User login
Risk of stent infection low, but may be underreported
Infections of coronary stents appear to be uncommon, but it is not clear if they are often missed, underreported, or truly rare, according to a new analysis.
In a search of multiple databases, 79 cases of coronary stent infections (CSI) were found in 65 published reports, according to Venkatakrishnan Ramakumar, MBBS, MD, department of cardiology, All India Institute of Medical Sciences, New Delhi.
Over the period of evaluation, which had no defined starting point but stretched to November 2021, the 79 infections reported worldwide occurred when millions of percutaneous coronary intervention (PCI) procedures were performed. In the United States alone, the current estimated annual number of PCIs is 600,000, according to an article published in the Journal of the American Heart Association.
If the number of reported CSI cases represented even a modest fraction of those that occurred, the risk would still be almost negligible. Yet, Dr. Ramakumar insisted that there has been little attention paid to the potential for CSI, creating a situation in which many or almost all cases are simply being missed.
“We do not know how many infections have gone unrecognized,” Dr. Ramakumar said in presenting his results at the Cardiovascular Research Technologies conference, sponsored by MedStar Heart & Vascular Institute. And even if they are identified and promptly treated, there “is the potential for a publication bias,” he added, referring to the reluctance of investigators to submit and publishers to accept manuscripts with negative results.
Regardless of the frequency with which they occur, CSI is associated with bad outcomes, according to the data evaluated by Dr. Ramakumar. On the basis of in-hospital mortality, the primary endpoint of this analysis, the rate of death in patients developing CSI was 30.3%.
Successful treatment varied by hospital type
This risk was not uniform. Rather, rates of in-hospital mortality and proportion of patients treated successfully varied substantially by type of hospital. At private teaching hospitals for example, successful treatment – whether medical alone or followed by bailout surgery – was 80%. The rates fell to 40% at public teaching hospitals and then to 25% at private nonteaching hospitals.
The full-text articles included in this analysis were evaluated and selected by two reviewers working independently. A CSI diagnosis made clinically or with imaging and treatment outcomes were among criteria for the case studies to be included. Dr. Ramakumar said the study, which he claimed is the largest systematic review of CSI ever conducted, has been registered with PROSPERO, an international prospective registry of systematic reviews.
The presenting symptom was fever in 72% of cases and chest pain in the others, although there was one asymptomatic CSI reported. On angiography, 62% had a concomitant mycotic aneurysm. Intramyocardial abscess (13.9%), rupture (11.3%), and coronary fistula (7.5%) were also common findings, but no angiographic abnormalities could be identified in 53% of patients.
Following PCI, most CSI developed within 8 days (43%) or the first month (23%), but CSI was reported more than 6 months after the procedure in 19%. Complex PCI accounted for 51% of cases. Of stent types, 56% were drug eluting and 13% were bare metal.
When comparing characteristics of those who survived CSI with those who did not, most (89%) of those with a non–ST-segment elevated acute coronary syndrome ultimately survived, while survival from CSI in those with structural heart disease was only 17%.
Microbiological findings were not a criterion for study inclusion, but Staphylococcus species accounted for 65% of the infections for which positive cultures were reported. Pseudomonas accounted for 13%. Less than 4% (3.8%) tested positive for multiple pathogens. A small proportion of patients had unusual infectious organisms.
As part of this analysis, the investigators developed an artificial intelligence model to predict CSI based on patient characteristics and other variables. However, the specificity of only around 70% led Dr. Ramakumar to conclude that it does not yet have practical value.
However, he believes that better methodology to detect CSI is needed, and he proposed a diagnostic algorithm that he believes would both improve detection rates and accelerate the time to diagnosis.
Algorithm proposed for detection of CSI
In this algorithm, the first step in symptomatic patients with a positive blood culture suspected of CSI is imaging, such as transthoracic echocardiography, to identify features of infective endocarditis or endarteritis. If the imaging is positive, further imaging, such as PET, that supports the diagnosis, should be adequate to support a diagnosis and treatment.
If initial imaging is negative, alternative diagnoses should be considered, but Dr. Ramakumar advised repeat imaging after 48 hours if symptoms persist and no other causes are found.
Dr. Ramakumar acknowledged the many limitations of this analysis, including the small sample size and the challenges of assembling coherent data from case reports with variable types of information submitted during different eras of PCI evolution. However, reiterating that CSI might be frequently missed, he emphasized that this problem might be bigger than currently understood.
It is difficult to rule out any possibility that CSI is frequently missed, but Andrew Sharp, MD, PhD, a consultant interventional cardiologist at the University Hospital of Wales, Cardiff, is skeptical.
“One might think this is a potential problem, but I cannot think of one patient in whom this has occurred,” Dr. Sharp said in an interview. He is fairly confident that they are extremely rare.
“When there is infection associated with a foreign body, such as a pacemaker, they do not typically resolve by themselves,” he explained. “Often the device has to be removed. If this was true for CSI, then I think we would be aware of these complications.”
However, he praised the investigators for taking a look at CSI in a systematic approach. An invited panelist during the CRT featured research, which is where these data were presented, Dr. Sharp was more interested in understanding why they do not occur now that data are available to suggest they are rare.
“Is there something in the coronary environment, such as the consistent blood flow, that protects against infection?” he asked. CSI is a valid area of further research, according to Dr. Sharp, but he does not consider infected stents to be a common threat based on his own sizable case series.
Dr. Ramakumar and Dr. Sharp reported no potential conflicts of interest.
Infections of coronary stents appear to be uncommon, but it is not clear if they are often missed, underreported, or truly rare, according to a new analysis.
In a search of multiple databases, 79 cases of coronary stent infections (CSI) were found in 65 published reports, according to Venkatakrishnan Ramakumar, MBBS, MD, department of cardiology, All India Institute of Medical Sciences, New Delhi.
Over the period of evaluation, which had no defined starting point but stretched to November 2021, the 79 infections reported worldwide occurred when millions of percutaneous coronary intervention (PCI) procedures were performed. In the United States alone, the current estimated annual number of PCIs is 600,000, according to an article published in the Journal of the American Heart Association.
If the number of reported CSI cases represented even a modest fraction of those that occurred, the risk would still be almost negligible. Yet, Dr. Ramakumar insisted that there has been little attention paid to the potential for CSI, creating a situation in which many or almost all cases are simply being missed.
“We do not know how many infections have gone unrecognized,” Dr. Ramakumar said in presenting his results at the Cardiovascular Research Technologies conference, sponsored by MedStar Heart & Vascular Institute. And even if they are identified and promptly treated, there “is the potential for a publication bias,” he added, referring to the reluctance of investigators to submit and publishers to accept manuscripts with negative results.
Regardless of the frequency with which they occur, CSI is associated with bad outcomes, according to the data evaluated by Dr. Ramakumar. On the basis of in-hospital mortality, the primary endpoint of this analysis, the rate of death in patients developing CSI was 30.3%.
Successful treatment varied by hospital type
This risk was not uniform. Rather, rates of in-hospital mortality and proportion of patients treated successfully varied substantially by type of hospital. At private teaching hospitals for example, successful treatment – whether medical alone or followed by bailout surgery – was 80%. The rates fell to 40% at public teaching hospitals and then to 25% at private nonteaching hospitals.
The full-text articles included in this analysis were evaluated and selected by two reviewers working independently. A CSI diagnosis made clinically or with imaging and treatment outcomes were among criteria for the case studies to be included. Dr. Ramakumar said the study, which he claimed is the largest systematic review of CSI ever conducted, has been registered with PROSPERO, an international prospective registry of systematic reviews.
The presenting symptom was fever in 72% of cases and chest pain in the others, although there was one asymptomatic CSI reported. On angiography, 62% had a concomitant mycotic aneurysm. Intramyocardial abscess (13.9%), rupture (11.3%), and coronary fistula (7.5%) were also common findings, but no angiographic abnormalities could be identified in 53% of patients.
Following PCI, most CSI developed within 8 days (43%) or the first month (23%), but CSI was reported more than 6 months after the procedure in 19%. Complex PCI accounted for 51% of cases. Of stent types, 56% were drug eluting and 13% were bare metal.
When comparing characteristics of those who survived CSI with those who did not, most (89%) of those with a non–ST-segment elevated acute coronary syndrome ultimately survived, while survival from CSI in those with structural heart disease was only 17%.
Microbiological findings were not a criterion for study inclusion, but Staphylococcus species accounted for 65% of the infections for which positive cultures were reported. Pseudomonas accounted for 13%. Less than 4% (3.8%) tested positive for multiple pathogens. A small proportion of patients had unusual infectious organisms.
As part of this analysis, the investigators developed an artificial intelligence model to predict CSI based on patient characteristics and other variables. However, the specificity of only around 70% led Dr. Ramakumar to conclude that it does not yet have practical value.
However, he believes that better methodology to detect CSI is needed, and he proposed a diagnostic algorithm that he believes would both improve detection rates and accelerate the time to diagnosis.
Algorithm proposed for detection of CSI
In this algorithm, the first step in symptomatic patients with a positive blood culture suspected of CSI is imaging, such as transthoracic echocardiography, to identify features of infective endocarditis or endarteritis. If the imaging is positive, further imaging, such as PET, that supports the diagnosis, should be adequate to support a diagnosis and treatment.
If initial imaging is negative, alternative diagnoses should be considered, but Dr. Ramakumar advised repeat imaging after 48 hours if symptoms persist and no other causes are found.
Dr. Ramakumar acknowledged the many limitations of this analysis, including the small sample size and the challenges of assembling coherent data from case reports with variable types of information submitted during different eras of PCI evolution. However, reiterating that CSI might be frequently missed, he emphasized that this problem might be bigger than currently understood.
It is difficult to rule out any possibility that CSI is frequently missed, but Andrew Sharp, MD, PhD, a consultant interventional cardiologist at the University Hospital of Wales, Cardiff, is skeptical.
“One might think this is a potential problem, but I cannot think of one patient in whom this has occurred,” Dr. Sharp said in an interview. He is fairly confident that they are extremely rare.
“When there is infection associated with a foreign body, such as a pacemaker, they do not typically resolve by themselves,” he explained. “Often the device has to be removed. If this was true for CSI, then I think we would be aware of these complications.”
However, he praised the investigators for taking a look at CSI in a systematic approach. An invited panelist during the CRT featured research, which is where these data were presented, Dr. Sharp was more interested in understanding why they do not occur now that data are available to suggest they are rare.
“Is there something in the coronary environment, such as the consistent blood flow, that protects against infection?” he asked. CSI is a valid area of further research, according to Dr. Sharp, but he does not consider infected stents to be a common threat based on his own sizable case series.
Dr. Ramakumar and Dr. Sharp reported no potential conflicts of interest.
Infections of coronary stents appear to be uncommon, but it is not clear if they are often missed, underreported, or truly rare, according to a new analysis.
In a search of multiple databases, 79 cases of coronary stent infections (CSI) were found in 65 published reports, according to Venkatakrishnan Ramakumar, MBBS, MD, department of cardiology, All India Institute of Medical Sciences, New Delhi.
Over the period of evaluation, which had no defined starting point but stretched to November 2021, the 79 infections reported worldwide occurred when millions of percutaneous coronary intervention (PCI) procedures were performed. In the United States alone, the current estimated annual number of PCIs is 600,000, according to an article published in the Journal of the American Heart Association.
If the number of reported CSI cases represented even a modest fraction of those that occurred, the risk would still be almost negligible. Yet, Dr. Ramakumar insisted that there has been little attention paid to the potential for CSI, creating a situation in which many or almost all cases are simply being missed.
“We do not know how many infections have gone unrecognized,” Dr. Ramakumar said in presenting his results at the Cardiovascular Research Technologies conference, sponsored by MedStar Heart & Vascular Institute. And even if they are identified and promptly treated, there “is the potential for a publication bias,” he added, referring to the reluctance of investigators to submit and publishers to accept manuscripts with negative results.
Regardless of the frequency with which they occur, CSI is associated with bad outcomes, according to the data evaluated by Dr. Ramakumar. On the basis of in-hospital mortality, the primary endpoint of this analysis, the rate of death in patients developing CSI was 30.3%.
Successful treatment varied by hospital type
This risk was not uniform. Rather, rates of in-hospital mortality and proportion of patients treated successfully varied substantially by type of hospital. At private teaching hospitals for example, successful treatment – whether medical alone or followed by bailout surgery – was 80%. The rates fell to 40% at public teaching hospitals and then to 25% at private nonteaching hospitals.
The full-text articles included in this analysis were evaluated and selected by two reviewers working independently. A CSI diagnosis made clinically or with imaging and treatment outcomes were among criteria for the case studies to be included. Dr. Ramakumar said the study, which he claimed is the largest systematic review of CSI ever conducted, has been registered with PROSPERO, an international prospective registry of systematic reviews.
The presenting symptom was fever in 72% of cases and chest pain in the others, although there was one asymptomatic CSI reported. On angiography, 62% had a concomitant mycotic aneurysm. Intramyocardial abscess (13.9%), rupture (11.3%), and coronary fistula (7.5%) were also common findings, but no angiographic abnormalities could be identified in 53% of patients.
Following PCI, most CSI developed within 8 days (43%) or the first month (23%), but CSI was reported more than 6 months after the procedure in 19%. Complex PCI accounted for 51% of cases. Of stent types, 56% were drug eluting and 13% were bare metal.
When comparing characteristics of those who survived CSI with those who did not, most (89%) of those with a non–ST-segment elevated acute coronary syndrome ultimately survived, while survival from CSI in those with structural heart disease was only 17%.
Microbiological findings were not a criterion for study inclusion, but Staphylococcus species accounted for 65% of the infections for which positive cultures were reported. Pseudomonas accounted for 13%. Less than 4% (3.8%) tested positive for multiple pathogens. A small proportion of patients had unusual infectious organisms.
As part of this analysis, the investigators developed an artificial intelligence model to predict CSI based on patient characteristics and other variables. However, the specificity of only around 70% led Dr. Ramakumar to conclude that it does not yet have practical value.
However, he believes that better methodology to detect CSI is needed, and he proposed a diagnostic algorithm that he believes would both improve detection rates and accelerate the time to diagnosis.
Algorithm proposed for detection of CSI
In this algorithm, the first step in symptomatic patients with a positive blood culture suspected of CSI is imaging, such as transthoracic echocardiography, to identify features of infective endocarditis or endarteritis. If the imaging is positive, further imaging, such as PET, that supports the diagnosis, should be adequate to support a diagnosis and treatment.
If initial imaging is negative, alternative diagnoses should be considered, but Dr. Ramakumar advised repeat imaging after 48 hours if symptoms persist and no other causes are found.
Dr. Ramakumar acknowledged the many limitations of this analysis, including the small sample size and the challenges of assembling coherent data from case reports with variable types of information submitted during different eras of PCI evolution. However, reiterating that CSI might be frequently missed, he emphasized that this problem might be bigger than currently understood.
It is difficult to rule out any possibility that CSI is frequently missed, but Andrew Sharp, MD, PhD, a consultant interventional cardiologist at the University Hospital of Wales, Cardiff, is skeptical.
“One might think this is a potential problem, but I cannot think of one patient in whom this has occurred,” Dr. Sharp said in an interview. He is fairly confident that they are extremely rare.
“When there is infection associated with a foreign body, such as a pacemaker, they do not typically resolve by themselves,” he explained. “Often the device has to be removed. If this was true for CSI, then I think we would be aware of these complications.”
However, he praised the investigators for taking a look at CSI in a systematic approach. An invited panelist during the CRT featured research, which is where these data were presented, Dr. Sharp was more interested in understanding why they do not occur now that data are available to suggest they are rare.
“Is there something in the coronary environment, such as the consistent blood flow, that protects against infection?” he asked. CSI is a valid area of further research, according to Dr. Sharp, but he does not consider infected stents to be a common threat based on his own sizable case series.
Dr. Ramakumar and Dr. Sharp reported no potential conflicts of interest.
FROM CRT 2023
Integrating intestinal ultrasound into inflammatory bowel disease training and practice in the United States
Evolving endpoints and treat-to-target strategies in inflammatory bowel disease (IBD) incorporate a need for more frequent assessments of the disease, including objective measures of inflammation.1,2 Intestinal ultrasound (IUS) is a noninvasive, well-tolerated,3 repeatable, point-of-care (POC) test that is highly sensitive and specific in detection of bowel inflammation, transmural healing,4,5 and response to therapy in both Crohn’s disease (CD) and ulcerative colitis (UC).6-8 As IUS is taking hold in the United States, there is a great need to teach the next generation of gastroenterologists about its value, how to incorporate it into clinical practice, and how to become appropriately trained and maintain competency.
Why incorporate IUS in the United States now?
As IBD management has evolved, so has the appreciation for the value of bedside IUS as a tool that addresses very real needs for the field. Unlike other parts of the world in which ultrasound skills are part of the training curriculum, this has not been the case in internal medicine and gastroenterology training in the United States. In addition, there have been no specific billing codes or clear renumeration processes outlined for IUS,9 nor have there been any local training opportunities. Because of these challenges, it was not until recently that several leaders in IBD in the United States championed the potential of this technology and incorporated it into IBD management. Subsequently, a number of gastroenterologists have been trained and are now leading the effort to disseminate this tool throughout the United States. A consequence of these efforts resulted in support from the Helmsley Charitable Trust (Helmsley) and the creation of the Intestinal Ultrasound Group of the United States and Canada to address the gaps unique to North America as well as to strengthen the quality of IUS research through collaborations across the continent.
What is IUS, and when is it performed?
IUS is a sonographic exam performed by a gastroenterology-trained professional who scans the abdominal wall (and perineum when the rectum and perineal disease is evaluated), using both a convex low-frequency probe and linear high-frequency probe to evaluate the small intestine, colon, and rectum. The bowel is composed of five layers with alternating hyperechoic and hypoechoic layers: the mucosal-lumen interface (not a true part of the bowel wall), deep mucosa, submucosa, muscularis propria, and serosa. (Figure)
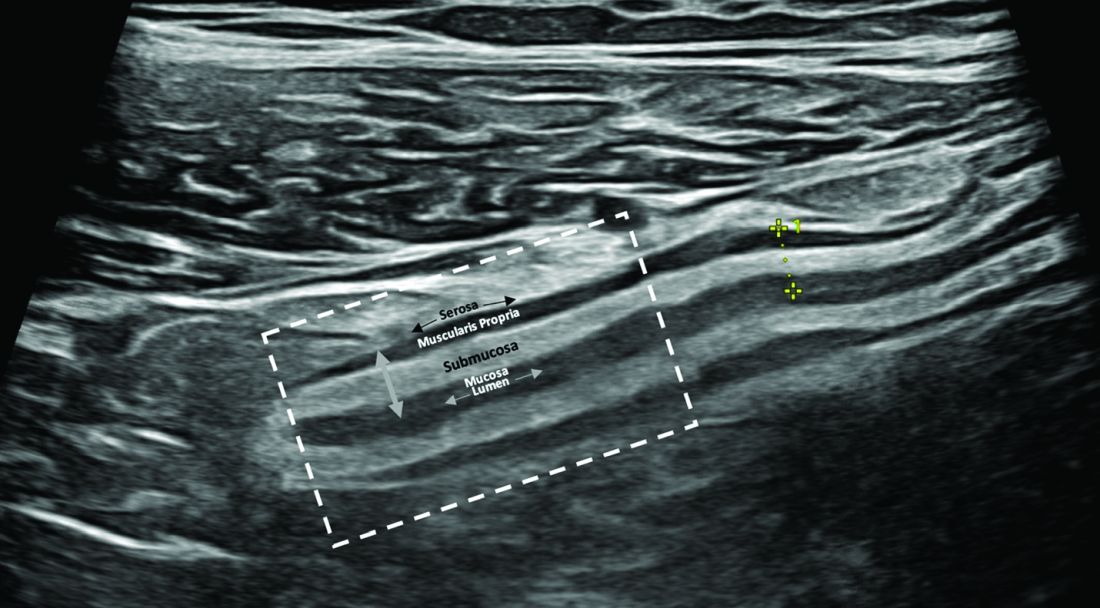
The most sensitive parameter for assessment of IBD activity is bowel wall thickness (≤ 3 mm in the small bowel and colon and ≤ 4 mm in the rectum are considered normal in adults).8,10 The second key parameter is the assessment of vascularization, in which presence of hyperemia suggests active disease.11 There are a number of indices to quantify hyperemia, with the most widely used being the Limberg score.12 Additional parameters include assessment of loss of the delineation of the bowel wall layers (loss of stratification signifies active inflammation), increased thickness of the submucosa,13 increased mesenteric fatty proliferation (with increased inflammation, mesenteric fat proliferation will appear as a hyperechoic area surrounding the bowel), lymphadenopathy, bowel strictures, and extramural complications such as fistulae and abscess. Shear wave elastography may be an effective way to differentiate severe fibrotic strictures, but this is an area that requires more investigation.14
IUS has been shown to be an excellent tool in not only assessing disease activity and disease complication (with higher sensitivity than the Harvey-Bradshaw Index, serum C-reactive protein),15 but, unique to IUS, can provide early prediction of response in moderate to severe active UC.6,7 This has also been shown with transperineal ultrasound in patients with UC, with the ability to predict response to therapy as early as 1 week from induction therapy.16 Furthermore, it can be used to assess transmural healing, which has been shown to be associated with improved outcomes in Crohn’s patient, such as lower rates of hospitalizations, surgery, medication escalation, and need for corticosteroids.17 IUS is associated with great patient satisfaction and greater understanding of disease-related symptoms when the patient sees the inflammation of the bowel. (Table)
How can you get trained in IUS?
Training in IUS varies across the globe, from incorporation of IUS into the standard training curriculum to available training programs that can be followed and attended outside of medical training. In the United States, interested gastroenterologists can now be trained by becoming a member of the International Bowel Ultrasound Group (IBUS Group) and applying to the workshops now available. The IBUS Group has developed an IUS-specific training curriculum over the last 16 years, which is comprised of three modules: a 2-day hands-on workshop (Module 1) with final examination of theoretical competency, a preceptorship at an “expert center” with an experienced sonographer for a total of 4 weeks to complete 40 supervised IUS examinations (Module 2), and didactics and a final examination (Module 3). Also with support from Helmsley, the first Module 1 to be offered in the United States was hosted at Mount Sinai Medical Center in New York in 2022, the second was hosted at the University of Chicago in March 2023, and the third is planned to take place at Cedars-Sinai Medical Center in Los Angeles in March 2024.18 With the growing interest and demand for IUS training in the United States, U.S. experts are working to develop new training options that will be less time consuming, scalable, and still provide appropriate training and competency assessment.
How do you integrate IUS into your practice?
The keys to integrating IUS are a section chief or practice manager’s support of a trainee or faculty member for both funding of equipment and protected time for training and building of the program, as well as a permissive environment and collegial relationship with radiology. An ultrasound machine and additional transducers may range in price from $50,000-$120,000. Funding may be a limiting step for many, however. A detailed business plan is imperative to the success and investment of funds in an IUS program. With current billing practices in place that include ”limited abdominal ultrasound” (76705) and “Doppler ultrasound of the abdomen” (93975),19 reimbursement should include a technical fee, professional fee, and if in a hospital-based clinic, a facility fee. IUS pro-fee combined with technical fee is reimbursed at approximately 0.80 relative value units. When possible, the facility fee is included for approximately $800 per IUS visit. For billing and compliance with HIPAA, all billed IUS images must be stored in a durable and accessible format. It is recommended that the images and cine loops be digitally stored to the same or similar platform used by radiologists at the same institution. This requires early communication with the local information technology department for the connection of an ultrasound machine to the storage platform and/or electronic health record. Reporting results should be standardized with unique or otherwise available IUS templates, which also satisfy all billing components.9 The flow for incorporation of IUS into practice can be at the same time patients are seen during their visit, or alternatively, in a dedicated IUS clinic in which patients are referred by other providers and scheduled back to back.
Conclusions
In summary, the confluence of treat-to-target strategies in IBD, new treatment options in IBD, and successful efforts to translate IUS training and billing practices to the United States portends a great future for the field and for our patients.
Dr. Cleveland and Dr. Rubin, of the University of Chicago’s Inflammatory Bowel Disease Center, are speakers for Samsung/Boston Imaging.
References
1. Turner D et al. Gastroenterology. Apr 2021;160(5):1570-83. doi: 10.1053/j.gastro.2020.12.031
2. Hart AL and Rubin DT. Gastroenterology. Apr 2022;162(5):1367-9. doi: 10.1053/j.gastro.2022.02.013
3. Rajagopalan A et al. JGH Open. Apr 2020;4(2):267-72. doi: 10.1002/jgh3.12268
4. Calabrese E et al. Clin Gastroenterol Hepatol. Apr 2022;20(4):e711-22. doi: 10.1016/j.cgh.2021.03.030
5. Ripolles T et al. Inflamm Bowel Dis. Oct 2016;22(10):2465-73. doi10.1097/MIB.0000000000000882
6. Maaser C et al. Gut. Sep 2020;69(9):1629-36. doi: 10.1136/gutjnl-2019-319451
7. Ilvemark J et al. J Crohns Colitis. Nov 23 2022;16(11):1725-34. doi: 10.1093/ecco-jcc/jjac083
8. Sagami S et al. Aliment Pharmacol Ther. Jun 2020;51(12):1373-83. doi: 10.1111/apt.15767
9. Dolinger MT et al. Guide to Intestinal Ultrasound Credentialing, Documentation, and Billing for Gastroenterologists in the United States. Am J Gastroenterol. 2023.
