User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Seeking help for burnout may be a gamble for doctors
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
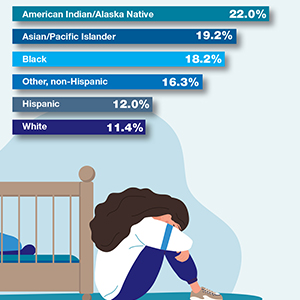
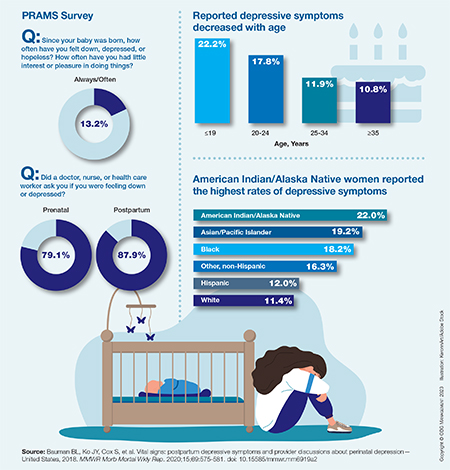
Self-reported symptoms of postpartum depression in the United States, 2018


How ob.gyn. programs provide abortion training post Dobbs
to fulfill required clinical rotations in the procedure.
The Accreditation Council for Graduate Medical Education requires ob.gyn. residents – unless they have a religious or moral exemption – to undergo abortion training to complete their programs. In states with bans or restrictions on family planning services or abortions, resident training must be received at institutions that are out of state.
Some residency programs are just beginning to coordinate out-of-state training, while others are further along in their offerings. There’s no formal matching process, and it remains unclear who will cover the costs of residents training elsewhere for a month.
These uncertainties, along with lack of coordination about malpractice, clinical rotations, and limited faculty, leave some program directors skeptical they’ll be able to keep up with demand for out-of-state slots. They are also wary of harming their own residents’ educational and clinical opportunities.
A 3rd-year ob.gyn. resident, who didn’t want to give her name or residency program for fear of backlash against her home institution, told this news organization that the Catholic-affiliated site is trying to avoid drawing attention to its minimal abortion training in a restrictive Midwest state. She knew after the Supreme Court’s decision in Dobbs v. Jackson she’d have to look outside the program for more complex abortion training.
While she could learn dilation and curettage or other first-trimester or early–second-trimester procedures at the Midwest program, she said she couldn’t learn dilation and evacuation.
A mentor at her program connected her with a residency program at the University of New Mexico, where she recently started a 5-week family planning rotation. She is the first out-of-state resident hosted by UNM. Currently, UNM has six ob.gyn. residents per class year, for a total of 36, and six family planning fellows.
The ob.gyn. resident is staying with a friend at no cost, and her home institution still pays her salary. But she still must pay the mortgage on a home she can’t live in while away and misses being part of a community where she’s built a life over the past 2 years.
“There’s a part of you that’s just angry that you can’t do this for the women ... in your state,” she said. “Unfortunately, there isn’t a formalized program for ob.gyn. residents interested in more advanced training to be matched with a program that has the ability to offer that training. It’s very much a word-of-mouth and who-you-know situation. For people without those connections, it can be difficult to obtain this training unless they are interested in a formal fellowship.”
This year, about 1,500 ob.gyn. residents matched into 280 residency programs, according to the National Residency Matching Program.
Alyssa Colwill, MD, assistant professor of obstetrics and gynecology at Oregon Health and Science University and director of the ob.gyn. Ryan Residency Program at OHSU, estimated that 1,000 ob.gyn. residents per year will seek out-of-state abortion training. The estimate is based on the number of residents in programs in states with restrictions.
The Ryan Program, which began in 1999, helps ob.gyn. residency programs provide training in abortion and contraception care (family planning) as a required rotation.
Connecting programs
Ryan-affiliated residencies have been helping connect programs in states with abortion bans and restrictions to programs in states with more liberal laws.
Twelve of the 100 Ryan programs sent residents out of state in the past academic year, and 15 will follow this year. More are expected soon, said Kristin Simonson, MA, director of programs and operations at the Ryan Residency Program, headquartered at the Bixby Center for Global Reproductive Health at the University of California, San Francisco.
Before the Dobbs decision, very few programs considered next steps to train ob.gyn. residents if abortions became illegal, Ms. Simonson said. “I think a lot of people just kind of were waiting and seeing ... and hoping that they wouldn’t have to make any drastic plans. It was hard to motivate people to have a plan B ready to go,” she said.
“Almost all of us working in this field had a really bad feeling,” said Courtney G. Forbis, MD, UNM assistant professor of ob.gyn. and Ryan Residency director. She and colleagues began planning for the future months ahead of the court decision. But the program wasn’t able to begin accepting out-of-state residents until now, she said. “We are trying to use this experience to see what we can accommodate in the future.”
OHSU also began planning for alternative training when it learned of the leaked Supreme Court decision, Dr. Colwill said. “We decided that we had the bandwidth and opportunity to train more individuals that were going to lose access to services and educational opportunities,” she said.
The university ran a 4-week test rotation last fall. So far, six residents and one fellow have come from out of the state, said Dr. Colwill. OHSU hopes to have 10 more in the coming year. The out-of-state learners will join 32 ob.gyn. residents and 12 fellows who were already in the program, she said.
To ease residents’ integration into an away program, the Ryan Program – along with the American College of Obstetricians and Gynecologists, the Council on Resident Education in Obstetrics and Gynecology, and Innovating Education in Reproductive Health – recently began offering a free, web-based patient-centered abortion education curriculum.
The course supplements in-person clinical training in abortion care and prepares residents traveling and transitioning into another program to begin learning new skills on their first day, AnnaMarie Connolly, MD, ACOG’s chief of education and academic affairs, said in a prepared statement.
Training costs
Residents and their institutions also face additional costs. The home institution that loses a resident for a few weeks to a month has to determine how to cover the care not provided while they are away, Ms. Simonson said. Residents may incur expenses for transportation, housing, food, and other things while out of state.
OHSU covers transportation and housing through its abortion care and training fund, but there are other factors to consider, Dr. Colwill said. For example, the home and host programs have to coordinate licensing, malpractice, and line up rotation dates, she said.
Among other complications, UNM wasn’t able to set up an agreement so that its new resident could participate in a rotation at Planned Parenthood. “We have the clinical volume to accommodate another learner,” Dr. Forbis said. But the program has to balance resources, such as “trying to make sure we don’t have one faculty [member] assigned to too many learners at one time,” she said.
Given the logistic and financial challenges, programs may not be able to ensure that all residents who need abortion training receive it, said Ms. Simonson.
The Ryan Program, for instance, can’t help the more than 100 residency programs in states where abortions are currently illegal, she said.
UNM is trying to partner with specific programs, such as those in the state of Texas where abortion is banned, to train its residents each year, Dr. Forbis said.
OHSU also will look for opportunities to train as many residents as possible, Dr. Colwill said, “but I don’t think we’ll ever be able to fill that gap of 1,000 residents that need this training.”
A version of this article first appeared on Medscape.com.
to fulfill required clinical rotations in the procedure.
The Accreditation Council for Graduate Medical Education requires ob.gyn. residents – unless they have a religious or moral exemption – to undergo abortion training to complete their programs. In states with bans or restrictions on family planning services or abortions, resident training must be received at institutions that are out of state.
Some residency programs are just beginning to coordinate out-of-state training, while others are further along in their offerings. There’s no formal matching process, and it remains unclear who will cover the costs of residents training elsewhere for a month.
These uncertainties, along with lack of coordination about malpractice, clinical rotations, and limited faculty, leave some program directors skeptical they’ll be able to keep up with demand for out-of-state slots. They are also wary of harming their own residents’ educational and clinical opportunities.
A 3rd-year ob.gyn. resident, who didn’t want to give her name or residency program for fear of backlash against her home institution, told this news organization that the Catholic-affiliated site is trying to avoid drawing attention to its minimal abortion training in a restrictive Midwest state. She knew after the Supreme Court’s decision in Dobbs v. Jackson she’d have to look outside the program for more complex abortion training.
While she could learn dilation and curettage or other first-trimester or early–second-trimester procedures at the Midwest program, she said she couldn’t learn dilation and evacuation.
A mentor at her program connected her with a residency program at the University of New Mexico, where she recently started a 5-week family planning rotation. She is the first out-of-state resident hosted by UNM. Currently, UNM has six ob.gyn. residents per class year, for a total of 36, and six family planning fellows.
The ob.gyn. resident is staying with a friend at no cost, and her home institution still pays her salary. But she still must pay the mortgage on a home she can’t live in while away and misses being part of a community where she’s built a life over the past 2 years.
“There’s a part of you that’s just angry that you can’t do this for the women ... in your state,” she said. “Unfortunately, there isn’t a formalized program for ob.gyn. residents interested in more advanced training to be matched with a program that has the ability to offer that training. It’s very much a word-of-mouth and who-you-know situation. For people without those connections, it can be difficult to obtain this training unless they are interested in a formal fellowship.”
This year, about 1,500 ob.gyn. residents matched into 280 residency programs, according to the National Residency Matching Program.
Alyssa Colwill, MD, assistant professor of obstetrics and gynecology at Oregon Health and Science University and director of the ob.gyn. Ryan Residency Program at OHSU, estimated that 1,000 ob.gyn. residents per year will seek out-of-state abortion training. The estimate is based on the number of residents in programs in states with restrictions.
The Ryan Program, which began in 1999, helps ob.gyn. residency programs provide training in abortion and contraception care (family planning) as a required rotation.
Connecting programs
Ryan-affiliated residencies have been helping connect programs in states with abortion bans and restrictions to programs in states with more liberal laws.
Twelve of the 100 Ryan programs sent residents out of state in the past academic year, and 15 will follow this year. More are expected soon, said Kristin Simonson, MA, director of programs and operations at the Ryan Residency Program, headquartered at the Bixby Center for Global Reproductive Health at the University of California, San Francisco.
Before the Dobbs decision, very few programs considered next steps to train ob.gyn. residents if abortions became illegal, Ms. Simonson said. “I think a lot of people just kind of were waiting and seeing ... and hoping that they wouldn’t have to make any drastic plans. It was hard to motivate people to have a plan B ready to go,” she said.
“Almost all of us working in this field had a really bad feeling,” said Courtney G. Forbis, MD, UNM assistant professor of ob.gyn. and Ryan Residency director. She and colleagues began planning for the future months ahead of the court decision. But the program wasn’t able to begin accepting out-of-state residents until now, she said. “We are trying to use this experience to see what we can accommodate in the future.”
OHSU also began planning for alternative training when it learned of the leaked Supreme Court decision, Dr. Colwill said. “We decided that we had the bandwidth and opportunity to train more individuals that were going to lose access to services and educational opportunities,” she said.
The university ran a 4-week test rotation last fall. So far, six residents and one fellow have come from out of the state, said Dr. Colwill. OHSU hopes to have 10 more in the coming year. The out-of-state learners will join 32 ob.gyn. residents and 12 fellows who were already in the program, she said.
To ease residents’ integration into an away program, the Ryan Program – along with the American College of Obstetricians and Gynecologists, the Council on Resident Education in Obstetrics and Gynecology, and Innovating Education in Reproductive Health – recently began offering a free, web-based patient-centered abortion education curriculum.
The course supplements in-person clinical training in abortion care and prepares residents traveling and transitioning into another program to begin learning new skills on their first day, AnnaMarie Connolly, MD, ACOG’s chief of education and academic affairs, said in a prepared statement.
Training costs
Residents and their institutions also face additional costs. The home institution that loses a resident for a few weeks to a month has to determine how to cover the care not provided while they are away, Ms. Simonson said. Residents may incur expenses for transportation, housing, food, and other things while out of state.
OHSU covers transportation and housing through its abortion care and training fund, but there are other factors to consider, Dr. Colwill said. For example, the home and host programs have to coordinate licensing, malpractice, and line up rotation dates, she said.
Among other complications, UNM wasn’t able to set up an agreement so that its new resident could participate in a rotation at Planned Parenthood. “We have the clinical volume to accommodate another learner,” Dr. Forbis said. But the program has to balance resources, such as “trying to make sure we don’t have one faculty [member] assigned to too many learners at one time,” she said.
Given the logistic and financial challenges, programs may not be able to ensure that all residents who need abortion training receive it, said Ms. Simonson.
The Ryan Program, for instance, can’t help the more than 100 residency programs in states where abortions are currently illegal, she said.
UNM is trying to partner with specific programs, such as those in the state of Texas where abortion is banned, to train its residents each year, Dr. Forbis said.
OHSU also will look for opportunities to train as many residents as possible, Dr. Colwill said, “but I don’t think we’ll ever be able to fill that gap of 1,000 residents that need this training.”
A version of this article first appeared on Medscape.com.
to fulfill required clinical rotations in the procedure.
The Accreditation Council for Graduate Medical Education requires ob.gyn. residents – unless they have a religious or moral exemption – to undergo abortion training to complete their programs. In states with bans or restrictions on family planning services or abortions, resident training must be received at institutions that are out of state.
Some residency programs are just beginning to coordinate out-of-state training, while others are further along in their offerings. There’s no formal matching process, and it remains unclear who will cover the costs of residents training elsewhere for a month.
These uncertainties, along with lack of coordination about malpractice, clinical rotations, and limited faculty, leave some program directors skeptical they’ll be able to keep up with demand for out-of-state slots. They are also wary of harming their own residents’ educational and clinical opportunities.
A 3rd-year ob.gyn. resident, who didn’t want to give her name or residency program for fear of backlash against her home institution, told this news organization that the Catholic-affiliated site is trying to avoid drawing attention to its minimal abortion training in a restrictive Midwest state. She knew after the Supreme Court’s decision in Dobbs v. Jackson she’d have to look outside the program for more complex abortion training.
While she could learn dilation and curettage or other first-trimester or early–second-trimester procedures at the Midwest program, she said she couldn’t learn dilation and evacuation.
A mentor at her program connected her with a residency program at the University of New Mexico, where she recently started a 5-week family planning rotation. She is the first out-of-state resident hosted by UNM. Currently, UNM has six ob.gyn. residents per class year, for a total of 36, and six family planning fellows.
The ob.gyn. resident is staying with a friend at no cost, and her home institution still pays her salary. But she still must pay the mortgage on a home she can’t live in while away and misses being part of a community where she’s built a life over the past 2 years.
“There’s a part of you that’s just angry that you can’t do this for the women ... in your state,” she said. “Unfortunately, there isn’t a formalized program for ob.gyn. residents interested in more advanced training to be matched with a program that has the ability to offer that training. It’s very much a word-of-mouth and who-you-know situation. For people without those connections, it can be difficult to obtain this training unless they are interested in a formal fellowship.”
This year, about 1,500 ob.gyn. residents matched into 280 residency programs, according to the National Residency Matching Program.
Alyssa Colwill, MD, assistant professor of obstetrics and gynecology at Oregon Health and Science University and director of the ob.gyn. Ryan Residency Program at OHSU, estimated that 1,000 ob.gyn. residents per year will seek out-of-state abortion training. The estimate is based on the number of residents in programs in states with restrictions.
The Ryan Program, which began in 1999, helps ob.gyn. residency programs provide training in abortion and contraception care (family planning) as a required rotation.
Connecting programs
Ryan-affiliated residencies have been helping connect programs in states with abortion bans and restrictions to programs in states with more liberal laws.
Twelve of the 100 Ryan programs sent residents out of state in the past academic year, and 15 will follow this year. More are expected soon, said Kristin Simonson, MA, director of programs and operations at the Ryan Residency Program, headquartered at the Bixby Center for Global Reproductive Health at the University of California, San Francisco.
Before the Dobbs decision, very few programs considered next steps to train ob.gyn. residents if abortions became illegal, Ms. Simonson said. “I think a lot of people just kind of were waiting and seeing ... and hoping that they wouldn’t have to make any drastic plans. It was hard to motivate people to have a plan B ready to go,” she said.
“Almost all of us working in this field had a really bad feeling,” said Courtney G. Forbis, MD, UNM assistant professor of ob.gyn. and Ryan Residency director. She and colleagues began planning for the future months ahead of the court decision. But the program wasn’t able to begin accepting out-of-state residents until now, she said. “We are trying to use this experience to see what we can accommodate in the future.”
OHSU also began planning for alternative training when it learned of the leaked Supreme Court decision, Dr. Colwill said. “We decided that we had the bandwidth and opportunity to train more individuals that were going to lose access to services and educational opportunities,” she said.
The university ran a 4-week test rotation last fall. So far, six residents and one fellow have come from out of the state, said Dr. Colwill. OHSU hopes to have 10 more in the coming year. The out-of-state learners will join 32 ob.gyn. residents and 12 fellows who were already in the program, she said.
To ease residents’ integration into an away program, the Ryan Program – along with the American College of Obstetricians and Gynecologists, the Council on Resident Education in Obstetrics and Gynecology, and Innovating Education in Reproductive Health – recently began offering a free, web-based patient-centered abortion education curriculum.
The course supplements in-person clinical training in abortion care and prepares residents traveling and transitioning into another program to begin learning new skills on their first day, AnnaMarie Connolly, MD, ACOG’s chief of education and academic affairs, said in a prepared statement.
Training costs
Residents and their institutions also face additional costs. The home institution that loses a resident for a few weeks to a month has to determine how to cover the care not provided while they are away, Ms. Simonson said. Residents may incur expenses for transportation, housing, food, and other things while out of state.
OHSU covers transportation and housing through its abortion care and training fund, but there are other factors to consider, Dr. Colwill said. For example, the home and host programs have to coordinate licensing, malpractice, and line up rotation dates, she said.
