User login
Postcolonoscopy colorectal cancers had unique features
Postcolonoscopy colorectal cancers were more likely to arise in the proximal colon and to show microsatellite instability, according to the results of a retrospective population-based study of 168 adults with incident colorectal cancers.
In all, 64% of postcolonoscopy colorectal cancers were located in the proximal colon, compared with 44% of detected colorectal cancers (P = .016), reported Niloy Jewel Samadder, MD, of the University of Utah in Salt Lake City, together with his associates. Furthermore, microsatellite instability (MSI) was detected in 32% of postcolonoscopy colorectal cancers, versus 13% of detected colorectal cancers (P = .005). These findings may point to differences in the underlying biology of postcolonoscopy colorectal cancers and detected colorectal cancers, they said. Studies are needed “to determine if postcolonoscopy cancers arise through a specific genetic pathway that may accelerate neoplastic progression,” they wrote in Clinical Gastroenterology and Hepatology.
Postcolonoscopy colorectal cancers are a “small but clinically important subset of colorectal cancers” that are diagnosed after the patient has a colonoscopy in which no cancer is detected, the researchers noted. These cancers have an estimated global prevalence ranging from 3% to 9% and an estimated pooled prevalence of 3.7% (Am J Gastroenterol. 2014;109:1375-89). Risk factors for postcolonoscopy colorectal cancers include low adenoma detection rates, rural facilities, and care by physicians who are not gastroenterologists. However, tumor-specific and patient-specific factors, including location within the colon and superior survival, compared with detected cancers, raises the possibility of underlying molecular differences related to tumorigenesis, the researchers said.
To investigate this idea, they retrospectively analyzed data from residents of Utah between 50 and 80 years old who had a colonoscopy between, Feb. 15, 1995, and Jan. 31, 2009, at one of two large clinical facilities in Utah (Intermountain Healthcare or the University of Utah Health Sciences). Using a state population-based database, they merged medical information from these patients with cancer histories from the Utah Cancer Registry. This enabled them to compare all 84 postcolonoscopy colorectal cancers (defined as those detected within 6-60 months of colonoscopy) with tissue available for analysis with 84 detected colorectal cancers (detected within 6 months of a colonoscopy).
In the multivariable analysis, MSI was the only molecular feature that was significantly more frequent in postcolonoscopy versus detected colorectal cancers (odds ratio, 4.20; 95% confidence interval, 1.58-11.14). However, postcolonoscopy colorectal cancers were significantly more likely to be early stage (86% versus 69% for detected colorectal cancers; P = .040). Five-year survival did not significantly differ between the groups.
“The molecular signatures of postcolonoscopy colorectal cancers in our study overlap with those of sporadic MSI and serrated pathways, suggesting these mechanisms play a disproportionate role in postcolonoscopy colorectal cancers.” the researchers said. “Additional studies are needed to determine whether these postcolonoscopy colorectal cancers arise through a familial cancer pathway and/or serrated neoplastic pathway of sporadic lesions.
Funders included the American College of Gastroenterology, the National Cancer Institute, the Huntsman Cancer Foundation, the University of Utah, and the Utah Department of Health. Dr. Samadder reported consulting relationships with Cancer Prevention Pharmaceuticals and Janssen Research and Development. The other researchers reported having no conflicts of interest.
SOURCE: Samadder NJ et al. Clin Gastroenterol Hepatol. 2019 Mar 28. doi: 10.1016/j.cgh.2019.02.040.
Postcolonoscopy colorectal cancers were more likely to arise in the proximal colon and to show microsatellite instability, according to the results of a retrospective population-based study of 168 adults with incident colorectal cancers.
In all, 64% of postcolonoscopy colorectal cancers were located in the proximal colon, compared with 44% of detected colorectal cancers (P = .016), reported Niloy Jewel Samadder, MD, of the University of Utah in Salt Lake City, together with his associates. Furthermore, microsatellite instability (MSI) was detected in 32% of postcolonoscopy colorectal cancers, versus 13% of detected colorectal cancers (P = .005). These findings may point to differences in the underlying biology of postcolonoscopy colorectal cancers and detected colorectal cancers, they said. Studies are needed “to determine if postcolonoscopy cancers arise through a specific genetic pathway that may accelerate neoplastic progression,” they wrote in Clinical Gastroenterology and Hepatology.
Postcolonoscopy colorectal cancers are a “small but clinically important subset of colorectal cancers” that are diagnosed after the patient has a colonoscopy in which no cancer is detected, the researchers noted. These cancers have an estimated global prevalence ranging from 3% to 9% and an estimated pooled prevalence of 3.7% (Am J Gastroenterol. 2014;109:1375-89). Risk factors for postcolonoscopy colorectal cancers include low adenoma detection rates, rural facilities, and care by physicians who are not gastroenterologists. However, tumor-specific and patient-specific factors, including location within the colon and superior survival, compared with detected cancers, raises the possibility of underlying molecular differences related to tumorigenesis, the researchers said.
To investigate this idea, they retrospectively analyzed data from residents of Utah between 50 and 80 years old who had a colonoscopy between, Feb. 15, 1995, and Jan. 31, 2009, at one of two large clinical facilities in Utah (Intermountain Healthcare or the University of Utah Health Sciences). Using a state population-based database, they merged medical information from these patients with cancer histories from the Utah Cancer Registry. This enabled them to compare all 84 postcolonoscopy colorectal cancers (defined as those detected within 6-60 months of colonoscopy) with tissue available for analysis with 84 detected colorectal cancers (detected within 6 months of a colonoscopy).
In the multivariable analysis, MSI was the only molecular feature that was significantly more frequent in postcolonoscopy versus detected colorectal cancers (odds ratio, 4.20; 95% confidence interval, 1.58-11.14). However, postcolonoscopy colorectal cancers were significantly more likely to be early stage (86% versus 69% for detected colorectal cancers; P = .040). Five-year survival did not significantly differ between the groups.
“The molecular signatures of postcolonoscopy colorectal cancers in our study overlap with those of sporadic MSI and serrated pathways, suggesting these mechanisms play a disproportionate role in postcolonoscopy colorectal cancers.” the researchers said. “Additional studies are needed to determine whether these postcolonoscopy colorectal cancers arise through a familial cancer pathway and/or serrated neoplastic pathway of sporadic lesions.
Funders included the American College of Gastroenterology, the National Cancer Institute, the Huntsman Cancer Foundation, the University of Utah, and the Utah Department of Health. Dr. Samadder reported consulting relationships with Cancer Prevention Pharmaceuticals and Janssen Research and Development. The other researchers reported having no conflicts of interest.
SOURCE: Samadder NJ et al. Clin Gastroenterol Hepatol. 2019 Mar 28. doi: 10.1016/j.cgh.2019.02.040.
Postcolonoscopy colorectal cancers were more likely to arise in the proximal colon and to show microsatellite instability, according to the results of a retrospective population-based study of 168 adults with incident colorectal cancers.
In all, 64% of postcolonoscopy colorectal cancers were located in the proximal colon, compared with 44% of detected colorectal cancers (P = .016), reported Niloy Jewel Samadder, MD, of the University of Utah in Salt Lake City, together with his associates. Furthermore, microsatellite instability (MSI) was detected in 32% of postcolonoscopy colorectal cancers, versus 13% of detected colorectal cancers (P = .005). These findings may point to differences in the underlying biology of postcolonoscopy colorectal cancers and detected colorectal cancers, they said. Studies are needed “to determine if postcolonoscopy cancers arise through a specific genetic pathway that may accelerate neoplastic progression,” they wrote in Clinical Gastroenterology and Hepatology.
Postcolonoscopy colorectal cancers are a “small but clinically important subset of colorectal cancers” that are diagnosed after the patient has a colonoscopy in which no cancer is detected, the researchers noted. These cancers have an estimated global prevalence ranging from 3% to 9% and an estimated pooled prevalence of 3.7% (Am J Gastroenterol. 2014;109:1375-89). Risk factors for postcolonoscopy colorectal cancers include low adenoma detection rates, rural facilities, and care by physicians who are not gastroenterologists. However, tumor-specific and patient-specific factors, including location within the colon and superior survival, compared with detected cancers, raises the possibility of underlying molecular differences related to tumorigenesis, the researchers said.
To investigate this idea, they retrospectively analyzed data from residents of Utah between 50 and 80 years old who had a colonoscopy between, Feb. 15, 1995, and Jan. 31, 2009, at one of two large clinical facilities in Utah (Intermountain Healthcare or the University of Utah Health Sciences). Using a state population-based database, they merged medical information from these patients with cancer histories from the Utah Cancer Registry. This enabled them to compare all 84 postcolonoscopy colorectal cancers (defined as those detected within 6-60 months of colonoscopy) with tissue available for analysis with 84 detected colorectal cancers (detected within 6 months of a colonoscopy).
In the multivariable analysis, MSI was the only molecular feature that was significantly more frequent in postcolonoscopy versus detected colorectal cancers (odds ratio, 4.20; 95% confidence interval, 1.58-11.14). However, postcolonoscopy colorectal cancers were significantly more likely to be early stage (86% versus 69% for detected colorectal cancers; P = .040). Five-year survival did not significantly differ between the groups.
“The molecular signatures of postcolonoscopy colorectal cancers in our study overlap with those of sporadic MSI and serrated pathways, suggesting these mechanisms play a disproportionate role in postcolonoscopy colorectal cancers.” the researchers said. “Additional studies are needed to determine whether these postcolonoscopy colorectal cancers arise through a familial cancer pathway and/or serrated neoplastic pathway of sporadic lesions.
Funders included the American College of Gastroenterology, the National Cancer Institute, the Huntsman Cancer Foundation, the University of Utah, and the Utah Department of Health. Dr. Samadder reported consulting relationships with Cancer Prevention Pharmaceuticals and Janssen Research and Development. The other researchers reported having no conflicts of interest.
SOURCE: Samadder NJ et al. Clin Gastroenterol Hepatol. 2019 Mar 28. doi: 10.1016/j.cgh.2019.02.040.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Getting high heightens stroke, arrhythmia risks
Stoners, beware: , and people with cannabis use disorder are at a 50% greater risk of being hospitalized for arrhythmias, according to new research presented at the American Heart Association Scientific Sessions 2019.
An analysis of pooled data on nearly 44,000 participants in a cross-sectional survey showed that, among the 13.6% who reported using marijuana within the last 30 days, the adjusted odds ratio for young-onset stroke (aged 18-44 years), compared with non-users, was 2.75, reported Tarang Parekh, MBBS, a health policy researcher of George Mason University in Fairfax, Va., and colleagues.
In a separate study, a retrospective analysis of national inpatient data showed that people diagnosed with cannabis use disorder – a pathological pattern of impaired control, social impairment, risky behavior or physiological adaptation similar in nature to alcoholism – had a 47%-52% increased likelihood of hospitalization for an arrhythmia, reported Rikinkumar S. Patel, MD, a psychiatry resident at Griffin Memorial Hospital in Norman, Okla.
“As these [cannabis] products become increasingly used across the country, getting clearer, scientifically rigorous data is going to be important as we try to understand the overall health effects of cannabis,” said AHA President Robert Harrington, MD, of Stanford (Calif.) University in a statement.
Currently, use of both medical and recreational marijuana is fully legal in 11 U.S. states and the District of Columbia. Medical marijuana is legal with recreational use decriminalized (or penalties reduced) in 28 other states, and totally illegal in 11 other states, according to employee screening firm DISA Global Solutions.
Stroke study
In an oral presentation with simultaneous publication in the AHA journal Stroke, Dr. Parekh and colleagues presented an analysis of pooled data from the Behavioral Risk Factor Surveillance System (BRFSS), a nationally representative cross-sectional survey collected by the Centers for Disease Control and Prevention in 2016 and 2017.
They looked at baseline sociodemographic data and created multivariable logistic regression models with state fixed effects to determine whether marijuana use within the last 30 days was associated with young-onset stroke.
They identified 43,860 participants representing a weighted sample of 35.5 million Americans. Of the sample, 63.3% were male, and 13.6 % of all participants reported using marijuana in the last 30 days.
They found in an unadjusted model that marijuana users had an odds ratio for stroke, compared with nonusers, of 1.59 (P less than.1), and in a model adjusted for demographic factors (gender, race, ethnicity, and education) the OR increased to 1.76 (P less than .05).
When they threw risk behavior into the model (physical activity, body mass index, heavy drinking, and cigarette smoking), they saw that the OR for stroke shot up to 2.75 (P less than .01).
“Physicians should ask patients if they use cannabis and counsel them about its potential stroke risk as part of regular doctor visits,” Dr. Parekh said in a statement.
Arrhythmias study
Based on recent studies suggesting that cannabis use may trigger cardiovascular events, Dr. Patel and colleagues studied whether cannabis use disorder may be related to arrhythmias, approaching the question through hospital records.
“The effects of using cannabis are seen within 15 minutes and last for around 3 hours. At lower doses, it is linked to a rapid heartbeat. At higher doses, it is linked to a too-slow heartbeat,” he said in a statement.
Dr. Patel and colleagues conducted a retrospective analysis of the Nationwide Inpatient Sample from 2010-2014, a period during which medical marijuana became legal in several states and recreational marijuana became legal in Colorado and Washington. The sample is a database maintained by the Healthcare Cost and Utilization Project of the U.S. Office of Disease Prevention and Health Promotion.
They identified 570,557 patients aged 15-54 years with a primary diagnosis of arrhythmia, and compared them with a sample of 67,662,082 patients hospitalized with no arrhythmia diagnosed during the same period.
They found a 2.6% incidence of cannabis use disorder among patients hospitalized for arrhythmias. Patients with cannabis use disorder tended to be younger (15- to 24-years-old; OR, 4.23), male (OR, 1.70) and African American (OR, 2.70).
In regression analysis adjusted for demographics and comorbidities, cannabis use disorder was associated with higher odds of arrhythmia hospitalization in young patients, at 1.28 times among 15- to 24-year-olds (95% confidence interval, 1.229-1.346) and 1.52 times for 25- to 34-year-olds (95% CI, 1.469-1.578).
“As medical and recreational cannabis is legalized in many states, it is important to know the difference between therapeutic cannabis dosing for medical purposes and the consequences of cannabis abuse. We urgently need additional research to understand these issues,” Dr. Patel said.
“It’s not proving that there’s a direct link, but it’s raising a suggestion in an observational analysis that [this] indeed might be the case. What that means for clinicians is that, if you’re seeing a patient who is presenting with a symptomatic arrhythmia, adding cannabis usage to your list of questions as you begin to try to understand possible precipitating factors for this arrhythmia seems to be a reasonable thing to do,” Dr. Harrington commented.
Stoners, beware: , and people with cannabis use disorder are at a 50% greater risk of being hospitalized for arrhythmias, according to new research presented at the American Heart Association Scientific Sessions 2019.
An analysis of pooled data on nearly 44,000 participants in a cross-sectional survey showed that, among the 13.6% who reported using marijuana within the last 30 days, the adjusted odds ratio for young-onset stroke (aged 18-44 years), compared with non-users, was 2.75, reported Tarang Parekh, MBBS, a health policy researcher of George Mason University in Fairfax, Va., and colleagues.
In a separate study, a retrospective analysis of national inpatient data showed that people diagnosed with cannabis use disorder – a pathological pattern of impaired control, social impairment, risky behavior or physiological adaptation similar in nature to alcoholism – had a 47%-52% increased likelihood of hospitalization for an arrhythmia, reported Rikinkumar S. Patel, MD, a psychiatry resident at Griffin Memorial Hospital in Norman, Okla.
“As these [cannabis] products become increasingly used across the country, getting clearer, scientifically rigorous data is going to be important as we try to understand the overall health effects of cannabis,” said AHA President Robert Harrington, MD, of Stanford (Calif.) University in a statement.
Currently, use of both medical and recreational marijuana is fully legal in 11 U.S. states and the District of Columbia. Medical marijuana is legal with recreational use decriminalized (or penalties reduced) in 28 other states, and totally illegal in 11 other states, according to employee screening firm DISA Global Solutions.
Stroke study
In an oral presentation with simultaneous publication in the AHA journal Stroke, Dr. Parekh and colleagues presented an analysis of pooled data from the Behavioral Risk Factor Surveillance System (BRFSS), a nationally representative cross-sectional survey collected by the Centers for Disease Control and Prevention in 2016 and 2017.
They looked at baseline sociodemographic data and created multivariable logistic regression models with state fixed effects to determine whether marijuana use within the last 30 days was associated with young-onset stroke.
They identified 43,860 participants representing a weighted sample of 35.5 million Americans. Of the sample, 63.3% were male, and 13.6 % of all participants reported using marijuana in the last 30 days.
They found in an unadjusted model that marijuana users had an odds ratio for stroke, compared with nonusers, of 1.59 (P less than.1), and in a model adjusted for demographic factors (gender, race, ethnicity, and education) the OR increased to 1.76 (P less than .05).
When they threw risk behavior into the model (physical activity, body mass index, heavy drinking, and cigarette smoking), they saw that the OR for stroke shot up to 2.75 (P less than .01).
“Physicians should ask patients if they use cannabis and counsel them about its potential stroke risk as part of regular doctor visits,” Dr. Parekh said in a statement.
Arrhythmias study
Based on recent studies suggesting that cannabis use may trigger cardiovascular events, Dr. Patel and colleagues studied whether cannabis use disorder may be related to arrhythmias, approaching the question through hospital records.
“The effects of using cannabis are seen within 15 minutes and last for around 3 hours. At lower doses, it is linked to a rapid heartbeat. At higher doses, it is linked to a too-slow heartbeat,” he said in a statement.
Dr. Patel and colleagues conducted a retrospective analysis of the Nationwide Inpatient Sample from 2010-2014, a period during which medical marijuana became legal in several states and recreational marijuana became legal in Colorado and Washington. The sample is a database maintained by the Healthcare Cost and Utilization Project of the U.S. Office of Disease Prevention and Health Promotion.
They identified 570,557 patients aged 15-54 years with a primary diagnosis of arrhythmia, and compared them with a sample of 67,662,082 patients hospitalized with no arrhythmia diagnosed during the same period.
They found a 2.6% incidence of cannabis use disorder among patients hospitalized for arrhythmias. Patients with cannabis use disorder tended to be younger (15- to 24-years-old; OR, 4.23), male (OR, 1.70) and African American (OR, 2.70).
In regression analysis adjusted for demographics and comorbidities, cannabis use disorder was associated with higher odds of arrhythmia hospitalization in young patients, at 1.28 times among 15- to 24-year-olds (95% confidence interval, 1.229-1.346) and 1.52 times for 25- to 34-year-olds (95% CI, 1.469-1.578).
