User login
Medicine grapples with physician suicide
One day in 1986, a medical school classmate handed Dr. Robert P. Bright a gun that she intended to use to kill herself. She asked him to hold on to it for her and to keep quiet about her sense of hopelessness.
“She didn’t want anybody in the medical school to know; it was all hidden and hush-hushed,” recalled Dr. Bright, who is now a psychiatrist at the Mayo Clinic, Scottsdale, Ariz. “I was trying to juggle that with the issue of safety.”
He honored his classmate’s request for confidentiality, but he sought advice from the medical school dean about what to do. Before long, his classmate sought help from a psychiatrist and got better with medication and psychotherapy. “It turned out well, thank goodness,” Dr. Bright said.
Similar stories of despair among medical students and physicians don’t always end well. The American Foundation for Suicide Prevention estimates that 300-400 U.S. physicians commit suicide each year, about one per day. Suicide deaths are 250%-400% higher among female physicians, compared with women in other professions, and 70% higher among male physicians, compared with men in other professions. Major depression is a common risk factor, along with bipolar disorder and substance abuse.
Depression and other mood disorders may be underrecognized and inadequately treated in physicians because they may be reluctant to seek treatment, may attempt to diagnose and treat themselves, or may seek and receive “VIP treatment” from health care providers, according to a review article coauthored by Dr. Bright (Current Psych. 2011;10:16-30).
“Physicians struggling with these things are very much in the closet about it,” he said. “It’s a sad reflection on the stigma that’s still in our country that people can’t come forth and say, ‘I’m struggling with depression or anxiety.’ ”
Researchers led by Dr. Katherine J. Gold at the University of Michigan used data from the National Violent Death Reporting System to evaluate suicide among physicians and found that job stressors “may impact physician identity and be a particular risk factor for which more attention is warranted” (Gen. Hosp. Psychiatry 2013;35:45-9).
Work dissatisfaction sent Dr. Pamela Wible into a tailspin early in her career. In 2004 she found herself in a suicidal state for about 6 weeks, “I stayed at home, crying myself into my pillow and I never sought help from my colleagues,” recalled Dr. Wible, a family physician in Eugene, Ore., who currently leads training sessions in medical student and physician suicide prevention. “I was not depressed before entering the medical profession, but [I had developed] constant thoughts of ‘Can I just disappear? What’s the easiest way to do this?’ I got to a place of complete surrender but I didn’t have the gun. I didn’t have the stockpile of pills. I didn’t have a follow-through on the plan.”
Instead of taking her own life, she “had an epiphany” and changed the way she practiced medicine. She said that owning her own clinic empowered her to “become the doctor I had originally described on my personal statement when I entered medical school.”
According to Dr. Charles F. Reynolds III, a psychiatrist at the University of Pittsburgh, reluctance to seek treatment can also be driven by concerns about the amount of time that treatment could take.
“As physicians, we often don’t appropriately take care of ourselves when it comes to issues like depression,” said Dr. Reynolds, who also directs the National Institute of Mental Health–sponsored Center of Excellence in the Prevention and Treatment of Late Life Mood Disorders. “We may still see it as a character weakness rather than as a medical illness that can be diagnosed and appropriately treated. Concerns about privacy also figure into the concerns of some physicians as well.”
Practicing in a rural area or small community can also be an obstacle to treatment, not only because of limited access to psychiatrists, but because the “patient” may be the only physician in town.
“As much as there’s stigma for everybody being voluntarily or involuntarily admitted [for suicidal ideation], it’s a little different when you’re a provider within the hospital where you’re seeking care,” Dr. Bright noted.
He said that if he had the opportunity to counsel physicians experiencing suicidal thoughts, he would “remind them of the medical nature of depression, that the brain is just another organ and the organ is not making chemicals just like the pancreas doesn’t make insulin in diabetes,” he said. “I’d also encourage them to get the treatment that they need. I would encourage compassion for themselves that they would give to anybody else in the same situation.”
He said that he would advise them to find a mental health provider “that they trust with confidentiality, and to reach out to other people for support. I would also let them know about the physician assistance programs that are available. There’s one through Vanderbilt (the Vanderbilt Center for Professional Health) and several others that specialize in working with physicians who are struggling with mental health or substance abuse or disruptive behavior.”
Dr. Reynolds’ core message to distressed physicians is that “you’re a better doctor for your patients, and a better father or mother for your family, if you’re taking good care of yourself,” he said. “It’s hard for you to take care of your patients if you’re not also taking care of yourself, if you’re burning out. Get help. Treatment works.”
Dr. Christine Moutier, chief medical officer of the American Foundation for Suicide Prevention, added that troubled physicians “should feel no shame for the fact that they’re in distress. Any of us can get there through a whole variety of different pathways that life presents. There’s science and data to support this experience as commonplace and having underpinnings that are of no fault to anyone. That’s the reality.”
Dr. Wible, who has lost several colleagues and physician friends to suicide, said that she hopes for a more transparent discussion of the topic by the medical profession. She presented on the topic at the 2014 annual scientific assembly of the American Academy of Family Physicians.
“The talk before mine was on Ebola, and every seat was taken” in the 900-seat room. When it came time for her presentation, “I maybe had 100 people in the room. Now, are physicians more likely to die from Ebola or from suicide? We are in a state of denial. If we don’t talk about suicide, we will continue to lose one or two medical students or doctors every day. The sooner we talk about this and connect with each other outside of a PowerPoint presentation, the sooner we’re going to solve this.”
After a physician in a large clinical department at the University of Pittsburgh took his own life several years ago, the chair of that department invited Dr. Reynolds to speak with his staff. The meeting “was primarily educational in nature, so we talked about the topic, to try to destigmatize and to educate people about the need for appropriate help-seeking,” recalled Dr. Reynolds, who is a former president of the American College of Psychiatrists. “If the leadership of a medical institution appropriately sanctions help-seeking behavior and treatment of mental disorders like depression, that’s going to make it okay for people to reach out and seek help rather than pushing it under the rug, so to speak. If the leadership says ‘this is a key thing and we don’t think you can function adequately as a medical student or as a physician if you’re not taking appropriate care of yourself,’ that helps to shift the culture.”
The ripple effect of that kind of message from health care administrators can’t be underestimated, said Dr. Moutier, who helped launch a suicide and depression awareness program at the University of California, San Diego (Acad. Med. 2012;87:320-6). She encouraged health care leaders to stage periodic grand rounds and lectures for their medical staff about physician well-being, burnout, and the risk of suicide. “If the leader is uncomfortable talking about these things, that’s a sign they should get a little education for themselves about [these topics],” she said.
Dr. Reynolds noted that certain state medical licensure boards including those for Arkansas and Pennsylvania have incorporated destigmatizing language into relicensure exams. “Some of them previously would ask questions such as whether the applicants had a history of a mental disorder like depression,” he said. “What you’re beginning to see now increasingly is that the state medical board will ask more generic questions, like ‘Do you have any conditions that would interfere with the practice of your specialty in medicine?’ This is a good thing.”
He said that he is optimistic about future of physician well-being, noting that the University of Pittsburgh and other medical schools have incorporated wellness principles into first-year curriculum. “We underscore the importance of students becoming sensitive to one another, learning how to recognize depression in each other and creating a culture in which students can encourage each other to engage in appropriate help-seeking,” Dr. Reynolds explained. “I think we are witnessing a shift in the culture of institutional medicine as we bring along new generations of physicians who are better educated about mental disorders and their treatment and issues related to suicide as we reach out to students, make counseling services available to them, educate them about these issues. That supports a cultural shift that gradually erodes the issue of stigma that has so long plagued appropriate help-seeking in medical institutions.”
Still, Dr. Wible said that she worries about the disaffected colleagues who reach out to her almost every day. “Just yesterday I got an e-mail from a physician in Oklahoma who told me they just lost three physicians to suicide in 1 month who were on probation with the medical board,” she said. “These are not defective physicians. These people need to be helped.”
Dr. Wible said that she favors holding periodic panel discussions on the topics of depression and physician suicide for medical students and physicians alike. “Let other physicians who’ve been depressed and suicidal sit in front of the room on the first week of medical school, or in a hospital once in a while, mandatory, where you listen to other well-respected physicians say, ‘yeah. I cried myself to sleep after I lost this patient,’ or ‘I had suicidal thoughts during a malpractice case.’ There are lots of reasons why physicians could be sad. They need to start talking about it publicly. Other medical students and physicians would then feel comfortable to raise their hands in the audience and say, ‘I felt the same way.’ ”
Suggested resources for help
American Foundation for Suicide Prevention (www.afsp.org/).
24-hour crisis line: 1-800-273-TALK (8255).
In 2008 the AFSP released a documentary about the problem of physician depression and suicide titled “Struggling in Silence,” which aired on public television stations nationwide and is available on DVD for $24.99.
Center for Patient and Professional Advocacy (www.mc.vanderbilt.edu/centers/cppa/index.php)
Depression and Bipolar Support Alliance (www.dbsalliance.org).
Federation of State Physician Health Programs Inc. (www.fsphp.org).
Vanderbilt Center for Professional Health (www.mc.vanderbilt.edu/cph).
The Mayo Clinic Program on Physician Well-Being (http://www.mayo.edu/research/centers-programs/physician-well-being-program/overview).
ePhysicianHealth.com, a program of the Ontario Medical Association (http://php.oma.org/ePhysicianHealth.html)
The Academic Medicine Handbook: A Guide to Achievement and Fulfillment for Academic Faculty, New York: Springer, 2013 (http://www.springer.com/medicine/internal/book/978-1-4614-5692-6)
[email protected]
On Twitter @dougbrunk
As a specialist in physician health, I shout out that we can never have too many articles on this heartbreaking tragedy that claims so many lives each year - and leaves so many devastated people in its wake.

|
| Dr. Michael F. Myers |
It is sobering and frightening that despite the excellent institutional and systemic changes outlined by Dr. Reynolds and Dr. Moutier and the moving first-hand testimonials of Dr. Bright and Dr. Wible, despairing doctors continue to die by their own hands. The loss of so many intelligent, highly trained, and compassionate caregivers is mind-numbing and unconscionable. We cannot afford to let down our guard.
As part of my research for a book in progress "When Physicians Kill Themselves: The Voices of Those They Leave Behind," I have been interviewing the family members and medical colleagues of doctors who have died by suicide.
One theme that is ascendant is how commonly the ailing physician has fallen through the cracks. Initially, he may not recognize or accept that he is burned out, depressed, or abusing alcohol and other drugs. When she does begin to understand what her symptoms suggest, the internalized stigma is so harsh and relentless that seeking help is out of the question. This drives self-medicating, but even when this does not occur and he consults a psychiatrist, punishing shame colors and works against forming a therapeutic alliance, accepting the diagnosis, keeping appointments, disclosing dangerous suicidality, adhering to medication, engaging in lifesaving psychotherapy and maintaining (or regaining) hope.
What makes matters worse is when the treating professional cuts corners (or enables self-defeating behaviors in the patient) and does not use the same judgment, monitoring, and vigilance that she uses with her nonphysician patients.
What I have found most disturbing in these narratives of my interviewees is how often their attempts to access their loved one's caregiver have fallen on deaf ears. This has to stop.
Dr. Wible says that she "had an epiphany" and changed the way she practiced medicine. It is our duty to reach out and help more physicians find their epiphany.
Dr. Michael F. Myers is professor of clinical psychiatry at SUNY Downstate Medical Center in Brooklyn, N.Y. He also is the coauthor (with Carla Fine) of "Touched by Suicide: Hope and Healing After Loss" and (with Dr. Glen O. Gabbard) of "The Physician as Patient: A Clinical Handbook for Mental Health Professionals."
As a specialist in physician health, I shout out that we can never have too many articles on this heartbreaking tragedy that claims so many lives each year - and leaves so many devastated people in its wake.

|
| Dr. Michael F. Myers |
It is sobering and frightening that despite the excellent institutional and systemic changes outlined by Dr. Reynolds and Dr. Moutier and the moving first-hand testimonials of Dr. Bright and Dr. Wible, despairing doctors continue to die by their own hands. The loss of so many intelligent, highly trained, and compassionate caregivers is mind-numbing and unconscionable. We cannot afford to let down our guard.
As part of my research for a book in progress "When Physicians Kill Themselves: The Voices of Those They Leave Behind," I have been interviewing the family members and medical colleagues of doctors who have died by suicide.
One theme that is ascendant is how commonly the ailing physician has fallen through the cracks. Initially, he may not recognize or accept that he is burned out, depressed, or abusing alcohol and other drugs. When she does begin to understand what her symptoms suggest, the internalized stigma is so harsh and relentless that seeking help is out of the question. This drives self-medicating, but even when this does not occur and he consults a psychiatrist, punishing shame colors and works against forming a therapeutic alliance, accepting the diagnosis, keeping appointments, disclosing dangerous suicidality, adhering to medication, engaging in lifesaving psychotherapy and maintaining (or regaining) hope.
What makes matters worse is when the treating professional cuts corners (or enables self-defeating behaviors in the patient) and does not use the same judgment, monitoring, and vigilance that she uses with her nonphysician patients.
What I have found most disturbing in these narratives of my interviewees is how often their attempts to access their loved one's caregiver have fallen on deaf ears. This has to stop.
Dr. Wible says that she "had an epiphany" and changed the way she practiced medicine. It is our duty to reach out and help more physicians find their epiphany.
Dr. Michael F. Myers is professor of clinical psychiatry at SUNY Downstate Medical Center in Brooklyn, N.Y. He also is the coauthor (with Carla Fine) of "Touched by Suicide: Hope and Healing After Loss" and (with Dr. Glen O. Gabbard) of "The Physician as Patient: A Clinical Handbook for Mental Health Professionals."
As a specialist in physician health, I shout out that we can never have too many articles on this heartbreaking tragedy that claims so many lives each year - and leaves so many devastated people in its wake.

|
| Dr. Michael F. Myers |
It is sobering and frightening that despite the excellent institutional and systemic changes outlined by Dr. Reynolds and Dr. Moutier and the moving first-hand testimonials of Dr. Bright and Dr. Wible, despairing doctors continue to die by their own hands. The loss of so many intelligent, highly trained, and compassionate caregivers is mind-numbing and unconscionable. We cannot afford to let down our guard.
As part of my research for a book in progress "When Physicians Kill Themselves: The Voices of Those They Leave Behind," I have been interviewing the family members and medical colleagues of doctors who have died by suicide.
One theme that is ascendant is how commonly the ailing physician has fallen through the cracks. Initially, he may not recognize or accept that he is burned out, depressed, or abusing alcohol and other drugs. When she does begin to understand what her symptoms suggest, the internalized stigma is so harsh and relentless that seeking help is out of the question. This drives self-medicating, but even when this does not occur and he consults a psychiatrist, punishing shame colors and works against forming a therapeutic alliance, accepting the diagnosis, keeping appointments, disclosing dangerous suicidality, adhering to medication, engaging in lifesaving psychotherapy and maintaining (or regaining) hope.
What makes matters worse is when the treating professional cuts corners (or enables self-defeating behaviors in the patient) and does not use the same judgment, monitoring, and vigilance that she uses with her nonphysician patients.
What I have found most disturbing in these narratives of my interviewees is how often their attempts to access their loved one's caregiver have fallen on deaf ears. This has to stop.
Dr. Wible says that she "had an epiphany" and changed the way she practiced medicine. It is our duty to reach out and help more physicians find their epiphany.
Dr. Michael F. Myers is professor of clinical psychiatry at SUNY Downstate Medical Center in Brooklyn, N.Y. He also is the coauthor (with Carla Fine) of "Touched by Suicide: Hope and Healing After Loss" and (with Dr. Glen O. Gabbard) of "The Physician as Patient: A Clinical Handbook for Mental Health Professionals."
One day in 1986, a medical school classmate handed Dr. Robert P. Bright a gun that she intended to use to kill herself. She asked him to hold on to it for her and to keep quiet about her sense of hopelessness.
“She didn’t want anybody in the medical school to know; it was all hidden and hush-hushed,” recalled Dr. Bright, who is now a psychiatrist at the Mayo Clinic, Scottsdale, Ariz. “I was trying to juggle that with the issue of safety.”
He honored his classmate’s request for confidentiality, but he sought advice from the medical school dean about what to do. Before long, his classmate sought help from a psychiatrist and got better with medication and psychotherapy. “It turned out well, thank goodness,” Dr. Bright said.
Similar stories of despair among medical students and physicians don’t always end well. The American Foundation for Suicide Prevention estimates that 300-400 U.S. physicians commit suicide each year, about one per day. Suicide deaths are 250%-400% higher among female physicians, compared with women in other professions, and 70% higher among male physicians, compared with men in other professions. Major depression is a common risk factor, along with bipolar disorder and substance abuse.
Depression and other mood disorders may be underrecognized and inadequately treated in physicians because they may be reluctant to seek treatment, may attempt to diagnose and treat themselves, or may seek and receive “VIP treatment” from health care providers, according to a review article coauthored by Dr. Bright (Current Psych. 2011;10:16-30).
“Physicians struggling with these things are very much in the closet about it,” he said. “It’s a sad reflection on the stigma that’s still in our country that people can’t come forth and say, ‘I’m struggling with depression or anxiety.’ ”
Researchers led by Dr. Katherine J. Gold at the University of Michigan used data from the National Violent Death Reporting System to evaluate suicide among physicians and found that job stressors “may impact physician identity and be a particular risk factor for which more attention is warranted” (Gen. Hosp. Psychiatry 2013;35:45-9).
Work dissatisfaction sent Dr. Pamela Wible into a tailspin early in her career. In 2004 she found herself in a suicidal state for about 6 weeks, “I stayed at home, crying myself into my pillow and I never sought help from my colleagues,” recalled Dr. Wible, a family physician in Eugene, Ore., who currently leads training sessions in medical student and physician suicide prevention. “I was not depressed before entering the medical profession, but [I had developed] constant thoughts of ‘Can I just disappear? What’s the easiest way to do this?’ I got to a place of complete surrender but I didn’t have the gun. I didn’t have the stockpile of pills. I didn’t have a follow-through on the plan.”
Instead of taking her own life, she “had an epiphany” and changed the way she practiced medicine. She said that owning her own clinic empowered her to “become the doctor I had originally described on my personal statement when I entered medical school.”
According to Dr. Charles F. Reynolds III, a psychiatrist at the University of Pittsburgh, reluctance to seek treatment can also be driven by concerns about the amount of time that treatment could take.
“As physicians, we often don’t appropriately take care of ourselves when it comes to issues like depression,” said Dr. Reynolds, who also directs the National Institute of Mental Health–sponsored Center of Excellence in the Prevention and Treatment of Late Life Mood Disorders. “We may still see it as a character weakness rather than as a medical illness that can be diagnosed and appropriately treated. Concerns about privacy also figure into the concerns of some physicians as well.”
Practicing in a rural area or small community can also be an obstacle to treatment, not only because of limited access to psychiatrists, but because the “patient” may be the only physician in town.
“As much as there’s stigma for everybody being voluntarily or involuntarily admitted [for suicidal ideation], it’s a little different when you’re a provider within the hospital where you’re seeking care,” Dr. Bright noted.
He said that if he had the opportunity to counsel physicians experiencing suicidal thoughts, he would “remind them of the medical nature of depression, that the brain is just another organ and the organ is not making chemicals just like the pancreas doesn’t make insulin in diabetes,” he said. “I’d also encourage them to get the treatment that they need. I would encourage compassion for themselves that they would give to anybody else in the same situation.”
He said that he would advise them to find a mental health provider “that they trust with confidentiality, and to reach out to other people for support. I would also let them know about the physician assistance programs that are available. There’s one through Vanderbilt (the Vanderbilt Center for Professional Health) and several others that specialize in working with physicians who are struggling with mental health or substance abuse or disruptive behavior.”
Dr. Reynolds’ core message to distressed physicians is that “you’re a better doctor for your patients, and a better father or mother for your family, if you’re taking good care of yourself,” he said. “It’s hard for you to take care of your patients if you’re not also taking care of yourself, if you’re burning out. Get help. Treatment works.”
Dr. Christine Moutier, chief medical officer of the American Foundation for Suicide Prevention, added that troubled physicians “should feel no shame for the fact that they’re in distress. Any of us can get there through a whole variety of different pathways that life presents. There’s science and data to support this experience as commonplace and having underpinnings that are of no fault to anyone. That’s the reality.”
Dr. Wible, who has lost several colleagues and physician friends to suicide, said that she hopes for a more transparent discussion of the topic by the medical profession. She presented on the topic at the 2014 annual scientific assembly of the American Academy of Family Physicians.
“The talk before mine was on Ebola, and every seat was taken” in the 900-seat room. When it came time for her presentation, “I maybe had 100 people in the room. Now, are physicians more likely to die from Ebola or from suicide? We are in a state of denial. If we don’t talk about suicide, we will continue to lose one or two medical students or doctors every day. The sooner we talk about this and connect with each other outside of a PowerPoint presentation, the sooner we’re going to solve this.”