Among other complications, UNM wasn’t able to set up an agreement so that its new resident could participate in a rotation at Planned Parenthood. “We have the clinical volume to accommodate another learner,” Dr. Forbis said. But the program has to balance resources, such as “trying to make sure we don’t have one faculty [member] assigned to too many learners at one time,” she said.
Given the logistic and financial challenges, programs may not be able to ensure that all residents who need abortion training receive it, said Ms. Simonson.
The Ryan Program, for instance, can’t help the more than 100 residency programs in states where abortions are currently illegal, she said.
UNM is trying to partner with specific programs, such as those in the state of Texas where abortion is banned, to train its residents each year, Dr. Forbis said.
OHSU also will look for opportunities to train as many residents as possible, Dr. Colwill said, “but I don’t think we’ll ever be able to fill that gap of 1,000 residents that need this training.”
A version of this article first appeared on Medscape.com.
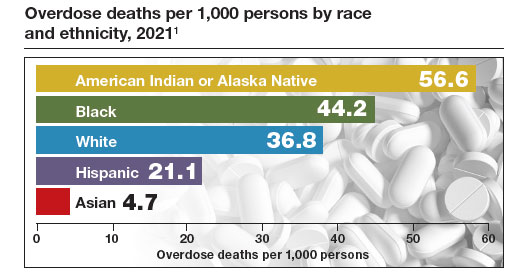
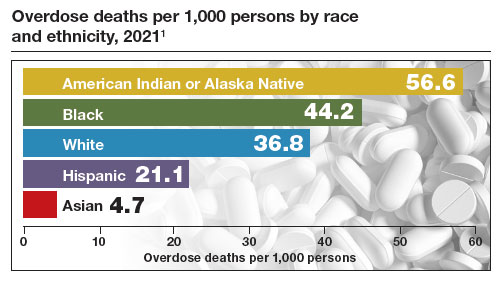
Medication treatment of opioid use disorder in primary care practice: Opportunities and limitations
The Centers for Disease Control and Prevention (CDC) reported 106,699 deaths in 2021 from drug overdose, with the majority being linked to synthetic opioids, including fentanyl and tramadol.1 This number compares with 42,795 deaths due to motor vehicle accidents and 48,183 deaths due to suicide in 2021.2,3 Most of the opioid overdose deaths occurred among people aged 25 to 64 years, the peak age of patients cared for by obstetrician-gynecologists. Among pregnant and postpartum persons, mortality due to drug overdose has increased by 81% between 2017 and 2020.4
Among pregnant and postpartum patients, drug overdose death is more common than suicide, and the risk for drug overdose death appears to be greatest in the year following delivery.5,6 In many cases, postpartum patients with OUD have had multiple contacts with the health care system prior to their death, showing that there is an opportunity for therapeutic intervention before the death occurred.7 Medication-assisted recovery for OUD involves a comprehensive array of interventions including medication, counseling, and social support. Medication treatment of OUD with BUP or methadone reduces the risk for death but is underutilized among patients with OUD.6,8 Recent federal legislation has removed restrictions on the use of BUP, increasing the opportunity for primary care clinicians to prescribe it for the treatment of OUD.9
Screening and diagnosis of OUD
Screening for OUD is recommended for patients who are at risk for opioid misuse (ie, those who are taking/have taken opioid medications). The OWLS (Overuse, Worrying, Losing interest, and feeling Slowed down, sluggish, or sedated) screening tool is used to detect prescription medication OUD and has 4 questions10:
1. In the past 3 months did you use your opioid medicines for other purposes—for example, to help you sleep or to help with stress or worry?
2. In the past 3 months did opioid medicines cause you to feel slowed down, sluggish, or sedated?
3. In the past 3 months did opioid medicines cause you to lose interest in your usual activities?
4. In the past 3 months did you worry about your use of opioid medicines?
Patient agreement with 3 or 4 questions indicates a positive screening test.
If the patient has a positive screening test, a formal diagnosis of OUD can be made using the 11 symptoms outlined in the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.11 The diagnosis of mild (2 to 3 symptoms), moderate (4 to 5 symptoms), or severe OUD (6 or more symptoms) is made based on the number of symptoms the patient reports.
Buprenorphine treatment of OUD in primary care
The role of primary care clinicians in the medication treatment of OUD is increasing. Using a nationwide system that tracks prescription medications, investigators reported that, in 2004, psychiatrists wrote 32.2% of all BUP prescriptions; in 2021, however, only 10% of such prescriptions were provided by psychiatrists, with most prescriptions written by non-psychiatrist physicians, nurse practitioners, and physician assistants that year.12 Innovative telehealth approaches to consultation and medication treatment of OUD are now available—one example is QuickMD.13 Such sites are designed to remove barriers to initiating medication treatment of OUD.
The role of primary care clinicians in the management of OUD using BUP and buprenorphine-naloxone (BUP-NAL) has increased due to many factors, including:
- the removal of US Food and Drug Administration (FDA) barriers to prescribing BUP
- the epidemic of OUD and the small size of the addiction specialist workforce, necessitating that primary care clinicians become engaged in the treatment of OUD
- an increase in unobserved initiation of BUP among ambulatory patients, and a parallel decrease in cases of observed initiation in addiction center settings
- the reframing of OUD as a chronic medical problem, with many similarities to diabetes, obesity, dyslipidemia, and hypertension.
Similar to other diseases managed by primary care clinicians, OUD requires long-term chronic treatment with a medicine that, if taken as directed, provides excellent outcomes. Primary care clinicians who prescribe BUP also can optimize longitudinal care for comorbid disorders such as hypertension and diabetes, which are prevalent in people with OUD.
In 2019, New Jersey implemented new guidelines for the treatment of OUD, removing prior authorization barriers, increasing reimbursement for office-based OUD treatment, and establishing regional centers of excellence. The implementation of the new guidelines was followed by a marked increase in BUP prescribers among primary care clinicians, emergency medicine physicians, and advanced practice clinicians.14
To estimate the public health impact of BUP prescribing by primary care clinicians, investigators simulated patient outcomes in 3 scenarios15:
1. primary care clinicians refer patients to addiction specialists for OUD treatment
2. primary care clinicians provide BUP services in their practice
3. primary care clinicians provide BUP and harm reduction kits containing syringes and wound care supplies in their practice.
Strategies 2 and 3 resulted in 14% fewer deaths due to opioid overdose, an increased life expectancy of approximately 2.7 years, and reduced hospital costs. For strategy 3, the incremental cost per life-year saved was $34,400. The investigators noted that prescribing BUP in primary care practice increases practice costs.15
Treatment with BUP reduces death from opioid overdose, improves patient health, decreases use of illicit opioids, and reduces patient cravings for opioids. BUP is a safe medication and is associated with fewer adverse effects than insulin or warfarin.16
Continue to: Methadone treatment of OUD...
Methadone treatment of OUD
Methadone is a full opioid agonist approved by the FDA for the treatment of severe pain or OUD. Methadone treatment of OUD is strictly regulated and typically is ordered and administered at an opioid treatment program that is federally licensed. Methadone for OUD treatment cannot be prescribed by a physician to a pharmacy, limiting its use in primary care practice. Methadone used to treat OUD is ordered and dispensed at opioid-treatment programs. Take-home doses of methadone may be available to patients after adherence to the regimen has been established. When used long-term, higher doses of methadone are associated with better adherence, but these higher doses can cause respiratory depression. In a study of 189 pregnant patients taking methadone to treat OUD, daily doses of 60 mg or greater were associated with better treatment retention at delivery and 60 days postpartum, as well as less use of nonprescription opioids.17 Under limited circumstances methadone can be ordered and dispensed for hospitalized patients with OUD.
Methadone is a pure opioid receptor agonist. Naloxone (NAL) is an opioid receptor antagonist. Buprenorphine (BUP) is a partial opioid receptor agonist-antagonist, which limits overdose risk. BUP often is combined with NAL as a combination formulation, which is thought to reduce the repurposing of BUP for non-prescribed uses. At appropriate treatment dosages, both methadone (≥60 mg) and BUP (≥ 16 mg) are highly effective for the treatment of OUD.1 For patients with health insurance, pharmacy benefits often provide some coverage for preferred products but no coverage for other products. Not all pharmacies carry BUP products. In a study of more than 5,000 pharmacies, approximately 60% reported that they carry and can dispense BUP medications.2
BUP monotherapy is available as generic sublingual tablets, buccal films (Belbuca), formulations for injection (Sublocade), and subcutaneous implants (Probuphine). BUPNAL is available as buccal films (Bunavail), sublingual films (Suboxone), and sublingual tablets (Zubsolv). For BUP-NAL combination productions, the following dose combinations have been reported to have similar effects: BUP-NAL 8 mg/2 mg sublingual film, BUP-NAL 5.7 mg/1.4 mg sublingual tablet, and BUP-NAL 4.2 mg/0.7 mg buccal film.3
When initiating BUP-monotherapy or BUP-NAL treatment for OUD, one approach for unobserved initiation is to instruct the patient to discontinue using opioid agonist drugs and wait for the onset of mild to moderate withdrawal symptoms. The purpose of this step is to avoid precipitating severe withdrawal symptoms caused by giving BUP or BUP-NAL to a patient who has recently used opioid drugs.
If BUP-NAL sublingual films (Suboxone) are prescribed following the onset of mild to moderate withdrawal symptoms, the patient can initiate therapy with a dose of 2 mg BUP/0.5 mg NAL or 4 mg BUP/1 mg NAL. At 60 to 120 minutes following the initial dose, if withdrawal symptoms persist, an additional dose of 4 mg BUP/1 mg NAL can be given. Thereafter, symptoms can be assessed every 60 to 120 minutes and additional doses administered to control symptoms. On the second day of therapy, a maximum of 16 mg of BUP is administered. Over the following days and weeks, if symptoms and cravings persist at a BUP dose of 16 mg, the total daily dose of BUP can be titrated up to 24 mg. For long-term treatment, a commonly prescribed daily dose is 16 mg BUP/4 mg NAL or 24 mg BUP/6 mg NAL. An absolute contraindication to BUP or BUP/NAL treatment is an allergy to the medication, and a relative contraindication is liver failure.
One potential complication of transmucosal BUP or BUP-NAL treatment is a dry mouth (xerostomia), which may contribute to dental disease.4 However, some experts question the quality of the data that contributed to the warning.5,6 Potential dental complications might be prevented by regular oral health examinations, daily flossing and teeth brushing, and stimulation of saliva by sugar-free gum or lozenges.
Primary care clinicians who initiate BUP or BUPNAL treatment for OUD often have a weekly visit with the patient during the initial phase of treatment and then every 3 to 4 weeks during maintenance therapy. Most patients need long-term treatment to achieve the goals of therapy, which include prevention of opioid overdose, reduction of cravings for nonprescription narcotics, and improvement in overall health. BUP and BUP-NAL treatment are effective without formal counseling, but counseling and social work support improve long-term adherence with treatment. Primary care clinicians who have experience with medication treatment of OUD report that their experience convinces them that medication treatment of OUD has similarities to the long-term treatment of diabetes, with antihyperglycemia medicines or the treatment of HIV infection with antiviral medications.
References
1. Mattick RP, Breen C, Kimber J, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014;CD002207.
2. Weiner SG, Qato DM, Faust JS, et al. Pharmacy availability of buprenorphine for opioid use disorder treatment in the U.S. JAMA Netw Open. 2023;6:E2316089.
3. Substance Abuse and Mental Health Services Administration (SAMHSA). Medications for opioid use disorder. SAMHSA website. Accessed August 21, 2023. https ://store.samhsa.gov/sites/default/files/SAMHSA_Digital_Download/PEP 21-02-01-002.pdf
4. FDA warns about dental problems with buprenorphine medicines dissolved in the mouth. FDA website. Accessed August 21, 2023. https ://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-about-dental-problems-buprenorphine-medicines-dissolved-mouth-treat-opioiduse-disorder#:~:text=What%20did%20FDA%20find%3F,medicines%20 dissolved%20in%20the%20mouth
5. Watson DP, Etmian S, Gastala N. Sublingual buprenorphine-naloxone exposure and dental disease. JAMA. 2023;329:1223-1224.
6. Brothers TD, Lewer D, Bonn M. Sublingual buprenorphine-naloxone exposure and dental disease. JAMA. 2023;329:1224.
Medication treatment of OUD in obstetrics
In the United States, the prevalence of OUD among pregnant patients hospitalized for delivery more than quadrupled from 1999 through 2014.18 BUP and methadone commonly are used to treat OUD during pregnancy.19 Among pregnant patients about 5% of buprenorphine prescriptions are written by obstetricians.20 An innovative approach to initiating BUP for pregnant patients with OUD is to use unobserved initiation, which involves outpatient discontinuation of nonprescription opioids to induce mild to moderate withdrawal symptoms followed by initiation of BUP treatment. In one cohort study, 55 pregnant patients used an unobserved outpatient protocol to initiate BUP treatment; 80% of the patients previously had used methadone or BUP. No patient experienced a precipitated withdrawal and 96% of patients returned for their office visit 1 week after initiation of treatment. Eighty-six percent of patients remained in treatment 3 months following initiation of BUP.21
Compared with methadone, BUP treatment during pregnancy may result in lower rates of neonatal abstinence syndrome. In one study of pregnant patients who were using methadone (n = 5,056) or BUP (n = 11,272) in late pregnancy, neonatal abstinence syndrome was diagnosed in 69.2% and 52.0% of newborns, respectively (adjusted relative risk, 0.73; 95% confidence interval, 0.71–0.75).22 In addition, compared with methadone, the use of BUP was associated with a reduced risk for low birth weight (14.9% vs 8.3%) and a lower risk for preterm birth (24.9% vs 14.4%). In this study, there were no differences in maternal obstetric outcomes when comparing BUP versus methadone treatment. Similar results have been reported in a meta-analysis analyzing the use of methadone and BUP during pregnancy.23 Studies performed to date have not shown an increased risk of congenital anomalies with the use of BUP-NAL during pregnancy.24,25
Although there may be differences in newborn outcomes with BUP and methadone, the American College of Obstetricians and Gynecologists does not recommend switching from methadone to BUP during pregnancy because precipitated withdrawal may occur.26 Based on recent studies, the American Society of Addiction Medicine has advised that it is safe to prescribe pregnant patients either BUP or BUP-NAL.27,28
Medication treatment of OUD with or without intensive counseling
The FDA recently reviewed literature related to the advantages and challenges of combining intensive counseling with medication treatment of OUD.29 The FDA noted that treatment saves lives and encouraged clinicians to initiate medication treatment of OUD or refer the patient to an appropriate clinician or treatment center. Combining medication treatment of OUD with intensive counseling is associated with greater treatment adherence and reduced health care costs. For example, in one study of 4,987 patients with OUD, initiation of counseling within 8 weeks of the start of medication treatment and a BUP dose of 16 mg or greater daily were associated with increased adherence to treatment.30 For patients receiving a BUP dose of less than 16 mg daily, treatment adherence with and without counseling was approximately 325 and 230 days, respectively. When the dose of BUP was 16 mg or greater, treatment adherence with and without counseling was approximately 405 and 320 days, respectively.30
Counseling should always be offered to patients initiating medication treatment of OUD. It should be noted that counseling alone is not a highly effective treatment for OUD.31 The FDA recently advised that the lack of availability of intensive counseling should not prevent clinicians from initiating BUP for the treatment of OUD.29 OUD is associated with a high mortalityrate and if counseling is not possible, medication treatment should be initiated. Substantial evidence demonstrates that medication treatment of OUD is associated with many benefits.16 The FDA advisory committee concluded that OUD treatment decisions should use shared decision making and be supportive and patient centered.29
The opportunities for medication treatment of OUD in primary care practice have expanded due to the recent FDA removal of restrictions on the use of BUP and heightened awareness of the positive public health impact of medication treatment. Challenges to the medication treatment of OUD remain, including stigmatization of OUD, barriers to insurance coverage for BUP, practice costs of treating OUD, and gaps in clinical education. For many pregnant patients, their main point of contact with health care is their obstetrician. By incorporating OUD treatment in pregnancy care, obstetricians will improve the health of the mother and newborn, contributing to the well-being of current and future generations. ●
Experts have recommended several interventions that may help reduce opioid overdose death.1 A consensus recommendation is that people who use drugs should be provided naloxone rescue medication and educated on the proper use of naloxone. Naloxone rescue medication is available in formulations for nasal or parenteral administration. The US Food and Drug Administration (FDA) recently has approved naloxone for over-the-counter status. The American Medical Association has provided a short web video on how to administer nasal naloxone.2 In a small pilot study, obstetricians offered every postpartum patient with naloxone administration education and a 2-dose nasal naloxone pack, with 76% of patients accepting the nasal naloxone pack.3
Many experts recommend that people who use drugs should be advised to never use them alone and to test a small amount of the drug to assess its potency. Many patients who use opioid drugs also take benzodiazepines, which can contribute to respiratory depression.4 Patients should avoid mixing drugs (eg, opioids and benzodiazepines). Some experts recommend that patients who use drugs should be provided take-home fentanyl test strips so they can evaluate their drugs for the presence of fentanyl, a medication that suppresses respiration and contributes to many overdose deaths. In addition, people who use drugs and are interested in reducing their use of drugs or managing overdose risk can be offered initiation of medication treatment of OUD.1
References
1. Wood E, Solomon ED, Hadland SE. Universal precautions for people at risk of opioid overdose in North America. JAMA Int Med. 2023;183:401-402.
