User login
Common Presentation for Complex Condition
A month ago, a 33-year-old woman noticed skin changes on her arms and face. The affected areas have recently begun to itch and burn—particularly, the patient notes, since she spent an extended period in the sun over the weekend. She has used an antifungal cream (nystatin) on the rash, to no avail.
The patient denies joint pain, fever, and malaise. She has a sister with multiple sclerosis, but her family history is otherwise unremarkable. The patient’s only medication is oral contraceptives, which she has taken since the birth of her first and only child last year.
EXAMINATION
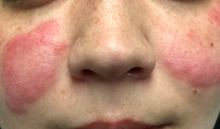
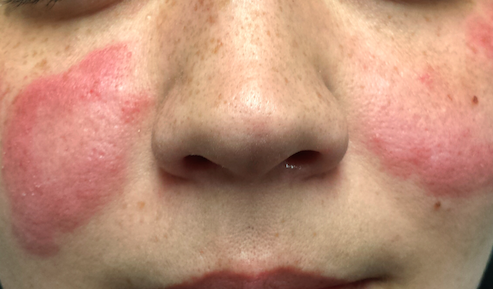
The patient is afebrile and in no particular distress. A florid red rash covers both cheeks, sparing the nose entirely. The margins are somewhat indurated and redder than the clearing centers. The follicular orifices are somewhat patulous, and a fine scale covers the affected areas of the face. The lateral brachial and triceps (sun-exposed) areas of both arms are similarly affected.
Punch biopsy reveals classic signs of lupus: vacuolar alteration of the basal cell layer, perivascular infiltrate around appendicial structures, and modest epidermal atrophy. Bloodwork yields no evidence of systemic lupus erythematosus (SLE).
What is the diagnosis?
DISCUSSION
Lupus, as a general topic, can be utterly confusing. Here are some facts that might help you make sense of the subject:
Purely cutaneous forms of lupus are commonly seen in dermatology; they manifest in sun-exposed skin as scaly annular lesions with clearing centers. Somewhat confusingly, however, cases of systemic lupus erythematosus (SLE) can present with similar cutaneous signs—and furthermore, patients with purely cutaneous lupus (subacute cutaneous lupus) may exhibit some systemic symptoms (just not enough to meet the strict criteria for SLE). Lupus in general is far more common in women than in men.
The “butterfly rash” seen in this case is uncommon but can occur in either cutaneous or systemic lupus. In most cases, though, this particular rash is a manifestation of seborrhea, psoriasis, rosacea or eczema—not lupus at all.
There are, of course, many other types of lupus. Another common form is discoid lupus (DLE), which manifests with round, scaly lesions on the head, neck, or ears; these are often misidentified as actinic keratosis, eczema, or psoriasis. DLE can be localized or generalized, purely cutaneous or a manifestation of SLE.
The key to diagnosis lies in first considering lupus in the differential and then biopsying the lesion (or sending the patient to someone who will). Once the clinical diagnosis is histologically confirmed (typical results include vacuolar interface dermatitis with sparse lymphocytic perivascular infiltrate), an immunologic workup is warranted. Also, depending on the predominant organ systems involved, the patient should be thoroughly evaluated by a dermatology or rheumatology specialist (or both).
Lupus is an autoimmune process, but in some ways it’s more useful to think of it as a form of vasculitis—which is why it can affect almost any organ system. The very first lupus patient I ever saw was in the psych ward having a psychotic break, which turned out to be secondary to a lupus-induced cerebritis. Since then, I’ve seen it affect the pericardium, kidneys, lungs, and joints. Lupus is even a major item in the differential of alopecia! SLE in particular is associated with an increase in thrombotic events and accounts for most early deaths from lupus (ie, within the first five years of diagnosis, when the cause of death is usually renal or pulmonary).
The patient in this case proved to have only cutaneous disease. She’ll respond nicely to a combination of sun protection and oral antimalarials (hydroxychloroquine) but will probably have recurrences every spring. Although unlikely to ever develop SLE, she is statistically more likely to develop other autoimmune diseases.
TAKE-HOME LEARNING POINTS
• Cutaneous lupus is more common than you might imagine. Lesions and eruptions in sun-exposed skin should prompt consideration of that item in the differential.
• Many forms of lupus have been identified, including neonatal lupus and overlapping syndromes involving lupus and lichen planus or even pemphigus.
• Though UV exposure is not always the cause, almost every type of lupus is worsened by UV light exposure.
• The differential for lupus is vast but includes psoriasis, sarcoidosis, dermatomyositis, and drug eruptions.
A month ago, a 33-year-old woman noticed skin changes on her arms and face. The affected areas have recently begun to itch and burn—particularly, the patient notes, since she spent an extended period in the sun over the weekend. She has used an antifungal cream (nystatin) on the rash, to no avail.
The patient denies joint pain, fever, and malaise. She has a sister with multiple sclerosis, but her family history is otherwise unremarkable. The patient’s only medication is oral contraceptives, which she has taken since the birth of her first and only child last year.
EXAMINATION
The patient is afebrile and in no particular distress. A florid red rash covers both cheeks, sparing the nose entirely. The margins are somewhat indurated and redder than the clearing centers. The follicular orifices are somewhat patulous, and a fine scale covers the affected areas of the face. The lateral brachial and triceps (sun-exposed) areas of both arms are similarly affected.
Punch biopsy reveals classic signs of lupus: vacuolar alteration of the basal cell layer, perivascular infiltrate around appendicial structures, and modest epidermal atrophy. Bloodwork yields no evidence of systemic lupus erythematosus (SLE).
What is the diagnosis?
DISCUSSION
Lupus, as a general topic, can be utterly confusing. Here are some facts that might help you make sense of the subject:
Purely cutaneous forms of lupus are commonly seen in dermatology; they manifest in sun-exposed skin as scaly annular lesions with clearing centers. Somewhat confusingly, however, cases of systemic lupus erythematosus (SLE) can present with similar cutaneous signs—and furthermore, patients with purely cutaneous lupus (subacute cutaneous lupus) may exhibit some systemic symptoms (just not enough to meet the strict criteria for SLE). Lupus in general is far more common in women than in men.
The “butterfly rash” seen in this case is uncommon but can occur in either cutaneous or systemic lupus. In most cases, though, this particular rash is a manifestation of seborrhea, psoriasis, rosacea or eczema—not lupus at all.
There are, of course, many other types of lupus. Another common form is discoid lupus (DLE), which manifests with round, scaly lesions on the head, neck, or ears; these are often misidentified as actinic keratosis, eczema, or psoriasis. DLE can be localized or generalized, purely cutaneous or a manifestation of SLE.
The key to diagnosis lies in first considering lupus in the differential and then biopsying the lesion (or sending the patient to someone who will). Once the clinical diagnosis is histologically confirmed (typical results include vacuolar interface dermatitis with sparse lymphocytic perivascular infiltrate), an immunologic workup is warranted. Also, depending on the predominant organ systems involved, the patient should be thoroughly evaluated by a dermatology or rheumatology specialist (or both).
Lupus is an autoimmune process, but in some ways it’s more useful to think of it as a form of vasculitis—which is why it can affect almost any organ system. The very first lupus patient I ever saw was in the psych ward having a psychotic break, which turned out to be secondary to a lupus-induced cerebritis. Since then, I’ve seen it affect the pericardium, kidneys, lungs, and joints. Lupus is even a major item in the differential of alopecia! SLE in particular is associated with an increase in thrombotic events and accounts for most early deaths from lupus (ie, within the first five years of diagnosis, when the cause of death is usually renal or pulmonary).
The patient in this case proved to have only cutaneous disease. She’ll respond nicely to a combination of sun protection and oral antimalarials (hydroxychloroquine) but will probably have recurrences every spring. Although unlikely to ever develop SLE, she is statistically more likely to develop other autoimmune diseases.
