User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
FDA approves new enzyme replacement therapy for Pompe disease
Pompe disease is a rare genetic disease that occurs in an estimated 1 in 40,000 births. It is caused by a genetic deficiency or dysfunction of the lysosomal enzyme acid alpha-glucosidase (GAA), which leads to a buildup of glycogen in skeletal and cardiac muscle cells, causing muscle weakness and premature death from respiratory failure or heart failure.
Nexviazyme, administered by intravenous infusion every 2 weeks, supplements GAA and helps reduce glycogen accumulation.
The approval of this product “brings patients with Pompe disease another enzyme replacement therapy option for this rare disease,” said Janet Maynard, MD, deputy director, Office of Rare Diseases, Pediatrics, Urologic and Reproductive Medicine, in the FDA’s Center for Drug Evaluation and Research, in a news release.
In 2010, the FDA approved alglucosidase alfa (Lumizyme) for the treatment of late-onset Pompe disease.
“The FDA will continue to work with stakeholders to advance the development of additional new, effective, and safe therapies for rare diseases, including Pompe disease,” said Dr. Maynard.
The approval is based on positive phase 3 data that demonstrated improvements in key disease burden measures, including respiratory function and walking disease, and that established the drug’s safety profile, Genzyme said in a news release.
The most common side effects were headache, fatigue, diarrhea, nausea, joint pain, dizziness, myalgia, pruritus, vomiting, dyspnea, erythema, paresthesia, and urticaria.
Serious reactions included hypersensitivity reactions, such as anaphylaxis, and infusion-associated reactions, including respiratory distress, chills, and pyrexia.
Patients susceptible to fluid volume overload or those with compromised cardiac or respiratory function may be at risk for serious acute cardiorespiratory failure.
The FDA granted Nexviazyme orphan drug designation, priority review, and breakthrough status.
Genzyme expects the new therapy to be available in the United States in the coming weeks and said it will be priced on par with Lumizyme.
A version of this article first appeared on Medscape.com.
Pompe disease is a rare genetic disease that occurs in an estimated 1 in 40,000 births. It is caused by a genetic deficiency or dysfunction of the lysosomal enzyme acid alpha-glucosidase (GAA), which leads to a buildup of glycogen in skeletal and cardiac muscle cells, causing muscle weakness and premature death from respiratory failure or heart failure.
Nexviazyme, administered by intravenous infusion every 2 weeks, supplements GAA and helps reduce glycogen accumulation.
The approval of this product “brings patients with Pompe disease another enzyme replacement therapy option for this rare disease,” said Janet Maynard, MD, deputy director, Office of Rare Diseases, Pediatrics, Urologic and Reproductive Medicine, in the FDA’s Center for Drug Evaluation and Research, in a news release.
In 2010, the FDA approved alglucosidase alfa (Lumizyme) for the treatment of late-onset Pompe disease.
“The FDA will continue to work with stakeholders to advance the development of additional new, effective, and safe therapies for rare diseases, including Pompe disease,” said Dr. Maynard.
The approval is based on positive phase 3 data that demonstrated improvements in key disease burden measures, including respiratory function and walking disease, and that established the drug’s safety profile, Genzyme said in a news release.
The most common side effects were headache, fatigue, diarrhea, nausea, joint pain, dizziness, myalgia, pruritus, vomiting, dyspnea, erythema, paresthesia, and urticaria.
Serious reactions included hypersensitivity reactions, such as anaphylaxis, and infusion-associated reactions, including respiratory distress, chills, and pyrexia.
Patients susceptible to fluid volume overload or those with compromised cardiac or respiratory function may be at risk for serious acute cardiorespiratory failure.
The FDA granted Nexviazyme orphan drug designation, priority review, and breakthrough status.
Genzyme expects the new therapy to be available in the United States in the coming weeks and said it will be priced on par with Lumizyme.
A version of this article first appeared on Medscape.com.
Pompe disease is a rare genetic disease that occurs in an estimated 1 in 40,000 births. It is caused by a genetic deficiency or dysfunction of the lysosomal enzyme acid alpha-glucosidase (GAA), which leads to a buildup of glycogen in skeletal and cardiac muscle cells, causing muscle weakness and premature death from respiratory failure or heart failure.
Nexviazyme, administered by intravenous infusion every 2 weeks, supplements GAA and helps reduce glycogen accumulation.
The approval of this product “brings patients with Pompe disease another enzyme replacement therapy option for this rare disease,” said Janet Maynard, MD, deputy director, Office of Rare Diseases, Pediatrics, Urologic and Reproductive Medicine, in the FDA’s Center for Drug Evaluation and Research, in a news release.
In 2010, the FDA approved alglucosidase alfa (Lumizyme) for the treatment of late-onset Pompe disease.
“The FDA will continue to work with stakeholders to advance the development of additional new, effective, and safe therapies for rare diseases, including Pompe disease,” said Dr. Maynard.
The approval is based on positive phase 3 data that demonstrated improvements in key disease burden measures, including respiratory function and walking disease, and that established the drug’s safety profile, Genzyme said in a news release.
The most common side effects were headache, fatigue, diarrhea, nausea, joint pain, dizziness, myalgia, pruritus, vomiting, dyspnea, erythema, paresthesia, and urticaria.
Serious reactions included hypersensitivity reactions, such as anaphylaxis, and infusion-associated reactions, including respiratory distress, chills, and pyrexia.
Patients susceptible to fluid volume overload or those with compromised cardiac or respiratory function may be at risk for serious acute cardiorespiratory failure.
The FDA granted Nexviazyme orphan drug designation, priority review, and breakthrough status.
Genzyme expects the new therapy to be available in the United States in the coming weeks and said it will be priced on par with Lumizyme.
A version of this article first appeared on Medscape.com.
CAG Clinical Practice Guideline: Vaccination in patients with IBD
The Canadian Association of Gastroenterology (CAG) has published a two-part clinical practice guideline for immunizing patients with inflammatory bowel disease (IBD) that covers both live and inactivated vaccines across pediatric and adult patients.
The guideline, which has been endorsed by the American Gastroenterological Association, is composed of recommendations drawn from a broader body of data than prior publications on the same topic, according to Eric I. Benchimol, MD, PhD, of the University of Ottawa and the University of Toronto, and colleagues.
“Previous guidelines on immunizations of patients with IBD considered only the limited available evidence of vaccine safety and effectiveness in IBD populations, and failed to consider the ample evidence available in the general population or in other immune-mediated inflammatory diseases when assessing the certainty of evidence or developing their recommendations,” they wrote in Gastroenterology.
Part 1: Live vaccine recommendations
The first part of the guideline includes seven recommendations for use of live vaccines in patients with IBD.
In this area, decision-making is largely dependent upon use of immunosuppressive therapy, which the investigators defined as “corticosteroids, thiopurines, biologics, small molecules such as JAK [Janus kinase] inhibitors, and combinations thereof,” with the caveat that “there is no standard definition of immunosuppression,” and “the degree to which immunosuppressive therapy causes clinically significant immunosuppression generally is dose related and varies by drug.”
Before offering specific recommendations, Dr. Benchimol and colleagues provided three general principles to abide by: 1. Clinicians should review each patient’s history of immunization and vaccine-preventable diseases at diagnosis and on a routine basis; 2. Appropriate vaccinations should ideally be given prior to starting immunosuppressive therapy; and 3. Immunosuppressive therapy (when urgently needed) should not be delayed so that immunizations can be given in advance.
“[Delaying therapy] could lead to more anticipated harms than benefits, due to the risk of progression of the inflammatory activity and resulting complications,” the investigators wrote.
Specific recommendations in the guideline address measles, mumps, and rubella (MMR); and varicella. Both vaccines are recommended for susceptible pediatric and adult patients not taking immunosuppressive therapy. In contrast, neither vaccine is recommended for immunosuppressed patients of any age. Certainty of evidence ranged from very low to moderate.
Concerning vaccination within the first 6 months of life for infants born of mothers taking biologics, the expert panel did not reach a consensus.
“[T]he group was unable to recommend for or against their routine use because the desirable and undesirable effects were closely balanced and the evidence on safety outcomes was insufficient to justify a recommendation,” wrote Dr. Benchimol and colleagues. “Health care providers should be cautious with the administration of live vaccines in the first year of life in the infants of mothers using biologics. These infants should be evaluated by clinicians with expertise in the impact of exposure to monoclonal antibody biologics in utero.”
Part 2: Inactivated vaccine recommendations
The second part of the guideline, by lead author Jennifer L. Jones, MD, of Dalhousie University, Queen Elizabeth II Health Sciences Center, Halifax, N.S., and colleagues, provides 15 recommendations for giving inactivated vaccines to patients with IBD.
The panel considered eight vaccines: Haemophilus influenzae type B (Hib); herpes zoster (HZ); hepatitis B; influenza; Streptococcus pneumoniae (pneumococcal vaccine); Neisseria meningitidis (meningococcal vaccine); human papillomavirus (HPV); and diphtheria, tetanus, and pertussis.
Generally, the above vaccines are recommended on an age-appropriate basis, regardless of immunosuppression status, albeit with varying levels of confidence. For example, the Hib vaccine is strongly recommended for pediatric patients 5 years and younger, whereas the same recommendation for older children and adults is conditional.
For several patient populations and vaccines, the guideline panel did not reach a consensus, including use of double-dose hepatitis B vaccine for immunosuppressed adults, timing seasonal flu shots with dosing of biologics, use of pneumococcal vaccines in nonimmunosuppressed patents without a risk factor for pneumococcal disease, use of meningococcal vaccines in adults not at risk for invasive meningococcal disease, and use of HPV vaccine in patients aged 27-45 years.
While immunosuppressive therapy is not a contraindication for giving inactivated vaccines, Dr. Jones and colleagues noted that immunosuppression may hinder vaccine responses.
“Given that patients with IBD on immunosuppressive therapy may have lower immune response to vaccine, further research will be needed to assess the safety and effectiveness of high-dose vs. standard-dose vaccination strategy,” they wrote, also noting that more work is needed to determine if accelerated vaccinations strategies may be feasible prior to initiation of immunosuppressive therapy.
Because of a lack of evidence, the guideline panel did not issue IBD-specific recommendations for vaccines against SARS-CoV-2; however, Dr. Jones and colleagues suggested that clinicians reference a CAG publication on the subject published earlier this year.
The guideline was supported by grants to the Canadian Association of Gastroenterology from the Canadian Institutes of Health Research’s Institute of Nutrition, Metabolism and Diabetes; and CANImmunize. Dr. Benchimol disclosed additional relationships with the Canadian Institutes of Health Research, Crohn’s and Colitis Canada; and the Canadian Child Health Clinician Scientist Program.
The Canadian Association of Gastroenterology (CAG) has published a two-part clinical practice guideline for immunizing patients with inflammatory bowel disease (IBD) that covers both live and inactivated vaccines across pediatric and adult patients.
The guideline, which has been endorsed by the American Gastroenterological Association, is composed of recommendations drawn from a broader body of data than prior publications on the same topic, according to Eric I. Benchimol, MD, PhD, of the University of Ottawa and the University of Toronto, and colleagues.
“Previous guidelines on immunizations of patients with IBD considered only the limited available evidence of vaccine safety and effectiveness in IBD populations, and failed to consider the ample evidence available in the general population or in other immune-mediated inflammatory diseases when assessing the certainty of evidence or developing their recommendations,” they wrote in Gastroenterology.
Part 1: Live vaccine recommendations
The first part of the guideline includes seven recommendations for use of live vaccines in patients with IBD.
In this area, decision-making is largely dependent upon use of immunosuppressive therapy, which the investigators defined as “corticosteroids, thiopurines, biologics, small molecules such as JAK [Janus kinase] inhibitors, and combinations thereof,” with the caveat that “there is no standard definition of immunosuppression,” and “the degree to which immunosuppressive therapy causes clinically significant immunosuppression generally is dose related and varies by drug.”
Before offering specific recommendations, Dr. Benchimol and colleagues provided three general principles to abide by: 1. Clinicians should review each patient’s history of immunization and vaccine-preventable diseases at diagnosis and on a routine basis; 2. Appropriate vaccinations should ideally be given prior to starting immunosuppressive therapy; and 3. Immunosuppressive therapy (when urgently needed) should not be delayed so that immunizations can be given in advance.
“[Delaying therapy] could lead to more anticipated harms than benefits, due to the risk of progression of the inflammatory activity and resulting complications,” the investigators wrote.
Specific recommendations in the guideline address measles, mumps, and rubella (MMR); and varicella. Both vaccines are recommended for susceptible pediatric and adult patients not taking immunosuppressive therapy. In contrast, neither vaccine is recommended for immunosuppressed patients of any age. Certainty of evidence ranged from very low to moderate.
Concerning vaccination within the first 6 months of life for infants born of mothers taking biologics, the expert panel did not reach a consensus.
“[T]he group was unable to recommend for or against their routine use because the desirable and undesirable effects were closely balanced and the evidence on safety outcomes was insufficient to justify a recommendation,” wrote Dr. Benchimol and colleagues. “Health care providers should be cautious with the administration of live vaccines in the first year of life in the infants of mothers using biologics. These infants should be evaluated by clinicians with expertise in the impact of exposure to monoclonal antibody biologics in utero.”
Part 2: Inactivated vaccine recommendations
The second part of the guideline, by lead author Jennifer L. Jones, MD, of Dalhousie University, Queen Elizabeth II Health Sciences Center, Halifax, N.S., and colleagues, provides 15 recommendations for giving inactivated vaccines to patients with IBD.
The panel considered eight vaccines: Haemophilus influenzae type B (Hib); herpes zoster (HZ); hepatitis B; influenza; Streptococcus pneumoniae (pneumococcal vaccine); Neisseria meningitidis (meningococcal vaccine); human papillomavirus (HPV); and diphtheria, tetanus, and pertussis.
Generally, the above vaccines are recommended on an age-appropriate basis, regardless of immunosuppression status, albeit with varying levels of confidence. For example, the Hib vaccine is strongly recommended for pediatric patients 5 years and younger, whereas the same recommendation for older children and adults is conditional.
For several patient populations and vaccines, the guideline panel did not reach a consensus, including use of double-dose hepatitis B vaccine for immunosuppressed adults, timing seasonal flu shots with dosing of biologics, use of pneumococcal vaccines in nonimmunosuppressed patents without a risk factor for pneumococcal disease, use of meningococcal vaccines in adults not at risk for invasive meningococcal disease, and use of HPV vaccine in patients aged 27-45 years.
While immunosuppressive therapy is not a contraindication for giving inactivated vaccines, Dr. Jones and colleagues noted that immunosuppression may hinder vaccine responses.
“Given that patients with IBD on immunosuppressive therapy may have lower immune response to vaccine, further research will be needed to assess the safety and effectiveness of high-dose vs. standard-dose vaccination strategy,” they wrote, also noting that more work is needed to determine if accelerated vaccinations strategies may be feasible prior to initiation of immunosuppressive therapy.