2. How to administer Naloxone. AMA website. Accessed August 28, 2023. https://www.ama-assn.org /delivering-care/overdose-epidemic/how-administer-naloxone
3. Naliboff JA, Tharpe N. Universal postpartum naloxone provision: a harm reduction quality improvement project. J Addict Med. 2022;17:360-362.
4. Kelly JC, Raghuraman N, Stout MJ, et al. Home induction of buprenorphine for treatment of opioid use disorder in pregnancy. Obstet Gynecol. 2021;138:655-659.
- Spencer MR, Miniño AM, Warner M. Drug overdose deaths in the United States, 20012021. NCHS Data Brief no 457. Hyattsville, MD, National Center for Health Statistics. 2022. NCHS Data Brief No. 457. Published December 2022. Accessed August 21, 2023. https://www.cdc.gov /nchs/products/databriefs/db457.htm
- US traffic deaths drop slightly in 2022 but still a ‘crisis.’ AP News website. Published April 20, 2023. Accessed August 21, 2023. https://apnews.com /article/traffic-deaths-distracted-driving-crisis -6db6471e273b275920b6c4f9eb7e493b
- Suicide statistics. American Foundation for Suicide Prevention website. Accessed August 21, 2023. https://afsp.org/suicide-statistics/
- Bruzelius E, Martins SS. US Trends in drug overdose mortality among pregnant and postpartum persons, 2017-2020. JAMA. 2022;328:2159-2161.
- Metz TD, Rovner P, Hoffman MC, et al. Maternal deaths from suicide and overdose in Colorado, 2004-2012. Obstet Gynecol. 2016;128:1233-1240.
- Schiff DM, Nielsen T, Terplan M, et al. Fatal and nonfatal overdose among pregnant and postpartum women in Massachusetts. Obstet Gynecol. 2018;132:466-474.
- Goldman-Mellor S, Margerison CE. Maternal drug-related death and suicide are leading causes of postpartum death in California. Am J Obstet Gynecol. 2019;221:489.e1-489.e9.
- Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550.
- Waiver elimination (MAT Act). SAMHSA website. Accessed August 21, 2023. https://www .samhsa.gov/medications-substance-use- disorders/removal-data-waiver-requirement
- Picco L, Middleton M, Bruno R, et al. Validation of the OWLS, a Screening Tool for Measuring Prescription Opioid Use Disorder in Primary Care. Pain Med. 2020;21:2757-2764.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013.
- Creedon TB, Ali MM, Schuman-Olivier Z. Trends in buprenorphine prescribing for opioid use disorder by psychiatrists in the US from 2003 to 2021. JAMA Health Forum. 2023;4:E230221.
- Quick MD website. Accessed August 21, 2023. https://quick.md/
- Treitler P, Nowels M, Samples H, et al. BUP utilization and prescribing among New Jersey Medicaid beneficiaries after adoption of initiatives designed to improve treatment access. JAMA Netw Open. 2023;6:E2312030.
- Jawa R, Tin Y, Nall S, et al. Estimated clinical outcomes and cost-effectiveness associated with provision of addiction treatment in US primary care clinics. JAMA Netw Open. 2023;6:E237888.
- Wakeman SE, Larochelle MR, Ameli O, et al. Comparative effectiveness of different treatment pathways of opioid use disorder. JAMA Netw Open. 2020;3:E1920622.
- Wilder CM, Hosta D, Winhusen T. Association of methadone dose with substance use and treatment retention in pregnant and postpartum women with opioid use disorder. J Subst Abuse Treat. 2017;80:33-36.
- Haight SC, Ko JY, Tong VT, et al. Opioid use disorder documented at delivery hospitalization - United States, 1999-2014. MMWR Morb Mortal Wkly Rep. 2018;67:845-849.
- Xu KY, Jones HE, Schiff DM, et al. Initiation and treatment discontinuation of medications for opioid use disorder in pregnant people compared with nonpregnant people. Obstet Gynecol. 2023;141:845-853.
- Kelly D, Krans EE. Medical specialty of buprenorphine prescribers for pregnant women with opioid use disorder. Am J Obstet Gynecol. 2019;220:502-503.
- Kelly JC, Raghuraman N, Stout MJ, et al. Home induction of buprenorphine for treatment of opioid use disorder in pregnancy. Obstet Gynecol. 2021;138:655-659.
- Suarez EA, Huybrechts KF, Straub L, et al. Buprenorphine versus methadone for opioid use disorder in pregnancy. N Engl J Med. 2022;387:2033-2044.
- Kinsella M, Halliday LO, Shaw M, et al. Buprenorphine compared with methadone in pregnancy: a systematic review and meta-analysis. Subst Use Misuse. 2022;57:1400-1416.
- Jumah NA, Edwards C, Balfour-Boehm J, et al. Observational study of the safety of buprenorphine-naloxone in pregnancy in a rural and remote population. BMJ Open. 2016;6:E011774.
- Mullins N, Galvin SL, Ramage M, et al. Buprenorphine and naloxone versus buprenorphine for opioid use disorder in pregnancy: a cohort study. J Addict Med. 2020;14:185-192.
- Opioid use and opioid use disorder in pregnancy. Committee Opinion No. 711. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2017;130:E81-E94.
- The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder: 2020 Focused Update. J Addict Med. 2020;14(2S suppl 1):1-91.
- Link HM, Jones H, Miller L, et al. Buprenorphinenaloxone use in pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol MFM. 2020;2:100179.
- Delphin-Rittmon ME, Cavazzoni P. US Food and Drug Administration website. https://www.fda .gov/media/168027/download
- Eren K, Schuster J, Herschell A, et al. Association of Counseling and Psychotherapy on retention in medication for addiction treatment within a large Medicaid population. J Addict Med. 2022;16:346353.
- Kakko J, Dybrandt Svanborg K, Kreek MJ, et al. 1-year retention and social function after buprenorphine-assisted relapse prevention treatment for heroin dependence in Sweden: a randomized, placebo-controlled trial. Lancet. 2003;361:662-668.
The Centers for Disease Control and Prevention (CDC) reported 106,699 deaths in 2021 from drug overdose, with the majority being linked to synthetic opioids, including fentanyl and tramadol.1 This number compares with 42,795 deaths due to motor vehicle accidents and 48,183 deaths due to suicide in 2021.2,3 Most of the opioid overdose deaths occurred among people aged 25 to 64 years, the peak age of patients cared for by obstetrician-gynecologists. Among pregnant and postpartum persons, mortality due to drug overdose has increased by 81% between 2017 and 2020.4
Among pregnant and postpartum patients, drug overdose death is more common than suicide, and the risk for drug overdose death appears to be greatest in the year following delivery.5,6 In many cases, postpartum patients with OUD have had multiple contacts with the health care system prior to their death, showing that there is an opportunity for therapeutic intervention before the death occurred.7 Medication-assisted recovery for OUD involves a comprehensive array of interventions including medication, counseling, and social support. Medication treatment of OUD with BUP or methadone reduces the risk for death but is underutilized among patients with OUD.6,8 Recent federal legislation has removed restrictions on the use of BUP, increasing the opportunity for primary care clinicians to prescribe it for the treatment of OUD.9
Screening and diagnosis of OUD
Screening for OUD is recommended for patients who are at risk for opioid misuse (ie, those who are taking/have taken opioid medications). The OWLS (Overuse, Worrying, Losing interest, and feeling Slowed down, sluggish, or sedated) screening tool is used to detect prescription medication OUD and has 4 questions10:
1. In the past 3 months did you use your opioid medicines for other purposes—for example, to help you sleep or to help with stress or worry?
2. In the past 3 months did opioid medicines cause you to feel slowed down, sluggish, or sedated?
3. In the past 3 months did opioid medicines cause you to lose interest in your usual activities?
4. In the past 3 months did you worry about your use of opioid medicines?
Patient agreement with 3 or 4 questions indicates a positive screening test.
If the patient has a positive screening test, a formal diagnosis of OUD can be made using the 11 symptoms outlined in the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.11 The diagnosis of mild (2 to 3 symptoms), moderate (4 to 5 symptoms), or severe OUD (6 or more symptoms) is made based on the number of symptoms the patient reports.

Buprenorphine treatment of OUD in primary care
The role of primary care clinicians in the medication treatment of OUD is increasing. Using a nationwide system that tracks prescription medications, investigators reported that, in 2004, psychiatrists wrote 32.2% of all BUP prescriptions; in 2021, however, only 10% of such prescriptions were provided by psychiatrists, with most prescriptions written by non-psychiatrist physicians, nurse practitioners, and physician assistants that year.12 Innovative telehealth approaches to consultation and medication treatment of OUD are now available—one example is QuickMD.13 Such sites are designed to remove barriers to initiating medication treatment of OUD.
The role of primary care clinicians in the management of OUD using BUP and buprenorphine-naloxone (BUP-NAL) has increased due to many factors, including:
- the removal of US Food and Drug Administration (FDA) barriers to prescribing BUP
- the epidemic of OUD and the small size of the addiction specialist workforce, necessitating that primary care clinicians become engaged in the treatment of OUD
- an increase in unobserved initiation of BUP among ambulatory patients, and a parallel decrease in cases of observed initiation in addiction center settings
- the reframing of OUD as a chronic medical problem, with many similarities to diabetes, obesity, dyslipidemia, and hypertension.
Similar to other diseases managed by primary care clinicians, OUD requires long-term chronic treatment with a medicine that, if taken as directed, provides excellent outcomes. Primary care clinicians who prescribe BUP also can optimize longitudinal care for comorbid disorders such as hypertension and diabetes, which are prevalent in people with OUD.
In 2019, New Jersey implemented new guidelines for the treatment of OUD, removing prior authorization barriers, increasing reimbursement for office-based OUD treatment, and establishing regional centers of excellence. The implementation of the new guidelines was followed by a marked increase in BUP prescribers among primary care clinicians, emergency medicine physicians, and advanced practice clinicians.14
To estimate the public health impact of BUP prescribing by primary care clinicians, investigators simulated patient outcomes in 3 scenarios15:
1. primary care clinicians refer patients to addiction specialists for OUD treatment
2. primary care clinicians provide BUP services in their practice
3. primary care clinicians provide BUP and harm reduction kits containing syringes and wound care supplies in their practice.
Strategies 2 and 3 resulted in 14% fewer deaths due to opioid overdose, an increased life expectancy of approximately 2.7 years, and reduced hospital costs. For strategy 3, the incremental cost per life-year saved was $34,400. The investigators noted that prescribing BUP in primary care practice increases practice costs.15
Treatment with BUP reduces death from opioid overdose, improves patient health, decreases use of illicit opioids, and reduces patient cravings for opioids. BUP is a safe medication and is associated with fewer adverse effects than insulin or warfarin.16
Continue to: Methadone treatment of OUD...
Methadone treatment of OUD
Methadone is a full opioid agonist approved by the FDA for the treatment of severe pain or OUD. Methadone treatment of OUD is strictly regulated and typically is ordered and administered at an opioid treatment program that is federally licensed. Methadone for OUD treatment cannot be prescribed by a physician to a pharmacy, limiting its use in primary care practice. Methadone used to treat OUD is ordered and dispensed at opioid-treatment programs. Take-home doses of methadone may be available to patients after adherence to the regimen has been established. When used long-term, higher doses of methadone are associated with better adherence, but these higher doses can cause respiratory depression. In a study of 189 pregnant patients taking methadone to treat OUD, daily doses of 60 mg or greater were associated with better treatment retention at delivery and 60 days postpartum, as well as less use of nonprescription opioids.17 Under limited circumstances methadone can be ordered and dispensed for hospitalized patients with OUD.
Methadone is a pure opioid receptor agonist. Naloxone (NAL) is an opioid receptor antagonist. Buprenorphine (BUP) is a partial opioid receptor agonist-antagonist, which limits overdose risk. BUP often is combined with NAL as a combination formulation, which is thought to reduce the repurposing of BUP for non-prescribed uses. At appropriate treatment dosages, both methadone (≥60 mg) and BUP (≥ 16 mg) are highly effective for the treatment of OUD.1 For patients with health insurance, pharmacy benefits often provide some coverage for preferred products but no coverage for other products. Not all pharmacies carry BUP products. In a study of more than 5,000 pharmacies, approximately 60% reported that they carry and can dispense BUP medications.2
BUP monotherapy is available as generic sublingual tablets, buccal films (Belbuca), formulations for injection (Sublocade), and subcutaneous implants (Probuphine). BUPNAL is available as buccal films (Bunavail), sublingual films (Suboxone), and sublingual tablets (Zubsolv). For BUP-NAL combination productions, the following dose combinations have been reported to have similar effects: BUP-NAL 8 mg/2 mg sublingual film, BUP-NAL 5.7 mg/1.4 mg sublingual tablet, and BUP-NAL 4.2 mg/0.7 mg buccal film.3
When initiating BUP-monotherapy or BUP-NAL treatment for OUD, one approach for unobserved initiation is to instruct the patient to discontinue using opioid agonist drugs and wait for the onset of mild to moderate withdrawal symptoms. The purpose of this step is to avoid precipitating severe withdrawal symptoms caused by giving BUP or BUP-NAL to a patient who has recently used opioid drugs.
If BUP-NAL sublingual films (Suboxone) are prescribed following the onset of mild to moderate withdrawal symptoms, the patient can initiate therapy with a dose of 2 mg BUP/0.5 mg NAL or 4 mg BUP/1 mg NAL. At 60 to 120 minutes following the initial dose, if withdrawal symptoms persist, an additional dose of 4 mg BUP/1 mg NAL can be given. Thereafter, symptoms can be assessed every 60 to 120 minutes and additional doses administered to control symptoms. On the second day of therapy, a maximum of 16 mg of BUP is administered. Over the following days and weeks, if symptoms and cravings persist at a BUP dose of 16 mg, the total daily dose of BUP can be titrated up to 24 mg. For long-term treatment, a commonly prescribed daily dose is 16 mg BUP/4 mg NAL or 24 mg BUP/6 mg NAL. An absolute contraindication to BUP or BUP/NAL treatment is an allergy to the medication, and a relative contraindication is liver failure.
One potential complication of transmucosal BUP or BUP-NAL treatment is a dry mouth (xerostomia), which may contribute to dental disease.4 However, some experts question the quality of the data that contributed to the warning.5,6 Potential dental complications might be prevented by regular oral health examinations, daily flossing and teeth brushing, and stimulation of saliva by sugar-free gum or lozenges.
Primary care clinicians who initiate BUP or BUPNAL treatment for OUD often have a weekly visit with the patient during the initial phase of treatment and then every 3 to 4 weeks during maintenance therapy. Most patients need long-term treatment to achieve the goals of therapy, which include prevention of opioid overdose, reduction of cravings for nonprescription narcotics, and improvement in overall health. BUP and BUP-NAL treatment are effective without formal counseling, but counseling and social work support improve long-term adherence with treatment. Primary care clinicians who have experience with medication treatment of OUD report that their experience convinces them that medication treatment of OUD has similarities to the long-term treatment of diabetes, with antihyperglycemia medicines or the treatment of HIV infection with antiviral medications.
References
1. Mattick RP, Breen C, Kimber J, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014;CD002207.
2. Weiner SG, Qato DM, Faust JS, et al. Pharmacy availability of buprenorphine for opioid use disorder treatment in the U.S. JAMA Netw Open. 2023;6:E2316089.
3. Substance Abuse and Mental Health Services Administration (SAMHSA). Medications for opioid use disorder. SAMHSA website. Accessed August 21, 2023. https ://store.samhsa.gov/sites/default/files/SAMHSA_Digital_Download/PEP 21-02-01-002.pdf
4. FDA warns about dental problems with buprenorphine medicines dissolved in the mouth. FDA website. Accessed August 21, 2023. https ://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-about-dental-problems-buprenorphine-medicines-dissolved-mouth-treat-opioiduse-disorder#:~:text=What%20did%20FDA%20find%3F,medicines%20 dissolved%20in%20the%20mouth
5. Watson DP, Etmian S, Gastala N. Sublingual buprenorphine-naloxone exposure and dental disease. JAMA. 2023;329:1223-1224.
