diagnosed with thrombosis in the end—to the risks of anticoagulation.
D-dimer blood tests have drawbacks, too. While a negative high-sensitivity D-dimer blood test in a patient with a low pre-test probability of DVT can effectively rule out a DVT, laboratory testing is not always immediately available in the ambulatory setting either.61 Additionally, false-positive rates are high, and positive D-dimer exams still require evaluation by Doppler ultrasound.
Given these limitations, performing an ultrasound at the bedside or in the exam room can allow for more timely and cost-effective care. In fact, research shows that a limited ultrasound, called the 2-region compression exam, which follows along the course of the common femoral vein and popliteal vein only, ignoring the femoral and calf veins, is highly accurate in assessing for proximal leg DVTs. As such, it has been adopted for POCUS use by emergency medicine physicians.62
Multiple studies show that physicians with minimal training can perform the 2-region compression exam with a high degree of accuracy when full-leg Doppler ultrasound was used as the gold standard.63,64 In these studies, hands-on training times ranged from only 10 minutes to 5 hours, and the exam could be performed in less than 4 minutes. A systematic review of 6 studies comparing emergency physician-performed ultrasound with radiology-performed ultrasound calculated an overall sensitivity of 0.95 (95% CI, 0.87-0.99) and specificity of 0.96 (95% CI, 0.87-0.99) for those performed by emergency physicians.65
The main concern with the 2-region compression exam is that it can miss a distal leg DVT. As stated earlier, distal DVTs are relatively benign and tend to resolve without treatment; however, up to 20% can progress to become a dangerous proximal leg DVT.58 Researchers have validated several methods by prospective trials to address this limitation.
Specifically, researchers have demonstrated that patients with a low pre-test probability of DVT per the Wells scoring system could have DVT effectively ruled out with a single 2-region compression ultrasound without further evaluation.66 In another study, researchers evaluated all patients (regardless of pretest probability) with a 2-point compression exam and found that those with negative exams could be followed with a second exam in 7 to 10 days without initiating anticoagulation. If the second one was negative, no further evaluation was needed.67,68
And finally, researchers demonstrated that a negative 2-point compression ultrasound in combination with a concurrent negative D-dimer test was effective at ruling out DVT, regardless of pre-test probability.69,70
A preferred approach
Given this data and the fact that in the ambulatory setting it is often easier and faster to perform a 2-region compression examination than to obtain a D-dimer laboratory test or a formal full-leg Doppler ultrasound, what follows is our preferred approach to a patient with suspected DVT in the outpatient setting (FIGURE 10).

We first assess pre-test probability using the Wells scoring system. We then perform the 2-region compression ultrasound. If the patient has low pre-test risk according to the Wells score, we rule out DVT. If the patient has moderate or high risk with a negative 2-region compression ultrasound, the patient gets a D-dimer test. If the D-dimer test is negative, we rule out DVT. If the D-dimer test is positive, we schedule the patient for a repeat 2-region compression ultrasound in 7 to 10 days. If at any time the 2-region compression evaluation is positive, we treat the patient for DVT.
CORRESPONDENCE
Paul Bornemann, MD, Palmetto Health Family Medicine Residency, Department of Family and Preventive Medicine, University of South Carolina School of Medicine, 3209 Colonial Drive, Columbia, SC 29203; [email protected].
Point-of-care ultrasound (POCUS) has been gaining greater traction in recent years as a way to quickly (and cost-effectively) assess for conditions including systolic dysfunction, pleural effusion, abdominal aortic aneurysms (AAAs), and deep vein thrombosis (DVT). It involves limited and specific ultrasound protocols performed at the bedside by the health care provider who is trying to answer a specific question and, thus, help guide treatment of the patient.
POCUS was first widely used by emergency physicians starting in the early 1990s with the widespread adoption of the Focused Assessment with Sonography in Trauma (FAST) scan.1,2 Since that time, POCUS has expanded beyond trauma applications and into family medicine.
One study assessed physicians’ perceptions of POCUS after its integration into a military family medicine clinic. The study showed that physicians perceived POCUS to be relatively easy to use, not overly time consuming, and of high value to the practice.3 In fact, the literature tells us that POCUS can help decrease the cost of health care and improve outcomes,4-7 while requiring a relatively brief training period.

If residencies are any indication, POCUS may be headed your way
Ultrasound units are becoming smaller and more affordable, and medical schools are increasingly incorporating ultrasound curricula into medical student training.8 As of 2016, only 6% of practicing FPs reported using non-obstetric POCUS in their practices.9 Similarly, a survey from 2015 reported that only 2% of family medicine residency programs had established POCUS curricula.10 However, 50% of respondents in the 2015 survey reported early-stage development or interest in developing a POCUS curriculum.
Since then a validated family medicine residency curriculum has been published,11 and the American Academy of Family Physicians (AAFP) recently released a POCUS Curriculum Guideline for residencies (https://www.aafp.org/dam/AAFP/documents/medical_education_residency/program_directors/Reprint290D_POCUS.pdf).
[polldaddy:9928416]


The potential applications of POCUS in family medicine are numerous and have been reviewed in several recent publications.12,13 In this article, we will review the evidence for the use of POCUS in 4 areas: the cardiovascular exam (FIGURES 1 and 2), the lung exam (FIGURES 3-6), the screening exam for AAAs (FIGURE 7), and the evaluation for DVT (FIGURES 8 and 9). (Obstetric and musculoskeletal applications have been sufficiently covered elsewhere.14-17) For all of these applications, POCUS is safe, accurate, and beneficial and can be performed with a relatively small amount of training by non-radiology specialists, including FPs (TABLEs 1 and 2).