TAKE-HOME LEARNING POINTS
• Cutaneous lupus is more common than you might imagine. Lesions and eruptions in sun-exposed skin should prompt consideration of that item in the differential.
• Many forms of lupus have been identified, including neonatal lupus and overlapping syndromes involving lupus and lichen planus or even pemphigus.
• Though UV exposure is not always the cause, almost every type of lupus is worsened by UV light exposure.
• The differential for lupus is vast but includes psoriasis, sarcoidosis, dermatomyositis, and drug eruptions.
A month ago, a 33-year-old woman noticed skin changes on her arms and face. The affected areas have recently begun to itch and burn—particularly, the patient notes, since she spent an extended period in the sun over the weekend. She has used an antifungal cream (nystatin) on the rash, to no avail.
The patient denies joint pain, fever, and malaise. She has a sister with multiple sclerosis, but her family history is otherwise unremarkable. The patient’s only medication is oral contraceptives, which she has taken since the birth of her first and only child last year.
EXAMINATION
The patient is afebrile and in no particular distress. A florid red rash covers both cheeks, sparing the nose entirely. The margins are somewhat indurated and redder than the clearing centers. The follicular orifices are somewhat patulous, and a fine scale covers the affected areas of the face. The lateral brachial and triceps (sun-exposed) areas of both arms are similarly affected.
Punch biopsy reveals classic signs of lupus: vacuolar alteration of the basal cell layer, perivascular infiltrate around appendicial structures, and modest epidermal atrophy. Bloodwork yields no evidence of systemic lupus erythematosus (SLE).
What is the diagnosis?
DISCUSSION
Lupus, as a general topic, can be utterly confusing. Here are some facts that might help you make sense of the subject:
Purely cutaneous forms of lupus are commonly seen in dermatology; they manifest in sun-exposed skin as scaly annular lesions with clearing centers. Somewhat confusingly, however, cases of systemic lupus erythematosus (SLE) can present with similar cutaneous signs—and furthermore, patients with purely cutaneous lupus (subacute cutaneous lupus) may exhibit some systemic symptoms (just not enough to meet the strict criteria for SLE). Lupus in general is far more common in women than in men.
The “butterfly rash” seen in this case is uncommon but can occur in either cutaneous or systemic lupus. In most cases, though, this particular rash is a manifestation of seborrhea, psoriasis, rosacea or eczema—not lupus at all.
There are, of course, many other types of lupus. Another common form is discoid lupus (DLE), which manifests with round, scaly lesions on the head, neck, or ears; these are often misidentified as actinic keratosis, eczema, or psoriasis. DLE can be localized or generalized, purely cutaneous or a manifestation of SLE.
The key to diagnosis lies in first considering lupus in the differential and then biopsying the lesion (or sending the patient to someone who will). Once the clinical diagnosis is histologically confirmed (typical results include vacuolar interface dermatitis with sparse lymphocytic perivascular infiltrate), an immunologic workup is warranted. Also, depending on the predominant organ systems involved, the patient should be thoroughly evaluated by a dermatology or rheumatology specialist (or both).
Lupus is an autoimmune process, but in some ways it’s more useful to think of it as a form of vasculitis—which is why it can affect almost any organ system. The very first lupus patient I ever saw was in the psych ward having a psychotic break, which turned out to be secondary to a lupus-induced cerebritis. Since then, I’ve seen it affect the pericardium, kidneys, lungs, and joints. Lupus is even a major item in the differential of alopecia! SLE in particular is associated with an increase in thrombotic events and accounts for most early deaths from lupus (ie, within the first five years of diagnosis, when the cause of death is usually renal or pulmonary).
The patient in this case proved to have only cutaneous disease. She’ll respond nicely to a combination of sun protection and oral antimalarials (hydroxychloroquine) but will probably have recurrences every spring. Although unlikely to ever develop SLE, she is statistically more likely to develop other autoimmune diseases.
TAKE-HOME LEARNING POINTS
• Cutaneous lupus is more common than you might imagine. Lesions and eruptions in sun-exposed skin should prompt consideration of that item in the differential.
• Many forms of lupus have been identified, including neonatal lupus and overlapping syndromes involving lupus and lichen planus or even pemphigus.
• Though UV exposure is not always the cause, almost every type of lupus is worsened by UV light exposure.
• The differential for lupus is vast but includes psoriasis, sarcoidosis, dermatomyositis, and drug eruptions.
Thrombolysis gap for stroke octogenarians disappears
NASHVILLE, TENN. – By 2010, U.S. octogenarians with acute ischemic stroke received intravenous thrombolytic treatment about as often as younger patients, showing that a sharp, age-related disparity in thrombolytic use a decade before had disappeared, based on comprehensive national data.
The 2010 data from the Nationwide Inpatient Sample further showed that the sex-based disparity in treatment with intravenous tissue plasminogen activator (TPA) seen in 2000 resolved as well from 2000 to 2010, but other disparities worsened with declines during the period in use of TPA at rural hospitals relative to urban hospitals and at nonteaching hospitals, compared with teaching hospitals, Dr. Michelle P. Lin said at the International Stroke Conference.
Perhaps most importantly, the statistics showed a “dramatic” increase in TPA use among all age groups during the decade ending in 2010, when thrombolytic therapy was administered to 3.5%-3.9% of adult patients regardless of their age, said Dr. Lin of the department of neurology at the University of Southern California in Los Angeles. In 2000, U.S. patients received TPA at less than a third of that rate.
Low TPA use in 2000 among patients aged 80 or older in part reflected the low number of octogenarians enrolled in the trials that documented the safety and efficacy of TPA for acute ischemicstroke patients, Dr. Lin said at the International Stroke Conference, sponsored by the American Heart Association.
Data from the National Inpatient Sample included information on the treatment received by 5,932,175 patients with acute ischemic stroke at more than 1,000 U.S. hospitals during 2000-2010. The age breakdown of the nearly 6 millionpatients showed 28% were aged 18-64 years, 37% were65-79, and 35% were 80 years or older.
In 2000, medical staffs administered intravenous treatment with TPA to 1.02% of these patients aged 18-64 years, 0.92% of patients aged 65-79 years, and 0.47% of patients aged 80 or older. By 2010, the annual rates of TPA use ran 3.61% in those 18-64 years, 3.87% among those 65-79 years, and 3.55% in patients 80 years or older. In an adjusted analysis, this translated into a greater than threefold increase in TPA use among the 18- to 64-year-olds, a nearly fourfold rise in patients 65-79 years, and a nearly sevenfold jump among those 80 or older, a 24% average annual increased rate among the oldest patients, who averaged 86 years old, Dr. Lin reported.
The data also showed that among the octogenarians during 2000-2005, TPA use in women lagged behind use in men by a relative 15%, but this completely disappeared during 2006-2010, when usage rates in men and women evened out. TPA use among African Americans, Hispanics, and Asians, compared with whites, remained significantly below the rate in whites throughout the decade, although the extent of the disparity narrowed for African Americans and Asians during the second half of the decade, compared with the first half.
Dr. Lin and her associates also analyzed TPA use relative to the demographic setting of the hospital and its teaching status. During 2000-2010, the relative usage of TPA at rural hospitals, compared with urban hospitals, fell from 65% of the comparator level to 36%. Among nonteaching hospitals, the rate of TPA use dropped from 52% of the teaching hospitals’ level to 49%.
Dr. Lin said she had no relevant financial disclosures.
[email protected]
On Twitter @mitchelzoler
NASHVILLE, TENN. – By 2010, U.S. octogenarians with acute ischemic stroke received intravenous thrombolytic treatment about as often as younger patients, showing that a sharp, age-related disparity in thrombolytic use a decade before had disappeared, based on comprehensive national data.
The 2010 data from the Nationwide Inpatient Sample further showed that the sex-based disparity in treatment with intravenous tissue plasminogen activator (TPA) seen in 2000 resolved as well from 2000 to 2010, but other disparities worsened with declines during the period in use of TPA at rural hospitals relative to urban hospitals and at nonteaching hospitals, compared with teaching hospitals, Dr. Michelle P. Lin said at the International Stroke Conference.