10. Maconi G et al. Ultraschall Med. Jun 2018;39(3):304-17. doi: 10.1055/s-0043-125329
11. Sasaki T et al. Scand J Gastroenterol. Mar 2014;49(3):295-301. doi: 10.3109/00365521.2013.871744
12. Limberg B. Z Gastroenterol. Jun 1999;37(6):495-508.
13. Miyoshi J et al. J Gastroenterol. Feb 2022;57(2):82-9. doi: 10.1007/s00535-021-01847-3
14. Chen YJ et al. Inflamm Bowel Dis. Sep 15 2018;24(10):2183-90. doi: 10.1093/ibd/izy115
15. Kucharzik T et al. Apr 2017;15(4):535-42e2. doi: 10.1016/j.cgh.2016.10.040
16. Sagami S et al. Aliment Pharmacol Ther. May 2022;55(10):1320-9. doi: 10.1111/apt.16817
17. Vaughan R et al. Aliment Pharmacol Ther. Jul 2022;56(1):84-94. doi: 10.1111/apt.16892
18. International Bowel Ultrasound Group. https://ibus-group.org/
19. American Medical Association. CPT (Current Procedural Terminology). https://www.ama-assn.org/amaone/cpt-current-procedural-terminology
Evolving endpoints and treat-to-target strategies in inflammatory bowel disease (IBD) incorporate a need for more frequent assessments of the disease, including objective measures of inflammation.1,2 Intestinal ultrasound (IUS) is a noninvasive, well-tolerated,3 repeatable, point-of-care (POC) test that is highly sensitive and specific in detection of bowel inflammation, transmural healing,4,5 and response to therapy in both Crohn’s disease (CD) and ulcerative colitis (UC).6-8 As IUS is taking hold in the United States, there is a great need to teach the next generation of gastroenterologists about its value, how to incorporate it into clinical practice, and how to become appropriately trained and maintain competency.
Why incorporate IUS in the United States now?
As IBD management has evolved, so has the appreciation for the value of bedside IUS as a tool that addresses very real needs for the field. Unlike other parts of the world in which ultrasound skills are part of the training curriculum, this has not been the case in internal medicine and gastroenterology training in the United States. In addition, there have been no specific billing codes or clear renumeration processes outlined for IUS,9 nor have there been any local training opportunities. Because of these challenges, it was not until recently that several leaders in IBD in the United States championed the potential of this technology and incorporated it into IBD management. Subsequently, a number of gastroenterologists have been trained and are now leading the effort to disseminate this tool throughout the United States. A consequence of these efforts resulted in support from the Helmsley Charitable Trust (Helmsley) and the creation of the Intestinal Ultrasound Group of the United States and Canada to address the gaps unique to North America as well as to strengthen the quality of IUS research through collaborations across the continent.
What is IUS, and when is it performed?
IUS is a sonographic exam performed by a gastroenterology-trained professional who scans the abdominal wall (and perineum when the rectum and perineal disease is evaluated), using both a convex low-frequency probe and linear high-frequency probe to evaluate the small intestine, colon, and rectum. The bowel is composed of five layers with alternating hyperechoic and hypoechoic layers: the mucosal-lumen interface (not a true part of the bowel wall), deep mucosa, submucosa, muscularis propria, and serosa. (Figure)

The most sensitive parameter for assessment of IBD activity is bowel wall thickness (≤ 3 mm in the small bowel and colon and ≤ 4 mm in the rectum are considered normal in adults).8,10 The second key parameter is the assessment of vascularization, in which presence of hyperemia suggests active disease.11 There are a number of indices to quantify hyperemia, with the most widely used being the Limberg score.12 Additional parameters include assessment of loss of the delineation of the bowel wall layers (loss of stratification signifies active inflammation), increased thickness of the submucosa,13 increased mesenteric fatty proliferation (with increased inflammation, mesenteric fat proliferation will appear as a hyperechoic area surrounding the bowel), lymphadenopathy, bowel strictures, and extramural complications such as fistulae and abscess. Shear wave elastography may be an effective way to differentiate severe fibrotic strictures, but this is an area that requires more investigation.14
IUS has been shown to be an excellent tool in not only assessing disease activity and disease complication (with higher sensitivity than the Harvey-Bradshaw Index, serum C-reactive protein),15 but, unique to IUS, can provide early prediction of response in moderate to severe active UC.6,7 This has also been shown with transperineal ultrasound in patients with UC, with the ability to predict response to therapy as early as 1 week from induction therapy.16 Furthermore, it can be used to assess transmural healing, which has been shown to be associated with improved outcomes in Crohn’s patient, such as lower rates of hospitalizations, surgery, medication escalation, and need for corticosteroids.17 IUS is associated with great patient satisfaction and greater understanding of disease-related symptoms when the patient sees the inflammation of the bowel. (Table)
How can you get trained in IUS?
Training in IUS varies across the globe, from incorporation of IUS into the standard training curriculum to available training programs that can be followed and attended outside of medical training. In the United States, interested gastroenterologists can now be trained by becoming a member of the International Bowel Ultrasound Group (IBUS Group) and applying to the workshops now available. The IBUS Group has developed an IUS-specific training curriculum over the last 16 years, which is comprised of three modules: a 2-day hands-on workshop (Module 1) with final examination of theoretical competency, a preceptorship at an “expert center” with an experienced sonographer for a total of 4 weeks to complete 40 supervised IUS examinations (Module 2), and didactics and a final examination (Module 3). Also with support from Helmsley, the first Module 1 to be offered in the United States was hosted at Mount Sinai Medical Center in New York in 2022, the second was hosted at the University of Chicago in March 2023, and the third is planned to take place at Cedars-Sinai Medical Center in Los Angeles in March 2024.18 With the growing interest and demand for IUS training in the United States, U.S. experts are working to develop new training options that will be less time consuming, scalable, and still provide appropriate training and competency assessment.
How do you integrate IUS into your practice?
The keys to integrating IUS are a section chief or practice manager’s support of a trainee or faculty member for both funding of equipment and protected time for training and building of the program, as well as a permissive environment and collegial relationship with radiology. An ultrasound machine and additional transducers may range in price from $50,000-$120,000. Funding may be a limiting step for many, however. A detailed business plan is imperative to the success and investment of funds in an IUS program. With current billing practices in place that include ”limited abdominal ultrasound” (76705) and “Doppler ultrasound of the abdomen” (93975),19 reimbursement should include a technical fee, professional fee, and if in a hospital-based clinic, a facility fee. IUS pro-fee combined with technical fee is reimbursed at approximately 0.80 relative value units. When possible, the facility fee is included for approximately $800 per IUS visit. For billing and compliance with HIPAA, all billed IUS images must be stored in a durable and accessible format. It is recommended that the images and cine loops be digitally stored to the same or similar platform used by radiologists at the same institution. This requires early communication with the local information technology department for the connection of an ultrasound machine to the storage platform and/or electronic health record. Reporting results should be standardized with unique or otherwise available IUS templates, which also satisfy all billing components.9 The flow for incorporation of IUS into practice can be at the same time patients are seen during their visit, or alternatively, in a dedicated IUS clinic in which patients are referred by other providers and scheduled back to back.
Conclusions
In summary, the confluence of treat-to-target strategies in IBD, new treatment options in IBD, and successful efforts to translate IUS training and billing practices to the United States portends a great future for the field and for our patients.
Dr. Cleveland and Dr. Rubin, of the University of Chicago’s Inflammatory Bowel Disease Center, are speakers for Samsung/Boston Imaging.
References
1. Turner D et al. Gastroenterology. Apr 2021;160(5):1570-83. doi: 10.1053/j.gastro.2020.12.031
2. Hart AL and Rubin DT. Gastroenterology. Apr 2022;162(5):1367-9. doi: 10.1053/j.gastro.2022.02.013
3. Rajagopalan A et al. JGH Open. Apr 2020;4(2):267-72. doi: 10.1002/jgh3.12268
4. Calabrese E et al. Clin Gastroenterol Hepatol. Apr 2022;20(4):e711-22. doi: 10.1016/j.cgh.2021.03.030
5. Ripolles T et al. Inflamm Bowel Dis. Oct 2016;22(10):2465-73. doi10.1097/MIB.0000000000000882
6. Maaser C et al. Gut. Sep 2020;69(9):1629-36. doi: 10.1136/gutjnl-2019-319451
7. Ilvemark J et al. J Crohns Colitis. Nov 23 2022;16(11):1725-34. doi: 10.1093/ecco-jcc/jjac083
8. Sagami S et al. Aliment Pharmacol Ther. Jun 2020;51(12):1373-83. doi: 10.1111/apt.15767
9. Dolinger MT et al. Guide to Intestinal Ultrasound Credentialing, Documentation, and Billing for Gastroenterologists in the United States. Am J Gastroenterol. 2023.
10. Maconi G et al. Ultraschall Med. Jun 2018;39(3):304-17. doi: 10.1055/s-0043-125329
11. Sasaki T et al. Scand J Gastroenterol. Mar 2014;49(3):295-301. doi: 10.3109/00365521.2013.871744
12. Limberg B. Z Gastroenterol. Jun 1999;37(6):495-508.
13. Miyoshi J et al. J Gastroenterol. Feb 2022;57(2):82-9. doi: 10.1007/s00535-021-01847-3
14. Chen YJ et al. Inflamm Bowel Dis. Sep 15 2018;24(10):2183-90. doi: 10.1093/ibd/izy115
15. Kucharzik T et al. Apr 2017;15(4):535-42e2. doi: 10.1016/j.cgh.2016.10.040
16. Sagami S et al. Aliment Pharmacol Ther. May 2022;55(10):1320-9. doi: 10.1111/apt.16817
17. Vaughan R et al. Aliment Pharmacol Ther. Jul 2022;56(1):84-94. doi: 10.1111/apt.16892
18. International Bowel Ultrasound Group. https://ibus-group.org/
19. American Medical Association. CPT (Current Procedural Terminology). https://www.ama-assn.org/amaone/cpt-current-procedural-terminology
Evolving endpoints and treat-to-target strategies in inflammatory bowel disease (IBD) incorporate a need for more frequent assessments of the disease, including objective measures of inflammation.1,2 Intestinal ultrasound (IUS) is a noninvasive, well-tolerated,3 repeatable, point-of-care (POC) test that is highly sensitive and specific in detection of bowel inflammation, transmural healing,4,5 and response to therapy in both Crohn’s disease (CD) and ulcerative colitis (UC).6-8 As IUS is taking hold in the United States, there is a great need to teach the next generation of gastroenterologists about its value, how to incorporate it into clinical practice, and how to become appropriately trained and maintain competency.
Why incorporate IUS in the United States now?
As IBD management has evolved, so has the appreciation for the value of bedside IUS as a tool that addresses very real needs for the field. Unlike other parts of the world in which ultrasound skills are part of the training curriculum, this has not been the case in internal medicine and gastroenterology training in the United States. In addition, there have been no specific billing codes or clear renumeration processes outlined for IUS,9 nor have there been any local training opportunities. Because of these challenges, it was not until recently that several leaders in IBD in the United States championed the potential of this technology and incorporated it into IBD management. Subsequently, a number of gastroenterologists have been trained and are now leading the effort to disseminate this tool throughout the United States. A consequence of these efforts resulted in support from the Helmsley Charitable Trust (Helmsley) and the creation of the Intestinal Ultrasound Group of the United States and Canada to address the gaps unique to North America as well as to strengthen the quality of IUS research through collaborations across the continent.
What is IUS, and when is it performed?
IUS is a sonographic exam performed by a gastroenterology-trained professional who scans the abdominal wall (and perineum when the rectum and perineal disease is evaluated), using both a convex low-frequency probe and linear high-frequency probe to evaluate the small intestine, colon, and rectum. The bowel is composed of five layers with alternating hyperechoic and hypoechoic layers: the mucosal-lumen interface (not a true part of the bowel wall), deep mucosa, submucosa, muscularis propria, and serosa. (Figure)

The most sensitive parameter for assessment of IBD activity is bowel wall thickness (≤ 3 mm in the small bowel and colon and ≤ 4 mm in the rectum are considered normal in adults).8,10 The second key parameter is the assessment of vascularization, in which presence of hyperemia suggests active disease.11 There are a number of indices to quantify hyperemia, with the most widely used being the Limberg score.12 Additional parameters include assessment of loss of the delineation of the bowel wall layers (loss of stratification signifies active inflammation), increased thickness of the submucosa,13 increased mesenteric fatty proliferation (with increased inflammation, mesenteric fat proliferation will appear as a hyperechoic area surrounding the bowel), lymphadenopathy, bowel strictures, and extramural complications such as fistulae and abscess. Shear wave elastography may be an effective way to differentiate severe fibrotic strictures, but this is an area that requires more investigation.14
IUS has been shown to be an excellent tool in not only assessing disease activity and disease complication (with higher sensitivity than the Harvey-Bradshaw Index, serum C-reactive protein),15 but, unique to IUS, can provide early prediction of response in moderate to severe active UC.6,7 This has also been shown with transperineal ultrasound in patients with UC, with the ability to predict response to therapy as early as 1 week from induction therapy.16 Furthermore, it can be used to assess transmural healing, which has been shown to be associated with improved outcomes in Crohn’s patient, such as lower rates of hospitalizations, surgery, medication escalation, and need for corticosteroids.17 IUS is associated with great patient satisfaction and greater understanding of disease-related symptoms when the patient sees the inflammation of the bowel. (Table)
How can you get trained in IUS?
Training in IUS varies across the globe, from incorporation of IUS into the standard training curriculum to available training programs that can be followed and attended outside of medical training. In the United States, interested gastroenterologists can now be trained by becoming a member of the International Bowel Ultrasound Group (IBUS Group) and applying to the workshops now available. The IBUS Group has developed an IUS-specific training curriculum over the last 16 years, which is comprised of three modules: a 2-day hands-on workshop (Module 1) with final examination of theoretical competency, a preceptorship at an “expert center” with an experienced sonographer for a total of 4 weeks to complete 40 supervised IUS examinations (Module 2), and didactics and a final examination (Module 3). Also with support from Helmsley, the first Module 1 to be offered in the United States was hosted at Mount Sinai Medical Center in New York in 2022, the second was hosted at the University of Chicago in March 2023, and the third is planned to take place at Cedars-Sinai Medical Center in Los Angeles in March 2024.18 With the growing interest and demand for IUS training in the United States, U.S. experts are working to develop new training options that will be less time consuming, scalable, and still provide appropriate training and competency assessment.
How do you integrate IUS into your practice?
The keys to integrating IUS are a section chief or practice manager’s support of a trainee or faculty member for both funding of equipment and protected time for training and building of the program, as well as a permissive environment and collegial relationship with radiology. An ultrasound machine and additional transducers may range in price from $50,000-$120,000. Funding may be a limiting step for many, however. A detailed business plan is imperative to the success and investment of funds in an IUS program. With current billing practices in place that include ”limited abdominal ultrasound” (76705) and “Doppler ultrasound of the abdomen” (93975),19 reimbursement should include a technical fee, professional fee, and if in a hospital-based clinic, a facility fee. IUS pro-fee combined with technical fee is reimbursed at approximately 0.80 relative value units. When possible, the facility fee is included for approximately $800 per IUS visit. For billing and compliance with HIPAA, all billed IUS images must be stored in a durable and accessible format. It is recommended that the images and cine loops be digitally stored to the same or similar platform used by radiologists at the same institution. This requires early communication with the local information technology department for the connection of an ultrasound machine to the storage platform and/or electronic health record. Reporting results should be standardized with unique or otherwise available IUS templates, which also satisfy all billing components.9 The flow for incorporation of IUS into practice can be at the same time patients are seen during their visit, or alternatively, in a dedicated IUS clinic in which patients are referred by other providers and scheduled back to back.
Conclusions
In summary, the confluence of treat-to-target strategies in IBD, new treatment options in IBD, and successful efforts to translate IUS training and billing practices to the United States portends a great future for the field and for our patients.
Dr. Cleveland and Dr. Rubin, of the University of Chicago’s Inflammatory Bowel Disease Center, are speakers for Samsung/Boston Imaging.
References
1. Turner D et al. Gastroenterology. Apr 2021;160(5):1570-83. doi: 10.1053/j.gastro.2020.12.031
2. Hart AL and Rubin DT. Gastroenterology. Apr 2022;162(5):1367-9. doi: 10.1053/j.gastro.2022.02.013
3. Rajagopalan A et al. JGH Open. Apr 2020;4(2):267-72. doi: 10.1002/jgh3.12268
4. Calabrese E et al. Clin Gastroenterol Hepatol. Apr 2022;20(4):e711-22. doi: 10.1016/j.cgh.2021.03.030
5. Ripolles T et al. Inflamm Bowel Dis. Oct 2016;22(10):2465-73. doi10.1097/MIB.0000000000000882
6. Maaser C et al. Gut. Sep 2020;69(9):1629-36. doi: 10.1136/gutjnl-2019-319451
7. Ilvemark J et al. J Crohns Colitis. Nov 23 2022;16(11):1725-34. doi: 10.1093/ecco-jcc/jjac083
8. Sagami S et al. Aliment Pharmacol Ther. Jun 2020;51(12):1373-83. doi: 10.1111/apt.15767
9. Dolinger MT et al. Guide to Intestinal Ultrasound Credentialing, Documentation, and Billing for Gastroenterologists in the United States. Am J Gastroenterol. 2023.
10. Maconi G et al. Ultraschall Med. Jun 2018;39(3):304-17. doi: 10.1055/s-0043-125329
11. Sasaki T et al. Scand J Gastroenterol. Mar 2014;49(3):295-301. doi: 10.3109/00365521.2013.871744
12. Limberg B. Z Gastroenterol. Jun 1999;37(6):495-508.
13. Miyoshi J et al. J Gastroenterol. Feb 2022;57(2):82-9. doi: 10.1007/s00535-021-01847-3
14. Chen YJ et al. Inflamm Bowel Dis. Sep 15 2018;24(10):2183-90. doi: 10.1093/ibd/izy115
15. Kucharzik T et al. Apr 2017;15(4):535-42e2. doi: 10.1016/j.cgh.2016.10.040
16. Sagami S et al. Aliment Pharmacol Ther. May 2022;55(10):1320-9. doi: 10.1111/apt.16817
17. Vaughan R et al. Aliment Pharmacol Ther. Jul 2022;56(1):84-94. doi: 10.1111/apt.16892
18. International Bowel Ultrasound Group. https://ibus-group.org/
19. American Medical Association. CPT (Current Procedural Terminology). https://www.ama-assn.org/amaone/cpt-current-procedural-terminology
A better MS measure?
“When you measure disability, what you really want to know is how things are changing in the patient’s life and not your perception of how they’re changing,” said Mark Gudesblatt, MD, who presented a study comparing the technique, called quantitative gait analysis, to other measures at a poster session during the annual meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS).
The device, called Protokinetics, has been used in clinical studies for Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, stroke, Friederich’s ataxia, and other conditions. The device is a digitized carpet that senses weight change and pressure as the individual walks.
“We can actually measure performance, and the performance is not just how fast you walk 25 feet. We’re measuring things that underlie how you walk: step length, step length variability, velocity, weight shift, how much time you spend on one leg. So it’s like listening to a symphony. We’re not measuring just the trumpets or the violins, we’re measuring everything,” said Dr. Gudesblatt, who is medical director of the Comprehensive MS Center at South Shore Neurologic Associates, Patchogue, N.Y.
Commonly used measures include the Expanded Disability Status Scale (EDSS), the 25-foot time walk (25’TW), and the Timed Up and Go (TUG).
Those measures are useful but don’t really measure up to clinical need, Dr. Gudesblatt said. “What you want is no evidence of disease activity, whether that’s multiple dimensions of thinking or multiple dimensions of walking, or changes on an MRI that are not the radiologist’s impression. Patients always say: ‘Doc, I’m worse.’ And we say: ‘Well, your exam is unchanged, your MRI has not changed. But they are worse for reasons – either their perception or their performance. So you can measure this very granularly, and you can relate it to their fear of falling, their balance confidence. This ups the game,” said Dr. Gudesblatt.
“And here’s where it gets even more interesting. You can use this for signatures of disease,” he added. The data can, for example, suggest that instead of Parkinson’s disease, a patient may have a Parkinson’s variant. “What we’re doing is showing how the 25-foot timed walk and Timed Up and Go are very traditional, conservative measures. They’re equivalent to the Pony Express. They’re good, but not where you want to be.”
Technology provides more sensitive, but more complex data
Digital tools to measure a variety of functions, including gait, cognition, and upper limb function are becoming increasingly common in MS, according to Catherine Larochelle, MD, PhD, who was asked for comment. “They are easily providing measures that are likely more sensitive and diverse and probably more meaningful about the daily functional status of a person than our usual EDSS,” said Dr. Larochelle, who is an associate professor at Université de Montréal.
The next step is to determine how best to use the complex data that such devices generate. “Lots of research is being done to better understand how to use the rich but complex data obtained with these tools to provide useful information to people with MS and their clinical team, to help guide shared clinical decisions, and likely accelerate and improve outcomes in clinical trials. So this is a very exciting new era in terms of clinical neurological assessment,” said Dr. Larochelle.
Granular gait analysis
Dr. Gudesblatt and colleagues analyzed retrospective data from 105 people with MS (69% female; average age, 53.7 years). Participants underwent all tests on the same day. The digital gait analysis captured velocity, double support, cadence, functional ambulation profile, gait variability index, and walk ratio over three trials conducted at preferred walking speed (PWS) and during dual task walking.
There were statistically significant relationships (P ≤ .01) between TUG and 25’TW (R2 = 0.62). There were also significant relationships between 25’TW and digital parameters measured at PWS: velocity (R2 = 0.63); double support (R2 = 0.74); cadence (R2 = 0.56); and gait variability index (R2 = 0.54). During dual task walking, there were relationships between 25’TW and velocity (R2 = 0.53); double support (R2 = 0.30); cadence (R2 = 0.43); and gait variability index (R2 = 0.46).
TUG values were significantly associated with gait parameters during PWS: velocity (R2 = 0.71); double support (R2 = 0.75); cadence (R2 = 0.43); gait variability index (R2 = 0.45); and walk ratio (R2 = 0.06). During dual task walking, TUG values were significantly associated with velocity (R2 = 0.55), double support (R2 = 0.21), cadence (R2 = 0.45), and gait variability index (R2 = 0.39).
“With the availability multiple effective disease modifying therapies and the future potential of restorative or reparative treatments, more granular, validated standardized outcome measures are urgently needed,” said Dr. Gudesblatt. Analysis of gait cycle can provide clinically useful information not adequately captured by the current, more traditional approaches of measuring outcomes in MS.
Dr. Gudesblatt and Dr. Larochelle have no relevant financial disclosures.
“When you measure disability, what you really want to know is how things are changing in the patient’s life and not your perception of how they’re changing,” said Mark Gudesblatt, MD, who presented a study comparing the technique, called quantitative gait analysis, to other measures at a poster session during the annual meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS).
The device, called Protokinetics, has been used in clinical studies for Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, stroke, Friederich’s ataxia, and other conditions. The device is a digitized carpet that senses weight change and pressure as the individual walks.
“We can actually measure performance, and the performance is not just how fast you walk 25 feet. We’re measuring things that underlie how you walk: step length, step length variability, velocity, weight shift, how much time you spend on one leg. So it’s like listening to a symphony. We’re not measuring just the trumpets or the violins, we’re measuring everything,” said Dr. Gudesblatt, who is medical director of the Comprehensive MS Center at South Shore Neurologic Associates, Patchogue, N.Y.
Commonly used measures include the Expanded Disability Status Scale (EDSS), the 25-foot time walk (25’TW), and the Timed Up and Go (TUG).
Those measures are useful but don’t really measure up to clinical need, Dr. Gudesblatt said. “What you want is no evidence of disease activity, whether that’s multiple dimensions of thinking or multiple dimensions of walking, or changes on an MRI that are not the radiologist’s impression. Patients always say: ‘Doc, I’m worse.’ And we say: ‘Well, your exam is unchanged, your MRI has not changed. But they are worse for reasons – either their perception or their performance. So you can measure this very granularly, and you can relate it to their fear of falling, their balance confidence. This ups the game,” said Dr. Gudesblatt.
“And here’s where it gets even more interesting. You can use this for signatures of disease,” he added. The data can, for example, suggest that instead of Parkinson’s disease, a patient may have a Parkinson’s variant. “What we’re doing is showing how the 25-foot timed walk and Timed Up and Go are very traditional, conservative measures. They’re equivalent to the Pony Express. They’re good, but not where you want to be.”
Technology provides more sensitive, but more complex data
Digital tools to measure a variety of functions, including gait, cognition, and upper limb function are becoming increasingly common in MS, according to Catherine Larochelle, MD, PhD, who was asked for comment. “They are easily providing measures that are likely more sensitive and diverse and probably more meaningful about the daily functional status of a person than our usual EDSS,” said Dr. Larochelle, who is an associate professor at Université de Montréal.