Just 2 hours of cardio POCUS training enhanced Dx accuracy
The American Society of Echocardiography (ASE) issued an expert consensus statement for focused cardiac ultrasound in 2013.18 The guideline supports non-cardiologists utilizing POCUS to assess for pericardial effusion and right and left ventricular enlargement, as well as to review global cardiac systolic function and intravascular volume status. Cardiovascular POCUS protocols are relatively easy to learn; even small amounts of training and practice can yield competency.
For example, a 2013 study showed that after 2 hours of training with a pocket ultrasound device, medical students and junior physicians inexperienced with POCUS were able to improve their diagnostic accuracy for heart failure from 50% to 75%.19 In another study, internal medicine residents with limited cardiac ultrasound training (ie, 20 practice exams) were able to detect decreased left ventricular ejection fraction using a handheld ultrasound device with 94% sensitivity and specificity in patients admitted to the hospital with acute decompensated heart failure.20 Similarly, after only 8 hours of training, a group of Norwegian general practitioners were able to obtain measurements of systolic function with a pocket ultrasound device that were not statistically different from a cardiologist’s measurements.21
In another study, rural FPs attended a 4-day course and then performed focused cardiac ultrasounds on primary care patients with a clinical indication for an echocardiogram.22 The scans were uploaded to a Web-based program for remote interpretation by a cardiologist. There was high concordance between the FPs’ interpretations of the focused cardiac ultrasounds and the cardiologist’s interpretations. Only 32% of the patients in the study group required a formal follow-up echocardiogram.
Kimura et al published a POCUS protocol for the rapid assessment of patients with heart failure, called the Cardiopulmonary Limited Ultrasound Exam (CLUE).23 The CLUE protocol utilizes 4 views to assess left ventricular systolic and diastolic function along with signs of pulmonary edema or systemic volume overload (TABLE 323). The presence of pulmonary edema or a plethoric inferior vena cava (IVC) was highly prognostic of in-hospital mortality. The CLUE protocol has been successfully used by novices including internal medicine residents after brief training (ie, up to 60 supervised scans) and can be performed in less than 5 minutes.24,25 
Inpatient use. In addition to its use as an outpatient diagnostic tool, POCUS may be able to help guide therapy in patients admitted to the hospital with heart failure. Increasing collapse of the IVC directly correlates with the amount of fluid volume removed during hemodialysis.26 Goonewardena et al showed that IVC collapsibility was an independent predictor of 30-day hospital readmission even when demographics, signs and symptoms, and volume of diuresis were otherwise equal.27 However, whether the use of IVC collapsibility to guide management improves outcomes in heart failure remains to be validated in a prospective trial.
More sensitive, specific than x-rays for pulmonary diagnoses
The chest x-ray has traditionally been the imaging modality of choice to evaluate primary care pulmonary complaints. However, POCUS can be more sensitive and specific than a chest x-ray for evaluating several pulmonary diagnoses including pleural effusion, pneumonia, and pulmonary edema.
Pleural effusion can be difficult to detect with a physical exam alone. A systematic review showed that the physical exam is not sensitive for effusions <300 mL and can have even lower utility in obese patients.28 While an upright lateral chest x-ray can accurately detect effusions as small as 50 mL, portable x-rays have sensitivities of only 53% to 71% for small- or moderate-sized effusions.29,30 Ultrasound, however, has a sensitivity of 97% for small effusions.31
A 2016 meta-analysis showed that POCUS had a pooled sensitivity and specificity of 94% and 98%, respectively, for pleural effusions, while chest x-ray had a pooled sensitivity and specificity of 51% and 91%, respectively, when compared with computed tomography (CT) and expert sonography.32 POCUS evaluation for pleural effusion is technically simple, and at least one study showed that even novice users can achieve high diagnostic accuracy after only 3 hours of training.33
Pneumonia is the eighth leading cause of death in the United States and the single leading cause of infectious disease death in children worldwide.34-36 Pneumonia is a difficult diagnosis to make based on a history and physical examination alone, and the Infectious Diseases Society of America recommends diagnostic imaging to make the diagnosis.37
The adult and pediatric literature clearly demonstrate that lung ultrasound is accurate at diagnosing pneumonia. In a 2015 meta-analysis of the pediatric literature, lung ultrasound had a sensitivity of 96% and a specificity of 93% and positive and negative likelihood ratios of 15.3 and 0.06, respectively.38 In adults, a 2016 meta-analysis of lung ultrasound showed a pooled sensitivity and specificity of 90% and 88%, respectively, with positive and negative likelihood ratios of 6.6 and 0.08, respectively.39
In 2015, a prospective study compared the accuracy of lung ultrasound and chest x-ray using CT as the gold standard.40 Lung ultrasound had a significantly better sensitivity of 82% compared to a sensitivity of 64% for chest x-ray. Specificities were comparable at 94% for ultrasound and 90% for chest x-ray.40
At least one study found novice sonographers to be accurate with lung POCUS for the diagnosis of pneumonia after only two 90-minute training sessions.41 Moreover, ultrasound has a more favorable safety profile, greater portability, and lower cost compared with chest x-ray and CT.
