User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
New COVID-19 cases rise again in children
The number of new COVID-19 cases in children increased for the second consecutive week in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
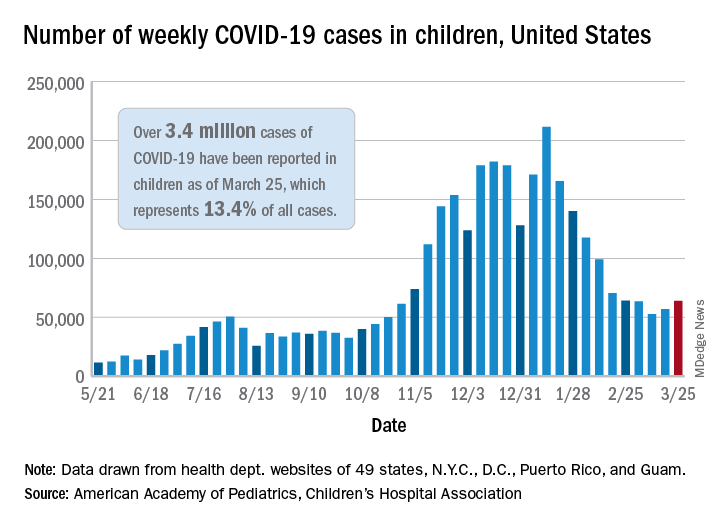
That brings the number of children infected with the coronavirus to over 3.4 million since the beginning of the pandemic, or 13.4% of all reported cases, the AAP and CHA said in their weekly COVID-19 report.
For just the week of March 19-25, however, the proportion of all cases occurring in children was quite a bit higher, 19.1%. That’s higher than at any other point during the pandemic, passing the previous high of 18.7% set just a week earlier, based on the data collected by AAP/CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The national infection rate was 4,525 cases per 100,000 children for the week of March 19-25, compared with 4,440 per 100,000 the previous week. States falling the farthest from that national mark were Hawaii at 1,101 per 100,000 and North Dakota at 8,848, the AAP and CHA said.
There was double-digit increase, 11, in the number of child deaths, as the total went from 268 to 279 despite Virginia’s revising its mortality data downward. The mortality rate for children remains 0.01%, and children represent only 0.06% of all COVID-19–related deaths in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting deaths by age, the report shows.
The state/local-level data show that Texas has the highest number of child deaths (48), followed by Arizona (26), New York City (22), California (16), and Illinois (16), while nine states and the District of Columbia have not yet reported a death, the AAP and CHA said.
The number of new COVID-19 cases in children increased for the second consecutive week in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

That brings the number of children infected with the coronavirus to over 3.4 million since the beginning of the pandemic, or 13.4% of all reported cases, the AAP and CHA said in their weekly COVID-19 report.
For just the week of March 19-25, however, the proportion of all cases occurring in children was quite a bit higher, 19.1%. That’s higher than at any other point during the pandemic, passing the previous high of 18.7% set just a week earlier, based on the data collected by AAP/CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The national infection rate was 4,525 cases per 100,000 children for the week of March 19-25, compared with 4,440 per 100,000 the previous week. States falling the farthest from that national mark were Hawaii at 1,101 per 100,000 and North Dakota at 8,848, the AAP and CHA said.
There was double-digit increase, 11, in the number of child deaths, as the total went from 268 to 279 despite Virginia’s revising its mortality data downward. The mortality rate for children remains 0.01%, and children represent only 0.06% of all COVID-19–related deaths in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting deaths by age, the report shows.
The state/local-level data show that Texas has the highest number of child deaths (48), followed by Arizona (26), New York City (22), California (16), and Illinois (16), while nine states and the District of Columbia have not yet reported a death, the AAP and CHA said.
The number of new COVID-19 cases in children increased for the second consecutive week in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

That brings the number of children infected with the coronavirus to over 3.4 million since the beginning of the pandemic, or 13.4% of all reported cases, the AAP and CHA said in their weekly COVID-19 report.
For just the week of March 19-25, however, the proportion of all cases occurring in children was quite a bit higher, 19.1%. That’s higher than at any other point during the pandemic, passing the previous high of 18.7% set just a week earlier, based on the data collected by AAP/CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The national infection rate was 4,525 cases per 100,000 children for the week of March 19-25, compared with 4,440 per 100,000 the previous week. States falling the farthest from that national mark were Hawaii at 1,101 per 100,000 and North Dakota at 8,848, the AAP and CHA said.
There was double-digit increase, 11, in the number of child deaths, as the total went from 268 to 279 despite Virginia’s revising its mortality data downward. The mortality rate for children remains 0.01%, and children represent only 0.06% of all COVID-19–related deaths in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting deaths by age, the report shows.
The state/local-level data show that Texas has the highest number of child deaths (48), followed by Arizona (26), New York City (22), California (16), and Illinois (16), while nine states and the District of Columbia have not yet reported a death, the AAP and CHA said.
FDA approves mirabegron to treat pediatric NDO
The Food and Drug Administration has expanded the indication for mirabegron (Myrbetriq/Myrbetriq Granules) to treat neurogenic detrusor overactivity (NDO), a bladder dysfunction related to neurologic impairment, in children aged 3 years and older.
This comes 1 year after the FDA approved solifenacin succinate, the first treatment of NDO in pediatric patients aged 2 years and older.
The approval of the drug for these new indications is a “positive step” for the treatment of NDO in young patients, Christine P. Nguyen, MD, director of the FDA’s Division of Urology, Obstetrics, and Gynecology, said in an FDA statement.
“Mirabegron, the active ingredient in Myrbetriq and Myrbetriq Granules, works by a different mechanism of action from the currently approved treatments, providing a new treatment option for these young patients. We remain committed to facilitating the development and approval of safe and effective therapies for pediatric NDO patients,” Dr. Nguyen said.
NDO is a bladder dysfunction that frequently occurs in patients with congenital conditions, such as spina bifida. It also occurs in people who suffer from other diseases or injuries of the nervous system, such as multiple sclerosis and spinal cord injury. Symptoms of the condition include urinary frequency and incontinence.
The condition is characterized by the overactivity of the bladder wall muscle, which is normally relaxed to allow storage of urine. Irregular bladder muscle contraction increases storage pressure and decreases the amount of urine the bladder can hold. This can also put the upper urinary tract at risk for deterioration and cause permanent damage to the kidneys.
The effectiveness of Myrbetriq and Myrbetriq Granules for pediatric NDO was determined in a study of 86 children and adolescents aged 3-17 years. The researchers found that after 24 weeks of treatment, the drug improved the patients’ bladder capacity, reduced the number of bladder wall muscle contractions, and improved the volume of urine that could be held. It also reduced the daily number of episodes of leakage.
Side effects of Myrbetriq and Myrbetriq Granules include urinary tract infection, cold symptoms, angioedema, constipation, and headache. The FDA said the drug may also increase blood pressure and may worsen blood pressure in patients who have a history of hypertension.
The FDA approved mirabegron in 2012 to treat overactive bladder in adults.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has expanded the indication for mirabegron (Myrbetriq/Myrbetriq Granules) to treat neurogenic detrusor overactivity (NDO), a bladder dysfunction related to neurologic impairment, in children aged 3 years and older.
This comes 1 year after the FDA approved solifenacin succinate, the first treatment of NDO in pediatric patients aged 2 years and older.
The approval of the drug for these new indications is a “positive step” for the treatment of NDO in young patients, Christine P. Nguyen, MD, director of the FDA’s Division of Urology, Obstetrics, and Gynecology, said in an FDA statement.
“Mirabegron, the active ingredient in Myrbetriq and Myrbetriq Granules, works by a different mechanism of action from the currently approved treatments, providing a new treatment option for these young patients. We remain committed to facilitating the development and approval of safe and effective therapies for pediatric NDO patients,” Dr. Nguyen said.
NDO is a bladder dysfunction that frequently occurs in patients with congenital conditions, such as spina bifida. It also occurs in people who suffer from other diseases or injuries of the nervous system, such as multiple sclerosis and spinal cord injury. Symptoms of the condition include urinary frequency and incontinence.
The condition is characterized by the overactivity of the bladder wall muscle, which is normally relaxed to allow storage of urine. Irregular bladder muscle contraction increases storage pressure and decreases the amount of urine the bladder can hold. This can also put the upper urinary tract at risk for deterioration and cause permanent damage to the kidneys.
The effectiveness of Myrbetriq and Myrbetriq Granules for pediatric NDO was determined in a study of 86 children and adolescents aged 3-17 years. The researchers found that after 24 weeks of treatment, the drug improved the patients’ bladder capacity, reduced the number of bladder wall muscle contractions, and improved the volume of urine that could be held. It also reduced the daily number of episodes of leakage.
Side effects of Myrbetriq and Myrbetriq Granules include urinary tract infection, cold symptoms, angioedema, constipation, and headache. The FDA said the drug may also increase blood pressure and may worsen blood pressure in patients who have a history of hypertension.
The FDA approved mirabegron in 2012 to treat overactive bladder in adults.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has expanded the indication for mirabegron (Myrbetriq/Myrbetriq Granules) to treat neurogenic detrusor overactivity (NDO), a bladder dysfunction related to neurologic impairment, in children aged 3 years and older.
This comes 1 year after the FDA approved solifenacin succinate, the first treatment of NDO in pediatric patients aged 2 years and older.
The approval of the drug for these new indications is a “positive step” for the treatment of NDO in young patients, Christine P. Nguyen, MD, director of the FDA’s Division of Urology, Obstetrics, and Gynecology, said in an FDA statement.
