User login
Most transgender teens not willing to delay hormone therapy to preserve fertility
TORONTO – The majority of transgender youth attending a pediatric gender clinic were not willing to delay starting hormone therapy in order to pursue fertility preservation, according to a survey study presented during a poster session at the Pediatric Academic Societies annual meeting.
Five percent of 66 young people and 33% of 52 parents surveyed during a visit to a hospital-based gender clinic agreed with the statement: “I would choose to delay hormone therapy to undergo fertility preservation (for my child) if asked today.”
Further, 70% of youth agreed that discomfort with a part of the body they don’t identify with was a factor that influenced their decision or thoughts about fertility preservation. Religious, financial, ethical, and demographic factors were not associated with willingness to delay treatment for fertility concerns.
“While hormone therapy has drastically improved the lives of countless transgender and gender nonconforming youth, its impact on fertility can unfairly force individuals to decide at a very early age whether or not they should preserve the ability to be a biological parent one day,” Rebecca Persky, MD, said in a press release. Dr. Persky, a former Children’s Hospital of Philadelphia (CHOP) resident, is now is a pediatric endocrinology fellow at the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
A slightly greater proportion of youth (20%) and 12% of parents agreed it was important to have biological children or grandchildren. For those youth who did want to preserve the option of having biological children, that desire was associated with perceiving it as important to their parents (odds ratio, 6.07; P less than .05).
“We didn’t ask any questions about adoption of children or grandchildren, so that might have yielded different results if we had,” Dr. Persky acknowledged in an interview.
A lack of information about whether hormone therapy definitely prevents biologic fertility was associated with parents’ willingness to delay treatment for fertility preservation (OR: 24.57, P less than .05), yet 62% of parents said they felt their children were able to “make a meaningful decision about taking steps to preserve fertility at this point in (his/her/their) life.”
“I thought delaying treatment would be one of the biggest barriers, but even when we asked them if they wanted to preserve their fertility while not delaying or changing their hormone therapy, only the minority [33%] said they would be interested in that,” Dr. Persky said in an interview. “It kind of argues that a lot of these kids just don’t want to have biological children.”
She noted, however, that one limitation of the study was that many of the children surveyed already were receiving hormone therapy such that the questions engaged more on a theoretical level than a practical one.
“Not surprisingly, the strongest factor in the parents’ decisions was whether or not it was important to their child to have biological children,” said Dr. Persky.
The researchers surveyed 66 transgender and gender nonconforming youth who presented for care at the Gender and Sexuality Development Clinic at CHOP. After the findings were released, it was noted by several concerned parties on Twitter that because of the location of the study, the sample was a decidedly selected one.
The mean age was 17 years of patients and 63% of the sample were assigned female sex at birth. The mean age in the 52 parents surveyed was 48 years. The survey included 36 items on knowledge of fertility preservation, the desire to have biological children, and other factors that may affect the decision to pursue fertility preservation.
Gender-specific and age-specific analyses have not been completed, but are in the works, said Dr. Persky, who acknowledged that the area requires more qualitative research.
The authors reported no conflicts of interest.
TORONTO – The majority of transgender youth attending a pediatric gender clinic were not willing to delay starting hormone therapy in order to pursue fertility preservation, according to a survey study presented during a poster session at the Pediatric Academic Societies annual meeting.
Five percent of 66 young people and 33% of 52 parents surveyed during a visit to a hospital-based gender clinic agreed with the statement: “I would choose to delay hormone therapy to undergo fertility preservation (for my child) if asked today.”
Further, 70% of youth agreed that discomfort with a part of the body they don’t identify with was a factor that influenced their decision or thoughts about fertility preservation. Religious, financial, ethical, and demographic factors were not associated with willingness to delay treatment for fertility concerns.
“While hormone therapy has drastically improved the lives of countless transgender and gender nonconforming youth, its impact on fertility can unfairly force individuals to decide at a very early age whether or not they should preserve the ability to be a biological parent one day,” Rebecca Persky, MD, said in a press release. Dr. Persky, a former Children’s Hospital of Philadelphia (CHOP) resident, is now is a pediatric endocrinology fellow at the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
A slightly greater proportion of youth (20%) and 12% of parents agreed it was important to have biological children or grandchildren. For those youth who did want to preserve the option of having biological children, that desire was associated with perceiving it as important to their parents (odds ratio, 6.07; P less than .05).
“We didn’t ask any questions about adoption of children or grandchildren, so that might have yielded different results if we had,” Dr. Persky acknowledged in an interview.
A lack of information about whether hormone therapy definitely prevents biologic fertility was associated with parents’ willingness to delay treatment for fertility preservation (OR: 24.57, P less than .05), yet 62% of parents said they felt their children were able to “make a meaningful decision about taking steps to preserve fertility at this point in (his/her/their) life.”
“I thought delaying treatment would be one of the biggest barriers, but even when we asked them if they wanted to preserve their fertility while not delaying or changing their hormone therapy, only the minority [33%] said they would be interested in that,” Dr. Persky said in an interview. “It kind of argues that a lot of these kids just don’t want to have biological children.”
She noted, however, that one limitation of the study was that many of the children surveyed already were receiving hormone therapy such that the questions engaged more on a theoretical level than a practical one.
“Not surprisingly, the strongest factor in the parents’ decisions was whether or not it was important to their child to have biological children,” said Dr. Persky.
The researchers surveyed 66 transgender and gender nonconforming youth who presented for care at the Gender and Sexuality Development Clinic at CHOP. After the findings were released, it was noted by several concerned parties on Twitter that because of the location of the study, the sample was a decidedly selected one.
The mean age was 17 years of patients and 63% of the sample were assigned female sex at birth. The mean age in the 52 parents surveyed was 48 years. The survey included 36 items on knowledge of fertility preservation, the desire to have biological children, and other factors that may affect the decision to pursue fertility preservation.
Gender-specific and age-specific analyses have not been completed, but are in the works, said Dr. Persky, who acknowledged that the area requires more qualitative research.
The authors reported no conflicts of interest.
TORONTO – The majority of transgender youth attending a pediatric gender clinic were not willing to delay starting hormone therapy in order to pursue fertility preservation, according to a survey study presented during a poster session at the Pediatric Academic Societies annual meeting.
Five percent of 66 young people and 33% of 52 parents surveyed during a visit to a hospital-based gender clinic agreed with the statement: “I would choose to delay hormone therapy to undergo fertility preservation (for my child) if asked today.”
Further, 70% of youth agreed that discomfort with a part of the body they don’t identify with was a factor that influenced their decision or thoughts about fertility preservation. Religious, financial, ethical, and demographic factors were not associated with willingness to delay treatment for fertility concerns.
“While hormone therapy has drastically improved the lives of countless transgender and gender nonconforming youth, its impact on fertility can unfairly force individuals to decide at a very early age whether or not they should preserve the ability to be a biological parent one day,” Rebecca Persky, MD, said in a press release. Dr. Persky, a former Children’s Hospital of Philadelphia (CHOP) resident, is now is a pediatric endocrinology fellow at the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
A slightly greater proportion of youth (20%) and 12% of parents agreed it was important to have biological children or grandchildren. For those youth who did want to preserve the option of having biological children, that desire was associated with perceiving it as important to their parents (odds ratio, 6.07; P less than .05).
“We didn’t ask any questions about adoption of children or grandchildren, so that might have yielded different results if we had,” Dr. Persky acknowledged in an interview.
A lack of information about whether hormone therapy definitely prevents biologic fertility was associated with parents’ willingness to delay treatment for fertility preservation (OR: 24.57, P less than .05), yet 62% of parents said they felt their children were able to “make a meaningful decision about taking steps to preserve fertility at this point in (his/her/their) life.”
“I thought delaying treatment would be one of the biggest barriers, but even when we asked them if they wanted to preserve their fertility while not delaying or changing their hormone therapy, only the minority [33%] said they would be interested in that,” Dr. Persky said in an interview. “It kind of argues that a lot of these kids just don’t want to have biological children.”
She noted, however, that one limitation of the study was that many of the children surveyed already were receiving hormone therapy such that the questions engaged more on a theoretical level than a practical one.
“Not surprisingly, the strongest factor in the parents’ decisions was whether or not it was important to their child to have biological children,” said Dr. Persky.
The researchers surveyed 66 transgender and gender nonconforming youth who presented for care at the Gender and Sexuality Development Clinic at CHOP. After the findings were released, it was noted by several concerned parties on Twitter that because of the location of the study, the sample was a decidedly selected one.
The mean age was 17 years of patients and 63% of the sample were assigned female sex at birth. The mean age in the 52 parents surveyed was 48 years. The survey included 36 items on knowledge of fertility preservation, the desire to have biological children, and other factors that may affect the decision to pursue fertility preservation.
Gender-specific and age-specific analyses have not been completed, but are in the works, said Dr. Persky, who acknowledged that the area requires more qualitative research.
The authors reported no conflicts of interest.
REPORTING FROM PAS 18
Key clinical point:
Major finding: Five percent of youth and 33% of parents were willing to delay hormone therapy to undergo fertility preservation.
Study details: Survey study of 66 young people attending a pediatric gender and sexuality clinic and 52 parents of those individuals.
Disclosures: The authors reported no conflicts of interest.
More testing of febrile infants at teaching vs. community hospitals, but similar outcomes
TORONTO – according to a study presented at the Pediatric Academic Societies annual meeting.
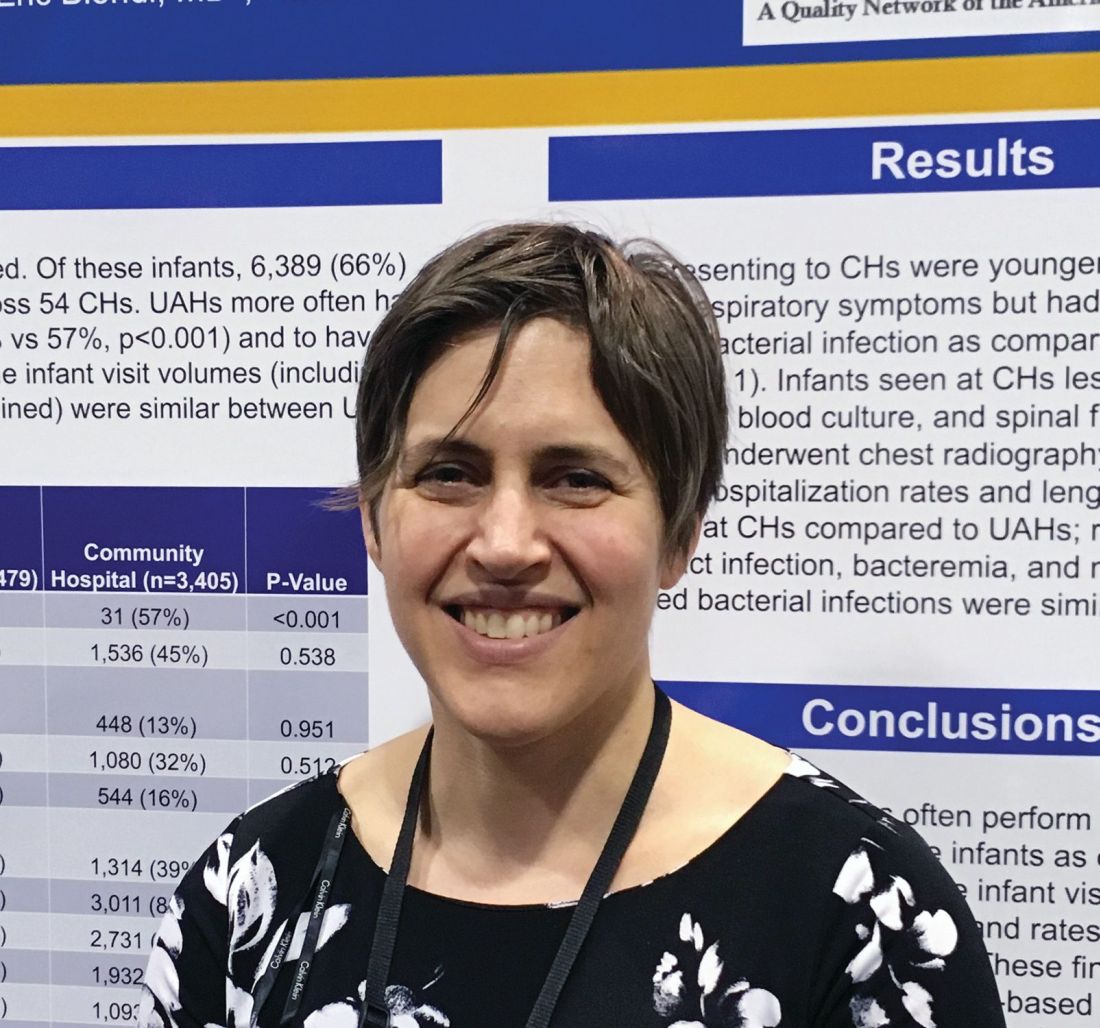
“The community hospitals are doing less procedures on the infants, but with basically the exact same outcomes,” said Beth C. Natt, MD, MPH, director of pediatric hospital medicine at Bridgeport (Conn.) Hospital.
Babies who presented to university-affiliated hospitals were more likely to be hospitalized (70% vs. 67%; P = .001) than were those at community hospitals, but had a similar likelihood of being diagnosed with bacteremia, meningitis, or urinary tract infection. The rates of missed bacterial infection were 0.8% for teaching hospitals and 1% for community hospitals (P = .346).
“There is some thought that in community settings, because we’re not completing the workup in the standard, protocolized way seen at teaching hospitals, we might be doing wrong by the children, but these data show we’re actually doing just fine,” Dr. Natt said in an interview.
She and her colleagues reviewed 9,884 febrile infant evaluations occurring at 132 hospitals participating in the Reducing Excessive Variation in the Infant Sepsis Evaluation (REVISE) quality improvement project. Two-thirds of the infants (n = 6,479) were evaluated across 78 university-affiliated hospitals and 3,405 (or 34%) were seen at 54 community hospitals. Hospital status was self-reported.
The teaching hospitals more often had at least one pediatric emergency medicine provider, compared with community hospitals (90% vs. 57%; P = .001) and were more likely to see babies between 7 and 30 days old (90% vs. 57%; P = .001). They also were more likely to obtain urine cultures (92% vs. 88%; P = 0.001), blood cultures (84% vs. 80%; P = .001), and cerebral spinal fluid cultures (62% vs. 57%; P = .001).
On the other hand, community hospitals were significantly more likely to see children presenting with respiratory symptoms (39% vs. 36% for teaching hospitals; P = .014), and were more likely to order chest x-rays on febrile infants (32% vs. 24% for university-affiliated hospitals; P = .001).
“As a community hospitalist, the results weren’t that surprising to me,” said Dr. Natt. “If anything was surprising it was how often we were doing chest x-rays, but I think that had to do with the fact that we had more children with respiratory symptoms coming to community hospitals.
“The American Academy of Pediatrics guidelines for fever were written last in 1993, when I was in high school, so they are very due to be revised,” said Dr. Natt. “I suspect the new guidelines will have us doing fewer spinal taps in children and more watchful waiting.”
TORONTO – according to a study presented at the Pediatric Academic Societies annual meeting.
“The community hospitals are doing less procedures on the infants, but with basically the exact same outcomes,” said Beth C. Natt, MD, MPH, director of pediatric hospital medicine at Bridgeport (Conn.) Hospital.
Babies who presented to university-affiliated hospitals were more likely to be hospitalized (70% vs. 67%; P = .001) than were those at community hospitals, but had a similar likelihood of being diagnosed with bacteremia, meningitis, or urinary tract infection. The rates of missed bacterial infection were 0.8% for teaching hospitals and 1% for community hospitals (P = .346).
“There is some thought that in community settings, because we’re not completing the workup in the standard, protocolized way seen at teaching hospitals, we might be doing wrong by the children, but these data show we’re actually doing just fine,” Dr. Natt said in an interview.
She and her colleagues reviewed 9,884 febrile infant evaluations occurring at 132 hospitals participating in the Reducing Excessive Variation in the Infant Sepsis Evaluation (REVISE) quality improvement project. Two-thirds of the infants (n = 6,479) were evaluated across 78 university-affiliated hospitals and 3,405 (or 34%) were seen at 54 community hospitals. Hospital status was self-reported.