“As medical and recreational cannabis is legalized in many states, it is important to know the difference between therapeutic cannabis dosing for medical purposes and the consequences of cannabis abuse. We urgently need additional research to understand these issues,” Dr. Patel said.
“It’s not proving that there’s a direct link, but it’s raising a suggestion in an observational analysis that [this] indeed might be the case. What that means for clinicians is that, if you’re seeing a patient who is presenting with a symptomatic arrhythmia, adding cannabis usage to your list of questions as you begin to try to understand possible precipitating factors for this arrhythmia seems to be a reasonable thing to do,” Dr. Harrington commented.
Stoners, beware: , and people with cannabis use disorder are at a 50% greater risk of being hospitalized for arrhythmias, according to new research presented at the American Heart Association Scientific Sessions 2019.
An analysis of pooled data on nearly 44,000 participants in a cross-sectional survey showed that, among the 13.6% who reported using marijuana within the last 30 days, the adjusted odds ratio for young-onset stroke (aged 18-44 years), compared with non-users, was 2.75, reported Tarang Parekh, MBBS, a health policy researcher of George Mason University in Fairfax, Va., and colleagues.
In a separate study, a retrospective analysis of national inpatient data showed that people diagnosed with cannabis use disorder – a pathological pattern of impaired control, social impairment, risky behavior or physiological adaptation similar in nature to alcoholism – had a 47%-52% increased likelihood of hospitalization for an arrhythmia, reported Rikinkumar S. Patel, MD, a psychiatry resident at Griffin Memorial Hospital in Norman, Okla.
“As these [cannabis] products become increasingly used across the country, getting clearer, scientifically rigorous data is going to be important as we try to understand the overall health effects of cannabis,” said AHA President Robert Harrington, MD, of Stanford (Calif.) University in a statement.
Currently, use of both medical and recreational marijuana is fully legal in 11 U.S. states and the District of Columbia. Medical marijuana is legal with recreational use decriminalized (or penalties reduced) in 28 other states, and totally illegal in 11 other states, according to employee screening firm DISA Global Solutions.
Stroke study
In an oral presentation with simultaneous publication in the AHA journal Stroke, Dr. Parekh and colleagues presented an analysis of pooled data from the Behavioral Risk Factor Surveillance System (BRFSS), a nationally representative cross-sectional survey collected by the Centers for Disease Control and Prevention in 2016 and 2017.
They looked at baseline sociodemographic data and created multivariable logistic regression models with state fixed effects to determine whether marijuana use within the last 30 days was associated with young-onset stroke.
They identified 43,860 participants representing a weighted sample of 35.5 million Americans. Of the sample, 63.3% were male, and 13.6 % of all participants reported using marijuana in the last 30 days.
They found in an unadjusted model that marijuana users had an odds ratio for stroke, compared with nonusers, of 1.59 (P less than.1), and in a model adjusted for demographic factors (gender, race, ethnicity, and education) the OR increased to 1.76 (P less than .05).
When they threw risk behavior into the model (physical activity, body mass index, heavy drinking, and cigarette smoking), they saw that the OR for stroke shot up to 2.75 (P less than .01).
“Physicians should ask patients if they use cannabis and counsel them about its potential stroke risk as part of regular doctor visits,” Dr. Parekh said in a statement.
Arrhythmias study
Based on recent studies suggesting that cannabis use may trigger cardiovascular events, Dr. Patel and colleagues studied whether cannabis use disorder may be related to arrhythmias, approaching the question through hospital records.
“The effects of using cannabis are seen within 15 minutes and last for around 3 hours. At lower doses, it is linked to a rapid heartbeat. At higher doses, it is linked to a too-slow heartbeat,” he said in a statement.
Dr. Patel and colleagues conducted a retrospective analysis of the Nationwide Inpatient Sample from 2010-2014, a period during which medical marijuana became legal in several states and recreational marijuana became legal in Colorado and Washington. The sample is a database maintained by the Healthcare Cost and Utilization Project of the U.S. Office of Disease Prevention and Health Promotion.
They identified 570,557 patients aged 15-54 years with a primary diagnosis of arrhythmia, and compared them with a sample of 67,662,082 patients hospitalized with no arrhythmia diagnosed during the same period.
They found a 2.6% incidence of cannabis use disorder among patients hospitalized for arrhythmias. Patients with cannabis use disorder tended to be younger (15- to 24-years-old; OR, 4.23), male (OR, 1.70) and African American (OR, 2.70).
In regression analysis adjusted for demographics and comorbidities, cannabis use disorder was associated with higher odds of arrhythmia hospitalization in young patients, at 1.28 times among 15- to 24-year-olds (95% confidence interval, 1.229-1.346) and 1.52 times for 25- to 34-year-olds (95% CI, 1.469-1.578).
“As medical and recreational cannabis is legalized in many states, it is important to know the difference between therapeutic cannabis dosing for medical purposes and the consequences of cannabis abuse. We urgently need additional research to understand these issues,” Dr. Patel said.
“It’s not proving that there’s a direct link, but it’s raising a suggestion in an observational analysis that [this] indeed might be the case. What that means for clinicians is that, if you’re seeing a patient who is presenting with a symptomatic arrhythmia, adding cannabis usage to your list of questions as you begin to try to understand possible precipitating factors for this arrhythmia seems to be a reasonable thing to do,” Dr. Harrington commented.
REPORTING FROM AHA 2019
Predictors of HA1c Goal Attainment in Patients Treated With Insulin at a VA Pharmacist-Managed Insulin Clinic (FULL)
Showing up to appointments and adherence to treatment recommendations correlated with glycemic goal attainment for patients.
About 30.3 million Americans (9.4%) have diabetes mellitus (DM).1 Veterans are disproportionately affected—about 1 in 4 of those who receive US Department of Veterans Affairs (VA) care have DM.2 The consequences of uncontrolled DM include microvascular complications (eg, retinopathy, neuropathy, and nephropathy) and macrovascular complications (eg, cardiovascular disease).
The American Diabetes Association (ADA) recommends achieving a goal hemoglobin A1c (HbA1c) level of < 7% to prevent these complications. However, a goal of < 8% HbA1c may be more appropriate for certain patients when a more strict goal may be impractical or have the potential to cause harm.3 Furthermore, guidelines developed by the VA and the US Department of Defense suggest a target HbA1c range of 7.0% to 8.5% for patients with established microvascular or macrovascular disease, comorbid conditions, or a life expectancy of 5 to 10 years.4
Despite the existence of evidence showing the importance of glycemic control in preventing morbidity and mortality associated with DM, many patients have inadequate glycemic control. Diabetes mellitus is the seventh leading cause of death in the US. Moreover, DM is a known risk factor for heart disease, stroke, and kidney disease, which are the first, fifth, and ninth leading causes of death in the US, respectively.5
Because DM management requires ongoing and comprehensive maintenance and monitoring, the ADA supports a collaborative, multidisciplinary, and patient-centered approach to delivery of care.3 Collaborative teams involving pharmacists have been shown to improve outcomes and cost savings for chronic diseases, including DM.6-12 In 1995, the VA launched a national policy providing clinical pharmacists with prescribing privileges that would aid in the provision of coordinated medication management for patients with chronic illnesses.13 The policy created a framework for collaborative drug therapy management (CDTM) models, which grants pharmacists the ability to perform patient assessments, order laboratory tests, and modify medications within a scope of practice.
Since the initiation of these services, several examples of successful DM management services using clinical pharmacists within the VA exist in the literature.14-16 However, even with intensive chronic disease and drug therapy management, not all patients who enroll in these services successfully reach clinical goals. Although these pharmacist-driven services seem to demonstrate overall benefit and cost savings to veteran patients and the VA system, little published data exist to help determine patient behaviors that are associated with glycemic goal attainment when using these services.
At the Corporal Michael J. Crescenz VA Medical Center in (CMCVAMC) Philadelphia, Pennsylvania, where this study was performed, primary care providers may refer patients with uncontrolled DM to the pharmacist disease state management (DSM) clinic. The clinic is a form of a CDTM and receives numerous referrals per year, with many patients discharged for successfully meeting glycemic targets.
However, a percentage of patients fail to attain glycemic goals despite involvement in this clinic. We observed specific patient behaviors that delayed glycemic goal attainment. This study examined whether these behaviors correlated with prolonged glycemic goal attainment. The purpose of this study was to identify patient behaviors that led to glycemic goal attainment in insulin-treated patients referred to this pharmacist DSM clinic.
Methods
This study was performed as a single-center retrospective chart review. The protocol and data collection documents were approved by the CMCVAMC Institutional Review Board. It included patients referred to a pharmacist-led DSM clinic for insulin titration/optimization from January 1, 2011 through December 31, 2012. Data were collected through June 30, 2013, to allow for 6 months after the last referral date of December 31, 2012.
This study included patients who were on insulin therapy at the time of pharmacy consult, who attended at least 3 consecutive pharmacy DSM clinic visits, and had an HbA1c ≥ 8% at the time of initial clinic consult. Patients who failed to have 3 consecutive pharmacy DSM clinic visits, were insulin-naïve at the time of referral, aged ≥ 90, lacked at least 1 follow-up HbA1c result while enrolled in the clinic, or had HbA1c < 8% were excluded.
Among the patients who met eligibility criteria, charts within the Computerized Patient Record System (CPRS) were reviewed in a chronologic order within the respective study time frame. A convenience sample of 100 patients were enrolled in each treatment arm: the goal-attained arm or the goal-not-attained arm.
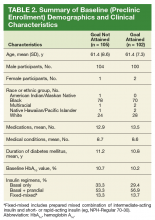
The primary study variable was HbA1c goal attainment, which was defined in this investigation as at least 1 HbA1c reading of < 8% while enrolled in the DSM clinic during the review period. Secondary variables included specific patient factors such as optimal frequency of self-monitoring of blood glucose (SMBG) testing, adherence to pharmacist’s instructions for changes to glucose-lowering medications, adherence to bringing glucose meter/glucose log book to clinic appointments, and percentage of visits attended. Definitions for each variable are provided in Table 1.
We hypothesized that patients who were more adherent to treatment plans, regularly attend clinic visits, and appropriately monitor their glucose levels were more likely to meet their glycemic goals.
Statistical Analysis
Univariate descriptive statistics described the individual variables/predictors of HbA1c goal attainment. As the study’s purpose was to identify patient factors and characteristics associated with HbA1c goal attainment, a logistic regression model framework was used for all covariates to evaluate each measured variable’s independent association with HbA1c. The univariate tests were used to compare patient characteristics between the 2 study groups: Pearson chi-square test was used for nominal data, and a paired t test (for normally distributed data) or Wilcoxon rank sum test (for non-normally distributed data) was used for continuous variables. Variables having a P value < .2 underwent a multivariate analysis stepwise logistic regression model to identify patient factors and characteristics associated with HbA1c goal attainment. A Fisher exact test was used to determine gender effect on HbA1c goal attainment, categoric variables were analyzed using Pearson chi-square test, and an unpaired t test was used for continuous data. The backward elimination approach to inclusion of variables in the model was used to build the most parsimonious and best-fitting model, and the Hosmer-Lemeshow goodness-of-fit tests was used to assess model fit. Data analyses were performed using IBM SPSS, version 18.0 (Armonk, NY).
Results
Five hundred eighty-four patient records were reviewed, and 207 patients met inclusion criteria: 102 patient records were reviewed for the goal-attained arm, and 105 patient records for the goal-not-attained arm. Most patients were excluded from the analysis due to not having 3 consecutive visits during the specified period or having an HbA1c of < 8% at the time of referral to the pharmacist DSM clinic.
The patients in this study had type 2 diabetes for about 11 years, were overwhelmingly male (99%), were aged about 61 years, and were taking on average 13 medications at the time of referral to the pharmacist DSM clinic. Mean HbA1c at time of enrollment was slightly higher in the goal-not-attained arm vs goal-attained arm (10.7% vs 10.2%, respectively), but the difference was not statistically significant (P = .066). A little more than half the patients in both study arms were on basal + prandial insulin regimens (Table 2).
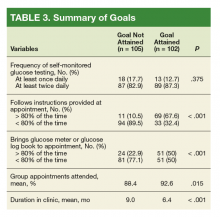
Patients who attained their goal HbA1cwere more likely to bring their glucose meter/glucose log book to at least 80% of their appointments (P < .001). Additionally, this same cohort followed insulin dosing instructions at least 80% of the time (P < .001).
Five variables were included in the multivariate analysis because they had a P value ≤ .2 in univariate analyses: (1) patient adherence to instructions (P < .001); (2) duration in clinic (P < .001); (3) patient bringingglucose meter or glucose log to appointments (P < .001); (4) percentage of scheduled appointments patient attended (P = .015); and (5) baseline HbA1c (P = .066).
Discussion
The development and constant modification of clinical practicing guidelines has made DM treatment a focus and priority.3,4 Additionally, the collaborative approach to health care and creation of VA pharmacist-driven services have demonstrated successful patient outcomes.6-16 Despite these efforts, further insight is needed to improve the management of DM. Our study identified specific behavioral factors that correlated to veteran patients to attaining their HbA1c goal of < 8% within a VA pharmacist DSM clinic. Additionally, it highlighted factors that contributed to patients not achieving their glycemic goals.
Our univariate analysis showed behaviors such as showing up for appointments and following directions regimens to correlate with glycemic goal attainment. However, following directions was the only behavioral factor that correlated to glycemic goal attainment in our multivariate analysis. Additionally, our findings indicated that factors for HbA1c goal attainment included patients who brought their glucose meter/glucose log book and attended clinic appointments at least 80% of the time, respectively.
These findings can help further refine the process for identifying patients who are most likely to achieve glycemic goals when referred to pharmacist DSM clinics or to any DM treatment program. Assessment of a patient’s motivation and ability to attend clinic appointments, bring their glucose meter/glucose log book, and to follow instructions provided at these appointments are reasonable screening questions to ask before referring that patient to a diabetes care program or service. Currently, this is not performed during the consult process to the pharmacist DSM clinic at the respective VA.
Additionally, our findings show that patients who met goal did so, on average, within 6 months of referral to the pharmacist DSM clinic. This finding may have occurred because patients who successfully reach HbA1c goal in 2 consecutive checks are discharged from the clinic. Patients who do not meet this goal continue with the clinic, thus increasing their duration of enrollment in this service. This finding could help clinical pharmacists estimate how long patients will be followed by the service, thus allowing for a more accurate estimation of workload and clinic capacity. Additionally, this finding provides insight if the patient should remain in clinic or be transferred to another program. Our findings aligned with previous studies showing the link between patient behaviors and glycemic goal attainment.17-19
Limitations
This study has a few notable limitations. First, it is limited to 1 VA medical center, so our findings may not be extrapolated easily to other institutions of the Veterans Health Administration. Ideally, future studies centered on identifying factors that lead to successful glycemic goal attainment would be helpful from multiple VA institutions. This would encourage more factors to be identified and trends to be strengthened. Ultimately, this would allow for more global changes to the consult process from primary care to pharmacist DSM clinics nationally vs at a local VA institution. Additionally, this study was limited to a specific retrospective time frame, therefore limiting its ability to identify trends. This study also relied on some subjective factors, such as the patient’s self-report of properly following the clinic instructions. Another limitation was that our investigation was not designed to characterize the specific pharmacist’s interventions that improved glycemic control. Future studies would benefit from the inclusion of specific interventions and their effect on glycemic goal attainment.
Conclusion
This retrospective study offers insight to specific patient behavioral factors that correlate with glycemic goal attainment in a VA pharmacist DSM clinic. Behavioral factors linked to HbA1c goal attainment of < 8% included appointment keeping, bringing glucose meter/glucose log book at least 80% of the time to these appointments, and following clinic instructions. This investigation also found that patients who attain glycemic goals generally do so within 6 months of enrollment. Furthermore, this study provided insight that following the clinic instructions a majority of the time strongly contributes to glycemic goal attainment. We believe that an assessment of patients’ behaviors prior to referrals to diabetes management programs will yield useful information about possible barriers to glycemic goal attainment.
1. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Published 2017. Accessed September 25, 2018.
2. Gaspar JL, Dahlke ME, Kasper B. Efficacy of patient aligned care team pharmacist service in reaching diabetes and hyperlipidemia treatment goals. Fed Pract. 2015;32(11):42-47.
3. American Diabetes Association. Standards of medical care in diabetes—2017. Diabetes Care. 2017;40(suppl 1):S6-S135.
4. US Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. https://www.healthquality.va.gov/guidelines/CD/diabetes/VADoDDMCPGFinal508.pdf. Published April 2017. Accessed September 7, 2018.
5. Centers for Disease Control and Prevention. Deaths: leading causes for 2014. Natl Vital Stat Rep. 2016;65(5):1-96.
6. Nigro SC, Garwood CL, Berlie H, et al. Clinical pharmacists as key members of the patient-centered medical home: an opinion statement of the Ambulatory Care Practice and Research Network of the American College of Clinical Pharmacy. Pharmacotherapy. 2014;34(1):96-108.
7. Smith M, Bates DW, Bodenheimer T, et al. Why pharmacists belong in the medical home. Health Aff (Millwood). 2010;29(5):906-913.
8. Chisholm-Burns MA, Kim Lee J, Spivey CA, et al. US Pharmacists’ effect as team members on patient care. Med Care. 2010;48(10):923-933.
9. Wubben DP, Vivian EM. Effects of pharmacist outpatient interventions on adults with diabetes mellitus: a systematic review. Pharmacotherapy. 2008;28(4):421-436.
10. Touchette DR, Doloresco F, Suda KJ, et al. Economic evaluations of clinical pharmacy services: 2006-2010. Pharmacotherapy. 2014;34(8):771-793.
11. Giberson S, Yoder S, Lee MP. Improving patient and health system outcomes through advanced pharmacy practice. A report of the U.S. Surgeon General. American College of Clinical Pharmacy. https://www.accp.com/docs/positions/misc/Improving_Patient_and_Health_System_Outcomes.pdf. Published December 2011. Accessed September 10, 2018.
12. Isetts BJ, Schondelmeyer SW, Artz MB, et al. Clinical and economic outcomes of medication therapy management services: the Minnesota experience. J Am Pharm Assoc (2003). 2008;48(2):203-211.
13. Ourth H, Groppi J, Morreale AP, Quicci-Roberts K. Clinical pharmacist prescribing activities in the Veterans Health Administration. Am J Health Syst Pharm. 2016;73(18):1406-1415.
