User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Flu activity continues to decline
The 2017-2018 flu season continued to loosen its grip on the country as both outpatient activity and pediatric deaths dropped during the week ending March 3, according to the Centers for Disease Control and Prevention.
After five consecutive weeks of double-digit pediatric deaths related to influenza-like illness (ILI), five deaths were reported for the week ending March 3, four of which occurred in previous weeks. The total for the 2017-2018 season is now 119, the CDC said in its weekly surveillance report.
The proportion of outpatient visits for ILI was 3.7% for the week, which is down from 4.9% the week before and less than half of the seasonal high of 7.5% that was recorded for the week of Feb. 3, CDC data show. The national baseline level of outpatient activity is 2.2%.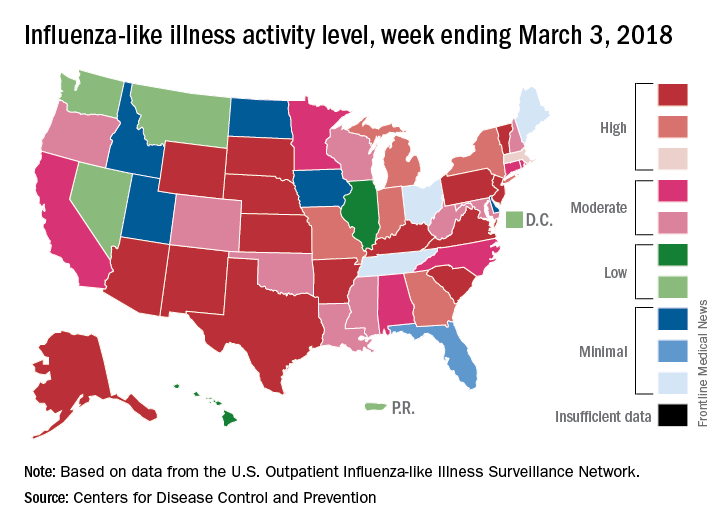
The cumulative hospitalization rate for the 2017-2018 flu season climbed from 84.2 the previous week to 86.3 per 100,000 population – well above the rate of 57.2 per 100,000 that was recorded for the corresponding week of the hospitalization-record-setting 2014-2015 season, FluView data show.
The 2017-2018 flu season continued to loosen its grip on the country as both outpatient activity and pediatric deaths dropped during the week ending March 3, according to the Centers for Disease Control and Prevention.
After five consecutive weeks of double-digit pediatric deaths related to influenza-like illness (ILI), five deaths were reported for the week ending March 3, four of which occurred in previous weeks. The total for the 2017-2018 season is now 119, the CDC said in its weekly surveillance report.
The proportion of outpatient visits for ILI was 3.7% for the week, which is down from 4.9% the week before and less than half of the seasonal high of 7.5% that was recorded for the week of Feb. 3, CDC data show. The national baseline level of outpatient activity is 2.2%.
The cumulative hospitalization rate for the 2017-2018 flu season climbed from 84.2 the previous week to 86.3 per 100,000 population – well above the rate of 57.2 per 100,000 that was recorded for the corresponding week of the hospitalization-record-setting 2014-2015 season, FluView data show.
The 2017-2018 flu season continued to loosen its grip on the country as both outpatient activity and pediatric deaths dropped during the week ending March 3, according to the Centers for Disease Control and Prevention.
After five consecutive weeks of double-digit pediatric deaths related to influenza-like illness (ILI), five deaths were reported for the week ending March 3, four of which occurred in previous weeks. The total for the 2017-2018 season is now 119, the CDC said in its weekly surveillance report.
The proportion of outpatient visits for ILI was 3.7% for the week, which is down from 4.9% the week before and less than half of the seasonal high of 7.5% that was recorded for the week of Feb. 3, CDC data show. The national baseline level of outpatient activity is 2.2%.
The cumulative hospitalization rate for the 2017-2018 flu season climbed from 84.2 the previous week to 86.3 per 100,000 population – well above the rate of 57.2 per 100,000 that was recorded for the corresponding week of the hospitalization-record-setting 2014-2015 season, FluView data show.
Peanut is most prevalent culprit in anaphylaxis PICU admits
ORLANDO – Food was found to be the most commonly identified trigger, with peanuts the most prevalent food cause, in what researchers say is the largest comprehensive review of anaphylaxis episodes in North America that led to pediatric intensive-care unit stays.
Researchers examined the Virtual Pediatrics Systems database, an international database of pediatric intensive care unit (PICU) information, said Carla M. Davis, MD, a pediatrician at Baylor College of Medicine, Houston. During 2010-2015, there were 1,989 pediatric anaphylaxis admissions to these units in North America, she reported at the joint congress of the American Academy of Allergy, Asthma and Immunology and the World Asthma Organization.
“Because anaphylaxis is one of the most severe consequences of allergic disease, we decided that this study needed to be done to see really what the landscape was in the most critically ill children,” she said.
Peanuts accounted for 45% of the food triggers, followed by tree nuts and seeds at 19%, and milk at 10%.
Common causes aside from food included drug, blood products, and venom, Dr. Davis said.
Anaphylaxis accounted for 0.3% of all PICU admissions over the 5-year period, researchers found. Dr. Davis said this was “higher than what we anticipated.”
The overall mortality rate was 1%, and researchers found that peanuts and dairy were main causes of death of all the food-induced cases.
Anaphylaxis occurred more often in children ages 6-18 years than in kids of other ages and was least common among those aged 2-5 years. Asian children were disproportionately represented among the PICU anaphylaxis patients, but the mortality rate didn’t vary by any demographic factors.
Admissions were most likely to happen in the fall and were more common in the Northeast and Western regions of the United States, Dr. Davis reported.
She said the deep look at the causes of these severe cases should help drive home the importance of counseling patients and families about prevention.
“For patients that have had a history of an allergic reaction to food or medication, but specifically food, I think really stressing avoidance measures will be something that will be very helpful, as well as counseling about epinephrine injectors and carrying them is going to help,” she said. “I think having a little more knowledge, pediatricians should be able to counsel and refer to allergists when they don’t feel they have all the necessary skills.”
Dr. Davis reports financial relationships with Aimmune Therapeutics and DBV Technologies.
SOURCE: Davis CM et al. 2018 AAAAI/WAO Joint Congress Abstract 775.
ORLANDO – Food was found to be the most commonly identified trigger, with peanuts the most prevalent food cause, in what researchers say is the largest comprehensive review of anaphylaxis episodes in North America that led to pediatric intensive-care unit stays.
Researchers examined the Virtual Pediatrics Systems database, an international database of pediatric intensive care unit (PICU) information, said Carla M. Davis, MD, a pediatrician at Baylor College of Medicine, Houston. During 2010-2015, there were 1,989 pediatric anaphylaxis admissions to these units in North America, she reported at the joint congress of the American Academy of Allergy, Asthma and Immunology and the World Asthma Organization.
“Because anaphylaxis is one of the most severe consequences of allergic disease, we decided that this study needed to be done to see really what the landscape was in the most critically ill children,” she said.
Peanuts accounted for 45% of the food triggers, followed by tree nuts and seeds at 19%, and milk at 10%.
Common causes aside from food included drug, blood products, and venom, Dr. Davis said.
Anaphylaxis accounted for 0.3% of all PICU admissions over the 5-year period, researchers found. Dr. Davis said this was “higher than what we anticipated.”
The overall mortality rate was 1%, and researchers found that peanuts and dairy were main causes of death of all the food-induced cases.
Anaphylaxis occurred more often in children ages 6-18 years than in kids of other ages and was least common among those aged 2-5 years. Asian children were disproportionately represented among the PICU anaphylaxis patients, but the mortality rate didn’t vary by any demographic factors.
Admissions were most likely to happen in the fall and were more common in the Northeast and Western regions of the United States, Dr. Davis reported.
She said the deep look at the causes of these severe cases should help drive home the importance of counseling patients and families about prevention.
“For patients that have had a history of an allergic reaction to food or medication, but specifically food, I think really stressing avoidance measures will be something that will be very helpful, as well as counseling about epinephrine injectors and carrying them is going to help,” she said. “I think having a little more knowledge, pediatricians should be able to counsel and refer to allergists when they don’t feel they have all the necessary skills.”
Dr. Davis reports financial relationships with Aimmune Therapeutics and DBV Technologies.
SOURCE: Davis CM et al. 2018 AAAAI/WAO Joint Congress Abstract 775.
ORLANDO – Food was found to be the most commonly identified trigger, with peanuts the most prevalent food cause, in what researchers say is the largest comprehensive review of anaphylaxis episodes in North America that led to pediatric intensive-care unit stays.
Researchers examined the Virtual Pediatrics Systems database, an international database of pediatric intensive care unit (PICU) information, said Carla M. Davis, MD, a pediatrician at Baylor College of Medicine, Houston. During 2010-2015, there were 1,989 pediatric anaphylaxis admissions to these units in North America, she reported at the joint congress of the American Academy of Allergy, Asthma and Immunology and the World Asthma Organization.
“Because anaphylaxis is one of the most severe consequences of allergic disease, we decided that this study needed to be done to see really what the landscape was in the most critically ill children,” she said.
Peanuts accounted for 45% of the food triggers, followed by tree nuts and seeds at 19%, and milk at 10%.
Common causes aside from food included drug, blood products, and venom, Dr. Davis said.
Anaphylaxis accounted for 0.3% of all PICU admissions over the 5-year period, researchers found. Dr. Davis said this was “higher than what we anticipated.”
The overall mortality rate was 1%, and researchers found that peanuts and dairy were main causes of death of all the food-induced cases.
Anaphylaxis occurred more often in children ages 6-18 years than in kids of other ages and was least common among those aged 2-5 years. Asian children were disproportionately represented among the PICU anaphylaxis patients, but the mortality rate didn’t vary by any demographic factors.
Admissions were most likely to happen in the fall and were more common in the Northeast and Western regions of the United States, Dr. Davis reported.
She said the deep look at the causes of these severe cases should help drive home the importance of counseling patients and families about prevention.
“For patients that have had a history of an allergic reaction to food or medication, but specifically food, I think really stressing avoidance measures will be something that will be very helpful, as well as counseling about epinephrine injectors and carrying them is going to help,” she said. “I think having a little more knowledge, pediatricians should be able to counsel and refer to allergists when they don’t feel they have all the necessary skills.”
