User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
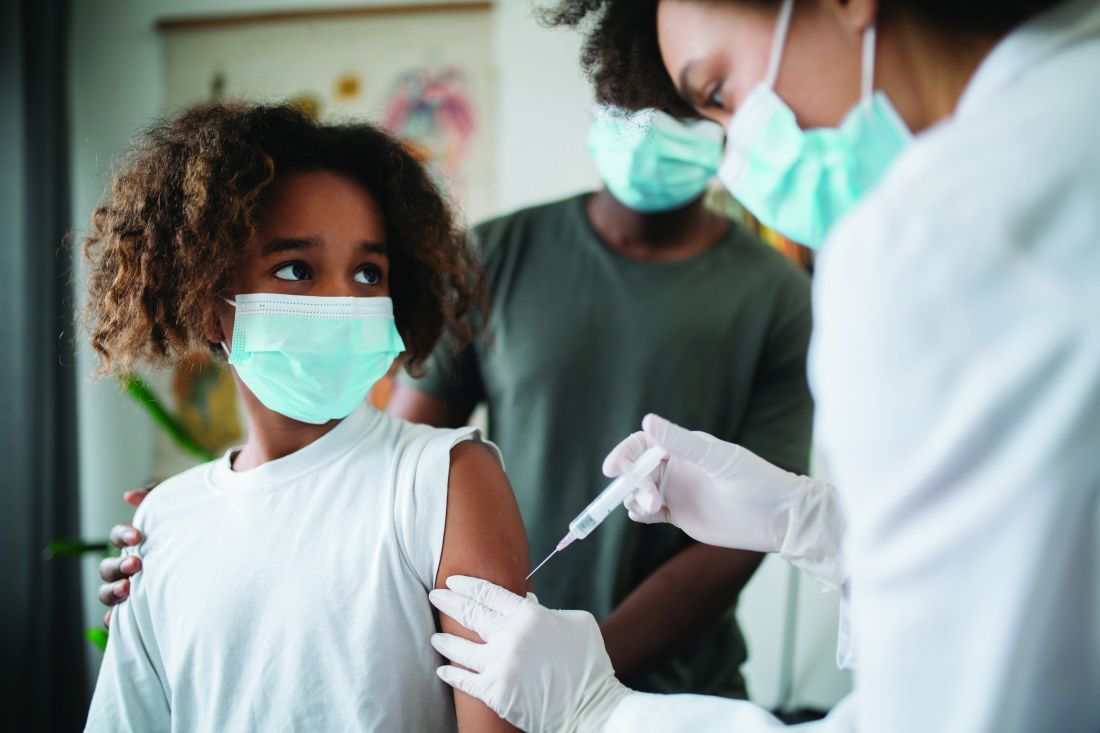
COVID cases spike as questions remain about Omicron’s threat
The best way to stay protected is by getting vaccinated and boosted, they said.
“For the unvaccinated, you’re looking at a winter of severe illness and death – for yourselves, families, and the hospitals who may soon overwhelm,” White House COVID-19 Response Coordinator Jeff Zients said at a news briefing Dec. 17. “We need the American people to do their part.”
The Omicron variant has been detected in at least 39 states and 75 countries, according to CDC director Rochelle Walensky, MD.
The strain is more transmissible than the already highly infectious Delta variant, and although there was early evidence that it caused more mild disease, she said that is likely because many of those infected have been vaccinated and boosted.
“Although Delta continues to circulate widely in the United States, Omicron is increasing rapidly and we expect it to become the dominant strain in the United States, as it has in other countries, in the coming weeks,” Dr. Walensky said.
The United States is averaging close to 1,300 deaths from COVID-19 each day. New cases, deaths, and hospitalizations are higher now than in the previous winter – before vaccines were so widely available. The New York Times reported on Dec. 17 that new infections in Connecticut and Maine have grown 150% in the past 2 weeks, and Ohio and Indiana are seeing hospitalization rates nearing the worst of 2020-2021’s winter surge.
Dueling reports released recently gave cause for relief and concern about Omicron.
A study from South Africa released on Dec. 14 shows lower hospitalizations during the first 3 weeks of the Omicron wave than during earlier waves from other variants. That’s the good news.
The concerning news is out of the United Kingdom, where Imperial College London reported Dec. 17 that the risk of reinfection with COVID-19 from Omicron is more than 5 times as high and that cases of Omicron-based COVID-19 are doubling every 2 days.
What’s more, the study “finds no evidence of Omicron having lower severity than Delta, judged by either the proportion of people testing positive who report symptoms, or by the proportion of cases seeking hospital care after infection. However, hospitalization data remains very limited at this time,” the researchers said.
“We have no evidence that the virus itself is more mild,” Eric Topol, MD, executive vice president of Scripps Research and editor-in-chief of Medscape, told PBS NewsHour. “Until we have that, we have to assume that people who don’t have any protection are highly vulnerable to getting very ill.”
The White House COVID-19 team continues to urge parents and guardians to get their children vaccinated, especially in anticipation of a post-holiday spike. Dr. Walensky said the CDC’s vaccine advisory board met on Dec. 16 to continue the safety discussion about COVID-19 vaccinations in children.
So far, 20 million children under 17 and 5 million under 11 have received their shots.
“Looking specifically at vaccine safety data from over 50,000 children 5-11 years old, we found no evidence of serious safety concerns,” Dr. Walensky said.
Top infectious disease expert Anthony S. Fauci, MD, highlighted the importance of getting vaccinated and boosted to avoid serious disease from Delta and Omicron.
“We’re in a situation where we are now facing a very important Delta surge and we are looking over our shoulder at an oncoming Omicron surge,” he said. “The optimum protection is fully vaccinated plus a boost.”
A version of this article first appeared on WebMD.com.
The best way to stay protected is by getting vaccinated and boosted, they said.
“For the unvaccinated, you’re looking at a winter of severe illness and death – for yourselves, families, and the hospitals who may soon overwhelm,” White House COVID-19 Response Coordinator Jeff Zients said at a news briefing Dec. 17. “We need the American people to do their part.”
The Omicron variant has been detected in at least 39 states and 75 countries, according to CDC director Rochelle Walensky, MD.
The strain is more transmissible than the already highly infectious Delta variant, and although there was early evidence that it caused more mild disease, she said that is likely because many of those infected have been vaccinated and boosted.
“Although Delta continues to circulate widely in the United States, Omicron is increasing rapidly and we expect it to become the dominant strain in the United States, as it has in other countries, in the coming weeks,” Dr. Walensky said.
The United States is averaging close to 1,300 deaths from COVID-19 each day. New cases, deaths, and hospitalizations are higher now than in the previous winter – before vaccines were so widely available. The New York Times reported on Dec. 17 that new infections in Connecticut and Maine have grown 150% in the past 2 weeks, and Ohio and Indiana are seeing hospitalization rates nearing the worst of 2020-2021’s winter surge.
Dueling reports released recently gave cause for relief and concern about Omicron.
A study from South Africa released on Dec. 14 shows lower hospitalizations during the first 3 weeks of the Omicron wave than during earlier waves from other variants. That’s the good news.
The concerning news is out of the United Kingdom, where Imperial College London reported Dec. 17 that the risk of reinfection with COVID-19 from Omicron is more than 5 times as high and that cases of Omicron-based COVID-19 are doubling every 2 days.
What’s more, the study “finds no evidence of Omicron having lower severity than Delta, judged by either the proportion of people testing positive who report symptoms, or by the proportion of cases seeking hospital care after infection. However, hospitalization data remains very limited at this time,” the researchers said.
“We have no evidence that the virus itself is more mild,” Eric Topol, MD, executive vice president of Scripps Research and editor-in-chief of Medscape, told PBS NewsHour. “Until we have that, we have to assume that people who don’t have any protection are highly vulnerable to getting very ill.”
The White House COVID-19 team continues to urge parents and guardians to get their children vaccinated, especially in anticipation of a post-holiday spike. Dr. Walensky said the CDC’s vaccine advisory board met on Dec. 16 to continue the safety discussion about COVID-19 vaccinations in children.
So far, 20 million children under 17 and 5 million under 11 have received their shots.
“Looking specifically at vaccine safety data from over 50,000 children 5-11 years old, we found no evidence of serious safety concerns,” Dr. Walensky said.
Top infectious disease expert Anthony S. Fauci, MD, highlighted the importance of getting vaccinated and boosted to avoid serious disease from Delta and Omicron.
“We’re in a situation where we are now facing a very important Delta surge and we are looking over our shoulder at an oncoming Omicron surge,” he said. “The optimum protection is fully vaccinated plus a boost.”
A version of this article first appeared on WebMD.com.
The best way to stay protected is by getting vaccinated and boosted, they said.
“For the unvaccinated, you’re looking at a winter of severe illness and death – for yourselves, families, and the hospitals who may soon overwhelm,” White House COVID-19 Response Coordinator Jeff Zients said at a news briefing Dec. 17. “We need the American people to do their part.”
The Omicron variant has been detected in at least 39 states and 75 countries, according to CDC director Rochelle Walensky, MD.
The strain is more transmissible than the already highly infectious Delta variant, and although there was early evidence that it caused more mild disease, she said that is likely because many of those infected have been vaccinated and boosted.
“Although Delta continues to circulate widely in the United States, Omicron is increasing rapidly and we expect it to become the dominant strain in the United States, as it has in other countries, in the coming weeks,” Dr. Walensky said.
The United States is averaging close to 1,300 deaths from COVID-19 each day. New cases, deaths, and hospitalizations are higher now than in the previous winter – before vaccines were so widely available. The New York Times reported on Dec. 17 that new infections in Connecticut and Maine have grown 150% in the past 2 weeks, and Ohio and Indiana are seeing hospitalization rates nearing the worst of 2020-2021’s winter surge.
Dueling reports released recently gave cause for relief and concern about Omicron.
A study from South Africa released on Dec. 14 shows lower hospitalizations during the first 3 weeks of the Omicron wave than during earlier waves from other variants. That’s the good news.
The concerning news is out of the United Kingdom, where Imperial College London reported Dec. 17 that the risk of reinfection with COVID-19 from Omicron is more than 5 times as high and that cases of Omicron-based COVID-19 are doubling every 2 days.
What’s more, the study “finds no evidence of Omicron having lower severity than Delta, judged by either the proportion of people testing positive who report symptoms, or by the proportion of cases seeking hospital care after infection. However, hospitalization data remains very limited at this time,” the researchers said.
“We have no evidence that the virus itself is more mild,” Eric Topol, MD, executive vice president of Scripps Research and editor-in-chief of Medscape, told PBS NewsHour. “Until we have that, we have to assume that people who don’t have any protection are highly vulnerable to getting very ill.”
The White House COVID-19 team continues to urge parents and guardians to get their children vaccinated, especially in anticipation of a post-holiday spike. Dr. Walensky said the CDC’s vaccine advisory board met on Dec. 16 to continue the safety discussion about COVID-19 vaccinations in children.
So far, 20 million children under 17 and 5 million under 11 have received their shots.
“Looking specifically at vaccine safety data from over 50,000 children 5-11 years old, we found no evidence of serious safety concerns,” Dr. Walensky said.
Top infectious disease expert Anthony S. Fauci, MD, highlighted the importance of getting vaccinated and boosted to avoid serious disease from Delta and Omicron.
“We’re in a situation where we are now facing a very important Delta surge and we are looking over our shoulder at an oncoming Omicron surge,” he said. “The optimum protection is fully vaccinated plus a boost.”
A version of this article first appeared on WebMD.com.
Small myocarditis risk now seen for adenovirus-based COVID-19 vaccine
The first large population study to investigate the association between different COVID-19 vaccines types and cardiac effects and adverse events shows a small increase in the risk for acute myocarditis with both the mRNA-based vaccines and – in what may a first in the literature – an adenovirus-vector vaccine.
The excess risk was seen following the first dose of the ChAdOc1 (AstraZeneca/Oxford), the adenovirus-based vaccine, and the mRNA-based BNT162b2 (Pfizer/BioNTech). It was observed after first and second doses of the mRNA-1273 (Moderna) vaccine.
The incidence rate ratios for myocarditis 1-7 days after the first AstraZeneca, Pfizer, and Moderna injections were 1.76, 1.45, and 8.38, respectively, and 23.1 after the second dose of the Moderna vaccine.
“There’s a bit more uncertainty and worry about mRNA vaccines because it’s quite a new vector for vaccination and, therefore, there’s been more focus on the potential side effects,” said Nicholas Mills, MD.
“But it doesn’t surprise me the signal is present for all types of vaccines because they’re designed to generate a systemic immune response and that is, unfortunately, where you can cause small risks for immune-mediated illnesses like myocarditis,” Dr. Mills, from the University of Edinburgh, told this news organization. Dr. Mills is a coauthor on the study, published Dec. 14 in Nature Medicine.
To put the risks in context, the group estimated between 1 and 10 additional myocarditis hospitalizations or deaths per 1 million people vaccinated, but 40 excess myocarditis events per million following a positive SARS-CoV-2 test result.
As reported, rates of excess myocarditis events associated with a first dose were 2 per million injections of the AstraZeneca vaccine, 1 per million for the Pfizer vaccine, and 6 per million with the Moderna vaccine.
Following a second dose, there were 10 additional myocarditis events per million people receiving the Moderna vaccine and none among recipients of the AstraZeneca or Pfizer vaccines.
“It was particularly seen within the first 7 days of the first dose, which is very consistent with what we see in people who have viral myocarditis,” Dr. Mills said. “So it looks like a real signal but it’s very small.”
The results are in line with previous studies of the Pfizer vaccine in Israel and studies of the Moderna vaccine in the United States, Biykem Bozkurt, MD, PhD, professor of medicine at Baylor College of Medicine, Houston, told this news organization.
“What this paper does is confirm that cardiovascular complications – and they are only looking at a small component of those cardiovascular complications – are markedly higher with the COVID-19 infection than with the vaccines,” she said.
It also adds a new twist to the search for the mechanisms of myocarditis, which has focused on the immunogenicity of the RNA in the Pfizer and Moderna vaccines but also hypothesized that molecular mimicry between the SARS-CoV-2 spike glycoprotein and cell antigens, antibody production against cardiac proteins, and testosterone may play a role.
“But now it doesn’t look like the risk is solely confined to the mRNA vaccine platform because it’s also happening with the adenovirus,” Dr. Bozkurt said. “The mechanisms require future experimental and clinical research and we’ll need more granular data with cohorts that are closely followed up as well as subclinical follow-up.”
James de Lemos, MD, professor of medicine at the University of Texas Southwestern Medical Center, Dallas, and cochair of the American Heart Association’s COVID-19 CVD Registry, said he was also not surprised by a myocarditis signal with AstraZeneca’s adenovirus vaccine.
“Looking at relative risks has biological implications, but the clinical and public health implications are that the absolute risk with the adenovirus is trivial. And you see that with their estimations of absolute risk where it’s literally sort of a needle in the haystack of 1 or 2 per million,” he said in an interview.
Large-scale data
The investigators examined the rates of hospital admission or death from myocarditis, pericarditis, and cardiac arrhythmia in the 28 days following SARS-CoV-2 vaccination or infection by linking the English National Immunisation Database of COVID-19 vaccination with a national patient-level health care database of 38.6 million people, aged 16 years or older, vaccinated from Dec.1, 2020, to Aug. 24, 2021.
The number of people admitted to the hospital or who died during the study period was 1,615 for myocarditis, 1,574 for pericarditis, and 385,508 for cardiac arrhythmia.
There was no evidence of an increased risk for pericarditis or cardiac arrhythmia following vaccination, except for arrhythmia in the 28 days following a second dose of the Moderna vaccine (IRR, 1.46).
In contrast, the risk was increased for pericarditis (IRR, 2.79) and cardiac arrhythmia (IRR, 5.35) in the 28 days following a positive SARS-CoV-2 test result.
Although the scale of the analysis allows for more precise estimates than what’s been possible in smaller data sets, there is the challenge of diagnosing COVID-19 from billing codes and the potential for ascertainment bias, noted Dr. de Lemos.
“Having said that, I think it’s a really important study, because it’s the first study to put the incidence in context in the same general population the risks of myocarditis with various vaccines and with COVID-19,” he said.
“That’s really important and provides a lot of reassurance for those who are trying to balance the risks and benefits of vaccination.”
Analyses by sex and age
A subgroup analysis by age showed increased risks for myocarditis with the mRNA vaccines only in those younger than 40, whereas no association was found with the Oxford adenovirus vaccine.
“We’re not seeing any signal here that would make us change the recommendation for vaccination in children as a consequence of this risk,” Dr. Mills said during a press briefing.
Dr. Bozkurt pointed out, however, that the estimated excess in myocarditis events following a second dose of the Moderna vaccine in these younger adults reportedly exceeded that for SARS-CoV-2 infection (15 per million vs. 10 per million).
“For that age group, it’s concerning and needs further clarification. This hasn’t been seen before,” she said.
The average age was 39 years for those receiving two doses of the Moderna vaccine and 55 for recipients of the Pfizer and Oxford vaccines. The Moderna vaccine wasn’t rolled out until April 2021 in the United Kingdom, the authors noted, so the number of patients who received this vaccine is lower.
Although reports have suggested young males are at greater risk for myocarditis after vaccination, an analysis by sex found that women had an increased risk for myocarditis after a first dose of the AstraZeneca (IRR, 1.40) and Pfizer (IRR, 1.54) vaccines and following a positive COVID-19 test result (IRR, 11.00).
“Women being at increased risk is rather a new message,” Dr. Bozkurt said. “But the incidence rate ratios are being compared against the unvaccinated, so when you see the increase in women, it doesn’t mean it’s increased against men. It would be helpful for sex-specific incidence rate ratios to be reported for younger age subgroups, such as ages 16-20 and 20-30, to determine whether there’s an increased risk for males compared to females at younger ages.”
Age and sex differences are huge questions, but “I think we’ll learn a lot about myocarditis in general from what is going to be an explosion of research into the vaccine-associated causes,” Dr. de Lemos said.
“That will help us understand myocarditis more broadly and prepare us for the next generation of vaccines, which inevitably will be mRNA based.”
Dr. Mills reported having no relevant disclosures. Dr. Bozkurt reported consulting for Bayer and scPharmaceuticals and serving on a clinical-events committee for a trial supported by Abbott Pharmaceuticals and on a data and safety monitoring board for a trial supported by Liva Nova Pharmaceuticals. Dr. De Lemos reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The first large population study to investigate the association between different COVID-19 vaccines types and cardiac effects and adverse events shows a small increase in the risk for acute myocarditis with both the mRNA-based vaccines and – in what may a first in the literature – an adenovirus-vector vaccine.
The excess risk was seen following the first dose of the ChAdOc1 (AstraZeneca/Oxford), the adenovirus-based vaccine, and the mRNA-based BNT162b2 (Pfizer/BioNTech). It was observed after first and second doses of the mRNA-1273 (Moderna) vaccine.
The incidence rate ratios for myocarditis 1-7 days after the first AstraZeneca, Pfizer, and Moderna injections were 1.76, 1.45, and 8.38, respectively, and 23.1 after the second dose of the Moderna vaccine.
“There’s a bit more uncertainty and worry about mRNA vaccines because it’s quite a new vector for vaccination and, therefore, there’s been more focus on the potential side effects,” said Nicholas Mills, MD.
“But it doesn’t surprise me the signal is present for all types of vaccines because they’re designed to generate a systemic immune response and that is, unfortunately, where you can cause small risks for immune-mediated illnesses like myocarditis,” Dr. Mills, from the University of Edinburgh, told this news organization. Dr. Mills is a coauthor on the study, published Dec. 14 in Nature Medicine.
To put the risks in context, the group estimated between 1 and 10 additional myocarditis hospitalizations or deaths per 1 million people vaccinated, but 40 excess myocarditis events per million following a positive SARS-CoV-2 test result.