The teaching hospitals more often had at least one pediatric emergency medicine provider, compared with community hospitals (90% vs. 57%; P = .001) and were more likely to see babies between 7 and 30 days old (90% vs. 57%; P = .001). They also were more likely to obtain urine cultures (92% vs. 88%; P = 0.001), blood cultures (84% vs. 80%; P = .001), and cerebral spinal fluid cultures (62% vs. 57%; P = .001).
On the other hand, community hospitals were significantly more likely to see children presenting with respiratory symptoms (39% vs. 36% for teaching hospitals; P = .014), and were more likely to order chest x-rays on febrile infants (32% vs. 24% for university-affiliated hospitals; P = .001).
“As a community hospitalist, the results weren’t that surprising to me,” said Dr. Natt. “If anything was surprising it was how often we were doing chest x-rays, but I think that had to do with the fact that we had more children with respiratory symptoms coming to community hospitals.
“The American Academy of Pediatrics guidelines for fever were written last in 1993, when I was in high school, so they are very due to be revised,” said Dr. Natt. “I suspect the new guidelines will have us doing fewer spinal taps in children and more watchful waiting.”
TORONTO – according to a study presented at the Pediatric Academic Societies annual meeting.
“The community hospitals are doing less procedures on the infants, but with basically the exact same outcomes,” said Beth C. Natt, MD, MPH, director of pediatric hospital medicine at Bridgeport (Conn.) Hospital.
Babies who presented to university-affiliated hospitals were more likely to be hospitalized (70% vs. 67%; P = .001) than were those at community hospitals, but had a similar likelihood of being diagnosed with bacteremia, meningitis, or urinary tract infection. The rates of missed bacterial infection were 0.8% for teaching hospitals and 1% for community hospitals (P = .346).
“There is some thought that in community settings, because we’re not completing the workup in the standard, protocolized way seen at teaching hospitals, we might be doing wrong by the children, but these data show we’re actually doing just fine,” Dr. Natt said in an interview.
She and her colleagues reviewed 9,884 febrile infant evaluations occurring at 132 hospitals participating in the Reducing Excessive Variation in the Infant Sepsis Evaluation (REVISE) quality improvement project. Two-thirds of the infants (n = 6,479) were evaluated across 78 university-affiliated hospitals and 3,405 (or 34%) were seen at 54 community hospitals. Hospital status was self-reported.
The teaching hospitals more often had at least one pediatric emergency medicine provider, compared with community hospitals (90% vs. 57%; P = .001) and were more likely to see babies between 7 and 30 days old (90% vs. 57%; P = .001). They also were more likely to obtain urine cultures (92% vs. 88%; P = 0.001), blood cultures (84% vs. 80%; P = .001), and cerebral spinal fluid cultures (62% vs. 57%; P = .001).
On the other hand, community hospitals were significantly more likely to see children presenting with respiratory symptoms (39% vs. 36% for teaching hospitals; P = .014), and were more likely to order chest x-rays on febrile infants (32% vs. 24% for university-affiliated hospitals; P = .001).
“As a community hospitalist, the results weren’t that surprising to me,” said Dr. Natt. “If anything was surprising it was how often we were doing chest x-rays, but I think that had to do with the fact that we had more children with respiratory symptoms coming to community hospitals.
“The American Academy of Pediatrics guidelines for fever were written last in 1993, when I was in high school, so they are very due to be revised,” said Dr. Natt. “I suspect the new guidelines will have us doing fewer spinal taps in children and more watchful waiting.”
AT PAS 18
Key clinical point: University-affiliated hospitals do more invasive testing in febrile infants, but have outcomes similar to those of community hospitals.
Major finding: The rate of missed bacterial infection did not differ between hospital types: 0.8% for teaching hospitals and 1% for community hospitals (P = .346).
Study details: Review of 9,884 febrile infant evaluations occurring at 132 hospitals, 66% of which were university-affiliated hospitals and 34% of which were community hospitals.
Disclosures: The investigators reported no conflicts of interest.
Pediatric inpatient seizures treated quickly with new intervention
TORONTO – Researchers at UCSF Benioff Children’s Hospital in San Francisco implemented a novel intervention that leveraged existing in-room technology to expedite antiepileptic drug administration to inpatients having a seizure.
With the quality initiative, they were able to decrease median time from seizure onset to benzodiazepine (BZD) administration from 7 minutes (preintervention) to 2 minutes (post intervention) and reduce the median time from order to administration of second-phase non-BZDs from 28 minutes to 11 minutes.
“Leveraging existing patient room technology to mobilize pharmacy to the bedside expedited non-BZD administration by 60%,” reported principal investigator Arpi Bekmezian, MD, a pediatric hospitalist and medical director of quality and safety at Benioff Children’s Hospital. She presented the findings at the Pediatric Academic Societies annual meeting.
“Furthermore, the rapid-response seizure rescue process may have created an increased sense of urgency helping to expedite initial BZD administration by 70%. ... This may have prevented the need for second-phase therapy and progression to status epilepticus, potentially minimizing the risk of neuronal injury, and all without the additional resources of a Code team.”
Early and rapid escalation of treatment is critical to prevent neuronal injury in patients with status epilepticus. Guidelines recommend initial antiepileptic therapy at 5 minutes, with rapid escalation to second-phase therapy if the seizure persists.
Preintervention baseline data from UCSF Benioff Children’s indicated a 7-minute lag time from seizure onset to BZD therapy and a 28-minute lag from order to administration of non-BZDs (phenobarbital, phenytoin, levetiracetam, valproic acid). Other studies have shown significantly greater delays to antiepileptic treatment.
“That was just too long, and it matched our clinical experience of being at the bedside of a seizing patient and wondering why the medication was taking so long to arrive from the pharmacy.”
The researchers set out to reduce time to BZD administration from 7 minutes to 5 minutes or less and to reduce time to second-phase non-BZD administration to less than 10 minutes. To accomplish this, a multidisciplinary team that included leadership from physicians, pharmacy, and nursing defined primary and secondary drivers of efficiency, with interventions targeting both team communication and medication delivery.
The intervention period lasted 16 months, during which time there were 61 seizure events requiring urgent antiepileptic treatment. Complete data were available for 57 seizures.
Among the interventions they implemented was to stock all medication-dispensing stations with intranasal/buccal BZD available on “nursing override” for easy access and administration.
Because non-BZDs require pharmacy compounding, and the main pharmacy receives many STAT orders with competing priorities, they developed a hospitalwide “seizure rescue” (SR) process by using patient-room staff terminals to activate a dedicated individual from the pharmacy, who would then report to the bedside with a backpack stocked with non-BZDs ready to compound. Nurses were trained to press the SR button for any seizure that may require urgent therapy.
“We didn’t want nurses to waste time on the phone [calling pharmacy], and we considered calling a Code, but we couldn’t really justify the resource utilization as most of these patients didn’t have respiratory compromise, and they didn’t need the whole Code team,” said Dr. Bekmezian. She noted that her hospital strongly discourages bedside compounding by nursing staff.
Instead, they realized they could easily reprogram the patient-room electronic staff terminals to have a dedicated SR button that would directly alert a dedicated pharmacist carrying the SR phone. The pharmacist could then swipe and confirm that they received the alert and let the nurse know they were on the way, “and this would free up the nurse to go ahead and obtain the benzodiazepines and administer them as pharmacy made their way to the room.”
“To our knowledge, this is the first study to report expediting antiepileptic drug delivery to patients in the hospital,” said Dr. Bekmezian. She noted that less than 50% of cases actually required pharmacist response, “but the pharmacy staff chose to be activated earlier in the management algorithm to avoid delays in treatment.”
UCSF Children’s Hospital San Francisco campus is a 183-bed, tertiary care, teaching children’s hospital that has pediatric, neonatal, and cardiac intensive care units and set-down units. They provide liver, bone marrow, kidney, and cardiac transplantation and have more than 10,000 annual admissions.
The investigators reported no conflicts of interest.
SOURCE: Bekmezian A et al. PAS 2018. Abstract 3545.3.
TORONTO – Researchers at UCSF Benioff Children’s Hospital in San Francisco implemented a novel intervention that leveraged existing in-room technology to expedite antiepileptic drug administration to inpatients having a seizure.
With the quality initiative, they were able to decrease median time from seizure onset to benzodiazepine (BZD) administration from 7 minutes (preintervention) to 2 minutes (post intervention) and reduce the median time from order to administration of second-phase non-BZDs from 28 minutes to 11 minutes.
“Leveraging existing patient room technology to mobilize pharmacy to the bedside expedited non-BZD administration by 60%,” reported principal investigator Arpi Bekmezian, MD, a pediatric hospitalist and medical director of quality and safety at Benioff Children’s Hospital. She presented the findings at the Pediatric Academic Societies annual meeting.
“Furthermore, the rapid-response seizure rescue process may have created an increased sense of urgency helping to expedite initial BZD administration by 70%. ... This may have prevented the need for second-phase therapy and progression to status epilepticus, potentially minimizing the risk of neuronal injury, and all without the additional resources of a Code team.”
Early and rapid escalation of treatment is critical to prevent neuronal injury in patients with status epilepticus. Guidelines recommend initial antiepileptic therapy at 5 minutes, with rapid escalation to second-phase therapy if the seizure persists.
Preintervention baseline data from UCSF Benioff Children’s indicated a 7-minute lag time from seizure onset to BZD therapy and a 28-minute lag from order to administration of non-BZDs (phenobarbital, phenytoin, levetiracetam, valproic acid). Other studies have shown significantly greater delays to antiepileptic treatment.
“That was just too long, and it matched our clinical experience of being at the bedside of a seizing patient and wondering why the medication was taking so long to arrive from the pharmacy.”
The researchers set out to reduce time to BZD administration from 7 minutes to 5 minutes or less and to reduce time to second-phase non-BZD administration to less than 10 minutes. To accomplish this, a multidisciplinary team that included leadership from physicians, pharmacy, and nursing defined primary and secondary drivers of efficiency, with interventions targeting both team communication and medication delivery.
The intervention period lasted 16 months, during which time there were 61 seizure events requiring urgent antiepileptic treatment. Complete data were available for 57 seizures.
Among the interventions they implemented was to stock all medication-dispensing stations with intranasal/buccal BZD available on “nursing override” for easy access and administration.
Because non-BZDs require pharmacy compounding, and the main pharmacy receives many STAT orders with competing priorities, they developed a hospitalwide “seizure rescue” (SR) process by using patient-room staff terminals to activate a dedicated individual from the pharmacy, who would then report to the bedside with a backpack stocked with non-BZDs ready to compound. Nurses were trained to press the SR button for any seizure that may require urgent therapy.
“We didn’t want nurses to waste time on the phone [calling pharmacy], and we considered calling a Code, but we couldn’t really justify the resource utilization as most of these patients didn’t have respiratory compromise, and they didn’t need the whole Code team,” said Dr. Bekmezian. She noted that her hospital strongly discourages bedside compounding by nursing staff.
Instead, they realized they could easily reprogram the patient-room electronic staff terminals to have a dedicated SR button that would directly alert a dedicated pharmacist carrying the SR phone. The pharmacist could then swipe and confirm that they received the alert and let the nurse know they were on the way, “and this would free up the nurse to go ahead and obtain the benzodiazepines and administer them as pharmacy made their way to the room.”
“To our knowledge, this is the first study to report expediting antiepileptic drug delivery to patients in the hospital,” said Dr. Bekmezian. She noted that less than 50% of cases actually required pharmacist response, “but the pharmacy staff chose to be activated earlier in the management algorithm to avoid delays in treatment.”
UCSF Children’s Hospital San Francisco campus is a 183-bed, tertiary care, teaching children’s hospital that has pediatric, neonatal, and cardiac intensive care units and set-down units. They provide liver, bone marrow, kidney, and cardiac transplantation and have more than 10,000 annual admissions.
The investigators reported no conflicts of interest.
SOURCE: Bekmezian A et al. PAS 2018. Abstract 3545.3.
TORONTO – Researchers at UCSF Benioff Children’s Hospital in San Francisco implemented a novel intervention that leveraged existing in-room technology to expedite antiepileptic drug administration to inpatients having a seizure.
With the quality initiative, they were able to decrease median time from seizure onset to benzodiazepine (BZD) administration from 7 minutes (preintervention) to 2 minutes (post intervention) and reduce the median time from order to administration of second-phase non-BZDs from 28 minutes to 11 minutes.
“Leveraging existing patient room technology to mobilize pharmacy to the bedside expedited non-BZD administration by 60%,” reported principal investigator Arpi Bekmezian, MD, a pediatric hospitalist and medical director of quality and safety at Benioff Children’s Hospital. She presented the findings at the Pediatric Academic Societies annual meeting.
“Furthermore, the rapid-response seizure rescue process may have created an increased sense of urgency helping to expedite initial BZD administration by 70%. ... This may have prevented the need for second-phase therapy and progression to status epilepticus, potentially minimizing the risk of neuronal injury, and all without the additional resources of a Code team.”
Early and rapid escalation of treatment is critical to prevent neuronal injury in patients with status epilepticus. Guidelines recommend initial antiepileptic therapy at 5 minutes, with rapid escalation to second-phase therapy if the seizure persists.
Preintervention baseline data from UCSF Benioff Children’s indicated a 7-minute lag time from seizure onset to BZD therapy and a 28-minute lag from order to administration of non-BZDs (phenobarbital, phenytoin, levetiracetam, valproic acid). Other studies have shown significantly greater delays to antiepileptic treatment.
“That was just too long, and it matched our clinical experience of being at the bedside of a seizing patient and wondering why the medication was taking so long to arrive from the pharmacy.”
The researchers set out to reduce time to BZD administration from 7 minutes to 5 minutes or less and to reduce time to second-phase non-BZD administration to less than 10 minutes. To accomplish this, a multidisciplinary team that included leadership from physicians, pharmacy, and nursing defined primary and secondary drivers of efficiency, with interventions targeting both team communication and medication delivery.
The intervention period lasted 16 months, during which time there were 61 seizure events requiring urgent antiepileptic treatment. Complete data were available for 57 seizures.
Among the interventions they implemented was to stock all medication-dispensing stations with intranasal/buccal BZD available on “nursing override” for easy access and administration.
Because non-BZDs require pharmacy compounding, and the main pharmacy receives many STAT orders with competing priorities, they developed a hospitalwide “seizure rescue” (SR) process by using patient-room staff terminals to activate a dedicated individual from the pharmacy, who would then report to the bedside with a backpack stocked with non-BZDs ready to compound. Nurses were trained to press the SR button for any seizure that may require urgent therapy.
“We didn’t want nurses to waste time on the phone [calling pharmacy], and we considered calling a Code, but we couldn’t really justify the resource utilization as most of these patients didn’t have respiratory compromise, and they didn’t need the whole Code team,” said Dr. Bekmezian. She noted that her hospital strongly discourages bedside compounding by nursing staff.
Instead, they realized they could easily reprogram the patient-room electronic staff terminals to have a dedicated SR button that would directly alert a dedicated pharmacist carrying the SR phone. The pharmacist could then swipe and confirm that they received the alert and let the nurse know they were on the way, “and this would free up the nurse to go ahead and obtain the benzodiazepines and administer them as pharmacy made their way to the room.”
“To our knowledge, this is the first study to report expediting antiepileptic drug delivery to patients in the hospital,” said Dr. Bekmezian. She noted that less than 50% of cases actually required pharmacist response, “but the pharmacy staff chose to be activated earlier in the management algorithm to avoid delays in treatment.”
UCSF Children’s Hospital San Francisco campus is a 183-bed, tertiary care, teaching children’s hospital that has pediatric, neonatal, and cardiac intensive care units and set-down units. They provide liver, bone marrow, kidney, and cardiac transplantation and have more than 10,000 annual admissions.
The investigators reported no conflicts of interest.
SOURCE: Bekmezian A et al. PAS 2018. Abstract 3545.3.
AT PAS 2018
Key clinical point: An intervention to speed delivery of antiepileptic drugs significantly reduced time to treatment.
Major finding: Median time from seizure onset to benzodiazepine (BZD) administration fell from 7 minutes preintervention to 2 minutes post intervention, and median time from order to administration of non-BZDs dropped from 28 minutes to 11 minutes.
Study details: A prospective, multicenter study of 57 seizure events during a 16-month period.
Disclosures: The investigators reported no conflicts of interest.
Source: Bekmezian A et al. PAS 2018. Abstract 3545.3.