After a physician in a large clinical department at the University of Pittsburgh took his own life several years ago, the chair of that department invited Dr. Reynolds to speak with his staff. The meeting “was primarily educational in nature, so we talked about the topic, to try to destigmatize and to educate people about the need for appropriate help-seeking,” recalled Dr. Reynolds, who is a former president of the American College of Psychiatrists. “If the leadership of a medical institution appropriately sanctions help-seeking behavior and treatment of mental disorders like depression, that’s going to make it okay for people to reach out and seek help rather than pushing it under the rug, so to speak. If the leadership says ‘this is a key thing and we don’t think you can function adequately as a medical student or as a physician if you’re not taking appropriate care of yourself,’ that helps to shift the culture.”
The ripple effect of that kind of message from health care administrators can’t be underestimated, said Dr. Moutier, who helped launch a suicide and depression awareness program at the University of California, San Diego (Acad. Med. 2012;87:320-6). She encouraged health care leaders to stage periodic grand rounds and lectures for their medical staff about physician well-being, burnout, and the risk of suicide. “If the leader is uncomfortable talking about these things, that’s a sign they should get a little education for themselves about [these topics],” she said.
Dr. Reynolds noted that certain state medical licensure boards including those for Arkansas and Pennsylvania have incorporated destigmatizing language into relicensure exams. “Some of them previously would ask questions such as whether the applicants had a history of a mental disorder like depression,” he said. “What you’re beginning to see now increasingly is that the state medical board will ask more generic questions, like ‘Do you have any conditions that would interfere with the practice of your specialty in medicine?’ This is a good thing.”
He said that he is optimistic about future of physician well-being, noting that the University of Pittsburgh and other medical schools have incorporated wellness principles into first-year curriculum. “We underscore the importance of students becoming sensitive to one another, learning how to recognize depression in each other and creating a culture in which students can encourage each other to engage in appropriate help-seeking,” Dr. Reynolds explained. “I think we are witnessing a shift in the culture of institutional medicine as we bring along new generations of physicians who are better educated about mental disorders and their treatment and issues related to suicide as we reach out to students, make counseling services available to them, educate them about these issues. That supports a cultural shift that gradually erodes the issue of stigma that has so long plagued appropriate help-seeking in medical institutions.”
Still, Dr. Wible said that she worries about the disaffected colleagues who reach out to her almost every day. “Just yesterday I got an e-mail from a physician in Oklahoma who told me they just lost three physicians to suicide in 1 month who were on probation with the medical board,” she said. “These are not defective physicians. These people need to be helped.”
Dr. Wible said that she favors holding periodic panel discussions on the topics of depression and physician suicide for medical students and physicians alike. “Let other physicians who’ve been depressed and suicidal sit in front of the room on the first week of medical school, or in a hospital once in a while, mandatory, where you listen to other well-respected physicians say, ‘yeah. I cried myself to sleep after I lost this patient,’ or ‘I had suicidal thoughts during a malpractice case.’ There are lots of reasons why physicians could be sad. They need to start talking about it publicly. Other medical students and physicians would then feel comfortable to raise their hands in the audience and say, ‘I felt the same way.’ ”
Suggested resources for help
American Foundation for Suicide Prevention (www.afsp.org/).
24-hour crisis line: 1-800-273-TALK (8255).
In 2008 the AFSP released a documentary about the problem of physician depression and suicide titled “Struggling in Silence,” which aired on public television stations nationwide and is available on DVD for $24.99.
Center for Patient and Professional Advocacy (www.mc.vanderbilt.edu/centers/cppa/index.php)
Depression and Bipolar Support Alliance (www.dbsalliance.org).
Federation of State Physician Health Programs Inc. (www.fsphp.org).
Vanderbilt Center for Professional Health (www.mc.vanderbilt.edu/cph).
The Mayo Clinic Program on Physician Well-Being (http://www.mayo.edu/research/centers-programs/physician-well-being-program/overview).
ePhysicianHealth.com, a program of the Ontario Medical Association (http://php.oma.org/ePhysicianHealth.html)
The Academic Medicine Handbook: A Guide to Achievement and Fulfillment for Academic Faculty, New York: Springer, 2013 (http://www.springer.com/medicine/internal/book/978-1-4614-5692-6)
[email protected]
On Twitter @dougbrunk
One day in 1986, a medical school classmate handed Dr. Robert P. Bright a gun that she intended to use to kill herself. She asked him to hold on to it for her and to keep quiet about her sense of hopelessness.
“She didn’t want anybody in the medical school to know; it was all hidden and hush-hushed,” recalled Dr. Bright, who is now a psychiatrist at the Mayo Clinic, Scottsdale, Ariz. “I was trying to juggle that with the issue of safety.”
He honored his classmate’s request for confidentiality, but he sought advice from the medical school dean about what to do. Before long, his classmate sought help from a psychiatrist and got better with medication and psychotherapy. “It turned out well, thank goodness,” Dr. Bright said.
Similar stories of despair among medical students and physicians don’t always end well. The American Foundation for Suicide Prevention estimates that 300-400 U.S. physicians commit suicide each year, about one per day. Suicide deaths are 250%-400% higher among female physicians, compared with women in other professions, and 70% higher among male physicians, compared with men in other professions. Major depression is a common risk factor, along with bipolar disorder and substance abuse.
Depression and other mood disorders may be underrecognized and inadequately treated in physicians because they may be reluctant to seek treatment, may attempt to diagnose and treat themselves, or may seek and receive “VIP treatment” from health care providers, according to a review article coauthored by Dr. Bright (Current Psych. 2011;10:16-30).
“Physicians struggling with these things are very much in the closet about it,” he said. “It’s a sad reflection on the stigma that’s still in our country that people can’t come forth and say, ‘I’m struggling with depression or anxiety.’ ”
Researchers led by Dr. Katherine J. Gold at the University of Michigan used data from the National Violent Death Reporting System to evaluate suicide among physicians and found that job stressors “may impact physician identity and be a particular risk factor for which more attention is warranted” (Gen. Hosp. Psychiatry 2013;35:45-9).
Work dissatisfaction sent Dr. Pamela Wible into a tailspin early in her career. In 2004 she found herself in a suicidal state for about 6 weeks, “I stayed at home, crying myself into my pillow and I never sought help from my colleagues,” recalled Dr. Wible, a family physician in Eugene, Ore., who currently leads training sessions in medical student and physician suicide prevention. “I was not depressed before entering the medical profession, but [I had developed] constant thoughts of ‘Can I just disappear? What’s the easiest way to do this?’ I got to a place of complete surrender but I didn’t have the gun. I didn’t have the stockpile of pills. I didn’t have a follow-through on the plan.”
Instead of taking her own life, she “had an epiphany” and changed the way she practiced medicine. She said that owning her own clinic empowered her to “become the doctor I had originally described on my personal statement when I entered medical school.”
According to Dr. Charles F. Reynolds III, a psychiatrist at the University of Pittsburgh, reluctance to seek treatment can also be driven by concerns about the amount of time that treatment could take.
“As physicians, we often don’t appropriately take care of ourselves when it comes to issues like depression,” said Dr. Reynolds, who also directs the National Institute of Mental Health–sponsored Center of Excellence in the Prevention and Treatment of Late Life Mood Disorders. “We may still see it as a character weakness rather than as a medical illness that can be diagnosed and appropriately treated. Concerns about privacy also figure into the concerns of some physicians as well.”
Practicing in a rural area or small community can also be an obstacle to treatment, not only because of limited access to psychiatrists, but because the “patient” may be the only physician in town.
“As much as there’s stigma for everybody being voluntarily or involuntarily admitted [for suicidal ideation], it’s a little different when you’re a provider within the hospital where you’re seeking care,” Dr. Bright noted.
He said that if he had the opportunity to counsel physicians experiencing suicidal thoughts, he would “remind them of the medical nature of depression, that the brain is just another organ and the organ is not making chemicals just like the pancreas doesn’t make insulin in diabetes,” he said. “I’d also encourage them to get the treatment that they need. I would encourage compassion for themselves that they would give to anybody else in the same situation.”
He said that he would advise them to find a mental health provider “that they trust with confidentiality, and to reach out to other people for support. I would also let them know about the physician assistance programs that are available. There’s one through Vanderbilt (the Vanderbilt Center for Professional Health) and several others that specialize in working with physicians who are struggling with mental health or substance abuse or disruptive behavior.”
Dr. Reynolds’ core message to distressed physicians is that “you’re a better doctor for your patients, and a better father or mother for your family, if you’re taking good care of yourself,” he said. “It’s hard for you to take care of your patients if you’re not also taking care of yourself, if you’re burning out. Get help. Treatment works.”
Dr. Christine Moutier, chief medical officer of the American Foundation for Suicide Prevention, added that troubled physicians “should feel no shame for the fact that they’re in distress. Any of us can get there through a whole variety of different pathways that life presents. There’s science and data to support this experience as commonplace and having underpinnings that are of no fault to anyone. That’s the reality.”
Dr. Wible, who has lost several colleagues and physician friends to suicide, said that she hopes for a more transparent discussion of the topic by the medical profession. She presented on the topic at the 2014 annual scientific assembly of the American Academy of Family Physicians.
“The talk before mine was on Ebola, and every seat was taken” in the 900-seat room. When it came time for her presentation, “I maybe had 100 people in the room. Now, are physicians more likely to die from Ebola or from suicide? We are in a state of denial. If we don’t talk about suicide, we will continue to lose one or two medical students or doctors every day. The sooner we talk about this and connect with each other outside of a PowerPoint presentation, the sooner we’re going to solve this.”
After a physician in a large clinical department at the University of Pittsburgh took his own life several years ago, the chair of that department invited Dr. Reynolds to speak with his staff. The meeting “was primarily educational in nature, so we talked about the topic, to try to destigmatize and to educate people about the need for appropriate help-seeking,” recalled Dr. Reynolds, who is a former president of the American College of Psychiatrists. “If the leadership of a medical institution appropriately sanctions help-seeking behavior and treatment of mental disorders like depression, that’s going to make it okay for people to reach out and seek help rather than pushing it under the rug, so to speak. If the leadership says ‘this is a key thing and we don’t think you can function adequately as a medical student or as a physician if you’re not taking appropriate care of yourself,’ that helps to shift the culture.”
The ripple effect of that kind of message from health care administrators can’t be underestimated, said Dr. Moutier, who helped launch a suicide and depression awareness program at the University of California, San Diego (Acad. Med. 2012;87:320-6). She encouraged health care leaders to stage periodic grand rounds and lectures for their medical staff about physician well-being, burnout, and the risk of suicide. “If the leader is uncomfortable talking about these things, that’s a sign they should get a little education for themselves about [these topics],” she said.
Dr. Reynolds noted that certain state medical licensure boards including those for Arkansas and Pennsylvania have incorporated destigmatizing language into relicensure exams. “Some of them previously would ask questions such as whether the applicants had a history of a mental disorder like depression,” he said. “What you’re beginning to see now increasingly is that the state medical board will ask more generic questions, like ‘Do you have any conditions that would interfere with the practice of your specialty in medicine?’ This is a good thing.”
He said that he is optimistic about future of physician well-being, noting that the University of Pittsburgh and other medical schools have incorporated wellness principles into first-year curriculum. “We underscore the importance of students becoming sensitive to one another, learning how to recognize depression in each other and creating a culture in which students can encourage each other to engage in appropriate help-seeking,” Dr. Reynolds explained. “I think we are witnessing a shift in the culture of institutional medicine as we bring along new generations of physicians who are better educated about mental disorders and their treatment and issues related to suicide as we reach out to students, make counseling services available to them, educate them about these issues. That supports a cultural shift that gradually erodes the issue of stigma that has so long plagued appropriate help-seeking in medical institutions.”
Still, Dr. Wible said that she worries about the disaffected colleagues who reach out to her almost every day. “Just yesterday I got an e-mail from a physician in Oklahoma who told me they just lost three physicians to suicide in 1 month who were on probation with the medical board,” she said. “These are not defective physicians. These people need to be helped.”
Dr. Wible said that she favors holding periodic panel discussions on the topics of depression and physician suicide for medical students and physicians alike. “Let other physicians who’ve been depressed and suicidal sit in front of the room on the first week of medical school, or in a hospital once in a while, mandatory, where you listen to other well-respected physicians say, ‘yeah. I cried myself to sleep after I lost this patient,’ or ‘I had suicidal thoughts during a malpractice case.’ There are lots of reasons why physicians could be sad. They need to start talking about it publicly. Other medical students and physicians would then feel comfortable to raise their hands in the audience and say, ‘I felt the same way.’ ”
Suggested resources for help
American Foundation for Suicide Prevention (www.afsp.org/).
24-hour crisis line: 1-800-273-TALK (8255).
In 2008 the AFSP released a documentary about the problem of physician depression and suicide titled “Struggling in Silence,” which aired on public television stations nationwide and is available on DVD for $24.99.
Center for Patient and Professional Advocacy (www.mc.vanderbilt.edu/centers/cppa/index.php)
Depression and Bipolar Support Alliance (www.dbsalliance.org).
Federation of State Physician Health Programs Inc. (www.fsphp.org).
Vanderbilt Center for Professional Health (www.mc.vanderbilt.edu/cph).
The Mayo Clinic Program on Physician Well-Being (http://www.mayo.edu/research/centers-programs/physician-well-being-program/overview).
ePhysicianHealth.com, a program of the Ontario Medical Association (http://php.oma.org/ePhysicianHealth.html)
The Academic Medicine Handbook: A Guide to Achievement and Fulfillment for Academic Faculty, New York: Springer, 2013 (http://www.springer.com/medicine/internal/book/978-1-4614-5692-6)
[email protected]
On Twitter @dougbrunk
Measures predict outcomes of chronic HCV with compensated cirrhosis
Readily available clinical measures can be used to reliably predict long-term outcome in patients with chronic HCV infections and well-compensated advanced liver disease, Dr. Adriaan J van der Meer, of Erasmus University Medical Center, Rotterdam, The Netherlands, and his colleagues report.
The researchers devised risk scores for mortality and for cirrhosis-related complications from a cohort of 405 patients, 100 of whom died during about 8 years of follow up. They then applied the model to 296 patients, 59 of whom died during 6 years of follow up. Independent predictive factors included age, male sex, platelet count, and aspartate aminotransferase/alanine aminotransferase ratio, the researcher said in an article published in the January issue of Gut ( Gut 2015;64:322-331).
Click here to read the article in Gut: http://gut.bmj.com/content/64/2/322.abstract
Readily available clinical measures can be used to reliably predict long-term outcome in patients with chronic HCV infections and well-compensated advanced liver disease, Dr. Adriaan J van der Meer, of Erasmus University Medical Center, Rotterdam, The Netherlands, and his colleagues report.
The researchers devised risk scores for mortality and for cirrhosis-related complications from a cohort of 405 patients, 100 of whom died during about 8 years of follow up. They then applied the model to 296 patients, 59 of whom died during 6 years of follow up. Independent predictive factors included age, male sex, platelet count, and aspartate aminotransferase/alanine aminotransferase ratio, the researcher said in an article published in the January issue of Gut ( Gut 2015;64:322-331).
Click here to read the article in Gut: http://gut.bmj.com/content/64/2/322.abstract
Readily available clinical measures can be used to reliably predict long-term outcome in patients with chronic HCV infections and well-compensated advanced liver disease, Dr. Adriaan J van der Meer, of Erasmus University Medical Center, Rotterdam, The Netherlands, and his colleagues report.
The researchers devised risk scores for mortality and for cirrhosis-related complications from a cohort of 405 patients, 100 of whom died during about 8 years of follow up. They then applied the model to 296 patients, 59 of whom died during 6 years of follow up. Independent predictive factors included age, male sex, platelet count, and aspartate aminotransferase/alanine aminotransferase ratio, the researcher said in an article published in the January issue of Gut ( Gut 2015;64:322-331).
Click here to read the article in Gut: http://gut.bmj.com/content/64/2/322.abstract
Tiny Lesion Turns Troublesome After Trauma
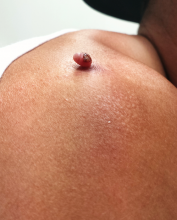
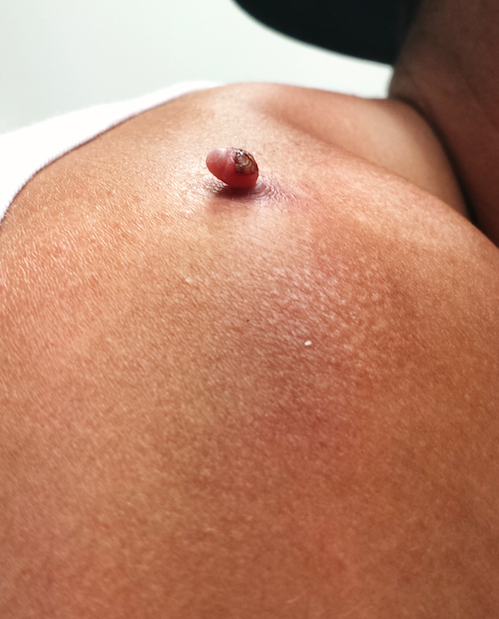
Four months ago, this 23-year-old man developed a lesion on his right shoulder. It appeared, as he recalls, over the course of a week and has subsequently grown. The lesion, which is now rather large, bleeds copiously with minor trauma. It causes only modest discomfort to the patient but considerable worry to his family.
The lesion was originally tiny and tag-like; the patient initially mistook it for a tick and tried to pull it off. Not only did that fail to work, it also seemed to irritate the lesion. At that point, it started to swell, eventually transforming into the lesion he presents with today.
The patient’s history is otherwise uneventful, and he reports taking no medications. He has had minimal sun exposure but says he tans easily when he does get some sun.
EXAMINATION
The lesion, measuring 5 mm x 2.5 mm, is a dark red and pedunculated papule on the crown of the right shoulder. It looks edematous and feels firm. The patient has otherwise unremarkable type IV skin.
Shave biopsy is performed, using a double-edged razor to make a shallow concave defect under the lesion. The wound is cauterized.
What is the diagnosis?
DISCUSSION
The pathology report confirmed the clinical suspicion of pyogenic granuloma, which, ironically, is neither pyogenic nor granulomatous. The condition acquired this name more than 100 years ago, based on assumptions about its origin. Microscopic examination revealed the highly vascular nature of these lesions, showing a field full of circles that represented the truncated ends of bundles of capillaries and venules. Although they are also called lobular capillary hemangiomas, the term pyogenic granuloma (PG) is still more commonly used.
PGs commonly manifest in the patient’s second or third decade of life, typically on extremities, chest, and nipples. This patient’s story is typical: His lesion began as a tag or wart that he then traumatized, creating a situation in which the body attempts (in vain) to heal the wound. Undisturbed, nearly all PGs would eventually wither and resolve with minimal scarring; however, that process is prolonged when the patient fails to “leave it alone.” (This is especially true in the case of young children.)
While this presentation is characteristic, there are other circumstances in which PGs develop. One is as a consequence of taking certain medications (eg, retinoids, antiretrovirals, and certain chemotherapy drugs). PGs are also commonly seen in the oral cavity of pregnant women in the third trimester and on the end of the umbilical stump in many newborns. Ingrown toenails are another common site; PGs will appear as glistening red, friable buttons of vascular tissue in the perionychial skin adjacent to the affected portion of the nail.
Shave biopsy is standard in such cases, not only to produce a cure but also to establish, via pathologic examination of the tissue, the correct diagnosis. (Nodular melanoma is the most prominent item in the differential; to miss that diagnosis would have dire consequences.) In terms of treatment, mere cautery or cryotherapy will not work and excision is seldom necessary. A deep shave will capture the entire lesion; if any remains, electrodessication and curettage will take care of it. Prior to such procedures, the patient (and family) needs to understand that scarring and pigment loss will occur.
In cases associated with medications or with ingrown toenails, the “cure” would be to withdraw the offending medications—although this is not always advisable for other, obvious reasons.
TAKE-HOME LEARNING POINTS
• Pyogenic granulomas (PG) have nothing to do with infection, nor are they truly granulomatous.
• PGs bleed readily with minor trauma and are often swollen and occasionally painful.
• PGs appear to represent, in most cases, the body’s frustrated attempt to heal a wound, most often a prick or pinch of a pre-existing lesion (eg, tag or mole).
• PGs are also seen in other contexts, such as with use of certain medications or in association with pregnancy.
• If treatment is attempted, the resulting specimen must be submitted for pathologic examination, since other lesions can mimic PGs (of most concern, nodular melanoma).
Four months ago, this 23-year-old man developed a lesion on his right shoulder. It appeared, as he recalls, over the course of a week and has subsequently grown. The lesion, which is now rather large, bleeds copiously with minor trauma. It causes only modest discomfort to the patient but considerable worry to his family.
The lesion was originally tiny and tag-like; the patient initially mistook it for a tick and tried to pull it off. Not only did that fail to work, it also seemed to irritate the lesion. At that point, it started to swell, eventually transforming into the lesion he presents with today.
The patient’s history is otherwise uneventful, and he reports taking no medications. He has had minimal sun exposure but says he tans easily when he does get some sun.
EXAMINATION
The lesion, measuring 5 mm x 2.5 mm, is a dark red and pedunculated papule on the crown of the right shoulder. It looks edematous and feels firm. The patient has otherwise unremarkable type IV skin.
Shave biopsy is performed, using a double-edged razor to make a shallow concave defect under the lesion. The wound is cauterized.
What is the diagnosis?
