User login
Neurology Reviews covers innovative and emerging news in neurology and neuroscience every month, with a focus on practical approaches to treating Parkinson's disease, epilepsy, headache, stroke, multiple sclerosis, Alzheimer's disease, and other neurologic disorders.
PML
Progressive multifocal leukoencephalopathy
Rituxan
The leading independent newspaper covering neurology news and commentary.
Is there a doctor on the plane? Tips for providing in-flight assistance
In most cases, passengers on an airline flight are representative of the general population, which means that anyone could have an emergency at any time.
as determined on the basis of in-flight medical emergencies that resulted in calls to a physician-directed medical communications center, said Amy Faith Ho, MD, MPH of Integrative Emergency Services, Dallas–Fort Worth, in a presentation at the annual meeting of the American College of Emergency Physicians.
The study authors reviewed records of 11,920 in-flight medical emergencies between Jan. 1, 2008, and Oct. 31, 2010. The data showed that physician passengers provided medical assistance in nearly half of in-flight emergencies (48.1%) and that flights were diverted because of the emergency in 7.3% of cases.
The majority of the in-flight emergencies involved syncope or presyncope (37.4% of cases), followed by respiratory symptoms (12.1%) and nausea or vomiting (9.5%), according to the study.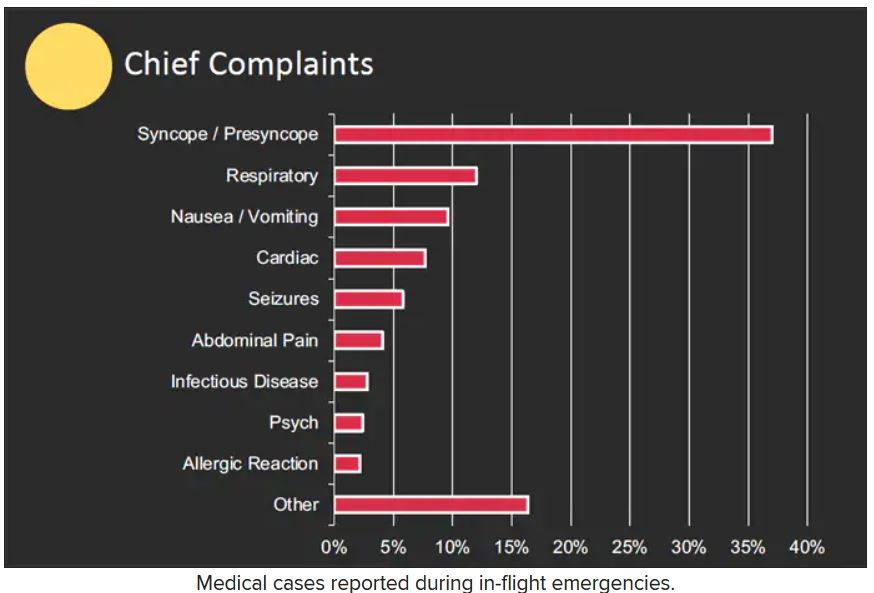
When a physician is faced with an in-flight emergency, the medical team includes the physician himself, medical ground control, and the flight attendants, said Dr. Ho. Requirements may vary among airlines, but all flight attendants will be trained in cardiopulmonary resuscitation (CPR) or basic life support, as well as use of automated external defibrillators (AEDs).
Physician call centers (medical ground control) can provide additional assistance remotely, she said.
The in-flight medical bag
Tools in a physician’s in-flight toolbox start with the first-aid kit. Airplanes also have an emergency medical kit (EMK), an oxygen tank, and an AED.
The minimum EMK contents are mandated by the Federal Aviation Administration, said Dr. Ho. The standard equipment includes a stethoscope, a sphygmomanometer, and three sizes of oropharyngeal airways. Other items include self-inflating manual resuscitation devices and CPR masks in thee sizes, alcohol sponges, gloves, adhesive tape, scissors, a tourniquet, as well as saline solution, needles, syringes, and an intravenous administration set consisting of tubing and two Y connectors.
An EMK also should contain the following medications: nonnarcotic analgesic tablets, antihistamine tablets, an injectable antihistamine, atropine, aspirin tablets, a bronchodilator, and epinephrine (both 1:1000; 1 injectable cc and 1:10,000; two injectable cc). Nitroglycerin tablets and 5 cc of 20 mg/mL injectable cardiac lidocaine are part of the mandated kit as well, according to Dr. Ho.
Some airlines carry additional supplies on all their flights, said Dr. Ho. Notably, American Airlines and British Airways carry EpiPens for adults and children, as well as opioid reversal medication (naloxone) and glucose for managing low blood sugar. American Airlines and Delta stock antiemetics, and Delta also carries naloxone. British Airways is unique in stocking additional cardiac medications, both oral and injectable.
How to handle an in-flight emergency
Physicians should always carry a copy of their medical license when traveling for documentation by the airline if they assist in a medical emergency during a flight, Dr. Ho emphasized. “Staff” personnel should be used. These include the flight attendants, medical ground control, and other passengers who might have useful skills, such as nursing, the ability to perform CPR, or therapy/counseling to calm a frightened patient. If needed, “crowdsource additional supplies from passengers,” such as a glucometer or pulse oximeter.
Legal lessons
Physicians are not obligated to assist during an in-flight medical emergency, said Dr. Ho. Legal jurisdiction can vary. In the United States, a bystander who assists in an emergency is generally protected by Good Samaritan laws; for international airlines, the laws may vary; those where the airline is based usually apply.
The Aviation Medical Assistance Act, passed in 1998, protects individuals from being sued for negligence while providing medical assistance, “unless the individual, while rendering such assistance, is guilty of gross negligence of willful misconduct,” Dr. Ho noted. The Aviation Medical Assistance Act also protects the airline itself “if the carrier in good faith believes that the passenger is a medically qualified individual.”
Dr. Ho disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In most cases, passengers on an airline flight are representative of the general population, which means that anyone could have an emergency at any time.
as determined on the basis of in-flight medical emergencies that resulted in calls to a physician-directed medical communications center, said Amy Faith Ho, MD, MPH of Integrative Emergency Services, Dallas–Fort Worth, in a presentation at the annual meeting of the American College of Emergency Physicians.
The study authors reviewed records of 11,920 in-flight medical emergencies between Jan. 1, 2008, and Oct. 31, 2010. The data showed that physician passengers provided medical assistance in nearly half of in-flight emergencies (48.1%) and that flights were diverted because of the emergency in 7.3% of cases.
The majority of the in-flight emergencies involved syncope or presyncope (37.4% of cases), followed by respiratory symptoms (12.1%) and nausea or vomiting (9.5%), according to the study.
When a physician is faced with an in-flight emergency, the medical team includes the physician himself, medical ground control, and the flight attendants, said Dr. Ho. Requirements may vary among airlines, but all flight attendants will be trained in cardiopulmonary resuscitation (CPR) or basic life support, as well as use of automated external defibrillators (AEDs).
Physician call centers (medical ground control) can provide additional assistance remotely, she said.
The in-flight medical bag
Tools in a physician’s in-flight toolbox start with the first-aid kit. Airplanes also have an emergency medical kit (EMK), an oxygen tank, and an AED.
The minimum EMK contents are mandated by the Federal Aviation Administration, said Dr. Ho. The standard equipment includes a stethoscope, a sphygmomanometer, and three sizes of oropharyngeal airways. Other items include self-inflating manual resuscitation devices and CPR masks in thee sizes, alcohol sponges, gloves, adhesive tape, scissors, a tourniquet, as well as saline solution, needles, syringes, and an intravenous administration set consisting of tubing and two Y connectors.
An EMK also should contain the following medications: nonnarcotic analgesic tablets, antihistamine tablets, an injectable antihistamine, atropine, aspirin tablets, a bronchodilator, and epinephrine (both 1:1000; 1 injectable cc and 1:10,000; two injectable cc). Nitroglycerin tablets and 5 cc of 20 mg/mL injectable cardiac lidocaine are part of the mandated kit as well, according to Dr. Ho.
Some airlines carry additional supplies on all their flights, said Dr. Ho. Notably, American Airlines and British Airways carry EpiPens for adults and children, as well as opioid reversal medication (naloxone) and glucose for managing low blood sugar. American Airlines and Delta stock antiemetics, and Delta also carries naloxone. British Airways is unique in stocking additional cardiac medications, both oral and injectable.
How to handle an in-flight emergency
Physicians should always carry a copy of their medical license when traveling for documentation by the airline if they assist in a medical emergency during a flight, Dr. Ho emphasized. “Staff” personnel should be used. These include the flight attendants, medical ground control, and other passengers who might have useful skills, such as nursing, the ability to perform CPR, or therapy/counseling to calm a frightened patient. If needed, “crowdsource additional supplies from passengers,” such as a glucometer or pulse oximeter.
Legal lessons
Physicians are not obligated to assist during an in-flight medical emergency, said Dr. Ho. Legal jurisdiction can vary. In the United States, a bystander who assists in an emergency is generally protected by Good Samaritan laws; for international airlines, the laws may vary; those where the airline is based usually apply.
The Aviation Medical Assistance Act, passed in 1998, protects individuals from being sued for negligence while providing medical assistance, “unless the individual, while rendering such assistance, is guilty of gross negligence of willful misconduct,” Dr. Ho noted. The Aviation Medical Assistance Act also protects the airline itself “if the carrier in good faith believes that the passenger is a medically qualified individual.”
Dr. Ho disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In most cases, passengers on an airline flight are representative of the general population, which means that anyone could have an emergency at any time.
as determined on the basis of in-flight medical emergencies that resulted in calls to a physician-directed medical communications center, said Amy Faith Ho, MD, MPH of Integrative Emergency Services, Dallas–Fort Worth, in a presentation at the annual meeting of the American College of Emergency Physicians.
The study authors reviewed records of 11,920 in-flight medical emergencies between Jan. 1, 2008, and Oct. 31, 2010. The data showed that physician passengers provided medical assistance in nearly half of in-flight emergencies (48.1%) and that flights were diverted because of the emergency in 7.3% of cases.
The majority of the in-flight emergencies involved syncope or presyncope (37.4% of cases), followed by respiratory symptoms (12.1%) and nausea or vomiting (9.5%), according to the study.
When a physician is faced with an in-flight emergency, the medical team includes the physician himself, medical ground control, and the flight attendants, said Dr. Ho. Requirements may vary among airlines, but all flight attendants will be trained in cardiopulmonary resuscitation (CPR) or basic life support, as well as use of automated external defibrillators (AEDs).
Physician call centers (medical ground control) can provide additional assistance remotely, she said.
The in-flight medical bag
Tools in a physician’s in-flight toolbox start with the first-aid kit. Airplanes also have an emergency medical kit (EMK), an oxygen tank, and an AED.
The minimum EMK contents are mandated by the Federal Aviation Administration, said Dr. Ho. The standard equipment includes a stethoscope, a sphygmomanometer, and three sizes of oropharyngeal airways. Other items include self-inflating manual resuscitation devices and CPR masks in thee sizes, alcohol sponges, gloves, adhesive tape, scissors, a tourniquet, as well as saline solution, needles, syringes, and an intravenous administration set consisting of tubing and two Y connectors.
An EMK also should contain the following medications: nonnarcotic analgesic tablets, antihistamine tablets, an injectable antihistamine, atropine, aspirin tablets, a bronchodilator, and epinephrine (both 1:1000; 1 injectable cc and 1:10,000; two injectable cc). Nitroglycerin tablets and 5 cc of 20 mg/mL injectable cardiac lidocaine are part of the mandated kit as well, according to Dr. Ho.
Some airlines carry additional supplies on all their flights, said Dr. Ho. Notably, American Airlines and British Airways carry EpiPens for adults and children, as well as opioid reversal medication (naloxone) and glucose for managing low blood sugar. American Airlines and Delta stock antiemetics, and Delta also carries naloxone. British Airways is unique in stocking additional cardiac medications, both oral and injectable.
How to handle an in-flight emergency
Physicians should always carry a copy of their medical license when traveling for documentation by the airline if they assist in a medical emergency during a flight, Dr. Ho emphasized. “Staff” personnel should be used. These include the flight attendants, medical ground control, and other passengers who might have useful skills, such as nursing, the ability to perform CPR, or therapy/counseling to calm a frightened patient. If needed, “crowdsource additional supplies from passengers,” such as a glucometer or pulse oximeter.
Legal lessons
Physicians are not obligated to assist during an in-flight medical emergency, said Dr. Ho. Legal jurisdiction can vary. In the United States, a bystander who assists in an emergency is generally protected by Good Samaritan laws; for international airlines, the laws may vary; those where the airline is based usually apply.
The Aviation Medical Assistance Act, passed in 1998, protects individuals from being sued for negligence while providing medical assistance, “unless the individual, while rendering such assistance, is guilty of gross negligence of willful misconduct,” Dr. Ho noted. The Aviation Medical Assistance Act also protects the airline itself “if the carrier in good faith believes that the passenger is a medically qualified individual.”
Dr. Ho disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACEP 2022
OSA raises risk of atrial fibrillation and stroke
compared with controls, based on data from 303 individuals.
OSA has become a common chronic disease, and cardiovascular diseases including AFib also are known independent risk factors associated with OSA, Anna Hojager, MD, of Zealand University Hospital, Roskilde, Denmark, and colleagues wrote. Previous studies have shown a significant increase in AFib risk in OSA patients with severe disease, but the prevalence of undiagnosed AFib in OSA patients has not been explored.
In a study published in Sleep Medicine, the researchers enrolled 238 adults with severe OSA (based on apnea-hypopnea index of 15 or higher) and 65 with mild or no OSA (based on an AHI of less than 15). The mean AHI across all participants was 34.2, and ranged from 0.2 to 115.8.
Participants underwent heart rhythm monitoring using a home system or standard ECG for 7 days; they were instructed to carry the device at all times except when showering or sweating heavily. The primary outcome was the detection of AFib, defined as at least one period of 30 seconds or longer with an irregular heart rhythm but without detectable evidence of another diagnosis. Sleep was assessed for one night using a portable sleep monitoring device. All participants were examined at baseline and measured for blood pressure, body mass index, waist-to-hip ratio, and ECG.
