User login
IDSA updates COVID guidelines for antibodies, antivirals, other drugs
An infectious disease expert panel cautions against routine use of bamlanivimab (Eli Lilly) and notes that remdesivir (Veklury) can shorten the clinical course of COVID-19 – which could be critical “as hospitals fill up” across the United States.
The group also said the monoclonal antibodies approved for emergency use by the Food and Drug Administration and still in development hold promise, although more clinical trial data are needed.
These and other recommendations appear in updated guidelines from the Infectious Diseases Society of America, released Nov. 18 and Nov. 22.
A conditional ‘no’ on routine bamlanivimab
“The guideline panel gave a conditional recommendation against the routine use of bamlanivimab,” Adarsh Bhimraj, MD, cochair of the IDSA COVID-19 Treatment and Management Guidelines Expert Panel, said.
On Nov. 10, the FDA issued an emergency-use authorization (EUA) for bamlanivimab for use in ambulatory patients with mild to moderate COVID-19.
“We did have a remark that it may be used in patients who have increased risk of severe COVID-19, as it is outlined in the FDA emergency-use authorization issued last week,” he said. He added that use should follow an informed discussion between provider and patient, one in which “the patient puts a very high value on the uncertain benefits and a low value on uncertain adverse events.”
The panel’s rationale was based in part on interim analysis of the phase 2 BLAZE-1 trial, which found 1.6% of people randomly assigned to bamlanivimab had an emergency department visit or hospitalization compared with 6.3% of those receiving a placebo.
“We thought the estimate was too fragile because the number in each arm was very low. Even a small change in these numbers could make the difference nonsignificant,” said Bhimraj, head of the neurologic infectious diseases section in the department of infectious diseases at the Cleveland (Ohio) Clinic.
Awaiting more data on antibody combination
On November 21, the FDA granted an EUA to the casirivimab and imbdevimab monoclonal antibody combination (Regeneron), indicated to treated mild to moderate COVID-19.
“Surprisingly, the preliminary results released in the EUA look a lot like bamlanivimab,” Dr. Bhimraj said.
Unlike bamlanivimab, for which trial details were published, the panel does not yet have the totality of data on casirivimab and imbdevimab, and therefore is not yet making a recommendation. “We want to be cautious as a guideline panel. We are anxiously awaiting the full publication,” he added.
“I do think these monoclonal antibodies show potential for benefit, but as Dr. Bhimraj said, it’s very difficult with the relatively small numbers we’re talking about,” said Rajesh T. Gandhi, MD, cochair of the IDSA COVID-19 Treatment and Management Guidelines Expert Panel.
Remaining questions include the degree of efficacy these antibody therapies will have, as well as which patients are most likely to benefit, added Dr. Gandhi, who is also a professor of medicine at Harvard Medical School and director of HIV Clinical Services and Education at Massachusetts General Hospital, both in Boston.
Furthermore, although there appear to be adequate supplies of remdesivir and dexamethasone, for example, availability and distribution of monoclonal antibodies could present logistic challenges. Prioritizing which high-risk patients receive this therapy and ensuring equity and access to communities most affected by COVID-19, including minority and low socioeconomic populations, need to be addressed, Dr. Gandhi said.
Remdesivir recommended to shorten hospital stays
The panel’s recommendations regarding the use of remdesivir “has largely remained the same,” Dr. Gandhi said. Evidence indicates recovery is faster with remdesivir at 10 days vs 15 days in people taking a placebo.
In the ACTT-1 trial, for example, participants in the treatment group recovered in a median 10 days versus 15 days in the placebo group.
Therefore, the IDSA panel continues to recommend remdesivir treatment for hospitalized patients with COVID-19.
“As hospitals around the United States fill up, the IDSA panel believes the effect of remdesivir on speeding up recovery could be an important benefit, and that is why we continue to suggest its use,” Dr. Gandhi said.
When asked about the World Health Organization–sponsored trial that showed no benefit in terms of mortality, he replied, “Remdesivir is not a home run – we need better drugs.”
A recommendation against lopinavir and ritonavir
In contrast, the panel recommends against use of the lopinavir/ritonavir protease inhibitor combination therapy, based in part on data from a preprint of the Solidarity study.
The open-label Solidarity trial in 30 countries, sponsored by WHO, assessed hydroxychloroquine, interferon, lopinavir/ritonavir, and remdesivir in people hospitalized with COVID-19.
None of these drugs showed an effect on mortality, Dr. Gandhi said. “Better medicines that improve survival are clearly needed.”
Dexamethasone remains the only agent demonstrated to reduce mortality in people hospitalized with COVID-19, he added.
Tocilizumab not for routine use
After critical review of the studies that have emerged since the last IDSA recommendation regarding tocilizumab (Actemra) in September, “the panel still stood with the recommendation against routine use of tocilizumab in hospitalized patients with COVID-19,” Dr. Bhimraj said.
The guidance is based on trials including COVACTA and EMPACTA. Treatment with tocilizumab was not associated with significant differences in mortality. In these and other studies, “we did not really find a significant difference, and that was the reason for the conditional recommendation against routine use of tocilizumab in hospitalized patients,” Dr. Bhimraj said.
Also, although the trials were blinded, “we know treatment with tocilizumab can cause a reduction in C-reactive protein levels,” which could indicate to researchers which participants were receiving active treatment versus placebo, he said.
Jury still out on baricitinib, remdesivir combination
The FDA granted an EUA to the combination of remdesivir and baricitinib (Olumiant) on Nov. 19. However, the IDSA panel is reserving its recommendation on this therapeutic combination until more data emerge.
“We still don’t have complete results of the ACTT-2 study, and the information we do have is what is available in the EUA,” Dr. Bhimraj said. The panel expects to issue guidance once the totality of data become available.
Unanswered questions include why investigators chose a 4-mg dose of baricitinib – twice the 2-mg dose commonly used for treating rheumatoid arthritis – and how many patients in the trial also were treated with steroids.
Dr. Gandhi agreed that the proportion of patients taking a steroid is “really an important consideration.” He added that dexamethasone has become standard of care because it reduces mortality, as well as the number of people requiring oxygen. He said it will be important to know how the baricitinib/remdesivir combination compares with dexamethasone.
“You don’t want to give a drug with less certain benefit over a drug for which there is more certain benefit,” Dr. Gandhi said.
Future possibilities
“The monoclonal antibodies are important to continue studying, particularly in combinations,” Dr. Gandhi said. Researchers are investigating formulations other than IV infusion to make therapy more convenient. For example, a subcutaneous injection like insulin would make administration at home more of a possibility.
Investigators are also looking at oral antiviral therapy, inhaled antivirals, and the promise of using interferon therapy. Dr. Gandhi added there is also “a lot of work around medications to reduce the excess inflammation that drives very severe COVID-19.”
‘Exciting news’ on AstraZeneca vaccine
Although not part of the IDSA guidelines, “we saw the news from AstraZeneca this morning, which is exciting,” Dr. Gandhi said during a media briefing.
Unlike the Pfizer and Moderna messenger RNA vaccines, which use the genetic material of the virus to make the virus proteins that elicit an immune response, the AstraZeneca/Oxford University vaccine uses a viral vector to carry the SARS-CoV-2 protein, to which the body produces an immune response.
“I’m thrilled that several different vaccines are showing important effects at rates higher than the FDA benchmark of 50%, and these are well exceeding that,” Dr. Gandhi said.
“One interesting thing from the [AstraZeneca] press release is they show asymptomatic infection being reduced,” he added. “That is critical because we know a lot of transmission of SARS-CoV-2 comes from asymptomatic people.”
Reasons for optimism
In response to a question about whether the experts feel more optimistic about COVID-19, Dr. Bhimraj said he is cautiously optimistic. “We have made tremendous progress in therapeutic agents, and in how the world has come together in the middle of a catastrophe to collaborate, setting our differences apart, to do trials. That is commendable.”
Dr. Gandhi said he felt more optimistic than he did in the spring. He pointed out that physicians and researchers know a lot more about potential blood clotting complications, how to support patients through severe COVID-19 and keep them off a ventilator whenever possible, and how to provide dexamethasone to reduce the risk of death.
Those benefits are in hospitalized patients, however, and “we need ways to prevent people from getting into the hospital, and that is why we are looking at the monoclonal antibodies,” Dr. Gandhi said. “If borne out in larger trials, that will be a major advance.”
“We need to keep our focus on prevention and go back to our idea of flattening the curve. That is critical so our health care systems do not get overwhelmed during this massive surge we are in,” Dr. Gandhi said. “So masking and social distancing are just as important as they always have been.”
Dr. Bhimraj disclosed no relevant financial relationships. Dr. Gandhi has no disclosures for the past 12 months; in the past 3 years, he has served on scientific advisory boards for Gilead and Merck.
This article first appeared on Medscape.com.
An infectious disease expert panel cautions against routine use of bamlanivimab (Eli Lilly) and notes that remdesivir (Veklury) can shorten the clinical course of COVID-19 – which could be critical “as hospitals fill up” across the United States.
The group also said the monoclonal antibodies approved for emergency use by the Food and Drug Administration and still in development hold promise, although more clinical trial data are needed.
These and other recommendations appear in updated guidelines from the Infectious Diseases Society of America, released Nov. 18 and Nov. 22.
A conditional ‘no’ on routine bamlanivimab
“The guideline panel gave a conditional recommendation against the routine use of bamlanivimab,” Adarsh Bhimraj, MD, cochair of the IDSA COVID-19 Treatment and Management Guidelines Expert Panel, said.
On Nov. 10, the FDA issued an emergency-use authorization (EUA) for bamlanivimab for use in ambulatory patients with mild to moderate COVID-19.
“We did have a remark that it may be used in patients who have increased risk of severe COVID-19, as it is outlined in the FDA emergency-use authorization issued last week,” he said. He added that use should follow an informed discussion between provider and patient, one in which “the patient puts a very high value on the uncertain benefits and a low value on uncertain adverse events.”
The panel’s rationale was based in part on interim analysis of the phase 2 BLAZE-1 trial, which found 1.6% of people randomly assigned to bamlanivimab had an emergency department visit or hospitalization compared with 6.3% of those receiving a placebo.
“We thought the estimate was too fragile because the number in each arm was very low. Even a small change in these numbers could make the difference nonsignificant,” said Bhimraj, head of the neurologic infectious diseases section in the department of infectious diseases at the Cleveland (Ohio) Clinic.
Awaiting more data on antibody combination
On November 21, the FDA granted an EUA to the casirivimab and imbdevimab monoclonal antibody combination (Regeneron), indicated to treated mild to moderate COVID-19.
“Surprisingly, the preliminary results released in the EUA look a lot like bamlanivimab,” Dr. Bhimraj said.
Unlike bamlanivimab, for which trial details were published, the panel does not yet have the totality of data on casirivimab and imbdevimab, and therefore is not yet making a recommendation. “We want to be cautious as a guideline panel. We are anxiously awaiting the full publication,” he added.
“I do think these monoclonal antibodies show potential for benefit, but as Dr. Bhimraj said, it’s very difficult with the relatively small numbers we’re talking about,” said Rajesh T. Gandhi, MD, cochair of the IDSA COVID-19 Treatment and Management Guidelines Expert Panel.
Remaining questions include the degree of efficacy these antibody therapies will have, as well as which patients are most likely to benefit, added Dr. Gandhi, who is also a professor of medicine at Harvard Medical School and director of HIV Clinical Services and Education at Massachusetts General Hospital, both in Boston.
Furthermore, although there appear to be adequate supplies of remdesivir and dexamethasone, for example, availability and distribution of monoclonal antibodies could present logistic challenges. Prioritizing which high-risk patients receive this therapy and ensuring equity and access to communities most affected by COVID-19, including minority and low socioeconomic populations, need to be addressed, Dr. Gandhi said.
Remdesivir recommended to shorten hospital stays
The panel’s recommendations regarding the use of remdesivir “has largely remained the same,” Dr. Gandhi said. Evidence indicates recovery is faster with remdesivir at 10 days vs 15 days in people taking a placebo.
In the ACTT-1 trial, for example, participants in the treatment group recovered in a median 10 days versus 15 days in the placebo group.
Therefore, the IDSA panel continues to recommend remdesivir treatment for hospitalized patients with COVID-19.
“As hospitals around the United States fill up, the IDSA panel believes the effect of remdesivir on speeding up recovery could be an important benefit, and that is why we continue to suggest its use,” Dr. Gandhi said.
When asked about the World Health Organization–sponsored trial that showed no benefit in terms of mortality, he replied, “Remdesivir is not a home run – we need better drugs.”
A recommendation against lopinavir and ritonavir
In contrast, the panel recommends against use of the lopinavir/ritonavir protease inhibitor combination therapy, based in part on data from a preprint of the Solidarity study.
The open-label Solidarity trial in 30 countries, sponsored by WHO, assessed hydroxychloroquine, interferon, lopinavir/ritonavir, and remdesivir in people hospitalized with COVID-19.
None of these drugs showed an effect on mortality, Dr. Gandhi said. “Better medicines that improve survival are clearly needed.”
Dexamethasone remains the only agent demonstrated to reduce mortality in people hospitalized with COVID-19, he added.
Tocilizumab not for routine use
After critical review of the studies that have emerged since the last IDSA recommendation regarding tocilizumab (Actemra) in September, “the panel still stood with the recommendation against routine use of tocilizumab in hospitalized patients with COVID-19,” Dr. Bhimraj said.
The guidance is based on trials including COVACTA and EMPACTA. Treatment with tocilizumab was not associated with significant differences in mortality. In these and other studies, “we did not really find a significant difference, and that was the reason for the conditional recommendation against routine use of tocilizumab in hospitalized patients,” Dr. Bhimraj said.
Also, although the trials were blinded, “we know treatment with tocilizumab can cause a reduction in C-reactive protein levels,” which could indicate to researchers which participants were receiving active treatment versus placebo, he said.
Jury still out on baricitinib, remdesivir combination
The FDA granted an EUA to the combination of remdesivir and baricitinib (Olumiant) on Nov. 19. However, the IDSA panel is reserving its recommendation on this therapeutic combination until more data emerge.
“We still don’t have complete results of the ACTT-2 study, and the information we do have is what is available in the EUA,” Dr. Bhimraj said. The panel expects to issue guidance once the totality of data become available.
Unanswered questions include why investigators chose a 4-mg dose of baricitinib – twice the 2-mg dose commonly used for treating rheumatoid arthritis – and how many patients in the trial also were treated with steroids.
Dr. Gandhi agreed that the proportion of patients taking a steroid is “really an important consideration.” He added that dexamethasone has become standard of care because it reduces mortality, as well as the number of people requiring oxygen. He said it will be important to know how the baricitinib/remdesivir combination compares with dexamethasone.
“You don’t want to give a drug with less certain benefit over a drug for which there is more certain benefit,” Dr. Gandhi said.
Future possibilities
“The monoclonal antibodies are important to continue studying, particularly in combinations,” Dr. Gandhi said. Researchers are investigating formulations other than IV infusion to make therapy more convenient. For example, a subcutaneous injection like insulin would make administration at home more of a possibility.
Investigators are also looking at oral antiviral therapy, inhaled antivirals, and the promise of using interferon therapy. Dr. Gandhi added there is also “a lot of work around medications to reduce the excess inflammation that drives very severe COVID-19.”
‘Exciting news’ on AstraZeneca vaccine
Although not part of the IDSA guidelines, “we saw the news from AstraZeneca this morning, which is exciting,” Dr. Gandhi said during a media briefing.
Unlike the Pfizer and Moderna messenger RNA vaccines, which use the genetic material of the virus to make the virus proteins that elicit an immune response, the AstraZeneca/Oxford University vaccine uses a viral vector to carry the SARS-CoV-2 protein, to which the body produces an immune response.
“I’m thrilled that several different vaccines are showing important effects at rates higher than the FDA benchmark of 50%, and these are well exceeding that,” Dr. Gandhi said.
“One interesting thing from the [AstraZeneca] press release is they show asymptomatic infection being reduced,” he added. “That is critical because we know a lot of transmission of SARS-CoV-2 comes from asymptomatic people.”
Reasons for optimism
In response to a question about whether the experts feel more optimistic about COVID-19, Dr. Bhimraj said he is cautiously optimistic. “We have made tremendous progress in therapeutic agents, and in how the world has come together in the middle of a catastrophe to collaborate, setting our differences apart, to do trials. That is commendable.”
Dr. Gandhi said he felt more optimistic than he did in the spring. He pointed out that physicians and researchers know a lot more about potential blood clotting complications, how to support patients through severe COVID-19 and keep them off a ventilator whenever possible, and how to provide dexamethasone to reduce the risk of death.
Those benefits are in hospitalized patients, however, and “we need ways to prevent people from getting into the hospital, and that is why we are looking at the monoclonal antibodies,” Dr. Gandhi said. “If borne out in larger trials, that will be a major advance.”
“We need to keep our focus on prevention and go back to our idea of flattening the curve. That is critical so our health care systems do not get overwhelmed during this massive surge we are in,” Dr. Gandhi said. “So masking and social distancing are just as important as they always have been.”
Dr. Bhimraj disclosed no relevant financial relationships. Dr. Gandhi has no disclosures for the past 12 months; in the past 3 years, he has served on scientific advisory boards for Gilead and Merck.
This article first appeared on Medscape.com.
An infectious disease expert panel cautions against routine use of bamlanivimab (Eli Lilly) and notes that remdesivir (Veklury) can shorten the clinical course of COVID-19 – which could be critical “as hospitals fill up” across the United States.
The group also said the monoclonal antibodies approved for emergency use by the Food and Drug Administration and still in development hold promise, although more clinical trial data are needed.
These and other recommendations appear in updated guidelines from the Infectious Diseases Society of America, released Nov. 18 and Nov. 22.
A conditional ‘no’ on routine bamlanivimab
“The guideline panel gave a conditional recommendation against the routine use of bamlanivimab,” Adarsh Bhimraj, MD, cochair of the IDSA COVID-19 Treatment and Management Guidelines Expert Panel, said.
On Nov. 10, the FDA issued an emergency-use authorization (EUA) for bamlanivimab for use in ambulatory patients with mild to moderate COVID-19.
“We did have a remark that it may be used in patients who have increased risk of severe COVID-19, as it is outlined in the FDA emergency-use authorization issued last week,” he said. He added that use should follow an informed discussion between provider and patient, one in which “the patient puts a very high value on the uncertain benefits and a low value on uncertain adverse events.”
The panel’s rationale was based in part on interim analysis of the phase 2 BLAZE-1 trial, which found 1.6% of people randomly assigned to bamlanivimab had an emergency department visit or hospitalization compared with 6.3% of those receiving a placebo.
“We thought the estimate was too fragile because the number in each arm was very low. Even a small change in these numbers could make the difference nonsignificant,” said Bhimraj, head of the neurologic infectious diseases section in the department of infectious diseases at the Cleveland (Ohio) Clinic.
Awaiting more data on antibody combination
On November 21, the FDA granted an EUA to the casirivimab and imbdevimab monoclonal antibody combination (Regeneron), indicated to treated mild to moderate COVID-19.
“Surprisingly, the preliminary results released in the EUA look a lot like bamlanivimab,” Dr. Bhimraj said.
Unlike bamlanivimab, for which trial details were published, the panel does not yet have the totality of data on casirivimab and imbdevimab, and therefore is not yet making a recommendation. “We want to be cautious as a guideline panel. We are anxiously awaiting the full publication,” he added.
“I do think these monoclonal antibodies show potential for benefit, but as Dr. Bhimraj said, it’s very difficult with the relatively small numbers we’re talking about,” said Rajesh T. Gandhi, MD, cochair of the IDSA COVID-19 Treatment and Management Guidelines Expert Panel.
Remaining questions include the degree of efficacy these antibody therapies will have, as well as which patients are most likely to benefit, added Dr. Gandhi, who is also a professor of medicine at Harvard Medical School and director of HIV Clinical Services and Education at Massachusetts General Hospital, both in Boston.
Furthermore, although there appear to be adequate supplies of remdesivir and dexamethasone, for example, availability and distribution of monoclonal antibodies could present logistic challenges. Prioritizing which high-risk patients receive this therapy and ensuring equity and access to communities most affected by COVID-19, including minority and low socioeconomic populations, need to be addressed, Dr. Gandhi said.
Remdesivir recommended to shorten hospital stays
The panel’s recommendations regarding the use of remdesivir “has largely remained the same,” Dr. Gandhi said. Evidence indicates recovery is faster with remdesivir at 10 days vs 15 days in people taking a placebo.
In the ACTT-1 trial, for example, participants in the treatment group recovered in a median 10 days versus 15 days in the placebo group.
Therefore, the IDSA panel continues to recommend remdesivir treatment for hospitalized patients with COVID-19.
“As hospitals around the United States fill up, the IDSA panel believes the effect of remdesivir on speeding up recovery could be an important benefit, and that is why we continue to suggest its use,” Dr. Gandhi said.