DISCUSSION
The pathology report confirmed the clinical suspicion of pyogenic granuloma, which, ironically, is neither pyogenic nor granulomatous. The condition acquired this name more than 100 years ago, based on assumptions about its origin. Microscopic examination revealed the highly vascular nature of these lesions, showing a field full of circles that represented the truncated ends of bundles of capillaries and venules. Although they are also called lobular capillary hemangiomas, the term pyogenic granuloma (PG) is still more commonly used.
PGs commonly manifest in the patient’s second or third decade of life, typically on extremities, chest, and nipples. This patient’s story is typical: His lesion began as a tag or wart that he then traumatized, creating a situation in which the body attempts (in vain) to heal the wound. Undisturbed, nearly all PGs would eventually wither and resolve with minimal scarring; however, that process is prolonged when the patient fails to “leave it alone.” (This is especially true in the case of young children.)
While this presentation is characteristic, there are other circumstances in which PGs develop. One is as a consequence of taking certain medications (eg, retinoids, antiretrovirals, and certain chemotherapy drugs). PGs are also commonly seen in the oral cavity of pregnant women in the third trimester and on the end of the umbilical stump in many newborns. Ingrown toenails are another common site; PGs will appear as glistening red, friable buttons of vascular tissue in the perionychial skin adjacent to the affected portion of the nail.
Shave biopsy is standard in such cases, not only to produce a cure but also to establish, via pathologic examination of the tissue, the correct diagnosis. (Nodular melanoma is the most prominent item in the differential; to miss that diagnosis would have dire consequences.) In terms of treatment, mere cautery or cryotherapy will not work and excision is seldom necessary. A deep shave will capture the entire lesion; if any remains, electrodessication and curettage will take care of it. Prior to such procedures, the patient (and family) needs to understand that scarring and pigment loss will occur.
In cases associated with medications or with ingrown toenails, the “cure” would be to withdraw the offending medications—although this is not always advisable for other, obvious reasons.
TAKE-HOME LEARNING POINTS
• Pyogenic granulomas (PG) have nothing to do with infection, nor are they truly granulomatous.
• PGs bleed readily with minor trauma and are often swollen and occasionally painful.
• PGs appear to represent, in most cases, the body’s frustrated attempt to heal a wound, most often a prick or pinch of a pre-existing lesion (eg, tag or mole).
• PGs are also seen in other contexts, such as with use of certain medications or in association with pregnancy.
• If treatment is attempted, the resulting specimen must be submitted for pathologic examination, since other lesions can mimic PGs (of most concern, nodular melanoma).
Four months ago, this 23-year-old man developed a lesion on his right shoulder. It appeared, as he recalls, over the course of a week and has subsequently grown. The lesion, which is now rather large, bleeds copiously with minor trauma. It causes only modest discomfort to the patient but considerable worry to his family.
The lesion was originally tiny and tag-like; the patient initially mistook it for a tick and tried to pull it off. Not only did that fail to work, it also seemed to irritate the lesion. At that point, it started to swell, eventually transforming into the lesion he presents with today.
The patient’s history is otherwise uneventful, and he reports taking no medications. He has had minimal sun exposure but says he tans easily when he does get some sun.
EXAMINATION
The lesion, measuring 5 mm x 2.5 mm, is a dark red and pedunculated papule on the crown of the right shoulder. It looks edematous and feels firm. The patient has otherwise unremarkable type IV skin.
Shave biopsy is performed, using a double-edged razor to make a shallow concave defect under the lesion. The wound is cauterized.
What is the diagnosis?
DISCUSSION
The pathology report confirmed the clinical suspicion of pyogenic granuloma, which, ironically, is neither pyogenic nor granulomatous. The condition acquired this name more than 100 years ago, based on assumptions about its origin. Microscopic examination revealed the highly vascular nature of these lesions, showing a field full of circles that represented the truncated ends of bundles of capillaries and venules. Although they are also called lobular capillary hemangiomas, the term pyogenic granuloma (PG) is still more commonly used.
PGs commonly manifest in the patient’s second or third decade of life, typically on extremities, chest, and nipples. This patient’s story is typical: His lesion began as a tag or wart that he then traumatized, creating a situation in which the body attempts (in vain) to heal the wound. Undisturbed, nearly all PGs would eventually wither and resolve with minimal scarring; however, that process is prolonged when the patient fails to “leave it alone.” (This is especially true in the case of young children.)
While this presentation is characteristic, there are other circumstances in which PGs develop. One is as a consequence of taking certain medications (eg, retinoids, antiretrovirals, and certain chemotherapy drugs). PGs are also commonly seen in the oral cavity of pregnant women in the third trimester and on the end of the umbilical stump in many newborns. Ingrown toenails are another common site; PGs will appear as glistening red, friable buttons of vascular tissue in the perionychial skin adjacent to the affected portion of the nail.
Shave biopsy is standard in such cases, not only to produce a cure but also to establish, via pathologic examination of the tissue, the correct diagnosis. (Nodular melanoma is the most prominent item in the differential; to miss that diagnosis would have dire consequences.) In terms of treatment, mere cautery or cryotherapy will not work and excision is seldom necessary. A deep shave will capture the entire lesion; if any remains, electrodessication and curettage will take care of it. Prior to such procedures, the patient (and family) needs to understand that scarring and pigment loss will occur.
In cases associated with medications or with ingrown toenails, the “cure” would be to withdraw the offending medications—although this is not always advisable for other, obvious reasons.
TAKE-HOME LEARNING POINTS
• Pyogenic granulomas (PG) have nothing to do with infection, nor are they truly granulomatous.
• PGs bleed readily with minor trauma and are often swollen and occasionally painful.
• PGs appear to represent, in most cases, the body’s frustrated attempt to heal a wound, most often a prick or pinch of a pre-existing lesion (eg, tag or mole).
• PGs are also seen in other contexts, such as with use of certain medications or in association with pregnancy.
• If treatment is attempted, the resulting specimen must be submitted for pathologic examination, since other lesions can mimic PGs (of most concern, nodular melanoma).
Studies shed new light on HSPC mobilization

in the bone marrow
Two new studies have revealed elements that are key to hematopoietic stem and progenitor cell (HSPC) mobilization.
In one study, investigators discovered that elevated levels of the peptide hormone angiotensin II increases HSPC mobilization in the context of vasculopathy and sickle cell disease (SCD).
In the other study, researchers found that p62, an autophagy regulator and signal organizer, is required to maintain HSPC retention in the bone marrow.
Jose Cancelas, MD, PhD, of the University of Cincinnati College of Medicine in Ohio, is the corresponding author on both studies.
In the first paper, published in Nature Communications, Dr Cancelas and his colleagues noted that patients with vasculopathies have an increase in circulating HSPCs.
“This phenomenon may represent a stress response contributing to vascular damage repair,” he said. “So the question becomes, how can we learn from these patients?”
Using mouse models of vasculopathy and vasculopathy-associated SCD, Dr Cancelas and his colleagues showed that acute and chronic elevated levels of angiotensin II resulted in an increased pool of HSPCs.
And when the researchers administered anti-angiotensin therapy, the pool of HSPCs decreased in mice and humans with SCD.
“These results indicate a new role for angiotensin in hematopoietic stem and progenitor cell trafficking under pathological conditions and define the hematopoietic consequences of anti-angiotensin therapy in vascular disease and sickle cell disease,” Dr Cancelas said.
“Every year, millions of patients receive anti-angiotensin therapies due to the harmful effects associated with chronic hyperangiotensinemia in cardiac, renal, or liver failure. Our study shows that this anti-angiotensin therapy modulates the levels of circulating stem cells and progenitors.”
In the second paper, published in Cell Reports, Dr Cancelas and his colleagues examined the role that p62 plays in HSPC mobilization.
The investigators found that, when p62 is lost in osteoblasts, mice develop a condition similar to osteoporosis in humans.
The osteoblasts cannot degrade inflammatory signals coming from macrophages. And as a consequence, the deficient osteoblasts secrete inflammatory signals that impair the retention of HSPCs in the bone marrow and allow their escape to the circulation.
Specifically, the team found that macrophages activate osteoblastic NF-kB, which results in osteopenia and HSPC egress. And p62 negatively regulates osteoblastic NF-kB activation.
Dr Cancelas noted that patients with inflammatory diseases often have osteopenia. So this research may provide insight into that phenomenon and help explain why patients with chronic inflammatory diseases have higher levels of circulating HSPCs. ![]()

in the bone marrow
Two new studies have revealed elements that are key to hematopoietic stem and progenitor cell (HSPC) mobilization.
In one study, investigators discovered that elevated levels of the peptide hormone angiotensin II increases HSPC mobilization in the context of vasculopathy and sickle cell disease (SCD).
In the other study, researchers found that p62, an autophagy regulator and signal organizer, is required to maintain HSPC retention in the bone marrow.
Jose Cancelas, MD, PhD, of the University of Cincinnati College of Medicine in Ohio, is the corresponding author on both studies.
In the first paper, published in Nature Communications, Dr Cancelas and his colleagues noted that patients with vasculopathies have an increase in circulating HSPCs.
“This phenomenon may represent a stress response contributing to vascular damage repair,” he said. “So the question becomes, how can we learn from these patients?”
Using mouse models of vasculopathy and vasculopathy-associated SCD, Dr Cancelas and his colleagues showed that acute and chronic elevated levels of angiotensin II resulted in an increased pool of HSPCs.
And when the researchers administered anti-angiotensin therapy, the pool of HSPCs decreased in mice and humans with SCD.
“These results indicate a new role for angiotensin in hematopoietic stem and progenitor cell trafficking under pathological conditions and define the hematopoietic consequences of anti-angiotensin therapy in vascular disease and sickle cell disease,” Dr Cancelas said.
“Every year, millions of patients receive anti-angiotensin therapies due to the harmful effects associated with chronic hyperangiotensinemia in cardiac, renal, or liver failure. Our study shows that this anti-angiotensin therapy modulates the levels of circulating stem cells and progenitors.”
In the second paper, published in Cell Reports, Dr Cancelas and his colleagues examined the role that p62 plays in HSPC mobilization.
The investigators found that, when p62 is lost in osteoblasts, mice develop a condition similar to osteoporosis in humans.
The osteoblasts cannot degrade inflammatory signals coming from macrophages. And as a consequence, the deficient osteoblasts secrete inflammatory signals that impair the retention of HSPCs in the bone marrow and allow their escape to the circulation.
Specifically, the team found that macrophages activate osteoblastic NF-kB, which results in osteopenia and HSPC egress. And p62 negatively regulates osteoblastic NF-kB activation.
Dr Cancelas noted that patients with inflammatory diseases often have osteopenia. So this research may provide insight into that phenomenon and help explain why patients with chronic inflammatory diseases have higher levels of circulating HSPCs. ![]()

in the bone marrow
Two new studies have revealed elements that are key to hematopoietic stem and progenitor cell (HSPC) mobilization.
In one study, investigators discovered that elevated levels of the peptide hormone angiotensin II increases HSPC mobilization in the context of vasculopathy and sickle cell disease (SCD).
In the other study, researchers found that p62, an autophagy regulator and signal organizer, is required to maintain HSPC retention in the bone marrow.
Jose Cancelas, MD, PhD, of the University of Cincinnati College of Medicine in Ohio, is the corresponding author on both studies.
In the first paper, published in Nature Communications, Dr Cancelas and his colleagues noted that patients with vasculopathies have an increase in circulating HSPCs.
“This phenomenon may represent a stress response contributing to vascular damage repair,” he said. “So the question becomes, how can we learn from these patients?”
Using mouse models of vasculopathy and vasculopathy-associated SCD, Dr Cancelas and his colleagues showed that acute and chronic elevated levels of angiotensin II resulted in an increased pool of HSPCs.
And when the researchers administered anti-angiotensin therapy, the pool of HSPCs decreased in mice and humans with SCD.
“These results indicate a new role for angiotensin in hematopoietic stem and progenitor cell trafficking under pathological conditions and define the hematopoietic consequences of anti-angiotensin therapy in vascular disease and sickle cell disease,” Dr Cancelas said.
“Every year, millions of patients receive anti-angiotensin therapies due to the harmful effects associated with chronic hyperangiotensinemia in cardiac, renal, or liver failure. Our study shows that this anti-angiotensin therapy modulates the levels of circulating stem cells and progenitors.”
In the second paper, published in Cell Reports, Dr Cancelas and his colleagues examined the role that p62 plays in HSPC mobilization.
The investigators found that, when p62 is lost in osteoblasts, mice develop a condition similar to osteoporosis in humans.
The osteoblasts cannot degrade inflammatory signals coming from macrophages. And as a consequence, the deficient osteoblasts secrete inflammatory signals that impair the retention of HSPCs in the bone marrow and allow their escape to the circulation.
Specifically, the team found that macrophages activate osteoblastic NF-kB, which results in osteopenia and HSPC egress. And p62 negatively regulates osteoblastic NF-kB activation.
Dr Cancelas noted that patients with inflammatory diseases often have osteopenia. So this research may provide insight into that phenomenon and help explain why patients with chronic inflammatory diseases have higher levels of circulating HSPCs. ![]()
EC supports continued use of ponatinib

Credit: Rhoda Baer
The European Commission (EC) has concluded that ponatinib (Iclusig) should continue to be prescribed in accordance with its already approved indications.
After trial results suggested the drug poses an increased risk of thrombotic events, the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) conducted a review of available ponatinib data.
Results of that review suggested the benefits of ponatinib outweigh the risks. So the committee said the drug should be prescribed as indicated.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recently endorsed this recommendation, and, now, the EC has followed suit. The EC’s decision is legally binding.
Ponatinib is approved in the European Union to treat adults with chronic phase, accelerated phase, or blast phase chronic myeloid leukemia who are resistant to dasatinib or nilotinib, who are intolerant to dasatinib or nilotinib and for whom subsequent treatment with imatinib is not clinically appropriate, or who have the T315I mutation.
The drug is also approved to treat adults with Philadelphia-chromosome positive acute lymphoblastic leukemia who are resistant to dasatinib, who cannot tolerate dasatinib and subsequent treatment with imatinib is not clinically appropriate, or who have the T315I mutation.
In October 2013, extended follow-up data from the PACE trial revealed that ponatinib-treated patients had a higher incidence of arterial and venous thrombotic events than was observed when the drug first gained approval. So one ponatinib trial was discontinued, and the rest were placed on partial clinical hold.
Soon after, ponatinib was pulled from the US market. The drug ultimately returned to the marketplace with new recommendations designed to decrease the risk of thrombotic events.
The EMA also revised its recommendations for ponatinib—discouraging use of the drug in certain patients, providing advice for managing comorbidities, and suggesting patient monitoring—but kept the drug on the market.
In October 2014, the PRAC concluded its 11-month review of ponatinib data, confirming that the benefit-risk profile of the drug was favorable in its approved indications and recommending that the indications remain unchanged.
However, the PRAC also said the risk of vascular occlusive events with ponatinib is likely dose-related. So the committee recommended that healthcare professionals monitor ponatinib-treated patients and consider dose reductions or discontinuing the drug in certain patients.
The CHMP endorsed these recommendations, and, now, the EC has as well. This is a legally binding decision for ponatinib to continue to be prescribed in Europe in accordance with its already approved indications.
Ponatinib is being developed by ARIAD Pharmaceuticals, Inc. ![]()

Credit: Rhoda Baer
The European Commission (EC) has concluded that ponatinib (Iclusig) should continue to be prescribed in accordance with its already approved indications.
After trial results suggested the drug poses an increased risk of thrombotic events, the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) conducted a review of available ponatinib data.
Results of that review suggested the benefits of ponatinib outweigh the risks. So the committee said the drug should be prescribed as indicated.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recently endorsed this recommendation, and, now, the EC has followed suit. The EC’s decision is legally binding.
Ponatinib is approved in the European Union to treat adults with chronic phase, accelerated phase, or blast phase chronic myeloid leukemia who are resistant to dasatinib or nilotinib, who are intolerant to dasatinib or nilotinib and for whom subsequent treatment with imatinib is not clinically appropriate, or who have the T315I mutation.
The drug is also approved to treat adults with Philadelphia-chromosome positive acute lymphoblastic leukemia who are resistant to dasatinib, who cannot tolerate dasatinib and subsequent treatment with imatinib is not clinically appropriate, or who have the T315I mutation.
In October 2013, extended follow-up data from the PACE trial revealed that ponatinib-treated patients had a higher incidence of arterial and venous thrombotic events than was observed when the drug first gained approval. So one ponatinib trial was discontinued, and the rest were placed on partial clinical hold.
Soon after, ponatinib was pulled from the US market. The drug ultimately returned to the marketplace with new recommendations designed to decrease the risk of thrombotic events.
The EMA also revised its recommendations for ponatinib—discouraging use of the drug in certain patients, providing advice for managing comorbidities, and suggesting patient monitoring—but kept the drug on the market.
In October 2014, the PRAC concluded its 11-month review of ponatinib data, confirming that the benefit-risk profile of the drug was favorable in its approved indications and recommending that the indications remain unchanged.
However, the PRAC also said the risk of vascular occlusive events with ponatinib is likely dose-related. So the committee recommended that healthcare professionals monitor ponatinib-treated patients and consider dose reductions or discontinuing the drug in certain patients.
The CHMP endorsed these recommendations, and, now, the EC has as well. This is a legally binding decision for ponatinib to continue to be prescribed in Europe in accordance with its already approved indications.
Ponatinib is being developed by ARIAD Pharmaceuticals, Inc. ![]()

Credit: Rhoda Baer
The European Commission (EC) has concluded that ponatinib (Iclusig) should continue to be prescribed in accordance with its already approved indications.
After trial results suggested the drug poses an increased risk of thrombotic events, the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) conducted a review of available ponatinib data.
Results of that review suggested the benefits of ponatinib outweigh the risks. So the committee said the drug should be prescribed as indicated.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recently endorsed this recommendation, and, now, the EC has followed suit. The EC’s decision is legally binding.
Ponatinib is approved in the European Union to treat adults with chronic phase, accelerated phase, or blast phase chronic myeloid leukemia who are resistant to dasatinib or nilotinib, who are intolerant to dasatinib or nilotinib and for whom subsequent treatment with imatinib is not clinically appropriate, or who have the T315I mutation.
The drug is also approved to treat adults with Philadelphia-chromosome positive acute lymphoblastic leukemia who are resistant to dasatinib, who cannot tolerate dasatinib and subsequent treatment with imatinib is not clinically appropriate, or who have the T315I mutation.
In October 2013, extended follow-up data from the PACE trial revealed that ponatinib-treated patients had a higher incidence of arterial and venous thrombotic events than was observed when the drug first gained approval. So one ponatinib trial was discontinued, and the rest were placed on partial clinical hold.
Soon after, ponatinib was pulled from the US market. The drug ultimately returned to the marketplace with new recommendations designed to decrease the risk of thrombotic events.
The EMA also revised its recommendations for ponatinib—discouraging use of the drug in certain patients, providing advice for managing comorbidities, and suggesting patient monitoring—but kept the drug on the market.
In October 2014, the PRAC concluded its 11-month review of ponatinib data, confirming that the benefit-risk profile of the drug was favorable in its approved indications and recommending that the indications remain unchanged.
However, the PRAC also said the risk of vascular occlusive events with ponatinib is likely dose-related. So the committee recommended that healthcare professionals monitor ponatinib-treated patients and consider dose reductions or discontinuing the drug in certain patients.
The CHMP endorsed these recommendations, and, now, the EC has as well. This is a legally binding decision for ponatinib to continue to be prescribed in Europe in accordance with its already approved indications.
Ponatinib is being developed by ARIAD Pharmaceuticals, Inc. ![]()
Combining bed nets and vaccines may worsen malaria risk

Credit: Caitlin Kleiboer
New research suggests that combining the use of malaria vaccines and insecticide-treated bed nets may actually increase the incidence of malaria.
The researchers used a mathematical model of malaria transmission to examine potential interactions between vaccines and bed nets.
They found that using insecticide-treated bed nets along with pre-erythrocytic vaccines (PEVs) or blood-stage vaccines (BSVs) increased the number of malaria cases.
However, using bed nets in conjunction with transmission-blocking vaccines (TBVs) resulted in fewer cases of malaria and increased the probability of eliminating the disease.
“The joint use of bed nets and vaccines will not always lead to consistent increases in the efficacy of malaria control,” said study author Mercedes Pascual, PhD, of the University of Michigan in Ann Arbor.
“Specifically, our study suggests that the combined use of some malaria vaccines with bed nets can lead to increased morbidity and mortality in older age classes.”
Dr Pascual and her colleagues described this research in Proceedings of the National Academy of Sciences.
The team noted that the malaria vaccine candidates currently under development fall into 3 categories, each focusing on a different stage of the malaria life cycle.
PEVs aim to reduce the chances that a person will be infected when bitten by a disease-carrying mosquito. BSVs don’t block infection but try to reduce the level of disease severity and the number of fatalities.
And TBVs don’t protect vaccinated individuals against infection or illness, but they prevent mosquitoes from spreading the disease to others after biting a vaccinated person.
Dr Pascual and her colleagues found that using bed nets in communities treated with BSVs can increase levels of morbidity—to levels even higher than those expected in the absence of nets. Furthermore, BSVs can’t promote malaria elimination on their own.