Overall, AFib occurred in 21 patients with moderate to severe OSA and 1 patient with mild/no OSA (8.8% vs. 1.5%, P = .045). The majority of patients across both groups had hypertension (66%) and dyslipidemia (77.6%), but the severe OSA group was more likely to be dysregulated and to have unknown prediabetes. Participants who were deemed candidates for anticoagulation therapy were referred for additional treatment. None of the 22 total patients with AFib had heart failure with reduced ejection fraction, and 68.2% had normal ejection fraction and ventricle function.
The researchers noted that no guidelines currently exist for systematic opportunistic screening for comorbidities in OSA patients, although the American Academy of Sleep Medicine recommends patient education as part of a multidisciplinary chronic disease management strategy. The high prevalence of AFib in OSA patients, as seen in the current study, “might warrant a recommendation of screening for paroxysmal [AFib] and could be valuable in the management of modifiable cardiovascular risk factors in patients with OSA,” they wrote.
The study findings were limited by several factors including the observational design and absence of polysomnography to assess OSA, the researchers noted. However, the study has the highest known prevalence of silent AFib in patients with moderate to severe OSA, and supports the value of screening and management for known comorbidities of OSA.
The study received no outside funding. The researchers had no financial conflicts to disclose.
compared with controls, based on data from 303 individuals.
OSA has become a common chronic disease, and cardiovascular diseases including AFib also are known independent risk factors associated with OSA, Anna Hojager, MD, of Zealand University Hospital, Roskilde, Denmark, and colleagues wrote. Previous studies have shown a significant increase in AFib risk in OSA patients with severe disease, but the prevalence of undiagnosed AFib in OSA patients has not been explored.
In a study published in Sleep Medicine, the researchers enrolled 238 adults with severe OSA (based on apnea-hypopnea index of 15 or higher) and 65 with mild or no OSA (based on an AHI of less than 15). The mean AHI across all participants was 34.2, and ranged from 0.2 to 115.8.
Participants underwent heart rhythm monitoring using a home system or standard ECG for 7 days; they were instructed to carry the device at all times except when showering or sweating heavily. The primary outcome was the detection of AFib, defined as at least one period of 30 seconds or longer with an irregular heart rhythm but without detectable evidence of another diagnosis. Sleep was assessed for one night using a portable sleep monitoring device. All participants were examined at baseline and measured for blood pressure, body mass index, waist-to-hip ratio, and ECG.
Overall, AFib occurred in 21 patients with moderate to severe OSA and 1 patient with mild/no OSA (8.8% vs. 1.5%, P = .045). The majority of patients across both groups had hypertension (66%) and dyslipidemia (77.6%), but the severe OSA group was more likely to be dysregulated and to have unknown prediabetes. Participants who were deemed candidates for anticoagulation therapy were referred for additional treatment. None of the 22 total patients with AFib had heart failure with reduced ejection fraction, and 68.2% had normal ejection fraction and ventricle function.
The researchers noted that no guidelines currently exist for systematic opportunistic screening for comorbidities in OSA patients, although the American Academy of Sleep Medicine recommends patient education as part of a multidisciplinary chronic disease management strategy. The high prevalence of AFib in OSA patients, as seen in the current study, “might warrant a recommendation of screening for paroxysmal [AFib] and could be valuable in the management of modifiable cardiovascular risk factors in patients with OSA,” they wrote.
The study findings were limited by several factors including the observational design and absence of polysomnography to assess OSA, the researchers noted. However, the study has the highest known prevalence of silent AFib in patients with moderate to severe OSA, and supports the value of screening and management for known comorbidities of OSA.
The study received no outside funding. The researchers had no financial conflicts to disclose.
compared with controls, based on data from 303 individuals.
OSA has become a common chronic disease, and cardiovascular diseases including AFib also are known independent risk factors associated with OSA, Anna Hojager, MD, of Zealand University Hospital, Roskilde, Denmark, and colleagues wrote. Previous studies have shown a significant increase in AFib risk in OSA patients with severe disease, but the prevalence of undiagnosed AFib in OSA patients has not been explored.
In a study published in Sleep Medicine, the researchers enrolled 238 adults with severe OSA (based on apnea-hypopnea index of 15 or higher) and 65 with mild or no OSA (based on an AHI of less than 15). The mean AHI across all participants was 34.2, and ranged from 0.2 to 115.8.
Participants underwent heart rhythm monitoring using a home system or standard ECG for 7 days; they were instructed to carry the device at all times except when showering or sweating heavily. The primary outcome was the detection of AFib, defined as at least one period of 30 seconds or longer with an irregular heart rhythm but without detectable evidence of another diagnosis. Sleep was assessed for one night using a portable sleep monitoring device. All participants were examined at baseline and measured for blood pressure, body mass index, waist-to-hip ratio, and ECG.
Overall, AFib occurred in 21 patients with moderate to severe OSA and 1 patient with mild/no OSA (8.8% vs. 1.5%, P = .045). The majority of patients across both groups had hypertension (66%) and dyslipidemia (77.6%), but the severe OSA group was more likely to be dysregulated and to have unknown prediabetes. Participants who were deemed candidates for anticoagulation therapy were referred for additional treatment. None of the 22 total patients with AFib had heart failure with reduced ejection fraction, and 68.2% had normal ejection fraction and ventricle function.
The researchers noted that no guidelines currently exist for systematic opportunistic screening for comorbidities in OSA patients, although the American Academy of Sleep Medicine recommends patient education as part of a multidisciplinary chronic disease management strategy. The high prevalence of AFib in OSA patients, as seen in the current study, “might warrant a recommendation of screening for paroxysmal [AFib] and could be valuable in the management of modifiable cardiovascular risk factors in patients with OSA,” they wrote.
The study findings were limited by several factors including the observational design and absence of polysomnography to assess OSA, the researchers noted. However, the study has the highest known prevalence of silent AFib in patients with moderate to severe OSA, and supports the value of screening and management for known comorbidities of OSA.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM SLEEP MEDICINE
Nutrition for cognition: A missed opportunity in U.S. seniors?
, new research shows. Researchers assessed the memory function of more than 3,500 persons who used SNAP or did not use SNAP over a period of 20 years. They found that those who didn’t use the food benefits program experienced 2 more years of cognitive aging compared with program users.
Of the 3,555 individuals included in the study, all were eligible to use the benefits, but only 559 did, leaving 2,996 participants who did not take advantage of the program.
Low program participation levels translate into a missed opportunity to prevent dementia, said study investigator Adina Zeki Al Hazzouri, PhD, assistant professor of epidemiology at the Columbia Aging Center at Columbia University Mailman School of Public Health in New York.
She said that prior research has shown that stigma may prevent older Americans from using SNAP. “Educational programs are needed to reduce the stigma that the public holds towards SNAP use,” she said.
Policy change could increase usage among older individuals, Dr. Zeki Al Hazzouri noted. Such changes could include simplifying enrollment and reporting procedures, shortening recertification periods, and increasing benefit levels.
The study was published online in Neurology.
Memory preservation
Dr. Zeki Al Hazzouri and her team assessed respondents from the Health and Retirement Study (HRS), a representative sample of Americans aged 50 and older. All respondents who were eligible to participate in SNAP in 1996 were followed every 2 years until 2016.
At each assessment, HRS respondents completed memory tests, including immediate and delayed word recall. For those who were too impaired to complete the interview, proxy informants – typically, their spouses or family members – assessed the memory and cognition of their family members using validated instruments, such as the 16-item Informant Questionnaire for Cognitive Decline.
Investigators used a validated memory function composite score, which is benchmarked against the memory assessments and evaluations of the Aging, Demographics, and Memory Study (ADAMS) cohort.
The team found that compared with nonusers, SNAP users were more likely to be women, Black, and born in the southern United States. They were less likely to be married and had more chronic conditions, such as high blood pressure, diabetes, cancer, heart problems, psychiatric problems, and arthritis.
One important study limitation was that SNAP use was measured only once during the study, the investigators noted. Ideally, Dr. Zeki Al Hazzouri said, future research would examine cumulative SNAP use history and explore the pathways that might account for the association between SNAP use and memory decline.
While findings suggest that there were no significant differences in baseline memory function between SNAP users and nonusers, users experienced approximately 2 fewer years of cognitive aging over a 10-year period than those who didn’t use the program.
Dr. Zeki Al Hazzouri speculated that SNAP benefits may slow cognitive aging by contributing to overall brain health and that, in comparison with nonusers, SNAP users absorb more nutrients, which promote neuronal integrity.
The investigators theorized that SNAP benefits may reduce stress from financial hardship, which has been linked to premature cognitive aging in other research.
“SNAP may also increase the purchasing power and investment in other health preserving behaviors, but also resulting in better access to care, which may in turn result in better disease management and management of risk factors for cognitive function,” the investigators wrote.
An underutilized program
In an accompanying editorial, Steven Albert, PhD, Philip B. Hallen Endowed Chair in Community Health and Social Justice at the University of Pittsburgh, noted that in 2020, among households with people aged 50 and older in the United States, more than 9 million Americans experienced food insecurity.
Furthermore, he pointed out, research from 2018 showed that 71% of people aged 60 and older who met income eligibility for SNAP did not participate in the program. “SNAP is an underutilized food security program involving substantial income supplements for older people with low incomes.
“Against the backdrop of so many failures of pharmacotherapy for dementia and the so far inexorable increase in the prevalence of dementia due to population aging, are we missing an opportunity to support cognitive health by failing to enroll the 14 million Americans who are over age 60 and eligible for SNAP but who do not participate?” Dr. Albert asked. He suggested that it would be helpful to determine this through a randomized promotion trial.
The study was funded by the National Institute on Aging. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Researchers assessed the memory function of more than 3,500 persons who used SNAP or did not use SNAP over a period of 20 years. They found that those who didn’t use the food benefits program experienced 2 more years of cognitive aging compared with program users.
Of the 3,555 individuals included in the study, all were eligible to use the benefits, but only 559 did, leaving 2,996 participants who did not take advantage of the program.
Low program participation levels translate into a missed opportunity to prevent dementia, said study investigator Adina Zeki Al Hazzouri, PhD, assistant professor of epidemiology at the Columbia Aging Center at Columbia University Mailman School of Public Health in New York.
She said that prior research has shown that stigma may prevent older Americans from using SNAP. “Educational programs are needed to reduce the stigma that the public holds towards SNAP use,” she said.
Policy change could increase usage among older individuals, Dr. Zeki Al Hazzouri noted. Such changes could include simplifying enrollment and reporting procedures, shortening recertification periods, and increasing benefit levels.
The study was published online in Neurology.
Memory preservation
Dr. Zeki Al Hazzouri and her team assessed respondents from the Health and Retirement Study (HRS), a representative sample of Americans aged 50 and older. All respondents who were eligible to participate in SNAP in 1996 were followed every 2 years until 2016.
At each assessment, HRS respondents completed memory tests, including immediate and delayed word recall. For those who were too impaired to complete the interview, proxy informants – typically, their spouses or family members – assessed the memory and cognition of their family members using validated instruments, such as the 16-item Informant Questionnaire for Cognitive Decline.
Investigators used a validated memory function composite score, which is benchmarked against the memory assessments and evaluations of the Aging, Demographics, and Memory Study (ADAMS) cohort.
The team found that compared with nonusers, SNAP users were more likely to be women, Black, and born in the southern United States. They were less likely to be married and had more chronic conditions, such as high blood pressure, diabetes, cancer, heart problems, psychiatric problems, and arthritis.
One important study limitation was that SNAP use was measured only once during the study, the investigators noted. Ideally, Dr. Zeki Al Hazzouri said, future research would examine cumulative SNAP use history and explore the pathways that might account for the association between SNAP use and memory decline.
While findings suggest that there were no significant differences in baseline memory function between SNAP users and nonusers, users experienced approximately 2 fewer years of cognitive aging over a 10-year period than those who didn’t use the program.
Dr. Zeki Al Hazzouri speculated that SNAP benefits may slow cognitive aging by contributing to overall brain health and that, in comparison with nonusers, SNAP users absorb more nutrients, which promote neuronal integrity.
The investigators theorized that SNAP benefits may reduce stress from financial hardship, which has been linked to premature cognitive aging in other research.
“SNAP may also increase the purchasing power and investment in other health preserving behaviors, but also resulting in better access to care, which may in turn result in better disease management and management of risk factors for cognitive function,” the investigators wrote.
An underutilized program
In an accompanying editorial, Steven Albert, PhD, Philip B. Hallen Endowed Chair in Community Health and Social Justice at the University of Pittsburgh, noted that in 2020, among households with people aged 50 and older in the United States, more than 9 million Americans experienced food insecurity.
Furthermore, he pointed out, research from 2018 showed that 71% of people aged 60 and older who met income eligibility for SNAP did not participate in the program. “SNAP is an underutilized food security program involving substantial income supplements for older people with low incomes.
“Against the backdrop of so many failures of pharmacotherapy for dementia and the so far inexorable increase in the prevalence of dementia due to population aging, are we missing an opportunity to support cognitive health by failing to enroll the 14 million Americans who are over age 60 and eligible for SNAP but who do not participate?” Dr. Albert asked. He suggested that it would be helpful to determine this through a randomized promotion trial.
The study was funded by the National Institute on Aging. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Researchers assessed the memory function of more than 3,500 persons who used SNAP or did not use SNAP over a period of 20 years. They found that those who didn’t use the food benefits program experienced 2 more years of cognitive aging compared with program users.
Of the 3,555 individuals included in the study, all were eligible to use the benefits, but only 559 did, leaving 2,996 participants who did not take advantage of the program.
Low program participation levels translate into a missed opportunity to prevent dementia, said study investigator Adina Zeki Al Hazzouri, PhD, assistant professor of epidemiology at the Columbia Aging Center at Columbia University Mailman School of Public Health in New York.
She said that prior research has shown that stigma may prevent older Americans from using SNAP. “Educational programs are needed to reduce the stigma that the public holds towards SNAP use,” she said.
Policy change could increase usage among older individuals, Dr. Zeki Al Hazzouri noted. Such changes could include simplifying enrollment and reporting procedures, shortening recertification periods, and increasing benefit levels.
The study was published online in Neurology.
Memory preservation
Dr. Zeki Al Hazzouri and her team assessed respondents from the Health and Retirement Study (HRS), a representative sample of Americans aged 50 and older. All respondents who were eligible to participate in SNAP in 1996 were followed every 2 years until 2016.