When asked about the World Health Organization–sponsored trial that showed no benefit in terms of mortality, he replied, “Remdesivir is not a home run – we need better drugs.”
A recommendation against lopinavir and ritonavir
In contrast, the panel recommends against use of the lopinavir/ritonavir protease inhibitor combination therapy, based in part on data from a preprint of the Solidarity study.
The open-label Solidarity trial in 30 countries, sponsored by WHO, assessed hydroxychloroquine, interferon, lopinavir/ritonavir, and remdesivir in people hospitalized with COVID-19.
None of these drugs showed an effect on mortality, Dr. Gandhi said. “Better medicines that improve survival are clearly needed.”
Dexamethasone remains the only agent demonstrated to reduce mortality in people hospitalized with COVID-19, he added.
Tocilizumab not for routine use
After critical review of the studies that have emerged since the last IDSA recommendation regarding tocilizumab (Actemra) in September, “the panel still stood with the recommendation against routine use of tocilizumab in hospitalized patients with COVID-19,” Dr. Bhimraj said.
The guidance is based on trials including COVACTA and EMPACTA. Treatment with tocilizumab was not associated with significant differences in mortality. In these and other studies, “we did not really find a significant difference, and that was the reason for the conditional recommendation against routine use of tocilizumab in hospitalized patients,” Dr. Bhimraj said.
Also, although the trials were blinded, “we know treatment with tocilizumab can cause a reduction in C-reactive protein levels,” which could indicate to researchers which participants were receiving active treatment versus placebo, he said.
Jury still out on baricitinib, remdesivir combination
The FDA granted an EUA to the combination of remdesivir and baricitinib (Olumiant) on Nov. 19. However, the IDSA panel is reserving its recommendation on this therapeutic combination until more data emerge.
“We still don’t have complete results of the ACTT-2 study, and the information we do have is what is available in the EUA,” Dr. Bhimraj said. The panel expects to issue guidance once the totality of data become available.
Unanswered questions include why investigators chose a 4-mg dose of baricitinib – twice the 2-mg dose commonly used for treating rheumatoid arthritis – and how many patients in the trial also were treated with steroids.
Dr. Gandhi agreed that the proportion of patients taking a steroid is “really an important consideration.” He added that dexamethasone has become standard of care because it reduces mortality, as well as the number of people requiring oxygen. He said it will be important to know how the baricitinib/remdesivir combination compares with dexamethasone.
“You don’t want to give a drug with less certain benefit over a drug for which there is more certain benefit,” Dr. Gandhi said.
Future possibilities
“The monoclonal antibodies are important to continue studying, particularly in combinations,” Dr. Gandhi said. Researchers are investigating formulations other than IV infusion to make therapy more convenient. For example, a subcutaneous injection like insulin would make administration at home more of a possibility.
Investigators are also looking at oral antiviral therapy, inhaled antivirals, and the promise of using interferon therapy. Dr. Gandhi added there is also “a lot of work around medications to reduce the excess inflammation that drives very severe COVID-19.”
‘Exciting news’ on AstraZeneca vaccine
Although not part of the IDSA guidelines, “we saw the news from AstraZeneca this morning, which is exciting,” Dr. Gandhi said during a media briefing.
Unlike the Pfizer and Moderna messenger RNA vaccines, which use the genetic material of the virus to make the virus proteins that elicit an immune response, the AstraZeneca/Oxford University vaccine uses a viral vector to carry the SARS-CoV-2 protein, to which the body produces an immune response.
“I’m thrilled that several different vaccines are showing important effects at rates higher than the FDA benchmark of 50%, and these are well exceeding that,” Dr. Gandhi said.
“One interesting thing from the [AstraZeneca] press release is they show asymptomatic infection being reduced,” he added. “That is critical because we know a lot of transmission of SARS-CoV-2 comes from asymptomatic people.”
Reasons for optimism
In response to a question about whether the experts feel more optimistic about COVID-19, Dr. Bhimraj said he is cautiously optimistic. “We have made tremendous progress in therapeutic agents, and in how the world has come together in the middle of a catastrophe to collaborate, setting our differences apart, to do trials. That is commendable.”
Dr. Gandhi said he felt more optimistic than he did in the spring. He pointed out that physicians and researchers know a lot more about potential blood clotting complications, how to support patients through severe COVID-19 and keep them off a ventilator whenever possible, and how to provide dexamethasone to reduce the risk of death.
Those benefits are in hospitalized patients, however, and “we need ways to prevent people from getting into the hospital, and that is why we are looking at the monoclonal antibodies,” Dr. Gandhi said. “If borne out in larger trials, that will be a major advance.”
“We need to keep our focus on prevention and go back to our idea of flattening the curve. That is critical so our health care systems do not get overwhelmed during this massive surge we are in,” Dr. Gandhi said. “So masking and social distancing are just as important as they always have been.”
Dr. Bhimraj disclosed no relevant financial relationships. Dr. Gandhi has no disclosures for the past 12 months; in the past 3 years, he has served on scientific advisory boards for Gilead and Merck.
This article first appeared on Medscape.com.
Stopping methotrexate, staying on etanercept provides best RA outcomes after remission
In patients with RA whose disease is well controlled by methotrexate combined with etanercept, withdrawal of methotrexate led to long-term outcomes that were nearly as good as continuation of combination therapy. The finding comes from the randomized, controlled SEAM-RA trial that sought to address weaknesses of previous studies. It included a long lead-in time with stringent criteria to ensure that participants had very good disease control.
Both the American College of Rheumatology and the European League Against Rheumatism recommend tapering medication in RA patients who are in long-term remission, but there is no established optimal strategy.
“There have been some prior RA trials that have looked at therapy reduction or withdrawal, but most did not use a very stringent definition of how well people were when they began. Were they in remission, or only in low disease activity?” said Jeffrey R. Curtis, MD, during a presentation of the results at the virtual annual meeting of the ACR. The study was also published online Nov. 18 in Arthritis & Rheumatology.
Stringent remission criteria
The key feature of the trial was the 6-month run-in period, when subjects were taking 50 mg etanercept once per week and 10-25 mg of oral methotrexate once per week, and had to complete at least three visits. They were excluded from the ensuing randomization if they had a Simplified Disease Activity Index (SDAI) score >3.3 and ≤11 at two or more visits, had an SDAI >11 at any time during the run-in period, or had an SDAI >3.3 at the third run-in visit.
“We [wanted them] to be doing quite well for a long period of time. That was empirically confirmed under observation as part of the lead-in period, and even before that, the clinical investigator had to affirm that they believed the patient was doing well for 6 or more months even before they were screened to enter the trial,” said Dr. Curtis, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham.
Once enrolled in the trial, patients were randomized 2:2:1 to continuing etanercept only (n = 101), continuing methotrexate only (n = 101), or continuing both medications (n = 51). Patients were eligible for rescue after randomization if they had an SDAI score >11 at any time, SDAI between 3.3 and 11 on three separate visits, or between 3.3 and 11 at two consecutive visits at least 2 weeks apart. About three-quarters of patients in each treatment arm were female, with a mean age of about 55 years, and 82%-91% were White.
Good remission recovery with rescue therapy
At week 48, 28.7% of the methotrexate-only group were in remission (SDAI ≤3.3), compared with 49.5% of the etanercept-only group (P = .004) and 52.9% of the combination group (P = .006). Time to disease worsening was shorter in the methotrexate-only group (median, 198 days) than in the etanercept-only group (median, not estimable; P < .001) and the combined therapy group (median, not estimable; P < .001).
The researchers also found that most patients who underwent rescue therapy once again achieved remission, including 71% of the methotrexate-only group, 75% of the etanercept-only group, and 80% of the combination therapy group. There was no between-group differences in the time required to reattain remission.
The high rate of remission recovery was a good sign, Dr. Curtis said. “To me as a clinician, the risk to try [withdrawing a medication] is quite low because the likelihood you can regain where you were before is quite good. It’s obviously more successful if you stop methotrexate and continue etanercept than if you do the reverse, but to me, this is quite a practical trial, and in fact the rigor of the inclusion criteria are much more like the patients I’m talking to about stopping therapy than some of the past studies in this regard. I think it’s quite useful in terms of generalizability. We want people that are doing this well or close to it before we take away medication.”
Positive reactions from rheumatologists
The reaction from the viewing audience was also positive. “I think this study fills a big data gap for what we do in clinical practice,” wrote Janet Pope, MD, in comments during the session.
Dr. Pope, who is a professor of medicine at the University of Western Ontario and head of rheumatology at St. Joseph’s Health Centre, both in London, said that the results build on previous work, including the CAMEO study, which showed that discontinuation of methotrexate in patients taking methotrexate and etanercept failed to achieve noninferiority to continuation of both medications, and the PRIZE study, which showed that continuing combination therapy at a reduced dose led to better outcomes than did switching to methotrexate alone or placebo. “This may be for some patients what they prefer if they don’t tolerate methotrexate,” she added.
“It’s wonderful to have these data to counsel patients. This is something we face every day,” wrote Elizabeth Wahl, MD, who is an acting assistant professor at the University of Washington, Seattle, and acting chief of rheumatology at the VA Puget Sound Healthcare System.
The study was funded by Amgen. Dr. Curtis has received grants or research support from AbbVie, Amgen, Bristol-Myers Squibb, Corrona, Janssen, Lilly, Myriad, Pfizer, Regeneron, Roche, and UCB. Dr. Pope consults for a variety of pharmaceutical companies. Dr. Wahl has no relevant financial disclosures.
SOURCE: Curtis JR et al. Arthritis Rheumatol. 2020;72(suppl 10), Abstract 0939.
In patients with RA whose disease is well controlled by methotrexate combined with etanercept, withdrawal of methotrexate led to long-term outcomes that were nearly as good as continuation of combination therapy. The finding comes from the randomized, controlled SEAM-RA trial that sought to address weaknesses of previous studies. It included a long lead-in time with stringent criteria to ensure that participants had very good disease control.
Both the American College of Rheumatology and the European League Against Rheumatism recommend tapering medication in RA patients who are in long-term remission, but there is no established optimal strategy.
“There have been some prior RA trials that have looked at therapy reduction or withdrawal, but most did not use a very stringent definition of how well people were when they began. Were they in remission, or only in low disease activity?” said Jeffrey R. Curtis, MD, during a presentation of the results at the virtual annual meeting of the ACR. The study was also published online Nov. 18 in Arthritis & Rheumatology.
Stringent remission criteria
The key feature of the trial was the 6-month run-in period, when subjects were taking 50 mg etanercept once per week and 10-25 mg of oral methotrexate once per week, and had to complete at least three visits. They were excluded from the ensuing randomization if they had a Simplified Disease Activity Index (SDAI) score >3.3 and ≤11 at two or more visits, had an SDAI >11 at any time during the run-in period, or had an SDAI >3.3 at the third run-in visit.
“We [wanted them] to be doing quite well for a long period of time. That was empirically confirmed under observation as part of the lead-in period, and even before that, the clinical investigator had to affirm that they believed the patient was doing well for 6 or more months even before they were screened to enter the trial,” said Dr. Curtis, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham.
Once enrolled in the trial, patients were randomized 2:2:1 to continuing etanercept only (n = 101), continuing methotrexate only (n = 101), or continuing both medications (n = 51). Patients were eligible for rescue after randomization if they had an SDAI score >11 at any time, SDAI between 3.3 and 11 on three separate visits, or between 3.3 and 11 at two consecutive visits at least 2 weeks apart. About three-quarters of patients in each treatment arm were female, with a mean age of about 55 years, and 82%-91% were White.
Good remission recovery with rescue therapy
At week 48, 28.7% of the methotrexate-only group were in remission (SDAI ≤3.3), compared with 49.5% of the etanercept-only group (P = .004) and 52.9% of the combination group (P = .006). Time to disease worsening was shorter in the methotrexate-only group (median, 198 days) than in the etanercept-only group (median, not estimable; P < .001) and the combined therapy group (median, not estimable; P < .001).
The researchers also found that most patients who underwent rescue therapy once again achieved remission, including 71% of the methotrexate-only group, 75% of the etanercept-only group, and 80% of the combination therapy group. There was no between-group differences in the time required to reattain remission.
The high rate of remission recovery was a good sign, Dr. Curtis said. “To me as a clinician, the risk to try [withdrawing a medication] is quite low because the likelihood you can regain where you were before is quite good. It’s obviously more successful if you stop methotrexate and continue etanercept than if you do the reverse, but to me, this is quite a practical trial, and in fact the rigor of the inclusion criteria are much more like the patients I’m talking to about stopping therapy than some of the past studies in this regard. I think it’s quite useful in terms of generalizability. We want people that are doing this well or close to it before we take away medication.”
Positive reactions from rheumatologists
The reaction from the viewing audience was also positive. “I think this study fills a big data gap for what we do in clinical practice,” wrote Janet Pope, MD, in comments during the session.
Dr. Pope, who is a professor of medicine at the University of Western Ontario and head of rheumatology at St. Joseph’s Health Centre, both in London, said that the results build on previous work, including the CAMEO study, which showed that discontinuation of methotrexate in patients taking methotrexate and etanercept failed to achieve noninferiority to continuation of both medications, and the PRIZE study, which showed that continuing combination therapy at a reduced dose led to better outcomes than did switching to methotrexate alone or placebo. “This may be for some patients what they prefer if they don’t tolerate methotrexate,” she added.
“It’s wonderful to have these data to counsel patients. This is something we face every day,” wrote Elizabeth Wahl, MD, who is an acting assistant professor at the University of Washington, Seattle, and acting chief of rheumatology at the VA Puget Sound Healthcare System.
The study was funded by Amgen. Dr. Curtis has received grants or research support from AbbVie, Amgen, Bristol-Myers Squibb, Corrona, Janssen, Lilly, Myriad, Pfizer, Regeneron, Roche, and UCB. Dr. Pope consults for a variety of pharmaceutical companies. Dr. Wahl has no relevant financial disclosures.
SOURCE: Curtis JR et al. Arthritis Rheumatol. 2020;72(suppl 10), Abstract 0939.
In patients with RA whose disease is well controlled by methotrexate combined with etanercept, withdrawal of methotrexate led to long-term outcomes that were nearly as good as continuation of combination therapy. The finding comes from the randomized, controlled SEAM-RA trial that sought to address weaknesses of previous studies. It included a long lead-in time with stringent criteria to ensure that participants had very good disease control.
Both the American College of Rheumatology and the European League Against Rheumatism recommend tapering medication in RA patients who are in long-term remission, but there is no established optimal strategy.
“There have been some prior RA trials that have looked at therapy reduction or withdrawal, but most did not use a very stringent definition of how well people were when they began. Were they in remission, or only in low disease activity?” said Jeffrey R. Curtis, MD, during a presentation of the results at the virtual annual meeting of the ACR. The study was also published online Nov. 18 in Arthritis & Rheumatology.
Stringent remission criteria
The key feature of the trial was the 6-month run-in period, when subjects were taking 50 mg etanercept once per week and 10-25 mg of oral methotrexate once per week, and had to complete at least three visits. They were excluded from the ensuing randomization if they had a Simplified Disease Activity Index (SDAI) score >3.3 and ≤11 at two or more visits, had an SDAI >11 at any time during the run-in period, or had an SDAI >3.3 at the third run-in visit.
“We [wanted them] to be doing quite well for a long period of time. That was empirically confirmed under observation as part of the lead-in period, and even before that, the clinical investigator had to affirm that they believed the patient was doing well for 6 or more months even before they were screened to enter the trial,” said Dr. Curtis, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham.
Once enrolled in the trial, patients were randomized 2:2:1 to continuing etanercept only (n = 101), continuing methotrexate only (n = 101), or continuing both medications (n = 51). Patients were eligible for rescue after randomization if they had an SDAI score >11 at any time, SDAI between 3.3 and 11 on three separate visits, or between 3.3 and 11 at two consecutive visits at least 2 weeks apart. About three-quarters of patients in each treatment arm were female, with a mean age of about 55 years, and 82%-91% were White.
Good remission recovery with rescue therapy
At week 48, 28.7% of the methotrexate-only group were in remission (SDAI ≤3.3), compared with 49.5% of the etanercept-only group (P = .004) and 52.9% of the combination group (P = .006). Time to disease worsening was shorter in the methotrexate-only group (median, 198 days) than in the etanercept-only group (median, not estimable; P < .001) and the combined therapy group (median, not estimable; P < .001).
The researchers also found that most patients who underwent rescue therapy once again achieved remission, including 71% of the methotrexate-only group, 75% of the etanercept-only group, and 80% of the combination therapy group. There was no between-group differences in the time required to reattain remission.
The high rate of remission recovery was a good sign, Dr. Curtis said. “To me as a clinician, the risk to try [withdrawing a medication] is quite low because the likelihood you can regain where you were before is quite good. It’s obviously more successful if you stop methotrexate and continue etanercept than if you do the reverse, but to me, this is quite a practical trial, and in fact the rigor of the inclusion criteria are much more like the patients I’m talking to about stopping therapy than some of the past studies in this regard. I think it’s quite useful in terms of generalizability. We want people that are doing this well or close to it before we take away medication.”
Positive reactions from rheumatologists
The reaction from the viewing audience was also positive. “I think this study fills a big data gap for what we do in clinical practice,” wrote Janet Pope, MD, in comments during the session.
Dr. Pope, who is a professor of medicine at the University of Western Ontario and head of rheumatology at St. Joseph’s Health Centre, both in London, said that the results build on previous work, including the CAMEO study, which showed that discontinuation of methotrexate in patients taking methotrexate and etanercept failed to achieve noninferiority to continuation of both medications, and the PRIZE study, which showed that continuing combination therapy at a reduced dose led to better outcomes than did switching to methotrexate alone or placebo. “This may be for some patients what they prefer if they don’t tolerate methotrexate,” she added.
“It’s wonderful to have these data to counsel patients. This is something we face every day,” wrote Elizabeth Wahl, MD, who is an acting assistant professor at the University of Washington, Seattle, and acting chief of rheumatology at the VA Puget Sound Healthcare System.
The study was funded by Amgen. Dr. Curtis has received grants or research support from AbbVie, Amgen, Bristol-Myers Squibb, Corrona, Janssen, Lilly, Myriad, Pfizer, Regeneron, Roche, and UCB. Dr. Pope consults for a variety of pharmaceutical companies. Dr. Wahl has no relevant financial disclosures.
SOURCE: Curtis JR et al. Arthritis Rheumatol. 2020;72(suppl 10), Abstract 0939.
FROM ACR 2020
Finerenone’s heart benefits hold up in T2D patients without CVD
Finerenone, the first nonsteroidal mineralocorticoid receptor antagonist to complete a phase 3 trial, showed cardiovascular benefits in patients with type 2 diabetes and chronic kidney disease, regardless of whether they entered the study with a history of cardiovascular disease, in follow-up analyses of the FIDELIO-DKD trial, which included 5,674 patients.
“Finerenone demonstrated benefits for primary and secondary cardiovascular disease protection,” said Gerasimos Filippatos, MD, at the American Heart Association scientific sessions. Finerenone treatment cut the rate of cardiovascular death, nonfatal MI or stroke, or heart failure hospitalization, when compared with placebo, by a relative 15% among patients with a history of cardiovascular disease (CVD), and by a relative 14% in patients without this history, differences that met a statistical test for consistency. But the absolute, drug-associated increments in benefit over placebo differed between the two CVD subgroups because of a sharp underlying difference in event rates.
In contrast, the analyses reported by Dr. Filippatos and associates from the FIDELIO-DKD study showed significant heterogeneity based on the presence or absence of CVD for the study’s primary endpoint, a composite renal metric that tallied the combined rate of death from renal causes, renal failure, or a sustained drop in estimated glomerular filtration rate of at least 40%. Researchers enrolled patients into FIDELIO-DKD based on having type 2 diabetes (T2D) and chronic kidney disease (CKD). The prevalence of a history of CVD was 46%.
Among patients with a history of CVD, the composite adverse CVD outcome occurred at a rate of 8.5/100 patient-years in patients on placebo and in 7.18/100 patients years among those on finerenone during a median of 2.6 years of follow-up, a 1.32/100–patient-year absolute between-group difference. Among patients in a primary prevention setting, incident CVD event rates during follow-up were roughly half that in the secondary prevention patients. The upshot was that, in the placebo group, the rate was 3.92/100 patient- years, and in those on finerenone was 3.43/100 patient-years, a 0.49/100–patient-year absolute difference.
CVD history produced heterogeneity for the primary endpoint
In the analysis that focused on the study’s primary, renal endpoint, among patients identified as having CVD at study entry, the outcome occurred at a rate of 9.06/100 patient-years in the placebo subgroup and at a rate of 6.6/100 patient years in those who received finerenone, a significant 30% relative risk reduction and an absolute between-group difference of 2.46/100 patient-years.
In contrast, among patients without a CVD history, the composite renal endpoint occurred at a rate of 9.1/100 patient-years in the placebo patients and 8.42/100 patient-years in those on finerenone, a 6% relative risk reduction that was not significant, and a 0.68/100–patient-year absolute difference. This disparity in the primary event rate between the two treatment arms reached statistical significance (P = .016), the investigators reported in the published version of the report in Circulation that simultaneously appeared online.
