User login
The Official Newspaper of the American Association for Thoracic Surgery
VIDEO: Expert discusses VATS thymectomy for myasthenia gravis
PHOENIX – In the clinical experience of Dr. Joshua R. Sonett, VATS thymectomy for myasthenia gravis is best performed in a bilateral thoracoscopic fashion.
In this approach, surgeons do about 95% of the operation on the left side to form a maximal thymectomy, “and finish taking out the specimen on the right side, making sure we can see both phrenic nerves in their entirety,” Dr. Sonett, chief of general thoracic surgery at Columbia University Medical Center, New York, said in a video interview at the annual meeting of the Society of Thoracic Surgeons.
Although there is no proof to date that thymectomy improves long-term outcomes for patients with myasthenia gravis, results from a large, international trial sponsored by the National Institutes of Health are expected to inform clinical practice about this topic, said Dr. Sonett, who is also director of the university’s high-risk lung assessment program.
Dr. Sonett reported having no relevant financial conflicts.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – In the clinical experience of Dr. Joshua R. Sonett, VATS thymectomy for myasthenia gravis is best performed in a bilateral thoracoscopic fashion.
In this approach, surgeons do about 95% of the operation on the left side to form a maximal thymectomy, “and finish taking out the specimen on the right side, making sure we can see both phrenic nerves in their entirety,” Dr. Sonett, chief of general thoracic surgery at Columbia University Medical Center, New York, said in a video interview at the annual meeting of the Society of Thoracic Surgeons.
Although there is no proof to date that thymectomy improves long-term outcomes for patients with myasthenia gravis, results from a large, international trial sponsored by the National Institutes of Health are expected to inform clinical practice about this topic, said Dr. Sonett, who is also director of the university’s high-risk lung assessment program.
Dr. Sonett reported having no relevant financial conflicts.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – In the clinical experience of Dr. Joshua R. Sonett, VATS thymectomy for myasthenia gravis is best performed in a bilateral thoracoscopic fashion.
In this approach, surgeons do about 95% of the operation on the left side to form a maximal thymectomy, “and finish taking out the specimen on the right side, making sure we can see both phrenic nerves in their entirety,” Dr. Sonett, chief of general thoracic surgery at Columbia University Medical Center, New York, said in a video interview at the annual meeting of the Society of Thoracic Surgeons.
Although there is no proof to date that thymectomy improves long-term outcomes for patients with myasthenia gravis, results from a large, international trial sponsored by the National Institutes of Health are expected to inform clinical practice about this topic, said Dr. Sonett, who is also director of the university’s high-risk lung assessment program.
Dr. Sonett reported having no relevant financial conflicts.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYIS FROM THE STS ANNUAL MEETING
STS: BMI impacts risk for complications after lung resection
PHOENIX – Being underweight is associated with a substantially increased risk of complications following lung resection for cancer, results from a large database study found.
“This is not generally known among surgeons or their patients,” Dr. Trevor Williams said in an interview before the annual meeting of the Society of Thoracic Surgeons. “Studies are conflicting about the relationship of BMI [body mass index] to surgical outcomes. Most of the previous studies simply categorize BMI as overweight or not. We’ve stratified based on World Health Organization categories to get a more precise look at BMI.”
Dr. Williams, a surgeon at the University of Chicago Medical Center, and his associates evaluated 41,446 patients in the STS General Thoracic Surgery Database who underwent elective anatomic lung resection for cancer between 2009 and 2014. Their mean age was 68 years, and 53% were female. The researchers performed multivariable analysis after adjusting for validated STS risk model covariates, including gender and spirometry.
According to WHO criteria for BMI, 3% were underweight (less than 18.5 kg/m2); 33.5% were normal weight (18.5-24.9 kg/m2); 35.4% were overweight (25-29.9 kg/m2); 18.1% were obese I (30-34.9 kg/m2); 6.4% were obese II (35-39.9 kg/m2), and 3.6% were obese III (40 kg/m2 or greater). Dr. Williams and his associates observed that women were more often underweight, compared with men (4.1% vs. 1.8%, respectively; P less than .001), and underweight patients more often had chronic obstructive pulmonary disease (51.7% vs. 35.2%; P less than .001). Pulmonary complication rates were higher among underweight and obese III patients (P less than .001), while being underweight was also associated with higher rates of infections and any surgical complications.
Multivariable analysis revealed that pulmonary and any postoperative complications were more common among underweight patients (odds ratio, 1.44 and OR, 1.41, respectively), while any major complication was more common among obese III patients (OR, 1.18). Overweight and obese I-II patients were less likely to have any postoperative and pulmonary complications, compared with patients who had a normal BMI. “The finding of underweight patients being such a high-risk patient population is suggested in the literature but not demonstrated as clearly as in this study,” Dr. Williams said. “A truly surprising finding was that obese patients actually have a lower risk of pulmonary and overall complications than ‘normal’-BMI patients.”
He concluded that according to the current analysis, “careful risk assessment is appropriate when considering operating on underweight patients. Whether there are interventions that could be instituted to improve an individual’s risk profile has not been determined. Any preconceived notions about not operating on obese patients due to elevated risk appear to be unfounded.”
Dr. Williams reported having no financial disclosures.
PHOENIX – Being underweight is associated with a substantially increased risk of complications following lung resection for cancer, results from a large database study found.
“This is not generally known among surgeons or their patients,” Dr. Trevor Williams said in an interview before the annual meeting of the Society of Thoracic Surgeons. “Studies are conflicting about the relationship of BMI [body mass index] to surgical outcomes. Most of the previous studies simply categorize BMI as overweight or not. We’ve stratified based on World Health Organization categories to get a more precise look at BMI.”
Dr. Williams, a surgeon at the University of Chicago Medical Center, and his associates evaluated 41,446 patients in the STS General Thoracic Surgery Database who underwent elective anatomic lung resection for cancer between 2009 and 2014. Their mean age was 68 years, and 53% were female. The researchers performed multivariable analysis after adjusting for validated STS risk model covariates, including gender and spirometry.
According to WHO criteria for BMI, 3% were underweight (less than 18.5 kg/m2); 33.5% were normal weight (18.5-24.9 kg/m2); 35.4% were overweight (25-29.9 kg/m2); 18.1% were obese I (30-34.9 kg/m2); 6.4% were obese II (35-39.9 kg/m2), and 3.6% were obese III (40 kg/m2 or greater). Dr. Williams and his associates observed that women were more often underweight, compared with men (4.1% vs. 1.8%, respectively; P less than .001), and underweight patients more often had chronic obstructive pulmonary disease (51.7% vs. 35.2%; P less than .001). Pulmonary complication rates were higher among underweight and obese III patients (P less than .001), while being underweight was also associated with higher rates of infections and any surgical complications.
Multivariable analysis revealed that pulmonary and any postoperative complications were more common among underweight patients (odds ratio, 1.44 and OR, 1.41, respectively), while any major complication was more common among obese III patients (OR, 1.18). Overweight and obese I-II patients were less likely to have any postoperative and pulmonary complications, compared with patients who had a normal BMI. “The finding of underweight patients being such a high-risk patient population is suggested in the literature but not demonstrated as clearly as in this study,” Dr. Williams said. “A truly surprising finding was that obese patients actually have a lower risk of pulmonary and overall complications than ‘normal’-BMI patients.”
He concluded that according to the current analysis, “careful risk assessment is appropriate when considering operating on underweight patients. Whether there are interventions that could be instituted to improve an individual’s risk profile has not been determined. Any preconceived notions about not operating on obese patients due to elevated risk appear to be unfounded.”
Dr. Williams reported having no financial disclosures.
PHOENIX – Being underweight is associated with a substantially increased risk of complications following lung resection for cancer, results from a large database study found.
“This is not generally known among surgeons or their patients,” Dr. Trevor Williams said in an interview before the annual meeting of the Society of Thoracic Surgeons. “Studies are conflicting about the relationship of BMI [body mass index] to surgical outcomes. Most of the previous studies simply categorize BMI as overweight or not. We’ve stratified based on World Health Organization categories to get a more precise look at BMI.”
Dr. Williams, a surgeon at the University of Chicago Medical Center, and his associates evaluated 41,446 patients in the STS General Thoracic Surgery Database who underwent elective anatomic lung resection for cancer between 2009 and 2014. Their mean age was 68 years, and 53% were female. The researchers performed multivariable analysis after adjusting for validated STS risk model covariates, including gender and spirometry.