Perhaps most importantly, the statistics showed a “dramatic” increase in TPA use among all age groups during the decade ending in 2010, when thrombolytic therapy was administered to 3.5%-3.9% of adult patients regardless of their age, said Dr. Lin of the department of neurology at the University of Southern California in Los Angeles. In 2000, U.S. patients received TPA at less than a third of that rate.
Low TPA use in 2000 among patients aged 80 or older in part reflected the low number of octogenarians enrolled in the trials that documented the safety and efficacy of TPA for acute ischemicstroke patients, Dr. Lin said at the International Stroke Conference, sponsored by the American Heart Association.
Data from the National Inpatient Sample included information on the treatment received by 5,932,175 patients with acute ischemic stroke at more than 1,000 U.S. hospitals during 2000-2010. The age breakdown of the nearly 6 millionpatients showed 28% were aged 18-64 years, 37% were65-79, and 35% were 80 years or older.
In 2000, medical staffs administered intravenous treatment with TPA to 1.02% of these patients aged 18-64 years, 0.92% of patients aged 65-79 years, and 0.47% of patients aged 80 or older. By 2010, the annual rates of TPA use ran 3.61% in those 18-64 years, 3.87% among those 65-79 years, and 3.55% in patients 80 years or older. In an adjusted analysis, this translated into a greater than threefold increase in TPA use among the 18- to 64-year-olds, a nearly fourfold rise in patients 65-79 years, and a nearly sevenfold jump among those 80 or older, a 24% average annual increased rate among the oldest patients, who averaged 86 years old, Dr. Lin reported.
The data also showed that among the octogenarians during 2000-2005, TPA use in women lagged behind use in men by a relative 15%, but this completely disappeared during 2006-2010, when usage rates in men and women evened out. TPA use among African Americans, Hispanics, and Asians, compared with whites, remained significantly below the rate in whites throughout the decade, although the extent of the disparity narrowed for African Americans and Asians during the second half of the decade, compared with the first half.
Dr. Lin and her associates also analyzed TPA use relative to the demographic setting of the hospital and its teaching status. During 2000-2010, the relative usage of TPA at rural hospitals, compared with urban hospitals, fell from 65% of the comparator level to 36%. Among nonteaching hospitals, the rate of TPA use dropped from 52% of the teaching hospitals’ level to 49%.
Dr. Lin said she had no relevant financial disclosures.
[email protected]
On Twitter @mitchelzoler
NASHVILLE, TENN. – By 2010, U.S. octogenarians with acute ischemic stroke received intravenous thrombolytic treatment about as often as younger patients, showing that a sharp, age-related disparity in thrombolytic use a decade before had disappeared, based on comprehensive national data.
The 2010 data from the Nationwide Inpatient Sample further showed that the sex-based disparity in treatment with intravenous tissue plasminogen activator (TPA) seen in 2000 resolved as well from 2000 to 2010, but other disparities worsened with declines during the period in use of TPA at rural hospitals relative to urban hospitals and at nonteaching hospitals, compared with teaching hospitals, Dr. Michelle P. Lin said at the International Stroke Conference.
Perhaps most importantly, the statistics showed a “dramatic” increase in TPA use among all age groups during the decade ending in 2010, when thrombolytic therapy was administered to 3.5%-3.9% of adult patients regardless of their age, said Dr. Lin of the department of neurology at the University of Southern California in Los Angeles. In 2000, U.S. patients received TPA at less than a third of that rate.
Low TPA use in 2000 among patients aged 80 or older in part reflected the low number of octogenarians enrolled in the trials that documented the safety and efficacy of TPA for acute ischemicstroke patients, Dr. Lin said at the International Stroke Conference, sponsored by the American Heart Association.
Data from the National Inpatient Sample included information on the treatment received by 5,932,175 patients with acute ischemic stroke at more than 1,000 U.S. hospitals during 2000-2010. The age breakdown of the nearly 6 millionpatients showed 28% were aged 18-64 years, 37% were65-79, and 35% were 80 years or older.
In 2000, medical staffs administered intravenous treatment with TPA to 1.02% of these patients aged 18-64 years, 0.92% of patients aged 65-79 years, and 0.47% of patients aged 80 or older. By 2010, the annual rates of TPA use ran 3.61% in those 18-64 years, 3.87% among those 65-79 years, and 3.55% in patients 80 years or older. In an adjusted analysis, this translated into a greater than threefold increase in TPA use among the 18- to 64-year-olds, a nearly fourfold rise in patients 65-79 years, and a nearly sevenfold jump among those 80 or older, a 24% average annual increased rate among the oldest patients, who averaged 86 years old, Dr. Lin reported.
The data also showed that among the octogenarians during 2000-2005, TPA use in women lagged behind use in men by a relative 15%, but this completely disappeared during 2006-2010, when usage rates in men and women evened out. TPA use among African Americans, Hispanics, and Asians, compared with whites, remained significantly below the rate in whites throughout the decade, although the extent of the disparity narrowed for African Americans and Asians during the second half of the decade, compared with the first half.
Dr. Lin and her associates also analyzed TPA use relative to the demographic setting of the hospital and its teaching status. During 2000-2010, the relative usage of TPA at rural hospitals, compared with urban hospitals, fell from 65% of the comparator level to 36%. Among nonteaching hospitals, the rate of TPA use dropped from 52% of the teaching hospitals’ level to 49%.
Dr. Lin said she had no relevant financial disclosures.
[email protected]
On Twitter @mitchelzoler
AT THE INTERNATIONAL STROKE CONFERENCE
Key clinical point: By 2010, U.S. ischemic stroke patients aged 80 or older received thrombolytic treatment as often as younger patients.
Major finding: In 2010, thrombolysis was used to treat 3.55% of U.S. stroke patients 80 years or older, 3.87% of those 65-79, and 3.61% of those 18-64.
Data source: The U.S. National Inpatient Sample for 2000-2010, which included 5,932,175 adults with acute ischemic stroke.
Disclosures: Dr. Lin said she had no relevant financial disclosures.
No benefit from amonafide for patients with secondary AML
For patients with secondary acute myeloid leukemia, induction therapy with cytarabine in combination with amonafide L-malate did not improve complete remission rates over the standard treatment of cytarabine and daunorubicin, investigators reported online March 2 in the Journal of Clinical Oncology.
Between January 2008 and August 2010, 433 patients with previously untreated secondary AML were randomly assigned to receive an intravenous infusion of cytarabine once per day for 7 days, plus either amonafide or daunorubicin for 4 days. The complete remission rate was 46% in the amonafide-plus-cytarabine group and 45% in the daunorubicin-plus-cytarabine group (P = .81), reported Dr. Richard M. Stone of Dana-Farber Cancer Institute, Boston, and associates.
“Although amonafide does not seem to provide benefit over standard induction therapy with daunorubicin, analyses of the data from this clinical trial may provide critical background information for tests of subsequent drugs that may have promise in this important disease subgroup, such as the liposomal-encapsulated daunorubicin/cytarabine CPX-351, the nuclear export inhibitor KPT-330, or the BCL-2 antagonist ABT-199,” they concluded.
Read the complete article here: (doi:10.1200/JCO.2014.57.0952).
For patients with secondary acute myeloid leukemia, induction therapy with cytarabine in combination with amonafide L-malate did not improve complete remission rates over the standard treatment of cytarabine and daunorubicin, investigators reported online March 2 in the Journal of Clinical Oncology.
Between January 2008 and August 2010, 433 patients with previously untreated secondary AML were randomly assigned to receive an intravenous infusion of cytarabine once per day for 7 days, plus either amonafide or daunorubicin for 4 days. The complete remission rate was 46% in the amonafide-plus-cytarabine group and 45% in the daunorubicin-plus-cytarabine group (P = .81), reported Dr. Richard M. Stone of Dana-Farber Cancer Institute, Boston, and associates.