“The totality of evidence suggests that finerenone could be used in patients with T2D with or without a history of CVD,” explained Dr. Filippatos in an interview. “The P-interaction for the composite kidney outcome is significant, but it is not corrected for multiple testing; therefore, it might be a false-chance finding and must be interpreted cautiously.
Furthermore, in another prespecified kidney composite outcome the results were consistent in patients with and without a history of CVD. In sum, all the FIDELIO-DKD analyses so far are “suggestive of a beneficial effect in patients without a history of CVD.”
Despite these patients receiving guideline directed therapies, “there remains a high unmet medical need in patients with T2D and CKD,” added Dr. Filippatos, professor of cardiology at the University of Athens. “We use multiple treatments for patients with heart failure, and we should use the same mindset for treating patients with T2D and CKD. The costs of dialysis and kidney transplant are very high, so it is important to consider options that slow progression of CKD in these patients.”
In FIDELIO-DKD, virtually all patients were on background therapy with a renin-angiotensin-system (RAS) inhibitor, so the trial’s results suggest that treatment should at least involve dual therapy with finerenone and a RAS inhibitor. Fewer than 5% were on background therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor, a drug class recently established as another key agent for treating CKD in patients with T2D, setting up the prospect for triple therapy, although this approach has not yet undergone prospective testing.
Combining RAS inhibition, finerenone, and an SGLT2 inhibitor is “potentially a marriage made in diabetes heaven,” commented Deepak L. Bhatt, MD, a professor of medicine at Harvard Medical School, Boston, who has not participated in finerenone studies.
Finerenone looks better for safety
Regardless of subgroup analyses based on history of CVD, the findings from all patients enrolled in FIDELIO-DKD were positive for the both the primary renal outcome and key secondary outcome of composite CVD events. In the total randomized cohort, treatment with finerenone on top of optimized treatment with an ACE inhibitor or angiotensin receptor blocker (RAS inhibition) led to a significant 18% relative risk reduction, compared with placebo, for the primary renal endpoint, and a significant 14% relative drop in the key secondary CVD outcome. Those results were published in October in the New England Journal of Medicine.
For treating patients with T2D and CKD ,finerenone overall “looks like a major advance,” Dr. Bhatt said in an interview.
In addition to the positive efficacy results, several experts also focused on what they saw as superior safety of finerenone in the trial, compared with the historical safety of the steroidal mineralocorticoid receptor antagonists (MRAs) now in use: spironolactone and eplerenone.
“I’m a big believer in spironolactone, but it has issues with side effects, and eplerenone never seemed to catch on,” said Dr. Bhatt, who is also executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
“A lot of physicians like these MRAs, but acknowledge that side effects have kept these drugs from being used to the extent they should.” The existing MRAs, especially spironolactone, have become a key drug class for treating heart failure with reduced ejection fraction (and, some claim, for also treating heart failure with preserved ejection fraction), as well as treatment-resistant hypertension and primary aldosteronism. By design, FIDELIO-DKD did not enroll patients with heart failure because treatment with an MRA is indicated for those with heart failure with reduced ejection fraction.
The spironolactone adverse effect that generates the greatest concern is hyperkalemia. During his discussion of FIDELIO-DKD as designated discussant, Christoph Wanner, MD, noted a recent study in which the incidence of hyperkalemia severe enough to cause study discontinuation was 23% among patients treated with spironolactone for heart failure, which contrasts with the 2.3% rate in FIDELIO-DKD among finerenone recipients. This hyperkalemia incidence from finerenone also improved on the historical performance of other drugs, like aliskiren (Tekturna), said Dr. Wanner, professor and head of nephrology at the University of Würzburg (Germany).
The FIDELIO-DKD results place finerenone alongside the RAS- and SGLT2-inhibitor drug classes as appropriate treatments for most patients with T2D and CKD. “We have entered a new era of effective treatment for diabetic kidney disease,” Dr. Wanner declared.
“The overall safety profile of finerenone looked better, including hyperkalemia,” said Dr. Bhatt. “Hyperkalemia with spironolactone is not necessarily as bad as the perception. With careful monitoring of spironolactone, the hyperkalemia is manageable. But the perception is that it’s bad, and along with gynecomastia it’s a real killer.”
While some dismiss gynecomastia as a major concern (for men) with spironolactone treatment, “if medical students learn one thing about spironolactone, it’s that it can cause gynecomastia,” adding to the negative image that the approved MRAs carry, Dr. Bhatt said.
“The hyperkalemia was manageable. This is very important because of past problems with potassium when using spironolactone,” Dr. Filippatos said. Finerenone also looks “more cardiorenal protective” than the steroidal MRAs, exerting renal benefits in FIDELIO-DKD never previously described for a steroidal MRA.
Some of the uncertainty about the efficacy of finerenone in patients with a history of cardiovascular disease will lift when results are available in about another year from the FIGARO-DKD pivotal trial of finerenone, which enrolled more than 7,000 patients with T2D and CKD (entry criteria very similar to FIDELIO-CKD). A big difference is that FIGARO-DKD has a composite CVD event metric as its primary endpoint, and includes hospitalization for heart failure as one facet of the composite.
FIDELIO-DKD was sponsored by Bayer. Dr. Filippatos has been a lecturer on behalf of, served as a researcher for, or both for Bayer and also for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Bayer and also from several other companies, and he also is an adviser to several companies. Dr. Wanner has received honoraria from Bayer, and also from AstraZeneca, Boehringer Ingelheim, FMC, Gilead, GlaxoSmithKline, Lilly, and Merck.
Finerenone, the first nonsteroidal mineralocorticoid receptor antagonist to complete a phase 3 trial, showed cardiovascular benefits in patients with type 2 diabetes and chronic kidney disease, regardless of whether they entered the study with a history of cardiovascular disease, in follow-up analyses of the FIDELIO-DKD trial, which included 5,674 patients.
“Finerenone demonstrated benefits for primary and secondary cardiovascular disease protection,” said Gerasimos Filippatos, MD, at the American Heart Association scientific sessions. Finerenone treatment cut the rate of cardiovascular death, nonfatal MI or stroke, or heart failure hospitalization, when compared with placebo, by a relative 15% among patients with a history of cardiovascular disease (CVD), and by a relative 14% in patients without this history, differences that met a statistical test for consistency. But the absolute, drug-associated increments in benefit over placebo differed between the two CVD subgroups because of a sharp underlying difference in event rates.
In contrast, the analyses reported by Dr. Filippatos and associates from the FIDELIO-DKD study showed significant heterogeneity based on the presence or absence of CVD for the study’s primary endpoint, a composite renal metric that tallied the combined rate of death from renal causes, renal failure, or a sustained drop in estimated glomerular filtration rate of at least 40%. Researchers enrolled patients into FIDELIO-DKD based on having type 2 diabetes (T2D) and chronic kidney disease (CKD). The prevalence of a history of CVD was 46%.
Among patients with a history of CVD, the composite adverse CVD outcome occurred at a rate of 8.5/100 patient-years in patients on placebo and in 7.18/100 patients years among those on finerenone during a median of 2.6 years of follow-up, a 1.32/100–patient-year absolute between-group difference. Among patients in a primary prevention setting, incident CVD event rates during follow-up were roughly half that in the secondary prevention patients. The upshot was that, in the placebo group, the rate was 3.92/100 patient- years, and in those on finerenone was 3.43/100 patient-years, a 0.49/100–patient-year absolute difference.
CVD history produced heterogeneity for the primary endpoint
In the analysis that focused on the study’s primary, renal endpoint, among patients identified as having CVD at study entry, the outcome occurred at a rate of 9.06/100 patient-years in the placebo subgroup and at a rate of 6.6/100 patient years in those who received finerenone, a significant 30% relative risk reduction and an absolute between-group difference of 2.46/100 patient-years.
In contrast, among patients without a CVD history, the composite renal endpoint occurred at a rate of 9.1/100 patient-years in the placebo patients and 8.42/100 patient-years in those on finerenone, a 6% relative risk reduction that was not significant, and a 0.68/100–patient-year absolute difference. This disparity in the primary event rate between the two treatment arms reached statistical significance (P = .016), the investigators reported in the published version of the report in Circulation that simultaneously appeared online.
“The totality of evidence suggests that finerenone could be used in patients with T2D with or without a history of CVD,” explained Dr. Filippatos in an interview. “The P-interaction for the composite kidney outcome is significant, but it is not corrected for multiple testing; therefore, it might be a false-chance finding and must be interpreted cautiously.
Furthermore, in another prespecified kidney composite outcome the results were consistent in patients with and without a history of CVD. In sum, all the FIDELIO-DKD analyses so far are “suggestive of a beneficial effect in patients without a history of CVD.”
Despite these patients receiving guideline directed therapies, “there remains a high unmet medical need in patients with T2D and CKD,” added Dr. Filippatos, professor of cardiology at the University of Athens. “We use multiple treatments for patients with heart failure, and we should use the same mindset for treating patients with T2D and CKD. The costs of dialysis and kidney transplant are very high, so it is important to consider options that slow progression of CKD in these patients.”
In FIDELIO-DKD, virtually all patients were on background therapy with a renin-angiotensin-system (RAS) inhibitor, so the trial’s results suggest that treatment should at least involve dual therapy with finerenone and a RAS inhibitor. Fewer than 5% were on background therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor, a drug class recently established as another key agent for treating CKD in patients with T2D, setting up the prospect for triple therapy, although this approach has not yet undergone prospective testing.
Combining RAS inhibition, finerenone, and an SGLT2 inhibitor is “potentially a marriage made in diabetes heaven,” commented Deepak L. Bhatt, MD, a professor of medicine at Harvard Medical School, Boston, who has not participated in finerenone studies.
Finerenone looks better for safety
Regardless of subgroup analyses based on history of CVD, the findings from all patients enrolled in FIDELIO-DKD were positive for the both the primary renal outcome and key secondary outcome of composite CVD events. In the total randomized cohort, treatment with finerenone on top of optimized treatment with an ACE inhibitor or angiotensin receptor blocker (RAS inhibition) led to a significant 18% relative risk reduction, compared with placebo, for the primary renal endpoint, and a significant 14% relative drop in the key secondary CVD outcome. Those results were published in October in the New England Journal of Medicine.
For treating patients with T2D and CKD ,finerenone overall “looks like a major advance,” Dr. Bhatt said in an interview.
In addition to the positive efficacy results, several experts also focused on what they saw as superior safety of finerenone in the trial, compared with the historical safety of the steroidal mineralocorticoid receptor antagonists (MRAs) now in use: spironolactone and eplerenone.
“I’m a big believer in spironolactone, but it has issues with side effects, and eplerenone never seemed to catch on,” said Dr. Bhatt, who is also executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
“A lot of physicians like these MRAs, but acknowledge that side effects have kept these drugs from being used to the extent they should.” The existing MRAs, especially spironolactone, have become a key drug class for treating heart failure with reduced ejection fraction (and, some claim, for also treating heart failure with preserved ejection fraction), as well as treatment-resistant hypertension and primary aldosteronism. By design, FIDELIO-DKD did not enroll patients with heart failure because treatment with an MRA is indicated for those with heart failure with reduced ejection fraction.
The spironolactone adverse effect that generates the greatest concern is hyperkalemia. During his discussion of FIDELIO-DKD as designated discussant, Christoph Wanner, MD, noted a recent study in which the incidence of hyperkalemia severe enough to cause study discontinuation was 23% among patients treated with spironolactone for heart failure, which contrasts with the 2.3% rate in FIDELIO-DKD among finerenone recipients. This hyperkalemia incidence from finerenone also improved on the historical performance of other drugs, like aliskiren (Tekturna), said Dr. Wanner, professor and head of nephrology at the University of Würzburg (Germany).
The FIDELIO-DKD results place finerenone alongside the RAS- and SGLT2-inhibitor drug classes as appropriate treatments for most patients with T2D and CKD. “We have entered a new era of effective treatment for diabetic kidney disease,” Dr. Wanner declared.
“The overall safety profile of finerenone looked better, including hyperkalemia,” said Dr. Bhatt. “Hyperkalemia with spironolactone is not necessarily as bad as the perception. With careful monitoring of spironolactone, the hyperkalemia is manageable. But the perception is that it’s bad, and along with gynecomastia it’s a real killer.”
While some dismiss gynecomastia as a major concern (for men) with spironolactone treatment, “if medical students learn one thing about spironolactone, it’s that it can cause gynecomastia,” adding to the negative image that the approved MRAs carry, Dr. Bhatt said.
“The hyperkalemia was manageable. This is very important because of past problems with potassium when using spironolactone,” Dr. Filippatos said. Finerenone also looks “more cardiorenal protective” than the steroidal MRAs, exerting renal benefits in FIDELIO-DKD never previously described for a steroidal MRA.
Some of the uncertainty about the efficacy of finerenone in patients with a history of cardiovascular disease will lift when results are available in about another year from the FIGARO-DKD pivotal trial of finerenone, which enrolled more than 7,000 patients with T2D and CKD (entry criteria very similar to FIDELIO-CKD). A big difference is that FIGARO-DKD has a composite CVD event metric as its primary endpoint, and includes hospitalization for heart failure as one facet of the composite.
FIDELIO-DKD was sponsored by Bayer. Dr. Filippatos has been a lecturer on behalf of, served as a researcher for, or both for Bayer and also for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Bayer and also from several other companies, and he also is an adviser to several companies. Dr. Wanner has received honoraria from Bayer, and also from AstraZeneca, Boehringer Ingelheim, FMC, Gilead, GlaxoSmithKline, Lilly, and Merck.
Finerenone, the first nonsteroidal mineralocorticoid receptor antagonist to complete a phase 3 trial, showed cardiovascular benefits in patients with type 2 diabetes and chronic kidney disease, regardless of whether they entered the study with a history of cardiovascular disease, in follow-up analyses of the FIDELIO-DKD trial, which included 5,674 patients.
“Finerenone demonstrated benefits for primary and secondary cardiovascular disease protection,” said Gerasimos Filippatos, MD, at the American Heart Association scientific sessions. Finerenone treatment cut the rate of cardiovascular death, nonfatal MI or stroke, or heart failure hospitalization, when compared with placebo, by a relative 15% among patients with a history of cardiovascular disease (CVD), and by a relative 14% in patients without this history, differences that met a statistical test for consistency. But the absolute, drug-associated increments in benefit over placebo differed between the two CVD subgroups because of a sharp underlying difference in event rates.
In contrast, the analyses reported by Dr. Filippatos and associates from the FIDELIO-DKD study showed significant heterogeneity based on the presence or absence of CVD for the study’s primary endpoint, a composite renal metric that tallied the combined rate of death from renal causes, renal failure, or a sustained drop in estimated glomerular filtration rate of at least 40%. Researchers enrolled patients into FIDELIO-DKD based on having type 2 diabetes (T2D) and chronic kidney disease (CKD). The prevalence of a history of CVD was 46%.
Among patients with a history of CVD, the composite adverse CVD outcome occurred at a rate of 8.5/100 patient-years in patients on placebo and in 7.18/100 patients years among those on finerenone during a median of 2.6 years of follow-up, a 1.32/100–patient-year absolute between-group difference. Among patients in a primary prevention setting, incident CVD event rates during follow-up were roughly half that in the secondary prevention patients. The upshot was that, in the placebo group, the rate was 3.92/100 patient- years, and in those on finerenone was 3.43/100 patient-years, a 0.49/100–patient-year absolute difference.
CVD history produced heterogeneity for the primary endpoint
In the analysis that focused on the study’s primary, renal endpoint, among patients identified as having CVD at study entry, the outcome occurred at a rate of 9.06/100 patient-years in the placebo subgroup and at a rate of 6.6/100 patient years in those who received finerenone, a significant 30% relative risk reduction and an absolute between-group difference of 2.46/100 patient-years.
In contrast, among patients without a CVD history, the composite renal endpoint occurred at a rate of 9.1/100 patient-years in the placebo patients and 8.42/100 patient-years in those on finerenone, a 6% relative risk reduction that was not significant, and a 0.68/100–patient-year absolute difference. This disparity in the primary event rate between the two treatment arms reached statistical significance (P = .016), the investigators reported in the published version of the report in Circulation that simultaneously appeared online.
“The totality of evidence suggests that finerenone could be used in patients with T2D with or without a history of CVD,” explained Dr. Filippatos in an interview. “The P-interaction for the composite kidney outcome is significant, but it is not corrected for multiple testing; therefore, it might be a false-chance finding and must be interpreted cautiously.
Furthermore, in another prespecified kidney composite outcome the results were consistent in patients with and without a history of CVD. In sum, all the FIDELIO-DKD analyses so far are “suggestive of a beneficial effect in patients without a history of CVD.”
Despite these patients receiving guideline directed therapies, “there remains a high unmet medical need in patients with T2D and CKD,” added Dr. Filippatos, professor of cardiology at the University of Athens. “We use multiple treatments for patients with heart failure, and we should use the same mindset for treating patients with T2D and CKD. The costs of dialysis and kidney transplant are very high, so it is important to consider options that slow progression of CKD in these patients.”
In FIDELIO-DKD, virtually all patients were on background therapy with a renin-angiotensin-system (RAS) inhibitor, so the trial’s results suggest that treatment should at least involve dual therapy with finerenone and a RAS inhibitor. Fewer than 5% were on background therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor, a drug class recently established as another key agent for treating CKD in patients with T2D, setting up the prospect for triple therapy, although this approach has not yet undergone prospective testing.
Combining RAS inhibition, finerenone, and an SGLT2 inhibitor is “potentially a marriage made in diabetes heaven,” commented Deepak L. Bhatt, MD, a professor of medicine at Harvard Medical School, Boston, who has not participated in finerenone studies.
Finerenone looks better for safety
Regardless of subgroup analyses based on history of CVD, the findings from all patients enrolled in FIDELIO-DKD were positive for the both the primary renal outcome and key secondary outcome of composite CVD events. In the total randomized cohort, treatment with finerenone on top of optimized treatment with an ACE inhibitor or angiotensin receptor blocker (RAS inhibition) led to a significant 18% relative risk reduction, compared with placebo, for the primary renal endpoint, and a significant 14% relative drop in the key secondary CVD outcome. Those results were published in October in the New England Journal of Medicine.
For treating patients with T2D and CKD ,finerenone overall “looks like a major advance,” Dr. Bhatt said in an interview.
In addition to the positive efficacy results, several experts also focused on what they saw as superior safety of finerenone in the trial, compared with the historical safety of the steroidal mineralocorticoid receptor antagonists (MRAs) now in use: spironolactone and eplerenone.
“I’m a big believer in spironolactone, but it has issues with side effects, and eplerenone never seemed to catch on,” said Dr. Bhatt, who is also executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
“A lot of physicians like these MRAs, but acknowledge that side effects have kept these drugs from being used to the extent they should.” The existing MRAs, especially spironolactone, have become a key drug class for treating heart failure with reduced ejection fraction (and, some claim, for also treating heart failure with preserved ejection fraction), as well as treatment-resistant hypertension and primary aldosteronism. By design, FIDELIO-DKD did not enroll patients with heart failure because treatment with an MRA is indicated for those with heart failure with reduced ejection fraction.
The spironolactone adverse effect that generates the greatest concern is hyperkalemia. During his discussion of FIDELIO-DKD as designated discussant, Christoph Wanner, MD, noted a recent study in which the incidence of hyperkalemia severe enough to cause study discontinuation was 23% among patients treated with spironolactone for heart failure, which contrasts with the 2.3% rate in FIDELIO-DKD among finerenone recipients. This hyperkalemia incidence from finerenone also improved on the historical performance of other drugs, like aliskiren (Tekturna), said Dr. Wanner, professor and head of nephrology at the University of Würzburg (Germany).
The FIDELIO-DKD results place finerenone alongside the RAS- and SGLT2-inhibitor drug classes as appropriate treatments for most patients with T2D and CKD. “We have entered a new era of effective treatment for diabetic kidney disease,” Dr. Wanner declared.
“The overall safety profile of finerenone looked better, including hyperkalemia,” said Dr. Bhatt. “Hyperkalemia with spironolactone is not necessarily as bad as the perception. With careful monitoring of spironolactone, the hyperkalemia is manageable. But the perception is that it’s bad, and along with gynecomastia it’s a real killer.”
While some dismiss gynecomastia as a major concern (for men) with spironolactone treatment, “if medical students learn one thing about spironolactone, it’s that it can cause gynecomastia,” adding to the negative image that the approved MRAs carry, Dr. Bhatt said.
“The hyperkalemia was manageable. This is very important because of past problems with potassium when using spironolactone,” Dr. Filippatos said. Finerenone also looks “more cardiorenal protective” than the steroidal MRAs, exerting renal benefits in FIDELIO-DKD never previously described for a steroidal MRA.