PEVs can promote malaria elimination, but the researchers found regions of decreased morbidity when PEV vaccination levels were low and increased morbidity when PEV vaccination levels were high.
This suggests that higher levels of PEV coverage and bed net use could result in malaria elimination, but it would involve crossing a peak of enhanced morbidity.
Finally, the researchers found that using bed nets in communities treated with TBVs always leads to significant decreases in morbidity and increases the probability of malaria elimination.
“Ironically, the vaccines that work best with bed nets are the ones that do not protect the vaccinated host—the bed net does that—but instead block transmission of malaria in mosquitoes that have found an opportunity to bite vaccinated hosts,” said study author Yael Artzy-Randrup, PhD, of the University of Amsterdam in The Netherlands.
Unraveling the interactions between bed nets and vaccines is especially challenging due to the complex and transient nature of malaria immunity, the researchers noted.
A child’s first malaria infection can result in severe, sometimes fatal, illness. If the child survives, he or she will gain partial immunity that reduces the risk of severe illness in the future.
Additional bites from infected mosquitoes can help the child retain that immunity, which would otherwise wane after 1 to 2 years. But the combination of bed nets and certain vaccines can undermine that natural immunity.
“This complexity is at the heart of why it has been so hard to develop any sort of malaria vaccine,” said study author Andrew Dobson, DPhil, of Princeton University in New Jersey. ![]()

Credit: Caitlin Kleiboer
New research suggests that combining the use of malaria vaccines and insecticide-treated bed nets may actually increase the incidence of malaria.
The researchers used a mathematical model of malaria transmission to examine potential interactions between vaccines and bed nets.
They found that using insecticide-treated bed nets along with pre-erythrocytic vaccines (PEVs) or blood-stage vaccines (BSVs) increased the number of malaria cases.
However, using bed nets in conjunction with transmission-blocking vaccines (TBVs) resulted in fewer cases of malaria and increased the probability of eliminating the disease.
“The joint use of bed nets and vaccines will not always lead to consistent increases in the efficacy of malaria control,” said study author Mercedes Pascual, PhD, of the University of Michigan in Ann Arbor.
“Specifically, our study suggests that the combined use of some malaria vaccines with bed nets can lead to increased morbidity and mortality in older age classes.”
Dr Pascual and her colleagues described this research in Proceedings of the National Academy of Sciences.
The team noted that the malaria vaccine candidates currently under development fall into 3 categories, each focusing on a different stage of the malaria life cycle.
PEVs aim to reduce the chances that a person will be infected when bitten by a disease-carrying mosquito. BSVs don’t block infection but try to reduce the level of disease severity and the number of fatalities.
And TBVs don’t protect vaccinated individuals against infection or illness, but they prevent mosquitoes from spreading the disease to others after biting a vaccinated person.
Dr Pascual and her colleagues found that using bed nets in communities treated with BSVs can increase levels of morbidity—to levels even higher than those expected in the absence of nets. Furthermore, BSVs can’t promote malaria elimination on their own.
PEVs can promote malaria elimination, but the researchers found regions of decreased morbidity when PEV vaccination levels were low and increased morbidity when PEV vaccination levels were high.
This suggests that higher levels of PEV coverage and bed net use could result in malaria elimination, but it would involve crossing a peak of enhanced morbidity.
Finally, the researchers found that using bed nets in communities treated with TBVs always leads to significant decreases in morbidity and increases the probability of malaria elimination.
“Ironically, the vaccines that work best with bed nets are the ones that do not protect the vaccinated host—the bed net does that—but instead block transmission of malaria in mosquitoes that have found an opportunity to bite vaccinated hosts,” said study author Yael Artzy-Randrup, PhD, of the University of Amsterdam in The Netherlands.
Unraveling the interactions between bed nets and vaccines is especially challenging due to the complex and transient nature of malaria immunity, the researchers noted.
A child’s first malaria infection can result in severe, sometimes fatal, illness. If the child survives, he or she will gain partial immunity that reduces the risk of severe illness in the future.
Additional bites from infected mosquitoes can help the child retain that immunity, which would otherwise wane after 1 to 2 years. But the combination of bed nets and certain vaccines can undermine that natural immunity.
“This complexity is at the heart of why it has been so hard to develop any sort of malaria vaccine,” said study author Andrew Dobson, DPhil, of Princeton University in New Jersey. ![]()

Credit: Caitlin Kleiboer
New research suggests that combining the use of malaria vaccines and insecticide-treated bed nets may actually increase the incidence of malaria.
The researchers used a mathematical model of malaria transmission to examine potential interactions between vaccines and bed nets.
They found that using insecticide-treated bed nets along with pre-erythrocytic vaccines (PEVs) or blood-stage vaccines (BSVs) increased the number of malaria cases.
However, using bed nets in conjunction with transmission-blocking vaccines (TBVs) resulted in fewer cases of malaria and increased the probability of eliminating the disease.
“The joint use of bed nets and vaccines will not always lead to consistent increases in the efficacy of malaria control,” said study author Mercedes Pascual, PhD, of the University of Michigan in Ann Arbor.
“Specifically, our study suggests that the combined use of some malaria vaccines with bed nets can lead to increased morbidity and mortality in older age classes.”
Dr Pascual and her colleagues described this research in Proceedings of the National Academy of Sciences.
The team noted that the malaria vaccine candidates currently under development fall into 3 categories, each focusing on a different stage of the malaria life cycle.
PEVs aim to reduce the chances that a person will be infected when bitten by a disease-carrying mosquito. BSVs don’t block infection but try to reduce the level of disease severity and the number of fatalities.
And TBVs don’t protect vaccinated individuals against infection or illness, but they prevent mosquitoes from spreading the disease to others after biting a vaccinated person.
Dr Pascual and her colleagues found that using bed nets in communities treated with BSVs can increase levels of morbidity—to levels even higher than those expected in the absence of nets. Furthermore, BSVs can’t promote malaria elimination on their own.
PEVs can promote malaria elimination, but the researchers found regions of decreased morbidity when PEV vaccination levels were low and increased morbidity when PEV vaccination levels were high.
This suggests that higher levels of PEV coverage and bed net use could result in malaria elimination, but it would involve crossing a peak of enhanced morbidity.
Finally, the researchers found that using bed nets in communities treated with TBVs always leads to significant decreases in morbidity and increases the probability of malaria elimination.
“Ironically, the vaccines that work best with bed nets are the ones that do not protect the vaccinated host—the bed net does that—but instead block transmission of malaria in mosquitoes that have found an opportunity to bite vaccinated hosts,” said study author Yael Artzy-Randrup, PhD, of the University of Amsterdam in The Netherlands.
Unraveling the interactions between bed nets and vaccines is especially challenging due to the complex and transient nature of malaria immunity, the researchers noted.
A child’s first malaria infection can result in severe, sometimes fatal, illness. If the child survives, he or she will gain partial immunity that reduces the risk of severe illness in the future.
Additional bites from infected mosquitoes can help the child retain that immunity, which would otherwise wane after 1 to 2 years. But the combination of bed nets and certain vaccines can undermine that natural immunity.
“This complexity is at the heart of why it has been so hard to develop any sort of malaria vaccine,” said study author Andrew Dobson, DPhil, of Princeton University in New Jersey. ![]()
Sofosbuvir and ribavirin effective in transplant patients with compensated recurrent HCV
Patients who develop HCV infections after liver transplant may respond to a 24-week course of sofosbuvir and ribavirin, Dr. Michael Charlton, of the Mayo Clinic in Rochester, Minn., and his colleagues reported.
The researchers enrolled and treated 40 liver transplant patients with compensated recurrent HCV infection of any genotype; 83% had HCV genotype 1, 40% had cirrhosis (based on biopsy), and 88% had been previously treated with interferon. All patients received 24 weeks of sofosbuvir 400 mg daily and ribavirin starting at 400 mg daily, which was adjusted according to creatinine clearance and hemoglobin values, the researchers said in the January 2015 issue of Gastroenterology.
After 12 weeks, 28 of 40 had a sustained virologic response (70%; 90% confidence interval: 56%−82%). Relapse accounted for all cases of virologic failure. No patients had detectable viral resistance during or after treatment.
Click here to read the study: http://www.ncbi.nlm.nih.gov/pubmed/25304641
Patients who develop HCV infections after liver transplant may respond to a 24-week course of sofosbuvir and ribavirin, Dr. Michael Charlton, of the Mayo Clinic in Rochester, Minn., and his colleagues reported.
The researchers enrolled and treated 40 liver transplant patients with compensated recurrent HCV infection of any genotype; 83% had HCV genotype 1, 40% had cirrhosis (based on biopsy), and 88% had been previously treated with interferon. All patients received 24 weeks of sofosbuvir 400 mg daily and ribavirin starting at 400 mg daily, which was adjusted according to creatinine clearance and hemoglobin values, the researchers said in the January 2015 issue of Gastroenterology.
After 12 weeks, 28 of 40 had a sustained virologic response (70%; 90% confidence interval: 56%−82%). Relapse accounted for all cases of virologic failure. No patients had detectable viral resistance during or after treatment.
Click here to read the study: http://www.ncbi.nlm.nih.gov/pubmed/25304641
Patients who develop HCV infections after liver transplant may respond to a 24-week course of sofosbuvir and ribavirin, Dr. Michael Charlton, of the Mayo Clinic in Rochester, Minn., and his colleagues reported.
The researchers enrolled and treated 40 liver transplant patients with compensated recurrent HCV infection of any genotype; 83% had HCV genotype 1, 40% had cirrhosis (based on biopsy), and 88% had been previously treated with interferon. All patients received 24 weeks of sofosbuvir 400 mg daily and ribavirin starting at 400 mg daily, which was adjusted according to creatinine clearance and hemoglobin values, the researchers said in the January 2015 issue of Gastroenterology.
After 12 weeks, 28 of 40 had a sustained virologic response (70%; 90% confidence interval: 56%−82%). Relapse accounted for all cases of virologic failure. No patients had detectable viral resistance during or after treatment.
Click here to read the study: http://www.ncbi.nlm.nih.gov/pubmed/25304641
Sofosbuvir and ribavirin prevent HCV recurrence after liver transplantation
Sofosbuvir and ribavirin given before liver transplantation prevented most cases of post-transplant HCV recurrence, according to Dr. Michael P. Curry, of Beth Israel Deaconess Medical Center, Boston, and his colleagues.
Up to 48 weeks of sofosbuvir (400 mg) and ribavirin were given to hepatocellular carcinoma patients on organ transplant waitlists. The patients had HCV of any genotype and cirrhosis (Child–Turcotte–Pugh score of 7 or less). The primary end point of the study (ClinicalTrials.gov: NCT01559844) was the proportion of 43 patients who had HCV-RNA levels of less than 25 IU/ml at transplant and at 12 weeks after transplant.
Of the 43 patients, 30 (70%) had a post-transplantation virologic response at 12 weeks, 10 (23%) had recurrent infection, and 3 (7%) died, the researchers reported in the January issue of Gastroenterology.
Click here to read the entire article: http://www.gastrojournal.org/article/S0016-5085%2814%2901145-7/fulltext
Sofosbuvir and ribavirin given before liver transplantation prevented most cases of post-transplant HCV recurrence, according to Dr. Michael P. Curry, of Beth Israel Deaconess Medical Center, Boston, and his colleagues.
Up to 48 weeks of sofosbuvir (400 mg) and ribavirin were given to hepatocellular carcinoma patients on organ transplant waitlists. The patients had HCV of any genotype and cirrhosis (Child–Turcotte–Pugh score of 7 or less). The primary end point of the study (ClinicalTrials.gov: NCT01559844) was the proportion of 43 patients who had HCV-RNA levels of less than 25 IU/ml at transplant and at 12 weeks after transplant.
Of the 43 patients, 30 (70%) had a post-transplantation virologic response at 12 weeks, 10 (23%) had recurrent infection, and 3 (7%) died, the researchers reported in the January issue of Gastroenterology.
Click here to read the entire article: http://www.gastrojournal.org/article/S0016-5085%2814%2901145-7/fulltext
Sofosbuvir and ribavirin given before liver transplantation prevented most cases of post-transplant HCV recurrence, according to Dr. Michael P. Curry, of Beth Israel Deaconess Medical Center, Boston, and his colleagues.
Up to 48 weeks of sofosbuvir (400 mg) and ribavirin were given to hepatocellular carcinoma patients on organ transplant waitlists. The patients had HCV of any genotype and cirrhosis (Child–Turcotte–Pugh score of 7 or less). The primary end point of the study (ClinicalTrials.gov: NCT01559844) was the proportion of 43 patients who had HCV-RNA levels of less than 25 IU/ml at transplant and at 12 weeks after transplant.
Of the 43 patients, 30 (70%) had a post-transplantation virologic response at 12 weeks, 10 (23%) had recurrent infection, and 3 (7%) died, the researchers reported in the January issue of Gastroenterology.
Click here to read the entire article: http://www.gastrojournal.org/article/S0016-5085%2814%2901145-7/fulltext
Residents looking to work in larger cities
Since 2008, residents’ preference for their future practice location has shifted from smaller cities and rural areas to large population centers, according to findings reported by physician recruitment firm Merritt Hawkins.
In a survey of residents who completed their training in 2014, 24% said that they wanted to practice in a community with a population of more than 1 million, compared with 6% in 2008, while 23% of residents chose the next-highest level of population – 500,001 to 1 million – compared with 15% in 2008, according to Merritt Hawkins.
As for the smaller communities, residents who wanted to practice in a area of 50,000 or fewer dropped from 17% in 2008 to 7% in 2014. Support for communities of 50,001-100,000 fell from 19% in 2008 to 10% in 2014, the company said. Only 1% of residents wanted to practice in a community of 10,000 people or fewer in 2014.
Residents’ reservations about practicing in rural areas more often are related to their “concerns about being on a clinical ‘island’ without specialty support, information technology, and other resources than they may be about the amenities of rural communities,” Merritt Hawkins said in its analysis of the 1,208 survey responses.
Since 2008, residents’ preference for their future practice location has shifted from smaller cities and rural areas to large population centers, according to findings reported by physician recruitment firm Merritt Hawkins.
In a survey of residents who completed their training in 2014, 24% said that they wanted to practice in a community with a population of more than 1 million, compared with 6% in 2008, while 23% of residents chose the next-highest level of population – 500,001 to 1 million – compared with 15% in 2008, according to Merritt Hawkins.

As for the smaller communities, residents who wanted to practice in a area of 50,000 or fewer dropped from 17% in 2008 to 7% in 2014. Support for communities of 50,001-100,000 fell from 19% in 2008 to 10% in 2014, the company said. Only 1% of residents wanted to practice in a community of 10,000 people or fewer in 2014.
Residents’ reservations about practicing in rural areas more often are related to their “concerns about being on a clinical ‘island’ without specialty support, information technology, and other resources than they may be about the amenities of rural communities,” Merritt Hawkins said in its analysis of the 1,208 survey responses.
Since 2008, residents’ preference for their future practice location has shifted from smaller cities and rural areas to large population centers, according to findings reported by physician recruitment firm Merritt Hawkins.
In a survey of residents who completed their training in 2014, 24% said that they wanted to practice in a community with a population of more than 1 million, compared with 6% in 2008, while 23% of residents chose the next-highest level of population – 500,001 to 1 million – compared with 15% in 2008, according to Merritt Hawkins.

As for the smaller communities, residents who wanted to practice in a area of 50,000 or fewer dropped from 17% in 2008 to 7% in 2014. Support for communities of 50,001-100,000 fell from 19% in 2008 to 10% in 2014, the company said. Only 1% of residents wanted to practice in a community of 10,000 people or fewer in 2014.
Residents’ reservations about practicing in rural areas more often are related to their “concerns about being on a clinical ‘island’ without specialty support, information technology, and other resources than they may be about the amenities of rural communities,” Merritt Hawkins said in its analysis of the 1,208 survey responses.
Coexisting Frailty, Cognitive Impairment, and Heart Failure: Implications for Clinical Care
From the Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA.
Abstract
- Objective: To review some of the proposed pathways that increase frailty risk in older persons with heart failure and to discuss tools that may be used to assess for changes in physical and cognitive functioning in this population in order to assist with appropriate and timely intervention.
- Methods: Review of the literature.
- Results: Heart failure is the only cardiovascular disease that is increasing by epidemic proportions, largely due to an aging society and therapeutic advances in disease management. Because heart failure is largely a cardiogeriatric syndrome, age-related syndromes such as frailty and cognitive impairment are common in heart failure patients. Compared with age-matched counterparts, older adults with heart failure 4 to 6 times more likely to be frail or cognitively impaired. The reason for the high prevalence of frailty and cognitive impairment in this population is not well known but may likely reflect the synergistic effects of heart failure and aging, which may heighten vulnerability to stressors and accelerate loss of physiologic reserve. Despite the high prevalence of frailty and cognitive impairment in the heart failure population, these conditions are not routinely screened for in clinical practice settings and guidelines on optimal assessment strategies are lacking.
- Conclusion: Persons with heart failure are at an increased risk for frailty, which may worsen symptoms, impair self-management, and lead to worse heart failure outcomes. Early detection of frailty and cognitive impairment may be an opportunity for intervention and a key strategy for improving clinical outcomes in older adults with heart failure.
Approximately 5.7 million persons in the United States are diagnosed with heart failure, and the number of reported new cases is expected to increase to over 700,000 cases annually by the year 2040 [1]. This rising incidence is fueled by an aging population; by the year 2030, 1 in 5 Americans will be over 65 years of age [2]. Heart failure is prevalent among those 65 years of age and older and is the most common reason for hospitalization in this age-group. High readmission rates, approaching 50% over 6 months, are a major contributor to the the escalating economic burden associated with heart failure [3].
Persons with heart failure are more likely to be frail and experience cognitive impairment than their age-matched counterparts without heart failure. The reasons for this are not well known but may be related to hemodynamic, vascular, and inflammatory changes occurring as heart failure progresses. In this paper, we review the link between frailty and cognitive impairment in heart failure, instruments that may be useful for early detection, and interventions such as exercise that may be beneficial for attenuating both conditions.
Frailty in Heart Failure
Epidemiology
Frailty is a heightened vulnerability to stressors in the presence of low physiological reserve [4]. When exposed to stressors, persons who are frail have a much higher probability for disproportionate decompensation, negative events, functional decline, disability, and mortality [5]. Among persons with heart failure, frailty may predispose them to decompensate at a lower threshold, requiring more frequent hospitalizations. Persons with heart failure are more likely to be frail than their age-matched counterparts without heart failure [6,7].
Frailty is a powerful predictor of poor clinical outcomes and mortality in cardiovascular disease [8,9]. Compared with the non-frail, frail persons with heart failure have increased rates of mortality (16.9% vs 4.8%) and increased rates of heart failure hospitalization (20.5% vs 13.3%) [10]. Frailty has also been shown to predict falls, disability, and hospitalization in heart failure patients [6,9,11] and was found to have a negative linear relationship with health-related quality of life [12]. Frail heart failure patients are also more likely to have comorbidities such diabetes mellitus, chronic obstructive pulmonary disease, atrial fibrillation, depression, anemia, and chronic kidney disease [9,13].
Pathophysiology
There is significant overlap in the underlying pathological mechanisms of heart failure and frailty. Symptoms of heart failure, such as dyspnea, fatigue, and muscle loss, mirror components that occur with frailty. Further, cardiac cachexia, a metabolic syndrome in advanced heart failure characterized by a loss of muscle mass, is very similar to the sarcopenia that occurs in frailty.
Frailty, characterized by an increased physiologic vulnerability to stressors, may predispose frail persons with heart failure to exacerbation and worsening of heart failure due to greater susceptibility to the harmful pathophysiologic processes in heart failure, such as inflammation and autonomic dysfunction. Proposed pathophysiologic pathways in frailty include free radicals and oxidative stress, cumulative DNA damage, decreased telomere length, and nuclear fragmentation [14,15]. Frailty has been associated with low-grade chronic inflammation and increased inflammatory cytokines, such as C-reactive protein, tumor necrosis factor–alpha (TNFα), interleukin-6 (IL-6)and fibrinogen [16–18]. Heart failure also is associated with a low-grade and chronic cardiac inflammatory response that is correlated with disease progression [19].
Inflammation. IL-6 is detectable in a higher proportion of persons who are frail compared to non-frail [16] and is the most highly correlated biomarker with frailty. In addition, among those with detectable IL-6 levels, those categorized as frail have higher IL-6 levels compared to those who are non-frail [16,20]. Individuals categorized as frail were found to have significantly higher levels of TNFα than those who were non-frail [16,20]. Increased IL-6 levels are associated with decreased muscle strength, while increased TNFα levels are associated with decreased skeletal muscle protein synthesis [21,22], thus contributing to frailty.
Oxidative stress. Protein carbonyls result from protein oxidation promoted by reactive oxygen species and are markers of oxidative stress. Protein carbonylation is implicated in the pathogenesis of the loss of skeletal muscle mass; high serum protein carbonyls are associated with poor grip strength [23]. 8-OHdG is an oxidized nucleoside indicative of oxidative damage to DNA and a measure of oxidative stress. Accumulation of 8-OHdG in skeletal muscle leads to loss of muscle mass and is associated with decreased hand grip strength in the elderly [24]. Higher serum levels of 8-OHdG are present in older adults who are frail as compared to those who are non-frail [25].