The next step is to determine how best to use the complex data that such devices generate. “Lots of research is being done to better understand how to use the rich but complex data obtained with these tools to provide useful information to people with MS and their clinical team, to help guide shared clinical decisions, and likely accelerate and improve outcomes in clinical trials. So this is a very exciting new era in terms of clinical neurological assessment,” said Dr. Larochelle.
Granular gait analysis
Dr. Gudesblatt and colleagues analyzed retrospective data from 105 people with MS (69% female; average age, 53.7 years). Participants underwent all tests on the same day. The digital gait analysis captured velocity, double support, cadence, functional ambulation profile, gait variability index, and walk ratio over three trials conducted at preferred walking speed (PWS) and during dual task walking.
There were statistically significant relationships (P ≤ .01) between TUG and 25’TW (R2 = 0.62). There were also significant relationships between 25’TW and digital parameters measured at PWS: velocity (R2 = 0.63); double support (R2 = 0.74); cadence (R2 = 0.56); and gait variability index (R2 = 0.54). During dual task walking, there were relationships between 25’TW and velocity (R2 = 0.53); double support (R2 = 0.30); cadence (R2 = 0.43); and gait variability index (R2 = 0.46).
TUG values were significantly associated with gait parameters during PWS: velocity (R2 = 0.71); double support (R2 = 0.75); cadence (R2 = 0.43); gait variability index (R2 = 0.45); and walk ratio (R2 = 0.06). During dual task walking, TUG values were significantly associated with velocity (R2 = 0.55), double support (R2 = 0.21), cadence (R2 = 0.45), and gait variability index (R2 = 0.39).
“With the availability multiple effective disease modifying therapies and the future potential of restorative or reparative treatments, more granular, validated standardized outcome measures are urgently needed,” said Dr. Gudesblatt. Analysis of gait cycle can provide clinically useful information not adequately captured by the current, more traditional approaches of measuring outcomes in MS.
Dr. Gudesblatt and Dr. Larochelle have no relevant financial disclosures.
“When you measure disability, what you really want to know is how things are changing in the patient’s life and not your perception of how they’re changing,” said Mark Gudesblatt, MD, who presented a study comparing the technique, called quantitative gait analysis, to other measures at a poster session during the annual meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS).
The device, called Protokinetics, has been used in clinical studies for Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, stroke, Friederich’s ataxia, and other conditions. The device is a digitized carpet that senses weight change and pressure as the individual walks.
“We can actually measure performance, and the performance is not just how fast you walk 25 feet. We’re measuring things that underlie how you walk: step length, step length variability, velocity, weight shift, how much time you spend on one leg. So it’s like listening to a symphony. We’re not measuring just the trumpets or the violins, we’re measuring everything,” said Dr. Gudesblatt, who is medical director of the Comprehensive MS Center at South Shore Neurologic Associates, Patchogue, N.Y.
Commonly used measures include the Expanded Disability Status Scale (EDSS), the 25-foot time walk (25’TW), and the Timed Up and Go (TUG).
Those measures are useful but don’t really measure up to clinical need, Dr. Gudesblatt said. “What you want is no evidence of disease activity, whether that’s multiple dimensions of thinking or multiple dimensions of walking, or changes on an MRI that are not the radiologist’s impression. Patients always say: ‘Doc, I’m worse.’ And we say: ‘Well, your exam is unchanged, your MRI has not changed. But they are worse for reasons – either their perception or their performance. So you can measure this very granularly, and you can relate it to their fear of falling, their balance confidence. This ups the game,” said Dr. Gudesblatt.
“And here’s where it gets even more interesting. You can use this for signatures of disease,” he added. The data can, for example, suggest that instead of Parkinson’s disease, a patient may have a Parkinson’s variant. “What we’re doing is showing how the 25-foot timed walk and Timed Up and Go are very traditional, conservative measures. They’re equivalent to the Pony Express. They’re good, but not where you want to be.”
Technology provides more sensitive, but more complex data
Digital tools to measure a variety of functions, including gait, cognition, and upper limb function are becoming increasingly common in MS, according to Catherine Larochelle, MD, PhD, who was asked for comment. “They are easily providing measures that are likely more sensitive and diverse and probably more meaningful about the daily functional status of a person than our usual EDSS,” said Dr. Larochelle, who is an associate professor at Université de Montréal.
The next step is to determine how best to use the complex data that such devices generate. “Lots of research is being done to better understand how to use the rich but complex data obtained with these tools to provide useful information to people with MS and their clinical team, to help guide shared clinical decisions, and likely accelerate and improve outcomes in clinical trials. So this is a very exciting new era in terms of clinical neurological assessment,” said Dr. Larochelle.
Granular gait analysis
Dr. Gudesblatt and colleagues analyzed retrospective data from 105 people with MS (69% female; average age, 53.7 years). Participants underwent all tests on the same day. The digital gait analysis captured velocity, double support, cadence, functional ambulation profile, gait variability index, and walk ratio over three trials conducted at preferred walking speed (PWS) and during dual task walking.
There were statistically significant relationships (P ≤ .01) between TUG and 25’TW (R2 = 0.62). There were also significant relationships between 25’TW and digital parameters measured at PWS: velocity (R2 = 0.63); double support (R2 = 0.74); cadence (R2 = 0.56); and gait variability index (R2 = 0.54). During dual task walking, there were relationships between 25’TW and velocity (R2 = 0.53); double support (R2 = 0.30); cadence (R2 = 0.43); and gait variability index (R2 = 0.46).
TUG values were significantly associated with gait parameters during PWS: velocity (R2 = 0.71); double support (R2 = 0.75); cadence (R2 = 0.43); gait variability index (R2 = 0.45); and walk ratio (R2 = 0.06). During dual task walking, TUG values were significantly associated with velocity (R2 = 0.55), double support (R2 = 0.21), cadence (R2 = 0.45), and gait variability index (R2 = 0.39).
“With the availability multiple effective disease modifying therapies and the future potential of restorative or reparative treatments, more granular, validated standardized outcome measures are urgently needed,” said Dr. Gudesblatt. Analysis of gait cycle can provide clinically useful information not adequately captured by the current, more traditional approaches of measuring outcomes in MS.
Dr. Gudesblatt and Dr. Larochelle have no relevant financial disclosures.
FROM ACTRIMS FORUM 2023
Thyroid hormones predict psychotic depression in MDD patients
Thyroid dysfunction is common among major depressive disorder (MDD) patients, but its relationship with the psychotic depression (PD) subtype has not been well studied, wrote Pu Peng, of The Second Xiangya Hospital of Central South University, Changsha, Hunan, China, and colleagues.
Given the significant negative consequences of PD in MDD, including comorbid psychosis, suicidal attempts, and worse prognosis, more ways to identify PD risk factors in MDD are needed, they said. Previous research suggests a role for thyroid hormones in the pathophysiology of PD, but data on specific associations are limited, they noted.
In a study published in Psychiatry Research, the authors recruited 1,718 adults aged 18-60 years with MDD who were treated at a single center. The median age was 34 years, 66% were female, and 10% were identified with PD.
Clinical symptoms were identified using the positive subscale of the Positive and Negative Symptom Scale (PANSS-P), Hamilton Anxiety Rating Scale (HAMA), and Hamilton Depression Rating Scale (HAMD). The median PANSS-P score was 7. The researchers measured serum levels of thyroid stimulating hormone (TSH), anti-thyroglobulin (TgAb), and thyroid peroxidases antibody (TPOAb). Subclinical hyperthyroidism (SCH) was defined as TSH levels greater than 8.0 uIU/L and FT4 within normal values.
Overall, the prevalence of SCH, abnormal TgAb, TPOAb, FT3, and FT4 were 13%, 17%, 25%, <0.1%, and 0.3%, respectively. Serum TSH levels, TgAb levels, and TPOAb levels were significantly higher in PD patients than in non-PD patients. No differences appeared in FT3 and FT4 levels between the two groups.
In a multivariate analysis, subclinical hypothyroidism was associated with a ninefold increased risk of PD (odds ratio, 9.32) as were abnormal TPOAb (OR, 1.89) and abnormal TgAb (OR, 2.09).
The findings were limited by several factors including the cross-sectional design, and the inclusion of participants from only a single center in China, which may limit generalizability, the researchers noted.
In addition, “It should be noted that the association between thyroid hormones and PD was small to moderate and the underlying mechanism remained unexplored,” they said. Other limitations include the use of only 17 of the 20 HAMD items and the lack of data on the relationship between anxiety and depressive features and thyroid dysfunction, they wrote.
More research is needed to confirm the findings in other populations, however; the results suggest that regular thyroid function tests may help with early detection of PD in MDD patients, they concluded.
The study was funded by the CAS Pioneer Hundred Talents Program and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
Thyroid dysfunction is common among major depressive disorder (MDD) patients, but its relationship with the psychotic depression (PD) subtype has not been well studied, wrote Pu Peng, of The Second Xiangya Hospital of Central South University, Changsha, Hunan, China, and colleagues.
Given the significant negative consequences of PD in MDD, including comorbid psychosis, suicidal attempts, and worse prognosis, more ways to identify PD risk factors in MDD are needed, they said. Previous research suggests a role for thyroid hormones in the pathophysiology of PD, but data on specific associations are limited, they noted.
In a study published in Psychiatry Research, the authors recruited 1,718 adults aged 18-60 years with MDD who were treated at a single center. The median age was 34 years, 66% were female, and 10% were identified with PD.
Clinical symptoms were identified using the positive subscale of the Positive and Negative Symptom Scale (PANSS-P), Hamilton Anxiety Rating Scale (HAMA), and Hamilton Depression Rating Scale (HAMD). The median PANSS-P score was 7. The researchers measured serum levels of thyroid stimulating hormone (TSH), anti-thyroglobulin (TgAb), and thyroid peroxidases antibody (TPOAb). Subclinical hyperthyroidism (SCH) was defined as TSH levels greater than 8.0 uIU/L and FT4 within normal values.
Overall, the prevalence of SCH, abnormal TgAb, TPOAb, FT3, and FT4 were 13%, 17%, 25%, <0.1%, and 0.3%, respectively. Serum TSH levels, TgAb levels, and TPOAb levels were significantly higher in PD patients than in non-PD patients. No differences appeared in FT3 and FT4 levels between the two groups.
In a multivariate analysis, subclinical hypothyroidism was associated with a ninefold increased risk of PD (odds ratio, 9.32) as were abnormal TPOAb (OR, 1.89) and abnormal TgAb (OR, 2.09).
The findings were limited by several factors including the cross-sectional design, and the inclusion of participants from only a single center in China, which may limit generalizability, the researchers noted.
In addition, “It should be noted that the association between thyroid hormones and PD was small to moderate and the underlying mechanism remained unexplored,” they said. Other limitations include the use of only 17 of the 20 HAMD items and the lack of data on the relationship between anxiety and depressive features and thyroid dysfunction, they wrote.
More research is needed to confirm the findings in other populations, however; the results suggest that regular thyroid function tests may help with early detection of PD in MDD patients, they concluded.
The study was funded by the CAS Pioneer Hundred Talents Program and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
Thyroid dysfunction is common among major depressive disorder (MDD) patients, but its relationship with the psychotic depression (PD) subtype has not been well studied, wrote Pu Peng, of The Second Xiangya Hospital of Central South University, Changsha, Hunan, China, and colleagues.
Given the significant negative consequences of PD in MDD, including comorbid psychosis, suicidal attempts, and worse prognosis, more ways to identify PD risk factors in MDD are needed, they said. Previous research suggests a role for thyroid hormones in the pathophysiology of PD, but data on specific associations are limited, they noted.
In a study published in Psychiatry Research, the authors recruited 1,718 adults aged 18-60 years with MDD who were treated at a single center. The median age was 34 years, 66% were female, and 10% were identified with PD.
Clinical symptoms were identified using the positive subscale of the Positive and Negative Symptom Scale (PANSS-P), Hamilton Anxiety Rating Scale (HAMA), and Hamilton Depression Rating Scale (HAMD). The median PANSS-P score was 7. The researchers measured serum levels of thyroid stimulating hormone (TSH), anti-thyroglobulin (TgAb), and thyroid peroxidases antibody (TPOAb). Subclinical hyperthyroidism (SCH) was defined as TSH levels greater than 8.0 uIU/L and FT4 within normal values.
Overall, the prevalence of SCH, abnormal TgAb, TPOAb, FT3, and FT4 were 13%, 17%, 25%, <0.1%, and 0.3%, respectively. Serum TSH levels, TgAb levels, and TPOAb levels were significantly higher in PD patients than in non-PD patients. No differences appeared in FT3 and FT4 levels between the two groups.
In a multivariate analysis, subclinical hypothyroidism was associated with a ninefold increased risk of PD (odds ratio, 9.32) as were abnormal TPOAb (OR, 1.89) and abnormal TgAb (OR, 2.09).
The findings were limited by several factors including the cross-sectional design, and the inclusion of participants from only a single center in China, which may limit generalizability, the researchers noted.
In addition, “It should be noted that the association between thyroid hormones and PD was small to moderate and the underlying mechanism remained unexplored,” they said. Other limitations include the use of only 17 of the 20 HAMD items and the lack of data on the relationship between anxiety and depressive features and thyroid dysfunction, they wrote.
More research is needed to confirm the findings in other populations, however; the results suggest that regular thyroid function tests may help with early detection of PD in MDD patients, they concluded.
The study was funded by the CAS Pioneer Hundred Talents Program and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRY RESEARCH
Empathy meltdown? Why burnout busts your empathy levels
Compassion is borne out of a sense of empathy – the ability to understand and share the feelings of others. Studies on empathy show it to be crucial to quality health care and not just for patients.
In one study on empathy ratings among doctors, 87% of the public believe that compassion, or a clear and obvious desire to relieve suffering, is the most critical factor when choosing a doctor. In fact, it eclipses travel time, wait time, and cost on the list of sought-after physician features.
Wendie Trubow, MD, an ob.gyn. in Newton, Mass., with over 25 years of experience in the medical field, says empathy has absolutely helped her be a better physician.
“Patients consistently mention how grateful they are that someone has listened to them and validated them,” she says. “When patients feel heard and validated, they are more likely to communicate openly, and this raises the potential of being able to create treatment plans that they will actually participate in. Ultimately, it enriches patient care.”
Mohammadreza Hojat, PhD, research professor of psychiatry and human behavior at the Asano-Gonnella Center for Research in Medical Education and Health Care at Thomas Jefferson University in Philadelphia, says that empirical research he and colleagues have done on empathy in health profession education and patient care over the past 20 years shows that empathic engagement in patient care is reciprocally beneficial for both clinicians and patients.
For example, Dr. Hojat notes that in one study, diabetic patients treated by empathic physicians (measured by the Jefferson Scale of Empathy) had more control over their disease when measured with laboratory test results such as hemoglobin A1c and LDL-C. In another, patients with diabetes treated by more empathic physicians had significantly lower rates of acute metabolic complications that required hospitalization.
For physicians, empathic relationships with your patients lead to fewer disputes, higher reimbursements, greater patient satisfaction, fewer malpractice lawsuits, and a more rewarding experience treating patients.
Different types of empathy
The importance of empathy in doctoring is evident, but Dr. Hojat says it’s crucial to differentiate between clinical empathy and emotional empathy. One can enhance care, while the other, when overused, may lead to physician burnout.
In fact, he says, clinical empathy and emotional empathy have different consequences in a medical setting.
“The relationship between clinical empathy and clinical outcomes is linear, meaning that more empathic engagement leads to more positive clinical outcomes,” says Dr. Hojat. “However, the relationship between emotional empathy and clinical outcomes is curvilinear, or an inverted U shape, similar to the association between anxiety and performance, meaning that limited emotional empathy or limited sympathetic engagement could be helpful, but its overabundance can hamper clinical relationships and objective clinical decision-making.”
The takeaway is that when physicians don’t regulate their emotional empathy, it becomes an obstacle to clinical empathy, ultimately detrimental to health care outcomes.
When burnout hinders empathy
Of course, the reverse is also true – burnout can make it harder for physicians to muster up empathy of any kind toward their patients. At least 53% of physicians show one or more symptoms of burnout, such as exhaustion, questioning the point of the work, cynicism, sarcasm, and the need to “vent” about patients or the job, according to Medscape’s ‘I Cry but No One Cares’: Physician Burnout & Depression Report 2023.
Venting about patients can also be called “compassion fatigue,” which is a sign that your ability to empathize with patients is compromised. You can still practice medicine, but you’re not operating anywhere close to your optimum abilities.
“Generally, physicians who are burned out struggle with empathy since it’s exactly what they’re missing for themselves, and [they] often find it difficult to generate,” says Dr. Trubow.
How to manage burnout and boost your empathy
Burnout can happen for various reasons – pressure to cycle through scores of patients, too many bureaucratic tasks, less autonomy, frustration with electronic health record requirements, and too many work hours, according to the Medscape report.
A report in Family Practice Management finds there are two main goals for physicians to tackle when trying to reduce burnout symptoms: Lower your stress levels and improve your ability to recharge your energy accounts.
“For physicians experiencing burnout [and thus, a lack of empathy], the best approach to this situation is to first take a break and evaluate whether there are any structures to put in place to improve the situation; this can often improve a provider’s empathy,” says Dr. Trubow.
For example, physicians can look at ways to alleviate burnout by investing in leadership development, finding flexible work arrangements, reducing technological burdens, and limiting nonclinical activities.
Other strategies that can build up your reserves include connecting with colleagues, gaining a greater sense of control over your work, and having opportunities to grow and excel in your field. This requires not only a personal approach by physicians, but a buy-in at an institutional level as well.
In Medscape’s report, where 65% of physicians say burnout affects their relationships, physicians’ coping methods include exercise, time with family and friends, time alone, sleep, music, and meditation.
“Clinical empathy must be placed in the realm of ‘evidence-based’ medicine,” says Dr. Hojat. “Given our research findings that clinical empathy tends to erode as students progress through medical school, it is important that assessment and enhancement of clinical empathy be integrated into formal educational curriculum of medical schools and postgraduate training programs for professional development of physicians–in-training and –in-practice.”
“Burnout also leads to a large swath of physicians who aren’t as empathetic toward their patients as they could be.”
–Danielle Ofri, “What Doctors Feel: How Emotions Affect the Practice of Medicine”.
A version of this article first appeared on Medscape.com.
Compassion is borne out of a sense of empathy – the ability to understand and share the feelings of others. Studies on empathy show it to be crucial to quality health care and not just for patients.
In one study on empathy ratings among doctors, 87% of the public believe that compassion, or a clear and obvious desire to relieve suffering, is the most critical factor when choosing a doctor. In fact, it eclipses travel time, wait time, and cost on the list of sought-after physician features.
Wendie Trubow, MD, an ob.gyn. in Newton, Mass., with over 25 years of experience in the medical field, says empathy has absolutely helped her be a better physician.
“Patients consistently mention how grateful they are that someone has listened to them and validated them,” she says. “When patients feel heard and validated, they are more likely to communicate openly, and this raises the potential of being able to create treatment plans that they will actually participate in. Ultimately, it enriches patient care.”
Mohammadreza Hojat, PhD, research professor of psychiatry and human behavior at the Asano-Gonnella Center for Research in Medical Education and Health Care at Thomas Jefferson University in Philadelphia, says that empirical research he and colleagues have done on empathy in health profession education and patient care over the past 20 years shows that empathic engagement in patient care is reciprocally beneficial for both clinicians and patients.
For example, Dr. Hojat notes that in one study, diabetic patients treated by empathic physicians (measured by the Jefferson Scale of Empathy) had more control over their disease when measured with laboratory test results such as hemoglobin A1c and LDL-C. In another, patients with diabetes treated by more empathic physicians had significantly lower rates of acute metabolic complications that required hospitalization.
For physicians, empathic relationships with your patients lead to fewer disputes, higher reimbursements, greater patient satisfaction, fewer malpractice lawsuits, and a more rewarding experience treating patients.
Different types of empathy
The importance of empathy in doctoring is evident, but Dr. Hojat says it’s crucial to differentiate between clinical empathy and emotional empathy. One can enhance care, while the other, when overused, may lead to physician burnout.
In fact, he says, clinical empathy and emotional empathy have different consequences in a medical setting.
“The relationship between clinical empathy and clinical outcomes is linear, meaning that more empathic engagement leads to more positive clinical outcomes,” says Dr. Hojat. “However, the relationship between emotional empathy and clinical outcomes is curvilinear, or an inverted U shape, similar to the association between anxiety and performance, meaning that limited emotional empathy or limited sympathetic engagement could be helpful, but its overabundance can hamper clinical relationships and objective clinical decision-making.”
The takeaway is that when physicians don’t regulate their emotional empathy, it becomes an obstacle to clinical empathy, ultimately detrimental to health care outcomes.
When burnout hinders empathy
Of course, the reverse is also true – burnout can make it harder for physicians to muster up empathy of any kind toward their patients. At least 53% of physicians show one or more symptoms of burnout, such as exhaustion, questioning the point of the work, cynicism, sarcasm, and the need to “vent” about patients or the job, according to Medscape’s ‘I Cry but No One Cares’: Physician Burnout & Depression Report 2023.
Venting about patients can also be called “compassion fatigue,” which is a sign that your ability to empathize with patients is compromised. You can still practice medicine, but you’re not operating anywhere close to your optimum abilities.
“Generally, physicians who are burned out struggle with empathy since it’s exactly what they’re missing for themselves, and [they] often find it difficult to generate,” says Dr. Trubow.
How to manage burnout and boost your empathy
Burnout can happen for various reasons – pressure to cycle through scores of patients, too many bureaucratic tasks, less autonomy, frustration with electronic health record requirements, and too many work hours, according to the Medscape report.
A report in Family Practice Management finds there are two main goals for physicians to tackle when trying to reduce burnout symptoms: Lower your stress levels and improve your ability to recharge your energy accounts.
“For physicians experiencing burnout [and thus, a lack of empathy], the best approach to this situation is to first take a break and evaluate whether there are any structures to put in place to improve the situation; this can often improve a provider’s empathy,” says Dr. Trubow.
For example, physicians can look at ways to alleviate burnout by investing in leadership development, finding flexible work arrangements, reducing technological burdens, and limiting nonclinical activities.
Other strategies that can build up your reserves include connecting with colleagues, gaining a greater sense of control over your work, and having opportunities to grow and excel in your field. This requires not only a personal approach by physicians, but a buy-in at an institutional level as well.
In Medscape’s report, where 65% of physicians say burnout affects their relationships, physicians’ coping methods include exercise, time with family and friends, time alone, sleep, music, and meditation.
“Clinical empathy must be placed in the realm of ‘evidence-based’ medicine,” says Dr. Hojat. “Given our research findings that clinical empathy tends to erode as students progress through medical school, it is important that assessment and enhancement of clinical empathy be integrated into formal educational curriculum of medical schools and postgraduate training programs for professional development of physicians–in-training and –in-practice.”
“Burnout also leads to a large swath of physicians who aren’t as empathetic toward their patients as they could be.”
–Danielle Ofri, “What Doctors Feel: How Emotions Affect the Practice of Medicine”.
A version of this article first appeared on Medscape.com.
Compassion is borne out of a sense of empathy – the ability to understand and share the feelings of others. Studies on empathy show it to be crucial to quality health care and not just for patients.
In one study on empathy ratings among doctors, 87% of the public believe that compassion, or a clear and obvious desire to relieve suffering, is the most critical factor when choosing a doctor. In fact, it eclipses travel time, wait time, and cost on the list of sought-after physician features.
Wendie Trubow, MD, an ob.gyn. in Newton, Mass., with over 25 years of experience in the medical field, says empathy has absolutely helped her be a better physician.
“Patients consistently mention how grateful they are that someone has listened to them and validated them,” she says. “When patients feel heard and validated, they are more likely to communicate openly, and this raises the potential of being able to create treatment plans that they will actually participate in. Ultimately, it enriches patient care.”
Mohammadreza Hojat, PhD, research professor of psychiatry and human behavior at the Asano-Gonnella Center for Research in Medical Education and Health Care at Thomas Jefferson University in Philadelphia, says that empirical research he and colleagues have done on empathy in health profession education and patient care over the past 20 years shows that empathic engagement in patient care is reciprocally beneficial for both clinicians and patients.
For example, Dr. Hojat notes that in one study, diabetic patients treated by empathic physicians (measured by the Jefferson Scale of Empathy) had more control over their disease when measured with laboratory test results such as hemoglobin A1c and LDL-C. In another, patients with diabetes treated by more empathic physicians had significantly lower rates of acute metabolic complications that required hospitalization.
For physicians, empathic relationships with your patients lead to fewer disputes, higher reimbursements, greater patient satisfaction, fewer malpractice lawsuits, and a more rewarding experience treating patients.
Different types of empathy
The importance of empathy in doctoring is evident, but Dr. Hojat says it’s crucial to differentiate between clinical empathy and emotional empathy. One can enhance care, while the other, when overused, may lead to physician burnout.
In fact, he says, clinical empathy and emotional empathy have different consequences in a medical setting.
“The relationship between clinical empathy and clinical outcomes is linear, meaning that more empathic engagement leads to more positive clinical outcomes,” says Dr. Hojat. “However, the relationship between emotional empathy and clinical outcomes is curvilinear, or an inverted U shape, similar to the association between anxiety and performance, meaning that limited emotional empathy or limited sympathetic engagement could be helpful, but its overabundance can hamper clinical relationships and objective clinical decision-making.”