Study spotlights risk factors for albuminuria in youth with T2DM
TORONTO – When Brandy Wicklow, MD, began her pediatric endocrinology fellowship at McGill University in 2006, about 12 per 100,000 children in Manitoba, Canada, were diagnosed with type 2 diabetes mellitus each year. By 2016 that rate had more than doubled, to 26 per 100,000 children.
“If you look just at indigenous youth in our province, it’s probably one of the highest rates ever reported, with 95 per 100,000 Manitoba First Nation children diagnosed with type 2 diabetes,” said Dr. Wicklow, a pediatric endocrinologist at the University of Manitoba and the Children’s Hospital Research Institute of Manitoba.
Many indigenous populations also face an increased risk for primary renal disease. One study reviewed the charts 90 of Canadian First Nation children and adolescents with T2DM (Diabetes Care. 2009;32[5]:786-90). Of 10 who had renal biopsies performed, nine had immune complex disease/glomerulosclerosis, two had mild diabetes-related lesions, and seven had focal segmental glomerulosclerosis (FSGS); yet none had classic nephropathy. An analysis of Chinese youth that included 216 renal biopsies yielded similar findings (Intl Urol Nephrol. 2012;45[1]:173-9).
It’s also known that early-onset T2DM is associated with substantially increased incidence of end-stage renal disease (ESRD) and mortality in middle age. For example, one study of Pima Indians found that those who were diagnosed with T2DM earlier than 20 years of age had a one in five chance of developing ESRD, while those who were diagnosed at age 20 years or older had a one in two chance of ESRD (JAMA. 2006;296[4]:421-6). In a separate analysis, researchers estimated the remaining lifetime risks for ESRD among Aboriginal people in Australia with and without diabetes (Diabetes Res Clin Pract. 2014;103[3]:e24-6). The value for young adults with diabetes was high, about one in two at the age of 30 years, while it decreased with age to one in seven at 60 years.
“One of the first biomarkers we see in terms of renal disease in kids with T2DM is albuminuria,” Dr. Wicklow said at the Pediatric Academic Societies meeting. “The question is, why do kids with type 2 get more renal disease than kids with type 1 diabetes?” The SEARCH for Diabetes in Youth (SEARCH) study from 2006 found that hypertension, increased body mass index, increased weight circumference, and increased lipids were factors, while the SEARCH study from 2015 found that ethnicity, increased weight to height ratio, and mean arterial pressure were factors.
“Insulin resistance is significantly associated with albuminuria,” Dr. Wicklow continued. “It’s also been shown to be associated with hyperfiltration. Some of the markers of insulin resistance are important but they make up about 19% of the variance between type 1 and type 2, which means there are other variables that we’re not measuring.”
Enter ICARE (Improving Renal Complications in Adolescents with Type 2 Diabetes through Research), an ongoing prospective cohort study that Dr. Wicklow and her associates launched in 2014 at eight centers in Canada. It aims to examine the biopsychosocial risk factors for albuminuria in youth with T2DM and the mechanisms for renal injury. “Our theoretical framework was that biological exposures that we are aware of, such as glycemic control, hypertension, and lipids, would all be important in the development of albuminuria and renal disease in kids,” said Dr. Wicklow, who is the study’s coprimary investigator along with Allison Dart, MD. “But what we thought was novel was that psychological exposures either as socioeconomic status or as mental health factors would also directly impinge on renal health with respect to chronic inflammation in the body, inflammation in the kidneys, and long-term kidney damage.”
The first phase of ICARE involved a detailed phenotypic assessment of youth, including anthropometrics, biochemistry, 24-hour ambulatory blood pressure monitoring, overnight urine collections for albumin excretion, renal ultrasound, and iohexol-derived glomerular filtration rate (GFR). Phase 2 included an evaluation of psychological factors, including hair-derived cortisol; validated questionnaires for perceived stress, distress, and resiliency; and a detailed evaluation of systemic and urine inflammatory biomarkers. Annual follow-up is carried out to assess temporal associations between clinical risk factors and renal outcomes, including progression of albuminuria.
At the meeting, Dr. Wicklow reported on 187 youth enrolled to date. Of these, 96% were of indigenous ethnicity, 57 had albuminuria and 130 did not, and the mean ages of the groups were 16 years and 15 years, respectively. At baseline, a higher proportion of those in the albuminuria group were female (74% vs. 64% of those in the no albuminuria group, respectively), had a higher mean hemoglobin A1c (11% vs. 9%), and had hypertension (94% vs. 72%). She noted that upon presentation to the clinic, only 23% of participants had HbA1c levels less than 7%, only 26% had ranges between 7% and 9%, and about 40% did not have any hypertension. Of those who did, 27% had nighttime-only hypertension, and only 2% had daytime-only hypertension.
“The other risk factor these kids have for developing ESRD is that the majority were exposed to diabetes in pregnancy,” Dr. Wicklow said. “Murine models of maternal diabetes exposure have demonstrated that offspring have small kidneys, less ureteric bud branching, and a lower number of nephrons. Most of the human clinical cohort studies look at associations between development of diabetes and parental hypertension, maternal smoking, and maternal education. There is likely an impact at birth that sets these kids up for development of type 2 diabetes.”
In addition, results from clinical cohort studies have found that depression, mental stress, and distress are high in youth with T2DM. “Preliminary data suggest that if you have positive mental health, or coping strategies, or someone has worked through this with you and you are resilient, you might benefit in terms of overall glycemic control,” she said. For example, ICARE investigators have found that the higher the score on the Kessler Psychological Distress Scale (K6), the greater the risk of renal inflammation as measured by monocyte chemotactic protein-1 (MCP-1; P = .02). “Mental health seems to be something that can directly impact your health from a biological standpoint, and we might be able to find biomarkers of that risk,” Dr. Wicklow said. “Where does the stress come from? Most of my patients are indigenous, so it’s not surprising that the history in Canada of colonization of residential schools has left a lasting impression on these families and communities in terms of loss of language, loss of culture, and loss of land. There’s a community-based stress and a family-based stress that these children feel.”
Social factors also play a big role. She presented baseline findings from 196 youth with T2DM and 456 with T1DM, including measures such as the Socioeconomic Factor Index-Version 2 (SEFI-2), a way to assess socioeconomic characteristics based on Canadian Census data that reflects nonmedical social determinants of health. “It looks at factors like number of rooms in the house, single-parent households, maternal education attainment, and family income,” Dr. Wicklow explained. “The higher the SEFI-2 score, the lower your socioeconomic status is for the area you live in. Kids with T2DM generally live in areas of lower SES and lower socioeconomic index. They often live far away from health care providers. Many do not attend school and many are not with their biologic families, so we’ve had a lot of issues addressing child and family services, in particular in the phase of a chronic illness where our expectation is one thing and the family’s and community’s expectations of what’s realistic in terms of treatment and goals is another. We also have a lot of adolescent pregnancies.”
To date, about 80% of youth with T1D have seen a health care provider within the first year after transition from the pediatric diabetes clinic, compared with just over 50% of kids with T2D. “We transition youth with T1DM to internists, while our youth with T2DM go to itinerant physicians often back in their communities and/or rural family physicians,” she said. Between baseline and year 2, the rate of hospital admissions remained similar among T1DM at 11.6 and 11.8 admissions per 100 patient-years, respectively, but the number of hospital admissions for T2DM patients jumped from 20.1 to 25.5 admissions per 100 patient-years. “Kids with type 2 are showing up in the hospital a lot more than those with type 1 diabetes, but not for diabetes-related diagnoses,” Dr. Wicklow said. “We’re starting to look through the data now, and most of our kids are showing up with mental health complaints and issues. That’s why they’re getting hospitalized.”
Among ICARE study participants who have completed 3 years of follow-up, about 52% had albuminuria at their baseline visit and 48% sustained albuminuria throughout the study. About 26% progressed from normal levels of albuminuria to microalbuminuria, from microalbuminuria to macroalbuminuria, or from normal levels of albuminuria to macroalbuminuria. In addition, 16% persisted in the category that they were in, and 10% regressed. “The good news is, some of our kids get better over time,” Dr. Wicklow said. “The bad news is that the majority do not.”
Going forward, Dr. Wicklow and her associates work with an ICARE advisory group composed of children and families “who sit with us and talk about what mental health needs might be important, and how we should organize our study in a follow-up of the kids, to try and answer some of the questions that are important,” she said. “Working with the concept of the study’s theoretical framework, they acknowledged that the biological exposures are important, but they were also concerned about food security, finding strength/resilience within the community, and finding coping factors in terms of keeping themselves healthy with their diabetes. For some communities, they are concerned with basic needs. We’re working with them to help them progress, and to figure out how to best study children with type 2 diabetes.”
ICARE has received support from Diabetes Canada, Research Manitoba, the Canadian Institutes of Health Research, the Children’s Hospital Research Institute of Manitoba (specifically the Diabetes Research Envisioned and Accomplished in Manitoba (DREAM) theme), and the University of Manitoba. Dr. Wicklow reported having no financial disclosures.
TORONTO – When Brandy Wicklow, MD, began her pediatric endocrinology fellowship at McGill University in 2006, about 12 per 100,000 children in Manitoba, Canada, were diagnosed with type 2 diabetes mellitus each year. By 2016 that rate had more than doubled, to 26 per 100,000 children.
“If you look just at indigenous youth in our province, it’s probably one of the highest rates ever reported, with 95 per 100,000 Manitoba First Nation children diagnosed with type 2 diabetes,” said Dr. Wicklow, a pediatric endocrinologist at the University of Manitoba and the Children’s Hospital Research Institute of Manitoba.
Many indigenous populations also face an increased risk for primary renal disease. One study reviewed the charts 90 of Canadian First Nation children and adolescents with T2DM (Diabetes Care. 2009;32[5]:786-90). Of 10 who had renal biopsies performed, nine had immune complex disease/glomerulosclerosis, two had mild diabetes-related lesions, and seven had focal segmental glomerulosclerosis (FSGS); yet none had classic nephropathy. An analysis of Chinese youth that included 216 renal biopsies yielded similar findings (Intl Urol Nephrol. 2012;45[1]:173-9).
It’s also known that early-onset T2DM is associated with substantially increased incidence of end-stage renal disease (ESRD) and mortality in middle age. For example, one study of Pima Indians found that those who were diagnosed with T2DM earlier than 20 years of age had a one in five chance of developing ESRD, while those who were diagnosed at age 20 years or older had a one in two chance of ESRD (JAMA. 2006;296[4]:421-6). In a separate analysis, researchers estimated the remaining lifetime risks for ESRD among Aboriginal people in Australia with and without diabetes (Diabetes Res Clin Pract. 2014;103[3]:e24-6). The value for young adults with diabetes was high, about one in two at the age of 30 years, while it decreased with age to one in seven at 60 years.
“One of the first biomarkers we see in terms of renal disease in kids with T2DM is albuminuria,” Dr. Wicklow said at the Pediatric Academic Societies meeting. “The question is, why do kids with type 2 get more renal disease than kids with type 1 diabetes?” The SEARCH for Diabetes in Youth (SEARCH) study from 2006 found that hypertension, increased body mass index, increased weight circumference, and increased lipids were factors, while the SEARCH study from 2015 found that ethnicity, increased weight to height ratio, and mean arterial pressure were factors.
“Insulin resistance is significantly associated with albuminuria,” Dr. Wicklow continued. “It’s also been shown to be associated with hyperfiltration. Some of the markers of insulin resistance are important but they make up about 19% of the variance between type 1 and type 2, which means there are other variables that we’re not measuring.”
Enter ICARE (Improving Renal Complications in Adolescents with Type 2 Diabetes through Research), an ongoing prospective cohort study that Dr. Wicklow and her associates launched in 2014 at eight centers in Canada. It aims to examine the biopsychosocial risk factors for albuminuria in youth with T2DM and the mechanisms for renal injury. “Our theoretical framework was that biological exposures that we are aware of, such as glycemic control, hypertension, and lipids, would all be important in the development of albuminuria and renal disease in kids,” said Dr. Wicklow, who is the study’s coprimary investigator along with Allison Dart, MD. “But what we thought was novel was that psychological exposures either as socioeconomic status or as mental health factors would also directly impinge on renal health with respect to chronic inflammation in the body, inflammation in the kidneys, and long-term kidney damage.”
The first phase of ICARE involved a detailed phenotypic assessment of youth, including anthropometrics, biochemistry, 24-hour ambulatory blood pressure monitoring, overnight urine collections for albumin excretion, renal ultrasound, and iohexol-derived glomerular filtration rate (GFR). Phase 2 included an evaluation of psychological factors, including hair-derived cortisol; validated questionnaires for perceived stress, distress, and resiliency; and a detailed evaluation of systemic and urine inflammatory biomarkers. Annual follow-up is carried out to assess temporal associations between clinical risk factors and renal outcomes, including progression of albuminuria.
At the meeting, Dr. Wicklow reported on 187 youth enrolled to date. Of these, 96% were of indigenous ethnicity, 57 had albuminuria and 130 did not, and the mean ages of the groups were 16 years and 15 years, respectively. At baseline, a higher proportion of those in the albuminuria group were female (74% vs. 64% of those in the no albuminuria group, respectively), had a higher mean hemoglobin A1c (11% vs. 9%), and had hypertension (94% vs. 72%). She noted that upon presentation to the clinic, only 23% of participants had HbA1c levels less than 7%, only 26% had ranges between 7% and 9%, and about 40% did not have any hypertension. Of those who did, 27% had nighttime-only hypertension, and only 2% had daytime-only hypertension.
“The other risk factor these kids have for developing ESRD is that the majority were exposed to diabetes in pregnancy,” Dr. Wicklow said. “Murine models of maternal diabetes exposure have demonstrated that offspring have small kidneys, less ureteric bud branching, and a lower number of nephrons. Most of the human clinical cohort studies look at associations between development of diabetes and parental hypertension, maternal smoking, and maternal education. There is likely an impact at birth that sets these kids up for development of type 2 diabetes.”
In addition, results from clinical cohort studies have found that depression, mental stress, and distress are high in youth with T2DM. “Preliminary data suggest that if you have positive mental health, or coping strategies, or someone has worked through this with you and you are resilient, you might benefit in terms of overall glycemic control,” she said. For example, ICARE investigators have found that the higher the score on the Kessler Psychological Distress Scale (K6), the greater the risk of renal inflammation as measured by monocyte chemotactic protein-1 (MCP-1; P = .02). “Mental health seems to be something that can directly impact your health from a biological standpoint, and we might be able to find biomarkers of that risk,” Dr. Wicklow said. “Where does the stress come from? Most of my patients are indigenous, so it’s not surprising that the history in Canada of colonization of residential schools has left a lasting impression on these families and communities in terms of loss of language, loss of culture, and loss of land. There’s a community-based stress and a family-based stress that these children feel.”
Social factors also play a big role. She presented baseline findings from 196 youth with T2DM and 456 with T1DM, including measures such as the Socioeconomic Factor Index-Version 2 (SEFI-2), a way to assess socioeconomic characteristics based on Canadian Census data that reflects nonmedical social determinants of health. “It looks at factors like number of rooms in the house, single-parent households, maternal education attainment, and family income,” Dr. Wicklow explained. “The higher the SEFI-2 score, the lower your socioeconomic status is for the area you live in. Kids with T2DM generally live in areas of lower SES and lower socioeconomic index. They often live far away from health care providers. Many do not attend school and many are not with their biologic families, so we’ve had a lot of issues addressing child and family services, in particular in the phase of a chronic illness where our expectation is one thing and the family’s and community’s expectations of what’s realistic in terms of treatment and goals is another. We also have a lot of adolescent pregnancies.”
To date, about 80% of youth with T1D have seen a health care provider within the first year after transition from the pediatric diabetes clinic, compared with just over 50% of kids with T2D. “We transition youth with T1DM to internists, while our youth with T2DM go to itinerant physicians often back in their communities and/or rural family physicians,” she said. Between baseline and year 2, the rate of hospital admissions remained similar among T1DM at 11.6 and 11.8 admissions per 100 patient-years, respectively, but the number of hospital admissions for T2DM patients jumped from 20.1 to 25.5 admissions per 100 patient-years. “Kids with type 2 are showing up in the hospital a lot more than those with type 1 diabetes, but not for diabetes-related diagnoses,” Dr. Wicklow said. “We’re starting to look through the data now, and most of our kids are showing up with mental health complaints and issues. That’s why they’re getting hospitalized.”