Because of a lack of evidence, the guideline panel did not issue IBD-specific recommendations for vaccines against SARS-CoV-2; however, Dr. Jones and colleagues suggested that clinicians reference a CAG publication on the subject published earlier this year.
The guideline was supported by grants to the Canadian Association of Gastroenterology from the Canadian Institutes of Health Research’s Institute of Nutrition, Metabolism and Diabetes; and CANImmunize. Dr. Benchimol disclosed additional relationships with the Canadian Institutes of Health Research, Crohn’s and Colitis Canada; and the Canadian Child Health Clinician Scientist Program.
The Canadian Association of Gastroenterology (CAG) has published a two-part clinical practice guideline for immunizing patients with inflammatory bowel disease (IBD) that covers both live and inactivated vaccines across pediatric and adult patients.
The guideline, which has been endorsed by the American Gastroenterological Association, is composed of recommendations drawn from a broader body of data than prior publications on the same topic, according to Eric I. Benchimol, MD, PhD, of the University of Ottawa and the University of Toronto, and colleagues.
“Previous guidelines on immunizations of patients with IBD considered only the limited available evidence of vaccine safety and effectiveness in IBD populations, and failed to consider the ample evidence available in the general population or in other immune-mediated inflammatory diseases when assessing the certainty of evidence or developing their recommendations,” they wrote in Gastroenterology.
Part 1: Live vaccine recommendations
The first part of the guideline includes seven recommendations for use of live vaccines in patients with IBD.
In this area, decision-making is largely dependent upon use of immunosuppressive therapy, which the investigators defined as “corticosteroids, thiopurines, biologics, small molecules such as JAK [Janus kinase] inhibitors, and combinations thereof,” with the caveat that “there is no standard definition of immunosuppression,” and “the degree to which immunosuppressive therapy causes clinically significant immunosuppression generally is dose related and varies by drug.”
Before offering specific recommendations, Dr. Benchimol and colleagues provided three general principles to abide by: 1. Clinicians should review each patient’s history of immunization and vaccine-preventable diseases at diagnosis and on a routine basis; 2. Appropriate vaccinations should ideally be given prior to starting immunosuppressive therapy; and 3. Immunosuppressive therapy (when urgently needed) should not be delayed so that immunizations can be given in advance.
“[Delaying therapy] could lead to more anticipated harms than benefits, due to the risk of progression of the inflammatory activity and resulting complications,” the investigators wrote.
Specific recommendations in the guideline address measles, mumps, and rubella (MMR); and varicella. Both vaccines are recommended for susceptible pediatric and adult patients not taking immunosuppressive therapy. In contrast, neither vaccine is recommended for immunosuppressed patients of any age. Certainty of evidence ranged from very low to moderate.
Concerning vaccination within the first 6 months of life for infants born of mothers taking biologics, the expert panel did not reach a consensus.
“[T]he group was unable to recommend for or against their routine use because the desirable and undesirable effects were closely balanced and the evidence on safety outcomes was insufficient to justify a recommendation,” wrote Dr. Benchimol and colleagues. “Health care providers should be cautious with the administration of live vaccines in the first year of life in the infants of mothers using biologics. These infants should be evaluated by clinicians with expertise in the impact of exposure to monoclonal antibody biologics in utero.”
Part 2: Inactivated vaccine recommendations
The second part of the guideline, by lead author Jennifer L. Jones, MD, of Dalhousie University, Queen Elizabeth II Health Sciences Center, Halifax, N.S., and colleagues, provides 15 recommendations for giving inactivated vaccines to patients with IBD.
The panel considered eight vaccines: Haemophilus influenzae type B (Hib); herpes zoster (HZ); hepatitis B; influenza; Streptococcus pneumoniae (pneumococcal vaccine); Neisseria meningitidis (meningococcal vaccine); human papillomavirus (HPV); and diphtheria, tetanus, and pertussis.
Generally, the above vaccines are recommended on an age-appropriate basis, regardless of immunosuppression status, albeit with varying levels of confidence. For example, the Hib vaccine is strongly recommended for pediatric patients 5 years and younger, whereas the same recommendation for older children and adults is conditional.
For several patient populations and vaccines, the guideline panel did not reach a consensus, including use of double-dose hepatitis B vaccine for immunosuppressed adults, timing seasonal flu shots with dosing of biologics, use of pneumococcal vaccines in nonimmunosuppressed patents without a risk factor for pneumococcal disease, use of meningococcal vaccines in adults not at risk for invasive meningococcal disease, and use of HPV vaccine in patients aged 27-45 years.
While immunosuppressive therapy is not a contraindication for giving inactivated vaccines, Dr. Jones and colleagues noted that immunosuppression may hinder vaccine responses.
“Given that patients with IBD on immunosuppressive therapy may have lower immune response to vaccine, further research will be needed to assess the safety and effectiveness of high-dose vs. standard-dose vaccination strategy,” they wrote, also noting that more work is needed to determine if accelerated vaccinations strategies may be feasible prior to initiation of immunosuppressive therapy.
Because of a lack of evidence, the guideline panel did not issue IBD-specific recommendations for vaccines against SARS-CoV-2; however, Dr. Jones and colleagues suggested that clinicians reference a CAG publication on the subject published earlier this year.
The guideline was supported by grants to the Canadian Association of Gastroenterology from the Canadian Institutes of Health Research’s Institute of Nutrition, Metabolism and Diabetes; and CANImmunize. Dr. Benchimol disclosed additional relationships with the Canadian Institutes of Health Research, Crohn’s and Colitis Canada; and the Canadian Child Health Clinician Scientist Program.
FROM GASTROENTEROLOGY
Ultraprocessed foods comprise most of the calories for youths
In the 2 decades from 1999 to 2018, ultraprocessed foods consistently accounted for the majority of energy intake by American young people, a large cross-sectional study of National Health and Nutrition Examination Survey (NHANES) data shows.
In young people aged 2-19 years, the estimated percentage of total energy from consumption of ultraprocessed foods increased from 61.4% to 67.0%, for a difference of 5.6% (95% confidence interval [CI] 3.5-7.7, P < .001 for trend), according to Lu Wang, PhD, MPH, a postdoctoral fellow at the Friedman School of Nutrition Science and Policy at Tufts University in Boston, and colleagues.
In contrast, total energy from non- or minimally processed foods decreased from 28.8% to 23.5% (difference −5.3%, 95% CI, −7.5 to −3.2, P < .001 for trend).
“The estimated percentage of energy consumed from ultraprocessed foods increased from 1999 to 2018, with an increasing trend in ready-to-heat and -eat mixed dishes and a decreasing trend in sugar-sweetened beverages,” the authors wrote. The report was published online Aug. 10 in JAMA.
The findings held regardless of the educational and socioeconomic status of the children’s parents.
Significant disparities by race and ethnicity emerged, however, with the ultraprocessed food phenomenon more marked in non-Hispanic Black youths and Mexican-American youths than in their non-Hispanic White counterparts. “Targeted marketing of junk foods toward racial/ethnic minority youths may partly contribute to such differences,” the authors wrote. “However, persistently lower consumption of ultraprocessed foods among Mexican-American youths may reflect more home cooking among Hispanic families.”
Among non-Hispanic Black youths consumption rose from 62.2% to 72.5% (difference 10.3%, 95% CI, 6.8-13.8) and among Mexican-American youths from 55.8% to 63.5% (difference 7.6%, 95% CI, 4.4-10.9). In non-Hispanic White youths intake rose from 63.4% to 68.6% (difference 5.2%, 95% CI, 2.1-8.3, P = .04 for trends).
In addition, a higher consumption of ultraprocessed foods among school-aged youths than among preschool children aged 2-5 years may reflect increased marketing, availability, and selection of ultraprocessed foods for older youths, the authors noted.
Food processing, with its potential adverse effects, may need to be considered as a food dimension in addition to nutrients and food groups in future dietary recommendations and food policies, they added.
“An increasing number of studies are showing a link between ultraprocessed food consumption and adverse health outcomes in children,” corresponding author Fang Fang Zhang, MD, PhD, Neely Family Professor and associate professor at Tufts’ Friedman School of Nutrition Science and Policy, said in an interview. “Health care providers can play a larger role in encouraging patients – and their parents – to replace unhealthy ultraprocessed foods such as ultraprocessed sweet bakery products with healthy unprocessed or minimally processed foods in their diet such as less processed whole grains. “
In Dr. Zhang’s view, teachers also have a part to play in promoting nutrition literacy. “Schools can play an important role in empowering children with knowledge and skills to make healthy food choices,” she said. “Nutrition literacy should be an integral part of the health education curriculum in all K-12 schools.”
Commenting on the study but not involved in it, Michelle Katzow, MD, a pediatrician/obesity medicine specialist and assistant professor at the Feinstein Institutes for Medical Research in Manhasset, N.Y., said the work highlights an often overlooked aspect of the modern American diet that may well be contributing to poor health outcomes in young people.
“It suggests that even as the science advances and we learn more about the adverse health effects of ultraprocessed foods, public health efforts to improve nutrition and food quality in children have not been successful,” she said in an interview. “This is because it is so hard for public health advocates to compete with the food industry, which stands to really benefit financially from hooking kids on processed foods that are not good for their health.”
Dr. Katzow added that the observed racial/ethnic disparities are not surprising in light of a growing body of evidence that racism exists in food marketing. “We need to put forward policies that regulate the food industry, particularly in relation to its most susceptible targets, our kids.”
Study details
The serial cross-sectional analysis used 24-hour dietary recall data from a nationally representative sample from 10 NHANES cycles for the range of 1999-2000 to 2017-2018. The weighted mean age of the cohort was 10.7 years and 49.1% were girls.
Among the subgroups of ultraprocessed foods, the estimated percentage of energy from ready-to-heat and ready-to-eat mixed dishes increased from 2.2% to 11.2% (difference 8.9%; 95%, CI, 7.7-10.2).
Energy from sweets and sweet snacks increased from 10.7% to 12.9% (difference 2.3%; 95% CI, 1.0-3.6), but the estimated percentage of energy decreased for sugar-sweetened beverages from 10.8% to 5.3% (difference −5.5%; 95% CI, −6.5 to −4.5).
In other categories, estimated energy intake from processed fats and oils, condiments, and sauces fell from 7.1% to 4.0% (difference −3.1%; 95% CI, −3.7 to −2.6, all P < .05 for trend).
Not surprisingly, ultraprocessed foods had an overall poorer nutrient profile than that of nonultraprocessed, although they often contained less saturated fat, and they also contained more carbohydrates, mostly from low-quality sources with added sugars and low levels of dietary fiber and protein.
And despite a higher total folate content in ultraprocessed foods because of fortification, higher-level consumers took in less total folate owing to their lower consumption of whole foods.
The authors cautioned that in addition to poor nutrient profiles, processing itself may harm health by changing the physical structure and chemical composition of food, which could lead to elevated glycemic response and reduced satiety. Furthermore, recent research has linked food additives such as emulsifiers, stabilizers, and artificial sweeteners to adverse metabolomic effects and obesity risk. Pointing to the recent success of efforts to reduce consumption of sugary beverages, Dr. Zhang said, “We need to mobilize the same energy and level of commitment when it comes to other unhealthy ultraprocessed foods such as cakes, cookies, doughnuts, and brownies.”
The trends identified by the Tufts study “are concerning and potentially have major public health significance,” according to an accompanying JAMA editorial.
“Better dietary assessment methods are needed to document trends and understand the unique role of ultraprocessed foods to inform future evidence-based policy and dietary recommendations,” wrote Katie A. Meyer, ScD, and Lindsey Smith Taillie, PhD, of the Gillings School of Global Public Health at the University of North Carolina in Chapel Hill.
The editorialists share the authors’ view that “a conceptual advancement would be to consider the level and characteristics of processing as just one of multiple dimensions (including nutrients and food groups) used to classify foods as healthy or unhealthy.” They pointed out that the Pan American Health Organization already recommends targeting products that are ultraprocessed and high in concerning add-in nutrients.
They cautioned, however, that the classification of ultraprocessed foods will not be easy because it requires data on a full list of ingredients, and the effects of processing generally cannot be separated from the composite nutrients of ultraprocessed foods.
This presents a challenge for national food consumption research “given that most large epidemiological studies rely on food frequency questionnaires that lack the information necessary to classify processing levels,” they wrote.
This study was supported by the National Institutes of Health and the São Paulo Research Foundation. Coauthor Dariush Mozaffarian, MD, a cardiologist at Tufts University, disclosed support from the Bill & Melinda Gates Foundation, the National Institutes of Health, and the Rockefeller Foundation as well as personal fees from several commercial companies. He has served on several scientific advisory boards and received royalties from UpToDate, all outside of the submitted work. Dr. Meyer reported a grant from choline manufacturer Balchem. Dr. Taillie reported funding from Bloomberg Philanthropies. Dr. Zhang had no disclosures. Dr. Katzow disclosed no competing interests.
In the 2 decades from 1999 to 2018, ultraprocessed foods consistently accounted for the majority of energy intake by American young people, a large cross-sectional study of National Health and Nutrition Examination Survey (NHANES) data shows.
In young people aged 2-19 years, the estimated percentage of total energy from consumption of ultraprocessed foods increased from 61.4% to 67.0%, for a difference of 5.6% (95% confidence interval [CI] 3.5-7.7, P < .001 for trend), according to Lu Wang, PhD, MPH, a postdoctoral fellow at the Friedman School of Nutrition Science and Policy at Tufts University in Boston, and colleagues.
In contrast, total energy from non- or minimally processed foods decreased from 28.8% to 23.5% (difference −5.3%, 95% CI, −7.5 to −3.2, P < .001 for trend).
“The estimated percentage of energy consumed from ultraprocessed foods increased from 1999 to 2018, with an increasing trend in ready-to-heat and -eat mixed dishes and a decreasing trend in sugar-sweetened beverages,” the authors wrote. The report was published online Aug. 10 in JAMA.
The findings held regardless of the educational and socioeconomic status of the children’s parents.
Significant disparities by race and ethnicity emerged, however, with the ultraprocessed food phenomenon more marked in non-Hispanic Black youths and Mexican-American youths than in their non-Hispanic White counterparts. “Targeted marketing of junk foods toward racial/ethnic minority youths may partly contribute to such differences,” the authors wrote. “However, persistently lower consumption of ultraprocessed foods among Mexican-American youths may reflect more home cooking among Hispanic families.”
Among non-Hispanic Black youths consumption rose from 62.2% to 72.5% (difference 10.3%, 95% CI, 6.8-13.8) and among Mexican-American youths from 55.8% to 63.5% (difference 7.6%, 95% CI, 4.4-10.9). In non-Hispanic White youths intake rose from 63.4% to 68.6% (difference 5.2%, 95% CI, 2.1-8.3, P = .04 for trends).