Dr. Davis reports financial relationships with Aimmune Therapeutics and DBV Technologies.
SOURCE: Davis CM et al. 2018 AAAAI/WAO Joint Congress Abstract 775.
REPORTING FROM AAAAI/WAO JOINT CONGRESS
Key clinical point:
Major finding: Researchers found that 45% of these PICU stays from food were caused by reactions to peanuts.
Study details: A review of 1,989 cases during 2010-2015 in the Virtual Pediatrics Systems database, which collects international PICU information.
Disclosures: Dr. Davis reports financial relationships with Aimmune Therapeutics and DBV Technologies.
Source: Davis CM et al. 2018 AAAAI/WAO Joint Congress, Abstract 775.
Phosphodiesterase-5 inhibitors often prescribed inappropriately
While most veterans with pulmonary hypertension are treated in accordance with clinical guidelines, almost two-thirds who are prescribed therapy are being treated with pulmonary vasodilators inappropriately, an analysis of veteran prescription data reveals.
Little was known about how pulmonary vasodilators were used in practice prior to the publication of this study. While pulmonary vasodilators are considered effective for group 1 pulmonary hypertension (PH), clinical guidelines and advice from the Choosing Wisely campaign recommend against their routine use for PH patients classified into the most common types of PH – groups 2 and 3 – because of a lack of benefit, potential for harm, and high cost, the authors wrote. The report was published in Annals of the American Thoracic Society.
The new analysis shows that patients with PH are potentially being exposed to unnecessary harm, according to study author Renda Soylemez Wiener, MD, MPH, of the Center for Healthcare Organization & Implementation Research at Bedford (Mass.) Veterans Affairs Medical Center, and her colleagues. Their findings also reveal that inappropriate prescribing of pulmonary vasodilators, mostly by specialist clinicians, is contributing to the financial burden of an already stretched health system.
The research team looked at prescription data for veterans prescribed a phosphodiesterase-5 inhibitor (PDE5i), which causes pulmonary vasodilation, between 2005 and 2012 at any VA site. The primary outcome of the study was the proportion of patients who received potentially inappropriate PDE5i as classified in guideline recommendations. Patients with group 1 PH were deemed to have been treated appropriately, while those with group 2 and 3 PH were deemed to have been potentially treated inappropriately. Those with groups 4 and 5 PH were thought to have received treatment of “uncertain value.”
In a chart abstraction analysis from a randomly selected subset of PDE5i-treated patients, half (110/230, 47.8% [41.3%-54.5%]) had documented right heart catheterization to confirm the presence of PH. After factoring this into their algorithm, the investigators determined that only 11.7% [8.0%-16.8%] of these patients received clearly appropriate treatment.
Over the 8-year study period, the number of patients with PH group 2 or 3 prescribed PDE5i rose more than 14-fold, the researchers said. They speculated that this figure was likely to continue to rise with the increasing use of echocardiography and detection of PH.
According to the authors, the cost of treating one PH patient for 1 year with PDE5i therapy was between $10,000 and $13,000.
The 1,711 PH patients classified as being treated inappropriately in the study translated into a cost of over $20 million, if each patient were treated for only 1 year, but many of the patients were treated for a longer period of time.
The researchers suggested that there were several reasons why clinicians might choose to deviate from the guidelines, including lacking familiarity with them or disagreeing with them.
“While guidelines do allow trials of PDE5i in treatment for groups 2 or 3 PH on a case-by-case basis after consultation with a PH expert and a confirmatory [right heart catheterization], even PH experts disagree about whether a trial of PDE5i therapy is reasonable and appropriate for patients with group 3 PH,” they wrote.
They may also overestimate the potential benefits of treatment and/or underestimate potential harm.
Clinicians may believe that guidelines developed for a general population do not apply to the patients they are treating.
“It is understandable why clinicians may offer unproven therapies like PDE5i in hopes of providing relief to very sick patients with groups 2 or 3 PH, especially if they do not believe the recommendation applies to their individual patient or they are not convinced about the potential harms of pulmonary vasodilators,” they said.
The authors expressed concern about VA clinicians’ allowing patients to take PDE5i therapy that had been initially prescribed by clinicians outside of VA hospitals. The researchers said such drugs, which potentially had been prescribed inappropriately, “were continued by VA clinicians without much apparent scrutiny.”
The chart abstraction analysis also showed that specialists prescribed the majority of potentially inappropriate PDE5i treatment, suggesting “that other interventions to prevent inappropriate use may be required.”
The researchers concluded that “[the] time has come to develop interventions to optimize prescribing for PH in order to improve the value, quality, and safety of care.”
One potential intervention suggested by the researchers was to require patients with PH to be evaluated at a PH expert center, as recommended by treatment guidelines.
The study was funded by the Department of Veterans Affairs with resources from the Edith Nourse Rogers Memorial VA Hospital. Elizabeth S. Klings, MD, one of the study’s authors, declared receiving research support from several pharmaceutical companies.
SOURCE: Wiener RS et al. Ann Am Thorac Soc. 2018 Feb 27. doi: 10.1513/AnnalsATS.201710-762OC.
While most veterans with pulmonary hypertension are treated in accordance with clinical guidelines, almost two-thirds who are prescribed therapy are being treated with pulmonary vasodilators inappropriately, an analysis of veteran prescription data reveals.
Little was known about how pulmonary vasodilators were used in practice prior to the publication of this study. While pulmonary vasodilators are considered effective for group 1 pulmonary hypertension (PH), clinical guidelines and advice from the Choosing Wisely campaign recommend against their routine use for PH patients classified into the most common types of PH – groups 2 and 3 – because of a lack of benefit, potential for harm, and high cost, the authors wrote. The report was published in Annals of the American Thoracic Society.
The new analysis shows that patients with PH are potentially being exposed to unnecessary harm, according to study author Renda Soylemez Wiener, MD, MPH, of the Center for Healthcare Organization & Implementation Research at Bedford (Mass.) Veterans Affairs Medical Center, and her colleagues. Their findings also reveal that inappropriate prescribing of pulmonary vasodilators, mostly by specialist clinicians, is contributing to the financial burden of an already stretched health system.
The research team looked at prescription data for veterans prescribed a phosphodiesterase-5 inhibitor (PDE5i), which causes pulmonary vasodilation, between 2005 and 2012 at any VA site. The primary outcome of the study was the proportion of patients who received potentially inappropriate PDE5i as classified in guideline recommendations. Patients with group 1 PH were deemed to have been treated appropriately, while those with group 2 and 3 PH were deemed to have been potentially treated inappropriately. Those with groups 4 and 5 PH were thought to have received treatment of “uncertain value.”
In a chart abstraction analysis from a randomly selected subset of PDE5i-treated patients, half (110/230, 47.8% [41.3%-54.5%]) had documented right heart catheterization to confirm the presence of PH. After factoring this into their algorithm, the investigators determined that only 11.7% [8.0%-16.8%] of these patients received clearly appropriate treatment.
Over the 8-year study period, the number of patients with PH group 2 or 3 prescribed PDE5i rose more than 14-fold, the researchers said. They speculated that this figure was likely to continue to rise with the increasing use of echocardiography and detection of PH.
According to the authors, the cost of treating one PH patient for 1 year with PDE5i therapy was between $10,000 and $13,000.
The 1,711 PH patients classified as being treated inappropriately in the study translated into a cost of over $20 million, if each patient were treated for only 1 year, but many of the patients were treated for a longer period of time.
The researchers suggested that there were several reasons why clinicians might choose to deviate from the guidelines, including lacking familiarity with them or disagreeing with them.
“While guidelines do allow trials of PDE5i in treatment for groups 2 or 3 PH on a case-by-case basis after consultation with a PH expert and a confirmatory [right heart catheterization], even PH experts disagree about whether a trial of PDE5i therapy is reasonable and appropriate for patients with group 3 PH,” they wrote.
They may also overestimate the potential benefits of treatment and/or underestimate potential harm.
Clinicians may believe that guidelines developed for a general population do not apply to the patients they are treating.
“It is understandable why clinicians may offer unproven therapies like PDE5i in hopes of providing relief to very sick patients with groups 2 or 3 PH, especially if they do not believe the recommendation applies to their individual patient or they are not convinced about the potential harms of pulmonary vasodilators,” they said.
The authors expressed concern about VA clinicians’ allowing patients to take PDE5i therapy that had been initially prescribed by clinicians outside of VA hospitals. The researchers said such drugs, which potentially had been prescribed inappropriately, “were continued by VA clinicians without much apparent scrutiny.”
The chart abstraction analysis also showed that specialists prescribed the majority of potentially inappropriate PDE5i treatment, suggesting “that other interventions to prevent inappropriate use may be required.”
The researchers concluded that “[the] time has come to develop interventions to optimize prescribing for PH in order to improve the value, quality, and safety of care.”
One potential intervention suggested by the researchers was to require patients with PH to be evaluated at a PH expert center, as recommended by treatment guidelines.
The study was funded by the Department of Veterans Affairs with resources from the Edith Nourse Rogers Memorial VA Hospital. Elizabeth S. Klings, MD, one of the study’s authors, declared receiving research support from several pharmaceutical companies.
SOURCE: Wiener RS et al. Ann Am Thorac Soc. 2018 Feb 27. doi: 10.1513/AnnalsATS.201710-762OC.
While most veterans with pulmonary hypertension are treated in accordance with clinical guidelines, almost two-thirds who are prescribed therapy are being treated with pulmonary vasodilators inappropriately, an analysis of veteran prescription data reveals.
Little was known about how pulmonary vasodilators were used in practice prior to the publication of this study. While pulmonary vasodilators are considered effective for group 1 pulmonary hypertension (PH), clinical guidelines and advice from the Choosing Wisely campaign recommend against their routine use for PH patients classified into the most common types of PH – groups 2 and 3 – because of a lack of benefit, potential for harm, and high cost, the authors wrote. The report was published in Annals of the American Thoracic Society.
The new analysis shows that patients with PH are potentially being exposed to unnecessary harm, according to study author Renda Soylemez Wiener, MD, MPH, of the Center for Healthcare Organization & Implementation Research at Bedford (Mass.) Veterans Affairs Medical Center, and her colleagues. Their findings also reveal that inappropriate prescribing of pulmonary vasodilators, mostly by specialist clinicians, is contributing to the financial burden of an already stretched health system.