As reported, rates of excess myocarditis events associated with a first dose were 2 per million injections of the AstraZeneca vaccine, 1 per million for the Pfizer vaccine, and 6 per million with the Moderna vaccine.
Following a second dose, there were 10 additional myocarditis events per million people receiving the Moderna vaccine and none among recipients of the AstraZeneca or Pfizer vaccines.
“It was particularly seen within the first 7 days of the first dose, which is very consistent with what we see in people who have viral myocarditis,” Dr. Mills said. “So it looks like a real signal but it’s very small.”
The results are in line with previous studies of the Pfizer vaccine in Israel and studies of the Moderna vaccine in the United States, Biykem Bozkurt, MD, PhD, professor of medicine at Baylor College of Medicine, Houston, told this news organization.
“What this paper does is confirm that cardiovascular complications – and they are only looking at a small component of those cardiovascular complications – are markedly higher with the COVID-19 infection than with the vaccines,” she said.
It also adds a new twist to the search for the mechanisms of myocarditis, which has focused on the immunogenicity of the RNA in the Pfizer and Moderna vaccines but also hypothesized that molecular mimicry between the SARS-CoV-2 spike glycoprotein and cell antigens, antibody production against cardiac proteins, and testosterone may play a role.
“But now it doesn’t look like the risk is solely confined to the mRNA vaccine platform because it’s also happening with the adenovirus,” Dr. Bozkurt said. “The mechanisms require future experimental and clinical research and we’ll need more granular data with cohorts that are closely followed up as well as subclinical follow-up.”
James de Lemos, MD, professor of medicine at the University of Texas Southwestern Medical Center, Dallas, and cochair of the American Heart Association’s COVID-19 CVD Registry, said he was also not surprised by a myocarditis signal with AstraZeneca’s adenovirus vaccine.
“Looking at relative risks has biological implications, but the clinical and public health implications are that the absolute risk with the adenovirus is trivial. And you see that with their estimations of absolute risk where it’s literally sort of a needle in the haystack of 1 or 2 per million,” he said in an interview.
Large-scale data
The investigators examined the rates of hospital admission or death from myocarditis, pericarditis, and cardiac arrhythmia in the 28 days following SARS-CoV-2 vaccination or infection by linking the English National Immunisation Database of COVID-19 vaccination with a national patient-level health care database of 38.6 million people, aged 16 years or older, vaccinated from Dec.1, 2020, to Aug. 24, 2021.
The number of people admitted to the hospital or who died during the study period was 1,615 for myocarditis, 1,574 for pericarditis, and 385,508 for cardiac arrhythmia.
There was no evidence of an increased risk for pericarditis or cardiac arrhythmia following vaccination, except for arrhythmia in the 28 days following a second dose of the Moderna vaccine (IRR, 1.46).
In contrast, the risk was increased for pericarditis (IRR, 2.79) and cardiac arrhythmia (IRR, 5.35) in the 28 days following a positive SARS-CoV-2 test result.
Although the scale of the analysis allows for more precise estimates than what’s been possible in smaller data sets, there is the challenge of diagnosing COVID-19 from billing codes and the potential for ascertainment bias, noted Dr. de Lemos.
“Having said that, I think it’s a really important study, because it’s the first study to put the incidence in context in the same general population the risks of myocarditis with various vaccines and with COVID-19,” he said.
“That’s really important and provides a lot of reassurance for those who are trying to balance the risks and benefits of vaccination.”
Analyses by sex and age
A subgroup analysis by age showed increased risks for myocarditis with the mRNA vaccines only in those younger than 40, whereas no association was found with the Oxford adenovirus vaccine.
“We’re not seeing any signal here that would make us change the recommendation for vaccination in children as a consequence of this risk,” Dr. Mills said during a press briefing.
Dr. Bozkurt pointed out, however, that the estimated excess in myocarditis events following a second dose of the Moderna vaccine in these younger adults reportedly exceeded that for SARS-CoV-2 infection (15 per million vs. 10 per million).
“For that age group, it’s concerning and needs further clarification. This hasn’t been seen before,” she said.
The average age was 39 years for those receiving two doses of the Moderna vaccine and 55 for recipients of the Pfizer and Oxford vaccines. The Moderna vaccine wasn’t rolled out until April 2021 in the United Kingdom, the authors noted, so the number of patients who received this vaccine is lower.
Although reports have suggested young males are at greater risk for myocarditis after vaccination, an analysis by sex found that women had an increased risk for myocarditis after a first dose of the AstraZeneca (IRR, 1.40) and Pfizer (IRR, 1.54) vaccines and following a positive COVID-19 test result (IRR, 11.00).
“Women being at increased risk is rather a new message,” Dr. Bozkurt said. “But the incidence rate ratios are being compared against the unvaccinated, so when you see the increase in women, it doesn’t mean it’s increased against men. It would be helpful for sex-specific incidence rate ratios to be reported for younger age subgroups, such as ages 16-20 and 20-30, to determine whether there’s an increased risk for males compared to females at younger ages.”
Age and sex differences are huge questions, but “I think we’ll learn a lot about myocarditis in general from what is going to be an explosion of research into the vaccine-associated causes,” Dr. de Lemos said.
“That will help us understand myocarditis more broadly and prepare us for the next generation of vaccines, which inevitably will be mRNA based.”
Dr. Mills reported having no relevant disclosures. Dr. Bozkurt reported consulting for Bayer and scPharmaceuticals and serving on a clinical-events committee for a trial supported by Abbott Pharmaceuticals and on a data and safety monitoring board for a trial supported by Liva Nova Pharmaceuticals. Dr. De Lemos reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The first large population study to investigate the association between different COVID-19 vaccines types and cardiac effects and adverse events shows a small increase in the risk for acute myocarditis with both the mRNA-based vaccines and – in what may a first in the literature – an adenovirus-vector vaccine.
The excess risk was seen following the first dose of the ChAdOc1 (AstraZeneca/Oxford), the adenovirus-based vaccine, and the mRNA-based BNT162b2 (Pfizer/BioNTech). It was observed after first and second doses of the mRNA-1273 (Moderna) vaccine.
The incidence rate ratios for myocarditis 1-7 days after the first AstraZeneca, Pfizer, and Moderna injections were 1.76, 1.45, and 8.38, respectively, and 23.1 after the second dose of the Moderna vaccine.
“There’s a bit more uncertainty and worry about mRNA vaccines because it’s quite a new vector for vaccination and, therefore, there’s been more focus on the potential side effects,” said Nicholas Mills, MD.
“But it doesn’t surprise me the signal is present for all types of vaccines because they’re designed to generate a systemic immune response and that is, unfortunately, where you can cause small risks for immune-mediated illnesses like myocarditis,” Dr. Mills, from the University of Edinburgh, told this news organization. Dr. Mills is a coauthor on the study, published Dec. 14 in Nature Medicine.
To put the risks in context, the group estimated between 1 and 10 additional myocarditis hospitalizations or deaths per 1 million people vaccinated, but 40 excess myocarditis events per million following a positive SARS-CoV-2 test result.
As reported, rates of excess myocarditis events associated with a first dose were 2 per million injections of the AstraZeneca vaccine, 1 per million for the Pfizer vaccine, and 6 per million with the Moderna vaccine.
Following a second dose, there were 10 additional myocarditis events per million people receiving the Moderna vaccine and none among recipients of the AstraZeneca or Pfizer vaccines.
“It was particularly seen within the first 7 days of the first dose, which is very consistent with what we see in people who have viral myocarditis,” Dr. Mills said. “So it looks like a real signal but it’s very small.”
The results are in line with previous studies of the Pfizer vaccine in Israel and studies of the Moderna vaccine in the United States, Biykem Bozkurt, MD, PhD, professor of medicine at Baylor College of Medicine, Houston, told this news organization.
“What this paper does is confirm that cardiovascular complications – and they are only looking at a small component of those cardiovascular complications – are markedly higher with the COVID-19 infection than with the vaccines,” she said.
It also adds a new twist to the search for the mechanisms of myocarditis, which has focused on the immunogenicity of the RNA in the Pfizer and Moderna vaccines but also hypothesized that molecular mimicry between the SARS-CoV-2 spike glycoprotein and cell antigens, antibody production against cardiac proteins, and testosterone may play a role.
“But now it doesn’t look like the risk is solely confined to the mRNA vaccine platform because it’s also happening with the adenovirus,” Dr. Bozkurt said. “The mechanisms require future experimental and clinical research and we’ll need more granular data with cohorts that are closely followed up as well as subclinical follow-up.”
James de Lemos, MD, professor of medicine at the University of Texas Southwestern Medical Center, Dallas, and cochair of the American Heart Association’s COVID-19 CVD Registry, said he was also not surprised by a myocarditis signal with AstraZeneca’s adenovirus vaccine.
“Looking at relative risks has biological implications, but the clinical and public health implications are that the absolute risk with the adenovirus is trivial. And you see that with their estimations of absolute risk where it’s literally sort of a needle in the haystack of 1 or 2 per million,” he said in an interview.
Large-scale data
The investigators examined the rates of hospital admission or death from myocarditis, pericarditis, and cardiac arrhythmia in the 28 days following SARS-CoV-2 vaccination or infection by linking the English National Immunisation Database of COVID-19 vaccination with a national patient-level health care database of 38.6 million people, aged 16 years or older, vaccinated from Dec.1, 2020, to Aug. 24, 2021.
The number of people admitted to the hospital or who died during the study period was 1,615 for myocarditis, 1,574 for pericarditis, and 385,508 for cardiac arrhythmia.
There was no evidence of an increased risk for pericarditis or cardiac arrhythmia following vaccination, except for arrhythmia in the 28 days following a second dose of the Moderna vaccine (IRR, 1.46).
In contrast, the risk was increased for pericarditis (IRR, 2.79) and cardiac arrhythmia (IRR, 5.35) in the 28 days following a positive SARS-CoV-2 test result.
Although the scale of the analysis allows for more precise estimates than what’s been possible in smaller data sets, there is the challenge of diagnosing COVID-19 from billing codes and the potential for ascertainment bias, noted Dr. de Lemos.
“Having said that, I think it’s a really important study, because it’s the first study to put the incidence in context in the same general population the risks of myocarditis with various vaccines and with COVID-19,” he said.
“That’s really important and provides a lot of reassurance for those who are trying to balance the risks and benefits of vaccination.”
Analyses by sex and age
A subgroup analysis by age showed increased risks for myocarditis with the mRNA vaccines only in those younger than 40, whereas no association was found with the Oxford adenovirus vaccine.
“We’re not seeing any signal here that would make us change the recommendation for vaccination in children as a consequence of this risk,” Dr. Mills said during a press briefing.
Dr. Bozkurt pointed out, however, that the estimated excess in myocarditis events following a second dose of the Moderna vaccine in these younger adults reportedly exceeded that for SARS-CoV-2 infection (15 per million vs. 10 per million).
“For that age group, it’s concerning and needs further clarification. This hasn’t been seen before,” she said.
The average age was 39 years for those receiving two doses of the Moderna vaccine and 55 for recipients of the Pfizer and Oxford vaccines. The Moderna vaccine wasn’t rolled out until April 2021 in the United Kingdom, the authors noted, so the number of patients who received this vaccine is lower.
Although reports have suggested young males are at greater risk for myocarditis after vaccination, an analysis by sex found that women had an increased risk for myocarditis after a first dose of the AstraZeneca (IRR, 1.40) and Pfizer (IRR, 1.54) vaccines and following a positive COVID-19 test result (IRR, 11.00).
“Women being at increased risk is rather a new message,” Dr. Bozkurt said. “But the incidence rate ratios are being compared against the unvaccinated, so when you see the increase in women, it doesn’t mean it’s increased against men. It would be helpful for sex-specific incidence rate ratios to be reported for younger age subgroups, such as ages 16-20 and 20-30, to determine whether there’s an increased risk for males compared to females at younger ages.”
Age and sex differences are huge questions, but “I think we’ll learn a lot about myocarditis in general from what is going to be an explosion of research into the vaccine-associated causes,” Dr. de Lemos said.
“That will help us understand myocarditis more broadly and prepare us for the next generation of vaccines, which inevitably will be mRNA based.”
Dr. Mills reported having no relevant disclosures. Dr. Bozkurt reported consulting for Bayer and scPharmaceuticals and serving on a clinical-events committee for a trial supported by Abbott Pharmaceuticals and on a data and safety monitoring board for a trial supported by Liva Nova Pharmaceuticals. Dr. De Lemos reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM NATURE MEDICINE
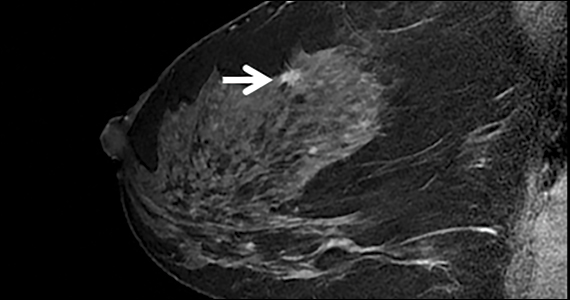
Postmenopausal women with early breast cancer can go chemo-free
New results from the phase 3 RxPONDER trial add to mounting evidence that most postmenopausal women with early-stage breast cancer derive no added benefits from chemotherapy and can be effectively treated with endocrine therapy alone.
The study, published in The New England Journal of Medicine, conversely shows that premenopausal women do benefit from adjuvant chemotherapy, theorized by many to largely be the result of chemotherapy-induced ovarian function suppression.
The RxPONDER trial results are in line with those from the practice-changing TAILORx trial and underscore that “postmenopausal women with 1 to 3 positive nodes and [a recurrence score] of 0 to 25 can likely safely forgo adjuvant chemotherapy without compromising invasive disease-free survival,” first author Kevin Kalinsky, MD, of the Winship Cancer Institute at Emory University, Atlanta, told this news organization. “This will save tens of thousands of women the time, expense, and potentially harmful side effects that can be associated with chemotherapy infusions.”
However, the authors note, “premenopausal women with 1-3 positive lymph nodes had a significant benefit from chemotherapy.”
The study, conducted by the Southwest Oncology Group (SWOG) Cancer Research Network, involved 5,018 women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer with one to three positive axillary lymph nodes – a breast cancer profile that represents approximately 20% of cases in the U.S.
All women had recurrence scores on the 100-point 21-gene breast cancer assay (Oncotype Dx) under 25, which is considered the lowest risk of recurrence. Patients were randomized to treatment with endocrine therapy only (n = 2,507) or chemotherapy followed by endocrine therapy (n = 2,511).
After a median follow-up of 5.3 years, women treated with adjunctive chemotherapy plus endocrine therapy exhibited no significant improvements in invasive disease-free survival compared to those who received endocrine therapy alone.
A prespecified analysis stratifying women by menopausal status underscored those results among postmenopausal women. In this cohort, researchers reported invasive disease-free survival was 91.9% in the endocrine-only group and 91.3% in the chemotherapy group (HR, 1.02; P = .89), indicating no benefit of the adjunctive chemotherapy.
However, among premenopausal women, the invasive disease-free survival rate was significantly higher with the addition of chemotherapy – 89.0% with endocrine-only therapy and 93.9% with both therapies (HR, 0.60; P = .002). Increases in distant relapse-free survival observed in the dual-therapy group similarly favored adding chemotherapy (HR, 0.58; P = .009).
Even when the authors further stratified the women into recurrence scores of 0 to 13 or 14 to 25, the results remained consistent. Postmenopausal women in each of the recurrence score groups continued to show no difference in invasive disease recurrence, new primary cancer, or death from chemotherapy (HR, 1.01 for each score group). Conversely, premenopausal women showed significant improvements in those outcomes when chemotherapy was added to endocrine therapy.
To what degree were the effects observed in premenopausal women the result of chemotherapy-induced ovarian suppression?
“I think it’s fair to say it’s the most interesting question right now in early-stage breast cancer for ER-positive tumors,” Harold Burstein, MD, of the Dana-Farber Cancer Institute and Harvard Medical School, Boston, said during a debate at the recent San Antonio Breast Cancer Symposium.
According to Sibylle Loibl, MD, PhD, when it comes to the use of chemotherapy, “age matters.”
“I strongly believe the biology of tumors is different in younger women with HR-positive/HER2-negative breast cancer,” Dr. Loibl, an associate professor at the University of Frankfurt, said during the debate. “It’s a different disease and the effects of chemotherapy are different.”
In young women, chemotherapy has “a direct cytotoxic effect, which cannot be neglected, and an endocrine effect on ovarian function suppression,” Dr. Loibl added. “I think both are needed in young premenopausal patients.”
According to the RxPONDER authors, “whether a chemotherapy benefit in premenopausal women is due to both direct cytocidal effects and treatment-induced menopause remains unclear,” but they noted that “it is possible that the contribution of these mechanisms may vary according to age.”
Further complicating matters, Dr. Loibl added, is that age appears to be poorly represented in genetic diagnostic tools.
“I think the gene expression profiles we are currently using as standard diagnostic tools do not capture the right biology for our premenopausal patients,” she said. “We have to keep in mind that these tests were designed and validated in postmenopausal patients and were only retrospectively used in premenopausal patients.”
The study was funded by the National Cancer Institute and others. Dr. Loibl has received honoraria from Prime and Chugai and numerous institutional research grants.
A version of this article first appeared on Medscape.com.
New results from the phase 3 RxPONDER trial add to mounting evidence that most postmenopausal women with early-stage breast cancer derive no added benefits from chemotherapy and can be effectively treated with endocrine therapy alone.
The study, published in The New England Journal of Medicine, conversely shows that premenopausal women do benefit from adjuvant chemotherapy, theorized by many to largely be the result of chemotherapy-induced ovarian function suppression.
The RxPONDER trial results are in line with those from the practice-changing TAILORx trial and underscore that “postmenopausal women with 1 to 3 positive nodes and [a recurrence score] of 0 to 25 can likely safely forgo adjuvant chemotherapy without compromising invasive disease-free survival,” first author Kevin Kalinsky, MD, of the Winship Cancer Institute at Emory University, Atlanta, told this news organization. “This will save tens of thousands of women the time, expense, and potentially harmful side effects that can be associated with chemotherapy infusions.”
However, the authors note, “premenopausal women with 1-3 positive lymph nodes had a significant benefit from chemotherapy.”
The study, conducted by the Southwest Oncology Group (SWOG) Cancer Research Network, involved 5,018 women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer with one to three positive axillary lymph nodes – a breast cancer profile that represents approximately 20% of cases in the U.S.
All women had recurrence scores on the 100-point 21-gene breast cancer assay (Oncotype Dx) under 25, which is considered the lowest risk of recurrence. Patients were randomized to treatment with endocrine therapy only (n = 2,507) or chemotherapy followed by endocrine therapy (n = 2,511).
After a median follow-up of 5.3 years, women treated with adjunctive chemotherapy plus endocrine therapy exhibited no significant improvements in invasive disease-free survival compared to those who received endocrine therapy alone.
A prespecified analysis stratifying women by menopausal status underscored those results among postmenopausal women. In this cohort, researchers reported invasive disease-free survival was 91.9% in the endocrine-only group and 91.3% in the chemotherapy group (HR, 1.02; P = .89), indicating no benefit of the adjunctive chemotherapy.
However, among premenopausal women, the invasive disease-free survival rate was significantly higher with the addition of chemotherapy – 89.0% with endocrine-only therapy and 93.9% with both therapies (HR, 0.60; P = .002). Increases in distant relapse-free survival observed in the dual-therapy group similarly favored adding chemotherapy (HR, 0.58; P = .009).
Even when the authors further stratified the women into recurrence scores of 0 to 13 or 14 to 25, the results remained consistent. Postmenopausal women in each of the recurrence score groups continued to show no difference in invasive disease recurrence, new primary cancer, or death from chemotherapy (HR, 1.01 for each score group). Conversely, premenopausal women showed significant improvements in those outcomes when chemotherapy was added to endocrine therapy.