Measurement of Frailty in the Clinical Setting
Frailty has been conceptualized in a number of studies using different models and measures; however, there continues to be a lack of consensus on the definition and operationalization of frailty. Prior research has led to the development of several validated models of frailty that have demonstrated good prediction of adverse outcomes in older adults. Some models, such as the Fried phenotype [6], focus solely on the physical dimension, while other models take a multidimensional approach.Single-item measures (eg, gait speed, 6-minute walk test, handgrip strength) are also commonly used to screen for frailty, but a frailty measure that incorporates more than 1 physical dimension may be more sensitive and reliable. In our opinion, the ideal measure of frailty would consist of a brief assessment that can be serially performed in most clinical practice settings that can identify change in function over time. The incorporation of sensitive physical function measures that can detect frailty early has the potential to slow physical function decline by preserving physiological thresholds.
Cognitive Impairment in Heart Failure
Epidemiology
Cognitive impairment occurs frequently in patients with heart failure, and the presence of cognitive impairment in persons with heart failure has been shown to heighten risk for adverse clinical outcomes, disability, poor quality of life, and mortality [26,27]. Heart failure negatively influences cognitive functioning in most domains [28–32]. The most common domains adversely affected by heart failure and aging are memory and executive function. Deficits in these domains can substantially diminish patient ability to carry out essential self-care behaviors [30,32].
The most common form of cognitive impairment seen in patients with heart failure is mild cognitive impairment (MCI), which is a measurable deficit with memory or another core cognitive domain. Up to 60% of persons with heart failure have been reported to have MCI. Patients with MCI have cognitive deficits that are more pronounced than those seen in normal aging, but lack other symptoms of dementia, such as impaired judgment or reasoning. MCI often will not impede patients’ ability to carry out the activities of daily living (ADLs) independently, but patients may have difficulty in performing some instrumental activities of daily living (IADLs), such as remembering medications, scheduling provider appointments. Dementia, a decline in cognitive ability severe enough to hinder an individual’s ability to perform ADLs or IADLs or engage in social activities or occupational responsibilities, occurs in approximately 25% of persons with heart failure [33].
Persons with heart failure have a fourfold greater likelihood of developing CI than persons without heart failure. Several cohort studies have shown that persons with heart failure had lower performance on cognitive tests than individuals without heart failure [34,35] and were 50% more likely to progress to dementia.
Assessment Tools
Although a comprehensive neurocognitive battery would aid in detecting cognitive impairment in heart failure, few clinical practice settings have the resources to perform such a detailed and time-consuming measurement. Most studies in heart failure have relied on global screening questionnaires such as the Mini-Mental State Examination (MMSE) [36] to assess cognitive functioning in persons with heart failure and in other cardiovascular disorders. Global cognitive measures, however, often lack sensitivity for detecting subtle cognitive deficits such as seen in MCI [28–30]. Screening that measures executive function may be the most beneficial for busy clinical settings, since declines in this domain are well established as contributing to poor outcomes in persons with heart failure.
The Montreal Cognitive Assessment (MoCA) is a rapid screening test designed to detect MCI. It assesses different cognitive domains, including attention, memory, language, and executive function [37]. The MoCA lends itself to use in clinical setting because it is brief, requires little training to administer, and is easy to score. This instrument has been used successfully to assess MCI in persons with heart failure and may be more sensitive than the MMSE in identifying clinically relevant cognitive dysfunction. In 2013 study, Cameron et al [38] administered the MMSE and MoCA to 93 hospitalized heart failure patients and found that the MoCA classified 41% of patients as cognitively impaired that were not classified using the MMSE. For persons with a vascular cognitive deficit, the MMSE has been portrayed as an inadequate screening test due to lack of sensitivity for visuospatial and executive function deficits. Because the MoCA was designed to be more sensitive to such deficits, it may be a superior screening method for persons with heart failure. Although previous studies support the use of the MoCA in persons with heart failure, more research is needed in larger, more diverse heart failure samples with a wide range of cognitive deficits.
A Reasonable Clinical Assessment Approach
Considering the link between heart failure, frailty, and MCI, incorporating simple physical performance measures with cognitive screening may be an effective strategy to identify persons at risk for frailty. Two clinically relevant physical performance-based measures of frailty are proposed: the Fried phenotype (mentioned earlier) and the Short Physical Performance Battery (SBBP). In addition, cognitive screening using the MoCA is recommended as part of the routine examination for determining possible MCI or more severe cognitive deficits. The predictive validity of measuring physical frailty is enhanced when cognitive impairment is included in the assessment [36,39].
The performance-based measures included in this review have previously demonstrated excellent psychometric properties as well as sensitivity for change that is clinically meaningful. Minimal detectable change (MDC), a threshold score that refers to the minimal amount of change outside of error that reflects true change by a patient between 2 time points (rather than variation in measurement), is important for interpreting level of risk for frailty and is included for each instrument [40,41]. If a more brief frailty examination is needed, cut-points for gait speed and handgrip have been used effectively in a number of studies as a threshold for determining frailty, including in older patients with cardiovascular disease and in heart failure [8,42,43].
Fried Frailty Phenotype
The Fried phenotype is an appropriate method of measuring frailty in a clinical setting due to its wide application across diverse populations and consistent identification of adverse outcomes [44]. This model is derived from a frailty model proposed by Fried et al [6] in which a phenotypic cycle exists that includes disease, sarcopenia, decreased walking speed, chronic undernutrition, decreased total energy expenditure, senescent musculoskeletal changes, decreased resting metabolic rate, weight loss and decreased maximal oxygen consumption. Frailty exists when a critical mass of these cycle components are identified in an individual [6].
To validate the model, Fried et al used data from the Cardiovascular Health Study and used the model to show association with a 3-year and 7-year incidence of mobility and ADL disability among 4317 community-dwelling men and women aged 65 years and older, independent of comorbidities. Several studies have directly tested the frailty phenotype model alone and in comparison to other models of frailty in large prospective studies across different populations, such as the Survey of Health, Aging and Retirement in Europe (SHARE) [45], the European Male Aging Study [46], and the Canadian Health Study of Aging [47]. While these studies found the prevalence of frailty to vary across the populations, they all validated the Fried model and found no significant differences in the predictive ability of the Fried model and other models of frailty. The Frailty Consensus conference evaluated the different models of frailty and determined that the Fried model is a validated construct of frailty and is acceptable for use in the identification of individuals who are frail or likely to become frail [48]. Thus, the Fried et al frailty phenotype model is considered to be a standard measure of frailty in older individuals.
Short Physical Performance Battery
In other chronic illness populations, the SPPB has also been used as a predictor of outcomes before, during, or after hospitalization. Valpato et al [53], for example, used the SPPB to assess older adults (mean age, 78 yr) admitted to the hospital with a diagnosis of heart failure (64%), pneumonia (13%), chronic obstructive pulmonary disease (16%), or minor stroke (6.6%) at admission (baseline) and discharge. Patients with the lowest SPPB quartile scores at hospital discharge had a fivefold greater risk of rehospitalization or mortality compared to the highest quartile. In addition, those who had an early decline in SPPB scores 1 month after hospital discharge had greater limitations in performing activities of daily living and a significantly greater probability of being re-hospitalized or death during the 1-year follow-up period. These studies suggest that the SPPB at the first follow-up outpatient visit following hospital discharge may be beneficial for identifying need for further intervention or the need for more frequent follow-up care. Although the SPPB is not part of the Fried et al phenotype, it may provide additional information concerning risk for falls and lower extremity strength that may be beneficial in the evaluation of some persons with heart failure [54]. The SPPB along with instructions and normative data are available for clinical use at no charge at www.grc.nia.nih.gov/branches/ledb/sppb/index.htm.
Interventions for Frailty in Heart Failure
Interventions to address frailty have included exercise training, comprehensive geriatric assessment and management services, social support systems, nutrition, and drugs; however, few intervention studies have examined frailty in heart failure [8]. Restoration of physical function through aerobic exercise and resistance training has shown benefit in frail older adults [55–57] and in persons with heart failure [58]. Maintaining and/or restoring physical function through aerobic and resistance exercise training may be the key to preventing further decline or potentially reversing frailty in older adults with heart failure.
Aerobic exercise has been shown to be beneficial for both frail older adults and frail persons with heart failure [18]. In a study of community-dwelling frail older adults aged 65 and older, a combined aerobic and resistance exercise intervention, performed over 16 weeks, demonstrated significant improvement in frailty scores during the 1-year follow-up in contrast to worsening frailty measures in the control group [57].
Older adults with heart failure experience a much lower exercise tolerance largely due to a 50% to 75% decrease in aerobic capacity in addition to the well-known alterations in peripheral musculoskeletal performance that contribute to fatigue and greater symptom severity. Aerobic exercise has been shown to be beneficial for most heart failure patients by altering the peripheral and central mechanisms, such as inflammatory cytokines, that contribute to heart failure exacerbations, worsened symptom severity, and poor clinical outcomes [59–62].Lower rates of hospitalization, improved physical function, and enhanced health-related quality of life are reported in heart failure patients who routinely exercise [59]. Resistance training has been shown to improve physical function in frail older adults [55]. Further, the use of TheraBand exercise bands in resistance training demonstrated improvement in physical function among frail older adults [56].
Exercise also appears to exert a positive effect on cognition, particularly executive functioning, and may also have a protective effect against cognitive decline with aging and among those with heart failure. The underlying mechanism for improvement in cognition remains poorly understood but is likely related to improved cardiac function, cerebral perfusion, and oxygenation, although this has not been clearly established. Larson et al (2006) evaluated the frequency of participation in a variety of physical activities (eg, walking, bicycling and swimming) over 6 years in 1740 older adults [63]. Older adults who exercised more than 3 times per week during initial assessment were 34% less likely to be diagnosed with dementia than those who exercised fewer than 3 times per week. Several meta-analyses in recent years have shown a consistent and positive relationship between aerobic exercise and cognition [64,65]. Importantly, findings from meta-analyses have shown a moderate effect size (> 0.5) from aerobic training, which was similar for normal and cognitively impaired adults [64].
Implications for Clinical Care
A systematic assessment performed periodically using physical and cognitive measures that may identify prefrailty may be the best strategy for preventing further functional loss, limitations, and disability in persons with heart failure. Persons with heart failure ideally should be evaluated annually for physical function, since a decline has been consistently shown to be a strong predictor of adverse health outcomes, disability, and death [6,66]. Cognitive function should also be assessed routinely in persons with heart failure, particularly when first diagnosed, when changes in treatment regimen occur, and with worsening disease severity, since these events have been shown to occur before changes in cognition [31]. Incorporating geriatric performance-based measures in heart failure management would allow for more treatment strategies aimed at improving physical function, cognitive outcomes, and quality of life. Further, identifying frailty in heart failure is an important component of clinical decision-making when determining if a patient can tolerate therapies such as implantable defibrillators, cardiac resynchronization therapy, or left ventricular assist device placement.
In older adults, performance measures are well established and commonly used as part of geriatric assessment to evaluate physical and cognitive functioning. Performance-based measures may be particularly beneficial in older adults with heart failure to monitor serial changes in physical function. Performance measures in clinical settings require staff time but little training, space, equipment, or risk. As performance measures become more common in practice settings, MDC thresholds may need to be re-evaluated based on the characteristics of the population [67].
For persons with heart failure whose screening outcomes suggest MCI, more comprehensive neuropsychological testing should be available as well as provision of resources to optimize functional independence. Early identification of impaired cognition may lower risk of poor self-management through simplification of medication regimens or providing resources to help manage other regimens essential for optimal heart failure care. It is also important to recognize that depressive symptoms are common in persons with heart failure and are highly correlated with cognitive impairment in this population. Screening for depressive symptoms therefore, may also enhance identification of persons with heart failure at risk for frailty [4,28].
Conclusion
Effective appraisal and development of effective interventions are essential in older adults with heart failure who are at high risk for frailty and cognitive impairment. This will become increasingly important as the population ages and the incidence of heart failure rises proportionately. Although curative treatments for frailty and cognitive impairment are not available, interdisciplinary interventions such as exercise and comprehensive geriatric assessment may improve outcomes in older persons with heart failure [68]. Information gained from objective, simple, inexpensive physical performance measures, when used in combination with cognitive screening, may enhance the ability to evaluate change that signal onset of frailty or cognitive impairment [54,69,70]. The high morbidity and mortality associated with frailty and cognitive impairment indicate that it should be a priority for future research as a strategy to improve clinical outcomes, enhance quality of life, and lower health care costs in this growing population.
Corresponding author: Rebecca Gary, PhD, RN, Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA 30322, [email protected].
Funding/support: B. Butts was partially funded for this work through National Institutes of Health/National
Institute of Nursing Research Grant #T32NR012715.
1. Velagaleti RS and Vasan RS. Heart failure in the twenty-first century: is it a coronary artery disease or hypertension problem. Cardiol Clin 2007;25:487–95.
2. Vincent GK, Velkoff VA. The next four decades. The older population in the United States: 2010 to 2050. United States Census Bureau Report No: P25-1138. U.S. Department of Commerce; May 2010.
3. Butler J, Kalogeropoulos A. Worsening heart failure hospitalization epidemic we do not know how to prevent and we do not know how to treat. J Am Coll Cardiol 2008;52:435–7.
4. Gary R. Evaluation of frailty in older adults with cardiovascular disease: incorporating physical performance measures. J Cardiovasc Nurs 2012;27:120–131.
5. Shamliyan T, Talley KM, Ramakrishnan R, Kane RL. Association of frailty with survival: a systematic literature review. Age Res Rev 2013;12:719–36.
6. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol Med Sci 2001; 56:M146–M156.
7. Newman AB, Gottdiener JS, Mcburnie MA, et al. Associations of subclinical cardiovascular disease with frailty. J Gerontol A Biol Sci Med Sci 2001;56:M158–66.
8. Afilalo J, Karunananthan S, Eisenberg MJ, et al. Role of frailty in patients with cardiovascular disease. Am J Cardiol 2009; 103:1616–21.
9. Cacciatore F, Abete P, Maella F, et al. Frailty predicts long-term mortality in elderly subjects with chronic heart failure. Eur J Clin Invest 2008;35:723–30.
10. Lupón J, González B, Santaeugenia S, et al. Prognostic implication of frailty and depressive symptoms in an outpatient population with heart failure. Rev Españ Cardiol 2008;61:835–42.
11. Rich MW. Heart failure in the oldest patients: the impact of comorbid conditions. Am J Geriatr Cardiol 2007;14:134–41.
12. Buck HG, Riegel B. The impact of frailty on health related quality of life in heart failure. Eur J Cardiovasc Nurs 2011;10:159–66
13. Boxer RS, Shah KB Kenny AM. Frailty and prognosis in advanced heart failure. Curr Opin Supp Pall Care 2014;8:25–9.
14. Afilalo J, Sebag IA, Chalifour LE, et al. Age-related changes in lamin A/C expression in cardiomyocytes. Am J Physiol Heart Circ Physiol 2007;293:H1451–6.
15. Walston J. Frailty—the search for underlying causes. Sci Aging Know Environ 2004;2004:e4.
16. Hubbard RE, O’Mahony MS, Savva GM, et al. Inflammation and frailty measures in older people. J Cell Mol Med 2009; 13:3103–9.
17. Hubbard RE Woodhouse KW. Frailty, inflammation and the elderly. Biogerontol 2010;11:635–41.
18. Baptista G, Dupuy A-M, Jaussent A, et al. Low-grade chronic inflammation and superoxide anion production by NADPH oxidase are the main determinants of physical frailty in older adults. Free Rad Res 2012;46:1108–14.
19. Abbate A. The heart on fire: Inflammasome and cardiomyopathy. Exper Physiol 2013;98:385.
20. Collerton J, Martin-Ruiz C, Davies K, et al. Frailty and the role of inflammation, immunosenescence and cellular ageing in the very old: Cross-sectional findings from the Newcastle 85+ Study. Mech Age Devel 2012;133:456–66.
21. Ferrucci L, Harris TB, Guralnik JM, et al. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc 1999;47:639–46.
22. Toth MJ, Matthews DE, Tracy RP and Previs MJ. Age-related differences in skeletal muscle protein synthesis: relation to markers of immune activation. Am j Physiol Endocrin Metab 2005;288:E883–91.
23. Howard C, Ferrucci L, Sun K, et al. Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol 2007;103:17–20.
24. Muzembo BA, Nagano Y, Eitoku M, et al. A cross-sectional assessment of oxidative DNA damage and muscle strength among elderly people living in the community. Envir Health Prev Med 2014;19:21–9.
25. Wu I-C, Shiesh S-C, Kuo P-H and Lin X-Z. High oxidative stress is correlated with frailty in elderly Chinese. J Am Geriatr Soc 2009;57:1666–71.
26. Alosco ML, Spitznagel MB, Cohen R, et al. Cognitive impairment is independently associated with reduced instrumental activities of daily living in persons with heart failure. J Cardiovasc Nurs 2012;27:44–50.
27. Feola M, Rosso GL, Peano M, et al. Correlation between cognitive impairment and prognostic parameters in patients with congestive heart failure. Arch Med Res 2007;38:234–9.
28. Pressler SJ, Subramanian U, Kareken D, et al. Cognitive deficits in chronic heart failure. Nurs Res 2010;59:127–39.
29. Pressler SJ, Kim J, Riley P, et al. Memory dysfunction, psychomotor slowing, and decreased executive function predict mortality in patients with heart failure and low ejection fraction. J Cardiac Fail 2010;16:750–60.
30. Pressler SJ, Subramanian U, Kareken D, et al. Cognitive deficits and health-related quality of life in chronic heart failure. J Cardiovasc Nurs 2010;25:189–98.
31. Hajduk AM, Lemon SC, Mcmanus DD, et al. Cognitive impairment and self-care in heart failure. Clin Epidemiol 2013; 24:407–16.
32. Dardiotis E, Giamouzis G, Mastrogiannis D, et al. Cognitive impairment in heart failure. Cardiol Res Prac 2012; 2012:595821.
33. Petersen RC and O’brien J. Mild cognitive impairment should be considered for DSM-V. J Geriatr Psychiatry Neurol 2006; 19:147–54.
34. Hjelm C, Dahl A, Broström A, et al. The influence of heart failure on longitudinal changes in cognition among individuals 80 years of age and older. J Clin Nurs 2012; 21:994–1003.
35. Almeida OP, Garrido GJ, Beer C, et al. Cognitive and brain changes associated with ischaemic heart disease and heart failure. Eur Heart J 2012;33:1769–76.
36. Folstein MF, Folstein SE, McHugh PR. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189–98.
37. Nasveddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005;53:695–699.
38. Cameron J, Worrall-Carter L, Page K, et al. Screening for mild cognitive impairment in patients with heart failure: Montreal cognitive assessment versus mini mental state exam. Eur J Cardiovasc Nurs 2013;12:252–60.
39. Avila-Funes JA, Amieva H, Barberger-Gateau P, et al. Cognitive impairment improves the predictive validity of the phenotype of frailty for adverse health outcomes: the three-city study. J Am Geriatr Soc 57:453–61.
40. Perera S, Mody SH, Woodman RC, Studenski SA. Meaningful change and responsiveness in common physical performance measures in older adults. Am Geriatr Soc 2006;54:743–9.
41. Kwon S, Perera S, Pahor M, et al. What is a meaningful change in physical performance? Findings from a clinical trial in older adults (the LIFE-P study). J Nutr Health Aging 2009;13:538–44.
42. Abellan Van Kan G, Rolland Y, Houles M, et al. The assessment of frailty in older adults. Clin Geriatr Med 2010; 26:275–86.
43. Pahor M, Manini T, Cesari M. Sarcopenia: clinical evaluation, biological markers and other evaluation tools. J Nutr Health Aging 2009;13:724–8.
44. Gary R. Evaluation of frailty in older adults with cardiovascular disease. J Cardiovasc Nurs 2012;27:120–31.
45. Macklai NS, Spagnoli J, Junod J, Santos-Eggimann B. Prospective association of the SHARE-operationalized frailty phenotype with adverse health outcomes: evidence from 620+ community-dwelling Europeans living in 11 countries. BMC Geriatr 2013;13:1–9.
46. Ravinrarajah R, Lee DM, Pye SR, et al. The ability of three different models of frailty to predict all-cause mortality: Results from the European Male Aging Study (EMAS). Arch Gerontol Geriatr 2013;57:360–8.
47. Rockwood K, Andrew M and Mitnitski A. A comparison of two approaches to measuring frailty in elderly people. J Gerontol Med Sci 2007;62:738–43.
48. Morley JE, Vellas B, Van Kan GA, et al. Frailty consensus: a call to action. J Am Med Dir Assoc 2013;14:392–7.
49. Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol 1994;49:M85–94.
50. Guralnik JM, Ferrucci L, Simonsick EM, et al. Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N Engl J Med 1995;332:556–61.
51. Di Bari M, Pozzi C, Cavallini Mc, et al. The diagnosis of heart failure in the community. Comparative validation of four sets of criteria in unselected older adults: the ICARe Dicomano Study. J Am Coll Cardiol 2004;44:1601–08.