The research team looked at prescription data for veterans prescribed a phosphodiesterase-5 inhibitor (PDE5i), which causes pulmonary vasodilation, between 2005 and 2012 at any VA site. The primary outcome of the study was the proportion of patients who received potentially inappropriate PDE5i as classified in guideline recommendations. Patients with group 1 PH were deemed to have been treated appropriately, while those with group 2 and 3 PH were deemed to have been potentially treated inappropriately. Those with groups 4 and 5 PH were thought to have received treatment of “uncertain value.”
In a chart abstraction analysis from a randomly selected subset of PDE5i-treated patients, half (110/230, 47.8% [41.3%-54.5%]) had documented right heart catheterization to confirm the presence of PH. After factoring this into their algorithm, the investigators determined that only 11.7% [8.0%-16.8%] of these patients received clearly appropriate treatment.
Over the 8-year study period, the number of patients with PH group 2 or 3 prescribed PDE5i rose more than 14-fold, the researchers said. They speculated that this figure was likely to continue to rise with the increasing use of echocardiography and detection of PH.
According to the authors, the cost of treating one PH patient for 1 year with PDE5i therapy was between $10,000 and $13,000.
The 1,711 PH patients classified as being treated inappropriately in the study translated into a cost of over $20 million, if each patient were treated for only 1 year, but many of the patients were treated for a longer period of time.
The researchers suggested that there were several reasons why clinicians might choose to deviate from the guidelines, including lacking familiarity with them or disagreeing with them.
“While guidelines do allow trials of PDE5i in treatment for groups 2 or 3 PH on a case-by-case basis after consultation with a PH expert and a confirmatory [right heart catheterization], even PH experts disagree about whether a trial of PDE5i therapy is reasonable and appropriate for patients with group 3 PH,” they wrote.
They may also overestimate the potential benefits of treatment and/or underestimate potential harm.
Clinicians may believe that guidelines developed for a general population do not apply to the patients they are treating.
“It is understandable why clinicians may offer unproven therapies like PDE5i in hopes of providing relief to very sick patients with groups 2 or 3 PH, especially if they do not believe the recommendation applies to their individual patient or they are not convinced about the potential harms of pulmonary vasodilators,” they said.
The authors expressed concern about VA clinicians’ allowing patients to take PDE5i therapy that had been initially prescribed by clinicians outside of VA hospitals. The researchers said such drugs, which potentially had been prescribed inappropriately, “were continued by VA clinicians without much apparent scrutiny.”
The chart abstraction analysis also showed that specialists prescribed the majority of potentially inappropriate PDE5i treatment, suggesting “that other interventions to prevent inappropriate use may be required.”
The researchers concluded that “[the] time has come to develop interventions to optimize prescribing for PH in order to improve the value, quality, and safety of care.”
One potential intervention suggested by the researchers was to require patients with PH to be evaluated at a PH expert center, as recommended by treatment guidelines.
The study was funded by the Department of Veterans Affairs with resources from the Edith Nourse Rogers Memorial VA Hospital. Elizabeth S. Klings, MD, one of the study’s authors, declared receiving research support from several pharmaceutical companies.
SOURCE: Wiener RS et al. Ann Am Thorac Soc. 2018 Feb 27. doi: 10.1513/AnnalsATS.201710-762OC.
FROM ANNALS OF THE AMERICAN THORACIC SOCIETY
Key clinical point: Inappropriate prescribing of phosphodiesterase-5 inhibitor (PDE5i) therapy has risen 14-fold in 8 years, at a cost of $20 million per year.
Major finding:
Study details: A retrospective analysis of veterans prescribed phosphodiesterase-5 inhibitors for pulmonary hypertension between 2005 and 2012.
Disclosures: The study was funded by the Department of Veterans Affairs with resources from the Edith Nourse Rogers Memorial Veterans Hospital. Elizabeth S. Klings, MD, one of the study’s authors, declared receiving research support from several pharmaceutical companies.
Source: Wiener RS et al. Ann Am Thorac Soc. 2018 Feb 27. doi: 10.1513/AnnalsATS.201710-762OC.
Climate change is worsening allergies, expert says
ORLANDO – Climate change is not just eroding coastlines and threatening seaside cities and taking lives with increasingly powerful hurricanes, but appears to be contributing to increases in allergy and asthma, an expert told the audience at the joint congress of the American Academy of Allergy, Asthma and Immunology and the World Asthma Organization.
Longer pollen seasons, allergens unleashed by felled trees and ripped-up plants, mold growth following floods, and irritants launched into the air by wildfires are some of the concerns that should be alarming physicians and policy makers, said Nelson A. Rosario, MD, PhD, professor of pediatrics at Federal University of Paraná (Brazil).
And evidence suggests that his fellow allergists and their patients agree.
A 2015 international survey found that 80% of rhinitis patients blamed climate change for contributing to their symptoms.
Only 11% said that climate change wasn’t relevant at all. Asked how patients have been affected by climate change, about two-thirds said “increased care for allergic sensitization and symptoms on exposure to plants or mold.”
Science supports these views, Dr. Rosario said.
A 2011 study of North American pollen seasons found that some cities had significant increases of 11-27 days, compared with 15 years before.
And a 2017 review noted the impacts of the consequences of climate change, from increased allergies due to heavy precipitation events, asthma prompted by intense tropical cyclones, and allergic conditions caused by extremely high sea levels.
Dr. Rosario suggested that, rather than wait for official agencies to take action, physicians need to adapt and help their patients adapt. A team of doctors wrote in 2013 that while “improved governmental controls” could lead to cleaner air, they “meet strong opposition because of their effect on business and productivity.” So, they said, the allergy community should adjust, by “anticipating the needs of patients and by adopting practices and research methods to meet changing environmental conditions.”
Dr. Rosario urged physicians to think of the climate-change effects on allergy and asthma as a “collective action” problem, not an individual one.
“The consequences will come,” he said. “There must be international cooperation.”
ORLANDO – Climate change is not just eroding coastlines and threatening seaside cities and taking lives with increasingly powerful hurricanes, but appears to be contributing to increases in allergy and asthma, an expert told the audience at the joint congress of the American Academy of Allergy, Asthma and Immunology and the World Asthma Organization.
Longer pollen seasons, allergens unleashed by felled trees and ripped-up plants, mold growth following floods, and irritants launched into the air by wildfires are some of the concerns that should be alarming physicians and policy makers, said Nelson A. Rosario, MD, PhD, professor of pediatrics at Federal University of Paraná (Brazil).
And evidence suggests that his fellow allergists and their patients agree.
A 2015 international survey found that 80% of rhinitis patients blamed climate change for contributing to their symptoms.
Only 11% said that climate change wasn’t relevant at all. Asked how patients have been affected by climate change, about two-thirds said “increased care for allergic sensitization and symptoms on exposure to plants or mold.”
Science supports these views, Dr. Rosario said.
A 2011 study of North American pollen seasons found that some cities had significant increases of 11-27 days, compared with 15 years before.
And a 2017 review noted the impacts of the consequences of climate change, from increased allergies due to heavy precipitation events, asthma prompted by intense tropical cyclones, and allergic conditions caused by extremely high sea levels.
Dr. Rosario suggested that, rather than wait for official agencies to take action, physicians need to adapt and help their patients adapt. A team of doctors wrote in 2013 that while “improved governmental controls” could lead to cleaner air, they “meet strong opposition because of their effect on business and productivity.” So, they said, the allergy community should adjust, by “anticipating the needs of patients and by adopting practices and research methods to meet changing environmental conditions.”
Dr. Rosario urged physicians to think of the climate-change effects on allergy and asthma as a “collective action” problem, not an individual one.
“The consequences will come,” he said. “There must be international cooperation.”
ORLANDO – Climate change is not just eroding coastlines and threatening seaside cities and taking lives with increasingly powerful hurricanes, but appears to be contributing to increases in allergy and asthma, an expert told the audience at the joint congress of the American Academy of Allergy, Asthma and Immunology and the World Asthma Organization.
Longer pollen seasons, allergens unleashed by felled trees and ripped-up plants, mold growth following floods, and irritants launched into the air by wildfires are some of the concerns that should be alarming physicians and policy makers, said Nelson A. Rosario, MD, PhD, professor of pediatrics at Federal University of Paraná (Brazil).
And evidence suggests that his fellow allergists and their patients agree.
A 2015 international survey found that 80% of rhinitis patients blamed climate change for contributing to their symptoms.
Only 11% said that climate change wasn’t relevant at all. Asked how patients have been affected by climate change, about two-thirds said “increased care for allergic sensitization and symptoms on exposure to plants or mold.”
Science supports these views, Dr. Rosario said.
A 2011 study of North American pollen seasons found that some cities had significant increases of 11-27 days, compared with 15 years before.
And a 2017 review noted the impacts of the consequences of climate change, from increased allergies due to heavy precipitation events, asthma prompted by intense tropical cyclones, and allergic conditions caused by extremely high sea levels.
Dr. Rosario suggested that, rather than wait for official agencies to take action, physicians need to adapt and help their patients adapt. A team of doctors wrote in 2013 that while “improved governmental controls” could lead to cleaner air, they “meet strong opposition because of their effect on business and productivity.” So, they said, the allergy community should adjust, by “anticipating the needs of patients and by adopting practices and research methods to meet changing environmental conditions.”
Dr. Rosario urged physicians to think of the climate-change effects on allergy and asthma as a “collective action” problem, not an individual one.
“The consequences will come,” he said. “There must be international cooperation.”
EXPERT ANALYSIS FROM AAAAI/WAO JOINT CONGRESS
Flu activity takes another turn for the better
Outpatient influenza-like illness activity continues to drop, but pediatric deaths for 2017-2018 are already higher than either of the last two entire seasons, according to the Centers for Disease and Prevention.