14. Taveira TH, Friedmann PD, Cohen LB, et al. Pharmacist-led group medical appointment model in type 2 diabetes. Diabetes Educ. 2010;36(1):109-117.
15. Edwards KL, Hadley RL, Baby N, Yeary JC, Chastain LM, Brown CD. Utilizing clinical pharmacy specialists to address access to care barriers in the veteran population for the management of diabetes. J Pharm Pract. 2017;30(4):412-418.
16. Cripps RJ, Gourley ES, Johnson W, et al. An evaluation of diabetes-related measures of control after 6 months of clinical pharmacy specialist intervention. J Pharm Prac. 2011;24(3):332-338.
17. Jones H, Edwards L, Vallis TM, et al; Diabetes Stages of Change (DiSC) Study. Changes in diabetes self-care behaviors make a difference in glycemic control. Diabetes Care. 2003;26(3):732-737.
18. Schetman JM, Schorling JB, Voss JD. Appointment adherence and disparities in outcomes among patients with diabetes. J Gen Intern Med. 2008;23(10):1685-1687.
19. Rhee, MK, Slocum W, Zeimer DC, et al. Patient adherence improves glycemic control. Diabetes Educ. 2005;31(2):240-250.
Showing up to appointments and adherence to treatment recommendations correlated with glycemic goal attainment for patients.
Showing up to appointments and adherence to treatment recommendations correlated with glycemic goal attainment for patients.
About 30.3 million Americans (9.4%) have diabetes mellitus (DM).1 Veterans are disproportionately affected—about 1 in 4 of those who receive US Department of Veterans Affairs (VA) care have DM.2 The consequences of uncontrolled DM include microvascular complications (eg, retinopathy, neuropathy, and nephropathy) and macrovascular complications (eg, cardiovascular disease).
The American Diabetes Association (ADA) recommends achieving a goal hemoglobin A1c (HbA1c) level of < 7% to prevent these complications. However, a goal of < 8% HbA1c may be more appropriate for certain patients when a more strict goal may be impractical or have the potential to cause harm.3 Furthermore, guidelines developed by the VA and the US Department of Defense suggest a target HbA1c range of 7.0% to 8.5% for patients with established microvascular or macrovascular disease, comorbid conditions, or a life expectancy of 5 to 10 years.4
Despite the existence of evidence showing the importance of glycemic control in preventing morbidity and mortality associated with DM, many patients have inadequate glycemic control. Diabetes mellitus is the seventh leading cause of death in the US. Moreover, DM is a known risk factor for heart disease, stroke, and kidney disease, which are the first, fifth, and ninth leading causes of death in the US, respectively.5
Because DM management requires ongoing and comprehensive maintenance and monitoring, the ADA supports a collaborative, multidisciplinary, and patient-centered approach to delivery of care.3 Collaborative teams involving pharmacists have been shown to improve outcomes and cost savings for chronic diseases, including DM.6-12 In 1995, the VA launched a national policy providing clinical pharmacists with prescribing privileges that would aid in the provision of coordinated medication management for patients with chronic illnesses.13 The policy created a framework for collaborative drug therapy management (CDTM) models, which grants pharmacists the ability to perform patient assessments, order laboratory tests, and modify medications within a scope of practice.
Since the initiation of these services, several examples of successful DM management services using clinical pharmacists within the VA exist in the literature.14-16 However, even with intensive chronic disease and drug therapy management, not all patients who enroll in these services successfully reach clinical goals. Although these pharmacist-driven services seem to demonstrate overall benefit and cost savings to veteran patients and the VA system, little published data exist to help determine patient behaviors that are associated with glycemic goal attainment when using these services.
At the Corporal Michael J. Crescenz VA Medical Center in (CMCVAMC) Philadelphia, Pennsylvania, where this study was performed, primary care providers may refer patients with uncontrolled DM to the pharmacist disease state management (DSM) clinic. The clinic is a form of a CDTM and receives numerous referrals per year, with many patients discharged for successfully meeting glycemic targets.
However, a percentage of patients fail to attain glycemic goals despite involvement in this clinic. We observed specific patient behaviors that delayed glycemic goal attainment. This study examined whether these behaviors correlated with prolonged glycemic goal attainment. The purpose of this study was to identify patient behaviors that led to glycemic goal attainment in insulin-treated patients referred to this pharmacist DSM clinic.
Methods
This study was performed as a single-center retrospective chart review. The protocol and data collection documents were approved by the CMCVAMC Institutional Review Board. It included patients referred to a pharmacist-led DSM clinic for insulin titration/optimization from January 1, 2011 through December 31, 2012. Data were collected through June 30, 2013, to allow for 6 months after the last referral date of December 31, 2012.
This study included patients who were on insulin therapy at the time of pharmacy consult, who attended at least 3 consecutive pharmacy DSM clinic visits, and had an HbA1c ≥ 8% at the time of initial clinic consult. Patients who failed to have 3 consecutive pharmacy DSM clinic visits, were insulin-naïve at the time of referral, aged ≥ 90, lacked at least 1 follow-up HbA1c result while enrolled in the clinic, or had HbA1c < 8% were excluded.
Among the patients who met eligibility criteria, charts within the Computerized Patient Record System (CPRS) were reviewed in a chronologic order within the respective study time frame. A convenience sample of 100 patients were enrolled in each treatment arm: the goal-attained arm or the goal-not-attained arm.
The primary study variable was HbA1c goal attainment, which was defined in this investigation as at least 1 HbA1c reading of < 8% while enrolled in the DSM clinic during the review period. Secondary variables included specific patient factors such as optimal frequency of self-monitoring of blood glucose (SMBG) testing, adherence to pharmacist’s instructions for changes to glucose-lowering medications, adherence to bringing glucose meter/glucose log book to clinic appointments, and percentage of visits attended. Definitions for each variable are provided in Table 1.
We hypothesized that patients who were more adherent to treatment plans, regularly attend clinic visits, and appropriately monitor their glucose levels were more likely to meet their glycemic goals.
Statistical Analysis
Univariate descriptive statistics described the individual variables/predictors of HbA1c goal attainment. As the study’s purpose was to identify patient factors and characteristics associated with HbA1c goal attainment, a logistic regression model framework was used for all covariates to evaluate each measured variable’s independent association with HbA1c. The univariate tests were used to compare patient characteristics between the 2 study groups: Pearson chi-square test was used for nominal data, and a paired t test (for normally distributed data) or Wilcoxon rank sum test (for non-normally distributed data) was used for continuous variables. Variables having a P value < .2 underwent a multivariate analysis stepwise logistic regression model to identify patient factors and characteristics associated with HbA1c goal attainment. A Fisher exact test was used to determine gender effect on HbA1c goal attainment, categoric variables were analyzed using Pearson chi-square test, and an unpaired t test was used for continuous data. The backward elimination approach to inclusion of variables in the model was used to build the most parsimonious and best-fitting model, and the Hosmer-Lemeshow goodness-of-fit tests was used to assess model fit. Data analyses were performed using IBM SPSS, version 18.0 (Armonk, NY).
Results
Five hundred eighty-four patient records were reviewed, and 207 patients met inclusion criteria: 102 patient records were reviewed for the goal-attained arm, and 105 patient records for the goal-not-attained arm. Most patients were excluded from the analysis due to not having 3 consecutive visits during the specified period or having an HbA1c of < 8% at the time of referral to the pharmacist DSM clinic.
The patients in this study had type 2 diabetes for about 11 years, were overwhelmingly male (99%), were aged about 61 years, and were taking on average 13 medications at the time of referral to the pharmacist DSM clinic. Mean HbA1c at time of enrollment was slightly higher in the goal-not-attained arm vs goal-attained arm (10.7% vs 10.2%, respectively), but the difference was not statistically significant (P = .066). A little more than half the patients in both study arms were on basal + prandial insulin regimens (Table 2).
Patients who attained their goal HbA1cwere more likely to bring their glucose meter/glucose log book to at least 80% of their appointments (P < .001). Additionally, this same cohort followed insulin dosing instructions at least 80% of the time (P < .001).
Five variables were included in the multivariate analysis because they had a P value ≤ .2 in univariate analyses: (1) patient adherence to instructions (P < .001); (2) duration in clinic (P < .001); (3) patient bringingglucose meter or glucose log to appointments (P < .001); (4) percentage of scheduled appointments patient attended (P = .015); and (5) baseline HbA1c (P = .066).
Discussion
The development and constant modification of clinical practicing guidelines has made DM treatment a focus and priority.3,4 Additionally, the collaborative approach to health care and creation of VA pharmacist-driven services have demonstrated successful patient outcomes.6-16 Despite these efforts, further insight is needed to improve the management of DM. Our study identified specific behavioral factors that correlated to veteran patients to attaining their HbA1c goal of < 8% within a VA pharmacist DSM clinic. Additionally, it highlighted factors that contributed to patients not achieving their glycemic goals.
Our univariate analysis showed behaviors such as showing up for appointments and following directions regimens to correlate with glycemic goal attainment. However, following directions was the only behavioral factor that correlated to glycemic goal attainment in our multivariate analysis. Additionally, our findings indicated that factors for HbA1c goal attainment included patients who brought their glucose meter/glucose log book and attended clinic appointments at least 80% of the time, respectively.
These findings can help further refine the process for identifying patients who are most likely to achieve glycemic goals when referred to pharmacist DSM clinics or to any DM treatment program. Assessment of a patient’s motivation and ability to attend clinic appointments, bring their glucose meter/glucose log book, and to follow instructions provided at these appointments are reasonable screening questions to ask before referring that patient to a diabetes care program or service. Currently, this is not performed during the consult process to the pharmacist DSM clinic at the respective VA.
Additionally, our findings show that patients who met goal did so, on average, within 6 months of referral to the pharmacist DSM clinic. This finding may have occurred because patients who successfully reach HbA1c goal in 2 consecutive checks are discharged from the clinic. Patients who do not meet this goal continue with the clinic, thus increasing their duration of enrollment in this service. This finding could help clinical pharmacists estimate how long patients will be followed by the service, thus allowing for a more accurate estimation of workload and clinic capacity. Additionally, this finding provides insight if the patient should remain in clinic or be transferred to another program. Our findings aligned with previous studies showing the link between patient behaviors and glycemic goal attainment.17-19
Limitations
This study has a few notable limitations. First, it is limited to 1 VA medical center, so our findings may not be extrapolated easily to other institutions of the Veterans Health Administration. Ideally, future studies centered on identifying factors that lead to successful glycemic goal attainment would be helpful from multiple VA institutions. This would encourage more factors to be identified and trends to be strengthened. Ultimately, this would allow for more global changes to the consult process from primary care to pharmacist DSM clinics nationally vs at a local VA institution. Additionally, this study was limited to a specific retrospective time frame, therefore limiting its ability to identify trends. This study also relied on some subjective factors, such as the patient’s self-report of properly following the clinic instructions. Another limitation was that our investigation was not designed to characterize the specific pharmacist’s interventions that improved glycemic control. Future studies would benefit from the inclusion of specific interventions and their effect on glycemic goal attainment.
Conclusion
This retrospective study offers insight to specific patient behavioral factors that correlate with glycemic goal attainment in a VA pharmacist DSM clinic. Behavioral factors linked to HbA1c goal attainment of < 8% included appointment keeping, bringing glucose meter/glucose log book at least 80% of the time to these appointments, and following clinic instructions. This investigation also found that patients who attain glycemic goals generally do so within 6 months of enrollment. Furthermore, this study provided insight that following the clinic instructions a majority of the time strongly contributes to glycemic goal attainment. We believe that an assessment of patients’ behaviors prior to referrals to diabetes management programs will yield useful information about possible barriers to glycemic goal attainment.
About 30.3 million Americans (9.4%) have diabetes mellitus (DM).1 Veterans are disproportionately affected—about 1 in 4 of those who receive US Department of Veterans Affairs (VA) care have DM.2 The consequences of uncontrolled DM include microvascular complications (eg, retinopathy, neuropathy, and nephropathy) and macrovascular complications (eg, cardiovascular disease).
The American Diabetes Association (ADA) recommends achieving a goal hemoglobin A1c (HbA1c) level of < 7% to prevent these complications. However, a goal of < 8% HbA1c may be more appropriate for certain patients when a more strict goal may be impractical or have the potential to cause harm.3 Furthermore, guidelines developed by the VA and the US Department of Defense suggest a target HbA1c range of 7.0% to 8.5% for patients with established microvascular or macrovascular disease, comorbid conditions, or a life expectancy of 5 to 10 years.4
Despite the existence of evidence showing the importance of glycemic control in preventing morbidity and mortality associated with DM, many patients have inadequate glycemic control. Diabetes mellitus is the seventh leading cause of death in the US. Moreover, DM is a known risk factor for heart disease, stroke, and kidney disease, which are the first, fifth, and ninth leading causes of death in the US, respectively.5
Because DM management requires ongoing and comprehensive maintenance and monitoring, the ADA supports a collaborative, multidisciplinary, and patient-centered approach to delivery of care.3 Collaborative teams involving pharmacists have been shown to improve outcomes and cost savings for chronic diseases, including DM.6-12 In 1995, the VA launched a national policy providing clinical pharmacists with prescribing privileges that would aid in the provision of coordinated medication management for patients with chronic illnesses.13 The policy created a framework for collaborative drug therapy management (CDTM) models, which grants pharmacists the ability to perform patient assessments, order laboratory tests, and modify medications within a scope of practice.
Since the initiation of these services, several examples of successful DM management services using clinical pharmacists within the VA exist in the literature.14-16 However, even with intensive chronic disease and drug therapy management, not all patients who enroll in these services successfully reach clinical goals. Although these pharmacist-driven services seem to demonstrate overall benefit and cost savings to veteran patients and the VA system, little published data exist to help determine patient behaviors that are associated with glycemic goal attainment when using these services.
At the Corporal Michael J. Crescenz VA Medical Center in (CMCVAMC) Philadelphia, Pennsylvania, where this study was performed, primary care providers may refer patients with uncontrolled DM to the pharmacist disease state management (DSM) clinic. The clinic is a form of a CDTM and receives numerous referrals per year, with many patients discharged for successfully meeting glycemic targets.
However, a percentage of patients fail to attain glycemic goals despite involvement in this clinic. We observed specific patient behaviors that delayed glycemic goal attainment. This study examined whether these behaviors correlated with prolonged glycemic goal attainment. The purpose of this study was to identify patient behaviors that led to glycemic goal attainment in insulin-treated patients referred to this pharmacist DSM clinic.
Methods
This study was performed as a single-center retrospective chart review. The protocol and data collection documents were approved by the CMCVAMC Institutional Review Board. It included patients referred to a pharmacist-led DSM clinic for insulin titration/optimization from January 1, 2011 through December 31, 2012. Data were collected through June 30, 2013, to allow for 6 months after the last referral date of December 31, 2012.
This study included patients who were on insulin therapy at the time of pharmacy consult, who attended at least 3 consecutive pharmacy DSM clinic visits, and had an HbA1c ≥ 8% at the time of initial clinic consult. Patients who failed to have 3 consecutive pharmacy DSM clinic visits, were insulin-naïve at the time of referral, aged ≥ 90, lacked at least 1 follow-up HbA1c result while enrolled in the clinic, or had HbA1c < 8% were excluded.
Among the patients who met eligibility criteria, charts within the Computerized Patient Record System (CPRS) were reviewed in a chronologic order within the respective study time frame. A convenience sample of 100 patients were enrolled in each treatment arm: the goal-attained arm or the goal-not-attained arm.
The primary study variable was HbA1c goal attainment, which was defined in this investigation as at least 1 HbA1c reading of < 8% while enrolled in the DSM clinic during the review period. Secondary variables included specific patient factors such as optimal frequency of self-monitoring of blood glucose (SMBG) testing, adherence to pharmacist’s instructions for changes to glucose-lowering medications, adherence to bringing glucose meter/glucose log book to clinic appointments, and percentage of visits attended. Definitions for each variable are provided in Table 1.
We hypothesized that patients who were more adherent to treatment plans, regularly attend clinic visits, and appropriately monitor their glucose levels were more likely to meet their glycemic goals.
Statistical Analysis
Univariate descriptive statistics described the individual variables/predictors of HbA1c goal attainment. As the study’s purpose was to identify patient factors and characteristics associated with HbA1c goal attainment, a logistic regression model framework was used for all covariates to evaluate each measured variable’s independent association with HbA1c. The univariate tests were used to compare patient characteristics between the 2 study groups: Pearson chi-square test was used for nominal data, and a paired t test (for normally distributed data) or Wilcoxon rank sum test (for non-normally distributed data) was used for continuous variables. Variables having a P value < .2 underwent a multivariate analysis stepwise logistic regression model to identify patient factors and characteristics associated with HbA1c goal attainment. A Fisher exact test was used to determine gender effect on HbA1c goal attainment, categoric variables were analyzed using Pearson chi-square test, and an unpaired t test was used for continuous data. The backward elimination approach to inclusion of variables in the model was used to build the most parsimonious and best-fitting model, and the Hosmer-Lemeshow goodness-of-fit tests was used to assess model fit. Data analyses were performed using IBM SPSS, version 18.0 (Armonk, NY).
Results
Five hundred eighty-four patient records were reviewed, and 207 patients met inclusion criteria: 102 patient records were reviewed for the goal-attained arm, and 105 patient records for the goal-not-attained arm. Most patients were excluded from the analysis due to not having 3 consecutive visits during the specified period or having an HbA1c of < 8% at the time of referral to the pharmacist DSM clinic.
The patients in this study had type 2 diabetes for about 11 years, were overwhelmingly male (99%), were aged about 61 years, and were taking on average 13 medications at the time of referral to the pharmacist DSM clinic. Mean HbA1c at time of enrollment was slightly higher in the goal-not-attained arm vs goal-attained arm (10.7% vs 10.2%, respectively), but the difference was not statistically significant (P = .066). A little more than half the patients in both study arms were on basal + prandial insulin regimens (Table 2).
Patients who attained their goal HbA1cwere more likely to bring their glucose meter/glucose log book to at least 80% of their appointments (P < .001). Additionally, this same cohort followed insulin dosing instructions at least 80% of the time (P < .001).
Five variables were included in the multivariate analysis because they had a P value ≤ .2 in univariate analyses: (1) patient adherence to instructions (P < .001); (2) duration in clinic (P < .001); (3) patient bringingglucose meter or glucose log to appointments (P < .001); (4) percentage of scheduled appointments patient attended (P = .015); and (5) baseline HbA1c (P = .066).