At each assessment, HRS respondents completed memory tests, including immediate and delayed word recall. For those who were too impaired to complete the interview, proxy informants – typically, their spouses or family members – assessed the memory and cognition of their family members using validated instruments, such as the 16-item Informant Questionnaire for Cognitive Decline.
Investigators used a validated memory function composite score, which is benchmarked against the memory assessments and evaluations of the Aging, Demographics, and Memory Study (ADAMS) cohort.
The team found that compared with nonusers, SNAP users were more likely to be women, Black, and born in the southern United States. They were less likely to be married and had more chronic conditions, such as high blood pressure, diabetes, cancer, heart problems, psychiatric problems, and arthritis.
One important study limitation was that SNAP use was measured only once during the study, the investigators noted. Ideally, Dr. Zeki Al Hazzouri said, future research would examine cumulative SNAP use history and explore the pathways that might account for the association between SNAP use and memory decline.
While findings suggest that there were no significant differences in baseline memory function between SNAP users and nonusers, users experienced approximately 2 fewer years of cognitive aging over a 10-year period than those who didn’t use the program.
Dr. Zeki Al Hazzouri speculated that SNAP benefits may slow cognitive aging by contributing to overall brain health and that, in comparison with nonusers, SNAP users absorb more nutrients, which promote neuronal integrity.
The investigators theorized that SNAP benefits may reduce stress from financial hardship, which has been linked to premature cognitive aging in other research.
“SNAP may also increase the purchasing power and investment in other health preserving behaviors, but also resulting in better access to care, which may in turn result in better disease management and management of risk factors for cognitive function,” the investigators wrote.
An underutilized program
In an accompanying editorial, Steven Albert, PhD, Philip B. Hallen Endowed Chair in Community Health and Social Justice at the University of Pittsburgh, noted that in 2020, among households with people aged 50 and older in the United States, more than 9 million Americans experienced food insecurity.
Furthermore, he pointed out, research from 2018 showed that 71% of people aged 60 and older who met income eligibility for SNAP did not participate in the program. “SNAP is an underutilized food security program involving substantial income supplements for older people with low incomes.
“Against the backdrop of so many failures of pharmacotherapy for dementia and the so far inexorable increase in the prevalence of dementia due to population aging, are we missing an opportunity to support cognitive health by failing to enroll the 14 million Americans who are over age 60 and eligible for SNAP but who do not participate?” Dr. Albert asked. He suggested that it would be helpful to determine this through a randomized promotion trial.
The study was funded by the National Institute on Aging. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From Neurology
Repeat COVID infection doubles mortality risk
Getting COVID-19 a second time doubles a person’s chance of dying and triples the likelihood of being hospitalized in the next 6 months, a new study found.
Vaccination and booster status did not improve survival or hospitalization rates among people who were infected more than once.
“Reinfection with COVID-19 increases the risk of both acute outcomes and long COVID,” study author Ziyad Al-Aly, MD, told Reuters. “This was evident in unvaccinated, vaccinated and boosted people.”
The study was published in the journal Nature Medicine.
Researchers analyzed U.S. Department of Veterans Affairs data, including 443,588 people with a first infection of SARS-CoV-2, 40,947 people who were infected two or more times, and 5.3 million people who had not been infected with coronavirus, whose data served as the control group.
“During the past few months, there’s been an air of invincibility among people who have had COVID-19 or their vaccinations and boosters, and especially among people who have had an infection and also received vaccines; some people started to [refer] to these individuals as having a sort of superimmunity to the virus,” Dr. Al-Aly said in a press release from Washington University in St. Louis. “Without ambiguity, our research showed that getting an infection a second, third or fourth time contributes to additional health risks in the acute phase, meaning the first 30 days after infection, and in the months beyond, meaning the long COVID phase.”
Being infected with COVID-19 more than once also dramatically increased the risk of developing lung problems, heart conditions, or brain conditions. The heightened risks persisted for 6 months.
Researchers said a limitation of their study was that data primarily came from White males.
An expert not involved in the study told Reuters that the Veterans Affairs population does not reflect the general population. Patients at VA health facilities are generally older with more than normal health complications, said John Moore, PhD, a professor of microbiology and immunology at Weill Cornell Medicine, New York.
Dr. Al-Aly encouraged people to be vigilant as they plan for the holiday season, Reuters reported.
“We had started seeing a lot of patients coming to the clinic with an air of invincibility,” he told Reuters. “They wondered, ‘Does getting a reinfection really matter?’ The answer is yes, it absolutely does.”
A version of this article first appeared on WebMD.com.
Getting COVID-19 a second time doubles a person’s chance of dying and triples the likelihood of being hospitalized in the next 6 months, a new study found.
Vaccination and booster status did not improve survival or hospitalization rates among people who were infected more than once.
“Reinfection with COVID-19 increases the risk of both acute outcomes and long COVID,” study author Ziyad Al-Aly, MD, told Reuters. “This was evident in unvaccinated, vaccinated and boosted people.”
The study was published in the journal Nature Medicine.
Researchers analyzed U.S. Department of Veterans Affairs data, including 443,588 people with a first infection of SARS-CoV-2, 40,947 people who were infected two or more times, and 5.3 million people who had not been infected with coronavirus, whose data served as the control group.
“During the past few months, there’s been an air of invincibility among people who have had COVID-19 or their vaccinations and boosters, and especially among people who have had an infection and also received vaccines; some people started to [refer] to these individuals as having a sort of superimmunity to the virus,” Dr. Al-Aly said in a press release from Washington University in St. Louis. “Without ambiguity, our research showed that getting an infection a second, third or fourth time contributes to additional health risks in the acute phase, meaning the first 30 days after infection, and in the months beyond, meaning the long COVID phase.”
Being infected with COVID-19 more than once also dramatically increased the risk of developing lung problems, heart conditions, or brain conditions. The heightened risks persisted for 6 months.
Researchers said a limitation of their study was that data primarily came from White males.
An expert not involved in the study told Reuters that the Veterans Affairs population does not reflect the general population. Patients at VA health facilities are generally older with more than normal health complications, said John Moore, PhD, a professor of microbiology and immunology at Weill Cornell Medicine, New York.
Dr. Al-Aly encouraged people to be vigilant as they plan for the holiday season, Reuters reported.
“We had started seeing a lot of patients coming to the clinic with an air of invincibility,” he told Reuters. “They wondered, ‘Does getting a reinfection really matter?’ The answer is yes, it absolutely does.”
A version of this article first appeared on WebMD.com.
Getting COVID-19 a second time doubles a person’s chance of dying and triples the likelihood of being hospitalized in the next 6 months, a new study found.
Vaccination and booster status did not improve survival or hospitalization rates among people who were infected more than once.
“Reinfection with COVID-19 increases the risk of both acute outcomes and long COVID,” study author Ziyad Al-Aly, MD, told Reuters. “This was evident in unvaccinated, vaccinated and boosted people.”
The study was published in the journal Nature Medicine.
Researchers analyzed U.S. Department of Veterans Affairs data, including 443,588 people with a first infection of SARS-CoV-2, 40,947 people who were infected two or more times, and 5.3 million people who had not been infected with coronavirus, whose data served as the control group.
“During the past few months, there’s been an air of invincibility among people who have had COVID-19 or their vaccinations and boosters, and especially among people who have had an infection and also received vaccines; some people started to [refer] to these individuals as having a sort of superimmunity to the virus,” Dr. Al-Aly said in a press release from Washington University in St. Louis. “Without ambiguity, our research showed that getting an infection a second, third or fourth time contributes to additional health risks in the acute phase, meaning the first 30 days after infection, and in the months beyond, meaning the long COVID phase.”
Being infected with COVID-19 more than once also dramatically increased the risk of developing lung problems, heart conditions, or brain conditions. The heightened risks persisted for 6 months.
Researchers said a limitation of their study was that data primarily came from White males.
An expert not involved in the study told Reuters that the Veterans Affairs population does not reflect the general population. Patients at VA health facilities are generally older with more than normal health complications, said John Moore, PhD, a professor of microbiology and immunology at Weill Cornell Medicine, New York.
Dr. Al-Aly encouraged people to be vigilant as they plan for the holiday season, Reuters reported.
“We had started seeing a lot of patients coming to the clinic with an air of invincibility,” he told Reuters. “They wondered, ‘Does getting a reinfection really matter?’ The answer is yes, it absolutely does.”
A version of this article first appeared on WebMD.com.
FROM NATURE MEDICINE
The Charlie Brown tree
I put a Christmas tree up early in November.
It’s not like it’s a real tree, or even a fancy one. For that matter, I’m Jewish.
Growing up in the 1970s one thing that could be relied on every year was the Charlie Brown Christmas special. It never changed. By age 5 you knew most of the lines, and loved the highlight when Charlie Brown brings home the saddest-looking tree ever, which collapses when he puts a single bauble on it.
Years ago, my kids gave me a Charlie Brown tree as a gift. It even plays the late Vince Guaraldi’s immortal Peanuts theme when you push a button. I forgot about it for a few years, then discovered it, and immediately brought it to my office.
I’m not a fan of holiday creep, where they move up earlier in the year, so I used to put it up after Thanksgiving. But we close the office 2-3 weeks later for the rest of the year. I like the tree, my staff likes the tree, and my patients like the tree, so I just started putting it up in early November so we can enjoy it for a month.
It’s whimsical and brings back memories of innocence, childhood, and (of course) Peanuts. It sets a cheerful tone when you see it there. Very few of my patients can resist pressing the button and playing the music as they go by.
The start of a new year is a relatively arbitrary date, chosen long ago. But its approach is always a reminder that life goes on. We continue our trips around the sun. Good times and bad times come and go, but time never stops.
In bad years the tree reminds me that it’s coming to an end, and to look toward the next. In good years it reminds me that it’s time to be ready for the surprises of the coming one.
In mid-December, after the patients are done for the last day of the year, I quietly put it away. It’s a vaguely somber moment, but at the same time I’m glad to know I now have 2-3 weeks of home time. It mostly involves working at my desk and returning phone calls, but there’s also time to relax with my kids, do jigsaw puzzles, and enjoy the Phoenix winter weather as a break before the next round starts.
To those who disagree with my choice of decoration or its timing, I simply respond: “Good grief!”
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I put a Christmas tree up early in November.
It’s not like it’s a real tree, or even a fancy one. For that matter, I’m Jewish.
Growing up in the 1970s one thing that could be relied on every year was the Charlie Brown Christmas special. It never changed. By age 5 you knew most of the lines, and loved the highlight when Charlie Brown brings home the saddest-looking tree ever, which collapses when he puts a single bauble on it.
Years ago, my kids gave me a Charlie Brown tree as a gift. It even plays the late Vince Guaraldi’s immortal Peanuts theme when you push a button. I forgot about it for a few years, then discovered it, and immediately brought it to my office.
I’m not a fan of holiday creep, where they move up earlier in the year, so I used to put it up after Thanksgiving. But we close the office 2-3 weeks later for the rest of the year. I like the tree, my staff likes the tree, and my patients like the tree, so I just started putting it up in early November so we can enjoy it for a month.
It’s whimsical and brings back memories of innocence, childhood, and (of course) Peanuts. It sets a cheerful tone when you see it there. Very few of my patients can resist pressing the button and playing the music as they go by.
The start of a new year is a relatively arbitrary date, chosen long ago. But its approach is always a reminder that life goes on. We continue our trips around the sun. Good times and bad times come and go, but time never stops.
In bad years the tree reminds me that it’s coming to an end, and to look toward the next. In good years it reminds me that it’s time to be ready for the surprises of the coming one.
In mid-December, after the patients are done for the last day of the year, I quietly put it away. It’s a vaguely somber moment, but at the same time I’m glad to know I now have 2-3 weeks of home time. It mostly involves working at my desk and returning phone calls, but there’s also time to relax with my kids, do jigsaw puzzles, and enjoy the Phoenix winter weather as a break before the next round starts.
To those who disagree with my choice of decoration or its timing, I simply respond: “Good grief!”
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I put a Christmas tree up early in November.
It’s not like it’s a real tree, or even a fancy one. For that matter, I’m Jewish.
Growing up in the 1970s one thing that could be relied on every year was the Charlie Brown Christmas special. It never changed. By age 5 you knew most of the lines, and loved the highlight when Charlie Brown brings home the saddest-looking tree ever, which collapses when he puts a single bauble on it.
Years ago, my kids gave me a Charlie Brown tree as a gift. It even plays the late Vince Guaraldi’s immortal Peanuts theme when you push a button. I forgot about it for a few years, then discovered it, and immediately brought it to my office.
I’m not a fan of holiday creep, where they move up earlier in the year, so I used to put it up after Thanksgiving. But we close the office 2-3 weeks later for the rest of the year. I like the tree, my staff likes the tree, and my patients like the tree, so I just started putting it up in early November so we can enjoy it for a month.
It’s whimsical and brings back memories of innocence, childhood, and (of course) Peanuts. It sets a cheerful tone when you see it there. Very few of my patients can resist pressing the button and playing the music as they go by.
The start of a new year is a relatively arbitrary date, chosen long ago. But its approach is always a reminder that life goes on. We continue our trips around the sun. Good times and bad times come and go, but time never stops.
In bad years the tree reminds me that it’s coming to an end, and to look toward the next. In good years it reminds me that it’s time to be ready for the surprises of the coming one.
In mid-December, after the patients are done for the last day of the year, I quietly put it away. It’s a vaguely somber moment, but at the same time I’m glad to know I now have 2-3 weeks of home time. It mostly involves working at my desk and returning phone calls, but there’s also time to relax with my kids, do jigsaw puzzles, and enjoy the Phoenix winter weather as a break before the next round starts.
To those who disagree with my choice of decoration or its timing, I simply respond: “Good grief!”
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
First recommendations for cancer screening in myositis issued
AT ACR 2022
PHILADELPHIA – The first consensus screening guidelines for patients with idiopathic inflammatory myopathy (IIM) provide recommendations on risk stratification for individuals, basic and enhanced screening protocols, and screening frequency.
The recommendations, issued by the International Myositis Assessment and Clinical Studies Group (IMACS), stratify cancer risk for individual patients into low, intermediate, or high categories based on the IIM disease subtype, autoantibody status, and clinical features, reported Alexander Oldroyd, PhD, MSc, MBChB of the University of Manchester, England.