Some of the uncertainty about the efficacy of finerenone in patients with a history of cardiovascular disease will lift when results are available in about another year from the FIGARO-DKD pivotal trial of finerenone, which enrolled more than 7,000 patients with T2D and CKD (entry criteria very similar to FIDELIO-CKD). A big difference is that FIGARO-DKD has a composite CVD event metric as its primary endpoint, and includes hospitalization for heart failure as one facet of the composite.
FIDELIO-DKD was sponsored by Bayer. Dr. Filippatos has been a lecturer on behalf of, served as a researcher for, or both for Bayer and also for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Bayer and also from several other companies, and he also is an adviser to several companies. Dr. Wanner has received honoraria from Bayer, and also from AstraZeneca, Boehringer Ingelheim, FMC, Gilead, GlaxoSmithKline, Lilly, and Merck.
FROM AHA 2020
Circulating miRNA could be a potential biomarker for early diagnosis of MM
Circulating microRNAs (miRNAs) could be a potential noninvasive biomarker for early diagnosis of multiple myeloma (MM), according to the results of a meta-analysis published online in the Journal of Bone Oncology.
In recent years, because of the rise of the miRNA technique, many scholars have studied its value in the diagnosis of MM, and have obtained good but inconsistent results, according to Shuai-Shuai Gao, of the Xi’an (China) Daxing Hospital, and colleagues. For this reason, they conducted the meta-analysis in order to more clearly determine the role of miRNA in the early diagnosis of MM. The meta-analysis ultimately assessed 32 studies from 15 articles comprising 2,053 MM patients and 1,118 healthy controls.
All the included studies involved newly diagnosed MM patients and healthy controls; the obtained miRNA was derived from serum or plasma specimens; and the report contained relevant statistics such as sensitivity, specificity, and area-under-the-curve values.
High sensitivity and specificity
The researchers found that the overall sensitivity and specificity of using miRNAs for the diagnosis of MM were 0.81 and 0.85, respectively. In addition, the overall positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the curve were 5.5, 0.22, 25 and 0.90, respectively.
A subgroup analysis showed that the down-regulation of miRNA clusters with larger samples size of plasma type could carry out a better diagnostic accuracy of MM patients, according to the authors.
“[Circulating miRNAs] not only had high sensitivity and strong specificity, but also had noninvasive and no radiation risks. It is worth continuing to optimize its practicality. In the future, multicenter, more rigorous, and high-quality case-control studies are still needed in clinical practice to improve the efficacy of circulating miRNA in the early diagnosis of MM,” the researchers concluded.
The study did not receive any outside funding and the researchers reported that they had no conflicts.
SOURCE: Gao S-S et al. J Bone Oncol. 2020 Oct 21. doi: 10.1016/j.jbo.2020.100327.
Circulating microRNAs (miRNAs) could be a potential noninvasive biomarker for early diagnosis of multiple myeloma (MM), according to the results of a meta-analysis published online in the Journal of Bone Oncology.
In recent years, because of the rise of the miRNA technique, many scholars have studied its value in the diagnosis of MM, and have obtained good but inconsistent results, according to Shuai-Shuai Gao, of the Xi’an (China) Daxing Hospital, and colleagues. For this reason, they conducted the meta-analysis in order to more clearly determine the role of miRNA in the early diagnosis of MM. The meta-analysis ultimately assessed 32 studies from 15 articles comprising 2,053 MM patients and 1,118 healthy controls.
All the included studies involved newly diagnosed MM patients and healthy controls; the obtained miRNA was derived from serum or plasma specimens; and the report contained relevant statistics such as sensitivity, specificity, and area-under-the-curve values.
High sensitivity and specificity
The researchers found that the overall sensitivity and specificity of using miRNAs for the diagnosis of MM were 0.81 and 0.85, respectively. In addition, the overall positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the curve were 5.5, 0.22, 25 and 0.90, respectively.
A subgroup analysis showed that the down-regulation of miRNA clusters with larger samples size of plasma type could carry out a better diagnostic accuracy of MM patients, according to the authors.
“[Circulating miRNAs] not only had high sensitivity and strong specificity, but also had noninvasive and no radiation risks. It is worth continuing to optimize its practicality. In the future, multicenter, more rigorous, and high-quality case-control studies are still needed in clinical practice to improve the efficacy of circulating miRNA in the early diagnosis of MM,” the researchers concluded.
The study did not receive any outside funding and the researchers reported that they had no conflicts.
SOURCE: Gao S-S et al. J Bone Oncol. 2020 Oct 21. doi: 10.1016/j.jbo.2020.100327.
Circulating microRNAs (miRNAs) could be a potential noninvasive biomarker for early diagnosis of multiple myeloma (MM), according to the results of a meta-analysis published online in the Journal of Bone Oncology.
In recent years, because of the rise of the miRNA technique, many scholars have studied its value in the diagnosis of MM, and have obtained good but inconsistent results, according to Shuai-Shuai Gao, of the Xi’an (China) Daxing Hospital, and colleagues. For this reason, they conducted the meta-analysis in order to more clearly determine the role of miRNA in the early diagnosis of MM. The meta-analysis ultimately assessed 32 studies from 15 articles comprising 2,053 MM patients and 1,118 healthy controls.
All the included studies involved newly diagnosed MM patients and healthy controls; the obtained miRNA was derived from serum or plasma specimens; and the report contained relevant statistics such as sensitivity, specificity, and area-under-the-curve values.
High sensitivity and specificity
The researchers found that the overall sensitivity and specificity of using miRNAs for the diagnosis of MM were 0.81 and 0.85, respectively. In addition, the overall positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the curve were 5.5, 0.22, 25 and 0.90, respectively.
A subgroup analysis showed that the down-regulation of miRNA clusters with larger samples size of plasma type could carry out a better diagnostic accuracy of MM patients, according to the authors.
“[Circulating miRNAs] not only had high sensitivity and strong specificity, but also had noninvasive and no radiation risks. It is worth continuing to optimize its practicality. In the future, multicenter, more rigorous, and high-quality case-control studies are still needed in clinical practice to improve the efficacy of circulating miRNA in the early diagnosis of MM,” the researchers concluded.
The study did not receive any outside funding and the researchers reported that they had no conflicts.
SOURCE: Gao S-S et al. J Bone Oncol. 2020 Oct 21. doi: 10.1016/j.jbo.2020.100327.
FROM THE JOURNAL OF BONE ONCOLOGY
Sharing mental health notes with patients: Differentiating between access and accessibility
As a result of technology, data to which individuals have long had a legal right of access are now easily accessible. The advent of secure online portals has converted what was once largely a hypothetical proposition – i.e., ready patient access to clinical records – into the increasing reality of open notes. Unsurprisingly, this movement toward greater transparency carries special implications and complexities for mental health professionals concerned with balancing patient autonomy and safety.1
In setting clinician expectations for incorporating clinical note-sharing into patient care, it is important to clarify the already robust legal rights of patients to obtain copies of their treatment records, including in the mental health context. For example, the provision of access to one’s own health information is one of the rare circumstances in which the HIPAA Privacy Rule compels, rather than merely permits, health care organizations to disclose records to external parties.2 This requirement accords with a prevailing principle of medical ethics that records should be furnished to patients or their designees upon request.3
While HIPAA provides mechanisms for denying individuals access to their health records when a licensed health care professional determines that such access is “reasonably likely” to jeopardize someone’s life or physical safety, this standard sets a high bar of physical endangerment. A psychiatrist or other physician’s concern that sharing clinical notes would be therapeutically suboptimal, discomforting, emotionally harmful, confusing, or deleterious to the doctor-patient relationship would not constitute sufficient legal grounds for denial of access.4
Moreover, because access is framed as a patient right, denial on the aforementioned safety grounds implicates due process requirements to include a written explanation and appellate review by a designated disinterested official. Importantly, this federally mandated right of access would preempt or override any contrary state law classified as less protective of patient prerogatives.
Contrary to popular misconception, mental health notes are not necessarily entitled to a greater degree of secrecy vis-à-vis patients than records pertaining to physical health. The HIPAA Privacy Rule, for example, generally does not distinguish between medical and mental health information, nor does it provide special rules for the latter.5 Specifically, patients’ access rights remain unchanged regardless of whether the requested record is classified as medical or psychiatric.
One narrow exception involves “psychotherapy notes,” but the legal definition of this term is considerably more restrictive than in common psychiatric usage. Psychotherapy notes under health privacy law include therapists’ private, desk-drawer notes on counseling sessions intended for personal use as memory joggers. They explicitly exclude any discussion of treatment modalities, diagnosis, or clinical progress. To retain their protected status, psychotherapy notes must be kept separate and apart from the patient chart. Indeed, a primary reason why such notes are exempted from patient access is because they have so little legitimate use other than to their creator.
Integrating the proliferation of open notes into practice regimens will likely pose a continuing and growing challenge for mental health professionals. From a medicolegal perspective, however, even psychiatrists who are understandably hesitant about widespread note-sharing would do well to remember that patients’ underlying right of access is generally well established, and is merely being operationalized and automated via web portals. Ultimately, adapting clinical record-keeping to both anticipate patient access – and, where possible, leverage it to improve care – may prove the most effective path forward for practicing psychiatry in the digital age.
Lt. Col. Kels teaches at the U.S. Army Medical Center of Excellence, and Dr. Kels teaches and practices psychiatry at the University of the Incarnate Word School of Osteopathic Medicine, both in San Antonio, Tex. Neither Lt. Col. Kels nor Dr. Kels have any conflicts of interest to disclose. Opinions expressed in this article are those of the authors alone and do not necessarily reflect those of any government agency.
References
1. Lancet Psychiatry. 2020;7(11):924-5.
2. 45 CFR Parts 160 and 164, subparts A and E.
3. American Medical Association. Code of medical ethics, opinion 3.3.1: management of medical records.
4. Department of Health & Human Services. Individuals’ right under HIPAA to access their health information.
5. Department of Health & Human Services. HIPAA privacy rule and sharing information related to mental health.
As a result of technology, data to which individuals have long had a legal right of access are now easily accessible. The advent of secure online portals has converted what was once largely a hypothetical proposition – i.e., ready patient access to clinical records – into the increasing reality of open notes. Unsurprisingly, this movement toward greater transparency carries special implications and complexities for mental health professionals concerned with balancing patient autonomy and safety.1
In setting clinician expectations for incorporating clinical note-sharing into patient care, it is important to clarify the already robust legal rights of patients to obtain copies of their treatment records, including in the mental health context. For example, the provision of access to one’s own health information is one of the rare circumstances in which the HIPAA Privacy Rule compels, rather than merely permits, health care organizations to disclose records to external parties.2 This requirement accords with a prevailing principle of medical ethics that records should be furnished to patients or their designees upon request.3
While HIPAA provides mechanisms for denying individuals access to their health records when a licensed health care professional determines that such access is “reasonably likely” to jeopardize someone’s life or physical safety, this standard sets a high bar of physical endangerment. A psychiatrist or other physician’s concern that sharing clinical notes would be therapeutically suboptimal, discomforting, emotionally harmful, confusing, or deleterious to the doctor-patient relationship would not constitute sufficient legal grounds for denial of access.4
Moreover, because access is framed as a patient right, denial on the aforementioned safety grounds implicates due process requirements to include a written explanation and appellate review by a designated disinterested official. Importantly, this federally mandated right of access would preempt or override any contrary state law classified as less protective of patient prerogatives.
Contrary to popular misconception, mental health notes are not necessarily entitled to a greater degree of secrecy vis-à-vis patients than records pertaining to physical health. The HIPAA Privacy Rule, for example, generally does not distinguish between medical and mental health information, nor does it provide special rules for the latter.5 Specifically, patients’ access rights remain unchanged regardless of whether the requested record is classified as medical or psychiatric.
One narrow exception involves “psychotherapy notes,” but the legal definition of this term is considerably more restrictive than in common psychiatric usage. Psychotherapy notes under health privacy law include therapists’ private, desk-drawer notes on counseling sessions intended for personal use as memory joggers. They explicitly exclude any discussion of treatment modalities, diagnosis, or clinical progress. To retain their protected status, psychotherapy notes must be kept separate and apart from the patient chart. Indeed, a primary reason why such notes are exempted from patient access is because they have so little legitimate use other than to their creator.
Integrating the proliferation of open notes into practice regimens will likely pose a continuing and growing challenge for mental health professionals. From a medicolegal perspective, however, even psychiatrists who are understandably hesitant about widespread note-sharing would do well to remember that patients’ underlying right of access is generally well established, and is merely being operationalized and automated via web portals. Ultimately, adapting clinical record-keeping to both anticipate patient access – and, where possible, leverage it to improve care – may prove the most effective path forward for practicing psychiatry in the digital age.
Lt. Col. Kels teaches at the U.S. Army Medical Center of Excellence, and Dr. Kels teaches and practices psychiatry at the University of the Incarnate Word School of Osteopathic Medicine, both in San Antonio, Tex. Neither Lt. Col. Kels nor Dr. Kels have any conflicts of interest to disclose. Opinions expressed in this article are those of the authors alone and do not necessarily reflect those of any government agency.
References
1. Lancet Psychiatry. 2020;7(11):924-5.
2. 45 CFR Parts 160 and 164, subparts A and E.
3. American Medical Association. Code of medical ethics, opinion 3.3.1: management of medical records.
4. Department of Health & Human Services. Individuals’ right under HIPAA to access their health information.
5. Department of Health & Human Services. HIPAA privacy rule and sharing information related to mental health.
As a result of technology, data to which individuals have long had a legal right of access are now easily accessible. The advent of secure online portals has converted what was once largely a hypothetical proposition – i.e., ready patient access to clinical records – into the increasing reality of open notes. Unsurprisingly, this movement toward greater transparency carries special implications and complexities for mental health professionals concerned with balancing patient autonomy and safety.1
In setting clinician expectations for incorporating clinical note-sharing into patient care, it is important to clarify the already robust legal rights of patients to obtain copies of their treatment records, including in the mental health context. For example, the provision of access to one’s own health information is one of the rare circumstances in which the HIPAA Privacy Rule compels, rather than merely permits, health care organizations to disclose records to external parties.2 This requirement accords with a prevailing principle of medical ethics that records should be furnished to patients or their designees upon request.3
While HIPAA provides mechanisms for denying individuals access to their health records when a licensed health care professional determines that such access is “reasonably likely” to jeopardize someone’s life or physical safety, this standard sets a high bar of physical endangerment. A psychiatrist or other physician’s concern that sharing clinical notes would be therapeutically suboptimal, discomforting, emotionally harmful, confusing, or deleterious to the doctor-patient relationship would not constitute sufficient legal grounds for denial of access.4
Moreover, because access is framed as a patient right, denial on the aforementioned safety grounds implicates due process requirements to include a written explanation and appellate review by a designated disinterested official. Importantly, this federally mandated right of access would preempt or override any contrary state law classified as less protective of patient prerogatives.
Contrary to popular misconception, mental health notes are not necessarily entitled to a greater degree of secrecy vis-à-vis patients than records pertaining to physical health. The HIPAA Privacy Rule, for example, generally does not distinguish between medical and mental health information, nor does it provide special rules for the latter.5 Specifically, patients’ access rights remain unchanged regardless of whether the requested record is classified as medical or psychiatric.
One narrow exception involves “psychotherapy notes,” but the legal definition of this term is considerably more restrictive than in common psychiatric usage. Psychotherapy notes under health privacy law include therapists’ private, desk-drawer notes on counseling sessions intended for personal use as memory joggers. They explicitly exclude any discussion of treatment modalities, diagnosis, or clinical progress. To retain their protected status, psychotherapy notes must be kept separate and apart from the patient chart. Indeed, a primary reason why such notes are exempted from patient access is because they have so little legitimate use other than to their creator.
Integrating the proliferation of open notes into practice regimens will likely pose a continuing and growing challenge for mental health professionals. From a medicolegal perspective, however, even psychiatrists who are understandably hesitant about widespread note-sharing would do well to remember that patients’ underlying right of access is generally well established, and is merely being operationalized and automated via web portals. Ultimately, adapting clinical record-keeping to both anticipate patient access – and, where possible, leverage it to improve care – may prove the most effective path forward for practicing psychiatry in the digital age.
Lt. Col. Kels teaches at the U.S. Army Medical Center of Excellence, and Dr. Kels teaches and practices psychiatry at the University of the Incarnate Word School of Osteopathic Medicine, both in San Antonio, Tex. Neither Lt. Col. Kels nor Dr. Kels have any conflicts of interest to disclose. Opinions expressed in this article are those of the authors alone and do not necessarily reflect those of any government agency.
References
1. Lancet Psychiatry. 2020;7(11):924-5.
2. 45 CFR Parts 160 and 164, subparts A and E.
3. American Medical Association. Code of medical ethics, opinion 3.3.1: management of medical records.
4. Department of Health & Human Services. Individuals’ right under HIPAA to access their health information.
5. Department of Health & Human Services. HIPAA privacy rule and sharing information related to mental health.
Equitable Post-COVID-19 Care: A Practical Framework to Integrate Health Equity in Diabetes Management
From T1D Exchange, Boston, MA (Dr. Ebekozien, Dr. Odugbesan, and Nicole Rioles); Barbara Davis Center, University of Colorado, Boulder, CO (Dr. Majidi); Cincinnati Children’s Hospital Medical Center, Cincinnati, OH (Dr. Jones); and Nationwide Children’s Hospital, Columbus, OH (Dr. Kamboj)
Health equity has been described as the opportunity for all persons to obtain their highest level of health possible.1 Unfortunately, even with advances in technology and care practices, disparities persist in health care outcomes. Disparities in prevalence, prognosis, and outcomes still exist in diabetes management.2 Non-Hispanic Black and/or Hispanic populations are more likely to have worse glycemic control,3,4 to encounter more barriers in access to care,5 and to have higher levels of acute complications,4 and to use advanced technologies less frequently.4 Diabetes is one of the preexisting conditions that increase morbidity and mortality in COVID-19.6,7 Unfortunately, adverse outcomes from COVID-19 also disproportionately impact a specific vulnerable population.8,9 The urgent transition to managing diabetes remotely during the COVID-19 pandemic may exacerbate long-term inequities because some vulnerable patients might not have access to technology devices necessary for effective remote management.
Here, we describe how quality improvement (QI) tools and principles can be adapted into a framework for advancing health equity. Specifically, we describe a 10-step framework that may be applied in diabetes care management to achieve improvement, using a hypothetical example of increasing use of continuous glucose monitors (CGMs) among patients with type 1 diabetes mellitus.10 This framework was developed to address the literature gap on practical ways health care providers can address inequities using QI principles, and was implemented by 1 of the authors at a local public health department.11 The framework’s iterative and comprehensive design makes it ideal for addressing inequities in chronic diseases like diabetes, which have multiple root causes with no easy solutions. The improvement program pilot received a national model practice award.11,12
10-Step Framework
Step 1: Review program/project baseline data for existing disparities. Diabetes programs and routine QI processes encourage existing data review to determine how effective the current system is working and if the existing process has a predictable pattern.13,14 Our equity-revised framework proposes a more in-depth review to stratify baseline data based on factors that might contribute to inequities, including race, age, income levels, ethnicity, language, sexual orientation, insurance type, and zip code. This process will identify patients not served or unfairly impacted due to socioeconomic factors. For example, using the hypothetical example of improving CGM use, a team completes a preliminary data review and determines that baseline CGM use is 30% in the clinic population. However, in a review to assess for disparities, they also identify that patients on public insurance have a significantly lower CGM uptake of only 15%.
Step 2: Build an equitable project team, including patients with lived experiences. Routine projects typically have clinicians, administrative staff, and analytic staff as members of their team. In a post-COVID-19 world, every team needs to learn directly from people impacted and share decision-making power. The traditional approach to receiving feedback has generally been to collect responses using surveys or focus groups. We propose that individuals/families who are disproportionately impacted be included as active members on QI teams. For example, in the hypothetical example of the CGM project, team members would include patients with type 1 diabetes who are on public insurance and their families.
Step 3: Develop equity-focused goals. The traditional program involves the development of aims that are SMART (specific, measurable, achievable, realistic, time-bound).15 The proposed framework encourages the inclusion of equity-revised goals (SMARTer) using insights from Steps 1 and 2. For example, your typical smart goal might be to increase the percentage of patients using CGM by 20% in 6 months, while a SMARTer goal would be to increase the proportion of patients using CGM by 20% and reduce the disparities among public and private insurance patients by 30% in 6 months.
Step 4: Identify inequitable processes/pathways. Traditional QI programs might use a process map or flow diagram to depict the current state of a process visually.16 For example, in Figure 1, the process map diagram depicts some differences in the process for patients with public insurance as opposed to those with private insurance. The framework also advocates for using visual tools like process maps to depict how there might be inequitable pathways in a system. Visually identifying inequitable processes/pathways can help a team see barriers, address challenges, and pilot innovative solutions.
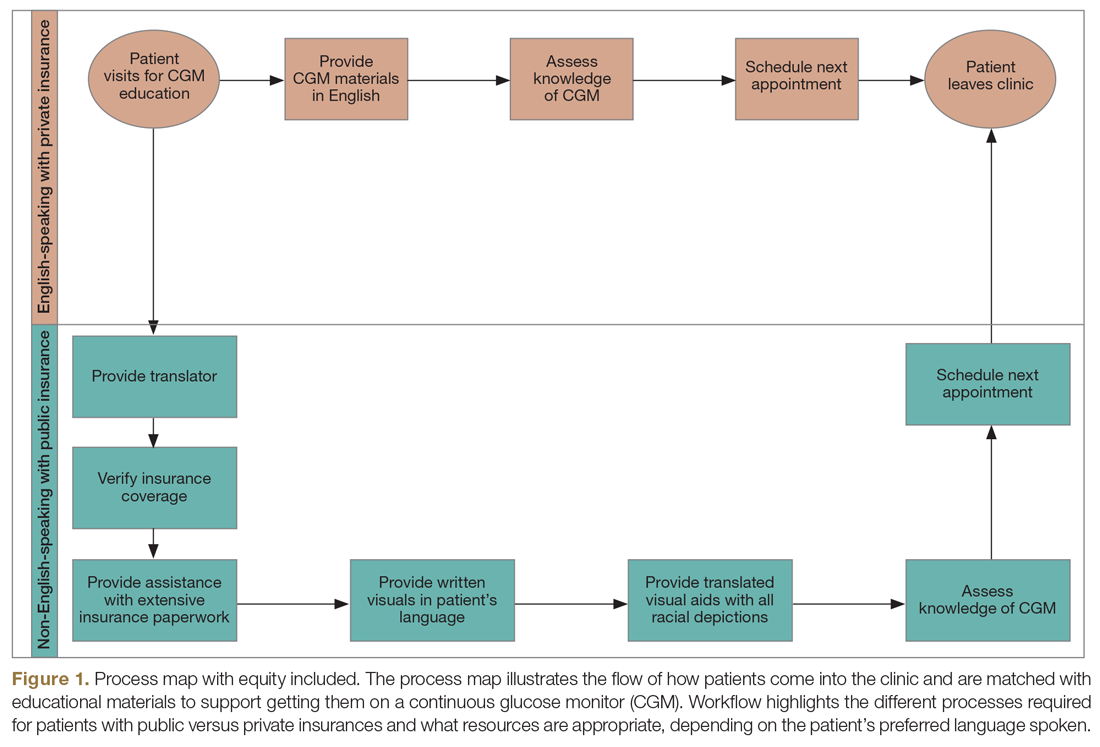
Step 5: Identify how socioeconomic factors are contributing to the current outcome. A good understanding of factors that contribute to the problem is an essential part of finding fundamental solutions. The fishbone diagram16 is a visualization tool used to identify contributing factors. When investigating contributing factors, it is commonplace to identify factors that fit into 1 of 5 categories: people, process, place, product, and policies (5 Ps). An equity-focused process will include equity as a new major factor category, and the socioeconomic impacts that contribute to inequities will be brainstormed and visually represented. For example, in the hypothetical CGM improvement example, an equity contributing factor is extensive CGM application paperwork for patients on public insurance as compared to those on private insurance. Figure 2 shows equity integrated into a fishbone diagram.
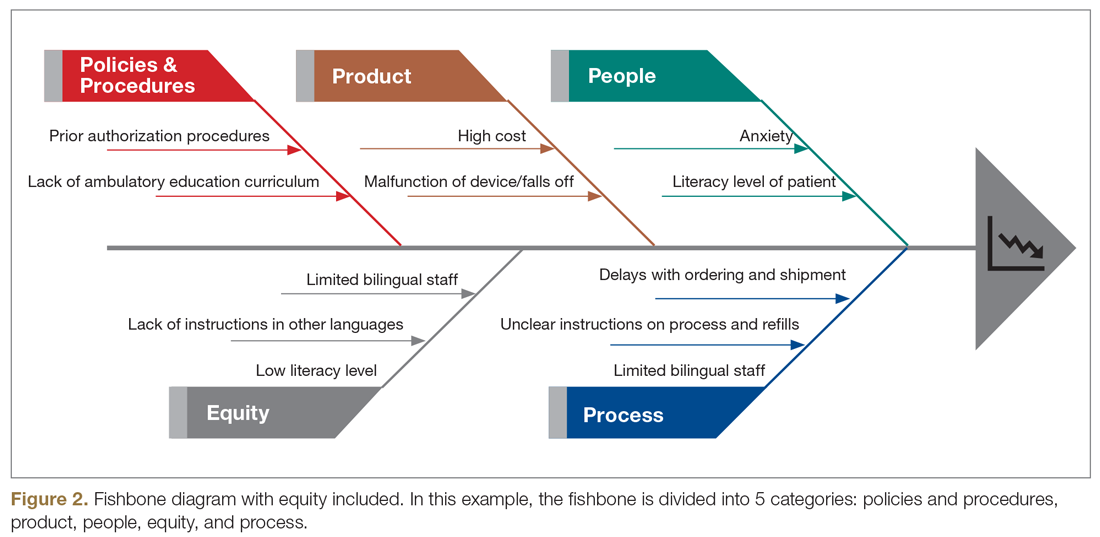
Step 6: Brainstorm possible improvements. Potential improvement ideas for the hypothetical CGM example might include redesigning the existing workflow, piloting CGM educational classes, and using a CGM barrier assessment tool to identify and address barriers to adoption.
Step 7: Use the decision matrix with equity as a criterion to prioritize improvement ideas. Decision matrix15 is a great tool that is frequently used to help teams prioritize potential ideas. Project team members must decide what criteria are important in prioritizing ideas to implement. Common criteria include implementation cost, time, and resources, but in addition to the common criteria, the team can specify ”impact on equity” as one of their criteria, alongside other standard criteria like impact.
Step 8: Test one small change at a time. This step is consistent with other traditional improvement models using the Plan, Do, Study, Act (PDSA) model for improvement.17 During this phase, the team should make predictions on the expected impact of the intervention on outcomes. For example, in the hypothetical example, the team predicts that testing and expanding CGM classes will reduce disparities among public versus private health insurance users by 5% and increase overall CGM uptake by 10%.
Step 9: Measure and compare results with predictions to identify inequitable practices or consequences. After each test of change, the team should review the results, including implementation cost considerations, and compare them to the predictions in the earlier step. The team should also document the potential reasons why their predictions were correct or inaccurate, and whether there were any unforeseen outcomes from the intervention.
Step 10: Celebrate small wins and repeat the process. Making fundamental and equitable changes takes time. This framework aimed at undoing inequities, particularly those inequities that have been amplified by the COVID-19 pandemic, is iterative and ongoing.18,19 Not every test of change will impact the outcome or reduce inequity, but over time, each change will impact the next, generating sustainable effects.
Conclusion
There are ongoing studies examining the adverse outcomes and potential health inequities for patients with diabetes impacted by COVID-19.20 Health care providers need to plan for post-COVID-19 care, keeping in mind that the pandemic might worsen already existing health disparities in diabetes management.3,4,21 This work will involve an intentional approach to address structural and systemic racism.22 Therefore, the work of building health equity solutions must be rooted in racial justice, economic equity, and equitable access to health care and education.
Initiatives like this are currently being funded through foundation grants as well as state and federal research or program grants. Regional and national payors, including the Centers for Medicare & Medicaid Services, are currently piloting long-term sustainable funding models through programs like accountable care organizations and the Accountable Health Communities Model.23
Health systems can successfully address health equity and racial justice, using a framework as described above, to identify determinants of health, develop policies to expand access to care for the most vulnerable patients, distribute decision-making power, and train staff by naming structural racism as a driver of health inequities.
Acknowledgment: The authors acknowledge the contributions of patients, families, diabetes care teams, and collaborators within the T1D Exchange Quality Improvement Collaborative, who continually seek to improve care and outcomes for people living with diabetes.
Corresponding author: Osagie Ebekozien, MD, 11 Avenue De La Fayette, Boston, MA 02115; [email protected].
Financial disclosures: None.
Funding: T1D Exchange QI Collaborative is funded by The Leona M. and Harry B. Helmsley Charitable Trust. No specific funding was received for this manuscript or the development of this framework.
Keywords: type 1 diabetes; quality improvement; QI framework; racial justice; health disparities.
1. American Public Health Association Health Equity web site. https://www.apha.org/topics-and-issues/health-equity. Accessed June 4, 2020.
2. Lado J, Lipman T. Racial and ethnic disparities in the incidence, treatment, and outcomes of youth with type 1 diabetes. Endocrinol Metab Clin North Am. 2016;45:453-461.
3. Kahkoska AR, Shay CM, Crandell J, et al. Association of race and ethnicity with glycemic control and hemoglobin A1c levels in youth with type 1 diabetes. JAMA Netw Open. 2018;1:e181851.
4. Willi SM, Miller KM, DiMeglio LA, et al; T1D Exchange Clinic Network. Racial-ethnic disparities in management and outcomes among children with type 1 diabetes. Pediatrics. 2015;135:424-434.
5. Valenzuela JM, Seid M, Waitzfelder B, et al. Prevalence of and disparities in barriers to care experienced by youth with type 1 diabetes. J Pediatr. 2014;164:1369-1375.
6. Hussain A, Bhowmik B, do Vale Moreira NC. COVID-19 and diabetes: Knowledge in progress. Diabetes Res Clin Pract. 2020;162:108142.
7. Bhatraju PK, Ghassemieh BJ, Nichols M, et al. Covid-19 in critically ill patients in the Seattle Region - case series. N Engl J Med. 2020;382:2012-2022.
8. Laurencin CT, McClinton A. The COVID-19 pandemic: a call to action to identify and address racial and ethnic disparities. J Racial Ethn Health Disparities. 2020;7:398-402.
9. Shah M, Sachdeva M, Dodiuk-Gad RP. COVID-19 and racial disparities. J Am Acad Dermatol. 2020;83:e35.
10. Ebekozien O, Rioles N, DeSalvo D, et al. Improving continuous glucose monitoring (CGM) use across national centers: results from the T1D Exchange Quality Improvement Collaborative (T1DX-QI). Diabetes. 2020;69(Supplement 1):145-LB.
11. Ebekozien O. QI methodology to address health equity. Presented at American Society of Quality BOSCON 2018; Boston, MA; March 19 and 20, 2018.
12. 2019 Model Practice Award, Building A Culture of Improvement. National Association of County and City Health Officials web site. www.naccho.org/membership/awards/model-practices. Accessed June 4, 2020.
13. Nuckols TK, Keeler E, Anderson LJ, et al. Economic evaluation of quality improvement interventions designed to improve glycemic control in diabetes: a systematic review and weighted regression analysis. Diabetes Care. 2018;41:985‐993.
14. Rossi MC, Nicolucci A, Arcangeli A, et al. Baseline quality-of-care data from a quality-improvement program implemented by a network of diabetes outpatient clinics. Diabetes Care. 2008;31:2166‐2168.
15. McQuillan RF, Silver SA, Harel Z, et al. How to measure and interpret quality improvement data. Clin J Am Soc Nephrol. 2016;11:908-914.
16. Siddiqi FS. Quality improvement in diabetes care: time for us to step up? Can J Diabetes. 2019;43:233.
17. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan-do-study-act method to improve quality in healthcare. BMJ Qual Saf. 2014;23:290‐298.
18. Ferdinand KC, Nasser SA. African American COVID-19 mortality: a sentinel event. J Am Coll Cardiol. 2020;75:2746-2748..
19. Muniyappa R, Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab. 2020;318:E736-E741.
20. Ebekozien OA, Noor N, Gallagher MP, Alonso GT. Type 1 diabetes and COVID-19: preliminary findings from a multicenter surveillance study in the U.S. Diabetes Care. 2020;43:e83-e85.
21. Majidi S, Ebekozien O, Noor N, et al. Inequities in health outcomes among patients in the T1D Exchange-QI Collaborative. Diabetes. 2020;69(Supplement 1):1220-P. https://doi.org/10.2337/ db20-1220.-P.
22. Williams DR, Mohammed SA. Discrimination and racial disparities in health: evidence and needed research. J Behav Med. 2009;32:20-47.
23. Centers for Medicare & Medicaid Services. Accountable Health Communities Model. CMS.gov web site. https://innovation.cms.gov/innovation-models/ahcm. Accessed October 10, 2020.
From T1D Exchange, Boston, MA (Dr. Ebekozien, Dr. Odugbesan, and Nicole Rioles); Barbara Davis Center, University of Colorado, Boulder, CO (Dr. Majidi); Cincinnati Children’s Hospital Medical Center, Cincinnati, OH (Dr. Jones); and Nationwide Children’s Hospital, Columbus, OH (Dr. Kamboj)
Health equity has been described as the opportunity for all persons to obtain their highest level of health possible.1 Unfortunately, even with advances in technology and care practices, disparities persist in health care outcomes. Disparities in prevalence, prognosis, and outcomes still exist in diabetes management.2 Non-Hispanic Black and/or Hispanic populations are more likely to have worse glycemic control,3,4 to encounter more barriers in access to care,5 and to have higher levels of acute complications,4 and to use advanced technologies less frequently.4 Diabetes is one of the preexisting conditions that increase morbidity and mortality in COVID-19.6,7 Unfortunately, adverse outcomes from COVID-19 also disproportionately impact a specific vulnerable population.8,9 The urgent transition to managing diabetes remotely during the COVID-19 pandemic may exacerbate long-term inequities because some vulnerable patients might not have access to technology devices necessary for effective remote management.
Here, we describe how quality improvement (QI) tools and principles can be adapted into a framework for advancing health equity. Specifically, we describe a 10-step framework that may be applied in diabetes care management to achieve improvement, using a hypothetical example of increasing use of continuous glucose monitors (CGMs) among patients with type 1 diabetes mellitus.10 This framework was developed to address the literature gap on practical ways health care providers can address inequities using QI principles, and was implemented by 1 of the authors at a local public health department.11 The framework’s iterative and comprehensive design makes it ideal for addressing inequities in chronic diseases like diabetes, which have multiple root causes with no easy solutions. The improvement program pilot received a national model practice award.11,12
10-Step Framework
Step 1: Review program/project baseline data for existing disparities. Diabetes programs and routine QI processes encourage existing data review to determine how effective the current system is working and if the existing process has a predictable pattern.13,14 Our equity-revised framework proposes a more in-depth review to stratify baseline data based on factors that might contribute to inequities, including race, age, income levels, ethnicity, language, sexual orientation, insurance type, and zip code. This process will identify patients not served or unfairly impacted due to socioeconomic factors. For example, using the hypothetical example of improving CGM use, a team completes a preliminary data review and determines that baseline CGM use is 30% in the clinic population. However, in a review to assess for disparities, they also identify that patients on public insurance have a significantly lower CGM uptake of only 15%.
Step 2: Build an equitable project team, including patients with lived experiences. Routine projects typically have clinicians, administrative staff, and analytic staff as members of their team. In a post-COVID-19 world, every team needs to learn directly from people impacted and share decision-making power. The traditional approach to receiving feedback has generally been to collect responses using surveys or focus groups. We propose that individuals/families who are disproportionately impacted be included as active members on QI teams. For example, in the hypothetical example of the CGM project, team members would include patients with type 1 diabetes who are on public insurance and their families.
Step 3: Develop equity-focused goals. The traditional program involves the development of aims that are SMART (specific, measurable, achievable, realistic, time-bound).15 The proposed framework encourages the inclusion of equity-revised goals (SMARTer) using insights from Steps 1 and 2. For example, your typical smart goal might be to increase the percentage of patients using CGM by 20% in 6 months, while a SMARTer goal would be to increase the proportion of patients using CGM by 20% and reduce the disparities among public and private insurance patients by 30% in 6 months.
Step 4: Identify inequitable processes/pathways. Traditional QI programs might use a process map or flow diagram to depict the current state of a process visually.16 For example, in Figure 1, the process map diagram depicts some differences in the process for patients with public insurance as opposed to those with private insurance. The framework also advocates for using visual tools like process maps to depict how there might be inequitable pathways in a system. Visually identifying inequitable processes/pathways can help a team see barriers, address challenges, and pilot innovative solutions.

Step 5: Identify how socioeconomic factors are contributing to the current outcome. A good understanding of factors that contribute to the problem is an essential part of finding fundamental solutions. The fishbone diagram16 is a visualization tool used to identify contributing factors. When investigating contributing factors, it is commonplace to identify factors that fit into 1 of 5 categories: people, process, place, product, and policies (5 Ps). An equity-focused process will include equity as a new major factor category, and the socioeconomic impacts that contribute to inequities will be brainstormed and visually represented. For example, in the hypothetical CGM improvement example, an equity contributing factor is extensive CGM application paperwork for patients on public insurance as compared to those on private insurance. Figure 2 shows equity integrated into a fishbone diagram.

Step 6: Brainstorm possible improvements. Potential improvement ideas for the hypothetical CGM example might include redesigning the existing workflow, piloting CGM educational classes, and using a CGM barrier assessment tool to identify and address barriers to adoption.
Step 7: Use the decision matrix with equity as a criterion to prioritize improvement ideas. Decision matrix15 is a great tool that is frequently used to help teams prioritize potential ideas. Project team members must decide what criteria are important in prioritizing ideas to implement. Common criteria include implementation cost, time, and resources, but in addition to the common criteria, the team can specify ”impact on equity” as one of their criteria, alongside other standard criteria like impact.
Step 8: Test one small change at a time. This step is consistent with other traditional improvement models using the Plan, Do, Study, Act (PDSA) model for improvement.17 During this phase, the team should make predictions on the expected impact of the intervention on outcomes. For example, in the hypothetical example, the team predicts that testing and expanding CGM classes will reduce disparities among public versus private health insurance users by 5% and increase overall CGM uptake by 10%.
Step 9: Measure and compare results with predictions to identify inequitable practices or consequences. After each test of change, the team should review the results, including implementation cost considerations, and compare them to the predictions in the earlier step. The team should also document the potential reasons why their predictions were correct or inaccurate, and whether there were any unforeseen outcomes from the intervention.
Step 10: Celebrate small wins and repeat the process. Making fundamental and equitable changes takes time. This framework aimed at undoing inequities, particularly those inequities that have been amplified by the COVID-19 pandemic, is iterative and ongoing.18,19 Not every test of change will impact the outcome or reduce inequity, but over time, each change will impact the next, generating sustainable effects.
Conclusion
There are ongoing studies examining the adverse outcomes and potential health inequities for patients with diabetes impacted by COVID-19.20 Health care providers need to plan for post-COVID-19 care, keeping in mind that the pandemic might worsen already existing health disparities in diabetes management.3,4,21 This work will involve an intentional approach to address structural and systemic racism.22 Therefore, the work of building health equity solutions must be rooted in racial justice, economic equity, and equitable access to health care and education.
Initiatives like this are currently being funded through foundation grants as well as state and federal research or program grants. Regional and national payors, including the Centers for Medicare & Medicaid Services, are currently piloting long-term sustainable funding models through programs like accountable care organizations and the Accountable Health Communities Model.23
Health systems can successfully address health equity and racial justice, using a framework as described above, to identify determinants of health, develop policies to expand access to care for the most vulnerable patients, distribute decision-making power, and train staff by naming structural racism as a driver of health inequities.
Acknowledgment: The authors acknowledge the contributions of patients, families, diabetes care teams, and collaborators within the T1D Exchange Quality Improvement Collaborative, who continually seek to improve care and outcomes for people living with diabetes.
Corresponding author: Osagie Ebekozien, MD, 11 Avenue De La Fayette, Boston, MA 02115; [email protected].
Financial disclosures: None.
Funding: T1D Exchange QI Collaborative is funded by The Leona M. and Harry B. Helmsley Charitable Trust. No specific funding was received for this manuscript or the development of this framework.
Keywords: type 1 diabetes; quality improvement; QI framework; racial justice; health disparities.
From T1D Exchange, Boston, MA (Dr. Ebekozien, Dr. Odugbesan, and Nicole Rioles); Barbara Davis Center, University of Colorado, Boulder, CO (Dr. Majidi); Cincinnati Children’s Hospital Medical Center, Cincinnati, OH (Dr. Jones); and Nationwide Children’s Hospital, Columbus, OH (Dr. Kamboj)
Health equity has been described as the opportunity for all persons to obtain their highest level of health possible.1 Unfortunately, even with advances in technology and care practices, disparities persist in health care outcomes. Disparities in prevalence, prognosis, and outcomes still exist in diabetes management.2 Non-Hispanic Black and/or Hispanic populations are more likely to have worse glycemic control,3,4 to encounter more barriers in access to care,5 and to have higher levels of acute complications,4 and to use advanced technologies less frequently.4 Diabetes is one of the preexisting conditions that increase morbidity and mortality in COVID-19.6,7 Unfortunately, adverse outcomes from COVID-19 also disproportionately impact a specific vulnerable population.8,9 The urgent transition to managing diabetes remotely during the COVID-19 pandemic may exacerbate long-term inequities because some vulnerable patients might not have access to technology devices necessary for effective remote management.