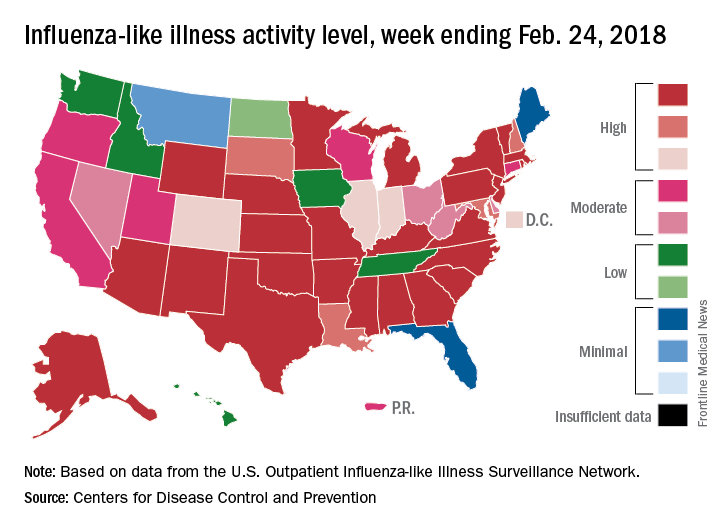
An additional 17 influenza-like illness-related (ILI) pediatric deaths were reported during the week ending Feb. 24, eight of which occurred in previous weeks. That brings the total to 114 for the 2017-2018 flu season so far, compared with 110 for the entire 2016-2017 season and 93 for the 2015-2016 season, the CDC reported Mar. 2.
The proportion of outpatient visits for ILI took another big drop, falling to 5.0% for the week, which was down from 6.4% the previous week and the seasonal high of 7.4% the 2 weeks before that (Feb. 10 and Feb. 3), CDC data show.
Flu-related hospitalizations, however, continued to rise to new highs, as the cumulative rate hit 81.7 per 100,000 population. In 2014-2015, the season with the highest number of hospitalizations since the CDC started keeping track, the cumulative rate for the corresponding week was 55.9 per 100,000, according to the CDC’s Fluview website.
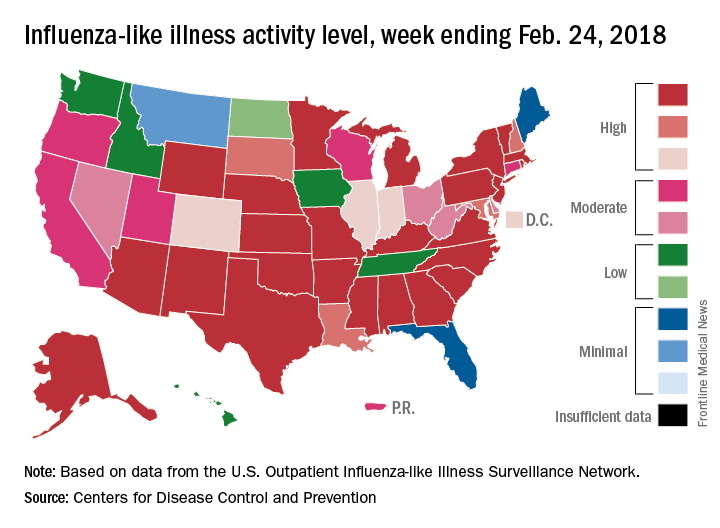
The map of state-reported ILI activity shows that 25 states are at level 10 on the CDC’s 1-10 scale, which is down from 33 the week before. Eight other states and the District of Columbia were in the “high” range with activity at levels 8 and 9 for the week ending Feb. 24, the CDC said.
Outpatient influenza-like illness activity continues to drop, but pediatric deaths for 2017-2018 are already higher than either of the last two entire seasons, according to the Centers for Disease and Prevention.

An additional 17 influenza-like illness-related (ILI) pediatric deaths were reported during the week ending Feb. 24, eight of which occurred in previous weeks. That brings the total to 114 for the 2017-2018 flu season so far, compared with 110 for the entire 2016-2017 season and 93 for the 2015-2016 season, the CDC reported Mar. 2.
The proportion of outpatient visits for ILI took another big drop, falling to 5.0% for the week, which was down from 6.4% the previous week and the seasonal high of 7.4% the 2 weeks before that (Feb. 10 and Feb. 3), CDC data show.
Flu-related hospitalizations, however, continued to rise to new highs, as the cumulative rate hit 81.7 per 100,000 population. In 2014-2015, the season with the highest number of hospitalizations since the CDC started keeping track, the cumulative rate for the corresponding week was 55.9 per 100,000, according to the CDC’s Fluview website.
The map of state-reported ILI activity shows that 25 states are at level 10 on the CDC’s 1-10 scale, which is down from 33 the week before. Eight other states and the District of Columbia were in the “high” range with activity at levels 8 and 9 for the week ending Feb. 24, the CDC said.
Outpatient influenza-like illness activity continues to drop, but pediatric deaths for 2017-2018 are already higher than either of the last two entire seasons, according to the Centers for Disease and Prevention.

An additional 17 influenza-like illness-related (ILI) pediatric deaths were reported during the week ending Feb. 24, eight of which occurred in previous weeks. That brings the total to 114 for the 2017-2018 flu season so far, compared with 110 for the entire 2016-2017 season and 93 for the 2015-2016 season, the CDC reported Mar. 2.
The proportion of outpatient visits for ILI took another big drop, falling to 5.0% for the week, which was down from 6.4% the previous week and the seasonal high of 7.4% the 2 weeks before that (Feb. 10 and Feb. 3), CDC data show.
Flu-related hospitalizations, however, continued to rise to new highs, as the cumulative rate hit 81.7 per 100,000 population. In 2014-2015, the season with the highest number of hospitalizations since the CDC started keeping track, the cumulative rate for the corresponding week was 55.9 per 100,000, according to the CDC’s Fluview website.
The map of state-reported ILI activity shows that 25 states are at level 10 on the CDC’s 1-10 scale, which is down from 33 the week before. Eight other states and the District of Columbia were in the “high” range with activity at levels 8 and 9 for the week ending Feb. 24, the CDC said.
App collects allergy symptoms in real time
ORLANDO – Use of an app combining patient-reported symptoms with local environmental triggers led patients to take action to improve their health, Penny Jones, PhD, reported at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization.
AirRater is a smartphone app and data collection network that includes information on air particulates, daily pollen and fungi counts, temperature, and planned burn locations. Patients enter their respiratory symptoms, which are correlated with local environmental conditions, according to Dr. Jones, a postdoctoral fellow at the University of Tasmania (Australia) in Hobart.
Most of the environmental data are gathered from government agencies; however, researchers collect pollen and fungi counts at their own stations.
Patients do not see the environmental data until they’ve logged in their symptoms so that their reports aren’t biased by that information, Dr. Jones said, adding that the app also sends notifications when pollen and pollutant levels are high.
“It’s an environmental monitoring system coupled with a smartphone app designed to help people with allergies and asthma make better decisions around their health,” Dr. Jones said.
The AirRater network and app are now operating in both Tasmania and Canberra, Australia.
There are more than 6,000 users, and data from surveys show that it is having an effect, Dr. Jones said. About 40% of users said they have changed their behavior in some way because of information provided by the app, including staying indoors, taking preventive medication, or speaking with their doctors.“It does appear that people are generally finding it a useful tool,” she said.
In a pilot study, researchers found that several environmental triggers were significantly correlated with exacerbation of patient symptoms, including maximum temperature (P < .001), particulate pollution (P < .001), relative humidity (P = .01), birch pollen (P = .006), and cypress pollen (P = .004).
Researchers plan to expand use of the network and app to other parts of Australia and are working to refine the understanding of aerobiological symptom drivers through DNA analysis of airborne particles. Their goal is to be able to identify personalized drivers of sensitivities, she said.
“We’ll keep working on this,” Dr. Jones said. “But we think that certainly has promise.”
The investigators reported no financial conflicts of interest, and the study had no outside funding.
SOURCE: Jones P et al. AAAAI/WAO Joint Congress, Abstract 270.
ORLANDO – Use of an app combining patient-reported symptoms with local environmental triggers led patients to take action to improve their health, Penny Jones, PhD, reported at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization.
AirRater is a smartphone app and data collection network that includes information on air particulates, daily pollen and fungi counts, temperature, and planned burn locations. Patients enter their respiratory symptoms, which are correlated with local environmental conditions, according to Dr. Jones, a postdoctoral fellow at the University of Tasmania (Australia) in Hobart.
Most of the environmental data are gathered from government agencies; however, researchers collect pollen and fungi counts at their own stations.
Patients do not see the environmental data until they’ve logged in their symptoms so that their reports aren’t biased by that information, Dr. Jones said, adding that the app also sends notifications when pollen and pollutant levels are high.
“It’s an environmental monitoring system coupled with a smartphone app designed to help people with allergies and asthma make better decisions around their health,” Dr. Jones said.
The AirRater network and app are now operating in both Tasmania and Canberra, Australia.
There are more than 6,000 users, and data from surveys show that it is having an effect, Dr. Jones said. About 40% of users said they have changed their behavior in some way because of information provided by the app, including staying indoors, taking preventive medication, or speaking with their doctors.“It does appear that people are generally finding it a useful tool,” she said.
In a pilot study, researchers found that several environmental triggers were significantly correlated with exacerbation of patient symptoms, including maximum temperature (P < .001), particulate pollution (P < .001), relative humidity (P = .01), birch pollen (P = .006), and cypress pollen (P = .004).
Researchers plan to expand use of the network and app to other parts of Australia and are working to refine the understanding of aerobiological symptom drivers through DNA analysis of airborne particles. Their goal is to be able to identify personalized drivers of sensitivities, she said.
“We’ll keep working on this,” Dr. Jones said. “But we think that certainly has promise.”
The investigators reported no financial conflicts of interest, and the study had no outside funding.
SOURCE: Jones P et al. AAAAI/WAO Joint Congress, Abstract 270.
ORLANDO – Use of an app combining patient-reported symptoms with local environmental triggers led patients to take action to improve their health, Penny Jones, PhD, reported at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization.
AirRater is a smartphone app and data collection network that includes information on air particulates, daily pollen and fungi counts, temperature, and planned burn locations. Patients enter their respiratory symptoms, which are correlated with local environmental conditions, according to Dr. Jones, a postdoctoral fellow at the University of Tasmania (Australia) in Hobart.
Most of the environmental data are gathered from government agencies; however, researchers collect pollen and fungi counts at their own stations.