Among ICARE study participants who have completed 3 years of follow-up, about 52% had albuminuria at their baseline visit and 48% sustained albuminuria throughout the study. About 26% progressed from normal levels of albuminuria to microalbuminuria, from microalbuminuria to macroalbuminuria, or from normal levels of albuminuria to macroalbuminuria. In addition, 16% persisted in the category that they were in, and 10% regressed. “The good news is, some of our kids get better over time,” Dr. Wicklow said. “The bad news is that the majority do not.”
Going forward, Dr. Wicklow and her associates work with an ICARE advisory group composed of children and families “who sit with us and talk about what mental health needs might be important, and how we should organize our study in a follow-up of the kids, to try and answer some of the questions that are important,” she said. “Working with the concept of the study’s theoretical framework, they acknowledged that the biological exposures are important, but they were also concerned about food security, finding strength/resilience within the community, and finding coping factors in terms of keeping themselves healthy with their diabetes. For some communities, they are concerned with basic needs. We’re working with them to help them progress, and to figure out how to best study children with type 2 diabetes.”
ICARE has received support from Diabetes Canada, Research Manitoba, the Canadian Institutes of Health Research, the Children’s Hospital Research Institute of Manitoba (specifically the Diabetes Research Envisioned and Accomplished in Manitoba (DREAM) theme), and the University of Manitoba. Dr. Wicklow reported having no financial disclosures.
TORONTO – When Brandy Wicklow, MD, began her pediatric endocrinology fellowship at McGill University in 2006, about 12 per 100,000 children in Manitoba, Canada, were diagnosed with type 2 diabetes mellitus each year. By 2016 that rate had more than doubled, to 26 per 100,000 children.
“If you look just at indigenous youth in our province, it’s probably one of the highest rates ever reported, with 95 per 100,000 Manitoba First Nation children diagnosed with type 2 diabetes,” said Dr. Wicklow, a pediatric endocrinologist at the University of Manitoba and the Children’s Hospital Research Institute of Manitoba.
Many indigenous populations also face an increased risk for primary renal disease. One study reviewed the charts 90 of Canadian First Nation children and adolescents with T2DM (Diabetes Care. 2009;32[5]:786-90). Of 10 who had renal biopsies performed, nine had immune complex disease/glomerulosclerosis, two had mild diabetes-related lesions, and seven had focal segmental glomerulosclerosis (FSGS); yet none had classic nephropathy. An analysis of Chinese youth that included 216 renal biopsies yielded similar findings (Intl Urol Nephrol. 2012;45[1]:173-9).
It’s also known that early-onset T2DM is associated with substantially increased incidence of end-stage renal disease (ESRD) and mortality in middle age. For example, one study of Pima Indians found that those who were diagnosed with T2DM earlier than 20 years of age had a one in five chance of developing ESRD, while those who were diagnosed at age 20 years or older had a one in two chance of ESRD (JAMA. 2006;296[4]:421-6). In a separate analysis, researchers estimated the remaining lifetime risks for ESRD among Aboriginal people in Australia with and without diabetes (Diabetes Res Clin Pract. 2014;103[3]:e24-6). The value for young adults with diabetes was high, about one in two at the age of 30 years, while it decreased with age to one in seven at 60 years.
“One of the first biomarkers we see in terms of renal disease in kids with T2DM is albuminuria,” Dr. Wicklow said at the Pediatric Academic Societies meeting. “The question is, why do kids with type 2 get more renal disease than kids with type 1 diabetes?” The SEARCH for Diabetes in Youth (SEARCH) study from 2006 found that hypertension, increased body mass index, increased weight circumference, and increased lipids were factors, while the SEARCH study from 2015 found that ethnicity, increased weight to height ratio, and mean arterial pressure were factors.
“Insulin resistance is significantly associated with albuminuria,” Dr. Wicklow continued. “It’s also been shown to be associated with hyperfiltration. Some of the markers of insulin resistance are important but they make up about 19% of the variance between type 1 and type 2, which means there are other variables that we’re not measuring.”
Enter ICARE (Improving Renal Complications in Adolescents with Type 2 Diabetes through Research), an ongoing prospective cohort study that Dr. Wicklow and her associates launched in 2014 at eight centers in Canada. It aims to examine the biopsychosocial risk factors for albuminuria in youth with T2DM and the mechanisms for renal injury. “Our theoretical framework was that biological exposures that we are aware of, such as glycemic control, hypertension, and lipids, would all be important in the development of albuminuria and renal disease in kids,” said Dr. Wicklow, who is the study’s coprimary investigator along with Allison Dart, MD. “But what we thought was novel was that psychological exposures either as socioeconomic status or as mental health factors would also directly impinge on renal health with respect to chronic inflammation in the body, inflammation in the kidneys, and long-term kidney damage.”
The first phase of ICARE involved a detailed phenotypic assessment of youth, including anthropometrics, biochemistry, 24-hour ambulatory blood pressure monitoring, overnight urine collections for albumin excretion, renal ultrasound, and iohexol-derived glomerular filtration rate (GFR). Phase 2 included an evaluation of psychological factors, including hair-derived cortisol; validated questionnaires for perceived stress, distress, and resiliency; and a detailed evaluation of systemic and urine inflammatory biomarkers. Annual follow-up is carried out to assess temporal associations between clinical risk factors and renal outcomes, including progression of albuminuria.
At the meeting, Dr. Wicklow reported on 187 youth enrolled to date. Of these, 96% were of indigenous ethnicity, 57 had albuminuria and 130 did not, and the mean ages of the groups were 16 years and 15 years, respectively. At baseline, a higher proportion of those in the albuminuria group were female (74% vs. 64% of those in the no albuminuria group, respectively), had a higher mean hemoglobin A1c (11% vs. 9%), and had hypertension (94% vs. 72%). She noted that upon presentation to the clinic, only 23% of participants had HbA1c levels less than 7%, only 26% had ranges between 7% and 9%, and about 40% did not have any hypertension. Of those who did, 27% had nighttime-only hypertension, and only 2% had daytime-only hypertension.
“The other risk factor these kids have for developing ESRD is that the majority were exposed to diabetes in pregnancy,” Dr. Wicklow said. “Murine models of maternal diabetes exposure have demonstrated that offspring have small kidneys, less ureteric bud branching, and a lower number of nephrons. Most of the human clinical cohort studies look at associations between development of diabetes and parental hypertension, maternal smoking, and maternal education. There is likely an impact at birth that sets these kids up for development of type 2 diabetes.”
In addition, results from clinical cohort studies have found that depression, mental stress, and distress are high in youth with T2DM. “Preliminary data suggest that if you have positive mental health, or coping strategies, or someone has worked through this with you and you are resilient, you might benefit in terms of overall glycemic control,” she said. For example, ICARE investigators have found that the higher the score on the Kessler Psychological Distress Scale (K6), the greater the risk of renal inflammation as measured by monocyte chemotactic protein-1 (MCP-1; P = .02). “Mental health seems to be something that can directly impact your health from a biological standpoint, and we might be able to find biomarkers of that risk,” Dr. Wicklow said. “Where does the stress come from? Most of my patients are indigenous, so it’s not surprising that the history in Canada of colonization of residential schools has left a lasting impression on these families and communities in terms of loss of language, loss of culture, and loss of land. There’s a community-based stress and a family-based stress that these children feel.”
Social factors also play a big role. She presented baseline findings from 196 youth with T2DM and 456 with T1DM, including measures such as the Socioeconomic Factor Index-Version 2 (SEFI-2), a way to assess socioeconomic characteristics based on Canadian Census data that reflects nonmedical social determinants of health. “It looks at factors like number of rooms in the house, single-parent households, maternal education attainment, and family income,” Dr. Wicklow explained. “The higher the SEFI-2 score, the lower your socioeconomic status is for the area you live in. Kids with T2DM generally live in areas of lower SES and lower socioeconomic index. They often live far away from health care providers. Many do not attend school and many are not with their biologic families, so we’ve had a lot of issues addressing child and family services, in particular in the phase of a chronic illness where our expectation is one thing and the family’s and community’s expectations of what’s realistic in terms of treatment and goals is another. We also have a lot of adolescent pregnancies.”
To date, about 80% of youth with T1D have seen a health care provider within the first year after transition from the pediatric diabetes clinic, compared with just over 50% of kids with T2D. “We transition youth with T1DM to internists, while our youth with T2DM go to itinerant physicians often back in their communities and/or rural family physicians,” she said. Between baseline and year 2, the rate of hospital admissions remained similar among T1DM at 11.6 and 11.8 admissions per 100 patient-years, respectively, but the number of hospital admissions for T2DM patients jumped from 20.1 to 25.5 admissions per 100 patient-years. “Kids with type 2 are showing up in the hospital a lot more than those with type 1 diabetes, but not for diabetes-related diagnoses,” Dr. Wicklow said. “We’re starting to look through the data now, and most of our kids are showing up with mental health complaints and issues. That’s why they’re getting hospitalized.”
Among ICARE study participants who have completed 3 years of follow-up, about 52% had albuminuria at their baseline visit and 48% sustained albuminuria throughout the study. About 26% progressed from normal levels of albuminuria to microalbuminuria, from microalbuminuria to macroalbuminuria, or from normal levels of albuminuria to macroalbuminuria. In addition, 16% persisted in the category that they were in, and 10% regressed. “The good news is, some of our kids get better over time,” Dr. Wicklow said. “The bad news is that the majority do not.”
Going forward, Dr. Wicklow and her associates work with an ICARE advisory group composed of children and families “who sit with us and talk about what mental health needs might be important, and how we should organize our study in a follow-up of the kids, to try and answer some of the questions that are important,” she said. “Working with the concept of the study’s theoretical framework, they acknowledged that the biological exposures are important, but they were also concerned about food security, finding strength/resilience within the community, and finding coping factors in terms of keeping themselves healthy with their diabetes. For some communities, they are concerned with basic needs. We’re working with them to help them progress, and to figure out how to best study children with type 2 diabetes.”
ICARE has received support from Diabetes Canada, Research Manitoba, the Canadian Institutes of Health Research, the Children’s Hospital Research Institute of Manitoba (specifically the Diabetes Research Envisioned and Accomplished in Manitoba (DREAM) theme), and the University of Manitoba. Dr. Wicklow reported having no financial disclosures.
AT PAS 18
Key clinical point: Mental health may indirectly increase inflammation, which contributes to kidney health.
Major finding: The higher the score on the Kessler Psychological Distress Scale (K6), the greater the risk of renal inflammation as measured by MCP-1 (P = .02).
Study details: Preliminary results from ICARE (Improving Renal Complications in Adolescents with Type 2 Diabetes through Research), an ongoing prospective cohort study.
Disclosures: ICARE has received support from Diabetes Canada, Research Manitoba, the Canadian Institutes of Health Research, the Children’s Hospital Research Institute of Manitoba (specifically the Diabetes Research Envisioned and Accomplished in Manitoba [DREAM] theme), and the University of Manitoba. Dr. Wicklow reported having no financial disclosures.
App found to improve quality of life for families of premature infants
TORONTO – Significant improvement in quality of life was observed in neonatal ICU families using the PreeMe+You app, preliminary results from a two-center study showed.
“NICU time is stressful,” one of the study authors, Abigail Whitney, said at the Pediatric Academic Societies annual meeting. “With the birth of a preterm infant, parents are often quickly transitioned into the role of becoming a parent much sooner and in much different circumstances than they might have anticipated. Parents have reported feelings of isolation, alienation, and insecurity in the parental role while in the NICU. Studies have shown that interventions that engage parents in their infant’s progress can decrease parental stress and anxiety, increase positive parent-infant interaction, and even reduce the infant’s length of stay. Also, with advancing technology there has been a push to find ways to use mobile technology to help parents balance engaging with their infant with the rest of their busy lives.”
In a study overseen by PreeMe+You’s chief medical expert, Bree Andrews, MD, MPH, Ms. Whitney and her associates administered the app to 48 families at either the University of Chicago Medicine Comer Children’s Hospital NICU or the Evanston Hospital NICU to assess readiness for using mobile technologies at the bedside. All families were recommended by a child life specialist who identified families who might be interested in using something like PreeMe+You. They excluded any families that were currently involved with child and family services, those with an infant younger than 7 days old, those whose child required escalation of care or upcoming surgeries, and those whose infant was over 37 weeks’ gestation.
First, the researchers briefed NICU staff about the study at charge nurse meetings, faculty meetings, and daily huddles for 2 weeks before first enrollment. “We did this knowing that parents might go to their nurses or doctors about how to answer specific questions within the app, or maybe want to learn more about a certain topic they learned from PreeMe+You,” Ms. Whitney said.
Data measurements included the PreeMe+You composite survey, which pulled questions from the Fragile Infant Parent Readiness Evaluation (FIPRE) and the NICU Parent Risk Evaluation and Engagement Model and Instrument (PREEMI). “We also included additional questions about technology use and capacity, as well as the PedsQL [Pediatric Quality of Life Inventory] Family Impact Module to assess parental quality of life throughout the study,” she said.
Over a period of 9 months, the researchers collected 153 quality of life measurements from 48 families. Of these, 48 occurred at enrollment, 23 occurred less than 1 week after enrollment, 30 occurred 1-2 weeks after enrollment, 28 occurred 3-4 weeks after enrollment, and 24 occurred 4 weeks or more after enrollment. By study closure, the researchers had follow-up data on 44 of the 48 families. The average gestational age at birth was 29.3 weeks, the average day of life at enrollment was 25.4, and the average birth weight was 1,280 grams.
On the app’s composite survey, 14.6% “agreed” and 79.2% “strongly agreed” that they were currently using a smart phone or tablet to look for information about preemies/NICU on the Internet, and about half “agreed” or “strongly agreed” (27.1% and 33.3%, respectively) that they spent more than 30 minutes per week looking up information about their NICU baby online. Nearly all families “agreed” or “strongly agreed” (14.6% and 85.4%) that they had a smart phone or tablet for Internet use in the NICU, and nearly all “agreed” or “strongly agreed” (33.3% and 62.5%) that having an app at the NICU bedside/home would be helpful. “This showed us that families were ready to use technology and interested in something like PreeMe+You at the bedside,” Ms. Whitney said.
At the time of study enrollment, 12 were in the purple stage, 8 were in the blue stage, 19 infants were in the orange stage, and 9 were in the yellow stage. Ms. Whitney reported that based on the PedsQL Family Impact Module, 35 of the 44 families showed increased quality of life functionality after participating in the study. This change was significant, with a P value of .001. Improvements were seen in the measure’s eight domains (physical, emotional, social, cognitive, communication, worry, daily activities, and family relationship functionality). “We saw increases across all of the domains based on how long the parents had been using the app,” Ms. Whitney said. “We found the biggest increase in quality of life in families of babies born less than 25 weeks’ gestational age, those born 25-26 weeks gestational age, those born 27-28 weeks gestational age, and those born 33-37 weeks gestational age. We are encouraged to see some of these quality of life changes in some of the earliest-born gestation babies because these are presumably the families that would have the longest time to go in the NICU and could benefit the most from using an app like PreeMe+You.”
She acknowledged certain limitations of the study, including the fact that it was conducted in two NICUs, “and we definitely need more comparisons to look at the natural trajectory of quality of life changes while families are in the NICU. Also, all of the families enrolled in our study had access to a research team that checked in with them weekly. In the real world, PreeMe+You would probably be self-guided.” Going forward, PreeMe+You plans to include additional features to give parents more self-guidance, making it easier for them to interact and partner with their baby’s medical team.
Funding for the study was provided by the Bucksbaum Institute for Clinical Excellence. Ms. Whitney was supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases.
TORONTO – Significant improvement in quality of life was observed in neonatal ICU families using the PreeMe+You app, preliminary results from a two-center study showed.
“NICU time is stressful,” one of the study authors, Abigail Whitney, said at the Pediatric Academic Societies annual meeting. “With the birth of a preterm infant, parents are often quickly transitioned into the role of becoming a parent much sooner and in much different circumstances than they might have anticipated. Parents have reported feelings of isolation, alienation, and insecurity in the parental role while in the NICU. Studies have shown that interventions that engage parents in their infant’s progress can decrease parental stress and anxiety, increase positive parent-infant interaction, and even reduce the infant’s length of stay. Also, with advancing technology there has been a push to find ways to use mobile technology to help parents balance engaging with their infant with the rest of their busy lives.”