To what degree were the effects observed in premenopausal women the result of chemotherapy-induced ovarian suppression?
“I think it’s fair to say it’s the most interesting question right now in early-stage breast cancer for ER-positive tumors,” Harold Burstein, MD, of the Dana-Farber Cancer Institute and Harvard Medical School, Boston, said during a debate at the recent San Antonio Breast Cancer Symposium.
According to Sibylle Loibl, MD, PhD, when it comes to the use of chemotherapy, “age matters.”
“I strongly believe the biology of tumors is different in younger women with HR-positive/HER2-negative breast cancer,” Dr. Loibl, an associate professor at the University of Frankfurt, said during the debate. “It’s a different disease and the effects of chemotherapy are different.”
In young women, chemotherapy has “a direct cytotoxic effect, which cannot be neglected, and an endocrine effect on ovarian function suppression,” Dr. Loibl added. “I think both are needed in young premenopausal patients.”
According to the RxPONDER authors, “whether a chemotherapy benefit in premenopausal women is due to both direct cytocidal effects and treatment-induced menopause remains unclear,” but they noted that “it is possible that the contribution of these mechanisms may vary according to age.”
Further complicating matters, Dr. Loibl added, is that age appears to be poorly represented in genetic diagnostic tools.
“I think the gene expression profiles we are currently using as standard diagnostic tools do not capture the right biology for our premenopausal patients,” she said. “We have to keep in mind that these tests were designed and validated in postmenopausal patients and were only retrospectively used in premenopausal patients.”
The study was funded by the National Cancer Institute and others. Dr. Loibl has received honoraria from Prime and Chugai and numerous institutional research grants.
A version of this article first appeared on Medscape.com.
New results from the phase 3 RxPONDER trial add to mounting evidence that most postmenopausal women with early-stage breast cancer derive no added benefits from chemotherapy and can be effectively treated with endocrine therapy alone.
The study, published in The New England Journal of Medicine, conversely shows that premenopausal women do benefit from adjuvant chemotherapy, theorized by many to largely be the result of chemotherapy-induced ovarian function suppression.
The RxPONDER trial results are in line with those from the practice-changing TAILORx trial and underscore that “postmenopausal women with 1 to 3 positive nodes and [a recurrence score] of 0 to 25 can likely safely forgo adjuvant chemotherapy without compromising invasive disease-free survival,” first author Kevin Kalinsky, MD, of the Winship Cancer Institute at Emory University, Atlanta, told this news organization. “This will save tens of thousands of women the time, expense, and potentially harmful side effects that can be associated with chemotherapy infusions.”
However, the authors note, “premenopausal women with 1-3 positive lymph nodes had a significant benefit from chemotherapy.”
The study, conducted by the Southwest Oncology Group (SWOG) Cancer Research Network, involved 5,018 women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer with one to three positive axillary lymph nodes – a breast cancer profile that represents approximately 20% of cases in the U.S.
All women had recurrence scores on the 100-point 21-gene breast cancer assay (Oncotype Dx) under 25, which is considered the lowest risk of recurrence. Patients were randomized to treatment with endocrine therapy only (n = 2,507) or chemotherapy followed by endocrine therapy (n = 2,511).
After a median follow-up of 5.3 years, women treated with adjunctive chemotherapy plus endocrine therapy exhibited no significant improvements in invasive disease-free survival compared to those who received endocrine therapy alone.
A prespecified analysis stratifying women by menopausal status underscored those results among postmenopausal women. In this cohort, researchers reported invasive disease-free survival was 91.9% in the endocrine-only group and 91.3% in the chemotherapy group (HR, 1.02; P = .89), indicating no benefit of the adjunctive chemotherapy.
However, among premenopausal women, the invasive disease-free survival rate was significantly higher with the addition of chemotherapy – 89.0% with endocrine-only therapy and 93.9% with both therapies (HR, 0.60; P = .002). Increases in distant relapse-free survival observed in the dual-therapy group similarly favored adding chemotherapy (HR, 0.58; P = .009).
Even when the authors further stratified the women into recurrence scores of 0 to 13 or 14 to 25, the results remained consistent. Postmenopausal women in each of the recurrence score groups continued to show no difference in invasive disease recurrence, new primary cancer, or death from chemotherapy (HR, 1.01 for each score group). Conversely, premenopausal women showed significant improvements in those outcomes when chemotherapy was added to endocrine therapy.
To what degree were the effects observed in premenopausal women the result of chemotherapy-induced ovarian suppression?
“I think it’s fair to say it’s the most interesting question right now in early-stage breast cancer for ER-positive tumors,” Harold Burstein, MD, of the Dana-Farber Cancer Institute and Harvard Medical School, Boston, said during a debate at the recent San Antonio Breast Cancer Symposium.
According to Sibylle Loibl, MD, PhD, when it comes to the use of chemotherapy, “age matters.”
“I strongly believe the biology of tumors is different in younger women with HR-positive/HER2-negative breast cancer,” Dr. Loibl, an associate professor at the University of Frankfurt, said during the debate. “It’s a different disease and the effects of chemotherapy are different.”
In young women, chemotherapy has “a direct cytotoxic effect, which cannot be neglected, and an endocrine effect on ovarian function suppression,” Dr. Loibl added. “I think both are needed in young premenopausal patients.”
According to the RxPONDER authors, “whether a chemotherapy benefit in premenopausal women is due to both direct cytocidal effects and treatment-induced menopause remains unclear,” but they noted that “it is possible that the contribution of these mechanisms may vary according to age.”
Further complicating matters, Dr. Loibl added, is that age appears to be poorly represented in genetic diagnostic tools.
“I think the gene expression profiles we are currently using as standard diagnostic tools do not capture the right biology for our premenopausal patients,” she said. “We have to keep in mind that these tests were designed and validated in postmenopausal patients and were only retrospectively used in premenopausal patients.”
The study was funded by the National Cancer Institute and others. Dr. Loibl has received honoraria from Prime and Chugai and numerous institutional research grants.
A version of this article first appeared on Medscape.com.
Cardiovascular effects of breast cancer treatment vary based on weight, menopausal status
For example, certain chemotherapy drugs may confer higher risk in breast cancer survivors of normal weight, whereas they may lower stroke risk in those who are obese, according to Heather Greenlee, ND, PhD, a public health researcher and naturopathic physician with the Fred Hutchinson Cancer Research Center in Seattle.
In postmenopausal women with breast cancer, aromatase inhibitors may increase cardiovascular risk, while tamoxifen appears to reduce the risk of incident dyslipidemia, she said.
The findings are from separate analyses of data from studies presented during a poster discussion session at the symposium.
Breast cancer treatment and cardiovascular effects: The role of weight
In one analysis, Dr. Greenlee and colleagues examined outcomes in 13,582 breast cancer survivors with a median age of 60 years and median follow-up of 7 years to assess whether cardiovascular disease (CVD) risk associated with specific breast cancer therapies varies by body mass index (BMI) category at diagnosis.
Many routinely used breast cancer therapies are cardiotoxic, and being overweight or obese are known risk factors for CVD, but few studies have assessed whether BMI modifies the effect of these treatment on cardiovascular risk, Dr. Greenlee explained.
After adjusting for baseline demographic and health-related factors, and other breast cancer treatment, they found that:
- Ischemic heart disease risk was higher among normal-weight women who received anthracyclines, compared with those who did not (hazard ratio, 4.2). No other risk associations were observed for other breast cancer therapies and BMI groups.
- Heart failure/cardiomyopathy risk was higher among women with normal weight who received anthracyclines, cyclophosphamides, or left-sided radiation, compared with those who did not (HRs, 5.24, 3.27, and 2.05, respectively), and among overweight women who received anthracyclines, compared with those who did not (HR, 2.18). No risk associations were observed for women who received trastuzumab, taxanes, endocrine therapy, or radiation on any side, and no risk associations were observed for women who were obese.
- Stroke risk was higher in normal-weight women who received taxanes, cyclophosphamides, or left-sided radiation versus those who did not (HRs, 2.14, 2.35, and 1.31, respectively), and stroke risk was lower in obese women who received anthracyclines, taxanes, or cyclophosphamide, compared with those who did not (HRs, 0.32, 0.41, and 0.29, respectively). No risk associations were observed for trastuzumab, endocrine therapy, or radiation on any side, and no risk associations were observed for women who were overweight.
The lack of associations noted between treatments and heart failure risk among obese patients could be caused by the “obesity paradox” observed in prior obese populations, the investigators noted, adding that additional analyses are planned to “examine whether different dosage and duration of breast cancer therapy exposures across the BMI groups contributed to these risk associations.”
Breast cancer treatment and cardiometabolic effects: The role of menopausal status
In a separate analysis, Dr. Greenlee and colleagues looked at the association between endocrine therapies and cardiometabolic risk based on menopausal status.
Endocrine therapy is associated with CVD in breast cancer survivors and may be associated with developing cardiometabolic risk factors like diabetes, dyslipidemia, and hypertension, they noted, explaining that tamoxifen has mixed estrogenic and antiestrogenic activity, while aromatase inhibitors deplete endogenous estrogen.
Since most studies have compared tamoxifen with aromatase inhibitor use, it has been a challenge challenging to discern the effects of each, Dr. Greenlee said.
She and her colleagues reviewed records for 14,942 breast cancer survivors who were diagnosed between 2005 and 2013. The patients had a mean age of 61 years at baseline, and 24.9% were premenopausal at the time of diagnosis. Of the premenopausal women, 27.3% used tamoxifen, 19.2% used aromatase inhibitors, and 53.5% did not use endocrine therapy, and of the postmenopausal women, 6.6% used tamoxifen, 47.7% used aromatase inhibitors, and 45.7% did not use endocrine therapy.
After adjusting for baseline demographics and health factors, the investigators found that:
- The use of tamoxifen or aromatase inhibitors was not associated with a risk of developing diabetes, dyslipidemia, or hypertension in premenopausal women, or with a risk of developing diabetes or hypertension in postmenopausal women.
- The risk of dyslipidemia was higher in postmenopausal aromatase inhibitor users, and lower in postmenopausal tamoxifen users, compared with postmenopausal non-users of endocrine therapy (HRs, 1.15 and 0.75, respectively).
The lack of associations between endocrine therapy and CVD risk in premenopausal women may be from low power, Dr. Greenlee said, noting that analyses in larger sample sizes are needed.
She and her colleagues plan to conduct further analyses looking at treatment dosage and duration, and comparing steroidal versus nonsteroidal aromatase inhibitors.
Future studies should examine the implications of these associations on long-term CVD and how best to manage lipid profiles in postmenopausal breast cancer survivors who have a history of endocrine therapy treatment, they concluded.
This research was funded by grants from the National Cancer Institute.
For example, certain chemotherapy drugs may confer higher risk in breast cancer survivors of normal weight, whereas they may lower stroke risk in those who are obese, according to Heather Greenlee, ND, PhD, a public health researcher and naturopathic physician with the Fred Hutchinson Cancer Research Center in Seattle.
In postmenopausal women with breast cancer, aromatase inhibitors may increase cardiovascular risk, while tamoxifen appears to reduce the risk of incident dyslipidemia, she said.
The findings are from separate analyses of data from studies presented during a poster discussion session at the symposium.
Breast cancer treatment and cardiovascular effects: The role of weight
In one analysis, Dr. Greenlee and colleagues examined outcomes in 13,582 breast cancer survivors with a median age of 60 years and median follow-up of 7 years to assess whether cardiovascular disease (CVD) risk associated with specific breast cancer therapies varies by body mass index (BMI) category at diagnosis.
Many routinely used breast cancer therapies are cardiotoxic, and being overweight or obese are known risk factors for CVD, but few studies have assessed whether BMI modifies the effect of these treatment on cardiovascular risk, Dr. Greenlee explained.
After adjusting for baseline demographic and health-related factors, and other breast cancer treatment, they found that:
- Ischemic heart disease risk was higher among normal-weight women who received anthracyclines, compared with those who did not (hazard ratio, 4.2). No other risk associations were observed for other breast cancer therapies and BMI groups.
- Heart failure/cardiomyopathy risk was higher among women with normal weight who received anthracyclines, cyclophosphamides, or left-sided radiation, compared with those who did not (HRs, 5.24, 3.27, and 2.05, respectively), and among overweight women who received anthracyclines, compared with those who did not (HR, 2.18). No risk associations were observed for women who received trastuzumab, taxanes, endocrine therapy, or radiation on any side, and no risk associations were observed for women who were obese.
- Stroke risk was higher in normal-weight women who received taxanes, cyclophosphamides, or left-sided radiation versus those who did not (HRs, 2.14, 2.35, and 1.31, respectively), and stroke risk was lower in obese women who received anthracyclines, taxanes, or cyclophosphamide, compared with those who did not (HRs, 0.32, 0.41, and 0.29, respectively). No risk associations were observed for trastuzumab, endocrine therapy, or radiation on any side, and no risk associations were observed for women who were overweight.
The lack of associations noted between treatments and heart failure risk among obese patients could be caused by the “obesity paradox” observed in prior obese populations, the investigators noted, adding that additional analyses are planned to “examine whether different dosage and duration of breast cancer therapy exposures across the BMI groups contributed to these risk associations.”
Breast cancer treatment and cardiometabolic effects: The role of menopausal status
In a separate analysis, Dr. Greenlee and colleagues looked at the association between endocrine therapies and cardiometabolic risk based on menopausal status.
Endocrine therapy is associated with CVD in breast cancer survivors and may be associated with developing cardiometabolic risk factors like diabetes, dyslipidemia, and hypertension, they noted, explaining that tamoxifen has mixed estrogenic and antiestrogenic activity, while aromatase inhibitors deplete endogenous estrogen.
Since most studies have compared tamoxifen with aromatase inhibitor use, it has been a challenge challenging to discern the effects of each, Dr. Greenlee said.
She and her colleagues reviewed records for 14,942 breast cancer survivors who were diagnosed between 2005 and 2013. The patients had a mean age of 61 years at baseline, and 24.9% were premenopausal at the time of diagnosis. Of the premenopausal women, 27.3% used tamoxifen, 19.2% used aromatase inhibitors, and 53.5% did not use endocrine therapy, and of the postmenopausal women, 6.6% used tamoxifen, 47.7% used aromatase inhibitors, and 45.7% did not use endocrine therapy.
After adjusting for baseline demographics and health factors, the investigators found that:
- The use of tamoxifen or aromatase inhibitors was not associated with a risk of developing diabetes, dyslipidemia, or hypertension in premenopausal women, or with a risk of developing diabetes or hypertension in postmenopausal women.
- The risk of dyslipidemia was higher in postmenopausal aromatase inhibitor users, and lower in postmenopausal tamoxifen users, compared with postmenopausal non-users of endocrine therapy (HRs, 1.15 and 0.75, respectively).
The lack of associations between endocrine therapy and CVD risk in premenopausal women may be from low power, Dr. Greenlee said, noting that analyses in larger sample sizes are needed.
She and her colleagues plan to conduct further analyses looking at treatment dosage and duration, and comparing steroidal versus nonsteroidal aromatase inhibitors.
Future studies should examine the implications of these associations on long-term CVD and how best to manage lipid profiles in postmenopausal breast cancer survivors who have a history of endocrine therapy treatment, they concluded.
This research was funded by grants from the National Cancer Institute.
For example, certain chemotherapy drugs may confer higher risk in breast cancer survivors of normal weight, whereas they may lower stroke risk in those who are obese, according to Heather Greenlee, ND, PhD, a public health researcher and naturopathic physician with the Fred Hutchinson Cancer Research Center in Seattle.
In postmenopausal women with breast cancer, aromatase inhibitors may increase cardiovascular risk, while tamoxifen appears to reduce the risk of incident dyslipidemia, she said.
The findings are from separate analyses of data from studies presented during a poster discussion session at the symposium.
Breast cancer treatment and cardiovascular effects: The role of weight
In one analysis, Dr. Greenlee and colleagues examined outcomes in 13,582 breast cancer survivors with a median age of 60 years and median follow-up of 7 years to assess whether cardiovascular disease (CVD) risk associated with specific breast cancer therapies varies by body mass index (BMI) category at diagnosis.
Many routinely used breast cancer therapies are cardiotoxic, and being overweight or obese are known risk factors for CVD, but few studies have assessed whether BMI modifies the effect of these treatment on cardiovascular risk, Dr. Greenlee explained.
After adjusting for baseline demographic and health-related factors, and other breast cancer treatment, they found that:
- Ischemic heart disease risk was higher among normal-weight women who received anthracyclines, compared with those who did not (hazard ratio, 4.2). No other risk associations were observed for other breast cancer therapies and BMI groups.
- Heart failure/cardiomyopathy risk was higher among women with normal weight who received anthracyclines, cyclophosphamides, or left-sided radiation, compared with those who did not (HRs, 5.24, 3.27, and 2.05, respectively), and among overweight women who received anthracyclines, compared with those who did not (HR, 2.18). No risk associations were observed for women who received trastuzumab, taxanes, endocrine therapy, or radiation on any side, and no risk associations were observed for women who were obese.
- Stroke risk was higher in normal-weight women who received taxanes, cyclophosphamides, or left-sided radiation versus those who did not (HRs, 2.14, 2.35, and 1.31, respectively), and stroke risk was lower in obese women who received anthracyclines, taxanes, or cyclophosphamide, compared with those who did not (HRs, 0.32, 0.41, and 0.29, respectively). No risk associations were observed for trastuzumab, endocrine therapy, or radiation on any side, and no risk associations were observed for women who were overweight.
The lack of associations noted between treatments and heart failure risk among obese patients could be caused by the “obesity paradox” observed in prior obese populations, the investigators noted, adding that additional analyses are planned to “examine whether different dosage and duration of breast cancer therapy exposures across the BMI groups contributed to these risk associations.”
Breast cancer treatment and cardiometabolic effects: The role of menopausal status
In a separate analysis, Dr. Greenlee and colleagues looked at the association between endocrine therapies and cardiometabolic risk based on menopausal status.
Endocrine therapy is associated with CVD in breast cancer survivors and may be associated with developing cardiometabolic risk factors like diabetes, dyslipidemia, and hypertension, they noted, explaining that tamoxifen has mixed estrogenic and antiestrogenic activity, while aromatase inhibitors deplete endogenous estrogen.
Since most studies have compared tamoxifen with aromatase inhibitor use, it has been a challenge challenging to discern the effects of each, Dr. Greenlee said.
She and her colleagues reviewed records for 14,942 breast cancer survivors who were diagnosed between 2005 and 2013. The patients had a mean age of 61 years at baseline, and 24.9% were premenopausal at the time of diagnosis. Of the premenopausal women, 27.3% used tamoxifen, 19.2% used aromatase inhibitors, and 53.5% did not use endocrine therapy, and of the postmenopausal women, 6.6% used tamoxifen, 47.7% used aromatase inhibitors, and 45.7% did not use endocrine therapy.
After adjusting for baseline demographics and health factors, the investigators found that:
- The use of tamoxifen or aromatase inhibitors was not associated with a risk of developing diabetes, dyslipidemia, or hypertension in premenopausal women, or with a risk of developing diabetes or hypertension in postmenopausal women.
- The risk of dyslipidemia was higher in postmenopausal aromatase inhibitor users, and lower in postmenopausal tamoxifen users, compared with postmenopausal non-users of endocrine therapy (HRs, 1.15 and 0.75, respectively).
The lack of associations between endocrine therapy and CVD risk in premenopausal women may be from low power, Dr. Greenlee said, noting that analyses in larger sample sizes are needed.
She and her colleagues plan to conduct further analyses looking at treatment dosage and duration, and comparing steroidal versus nonsteroidal aromatase inhibitors.