Patients do not see the environmental data until they’ve logged in their symptoms so that their reports aren’t biased by that information, Dr. Jones said, adding that the app also sends notifications when pollen and pollutant levels are high.
“It’s an environmental monitoring system coupled with a smartphone app designed to help people with allergies and asthma make better decisions around their health,” Dr. Jones said.
The AirRater network and app are now operating in both Tasmania and Canberra, Australia.
There are more than 6,000 users, and data from surveys show that it is having an effect, Dr. Jones said. About 40% of users said they have changed their behavior in some way because of information provided by the app, including staying indoors, taking preventive medication, or speaking with their doctors.“It does appear that people are generally finding it a useful tool,” she said.
In a pilot study, researchers found that several environmental triggers were significantly correlated with exacerbation of patient symptoms, including maximum temperature (P < .001), particulate pollution (P < .001), relative humidity (P = .01), birch pollen (P = .006), and cypress pollen (P = .004).
Researchers plan to expand use of the network and app to other parts of Australia and are working to refine the understanding of aerobiological symptom drivers through DNA analysis of airborne particles. Their goal is to be able to identify personalized drivers of sensitivities, she said.
“We’ll keep working on this,” Dr. Jones said. “But we think that certainly has promise.”
The investigators reported no financial conflicts of interest, and the study had no outside funding.
SOURCE: Jones P et al. AAAAI/WAO Joint Congress, Abstract 270.
REPORTING FROM THE AAAAI/WAO JOINT CONGRESS
Key clinical point: A smartphone app has been developed to collect patient allergy and asthma symptoms and fuse that information with environmental data to find correlations.
Major finding: About 40% of app users said information provided by the app prompted them to take preventive action.
Study details: A survey of 6,000 app users and a retrospective study correlating reported allergy and asthma symptoms with real-time environmental data.
Disclosures: The investigators reported no financial conflicts of interest, and the study had no outside funding.
Source: Jones P et al. AAAAI/WAO Joint Congress, Abstract 270.
‘Modified rush’ immunotherapy delivers good results
ORLANDO – Patients with allergic rhinitis undergoing allergen immunotherapy achieved the goal of their monthly maintenance dose at a 20% higher rate when they underwent went a “modified rush” protocol, compared with those who received the therapy with the much slower conventional approach.
The findings offer proof that the faster approach is safe, more effective, and should be more widely used even though many allergists continue to be hesitant to do so, according to Christine Schafer, MD, clinical assistant professor at Michigan State University, East Lansing and a private practice allergist in Grand Rapids, Mich.
“The beauty of it is you’ve consolidated 3 months into a day,” said Dr. Schafer, who added that patients also feel better faster. “They don’t have to have this going every week for 3 months not feeling any better.”
Dr. Schafer and her colleagues reviewed a year’s worth of results for patients undergoing conventional immunotherapy or a “modified rush” protocol in 2014. They found that the conventional approach produced a 64.5% success rate, compared with an 84.4% success rate for those doing the faster protocol (P less than .001).
Typically, allergen immunotherapy takes 6 months, with patients making weekly visits with gradually increasing doses. The weekly visits can be a burden, and many patients drop out. The modified rush protocol seems to be easier for patients to accomplish, Dr. Schafer said at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization.
The study compared 231 patients on the faster protocol with 392 treated with the traditional approach.
On the “modified rush” protocol, patients aged 12-15 years took preventive medications – 20 mg of prednisone and 150 mg of ranitidine 2 days before their appointment and again the day after. In addition, they took 10 mg of cetirizine and 5 mg of montelukast 2 days before their appointment. Patients also took those same medications the morning of the treatment.
For patients 16 and older, the dose of prednisone was 30 mg and the dose of montelukast was 10 mg.
The protocol consisted of eight shots, starting at a more diluted dose than the conventional protocol, as an additional safety precaution. The shots were given first every 15 minutes, then every 30 minutes or an hour as the dose increases. Once the treatment was over, the dilution was 1:10, a milestone not reached until 3 months under the conventional approach.
An hour or two after the treatment, all patients received 20 mg of prednisone.
Patients resumed the normal conventional protocol after the first visit.
Reactions were rare under the “modified rush” protocol, with 6 patients experiencing flushing but nothing else, 21 with grade 1 reactions, 3 with grade 2, and no reactions worse than that. Five patients needed epinephrine.
Dr. Schafer said the approach may hold appeal because, even though it’s accelerated, there is a slower lead-in to the maintenance dose.
“If you ask allergists, ‘Do you rush?’ they’ll say, ‘No, no, no, I don’t rush. It’s too risky.’” she said. “Hence, modified rush.”
SOURCE: Schafer C et al. AAAAI/WAO Joint Congress abstract 520.
ORLANDO – Patients with allergic rhinitis undergoing allergen immunotherapy achieved the goal of their monthly maintenance dose at a 20% higher rate when they underwent went a “modified rush” protocol, compared with those who received the therapy with the much slower conventional approach.
The findings offer proof that the faster approach is safe, more effective, and should be more widely used even though many allergists continue to be hesitant to do so, according to Christine Schafer, MD, clinical assistant professor at Michigan State University, East Lansing and a private practice allergist in Grand Rapids, Mich.
“The beauty of it is you’ve consolidated 3 months into a day,” said Dr. Schafer, who added that patients also feel better faster. “They don’t have to have this going every week for 3 months not feeling any better.”
Dr. Schafer and her colleagues reviewed a year’s worth of results for patients undergoing conventional immunotherapy or a “modified rush” protocol in 2014. They found that the conventional approach produced a 64.5% success rate, compared with an 84.4% success rate for those doing the faster protocol (P less than .001).
Typically, allergen immunotherapy takes 6 months, with patients making weekly visits with gradually increasing doses. The weekly visits can be a burden, and many patients drop out. The modified rush protocol seems to be easier for patients to accomplish, Dr. Schafer said at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization.
The study compared 231 patients on the faster protocol with 392 treated with the traditional approach.
On the “modified rush” protocol, patients aged 12-15 years took preventive medications – 20 mg of prednisone and 150 mg of ranitidine 2 days before their appointment and again the day after. In addition, they took 10 mg of cetirizine and 5 mg of montelukast 2 days before their appointment. Patients also took those same medications the morning of the treatment.
For patients 16 and older, the dose of prednisone was 30 mg and the dose of montelukast was 10 mg.
The protocol consisted of eight shots, starting at a more diluted dose than the conventional protocol, as an additional safety precaution. The shots were given first every 15 minutes, then every 30 minutes or an hour as the dose increases. Once the treatment was over, the dilution was 1:10, a milestone not reached until 3 months under the conventional approach.
An hour or two after the treatment, all patients received 20 mg of prednisone.
Patients resumed the normal conventional protocol after the first visit.
Reactions were rare under the “modified rush” protocol, with 6 patients experiencing flushing but nothing else, 21 with grade 1 reactions, 3 with grade 2, and no reactions worse than that. Five patients needed epinephrine.
Dr. Schafer said the approach may hold appeal because, even though it’s accelerated, there is a slower lead-in to the maintenance dose.
“If you ask allergists, ‘Do you rush?’ they’ll say, ‘No, no, no, I don’t rush. It’s too risky.’” she said. “Hence, modified rush.”
SOURCE: Schafer C et al. AAAAI/WAO Joint Congress abstract 520.
ORLANDO – Patients with allergic rhinitis undergoing allergen immunotherapy achieved the goal of their monthly maintenance dose at a 20% higher rate when they underwent went a “modified rush” protocol, compared with those who received the therapy with the much slower conventional approach.
The findings offer proof that the faster approach is safe, more effective, and should be more widely used even though many allergists continue to be hesitant to do so, according to Christine Schafer, MD, clinical assistant professor at Michigan State University, East Lansing and a private practice allergist in Grand Rapids, Mich.
“The beauty of it is you’ve consolidated 3 months into a day,” said Dr. Schafer, who added that patients also feel better faster. “They don’t have to have this going every week for 3 months not feeling any better.”
Dr. Schafer and her colleagues reviewed a year’s worth of results for patients undergoing conventional immunotherapy or a “modified rush” protocol in 2014. They found that the conventional approach produced a 64.5% success rate, compared with an 84.4% success rate for those doing the faster protocol (P less than .001).
Typically, allergen immunotherapy takes 6 months, with patients making weekly visits with gradually increasing doses. The weekly visits can be a burden, and many patients drop out. The modified rush protocol seems to be easier for patients to accomplish, Dr. Schafer said at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization.
The study compared 231 patients on the faster protocol with 392 treated with the traditional approach.
On the “modified rush” protocol, patients aged 12-15 years took preventive medications – 20 mg of prednisone and 150 mg of ranitidine 2 days before their appointment and again the day after. In addition, they took 10 mg of cetirizine and 5 mg of montelukast 2 days before their appointment. Patients also took those same medications the morning of the treatment.
For patients 16 and older, the dose of prednisone was 30 mg and the dose of montelukast was 10 mg.
The protocol consisted of eight shots, starting at a more diluted dose than the conventional protocol, as an additional safety precaution. The shots were given first every 15 minutes, then every 30 minutes or an hour as the dose increases. Once the treatment was over, the dilution was 1:10, a milestone not reached until 3 months under the conventional approach.
An hour or two after the treatment, all patients received 20 mg of prednisone.
Patients resumed the normal conventional protocol after the first visit.
Reactions were rare under the “modified rush” protocol, with 6 patients experiencing flushing but nothing else, 21 with grade 1 reactions, 3 with grade 2, and no reactions worse than that. Five patients needed epinephrine.
Dr. Schafer said the approach may hold appeal because, even though it’s accelerated, there is a slower lead-in to the maintenance dose.
“If you ask allergists, ‘Do you rush?’ they’ll say, ‘No, no, no, I don’t rush. It’s too risky.’” she said. “Hence, modified rush.”
SOURCE: Schafer C et al. AAAAI/WAO Joint Congress abstract 520.
REPORTING FROM AAAAI/WAO JOINT CONGRESS
Key clinical point: Completing an immunotherapy regimen on a ‘modified rush’ basis leads to better adherence.
Major finding: A total of 84.4% completed the ‘modified rush’ protocol, vs. 65.4% for the conventional protocol.