6. Brothers TD, Lewer D, Bonn M. Sublingual buprenorphine-naloxone exposure and dental disease. JAMA. 2023;329:1224.
Medication treatment of OUD in obstetrics
In the United States, the prevalence of OUD among pregnant patients hospitalized for delivery more than quadrupled from 1999 through 2014.18 BUP and methadone commonly are used to treat OUD during pregnancy.19 Among pregnant patients about 5% of buprenorphine prescriptions are written by obstetricians.20 An innovative approach to initiating BUP for pregnant patients with OUD is to use unobserved initiation, which involves outpatient discontinuation of nonprescription opioids to induce mild to moderate withdrawal symptoms followed by initiation of BUP treatment. In one cohort study, 55 pregnant patients used an unobserved outpatient protocol to initiate BUP treatment; 80% of the patients previously had used methadone or BUP. No patient experienced a precipitated withdrawal and 96% of patients returned for their office visit 1 week after initiation of treatment. Eighty-six percent of patients remained in treatment 3 months following initiation of BUP.21
Compared with methadone, BUP treatment during pregnancy may result in lower rates of neonatal abstinence syndrome. In one study of pregnant patients who were using methadone (n = 5,056) or BUP (n = 11,272) in late pregnancy, neonatal abstinence syndrome was diagnosed in 69.2% and 52.0% of newborns, respectively (adjusted relative risk, 0.73; 95% confidence interval, 0.71–0.75).22 In addition, compared with methadone, the use of BUP was associated with a reduced risk for low birth weight (14.9% vs 8.3%) and a lower risk for preterm birth (24.9% vs 14.4%). In this study, there were no differences in maternal obstetric outcomes when comparing BUP versus methadone treatment. Similar results have been reported in a meta-analysis analyzing the use of methadone and BUP during pregnancy.23 Studies performed to date have not shown an increased risk of congenital anomalies with the use of BUP-NAL during pregnancy.24,25
Although there may be differences in newborn outcomes with BUP and methadone, the American College of Obstetricians and Gynecologists does not recommend switching from methadone to BUP during pregnancy because precipitated withdrawal may occur.26 Based on recent studies, the American Society of Addiction Medicine has advised that it is safe to prescribe pregnant patients either BUP or BUP-NAL.27,28
Medication treatment of OUD with or without intensive counseling
The FDA recently reviewed literature related to the advantages and challenges of combining intensive counseling with medication treatment of OUD.29 The FDA noted that treatment saves lives and encouraged clinicians to initiate medication treatment of OUD or refer the patient to an appropriate clinician or treatment center. Combining medication treatment of OUD with intensive counseling is associated with greater treatment adherence and reduced health care costs. For example, in one study of 4,987 patients with OUD, initiation of counseling within 8 weeks of the start of medication treatment and a BUP dose of 16 mg or greater daily were associated with increased adherence to treatment.30 For patients receiving a BUP dose of less than 16 mg daily, treatment adherence with and without counseling was approximately 325 and 230 days, respectively. When the dose of BUP was 16 mg or greater, treatment adherence with and without counseling was approximately 405 and 320 days, respectively.30
Counseling should always be offered to patients initiating medication treatment of OUD. It should be noted that counseling alone is not a highly effective treatment for OUD.31 The FDA recently advised that the lack of availability of intensive counseling should not prevent clinicians from initiating BUP for the treatment of OUD.29 OUD is associated with a high mortalityrate and if counseling is not possible, medication treatment should be initiated. Substantial evidence demonstrates that medication treatment of OUD is associated with many benefits.16 The FDA advisory committee concluded that OUD treatment decisions should use shared decision making and be supportive and patient centered.29
The opportunities for medication treatment of OUD in primary care practice have expanded due to the recent FDA removal of restrictions on the use of BUP and heightened awareness of the positive public health impact of medication treatment. Challenges to the medication treatment of OUD remain, including stigmatization of OUD, barriers to insurance coverage for BUP, practice costs of treating OUD, and gaps in clinical education. For many pregnant patients, their main point of contact with health care is their obstetrician. By incorporating OUD treatment in pregnancy care, obstetricians will improve the health of the mother and newborn, contributing to the well-being of current and future generations. ●
Experts have recommended several interventions that may help reduce opioid overdose death.1 A consensus recommendation is that people who use drugs should be provided naloxone rescue medication and educated on the proper use of naloxone. Naloxone rescue medication is available in formulations for nasal or parenteral administration. The US Food and Drug Administration (FDA) recently has approved naloxone for over-the-counter status. The American Medical Association has provided a short web video on how to administer nasal naloxone.2 In a small pilot study, obstetricians offered every postpartum patient with naloxone administration education and a 2-dose nasal naloxone pack, with 76% of patients accepting the nasal naloxone pack.3
Many experts recommend that people who use drugs should be advised to never use them alone and to test a small amount of the drug to assess its potency. Many patients who use opioid drugs also take benzodiazepines, which can contribute to respiratory depression.4 Patients should avoid mixing drugs (eg, opioids and benzodiazepines). Some experts recommend that patients who use drugs should be provided take-home fentanyl test strips so they can evaluate their drugs for the presence of fentanyl, a medication that suppresses respiration and contributes to many overdose deaths. In addition, people who use drugs and are interested in reducing their use of drugs or managing overdose risk can be offered initiation of medication treatment of OUD.1
References
1. Wood E, Solomon ED, Hadland SE. Universal precautions for people at risk of opioid overdose in North America. JAMA Int Med. 2023;183:401-402.
2. How to administer Naloxone. AMA website. Accessed August 28, 2023. https://www.ama-assn.org /delivering-care/overdose-epidemic/how-administer-naloxone
3. Naliboff JA, Tharpe N. Universal postpartum naloxone provision: a harm reduction quality improvement project. J Addict Med. 2022;17:360-362.
4. Kelly JC, Raghuraman N, Stout MJ, et al. Home induction of buprenorphine for treatment of opioid use disorder in pregnancy. Obstet Gynecol. 2021;138:655-659.
The Centers for Disease Control and Prevention (CDC) reported 106,699 deaths in 2021 from drug overdose, with the majority being linked to synthetic opioids, including fentanyl and tramadol.1 This number compares with 42,795 deaths due to motor vehicle accidents and 48,183 deaths due to suicide in 2021.2,3 Most of the opioid overdose deaths occurred among people aged 25 to 64 years, the peak age of patients cared for by obstetrician-gynecologists. Among pregnant and postpartum persons, mortality due to drug overdose has increased by 81% between 2017 and 2020.4
Among pregnant and postpartum patients, drug overdose death is more common than suicide, and the risk for drug overdose death appears to be greatest in the year following delivery.5,6 In many cases, postpartum patients with OUD have had multiple contacts with the health care system prior to their death, showing that there is an opportunity for therapeutic intervention before the death occurred.7 Medication-assisted recovery for OUD involves a comprehensive array of interventions including medication, counseling, and social support. Medication treatment of OUD with BUP or methadone reduces the risk for death but is underutilized among patients with OUD.6,8 Recent federal legislation has removed restrictions on the use of BUP, increasing the opportunity for primary care clinicians to prescribe it for the treatment of OUD.9
Screening and diagnosis of OUD
Screening for OUD is recommended for patients who are at risk for opioid misuse (ie, those who are taking/have taken opioid medications). The OWLS (Overuse, Worrying, Losing interest, and feeling Slowed down, sluggish, or sedated) screening tool is used to detect prescription medication OUD and has 4 questions10:
1. In the past 3 months did you use your opioid medicines for other purposes—for example, to help you sleep or to help with stress or worry?
2. In the past 3 months did opioid medicines cause you to feel slowed down, sluggish, or sedated?
3. In the past 3 months did opioid medicines cause you to lose interest in your usual activities?
4. In the past 3 months did you worry about your use of opioid medicines?
Patient agreement with 3 or 4 questions indicates a positive screening test.
If the patient has a positive screening test, a formal diagnosis of OUD can be made using the 11 symptoms outlined in the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.11 The diagnosis of mild (2 to 3 symptoms), moderate (4 to 5 symptoms), or severe OUD (6 or more symptoms) is made based on the number of symptoms the patient reports.

Buprenorphine treatment of OUD in primary care
The role of primary care clinicians in the medication treatment of OUD is increasing. Using a nationwide system that tracks prescription medications, investigators reported that, in 2004, psychiatrists wrote 32.2% of all BUP prescriptions; in 2021, however, only 10% of such prescriptions were provided by psychiatrists, with most prescriptions written by non-psychiatrist physicians, nurse practitioners, and physician assistants that year.12 Innovative telehealth approaches to consultation and medication treatment of OUD are now available—one example is QuickMD.13 Such sites are designed to remove barriers to initiating medication treatment of OUD.
The role of primary care clinicians in the management of OUD using BUP and buprenorphine-naloxone (BUP-NAL) has increased due to many factors, including:
- the removal of US Food and Drug Administration (FDA) barriers to prescribing BUP
- the epidemic of OUD and the small size of the addiction specialist workforce, necessitating that primary care clinicians become engaged in the treatment of OUD
- an increase in unobserved initiation of BUP among ambulatory patients, and a parallel decrease in cases of observed initiation in addiction center settings
- the reframing of OUD as a chronic medical problem, with many similarities to diabetes, obesity, dyslipidemia, and hypertension.
Similar to other diseases managed by primary care clinicians, OUD requires long-term chronic treatment with a medicine that, if taken as directed, provides excellent outcomes. Primary care clinicians who prescribe BUP also can optimize longitudinal care for comorbid disorders such as hypertension and diabetes, which are prevalent in people with OUD.
In 2019, New Jersey implemented new guidelines for the treatment of OUD, removing prior authorization barriers, increasing reimbursement for office-based OUD treatment, and establishing regional centers of excellence. The implementation of the new guidelines was followed by a marked increase in BUP prescribers among primary care clinicians, emergency medicine physicians, and advanced practice clinicians.14
To estimate the public health impact of BUP prescribing by primary care clinicians, investigators simulated patient outcomes in 3 scenarios15:
1. primary care clinicians refer patients to addiction specialists for OUD treatment
2. primary care clinicians provide BUP services in their practice
3. primary care clinicians provide BUP and harm reduction kits containing syringes and wound care supplies in their practice.
Strategies 2 and 3 resulted in 14% fewer deaths due to opioid overdose, an increased life expectancy of approximately 2.7 years, and reduced hospital costs. For strategy 3, the incremental cost per life-year saved was $34,400. The investigators noted that prescribing BUP in primary care practice increases practice costs.15
Treatment with BUP reduces death from opioid overdose, improves patient health, decreases use of illicit opioids, and reduces patient cravings for opioids. BUP is a safe medication and is associated with fewer adverse effects than insulin or warfarin.16
Continue to: Methadone treatment of OUD...
Methadone treatment of OUD
Methadone is a full opioid agonist approved by the FDA for the treatment of severe pain or OUD. Methadone treatment of OUD is strictly regulated and typically is ordered and administered at an opioid treatment program that is federally licensed. Methadone for OUD treatment cannot be prescribed by a physician to a pharmacy, limiting its use in primary care practice. Methadone used to treat OUD is ordered and dispensed at opioid-treatment programs. Take-home doses of methadone may be available to patients after adherence to the regimen has been established. When used long-term, higher doses of methadone are associated with better adherence, but these higher doses can cause respiratory depression. In a study of 189 pregnant patients taking methadone to treat OUD, daily doses of 60 mg or greater were associated with better treatment retention at delivery and 60 days postpartum, as well as less use of nonprescription opioids.17 Under limited circumstances methadone can be ordered and dispensed for hospitalized patients with OUD.
Methadone is a pure opioid receptor agonist. Naloxone (NAL) is an opioid receptor antagonist. Buprenorphine (BUP) is a partial opioid receptor agonist-antagonist, which limits overdose risk. BUP often is combined with NAL as a combination formulation, which is thought to reduce the repurposing of BUP for non-prescribed uses. At appropriate treatment dosages, both methadone (≥60 mg) and BUP (≥ 16 mg) are highly effective for the treatment of OUD.1 For patients with health insurance, pharmacy benefits often provide some coverage for preferred products but no coverage for other products. Not all pharmacies carry BUP products. In a study of more than 5,000 pharmacies, approximately 60% reported that they carry and can dispense BUP medications.2
BUP monotherapy is available as generic sublingual tablets, buccal films (Belbuca), formulations for injection (Sublocade), and subcutaneous implants (Probuphine). BUPNAL is available as buccal films (Bunavail), sublingual films (Suboxone), and sublingual tablets (Zubsolv). For BUP-NAL combination productions, the following dose combinations have been reported to have similar effects: BUP-NAL 8 mg/2 mg sublingual film, BUP-NAL 5.7 mg/1.4 mg sublingual tablet, and BUP-NAL 4.2 mg/0.7 mg buccal film.3
When initiating BUP-monotherapy or BUP-NAL treatment for OUD, one approach for unobserved initiation is to instruct the patient to discontinue using opioid agonist drugs and wait for the onset of mild to moderate withdrawal symptoms. The purpose of this step is to avoid precipitating severe withdrawal symptoms caused by giving BUP or BUP-NAL to a patient who has recently used opioid drugs.
If BUP-NAL sublingual films (Suboxone) are prescribed following the onset of mild to moderate withdrawal symptoms, the patient can initiate therapy with a dose of 2 mg BUP/0.5 mg NAL or 4 mg BUP/1 mg NAL. At 60 to 120 minutes following the initial dose, if withdrawal symptoms persist, an additional dose of 4 mg BUP/1 mg NAL can be given. Thereafter, symptoms can be assessed every 60 to 120 minutes and additional doses administered to control symptoms. On the second day of therapy, a maximum of 16 mg of BUP is administered. Over the following days and weeks, if symptoms and cravings persist at a BUP dose of 16 mg, the total daily dose of BUP can be titrated up to 24 mg. For long-term treatment, a commonly prescribed daily dose is 16 mg BUP/4 mg NAL or 24 mg BUP/6 mg NAL. An absolute contraindication to BUP or BUP/NAL treatment is an allergy to the medication, and a relative contraindication is liver failure.
One potential complication of transmucosal BUP or BUP-NAL treatment is a dry mouth (xerostomia), which may contribute to dental disease.4 However, some experts question the quality of the data that contributed to the warning.5,6 Potential dental complications might be prevented by regular oral health examinations, daily flossing and teeth brushing, and stimulation of saliva by sugar-free gum or lozenges.
Primary care clinicians who initiate BUP or BUPNAL treatment for OUD often have a weekly visit with the patient during the initial phase of treatment and then every 3 to 4 weeks during maintenance therapy. Most patients need long-term treatment to achieve the goals of therapy, which include prevention of opioid overdose, reduction of cravings for nonprescription narcotics, and improvement in overall health. BUP and BUP-NAL treatment are effective without formal counseling, but counseling and social work support improve long-term adherence with treatment. Primary care clinicians who have experience with medication treatment of OUD report that their experience convinces them that medication treatment of OUD has similarities to the long-term treatment of diabetes, with antihyperglycemia medicines or the treatment of HIV infection with antiviral medications.
References
1. Mattick RP, Breen C, Kimber J, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014;CD002207.
2. Weiner SG, Qato DM, Faust JS, et al. Pharmacy availability of buprenorphine for opioid use disorder treatment in the U.S. JAMA Netw Open. 2023;6:E2316089.
3. Substance Abuse and Mental Health Services Administration (SAMHSA). Medications for opioid use disorder. SAMHSA website. Accessed August 21, 2023. https ://store.samhsa.gov/sites/default/files/SAMHSA_Digital_Download/PEP 21-02-01-002.pdf
4. FDA warns about dental problems with buprenorphine medicines dissolved in the mouth. FDA website. Accessed August 21, 2023. https ://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-about-dental-problems-buprenorphine-medicines-dissolved-mouth-treat-opioiduse-disorder#:~:text=What%20did%20FDA%20find%3F,medicines%20 dissolved%20in%20the%20mouth
5. Watson DP, Etmian S, Gastala N. Sublingual buprenorphine-naloxone exposure and dental disease. JAMA. 2023;329:1223-1224.
6. Brothers TD, Lewer D, Bonn M. Sublingual buprenorphine-naloxone exposure and dental disease. JAMA. 2023;329:1224.
Medication treatment of OUD in obstetrics
In the United States, the prevalence of OUD among pregnant patients hospitalized for delivery more than quadrupled from 1999 through 2014.18 BUP and methadone commonly are used to treat OUD during pregnancy.19 Among pregnant patients about 5% of buprenorphine prescriptions are written by obstetricians.20 An innovative approach to initiating BUP for pregnant patients with OUD is to use unobserved initiation, which involves outpatient discontinuation of nonprescription opioids to induce mild to moderate withdrawal symptoms followed by initiation of BUP treatment. In one cohort study, 55 pregnant patients used an unobserved outpatient protocol to initiate BUP treatment; 80% of the patients previously had used methadone or BUP. No patient experienced a precipitated withdrawal and 96% of patients returned for their office visit 1 week after initiation of treatment. Eighty-six percent of patients remained in treatment 3 months following initiation of BUP.21
Compared with methadone, BUP treatment during pregnancy may result in lower rates of neonatal abstinence syndrome. In one study of pregnant patients who were using methadone (n = 5,056) or BUP (n = 11,272) in late pregnancy, neonatal abstinence syndrome was diagnosed in 69.2% and 52.0% of newborns, respectively (adjusted relative risk, 0.73; 95% confidence interval, 0.71–0.75).22 In addition, compared with methadone, the use of BUP was associated with a reduced risk for low birth weight (14.9% vs 8.3%) and a lower risk for preterm birth (24.9% vs 14.4%). In this study, there were no differences in maternal obstetric outcomes when comparing BUP versus methadone treatment. Similar results have been reported in a meta-analysis analyzing the use of methadone and BUP during pregnancy.23 Studies performed to date have not shown an increased risk of congenital anomalies with the use of BUP-NAL during pregnancy.24,25
Although there may be differences in newborn outcomes with BUP and methadone, the American College of Obstetricians and Gynecologists does not recommend switching from methadone to BUP during pregnancy because precipitated withdrawal may occur.26 Based on recent studies, the American Society of Addiction Medicine has advised that it is safe to prescribe pregnant patients either BUP or BUP-NAL.27,28
Medication treatment of OUD with or without intensive counseling
The FDA recently reviewed literature related to the advantages and challenges of combining intensive counseling with medication treatment of OUD.29 The FDA noted that treatment saves lives and encouraged clinicians to initiate medication treatment of OUD or refer the patient to an appropriate clinician or treatment center. Combining medication treatment of OUD with intensive counseling is associated with greater treatment adherence and reduced health care costs. For example, in one study of 4,987 patients with OUD, initiation of counseling within 8 weeks of the start of medication treatment and a BUP dose of 16 mg or greater daily were associated with increased adherence to treatment.30 For patients receiving a BUP dose of less than 16 mg daily, treatment adherence with and without counseling was approximately 325 and 230 days, respectively. When the dose of BUP was 16 mg or greater, treatment adherence with and without counseling was approximately 405 and 320 days, respectively.30
Counseling should always be offered to patients initiating medication treatment of OUD. It should be noted that counseling alone is not a highly effective treatment for OUD.31 The FDA recently advised that the lack of availability of intensive counseling should not prevent clinicians from initiating BUP for the treatment of OUD.29 OUD is associated with a high mortalityrate and if counseling is not possible, medication treatment should be initiated. Substantial evidence demonstrates that medication treatment of OUD is associated with many benefits.16 The FDA advisory committee concluded that OUD treatment decisions should use shared decision making and be supportive and patient centered.29
The opportunities for medication treatment of OUD in primary care practice have expanded due to the recent FDA removal of restrictions on the use of BUP and heightened awareness of the positive public health impact of medication treatment. Challenges to the medication treatment of OUD remain, including stigmatization of OUD, barriers to insurance coverage for BUP, practice costs of treating OUD, and gaps in clinical education. For many pregnant patients, their main point of contact with health care is their obstetrician. By incorporating OUD treatment in pregnancy care, obstetricians will improve the health of the mother and newborn, contributing to the well-being of current and future generations. ●
Experts have recommended several interventions that may help reduce opioid overdose death.1 A consensus recommendation is that people who use drugs should be provided naloxone rescue medication and educated on the proper use of naloxone. Naloxone rescue medication is available in formulations for nasal or parenteral administration. The US Food and Drug Administration (FDA) recently has approved naloxone for over-the-counter status. The American Medical Association has provided a short web video on how to administer nasal naloxone.2 In a small pilot study, obstetricians offered every postpartum patient with naloxone administration education and a 2-dose nasal naloxone pack, with 76% of patients accepting the nasal naloxone pack.3
Many experts recommend that people who use drugs should be advised to never use them alone and to test a small amount of the drug to assess its potency. Many patients who use opioid drugs also take benzodiazepines, which can contribute to respiratory depression.4 Patients should avoid mixing drugs (eg, opioids and benzodiazepines). Some experts recommend that patients who use drugs should be provided take-home fentanyl test strips so they can evaluate their drugs for the presence of fentanyl, a medication that suppresses respiration and contributes to many overdose deaths. In addition, people who use drugs and are interested in reducing their use of drugs or managing overdose risk can be offered initiation of medication treatment of OUD.1
References
1. Wood E, Solomon ED, Hadland SE. Universal precautions for people at risk of opioid overdose in North America. JAMA Int Med. 2023;183:401-402.