In addition, a higher consumption of ultraprocessed foods among school-aged youths than among preschool children aged 2-5 years may reflect increased marketing, availability, and selection of ultraprocessed foods for older youths, the authors noted.
Food processing, with its potential adverse effects, may need to be considered as a food dimension in addition to nutrients and food groups in future dietary recommendations and food policies, they added.
“An increasing number of studies are showing a link between ultraprocessed food consumption and adverse health outcomes in children,” corresponding author Fang Fang Zhang, MD, PhD, Neely Family Professor and associate professor at Tufts’ Friedman School of Nutrition Science and Policy, said in an interview. “Health care providers can play a larger role in encouraging patients – and their parents – to replace unhealthy ultraprocessed foods such as ultraprocessed sweet bakery products with healthy unprocessed or minimally processed foods in their diet such as less processed whole grains. “
In Dr. Zhang’s view, teachers also have a part to play in promoting nutrition literacy. “Schools can play an important role in empowering children with knowledge and skills to make healthy food choices,” she said. “Nutrition literacy should be an integral part of the health education curriculum in all K-12 schools.”
Commenting on the study but not involved in it, Michelle Katzow, MD, a pediatrician/obesity medicine specialist and assistant professor at the Feinstein Institutes for Medical Research in Manhasset, N.Y., said the work highlights an often overlooked aspect of the modern American diet that may well be contributing to poor health outcomes in young people.
“It suggests that even as the science advances and we learn more about the adverse health effects of ultraprocessed foods, public health efforts to improve nutrition and food quality in children have not been successful,” she said in an interview. “This is because it is so hard for public health advocates to compete with the food industry, which stands to really benefit financially from hooking kids on processed foods that are not good for their health.”
Dr. Katzow added that the observed racial/ethnic disparities are not surprising in light of a growing body of evidence that racism exists in food marketing. “We need to put forward policies that regulate the food industry, particularly in relation to its most susceptible targets, our kids.”
Study details
The serial cross-sectional analysis used 24-hour dietary recall data from a nationally representative sample from 10 NHANES cycles for the range of 1999-2000 to 2017-2018. The weighted mean age of the cohort was 10.7 years and 49.1% were girls.
Among the subgroups of ultraprocessed foods, the estimated percentage of energy from ready-to-heat and ready-to-eat mixed dishes increased from 2.2% to 11.2% (difference 8.9%; 95%, CI, 7.7-10.2).
Energy from sweets and sweet snacks increased from 10.7% to 12.9% (difference 2.3%; 95% CI, 1.0-3.6), but the estimated percentage of energy decreased for sugar-sweetened beverages from 10.8% to 5.3% (difference −5.5%; 95% CI, −6.5 to −4.5).
In other categories, estimated energy intake from processed fats and oils, condiments, and sauces fell from 7.1% to 4.0% (difference −3.1%; 95% CI, −3.7 to −2.6, all P < .05 for trend).
Not surprisingly, ultraprocessed foods had an overall poorer nutrient profile than that of nonultraprocessed, although they often contained less saturated fat, and they also contained more carbohydrates, mostly from low-quality sources with added sugars and low levels of dietary fiber and protein.
And despite a higher total folate content in ultraprocessed foods because of fortification, higher-level consumers took in less total folate owing to their lower consumption of whole foods.
The authors cautioned that in addition to poor nutrient profiles, processing itself may harm health by changing the physical structure and chemical composition of food, which could lead to elevated glycemic response and reduced satiety. Furthermore, recent research has linked food additives such as emulsifiers, stabilizers, and artificial sweeteners to adverse metabolomic effects and obesity risk. Pointing to the recent success of efforts to reduce consumption of sugary beverages, Dr. Zhang said, “We need to mobilize the same energy and level of commitment when it comes to other unhealthy ultraprocessed foods such as cakes, cookies, doughnuts, and brownies.”
The trends identified by the Tufts study “are concerning and potentially have major public health significance,” according to an accompanying JAMA editorial.
“Better dietary assessment methods are needed to document trends and understand the unique role of ultraprocessed foods to inform future evidence-based policy and dietary recommendations,” wrote Katie A. Meyer, ScD, and Lindsey Smith Taillie, PhD, of the Gillings School of Global Public Health at the University of North Carolina in Chapel Hill.
The editorialists share the authors’ view that “a conceptual advancement would be to consider the level and characteristics of processing as just one of multiple dimensions (including nutrients and food groups) used to classify foods as healthy or unhealthy.” They pointed out that the Pan American Health Organization already recommends targeting products that are ultraprocessed and high in concerning add-in nutrients.
They cautioned, however, that the classification of ultraprocessed foods will not be easy because it requires data on a full list of ingredients, and the effects of processing generally cannot be separated from the composite nutrients of ultraprocessed foods.
This presents a challenge for national food consumption research “given that most large epidemiological studies rely on food frequency questionnaires that lack the information necessary to classify processing levels,” they wrote.
This study was supported by the National Institutes of Health and the São Paulo Research Foundation. Coauthor Dariush Mozaffarian, MD, a cardiologist at Tufts University, disclosed support from the Bill & Melinda Gates Foundation, the National Institutes of Health, and the Rockefeller Foundation as well as personal fees from several commercial companies. He has served on several scientific advisory boards and received royalties from UpToDate, all outside of the submitted work. Dr. Meyer reported a grant from choline manufacturer Balchem. Dr. Taillie reported funding from Bloomberg Philanthropies. Dr. Zhang had no disclosures. Dr. Katzow disclosed no competing interests.
In the 2 decades from 1999 to 2018, ultraprocessed foods consistently accounted for the majority of energy intake by American young people, a large cross-sectional study of National Health and Nutrition Examination Survey (NHANES) data shows.
In young people aged 2-19 years, the estimated percentage of total energy from consumption of ultraprocessed foods increased from 61.4% to 67.0%, for a difference of 5.6% (95% confidence interval [CI] 3.5-7.7, P < .001 for trend), according to Lu Wang, PhD, MPH, a postdoctoral fellow at the Friedman School of Nutrition Science and Policy at Tufts University in Boston, and colleagues.
In contrast, total energy from non- or minimally processed foods decreased from 28.8% to 23.5% (difference −5.3%, 95% CI, −7.5 to −3.2, P < .001 for trend).
“The estimated percentage of energy consumed from ultraprocessed foods increased from 1999 to 2018, with an increasing trend in ready-to-heat and -eat mixed dishes and a decreasing trend in sugar-sweetened beverages,” the authors wrote. The report was published online Aug. 10 in JAMA.
The findings held regardless of the educational and socioeconomic status of the children’s parents.
Significant disparities by race and ethnicity emerged, however, with the ultraprocessed food phenomenon more marked in non-Hispanic Black youths and Mexican-American youths than in their non-Hispanic White counterparts. “Targeted marketing of junk foods toward racial/ethnic minority youths may partly contribute to such differences,” the authors wrote. “However, persistently lower consumption of ultraprocessed foods among Mexican-American youths may reflect more home cooking among Hispanic families.”
Among non-Hispanic Black youths consumption rose from 62.2% to 72.5% (difference 10.3%, 95% CI, 6.8-13.8) and among Mexican-American youths from 55.8% to 63.5% (difference 7.6%, 95% CI, 4.4-10.9). In non-Hispanic White youths intake rose from 63.4% to 68.6% (difference 5.2%, 95% CI, 2.1-8.3, P = .04 for trends).
In addition, a higher consumption of ultraprocessed foods among school-aged youths than among preschool children aged 2-5 years may reflect increased marketing, availability, and selection of ultraprocessed foods for older youths, the authors noted.
Food processing, with its potential adverse effects, may need to be considered as a food dimension in addition to nutrients and food groups in future dietary recommendations and food policies, they added.
“An increasing number of studies are showing a link between ultraprocessed food consumption and adverse health outcomes in children,” corresponding author Fang Fang Zhang, MD, PhD, Neely Family Professor and associate professor at Tufts’ Friedman School of Nutrition Science and Policy, said in an interview. “Health care providers can play a larger role in encouraging patients – and their parents – to replace unhealthy ultraprocessed foods such as ultraprocessed sweet bakery products with healthy unprocessed or minimally processed foods in their diet such as less processed whole grains. “
In Dr. Zhang’s view, teachers also have a part to play in promoting nutrition literacy. “Schools can play an important role in empowering children with knowledge and skills to make healthy food choices,” she said. “Nutrition literacy should be an integral part of the health education curriculum in all K-12 schools.”
Commenting on the study but not involved in it, Michelle Katzow, MD, a pediatrician/obesity medicine specialist and assistant professor at the Feinstein Institutes for Medical Research in Manhasset, N.Y., said the work highlights an often overlooked aspect of the modern American diet that may well be contributing to poor health outcomes in young people.
“It suggests that even as the science advances and we learn more about the adverse health effects of ultraprocessed foods, public health efforts to improve nutrition and food quality in children have not been successful,” she said in an interview. “This is because it is so hard for public health advocates to compete with the food industry, which stands to really benefit financially from hooking kids on processed foods that are not good for their health.”
Dr. Katzow added that the observed racial/ethnic disparities are not surprising in light of a growing body of evidence that racism exists in food marketing. “We need to put forward policies that regulate the food industry, particularly in relation to its most susceptible targets, our kids.”
Study details
The serial cross-sectional analysis used 24-hour dietary recall data from a nationally representative sample from 10 NHANES cycles for the range of 1999-2000 to 2017-2018. The weighted mean age of the cohort was 10.7 years and 49.1% were girls.
Among the subgroups of ultraprocessed foods, the estimated percentage of energy from ready-to-heat and ready-to-eat mixed dishes increased from 2.2% to 11.2% (difference 8.9%; 95%, CI, 7.7-10.2).
Energy from sweets and sweet snacks increased from 10.7% to 12.9% (difference 2.3%; 95% CI, 1.0-3.6), but the estimated percentage of energy decreased for sugar-sweetened beverages from 10.8% to 5.3% (difference −5.5%; 95% CI, −6.5 to −4.5).
In other categories, estimated energy intake from processed fats and oils, condiments, and sauces fell from 7.1% to 4.0% (difference −3.1%; 95% CI, −3.7 to −2.6, all P < .05 for trend).
Not surprisingly, ultraprocessed foods had an overall poorer nutrient profile than that of nonultraprocessed, although they often contained less saturated fat, and they also contained more carbohydrates, mostly from low-quality sources with added sugars and low levels of dietary fiber and protein.
And despite a higher total folate content in ultraprocessed foods because of fortification, higher-level consumers took in less total folate owing to their lower consumption of whole foods.
The authors cautioned that in addition to poor nutrient profiles, processing itself may harm health by changing the physical structure and chemical composition of food, which could lead to elevated glycemic response and reduced satiety. Furthermore, recent research has linked food additives such as emulsifiers, stabilizers, and artificial sweeteners to adverse metabolomic effects and obesity risk. Pointing to the recent success of efforts to reduce consumption of sugary beverages, Dr. Zhang said, “We need to mobilize the same energy and level of commitment when it comes to other unhealthy ultraprocessed foods such as cakes, cookies, doughnuts, and brownies.”
The trends identified by the Tufts study “are concerning and potentially have major public health significance,” according to an accompanying JAMA editorial.
“Better dietary assessment methods are needed to document trends and understand the unique role of ultraprocessed foods to inform future evidence-based policy and dietary recommendations,” wrote Katie A. Meyer, ScD, and Lindsey Smith Taillie, PhD, of the Gillings School of Global Public Health at the University of North Carolina in Chapel Hill.
The editorialists share the authors’ view that “a conceptual advancement would be to consider the level and characteristics of processing as just one of multiple dimensions (including nutrients and food groups) used to classify foods as healthy or unhealthy.” They pointed out that the Pan American Health Organization already recommends targeting products that are ultraprocessed and high in concerning add-in nutrients.
They cautioned, however, that the classification of ultraprocessed foods will not be easy because it requires data on a full list of ingredients, and the effects of processing generally cannot be separated from the composite nutrients of ultraprocessed foods.
This presents a challenge for national food consumption research “given that most large epidemiological studies rely on food frequency questionnaires that lack the information necessary to classify processing levels,” they wrote.
This study was supported by the National Institutes of Health and the São Paulo Research Foundation. Coauthor Dariush Mozaffarian, MD, a cardiologist at Tufts University, disclosed support from the Bill & Melinda Gates Foundation, the National Institutes of Health, and the Rockefeller Foundation as well as personal fees from several commercial companies. He has served on several scientific advisory boards and received royalties from UpToDate, all outside of the submitted work. Dr. Meyer reported a grant from choline manufacturer Balchem. Dr. Taillie reported funding from Bloomberg Philanthropies. Dr. Zhang had no disclosures. Dr. Katzow disclosed no competing interests.
FROM JAMA
Surge of new child COVID cases continues for 6th consecutive week
The current COVID-19 surge has brought new cases in children to their highest level since February, according to a new report.
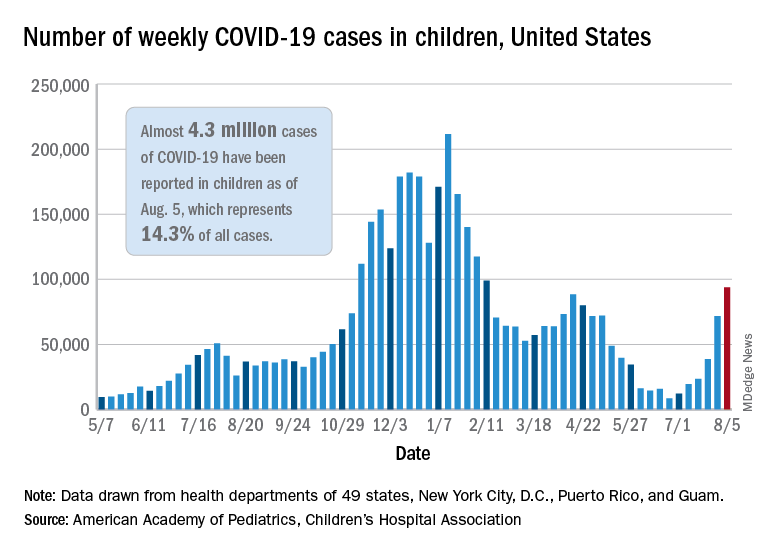
New pediatric cases rose for the 6th straight week, with almost 94,000 reported for the week ending Aug. 5.
That weekly total was up by 31% over the previous week and by over 1,000% since late June, when the new-case figure was at its lowest point (8,447) since early in the pandemic, the American Academy of Pediatrics and the Children’s Hospital Association said. COVID-related deaths – 13 for the week – were also higher than at any time since March 2021.
Almost 4.3 million children have been infected with SARS-CoV-2, which is 14.3% of all cases reported in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. Children represented 15.0% of the new cases reported in those jurisdictions during the week ending Aug. 5, the AAP and CHA said in their weekly report.