Pulmonary edema. Lung ultrasound can identify interstitial pulmonary edema via artifacts called B lines, which are produced by the reverberation of sound waves from the pleura due to the widening of the fluid-filled interlobular septa. These are distinctly different from the A-line pattern of repeating horizontal lines that is seen with normal lungs, making lung ultrasound more accurate than chest x-ray for identification of pulmonary edema.42,43 When final diagnosis via blinded chart review is used as the reference standard, bilateral B lines on a lung ultrasound image have a sensitivity of 86% to 100% and a specificity of 92% to 98% for the diagnosis of pulmonary edema compared to chest x-ray’s sensitivity of 56.9% and specificity of 89.2%.44 There is also a linear correlation between the number of B lines present and the extent of pulmonary edema.42,45,46 The number of B lines decreases in real time as volume is removed in dialysis patients.47
POCUS evaluation for B lines can be learned very quickly. Exams of novices who have performed only 5 prior exams correlate highly with those of experts who have performed more than 100 exams.48
Simple, efficient screening method for abdominal aortic aneurysm
AAAs are present in up to 7% of men over the age of 50.49 The mortality rate of a ruptured AAA is as high as 80% to 95%.50 There is, however, a long prodromal period when interventions can make a significant difference, which is why accurate screening is so important.
AAA screening with ultrasound has been shown to decrease mortality.51 The current recommendation of the US Preventive Services Task Force (USPSTF) is a one-time AAA screening for all men ages 65 to 75 years who have ever smoked (Grade B).52 Despite the recommendations of the USPSTF, screening rates are low. One study found that only 9% of eligible patients in primary care practices received appropriate screening.51
Ultrasound performed by specialists is known to be an excellent screening test for AAA with a sensitivity of 98.9% and a specificity of 99.9%.53 POCUS use by emergency medicine physicians for the evaluation of symptomatic AAA is well established in the literature. A meta-analysis including 7 studies and 655 patients showed a pooled sensitivity of 99% and a specificity of 98%.54 Multiple studies also support primary care physicians performing POCUS AAA screening in the clinic setting.
For example, a 2012 prospective, observational study performed in Canada compared office-based ultrasound screening exams performed by a rural FP to scans performed in the hospital on the same patients.55 The physician completed 50 training examinations. The average discrepancy in aorta diameters between the 2 was only 2 mm, which is clinically insignificant, and the office-based scans had a sensitivity and specificity of 100%.
Similarly, a second FP study performed in Barcelona, showed that an FP who performed POCUS AAA screening had 100% concordance with a radiologist.56 Additionally, POCUS screening for AAA was not time consuming; it was performed in under 4 minutes per patient.55,57
Ruling out DVT
DVT is a relatively rare occurrence in the ambulatory setting. However, patients who present with a painful, swollen lower extremity are much more common, and DVT must be considered and ruled out in these situations.
Although isolated distal DVTs that occur in the calf veins are usually self-limited and have a very low risk of embolization, they can progress to proximal DVTs of the thigh veins up to 20% of time.58,59 Similarly, thrombophlebitis of the superficial lower extremity veins rarely embolizes, but can progress to a proximal DVT, especially if large segments are involved or if the segments are within 5 cm of the junction to the deep venous system.59 The risk of missing a proximal leg DVT is high because embolization occurs up to 60% of the time if the DVT is left untreated.60
The current standard for diagnosis of DVT is the lower extremity Doppler ultrasound examination, but obtaining same-day Doppler evaluations can be difficult in the ambulatory setting. In these instances, the American College of Chest Physicians (ACCP) recommends that even low-risk patients receive anticoagulation pending the evaluation if it cannot be obtained in the first 24 hours.59 This approach not only increases the cost of care, but also exposes patients—many of whom will not be diagnosed with thrombosis in the end—to the risks of anticoagulation.
D-dimer blood tests have drawbacks, too. While a negative high-sensitivity D-dimer blood test in a patient with a low pre-test probability of DVT can effectively rule out a DVT, laboratory testing is not always immediately available in the ambulatory setting either.61 Additionally, false-positive rates are high, and positive D-dimer exams still require evaluation by Doppler ultrasound.
Given these limitations, performing an ultrasound at the bedside or in the exam room can allow for more timely and cost-effective care. In fact, research shows that a limited ultrasound, called the 2-region compression exam, which follows along the course of the common femoral vein and popliteal vein only, ignoring the femoral and calf veins, is highly accurate in assessing for proximal leg DVTs. As such, it has been adopted for POCUS use by emergency medicine physicians.62
Multiple studies show that physicians with minimal training can perform the 2-region compression exam with a high degree of accuracy when full-leg Doppler ultrasound was used as the gold standard.63,64 In these studies, hands-on training times ranged from only 10 minutes to 5 hours, and the exam could be performed in less than 4 minutes. A systematic review of 6 studies comparing emergency physician-performed ultrasound with radiology-performed ultrasound calculated an overall sensitivity of 0.95 (95% CI, 0.87-0.99) and specificity of 0.96 (95% CI, 0.87-0.99) for those performed by emergency physicians.65
The main concern with the 2-region compression exam is that it can miss a distal leg DVT. As stated earlier, distal DVTs are relatively benign and tend to resolve without treatment; however, up to 20% can progress to become a dangerous proximal leg DVT.58 Researchers have validated several methods by prospective trials to address this limitation.