According to WHO criteria for BMI, 3% were underweight (less than 18.5 kg/m2); 33.5% were normal weight (18.5-24.9 kg/m2); 35.4% were overweight (25-29.9 kg/m2); 18.1% were obese I (30-34.9 kg/m2); 6.4% were obese II (35-39.9 kg/m2), and 3.6% were obese III (40 kg/m2 or greater). Dr. Williams and his associates observed that women were more often underweight, compared with men (4.1% vs. 1.8%, respectively; P less than .001), and underweight patients more often had chronic obstructive pulmonary disease (51.7% vs. 35.2%; P less than .001). Pulmonary complication rates were higher among underweight and obese III patients (P less than .001), while being underweight was also associated with higher rates of infections and any surgical complications.
Multivariable analysis revealed that pulmonary and any postoperative complications were more common among underweight patients (odds ratio, 1.44 and OR, 1.41, respectively), while any major complication was more common among obese III patients (OR, 1.18). Overweight and obese I-II patients were less likely to have any postoperative and pulmonary complications, compared with patients who had a normal BMI. “The finding of underweight patients being such a high-risk patient population is suggested in the literature but not demonstrated as clearly as in this study,” Dr. Williams said. “A truly surprising finding was that obese patients actually have a lower risk of pulmonary and overall complications than ‘normal’-BMI patients.”
He concluded that according to the current analysis, “careful risk assessment is appropriate when considering operating on underweight patients. Whether there are interventions that could be instituted to improve an individual’s risk profile has not been determined. Any preconceived notions about not operating on obese patients due to elevated risk appear to be unfounded.”
Dr. Williams reported having no financial disclosures.
AT THE STS ANNUAL MEETING
Key clinical point: Careful risk assessment is appropriate when considering performing lung resection on underweight patients.
Major finding: Multivariable analysis revealed that pulmonary and any postoperative complications were more common among underweight patients (OR, 1.44 and OR, 1.41, respectively), while any major complication was more common among obese III patients (OR, 1.18).
Data source: An analysis of 41,446 patients in the STS General Thoracic Surgery Database who underwent elective lung resection for cancer between 2009 and 2014.
Disclosures: Dr. Williams reported having no financial disclosures.
VIDEO: Novel imaging technique helps hunt for pulmonary lesions
PHOENIX – Each year more than 250,000 patients present with ground-glass opacities and other solitary pulmonary nodules, and they are difficult to locate.
“There’s been a need for our field to develop new technologies to find these nodules in the OR,” Dr. Sunil Singhal said in a video interview at the annual meeting of the Society of Thoracic Surgeons. “The fallback plan has always been that we can make a thoracotomy. Some studies have shown that in about one out of every two cases you end up opening a patient just to find a tiny little nodule.”
Dr. Singhal of the division of cardiothoracic surgery at the University of Pennsylvania School of Medicine, Philadelphia, discussed preoperative and intraoperative localization methods, including an investigational technology in which patients receive an intravascular dye that localizes the pulmonary tumor. “When we put our video-assisted thoracoscopic surgery camera in, the tumors are glowing,” he said. “We can then do a localized wedge excision and confirm margins of the staple line. We’ve done this [in] about 80 patients, and it’s been non-toxic, very safe, and very effective. Our biggest limitation has been our depth of penetration.”
Dr. Singhal reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – Each year more than 250,000 patients present with ground-glass opacities and other solitary pulmonary nodules, and they are difficult to locate.
“There’s been a need for our field to develop new technologies to find these nodules in the OR,” Dr. Sunil Singhal said in a video interview at the annual meeting of the Society of Thoracic Surgeons. “The fallback plan has always been that we can make a thoracotomy. Some studies have shown that in about one out of every two cases you end up opening a patient just to find a tiny little nodule.”
Dr. Singhal of the division of cardiothoracic surgery at the University of Pennsylvania School of Medicine, Philadelphia, discussed preoperative and intraoperative localization methods, including an investigational technology in which patients receive an intravascular dye that localizes the pulmonary tumor. “When we put our video-assisted thoracoscopic surgery camera in, the tumors are glowing,” he said. “We can then do a localized wedge excision and confirm margins of the staple line. We’ve done this [in] about 80 patients, and it’s been non-toxic, very safe, and very effective. Our biggest limitation has been our depth of penetration.”
Dr. Singhal reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – Each year more than 250,000 patients present with ground-glass opacities and other solitary pulmonary nodules, and they are difficult to locate.
“There’s been a need for our field to develop new technologies to find these nodules in the OR,” Dr. Sunil Singhal said in a video interview at the annual meeting of the Society of Thoracic Surgeons. “The fallback plan has always been that we can make a thoracotomy. Some studies have shown that in about one out of every two cases you end up opening a patient just to find a tiny little nodule.”
Dr. Singhal of the division of cardiothoracic surgery at the University of Pennsylvania School of Medicine, Philadelphia, discussed preoperative and intraoperative localization methods, including an investigational technology in which patients receive an intravascular dye that localizes the pulmonary tumor. “When we put our video-assisted thoracoscopic surgery camera in, the tumors are glowing,” he said. “We can then do a localized wedge excision and confirm margins of the staple line. We’ve done this [in] about 80 patients, and it’s been non-toxic, very safe, and very effective. Our biggest limitation has been our depth of penetration.”
Dr. Singhal reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE STS ANNUAL MEETING
VIDEO: One in five hospital patients get health care–acquired infection
PHOENIX – If you happen to believe that the impact of health care–acquired infections is insignificant, think again. According to Dr. Kevin W. Lobdell, health care–acquired infections (HAIs) cause more deaths each year in the United States than breast cancer, lung cancer, and AIDS combined.
“If you look at hospitalized patients, one in five will acquire a health care–acquired infection,” Dr. Lobdell of the Sanger Heart and Vascular Institute at Carolinas Health System, Charlotte, N.C., said in a video interview at the annual meeting of the Society of Thoracic Surgeons. “With respect to length of stay, that goes from 5 days on average for a normal, uninfected patient, to 22 days if they’ve had an infection. The mortality rate can be as high as 6% in those people that have developed infections, so that in itself is an enormous burden.”
He went on to discuss the most common HAIs in the hospital setting and noted that combating them involves strategies that consider people, the environment, and technology. He predicted that in coming years clinicians will have a better “analytic capability to understand what we’ve done in the past and what correlates with success in the future, and then be able to implement and learn from that.”
Dr. Lobdell reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – If you happen to believe that the impact of health care–acquired infections is insignificant, think again. According to Dr. Kevin W. Lobdell, health care–acquired infections (HAIs) cause more deaths each year in the United States than breast cancer, lung cancer, and AIDS combined.
“If you look at hospitalized patients, one in five will acquire a health care–acquired infection,” Dr. Lobdell of the Sanger Heart and Vascular Institute at Carolinas Health System, Charlotte, N.C., said in a video interview at the annual meeting of the Society of Thoracic Surgeons. “With respect to length of stay, that goes from 5 days on average for a normal, uninfected patient, to 22 days if they’ve had an infection. The mortality rate can be as high as 6% in those people that have developed infections, so that in itself is an enormous burden.”
He went on to discuss the most common HAIs in the hospital setting and noted that combating them involves strategies that consider people, the environment, and technology. He predicted that in coming years clinicians will have a better “analytic capability to understand what we’ve done in the past and what correlates with success in the future, and then be able to implement and learn from that.”
Dr. Lobdell reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – If you happen to believe that the impact of health care–acquired infections is insignificant, think again. According to Dr. Kevin W. Lobdell, health care–acquired infections (HAIs) cause more deaths each year in the United States than breast cancer, lung cancer, and AIDS combined.
“If you look at hospitalized patients, one in five will acquire a health care–acquired infection,” Dr. Lobdell of the Sanger Heart and Vascular Institute at Carolinas Health System, Charlotte, N.C., said in a video interview at the annual meeting of the Society of Thoracic Surgeons. “With respect to length of stay, that goes from 5 days on average for a normal, uninfected patient, to 22 days if they’ve had an infection. The mortality rate can be as high as 6% in those people that have developed infections, so that in itself is an enormous burden.”
He went on to discuss the most common HAIs in the hospital setting and noted that combating them involves strategies that consider people, the environment, and technology. He predicted that in coming years clinicians will have a better “analytic capability to understand what we’ve done in the past and what correlates with success in the future, and then be able to implement and learn from that.”
Dr. Lobdell reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE STS ANNUAL MEETING
VIDEO: Preventing healthcare acquired infections after CT surgery
PHOENIX – More and more attention is being paid to preventing healthcare acquired infections (HAIs) in the hospital setting, and the role of HAIs in cardiothoracic surgery is a particlularly important area of focus.
“The good news is that cardiothoracic surgeons are really good at preventing infections. There’s been a lot of pressure over the past many years to report infections after cardiothoracic surgery, and so they’ve gotten a lot of things right,” Dr. Emily Landon said in a video interview at the annual meeting of the Society of Thoracic Surgeons.