“Although amonafide does not seem to provide benefit over standard induction therapy with daunorubicin, analyses of the data from this clinical trial may provide critical background information for tests of subsequent drugs that may have promise in this important disease subgroup, such as the liposomal-encapsulated daunorubicin/cytarabine CPX-351, the nuclear export inhibitor KPT-330, or the BCL-2 antagonist ABT-199,” they concluded.
Read the complete article here: (doi:10.1200/JCO.2014.57.0952).
For patients with secondary acute myeloid leukemia, induction therapy with cytarabine in combination with amonafide L-malate did not improve complete remission rates over the standard treatment of cytarabine and daunorubicin, investigators reported online March 2 in the Journal of Clinical Oncology.
Between January 2008 and August 2010, 433 patients with previously untreated secondary AML were randomly assigned to receive an intravenous infusion of cytarabine once per day for 7 days, plus either amonafide or daunorubicin for 4 days. The complete remission rate was 46% in the amonafide-plus-cytarabine group and 45% in the daunorubicin-plus-cytarabine group (P = .81), reported Dr. Richard M. Stone of Dana-Farber Cancer Institute, Boston, and associates.
“Although amonafide does not seem to provide benefit over standard induction therapy with daunorubicin, analyses of the data from this clinical trial may provide critical background information for tests of subsequent drugs that may have promise in this important disease subgroup, such as the liposomal-encapsulated daunorubicin/cytarabine CPX-351, the nuclear export inhibitor KPT-330, or the BCL-2 antagonist ABT-199,” they concluded.
Read the complete article here: (doi:10.1200/JCO.2014.57.0952).
General anesthesia linked to worsened stroke outcomes
NASHVILLE, TENN. – When acute ischemic stroke patients undergo an emergency endovascular procedure is it best done with general anesthesia or nongeneral anesthesia?
A post hoc analysis of data collected by a Dutch randomized, controlled trial of intra-arterial therapy suggested that nongeneral anesthesia was associated with substantially better patient outcomes, and the findings convinced the Dutch investigators who ran the study to stick with nongeneral anesthesia as their default approach.
In MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands) (N. Engl. J. Med. 2015;372:11-20), 216 acute ischemic stroke patients underwent intra-arterial treatment following randomization. Among these patients, 79 were treated with general anesthesia and 137 with nongeneral anesthesia. The anesthesia choice was made on a case-by-case basis by each participating interventionalist.
The study’s primary endpoint – 90-day status rated as a good function based on a modified Rankin sale score of 0-2 – occurred in 38% of the intra-arterial patients treated with nongeneral anesthesia, 23% of the intra-arterial patients treated with general anesthesia, and 19% among the control patients who received standard treatment without intra-arterial intervention.
“The effect on outcome that we found with intra-arterial treatment in MR CLEAN was not observed in the subgroup of patients treated with general anesthesia,” said Dr. Olvert A. Berkhemer at the International Stroke Conference. The analysis also showed that patients in the general and nongeneral anesthesia subgroups had similar stroke severity as measured by their National Institutes of Health Stroke Scale score, said Dr. Berkhemer, a researcher at the Academic Medical Center in Amsterdam.
But U.S. stroke specialists who heard the report cautioned that unidentified confounders might explain the results, and they also expressed skepticism that the Dutch observations would deter U.S. interventionalists from continuing to use general anesthesia when they perform endovascular procedures.
“The big concern [about this analysis] is that there may have been some things about the general anesthesia patients that they did not account for. I suspect there is a huge bias, that general anesthesia patients were sicker,” said Dr. Bruce Ovbiagele, professor and chief of neurology at the Medical University of South Carolina in Charleston. “At my institution they have used nongeneral anesthesia, but I have been at other places where they usually use general anesthesia; it is variable,” Dr. Ovbiagele added.
Dr. Berkhemer’s analysis also showed that general anesthesia linked with a delayed start to treatment, but without resulting in a significant difference in time to reperfusion. He noted that “sometimes you cannot do the procedure without general anesthesia,” and in MR CLEAN 6 of the 137 intra-arterial patients who started with nongeneral anesthesia eventually received general anesthesia because of discomfort and pain, Dr. Berkhemer said at the conference, sponsored by the American Heart Association.
He speculated that patients who did not receive general anesthesia did better because they did not undergo acute episodes of reduced blood pressure caused by the hypotensive effect of general anesthesia.
The findings reinforced the approach already used in most of the Dutch centers that participated in MR CLEAN, where nongeneral anesthesia is preferred when possible. “In our center we always use nongeneral anesthesia, we are happy with that and we are not going to change,” said Dr. Diederik W.J. Dippel, lead investigator of MR CLEAN and professor of neurology at Erasmus University Medical Center in Rotterdam, The Netherlands. This prejudice against general anesthesia would make it hard to run a trial in The Netherlands that matched the two anesthesia approaches against each other, Dr. Dippel added.
On Twitter @mitchelzoler
Results from prior analyses had also shown better outcomes of acute ischemic stroke patients undergoing endovascular intervention when they avoided general anesthesia. A notable feature of Dr. Berkhemer’s analysis was that the stroke severity levels were well balanced between the patients who received general anesthesia and those who did not. But many other factors aside from stroke severity can affect whether or not a patient receives general anesthesia.

|
Dr. Larry B. Goldstein |
Although the outcome differences seen in this analysis were striking, many factors could have contributed. Patients received different drugs, patients may have had widely divergent clinical states despite their similar stroke severity, and the people performing the procedures were different. The many variables make it very hard to pinpoint the cause of the different outcomes.
At my institution, Duke, the interventionalists who work on acute ischemic stroke patients almost exclusively use general anesthesia. That’s because of their concern about how patients will act during a very delicate procedure. For example, stroke patients can have an aphasia that makes it hard for them to respond to requests to do something specific during the procedure. I am very skeptical that my colleagues will decide to switch to using no general anesthesia based on the results of this new analysis.
I agree with the Dutch investigators that a randomized, controlled trial of general anesthesia or no general anesthesia would be very hard to perform because individual interventionalists would need to believe there is equipoise between the two anesthesia approaches. Most interventionalists right now probably believe the approach they have always used remains best and so would be unwilling to participate in a randomized controlled trial.
Dr. Larry B. Goldstein is professor of neurology and chief of the stroke center at Duke University in Durham, N.C. He had no relevant disclosures. He made these comments in an interview.
Results from prior analyses had also shown better outcomes of acute ischemic stroke patients undergoing endovascular intervention when they avoided general anesthesia. A notable feature of Dr. Berkhemer’s analysis was that the stroke severity levels were well balanced between the patients who received general anesthesia and those who did not. But many other factors aside from stroke severity can affect whether or not a patient receives general anesthesia.

|
Dr. Larry B. Goldstein |
Although the outcome differences seen in this analysis were striking, many factors could have contributed. Patients received different drugs, patients may have had widely divergent clinical states despite their similar stroke severity, and the people performing the procedures were different. The many variables make it very hard to pinpoint the cause of the different outcomes.
At my institution, Duke, the interventionalists who work on acute ischemic stroke patients almost exclusively use general anesthesia. That’s because of their concern about how patients will act during a very delicate procedure. For example, stroke patients can have an aphasia that makes it hard for them to respond to requests to do something specific during the procedure. I am very skeptical that my colleagues will decide to switch to using no general anesthesia based on the results of this new analysis.
I agree with the Dutch investigators that a randomized, controlled trial of general anesthesia or no general anesthesia would be very hard to perform because individual interventionalists would need to believe there is equipoise between the two anesthesia approaches. Most interventionalists right now probably believe the approach they have always used remains best and so would be unwilling to participate in a randomized controlled trial.
Dr. Larry B. Goldstein is professor of neurology and chief of the stroke center at Duke University in Durham, N.C. He had no relevant disclosures. He made these comments in an interview.