Study details: Retrospective chart review of 623 patients who received allergen immunotherapy via the conventional, 6-month approach or a modified, faster approach.
Disclosures: The investigators reported no conflicts of interest.
Source: Schafer C et al. AAAAI/WAO Joint Congress abstract 520.
Does boosting inhaled glucocorticoids avoid asthma exacerbations?
in preventing the exacerbation from occurring, according to the results of two trials in adults and children.
Presented at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization and simultaneously published in the March 3 online edition of the New England Journal of Medicine, one study explored the effect of quadrupling the inhaled glucocorticoid dose in adults and adolescents with asthma, while the other looked at quintupling the dose in children.
At 1 year, there was a significantly lower incidence of severe asthma exacerbations in the group who used the higher dose of inhaled glucocorticoids (45% vs. 52%; hazard ratio, 0.80; P = .001) after adjusting for age, sex, and peak flow measures at randomization.
Researchers also saw a lower percentage of participants using systemic glucocorticoids in the quadruple-dose group compared with the normal-dose group (33% vs. 40%), and the quadruple-dose group also showed a 14% lower incidence of unscheduled health care consultations.
At the end of the 12-month follow-up, the estimated mean total dose of inhaled glucocorticoids was 385 mg in the quadruple-dose group and 328 mg in the normal-dose group.
The most common serious adverse event was hospitalization for asthma, which occurred three times in the quadruple-dose group and 18 times in the normal-dose group. However the incidence of oral candidiasis and dysphonia – both potentially treatment related – was significantly higher in the quadruple-dose group (36 events vs. 9 events).
Overall, the number needed to treat with the quadruple dose to prevent one severe asthma exacerbation was 15.
“Given the potential benefit with respect to preventing exacerbations and in view of the toxic effects of inhaled glucocorticoids and the biases that may have been introduced by the absence of blinding, individual practitioners, patients, and guideline committees will need to consider whether the magnitude of the reduction achieved is clinically meaningful,” wrote Tricia McKeever, PhD, from the department of epidemiology and public health at the University of Nottingham (United Kingdom) and her coauthors.
The second study, which was double blinded, investigated whether quintupling the dose of inhaled glucocorticoids might avoid exacerbations in children. They randomized 254 children who had mild-moderate persistent asthma and had had at least one exacerbation treated with systemic glucocorticoids in the previous year to manage “yellow zone” early warning signs with either normal dose or five times their usual dose of inhaled glucocorticoids.
The rate of severe asthma exacerbations did not differ significantly between the quintuple-dose and normal-dose groups at the 1-year follow-up (0.48 vs. 0.37; P = 0.3), nor did the time to the first severe exacerbation or the rate of emergency department or urgent care visits.
The four hospitalizations for asthma all occurred in the high-dose group. However, there was a lower growth rate seen in children in the high-dose group than in the low-dose group (5.43 cm/yr vs. 5.65 cm/yr; P = .06). There were no significant differences between the two groups in other adverse events.
However, Daniel J. Jackson, MD, and his coauthors noted that there were fewer yellow zone episodes and fewer exacerbations in both groups than they had anticipated.
“It is important to recognize that our findings are specific to school-age children with mild-to-moderate persistent asthma regularly treated with daily low-dose inhaled glucocorticoids (with good adherence),” wrote Dr. Jackson from the department of pediatrics at the University of Wisconsin–Madison and his coauthors.
The first study was supported by the National Institute for Health Research. Six authors declared grants, personal fees and other funding and support from the pharmaceutical industry outside the submitted work.
The second study was supported by the National Heart, Lung, and Blood Institute. Fifteen authors declared grants, personal fees and other funding from the pharmaceutical industry, as well as other private industry, outside the submitted work. Several also declared grants from organizations including the National Institutes of Health.
SOURCE: McKeeve T et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMoa1714257; Jackson DJ et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJM0a1710988.
These two trials address the important question of whether substantial escalation of regularly used inhaled glucocorticoids prevents exacerbations if started at the first sign of deterioration, as this so-called yellow zone has long been thought the perfect time to initiate more aggressive care. However glucocorticoids have serious side effects, and there is some preclinical evidence that they may enhance viral replication
One trial shows that escalating dose in this yellow zone does not prevent exacerbations in children with the early signs of asthma instability. The second trial is more complex and more controversial, as the open-label design may have biased the outcome, and the degree of benefit is debatable.
Together, these studies suggest that high doses of inhaled glucocorticoids either do not prevent exacerbations or only do so in a small subgroup of patients with as-yet-undefined baseline and exacerbation characteristics.
Philip G. Bardin, PhD, is from the Monash Lung and Sleep Unit at the Monash University Medical Centre in Melbourne, Australia. These comments are taken from an accompanying editorial (N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMe1800152). Dr. Bardin reported personal fees from GlaxoSmithKline outside the submitted work.
These two trials address the important question of whether substantial escalation of regularly used inhaled glucocorticoids prevents exacerbations if started at the first sign of deterioration, as this so-called yellow zone has long been thought the perfect time to initiate more aggressive care. However glucocorticoids have serious side effects, and there is some preclinical evidence that they may enhance viral replication
One trial shows that escalating dose in this yellow zone does not prevent exacerbations in children with the early signs of asthma instability. The second trial is more complex and more controversial, as the open-label design may have biased the outcome, and the degree of benefit is debatable.
Together, these studies suggest that high doses of inhaled glucocorticoids either do not prevent exacerbations or only do so in a small subgroup of patients with as-yet-undefined baseline and exacerbation characteristics.
Philip G. Bardin, PhD, is from the Monash Lung and Sleep Unit at the Monash University Medical Centre in Melbourne, Australia. These comments are taken from an accompanying editorial (N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMe1800152). Dr. Bardin reported personal fees from GlaxoSmithKline outside the submitted work.
These two trials address the important question of whether substantial escalation of regularly used inhaled glucocorticoids prevents exacerbations if started at the first sign of deterioration, as this so-called yellow zone has long been thought the perfect time to initiate more aggressive care. However glucocorticoids have serious side effects, and there is some preclinical evidence that they may enhance viral replication
One trial shows that escalating dose in this yellow zone does not prevent exacerbations in children with the early signs of asthma instability. The second trial is more complex and more controversial, as the open-label design may have biased the outcome, and the degree of benefit is debatable.
Together, these studies suggest that high doses of inhaled glucocorticoids either do not prevent exacerbations or only do so in a small subgroup of patients with as-yet-undefined baseline and exacerbation characteristics.
Philip G. Bardin, PhD, is from the Monash Lung and Sleep Unit at the Monash University Medical Centre in Melbourne, Australia. These comments are taken from an accompanying editorial (N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMe1800152). Dr. Bardin reported personal fees from GlaxoSmithKline outside the submitted work.
in preventing the exacerbation from occurring, according to the results of two trials in adults and children.
Presented at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization and simultaneously published in the March 3 online edition of the New England Journal of Medicine, one study explored the effect of quadrupling the inhaled glucocorticoid dose in adults and adolescents with asthma, while the other looked at quintupling the dose in children.
At 1 year, there was a significantly lower incidence of severe asthma exacerbations in the group who used the higher dose of inhaled glucocorticoids (45% vs. 52%; hazard ratio, 0.80; P = .001) after adjusting for age, sex, and peak flow measures at randomization.
Researchers also saw a lower percentage of participants using systemic glucocorticoids in the quadruple-dose group compared with the normal-dose group (33% vs. 40%), and the quadruple-dose group also showed a 14% lower incidence of unscheduled health care consultations.
At the end of the 12-month follow-up, the estimated mean total dose of inhaled glucocorticoids was 385 mg in the quadruple-dose group and 328 mg in the normal-dose group.
The most common serious adverse event was hospitalization for asthma, which occurred three times in the quadruple-dose group and 18 times in the normal-dose group. However the incidence of oral candidiasis and dysphonia – both potentially treatment related – was significantly higher in the quadruple-dose group (36 events vs. 9 events).
Overall, the number needed to treat with the quadruple dose to prevent one severe asthma exacerbation was 15.
“Given the potential benefit with respect to preventing exacerbations and in view of the toxic effects of inhaled glucocorticoids and the biases that may have been introduced by the absence of blinding, individual practitioners, patients, and guideline committees will need to consider whether the magnitude of the reduction achieved is clinically meaningful,” wrote Tricia McKeever, PhD, from the department of epidemiology and public health at the University of Nottingham (United Kingdom) and her coauthors.
The second study, which was double blinded, investigated whether quintupling the dose of inhaled glucocorticoids might avoid exacerbations in children. They randomized 254 children who had mild-moderate persistent asthma and had had at least one exacerbation treated with systemic glucocorticoids in the previous year to manage “yellow zone” early warning signs with either normal dose or five times their usual dose of inhaled glucocorticoids.
The rate of severe asthma exacerbations did not differ significantly between the quintuple-dose and normal-dose groups at the 1-year follow-up (0.48 vs. 0.37; P = 0.3), nor did the time to the first severe exacerbation or the rate of emergency department or urgent care visits.
The four hospitalizations for asthma all occurred in the high-dose group. However, there was a lower growth rate seen in children in the high-dose group than in the low-dose group (5.43 cm/yr vs. 5.65 cm/yr; P = .06). There were no significant differences between the two groups in other adverse events.
However, Daniel J. Jackson, MD, and his coauthors noted that there were fewer yellow zone episodes and fewer exacerbations in both groups than they had anticipated.
“It is important to recognize that our findings are specific to school-age children with mild-to-moderate persistent asthma regularly treated with daily low-dose inhaled glucocorticoids (with good adherence),” wrote Dr. Jackson from the department of pediatrics at the University of Wisconsin–Madison and his coauthors.
The first study was supported by the National Institute for Health Research. Six authors declared grants, personal fees and other funding and support from the pharmaceutical industry outside the submitted work.
The second study was supported by the National Heart, Lung, and Blood Institute. Fifteen authors declared grants, personal fees and other funding from the pharmaceutical industry, as well as other private industry, outside the submitted work. Several also declared grants from organizations including the National Institutes of Health.
SOURCE: McKeeve T et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMoa1714257; Jackson DJ et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJM0a1710988.
in preventing the exacerbation from occurring, according to the results of two trials in adults and children.