Specifically, researchers have demonstrated that patients with a low pre-test probability of DVT per the Wells scoring system could have DVT effectively ruled out with a single 2-region compression ultrasound without further evaluation.66 In another study, researchers evaluated all patients (regardless of pretest probability) with a 2-point compression exam and found that those with negative exams could be followed with a second exam in 7 to 10 days without initiating anticoagulation. If the second one was negative, no further evaluation was needed.67,68
And finally, researchers demonstrated that a negative 2-point compression ultrasound in combination with a concurrent negative D-dimer test was effective at ruling out DVT, regardless of pre-test probability.69,70
A preferred approach
Given this data and the fact that in the ambulatory setting it is often easier and faster to perform a 2-region compression examination than to obtain a D-dimer laboratory test or a formal full-leg Doppler ultrasound, what follows is our preferred approach to a patient with suspected DVT in the outpatient setting (FIGURE 10).

We first assess pre-test probability using the Wells scoring system. We then perform the 2-region compression ultrasound. If the patient has low pre-test risk according to the Wells score, we rule out DVT. If the patient has moderate or high risk with a negative 2-region compression ultrasound, the patient gets a D-dimer test. If the D-dimer test is negative, we rule out DVT. If the D-dimer test is positive, we schedule the patient for a repeat 2-region compression ultrasound in 7 to 10 days. If at any time the 2-region compression evaluation is positive, we treat the patient for DVT.
CORRESPONDENCE
Paul Bornemann, MD, Palmetto Health Family Medicine Residency, Department of Family and Preventive Medicine, University of South Carolina School of Medicine, 3209 Colonial Drive, Columbia, SC 29203; [email protected].
1. Hahn RG, Davies TC, Rodney WM. Diagnostic ultrasound in general practice. Fam Pract. 1988;5:129-135.
2. Deutchman ME, Hahn RG, Rodney WMM. Diagnostic ultrasound imaging by physicians of first contact: extending the family medicine experience into emergency medicine. Ann Emerg Med. 1993;22:594-596.
3. Bornemann P, Bornemann G. Military family physicians’ perceptions of a pocket point-of-care ultrasound device in clinical practice. Mil Med. 2014;179:1474-1477.
4. Smith-Bindman R, Aubin C, Bailitz J, et al. Ultrasonography versus computed tomography for suspected nephrolithiasis. N Engl J Med. 2014;371:1100-1110.
5. Parker L, Nazarian LN, Carrino JA, et al. Musculoskeletal imaging: medicare use, costs, and potential for cost substitution. J Am Coll Radiol. 2008;5:182-188.
6. Gordon CE, Feller-Kopman D, Balk EM, et al. Pneumothorax following thoracentesis: a systematic review and meta-analysis. Arch Intern Med. 2010;170:332-339.
7. Calvert N, Hind D, McWilliams RG, et al. The effectiveness and cost-effectiveness of ultrasound locating devices for central venous access: a systematic review and economic evaluation. Health Technol Assess. 2003;7:1-84.
8. Hoppmann RA, Rao VV, Bell F, et al. The evolution of an integrated ultrasound curriculum (iUSC) for medical students: 9-year experience. Crit Ultrasound J. 2015;7:18.
9. Clinical procedures performed by physicians at their practice. American Academy of Family Physicians Member Census, December 31, 2016. Available at: http://www.aafp.org/about/the-aafp/family-medicine-facts/table-12(rev).html. Accessed June 26, 2017.
10. Hall JW, Holman H, Bornemann P, et al. Point of care ultrasound in family medicine residency programs: a CERA study. Fam Med. 2015;47:706-711.
11. Bornemann P. Assessment of a novel point-of-care ultrasound curriculum’s effect on competency measures in family medicine graduate medical education. J Ultrasound Med. 2017;36:1205-1211.
12. Steinmetz P, Oleskevich S. The benefits of doing ultrasound exams in your office. J Fam Pract. 2016;65:517-523.
13. Flick D. Bedside ultrasound education in family medicine. J Ultrasound Med. 2016;35:1369-1371.
14. Dresang LT, Rodney WM, Rodney KM. Prenatal ultrasound: a tale of two cities. J Natl Med Assoc. 2006;98:167-171.
15. Dresang LT, Rodney WM, Dees J. Teaching prenatal ultrasound to family medicine residents. Fam Med. 2004;36:98-107.
16. Rodney WM, Deutchman ME, Hartman KJ, et al. Obstetric ultrasound by family physicians. J Fam Pract. 1992;34:186-194.
17. Broadhurst NA, Simmons N. Musculoskeletal ultrasound - used to best advantage. Aust Fam Physician. 2007;36:430-432.
18. Spencer KT, Kimura BJ, Korcarz CE, et al. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:567-581.
19. Panoulas VF, Daigeler AL, Malaweera AS, et al. Pocket-size hand-held cardiac ultrasound as an adjunct to clinical examination in the hands of medical students and junior doctors. Eur Heart J Cardiovasc Imaging. 2013;14:323-330.
20. Razi R, Estrada JR, Doll J, et al. Bedside hand-carried ultrasound by internal medicine residents versus traditional clinical assessment for the identification of systolic dysfunction in patients admitted with decompensated heart failure. J Am Soc Echocardiogr. 2011;24:1319-1324.