In a study overseen by PreeMe+You’s chief medical expert, Bree Andrews, MD, MPH, Ms. Whitney and her associates administered the app to 48 families at either the University of Chicago Medicine Comer Children’s Hospital NICU or the Evanston Hospital NICU to assess readiness for using mobile technologies at the bedside. All families were recommended by a child life specialist who identified families who might be interested in using something like PreeMe+You. They excluded any families that were currently involved with child and family services, those with an infant younger than 7 days old, those whose child required escalation of care or upcoming surgeries, and those whose infant was over 37 weeks’ gestation.
First, the researchers briefed NICU staff about the study at charge nurse meetings, faculty meetings, and daily huddles for 2 weeks before first enrollment. “We did this knowing that parents might go to their nurses or doctors about how to answer specific questions within the app, or maybe want to learn more about a certain topic they learned from PreeMe+You,” Ms. Whitney said.
Data measurements included the PreeMe+You composite survey, which pulled questions from the Fragile Infant Parent Readiness Evaluation (FIPRE) and the NICU Parent Risk Evaluation and Engagement Model and Instrument (PREEMI). “We also included additional questions about technology use and capacity, as well as the PedsQL [Pediatric Quality of Life Inventory] Family Impact Module to assess parental quality of life throughout the study,” she said.
Over a period of 9 months, the researchers collected 153 quality of life measurements from 48 families. Of these, 48 occurred at enrollment, 23 occurred less than 1 week after enrollment, 30 occurred 1-2 weeks after enrollment, 28 occurred 3-4 weeks after enrollment, and 24 occurred 4 weeks or more after enrollment. By study closure, the researchers had follow-up data on 44 of the 48 families. The average gestational age at birth was 29.3 weeks, the average day of life at enrollment was 25.4, and the average birth weight was 1,280 grams.
On the app’s composite survey, 14.6% “agreed” and 79.2% “strongly agreed” that they were currently using a smart phone or tablet to look for information about preemies/NICU on the Internet, and about half “agreed” or “strongly agreed” (27.1% and 33.3%, respectively) that they spent more than 30 minutes per week looking up information about their NICU baby online. Nearly all families “agreed” or “strongly agreed” (14.6% and 85.4%) that they had a smart phone or tablet for Internet use in the NICU, and nearly all “agreed” or “strongly agreed” (33.3% and 62.5%) that having an app at the NICU bedside/home would be helpful. “This showed us that families were ready to use technology and interested in something like PreeMe+You at the bedside,” Ms. Whitney said.
At the time of study enrollment, 12 were in the purple stage, 8 were in the blue stage, 19 infants were in the orange stage, and 9 were in the yellow stage. Ms. Whitney reported that based on the PedsQL Family Impact Module, 35 of the 44 families showed increased quality of life functionality after participating in the study. This change was significant, with a P value of .001. Improvements were seen in the measure’s eight domains (physical, emotional, social, cognitive, communication, worry, daily activities, and family relationship functionality). “We saw increases across all of the domains based on how long the parents had been using the app,” Ms. Whitney said. “We found the biggest increase in quality of life in families of babies born less than 25 weeks’ gestational age, those born 25-26 weeks gestational age, those born 27-28 weeks gestational age, and those born 33-37 weeks gestational age. We are encouraged to see some of these quality of life changes in some of the earliest-born gestation babies because these are presumably the families that would have the longest time to go in the NICU and could benefit the most from using an app like PreeMe+You.”
She acknowledged certain limitations of the study, including the fact that it was conducted in two NICUs, “and we definitely need more comparisons to look at the natural trajectory of quality of life changes while families are in the NICU. Also, all of the families enrolled in our study had access to a research team that checked in with them weekly. In the real world, PreeMe+You would probably be self-guided.” Going forward, PreeMe+You plans to include additional features to give parents more self-guidance, making it easier for them to interact and partner with their baby’s medical team.
Funding for the study was provided by the Bucksbaum Institute for Clinical Excellence. Ms. Whitney was supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases.
TORONTO – Significant improvement in quality of life was observed in neonatal ICU families using the PreeMe+You app, preliminary results from a two-center study showed.
“NICU time is stressful,” one of the study authors, Abigail Whitney, said at the Pediatric Academic Societies annual meeting. “With the birth of a preterm infant, parents are often quickly transitioned into the role of becoming a parent much sooner and in much different circumstances than they might have anticipated. Parents have reported feelings of isolation, alienation, and insecurity in the parental role while in the NICU. Studies have shown that interventions that engage parents in their infant’s progress can decrease parental stress and anxiety, increase positive parent-infant interaction, and even reduce the infant’s length of stay. Also, with advancing technology there has been a push to find ways to use mobile technology to help parents balance engaging with their infant with the rest of their busy lives.”
In a study overseen by PreeMe+You’s chief medical expert, Bree Andrews, MD, MPH, Ms. Whitney and her associates administered the app to 48 families at either the University of Chicago Medicine Comer Children’s Hospital NICU or the Evanston Hospital NICU to assess readiness for using mobile technologies at the bedside. All families were recommended by a child life specialist who identified families who might be interested in using something like PreeMe+You. They excluded any families that were currently involved with child and family services, those with an infant younger than 7 days old, those whose child required escalation of care or upcoming surgeries, and those whose infant was over 37 weeks’ gestation.
First, the researchers briefed NICU staff about the study at charge nurse meetings, faculty meetings, and daily huddles for 2 weeks before first enrollment. “We did this knowing that parents might go to their nurses or doctors about how to answer specific questions within the app, or maybe want to learn more about a certain topic they learned from PreeMe+You,” Ms. Whitney said.
Data measurements included the PreeMe+You composite survey, which pulled questions from the Fragile Infant Parent Readiness Evaluation (FIPRE) and the NICU Parent Risk Evaluation and Engagement Model and Instrument (PREEMI). “We also included additional questions about technology use and capacity, as well as the PedsQL [Pediatric Quality of Life Inventory] Family Impact Module to assess parental quality of life throughout the study,” she said.
Over a period of 9 months, the researchers collected 153 quality of life measurements from 48 families. Of these, 48 occurred at enrollment, 23 occurred less than 1 week after enrollment, 30 occurred 1-2 weeks after enrollment, 28 occurred 3-4 weeks after enrollment, and 24 occurred 4 weeks or more after enrollment. By study closure, the researchers had follow-up data on 44 of the 48 families. The average gestational age at birth was 29.3 weeks, the average day of life at enrollment was 25.4, and the average birth weight was 1,280 grams.
On the app’s composite survey, 14.6% “agreed” and 79.2% “strongly agreed” that they were currently using a smart phone or tablet to look for information about preemies/NICU on the Internet, and about half “agreed” or “strongly agreed” (27.1% and 33.3%, respectively) that they spent more than 30 minutes per week looking up information about their NICU baby online. Nearly all families “agreed” or “strongly agreed” (14.6% and 85.4%) that they had a smart phone or tablet for Internet use in the NICU, and nearly all “agreed” or “strongly agreed” (33.3% and 62.5%) that having an app at the NICU bedside/home would be helpful. “This showed us that families were ready to use technology and interested in something like PreeMe+You at the bedside,” Ms. Whitney said.
At the time of study enrollment, 12 were in the purple stage, 8 were in the blue stage, 19 infants were in the orange stage, and 9 were in the yellow stage. Ms. Whitney reported that based on the PedsQL Family Impact Module, 35 of the 44 families showed increased quality of life functionality after participating in the study. This change was significant, with a P value of .001. Improvements were seen in the measure’s eight domains (physical, emotional, social, cognitive, communication, worry, daily activities, and family relationship functionality). “We saw increases across all of the domains based on how long the parents had been using the app,” Ms. Whitney said. “We found the biggest increase in quality of life in families of babies born less than 25 weeks’ gestational age, those born 25-26 weeks gestational age, those born 27-28 weeks gestational age, and those born 33-37 weeks gestational age. We are encouraged to see some of these quality of life changes in some of the earliest-born gestation babies because these are presumably the families that would have the longest time to go in the NICU and could benefit the most from using an app like PreeMe+You.”
She acknowledged certain limitations of the study, including the fact that it was conducted in two NICUs, “and we definitely need more comparisons to look at the natural trajectory of quality of life changes while families are in the NICU. Also, all of the families enrolled in our study had access to a research team that checked in with them weekly. In the real world, PreeMe+You would probably be self-guided.” Going forward, PreeMe+You plans to include additional features to give parents more self-guidance, making it easier for them to interact and partner with their baby’s medical team.
Funding for the study was provided by the Bucksbaum Institute for Clinical Excellence. Ms. Whitney was supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases.
REPORTING FROM PAS 2018
Key clinical point:
Major finding: In all, 35 of the 44 families showed increased quality of life functionality, based on the PedsQL Family Impact Module (P = .001).
Study details: A two-center study of 44 families with premature infants intended to assess readiness for using mobile technologies at the bedside.
Disclosures: Funding for the study was provided by the Bucksbaum Institute for Clinical Excellence. Ms. Whitney was supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases.
Trio of blood biomarkers elevated in children with LRTIs
TORONTO – While C-reactive protein, procalcitonin, and proadrenomedullin are associated with development of severe clinical outcomes in children with lower respiratory tract infections, proadrenomedullin is most strongly associated with disease severity, preliminary results from a prospective cohort study showed.
“Despite the fact that pneumonia guidelines call the site of care decision the most important decision in the management of pediatric pneumonia, no validated risk stratification tools exist for pediatric lower respiratory tract infections (LRTI),” lead study author Todd A. Florin, MD, said at the annual Pediatric Academic Societies meeting. “Biomarkers offer an objective means of classifying disease severity and clinical outcomes.”
PCT is a precursor of calcitonin secreted by the thyroid, lung, and intestine in response to bacterial infections. It also has been shown to be associated with adverse outcomes and mortality in adults, with results generally suggesting that it is a stronger predictor of severity than CRP. “There is limited data on the association of CRP or PCT with severe outcomes in children with LRTIs,” Dr. Florin noted. “One recent U.S. study of 532 children did demonstrate an association of elevated PCT with ICU admission, chest drainage, and hospital length of stay in children with [community-acquired pneumonia] CAP.”
ProADM, meanwhile, is a vasodilatory peptide with antimicrobial and anti-inflammatory functions synthesized during severe infections. It has a half-life of several hours and has been shown to be associated with disease severity in adults with LRTI. Recent studies have shown that it has improved prognostication over WBC, CRP, and PCT. “In two small studies of children with pneumonia, proADM levels were significantly elevated in children with complicated pneumonia, compared to those with uncomplicated pneumonia,” Dr. Florin said. “Although all three of these markers demonstrate promise in predicting severe outcomes in adults with LRTIs, very few studies have examined their association with disease severity in pediatric disease. Therefore, the aim of the current analysis was to determine the association between blood biomarkers and disease severity in children who present to the ED with lower respiratory tract infections.”
In a study known as Catalyzing Ambulatory Research in Pneumonia Etiology and Diagnostic Innovations in Emergency Medicine (CARPE DIEM), he and his associates performed a prospective cohort analysis of children with suspected CAP who were admitted to the Cincinnati Children’s Hospital ED between July 2012 and December 2017. They limited the analysis to children aged 3 months to 18 years with signs and symptoms of an LRTI, and all eligible patients were required to have a chest radiograph ordered for suspicion of CAP. They excluded children hospitalized within 14 days prior to the index ED visit, immunodeficient or immunosuppressed children, those with a history of aspiration or aspiration pneumonia, and those who weighed less than 5 kg because of blood drawing maximums. Biomarkers were measured only in children with focal findings on chest x-ray in the ED. The primary outcome was disease severity: mild (defined as discharged home), moderate (defined as hospitalized, but not severe) and severe (defined as having an ICU length of stay of greater than 48 hours, chest drainage, severe sepsis, noninvasive positive pressure ventilation, intubation, vasoactive infusions, or death). Biomarkers were obtained at the time of presentation to the ED, prior to the occurrence of clinical outcomes.
Over a period of 4.5 years, the researchers enrolled 1,142 patients. Of these, 478 had focal findings on chest x-ray and blood obtained. The median age of these 478 children was 4.4 years, 52% were male, and 82% had all three biomarkers performed. Specifically, 456 had CRP and PCT performed, while 358 had proADM performed. “Not every child had every marker performed due to challenges in obtaining sufficient blood for all three biomarkers in some children,” Dr. Florin explained.
Preliminary data that Dr. Florin presented at PAS found that the median CRP, PCT, and proADM did not differ by gender, race, ethnicity, or insurance status. “In addition, there were not significant differences in the distribution of disease severity by biomarker performed, with approximately 27% of patients being classified as mild, 66% as moderate, and 7% as severe,” he said.
The median CRP was 2.4 ng/mL in those with mild disease, 2.5 ng/mL in those with moderate disease, and 6.25 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease reaching statistical significance (P = .002). The median PCT was 0.16 ng/mL in those with mild disease, 0.26 ng/mL in those with moderate disease, and 0.49 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease reaching statistical significance (P = .047). Meanwhile, the median proADM was 0.53 ng/mL in those with mild disease, 0.59 ng/mL in those with moderate disease, and 0.81 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease also reaching statistical significance (P less than .0001).
Next, the researchers performed logistic regression of each biomarker individually and in combination. They found that and had the best ability to discriminate those developing severe vs. nonsevere disease (area under the receiving operating curve of 0.72, vs. 0.67 and 0.60, respectively). When CRP and PCT markers were combined with proADM, they were no longer associated with severe disease, while a strong association with proADM remained significant.
Dr. Florin acknowledged certain limitations of the study, including the fact that requiring collection of blood samples may have resulted in an enrollment bias toward patients receiving phlebotomy or IV line placement in the ED. “In addition, the children in the moderate-severity group are likely more heterogeneous than the other two severity groups,” he said. “Finally, given that this is a single-center study, we had a relatively small number of outcomes for some of the individual severity measures, which may have limited power and precision.”
He concluded his presentation by saying that he is “cautiously optimistic” about the study results. “As is the case in many biomarker studies, I do not anticipate that any single biomarker will be the magic bullet for predicting disease severity in pediatric CAP,” Dr. Florin said. “It will likely be a combination of clinical factors and several biomarkers that will achieve optimal prognostic ability. That said, our results suggest that similar to adult studies, proADM appears to have the strongest association with severe disease, compared with CRP and PCT. Combinations of biomarkers did not perform better than proADM alone. With the advent of rapid point-of-care diagnostics, these markers may have a role in management and site-of-care decisions for children with LRTI.”
The study received funding support from the Gerber Foundation, the National Institute of Allergy and Infectious Diseases, and Cincinnati Children’s Hospital Medical Center. Dr. Florin reported having no financial disclosures.
TORONTO – While C-reactive protein, procalcitonin, and proadrenomedullin are associated with development of severe clinical outcomes in children with lower respiratory tract infections, proadrenomedullin is most strongly associated with disease severity, preliminary results from a prospective cohort study showed.
“Despite the fact that pneumonia guidelines call the site of care decision the most important decision in the management of pediatric pneumonia, no validated risk stratification tools exist for pediatric lower respiratory tract infections (LRTI),” lead study author Todd A. Florin, MD, said at the annual Pediatric Academic Societies meeting. “Biomarkers offer an objective means of classifying disease severity and clinical outcomes.”
PCT is a precursor of calcitonin secreted by the thyroid, lung, and intestine in response to bacterial infections. It also has been shown to be associated with adverse outcomes and mortality in adults, with results generally suggesting that it is a stronger predictor of severity than CRP. “There is limited data on the association of CRP or PCT with severe outcomes in children with LRTIs,” Dr. Florin noted. “One recent U.S. study of 532 children did demonstrate an association of elevated PCT with ICU admission, chest drainage, and hospital length of stay in children with [community-acquired pneumonia] CAP.”
ProADM, meanwhile, is a vasodilatory peptide with antimicrobial and anti-inflammatory functions synthesized during severe infections. It has a half-life of several hours and has been shown to be associated with disease severity in adults with LRTI. Recent studies have shown that it has improved prognostication over WBC, CRP, and PCT. “In two small studies of children with pneumonia, proADM levels were significantly elevated in children with complicated pneumonia, compared to those with uncomplicated pneumonia,” Dr. Florin said. “Although all three of these markers demonstrate promise in predicting severe outcomes in adults with LRTIs, very few studies have examined their association with disease severity in pediatric disease. Therefore, the aim of the current analysis was to determine the association between blood biomarkers and disease severity in children who present to the ED with lower respiratory tract infections.”