“Mirabegron, the active ingredient in Myrbetriq and Myrbetriq Granules, works by a different mechanism of action from the currently approved treatments, providing a new treatment option for these young patients. We remain committed to facilitating the development and approval of safe and effective therapies for pediatric NDO patients,” Dr. Nguyen said.
NDO is a bladder dysfunction that frequently occurs in patients with congenital conditions, such as spina bifida. It also occurs in people who suffer from other diseases or injuries of the nervous system, such as multiple sclerosis and spinal cord injury. Symptoms of the condition include urinary frequency and incontinence.
The condition is characterized by the overactivity of the bladder wall muscle, which is normally relaxed to allow storage of urine. Irregular bladder muscle contraction increases storage pressure and decreases the amount of urine the bladder can hold. This can also put the upper urinary tract at risk for deterioration and cause permanent damage to the kidneys.
The effectiveness of Myrbetriq and Myrbetriq Granules for pediatric NDO was determined in a study of 86 children and adolescents aged 3-17 years. The researchers found that after 24 weeks of treatment, the drug improved the patients’ bladder capacity, reduced the number of bladder wall muscle contractions, and improved the volume of urine that could be held. It also reduced the daily number of episodes of leakage.
Side effects of Myrbetriq and Myrbetriq Granules include urinary tract infection, cold symptoms, angioedema, constipation, and headache. The FDA said the drug may also increase blood pressure and may worsen blood pressure in patients who have a history of hypertension.
The FDA approved mirabegron in 2012 to treat overactive bladder in adults.
A version of this article first appeared on Medscape.com.
Child abuse tied to persistent inflammation in later life
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Politics has no place here,’ AAP says about transgender care
The American Academy of Pediatrics (AAP) released a statement condemning state legislation introduced across the country that would prohibit the medical treatment of children who identify as transgender.
“Politics has no place here. Transgender children, like all children, just want to belong. We will fight state by state, in the courts, and on the national stage to make sure they know they do,” said the American Academy of Pediatrics, an organization that recommends that youth who identify as transgender have access to comprehensive, gender-affirming and “developmentally appropriate” health care.
There has been a lot of controversy among physicians surrounding gender-affirming care for children. In 2019, three separate groups of physicians questioned the hormonal treatment of children and adolescents with gender dysphoria, saying more research is needed to support gender-affirming care recommendations from groups such as the AAP. In addition, California-based endocrinologist Michael K. Laidlaw, MD, called the use of puberty blockers – medication used to delay or prevent the onset of puberty – an “experiment” and “public health problem.”
Some of the bills circulating would prohibit gender-affirming care for children and penalize pediatricians and other physicians for providing such care. Other bills would prevent transgender youth from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Jason Rafferty, MD, a pediatrician who authored AAP’s 2018 policy statement encouraging pediatricians to provide gender-affirmative care to children and adolescents, said in an interview that these proposed state bills are targeting a vulnerable population at a vulnerable time.
“You have this marginalized group and right in the midst of a pandemic – and [social isolation] – and you have these policies coming out that are really targeting the most vulnerable,” said Dr. Rafferty, who practices at the Adolescent Healthcare Center at Hasbro Children’s Hospital, Providence, R.I. “Some of the proposed policies would ensure that some of these transgender kids wouldn’t have access to basic medical care [or access to areas] where pediatricians are affirming these kids and creating a safe environment for them.”
M. Brett Cooper, MD, of pediatrics at UT Southwestern Medical Center, Dallas, said in an interview that the proposed state legislations are based on personal beliefs, not evidence-based care.
He said the AAP is speaking out because many of these proposed state legislatures are advancing bills that would affect gender-diverse children.
“Many of these bills are making it close to passing both chambers of a state legislature and thus sent to the governor for approval,” Dr. Cooper said. “In past legislative sessions, bills such as these have never it made out of committee in either a state House or state Senate.”
Early in March, South Dakota Governor Kristi Noem tweeted that she was “excited to sign” a bill that would prevent children who are transgender from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Meanwhile, previous studies have shown that teens whose gender identity does not align with their sex assigned at birth were more likely to report a suicide attempt, compared with cisgender youth. A 2015 study has attributed this increased risk of suicide to marginalization, social exclusion, and trauma. The bills being proposed may exacerbate this, according to Dr. Rafferty.
These bills point in the direction of approaches and models that research shows may be harmful to transgender children, he said.
“Beyond that, policies that are out there affecting participation in sports and other activities really just draw attention to these youth in unnecessary ways and really puts their physical and mental health at risk in a school environment,” he added.
“Whether it’s health care, school, community, or home [these policies] just really create a very hostile and dangerous environment toward a population that already is at significant risk,” Dr. Rafferty said.
The AAP’s clinical care guidelines for children who identify as transgender state that pediatricians who provide gender-affirmative care and speak with children and their families about gender issues help foster an environment of nurturance and support.
“Many people think that the care provided to gender diverse young people is rogue,” Dr. Cooper said. “Pediatric gender clinics are often either multidisciplinary or have a readily accessible team, including social work, mental health, and the physician. Medical care for gender diverse youth follows well-established standards of care.”
Dr. Rafferty said it is important for care providers to support children and teenagers who identify as transgender by creating a safe space and helping them identify what they need.
“There’s a lot out there that really tries to unite gender and biological sex together, saying that gender can be reduced to your anatomy,” Dr. Rafferty said. “But I think what we’re coming to appreciate is that our sense of identity is much more complex than simply our genes or biology.”
Dr. Rafferty is a member of the American Academy of Pediatrics, the Society of Adolescent Health and Medicine, and the American Academy of Child and Adolescent Psychiatry. Dr. Cooper is a columnist for Pediatric News and is on the board of trustees for the Texas Medical Association.
The American Academy of Pediatrics (AAP) released a statement condemning state legislation introduced across the country that would prohibit the medical treatment of children who identify as transgender.
“Politics has no place here. Transgender children, like all children, just want to belong. We will fight state by state, in the courts, and on the national stage to make sure they know they do,” said the American Academy of Pediatrics, an organization that recommends that youth who identify as transgender have access to comprehensive, gender-affirming and “developmentally appropriate” health care.
There has been a lot of controversy among physicians surrounding gender-affirming care for children. In 2019, three separate groups of physicians questioned the hormonal treatment of children and adolescents with gender dysphoria, saying more research is needed to support gender-affirming care recommendations from groups such as the AAP. In addition, California-based endocrinologist Michael K. Laidlaw, MD, called the use of puberty blockers – medication used to delay or prevent the onset of puberty – an “experiment” and “public health problem.”
Some of the bills circulating would prohibit gender-affirming care for children and penalize pediatricians and other physicians for providing such care. Other bills would prevent transgender youth from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Jason Rafferty, MD, a pediatrician who authored AAP’s 2018 policy statement encouraging pediatricians to provide gender-affirmative care to children and adolescents, said in an interview that these proposed state bills are targeting a vulnerable population at a vulnerable time.
“You have this marginalized group and right in the midst of a pandemic – and [social isolation] – and you have these policies coming out that are really targeting the most vulnerable,” said Dr. Rafferty, who practices at the Adolescent Healthcare Center at Hasbro Children’s Hospital, Providence, R.I. “Some of the proposed policies would ensure that some of these transgender kids wouldn’t have access to basic medical care [or access to areas] where pediatricians are affirming these kids and creating a safe environment for them.”
M. Brett Cooper, MD, of pediatrics at UT Southwestern Medical Center, Dallas, said in an interview that the proposed state legislations are based on personal beliefs, not evidence-based care.
He said the AAP is speaking out because many of these proposed state legislatures are advancing bills that would affect gender-diverse children.
“Many of these bills are making it close to passing both chambers of a state legislature and thus sent to the governor for approval,” Dr. Cooper said. “In past legislative sessions, bills such as these have never it made out of committee in either a state House or state Senate.”
Early in March, South Dakota Governor Kristi Noem tweeted that she was “excited to sign” a bill that would prevent children who are transgender from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Meanwhile, previous studies have shown that teens whose gender identity does not align with their sex assigned at birth were more likely to report a suicide attempt, compared with cisgender youth. A 2015 study has attributed this increased risk of suicide to marginalization, social exclusion, and trauma. The bills being proposed may exacerbate this, according to Dr. Rafferty.
These bills point in the direction of approaches and models that research shows may be harmful to transgender children, he said.
“Beyond that, policies that are out there affecting participation in sports and other activities really just draw attention to these youth in unnecessary ways and really puts their physical and mental health at risk in a school environment,” he added.
“Whether it’s health care, school, community, or home [these policies] just really create a very hostile and dangerous environment toward a population that already is at significant risk,” Dr. Rafferty said.
The AAP’s clinical care guidelines for children who identify as transgender state that pediatricians who provide gender-affirmative care and speak with children and their families about gender issues help foster an environment of nurturance and support.
“Many people think that the care provided to gender diverse young people is rogue,” Dr. Cooper said. “Pediatric gender clinics are often either multidisciplinary or have a readily accessible team, including social work, mental health, and the physician. Medical care for gender diverse youth follows well-established standards of care.”
Dr. Rafferty said it is important for care providers to support children and teenagers who identify as transgender by creating a safe space and helping them identify what they need.
“There’s a lot out there that really tries to unite gender and biological sex together, saying that gender can be reduced to your anatomy,” Dr. Rafferty said. “But I think what we’re coming to appreciate is that our sense of identity is much more complex than simply our genes or biology.”
Dr. Rafferty is a member of the American Academy of Pediatrics, the Society of Adolescent Health and Medicine, and the American Academy of Child and Adolescent Psychiatry. Dr. Cooper is a columnist for Pediatric News and is on the board of trustees for the Texas Medical Association.
The American Academy of Pediatrics (AAP) released a statement condemning state legislation introduced across the country that would prohibit the medical treatment of children who identify as transgender.