Results from prior analyses had also shown better outcomes of acute ischemic stroke patients undergoing endovascular intervention when they avoided general anesthesia. A notable feature of Dr. Berkhemer’s analysis was that the stroke severity levels were well balanced between the patients who received general anesthesia and those who did not. But many other factors aside from stroke severity can affect whether or not a patient receives general anesthesia.

|
Dr. Larry B. Goldstein |
Although the outcome differences seen in this analysis were striking, many factors could have contributed. Patients received different drugs, patients may have had widely divergent clinical states despite their similar stroke severity, and the people performing the procedures were different. The many variables make it very hard to pinpoint the cause of the different outcomes.
At my institution, Duke, the interventionalists who work on acute ischemic stroke patients almost exclusively use general anesthesia. That’s because of their concern about how patients will act during a very delicate procedure. For example, stroke patients can have an aphasia that makes it hard for them to respond to requests to do something specific during the procedure. I am very skeptical that my colleagues will decide to switch to using no general anesthesia based on the results of this new analysis.
I agree with the Dutch investigators that a randomized, controlled trial of general anesthesia or no general anesthesia would be very hard to perform because individual interventionalists would need to believe there is equipoise between the two anesthesia approaches. Most interventionalists right now probably believe the approach they have always used remains best and so would be unwilling to participate in a randomized controlled trial.
Dr. Larry B. Goldstein is professor of neurology and chief of the stroke center at Duke University in Durham, N.C. He had no relevant disclosures. He made these comments in an interview.
NASHVILLE, TENN. – When acute ischemic stroke patients undergo an emergency endovascular procedure is it best done with general anesthesia or nongeneral anesthesia?
A post hoc analysis of data collected by a Dutch randomized, controlled trial of intra-arterial therapy suggested that nongeneral anesthesia was associated with substantially better patient outcomes, and the findings convinced the Dutch investigators who ran the study to stick with nongeneral anesthesia as their default approach.
In MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands) (N. Engl. J. Med. 2015;372:11-20), 216 acute ischemic stroke patients underwent intra-arterial treatment following randomization. Among these patients, 79 were treated with general anesthesia and 137 with nongeneral anesthesia. The anesthesia choice was made on a case-by-case basis by each participating interventionalist.
The study’s primary endpoint – 90-day status rated as a good function based on a modified Rankin sale score of 0-2 – occurred in 38% of the intra-arterial patients treated with nongeneral anesthesia, 23% of the intra-arterial patients treated with general anesthesia, and 19% among the control patients who received standard treatment without intra-arterial intervention.
“The effect on outcome that we found with intra-arterial treatment in MR CLEAN was not observed in the subgroup of patients treated with general anesthesia,” said Dr. Olvert A. Berkhemer at the International Stroke Conference. The analysis also showed that patients in the general and nongeneral anesthesia subgroups had similar stroke severity as measured by their National Institutes of Health Stroke Scale score, said Dr. Berkhemer, a researcher at the Academic Medical Center in Amsterdam.
But U.S. stroke specialists who heard the report cautioned that unidentified confounders might explain the results, and they also expressed skepticism that the Dutch observations would deter U.S. interventionalists from continuing to use general anesthesia when they perform endovascular procedures.
“The big concern [about this analysis] is that there may have been some things about the general anesthesia patients that they did not account for. I suspect there is a huge bias, that general anesthesia patients were sicker,” said Dr. Bruce Ovbiagele, professor and chief of neurology at the Medical University of South Carolina in Charleston. “At my institution they have used nongeneral anesthesia, but I have been at other places where they usually use general anesthesia; it is variable,” Dr. Ovbiagele added.
Dr. Berkhemer’s analysis also showed that general anesthesia linked with a delayed start to treatment, but without resulting in a significant difference in time to reperfusion. He noted that “sometimes you cannot do the procedure without general anesthesia,” and in MR CLEAN 6 of the 137 intra-arterial patients who started with nongeneral anesthesia eventually received general anesthesia because of discomfort and pain, Dr. Berkhemer said at the conference, sponsored by the American Heart Association.
He speculated that patients who did not receive general anesthesia did better because they did not undergo acute episodes of reduced blood pressure caused by the hypotensive effect of general anesthesia.
The findings reinforced the approach already used in most of the Dutch centers that participated in MR CLEAN, where nongeneral anesthesia is preferred when possible. “In our center we always use nongeneral anesthesia, we are happy with that and we are not going to change,” said Dr. Diederik W.J. Dippel, lead investigator of MR CLEAN and professor of neurology at Erasmus University Medical Center in Rotterdam, The Netherlands. This prejudice against general anesthesia would make it hard to run a trial in The Netherlands that matched the two anesthesia approaches against each other, Dr. Dippel added.
On Twitter @mitchelzoler
NASHVILLE, TENN. – When acute ischemic stroke patients undergo an emergency endovascular procedure is it best done with general anesthesia or nongeneral anesthesia?
A post hoc analysis of data collected by a Dutch randomized, controlled trial of intra-arterial therapy suggested that nongeneral anesthesia was associated with substantially better patient outcomes, and the findings convinced the Dutch investigators who ran the study to stick with nongeneral anesthesia as their default approach.
In MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands) (N. Engl. J. Med. 2015;372:11-20), 216 acute ischemic stroke patients underwent intra-arterial treatment following randomization. Among these patients, 79 were treated with general anesthesia and 137 with nongeneral anesthesia. The anesthesia choice was made on a case-by-case basis by each participating interventionalist.
The study’s primary endpoint – 90-day status rated as a good function based on a modified Rankin sale score of 0-2 – occurred in 38% of the intra-arterial patients treated with nongeneral anesthesia, 23% of the intra-arterial patients treated with general anesthesia, and 19% among the control patients who received standard treatment without intra-arterial intervention.
“The effect on outcome that we found with intra-arterial treatment in MR CLEAN was not observed in the subgroup of patients treated with general anesthesia,” said Dr. Olvert A. Berkhemer at the International Stroke Conference. The analysis also showed that patients in the general and nongeneral anesthesia subgroups had similar stroke severity as measured by their National Institutes of Health Stroke Scale score, said Dr. Berkhemer, a researcher at the Academic Medical Center in Amsterdam.
But U.S. stroke specialists who heard the report cautioned that unidentified confounders might explain the results, and they also expressed skepticism that the Dutch observations would deter U.S. interventionalists from continuing to use general anesthesia when they perform endovascular procedures.
“The big concern [about this analysis] is that there may have been some things about the general anesthesia patients that they did not account for. I suspect there is a huge bias, that general anesthesia patients were sicker,” said Dr. Bruce Ovbiagele, professor and chief of neurology at the Medical University of South Carolina in Charleston. “At my institution they have used nongeneral anesthesia, but I have been at other places where they usually use general anesthesia; it is variable,” Dr. Ovbiagele added.
Dr. Berkhemer’s analysis also showed that general anesthesia linked with a delayed start to treatment, but without resulting in a significant difference in time to reperfusion. He noted that “sometimes you cannot do the procedure without general anesthesia,” and in MR CLEAN 6 of the 137 intra-arterial patients who started with nongeneral anesthesia eventually received general anesthesia because of discomfort and pain, Dr. Berkhemer said at the conference, sponsored by the American Heart Association.
He speculated that patients who did not receive general anesthesia did better because they did not undergo acute episodes of reduced blood pressure caused by the hypotensive effect of general anesthesia.
The findings reinforced the approach already used in most of the Dutch centers that participated in MR CLEAN, where nongeneral anesthesia is preferred when possible. “In our center we always use nongeneral anesthesia, we are happy with that and we are not going to change,” said Dr. Diederik W.J. Dippel, lead investigator of MR CLEAN and professor of neurology at Erasmus University Medical Center in Rotterdam, The Netherlands. This prejudice against general anesthesia would make it hard to run a trial in The Netherlands that matched the two anesthesia approaches against each other, Dr. Dippel added.