“However, patients that undergo cardiothoracic surgery are still at risk of the infections that plague everyone in hospitals ... all of these are a problem based on whatever the hospital’s current situation is.”
Dr. Landon, who is the medical director of antimicrobial stewardship and infection control at University of Chicago Medicine, Chicago, discussed how cardiothroacic surgeons can maintain their own good outcomes and how they can have a postive impact outside the OR on protecting their patients after surgery.
Dr. Landon reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – More and more attention is being paid to preventing healthcare acquired infections (HAIs) in the hospital setting, and the role of HAIs in cardiothoracic surgery is a particlularly important area of focus.
“The good news is that cardiothoracic surgeons are really good at preventing infections. There’s been a lot of pressure over the past many years to report infections after cardiothoracic surgery, and so they’ve gotten a lot of things right,” Dr. Emily Landon said in a video interview at the annual meeting of the Society of Thoracic Surgeons.
“However, patients that undergo cardiothoracic surgery are still at risk of the infections that plague everyone in hospitals ... all of these are a problem based on whatever the hospital’s current situation is.”
Dr. Landon, who is the medical director of antimicrobial stewardship and infection control at University of Chicago Medicine, Chicago, discussed how cardiothroacic surgeons can maintain their own good outcomes and how they can have a postive impact outside the OR on protecting their patients after surgery.
Dr. Landon reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PHOENIX – More and more attention is being paid to preventing healthcare acquired infections (HAIs) in the hospital setting, and the role of HAIs in cardiothoracic surgery is a particlularly important area of focus.
“The good news is that cardiothoracic surgeons are really good at preventing infections. There’s been a lot of pressure over the past many years to report infections after cardiothoracic surgery, and so they’ve gotten a lot of things right,” Dr. Emily Landon said in a video interview at the annual meeting of the Society of Thoracic Surgeons.
“However, patients that undergo cardiothoracic surgery are still at risk of the infections that plague everyone in hospitals ... all of these are a problem based on whatever the hospital’s current situation is.”
Dr. Landon, who is the medical director of antimicrobial stewardship and infection control at University of Chicago Medicine, Chicago, discussed how cardiothroacic surgeons can maintain their own good outcomes and how they can have a postive impact outside the OR on protecting their patients after surgery.
Dr. Landon reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE STS ANNUAL MEETING
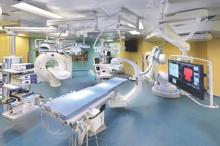
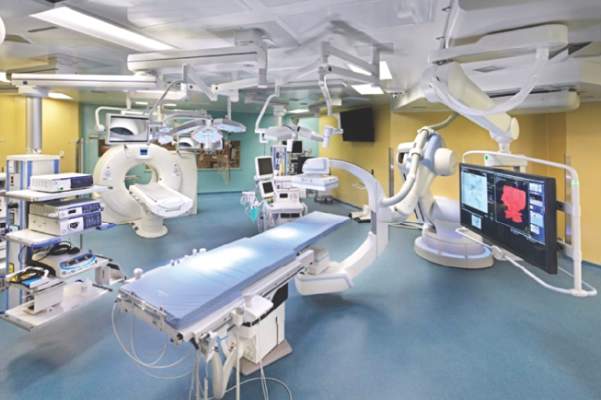
STS: Hybrid thoracic suite leverages CT’s imaging sensitivity
PHOENIX – Using CT imaging to detect lung cancers in people at high risk for developing it has made it possible to find small tumors with substantially increased sensitivity than is possible with radiography, However, this approach has posed a new challenge to thoracic surgeons: How to visualize these nodules – subcentimeter and nonpalpable – for biopsy or for resection?
The answer may be the hybrid thoracic operating room developed by Dr. Kazuhiro Yasufuku and his associates at Toronto General Hospital, a novel surgical suite that he described at the annual meeting of the Society of Thoracic Surgeons.
Dr. Yasufuku and his team began using the hybrid operating room on an investigational basis in 2013 and have now done about 50 cases as part of several research protocols. The trials address the feasibility of resection, biopsy, and nodule localization, as well as whether the hybrid approach reduces the amount of radiation exposure to both patients and to the surgical team, he said. They plan to report some of their initial results later this year.
The Toronto group assembled the hybrid array of equipment into a single operating room that includes both a dual-source, dual-energy CT scanner and a robotic cone-beam CT scanner, equipment for minimally invasive procedures including video-assisted thoracoscopic and robotic surgery, and advanced endoscopic technology including endobronchial ultrasound and navigational bronchoscopy. “We use innovative methods that we already know about, but bring them all together” within a single space, Dr. Yasufuku explained. “Rather than having patients go to several locations, we can do everything at the same time in one room.”
Perhaps the most novel aspect of this operating room is inclusion of a robotic cone-beam CT scanner, which uses mobile, flat CT-imaging panels that overcome the limitations of a conventional, fixed CT scanner. “They scan the patient and then we can retract them and get them out of the way” to better facilitate surgery, he said in an interview.
“We do not have a culture in thoracic surgery of using imaging during surgery,” said Dr. Yasufuku, director of the interventional thoracic surgery program at the University of Toronto. Hybrid operating rooms using noninvasive or minimally invasive equipment and procedures have become commonplace for cardiovascular surgeons and cardiac interventionalists, but this approach has generally not yet been applied to thoracic surgery for cancer, in large part because of the imaging limitations, he said. “It is difficult to perform video-assisted thorascopic surgery using fixed CT.”
Bronchoscopic technologies provide additional, important tools for minimally invasive thoracic surgery. “We use the hybrid operating room to mark small [nonpalpable] lesions.” One approach to marking is to place a microcoil within the nodule with a percutaneous needle. Another approach is to tag the nodule with a fluorescent dye using navigational bronchoscopy.
Dr. Yasufuku also emphasized that the hybrid operating room will also be valuable when new, minimally invasive, nonsurgical therapeutic options for treatment of lung cancer become available in the near future.
Dr. Yasufuku said that he had no relevant disclosures.
On Twitter @mitchelzoler
PHOENIX – Using CT imaging to detect lung cancers in people at high risk for developing it has made it possible to find small tumors with substantially increased sensitivity than is possible with radiography, However, this approach has posed a new challenge to thoracic surgeons: How to visualize these nodules – subcentimeter and nonpalpable – for biopsy or for resection?
The answer may be the hybrid thoracic operating room developed by Dr. Kazuhiro Yasufuku and his associates at Toronto General Hospital, a novel surgical suite that he described at the annual meeting of the Society of Thoracic Surgeons.
Dr. Yasufuku and his team began using the hybrid operating room on an investigational basis in 2013 and have now done about 50 cases as part of several research protocols. The trials address the feasibility of resection, biopsy, and nodule localization, as well as whether the hybrid approach reduces the amount of radiation exposure to both patients and to the surgical team, he said. They plan to report some of their initial results later this year.
The Toronto group assembled the hybrid array of equipment into a single operating room that includes both a dual-source, dual-energy CT scanner and a robotic cone-beam CT scanner, equipment for minimally invasive procedures including video-assisted thoracoscopic and robotic surgery, and advanced endoscopic technology including endobronchial ultrasound and navigational bronchoscopy. “We use innovative methods that we already know about, but bring them all together” within a single space, Dr. Yasufuku explained. “Rather than having patients go to several locations, we can do everything at the same time in one room.”
Perhaps the most novel aspect of this operating room is inclusion of a robotic cone-beam CT scanner, which uses mobile, flat CT-imaging panels that overcome the limitations of a conventional, fixed CT scanner. “They scan the patient and then we can retract them and get them out of the way” to better facilitate surgery, he said in an interview.
“We do not have a culture in thoracic surgery of using imaging during surgery,” said Dr. Yasufuku, director of the interventional thoracic surgery program at the University of Toronto. Hybrid operating rooms using noninvasive or minimally invasive equipment and procedures have become commonplace for cardiovascular surgeons and cardiac interventionalists, but this approach has generally not yet been applied to thoracic surgery for cancer, in large part because of the imaging limitations, he said. “It is difficult to perform video-assisted thorascopic surgery using fixed CT.”
Bronchoscopic technologies provide additional, important tools for minimally invasive thoracic surgery. “We use the hybrid operating room to mark small [nonpalpable] lesions.” One approach to marking is to place a microcoil within the nodule with a percutaneous needle. Another approach is to tag the nodule with a fluorescent dye using navigational bronchoscopy.
Dr. Yasufuku also emphasized that the hybrid operating room will also be valuable when new, minimally invasive, nonsurgical therapeutic options for treatment of lung cancer become available in the near future.