“There’s a big unmet need for cancer screening. One in four adults with myositis has cancer, either 3 years before or after a diagnosis of myositis. It’s one of the leading causes of death in these patients, and they’re overwhelmingly diagnosed at a late stage, so we need standardized approaches to get early diagnosis,” he said in an interview at the annual meeting of the American College of Rheumatology.
Sharon Kolasinski, MD, of the University of Pennsylvania in Philadelphia, said in an interview that the guideline is a welcome development for rheumatologists. Dr. Kolasinski moderated the session where Dr. Oldroyd described the guideline, but she was not involved in its formulation.
“I think that we all have wondered for a very long time: What is the optimal cancer screening for myositis patients? We all worry that the onset of their diseases is associated with a coincident cancer, or that they will develop it soon,” she said.
Dr. Oldroyd emphasized that all patients with myositis have elevated risk for cancer compared with the general population and that the guideline categories of low, intermediate, and high are relative only to patients with IIM.
International consensus
The data on which the recommendations are based come from a systematic review and meta-analysis by Dr. Oldroyd and colleagues of 69 studies on cancer risk factors and 9 on IIM-specific cancer screening.
The authors of that paper found that the dermatomyositis subtype, older age, male sex, dysphagia, cutaneous ulceration and antitranscriptional intermediary factor-1 gamma (anti-TIF1-gamma) positivity were associated with significantly increased risk of cancer.
In contrast, polymyositis and clinically amyopathic dermatomyositis subtypes, Raynaud’s phenomenon, interstitial lung disease, very high serum creatine kinase or lactate dehydrogenase levels, and positivity for anti-Jo1 or anti-EJ antibodies were associated with significantly reduced risk of cancer.
The consensus recommendations were developed with anonymous contributions from 75 expert participants in 22 countries, with additional input from 3 patient partners.
Do this
The guideline lists 18 recommendations, of which 13 are strong and 5 are conditional.
An example of a strong recommendation is number 3, based on a moderate level of evidences:
“All adult IIM patients, irrespective of cancer risk, should continue to participate in country/region-specific age and sex appropriate cancer screening programs,” the guideline recommends.
Patients with verified inclusion body myositis or juvenile-onset IIM do not, however, require routine screening for myositis-associated cancer, the guideline says (recommendations 1 and 2).
There are also recommendations that all adults with new-onset IIM be tested for myositis-specific and myositis-associated autoantibodies to assist in stratifying patients by risk category.
The guideline divides screening recommendations into basic and enhanced. The basic screening should include a comprehensive history and physical exam, complete blood count, liver functions tests, erythrocyte sedimentation rates/plasma viscosity, serum protein electrophoresis, urinalysis, and chest x-ray.
Adults with IIM who are determined to be at low risk for IIM-related cancer should have basic cancer screening at the time of IIM diagnosis. Adults with intermediate risk should undergo both basic and enhanced screening at the time of IIM diagnosis, and those with high risk should undergo enhanced screening at the time of myositis diagnosis, with basic screening annually for 3 years, the recommendations say.
Consider doing this
Conditional recommendations (“clinicians should consider ...”) include the use of PET/CT for adults at high risk for cancer when an underlying cancer has not been detected at the time of IIM diagnosis. They also include a single screening test for anti-TIF1-gamma positive dermatomyositis patients whose disease onset was after age 40 and who have at least one additional risk factor.
Also conditionally recommended are upper and lower gastrointestinal endoscopy for patients at high risk when an underlying cancer is not found at the time of IIM diagnosis, nasoendoscopy in geographical regions with elevated risk for nasopharyngeal cancers, and screening for all IIM patients with red-flag symptoms or clinical features of cancer, including unexplained weight loss, family history of cancer, smoking, unexplained fever, or night sweats.
Guided steps
“I think clinicians have a lot of questions such as, ‘well, what should I do, when should I do it?’ These are important clinical questions, and we need guidance about this. We need to balance comprehensiveness with cost-effectiveness, and we need expert opinion about what steps we should take now and which should we take later,” Dr. Kolasinski said.
The guideline development process was supported by the University of Manchester, IMACS, National Institute for Health Research (United Kingdom), National Institutes of Health, National Health Service Northern Care Alliance, The Myositis Association, Myositis UK, University of Pittsburgh, Versus Arthritis, and the Center for Musculoskeletal Research. Dr. Oldroyd and Dr. Kolasinski reported having no relevant conflicts of interest.
AT ACR 2022
PHILADELPHIA – The first consensus screening guidelines for patients with idiopathic inflammatory myopathy (IIM) provide recommendations on risk stratification for individuals, basic and enhanced screening protocols, and screening frequency.
The recommendations, issued by the International Myositis Assessment and Clinical Studies Group (IMACS), stratify cancer risk for individual patients into low, intermediate, or high categories based on the IIM disease subtype, autoantibody status, and clinical features, reported Alexander Oldroyd, PhD, MSc, MBChB of the University of Manchester, England.
“There’s a big unmet need for cancer screening. One in four adults with myositis has cancer, either 3 years before or after a diagnosis of myositis. It’s one of the leading causes of death in these patients, and they’re overwhelmingly diagnosed at a late stage, so we need standardized approaches to get early diagnosis,” he said in an interview at the annual meeting of the American College of Rheumatology.
Sharon Kolasinski, MD, of the University of Pennsylvania in Philadelphia, said in an interview that the guideline is a welcome development for rheumatologists. Dr. Kolasinski moderated the session where Dr. Oldroyd described the guideline, but she was not involved in its formulation.
“I think that we all have wondered for a very long time: What is the optimal cancer screening for myositis patients? We all worry that the onset of their diseases is associated with a coincident cancer, or that they will develop it soon,” she said.
Dr. Oldroyd emphasized that all patients with myositis have elevated risk for cancer compared with the general population and that the guideline categories of low, intermediate, and high are relative only to patients with IIM.
International consensus
The data on which the recommendations are based come from a systematic review and meta-analysis by Dr. Oldroyd and colleagues of 69 studies on cancer risk factors and 9 on IIM-specific cancer screening.
The authors of that paper found that the dermatomyositis subtype, older age, male sex, dysphagia, cutaneous ulceration and antitranscriptional intermediary factor-1 gamma (anti-TIF1-gamma) positivity were associated with significantly increased risk of cancer.
In contrast, polymyositis and clinically amyopathic dermatomyositis subtypes, Raynaud’s phenomenon, interstitial lung disease, very high serum creatine kinase or lactate dehydrogenase levels, and positivity for anti-Jo1 or anti-EJ antibodies were associated with significantly reduced risk of cancer.
The consensus recommendations were developed with anonymous contributions from 75 expert participants in 22 countries, with additional input from 3 patient partners.
Do this
The guideline lists 18 recommendations, of which 13 are strong and 5 are conditional.
An example of a strong recommendation is number 3, based on a moderate level of evidences:
“All adult IIM patients, irrespective of cancer risk, should continue to participate in country/region-specific age and sex appropriate cancer screening programs,” the guideline recommends.
Patients with verified inclusion body myositis or juvenile-onset IIM do not, however, require routine screening for myositis-associated cancer, the guideline says (recommendations 1 and 2).
There are also recommendations that all adults with new-onset IIM be tested for myositis-specific and myositis-associated autoantibodies to assist in stratifying patients by risk category.
The guideline divides screening recommendations into basic and enhanced. The basic screening should include a comprehensive history and physical exam, complete blood count, liver functions tests, erythrocyte sedimentation rates/plasma viscosity, serum protein electrophoresis, urinalysis, and chest x-ray.
Adults with IIM who are determined to be at low risk for IIM-related cancer should have basic cancer screening at the time of IIM diagnosis. Adults with intermediate risk should undergo both basic and enhanced screening at the time of IIM diagnosis, and those with high risk should undergo enhanced screening at the time of myositis diagnosis, with basic screening annually for 3 years, the recommendations say.
Consider doing this
Conditional recommendations (“clinicians should consider ...”) include the use of PET/CT for adults at high risk for cancer when an underlying cancer has not been detected at the time of IIM diagnosis. They also include a single screening test for anti-TIF1-gamma positive dermatomyositis patients whose disease onset was after age 40 and who have at least one additional risk factor.
Also conditionally recommended are upper and lower gastrointestinal endoscopy for patients at high risk when an underlying cancer is not found at the time of IIM diagnosis, nasoendoscopy in geographical regions with elevated risk for nasopharyngeal cancers, and screening for all IIM patients with red-flag symptoms or clinical features of cancer, including unexplained weight loss, family history of cancer, smoking, unexplained fever, or night sweats.
Guided steps
“I think clinicians have a lot of questions such as, ‘well, what should I do, when should I do it?’ These are important clinical questions, and we need guidance about this. We need to balance comprehensiveness with cost-effectiveness, and we need expert opinion about what steps we should take now and which should we take later,” Dr. Kolasinski said.
The guideline development process was supported by the University of Manchester, IMACS, National Institute for Health Research (United Kingdom), National Institutes of Health, National Health Service Northern Care Alliance, The Myositis Association, Myositis UK, University of Pittsburgh, Versus Arthritis, and the Center for Musculoskeletal Research. Dr. Oldroyd and Dr. Kolasinski reported having no relevant conflicts of interest.
AT ACR 2022
PHILADELPHIA – The first consensus screening guidelines for patients with idiopathic inflammatory myopathy (IIM) provide recommendations on risk stratification for individuals, basic and enhanced screening protocols, and screening frequency.
The recommendations, issued by the International Myositis Assessment and Clinical Studies Group (IMACS), stratify cancer risk for individual patients into low, intermediate, or high categories based on the IIM disease subtype, autoantibody status, and clinical features, reported Alexander Oldroyd, PhD, MSc, MBChB of the University of Manchester, England.
“There’s a big unmet need for cancer screening. One in four adults with myositis has cancer, either 3 years before or after a diagnosis of myositis. It’s one of the leading causes of death in these patients, and they’re overwhelmingly diagnosed at a late stage, so we need standardized approaches to get early diagnosis,” he said in an interview at the annual meeting of the American College of Rheumatology.
Sharon Kolasinski, MD, of the University of Pennsylvania in Philadelphia, said in an interview that the guideline is a welcome development for rheumatologists. Dr. Kolasinski moderated the session where Dr. Oldroyd described the guideline, but she was not involved in its formulation.
“I think that we all have wondered for a very long time: What is the optimal cancer screening for myositis patients? We all worry that the onset of their diseases is associated with a coincident cancer, or that they will develop it soon,” she said.
Dr. Oldroyd emphasized that all patients with myositis have elevated risk for cancer compared with the general population and that the guideline categories of low, intermediate, and high are relative only to patients with IIM.
International consensus
The data on which the recommendations are based come from a systematic review and meta-analysis by Dr. Oldroyd and colleagues of 69 studies on cancer risk factors and 9 on IIM-specific cancer screening.
The authors of that paper found that the dermatomyositis subtype, older age, male sex, dysphagia, cutaneous ulceration and antitranscriptional intermediary factor-1 gamma (anti-TIF1-gamma) positivity were associated with significantly increased risk of cancer.
In contrast, polymyositis and clinically amyopathic dermatomyositis subtypes, Raynaud’s phenomenon, interstitial lung disease, very high serum creatine kinase or lactate dehydrogenase levels, and positivity for anti-Jo1 or anti-EJ antibodies were associated with significantly reduced risk of cancer.
The consensus recommendations were developed with anonymous contributions from 75 expert participants in 22 countries, with additional input from 3 patient partners.
Do this
The guideline lists 18 recommendations, of which 13 are strong and 5 are conditional.
An example of a strong recommendation is number 3, based on a moderate level of evidences:
“All adult IIM patients, irrespective of cancer risk, should continue to participate in country/region-specific age and sex appropriate cancer screening programs,” the guideline recommends.
Patients with verified inclusion body myositis or juvenile-onset IIM do not, however, require routine screening for myositis-associated cancer, the guideline says (recommendations 1 and 2).
There are also recommendations that all adults with new-onset IIM be tested for myositis-specific and myositis-associated autoantibodies to assist in stratifying patients by risk category.
The guideline divides screening recommendations into basic and enhanced. The basic screening should include a comprehensive history and physical exam, complete blood count, liver functions tests, erythrocyte sedimentation rates/plasma viscosity, serum protein electrophoresis, urinalysis, and chest x-ray.
Adults with IIM who are determined to be at low risk for IIM-related cancer should have basic cancer screening at the time of IIM diagnosis. Adults with intermediate risk should undergo both basic and enhanced screening at the time of IIM diagnosis, and those with high risk should undergo enhanced screening at the time of myositis diagnosis, with basic screening annually for 3 years, the recommendations say.
Consider doing this
Conditional recommendations (“clinicians should consider ...”) include the use of PET/CT for adults at high risk for cancer when an underlying cancer has not been detected at the time of IIM diagnosis. They also include a single screening test for anti-TIF1-gamma positive dermatomyositis patients whose disease onset was after age 40 and who have at least one additional risk factor.
Also conditionally recommended are upper and lower gastrointestinal endoscopy for patients at high risk when an underlying cancer is not found at the time of IIM diagnosis, nasoendoscopy in geographical regions with elevated risk for nasopharyngeal cancers, and screening for all IIM patients with red-flag symptoms or clinical features of cancer, including unexplained weight loss, family history of cancer, smoking, unexplained fever, or night sweats.
Guided steps
“I think clinicians have a lot of questions such as, ‘well, what should I do, when should I do it?’ These are important clinical questions, and we need guidance about this. We need to balance comprehensiveness with cost-effectiveness, and we need expert opinion about what steps we should take now and which should we take later,” Dr. Kolasinski said.
The guideline development process was supported by the University of Manchester, IMACS, National Institute for Health Research (United Kingdom), National Institutes of Health, National Health Service Northern Care Alliance, The Myositis Association, Myositis UK, University of Pittsburgh, Versus Arthritis, and the Center for Musculoskeletal Research. Dr. Oldroyd and Dr. Kolasinski reported having no relevant conflicts of interest.
No benefit of rivaroxaban in COVID outpatients: PREVENT-HD
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
FROM AHA 2022
Glioblastoma spreading strategies discovered
Every year, around 7,000 people in Germany develop a brain tumor, and around half of those cases involve a glioblastoma, a particularly aggressive form of the disease. Glioblastomas are incurable, but advances are being made in both diagnostics and therapy.