On Twitter @mitchelzoler
AT THE INTERNATIONAL STROKE CONFERENCE
Key clinical point: Post hoc analysis of acute ischemic stroke patients treated intra-arterially suggests that avoiding general anesthesia produces better long-term outcomes.
Major finding: After 90 days, 38% of patients treated without general anesthesia and 23% of those who received general anesthesia had good outcomes.
Data source: Analysis of 216 patients in the Dutch MR CLEAN trial who underwent treatment for acute ischemic stroke.
Disclosures: Dr. Berkhemer, Dr. Dippel, and Dr. Ovbiagele had no disclosures.
LISTEN NOW: David Pressel, MD, PHD, FHM, discusses violence in hospitals
DAVID PRESSEL, MD, PHD, FHM, medical director of inpatient services at Nemours Children’s Health System, talks about the nature of violence in hospitals and a training program he has helped put into place at his center.
DAVID PRESSEL, MD, PHD, FHM, medical director of inpatient services at Nemours Children’s Health System, talks about the nature of violence in hospitals and a training program he has helped put into place at his center.
DAVID PRESSEL, MD, PHD, FHM, medical director of inpatient services at Nemours Children’s Health System, talks about the nature of violence in hospitals and a training program he has helped put into place at his center.
LISTEN NOW: David Lichtman, PA, explains factors to determine when hospitalists perform procedures
DAVID LICHTMAN, PA, a hospitalist and director of the Johns Hopkins Central Procedure Service, explains the complicated set of factors used by
individual hospitals to determine which procedures fall under the scope of their HM practitioners.
DAVID LICHTMAN, PA, a hospitalist and director of the Johns Hopkins Central Procedure Service, explains the complicated set of factors used by
individual hospitals to determine which procedures fall under the scope of their HM practitioners.
DAVID LICHTMAN, PA, a hospitalist and director of the Johns Hopkins Central Procedure Service, explains the complicated set of factors used by
individual hospitals to determine which procedures fall under the scope of their HM practitioners.
LISTEN NOW: Monal Shah, MD, discusses exceptions for VTE admissions
Although patients with blood clots are oftentimes not admitted to the hospital, there are some exceptions. Monal Shah, MD, physician advisor for Parkland Hospital in Dallas, Texas, and the former section head of hospital medicine at the University of Texas Southwestern Medical Center, discusses some exceptions.
Although patients with blood clots are oftentimes not admitted to the hospital, there are some exceptions. Monal Shah, MD, physician advisor for Parkland Hospital in Dallas, Texas, and the former section head of hospital medicine at the University of Texas Southwestern Medical Center, discusses some exceptions.
Although patients with blood clots are oftentimes not admitted to the hospital, there are some exceptions. Monal Shah, MD, physician advisor for Parkland Hospital in Dallas, Texas, and the former section head of hospital medicine at the University of Texas Southwestern Medical Center, discusses some exceptions.
LISTEN NOW: Adam E. Fall, MD, SFHM, discusses VTE management in an era of technology
Adam E. Fall, MD, SFHM, who recently worked as the senior regional medical director for hospital medicine at TeamHealth in Chattanooga, Tenn., discusses the importance of allowing nuance and gradation to govern patient care in the treatment of thromboembolism in an era where hospitalists can be overly reliant on electronic medical records.
Adam E. Fall, MD, SFHM, who recently worked as the senior regional medical director for hospital medicine at TeamHealth in Chattanooga, Tenn., discusses the importance of allowing nuance and gradation to govern patient care in the treatment of thromboembolism in an era where hospitalists can be overly reliant on electronic medical records.
Adam E. Fall, MD, SFHM, who recently worked as the senior regional medical director for hospital medicine at TeamHealth in Chattanooga, Tenn., discusses the importance of allowing nuance and gradation to govern patient care in the treatment of thromboembolism in an era where hospitalists can be overly reliant on electronic medical records.
Tip-Top Tactics for Bedside Procedure Training
David Lichtman, PA, director of the Johns Hopkins Central Procedure Service in Baltimore, Md., says bedside procedure training should be consistent and thorough, regardless of whether the trainee is a medical student, a resident, a fellow, or an established physician. He is a strong advocate for training that includes well-designed computer coursework, evaluates practitioners from start to finish, and demonstrates that they are meeting established benchmarks.
“That’s what keeps patients safe,” he says.
Experienced, capable, and proven educators are also critical.
“Let’s face it: Not everybody is a very good teacher,” he adds.
Currently, many medical residents can do rotations that will give them hands-on experience. But some physicians question why certain procedures are still being taught to internal medicine residents if the ABIM no longer requires hands-on experience. Other programs may simply lack the resources, including experienced supervisors, to provide proper training.
The demand for more training is clearly there, however. Sally Wang, MD, FHM, director of procedure education at Brigham and Women’s Hospital and a clinical instructor at Harvard Medical School in Boston, co-leads the procedures pre-course at the SHM annual meeting. She compares the logistically complicated event to throwing a wedding. It consistently sells out despite having doubled in size, to 60 slots for a basic procedure course and 60 slots for a second course that emphasizes ultrasound. At the HM14 pre-course in Las Vegas, Dr. Wang counted enough people on the waiting list to fill an additional course.
“It was mind-boggling,” she says.
Several companies have taken notice of the pent-up demand and are offering their own courses and workshops. Joshua Lenchus, DO, RPh, FACP, SFHM, medical director of the University of Miami-Jackson Memorial Hospital’s procedure service, and others see many of these offerings as introductions only, however. At the University of Miami, he says, his rigorous, simulation-based invasive bedside procedures curriculum is mandatory for all internal medicine residents. The curriculum includes central line, thoracentesis, paracentesis, lumbar puncture, and sometimes arthrocentesis as its core procedures, though Dr. Lenchus says others can easily be added. This fall, for instance, he plans to add chest tube and arterial line placement.
He notes a dramatic reduction in central line placement and thoracentesis complications after his team began performing them to the “four pillars” of his program. Rigorous simulation-based training, strict adherence to a critical skills checklist, consistent use of ultrasound, and direct supervision can form a very effective bundle of safety measures, he says, just like a combination of seat belts, speed reduction, and other precautions can lead to fewer automobile-associated injuries and deaths. TH
David Lichtman, PA, director of the Johns Hopkins Central Procedure Service in Baltimore, Md., says bedside procedure training should be consistent and thorough, regardless of whether the trainee is a medical student, a resident, a fellow, or an established physician. He is a strong advocate for training that includes well-designed computer coursework, evaluates practitioners from start to finish, and demonstrates that they are meeting established benchmarks.
“That’s what keeps patients safe,” he says.
Experienced, capable, and proven educators are also critical.
“Let’s face it: Not everybody is a very good teacher,” he adds.
Currently, many medical residents can do rotations that will give them hands-on experience. But some physicians question why certain procedures are still being taught to internal medicine residents if the ABIM no longer requires hands-on experience. Other programs may simply lack the resources, including experienced supervisors, to provide proper training.
The demand for more training is clearly there, however. Sally Wang, MD, FHM, director of procedure education at Brigham and Women’s Hospital and a clinical instructor at Harvard Medical School in Boston, co-leads the procedures pre-course at the SHM annual meeting. She compares the logistically complicated event to throwing a wedding. It consistently sells out despite having doubled in size, to 60 slots for a basic procedure course and 60 slots for a second course that emphasizes ultrasound. At the HM14 pre-course in Las Vegas, Dr. Wang counted enough people on the waiting list to fill an additional course.
“It was mind-boggling,” she says.
Several companies have taken notice of the pent-up demand and are offering their own courses and workshops. Joshua Lenchus, DO, RPh, FACP, SFHM, medical director of the University of Miami-Jackson Memorial Hospital’s procedure service, and others see many of these offerings as introductions only, however. At the University of Miami, he says, his rigorous, simulation-based invasive bedside procedures curriculum is mandatory for all internal medicine residents. The curriculum includes central line, thoracentesis, paracentesis, lumbar puncture, and sometimes arthrocentesis as its core procedures, though Dr. Lenchus says others can easily be added. This fall, for instance, he plans to add chest tube and arterial line placement.