“Politics has no place here. Transgender children, like all children, just want to belong. We will fight state by state, in the courts, and on the national stage to make sure they know they do,” said the American Academy of Pediatrics, an organization that recommends that youth who identify as transgender have access to comprehensive, gender-affirming and “developmentally appropriate” health care.
There has been a lot of controversy among physicians surrounding gender-affirming care for children. In 2019, three separate groups of physicians questioned the hormonal treatment of children and adolescents with gender dysphoria, saying more research is needed to support gender-affirming care recommendations from groups such as the AAP. In addition, California-based endocrinologist Michael K. Laidlaw, MD, called the use of puberty blockers – medication used to delay or prevent the onset of puberty – an “experiment” and “public health problem.”
Some of the bills circulating would prohibit gender-affirming care for children and penalize pediatricians and other physicians for providing such care. Other bills would prevent transgender youth from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Jason Rafferty, MD, a pediatrician who authored AAP’s 2018 policy statement encouraging pediatricians to provide gender-affirmative care to children and adolescents, said in an interview that these proposed state bills are targeting a vulnerable population at a vulnerable time.
“You have this marginalized group and right in the midst of a pandemic – and [social isolation] – and you have these policies coming out that are really targeting the most vulnerable,” said Dr. Rafferty, who practices at the Adolescent Healthcare Center at Hasbro Children’s Hospital, Providence, R.I. “Some of the proposed policies would ensure that some of these transgender kids wouldn’t have access to basic medical care [or access to areas] where pediatricians are affirming these kids and creating a safe environment for them.”
M. Brett Cooper, MD, of pediatrics at UT Southwestern Medical Center, Dallas, said in an interview that the proposed state legislations are based on personal beliefs, not evidence-based care.
He said the AAP is speaking out because many of these proposed state legislatures are advancing bills that would affect gender-diverse children.
“Many of these bills are making it close to passing both chambers of a state legislature and thus sent to the governor for approval,” Dr. Cooper said. “In past legislative sessions, bills such as these have never it made out of committee in either a state House or state Senate.”
Early in March, South Dakota Governor Kristi Noem tweeted that she was “excited to sign” a bill that would prevent children who are transgender from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Meanwhile, previous studies have shown that teens whose gender identity does not align with their sex assigned at birth were more likely to report a suicide attempt, compared with cisgender youth. A 2015 study has attributed this increased risk of suicide to marginalization, social exclusion, and trauma. The bills being proposed may exacerbate this, according to Dr. Rafferty.
These bills point in the direction of approaches and models that research shows may be harmful to transgender children, he said.
“Beyond that, policies that are out there affecting participation in sports and other activities really just draw attention to these youth in unnecessary ways and really puts their physical and mental health at risk in a school environment,” he added.
“Whether it’s health care, school, community, or home [these policies] just really create a very hostile and dangerous environment toward a population that already is at significant risk,” Dr. Rafferty said.
The AAP’s clinical care guidelines for children who identify as transgender state that pediatricians who provide gender-affirmative care and speak with children and their families about gender issues help foster an environment of nurturance and support.
“Many people think that the care provided to gender diverse young people is rogue,” Dr. Cooper said. “Pediatric gender clinics are often either multidisciplinary or have a readily accessible team, including social work, mental health, and the physician. Medical care for gender diverse youth follows well-established standards of care.”
Dr. Rafferty said it is important for care providers to support children and teenagers who identify as transgender by creating a safe space and helping them identify what they need.
“There’s a lot out there that really tries to unite gender and biological sex together, saying that gender can be reduced to your anatomy,” Dr. Rafferty said. “But I think what we’re coming to appreciate is that our sense of identity is much more complex than simply our genes or biology.”
Dr. Rafferty is a member of the American Academy of Pediatrics, the Society of Adolescent Health and Medicine, and the American Academy of Child and Adolescent Psychiatry. Dr. Cooper is a columnist for Pediatric News and is on the board of trustees for the Texas Medical Association.
COVID-19 ‘long-haul’ symptoms overlap with ME/CFS
People experiencing long-term symptoms following acute COVID-19 infection are increasingly meeting criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a phenomenon that highlights the need for unified research and clinical approaches, speakers said at a press briefing March 25 held by the advocacy group MEAction.
“Post-COVID lingering illness was predictable. Similar lingering fatigue syndromes have been reported in the scientific literature for nearly 100 years, following a variety of well-documented infections with viruses, bacteria, fungi, and even protozoa,” said Anthony Komaroff, MD, professor of medicine at Harvard Medical School, Boston.
Core criteria for ME/CFS established by the Institute of Medicine in 2015 include substantial decrement in functioning for at least 6 months, postexertional malaise (PEM), or a worsening of symptoms following even minor exertion (often described as “crashes”), unrefreshing sleep, and cognitive impairment and/or orthostatic intolerance.
Patients with ME/CFS also commonly experience painful headaches, muscle or joint aches, and allergies/other sensitivities. Although many patients can trace their symptoms to an initiating infection, “the cause is often unclear because the diagnosis is often delayed for months or years after symptom onset,” said Lucinda Bateman, MD, founder of the Bateman Horne Center, Salt Lake City, who leads a clinician coalition that aims to improve ME/CFS management.
In an international survey of 3762 COVID-19 “long-haulers” published in a preprint in December of 2020, the most frequent symptoms reported at least 6 months after illness onset were fatigue in 78%, PEM in 72%, and cognitive dysfunction (“brain fog”) in 55%. At the time of the survey, 45% reported requiring reduced work schedules because of their illness, and 22% reported being unable to work at all.
Dr. Bateman said those findings align with her experience so far with 12 COVID-19 “long haulers” who self-referred to her ME/CFS and fibromyalgia specialty clinic. Nine of the 12 met criteria for postural orthostatic tachycardia syndrome (POTS) based on the 10-minute NASA Lean Test, she said, and half also met the 2016 American College of Rheumatology criteria for fibromyalgia.
“Some were severely impaired. We suspect a small fiber polyneuropathy in about half, and mast cell activation syndrome in more than half. We look forward to doing more testing,” Dr. Bateman said.
To be sure, Dr. Komaroff noted, there are some differences. “Long COVID” patients will often experience breathlessness and ongoing anosmia (loss of taste and smell), which aren’t typical of ME/CFS.
But, he said, “many of the symptoms are quite similar ... My guess is that ME/CFS is an illness with a final common pathway that can be triggered by different things,” said Dr. Komaroff, a senior physician at Brigham and Women’s Hospital in Boston, and editor-in-chief of the Harvard Health Letter.
Based on previous data about CFS suggesting a 10% rate of symptoms persisting at least a year following a variety of infectious agents and the predicted 200 million COVID-19 cases globally by the end of 2021, Dr. Komaroff estimated that about 20 million cases of “long COVID” would be expected in the next year.
‘A huge investment’
On the research side, the National Institutes of Health recently appropriated $1.15 billion dollars over the next 4 years to investigate “the heterogeneity in the recovery process after COVID and to develop treatments for those suffering from [postacute COVID-19 syndrome]” according to a Feb. 5, 2021, blog from the National Institute of Neurological Disorders and Stroke (NINDS).
That same day, another NINDS blog announced “new resources for large-scale ME/CFS research” and emphasized the tie-in with long–COVID-19 syndrome.
“That’s a huge investment. In my opinion, there will be several lingering illnesses following COVID,” Dr. Komaroff said, adding, “It’s my bet that long COVID will prove to be caused by certain kinds of abnormalities in the brain, some of the same abnormalities already identified in ME/CFS. Research will determine whether that’s right or wrong.”
In 2017, NINDS had announced a large increase in funding for ME/CFS research, including the creation of four dedicated research centers. In April 2019, NINDS held a 2-day conference highlighting that ongoing work, as reported by Medscape Medical News.
During the briefing, NINDS clinical director Avindra Nath, MD, described a comprehensive ongoing ME/CFS intramural study he’s been leading since 2016.
He’s now also overseeing two long–COVID-19 studies, one of which has a protocol similar to that of the ME/CFS study and will include individuals who are still experiencing long-term symptoms following confirmed cases of COVID-19. The aim is to screen about 1,300 patients. Several task forces are now examining all of these data together.
“Each aspect is now being analyzed … What we learn from one applies to the other,” Dr. Nath said.
Advice for clinicians
In interviews, Dr. Bateman and Dr. Nath offered clinical advice for managing patients who meet ME/CFS criteria, whether they had confirmed or suspected COVID-19, a different infection, or unknown trigger(s).
Dr. Bateman advised that clinicians assess patients for each of the symptoms individually. “Besides exercise intolerance and PEM, the most commonly missed is orthostatic intolerance. It really doesn’t matter what the cause is, it’s amenable to supportive treatment. It’s one aspect of the illness that contributes to severely impaired function. My plea to all physicians would be for sure to assess for [orthostatic intolerance], and gain an understanding about activity management and avoiding PEM symptoms.”
Dr. Nath noted that an often-challenging situation is when tests for the infectious agent and other blood work come back negative, yet the patient still reports multiple debilitating symptoms. This has been a particular issue with long COVID-19, since many patients became ill early in the pandemic before the polymerase chain reaction (PCR) tests for SARS-CoV-2 were widely available.
“The physician can only order tests that are available at their labs. I think what the physician should do is handle symptoms symptomatically but also refer patients to specialists who are taking care of these patients or to research studies,” he said.
Dr. Bateman added, “Whether they had a documented COVID infection – we just have to let go of that in 2020. Way too many people didn’t have access to a test or the timing wasn’t amenable. If people meet criteria for ME/CFS, it’s irrelevant … It’s mainly a clinical diagnosis. It’s not reliant on identifying the infectious trigger.”