Another measure that has been trending upward recently is vaccine initiation among 12- to 15-year-olds, although the latest weekly total is still well below the high of 1.4 million seen in May. First-time vaccinations reached almost 411,000 for the week of Aug. 3-9, marking the fourth consecutive increase in that age group, the Centers for Disease Control and Prevention said on its COVID Data Tracker. Vaccinations also increased, although more modestly, for 16- and 17-year-olds in the most recent week.
Cumulative figures for children aged 12-17 show that almost 10.4 million have received at least one dose and that 7.7 million are fully vaccinated as of Aug. 9. By age group, 42.2% of those aged 12-15 have received at least one dose, and 30.4% have completed the vaccine regimen. Among those aged 16-17 years, 52.2% have gotten their first dose, and 41.4% are fully vaccinated, according to the COVID Data Tracker.
Looking at vaccination rates on the state level shows that only 20% of children aged 12-17 in Wyoming and 21% in Mississippi have gotten at least one dose as of Aug. 4, while Massachusetts is up to 68% and Vermont reports 70%. Rates for full vaccination range from 11% in Mississippi and Alabama to 61% in Vermont, based on an AAP analysis of CDC data, which is not available for Idaho.
The current COVID-19 surge has brought new cases in children to their highest level since February, according to a new report.

New pediatric cases rose for the 6th straight week, with almost 94,000 reported for the week ending Aug. 5.
That weekly total was up by 31% over the previous week and by over 1,000% since late June, when the new-case figure was at its lowest point (8,447) since early in the pandemic, the American Academy of Pediatrics and the Children’s Hospital Association said. COVID-related deaths – 13 for the week – were also higher than at any time since March 2021.
Almost 4.3 million children have been infected with SARS-CoV-2, which is 14.3% of all cases reported in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. Children represented 15.0% of the new cases reported in those jurisdictions during the week ending Aug. 5, the AAP and CHA said in their weekly report.
Another measure that has been trending upward recently is vaccine initiation among 12- to 15-year-olds, although the latest weekly total is still well below the high of 1.4 million seen in May. First-time vaccinations reached almost 411,000 for the week of Aug. 3-9, marking the fourth consecutive increase in that age group, the Centers for Disease Control and Prevention said on its COVID Data Tracker. Vaccinations also increased, although more modestly, for 16- and 17-year-olds in the most recent week.
Cumulative figures for children aged 12-17 show that almost 10.4 million have received at least one dose and that 7.7 million are fully vaccinated as of Aug. 9. By age group, 42.2% of those aged 12-15 have received at least one dose, and 30.4% have completed the vaccine regimen. Among those aged 16-17 years, 52.2% have gotten their first dose, and 41.4% are fully vaccinated, according to the COVID Data Tracker.
Looking at vaccination rates on the state level shows that only 20% of children aged 12-17 in Wyoming and 21% in Mississippi have gotten at least one dose as of Aug. 4, while Massachusetts is up to 68% and Vermont reports 70%. Rates for full vaccination range from 11% in Mississippi and Alabama to 61% in Vermont, based on an AAP analysis of CDC data, which is not available for Idaho.
The current COVID-19 surge has brought new cases in children to their highest level since February, according to a new report.

New pediatric cases rose for the 6th straight week, with almost 94,000 reported for the week ending Aug. 5.
That weekly total was up by 31% over the previous week and by over 1,000% since late June, when the new-case figure was at its lowest point (8,447) since early in the pandemic, the American Academy of Pediatrics and the Children’s Hospital Association said. COVID-related deaths – 13 for the week – were also higher than at any time since March 2021.
Almost 4.3 million children have been infected with SARS-CoV-2, which is 14.3% of all cases reported in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. Children represented 15.0% of the new cases reported in those jurisdictions during the week ending Aug. 5, the AAP and CHA said in their weekly report.
Another measure that has been trending upward recently is vaccine initiation among 12- to 15-year-olds, although the latest weekly total is still well below the high of 1.4 million seen in May. First-time vaccinations reached almost 411,000 for the week of Aug. 3-9, marking the fourth consecutive increase in that age group, the Centers for Disease Control and Prevention said on its COVID Data Tracker. Vaccinations also increased, although more modestly, for 16- and 17-year-olds in the most recent week.
Cumulative figures for children aged 12-17 show that almost 10.4 million have received at least one dose and that 7.7 million are fully vaccinated as of Aug. 9. By age group, 42.2% of those aged 12-15 have received at least one dose, and 30.4% have completed the vaccine regimen. Among those aged 16-17 years, 52.2% have gotten their first dose, and 41.4% are fully vaccinated, according to the COVID Data Tracker.
Looking at vaccination rates on the state level shows that only 20% of children aged 12-17 in Wyoming and 21% in Mississippi have gotten at least one dose as of Aug. 4, while Massachusetts is up to 68% and Vermont reports 70%. Rates for full vaccination range from 11% in Mississippi and Alabama to 61% in Vermont, based on an AAP analysis of CDC data, which is not available for Idaho.
Heart doc offering ‘fountain of youth’ jailed for 6 1/2 years
Cardiologist Samirkumar J. Shah, MD, was sentenced to 78 months in prison after his conviction on two counts of federal health care fraud involving more than $13 million.
As part of his sentence, Dr. Shah, 58, of Fox Chapel, Pa., must pay $1.7 million in restitution and other penalties and undergo 3 years of supervised release after prison.
“Dr. Shah risked the health of his patients so he could make millions of dollars through unnecessary procedures, and lied and fabricated records for years to perpetuate his fraud scheme,” acting U.S. Attorney Stephen R. Kaufman said in an Aug. 5 statement from the Department of Justice.
As previously reported, Dr. Shah was convicted June 14, 2019, of submitting fraudulent claims to private and federal insurance programs between 2008 and 2013 for external counterpulsation (ECP) therapy, a lower limb compression treatment approved for patients with coronary artery disease and refractory angina.
Dr. Shah, however, advertised ECP as the “fountain of youth,” claimed it made patients “younger and smarter,” and offered the treatment for conditions such as obesity, hypertension, hypotension, diabetes, and erectile dysfunction.
Patients were required to undergo diagnostic ultrasounds as a precautionary measure prior to starting ECP, but witness testimony established that Dr. Shah did not review any of the imaging before approving new patients for ECP, placing his patients at risk for serious injury or even death, the DOJ stated.
The evidence also showed that Dr. Shah double-billed insurers, routinely submitted fabricated patient files, and made false statements concerning his practice, patient population, recording keeping, and compliance with coverage guidelines, the government said.
During the scheme, Dr. Shah submitted ECP-related claims for Medicare Part B, UPMC Health Plan, Highmark Blue Cross Blue Shield, and Gateway Health Plan beneficiaries totalling more than $13 million and received reimbursement payments in excess of $3.5 million.
“Rather than upholding the oath he swore and providing care for patients who trusted him, this defendant misled patients and drained critical Medicaid funds from families who needed it,” said Attorney General Josh Shapiro. “We will not let anyone put their patients’ lives at risk for a profit.”
“Today’s sentence holds Mr. Shah accountable for his appalling actions,” said FBI Pittsburgh Special Agent in Charge Mike Nordwall. “Mr. Shah used his position as a doctor to illegally profit from a health care program paid for by taxpayers. Fraud of this magnitude will not be tolerated.”
Dr. Shah has been in custody since July 15, 2021, after skipping out on his original July 14 sentencing date. The Tribune-Review reported that Dr. Shah filed a last-minute request for a continuance, claiming he had an adverse reaction to the Pfizer COVID-19 vaccination and was advised by his doctor that he needed “strict bedrest for at least 6 weeks.”
Dr. Shah reportedly turned himself after presiding U.S. District Judge David S. Cercone denied the motion and issued an arrest warrant.
A version of this article first appeared on Medscape.com.
Cardiologist Samirkumar J. Shah, MD, was sentenced to 78 months in prison after his conviction on two counts of federal health care fraud involving more than $13 million.
As part of his sentence, Dr. Shah, 58, of Fox Chapel, Pa., must pay $1.7 million in restitution and other penalties and undergo 3 years of supervised release after prison.
“Dr. Shah risked the health of his patients so he could make millions of dollars through unnecessary procedures, and lied and fabricated records for years to perpetuate his fraud scheme,” acting U.S. Attorney Stephen R. Kaufman said in an Aug. 5 statement from the Department of Justice.
As previously reported, Dr. Shah was convicted June 14, 2019, of submitting fraudulent claims to private and federal insurance programs between 2008 and 2013 for external counterpulsation (ECP) therapy, a lower limb compression treatment approved for patients with coronary artery disease and refractory angina.
Dr. Shah, however, advertised ECP as the “fountain of youth,” claimed it made patients “younger and smarter,” and offered the treatment for conditions such as obesity, hypertension, hypotension, diabetes, and erectile dysfunction.
Patients were required to undergo diagnostic ultrasounds as a precautionary measure prior to starting ECP, but witness testimony established that Dr. Shah did not review any of the imaging before approving new patients for ECP, placing his patients at risk for serious injury or even death, the DOJ stated.
The evidence also showed that Dr. Shah double-billed insurers, routinely submitted fabricated patient files, and made false statements concerning his practice, patient population, recording keeping, and compliance with coverage guidelines, the government said.
During the scheme, Dr. Shah submitted ECP-related claims for Medicare Part B, UPMC Health Plan, Highmark Blue Cross Blue Shield, and Gateway Health Plan beneficiaries totalling more than $13 million and received reimbursement payments in excess of $3.5 million.
“Rather than upholding the oath he swore and providing care for patients who trusted him, this defendant misled patients and drained critical Medicaid funds from families who needed it,” said Attorney General Josh Shapiro. “We will not let anyone put their patients’ lives at risk for a profit.”
“Today’s sentence holds Mr. Shah accountable for his appalling actions,” said FBI Pittsburgh Special Agent in Charge Mike Nordwall. “Mr. Shah used his position as a doctor to illegally profit from a health care program paid for by taxpayers. Fraud of this magnitude will not be tolerated.”
Dr. Shah has been in custody since July 15, 2021, after skipping out on his original July 14 sentencing date. The Tribune-Review reported that Dr. Shah filed a last-minute request for a continuance, claiming he had an adverse reaction to the Pfizer COVID-19 vaccination and was advised by his doctor that he needed “strict bedrest for at least 6 weeks.”
Dr. Shah reportedly turned himself after presiding U.S. District Judge David S. Cercone denied the motion and issued an arrest warrant.
A version of this article first appeared on Medscape.com.
Cardiologist Samirkumar J. Shah, MD, was sentenced to 78 months in prison after his conviction on two counts of federal health care fraud involving more than $13 million.
As part of his sentence, Dr. Shah, 58, of Fox Chapel, Pa., must pay $1.7 million in restitution and other penalties and undergo 3 years of supervised release after prison.
“Dr. Shah risked the health of his patients so he could make millions of dollars through unnecessary procedures, and lied and fabricated records for years to perpetuate his fraud scheme,” acting U.S. Attorney Stephen R. Kaufman said in an Aug. 5 statement from the Department of Justice.
As previously reported, Dr. Shah was convicted June 14, 2019, of submitting fraudulent claims to private and federal insurance programs between 2008 and 2013 for external counterpulsation (ECP) therapy, a lower limb compression treatment approved for patients with coronary artery disease and refractory angina.
Dr. Shah, however, advertised ECP as the “fountain of youth,” claimed it made patients “younger and smarter,” and offered the treatment for conditions such as obesity, hypertension, hypotension, diabetes, and erectile dysfunction.
Patients were required to undergo diagnostic ultrasounds as a precautionary measure prior to starting ECP, but witness testimony established that Dr. Shah did not review any of the imaging before approving new patients for ECP, placing his patients at risk for serious injury or even death, the DOJ stated.
The evidence also showed that Dr. Shah double-billed insurers, routinely submitted fabricated patient files, and made false statements concerning his practice, patient population, recording keeping, and compliance with coverage guidelines, the government said.
During the scheme, Dr. Shah submitted ECP-related claims for Medicare Part B, UPMC Health Plan, Highmark Blue Cross Blue Shield, and Gateway Health Plan beneficiaries totalling more than $13 million and received reimbursement payments in excess of $3.5 million.
“Rather than upholding the oath he swore and providing care for patients who trusted him, this defendant misled patients and drained critical Medicaid funds from families who needed it,” said Attorney General Josh Shapiro. “We will not let anyone put their patients’ lives at risk for a profit.”
“Today’s sentence holds Mr. Shah accountable for his appalling actions,” said FBI Pittsburgh Special Agent in Charge Mike Nordwall. “Mr. Shah used his position as a doctor to illegally profit from a health care program paid for by taxpayers. Fraud of this magnitude will not be tolerated.”
Dr. Shah has been in custody since July 15, 2021, after skipping out on his original July 14 sentencing date. The Tribune-Review reported that Dr. Shah filed a last-minute request for a continuance, claiming he had an adverse reaction to the Pfizer COVID-19 vaccination and was advised by his doctor that he needed “strict bedrest for at least 6 weeks.”
Dr. Shah reportedly turned himself after presiding U.S. District Judge David S. Cercone denied the motion and issued an arrest warrant.
A version of this article first appeared on Medscape.com.
Medical residents need breastfeeding support too
As working mothers with babies in tow when the COVID-19 crisis struck, countless uncertainties threatened our already precarious work-life balance. We suddenly had many questions:
“If my daycare closes, what will I do for childcare?”
“How do I navigate diaper changes, feedings, and naps with my hectic remote work schedule?”
“If I’m constantly interrupted during the day, should I skip sleep to catch up on work and not let my colleagues down?”
As professionals who work closely with medical trainees, we knew our parenting dilemmas were being experienced even more acutely by our frontline worker colleagues.
Medical training is an increasingly common time to start a family. In a recent study, 34% of trainees in Harvard-affiliated residency programs became parents during training, and another 52% planned to do so. Trainees have higher breastfeeding initiation rates but lower continuation rates than the general population. Early nursing cessation among trainees is well documented nationally and is most often attributed to work-related barriers. These barriers range from insufficient time and limited access to facilities to a lack of support and discrimination by supervisors and peers.
This trend does not discriminate by specialty. Even among training programs known to be “family friendly,” the average duration of nursing is just 4.5 months. Residents of color are disproportionately affected by inadequate support. Studies show that Black parents breastfeed at lower rates than White parents. This has been largely attributed to structural racism and implicit bias, such as Black parents receiving less assistance initiating nursing after delivery. Adequate lactation support and inclusivity are also lacking for transgender parents who choose to breastfeed or chestfeed.
The very nature of residency training, which includes shifts that can span more than 24 hours, conflicts with many health-promoting behaviors like sleeping and eating well. However, its interference with lactation is correlated with gender. Women are disproportionately affected by the negative outcomes of unmet lactation goals. These include work-life imbalance, career dissatisfaction, and negative emotions. In a study of pediatric residents, one in four did not achieve their breastfeeding goals. Respondents reported feeling “sad, devastated, defeated, disappointed, guilty, embarrassed, frustrated, angry, like a failure, and inadequate.” Among physician mothers more broadly, discrimination related to pregnancy, parental leave, and nursing is associated with higher self-reported burnout.