In a study known as Catalyzing Ambulatory Research in Pneumonia Etiology and Diagnostic Innovations in Emergency Medicine (CARPE DIEM), he and his associates performed a prospective cohort analysis of children with suspected CAP who were admitted to the Cincinnati Children’s Hospital ED between July 2012 and December 2017. They limited the analysis to children aged 3 months to 18 years with signs and symptoms of an LRTI, and all eligible patients were required to have a chest radiograph ordered for suspicion of CAP. They excluded children hospitalized within 14 days prior to the index ED visit, immunodeficient or immunosuppressed children, those with a history of aspiration or aspiration pneumonia, and those who weighed less than 5 kg because of blood drawing maximums. Biomarkers were measured only in children with focal findings on chest x-ray in the ED. The primary outcome was disease severity: mild (defined as discharged home), moderate (defined as hospitalized, but not severe) and severe (defined as having an ICU length of stay of greater than 48 hours, chest drainage, severe sepsis, noninvasive positive pressure ventilation, intubation, vasoactive infusions, or death). Biomarkers were obtained at the time of presentation to the ED, prior to the occurrence of clinical outcomes.
Over a period of 4.5 years, the researchers enrolled 1,142 patients. Of these, 478 had focal findings on chest x-ray and blood obtained. The median age of these 478 children was 4.4 years, 52% were male, and 82% had all three biomarkers performed. Specifically, 456 had CRP and PCT performed, while 358 had proADM performed. “Not every child had every marker performed due to challenges in obtaining sufficient blood for all three biomarkers in some children,” Dr. Florin explained.
Preliminary data that Dr. Florin presented at PAS found that the median CRP, PCT, and proADM did not differ by gender, race, ethnicity, or insurance status. “In addition, there were not significant differences in the distribution of disease severity by biomarker performed, with approximately 27% of patients being classified as mild, 66% as moderate, and 7% as severe,” he said.
The median CRP was 2.4 ng/mL in those with mild disease, 2.5 ng/mL in those with moderate disease, and 6.25 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease reaching statistical significance (P = .002). The median PCT was 0.16 ng/mL in those with mild disease, 0.26 ng/mL in those with moderate disease, and 0.49 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease reaching statistical significance (P = .047). Meanwhile, the median proADM was 0.53 ng/mL in those with mild disease, 0.59 ng/mL in those with moderate disease, and 0.81 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease also reaching statistical significance (P less than .0001).
Next, the researchers performed logistic regression of each biomarker individually and in combination. They found that and had the best ability to discriminate those developing severe vs. nonsevere disease (area under the receiving operating curve of 0.72, vs. 0.67 and 0.60, respectively). When CRP and PCT markers were combined with proADM, they were no longer associated with severe disease, while a strong association with proADM remained significant.
Dr. Florin acknowledged certain limitations of the study, including the fact that requiring collection of blood samples may have resulted in an enrollment bias toward patients receiving phlebotomy or IV line placement in the ED. “In addition, the children in the moderate-severity group are likely more heterogeneous than the other two severity groups,” he said. “Finally, given that this is a single-center study, we had a relatively small number of outcomes for some of the individual severity measures, which may have limited power and precision.”
He concluded his presentation by saying that he is “cautiously optimistic” about the study results. “As is the case in many biomarker studies, I do not anticipate that any single biomarker will be the magic bullet for predicting disease severity in pediatric CAP,” Dr. Florin said. “It will likely be a combination of clinical factors and several biomarkers that will achieve optimal prognostic ability. That said, our results suggest that similar to adult studies, proADM appears to have the strongest association with severe disease, compared with CRP and PCT. Combinations of biomarkers did not perform better than proADM alone. With the advent of rapid point-of-care diagnostics, these markers may have a role in management and site-of-care decisions for children with LRTI.”
The study received funding support from the Gerber Foundation, the National Institute of Allergy and Infectious Diseases, and Cincinnati Children’s Hospital Medical Center. Dr. Florin reported having no financial disclosures.
TORONTO – While C-reactive protein, procalcitonin, and proadrenomedullin are associated with development of severe clinical outcomes in children with lower respiratory tract infections, proadrenomedullin is most strongly associated with disease severity, preliminary results from a prospective cohort study showed.
“Despite the fact that pneumonia guidelines call the site of care decision the most important decision in the management of pediatric pneumonia, no validated risk stratification tools exist for pediatric lower respiratory tract infections (LRTI),” lead study author Todd A. Florin, MD, said at the annual Pediatric Academic Societies meeting. “Biomarkers offer an objective means of classifying disease severity and clinical outcomes.”
PCT is a precursor of calcitonin secreted by the thyroid, lung, and intestine in response to bacterial infections. It also has been shown to be associated with adverse outcomes and mortality in adults, with results generally suggesting that it is a stronger predictor of severity than CRP. “There is limited data on the association of CRP or PCT with severe outcomes in children with LRTIs,” Dr. Florin noted. “One recent U.S. study of 532 children did demonstrate an association of elevated PCT with ICU admission, chest drainage, and hospital length of stay in children with [community-acquired pneumonia] CAP.”
ProADM, meanwhile, is a vasodilatory peptide with antimicrobial and anti-inflammatory functions synthesized during severe infections. It has a half-life of several hours and has been shown to be associated with disease severity in adults with LRTI. Recent studies have shown that it has improved prognostication over WBC, CRP, and PCT. “In two small studies of children with pneumonia, proADM levels were significantly elevated in children with complicated pneumonia, compared to those with uncomplicated pneumonia,” Dr. Florin said. “Although all three of these markers demonstrate promise in predicting severe outcomes in adults with LRTIs, very few studies have examined their association with disease severity in pediatric disease. Therefore, the aim of the current analysis was to determine the association between blood biomarkers and disease severity in children who present to the ED with lower respiratory tract infections.”
In a study known as Catalyzing Ambulatory Research in Pneumonia Etiology and Diagnostic Innovations in Emergency Medicine (CARPE DIEM), he and his associates performed a prospective cohort analysis of children with suspected CAP who were admitted to the Cincinnati Children’s Hospital ED between July 2012 and December 2017. They limited the analysis to children aged 3 months to 18 years with signs and symptoms of an LRTI, and all eligible patients were required to have a chest radiograph ordered for suspicion of CAP. They excluded children hospitalized within 14 days prior to the index ED visit, immunodeficient or immunosuppressed children, those with a history of aspiration or aspiration pneumonia, and those who weighed less than 5 kg because of blood drawing maximums. Biomarkers were measured only in children with focal findings on chest x-ray in the ED. The primary outcome was disease severity: mild (defined as discharged home), moderate (defined as hospitalized, but not severe) and severe (defined as having an ICU length of stay of greater than 48 hours, chest drainage, severe sepsis, noninvasive positive pressure ventilation, intubation, vasoactive infusions, or death). Biomarkers were obtained at the time of presentation to the ED, prior to the occurrence of clinical outcomes.
Over a period of 4.5 years, the researchers enrolled 1,142 patients. Of these, 478 had focal findings on chest x-ray and blood obtained. The median age of these 478 children was 4.4 years, 52% were male, and 82% had all three biomarkers performed. Specifically, 456 had CRP and PCT performed, while 358 had proADM performed. “Not every child had every marker performed due to challenges in obtaining sufficient blood for all three biomarkers in some children,” Dr. Florin explained.
Preliminary data that Dr. Florin presented at PAS found that the median CRP, PCT, and proADM did not differ by gender, race, ethnicity, or insurance status. “In addition, there were not significant differences in the distribution of disease severity by biomarker performed, with approximately 27% of patients being classified as mild, 66% as moderate, and 7% as severe,” he said.
The median CRP was 2.4 ng/mL in those with mild disease, 2.5 ng/mL in those with moderate disease, and 6.25 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease reaching statistical significance (P = .002). The median PCT was 0.16 ng/mL in those with mild disease, 0.26 ng/mL in those with moderate disease, and 0.49 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease reaching statistical significance (P = .047). Meanwhile, the median proADM was 0.53 ng/mL in those with mild disease, 0.59 ng/mL in those with moderate disease, and 0.81 ng/mL in those with severe disease, with the difference between the two subclasses of nonsevere disease and moderate disease and severe disease also reaching statistical significance (P less than .0001).
Next, the researchers performed logistic regression of each biomarker individually and in combination. They found that and had the best ability to discriminate those developing severe vs. nonsevere disease (area under the receiving operating curve of 0.72, vs. 0.67 and 0.60, respectively). When CRP and PCT markers were combined with proADM, they were no longer associated with severe disease, while a strong association with proADM remained significant.
Dr. Florin acknowledged certain limitations of the study, including the fact that requiring collection of blood samples may have resulted in an enrollment bias toward patients receiving phlebotomy or IV line placement in the ED. “In addition, the children in the moderate-severity group are likely more heterogeneous than the other two severity groups,” he said. “Finally, given that this is a single-center study, we had a relatively small number of outcomes for some of the individual severity measures, which may have limited power and precision.”
He concluded his presentation by saying that he is “cautiously optimistic” about the study results. “As is the case in many biomarker studies, I do not anticipate that any single biomarker will be the magic bullet for predicting disease severity in pediatric CAP,” Dr. Florin said. “It will likely be a combination of clinical factors and several biomarkers that will achieve optimal prognostic ability. That said, our results suggest that similar to adult studies, proADM appears to have the strongest association with severe disease, compared with CRP and PCT. Combinations of biomarkers did not perform better than proADM alone. With the advent of rapid point-of-care diagnostics, these markers may have a role in management and site-of-care decisions for children with LRTI.”
The study received funding support from the Gerber Foundation, the National Institute of Allergy and Infectious Diseases, and Cincinnati Children’s Hospital Medical Center. Dr. Florin reported having no financial disclosures.
AT PAS 18
Key clinical point: Blood biomarkers such as C-reactive protein (CRP), procalcitonin (PCT), and proadrenomedullin (proADM) may have a role in management and site-of-care decisions for children with LRTIs.
Major finding: The proADM alone was associated with the largest odds for severe disease (OR 13.1), compared with CRP alone (OR 1.6) and PCT alone (OR 1.4).
Study details: Preliminary results from prospective cohort analysis of 478 children with suspected community-acquired pneumonia who were admitted to the Cincinnati Children’s Hospital ED.
Disclosures: The study received funding support from the Gerber Foundation, the National Institute of Allergy and Infectious Diseases, and Cincinnati Children’s Hospital Medical Center. Dr. Florin reported having no financial disclosures.
Brief stress management training helps teens with mental health complaints
TORONTO – A brief stress management intervention left a lasting effect on adolescents referred for mental health–related complaints, significantly reducing perceived distress, and heart rate variability. The majority of recipients expressed interest in additional training at follow-up.
Elizabeth B. Mason, MD, and her colleagues from Rainbow Babies and Children’s Hospital in Cleveland, reported in a poster presentation at the Pediatric Academic Societies annual meeting.
The vast majority of participants (96.5%) were African American, mean age was 16 years, and 36% were male. Cutoff criteria for generalized anxiety on the SCARED was met by 35 (41%) participants, and 23 (27%) participants scored positive for depression on the PHQ-A.
Following completion of preintervention questionnaires, 50 of 86 (58%) participants received psychoeducation from an adolescent medicine fellow or a pediatric psychologist on the effect stress has on the body, training in diaphragmatic breathing and progressive muscle relaxation, and no-cost/low-cost exercise options. Study participants also engaged in a peripheral biofeedback program called Unyte that has been shown to improve heart rate variability. The remaining 36 participants received no training and served as controls.
Those in the intervention group had significantly lower SUDS scores postintervention than did the control group. Heart rate variability coherence rates also decreased significantly postintervention in those who received the intervention, compared with controls.
When reached by phone 1 week after the session, 92% of participants said they found the intervention helpful and felt more relaxed, and 44% expressed interest in additional relaxation training.
The investigators concluded that “the results of this study suggest that a brief stress management intervention in an urban adolescent medicine clinic is effective at decreasing subjective distress and improving heart rate variability coherence rates. Future studies should include a control group and longer-term follow-up.”
TORONTO – A brief stress management intervention left a lasting effect on adolescents referred for mental health–related complaints, significantly reducing perceived distress, and heart rate variability. The majority of recipients expressed interest in additional training at follow-up.
Elizabeth B. Mason, MD, and her colleagues from Rainbow Babies and Children’s Hospital in Cleveland, reported in a poster presentation at the Pediatric Academic Societies annual meeting.
The vast majority of participants (96.5%) were African American, mean age was 16 years, and 36% were male. Cutoff criteria for generalized anxiety on the SCARED was met by 35 (41%) participants, and 23 (27%) participants scored positive for depression on the PHQ-A.
Following completion of preintervention questionnaires, 50 of 86 (58%) participants received psychoeducation from an adolescent medicine fellow or a pediatric psychologist on the effect stress has on the body, training in diaphragmatic breathing and progressive muscle relaxation, and no-cost/low-cost exercise options. Study participants also engaged in a peripheral biofeedback program called Unyte that has been shown to improve heart rate variability. The remaining 36 participants received no training and served as controls.
Those in the intervention group had significantly lower SUDS scores postintervention than did the control group. Heart rate variability coherence rates also decreased significantly postintervention in those who received the intervention, compared with controls.
When reached by phone 1 week after the session, 92% of participants said they found the intervention helpful and felt more relaxed, and 44% expressed interest in additional relaxation training.
The investigators concluded that “the results of this study suggest that a brief stress management intervention in an urban adolescent medicine clinic is effective at decreasing subjective distress and improving heart rate variability coherence rates. Future studies should include a control group and longer-term follow-up.”
TORONTO – A brief stress management intervention left a lasting effect on adolescents referred for mental health–related complaints, significantly reducing perceived distress, and heart rate variability. The majority of recipients expressed interest in additional training at follow-up.
Elizabeth B. Mason, MD, and her colleagues from Rainbow Babies and Children’s Hospital in Cleveland, reported in a poster presentation at the Pediatric Academic Societies annual meeting.
The vast majority of participants (96.5%) were African American, mean age was 16 years, and 36% were male. Cutoff criteria for generalized anxiety on the SCARED was met by 35 (41%) participants, and 23 (27%) participants scored positive for depression on the PHQ-A.
Following completion of preintervention questionnaires, 50 of 86 (58%) participants received psychoeducation from an adolescent medicine fellow or a pediatric psychologist on the effect stress has on the body, training in diaphragmatic breathing and progressive muscle relaxation, and no-cost/low-cost exercise options. Study participants also engaged in a peripheral biofeedback program called Unyte that has been shown to improve heart rate variability. The remaining 36 participants received no training and served as controls.
Those in the intervention group had significantly lower SUDS scores postintervention than did the control group. Heart rate variability coherence rates also decreased significantly postintervention in those who received the intervention, compared with controls.
When reached by phone 1 week after the session, 92% of participants said they found the intervention helpful and felt more relaxed, and 44% expressed interest in additional relaxation training.
The investigators concluded that “the results of this study suggest that a brief stress management intervention in an urban adolescent medicine clinic is effective at decreasing subjective distress and improving heart rate variability coherence rates. Future studies should include a control group and longer-term follow-up.”
REPORTING FROM PAS 2018
Key clinical point: A brief stress management intervention had a lasting effect on adolescents referred for mental health–related complaints.
Major finding: Nearly all participants found the intervention helpful and felt more relaxed and 44% were interested in additional training.
Study details: A randomized trial including 86 adolescents (mean age, 16 years) referred for anxiety, somatic complaints, or difficulty managing mood.
Disclosures: The investigators reported no conflict of interest.
Simple tool improves inpatient influenza vaccination rates
TORONTO –
“When we looked at the immunization status of children in New York City, we found that one of the vaccines most commonly missed was influenza vaccine, especially from 2011 through 2014,” one of the study authors, Anmol Goyal, MD, of SUNY Downstate Medical Center, Brooklyn, N.Y., said in an interview at the Pediatric Academic Societies meeting.