The takeaway is that when physicians don’t regulate their emotional empathy, it becomes an obstacle to clinical empathy, ultimately detrimental to health care outcomes.
When burnout hinders empathy
Of course, the reverse is also true – burnout can make it harder for physicians to muster up empathy of any kind toward their patients. At least 53% of physicians show one or more symptoms of burnout, such as exhaustion, questioning the point of the work, cynicism, sarcasm, and the need to “vent” about patients or the job, according to Medscape’s ‘I Cry but No One Cares’: Physician Burnout & Depression Report 2023.
Venting about patients can also be called “compassion fatigue,” which is a sign that your ability to empathize with patients is compromised. You can still practice medicine, but you’re not operating anywhere close to your optimum abilities.
“Generally, physicians who are burned out struggle with empathy since it’s exactly what they’re missing for themselves, and [they] often find it difficult to generate,” says Dr. Trubow.
How to manage burnout and boost your empathy
Burnout can happen for various reasons – pressure to cycle through scores of patients, too many bureaucratic tasks, less autonomy, frustration with electronic health record requirements, and too many work hours, according to the Medscape report.
A report in Family Practice Management finds there are two main goals for physicians to tackle when trying to reduce burnout symptoms: Lower your stress levels and improve your ability to recharge your energy accounts.
“For physicians experiencing burnout [and thus, a lack of empathy], the best approach to this situation is to first take a break and evaluate whether there are any structures to put in place to improve the situation; this can often improve a provider’s empathy,” says Dr. Trubow.
For example, physicians can look at ways to alleviate burnout by investing in leadership development, finding flexible work arrangements, reducing technological burdens, and limiting nonclinical activities.
Other strategies that can build up your reserves include connecting with colleagues, gaining a greater sense of control over your work, and having opportunities to grow and excel in your field. This requires not only a personal approach by physicians, but a buy-in at an institutional level as well.
In Medscape’s report, where 65% of physicians say burnout affects their relationships, physicians’ coping methods include exercise, time with family and friends, time alone, sleep, music, and meditation.
“Clinical empathy must be placed in the realm of ‘evidence-based’ medicine,” says Dr. Hojat. “Given our research findings that clinical empathy tends to erode as students progress through medical school, it is important that assessment and enhancement of clinical empathy be integrated into formal educational curriculum of medical schools and postgraduate training programs for professional development of physicians–in-training and –in-practice.”
“Burnout also leads to a large swath of physicians who aren’t as empathetic toward their patients as they could be.”
–Danielle Ofri, “What Doctors Feel: How Emotions Affect the Practice of Medicine”.
A version of this article first appeared on Medscape.com.
Childhood trauma tied to increased Parkinson’s disease severity
new research shows.
Results of the first study to evaluate the relationship between childhood trauma and PD investigators found that the relationship appears to be dose dependent. Patients with PD who reported more than one ACE all experienced a statistically significant decrease in QOL, and for each additional ACE, there was significant worsening of motor symptoms.
This study supports a recent-call to-action paper in JAMA Neurology encouraging adoption of “trauma-informed neurology,” study investigator Indu Subramanian, MD, clinical professor, department of neurology, University of California, Los Angeles, said in an interview.
“We need to start asking about ACEs in everyone. It should be part of our medical intake,” said Dr. Subramanian, who is also the director of the Southwest Parkinson’s Disease Research, Education, and Clinical Center, West Los Angeles Veterans Affairs Medical Center.
The study was published online in Neurology: Clinical Practice.
Hard on the mind and body
A robust body of literature has clearly established a connection between ACEs, which include physical and emotional abuse, neglect, and household dysfunction, and negative physical health outcomes across the lifespan. These include stroke, dementia, diabetes, cancer, cardiovascular disease, autoimmune disorders, hypertension, and premature death as well as psychosocial health outcomes such as anxiety, depression, substance use, and suicide.
However, until now, the effects of childhood trauma have not been evaluated in a PD population.
As part of the MVP study, 712 adults with PD responded to an online survey asking about childhood trauma.
As anticipated, patients with the least reported childhood trauma reported the highest current QOL and lowest patient-reported motor and nonmotor symptom burden compared with peers with higher reported childhood trauma, the researchers reported.
PD symptom burden increased and QOL decreased as the number of ACEs increased.
Patients with ACE scores of 4 or higher reported greater PD symptom severity for 45% of the variables assessed, including apathy, muscle pain, daytime sleepiness, restless leg syndrome, depression, fatigue, comprehension, and anxiety (P < .05), compared with peers with trauma scores of 0.
Limitations of the study included the cross-sectional nature, which prevents making any causal determinations. Also, the ACE questionnaire, because it is self-reported and a retrospective collection of data, introduces the risk for recall bias. In addition, 65% of respondents were women, and racial and ethnic minority groups were not well represented.
Looking ahead, Dr. Subramanian and coauthors believe future research should “attempt to include more diverse populations, attempt improve the response rate of these sensitive questions and, most importantly, determine whether the adverse outcomes associated with childhood trauma can be mitigated with lifestyle modification, psychosocial support, and intervention in adulthood.”
“As a trauma-informed approach, something sorely lacking yet needed in the field of movement disorders, clinicians can proactively screen for ACEs while being mindful to avoid retraumatization,” they suggested. “They can begin to identify how ACEs may physiologically contribute to PD symptom and focus on targeting appropriate interventions that may improve outcomes.”
Life experiences matter
In a comment, Michael S. Okun, MD, medical advisor, Parkinson’s Foundation, and director of the Norman Fixel Institute for Neurological Diseases, University of Florida Health, Gainesville, said that “the idea that childhood trauma could be associated with a mild increase in severity of Parkinson’s symptoms such as apathy, pain, sleepiness and depression is fascinating.”
“We should however temper our enthusiasm for the results of this study because they were obtained through a direct patient survey, and not collected from large well characterized medical database,” Dr. Okun said.
He added” “If the data on childhood trauma and Parkinson’s can be replicated, we must ask why this could be?
“For Parkinson clinicians this as a reminder of how important obtaining a complete life history can be when strategizing on a plan to reduce motor and nonmotor Parkinson symptoms. Life experiences matter and can impact symptoms,” Dr. Okun said.
The MVP study was initiated with support of the National Center for Complementary and Integrative Health. The ongoing data collection has been supported by a donation from Sondra and Bill Fondren. Dr. Subramanian and Dr. Okun disclosed no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
new research shows.
Results of the first study to evaluate the relationship between childhood trauma and PD investigators found that the relationship appears to be dose dependent. Patients with PD who reported more than one ACE all experienced a statistically significant decrease in QOL, and for each additional ACE, there was significant worsening of motor symptoms.
This study supports a recent-call to-action paper in JAMA Neurology encouraging adoption of “trauma-informed neurology,” study investigator Indu Subramanian, MD, clinical professor, department of neurology, University of California, Los Angeles, said in an interview.
“We need to start asking about ACEs in everyone. It should be part of our medical intake,” said Dr. Subramanian, who is also the director of the Southwest Parkinson’s Disease Research, Education, and Clinical Center, West Los Angeles Veterans Affairs Medical Center.
The study was published online in Neurology: Clinical Practice.
Hard on the mind and body
A robust body of literature has clearly established a connection between ACEs, which include physical and emotional abuse, neglect, and household dysfunction, and negative physical health outcomes across the lifespan. These include stroke, dementia, diabetes, cancer, cardiovascular disease, autoimmune disorders, hypertension, and premature death as well as psychosocial health outcomes such as anxiety, depression, substance use, and suicide.
However, until now, the effects of childhood trauma have not been evaluated in a PD population.
As part of the MVP study, 712 adults with PD responded to an online survey asking about childhood trauma.
As anticipated, patients with the least reported childhood trauma reported the highest current QOL and lowest patient-reported motor and nonmotor symptom burden compared with peers with higher reported childhood trauma, the researchers reported.
PD symptom burden increased and QOL decreased as the number of ACEs increased.
Patients with ACE scores of 4 or higher reported greater PD symptom severity for 45% of the variables assessed, including apathy, muscle pain, daytime sleepiness, restless leg syndrome, depression, fatigue, comprehension, and anxiety (P < .05), compared with peers with trauma scores of 0.
Limitations of the study included the cross-sectional nature, which prevents making any causal determinations. Also, the ACE questionnaire, because it is self-reported and a retrospective collection of data, introduces the risk for recall bias. In addition, 65% of respondents were women, and racial and ethnic minority groups were not well represented.
Looking ahead, Dr. Subramanian and coauthors believe future research should “attempt to include more diverse populations, attempt improve the response rate of these sensitive questions and, most importantly, determine whether the adverse outcomes associated with childhood trauma can be mitigated with lifestyle modification, psychosocial support, and intervention in adulthood.”
“As a trauma-informed approach, something sorely lacking yet needed in the field of movement disorders, clinicians can proactively screen for ACEs while being mindful to avoid retraumatization,” they suggested. “They can begin to identify how ACEs may physiologically contribute to PD symptom and focus on targeting appropriate interventions that may improve outcomes.”
Life experiences matter
In a comment, Michael S. Okun, MD, medical advisor, Parkinson’s Foundation, and director of the Norman Fixel Institute for Neurological Diseases, University of Florida Health, Gainesville, said that “the idea that childhood trauma could be associated with a mild increase in severity of Parkinson’s symptoms such as apathy, pain, sleepiness and depression is fascinating.”
“We should however temper our enthusiasm for the results of this study because they were obtained through a direct patient survey, and not collected from large well characterized medical database,” Dr. Okun said.
He added” “If the data on childhood trauma and Parkinson’s can be replicated, we must ask why this could be?
“For Parkinson clinicians this as a reminder of how important obtaining a complete life history can be when strategizing on a plan to reduce motor and nonmotor Parkinson symptoms. Life experiences matter and can impact symptoms,” Dr. Okun said.
The MVP study was initiated with support of the National Center for Complementary and Integrative Health. The ongoing data collection has been supported by a donation from Sondra and Bill Fondren. Dr. Subramanian and Dr. Okun disclosed no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
new research shows.
Results of the first study to evaluate the relationship between childhood trauma and PD investigators found that the relationship appears to be dose dependent. Patients with PD who reported more than one ACE all experienced a statistically significant decrease in QOL, and for each additional ACE, there was significant worsening of motor symptoms.
This study supports a recent-call to-action paper in JAMA Neurology encouraging adoption of “trauma-informed neurology,” study investigator Indu Subramanian, MD, clinical professor, department of neurology, University of California, Los Angeles, said in an interview.
“We need to start asking about ACEs in everyone. It should be part of our medical intake,” said Dr. Subramanian, who is also the director of the Southwest Parkinson’s Disease Research, Education, and Clinical Center, West Los Angeles Veterans Affairs Medical Center.
The study was published online in Neurology: Clinical Practice.
Hard on the mind and body
A robust body of literature has clearly established a connection between ACEs, which include physical and emotional abuse, neglect, and household dysfunction, and negative physical health outcomes across the lifespan. These include stroke, dementia, diabetes, cancer, cardiovascular disease, autoimmune disorders, hypertension, and premature death as well as psychosocial health outcomes such as anxiety, depression, substance use, and suicide.
However, until now, the effects of childhood trauma have not been evaluated in a PD population.
As part of the MVP study, 712 adults with PD responded to an online survey asking about childhood trauma.
As anticipated, patients with the least reported childhood trauma reported the highest current QOL and lowest patient-reported motor and nonmotor symptom burden compared with peers with higher reported childhood trauma, the researchers reported.
PD symptom burden increased and QOL decreased as the number of ACEs increased.
Patients with ACE scores of 4 or higher reported greater PD symptom severity for 45% of the variables assessed, including apathy, muscle pain, daytime sleepiness, restless leg syndrome, depression, fatigue, comprehension, and anxiety (P < .05), compared with peers with trauma scores of 0.
Limitations of the study included the cross-sectional nature, which prevents making any causal determinations. Also, the ACE questionnaire, because it is self-reported and a retrospective collection of data, introduces the risk for recall bias. In addition, 65% of respondents were women, and racial and ethnic minority groups were not well represented.
Looking ahead, Dr. Subramanian and coauthors believe future research should “attempt to include more diverse populations, attempt improve the response rate of these sensitive questions and, most importantly, determine whether the adverse outcomes associated with childhood trauma can be mitigated with lifestyle modification, psychosocial support, and intervention in adulthood.”
“As a trauma-informed approach, something sorely lacking yet needed in the field of movement disorders, clinicians can proactively screen for ACEs while being mindful to avoid retraumatization,” they suggested. “They can begin to identify how ACEs may physiologically contribute to PD symptom and focus on targeting appropriate interventions that may improve outcomes.”
Life experiences matter
In a comment, Michael S. Okun, MD, medical advisor, Parkinson’s Foundation, and director of the Norman Fixel Institute for Neurological Diseases, University of Florida Health, Gainesville, said that “the idea that childhood trauma could be associated with a mild increase in severity of Parkinson’s symptoms such as apathy, pain, sleepiness and depression is fascinating.”
“We should however temper our enthusiasm for the results of this study because they were obtained through a direct patient survey, and not collected from large well characterized medical database,” Dr. Okun said.
He added” “If the data on childhood trauma and Parkinson’s can be replicated, we must ask why this could be?
“For Parkinson clinicians this as a reminder of how important obtaining a complete life history can be when strategizing on a plan to reduce motor and nonmotor Parkinson symptoms. Life experiences matter and can impact symptoms,” Dr. Okun said.
The MVP study was initiated with support of the National Center for Complementary and Integrative Health. The ongoing data collection has been supported by a donation from Sondra and Bill Fondren. Dr. Subramanian and Dr. Okun disclosed no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY: CLINICAL PRACTICE
Does CRC risk in IBD extend to close family members?
new research suggests.
In a large Swedish study, a history of IBD among first-degree relatives was not associated with an increased risk of CRC, even when considering various characteristics of IBD and CRC history.
The findings suggest that extra screening for CRC may not be needed for children, siblings, or parents of those with IBD, say the study authors, led by Kai Wang, MD, PhD, with Harvard School of Public Health, Boston. The findings strengthen the theory that it’s inflammation or atypism of the colon of people with IBD that confers the increased CRC risk.
“There is nothing in this study that changes our existing practice,” said Ashwin Ananthakrishnan, MD, MPH, with Massachusetts General Hospital and Harvard Medical School in Boston, who was not involved in the research. “It is already the thought that inflammation in IBD increases risk of cancer,” which would not increase CRC risk among family members.
The study appeared in the International Journal of Cancer.
Patients with IBD are known to be at increased risk for CRC. However, the association between family history of IBD and CRC risk remains less clear. Current CRC screening recommendations are the same for patients who have family members with IBD and for those who do not.
The Swedish nationwide case-control study included 69,659 individuals with CRC, of whom 1,599 (2.3%) had IBD, and 343,032 matched control persons who did not have CRC, of whom 1,477 (0.4%) had IBD.
Overall, 2.2% of CRC case patients and control patients had at least one first-degree relative who had a history of IBD.
After adjusting for family history of CRC, the authors did not find an increase in risk for CRC among first-degree relatives of people with IBD (odds ratio, 0.96; 95% confidence interval, 0.91-1.02).
The null association was consistently observed regardless of IBD subtype (Crohn’s disease or ulcerative colitis), the number of first-degree relatives with IBD, age at first IBD diagnosis, maximum location or extent of IBD, or type of relative (parent, sibling, or offspring). The null association remained for early-onset CRC diagnosed before age 50.
Overall, these findings suggest that IBD and CRC may not have substantial familial clustering or shared genetic susceptibility and provide “robust evidence that a family history of IBD did not increase the risk of CRC, supporting use of the same routine CRC screening strategy in offspring, siblings, and parents of IBD patients as in the general population,” Dr. Wang and colleagues conclude.
This “well-done” study is one of the largest to date to evaluate first-degree relatives of IBD patients and their risk of CRC, said Shannon Chang, MD, with NYU Langone Health Inflammatory Bowel Disease Center, who wasn’t involved in the research.
The findings are reassuring, as the authors assessed several factors and found that family members of patients with IBD are not at higher risk for CRC, compared with the general population, Dr. Chang added.
Support for the study was provided by the National Institutes of Health, the American Cancer Society, ALF funding, the Swedish Research Council, and the Swedish Cancer Foundation. Dr. Wang, Dr. Chang, and Dr. Ananthakrishnan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In a large Swedish study, a history of IBD among first-degree relatives was not associated with an increased risk of CRC, even when considering various characteristics of IBD and CRC history.
The findings suggest that extra screening for CRC may not be needed for children, siblings, or parents of those with IBD, say the study authors, led by Kai Wang, MD, PhD, with Harvard School of Public Health, Boston. The findings strengthen the theory that it’s inflammation or atypism of the colon of people with IBD that confers the increased CRC risk.
“There is nothing in this study that changes our existing practice,” said Ashwin Ananthakrishnan, MD, MPH, with Massachusetts General Hospital and Harvard Medical School in Boston, who was not involved in the research. “It is already the thought that inflammation in IBD increases risk of cancer,” which would not increase CRC risk among family members.
The study appeared in the International Journal of Cancer.
Patients with IBD are known to be at increased risk for CRC. However, the association between family history of IBD and CRC risk remains less clear. Current CRC screening recommendations are the same for patients who have family members with IBD and for those who do not.
The Swedish nationwide case-control study included 69,659 individuals with CRC, of whom 1,599 (2.3%) had IBD, and 343,032 matched control persons who did not have CRC, of whom 1,477 (0.4%) had IBD.
Overall, 2.2% of CRC case patients and control patients had at least one first-degree relative who had a history of IBD.
After adjusting for family history of CRC, the authors did not find an increase in risk for CRC among first-degree relatives of people with IBD (odds ratio, 0.96; 95% confidence interval, 0.91-1.02).
The null association was consistently observed regardless of IBD subtype (Crohn’s disease or ulcerative colitis), the number of first-degree relatives with IBD, age at first IBD diagnosis, maximum location or extent of IBD, or type of relative (parent, sibling, or offspring). The null association remained for early-onset CRC diagnosed before age 50.
Overall, these findings suggest that IBD and CRC may not have substantial familial clustering or shared genetic susceptibility and provide “robust evidence that a family history of IBD did not increase the risk of CRC, supporting use of the same routine CRC screening strategy in offspring, siblings, and parents of IBD patients as in the general population,” Dr. Wang and colleagues conclude.
This “well-done” study is one of the largest to date to evaluate first-degree relatives of IBD patients and their risk of CRC, said Shannon Chang, MD, with NYU Langone Health Inflammatory Bowel Disease Center, who wasn’t involved in the research.
The findings are reassuring, as the authors assessed several factors and found that family members of patients with IBD are not at higher risk for CRC, compared with the general population, Dr. Chang added.
Support for the study was provided by the National Institutes of Health, the American Cancer Society, ALF funding, the Swedish Research Council, and the Swedish Cancer Foundation. Dr. Wang, Dr. Chang, and Dr. Ananthakrishnan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In a large Swedish study, a history of IBD among first-degree relatives was not associated with an increased risk of CRC, even when considering various characteristics of IBD and CRC history.
The findings suggest that extra screening for CRC may not be needed for children, siblings, or parents of those with IBD, say the study authors, led by Kai Wang, MD, PhD, with Harvard School of Public Health, Boston. The findings strengthen the theory that it’s inflammation or atypism of the colon of people with IBD that confers the increased CRC risk.
“There is nothing in this study that changes our existing practice,” said Ashwin Ananthakrishnan, MD, MPH, with Massachusetts General Hospital and Harvard Medical School in Boston, who was not involved in the research. “It is already the thought that inflammation in IBD increases risk of cancer,” which would not increase CRC risk among family members.
The study appeared in the International Journal of Cancer.
Patients with IBD are known to be at increased risk for CRC. However, the association between family history of IBD and CRC risk remains less clear. Current CRC screening recommendations are the same for patients who have family members with IBD and for those who do not.
The Swedish nationwide case-control study included 69,659 individuals with CRC, of whom 1,599 (2.3%) had IBD, and 343,032 matched control persons who did not have CRC, of whom 1,477 (0.4%) had IBD.
Overall, 2.2% of CRC case patients and control patients had at least one first-degree relative who had a history of IBD.
After adjusting for family history of CRC, the authors did not find an increase in risk for CRC among first-degree relatives of people with IBD (odds ratio, 0.96; 95% confidence interval, 0.91-1.02).
The null association was consistently observed regardless of IBD subtype (Crohn’s disease or ulcerative colitis), the number of first-degree relatives with IBD, age at first IBD diagnosis, maximum location or extent of IBD, or type of relative (parent, sibling, or offspring). The null association remained for early-onset CRC diagnosed before age 50.
Overall, these findings suggest that IBD and CRC may not have substantial familial clustering or shared genetic susceptibility and provide “robust evidence that a family history of IBD did not increase the risk of CRC, supporting use of the same routine CRC screening strategy in offspring, siblings, and parents of IBD patients as in the general population,” Dr. Wang and colleagues conclude.
This “well-done” study is one of the largest to date to evaluate first-degree relatives of IBD patients and their risk of CRC, said Shannon Chang, MD, with NYU Langone Health Inflammatory Bowel Disease Center, who wasn’t involved in the research.
The findings are reassuring, as the authors assessed several factors and found that family members of patients with IBD are not at higher risk for CRC, compared with the general population, Dr. Chang added.
Support for the study was provided by the National Institutes of Health, the American Cancer Society, ALF funding, the Swedish Research Council, and the Swedish Cancer Foundation. Dr. Wang, Dr. Chang, and Dr. Ananthakrishnan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE INTERNATIONAL JOURNAL OF CANCER
New coding policies to prevent surprise billing for CRC screening
The Departments of Labor, Health & Human Services, and the Treasury issued guidance in 2022 that plans and insurers “must cover and may not impose cost sharing with respect to a colonoscopy conducted after a positive non-invasive stool-based screening test” for plan or policy years1 beginning on or after May 31, 2022, and, further, “may not impose cost-sharing with respect to a polyp removal during a colonoscopy performed as a screening procedure.”2 So why are so many patients still being charged fees for these screening services? In many cases, the answer comes down to missing code modifiers.
Commercial insurers want you to use modifier 33
AGA spoke to Elevance (formerly Anthem), Cigna, Aetna, and Blue Cross Blue Shield Association about how physicians should report colorectal cancer screening procedures and tests. They said using the 33 modifier (preventive service) is essential for their systems to trigger the screening benefits for beneficiaries. Without the 33 modifier, the claim will be processed as a diagnostic service, and coinsurance may apply.
According to the CPT manual, modifier 33 should be used “when the primary purpose of the service is the delivery of an evidence-based service in accordance with a U.S. Preventive Services Task Force A or B rating in effect and other preventive services identified in preventive mandates (legislative or regulatory) ...” Use modifier 33 with colonoscopies that start out as screening procedures and with colonoscopies following a positive non-invasive stool-based test, like fecal immunochemical test (FIT) or Cologuard™ multi-target stool DNA test.
It is important to note that modifier 33 won’t ensure all screening colonoscopy claims are paid, because not all commercial plans are required to cover 100 percent of the costs of CRC screening tests and procedures. For example, employer-sponsored insurance plans and legacy plans can choose not to adopt the expanded CRC benefits. Patients who are covered under these plans may not be aware that their CRC test or procedure will not be fully covered. These patients may still receive a “surprise” bill if their screening colonoscopy requires removal of polyps or if they have a colonoscopy following a positive non-invasive CRC test.
Medicare wants you to use modifiers PT and KX, but not together
CMS uses Healthcare Common Procedural Coding System (HCPCS) codes to differentiate between screening and diagnostic colonoscopies to apply screening benefits. For Medicare beneficiaries who choose colonoscopy as their CRC screening, use HCPCS code G0105 (Colorectal cancer screening; colonoscopy on individual at high risk) or G0121 (Colorectal cancer screening; colonoscopy on individual not meeting the criteria for high risk) for screening colonoscopies as appropriate. No modifier is necessary with G0105 or G0121.
Effective for claims with dates of service on or after 1/1/2023, use the appropriate HCPCS codes G0105 or G0121 with the KX modifier for colonoscopy following a positive result for any of the following non-invasive stool-based CRC screening tests:
• Screening guaiac-based fecal occult blood test (gFOBT) (CPT 82270)
• Screening immunoassay-based fecal occult blood test (iFOBT) (HCPCS G0328)
• Cologuard™ – multi-target stool DNA (sDNA) test (CPT 81528)
According to the guidance in the CMS Manual System, if modifier KX is not added to G0105 or G0121 for colonoscopy following a positive non-invasive stool-based test, Medicare will return the screening colonoscopy claim as “unprocessable.”3 If this happens, add modifier KX and resubmit the claim.
If polyps are removed during a screening colonoscopy, use the appropriate CPT code (45380, 45384, 45385, 45388) and add modifier PT (colorectal cancer screening test; converted to diagnostic test or other procedure) to each CPT code for Medicare. However, it is important to note that if a polyp is removed during a screening colonoscopy, the Medicare beneficiary is responsible for 15% of the cost from 2023 to 2026. This falls to 10% of the cost from 2027 to 2029, and by 2030 it will be covered 100% by Medicare. Some Medicare beneficiaries are not aware that Medicare has not fully eliminated the coinsurance responsibility yet.