Dr. Yasufuku said that he had no relevant disclosures.
On Twitter @mitchelzoler
PHOENIX – Using CT imaging to detect lung cancers in people at high risk for developing it has made it possible to find small tumors with substantially increased sensitivity than is possible with radiography, However, this approach has posed a new challenge to thoracic surgeons: How to visualize these nodules – subcentimeter and nonpalpable – for biopsy or for resection?
The answer may be the hybrid thoracic operating room developed by Dr. Kazuhiro Yasufuku and his associates at Toronto General Hospital, a novel surgical suite that he described at the annual meeting of the Society of Thoracic Surgeons.
Dr. Yasufuku and his team began using the hybrid operating room on an investigational basis in 2013 and have now done about 50 cases as part of several research protocols. The trials address the feasibility of resection, biopsy, and nodule localization, as well as whether the hybrid approach reduces the amount of radiation exposure to both patients and to the surgical team, he said. They plan to report some of their initial results later this year.
The Toronto group assembled the hybrid array of equipment into a single operating room that includes both a dual-source, dual-energy CT scanner and a robotic cone-beam CT scanner, equipment for minimally invasive procedures including video-assisted thoracoscopic and robotic surgery, and advanced endoscopic technology including endobronchial ultrasound and navigational bronchoscopy. “We use innovative methods that we already know about, but bring them all together” within a single space, Dr. Yasufuku explained. “Rather than having patients go to several locations, we can do everything at the same time in one room.”
Perhaps the most novel aspect of this operating room is inclusion of a robotic cone-beam CT scanner, which uses mobile, flat CT-imaging panels that overcome the limitations of a conventional, fixed CT scanner. “They scan the patient and then we can retract them and get them out of the way” to better facilitate surgery, he said in an interview.
“We do not have a culture in thoracic surgery of using imaging during surgery,” said Dr. Yasufuku, director of the interventional thoracic surgery program at the University of Toronto. Hybrid operating rooms using noninvasive or minimally invasive equipment and procedures have become commonplace for cardiovascular surgeons and cardiac interventionalists, but this approach has generally not yet been applied to thoracic surgery for cancer, in large part because of the imaging limitations, he said. “It is difficult to perform video-assisted thorascopic surgery using fixed CT.”
Bronchoscopic technologies provide additional, important tools for minimally invasive thoracic surgery. “We use the hybrid operating room to mark small [nonpalpable] lesions.” One approach to marking is to place a microcoil within the nodule with a percutaneous needle. Another approach is to tag the nodule with a fluorescent dye using navigational bronchoscopy.
Dr. Yasufuku also emphasized that the hybrid operating room will also be valuable when new, minimally invasive, nonsurgical therapeutic options for treatment of lung cancer become available in the near future.
Dr. Yasufuku said that he had no relevant disclosures.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE STS ANNUAL MEETING
VIDEO: Which lesions are best for bronchoscopic endoluminal treatment?
PHOENIX – According to Dr. Moishe Liberman, promising lesions for bronchoscopic endoluminal treatment include endobronchial lesions and intraluminal exophytic tumors within the trachea or main bronchus, provided that the distal airway lumen is visible and you can get past the tumor with a flexible endoscope.
“We always teach the fellows that if you get pus back when you’re trying to get around the tumor or play with the tumor, you’re usually going to have a very good result,” said Dr. Liberman, a thoracic surgeon who directs the endoscopic tracheo-bronchial and oesophageal center at the Centre hospitalier de l’Université de Montréal, Quebec, Canada. “If you play with the tumor and you get the tumor out and you get nothing back, usually the CT scan or the X-ray postoperatively is going to look just like it did preoperatively, even though endoscopically you might have a good result.”
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Central lesions are also excellent candidates for endoluminal therapy, he said in a video interview at the meeting. Distal lesions in the small bronchi “are candidates but are much more difficult and require more specialized tools. The shorter the lesion, the more likely you are to have good success.”
Available options for delivering energy endoscopically include electrocautery, argon plasma coagulation, laser, and cryotherapy. A disadvantage of all of the thermal modalities except for cryotherapy “include the potential for airway fire and you have to work with low FiO2s [fraction of inspired oxygen],” Dr. Liberman noted. “A lot of these patients need high FiO2s to saturate, so I think that’s always an issue. We never go on cardiopulmonary bypass to do these cases and we never cannulate patients to do these cases. You also have to worry about gas emboli, especially when you open up big vessels. These modalities can also cause inadvertent airway injury, delayed effects, and bronchoscope damage.”
In general, he continued, laser-tissue interactions depend on the power and the wavelength of the laser as well as the color and the water content of the target tissue. “The power density of the wavelength you choose determines its ability to cut, coagulate, or vaporize the tissue,” he said.
“As the power density increases, the laser fiber approaches the target tissue. Power density is more important than the energy delivered.”
The Nd:YAG (neodymium-doped yttrium aluminium garnet) laser, which causes more destruction in the deep tissue than on the surface, is the most common laser used in interventional airway procedures, he said. Two other commonly used lasers include the KTP (potassium titanyl phosphate) and the CO2. “I like CO2s a lot for upper airway and subglottic problems as well as vocal cord problems,” Dr. Liberman said. “It’s very precise and has low penetration. The Nd:YAG is very good for deep penetration. You need familiarity with these. I don’t think you can just take one of these off the shelf if you’ve never used it before. Sometimes your ENT [ear nose and throat] or urology colleagues can help you, because they’re using a lot more of these lasers than we are.”
Contraindications for laser bronchoscopy include operable lesions. Dr. Liberman said that while he and his associates use lasers in a preoperative setting, “we’re very careful not to damage proximal or distal airway when we know we’re going to do a sleeve resection or pneumonectomy.”
Other contraindications for laser bronchoscopy include patients with a poor short-term prognosis, severe coagulation disorder, extrinsic airway obstruction, tracheoesophageal fistula or T-Med fistula, those with extensive submucosal disease causing obstruction, and those with lesion adjacent to the esophagus or to a major vessel.
Dr. Liberman reported having received research grants from Ethicon, Boston Scientific, Olympus, Covidien, and Baxter.
PHOENIX – According to Dr. Moishe Liberman, promising lesions for bronchoscopic endoluminal treatment include endobronchial lesions and intraluminal exophytic tumors within the trachea or main bronchus, provided that the distal airway lumen is visible and you can get past the tumor with a flexible endoscope.
“We always teach the fellows that if you get pus back when you’re trying to get around the tumor or play with the tumor, you’re usually going to have a very good result,” said Dr. Liberman, a thoracic surgeon who directs the endoscopic tracheo-bronchial and oesophageal center at the Centre hospitalier de l’Université de Montréal, Quebec, Canada. “If you play with the tumor and you get the tumor out and you get nothing back, usually the CT scan or the X-ray postoperatively is going to look just like it did preoperatively, even though endoscopically you might have a good result.”
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Central lesions are also excellent candidates for endoluminal therapy, he said in a video interview at the meeting. Distal lesions in the small bronchi “are candidates but are much more difficult and require more specialized tools. The shorter the lesion, the more likely you are to have good success.”
Available options for delivering energy endoscopically include electrocautery, argon plasma coagulation, laser, and cryotherapy. A disadvantage of all of the thermal modalities except for cryotherapy “include the potential for airway fire and you have to work with low FiO2s [fraction of inspired oxygen],” Dr. Liberman noted. “A lot of these patients need high FiO2s to saturate, so I think that’s always an issue. We never go on cardiopulmonary bypass to do these cases and we never cannulate patients to do these cases. You also have to worry about gas emboli, especially when you open up big vessels. These modalities can also cause inadvertent airway injury, delayed effects, and bronchoscope damage.”
In general, he continued, laser-tissue interactions depend on the power and the wavelength of the laser as well as the color and the water content of the target tissue. “The power density of the wavelength you choose determines its ability to cut, coagulate, or vaporize the tissue,” he said.
“As the power density increases, the laser fiber approaches the target tissue. Power density is more important than the energy delivered.”
The Nd:YAG (neodymium-doped yttrium aluminium garnet) laser, which causes more destruction in the deep tissue than on the surface, is the most common laser used in interventional airway procedures, he said. Two other commonly used lasers include the KTP (potassium titanyl phosphate) and the CO2. “I like CO2s a lot for upper airway and subglottic problems as well as vocal cord problems,” Dr. Liberman said. “It’s very precise and has low penetration. The Nd:YAG is very good for deep penetration. You need familiarity with these. I don’t think you can just take one of these off the shelf if you’ve never used it before. Sometimes your ENT [ear nose and throat] or urology colleagues can help you, because they’re using a lot more of these lasers than we are.”