This news organization spoke to Wolfgang Wick, MD, medical director of the neurologic clinic at UKHD, about how glioblastomas are treated; the role that vaccinations, recombinant proteins, and parvoviruses play; and what therapeutic approaches might be derived from the discovery of this method by which glioblastomas spread.
Question: Glioblastomas spread through the brain like a fungal network. So how would a glioblastoma currently be treated? The tumor can only be partially removed through surgery.
Answer: Nevertheless, glioblastoma would be operated on. This would have a significant effect. Relieving the strain of the main tumor mass, without generating a new deficit, is prognostically very good for the patient concerned. However, surgery on glioblastoma is never curative.
The reason a cure is not possible is down to the special form and spread of the glioblastoma. Nevertheless, an operation helps. This seems to be because removing the main tumor mass maybe has a positive immunological effect. But it may also be connected to the tumor’s network communication. The surgical intervention stimulates the network by increasing resistance.
If the main tumor mass is decreased through a surgical procedure, this results in an at least temporarily improved starting position for the patient until the mass regenerates. This could also be connected to the fact that tumor communication is not unregulated but is rather in accordance with a certain hierarchy and order, which requires a certain structure and mass.
The other aspect is that support can be requested via this communication. You can imagine that a cell connected to another cell via a conduit receives help from this other cell in the form of organelles by exchanging ions and that, for example, stress or toxicity can be much better balanced out in large networks than in small networks. That means that external attacks, such as a surgical intervention, can be much better balanced by a well-organized network than by isolated cells.
Resistance to chemotherapy
Q: How do irradiation and chemotherapy rank in the treatment of glioblastomas?
A: Irradiation is another therapeutic approach. It causes cells to be stuck in the growth phase of the cell cycle. The cells are not killed through radiation, but they are practically halted. And this arrest of the cell cycle is often sufficient to help people with glioblastomas for a very long time. But the same is true for irradiation as for surgery. This deep network of cells cannot be addressed.
Attempts have been made in the past to reduce the radiation dose to the extent that the brain is no longer damaged by it, but this low dose was then not sufficient to exert any control. If you want to control the tumor, the dose must be high and the volume must be correspondingly low, since there is a clear limit.
Every patient is offered alkylating chemotherapy. At the moment, just one substance is used here in the primary therapy: temozolomide. The problem with this is that two-thirds of tumors in all cells exhibit a resistance to this alkylating chemotherapy, which means that the efficacy of this therapy is highly limited in two-thirds of patients.
In the one-third of patients in whom this resistance is not present, the chemotherapy works fairly well. But even then, it is unfortunately only a matter of time until there is a relapse or disease progression. In my practice, this has always been the case, but there are people who have been living with this disease for 20 years now. There seem to be tumor cells that calmly and silently survive this phase of chemotherapy and then restart the cell cycle at some point.
Q: What do you think of alternating electric fields as a therapy option?
A: Therapy with alternating electric fields is currently being used and offered to patients. This means that patients who have survived well through radiochemotherapy should also be offered treatment with alternating electric fields.
However, what happens in this process is not as well understood as with other therapies. It is assumed that the cell cycle, i.e., cell division, is altered by disrupting the mitotic spindle. But you can imagine, and this is now speculation, but quite sound speculation I believe, that alternating electric fields also cause a certain amount of confusion in the previously described networks. But this still needs to be investigated in more detail.
It is not implausible. We know that such alternating electric fields disturb the organization of cell organelles. And we also know that for this communication, we need fairly good order and also organization. This would definitely be a starting point on the way to understanding why this therapy potentially shows a certain effect in some patients.
Nerve cell precursors
Q: Scientists from the UKHD and the DKFZ have discovered a new glioblastoma spreading strategy and have learned that the tumor cells imitate the properties and movement patterns of nerve cells. They are labeling the results a “milestone in the field of cancer neuroscience.” Could you explain a bit more?
A: Glioblastoma does not grow on its own as a solid mass, but instead, the entire brain is affected by the disease. The question of how the tumor’s individual cells move the main tumor mass from afar, how they get there, how they continue to be supplied, and what their interaction partners are – an entirely new light has been shed on all of this in our work.
The development of tumor cell mobility has been recognized as a remnant of brain development. The tumor cells have retained properties that the precursor cells for nervous-system development require for an organized nervous system to emerge from just a few cells. This means that the tumor cells copy or eventually retain properties of the nerve-cell precursors that, unlike mature nerve cells, are mobile to a fairly high degree.
Mobility here means that it can advance along a network, despite said network being very densely packed. This also means that certain processes, such as releasing and then continuing to move again, must function and that the communication regarding the original disease must be maintained.
First, we understand what the different glioblastoma cell types do, which molecular properties are associated with which behaviors, and which cell type (namely the swarming cells) is responsible for the invasive tumor growth. In contrast, the network-forming cell type, which only develops from these, is responsible for the resistance.
Interrupting communication
Q: Which starting points for new therapies do you see?
A: In terms of new therapies, these movement phenomena are one good starting point. The other starting point – I find this one much more interesting – is that the programming steps that these tumor cells use [are] no longer needed. This is because our mature nervous system no longer requires this program, which was necessary for the mobility of cells in development.
Our central nervous system exhibits little cell movement. This is to do with programs of nervous-system development that are switched off in the mature nervous system. But they are then reactivated or remain active in the tumor cells. This process reveals potential starting points for therapy.
Addressing the movement of cells, that has been investigated for the last 20 years, but it seems to have an extraordinarily high number of side effects, because these movement mechanisms are also important for other, healthy cells in the body. For example, digestive mechanisms and other proliferation mechanisms, on mucous membranes, in the blood system, in the bone marrow, are then affected and no longer function.
There is another possible approach: the more-or-less specific interaction between the nerve cells and the tumor cells also offers starting points for therapies, from our point of view. The key word is epilepsy treatment. We know that people with brain tumors suffer badly, or worse than usual, from epileptic seizures. This was often regarded purely as a pressure problem. There is a disruptive element in the brain, and this causes the electrical activity in the brain to become disorganized. For some people, this can lead to seizures in certain situations.
The communication between tumor cells and nerve cells takes place via transmission substances, e.g., through the neurotransmitter glutamate. Now you can consider whether a “surplus” of communication, such as an excessively strong stimulus, can trigger epileptic seizures.
In this work, we demonstrate that by interrupting this communication, we can also prevent the movement of these cells and the growth, the proliferation, of these cells.
Q: What is the significance of parvoviruses for therapy?
A: The major topic for cancer is immunotherapy. And one option for performing immunotherapies lies with viruses. Parvoviruses are a plausible therapy for proliferating cells.
Parvoviruses are usually administered locally. This means that a surgical cavity is infected with the viruses and the tumor cells that remain after an operation will then hopefully be killed off by these viruses.
This is the first step and the immediate effect of virus therapy. The attempt is made to kill off cells in the same way as with a medication. The advantage of viruses is the high specificity, i.e., only dividing cells will be attacked. In addition, parvoviruses are so small that they can also spread well and circulate through the brain.
The second reason for immunotherapy is that when killing off cells with viruses, antigens are often released that otherwise would not be, depending on the virus. But it’s the case with parvoviruses. They integrate with the virus’s genetic material. When cells rupture, certain proteins are then revealed, hybrids of viruses and the human genome, and these are attractive to the immune system.
There is a whole range of studies on this subject. However, there are currently no randomized studies that directly compare the therapies. But the expectation is that the use of parvoviruses could be a good addition to therapy.
One limitation that should be mentioned is that the use of viruses may be beneficial for some patients, but it will not have an effect in every patient. What is exciting about parvoviruses is that these viruses can be injected via the bloodstream and still achieve a good effect in the brain.
Protein APG101
Q: How relevant is the recombinant protein APG101 to therapy?
A: APG101 is a protein that simulates the cell-death receptor CD95 and binds with a stable antibody fragment. By doing so, it blocks the signaling pathway between CD95 ligand and receptor. The interaction between the CD95 ligand and the CD95 receptor activates an intracellular signaling pathway, which in turn stimulates the invasive growth and migration of tumor cells.
APG101 blocks the CD95 ligand and thereby prevents the activation of the CD95 signaling pathway, which leads to a reduction in the invasive cell growth and migration.
Apoptosis, programmed cell death, is a system we have used throughout our evolution to kill off the cell components we no longer need. During tumor development, this system is perverted, so to speak. Here, the stimulation of this system does not actually lead to cell death but rather to cell movement (i.e., to cell mobility). And in principle, APG101 blocks this mobility.
To date, I only know of three studies in which the medication has been used for tumors. One study was published 8 years ago. We demonstrated that we can achieve a relatively good effect with APG101 in connection with repeat irradiation, compared with repeat irradiation alone. We consider this effect to most likely be due to this influence on cell mobility.
There is a study on primary therapy: a four-arm study by the Neuro-Oncological Working Group. The results are still not available, however. In addition, a study on primary therapy with APG101 is currently being conducted in China. It is investigating whether the mechanism of action influences mobility. Whether it will be pushed through as therapy remains to be seen.
Vaccinations and antigens
Q: Vaccinations are of course a part of immunotherapy. What is their status?
A: We are looking at the IDH1 protein, which is present in mutated form in a group of brain tumors, as a very good target for a vaccine. The reason is that the protein is present in its mutated form in every cell of the tumor but not in healthy cells. That is a prerequisite for immunotherapy.
We started a study with peptides a few years ago. These peptides are injected under the skin on the stomach and leg. They cause an immune response systemically and in the brain tumor. This immune response may cause an inflammatory reaction (we can demonstrate this inflammatory reaction). And in this noncontrolled study, the approach was successful, at least compared to historical controls. There is no randomized study with treatment-naive control patients.
However, we are cautious because we know that peptide, unlike CAR T cells or RNA-based vaccines, for example, only triggers a relatively small immune response in many patients. The scale of the immune response is important, rather than the specificity. The scale is probably not large enough in most patients for a long-term effect to be expected.
But there are exceptions. Patients we vaccinated many years ago still have a very remarkable immune status. But we also have patients in whom an immune status cannot even be seen anymore, after just a short period of time.
Therefore, our aim is to perform the immune strategy with more effective, stronger measures – not more specific, but stronger. Unfortunately, it is often the case with glioblastomas that there is not a single antigen that can be vaccinated against. Instead, a relatively large cocktail is needed, which unfortunately also often varies from patient to patient. The conditions are difficult.
Q: You mentioned that glioblastomas can be classified into subgroups. Does this improve the prognosis?
A: Yes, in certain subgroups the prognosis improves. That is the case with those usually very small groups that are molecularly well defined. I believe that by better understanding the individual groups, we have succeeded in making major progress in those groups. But where there is light, there is also shadow. We know that there are many groups with which we have not achieved a great deal.
Fundamental research leads to a better understanding, and the next step in this is to be able to adapt the therapy. Instead of it being one therapy for everyone, it will become a part of various differing therapies for these quite different groups. We are making a lot of progress with individual groups. But unfortunately, we have not come quite as far as we want with many patients.
This article was translated from the Medscape German edition. A version of this article first appeared on Medscape.com.
Every year, around 7,000 people in Germany develop a brain tumor, and around half of those cases involve a glioblastoma, a particularly aggressive form of the disease. Glioblastomas are incurable, but advances are being made in both diagnostics and therapy.
This news organization spoke to Wolfgang Wick, MD, medical director of the neurologic clinic at UKHD, about how glioblastomas are treated; the role that vaccinations, recombinant proteins, and parvoviruses play; and what therapeutic approaches might be derived from the discovery of this method by which glioblastomas spread.
Question: Glioblastomas spread through the brain like a fungal network. So how would a glioblastoma currently be treated? The tumor can only be partially removed through surgery.
Answer: Nevertheless, glioblastoma would be operated on. This would have a significant effect. Relieving the strain of the main tumor mass, without generating a new deficit, is prognostically very good for the patient concerned. However, surgery on glioblastoma is never curative.
The reason a cure is not possible is down to the special form and spread of the glioblastoma. Nevertheless, an operation helps. This seems to be because removing the main tumor mass maybe has a positive immunological effect. But it may also be connected to the tumor’s network communication. The surgical intervention stimulates the network by increasing resistance.
If the main tumor mass is decreased through a surgical procedure, this results in an at least temporarily improved starting position for the patient until the mass regenerates. This could also be connected to the fact that tumor communication is not unregulated but is rather in accordance with a certain hierarchy and order, which requires a certain structure and mass.
The other aspect is that support can be requested via this communication. You can imagine that a cell connected to another cell via a conduit receives help from this other cell in the form of organelles by exchanging ions and that, for example, stress or toxicity can be much better balanced out in large networks than in small networks. That means that external attacks, such as a surgical intervention, can be much better balanced by a well-organized network than by isolated cells.
Resistance to chemotherapy
Q: How do irradiation and chemotherapy rank in the treatment of glioblastomas?
A: Irradiation is another therapeutic approach. It causes cells to be stuck in the growth phase of the cell cycle. The cells are not killed through radiation, but they are practically halted. And this arrest of the cell cycle is often sufficient to help people with glioblastomas for a very long time. But the same is true for irradiation as for surgery. This deep network of cells cannot be addressed.
Attempts have been made in the past to reduce the radiation dose to the extent that the brain is no longer damaged by it, but this low dose was then not sufficient to exert any control. If you want to control the tumor, the dose must be high and the volume must be correspondingly low, since there is a clear limit.
Every patient is offered alkylating chemotherapy. At the moment, just one substance is used here in the primary therapy: temozolomide. The problem with this is that two-thirds of tumors in all cells exhibit a resistance to this alkylating chemotherapy, which means that the efficacy of this therapy is highly limited in two-thirds of patients.
In the one-third of patients in whom this resistance is not present, the chemotherapy works fairly well. But even then, it is unfortunately only a matter of time until there is a relapse or disease progression. In my practice, this has always been the case, but there are people who have been living with this disease for 20 years now. There seem to be tumor cells that calmly and silently survive this phase of chemotherapy and then restart the cell cycle at some point.
Q: What do you think of alternating electric fields as a therapy option?
A: Therapy with alternating electric fields is currently being used and offered to patients. This means that patients who have survived well through radiochemotherapy should also be offered treatment with alternating electric fields.