What to do if your patient gets an unexpected bill
If your patient gets an unexpected bill and you coded the procedure correctly with the correct modifier, direct them to the AGA GI Patient Care Center’s “Colorectal cancer screening: what to expect when paying” resource for help with next steps.4
The authors have no conflicts to declare.
References
1. U.S. Department of Labor (2022, Jan. 10) FAQs About Affordable Care Act Implementation Part 51. https://www.dol.gov/sites/dolgov/files/EBSA/about-ebsa/our-activities/resource-center/faqs/aca-part-51.pdf
2. Centers for Medicare and Medicaid Services (n.d.) Affordable Care Act Implementation FAQs - Set 12. https://www.cms.gov/CCIIO/Resources/Fact-Sheets-and-FAQs/aca_implementation_faqs12.
3. Centers for Medicare and Medicaid Services (2023, Jan. 27) CMS Manual System Pub 100-03 Medicare National Coverage Determinations Transmittal 11824. https://www.cms.gov/files/document/r11824ncd.pdf.
4. American Gastroenterological Association (2023, Feb. 21) AGA GI Patient Center Colorectal Cancer Screening: What to expect when paying. https://patient.gastro.org/paying-for-your-colonoscopy/.
The Departments of Labor, Health & Human Services, and the Treasury issued guidance in 2022 that plans and insurers “must cover and may not impose cost sharing with respect to a colonoscopy conducted after a positive non-invasive stool-based screening test” for plan or policy years1 beginning on or after May 31, 2022, and, further, “may not impose cost-sharing with respect to a polyp removal during a colonoscopy performed as a screening procedure.”2 So why are so many patients still being charged fees for these screening services? In many cases, the answer comes down to missing code modifiers.
Commercial insurers want you to use modifier 33
AGA spoke to Elevance (formerly Anthem), Cigna, Aetna, and Blue Cross Blue Shield Association about how physicians should report colorectal cancer screening procedures and tests. They said using the 33 modifier (preventive service) is essential for their systems to trigger the screening benefits for beneficiaries. Without the 33 modifier, the claim will be processed as a diagnostic service, and coinsurance may apply.
According to the CPT manual, modifier 33 should be used “when the primary purpose of the service is the delivery of an evidence-based service in accordance with a U.S. Preventive Services Task Force A or B rating in effect and other preventive services identified in preventive mandates (legislative or regulatory) ...” Use modifier 33 with colonoscopies that start out as screening procedures and with colonoscopies following a positive non-invasive stool-based test, like fecal immunochemical test (FIT) or Cologuard™ multi-target stool DNA test.
It is important to note that modifier 33 won’t ensure all screening colonoscopy claims are paid, because not all commercial plans are required to cover 100 percent of the costs of CRC screening tests and procedures. For example, employer-sponsored insurance plans and legacy plans can choose not to adopt the expanded CRC benefits. Patients who are covered under these plans may not be aware that their CRC test or procedure will not be fully covered. These patients may still receive a “surprise” bill if their screening colonoscopy requires removal of polyps or if they have a colonoscopy following a positive non-invasive CRC test.
Medicare wants you to use modifiers PT and KX, but not together
CMS uses Healthcare Common Procedural Coding System (HCPCS) codes to differentiate between screening and diagnostic colonoscopies to apply screening benefits. For Medicare beneficiaries who choose colonoscopy as their CRC screening, use HCPCS code G0105 (Colorectal cancer screening; colonoscopy on individual at high risk) or G0121 (Colorectal cancer screening; colonoscopy on individual not meeting the criteria for high risk) for screening colonoscopies as appropriate. No modifier is necessary with G0105 or G0121.
Effective for claims with dates of service on or after 1/1/2023, use the appropriate HCPCS codes G0105 or G0121 with the KX modifier for colonoscopy following a positive result for any of the following non-invasive stool-based CRC screening tests:
• Screening guaiac-based fecal occult blood test (gFOBT) (CPT 82270)
• Screening immunoassay-based fecal occult blood test (iFOBT) (HCPCS G0328)
• Cologuard™ – multi-target stool DNA (sDNA) test (CPT 81528)
According to the guidance in the CMS Manual System, if modifier KX is not added to G0105 or G0121 for colonoscopy following a positive non-invasive stool-based test, Medicare will return the screening colonoscopy claim as “unprocessable.”3 If this happens, add modifier KX and resubmit the claim.
If polyps are removed during a screening colonoscopy, use the appropriate CPT code (45380, 45384, 45385, 45388) and add modifier PT (colorectal cancer screening test; converted to diagnostic test or other procedure) to each CPT code for Medicare. However, it is important to note that if a polyp is removed during a screening colonoscopy, the Medicare beneficiary is responsible for 15% of the cost from 2023 to 2026. This falls to 10% of the cost from 2027 to 2029, and by 2030 it will be covered 100% by Medicare. Some Medicare beneficiaries are not aware that Medicare has not fully eliminated the coinsurance responsibility yet.

What to do if your patient gets an unexpected bill
If your patient gets an unexpected bill and you coded the procedure correctly with the correct modifier, direct them to the AGA GI Patient Care Center’s “Colorectal cancer screening: what to expect when paying” resource for help with next steps.4
The authors have no conflicts to declare.
References
1. U.S. Department of Labor (2022, Jan. 10) FAQs About Affordable Care Act Implementation Part 51. https://www.dol.gov/sites/dolgov/files/EBSA/about-ebsa/our-activities/resource-center/faqs/aca-part-51.pdf
2. Centers for Medicare and Medicaid Services (n.d.) Affordable Care Act Implementation FAQs - Set 12. https://www.cms.gov/CCIIO/Resources/Fact-Sheets-and-FAQs/aca_implementation_faqs12.
3. Centers for Medicare and Medicaid Services (2023, Jan. 27) CMS Manual System Pub 100-03 Medicare National Coverage Determinations Transmittal 11824. https://www.cms.gov/files/document/r11824ncd.pdf.
4. American Gastroenterological Association (2023, Feb. 21) AGA GI Patient Center Colorectal Cancer Screening: What to expect when paying. https://patient.gastro.org/paying-for-your-colonoscopy/.
The Departments of Labor, Health & Human Services, and the Treasury issued guidance in 2022 that plans and insurers “must cover and may not impose cost sharing with respect to a colonoscopy conducted after a positive non-invasive stool-based screening test” for plan or policy years1 beginning on or after May 31, 2022, and, further, “may not impose cost-sharing with respect to a polyp removal during a colonoscopy performed as a screening procedure.”2 So why are so many patients still being charged fees for these screening services? In many cases, the answer comes down to missing code modifiers.
Commercial insurers want you to use modifier 33
AGA spoke to Elevance (formerly Anthem), Cigna, Aetna, and Blue Cross Blue Shield Association about how physicians should report colorectal cancer screening procedures and tests. They said using the 33 modifier (preventive service) is essential for their systems to trigger the screening benefits for beneficiaries. Without the 33 modifier, the claim will be processed as a diagnostic service, and coinsurance may apply.
According to the CPT manual, modifier 33 should be used “when the primary purpose of the service is the delivery of an evidence-based service in accordance with a U.S. Preventive Services Task Force A or B rating in effect and other preventive services identified in preventive mandates (legislative or regulatory) ...” Use modifier 33 with colonoscopies that start out as screening procedures and with colonoscopies following a positive non-invasive stool-based test, like fecal immunochemical test (FIT) or Cologuard™ multi-target stool DNA test.
It is important to note that modifier 33 won’t ensure all screening colonoscopy claims are paid, because not all commercial plans are required to cover 100 percent of the costs of CRC screening tests and procedures. For example, employer-sponsored insurance plans and legacy plans can choose not to adopt the expanded CRC benefits. Patients who are covered under these plans may not be aware that their CRC test or procedure will not be fully covered. These patients may still receive a “surprise” bill if their screening colonoscopy requires removal of polyps or if they have a colonoscopy following a positive non-invasive CRC test.
Medicare wants you to use modifiers PT and KX, but not together
CMS uses Healthcare Common Procedural Coding System (HCPCS) codes to differentiate between screening and diagnostic colonoscopies to apply screening benefits. For Medicare beneficiaries who choose colonoscopy as their CRC screening, use HCPCS code G0105 (Colorectal cancer screening; colonoscopy on individual at high risk) or G0121 (Colorectal cancer screening; colonoscopy on individual not meeting the criteria for high risk) for screening colonoscopies as appropriate. No modifier is necessary with G0105 or G0121.
Effective for claims with dates of service on or after 1/1/2023, use the appropriate HCPCS codes G0105 or G0121 with the KX modifier for colonoscopy following a positive result for any of the following non-invasive stool-based CRC screening tests:
• Screening guaiac-based fecal occult blood test (gFOBT) (CPT 82270)
• Screening immunoassay-based fecal occult blood test (iFOBT) (HCPCS G0328)
• Cologuard™ – multi-target stool DNA (sDNA) test (CPT 81528)
According to the guidance in the CMS Manual System, if modifier KX is not added to G0105 or G0121 for colonoscopy following a positive non-invasive stool-based test, Medicare will return the screening colonoscopy claim as “unprocessable.”3 If this happens, add modifier KX and resubmit the claim.
If polyps are removed during a screening colonoscopy, use the appropriate CPT code (45380, 45384, 45385, 45388) and add modifier PT (colorectal cancer screening test; converted to diagnostic test or other procedure) to each CPT code for Medicare. However, it is important to note that if a polyp is removed during a screening colonoscopy, the Medicare beneficiary is responsible for 15% of the cost from 2023 to 2026. This falls to 10% of the cost from 2027 to 2029, and by 2030 it will be covered 100% by Medicare. Some Medicare beneficiaries are not aware that Medicare has not fully eliminated the coinsurance responsibility yet.

What to do if your patient gets an unexpected bill
If your patient gets an unexpected bill and you coded the procedure correctly with the correct modifier, direct them to the AGA GI Patient Care Center’s “Colorectal cancer screening: what to expect when paying” resource for help with next steps.4
The authors have no conflicts to declare.
References
1. U.S. Department of Labor (2022, Jan. 10) FAQs About Affordable Care Act Implementation Part 51. https://www.dol.gov/sites/dolgov/files/EBSA/about-ebsa/our-activities/resource-center/faqs/aca-part-51.pdf
2. Centers for Medicare and Medicaid Services (n.d.) Affordable Care Act Implementation FAQs - Set 12. https://www.cms.gov/CCIIO/Resources/Fact-Sheets-and-FAQs/aca_implementation_faqs12.
3. Centers for Medicare and Medicaid Services (2023, Jan. 27) CMS Manual System Pub 100-03 Medicare National Coverage Determinations Transmittal 11824. https://www.cms.gov/files/document/r11824ncd.pdf.
4. American Gastroenterological Association (2023, Feb. 21) AGA GI Patient Center Colorectal Cancer Screening: What to expect when paying. https://patient.gastro.org/paying-for-your-colonoscopy/.
Epithelioma Cuniculatum (Plantar Verrucous Carcinoma): A Systematic Review of Treatment Options
Verrucous carcinoma (VC) is an uncommon type of well-differentiated squamous cell carcinoma (SCC) that most commonly affects men in the fifth to sixth decades of life. 1 The tumor grows slowly over a decade or more and does not frequently metastasize but has a high propensity for recurrence and local invasion. 2 There are 3 main subtypes of VC classified by anatomic site: oral florid papillomatosis (oral cavity), Buschke-Lowenstein tumor (anogenital region), and epithelioma cuniculatum (EC)(feet). 3 Epithelioma cuniculatum, also known as carcinoma cuniculatum or papillomatosis cutis carcinoides, most commonly presents as a solitary, warty or cauliflowerlike, exophytic mass with keratin-filled sinus tracts and malodorous discharge. 4 Diabetic foot ulcers and chronic inflammatory conditions are predisposing risk factors for EC, and it can result in difficulty walking/immobility, pain, and bleeding depending on anatomic involvement. 5-9
The differential diagnosis for VC includes refractory verruca vulgaris, clavus, SCC, keratoacanthoma, deep fungal or mycobacterial infection, eccrine poroma or porocarcinoma, amelanotic melanoma, and sarcoma.10-13 The slow-growing nature of VC, sampling error of superficial biopsies, and minimal cytological atypia on histologic examination can contribute to delayed diagnosis and appropriate treatment.14 Characteristic histologic features include hyperkeratosis, papillomatosis, marked acanthosis, broad blunt-ended rete ridges with a “bulldozing” architecture, and minimal cytologic atypia and mitoses.5,6 In some cases, pleomorphism and glassy eosinophilic cytoplasmic changes may be more pronounced than that of a common wart though less dramatic than that of conventional SCCs.15 Antigen Ki-67 and tumor protein p53 have been proposed to help differentiate between common plantar verruca, VC, and SCC, but the histologic diagnosis remains challenging, and repeat histopathologic examination often is required.16-19 Following diagnosis, computed tomography or magnetic resonance imaging may be necessary to determine tumor extension and assess for deep tissue and bony involvement.20-22
Treatment of EC is particularly challenging because of the anatomic location and need for margin control while maintaining adequate function, preserving healthy tissue, and providing coverage of defects. Surgical excision of EC is the first-line treatment, most commonly by wide local excision (WLE) or amputation. Mohs micrographic surgery (MMS) also has been utilized. One review found no recurrences in 5 cases of EC treated with MMS.23 As MMS is a tissue-sparing technique, this is a valuable modality for sites of functional importance such as the feet. Herein, we review various reported EC treatment modalities and outcomes, with an emphasis on recurrence rates for WLE and MMS.
METHODS
A systematic literature review of PubMed articles indexed for MEDLINE, as well as databases including the Cochrane Library, Web of Science, and Cumulative Index to Nursing and Allied Health Literature (CINAHL), was performed on January 14, 2020. Two authors (S.S.D. and S.V.C.) independently screened results using the search terms (plantar OR foot) AND (verrucous carcinoma OR epithelioma cuniculatum OR carcinoma cuniculatum). The search terms were chosen according to MeSH subject headings. All articles from the start date of the databases through the search date were screened, and articles pertaining to VC, EC, or carcinoma cuniculatum located on the foot were included. Of these, non–English-language articles were translated and included. Articles reporting VC on a site other than the foot (eg, the oral cavity) or benign verrucous skin lesions were excluded. The reference lists for all articles also were reviewed for additional reports that were absent from the initial search using both included and excluded articles. A full-text review was performed on 221 articles published between 1954 and 2019 per the PRISMA guidelines (Figure).
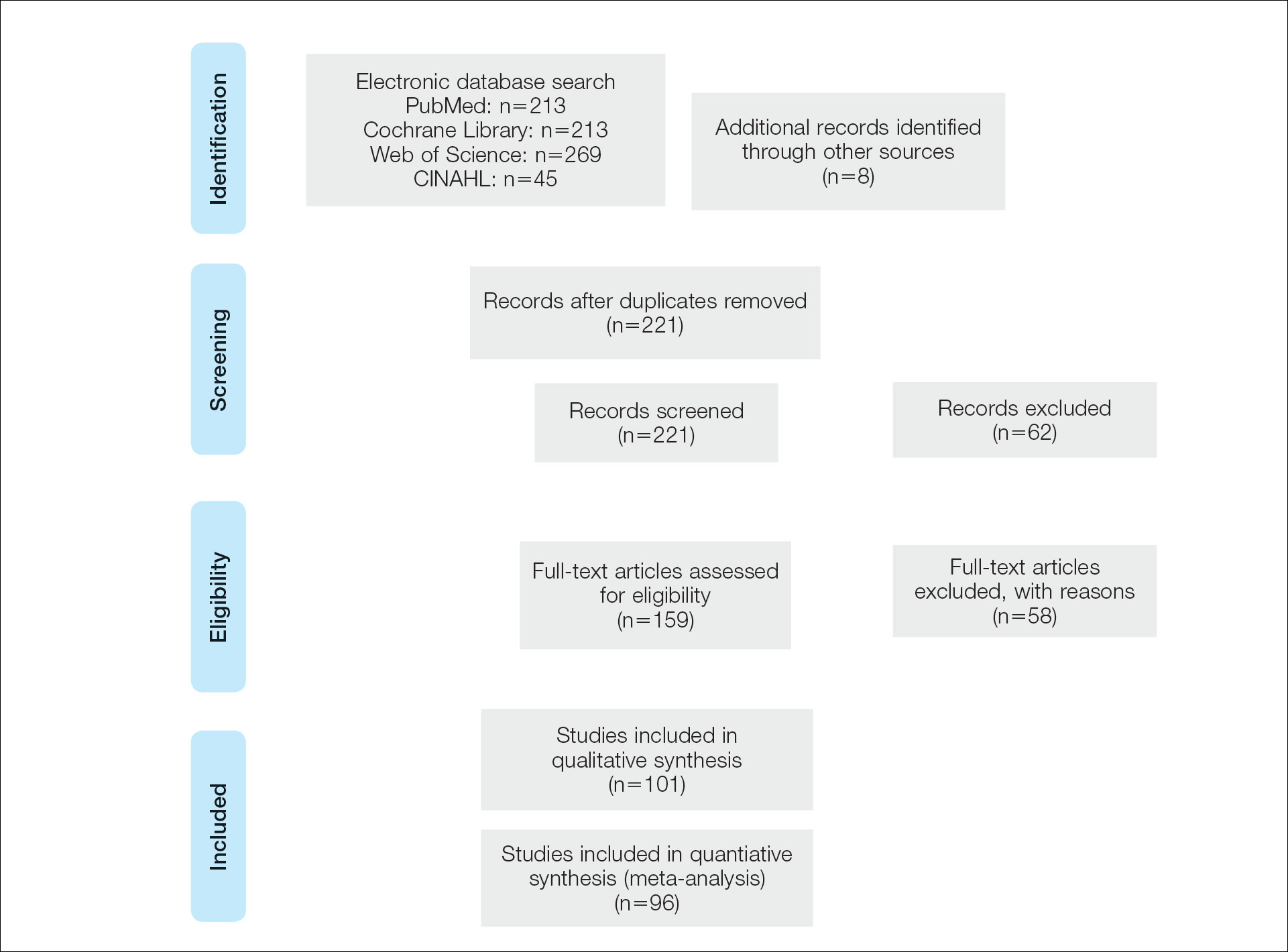
A total of 101 articles were included in the study for qualitative analysis. Nearly all articles identified were case reports, giving an evidence level of 5 by the Centre for Evidence-Based Medicine rating scale. Five articles reported data on multiple patients without individual demographic or clinical details and were excluded from analysis. Of the remaining 96 articles, information about patient characteristics, tumor size, treatment modality, and recurrence were extracted for 115 cases.
RESULTS
Of the 115 cases that were reviewed, 81 (70%) were male and 33 (29%) were female with a male-to-female ratio of 2.4:1. Ages of the patients ranged from 18 to 88 years; the mean and median age was 56 years. Nearly all reported cases of EC affected the plantar surface of one foot, with 4 reports of tumors affecting both feet.24-27 One case affecting both feet reported known exposure to lead arsenate pesticides27; all others were associated with a clinical history of chronic ulcers or warts persisting for several years to decades. Other less common sites of EC included the dorsal foot, interdigital web space, and subungual digit.28-30 The most common location reported was the anterior ball of the foot. Tumors were reported to arise within pre-existing lesions, such as hypertrophic lichen planus or chronic foot wounds associated with diabetes mellitus or leprosy.31-35 Tumor size ranged from 1 to 22 cm with a median of 4.5 cm.
Eight cases were reported to be associated with human papillomavirus; low-risk types 6 and 11 and high-risk types 16 and 18 were found in 6 cases.36-41 Two cases reported association with human papillomavirus type 2.7,42
Metastases to dermal and subdermal lymphatics, regional lymph nodes, and the lungs were reported in 3 cases, repectively.43-45 Of these, one primary tumor had received low-dose irradiation in the form of X-ray therapy.45
Treatment Modalities
The cases of EC that we reviewed included treatment with surgical and systemic therapies as well as other modalities such as acitretin, interferon alfa, topical imiquimod, curettage, debridement, electrodesiccation, and radiation. The Table includes a complete summary of the treatments we analyzed.
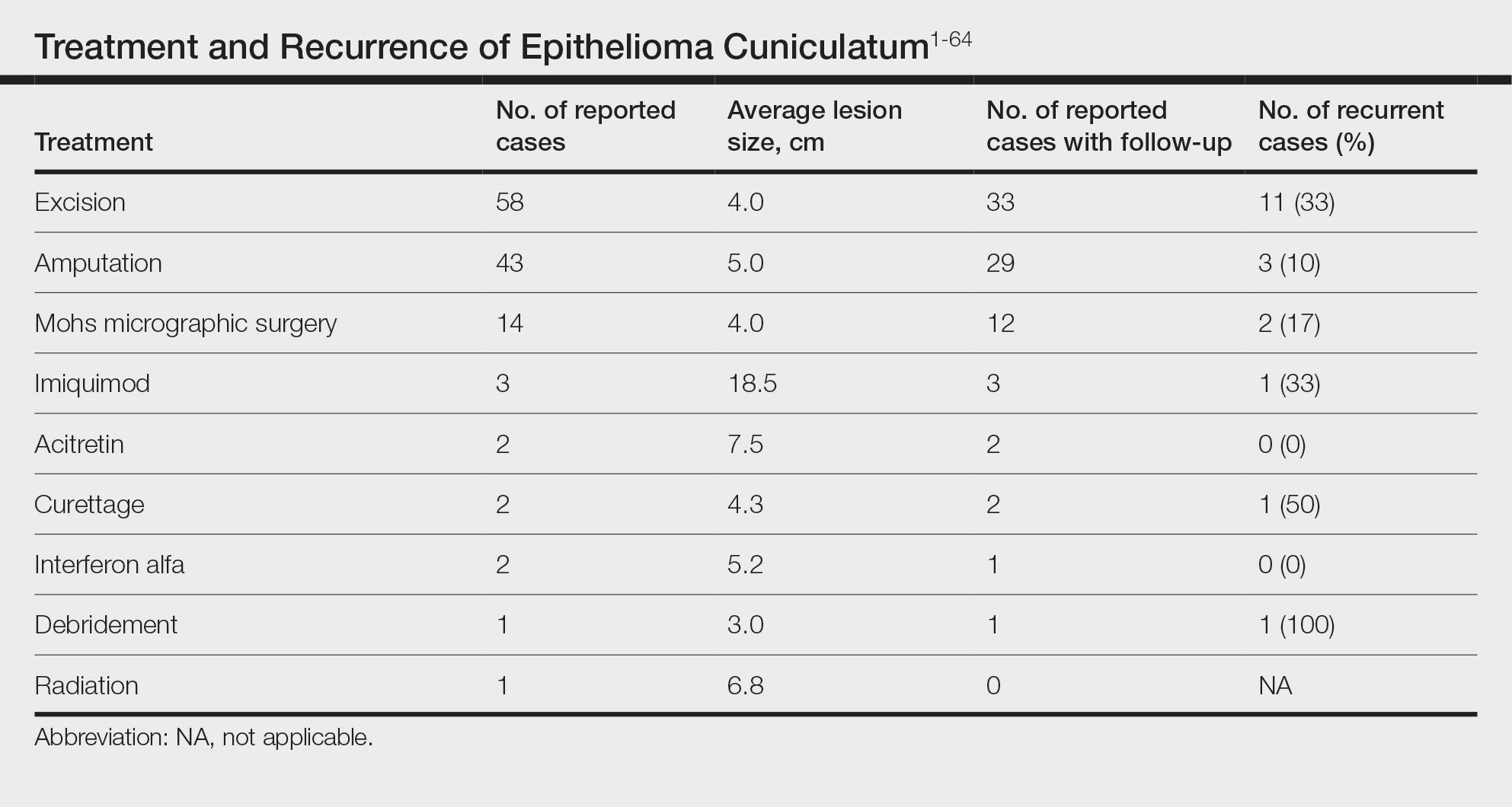
Surgical Therapy—The majority (91% [105/115]) of cases were treated surgically. The most common treatment modality was WLE (50% [58/115]), followed by amputation (37% [43/115]) and MMS (12% [14/115]).
Wide local excision was the most frequently reported treatment, with excision margins of at least 5 mm to 1 cm.48 Incidence of recurrence was reported for 57% (33/58) of cases treated with WLE; of these, the recurrence rate was 33% (11/33). For patients with EC recurrence, the most common secondary treatment was repeat excision with wider margins (1–2 cm) or amputation (5/11).49-52 Few postoperative complications were reported but included pain, infection, and difficulty walking, which were mostly associated with repair modality (eg, split-thickness skin grafts, rotational flaps).53 Amputation was the second most common treatment modality, with a 67% (29/43) incidence of recurrence. Types of amputation included transmetatarsal ray amputation (7/43 [16%]), foot or forefoot amputation (2/43 [5%]), above-the-knee amputation (1/43 [2%]), and below-the-knee amputation (1/43 [2%]). Complications associated with amputation included infection and requirement of prosthetics for ambulation. Split-thickness skin grafts and rotational flaps were the most common surgical repairs performed.52,53
Mohs micrographic surgery was the least frequently reported surgical treatment modality. Both traditional MMS on fresh tissue and “slow Mohs,” with formalin-fixed paraffin embedded tissue examination over several days, were performed for EC with horizontal en face sectioning.54-56 Incidence of recurrence was reported for 86% (12/14) of MMS cases. Of these, recurrence was seen in 17% (2/12) that utilized a flat horizontal processing of tissue sections coupled with saucerlike excisions to enable examination of the entire undersurface and margins. In one case, the patient was treated with MMS with recurrence noted 1 month later; thus, repeat MMS was performed, and the tumor was found to be entwined around the flexor tendon.57 The tendon was removed, and clear margins were obtained. Follow-up 3 years after the second MMS revealed no signs of recurrence.57 In the other case, the patient had a particularly aggressive course with bilateral VC in the setting of diabetic ulcers that was treated with WLE prior to MMS and recurrence still noted after MMS.26 No complications were reported with MMS.