Future studies should examine the implications of these associations on long-term CVD and how best to manage lipid profiles in postmenopausal breast cancer survivors who have a history of endocrine therapy treatment, they concluded.
This research was funded by grants from the National Cancer Institute.
FROM SABCS 2021
WPATH draft on gender dysphoria ‘skewed and misses urgent issues’
New draft guidance from the World Professional Association for Transgender Health (WPATH) is raising serious concerns among professionals caring for people with gender dysphoria, prompting claims that WPATH is an organization “captured by activists.”
Experts in adolescent and child psychology, as well as pediatric health, have expressed dismay that the WPATH Standards of Care (SOC) 8 appear to miss some of the most urgent issues in the field of transgender medicine and are considered to express a radical and unreserved leaning towards “gender-affirmation.”
The WPATH SOC 8 document is available for view and comment until Dec. 16 until 11.59 PM EST, after which time revisions will be made and the final version published.
Despite repeated attempts by this news organization to seek clarification on certain aspects of the guidance from members of the WPATH SOC 8 committee, requests were declined “until the guidance is finalized.”
According to the WPATH website, the SOC 8 aims to provide “clinical guidance for health professionals to assist transgender and gender diverse people with safe and effective pathways” to manage their gender dysphoria and potentially transition.
Such pathways may relate to primary care, gynecologic and urologic care, reproductive options, voice and communication therapy, mental health services, and hormonal or surgical treatments, among others.
WPATH adds that it was felt necessary to revise the existing SOC 7 (published in 2012) because of recent “globally unprecedented increase and visibility of transgender and gender-diverse people seeking support and gender-affirming medical treatment.”
Gender-affirming medical treatment means different things at different ages. In the case of kids with gender dysphoria who have not yet entered puberty associated with their birth sex, this might include prescribing so-called “puberty blockers” to delay natural puberty – gonadotrophin-releasing hormone analogs that are licensed for use in precocious puberty in children. Such agents have not been licensed for use in children with gender dysphoria, however, so any use for this purpose is off-label.
Following puberty blockade – or in cases where adolescents have already undergone natural puberty – the next step is to begin cross-sex hormones. So, for a female patient who wants to transition to male (FTM), that would be lifelong testosterone, and for a male who wants to be female (MTF), it involves lifelong estrogen. Again, use of such hormones in transgender individuals is entirely off-label.
Just last month, two of America’s leading experts on transgender medicine, both psychologists – including one who is transgender – told this news organization they were concerned that the quality of the evaluations of youth with gender dysphoria are being stifled by activists who are worried that open discussions will further stigmatize trans individuals.
They subsequently wrote an op-ed on the topic entitled, “The mental health establishment is failing trans kids,” which was finally published in the Washington Post on Nov. 24, after numerous other mainstream U.S. media outlets had rejected it.
New SOC 8 ‘is not evidence based,’ should not be new ‘gold standard’
One expert says the draft SOC 8 lacks balance and does not address certain issues, while paying undue attention to others that detract from real questions facing the field of transgender medicine, both in the United States and around the world.
Julia Mason, MD, is a pediatrician based in Gresham, Oregon, with a special interest in children and adolescents experiencing gender dysphoria. “The SOC 8 shows us that WPATH remains captured by activists,” she asserts.
Dr. Mason questions the integrity of WPATH based on what she has read in the draft SOC 8.
“We need a serious organization to take a sober look at the evidence, and that is why we have established the Society for Evidence-Based Gender Medicine [SEGM],” she noted. “This is what we do – we are looking at all of the evidence.”
Dr. Mason is a clinical advisor to SEGM, an organization set-up to evaluate current interventions and evidence on gender dysphoria.
The pediatrician has particular concerns regarding the child and adolescent chapters in the draft SOC 8. The adolescent chapter states: “Guidelines are meant to provide a gold standard based on the available evidence at this moment of time.”
Dr. Mason disputes this assertion. “This document should not be the new gold standard going forward, primarily because it is not evidence based.”
In an interview, Dr. Mason explained that WPATH say they used the “Delphi consensus process” to determine their recommendations, but “this process is designed for use with a panel of experts when evidence is lacking. I would say they didn’t have a panel of experts. They largely had a panel of activists, with a few experts.”
There is no mention, for example, of England’s National Institute for Health and Care Excellence (NICE) evidence reviews on puberty blockers and cross-sex hormones from earlier this year. These reviews determined that no studies have compared cross-sex hormones or puberty blockers with a control group and all follow-up periods for cross-sex hormones were relatively short.
This disappoints Dr. Mason: “These are significant; they are important documents.”
And much of the evidence quoted comes from the well-known and often-quoted “Dutch-protocol” study of 2011, in which the children studied were much younger at the time of their gender dysphoria, compared with the many adolescents who make up the current surge in presentation at gender clinics worldwide, she adds.
Rapid-onset GD: adolescents presenting late with little history
Dr. Mason also stresses that the SOC 8 does not address the most urgent issues in transgender medicine today, mainly because it does not address rapid-onset gender dysphoria (ROGD): “This is the dilemma of the 21st century; it’s new.”
ROGD – a term first coined in 2018 by researcher Lisa Littman, MD, MPH, now president of the Institute for Comprehensive Gender Dysphoria Research (ICGDR) – refers to the phenomena of adolescents expressing a desire to transition from their birth sex after little or no apparent previous indication.
However, the SOC 8 does make reference to aspects of adolescent development that might impact their decision-making processes around gender identity during teen years. The chapter on adolescents reads: “... adolescence is also often associated with increased risk-taking behaviors. Along with these notable changes ... individuation from parents ... [there is] often a heightened focus on peer relationships, which can be both positive and detrimental.”
The guidance goes on to point out that “it is critical to understand how all of these aspects of development may impact the decision-making for a given young person within their specific cultural context.”
Desistance and detransitioning not adequately addressed
Dr. Mason also says there is little mention “about detransitioning in this SOC [8], and ‘gender dysphoria’ and ‘trans’ are terms that are not defined.”
Likewise, there is no mention of desistance, she highlights, which is when individuals naturally resolve their dysphoria around their birth sex as they grow older.
The most recent published data seen by this news organization relates to a study from March 2021 that showed nearly 88% of boys who struggled with gender identity in childhood (approximate mean age 8 years and follow-up at approximate mean age 20 years) desisted. It reads: “Of the 139 participants, 17 (12.2%) were classified as ‘persisters’ and the remaining 122 (87.8%) were classified as desisters.”
“Most children with gender dysphoria will desist and lose their concept of themselves as being the opposite gender,” Dr. Mason explains. “This is the safest path for a child – desistance.”
“Transition can turn a healthy young person into a lifelong medical patient and has significant health risks,” she emphasizes, stressing that transition has not been shown to decrease the probability of suicide, or attempts at suicide, despite myriad claims saying otherwise.
“Before we were routinely transitioning kids at school, the vast majority of children grew out of their gender dysphoria. This history is not recognized at all in these SOC [8],” she maintains.
Ken Zucker, PhD, CPsych, an author of the study of desistance in boys, says the terms desistence and persistence of gender dysphoria have caused some consternation in certain circles.
An editor of the Archives of Sexual Behavior and professor in the department of psychiatry, University of Toronto, Dr. Zucker has published widely on the topic.
He told this news organization: “The terms persistence and desistance have become verboten among the WPATH cognoscenti. Perhaps the contributors to SOC 8 have come up with alternative descriptors.”
“The term ‘desistance’ is particularly annoying to some of the gender-affirming clinicians, because they don’t believe that desistance is bona fide,” Dr. Zucker points out.
“The desistance resisters are like anti-vaxxers – nothing one can provide as evidence for the efficacy of vaccines is sufficient. There will always be a new objection.”
Other mental health issues, in particular ADHD and autism
It is also widely acknowledged that there is a higher rate of neurodevelopmental and psychiatric diagnoses in individuals with gender dysphoria. For example, one 2020 study found that transgender people were three to six times as likely to be autistic as cisgender people (those whose gender is aligned with their birth sex).
Statement one in the chapter on adolescents in draft WPATH SOC 8 does give a nod to this, pointing out that health professionals working with gender diverse adolescents “should receive training and develop expertise in autism spectrum disorders and other neurodiversity conditions.”
It also notes that in some cases “a more extended assessment process may be useful, such as for youth with more complex presentations (e.g., complicated mental health histories, co-occurring autism spectrum characteristics in particular) and an absence of experienced childhood gender incongruence.”
However, Dr. Mason stresses that underlying mental health issues are central to addressing how to manage a significant number of these patients.
“If a young person has ADHD or autism, they are not ready to make decisions about the rest of their life at age 18. Even a neurotypical young person is still developing their frontal cortex in their early 20s, and it takes longer for those with ADHD or on the autism spectrum.”
She firmly believes that the guidance does not give sufficient consideration to comorbidities in people over the age of 18.
According to their [SOC 8] guidelines, “once someone is 18 they are ready for anything,” says Dr. Mason.
Offering some explanation for the increased prevalence of ADHD and autism in those with gender dysphoria, Dr. Mason notes that children can have “hyperfocus” and those with autism will fixate on a particular area of interest. “If a child is unhappy in their life, and this can be more likely if someone is neuro-atypical, then it is likely that the individual might go online and find this one solution [for example, a transgender identity] that seems to fix everything.”
Perceptions of femininity and masculinity can also be extra challenging for a child with autism, Dr. Mason says. “It is relatively easy for an autistic girl to feel like she should be a boy because the rules of femininity are composed of nonverbal, subtle behaviors that can be difficult to pick up on,” she points out. “An autistic child who isn’t particularly good at nonverbal communication might not pick up on these and thus feel they are not very ‘female.’”
“There’s a whole lot of grass-is-greener-type thinking. Girls think boys have an easier life, and boys think girls have an easier life. I know some detransitioners who have spoken eloquently about realizing their mistake on this,” she adds.
Other parts of the SOC 8 that Dr. Mason disagrees with include the recommendation in the adolescent chapter that 14-year-olds are mature enough to start cross-sex hormones, that is, giving testosterone to a female who wants to transition to male or estrogen to a male who wishes to transition to female. “I think that’s far too young,” she asserts.
And she points out that the document states 17-year-olds are ready for genital reassignment surgery. Again, she believes this is far too young.
“Also, the SOC 8 document does not clarify who is appropriate for surgery. Whenever surgery is discussed, it becomes very vague,” she said.
A version of this article first appeared on Medscape.com.
New draft guidance from the World Professional Association for Transgender Health (WPATH) is raising serious concerns among professionals caring for people with gender dysphoria, prompting claims that WPATH is an organization “captured by activists.”
Experts in adolescent and child psychology, as well as pediatric health, have expressed dismay that the WPATH Standards of Care (SOC) 8 appear to miss some of the most urgent issues in the field of transgender medicine and are considered to express a radical and unreserved leaning towards “gender-affirmation.”
The WPATH SOC 8 document is available for view and comment until Dec. 16 until 11.59 PM EST, after which time revisions will be made and the final version published.
Despite repeated attempts by this news organization to seek clarification on certain aspects of the guidance from members of the WPATH SOC 8 committee, requests were declined “until the guidance is finalized.”
According to the WPATH website, the SOC 8 aims to provide “clinical guidance for health professionals to assist transgender and gender diverse people with safe and effective pathways” to manage their gender dysphoria and potentially transition.
Such pathways may relate to primary care, gynecologic and urologic care, reproductive options, voice and communication therapy, mental health services, and hormonal or surgical treatments, among others.
WPATH adds that it was felt necessary to revise the existing SOC 7 (published in 2012) because of recent “globally unprecedented increase and visibility of transgender and gender-diverse people seeking support and gender-affirming medical treatment.”
Gender-affirming medical treatment means different things at different ages. In the case of kids with gender dysphoria who have not yet entered puberty associated with their birth sex, this might include prescribing so-called “puberty blockers” to delay natural puberty – gonadotrophin-releasing hormone analogs that are licensed for use in precocious puberty in children. Such agents have not been licensed for use in children with gender dysphoria, however, so any use for this purpose is off-label.
Following puberty blockade – or in cases where adolescents have already undergone natural puberty – the next step is to begin cross-sex hormones. So, for a female patient who wants to transition to male (FTM), that would be lifelong testosterone, and for a male who wants to be female (MTF), it involves lifelong estrogen. Again, use of such hormones in transgender individuals is entirely off-label.
Just last month, two of America’s leading experts on transgender medicine, both psychologists – including one who is transgender – told this news organization they were concerned that the quality of the evaluations of youth with gender dysphoria are being stifled by activists who are worried that open discussions will further stigmatize trans individuals.
They subsequently wrote an op-ed on the topic entitled, “The mental health establishment is failing trans kids,” which was finally published in the Washington Post on Nov. 24, after numerous other mainstream U.S. media outlets had rejected it.
New SOC 8 ‘is not evidence based,’ should not be new ‘gold standard’
One expert says the draft SOC 8 lacks balance and does not address certain issues, while paying undue attention to others that detract from real questions facing the field of transgender medicine, both in the United States and around the world.
Julia Mason, MD, is a pediatrician based in Gresham, Oregon, with a special interest in children and adolescents experiencing gender dysphoria. “The SOC 8 shows us that WPATH remains captured by activists,” she asserts.
Dr. Mason questions the integrity of WPATH based on what she has read in the draft SOC 8.
“We need a serious organization to take a sober look at the evidence, and that is why we have established the Society for Evidence-Based Gender Medicine [SEGM],” she noted. “This is what we do – we are looking at all of the evidence.”
Dr. Mason is a clinical advisor to SEGM, an organization set-up to evaluate current interventions and evidence on gender dysphoria.
The pediatrician has particular concerns regarding the child and adolescent chapters in the draft SOC 8. The adolescent chapter states: “Guidelines are meant to provide a gold standard based on the available evidence at this moment of time.”
Dr. Mason disputes this assertion. “This document should not be the new gold standard going forward, primarily because it is not evidence based.”
In an interview, Dr. Mason explained that WPATH say they used the “Delphi consensus process” to determine their recommendations, but “this process is designed for use with a panel of experts when evidence is lacking. I would say they didn’t have a panel of experts. They largely had a panel of activists, with a few experts.”
There is no mention, for example, of England’s National Institute for Health and Care Excellence (NICE) evidence reviews on puberty blockers and cross-sex hormones from earlier this year. These reviews determined that no studies have compared cross-sex hormones or puberty blockers with a control group and all follow-up periods for cross-sex hormones were relatively short.
This disappoints Dr. Mason: “These are significant; they are important documents.”
And much of the evidence quoted comes from the well-known and often-quoted “Dutch-protocol” study of 2011, in which the children studied were much younger at the time of their gender dysphoria, compared with the many adolescents who make up the current surge in presentation at gender clinics worldwide, she adds.
Rapid-onset GD: adolescents presenting late with little history
Dr. Mason also stresses that the SOC 8 does not address the most urgent issues in transgender medicine today, mainly because it does not address rapid-onset gender dysphoria (ROGD): “This is the dilemma of the 21st century; it’s new.”
ROGD – a term first coined in 2018 by researcher Lisa Littman, MD, MPH, now president of the Institute for Comprehensive Gender Dysphoria Research (ICGDR) – refers to the phenomena of adolescents expressing a desire to transition from their birth sex after little or no apparent previous indication.
However, the SOC 8 does make reference to aspects of adolescent development that might impact their decision-making processes around gender identity during teen years. The chapter on adolescents reads: “... adolescence is also often associated with increased risk-taking behaviors. Along with these notable changes ... individuation from parents ... [there is] often a heightened focus on peer relationships, which can be both positive and detrimental.”
The guidance goes on to point out that “it is critical to understand how all of these aspects of development may impact the decision-making for a given young person within their specific cultural context.”
Desistance and detransitioning not adequately addressed
Dr. Mason also says there is little mention “about detransitioning in this SOC [8], and ‘gender dysphoria’ and ‘trans’ are terms that are not defined.”
Likewise, there is no mention of desistance, she highlights, which is when individuals naturally resolve their dysphoria around their birth sex as they grow older.
The most recent published data seen by this news organization relates to a study from March 2021 that showed nearly 88% of boys who struggled with gender identity in childhood (approximate mean age 8 years and follow-up at approximate mean age 20 years) desisted. It reads: “Of the 139 participants, 17 (12.2%) were classified as ‘persisters’ and the remaining 122 (87.8%) were classified as desisters.”
“Most children with gender dysphoria will desist and lose their concept of themselves as being the opposite gender,” Dr. Mason explains. “This is the safest path for a child – desistance.”
“Transition can turn a healthy young person into a lifelong medical patient and has significant health risks,” she emphasizes, stressing that transition has not been shown to decrease the probability of suicide, or attempts at suicide, despite myriad claims saying otherwise.
“Before we were routinely transitioning kids at school, the vast majority of children grew out of their gender dysphoria. This history is not recognized at all in these SOC [8],” she maintains.
Ken Zucker, PhD, CPsych, an author of the study of desistance in boys, says the terms desistence and persistence of gender dysphoria have caused some consternation in certain circles.
An editor of the Archives of Sexual Behavior and professor in the department of psychiatry, University of Toronto, Dr. Zucker has published widely on the topic.
He told this news organization: “The terms persistence and desistance have become verboten among the WPATH cognoscenti. Perhaps the contributors to SOC 8 have come up with alternative descriptors.”
“The term ‘desistance’ is particularly annoying to some of the gender-affirming clinicians, because they don’t believe that desistance is bona fide,” Dr. Zucker points out.
“The desistance resisters are like anti-vaxxers – nothing one can provide as evidence for the efficacy of vaccines is sufficient. There will always be a new objection.”
Other mental health issues, in particular ADHD and autism
It is also widely acknowledged that there is a higher rate of neurodevelopmental and psychiatric diagnoses in individuals with gender dysphoria. For example, one 2020 study found that transgender people were three to six times as likely to be autistic as cisgender people (those whose gender is aligned with their birth sex).
Statement one in the chapter on adolescents in draft WPATH SOC 8 does give a nod to this, pointing out that health professionals working with gender diverse adolescents “should receive training and develop expertise in autism spectrum disorders and other neurodiversity conditions.”
It also notes that in some cases “a more extended assessment process may be useful, such as for youth with more complex presentations (e.g., complicated mental health histories, co-occurring autism spectrum characteristics in particular) and an absence of experienced childhood gender incongruence.”
However, Dr. Mason stresses that underlying mental health issues are central to addressing how to manage a significant number of these patients.
“If a young person has ADHD or autism, they are not ready to make decisions about the rest of their life at age 18. Even a neurotypical young person is still developing their frontal cortex in their early 20s, and it takes longer for those with ADHD or on the autism spectrum.”
She firmly believes that the guidance does not give sufficient consideration to comorbidities in people over the age of 18.
According to their [SOC 8] guidelines, “once someone is 18 they are ready for anything,” says Dr. Mason.
Offering some explanation for the increased prevalence of ADHD and autism in those with gender dysphoria, Dr. Mason notes that children can have “hyperfocus” and those with autism will fixate on a particular area of interest. “If a child is unhappy in their life, and this can be more likely if someone is neuro-atypical, then it is likely that the individual might go online and find this one solution [for example, a transgender identity] that seems to fix everything.”
Perceptions of femininity and masculinity can also be extra challenging for a child with autism, Dr. Mason says. “It is relatively easy for an autistic girl to feel like she should be a boy because the rules of femininity are composed of nonverbal, subtle behaviors that can be difficult to pick up on,” she points out. “An autistic child who isn’t particularly good at nonverbal communication might not pick up on these and thus feel they are not very ‘female.’”
“There’s a whole lot of grass-is-greener-type thinking. Girls think boys have an easier life, and boys think girls have an easier life. I know some detransitioners who have spoken eloquently about realizing their mistake on this,” she adds.
Other parts of the SOC 8 that Dr. Mason disagrees with include the recommendation in the adolescent chapter that 14-year-olds are mature enough to start cross-sex hormones, that is, giving testosterone to a female who wants to transition to male or estrogen to a male who wishes to transition to female. “I think that’s far too young,” she asserts.