“Given this year’s epidemic of influenza and the increasing deaths, we decided to look back on interventions we had done in the past to see if any can be reimplemented to help improve the vaccination status for these children,” he said. “The national goal is 80%, but if we look at the recent trend, even though we have been able to improve vaccination status, it is still below the national goal.” For example, he said, according to New York Department of Health data, the 2012-2013 influenza vaccination rates in New York City were 65% among children 6 months to 5 years old, 47% among those 5-8 years old, and 31% among those 9-18 years old, which were well below the national goal.
In an effort to improve influenza vaccine access, lead author Stephan Kohlhoff, MD, a pediatric infectious disease specialist at the medical center, and his associates, implemented a simple vaccine screening tool to use in the inpatient setting as an opportunity to improve vaccination rates among children in New York City. It consisted of nursing staff assessing the patient’s influenza immunization status on admission and conducting source verification using the citywide immunization registry, or with vaccine cards brought by parents or guardians during admission. Influenza vaccine was administered as a standing order before discharge, unless refused by the parents or guardians. The study population comprised 602 patients between the ages of 6 months and 21 years who were admitted to the inpatient unit during 2 months of the influenza season (November and December) from 2011 to 2013.
Dr. Goyal, a second-year pediatric resident at the medical center, reported that the influenza vaccination status on admission was positive in only 31% of children in 2011, 30% in 2012, and 34% in 2013. The vaccine screening tool was implemented in 64% of admitted children in 2012 and 70% in 2013. Following implementation, the researchers observed a 5% increase in immunization rates in 2012 and an 11% increase in 2013, with an overall increase of 8% over 2 years (P less than .001). He was quick to point out that the influenza rate could have been improved by an additional 22% had 77% of patients not refused vaccination.
“Unfortunately, as our primary objective was to assess the utility of our screening tool in improving inpatient immunization status, we had very limited data points toward refusal of vaccine,” Dr. Goyal said. “Some of the reasons for refusal that were gathered during screening included preferred vaccination by their primary care provider after discharge. Or, maybe they don’t want the vaccine because they feel that the vaccine will make their kids sick. We don’t have enough data to point to any particular reason. This study provides information on acceptance rate of inpatient immunization, which may be useful for implementing additional educational initiatives to overcome potential barriers and help us reach our national goal.”
The researchers reported having no financial disclosures.
TORONTO –
“When we looked at the immunization status of children in New York City, we found that one of the vaccines most commonly missed was influenza vaccine, especially from 2011 through 2014,” one of the study authors, Anmol Goyal, MD, of SUNY Downstate Medical Center, Brooklyn, N.Y., said in an interview at the Pediatric Academic Societies meeting.
“Given this year’s epidemic of influenza and the increasing deaths, we decided to look back on interventions we had done in the past to see if any can be reimplemented to help improve the vaccination status for these children,” he said. “The national goal is 80%, but if we look at the recent trend, even though we have been able to improve vaccination status, it is still below the national goal.” For example, he said, according to New York Department of Health data, the 2012-2013 influenza vaccination rates in New York City were 65% among children 6 months to 5 years old, 47% among those 5-8 years old, and 31% among those 9-18 years old, which were well below the national goal.
In an effort to improve influenza vaccine access, lead author Stephan Kohlhoff, MD, a pediatric infectious disease specialist at the medical center, and his associates, implemented a simple vaccine screening tool to use in the inpatient setting as an opportunity to improve vaccination rates among children in New York City. It consisted of nursing staff assessing the patient’s influenza immunization status on admission and conducting source verification using the citywide immunization registry, or with vaccine cards brought by parents or guardians during admission. Influenza vaccine was administered as a standing order before discharge, unless refused by the parents or guardians. The study population comprised 602 patients between the ages of 6 months and 21 years who were admitted to the inpatient unit during 2 months of the influenza season (November and December) from 2011 to 2013.
Dr. Goyal, a second-year pediatric resident at the medical center, reported that the influenza vaccination status on admission was positive in only 31% of children in 2011, 30% in 2012, and 34% in 2013. The vaccine screening tool was implemented in 64% of admitted children in 2012 and 70% in 2013. Following implementation, the researchers observed a 5% increase in immunization rates in 2012 and an 11% increase in 2013, with an overall increase of 8% over 2 years (P less than .001). He was quick to point out that the influenza rate could have been improved by an additional 22% had 77% of patients not refused vaccination.
“Unfortunately, as our primary objective was to assess the utility of our screening tool in improving inpatient immunization status, we had very limited data points toward refusal of vaccine,” Dr. Goyal said. “Some of the reasons for refusal that were gathered during screening included preferred vaccination by their primary care provider after discharge. Or, maybe they don’t want the vaccine because they feel that the vaccine will make their kids sick. We don’t have enough data to point to any particular reason. This study provides information on acceptance rate of inpatient immunization, which may be useful for implementing additional educational initiatives to overcome potential barriers and help us reach our national goal.”
The researchers reported having no financial disclosures.
TORONTO –
“When we looked at the immunization status of children in New York City, we found that one of the vaccines most commonly missed was influenza vaccine, especially from 2011 through 2014,” one of the study authors, Anmol Goyal, MD, of SUNY Downstate Medical Center, Brooklyn, N.Y., said in an interview at the Pediatric Academic Societies meeting.
“Given this year’s epidemic of influenza and the increasing deaths, we decided to look back on interventions we had done in the past to see if any can be reimplemented to help improve the vaccination status for these children,” he said. “The national goal is 80%, but if we look at the recent trend, even though we have been able to improve vaccination status, it is still below the national goal.” For example, he said, according to New York Department of Health data, the 2012-2013 influenza vaccination rates in New York City were 65% among children 6 months to 5 years old, 47% among those 5-8 years old, and 31% among those 9-18 years old, which were well below the national goal.
In an effort to improve influenza vaccine access, lead author Stephan Kohlhoff, MD, a pediatric infectious disease specialist at the medical center, and his associates, implemented a simple vaccine screening tool to use in the inpatient setting as an opportunity to improve vaccination rates among children in New York City. It consisted of nursing staff assessing the patient’s influenza immunization status on admission and conducting source verification using the citywide immunization registry, or with vaccine cards brought by parents or guardians during admission. Influenza vaccine was administered as a standing order before discharge, unless refused by the parents or guardians. The study population comprised 602 patients between the ages of 6 months and 21 years who were admitted to the inpatient unit during 2 months of the influenza season (November and December) from 2011 to 2013.
Dr. Goyal, a second-year pediatric resident at the medical center, reported that the influenza vaccination status on admission was positive in only 31% of children in 2011, 30% in 2012, and 34% in 2013. The vaccine screening tool was implemented in 64% of admitted children in 2012 and 70% in 2013. Following implementation, the researchers observed a 5% increase in immunization rates in 2012 and an 11% increase in 2013, with an overall increase of 8% over 2 years (P less than .001). He was quick to point out that the influenza rate could have been improved by an additional 22% had 77% of patients not refused vaccination.
“Unfortunately, as our primary objective was to assess the utility of our screening tool in improving inpatient immunization status, we had very limited data points toward refusal of vaccine,” Dr. Goyal said. “Some of the reasons for refusal that were gathered during screening included preferred vaccination by their primary care provider after discharge. Or, maybe they don’t want the vaccine because they feel that the vaccine will make their kids sick. We don’t have enough data to point to any particular reason. This study provides information on acceptance rate of inpatient immunization, which may be useful for implementing additional educational initiatives to overcome potential barriers and help us reach our national goal.”
The researchers reported having no financial disclosures.
AT PAS 18
Key clinical point: The inpatient setting can be used to successfully improve influenza vaccine rates.
Major finding: Following implementation of a simple inpatient vaccine screening tool, a 5% increase in immunization rates occurred in 2012 and an 11% increase occurred in 2013.
Study details: A review of 602 patients between the ages of 6 months and 21 years who were admitted to the inpatient unit during 2 months of the influenza season (November and December) from 2011 to 2013.
Disclosures: The researchers reported having no financial disclosures.
Patient adjustments needed for closed-loop insulin delivery
TORONTO – Closed-loop insulin delivery is expected to become the standard of care in type 1 diabetes mellitus (T1DM), but there are multiple barriers that patients need to overcome.
“Many people who are potentially going to be using closed-loop systems are enthusiastic but have unrealistic expectations of how the systems are going to perform, and there are many barriers to uptake and optimal use that we still haven’t quite figured out,” said Korey K. Hood, PhD, a professor in the departments of pediatrics and psychiatry & behavioral sciences at Stanford (Calif.) University.
In a session dedicated to all aspects of closed-loop automated insulin delivery at the Pediatric Academic Societies annual meeting, Dr. Hood offered comments on patient and family factors important to the uptake and use of closed-loop technologies. His research at Stanford is focused on understanding the psychosocial aspects of diabetes management and how these factors contribute to disease outcomes.
Closed-loop insulin delivery refers to technologies that combine automated glucose monitoring (AGM) with an algorithm to determine insulin needs and an insulin delivery device. Sometimes called an “artificial pancreas” or “bionic pancreas,” closed-loop insulin delivery is considered a significant advance in the management of T1DM, relegating daily finger sticks and nighttime hypoglycemia to things of the past.
In a recent meta-analysis of randomized clinical trials, use of any automated device added nearly 2.5 hours of time in near normoglycemia over 24 hours in patients with TIDM, compared with any other type of insulin-based treatment (BMJ. 2018. doi: 10.1136/bmj.k1310). The benefit was primarily based on better glucose control in the overnight period.
In September 2016, the Food and Drug Administration approved the MiniMed 670G Insulin Pump System (Medtronic), the first hybrid automated insulin delivery device for T1DM and the only one approved in the United States. The system is intended for subcutaneous continuous glucose monitoring (CGM) and continuous delivery of basal insulin and administration of insulin for the management of T1DM in persons 14 years of age and older.
Barriers from different perspectives
Barriers to uptake and use are common for the devices that are components of closed-loop systems. In a survey of 1,503 adults with TIDM, Dr. Hood’s group found a wide range of barriers to adoption of CGM or insulin pumps that could potentially also impact use of closed-loop systems (Diabetes Care. 2017;40:181-7). Some were nonmodifiable, like costs, but most were modifiable.
“Many people talk about the hassle of wearing devices. They don’t like having multiple devices on their bodies. They don’t always like the way that they look, and so these are things that we can have some kind of impact on and need to be paying attention to,” he said.
“The younger participants indicated a lot more barriers to using devices, and as they got older, they indicated fewer barriers. But what was also interesting is that the younger participants also indicated a lot more diabetes distress. As time went on, that was less of a factor in whether or not people were using diabetes devices,” reported Dr. Hood.
Not surprisingly, he added, was that younger participants had more favorable views of technology in general. “But they had less favorable views of diabetes technology [than older participants], so they’re really not crazy about using these devices.”
Dr. Hood’s group has also studied whether patient-reported barriers to CGM use align with what clinicians perceive to be patient-related barriers (Diabetes Sci Technol. 2017;11[3]484-92). Similar to the patients, clinicians most frequently endorsed the perception that patients dislike having the device on their body. However, other things they felt their patients worried about were the alarms on the device and the difficulty in understanding its features, neither of which patients considered a primary barrier to CGM or insulin pump adoption.
“So, we need to be cautious and mindful as we move forward that there are mismatches between the patient-reported and clinician-reported barriers,” said Dr. Hood. “Our response, often, is to teach and to provide some kind of education, when that’s not necessarily what the patient is asking for.”
Would you use it?
In 2017, a group of investigators conducted a qualitative study of 284 participants, ranging in age from 8 to 86 years, with T1DM. The researchers used structured interviews or focus groups to explore expectations, desired features, potential benefits, and perceived burdens of automated insulin delivery systems (Diabetes Care. 2017;40[11]:1453-61).
“We were interested in children, adolescents, and adults with type 1, and then also the partners of the adults and the parents of the youth,” he explained.
“The findings revealed three themes identified as pressing for the uptake of automated insulin delivery: considerations of trust and control, system features, and concerns and barriers to adoption.
“For children, the areas of most concern revolved around specific social situations. Adolescents, on the other hand, were more concerned about the physical features of the device, the wearability, the discreetness of using it, and the comfort,” said Dr. Hood.
Adults and parents were much more interested in device accuracy, safety, adaptability, and algorithm quality. “For the kids and teens, not surprisingly, this wasn’t high on their list,” he added.
A clear indication of the unrealistic expectations surrounding this technology came from a 2018 study of almost 200 family members, which found that “reducing the constant concerns about diabetes, relieving family stress, and improving overall family relationships” were the three major areas the participants hoped would be helped with automated insulin delivery (Diabetes Technol Ther. 2018;20[3]:222-8).
“If we come up with a device that does this, then I think we will have fixed everything!” Dr. Hood said, adding that it really highlights the “very high hopes and expectations” of what closed-loop systems should deliver.
Device readiness is another area researchers have studied in the run-up to closed-loop systems. “The idea that everybody’s going to be ready to start in the same way, I think, is going to set us up for some failures,” he noted, adding that, in general, parents are much more enthusiastic about this than pediatric patients.
“Individuals who had been using diabetes devices – and some had already been in closed-loop studies – They had more realistic expectations of what these systems are going to be, because they knew that it wasn’t going to be a complete fix. Whereas others with more limited experience with component devices and these systems had much higher expectations and reported a fair amount of dissatisfaction at the end of the study because it didn’t do everything that they wanted it to do.”
Waves of uptake
“Ultimately, a closed-loop system is going to be judged by whether it can increase time on target and reduce cognitive burden,” said Dr. Hood. He finished his talk with some projections about the future of closed-loop systems. “I think we’re going to probably to have different waves, or types, of closed-loop users.”
The first wave will be the group that’s already “sold” on the idea, which might encompass about 15% of patients. The second wave, which might represent about 30% of the relevant patient population, will be those who are sold on the idea and will likely use it but will have high expectations of the system’s ease of use and effectiveness and thus are highly likely to discontinue its use if those expectations are not met.
“The third wave will be those who might use a closed-loop system but might be unaware of them currently and will need a fair amount of education.” And, finally, the fourth group are unlikely to ever use closed-loop insulin delivery. “They are a group that feels burned by previous generations of systems, and I think that they may not perceive benefit,” Dr. Hood suggested.
“But all of this is to say that I do think that a tailored experience, and one that is focused on different profiles, can optimize both the uptake and the use of these systems.”
Dr. Hood reported receiving grant/research support from Dexcom and being a consultant for Lilly Innovation Center, J&J Diabetes Institute, and Bigfoot Biomedical.
TORONTO – Closed-loop insulin delivery is expected to become the standard of care in type 1 diabetes mellitus (T1DM), but there are multiple barriers that patients need to overcome.
“Many people who are potentially going to be using closed-loop systems are enthusiastic but have unrealistic expectations of how the systems are going to perform, and there are many barriers to uptake and optimal use that we still haven’t quite figured out,” said Korey K. Hood, PhD, a professor in the departments of pediatrics and psychiatry & behavioral sciences at Stanford (Calif.) University.
In a session dedicated to all aspects of closed-loop automated insulin delivery at the Pediatric Academic Societies annual meeting, Dr. Hood offered comments on patient and family factors important to the uptake and use of closed-loop technologies. His research at Stanford is focused on understanding the psychosocial aspects of diabetes management and how these factors contribute to disease outcomes.
Closed-loop insulin delivery refers to technologies that combine automated glucose monitoring (AGM) with an algorithm to determine insulin needs and an insulin delivery device. Sometimes called an “artificial pancreas” or “bionic pancreas,” closed-loop insulin delivery is considered a significant advance in the management of T1DM, relegating daily finger sticks and nighttime hypoglycemia to things of the past.
In a recent meta-analysis of randomized clinical trials, use of any automated device added nearly 2.5 hours of time in near normoglycemia over 24 hours in patients with TIDM, compared with any other type of insulin-based treatment (BMJ. 2018. doi: 10.1136/bmj.k1310). The benefit was primarily based on better glucose control in the overnight period.
In September 2016, the Food and Drug Administration approved the MiniMed 670G Insulin Pump System (Medtronic), the first hybrid automated insulin delivery device for T1DM and the only one approved in the United States. The system is intended for subcutaneous continuous glucose monitoring (CGM) and continuous delivery of basal insulin and administration of insulin for the management of T1DM in persons 14 years of age and older.