2. How to administer Naloxone. AMA website. Accessed August 28, 2023. https://www.ama-assn.org /delivering-care/overdose-epidemic/how-administer-naloxone
3. Naliboff JA, Tharpe N. Universal postpartum naloxone provision: a harm reduction quality improvement project. J Addict Med. 2022;17:360-362.
4. Kelly JC, Raghuraman N, Stout MJ, et al. Home induction of buprenorphine for treatment of opioid use disorder in pregnancy. Obstet Gynecol. 2021;138:655-659.
- Spencer MR, Miniño AM, Warner M. Drug overdose deaths in the United States, 20012021. NCHS Data Brief no 457. Hyattsville, MD, National Center for Health Statistics. 2022. NCHS Data Brief No. 457. Published December 2022. Accessed August 21, 2023. https://www.cdc.gov /nchs/products/databriefs/db457.htm
- US traffic deaths drop slightly in 2022 but still a ‘crisis.’ AP News website. Published April 20, 2023. Accessed August 21, 2023. https://apnews.com /article/traffic-deaths-distracted-driving-crisis -6db6471e273b275920b6c4f9eb7e493b
- Suicide statistics. American Foundation for Suicide Prevention website. Accessed August 21, 2023. https://afsp.org/suicide-statistics/
- Bruzelius E, Martins SS. US Trends in drug overdose mortality among pregnant and postpartum persons, 2017-2020. JAMA. 2022;328:2159-2161.
- Metz TD, Rovner P, Hoffman MC, et al. Maternal deaths from suicide and overdose in Colorado, 2004-2012. Obstet Gynecol. 2016;128:1233-1240.
- Schiff DM, Nielsen T, Terplan M, et al. Fatal and nonfatal overdose among pregnant and postpartum women in Massachusetts. Obstet Gynecol. 2018;132:466-474.
- Goldman-Mellor S, Margerison CE. Maternal drug-related death and suicide are leading causes of postpartum death in California. Am J Obstet Gynecol. 2019;221:489.e1-489.e9.
- Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550.
- Waiver elimination (MAT Act). SAMHSA website. Accessed August 21, 2023. https://www .samhsa.gov/medications-substance-use- disorders/removal-data-waiver-requirement
- Picco L, Middleton M, Bruno R, et al. Validation of the OWLS, a Screening Tool for Measuring Prescription Opioid Use Disorder in Primary Care. Pain Med. 2020;21:2757-2764.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013.
- Creedon TB, Ali MM, Schuman-Olivier Z. Trends in buprenorphine prescribing for opioid use disorder by psychiatrists in the US from 2003 to 2021. JAMA Health Forum. 2023;4:E230221.
- Quick MD website. Accessed August 21, 2023. https://quick.md/
- Treitler P, Nowels M, Samples H, et al. BUP utilization and prescribing among New Jersey Medicaid beneficiaries after adoption of initiatives designed to improve treatment access. JAMA Netw Open. 2023;6:E2312030.
- Jawa R, Tin Y, Nall S, et al. Estimated clinical outcomes and cost-effectiveness associated with provision of addiction treatment in US primary care clinics. JAMA Netw Open. 2023;6:E237888.
- Wakeman SE, Larochelle MR, Ameli O, et al. Comparative effectiveness of different treatment pathways of opioid use disorder. JAMA Netw Open. 2020;3:E1920622.
- Wilder CM, Hosta D, Winhusen T. Association of methadone dose with substance use and treatment retention in pregnant and postpartum women with opioid use disorder. J Subst Abuse Treat. 2017;80:33-36.
- Haight SC, Ko JY, Tong VT, et al. Opioid use disorder documented at delivery hospitalization - United States, 1999-2014. MMWR Morb Mortal Wkly Rep. 2018;67:845-849.
- Xu KY, Jones HE, Schiff DM, et al. Initiation and treatment discontinuation of medications for opioid use disorder in pregnant people compared with nonpregnant people. Obstet Gynecol. 2023;141:845-853.
- Kelly D, Krans EE. Medical specialty of buprenorphine prescribers for pregnant women with opioid use disorder. Am J Obstet Gynecol. 2019;220:502-503.
- Kelly JC, Raghuraman N, Stout MJ, et al. Home induction of buprenorphine for treatment of opioid use disorder in pregnancy. Obstet Gynecol. 2021;138:655-659.
- Suarez EA, Huybrechts KF, Straub L, et al. Buprenorphine versus methadone for opioid use disorder in pregnancy. N Engl J Med. 2022;387:2033-2044.
- Kinsella M, Halliday LO, Shaw M, et al. Buprenorphine compared with methadone in pregnancy: a systematic review and meta-analysis. Subst Use Misuse. 2022;57:1400-1416.
- Jumah NA, Edwards C, Balfour-Boehm J, et al. Observational study of the safety of buprenorphine-naloxone in pregnancy in a rural and remote population. BMJ Open. 2016;6:E011774.
- Mullins N, Galvin SL, Ramage M, et al. Buprenorphine and naloxone versus buprenorphine for opioid use disorder in pregnancy: a cohort study. J Addict Med. 2020;14:185-192.
- Opioid use and opioid use disorder in pregnancy. Committee Opinion No. 711. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2017;130:E81-E94.
- The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder: 2020 Focused Update. J Addict Med. 2020;14(2S suppl 1):1-91.
- Link HM, Jones H, Miller L, et al. Buprenorphinenaloxone use in pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol MFM. 2020;2:100179.
- Delphin-Rittmon ME, Cavazzoni P. US Food and Drug Administration website. https://www.fda .gov/media/168027/download
- Eren K, Schuster J, Herschell A, et al. Association of Counseling and Psychotherapy on retention in medication for addiction treatment within a large Medicaid population. J Addict Med. 2022;16:346353.
- Kakko J, Dybrandt Svanborg K, Kreek MJ, et al. 1-year retention and social function after buprenorphine-assisted relapse prevention treatment for heroin dependence in Sweden: a randomized, placebo-controlled trial. Lancet. 2003;361:662-668.
- Spencer MR, Miniño AM, Warner M. Drug overdose deaths in the United States, 20012021. NCHS Data Brief no 457. Hyattsville, MD, National Center for Health Statistics. 2022. NCHS Data Brief No. 457. Published December 2022. Accessed August 21, 2023. https://www.cdc.gov /nchs/products/databriefs/db457.htm
- US traffic deaths drop slightly in 2022 but still a ‘crisis.’ AP News website. Published April 20, 2023. Accessed August 21, 2023. https://apnews.com /article/traffic-deaths-distracted-driving-crisis -6db6471e273b275920b6c4f9eb7e493b
- Suicide statistics. American Foundation for Suicide Prevention website. Accessed August 21, 2023. https://afsp.org/suicide-statistics/
- Bruzelius E, Martins SS. US Trends in drug overdose mortality among pregnant and postpartum persons, 2017-2020. JAMA. 2022;328:2159-2161.
- Metz TD, Rovner P, Hoffman MC, et al. Maternal deaths from suicide and overdose in Colorado, 2004-2012. Obstet Gynecol. 2016;128:1233-1240.
- Schiff DM, Nielsen T, Terplan M, et al. Fatal and nonfatal overdose among pregnant and postpartum women in Massachusetts. Obstet Gynecol. 2018;132:466-474.
- Goldman-Mellor S, Margerison CE. Maternal drug-related death and suicide are leading causes of postpartum death in California. Am J Obstet Gynecol. 2019;221:489.e1-489.e9.
- Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550.
- Waiver elimination (MAT Act). SAMHSA website. Accessed August 21, 2023. https://www .samhsa.gov/medications-substance-use- disorders/removal-data-waiver-requirement
- Picco L, Middleton M, Bruno R, et al. Validation of the OWLS, a Screening Tool for Measuring Prescription Opioid Use Disorder in Primary Care. Pain Med. 2020;21:2757-2764.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013.
- Creedon TB, Ali MM, Schuman-Olivier Z. Trends in buprenorphine prescribing for opioid use disorder by psychiatrists in the US from 2003 to 2021. JAMA Health Forum. 2023;4:E230221.
- Quick MD website. Accessed August 21, 2023. https://quick.md/
- Treitler P, Nowels M, Samples H, et al. BUP utilization and prescribing among New Jersey Medicaid beneficiaries after adoption of initiatives designed to improve treatment access. JAMA Netw Open. 2023;6:E2312030.
- Jawa R, Tin Y, Nall S, et al. Estimated clinical outcomes and cost-effectiveness associated with provision of addiction treatment in US primary care clinics. JAMA Netw Open. 2023;6:E237888.
- Wakeman SE, Larochelle MR, Ameli O, et al. Comparative effectiveness of different treatment pathways of opioid use disorder. JAMA Netw Open. 2020;3:E1920622.
- Wilder CM, Hosta D, Winhusen T. Association of methadone dose with substance use and treatment retention in pregnant and postpartum women with opioid use disorder. J Subst Abuse Treat. 2017;80:33-36.
- Haight SC, Ko JY, Tong VT, et al. Opioid use disorder documented at delivery hospitalization - United States, 1999-2014. MMWR Morb Mortal Wkly Rep. 2018;67:845-849.
- Xu KY, Jones HE, Schiff DM, et al. Initiation and treatment discontinuation of medications for opioid use disorder in pregnant people compared with nonpregnant people. Obstet Gynecol. 2023;141:845-853.
- Kelly D, Krans EE. Medical specialty of buprenorphine prescribers for pregnant women with opioid use disorder. Am J Obstet Gynecol. 2019;220:502-503.
- Kelly JC, Raghuraman N, Stout MJ, et al. Home induction of buprenorphine for treatment of opioid use disorder in pregnancy. Obstet Gynecol. 2021;138:655-659.
- Suarez EA, Huybrechts KF, Straub L, et al. Buprenorphine versus methadone for opioid use disorder in pregnancy. N Engl J Med. 2022;387:2033-2044.
- Kinsella M, Halliday LO, Shaw M, et al. Buprenorphine compared with methadone in pregnancy: a systematic review and meta-analysis. Subst Use Misuse. 2022;57:1400-1416.
- Jumah NA, Edwards C, Balfour-Boehm J, et al. Observational study of the safety of buprenorphine-naloxone in pregnancy in a rural and remote population. BMJ Open. 2016;6:E011774.
- Mullins N, Galvin SL, Ramage M, et al. Buprenorphine and naloxone versus buprenorphine for opioid use disorder in pregnancy: a cohort study. J Addict Med. 2020;14:185-192.
- Opioid use and opioid use disorder in pregnancy. Committee Opinion No. 711. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2017;130:E81-E94.
- The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder: 2020 Focused Update. J Addict Med. 2020;14(2S suppl 1):1-91.
- Link HM, Jones H, Miller L, et al. Buprenorphinenaloxone use in pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol MFM. 2020;2:100179.
- Delphin-Rittmon ME, Cavazzoni P. US Food and Drug Administration website. https://www.fda .gov/media/168027/download
- Eren K, Schuster J, Herschell A, et al. Association of Counseling and Psychotherapy on retention in medication for addiction treatment within a large Medicaid population. J Addict Med. 2022;16:346353.
- Kakko J, Dybrandt Svanborg K, Kreek MJ, et al. 1-year retention and social function after buprenorphine-assisted relapse prevention treatment for heroin dependence in Sweden: a randomized, placebo-controlled trial. Lancet. 2003;361:662-668.
Nurses maintain more stigma toward pregnant women with OUD
Opioid use disorder among pregnant women continues to rise, and untreated opioid use is associated with complications including preterm delivery, placental abruption, and stillbirth, wrote Alexis Braverman, MD, of the University of Illinois, Chicago, and colleagues. However, many perinatal women who seek care and medications for opioid use disorder (OUD) report stigma that limits their ability to reduce these risks.
In a study published in the American Journal on Addictions , the researchers conducted an anonymous survey of 132 health care workers at six outpatient locations and a main hospital of an urban medical center. The survey was designed to assess attitudes toward pregnant women who were using opioids. The 119 complete responses in the final analysis included 40 nurses and 79 clinicians across ob.gyn., family medicine, and pediatrics. A total of 19 respondents were waivered to prescribe outpatient buprenorphine for OUD.
Nurses were significantly less likely than clinicians to agree that OUD is a chronic illness, to feel sympathy for women who use opioids during pregnancy, and to see pregnancy as an opportunity for behavior change (P = .000, P = .003, and P = .001, respectively).
Overall, family medicine providers and clinicians with 11-20 years of practice experience were significantly more sympathetic to pregnant women who used opioids, compared with providers from other departments and with fewer years of practice (P = .025 and P = .039, respectively).
Providers in pediatrics departments were significantly more likely than those from other departments to agree strongly with feeling anger at pregnant women who use opioids (P = .009), and that these women should not be allowed to parent (P = .013). However, providers in pediatrics were significantly more comfortable than those in other departments with discussing the involvement of social services in patient care (P = .020) and with counseling patients on neonatal opioid withdrawal syndrome, known as NOWS (P = .027).
“We hypothesize that nurses who perform more acute, inpatient work rather than outpatient work may not be exposed as frequently to a patient’s personal progress on their journey with OUD,” and therefore might not be exposed to the rewarding experiences and progress made by patients, the researchers wrote in their discussion.
However, the overall low level of comfort in discussing NOWS and social service involvement across provider groups (one-quarter for pediatrics, one-fifth for ob.gyn, and one-sixth for family medicine) highlights the need for further training in this area, they said.
The findings were limited by several factors, including the potential for responder bias; however, the results identify a need for greater training in stigma reduction and in counseling families on issues related to OUD, the researchers said. More studies are needed to examine attitude changes after the implementation of stigma reduction strategies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Opioid use disorder among pregnant women continues to rise, and untreated opioid use is associated with complications including preterm delivery, placental abruption, and stillbirth, wrote Alexis Braverman, MD, of the University of Illinois, Chicago, and colleagues. However, many perinatal women who seek care and medications for opioid use disorder (OUD) report stigma that limits their ability to reduce these risks.
In a study published in the American Journal on Addictions , the researchers conducted an anonymous survey of 132 health care workers at six outpatient locations and a main hospital of an urban medical center. The survey was designed to assess attitudes toward pregnant women who were using opioids. The 119 complete responses in the final analysis included 40 nurses and 79 clinicians across ob.gyn., family medicine, and pediatrics. A total of 19 respondents were waivered to prescribe outpatient buprenorphine for OUD.
Nurses were significantly less likely than clinicians to agree that OUD is a chronic illness, to feel sympathy for women who use opioids during pregnancy, and to see pregnancy as an opportunity for behavior change (P = .000, P = .003, and P = .001, respectively).
Overall, family medicine providers and clinicians with 11-20 years of practice experience were significantly more sympathetic to pregnant women who used opioids, compared with providers from other departments and with fewer years of practice (P = .025 and P = .039, respectively).
Providers in pediatrics departments were significantly more likely than those from other departments to agree strongly with feeling anger at pregnant women who use opioids (P = .009), and that these women should not be allowed to parent (P = .013). However, providers in pediatrics were significantly more comfortable than those in other departments with discussing the involvement of social services in patient care (P = .020) and with counseling patients on neonatal opioid withdrawal syndrome, known as NOWS (P = .027).
“We hypothesize that nurses who perform more acute, inpatient work rather than outpatient work may not be exposed as frequently to a patient’s personal progress on their journey with OUD,” and therefore might not be exposed to the rewarding experiences and progress made by patients, the researchers wrote in their discussion.