And she points out that the document states 17-year-olds are ready for genital reassignment surgery. Again, she believes this is far too young.
“Also, the SOC 8 document does not clarify who is appropriate for surgery. Whenever surgery is discussed, it becomes very vague,” she said.
A version of this article first appeared on Medscape.com.
New draft guidance from the World Professional Association for Transgender Health (WPATH) is raising serious concerns among professionals caring for people with gender dysphoria, prompting claims that WPATH is an organization “captured by activists.”
Experts in adolescent and child psychology, as well as pediatric health, have expressed dismay that the WPATH Standards of Care (SOC) 8 appear to miss some of the most urgent issues in the field of transgender medicine and are considered to express a radical and unreserved leaning towards “gender-affirmation.”
The WPATH SOC 8 document is available for view and comment until Dec. 16 until 11.59 PM EST, after which time revisions will be made and the final version published.
Despite repeated attempts by this news organization to seek clarification on certain aspects of the guidance from members of the WPATH SOC 8 committee, requests were declined “until the guidance is finalized.”
According to the WPATH website, the SOC 8 aims to provide “clinical guidance for health professionals to assist transgender and gender diverse people with safe and effective pathways” to manage their gender dysphoria and potentially transition.
Such pathways may relate to primary care, gynecologic and urologic care, reproductive options, voice and communication therapy, mental health services, and hormonal or surgical treatments, among others.
WPATH adds that it was felt necessary to revise the existing SOC 7 (published in 2012) because of recent “globally unprecedented increase and visibility of transgender and gender-diverse people seeking support and gender-affirming medical treatment.”
Gender-affirming medical treatment means different things at different ages. In the case of kids with gender dysphoria who have not yet entered puberty associated with their birth sex, this might include prescribing so-called “puberty blockers” to delay natural puberty – gonadotrophin-releasing hormone analogs that are licensed for use in precocious puberty in children. Such agents have not been licensed for use in children with gender dysphoria, however, so any use for this purpose is off-label.
Following puberty blockade – or in cases where adolescents have already undergone natural puberty – the next step is to begin cross-sex hormones. So, for a female patient who wants to transition to male (FTM), that would be lifelong testosterone, and for a male who wants to be female (MTF), it involves lifelong estrogen. Again, use of such hormones in transgender individuals is entirely off-label.
Just last month, two of America’s leading experts on transgender medicine, both psychologists – including one who is transgender – told this news organization they were concerned that the quality of the evaluations of youth with gender dysphoria are being stifled by activists who are worried that open discussions will further stigmatize trans individuals.
They subsequently wrote an op-ed on the topic entitled, “The mental health establishment is failing trans kids,” which was finally published in the Washington Post on Nov. 24, after numerous other mainstream U.S. media outlets had rejected it.
New SOC 8 ‘is not evidence based,’ should not be new ‘gold standard’
One expert says the draft SOC 8 lacks balance and does not address certain issues, while paying undue attention to others that detract from real questions facing the field of transgender medicine, both in the United States and around the world.
Julia Mason, MD, is a pediatrician based in Gresham, Oregon, with a special interest in children and adolescents experiencing gender dysphoria. “The SOC 8 shows us that WPATH remains captured by activists,” she asserts.
Dr. Mason questions the integrity of WPATH based on what she has read in the draft SOC 8.
“We need a serious organization to take a sober look at the evidence, and that is why we have established the Society for Evidence-Based Gender Medicine [SEGM],” she noted. “This is what we do – we are looking at all of the evidence.”
Dr. Mason is a clinical advisor to SEGM, an organization set-up to evaluate current interventions and evidence on gender dysphoria.
The pediatrician has particular concerns regarding the child and adolescent chapters in the draft SOC 8. The adolescent chapter states: “Guidelines are meant to provide a gold standard based on the available evidence at this moment of time.”
Dr. Mason disputes this assertion. “This document should not be the new gold standard going forward, primarily because it is not evidence based.”
In an interview, Dr. Mason explained that WPATH say they used the “Delphi consensus process” to determine their recommendations, but “this process is designed for use with a panel of experts when evidence is lacking. I would say they didn’t have a panel of experts. They largely had a panel of activists, with a few experts.”
There is no mention, for example, of England’s National Institute for Health and Care Excellence (NICE) evidence reviews on puberty blockers and cross-sex hormones from earlier this year. These reviews determined that no studies have compared cross-sex hormones or puberty blockers with a control group and all follow-up periods for cross-sex hormones were relatively short.
This disappoints Dr. Mason: “These are significant; they are important documents.”
And much of the evidence quoted comes from the well-known and often-quoted “Dutch-protocol” study of 2011, in which the children studied were much younger at the time of their gender dysphoria, compared with the many adolescents who make up the current surge in presentation at gender clinics worldwide, she adds.
Rapid-onset GD: adolescents presenting late with little history
Dr. Mason also stresses that the SOC 8 does not address the most urgent issues in transgender medicine today, mainly because it does not address rapid-onset gender dysphoria (ROGD): “This is the dilemma of the 21st century; it’s new.”
ROGD – a term first coined in 2018 by researcher Lisa Littman, MD, MPH, now president of the Institute for Comprehensive Gender Dysphoria Research (ICGDR) – refers to the phenomena of adolescents expressing a desire to transition from their birth sex after little or no apparent previous indication.
However, the SOC 8 does make reference to aspects of adolescent development that might impact their decision-making processes around gender identity during teen years. The chapter on adolescents reads: “... adolescence is also often associated with increased risk-taking behaviors. Along with these notable changes ... individuation from parents ... [there is] often a heightened focus on peer relationships, which can be both positive and detrimental.”
The guidance goes on to point out that “it is critical to understand how all of these aspects of development may impact the decision-making for a given young person within their specific cultural context.”
Desistance and detransitioning not adequately addressed
Dr. Mason also says there is little mention “about detransitioning in this SOC [8], and ‘gender dysphoria’ and ‘trans’ are terms that are not defined.”
Likewise, there is no mention of desistance, she highlights, which is when individuals naturally resolve their dysphoria around their birth sex as they grow older.
The most recent published data seen by this news organization relates to a study from March 2021 that showed nearly 88% of boys who struggled with gender identity in childhood (approximate mean age 8 years and follow-up at approximate mean age 20 years) desisted. It reads: “Of the 139 participants, 17 (12.2%) were classified as ‘persisters’ and the remaining 122 (87.8%) were classified as desisters.”
“Most children with gender dysphoria will desist and lose their concept of themselves as being the opposite gender,” Dr. Mason explains. “This is the safest path for a child – desistance.”
“Transition can turn a healthy young person into a lifelong medical patient and has significant health risks,” she emphasizes, stressing that transition has not been shown to decrease the probability of suicide, or attempts at suicide, despite myriad claims saying otherwise.
“Before we were routinely transitioning kids at school, the vast majority of children grew out of their gender dysphoria. This history is not recognized at all in these SOC [8],” she maintains.
Ken Zucker, PhD, CPsych, an author of the study of desistance in boys, says the terms desistence and persistence of gender dysphoria have caused some consternation in certain circles.
An editor of the Archives of Sexual Behavior and professor in the department of psychiatry, University of Toronto, Dr. Zucker has published widely on the topic.
He told this news organization: “The terms persistence and desistance have become verboten among the WPATH cognoscenti. Perhaps the contributors to SOC 8 have come up with alternative descriptors.”
“The term ‘desistance’ is particularly annoying to some of the gender-affirming clinicians, because they don’t believe that desistance is bona fide,” Dr. Zucker points out.
“The desistance resisters are like anti-vaxxers – nothing one can provide as evidence for the efficacy of vaccines is sufficient. There will always be a new objection.”
Other mental health issues, in particular ADHD and autism
It is also widely acknowledged that there is a higher rate of neurodevelopmental and psychiatric diagnoses in individuals with gender dysphoria. For example, one 2020 study found that transgender people were three to six times as likely to be autistic as cisgender people (those whose gender is aligned with their birth sex).
Statement one in the chapter on adolescents in draft WPATH SOC 8 does give a nod to this, pointing out that health professionals working with gender diverse adolescents “should receive training and develop expertise in autism spectrum disorders and other neurodiversity conditions.”
It also notes that in some cases “a more extended assessment process may be useful, such as for youth with more complex presentations (e.g., complicated mental health histories, co-occurring autism spectrum characteristics in particular) and an absence of experienced childhood gender incongruence.”
However, Dr. Mason stresses that underlying mental health issues are central to addressing how to manage a significant number of these patients.
“If a young person has ADHD or autism, they are not ready to make decisions about the rest of their life at age 18. Even a neurotypical young person is still developing their frontal cortex in their early 20s, and it takes longer for those with ADHD or on the autism spectrum.”
She firmly believes that the guidance does not give sufficient consideration to comorbidities in people over the age of 18.
According to their [SOC 8] guidelines, “once someone is 18 they are ready for anything,” says Dr. Mason.
Offering some explanation for the increased prevalence of ADHD and autism in those with gender dysphoria, Dr. Mason notes that children can have “hyperfocus” and those with autism will fixate on a particular area of interest. “If a child is unhappy in their life, and this can be more likely if someone is neuro-atypical, then it is likely that the individual might go online and find this one solution [for example, a transgender identity] that seems to fix everything.”
Perceptions of femininity and masculinity can also be extra challenging for a child with autism, Dr. Mason says. “It is relatively easy for an autistic girl to feel like she should be a boy because the rules of femininity are composed of nonverbal, subtle behaviors that can be difficult to pick up on,” she points out. “An autistic child who isn’t particularly good at nonverbal communication might not pick up on these and thus feel they are not very ‘female.’”
“There’s a whole lot of grass-is-greener-type thinking. Girls think boys have an easier life, and boys think girls have an easier life. I know some detransitioners who have spoken eloquently about realizing their mistake on this,” she adds.
Other parts of the SOC 8 that Dr. Mason disagrees with include the recommendation in the adolescent chapter that 14-year-olds are mature enough to start cross-sex hormones, that is, giving testosterone to a female who wants to transition to male or estrogen to a male who wishes to transition to female. “I think that’s far too young,” she asserts.
And she points out that the document states 17-year-olds are ready for genital reassignment surgery. Again, she believes this is far too young.
“Also, the SOC 8 document does not clarify who is appropriate for surgery. Whenever surgery is discussed, it becomes very vague,” she said.
A version of this article first appeared on Medscape.com.
Study finds more adverse maternal outcomes in women with disabilities
Women with physical, intellectual, and sensory disabilities had higher risk for almost all pregnancy complications, obstetric interventions, and adverse outcomes, including severe maternal morbidity (SMM) and mortality compared to women without disabilities, according to an analysis of a large, retrospective cohort.
The findings, published in JAMA Network Open (2021;4[12]:e2138414 doi: 10.1001/jamanetworkopen.2021.38414), “may be a direct reflection of the challenges women with all types of disabilities face when accessing and receiving care, which is likely compounded by poorer preconception health,” suggested lead author Jessica L. Gleason, PhD, MPH, and co-authors, all from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, Md.
“Women with disabilities have long been ignored in obstetric research and clinical practice,” added Hilary K. Brown, PhD, from the University of Toronto, in an accompanying editorial. “Inclusion of disability indicators needs to be the norm – not the exception – in health administrative data so that these disparities can be regularly tracked and addressed.”
The investigators used data from the Consortium on Safe Labor (CSL), a retrospective cohort of deliveries from 12 U.S. clinical centers between Jan. 2002 and Jan. 2008, to analyze obstetric interventions and adverse maternal outcomes in women with and without disabilities.
The analysis included a total of 223,385 women, mean age 27.6 years, of whom 2,074 (0.9%) had a disability, and 221,311 did not. Among those with disabilities, 1,733 (83.5%) were physical, 91 (4.4%) were intellectual, and 250 (12.1%) were sensory. While almost half (49.4%) of the women were White, 22.5% were Black, 17.5% were Hispanic, and 4.1% were Asian or Pacific Islander.
Outcomes were analyzed with three composite measures:
- Pregnancy-related complications (pregnancy-related hypertensive diseases, gestational diabetes, placental abruption, placenta previa, premature rupture of membranes, preterm PROM);
- All labor, delivery, and postpartum complications (chorioamnionitis, hemorrhage, blood transfusion, thromboembolism, postpartum fever, infection, cardiovascular events, cardiomyopathy, and maternal death);
- SMM only, including severe pre-eclampsia/eclampsia, hemorrhage, thromboembolism, fever, infection, cardiomyopathy, and cardiovascular events during labor and delivery.
After adjustment for covariates, women with disabilities had higher risk of pregnancy-related complications. This included a 48% higher risk of mild pre-eclampsia and double the risk of severe pre-eclampsia/eclampsia. The composite risk of any pregnancy complication was 27% higher for women with physical disabilities, 49% higher for women with intellectual disabilities, and 53% higher for women with sensory disabilities.
The findings were similar for labor, delivery, and postpartum complications, showing women with disabilities had higher risk for a range of obstetrical interventions, including cesarean delivery – both planned and intrapartum (aRR, 1.34). Additionally, women with disabilities were less likely to have a cesarean delivery that was “solely clinically indicated” (aRR, 0.79), and more likely to have a cesarean delivery for “softer” mixed indication (aRR, 1.16), “supporting a possible overuse of cesarean delivery among women with disability,” they suggested.
Women with disabilities also had a higher risk of postpartum hemorrhage (aRR, 1.27), blood transfusion (aRR, 1.64), and maternal mortality (aRR, 11.19), as well as individual markers of severe maternal morbidity, such as cardiovascular events (aRR, 4.02), infection (aRR, 2.69), and venous thromboembolism (aRR, 6.08).
The authors speculate that the increased risks for women with disabilities “may be the result of a combination of independent risk factors, including the higher rate of obstetric intervention via cesarean delivery, under-recognition of women with disabilities as a population with higher-risk pregnancies, and lack of health care practitioner knowledge or comfort in managing pregnancies among women with disabilities.”
Dr. Brown noted in her commentary that there is a need for better education of health care professionals in this area. “Given that 12% of reproductive-aged women have a disability, that pregnancy rates are similar among women with and without disabilities, and that women with disabilities are at elevated risk of a range of adverse maternal outcomes, including severe maternal morbidity and maternal mortality, disability modules should be a mandatory component of education for obstetricians and midwives as well as other obstetrical health care professionals.”
Calling the study “a serious wake-up call,” Monika Mitra, PhD, told this publication that the findings highlight the need for “urgent attention” on improving obstetric care for people with disabilities “with a focus on accessibility and inclusion, changing clinical practice to better serve disabled people, integrating disability-related training for health care practitioners, and developing evidence-based interventions to support people with disabilities during this time.” The associate professor and director of the Lurie Institute for Disability Policy, in Brandeis University, Waltham, Mass. said the risk factors for poor outcomes are present early in pregnancy or even preconception. “We know that disabled women report barriers in accessing health care and receive lower-quality care compared to nondisabled women and are more likely to experience poverty, housing and food insecurity, educational and employment barriers, abuse, chronic health conditions, and mental illness than women without disabilities.”
She noted that the study’s sample of people with disabilities was small, and the measure of disability used was based on ICD-9 codes, which captures only severe disabilities. “As noted in the commentary by [Dr.] Brown, our standard sources of health administrative data do not give us the full picture on disability, and we need other, more equitable ways of identifying disability based, for example, on self-reports of activity or participation limitations if we are to be able to understand the effects on obstetric outcomes of health and health care disparities and of social determinants of health. Moreover, researchers have generally not yet begun to incorporate knowledge of the experiences of transgender people during pregnancy, which will impact our measures and study of obstetric outcomes among people with disabilities as well as the language we use.”
The study was supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). The study authors and Dr. Brown reported no conflicts of interest. Dr. Mitra receives funding from the NICHD and the National Institute on Disability, Independent Living for research on pregnancy outcomes among people with disabilities.
Women with physical, intellectual, and sensory disabilities had higher risk for almost all pregnancy complications, obstetric interventions, and adverse outcomes, including severe maternal morbidity (SMM) and mortality compared to women without disabilities, according to an analysis of a large, retrospective cohort.
The findings, published in JAMA Network Open (2021;4[12]:e2138414 doi: 10.1001/jamanetworkopen.2021.38414), “may be a direct reflection of the challenges women with all types of disabilities face when accessing and receiving care, which is likely compounded by poorer preconception health,” suggested lead author Jessica L. Gleason, PhD, MPH, and co-authors, all from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, Md.
“Women with disabilities have long been ignored in obstetric research and clinical practice,” added Hilary K. Brown, PhD, from the University of Toronto, in an accompanying editorial. “Inclusion of disability indicators needs to be the norm – not the exception – in health administrative data so that these disparities can be regularly tracked and addressed.”
The investigators used data from the Consortium on Safe Labor (CSL), a retrospective cohort of deliveries from 12 U.S. clinical centers between Jan. 2002 and Jan. 2008, to analyze obstetric interventions and adverse maternal outcomes in women with and without disabilities.
The analysis included a total of 223,385 women, mean age 27.6 years, of whom 2,074 (0.9%) had a disability, and 221,311 did not. Among those with disabilities, 1,733 (83.5%) were physical, 91 (4.4%) were intellectual, and 250 (12.1%) were sensory. While almost half (49.4%) of the women were White, 22.5% were Black, 17.5% were Hispanic, and 4.1% were Asian or Pacific Islander.
Outcomes were analyzed with three composite measures:
- Pregnancy-related complications (pregnancy-related hypertensive diseases, gestational diabetes, placental abruption, placenta previa, premature rupture of membranes, preterm PROM);
- All labor, delivery, and postpartum complications (chorioamnionitis, hemorrhage, blood transfusion, thromboembolism, postpartum fever, infection, cardiovascular events, cardiomyopathy, and maternal death);
- SMM only, including severe pre-eclampsia/eclampsia, hemorrhage, thromboembolism, fever, infection, cardiomyopathy, and cardiovascular events during labor and delivery.
After adjustment for covariates, women with disabilities had higher risk of pregnancy-related complications. This included a 48% higher risk of mild pre-eclampsia and double the risk of severe pre-eclampsia/eclampsia. The composite risk of any pregnancy complication was 27% higher for women with physical disabilities, 49% higher for women with intellectual disabilities, and 53% higher for women with sensory disabilities.
The findings were similar for labor, delivery, and postpartum complications, showing women with disabilities had higher risk for a range of obstetrical interventions, including cesarean delivery – both planned and intrapartum (aRR, 1.34). Additionally, women with disabilities were less likely to have a cesarean delivery that was “solely clinically indicated” (aRR, 0.79), and more likely to have a cesarean delivery for “softer” mixed indication (aRR, 1.16), “supporting a possible overuse of cesarean delivery among women with disability,” they suggested.
Women with disabilities also had a higher risk of postpartum hemorrhage (aRR, 1.27), blood transfusion (aRR, 1.64), and maternal mortality (aRR, 11.19), as well as individual markers of severe maternal morbidity, such as cardiovascular events (aRR, 4.02), infection (aRR, 2.69), and venous thromboembolism (aRR, 6.08).
The authors speculate that the increased risks for women with disabilities “may be the result of a combination of independent risk factors, including the higher rate of obstetric intervention via cesarean delivery, under-recognition of women with disabilities as a population with higher-risk pregnancies, and lack of health care practitioner knowledge or comfort in managing pregnancies among women with disabilities.”
Dr. Brown noted in her commentary that there is a need for better education of health care professionals in this area. “Given that 12% of reproductive-aged women have a disability, that pregnancy rates are similar among women with and without disabilities, and that women with disabilities are at elevated risk of a range of adverse maternal outcomes, including severe maternal morbidity and maternal mortality, disability modules should be a mandatory component of education for obstetricians and midwives as well as other obstetrical health care professionals.”
Calling the study “a serious wake-up call,” Monika Mitra, PhD, told this publication that the findings highlight the need for “urgent attention” on improving obstetric care for people with disabilities “with a focus on accessibility and inclusion, changing clinical practice to better serve disabled people, integrating disability-related training for health care practitioners, and developing evidence-based interventions to support people with disabilities during this time.” The associate professor and director of the Lurie Institute for Disability Policy, in Brandeis University, Waltham, Mass. said the risk factors for poor outcomes are present early in pregnancy or even preconception. “We know that disabled women report barriers in accessing health care and receive lower-quality care compared to nondisabled women and are more likely to experience poverty, housing and food insecurity, educational and employment barriers, abuse, chronic health conditions, and mental illness than women without disabilities.”
She noted that the study’s sample of people with disabilities was small, and the measure of disability used was based on ICD-9 codes, which captures only severe disabilities. “As noted in the commentary by [Dr.] Brown, our standard sources of health administrative data do not give us the full picture on disability, and we need other, more equitable ways of identifying disability based, for example, on self-reports of activity or participation limitations if we are to be able to understand the effects on obstetric outcomes of health and health care disparities and of social determinants of health. Moreover, researchers have generally not yet begun to incorporate knowledge of the experiences of transgender people during pregnancy, which will impact our measures and study of obstetric outcomes among people with disabilities as well as the language we use.”
The study was supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). The study authors and Dr. Brown reported no conflicts of interest. Dr. Mitra receives funding from the NICHD and the National Institute on Disability, Independent Living for research on pregnancy outcomes among people with disabilities.
Women with physical, intellectual, and sensory disabilities had higher risk for almost all pregnancy complications, obstetric interventions, and adverse outcomes, including severe maternal morbidity (SMM) and mortality compared to women without disabilities, according to an analysis of a large, retrospective cohort.
The findings, published in JAMA Network Open (2021;4[12]:e2138414 doi: 10.1001/jamanetworkopen.2021.38414), “may be a direct reflection of the challenges women with all types of disabilities face when accessing and receiving care, which is likely compounded by poorer preconception health,” suggested lead author Jessica L. Gleason, PhD, MPH, and co-authors, all from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, Md.
“Women with disabilities have long been ignored in obstetric research and clinical practice,” added Hilary K. Brown, PhD, from the University of Toronto, in an accompanying editorial. “Inclusion of disability indicators needs to be the norm – not the exception – in health administrative data so that these disparities can be regularly tracked and addressed.”
The investigators used data from the Consortium on Safe Labor (CSL), a retrospective cohort of deliveries from 12 U.S. clinical centers between Jan. 2002 and Jan. 2008, to analyze obstetric interventions and adverse maternal outcomes in women with and without disabilities.
The analysis included a total of 223,385 women, mean age 27.6 years, of whom 2,074 (0.9%) had a disability, and 221,311 did not. Among those with disabilities, 1,733 (83.5%) were physical, 91 (4.4%) were intellectual, and 250 (12.1%) were sensory. While almost half (49.4%) of the women were White, 22.5% were Black, 17.5% were Hispanic, and 4.1% were Asian or Pacific Islander.
Outcomes were analyzed with three composite measures:
- Pregnancy-related complications (pregnancy-related hypertensive diseases, gestational diabetes, placental abruption, placenta previa, premature rupture of membranes, preterm PROM);
- All labor, delivery, and postpartum complications (chorioamnionitis, hemorrhage, blood transfusion, thromboembolism, postpartum fever, infection, cardiovascular events, cardiomyopathy, and maternal death);
- SMM only, including severe pre-eclampsia/eclampsia, hemorrhage, thromboembolism, fever, infection, cardiomyopathy, and cardiovascular events during labor and delivery.
After adjustment for covariates, women with disabilities had higher risk of pregnancy-related complications. This included a 48% higher risk of mild pre-eclampsia and double the risk of severe pre-eclampsia/eclampsia. The composite risk of any pregnancy complication was 27% higher for women with physical disabilities, 49% higher for women with intellectual disabilities, and 53% higher for women with sensory disabilities.
The findings were similar for labor, delivery, and postpartum complications, showing women with disabilities had higher risk for a range of obstetrical interventions, including cesarean delivery – both planned and intrapartum (aRR, 1.34). Additionally, women with disabilities were less likely to have a cesarean delivery that was “solely clinically indicated” (aRR, 0.79), and more likely to have a cesarean delivery for “softer” mixed indication (aRR, 1.16), “supporting a possible overuse of cesarean delivery among women with disability,” they suggested.
Women with disabilities also had a higher risk of postpartum hemorrhage (aRR, 1.27), blood transfusion (aRR, 1.64), and maternal mortality (aRR, 11.19), as well as individual markers of severe maternal morbidity, such as cardiovascular events (aRR, 4.02), infection (aRR, 2.69), and venous thromboembolism (aRR, 6.08).
The authors speculate that the increased risks for women with disabilities “may be the result of a combination of independent risk factors, including the higher rate of obstetric intervention via cesarean delivery, under-recognition of women with disabilities as a population with higher-risk pregnancies, and lack of health care practitioner knowledge or comfort in managing pregnancies among women with disabilities.”
Dr. Brown noted in her commentary that there is a need for better education of health care professionals in this area. “Given that 12% of reproductive-aged women have a disability, that pregnancy rates are similar among women with and without disabilities, and that women with disabilities are at elevated risk of a range of adverse maternal outcomes, including severe maternal morbidity and maternal mortality, disability modules should be a mandatory component of education for obstetricians and midwives as well as other obstetrical health care professionals.”
Calling the study “a serious wake-up call,” Monika Mitra, PhD, told this publication that the findings highlight the need for “urgent attention” on improving obstetric care for people with disabilities “with a focus on accessibility and inclusion, changing clinical practice to better serve disabled people, integrating disability-related training for health care practitioners, and developing evidence-based interventions to support people with disabilities during this time.” The associate professor and director of the Lurie Institute for Disability Policy, in Brandeis University, Waltham, Mass. said the risk factors for poor outcomes are present early in pregnancy or even preconception. “We know that disabled women report barriers in accessing health care and receive lower-quality care compared to nondisabled women and are more likely to experience poverty, housing and food insecurity, educational and employment barriers, abuse, chronic health conditions, and mental illness than women without disabilities.”
She noted that the study’s sample of people with disabilities was small, and the measure of disability used was based on ICD-9 codes, which captures only severe disabilities. “As noted in the commentary by [Dr.] Brown, our standard sources of health administrative data do not give us the full picture on disability, and we need other, more equitable ways of identifying disability based, for example, on self-reports of activity or participation limitations if we are to be able to understand the effects on obstetric outcomes of health and health care disparities and of social determinants of health. Moreover, researchers have generally not yet begun to incorporate knowledge of the experiences of transgender people during pregnancy, which will impact our measures and study of obstetric outcomes among people with disabilities as well as the language we use.”
The study was supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). The study authors and Dr. Brown reported no conflicts of interest. Dr. Mitra receives funding from the NICHD and the National Institute on Disability, Independent Living for research on pregnancy outcomes among people with disabilities.
CDC panel backs mRNA COVID vaccines over J&J because of clot risk
because the Johnson & Johnson shot carries the risk of a rare but potentially fatal side effect that causes blood clots and bleeding in the brain.
In an emergency meeting on December 16, the CDC’s Advisory Committee on Immunization Practices, or ACIP, voted unanimously (15-0) to state a preference for the mRNA vaccines over the Johnson & Johnson shot. The vote came after the panel heard a safety update on cases of thrombosis with thrombocytopenia syndrome, or TTS, a condition that causes large clots that deplete the blood of platelets, resulting in uncontrolled bleeding.
The move brings the United States in line with other wealthy countries. In May, Denmark dropped the Johnson & Johnson shot from its vaccination program because of this risk. Australia and Greece have limited the use of a similar vaccine, made by AstraZeneca, in younger people because of the TTS risk. Both vaccines use the envelope of a different kind of virus, called an adenovirus, to sneak the vaccine instructions into cells. On Dec. 16, health officials said they had determined that TTS was likely due to a class effect, meaning it happens with all adenovirus vector vaccines.
The risk of dying from TTS after a Johnson & Johnson shot is extremely rare. There is an estimated 1 death for every 2 million doses of the vaccine given in the general population. That risk is higher for women ages 30 to 49, rising to about 2 deaths for every 1 million doses given in this age group. There’s no question that the Johnson & Johnson shot has saved many more lives than it has taken, experts said
Still, the committee previously paused the use of the Johnson & Johnson vaccine in April after the first cases of TTS came to light. That pause was lifted just 10 days later, after a new warning was added to the vaccine’s label to raise awareness of the risk.
In updating the safety information on Johnson & Johnson, the panel noted that the warning label had not sufficiently lowered the risk of death from TTS. Doctors seem to be aware of the condition because none of the patients who had developed TTS had been treated with the blood thinner heparin, which can make the syndrome worse. But patients continued to die even after the label was added, the panel noted, because TTS can progress so quickly that doctors simply don’t have time to treat it.
For that reason, and because there are other, safer vaccines available, the panel decided to make what’s called a preferential statement, saying the Pfizer and Moderna mRNA vaccines should be preferred over Johnson & Johnson.
The statement leaves the J&J vaccine on the market and available to patients who are at risk of a severe allergic reaction to the mRNA vaccines. It also means that people can still choose the J&J vaccine if they still want it after being informed about the risks.
About 17 million first doses and 900,000 second doses of the Johnson & Johnson vaccine have been given in the United States. Through the end of August, 54 cases of thrombosis with thrombocytopenia syndrome (TTS) have occurred after the J&J shots in the United States. Nearly half of those were in women ages 30 to 49. There have been nine deaths from TTS after Johnson & Johnson shots.
A version of this article first appeared on WebMD.com.
because the Johnson & Johnson shot carries the risk of a rare but potentially fatal side effect that causes blood clots and bleeding in the brain.
In an emergency meeting on December 16, the CDC’s Advisory Committee on Immunization Practices, or ACIP, voted unanimously (15-0) to state a preference for the mRNA vaccines over the Johnson & Johnson shot. The vote came after the panel heard a safety update on cases of thrombosis with thrombocytopenia syndrome, or TTS, a condition that causes large clots that deplete the blood of platelets, resulting in uncontrolled bleeding.
The move brings the United States in line with other wealthy countries. In May, Denmark dropped the Johnson & Johnson shot from its vaccination program because of this risk. Australia and Greece have limited the use of a similar vaccine, made by AstraZeneca, in younger people because of the TTS risk. Both vaccines use the envelope of a different kind of virus, called an adenovirus, to sneak the vaccine instructions into cells. On Dec. 16, health officials said they had determined that TTS was likely due to a class effect, meaning it happens with all adenovirus vector vaccines.
The risk of dying from TTS after a Johnson & Johnson shot is extremely rare. There is an estimated 1 death for every 2 million doses of the vaccine given in the general population. That risk is higher for women ages 30 to 49, rising to about 2 deaths for every 1 million doses given in this age group. There’s no question that the Johnson & Johnson shot has saved many more lives than it has taken, experts said
Still, the committee previously paused the use of the Johnson & Johnson vaccine in April after the first cases of TTS came to light. That pause was lifted just 10 days later, after a new warning was added to the vaccine’s label to raise awareness of the risk.
In updating the safety information on Johnson & Johnson, the panel noted that the warning label had not sufficiently lowered the risk of death from TTS. Doctors seem to be aware of the condition because none of the patients who had developed TTS had been treated with the blood thinner heparin, which can make the syndrome worse. But patients continued to die even after the label was added, the panel noted, because TTS can progress so quickly that doctors simply don’t have time to treat it.
For that reason, and because there are other, safer vaccines available, the panel decided to make what’s called a preferential statement, saying the Pfizer and Moderna mRNA vaccines should be preferred over Johnson & Johnson.
The statement leaves the J&J vaccine on the market and available to patients who are at risk of a severe allergic reaction to the mRNA vaccines. It also means that people can still choose the J&J vaccine if they still want it after being informed about the risks.
About 17 million first doses and 900,000 second doses of the Johnson & Johnson vaccine have been given in the United States. Through the end of August, 54 cases of thrombosis with thrombocytopenia syndrome (TTS) have occurred after the J&J shots in the United States. Nearly half of those were in women ages 30 to 49. There have been nine deaths from TTS after Johnson & Johnson shots.
A version of this article first appeared on WebMD.com.
because the Johnson & Johnson shot carries the risk of a rare but potentially fatal side effect that causes blood clots and bleeding in the brain.
In an emergency meeting on December 16, the CDC’s Advisory Committee on Immunization Practices, or ACIP, voted unanimously (15-0) to state a preference for the mRNA vaccines over the Johnson & Johnson shot. The vote came after the panel heard a safety update on cases of thrombosis with thrombocytopenia syndrome, or TTS, a condition that causes large clots that deplete the blood of platelets, resulting in uncontrolled bleeding.
The move brings the United States in line with other wealthy countries. In May, Denmark dropped the Johnson & Johnson shot from its vaccination program because of this risk. Australia and Greece have limited the use of a similar vaccine, made by AstraZeneca, in younger people because of the TTS risk. Both vaccines use the envelope of a different kind of virus, called an adenovirus, to sneak the vaccine instructions into cells. On Dec. 16, health officials said they had determined that TTS was likely due to a class effect, meaning it happens with all adenovirus vector vaccines.
The risk of dying from TTS after a Johnson & Johnson shot is extremely rare. There is an estimated 1 death for every 2 million doses of the vaccine given in the general population. That risk is higher for women ages 30 to 49, rising to about 2 deaths for every 1 million doses given in this age group. There’s no question that the Johnson & Johnson shot has saved many more lives than it has taken, experts said
Still, the committee previously paused the use of the Johnson & Johnson vaccine in April after the first cases of TTS came to light. That pause was lifted just 10 days later, after a new warning was added to the vaccine’s label to raise awareness of the risk.
In updating the safety information on Johnson & Johnson, the panel noted that the warning label had not sufficiently lowered the risk of death from TTS. Doctors seem to be aware of the condition because none of the patients who had developed TTS had been treated with the blood thinner heparin, which can make the syndrome worse. But patients continued to die even after the label was added, the panel noted, because TTS can progress so quickly that doctors simply don’t have time to treat it.
For that reason, and because there are other, safer vaccines available, the panel decided to make what’s called a preferential statement, saying the Pfizer and Moderna mRNA vaccines should be preferred over Johnson & Johnson.
The statement leaves the J&J vaccine on the market and available to patients who are at risk of a severe allergic reaction to the mRNA vaccines. It also means that people can still choose the J&J vaccine if they still want it after being informed about the risks.
About 17 million first doses and 900,000 second doses of the Johnson & Johnson vaccine have been given in the United States. Through the end of August, 54 cases of thrombosis with thrombocytopenia syndrome (TTS) have occurred after the J&J shots in the United States. Nearly half of those were in women ages 30 to 49. There have been nine deaths from TTS after Johnson & Johnson shots.
A version of this article first appeared on WebMD.com.
Infectious disease pop quiz: Clinical challenge #6 for the ObGyn
Which vaccines are contraindicated in pregnancy?
Continue to the answer...
Live virus vaccines should not be used in pregnancy because of the possibility of teratogenic effects. Live agents include the measles, mumps, and rubella (MMR) vaccine; live influenza vaccine (FluMist); oral polio vaccine; BCG (bacille Calmette-Guerin) vaccine; yellow fever vaccine; and smallpox vaccine.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
Which vaccines are contraindicated in pregnancy?
Continue to the answer...
Live virus vaccines should not be used in pregnancy because of the possibility of teratogenic effects. Live agents include the measles, mumps, and rubella (MMR) vaccine; live influenza vaccine (FluMist); oral polio vaccine; BCG (bacille Calmette-Guerin) vaccine; yellow fever vaccine; and smallpox vaccine.
Which vaccines are contraindicated in pregnancy?
Continue to the answer...
Live virus vaccines should not be used in pregnancy because of the possibility of teratogenic effects. Live agents include the measles, mumps, and rubella (MMR) vaccine; live influenza vaccine (FluMist); oral polio vaccine; BCG (bacille Calmette-Guerin) vaccine; yellow fever vaccine; and smallpox vaccine.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
Califf plans work on opioids, accelerated approvals on return to FDA
Robert M. Califf, MD, plans to take a close look at federal policies on opioid prescriptions in his expected second turn as the top U.S. regulator of medical products, as well as keep closer tabs on the performance of drugs cleared with accelerated approvals.
Dr. Califf on Tuesday fielded questions at a Senate hearing about his nomination by President Joe Biden to serve as administrator of the U.S. Food and Drug Administration, a role in which he served in the Obama administration. He also spoke about the need to bolster the nation’s ability to maintain an adequate supply of key medical products, including drugs.
Members of the Senate Health, Education, Labor and Pensions Committee, which is handling Dr. Califf’s nomination, were largely cordial and supportive during the hearing. Sen. Patty Murray (D-Wash.), the committee chair, and the panel’s top Republican, Sen. Richard Burr of North Carolina, addressed Dr. Califf during the hearing as if he would soon serve again as the FDA’s leader. Both were among the senators who voted 89-4 to confirm Dr. Califf in a February 2016 vote.
Dr. Califf “was previously confirmed to lead FDA in an overwhelming bipartisan vote, and I look forward to working with him again to ensure FDA continues to protect families across the country, uphold the gold standard of safety and effectiveness, and put science and data first,” Sen. Murray said.
Less enthusiastic about Dr. Califf was Sen. Bernie Sanders (I-VT), who was among the seven senators who did not vote on Dr. Califf’s nomination in 2016.
Sen. Sanders objected in 2016 to Dr. Califf’s ties to the pharmaceutical industry, and he did so again Tuesday. A noted leader in conducting clinical trials, Dr. Califf has worked with many drugmakers. But at the hearing, Dr. Califf said he concurs with Sen. Sanders on an idea strongly opposed by the pharmaceutical industry.
In response to Sen. Sanders’ question, Dr. Califf said he already is “on record as being in favor of Medicare negotiating with the industry on prices.”
The FDA would not take direct part in negotiations, as this work would be handled by the Centers for Medicare & Medicaid Services. Democrats want to give Medicare some negotiating authority through their sweeping Build Back Better Act.
People in the United States are dismayed over both the cost of prescription drugs and the widespread distribution of prescription painkillers that helped fuel the current opioid epidemic, Sen. Sanders told Dr. Califf. Many people will be concerned about an FDA commissioner who has benefited from close ties to the industry, Sen. Sanders said.
“How are they going to believe that you’re going to be an independent and strong voice against this enormously powerful, special interest?” Sen. Sanders asked.
“I’m totally with you on the concept that the price of pharmaceuticals is way too high in this country,” Dr. Califf said in reply.
Dr. Califf was paid $2.7 million in salary and bonus by Verily Life Sciences, the biomedical research organization operated by Alphabet, parent company of Google, according to his federal financial disclosure. He also reported holding board positions with pharmaceutical companies AmyriAD and Centessa Pharmaceuticals.
Bloomberg Government reported that Dr. Califf has ties to about 16 other research organizations and biotech companies. Bloomberg Government also said that, in his earlier FDA service, Dr. Califf kept a whiteboard in his office that listed all the activities and projects that required his recusal, citing as a source Howard Sklamberg, who was a deputy commissioner under Dr. Califf.
“He was very, very, very careful,” Mr. Sklamberg, who’s now an attorney at Arnold & Porter LLP, told Bloomberg Government.
‘Work to do’ on opioids
Senators looped back repeatedly to the topic of opioids during Dr. Califf’s hearing, reflecting deep concerns about the FDA’s efforts to warn of the risks of prescription painkillers.
There were an estimated 100,306 drug overdose deaths in the United States in the 12 months ending in April, an increase of 28.5% from the 78,056 deaths during the same period the year before, according to the Centers for Disease Control and Prevention.