Presented at the joint congress of the American Academy of Allergy, Asthma, and Immunology and the World Asthma Organization and simultaneously published in the March 3 online edition of the New England Journal of Medicine, one study explored the effect of quadrupling the inhaled glucocorticoid dose in adults and adolescents with asthma, while the other looked at quintupling the dose in children.
At 1 year, there was a significantly lower incidence of severe asthma exacerbations in the group who used the higher dose of inhaled glucocorticoids (45% vs. 52%; hazard ratio, 0.80; P = .001) after adjusting for age, sex, and peak flow measures at randomization.
Researchers also saw a lower percentage of participants using systemic glucocorticoids in the quadruple-dose group compared with the normal-dose group (33% vs. 40%), and the quadruple-dose group also showed a 14% lower incidence of unscheduled health care consultations.
At the end of the 12-month follow-up, the estimated mean total dose of inhaled glucocorticoids was 385 mg in the quadruple-dose group and 328 mg in the normal-dose group.
The most common serious adverse event was hospitalization for asthma, which occurred three times in the quadruple-dose group and 18 times in the normal-dose group. However the incidence of oral candidiasis and dysphonia – both potentially treatment related – was significantly higher in the quadruple-dose group (36 events vs. 9 events).
Overall, the number needed to treat with the quadruple dose to prevent one severe asthma exacerbation was 15.
“Given the potential benefit with respect to preventing exacerbations and in view of the toxic effects of inhaled glucocorticoids and the biases that may have been introduced by the absence of blinding, individual practitioners, patients, and guideline committees will need to consider whether the magnitude of the reduction achieved is clinically meaningful,” wrote Tricia McKeever, PhD, from the department of epidemiology and public health at the University of Nottingham (United Kingdom) and her coauthors.
The second study, which was double blinded, investigated whether quintupling the dose of inhaled glucocorticoids might avoid exacerbations in children. They randomized 254 children who had mild-moderate persistent asthma and had had at least one exacerbation treated with systemic glucocorticoids in the previous year to manage “yellow zone” early warning signs with either normal dose or five times their usual dose of inhaled glucocorticoids.
The rate of severe asthma exacerbations did not differ significantly between the quintuple-dose and normal-dose groups at the 1-year follow-up (0.48 vs. 0.37; P = 0.3), nor did the time to the first severe exacerbation or the rate of emergency department or urgent care visits.
The four hospitalizations for asthma all occurred in the high-dose group. However, there was a lower growth rate seen in children in the high-dose group than in the low-dose group (5.43 cm/yr vs. 5.65 cm/yr; P = .06). There were no significant differences between the two groups in other adverse events.
However, Daniel J. Jackson, MD, and his coauthors noted that there were fewer yellow zone episodes and fewer exacerbations in both groups than they had anticipated.
“It is important to recognize that our findings are specific to school-age children with mild-to-moderate persistent asthma regularly treated with daily low-dose inhaled glucocorticoids (with good adherence),” wrote Dr. Jackson from the department of pediatrics at the University of Wisconsin–Madison and his coauthors.
The first study was supported by the National Institute for Health Research. Six authors declared grants, personal fees and other funding and support from the pharmaceutical industry outside the submitted work.
The second study was supported by the National Heart, Lung, and Blood Institute. Fifteen authors declared grants, personal fees and other funding from the pharmaceutical industry, as well as other private industry, outside the submitted work. Several also declared grants from organizations including the National Institutes of Health.
SOURCE: McKeeve T et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMoa1714257; Jackson DJ et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJM0a1710988.
FROM AAAAI/WAO JOINT CONGRESS
Key clinical point: Escalating the dose of inhaled glucocorticoids at the first early warnings of an asthma exacerbation may not significantly reduce the likelihood of the exacerbation occurring.
Major finding: Fifteen individuals would need to quadruple their dose of inhaled glucocorticoids to avoid one asthma exacerbation.
Data source: Two randomized, controlled trials in 1,992 adolescents and adults and 254 children with asthma.
Disclosures: The first study was supported by the National Institute for Health Research. Six authors declared grants, personal fees, and other funding and support from the pharmaceutical industry outside the submitted work. The second study was supported by the National Heart, Lung, and Blood Institute. Fifteen authors declared grants, personal fees, and other funding from the pharmaceutical industry, as well as other private industry, outside the submitted work. Several also declared grants from organizations including the National Institutes of Health. No other conflicts of interest were declared.
Sources: McKeeve T et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJMoa1714257; Jackson DJ et al. N Engl J Med. 2018 Mar 3. doi: 10.1056/NEJM0a1710988.
Intermittent dosing cuts time to extubation for surgical patients
SAN ANTONIO – according to a preliminary analysis of a randomized trial.
Additionally, the researchers found that much lower amounts of sedation and analgesia were given to patients who underwent intermittent dosing, compared with patients who received a continuous infusion.
Of the 95 patients in the trial, 39 were randomized to intermittent dosing and 56 to the control group of continuous infusion, with the drugs midazolam and fentanyl having been given to both groups.
Mean mechanical ventilation time was 65 hours in the intermittent dosing arm, versus 111 hours in the continuous infusion arm (P less than 0.03), noted Dr. Sich, a fourth-year general surgery resident at Abington Memorial Hospital, Abington, Pa., during his presentation.
Patients in the continuous infusions arm of the trial received a mean of 73.1 mg of midazolam, compared with 18 mg for the intermittent dosing arm, a difference that approached very closely to statistical significance (P = 0.06) and was thrown off in the latest iteration by an outlier, Dr. Sich explained. The relative difference between the mean fentanyl doses administered was even greater between the two groups, with 5,848 mcg given to patients in the control group, versus the 942 mcg given to participants in the intermittent dosing group (P less than 0.01).
“This is a new way to use an old drug, and it really might be beneficial, and can even be used as first-line therapy and a way to keep patients awake and off the ventilator,” said Dr. Sich, referring to the intermittent dosing. Continuous infusions leave patients oversedated and prolong ventilation time.
“What we propose, rather, is using a sliding-scale intermittent pain and sedation regimen,” he said. “We believe that it won’t compromise patient care and won’t compromise patient comfort, and it will lead to shorter mechanical ventilation times for surgical patients than continuous infusions.”
Dr. Sich also pointed out that there was no difference in time spent at target levels of sedation and analgesia between the two trial groups. Referring to this finding, he noted that “we wanted to make sure that in the intermittent arm we’re giving them less drug, but we don’t want them to be [less comfortable].”
One potential drawback to the intermittent dosing approach is that it is more nursing intensive, according to Dr. Sich, since it is based on a nursing treatment protocol to give medications every hour.
Intermittent dosing is “more hands-on” than a typical continuous infusion approach and so was more challenging for nurses who, per the treatment protocol, had to give medications every hour, he explained. However, “when they saw the data in the months and year as we’ve been going on, they’re actually quite proud of our work and their work.”
Gilman Baker Allen, MD, a pulmonologist and intensivist at the University of Vermont Medical Center, Burlington, said the study was “terrific work” and acknowledged the importance of gauging nurse satisfaction with the protocol.
“I think that when you feed this kind of data back to nursing staff, they may not be satisfied with the intensity of the work, but when they see the rewards at the end, it oftentimes is a very positive experience,” said Dr. Allen, who moderated the session.
Dr. Sich and his colleagues had no financial disclosures or conflicts of interest related to the study.
SOURCE: Sich N et al. CCC47, Abstract 18.
SAN ANTONIO – according to a preliminary analysis of a randomized trial.
Additionally, the researchers found that much lower amounts of sedation and analgesia were given to patients who underwent intermittent dosing, compared with patients who received a continuous infusion.
Of the 95 patients in the trial, 39 were randomized to intermittent dosing and 56 to the control group of continuous infusion, with the drugs midazolam and fentanyl having been given to both groups.
Mean mechanical ventilation time was 65 hours in the intermittent dosing arm, versus 111 hours in the continuous infusion arm (P less than 0.03), noted Dr. Sich, a fourth-year general surgery resident at Abington Memorial Hospital, Abington, Pa., during his presentation.
Patients in the continuous infusions arm of the trial received a mean of 73.1 mg of midazolam, compared with 18 mg for the intermittent dosing arm, a difference that approached very closely to statistical significance (P = 0.06) and was thrown off in the latest iteration by an outlier, Dr. Sich explained. The relative difference between the mean fentanyl doses administered was even greater between the two groups, with 5,848 mcg given to patients in the control group, versus the 942 mcg given to participants in the intermittent dosing group (P less than 0.01).
“This is a new way to use an old drug, and it really might be beneficial, and can even be used as first-line therapy and a way to keep patients awake and off the ventilator,” said Dr. Sich, referring to the intermittent dosing. Continuous infusions leave patients oversedated and prolong ventilation time.
“What we propose, rather, is using a sliding-scale intermittent pain and sedation regimen,” he said. “We believe that it won’t compromise patient care and won’t compromise patient comfort, and it will lead to shorter mechanical ventilation times for surgical patients than continuous infusions.”
Dr. Sich also pointed out that there was no difference in time spent at target levels of sedation and analgesia between the two trial groups. Referring to this finding, he noted that “we wanted to make sure that in the intermittent arm we’re giving them less drug, but we don’t want them to be [less comfortable].”
One potential drawback to the intermittent dosing approach is that it is more nursing intensive, according to Dr. Sich, since it is based on a nursing treatment protocol to give medications every hour.
Intermittent dosing is “more hands-on” than a typical continuous infusion approach and so was more challenging for nurses who, per the treatment protocol, had to give medications every hour, he explained. However, “when they saw the data in the months and year as we’ve been going on, they’re actually quite proud of our work and their work.”
Gilman Baker Allen, MD, a pulmonologist and intensivist at the University of Vermont Medical Center, Burlington, said the study was “terrific work” and acknowledged the importance of gauging nurse satisfaction with the protocol.
“I think that when you feed this kind of data back to nursing staff, they may not be satisfied with the intensity of the work, but when they see the rewards at the end, it oftentimes is a very positive experience,” said Dr. Allen, who moderated the session.
Dr. Sich and his colleagues had no financial disclosures or conflicts of interest related to the study.
SOURCE: Sich N et al. CCC47, Abstract 18.
SAN ANTONIO – according to a preliminary analysis of a randomized trial.