Dr. Komaroff, who began caring for then-termed “chronic fatigue syndrome” patients and researching the condition more than 30 years ago, said that “every cloud has its silver lining. The increased focus on postinfectious fatigue syndrome is a silver lining in my mind around the terrible dark cloud that is the pandemic of COVID.”
Dr. Komaroff has received personal fees from Serimmune Inc., Ono Pharma, and Deallus, and grants from the NIH. Dr. Bateman is employed by the Bateman Horne Center, which receives grants from the NIH, and fees from Exagen Inc., and Teva Pharmaceutical. Dr. Nath is an NIH employee.
A version of this article first appeared on Medscape.com.
People experiencing long-term symptoms following acute COVID-19 infection are increasingly meeting criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a phenomenon that highlights the need for unified research and clinical approaches, speakers said at a press briefing March 25 held by the advocacy group MEAction.
“Post-COVID lingering illness was predictable. Similar lingering fatigue syndromes have been reported in the scientific literature for nearly 100 years, following a variety of well-documented infections with viruses, bacteria, fungi, and even protozoa,” said Anthony Komaroff, MD, professor of medicine at Harvard Medical School, Boston.
Core criteria for ME/CFS established by the Institute of Medicine in 2015 include substantial decrement in functioning for at least 6 months, postexertional malaise (PEM), or a worsening of symptoms following even minor exertion (often described as “crashes”), unrefreshing sleep, and cognitive impairment and/or orthostatic intolerance.
Patients with ME/CFS also commonly experience painful headaches, muscle or joint aches, and allergies/other sensitivities. Although many patients can trace their symptoms to an initiating infection, “the cause is often unclear because the diagnosis is often delayed for months or years after symptom onset,” said Lucinda Bateman, MD, founder of the Bateman Horne Center, Salt Lake City, who leads a clinician coalition that aims to improve ME/CFS management.
In an international survey of 3762 COVID-19 “long-haulers” published in a preprint in December of 2020, the most frequent symptoms reported at least 6 months after illness onset were fatigue in 78%, PEM in 72%, and cognitive dysfunction (“brain fog”) in 55%. At the time of the survey, 45% reported requiring reduced work schedules because of their illness, and 22% reported being unable to work at all.
Dr. Bateman said those findings align with her experience so far with 12 COVID-19 “long haulers” who self-referred to her ME/CFS and fibromyalgia specialty clinic. Nine of the 12 met criteria for postural orthostatic tachycardia syndrome (POTS) based on the 10-minute NASA Lean Test, she said, and half also met the 2016 American College of Rheumatology criteria for fibromyalgia.
“Some were severely impaired. We suspect a small fiber polyneuropathy in about half, and mast cell activation syndrome in more than half. We look forward to doing more testing,” Dr. Bateman said.
To be sure, Dr. Komaroff noted, there are some differences. “Long COVID” patients will often experience breathlessness and ongoing anosmia (loss of taste and smell), which aren’t typical of ME/CFS.
But, he said, “many of the symptoms are quite similar ... My guess is that ME/CFS is an illness with a final common pathway that can be triggered by different things,” said Dr. Komaroff, a senior physician at Brigham and Women’s Hospital in Boston, and editor-in-chief of the Harvard Health Letter.
Based on previous data about CFS suggesting a 10% rate of symptoms persisting at least a year following a variety of infectious agents and the predicted 200 million COVID-19 cases globally by the end of 2021, Dr. Komaroff estimated that about 20 million cases of “long COVID” would be expected in the next year.
‘A huge investment’
On the research side, the National Institutes of Health recently appropriated $1.15 billion dollars over the next 4 years to investigate “the heterogeneity in the recovery process after COVID and to develop treatments for those suffering from [postacute COVID-19 syndrome]” according to a Feb. 5, 2021, blog from the National Institute of Neurological Disorders and Stroke (NINDS).
That same day, another NINDS blog announced “new resources for large-scale ME/CFS research” and emphasized the tie-in with long–COVID-19 syndrome.
“That’s a huge investment. In my opinion, there will be several lingering illnesses following COVID,” Dr. Komaroff said, adding, “It’s my bet that long COVID will prove to be caused by certain kinds of abnormalities in the brain, some of the same abnormalities already identified in ME/CFS. Research will determine whether that’s right or wrong.”
In 2017, NINDS had announced a large increase in funding for ME/CFS research, including the creation of four dedicated research centers. In April 2019, NINDS held a 2-day conference highlighting that ongoing work, as reported by Medscape Medical News.
During the briefing, NINDS clinical director Avindra Nath, MD, described a comprehensive ongoing ME/CFS intramural study he’s been leading since 2016.
He’s now also overseeing two long–COVID-19 studies, one of which has a protocol similar to that of the ME/CFS study and will include individuals who are still experiencing long-term symptoms following confirmed cases of COVID-19. The aim is to screen about 1,300 patients. Several task forces are now examining all of these data together.
“Each aspect is now being analyzed … What we learn from one applies to the other,” Dr. Nath said.
Advice for clinicians
In interviews, Dr. Bateman and Dr. Nath offered clinical advice for managing patients who meet ME/CFS criteria, whether they had confirmed or suspected COVID-19, a different infection, or unknown trigger(s).
Dr. Bateman advised that clinicians assess patients for each of the symptoms individually. “Besides exercise intolerance and PEM, the most commonly missed is orthostatic intolerance. It really doesn’t matter what the cause is, it’s amenable to supportive treatment. It’s one aspect of the illness that contributes to severely impaired function. My plea to all physicians would be for sure to assess for [orthostatic intolerance], and gain an understanding about activity management and avoiding PEM symptoms.”
Dr. Nath noted that an often-challenging situation is when tests for the infectious agent and other blood work come back negative, yet the patient still reports multiple debilitating symptoms. This has been a particular issue with long COVID-19, since many patients became ill early in the pandemic before the polymerase chain reaction (PCR) tests for SARS-CoV-2 were widely available.
“The physician can only order tests that are available at their labs. I think what the physician should do is handle symptoms symptomatically but also refer patients to specialists who are taking care of these patients or to research studies,” he said.
Dr. Bateman added, “Whether they had a documented COVID infection – we just have to let go of that in 2020. Way too many people didn’t have access to a test or the timing wasn’t amenable. If people meet criteria for ME/CFS, it’s irrelevant … It’s mainly a clinical diagnosis. It’s not reliant on identifying the infectious trigger.”
Dr. Komaroff, who began caring for then-termed “chronic fatigue syndrome” patients and researching the condition more than 30 years ago, said that “every cloud has its silver lining. The increased focus on postinfectious fatigue syndrome is a silver lining in my mind around the terrible dark cloud that is the pandemic of COVID.”
Dr. Komaroff has received personal fees from Serimmune Inc., Ono Pharma, and Deallus, and grants from the NIH. Dr. Bateman is employed by the Bateman Horne Center, which receives grants from the NIH, and fees from Exagen Inc., and Teva Pharmaceutical. Dr. Nath is an NIH employee.
A version of this article first appeared on Medscape.com.
People experiencing long-term symptoms following acute COVID-19 infection are increasingly meeting criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a phenomenon that highlights the need for unified research and clinical approaches, speakers said at a press briefing March 25 held by the advocacy group MEAction.
“Post-COVID lingering illness was predictable. Similar lingering fatigue syndromes have been reported in the scientific literature for nearly 100 years, following a variety of well-documented infections with viruses, bacteria, fungi, and even protozoa,” said Anthony Komaroff, MD, professor of medicine at Harvard Medical School, Boston.
Core criteria for ME/CFS established by the Institute of Medicine in 2015 include substantial decrement in functioning for at least 6 months, postexertional malaise (PEM), or a worsening of symptoms following even minor exertion (often described as “crashes”), unrefreshing sleep, and cognitive impairment and/or orthostatic intolerance.
Patients with ME/CFS also commonly experience painful headaches, muscle or joint aches, and allergies/other sensitivities. Although many patients can trace their symptoms to an initiating infection, “the cause is often unclear because the diagnosis is often delayed for months or years after symptom onset,” said Lucinda Bateman, MD, founder of the Bateman Horne Center, Salt Lake City, who leads a clinician coalition that aims to improve ME/CFS management.
In an international survey of 3762 COVID-19 “long-haulers” published in a preprint in December of 2020, the most frequent symptoms reported at least 6 months after illness onset were fatigue in 78%, PEM in 72%, and cognitive dysfunction (“brain fog”) in 55%. At the time of the survey, 45% reported requiring reduced work schedules because of their illness, and 22% reported being unable to work at all.
Dr. Bateman said those findings align with her experience so far with 12 COVID-19 “long haulers” who self-referred to her ME/CFS and fibromyalgia specialty clinic. Nine of the 12 met criteria for postural orthostatic tachycardia syndrome (POTS) based on the 10-minute NASA Lean Test, she said, and half also met the 2016 American College of Rheumatology criteria for fibromyalgia.
“Some were severely impaired. We suspect a small fiber polyneuropathy in about half, and mast cell activation syndrome in more than half. We look forward to doing more testing,” Dr. Bateman said.
To be sure, Dr. Komaroff noted, there are some differences. “Long COVID” patients will often experience breathlessness and ongoing anosmia (loss of taste and smell), which aren’t typical of ME/CFS.
But, he said, “many of the symptoms are quite similar ... My guess is that ME/CFS is an illness with a final common pathway that can be triggered by different things,” said Dr. Komaroff, a senior physician at Brigham and Women’s Hospital in Boston, and editor-in-chief of the Harvard Health Letter.
Based on previous data about CFS suggesting a 10% rate of symptoms persisting at least a year following a variety of infectious agents and the predicted 200 million COVID-19 cases globally by the end of 2021, Dr. Komaroff estimated that about 20 million cases of “long COVID” would be expected in the next year.