However, what happens in this process is not as well understood as with other therapies. It is assumed that the cell cycle, i.e., cell division, is altered by disrupting the mitotic spindle. But you can imagine, and this is now speculation, but quite sound speculation I believe, that alternating electric fields also cause a certain amount of confusion in the previously described networks. But this still needs to be investigated in more detail.
It is not implausible. We know that such alternating electric fields disturb the organization of cell organelles. And we also know that for this communication, we need fairly good order and also organization. This would definitely be a starting point on the way to understanding why this therapy potentially shows a certain effect in some patients.
Nerve cell precursors
Q: Scientists from the UKHD and the DKFZ have discovered a new glioblastoma spreading strategy and have learned that the tumor cells imitate the properties and movement patterns of nerve cells. They are labeling the results a “milestone in the field of cancer neuroscience.” Could you explain a bit more?
A: Glioblastoma does not grow on its own as a solid mass, but instead, the entire brain is affected by the disease. The question of how the tumor’s individual cells move the main tumor mass from afar, how they get there, how they continue to be supplied, and what their interaction partners are – an entirely new light has been shed on all of this in our work.
The development of tumor cell mobility has been recognized as a remnant of brain development. The tumor cells have retained properties that the precursor cells for nervous-system development require for an organized nervous system to emerge from just a few cells. This means that the tumor cells copy or eventually retain properties of the nerve-cell precursors that, unlike mature nerve cells, are mobile to a fairly high degree.
Mobility here means that it can advance along a network, despite said network being very densely packed. This also means that certain processes, such as releasing and then continuing to move again, must function and that the communication regarding the original disease must be maintained.
First, we understand what the different glioblastoma cell types do, which molecular properties are associated with which behaviors, and which cell type (namely the swarming cells) is responsible for the invasive tumor growth. In contrast, the network-forming cell type, which only develops from these, is responsible for the resistance.
Interrupting communication
Q: Which starting points for new therapies do you see?
A: In terms of new therapies, these movement phenomena are one good starting point. The other starting point – I find this one much more interesting – is that the programming steps that these tumor cells use [are] no longer needed. This is because our mature nervous system no longer requires this program, which was necessary for the mobility of cells in development.
Our central nervous system exhibits little cell movement. This is to do with programs of nervous-system development that are switched off in the mature nervous system. But they are then reactivated or remain active in the tumor cells. This process reveals potential starting points for therapy.
Addressing the movement of cells, that has been investigated for the last 20 years, but it seems to have an extraordinarily high number of side effects, because these movement mechanisms are also important for other, healthy cells in the body. For example, digestive mechanisms and other proliferation mechanisms, on mucous membranes, in the blood system, in the bone marrow, are then affected and no longer function.
There is another possible approach: the more-or-less specific interaction between the nerve cells and the tumor cells also offers starting points for therapies, from our point of view. The key word is epilepsy treatment. We know that people with brain tumors suffer badly, or worse than usual, from epileptic seizures. This was often regarded purely as a pressure problem. There is a disruptive element in the brain, and this causes the electrical activity in the brain to become disorganized. For some people, this can lead to seizures in certain situations.
The communication between tumor cells and nerve cells takes place via transmission substances, e.g., through the neurotransmitter glutamate. Now you can consider whether a “surplus” of communication, such as an excessively strong stimulus, can trigger epileptic seizures.
In this work, we demonstrate that by interrupting this communication, we can also prevent the movement of these cells and the growth, the proliferation, of these cells.
Q: What is the significance of parvoviruses for therapy?
A: The major topic for cancer is immunotherapy. And one option for performing immunotherapies lies with viruses. Parvoviruses are a plausible therapy for proliferating cells.
Parvoviruses are usually administered locally. This means that a surgical cavity is infected with the viruses and the tumor cells that remain after an operation will then hopefully be killed off by these viruses.
This is the first step and the immediate effect of virus therapy. The attempt is made to kill off cells in the same way as with a medication. The advantage of viruses is the high specificity, i.e., only dividing cells will be attacked. In addition, parvoviruses are so small that they can also spread well and circulate through the brain.
The second reason for immunotherapy is that when killing off cells with viruses, antigens are often released that otherwise would not be, depending on the virus. But it’s the case with parvoviruses. They integrate with the virus’s genetic material. When cells rupture, certain proteins are then revealed, hybrids of viruses and the human genome, and these are attractive to the immune system.
There is a whole range of studies on this subject. However, there are currently no randomized studies that directly compare the therapies. But the expectation is that the use of parvoviruses could be a good addition to therapy.
One limitation that should be mentioned is that the use of viruses may be beneficial for some patients, but it will not have an effect in every patient. What is exciting about parvoviruses is that these viruses can be injected via the bloodstream and still achieve a good effect in the brain.
Protein APG101
Q: How relevant is the recombinant protein APG101 to therapy?
A: APG101 is a protein that simulates the cell-death receptor CD95 and binds with a stable antibody fragment. By doing so, it blocks the signaling pathway between CD95 ligand and receptor. The interaction between the CD95 ligand and the CD95 receptor activates an intracellular signaling pathway, which in turn stimulates the invasive growth and migration of tumor cells.
APG101 blocks the CD95 ligand and thereby prevents the activation of the CD95 signaling pathway, which leads to a reduction in the invasive cell growth and migration.
Apoptosis, programmed cell death, is a system we have used throughout our evolution to kill off the cell components we no longer need. During tumor development, this system is perverted, so to speak. Here, the stimulation of this system does not actually lead to cell death but rather to cell movement (i.e., to cell mobility). And in principle, APG101 blocks this mobility.
To date, I only know of three studies in which the medication has been used for tumors. One study was published 8 years ago. We demonstrated that we can achieve a relatively good effect with APG101 in connection with repeat irradiation, compared with repeat irradiation alone. We consider this effect to most likely be due to this influence on cell mobility.
There is a study on primary therapy: a four-arm study by the Neuro-Oncological Working Group. The results are still not available, however. In addition, a study on primary therapy with APG101 is currently being conducted in China. It is investigating whether the mechanism of action influences mobility. Whether it will be pushed through as therapy remains to be seen.
Vaccinations and antigens
Q: Vaccinations are of course a part of immunotherapy. What is their status?
A: We are looking at the IDH1 protein, which is present in mutated form in a group of brain tumors, as a very good target for a vaccine. The reason is that the protein is present in its mutated form in every cell of the tumor but not in healthy cells. That is a prerequisite for immunotherapy.
We started a study with peptides a few years ago. These peptides are injected under the skin on the stomach and leg. They cause an immune response systemically and in the brain tumor. This immune response may cause an inflammatory reaction (we can demonstrate this inflammatory reaction). And in this noncontrolled study, the approach was successful, at least compared to historical controls. There is no randomized study with treatment-naive control patients.
However, we are cautious because we know that peptide, unlike CAR T cells or RNA-based vaccines, for example, only triggers a relatively small immune response in many patients. The scale of the immune response is important, rather than the specificity. The scale is probably not large enough in most patients for a long-term effect to be expected.
But there are exceptions. Patients we vaccinated many years ago still have a very remarkable immune status. But we also have patients in whom an immune status cannot even be seen anymore, after just a short period of time.
Therefore, our aim is to perform the immune strategy with more effective, stronger measures – not more specific, but stronger. Unfortunately, it is often the case with glioblastomas that there is not a single antigen that can be vaccinated against. Instead, a relatively large cocktail is needed, which unfortunately also often varies from patient to patient. The conditions are difficult.
Q: You mentioned that glioblastomas can be classified into subgroups. Does this improve the prognosis?
A: Yes, in certain subgroups the prognosis improves. That is the case with those usually very small groups that are molecularly well defined. I believe that by better understanding the individual groups, we have succeeded in making major progress in those groups. But where there is light, there is also shadow. We know that there are many groups with which we have not achieved a great deal.
Fundamental research leads to a better understanding, and the next step in this is to be able to adapt the therapy. Instead of it being one therapy for everyone, it will become a part of various differing therapies for these quite different groups. We are making a lot of progress with individual groups. But unfortunately, we have not come quite as far as we want with many patients.
This article was translated from the Medscape German edition. A version of this article first appeared on Medscape.com.
Every year, around 7,000 people in Germany develop a brain tumor, and around half of those cases involve a glioblastoma, a particularly aggressive form of the disease. Glioblastomas are incurable, but advances are being made in both diagnostics and therapy.
This news organization spoke to Wolfgang Wick, MD, medical director of the neurologic clinic at UKHD, about how glioblastomas are treated; the role that vaccinations, recombinant proteins, and parvoviruses play; and what therapeutic approaches might be derived from the discovery of this method by which glioblastomas spread.
Question: Glioblastomas spread through the brain like a fungal network. So how would a glioblastoma currently be treated? The tumor can only be partially removed through surgery.
Answer: Nevertheless, glioblastoma would be operated on. This would have a significant effect. Relieving the strain of the main tumor mass, without generating a new deficit, is prognostically very good for the patient concerned. However, surgery on glioblastoma is never curative.
The reason a cure is not possible is down to the special form and spread of the glioblastoma. Nevertheless, an operation helps. This seems to be because removing the main tumor mass maybe has a positive immunological effect. But it may also be connected to the tumor’s network communication. The surgical intervention stimulates the network by increasing resistance.
If the main tumor mass is decreased through a surgical procedure, this results in an at least temporarily improved starting position for the patient until the mass regenerates. This could also be connected to the fact that tumor communication is not unregulated but is rather in accordance with a certain hierarchy and order, which requires a certain structure and mass.
The other aspect is that support can be requested via this communication. You can imagine that a cell connected to another cell via a conduit receives help from this other cell in the form of organelles by exchanging ions and that, for example, stress or toxicity can be much better balanced out in large networks than in small networks. That means that external attacks, such as a surgical intervention, can be much better balanced by a well-organized network than by isolated cells.
Resistance to chemotherapy
Q: How do irradiation and chemotherapy rank in the treatment of glioblastomas?
A: Irradiation is another therapeutic approach. It causes cells to be stuck in the growth phase of the cell cycle. The cells are not killed through radiation, but they are practically halted. And this arrest of the cell cycle is often sufficient to help people with glioblastomas for a very long time. But the same is true for irradiation as for surgery. This deep network of cells cannot be addressed.
Attempts have been made in the past to reduce the radiation dose to the extent that the brain is no longer damaged by it, but this low dose was then not sufficient to exert any control. If you want to control the tumor, the dose must be high and the volume must be correspondingly low, since there is a clear limit.
Every patient is offered alkylating chemotherapy. At the moment, just one substance is used here in the primary therapy: temozolomide. The problem with this is that two-thirds of tumors in all cells exhibit a resistance to this alkylating chemotherapy, which means that the efficacy of this therapy is highly limited in two-thirds of patients.
In the one-third of patients in whom this resistance is not present, the chemotherapy works fairly well. But even then, it is unfortunately only a matter of time until there is a relapse or disease progression. In my practice, this has always been the case, but there are people who have been living with this disease for 20 years now. There seem to be tumor cells that calmly and silently survive this phase of chemotherapy and then restart the cell cycle at some point.
Q: What do you think of alternating electric fields as a therapy option?
A: Therapy with alternating electric fields is currently being used and offered to patients. This means that patients who have survived well through radiochemotherapy should also be offered treatment with alternating electric fields.
However, what happens in this process is not as well understood as with other therapies. It is assumed that the cell cycle, i.e., cell division, is altered by disrupting the mitotic spindle. But you can imagine, and this is now speculation, but quite sound speculation I believe, that alternating electric fields also cause a certain amount of confusion in the previously described networks. But this still needs to be investigated in more detail.
It is not implausible. We know that such alternating electric fields disturb the organization of cell organelles. And we also know that for this communication, we need fairly good order and also organization. This would definitely be a starting point on the way to understanding why this therapy potentially shows a certain effect in some patients.
Nerve cell precursors
Q: Scientists from the UKHD and the DKFZ have discovered a new glioblastoma spreading strategy and have learned that the tumor cells imitate the properties and movement patterns of nerve cells. They are labeling the results a “milestone in the field of cancer neuroscience.” Could you explain a bit more?
A: Glioblastoma does not grow on its own as a solid mass, but instead, the entire brain is affected by the disease. The question of how the tumor’s individual cells move the main tumor mass from afar, how they get there, how they continue to be supplied, and what their interaction partners are – an entirely new light has been shed on all of this in our work.
The development of tumor cell mobility has been recognized as a remnant of brain development. The tumor cells have retained properties that the precursor cells for nervous-system development require for an organized nervous system to emerge from just a few cells. This means that the tumor cells copy or eventually retain properties of the nerve-cell precursors that, unlike mature nerve cells, are mobile to a fairly high degree.
Mobility here means that it can advance along a network, despite said network being very densely packed. This also means that certain processes, such as releasing and then continuing to move again, must function and that the communication regarding the original disease must be maintained.
First, we understand what the different glioblastoma cell types do, which molecular properties are associated with which behaviors, and which cell type (namely the swarming cells) is responsible for the invasive tumor growth. In contrast, the network-forming cell type, which only develops from these, is responsible for the resistance.
Interrupting communication
Q: Which starting points for new therapies do you see?
A: In terms of new therapies, these movement phenomena are one good starting point. The other starting point – I find this one much more interesting – is that the programming steps that these tumor cells use [are] no longer needed. This is because our mature nervous system no longer requires this program, which was necessary for the mobility of cells in development.
Our central nervous system exhibits little cell movement. This is to do with programs of nervous-system development that are switched off in the mature nervous system. But they are then reactivated or remain active in the tumor cells. This process reveals potential starting points for therapy.
Addressing the movement of cells, that has been investigated for the last 20 years, but it seems to have an extraordinarily high number of side effects, because these movement mechanisms are also important for other, healthy cells in the body. For example, digestive mechanisms and other proliferation mechanisms, on mucous membranes, in the blood system, in the bone marrow, are then affected and no longer function.
There is another possible approach: the more-or-less specific interaction between the nerve cells and the tumor cells also offers starting points for therapies, from our point of view. The key word is epilepsy treatment. We know that people with brain tumors suffer badly, or worse than usual, from epileptic seizures. This was often regarded purely as a pressure problem. There is a disruptive element in the brain, and this causes the electrical activity in the brain to become disorganized. For some people, this can lead to seizures in certain situations.