Navigating nursing during residency training has more than just emotional and psychological consequences – it also has professional ones. Pursuing personal lactation goals can delay residency program completion and board certification, influence specialty selection, negatively impact research productivity, impede career advancement, and lead to misgivings about career choice.
Trainees and their families are not the only ones harmed by inadequate support in residency programs. Patients and their families are affected, too. Research suggests that physicians’ personal breastfeeding practices affect the advice they give to patients. Those who receive lactation support are more likely to help patients meet their own goals. In the previously mentioned study of pediatric residents, more than 90% of the 400 respondents said their own or their partner’s nursing experience affected their interaction with lactating patients in their clinic or hospital.
Increased lactation support is a straightforward, low-cost, high-impact intervention. It benefits trainee well-being, satisfaction, workflow, and future patient care. The Accreditation Council for Graduate Medical Education mandated in July 2019 that all residency programs provide adequate lactation facilities – including refrigeration capabilities and proximity for safe patient care. However, to our knowledge, rates of compliance with this new policy and citation for noncompliance have yet to be seen. Regardless, facilities alone are not enough. Residency programs should develop and enforce formal lactation policies.
Several institutions have successfully piloted such policies in recent years. One in particular from the University of Michigan’s surgery residency program inspired the development of a lactation policy within the internal medicine residency at our institution. These policies designate appropriate spaces at each clinical rotation site, clarify that residents are encouraged to take pumping breaks as needed – in coordination with clinical teams so as not to compromise patient care – and communicate support from supervisors.
Our program also established an informal peer mentoring program. Residents with experience pumping at work pair up with newer trainees. The policy benefits residents who wish to chestfeed or breastfeed, normalizes lactation, and empowers trainees by diminishing the need to ask for individual accommodations. It also costs the program nothing.
As more women enter medicine and more trainees become parents during residency, the need for support in this area will only continue to grow. The widespread lack of such resources, and the fact that clean and private facilities are only now being mandated, is symbolic. If even this basic need is rarely acknowledged or met, what other resident needs are being neglected?
A version of this article first appeared on Medscape.com.
As working mothers with babies in tow when the COVID-19 crisis struck, countless uncertainties threatened our already precarious work-life balance. We suddenly had many questions:
“If my daycare closes, what will I do for childcare?”
“How do I navigate diaper changes, feedings, and naps with my hectic remote work schedule?”
“If I’m constantly interrupted during the day, should I skip sleep to catch up on work and not let my colleagues down?”
As professionals who work closely with medical trainees, we knew our parenting dilemmas were being experienced even more acutely by our frontline worker colleagues.
Medical training is an increasingly common time to start a family. In a recent study, 34% of trainees in Harvard-affiliated residency programs became parents during training, and another 52% planned to do so. Trainees have higher breastfeeding initiation rates but lower continuation rates than the general population. Early nursing cessation among trainees is well documented nationally and is most often attributed to work-related barriers. These barriers range from insufficient time and limited access to facilities to a lack of support and discrimination by supervisors and peers.
This trend does not discriminate by specialty. Even among training programs known to be “family friendly,” the average duration of nursing is just 4.5 months. Residents of color are disproportionately affected by inadequate support. Studies show that Black parents breastfeed at lower rates than White parents. This has been largely attributed to structural racism and implicit bias, such as Black parents receiving less assistance initiating nursing after delivery. Adequate lactation support and inclusivity are also lacking for transgender parents who choose to breastfeed or chestfeed.
The very nature of residency training, which includes shifts that can span more than 24 hours, conflicts with many health-promoting behaviors like sleeping and eating well. However, its interference with lactation is correlated with gender. Women are disproportionately affected by the negative outcomes of unmet lactation goals. These include work-life imbalance, career dissatisfaction, and negative emotions. In a study of pediatric residents, one in four did not achieve their breastfeeding goals. Respondents reported feeling “sad, devastated, defeated, disappointed, guilty, embarrassed, frustrated, angry, like a failure, and inadequate.” Among physician mothers more broadly, discrimination related to pregnancy, parental leave, and nursing is associated with higher self-reported burnout.
Navigating nursing during residency training has more than just emotional and psychological consequences – it also has professional ones. Pursuing personal lactation goals can delay residency program completion and board certification, influence specialty selection, negatively impact research productivity, impede career advancement, and lead to misgivings about career choice.
Trainees and their families are not the only ones harmed by inadequate support in residency programs. Patients and their families are affected, too. Research suggests that physicians’ personal breastfeeding practices affect the advice they give to patients. Those who receive lactation support are more likely to help patients meet their own goals. In the previously mentioned study of pediatric residents, more than 90% of the 400 respondents said their own or their partner’s nursing experience affected their interaction with lactating patients in their clinic or hospital.
Increased lactation support is a straightforward, low-cost, high-impact intervention. It benefits trainee well-being, satisfaction, workflow, and future patient care. The Accreditation Council for Graduate Medical Education mandated in July 2019 that all residency programs provide adequate lactation facilities – including refrigeration capabilities and proximity for safe patient care. However, to our knowledge, rates of compliance with this new policy and citation for noncompliance have yet to be seen. Regardless, facilities alone are not enough. Residency programs should develop and enforce formal lactation policies.
Several institutions have successfully piloted such policies in recent years. One in particular from the University of Michigan’s surgery residency program inspired the development of a lactation policy within the internal medicine residency at our institution. These policies designate appropriate spaces at each clinical rotation site, clarify that residents are encouraged to take pumping breaks as needed – in coordination with clinical teams so as not to compromise patient care – and communicate support from supervisors.
Our program also established an informal peer mentoring program. Residents with experience pumping at work pair up with newer trainees. The policy benefits residents who wish to chestfeed or breastfeed, normalizes lactation, and empowers trainees by diminishing the need to ask for individual accommodations. It also costs the program nothing.
As more women enter medicine and more trainees become parents during residency, the need for support in this area will only continue to grow. The widespread lack of such resources, and the fact that clean and private facilities are only now being mandated, is symbolic. If even this basic need is rarely acknowledged or met, what other resident needs are being neglected?
A version of this article first appeared on Medscape.com.
As working mothers with babies in tow when the COVID-19 crisis struck, countless uncertainties threatened our already precarious work-life balance. We suddenly had many questions:
“If my daycare closes, what will I do for childcare?”
“How do I navigate diaper changes, feedings, and naps with my hectic remote work schedule?”
“If I’m constantly interrupted during the day, should I skip sleep to catch up on work and not let my colleagues down?”
As professionals who work closely with medical trainees, we knew our parenting dilemmas were being experienced even more acutely by our frontline worker colleagues.
Medical training is an increasingly common time to start a family. In a recent study, 34% of trainees in Harvard-affiliated residency programs became parents during training, and another 52% planned to do so. Trainees have higher breastfeeding initiation rates but lower continuation rates than the general population. Early nursing cessation among trainees is well documented nationally and is most often attributed to work-related barriers. These barriers range from insufficient time and limited access to facilities to a lack of support and discrimination by supervisors and peers.
This trend does not discriminate by specialty. Even among training programs known to be “family friendly,” the average duration of nursing is just 4.5 months. Residents of color are disproportionately affected by inadequate support. Studies show that Black parents breastfeed at lower rates than White parents. This has been largely attributed to structural racism and implicit bias, such as Black parents receiving less assistance initiating nursing after delivery. Adequate lactation support and inclusivity are also lacking for transgender parents who choose to breastfeed or chestfeed.
The very nature of residency training, which includes shifts that can span more than 24 hours, conflicts with many health-promoting behaviors like sleeping and eating well. However, its interference with lactation is correlated with gender. Women are disproportionately affected by the negative outcomes of unmet lactation goals. These include work-life imbalance, career dissatisfaction, and negative emotions. In a study of pediatric residents, one in four did not achieve their breastfeeding goals. Respondents reported feeling “sad, devastated, defeated, disappointed, guilty, embarrassed, frustrated, angry, like a failure, and inadequate.” Among physician mothers more broadly, discrimination related to pregnancy, parental leave, and nursing is associated with higher self-reported burnout.
Navigating nursing during residency training has more than just emotional and psychological consequences – it also has professional ones. Pursuing personal lactation goals can delay residency program completion and board certification, influence specialty selection, negatively impact research productivity, impede career advancement, and lead to misgivings about career choice.
Trainees and their families are not the only ones harmed by inadequate support in residency programs. Patients and their families are affected, too. Research suggests that physicians’ personal breastfeeding practices affect the advice they give to patients. Those who receive lactation support are more likely to help patients meet their own goals. In the previously mentioned study of pediatric residents, more than 90% of the 400 respondents said their own or their partner’s nursing experience affected their interaction with lactating patients in their clinic or hospital.
Increased lactation support is a straightforward, low-cost, high-impact intervention. It benefits trainee well-being, satisfaction, workflow, and future patient care. The Accreditation Council for Graduate Medical Education mandated in July 2019 that all residency programs provide adequate lactation facilities – including refrigeration capabilities and proximity for safe patient care. However, to our knowledge, rates of compliance with this new policy and citation for noncompliance have yet to be seen. Regardless, facilities alone are not enough. Residency programs should develop and enforce formal lactation policies.
Several institutions have successfully piloted such policies in recent years. One in particular from the University of Michigan’s surgery residency program inspired the development of a lactation policy within the internal medicine residency at our institution. These policies designate appropriate spaces at each clinical rotation site, clarify that residents are encouraged to take pumping breaks as needed – in coordination with clinical teams so as not to compromise patient care – and communicate support from supervisors.
Our program also established an informal peer mentoring program. Residents with experience pumping at work pair up with newer trainees. The policy benefits residents who wish to chestfeed or breastfeed, normalizes lactation, and empowers trainees by diminishing the need to ask for individual accommodations. It also costs the program nothing.
As more women enter medicine and more trainees become parents during residency, the need for support in this area will only continue to grow. The widespread lack of such resources, and the fact that clean and private facilities are only now being mandated, is symbolic. If even this basic need is rarely acknowledged or met, what other resident needs are being neglected?
A version of this article first appeared on Medscape.com.
Better CNS control in children with ALL: ‘Goldilocks’ approach
Treatment of central nervous system involvement in pediatric acute lymphoblastic leukemia (ALL) needs to be based on risk, and should no longer be applied in a one-size-fits all approach, say experts writing in an editorial published July 29 in the journal Blood.
“Because cure rates now exceed 90%, using neurotoxic drugs in non–risk-adapted protocols is unacceptable and a paradigm shift in treating CNS ALL is required,” write pediatric leukemia researchers Christina Halsey, PhD, of the University of Glasgow and Gabriele Escherich, MD, of the University Medical Center Hamburg-Eppendorf (Germany).
“We want to reach a Goldilocks point: not too much, not too little, but just right for every child,” they write.
The problem is that “the absence of clinically useful biomarkers prevents accurate risk stratification, resulting in universal use of intensive CNS-directed therapy. This therapy is likely to overtreat many children, exposing them to an unnecessary risk of toxicity,” including long-term cognitive deficits in 20%-40% of them, they point out.
The editorial accompanied a new study in which investigators in China focused on improving CNS control in children with ALL, which the editorialists said was step in the right direction.
In the Chinese Children’s Cancer Group ALL-2015 trial, investigators found that prephase dexamethasone, delayed intrathecal therapy, intravenous anesthesia to reduce traumatic lumbar punctures, and flow cytometry to accurately ascertain initial CNS involvement may improve CNS control.
The trial included 7,640 consecutive children up to age 18 treated from 2015 to 2019 across 20 major medical centers in China. Children received conventional remission induction and subsequent risk-directed therapy, including 16-22 triple intrathecal treatments. Prophylactic cranial irradiation was not used.
The 5-year event-free survival was 80.3% and overall survival 91.1%. The cumulative risk of isolated CNS relapse was 1.9% and of any CNS relapse 2.7%, comparable to reports from other major study groups, both with and without cranial radiation.
“We attributed our relatively good CNS control to the prephase treatment with dexamethasone, which reduced leukemia cells in blood and the CNS, and to the delayed intrathecal therapy until all (or a large proportion) of circulating leukemic blasts were cleared, thus reducing the consequence of traumatic lumbar puncture with blasts,” said the investigators, led by Jingyan Tang, MD, a hematologist/oncologist at the Shanghai (China) Children’s Medical Center.
“This approach of delayed administration of initial intrathecal therapy after prephase steroid treatment, if confirmed successful by additional studies, can be adopted readily,” they say.
The editorialists concur. The low rates of CNS relapse, despite omission of radiotherapy and inclusion of high-risk subgroups, “might suggest a potential protective effect of steroids before diagnostic lumbar puncture,” they said.
“However, flow cytometry is not sensitive enough to track disease response over time. In the bone marrow, minimal residual disease (MRD) is used to identify children at high or low risk of relapse and modify therapy accordingly. We desperately need a minimal residual disease equivalent for CNS leukemia to allow us to tailor therapy,” Dr. Halsey and Dr. Escherich say.
It’s not surprising that the use of anesthesia led to fewer traumatic lumbar punctures than in “frightened child[ren] undergoing such a painful procedure without anesthesia,” the study team notes. Its correlation with lower CNS relapses is probably because drug delivery was more accurate in sedated children, the editorialists add.
Female sex was also protective against relapse in cases where general anesthesia wasn’t used for lumbar puncture. “One could speculate that it is more difficult to restrict male patients than female patients for successful intrathecal therapy if they were not undergoing anesthesia during the procedure,” the investigators write.
“Unfortunately,” the editorialists add, rapid adoption of anesthesia for lumbar punctures “is tempered by the recent observation that repeated general anesthesia in children with ALL is associated with increased neurotoxicity.”
The work was supported by grants from the National Natural Science Foundation of China, National Cancer Institute, and others. The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Treatment of central nervous system involvement in pediatric acute lymphoblastic leukemia (ALL) needs to be based on risk, and should no longer be applied in a one-size-fits all approach, say experts writing in an editorial published July 29 in the journal Blood.
“Because cure rates now exceed 90%, using neurotoxic drugs in non–risk-adapted protocols is unacceptable and a paradigm shift in treating CNS ALL is required,” write pediatric leukemia researchers Christina Halsey, PhD, of the University of Glasgow and Gabriele Escherich, MD, of the University Medical Center Hamburg-Eppendorf (Germany).
“We want to reach a Goldilocks point: not too much, not too little, but just right for every child,” they write.
The problem is that “the absence of clinically useful biomarkers prevents accurate risk stratification, resulting in universal use of intensive CNS-directed therapy. This therapy is likely to overtreat many children, exposing them to an unnecessary risk of toxicity,” including long-term cognitive deficits in 20%-40% of them, they point out.
The editorial accompanied a new study in which investigators in China focused on improving CNS control in children with ALL, which the editorialists said was step in the right direction.
In the Chinese Children’s Cancer Group ALL-2015 trial, investigators found that prephase dexamethasone, delayed intrathecal therapy, intravenous anesthesia to reduce traumatic lumbar punctures, and flow cytometry to accurately ascertain initial CNS involvement may improve CNS control.
The trial included 7,640 consecutive children up to age 18 treated from 2015 to 2019 across 20 major medical centers in China. Children received conventional remission induction and subsequent risk-directed therapy, including 16-22 triple intrathecal treatments. Prophylactic cranial irradiation was not used.
The 5-year event-free survival was 80.3% and overall survival 91.1%. The cumulative risk of isolated CNS relapse was 1.9% and of any CNS relapse 2.7%, comparable to reports from other major study groups, both with and without cranial radiation.
“We attributed our relatively good CNS control to the prephase treatment with dexamethasone, which reduced leukemia cells in blood and the CNS, and to the delayed intrathecal therapy until all (or a large proportion) of circulating leukemic blasts were cleared, thus reducing the consequence of traumatic lumbar puncture with blasts,” said the investigators, led by Jingyan Tang, MD, a hematologist/oncologist at the Shanghai (China) Children’s Medical Center.
“This approach of delayed administration of initial intrathecal therapy after prephase steroid treatment, if confirmed successful by additional studies, can be adopted readily,” they say.
The editorialists concur. The low rates of CNS relapse, despite omission of radiotherapy and inclusion of high-risk subgroups, “might suggest a potential protective effect of steroids before diagnostic lumbar puncture,” they said.
“However, flow cytometry is not sensitive enough to track disease response over time. In the bone marrow, minimal residual disease (MRD) is used to identify children at high or low risk of relapse and modify therapy accordingly. We desperately need a minimal residual disease equivalent for CNS leukemia to allow us to tailor therapy,” Dr. Halsey and Dr. Escherich say.
It’s not surprising that the use of anesthesia led to fewer traumatic lumbar punctures than in “frightened child[ren] undergoing such a painful procedure without anesthesia,” the study team notes. Its correlation with lower CNS relapses is probably because drug delivery was more accurate in sedated children, the editorialists add.
Female sex was also protective against relapse in cases where general anesthesia wasn’t used for lumbar puncture. “One could speculate that it is more difficult to restrict male patients than female patients for successful intrathecal therapy if they were not undergoing anesthesia during the procedure,” the investigators write.
“Unfortunately,” the editorialists add, rapid adoption of anesthesia for lumbar punctures “is tempered by the recent observation that repeated general anesthesia in children with ALL is associated with increased neurotoxicity.”
The work was supported by grants from the National Natural Science Foundation of China, National Cancer Institute, and others. The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Treatment of central nervous system involvement in pediatric acute lymphoblastic leukemia (ALL) needs to be based on risk, and should no longer be applied in a one-size-fits all approach, say experts writing in an editorial published July 29 in the journal Blood.
“Because cure rates now exceed 90%, using neurotoxic drugs in non–risk-adapted protocols is unacceptable and a paradigm shift in treating CNS ALL is required,” write pediatric leukemia researchers Christina Halsey, PhD, of the University of Glasgow and Gabriele Escherich, MD, of the University Medical Center Hamburg-Eppendorf (Germany).
“We want to reach a Goldilocks point: not too much, not too little, but just right for every child,” they write.
The problem is that “the absence of clinically useful biomarkers prevents accurate risk stratification, resulting in universal use of intensive CNS-directed therapy. This therapy is likely to overtreat many children, exposing them to an unnecessary risk of toxicity,” including long-term cognitive deficits in 20%-40% of them, they point out.
The editorial accompanied a new study in which investigators in China focused on improving CNS control in children with ALL, which the editorialists said was step in the right direction.
In the Chinese Children’s Cancer Group ALL-2015 trial, investigators found that prephase dexamethasone, delayed intrathecal therapy, intravenous anesthesia to reduce traumatic lumbar punctures, and flow cytometry to accurately ascertain initial CNS involvement may improve CNS control.
The trial included 7,640 consecutive children up to age 18 treated from 2015 to 2019 across 20 major medical centers in China. Children received conventional remission induction and subsequent risk-directed therapy, including 16-22 triple intrathecal treatments. Prophylactic cranial irradiation was not used.
The 5-year event-free survival was 80.3% and overall survival 91.1%. The cumulative risk of isolated CNS relapse was 1.9% and of any CNS relapse 2.7%, comparable to reports from other major study groups, both with and without cranial radiation.
“We attributed our relatively good CNS control to the prephase treatment with dexamethasone, which reduced leukemia cells in blood and the CNS, and to the delayed intrathecal therapy until all (or a large proportion) of circulating leukemic blasts were cleared, thus reducing the consequence of traumatic lumbar puncture with blasts,” said the investigators, led by Jingyan Tang, MD, a hematologist/oncologist at the Shanghai (China) Children’s Medical Center.
“This approach of delayed administration of initial intrathecal therapy after prephase steroid treatment, if confirmed successful by additional studies, can be adopted readily,” they say.
The editorialists concur. The low rates of CNS relapse, despite omission of radiotherapy and inclusion of high-risk subgroups, “might suggest a potential protective effect of steroids before diagnostic lumbar puncture,” they said.
“However, flow cytometry is not sensitive enough to track disease response over time. In the bone marrow, minimal residual disease (MRD) is used to identify children at high or low risk of relapse and modify therapy accordingly. We desperately need a minimal residual disease equivalent for CNS leukemia to allow us to tailor therapy,” Dr. Halsey and Dr. Escherich say.
It’s not surprising that the use of anesthesia led to fewer traumatic lumbar punctures than in “frightened child[ren] undergoing such a painful procedure without anesthesia,” the study team notes. Its correlation with lower CNS relapses is probably because drug delivery was more accurate in sedated children, the editorialists add.
Female sex was also protective against relapse in cases where general anesthesia wasn’t used for lumbar puncture. “One could speculate that it is more difficult to restrict male patients than female patients for successful intrathecal therapy if they were not undergoing anesthesia during the procedure,” the investigators write.
“Unfortunately,” the editorialists add, rapid adoption of anesthesia for lumbar punctures “is tempered by the recent observation that repeated general anesthesia in children with ALL is associated with increased neurotoxicity.”
The work was supported by grants from the National Natural Science Foundation of China, National Cancer Institute, and others. The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Global youth depression and anxiety doubled during pandemic
The COVID-19 pandemic doubled international rates of child and adolescent psychological disorders, according to results of a meta-analysis.
In the first year of the pandemic, an estimated one in four youth across various regions of the globe experienced clinically elevated depression symptoms, while one in five experienced clinically elevated anxiety symptoms. These pooled estimates, which increased over time, are double prepandemic estimates, according to Nicole Racine, PhD, RPsych, a clinical psychologist at the University of Calgary (Alta.) and colleagues.
Their meta-analysis of 29 studies, comprising 80,879 young people worldwide aged 18 years or less, found pooled prevalence estimates of clinically elevated youth depression and anxiety of 25.2% (95% confidence interval, 21.2%-29.7%) and 20.5% (95% CI, 17.2%-24.4%), respectively.
“The prevalence of depression and anxiety symptoms during COVID-19 [has] doubled, compared with prepandemic estimates, and moderator analyses revealed that prevalence rates were higher when collected later in the pandemic, in older adolescents, and in girls,” the researchers write online in JAMA Pediatrics.
Prepandemic estimates of clinically significant generalized anxiety and depressive symptoms in large youth cohorts were approximately 11.6% and 12.9%, respectively, the authors say.
The increases revealed in these international findings have implications for targeted mental health resource planning.
“One difficulty in the literature is that there are large discrepancies on the prevalence of child depression and anxiety during the COVID-19 pandemic, with published rates between 2% and 68%,” corresponding author Sheri Madigan, PhD, RPsych, of the University of Calgary department of psychology, said in an interview. “By conducting a synthesis of the 29 studies on over 80,000 children, we were able to determine that, on average across these studies, 25% of youth are experiencing depression and 20% are experiencing anxiety during the COVID-19 pandemic.”
The cohort
The mean age in the combined global cohort was 13 years (range 4.1-17.6 ), and the mean proportion of females was 52.7% (standard deviation) 12.3%). The findings were based on international data published from Jan. 1, 2020, to Feb. 16, 2021, in studies conducted in the Middle East (n = 1), Europe (n = 4), South America (n = 2), North America (n = 6), and East Asia (n = 16). Notably absent were data from most of Latin America and the Middle East, Africa, South East Asia, and the Pacific Islands.
As the year progressed, the prevalence of depressive symptoms rose (b = .26; 95% CI, .06-46) with the number of months elapsed. Prevalence rates also rose as both age (b = 0.08, 95% CI, 0.01-0.15), and the percentage of females in samples increased (b = .03; 95% CI, 0.01-0.05).
The authors surmise that this cumulative worsening might be because of prolonged social isolation, family financial difficulties, missed milestones, and school disruptions, which are compounded over time. A second possibility is that studies conducted in the earlier months of the pandemic were more likely to be conducted in East Asia, where the self-reported prevalence of mental health symptoms tends to be lower.
The findings highlight an urgent need for intervention and recovery efforts and also indicate the need to consider individual differences when determining targets for intervention, including age, sex, and exposure to COVID-19 stressors), they add.
Even more concerning, recent data from the Centers for Disease Control and Prevention suggest that the pandemic spurred an increase in suspected suicide attempts by teenage girls. In the United Kingdom, acute mental health presentations to emergency care tripled over 2019 at one pediatric facility during the pandemic.
The authors attribute the toll on the psychological well-being of the world’s young people to pandemic-mandated restrictions. Those entailed loss of peer interactions, social isolation, and reduced contact with support figures such as teachers, and, “In addition, schools are often a primary location for receiving psychological services, with 80% of children relying on school-based services to address their mental health needs.” For many children, these services were rendered unavailable owing to school closures, Dr. Madigan and associates write.
In the context of clinical practice, doctors play a critical role. “With school closures, the physician’s office may be the only mental health checkpoint for youth,” Dr. Madigan said “So I recommend that family physicians screen for, and/or ask children and youth, about their mental health.”
On the home front, emerging research suggests that a predictable home environment can protect children’s mental well-being, with less depression and fewer behavioral problems observed in families adhering to regular routines during COVID-19. “Thus, a tangible solution to help mitigate the adverse effects of COVID-19 on youth is working with children and families to implement consistent and predictable routines around schoolwork, sleep, screen use, and physical activity,” the authors write.
They also point to the need for research on the long-term effects of the pandemic on mental health, including studies in order to “augment understanding of the implications of this crisis on the mental health trajectories of today’s children and youth.”
In an accompanying editorial, Tami D. Benton, MD, psychiatrist-in-chief at Children’s Hospital of Philadelphia, and colleagues, who were not involved in the meta-analysis, note certain limitations to the study. First, the included studies are based on self- or parent-reported symptoms. Second, the studies, more than half of which (55.2%) were done in China, may not be generalizable to all regions of the world, where 90% of children live in low- or middle-income countries.
Still, they write,“The increased mental health needs identified in the meta-analysis call for immediate action for every country. Our responses must consider the range of child mental health infrastructures available, which vary across countries, with some having well-developed and coordinated mental health services, while others have informal, limited, underfunded, or fragmented systems of care.”
Empirically supported and culturally appropriate intervention strategies for children and families according to countries and communities will be crucial, they stress.
“This meta-analysis provides the most complete evidence to date on the toll the COVID-19 pandemic has taken on child and adolescent mental health,” said Katie A. McLaughlin, PhD, a professor of psychology at Harvard University in Boston, who was not involved in the study. “The results confirm the substantial increases in symptoms of youth depression and anxiety that many clinicians and researchers have observed during the pandemic and highlight the critical need for greater investments in mental health services for children and adolescents.”
This study received no specific funding other than research support to the investigators from nonprivate entities. The authors disclosed no relevant conflicts of interest. Dr. Benton and associates and Dr. McLaughlin declared no competing interests.
The COVID-19 pandemic doubled international rates of child and adolescent psychological disorders, according to results of a meta-analysis.
In the first year of the pandemic, an estimated one in four youth across various regions of the globe experienced clinically elevated depression symptoms, while one in five experienced clinically elevated anxiety symptoms. These pooled estimates, which increased over time, are double prepandemic estimates, according to Nicole Racine, PhD, RPsych, a clinical psychologist at the University of Calgary (Alta.) and colleagues.
Their meta-analysis of 29 studies, comprising 80,879 young people worldwide aged 18 years or less, found pooled prevalence estimates of clinically elevated youth depression and anxiety of 25.2% (95% confidence interval, 21.2%-29.7%) and 20.5% (95% CI, 17.2%-24.4%), respectively.
“The prevalence of depression and anxiety symptoms during COVID-19 [has] doubled, compared with prepandemic estimates, and moderator analyses revealed that prevalence rates were higher when collected later in the pandemic, in older adolescents, and in girls,” the researchers write online in JAMA Pediatrics.
Prepandemic estimates of clinically significant generalized anxiety and depressive symptoms in large youth cohorts were approximately 11.6% and 12.9%, respectively, the authors say.
The increases revealed in these international findings have implications for targeted mental health resource planning.
“One difficulty in the literature is that there are large discrepancies on the prevalence of child depression and anxiety during the COVID-19 pandemic, with published rates between 2% and 68%,” corresponding author Sheri Madigan, PhD, RPsych, of the University of Calgary department of psychology, said in an interview. “By conducting a synthesis of the 29 studies on over 80,000 children, we were able to determine that, on average across these studies, 25% of youth are experiencing depression and 20% are experiencing anxiety during the COVID-19 pandemic.”
The cohort
The mean age in the combined global cohort was 13 years (range 4.1-17.6 ), and the mean proportion of females was 52.7% (standard deviation) 12.3%). The findings were based on international data published from Jan. 1, 2020, to Feb. 16, 2021, in studies conducted in the Middle East (n = 1), Europe (n = 4), South America (n = 2), North America (n = 6), and East Asia (n = 16). Notably absent were data from most of Latin America and the Middle East, Africa, South East Asia, and the Pacific Islands.
As the year progressed, the prevalence of depressive symptoms rose (b = .26; 95% CI, .06-46) with the number of months elapsed. Prevalence rates also rose as both age (b = 0.08, 95% CI, 0.01-0.15), and the percentage of females in samples increased (b = .03; 95% CI, 0.01-0.05).
The authors surmise that this cumulative worsening might be because of prolonged social isolation, family financial difficulties, missed milestones, and school disruptions, which are compounded over time. A second possibility is that studies conducted in the earlier months of the pandemic were more likely to be conducted in East Asia, where the self-reported prevalence of mental health symptoms tends to be lower.
The findings highlight an urgent need for intervention and recovery efforts and also indicate the need to consider individual differences when determining targets for intervention, including age, sex, and exposure to COVID-19 stressors), they add.
Even more concerning, recent data from the Centers for Disease Control and Prevention suggest that the pandemic spurred an increase in suspected suicide attempts by teenage girls. In the United Kingdom, acute mental health presentations to emergency care tripled over 2019 at one pediatric facility during the pandemic.
The authors attribute the toll on the psychological well-being of the world’s young people to pandemic-mandated restrictions. Those entailed loss of peer interactions, social isolation, and reduced contact with support figures such as teachers, and, “In addition, schools are often a primary location for receiving psychological services, with 80% of children relying on school-based services to address their mental health needs.” For many children, these services were rendered unavailable owing to school closures, Dr. Madigan and associates write.
In the context of clinical practice, doctors play a critical role. “With school closures, the physician’s office may be the only mental health checkpoint for youth,” Dr. Madigan said “So I recommend that family physicians screen for, and/or ask children and youth, about their mental health.”
On the home front, emerging research suggests that a predictable home environment can protect children’s mental well-being, with less depression and fewer behavioral problems observed in families adhering to regular routines during COVID-19. “Thus, a tangible solution to help mitigate the adverse effects of COVID-19 on youth is working with children and families to implement consistent and predictable routines around schoolwork, sleep, screen use, and physical activity,” the authors write.
They also point to the need for research on the long-term effects of the pandemic on mental health, including studies in order to “augment understanding of the implications of this crisis on the mental health trajectories of today’s children and youth.”
In an accompanying editorial, Tami D. Benton, MD, psychiatrist-in-chief at Children’s Hospital of Philadelphia, and colleagues, who were not involved in the meta-analysis, note certain limitations to the study. First, the included studies are based on self- or parent-reported symptoms. Second, the studies, more than half of which (55.2%) were done in China, may not be generalizable to all regions of the world, where 90% of children live in low- or middle-income countries.
Still, they write,“The increased mental health needs identified in the meta-analysis call for immediate action for every country. Our responses must consider the range of child mental health infrastructures available, which vary across countries, with some having well-developed and coordinated mental health services, while others have informal, limited, underfunded, or fragmented systems of care.”
Empirically supported and culturally appropriate intervention strategies for children and families according to countries and communities will be crucial, they stress.
“This meta-analysis provides the most complete evidence to date on the toll the COVID-19 pandemic has taken on child and adolescent mental health,” said Katie A. McLaughlin, PhD, a professor of psychology at Harvard University in Boston, who was not involved in the study. “The results confirm the substantial increases in symptoms of youth depression and anxiety that many clinicians and researchers have observed during the pandemic and highlight the critical need for greater investments in mental health services for children and adolescents.”
This study received no specific funding other than research support to the investigators from nonprivate entities. The authors disclosed no relevant conflicts of interest. Dr. Benton and associates and Dr. McLaughlin declared no competing interests.
The COVID-19 pandemic doubled international rates of child and adolescent psychological disorders, according to results of a meta-analysis.
In the first year of the pandemic, an estimated one in four youth across various regions of the globe experienced clinically elevated depression symptoms, while one in five experienced clinically elevated anxiety symptoms. These pooled estimates, which increased over time, are double prepandemic estimates, according to Nicole Racine, PhD, RPsych, a clinical psychologist at the University of Calgary (Alta.) and colleagues.
Their meta-analysis of 29 studies, comprising 80,879 young people worldwide aged 18 years or less, found pooled prevalence estimates of clinically elevated youth depression and anxiety of 25.2% (95% confidence interval, 21.2%-29.7%) and 20.5% (95% CI, 17.2%-24.4%), respectively.
“The prevalence of depression and anxiety symptoms during COVID-19 [has] doubled, compared with prepandemic estimates, and moderator analyses revealed that prevalence rates were higher when collected later in the pandemic, in older adolescents, and in girls,” the researchers write online in JAMA Pediatrics.
Prepandemic estimates of clinically significant generalized anxiety and depressive symptoms in large youth cohorts were approximately 11.6% and 12.9%, respectively, the authors say.
The increases revealed in these international findings have implications for targeted mental health resource planning.
“One difficulty in the literature is that there are large discrepancies on the prevalence of child depression and anxiety during the COVID-19 pandemic, with published rates between 2% and 68%,” corresponding author Sheri Madigan, PhD, RPsych, of the University of Calgary department of psychology, said in an interview. “By conducting a synthesis of the 29 studies on over 80,000 children, we were able to determine that, on average across these studies, 25% of youth are experiencing depression and 20% are experiencing anxiety during the COVID-19 pandemic.”
The cohort
The mean age in the combined global cohort was 13 years (range 4.1-17.6 ), and the mean proportion of females was 52.7% (standard deviation) 12.3%). The findings were based on international data published from Jan. 1, 2020, to Feb. 16, 2021, in studies conducted in the Middle East (n = 1), Europe (n = 4), South America (n = 2), North America (n = 6), and East Asia (n = 16). Notably absent were data from most of Latin America and the Middle East, Africa, South East Asia, and the Pacific Islands.
As the year progressed, the prevalence of depressive symptoms rose (b = .26; 95% CI, .06-46) with the number of months elapsed. Prevalence rates also rose as both age (b = 0.08, 95% CI, 0.01-0.15), and the percentage of females in samples increased (b = .03; 95% CI, 0.01-0.05).
The authors surmise that this cumulative worsening might be because of prolonged social isolation, family financial difficulties, missed milestones, and school disruptions, which are compounded over time. A second possibility is that studies conducted in the earlier months of the pandemic were more likely to be conducted in East Asia, where the self-reported prevalence of mental health symptoms tends to be lower.
The findings highlight an urgent need for intervention and recovery efforts and also indicate the need to consider individual differences when determining targets for intervention, including age, sex, and exposure to COVID-19 stressors), they add.
Even more concerning, recent data from the Centers for Disease Control and Prevention suggest that the pandemic spurred an increase in suspected suicide attempts by teenage girls. In the United Kingdom, acute mental health presentations to emergency care tripled over 2019 at one pediatric facility during the pandemic.
The authors attribute the toll on the psychological well-being of the world’s young people to pandemic-mandated restrictions. Those entailed loss of peer interactions, social isolation, and reduced contact with support figures such as teachers, and, “In addition, schools are often a primary location for receiving psychological services, with 80% of children relying on school-based services to address their mental health needs.” For many children, these services were rendered unavailable owing to school closures, Dr. Madigan and associates write.
In the context of clinical practice, doctors play a critical role. “With school closures, the physician’s office may be the only mental health checkpoint for youth,” Dr. Madigan said “So I recommend that family physicians screen for, and/or ask children and youth, about their mental health.”
On the home front, emerging research suggests that a predictable home environment can protect children’s mental well-being, with less depression and fewer behavioral problems observed in families adhering to regular routines during COVID-19. “Thus, a tangible solution to help mitigate the adverse effects of COVID-19 on youth is working with children and families to implement consistent and predictable routines around schoolwork, sleep, screen use, and physical activity,” the authors write.
They also point to the need for research on the long-term effects of the pandemic on mental health, including studies in order to “augment understanding of the implications of this crisis on the mental health trajectories of today’s children and youth.”
In an accompanying editorial, Tami D. Benton, MD, psychiatrist-in-chief at Children’s Hospital of Philadelphia, and colleagues, who were not involved in the meta-analysis, note certain limitations to the study. First, the included studies are based on self- or parent-reported symptoms. Second, the studies, more than half of which (55.2%) were done in China, may not be generalizable to all regions of the world, where 90% of children live in low- or middle-income countries.
Still, they write,“The increased mental health needs identified in the meta-analysis call for immediate action for every country. Our responses must consider the range of child mental health infrastructures available, which vary across countries, with some having well-developed and coordinated mental health services, while others have informal, limited, underfunded, or fragmented systems of care.”
Empirically supported and culturally appropriate intervention strategies for children and families according to countries and communities will be crucial, they stress.
“This meta-analysis provides the most complete evidence to date on the toll the COVID-19 pandemic has taken on child and adolescent mental health,” said Katie A. McLaughlin, PhD, a professor of psychology at Harvard University in Boston, who was not involved in the study. “The results confirm the substantial increases in symptoms of youth depression and anxiety that many clinicians and researchers have observed during the pandemic and highlight the critical need for greater investments in mental health services for children and adolescents.”
This study received no specific funding other than research support to the investigators from nonprivate entities. The authors disclosed no relevant conflicts of interest. Dr. Benton and associates and Dr. McLaughlin declared no competing interests.
Exposure to marijuana smoke linked to increased risk of respiratory infections in children
Exposure to secondhand marijuana smoke is more strongly associated with viral respiratory infections in children, compared with children who were exposed to tobacco smoke and those with no smoke exposure, new research shows.
“The findings of this study are interesting and pleasantly raise further questions,” said Kristen Miller, MD, attending physician in the division of pulmonary and sleep medicine at Children’s Hospital of Philadelphia, who was not involved in the study. “Given the robust literature regarding secondhand smoke exposure and the current landscape surrounding marijuana, this is a timely study to evaluate the prevalence of marijuana use and the associated effects of marijuana exposure among children.”
Prior research has linked primary marijuana use with respiratory effects. A 2020 study associated cannabis use with an increased risk of severe bronchitis, lung hyperinflation, and increased central airway resistance. However, according to the Centers for Disease Control and Prevention, there are still a lot of unanswered questions surrounding secondhand marijuana smoke exposure and its effects.
“If kids are exposed to enough secondhand smoke, regardless of what the substance is, they’re going to have some negative health outcomes with it,” study author Adam Johnson, MD, of Wake Forest University, Winston-Salem, N.C., said in an interview.
The study, published in Pediatric Research, looked at rates of reported ED and urgent care visits and specific illnesses – such as otitis media, viral respiratory infections, and asthma exacerbations – among children with marijuana exposure and tobacco exposure.
For the study, Dr. Johnson and colleagues surveyed 1,500 parents and caregivers who went to an academic children’s hospital between Dec. 1, 2015, and July 30, 2017. Researchers found that children exposed to marijuana smoke had higher rates of ED visits at 2.21 within the past 12 months, compared with those exposed to tobacco smoke (2.14 within the past 12 months) and those with no smoke exposure (1.94 within the past 12 months). However, the difference in these visits were not statistically significant.
Researchers saw that children exposed to secondhand marijuana smoke saw a 30% increase in viral respiratory infections, compared with those who were not exposed to tobacco or marijuana smoke, Dr. Johnson said. Caregivers who smoked marijuana reported a rate of 1.31 viral infections in their children within the last year. Meanwhile those who smoked tobacco reported a rate of 1.00 infections within the last 12 months and caregivers who did not smoke reported 1.04 infections within the year.
“It suggests that components in marijuana smoke may depress the body’s immune responses to viral infections in children,” Dr. Miller said in an interview.
When it came to otitis media episodes, children exposed to marijuana had a rate of 0.96 episodes within the past 12 months. Children experiencing secondhand tobacco smoke had a rate of 0.83 episodes and those with no smoke exposure had 0.75 episodes within the past 12 months. Researchers did not note this difference as statistically significant.
When it came to asthma exacerbations, children exposed to marijuana smoke also had statistically insignificantly higher rates of exacerbations, compared with those exposed to tobacco smoke and those not exposed to smoke.
“I think it was surprising that the survey results found that marijuana seemed to be more strongly associated with the viral respiratory infections than tobacco,” Dr. Johnson said. “We know that secondhand tobacco smoke exposure in kids does lead to things like otitis media or ear infections, asthma attacks, and other processes, including colds. It was interesting that we didn’t find that association [in the new study], but we found that with marijuana.”
Dr. Johnson said the findings are especially concerning with increases in the acceptance and accessibility of marijuana as it becomes legalized in many states.
A 2015 study examined the effect of secondhand marijuana smoke exposure. Researchers found that exposure to secondhand marijuana smoke can increase heart rate, have mild to moderate sedative effects and can produce detectable cannabinoid levels in blood and urine. However, another study published in 2012 found that low to moderate primary marijuana use is less harmful to users’ lungs than tobacco exposure.
Dr. Miller added that little is known about how exposure to marijuana smoke can affect the innate responses to pathogens and there is a need to “study this in more detail” to figure out if secondhand marijuana smoke is a risk factor for either an increase in respiratory virus infections or their severity.
“These questions could have considerable implications for the health of our children and public health measures regarding marijuana use,” she explained. “As documented marijuana use increases, health care providers need to be aware of the effects of marijuana use and exposure.”
Neither Dr. Johnson nor Dr. Miller has any relevant financial disclosures.
Exposure to secondhand marijuana smoke is more strongly associated with viral respiratory infections in children, compared with children who were exposed to tobacco smoke and those with no smoke exposure, new research shows.
“The findings of this study are interesting and pleasantly raise further questions,” said Kristen Miller, MD, attending physician in the division of pulmonary and sleep medicine at Children’s Hospital of Philadelphia, who was not involved in the study. “Given the robust literature regarding secondhand smoke exposure and the current landscape surrounding marijuana, this is a timely study to evaluate the prevalence of marijuana use and the associated effects of marijuana exposure among children.”
Prior research has linked primary marijuana use with respiratory effects. A 2020 study associated cannabis use with an increased risk of severe bronchitis, lung hyperinflation, and increased central airway resistance. However, according to the Centers for Disease Control and Prevention, there are still a lot of unanswered questions surrounding secondhand marijuana smoke exposure and its effects.
“If kids are exposed to enough secondhand smoke, regardless of what the substance is, they’re going to have some negative health outcomes with it,” study author Adam Johnson, MD, of Wake Forest University, Winston-Salem, N.C., said in an interview.
The study, published in Pediatric Research, looked at rates of reported ED and urgent care visits and specific illnesses – such as otitis media, viral respiratory infections, and asthma exacerbations – among children with marijuana exposure and tobacco exposure.
For the study, Dr. Johnson and colleagues surveyed 1,500 parents and caregivers who went to an academic children’s hospital between Dec. 1, 2015, and July 30, 2017. Researchers found that children exposed to marijuana smoke had higher rates of ED visits at 2.21 within the past 12 months, compared with those exposed to tobacco smoke (2.14 within the past 12 months) and those with no smoke exposure (1.94 within the past 12 months). However, the difference in these visits were not statistically significant.
Researchers saw that children exposed to secondhand marijuana smoke saw a 30% increase in viral respiratory infections, compared with those who were not exposed to tobacco or marijuana smoke, Dr. Johnson said. Caregivers who smoked marijuana reported a rate of 1.31 viral infections in their children within the last year. Meanwhile those who smoked tobacco reported a rate of 1.00 infections within the last 12 months and caregivers who did not smoke reported 1.04 infections within the year.
“It suggests that components in marijuana smoke may depress the body’s immune responses to viral infections in children,” Dr. Miller said in an interview.
When it came to otitis media episodes, children exposed to marijuana had a rate of 0.96 episodes within the past 12 months. Children experiencing secondhand tobacco smoke had a rate of 0.83 episodes and those with no smoke exposure had 0.75 episodes within the past 12 months. Researchers did not note this difference as statistically significant.
When it came to asthma exacerbations, children exposed to marijuana smoke also had statistically insignificantly higher rates of exacerbations, compared with those exposed to tobacco smoke and those not exposed to smoke.
“I think it was surprising that the survey results found that marijuana seemed to be more strongly associated with the viral respiratory infections than tobacco,” Dr. Johnson said. “We know that secondhand tobacco smoke exposure in kids does lead to things like otitis media or ear infections, asthma attacks, and other processes, including colds. It was interesting that we didn’t find that association [in the new study], but we found that with marijuana.”
Dr. Johnson said the findings are especially concerning with increases in the acceptance and accessibility of marijuana as it becomes legalized in many states.
A 2015 study examined the effect of secondhand marijuana smoke exposure. Researchers found that exposure to secondhand marijuana smoke can increase heart rate, have mild to moderate sedative effects and can produce detectable cannabinoid levels in blood and urine. However, another study published in 2012 found that low to moderate primary marijuana use is less harmful to users’ lungs than tobacco exposure.
Dr. Miller added that little is known about how exposure to marijuana smoke can affect the innate responses to pathogens and there is a need to “study this in more detail” to figure out if secondhand marijuana smoke is a risk factor for either an increase in respiratory virus infections or their severity.
“These questions could have considerable implications for the health of our children and public health measures regarding marijuana use,” she explained. “As documented marijuana use increases, health care providers need to be aware of the effects of marijuana use and exposure.”
Neither Dr. Johnson nor Dr. Miller has any relevant financial disclosures.
Exposure to secondhand marijuana smoke is more strongly associated with viral respiratory infections in children, compared with children who were exposed to tobacco smoke and those with no smoke exposure, new research shows.
“The findings of this study are interesting and pleasantly raise further questions,” said Kristen Miller, MD, attending physician in the division of pulmonary and sleep medicine at Children’s Hospital of Philadelphia, who was not involved in the study. “Given the robust literature regarding secondhand smoke exposure and the current landscape surrounding marijuana, this is a timely study to evaluate the prevalence of marijuana use and the associated effects of marijuana exposure among children.”
Prior research has linked primary marijuana use with respiratory effects. A 2020 study associated cannabis use with an increased risk of severe bronchitis, lung hyperinflation, and increased central airway resistance. However, according to the Centers for Disease Control and Prevention, there are still a lot of unanswered questions surrounding secondhand marijuana smoke exposure and its effects.
“If kids are exposed to enough secondhand smoke, regardless of what the substance is, they’re going to have some negative health outcomes with it,” study author Adam Johnson, MD, of Wake Forest University, Winston-Salem, N.C., said in an interview.
The study, published in Pediatric Research, looked at rates of reported ED and urgent care visits and specific illnesses – such as otitis media, viral respiratory infections, and asthma exacerbations – among children with marijuana exposure and tobacco exposure.
For the study, Dr. Johnson and colleagues surveyed 1,500 parents and caregivers who went to an academic children’s hospital between Dec. 1, 2015, and July 30, 2017. Researchers found that children exposed to marijuana smoke had higher rates of ED visits at 2.21 within the past 12 months, compared with those exposed to tobacco smoke (2.14 within the past 12 months) and those with no smoke exposure (1.94 within the past 12 months). However, the difference in these visits were not statistically significant.
Researchers saw that children exposed to secondhand marijuana smoke saw a 30% increase in viral respiratory infections, compared with those who were not exposed to tobacco or marijuana smoke, Dr. Johnson said. Caregivers who smoked marijuana reported a rate of 1.31 viral infections in their children within the last year. Meanwhile those who smoked tobacco reported a rate of 1.00 infections within the last 12 months and caregivers who did not smoke reported 1.04 infections within the year.
“It suggests that components in marijuana smoke may depress the body’s immune responses to viral infections in children,” Dr. Miller said in an interview.
When it came to otitis media episodes, children exposed to marijuana had a rate of 0.96 episodes within the past 12 months. Children experiencing secondhand tobacco smoke had a rate of 0.83 episodes and those with no smoke exposure had 0.75 episodes within the past 12 months. Researchers did not note this difference as statistically significant.
When it came to asthma exacerbations, children exposed to marijuana smoke also had statistically insignificantly higher rates of exacerbations, compared with those exposed to tobacco smoke and those not exposed to smoke.
“I think it was surprising that the survey results found that marijuana seemed to be more strongly associated with the viral respiratory infections than tobacco,” Dr. Johnson said. “We know that secondhand tobacco smoke exposure in kids does lead to things like otitis media or ear infections, asthma attacks, and other processes, including colds. It was interesting that we didn’t find that association [in the new study], but we found that with marijuana.”
Dr. Johnson said the findings are especially concerning with increases in the acceptance and accessibility of marijuana as it becomes legalized in many states.
A 2015 study examined the effect of secondhand marijuana smoke exposure. Researchers found that exposure to secondhand marijuana smoke can increase heart rate, have mild to moderate sedative effects and can produce detectable cannabinoid levels in blood and urine. However, another study published in 2012 found that low to moderate primary marijuana use is less harmful to users’ lungs than tobacco exposure.
Dr. Miller added that little is known about how exposure to marijuana smoke can affect the innate responses to pathogens and there is a need to “study this in more detail” to figure out if secondhand marijuana smoke is a risk factor for either an increase in respiratory virus infections or their severity.
“These questions could have considerable implications for the health of our children and public health measures regarding marijuana use,” she explained. “As documented marijuana use increases, health care providers need to be aware of the effects of marijuana use and exposure.”
Neither Dr. Johnson nor Dr. Miller has any relevant financial disclosures.
FROM PEDIATRIC RESEARCH
CDC: Vaccination may cut risk of COVID reinfection in half
The Centers for Disease Control and Prevention has recommended that everyone get a COVID-19 vaccine, even if they’ve had the virus before. Yet many skeptics have held off getting the shots, believing that immunity generated by their previous infection will protect them if they should encounter the virus again.
A new study published in the CDC’s Morbidity and Mortality Weekly Report pokes holes in this notion. It shows people who have recovered from COVID-19 but haven’t been vaccinated have more than double the risk of testing positive for the virus again, compared with someone who was vaccinated after an initial infection.
The study looked at 738 Kentucky residents who had an initial bout of COVID-19 in 2020. About 250 of them tested positive for COVID-19 a second time between May and July of 2021, when the Delta variant became dominant in the United States.
The study matched each person who’d been reinfected with two people of the same sex and roughly the same age who had caught their initial COVID infection within the same week. The researchers then cross-matched those cases with data from Kentucky’s Immunization Registry.
They found that those who were unvaccinated had more than double the risk of being reinfected during the Delta wave. Partial vaccination appeared to have no significant impact on the risk of reinfection.
Among those who were reinfected, 20% were fully vaccinated, while 34% of those who did not get reinfected were fully vaccinated.
The study is observational, meaning it can’t show cause and effect; and the researchers had no information on the severity of the infections. Alyson Cavanaugh, PhD, a member of the CDC’s Epidemic Intelligence Service who led the study, said it is possible that some of the people who tested positive a second time had asymptomatic infections that were picked up through routine screening.
Still, the study backs up previous research and suggests that vaccination offers important additional protection.
“Our laboratory studies have shown that there’s an added benefit of vaccine for people who’ve had previous COVID-19. This is a real-world, epidemiologic study that found that among people who’d previously already had COVID-19, those who were vaccinated had lower odds of being reinfected,” Dr. Cavanaugh said.
“If you have had COVID-19 before, please still get vaccinated,” said CDC Director Rochelle Walensky, MD, in a written media statement. “This study shows you are twice as likely to get infected again if you are unvaccinated. Getting the vaccine is the best way to protect yourself and others around you, especially as the more contagious Delta variant spreads around the country.”
In a White House COVID-19 Response Team briefing in May, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease, explained why vaccines create stronger immunity than infection. He highlighted new research showing that two doses of an mRNA vaccine produce levels of neutralizing antibodies that are up to 10 times higher than the levels found in the blood of people who’ve recovered from COVID-19. Vaccines also enhance B cells and T cells in people who’ve recovered from COVID-19, which broadens the spectrum of protection and helps to fend off variants.
The study has some important limitations, which the authors acknowledged. The first is that second infections weren’t confirmed with genetic sequencing, so the researchers couldn’t definitively tell if a person tested positive a second time because they caught a new virus, or if they were somehow still shedding virus from their first infection. Given that the tests were at least 5 months apart, though, the researchers think reinfection is the most likely explanation.
Another bias in the study could have something to do with vaccination. Vaccinated people may have been less likely to be tested for COVID-19 after their vaccines, so the association or reinfection with a lack of vaccination may be overestimated.
Also, people who were vaccinated at federal sites or in another state were not logged in the state’s immunization registry, which may have skewed the data.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has recommended that everyone get a COVID-19 vaccine, even if they’ve had the virus before. Yet many skeptics have held off getting the shots, believing that immunity generated by their previous infection will protect them if they should encounter the virus again.
A new study published in the CDC’s Morbidity and Mortality Weekly Report pokes holes in this notion. It shows people who have recovered from COVID-19 but haven’t been vaccinated have more than double the risk of testing positive for the virus again, compared with someone who was vaccinated after an initial infection.
The study looked at 738 Kentucky residents who had an initial bout of COVID-19 in 2020. About 250 of them tested positive for COVID-19 a second time between May and July of 2021, when the Delta variant became dominant in the United States.
The study matched each person who’d been reinfected with two people of the same sex and roughly the same age who had caught their initial COVID infection within the same week. The researchers then cross-matched those cases with data from Kentucky’s Immunization Registry.
They found that those who were unvaccinated had more than double the risk of being reinfected during the Delta wave. Partial vaccination appeared to have no significant impact on the risk of reinfection.
Among those who were reinfected, 20% were fully vaccinated, while 34% of those who did not get reinfected were fully vaccinated.
The study is observational, meaning it can’t show cause and effect; and the researchers had no information on the severity of the infections. Alyson Cavanaugh, PhD, a member of the CDC’s Epidemic Intelligence Service who led the study, said it is possible that some of the people who tested positive a second time had asymptomatic infections that were picked up through routine screening.
Still, the study backs up previous research and suggests that vaccination offers important additional protection.
“Our laboratory studies have shown that there’s an added benefit of vaccine for people who’ve had previous COVID-19. This is a real-world, epidemiologic study that found that among people who’d previously already had COVID-19, those who were vaccinated had lower odds of being reinfected,” Dr. Cavanaugh said.
“If you have had COVID-19 before, please still get vaccinated,” said CDC Director Rochelle Walensky, MD, in a written media statement. “This study shows you are twice as likely to get infected again if you are unvaccinated. Getting the vaccine is the best way to protect yourself and others around you, especially as the more contagious Delta variant spreads around the country.”
In a White House COVID-19 Response Team briefing in May, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease, explained why vaccines create stronger immunity than infection. He highlighted new research showing that two doses of an mRNA vaccine produce levels of neutralizing antibodies that are up to 10 times higher than the levels found in the blood of people who’ve recovered from COVID-19. Vaccines also enhance B cells and T cells in people who’ve recovered from COVID-19, which broadens the spectrum of protection and helps to fend off variants.
The study has some important limitations, which the authors acknowledged. The first is that second infections weren’t confirmed with genetic sequencing, so the researchers couldn’t definitively tell if a person tested positive a second time because they caught a new virus, or if they were somehow still shedding virus from their first infection. Given that the tests were at least 5 months apart, though, the researchers think reinfection is the most likely explanation.
Another bias in the study could have something to do with vaccination. Vaccinated people may have been less likely to be tested for COVID-19 after their vaccines, so the association or reinfection with a lack of vaccination may be overestimated.
Also, people who were vaccinated at federal sites or in another state were not logged in the state’s immunization registry, which may have skewed the data.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has recommended that everyone get a COVID-19 vaccine, even if they’ve had the virus before. Yet many skeptics have held off getting the shots, believing that immunity generated by their previous infection will protect them if they should encounter the virus again.
A new study published in the CDC’s Morbidity and Mortality Weekly Report pokes holes in this notion. It shows people who have recovered from COVID-19 but haven’t been vaccinated have more than double the risk of testing positive for the virus again, compared with someone who was vaccinated after an initial infection.
The study looked at 738 Kentucky residents who had an initial bout of COVID-19 in 2020. About 250 of them tested positive for COVID-19 a second time between May and July of 2021, when the Delta variant became dominant in the United States.
The study matched each person who’d been reinfected with two people of the same sex and roughly the same age who had caught their initial COVID infection within the same week. The researchers then cross-matched those cases with data from Kentucky’s Immunization Registry.
They found that those who were unvaccinated had more than double the risk of being reinfected during the Delta wave. Partial vaccination appeared to have no significant impact on the risk of reinfection.
Among those who were reinfected, 20% were fully vaccinated, while 34% of those who did not get reinfected were fully vaccinated.
The study is observational, meaning it can’t show cause and effect; and the researchers had no information on the severity of the infections. Alyson Cavanaugh, PhD, a member of the CDC’s Epidemic Intelligence Service who led the study, said it is possible that some of the people who tested positive a second time had asymptomatic infections that were picked up through routine screening.
Still, the study backs up previous research and suggests that vaccination offers important additional protection.
“Our laboratory studies have shown that there’s an added benefit of vaccine for people who’ve had previous COVID-19. This is a real-world, epidemiologic study that found that among people who’d previously already had COVID-19, those who were vaccinated had lower odds of being reinfected,” Dr. Cavanaugh said.
“If you have had COVID-19 before, please still get vaccinated,” said CDC Director Rochelle Walensky, MD, in a written media statement. “This study shows you are twice as likely to get infected again if you are unvaccinated. Getting the vaccine is the best way to protect yourself and others around you, especially as the more contagious Delta variant spreads around the country.”
In a White House COVID-19 Response Team briefing in May, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease, explained why vaccines create stronger immunity than infection. He highlighted new research showing that two doses of an mRNA vaccine produce levels of neutralizing antibodies that are up to 10 times higher than the levels found in the blood of people who’ve recovered from COVID-19. Vaccines also enhance B cells and T cells in people who’ve recovered from COVID-19, which broadens the spectrum of protection and helps to fend off variants.
The study has some important limitations, which the authors acknowledged. The first is that second infections weren’t confirmed with genetic sequencing, so the researchers couldn’t definitively tell if a person tested positive a second time because they caught a new virus, or if they were somehow still shedding virus from their first infection. Given that the tests were at least 5 months apart, though, the researchers think reinfection is the most likely explanation.
Another bias in the study could have something to do with vaccination. Vaccinated people may have been less likely to be tested for COVID-19 after their vaccines, so the association or reinfection with a lack of vaccination may be overestimated.
Also, people who were vaccinated at federal sites or in another state were not logged in the state’s immunization registry, which may have skewed the data.
A version of this article first appeared on Medscape.com.