21. Mjølstad OC, Snare SR, Folkvord L, et al. Assessment of left ventricular function by GPs using pocket-sized ultrasound. Fam Pract. 2012;29:534-540.
22. Evangelista A, Galuppo V, Méndez J, et al. Hand-held cardiac ultrasound screening performed by family doctors with remote expert support interpretation. Heart. 2016;102:376-382.
23. Kimura BJ, Yogo N, O’Connell CW, et al. Cardiopulmonary limited ultrasound examination for “quick-look” bedside application. Am J Cardiol. 2011;108:586-590.
24. Kimura BJ, Amundson SA, Phan JN, et al. Observations during development of an internal medicine residency training program in cardiovascular limited ultrasound examination. J Hosp Med. 2012;7:537-542.
25. Kimura BJ, Shaw DJ, Amundson SA, et al. Cardiac limited ultrasound examination techniques to augment the bedside cardiac physical examination. J Ultrasound Med. 2015;34:1683-1690.
26. Brennan JM, Ronan A, Goonewardena S, et al. Handcarried ultrasound measurement of the inferior vena cava for assessment of intravascular volume status in the outpatient hemodialysis clinic. Clin J Am Soc Nephrol. 2006;1:749-753.
27. Goonewardena SN, Gemignani A, Ronan A, et al. Comparison of hand-carried ultrasound assessment of the inferior vena cava and N-terminal pro-brain natriuretic peptide for predicting readmission after hospitalization for acute decompensated heart failure. JACC Cardiovasc Imaging. 2008;1:595-601.
28. Wong CL, Holroyd-Leduc J, Straus SE. Does this patient have a pleural effusion? JAMA. 2009;301:309-317.
29. Blackmore CC, Black WC, Dallas RV, et al. Pleural fluid volume estimation: a chest radiograph prediction rule. Acad Radiol. 1996;3:103-109.
30. Kitazono MT, Lau CT, Parada AN, et al. Differentiation of pleural effusions from parenchymal opacities: accuracy of bedside chest radiography. Am J Roentgenol. 2010;194:407-412.
31. Kalokairinou-Motogna M, Maratou K, Paianid I, et al. Application of color Doppler ultrasound in the study of small pleural effusion. Med Ultrason. 2010;12:12-16.
32. Yousefifard M, Baikpour M, Ghelichkhani P, et al. Screening performance characteristic of ultrasonography and radiography in detection of pleural effusion; a meta-analysis. Emerg (Tehran, Iran). 2016;4:1-10.
33. Begot E, Grumann A, Duvoid T, et al. Ultrasonographic identification and semiquantitative assessment of unloculated pleural effusions in critically ill patients by residents after a focused training. Intensive Care Med. 2014;40:1475-1480.
34. World Health Organization. Pneumonia. Fact Sheet No. 331. Available at: http://www.who.int/mediacentre/factsheets/fs331/en/. Accessed June 26, 2017.
35. Gereige RS, Laufer PM. Pneumonia. Pediatr Rev. 2013;34:438-456.
36. National Center for Health Statistics. Leading causes of death. https://www.cdc.gov/nchs/fastats/leading-causes-of-death.htm. Accessed July 2, 2017.
37. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-S72.
38. Pereda MA, Chavez MA, Hooper-Miele CC, et al. Lung ultrasound for the diagnosis of pneumonia in children: a meta-analysis. Pediatrics. 2015;135:714-722.
39. Xia Y, Ying Y, Wang S, et al. Effectiveness of lung ultrasonography for diagnosis of pneumonia in adults: a systematic review and meta-analysis. J Thorac Dis. 2016;8:2822-2831.
40. Nazerian P, Volpicelli G, Vanni S, et al. Accuracy of lung ultrasound for the diagnosis of consolidations when compared to chest computed tomography. Am J Emerg Med. 2015;33:620-625.
41. Filopei J, Siedenburg H, Rattner P, et al. Impact of pocket ultrasound use by internal medicine housestaff in the diagnosis of dyspnea. J Hosp Med. 2014;9:594-597.
42. Lichtenstein D, Mezière G. A lung ultrasound sign allowing bedside distinction between pulmonary edema and COPD: the comet-tail artifact. Intensive Care Med. 1998;24:1331-1334.
43. Gargani L, Volpicelli G. How I do it: lung ultrasound. Cardiovasc Ultrasound. 2014;12:25.
44. Martindale JL, Wakai A, Collins SP, et al. Diagnosing acute heart failure in the emergency department: a systematic review and meta-analysis. Acad Emerg Med. 2016;23:223-242.
45. Volpicelli G, Mussa A, Garofalo G, et al. Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. Am J Emerg Med. 2006;24:689-696.
46. Picano E, Frassi F, Agricola E, et al. Ultrasound lung comets: a clinically useful sign of extravascular lung water. J Am Soc Echocardiogr. 2006;19:356-363.
47. Noble VE, Murray AF, Capp R, et al. Ultrasound assessment for extravascular lung water in patients undergoing hemodialysis: time course for resolution. Chest. 2009;135:1433-1439.
48. Gullett J, Donnelly JP, Sinert R, et al. Interobserver agreement in the evaluation of B-lines using bedside ultrasound. J Crit Care. 2015;30:1395-1399.
49. Guirguis-Blake JM, Beil TL, Sun X, et al. Primary Care Screening for Abdominal Aortic Aneurysm: A Systematic Evidence Review for the U.S. Preventive Services Task Force. Evidence Syntheses No. 109. Rockville, MD; 2014.