Contraindications for laser bronchoscopy include operable lesions. Dr. Liberman said that while he and his associates use lasers in a preoperative setting, “we’re very careful not to damage proximal or distal airway when we know we’re going to do a sleeve resection or pneumonectomy.”
Other contraindications for laser bronchoscopy include patients with a poor short-term prognosis, severe coagulation disorder, extrinsic airway obstruction, tracheoesophageal fistula or T-Med fistula, those with extensive submucosal disease causing obstruction, and those with lesion adjacent to the esophagus or to a major vessel.
Dr. Liberman reported having received research grants from Ethicon, Boston Scientific, Olympus, Covidien, and Baxter.
PHOENIX – According to Dr. Moishe Liberman, promising lesions for bronchoscopic endoluminal treatment include endobronchial lesions and intraluminal exophytic tumors within the trachea or main bronchus, provided that the distal airway lumen is visible and you can get past the tumor with a flexible endoscope.
“We always teach the fellows that if you get pus back when you’re trying to get around the tumor or play with the tumor, you’re usually going to have a very good result,” said Dr. Liberman, a thoracic surgeon who directs the endoscopic tracheo-bronchial and oesophageal center at the Centre hospitalier de l’Université de Montréal, Quebec, Canada. “If you play with the tumor and you get the tumor out and you get nothing back, usually the CT scan or the X-ray postoperatively is going to look just like it did preoperatively, even though endoscopically you might have a good result.”
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Central lesions are also excellent candidates for endoluminal therapy, he said in a video interview at the meeting. Distal lesions in the small bronchi “are candidates but are much more difficult and require more specialized tools. The shorter the lesion, the more likely you are to have good success.”
Available options for delivering energy endoscopically include electrocautery, argon plasma coagulation, laser, and cryotherapy. A disadvantage of all of the thermal modalities except for cryotherapy “include the potential for airway fire and you have to work with low FiO2s [fraction of inspired oxygen],” Dr. Liberman noted. “A lot of these patients need high FiO2s to saturate, so I think that’s always an issue. We never go on cardiopulmonary bypass to do these cases and we never cannulate patients to do these cases. You also have to worry about gas emboli, especially when you open up big vessels. These modalities can also cause inadvertent airway injury, delayed effects, and bronchoscope damage.”
In general, he continued, laser-tissue interactions depend on the power and the wavelength of the laser as well as the color and the water content of the target tissue. “The power density of the wavelength you choose determines its ability to cut, coagulate, or vaporize the tissue,” he said.
“As the power density increases, the laser fiber approaches the target tissue. Power density is more important than the energy delivered.”
The Nd:YAG (neodymium-doped yttrium aluminium garnet) laser, which causes more destruction in the deep tissue than on the surface, is the most common laser used in interventional airway procedures, he said. Two other commonly used lasers include the KTP (potassium titanyl phosphate) and the CO2. “I like CO2s a lot for upper airway and subglottic problems as well as vocal cord problems,” Dr. Liberman said. “It’s very precise and has low penetration. The Nd:YAG is very good for deep penetration. You need familiarity with these. I don’t think you can just take one of these off the shelf if you’ve never used it before. Sometimes your ENT [ear nose and throat] or urology colleagues can help you, because they’re using a lot more of these lasers than we are.”
Contraindications for laser bronchoscopy include operable lesions. Dr. Liberman said that while he and his associates use lasers in a preoperative setting, “we’re very careful not to damage proximal or distal airway when we know we’re going to do a sleeve resection or pneumonectomy.”
Other contraindications for laser bronchoscopy include patients with a poor short-term prognosis, severe coagulation disorder, extrinsic airway obstruction, tracheoesophageal fistula or T-Med fistula, those with extensive submucosal disease causing obstruction, and those with lesion adjacent to the esophagus or to a major vessel.
Dr. Liberman reported having received research grants from Ethicon, Boston Scientific, Olympus, Covidien, and Baxter.
EXPERT ANALYSIS FROM THE STS ANNUAL MEETING
FDA approves marketing of sutureless aortic valve
LivaNova announced that the U.S. Food and Drug Administration has announced the approval of their Perceval Sutureless Heart Valve. The approval was issued Jan. 8 and is effective immediately, and LivaNova indicated that it will begin commercial distribution of the device in the United States over the coming quarter.
According to an FDA summary document, the Perceval Sutureless Heart Valve is a bioprosthetic valve designed to replace a diseased native or a malfunctioning prosthetic aortic valve via open heart surgery. The self-expanding stent frame consists of a tissue component made from bovine pericardium and a self-expandable nitinol stent, along with a dedicated delivery system that allows physicians to position and anchor the valve suturelessly.
The Perceval heart valve is supplied unmounted and must be loaded onto an accompanying holder by reducing the valve size using a supplied polycarbonate collapser. The holder is used for sternal approaches and includes a rigid shaft with an end section that houses the valve prosthesis during delivery. A separate holder, approximately 3 cm longer, is also available for minimally invasive procedures. After implantation, the physician uses the a post-dilation catheter to expand the valve in situ.
Perceval’s approval was based upon the results of a pivotal European study performed to assess the device, the CAVALIER (Safety and Effectiveness Study of Perceval S Valve for Extended CE Mark) trial. The prospective, multicenter, nonrandomized clinical study was conducted at 26 investigational sites in Austria, Belgium, England, France, Germany, and the Netherlands.
Patients were treated between Feb. 23, 2010, and Sept. 30, 2013, and the database for the PDA-assessed data collected through Nov. 5, 2014, and included 658 patients.
The differences between the New York Heart Association (NYHA) class at 12 months and the baseline were calculated. In total, 77.5% of patients showed a decrease of NYHA equal to at least one class, whereas 19.7% of patients remained stable over the time. Only 2.8% of patients showed a worsened clinical status.
Reduction in mean gradients and increase in effective orifice area were both observed at 1 year follow-up, according to the FDA report.
At the 1-, 2-, and 3-year follow-up time points, 75.1% or greater of the implanted patients with available data had improved by one to three classes and at the 4-year follow-up time point 72.6% of the patients had improved by one to three classes, according to the report. In addition, at the 1-, 2-, and 3-year follow-up time points, 92.9% or greater of the patients were in NYHA Class I and Class II, and at the 4-year follow-up time point, 86.3% of the patients were in NYHA Class I and Class II.
Although there was a predominance of women in the study population (64.4%), patients of both sexes demonstrated acceptable hemodynamic outcomes and significant improvement in functional status, according to the FDA summary.
Sutureless valves are considered to have a promising future according to a recent international consensus panel recommendationpublished online in the European Journal of Cardio-Thoracic Surgery (2015 Oct 29. doi: 10.1093/ejcts/ezv369). The report assessed various benefits of sutureless and rapid deployment technology, and concluded these devices “may represent a helpful tool in aortic valve replacement for patients requiring a biological valve.” However, further evidence will be needed to reaffirm the benefit of sutureless and rapid deployment valves, they concluded.
LivaNova announced that the U.S. Food and Drug Administration has announced the approval of their Perceval Sutureless Heart Valve. The approval was issued Jan. 8 and is effective immediately, and LivaNova indicated that it will begin commercial distribution of the device in the United States over the coming quarter.
According to an FDA summary document, the Perceval Sutureless Heart Valve is a bioprosthetic valve designed to replace a diseased native or a malfunctioning prosthetic aortic valve via open heart surgery. The self-expanding stent frame consists of a tissue component made from bovine pericardium and a self-expandable nitinol stent, along with a dedicated delivery system that allows physicians to position and anchor the valve suturelessly.
The Perceval heart valve is supplied unmounted and must be loaded onto an accompanying holder by reducing the valve size using a supplied polycarbonate collapser. The holder is used for sternal approaches and includes a rigid shaft with an end section that houses the valve prosthesis during delivery. A separate holder, approximately 3 cm longer, is also available for minimally invasive procedures. After implantation, the physician uses the a post-dilation catheter to expand the valve in situ.
Perceval’s approval was based upon the results of a pivotal European study performed to assess the device, the CAVALIER (Safety and Effectiveness Study of Perceval S Valve for Extended CE Mark) trial. The prospective, multicenter, nonrandomized clinical study was conducted at 26 investigational sites in Austria, Belgium, England, France, Germany, and the Netherlands.
Patients were treated between Feb. 23, 2010, and Sept. 30, 2013, and the database for the PDA-assessed data collected through Nov. 5, 2014, and included 658 patients.
The differences between the New York Heart Association (NYHA) class at 12 months and the baseline were calculated. In total, 77.5% of patients showed a decrease of NYHA equal to at least one class, whereas 19.7% of patients remained stable over the time. Only 2.8% of patients showed a worsened clinical status.