Overall, recurrence was most frequently reported with WLE (11/33 [33%]), followed by MMS (2/12 [17%]) and amputation (3/29 [10%]). When comparing WLE and amputation, the relationship between treatment modality and recurrence was statistically significant using a χ2 test of independence (χ2=4.7; P=.03). However, results were not significant with Yates correction for continuity (χ2=3.4; P=.06). The χ2 test of independence showed no significant association between treatment method and recurrence when comparing WLE with MMS (χ2=1.2; P=.28). Reported follow-up times varied greatly from a few months to 10 years.
Systemic Therapy—Of the total cases, only 2 cases reported treatment with acitretin and 2 utilized interferon alfa.58,59 In one case, treatment of EC with interferon alfa alone required more aggressive therapy (ie, amputation).58 Neither of the 2 cases using acitretin reported recurrence.59,60 Complications of acitretin therapy included cheilitis and transaminitis.60
Other Treatment Modalities—Three cases utilized imiquimod, with 2 cases of imiquimod monotherapy and 1 case of imiquimod in combination with electrodesiccation and WLE.37 One of the cases of EC treated with imiquimod monotherapy recurred and required WLE.61
There were reports of other treatments including curettage alone (2% [2/115]),40,62 debridement alone (1% [1/115]),40 electrodesiccation (1% [1/115]),37 and radiation (1% [1/115]).43 Recurrence was found with curettage alone and debridement alone. Electrodesiccation was reported in conjunction with WLE without recurrence. Radiation was used to treat a case of VC that had metastasized to the lymph nodes; no follow-up was described.43
COMMENT
Epithelioma cuniculatum is an indolent malignancy of the plantar foot that likely is frequently underdiagnosed or misdiagnosed because of location, sampling error, and challenges in histopathologic diagnosis. Once diagnosed, surgical removal with margin control is the first-line therapy for EC. Our review found a number of surgical, systemic, and other treatment modalities that have been used to treat EC, but there remains a lack of evidence to provide clear guidelines as to which therapies are most effective. Current data on the treatment of EC largely are limited to case reports and case series. To date, there are no reports of higher-quality studies or randomized controlled trials to assess the efficacy of various treatment modalities.
Our review found that WLE is the most common treatment modality for EC, followed by amputation and MMS. Three cases43-45 that reported metastasis to lymph nodes also were treated with fine-needle aspiration or biopsy, and it is recommended that sentinel lymph node biopsy be performed when there is a history of radiation exposure or clinically and sonographically unsuspicious lymph nodes, while dissection of regional nodes should be performed if lymph node metastasis is suspected.53 Additional treatments reported included acitretin, interferon alfa, topical imiquimod, curettage, debridement, and electrodesiccation, but because of the limited number of cases and variable efficacy, no conclusions can be made on the utility of these alternative modalities.
The lowest rate of reported recurrence was found with amputation, followed by MMS and WLE. Amputation is the most aggressive treatment option, but its superiority in lower recurrence rates was not statistically significant when compared with either WLE or MMS after Yates correction. Despite treatment with radical surgery, recurrence is still possible and may be associated with factors including greater size (>2 cm) and depth (>4 mm), poor histologic differentiation, perineural involvement, failure of previous treatments, and immunosuppression.63 No statistically significant difference in recurrence rates was found among surgical methods, though data trended toward lower rates of recurrence with MMS compared with WLE, as recurrence with MMS was only reported in 2 cases.25,56
The efficacy of MMS is well documented for tumors with contiguous growth and enables maximum preservation of normal tissue structure and function with complete margin visualization. Thus, our results are in agreement with those of prior studies,54-56,64 suggesting that MMS is associated with lower recurrence rates for EC than WLE. Future studies and reporting of MMS for EC are particularly important because of the functional importance of the plantar foot.
It is important to note that there are local and systemic risk factors that increase the likelihood of developing EC and facilitate tumor growth, including antecedent trauma to the lesion site, chronic irritation or infection, and immunosuppression (HIV related or iatrogenic medication induced). These risk factors may play a role in the treatment modality utilized (eg, more aggressive EC may be treated with amputation instead of WLE). Underlying patient comorbidities could potentially affect recurrence rates, which is a variable we could not control for in our analysis.
Our findings are limited by study design, with supporting evidence consisting of case reports and series. The review is limited by interstudy variability and heterogeneity of results. Additionally, recurrence is not reported in all cases and may be a source of sampling bias. Further complicating the generalizability of these results is the lack of follow-up to evaluate morbidity and quality of life after treatment.
CONCLUSION
This review suggests that MMS is associated with lower recurrence rates than WLE for the treatment of EC. Further investigation of MMS for EC with appropriate follow-up is necessary to identify whether MMS is associated with lower recurrence and less functional impairment. Nonsurgical treatments, including topical imiquimod, interferon alfa, and acitretin, may be useful in cases where surgical therapies are contraindicated, but there is little evidence to support these treatment modalities. Treatment guidelines for EC are not established, and appropriate treatment guidelines should be developed in the future.
- McKee PH, Wilkinson JD, Black MM, et al. Carcinoma (epithelioma) cuniculatum: a clinicopathological study of nineteen cases and review of the literature. Histopathology. 1981;5:425-436.
- Aird I, Johnson HD, Lennox B, et al. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245-250.
- Seremet S, Erdemir AT, Kiremitci U, et al. Unusually early-onset plantar verrucous carcinoma. Cutis. 2019;104:34-36.
- Spyriounis PK, Tentis D, Sparveri IF, et al. Plantar epithelioma cuniculatum. a case report with review of the literature. Eur J Plast Surg. 2004;27:253-256.
- Ho J, Diven G, Bu J, et al. An ulcerating verrucous plaque on the foot. verrucous carcinoma (epithelioma cuniculatum). Arch Dermatol. 2000;136:547-548, 550-551.
- Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
- Zielonka E, Goldschmidt D, de Fontaine S. Verrucous carcinoma or epithelioma cuniculatum plantare. Eur J Surg Oncol. 1997;23:86-87.
- Dogan G, Oram Y, Hazneci E, et al. Three cases of verrucous carcinoma. Australas J Dermatol. 1998;39:251-254.
- Schwartz RA, Burgess GH. Verrucous carcinoma of the foot. J Surg Oncol. 1980;14:333-339.
- McKay C, McBride P, Muir J. Plantar verrucous carcinoma masquerading as toe web intertrigo. Australas J Dermatol. 2012;53:2010-2012.
- Shenoy AS, Waghmare RS, Kavishwar VS, et al. Carcinoma cuniculatum of foot. Foot. 2011;21:207-208.
- Lozzi G, Perris K. Carcinoma cuniculatum. CMAJ. 2007;177:249-251.
- Schein O, Orenstein A, Bar-Meir E. Plantar verrucous carcicoma (epithelioma cuniculatum): rare form of the common wart. Isr Med Assoc J. 2006;8:885.
- Rheingold LM, Roth LM. Carcinoma of the skin of the foot exhibiting some verrucous features. Plast Reconstr Surg. 1978;61:605-609.
- Klima M, Kurtis B, Jordan PH. Verrucous carcinoma of skin. J Cutan Pathol. 1980;7:88-98.
- Nakamura Y, Kashiwagi K, Nakamura A, et al. Verrucous carcinoma of the foot diagnosed using p53 and Ki-67 immunostaining in a patient with diabetic neuropathy. Am J Dermatopathol. 2015;37:257-259.
- Costache M, Desa LT, Mitrache LE, et al. Cutaneous verrucous carcinoma—report of three cases with review of literature. Rom J Morphol Embryol. 2014;55:383-388.
- Terada T. Verrucous carcinoma of the skin: a report on 5 Japanese cases. Ann Diagn Pathol. 2011;15:175-180.
- Noel JC, Heenen M, Peny MO, et al. Proliferating cell nuclear antigen distribution in verrucous carcinoma of the skin. Br J Dermatol. 1995;133:868-873.
- García-Gavín J, González-Vilas D, Rodríguez-Pazos L, et al. Verrucous carcinoma of the foot affecting the bone: utility of the computed tomography scanner. Dermatol Online J. 2010;16:3-5.
- Wasserman PL, Taylor RC, Pinillia J, et al. Verrucous carcinoma of the foot and enhancement assessment by MRI. Skeletal Radiol. 2009;38:393-395.
- Bhushan MH, Ferguson JE, Hutchinson CE. Carcinoma cuniculatum of the foot assessed by magnetic resonance scanning. Clin Exp Dermatol. 2001;26:419-422.
- Penera KE, Manji KA, Craig AB, et al. Atypical presentation of verrucous carcinoma: a case study and review of the literature. Foot Ankle Spec. 2013;6:318-322.
- Suen K, Wijeratne S, Patrikios J. An unusual case of bilateral verrucous carcinoma of the foot (epithelioma cuniculatum). J Surg Case Rep. 2012;2012:rjs020.
- Riccio C, King K, Elston JB, et al. Bilateral plantar verrucous carcinoma. Eplasty. 2016;16:ic46.
- Di Palma V, Stone JP, Schell A, et al. Mistaken diabetic ulcers: a case of bilateral foot verrucous carcinoma. Case Rep Dermatol Med. 2018;2018:4192657.
- Seehafer JR, Muller SA, Dicken CH. Bilateral verrucous carcinoma of the feet. Orthop Surv. 1979;3:205.
- Tosti A, Morelli R, Fanti PA, et al. Carcinoma cuniculatum of the nail apparatus: report of three cases. Dermatology. 1993;186:217-221.
- Melo CR, Melo IS, Souza LP. Epithelioma cuniculatum, a verrucous carcinoma of the foot. report of 2 cases. Dermatologica. 1981;163:338-342.
- Van Geertruyden JP, Olemans C, Laporte M, et al. Verrucous carcinoma of the nail bed. Foot Ankle Int. 1998;19:327-328.
- Thakur BK, Verma S, Raphael V. Verrucous carcinoma developing in a long standing case of ulcerative lichen planus of sole: a rare case report. J Eur Acad Dermatol Venereol. 2015;29:399-401.
- Mayron R, Grimwood RE, Siegle RJ, et al. Verrucous carcinoma arising in ulcerative lichen planus of the soles. J Dermatol Surg Oncol. 1988;14:547-551.
- Boussofara L, Belajouza-Noueiri C, Ghariani N, et al. Verrucous epidermoid carcinoma as a complication in cutaneous lichen planus [article in French]. Ann Dermatol Venereol. 2006;133:404-405.
- Khullar G, Mittal S, Sharma S. Verrucous carcinoma on the foot arising in a chronic neuropathic ulcer of leprosy. Australas J Dermatol. 2019;60:245-246.
- Ochsner PE, Hausman R, Olsthoorn PGM. Epithelioma cunicalutum developing in a neuropathic ulcer of leprous etiology. Arch Orthop Trauma Surg. 1979;94:227-231.
- Ray R, Bhagat A, Vasudevan B, et al. A rare case of plantar epithelioma cuniculatum arising from a wart. Indian J Dermatol. 2015;60:485-487.
- Imko-Walczuk B, Cegielska A, Placek W, et al. Human papillomavirus-related verrucous carcinoma in a renal transplant patient after long-term immunosuppression: a case report. Transplant Proc. 2014;46:2916-2919.
- Floristán MU, Feltes RA, Sáenz JC, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 18. Actas Dermosifiliogr. 2009;100:433-435.
- Sasaoka R, Morimura T, Mihara M, et al. Detection of human pupillomavirus type 16 DNA in two cases of verriicous carcinoma of the foot. Br J Dermatol. 1996;134:983984.
- Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
- Knobler RM, Schneider S, Neumann RA, et al. DNA dot‐blot hybridization implicates human papillomavirus type 11‐DNA in epithelioma cuniculatum. J Med Virol. 1989;29:33-37.
- Noel JC, Peny MO, Detremmerie O, et al. Demonstration of human papillomavirus type 2 in a verrucous carcinoma of the foot. Dermatology. 1993;187:58-61.
- Jungmann J, Vogt T, Müller CSL. Giant verrucous carcinoma of the lower extremity in women with dementia. BMJ Case Rep. 2012;2012:bcr2012006357.
- McKee PH, Wilkinson JD, Corbett MF, et al. Carcinoma cuniculatum: a case metastasizing to skin and lymph nodes. Clin Exp Dermatol. 1981;6:613-618.
- Owen WR, Wolfe ID, Burnett JW, et al. Epithelioma cuniculatum. South Med J. 1978;71:477-479.
- Patel AN, Bedforth N, Varma S. Pain-free treatment of carcinoma cuniculatum on the heel using Mohs micrographic surgery and ultrasonography-guided sciatic nerve block. Clin Exp Dermatol. 2013;38:569-571.
- Padilla RS, Bailin PL, Howard WR, et al. Verrucous carcinoma of the skin and its management by Mohs’ surgery. Plast Reconstr Surg. 1984;73:442-447.
- Kotwal M, Poflee S, Bobhate S. Carcinoma cuniculatum at various anatomical sites. Indian J Dermatol. 2005;50:216-220.
- Arefi M, Philipone E, Caprioli R, et al. A case of verrucous carcinoma (epithelioma cuniculatum) of the heel mimicking infected epidermal cyst and gout. Foot Ankle Spec. 2008;1:297-299.
- Trebing D, Brunner M, Kröning Y, et al. Young man with verrucous heel tumor [article in German]. J Dtsch Dermatol Ges. 2003;9:739-741.
- Thompson SG. Epithelioma cuniculatum: an unusual tumour of the foot. Br J Plast Surg. 1965;18:214-217.
- Thomas EJ, Graves NC, Meritt SM. Carcinoma cuniculatum: an atypical presentation in the foot. J Foot Ankle Surg. 2014;53:356-359.
- Koch H, Kowatsch E, Hödl S, et al. Verrucous carcinoma of the skin: long-term follow-up results following surgical therapy. Dermatol Surg. 2004;30:1124-1130.
- Mallatt BD, Ceilley RI, Dryer RF. Management of verrucous carcinoma on a foot by a combination of chemosurgery and plastic repair: report of a case. J Dermatol Surg Oncol. 1980;6:532-534.
- Mohs FE, Sahl WJ. Chemosurgery for verrucous carcinoma. J Dermatol Surg Oncol. 1979;5:302-306.
- Alkalay R, Alcalay J, Shiri J. Plantar verrucous carcinoma treated with Mohs micrographic surgery: a case report and literature review. J Drugs Dermatol. 2006;5:68-73.
- Mora RG. Microscopically controlled surgery (Mohs’ chemosurgery) for treatment of verrucous squamous cell carcinoma of the foot (epithelioma cuniculatum). J Am Acad Dermatol. 1983;8:354-362.
- Risse L, Negrier P, Dang PM, et al. Treatment of verrucous carcinoma with recombinant alfa-interferon. Dermatology. 1995;190:142-144.
- Rogozin´ski TT, Schwartz RA, Towpik E. Verrucous carcinoma in Unna-Thost hyperkeratosis of the palms and soles. J Am Acad Dermatol. 1994;31:1061-1062.
- Kuan YZ, Hsu HC, Kuo TT, et al. Multiple verrucous carcinomas treated with acitretin. J Am Acad Dermatol. 2007;56(2 suppl):S29-S32.
- Schalock PC, Kornik RI, Baughman RD, et al. Treatment of verrucous carcinoma with topical imiquimod. J Am Acad Dermatol. 2006;54:233-234.
- Brown SM, Freeman RG. Epithelioma cuniculatum. Arch Dermatol. 1976;112:1295-1296.
- Rowe DE, Carroll RJ, Day CL, et al. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
- Swanson NA, Taylor WB. Plantar verrucous carcinoma: literature review and treatment by the Mohs’ chemosurgery technique. Arch Dermatol. 1980;116:794-797.
Verrucous carcinoma (VC) is an uncommon type of well-differentiated squamous cell carcinoma (SCC) that most commonly affects men in the fifth to sixth decades of life. 1 The tumor grows slowly over a decade or more and does not frequently metastasize but has a high propensity for recurrence and local invasion. 2 There are 3 main subtypes of VC classified by anatomic site: oral florid papillomatosis (oral cavity), Buschke-Lowenstein tumor (anogenital region), and epithelioma cuniculatum (EC)(feet). 3 Epithelioma cuniculatum, also known as carcinoma cuniculatum or papillomatosis cutis carcinoides, most commonly presents as a solitary, warty or cauliflowerlike, exophytic mass with keratin-filled sinus tracts and malodorous discharge. 4 Diabetic foot ulcers and chronic inflammatory conditions are predisposing risk factors for EC, and it can result in difficulty walking/immobility, pain, and bleeding depending on anatomic involvement. 5-9
The differential diagnosis for VC includes refractory verruca vulgaris, clavus, SCC, keratoacanthoma, deep fungal or mycobacterial infection, eccrine poroma or porocarcinoma, amelanotic melanoma, and sarcoma.10-13 The slow-growing nature of VC, sampling error of superficial biopsies, and minimal cytological atypia on histologic examination can contribute to delayed diagnosis and appropriate treatment.14 Characteristic histologic features include hyperkeratosis, papillomatosis, marked acanthosis, broad blunt-ended rete ridges with a “bulldozing” architecture, and minimal cytologic atypia and mitoses.5,6 In some cases, pleomorphism and glassy eosinophilic cytoplasmic changes may be more pronounced than that of a common wart though less dramatic than that of conventional SCCs.15 Antigen Ki-67 and tumor protein p53 have been proposed to help differentiate between common plantar verruca, VC, and SCC, but the histologic diagnosis remains challenging, and repeat histopathologic examination often is required.16-19 Following diagnosis, computed tomography or magnetic resonance imaging may be necessary to determine tumor extension and assess for deep tissue and bony involvement.20-22
Treatment of EC is particularly challenging because of the anatomic location and need for margin control while maintaining adequate function, preserving healthy tissue, and providing coverage of defects. Surgical excision of EC is the first-line treatment, most commonly by wide local excision (WLE) or amputation. Mohs micrographic surgery (MMS) also has been utilized. One review found no recurrences in 5 cases of EC treated with MMS.23 As MMS is a tissue-sparing technique, this is a valuable modality for sites of functional importance such as the feet. Herein, we review various reported EC treatment modalities and outcomes, with an emphasis on recurrence rates for WLE and MMS.
METHODS
A systematic literature review of PubMed articles indexed for MEDLINE, as well as databases including the Cochrane Library, Web of Science, and Cumulative Index to Nursing and Allied Health Literature (CINAHL), was performed on January 14, 2020. Two authors (S.S.D. and S.V.C.) independently screened results using the search terms (plantar OR foot) AND (verrucous carcinoma OR epithelioma cuniculatum OR carcinoma cuniculatum). The search terms were chosen according to MeSH subject headings. All articles from the start date of the databases through the search date were screened, and articles pertaining to VC, EC, or carcinoma cuniculatum located on the foot were included. Of these, non–English-language articles were translated and included. Articles reporting VC on a site other than the foot (eg, the oral cavity) or benign verrucous skin lesions were excluded. The reference lists for all articles also were reviewed for additional reports that were absent from the initial search using both included and excluded articles. A full-text review was performed on 221 articles published between 1954 and 2019 per the PRISMA guidelines (Figure).

A total of 101 articles were included in the study for qualitative analysis. Nearly all articles identified were case reports, giving an evidence level of 5 by the Centre for Evidence-Based Medicine rating scale. Five articles reported data on multiple patients without individual demographic or clinical details and were excluded from analysis. Of the remaining 96 articles, information about patient characteristics, tumor size, treatment modality, and recurrence were extracted for 115 cases.
RESULTS
Of the 115 cases that were reviewed, 81 (70%) were male and 33 (29%) were female with a male-to-female ratio of 2.4:1. Ages of the patients ranged from 18 to 88 years; the mean and median age was 56 years. Nearly all reported cases of EC affected the plantar surface of one foot, with 4 reports of tumors affecting both feet.24-27 One case affecting both feet reported known exposure to lead arsenate pesticides27; all others were associated with a clinical history of chronic ulcers or warts persisting for several years to decades. Other less common sites of EC included the dorsal foot, interdigital web space, and subungual digit.28-30 The most common location reported was the anterior ball of the foot. Tumors were reported to arise within pre-existing lesions, such as hypertrophic lichen planus or chronic foot wounds associated with diabetes mellitus or leprosy.31-35 Tumor size ranged from 1 to 22 cm with a median of 4.5 cm.
Eight cases were reported to be associated with human papillomavirus; low-risk types 6 and 11 and high-risk types 16 and 18 were found in 6 cases.36-41 Two cases reported association with human papillomavirus type 2.7,42
Metastases to dermal and subdermal lymphatics, regional lymph nodes, and the lungs were reported in 3 cases, repectively.43-45 Of these, one primary tumor had received low-dose irradiation in the form of X-ray therapy.45
Treatment Modalities
The cases of EC that we reviewed included treatment with surgical and systemic therapies as well as other modalities such as acitretin, interferon alfa, topical imiquimod, curettage, debridement, electrodesiccation, and radiation. The Table includes a complete summary of the treatments we analyzed.

Surgical Therapy—The majority (91% [105/115]) of cases were treated surgically. The most common treatment modality was WLE (50% [58/115]), followed by amputation (37% [43/115]) and MMS (12% [14/115]).
Wide local excision was the most frequently reported treatment, with excision margins of at least 5 mm to 1 cm.48 Incidence of recurrence was reported for 57% (33/58) of cases treated with WLE; of these, the recurrence rate was 33% (11/33). For patients with EC recurrence, the most common secondary treatment was repeat excision with wider margins (1–2 cm) or amputation (5/11).49-52 Few postoperative complications were reported but included pain, infection, and difficulty walking, which were mostly associated with repair modality (eg, split-thickness skin grafts, rotational flaps).53 Amputation was the second most common treatment modality, with a 67% (29/43) incidence of recurrence. Types of amputation included transmetatarsal ray amputation (7/43 [16%]), foot or forefoot amputation (2/43 [5%]), above-the-knee amputation (1/43 [2%]), and below-the-knee amputation (1/43 [2%]). Complications associated with amputation included infection and requirement of prosthetics for ambulation. Split-thickness skin grafts and rotational flaps were the most common surgical repairs performed.52,53
Mohs micrographic surgery was the least frequently reported surgical treatment modality. Both traditional MMS on fresh tissue and “slow Mohs,” with formalin-fixed paraffin embedded tissue examination over several days, were performed for EC with horizontal en face sectioning.54-56 Incidence of recurrence was reported for 86% (12/14) of MMS cases. Of these, recurrence was seen in 17% (2/12) that utilized a flat horizontal processing of tissue sections coupled with saucerlike excisions to enable examination of the entire undersurface and margins. In one case, the patient was treated with MMS with recurrence noted 1 month later; thus, repeat MMS was performed, and the tumor was found to be entwined around the flexor tendon.57 The tendon was removed, and clear margins were obtained. Follow-up 3 years after the second MMS revealed no signs of recurrence.57 In the other case, the patient had a particularly aggressive course with bilateral VC in the setting of diabetic ulcers that was treated with WLE prior to MMS and recurrence still noted after MMS.26 No complications were reported with MMS.
Overall, recurrence was most frequently reported with WLE (11/33 [33%]), followed by MMS (2/12 [17%]) and amputation (3/29 [10%]). When comparing WLE and amputation, the relationship between treatment modality and recurrence was statistically significant using a χ2 test of independence (χ2=4.7; P=.03). However, results were not significant with Yates correction for continuity (χ2=3.4; P=.06). The χ2 test of independence showed no significant association between treatment method and recurrence when comparing WLE with MMS (χ2=1.2; P=.28). Reported follow-up times varied greatly from a few months to 10 years.
Systemic Therapy—Of the total cases, only 2 cases reported treatment with acitretin and 2 utilized interferon alfa.58,59 In one case, treatment of EC with interferon alfa alone required more aggressive therapy (ie, amputation).58 Neither of the 2 cases using acitretin reported recurrence.59,60 Complications of acitretin therapy included cheilitis and transaminitis.60
Other Treatment Modalities—Three cases utilized imiquimod, with 2 cases of imiquimod monotherapy and 1 case of imiquimod in combination with electrodesiccation and WLE.37 One of the cases of EC treated with imiquimod monotherapy recurred and required WLE.61
There were reports of other treatments including curettage alone (2% [2/115]),40,62 debridement alone (1% [1/115]),40 electrodesiccation (1% [1/115]),37 and radiation (1% [1/115]).43 Recurrence was found with curettage alone and debridement alone. Electrodesiccation was reported in conjunction with WLE without recurrence. Radiation was used to treat a case of VC that had metastasized to the lymph nodes; no follow-up was described.43
COMMENT
Epithelioma cuniculatum is an indolent malignancy of the plantar foot that likely is frequently underdiagnosed or misdiagnosed because of location, sampling error, and challenges in histopathologic diagnosis. Once diagnosed, surgical removal with margin control is the first-line therapy for EC. Our review found a number of surgical, systemic, and other treatment modalities that have been used to treat EC, but there remains a lack of evidence to provide clear guidelines as to which therapies are most effective. Current data on the treatment of EC largely are limited to case reports and case series. To date, there are no reports of higher-quality studies or randomized controlled trials to assess the efficacy of various treatment modalities.