Dr. Califf said he plans to focus on what information the FDA conveys to the public about the risks of prescription painkillers, including a look at what the labels for these products say.
“I am committed to do a comprehensive review of the status of opioids, early in my tenure,” Dr. Califf said.
Dr. Califf indicated that physicians are still too quick to provide excess doses of these medicines, despite years of efforts to restrain their use. He said he knows relatives who were given 30-day prescriptions for opioids after minor surgery.
“So I know we have work to do,” Dr. Califf said.
Concerns about the FDA’s previous work in managing opioids has led to protests from a few Democratic senators about the prospect of President Biden nominating the acting FDA commissioner, Janet Woodcock, MD, for the permanent post.
At the hearing, Sen. Ben Ray Luján (D-NM) raised the case of the FDA’s approval of the powerful Zohydro painkiller. The agency approved that drug despite an 11-2 vote against it by the FDA’s Anesthetic and Analgesic Drug Products Advisory Committee.
Sen. Luján asked Dr. Califf what he would do if an FDA advisory committee voted “overwhelmingly” against recommending approval of a medicine, as happened in the Zohydro case.
While not mentioned by Sen. Luján in this exchange during the hearing with Dr. Califf, the FDA staff’s rejection of recommendations of advisory committees has been a growing concern among researchers.
The agency last year approved aducanumab (Aduhelm, Biogen), a drug for Alzheimer’s disease, dismissing the advice of its Peripheral and Central Nervous System Drugs Advisory Committee. That decision triggered the resignation of several members of the panel. The FDA staff also earlier rejected the conclusion the majority of members of the same advisory committee offered in 2016 on eteplirsen (Exondys 51, Sarepta), a drug for Duchenne muscular dystrophy.
Dr. Califf told Sen. Luján he had done recent research into how often the FDA staff does not concur with the recommendations of an advisory committee. He said the FDA takes a different course of action in about 25% of cases. In about three-quarters of those cases, the FDA staff opts for a “more stringent” approach regarding allowing the public access to the drug, as opposed to a more generous one as seen in the Zohydro, Aduhelm, and Exondys 51 cases.
Still, Dr. Califf said that when there’s an 11-2 advisory committee vote against recommendation of a product, “the leaders at FDA really need to take a close look” at what’s happening.
Question on accelerated approvals
The FDA’s approval of aducanumab drew attention to a debate already underway about conditional clearances known as accelerated approvals.
The FDA has used this path since the 1990s to speed access to drugs for serious conditions. The trade-off for early access is that the agency sometimes makes the wrong call based on initial findings, and clears a medicine later found not to benefit patients as expected.
The FDA’s cancer division is in the midst of public efforts to address cases where drugmakers have not been able to deliver studies that support accelerated approvals of their oncology drugs. In addition, the Office of Inspector General of the U.S. Department of Health & Human Services announced in August that it is reviewing the FDA’s handling of the accelerated approval process.
At Tuesday’s hearing, Sen. Burr grilled Dr. Califf about how he would respond to calls to change how the FDA handles the accelerated-approval process.
“Can you commit to me and to patients who may rely on cutting-edge treatments that you will not support efforts to narrow this pathway or raise the bar for drugs to be approved under those pathways?” Burr asked Califf.
Dr. Califf responded by saying he was “a fan of accelerated approval – for the right conditions.”
Earlier, in his opening statement, Dr. Califf had said his mother benefited directly from the accelerated approval of new drugs for multiple myeloma. Dr. Califf told Sen. Burr that he had spent “countless hours with patient groups” and understands the need to speed the approval of medicines for serious diseases.
But the FDA also has to make sure it holds up its end of the bargain struck with accelerated approvals. This involves checking on how these medicines work once they are marketed.
“We’re accepting that there’s more uncertainty,” Dr. Califf said. “That means we’ve got to have a better system to evaluate these products as they’re used on the market. And I think there are ways that we can do that now. Technology is making this possible in ways that it just was not possible before.”
Worries about the medical supply chain
Sen. Susan Collins (R-Maine) asked Dr. Califf about the vulnerability of the U.S. medical system to disruptions of the supply chain. She raised concerns about China’s dominance in antibiotic manufacturing as an example. She asked if Congress could do more to encourage domestic manufacturing of medical supplies, such as by offering tax incentives.
Dr. Califf told Sen. Collins he shared her concern about the U.S. manufacturing of ingredients used in both branded and generic drugs. He said he recently has served on a committee of the National Academy of Medicine that is examining supply chain issues.
This committee will soon release a report with specific recommendations, Dr. Califf said.
“We don’t have enough competitive entities in what’s become sort of a commodity business” of drug manufacturing, Dr. Califf said. “So we need a number of steps to make the system more resilient.”
A version of this article first appeared on Medscape.com.
Robert M. Califf, MD, plans to take a close look at federal policies on opioid prescriptions in his expected second turn as the top U.S. regulator of medical products, as well as keep closer tabs on the performance of drugs cleared with accelerated approvals.
Dr. Califf on Tuesday fielded questions at a Senate hearing about his nomination by President Joe Biden to serve as administrator of the U.S. Food and Drug Administration, a role in which he served in the Obama administration. He also spoke about the need to bolster the nation’s ability to maintain an adequate supply of key medical products, including drugs.
Members of the Senate Health, Education, Labor and Pensions Committee, which is handling Dr. Califf’s nomination, were largely cordial and supportive during the hearing. Sen. Patty Murray (D-Wash.), the committee chair, and the panel’s top Republican, Sen. Richard Burr of North Carolina, addressed Dr. Califf during the hearing as if he would soon serve again as the FDA’s leader. Both were among the senators who voted 89-4 to confirm Dr. Califf in a February 2016 vote.
Dr. Califf “was previously confirmed to lead FDA in an overwhelming bipartisan vote, and I look forward to working with him again to ensure FDA continues to protect families across the country, uphold the gold standard of safety and effectiveness, and put science and data first,” Sen. Murray said.
Less enthusiastic about Dr. Califf was Sen. Bernie Sanders (I-VT), who was among the seven senators who did not vote on Dr. Califf’s nomination in 2016.
Sen. Sanders objected in 2016 to Dr. Califf’s ties to the pharmaceutical industry, and he did so again Tuesday. A noted leader in conducting clinical trials, Dr. Califf has worked with many drugmakers. But at the hearing, Dr. Califf said he concurs with Sen. Sanders on an idea strongly opposed by the pharmaceutical industry.
In response to Sen. Sanders’ question, Dr. Califf said he already is “on record as being in favor of Medicare negotiating with the industry on prices.”
The FDA would not take direct part in negotiations, as this work would be handled by the Centers for Medicare & Medicaid Services. Democrats want to give Medicare some negotiating authority through their sweeping Build Back Better Act.
People in the United States are dismayed over both the cost of prescription drugs and the widespread distribution of prescription painkillers that helped fuel the current opioid epidemic, Sen. Sanders told Dr. Califf. Many people will be concerned about an FDA commissioner who has benefited from close ties to the industry, Sen. Sanders said.
“How are they going to believe that you’re going to be an independent and strong voice against this enormously powerful, special interest?” Sen. Sanders asked.
“I’m totally with you on the concept that the price of pharmaceuticals is way too high in this country,” Dr. Califf said in reply.
Dr. Califf was paid $2.7 million in salary and bonus by Verily Life Sciences, the biomedical research organization operated by Alphabet, parent company of Google, according to his federal financial disclosure. He also reported holding board positions with pharmaceutical companies AmyriAD and Centessa Pharmaceuticals.
Bloomberg Government reported that Dr. Califf has ties to about 16 other research organizations and biotech companies. Bloomberg Government also said that, in his earlier FDA service, Dr. Califf kept a whiteboard in his office that listed all the activities and projects that required his recusal, citing as a source Howard Sklamberg, who was a deputy commissioner under Dr. Califf.
“He was very, very, very careful,” Mr. Sklamberg, who’s now an attorney at Arnold & Porter LLP, told Bloomberg Government.
‘Work to do’ on opioids
Senators looped back repeatedly to the topic of opioids during Dr. Califf’s hearing, reflecting deep concerns about the FDA’s efforts to warn of the risks of prescription painkillers.
There were an estimated 100,306 drug overdose deaths in the United States in the 12 months ending in April, an increase of 28.5% from the 78,056 deaths during the same period the year before, according to the Centers for Disease Control and Prevention.
Dr. Califf said he plans to focus on what information the FDA conveys to the public about the risks of prescription painkillers, including a look at what the labels for these products say.
“I am committed to do a comprehensive review of the status of opioids, early in my tenure,” Dr. Califf said.
Dr. Califf indicated that physicians are still too quick to provide excess doses of these medicines, despite years of efforts to restrain their use. He said he knows relatives who were given 30-day prescriptions for opioids after minor surgery.
“So I know we have work to do,” Dr. Califf said.
Concerns about the FDA’s previous work in managing opioids has led to protests from a few Democratic senators about the prospect of President Biden nominating the acting FDA commissioner, Janet Woodcock, MD, for the permanent post.
At the hearing, Sen. Ben Ray Luján (D-NM) raised the case of the FDA’s approval of the powerful Zohydro painkiller. The agency approved that drug despite an 11-2 vote against it by the FDA’s Anesthetic and Analgesic Drug Products Advisory Committee.
Sen. Luján asked Dr. Califf what he would do if an FDA advisory committee voted “overwhelmingly” against recommending approval of a medicine, as happened in the Zohydro case.
While not mentioned by Sen. Luján in this exchange during the hearing with Dr. Califf, the FDA staff’s rejection of recommendations of advisory committees has been a growing concern among researchers.
The agency last year approved aducanumab (Aduhelm, Biogen), a drug for Alzheimer’s disease, dismissing the advice of its Peripheral and Central Nervous System Drugs Advisory Committee. That decision triggered the resignation of several members of the panel. The FDA staff also earlier rejected the conclusion the majority of members of the same advisory committee offered in 2016 on eteplirsen (Exondys 51, Sarepta), a drug for Duchenne muscular dystrophy.
Dr. Califf told Sen. Luján he had done recent research into how often the FDA staff does not concur with the recommendations of an advisory committee. He said the FDA takes a different course of action in about 25% of cases. In about three-quarters of those cases, the FDA staff opts for a “more stringent” approach regarding allowing the public access to the drug, as opposed to a more generous one as seen in the Zohydro, Aduhelm, and Exondys 51 cases.
Still, Dr. Califf said that when there’s an 11-2 advisory committee vote against recommendation of a product, “the leaders at FDA really need to take a close look” at what’s happening.
Question on accelerated approvals
The FDA’s approval of aducanumab drew attention to a debate already underway about conditional clearances known as accelerated approvals.
The FDA has used this path since the 1990s to speed access to drugs for serious conditions. The trade-off for early access is that the agency sometimes makes the wrong call based on initial findings, and clears a medicine later found not to benefit patients as expected.
The FDA’s cancer division is in the midst of public efforts to address cases where drugmakers have not been able to deliver studies that support accelerated approvals of their oncology drugs. In addition, the Office of Inspector General of the U.S. Department of Health & Human Services announced in August that it is reviewing the FDA’s handling of the accelerated approval process.
At Tuesday’s hearing, Sen. Burr grilled Dr. Califf about how he would respond to calls to change how the FDA handles the accelerated-approval process.
“Can you commit to me and to patients who may rely on cutting-edge treatments that you will not support efforts to narrow this pathway or raise the bar for drugs to be approved under those pathways?” Burr asked Califf.
Dr. Califf responded by saying he was “a fan of accelerated approval – for the right conditions.”
Earlier, in his opening statement, Dr. Califf had said his mother benefited directly from the accelerated approval of new drugs for multiple myeloma. Dr. Califf told Sen. Burr that he had spent “countless hours with patient groups” and understands the need to speed the approval of medicines for serious diseases.
But the FDA also has to make sure it holds up its end of the bargain struck with accelerated approvals. This involves checking on how these medicines work once they are marketed.
“We’re accepting that there’s more uncertainty,” Dr. Califf said. “That means we’ve got to have a better system to evaluate these products as they’re used on the market. And I think there are ways that we can do that now. Technology is making this possible in ways that it just was not possible before.”
Worries about the medical supply chain
Sen. Susan Collins (R-Maine) asked Dr. Califf about the vulnerability of the U.S. medical system to disruptions of the supply chain. She raised concerns about China’s dominance in antibiotic manufacturing as an example. She asked if Congress could do more to encourage domestic manufacturing of medical supplies, such as by offering tax incentives.
Dr. Califf told Sen. Collins he shared her concern about the U.S. manufacturing of ingredients used in both branded and generic drugs. He said he recently has served on a committee of the National Academy of Medicine that is examining supply chain issues.
This committee will soon release a report with specific recommendations, Dr. Califf said.
“We don’t have enough competitive entities in what’s become sort of a commodity business” of drug manufacturing, Dr. Califf said. “So we need a number of steps to make the system more resilient.”
A version of this article first appeared on Medscape.com.
Robert M. Califf, MD, plans to take a close look at federal policies on opioid prescriptions in his expected second turn as the top U.S. regulator of medical products, as well as keep closer tabs on the performance of drugs cleared with accelerated approvals.
Dr. Califf on Tuesday fielded questions at a Senate hearing about his nomination by President Joe Biden to serve as administrator of the U.S. Food and Drug Administration, a role in which he served in the Obama administration. He also spoke about the need to bolster the nation’s ability to maintain an adequate supply of key medical products, including drugs.
Members of the Senate Health, Education, Labor and Pensions Committee, which is handling Dr. Califf’s nomination, were largely cordial and supportive during the hearing. Sen. Patty Murray (D-Wash.), the committee chair, and the panel’s top Republican, Sen. Richard Burr of North Carolina, addressed Dr. Califf during the hearing as if he would soon serve again as the FDA’s leader. Both were among the senators who voted 89-4 to confirm Dr. Califf in a February 2016 vote.
Dr. Califf “was previously confirmed to lead FDA in an overwhelming bipartisan vote, and I look forward to working with him again to ensure FDA continues to protect families across the country, uphold the gold standard of safety and effectiveness, and put science and data first,” Sen. Murray said.
Less enthusiastic about Dr. Califf was Sen. Bernie Sanders (I-VT), who was among the seven senators who did not vote on Dr. Califf’s nomination in 2016.
Sen. Sanders objected in 2016 to Dr. Califf’s ties to the pharmaceutical industry, and he did so again Tuesday. A noted leader in conducting clinical trials, Dr. Califf has worked with many drugmakers. But at the hearing, Dr. Califf said he concurs with Sen. Sanders on an idea strongly opposed by the pharmaceutical industry.
In response to Sen. Sanders’ question, Dr. Califf said he already is “on record as being in favor of Medicare negotiating with the industry on prices.”
The FDA would not take direct part in negotiations, as this work would be handled by the Centers for Medicare & Medicaid Services. Democrats want to give Medicare some negotiating authority through their sweeping Build Back Better Act.
People in the United States are dismayed over both the cost of prescription drugs and the widespread distribution of prescription painkillers that helped fuel the current opioid epidemic, Sen. Sanders told Dr. Califf. Many people will be concerned about an FDA commissioner who has benefited from close ties to the industry, Sen. Sanders said.
“How are they going to believe that you’re going to be an independent and strong voice against this enormously powerful, special interest?” Sen. Sanders asked.
“I’m totally with you on the concept that the price of pharmaceuticals is way too high in this country,” Dr. Califf said in reply.
Dr. Califf was paid $2.7 million in salary and bonus by Verily Life Sciences, the biomedical research organization operated by Alphabet, parent company of Google, according to his federal financial disclosure. He also reported holding board positions with pharmaceutical companies AmyriAD and Centessa Pharmaceuticals.
Bloomberg Government reported that Dr. Califf has ties to about 16 other research organizations and biotech companies. Bloomberg Government also said that, in his earlier FDA service, Dr. Califf kept a whiteboard in his office that listed all the activities and projects that required his recusal, citing as a source Howard Sklamberg, who was a deputy commissioner under Dr. Califf.
“He was very, very, very careful,” Mr. Sklamberg, who’s now an attorney at Arnold & Porter LLP, told Bloomberg Government.
‘Work to do’ on opioids
Senators looped back repeatedly to the topic of opioids during Dr. Califf’s hearing, reflecting deep concerns about the FDA’s efforts to warn of the risks of prescription painkillers.
There were an estimated 100,306 drug overdose deaths in the United States in the 12 months ending in April, an increase of 28.5% from the 78,056 deaths during the same period the year before, according to the Centers for Disease Control and Prevention.
Dr. Califf said he plans to focus on what information the FDA conveys to the public about the risks of prescription painkillers, including a look at what the labels for these products say.
“I am committed to do a comprehensive review of the status of opioids, early in my tenure,” Dr. Califf said.
Dr. Califf indicated that physicians are still too quick to provide excess doses of these medicines, despite years of efforts to restrain their use. He said he knows relatives who were given 30-day prescriptions for opioids after minor surgery.
“So I know we have work to do,” Dr. Califf said.
Concerns about the FDA’s previous work in managing opioids has led to protests from a few Democratic senators about the prospect of President Biden nominating the acting FDA commissioner, Janet Woodcock, MD, for the permanent post.
At the hearing, Sen. Ben Ray Luján (D-NM) raised the case of the FDA’s approval of the powerful Zohydro painkiller. The agency approved that drug despite an 11-2 vote against it by the FDA’s Anesthetic and Analgesic Drug Products Advisory Committee.
Sen. Luján asked Dr. Califf what he would do if an FDA advisory committee voted “overwhelmingly” against recommending approval of a medicine, as happened in the Zohydro case.
While not mentioned by Sen. Luján in this exchange during the hearing with Dr. Califf, the FDA staff’s rejection of recommendations of advisory committees has been a growing concern among researchers.
The agency last year approved aducanumab (Aduhelm, Biogen), a drug for Alzheimer’s disease, dismissing the advice of its Peripheral and Central Nervous System Drugs Advisory Committee. That decision triggered the resignation of several members of the panel. The FDA staff also earlier rejected the conclusion the majority of members of the same advisory committee offered in 2016 on eteplirsen (Exondys 51, Sarepta), a drug for Duchenne muscular dystrophy.
Dr. Califf told Sen. Luján he had done recent research into how often the FDA staff does not concur with the recommendations of an advisory committee. He said the FDA takes a different course of action in about 25% of cases. In about three-quarters of those cases, the FDA staff opts for a “more stringent” approach regarding allowing the public access to the drug, as opposed to a more generous one as seen in the Zohydro, Aduhelm, and Exondys 51 cases.
Still, Dr. Califf said that when there’s an 11-2 advisory committee vote against recommendation of a product, “the leaders at FDA really need to take a close look” at what’s happening.
Question on accelerated approvals
The FDA’s approval of aducanumab drew attention to a debate already underway about conditional clearances known as accelerated approvals.
The FDA has used this path since the 1990s to speed access to drugs for serious conditions. The trade-off for early access is that the agency sometimes makes the wrong call based on initial findings, and clears a medicine later found not to benefit patients as expected.
The FDA’s cancer division is in the midst of public efforts to address cases where drugmakers have not been able to deliver studies that support accelerated approvals of their oncology drugs. In addition, the Office of Inspector General of the U.S. Department of Health & Human Services announced in August that it is reviewing the FDA’s handling of the accelerated approval process.
At Tuesday’s hearing, Sen. Burr grilled Dr. Califf about how he would respond to calls to change how the FDA handles the accelerated-approval process.
“Can you commit to me and to patients who may rely on cutting-edge treatments that you will not support efforts to narrow this pathway or raise the bar for drugs to be approved under those pathways?” Burr asked Califf.
Dr. Califf responded by saying he was “a fan of accelerated approval – for the right conditions.”