However, the overall low level of comfort in discussing NOWS and social service involvement across provider groups (one-quarter for pediatrics, one-fifth for ob.gyn, and one-sixth for family medicine) highlights the need for further training in this area, they said.
The findings were limited by several factors, including the potential for responder bias; however, the results identify a need for greater training in stigma reduction and in counseling families on issues related to OUD, the researchers said. More studies are needed to examine attitude changes after the implementation of stigma reduction strategies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Opioid use disorder among pregnant women continues to rise, and untreated opioid use is associated with complications including preterm delivery, placental abruption, and stillbirth, wrote Alexis Braverman, MD, of the University of Illinois, Chicago, and colleagues. However, many perinatal women who seek care and medications for opioid use disorder (OUD) report stigma that limits their ability to reduce these risks.
In a study published in the American Journal on Addictions , the researchers conducted an anonymous survey of 132 health care workers at six outpatient locations and a main hospital of an urban medical center. The survey was designed to assess attitudes toward pregnant women who were using opioids. The 119 complete responses in the final analysis included 40 nurses and 79 clinicians across ob.gyn., family medicine, and pediatrics. A total of 19 respondents were waivered to prescribe outpatient buprenorphine for OUD.
Nurses were significantly less likely than clinicians to agree that OUD is a chronic illness, to feel sympathy for women who use opioids during pregnancy, and to see pregnancy as an opportunity for behavior change (P = .000, P = .003, and P = .001, respectively).
Overall, family medicine providers and clinicians with 11-20 years of practice experience were significantly more sympathetic to pregnant women who used opioids, compared with providers from other departments and with fewer years of practice (P = .025 and P = .039, respectively).
Providers in pediatrics departments were significantly more likely than those from other departments to agree strongly with feeling anger at pregnant women who use opioids (P = .009), and that these women should not be allowed to parent (P = .013). However, providers in pediatrics were significantly more comfortable than those in other departments with discussing the involvement of social services in patient care (P = .020) and with counseling patients on neonatal opioid withdrawal syndrome, known as NOWS (P = .027).
“We hypothesize that nurses who perform more acute, inpatient work rather than outpatient work may not be exposed as frequently to a patient’s personal progress on their journey with OUD,” and therefore might not be exposed to the rewarding experiences and progress made by patients, the researchers wrote in their discussion.
However, the overall low level of comfort in discussing NOWS and social service involvement across provider groups (one-quarter for pediatrics, one-fifth for ob.gyn, and one-sixth for family medicine) highlights the need for further training in this area, they said.
The findings were limited by several factors, including the potential for responder bias; however, the results identify a need for greater training in stigma reduction and in counseling families on issues related to OUD, the researchers said. More studies are needed to examine attitude changes after the implementation of stigma reduction strategies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM THE AMERICAN JOURNAL ON ADDICTIONS
AI mammogram screening is equivalent to human readers
, a radiology and biomedical imaging professor at Yale University in New Haven, Conn.
The reason is because AI is proving to be as good as humans in interpreting mammograms, at least in the research setting.
In one of the latest reports, published online in Radiology, British investigators found that the performance of a commercially available AI system (INSIGHT MMG version 1.1.7.1 – Lunit) was essentially equivalent to over 500 specialized readers. The results are in line with other recent AI studies.
Double reading – having mammograms read by two clinicians to increase cancer detection rates – is common in the United Kingdom and elsewhere in Europe.
The British team compared the performance of 552 readers with Lunit’s AI program on the Personal Performance in Mammographic Screening exam, a quality assurance test which mammogram readers in the United Kingdom are required to take twice a year. Readers assign a malignancy score to 60 challenging cases, a mix of normal breasts and breasts with benign and cancerous lesions. The study included two test sessions for a total of 120 breast screenings.
Fifty-seven percent of the readers in the study were board-certified radiologists, 37% were radiographers, and 6% were breast clinicians. Each read at least 5,000 mammograms a year.
There was no difference in overall performance between the AI program and the human readers (AUC 0.93 vs. 0.88, P = .15).
Commenting in an editorial published with the investigation, Dr. Philpotts said the results “suggest that AI could confidently act as a second reader to decrease workloads.”
As for the United States, where double reading is generally not done, she pointed out that “many U.S. radiologists interpreting mammograms are nonspecialized and do not read high volumes of mammograms. Thus, the AI system evaluated in the study “could be used as a supplemental tool to aid the performance of readers in the United States or in other countries where screening programs use a single reading.”
There was also no difference in sensitivity between AI and human readers (84% vs. 90%, P = .34), but the AI algorithm had a higher specificity (89% vs. 76%, P = .003).
Using AI recall scores that matched the average human reader performance (90% sensitivity, 76% specificity), there was no difference with AI in regard to sensitivity (91%, P = .73) or specificity (77%, P = .85), but the investigators noted the power of the analysis was limited.
Overall, “diagnostic performance of AI was comparable with that of the average human reader.” It seems “increasingly likely that AI will eventually play a part in the interpretation of screening mammograms,” said investigators led by Yan Chen, PhD, of the Nottingham Breast Institute in England.
“That the AI system was able to match the performance of the average reader in this specialized group of mammogram readers indicates the robustness of this AI algorithm,” Dr. Philpotts said.
However, there are some caveats.
For one, the system was designed for 2D mammography, the current standard of care in the United Kingdom, while digital breast tomosynthesis (DBT) is replacing 2D mammography in the United States.
In the United States, “AI algorithms specific to DBT are necessary and will need to be reliable and reproducible to be embraced by radiologists,” Dr. Philpotts said.
Also in the United Kingdom, screening is performed at 3-year intervals in women aged 50-70 years old, which means that the study population was enriched for older women with less-dense breasts. Screening generally starts earlier in the United States and includes premenopausal women with denser breasts.
A recent study from Korea, where many women have dense breasts, found that 2D mammography and supplementary ultrasound outperformed AI for cancer detection.
“This underscores the challenges of finding cancers in dense breasts, which plague both radiologists and AI alike, and provides evidence that breast density is an important factor to consider when evaluating AI performance,” Dr. Philpotts said.
The work was funded by Lunit, the maker of the AI program used in the study. The investigators and Dr. Philpotts had no disclosures.
, a radiology and biomedical imaging professor at Yale University in New Haven, Conn.
The reason is because AI is proving to be as good as humans in interpreting mammograms, at least in the research setting.
In one of the latest reports, published online in Radiology, British investigators found that the performance of a commercially available AI system (INSIGHT MMG version 1.1.7.1 – Lunit) was essentially equivalent to over 500 specialized readers. The results are in line with other recent AI studies.
Double reading – having mammograms read by two clinicians to increase cancer detection rates – is common in the United Kingdom and elsewhere in Europe.
The British team compared the performance of 552 readers with Lunit’s AI program on the Personal Performance in Mammographic Screening exam, a quality assurance test which mammogram readers in the United Kingdom are required to take twice a year. Readers assign a malignancy score to 60 challenging cases, a mix of normal breasts and breasts with benign and cancerous lesions. The study included two test sessions for a total of 120 breast screenings.
Fifty-seven percent of the readers in the study were board-certified radiologists, 37% were radiographers, and 6% were breast clinicians. Each read at least 5,000 mammograms a year.
There was no difference in overall performance between the AI program and the human readers (AUC 0.93 vs. 0.88, P = .15).
Commenting in an editorial published with the investigation, Dr. Philpotts said the results “suggest that AI could confidently act as a second reader to decrease workloads.”
As for the United States, where double reading is generally not done, she pointed out that “many U.S. radiologists interpreting mammograms are nonspecialized and do not read high volumes of mammograms. Thus, the AI system evaluated in the study “could be used as a supplemental tool to aid the performance of readers in the United States or in other countries where screening programs use a single reading.”
There was also no difference in sensitivity between AI and human readers (84% vs. 90%, P = .34), but the AI algorithm had a higher specificity (89% vs. 76%, P = .003).
Using AI recall scores that matched the average human reader performance (90% sensitivity, 76% specificity), there was no difference with AI in regard to sensitivity (91%, P = .73) or specificity (77%, P = .85), but the investigators noted the power of the analysis was limited.
Overall, “diagnostic performance of AI was comparable with that of the average human reader.” It seems “increasingly likely that AI will eventually play a part in the interpretation of screening mammograms,” said investigators led by Yan Chen, PhD, of the Nottingham Breast Institute in England.
“That the AI system was able to match the performance of the average reader in this specialized group of mammogram readers indicates the robustness of this AI algorithm,” Dr. Philpotts said.
However, there are some caveats.
For one, the system was designed for 2D mammography, the current standard of care in the United Kingdom, while digital breast tomosynthesis (DBT) is replacing 2D mammography in the United States.
In the United States, “AI algorithms specific to DBT are necessary and will need to be reliable and reproducible to be embraced by radiologists,” Dr. Philpotts said.
Also in the United Kingdom, screening is performed at 3-year intervals in women aged 50-70 years old, which means that the study population was enriched for older women with less-dense breasts. Screening generally starts earlier in the United States and includes premenopausal women with denser breasts.
A recent study from Korea, where many women have dense breasts, found that 2D mammography and supplementary ultrasound outperformed AI for cancer detection.
“This underscores the challenges of finding cancers in dense breasts, which plague both radiologists and AI alike, and provides evidence that breast density is an important factor to consider when evaluating AI performance,” Dr. Philpotts said.
The work was funded by Lunit, the maker of the AI program used in the study. The investigators and Dr. Philpotts had no disclosures.
, a radiology and biomedical imaging professor at Yale University in New Haven, Conn.
The reason is because AI is proving to be as good as humans in interpreting mammograms, at least in the research setting.
In one of the latest reports, published online in Radiology, British investigators found that the performance of a commercially available AI system (INSIGHT MMG version 1.1.7.1 – Lunit) was essentially equivalent to over 500 specialized readers. The results are in line with other recent AI studies.
Double reading – having mammograms read by two clinicians to increase cancer detection rates – is common in the United Kingdom and elsewhere in Europe.
The British team compared the performance of 552 readers with Lunit’s AI program on the Personal Performance in Mammographic Screening exam, a quality assurance test which mammogram readers in the United Kingdom are required to take twice a year. Readers assign a malignancy score to 60 challenging cases, a mix of normal breasts and breasts with benign and cancerous lesions. The study included two test sessions for a total of 120 breast screenings.
Fifty-seven percent of the readers in the study were board-certified radiologists, 37% were radiographers, and 6% were breast clinicians. Each read at least 5,000 mammograms a year.
There was no difference in overall performance between the AI program and the human readers (AUC 0.93 vs. 0.88, P = .15).
Commenting in an editorial published with the investigation, Dr. Philpotts said the results “suggest that AI could confidently act as a second reader to decrease workloads.”
As for the United States, where double reading is generally not done, she pointed out that “many U.S. radiologists interpreting mammograms are nonspecialized and do not read high volumes of mammograms. Thus, the AI system evaluated in the study “could be used as a supplemental tool to aid the performance of readers in the United States or in other countries where screening programs use a single reading.”
There was also no difference in sensitivity between AI and human readers (84% vs. 90%, P = .34), but the AI algorithm had a higher specificity (89% vs. 76%, P = .003).
Using AI recall scores that matched the average human reader performance (90% sensitivity, 76% specificity), there was no difference with AI in regard to sensitivity (91%, P = .73) or specificity (77%, P = .85), but the investigators noted the power of the analysis was limited.
Overall, “diagnostic performance of AI was comparable with that of the average human reader.” It seems “increasingly likely that AI will eventually play a part in the interpretation of screening mammograms,” said investigators led by Yan Chen, PhD, of the Nottingham Breast Institute in England.
“That the AI system was able to match the performance of the average reader in this specialized group of mammogram readers indicates the robustness of this AI algorithm,” Dr. Philpotts said.
However, there are some caveats.
For one, the system was designed for 2D mammography, the current standard of care in the United Kingdom, while digital breast tomosynthesis (DBT) is replacing 2D mammography in the United States.
In the United States, “AI algorithms specific to DBT are necessary and will need to be reliable and reproducible to be embraced by radiologists,” Dr. Philpotts said.
Also in the United Kingdom, screening is performed at 3-year intervals in women aged 50-70 years old, which means that the study population was enriched for older women with less-dense breasts. Screening generally starts earlier in the United States and includes premenopausal women with denser breasts.
A recent study from Korea, where many women have dense breasts, found that 2D mammography and supplementary ultrasound outperformed AI for cancer detection.
“This underscores the challenges of finding cancers in dense breasts, which plague both radiologists and AI alike, and provides evidence that breast density is an important factor to consider when evaluating AI performance,” Dr. Philpotts said.
The work was funded by Lunit, the maker of the AI program used in the study. The investigators and Dr. Philpotts had no disclosures.
FROM RADIOLOGY
New European restrictions on topiramate in pregnancy
While it’s well known that topiramate can cause major congenital malformations and fetal growth restriction when used during pregnancy, recent data also suggest a possibly increased risk for neurodevelopmental disorders when topiramate is used during pregnancy, the EMA said in a statement.
The data include two observational studies that showed children born to mothers with epilepsy and who were exposed to topiramate in the womb may have a two- to threefold higher risk for neurodevelopmental disorders, in particular autism spectrum disorders (ASD), intellectual disability, or attention deficit hyperactivity disorder (ADHD), compared with children born to mothers with epilepsy not taking antiepileptic medication.
For patients using topiramate for the treatment of epilepsy, the PRAC now recommends that the medicine not be used during pregnancy unless no other suitable treatment is available.
The PRAC had also recommended a pregnancy prevention program to avoid exposure of the developing fetus to topiramate. “These measures will inform any woman or girl who is able to have children of the risks of taking topiramate during pregnancy and the need to avoid becoming pregnant while taking topiramate,” the EMA said.
Regardless of indication, the agency said topiramate should be used in women of childbearing age only when the following conditions of the pregnancy prevention program are met:
- A pregnancy test before starting treatment.
- Counseling about the risks of topiramate treatment and the need for highly effective contraception throughout treatment.
- A review of ongoing treatment at least annually by completion of a risk awareness form.
The PRAC recommends that health care professionals ensure women of childbearing age are fully aware of the risks of taking topiramate during pregnancy. The committee noted that alternative treatment options should be considered and the need for topiramate treatment should be reassessed at least annually.
The product information for topiramate-containing medicines will be updated to further highlight the risks for neurodevelopmental disorders and the additional safety measures to be taken.
Patients and health care professionals will be provided with educational materials regarding the risks of using topiramate during pregnancy, and a patient card will be provided to the patient with each medicine package. A visible warning will also be added to the outer packaging of the medicine.
The new PRAC recommendations will be sent to the Coordination Group for Mutual Recognition and Decentralised Procedures – Human (CMDh), which will adopt a position.
A version of this article first appeared on Medscape.com.
While it’s well known that topiramate can cause major congenital malformations and fetal growth restriction when used during pregnancy, recent data also suggest a possibly increased risk for neurodevelopmental disorders when topiramate is used during pregnancy, the EMA said in a statement.
The data include two observational studies that showed children born to mothers with epilepsy and who were exposed to topiramate in the womb may have a two- to threefold higher risk for neurodevelopmental disorders, in particular autism spectrum disorders (ASD), intellectual disability, or attention deficit hyperactivity disorder (ADHD), compared with children born to mothers with epilepsy not taking antiepileptic medication.
For patients using topiramate for the treatment of epilepsy, the PRAC now recommends that the medicine not be used during pregnancy unless no other suitable treatment is available.
The PRAC had also recommended a pregnancy prevention program to avoid exposure of the developing fetus to topiramate. “These measures will inform any woman or girl who is able to have children of the risks of taking topiramate during pregnancy and the need to avoid becoming pregnant while taking topiramate,” the EMA said.
Regardless of indication, the agency said topiramate should be used in women of childbearing age only when the following conditions of the pregnancy prevention program are met:
- A pregnancy test before starting treatment.
- Counseling about the risks of topiramate treatment and the need for highly effective contraception throughout treatment.
- A review of ongoing treatment at least annually by completion of a risk awareness form.
The PRAC recommends that health care professionals ensure women of childbearing age are fully aware of the risks of taking topiramate during pregnancy. The committee noted that alternative treatment options should be considered and the need for topiramate treatment should be reassessed at least annually.
The product information for topiramate-containing medicines will be updated to further highlight the risks for neurodevelopmental disorders and the additional safety measures to be taken.
Patients and health care professionals will be provided with educational materials regarding the risks of using topiramate during pregnancy, and a patient card will be provided to the patient with each medicine package. A visible warning will also be added to the outer packaging of the medicine.
The new PRAC recommendations will be sent to the Coordination Group for Mutual Recognition and Decentralised Procedures – Human (CMDh), which will adopt a position.
A version of this article first appeared on Medscape.com.
While it’s well known that topiramate can cause major congenital malformations and fetal growth restriction when used during pregnancy, recent data also suggest a possibly increased risk for neurodevelopmental disorders when topiramate is used during pregnancy, the EMA said in a statement.
The data include two observational studies that showed children born to mothers with epilepsy and who were exposed to topiramate in the womb may have a two- to threefold higher risk for neurodevelopmental disorders, in particular autism spectrum disorders (ASD), intellectual disability, or attention deficit hyperactivity disorder (ADHD), compared with children born to mothers with epilepsy not taking antiepileptic medication.
For patients using topiramate for the treatment of epilepsy, the PRAC now recommends that the medicine not be used during pregnancy unless no other suitable treatment is available.