Here, we describe how quality improvement (QI) tools and principles can be adapted into a framework for advancing health equity. Specifically, we describe a 10-step framework that may be applied in diabetes care management to achieve improvement, using a hypothetical example of increasing use of continuous glucose monitors (CGMs) among patients with type 1 diabetes mellitus.10 This framework was developed to address the literature gap on practical ways health care providers can address inequities using QI principles, and was implemented by 1 of the authors at a local public health department.11 The framework’s iterative and comprehensive design makes it ideal for addressing inequities in chronic diseases like diabetes, which have multiple root causes with no easy solutions. The improvement program pilot received a national model practice award.11,12
10-Step Framework
Step 1: Review program/project baseline data for existing disparities. Diabetes programs and routine QI processes encourage existing data review to determine how effective the current system is working and if the existing process has a predictable pattern.13,14 Our equity-revised framework proposes a more in-depth review to stratify baseline data based on factors that might contribute to inequities, including race, age, income levels, ethnicity, language, sexual orientation, insurance type, and zip code. This process will identify patients not served or unfairly impacted due to socioeconomic factors. For example, using the hypothetical example of improving CGM use, a team completes a preliminary data review and determines that baseline CGM use is 30% in the clinic population. However, in a review to assess for disparities, they also identify that patients on public insurance have a significantly lower CGM uptake of only 15%.
Step 2: Build an equitable project team, including patients with lived experiences. Routine projects typically have clinicians, administrative staff, and analytic staff as members of their team. In a post-COVID-19 world, every team needs to learn directly from people impacted and share decision-making power. The traditional approach to receiving feedback has generally been to collect responses using surveys or focus groups. We propose that individuals/families who are disproportionately impacted be included as active members on QI teams. For example, in the hypothetical example of the CGM project, team members would include patients with type 1 diabetes who are on public insurance and their families.
Step 3: Develop equity-focused goals. The traditional program involves the development of aims that are SMART (specific, measurable, achievable, realistic, time-bound).15 The proposed framework encourages the inclusion of equity-revised goals (SMARTer) using insights from Steps 1 and 2. For example, your typical smart goal might be to increase the percentage of patients using CGM by 20% in 6 months, while a SMARTer goal would be to increase the proportion of patients using CGM by 20% and reduce the disparities among public and private insurance patients by 30% in 6 months.
Step 4: Identify inequitable processes/pathways. Traditional QI programs might use a process map or flow diagram to depict the current state of a process visually.16 For example, in Figure 1, the process map diagram depicts some differences in the process for patients with public insurance as opposed to those with private insurance. The framework also advocates for using visual tools like process maps to depict how there might be inequitable pathways in a system. Visually identifying inequitable processes/pathways can help a team see barriers, address challenges, and pilot innovative solutions.

Step 5: Identify how socioeconomic factors are contributing to the current outcome. A good understanding of factors that contribute to the problem is an essential part of finding fundamental solutions. The fishbone diagram16 is a visualization tool used to identify contributing factors. When investigating contributing factors, it is commonplace to identify factors that fit into 1 of 5 categories: people, process, place, product, and policies (5 Ps). An equity-focused process will include equity as a new major factor category, and the socioeconomic impacts that contribute to inequities will be brainstormed and visually represented. For example, in the hypothetical CGM improvement example, an equity contributing factor is extensive CGM application paperwork for patients on public insurance as compared to those on private insurance. Figure 2 shows equity integrated into a fishbone diagram.

Step 6: Brainstorm possible improvements. Potential improvement ideas for the hypothetical CGM example might include redesigning the existing workflow, piloting CGM educational classes, and using a CGM barrier assessment tool to identify and address barriers to adoption.
Step 7: Use the decision matrix with equity as a criterion to prioritize improvement ideas. Decision matrix15 is a great tool that is frequently used to help teams prioritize potential ideas. Project team members must decide what criteria are important in prioritizing ideas to implement. Common criteria include implementation cost, time, and resources, but in addition to the common criteria, the team can specify ”impact on equity” as one of their criteria, alongside other standard criteria like impact.
Step 8: Test one small change at a time. This step is consistent with other traditional improvement models using the Plan, Do, Study, Act (PDSA) model for improvement.17 During this phase, the team should make predictions on the expected impact of the intervention on outcomes. For example, in the hypothetical example, the team predicts that testing and expanding CGM classes will reduce disparities among public versus private health insurance users by 5% and increase overall CGM uptake by 10%.
Step 9: Measure and compare results with predictions to identify inequitable practices or consequences. After each test of change, the team should review the results, including implementation cost considerations, and compare them to the predictions in the earlier step. The team should also document the potential reasons why their predictions were correct or inaccurate, and whether there were any unforeseen outcomes from the intervention.
Step 10: Celebrate small wins and repeat the process. Making fundamental and equitable changes takes time. This framework aimed at undoing inequities, particularly those inequities that have been amplified by the COVID-19 pandemic, is iterative and ongoing.18,19 Not every test of change will impact the outcome or reduce inequity, but over time, each change will impact the next, generating sustainable effects.
Conclusion
There are ongoing studies examining the adverse outcomes and potential health inequities for patients with diabetes impacted by COVID-19.20 Health care providers need to plan for post-COVID-19 care, keeping in mind that the pandemic might worsen already existing health disparities in diabetes management.3,4,21 This work will involve an intentional approach to address structural and systemic racism.22 Therefore, the work of building health equity solutions must be rooted in racial justice, economic equity, and equitable access to health care and education.
Initiatives like this are currently being funded through foundation grants as well as state and federal research or program grants. Regional and national payors, including the Centers for Medicare & Medicaid Services, are currently piloting long-term sustainable funding models through programs like accountable care organizations and the Accountable Health Communities Model.23
Health systems can successfully address health equity and racial justice, using a framework as described above, to identify determinants of health, develop policies to expand access to care for the most vulnerable patients, distribute decision-making power, and train staff by naming structural racism as a driver of health inequities.
Acknowledgment: The authors acknowledge the contributions of patients, families, diabetes care teams, and collaborators within the T1D Exchange Quality Improvement Collaborative, who continually seek to improve care and outcomes for people living with diabetes.
Corresponding author: Osagie Ebekozien, MD, 11 Avenue De La Fayette, Boston, MA 02115; [email protected].
Financial disclosures: None.
Funding: T1D Exchange QI Collaborative is funded by The Leona M. and Harry B. Helmsley Charitable Trust. No specific funding was received for this manuscript or the development of this framework.
Keywords: type 1 diabetes; quality improvement; QI framework; racial justice; health disparities.
1. American Public Health Association Health Equity web site. https://www.apha.org/topics-and-issues/health-equity. Accessed June 4, 2020.
2. Lado J, Lipman T. Racial and ethnic disparities in the incidence, treatment, and outcomes of youth with type 1 diabetes. Endocrinol Metab Clin North Am. 2016;45:453-461.
3. Kahkoska AR, Shay CM, Crandell J, et al. Association of race and ethnicity with glycemic control and hemoglobin A1c levels in youth with type 1 diabetes. JAMA Netw Open. 2018;1:e181851.
4. Willi SM, Miller KM, DiMeglio LA, et al; T1D Exchange Clinic Network. Racial-ethnic disparities in management and outcomes among children with type 1 diabetes. Pediatrics. 2015;135:424-434.
5. Valenzuela JM, Seid M, Waitzfelder B, et al. Prevalence of and disparities in barriers to care experienced by youth with type 1 diabetes. J Pediatr. 2014;164:1369-1375.
6. Hussain A, Bhowmik B, do Vale Moreira NC. COVID-19 and diabetes: Knowledge in progress. Diabetes Res Clin Pract. 2020;162:108142.
7. Bhatraju PK, Ghassemieh BJ, Nichols M, et al. Covid-19 in critically ill patients in the Seattle Region - case series. N Engl J Med. 2020;382:2012-2022.
8. Laurencin CT, McClinton A. The COVID-19 pandemic: a call to action to identify and address racial and ethnic disparities. J Racial Ethn Health Disparities. 2020;7:398-402.
9. Shah M, Sachdeva M, Dodiuk-Gad RP. COVID-19 and racial disparities. J Am Acad Dermatol. 2020;83:e35.
10. Ebekozien O, Rioles N, DeSalvo D, et al. Improving continuous glucose monitoring (CGM) use across national centers: results from the T1D Exchange Quality Improvement Collaborative (T1DX-QI). Diabetes. 2020;69(Supplement 1):145-LB.
11. Ebekozien O. QI methodology to address health equity. Presented at American Society of Quality BOSCON 2018; Boston, MA; March 19 and 20, 2018.
12. 2019 Model Practice Award, Building A Culture of Improvement. National Association of County and City Health Officials web site. www.naccho.org/membership/awards/model-practices. Accessed June 4, 2020.
13. Nuckols TK, Keeler E, Anderson LJ, et al. Economic evaluation of quality improvement interventions designed to improve glycemic control in diabetes: a systematic review and weighted regression analysis. Diabetes Care. 2018;41:985‐993.
14. Rossi MC, Nicolucci A, Arcangeli A, et al. Baseline quality-of-care data from a quality-improvement program implemented by a network of diabetes outpatient clinics. Diabetes Care. 2008;31:2166‐2168.
15. McQuillan RF, Silver SA, Harel Z, et al. How to measure and interpret quality improvement data. Clin J Am Soc Nephrol. 2016;11:908-914.
16. Siddiqi FS. Quality improvement in diabetes care: time for us to step up? Can J Diabetes. 2019;43:233.
17. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan-do-study-act method to improve quality in healthcare. BMJ Qual Saf. 2014;23:290‐298.
18. Ferdinand KC, Nasser SA. African American COVID-19 mortality: a sentinel event. J Am Coll Cardiol. 2020;75:2746-2748..
19. Muniyappa R, Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab. 2020;318:E736-E741.
20. Ebekozien OA, Noor N, Gallagher MP, Alonso GT. Type 1 diabetes and COVID-19: preliminary findings from a multicenter surveillance study in the U.S. Diabetes Care. 2020;43:e83-e85.
21. Majidi S, Ebekozien O, Noor N, et al. Inequities in health outcomes among patients in the T1D Exchange-QI Collaborative. Diabetes. 2020;69(Supplement 1):1220-P. https://doi.org/10.2337/ db20-1220.-P.
22. Williams DR, Mohammed SA. Discrimination and racial disparities in health: evidence and needed research. J Behav Med. 2009;32:20-47.
23. Centers for Medicare & Medicaid Services. Accountable Health Communities Model. CMS.gov web site. https://innovation.cms.gov/innovation-models/ahcm. Accessed October 10, 2020.
1. American Public Health Association Health Equity web site. https://www.apha.org/topics-and-issues/health-equity. Accessed June 4, 2020.
2. Lado J, Lipman T. Racial and ethnic disparities in the incidence, treatment, and outcomes of youth with type 1 diabetes. Endocrinol Metab Clin North Am. 2016;45:453-461.
3. Kahkoska AR, Shay CM, Crandell J, et al. Association of race and ethnicity with glycemic control and hemoglobin A1c levels in youth with type 1 diabetes. JAMA Netw Open. 2018;1:e181851.
4. Willi SM, Miller KM, DiMeglio LA, et al; T1D Exchange Clinic Network. Racial-ethnic disparities in management and outcomes among children with type 1 diabetes. Pediatrics. 2015;135:424-434.
5. Valenzuela JM, Seid M, Waitzfelder B, et al. Prevalence of and disparities in barriers to care experienced by youth with type 1 diabetes. J Pediatr. 2014;164:1369-1375.
6. Hussain A, Bhowmik B, do Vale Moreira NC. COVID-19 and diabetes: Knowledge in progress. Diabetes Res Clin Pract. 2020;162:108142.
7. Bhatraju PK, Ghassemieh BJ, Nichols M, et al. Covid-19 in critically ill patients in the Seattle Region - case series. N Engl J Med. 2020;382:2012-2022.
8. Laurencin CT, McClinton A. The COVID-19 pandemic: a call to action to identify and address racial and ethnic disparities. J Racial Ethn Health Disparities. 2020;7:398-402.
9. Shah M, Sachdeva M, Dodiuk-Gad RP. COVID-19 and racial disparities. J Am Acad Dermatol. 2020;83:e35.
10. Ebekozien O, Rioles N, DeSalvo D, et al. Improving continuous glucose monitoring (CGM) use across national centers: results from the T1D Exchange Quality Improvement Collaborative (T1DX-QI). Diabetes. 2020;69(Supplement 1):145-LB.
11. Ebekozien O. QI methodology to address health equity. Presented at American Society of Quality BOSCON 2018; Boston, MA; March 19 and 20, 2018.
12. 2019 Model Practice Award, Building A Culture of Improvement. National Association of County and City Health Officials web site. www.naccho.org/membership/awards/model-practices. Accessed June 4, 2020.
13. Nuckols TK, Keeler E, Anderson LJ, et al. Economic evaluation of quality improvement interventions designed to improve glycemic control in diabetes: a systematic review and weighted regression analysis. Diabetes Care. 2018;41:985‐993.
14. Rossi MC, Nicolucci A, Arcangeli A, et al. Baseline quality-of-care data from a quality-improvement program implemented by a network of diabetes outpatient clinics. Diabetes Care. 2008;31:2166‐2168.
15. McQuillan RF, Silver SA, Harel Z, et al. How to measure and interpret quality improvement data. Clin J Am Soc Nephrol. 2016;11:908-914.
16. Siddiqi FS. Quality improvement in diabetes care: time for us to step up? Can J Diabetes. 2019;43:233.
17. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan-do-study-act method to improve quality in healthcare. BMJ Qual Saf. 2014;23:290‐298.
18. Ferdinand KC, Nasser SA. African American COVID-19 mortality: a sentinel event. J Am Coll Cardiol. 2020;75:2746-2748..
19. Muniyappa R, Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab. 2020;318:E736-E741.
20. Ebekozien OA, Noor N, Gallagher MP, Alonso GT. Type 1 diabetes and COVID-19: preliminary findings from a multicenter surveillance study in the U.S. Diabetes Care. 2020;43:e83-e85.
21. Majidi S, Ebekozien O, Noor N, et al. Inequities in health outcomes among patients in the T1D Exchange-QI Collaborative. Diabetes. 2020;69(Supplement 1):1220-P. https://doi.org/10.2337/ db20-1220.-P.
22. Williams DR, Mohammed SA. Discrimination and racial disparities in health: evidence and needed research. J Behav Med. 2009;32:20-47.
23. Centers for Medicare & Medicaid Services. Accountable Health Communities Model. CMS.gov web site. https://innovation.cms.gov/innovation-models/ahcm. Accessed October 10, 2020.
Telepsychiatry poised to thrive after the pandemic
Once the fog lifts on a global pandemic that led to an explosion in telehealth visits in 2020, mental health experts expect virtual and in-person visits to merge to become a standard model of care in clinical psychiatry.
Hybrid care is the future, Peter Yellowlees, MBBS, MD, a professor of psychiatry and chief wellness officer at the University of California, Davis, said in an interview. “I’ve been working this way for several years – where all of my patients get to see me in person or online, or both.”
The model’s increasing popularity reflects a major shift toward virtual consults. Telemedicine offers safer, quicker, and less expensive alternatives, Steven Chan, MD, MBA, of Stanford (Calif.) University, said in an interview. “This continuity is essential to helping to reduce emergency room visits, reduce inpatient hospitalizations and readmissions, and improving adherence to treatment,” Dr. Chan said. State and federal regulators’ actions to lift certain licensing and prescribing restrictions and expand coverage made it easier for clinical psychiatrists to offer and get paid for these services.
The catch is that no one knows whether these easements will remain in place once COVID-19 recedes, ending the national public health emergency, Jay H. Shore, MD, MPH, chairperson of the American Psychiatric Association telepsychiatry committee, and professor and director of telemedicine programming at the University of Colorado at Denver, Aurora’s department of psychiatry, said in an interview. “These all temporarily changed during this time period, but we have no idea when they’re going to end or if they’re going to continue off of COVID. So now, there’s a lot of uncertainty.”
‘Suite of different technologies’
New freedoms to deliver telehealth care left some practices scrambling to adopt or refine technology. Once COVID-19 hit, “I can’t think of a psychiatrist or provider or institution that didn’t have to rapidly virtualize and do at least some video conferencing,” Dr. Shore said. This immediate shift signaled a key move toward hybrid patient-doctor relations. “It means you hold a relationship with your patients through multiple different mediums, email, portal, telephone, in person. It’s not just about in-person versus video, it’s about a suite of different technologies.”
Dr. Shore began practicing telehealth 20 years ago, long before the age of COVID. He’s since established rewarding relationships with patients he’s never met. “I’ve done everything from medical management to long-term psychotherapy, group psychotherapy, and I’ve been successful with different populations.”
Many nuances exist around matching the right patient with the right videoconferencing adaptation, Dr. Shore continued. However, “in general, the literature supports that you can get equal clinical outcomes with telehealth versus in-person treatment.”
‘I see their garden’
While some may eschew the idea of providing care over a virtual platform, other physicians see it as an insightful window into a person’s mental state. “I think some are actually quite surprised by how much good care you can give using video,” said Dr. Yellowlees. Unlike a phone conversation, video allows you to see a person’s home. “The beauty of a video is not just that you can see the person, you can also look around their home, and learn more about them.”
In his own visits, he asks patients to take him on a virtual “walk” through their house, provided there are no confidentiality issues.
“I get to meet their pets, the carers, the spouses; I get to see their garden. I get to see what their interests are from looking at the paintings on their walls. I learn more about my patients that way. If you use video to purely see people from the neck up, then that’s fine, but I think you can also use it to your advantage, seeing people at home.” He also encourages patients to do visits in their cars – as long as the windows are shut, they’re in a safe area, and most importantly, they’re not driving.
Nina Vasan, MD, MBA, who treats patients at Stanford and in a concierge private practice for executives, agrees that seeing patients in the home offers a more direct view of patients’ lives. “These little extra pieces of information are things that we didn’t get before COVID, when patients would come by themselves into the office week after week. I do feel like I know some patients much better by being able to see their surroundings and home interactions,” Dr. Vasan, founder and executive director of Brainstorm: The Stanford Lab for Mental Health Innovation and chair of the APA’s committee on innovation, said in an interview.
She uses Zoom for her sessions, and regularly texts with her concierge patients. In both clinics, where most visits have gone virtual, “the no-show rate is near zero. Before COVID, it was around 10%-20%.”
Safety is a key advantage of virtual visits, noted Dr. Shore. “It’s certainly better than some of the risks of seeing people in person.”
Lack of integration causes frustration
Some challenges exist around access and adaptation of virtual technology. The pandemic’s sudden onslaught left many clinicians scrambling to adjust to a virtual format without any training or tools to support them. “This has caused some stress,” Dr. Shore said.
Electronic health record systems may or may not integrate video visits, noted Dr. Chan. “For instance, I work at a health system where mental health questionnaires, imaging systems, notes, and video visit scheduling are housed in separate systems.”
Without a seamless national, integrated system in place, physicians are often left to couple different IT formats together. “There are some systems that work pretty well, and there are some that are really clunky where you’re forcing the two together, so that’s a challenge,” Dr. Shore said. Providers may be using one system for their office and another system to provide care. “They may be using different EHRs and different teleconferencing platforms. That adds complexity.” Video conferencing platforms aren’t as challenging to use as EHRs. However, if some people are self-taught, there’s no way of knowing if they’re using best practices, he added.
Many health systems do offer video built into EMRs. Companies such as Epic have provided integrations with Vidyo, Amwell, and other platforms, noted Dr. Chan.
An integrated platform is useful if done well – but isn’t necessarily essential, according to Dr. Yellowlees. In his own setup, he signs a laptop into an EMR and uses an iPad to communicate with patients via video on the iPad and all video on the phone.
“The big advantage of telemedicine is you can type your notes in a socially appropriate way while talking to the patient on video. I do that, and it saves me a considerable amount of time. I don’t have to spend time after a consultation typing up notes.”
Still, others may struggle with bandwidth or connectivity. “Perhaps there’s a problem with privacy or the types of patients you’re dealing with,” Dr. Shore said. Older patients in a nursing home, for example, may require a team of people that works onsite. Suddenly, their care has to transition to a virtual system. “You may need to figure how to put the right team together to do straightforward and individual interactions,” he said.
Virtual care also suffers from the “digital divide,” an issue that predates COVID-19, said Dr. Shore. Not all patients have access to bandwidth and the technology to see clinicians. Other lack expertise and comfort with using the technology, or can’t afford the equipment necessary to bring them online. The pandemic has highlighted all of these disparities, he emphasized.
Dr. Chan offered that the digital divide is part of a larger socioeconomic divide, where barriers to any care – including transportation for in-person care – exist.