Reduction in mean gradients and increase in effective orifice area were both observed at 1 year follow-up, according to the FDA report.
At the 1-, 2-, and 3-year follow-up time points, 75.1% or greater of the implanted patients with available data had improved by one to three classes and at the 4-year follow-up time point 72.6% of the patients had improved by one to three classes, according to the report. In addition, at the 1-, 2-, and 3-year follow-up time points, 92.9% or greater of the patients were in NYHA Class I and Class II, and at the 4-year follow-up time point, 86.3% of the patients were in NYHA Class I and Class II.
Although there was a predominance of women in the study population (64.4%), patients of both sexes demonstrated acceptable hemodynamic outcomes and significant improvement in functional status, according to the FDA summary.
Sutureless valves are considered to have a promising future according to a recent international consensus panel recommendationpublished online in the European Journal of Cardio-Thoracic Surgery (2015 Oct 29. doi: 10.1093/ejcts/ezv369). The report assessed various benefits of sutureless and rapid deployment technology, and concluded these devices “may represent a helpful tool in aortic valve replacement for patients requiring a biological valve.” However, further evidence will be needed to reaffirm the benefit of sutureless and rapid deployment valves, they concluded.
LivaNova announced that the U.S. Food and Drug Administration has announced the approval of their Perceval Sutureless Heart Valve. The approval was issued Jan. 8 and is effective immediately, and LivaNova indicated that it will begin commercial distribution of the device in the United States over the coming quarter.
According to an FDA summary document, the Perceval Sutureless Heart Valve is a bioprosthetic valve designed to replace a diseased native or a malfunctioning prosthetic aortic valve via open heart surgery. The self-expanding stent frame consists of a tissue component made from bovine pericardium and a self-expandable nitinol stent, along with a dedicated delivery system that allows physicians to position and anchor the valve suturelessly.
The Perceval heart valve is supplied unmounted and must be loaded onto an accompanying holder by reducing the valve size using a supplied polycarbonate collapser. The holder is used for sternal approaches and includes a rigid shaft with an end section that houses the valve prosthesis during delivery. A separate holder, approximately 3 cm longer, is also available for minimally invasive procedures. After implantation, the physician uses the a post-dilation catheter to expand the valve in situ.
Perceval’s approval was based upon the results of a pivotal European study performed to assess the device, the CAVALIER (Safety and Effectiveness Study of Perceval S Valve for Extended CE Mark) trial. The prospective, multicenter, nonrandomized clinical study was conducted at 26 investigational sites in Austria, Belgium, England, France, Germany, and the Netherlands.
Patients were treated between Feb. 23, 2010, and Sept. 30, 2013, and the database for the PDA-assessed data collected through Nov. 5, 2014, and included 658 patients.
The differences between the New York Heart Association (NYHA) class at 12 months and the baseline were calculated. In total, 77.5% of patients showed a decrease of NYHA equal to at least one class, whereas 19.7% of patients remained stable over the time. Only 2.8% of patients showed a worsened clinical status.
Reduction in mean gradients and increase in effective orifice area were both observed at 1 year follow-up, according to the FDA report.
At the 1-, 2-, and 3-year follow-up time points, 75.1% or greater of the implanted patients with available data had improved by one to three classes and at the 4-year follow-up time point 72.6% of the patients had improved by one to three classes, according to the report. In addition, at the 1-, 2-, and 3-year follow-up time points, 92.9% or greater of the patients were in NYHA Class I and Class II, and at the 4-year follow-up time point, 86.3% of the patients were in NYHA Class I and Class II.
Although there was a predominance of women in the study population (64.4%), patients of both sexes demonstrated acceptable hemodynamic outcomes and significant improvement in functional status, according to the FDA summary.
Sutureless valves are considered to have a promising future according to a recent international consensus panel recommendationpublished online in the European Journal of Cardio-Thoracic Surgery (2015 Oct 29. doi: 10.1093/ejcts/ezv369). The report assessed various benefits of sutureless and rapid deployment technology, and concluded these devices “may represent a helpful tool in aortic valve replacement for patients requiring a biological valve.” However, further evidence will be needed to reaffirm the benefit of sutureless and rapid deployment valves, they concluded.
Patient records requests: New regs clarify physician responsibilities
The age of the information-empowered patient is upon us. Not only do patients bring the results of their Internet research when they come to the office, they also want to take a record of the clinical encounter with them when they leave.
New HIPAA guidance issued in January by the Health & Human Services Department’s Office of Civil Rights (OCR) aims to help clinicians know how to respond and with what information; it also addresses when patients can be charged for the information.
In the past, physicians and other providers had to “wing it” when it came to unclear rules about patient’s data requests, said Dianne J. Bourque, a Boston health law and HIPAA compliance attorney. “Prior to this, there may not have been readily available guidance that would drill down” to address specific concerns.
When it comes to systems security, physicians and other health providers do not have to put their health IT systems at risk in an effort to meet a request for patient records. For example, Mrs. Smith requests that her protected health information (PHI) be copied onto a thumb drive that she has provided.
In most cases, a covered entity must provide data access in the manner requested by the patient. But the updated guidance states that health providers are not expected to tolerate “unacceptable levels of risk to the security of the PHI on its systems” in responding to requests.
Unlike system security, patient security does not trump patient access. If Mr. Black requests that his records by emailed to him, but a connection cannot be made secure, physicians are still required to send the data.
While OCR requires HIPAA-covered entities to implement reasonable safeguards to protect PHI while in transit, patients have a right to receive a copy of records by unencrypted email if they so wish.
To comply with the new rules, be sure to warn patients of the risks, and confirm that they still want their PHI by unencrypted email. If confirmed, you must comply with the request. This clarification relieves doctors of potential breach notification and liability if the data is intercepted in transit.
The guidance also clarifies how to deliver patients’ data. If PHI is maintained electronically, physicians and other HIPAA-covered entities must be able provide it to patients electronically.
“Because you hold it electronically, you can’t say, ‘Forget it, you have to have paper,’” Ms. Bourque said. “You lose that option when you keep [data] electronically. Maybe you have to go buy a scanner and scan [the document] and email it, but you can’t charge [patients] for the scanner.”
The new guidance also allows patients to get results directly from a clinical laboratory; however, labs are not required to interpret test results. Rather, patients are encouraged to reach out to their physician for such insights.
Overall, the access guidelines appear reasonable and hopefully will relieve hassles for patients in obtaining their health information, said Dr. Sam Slishman, an emergency physician for Sierra Vista Hospital in San Luis Obispo, Calif., and co-founder of Pre-R, a service that provides in-home visits. Dr. Slishman does not foresee the guidance having much impact on his practices.
“It’s crazy to me that patients have to struggle to retrieve their records at all,” he said in an interview. “I routinely send my patients home with at least their lab tests and copies of their radiology reports so they have something to bring to their [primary care physicians]. If they want more, I hand it to them.”
Dr. Rocky D. Bilhartz, an interventional cardiologist in private practice in College Station, Tex., said that he has concerns about the guidelines. Specifically, that doctors may charge a fee to cover the cost of copying records, but that they cannot charge for the cost of searching and retrieving data, said Dr. Bilhartz, who is founder of ECGsource, an online cardiovascular medical education resource.
“Record requests can take significant time for staff to filter through and gather,” he said in an interview. “That time should be reimbursable ... If updated provisions prohibit charging for time spent compiling records, it seems those provisions are a bit out of touch with understanding what those of us on the ground floor must do when a request is received.”
But Dr. Bilhartz acknowledged that he would be unlikely to charge patients for “reasonable” data requests.
“I’m in private practice ... and because of that, I have more market-driven accountability to all my patients,” he said. “Why would I nickel and dime people who I would want to be satisfied patients? For reasonable requests, I would just provide records for free.”
Ms. Bourque notes that while the clarifications are primarily positive for health providers, they present a double-edged sword.
“The good side is that, it has all this detail and it’s really helpful and makes things easier when you have a tricky access request and don’t know what to do,” she said. “The flip side is that once it’s out there, they expect you to read it and pay attention. You start running out of excuses for why you didn’t comply with the access right or why you got it wrong.”
On Twitter @legal_med
The age of the information-empowered patient is upon us. Not only do patients bring the results of their Internet research when they come to the office, they also want to take a record of the clinical encounter with them when they leave.
New HIPAA guidance issued in January by the Health & Human Services Department’s Office of Civil Rights (OCR) aims to help clinicians know how to respond and with what information; it also addresses when patients can be charged for the information.