The PRAC had also recommended a pregnancy prevention program to avoid exposure of the developing fetus to topiramate. “These measures will inform any woman or girl who is able to have children of the risks of taking topiramate during pregnancy and the need to avoid becoming pregnant while taking topiramate,” the EMA said.
Regardless of indication, the agency said topiramate should be used in women of childbearing age only when the following conditions of the pregnancy prevention program are met:
- A pregnancy test before starting treatment.
- Counseling about the risks of topiramate treatment and the need for highly effective contraception throughout treatment.
- A review of ongoing treatment at least annually by completion of a risk awareness form.
The PRAC recommends that health care professionals ensure women of childbearing age are fully aware of the risks of taking topiramate during pregnancy. The committee noted that alternative treatment options should be considered and the need for topiramate treatment should be reassessed at least annually.
The product information for topiramate-containing medicines will be updated to further highlight the risks for neurodevelopmental disorders and the additional safety measures to be taken.
Patients and health care professionals will be provided with educational materials regarding the risks of using topiramate during pregnancy, and a patient card will be provided to the patient with each medicine package. A visible warning will also be added to the outer packaging of the medicine.
The new PRAC recommendations will be sent to the Coordination Group for Mutual Recognition and Decentralised Procedures – Human (CMDh), which will adopt a position.
A version of this article first appeared on Medscape.com.
New Moderna vaccine to work against recent COVID variant
“The company said its shot generated an 8.7-fold increase in neutralizing antibodies in humans against BA.2.86, which is being tracked by the World Health Organization and the U.S. Centers for Disease Control and Prevention,” Reuters reported.
“We think this is news people will want to hear as they prepare to go out and get their fall boosters,” Jacqueline Miller, Moderna head of infectious diseases, told the news agency.
The CDC said that the BA.2.86 variant might be more likely to infect people who have already had COVID or previous vaccinations. BA.2.86 is an Omicron variant. It has undergone more mutations than XBB.1.5, which has dominated most of this year and was the intended target of the updated shots.
BA.2.86 does not have a strong presence in the United States yet. However, officials are concerned about its high number of mutations, NBC News reported.
The FDA is expected to approve the new Moderna shot by early October.
Pfizer told NBC that its updated booster also generated a strong antibody response against Omicron variants, including BA.2.86.
COVID-19 cases and hospitalizations have been increasing in the U.S. because of the rise of several variants.
Experts told Reuters that BA.2.86 probably won’t cause a wave of severe disease and death because immunity has been built up around the world through previous infections and mass vaccinations.
A version of this article appeared on WebMD.com.
“The company said its shot generated an 8.7-fold increase in neutralizing antibodies in humans against BA.2.86, which is being tracked by the World Health Organization and the U.S. Centers for Disease Control and Prevention,” Reuters reported.
“We think this is news people will want to hear as they prepare to go out and get their fall boosters,” Jacqueline Miller, Moderna head of infectious diseases, told the news agency.
The CDC said that the BA.2.86 variant might be more likely to infect people who have already had COVID or previous vaccinations. BA.2.86 is an Omicron variant. It has undergone more mutations than XBB.1.5, which has dominated most of this year and was the intended target of the updated shots.
BA.2.86 does not have a strong presence in the United States yet. However, officials are concerned about its high number of mutations, NBC News reported.
The FDA is expected to approve the new Moderna shot by early October.
Pfizer told NBC that its updated booster also generated a strong antibody response against Omicron variants, including BA.2.86.
COVID-19 cases and hospitalizations have been increasing in the U.S. because of the rise of several variants.
Experts told Reuters that BA.2.86 probably won’t cause a wave of severe disease and death because immunity has been built up around the world through previous infections and mass vaccinations.
A version of this article appeared on WebMD.com.
“The company said its shot generated an 8.7-fold increase in neutralizing antibodies in humans against BA.2.86, which is being tracked by the World Health Organization and the U.S. Centers for Disease Control and Prevention,” Reuters reported.
“We think this is news people will want to hear as they prepare to go out and get their fall boosters,” Jacqueline Miller, Moderna head of infectious diseases, told the news agency.
The CDC said that the BA.2.86 variant might be more likely to infect people who have already had COVID or previous vaccinations. BA.2.86 is an Omicron variant. It has undergone more mutations than XBB.1.5, which has dominated most of this year and was the intended target of the updated shots.
BA.2.86 does not have a strong presence in the United States yet. However, officials are concerned about its high number of mutations, NBC News reported.
The FDA is expected to approve the new Moderna shot by early October.
Pfizer told NBC that its updated booster also generated a strong antibody response against Omicron variants, including BA.2.86.
COVID-19 cases and hospitalizations have been increasing in the U.S. because of the rise of several variants.
Experts told Reuters that BA.2.86 probably won’t cause a wave of severe disease and death because immunity has been built up around the world through previous infections and mass vaccinations.
A version of this article appeared on WebMD.com.
Ketogenic diet short-term may benefit women with PCOS
Ketogenic diets may improve reproductive hormone levels in women with polycystic ovary syndrome (PCOS), new research suggests.
In the first-ever systematic review and meta-analysis of clinical trials on the association, ketogenic diets followed for 45 days to 24 weeks showed improvements in the luteinizing hormone (LH)/follicle-stimulating hormone (FSH) ratio, serum free testosterone, and serum sex hormone binding globulin (SHBG).
Previous evidence supporting ketogenic diets in PCOS has been “relatively patchy,” and although there have been reviews on the topic, this is the first meta-analysis, write Karniza Khalid, MD, of the National Institutes of Health, Ministry of Health Malaysia, and colleagues.
Study co-author Syed A.A. Rizvi, MD, PhD, told this news organization: “Our paper supports the positive effects of short-term ketogenic diets on hormonal imbalances commonly associated with PCOS, a complex disease state associated with a multitude of presenting symptoms among individuals. Based on the presentation and individual patient circumstances, besides pharmacologic treatment, lifestyle changes and a ketogenic diet can lead to even faster improvements.”
However, Dr. Rizvi, a professor at the College of Biomedical Sciences, Larkin University, Miami, cautioned: “I would highly recommend a keto diet to women suffering from PCOS, but we all know every person has a different situation. Some may not want to change their diet, some may not be able to afford it, and for some it is just too much work. ... This is why any lifestyle change has to be discussed and planned carefully between patients and their health care providers.”
The findings were published online in the Journal of the Endocrine Society.
The literature search yielded seven qualifying studies of ketogenic diets, generally defined as a daily carbohydrate intake below 50 g while allowing variable amounts of fat and protein. A total of 170 participants were enrolled in the studies from Italy, China, and the United States.
Pooled data showed a significant association between ketogenic diet and reduced LH/FSH ratio (P < .001) and free testosterone (P < .001). There was also a significant increase in circulating SHBG (P = .002).
On the other hand, serum progesterone levels did not change significantly (P = .353).
Weight loss, a secondary outcome, was significantly greater with the ketogenic diet (P < .001).
“Since low-carbohydrate diets have shown to be effective in addressing obesity and type 2 diabetes, it makes sense that they would also be helpful to the patients with PCOS, and in fact, it has been the case,” Dr. Rizvi noted.
The exact mechanisms for the hormonal effects aren’t clear, but one theory is that the reduction in hyperinsulinemia from the ketogenic diet decreases stimulation of ovarian androgen production and increases SHBG levels. Another is that the physiologic ketosis induced by low carbohydrate intake reduces both circulating insulin and insulin-like growth factor-1, thereby suppressing the stimulus on the production of both ovarian and adrenal androgens.
The analysis didn’t include pregnancy rates. However, Dr. Rizvi noted, “there have been published studies showing that [patients with] PCOS on keto diets have significantly improved pregnancy rates, also including via [in vitro fertilization].”
The study received no outside funding. The authors have reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Ketogenic diets may improve reproductive hormone levels in women with polycystic ovary syndrome (PCOS), new research suggests.
In the first-ever systematic review and meta-analysis of clinical trials on the association, ketogenic diets followed for 45 days to 24 weeks showed improvements in the luteinizing hormone (LH)/follicle-stimulating hormone (FSH) ratio, serum free testosterone, and serum sex hormone binding globulin (SHBG).
Previous evidence supporting ketogenic diets in PCOS has been “relatively patchy,” and although there have been reviews on the topic, this is the first meta-analysis, write Karniza Khalid, MD, of the National Institutes of Health, Ministry of Health Malaysia, and colleagues.
Study co-author Syed A.A. Rizvi, MD, PhD, told this news organization: “Our paper supports the positive effects of short-term ketogenic diets on hormonal imbalances commonly associated with PCOS, a complex disease state associated with a multitude of presenting symptoms among individuals. Based on the presentation and individual patient circumstances, besides pharmacologic treatment, lifestyle changes and a ketogenic diet can lead to even faster improvements.”
However, Dr. Rizvi, a professor at the College of Biomedical Sciences, Larkin University, Miami, cautioned: “I would highly recommend a keto diet to women suffering from PCOS, but we all know every person has a different situation. Some may not want to change their diet, some may not be able to afford it, and for some it is just too much work. ... This is why any lifestyle change has to be discussed and planned carefully between patients and their health care providers.”
The findings were published online in the Journal of the Endocrine Society.
The literature search yielded seven qualifying studies of ketogenic diets, generally defined as a daily carbohydrate intake below 50 g while allowing variable amounts of fat and protein. A total of 170 participants were enrolled in the studies from Italy, China, and the United States.
Pooled data showed a significant association between ketogenic diet and reduced LH/FSH ratio (P < .001) and free testosterone (P < .001). There was also a significant increase in circulating SHBG (P = .002).
On the other hand, serum progesterone levels did not change significantly (P = .353).
Weight loss, a secondary outcome, was significantly greater with the ketogenic diet (P < .001).
“Since low-carbohydrate diets have shown to be effective in addressing obesity and type 2 diabetes, it makes sense that they would also be helpful to the patients with PCOS, and in fact, it has been the case,” Dr. Rizvi noted.
The exact mechanisms for the hormonal effects aren’t clear, but one theory is that the reduction in hyperinsulinemia from the ketogenic diet decreases stimulation of ovarian androgen production and increases SHBG levels. Another is that the physiologic ketosis induced by low carbohydrate intake reduces both circulating insulin and insulin-like growth factor-1, thereby suppressing the stimulus on the production of both ovarian and adrenal androgens.
The analysis didn’t include pregnancy rates. However, Dr. Rizvi noted, “there have been published studies showing that [patients with] PCOS on keto diets have significantly improved pregnancy rates, also including via [in vitro fertilization].”
The study received no outside funding. The authors have reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Ketogenic diets may improve reproductive hormone levels in women with polycystic ovary syndrome (PCOS), new research suggests.
In the first-ever systematic review and meta-analysis of clinical trials on the association, ketogenic diets followed for 45 days to 24 weeks showed improvements in the luteinizing hormone (LH)/follicle-stimulating hormone (FSH) ratio, serum free testosterone, and serum sex hormone binding globulin (SHBG).
Previous evidence supporting ketogenic diets in PCOS has been “relatively patchy,” and although there have been reviews on the topic, this is the first meta-analysis, write Karniza Khalid, MD, of the National Institutes of Health, Ministry of Health Malaysia, and colleagues.
Study co-author Syed A.A. Rizvi, MD, PhD, told this news organization: “Our paper supports the positive effects of short-term ketogenic diets on hormonal imbalances commonly associated with PCOS, a complex disease state associated with a multitude of presenting symptoms among individuals. Based on the presentation and individual patient circumstances, besides pharmacologic treatment, lifestyle changes and a ketogenic diet can lead to even faster improvements.”
However, Dr. Rizvi, a professor at the College of Biomedical Sciences, Larkin University, Miami, cautioned: “I would highly recommend a keto diet to women suffering from PCOS, but we all know every person has a different situation. Some may not want to change their diet, some may not be able to afford it, and for some it is just too much work. ... This is why any lifestyle change has to be discussed and planned carefully between patients and their health care providers.”
The findings were published online in the Journal of the Endocrine Society.
The literature search yielded seven qualifying studies of ketogenic diets, generally defined as a daily carbohydrate intake below 50 g while allowing variable amounts of fat and protein. A total of 170 participants were enrolled in the studies from Italy, China, and the United States.
Pooled data showed a significant association between ketogenic diet and reduced LH/FSH ratio (P < .001) and free testosterone (P < .001). There was also a significant increase in circulating SHBG (P = .002).
On the other hand, serum progesterone levels did not change significantly (P = .353).
Weight loss, a secondary outcome, was significantly greater with the ketogenic diet (P < .001).
“Since low-carbohydrate diets have shown to be effective in addressing obesity and type 2 diabetes, it makes sense that they would also be helpful to the patients with PCOS, and in fact, it has been the case,” Dr. Rizvi noted.
The exact mechanisms for the hormonal effects aren’t clear, but one theory is that the reduction in hyperinsulinemia from the ketogenic diet decreases stimulation of ovarian androgen production and increases SHBG levels. Another is that the physiologic ketosis induced by low carbohydrate intake reduces both circulating insulin and insulin-like growth factor-1, thereby suppressing the stimulus on the production of both ovarian and adrenal androgens.
The analysis didn’t include pregnancy rates. However, Dr. Rizvi noted, “there have been published studies showing that [patients with] PCOS on keto diets have significantly improved pregnancy rates, also including via [in vitro fertilization].”
The study received no outside funding. The authors have reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Treating fractures in elderly patients: Beyond the broken bone
While half the fracture-prevention battle is getting people diagnosed with low bone density, nearly 80% of older Americans who suffer bone breaks are not tested or treated for osteoporosis. Fractures associated with aging and diminished bone mineral density exact an enormous toll on patients’ lives and cost the health care system billions of dollars annually according to Bone Health and Osteoporosis: A Report of the Surgeon General. But current gaps in patient education and bone density screening are huge.
“It’s concerning that older patients at risk for fracture are often not screened to determine their risk factors contributing to osteoporosis and patients are not educated about fracture prevention,” said Meryl S. LeBoff, MD, an endocrinologist at Brigham and Women’s Hospital, and chief of calcium and bone section, and professor of medicine, at Harvard Medical School, Boston. “Furthermore, the majority of highest-risk women and men who do have fractures are not screened and they do not receive effective, [Food and Drug Administration]–approved therapies.”
Recent guidelines
Screening with dual-energy x-ray absorptiometry (DEXA) is recommended for all women at age 65 and all men at age 70. But the occasion of a fracture in an older person who has not yet met these age thresholds should prompt a bone density assessment.
“Doctors need to stress that one in two women and one in four men over age 50 will have a fracture in their remaining lifetimes,” Dr. LeBoff said. ”Primary care doctors play a critical role in ordering timely bone densitometry for both sexes.
If an older patient has been treated for a fracture, the main goal going forward is to prevent another one, for which the risk is highest in the 2 years after the incident fracture.”
According to Kendall F. Moseley, MD, clinical director of the division of endocrinology, diabetes & metabolism at Johns Hopkins Medicine in Baltimore, “Elderly patients need to understand that a fracture at their age is like a heart attack of the bone,” she said, adding that just as cardiovascular risk factors such as high blood pressure and blood lipids are silent before a stroke or infarction, the bone thinning of old age is also silent.
Endocrinologist Jennifer J. Kelly, DO, director of the metabolic bone program and an associate professor at the University of Vermont Medical Center in Burlington, said a fracture in anyone over age 50 that appears not to have resulted from a traumatic blow, is a compelling reason to order a DEXA exam.
Nahid J. Rianon, MBBS/MD, DrPH, assistant professor of the division of geriatric medicine at the UTHealth McGovern Medical School, Houston, goes further: “Any fracture in someone age 50 and older warrants screening for osteoporosis. And if the fracture is nontraumatic, that is by definition a clinical diagnosis of osteoporosis regardless of normal results on bone density tests and they should be treated medically. There are aspects of bone that we still can’t measure in the clinical setting.”
If DEXA is not accessible, fracture risk over the next 10 years can be evaluated based on multiple patient characteristics and medical history using the online FRAX calculator.
Just a 3% risk of hip fracture on FRAX is considered an indication to begin medical osteoporosis treatment in the United States regardless of bone density test results, Dr. Rianon said.
Fracture management
Whether a senior suffers a traumatic fracture or an osteoporosis-related fragility fracture, older age can impede the healing process in some. Senescence may also increase systemic proinflammatory status, according to Clark and colleagues, writing in Current Osteoporosis Reports.
They called for research to develop more directed treatment options for the elderly population.
Dr. Rianon noted that healing may also be affected by a decrease in muscle mass, which plays a role in holding the bone in place. “But it is still controversial how changing metabolic factors affect bone healing in the elderly.”
However, countered Dr. Kelly, fractures in elderly patients are not necessarily less likely to mend – if osteoporosis is not present. “Many heal very well – it really depends more upon their overall health and medical history. Whether or not a person requires surgery depends more upon the extent of the fracture and if the bone is able to align and heal appropriately without surgery.”
Fracture sites
Spine. According to the American Academy of Orthopedic Surgeons the earliest and most frequent site of fragility fractures in the elderly is the spine. Most vertebral fracture pain improves within 3 months without specific treatment. A short period of rest, limited analgesic use, and possible back bracing may help as the fractures heal on their own. But if pain is severe and persistent, vertebral augmentation with percutaneous kyphoplasty or vertebroplasty may be an option. These procedures, however, can destabilize surrounding discs because of the greater thickness of the injected cement.
Hip. The most dangerous fractures occur in the hip. These carry at least a 20% risk of death in the first postoperative year and must be treated surgically. Those in the proximal femur, the head, or the femoral neck will usually need hip replacement, but if the break is farther down, it may be repaired with cement, screws, plates, and rods.