52. Chiarantini D, Volpato S, Sioulis F, et al. Lower extremity performance measures predict long-term prognosis in older patients hospitalized for heart failure. J Cardiac Failure 2010; 16:390–5.
53. Volpato S, Cavalieri M, Sioulis F, et al. Predictive value of the short physical performance battery following hospitalization in older patients. J Gerontol A Biol Sci Med Sci 2011;66:89–96.
54. Studenski S, Perera S, Wallace D, et al. Physical performance measures in the clinical setting. J Am Geriatr Soc 2003; 51:314–22.
55. Binder EF, Schechtman KB, Ehsani AA, et al. Effects of exercise training on frailty in community-dwelling older adults: results of a randomized, controlled trial. J Am Geriatr Soc 2012; 50:1921–8.
56. Brown M, Sinacore DR, Ehsani AA, et al. Low-intensity exercise as a modifier of physical frailty in older adults. Arch Phys Med Rehab 2000;81:960–5.
57. Yamada M, Arai H, Sonoda T and Aoyama T. Community-based exercise program is cost-effective by preventing care and disability in Japanese frail older adults. J Am Med Dir Assoc 2012;13:507–11.
58. Gary RA, Cress ME, Higgins MK, et al. A combined aerobic and resistance exercise program improves physical functional performance in patients with heart failure: a pilot study. J Cardiovasc Nurs 2012;27:418–30.
59. De Meirelles L, Matsuura C, Resende AD, et al. Chronic exercise leads to antiaggregant, antioxidant and anti-inflammatory effects in heart failure patients. Eur J Prev Cardiol 2014;21:1225–32.
60. Feiereisen P, Vaillant M, Gilson G, Delagardelle C. Effects of different training modalities on circulating anabolic/catabolic markers in chronic heart failure. J Cardiopulm Rehab Prev 2013;33:303–8.
61. Smart NA, Steele M. The effect of physical training on systemic proinflammatory cytokine expression in heart failure patients: a systematic review. Congest Heart Fail 2011;17:110–4.
62. Nunes RB, Alves JP, Kessler LP, Lago PD. Aerobic exercsie improves the inflammatory profile correlated with cardiac remodeling and function in chronic heart failure rats. Clin Chest Med 2013;68:876–82.
63. Larson EB, Wang L, Bowen JD, et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann Intern Med 2006;144:73–8.
64. Colcombe S and Kramer AF. Fitness effects on the cognitive function of older adults: a meta-analytic study. Psychol Sci 2003;14:125–30.
65. Heyn P, Abreu BC, Ottenbacher KJ. The effects of exercise training on elderly persons with cognitive impairment and dementia: a meta-analysis. Arch Phys Med Rehab 2004; 85:1694–704.
66. Bautmans I, Vanpuyvelde K, Mets T. Sarcopenia and functional decline: pathophysiology, prevention and therapy. Acta Clinica Belgica 2009;64:303–16.
67. Guralnik JM, Ferrucci L, Pieper CF, et al. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J Gerontol A Biol Sci Med Sci 2000;55A:M221–M231.
68. Kramer AF, Erickson KI. Capitalizing on cortical plasticity: influence of physical activity on cognition and brain function. Trends Cogn Sci 2007;11:342–8.
69. Harkness K, Heckman GA, Mckelvie RS. The older patient with heart failure: high risk for frailty and cognitive impairment. Expert Rev Cardiovasc Ther 2012;10:779–95.
70. Waters DL, Baumgartner RN, Garry PJ, Vellas B. Advantages of dietary, exercise-related, and therapeutic interventions to prevent and treat sarcopenia in adult patients: an update. Clin Interv Aging 2010;5:259–70.
From the Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA.
Abstract
- Objective: To review some of the proposed pathways that increase frailty risk in older persons with heart failure and to discuss tools that may be used to assess for changes in physical and cognitive functioning in this population in order to assist with appropriate and timely intervention.
- Methods: Review of the literature.
- Results: Heart failure is the only cardiovascular disease that is increasing by epidemic proportions, largely due to an aging society and therapeutic advances in disease management. Because heart failure is largely a cardiogeriatric syndrome, age-related syndromes such as frailty and cognitive impairment are common in heart failure patients. Compared with age-matched counterparts, older adults with heart failure 4 to 6 times more likely to be frail or cognitively impaired. The reason for the high prevalence of frailty and cognitive impairment in this population is not well known but may likely reflect the synergistic effects of heart failure and aging, which may heighten vulnerability to stressors and accelerate loss of physiologic reserve. Despite the high prevalence of frailty and cognitive impairment in the heart failure population, these conditions are not routinely screened for in clinical practice settings and guidelines on optimal assessment strategies are lacking.
- Conclusion: Persons with heart failure are at an increased risk for frailty, which may worsen symptoms, impair self-management, and lead to worse heart failure outcomes. Early detection of frailty and cognitive impairment may be an opportunity for intervention and a key strategy for improving clinical outcomes in older adults with heart failure.
Approximately 5.7 million persons in the United States are diagnosed with heart failure, and the number of reported new cases is expected to increase to over 700,000 cases annually by the year 2040 [1]. This rising incidence is fueled by an aging population; by the year 2030, 1 in 5 Americans will be over 65 years of age [2]. Heart failure is prevalent among those 65 years of age and older and is the most common reason for hospitalization in this age-group. High readmission rates, approaching 50% over 6 months, are a major contributor to the the escalating economic burden associated with heart failure [3].
Persons with heart failure are more likely to be frail and experience cognitive impairment than their age-matched counterparts without heart failure. The reasons for this are not well known but may be related to hemodynamic, vascular, and inflammatory changes occurring as heart failure progresses. In this paper, we review the link between frailty and cognitive impairment in heart failure, instruments that may be useful for early detection, and interventions such as exercise that may be beneficial for attenuating both conditions.
Frailty in Heart Failure
Epidemiology
Frailty is a heightened vulnerability to stressors in the presence of low physiological reserve [4]. When exposed to stressors, persons who are frail have a much higher probability for disproportionate decompensation, negative events, functional decline, disability, and mortality [5]. Among persons with heart failure, frailty may predispose them to decompensate at a lower threshold, requiring more frequent hospitalizations. Persons with heart failure are more likely to be frail than their age-matched counterparts without heart failure [6,7].
Frailty is a powerful predictor of poor clinical outcomes and mortality in cardiovascular disease [8,9]. Compared with the non-frail, frail persons with heart failure have increased rates of mortality (16.9% vs 4.8%) and increased rates of heart failure hospitalization (20.5% vs 13.3%) [10]. Frailty has also been shown to predict falls, disability, and hospitalization in heart failure patients [6,9,11] and was found to have a negative linear relationship with health-related quality of life [12]. Frail heart failure patients are also more likely to have comorbidities such diabetes mellitus, chronic obstructive pulmonary disease, atrial fibrillation, depression, anemia, and chronic kidney disease [9,13].
Pathophysiology
There is significant overlap in the underlying pathological mechanisms of heart failure and frailty. Symptoms of heart failure, such as dyspnea, fatigue, and muscle loss, mirror components that occur with frailty. Further, cardiac cachexia, a metabolic syndrome in advanced heart failure characterized by a loss of muscle mass, is very similar to the sarcopenia that occurs in frailty.
Frailty, characterized by an increased physiologic vulnerability to stressors, may predispose frail persons with heart failure to exacerbation and worsening of heart failure due to greater susceptibility to the harmful pathophysiologic processes in heart failure, such as inflammation and autonomic dysfunction. Proposed pathophysiologic pathways in frailty include free radicals and oxidative stress, cumulative DNA damage, decreased telomere length, and nuclear fragmentation [14,15]. Frailty has been associated with low-grade chronic inflammation and increased inflammatory cytokines, such as C-reactive protein, tumor necrosis factor–alpha (TNFα), interleukin-6 (IL-6)and fibrinogen [16–18]. Heart failure also is associated with a low-grade and chronic cardiac inflammatory response that is correlated with disease progression [19].
Inflammation. IL-6 is detectable in a higher proportion of persons who are frail compared to non-frail [16] and is the most highly correlated biomarker with frailty. In addition, among those with detectable IL-6 levels, those categorized as frail have higher IL-6 levels compared to those who are non-frail [16,20]. Individuals categorized as frail were found to have significantly higher levels of TNFα than those who were non-frail [16,20]. Increased IL-6 levels are associated with decreased muscle strength, while increased TNFα levels are associated with decreased skeletal muscle protein synthesis [21,22], thus contributing to frailty.
Oxidative stress. Protein carbonyls result from protein oxidation promoted by reactive oxygen species and are markers of oxidative stress. Protein carbonylation is implicated in the pathogenesis of the loss of skeletal muscle mass; high serum protein carbonyls are associated with poor grip strength [23]. 8-OHdG is an oxidized nucleoside indicative of oxidative damage to DNA and a measure of oxidative stress. Accumulation of 8-OHdG in skeletal muscle leads to loss of muscle mass and is associated with decreased hand grip strength in the elderly [24]. Higher serum levels of 8-OHdG are present in older adults who are frail as compared to those who are non-frail [25].
Measurement of Frailty in the Clinical Setting
Frailty has been conceptualized in a number of studies using different models and measures; however, there continues to be a lack of consensus on the definition and operationalization of frailty. Prior research has led to the development of several validated models of frailty that have demonstrated good prediction of adverse outcomes in older adults. Some models, such as the Fried phenotype [6], focus solely on the physical dimension, while other models take a multidimensional approach.Single-item measures (eg, gait speed, 6-minute walk test, handgrip strength) are also commonly used to screen for frailty, but a frailty measure that incorporates more than 1 physical dimension may be more sensitive and reliable. In our opinion, the ideal measure of frailty would consist of a brief assessment that can be serially performed in most clinical practice settings that can identify change in function over time. The incorporation of sensitive physical function measures that can detect frailty early has the potential to slow physical function decline by preserving physiological thresholds.
Cognitive Impairment in Heart Failure
Epidemiology
Cognitive impairment occurs frequently in patients with heart failure, and the presence of cognitive impairment in persons with heart failure has been shown to heighten risk for adverse clinical outcomes, disability, poor quality of life, and mortality [26,27]. Heart failure negatively influences cognitive functioning in most domains [28–32]. The most common domains adversely affected by heart failure and aging are memory and executive function. Deficits in these domains can substantially diminish patient ability to carry out essential self-care behaviors [30,32].
The most common form of cognitive impairment seen in patients with heart failure is mild cognitive impairment (MCI), which is a measurable deficit with memory or another core cognitive domain. Up to 60% of persons with heart failure have been reported to have MCI. Patients with MCI have cognitive deficits that are more pronounced than those seen in normal aging, but lack other symptoms of dementia, such as impaired judgment or reasoning. MCI often will not impede patients’ ability to carry out the activities of daily living (ADLs) independently, but patients may have difficulty in performing some instrumental activities of daily living (IADLs), such as remembering medications, scheduling provider appointments. Dementia, a decline in cognitive ability severe enough to hinder an individual’s ability to perform ADLs or IADLs or engage in social activities or occupational responsibilities, occurs in approximately 25% of persons with heart failure [33].
Persons with heart failure have a fourfold greater likelihood of developing CI than persons without heart failure. Several cohort studies have shown that persons with heart failure had lower performance on cognitive tests than individuals without heart failure [34,35] and were 50% more likely to progress to dementia.
Assessment Tools
Although a comprehensive neurocognitive battery would aid in detecting cognitive impairment in heart failure, few clinical practice settings have the resources to perform such a detailed and time-consuming measurement. Most studies in heart failure have relied on global screening questionnaires such as the Mini-Mental State Examination (MMSE) [36] to assess cognitive functioning in persons with heart failure and in other cardiovascular disorders. Global cognitive measures, however, often lack sensitivity for detecting subtle cognitive deficits such as seen in MCI [28–30]. Screening that measures executive function may be the most beneficial for busy clinical settings, since declines in this domain are well established as contributing to poor outcomes in persons with heart failure.
The Montreal Cognitive Assessment (MoCA) is a rapid screening test designed to detect MCI. It assesses different cognitive domains, including attention, memory, language, and executive function [37]. The MoCA lends itself to use in clinical setting because it is brief, requires little training to administer, and is easy to score. This instrument has been used successfully to assess MCI in persons with heart failure and may be more sensitive than the MMSE in identifying clinically relevant cognitive dysfunction. In 2013 study, Cameron et al [38] administered the MMSE and MoCA to 93 hospitalized heart failure patients and found that the MoCA classified 41% of patients as cognitively impaired that were not classified using the MMSE. For persons with a vascular cognitive deficit, the MMSE has been portrayed as an inadequate screening test due to lack of sensitivity for visuospatial and executive function deficits. Because the MoCA was designed to be more sensitive to such deficits, it may be a superior screening method for persons with heart failure. Although previous studies support the use of the MoCA in persons with heart failure, more research is needed in larger, more diverse heart failure samples with a wide range of cognitive deficits.
A Reasonable Clinical Assessment Approach
Considering the link between heart failure, frailty, and MCI, incorporating simple physical performance measures with cognitive screening may be an effective strategy to identify persons at risk for frailty. Two clinically relevant physical performance-based measures of frailty are proposed: the Fried phenotype (mentioned earlier) and the Short Physical Performance Battery (SBBP). In addition, cognitive screening using the MoCA is recommended as part of the routine examination for determining possible MCI or more severe cognitive deficits. The predictive validity of measuring physical frailty is enhanced when cognitive impairment is included in the assessment [36,39].
The performance-based measures included in this review have previously demonstrated excellent psychometric properties as well as sensitivity for change that is clinically meaningful. Minimal detectable change (MDC), a threshold score that refers to the minimal amount of change outside of error that reflects true change by a patient between 2 time points (rather than variation in measurement), is important for interpreting level of risk for frailty and is included for each instrument [40,41]. If a more brief frailty examination is needed, cut-points for gait speed and handgrip have been used effectively in a number of studies as a threshold for determining frailty, including in older patients with cardiovascular disease and in heart failure [8,42,43].
Fried Frailty Phenotype
The Fried phenotype is an appropriate method of measuring frailty in a clinical setting due to its wide application across diverse populations and consistent identification of adverse outcomes [44]. This model is derived from a frailty model proposed by Fried et al [6] in which a phenotypic cycle exists that includes disease, sarcopenia, decreased walking speed, chronic undernutrition, decreased total energy expenditure, senescent musculoskeletal changes, decreased resting metabolic rate, weight loss and decreased maximal oxygen consumption. Frailty exists when a critical mass of these cycle components are identified in an individual [6].
To validate the model, Fried et al used data from the Cardiovascular Health Study and used the model to show association with a 3-year and 7-year incidence of mobility and ADL disability among 4317 community-dwelling men and women aged 65 years and older, independent of comorbidities. Several studies have directly tested the frailty phenotype model alone and in comparison to other models of frailty in large prospective studies across different populations, such as the Survey of Health, Aging and Retirement in Europe (SHARE) [45], the European Male Aging Study [46], and the Canadian Health Study of Aging [47]. While these studies found the prevalence of frailty to vary across the populations, they all validated the Fried model and found no significant differences in the predictive ability of the Fried model and other models of frailty. The Frailty Consensus conference evaluated the different models of frailty and determined that the Fried model is a validated construct of frailty and is acceptable for use in the identification of individuals who are frail or likely to become frail [48]. Thus, the Fried et al frailty phenotype model is considered to be a standard measure of frailty in older individuals.
Short Physical Performance Battery
In other chronic illness populations, the SPPB has also been used as a predictor of outcomes before, during, or after hospitalization. Valpato et al [53], for example, used the SPPB to assess older adults (mean age, 78 yr) admitted to the hospital with a diagnosis of heart failure (64%), pneumonia (13%), chronic obstructive pulmonary disease (16%), or minor stroke (6.6%) at admission (baseline) and discharge. Patients with the lowest SPPB quartile scores at hospital discharge had a fivefold greater risk of rehospitalization or mortality compared to the highest quartile. In addition, those who had an early decline in SPPB scores 1 month after hospital discharge had greater limitations in performing activities of daily living and a significantly greater probability of being re-hospitalized or death during the 1-year follow-up period. These studies suggest that the SPPB at the first follow-up outpatient visit following hospital discharge may be beneficial for identifying need for further intervention or the need for more frequent follow-up care. Although the SPPB is not part of the Fried et al phenotype, it may provide additional information concerning risk for falls and lower extremity strength that may be beneficial in the evaluation of some persons with heart failure [54]. The SPPB along with instructions and normative data are available for clinical use at no charge at www.grc.nia.nih.gov/branches/ledb/sppb/index.htm.
Interventions for Frailty in Heart Failure
Interventions to address frailty have included exercise training, comprehensive geriatric assessment and management services, social support systems, nutrition, and drugs; however, few intervention studies have examined frailty in heart failure [8]. Restoration of physical function through aerobic exercise and resistance training has shown benefit in frail older adults [55–57] and in persons with heart failure [58]. Maintaining and/or restoring physical function through aerobic and resistance exercise training may be the key to preventing further decline or potentially reversing frailty in older adults with heart failure.
Aerobic exercise has been shown to be beneficial for both frail older adults and frail persons with heart failure [18]. In a study of community-dwelling frail older adults aged 65 and older, a combined aerobic and resistance exercise intervention, performed over 16 weeks, demonstrated significant improvement in frailty scores during the 1-year follow-up in contrast to worsening frailty measures in the control group [57].
Older adults with heart failure experience a much lower exercise tolerance largely due to a 50% to 75% decrease in aerobic capacity in addition to the well-known alterations in peripheral musculoskeletal performance that contribute to fatigue and greater symptom severity. Aerobic exercise has been shown to be beneficial for most heart failure patients by altering the peripheral and central mechanisms, such as inflammatory cytokines, that contribute to heart failure exacerbations, worsened symptom severity, and poor clinical outcomes [59–62].Lower rates of hospitalization, improved physical function, and enhanced health-related quality of life are reported in heart failure patients who routinely exercise [59]. Resistance training has been shown to improve physical function in frail older adults [55]. Further, the use of TheraBand exercise bands in resistance training demonstrated improvement in physical function among frail older adults [56].
Exercise also appears to exert a positive effect on cognition, particularly executive functioning, and may also have a protective effect against cognitive decline with aging and among those with heart failure. The underlying mechanism for improvement in cognition remains poorly understood but is likely related to improved cardiac function, cerebral perfusion, and oxygenation, although this has not been clearly established. Larson et al (2006) evaluated the frequency of participation in a variety of physical activities (eg, walking, bicycling and swimming) over 6 years in 1740 older adults [63]. Older adults who exercised more than 3 times per week during initial assessment were 34% less likely to be diagnosed with dementia than those who exercised fewer than 3 times per week. Several meta-analyses in recent years have shown a consistent and positive relationship between aerobic exercise and cognition [64,65]. Importantly, findings from meta-analyses have shown a moderate effect size (> 0.5) from aerobic training, which was similar for normal and cognitively impaired adults [64].
Implications for Clinical Care
A systematic assessment performed periodically using physical and cognitive measures that may identify prefrailty may be the best strategy for preventing further functional loss, limitations, and disability in persons with heart failure. Persons with heart failure ideally should be evaluated annually for physical function, since a decline has been consistently shown to be a strong predictor of adverse health outcomes, disability, and death [6,66]. Cognitive function should also be assessed routinely in persons with heart failure, particularly when first diagnosed, when changes in treatment regimen occur, and with worsening disease severity, since these events have been shown to occur before changes in cognition [31]. Incorporating geriatric performance-based measures in heart failure management would allow for more treatment strategies aimed at improving physical function, cognitive outcomes, and quality of life. Further, identifying frailty in heart failure is an important component of clinical decision-making when determining if a patient can tolerate therapies such as implantable defibrillators, cardiac resynchronization therapy, or left ventricular assist device placement.
In older adults, performance measures are well established and commonly used as part of geriatric assessment to evaluate physical and cognitive functioning. Performance-based measures may be particularly beneficial in older adults with heart failure to monitor serial changes in physical function. Performance measures in clinical settings require staff time but little training, space, equipment, or risk. As performance measures become more common in practice settings, MDC thresholds may need to be re-evaluated based on the characteristics of the population [67].
For persons with heart failure whose screening outcomes suggest MCI, more comprehensive neuropsychological testing should be available as well as provision of resources to optimize functional independence. Early identification of impaired cognition may lower risk of poor self-management through simplification of medication regimens or providing resources to help manage other regimens essential for optimal heart failure care. It is also important to recognize that depressive symptoms are common in persons with heart failure and are highly correlated with cognitive impairment in this population. Screening for depressive symptoms therefore, may also enhance identification of persons with heart failure at risk for frailty [4,28].
Conclusion
Effective appraisal and development of effective interventions are essential in older adults with heart failure who are at high risk for frailty and cognitive impairment. This will become increasingly important as the population ages and the incidence of heart failure rises proportionately. Although curative treatments for frailty and cognitive impairment are not available, interdisciplinary interventions such as exercise and comprehensive geriatric assessment may improve outcomes in older persons with heart failure [68]. Information gained from objective, simple, inexpensive physical performance measures, when used in combination with cognitive screening, may enhance the ability to evaluate change that signal onset of frailty or cognitive impairment [54,69,70]. The high morbidity and mortality associated with frailty and cognitive impairment indicate that it should be a priority for future research as a strategy to improve clinical outcomes, enhance quality of life, and lower health care costs in this growing population.
Corresponding author: Rebecca Gary, PhD, RN, Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA 30322, [email protected].
Funding/support: B. Butts was partially funded for this work through National Institutes of Health/National
Institute of Nursing Research Grant #T32NR012715.
From the Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA.
Abstract
- Objective: To review some of the proposed pathways that increase frailty risk in older persons with heart failure and to discuss tools that may be used to assess for changes in physical and cognitive functioning in this population in order to assist with appropriate and timely intervention.
- Methods: Review of the literature.
- Results: Heart failure is the only cardiovascular disease that is increasing by epidemic proportions, largely due to an aging society and therapeutic advances in disease management. Because heart failure is largely a cardiogeriatric syndrome, age-related syndromes such as frailty and cognitive impairment are common in heart failure patients. Compared with age-matched counterparts, older adults with heart failure 4 to 6 times more likely to be frail or cognitively impaired. The reason for the high prevalence of frailty and cognitive impairment in this population is not well known but may likely reflect the synergistic effects of heart failure and aging, which may heighten vulnerability to stressors and accelerate loss of physiologic reserve. Despite the high prevalence of frailty and cognitive impairment in the heart failure population, these conditions are not routinely screened for in clinical practice settings and guidelines on optimal assessment strategies are lacking.
- Conclusion: Persons with heart failure are at an increased risk for frailty, which may worsen symptoms, impair self-management, and lead to worse heart failure outcomes. Early detection of frailty and cognitive impairment may be an opportunity for intervention and a key strategy for improving clinical outcomes in older adults with heart failure.
Approximately 5.7 million persons in the United States are diagnosed with heart failure, and the number of reported new cases is expected to increase to over 700,000 cases annually by the year 2040 [1]. This rising incidence is fueled by an aging population; by the year 2030, 1 in 5 Americans will be over 65 years of age [2]. Heart failure is prevalent among those 65 years of age and older and is the most common reason for hospitalization in this age-group. High readmission rates, approaching 50% over 6 months, are a major contributor to the the escalating economic burden associated with heart failure [3].
Persons with heart failure are more likely to be frail and experience cognitive impairment than their age-matched counterparts without heart failure. The reasons for this are not well known but may be related to hemodynamic, vascular, and inflammatory changes occurring as heart failure progresses. In this paper, we review the link between frailty and cognitive impairment in heart failure, instruments that may be useful for early detection, and interventions such as exercise that may be beneficial for attenuating both conditions.
Frailty in Heart Failure
Epidemiology
Frailty is a heightened vulnerability to stressors in the presence of low physiological reserve [4]. When exposed to stressors, persons who are frail have a much higher probability for disproportionate decompensation, negative events, functional decline, disability, and mortality [5]. Among persons with heart failure, frailty may predispose them to decompensate at a lower threshold, requiring more frequent hospitalizations. Persons with heart failure are more likely to be frail than their age-matched counterparts without heart failure [6,7].
Frailty is a powerful predictor of poor clinical outcomes and mortality in cardiovascular disease [8,9]. Compared with the non-frail, frail persons with heart failure have increased rates of mortality (16.9% vs 4.8%) and increased rates of heart failure hospitalization (20.5% vs 13.3%) [10]. Frailty has also been shown to predict falls, disability, and hospitalization in heart failure patients [6,9,11] and was found to have a negative linear relationship with health-related quality of life [12]. Frail heart failure patients are also more likely to have comorbidities such diabetes mellitus, chronic obstructive pulmonary disease, atrial fibrillation, depression, anemia, and chronic kidney disease [9,13].
Pathophysiology
There is significant overlap in the underlying pathological mechanisms of heart failure and frailty. Symptoms of heart failure, such as dyspnea, fatigue, and muscle loss, mirror components that occur with frailty. Further, cardiac cachexia, a metabolic syndrome in advanced heart failure characterized by a loss of muscle mass, is very similar to the sarcopenia that occurs in frailty.
Frailty, characterized by an increased physiologic vulnerability to stressors, may predispose frail persons with heart failure to exacerbation and worsening of heart failure due to greater susceptibility to the harmful pathophysiologic processes in heart failure, such as inflammation and autonomic dysfunction. Proposed pathophysiologic pathways in frailty include free radicals and oxidative stress, cumulative DNA damage, decreased telomere length, and nuclear fragmentation [14,15]. Frailty has been associated with low-grade chronic inflammation and increased inflammatory cytokines, such as C-reactive protein, tumor necrosis factor–alpha (TNFα), interleukin-6 (IL-6)and fibrinogen [16–18]. Heart failure also is associated with a low-grade and chronic cardiac inflammatory response that is correlated with disease progression [19].
Inflammation. IL-6 is detectable in a higher proportion of persons who are frail compared to non-frail [16] and is the most highly correlated biomarker with frailty. In addition, among those with detectable IL-6 levels, those categorized as frail have higher IL-6 levels compared to those who are non-frail [16,20]. Individuals categorized as frail were found to have significantly higher levels of TNFα than those who were non-frail [16,20]. Increased IL-6 levels are associated with decreased muscle strength, while increased TNFα levels are associated with decreased skeletal muscle protein synthesis [21,22], thus contributing to frailty.
Oxidative stress. Protein carbonyls result from protein oxidation promoted by reactive oxygen species and are markers of oxidative stress. Protein carbonylation is implicated in the pathogenesis of the loss of skeletal muscle mass; high serum protein carbonyls are associated with poor grip strength [23]. 8-OHdG is an oxidized nucleoside indicative of oxidative damage to DNA and a measure of oxidative stress. Accumulation of 8-OHdG in skeletal muscle leads to loss of muscle mass and is associated with decreased hand grip strength in the elderly [24]. Higher serum levels of 8-OHdG are present in older adults who are frail as compared to those who are non-frail [25].
Measurement of Frailty in the Clinical Setting
Frailty has been conceptualized in a number of studies using different models and measures; however, there continues to be a lack of consensus on the definition and operationalization of frailty. Prior research has led to the development of several validated models of frailty that have demonstrated good prediction of adverse outcomes in older adults. Some models, such as the Fried phenotype [6], focus solely on the physical dimension, while other models take a multidimensional approach.Single-item measures (eg, gait speed, 6-minute walk test, handgrip strength) are also commonly used to screen for frailty, but a frailty measure that incorporates more than 1 physical dimension may be more sensitive and reliable. In our opinion, the ideal measure of frailty would consist of a brief assessment that can be serially performed in most clinical practice settings that can identify change in function over time. The incorporation of sensitive physical function measures that can detect frailty early has the potential to slow physical function decline by preserving physiological thresholds.
Cognitive Impairment in Heart Failure
Epidemiology
Cognitive impairment occurs frequently in patients with heart failure, and the presence of cognitive impairment in persons with heart failure has been shown to heighten risk for adverse clinical outcomes, disability, poor quality of life, and mortality [26,27]. Heart failure negatively influences cognitive functioning in most domains [28–32]. The most common domains adversely affected by heart failure and aging are memory and executive function. Deficits in these domains can substantially diminish patient ability to carry out essential self-care behaviors [30,32].
The most common form of cognitive impairment seen in patients with heart failure is mild cognitive impairment (MCI), which is a measurable deficit with memory or another core cognitive domain. Up to 60% of persons with heart failure have been reported to have MCI. Patients with MCI have cognitive deficits that are more pronounced than those seen in normal aging, but lack other symptoms of dementia, such as impaired judgment or reasoning. MCI often will not impede patients’ ability to carry out the activities of daily living (ADLs) independently, but patients may have difficulty in performing some instrumental activities of daily living (IADLs), such as remembering medications, scheduling provider appointments. Dementia, a decline in cognitive ability severe enough to hinder an individual’s ability to perform ADLs or IADLs or engage in social activities or occupational responsibilities, occurs in approximately 25% of persons with heart failure [33].
Persons with heart failure have a fourfold greater likelihood of developing CI than persons without heart failure. Several cohort studies have shown that persons with heart failure had lower performance on cognitive tests than individuals without heart failure [34,35] and were 50% more likely to progress to dementia.
Assessment Tools
Although a comprehensive neurocognitive battery would aid in detecting cognitive impairment in heart failure, few clinical practice settings have the resources to perform such a detailed and time-consuming measurement. Most studies in heart failure have relied on global screening questionnaires such as the Mini-Mental State Examination (MMSE) [36] to assess cognitive functioning in persons with heart failure and in other cardiovascular disorders. Global cognitive measures, however, often lack sensitivity for detecting subtle cognitive deficits such as seen in MCI [28–30]. Screening that measures executive function may be the most beneficial for busy clinical settings, since declines in this domain are well established as contributing to poor outcomes in persons with heart failure.
The Montreal Cognitive Assessment (MoCA) is a rapid screening test designed to detect MCI. It assesses different cognitive domains, including attention, memory, language, and executive function [37]. The MoCA lends itself to use in clinical setting because it is brief, requires little training to administer, and is easy to score. This instrument has been used successfully to assess MCI in persons with heart failure and may be more sensitive than the MMSE in identifying clinically relevant cognitive dysfunction. In 2013 study, Cameron et al [38] administered the MMSE and MoCA to 93 hospitalized heart failure patients and found that the MoCA classified 41% of patients as cognitively impaired that were not classified using the MMSE. For persons with a vascular cognitive deficit, the MMSE has been portrayed as an inadequate screening test due to lack of sensitivity for visuospatial and executive function deficits. Because the MoCA was designed to be more sensitive to such deficits, it may be a superior screening method for persons with heart failure. Although previous studies support the use of the MoCA in persons with heart failure, more research is needed in larger, more diverse heart failure samples with a wide range of cognitive deficits.
A Reasonable Clinical Assessment Approach
Considering the link between heart failure, frailty, and MCI, incorporating simple physical performance measures with cognitive screening may be an effective strategy to identify persons at risk for frailty. Two clinically relevant physical performance-based measures of frailty are proposed: the Fried phenotype (mentioned earlier) and the Short Physical Performance Battery (SBBP). In addition, cognitive screening using the MoCA is recommended as part of the routine examination for determining possible MCI or more severe cognitive deficits. The predictive validity of measuring physical frailty is enhanced when cognitive impairment is included in the assessment [36,39].
The performance-based measures included in this review have previously demonstrated excellent psychometric properties as well as sensitivity for change that is clinically meaningful. Minimal detectable change (MDC), a threshold score that refers to the minimal amount of change outside of error that reflects true change by a patient between 2 time points (rather than variation in measurement), is important for interpreting level of risk for frailty and is included for each instrument [40,41]. If a more brief frailty examination is needed, cut-points for gait speed and handgrip have been used effectively in a number of studies as a threshold for determining frailty, including in older patients with cardiovascular disease and in heart failure [8,42,43].
Fried Frailty Phenotype
The Fried phenotype is an appropriate method of measuring frailty in a clinical setting due to its wide application across diverse populations and consistent identification of adverse outcomes [44]. This model is derived from a frailty model proposed by Fried et al [6] in which a phenotypic cycle exists that includes disease, sarcopenia, decreased walking speed, chronic undernutrition, decreased total energy expenditure, senescent musculoskeletal changes, decreased resting metabolic rate, weight loss and decreased maximal oxygen consumption. Frailty exists when a critical mass of these cycle components are identified in an individual [6].
To validate the model, Fried et al used data from the Cardiovascular Health Study and used the model to show association with a 3-year and 7-year incidence of mobility and ADL disability among 4317 community-dwelling men and women aged 65 years and older, independent of comorbidities. Several studies have directly tested the frailty phenotype model alone and in comparison to other models of frailty in large prospective studies across different populations, such as the Survey of Health, Aging and Retirement in Europe (SHARE) [45], the European Male Aging Study [46], and the Canadian Health Study of Aging [47]. While these studies found the prevalence of frailty to vary across the populations, they all validated the Fried model and found no significant differences in the predictive ability of the Fried model and other models of frailty. The Frailty Consensus conference evaluated the different models of frailty and determined that the Fried model is a validated construct of frailty and is acceptable for use in the identification of individuals who are frail or likely to become frail [48]. Thus, the Fried et al frailty phenotype model is considered to be a standard measure of frailty in older individuals.
Short Physical Performance Battery
In other chronic illness populations, the SPPB has also been used as a predictor of outcomes before, during, or after hospitalization. Valpato et al [53], for example, used the SPPB to assess older adults (mean age, 78 yr) admitted to the hospital with a diagnosis of heart failure (64%), pneumonia (13%), chronic obstructive pulmonary disease (16%), or minor stroke (6.6%) at admission (baseline) and discharge. Patients with the lowest SPPB quartile scores at hospital discharge had a fivefold greater risk of rehospitalization or mortality compared to the highest quartile. In addition, those who had an early decline in SPPB scores 1 month after hospital discharge had greater limitations in performing activities of daily living and a significantly greater probability of being re-hospitalized or death during the 1-year follow-up period. These studies suggest that the SPPB at the first follow-up outpatient visit following hospital discharge may be beneficial for identifying need for further intervention or the need for more frequent follow-up care. Although the SPPB is not part of the Fried et al phenotype, it may provide additional information concerning risk for falls and lower extremity strength that may be beneficial in the evaluation of some persons with heart failure [54]. The SPPB along with instructions and normative data are available for clinical use at no charge at www.grc.nia.nih.gov/branches/ledb/sppb/index.htm.
Interventions for Frailty in Heart Failure
Interventions to address frailty have included exercise training, comprehensive geriatric assessment and management services, social support systems, nutrition, and drugs; however, few intervention studies have examined frailty in heart failure [8]. Restoration of physical function through aerobic exercise and resistance training has shown benefit in frail older adults [55–57] and in persons with heart failure [58]. Maintaining and/or restoring physical function through aerobic and resistance exercise training may be the key to preventing further decline or potentially reversing frailty in older adults with heart failure.
Aerobic exercise has been shown to be beneficial for both frail older adults and frail persons with heart failure [18]. In a study of community-dwelling frail older adults aged 65 and older, a combined aerobic and resistance exercise intervention, performed over 16 weeks, demonstrated significant improvement in frailty scores during the 1-year follow-up in contrast to worsening frailty measures in the control group [57].
Older adults with heart failure experience a much lower exercise tolerance largely due to a 50% to 75% decrease in aerobic capacity in addition to the well-known alterations in peripheral musculoskeletal performance that contribute to fatigue and greater symptom severity. Aerobic exercise has been shown to be beneficial for most heart failure patients by altering the peripheral and central mechanisms, such as inflammatory cytokines, that contribute to heart failure exacerbations, worsened symptom severity, and poor clinical outcomes [59–62].Lower rates of hospitalization, improved physical function, and enhanced health-related quality of life are reported in heart failure patients who routinely exercise [59]. Resistance training has been shown to improve physical function in frail older adults [55]. Further, the use of TheraBand exercise bands in resistance training demonstrated improvement in physical function among frail older adults [56].
Exercise also appears to exert a positive effect on cognition, particularly executive functioning, and may also have a protective effect against cognitive decline with aging and among those with heart failure. The underlying mechanism for improvement in cognition remains poorly understood but is likely related to improved cardiac function, cerebral perfusion, and oxygenation, although this has not been clearly established. Larson et al (2006) evaluated the frequency of participation in a variety of physical activities (eg, walking, bicycling and swimming) over 6 years in 1740 older adults [63]. Older adults who exercised more than 3 times per week during initial assessment were 34% less likely to be diagnosed with dementia than those who exercised fewer than 3 times per week. Several meta-analyses in recent years have shown a consistent and positive relationship between aerobic exercise and cognition [64,65]. Importantly, findings from meta-analyses have shown a moderate effect size (> 0.5) from aerobic training, which was similar for normal and cognitively impaired adults [64].
Implications for Clinical Care
A systematic assessment performed periodically using physical and cognitive measures that may identify prefrailty may be the best strategy for preventing further functional loss, limitations, and disability in persons with heart failure. Persons with heart failure ideally should be evaluated annually for physical function, since a decline has been consistently shown to be a strong predictor of adverse health outcomes, disability, and death [6,66]. Cognitive function should also be assessed routinely in persons with heart failure, particularly when first diagnosed, when changes in treatment regimen occur, and with worsening disease severity, since these events have been shown to occur before changes in cognition [31]. Incorporating geriatric performance-based measures in heart failure management would allow for more treatment strategies aimed at improving physical function, cognitive outcomes, and quality of life. Further, identifying frailty in heart failure is an important component of clinical decision-making when determining if a patient can tolerate therapies such as implantable defibrillators, cardiac resynchronization therapy, or left ventricular assist device placement.
In older adults, performance measures are well established and commonly used as part of geriatric assessment to evaluate physical and cognitive functioning. Performance-based measures may be particularly beneficial in older adults with heart failure to monitor serial changes in physical function. Performance measures in clinical settings require staff time but little training, space, equipment, or risk. As performance measures become more common in practice settings, MDC thresholds may need to be re-evaluated based on the characteristics of the population [67].
For persons with heart failure whose screening outcomes suggest MCI, more comprehensive neuropsychological testing should be available as well as provision of resources to optimize functional independence. Early identification of impaired cognition may lower risk of poor self-management through simplification of medication regimens or providing resources to help manage other regimens essential for optimal heart failure care. It is also important to recognize that depressive symptoms are common in persons with heart failure and are highly correlated with cognitive impairment in this population. Screening for depressive symptoms therefore, may also enhance identification of persons with heart failure at risk for frailty [4,28].
Conclusion
Effective appraisal and development of effective interventions are essential in older adults with heart failure who are at high risk for frailty and cognitive impairment. This will become increasingly important as the population ages and the incidence of heart failure rises proportionately. Although curative treatments for frailty and cognitive impairment are not available, interdisciplinary interventions such as exercise and comprehensive geriatric assessment may improve outcomes in older persons with heart failure [68]. Information gained from objective, simple, inexpensive physical performance measures, when used in combination with cognitive screening, may enhance the ability to evaluate change that signal onset of frailty or cognitive impairment [54,69,70]. The high morbidity and mortality associated with frailty and cognitive impairment indicate that it should be a priority for future research as a strategy to improve clinical outcomes, enhance quality of life, and lower health care costs in this growing population.
Corresponding author: Rebecca Gary, PhD, RN, Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA 30322, [email protected].
Funding/support: B. Butts was partially funded for this work through National Institutes of Health/National
Institute of Nursing Research Grant #T32NR012715.
1. Velagaleti RS and Vasan RS. Heart failure in the twenty-first century: is it a coronary artery disease or hypertension problem. Cardiol Clin 2007;25:487–95.
2. Vincent GK, Velkoff VA. The next four decades. The older population in the United States: 2010 to 2050. United States Census Bureau Report No: P25-1138. U.S. Department of Commerce; May 2010.
3. Butler J, Kalogeropoulos A. Worsening heart failure hospitalization epidemic we do not know how to prevent and we do not know how to treat. J Am Coll Cardiol 2008;52:435–7.
4. Gary R. Evaluation of frailty in older adults with cardiovascular disease: incorporating physical performance measures. J Cardiovasc Nurs 2012;27:120–131.
5. Shamliyan T, Talley KM, Ramakrishnan R, Kane RL. Association of frailty with survival: a systematic literature review. Age Res Rev 2013;12:719–36.
6. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol Med Sci 2001; 56:M146–M156.
7. Newman AB, Gottdiener JS, Mcburnie MA, et al. Associations of subclinical cardiovascular disease with frailty. J Gerontol A Biol Sci Med Sci 2001;56:M158–66.
8. Afilalo J, Karunananthan S, Eisenberg MJ, et al. Role of frailty in patients with cardiovascular disease. Am J Cardiol 2009; 103:1616–21.
9. Cacciatore F, Abete P, Maella F, et al. Frailty predicts long-term mortality in elderly subjects with chronic heart failure. Eur J Clin Invest 2008;35:723–30.
10. Lupón J, González B, Santaeugenia S, et al. Prognostic implication of frailty and depressive symptoms in an outpatient population with heart failure. Rev Españ Cardiol 2008;61:835–42.
11. Rich MW. Heart failure in the oldest patients: the impact of comorbid conditions. Am J Geriatr Cardiol 2007;14:134–41.
12. Buck HG, Riegel B. The impact of frailty on health related quality of life in heart failure. Eur J Cardiovasc Nurs 2011;10:159–66
13. Boxer RS, Shah KB Kenny AM. Frailty and prognosis in advanced heart failure. Curr Opin Supp Pall Care 2014;8:25–9.
14. Afilalo J, Sebag IA, Chalifour LE, et al. Age-related changes in lamin A/C expression in cardiomyocytes. Am J Physiol Heart Circ Physiol 2007;293:H1451–6.
15. Walston J. Frailty—the search for underlying causes. Sci Aging Know Environ 2004;2004:e4.
16. Hubbard RE, O’Mahony MS, Savva GM, et al. Inflammation and frailty measures in older people. J Cell Mol Med 2009; 13:3103–9.
17. Hubbard RE Woodhouse KW. Frailty, inflammation and the elderly. Biogerontol 2010;11:635–41.
18. Baptista G, Dupuy A-M, Jaussent A, et al. Low-grade chronic inflammation and superoxide anion production by NADPH oxidase are the main determinants of physical frailty in older adults. Free Rad Res 2012;46:1108–14.