Earlier, in his opening statement, Dr. Califf had said his mother benefited directly from the accelerated approval of new drugs for multiple myeloma. Dr. Califf told Sen. Burr that he had spent “countless hours with patient groups” and understands the need to speed the approval of medicines for serious diseases.
But the FDA also has to make sure it holds up its end of the bargain struck with accelerated approvals. This involves checking on how these medicines work once they are marketed.
“We’re accepting that there’s more uncertainty,” Dr. Califf said. “That means we’ve got to have a better system to evaluate these products as they’re used on the market. And I think there are ways that we can do that now. Technology is making this possible in ways that it just was not possible before.”
Worries about the medical supply chain
Sen. Susan Collins (R-Maine) asked Dr. Califf about the vulnerability of the U.S. medical system to disruptions of the supply chain. She raised concerns about China’s dominance in antibiotic manufacturing as an example. She asked if Congress could do more to encourage domestic manufacturing of medical supplies, such as by offering tax incentives.
Dr. Califf told Sen. Collins he shared her concern about the U.S. manufacturing of ingredients used in both branded and generic drugs. He said he recently has served on a committee of the National Academy of Medicine that is examining supply chain issues.
This committee will soon release a report with specific recommendations, Dr. Califf said.
“We don’t have enough competitive entities in what’s become sort of a commodity business” of drug manufacturing, Dr. Califf said. “So we need a number of steps to make the system more resilient.”
A version of this article first appeared on Medscape.com.
Average-risk women with dense breasts—What breast screening is appropriate?
Text copyright DenseBreast-info.org.
Answer
A. For women with extremely dense breasts who are not otherwise at increased risk for breast cancer, screening magnetic resonance imaging (MRI) is preferred, plus her mammogram or tomosynthesis. If MRI is not an option, consider ultrasonography or contrast-enhanced mammography.
The same screening considerations apply to women with heterogeneously dense breasts; however, there is limited capacity for MRI or even ultrasound screening at many facilities. Research supports MRI in dense breasts, and abbreviated, lower-cost protocols have been validated that address some of the barriers to MRI.1 Although not yet widely available, abbreviated MRI will likely have a greater role in screening women with dense breasts who are not high risk. It is important to note that preauthorization from insurance may be required for screening MRI, and in most US states, deductibles and copays apply.
The exam
Contrast-enhanced MRI requires IV injection of gadolinium-based contrast to look at the anatomy and blood flow patterns of the breast tissue. The patient lies face down with the breasts placed in two rectangular openings, or “coils.” The exam takes place inside the tunnel of the scanner, with the head facing out.After initial images are obtained, the contrast agent is injected into a vein in the arm, and additional images are taken, which will show areas of enhancement. The exam takes about 20 to 40 minutes. An “abbreviated” MRI can be performed for screening in some centers, which uses fewer sequences and takes about 10 minutes.
Benefits
At least 40% of cancers are missed on mammography in women with dense breasts.2 MRI is the most widely studied technique using a contrast agent, and it produces the highest additional cancer detection of all the supplemental technologies to date, yielding, in the first year, 10-16 additional cancers per 1,000 women screened after mammography/tomosynthesis (reviewed in Berg et al.3). The cancer-detection benefit is seen across all breast density categories, even among average-risk women.4 There is no ionizing radiation, and it has been shown to reduce the rate of interval cancers (those detected due to symptoms after a negative screening mammogram), as well as the rate of late-stage disease. Axillary lymph nodes can be examined at the same screening exam.
While tomosynthesis improves cancer detection in women with fatty breasts, scattered fibroglandular breast tissue, and heterogeneously dense breasts, it does not significantly improve cancer detection in women with extremely dense breasts.5,6 Current American Cancer Society and National Comprehensive Cancer Network guidelines recommend annual screening MRI for women at high risk for breast cancer (regardless of breast density); however, increasingly, research supports the effectiveness of MRI in women with dense breasts who are otherwise considered average risk. A large randomized controlled trial in the Netherlands compared outcomes in women with extremely dense breasts invited to have screening MRI after negative mammography to those assigned to continue receiving screening mammography only. The incremental cancer detection rate was 16.5 per 1,000 (79/4,783) women screened with MRI in the first round7 and 6 per 1,000 women screened in the second round 2 years later.8 The interval cancer rate was 0.8 per 1,000 (4/4,783) women screened with MRI, compared with 4.9 per 1,000 (16/3,278) women who declined MRI and received mammography only.7
Screening ultrasound will show up to 3 additional cancers per 1,000 women screened after mammography/tomosynthesis (reviewed in Vourtsis and Berg9 and Berg and Vourtsis10), far lower than the added cancer-detection rate of MRI. Consider screening ultrasound for women who cannot tolerate or access screening MRI.11 Contrast-enhanced mammography (CEM) uses iodinated contrast (as in computed tomography). CEM is not widely available but appears to show cancer-detection similar to MRI. For further discussion, see Berg et al’s 2021 review.3
The FIGURE shows an example of an invasive cancer depicted on contrast-enhanced MRI in a 53-year-old woman with dense breasts and a family history of breast cancer that was not visible on tomosynthesis, even in retrospect, due to masking by dense tissue.

Considerations
Breast MRI increases callbacks even after mammography and ultrasound; however, such false alarms are reduced in subsequent screening rounds. MRI cannot be performed in women who have certain metal implants— some pacemakers or spinal fixation rods—and is not recommended for pregnant women. Claustrophobia may be an issue for some women. MRI is expensive and requires IV contrast. Gadolinium is known to accumulate in the brain, although the long-term effects of this are unknown and no harm has been shown.●
For more information, visit medically sourced DenseBreast-info.org. Comprehensive resources include a free CME opportunity, Dense Breasts and Supplemental Screening.
- Comstock CE, Gatsonis C, Newstead GM, et al. Comparison of abbreviated breast MRI vs digital breast tomosynthesis for breast cancer detection among women with dense breasts undergoing screening. JAMA. 2020;323:746-756. doi: 10.1001 /jama.2020.0572
- Kerlikowske K, Zhu W, Tosteson AN, et al. Identifying women with dense breasts at high risk for interval cancer: a cohort study. Ann Intern Med. 2015;162:673-681. doi: 10.7326/M14-1465.
- Berg WA, Rafferty EA, Friedewald SM, Hruska CB, Rahbar H. Screening Algorithms in Dense Breasts: AJR Expert Panel Narrative Review. AJR Am J Roentgenol. 2021;216:275-294. doi: 10.2214/AJR.20.24436.
- Kuhl CK, Strobel K, Bieling H, et al. Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology. 2017;283:361-370. doi: 10.1148/radiol.2016161444.
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315:1784-1786. doi: 10.1001/jama.2016.1708.
- Osteras BH, Martinsen ACT, Gullien R, et al. Digital mammography versus breast tomosynthesis: impact of breast density on diagnostic performance in population-based screening. Radiology. 2019;293:60-68. doi: 10.1148 /radiol.2019190425.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Veenhuizen SGA, de Lange SV, Bakker MF, et al. Supplemental breast MRI for women with extremely dense breasts: results of the second screening round of the DENSE trial. Radiology. 2021;299:278-286. doi: 10.1148/radiol.2021203633.
- Vourtsis A, Berg WA. Breast density implications and supplemental screening. Eur Radiol. 2019;29:1762-1777. doi: 10.1007/s00330-018-5668-8.
- Berg WA, Vourtsis A. Screening ultrasound using handheld or automated technique in women with dense breasts. J Breast Imaging. 2019;1:283-296.
- National Comprehensive Cancer Network. Breast Cancer Screening and Diagnosis (Version 1.2021). https://www.nccn. org/professionals/physician_gls/pdf/breast-screening.pdf. Accessed November 18, 2021.
Text copyright DenseBreast-info.org.
Answer
A. For women with extremely dense breasts who are not otherwise at increased risk for breast cancer, screening magnetic resonance imaging (MRI) is preferred, plus her mammogram or tomosynthesis. If MRI is not an option, consider ultrasonography or contrast-enhanced mammography.
The same screening considerations apply to women with heterogeneously dense breasts; however, there is limited capacity for MRI or even ultrasound screening at many facilities. Research supports MRI in dense breasts, and abbreviated, lower-cost protocols have been validated that address some of the barriers to MRI.1 Although not yet widely available, abbreviated MRI will likely have a greater role in screening women with dense breasts who are not high risk. It is important to note that preauthorization from insurance may be required for screening MRI, and in most US states, deductibles and copays apply.
The exam
Contrast-enhanced MRI requires IV injection of gadolinium-based contrast to look at the anatomy and blood flow patterns of the breast tissue. The patient lies face down with the breasts placed in two rectangular openings, or “coils.” The exam takes place inside the tunnel of the scanner, with the head facing out.After initial images are obtained, the contrast agent is injected into a vein in the arm, and additional images are taken, which will show areas of enhancement. The exam takes about 20 to 40 minutes. An “abbreviated” MRI can be performed for screening in some centers, which uses fewer sequences and takes about 10 minutes.
Benefits
At least 40% of cancers are missed on mammography in women with dense breasts.2 MRI is the most widely studied technique using a contrast agent, and it produces the highest additional cancer detection of all the supplemental technologies to date, yielding, in the first year, 10-16 additional cancers per 1,000 women screened after mammography/tomosynthesis (reviewed in Berg et al.3). The cancer-detection benefit is seen across all breast density categories, even among average-risk women.4 There is no ionizing radiation, and it has been shown to reduce the rate of interval cancers (those detected due to symptoms after a negative screening mammogram), as well as the rate of late-stage disease. Axillary lymph nodes can be examined at the same screening exam.
While tomosynthesis improves cancer detection in women with fatty breasts, scattered fibroglandular breast tissue, and heterogeneously dense breasts, it does not significantly improve cancer detection in women with extremely dense breasts.5,6 Current American Cancer Society and National Comprehensive Cancer Network guidelines recommend annual screening MRI for women at high risk for breast cancer (regardless of breast density); however, increasingly, research supports the effectiveness of MRI in women with dense breasts who are otherwise considered average risk. A large randomized controlled trial in the Netherlands compared outcomes in women with extremely dense breasts invited to have screening MRI after negative mammography to those assigned to continue receiving screening mammography only. The incremental cancer detection rate was 16.5 per 1,000 (79/4,783) women screened with MRI in the first round7 and 6 per 1,000 women screened in the second round 2 years later.8 The interval cancer rate was 0.8 per 1,000 (4/4,783) women screened with MRI, compared with 4.9 per 1,000 (16/3,278) women who declined MRI and received mammography only.7
Screening ultrasound will show up to 3 additional cancers per 1,000 women screened after mammography/tomosynthesis (reviewed in Vourtsis and Berg9 and Berg and Vourtsis10), far lower than the added cancer-detection rate of MRI. Consider screening ultrasound for women who cannot tolerate or access screening MRI.11 Contrast-enhanced mammography (CEM) uses iodinated contrast (as in computed tomography). CEM is not widely available but appears to show cancer-detection similar to MRI. For further discussion, see Berg et al’s 2021 review.3
The FIGURE shows an example of an invasive cancer depicted on contrast-enhanced MRI in a 53-year-old woman with dense breasts and a family history of breast cancer that was not visible on tomosynthesis, even in retrospect, due to masking by dense tissue.

Considerations
Breast MRI increases callbacks even after mammography and ultrasound; however, such false alarms are reduced in subsequent screening rounds. MRI cannot be performed in women who have certain metal implants— some pacemakers or spinal fixation rods—and is not recommended for pregnant women. Claustrophobia may be an issue for some women. MRI is expensive and requires IV contrast. Gadolinium is known to accumulate in the brain, although the long-term effects of this are unknown and no harm has been shown.●
For more information, visit medically sourced DenseBreast-info.org. Comprehensive resources include a free CME opportunity, Dense Breasts and Supplemental Screening.
Text copyright DenseBreast-info.org.
Answer
A. For women with extremely dense breasts who are not otherwise at increased risk for breast cancer, screening magnetic resonance imaging (MRI) is preferred, plus her mammogram or tomosynthesis. If MRI is not an option, consider ultrasonography or contrast-enhanced mammography.
The same screening considerations apply to women with heterogeneously dense breasts; however, there is limited capacity for MRI or even ultrasound screening at many facilities. Research supports MRI in dense breasts, and abbreviated, lower-cost protocols have been validated that address some of the barriers to MRI.1 Although not yet widely available, abbreviated MRI will likely have a greater role in screening women with dense breasts who are not high risk. It is important to note that preauthorization from insurance may be required for screening MRI, and in most US states, deductibles and copays apply.
The exam
Contrast-enhanced MRI requires IV injection of gadolinium-based contrast to look at the anatomy and blood flow patterns of the breast tissue. The patient lies face down with the breasts placed in two rectangular openings, or “coils.” The exam takes place inside the tunnel of the scanner, with the head facing out.After initial images are obtained, the contrast agent is injected into a vein in the arm, and additional images are taken, which will show areas of enhancement. The exam takes about 20 to 40 minutes. An “abbreviated” MRI can be performed for screening in some centers, which uses fewer sequences and takes about 10 minutes.
Benefits
At least 40% of cancers are missed on mammography in women with dense breasts.2 MRI is the most widely studied technique using a contrast agent, and it produces the highest additional cancer detection of all the supplemental technologies to date, yielding, in the first year, 10-16 additional cancers per 1,000 women screened after mammography/tomosynthesis (reviewed in Berg et al.3). The cancer-detection benefit is seen across all breast density categories, even among average-risk women.4 There is no ionizing radiation, and it has been shown to reduce the rate of interval cancers (those detected due to symptoms after a negative screening mammogram), as well as the rate of late-stage disease. Axillary lymph nodes can be examined at the same screening exam.
While tomosynthesis improves cancer detection in women with fatty breasts, scattered fibroglandular breast tissue, and heterogeneously dense breasts, it does not significantly improve cancer detection in women with extremely dense breasts.5,6 Current American Cancer Society and National Comprehensive Cancer Network guidelines recommend annual screening MRI for women at high risk for breast cancer (regardless of breast density); however, increasingly, research supports the effectiveness of MRI in women with dense breasts who are otherwise considered average risk. A large randomized controlled trial in the Netherlands compared outcomes in women with extremely dense breasts invited to have screening MRI after negative mammography to those assigned to continue receiving screening mammography only. The incremental cancer detection rate was 16.5 per 1,000 (79/4,783) women screened with MRI in the first round7 and 6 per 1,000 women screened in the second round 2 years later.8 The interval cancer rate was 0.8 per 1,000 (4/4,783) women screened with MRI, compared with 4.9 per 1,000 (16/3,278) women who declined MRI and received mammography only.7
Screening ultrasound will show up to 3 additional cancers per 1,000 women screened after mammography/tomosynthesis (reviewed in Vourtsis and Berg9 and Berg and Vourtsis10), far lower than the added cancer-detection rate of MRI. Consider screening ultrasound for women who cannot tolerate or access screening MRI.11 Contrast-enhanced mammography (CEM) uses iodinated contrast (as in computed tomography). CEM is not widely available but appears to show cancer-detection similar to MRI. For further discussion, see Berg et al’s 2021 review.3
The FIGURE shows an example of an invasive cancer depicted on contrast-enhanced MRI in a 53-year-old woman with dense breasts and a family history of breast cancer that was not visible on tomosynthesis, even in retrospect, due to masking by dense tissue.

Considerations
Breast MRI increases callbacks even after mammography and ultrasound; however, such false alarms are reduced in subsequent screening rounds. MRI cannot be performed in women who have certain metal implants— some pacemakers or spinal fixation rods—and is not recommended for pregnant women. Claustrophobia may be an issue for some women. MRI is expensive and requires IV contrast. Gadolinium is known to accumulate in the brain, although the long-term effects of this are unknown and no harm has been shown.●
For more information, visit medically sourced DenseBreast-info.org. Comprehensive resources include a free CME opportunity, Dense Breasts and Supplemental Screening.
- Comstock CE, Gatsonis C, Newstead GM, et al. Comparison of abbreviated breast MRI vs digital breast tomosynthesis for breast cancer detection among women with dense breasts undergoing screening. JAMA. 2020;323:746-756. doi: 10.1001 /jama.2020.0572
- Kerlikowske K, Zhu W, Tosteson AN, et al. Identifying women with dense breasts at high risk for interval cancer: a cohort study. Ann Intern Med. 2015;162:673-681. doi: 10.7326/M14-1465.
- Berg WA, Rafferty EA, Friedewald SM, Hruska CB, Rahbar H. Screening Algorithms in Dense Breasts: AJR Expert Panel Narrative Review. AJR Am J Roentgenol. 2021;216:275-294. doi: 10.2214/AJR.20.24436.
- Kuhl CK, Strobel K, Bieling H, et al. Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology. 2017;283:361-370. doi: 10.1148/radiol.2016161444.
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315:1784-1786. doi: 10.1001/jama.2016.1708.
- Osteras BH, Martinsen ACT, Gullien R, et al. Digital mammography versus breast tomosynthesis: impact of breast density on diagnostic performance in population-based screening. Radiology. 2019;293:60-68. doi: 10.1148 /radiol.2019190425.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Veenhuizen SGA, de Lange SV, Bakker MF, et al. Supplemental breast MRI for women with extremely dense breasts: results of the second screening round of the DENSE trial. Radiology. 2021;299:278-286. doi: 10.1148/radiol.2021203633.
- Vourtsis A, Berg WA. Breast density implications and supplemental screening. Eur Radiol. 2019;29:1762-1777. doi: 10.1007/s00330-018-5668-8.
- Berg WA, Vourtsis A. Screening ultrasound using handheld or automated technique in women with dense breasts. J Breast Imaging. 2019;1:283-296.
- National Comprehensive Cancer Network. Breast Cancer Screening and Diagnosis (Version 1.2021). https://www.nccn. org/professionals/physician_gls/pdf/breast-screening.pdf. Accessed November 18, 2021.
- Comstock CE, Gatsonis C, Newstead GM, et al. Comparison of abbreviated breast MRI vs digital breast tomosynthesis for breast cancer detection among women with dense breasts undergoing screening. JAMA. 2020;323:746-756. doi: 10.1001 /jama.2020.0572
- Kerlikowske K, Zhu W, Tosteson AN, et al. Identifying women with dense breasts at high risk for interval cancer: a cohort study. Ann Intern Med. 2015;162:673-681. doi: 10.7326/M14-1465.
- Berg WA, Rafferty EA, Friedewald SM, Hruska CB, Rahbar H. Screening Algorithms in Dense Breasts: AJR Expert Panel Narrative Review. AJR Am J Roentgenol. 2021;216:275-294. doi: 10.2214/AJR.20.24436.
- Kuhl CK, Strobel K, Bieling H, et al. Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology. 2017;283:361-370. doi: 10.1148/radiol.2016161444.
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315:1784-1786. doi: 10.1001/jama.2016.1708.
- Osteras BH, Martinsen ACT, Gullien R, et al. Digital mammography versus breast tomosynthesis: impact of breast density on diagnostic performance in population-based screening. Radiology. 2019;293:60-68. doi: 10.1148 /radiol.2019190425.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Veenhuizen SGA, de Lange SV, Bakker MF, et al. Supplemental breast MRI for women with extremely dense breasts: results of the second screening round of the DENSE trial. Radiology. 2021;299:278-286. doi: 10.1148/radiol.2021203633.
- Vourtsis A, Berg WA. Breast density implications and supplemental screening. Eur Radiol. 2019;29:1762-1777. doi: 10.1007/s00330-018-5668-8.
- Berg WA, Vourtsis A. Screening ultrasound using handheld or automated technique in women with dense breasts. J Breast Imaging. 2019;1:283-296.
- National Comprehensive Cancer Network. Breast Cancer Screening and Diagnosis (Version 1.2021). https://www.nccn. org/professionals/physician_gls/pdf/breast-screening.pdf. Accessed November 18, 2021.
Quiz developed in collaboration with