Distal radius. Outcomes of wrist fractures may be positive without surgical intervention, according to a recent retrospective analysis from Turkey by Yalin and colleagues. In a comparison of clinical outcomes in seniors aged 70-89 and assigned to cast immobilization or various surgical treatments for distal radius fractures, no statistically significant difference was found in patient-reported disability scores and range of motion values between casting and surgery in the first postoperative year.
Other sites. Fractures in the elderly are not uncommon in the shoulder, distal radius, cubitus, proximal humerus, and humerus. These fractures are often treated without surgery, but nevertheless signal a high risk for additional fractures.
Bone-enhancing medications
Even in the absence of diagnosed low bone density or osteoporosis, anabolic agents such as the synthetic human parathyroid hormones abaloparatide (Tymlos) and teriparatide (Forteo) may be used to help in some cases with a bad healing prognosis and may also be used for people undergoing surgeries such as a spinal fusion, but there are not clinical guidelines. “We receive referrals regularly for this treatment from our orthopedics colleagues, but it is considered an off-label use,” Dr. Kelly said.
The anabolics teriparatide and romosozumab (Evenity) have proved effective in lowering fractures in high-risk older women.
Post fracture
After recovering from a fracture, elderly people are strongly advised to make lifestyle changes to boost bone health and reduce risk of further fractures, said Willy M. Valencia, MD, a geriatrician-endocrinologist at the Cleveland Clinic. Apart from active daily living, he recommends several types of formal exercise to promote bone formation; increase muscle mass, strength, and flexibility; and improve endurance, balance, and gait. The National Institute on Aging outlines suitable exercise programs for seniors.
“These exercises will help reduce the risk of falling and to avoid more fractures,” he said. “Whether a patient has been exercising before the fracture or not, they may feel some reticence or reluctance to take up exercise afterwards because they’re afraid of having another fracture, but they should understand that their fracture risk increases if they remain sedentary. They should start slowly but they can’t be sitting all day.”
Even before it’s possible to exercise at the healing fracture site, added Dr. Rianon, its advisable to work other areas of the body. “Overall mobility is important, and exercising other parts of the body can stimulate strength and help prevent falling.”
In other postsurgical measures, a bone-friendly diet rich in calcium and vitamin D, as well as supplementation with these vital nutrients, is essential to lower the risk of falling.
Fall prevention is paramount, said Dr. Valencia. While exercise can improve, gait, balance, and endurance, logistical measures may also be necessary. Seniors may have to move to a one-floor domicile with no stairs to negotiate. At the very least, they need to fall-proof their daily lives by upgrading their eyeglasses and home lighting, eliminating obstacles and loose carpets, fixing bannisters, and installing bathroom handrails. Some may need assistive devices for walking, especially outdoors in slippery conditions.
At the end of the day, the role of the primary physician in screening for bone problems before fracture and postsurgical care is key. “Risk factors for osteoporosis and fracture risk must be added to the patient’s chart,” said Dr. Rianon. Added Dr. Moseley. “No matter how busy they are, my hope is that primary care physicians will not put patients’ bone health at the bottom of the clinical agenda.”
While half the fracture-prevention battle is getting people diagnosed with low bone density, nearly 80% of older Americans who suffer bone breaks are not tested or treated for osteoporosis. Fractures associated with aging and diminished bone mineral density exact an enormous toll on patients’ lives and cost the health care system billions of dollars annually according to Bone Health and Osteoporosis: A Report of the Surgeon General. But current gaps in patient education and bone density screening are huge.
“It’s concerning that older patients at risk for fracture are often not screened to determine their risk factors contributing to osteoporosis and patients are not educated about fracture prevention,” said Meryl S. LeBoff, MD, an endocrinologist at Brigham and Women’s Hospital, and chief of calcium and bone section, and professor of medicine, at Harvard Medical School, Boston. “Furthermore, the majority of highest-risk women and men who do have fractures are not screened and they do not receive effective, [Food and Drug Administration]–approved therapies.”
Recent guidelines
Screening with dual-energy x-ray absorptiometry (DEXA) is recommended for all women at age 65 and all men at age 70. But the occasion of a fracture in an older person who has not yet met these age thresholds should prompt a bone density assessment.
“Doctors need to stress that one in two women and one in four men over age 50 will have a fracture in their remaining lifetimes,” Dr. LeBoff said. ”Primary care doctors play a critical role in ordering timely bone densitometry for both sexes.
If an older patient has been treated for a fracture, the main goal going forward is to prevent another one, for which the risk is highest in the 2 years after the incident fracture.”
According to Kendall F. Moseley, MD, clinical director of the division of endocrinology, diabetes & metabolism at Johns Hopkins Medicine in Baltimore, “Elderly patients need to understand that a fracture at their age is like a heart attack of the bone,” she said, adding that just as cardiovascular risk factors such as high blood pressure and blood lipids are silent before a stroke or infarction, the bone thinning of old age is also silent.
Endocrinologist Jennifer J. Kelly, DO, director of the metabolic bone program and an associate professor at the University of Vermont Medical Center in Burlington, said a fracture in anyone over age 50 that appears not to have resulted from a traumatic blow, is a compelling reason to order a DEXA exam.
Nahid J. Rianon, MBBS/MD, DrPH, assistant professor of the division of geriatric medicine at the UTHealth McGovern Medical School, Houston, goes further: “Any fracture in someone age 50 and older warrants screening for osteoporosis. And if the fracture is nontraumatic, that is by definition a clinical diagnosis of osteoporosis regardless of normal results on bone density tests and they should be treated medically. There are aspects of bone that we still can’t measure in the clinical setting.”
If DEXA is not accessible, fracture risk over the next 10 years can be evaluated based on multiple patient characteristics and medical history using the online FRAX calculator.
Just a 3% risk of hip fracture on FRAX is considered an indication to begin medical osteoporosis treatment in the United States regardless of bone density test results, Dr. Rianon said.
Fracture management
Whether a senior suffers a traumatic fracture or an osteoporosis-related fragility fracture, older age can impede the healing process in some. Senescence may also increase systemic proinflammatory status, according to Clark and colleagues, writing in Current Osteoporosis Reports.
They called for research to develop more directed treatment options for the elderly population.
Dr. Rianon noted that healing may also be affected by a decrease in muscle mass, which plays a role in holding the bone in place. “But it is still controversial how changing metabolic factors affect bone healing in the elderly.”
However, countered Dr. Kelly, fractures in elderly patients are not necessarily less likely to mend – if osteoporosis is not present. “Many heal very well – it really depends more upon their overall health and medical history. Whether or not a person requires surgery depends more upon the extent of the fracture and if the bone is able to align and heal appropriately without surgery.”
Fracture sites
Spine. According to the American Academy of Orthopedic Surgeons the earliest and most frequent site of fragility fractures in the elderly is the spine. Most vertebral fracture pain improves within 3 months without specific treatment. A short period of rest, limited analgesic use, and possible back bracing may help as the fractures heal on their own. But if pain is severe and persistent, vertebral augmentation with percutaneous kyphoplasty or vertebroplasty may be an option. These procedures, however, can destabilize surrounding discs because of the greater thickness of the injected cement.
Hip. The most dangerous fractures occur in the hip. These carry at least a 20% risk of death in the first postoperative year and must be treated surgically. Those in the proximal femur, the head, or the femoral neck will usually need hip replacement, but if the break is farther down, it may be repaired with cement, screws, plates, and rods.
Distal radius. Outcomes of wrist fractures may be positive without surgical intervention, according to a recent retrospective analysis from Turkey by Yalin and colleagues. In a comparison of clinical outcomes in seniors aged 70-89 and assigned to cast immobilization or various surgical treatments for distal radius fractures, no statistically significant difference was found in patient-reported disability scores and range of motion values between casting and surgery in the first postoperative year.
Other sites. Fractures in the elderly are not uncommon in the shoulder, distal radius, cubitus, proximal humerus, and humerus. These fractures are often treated without surgery, but nevertheless signal a high risk for additional fractures.
Bone-enhancing medications
Even in the absence of diagnosed low bone density or osteoporosis, anabolic agents such as the synthetic human parathyroid hormones abaloparatide (Tymlos) and teriparatide (Forteo) may be used to help in some cases with a bad healing prognosis and may also be used for people undergoing surgeries such as a spinal fusion, but there are not clinical guidelines. “We receive referrals regularly for this treatment from our orthopedics colleagues, but it is considered an off-label use,” Dr. Kelly said.
The anabolics teriparatide and romosozumab (Evenity) have proved effective in lowering fractures in high-risk older women.
Post fracture
After recovering from a fracture, elderly people are strongly advised to make lifestyle changes to boost bone health and reduce risk of further fractures, said Willy M. Valencia, MD, a geriatrician-endocrinologist at the Cleveland Clinic. Apart from active daily living, he recommends several types of formal exercise to promote bone formation; increase muscle mass, strength, and flexibility; and improve endurance, balance, and gait. The National Institute on Aging outlines suitable exercise programs for seniors.
“These exercises will help reduce the risk of falling and to avoid more fractures,” he said. “Whether a patient has been exercising before the fracture or not, they may feel some reticence or reluctance to take up exercise afterwards because they’re afraid of having another fracture, but they should understand that their fracture risk increases if they remain sedentary. They should start slowly but they can’t be sitting all day.”
Even before it’s possible to exercise at the healing fracture site, added Dr. Rianon, its advisable to work other areas of the body. “Overall mobility is important, and exercising other parts of the body can stimulate strength and help prevent falling.”
In other postsurgical measures, a bone-friendly diet rich in calcium and vitamin D, as well as supplementation with these vital nutrients, is essential to lower the risk of falling.
Fall prevention is paramount, said Dr. Valencia. While exercise can improve, gait, balance, and endurance, logistical measures may also be necessary. Seniors may have to move to a one-floor domicile with no stairs to negotiate. At the very least, they need to fall-proof their daily lives by upgrading their eyeglasses and home lighting, eliminating obstacles and loose carpets, fixing bannisters, and installing bathroom handrails. Some may need assistive devices for walking, especially outdoors in slippery conditions.
At the end of the day, the role of the primary physician in screening for bone problems before fracture and postsurgical care is key. “Risk factors for osteoporosis and fracture risk must be added to the patient’s chart,” said Dr. Rianon. Added Dr. Moseley. “No matter how busy they are, my hope is that primary care physicians will not put patients’ bone health at the bottom of the clinical agenda.”
While half the fracture-prevention battle is getting people diagnosed with low bone density, nearly 80% of older Americans who suffer bone breaks are not tested or treated for osteoporosis. Fractures associated with aging and diminished bone mineral density exact an enormous toll on patients’ lives and cost the health care system billions of dollars annually according to Bone Health and Osteoporosis: A Report of the Surgeon General. But current gaps in patient education and bone density screening are huge.
“It’s concerning that older patients at risk for fracture are often not screened to determine their risk factors contributing to osteoporosis and patients are not educated about fracture prevention,” said Meryl S. LeBoff, MD, an endocrinologist at Brigham and Women’s Hospital, and chief of calcium and bone section, and professor of medicine, at Harvard Medical School, Boston. “Furthermore, the majority of highest-risk women and men who do have fractures are not screened and they do not receive effective, [Food and Drug Administration]–approved therapies.”
Recent guidelines
Screening with dual-energy x-ray absorptiometry (DEXA) is recommended for all women at age 65 and all men at age 70. But the occasion of a fracture in an older person who has not yet met these age thresholds should prompt a bone density assessment.
“Doctors need to stress that one in two women and one in four men over age 50 will have a fracture in their remaining lifetimes,” Dr. LeBoff said. ”Primary care doctors play a critical role in ordering timely bone densitometry for both sexes.
If an older patient has been treated for a fracture, the main goal going forward is to prevent another one, for which the risk is highest in the 2 years after the incident fracture.”
According to Kendall F. Moseley, MD, clinical director of the division of endocrinology, diabetes & metabolism at Johns Hopkins Medicine in Baltimore, “Elderly patients need to understand that a fracture at their age is like a heart attack of the bone,” she said, adding that just as cardiovascular risk factors such as high blood pressure and blood lipids are silent before a stroke or infarction, the bone thinning of old age is also silent.
Endocrinologist Jennifer J. Kelly, DO, director of the metabolic bone program and an associate professor at the University of Vermont Medical Center in Burlington, said a fracture in anyone over age 50 that appears not to have resulted from a traumatic blow, is a compelling reason to order a DEXA exam.
Nahid J. Rianon, MBBS/MD, DrPH, assistant professor of the division of geriatric medicine at the UTHealth McGovern Medical School, Houston, goes further: “Any fracture in someone age 50 and older warrants screening for osteoporosis. And if the fracture is nontraumatic, that is by definition a clinical diagnosis of osteoporosis regardless of normal results on bone density tests and they should be treated medically. There are aspects of bone that we still can’t measure in the clinical setting.”
If DEXA is not accessible, fracture risk over the next 10 years can be evaluated based on multiple patient characteristics and medical history using the online FRAX calculator.
Just a 3% risk of hip fracture on FRAX is considered an indication to begin medical osteoporosis treatment in the United States regardless of bone density test results, Dr. Rianon said.
Fracture management
Whether a senior suffers a traumatic fracture or an osteoporosis-related fragility fracture, older age can impede the healing process in some. Senescence may also increase systemic proinflammatory status, according to Clark and colleagues, writing in Current Osteoporosis Reports.
They called for research to develop more directed treatment options for the elderly population.
Dr. Rianon noted that healing may also be affected by a decrease in muscle mass, which plays a role in holding the bone in place. “But it is still controversial how changing metabolic factors affect bone healing in the elderly.”
However, countered Dr. Kelly, fractures in elderly patients are not necessarily less likely to mend – if osteoporosis is not present. “Many heal very well – it really depends more upon their overall health and medical history. Whether or not a person requires surgery depends more upon the extent of the fracture and if the bone is able to align and heal appropriately without surgery.”
Fracture sites
Spine. According to the American Academy of Orthopedic Surgeons the earliest and most frequent site of fragility fractures in the elderly is the spine. Most vertebral fracture pain improves within 3 months without specific treatment. A short period of rest, limited analgesic use, and possible back bracing may help as the fractures heal on their own. But if pain is severe and persistent, vertebral augmentation with percutaneous kyphoplasty or vertebroplasty may be an option. These procedures, however, can destabilize surrounding discs because of the greater thickness of the injected cement.
Hip. The most dangerous fractures occur in the hip. These carry at least a 20% risk of death in the first postoperative year and must be treated surgically. Those in the proximal femur, the head, or the femoral neck will usually need hip replacement, but if the break is farther down, it may be repaired with cement, screws, plates, and rods.
Distal radius. Outcomes of wrist fractures may be positive without surgical intervention, according to a recent retrospective analysis from Turkey by Yalin and colleagues. In a comparison of clinical outcomes in seniors aged 70-89 and assigned to cast immobilization or various surgical treatments for distal radius fractures, no statistically significant difference was found in patient-reported disability scores and range of motion values between casting and surgery in the first postoperative year.
Other sites. Fractures in the elderly are not uncommon in the shoulder, distal radius, cubitus, proximal humerus, and humerus. These fractures are often treated without surgery, but nevertheless signal a high risk for additional fractures.
Bone-enhancing medications
Even in the absence of diagnosed low bone density or osteoporosis, anabolic agents such as the synthetic human parathyroid hormones abaloparatide (Tymlos) and teriparatide (Forteo) may be used to help in some cases with a bad healing prognosis and may also be used for people undergoing surgeries such as a spinal fusion, but there are not clinical guidelines. “We receive referrals regularly for this treatment from our orthopedics colleagues, but it is considered an off-label use,” Dr. Kelly said.
The anabolics teriparatide and romosozumab (Evenity) have proved effective in lowering fractures in high-risk older women.
Post fracture
After recovering from a fracture, elderly people are strongly advised to make lifestyle changes to boost bone health and reduce risk of further fractures, said Willy M. Valencia, MD, a geriatrician-endocrinologist at the Cleveland Clinic. Apart from active daily living, he recommends several types of formal exercise to promote bone formation; increase muscle mass, strength, and flexibility; and improve endurance, balance, and gait. The National Institute on Aging outlines suitable exercise programs for seniors.
“These exercises will help reduce the risk of falling and to avoid more fractures,” he said. “Whether a patient has been exercising before the fracture or not, they may feel some reticence or reluctance to take up exercise afterwards because they’re afraid of having another fracture, but they should understand that their fracture risk increases if they remain sedentary. They should start slowly but they can’t be sitting all day.”
Even before it’s possible to exercise at the healing fracture site, added Dr. Rianon, its advisable to work other areas of the body. “Overall mobility is important, and exercising other parts of the body can stimulate strength and help prevent falling.”
In other postsurgical measures, a bone-friendly diet rich in calcium and vitamin D, as well as supplementation with these vital nutrients, is essential to lower the risk of falling.
Fall prevention is paramount, said Dr. Valencia. While exercise can improve, gait, balance, and endurance, logistical measures may also be necessary. Seniors may have to move to a one-floor domicile with no stairs to negotiate. At the very least, they need to fall-proof their daily lives by upgrading their eyeglasses and home lighting, eliminating obstacles and loose carpets, fixing bannisters, and installing bathroom handrails. Some may need assistive devices for walking, especially outdoors in slippery conditions.
At the end of the day, the role of the primary physician in screening for bone problems before fracture and postsurgical care is key. “Risk factors for osteoporosis and fracture risk must be added to the patient’s chart,” said Dr. Rianon. Added Dr. Moseley. “No matter how busy they are, my hope is that primary care physicians will not put patients’ bone health at the bottom of the clinical agenda.”