Barriers from different perspectives
Barriers to uptake and use are common for the devices that are components of closed-loop systems. In a survey of 1,503 adults with TIDM, Dr. Hood’s group found a wide range of barriers to adoption of CGM or insulin pumps that could potentially also impact use of closed-loop systems (Diabetes Care. 2017;40:181-7). Some were nonmodifiable, like costs, but most were modifiable.
“Many people talk about the hassle of wearing devices. They don’t like having multiple devices on their bodies. They don’t always like the way that they look, and so these are things that we can have some kind of impact on and need to be paying attention to,” he said.
“The younger participants indicated a lot more barriers to using devices, and as they got older, they indicated fewer barriers. But what was also interesting is that the younger participants also indicated a lot more diabetes distress. As time went on, that was less of a factor in whether or not people were using diabetes devices,” reported Dr. Hood.
Not surprisingly, he added, was that younger participants had more favorable views of technology in general. “But they had less favorable views of diabetes technology [than older participants], so they’re really not crazy about using these devices.”
Dr. Hood’s group has also studied whether patient-reported barriers to CGM use align with what clinicians perceive to be patient-related barriers (Diabetes Sci Technol. 2017;11[3]484-92). Similar to the patients, clinicians most frequently endorsed the perception that patients dislike having the device on their body. However, other things they felt their patients worried about were the alarms on the device and the difficulty in understanding its features, neither of which patients considered a primary barrier to CGM or insulin pump adoption.
“So, we need to be cautious and mindful as we move forward that there are mismatches between the patient-reported and clinician-reported barriers,” said Dr. Hood. “Our response, often, is to teach and to provide some kind of education, when that’s not necessarily what the patient is asking for.”
Would you use it?
In 2017, a group of investigators conducted a qualitative study of 284 participants, ranging in age from 8 to 86 years, with T1DM. The researchers used structured interviews or focus groups to explore expectations, desired features, potential benefits, and perceived burdens of automated insulin delivery systems (Diabetes Care. 2017;40[11]:1453-61).
“We were interested in children, adolescents, and adults with type 1, and then also the partners of the adults and the parents of the youth,” he explained.
“The findings revealed three themes identified as pressing for the uptake of automated insulin delivery: considerations of trust and control, system features, and concerns and barriers to adoption.
“For children, the areas of most concern revolved around specific social situations. Adolescents, on the other hand, were more concerned about the physical features of the device, the wearability, the discreetness of using it, and the comfort,” said Dr. Hood.
Adults and parents were much more interested in device accuracy, safety, adaptability, and algorithm quality. “For the kids and teens, not surprisingly, this wasn’t high on their list,” he added.
A clear indication of the unrealistic expectations surrounding this technology came from a 2018 study of almost 200 family members, which found that “reducing the constant concerns about diabetes, relieving family stress, and improving overall family relationships” were the three major areas the participants hoped would be helped with automated insulin delivery (Diabetes Technol Ther. 2018;20[3]:222-8).
“If we come up with a device that does this, then I think we will have fixed everything!” Dr. Hood said, adding that it really highlights the “very high hopes and expectations” of what closed-loop systems should deliver.
Device readiness is another area researchers have studied in the run-up to closed-loop systems. “The idea that everybody’s going to be ready to start in the same way, I think, is going to set us up for some failures,” he noted, adding that, in general, parents are much more enthusiastic about this than pediatric patients.
“Individuals who had been using diabetes devices – and some had already been in closed-loop studies – They had more realistic expectations of what these systems are going to be, because they knew that it wasn’t going to be a complete fix. Whereas others with more limited experience with component devices and these systems had much higher expectations and reported a fair amount of dissatisfaction at the end of the study because it didn’t do everything that they wanted it to do.”
Waves of uptake
“Ultimately, a closed-loop system is going to be judged by whether it can increase time on target and reduce cognitive burden,” said Dr. Hood. He finished his talk with some projections about the future of closed-loop systems. “I think we’re going to probably to have different waves, or types, of closed-loop users.”
The first wave will be the group that’s already “sold” on the idea, which might encompass about 15% of patients. The second wave, which might represent about 30% of the relevant patient population, will be those who are sold on the idea and will likely use it but will have high expectations of the system’s ease of use and effectiveness and thus are highly likely to discontinue its use if those expectations are not met.
“The third wave will be those who might use a closed-loop system but might be unaware of them currently and will need a fair amount of education.” And, finally, the fourth group are unlikely to ever use closed-loop insulin delivery. “They are a group that feels burned by previous generations of systems, and I think that they may not perceive benefit,” Dr. Hood suggested.
“But all of this is to say that I do think that a tailored experience, and one that is focused on different profiles, can optimize both the uptake and the use of these systems.”
Dr. Hood reported receiving grant/research support from Dexcom and being a consultant for Lilly Innovation Center, J&J Diabetes Institute, and Bigfoot Biomedical.
TORONTO – Closed-loop insulin delivery is expected to become the standard of care in type 1 diabetes mellitus (T1DM), but there are multiple barriers that patients need to overcome.
“Many people who are potentially going to be using closed-loop systems are enthusiastic but have unrealistic expectations of how the systems are going to perform, and there are many barriers to uptake and optimal use that we still haven’t quite figured out,” said Korey K. Hood, PhD, a professor in the departments of pediatrics and psychiatry & behavioral sciences at Stanford (Calif.) University.
In a session dedicated to all aspects of closed-loop automated insulin delivery at the Pediatric Academic Societies annual meeting, Dr. Hood offered comments on patient and family factors important to the uptake and use of closed-loop technologies. His research at Stanford is focused on understanding the psychosocial aspects of diabetes management and how these factors contribute to disease outcomes.
Closed-loop insulin delivery refers to technologies that combine automated glucose monitoring (AGM) with an algorithm to determine insulin needs and an insulin delivery device. Sometimes called an “artificial pancreas” or “bionic pancreas,” closed-loop insulin delivery is considered a significant advance in the management of T1DM, relegating daily finger sticks and nighttime hypoglycemia to things of the past.
In a recent meta-analysis of randomized clinical trials, use of any automated device added nearly 2.5 hours of time in near normoglycemia over 24 hours in patients with TIDM, compared with any other type of insulin-based treatment (BMJ. 2018. doi: 10.1136/bmj.k1310). The benefit was primarily based on better glucose control in the overnight period.
In September 2016, the Food and Drug Administration approved the MiniMed 670G Insulin Pump System (Medtronic), the first hybrid automated insulin delivery device for T1DM and the only one approved in the United States. The system is intended for subcutaneous continuous glucose monitoring (CGM) and continuous delivery of basal insulin and administration of insulin for the management of T1DM in persons 14 years of age and older.
Barriers from different perspectives
Barriers to uptake and use are common for the devices that are components of closed-loop systems. In a survey of 1,503 adults with TIDM, Dr. Hood’s group found a wide range of barriers to adoption of CGM or insulin pumps that could potentially also impact use of closed-loop systems (Diabetes Care. 2017;40:181-7). Some were nonmodifiable, like costs, but most were modifiable.
“Many people talk about the hassle of wearing devices. They don’t like having multiple devices on their bodies. They don’t always like the way that they look, and so these are things that we can have some kind of impact on and need to be paying attention to,” he said.
“The younger participants indicated a lot more barriers to using devices, and as they got older, they indicated fewer barriers. But what was also interesting is that the younger participants also indicated a lot more diabetes distress. As time went on, that was less of a factor in whether or not people were using diabetes devices,” reported Dr. Hood.
Not surprisingly, he added, was that younger participants had more favorable views of technology in general. “But they had less favorable views of diabetes technology [than older participants], so they’re really not crazy about using these devices.”
Dr. Hood’s group has also studied whether patient-reported barriers to CGM use align with what clinicians perceive to be patient-related barriers (Diabetes Sci Technol. 2017;11[3]484-92). Similar to the patients, clinicians most frequently endorsed the perception that patients dislike having the device on their body. However, other things they felt their patients worried about were the alarms on the device and the difficulty in understanding its features, neither of which patients considered a primary barrier to CGM or insulin pump adoption.
“So, we need to be cautious and mindful as we move forward that there are mismatches between the patient-reported and clinician-reported barriers,” said Dr. Hood. “Our response, often, is to teach and to provide some kind of education, when that’s not necessarily what the patient is asking for.”
Would you use it?
In 2017, a group of investigators conducted a qualitative study of 284 participants, ranging in age from 8 to 86 years, with T1DM. The researchers used structured interviews or focus groups to explore expectations, desired features, potential benefits, and perceived burdens of automated insulin delivery systems (Diabetes Care. 2017;40[11]:1453-61).
“We were interested in children, adolescents, and adults with type 1, and then also the partners of the adults and the parents of the youth,” he explained.
“The findings revealed three themes identified as pressing for the uptake of automated insulin delivery: considerations of trust and control, system features, and concerns and barriers to adoption.
“For children, the areas of most concern revolved around specific social situations. Adolescents, on the other hand, were more concerned about the physical features of the device, the wearability, the discreetness of using it, and the comfort,” said Dr. Hood.
Adults and parents were much more interested in device accuracy, safety, adaptability, and algorithm quality. “For the kids and teens, not surprisingly, this wasn’t high on their list,” he added.
A clear indication of the unrealistic expectations surrounding this technology came from a 2018 study of almost 200 family members, which found that “reducing the constant concerns about diabetes, relieving family stress, and improving overall family relationships” were the three major areas the participants hoped would be helped with automated insulin delivery (Diabetes Technol Ther. 2018;20[3]:222-8).
“If we come up with a device that does this, then I think we will have fixed everything!” Dr. Hood said, adding that it really highlights the “very high hopes and expectations” of what closed-loop systems should deliver.
Device readiness is another area researchers have studied in the run-up to closed-loop systems. “The idea that everybody’s going to be ready to start in the same way, I think, is going to set us up for some failures,” he noted, adding that, in general, parents are much more enthusiastic about this than pediatric patients.
“Individuals who had been using diabetes devices – and some had already been in closed-loop studies – They had more realistic expectations of what these systems are going to be, because they knew that it wasn’t going to be a complete fix. Whereas others with more limited experience with component devices and these systems had much higher expectations and reported a fair amount of dissatisfaction at the end of the study because it didn’t do everything that they wanted it to do.”
Waves of uptake
“Ultimately, a closed-loop system is going to be judged by whether it can increase time on target and reduce cognitive burden,” said Dr. Hood. He finished his talk with some projections about the future of closed-loop systems. “I think we’re going to probably to have different waves, or types, of closed-loop users.”
The first wave will be the group that’s already “sold” on the idea, which might encompass about 15% of patients. The second wave, which might represent about 30% of the relevant patient population, will be those who are sold on the idea and will likely use it but will have high expectations of the system’s ease of use and effectiveness and thus are highly likely to discontinue its use if those expectations are not met.
“The third wave will be those who might use a closed-loop system but might be unaware of them currently and will need a fair amount of education.” And, finally, the fourth group are unlikely to ever use closed-loop insulin delivery. “They are a group that feels burned by previous generations of systems, and I think that they may not perceive benefit,” Dr. Hood suggested.
“But all of this is to say that I do think that a tailored experience, and one that is focused on different profiles, can optimize both the uptake and the use of these systems.”
Dr. Hood reported receiving grant/research support from Dexcom and being a consultant for Lilly Innovation Center, J&J Diabetes Institute, and Bigfoot Biomedical.
EXPERT ANALYSIS FROM PAS 2018
HBV birth dose predicts vaccine adherence
TORONTO – Infants who do not receive the hepatitis B vaccine birth dose are less likely to be up-to-date recipients of recommended vaccines by 19 months, based on results from a retrospective study of more than 9,000 infants.
“As pediatricians, we should be mindful of that when we are meeting families after the birth hospitalization and start a conversation at that point around vaccines,” one of the study authors, Annika M. Hofstetter, MD, PhD, said in an interview at the Pediatric Academic Societies meeting.
Of the 9,080 infants, 51% were male, 49% were non-Hispanic white, 56% were covered by public health insurance, and 47% stayed in the hospital for 48 hours or longer. The researchers reported that 76% infants received the HBV during the birth hospitalization, and 54% of subjects completed the seven-vaccine series by age 19 months. They also found that (P less than .001). Infants who received the HBV birth dose were 2.9 times more likely to complete the seven-vaccine series by age 19 months, compared with those who did not receive the HBV birth dose.
“Parents are making their first vaccine decision during that birth hospitalization,” said Dr. Hofstetter, who also conducts immunization research studies at Seattle Children’s Research Institute. “It’s unclear what underlies this decision, such as specific parent concerns or the way in which we as providers in the hospital are communicating vaccine information to the families. It’s telling, and it will be interesting to further explore the factors that are determining whether a family gets the vaccine during the birth hospitalization or not, and how we as a pediatric community can start having effective vaccine conversations earlier.”
She acknowledged certain limitations of the study, including the potential for misclassification errors in vaccine reporting systems and the fact that no data were available on parental attitudes about vaccination. The researchers reported having no financial disclosures.
TORONTO – Infants who do not receive the hepatitis B vaccine birth dose are less likely to be up-to-date recipients of recommended vaccines by 19 months, based on results from a retrospective study of more than 9,000 infants.
“As pediatricians, we should be mindful of that when we are meeting families after the birth hospitalization and start a conversation at that point around vaccines,” one of the study authors, Annika M. Hofstetter, MD, PhD, said in an interview at the Pediatric Academic Societies meeting.
Of the 9,080 infants, 51% were male, 49% were non-Hispanic white, 56% were covered by public health insurance, and 47% stayed in the hospital for 48 hours or longer. The researchers reported that 76% infants received the HBV during the birth hospitalization, and 54% of subjects completed the seven-vaccine series by age 19 months. They also found that (P less than .001). Infants who received the HBV birth dose were 2.9 times more likely to complete the seven-vaccine series by age 19 months, compared with those who did not receive the HBV birth dose.
“Parents are making their first vaccine decision during that birth hospitalization,” said Dr. Hofstetter, who also conducts immunization research studies at Seattle Children’s Research Institute. “It’s unclear what underlies this decision, such as specific parent concerns or the way in which we as providers in the hospital are communicating vaccine information to the families. It’s telling, and it will be interesting to further explore the factors that are determining whether a family gets the vaccine during the birth hospitalization or not, and how we as a pediatric community can start having effective vaccine conversations earlier.”
She acknowledged certain limitations of the study, including the potential for misclassification errors in vaccine reporting systems and the fact that no data were available on parental attitudes about vaccination. The researchers reported having no financial disclosures.
TORONTO – Infants who do not receive the hepatitis B vaccine birth dose are less likely to be up-to-date recipients of recommended vaccines by 19 months, based on results from a retrospective study of more than 9,000 infants.
“As pediatricians, we should be mindful of that when we are meeting families after the birth hospitalization and start a conversation at that point around vaccines,” one of the study authors, Annika M. Hofstetter, MD, PhD, said in an interview at the Pediatric Academic Societies meeting.
Of the 9,080 infants, 51% were male, 49% were non-Hispanic white, 56% were covered by public health insurance, and 47% stayed in the hospital for 48 hours or longer. The researchers reported that 76% infants received the HBV during the birth hospitalization, and 54% of subjects completed the seven-vaccine series by age 19 months. They also found that (P less than .001). Infants who received the HBV birth dose were 2.9 times more likely to complete the seven-vaccine series by age 19 months, compared with those who did not receive the HBV birth dose.
“Parents are making their first vaccine decision during that birth hospitalization,” said Dr. Hofstetter, who also conducts immunization research studies at Seattle Children’s Research Institute. “It’s unclear what underlies this decision, such as specific parent concerns or the way in which we as providers in the hospital are communicating vaccine information to the families. It’s telling, and it will be interesting to further explore the factors that are determining whether a family gets the vaccine during the birth hospitalization or not, and how we as a pediatric community can start having effective vaccine conversations earlier.”
She acknowledged certain limitations of the study, including the potential for misclassification errors in vaccine reporting systems and the fact that no data were available on parental attitudes about vaccination. The researchers reported having no financial disclosures.
AT PAS 2018
Key clinical point: Likelihood of completing the 7-vaccine series at 19 months was higher among infants who received the HBV birth dose.
Major finding: Infants who received the HBV birth dose were 2.9 times more likely to complete the 7-vaccine series by age 19 months, compared with those who did not receive the HBV birth dose.
Study details: A retrospective review of 9,080 infants born weighing at least 2,000 grams who received hospitalization care between January 1, 2008 and December 31, 2013.
Disclosures: The researchers reported having no financial disclosures.