Our review found that WLE is the most common treatment modality for EC, followed by amputation and MMS. Three cases43-45 that reported metastasis to lymph nodes also were treated with fine-needle aspiration or biopsy, and it is recommended that sentinel lymph node biopsy be performed when there is a history of radiation exposure or clinically and sonographically unsuspicious lymph nodes, while dissection of regional nodes should be performed if lymph node metastasis is suspected.53 Additional treatments reported included acitretin, interferon alfa, topical imiquimod, curettage, debridement, and electrodesiccation, but because of the limited number of cases and variable efficacy, no conclusions can be made on the utility of these alternative modalities.
The lowest rate of reported recurrence was found with amputation, followed by MMS and WLE. Amputation is the most aggressive treatment option, but its superiority in lower recurrence rates was not statistically significant when compared with either WLE or MMS after Yates correction. Despite treatment with radical surgery, recurrence is still possible and may be associated with factors including greater size (>2 cm) and depth (>4 mm), poor histologic differentiation, perineural involvement, failure of previous treatments, and immunosuppression.63 No statistically significant difference in recurrence rates was found among surgical methods, though data trended toward lower rates of recurrence with MMS compared with WLE, as recurrence with MMS was only reported in 2 cases.25,56
The efficacy of MMS is well documented for tumors with contiguous growth and enables maximum preservation of normal tissue structure and function with complete margin visualization. Thus, our results are in agreement with those of prior studies,54-56,64 suggesting that MMS is associated with lower recurrence rates for EC than WLE. Future studies and reporting of MMS for EC are particularly important because of the functional importance of the plantar foot.
It is important to note that there are local and systemic risk factors that increase the likelihood of developing EC and facilitate tumor growth, including antecedent trauma to the lesion site, chronic irritation or infection, and immunosuppression (HIV related or iatrogenic medication induced). These risk factors may play a role in the treatment modality utilized (eg, more aggressive EC may be treated with amputation instead of WLE). Underlying patient comorbidities could potentially affect recurrence rates, which is a variable we could not control for in our analysis.
Our findings are limited by study design, with supporting evidence consisting of case reports and series. The review is limited by interstudy variability and heterogeneity of results. Additionally, recurrence is not reported in all cases and may be a source of sampling bias. Further complicating the generalizability of these results is the lack of follow-up to evaluate morbidity and quality of life after treatment.
CONCLUSION
This review suggests that MMS is associated with lower recurrence rates than WLE for the treatment of EC. Further investigation of MMS for EC with appropriate follow-up is necessary to identify whether MMS is associated with lower recurrence and less functional impairment. Nonsurgical treatments, including topical imiquimod, interferon alfa, and acitretin, may be useful in cases where surgical therapies are contraindicated, but there is little evidence to support these treatment modalities. Treatment guidelines for EC are not established, and appropriate treatment guidelines should be developed in the future.
Verrucous carcinoma (VC) is an uncommon type of well-differentiated squamous cell carcinoma (SCC) that most commonly affects men in the fifth to sixth decades of life. 1 The tumor grows slowly over a decade or more and does not frequently metastasize but has a high propensity for recurrence and local invasion. 2 There are 3 main subtypes of VC classified by anatomic site: oral florid papillomatosis (oral cavity), Buschke-Lowenstein tumor (anogenital region), and epithelioma cuniculatum (EC)(feet). 3 Epithelioma cuniculatum, also known as carcinoma cuniculatum or papillomatosis cutis carcinoides, most commonly presents as a solitary, warty or cauliflowerlike, exophytic mass with keratin-filled sinus tracts and malodorous discharge. 4 Diabetic foot ulcers and chronic inflammatory conditions are predisposing risk factors for EC, and it can result in difficulty walking/immobility, pain, and bleeding depending on anatomic involvement. 5-9
The differential diagnosis for VC includes refractory verruca vulgaris, clavus, SCC, keratoacanthoma, deep fungal or mycobacterial infection, eccrine poroma or porocarcinoma, amelanotic melanoma, and sarcoma.10-13 The slow-growing nature of VC, sampling error of superficial biopsies, and minimal cytological atypia on histologic examination can contribute to delayed diagnosis and appropriate treatment.14 Characteristic histologic features include hyperkeratosis, papillomatosis, marked acanthosis, broad blunt-ended rete ridges with a “bulldozing” architecture, and minimal cytologic atypia and mitoses.5,6 In some cases, pleomorphism and glassy eosinophilic cytoplasmic changes may be more pronounced than that of a common wart though less dramatic than that of conventional SCCs.15 Antigen Ki-67 and tumor protein p53 have been proposed to help differentiate between common plantar verruca, VC, and SCC, but the histologic diagnosis remains challenging, and repeat histopathologic examination often is required.16-19 Following diagnosis, computed tomography or magnetic resonance imaging may be necessary to determine tumor extension and assess for deep tissue and bony involvement.20-22
Treatment of EC is particularly challenging because of the anatomic location and need for margin control while maintaining adequate function, preserving healthy tissue, and providing coverage of defects. Surgical excision of EC is the first-line treatment, most commonly by wide local excision (WLE) or amputation. Mohs micrographic surgery (MMS) also has been utilized. One review found no recurrences in 5 cases of EC treated with MMS.23 As MMS is a tissue-sparing technique, this is a valuable modality for sites of functional importance such as the feet. Herein, we review various reported EC treatment modalities and outcomes, with an emphasis on recurrence rates for WLE and MMS.
METHODS
A systematic literature review of PubMed articles indexed for MEDLINE, as well as databases including the Cochrane Library, Web of Science, and Cumulative Index to Nursing and Allied Health Literature (CINAHL), was performed on January 14, 2020. Two authors (S.S.D. and S.V.C.) independently screened results using the search terms (plantar OR foot) AND (verrucous carcinoma OR epithelioma cuniculatum OR carcinoma cuniculatum). The search terms were chosen according to MeSH subject headings. All articles from the start date of the databases through the search date were screened, and articles pertaining to VC, EC, or carcinoma cuniculatum located on the foot were included. Of these, non–English-language articles were translated and included. Articles reporting VC on a site other than the foot (eg, the oral cavity) or benign verrucous skin lesions were excluded. The reference lists for all articles also were reviewed for additional reports that were absent from the initial search using both included and excluded articles. A full-text review was performed on 221 articles published between 1954 and 2019 per the PRISMA guidelines (Figure).

A total of 101 articles were included in the study for qualitative analysis. Nearly all articles identified were case reports, giving an evidence level of 5 by the Centre for Evidence-Based Medicine rating scale. Five articles reported data on multiple patients without individual demographic or clinical details and were excluded from analysis. Of the remaining 96 articles, information about patient characteristics, tumor size, treatment modality, and recurrence were extracted for 115 cases.
RESULTS
Of the 115 cases that were reviewed, 81 (70%) were male and 33 (29%) were female with a male-to-female ratio of 2.4:1. Ages of the patients ranged from 18 to 88 years; the mean and median age was 56 years. Nearly all reported cases of EC affected the plantar surface of one foot, with 4 reports of tumors affecting both feet.24-27 One case affecting both feet reported known exposure to lead arsenate pesticides27; all others were associated with a clinical history of chronic ulcers or warts persisting for several years to decades. Other less common sites of EC included the dorsal foot, interdigital web space, and subungual digit.28-30 The most common location reported was the anterior ball of the foot. Tumors were reported to arise within pre-existing lesions, such as hypertrophic lichen planus or chronic foot wounds associated with diabetes mellitus or leprosy.31-35 Tumor size ranged from 1 to 22 cm with a median of 4.5 cm.
Eight cases were reported to be associated with human papillomavirus; low-risk types 6 and 11 and high-risk types 16 and 18 were found in 6 cases.36-41 Two cases reported association with human papillomavirus type 2.7,42
Metastases to dermal and subdermal lymphatics, regional lymph nodes, and the lungs were reported in 3 cases, repectively.43-45 Of these, one primary tumor had received low-dose irradiation in the form of X-ray therapy.45
Treatment Modalities
The cases of EC that we reviewed included treatment with surgical and systemic therapies as well as other modalities such as acitretin, interferon alfa, topical imiquimod, curettage, debridement, electrodesiccation, and radiation. The Table includes a complete summary of the treatments we analyzed.

Surgical Therapy—The majority (91% [105/115]) of cases were treated surgically. The most common treatment modality was WLE (50% [58/115]), followed by amputation (37% [43/115]) and MMS (12% [14/115]).
Wide local excision was the most frequently reported treatment, with excision margins of at least 5 mm to 1 cm.48 Incidence of recurrence was reported for 57% (33/58) of cases treated with WLE; of these, the recurrence rate was 33% (11/33). For patients with EC recurrence, the most common secondary treatment was repeat excision with wider margins (1–2 cm) or amputation (5/11).49-52 Few postoperative complications were reported but included pain, infection, and difficulty walking, which were mostly associated with repair modality (eg, split-thickness skin grafts, rotational flaps).53 Amputation was the second most common treatment modality, with a 67% (29/43) incidence of recurrence. Types of amputation included transmetatarsal ray amputation (7/43 [16%]), foot or forefoot amputation (2/43 [5%]), above-the-knee amputation (1/43 [2%]), and below-the-knee amputation (1/43 [2%]). Complications associated with amputation included infection and requirement of prosthetics for ambulation. Split-thickness skin grafts and rotational flaps were the most common surgical repairs performed.52,53
Mohs micrographic surgery was the least frequently reported surgical treatment modality. Both traditional MMS on fresh tissue and “slow Mohs,” with formalin-fixed paraffin embedded tissue examination over several days, were performed for EC with horizontal en face sectioning.54-56 Incidence of recurrence was reported for 86% (12/14) of MMS cases. Of these, recurrence was seen in 17% (2/12) that utilized a flat horizontal processing of tissue sections coupled with saucerlike excisions to enable examination of the entire undersurface and margins. In one case, the patient was treated with MMS with recurrence noted 1 month later; thus, repeat MMS was performed, and the tumor was found to be entwined around the flexor tendon.57 The tendon was removed, and clear margins were obtained. Follow-up 3 years after the second MMS revealed no signs of recurrence.57 In the other case, the patient had a particularly aggressive course with bilateral VC in the setting of diabetic ulcers that was treated with WLE prior to MMS and recurrence still noted after MMS.26 No complications were reported with MMS.
Overall, recurrence was most frequently reported with WLE (11/33 [33%]), followed by MMS (2/12 [17%]) and amputation (3/29 [10%]). When comparing WLE and amputation, the relationship between treatment modality and recurrence was statistically significant using a χ2 test of independence (χ2=4.7; P=.03). However, results were not significant with Yates correction for continuity (χ2=3.4; P=.06). The χ2 test of independence showed no significant association between treatment method and recurrence when comparing WLE with MMS (χ2=1.2; P=.28). Reported follow-up times varied greatly from a few months to 10 years.
Systemic Therapy—Of the total cases, only 2 cases reported treatment with acitretin and 2 utilized interferon alfa.58,59 In one case, treatment of EC with interferon alfa alone required more aggressive therapy (ie, amputation).58 Neither of the 2 cases using acitretin reported recurrence.59,60 Complications of acitretin therapy included cheilitis and transaminitis.60
Other Treatment Modalities—Three cases utilized imiquimod, with 2 cases of imiquimod monotherapy and 1 case of imiquimod in combination with electrodesiccation and WLE.37 One of the cases of EC treated with imiquimod monotherapy recurred and required WLE.61
There were reports of other treatments including curettage alone (2% [2/115]),40,62 debridement alone (1% [1/115]),40 electrodesiccation (1% [1/115]),37 and radiation (1% [1/115]).43 Recurrence was found with curettage alone and debridement alone. Electrodesiccation was reported in conjunction with WLE without recurrence. Radiation was used to treat a case of VC that had metastasized to the lymph nodes; no follow-up was described.43
COMMENT
Epithelioma cuniculatum is an indolent malignancy of the plantar foot that likely is frequently underdiagnosed or misdiagnosed because of location, sampling error, and challenges in histopathologic diagnosis. Once diagnosed, surgical removal with margin control is the first-line therapy for EC. Our review found a number of surgical, systemic, and other treatment modalities that have been used to treat EC, but there remains a lack of evidence to provide clear guidelines as to which therapies are most effective. Current data on the treatment of EC largely are limited to case reports and case series. To date, there are no reports of higher-quality studies or randomized controlled trials to assess the efficacy of various treatment modalities.
Our review found that WLE is the most common treatment modality for EC, followed by amputation and MMS. Three cases43-45 that reported metastasis to lymph nodes also were treated with fine-needle aspiration or biopsy, and it is recommended that sentinel lymph node biopsy be performed when there is a history of radiation exposure or clinically and sonographically unsuspicious lymph nodes, while dissection of regional nodes should be performed if lymph node metastasis is suspected.53 Additional treatments reported included acitretin, interferon alfa, topical imiquimod, curettage, debridement, and electrodesiccation, but because of the limited number of cases and variable efficacy, no conclusions can be made on the utility of these alternative modalities.
The lowest rate of reported recurrence was found with amputation, followed by MMS and WLE. Amputation is the most aggressive treatment option, but its superiority in lower recurrence rates was not statistically significant when compared with either WLE or MMS after Yates correction. Despite treatment with radical surgery, recurrence is still possible and may be associated with factors including greater size (>2 cm) and depth (>4 mm), poor histologic differentiation, perineural involvement, failure of previous treatments, and immunosuppression.63 No statistically significant difference in recurrence rates was found among surgical methods, though data trended toward lower rates of recurrence with MMS compared with WLE, as recurrence with MMS was only reported in 2 cases.25,56
The efficacy of MMS is well documented for tumors with contiguous growth and enables maximum preservation of normal tissue structure and function with complete margin visualization. Thus, our results are in agreement with those of prior studies,54-56,64 suggesting that MMS is associated with lower recurrence rates for EC than WLE. Future studies and reporting of MMS for EC are particularly important because of the functional importance of the plantar foot.
It is important to note that there are local and systemic risk factors that increase the likelihood of developing EC and facilitate tumor growth, including antecedent trauma to the lesion site, chronic irritation or infection, and immunosuppression (HIV related or iatrogenic medication induced). These risk factors may play a role in the treatment modality utilized (eg, more aggressive EC may be treated with amputation instead of WLE). Underlying patient comorbidities could potentially affect recurrence rates, which is a variable we could not control for in our analysis.
Our findings are limited by study design, with supporting evidence consisting of case reports and series. The review is limited by interstudy variability and heterogeneity of results. Additionally, recurrence is not reported in all cases and may be a source of sampling bias. Further complicating the generalizability of these results is the lack of follow-up to evaluate morbidity and quality of life after treatment.
CONCLUSION
This review suggests that MMS is associated with lower recurrence rates than WLE for the treatment of EC. Further investigation of MMS for EC with appropriate follow-up is necessary to identify whether MMS is associated with lower recurrence and less functional impairment. Nonsurgical treatments, including topical imiquimod, interferon alfa, and acitretin, may be useful in cases where surgical therapies are contraindicated, but there is little evidence to support these treatment modalities. Treatment guidelines for EC are not established, and appropriate treatment guidelines should be developed in the future.
- McKee PH, Wilkinson JD, Black MM, et al. Carcinoma (epithelioma) cuniculatum: a clinicopathological study of nineteen cases and review of the literature. Histopathology. 1981;5:425-436.
- Aird I, Johnson HD, Lennox B, et al. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245-250.
- Seremet S, Erdemir AT, Kiremitci U, et al. Unusually early-onset plantar verrucous carcinoma. Cutis. 2019;104:34-36.
- Spyriounis PK, Tentis D, Sparveri IF, et al. Plantar epithelioma cuniculatum. a case report with review of the literature. Eur J Plast Surg. 2004;27:253-256.
- Ho J, Diven G, Bu J, et al. An ulcerating verrucous plaque on the foot. verrucous carcinoma (epithelioma cuniculatum). Arch Dermatol. 2000;136:547-548, 550-551.
- Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
- Zielonka E, Goldschmidt D, de Fontaine S. Verrucous carcinoma or epithelioma cuniculatum plantare. Eur J Surg Oncol. 1997;23:86-87.
- Dogan G, Oram Y, Hazneci E, et al. Three cases of verrucous carcinoma. Australas J Dermatol. 1998;39:251-254.
- Schwartz RA, Burgess GH. Verrucous carcinoma of the foot. J Surg Oncol. 1980;14:333-339.
- McKay C, McBride P, Muir J. Plantar verrucous carcinoma masquerading as toe web intertrigo. Australas J Dermatol. 2012;53:2010-2012.
- Shenoy AS, Waghmare RS, Kavishwar VS, et al. Carcinoma cuniculatum of foot. Foot. 2011;21:207-208.
- Lozzi G, Perris K. Carcinoma cuniculatum. CMAJ. 2007;177:249-251.
- Schein O, Orenstein A, Bar-Meir E. Plantar verrucous carcicoma (epithelioma cuniculatum): rare form of the common wart. Isr Med Assoc J. 2006;8:885.
- Rheingold LM, Roth LM. Carcinoma of the skin of the foot exhibiting some verrucous features. Plast Reconstr Surg. 1978;61:605-609.
- Klima M, Kurtis B, Jordan PH. Verrucous carcinoma of skin. J Cutan Pathol. 1980;7:88-98.
- Nakamura Y, Kashiwagi K, Nakamura A, et al. Verrucous carcinoma of the foot diagnosed using p53 and Ki-67 immunostaining in a patient with diabetic neuropathy. Am J Dermatopathol. 2015;37:257-259.
- Costache M, Desa LT, Mitrache LE, et al. Cutaneous verrucous carcinoma—report of three cases with review of literature. Rom J Morphol Embryol. 2014;55:383-388.
- Terada T. Verrucous carcinoma of the skin: a report on 5 Japanese cases. Ann Diagn Pathol. 2011;15:175-180.
- Noel JC, Heenen M, Peny MO, et al. Proliferating cell nuclear antigen distribution in verrucous carcinoma of the skin. Br J Dermatol. 1995;133:868-873.
- García-Gavín J, González-Vilas D, Rodríguez-Pazos L, et al. Verrucous carcinoma of the foot affecting the bone: utility of the computed tomography scanner. Dermatol Online J. 2010;16:3-5.
- Wasserman PL, Taylor RC, Pinillia J, et al. Verrucous carcinoma of the foot and enhancement assessment by MRI. Skeletal Radiol. 2009;38:393-395.
- Bhushan MH, Ferguson JE, Hutchinson CE. Carcinoma cuniculatum of the foot assessed by magnetic resonance scanning. Clin Exp Dermatol. 2001;26:419-422.
- Penera KE, Manji KA, Craig AB, et al. Atypical presentation of verrucous carcinoma: a case study and review of the literature. Foot Ankle Spec. 2013;6:318-322.
- Suen K, Wijeratne S, Patrikios J. An unusual case of bilateral verrucous carcinoma of the foot (epithelioma cuniculatum). J Surg Case Rep. 2012;2012:rjs020.
- Riccio C, King K, Elston JB, et al. Bilateral plantar verrucous carcinoma. Eplasty. 2016;16:ic46.
- Di Palma V, Stone JP, Schell A, et al. Mistaken diabetic ulcers: a case of bilateral foot verrucous carcinoma. Case Rep Dermatol Med. 2018;2018:4192657.
- Seehafer JR, Muller SA, Dicken CH. Bilateral verrucous carcinoma of the feet. Orthop Surv. 1979;3:205.
- Tosti A, Morelli R, Fanti PA, et al. Carcinoma cuniculatum of the nail apparatus: report of three cases. Dermatology. 1993;186:217-221.
- Melo CR, Melo IS, Souza LP. Epithelioma cuniculatum, a verrucous carcinoma of the foot. report of 2 cases. Dermatologica. 1981;163:338-342.
- Van Geertruyden JP, Olemans C, Laporte M, et al. Verrucous carcinoma of the nail bed. Foot Ankle Int. 1998;19:327-328.
- Thakur BK, Verma S, Raphael V. Verrucous carcinoma developing in a long standing case of ulcerative lichen planus of sole: a rare case report. J Eur Acad Dermatol Venereol. 2015;29:399-401.
- Mayron R, Grimwood RE, Siegle RJ, et al. Verrucous carcinoma arising in ulcerative lichen planus of the soles. J Dermatol Surg Oncol. 1988;14:547-551.
- Boussofara L, Belajouza-Noueiri C, Ghariani N, et al. Verrucous epidermoid carcinoma as a complication in cutaneous lichen planus [article in French]. Ann Dermatol Venereol. 2006;133:404-405.
- Khullar G, Mittal S, Sharma S. Verrucous carcinoma on the foot arising in a chronic neuropathic ulcer of leprosy. Australas J Dermatol. 2019;60:245-246.
- Ochsner PE, Hausman R, Olsthoorn PGM. Epithelioma cunicalutum developing in a neuropathic ulcer of leprous etiology. Arch Orthop Trauma Surg. 1979;94:227-231.
- Ray R, Bhagat A, Vasudevan B, et al. A rare case of plantar epithelioma cuniculatum arising from a wart. Indian J Dermatol. 2015;60:485-487.
- Imko-Walczuk B, Cegielska A, Placek W, et al. Human papillomavirus-related verrucous carcinoma in a renal transplant patient after long-term immunosuppression: a case report. Transplant Proc. 2014;46:2916-2919.
- Floristán MU, Feltes RA, Sáenz JC, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 18. Actas Dermosifiliogr. 2009;100:433-435.
- Sasaoka R, Morimura T, Mihara M, et al. Detection of human pupillomavirus type 16 DNA in two cases of verriicous carcinoma of the foot. Br J Dermatol. 1996;134:983984.
- Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
- Knobler RM, Schneider S, Neumann RA, et al. DNA dot‐blot hybridization implicates human papillomavirus type 11‐DNA in epithelioma cuniculatum. J Med Virol. 1989;29:33-37.
- Noel JC, Peny MO, Detremmerie O, et al. Demonstration of human papillomavirus type 2 in a verrucous carcinoma of the foot. Dermatology. 1993;187:58-61.
- Jungmann J, Vogt T, Müller CSL. Giant verrucous carcinoma of the lower extremity in women with dementia. BMJ Case Rep. 2012;2012:bcr2012006357.
- McKee PH, Wilkinson JD, Corbett MF, et al. Carcinoma cuniculatum: a case metastasizing to skin and lymph nodes. Clin Exp Dermatol. 1981;6:613-618.
- Owen WR, Wolfe ID, Burnett JW, et al. Epithelioma cuniculatum. South Med J. 1978;71:477-479.
- Patel AN, Bedforth N, Varma S. Pain-free treatment of carcinoma cuniculatum on the heel using Mohs micrographic surgery and ultrasonography-guided sciatic nerve block. Clin Exp Dermatol. 2013;38:569-571.
- Padilla RS, Bailin PL, Howard WR, et al. Verrucous carcinoma of the skin and its management by Mohs’ surgery. Plast Reconstr Surg. 1984;73:442-447.
- Kotwal M, Poflee S, Bobhate S. Carcinoma cuniculatum at various anatomical sites. Indian J Dermatol. 2005;50:216-220.
- Arefi M, Philipone E, Caprioli R, et al. A case of verrucous carcinoma (epithelioma cuniculatum) of the heel mimicking infected epidermal cyst and gout. Foot Ankle Spec. 2008;1:297-299.
- Trebing D, Brunner M, Kröning Y, et al. Young man with verrucous heel tumor [article in German]. J Dtsch Dermatol Ges. 2003;9:739-741.
- Thompson SG. Epithelioma cuniculatum: an unusual tumour of the foot. Br J Plast Surg. 1965;18:214-217.
- Thomas EJ, Graves NC, Meritt SM. Carcinoma cuniculatum: an atypical presentation in the foot. J Foot Ankle Surg. 2014;53:356-359.
- Koch H, Kowatsch E, Hödl S, et al. Verrucous carcinoma of the skin: long-term follow-up results following surgical therapy. Dermatol Surg. 2004;30:1124-1130.
- Mallatt BD, Ceilley RI, Dryer RF. Management of verrucous carcinoma on a foot by a combination of chemosurgery and plastic repair: report of a case. J Dermatol Surg Oncol. 1980;6:532-534.
- Mohs FE, Sahl WJ. Chemosurgery for verrucous carcinoma. J Dermatol Surg Oncol. 1979;5:302-306.
- Alkalay R, Alcalay J, Shiri J. Plantar verrucous carcinoma treated with Mohs micrographic surgery: a case report and literature review. J Drugs Dermatol. 2006;5:68-73.
- Mora RG. Microscopically controlled surgery (Mohs’ chemosurgery) for treatment of verrucous squamous cell carcinoma of the foot (epithelioma cuniculatum). J Am Acad Dermatol. 1983;8:354-362.