Additionally, the researchers found that much lower amounts of sedation and analgesia were given to patients who underwent intermittent dosing, compared with patients who received a continuous infusion.
Of the 95 patients in the trial, 39 were randomized to intermittent dosing and 56 to the control group of continuous infusion, with the drugs midazolam and fentanyl having been given to both groups.
Mean mechanical ventilation time was 65 hours in the intermittent dosing arm, versus 111 hours in the continuous infusion arm (P less than 0.03), noted Dr. Sich, a fourth-year general surgery resident at Abington Memorial Hospital, Abington, Pa., during his presentation.
Patients in the continuous infusions arm of the trial received a mean of 73.1 mg of midazolam, compared with 18 mg for the intermittent dosing arm, a difference that approached very closely to statistical significance (P = 0.06) and was thrown off in the latest iteration by an outlier, Dr. Sich explained. The relative difference between the mean fentanyl doses administered was even greater between the two groups, with 5,848 mcg given to patients in the control group, versus the 942 mcg given to participants in the intermittent dosing group (P less than 0.01).
“This is a new way to use an old drug, and it really might be beneficial, and can even be used as first-line therapy and a way to keep patients awake and off the ventilator,” said Dr. Sich, referring to the intermittent dosing. Continuous infusions leave patients oversedated and prolong ventilation time.
“What we propose, rather, is using a sliding-scale intermittent pain and sedation regimen,” he said. “We believe that it won’t compromise patient care and won’t compromise patient comfort, and it will lead to shorter mechanical ventilation times for surgical patients than continuous infusions.”
Dr. Sich also pointed out that there was no difference in time spent at target levels of sedation and analgesia between the two trial groups. Referring to this finding, he noted that “we wanted to make sure that in the intermittent arm we’re giving them less drug, but we don’t want them to be [less comfortable].”
One potential drawback to the intermittent dosing approach is that it is more nursing intensive, according to Dr. Sich, since it is based on a nursing treatment protocol to give medications every hour.
Intermittent dosing is “more hands-on” than a typical continuous infusion approach and so was more challenging for nurses who, per the treatment protocol, had to give medications every hour, he explained. However, “when they saw the data in the months and year as we’ve been going on, they’re actually quite proud of our work and their work.”
Gilman Baker Allen, MD, a pulmonologist and intensivist at the University of Vermont Medical Center, Burlington, said the study was “terrific work” and acknowledged the importance of gauging nurse satisfaction with the protocol.
“I think that when you feed this kind of data back to nursing staff, they may not be satisfied with the intensity of the work, but when they see the rewards at the end, it oftentimes is a very positive experience,” said Dr. Allen, who moderated the session.
Dr. Sich and his colleagues had no financial disclosures or conflicts of interest related to the study.
SOURCE: Sich N et al. CCC47, Abstract 18.
Reporting from CCC47
Key clinical point: Among patients requiring ventilation, intermittent administration of sedation and analgesia significantly reduced mechanical ventilation time and total amount of drugs versus a continuous infusion approach.
Major finding: Mean mechanical ventilation time was 65 hours in the intermittent dosing arm, versus 111 hours in the continuous infusion arm (P less than 0.03).
Data source: A single-blinded, randomized, controlled trial of 95 surgical patients requiring ventilation.
Disclosures: The authors reported no financial disclosures or conflicts of interest related to the study.
Source: Sich N et al. CCC47, Abstract 18.
On-demand nebulization in ICU equivalent to standard
SAN ANTONIO – with both medications, according to the results of a randomized clinical trial presented by Frederique Paulus, RN, PhD.
In this study, adverse events such as tachyarrhythmia and agitation were less frequent with the on-demand approach, in which patients receive nebulization based on strict clinical indications, Dr. Paulus reported at the Critical Care Congress sponsored by the Society for Critical Care Medicine. The study was published simultaneously in JAMA.
The on-demand approach may also be cost saving, she noted, citing an economic analysis underway that is not yet ready for publication.
“In our ICU, it will save us 350,000 Euros a year,” she said. “In the Netherlands, 40,000 patients will be mechanically ventilated in a year, so it will save us millions in the Netherlands alone.”
The study included adult ICU patients who were expected not to be extubated for at least 24 hours. Dr. Paulus presented the primary analysis of the study, which included data for 922 patients who were randomized either to the on-demand group (n = 455) or the routine nebulization group (n = 467) and completed follow-up.
Patients assigned to the on-demand group received acetylcysteine-containing solutions if they had thick or tenacious secretions, or salbutamol-containing solutions if wheezing was observed or suspected or when findings were suggestive of lower-airway obstruction, according to the paper, published in JAMA.
The primary outcome, number of ventilator-free days at day 28 of the study, was noninferior in the on-demand group versus the routine group, Dr. Paulus said.
The median number of ventilator-free days was 21 for the on-demand group and 20 for the routine group, said the paper.
The length of stay, mortality, and proportion of patients developing pulmonary complications did not differ between the two study arms, the investigators also reported in JAMA.
However, adverse events occurred in just 13.8% of the on-demand group, compared with 29.3% of the routine group (P less than .001), with the difference in adverse events mainly attributable to less tachyarrhythmia and agitation in the experimental group, according to the researchers.
Dr. Paulus and coauthors reported no conflicts of interest related to the study.
SOURCE: Paulus F et al. (van Meenen DMP et al.) JAMA. 2018 Feb. doi: 10.1001/jama.2018.0949.
I would suspect local practice patterns with nebulized acytylcysteine to vary even more widely than bronchodilator administration strategies.
I would suspect local practice patterns with nebulized acytylcysteine to vary even more widely than bronchodilator administration strategies.
I would suspect local practice patterns with nebulized acytylcysteine to vary even more widely than bronchodilator administration strategies.
SAN ANTONIO – with both medications, according to the results of a randomized clinical trial presented by Frederique Paulus, RN, PhD.
In this study, adverse events such as tachyarrhythmia and agitation were less frequent with the on-demand approach, in which patients receive nebulization based on strict clinical indications, Dr. Paulus reported at the Critical Care Congress sponsored by the Society for Critical Care Medicine. The study was published simultaneously in JAMA.
The on-demand approach may also be cost saving, she noted, citing an economic analysis underway that is not yet ready for publication.
“In our ICU, it will save us 350,000 Euros a year,” she said. “In the Netherlands, 40,000 patients will be mechanically ventilated in a year, so it will save us millions in the Netherlands alone.”
The study included adult ICU patients who were expected not to be extubated for at least 24 hours. Dr. Paulus presented the primary analysis of the study, which included data for 922 patients who were randomized either to the on-demand group (n = 455) or the routine nebulization group (n = 467) and completed follow-up.
Patients assigned to the on-demand group received acetylcysteine-containing solutions if they had thick or tenacious secretions, or salbutamol-containing solutions if wheezing was observed or suspected or when findings were suggestive of lower-airway obstruction, according to the paper, published in JAMA.
The primary outcome, number of ventilator-free days at day 28 of the study, was noninferior in the on-demand group versus the routine group, Dr. Paulus said.
The median number of ventilator-free days was 21 for the on-demand group and 20 for the routine group, said the paper.
The length of stay, mortality, and proportion of patients developing pulmonary complications did not differ between the two study arms, the investigators also reported in JAMA.
However, adverse events occurred in just 13.8% of the on-demand group, compared with 29.3% of the routine group (P less than .001), with the difference in adverse events mainly attributable to less tachyarrhythmia and agitation in the experimental group, according to the researchers.
Dr. Paulus and coauthors reported no conflicts of interest related to the study.
SOURCE: Paulus F et al. (van Meenen DMP et al.) JAMA. 2018 Feb. doi: 10.1001/jama.2018.0949.
SAN ANTONIO – with both medications, according to the results of a randomized clinical trial presented by Frederique Paulus, RN, PhD.
In this study, adverse events such as tachyarrhythmia and agitation were less frequent with the on-demand approach, in which patients receive nebulization based on strict clinical indications, Dr. Paulus reported at the Critical Care Congress sponsored by the Society for Critical Care Medicine. The study was published simultaneously in JAMA.
The on-demand approach may also be cost saving, she noted, citing an economic analysis underway that is not yet ready for publication.
“In our ICU, it will save us 350,000 Euros a year,” she said. “In the Netherlands, 40,000 patients will be mechanically ventilated in a year, so it will save us millions in the Netherlands alone.”
The study included adult ICU patients who were expected not to be extubated for at least 24 hours. Dr. Paulus presented the primary analysis of the study, which included data for 922 patients who were randomized either to the on-demand group (n = 455) or the routine nebulization group (n = 467) and completed follow-up.
Patients assigned to the on-demand group received acetylcysteine-containing solutions if they had thick or tenacious secretions, or salbutamol-containing solutions if wheezing was observed or suspected or when findings were suggestive of lower-airway obstruction, according to the paper, published in JAMA.
The primary outcome, number of ventilator-free days at day 28 of the study, was noninferior in the on-demand group versus the routine group, Dr. Paulus said.
The median number of ventilator-free days was 21 for the on-demand group and 20 for the routine group, said the paper.
The length of stay, mortality, and proportion of patients developing pulmonary complications did not differ between the two study arms, the investigators also reported in JAMA.
However, adverse events occurred in just 13.8% of the on-demand group, compared with 29.3% of the routine group (P less than .001), with the difference in adverse events mainly attributable to less tachyarrhythmia and agitation in the experimental group, according to the researchers.
Dr. Paulus and coauthors reported no conflicts of interest related to the study.
SOURCE: Paulus F et al. (van Meenen DMP et al.) JAMA. 2018 Feb. doi: 10.1001/jama.2018.0949.
REPORTING FROM CCC47
Key clinical point: Compared with routine nebulization with acetylcysteine or salbutamol, on-demand nebulization with acetylcysteine or salbutamol was noninferior among patients receiving invasive ventilation in the ICU.
Major finding: At day 28, the median number of ventilator-free days was 21 in the on-demand group and 20 in the routine care group.
Data source: Primary analysis of a randomized clinical trial including 922 adult patients who were expected to need at least 24 hours of invasive ventilation at one of seven ICUs in the Netherlands.
Disclosures: Authors reported no conflicts of interest related to the study.
Source: Paulus F et al. (van Meenen DMP et al.) JAMA. 2018 Feb. doi: 10.1001/jama.2018.0949.