Technology and access issues aside, insidious “Zoom fatigue” is affecting everyone right now. “That’s clearly real,” Dr. Shore said. “And it’s not just about videoconferencing; it’s about being in quarantine. We’re all in this virtual lockdown, where people are using the term ‘videoconferencing fatigue.’ It’s a complicated problem.”
To assist with telehealth implementation, the APA has issued practice guidance and a toolkit that includes an extensive set of educational materials, including 40 videos on various topics. It’s also hosted webinars on telehealth policies and written up standard operating procedures on this topic, Dr. Yellowlees said. “The most important thing is APA, like other organizations, is advocating to have the relaxation of regulations during COVID made permanent,” he said.
Reimbursement post pandemic
As virtual visits rose in 2020, so did billing for such services. According to America’s Health Insurance Plans, claims to private insurers exploded by more than 4,000% in 2020. AHIP last July reported that mental health conditions made up one-third or more than 33% of telehealth claims to private insurers during the pandemic. Dr. Yellowlees said the health insurers he’s dealt with “have been good about paying for telehealth visits during the COVID-declared emergency.”
Whether this coverage will continue once the public health emergency ends, is unclear, some contend. “My sense is that there will be a rollback of coverage, but it won’t be back to what we had prepandemic,” said Ateev Mehrotra, MD, MPH, who studies telemedicine trends, in an interview. Right now, there’s still a great deal of uncertainty about reimbursement, “and that’s what’s giving providers pause,” said Dr. Mehrotra, of the department of health care policy and medicine at Harvard Medical School and a hospitalist at Beth Israel Deaconess Medical Center, both in Boston.
AHIP will continue to support federal and state policies to further promote telehealth access during the public health emergency, spokesman David Allen said in a statement. “Insurance providers have independently shifted their policies to increase access to care and services, ranging from acute care needs and triage services to chronic disease management and behavioral health. We are still awaiting information on what changes will remain in place beyond the public health emergency,” he said.
Even before COVID-19, Mr. Allen noted, nearly all large employers (96%) offered access to telehealth services as a covered benefit in 2019. In May, the Kaiser Family Foundation reported that many insurers were reducing or lifting cost sharing for telehealth for limited time periods.
Some of the larger payers said they’re continuing benefits, although it’s not clear how long some benefits will remain in place.
UnitedHealthcare offers no-cost coverage for COVID-19 testing–related telehealth visits, but that benefit is set to expire once the public health emergency presumably ends on Jan. 20, 2021. Through Dec. 31, 2020, the payer said it would offer coverage with no cost sharing for telehealth visits related to COVID-19 treatment and expanded access to telehealth visits not related to COVID-19 through its network. Similarly, Anthem is waiving cost sharing for COVID-19 treatments via telehealth or in-person visits, and for telehealth visits not related to COVID-19 for Medicare members through the end of the year.
Anthem will continue to cover telehealth and encourage members and clinicians to use telehealth for behavioral health, a spokesperson said. Anthem also has a telehealth provider, LiveHealth Online, “another safe and effective way for members to see a doctor to receive health guidance from their home via mobile device or a computer with a webcam,” said the spokesperson.
Dr. Vasan hopes that insurers will increase coverage for telehealth and at the same rate as in-person visits, especially for mental health. “I have not felt that the quality of the clinic has decreased, and in fact, in some ways it’s gotten better, and insurance coverage should reflect this.”
Outlook for the hybrid model
As long as there’s COVID-19, psychiatry practices must remain virtual, at least for now, Dr. Shore said. “We will emerge from this pandemic, I suspect in bits and starts.” When that happens, practices will need to have a transition plan in place. “Once we get away from COVID, I don’t think our mental health will ever be the same again. We’ll have much more virtual technology along the lines of a hybrid model, where we’ll see patients in person, but we’ll use more technology to work with patients, but it will be more of a blend.” Practices will also have to address the regulatory, reimbursement, and prescribing conditions the new world offers.
Some practices are already discovering the benefits of relying less on a brick-and-mortar office.
Dr. Chan said his colleagues are finding that they no longer have to deal with the expense and upkeep of renting and furnishing office space. “Many are taking their practice virtual-only because the monthly recurring costs are so cheap, and they can see patients in distant, underserved communities.” In-person visits are now inconvenient and risky, he continued. They require expensive personal protective equipment and cleaning protocols. “Plus, there’s the risk that services must shut down when stay-at-home orders return or when a staff member gets infected.”
Physicians prescribing certain controlled substances will likely continue to use office space, once the public health emergency expires and face-to-face visits resume. “In such cases, they can rent office space part-time,” Dr. Chan added.
Dr. Vasan, also of the department of psychiatry at Stanford and chief medical officer of Real, hopes that such a model prevails. “I do miss seeing patients in person and think a hybrid will be a good balance.”
Most patients want it, as do the influx of Generation Z physicians coming into the profession, Dr. Yellowlees noted. These are young, technologically savvy doctors who grew up in the age of the Internet. “I think the silver lining of COVID is it led telemedicine past the tipping point, where both patients and providers are learning that it’s an appropriate way to get care, as long as you’re careful, use professional guidelines – and don’t drop your standards of care.”
Dr. Yellowlees and Dr. Shore are coauthors of Telepsychiatry and Health Technologies: A Guide for Mental Health Professionals (Washington: American Psychiatric Association Publishing, 2018), and receive royalties from the book. Dr. Shore also reported working with AccessCare and receiving royalties from Springer Press. Dr. Chan reported consulting for Orbit Health. Dr. Vasan reported no conflicts of interest. Dr. Mehrotra has received research funding from several U.S. agencies, including the National Institute of Mental Health and the National Institute of Neurological Disorders and Stroke.
Once the fog lifts on a global pandemic that led to an explosion in telehealth visits in 2020, mental health experts expect virtual and in-person visits to merge to become a standard model of care in clinical psychiatry.
Hybrid care is the future, Peter Yellowlees, MBBS, MD, a professor of psychiatry and chief wellness officer at the University of California, Davis, said in an interview. “I’ve been working this way for several years – where all of my patients get to see me in person or online, or both.”
The model’s increasing popularity reflects a major shift toward virtual consults. Telemedicine offers safer, quicker, and less expensive alternatives, Steven Chan, MD, MBA, of Stanford (Calif.) University, said in an interview. “This continuity is essential to helping to reduce emergency room visits, reduce inpatient hospitalizations and readmissions, and improving adherence to treatment,” Dr. Chan said. State and federal regulators’ actions to lift certain licensing and prescribing restrictions and expand coverage made it easier for clinical psychiatrists to offer and get paid for these services.
The catch is that no one knows whether these easements will remain in place once COVID-19 recedes, ending the national public health emergency, Jay H. Shore, MD, MPH, chairperson of the American Psychiatric Association telepsychiatry committee, and professor and director of telemedicine programming at the University of Colorado at Denver, Aurora’s department of psychiatry, said in an interview. “These all temporarily changed during this time period, but we have no idea when they’re going to end or if they’re going to continue off of COVID. So now, there’s a lot of uncertainty.”
‘Suite of different technologies’
New freedoms to deliver telehealth care left some practices scrambling to adopt or refine technology. Once COVID-19 hit, “I can’t think of a psychiatrist or provider or institution that didn’t have to rapidly virtualize and do at least some video conferencing,” Dr. Shore said. This immediate shift signaled a key move toward hybrid patient-doctor relations. “It means you hold a relationship with your patients through multiple different mediums, email, portal, telephone, in person. It’s not just about in-person versus video, it’s about a suite of different technologies.”
Dr. Shore began practicing telehealth 20 years ago, long before the age of COVID. He’s since established rewarding relationships with patients he’s never met. “I’ve done everything from medical management to long-term psychotherapy, group psychotherapy, and I’ve been successful with different populations.”
Many nuances exist around matching the right patient with the right videoconferencing adaptation, Dr. Shore continued. However, “in general, the literature supports that you can get equal clinical outcomes with telehealth versus in-person treatment.”
‘I see their garden’
While some may eschew the idea of providing care over a virtual platform, other physicians see it as an insightful window into a person’s mental state. “I think some are actually quite surprised by how much good care you can give using video,” said Dr. Yellowlees. Unlike a phone conversation, video allows you to see a person’s home. “The beauty of a video is not just that you can see the person, you can also look around their home, and learn more about them.”
In his own visits, he asks patients to take him on a virtual “walk” through their house, provided there are no confidentiality issues.
“I get to meet their pets, the carers, the spouses; I get to see their garden. I get to see what their interests are from looking at the paintings on their walls. I learn more about my patients that way. If you use video to purely see people from the neck up, then that’s fine, but I think you can also use it to your advantage, seeing people at home.” He also encourages patients to do visits in their cars – as long as the windows are shut, they’re in a safe area, and most importantly, they’re not driving.
Nina Vasan, MD, MBA, who treats patients at Stanford and in a concierge private practice for executives, agrees that seeing patients in the home offers a more direct view of patients’ lives. “These little extra pieces of information are things that we didn’t get before COVID, when patients would come by themselves into the office week after week. I do feel like I know some patients much better by being able to see their surroundings and home interactions,” Dr. Vasan, founder and executive director of Brainstorm: The Stanford Lab for Mental Health Innovation and chair of the APA’s committee on innovation, said in an interview.
She uses Zoom for her sessions, and regularly texts with her concierge patients. In both clinics, where most visits have gone virtual, “the no-show rate is near zero. Before COVID, it was around 10%-20%.”
Safety is a key advantage of virtual visits, noted Dr. Shore. “It’s certainly better than some of the risks of seeing people in person.”
Lack of integration causes frustration
Some challenges exist around access and adaptation of virtual technology. The pandemic’s sudden onslaught left many clinicians scrambling to adjust to a virtual format without any training or tools to support them. “This has caused some stress,” Dr. Shore said.
Electronic health record systems may or may not integrate video visits, noted Dr. Chan. “For instance, I work at a health system where mental health questionnaires, imaging systems, notes, and video visit scheduling are housed in separate systems.”
Without a seamless national, integrated system in place, physicians are often left to couple different IT formats together. “There are some systems that work pretty well, and there are some that are really clunky where you’re forcing the two together, so that’s a challenge,” Dr. Shore said. Providers may be using one system for their office and another system to provide care. “They may be using different EHRs and different teleconferencing platforms. That adds complexity.” Video conferencing platforms aren’t as challenging to use as EHRs. However, if some people are self-taught, there’s no way of knowing if they’re using best practices, he added.
Many health systems do offer video built into EMRs. Companies such as Epic have provided integrations with Vidyo, Amwell, and other platforms, noted Dr. Chan.
An integrated platform is useful if done well – but isn’t necessarily essential, according to Dr. Yellowlees. In his own setup, he signs a laptop into an EMR and uses an iPad to communicate with patients via video on the iPad and all video on the phone.
“The big advantage of telemedicine is you can type your notes in a socially appropriate way while talking to the patient on video. I do that, and it saves me a considerable amount of time. I don’t have to spend time after a consultation typing up notes.”
Still, others may struggle with bandwidth or connectivity. “Perhaps there’s a problem with privacy or the types of patients you’re dealing with,” Dr. Shore said. Older patients in a nursing home, for example, may require a team of people that works onsite. Suddenly, their care has to transition to a virtual system. “You may need to figure how to put the right team together to do straightforward and individual interactions,” he said.
Virtual care also suffers from the “digital divide,” an issue that predates COVID-19, said Dr. Shore. Not all patients have access to bandwidth and the technology to see clinicians. Other lack expertise and comfort with using the technology, or can’t afford the equipment necessary to bring them online. The pandemic has highlighted all of these disparities, he emphasized.
Dr. Chan offered that the digital divide is part of a larger socioeconomic divide, where barriers to any care – including transportation for in-person care – exist.
Technology and access issues aside, insidious “Zoom fatigue” is affecting everyone right now. “That’s clearly real,” Dr. Shore said. “And it’s not just about videoconferencing; it’s about being in quarantine. We’re all in this virtual lockdown, where people are using the term ‘videoconferencing fatigue.’ It’s a complicated problem.”
To assist with telehealth implementation, the APA has issued practice guidance and a toolkit that includes an extensive set of educational materials, including 40 videos on various topics. It’s also hosted webinars on telehealth policies and written up standard operating procedures on this topic, Dr. Yellowlees said. “The most important thing is APA, like other organizations, is advocating to have the relaxation of regulations during COVID made permanent,” he said.
Reimbursement post pandemic
As virtual visits rose in 2020, so did billing for such services. According to America’s Health Insurance Plans, claims to private insurers exploded by more than 4,000% in 2020. AHIP last July reported that mental health conditions made up one-third or more than 33% of telehealth claims to private insurers during the pandemic. Dr. Yellowlees said the health insurers he’s dealt with “have been good about paying for telehealth visits during the COVID-declared emergency.”
Whether this coverage will continue once the public health emergency ends, is unclear, some contend. “My sense is that there will be a rollback of coverage, but it won’t be back to what we had prepandemic,” said Ateev Mehrotra, MD, MPH, who studies telemedicine trends, in an interview. Right now, there’s still a great deal of uncertainty about reimbursement, “and that’s what’s giving providers pause,” said Dr. Mehrotra, of the department of health care policy and medicine at Harvard Medical School and a hospitalist at Beth Israel Deaconess Medical Center, both in Boston.
AHIP will continue to support federal and state policies to further promote telehealth access during the public health emergency, spokesman David Allen said in a statement. “Insurance providers have independently shifted their policies to increase access to care and services, ranging from acute care needs and triage services to chronic disease management and behavioral health. We are still awaiting information on what changes will remain in place beyond the public health emergency,” he said.
Even before COVID-19, Mr. Allen noted, nearly all large employers (96%) offered access to telehealth services as a covered benefit in 2019. In May, the Kaiser Family Foundation reported that many insurers were reducing or lifting cost sharing for telehealth for limited time periods.
Some of the larger payers said they’re continuing benefits, although it’s not clear how long some benefits will remain in place.
UnitedHealthcare offers no-cost coverage for COVID-19 testing–related telehealth visits, but that benefit is set to expire once the public health emergency presumably ends on Jan. 20, 2021. Through Dec. 31, 2020, the payer said it would offer coverage with no cost sharing for telehealth visits related to COVID-19 treatment and expanded access to telehealth visits not related to COVID-19 through its network. Similarly, Anthem is waiving cost sharing for COVID-19 treatments via telehealth or in-person visits, and for telehealth visits not related to COVID-19 for Medicare members through the end of the year.
Anthem will continue to cover telehealth and encourage members and clinicians to use telehealth for behavioral health, a spokesperson said. Anthem also has a telehealth provider, LiveHealth Online, “another safe and effective way for members to see a doctor to receive health guidance from their home via mobile device or a computer with a webcam,” said the spokesperson.
Dr. Vasan hopes that insurers will increase coverage for telehealth and at the same rate as in-person visits, especially for mental health. “I have not felt that the quality of the clinic has decreased, and in fact, in some ways it’s gotten better, and insurance coverage should reflect this.”
Outlook for the hybrid model
As long as there’s COVID-19, psychiatry practices must remain virtual, at least for now, Dr. Shore said. “We will emerge from this pandemic, I suspect in bits and starts.” When that happens, practices will need to have a transition plan in place. “Once we get away from COVID, I don’t think our mental health will ever be the same again. We’ll have much more virtual technology along the lines of a hybrid model, where we’ll see patients in person, but we’ll use more technology to work with patients, but it will be more of a blend.” Practices will also have to address the regulatory, reimbursement, and prescribing conditions the new world offers.
Some practices are already discovering the benefits of relying less on a brick-and-mortar office.
Dr. Chan said his colleagues are finding that they no longer have to deal with the expense and upkeep of renting and furnishing office space. “Many are taking their practice virtual-only because the monthly recurring costs are so cheap, and they can see patients in distant, underserved communities.” In-person visits are now inconvenient and risky, he continued. They require expensive personal protective equipment and cleaning protocols. “Plus, there’s the risk that services must shut down when stay-at-home orders return or when a staff member gets infected.”
Physicians prescribing certain controlled substances will likely continue to use office space, once the public health emergency expires and face-to-face visits resume. “In such cases, they can rent office space part-time,” Dr. Chan added.
Dr. Vasan, also of the department of psychiatry at Stanford and chief medical officer of Real, hopes that such a model prevails. “I do miss seeing patients in person and think a hybrid will be a good balance.”
Most patients want it, as do the influx of Generation Z physicians coming into the profession, Dr. Yellowlees noted. These are young, technologically savvy doctors who grew up in the age of the Internet. “I think the silver lining of COVID is it led telemedicine past the tipping point, where both patients and providers are learning that it’s an appropriate way to get care, as long as you’re careful, use professional guidelines – and don’t drop your standards of care.”
Dr. Yellowlees and Dr. Shore are coauthors of Telepsychiatry and Health Technologies: A Guide for Mental Health Professionals (Washington: American Psychiatric Association Publishing, 2018), and receive royalties from the book. Dr. Shore also reported working with AccessCare and receiving royalties from Springer Press. Dr. Chan reported consulting for Orbit Health. Dr. Vasan reported no conflicts of interest. Dr. Mehrotra has received research funding from several U.S. agencies, including the National Institute of Mental Health and the National Institute of Neurological Disorders and Stroke.
Once the fog lifts on a global pandemic that led to an explosion in telehealth visits in 2020, mental health experts expect virtual and in-person visits to merge to become a standard model of care in clinical psychiatry.
Hybrid care is the future, Peter Yellowlees, MBBS, MD, a professor of psychiatry and chief wellness officer at the University of California, Davis, said in an interview. “I’ve been working this way for several years – where all of my patients get to see me in person or online, or both.”
The model’s increasing popularity reflects a major shift toward virtual consults. Telemedicine offers safer, quicker, and less expensive alternatives, Steven Chan, MD, MBA, of Stanford (Calif.) University, said in an interview. “This continuity is essential to helping to reduce emergency room visits, reduce inpatient hospitalizations and readmissions, and improving adherence to treatment,” Dr. Chan said. State and federal regulators’ actions to lift certain licensing and prescribing restrictions and expand coverage made it easier for clinical psychiatrists to offer and get paid for these services.
The catch is that no one knows whether these easements will remain in place once COVID-19 recedes, ending the national public health emergency, Jay H. Shore, MD, MPH, chairperson of the American Psychiatric Association telepsychiatry committee, and professor and director of telemedicine programming at the University of Colorado at Denver, Aurora’s department of psychiatry, said in an interview. “These all temporarily changed during this time period, but we have no idea when they’re going to end or if they’re going to continue off of COVID. So now, there’s a lot of uncertainty.”
‘Suite of different technologies’
New freedoms to deliver telehealth care left some practices scrambling to adopt or refine technology. Once COVID-19 hit, “I can’t think of a psychiatrist or provider or institution that didn’t have to rapidly virtualize and do at least some video conferencing,” Dr. Shore said. This immediate shift signaled a key move toward hybrid patient-doctor relations. “It means you hold a relationship with your patients through multiple different mediums, email, portal, telephone, in person. It’s not just about in-person versus video, it’s about a suite of different technologies.”
Dr. Shore began practicing telehealth 20 years ago, long before the age of COVID. He’s since established rewarding relationships with patients he’s never met. “I’ve done everything from medical management to long-term psychotherapy, group psychotherapy, and I’ve been successful with different populations.”
Many nuances exist around matching the right patient with the right videoconferencing adaptation, Dr. Shore continued. However, “in general, the literature supports that you can get equal clinical outcomes with telehealth versus in-person treatment.”
‘I see their garden’
While some may eschew the idea of providing care over a virtual platform, other physicians see it as an insightful window into a person’s mental state. “I think some are actually quite surprised by how much good care you can give using video,” said Dr. Yellowlees. Unlike a phone conversation, video allows you to see a person’s home. “The beauty of a video is not just that you can see the person, you can also look around their home, and learn more about them.”
In his own visits, he asks patients to take him on a virtual “walk” through their house, provided there are no confidentiality issues.
“I get to meet their pets, the carers, the spouses; I get to see their garden. I get to see what their interests are from looking at the paintings on their walls. I learn more about my patients that way. If you use video to purely see people from the neck up, then that’s fine, but I think you can also use it to your advantage, seeing people at home.” He also encourages patients to do visits in their cars – as long as the windows are shut, they’re in a safe area, and most importantly, they’re not driving.
Nina Vasan, MD, MBA, who treats patients at Stanford and in a concierge private practice for executives, agrees that seeing patients in the home offers a more direct view of patients’ lives. “These little extra pieces of information are things that we didn’t get before COVID, when patients would come by themselves into the office week after week. I do feel like I know some patients much better by being able to see their surroundings and home interactions,” Dr. Vasan, founder and executive director of Brainstorm: The Stanford Lab for Mental Health Innovation and chair of the APA’s committee on innovation, said in an interview.
She uses Zoom for her sessions, and regularly texts with her concierge patients. In both clinics, where most visits have gone virtual, “the no-show rate is near zero. Before COVID, it was around 10%-20%.”
Safety is a key advantage of virtual visits, noted Dr. Shore. “It’s certainly better than some of the risks of seeing people in person.”