‘A huge investment’
On the research side, the National Institutes of Health recently appropriated $1.15 billion dollars over the next 4 years to investigate “the heterogeneity in the recovery process after COVID and to develop treatments for those suffering from [postacute COVID-19 syndrome]” according to a Feb. 5, 2021, blog from the National Institute of Neurological Disorders and Stroke (NINDS).
That same day, another NINDS blog announced “new resources for large-scale ME/CFS research” and emphasized the tie-in with long–COVID-19 syndrome.
“That’s a huge investment. In my opinion, there will be several lingering illnesses following COVID,” Dr. Komaroff said, adding, “It’s my bet that long COVID will prove to be caused by certain kinds of abnormalities in the brain, some of the same abnormalities already identified in ME/CFS. Research will determine whether that’s right or wrong.”
In 2017, NINDS had announced a large increase in funding for ME/CFS research, including the creation of four dedicated research centers. In April 2019, NINDS held a 2-day conference highlighting that ongoing work, as reported by Medscape Medical News.
During the briefing, NINDS clinical director Avindra Nath, MD, described a comprehensive ongoing ME/CFS intramural study he’s been leading since 2016.
He’s now also overseeing two long–COVID-19 studies, one of which has a protocol similar to that of the ME/CFS study and will include individuals who are still experiencing long-term symptoms following confirmed cases of COVID-19. The aim is to screen about 1,300 patients. Several task forces are now examining all of these data together.
“Each aspect is now being analyzed … What we learn from one applies to the other,” Dr. Nath said.
Advice for clinicians
In interviews, Dr. Bateman and Dr. Nath offered clinical advice for managing patients who meet ME/CFS criteria, whether they had confirmed or suspected COVID-19, a different infection, or unknown trigger(s).
Dr. Bateman advised that clinicians assess patients for each of the symptoms individually. “Besides exercise intolerance and PEM, the most commonly missed is orthostatic intolerance. It really doesn’t matter what the cause is, it’s amenable to supportive treatment. It’s one aspect of the illness that contributes to severely impaired function. My plea to all physicians would be for sure to assess for [orthostatic intolerance], and gain an understanding about activity management and avoiding PEM symptoms.”
Dr. Nath noted that an often-challenging situation is when tests for the infectious agent and other blood work come back negative, yet the patient still reports multiple debilitating symptoms. This has been a particular issue with long COVID-19, since many patients became ill early in the pandemic before the polymerase chain reaction (PCR) tests for SARS-CoV-2 were widely available.
“The physician can only order tests that are available at their labs. I think what the physician should do is handle symptoms symptomatically but also refer patients to specialists who are taking care of these patients or to research studies,” he said.
Dr. Bateman added, “Whether they had a documented COVID infection – we just have to let go of that in 2020. Way too many people didn’t have access to a test or the timing wasn’t amenable. If people meet criteria for ME/CFS, it’s irrelevant … It’s mainly a clinical diagnosis. It’s not reliant on identifying the infectious trigger.”
Dr. Komaroff, who began caring for then-termed “chronic fatigue syndrome” patients and researching the condition more than 30 years ago, said that “every cloud has its silver lining. The increased focus on postinfectious fatigue syndrome is a silver lining in my mind around the terrible dark cloud that is the pandemic of COVID.”
Dr. Komaroff has received personal fees from Serimmune Inc., Ono Pharma, and Deallus, and grants from the NIH. Dr. Bateman is employed by the Bateman Horne Center, which receives grants from the NIH, and fees from Exagen Inc., and Teva Pharmaceutical. Dr. Nath is an NIH employee.
A version of this article first appeared on Medscape.com.
Maternal caffeine consumption, even small amounts, may reduce neonatal size
For pregnant women, just half a cup of coffee a day may reduce neonatal birth size and body weight, according to a prospective study involving more than 2,500 women.
That’s only 50 mg of a caffeine day, which falls below the upper threshold of 200 mg set by the American College of Obstetricians and Gynecologists, lead author Jessica Gleason, PhD, MPH, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Bethesda, Md, and colleagues reported.
“Systematic reviews and meta-analyses have reported that maternal caffeine consumption, even in doses lower than 200 mg, is associated with a higher risk for low birth weight, small for gestational age (SGA), and fetal growth restriction, suggesting there may be no safe amount of caffeine during pregnancy,” the investigators wrote in JAMA Network Open.
Findings to date have been inconsistent, with a 2014 meta-analysis reporting contrary or null results in four out of nine studies.
Dr. Gleason and colleagues suggested that such discrepancies may be caused by uncontrolled confounding factors in some of the studies, such as smoking, as well as the inadequacy of self-reporting, which fails to incorporate variations in caffeine content between beverages, or differences in rates of metabolism between individuals.
“To our knowledge, no studies have examined the association between caffeine intake and neonatal anthropometric measures beyond weight, length, and head circumference, and few have analyzed plasma concentrations of caffeine and its metabolites or genetic variations in the rate of metabolism associated with neonatal size,” the investigators wrote.
Dr. Gleason and colleagues set out to address this knowledge gap with a prospective cohort study, including 2,055 nonsmoking women with low risk of birth defects who presented at 12 centers between 2009 and 2013. Mean participant age was 28.3 years and mean body mass index was 23.6. Races and ethnicities were represented almost evenly even across four groups: Hispanic (28.2%), White (27.4%), Black (25.2%), and Asian/Pacific Islander (19.2%). Rate of caffeine metabolism was defined by the single-nucleotide variant rs762551 (CYP1A2*1F), according to which, slightly more women had slow metabolism (52.7%) than fast metabolism (47.3%).
Women were enrolled at 8-13 weeks’ gestational age, at which time they underwent interviews and blood draws, allowing for measurement of caffeine and paraxanthine plasma levels, as well as self-reported caffeine consumption during the preceding week.
Over the course of six visits, fetal growth was observed via ultrasound. Medical records were used to determine birth weights and neonatal anthropometric measures, including fat and skin fold mass, body length, and circumferences of the thigh, arm, abdomen, and head.
Neonatal measurements were compared with plasma levels of caffeine and paraxanthine, both continuously and as quartiles (Q1, ≤ 28.3 ng/mL; Q2, 28.4-157.1 ng/mL; Q3, 157.2-658.8 ng/mL; Q4, > 658.8 ng/mL). Comparisons were also made with self-reported caffeine intake.
Women who reported drinking 1-50 mg of caffeine per day had neonates with smaller subscapular skin folds (beta = –0.14 mm; 95% confidence interval, –0.27 to -–0.01 mm), while those who reported more than 50 mg per day had newborns with lower birth weight (beta = –66 g; 95% CI, –121 to –10 g), and smaller circumferences of mid-upper thigh (beta = –0.32 cm; 95% CI, –0.55 to –0.09 cm), anterior thigh skin fold (beta = –0.24 mm; 95% CI, –0.47 to -.01 mm), and mid-upper arm (beta = –0.17 cm; 95% CI, –0.31 to –0.02 cm).
Caffeine plasma concentrations supported these findings.
Compared with women who had caffeine plasma concentrations in the lowest quartile, those in the highest quartile gave birth to neonates with shorter length (beta = –0.44 cm; P = .04 for trend) and lower body weight (beta = –84.3 g; P = .04 for trend), as well as smaller mid-upper arm circumference (beta = -0.25 cm; P = .02 for trend), mid-upper thigh circumference (beta = –0.29 cm; P = .07 for trend), and head circumference (beta = –0.28 cm; P < .001 for trend). A comparison of lower and upper paraxanthine quartiles revealed the similar trends, as did analyses of continuous measures.
“Our results suggest that caffeine consumption during pregnancy, even at levels much lower than the recommended 200 mg per day of caffeine may be associated with decreased fetal growth,” the investigators concluded.
Sarah W. Prager, MD, of the University of Washington, Seattle, suggested that the findings “do not demonstrate that caffeine has a clinically meaningful negative clinical impact on newborn size and weight.”
She noted that there was no difference in the rate of SGA between plasma caffeine quartiles, and that most patients were thin, which may not accurately represent the U.S. population.
“Based on these new data, my take home message to patients would be that increasing amounts of caffeine can have a small but real impact on the size of their baby at birth, though it is unlikely to result in a diagnosis of SGA,” she said. “Pregnant patients may want to limit caffeine intake even more than the ACOG recommendation of 200 mg per day.”
According to Robert M. Silver, MD, of the University of Utah Health Sciences Center, Salt Lake City, “data from this study are of high quality, owing to the prospective cohort design, large numbers, assessment of biomarkers, and sophisticated analyses.”
Still, he urged a cautious interpretation from a clinical perspective.
“It is important to not overreact to these data,” he said. “The decrease in fetal growth associated with caffeine is small and may prove to be clinically meaningless. Accordingly, clinical recommendations regarding caffeine intake during pregnancy should not be modified solely based on this study.”
Dr. Silver suggested that the findings deserve additional investigation.
“These observations warrant further research about the effects of caffeine exposure during pregnancy,” he said. “Ideally, studies should assess the effect of caffeine exposure on fetal growth in various pregnancy epochs as well as on neonatal and childhood growth.”
The study was funded by the Intramural Research Program of the NICHD. Dr. Gerlanc is an employee of The Prospective Group, which was contracted to provide statistical support.
For pregnant women, just half a cup of coffee a day may reduce neonatal birth size and body weight, according to a prospective study involving more than 2,500 women.
That’s only 50 mg of a caffeine day, which falls below the upper threshold of 200 mg set by the American College of Obstetricians and Gynecologists, lead author Jessica Gleason, PhD, MPH, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Bethesda, Md, and colleagues reported.
“Systematic reviews and meta-analyses have reported that maternal caffeine consumption, even in doses lower than 200 mg, is associated with a higher risk for low birth weight, small for gestational age (SGA), and fetal growth restriction, suggesting there may be no safe amount of caffeine during pregnancy,” the investigators wrote in JAMA Network Open.
Findings to date have been inconsistent, with a 2014 meta-analysis reporting contrary or null results in four out of nine studies.
Dr. Gleason and colleagues suggested that such discrepancies may be caused by uncontrolled confounding factors in some of the studies, such as smoking, as well as the inadequacy of self-reporting, which fails to incorporate variations in caffeine content between beverages, or differences in rates of metabolism between individuals.
“To our knowledge, no studies have examined the association between caffeine intake and neonatal anthropometric measures beyond weight, length, and head circumference, and few have analyzed plasma concentrations of caffeine and its metabolites or genetic variations in the rate of metabolism associated with neonatal size,” the investigators wrote.
Dr. Gleason and colleagues set out to address this knowledge gap with a prospective cohort study, including 2,055 nonsmoking women with low risk of birth defects who presented at 12 centers between 2009 and 2013. Mean participant age was 28.3 years and mean body mass index was 23.6. Races and ethnicities were represented almost evenly even across four groups: Hispanic (28.2%), White (27.4%), Black (25.2%), and Asian/Pacific Islander (19.2%). Rate of caffeine metabolism was defined by the single-nucleotide variant rs762551 (CYP1A2*1F), according to which, slightly more women had slow metabolism (52.7%) than fast metabolism (47.3%).
Women were enrolled at 8-13 weeks’ gestational age, at which time they underwent interviews and blood draws, allowing for measurement of caffeine and paraxanthine plasma levels, as well as self-reported caffeine consumption during the preceding week.
Over the course of six visits, fetal growth was observed via ultrasound. Medical records were used to determine birth weights and neonatal anthropometric measures, including fat and skin fold mass, body length, and circumferences of the thigh, arm, abdomen, and head.
Neonatal measurements were compared with plasma levels of caffeine and paraxanthine, both continuously and as quartiles (Q1, ≤ 28.3 ng/mL; Q2, 28.4-157.1 ng/mL; Q3, 157.2-658.8 ng/mL; Q4, > 658.8 ng/mL). Comparisons were also made with self-reported caffeine intake.
Women who reported drinking 1-50 mg of caffeine per day had neonates with smaller subscapular skin folds (beta = –0.14 mm; 95% confidence interval, –0.27 to -–0.01 mm), while those who reported more than 50 mg per day had newborns with lower birth weight (beta = –66 g; 95% CI, –121 to –10 g), and smaller circumferences of mid-upper thigh (beta = –0.32 cm; 95% CI, –0.55 to –0.09 cm), anterior thigh skin fold (beta = –0.24 mm; 95% CI, –0.47 to -.01 mm), and mid-upper arm (beta = –0.17 cm; 95% CI, –0.31 to –0.02 cm).
Caffeine plasma concentrations supported these findings.
Compared with women who had caffeine plasma concentrations in the lowest quartile, those in the highest quartile gave birth to neonates with shorter length (beta = –0.44 cm; P = .04 for trend) and lower body weight (beta = –84.3 g; P = .04 for trend), as well as smaller mid-upper arm circumference (beta = -0.25 cm; P = .02 for trend), mid-upper thigh circumference (beta = –0.29 cm; P = .07 for trend), and head circumference (beta = –0.28 cm; P < .001 for trend). A comparison of lower and upper paraxanthine quartiles revealed the similar trends, as did analyses of continuous measures.
“Our results suggest that caffeine consumption during pregnancy, even at levels much lower than the recommended 200 mg per day of caffeine may be associated with decreased fetal growth,” the investigators concluded.
Sarah W. Prager, MD, of the University of Washington, Seattle, suggested that the findings “do not demonstrate that caffeine has a clinically meaningful negative clinical impact on newborn size and weight.”
She noted that there was no difference in the rate of SGA between plasma caffeine quartiles, and that most patients were thin, which may not accurately represent the U.S. population.
“Based on these new data, my take home message to patients would be that increasing amounts of caffeine can have a small but real impact on the size of their baby at birth, though it is unlikely to result in a diagnosis of SGA,” she said. “Pregnant patients may want to limit caffeine intake even more than the ACOG recommendation of 200 mg per day.”
According to Robert M. Silver, MD, of the University of Utah Health Sciences Center, Salt Lake City, “data from this study are of high quality, owing to the prospective cohort design, large numbers, assessment of biomarkers, and sophisticated analyses.”
Still, he urged a cautious interpretation from a clinical perspective.
“It is important to not overreact to these data,” he said. “The decrease in fetal growth associated with caffeine is small and may prove to be clinically meaningless. Accordingly, clinical recommendations regarding caffeine intake during pregnancy should not be modified solely based on this study.”
Dr. Silver suggested that the findings deserve additional investigation.
“These observations warrant further research about the effects of caffeine exposure during pregnancy,” he said. “Ideally, studies should assess the effect of caffeine exposure on fetal growth in various pregnancy epochs as well as on neonatal and childhood growth.”
The study was funded by the Intramural Research Program of the NICHD. Dr. Gerlanc is an employee of The Prospective Group, which was contracted to provide statistical support.
For pregnant women, just half a cup of coffee a day may reduce neonatal birth size and body weight, according to a prospective study involving more than 2,500 women.
That’s only 50 mg of a caffeine day, which falls below the upper threshold of 200 mg set by the American College of Obstetricians and Gynecologists, lead author Jessica Gleason, PhD, MPH, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Bethesda, Md, and colleagues reported.
“Systematic reviews and meta-analyses have reported that maternal caffeine consumption, even in doses lower than 200 mg, is associated with a higher risk for low birth weight, small for gestational age (SGA), and fetal growth restriction, suggesting there may be no safe amount of caffeine during pregnancy,” the investigators wrote in JAMA Network Open.
Findings to date have been inconsistent, with a 2014 meta-analysis reporting contrary or null results in four out of nine studies.
Dr. Gleason and colleagues suggested that such discrepancies may be caused by uncontrolled confounding factors in some of the studies, such as smoking, as well as the inadequacy of self-reporting, which fails to incorporate variations in caffeine content between beverages, or differences in rates of metabolism between individuals.
“To our knowledge, no studies have examined the association between caffeine intake and neonatal anthropometric measures beyond weight, length, and head circumference, and few have analyzed plasma concentrations of caffeine and its metabolites or genetic variations in the rate of metabolism associated with neonatal size,” the investigators wrote.
Dr. Gleason and colleagues set out to address this knowledge gap with a prospective cohort study, including 2,055 nonsmoking women with low risk of birth defects who presented at 12 centers between 2009 and 2013. Mean participant age was 28.3 years and mean body mass index was 23.6. Races and ethnicities were represented almost evenly even across four groups: Hispanic (28.2%), White (27.4%), Black (25.2%), and Asian/Pacific Islander (19.2%). Rate of caffeine metabolism was defined by the single-nucleotide variant rs762551 (CYP1A2*1F), according to which, slightly more women had slow metabolism (52.7%) than fast metabolism (47.3%).
Women were enrolled at 8-13 weeks’ gestational age, at which time they underwent interviews and blood draws, allowing for measurement of caffeine and paraxanthine plasma levels, as well as self-reported caffeine consumption during the preceding week.
Over the course of six visits, fetal growth was observed via ultrasound. Medical records were used to determine birth weights and neonatal anthropometric measures, including fat and skin fold mass, body length, and circumferences of the thigh, arm, abdomen, and head.
Neonatal measurements were compared with plasma levels of caffeine and paraxanthine, both continuously and as quartiles (Q1, ≤ 28.3 ng/mL; Q2, 28.4-157.1 ng/mL; Q3, 157.2-658.8 ng/mL; Q4, > 658.8 ng/mL). Comparisons were also made with self-reported caffeine intake.
Women who reported drinking 1-50 mg of caffeine per day had neonates with smaller subscapular skin folds (beta = –0.14 mm; 95% confidence interval, –0.27 to -–0.01 mm), while those who reported more than 50 mg per day had newborns with lower birth weight (beta = –66 g; 95% CI, –121 to –10 g), and smaller circumferences of mid-upper thigh (beta = –0.32 cm; 95% CI, –0.55 to –0.09 cm), anterior thigh skin fold (beta = –0.24 mm; 95% CI, –0.47 to -.01 mm), and mid-upper arm (beta = –0.17 cm; 95% CI, –0.31 to –0.02 cm).
Caffeine plasma concentrations supported these findings.
Compared with women who had caffeine plasma concentrations in the lowest quartile, those in the highest quartile gave birth to neonates with shorter length (beta = –0.44 cm; P = .04 for trend) and lower body weight (beta = –84.3 g; P = .04 for trend), as well as smaller mid-upper arm circumference (beta = -0.25 cm; P = .02 for trend), mid-upper thigh circumference (beta = –0.29 cm; P = .07 for trend), and head circumference (beta = –0.28 cm; P < .001 for trend). A comparison of lower and upper paraxanthine quartiles revealed the similar trends, as did analyses of continuous measures.
“Our results suggest that caffeine consumption during pregnancy, even at levels much lower than the recommended 200 mg per day of caffeine may be associated with decreased fetal growth,” the investigators concluded.
Sarah W. Prager, MD, of the University of Washington, Seattle, suggested that the findings “do not demonstrate that caffeine has a clinically meaningful negative clinical impact on newborn size and weight.”
She noted that there was no difference in the rate of SGA between plasma caffeine quartiles, and that most patients were thin, which may not accurately represent the U.S. population.