50. Metcalfe D, Holt PJE, Thompson MM. The management of abdominal aortic aneurysms. BMJ. 2011;342:d1384.
51. Thompson SG, Ashton HA, Gao L, et al. Final follow-up of the Multicentre Aneurysm Screening Study (MASS) randomized trial of abdominal aortic aneurysm screening. Brit J Surg. 2012;99:1649-1656.
52. LeFevre ML. Screening for abdominal aortic aneurysm: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:281-290.
53. Lindholt JS, Vammen S, Juul S, et al. The validity of ultrasonographic scanning as screening method for abdominal aortic aneurysm. Eur J Vasc Endovasc Surg. 1999;17:472-475.
54. Rubano E, Mehta N, Caputo W, et al. Systematic review: emergency department bedside ultrasonography for diagnosing suspected abdominal aortic aneurysm. Acad Emerg Med. 2013;20:128-138.
55. Blois B. Office-based ultrasound screening for abdominal aortic aneurysm. Can Fam Physician. 2012;58:e172-e178.
56. Sisó-Almirall A, Gilabert Solé R, Bru Saumell C, et al. Feasibility of hand-held-ultrasonography in the screening of abdominal aortic aneurysms and abdominal aortic atherosclerosis. Med Clin (Barc). 2013;141:417-422.
57. Sisó-Almirall A, Kostov B, Navarro González M, et al. Abdominal aortic aneurysm screening program using hand-held ultrasound in primary healthcare. PLoS One. 2017;12:e0176877.
58. Philbrick JT, Becker DM. Calf deep venous thrombosis: a wolf in sheep’s clothing? Arch Intern Med. 1988;148:2131-2138.
59. Bates SM, Jaeschke R, Stevens SM, et al. Diagnosis of DVT: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e351S-418S.
60. Cushman M, Tsai AW, White RH, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19-25.
61. Wells PS, Anderson DR, Rodger M, et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 2003;349:1227-1235.
62. Lensing AW, Prandoni P, Brandjes D, et al. Detection of deep-vein thrombosis by real-time B-mode ultrasonography. N Engl J Med. 1989;320:342-345.
63. Crisp JG, Lovato LM, Jang TB. Compression ultrasonography of the lower extremity with portable vascular ultrasonography can accurately detect deep venous thrombosis in the emergency department. Ann Emerg Med. 2010;56:601-610.
64. Blaivas M, Lambert MJ, Harwood RA, et al. Lower-extremity doppler for deep venous thrombosis—can emergency physicians be accurate and fast? Acad Emerg Med. 2000;7:120-126.
65. Burnside PR, Brown MD, Kline JA. Systematic review of emergency physician-performed ultrasonography for lower-extremity deep vein thrombosis. Acad Emerg Med. 2008;15:493-498.
66. Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350:1795-1798.
67. Birdwell BG, Raskob GE, Whitsett TL, et al. The clinical validity of normal compression ultrasonography in outpatients suspected of having deep venous thrombosis. Ann Intern Med. 1998;128:1-7.
68. Cogo A, Lensing AW, Koopman MM, et al. Compression ultrasonography for diagnostic management of patients with clinically suspected deep vein thrombosis: prospective cohort study. BMJ. 1998;316:17-20.
69. Tick LW, Ton E, Van Voorthuizen T, et al. Practical diagnostic management of patients with clinically suspected deep vein thrombosis by clinical probability test, compression ultrasonography, and D-dimer test. Am J Med. 2002;113:630-635.
70. Stevens
1. Hahn RG, Davies TC, Rodney WM. Diagnostic ultrasound in general practice. Fam Pract. 1988;5:129-135.
2. Deutchman ME, Hahn RG, Rodney WMM. Diagnostic ultrasound imaging by physicians of first contact: extending the family medicine experience into emergency medicine. Ann Emerg Med. 1993;22:594-596.
3. Bornemann P, Bornemann G. Military family physicians’ perceptions of a pocket point-of-care ultrasound device in clinical practice. Mil Med. 2014;179:1474-1477.
4. Smith-Bindman R, Aubin C, Bailitz J, et al. Ultrasonography versus computed tomography for suspected nephrolithiasis. N Engl J Med. 2014;371:1100-1110.
5. Parker L, Nazarian LN, Carrino JA, et al. Musculoskeletal imaging: medicare use, costs, and potential for cost substitution. J Am Coll Radiol. 2008;5:182-188.
6. Gordon CE, Feller-Kopman D, Balk EM, et al. Pneumothorax following thoracentesis: a systematic review and meta-analysis. Arch Intern Med. 2010;170:332-339.
7. Calvert N, Hind D, McWilliams RG, et al. The effectiveness and cost-effectiveness of ultrasound locating devices for central venous access: a systematic review and economic evaluation. Health Technol Assess. 2003;7:1-84.
8. Hoppmann RA, Rao VV, Bell F, et al. The evolution of an integrated ultrasound curriculum (iUSC) for medical students: 9-year experience. Crit Ultrasound J. 2015;7:18.
9. Clinical procedures performed by physicians at their practice. American Academy of Family Physicians Member Census, December 31, 2016. Available at: http://www.aafp.org/about/the-aafp/family-medicine-facts/table-12(rev).html. Accessed June 26, 2017.