Lack of integration causes frustration
Some challenges exist around access and adaptation of virtual technology. The pandemic’s sudden onslaught left many clinicians scrambling to adjust to a virtual format without any training or tools to support them. “This has caused some stress,” Dr. Shore said.
Electronic health record systems may or may not integrate video visits, noted Dr. Chan. “For instance, I work at a health system where mental health questionnaires, imaging systems, notes, and video visit scheduling are housed in separate systems.”
Without a seamless national, integrated system in place, physicians are often left to couple different IT formats together. “There are some systems that work pretty well, and there are some that are really clunky where you’re forcing the two together, so that’s a challenge,” Dr. Shore said. Providers may be using one system for their office and another system to provide care. “They may be using different EHRs and different teleconferencing platforms. That adds complexity.” Video conferencing platforms aren’t as challenging to use as EHRs. However, if some people are self-taught, there’s no way of knowing if they’re using best practices, he added.
Many health systems do offer video built into EMRs. Companies such as Epic have provided integrations with Vidyo, Amwell, and other platforms, noted Dr. Chan.
An integrated platform is useful if done well – but isn’t necessarily essential, according to Dr. Yellowlees. In his own setup, he signs a laptop into an EMR and uses an iPad to communicate with patients via video on the iPad and all video on the phone.
“The big advantage of telemedicine is you can type your notes in a socially appropriate way while talking to the patient on video. I do that, and it saves me a considerable amount of time. I don’t have to spend time after a consultation typing up notes.”
Still, others may struggle with bandwidth or connectivity. “Perhaps there’s a problem with privacy or the types of patients you’re dealing with,” Dr. Shore said. Older patients in a nursing home, for example, may require a team of people that works onsite. Suddenly, their care has to transition to a virtual system. “You may need to figure how to put the right team together to do straightforward and individual interactions,” he said.
Virtual care also suffers from the “digital divide,” an issue that predates COVID-19, said Dr. Shore. Not all patients have access to bandwidth and the technology to see clinicians. Other lack expertise and comfort with using the technology, or can’t afford the equipment necessary to bring them online. The pandemic has highlighted all of these disparities, he emphasized.
Dr. Chan offered that the digital divide is part of a larger socioeconomic divide, where barriers to any care – including transportation for in-person care – exist.
Technology and access issues aside, insidious “Zoom fatigue” is affecting everyone right now. “That’s clearly real,” Dr. Shore said. “And it’s not just about videoconferencing; it’s about being in quarantine. We’re all in this virtual lockdown, where people are using the term ‘videoconferencing fatigue.’ It’s a complicated problem.”
To assist with telehealth implementation, the APA has issued practice guidance and a toolkit that includes an extensive set of educational materials, including 40 videos on various topics. It’s also hosted webinars on telehealth policies and written up standard operating procedures on this topic, Dr. Yellowlees said. “The most important thing is APA, like other organizations, is advocating to have the relaxation of regulations during COVID made permanent,” he said.
Reimbursement post pandemic
As virtual visits rose in 2020, so did billing for such services. According to America’s Health Insurance Plans, claims to private insurers exploded by more than 4,000% in 2020. AHIP last July reported that mental health conditions made up one-third or more than 33% of telehealth claims to private insurers during the pandemic. Dr. Yellowlees said the health insurers he’s dealt with “have been good about paying for telehealth visits during the COVID-declared emergency.”
Whether this coverage will continue once the public health emergency ends, is unclear, some contend. “My sense is that there will be a rollback of coverage, but it won’t be back to what we had prepandemic,” said Ateev Mehrotra, MD, MPH, who studies telemedicine trends, in an interview. Right now, there’s still a great deal of uncertainty about reimbursement, “and that’s what’s giving providers pause,” said Dr. Mehrotra, of the department of health care policy and medicine at Harvard Medical School and a hospitalist at Beth Israel Deaconess Medical Center, both in Boston.
AHIP will continue to support federal and state policies to further promote telehealth access during the public health emergency, spokesman David Allen said in a statement. “Insurance providers have independently shifted their policies to increase access to care and services, ranging from acute care needs and triage services to chronic disease management and behavioral health. We are still awaiting information on what changes will remain in place beyond the public health emergency,” he said.
Even before COVID-19, Mr. Allen noted, nearly all large employers (96%) offered access to telehealth services as a covered benefit in 2019. In May, the Kaiser Family Foundation reported that many insurers were reducing or lifting cost sharing for telehealth for limited time periods.
Some of the larger payers said they’re continuing benefits, although it’s not clear how long some benefits will remain in place.
UnitedHealthcare offers no-cost coverage for COVID-19 testing–related telehealth visits, but that benefit is set to expire once the public health emergency presumably ends on Jan. 20, 2021. Through Dec. 31, 2020, the payer said it would offer coverage with no cost sharing for telehealth visits related to COVID-19 treatment and expanded access to telehealth visits not related to COVID-19 through its network. Similarly, Anthem is waiving cost sharing for COVID-19 treatments via telehealth or in-person visits, and for telehealth visits not related to COVID-19 for Medicare members through the end of the year.
Anthem will continue to cover telehealth and encourage members and clinicians to use telehealth for behavioral health, a spokesperson said. Anthem also has a telehealth provider, LiveHealth Online, “another safe and effective way for members to see a doctor to receive health guidance from their home via mobile device or a computer with a webcam,” said the spokesperson.
Dr. Vasan hopes that insurers will increase coverage for telehealth and at the same rate as in-person visits, especially for mental health. “I have not felt that the quality of the clinic has decreased, and in fact, in some ways it’s gotten better, and insurance coverage should reflect this.”
Outlook for the hybrid model
As long as there’s COVID-19, psychiatry practices must remain virtual, at least for now, Dr. Shore said. “We will emerge from this pandemic, I suspect in bits and starts.” When that happens, practices will need to have a transition plan in place. “Once we get away from COVID, I don’t think our mental health will ever be the same again. We’ll have much more virtual technology along the lines of a hybrid model, where we’ll see patients in person, but we’ll use more technology to work with patients, but it will be more of a blend.” Practices will also have to address the regulatory, reimbursement, and prescribing conditions the new world offers.
Some practices are already discovering the benefits of relying less on a brick-and-mortar office.
Dr. Chan said his colleagues are finding that they no longer have to deal with the expense and upkeep of renting and furnishing office space. “Many are taking their practice virtual-only because the monthly recurring costs are so cheap, and they can see patients in distant, underserved communities.” In-person visits are now inconvenient and risky, he continued. They require expensive personal protective equipment and cleaning protocols. “Plus, there’s the risk that services must shut down when stay-at-home orders return or when a staff member gets infected.”
Physicians prescribing certain controlled substances will likely continue to use office space, once the public health emergency expires and face-to-face visits resume. “In such cases, they can rent office space part-time,” Dr. Chan added.
Dr. Vasan, also of the department of psychiatry at Stanford and chief medical officer of Real, hopes that such a model prevails. “I do miss seeing patients in person and think a hybrid will be a good balance.”
Most patients want it, as do the influx of Generation Z physicians coming into the profession, Dr. Yellowlees noted. These are young, technologically savvy doctors who grew up in the age of the Internet. “I think the silver lining of COVID is it led telemedicine past the tipping point, where both patients and providers are learning that it’s an appropriate way to get care, as long as you’re careful, use professional guidelines – and don’t drop your standards of care.”
Dr. Yellowlees and Dr. Shore are coauthors of Telepsychiatry and Health Technologies: A Guide for Mental Health Professionals (Washington: American Psychiatric Association Publishing, 2018), and receive royalties from the book. Dr. Shore also reported working with AccessCare and receiving royalties from Springer Press. Dr. Chan reported consulting for Orbit Health. Dr. Vasan reported no conflicts of interest. Dr. Mehrotra has received research funding from several U.S. agencies, including the National Institute of Mental Health and the National Institute of Neurological Disorders and Stroke.
A call to make four telehealth provisions permanent
Lawmakers, physicians, and advocates alike have hailed a relaxation of telehealth rules under the COVID-19 emergency declaration, and they’d like things to stay this way.
Regulators previously restricted telemedicine use “by insisting that you could only see patients in the state you’re licensed in, by not reimbursing as widely for telehealth, and by not allowing us to prescribe controlled substances. They also didn’t allow us to see patients on the phone. So, there’s very good reasons to keep those regulations permanently relaxed,” said Peter Yellowlees, MBBS, MD, a professor of psychiatry and chief wellness officer at the University of California, Davis.
In his view, changes should take place in four key areas:
- Licensing. “Traditionally, state medical boards have been very insistent that clinical psychiatrists license in the state the patient resides in. This means physicians must have licenses in many different states. It’s very restrictive, because physicians can’t follow patients from one state to another. Under COVID, we can do this, but physicians want these licensing changes to be made permanent.”
- Reimbursement. “In the past, federal regulators have only allowed reimbursement for telemedicine in very specific, defined rural areas and specified clinical environments. This rule has since been relaxed, allowing us to see patients anywhere, especially in their homes. This is another area that should become permanent. Payers should continue to pay telehealth services on par with in-person visits.”
- Telephony. “Psychiatrists and other physicians haven’t been traditionally paid for telephone visits. But there’s no doubt that telephone follow-up visits can be very beneficial, so while I wouldn’t personally see a new patient on the phone, I now follow up with them on the phone once I have gotten to know them, and this works well.”
- Prescribing. The Ryan Haight Online Pharmacy Consumer Protection Act of 2008 was introduced to stop overseas pharmacies prescribing narcotics. “It was very successful, but as a side effect, it stopped most physicians from prescribing controlled substances on video. With COVID, we can now do this. For psychiatry, this is very important because it means we can use video to treat people for addictions with medications like buprenorphine and [prescribe] stimulants for children with ADHD. The U.S. Drug Enforcement Administration should finalize regulations for the Ryan Haight Act to allow for the prescribing of controlled substances via telehealth without a prior in-person exam.”
The American Psychiatric Association has called for an extension of the telehealth waiver authority under COVID-19 beyond the emergency declaration to study its impact. It will continue to advocate to allow for telephone-only telehealth to be reimbursed at the same rate as live audio-video, said a spokesperson. “We also will continue to advocate for the removal of geographic and originating site restrictions in Medicare, which prevent Medicare patients from being seen in the home,” with some exceptions, the spokesperson said.
The APA has also issued guidance to practitioners seeking clarity on telehealth coverage and COVID-19.
Lawmakers, physicians, and advocates alike have hailed a relaxation of telehealth rules under the COVID-19 emergency declaration, and they’d like things to stay this way.
Regulators previously restricted telemedicine use “by insisting that you could only see patients in the state you’re licensed in, by not reimbursing as widely for telehealth, and by not allowing us to prescribe controlled substances. They also didn’t allow us to see patients on the phone. So, there’s very good reasons to keep those regulations permanently relaxed,” said Peter Yellowlees, MBBS, MD, a professor of psychiatry and chief wellness officer at the University of California, Davis.
In his view, changes should take place in four key areas:
- Licensing. “Traditionally, state medical boards have been very insistent that clinical psychiatrists license in the state the patient resides in. This means physicians must have licenses in many different states. It’s very restrictive, because physicians can’t follow patients from one state to another. Under COVID, we can do this, but physicians want these licensing changes to be made permanent.”
- Reimbursement. “In the past, federal regulators have only allowed reimbursement for telemedicine in very specific, defined rural areas and specified clinical environments. This rule has since been relaxed, allowing us to see patients anywhere, especially in their homes. This is another area that should become permanent. Payers should continue to pay telehealth services on par with in-person visits.”
- Telephony. “Psychiatrists and other physicians haven’t been traditionally paid for telephone visits. But there’s no doubt that telephone follow-up visits can be very beneficial, so while I wouldn’t personally see a new patient on the phone, I now follow up with them on the phone once I have gotten to know them, and this works well.”
- Prescribing. The Ryan Haight Online Pharmacy Consumer Protection Act of 2008 was introduced to stop overseas pharmacies prescribing narcotics. “It was very successful, but as a side effect, it stopped most physicians from prescribing controlled substances on video. With COVID, we can now do this. For psychiatry, this is very important because it means we can use video to treat people for addictions with medications like buprenorphine and [prescribe] stimulants for children with ADHD. The U.S. Drug Enforcement Administration should finalize regulations for the Ryan Haight Act to allow for the prescribing of controlled substances via telehealth without a prior in-person exam.”
The American Psychiatric Association has called for an extension of the telehealth waiver authority under COVID-19 beyond the emergency declaration to study its impact. It will continue to advocate to allow for telephone-only telehealth to be reimbursed at the same rate as live audio-video, said a spokesperson. “We also will continue to advocate for the removal of geographic and originating site restrictions in Medicare, which prevent Medicare patients from being seen in the home,” with some exceptions, the spokesperson said.
The APA has also issued guidance to practitioners seeking clarity on telehealth coverage and COVID-19.
Lawmakers, physicians, and advocates alike have hailed a relaxation of telehealth rules under the COVID-19 emergency declaration, and they’d like things to stay this way.
Regulators previously restricted telemedicine use “by insisting that you could only see patients in the state you’re licensed in, by not reimbursing as widely for telehealth, and by not allowing us to prescribe controlled substances. They also didn’t allow us to see patients on the phone. So, there’s very good reasons to keep those regulations permanently relaxed,” said Peter Yellowlees, MBBS, MD, a professor of psychiatry and chief wellness officer at the University of California, Davis.
In his view, changes should take place in four key areas:
- Licensing. “Traditionally, state medical boards have been very insistent that clinical psychiatrists license in the state the patient resides in. This means physicians must have licenses in many different states. It’s very restrictive, because physicians can’t follow patients from one state to another. Under COVID, we can do this, but physicians want these licensing changes to be made permanent.”
- Reimbursement. “In the past, federal regulators have only allowed reimbursement for telemedicine in very specific, defined rural areas and specified clinical environments. This rule has since been relaxed, allowing us to see patients anywhere, especially in their homes. This is another area that should become permanent. Payers should continue to pay telehealth services on par with in-person visits.”
- Telephony. “Psychiatrists and other physicians haven’t been traditionally paid for telephone visits. But there’s no doubt that telephone follow-up visits can be very beneficial, so while I wouldn’t personally see a new patient on the phone, I now follow up with them on the phone once I have gotten to know them, and this works well.”
- Prescribing. The Ryan Haight Online Pharmacy Consumer Protection Act of 2008 was introduced to stop overseas pharmacies prescribing narcotics. “It was very successful, but as a side effect, it stopped most physicians from prescribing controlled substances on video. With COVID, we can now do this. For psychiatry, this is very important because it means we can use video to treat people for addictions with medications like buprenorphine and [prescribe] stimulants for children with ADHD. The U.S. Drug Enforcement Administration should finalize regulations for the Ryan Haight Act to allow for the prescribing of controlled substances via telehealth without a prior in-person exam.”
The American Psychiatric Association has called for an extension of the telehealth waiver authority under COVID-19 beyond the emergency declaration to study its impact. It will continue to advocate to allow for telephone-only telehealth to be reimbursed at the same rate as live audio-video, said a spokesperson. “We also will continue to advocate for the removal of geographic and originating site restrictions in Medicare, which prevent Medicare patients from being seen in the home,” with some exceptions, the spokesperson said.
The APA has also issued guidance to practitioners seeking clarity on telehealth coverage and COVID-19.
2020 and the telehealth boom
This year saw an unprecedented rise in medical consults over virtual platforms, as the COVID-19 pandemic raged on in the United States and worldwide.
Statistics from major health care groups and payers underscore this effect. Polling 1,004 U.S. adults this fall, the American Psychiatric Association found that 31% had used telehealth services – with 72% reporting they had ventured into this mode of care over the last 6 months.
The Coronavirus Aid, Relief, and Economic Security Act, or CARES Act, was a major catalyst, waiving geographic and Medicare telehealth payment restrictions for mental health services during certain emergency periods. Medicare beneficiaries gained access to telehealth services – they could start seeing doctors via videoconferencing in their homes, regardless of location. The Centers for Medicare and Medicaid Services began paying doctors for telehealth services at the same rate as in-office visits for all diagnoses and issued a toolkit to promote adoption of telehealth coverage policies among state Medicaid agencies.
Most states responded, expanding telehealth in Medicaid programs and relaxing restrictions on provider licensing, online prescribing, and patient consent for telehealth, the Kaiser Family Foundation reported in May. Other federal agencies took actions during the public health emergency. The Drug Enforcement Administration allowed for the prescribing of controlled substances through telemedicine, and the U.S. Department of Health & Human Services’s Office for Civil Rights agreed not to impose penalties for noncompliance of HIPAA during video conferencing, provided that physicians were acting in the best interests of the patient.
“The benefits we’re seeing on both sides – for patients and for doctors – around convenience and access are wonderful,” Nina Vasan, MD, MBA, founder and executive director of Brainstorm: The Stanford Lab for Mental Health Innovation and chair of APA’s Committee on Innovation, said in an interview. Before COVID began, only a handful of clinicians were seeing patients via televideo at Stanford, said Dr. Vasan. “Now, almost everyone is. The forced uptake and change of behavior was something we’ve needed for years, and now that it has happened, I don’t see it going away.”
This year saw an unprecedented rise in medical consults over virtual platforms, as the COVID-19 pandemic raged on in the United States and worldwide.
Statistics from major health care groups and payers underscore this effect. Polling 1,004 U.S. adults this fall, the American Psychiatric Association found that 31% had used telehealth services – with 72% reporting they had ventured into this mode of care over the last 6 months.
The Coronavirus Aid, Relief, and Economic Security Act, or CARES Act, was a major catalyst, waiving geographic and Medicare telehealth payment restrictions for mental health services during certain emergency periods. Medicare beneficiaries gained access to telehealth services – they could start seeing doctors via videoconferencing in their homes, regardless of location. The Centers for Medicare and Medicaid Services began paying doctors for telehealth services at the same rate as in-office visits for all diagnoses and issued a toolkit to promote adoption of telehealth coverage policies among state Medicaid agencies.
Most states responded, expanding telehealth in Medicaid programs and relaxing restrictions on provider licensing, online prescribing, and patient consent for telehealth, the Kaiser Family Foundation reported in May. Other federal agencies took actions during the public health emergency. The Drug Enforcement Administration allowed for the prescribing of controlled substances through telemedicine, and the U.S. Department of Health & Human Services’s Office for Civil Rights agreed not to impose penalties for noncompliance of HIPAA during video conferencing, provided that physicians were acting in the best interests of the patient.
“The benefits we’re seeing on both sides – for patients and for doctors – around convenience and access are wonderful,” Nina Vasan, MD, MBA, founder and executive director of Brainstorm: The Stanford Lab for Mental Health Innovation and chair of APA’s Committee on Innovation, said in an interview. Before COVID began, only a handful of clinicians were seeing patients via televideo at Stanford, said Dr. Vasan. “Now, almost everyone is. The forced uptake and change of behavior was something we’ve needed for years, and now that it has happened, I don’t see it going away.”
This year saw an unprecedented rise in medical consults over virtual platforms, as the COVID-19 pandemic raged on in the United States and worldwide.
Statistics from major health care groups and payers underscore this effect. Polling 1,004 U.S. adults this fall, the American Psychiatric Association found that 31% had used telehealth services – with 72% reporting they had ventured into this mode of care over the last 6 months.
The Coronavirus Aid, Relief, and Economic Security Act, or CARES Act, was a major catalyst, waiving geographic and Medicare telehealth payment restrictions for mental health services during certain emergency periods. Medicare beneficiaries gained access to telehealth services – they could start seeing doctors via videoconferencing in their homes, regardless of location. The Centers for Medicare and Medicaid Services began paying doctors for telehealth services at the same rate as in-office visits for all diagnoses and issued a toolkit to promote adoption of telehealth coverage policies among state Medicaid agencies.
Most states responded, expanding telehealth in Medicaid programs and relaxing restrictions on provider licensing, online prescribing, and patient consent for telehealth, the Kaiser Family Foundation reported in May. Other federal agencies took actions during the public health emergency. The Drug Enforcement Administration allowed for the prescribing of controlled substances through telemedicine, and the U.S. Department of Health & Human Services’s Office for Civil Rights agreed not to impose penalties for noncompliance of HIPAA during video conferencing, provided that physicians were acting in the best interests of the patient.
“The benefits we’re seeing on both sides – for patients and for doctors – around convenience and access are wonderful,” Nina Vasan, MD, MBA, founder and executive director of Brainstorm: The Stanford Lab for Mental Health Innovation and chair of APA’s Committee on Innovation, said in an interview. Before COVID began, only a handful of clinicians were seeing patients via televideo at Stanford, said Dr. Vasan. “Now, almost everyone is. The forced uptake and change of behavior was something we’ve needed for years, and now that it has happened, I don’t see it going away.”
Pigment traits, sun sensitivity associated with risk of non-Hodgkin lymphomas and chronic lymphocytic leukemia
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
FROM CANCER MEDICINE

