19. Abbate A. The heart on fire: Inflammasome and cardiomyopathy. Exper Physiol 2013;98:385.
20. Collerton J, Martin-Ruiz C, Davies K, et al. Frailty and the role of inflammation, immunosenescence and cellular ageing in the very old: Cross-sectional findings from the Newcastle 85+ Study. Mech Age Devel 2012;133:456–66.
21. Ferrucci L, Harris TB, Guralnik JM, et al. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc 1999;47:639–46.
22. Toth MJ, Matthews DE, Tracy RP and Previs MJ. Age-related differences in skeletal muscle protein synthesis: relation to markers of immune activation. Am j Physiol Endocrin Metab 2005;288:E883–91.
23. Howard C, Ferrucci L, Sun K, et al. Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol 2007;103:17–20.
24. Muzembo BA, Nagano Y, Eitoku M, et al. A cross-sectional assessment of oxidative DNA damage and muscle strength among elderly people living in the community. Envir Health Prev Med 2014;19:21–9.
25. Wu I-C, Shiesh S-C, Kuo P-H and Lin X-Z. High oxidative stress is correlated with frailty in elderly Chinese. J Am Geriatr Soc 2009;57:1666–71.
26. Alosco ML, Spitznagel MB, Cohen R, et al. Cognitive impairment is independently associated with reduced instrumental activities of daily living in persons with heart failure. J Cardiovasc Nurs 2012;27:44–50.
27. Feola M, Rosso GL, Peano M, et al. Correlation between cognitive impairment and prognostic parameters in patients with congestive heart failure. Arch Med Res 2007;38:234–9.
28. Pressler SJ, Subramanian U, Kareken D, et al. Cognitive deficits in chronic heart failure. Nurs Res 2010;59:127–39.
29. Pressler SJ, Kim J, Riley P, et al. Memory dysfunction, psychomotor slowing, and decreased executive function predict mortality in patients with heart failure and low ejection fraction. J Cardiac Fail 2010;16:750–60.
30. Pressler SJ, Subramanian U, Kareken D, et al. Cognitive deficits and health-related quality of life in chronic heart failure. J Cardiovasc Nurs 2010;25:189–98.
31. Hajduk AM, Lemon SC, Mcmanus DD, et al. Cognitive impairment and self-care in heart failure. Clin Epidemiol 2013; 24:407–16.
32. Dardiotis E, Giamouzis G, Mastrogiannis D, et al. Cognitive impairment in heart failure. Cardiol Res Prac 2012; 2012:595821.
33. Petersen RC and O’brien J. Mild cognitive impairment should be considered for DSM-V. J Geriatr Psychiatry Neurol 2006; 19:147–54.
34. Hjelm C, Dahl A, Broström A, et al. The influence of heart failure on longitudinal changes in cognition among individuals 80 years of age and older. J Clin Nurs 2012; 21:994–1003.
35. Almeida OP, Garrido GJ, Beer C, et al. Cognitive and brain changes associated with ischaemic heart disease and heart failure. Eur Heart J 2012;33:1769–76.
36. Folstein MF, Folstein SE, McHugh PR. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189–98.
37. Nasveddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005;53:695–699.
38. Cameron J, Worrall-Carter L, Page K, et al. Screening for mild cognitive impairment in patients with heart failure: Montreal cognitive assessment versus mini mental state exam. Eur J Cardiovasc Nurs 2013;12:252–60.
39. Avila-Funes JA, Amieva H, Barberger-Gateau P, et al. Cognitive impairment improves the predictive validity of the phenotype of frailty for adverse health outcomes: the three-city study. J Am Geriatr Soc 57:453–61.
40. Perera S, Mody SH, Woodman RC, Studenski SA. Meaningful change and responsiveness in common physical performance measures in older adults. Am Geriatr Soc 2006;54:743–9.
41. Kwon S, Perera S, Pahor M, et al. What is a meaningful change in physical performance? Findings from a clinical trial in older adults (the LIFE-P study). J Nutr Health Aging 2009;13:538–44.
42. Abellan Van Kan G, Rolland Y, Houles M, et al. The assessment of frailty in older adults. Clin Geriatr Med 2010; 26:275–86.
43. Pahor M, Manini T, Cesari M. Sarcopenia: clinical evaluation, biological markers and other evaluation tools. J Nutr Health Aging 2009;13:724–8.
44. Gary R. Evaluation of frailty in older adults with cardiovascular disease. J Cardiovasc Nurs 2012;27:120–31.
45. Macklai NS, Spagnoli J, Junod J, Santos-Eggimann B. Prospective association of the SHARE-operationalized frailty phenotype with adverse health outcomes: evidence from 620+ community-dwelling Europeans living in 11 countries. BMC Geriatr 2013;13:1–9.
46. Ravinrarajah R, Lee DM, Pye SR, et al. The ability of three different models of frailty to predict all-cause mortality: Results from the European Male Aging Study (EMAS). Arch Gerontol Geriatr 2013;57:360–8.
47. Rockwood K, Andrew M and Mitnitski A. A comparison of two approaches to measuring frailty in elderly people. J Gerontol Med Sci 2007;62:738–43.
48. Morley JE, Vellas B, Van Kan GA, et al. Frailty consensus: a call to action. J Am Med Dir Assoc 2013;14:392–7.
49. Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol 1994;49:M85–94.
50. Guralnik JM, Ferrucci L, Simonsick EM, et al. Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N Engl J Med 1995;332:556–61.
51. Di Bari M, Pozzi C, Cavallini Mc, et al. The diagnosis of heart failure in the community. Comparative validation of four sets of criteria in unselected older adults: the ICARe Dicomano Study. J Am Coll Cardiol 2004;44:1601–08.
52. Chiarantini D, Volpato S, Sioulis F, et al. Lower extremity performance measures predict long-term prognosis in older patients hospitalized for heart failure. J Cardiac Failure 2010; 16:390–5.
53. Volpato S, Cavalieri M, Sioulis F, et al. Predictive value of the short physical performance battery following hospitalization in older patients. J Gerontol A Biol Sci Med Sci 2011;66:89–96.
54. Studenski S, Perera S, Wallace D, et al. Physical performance measures in the clinical setting. J Am Geriatr Soc 2003; 51:314–22.
55. Binder EF, Schechtman KB, Ehsani AA, et al. Effects of exercise training on frailty in community-dwelling older adults: results of a randomized, controlled trial. J Am Geriatr Soc 2012; 50:1921–8.
56. Brown M, Sinacore DR, Ehsani AA, et al. Low-intensity exercise as a modifier of physical frailty in older adults. Arch Phys Med Rehab 2000;81:960–5.
57. Yamada M, Arai H, Sonoda T and Aoyama T. Community-based exercise program is cost-effective by preventing care and disability in Japanese frail older adults. J Am Med Dir Assoc 2012;13:507–11.
58. Gary RA, Cress ME, Higgins MK, et al. A combined aerobic and resistance exercise program improves physical functional performance in patients with heart failure: a pilot study. J Cardiovasc Nurs 2012;27:418–30.
59. De Meirelles L, Matsuura C, Resende AD, et al. Chronic exercise leads to antiaggregant, antioxidant and anti-inflammatory effects in heart failure patients. Eur J Prev Cardiol 2014;21:1225–32.
60. Feiereisen P, Vaillant M, Gilson G, Delagardelle C. Effects of different training modalities on circulating anabolic/catabolic markers in chronic heart failure. J Cardiopulm Rehab Prev 2013;33:303–8.
61. Smart NA, Steele M. The effect of physical training on systemic proinflammatory cytokine expression in heart failure patients: a systematic review. Congest Heart Fail 2011;17:110–4.
62. Nunes RB, Alves JP, Kessler LP, Lago PD. Aerobic exercsie improves the inflammatory profile correlated with cardiac remodeling and function in chronic heart failure rats. Clin Chest Med 2013;68:876–82.
63. Larson EB, Wang L, Bowen JD, et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann Intern Med 2006;144:73–8.
64. Colcombe S and Kramer AF. Fitness effects on the cognitive function of older adults: a meta-analytic study. Psychol Sci 2003;14:125–30.
65. Heyn P, Abreu BC, Ottenbacher KJ. The effects of exercise training on elderly persons with cognitive impairment and dementia: a meta-analysis. Arch Phys Med Rehab 2004; 85:1694–704.
66. Bautmans I, Vanpuyvelde K, Mets T. Sarcopenia and functional decline: pathophysiology, prevention and therapy. Acta Clinica Belgica 2009;64:303–16.
67. Guralnik JM, Ferrucci L, Pieper CF, et al. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J Gerontol A Biol Sci Med Sci 2000;55A:M221–M231.
68. Kramer AF, Erickson KI. Capitalizing on cortical plasticity: influence of physical activity on cognition and brain function. Trends Cogn Sci 2007;11:342–8.
69. Harkness K, Heckman GA, Mckelvie RS. The older patient with heart failure: high risk for frailty and cognitive impairment. Expert Rev Cardiovasc Ther 2012;10:779–95.
70. Waters DL, Baumgartner RN, Garry PJ, Vellas B. Advantages of dietary, exercise-related, and therapeutic interventions to prevent and treat sarcopenia in adult patients: an update. Clin Interv Aging 2010;5:259–70.
1. Velagaleti RS and Vasan RS. Heart failure in the twenty-first century: is it a coronary artery disease or hypertension problem. Cardiol Clin 2007;25:487–95.
2. Vincent GK, Velkoff VA. The next four decades. The older population in the United States: 2010 to 2050. United States Census Bureau Report No: P25-1138. U.S. Department of Commerce; May 2010.
3. Butler J, Kalogeropoulos A. Worsening heart failure hospitalization epidemic we do not know how to prevent and we do not know how to treat. J Am Coll Cardiol 2008;52:435–7.
4. Gary R. Evaluation of frailty in older adults with cardiovascular disease: incorporating physical performance measures. J Cardiovasc Nurs 2012;27:120–131.
5. Shamliyan T, Talley KM, Ramakrishnan R, Kane RL. Association of frailty with survival: a systematic literature review. Age Res Rev 2013;12:719–36.
6. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol Med Sci 2001; 56:M146–M156.
7. Newman AB, Gottdiener JS, Mcburnie MA, et al. Associations of subclinical cardiovascular disease with frailty. J Gerontol A Biol Sci Med Sci 2001;56:M158–66.
8. Afilalo J, Karunananthan S, Eisenberg MJ, et al. Role of frailty in patients with cardiovascular disease. Am J Cardiol 2009; 103:1616–21.
9. Cacciatore F, Abete P, Maella F, et al. Frailty predicts long-term mortality in elderly subjects with chronic heart failure. Eur J Clin Invest 2008;35:723–30.
10. Lupón J, González B, Santaeugenia S, et al. Prognostic implication of frailty and depressive symptoms in an outpatient population with heart failure. Rev Españ Cardiol 2008;61:835–42.
11. Rich MW. Heart failure in the oldest patients: the impact of comorbid conditions. Am J Geriatr Cardiol 2007;14:134–41.
12. Buck HG, Riegel B. The impact of frailty on health related quality of life in heart failure. Eur J Cardiovasc Nurs 2011;10:159–66
13. Boxer RS, Shah KB Kenny AM. Frailty and prognosis in advanced heart failure. Curr Opin Supp Pall Care 2014;8:25–9.
14. Afilalo J, Sebag IA, Chalifour LE, et al. Age-related changes in lamin A/C expression in cardiomyocytes. Am J Physiol Heart Circ Physiol 2007;293:H1451–6.
15. Walston J. Frailty—the search for underlying causes. Sci Aging Know Environ 2004;2004:e4.
16. Hubbard RE, O’Mahony MS, Savva GM, et al. Inflammation and frailty measures in older people. J Cell Mol Med 2009; 13:3103–9.
17. Hubbard RE Woodhouse KW. Frailty, inflammation and the elderly. Biogerontol 2010;11:635–41.
18. Baptista G, Dupuy A-M, Jaussent A, et al. Low-grade chronic inflammation and superoxide anion production by NADPH oxidase are the main determinants of physical frailty in older adults. Free Rad Res 2012;46:1108–14.
19. Abbate A. The heart on fire: Inflammasome and cardiomyopathy. Exper Physiol 2013;98:385.
20. Collerton J, Martin-Ruiz C, Davies K, et al. Frailty and the role of inflammation, immunosenescence and cellular ageing in the very old: Cross-sectional findings from the Newcastle 85+ Study. Mech Age Devel 2012;133:456–66.
21. Ferrucci L, Harris TB, Guralnik JM, et al. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc 1999;47:639–46.
22. Toth MJ, Matthews DE, Tracy RP and Previs MJ. Age-related differences in skeletal muscle protein synthesis: relation to markers of immune activation. Am j Physiol Endocrin Metab 2005;288:E883–91.
23. Howard C, Ferrucci L, Sun K, et al. Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol 2007;103:17–20.
24. Muzembo BA, Nagano Y, Eitoku M, et al. A cross-sectional assessment of oxidative DNA damage and muscle strength among elderly people living in the community. Envir Health Prev Med 2014;19:21–9.
25. Wu I-C, Shiesh S-C, Kuo P-H and Lin X-Z. High oxidative stress is correlated with frailty in elderly Chinese. J Am Geriatr Soc 2009;57:1666–71.
26. Alosco ML, Spitznagel MB, Cohen R, et al. Cognitive impairment is independently associated with reduced instrumental activities of daily living in persons with heart failure. J Cardiovasc Nurs 2012;27:44–50.
27. Feola M, Rosso GL, Peano M, et al. Correlation between cognitive impairment and prognostic parameters in patients with congestive heart failure. Arch Med Res 2007;38:234–9.
28. Pressler SJ, Subramanian U, Kareken D, et al. Cognitive deficits in chronic heart failure. Nurs Res 2010;59:127–39.
29. Pressler SJ, Kim J, Riley P, et al. Memory dysfunction, psychomotor slowing, and decreased executive function predict mortality in patients with heart failure and low ejection fraction. J Cardiac Fail 2010;16:750–60.
30. Pressler SJ, Subramanian U, Kareken D, et al. Cognitive deficits and health-related quality of life in chronic heart failure. J Cardiovasc Nurs 2010;25:189–98.
31. Hajduk AM, Lemon SC, Mcmanus DD, et al. Cognitive impairment and self-care in heart failure. Clin Epidemiol 2013; 24:407–16.
32. Dardiotis E, Giamouzis G, Mastrogiannis D, et al. Cognitive impairment in heart failure. Cardiol Res Prac 2012; 2012:595821.
33. Petersen RC and O’brien J. Mild cognitive impairment should be considered for DSM-V. J Geriatr Psychiatry Neurol 2006; 19:147–54.
34. Hjelm C, Dahl A, Broström A, et al. The influence of heart failure on longitudinal changes in cognition among individuals 80 years of age and older. J Clin Nurs 2012; 21:994–1003.
35. Almeida OP, Garrido GJ, Beer C, et al. Cognitive and brain changes associated with ischaemic heart disease and heart failure. Eur Heart J 2012;33:1769–76.
36. Folstein MF, Folstein SE, McHugh PR. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189–98.
37. Nasveddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005;53:695–699.
38. Cameron J, Worrall-Carter L, Page K, et al. Screening for mild cognitive impairment in patients with heart failure: Montreal cognitive assessment versus mini mental state exam. Eur J Cardiovasc Nurs 2013;12:252–60.
39. Avila-Funes JA, Amieva H, Barberger-Gateau P, et al. Cognitive impairment improves the predictive validity of the phenotype of frailty for adverse health outcomes: the three-city study. J Am Geriatr Soc 57:453–61.
40. Perera S, Mody SH, Woodman RC, Studenski SA. Meaningful change and responsiveness in common physical performance measures in older adults. Am Geriatr Soc 2006;54:743–9.
41. Kwon S, Perera S, Pahor M, et al. What is a meaningful change in physical performance? Findings from a clinical trial in older adults (the LIFE-P study). J Nutr Health Aging 2009;13:538–44.
42. Abellan Van Kan G, Rolland Y, Houles M, et al. The assessment of frailty in older adults. Clin Geriatr Med 2010; 26:275–86.
43. Pahor M, Manini T, Cesari M. Sarcopenia: clinical evaluation, biological markers and other evaluation tools. J Nutr Health Aging 2009;13:724–8.
44. Gary R. Evaluation of frailty in older adults with cardiovascular disease. J Cardiovasc Nurs 2012;27:120–31.
45. Macklai NS, Spagnoli J, Junod J, Santos-Eggimann B. Prospective association of the SHARE-operationalized frailty phenotype with adverse health outcomes: evidence from 620+ community-dwelling Europeans living in 11 countries. BMC Geriatr 2013;13:1–9.
46. Ravinrarajah R, Lee DM, Pye SR, et al. The ability of three different models of frailty to predict all-cause mortality: Results from the European Male Aging Study (EMAS). Arch Gerontol Geriatr 2013;57:360–8.
47. Rockwood K, Andrew M and Mitnitski A. A comparison of two approaches to measuring frailty in elderly people. J Gerontol Med Sci 2007;62:738–43.
48. Morley JE, Vellas B, Van Kan GA, et al. Frailty consensus: a call to action. J Am Med Dir Assoc 2013;14:392–7.
49. Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol 1994;49:M85–94.
50. Guralnik JM, Ferrucci L, Simonsick EM, et al. Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N Engl J Med 1995;332:556–61.
51. Di Bari M, Pozzi C, Cavallini Mc, et al. The diagnosis of heart failure in the community. Comparative validation of four sets of criteria in unselected older adults: the ICARe Dicomano Study. J Am Coll Cardiol 2004;44:1601–08.
52. Chiarantini D, Volpato S, Sioulis F, et al. Lower extremity performance measures predict long-term prognosis in older patients hospitalized for heart failure. J Cardiac Failure 2010; 16:390–5.
53. Volpato S, Cavalieri M, Sioulis F, et al. Predictive value of the short physical performance battery following hospitalization in older patients. J Gerontol A Biol Sci Med Sci 2011;66:89–96.
54. Studenski S, Perera S, Wallace D, et al. Physical performance measures in the clinical setting. J Am Geriatr Soc 2003; 51:314–22.
55. Binder EF, Schechtman KB, Ehsani AA, et al. Effects of exercise training on frailty in community-dwelling older adults: results of a randomized, controlled trial. J Am Geriatr Soc 2012; 50:1921–8.
56. Brown M, Sinacore DR, Ehsani AA, et al. Low-intensity exercise as a modifier of physical frailty in older adults. Arch Phys Med Rehab 2000;81:960–5.
57. Yamada M, Arai H, Sonoda T and Aoyama T. Community-based exercise program is cost-effective by preventing care and disability in Japanese frail older adults. J Am Med Dir Assoc 2012;13:507–11.
58. Gary RA, Cress ME, Higgins MK, et al. A combined aerobic and resistance exercise program improves physical functional performance in patients with heart failure: a pilot study. J Cardiovasc Nurs 2012;27:418–30.
59. De Meirelles L, Matsuura C, Resende AD, et al. Chronic exercise leads to antiaggregant, antioxidant and anti-inflammatory effects in heart failure patients. Eur J Prev Cardiol 2014;21:1225–32.
60. Feiereisen P, Vaillant M, Gilson G, Delagardelle C. Effects of different training modalities on circulating anabolic/catabolic markers in chronic heart failure. J Cardiopulm Rehab Prev 2013;33:303–8.
61. Smart NA, Steele M. The effect of physical training on systemic proinflammatory cytokine expression in heart failure patients: a systematic review. Congest Heart Fail 2011;17:110–4.
62. Nunes RB, Alves JP, Kessler LP, Lago PD. Aerobic exercsie improves the inflammatory profile correlated with cardiac remodeling and function in chronic heart failure rats. Clin Chest Med 2013;68:876–82.
63. Larson EB, Wang L, Bowen JD, et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann Intern Med 2006;144:73–8.
64. Colcombe S and Kramer AF. Fitness effects on the cognitive function of older adults: a meta-analytic study. Psychol Sci 2003;14:125–30.
65. Heyn P, Abreu BC, Ottenbacher KJ. The effects of exercise training on elderly persons with cognitive impairment and dementia: a meta-analysis. Arch Phys Med Rehab 2004; 85:1694–704.
66. Bautmans I, Vanpuyvelde K, Mets T. Sarcopenia and functional decline: pathophysiology, prevention and therapy. Acta Clinica Belgica 2009;64:303–16.
67. Guralnik JM, Ferrucci L, Pieper CF, et al. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J Gerontol A Biol Sci Med Sci 2000;55A:M221–M231.
68. Kramer AF, Erickson KI. Capitalizing on cortical plasticity: influence of physical activity on cognition and brain function. Trends Cogn Sci 2007;11:342–8.
69. Harkness K, Heckman GA, Mckelvie RS. The older patient with heart failure: high risk for frailty and cognitive impairment. Expert Rev Cardiovasc Ther 2012;10:779–95.
70. Waters DL, Baumgartner RN, Garry PJ, Vellas B. Advantages of dietary, exercise-related, and therapeutic interventions to prevent and treat sarcopenia in adult patients: an update. Clin Interv Aging 2010;5:259–70.