- Risse L, Negrier P, Dang PM, et al. Treatment of verrucous carcinoma with recombinant alfa-interferon. Dermatology. 1995;190:142-144.
- Rogozin´ski TT, Schwartz RA, Towpik E. Verrucous carcinoma in Unna-Thost hyperkeratosis of the palms and soles. J Am Acad Dermatol. 1994;31:1061-1062.
- Kuan YZ, Hsu HC, Kuo TT, et al. Multiple verrucous carcinomas treated with acitretin. J Am Acad Dermatol. 2007;56(2 suppl):S29-S32.
- Schalock PC, Kornik RI, Baughman RD, et al. Treatment of verrucous carcinoma with topical imiquimod. J Am Acad Dermatol. 2006;54:233-234.
- Brown SM, Freeman RG. Epithelioma cuniculatum. Arch Dermatol. 1976;112:1295-1296.
- Rowe DE, Carroll RJ, Day CL, et al. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
- Swanson NA, Taylor WB. Plantar verrucous carcinoma: literature review and treatment by the Mohs’ chemosurgery technique. Arch Dermatol. 1980;116:794-797.
- McKee PH, Wilkinson JD, Black MM, et al. Carcinoma (epithelioma) cuniculatum: a clinicopathological study of nineteen cases and review of the literature. Histopathology. 1981;5:425-436.
- Aird I, Johnson HD, Lennox B, et al. Epithelioma cuniculatum: a variety of squamous carcinoma peculiar to the foot. Br J Surg. 1954;42:245-250.
- Seremet S, Erdemir AT, Kiremitci U, et al. Unusually early-onset plantar verrucous carcinoma. Cutis. 2019;104:34-36.
- Spyriounis PK, Tentis D, Sparveri IF, et al. Plantar epithelioma cuniculatum. a case report with review of the literature. Eur J Plast Surg. 2004;27:253-256.
- Ho J, Diven G, Bu J, et al. An ulcerating verrucous plaque on the foot. verrucous carcinoma (epithelioma cuniculatum). Arch Dermatol. 2000;136:547-548, 550-551.
- Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin): a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
- Zielonka E, Goldschmidt D, de Fontaine S. Verrucous carcinoma or epithelioma cuniculatum plantare. Eur J Surg Oncol. 1997;23:86-87.
- Dogan G, Oram Y, Hazneci E, et al. Three cases of verrucous carcinoma. Australas J Dermatol. 1998;39:251-254.
- Schwartz RA, Burgess GH. Verrucous carcinoma of the foot. J Surg Oncol. 1980;14:333-339.
- McKay C, McBride P, Muir J. Plantar verrucous carcinoma masquerading as toe web intertrigo. Australas J Dermatol. 2012;53:2010-2012.
- Shenoy AS, Waghmare RS, Kavishwar VS, et al. Carcinoma cuniculatum of foot. Foot. 2011;21:207-208.
- Lozzi G, Perris K. Carcinoma cuniculatum. CMAJ. 2007;177:249-251.
- Schein O, Orenstein A, Bar-Meir E. Plantar verrucous carcicoma (epithelioma cuniculatum): rare form of the common wart. Isr Med Assoc J. 2006;8:885.
- Rheingold LM, Roth LM. Carcinoma of the skin of the foot exhibiting some verrucous features. Plast Reconstr Surg. 1978;61:605-609.
- Klima M, Kurtis B, Jordan PH. Verrucous carcinoma of skin. J Cutan Pathol. 1980;7:88-98.
- Nakamura Y, Kashiwagi K, Nakamura A, et al. Verrucous carcinoma of the foot diagnosed using p53 and Ki-67 immunostaining in a patient with diabetic neuropathy. Am J Dermatopathol. 2015;37:257-259.
- Costache M, Desa LT, Mitrache LE, et al. Cutaneous verrucous carcinoma—report of three cases with review of literature. Rom J Morphol Embryol. 2014;55:383-388.
- Terada T. Verrucous carcinoma of the skin: a report on 5 Japanese cases. Ann Diagn Pathol. 2011;15:175-180.
- Noel JC, Heenen M, Peny MO, et al. Proliferating cell nuclear antigen distribution in verrucous carcinoma of the skin. Br J Dermatol. 1995;133:868-873.
- García-Gavín J, González-Vilas D, Rodríguez-Pazos L, et al. Verrucous carcinoma of the foot affecting the bone: utility of the computed tomography scanner. Dermatol Online J. 2010;16:3-5.
- Wasserman PL, Taylor RC, Pinillia J, et al. Verrucous carcinoma of the foot and enhancement assessment by MRI. Skeletal Radiol. 2009;38:393-395.
- Bhushan MH, Ferguson JE, Hutchinson CE. Carcinoma cuniculatum of the foot assessed by magnetic resonance scanning. Clin Exp Dermatol. 2001;26:419-422.
- Penera KE, Manji KA, Craig AB, et al. Atypical presentation of verrucous carcinoma: a case study and review of the literature. Foot Ankle Spec. 2013;6:318-322.
- Suen K, Wijeratne S, Patrikios J. An unusual case of bilateral verrucous carcinoma of the foot (epithelioma cuniculatum). J Surg Case Rep. 2012;2012:rjs020.
- Riccio C, King K, Elston JB, et al. Bilateral plantar verrucous carcinoma. Eplasty. 2016;16:ic46.
- Di Palma V, Stone JP, Schell A, et al. Mistaken diabetic ulcers: a case of bilateral foot verrucous carcinoma. Case Rep Dermatol Med. 2018;2018:4192657.
- Seehafer JR, Muller SA, Dicken CH. Bilateral verrucous carcinoma of the feet. Orthop Surv. 1979;3:205.
- Tosti A, Morelli R, Fanti PA, et al. Carcinoma cuniculatum of the nail apparatus: report of three cases. Dermatology. 1993;186:217-221.
- Melo CR, Melo IS, Souza LP. Epithelioma cuniculatum, a verrucous carcinoma of the foot. report of 2 cases. Dermatologica. 1981;163:338-342.
- Van Geertruyden JP, Olemans C, Laporte M, et al. Verrucous carcinoma of the nail bed. Foot Ankle Int. 1998;19:327-328.
- Thakur BK, Verma S, Raphael V. Verrucous carcinoma developing in a long standing case of ulcerative lichen planus of sole: a rare case report. J Eur Acad Dermatol Venereol. 2015;29:399-401.
- Mayron R, Grimwood RE, Siegle RJ, et al. Verrucous carcinoma arising in ulcerative lichen planus of the soles. J Dermatol Surg Oncol. 1988;14:547-551.
- Boussofara L, Belajouza-Noueiri C, Ghariani N, et al. Verrucous epidermoid carcinoma as a complication in cutaneous lichen planus [article in French]. Ann Dermatol Venereol. 2006;133:404-405.
- Khullar G, Mittal S, Sharma S. Verrucous carcinoma on the foot arising in a chronic neuropathic ulcer of leprosy. Australas J Dermatol. 2019;60:245-246.
- Ochsner PE, Hausman R, Olsthoorn PGM. Epithelioma cunicalutum developing in a neuropathic ulcer of leprous etiology. Arch Orthop Trauma Surg. 1979;94:227-231.
- Ray R, Bhagat A, Vasudevan B, et al. A rare case of plantar epithelioma cuniculatum arising from a wart. Indian J Dermatol. 2015;60:485-487.
- Imko-Walczuk B, Cegielska A, Placek W, et al. Human papillomavirus-related verrucous carcinoma in a renal transplant patient after long-term immunosuppression: a case report. Transplant Proc. 2014;46:2916-2919.
- Floristán MU, Feltes RA, Sáenz JC, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 18. Actas Dermosifiliogr. 2009;100:433-435.
- Sasaoka R, Morimura T, Mihara M, et al. Detection of human pupillomavirus type 16 DNA in two cases of verriicous carcinoma of the foot. Br J Dermatol. 1996;134:983984.
- Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
- Knobler RM, Schneider S, Neumann RA, et al. DNA dot‐blot hybridization implicates human papillomavirus type 11‐DNA in epithelioma cuniculatum. J Med Virol. 1989;29:33-37.
- Noel JC, Peny MO, Detremmerie O, et al. Demonstration of human papillomavirus type 2 in a verrucous carcinoma of the foot. Dermatology. 1993;187:58-61.
- Jungmann J, Vogt T, Müller CSL. Giant verrucous carcinoma of the lower extremity in women with dementia. BMJ Case Rep. 2012;2012:bcr2012006357.
- McKee PH, Wilkinson JD, Corbett MF, et al. Carcinoma cuniculatum: a case metastasizing to skin and lymph nodes. Clin Exp Dermatol. 1981;6:613-618.
- Owen WR, Wolfe ID, Burnett JW, et al. Epithelioma cuniculatum. South Med J. 1978;71:477-479.
- Patel AN, Bedforth N, Varma S. Pain-free treatment of carcinoma cuniculatum on the heel using Mohs micrographic surgery and ultrasonography-guided sciatic nerve block. Clin Exp Dermatol. 2013;38:569-571.
- Padilla RS, Bailin PL, Howard WR, et al. Verrucous carcinoma of the skin and its management by Mohs’ surgery. Plast Reconstr Surg. 1984;73:442-447.
- Kotwal M, Poflee S, Bobhate S. Carcinoma cuniculatum at various anatomical sites. Indian J Dermatol. 2005;50:216-220.
- Arefi M, Philipone E, Caprioli R, et al. A case of verrucous carcinoma (epithelioma cuniculatum) of the heel mimicking infected epidermal cyst and gout. Foot Ankle Spec. 2008;1:297-299.
- Trebing D, Brunner M, Kröning Y, et al. Young man with verrucous heel tumor [article in German]. J Dtsch Dermatol Ges. 2003;9:739-741.
- Thompson SG. Epithelioma cuniculatum: an unusual tumour of the foot. Br J Plast Surg. 1965;18:214-217.
- Thomas EJ, Graves NC, Meritt SM. Carcinoma cuniculatum: an atypical presentation in the foot. J Foot Ankle Surg. 2014;53:356-359.
- Koch H, Kowatsch E, Hödl S, et al. Verrucous carcinoma of the skin: long-term follow-up results following surgical therapy. Dermatol Surg. 2004;30:1124-1130.
- Mallatt BD, Ceilley RI, Dryer RF. Management of verrucous carcinoma on a foot by a combination of chemosurgery and plastic repair: report of a case. J Dermatol Surg Oncol. 1980;6:532-534.
- Mohs FE, Sahl WJ. Chemosurgery for verrucous carcinoma. J Dermatol Surg Oncol. 1979;5:302-306.
- Alkalay R, Alcalay J, Shiri J. Plantar verrucous carcinoma treated with Mohs micrographic surgery: a case report and literature review. J Drugs Dermatol. 2006;5:68-73.
- Mora RG. Microscopically controlled surgery (Mohs’ chemosurgery) for treatment of verrucous squamous cell carcinoma of the foot (epithelioma cuniculatum). J Am Acad Dermatol. 1983;8:354-362.
- Risse L, Negrier P, Dang PM, et al. Treatment of verrucous carcinoma with recombinant alfa-interferon. Dermatology. 1995;190:142-144.
- Rogozin´ski TT, Schwartz RA, Towpik E. Verrucous carcinoma in Unna-Thost hyperkeratosis of the palms and soles. J Am Acad Dermatol. 1994;31:1061-1062.
- Kuan YZ, Hsu HC, Kuo TT, et al. Multiple verrucous carcinomas treated with acitretin. J Am Acad Dermatol. 2007;56(2 suppl):S29-S32.
- Schalock PC, Kornik RI, Baughman RD, et al. Treatment of verrucous carcinoma with topical imiquimod. J Am Acad Dermatol. 2006;54:233-234.
- Brown SM, Freeman RG. Epithelioma cuniculatum. Arch Dermatol. 1976;112:1295-1296.
- Rowe DE, Carroll RJ, Day CL, et al. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
- Swanson NA, Taylor WB. Plantar verrucous carcinoma: literature review and treatment by the Mohs’ chemosurgery technique. Arch Dermatol. 1980;116:794-797.
Practice Points
- Because of its slow-growing nature and propensity for local invasion and recurrence, diagnosis of epithelioma cuniculatum (EC) often is delayed and therefore can be associated with notable morbidity.
- Wide local excision with 5-mm to 1-cm margins is considered standard of care and is the most commonly reported treatment of EC. Amputation may be required in cases with extensive local destruction.
- Mohs micrographic surgery is a viable option for treatment of EC, with more recent cases suggesting favorable outcomes regarding recurrence rates.
Can skin care aid use of diabetes devices?
Technologies that allow people to monitor blood sugar and automate the administration of insulin have radically transformed the lives of patients – and children in particular – with type 1 diabetes. But the devices often come with a cost: Insulin pumps and continuous glucose monitors can irritate the skin at the points of contact, causing some people to stop using their pumps or monitors altogether.
Regular use of lipid-rich skin creams can reduce eczema in children who use insulin pumps and continuous glucose monitors to manage type 1 diabetes, Danish researchers reported last month. The article is currently undergoing peer review at The Lancet Diabetes and Endocrinology, and the authors said they hope their approach will deter more children from abandoning diabetes technology.
“A simple thing can actually change a lot,” said Anna Korsgaard Berg, MD, a pediatrician who specializes in diabetes care at Copenhagen University Hospital’s Steno Diabetes Center in Herlev, Denmark, and a coauthor of the new study. “Not all skin reactions can be solved by the skin care program, but it can help improve the issue.”
More than 1.5 million children and adolescents worldwide live with type 1 diabetes, a condition that requires continuous insulin infusion. Insulin pumps meet this need in many wealthier countries, and are often used in combination with sensors that measure a child’s glucose level. Both the American Diabetes Association and the International Society for Adolescent and Pediatric Diabetes recommend insulin pumps and continuous glucose monitors as core treatment tools.
Dr. Berg and colleagues, who have previously shown that as many as 90% of children who use these devices experience some kind of skin reaction, want to minimize the rate of such discomfort in hopes that fewer children stop using the devices. According to a 2014 study, 18% of people with type 1 diabetes who stopped using continuous glucose monitors did so because of skin irritation.
Lather on that lipid-rich lotion
Dr. Berg and colleagues studied 170 children and adolescents with type 1 diabetes (average age, 11 years) who use insulin pumps, continuous glucose monitors, or both. From March 2020 to July 2021, 112 children (55 girls) employed a skin care program developed for the study, while the other 58 (34 girls) did not receive any skin care advice.
The skin care group received instructions about how to gently insert and remove their insulin pumps or glucose monitors, to minimize skin damage. They also were told to avoid disinfectants such as alcohol, which can irritate skin. The children in this group used a cream containing 70% lipids to help rehydrate their skin, applying the salve each day a device was not inserted into their skin.
Eczema can be a real problem for kids who use insulin pumps and continuous glucose monitors to manage type 1 diabetes. Researchers found that regular use of lipid-rich skin creams can reduce its incidence.
Although insulin pumps and glucose monitors are kept in place for longer periods of time than they once were, Dr. Berg and colleagues noted, users do periodically remove them when bathing or when undergoing medical tests that involve x-rays. On days when the devices were not in place for a period of time, children in the skin care group were encouraged to follow the protocol.
Study results
One-third of children in the skin care group developed eczema or experienced a wound, compared with almost half of the children in the control group, according to the researchers. The absolute difference in developing eczema or wounds between the two groups was 12.9 % (95% confidence interval, –28.7% to 2.9%).
Children in the skin care group were much less likely to develop wounds, the researchers found, when they focused only on wounds and not eczema (odds ratio, 0.29, 95% CI, 0.12-0.68).
Dr. Berg said she would like to explore whether other techniques, such as a combination of patches, adhesives, or other lotions, yield even better results.
“Anything that can help people use technology more consistently is better for both quality of life and diabetes outcomes,” said Priya Prahalad, MD, a specialist in pediatric endocrinology and diabetes at Stanford Medicine Children’s Health in Palo Alto and Sunnyvale, Calif.
Dr. Prahalad, who was not involved in the Danish study, said that although the sample sizes in the trial were relatively small, the data are “headed in the right direction.”
Pediatricians already recommend using moisturizing creams at the sites where pumps or glucose monitors are inserted into the skin, she noted. But the new study simply employed an especially moisturizing cream to mitigate skin damage.
Although one reason for skin irritation may be the repeated insertion and removal of devices, Dr. Berg and Dr. Prahalad stressed that the medical devices themselves may contain allergy-causing components. Device makers are not required to disclose what’s inside the boxes.
“I do not understand why the full content of a device is not by law mandatory to declare, when declaration by law is mandatory for many other products and drugs but not for medical devices,” Dr. Berg said.
Dr. Berg reports receiving lipid cream from Teva Pharmaceuticals and research support from Medtronic. Dr. Prahalad reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Technologies that allow people to monitor blood sugar and automate the administration of insulin have radically transformed the lives of patients – and children in particular – with type 1 diabetes. But the devices often come with a cost: Insulin pumps and continuous glucose monitors can irritate the skin at the points of contact, causing some people to stop using their pumps or monitors altogether.
Regular use of lipid-rich skin creams can reduce eczema in children who use insulin pumps and continuous glucose monitors to manage type 1 diabetes, Danish researchers reported last month. The article is currently undergoing peer review at The Lancet Diabetes and Endocrinology, and the authors said they hope their approach will deter more children from abandoning diabetes technology.
“A simple thing can actually change a lot,” said Anna Korsgaard Berg, MD, a pediatrician who specializes in diabetes care at Copenhagen University Hospital’s Steno Diabetes Center in Herlev, Denmark, and a coauthor of the new study. “Not all skin reactions can be solved by the skin care program, but it can help improve the issue.”
More than 1.5 million children and adolescents worldwide live with type 1 diabetes, a condition that requires continuous insulin infusion. Insulin pumps meet this need in many wealthier countries, and are often used in combination with sensors that measure a child’s glucose level. Both the American Diabetes Association and the International Society for Adolescent and Pediatric Diabetes recommend insulin pumps and continuous glucose monitors as core treatment tools.
Dr. Berg and colleagues, who have previously shown that as many as 90% of children who use these devices experience some kind of skin reaction, want to minimize the rate of such discomfort in hopes that fewer children stop using the devices. According to a 2014 study, 18% of people with type 1 diabetes who stopped using continuous glucose monitors did so because of skin irritation.
Lather on that lipid-rich lotion
Dr. Berg and colleagues studied 170 children and adolescents with type 1 diabetes (average age, 11 years) who use insulin pumps, continuous glucose monitors, or both. From March 2020 to July 2021, 112 children (55 girls) employed a skin care program developed for the study, while the other 58 (34 girls) did not receive any skin care advice.
The skin care group received instructions about how to gently insert and remove their insulin pumps or glucose monitors, to minimize skin damage. They also were told to avoid disinfectants such as alcohol, which can irritate skin. The children in this group used a cream containing 70% lipids to help rehydrate their skin, applying the salve each day a device was not inserted into their skin.
Eczema can be a real problem for kids who use insulin pumps and continuous glucose monitors to manage type 1 diabetes. Researchers found that regular use of lipid-rich skin creams can reduce its incidence.
Although insulin pumps and glucose monitors are kept in place for longer periods of time than they once were, Dr. Berg and colleagues noted, users do periodically remove them when bathing or when undergoing medical tests that involve x-rays. On days when the devices were not in place for a period of time, children in the skin care group were encouraged to follow the protocol.
Study results
One-third of children in the skin care group developed eczema or experienced a wound, compared with almost half of the children in the control group, according to the researchers. The absolute difference in developing eczema or wounds between the two groups was 12.9 % (95% confidence interval, –28.7% to 2.9%).
Children in the skin care group were much less likely to develop wounds, the researchers found, when they focused only on wounds and not eczema (odds ratio, 0.29, 95% CI, 0.12-0.68).
Dr. Berg said she would like to explore whether other techniques, such as a combination of patches, adhesives, or other lotions, yield even better results.
“Anything that can help people use technology more consistently is better for both quality of life and diabetes outcomes,” said Priya Prahalad, MD, a specialist in pediatric endocrinology and diabetes at Stanford Medicine Children’s Health in Palo Alto and Sunnyvale, Calif.
Dr. Prahalad, who was not involved in the Danish study, said that although the sample sizes in the trial were relatively small, the data are “headed in the right direction.”
Pediatricians already recommend using moisturizing creams at the sites where pumps or glucose monitors are inserted into the skin, she noted. But the new study simply employed an especially moisturizing cream to mitigate skin damage.
Although one reason for skin irritation may be the repeated insertion and removal of devices, Dr. Berg and Dr. Prahalad stressed that the medical devices themselves may contain allergy-causing components. Device makers are not required to disclose what’s inside the boxes.
“I do not understand why the full content of a device is not by law mandatory to declare, when declaration by law is mandatory for many other products and drugs but not for medical devices,” Dr. Berg said.
Dr. Berg reports receiving lipid cream from Teva Pharmaceuticals and research support from Medtronic. Dr. Prahalad reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Technologies that allow people to monitor blood sugar and automate the administration of insulin have radically transformed the lives of patients – and children in particular – with type 1 diabetes. But the devices often come with a cost: Insulin pumps and continuous glucose monitors can irritate the skin at the points of contact, causing some people to stop using their pumps or monitors altogether.
Regular use of lipid-rich skin creams can reduce eczema in children who use insulin pumps and continuous glucose monitors to manage type 1 diabetes, Danish researchers reported last month. The article is currently undergoing peer review at The Lancet Diabetes and Endocrinology, and the authors said they hope their approach will deter more children from abandoning diabetes technology.
“A simple thing can actually change a lot,” said Anna Korsgaard Berg, MD, a pediatrician who specializes in diabetes care at Copenhagen University Hospital’s Steno Diabetes Center in Herlev, Denmark, and a coauthor of the new study. “Not all skin reactions can be solved by the skin care program, but it can help improve the issue.”
More than 1.5 million children and adolescents worldwide live with type 1 diabetes, a condition that requires continuous insulin infusion. Insulin pumps meet this need in many wealthier countries, and are often used in combination with sensors that measure a child’s glucose level. Both the American Diabetes Association and the International Society for Adolescent and Pediatric Diabetes recommend insulin pumps and continuous glucose monitors as core treatment tools.
Dr. Berg and colleagues, who have previously shown that as many as 90% of children who use these devices experience some kind of skin reaction, want to minimize the rate of such discomfort in hopes that fewer children stop using the devices. According to a 2014 study, 18% of people with type 1 diabetes who stopped using continuous glucose monitors did so because of skin irritation.
Lather on that lipid-rich lotion
Dr. Berg and colleagues studied 170 children and adolescents with type 1 diabetes (average age, 11 years) who use insulin pumps, continuous glucose monitors, or both. From March 2020 to July 2021, 112 children (55 girls) employed a skin care program developed for the study, while the other 58 (34 girls) did not receive any skin care advice.
The skin care group received instructions about how to gently insert and remove their insulin pumps or glucose monitors, to minimize skin damage. They also were told to avoid disinfectants such as alcohol, which can irritate skin. The children in this group used a cream containing 70% lipids to help rehydrate their skin, applying the salve each day a device was not inserted into their skin.
Eczema can be a real problem for kids who use insulin pumps and continuous glucose monitors to manage type 1 diabetes. Researchers found that regular use of lipid-rich skin creams can reduce its incidence.
Although insulin pumps and glucose monitors are kept in place for longer periods of time than they once were, Dr. Berg and colleagues noted, users do periodically remove them when bathing or when undergoing medical tests that involve x-rays. On days when the devices were not in place for a period of time, children in the skin care group were encouraged to follow the protocol.
Study results
One-third of children in the skin care group developed eczema or experienced a wound, compared with almost half of the children in the control group, according to the researchers. The absolute difference in developing eczema or wounds between the two groups was 12.9 % (95% confidence interval, –28.7% to 2.9%).
Children in the skin care group were much less likely to develop wounds, the researchers found, when they focused only on wounds and not eczema (odds ratio, 0.29, 95% CI, 0.12-0.68).
Dr. Berg said she would like to explore whether other techniques, such as a combination of patches, adhesives, or other lotions, yield even better results.
“Anything that can help people use technology more consistently is better for both quality of life and diabetes outcomes,” said Priya Prahalad, MD, a specialist in pediatric endocrinology and diabetes at Stanford Medicine Children’s Health in Palo Alto and Sunnyvale, Calif.
Dr. Prahalad, who was not involved in the Danish study, said that although the sample sizes in the trial were relatively small, the data are “headed in the right direction.”
Pediatricians already recommend using moisturizing creams at the sites where pumps or glucose monitors are inserted into the skin, she noted. But the new study simply employed an especially moisturizing cream to mitigate skin damage.
Although one reason for skin irritation may be the repeated insertion and removal of devices, Dr. Berg and Dr. Prahalad stressed that the medical devices themselves may contain allergy-causing components. Device makers are not required to disclose what’s inside the boxes.
“I do not understand why the full content of a device is not by law mandatory to declare, when declaration by law is mandatory for many other products and drugs but not for medical devices,” Dr. Berg said.
Dr. Berg reports receiving lipid cream from Teva Pharmaceuticals and research support from Medtronic. Dr. Prahalad reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.