He notes a dramatic reduction in central line placement and thoracentesis complications after his team began performing them to the “four pillars” of his program. Rigorous simulation-based training, strict adherence to a critical skills checklist, consistent use of ultrasound, and direct supervision can form a very effective bundle of safety measures, he says, just like a combination of seat belts, speed reduction, and other precautions can lead to fewer automobile-associated injuries and deaths. TH
David Lichtman, PA, director of the Johns Hopkins Central Procedure Service in Baltimore, Md., says bedside procedure training should be consistent and thorough, regardless of whether the trainee is a medical student, a resident, a fellow, or an established physician. He is a strong advocate for training that includes well-designed computer coursework, evaluates practitioners from start to finish, and demonstrates that they are meeting established benchmarks.
“That’s what keeps patients safe,” he says.
Experienced, capable, and proven educators are also critical.
“Let’s face it: Not everybody is a very good teacher,” he adds.
Currently, many medical residents can do rotations that will give them hands-on experience. But some physicians question why certain procedures are still being taught to internal medicine residents if the ABIM no longer requires hands-on experience. Other programs may simply lack the resources, including experienced supervisors, to provide proper training.
The demand for more training is clearly there, however. Sally Wang, MD, FHM, director of procedure education at Brigham and Women’s Hospital and a clinical instructor at Harvard Medical School in Boston, co-leads the procedures pre-course at the SHM annual meeting. She compares the logistically complicated event to throwing a wedding. It consistently sells out despite having doubled in size, to 60 slots for a basic procedure course and 60 slots for a second course that emphasizes ultrasound. At the HM14 pre-course in Las Vegas, Dr. Wang counted enough people on the waiting list to fill an additional course.
“It was mind-boggling,” she says.
Several companies have taken notice of the pent-up demand and are offering their own courses and workshops. Joshua Lenchus, DO, RPh, FACP, SFHM, medical director of the University of Miami-Jackson Memorial Hospital’s procedure service, and others see many of these offerings as introductions only, however. At the University of Miami, he says, his rigorous, simulation-based invasive bedside procedures curriculum is mandatory for all internal medicine residents. The curriculum includes central line, thoracentesis, paracentesis, lumbar puncture, and sometimes arthrocentesis as its core procedures, though Dr. Lenchus says others can easily be added. This fall, for instance, he plans to add chest tube and arterial line placement.
He notes a dramatic reduction in central line placement and thoracentesis complications after his team began performing them to the “four pillars” of his program. Rigorous simulation-based training, strict adherence to a critical skills checklist, consistent use of ultrasound, and direct supervision can form a very effective bundle of safety measures, he says, just like a combination of seat belts, speed reduction, and other precautions can lead to fewer automobile-associated injuries and deaths. TH
‘Biodegradable’ CAR may aid transplant in AML

Photo courtesy of
BMT Tandem Meetings
SAN DIEGO—Researchers have developed a “biodegradable” chimeric antigen receptor (CAR) T-cell therapy that could potentially serve as a preparative regimen for acute myeloid leukemia (AML) patients undergoing allogeneic transplant.
The team created CAR T cells that target CD33 (CART33) and modified them with RNA so the cells would stop expressing CARs over time.
In mouse models of AML, the RNA-CART33 cells had an antileukemic effect and induced myeloablation.
The cells also stopped expressing CARs by the 2-week mark, which would allow for engraftment after allogeneic transplant, according to the researchers.
Saad S. Kenderian, MD, of the University of Pennsylvania in Philadelphia, presented this research at the 2015 BMT Tandem Meetings as one of the meeting’s “Best Abstracts” (abstract 1). The research was funded by Novartis.
“Allogeneic transplantation is the only potentially curative option in relapsed/refractory AML,” Dr Kenderian noted. “Outcomes are poor if patients are transplanted in residual disease . . . , and these patients are often considered transplant-ineligible. Therefore, novel therapies are desperately needed.”
With this in mind, Dr Kenderian and his colleagues set out to develop a CAR T-cell therapy targeting CD33, which is expressed on AML blasts.
The researchers created a CAR from the anti-CD33 single-chain fragment variable of gemtuzumab ozogamicin, 41BB costimulation, CD3ζ signaling domain, and a lentiviral (LV) vector. They transduced T cells with this construct and expanded them in culture using anti-CD3/CD28 magnetic beads.
The team then tested these CART33 cells in NSGS mice engrafted with primary AML blasts. The mice received CART33 cells, another CAR T-cell therapy known as CART123, or control T cells.
At 4 weeks, mice that had received CART33 or CART123 cells were entirely leukemia-free, but the disease continued to progress in mice that received control T cells.
Likewise, when the experiment ended at 200 days, survival was 100% among mice that received CART33 or CART123, but all of the control mice had died. And at 200 days, CAR T cells were still circulating in the CART33- and CART123-treated mice.
Next, the researchers administered CART33 cells to HIS-NSG mice engrafted with human bone marrow and found the treatment resulted in myeloablation. There was a significant reduction of CD34-positive cells in mice that received CART33 compared to mice that received control T cells or no treatment.
“So based on our preclinical data, when we treat refractory AML with lentivirally transduced CART33, that will result in myeloablation, eradication of AML, and persistence of these CARs,” Dr Kenderian said.
“If allogeneic transplantation is performed at this aplastic stage, it will likely lead to rejection of the graft by persisting CAR therapy, which also means that elimination of CARs is necessary prior to stem cell infusion.”
So the researchers decided to create a transiently expressed, mRNA-modified CAR based on CART33. They electroporated T cells with this construct, and the cells expressed CARs for up to 6 days.
In experiments with the MOLM14 cell line, RNA-modified CART33 cells exhibited transient but comparable killing ability as LV-transduced CART33.
The researchers then tested RNA-CART33 in combination with chemotherapy in vivo. They transplanted NSG mice with MOLM14 and treated them with cyclophosphamide plus RNA-CART33 or cyclophosphamide plus control T cells.
Combination RNA-CART33 and chemotherapy prompted stronger, more durable antileukemic activity than cyclophosphamide and control T cells. Furthermore, there was a significant improvement in survival among RNA-CART33-treated mice (P=0.01).
Finally, Dr Kenderian and his colleagues tested the effect of RNA-CART33 on hematopoiesis. The team treated NSGS mice with busulfan and transplanted them with T-cell-depleted bone marrow. Following engraftment, mice received RNA-CART33 cells, LV-CART33 cells, or control T cells.
The researchers followed the mice for 2 weeks and found that both RNA-CART33 and LV-CART33 induced myeloablation. And at 14 days, LV-CART33-treated mice were still expressing CARs, but RNA-CART33-treated mice were not.
“Based on our preclinical data, if we treat refractory AML with RNA-modified CART33, that results in myeloablation, anti-AML activity, and biodegradable, non-persisting CARs,” Dr Kenderian summarized.
“If allogeneic transplantation follows at this stage, it will likely lead to engraftment. Therefore, we conclude from this study that RNA-CART33 could be incorporated in novel conditioning regimens and will be tested in pilot phase 1 studies.” ![]()

Photo courtesy of
BMT Tandem Meetings
SAN DIEGO—Researchers have developed a “biodegradable” chimeric antigen receptor (CAR) T-cell therapy that could potentially serve as a preparative regimen for acute myeloid leukemia (AML) patients undergoing allogeneic transplant.
The team created CAR T cells that target CD33 (CART33) and modified them with RNA so the cells would stop expressing CARs over time.
In mouse models of AML, the RNA-CART33 cells had an antileukemic effect and induced myeloablation.
The cells also stopped expressing CARs by the 2-week mark, which would allow for engraftment after allogeneic transplant, according to the researchers.
Saad S. Kenderian, MD, of the University of Pennsylvania in Philadelphia, presented this research at the 2015 BMT Tandem Meetings as one of the meeting’s “Best Abstracts” (abstract 1). The research was funded by Novartis.
“Allogeneic transplantation is the only potentially curative option in relapsed/refractory AML,” Dr Kenderian noted. “Outcomes are poor if patients are transplanted in residual disease . . . , and these patients are often considered transplant-ineligible. Therefore, novel therapies are desperately needed.”
With this in mind, Dr Kenderian and his colleagues set out to develop a CAR T-cell therapy targeting CD33, which is expressed on AML blasts.
The researchers created a CAR from the anti-CD33 single-chain fragment variable of gemtuzumab ozogamicin, 41BB costimulation, CD3ζ signaling domain, and a lentiviral (LV) vector. They transduced T cells with this construct and expanded them in culture using anti-CD3/CD28 magnetic beads.
The team then tested these CART33 cells in NSGS mice engrafted with primary AML blasts. The mice received CART33 cells, another CAR T-cell therapy known as CART123, or control T cells.
At 4 weeks, mice that had received CART33 or CART123 cells were entirely leukemia-free, but the disease continued to progress in mice that received control T cells.
Likewise, when the experiment ended at 200 days, survival was 100% among mice that received CART33 or CART123, but all of the control mice had died. And at 200 days, CAR T cells were still circulating in the CART33- and CART123-treated mice.
Next, the researchers administered CART33 cells to HIS-NSG mice engrafted with human bone marrow and found the treatment resulted in myeloablation. There was a significant reduction of CD34-positive cells in mice that received CART33 compared to mice that received control T cells or no treatment.
“So based on our preclinical data, when we treat refractory AML with lentivirally transduced CART33, that will result in myeloablation, eradication of AML, and persistence of these CARs,” Dr Kenderian said.
“If allogeneic transplantation is performed at this aplastic stage, it will likely lead to rejection of the graft by persisting CAR therapy, which also means that elimination of CARs is necessary prior to stem cell infusion.”
So the researchers decided to create a transiently expressed, mRNA-modified CAR based on CART33. They electroporated T cells with this construct, and the cells expressed CARs for up to 6 days.
In experiments with the MOLM14 cell line, RNA-modified CART33 cells exhibited transient but comparable killing ability as LV-transduced CART33.
The researchers then tested RNA-CART33 in combination with chemotherapy in vivo. They transplanted NSG mice with MOLM14 and treated them with cyclophosphamide plus RNA-CART33 or cyclophosphamide plus control T cells.
Combination RNA-CART33 and chemotherapy prompted stronger, more durable antileukemic activity than cyclophosphamide and control T cells. Furthermore, there was a significant improvement in survival among RNA-CART33-treated mice (P=0.01).
Finally, Dr Kenderian and his colleagues tested the effect of RNA-CART33 on hematopoiesis. The team treated NSGS mice with busulfan and transplanted them with T-cell-depleted bone marrow. Following engraftment, mice received RNA-CART33 cells, LV-CART33 cells, or control T cells.
The researchers followed the mice for 2 weeks and found that both RNA-CART33 and LV-CART33 induced myeloablation. And at 14 days, LV-CART33-treated mice were still expressing CARs, but RNA-CART33-treated mice were not.
“Based on our preclinical data, if we treat refractory AML with RNA-modified CART33, that results in myeloablation, anti-AML activity, and biodegradable, non-persisting CARs,” Dr Kenderian summarized.
“If allogeneic transplantation follows at this stage, it will likely lead to engraftment. Therefore, we conclude from this study that RNA-CART33 could be incorporated in novel conditioning regimens and will be tested in pilot phase 1 studies.” ![]()

Photo courtesy of
BMT Tandem Meetings
SAN DIEGO—Researchers have developed a “biodegradable” chimeric antigen receptor (CAR) T-cell therapy that could potentially serve as a preparative regimen for acute myeloid leukemia (AML) patients undergoing allogeneic transplant.
The team created CAR T cells that target CD33 (CART33) and modified them with RNA so the cells would stop expressing CARs over time.
In mouse models of AML, the RNA-CART33 cells had an antileukemic effect and induced myeloablation.
The cells also stopped expressing CARs by the 2-week mark, which would allow for engraftment after allogeneic transplant, according to the researchers.
Saad S. Kenderian, MD, of the University of Pennsylvania in Philadelphia, presented this research at the 2015 BMT Tandem Meetings as one of the meeting’s “Best Abstracts” (abstract 1). The research was funded by Novartis.
“Allogeneic transplantation is the only potentially curative option in relapsed/refractory AML,” Dr Kenderian noted. “Outcomes are poor if patients are transplanted in residual disease . . . , and these patients are often considered transplant-ineligible. Therefore, novel therapies are desperately needed.”
With this in mind, Dr Kenderian and his colleagues set out to develop a CAR T-cell therapy targeting CD33, which is expressed on AML blasts.
The researchers created a CAR from the anti-CD33 single-chain fragment variable of gemtuzumab ozogamicin, 41BB costimulation, CD3ζ signaling domain, and a lentiviral (LV) vector. They transduced T cells with this construct and expanded them in culture using anti-CD3/CD28 magnetic beads.
The team then tested these CART33 cells in NSGS mice engrafted with primary AML blasts. The mice received CART33 cells, another CAR T-cell therapy known as CART123, or control T cells.
At 4 weeks, mice that had received CART33 or CART123 cells were entirely leukemia-free, but the disease continued to progress in mice that received control T cells.
Likewise, when the experiment ended at 200 days, survival was 100% among mice that received CART33 or CART123, but all of the control mice had died. And at 200 days, CAR T cells were still circulating in the CART33- and CART123-treated mice.
Next, the researchers administered CART33 cells to HIS-NSG mice engrafted with human bone marrow and found the treatment resulted in myeloablation. There was a significant reduction of CD34-positive cells in mice that received CART33 compared to mice that received control T cells or no treatment.
“So based on our preclinical data, when we treat refractory AML with lentivirally transduced CART33, that will result in myeloablation, eradication of AML, and persistence of these CARs,” Dr Kenderian said.
“If allogeneic transplantation is performed at this aplastic stage, it will likely lead to rejection of the graft by persisting CAR therapy, which also means that elimination of CARs is necessary prior to stem cell infusion.”
So the researchers decided to create a transiently expressed, mRNA-modified CAR based on CART33. They electroporated T cells with this construct, and the cells expressed CARs for up to 6 days.
In experiments with the MOLM14 cell line, RNA-modified CART33 cells exhibited transient but comparable killing ability as LV-transduced CART33.
The researchers then tested RNA-CART33 in combination with chemotherapy in vivo. They transplanted NSG mice with MOLM14 and treated them with cyclophosphamide plus RNA-CART33 or cyclophosphamide plus control T cells.
Combination RNA-CART33 and chemotherapy prompted stronger, more durable antileukemic activity than cyclophosphamide and control T cells. Furthermore, there was a significant improvement in survival among RNA-CART33-treated mice (P=0.01).
Finally, Dr Kenderian and his colleagues tested the effect of RNA-CART33 on hematopoiesis. The team treated NSGS mice with busulfan and transplanted them with T-cell-depleted bone marrow. Following engraftment, mice received RNA-CART33 cells, LV-CART33 cells, or control T cells.
The researchers followed the mice for 2 weeks and found that both RNA-CART33 and LV-CART33 induced myeloablation. And at 14 days, LV-CART33-treated mice were still expressing CARs, but RNA-CART33-treated mice were not.
“Based on our preclinical data, if we treat refractory AML with RNA-modified CART33, that results in myeloablation, anti-AML activity, and biodegradable, non-persisting CARs,” Dr Kenderian summarized.
“If allogeneic transplantation follows at this stage, it will likely lead to engraftment. Therefore, we conclude from this study that RNA-CART33 could be incorporated in novel conditioning regimens and will be tested in pilot phase 1 studies.” ![]()