In the past, physicians and other providers had to “wing it” when it came to unclear rules about patient’s data requests, said Dianne J. Bourque, a Boston health law and HIPAA compliance attorney. “Prior to this, there may not have been readily available guidance that would drill down” to address specific concerns.
When it comes to systems security, physicians and other health providers do not have to put their health IT systems at risk in an effort to meet a request for patient records. For example, Mrs. Smith requests that her protected health information (PHI) be copied onto a thumb drive that she has provided.
In most cases, a covered entity must provide data access in the manner requested by the patient. But the updated guidance states that health providers are not expected to tolerate “unacceptable levels of risk to the security of the PHI on its systems” in responding to requests.
Unlike system security, patient security does not trump patient access. If Mr. Black requests that his records by emailed to him, but a connection cannot be made secure, physicians are still required to send the data.
While OCR requires HIPAA-covered entities to implement reasonable safeguards to protect PHI while in transit, patients have a right to receive a copy of records by unencrypted email if they so wish.
To comply with the new rules, be sure to warn patients of the risks, and confirm that they still want their PHI by unencrypted email. If confirmed, you must comply with the request. This clarification relieves doctors of potential breach notification and liability if the data is intercepted in transit.
The guidance also clarifies how to deliver patients’ data. If PHI is maintained electronically, physicians and other HIPAA-covered entities must be able provide it to patients electronically.
“Because you hold it electronically, you can’t say, ‘Forget it, you have to have paper,’” Ms. Bourque said. “You lose that option when you keep [data] electronically. Maybe you have to go buy a scanner and scan [the document] and email it, but you can’t charge [patients] for the scanner.”
The new guidance also allows patients to get results directly from a clinical laboratory; however, labs are not required to interpret test results. Rather, patients are encouraged to reach out to their physician for such insights.
Overall, the access guidelines appear reasonable and hopefully will relieve hassles for patients in obtaining their health information, said Dr. Sam Slishman, an emergency physician for Sierra Vista Hospital in San Luis Obispo, Calif., and co-founder of Pre-R, a service that provides in-home visits. Dr. Slishman does not foresee the guidance having much impact on his practices.
“It’s crazy to me that patients have to struggle to retrieve their records at all,” he said in an interview. “I routinely send my patients home with at least their lab tests and copies of their radiology reports so they have something to bring to their [primary care physicians]. If they want more, I hand it to them.”
Dr. Rocky D. Bilhartz, an interventional cardiologist in private practice in College Station, Tex., said that he has concerns about the guidelines. Specifically, that doctors may charge a fee to cover the cost of copying records, but that they cannot charge for the cost of searching and retrieving data, said Dr. Bilhartz, who is founder of ECGsource, an online cardiovascular medical education resource.
“Record requests can take significant time for staff to filter through and gather,” he said in an interview. “That time should be reimbursable ... If updated provisions prohibit charging for time spent compiling records, it seems those provisions are a bit out of touch with understanding what those of us on the ground floor must do when a request is received.”
But Dr. Bilhartz acknowledged that he would be unlikely to charge patients for “reasonable” data requests.
“I’m in private practice ... and because of that, I have more market-driven accountability to all my patients,” he said. “Why would I nickel and dime people who I would want to be satisfied patients? For reasonable requests, I would just provide records for free.”
Ms. Bourque notes that while the clarifications are primarily positive for health providers, they present a double-edged sword.
“The good side is that, it has all this detail and it’s really helpful and makes things easier when you have a tricky access request and don’t know what to do,” she said. “The flip side is that once it’s out there, they expect you to read it and pay attention. You start running out of excuses for why you didn’t comply with the access right or why you got it wrong.”
On Twitter @legal_med
The age of the information-empowered patient is upon us. Not only do patients bring the results of their Internet research when they come to the office, they also want to take a record of the clinical encounter with them when they leave.
New HIPAA guidance issued in January by the Health & Human Services Department’s Office of Civil Rights (OCR) aims to help clinicians know how to respond and with what information; it also addresses when patients can be charged for the information.
In the past, physicians and other providers had to “wing it” when it came to unclear rules about patient’s data requests, said Dianne J. Bourque, a Boston health law and HIPAA compliance attorney. “Prior to this, there may not have been readily available guidance that would drill down” to address specific concerns.
When it comes to systems security, physicians and other health providers do not have to put their health IT systems at risk in an effort to meet a request for patient records. For example, Mrs. Smith requests that her protected health information (PHI) be copied onto a thumb drive that she has provided.
In most cases, a covered entity must provide data access in the manner requested by the patient. But the updated guidance states that health providers are not expected to tolerate “unacceptable levels of risk to the security of the PHI on its systems” in responding to requests.
Unlike system security, patient security does not trump patient access. If Mr. Black requests that his records by emailed to him, but a connection cannot be made secure, physicians are still required to send the data.
While OCR requires HIPAA-covered entities to implement reasonable safeguards to protect PHI while in transit, patients have a right to receive a copy of records by unencrypted email if they so wish.
To comply with the new rules, be sure to warn patients of the risks, and confirm that they still want their PHI by unencrypted email. If confirmed, you must comply with the request. This clarification relieves doctors of potential breach notification and liability if the data is intercepted in transit.
The guidance also clarifies how to deliver patients’ data. If PHI is maintained electronically, physicians and other HIPAA-covered entities must be able provide it to patients electronically.
“Because you hold it electronically, you can’t say, ‘Forget it, you have to have paper,’” Ms. Bourque said. “You lose that option when you keep [data] electronically. Maybe you have to go buy a scanner and scan [the document] and email it, but you can’t charge [patients] for the scanner.”
The new guidance also allows patients to get results directly from a clinical laboratory; however, labs are not required to interpret test results. Rather, patients are encouraged to reach out to their physician for such insights.
Overall, the access guidelines appear reasonable and hopefully will relieve hassles for patients in obtaining their health information, said Dr. Sam Slishman, an emergency physician for Sierra Vista Hospital in San Luis Obispo, Calif., and co-founder of Pre-R, a service that provides in-home visits. Dr. Slishman does not foresee the guidance having much impact on his practices.
“It’s crazy to me that patients have to struggle to retrieve their records at all,” he said in an interview. “I routinely send my patients home with at least their lab tests and copies of their radiology reports so they have something to bring to their [primary care physicians]. If they want more, I hand it to them.”
Dr. Rocky D. Bilhartz, an interventional cardiologist in private practice in College Station, Tex., said that he has concerns about the guidelines. Specifically, that doctors may charge a fee to cover the cost of copying records, but that they cannot charge for the cost of searching and retrieving data, said Dr. Bilhartz, who is founder of ECGsource, an online cardiovascular medical education resource.
“Record requests can take significant time for staff to filter through and gather,” he said in an interview. “That time should be reimbursable ... If updated provisions prohibit charging for time spent compiling records, it seems those provisions are a bit out of touch with understanding what those of us on the ground floor must do when a request is received.”
But Dr. Bilhartz acknowledged that he would be unlikely to charge patients for “reasonable” data requests.
“I’m in private practice ... and because of that, I have more market-driven accountability to all my patients,” he said. “Why would I nickel and dime people who I would want to be satisfied patients? For reasonable requests, I would just provide records for free.”
Ms. Bourque notes that while the clarifications are primarily positive for health providers, they present a double-edged sword.
“The good side is that, it has all this detail and it’s really helpful and makes things easier when you have a tricky access request and don’t know what to do,” she said. “The flip side is that once it’s out there, they expect you to read it and pay attention. You start running out of excuses for why you didn’t comply with the access right or why you got it wrong.”
On Twitter @legal_med
CMS: End of meaningful use imminent in 2016
Meaningful use is on its way out.
Andy Slavitt, acting administrator of the Centers for Medicare & Medicaid Services, told investors attending the annual J.P. Morgan Healthcare Conference that CMS is pulling back from the health care IT incentive program in the coming months.
“The meaningful use program as it has existed will now be effectively over and replaced with something better,” Mr. Slavitt said.*
“We have to get the hearts and minds of physicians back. I think we’ve lost them,” Mr. Slavitt said. He noted that, when the meaningful use incentive program began, few physicians and practices used electronic health records and concerns were that many would not willingly embrace information technology. Now that “virtually everywhere care is delivered has a computer,” it’s time to make health care technology serve beneficiaries and the physicians who serve them, Mr. Slavitt said.
The cost, however, was too high, Mr. Slavitt said. “As any physician will tell you, physician burden and frustration levels are real. Programs that are designed to improve often distract. Done poorly, measures are divorced from how physicians practice and add to the cynicism that the people who build these programs just don’t get it.”