10. Hall JW, Holman H, Bornemann P, et al. Point of care ultrasound in family medicine residency programs: a CERA study. Fam Med. 2015;47:706-711.
11. Bornemann P. Assessment of a novel point-of-care ultrasound curriculum’s effect on competency measures in family medicine graduate medical education. J Ultrasound Med. 2017;36:1205-1211.
12. Steinmetz P, Oleskevich S. The benefits of doing ultrasound exams in your office. J Fam Pract. 2016;65:517-523.
13. Flick D. Bedside ultrasound education in family medicine. J Ultrasound Med. 2016;35:1369-1371.
14. Dresang LT, Rodney WM, Rodney KM. Prenatal ultrasound: a tale of two cities. J Natl Med Assoc. 2006;98:167-171.
15. Dresang LT, Rodney WM, Dees J. Teaching prenatal ultrasound to family medicine residents. Fam Med. 2004;36:98-107.
16. Rodney WM, Deutchman ME, Hartman KJ, et al. Obstetric ultrasound by family physicians. J Fam Pract. 1992;34:186-194.
17. Broadhurst NA, Simmons N. Musculoskeletal ultrasound - used to best advantage. Aust Fam Physician. 2007;36:430-432.
18. Spencer KT, Kimura BJ, Korcarz CE, et al. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:567-581.
19. Panoulas VF, Daigeler AL, Malaweera AS, et al. Pocket-size hand-held cardiac ultrasound as an adjunct to clinical examination in the hands of medical students and junior doctors. Eur Heart J Cardiovasc Imaging. 2013;14:323-330.
20. Razi R, Estrada JR, Doll J, et al. Bedside hand-carried ultrasound by internal medicine residents versus traditional clinical assessment for the identification of systolic dysfunction in patients admitted with decompensated heart failure. J Am Soc Echocardiogr. 2011;24:1319-1324.
21. Mjølstad OC, Snare SR, Folkvord L, et al. Assessment of left ventricular function by GPs using pocket-sized ultrasound. Fam Pract. 2012;29:534-540.
22. Evangelista A, Galuppo V, Méndez J, et al. Hand-held cardiac ultrasound screening performed by family doctors with remote expert support interpretation. Heart. 2016;102:376-382.
23. Kimura BJ, Yogo N, O’Connell CW, et al. Cardiopulmonary limited ultrasound examination for “quick-look” bedside application. Am J Cardiol. 2011;108:586-590.
24. Kimura BJ, Amundson SA, Phan JN, et al. Observations during development of an internal medicine residency training program in cardiovascular limited ultrasound examination. J Hosp Med. 2012;7:537-542.
25. Kimura BJ, Shaw DJ, Amundson SA, et al. Cardiac limited ultrasound examination techniques to augment the bedside cardiac physical examination. J Ultrasound Med. 2015;34:1683-1690.
26. Brennan JM, Ronan A, Goonewardena S, et al. Handcarried ultrasound measurement of the inferior vena cava for assessment of intravascular volume status in the outpatient hemodialysis clinic. Clin J Am Soc Nephrol. 2006;1:749-753.
27. Goonewardena SN, Gemignani A, Ronan A, et al. Comparison of hand-carried ultrasound assessment of the inferior vena cava and N-terminal pro-brain natriuretic peptide for predicting readmission after hospitalization for acute decompensated heart failure. JACC Cardiovasc Imaging. 2008;1:595-601.
28. Wong CL, Holroyd-Leduc J, Straus SE. Does this patient have a pleural effusion? JAMA. 2009;301:309-317.
29. Blackmore CC, Black WC, Dallas RV, et al. Pleural fluid volume estimation: a chest radiograph prediction rule. Acad Radiol. 1996;3:103-109.
30. Kitazono MT, Lau CT, Parada AN, et al. Differentiation of pleural effusions from parenchymal opacities: accuracy of bedside chest radiography. Am J Roentgenol. 2010;194:407-412.
31. Kalokairinou-Motogna M, Maratou K, Paianid I, et al. Application of color Doppler ultrasound in the study of small pleural effusion. Med Ultrason. 2010;12:12-16.
32. Yousefifard M, Baikpour M, Ghelichkhani P, et al. Screening performance characteristic of ultrasonography and radiography in detection of pleural effusion; a meta-analysis. Emerg (Tehran, Iran). 2016;4:1-10.
33. Begot E, Grumann A, Duvoid T, et al. Ultrasonographic identification and semiquantitative assessment of unloculated pleural effusions in critically ill patients by residents after a focused training. Intensive Care Med. 2014;40:1475-1480.
34. World Health Organization. Pneumonia. Fact Sheet No. 331. Available at: http://www.who.int/mediacentre/factsheets/fs331/en/. Accessed June 26, 2017.
35. Gereige RS, Laufer PM. Pneumonia. Pediatr Rev. 2013;34:438-456.
36. National Center for Health Statistics. Leading causes of death. https://www.cdc.gov/nchs/fastats/leading-causes-of-death.htm. Accessed July 2, 2017.
37. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-S72.
38. Pereda MA, Chavez MA, Hooper-Miele CC, et al. Lung ultrasound for the diagnosis of pneumonia in children: a meta-analysis. Pediatrics. 2015;135:714-722.
39. Xia Y, Ying Y, Wang S, et al. Effectiveness of lung ultrasonography for diagnosis of pneumonia in adults: a systematic review and meta-analysis. J Thorac Dis. 2016;8:2822-2831.