“Based on these new data, my take home message to patients would be that increasing amounts of caffeine can have a small but real impact on the size of their baby at birth, though it is unlikely to result in a diagnosis of SGA,” she said. “Pregnant patients may want to limit caffeine intake even more than the ACOG recommendation of 200 mg per day.”
According to Robert M. Silver, MD, of the University of Utah Health Sciences Center, Salt Lake City, “data from this study are of high quality, owing to the prospective cohort design, large numbers, assessment of biomarkers, and sophisticated analyses.”
Still, he urged a cautious interpretation from a clinical perspective.
“It is important to not overreact to these data,” he said. “The decrease in fetal growth associated with caffeine is small and may prove to be clinically meaningless. Accordingly, clinical recommendations regarding caffeine intake during pregnancy should not be modified solely based on this study.”
Dr. Silver suggested that the findings deserve additional investigation.
“These observations warrant further research about the effects of caffeine exposure during pregnancy,” he said. “Ideally, studies should assess the effect of caffeine exposure on fetal growth in various pregnancy epochs as well as on neonatal and childhood growth.”
The study was funded by the Intramural Research Program of the NICHD. Dr. Gerlanc is an employee of The Prospective Group, which was contracted to provide statistical support.
FROM JAMA NETWORK OPEN
In U.S., lockdowns added 2 pounds per month
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Vitamin D may protect against COVID-19, especially in Black patients
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Senate confirms Murthy as Surgeon General
Seven Republicans – Bill Cassidy (La.), Susan Collins (Maine), Roger Marshall (Kan.), Susan Murkowski (Alaska), Rob Portman (Ohio), Mitt Romney (Utah), and Dan Sullivan (Alaska) – joined all the Democrats and independents in the 57-43 vote approving Dr. Murthy’s nomination.
Dr. Murthy, 43, previously served as the 19th Surgeon General, from December 2014 to April 2017, when he was asked to step down by President Donald J. Trump.
Surgeons General serve 4-year terms.
During his first tenure, Dr. Murthy issued the first-ever Surgeon General’s report on the crisis of addiction and issued a call to action to doctors to help battle the opioid crisis.
When Dr. Murthy was nominated by President-elect Joseph R. Biden Jr. in December, he was acting as cochair of the incoming administration’s COVID-19 transition advisory board.
Early in 2020, before the COVID-19 pandemic hit, Dr. Murthy published a timely book: “Together: The Healing Power of Human Connection in a Sometimes Lonely World”.
He earned his bachelor’s degree from Harvard and his MD and MBA degrees from Yale. He completed his internal medicine residency at Brigham and Women’s Hospital in Boston, where he also served as a hospitalist, and later joined Harvard Medical School as a faculty member in internal medicine.
He is married to Alice Chen, MD. The couple have two children.
A version of this article first appeared on WebMD.com.
Seven Republicans – Bill Cassidy (La.), Susan Collins (Maine), Roger Marshall (Kan.), Susan Murkowski (Alaska), Rob Portman (Ohio), Mitt Romney (Utah), and Dan Sullivan (Alaska) – joined all the Democrats and independents in the 57-43 vote approving Dr. Murthy’s nomination.
Dr. Murthy, 43, previously served as the 19th Surgeon General, from December 2014 to April 2017, when he was asked to step down by President Donald J. Trump.
Surgeons General serve 4-year terms.
During his first tenure, Dr. Murthy issued the first-ever Surgeon General’s report on the crisis of addiction and issued a call to action to doctors to help battle the opioid crisis.
When Dr. Murthy was nominated by President-elect Joseph R. Biden Jr. in December, he was acting as cochair of the incoming administration’s COVID-19 transition advisory board.
Early in 2020, before the COVID-19 pandemic hit, Dr. Murthy published a timely book: “Together: The Healing Power of Human Connection in a Sometimes Lonely World”.
He earned his bachelor’s degree from Harvard and his MD and MBA degrees from Yale. He completed his internal medicine residency at Brigham and Women’s Hospital in Boston, where he also served as a hospitalist, and later joined Harvard Medical School as a faculty member in internal medicine.
He is married to Alice Chen, MD. The couple have two children.
A version of this article first appeared on WebMD.com.
Seven Republicans – Bill Cassidy (La.), Susan Collins (Maine), Roger Marshall (Kan.), Susan Murkowski (Alaska), Rob Portman (Ohio), Mitt Romney (Utah), and Dan Sullivan (Alaska) – joined all the Democrats and independents in the 57-43 vote approving Dr. Murthy’s nomination.
Dr. Murthy, 43, previously served as the 19th Surgeon General, from December 2014 to April 2017, when he was asked to step down by President Donald J. Trump.
Surgeons General serve 4-year terms.
During his first tenure, Dr. Murthy issued the first-ever Surgeon General’s report on the crisis of addiction and issued a call to action to doctors to help battle the opioid crisis.
When Dr. Murthy was nominated by President-elect Joseph R. Biden Jr. in December, he was acting as cochair of the incoming administration’s COVID-19 transition advisory board.
Early in 2020, before the COVID-19 pandemic hit, Dr. Murthy published a timely book: “Together: The Healing Power of Human Connection in a Sometimes Lonely World”.
He earned his bachelor’s degree from Harvard and his MD and MBA degrees from Yale. He completed his internal medicine residency at Brigham and Women’s Hospital in Boston, where he also served as a hospitalist, and later joined Harvard Medical School as a faculty member in internal medicine.
He is married to Alice Chen, MD. The couple have two children.
A version of this article first appeared on WebMD.com.
Preterm infant supine sleep positioning becoming more common, but racial/ethnic disparities remain
Although supine sleep positioning of preterm infants is becoming more common, racial disparities remain, according to a retrospective analysis involving more than 66,000 mothers.
Non-Hispanic Black preterm infants were 39%-56% less likely to sleep on their backs than were non-Hispanic White preterm infants, reported lead author Sunah S. Hwang, MD, MPH, of the University Colorado, Aurora, and colleagues.
According to the investigators, these findings may explain, in part, why the risk of sudden unexpected infant death (SUID) is more than twofold higher among non-Hispanic Black preterm infants than non-Hispanic White preterm infants.
“During the first year of life, one of the most effective and modifiable parental behaviors that may reduce the risk for SUID is adhering to safe infant sleep practices, including supine sleep positioning or back-sleeping,” wrote Dr. Hwang and colleagues. The report is in the Journal of Pediatrics. “For the healthy-term population, research on the racial/ethnic disparity in adherence to safe sleep practices is robust, but for preterm infants who are at much higher risk for SUID, less is known.”
To address this knowledge gap, the investigators conducted a retrospective study using data from the Pregnancy Risk Assessment Monitoring System (PRAMS), a population-based perinatal surveillance system. The final dataset involved 66,131 mothers who gave birth to preterm infants in 16 states between 2000 and 2015. The sample size was weighted to 1,020,986 mothers.
The investigators evaluated annual marginal prevalence of supine sleep positioning among two cohorts: early preterm infants (gestational age less than 34 weeks) and late preterm infants (gestational age 34-36 weeks). The primary outcome was rate of supine sleep positioning, a practice that must have been followed consistently, excluding other positions (i.e. prone or side). Mothers were grouped by race/ethnicity into four categories: non-Hispanic Black, non-Hispanic White, Hispanic, and other. Several other maternal and infant characteristics were recorded, including marital status, maternal age, education, insurance prior to birth, history of previous live birth, insurance, method of delivery, birth weight, and sex.
From 2000 to 2015, the overall adjusted odds of supine sleep positioning increased by 8.5% in the early preterm group and 5.2% in the late preterm group. This intergroup difference may be due to disparate levels of in-hospital education, the investigators suggested.
“Perhaps the longer NICU hospitalization for early preterm infants compared with late preterm infants affords greater opportunities for parental education and engagement about safe sleep practices,” they wrote.
Among early preterm infants, odds percentages increased by 7.3%, 7.7%, and 10.0% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers, respectively. For late preterm infants, respective rates increased by 5.9%, 4.8%, and 5.8% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers.
Despite these improvements, racial disparities were still observed. Non-Hispanic Black mothers reported lower rates of supine sleep positioning for both early preterm infants (odds ratio [OR], 0.61; P less than .0001) and late preterm infants (OR, 0.44; P less than .0001) compared with non-Hispanic White mothers.
These disparities seem “to be in line with racial/ethnic disparity trends in infant mortality and in SUID rates that have persisted for decades among infants,” the investigators wrote.
To a lesser degree, and lacking statistical significance, Hispanic mothers reported lower odds of supine sleep positioning than the odds of White mothers for both early preterm infants (OR, 0.80; P = .1670) and late preterm infants (OR, 0.81; P = .1054).
According to Dr. Hwang and colleagues, more specific demographic data are needed to accurately describe supine sleep positioning rates among Hispanic mothers, partly because of the heterogeneity of this cohort.
“A large body of literature has shown significant variability by immigrant status and country of origin in several infant health outcomes among the Hispanic population,” the investigators wrote. “This study was unable to stratify the Hispanic cohort by these characteristics and thus the distribution of supine sleep positioning prevalence across different Hispanic subgroups could not be demonstrated in this study.”
The investigators also suggested that interventional studies are needed.
“Additional efforts to understand the barriers and facilitators to SSP [supine sleep positioning] adherence among all preterm infant caregivers, particularly non-Hispanic Black and Hispanic parents, are needed so that novel interventions can then be developed,” they wrote.
According to Denice Cora-Bramble, MD, MBA, chief diversity officer at Children’s National Hospital and professor of pediatrics at George Washington University, Washington, the observed improvements in supine sleep positioning may predict lower rates of infant mortality, but more work in the area is needed.