Soon, CMS will no longer reward health care providers for using technology, but will instead focus on patient outcomes through the merit-based incentive pay systems created by last year’s Medicare Access and CHIP Reauthorization Act (MACRA) legislation. In addition to asking physicians to work with health care IT innovators to create systems that work best according to their practice’s respective needs, CMS is calling on the private sector to create apps and analytic tools that will keep data secure while fostering true and widespread interoperability.
Anyone seeking to block data transfer will find CMS is not their friend. Mr. Slavitt said. “We’re deadly serious about interoperability. Technology companies that look for ways to practice data blocking in opposition to new regulations will find that it will not be tolerated.”
Dr. James L. Madara, CEO of the American Medical Association, echoed Mr. Slavitt’s comments on the current, negative impact of EHRs on physicians’ practices. He noted that many physicians are spending at least 2 hours each workday using their EHR and may click up to 4,000 times per 8-hour shift.
Dr. Madara outlined three AMA goals to help restore the physician-patient relationship. The first is to restructure the medical school curriculum, which he said essentially is the same as it has been for 100 years. New generations of physicians should be taught how to deliver collaborative care that includes telemedicine, more ambulatory care, and home care. Community-based partnerships, he said, would become key to treating chronic diseases like diabetes and would have to be factored into reimbursement models. The AMA also seeks to improve health outcomes and ensure thriving physician practices.
Central to the AMA’s plan for the future: Helping physicians restructure practice via technology. He announced that the AMA is a founding partner in the Silicon Valley (Calif.) based Health2047, a company focused on supporting health IT and other entrepreneurs in their efforts to provide physicians with digital tools that improve patient outcomes, among other innovations.
With MACRA set to go into full effect in 2019, Dr. Madara said that a “daunting” level of change is about to take place. Citing the successful shift to ICD-10, he said he was optimistic there would be positive changes, largely brought about through incentives to the private marketplace and by dropping meaningful use.
Although having metrics in health care delivery is important, Dr. Madara said that, up to this point, “We kinda got it wrong” with quality measures that are more processed based, rather than evidence based. “It was really great to hear about the move from meaningful use to a more aggregated program.”
*Correction, 1/12/2016: A previous version of this story included an incorrect start-up date for meaningful use changes.
On Twitter @whitneymcknight
Meaningful use is on its way out.
Andy Slavitt, acting administrator of the Centers for Medicare & Medicaid Services, told investors attending the annual J.P. Morgan Healthcare Conference that CMS is pulling back from the health care IT incentive program in the coming months.
“The meaningful use program as it has existed will now be effectively over and replaced with something better,” Mr. Slavitt said.*
“We have to get the hearts and minds of physicians back. I think we’ve lost them,” Mr. Slavitt said. He noted that, when the meaningful use incentive program began, few physicians and practices used electronic health records and concerns were that many would not willingly embrace information technology. Now that “virtually everywhere care is delivered has a computer,” it’s time to make health care technology serve beneficiaries and the physicians who serve them, Mr. Slavitt said.
The cost, however, was too high, Mr. Slavitt said. “As any physician will tell you, physician burden and frustration levels are real. Programs that are designed to improve often distract. Done poorly, measures are divorced from how physicians practice and add to the cynicism that the people who build these programs just don’t get it.”
Soon, CMS will no longer reward health care providers for using technology, but will instead focus on patient outcomes through the merit-based incentive pay systems created by last year’s Medicare Access and CHIP Reauthorization Act (MACRA) legislation. In addition to asking physicians to work with health care IT innovators to create systems that work best according to their practice’s respective needs, CMS is calling on the private sector to create apps and analytic tools that will keep data secure while fostering true and widespread interoperability.
Anyone seeking to block data transfer will find CMS is not their friend. Mr. Slavitt said. “We’re deadly serious about interoperability. Technology companies that look for ways to practice data blocking in opposition to new regulations will find that it will not be tolerated.”
Dr. James L. Madara, CEO of the American Medical Association, echoed Mr. Slavitt’s comments on the current, negative impact of EHRs on physicians’ practices. He noted that many physicians are spending at least 2 hours each workday using their EHR and may click up to 4,000 times per 8-hour shift.
Dr. Madara outlined three AMA goals to help restore the physician-patient relationship. The first is to restructure the medical school curriculum, which he said essentially is the same as it has been for 100 years. New generations of physicians should be taught how to deliver collaborative care that includes telemedicine, more ambulatory care, and home care. Community-based partnerships, he said, would become key to treating chronic diseases like diabetes and would have to be factored into reimbursement models. The AMA also seeks to improve health outcomes and ensure thriving physician practices.
Central to the AMA’s plan for the future: Helping physicians restructure practice via technology. He announced that the AMA is a founding partner in the Silicon Valley (Calif.) based Health2047, a company focused on supporting health IT and other entrepreneurs in their efforts to provide physicians with digital tools that improve patient outcomes, among other innovations.
With MACRA set to go into full effect in 2019, Dr. Madara said that a “daunting” level of change is about to take place. Citing the successful shift to ICD-10, he said he was optimistic there would be positive changes, largely brought about through incentives to the private marketplace and by dropping meaningful use.
Although having metrics in health care delivery is important, Dr. Madara said that, up to this point, “We kinda got it wrong” with quality measures that are more processed based, rather than evidence based. “It was really great to hear about the move from meaningful use to a more aggregated program.”
*Correction, 1/12/2016: A previous version of this story included an incorrect start-up date for meaningful use changes.
On Twitter @whitneymcknight
Meaningful use is on its way out.
Andy Slavitt, acting administrator of the Centers for Medicare & Medicaid Services, told investors attending the annual J.P. Morgan Healthcare Conference that CMS is pulling back from the health care IT incentive program in the coming months.
“The meaningful use program as it has existed will now be effectively over and replaced with something better,” Mr. Slavitt said.*
“We have to get the hearts and minds of physicians back. I think we’ve lost them,” Mr. Slavitt said. He noted that, when the meaningful use incentive program began, few physicians and practices used electronic health records and concerns were that many would not willingly embrace information technology. Now that “virtually everywhere care is delivered has a computer,” it’s time to make health care technology serve beneficiaries and the physicians who serve them, Mr. Slavitt said.
The cost, however, was too high, Mr. Slavitt said. “As any physician will tell you, physician burden and frustration levels are real. Programs that are designed to improve often distract. Done poorly, measures are divorced from how physicians practice and add to the cynicism that the people who build these programs just don’t get it.”
Soon, CMS will no longer reward health care providers for using technology, but will instead focus on patient outcomes through the merit-based incentive pay systems created by last year’s Medicare Access and CHIP Reauthorization Act (MACRA) legislation. In addition to asking physicians to work with health care IT innovators to create systems that work best according to their practice’s respective needs, CMS is calling on the private sector to create apps and analytic tools that will keep data secure while fostering true and widespread interoperability.
Anyone seeking to block data transfer will find CMS is not their friend. Mr. Slavitt said. “We’re deadly serious about interoperability. Technology companies that look for ways to practice data blocking in opposition to new regulations will find that it will not be tolerated.”
Dr. James L. Madara, CEO of the American Medical Association, echoed Mr. Slavitt’s comments on the current, negative impact of EHRs on physicians’ practices. He noted that many physicians are spending at least 2 hours each workday using their EHR and may click up to 4,000 times per 8-hour shift.
Dr. Madara outlined three AMA goals to help restore the physician-patient relationship. The first is to restructure the medical school curriculum, which he said essentially is the same as it has been for 100 years. New generations of physicians should be taught how to deliver collaborative care that includes telemedicine, more ambulatory care, and home care. Community-based partnerships, he said, would become key to treating chronic diseases like diabetes and would have to be factored into reimbursement models. The AMA also seeks to improve health outcomes and ensure thriving physician practices.
Central to the AMA’s plan for the future: Helping physicians restructure practice via technology. He announced that the AMA is a founding partner in the Silicon Valley (Calif.) based Health2047, a company focused on supporting health IT and other entrepreneurs in their efforts to provide physicians with digital tools that improve patient outcomes, among other innovations.
With MACRA set to go into full effect in 2019, Dr. Madara said that a “daunting” level of change is about to take place. Citing the successful shift to ICD-10, he said he was optimistic there would be positive changes, largely brought about through incentives to the private marketplace and by dropping meaningful use.
Although having metrics in health care delivery is important, Dr. Madara said that, up to this point, “We kinda got it wrong” with quality measures that are more processed based, rather than evidence based. “It was really great to hear about the move from meaningful use to a more aggregated program.”
*Correction, 1/12/2016: A previous version of this story included an incorrect start-up date for meaningful use changes.
On Twitter @whitneymcknight
FROM THE J.P. MORGAN HEALTHCARE CONFERENCE