40. Nazerian P, Volpicelli G, Vanni S, et al. Accuracy of lung ultrasound for the diagnosis of consolidations when compared to chest computed tomography. Am J Emerg Med. 2015;33:620-625.
41. Filopei J, Siedenburg H, Rattner P, et al. Impact of pocket ultrasound use by internal medicine housestaff in the diagnosis of dyspnea. J Hosp Med. 2014;9:594-597.
42. Lichtenstein D, Mezière G. A lung ultrasound sign allowing bedside distinction between pulmonary edema and COPD: the comet-tail artifact. Intensive Care Med. 1998;24:1331-1334.
43. Gargani L, Volpicelli G. How I do it: lung ultrasound. Cardiovasc Ultrasound. 2014;12:25.
44. Martindale JL, Wakai A, Collins SP, et al. Diagnosing acute heart failure in the emergency department: a systematic review and meta-analysis. Acad Emerg Med. 2016;23:223-242.
45. Volpicelli G, Mussa A, Garofalo G, et al. Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. Am J Emerg Med. 2006;24:689-696.
46. Picano E, Frassi F, Agricola E, et al. Ultrasound lung comets: a clinically useful sign of extravascular lung water. J Am Soc Echocardiogr. 2006;19:356-363.
47. Noble VE, Murray AF, Capp R, et al. Ultrasound assessment for extravascular lung water in patients undergoing hemodialysis: time course for resolution. Chest. 2009;135:1433-1439.
48. Gullett J, Donnelly JP, Sinert R, et al. Interobserver agreement in the evaluation of B-lines using bedside ultrasound. J Crit Care. 2015;30:1395-1399.
49. Guirguis-Blake JM, Beil TL, Sun X, et al. Primary Care Screening for Abdominal Aortic Aneurysm: A Systematic Evidence Review for the U.S. Preventive Services Task Force. Evidence Syntheses No. 109. Rockville, MD; 2014.
50. Metcalfe D, Holt PJE, Thompson MM. The management of abdominal aortic aneurysms. BMJ. 2011;342:d1384.
51. Thompson SG, Ashton HA, Gao L, et al. Final follow-up of the Multicentre Aneurysm Screening Study (MASS) randomized trial of abdominal aortic aneurysm screening. Brit J Surg. 2012;99:1649-1656.
52. LeFevre ML. Screening for abdominal aortic aneurysm: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:281-290.
53. Lindholt JS, Vammen S, Juul S, et al. The validity of ultrasonographic scanning as screening method for abdominal aortic aneurysm. Eur J Vasc Endovasc Surg. 1999;17:472-475.
54. Rubano E, Mehta N, Caputo W, et al. Systematic review: emergency department bedside ultrasonography for diagnosing suspected abdominal aortic aneurysm. Acad Emerg Med. 2013;20:128-138.
55. Blois B. Office-based ultrasound screening for abdominal aortic aneurysm. Can Fam Physician. 2012;58:e172-e178.
56. Sisó-Almirall A, Gilabert Solé R, Bru Saumell C, et al. Feasibility of hand-held-ultrasonography in the screening of abdominal aortic aneurysms and abdominal aortic atherosclerosis. Med Clin (Barc). 2013;141:417-422.
57. Sisó-Almirall A, Kostov B, Navarro González M, et al. Abdominal aortic aneurysm screening program using hand-held ultrasound in primary healthcare. PLoS One. 2017;12:e0176877.
58. Philbrick JT, Becker DM. Calf deep venous thrombosis: a wolf in sheep’s clothing? Arch Intern Med. 1988;148:2131-2138.
59. Bates SM, Jaeschke R, Stevens SM, et al. Diagnosis of DVT: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e351S-418S.
60. Cushman M, Tsai AW, White RH, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19-25.
61. Wells PS, Anderson DR, Rodger M, et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 2003;349:1227-1235.
62. Lensing AW, Prandoni P, Brandjes D, et al. Detection of deep-vein thrombosis by real-time B-mode ultrasonography. N Engl J Med. 1989;320:342-345.
63. Crisp JG, Lovato LM, Jang TB. Compression ultrasonography of the lower extremity with portable vascular ultrasonography can accurately detect deep venous thrombosis in the emergency department. Ann Emerg Med. 2010;56:601-610.
64. Blaivas M, Lambert MJ, Harwood RA, et al. Lower-extremity doppler for deep venous thrombosis—can emergency physicians be accurate and fast? Acad Emerg Med. 2000;7:120-126.
65. Burnside PR, Brown MD, Kline JA. Systematic review of emergency physician-performed ultrasonography for lower-extremity deep vein thrombosis. Acad Emerg Med. 2008;15:493-498.
66. Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350:1795-1798.
67. Birdwell BG, Raskob GE, Whitsett TL, et al. The clinical validity of normal compression ultrasonography in outpatients suspected of having deep venous thrombosis. Ann Intern Med. 1998;128:1-7.
68. Cogo A, Lensing AW, Koopman MM, et al. Compression ultrasonography for diagnostic management of patients with clinically suspected deep vein thrombosis: prospective cohort study. BMJ. 1998;316:17-20.
69. Tick LW, Ton E, Van Voorthuizen T, et al. Practical diagnostic management of patients with clinically suspected deep vein thrombosis by clinical probability test, compression ultrasonography, and D-dimer test. Am J Med. 2002;113:630-635.
70. Stevens