“In spite of improvement in infants’ supine sleep positioning during the study period, racial/ethnic disparities persisted among non-Hispanic Blacks and Hispanics,” Dr. Cora-Bramble said. “That there was improvement among the populations included in the study is significant because of the associated and expected decrease in infant mortality. However, the study results need to be evaluated within the context of [the study’s] limitations, such as the inclusion of only sixteen states in the data analysis. More research is needed to understand and effectively address the disparities highlighted in the study.”
The investigators and Dr. Cora-Bramble reported no conflicts of interest.
Although supine sleep positioning of preterm infants is becoming more common, racial disparities remain, according to a retrospective analysis involving more than 66,000 mothers.
Non-Hispanic Black preterm infants were 39%-56% less likely to sleep on their backs than were non-Hispanic White preterm infants, reported lead author Sunah S. Hwang, MD, MPH, of the University Colorado, Aurora, and colleagues.
According to the investigators, these findings may explain, in part, why the risk of sudden unexpected infant death (SUID) is more than twofold higher among non-Hispanic Black preterm infants than non-Hispanic White preterm infants.
“During the first year of life, one of the most effective and modifiable parental behaviors that may reduce the risk for SUID is adhering to safe infant sleep practices, including supine sleep positioning or back-sleeping,” wrote Dr. Hwang and colleagues. The report is in the Journal of Pediatrics. “For the healthy-term population, research on the racial/ethnic disparity in adherence to safe sleep practices is robust, but for preterm infants who are at much higher risk for SUID, less is known.”
To address this knowledge gap, the investigators conducted a retrospective study using data from the Pregnancy Risk Assessment Monitoring System (PRAMS), a population-based perinatal surveillance system. The final dataset involved 66,131 mothers who gave birth to preterm infants in 16 states between 2000 and 2015. The sample size was weighted to 1,020,986 mothers.
The investigators evaluated annual marginal prevalence of supine sleep positioning among two cohorts: early preterm infants (gestational age less than 34 weeks) and late preterm infants (gestational age 34-36 weeks). The primary outcome was rate of supine sleep positioning, a practice that must have been followed consistently, excluding other positions (i.e. prone or side). Mothers were grouped by race/ethnicity into four categories: non-Hispanic Black, non-Hispanic White, Hispanic, and other. Several other maternal and infant characteristics were recorded, including marital status, maternal age, education, insurance prior to birth, history of previous live birth, insurance, method of delivery, birth weight, and sex.
From 2000 to 2015, the overall adjusted odds of supine sleep positioning increased by 8.5% in the early preterm group and 5.2% in the late preterm group. This intergroup difference may be due to disparate levels of in-hospital education, the investigators suggested.
“Perhaps the longer NICU hospitalization for early preterm infants compared with late preterm infants affords greater opportunities for parental education and engagement about safe sleep practices,” they wrote.
Among early preterm infants, odds percentages increased by 7.3%, 7.7%, and 10.0% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers, respectively. For late preterm infants, respective rates increased by 5.9%, 4.8%, and 5.8% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers.
Despite these improvements, racial disparities were still observed. Non-Hispanic Black mothers reported lower rates of supine sleep positioning for both early preterm infants (odds ratio [OR], 0.61; P less than .0001) and late preterm infants (OR, 0.44; P less than .0001) compared with non-Hispanic White mothers.
These disparities seem “to be in line with racial/ethnic disparity trends in infant mortality and in SUID rates that have persisted for decades among infants,” the investigators wrote.
To a lesser degree, and lacking statistical significance, Hispanic mothers reported lower odds of supine sleep positioning than the odds of White mothers for both early preterm infants (OR, 0.80; P = .1670) and late preterm infants (OR, 0.81; P = .1054).
According to Dr. Hwang and colleagues, more specific demographic data are needed to accurately describe supine sleep positioning rates among Hispanic mothers, partly because of the heterogeneity of this cohort.
“A large body of literature has shown significant variability by immigrant status and country of origin in several infant health outcomes among the Hispanic population,” the investigators wrote. “This study was unable to stratify the Hispanic cohort by these characteristics and thus the distribution of supine sleep positioning prevalence across different Hispanic subgroups could not be demonstrated in this study.”
The investigators also suggested that interventional studies are needed.
“Additional efforts to understand the barriers and facilitators to SSP [supine sleep positioning] adherence among all preterm infant caregivers, particularly non-Hispanic Black and Hispanic parents, are needed so that novel interventions can then be developed,” they wrote.
According to Denice Cora-Bramble, MD, MBA, chief diversity officer at Children’s National Hospital and professor of pediatrics at George Washington University, Washington, the observed improvements in supine sleep positioning may predict lower rates of infant mortality, but more work in the area is needed.
“In spite of improvement in infants’ supine sleep positioning during the study period, racial/ethnic disparities persisted among non-Hispanic Blacks and Hispanics,” Dr. Cora-Bramble said. “That there was improvement among the populations included in the study is significant because of the associated and expected decrease in infant mortality. However, the study results need to be evaluated within the context of [the study’s] limitations, such as the inclusion of only sixteen states in the data analysis. More research is needed to understand and effectively address the disparities highlighted in the study.”
The investigators and Dr. Cora-Bramble reported no conflicts of interest.
Although supine sleep positioning of preterm infants is becoming more common, racial disparities remain, according to a retrospective analysis involving more than 66,000 mothers.
Non-Hispanic Black preterm infants were 39%-56% less likely to sleep on their backs than were non-Hispanic White preterm infants, reported lead author Sunah S. Hwang, MD, MPH, of the University Colorado, Aurora, and colleagues.
According to the investigators, these findings may explain, in part, why the risk of sudden unexpected infant death (SUID) is more than twofold higher among non-Hispanic Black preterm infants than non-Hispanic White preterm infants.
“During the first year of life, one of the most effective and modifiable parental behaviors that may reduce the risk for SUID is adhering to safe infant sleep practices, including supine sleep positioning or back-sleeping,” wrote Dr. Hwang and colleagues. The report is in the Journal of Pediatrics. “For the healthy-term population, research on the racial/ethnic disparity in adherence to safe sleep practices is robust, but for preterm infants who are at much higher risk for SUID, less is known.”
To address this knowledge gap, the investigators conducted a retrospective study using data from the Pregnancy Risk Assessment Monitoring System (PRAMS), a population-based perinatal surveillance system. The final dataset involved 66,131 mothers who gave birth to preterm infants in 16 states between 2000 and 2015. The sample size was weighted to 1,020,986 mothers.
The investigators evaluated annual marginal prevalence of supine sleep positioning among two cohorts: early preterm infants (gestational age less than 34 weeks) and late preterm infants (gestational age 34-36 weeks). The primary outcome was rate of supine sleep positioning, a practice that must have been followed consistently, excluding other positions (i.e. prone or side). Mothers were grouped by race/ethnicity into four categories: non-Hispanic Black, non-Hispanic White, Hispanic, and other. Several other maternal and infant characteristics were recorded, including marital status, maternal age, education, insurance prior to birth, history of previous live birth, insurance, method of delivery, birth weight, and sex.
From 2000 to 2015, the overall adjusted odds of supine sleep positioning increased by 8.5% in the early preterm group and 5.2% in the late preterm group. This intergroup difference may be due to disparate levels of in-hospital education, the investigators suggested.
“Perhaps the longer NICU hospitalization for early preterm infants compared with late preterm infants affords greater opportunities for parental education and engagement about safe sleep practices,” they wrote.
Among early preterm infants, odds percentages increased by 7.3%, 7.7%, and 10.0% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers, respectively. For late preterm infants, respective rates increased by 5.9%, 4.8%, and 5.8% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers.
Despite these improvements, racial disparities were still observed. Non-Hispanic Black mothers reported lower rates of supine sleep positioning for both early preterm infants (odds ratio [OR], 0.61; P less than .0001) and late preterm infants (OR, 0.44; P less than .0001) compared with non-Hispanic White mothers.
These disparities seem “to be in line with racial/ethnic disparity trends in infant mortality and in SUID rates that have persisted for decades among infants,” the investigators wrote.
To a lesser degree, and lacking statistical significance, Hispanic mothers reported lower odds of supine sleep positioning than the odds of White mothers for both early preterm infants (OR, 0.80; P = .1670) and late preterm infants (OR, 0.81; P = .1054).
According to Dr. Hwang and colleagues, more specific demographic data are needed to accurately describe supine sleep positioning rates among Hispanic mothers, partly because of the heterogeneity of this cohort.
“A large body of literature has shown significant variability by immigrant status and country of origin in several infant health outcomes among the Hispanic population,” the investigators wrote. “This study was unable to stratify the Hispanic cohort by these characteristics and thus the distribution of supine sleep positioning prevalence across different Hispanic subgroups could not be demonstrated in this study.”
The investigators also suggested that interventional studies are needed.
“Additional efforts to understand the barriers and facilitators to SSP [supine sleep positioning] adherence among all preterm infant caregivers, particularly non-Hispanic Black and Hispanic parents, are needed so that novel interventions can then be developed,” they wrote.
According to Denice Cora-Bramble, MD, MBA, chief diversity officer at Children’s National Hospital and professor of pediatrics at George Washington University, Washington, the observed improvements in supine sleep positioning may predict lower rates of infant mortality, but more work in the area is needed.
“In spite of improvement in infants’ supine sleep positioning during the study period, racial/ethnic disparities persisted among non-Hispanic Blacks and Hispanics,” Dr. Cora-Bramble said. “That there was improvement among the populations included in the study is significant because of the associated and expected decrease in infant mortality. However, the study results need to be evaluated within the context of [the study’s] limitations, such as the inclusion of only sixteen states in the data analysis. More research is needed to understand and effectively address the disparities highlighted in the study.”
The investigators and Dr. Cora-Bramble reported no conflicts of interest.
FROM JOURNAL OF PEDIATRICS