The communication between tumor cells and nerve cells takes place via transmission substances, e.g., through the neurotransmitter glutamate. Now you can consider whether a “surplus” of communication, such as an excessively strong stimulus, can trigger epileptic seizures.
In this work, we demonstrate that by interrupting this communication, we can also prevent the movement of these cells and the growth, the proliferation, of these cells.
Q: What is the significance of parvoviruses for therapy?
A: The major topic for cancer is immunotherapy. And one option for performing immunotherapies lies with viruses. Parvoviruses are a plausible therapy for proliferating cells.
Parvoviruses are usually administered locally. This means that a surgical cavity is infected with the viruses and the tumor cells that remain after an operation will then hopefully be killed off by these viruses.
This is the first step and the immediate effect of virus therapy. The attempt is made to kill off cells in the same way as with a medication. The advantage of viruses is the high specificity, i.e., only dividing cells will be attacked. In addition, parvoviruses are so small that they can also spread well and circulate through the brain.
The second reason for immunotherapy is that when killing off cells with viruses, antigens are often released that otherwise would not be, depending on the virus. But it’s the case with parvoviruses. They integrate with the virus’s genetic material. When cells rupture, certain proteins are then revealed, hybrids of viruses and the human genome, and these are attractive to the immune system.
There is a whole range of studies on this subject. However, there are currently no randomized studies that directly compare the therapies. But the expectation is that the use of parvoviruses could be a good addition to therapy.
One limitation that should be mentioned is that the use of viruses may be beneficial for some patients, but it will not have an effect in every patient. What is exciting about parvoviruses is that these viruses can be injected via the bloodstream and still achieve a good effect in the brain.
Protein APG101
Q: How relevant is the recombinant protein APG101 to therapy?
A: APG101 is a protein that simulates the cell-death receptor CD95 and binds with a stable antibody fragment. By doing so, it blocks the signaling pathway between CD95 ligand and receptor. The interaction between the CD95 ligand and the CD95 receptor activates an intracellular signaling pathway, which in turn stimulates the invasive growth and migration of tumor cells.
APG101 blocks the CD95 ligand and thereby prevents the activation of the CD95 signaling pathway, which leads to a reduction in the invasive cell growth and migration.
Apoptosis, programmed cell death, is a system we have used throughout our evolution to kill off the cell components we no longer need. During tumor development, this system is perverted, so to speak. Here, the stimulation of this system does not actually lead to cell death but rather to cell movement (i.e., to cell mobility). And in principle, APG101 blocks this mobility.
To date, I only know of three studies in which the medication has been used for tumors. One study was published 8 years ago. We demonstrated that we can achieve a relatively good effect with APG101 in connection with repeat irradiation, compared with repeat irradiation alone. We consider this effect to most likely be due to this influence on cell mobility.
There is a study on primary therapy: a four-arm study by the Neuro-Oncological Working Group. The results are still not available, however. In addition, a study on primary therapy with APG101 is currently being conducted in China. It is investigating whether the mechanism of action influences mobility. Whether it will be pushed through as therapy remains to be seen.
Vaccinations and antigens
Q: Vaccinations are of course a part of immunotherapy. What is their status?
A: We are looking at the IDH1 protein, which is present in mutated form in a group of brain tumors, as a very good target for a vaccine. The reason is that the protein is present in its mutated form in every cell of the tumor but not in healthy cells. That is a prerequisite for immunotherapy.
We started a study with peptides a few years ago. These peptides are injected under the skin on the stomach and leg. They cause an immune response systemically and in the brain tumor. This immune response may cause an inflammatory reaction (we can demonstrate this inflammatory reaction). And in this noncontrolled study, the approach was successful, at least compared to historical controls. There is no randomized study with treatment-naive control patients.
However, we are cautious because we know that peptide, unlike CAR T cells or RNA-based vaccines, for example, only triggers a relatively small immune response in many patients. The scale of the immune response is important, rather than the specificity. The scale is probably not large enough in most patients for a long-term effect to be expected.
But there are exceptions. Patients we vaccinated many years ago still have a very remarkable immune status. But we also have patients in whom an immune status cannot even be seen anymore, after just a short period of time.
Therefore, our aim is to perform the immune strategy with more effective, stronger measures – not more specific, but stronger. Unfortunately, it is often the case with glioblastomas that there is not a single antigen that can be vaccinated against. Instead, a relatively large cocktail is needed, which unfortunately also often varies from patient to patient. The conditions are difficult.
Q: You mentioned that glioblastomas can be classified into subgroups. Does this improve the prognosis?
A: Yes, in certain subgroups the prognosis improves. That is the case with those usually very small groups that are molecularly well defined. I believe that by better understanding the individual groups, we have succeeded in making major progress in those groups. But where there is light, there is also shadow. We know that there are many groups with which we have not achieved a great deal.
Fundamental research leads to a better understanding, and the next step in this is to be able to adapt the therapy. Instead of it being one therapy for everyone, it will become a part of various differing therapies for these quite different groups. We are making a lot of progress with individual groups. But unfortunately, we have not come quite as far as we want with many patients.
This article was translated from the Medscape German edition. A version of this article first appeared on Medscape.com.
Have you heard the one about the emergency dept. that called 911?
Who watches the ED staff?
We heard a really great joke recently, one we simply have to share.
A man in Seattle went to a therapist. “I’m depressed,” he says. “Depressed, overworked, and lonely.”
“Oh dear, that sounds quite serious,” the therapist replies. “Tell me all about it.”
“Life just seems so harsh and cruel,” the man explains. “The pandemic has caused 300,000 health care workers across the country to leave the industry.”
“Such as the doctor typically filling this role in the joke,” the therapist, who is not licensed to prescribe medicine, nods.
“Exactly! And with so many respiratory viruses circulating and COVID still hanging around, emergency departments all over the country are facing massive backups. People are waiting outside the hospital for hours, hoping a bed will open up. Things got so bad at a hospital near Seattle in October that a nurse called 911 on her own ED. Told the 911 operator to send the fire department to help out, since they were ‘drowning’ and ‘in dire straits.’ They had 45 patients waiting and only five nurses to take care of them.”
“That is quite serious,” the therapist says, scribbling down unseen notes.
“The fire chief did send a crew out, and they cleaned rooms, changed beds, and took vitals for 90 minutes until the crisis passed,” the man says. “But it’s only a matter of time before it happens again. The hospital president said they have 300 open positions, and literally no one has applied to work in the emergency department. Not one person.”
“And how does all this make you feel?” the therapist asks.
“I feel all alone,” the man says. “This world feels so threatening, like no one cares, and I have no idea what will come next. It’s so vague and uncertain.”
“Ah, I think I have a solution for you,” the therapist says. “Go to the emergency department at St. Michael Medical Center in Silverdale, near Seattle. They’ll get your bad mood all settled, and they’ll prescribe you the medicine you need to relax.”
The man bursts into tears. “You don’t understand,” he says. “I am the emergency department at St. Michael Medical Center.”
Good joke. Everybody laugh. Roll on snare drum. Curtains.
Myth buster: Supplements for cholesterol lowering
When it comes to that nasty low-density lipoprotein cholesterol, some people swear by supplements over statins as a holistic approach. Well, we’re busting the myth that those heart-healthy supplements are even effective in comparison.
Which supplements are we talking about? These six are always on sale at the pharmacy: fish oil, cinnamon, garlic, turmeric, plant sterols, and red yeast rice.
In a study presented at the recent American Heart Association scientific sessions, researchers compared these supplements’ effectiveness in lowering LDL cholesterol with low-dose rosuvastatin or placebo among 199 adults aged 40-75 years who didn’t have a personal history of cardiovascular disease.
Participants who took the statin for 28 days had an average of 24% decrease in total cholesterol and a 38% reduction in LDL cholesterol, while 28 days’ worth of the supplements did no better than the placebo in either measure. Compared with placebo, the plant sterols supplement notably lowered HDL cholesterol and the garlic supplement notably increased LDL cholesterol.
Even though there are other studies showing the validity of plant sterols and red yeast rice to lower LDL cholesterol, author Luke J. Laffin, MD, of the Cleveland Clinic noted that this study shows how supplement results can vary and that more research is needed to see the effect they truly have on cholesterol over time.
So, should you stop taking or recommending supplements for heart health or healthy cholesterol levels? Well, we’re not going to come to your house and raid your medicine cabinet, but the authors of this study are definitely not saying that you should rely on them.
Consider this myth mostly busted.
COVID dept. of unintended consequences, part 2
The surveillance testing programs conducted in the first year of the pandemic were, in theory, meant to keep everyone safer. Someone, apparently, forgot to explain that to the students of the University of Wyoming and the University of Idaho.
We’re all familiar with the drill: Students at the two schools had to undergo frequent COVID screening to keep the virus from spreading, thereby making everyone safer. Duck your head now, because here comes the unintended consequence.
The students who didn’t get COVID eventually, and perhaps not so surprisingly, “perceived that the mandatory testing policy decreased their risk of contracting COVID-19, and … this perception led to higher participation in COVID-risky events,” Chian Jones Ritten, PhD, and associates said in PNAS Nexus.
They surveyed 757 students from the Univ. of Washington and 517 from the Univ. of Idaho and found that those who were tested more frequently perceived that they were less likely to contract the virus. Those respondents also more frequently attended indoor gatherings, both small and large, and spent more time in restaurants and bars.
The investigators did not mince words: “From a public health standpoint, such behavior is problematic.”
Current parents/participants in the workforce might have other ideas about an appropriate response to COVID.
At this point, we probably should mention that appropriation is the second-most sincere form of flattery.
Who watches the ED staff?
We heard a really great joke recently, one we simply have to share.
A man in Seattle went to a therapist. “I’m depressed,” he says. “Depressed, overworked, and lonely.”
“Oh dear, that sounds quite serious,” the therapist replies. “Tell me all about it.”
“Life just seems so harsh and cruel,” the man explains. “The pandemic has caused 300,000 health care workers across the country to leave the industry.”
“Such as the doctor typically filling this role in the joke,” the therapist, who is not licensed to prescribe medicine, nods.
“Exactly! And with so many respiratory viruses circulating and COVID still hanging around, emergency departments all over the country are facing massive backups. People are waiting outside the hospital for hours, hoping a bed will open up. Things got so bad at a hospital near Seattle in October that a nurse called 911 on her own ED. Told the 911 operator to send the fire department to help out, since they were ‘drowning’ and ‘in dire straits.’ They had 45 patients waiting and only five nurses to take care of them.”
“That is quite serious,” the therapist says, scribbling down unseen notes.
“The fire chief did send a crew out, and they cleaned rooms, changed beds, and took vitals for 90 minutes until the crisis passed,” the man says. “But it’s only a matter of time before it happens again. The hospital president said they have 300 open positions, and literally no one has applied to work in the emergency department. Not one person.”
“And how does all this make you feel?” the therapist asks.
“I feel all alone,” the man says. “This world feels so threatening, like no one cares, and I have no idea what will come next. It’s so vague and uncertain.”
“Ah, I think I have a solution for you,” the therapist says. “Go to the emergency department at St. Michael Medical Center in Silverdale, near Seattle. They’ll get your bad mood all settled, and they’ll prescribe you the medicine you need to relax.”
The man bursts into tears. “You don’t understand,” he says. “I am the emergency department at St. Michael Medical Center.”
Good joke. Everybody laugh. Roll on snare drum. Curtains.
Myth buster: Supplements for cholesterol lowering
When it comes to that nasty low-density lipoprotein cholesterol, some people swear by supplements over statins as a holistic approach. Well, we’re busting the myth that those heart-healthy supplements are even effective in comparison.
Which supplements are we talking about? These six are always on sale at the pharmacy: fish oil, cinnamon, garlic, turmeric, plant sterols, and red yeast rice.
In a study presented at the recent American Heart Association scientific sessions, researchers compared these supplements’ effectiveness in lowering LDL cholesterol with low-dose rosuvastatin or placebo among 199 adults aged 40-75 years who didn’t have a personal history of cardiovascular disease.
Participants who took the statin for 28 days had an average of 24% decrease in total cholesterol and a 38% reduction in LDL cholesterol, while 28 days’ worth of the supplements did no better than the placebo in either measure. Compared with placebo, the plant sterols supplement notably lowered HDL cholesterol and the garlic supplement notably increased LDL cholesterol.
Even though there are other studies showing the validity of plant sterols and red yeast rice to lower LDL cholesterol, author Luke J. Laffin, MD, of the Cleveland Clinic noted that this study shows how supplement results can vary and that more research is needed to see the effect they truly have on cholesterol over time.
So, should you stop taking or recommending supplements for heart health or healthy cholesterol levels? Well, we’re not going to come to your house and raid your medicine cabinet, but the authors of this study are definitely not saying that you should rely on them.
Consider this myth mostly busted.
COVID dept. of unintended consequences, part 2
The surveillance testing programs conducted in the first year of the pandemic were, in theory, meant to keep everyone safer. Someone, apparently, forgot to explain that to the students of the University of Wyoming and the University of Idaho.
We’re all familiar with the drill: Students at the two schools had to undergo frequent COVID screening to keep the virus from spreading, thereby making everyone safer. Duck your head now, because here comes the unintended consequence.
The students who didn’t get COVID eventually, and perhaps not so surprisingly, “perceived that the mandatory testing policy decreased their risk of contracting COVID-19, and … this perception led to higher participation in COVID-risky events,” Chian Jones Ritten, PhD, and associates said in PNAS Nexus.
They surveyed 757 students from the Univ. of Washington and 517 from the Univ. of Idaho and found that those who were tested more frequently perceived that they were less likely to contract the virus. Those respondents also more frequently attended indoor gatherings, both small and large, and spent more time in restaurants and bars.
The investigators did not mince words: “From a public health standpoint, such behavior is problematic.”
Current parents/participants in the workforce might have other ideas about an appropriate response to COVID.
At this point, we probably should mention that appropriation is the second-most sincere form of flattery.
Who watches the ED staff?
We heard a really great joke recently, one we simply have to share.
A man in Seattle went to a therapist. “I’m depressed,” he says. “Depressed, overworked, and lonely.”
“Oh dear, that sounds quite serious,” the therapist replies. “Tell me all about it.”