Discussion
The development and constant modification of clinical practicing guidelines has made DM treatment a focus and priority.3,4 Additionally, the collaborative approach to health care and creation of VA pharmacist-driven services have demonstrated successful patient outcomes.6-16 Despite these efforts, further insight is needed to improve the management of DM. Our study identified specific behavioral factors that correlated to veteran patients to attaining their HbA1c goal of < 8% within a VA pharmacist DSM clinic. Additionally, it highlighted factors that contributed to patients not achieving their glycemic goals.
Our univariate analysis showed behaviors such as showing up for appointments and following directions regimens to correlate with glycemic goal attainment. However, following directions was the only behavioral factor that correlated to glycemic goal attainment in our multivariate analysis. Additionally, our findings indicated that factors for HbA1c goal attainment included patients who brought their glucose meter/glucose log book and attended clinic appointments at least 80% of the time, respectively.
These findings can help further refine the process for identifying patients who are most likely to achieve glycemic goals when referred to pharmacist DSM clinics or to any DM treatment program. Assessment of a patient’s motivation and ability to attend clinic appointments, bring their glucose meter/glucose log book, and to follow instructions provided at these appointments are reasonable screening questions to ask before referring that patient to a diabetes care program or service. Currently, this is not performed during the consult process to the pharmacist DSM clinic at the respective VA.
Additionally, our findings show that patients who met goal did so, on average, within 6 months of referral to the pharmacist DSM clinic. This finding may have occurred because patients who successfully reach HbA1c goal in 2 consecutive checks are discharged from the clinic. Patients who do not meet this goal continue with the clinic, thus increasing their duration of enrollment in this service. This finding could help clinical pharmacists estimate how long patients will be followed by the service, thus allowing for a more accurate estimation of workload and clinic capacity. Additionally, this finding provides insight if the patient should remain in clinic or be transferred to another program. Our findings aligned with previous studies showing the link between patient behaviors and glycemic goal attainment.17-19
Limitations
This study has a few notable limitations. First, it is limited to 1 VA medical center, so our findings may not be extrapolated easily to other institutions of the Veterans Health Administration. Ideally, future studies centered on identifying factors that lead to successful glycemic goal attainment would be helpful from multiple VA institutions. This would encourage more factors to be identified and trends to be strengthened. Ultimately, this would allow for more global changes to the consult process from primary care to pharmacist DSM clinics nationally vs at a local VA institution. Additionally, this study was limited to a specific retrospective time frame, therefore limiting its ability to identify trends. This study also relied on some subjective factors, such as the patient’s self-report of properly following the clinic instructions. Another limitation was that our investigation was not designed to characterize the specific pharmacist’s interventions that improved glycemic control. Future studies would benefit from the inclusion of specific interventions and their effect on glycemic goal attainment.
Conclusion
This retrospective study offers insight to specific patient behavioral factors that correlate with glycemic goal attainment in a VA pharmacist DSM clinic. Behavioral factors linked to HbA1c goal attainment of < 8% included appointment keeping, bringing glucose meter/glucose log book at least 80% of the time to these appointments, and following clinic instructions. This investigation also found that patients who attain glycemic goals generally do so within 6 months of enrollment. Furthermore, this study provided insight that following the clinic instructions a majority of the time strongly contributes to glycemic goal attainment. We believe that an assessment of patients’ behaviors prior to referrals to diabetes management programs will yield useful information about possible barriers to glycemic goal attainment.
1. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Published 2017. Accessed September 25, 2018.
2. Gaspar JL, Dahlke ME, Kasper B. Efficacy of patient aligned care team pharmacist service in reaching diabetes and hyperlipidemia treatment goals. Fed Pract. 2015;32(11):42-47.
3. American Diabetes Association. Standards of medical care in diabetes—2017. Diabetes Care. 2017;40(suppl 1):S6-S135.
4. US Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. https://www.healthquality.va.gov/guidelines/CD/diabetes/VADoDDMCPGFinal508.pdf. Published April 2017. Accessed September 7, 2018.
5. Centers for Disease Control and Prevention. Deaths: leading causes for 2014. Natl Vital Stat Rep. 2016;65(5):1-96.
6. Nigro SC, Garwood CL, Berlie H, et al. Clinical pharmacists as key members of the patient-centered medical home: an opinion statement of the Ambulatory Care Practice and Research Network of the American College of Clinical Pharmacy. Pharmacotherapy. 2014;34(1):96-108.
7. Smith M, Bates DW, Bodenheimer T, et al. Why pharmacists belong in the medical home. Health Aff (Millwood). 2010;29(5):906-913.
8. Chisholm-Burns MA, Kim Lee J, Spivey CA, et al. US Pharmacists’ effect as team members on patient care. Med Care. 2010;48(10):923-933.
9. Wubben DP, Vivian EM. Effects of pharmacist outpatient interventions on adults with diabetes mellitus: a systematic review. Pharmacotherapy. 2008;28(4):421-436.
10. Touchette DR, Doloresco F, Suda KJ, et al. Economic evaluations of clinical pharmacy services: 2006-2010. Pharmacotherapy. 2014;34(8):771-793.
11. Giberson S, Yoder S, Lee MP. Improving patient and health system outcomes through advanced pharmacy practice. A report of the U.S. Surgeon General. American College of Clinical Pharmacy. https://www.accp.com/docs/positions/misc/Improving_Patient_and_Health_System_Outcomes.pdf. Published December 2011. Accessed September 10, 2018.
12. Isetts BJ, Schondelmeyer SW, Artz MB, et al. Clinical and economic outcomes of medication therapy management services: the Minnesota experience. J Am Pharm Assoc (2003). 2008;48(2):203-211.
13. Ourth H, Groppi J, Morreale AP, Quicci-Roberts K. Clinical pharmacist prescribing activities in the Veterans Health Administration. Am J Health Syst Pharm. 2016;73(18):1406-1415.
14. Taveira TH, Friedmann PD, Cohen LB, et al. Pharmacist-led group medical appointment model in type 2 diabetes. Diabetes Educ. 2010;36(1):109-117.
15. Edwards KL, Hadley RL, Baby N, Yeary JC, Chastain LM, Brown CD. Utilizing clinical pharmacy specialists to address access to care barriers in the veteran population for the management of diabetes. J Pharm Pract. 2017;30(4):412-418.
16. Cripps RJ, Gourley ES, Johnson W, et al. An evaluation of diabetes-related measures of control after 6 months of clinical pharmacy specialist intervention. J Pharm Prac. 2011;24(3):332-338.
17. Jones H, Edwards L, Vallis TM, et al; Diabetes Stages of Change (DiSC) Study. Changes in diabetes self-care behaviors make a difference in glycemic control. Diabetes Care. 2003;26(3):732-737.
18. Schetman JM, Schorling JB, Voss JD. Appointment adherence and disparities in outcomes among patients with diabetes. J Gen Intern Med. 2008;23(10):1685-1687.
19. Rhee, MK, Slocum W, Zeimer DC, et al. Patient adherence improves glycemic control. Diabetes Educ. 2005;31(2):240-250.
1. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Published 2017. Accessed September 25, 2018.
2. Gaspar JL, Dahlke ME, Kasper B. Efficacy of patient aligned care team pharmacist service in reaching diabetes and hyperlipidemia treatment goals. Fed Pract. 2015;32(11):42-47.
3. American Diabetes Association. Standards of medical care in diabetes—2017. Diabetes Care. 2017;40(suppl 1):S6-S135.
4. US Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. https://www.healthquality.va.gov/guidelines/CD/diabetes/VADoDDMCPGFinal508.pdf. Published April 2017. Accessed September 7, 2018.
5. Centers for Disease Control and Prevention. Deaths: leading causes for 2014. Natl Vital Stat Rep. 2016;65(5):1-96.
6. Nigro SC, Garwood CL, Berlie H, et al. Clinical pharmacists as key members of the patient-centered medical home: an opinion statement of the Ambulatory Care Practice and Research Network of the American College of Clinical Pharmacy. Pharmacotherapy. 2014;34(1):96-108.
7. Smith M, Bates DW, Bodenheimer T, et al. Why pharmacists belong in the medical home. Health Aff (Millwood). 2010;29(5):906-913.
8. Chisholm-Burns MA, Kim Lee J, Spivey CA, et al. US Pharmacists’ effect as team members on patient care. Med Care. 2010;48(10):923-933.
9. Wubben DP, Vivian EM. Effects of pharmacist outpatient interventions on adults with diabetes mellitus: a systematic review. Pharmacotherapy. 2008;28(4):421-436.
10. Touchette DR, Doloresco F, Suda KJ, et al. Economic evaluations of clinical pharmacy services: 2006-2010. Pharmacotherapy. 2014;34(8):771-793.
11. Giberson S, Yoder S, Lee MP. Improving patient and health system outcomes through advanced pharmacy practice. A report of the U.S. Surgeon General. American College of Clinical Pharmacy. https://www.accp.com/docs/positions/misc/Improving_Patient_and_Health_System_Outcomes.pdf. Published December 2011. Accessed September 10, 2018.
12. Isetts BJ, Schondelmeyer SW, Artz MB, et al. Clinical and economic outcomes of medication therapy management services: the Minnesota experience. J Am Pharm Assoc (2003). 2008;48(2):203-211.
13. Ourth H, Groppi J, Morreale AP, Quicci-Roberts K. Clinical pharmacist prescribing activities in the Veterans Health Administration. Am J Health Syst Pharm. 2016;73(18):1406-1415.
14. Taveira TH, Friedmann PD, Cohen LB, et al. Pharmacist-led group medical appointment model in type 2 diabetes. Diabetes Educ. 2010;36(1):109-117.
15. Edwards KL, Hadley RL, Baby N, Yeary JC, Chastain LM, Brown CD. Utilizing clinical pharmacy specialists to address access to care barriers in the veteran population for the management of diabetes. J Pharm Pract. 2017;30(4):412-418.
16. Cripps RJ, Gourley ES, Johnson W, et al. An evaluation of diabetes-related measures of control after 6 months of clinical pharmacy specialist intervention. J Pharm Prac. 2011;24(3):332-338.
17. Jones H, Edwards L, Vallis TM, et al; Diabetes Stages of Change (DiSC) Study. Changes in diabetes self-care behaviors make a difference in glycemic control. Diabetes Care. 2003;26(3):732-737.
18. Schetman JM, Schorling JB, Voss JD. Appointment adherence and disparities in outcomes among patients with diabetes. J Gen Intern Med. 2008;23(10):1685-1687.
19. Rhee, MK, Slocum W, Zeimer DC, et al. Patient adherence improves glycemic control. Diabetes Educ. 2005;31(2):240-250.
Tiotropium/olodaterol improved lung function in steroid-free patients with COPD
New Orleans – compared with tiotropium alone, according to results of a pooled analysis of phase 3 trial data.
Lung function, symptoms, and quality of life were all improved irrespective of disease severity and baseline symptoms, and these benefits were apparent across multiple subgroups of non-ICS patients, investigator Peter M. Calverley, MBChB, of the University of Liverpool (England) reported at the annual meeting of the American College of Chest Physicians.
“Perhaps frustratingly, we can’t personalize [treatment] – everybody seemed to get benefit across the board, and the magnitude of that benefit was rather similar,” Dr. Calverley said in a podium presentation of the results.
The subgroup analysis was undertaken to fill a knowledge gap, according to Dr. Calverley, regarding the efficacy of combined long-acting muscarinic antagonist/long-acting beta2-agonist (LAMA/LABA) treatment in the sizable proportion of COPD patients who are not receiving ICS.
Patients in LAMA/LABA clinical trials are frequently taking multiple therapies, including ICS, he explained.
The post hoc analysis of pooled data from the TONADO 1 and 2 and OTEMTO 1 and 2 trials included a total of 1,596 patients with COPD who were not receiving ICS at trial enrollment.
The no-ICS patients receiving tiotropium/olodaterol had a significantly greater improvement in trough forced expiratory volume in 1 second (FEV1) response by week 12 of treatment, compared with patients receiving tiotropium alone (0.054 L; P less than .0001), the researcher reported.
The trough FEV1 improvement accruing to the tiotropium/olodaterol–treated patients was consistent across subgroups stratified according to GOLD stage and symptoms at baseline (such as Baseline Dyspnea Index), he added.
Transition Dyspnea Index (TDI) score at 12 weeks in these patients not receiving ICS was likewise superior in the tiotropium/olodaterol treated patients, compared with the tiotropium monotherapy group (.575; P less than .0001), while changes in St. George’s Respiratory Questionnaire (SGRQ) total scores also favored the combination treatment, according to the analyses presented.
Taken together, results of this post hoc analysis support the use of dual bronchodilator therapy to improve key COPD outcomes, compared with single bronchodilator therapy. In addition, this strategy should be considered as an earlier treatment option, according to Dr. Calverley.
“Clearly dual bronchodilator therapy improves the key outcomes, compared with an effective single bronchodilator, and this perhaps is why it’s being picked up as an earlier treatment option, and certainly in some guidelines like the current [National Institute for Health Care and Excellence] guidelines where I live in the U.K., it’s becoming standard of care for most people with COPD,” he said in his presentation.
Dr. Calverley reported grants and personal fees from GlaxoSmithKline; personal fees from AstraZeneca, Recipharm, and Zambon; and personal and other fees from Boehringer Ingelheim outside the submitted work.
SOURCE: Calverley PM et al. CHEST 2019. Abstract, doi: 10.1016/j.chest.2019.08.310.
New Orleans – compared with tiotropium alone, according to results of a pooled analysis of phase 3 trial data.
Lung function, symptoms, and quality of life were all improved irrespective of disease severity and baseline symptoms, and these benefits were apparent across multiple subgroups of non-ICS patients, investigator Peter M. Calverley, MBChB, of the University of Liverpool (England) reported at the annual meeting of the American College of Chest Physicians.
“Perhaps frustratingly, we can’t personalize [treatment] – everybody seemed to get benefit across the board, and the magnitude of that benefit was rather similar,” Dr. Calverley said in a podium presentation of the results.
The subgroup analysis was undertaken to fill a knowledge gap, according to Dr. Calverley, regarding the efficacy of combined long-acting muscarinic antagonist/long-acting beta2-agonist (LAMA/LABA) treatment in the sizable proportion of COPD patients who are not receiving ICS.
Patients in LAMA/LABA clinical trials are frequently taking multiple therapies, including ICS, he explained.
The post hoc analysis of pooled data from the TONADO 1 and 2 and OTEMTO 1 and 2 trials included a total of 1,596 patients with COPD who were not receiving ICS at trial enrollment.
The no-ICS patients receiving tiotropium/olodaterol had a significantly greater improvement in trough forced expiratory volume in 1 second (FEV1) response by week 12 of treatment, compared with patients receiving tiotropium alone (0.054 L; P less than .0001), the researcher reported.
The trough FEV1 improvement accruing to the tiotropium/olodaterol–treated patients was consistent across subgroups stratified according to GOLD stage and symptoms at baseline (such as Baseline Dyspnea Index), he added.
Transition Dyspnea Index (TDI) score at 12 weeks in these patients not receiving ICS was likewise superior in the tiotropium/olodaterol treated patients, compared with the tiotropium monotherapy group (.575; P less than .0001), while changes in St. George’s Respiratory Questionnaire (SGRQ) total scores also favored the combination treatment, according to the analyses presented.
Taken together, results of this post hoc analysis support the use of dual bronchodilator therapy to improve key COPD outcomes, compared with single bronchodilator therapy. In addition, this strategy should be considered as an earlier treatment option, according to Dr. Calverley.
“Clearly dual bronchodilator therapy improves the key outcomes, compared with an effective single bronchodilator, and this perhaps is why it’s being picked up as an earlier treatment option, and certainly in some guidelines like the current [National Institute for Health Care and Excellence] guidelines where I live in the U.K., it’s becoming standard of care for most people with COPD,” he said in his presentation.
Dr. Calverley reported grants and personal fees from GlaxoSmithKline; personal fees from AstraZeneca, Recipharm, and Zambon; and personal and other fees from Boehringer Ingelheim outside the submitted work.
SOURCE: Calverley PM et al. CHEST 2019. Abstract, doi: 10.1016/j.chest.2019.08.310.
New Orleans – compared with tiotropium alone, according to results of a pooled analysis of phase 3 trial data.
Lung function, symptoms, and quality of life were all improved irrespective of disease severity and baseline symptoms, and these benefits were apparent across multiple subgroups of non-ICS patients, investigator Peter M. Calverley, MBChB, of the University of Liverpool (England) reported at the annual meeting of the American College of Chest Physicians.
“Perhaps frustratingly, we can’t personalize [treatment] – everybody seemed to get benefit across the board, and the magnitude of that benefit was rather similar,” Dr. Calverley said in a podium presentation of the results.
The subgroup analysis was undertaken to fill a knowledge gap, according to Dr. Calverley, regarding the efficacy of combined long-acting muscarinic antagonist/long-acting beta2-agonist (LAMA/LABA) treatment in the sizable proportion of COPD patients who are not receiving ICS.
Patients in LAMA/LABA clinical trials are frequently taking multiple therapies, including ICS, he explained.
The post hoc analysis of pooled data from the TONADO 1 and 2 and OTEMTO 1 and 2 trials included a total of 1,596 patients with COPD who were not receiving ICS at trial enrollment.
The no-ICS patients receiving tiotropium/olodaterol had a significantly greater improvement in trough forced expiratory volume in 1 second (FEV1) response by week 12 of treatment, compared with patients receiving tiotropium alone (0.054 L; P less than .0001), the researcher reported.
The trough FEV1 improvement accruing to the tiotropium/olodaterol–treated patients was consistent across subgroups stratified according to GOLD stage and symptoms at baseline (such as Baseline Dyspnea Index), he added.
Transition Dyspnea Index (TDI) score at 12 weeks in these patients not receiving ICS was likewise superior in the tiotropium/olodaterol treated patients, compared with the tiotropium monotherapy group (.575; P less than .0001), while changes in St. George’s Respiratory Questionnaire (SGRQ) total scores also favored the combination treatment, according to the analyses presented.
Taken together, results of this post hoc analysis support the use of dual bronchodilator therapy to improve key COPD outcomes, compared with single bronchodilator therapy. In addition, this strategy should be considered as an earlier treatment option, according to Dr. Calverley.
“Clearly dual bronchodilator therapy improves the key outcomes, compared with an effective single bronchodilator, and this perhaps is why it’s being picked up as an earlier treatment option, and certainly in some guidelines like the current [National Institute for Health Care and Excellence] guidelines where I live in the U.K., it’s becoming standard of care for most people with COPD,” he said in his presentation.
Dr. Calverley reported grants and personal fees from GlaxoSmithKline; personal fees from AstraZeneca, Recipharm, and Zambon; and personal and other fees from Boehringer Ingelheim outside the submitted work.
SOURCE: Calverley PM et al. CHEST 2019. Abstract, doi: 10.1016/j.chest.2019.08.310.
REPORTING FROM CHEST 2019
Atezolizumab bests chemo in NSCLC patients with high PD-L1 expression
NATIONAL HARBOR, MD – interim results of a phase 3 trial suggest.
When compared with platinum-based chemotherapy, atezolizumab significantly improved OS in patients who had PD-L1 expression of 50% or greater on tumor cells (TC) or 10% or greater on tumor-infiltrating immune cells (IC).
Patients with lower levels of PD-L1 expression had a numerical, but not statistically significant, benefit in OS with atezolizumab.
Giuseppe Giaccone, MD, PhD, of Georgetown University, Washington, presented these results, from the IMpower110 trial, as a late-breaking abstract at the annual meeting of the Society for Immunotherapy of Cancer.
The trial enrolled treatment-naive, PD-L1–selected patients with stage IV NSCLC. Patients with EGFR-positive or ALK-positive NSCLC were excluded.
The overall population consisted of 554 patients with PD-L1 expression of at least 1% on TC or IC (TC1/2/3 or IC1/2/3). There were 328 patients with PD-L1 expression of at least 5% on TC or IC (TC2/3 or IC2/3) and 205 patients with PD-L1 expression of at least 50% on TC or 10% on IC (TC3 or IC3).
All patients were randomized to receive atezolizumab or platinum-based chemotherapy. Atezolizumab was given at 1,200 mg every 3 weeks until disease progression or loss of clinical benefit. In the chemotherapy arm, patients with nonsquamous NSCLC received cisplatin or carboplatin plus pemetrexed, and patients with squamous NSCLC received cisplatin or carboplatin plus gemcitabine. Chemotherapy was given for 4-6 cycles, after which patients received pemetrexed (nonsquamous) or best supportive care (squamous) until progression.
Baseline characteristics were similar between the treatment arms and across the PD-L1 subgroups.
Overall survival
OS was evaluated hierarchically, starting with the TC3 or IC3 population. This group had a significant improvement in OS with atezolizumab. At a median follow-up of 15.7 months, the median OS was 13.1 months with chemotherapy and 20.2 months with atezolizumab (hazard ratio, 0.59; P = .0106).
The TC2/3 or IC2/3 population had a numerical, but not significant, improvement in OS with atezolizumab. At a median follow-up of 15.2 months, the median OS was 14.9 months with chemotherapy and 18.2 months with atezolizumab (HR, 0.72; P = .0416).
“The prespecified alpha boundary was not crossed in this analysis,” Dr. Giaccone said. “Therefore, it cannot be considered statistically significant, although, numerically, there is clearly an advantage with atezolizumab versus chemotherapy.”
There was no significant improvement in OS in the overall population (TC1/2/3 or IC1/2/3). At a median follow-up of 13.4 months, the median OS was 14.1 months in the chemotherapy arm and 17.5 months in the atezolizumab arm (HR, 0.83; P = .1481).
Dr. Giaccone noted that 29.6% of patients in the atezolizumab arm and 49.5% of those in the chemotherapy arm received at least one subsequent cancer therapy. The proportion of patients that received second-line cancer therapies was similar across the PD-L1 subgroups.
Progression-free survival
Progression-free survival (PFS) was significantly better with atezolizumab, regardless of PD-L1 expression.
In the TC3/IC3 population, the median PFS was 5.0 months with chemotherapy and 8.1 months with atezolizumab (HR, 0.63; P = .0070). In the TC2/3 or IC2/3 group, the median PFS was 5.5 months with chemotherapy and 7.2 months with atezolizumab (HR, 0.67; P = .0030). In the overall population, the median PFS was 5.5 months with chemotherapy and 5.7 months with atezolizumab (HR, 0.77; P = .0104).
Safety
“The safety profile of atezolizumab was consistent with prior observations,” Dr. Giaccone said.
The incidence of treatment-related adverse events (AEs) was 90.2% in the atezolizumab arm and 94.7% in the chemotherapy arm. Rates of grade 3/4 related AEs were 12.9% and 44.1%, respectively. There was one grade 5 AE in the chemotherapy arm and none in the atezolizumab arm.
AEs that were more frequent with atezolizumab included increased aspartate aminotransferase, pruritus, and hypothyroidism.
This trial is sponsored by Hoffmann-La Roche, and Dr. Giaccone disclosed grants and nonfinancial support from the company.
SOURCE: Giaccone G et al. SITC 2019, Abstract O81.
NATIONAL HARBOR, MD – interim results of a phase 3 trial suggest.
When compared with platinum-based chemotherapy, atezolizumab significantly improved OS in patients who had PD-L1 expression of 50% or greater on tumor cells (TC) or 10% or greater on tumor-infiltrating immune cells (IC).
Patients with lower levels of PD-L1 expression had a numerical, but not statistically significant, benefit in OS with atezolizumab.
Giuseppe Giaccone, MD, PhD, of Georgetown University, Washington, presented these results, from the IMpower110 trial, as a late-breaking abstract at the annual meeting of the Society for Immunotherapy of Cancer.
The trial enrolled treatment-naive, PD-L1–selected patients with stage IV NSCLC. Patients with EGFR-positive or ALK-positive NSCLC were excluded.
The overall population consisted of 554 patients with PD-L1 expression of at least 1% on TC or IC (TC1/2/3 or IC1/2/3). There were 328 patients with PD-L1 expression of at least 5% on TC or IC (TC2/3 or IC2/3) and 205 patients with PD-L1 expression of at least 50% on TC or 10% on IC (TC3 or IC3).
All patients were randomized to receive atezolizumab or platinum-based chemotherapy. Atezolizumab was given at 1,200 mg every 3 weeks until disease progression or loss of clinical benefit. In the chemotherapy arm, patients with nonsquamous NSCLC received cisplatin or carboplatin plus pemetrexed, and patients with squamous NSCLC received cisplatin or carboplatin plus gemcitabine. Chemotherapy was given for 4-6 cycles, after which patients received pemetrexed (nonsquamous) or best supportive care (squamous) until progression.
Baseline characteristics were similar between the treatment arms and across the PD-L1 subgroups.
Overall survival
OS was evaluated hierarchically, starting with the TC3 or IC3 population. This group had a significant improvement in OS with atezolizumab. At a median follow-up of 15.7 months, the median OS was 13.1 months with chemotherapy and 20.2 months with atezolizumab (hazard ratio, 0.59; P = .0106).
The TC2/3 or IC2/3 population had a numerical, but not significant, improvement in OS with atezolizumab. At a median follow-up of 15.2 months, the median OS was 14.9 months with chemotherapy and 18.2 months with atezolizumab (HR, 0.72; P = .0416).
“The prespecified alpha boundary was not crossed in this analysis,” Dr. Giaccone said. “Therefore, it cannot be considered statistically significant, although, numerically, there is clearly an advantage with atezolizumab versus chemotherapy.”
There was no significant improvement in OS in the overall population (TC1/2/3 or IC1/2/3). At a median follow-up of 13.4 months, the median OS was 14.1 months in the chemotherapy arm and 17.5 months in the atezolizumab arm (HR, 0.83; P = .1481).
Dr. Giaccone noted that 29.6% of patients in the atezolizumab arm and 49.5% of those in the chemotherapy arm received at least one subsequent cancer therapy. The proportion of patients that received second-line cancer therapies was similar across the PD-L1 subgroups.
Progression-free survival
Progression-free survival (PFS) was significantly better with atezolizumab, regardless of PD-L1 expression.
In the TC3/IC3 population, the median PFS was 5.0 months with chemotherapy and 8.1 months with atezolizumab (HR, 0.63; P = .0070). In the TC2/3 or IC2/3 group, the median PFS was 5.5 months with chemotherapy and 7.2 months with atezolizumab (HR, 0.67; P = .0030). In the overall population, the median PFS was 5.5 months with chemotherapy and 5.7 months with atezolizumab (HR, 0.77; P = .0104).
Safety
“The safety profile of atezolizumab was consistent with prior observations,” Dr. Giaccone said.
The incidence of treatment-related adverse events (AEs) was 90.2% in the atezolizumab arm and 94.7% in the chemotherapy arm. Rates of grade 3/4 related AEs were 12.9% and 44.1%, respectively. There was one grade 5 AE in the chemotherapy arm and none in the atezolizumab arm.
AEs that were more frequent with atezolizumab included increased aspartate aminotransferase, pruritus, and hypothyroidism.
This trial is sponsored by Hoffmann-La Roche, and Dr. Giaccone disclosed grants and nonfinancial support from the company.
SOURCE: Giaccone G et al. SITC 2019, Abstract O81.
NATIONAL HARBOR, MD – interim results of a phase 3 trial suggest.
When compared with platinum-based chemotherapy, atezolizumab significantly improved OS in patients who had PD-L1 expression of 50% or greater on tumor cells (TC) or 10% or greater on tumor-infiltrating immune cells (IC).
Patients with lower levels of PD-L1 expression had a numerical, but not statistically significant, benefit in OS with atezolizumab.
Giuseppe Giaccone, MD, PhD, of Georgetown University, Washington, presented these results, from the IMpower110 trial, as a late-breaking abstract at the annual meeting of the Society for Immunotherapy of Cancer.
The trial enrolled treatment-naive, PD-L1–selected patients with stage IV NSCLC. Patients with EGFR-positive or ALK-positive NSCLC were excluded.
The overall population consisted of 554 patients with PD-L1 expression of at least 1% on TC or IC (TC1/2/3 or IC1/2/3). There were 328 patients with PD-L1 expression of at least 5% on TC or IC (TC2/3 or IC2/3) and 205 patients with PD-L1 expression of at least 50% on TC or 10% on IC (TC3 or IC3).
All patients were randomized to receive atezolizumab or platinum-based chemotherapy. Atezolizumab was given at 1,200 mg every 3 weeks until disease progression or loss of clinical benefit. In the chemotherapy arm, patients with nonsquamous NSCLC received cisplatin or carboplatin plus pemetrexed, and patients with squamous NSCLC received cisplatin or carboplatin plus gemcitabine. Chemotherapy was given for 4-6 cycles, after which patients received pemetrexed (nonsquamous) or best supportive care (squamous) until progression.
Baseline characteristics were similar between the treatment arms and across the PD-L1 subgroups.
Overall survival
OS was evaluated hierarchically, starting with the TC3 or IC3 population. This group had a significant improvement in OS with atezolizumab. At a median follow-up of 15.7 months, the median OS was 13.1 months with chemotherapy and 20.2 months with atezolizumab (hazard ratio, 0.59; P = .0106).
The TC2/3 or IC2/3 population had a numerical, but not significant, improvement in OS with atezolizumab. At a median follow-up of 15.2 months, the median OS was 14.9 months with chemotherapy and 18.2 months with atezolizumab (HR, 0.72; P = .0416).
“The prespecified alpha boundary was not crossed in this analysis,” Dr. Giaccone said. “Therefore, it cannot be considered statistically significant, although, numerically, there is clearly an advantage with atezolizumab versus chemotherapy.”
There was no significant improvement in OS in the overall population (TC1/2/3 or IC1/2/3). At a median follow-up of 13.4 months, the median OS was 14.1 months in the chemotherapy arm and 17.5 months in the atezolizumab arm (HR, 0.83; P = .1481).
Dr. Giaccone noted that 29.6% of patients in the atezolizumab arm and 49.5% of those in the chemotherapy arm received at least one subsequent cancer therapy. The proportion of patients that received second-line cancer therapies was similar across the PD-L1 subgroups.
Progression-free survival
Progression-free survival (PFS) was significantly better with atezolizumab, regardless of PD-L1 expression.
In the TC3/IC3 population, the median PFS was 5.0 months with chemotherapy and 8.1 months with atezolizumab (HR, 0.63; P = .0070). In the TC2/3 or IC2/3 group, the median PFS was 5.5 months with chemotherapy and 7.2 months with atezolizumab (HR, 0.67; P = .0030). In the overall population, the median PFS was 5.5 months with chemotherapy and 5.7 months with atezolizumab (HR, 0.77; P = .0104).
Safety
“The safety profile of atezolizumab was consistent with prior observations,” Dr. Giaccone said.
The incidence of treatment-related adverse events (AEs) was 90.2% in the atezolizumab arm and 94.7% in the chemotherapy arm. Rates of grade 3/4 related AEs were 12.9% and 44.1%, respectively. There was one grade 5 AE in the chemotherapy arm and none in the atezolizumab arm.
AEs that were more frequent with atezolizumab included increased aspartate aminotransferase, pruritus, and hypothyroidism.
This trial is sponsored by Hoffmann-La Roche, and Dr. Giaccone disclosed grants and nonfinancial support from the company.
SOURCE: Giaccone G et al. SITC 2019, Abstract O81.
REPORTING FROM SITC 2019
Psilocybin research is mind expanding
Microdosing tied to enhanced divergent thinking in the short term
COPENHAGEN – A single dose of psilocybin taken in a supportive social setting was associated with demonstrably enhanced empathy, creative thinking, and subjective well-being lasting for up to 7 days in an observational study, Kim P.C. Kuypers, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
The participants in this uncontrolled study were healthy individuals curious to experience psilocybin in a controlled environment, but the observed benefits are of special interest because of psilocybin’s potential role in the treatment of psychiatric disorders, including depression and PTSD, in which preliminary study results have been quite encouraging, observed Dr. Kuypers, a neuropsychologist at Maastricht (the Netherlands) University.
For example, hallmarks of depression include psychological inflexibility, negative thoughts, and disturbed empathy. As this study showed, psilocybin addresses all those issues, she said.
according to Dr. Kuypers.
She and her coinvestigators studied 55 participants at a psilocybin retreat. These are events sprouting up across Europe to accommodate people who are curious about psilocybin and interested in trying it in a supportive setting. The investigators administered structured tests of convergent and divergent thinking, emotional empathy, and satisfaction with life before participants ingested the so-called magic mushroom, the morning after, and again 7 days later. In the Netherlands, the above-ground portion of the plant is an illegal substance, but the underground stem is not. The average dose was 27 mg, slightly higher than is often used in controlled laboratory studies; however, there were no adverse events.
The results showed that divergent thinking was enhanced the morning after taking psilocybin, an effect that was not sustained at the 7-day mark. In contrast, convergent thinking, emotional empathy, and a satisfaction-with-life scores above baseline were maintained 7 days after ingestion.
Psilocybin is a 5-HT2A agonist. It is considered a classic psychedelic, as are LSD, mescaline, and ayahuasca. These agents are often used recreationally to broaden consciousness, for relaxation, and to amplify emotions. Cave paintings indicate that psilocybin has been used medicinally for 11,000 years. But even early hunter-gatherers could not possibly have envisioned the current phenomenon of psilocybin microdosing to enhance work performance as adopted by hard-driving Silicon Valley professionals and similarly motivated strivers elsewhere around the world.
Microdosing for enhanced performance
Microdosing of psychedelics, especially psilocybin and LSD, is the practice of taking a low dose – typically 1/10th of a full recreational dose, which is too small an amount to cause full-blown perceptual alterations – once every several days in order to stimulate productivity. Psychedelic microdosing has garnered considerable mass media attention as an increasingly popular practice among younger professionals in the fields of computer science, engineering, mathematics, and technology. But it hasn’t been subjected to much scientific scrutiny.
Dr. Kuypers and colleagues wanted to find out whether it is actually effective and whether there are negative effects. They conducted an online questionnaire survey during a 5-month period of 2018 that drew 1,116 respondents, 80% of whom were currently microdosing, while the other 20% were former microdosers who had stopped completely.
The most common motivation for microdosing was indeed to stimulate productivity through increased focus, energy, and creativity. Almost half of microdosers indicated that they designed their own dosing schedule – typically once every 2-4 days – and two-thirds of microdosers were oblivious as to the consumed dose.
The most common reasons that respondents stopped microdosing were negative experiences or loss of interest because of lack of efficacy. The negative experiences typically involved acute anxiety or other psychological symptoms limited to when they were under the influence (Int J Neuropsychopharmacol. 2019 Jul 1;22[7]:426-34).
A subset of survey respondents microdosed to alleviate symptoms of a physician-diagnosed mental or physical health disorder. They were the focus of a separate analysis.
Microdosing for mental, physical health problems
Four hundred and ten survey respondents reported microdosing to self-treat a total of 901 physician-diagnosed mental and 161 physiologic conditions. They were asked three efficacy questions: Did it work? Did symptoms disappear? Did your quality of life improve?
The responses were disorder-specific. Individuals with anxiety disorders, ADHD, migraine, and other pain syndromes were the only ones to consistently rate microdosing as more effective than conventional treatment. Microdosing was rated less effective for symptom relief than were full-dose psychedelics only in respondents with depression or anxiety; individuals with other mental disorders or with physiological disorders rated the two dosing strategies similarly (Front Psychiatry. 2019 Sep 13. doi: 10.3389/fpsyt.2019.00672).
Future randomized controlled trials are warranted to more accurately assess efficacy claims for various psychedelics, optimal dosing, disorder specificity, and how they stack up compared with standard therapies, she said.
Dr. Kuypers reported having no financial conflicts of interest regarding her studies, which were conducted free of commercial support.
Microdosing tied to enhanced divergent thinking in the short term
Microdosing tied to enhanced divergent thinking in the short term
COPENHAGEN – A single dose of psilocybin taken in a supportive social setting was associated with demonstrably enhanced empathy, creative thinking, and subjective well-being lasting for up to 7 days in an observational study, Kim P.C. Kuypers, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
The participants in this uncontrolled study were healthy individuals curious to experience psilocybin in a controlled environment, but the observed benefits are of special interest because of psilocybin’s potential role in the treatment of psychiatric disorders, including depression and PTSD, in which preliminary study results have been quite encouraging, observed Dr. Kuypers, a neuropsychologist at Maastricht (the Netherlands) University.
For example, hallmarks of depression include psychological inflexibility, negative thoughts, and disturbed empathy. As this study showed, psilocybin addresses all those issues, she said.
according to Dr. Kuypers.
She and her coinvestigators studied 55 participants at a psilocybin retreat. These are events sprouting up across Europe to accommodate people who are curious about psilocybin and interested in trying it in a supportive setting. The investigators administered structured tests of convergent and divergent thinking, emotional empathy, and satisfaction with life before participants ingested the so-called magic mushroom, the morning after, and again 7 days later. In the Netherlands, the above-ground portion of the plant is an illegal substance, but the underground stem is not. The average dose was 27 mg, slightly higher than is often used in controlled laboratory studies; however, there were no adverse events.
The results showed that divergent thinking was enhanced the morning after taking psilocybin, an effect that was not sustained at the 7-day mark. In contrast, convergent thinking, emotional empathy, and a satisfaction-with-life scores above baseline were maintained 7 days after ingestion.
Psilocybin is a 5-HT2A agonist. It is considered a classic psychedelic, as are LSD, mescaline, and ayahuasca. These agents are often used recreationally to broaden consciousness, for relaxation, and to amplify emotions. Cave paintings indicate that psilocybin has been used medicinally for 11,000 years. But even early hunter-gatherers could not possibly have envisioned the current phenomenon of psilocybin microdosing to enhance work performance as adopted by hard-driving Silicon Valley professionals and similarly motivated strivers elsewhere around the world.
Microdosing for enhanced performance
Microdosing of psychedelics, especially psilocybin and LSD, is the practice of taking a low dose – typically 1/10th of a full recreational dose, which is too small an amount to cause full-blown perceptual alterations – once every several days in order to stimulate productivity. Psychedelic microdosing has garnered considerable mass media attention as an increasingly popular practice among younger professionals in the fields of computer science, engineering, mathematics, and technology. But it hasn’t been subjected to much scientific scrutiny.
Dr. Kuypers and colleagues wanted to find out whether it is actually effective and whether there are negative effects. They conducted an online questionnaire survey during a 5-month period of 2018 that drew 1,116 respondents, 80% of whom were currently microdosing, while the other 20% were former microdosers who had stopped completely.
The most common motivation for microdosing was indeed to stimulate productivity through increased focus, energy, and creativity. Almost half of microdosers indicated that they designed their own dosing schedule – typically once every 2-4 days – and two-thirds of microdosers were oblivious as to the consumed dose.
The most common reasons that respondents stopped microdosing were negative experiences or loss of interest because of lack of efficacy. The negative experiences typically involved acute anxiety or other psychological symptoms limited to when they were under the influence (Int J Neuropsychopharmacol. 2019 Jul 1;22[7]:426-34).
A subset of survey respondents microdosed to alleviate symptoms of a physician-diagnosed mental or physical health disorder. They were the focus of a separate analysis.
Microdosing for mental, physical health problems
Four hundred and ten survey respondents reported microdosing to self-treat a total of 901 physician-diagnosed mental and 161 physiologic conditions. They were asked three efficacy questions: Did it work? Did symptoms disappear? Did your quality of life improve?
The responses were disorder-specific. Individuals with anxiety disorders, ADHD, migraine, and other pain syndromes were the only ones to consistently rate microdosing as more effective than conventional treatment. Microdosing was rated less effective for symptom relief than were full-dose psychedelics only in respondents with depression or anxiety; individuals with other mental disorders or with physiological disorders rated the two dosing strategies similarly (Front Psychiatry. 2019 Sep 13. doi: 10.3389/fpsyt.2019.00672).
Future randomized controlled trials are warranted to more accurately assess efficacy claims for various psychedelics, optimal dosing, disorder specificity, and how they stack up compared with standard therapies, she said.
Dr. Kuypers reported having no financial conflicts of interest regarding her studies, which were conducted free of commercial support.
COPENHAGEN – A single dose of psilocybin taken in a supportive social setting was associated with demonstrably enhanced empathy, creative thinking, and subjective well-being lasting for up to 7 days in an observational study, Kim P.C. Kuypers, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
The participants in this uncontrolled study were healthy individuals curious to experience psilocybin in a controlled environment, but the observed benefits are of special interest because of psilocybin’s potential role in the treatment of psychiatric disorders, including depression and PTSD, in which preliminary study results have been quite encouraging, observed Dr. Kuypers, a neuropsychologist at Maastricht (the Netherlands) University.
For example, hallmarks of depression include psychological inflexibility, negative thoughts, and disturbed empathy. As this study showed, psilocybin addresses all those issues, she said.
according to Dr. Kuypers.
She and her coinvestigators studied 55 participants at a psilocybin retreat. These are events sprouting up across Europe to accommodate people who are curious about psilocybin and interested in trying it in a supportive setting. The investigators administered structured tests of convergent and divergent thinking, emotional empathy, and satisfaction with life before participants ingested the so-called magic mushroom, the morning after, and again 7 days later. In the Netherlands, the above-ground portion of the plant is an illegal substance, but the underground stem is not. The average dose was 27 mg, slightly higher than is often used in controlled laboratory studies; however, there were no adverse events.
The results showed that divergent thinking was enhanced the morning after taking psilocybin, an effect that was not sustained at the 7-day mark. In contrast, convergent thinking, emotional empathy, and a satisfaction-with-life scores above baseline were maintained 7 days after ingestion.
Psilocybin is a 5-HT2A agonist. It is considered a classic psychedelic, as are LSD, mescaline, and ayahuasca. These agents are often used recreationally to broaden consciousness, for relaxation, and to amplify emotions. Cave paintings indicate that psilocybin has been used medicinally for 11,000 years. But even early hunter-gatherers could not possibly have envisioned the current phenomenon of psilocybin microdosing to enhance work performance as adopted by hard-driving Silicon Valley professionals and similarly motivated strivers elsewhere around the world.
Microdosing for enhanced performance
Microdosing of psychedelics, especially psilocybin and LSD, is the practice of taking a low dose – typically 1/10th of a full recreational dose, which is too small an amount to cause full-blown perceptual alterations – once every several days in order to stimulate productivity. Psychedelic microdosing has garnered considerable mass media attention as an increasingly popular practice among younger professionals in the fields of computer science, engineering, mathematics, and technology. But it hasn’t been subjected to much scientific scrutiny.
Dr. Kuypers and colleagues wanted to find out whether it is actually effective and whether there are negative effects. They conducted an online questionnaire survey during a 5-month period of 2018 that drew 1,116 respondents, 80% of whom were currently microdosing, while the other 20% were former microdosers who had stopped completely.
The most common motivation for microdosing was indeed to stimulate productivity through increased focus, energy, and creativity. Almost half of microdosers indicated that they designed their own dosing schedule – typically once every 2-4 days – and two-thirds of microdosers were oblivious as to the consumed dose.
The most common reasons that respondents stopped microdosing were negative experiences or loss of interest because of lack of efficacy. The negative experiences typically involved acute anxiety or other psychological symptoms limited to when they were under the influence (Int J Neuropsychopharmacol. 2019 Jul 1;22[7]:426-34).
A subset of survey respondents microdosed to alleviate symptoms of a physician-diagnosed mental or physical health disorder. They were the focus of a separate analysis.
Microdosing for mental, physical health problems
Four hundred and ten survey respondents reported microdosing to self-treat a total of 901 physician-diagnosed mental and 161 physiologic conditions. They were asked three efficacy questions: Did it work? Did symptoms disappear? Did your quality of life improve?
The responses were disorder-specific. Individuals with anxiety disorders, ADHD, migraine, and other pain syndromes were the only ones to consistently rate microdosing as more effective than conventional treatment. Microdosing was rated less effective for symptom relief than were full-dose psychedelics only in respondents with depression or anxiety; individuals with other mental disorders or with physiological disorders rated the two dosing strategies similarly (Front Psychiatry. 2019 Sep 13. doi: 10.3389/fpsyt.2019.00672).
Future randomized controlled trials are warranted to more accurately assess efficacy claims for various psychedelics, optimal dosing, disorder specificity, and how they stack up compared with standard therapies, she said.
Dr. Kuypers reported having no financial conflicts of interest regarding her studies, which were conducted free of commercial support.
EXPERT ANALYSIS FROM ECNP 2019
Study spotlights severe NAFLD in lean patients
BOSTON – For patients with nonalcoholic fatty liver disease (NAFLD), leaner may really be meaner, according to a recent French study involving more than 100,000 individuals of the general population.
NAFLD was uncommon among participants with normal bodyweight, but when NAFLD was present, lean patients had almost twice the risk of advanced fibrosis as that of overweight and obese patients, reported principal author Lawrence Serfaty, MD, chief of the department of hepatology at Strasbourg (France) University, who conducted the project with colleagues at the French public research organization, Inserm.
“Normally, NAFLD and [nonalcoholic steatohepatitis (NASH)] are part of metabolic syndrome,” Dr. Serfaty said in an interview at the annual meeting of the American Association for the Study of Liver Diseases. “But there are some patients with no metabolic risk factors who are lean and who may have NAFLD.”
To determine the prevalence and characteristics of these patients, the investigators drew data from the CONSTANCES cohort, which is composed of 118,664 members of the general public in France. After excluding those who withdrew consent, had a history of other liver diseases, or reported an excess of alcohol consumption (more than 20 g/day), the analyzed dataset included 102,344 subjects. Among these participants, NAFLD and advanced fibrosis were diagnosed with the Fatty Liver Index (FLI) and the Forns Index (FI), respectively, in which an FLI score greater than 60 indicated NAFLD and an FI score greater than 6.9 indicated advanced fibrosis.
Participants were sorted into three weight categories by body mass index: lean (BMI less than 25 kg/m2), overweight (BMI 25-30), or obese (BMI more than 30). In addition, a variety of other health measures were recorded, including presence of metabolic risk factors, such as diabetes or metabolic syndrome, and elevated alanine transaminase (ALT).
Analysis showed that the prevalence of NAFLD in the general population was 18.2%. Of those diagnosed with NAFLD, 2.6% had advanced fibrosis. As expected, Dr. Serfaty said, NAFLD was much less common among those who were of normal bodyweight, with lean patients accounting for only 1.9% of NAFLD diagnoses. However, when NAFLD was diagnosed in lean patients, it was more often severe. Among lean patients with NAFLD, 4.5% had advanced fibrosis, compared with 2.4% of overweight patients and 2.3% of obese patients.
Dr. Serfaty noted that lean patients with NAFLD tended to have fewer metabolic risk factors, but most had at least one. Lifestyle factors were likely to blame, he said, because lean patients with NAFLD were relatively heavy users of tobacco and alcohol, compared with obese or overweight patients. Cardiovascular disease was also more common among lean patients with NAFLD.
“There are probably other factors [that are different, such as] genetic factors, the microbiome, and maybe the immune system,” Dr. Serfaty said.
While drivers of NAFLD among lean patients remain to be clarified, Dr. Serfaty highlighted the importance of recognizing this unique patient population, and if encountered, not discounting the severity of disease based on a lack of other metabolic risk factors.
“It is very important to know that these patients exist because it is very difficult to identify these patients,” Dr. Serfaty said. “If you have a lean patient with NAFLD, normally you [might] say, it’s not very severe for this patient because he’s lean; but no, [that’s not correct], because maybe he has more advanced fibrosis. So you have to go through and check with other noninvasive markers to be sure that this patient does not have advanced fibrosis.”
Concerning the difficulty of identifying such patients in the first place, Dr. Serfaty suggested that elevated ALT may be the most reliable red flag because this laboratory abnormality occurred in more than half of the lean patients diagnosed with NAFLD, despite no excessive alcohol consumption or prior hepatitis. The investigators disclosed relationships with Gilead, AbbVie, Echosens, and others.
SOURCE: Serfaty L et al. The Liver Meeting 2019, Abstract 1188.
BOSTON – For patients with nonalcoholic fatty liver disease (NAFLD), leaner may really be meaner, according to a recent French study involving more than 100,000 individuals of the general population.
NAFLD was uncommon among participants with normal bodyweight, but when NAFLD was present, lean patients had almost twice the risk of advanced fibrosis as that of overweight and obese patients, reported principal author Lawrence Serfaty, MD, chief of the department of hepatology at Strasbourg (France) University, who conducted the project with colleagues at the French public research organization, Inserm.
“Normally, NAFLD and [nonalcoholic steatohepatitis (NASH)] are part of metabolic syndrome,” Dr. Serfaty said in an interview at the annual meeting of the American Association for the Study of Liver Diseases. “But there are some patients with no metabolic risk factors who are lean and who may have NAFLD.”
To determine the prevalence and characteristics of these patients, the investigators drew data from the CONSTANCES cohort, which is composed of 118,664 members of the general public in France. After excluding those who withdrew consent, had a history of other liver diseases, or reported an excess of alcohol consumption (more than 20 g/day), the analyzed dataset included 102,344 subjects. Among these participants, NAFLD and advanced fibrosis were diagnosed with the Fatty Liver Index (FLI) and the Forns Index (FI), respectively, in which an FLI score greater than 60 indicated NAFLD and an FI score greater than 6.9 indicated advanced fibrosis.
Participants were sorted into three weight categories by body mass index: lean (BMI less than 25 kg/m2), overweight (BMI 25-30), or obese (BMI more than 30). In addition, a variety of other health measures were recorded, including presence of metabolic risk factors, such as diabetes or metabolic syndrome, and elevated alanine transaminase (ALT).
Analysis showed that the prevalence of NAFLD in the general population was 18.2%. Of those diagnosed with NAFLD, 2.6% had advanced fibrosis. As expected, Dr. Serfaty said, NAFLD was much less common among those who were of normal bodyweight, with lean patients accounting for only 1.9% of NAFLD diagnoses. However, when NAFLD was diagnosed in lean patients, it was more often severe. Among lean patients with NAFLD, 4.5% had advanced fibrosis, compared with 2.4% of overweight patients and 2.3% of obese patients.
Dr. Serfaty noted that lean patients with NAFLD tended to have fewer metabolic risk factors, but most had at least one. Lifestyle factors were likely to blame, he said, because lean patients with NAFLD were relatively heavy users of tobacco and alcohol, compared with obese or overweight patients. Cardiovascular disease was also more common among lean patients with NAFLD.
“There are probably other factors [that are different, such as] genetic factors, the microbiome, and maybe the immune system,” Dr. Serfaty said.
While drivers of NAFLD among lean patients remain to be clarified, Dr. Serfaty highlighted the importance of recognizing this unique patient population, and if encountered, not discounting the severity of disease based on a lack of other metabolic risk factors.
“It is very important to know that these patients exist because it is very difficult to identify these patients,” Dr. Serfaty said. “If you have a lean patient with NAFLD, normally you [might] say, it’s not very severe for this patient because he’s lean; but no, [that’s not correct], because maybe he has more advanced fibrosis. So you have to go through and check with other noninvasive markers to be sure that this patient does not have advanced fibrosis.”
Concerning the difficulty of identifying such patients in the first place, Dr. Serfaty suggested that elevated ALT may be the most reliable red flag because this laboratory abnormality occurred in more than half of the lean patients diagnosed with NAFLD, despite no excessive alcohol consumption or prior hepatitis. The investigators disclosed relationships with Gilead, AbbVie, Echosens, and others.
SOURCE: Serfaty L et al. The Liver Meeting 2019, Abstract 1188.
BOSTON – For patients with nonalcoholic fatty liver disease (NAFLD), leaner may really be meaner, according to a recent French study involving more than 100,000 individuals of the general population.
NAFLD was uncommon among participants with normal bodyweight, but when NAFLD was present, lean patients had almost twice the risk of advanced fibrosis as that of overweight and obese patients, reported principal author Lawrence Serfaty, MD, chief of the department of hepatology at Strasbourg (France) University, who conducted the project with colleagues at the French public research organization, Inserm.
“Normally, NAFLD and [nonalcoholic steatohepatitis (NASH)] are part of metabolic syndrome,” Dr. Serfaty said in an interview at the annual meeting of the American Association for the Study of Liver Diseases. “But there are some patients with no metabolic risk factors who are lean and who may have NAFLD.”
To determine the prevalence and characteristics of these patients, the investigators drew data from the CONSTANCES cohort, which is composed of 118,664 members of the general public in France. After excluding those who withdrew consent, had a history of other liver diseases, or reported an excess of alcohol consumption (more than 20 g/day), the analyzed dataset included 102,344 subjects. Among these participants, NAFLD and advanced fibrosis were diagnosed with the Fatty Liver Index (FLI) and the Forns Index (FI), respectively, in which an FLI score greater than 60 indicated NAFLD and an FI score greater than 6.9 indicated advanced fibrosis.
Participants were sorted into three weight categories by body mass index: lean (BMI less than 25 kg/m2), overweight (BMI 25-30), or obese (BMI more than 30). In addition, a variety of other health measures were recorded, including presence of metabolic risk factors, such as diabetes or metabolic syndrome, and elevated alanine transaminase (ALT).
Analysis showed that the prevalence of NAFLD in the general population was 18.2%. Of those diagnosed with NAFLD, 2.6% had advanced fibrosis. As expected, Dr. Serfaty said, NAFLD was much less common among those who were of normal bodyweight, with lean patients accounting for only 1.9% of NAFLD diagnoses. However, when NAFLD was diagnosed in lean patients, it was more often severe. Among lean patients with NAFLD, 4.5% had advanced fibrosis, compared with 2.4% of overweight patients and 2.3% of obese patients.
Dr. Serfaty noted that lean patients with NAFLD tended to have fewer metabolic risk factors, but most had at least one. Lifestyle factors were likely to blame, he said, because lean patients with NAFLD were relatively heavy users of tobacco and alcohol, compared with obese or overweight patients. Cardiovascular disease was also more common among lean patients with NAFLD.
“There are probably other factors [that are different, such as] genetic factors, the microbiome, and maybe the immune system,” Dr. Serfaty said.
While drivers of NAFLD among lean patients remain to be clarified, Dr. Serfaty highlighted the importance of recognizing this unique patient population, and if encountered, not discounting the severity of disease based on a lack of other metabolic risk factors.
“It is very important to know that these patients exist because it is very difficult to identify these patients,” Dr. Serfaty said. “If you have a lean patient with NAFLD, normally you [might] say, it’s not very severe for this patient because he’s lean; but no, [that’s not correct], because maybe he has more advanced fibrosis. So you have to go through and check with other noninvasive markers to be sure that this patient does not have advanced fibrosis.”
Concerning the difficulty of identifying such patients in the first place, Dr. Serfaty suggested that elevated ALT may be the most reliable red flag because this laboratory abnormality occurred in more than half of the lean patients diagnosed with NAFLD, despite no excessive alcohol consumption or prior hepatitis. The investigators disclosed relationships with Gilead, AbbVie, Echosens, and others.
SOURCE: Serfaty L et al. The Liver Meeting 2019, Abstract 1188.
REPORTING FROM THE LIVER MEETING 2019
Lung cancer screening, early diagnosis still lower among blacks, Hispanics
SAN FRANCISCO – A review of lung cancer cases in New York suggests that 2011 guidelines issued by the National Comprehensive Cancer Network (NCCN) have made little difference in addressing ethnic and socioeconomic disparities in lung cancer diagnosis. The review showed some progress; however, . The researchers also found a reduced incidence of lung cancer only in whites.
The guidelines call for CT screening for smokers aged 55-74 years that have a 30 pack-year history within the past 15 years and were based on a lung cancer screening trial showing that annual low-dose CT led to a 20% reduction in lung cancer deaths, compared with chest x-ray (N Eng J Med. 2011;365:395-409).
Nonwhites “aren’t always exposed to screening opportunities, and may be taken care of by providers who haven’t necessarily bought into screening or aren’t necessarily aware of it. So the downstream effects of the lack of access to screening hurts them doubly, because they’re diagnosed later and they also have disparities in access to treatment,” said Elizabeth David, MD, in an interview. Dr. David is an associate professor of surgery of University of Southern California, Los Angeles. She comoderated a session at the annual congress of the American College of Surgeons, where the research was presented.
The findings underscore the need to get the word out to primary care providers in all communities that catching lung cancer early can make all the difference to patients. “The more we can do to talk about lung cancer as a treatable disease and a cancer that can become a chronically managed medical problem for patients, rather than a death sentence, which it has the misperception of being, the more we can encourage people to seek treatment when they have a symptom, instead of being afraid and doing nothing,” said Dr. David.
The researchers considered metropolitan New York to be a good test case to examine the impact of the screening guidelines since it is socioeconomically and racially diverse. “New York presents a microcosm of what’s going on nationally in other urban areas,” said Tamar Nobel, MD, from Mount Sinai Hospital, New York, who presented the study.
The researchers analyzed records from the New York State Cancer Registry from the period 2008-201 and compared that with data from 2012-2015 (total, n = 18,284). The incidence of lung cancer declined among whites from the first period to the second, at an average annual percent change of –1.5% (95% confidence interval, –1.0% to –2.0%). A similar result was seen in Hispanics (–1.7%; 95% CI, –0.4% to –2.9%), but not among blacks (–1.2%; not significant). There was an increase among Asians/Pacific Islanders (+1.4%; 95% CI, 0.6% to 2.2%).
There were also inequalities when it came to the odds of a stage I diagnosis. Although the percentages increased in whites (+3.5%; 95% CI, 0.6%-6.4%) and blacks (+6.0%; 95% CI, 2.3%-9.7%), the median percentage was higher among whites (27.6% versus 18.2%; no overlap among 95% CIs; odds ratio, 0.62; 95% CI, 0.56-0.68). Hispanics and Asians/Pacific Islanders experienced no significant changes in their odds of a stage I diagnosis, and both groups remained significantly less likely than whites to be diagnosed with stage I disease (OR, 0.72 and 0.86, respectively, both significant).
No funding was disclosed. Dr. David and Dr. Nobel have no relevant financial disclosures.
SOURCE: Nobel T et al. ACS Clinical Congress 2019. Abstract, doi: 10.1016/j.jamcollsurg.2019.08.607.
SAN FRANCISCO – A review of lung cancer cases in New York suggests that 2011 guidelines issued by the National Comprehensive Cancer Network (NCCN) have made little difference in addressing ethnic and socioeconomic disparities in lung cancer diagnosis. The review showed some progress; however, . The researchers also found a reduced incidence of lung cancer only in whites.
The guidelines call for CT screening for smokers aged 55-74 years that have a 30 pack-year history within the past 15 years and were based on a lung cancer screening trial showing that annual low-dose CT led to a 20% reduction in lung cancer deaths, compared with chest x-ray (N Eng J Med. 2011;365:395-409).
Nonwhites “aren’t always exposed to screening opportunities, and may be taken care of by providers who haven’t necessarily bought into screening or aren’t necessarily aware of it. So the downstream effects of the lack of access to screening hurts them doubly, because they’re diagnosed later and they also have disparities in access to treatment,” said Elizabeth David, MD, in an interview. Dr. David is an associate professor of surgery of University of Southern California, Los Angeles. She comoderated a session at the annual congress of the American College of Surgeons, where the research was presented.
The findings underscore the need to get the word out to primary care providers in all communities that catching lung cancer early can make all the difference to patients. “The more we can do to talk about lung cancer as a treatable disease and a cancer that can become a chronically managed medical problem for patients, rather than a death sentence, which it has the misperception of being, the more we can encourage people to seek treatment when they have a symptom, instead of being afraid and doing nothing,” said Dr. David.
The researchers considered metropolitan New York to be a good test case to examine the impact of the screening guidelines since it is socioeconomically and racially diverse. “New York presents a microcosm of what’s going on nationally in other urban areas,” said Tamar Nobel, MD, from Mount Sinai Hospital, New York, who presented the study.
The researchers analyzed records from the New York State Cancer Registry from the period 2008-201 and compared that with data from 2012-2015 (total, n = 18,284). The incidence of lung cancer declined among whites from the first period to the second, at an average annual percent change of –1.5% (95% confidence interval, –1.0% to –2.0%). A similar result was seen in Hispanics (–1.7%; 95% CI, –0.4% to –2.9%), but not among blacks (–1.2%; not significant). There was an increase among Asians/Pacific Islanders (+1.4%; 95% CI, 0.6% to 2.2%).
There were also inequalities when it came to the odds of a stage I diagnosis. Although the percentages increased in whites (+3.5%; 95% CI, 0.6%-6.4%) and blacks (+6.0%; 95% CI, 2.3%-9.7%), the median percentage was higher among whites (27.6% versus 18.2%; no overlap among 95% CIs; odds ratio, 0.62; 95% CI, 0.56-0.68). Hispanics and Asians/Pacific Islanders experienced no significant changes in their odds of a stage I diagnosis, and both groups remained significantly less likely than whites to be diagnosed with stage I disease (OR, 0.72 and 0.86, respectively, both significant).
No funding was disclosed. Dr. David and Dr. Nobel have no relevant financial disclosures.
SOURCE: Nobel T et al. ACS Clinical Congress 2019. Abstract, doi: 10.1016/j.jamcollsurg.2019.08.607.
SAN FRANCISCO – A review of lung cancer cases in New York suggests that 2011 guidelines issued by the National Comprehensive Cancer Network (NCCN) have made little difference in addressing ethnic and socioeconomic disparities in lung cancer diagnosis. The review showed some progress; however, . The researchers also found a reduced incidence of lung cancer only in whites.
The guidelines call for CT screening for smokers aged 55-74 years that have a 30 pack-year history within the past 15 years and were based on a lung cancer screening trial showing that annual low-dose CT led to a 20% reduction in lung cancer deaths, compared with chest x-ray (N Eng J Med. 2011;365:395-409).
Nonwhites “aren’t always exposed to screening opportunities, and may be taken care of by providers who haven’t necessarily bought into screening or aren’t necessarily aware of it. So the downstream effects of the lack of access to screening hurts them doubly, because they’re diagnosed later and they also have disparities in access to treatment,” said Elizabeth David, MD, in an interview. Dr. David is an associate professor of surgery of University of Southern California, Los Angeles. She comoderated a session at the annual congress of the American College of Surgeons, where the research was presented.
The findings underscore the need to get the word out to primary care providers in all communities that catching lung cancer early can make all the difference to patients. “The more we can do to talk about lung cancer as a treatable disease and a cancer that can become a chronically managed medical problem for patients, rather than a death sentence, which it has the misperception of being, the more we can encourage people to seek treatment when they have a symptom, instead of being afraid and doing nothing,” said Dr. David.
The researchers considered metropolitan New York to be a good test case to examine the impact of the screening guidelines since it is socioeconomically and racially diverse. “New York presents a microcosm of what’s going on nationally in other urban areas,” said Tamar Nobel, MD, from Mount Sinai Hospital, New York, who presented the study.
The researchers analyzed records from the New York State Cancer Registry from the period 2008-201 and compared that with data from 2012-2015 (total, n = 18,284). The incidence of lung cancer declined among whites from the first period to the second, at an average annual percent change of –1.5% (95% confidence interval, –1.0% to –2.0%). A similar result was seen in Hispanics (–1.7%; 95% CI, –0.4% to –2.9%), but not among blacks (–1.2%; not significant). There was an increase among Asians/Pacific Islanders (+1.4%; 95% CI, 0.6% to 2.2%).
There were also inequalities when it came to the odds of a stage I diagnosis. Although the percentages increased in whites (+3.5%; 95% CI, 0.6%-6.4%) and blacks (+6.0%; 95% CI, 2.3%-9.7%), the median percentage was higher among whites (27.6% versus 18.2%; no overlap among 95% CIs; odds ratio, 0.62; 95% CI, 0.56-0.68). Hispanics and Asians/Pacific Islanders experienced no significant changes in their odds of a stage I diagnosis, and both groups remained significantly less likely than whites to be diagnosed with stage I disease (OR, 0.72 and 0.86, respectively, both significant).
No funding was disclosed. Dr. David and Dr. Nobel have no relevant financial disclosures.
SOURCE: Nobel T et al. ACS Clinical Congress 2019. Abstract, doi: 10.1016/j.jamcollsurg.2019.08.607.
REPORTING FROM CLINICAL CONGRESS 2019
Orthopedic ambulatory surgery centers beat inpatient services on cost
NATIONAL HARBOR, MD. – Hospital outpatient departments (HOPDs) and (IPs) with similar levels of postoperative opioid use, according to a new study.
Fanta Waterman, PhD, director of medical and health sciences at Pacira Pharmaceuticals, and colleagues retrospectively published the results of their investigation in the Journal of Managed Care & Specialty Pharmacy supplement for the annual meeting of the Academy of Managed Care Pharmacy.
Investigators evaluated data from 126,172 commercially insured patients who underwent one of six orthopedic surgical procedures between April 2012 and December 2017. Using the Optum Research Database, they pooled data from patients who had received total knee arthroplasty (TKA), partial knee arthroplasty, total hip arthroplasty (THA), rotator cuff repair (RCR), total shoulder arthroplasty, and lumbar spine fusion.
More than half (51%) of the patients were male, and the patients averaged 58 years of age. Most patients who underwent any of the six surgical interventions had the procedures performed at IPs (68%), while 18% had their operations at HOPDs and 14% were perfomed at ASCs.
TKA, RCR, and THA were the most common procedures performed (32%, 27%, and 20%, respectively). While no fluctuation was observed in the total number of IP procedures performed during 2012-2017, researchers noted a marked increase in ASCs (58%) and HOPDs (15%).
At the 30-day mark, the total all-cause postsurgical costs associated with IPs ($44,566) were more than double that of HOPDs ($20,468) and ASCs ($19,110; P less than .001). Moreover, multivariate adjustment showed that postsurgical costs accrued 30 days after surgery for HOPDs and ASCs were 14% and 27% lower than IPs (P less than .001), respectively.
Additionally, each group exhibited similar evidence of opioid use in the 12-month period prior to undergoing surgery, ranging from 63% to 65%. Postsurgical opioid use among opioid-naive patients was the highest in the HOPD group at 96% prevalence, with IPs and ASCs trailing with 91% and 90% (P less than .001), respectively. However, the postsurgical prevalence of opioid use in patients who had used opioids before surgery was 95% for IPs and HOPDs and 82% for ASCs (P less than .001).
SOURCE: Waterman F et al. AMCP NEXUS 2019, Abstract U12.
NATIONAL HARBOR, MD. – Hospital outpatient departments (HOPDs) and (IPs) with similar levels of postoperative opioid use, according to a new study.
Fanta Waterman, PhD, director of medical and health sciences at Pacira Pharmaceuticals, and colleagues retrospectively published the results of their investigation in the Journal of Managed Care & Specialty Pharmacy supplement for the annual meeting of the Academy of Managed Care Pharmacy.
Investigators evaluated data from 126,172 commercially insured patients who underwent one of six orthopedic surgical procedures between April 2012 and December 2017. Using the Optum Research Database, they pooled data from patients who had received total knee arthroplasty (TKA), partial knee arthroplasty, total hip arthroplasty (THA), rotator cuff repair (RCR), total shoulder arthroplasty, and lumbar spine fusion.
More than half (51%) of the patients were male, and the patients averaged 58 years of age. Most patients who underwent any of the six surgical interventions had the procedures performed at IPs (68%), while 18% had their operations at HOPDs and 14% were perfomed at ASCs.
TKA, RCR, and THA were the most common procedures performed (32%, 27%, and 20%, respectively). While no fluctuation was observed in the total number of IP procedures performed during 2012-2017, researchers noted a marked increase in ASCs (58%) and HOPDs (15%).
At the 30-day mark, the total all-cause postsurgical costs associated with IPs ($44,566) were more than double that of HOPDs ($20,468) and ASCs ($19,110; P less than .001). Moreover, multivariate adjustment showed that postsurgical costs accrued 30 days after surgery for HOPDs and ASCs were 14% and 27% lower than IPs (P less than .001), respectively.
Additionally, each group exhibited similar evidence of opioid use in the 12-month period prior to undergoing surgery, ranging from 63% to 65%. Postsurgical opioid use among opioid-naive patients was the highest in the HOPD group at 96% prevalence, with IPs and ASCs trailing with 91% and 90% (P less than .001), respectively. However, the postsurgical prevalence of opioid use in patients who had used opioids before surgery was 95% for IPs and HOPDs and 82% for ASCs (P less than .001).
SOURCE: Waterman F et al. AMCP NEXUS 2019, Abstract U12.
NATIONAL HARBOR, MD. – Hospital outpatient departments (HOPDs) and (IPs) with similar levels of postoperative opioid use, according to a new study.
Fanta Waterman, PhD, director of medical and health sciences at Pacira Pharmaceuticals, and colleagues retrospectively published the results of their investigation in the Journal of Managed Care & Specialty Pharmacy supplement for the annual meeting of the Academy of Managed Care Pharmacy.
Investigators evaluated data from 126,172 commercially insured patients who underwent one of six orthopedic surgical procedures between April 2012 and December 2017. Using the Optum Research Database, they pooled data from patients who had received total knee arthroplasty (TKA), partial knee arthroplasty, total hip arthroplasty (THA), rotator cuff repair (RCR), total shoulder arthroplasty, and lumbar spine fusion.
More than half (51%) of the patients were male, and the patients averaged 58 years of age. Most patients who underwent any of the six surgical interventions had the procedures performed at IPs (68%), while 18% had their operations at HOPDs and 14% were perfomed at ASCs.
TKA, RCR, and THA were the most common procedures performed (32%, 27%, and 20%, respectively). While no fluctuation was observed in the total number of IP procedures performed during 2012-2017, researchers noted a marked increase in ASCs (58%) and HOPDs (15%).
At the 30-day mark, the total all-cause postsurgical costs associated with IPs ($44,566) were more than double that of HOPDs ($20,468) and ASCs ($19,110; P less than .001). Moreover, multivariate adjustment showed that postsurgical costs accrued 30 days after surgery for HOPDs and ASCs were 14% and 27% lower than IPs (P less than .001), respectively.
Additionally, each group exhibited similar evidence of opioid use in the 12-month period prior to undergoing surgery, ranging from 63% to 65%. Postsurgical opioid use among opioid-naive patients was the highest in the HOPD group at 96% prevalence, with IPs and ASCs trailing with 91% and 90% (P less than .001), respectively. However, the postsurgical prevalence of opioid use in patients who had used opioids before surgery was 95% for IPs and HOPDs and 82% for ASCs (P less than .001).
SOURCE: Waterman F et al. AMCP NEXUS 2019, Abstract U12.
REPORTING FROM AMCP NEXUS 2019
Adding mental health clinicians to your practice is full of benefits
NEW ORLEANS – The way Jay Rabinowitz, MD, MPH, sees it, providing mental and behavioral health care services in your primary care pediatrics practice is a win-win for patients, parents, and clinicians.
For one thing, children with mental and behavioral issues – especially depression and anxiety – make up a good chunk of any pediatrician’s workday. Dr. Rabinowitz, clinical professor of pediatrics at the University of Colorado, Aurora, said at the annual meeting of the American Academy of Pediatrics. “It is the most costly issue in children’s health care today.”
According to “Behavioral Health Integration in Pediatric Primary Care,” a report supported by the Milbank Memorial Fund, one in five children aged 9-17 years have a diagnosable psychiatric disorder, and up to 70% of children in the juvenile justice system have a mental health disorder. The report also found that the treatment of mental health disorders accounts for the most costly childhood medical expenditure, and that between 15% and 20% of children with psychiatric disorders receive specialty care; the rest see their primary care provider. A long-term cost analysis showed significant cost savings: $1 spent on collaborative care saves $6.50 on health care costs.
More recently, the Guidelines for Adolescent Depression in Primary Care (GLAD-PC) found that only 50% of adolescents with depression are diagnosed before reaching adulthood (Pediatrics. March 2018;141[3]:e20174081). As many as two out of three youth with depression are not identified by their pediatrician and do not receive any kind of care.
“Even when diagnosed, only half of these patients are treated appropriately,” said Dr. Rabinowitz, who also practices at Parker (Colo.) Pediatrics and Adolescents.
The guidelines also found that reliance on self-report depression checklists alone lead to substantial numbers of false-positive and false-negative cases. “Primary care providers will benefit from having access to ongoing consultation with mental health providers,” according to the guidelines.
“Integrative care was associated with significant decreases in depression scores, and improved response and remission rates at 12 months, compared with treatment as usual,” Dr. Rabinowitz said.
Providing mental health services in a primary care pediatrics setting also makes sense because there’s a shortage of psychiatrists and psychologists to see them, and it enables patients to get evaluated quicker. “It’s convenient, and it reduces stigma,” he added. “It’s a familiar setting, a familiar provider, and they’re more likely to initiate counseling. Nationwide, 50% of patients who are referred for mental health do not make their initial appointment. Think about that. If you had diabetics in your practice and only 50% would go to the endocrinologist, what would you think?”
How Dr. Rabinowitz and his partners got started
Dr. Rabinowitz and his colleagues created an integrated care model in 2008 by adding a psychologist to their practice, but before doing that, they asked parents of children with mental and behavioral health issues what type of insurance they had. Then they obtained a referral list from the family’s insurer and hoped for the best. “Sometimes I referred to someone I may not have heard of,” Dr. Rabinowitz said. “Usually I did not get follow-up reports, or even know for sure if the patient ever went.”
Today, Parker Pediatrics and Adolescents employs three doctoral-level psychologists: one full-time, one three-quarter time, and one half-time, as well as one master’s-level therapist who works half-time.
“On any given day, we have at least two counselors in our office,” said Lindsey Einhorn, PhD, a licensed clinical psychologist who joined the practice in 2011. She and her colleagues care for children and teens with ADHD, depression, anxiety, behavioral and adjustment disorders, drug counseling, behavioral addictions, social struggles such as bullying, obsessive compulsive disorder (OCD), loss, hair or eyelash pulling, mood dysregulation, and sibling conflict. They refer for educational testing, comprehensive psychological evaluations, difficult divorce cases, play therapy, complex cases requiring more than 20 sessions, and children of staff employed by the practice.
The practice features a separate waiting room for psychology patients and front office staff dedicated to managing their schedules. “For anyone who’s trying to make a psychology appointment but can’t be seen in an efficient manner or wants a different day or time, we keep an ongoing move-up list,” Dr. Einhorn said. “If a family calls to cancel an appointment, the front desk person who makes that cancellation will fill out a slip and give it to one of our psychology schedulers. That person will create a move-up list and start filling that appointment. If there’s a cancellation, it’s rare that it goes unfilled.”
Key forms for parents to complete include informed consent, a notice of privacy practices, a late cancel/no show policy, an initial intake agreement, and a summary of parent concerns.
Patient and clinician reaction
According to results from a recent survey of parents whose children were seen by a psychologist at Parker Pediatrics and Adolescents, 89% said it was important for their children to receive mental health services in the same location as their medical care, and 96% were satisfied with the services provided. In addition, 93% said that the experience benefited their child, 72% were satisfied with appointment times, and 55% expressed interest in virtual visits via telemedicine. Meanwhile, a survey of parents whose children have not been seen by a psychologist at the practice found that 65% knew a psychologist was on staff, and only 9% said that there were barriers to their child seeing a psychologist there.
Clinicians themselves benefit from having mental health specialists on site for referrals. “It enables you to be more efficient, and it saves time,” Dr. Rabinowitz said. “There’s knowledge and confidence gained, and it improves satisfaction because physicians don’t have to stay at the office later filling out referral forms. It meets the needs of your patients and their families, it attracts new patients, and you may be able to make some income on this.”
How to get started
Dr. Rabinowitz recommended that, once clinicians at a pediatric practice commit to expanding their services to include mental and behavioral health care, they should hold a corporate/partner meeting, assign responsibilities, and establish a timeline for implementation. “This is all very important,” he said. “Then you have to talk about what kind of arrangement you want to have. You could employ someone to join your practice, hire an independent contractor, establish a space share agreement, or have an out-of-office arrangement.”
For many years, clinicians at Parker Pediatrics and Adolescents had a psychologist perform ADHD evaluations on a consultative basis. “Then, as we saw a need for mental health services about a decade ago, we hired a part-time psychologist who did testing as well as counseling,” Dr. Rabinowitz said. “But that psychologist got very busy, so we hired a full-time psychologist. We continued to hire additional psychologists as need increased.”
Reimbursement issues
Numerous reimbursement barriers to providing mental health services in pediatric primary care exist, he noted, including a lack of payment if mental health codes are used, a lack of “incident to” payments in some areas of the country, existing reimbursement levels, and the fact that same-day billing of physical and mental health often is not allowed. “However, we have found that if we give flu shots during their mental health visit, [insurers] will cover the flu shot,” he said. “Reimbursement for screening is sometimes not covered very well.”
One reimbursement option is the fee-for-service/concierge model, “but that’s not an economic option for many,” he said. “You can’t see Medicaid patients in that model.” Joining a mental health networks is feasible, “but there is poor reimbursement,” he said. “It also creates another layer of administration.”
He recommends financial integration, “but you need to research your options because a lot of it is state dependent.” Other options include grants, insurance contracts, and seeking permission from Medicaid.
Mental health CPT codes that mental health clinicians at the practice commonly use include bill by time (CPT code 99214/15); psychotherapy session that lasts 16-37 minutes (CPT code 90832); psychotherapy session that lasts 38-52 minutes (CPT code 90834); and psychotherapy session that lasts more than 53 minutes (CPT code 90837). Clinicians also can bill by interactive complexity (CPT code 90785) and psychotherapy for crisis (CPT code 90839).
Dr. Rabinowitz and Dr. Einhorn reported having no financial disclosures.
NEW ORLEANS – The way Jay Rabinowitz, MD, MPH, sees it, providing mental and behavioral health care services in your primary care pediatrics practice is a win-win for patients, parents, and clinicians.
For one thing, children with mental and behavioral issues – especially depression and anxiety – make up a good chunk of any pediatrician’s workday. Dr. Rabinowitz, clinical professor of pediatrics at the University of Colorado, Aurora, said at the annual meeting of the American Academy of Pediatrics. “It is the most costly issue in children’s health care today.”
According to “Behavioral Health Integration in Pediatric Primary Care,” a report supported by the Milbank Memorial Fund, one in five children aged 9-17 years have a diagnosable psychiatric disorder, and up to 70% of children in the juvenile justice system have a mental health disorder. The report also found that the treatment of mental health disorders accounts for the most costly childhood medical expenditure, and that between 15% and 20% of children with psychiatric disorders receive specialty care; the rest see their primary care provider. A long-term cost analysis showed significant cost savings: $1 spent on collaborative care saves $6.50 on health care costs.
More recently, the Guidelines for Adolescent Depression in Primary Care (GLAD-PC) found that only 50% of adolescents with depression are diagnosed before reaching adulthood (Pediatrics. March 2018;141[3]:e20174081). As many as two out of three youth with depression are not identified by their pediatrician and do not receive any kind of care.
“Even when diagnosed, only half of these patients are treated appropriately,” said Dr. Rabinowitz, who also practices at Parker (Colo.) Pediatrics and Adolescents.
The guidelines also found that reliance on self-report depression checklists alone lead to substantial numbers of false-positive and false-negative cases. “Primary care providers will benefit from having access to ongoing consultation with mental health providers,” according to the guidelines.
“Integrative care was associated with significant decreases in depression scores, and improved response and remission rates at 12 months, compared with treatment as usual,” Dr. Rabinowitz said.
Providing mental health services in a primary care pediatrics setting also makes sense because there’s a shortage of psychiatrists and psychologists to see them, and it enables patients to get evaluated quicker. “It’s convenient, and it reduces stigma,” he added. “It’s a familiar setting, a familiar provider, and they’re more likely to initiate counseling. Nationwide, 50% of patients who are referred for mental health do not make their initial appointment. Think about that. If you had diabetics in your practice and only 50% would go to the endocrinologist, what would you think?”
How Dr. Rabinowitz and his partners got started
Dr. Rabinowitz and his colleagues created an integrated care model in 2008 by adding a psychologist to their practice, but before doing that, they asked parents of children with mental and behavioral health issues what type of insurance they had. Then they obtained a referral list from the family’s insurer and hoped for the best. “Sometimes I referred to someone I may not have heard of,” Dr. Rabinowitz said. “Usually I did not get follow-up reports, or even know for sure if the patient ever went.”
Today, Parker Pediatrics and Adolescents employs three doctoral-level psychologists: one full-time, one three-quarter time, and one half-time, as well as one master’s-level therapist who works half-time.
“On any given day, we have at least two counselors in our office,” said Lindsey Einhorn, PhD, a licensed clinical psychologist who joined the practice in 2011. She and her colleagues care for children and teens with ADHD, depression, anxiety, behavioral and adjustment disorders, drug counseling, behavioral addictions, social struggles such as bullying, obsessive compulsive disorder (OCD), loss, hair or eyelash pulling, mood dysregulation, and sibling conflict. They refer for educational testing, comprehensive psychological evaluations, difficult divorce cases, play therapy, complex cases requiring more than 20 sessions, and children of staff employed by the practice.
The practice features a separate waiting room for psychology patients and front office staff dedicated to managing their schedules. “For anyone who’s trying to make a psychology appointment but can’t be seen in an efficient manner or wants a different day or time, we keep an ongoing move-up list,” Dr. Einhorn said. “If a family calls to cancel an appointment, the front desk person who makes that cancellation will fill out a slip and give it to one of our psychology schedulers. That person will create a move-up list and start filling that appointment. If there’s a cancellation, it’s rare that it goes unfilled.”
Key forms for parents to complete include informed consent, a notice of privacy practices, a late cancel/no show policy, an initial intake agreement, and a summary of parent concerns.
Patient and clinician reaction
According to results from a recent survey of parents whose children were seen by a psychologist at Parker Pediatrics and Adolescents, 89% said it was important for their children to receive mental health services in the same location as their medical care, and 96% were satisfied with the services provided. In addition, 93% said that the experience benefited their child, 72% were satisfied with appointment times, and 55% expressed interest in virtual visits via telemedicine. Meanwhile, a survey of parents whose children have not been seen by a psychologist at the practice found that 65% knew a psychologist was on staff, and only 9% said that there were barriers to their child seeing a psychologist there.
Clinicians themselves benefit from having mental health specialists on site for referrals. “It enables you to be more efficient, and it saves time,” Dr. Rabinowitz said. “There’s knowledge and confidence gained, and it improves satisfaction because physicians don’t have to stay at the office later filling out referral forms. It meets the needs of your patients and their families, it attracts new patients, and you may be able to make some income on this.”
How to get started
Dr. Rabinowitz recommended that, once clinicians at a pediatric practice commit to expanding their services to include mental and behavioral health care, they should hold a corporate/partner meeting, assign responsibilities, and establish a timeline for implementation. “This is all very important,” he said. “Then you have to talk about what kind of arrangement you want to have. You could employ someone to join your practice, hire an independent contractor, establish a space share agreement, or have an out-of-office arrangement.”
For many years, clinicians at Parker Pediatrics and Adolescents had a psychologist perform ADHD evaluations on a consultative basis. “Then, as we saw a need for mental health services about a decade ago, we hired a part-time psychologist who did testing as well as counseling,” Dr. Rabinowitz said. “But that psychologist got very busy, so we hired a full-time psychologist. We continued to hire additional psychologists as need increased.”
Reimbursement issues
Numerous reimbursement barriers to providing mental health services in pediatric primary care exist, he noted, including a lack of payment if mental health codes are used, a lack of “incident to” payments in some areas of the country, existing reimbursement levels, and the fact that same-day billing of physical and mental health often is not allowed. “However, we have found that if we give flu shots during their mental health visit, [insurers] will cover the flu shot,” he said. “Reimbursement for screening is sometimes not covered very well.”
One reimbursement option is the fee-for-service/concierge model, “but that’s not an economic option for many,” he said. “You can’t see Medicaid patients in that model.” Joining a mental health networks is feasible, “but there is poor reimbursement,” he said. “It also creates another layer of administration.”
He recommends financial integration, “but you need to research your options because a lot of it is state dependent.” Other options include grants, insurance contracts, and seeking permission from Medicaid.
Mental health CPT codes that mental health clinicians at the practice commonly use include bill by time (CPT code 99214/15); psychotherapy session that lasts 16-37 minutes (CPT code 90832); psychotherapy session that lasts 38-52 minutes (CPT code 90834); and psychotherapy session that lasts more than 53 minutes (CPT code 90837). Clinicians also can bill by interactive complexity (CPT code 90785) and psychotherapy for crisis (CPT code 90839).
Dr. Rabinowitz and Dr. Einhorn reported having no financial disclosures.
NEW ORLEANS – The way Jay Rabinowitz, MD, MPH, sees it, providing mental and behavioral health care services in your primary care pediatrics practice is a win-win for patients, parents, and clinicians.
For one thing, children with mental and behavioral issues – especially depression and anxiety – make up a good chunk of any pediatrician’s workday. Dr. Rabinowitz, clinical professor of pediatrics at the University of Colorado, Aurora, said at the annual meeting of the American Academy of Pediatrics. “It is the most costly issue in children’s health care today.”
According to “Behavioral Health Integration in Pediatric Primary Care,” a report supported by the Milbank Memorial Fund, one in five children aged 9-17 years have a diagnosable psychiatric disorder, and up to 70% of children in the juvenile justice system have a mental health disorder. The report also found that the treatment of mental health disorders accounts for the most costly childhood medical expenditure, and that between 15% and 20% of children with psychiatric disorders receive specialty care; the rest see their primary care provider. A long-term cost analysis showed significant cost savings: $1 spent on collaborative care saves $6.50 on health care costs.
More recently, the Guidelines for Adolescent Depression in Primary Care (GLAD-PC) found that only 50% of adolescents with depression are diagnosed before reaching adulthood (Pediatrics. March 2018;141[3]:e20174081). As many as two out of three youth with depression are not identified by their pediatrician and do not receive any kind of care.
“Even when diagnosed, only half of these patients are treated appropriately,” said Dr. Rabinowitz, who also practices at Parker (Colo.) Pediatrics and Adolescents.
The guidelines also found that reliance on self-report depression checklists alone lead to substantial numbers of false-positive and false-negative cases. “Primary care providers will benefit from having access to ongoing consultation with mental health providers,” according to the guidelines.
“Integrative care was associated with significant decreases in depression scores, and improved response and remission rates at 12 months, compared with treatment as usual,” Dr. Rabinowitz said.
Providing mental health services in a primary care pediatrics setting also makes sense because there’s a shortage of psychiatrists and psychologists to see them, and it enables patients to get evaluated quicker. “It’s convenient, and it reduces stigma,” he added. “It’s a familiar setting, a familiar provider, and they’re more likely to initiate counseling. Nationwide, 50% of patients who are referred for mental health do not make their initial appointment. Think about that. If you had diabetics in your practice and only 50% would go to the endocrinologist, what would you think?”
How Dr. Rabinowitz and his partners got started
Dr. Rabinowitz and his colleagues created an integrated care model in 2008 by adding a psychologist to their practice, but before doing that, they asked parents of children with mental and behavioral health issues what type of insurance they had. Then they obtained a referral list from the family’s insurer and hoped for the best. “Sometimes I referred to someone I may not have heard of,” Dr. Rabinowitz said. “Usually I did not get follow-up reports, or even know for sure if the patient ever went.”
Today, Parker Pediatrics and Adolescents employs three doctoral-level psychologists: one full-time, one three-quarter time, and one half-time, as well as one master’s-level therapist who works half-time.
“On any given day, we have at least two counselors in our office,” said Lindsey Einhorn, PhD, a licensed clinical psychologist who joined the practice in 2011. She and her colleagues care for children and teens with ADHD, depression, anxiety, behavioral and adjustment disorders, drug counseling, behavioral addictions, social struggles such as bullying, obsessive compulsive disorder (OCD), loss, hair or eyelash pulling, mood dysregulation, and sibling conflict. They refer for educational testing, comprehensive psychological evaluations, difficult divorce cases, play therapy, complex cases requiring more than 20 sessions, and children of staff employed by the practice.
The practice features a separate waiting room for psychology patients and front office staff dedicated to managing their schedules. “For anyone who’s trying to make a psychology appointment but can’t be seen in an efficient manner or wants a different day or time, we keep an ongoing move-up list,” Dr. Einhorn said. “If a family calls to cancel an appointment, the front desk person who makes that cancellation will fill out a slip and give it to one of our psychology schedulers. That person will create a move-up list and start filling that appointment. If there’s a cancellation, it’s rare that it goes unfilled.”
Key forms for parents to complete include informed consent, a notice of privacy practices, a late cancel/no show policy, an initial intake agreement, and a summary of parent concerns.
Patient and clinician reaction
According to results from a recent survey of parents whose children were seen by a psychologist at Parker Pediatrics and Adolescents, 89% said it was important for their children to receive mental health services in the same location as their medical care, and 96% were satisfied with the services provided. In addition, 93% said that the experience benefited their child, 72% were satisfied with appointment times, and 55% expressed interest in virtual visits via telemedicine. Meanwhile, a survey of parents whose children have not been seen by a psychologist at the practice found that 65% knew a psychologist was on staff, and only 9% said that there were barriers to their child seeing a psychologist there.
Clinicians themselves benefit from having mental health specialists on site for referrals. “It enables you to be more efficient, and it saves time,” Dr. Rabinowitz said. “There’s knowledge and confidence gained, and it improves satisfaction because physicians don’t have to stay at the office later filling out referral forms. It meets the needs of your patients and their families, it attracts new patients, and you may be able to make some income on this.”
How to get started
Dr. Rabinowitz recommended that, once clinicians at a pediatric practice commit to expanding their services to include mental and behavioral health care, they should hold a corporate/partner meeting, assign responsibilities, and establish a timeline for implementation. “This is all very important,” he said. “Then you have to talk about what kind of arrangement you want to have. You could employ someone to join your practice, hire an independent contractor, establish a space share agreement, or have an out-of-office arrangement.”
For many years, clinicians at Parker Pediatrics and Adolescents had a psychologist perform ADHD evaluations on a consultative basis. “Then, as we saw a need for mental health services about a decade ago, we hired a part-time psychologist who did testing as well as counseling,” Dr. Rabinowitz said. “But that psychologist got very busy, so we hired a full-time psychologist. We continued to hire additional psychologists as need increased.”
Reimbursement issues
Numerous reimbursement barriers to providing mental health services in pediatric primary care exist, he noted, including a lack of payment if mental health codes are used, a lack of “incident to” payments in some areas of the country, existing reimbursement levels, and the fact that same-day billing of physical and mental health often is not allowed. “However, we have found that if we give flu shots during their mental health visit, [insurers] will cover the flu shot,” he said. “Reimbursement for screening is sometimes not covered very well.”
One reimbursement option is the fee-for-service/concierge model, “but that’s not an economic option for many,” he said. “You can’t see Medicaid patients in that model.” Joining a mental health networks is feasible, “but there is poor reimbursement,” he said. “It also creates another layer of administration.”
He recommends financial integration, “but you need to research your options because a lot of it is state dependent.” Other options include grants, insurance contracts, and seeking permission from Medicaid.
Mental health CPT codes that mental health clinicians at the practice commonly use include bill by time (CPT code 99214/15); psychotherapy session that lasts 16-37 minutes (CPT code 90832); psychotherapy session that lasts 38-52 minutes (CPT code 90834); and psychotherapy session that lasts more than 53 minutes (CPT code 90837). Clinicians also can bill by interactive complexity (CPT code 90785) and psychotherapy for crisis (CPT code 90839).
Dr. Rabinowitz and Dr. Einhorn reported having no financial disclosures.
EXPERT ANALYSIS FROM AAP 2019