“Life just seems so harsh and cruel,” the man explains. “The pandemic has caused 300,000 health care workers across the country to leave the industry.”
“Such as the doctor typically filling this role in the joke,” the therapist, who is not licensed to prescribe medicine, nods.
“Exactly! And with so many respiratory viruses circulating and COVID still hanging around, emergency departments all over the country are facing massive backups. People are waiting outside the hospital for hours, hoping a bed will open up. Things got so bad at a hospital near Seattle in October that a nurse called 911 on her own ED. Told the 911 operator to send the fire department to help out, since they were ‘drowning’ and ‘in dire straits.’ They had 45 patients waiting and only five nurses to take care of them.”
“That is quite serious,” the therapist says, scribbling down unseen notes.
“The fire chief did send a crew out, and they cleaned rooms, changed beds, and took vitals for 90 minutes until the crisis passed,” the man says. “But it’s only a matter of time before it happens again. The hospital president said they have 300 open positions, and literally no one has applied to work in the emergency department. Not one person.”
“And how does all this make you feel?” the therapist asks.
“I feel all alone,” the man says. “This world feels so threatening, like no one cares, and I have no idea what will come next. It’s so vague and uncertain.”
“Ah, I think I have a solution for you,” the therapist says. “Go to the emergency department at St. Michael Medical Center in Silverdale, near Seattle. They’ll get your bad mood all settled, and they’ll prescribe you the medicine you need to relax.”
The man bursts into tears. “You don’t understand,” he says. “I am the emergency department at St. Michael Medical Center.”
Good joke. Everybody laugh. Roll on snare drum. Curtains.
Myth buster: Supplements for cholesterol lowering
When it comes to that nasty low-density lipoprotein cholesterol, some people swear by supplements over statins as a holistic approach. Well, we’re busting the myth that those heart-healthy supplements are even effective in comparison.
Which supplements are we talking about? These six are always on sale at the pharmacy: fish oil, cinnamon, garlic, turmeric, plant sterols, and red yeast rice.
In a study presented at the recent American Heart Association scientific sessions, researchers compared these supplements’ effectiveness in lowering LDL cholesterol with low-dose rosuvastatin or placebo among 199 adults aged 40-75 years who didn’t have a personal history of cardiovascular disease.
Participants who took the statin for 28 days had an average of 24% decrease in total cholesterol and a 38% reduction in LDL cholesterol, while 28 days’ worth of the supplements did no better than the placebo in either measure. Compared with placebo, the plant sterols supplement notably lowered HDL cholesterol and the garlic supplement notably increased LDL cholesterol.
Even though there are other studies showing the validity of plant sterols and red yeast rice to lower LDL cholesterol, author Luke J. Laffin, MD, of the Cleveland Clinic noted that this study shows how supplement results can vary and that more research is needed to see the effect they truly have on cholesterol over time.
So, should you stop taking or recommending supplements for heart health or healthy cholesterol levels? Well, we’re not going to come to your house and raid your medicine cabinet, but the authors of this study are definitely not saying that you should rely on them.
Consider this myth mostly busted.
COVID dept. of unintended consequences, part 2
The surveillance testing programs conducted in the first year of the pandemic were, in theory, meant to keep everyone safer. Someone, apparently, forgot to explain that to the students of the University of Wyoming and the University of Idaho.
We’re all familiar with the drill: Students at the two schools had to undergo frequent COVID screening to keep the virus from spreading, thereby making everyone safer. Duck your head now, because here comes the unintended consequence.
The students who didn’t get COVID eventually, and perhaps not so surprisingly, “perceived that the mandatory testing policy decreased their risk of contracting COVID-19, and … this perception led to higher participation in COVID-risky events,” Chian Jones Ritten, PhD, and associates said in PNAS Nexus.
They surveyed 757 students from the Univ. of Washington and 517 from the Univ. of Idaho and found that those who were tested more frequently perceived that they were less likely to contract the virus. Those respondents also more frequently attended indoor gatherings, both small and large, and spent more time in restaurants and bars.
The investigators did not mince words: “From a public health standpoint, such behavior is problematic.”
Current parents/participants in the workforce might have other ideas about an appropriate response to COVID.
At this point, we probably should mention that appropriation is the second-most sincere form of flattery.
Traffic-related pollutant tied to increased dementia risk
Exposure to a traffic-related air pollutant significantly increases risk for dementia, new research suggests. Results from a meta-analysis, which included a total of more than 90 million people, showed
Particulate matter is a mixture of solid particles and liquid droplets from the burning of fossil fuels and nitrogen oxide, and also produced from road traffic exhaust.
While the research only showed an association between this type of air pollution and dementia risk, the estimates were consistent across the different analyses used.
“It’s rather sobering that there is this 3% relationship between incidence of dementia and the particulate matter and that it is such a precise estimate,” senior investigator Janet Martin, PharmD, MSc, associate professor of anesthesia & perioperative medicine and epidemiology & biostatistics at Western University’s, London, Ont., told this news organization.
The findings were published online in Neurology.
Conflicting results in past studies
Air pollution is a known risk factor for dementia, but studies attempting to pinpoint its exact impact have yielded conflicting results.
Researchers analyzed data from 17 studies with a total of 91.4 million individuals, 6% of whom had dementia. In addition to PM2.5, the investigators also assessed nitrogen oxides, which form smog, nitrogen dioxide, and ozone exposure.
After adjustments for other known risk factors, such as age and gender, results showed that dementia risk increased by 3% for every 1 m3 rise in PM2.5 exposure (adjusted hazard ratio, 1.03; 95% confidence interval, 1.02-1.05).
The associations between dementia and exposure to nitrogen oxides (HR, 1.05; 95% CI, 0.99-1.13), nitrogen dioxide (HR, 1.03; 95% CI, 1.00-1.07) and ozone (HR, 1.01; 95% CI, 0.91-1.11) did not reach statistical significance. However, the confidence intervals were wide enough that clinical relevance cannot be ruled out, Dr. Martin said.
The study did not examine how or if the duration of PM2.5 exposure affected dementia risk. In addition, the investigators were not able to identify a threshold above which dementia risk begins to rise.
The Environmental Pollution Agency considers average yearly exposures up to 12 mcg/m3 to be safe. The World Health Organization sets that limit lower, at 5 mcg/m3.
Dr. Martin noted that more studies are needed to explore those issues, as well as the mechanisms by which air pollutants contribute to the pathology of dementia. However, the clear link between fine particulate matter exposure and increased risk emphasizes the need to address air pollution as a modifiable risk factor for dementia.
“The rising tide of dementia is not something we can easily reverse,” Dr. Martin said. “The evidence has been so elusive for how to treat dementia once you have it, so our biggest opportunity is to prevent it.”
Results from a study published earlier in 2022 estimated that rates of dementia will triple worldwide and double in the United States by 2050 unless steps are taking to mitigate risk factors.
Research also suggests that improving air quality PM2.5 by just 10% results in a 14% decreased risk for dementia.
‘Impressive’ pattern
Paul Rosenberg, MD, codirector of the Memory and Alzheimer’s Treatment Center division of geriatric psychiatry at Johns Hopkins University, Baltimore, said that air pollution “is the most prominent environmental risk we’ve found” for dementia. It also “adds to many other lifestyle and comorbidity risks, such as lack of exercise, obesity, depression, hearing loss, etc,” said Dr. Rosenberg, who was not involved with the research.
He noted what was “most impressive” was that in most of the pooled studies, small particulate air pollution was associated with dementia. “The overall pattern is most impressive and the effect sizes quite consistent over most of the studies,” Dr. Rosenberg said.
The meta-analysis was unfunded. Dr. Martin and Dr. Rosenberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Exposure to a traffic-related air pollutant significantly increases risk for dementia, new research suggests. Results from a meta-analysis, which included a total of more than 90 million people, showed
Particulate matter is a mixture of solid particles and liquid droplets from the burning of fossil fuels and nitrogen oxide, and also produced from road traffic exhaust.
While the research only showed an association between this type of air pollution and dementia risk, the estimates were consistent across the different analyses used.
“It’s rather sobering that there is this 3% relationship between incidence of dementia and the particulate matter and that it is such a precise estimate,” senior investigator Janet Martin, PharmD, MSc, associate professor of anesthesia & perioperative medicine and epidemiology & biostatistics at Western University’s, London, Ont., told this news organization.
The findings were published online in Neurology.
Conflicting results in past studies
Air pollution is a known risk factor for dementia, but studies attempting to pinpoint its exact impact have yielded conflicting results.
Researchers analyzed data from 17 studies with a total of 91.4 million individuals, 6% of whom had dementia. In addition to PM2.5, the investigators also assessed nitrogen oxides, which form smog, nitrogen dioxide, and ozone exposure.
After adjustments for other known risk factors, such as age and gender, results showed that dementia risk increased by 3% for every 1 m3 rise in PM2.5 exposure (adjusted hazard ratio, 1.03; 95% confidence interval, 1.02-1.05).
The associations between dementia and exposure to nitrogen oxides (HR, 1.05; 95% CI, 0.99-1.13), nitrogen dioxide (HR, 1.03; 95% CI, 1.00-1.07) and ozone (HR, 1.01; 95% CI, 0.91-1.11) did not reach statistical significance. However, the confidence intervals were wide enough that clinical relevance cannot be ruled out, Dr. Martin said.
The study did not examine how or if the duration of PM2.5 exposure affected dementia risk. In addition, the investigators were not able to identify a threshold above which dementia risk begins to rise.
The Environmental Pollution Agency considers average yearly exposures up to 12 mcg/m3 to be safe. The World Health Organization sets that limit lower, at 5 mcg/m3.
Dr. Martin noted that more studies are needed to explore those issues, as well as the mechanisms by which air pollutants contribute to the pathology of dementia. However, the clear link between fine particulate matter exposure and increased risk emphasizes the need to address air pollution as a modifiable risk factor for dementia.
“The rising tide of dementia is not something we can easily reverse,” Dr. Martin said. “The evidence has been so elusive for how to treat dementia once you have it, so our biggest opportunity is to prevent it.”
Results from a study published earlier in 2022 estimated that rates of dementia will triple worldwide and double in the United States by 2050 unless steps are taking to mitigate risk factors.
Research also suggests that improving air quality PM2.5 by just 10% results in a 14% decreased risk for dementia.
‘Impressive’ pattern
Paul Rosenberg, MD, codirector of the Memory and Alzheimer’s Treatment Center division of geriatric psychiatry at Johns Hopkins University, Baltimore, said that air pollution “is the most prominent environmental risk we’ve found” for dementia. It also “adds to many other lifestyle and comorbidity risks, such as lack of exercise, obesity, depression, hearing loss, etc,” said Dr. Rosenberg, who was not involved with the research.
He noted what was “most impressive” was that in most of the pooled studies, small particulate air pollution was associated with dementia. “The overall pattern is most impressive and the effect sizes quite consistent over most of the studies,” Dr. Rosenberg said.
The meta-analysis was unfunded. Dr. Martin and Dr. Rosenberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Exposure to a traffic-related air pollutant significantly increases risk for dementia, new research suggests. Results from a meta-analysis, which included a total of more than 90 million people, showed
Particulate matter is a mixture of solid particles and liquid droplets from the burning of fossil fuels and nitrogen oxide, and also produced from road traffic exhaust.
While the research only showed an association between this type of air pollution and dementia risk, the estimates were consistent across the different analyses used.
“It’s rather sobering that there is this 3% relationship between incidence of dementia and the particulate matter and that it is such a precise estimate,” senior investigator Janet Martin, PharmD, MSc, associate professor of anesthesia & perioperative medicine and epidemiology & biostatistics at Western University’s, London, Ont., told this news organization.
The findings were published online in Neurology.
Conflicting results in past studies
Air pollution is a known risk factor for dementia, but studies attempting to pinpoint its exact impact have yielded conflicting results.
Researchers analyzed data from 17 studies with a total of 91.4 million individuals, 6% of whom had dementia. In addition to PM2.5, the investigators also assessed nitrogen oxides, which form smog, nitrogen dioxide, and ozone exposure.
After adjustments for other known risk factors, such as age and gender, results showed that dementia risk increased by 3% for every 1 m3 rise in PM2.5 exposure (adjusted hazard ratio, 1.03; 95% confidence interval, 1.02-1.05).
The associations between dementia and exposure to nitrogen oxides (HR, 1.05; 95% CI, 0.99-1.13), nitrogen dioxide (HR, 1.03; 95% CI, 1.00-1.07) and ozone (HR, 1.01; 95% CI, 0.91-1.11) did not reach statistical significance. However, the confidence intervals were wide enough that clinical relevance cannot be ruled out, Dr. Martin said.
The study did not examine how or if the duration of PM2.5 exposure affected dementia risk. In addition, the investigators were not able to identify a threshold above which dementia risk begins to rise.
The Environmental Pollution Agency considers average yearly exposures up to 12 mcg/m3 to be safe. The World Health Organization sets that limit lower, at 5 mcg/m3.
Dr. Martin noted that more studies are needed to explore those issues, as well as the mechanisms by which air pollutants contribute to the pathology of dementia. However, the clear link between fine particulate matter exposure and increased risk emphasizes the need to address air pollution as a modifiable risk factor for dementia.
“The rising tide of dementia is not something we can easily reverse,” Dr. Martin said. “The evidence has been so elusive for how to treat dementia once you have it, so our biggest opportunity is to prevent it.”
Results from a study published earlier in 2022 estimated that rates of dementia will triple worldwide and double in the United States by 2050 unless steps are taking to mitigate risk factors.
Research also suggests that improving air quality PM2.5 by just 10% results in a 14% decreased risk for dementia.
‘Impressive’ pattern
Paul Rosenberg, MD, codirector of the Memory and Alzheimer’s Treatment Center division of geriatric psychiatry at Johns Hopkins University, Baltimore, said that air pollution “is the most prominent environmental risk we’ve found” for dementia. It also “adds to many other lifestyle and comorbidity risks, such as lack of exercise, obesity, depression, hearing loss, etc,” said Dr. Rosenberg, who was not involved with the research.
He noted what was “most impressive” was that in most of the pooled studies, small particulate air pollution was associated with dementia. “The overall pattern is most impressive and the effect sizes quite consistent over most of the studies,” Dr. Rosenberg said.
The meta-analysis was unfunded. Dr. Martin and Dr. Rosenberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY